WO2011085575A1 - Ortho-heterocyclyl formanilide compounds, their synthesis methods and use - Google Patents
Ortho-heterocyclyl formanilide compounds, their synthesis methods and use Download PDFInfo
- Publication number
- WO2011085575A1 WO2011085575A1 PCT/CN2010/073303 CN2010073303W WO2011085575A1 WO 2011085575 A1 WO2011085575 A1 WO 2011085575A1 CN 2010073303 W CN2010073303 W CN 2010073303W WO 2011085575 A1 WO2011085575 A1 WO 2011085575A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- compound
- chloro
- synthesis
- bromo
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 Cc(cc(*)cc1C(*2)=NN=C2N)c1NC(c1cc(Br)n[n]1-c1ccccc1Cl)=O Chemical compound Cc(cc(*)cc1C(*2)=NN=C2N)c1NC(c1cc(Br)n[n]1-c1ccccc1Cl)=O 0.000 description 2
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/82—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with three ring hetero atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
Definitions
- the invention belongs to the field of pesticides, and particularly relates to an o-heterocyclic anilide compound which can be used as an insecticide and a preparation method thereof.
- the prevention and control of pests has always been the core field of pesticide scientific research.
- the widespread use of pesticides has led to the effective management of most pests.
- the problem of drug resistance caused by the use of organic synthetic insecticides has become more prominent. Therefore, low toxicity compounds acting on new targets have become an inevitable trend in pesticide research.
- the object of the present invention is to provide a class of o-heterocyclic anilide compounds which have excellent control effects against various pests and are characterized by high efficiency, safety and good environmental compatibility, so as to meet crop protection and high-efficiency and safe insecticides. Demand.
- Another object of the present invention is to provide a process for the preparation of the above compounds.
- Still another object of the invention is to provide the use of the above compounds in the preparation of insecticides.
- the object of the invention can be achieved by the following measures:
- 1 or 2 are independently hydrogen, halogen or cyano;
- Z is oxygen or sulfur;
- Ri or R 2 are each independently a C1 to C5 fluorenyl group, a C1 to C5 halogenated fluorenyl group, a hydroxyl group, a C1 to C5 decyloxy group, a Cl ⁇ C5 halogenated fluorenyloxy group, a fluorenyl group, a C1 to C5 thiol group, an amino group.
- C1 ⁇ C5 ⁇ amino group including hydrazine, hydrazine-diamino group), C1 ⁇ C5 halogenated hydrazine amino group, C1 ⁇ C5 oxiranylamino group, C1 ⁇ C5 decyl sulfonamide group, C1 ⁇ C5 acyl group, C1 ⁇ C5 Amido, halogen, cyano or thiocyano.
- the X or X 2 in the present invention are each independently preferably H, F, CI, Br or CN; X, or X 2 are each independently further preferably H, F, CI or Br; wherein 1 is most preferably CI, Br or CN, X 2 is most preferably CI, Br or CN.
- R 2 each independently preferably is C1 to C5 fluorenyl, hydroxy, C1 to C5 decyloxy, fluorenyl, C1 to C5 thiol, amino, C1 to C5 decylamino, C1 to C5 decyl sulfonate.
- An amide group, a C1 to C5 amide group, a halogen, a cyano group or a thiocyano group; and C1 to C5 in each of the above substituents may further preferably be C1 to C3. Specifically speaking!
- R 2 may be independently selected from the group consisting of: -CH 3 , -C 2 H 5 , -CH(CH 3 ) 2 , -OH, -OCH 3 , -OC 2 H 5 , -OCH (CH 3 2 , -SH, -SCH 3 , -SC 2 H 5 , -SCH(CH 3 ) 2 , -NH 2 , -NHCH 3 , -NHC 2 H 5 , -N(CH 3 ) 2 , -NHCH(CH 3 ) 2 , -NHOCH 3 -NHOC 2 H 5 , -NHOCH(CH 3 ) 2 , -N(CH 3 ) 2 -NHCF 3 -NHCHF 2 -NHCH 2 F, -NHCOCH 3 , -NHCOC 2 H 5 , - NHS0 2 CH 3 , -NHS0 2 C 2 H 5 , halogen, cyano or thiocyano group.
- Ri is a C1 to C5 fluorenyl group, a hydroxy group, a decyl group, a C1 to C5 thiol group, an amino group, a C1 to C5 fluorenylamino group, a C1 to C5 decyl sulfonylamino group, a C1 to C5 amide group, a halogen, a cyano group or a sulfur.
- the cyano group is most preferably a C1 to C3 fluorenyl group, a hydroxy group, a decyl group, a C1 to C3 thiol group, an amino group, a C1 to C3 fluorenylamino group, a C1 to C3 fluorenyl sulfonamide group, a Cl ⁇ C3 amide group, a halogen (preferably C1). Or Br), cyano or thiocyano.
- R 2 is further preferably a C1 to C5 mercapto group, an amino group, a C1 to C5 mercaptoamino group, a C1 to C5 mercaptosulfonylamino group, a Cl ⁇ C5 amido group, a halogen, a cyano group or a thiocyano group, and most preferably a C1 to C3 group.
- Base amino group, C1 ⁇ C3 oxime amino group, Cl ⁇ C3 decyl sulfonamide group, C1 ⁇ C3 amide group, halogen (preferably C1 or Br), cyano group or thiocyano group.
- Z is oxygen, 1 ⁇ is. 1 ⁇ . 5 thiol, amino, halogen, cyano or thiocyano group, when X is under neutral conditions, in a polar aprotic solvent, the intermediate M1 is reacted with the corresponding hydrazide reagent R ONHN ⁇ at room temperature Ring opening gives a compound of the formula (I 1 ), the reaction route of which is:
- Ri is a C1 to C5 halogenated fluorenyl group or a C1 to C5 acyl group
- it can also be produced according to the above formula.
- the compound II which is an amino group and a thiolation reagent, hydrazine It can be prepared by reacting an oxidizing reagent, a mercaptosulfonylating reagent or an acylating reagent.
- a compound which is a halogen can be prepared by reacting with sodium thiocyanate.
- the compound of the formula 12 can be reacted with a thiolation reagent.
- Ri is a C1 to C5 thiol group
- a compound of the formula 13 can be reacted with a thiolation reagent to prepare.
- intermediate M2 is reacted with carbon disulfide or halogenated hydrocarbon under basic conditions in a polar protic solvent at reflux temperature to obtain intermediate M3, and organic weak acid is used as a catalyst in the solvent.
- the M3 reaction is dehydrated and condensed to obtain a compound represented by the formula (I4); or the intermediate M1 is reacted with the corresponding reagent NH2NHCS to obtain M3, and then the organic weak acid is used as a catalyst in the solvent, and the M3 reaction is dehydrated and condensed to obtain a ring.
- a compound of the formula (I 4), the reaction route is:
- the preparation of the compound of formula I when Z is S can be prepared according to the above formula, and can be directly used when it is an anthracenyl group or a halogenated fluorenyl group.
- M2-M3-I4 when R is an amino group or other group (such as hydroxy, thiol, cyano), the M1-M3-I4 pathway can also be used.
- R is an amino group or other group (such as hydroxy, thiol, cyano)
- the M1-M3-I4 pathway can also be used.
- 1 ⁇ . 5 methoxy, C1 ⁇ C5 halogenated methoxy, C1 ⁇ C5 ⁇ amino, C1 ⁇ C5 halogenated guanidine amino group, C1 ⁇ C5 oxiranylamino group, C1 ⁇ C5 decyl sulfonamide group or C1 ⁇ C5 acyl group It can be prepared by further reaction with 14 using the corresponding guanidylation, sulfonamidation or acylation reagent.
- it is a thiocyano group a compound which is a halogen is reacted with sodium thiocyanate to prepare.
- the reaction route is:
- R 2 is as defined above, and is preferably a C1 to C5 fluorenyl group, a C1 to C5 halogenated fluorenyl group, a hydroxyl group, a decyl group, an amino group, a Cl ⁇ C5 fluorenylamino group, a C1 to C5 decyl sulfonylamino group or a C1 to C5 acyl group. Halogen, cyano or thiocyano.
- R 2 is a Cl ⁇ C5 oxime amino group, a C1 ⁇ C5 decyl sulfonamide group or a C1 ⁇ C5 acyl group
- the compound prepared by the above formula may be further used with the corresponding thiolation, sulfonamide or acylation reagent. Reaction preparation.
- R 2 is a thiocyano group
- a compound in which R 2 is a halogen is reacted with sodium thiocyanate to prepare.
- the acidic condition or the weak acid described in the present invention is an organic acid such as methanesulfonic acid or p-toluenesulfonic acid;
- the basic condition is a strong base or a weak base, and the strong base is sodium hydroxide, potassium hydroxide, sodium alkoxide or the like;
- the weak base is pyridine, triethylamine, etc.;
- the polar protic solvent is a solvent such as an alcohol, toluene or benzene; and the polar aprotic solvent is a solvent such as DMF.
- the solvent mentioned separately in the present invention may generally be a polar protic solvent or a polar aprotic solvent.
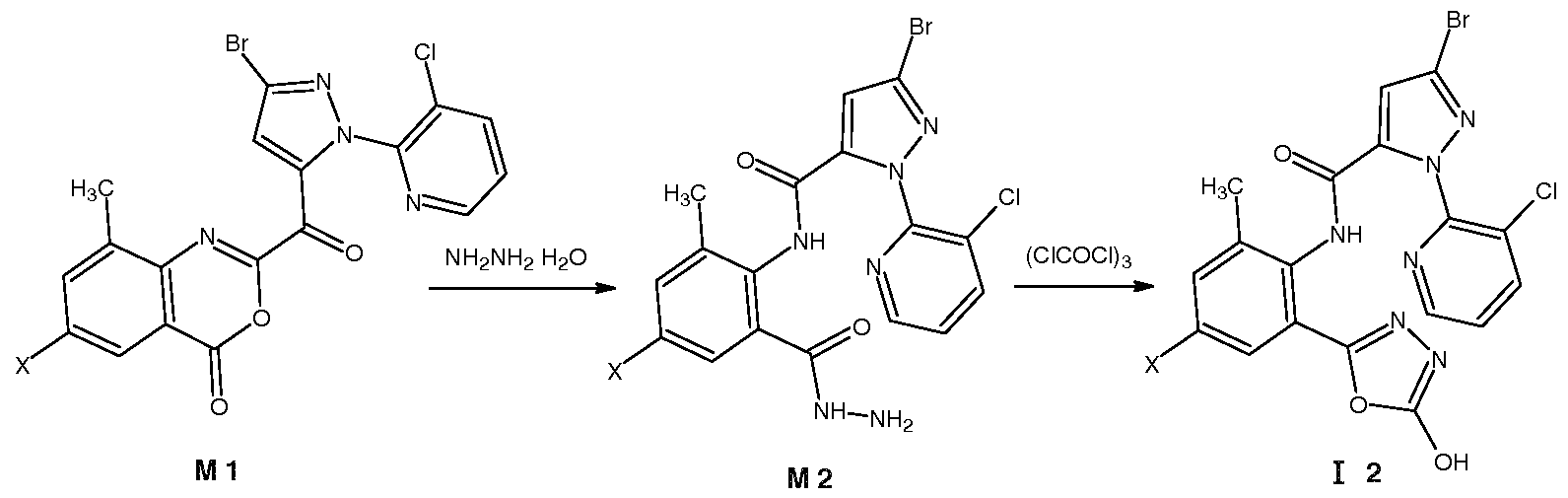
- Intermediate M2 is prepared by reacting raw material (Ml) with hydrazine hydrate at a temperature of -10 ⁇ 30 °C (refer to patent CN 200910033297),
- insects have excellent control activity against insects, and thus the compounds of the present invention can be used as an insecticide to protect plants such as agriculture and horticulture.
- the insects are lepidopteran pests such as cotton bollworm, beet armyworm, diamondback moth, cabbage caterpillar, rice leaf roller and rice stem borer, and homopteran pests such as spider mites, planthoppers, locusts, whiteflies, etc. , Diptera pests such as housefly, leaf miner, mosquito, etc., Orthoptera and coleoptera and other pests.
- the pests to which the compounds of the present invention are controll are not limited to the scope of the above examples.
- the compound of the present invention represented by the general formulae (I) and (?) when used as an insecticide in the fields of agriculture, horticulture, etc., it may be used alone or in the form of a pesticidal composition, such as by formula (I) Or the formula ( ⁇ ) is an active ingredient, and a pesticide adjuvant commonly used in the art is processed into an aqueous emulsion, a suspension, a water-dispersible granule, an emulsifiable concentrate, and the like.
- Commonly used pesticide additives include: liquid carriers such as water; organic solvents such as toluene, xylene, cyclohexane, methanol, butanol, ethylene glycol, acetone, dimethylformamide, diethyl ether, dimethyl sulfoxide, animals and Vegetable oils and fatty acids; commonly used surfactants such as emulsifiers and dispersants, including anionic surfactants, cationic surfactants, nonionic surfactants and amphoteric surfactants; other additives such as wetting agents, thickeners, etc. .
- the content in the insecticide may be selected from the range of 0.1% to 99.5%.
- the appropriate active ingredient content can be determined according to the form of the preparation and the method of administration.
- the aqueous emulsion contains from 5% to 50% by weight of the active ingredient, preferably in an amount of from 10% to 40%; in the suspension, from 5% to 50% of the active ingredient, preferably Its content is 5% to 40%.
- the compound of the present invention as an active ingredient can be uniformly mixed with an auxiliary agent such as a solvent and a surfactant, and can be diluted with water to a specified concentration during use.
- the water-dispersible granules can be prepared by mixing and pulverizing the compound of the present invention, a solid carrier, a surfactant, and the like as an active ingredient. Dilute with water when using.
- the processing method of the preparation is by no means limited to the above. A person skilled in the art can select an appropriate method depending on the active ingredient, the purpose of use, and the like.
- the insecticide of the present invention may contain other insecticides, acaricides, fungicides, insect growth regulators, plant growth, in addition to the compounds represented by the general formulae (I) and ( ⁇ ) as active ingredients. Any suitable active ingredient such as a regulator and a soil amendment.
- the insecticide of the present invention conventional application methods such as stem and leaf spray, surface application, soil treatment, and seed treatment can be selected.
- stem and leaf spray the compound represented by the general formulae (I) and ( ⁇ ) as an active ingredient can be used in an aqueous emulsion, a suspension, a water-dispersible granule, and a concentration range of 1 to 1000 mg/L.
- the emulsifiable concentrate preferably has a concentration of from 1 to 500 mg/L.
- novel o-heterocyclic anilide compound disclosed by the invention has excellent control effects on harmful insects, and therefore the compound can be used for preparing insecticides in the fields of agriculture, horticulture and the like, and has the advantages of high efficiency, low toxicity and environmental friendliness.
- the title compound was obtained by the synthesis of the compound of the compound N0.34.
- 25 parts of the compound of the present invention 6 parts of a wetting agent to sodium methyl fatty amide sulfonate, 2 parts of a suspending nonylphenol ethoxylate ether condensate, 6 parts of a tackifier carboxymethyl cellulose 1 part of preservative sodium salicylate, 2 parts of antifreeze propylene glycol, 1 part of antifoaming silicone oil, 57 parts of water, 25% suspension of active ingredient according to the suspension processing technology.
- test Example 1 Insecticidal effect on Plutella xylostella
- the 3rd instar larvae were selected and tested for insecticidal effects using a leaf-fed feeding method.
- the compounds of the examples of the present invention were each made into an insecticide.
- the obtained insecticide emulsifiable concentrate is diluted with purified water and uniformly mixed to obtain a desired concentration of the chemical solution.
- Select amaranth, wash and dry, use a puncher to make a leaf dish, dip in the liquid for 10 seconds, and then dry it naturally and put it into the dish.
- Each dish was connected to P. xylostella 3 instar larvae 10 times, repeated 3 times, the number of dead insects was investigated in the first ld and 3d, the mortality was counted, and the insecticidal effect was evaluated.
- Mortality statistics are shown in the table below.
- Test Example 2 Insecticidal effect on broad bean meal The 3 day old nymphs of Vicia faba, which were continuously raised in the room, were selected for the insecticidal effect test by the dipping method. According to the constitution of Formulation Example 3, the compounds of the present invention were each made into an insecticide. The obtained insecticide emulsifiable concentrate was diluted with purified water to prepare a 400 mg/L drug solution.
- the 3rd instar larvae of Chilo suppressalis were used, and the insecticidal effect test was carried out by using the rice seedling feeding method.
- the compounds of the examples of the present invention were each made into an insecticide.
- the obtained insecticide emulsifiable concentrate is diluted with purified water and uniformly mixed to obtain a desired concentration of the chemical solution.
- the rice seedlings were selected and divided into 10 strains/group, which were taken out in the liquid for 10 seconds, and then naturally dried to be placed in the finger tube. Each tube was connected to 10 larvae of the third instar larvae, and the larvae were counted three times.
- the number of dead insects was counted on the first ld, 3d, 5d, and 7d, and the mortality was evaluated to evaluate the insecticidal effect.
- Compound No. 11 had a mortality rate of 16.67, 36.67, 43.33, and 90.00% at the first ld, 3d, 5d, and 7d after treatment.
- Compound No. 34 had a mortality rate of 17.61, 34.61, 42.13, and 91.01% at ld, 3d, 5d, and 7d after treatment, respectively.
Landscapes
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Dentistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Plant Pathology (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Agronomy & Crop Science (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
说明书 邻杂环甲酰苯胺类化合物及其合成方法和应用 技术领域 Description o-heterocyclic anilide compound and synthesis method and application thereof
本发明属于农药领域, 具体涉及一种可以作为杀虫剂的邻杂环甲酰苯胺类化合物及 其制备方法。 害虫的防治一直以来是农药科学研究的核心领域,杀虫剂的普遍使用,使得多数虫害 得到了有效治理。随着杀虫剂应用规模的不断加大,有机合成杀虫剂在使用中产生的抗药 性等问题也愈发凸显。因此,作用于全新靶点的低毒化合物成为了杀虫剂研究的必然趋势。 The invention belongs to the field of pesticides, and particularly relates to an o-heterocyclic anilide compound which can be used as an insecticide and a preparation method thereof. The prevention and control of pests has always been the core field of pesticide scientific research. The widespread use of pesticides has led to the effective management of most pests. With the increasing use of pesticides, the problem of drug resistance caused by the use of organic synthetic insecticides has become more prominent. Therefore, low toxicity compounds acting on new targets have become an inevitable trend in pesticide research.
^:咖 ^:Cai
本发明的目的是提供针对各种害虫具有优良防治效果, 并具有高效、安全、环境相容 性好等特点的一类邻杂环甲酰苯胺类化合物, 以满足作物保护对高效安全杀虫剂的需求。 The object of the present invention is to provide a class of o-heterocyclic anilide compounds which have excellent control effects against various pests and are characterized by high efficiency, safety and good environmental compatibility, so as to meet crop protection and high-efficiency and safe insecticides. Demand.
本发明的另一目的是提供一种上述化合物的制备方法。 Another object of the present invention is to provide a process for the preparation of the above compounds.
本发明还有一个目的是提供上述化合物在制备杀虫剂方面的用途。 Still another object of the invention is to provide the use of the above compounds in the preparation of insecticides.
本发明的目的可以通过以下措施达到: The object of the invention can be achieved by the following measures:
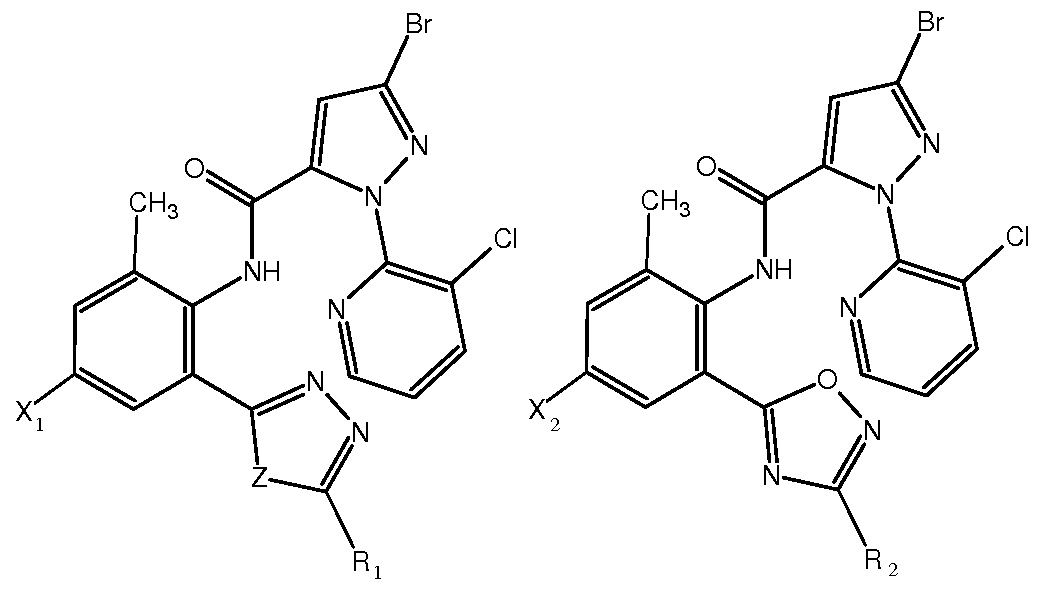
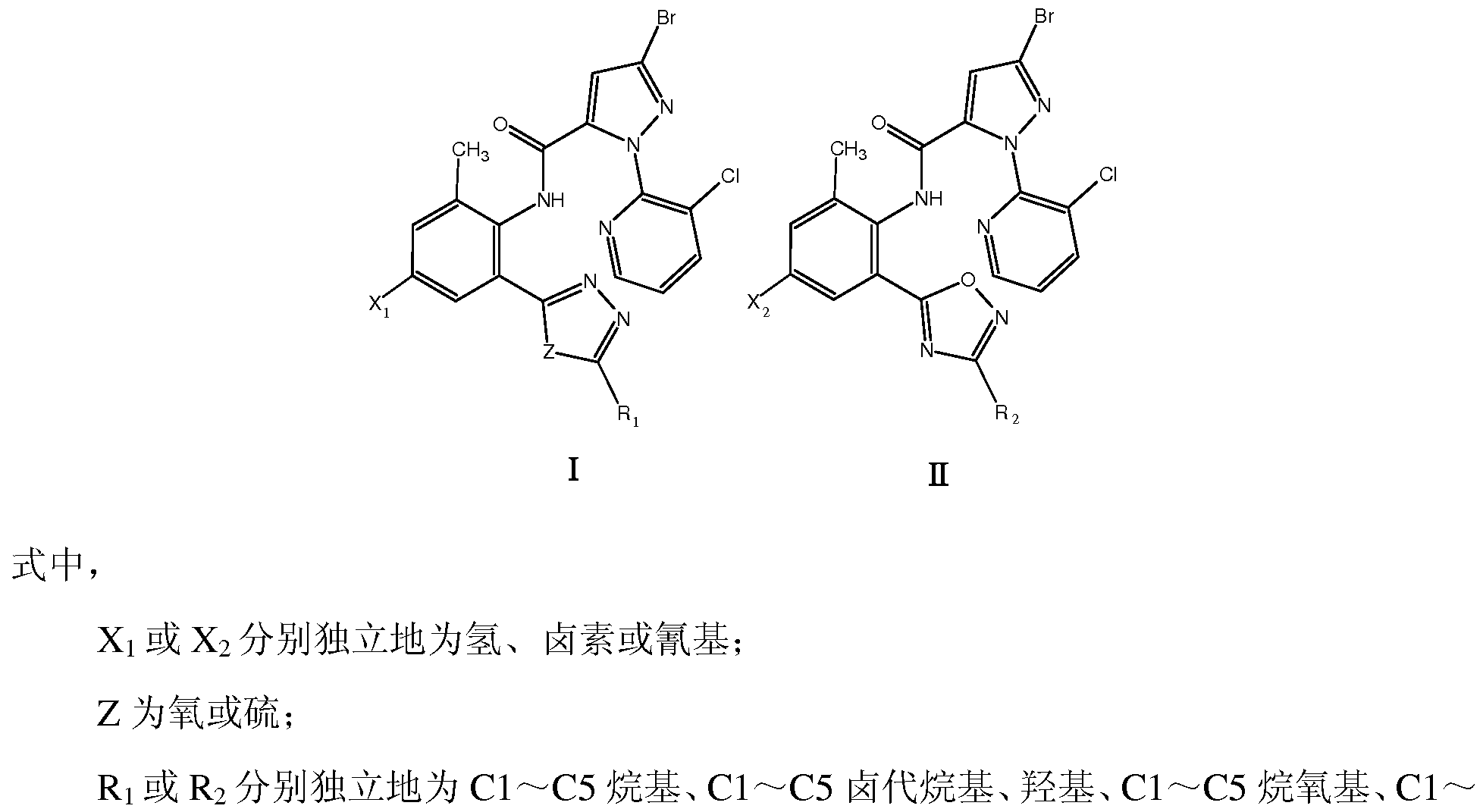
式 ( I ) 、 (Π ( I ) , (Π
I Π 式中, 1或 2分别独立地为氢、 卤素或氰基; Z为氧或硫; In the formula, 1 or 2 are independently hydrogen, halogen or cyano; Z is oxygen or sulfur;
Ri或 R2分别独立地为 C1〜C5垸基、 C1〜C5卤代垸基、羟基、 C1〜C5垸氧基、 Cl〜 C5卤代垸氧基、 巯基、 C1〜C5垸硫基、 氨基、 C1〜C5垸氨基 (包括 Ν,Ν-二垸氨基)、 C1〜C5卤代垸氨基、 C1〜C5垸氧氨基、 C1〜C5垸基磺酰胺基、 C1〜C5酰基、 C1〜C5 酰胺基、 卤素、 氰基或硫氰基。 Ri or R 2 are each independently a C1 to C5 fluorenyl group, a C1 to C5 halogenated fluorenyl group, a hydroxyl group, a C1 to C5 decyloxy group, a Cl~C5 halogenated fluorenyloxy group, a fluorenyl group, a C1 to C5 thiol group, an amino group. , C1~C5 垸amino group (including hydrazine, hydrazine-diamino group), C1~C5 halogenated hydrazine amino group, C1~C5 oxiranylamino group, C1~C5 decyl sulfonamide group, C1~C5 acyl group, C1~C5 Amido, halogen, cyano or thiocyano.
本发明中的 或 X2分别独立地优选为 H、 F、 CI 、 Br或 CN; X,或 X2分别独立地 进一步优选为 H、 F、 CI 或 Br; 其中 1最优选为 CI 、 Br或 CN, X2最优选为 CI 、 Br 或 CN。 The X or X 2 in the present invention are each independently preferably H, F, CI, Br or CN; X, or X 2 are each independently further preferably H, F, CI or Br; wherein 1 is most preferably CI, Br or CN, X 2 is most preferably CI, Br or CN.
本发明中的 或 R2分别独立地优选为 C1〜C5垸基、羟基、 C1〜C5垸氧基、巯基、 C1〜C5垸硫基、 氨基、 C1〜C5垸氨基、 C1〜C5垸基磺酰胺基、 C1〜C5酰胺基、 卤素、 氰基或硫氰基; 前述各取代基中的 C1〜C5可以进一步优选为 C1〜C3。 具体的讲!^或 R2可以分别独立地选自如下基团: -CH3、 -C2H5、 -CH(CH3)2、 -OH、 -OCH3、 -OC2H5、 -OCH(CH3)2、 -SH、 -SCH3、 -SC2H5、 -SCH(CH3)2、 -NH2、 -NHCH3、 -NHC2H5、 -N(CH3)2、 -NHCH(CH3)2、 -NHOCH3 -NHOC2H5、 -NHOCH(CH3)2 、 -N(CH3)2 -NHCF3 -NHCHF2 -NHCH2F、 -NHCOCH3、 -NHCOC2H5、 -NHS02CH3、 -NHS02C2H5、 卤素、 氰基或硫氰 基等。 In the present invention, or R 2 each independently preferably is C1 to C5 fluorenyl, hydroxy, C1 to C5 decyloxy, fluorenyl, C1 to C5 thiol, amino, C1 to C5 decylamino, C1 to C5 decyl sulfonate. An amide group, a C1 to C5 amide group, a halogen, a cyano group or a thiocyano group; and C1 to C5 in each of the above substituents may further preferably be C1 to C3. Specifically speaking! ^ or R 2 may be independently selected from the group consisting of: -CH 3 , -C 2 H 5 , -CH(CH 3 ) 2 , -OH, -OCH 3 , -OC 2 H 5 , -OCH (CH 3 2 , -SH, -SCH 3 , -SC 2 H 5 , -SCH(CH 3 ) 2 , -NH 2 , -NHCH 3 , -NHC 2 H 5 , -N(CH 3 ) 2 , -NHCH(CH 3 ) 2 , -NHOCH 3 -NHOC 2 H 5 , -NHOCH(CH 3 ) 2 , -N(CH 3 ) 2 -NHCF 3 -NHCHF 2 -NHCH 2 F, -NHCOCH 3 , -NHCOC 2 H 5 , - NHS0 2 CH 3 , -NHS0 2 C 2 H 5 , halogen, cyano or thiocyano group.
Ri进一步优选为 C1〜C5垸基、 羟基、 巯基、 C1〜C5垸硫基、 氨基、 C1〜C5垸氨 基、 C1〜C5 垸基磺酰胺基、 C1〜C5 酰胺基、 卤素、 氰基或硫氰基, 最优选为 C1〜C3 垸基、羟基、巯基、 C1〜C3垸硫基、氨基、 C1〜C3垸氨基、 C1〜C3垸基磺酰胺基、 Cl〜 C3酰胺基、 卤素 (优选 C1或 Br)、 氰基或硫氰基。 Further preferably, Ri is a C1 to C5 fluorenyl group, a hydroxy group, a decyl group, a C1 to C5 thiol group, an amino group, a C1 to C5 fluorenylamino group, a C1 to C5 decyl sulfonylamino group, a C1 to C5 amide group, a halogen, a cyano group or a sulfur. The cyano group is most preferably a C1 to C3 fluorenyl group, a hydroxy group, a decyl group, a C1 to C3 thiol group, an amino group, a C1 to C3 fluorenylamino group, a C1 to C3 fluorenyl sulfonamide group, a Cl~C3 amide group, a halogen (preferably C1). Or Br), cyano or thiocyano.
R2进一步优选为 C1〜C5垸基、氨基、 C1〜C5垸氨基、 C1〜C5垸基磺酰胺基、 Cl〜 C5酰胺基、 卤素、 氰基或硫氰基, 最优选为 C1〜C3垸基、 氨基、 C1〜C3垸氨基、 Cl〜 C3垸基磺酰胺基、 C1〜C3酰胺基、 卤素 (优选 C1或 Br)、 氰基或硫氰基。 R 2 is further preferably a C1 to C5 mercapto group, an amino group, a C1 to C5 mercaptoamino group, a C1 to C5 mercaptosulfonylamino group, a Cl~C5 amido group, a halogen, a cyano group or a thiocyano group, and most preferably a C1 to C3 group. Base, amino group, C1~C3 oxime amino group, Cl~C3 decyl sulfonamide group, C1~C3 amide group, halogen (preferably C1 or Br), cyano group or thiocyano group.
通式 ( I ) 和 (Π ) 所示的邻杂环甲酰苯胺类化合物的制备方法分别如下: The preparation methods of the o-heterocyclic anilide compounds represented by the general formulae (I) and (Π) are as follows:
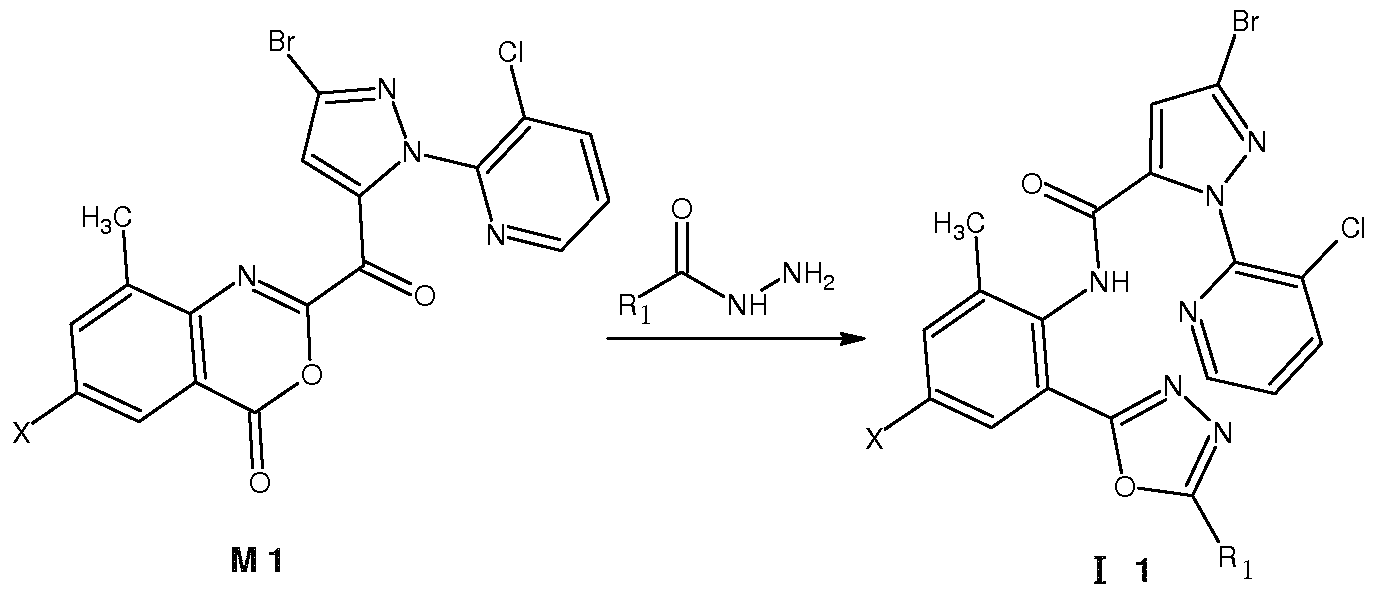
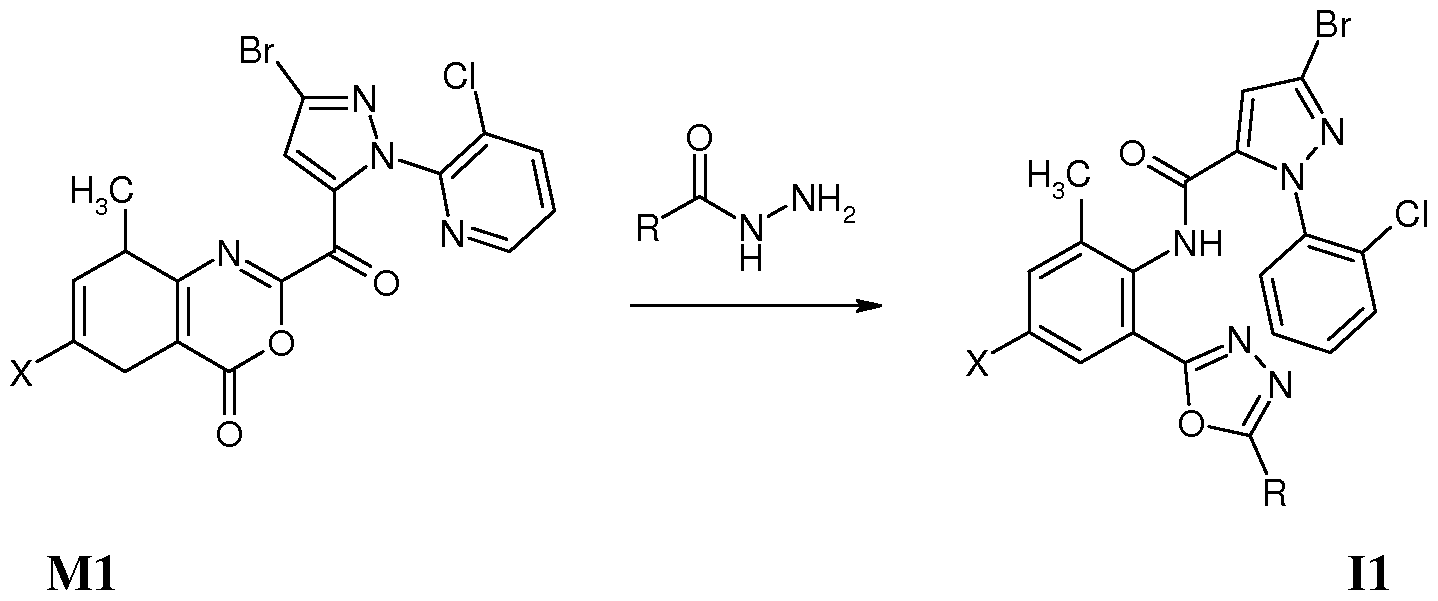
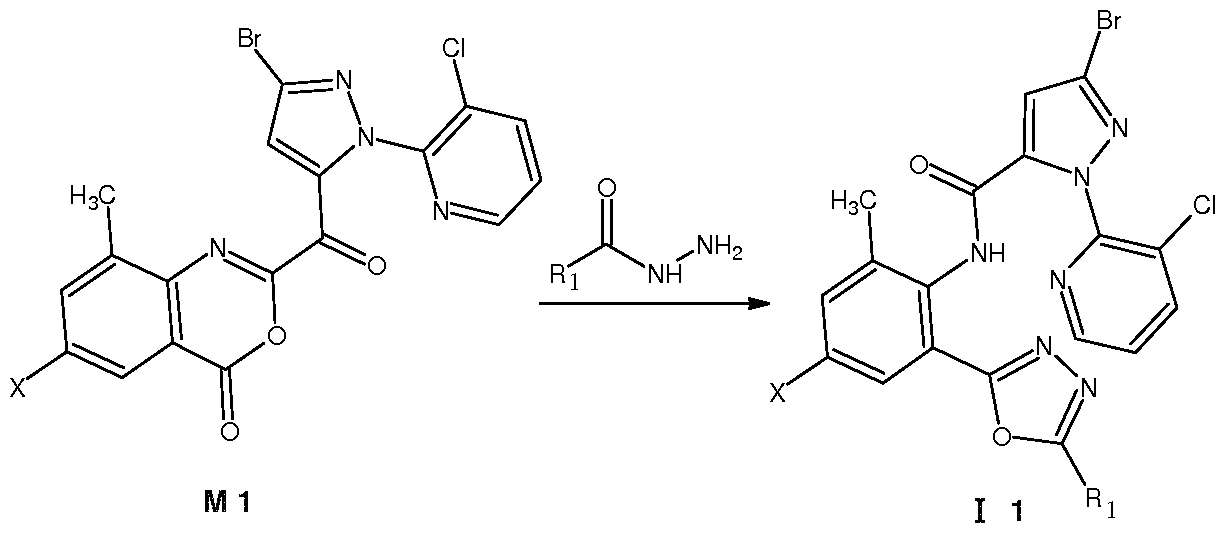
Z为氧, 1^为。1〜。5垸基、 氨基、 卤素、 氰基或硫氰基, X为 时, 在中性条件 下、 极性非质子溶剂中, 在室温下, 将中间体 Ml与相应的酰肼试剂 R ONHN ^反应 开环得到通式 ( I 1 ) 所示的化合物, 其反应路线为: Z is oxygen, 1^ is. 1~. 5 thiol, amino, halogen, cyano or thiocyano group, when X is under neutral conditions, in a polar aprotic solvent, the intermediate M1 is reacted with the corresponding hydrazide reagent R ONHN ^ at room temperature Ring opening gives a compound of the formula (I 1 ), the reaction route of which is:
Ri为 C1〜C5卤代垸基或 C1〜C5酰基时, 也可按上式进行制备。 为 C1〜C5垸氨基、 C1〜C5卤代垸氨基、 C1〜C5垸氧氨基、 C1〜C5垸基磺酰胺基或 C1〜C5酰基时, 将 为氨基的化合物 II与垸基化试剂、 垸氧化试剂、 垸基磺酰化试剂或酰基化试剂进行反应 即可制备。 为硫氰基时, 将 为卤素的化合物与硫氰化钠进行反应即可制备。 When Ri is a C1 to C5 halogenated fluorenyl group or a C1 to C5 acyl group, it can also be produced according to the above formula. When it is a C1~C5 oxime amino group, a C1~C5 halogenated oxime amino group, a C1~C5 oxime oxyamino group, a C1~C5 decyl sulfonamide group or a C1 ~C5 acyl group, the compound II which is an amino group and a thiolation reagent, hydrazine It can be prepared by reacting an oxidizing reagent, a mercaptosulfonylating reagent or an acylating reagent. When it is a thiocyano group, a compound which is a halogen can be prepared by reacting with sodium thiocyanate.
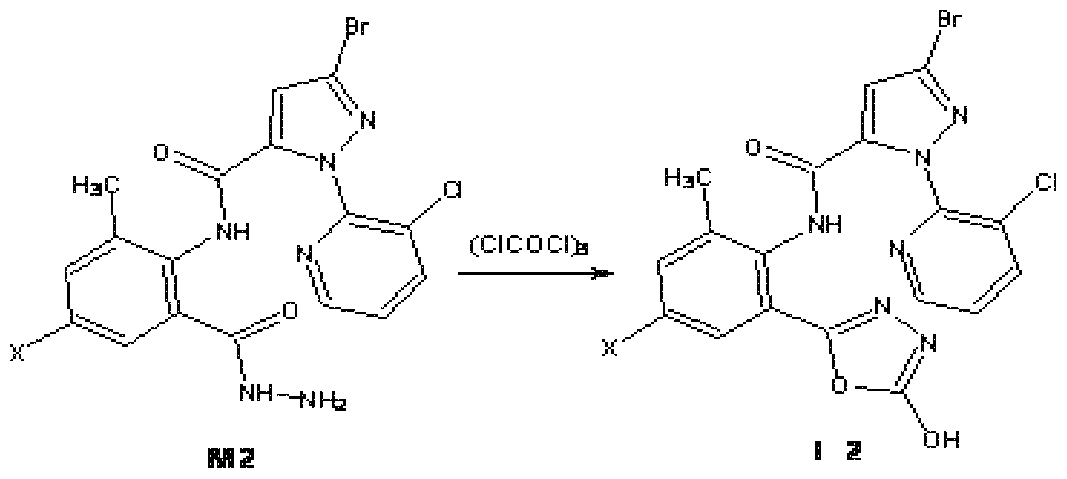
Z为氧, !^为羟基, X为 时, 先由中间体 Ml与水合肼反应制备中间体 M2, 在中 性条件下、 极性质子溶剂中, 在回流温度下, 中间体 M2 与三光气或光气反应闭环得到 通式 ( Z is oxygen, ! ^ is hydroxyl, when X is, firstly intermediate M1 is reacted with hydrazine hydrate to prepare intermediate M2. Under neutral conditions, polar protic solvent, intermediate M2 reacts with triphosgene or phosgene at reflux temperature. Get the formula
Ri为 C1〜C5垸氧基或 C1〜C5卤代垸氧基时, 将式 12化合物与垸基化试剂反应即可制 When Ri is a C1~C5 decyloxy group or a C1~C5 halogenated decyloxy group, the compound of the formula 12 can be reacted with a thiolation reagent.
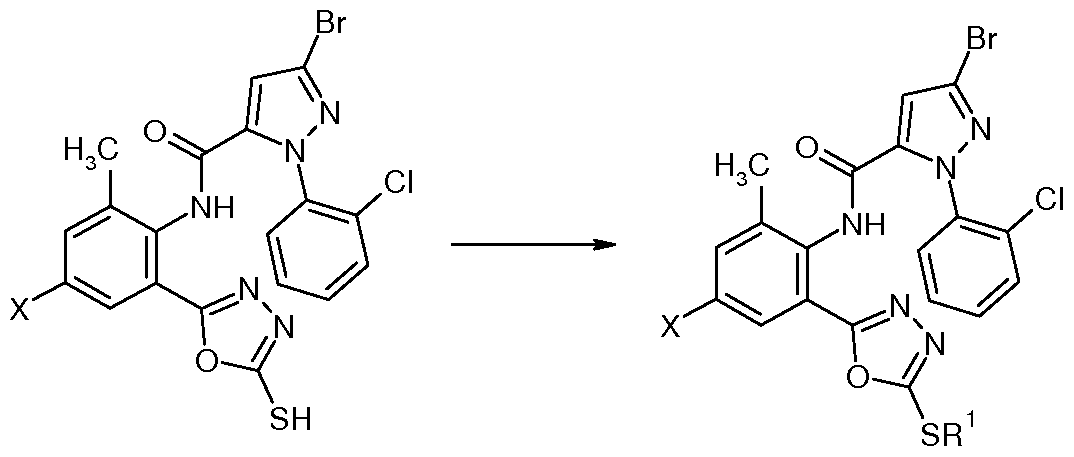
Z为氧, !^为巯基, X为 时, 在碱性条件下、 极性质子溶剂中, 在回流温度下, 中间体 M2与二硫化 应路线为: Z is oxygen, ! ^ is sulfhydryl, when X is, under basic conditions, polar protic solvent, at reflux temperature, intermediate M2 and disulfide should be routed as follows:
Ri为 C1〜C5垸硫基时, 将式 13化合物与垸基化试剂反应即可制备。 When Ri is a C1 to C5 thiol group, a compound of the formula 13 can be reacted with a thiolation reagent to prepare.
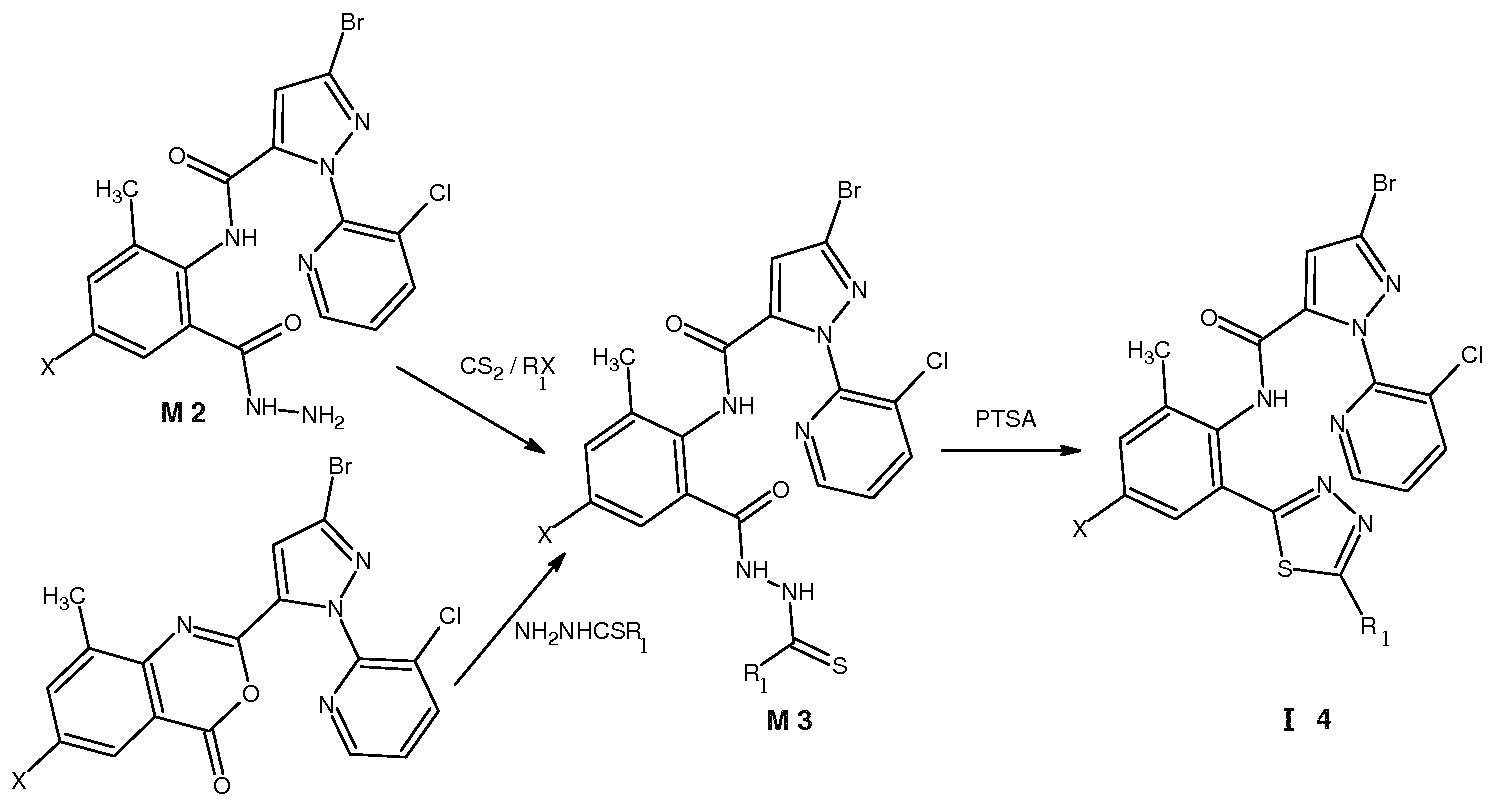
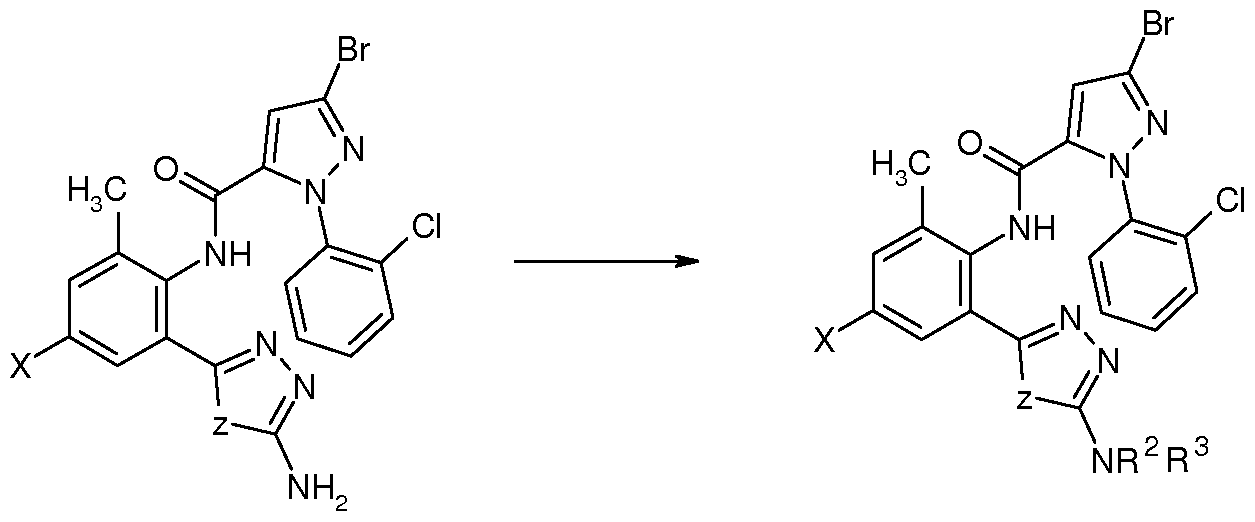
Z , X为 时, 在碱性条件下、 极性质子溶剂中, 在回流温度下, 用中间体 M2 与二硫化碳、 卤代烃反应, 得到中间体 M3, 再在溶剂中用有机弱酸作催化剂, M3反应 脱水缩合成环得到通式( I 4)所示的化合物;或者用中间体 Ml与相应的试剂 NH2NHCS 反应后得到 M3,再在溶剂中用有机弱酸作催化剂, M3反应脱水缩合成环得到通式( I 4) 所示的化合物, 反应路线为: When Z and X are used, intermediate M2 is reacted with carbon disulfide or halogenated hydrocarbon under basic conditions in a polar protic solvent at reflux temperature to obtain intermediate M3, and organic weak acid is used as a catalyst in the solvent. The M3 reaction is dehydrated and condensed to obtain a compound represented by the formula (I4); or the intermediate M1 is reacted with the corresponding reagent NH2NHCS to obtain M3, and then the organic weak acid is used as a catalyst in the solvent, and the M3 reaction is dehydrated and condensed to obtain a ring. A compound of the formula (I 4), the reaction route is:
Z为 S 时的式 I化合物的制备可以按上式制备, 如 为垸基或卤代垸基时可直接采用The preparation of the compound of formula I when Z is S can be prepared according to the above formula, and can be directly used when it is an anthracenyl group or a halogenated fluorenyl group.
M2-M3-I4的途径, R为氨基或其他基团(如羟基、巯基、氰基)时,也可以采用 M1-M3-I4 途径。 当 为。1〜。5垸氧基、 C1〜C5卤代垸氧基、 C1〜C5垸氨基、 C1〜C5卤代垸氨 基、 C1〜C5垸氧氨基、 C1〜C5垸基磺酰胺基或 C1〜C5酰基等时,可采用相应的垸基化、 磺酰胺基化或酰基化试剂与 14进一步反应制备。 当 为硫氰基时, 将 为卤素的化合 物与硫氰化钠进行反应即可制备。 The route of M2-M3-I4, when R is an amino group or other group (such as hydroxy, thiol, cyano), the M1-M3-I4 pathway can also be used. When. 1~. 5 methoxy, C1~C5 halogenated methoxy, C1~C5 垸amino, C1~C5 halogenated guanidine amino group, C1~C5 oxiranylamino group, C1~C5 decyl sulfonamide group or C1~C5 acyl group It can be prepared by further reaction with 14 using the corresponding guanidylation, sulfonamidation or acylation reagent. When it is a thiocyano group, a compound which is a halogen is reacted with sodium thiocyanate to prepare.
在碱性条件下、 极性质子溶剂中, 在 70-80 °C或回流温度下, 中间体 M4与相应的反 应试剂 HON=CNH2R2缩合成环得到通式 (Π ) 所示的化合物, 反应路线为: The intermediate M4 is condensed with the corresponding reagent HON=CNH 2 R 2 under basic conditions in a polar protic solvent at 70-80 ° C or reflux temperature to obtain a compound of the formula (Π). , the reaction route is:
R2的定义如上所述, 优选为 C1〜C5垸基、 C1〜C5卤代垸基、 羟基、 巯基、 氨基、 Cl〜 C5垸氨基、 C1〜C5垸基磺酰胺基或 C1〜C5酰基、 卤素、 氰基或硫氰基。 当 R2为 Cl〜 C5垸氨基、 C1〜C5垸基磺酰胺基或 C1〜C5酰基基团时, 可以采用上式制备的化合物进 一步与相应的垸基化、磺酰胺基化或酰基化试剂反应制备。 当 R2为硫氰基时, 将 R2为卤 素的化合物与硫氰化钠进行反应即可制备。 R 2 is as defined above, and is preferably a C1 to C5 fluorenyl group, a C1 to C5 halogenated fluorenyl group, a hydroxyl group, a decyl group, an amino group, a Cl~C5 fluorenylamino group, a C1 to C5 decyl sulfonylamino group or a C1 to C5 acyl group. Halogen, cyano or thiocyano. When R 2 is a Cl~C5 oxime amino group, a C1~C5 decyl sulfonamide group or a C1 ~C5 acyl group, the compound prepared by the above formula may be further used with the corresponding thiolation, sulfonamide or acylation reagent. Reaction preparation. When R 2 is a thiocyano group, a compound in which R 2 is a halogen is reacted with sodium thiocyanate to prepare.
本发明中所述的酸性条件或弱酸采用甲磺酸、对甲苯磺酸等有机酸;所述碱性条件采 用强碱或弱碱, 强碱为氢氧化钠、 氢氧化钾、 醇钠等; 所述的弱碱为吡啶、 三乙胺等; 所 述的极性质子溶剂为醇、 甲苯、 苯等溶剂; 所述的极性非质子溶剂为 DMF等溶剂。 本发 明单独提到的溶剂一般可以为极性质子溶剂, 也可以为极性非质子溶剂。 The acidic condition or the weak acid described in the present invention is an organic acid such as methanesulfonic acid or p-toluenesulfonic acid; the basic condition is a strong base or a weak base, and the strong base is sodium hydroxide, potassium hydroxide, sodium alkoxide or the like; The weak base is pyridine, triethylamine, etc.; The polar protic solvent is a solvent such as an alcohol, toluene or benzene; and the polar aprotic solvent is a solvent such as DMF. The solvent mentioned separately in the present invention may generally be a polar protic solvent or a polar aprotic solvent.
中间体 M2 由在 -10~30°C温度下, 将原料 (Ml ) 与水合肼反应制得 (参照专利 CN 200910033297), 反 Intermediate M2 is prepared by reacting raw material (Ml) with hydrazine hydrate at a temperature of -10~30 °C (refer to patent CN 200910033297),
M M 2 M M 2
通式 ( I ) 和 (Π ) 化合物对昆虫具有优良的防治活性, 因而本发明的化合物可用作 制备杀虫剂,进而保护农业、 园艺等植物。所述的昆虫有鳞翅目害虫如棉铃虫、甜菜夜蛾、 小菜蛾、 菜青虫、 稻纵卷叶螟和二化螟等, 同翅目害虫如叶蝉、 飞虱、 蚜虫、 粉虱等, 双 翅目害虫如家蝇、 潜叶蝇、 蚊类等, 直翅目和鞘翅目等害虫等。 当然, 本发明的化合物可 防治的有害生物不限于上述举例的范围。 The compounds of the formula (I) and (Π) have excellent control activity against insects, and thus the compounds of the present invention can be used as an insecticide to protect plants such as agriculture and horticulture. The insects are lepidopteran pests such as cotton bollworm, beet armyworm, diamondback moth, cabbage caterpillar, rice leaf roller and rice stem borer, and homopteran pests such as spider mites, planthoppers, locusts, whiteflies, etc. , Diptera pests such as housefly, leaf miner, mosquito, etc., Orthoptera and coleoptera and other pests. Of course, the pests to which the compounds of the present invention are controll are not limited to the scope of the above examples.
当由通式 ( I ) 和 (Π ) 表示的本发明的化合物用作农业、 园艺等领域的杀虫剂时, 可单独使用, 或以杀虫组合物的方式使用, 如以式 ( I ) 或式 (Π ) 为活性成分, 加上本 领域常用的农药助剂加工成水乳剂、 悬浮剂、 水分散颗粒剂、 乳油等。 When the compound of the present invention represented by the general formulae (I) and (?) is used as an insecticide in the fields of agriculture, horticulture, etc., it may be used alone or in the form of a pesticidal composition, such as by formula (I) Or the formula (Π) is an active ingredient, and a pesticide adjuvant commonly used in the art is processed into an aqueous emulsion, a suspension, a water-dispersible granule, an emulsifiable concentrate, and the like.
常用的农药助剂包括: 液体载体, 如水; 有机溶剂如甲苯、 二甲苯、 环己垸、 甲醇、 丁醇、 乙二醇、 丙酮、 二甲基甲酰胺、 乙醚、 二甲亚砜、 动物和植物油及脂肪酸; 常用的 表面活性剂如乳化剂和分散剂, 包括阴离子表面活性剂、 阳离子表面活性剂、 非离子表面 活性剂和两性表面活性剂; 其它助剂, 如湿润剂、 增稠剂等。 Commonly used pesticide additives include: liquid carriers such as water; organic solvents such as toluene, xylene, cyclohexane, methanol, butanol, ethylene glycol, acetone, dimethylformamide, diethyl ether, dimethyl sulfoxide, animals and Vegetable oils and fatty acids; commonly used surfactants such as emulsifiers and dispersants, including anionic surfactants, cationic surfactants, nonionic surfactants and amphoteric surfactants; other additives such as wetting agents, thickeners, etc. .
由通式 ( I ) 和 (Π )表示的本发明的化合物用作杀虫剂中的活性成分时, 在所述杀 虫剂中的含量可在 0 .1%至 99.5%的范围内进行选择, 并可根据制剂形式和施用方法确定适 当的活性成分含量。通常,在水乳剂中含有 5%至 50% (重量百分比,下同)所述的活性成分, 优选其含量为 10%至 40%; 在悬浮剂中含有 5%至 50%的活性成分, 优选其含量为 5%至 40% 。 When the compound of the present invention represented by the general formulae (I) and (?) is used as an active ingredient in the insecticide, the content in the insecticide may be selected from the range of 0.1% to 99.5%. The appropriate active ingredient content can be determined according to the form of the preparation and the method of administration. Usually, the aqueous emulsion contains from 5% to 50% by weight of the active ingredient, preferably in an amount of from 10% to 40%; in the suspension, from 5% to 50% of the active ingredient, preferably Its content is 5% to 40%.
例如, 对于所述水乳剂、 悬浮剂, 可将作为活性成分的本发明化合物与溶剂和表面 活性剂等助剂进行均匀混合而制成, 使用时可用水稀释到指定浓度。对于所述水分散颗粒 剂,可将作为活性成分的本发明化合物、固体载体和表面活性剂等混合并进行粉碎而制成, 使用时用水进行稀释。 当然, 制剂的加工方法绝不限于上述内容。 本领域的技术人员可根 据所述活性成分和使用目的等, 选择合适的方法。 For example, for the aqueous emulsion or suspending agent, the compound of the present invention as an active ingredient can be uniformly mixed with an auxiliary agent such as a solvent and a surfactant, and can be diluted with water to a specified concentration during use. The water-dispersible granules can be prepared by mixing and pulverizing the compound of the present invention, a solid carrier, a surfactant, and the like as an active ingredient. Dilute with water when using. Of course, the processing method of the preparation is by no means limited to the above. A person skilled in the art can select an appropriate method depending on the active ingredient, the purpose of use, and the like.
除了作为活性成分的由通式 ( I ) 和 (Π ) 表示的所述化合物之外, 本发明的杀虫剂 可包含其它杀虫剂、 杀螨剂、 杀菌剂、 昆虫生长调节剂、 植物生长调节剂和土壤改良剂等 任何适用的活性成分。 The insecticide of the present invention may contain other insecticides, acaricides, fungicides, insect growth regulators, plant growth, in addition to the compounds represented by the general formulae (I) and (Π) as active ingredients. Any suitable active ingredient such as a regulator and a soil amendment.
对于本发明的杀虫剂的使用, 可选择常用的施药方法, 如茎叶喷雾、 水面施用、 土壤 处理和种子处理等。 例如, 当采用茎叶喷雾时, 作为活性成分的由通式 ( I ) 和 (Π ) 表 示的化合物的可使用浓度范围为 1至 1000 mg/L 的水乳剂、 悬浮剂、 水分散颗粒剂、 乳油, 优选其浓度为 1 至 500 mg/L。 For the use of the insecticide of the present invention, conventional application methods such as stem and leaf spray, surface application, soil treatment, and seed treatment can be selected. For example, when a stem and leaf spray is used, the compound represented by the general formulae (I) and (Π) as an active ingredient can be used in an aqueous emulsion, a suspension, a water-dispersible granule, and a concentration range of 1 to 1000 mg/L. The emulsifiable concentrate preferably has a concentration of from 1 to 500 mg/L.
本发明公开的新型邻杂环甲酰苯胺类化合物对有害昆虫具有优良的防治效果, 因此该 化合物可用制备农业、 园艺等领域的杀虫剂, 具有高效、 低毒、 环境友好的优点。 The novel o-heterocyclic anilide compound disclosed by the invention has excellent control effects on harmful insects, and therefore the compound can be used for preparing insecticides in the fields of agriculture, horticulture and the like, and has the advantages of high efficiency, low toxicity and environmental friendliness.
为了便于对本发明的进一步了解, 下面提供的实施例对其做了更详细的说明。这些实 施例仅供叙述而并非用来限定本发明的范围或实施原则。 In order to facilitate a further understanding of the present invention, the embodiments provided below are described in more detail. These examples are for illustrative purposes only and are not intended to limit the scope or implementation of the invention.
实施例 1 : Example 1
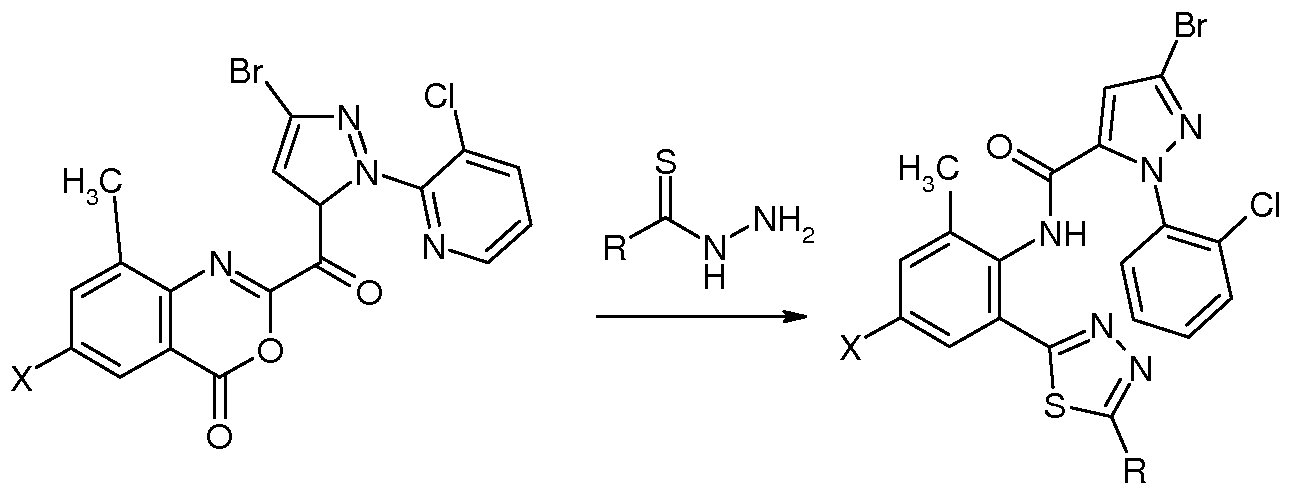
反应通 Reaction
化合物 NO.l (X=H, R=CH3): 3-溴 -N-{2-[2-(5-甲基 -1,3,4-噁二唑 )]-6-甲基苯基}-1-(3- 氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Compound NO.l (X=H, R=CH 3 ): 3-bromo-N-{2-[2-(5-methyl-1,3,4-oxadiazole)]-6-methylbenzene Synthesis of -1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide)
取 2.0 g (4.4 mmol) 2-(3-溴 -1-(3-氯 -2-吡啶基 )-1 H-吡唑) -5-甲基 -4 H-3,1-苯并噁嗪 -4- 酮 (Ml ) 悬浮于 40 mL DMF中, 加入 0.4 g乙酰肼, 室温反应过夜, 次日得澄清溶液, 搅拌下将反应液缓慢加入 100 mL水中, 缓慢析出固体, 继续搅拌 2 h, 过滤、 干燥得成 品 1.45 g。 化合物 NO.2 (X=CL, R=CH3 ): 3-溴 -N-{4-氯 -2-[2-(5-甲基 -1,3,4-噁二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 Take 2.0 g (4.4 mmol) of 2-(3-bromo-1-(3-chloro-2-pyridyl)-1 H-pyrazole)-5-methyl-4 H-3,1-benzoxazine -4- Ketone (Ml) was suspended in 40 mL of DMF, added with 0.4 g of acetohydrazide, and reacted at room temperature overnight. The solution was clarified the next day. The reaction solution was slowly added to 100 mL of water with stirring. The solid was slowly precipitated and stirring was continued for 2 h. Filtered and dried to obtain 1.45 g of finished product. Compound NO. 2 (X=CL, R=CH 3 ): 3-bromo-N-{4-chloro-2-[2-(5-methyl-1,3,4-oxadiazole)]-6 Synthesis of -methylphenyl}-1-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
按实施例 1 化合物 N0.1的合成方法, 2-(3-溴 -1-(3-氯 -2-吡啶基 )-1 H-吡唑) -7-氯 -5-甲基 -4 H-3,1-苯并噁嗪 -4-酮 (M1C1) 与乙酰肼反应得到标题化合物 (N0.2) 1.51 g。 化合物 N0.23 (X=CN, R= CH3): 3-溴 -N-{4-氰基 2-[2-(5-氨基 -1,3,4-噁二唑 )]-6-甲基 苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Synthesis of Compound N0.1 as in Example 1, 2-(3-bromo-1-(3-chloro-2-pyridyl)-1 H-pyrazole)-7-chloro-5-methyl-4H Reaction of -3,1-benzoxazin-4-one (M1C1) with acetohydrazide gave the title compound (N0.2) 1.51 g. Compound N0.23 (X=CN, R=CH 3 ): 3-bromo-N-{4-cyano 2-[2-(5-amino-1,3,4-oxadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例 1 化合物 N0.1的合成方法得到标题化合物。 实施例 2 : The title compound was obtained according to the procedure of the compound 10.1. Example 2:
反应通式: Reaction formula:
化合物 N0.3 (X=CL, R=OH) : 3-溴 -N-{4-氯 -2-[2-(5-羟基 -1,3,4-噁二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Compound N0.3 (X=CL, R=OH) : 3-bromo-N-{4-chloro-2-[2-(5-hydroxy-1,3,4-oxadiazole)]-6- Synthesis of phenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
将 l.O g (2.07 mmol) M2和 0.8 g (2.68 mol) 三光气加至 20 mL的甲苯中回流反 应 4 h, 浓缩后将滤液缓慢倾入水中, 搅拌 2 h以上, 静置, 过滤, 水洗至滤液 pH=7, 过 滤, 乙醚重结晶后即得 l.O g固体产品。 实施例 3 : Add 10 g (2.07 mmol) of M2 and 0.8 g (2.68 mol) of triphosgene to 20 mL of toluene and reflux for 4 h. After concentration, the filtrate was slowly poured into water, stirred for more than 2 h, allowed to stand, filtered, washed with water until The filtrate pH = 7, filtered, and recrystallized from diethyl ether to give 10 g of solid product. Example 3:
反应通式 Reaction formula
化合物 NO.4 (X=CL, R=SH) : 3-溴 -N-{4-氯 -2-[2-(5-巯基 -1,3,4-噁二唑 )]-6-甲基苯 基 }-l-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 Compound NO.4 (X=CL, R=SH) : 3-bromo-N-{4-chloro-2-[2-(5-fluorenyl-1,3,4-oxadiazole)]-6- Synthesis of phenyl}-l-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
将 0.97g (2.0 mmol) 中间体 M2与 5 mL二硫化碳加至 20 mL吡啶中, 加热至 50 V 恒温反应 3天, 结束后将此反应液倾入冰水中充分搅拌 2 h后静置, 等固体充分析出后过 滤, 醇洗、 水洗, 干燥得产品 0.85 g。 化合物 N0.7 (X=Br, R=SH) : 3-溴 -N-{4-溴 -2-[2-(5-巯基 -1,3,4-噁二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Add 0.97g (2.0 mmol) of intermediate M2 and 5 mL of carbon disulfide to 20 mL of pyridine, heat to 50 V for 3 days, and then pour the reaction into ice water and stir for 2 h, then let stand, wait for solid. After the analysis, the mixture was filtered, washed with alcohol, washed with water, and dried to obtain 0.85 g of product. Compound N0.7 (X=Br, R=SH) : 3-bromo-N-{4-bromo-2-[2-(5-fluorenyl-1,3,4-oxadiazole)]-6- Synthesis of phenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.4的合成方法进行合成得相应的标题化合物。 化合物 N0.24 (X=CN, R=SH): 3-溴 -N-{4-氰基 -2-[2-(5-巯基 -1,3,4-噁二唑 )]-6-甲基 苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 The title compound was obtained by the synthesis of the compound of Example N. Compound N0.24 (X=CN, R=SH) : 3-bromo-N-{4-cyano-2-[2-(5-fluorenyl-1,3,4-oxadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.4的合成方法进行合成得到相应的标题化合物。 实施例 4 : Synthesis according to the synthesis of the compound of Example N0.4 gave the corresponding title compound. Example 4:
反应通式 Reaction formula
13 1 3
化合物 N0.5 (X=CL, R^CHs): 3-溴 -N-{4-氯 -2-[2-(5-巯甲基 -1,3,4-噁二唑 )]-6-甲基 苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Compound N0.5 (X=CL, R^CHs): 3-bromo-N-{4-chloro-2-[2-(5-fluorenyl-1,3,4-oxadiazole)]-6 Synthesis of -methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
将 1.3 g (2.4 mmol) 13溶于 5 mL 10%的氢氧化钠溶液中,然后加入含碘甲垸 0.2 mL (3.0 mmol) 乙醇溶液 20 mL。 室温反应 10 h后倾入 50 mL水中, 搅拌 2 h后静置, 过滤, 水洗, 乙醇重结晶后干燥得相应的标题化合物 0.88 go 化合物 NO.6 (X=CL, R = S CH (CH3) 2): 3-溴 -N-{4-氯 -2-[2-(5-巯异丙基 -1,3,4-噁 二唑) ]-6-甲基苯基 }-l-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 Dissolve 1.3 g (2.4 mmol) of 1 3 in 5 mL of 10% sodium hydroxide solution, then add 0.2 mL of methyl iodide (3.0 Methyl) 20 mL of ethanol solution. After reacting for 10 h at room temperature, it was poured into 50 mL of water, stirred for 2 h, then allowed to stand, filtered, washed with water, and recrystallized from ethanol to give the corresponding title compound 0.88 ° Compound No. 6 (X=CL, R = S CH (CH 3 2 ): 3-bromo-N-{4-chloro-2-[2-(5-indoleisopropyl-1,3,4-oxadiazole)]-6-methylphenyl}-l- Synthesis of (3-Chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
按实施例化合物 N0.5的合成方法进行合成得相应的标题化合物 1.20 g。 化合物 NO.8 (X=Br, R^CHs): 3-溴 -N-{4-溴 -2-[2-(5-巯甲基 -1,3,4-噁二唑 )]-6-甲基 苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 The title compound 1.20 g was obtained by the title compound. Compound No. 8 (X=Br, R^CHs): 3-bromo-N-{4-bromo-2-[2-(5-fluorenyl-1,3,4-oxadiazole)]-6 Synthesis of -methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.5的合成方法进行合成得相应的标题化合物。 化合物 N0.25 (X=CN, : 3-溴 -N-{4-氰基 -2-[2-(5-巯甲基 -1,3,4-噁二唑 )]-6- 甲基苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 The title compound was obtained by the synthesis of the compound of Example N. Compound N0.25 (X=CN, : 3-bromo-N-{4-cyano-2-[2-(5-fluorenyl-1,3,4-oxadiazole)]-6-methylphenyl}-1-(3- Synthesis of Chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.5的合成方法进行合成得相应的标题化合物。 实施例 5 : The title compound was obtained by the synthesis of the compound of compound N0.5. Example 5:
反应通式 Reaction formula
M 3 1 4 M 3 1 4
化合物 NO.9 (X=CL, R=SCH3): 3-溴 -N-{4-氯 -2-[2-(5-巯甲基 -1,3,4-噻二唑 )]-6-甲基 苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Compound NO.9 (X=CL, R=SCH 3 ): 3-bromo-N-{4-chloro-2-[2-(5-fluorenyl-1,3,4-thiadiazole)]- Synthesis of 6-Methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
将 0.97 g ( 1.70 mmol) M3 (R为 SCH3)与 0.38 g ( 1.97 mmol)对甲苯磺酸加至 20 mL 甲苯中, 加热至回流反应 2 h, 脱溶后用环己垸重结晶, 干燥得相应的标题化合物 0.82 g。 化合物 N0.26 (X=CN, R=SCH3): 3-溴 -N-{4-氯 -2-[2-(5-巯甲基 -1,3,4-噻二唑 )]-6-甲 基苯基 }-l-(3-氯 -2-吡啶基 )-m-吡唑 -5-甲酰胺的合成 Add 0.97 g ( 1.70 mmol) M3 (R is SCH 3 ) and 0.38 g ( 1.97 mmol) of p-toluenesulfonic acid to 20 mL of toluene, heat to reflux for 2 h, desolvate and recrystallize with cyclohexane, dry. The corresponding title compound was obtained in 0.82 g. Compound N0.26 (X=CN, R=SCH 3 ): 3-bromo-N-{4-chloro-2-[2-(5-fluorenyl-1,3,4-thiadiazole)]- 6-A Synthesis of phenyl}-l-(3-chloro-2-pyridyl)-m-pyrazole-5-carboxamide
按实施例化合物 N0.9的合成方法进行合成得相应的标题化合物。 化合物 NO.10 (X=CL, R=NH2): 3-溴 -N-{4-氯 -2-[2-(5-氨基 -1,3,4-噻二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 The title compound was obtained by the synthesis of the compound of Example N. Compound NO. 10 (X=CL, R=NH 2 ) : 3-bromo-N-{4-chloro-2-[2-(5-amino-1,3,4-thiadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
将 0.2 mL甲基磺酸滴加至 20 mL含 1.0 g (2.73 mmol) M3 (R为 NH2) 的甲苯溶液 中, 加热至回流反应 3 h后冷至室温, 继续反应过夜, 次日将该反应液悬浮于 35 mL乙酸 乙酯中, 剧烈搅拌下加入 20 mL 10% 的氨水溶液, 过滤得相应的标题化合物 0.5 g。 化合物 N0.27 (X=Br, R=NH2): 3-溴 -N-{4-溴 -2-[2-(5-氨基 -1,3,4-噻二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 0.2 mL of methyl sulfonic acid was added dropwise to 20 mL of a toluene solution containing 1.0 g (2.73 mmol) of M3 (R is NH 2 ), heated to reflux for 3 h, cooled to room temperature, and the reaction was continued overnight. The reaction mixture was suspended in ethyl acetate (35 mL). Compound N0.27 (X=Br, R=NH 2 ): 3-bromo-N-{4-bromo-2-[2-(5-amino-1,3,4-thiadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 NO.10的合成方法进行合成得相应的标题化合物。 化合物 N0.28 (X=CN, R=NH2): 3-溴 -N-{4-氰基 -2-[2-(5-氨基 -1,3,4-噻二唑 )]-6-甲基 苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 The title compound was obtained by the synthesis of the compound of Example No.10. Compound N0.28 (X=CN, R=NH 2 ) : 3-bromo-N-{4-cyano-2-[2-(5-amino-1,3,4-thiadiazole)]-6 Synthesis of -methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 NO.10的合成方法进行合成得相应的标题化合物。 化合物 N0.39 (X=CN, R=CH3): 3-溴 -N-{4-氰基 -2-[2-(5-甲基 -1,3,4-噻二唑 )]-6-甲基 苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 The title compound was obtained by the synthesis of the compound of Example No.10. Compound N0.39 (X=CN, R=CH 3 ): 3-bromo-N-{4-cyano-2-[2-(5-methyl-1,3,4-thiadiazole)]- Synthesis of 6-Methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.9的合成方法进行合成得相应的标题化合物。 化合物 NO.40 (X=Br, R=OH): 3-溴 -N-{4-氰基 -2-[2-(5-甲基 -1,3,4-噻二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 The title compound was obtained by the synthesis of the compound of Example N. Compound NO. 40 (X = Br, R = OH): 3-bromo-N-{4-cyano-2-[2-(5-methyl-1,3,4-thiadiazole)]-6 Synthesis of -methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.9的合成方法进行合成得相应的标题化合物。 化合物 N0.41 (X=CI, R=SH): 3-溴 -N-{4-氰基 -2-[2-(5-甲基 -1,3,4-噻二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 The title compound was obtained by the synthesis of the compound of Example N. Compound N0.41 (X=CI, R=SH) : 3-bromo-N-{4-cyano-2-[2-(5-methyl-1,3,4-thiadiazole)]-6 Synthesis of -methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.9的合成方法进行合成得相应的标题化合物。 实施例 6 The title compound was obtained by the synthesis of the compound of Example N. Example 6
化合物 NO.ll (X=CL, R=CH3 ) : 3-溴 -N-{4-氯 -2-[2-(5-甲基 -1,2,4-噁二唑 )]-6-甲基苯 基 }-l-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 Compound NO.ll (X=CL, R=CH 3 ) : 3-bromo-N-{4-chloro-2-[2-(5-methyl-1,2,4-oxadiazole)]-6 Synthesis of -methylphenyl}-l-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
将 1.0 g (2.07 mmol) M4和 0.33 g (4.46 mol) Ν'-羟基乙脒加至 15 mL的乙醇中, 冰浴冷却至 0 °C后, 缓慢滴加乙醇钠的乙醇溶液(由 0.1 g的金属钠加至 10 mL的乙醇中 搅拌反应 30 min得到), 完后冷至室温保持反应 2 h, 加热回流反应过夜。 次日冷至室温 后, 过滤, 滤液缓慢倾入水中, 搅拌 2 h以上, 静置, 过滤, 水洗, 干燥即得白色固体产 品 0.6 g。 化合物 N0.12 (X=Br, R= CH3 ) : 3-溴 -N-{4-溴 -2-[2-(5-甲基 -1,2,4-噁二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Add 1.0 g (2.07 mmol) of M4 and 0.33 g (4.46 mol) of Ν'-hydroxyethyl hydrazine to 15 mL of ethanol, cool to 0 °C in an ice bath, and slowly add ethanol solution of sodium ethoxide (from 0.1 g). The sodium metal was added to 10 mL of ethanol and stirred for 30 min to obtain), and after cooling to room temperature, the reaction was kept for 2 h, and the reaction was heated under reflux overnight. After cooling to room temperature on the next day, it was filtered, and the filtrate was slowly poured into water, stirred for 2 h or more, allowed to stand, filtered, washed with water, and dried to give a white solid product (0.6 g). Compound N0.12 (X=Br, R=CH 3 ) : 3-bromo-N-{4-bromo-2-[2-(5-methyl-1,2,4-oxadiazole)]-6 Synthesis of -methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 NO.ll的合成方法进行合成得相应的标题化合物。 化合物 N0.29 (X=CL, R=CN) : 3-溴 -N-{4-氯 -2-[2-(5-氰基 -1,2,4-噁二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 The title compound was obtained by the synthesis of the compound of the compound of Example No. 11. Compound N0.29 (X=CL, R=CN): 3-bromo-N-{4-chloro-2-[2-(5-cyano-1,2,4-oxadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 NO.ll的合成方法, 将 M4与 HON=CNH2CN进行合成得相应的标 题化合物。 化合物 NO.30 (X=CL, R=NH2) : 3-溴 -N-{4-氯 -2-[2-(5-氨基 -1,2,4-噁二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 M4 was synthesized with HON=CNH 2 CN according to the method of the synthesis of the compound of the present invention. Compound NO. 30 (X=CL, R=NH 2 ) : 3-bromo-N-{4-chloro-2-[2-(5-amino-1,2,4-oxadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 NO.ll的合成方法, 将 M4与 HON=CNH2NH2进行合成得相应的标 题化合物。 M4 was synthesized with HON = CNH 2 NH 2 to give the corresponding title compound.
化合物 N0.45 (X=CN, R=Br) : 3-溴 -N-{4-氰基 -2-[2-(5-溴 -1,2,4-噁二唑 )]-6-甲基苯 基 }-l-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 Compound N0.45 (X=CN, R=Br) : 3-bromo-N-{4-cyano-2-[2-(5-bromo-1,2,4-oxadiazole)]-6- Methylbenzene Synthesis of }}-l-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
按实施例化合物 N0.29的合成方法, 将 M4与 HON=CNH2Br进行合成得相应的标题化 合物。 化合物 N0.46 (X=Br, R=SCN): 3-溴 -N-{4-溴 -2-[2-(5-硫氰基 -1,2,4-噁二唑 )]-6-甲基 苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 M4 was synthesized with HON=CNH 2 Br according to the method of the compound of Example N0.29 to give the corresponding title compound. Compound N0.46 (X=Br, R=SCN): 3-bromo-N-{4-bromo-2-[2-(5-thiocyano-1,2,4-oxadiazole)]-6 Synthesis of -methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
取 2.0g( 3.6mmol ) 3-溴 -N-{4-溴 -2-[2-(5-氯 -1,2,4-噁二唑 )]-6-甲基苯基}-1-(3-氯 -2-吡啶 基) -1H-吡唑 -5-甲酰胺悬浮于 20 mL DMF中, 加入 0.5g硫氰化钠, 室温反应过夜,, 减压 脱溶后, 水洗, 过滤, 环己垸重结晶, 干燥, 得相应的标题化合物 1.0g。 Take 2.0 g (3.6 mmol) of 3-bromo-N-{4-bromo-2-[2-(5-chloro-1,2,4-oxadiazole)]-6-methylphenyl}-1- (3-Chloro-2-pyridyl)-1H-pyrazole-5-carboxamide was suspended in 20 mL of DMF, added with 0.5 g of sodium thiocyanate, reacted at room temperature overnight, desolvated under reduced pressure, washed with water, filtered. The Cyclohexanide was recrystallized and dried to give the corresponding title compound 1.0 g.
化合物 N0.13 (X=CL, R=NH2): 3-溴 -N-{4-氯 -2-[2-(5-氨基 -1,3,4-噁二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Compound N0.13 (X=CL, R=NH 2 ) : 3-bromo-N-{4-chloro-2-[2-(5-amino-1,3,4-oxadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
取 2.0g( 4.2mmol )2-(3-溴 -1-(3-氯 -2-吡啶基 )-1 H-吡唑) -5-甲基 -4 H-3,1-苯并噁嗪 -4-酮 (Ml ) 悬浮于 40 mL DMF中, 加入 0.8g氨基脲, 室温反应过夜, 加入对甲苯磺酸 l.Og 和 20 mL 甲苯, 加热至回流反应 4h, 脱溶后用环己垸重结晶, 干燥得相应的标题化合物 (NO.13) 1.2g。 化合物 N0.14 (X=Br, R=NH2): 3-溴 -N-{4-溴 -2-[2-(5-氨基 -1,3,4-噁二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 按实施例化合物 NO.13的合成方法进行合成得相应的标题化合物。 化合物 N0.31 (X=CL, R=CN): 3-溴 -N-{4-氯 -2-[2-(5-氰基 -1,3,4-噁二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Take 2.0 g (4.2 mmol) of 2-(3-bromo-1-(3-chloro-2-pyridyl)-1 H-pyrazole)-5-methyl-4 H-3,1-benzoxazine -4-ketone (Ml) was suspended in 40 mL of DMF, added with 0.8 g of semicarbazide, reacted at room temperature overnight, added with 1.0 g of p-toluenesulfonic acid and 20 mL of toluene, heated to reflux for 4 h, desolvated with cyclohexane. It was recrystallized and dried to give the title compound (NO. 13). Compound N0.14 (X=Br, R=NH 2 ): 3-bromo-N-{4-bromo-2-[2-(5-amino-1,3,4-oxadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide The title compound was obtained by the synthesis of the compound of Example No.13. Compound N0.31 (X=CL, R=CN): 3-bromo-N-{4-chloro-2-[2-(5-cyano-1,3,4-oxadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
取 2.0g( 4.2mmol )2-(3-溴小 (3-氯 -2-吡啶基 )-1 H-吡唑) -5-甲基 -4 H-3,1-苯并噁嗪 -4-酮 (Ml ) 悬浮于 40 mL DMF中, 加入 0.8g CNCONHNH2, 室温反应过夜, 加入对甲苯磺酸 l.Og和 20 mL 甲苯, 加热至回流反应 4h, 脱溶后用环己垸重结晶, 干燥得相应的标题化合 物 (N0.31 ) 1.2g。 化合物 N0.32 (X=CL, R=CL) : 3-溴 -N-{4-氯 -2-[2-(5-氯 -1,3,4-噁二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Take 2.0 g (4.2 mmol) of 2-(3-bromo small (3-chloro-2-pyridyl)-1 H-pyrazole)-5-methyl-4 H-3,1-benzoxazine-4 - Ketone (Ml) was suspended in 40 mL of DMF, added with 0.8 g of CNCONHNH 2 , reacted at room temperature overnight, added with 1.0 g of p-toluenesulfonic acid and 20 mL of toluene, heated to reflux for 4 h, desolvated and recrystallized from cyclohexane. The title compound (N0.31) 1.2 g was obtained. Compound N0.32 (X=CL, R=CL): 3-bromo-N-{4-chloro-2-[2-(5-chloro-1,3,4-oxadiazole)]-6- Synthesis of phenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.31的合成方法, 将 Ml与 CLCONHNH2进行合成得相应的标题化合 物。 化合物 N0.33 (X=CL, R=SCN): 3-溴 -N-{4-氯 -2-[2-(5-硫氰基 -1,3,4-噁二唑 )]-6-甲基 苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 M1 was synthesized with CLCONHNH 2 according to the procedure of the compound of Example N0.31 to give the corresponding title compound. Compound N0.33 (X=CL, R=SCN): 3-bromo-N-{4-chloro-2-[2-(5-thiocyano-1,3,4-oxadiazole)]-6 Synthesis of -methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
取 2.0g( 3.8mmol ) 3-溴 -N-{4-氯 -2-[2-(5-氯 -1,3,4-噁二唑 )]-6-甲基苯基}-1-(3-氯 -2-吡啶 基) -1H-吡唑 -5-甲酰胺悬浮于 20 mL DMF中, 加入 0.5g硫氰化钠, 室温反应过夜,, 减压 脱溶后, 水洗, 过滤, 环己垸重结晶, 干燥, 得相应的标题化合物 1.3g。 化合物 N0.47 (X=Br, R=CN) : 3-溴 -N-{4-溴 -2-[2-(5-氰基 -1,3,4-噁二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Take 2.0 g (3.8 mmol) of 3-bromo-N-{4-chloro-2-[2-(5-chloro-1,3,4-oxadiazole)]-6-methylphenyl}-1- (3-Chloro-2-pyridyl)-1H-pyrazole-5-carboxamide was suspended in 20 mL of DMF, added with 0.5 g of sodium thiocyanate, reacted at room temperature overnight, desolvated under reduced pressure, washed with water, filtered. The Cyclohexanide was recrystallized and dried to give the corresponding title compound (1.3 g). Compound N0.47 (X=Br, R=CN): 3-bromo-N-{4-bromo-2-[2-(5-cyano-1,3,4-oxadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.31的合成方法, 将 Ml与 CNCONHNH2进行合成得相应的标题化合 物。 化合物 N0.48 (X=CN, R=SCN) : 3-溴 -N-{4-氰基 -2-[2-(5-硫氰基 -1,3,4-噁二唑 )]-6- 甲基苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 M1 was synthesized with CNCONHNH 2 according to the procedure of the compound of Example N0.31 to give the corresponding title compound. Compound N0.48 (X=CN, R=SCN): 3-bromo-N-{4-cyano-2-[2-(5-thiocyano-1,3,4-oxadiazole)]- Synthesis of 6-methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
取 2.0g( 3.7mmol ) 3-溴 -N-{4-氰基 -2-[2-(5-氯 -1,3,4-噁二唑 )]-6-甲基苯基}-1-(3-氯 -2-吡 啶基) -1H-吡唑 -5-甲酰胺悬浮于 20 mL DMF中, 加入 0.5g硫氰化钠, 室温反应过夜,, 减 压脱溶后, 水洗, 过滤, 环己垸重结晶, 干燥, 得相应的标题化合物 1.2g。 实施例 8 Take 2.0 g (3.7 mmol) of 3-bromo-N-{4-cyano-2-[2-(5-chloro-1,3,4-oxadiazole)]-6-methylphenyl}-1 -(3-Chloro-2-pyridyl)-1H-pyrazole-5-carboxamide was suspended in 20 mL of DMF, added with 0.5 g of sodium thiocyanate, allowed to react at room temperature overnight, desolvated under reduced pressure, washed with water, filtered The Cyclohexanide was recrystallized and dried to give the corresponding title compound. Example 8
反应通 Reaction
1 5 1 6 化合物 N0.15(X=CL,Z=0,R2=CH3 ): 3-溴 -N-{4-氯 -2-[2-(5-甲氨基 -1,3,4-噁二唑 )]-6- 甲基苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 1 5 1 6 Compound N0.15 (X=CL, Z=0, R 2 =CH 3 ): 3-bromo-N-{4-chloro-2-[2-(5-methylamino-1,3, Synthesis of 4-oxadiazole)]-6-methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
将2.0 § ( 411111101) 1 5 溶于 5 mL 10%的氢氧化钠溶液中,然后加入含碘甲垸 0.4 mL (6.0 mmol) 乙醇溶液 20 mL。 室温反应 10 h后倾入 50 mL水中, 搅拌 2 h后静置, 过滤, 水洗, 乙醇重结晶后干燥得相应的标题化合物 1.4 化合物 N0.16(X=CL,Z=S,R2=CH3 ): 3-溴 -N-{4-氯 -2-[2-(5-甲氨基 -1,3,4-噻二唑 )]-6- 甲基苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Dissolve 2.0 § ( 411111101) 1 5 in 5 mL of 10% sodium hydroxide solution, then add 20 mL of 0.4 mL (6.0 mmol) ethanol solution containing methyl iodide. After reacting at room temperature for 10 h, pour into 50 mL of water, stir for 2 h, then let stand, filter, wash with water, recrystallize from ethanol and dry to give the corresponding title compound 1.4 Compound N0.16 (X=CL, Z=S, R 2 =CH 3 ): 3-bromo-N-{4-chloro-2-[2-(5-methylamino-1,3,4-thiadiazole)]-6-methylphenyl}-1-(3- Synthesis of Chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.15的合成方法进行合成得相应的标题化合物。 化合物 N0.17 ( (X=CL, Z=0, R2=S02CH3 ): 3-溴 -N-{4-氯 -2-[2-(5-甲磺酰氨基 -1,3,4- 噁二唑 )]-6-甲基苯基 }-1-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 The title compound was obtained by the synthesis of the compound of Example N0.15. Compound N0.17 ((X=CL, Z=0, R 2 =S0 2 CH 3 ): 3-bromo-N-{4-chloro-2-[2-(5-methanesulfonylamino-1,3) ,4-oxadiazole)]-6-methylphenyl}-1-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
将 2.0 g (4mmol) I 5 溶于 20 mL甲苯禾口 lmL三乙胺溶液中, 然后加入 0.7g(6mmol)甲 基磺酰氯, 室温反应 2h后倾入 50 mL水中, 搅拌后静置, 分去水层, 减压蒸去溶剂, 乙醇 重结晶后干燥得相应的标题化合物 (N0.17) 1.8 化合物 N0.18 ( (X=CL, Z=S, R2=S02CH3 ): 3-溴 -N-{4-氯 -2-[2-(5-甲磺酰氨基 -1,3,4- 噻二唑 )]-6-甲基苯基 }-1-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 Dissolve 2.0 g (4 mmol) of I 5 in 20 mL of toluene and 1 mL of triethylamine solution, then add 0.7 g (6 mmol) of methylsulfonyl chloride, react at room temperature for 2 h, pour into 50 mL of water, stir, and let stand. the aqueous layer, after the solvent was distilled off under reduced pressure, dried and recrystallized from ethanol to give the corresponding title compound (N0.17) 1.8 compound N0.18 ((X = CL, Z = S, R 2 = S0 2 CH 3): 3 -Bromo-N-{4-chloro-2-[2-(5-methanesulfonylamino-1,3,4-thiadiazole)]-6-methylphenyl}-1-(3-chloro- Synthesis of 2-pyridyl)-1Η-pyrazole-5-carboxamide
按实施例化合物 N0.17的合成方法进行合成得相应的标题化合物。 化合物 NO.21 ( (X=CL , Z=0, R2=COCH3 ): 3-溴 -N-{4-氯 -2-[2-(5乙酰酰氨基 -1,3,4- 噁二唑 )]-6-甲基苯基 }-l-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 The title compound was obtained by the synthesis of the compound of Example N0.17. Compound NO.21 ((X=CL, Z=0, R 2 =COCH 3 ): 3-bromo-N-{4-chloro-2-[2-(5acetamido-1,3,4-oxine) Synthesis of oxazolidine]--6-methylphenyl}-l-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
将 2.0 g (4mmol) 1 5 溶于 20 mL甲苯禾 mL三乙胺溶液中, 然后加入 0.47g(6mmol)乙 酰氯, 室温反应 2h后倾入 50 mL水中, 搅拌后静置, 分去水层, 减压蒸去溶剂, 乙醇重结 晶后干燥得相应的标题化合物 (N0.21 ) 1.8 go 化合物 N0.22 ( (X=CL, Z=S, R2=COCH3 ): 3-溴 -N-{4-氯 -2-[2-(5-乙酰酰氨基 -1,3,4- 噻二唑 )]-6-甲基苯基 }-1-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 Dissolve 2.0 g (4 mmol) of 1 5 in 20 mL of toluene mL triethylamine solution, then add 0.47 g (6 mmol) of acetyl chloride, react at room temperature for 2 h, pour into 50 mL of water, stir, and let stand, remove the water layer. , evaporated and dried under reduced pressure to give the corresponding the solvent, the title compound was recrystallized from ethanol (N0.21) 1.8 go compound N0.22 ((X = CL, Z = S, R 2 = COCH 3): 3- bromo -N -{4-Chloro-2-[2-(5-acetylamido-1,3,4-thiadiazole)]-6-methylphenyl}-1-(3-chloro-2-pyridyl) Synthesis of -1Η-pyrazole-5-carboxamide
按上述化合物 N0.21的合成方法进行合成得相应的标题化合物。 实施例 9 The title compound is obtained by the synthesis of the above compound N0.21. Example 9
反应通式: Reaction formula:
化合物 N0.19 ( (X=CL, Z=0, R2= R3=CH3): 3-溴 -N-{4-氯 -2-[2-(5-二甲氨基 -1,3,4- 噁二唑 )]-6-甲基苯基 }-1-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 Compound N0.19 ((X=CL, Z=0, R 2 = R 3 =CH 3 ): 3-bromo-N-{4-chloro-2-[2-(5-dimethylamino-1,3) ,4-oxadiazole)]-6-methylphenyl}-1-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
将 2.0 g (4mmol) 1 5 加入 10 mL30%甲醛和 10mL甲酸溶液中, 回流反应 5h, 冷却后 加入 10%氢氧化钠水溶液至 pH=10,过滤,水洗涤,干燥,得到相应的标题化合物(N0.19) 1.6 g。 化合物 NO.20 ( (X=CL, Z=S, R^ R^CHs): 3-溴 -N-{4-氯 -2-[2-(5-二甲氨基 -1,3,4- 噻二唑 )]-6-甲基苯基 }-1-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 2.0 g (4 mmol) 1 5 was added to 10 mL of 30% formaldehyde and 10 mL of formic acid solution, and the reaction was refluxed for 5 h. After cooling, 10% aqueous sodium hydroxide solution was added to pH=10, filtered, washed with water and dried to give the title compound ( N0.19) 1.6 g. Compound NO.20 ((X=CL, Z=S, R^ R^CHs): 3-bromo-N-{4-chloro-2-[2-(5-dimethylamino-1,3,4- Synthesis of thiadiazole]]-6-methylphenyl}-1-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
按实施例化合物 N0.19的合成方法进行合成得相应的标题化合物。 The title compound was obtained by the synthesis of the compound of Example N0.19.
化合物 N0.34 (X=CL, R=CN): 3-溴 -N-{4-氯 -2-[2-(5-氰基 -1,3,4-噻二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 Compound N0.34 (X=CL, R=CN): 3-bromo-N-{4-chloro-2-[2-(5-cyano-1,3,4-thiadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
取 2.0g( 4.2mmol )2-(3-溴 -1-(3-氯 -2-吡啶基 )-1 H-吡唑) -5-甲基 -4 H-3,1-苯并噁嗪 -4-酮 (Ml ) 悬浮于 40 mL DMF中, 力口入 0.5gCNC=SNHNH2, 室温反应过夜, 加入对甲苯磺 酸 l.Og和 20 mL 甲苯, 加热至回流反应 4h, 脱溶后,水洗, 过滤, 用环己垸重结晶, 干 燥, 得相应的标题化合物 1.2g。 化合物 N0.35 (X=Br, R=CN) : 3-溴 -N-{4-溴 -2-[2-(5-氰基 -1,3,4-噻二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Take 2.0 g (4.2 mmol) of 2-(3-bromo-1-(3-chloro-2-pyridyl)-1 H-pyrazole)-5-methyl-4 H-3,1-benzoxazine -4-ketone (Ml) was suspended in 40 mL of DMF, and 0.5 g of CNC=SNHNH 2 was added to the solution. The reaction was carried out at room temperature overnight, and 1.0 g of p-toluenesulfonic acid and 20 mL of toluene were added, and heated to reflux for 4 h. After desolvation, It was washed with water, filtered, and then crystallised from hexanes, Compound N0.35 (X=Br, R=CN): 3-bromo-N-{4-bromo-2-[2-(5-cyano-1,3,4-thiadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.34的合成方法进行合成得相应的标题化合物。 The title compound was obtained by the synthesis of the compound of the compound N0.34.
化合物 N0.36 (X=CN, R=CL): 3-溴 -N-{4-氰基 -2-[2-(5-氯 -1,3,4-噻二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Compound N0.36 (X=CN, R=CL): 3-bromo-N-{4-cyano-2-[2-(5-chloro-1,3,4-thiadiazole)]-6- Synthesis of methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.34的合成方法,将 Ml与 CLC=SNHNH2进行合成,得相应的标题化 合物。 化合物 N0.37 (X=CL, R=CL) : 3-溴 -N-{4-氯 -2-[2-(5-氯 -1,3,4-噻二唑 )]-6-甲基苯 基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Synthesis of M1 and CLC=SNHNH 2 gave the corresponding title compound. Compound N0.37 (X=CL, R=CL): 3-bromo-N-{4-chloro-2-[2-(5-chloro-1,3,4-thiadiazole)]-6- Synthesis of phenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
按实施例化合物 N0.34的合成方法, 将 Ml与 CLCONHNH2进行合成得相应的标题化合 物。 化合物 N0.38 (X=CL, R=SCN): 3-溴 -N-{4-溴 -2-[2-(5-硫氰基 -1,3,4-噻二唑 )]-6-甲基 苯基 }-1-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 M1 was synthesized with CLCONHNH 2 to give the corresponding title compound. Compound N0.38 (X=CL, R=SCN): 3-bromo-N-{4-bromo-2-[2-(5-thiocyano-1,3,4-thiadiazole)]-6 Synthesis of -methylphenyl}-1-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
取 2.0g( 3.8mmol ) 3-溴 -N-{4-氯 -2-[2-(5-氯 -1,3,4-噻二唑 )]-6-甲基苯基}-1-(3-氯 -2-吡啶 基) -1H-吡唑 -5-甲酰胺悬浮于 20 mL DMF中, 加入 0.5g硫氰化钠, 室温反应过夜, 减压 脱溶后, 水洗, 过滤, 环己垸重结晶, 干燥, 得相应的标题化合物 1.0g。 实施例 11 Take 2.0 g (3.8 mmol) of 3-bromo-N-{4-chloro-2-[2-(5-chloro-1,3,4-thiadiazole)]-6-methylphenyl}-1- (3-Chloro-2-pyridinyl)-1H-pyrazole-5-carboxamide was suspended in 20 mL of DMF, added with 0.5 g of sodium thiocyanate, allowed to react at room temperature overnight, desolvated under reduced pressure, washed with water, filtered, and evaporated. The crystals were recrystallized and dried to give the corresponding title compound 1.0 g. Example 11
反应通式: Reaction formula:
化合物 N0.42 ( (X=Br , R =COCH3 ): 3-溴 -N-{4-溴 -2-[2-(5乙酰酰氨基 -1,2,4-噁二 睡)] -6-甲基苯基 }-1-(3-氯 -2-吡啶基 HH-吡唑 -5-甲酰胺的合成 Compound N0.42 ((X=Br , R =COCH3 ): 3-bromo-N-{4-bromo-2-[2-(5-acetylamido-1,2,4-carbazone)]-6 Synthesis of -methylphenyl}-1-(3-chloro-2-pyridylHH-pyrazole-5-carboxamide
将 2.2 g (4mmol) 18溶于 20 mL甲苯和 lmL三乙胺溶液中, 然后加入 0.47g(6mmol) 乙酰氯, 室温反应 2h后倾入 50 mL水中, 搅拌后静置, 分去水层, 减压蒸去溶剂, 乙醇 重结晶后干燥得相应的标题化合物 1.5 g o 化合物 N0.43 ( (X=CN , R^SOaCHs ): 3-溴 -N-{4-氰基 -2-[2-(5-甲磺酰氨基 -1,2,4- 噁二唑 )]-6-甲基苯基 }-1-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 2.2 g (4 mmol) 18 was dissolved in 20 mL of toluene and 1 mL of triethylamine solution, then 0.47 g (6 mmol) of acetyl chloride was added, reacted at room temperature for 2 h, poured into 50 mL of water, stirred, and allowed to stand, and the aqueous layer was separated. The solvent was evaporated under reduced pressure, and the residue was evaporated to crystalljjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjjj (5-Methanesulfonylamino-1,2,4-oxadiazole)]-6-methylphenyl}-1-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide Synthesis
将 2.0 g (4mmol) 18溶于 20 mL甲苯和 lmL三乙胺溶液中, 然后加入 0.7g(6mmol) 甲基磺酰氯, 室温反应 2h后倾入 50 mL水中, 搅拌后静置, 分去水层, 减压蒸去溶剂, 乙醇重结晶后干燥得相应的标题化合物 1.4 g。 化合物 N0.44(X=CN , R=NHCH3 ): 3-溴 -N-{4-氰基 -2-[2-(5-甲氨基 -1,2,4-噁二唑 )]-6- 甲基苯基 }-1-(3-氯 -2-吡啶基 )-1Η-吡唑 -5-甲酰胺的合成 Dissolve 2.0 g (4 mmol) of 18 in 20 mL of toluene and 1 mL of triethylamine solution, then add 0.7 g (6 mmol) of methylsulfonyl chloride, react at room temperature for 2 h, pour into 50 mL of water, stir, stand, and remove water. The solvent was evaporated under reduced pressure. Compound N0.44 (X=CN, R=NHCH 3 ): 3-bromo-N-{4-cyano-2-[2-(5-methylamino-1,2,4-oxadiazole)]- Synthesis of 6-Methylphenyl}-1-(3-chloro-2-pyridyl)-1Η-pyrazole-5-carboxamide
将 2.0 g (4mmol) I8溶于 5 mL 10%的氢氧化钠溶液中,然后加入含碘甲垸 0.4 mL(6.0 mmol) 乙醇溶液 20 mL。 室温反应 10 h后倾入 50 mL水中, 搅拌 2 h后静置, 过滤, 水 洗, 乙醇重结晶后干燥得相应的标题化合物 1.4 g。 各实施例所制得的化合物如表 1、 2所示: Dissolve 2.0 g (4 mmol) of I8 in 5 mL of 10% sodium hydroxide solution, then add 20 mL of 0.4 mL (6.0 mmol) ethanol solution containing methyl iodide. After reacting for 10 h at room temperature, it was poured into 50 mL of water, stirred for 2 h, then allowed to stand, filtered, washed with water, and recrystallized from ethanol to give the title compound 1.4 g. The compounds prepared in the examples are shown in Tables 1 and 2:
表 1 Table 1
通式 X z R General formula X z R
NO. 形貌 熔点 / V NO. Morphology Melting point / V
1 I H 0 CH3 白色粉末 258. 2-260. 11 IH 0 CH 3 white powder 258. 2-260. 1
2 I C1 0 CH3 白色粉末 202. 1-204. 22 I C1 0 CH 3 white powder 202. 1-204. 2
3 I C1 0 OH 浅黄色粉末 221. 9-224. 13 I C1 0 OH light yellow powder 221. 9-224. 1
4 I C1 0 SH 棕色粉末 169. 8-171. 64 I C1 0 SH Brown powder 169. 8-171. 6
5 I C1 0 SCH3 白色粉末 165. 2-167. 55 I C1 0 SCH 3 white powder 165. 2-167. 5
6 I C1 0 SCH (CH3) 2 白色粉末 230. 0-232. 56 I C1 0 SCH (CH 3 ) 2 white powder 230. 0-232. 5
7 I Br 0 SH 白色粉末 245. 5-248. 17 I Br 0 SH white powder 245. 5-248. 1
8 I Br 0 SCH3 白色粉末 192. 5-195. 38 I Br 0 SCH 3 white powder 192. 5-195. 3
9 I CI s SCH3 棕黄色粉末 119. 7-121. 19 I CI s SCH 3 brownish yellow powder 119. 7-121. 1
10 I CI s NH2 浅棕色粉末 317. 2-320. 910 I CI s NH 2 light brown powder 317. 2-320. 9
11 II CI CH3 白色粉末 172. 9-175. 511 II CI CH 3 white powder 172. 9-175. 5
12 II Br CH3 白色粉末 193. 5-196. 512 II Br CH 3 white powder 193. 5-196. 5
13 I CI 0 NH2 浅棕色粉末 255. 2-258. 013 I CI 0 NH 2 light brown powder 255. 2-258. 0
14 I Br 0 NH2 浅棕色粉末 278. 3-280. 114 I Br 0 NH 2 light brown powder 278. 3-280. 1
15 I CI 0 NHCH3 白色粉末 289. 5-293. 715 I CI 0 NHCH3 white powder 289. 5-293. 7
16 I CI s NHCH3 白色粉末 310. 0-313. 516 I CI s NHCH3 white powder 310. 0-313. 5
17 I CI 0 NHS02CH3 黄色粉末 254. 6-26017 I CI 0 NHS0 2 CH 3 yellow powder 254. 6-260
18 I CI s NHS02CH3 黄色粉末 273. 7-276. 518 I CI s NHS0 2 CH 3 yellow powder 273. 7-276. 5
19 I CI 0 N(CH3) 2 棕黄色粉末 310. 5-313. 7 19 I CI 0 N(CH 3 ) 2 brownish yellow powder 310. 5-313. 7
(He'ra)¾ -08 '(He 's) ^ίτ '(He ^)Z Z \ Z ¾ (He'ra)3⁄4 -08 '(He 's) ^ίτ '(He ^)Z Z \ Z 3⁄4
OVL - ςς-L '(Ηΐ L= Γ ^Ζί ΧηΖ '6. = 'Ρ)8/;ΖΛΗΪ ¾ l0.8- 8'(HI'S) 8.6 (9P-OSMa) OZ OVL - ςς-L '(Ηΐ L= Γ ^Ζί ΧηΖ '6. = 'Ρ)8/;ΖΛΗΪ 3⁄4 l0.8- 8'(HI'S) 8.6 ( 9 P-OSMa) OZ
(He'ra)½ -99T (He's)82'2'(He's)88'2'(Hrs)6r (H rra)2e'z.-ee'z. Hrs)27- (H ra)s9'z.-e8' (Hr9'7=r'p) -8'(Hrs)g8'6'(Hrs)gz.Oi(¾a3) 61 (He'ra)1⁄2 -99T (He's)82'2'(He's)88'2'(Hrs)6r (H rra)2e'z.-ee'z. Hrs)27- (H ra)s9'z .-e8' (Hr9'7=r'p) -8'(Hrs)g8'6'(Hrs)gz.Oi(3⁄4a3) 61
(He 's) '(He 's)£9 '(HZ ¾ (He 's) '(He 's)£9 '(HZ 3⁄4
WL - ZVL '(Ηΐ L= Γ 'P 'L'diZ '6. = 'P)9S.Z/(HI 'ra)78 — .8'(HI'S)09.6 (9P-OSMa) 81 WL - ZVL '(Ηΐ L= Γ 'P 'L'diZ '6. = 'P)9S.Z/(HI 'ra)78 — .8'(HI'S)09.6 ( 9 P-OSMa) 81
(He's)%'2'(He's)T8'2'(Hrs)6r (H ΐ¾ι)Κϋί7·/ (Ηΐ' ) ·ΖΛΐΚ¾ ·Δ— 6'Z/(Hr9 =fP)^-8'(Hrs)S6'6'(Hrs)8SOI(¾ D) LI (He's)%'2'(He's)T8'2'(Hrs)6r (H ΐ3⁄4ι)Κϋί7·/ (Ηΐ' ) ·ΖΛΐΚ3⁄4 ·Δ—6'Z/(Hr9 =fP)^-8'(Hrs) S6'6'(Hrs)8SOI(3⁄4 D) LI
WL -9YL '(Ηΐ L= Γ 'νΠί' L Z '6. = 'P)W(HI ¾ ί78ϋ0·8'(Ηΐ'») 6 (9P-OSMa) 91 WL -9YL '(Ηΐ L= Γ 'νΠί' LZ '6. = 'P)W(HI 3⁄4 ί78ϋ0·8'(Ηΐ'») 6 ( 9 P-OSMa) 91
SI SI
'(IK¾ W — ^ΖΛΗΐ¾ 9ΓΔ— 9 ΖΛΗΐ¾ ϋ9·ζ/(Ηΐ¾ι)ε9· — 00'8'(HI's)(y6(9P-OS]A[a) n '(IK3⁄4 W — ^ΖΛΗΐ3⁄4 9ΓΔ— 9 ΖΛΗΐ3⁄4 ϋ9·ζ/(Ηΐ3⁄4ι)ε9· — 00'8'(HI's)(y6( 9 P-OS]A[a) n
(IK's)0 '(IK's)Z Z/(H (IK's)0 '(IK's)Z Z/(H
¾ - — S9.Z/(HI'ra)9S — 68.ΖΛΗΐ¾ ·— 8 .8'(Ηΐ¾ .8— 87-8'(HI's)0r(H(9P-OS]A[a) £13⁄4 - — S9.Z/(HI'ra)9S — 68.ΖΛΗΐ3⁄4 ·— 8 .8'(Ηΐ3⁄4 .8— 87-8'(HI's)0r(H( 9 P-OS]A[a) £1
I'ra) — 67-z/(HI's)6 z (Hlt.8=r'P)98.Z/(Hrs)60.8'(Hr^=fP)97-8'(Hrs)0r(H(¾ D) Zl I'ra) — 67-z/(HI's)6 z (Hlt.8=r'P)98.Z/(Hrs)60.8'(Hr^=fP)97-8'(Hrs)0r(H(3(4) D) Zl
(He 's) LZZ '(He 's) £ςτ (He 's) LZZ '(He 's) £ςτ
'(Ηΐ 's) M L Z Zi. L-6V L XUZ^) m0.8'(m'ra) 8- 8'(HI's) 90Όΐ(¾ΧΟ) II ς9·ΖΛΗΐ¾ι)99ϋ6·ΖΛΗΐ¾ · — ζ ·8'(Ηΐ¾ι)^8— 89'8'(HI's)8 (H(9P-OS]A[a) 01'(Ηΐ 's) MLZ Zi. L-6V L XUZ^) m0.8'(m'ra) 8- 8'(HI's) 90Όΐ(3⁄4ΧΟ) II ς9·ΖΛΗΐ3⁄4ι)99ϋ6·ΖΛΗΐ3⁄4 · — ζ ·8' (Ηΐ3⁄4ι)^8— 89'8'(HI's)8 (H( 9 P-OS]A[a) 01
\UZ ¾ 90. - ZZ'L '(Ηΐ L= Γ ^ Z'L Z '6.L= I 'P) ' (Ηΐ ¾ ί78· — 00'8(9P-OS]A[a) 6 \UZ 3⁄4 90. - ZZ'L '(Ηΐ L= Γ ^ Z'L Z '6.L= I 'P) ' (Ηΐ 3⁄4 ί78· — 00'8( 9 P-OS]A[a) 6
Ηΐ'»)6ΐ.ΖΛΗΐ¾ κ.Δ— W/ (Hrs) .Z/(IK¾ SZ; — .Z/(Hr9.7=fP) .8'(Hrs)S6.6(¾ D) 8Ηΐ'»)6ΐ.ΖΛΗΐ3⁄4 κ.Δ— W/ (Hrs) .Z/(IK3⁄4 SZ; — .Z/(Hr9.7=fP) .8'(Hrs)S6.6(3⁄4 D) 8
.Z/(Hrs)7S.Z/(Hrs)7Z/Z/(Hr8. =r'P)98.Z/(Hrs)S6.Z/(Hlt.7=r'P) .8'(Hrs)00.6(9P-OS]A[a) L.Z/(Hrs)7S.Z/(Hrs)7Z/Z/(Hr8. =r'P)98.Z/(Hrs)S6.Z/(Hlt.7=r'P) .8'(Hrs )00.6( 9 P-OS]A[a) L
(ΙΚ^ θΓΐ— Sn'(IK¾ 06'I— 9 τ'(Ηΐ¾ 9ϋ6·ε'(Ηΐ¾ 9·9_(ΙΚ^ θΓΐ— Sn'(IK3⁄4 06'I— 9 τ'(Ηΐ3⁄4 9ϋ6·ε'(Ηΐ3⁄4 9·9_
98·9'(Ηΐ¾ κϋ ΖΛΗΐ'»)Ο ·ΖΛΐΚ¾ ¾·Δ— 88·Ζ/(Ηΐ'8·8ε=1·'Ρ) 0·8'(Ηΐ'»)8 8(9Ρ-Ο8]Α[α) 9 98·9'(Ηΐ3⁄4 κϋ ΖΛΗΐ'»)Ο ·ΖΛΐΚ3⁄4 3⁄4·Δ—88·Ζ/(Ηΐ'8·8ε=1·'Ρ) 0·8'(Ηΐ'»)8 8( 9 Ρ-Ο8 ]Α[α) 9
'(PK's)I8. '(Hrs)6rZ/(H s)8 z/(Hrs)IZ/ (Hr^=fP)S8.Z/(Hrs)97.8'(Hrs)76.6(¾ D) £ '(PK's)I8. '(Hrs)6rZ/(H s)8 z/(Hrs)IZ/ (Hr^=fP)S8.Z/(Hrs)97.8'(Hrs)76.6(3⁄4 D) £
's)07- (H ra)S9'Z.-2Z.- (Hrs)8r8'(Hrz.-rg-7=r'PP)0S'8'(Hrs)6S'8'(Hrs)0SOT(9P-OS]A[a) 's)07- (H ra)S9'Z.-2Z.- (Hrs)8r8'(Hrz.-rg-7=r'PP)0S'8'(Hrs)6S'8'(Hrs)0SOT( 9 P-OS]A[a)
(He 's) (He 's)
9Ζτ \m ¾ ii'L - i9.L '(Ηΐ 's) e0'8 '(Ηΐ 's) '(Ηΐ 's) Oi O\ '(Ηΐ 's) 9S 2l(9P-OS]A[a) £ 9Ζτ \m 3⁄4 ii'L - i9.L '(Ηΐ 's) e0'8 '(Ηΐ 's) '(Ηΐ 's) Oi O\ '(Ηΐ 's) 9S 2l( 9 P-OS]A [a) £
(Hrs)¾Or(Hrs)76'6'(zH9=f'HrP)0S'8 (Hrs)3⁄4Or(Hrs)76'6'(zH9=f'HrP)0S'8
'(H9=r'Hrp)z.r8'(zH9=r'Hrpp)6S'z.'(Hrs)e7- (HTs)z.e' (He's)gr (He's)68 (9p-os]A[a) Z '(H9=r'Hrp)z.r8'(zH9=r'Hrpp)6S'z.'(Hrs)e7- (HTs)ze'(He's)gr(He's)68 ( 9 p-os]A[ a) Z
(HI's)00O-'(IK's)9S (H (HI's)00O-'(IK's)9S (H
I I
(uidd) g (ΖΗΙΜ00£ H讓 Ht •ON (uidd) g (ΖΗΙΜ00£H let H t •ON
oz Oz
COCC.0/OlOZN3/X3d S.SS80/T10Z OAV (CDCl3)10.67(s,lH),9.93(s,lH),8.55(d,J=4.6,lH),8.02— 7.85(m,2H),7.62(s,lH),7.48-7.22(m,l H),7.19(s,lH),2.92(s,3H),2.31(s,3H) COCC.0/OlOZN3/X3d S.SS80/T10Z OAV (CDCl 3 ) 10.67 (s, lH), 9.93 (s, lH), 8.55 (d, J = 4.6, lH), 8.02 - 7.85 (m, 2H), 7.62 (s, lH), 7.48 - 7.22 (m , l H), 7.19 (s, lH), 2.92 (s, 3H), 2.31 (s, 3H)
(DMSO-d6) 9.80(s,lH),8.42— 7.94(m, lH),7.66(d, 7 =7.9, 2H),7.44(d, 7 =7.2, 1H), 7.53 - 7.11 (m, 2H), 2.74(s, 3H), 2.48 (s, 3H) (DMSO-d 6 ) 9.80 (s, lH), 8.42 - 7.94 (m, lH), 7.66 (d, 7 = 7.9, 2H), 7.44 (d, 7 = 7.2, 1H), 7.53 - 7.11 (m, 2H), 2.74(s, 3H), 2.48 (s, 3H)
(DMSO-d6)1.89(s,3H),2.15(s,3H),7.37(slH),7.43(s,lH),7.59(dd,lH,J=6Hz),8.17(d,lH,J=6H), 8.50(d,lH,J=6Hz),9.94(s,lH),10.23(s,lH) (DMSO-d6)10.50(s,lH),8.59(s,lH),8.50(dd,J=4.5,1.7,lH),8.18(s,lH),7.72— 7.65(m,2H),7.40(s, 1H),2.21(S,3H),1.82(S,1H) (DMSO-d 6 ) 1.89 (s, 3H), 2.15 (s, 3H), 7.37 (slH), 7.43 (s, lH), 7.59 (dd, lH, J = 6 Hz), 8.17 (d, lH, J =6H), 8.50 (d, lH, J = 6 Hz), 9.94 (s, lH), 10.23 (s, lH) (DMSO-d 6 ) 10.50 (s, lH), 8.59 (s, lH), 8.50 ( Dd, J = 4.5, 1.7, lH), 8.18 (s, lH), 7.72 - 7.65 (m, 2H), 7.40 (s, 1H), 2.21 (S, 3H), 1.82 (S, 1H)
(CDCl3)9.94(s,lH),8.46(s,lH),7.85(d,J=7.4,lH),7.71(s,lH),7.38(s,2H),7.19(s,lH),2.81(s,3H), 2.26(s,3H) (CDCl 3 ) 9.94 (s, lH), 8.46 (s, lH), 7.85 (d, J = 7.4, lH), 7.71 (s, lH), 7.38 (s, 2H), 7.19 (s, lH), 2.81(s,3H), 2.26(s,3H)
(CDCl3)9.94(s,lH),8.46(s,lH),7.85(d,J=7.4,lH),7.71(s,lH),7.38(s,2H),7.19(s,lH),2.81(s,3H), 2.26(s,3H) (CDCl 3 ) 9.94 (s, lH), 8.46 (s, lH), 7.85 (d, J = 7.4, lH), 7.71 (s, lH), 7.38 (s, 2H), 7.19 (s, lH), 2.81(s,3H), 2.26(s,3H)
(DMSO-d6)10.38(s,lH),8.68— 8.33(m,lH),8.27— 7.94(m,lH),7.92— 7.66(m,lH),7.65— 7.44(m,2 H),7.37(s,3H),2.18(s,3H) (DMSO-d 6 ) 10.38 (s, lH), 8.68 - 8.33 (m, lH), 8.27 - 7.94 (m, lH), 7.92 - 7.66 (m, lH), 7.65 - 7.44 (m, 2 H), 7.37(s,3H), 2.18(s,3H)
(DMSO-d6)10.38(s,lH),8.68— 8.33(m,lH),8.27— 7.94(m,lH),7.92— 7.66(m,lH),7.65— 7.44(m,2 H),7.37(s,3H),2.18(s,3H) (DMSO-d 6 ) 10.38 (s, lH), 8.68 - 8.33 (m, lH), 8.27 - 7.94 (m, lH), 7.92 - 7.66 (m, lH), 7.65 - 7.44 (m, 2 H), 7.37(s,3H), 2.18(s,3H)
(DMSO-d6) 10.50 (s, 1H), 8.64 (s, 1H), 8.13 (s, 1H), 7.75- 7.33 (m, 4H), 2.36 (s, 3H) (DMSO-d6)10.20(s,lH),8.58-8.13(m,lH),8.38-8.04(m,lH),7.93-7.58(m,lH),7.75-7.53(m,2 H),7.47(s,3H),2.40(s,3H) (DMSO-d 6 ) 10.50 (s, 1H), 8.64 (s, 1H), 8.13 (s, 1H), 7.75- 7.33 (m, 4H), 2.36 (s, 3H) (DMSO-d 6 ) 10.20 ( s, lH), 8.58-8.13 (m, lH), 8.38-8.04 (m, lH), 7.93-7.58 (m, lH), 7.75-7.53 (m, 2 H), 7.47 (s, 3H), 2.40 (s, 3H)
(DMSO-d6) 10.60 (s, 1H), 8.54 (s, 1H), 8.22 (s, 1H), 7.73 - 7.46 (m, 4H), 2.26 (s, 3H) (DMSO-d6)8.68(d,J=4.2,lH),8.15(s,lH),7.96(d,J=7.8,lH),7.84(s,lH),7.65(s,lH),7.31(s,lH), 2.56(s,3H) (DMSO-d 6 ) 10.60 (s, 1H), 8.54 (s, 1H), 8.22 (s, 1H), 7.73 - 7.46 (m, 4H), 2.26 (s, 3H) (DMSO-d 6 ) 8.68 ( d, J = 4.2, lH), 8.15 (s, lH), 7.96 (d, J = 7.8, lH), 7.84 (s, lH), 7.65 (s, lH), 7.31 (s, lH), 2.56 ( s, 3H)
(DMSO-d6) 10.40 (s, 1H), 8.64 (s, 1H), 8.32 (s, 1H), 7.85 - 7.53 (m, 4H), 2.56 (s, 3H) (DMSO-d6)8.55(d,J=4.2,lH),7.95(s,lH),7.86(d,J=7.8,lH),7.78(s,lH),7.64(s,lH),7.21(s,lH), 2.36(s,3H) (DMSO-d 6 ) 10.40 (s, 1H), 8.64 (s, 1H), 8.32 (s, 1H), 7.85 - 7.53 (m, 4H), 2.56 (s, 3H) (DMSO-d 6 ) 8.55 ( d, J = 4.2, lH), 7.95 (s, lH), 7.86 (d, J = 7.8, lH), 7.78 (s, lH), 7.64 (s, lH), 7.21 (s, lH), 2.36 ( s, 3H)
(DMSO-d6)8.85(d,J=4.2,lH),8.15(s,lH),7.89(d,J=7.8,lH),7.78(s,lH),7.64(s,lH),7.22(s,lH), 2.44(s,3H) (DMSO-d 6 ) 8.85 (d, J = 4.2, lH), 8.15 (s, lH), 7.89 (d, J = 7.8, lH), 7.78 (s, lH), 7.64 (s, lH), 7.22 (s,lH), 2.44(s,3H)
(DMSO-d6)8.75(d,J=4.2,lH),7.95(s,lH),7.86(d,J=7.8,lH),7.76(s,lH),7.64(s,lH),7.31(s,lH), 2.26(s,3H) (DMSO-d 6 ) 8.75 (d, J = 4.2, lH), 7.95 (s, lH), 7.86 (d, J = 7.8, lH), 7.76 (s, lH), 7.64 (s, lH), 7.31 (s,lH), 2.26(s,3H)
(DMSO-d6) 10.38 (s, 1H), 8.47 (s, 1H), 8.13 (s, 1H), 7.73 - 7.43 (m, 4H), 2.26 (s, 3H) (DMSO-d6) 10.50 (s, 1H), 8.54 (s, 1H), 8.13 (s, 1H), 7.63 - 7.33 (m, 4H), 2.35(s, 3H) (CDC13)10.56 (s,lH),8.65-8.44(m,lH),8.18-7.87 (m,2H), 7.55-7.38 (m, 2H), 7.21 (s, 1H), 2.63 (s, 3H), 2.30 (s, 3H) (DMSO-d 6 ) 10.38 (s, 1H), 8.47 (s, 1H), 8.13 (s, 1H), 7.73 - 7.43 (m, 4H), 2.26 (s, 3H) (DMSO-d 6 ) 10.50 ( s, 1H), 8.54 (s, 1H), 8.13 (s, 1H), 7.63 - 7.33 (m, 4H), 2.35(s, 3H) (CDC13) 10.56 (s, lH), 8.65-8.44 (m, lH), 8.18-7.87 (m, 2H), 7.55-7.38 (m, 2H), 7.21 (s, 1H), 2.63 (s, 3H), 2.30 (s, 3H)
(DMSO-d6)12.38 (s, 1H), 10.30 (s, 1H), 8.47 (s, 1H), 8.20 (s, 1H), 7.66 - 7.38 (m, 4H), 2.36 (s, 3H) (DMSO-d6) 12.38 (s, 1H), 10.30 (s, 1H), 8.47 (s, 1H), 8.20 (s, 1H), 7.66 - 7.38 (m, 4H), 2.36 (s, 3H)
(DMSO-d6)9.80(s,lH),8.83(d,J=4.2,lH),7.95(s,lH),7.86(d,J=7.8,lH),7.64(s,lH),7.54(s,lH),7 .ll(s,lH),2.38(s,3H) (DMSO-d6) 9.80 (s, lH), 8.83 (d, J = 4.2, 1H), 7.95 (s, lH), 7.86 (d, J = 7.8, lH), 7.64 (s, lH), 7.54 ( s,lH),7 .ll(s,lH), 2.38(s,3H)
(CDC13)10.88(s,lH),9.90(s,lH),8.65(d,J=4.6,lH),8.22- 7.86(m,2H),7.72(s,lH),7.46- 7.22(m,l H),7.19(s,lH),2.84(s,3H),2.31(s,3H) (CDC13) 10.88 (s, lH), 9.90 (s, lH), 8.65 (d, J = 4.6, lH), 8.22 - 7.86 (m, 2H), 7.72 (s, lH), 7.46 - 7.22 (m, l H), 7.19 (s, lH), 2.84 (s, 3H), 2.31 (s, 3H)
43 (DMSO-d6) 9.80(s,lH),8.42— 7.94(m, lH),7.66(d, J =7.9, 2H),7.30(d, J =7.2, 1H), 7.42 - 7.04 43 (DMSO-d6) 9.80 (s, lH), 8.42 - 7.94 (m, lH), 7.66 (d, J = 7.9, 2H), 7.30 (d, J = 7.2, 1H), 7.42 - 7.04
(m, 2H), 2.66(s, 3H), 2.48 (s, 3H) (m, 2H), 2.66(s, 3H), 2.48 (s, 3H)
44 (CDC13)10.38(s,lH),9.16(s,lH),8.25(s,lH),7.52(d,J=7.4,lH),7.73(s,lH),7.46(s,2H), 44 (CDC13) 10.38 (s, lH), 9.16 (s, lH), 8.25 (s, lH), 7.52 (d, J = 7.4, lH), 7.73 (s, lH), 7.46 (s, 2H),
7.21(s,lH),2.68(s,3H),2.20(s,3H) 7.21(s,lH), 2.68(s,3H), 2.20(s,3H)
45 (DMSO-d6) 10.81 (s, 1H), 8.74 (s, 1H), 8.03 (s, 1H), 7.95- 7.53 (m, 4H), 2.46 (s, 3H) 45 (DMSO-d6) 10.81 (s, 1H), 8.74 (s, 1H), 8.03 (s, 1H), 7.95- 7.53 (m, 4H), 2.46 (s, 3H)
46 (DMSO-d6) 10.42 (s, 1H), 8.53 (s, 1H), 8.10 (s, 1H), 7.75- 7.38 (m, 4H), 2.36 (s, 3H) 46 (DMSO-d6) 10.42 (s, 1H), 8.53 (s, 1H), 8.10 (s, 1H), 7.75- 7.38 (m, 4H), 2.36 (s, 3H)
47 (DMSO-d6)8.85(d,J=4.2,lH),7.65(s,lH),7.86(d,J=7.8,lH),7.76(s,lH),7.42(s,lH),7.31(s,lH),47 (DMSO-d6) 8.85 (d, J = 4.2, lH), 7.65 (s, lH), 7.86 (d, J = 7.8, lH), 7.76 (s, lH), 7.42 (s, lH), 7.31 (s,lH),
2.46(s,3H) 2.46(s,3H)
48 (DMSO-d6)8.55(d,J=4.2,lH),7.85(s,lH),7.86(d,J=7.8,lH),7.78(s,lH),7.64(s,lH),7.20(s,lH), 48 (DMSO-d6) 8.55 (d, J = 4.2, lH), 7.85 (s, lH), 7.86 (d, J = 7.8, lH), 7.78 (s, lH), 7.64 (s, lH), 7.20 (s,lH),
2.33(s,3H) 2.33(s,3H)
下面将叙述以本发明化合物为活性成分的制剂例, 所述的制剂例可用作农业、 园艺和 花卉栽培领域的杀虫剂。 但是, 本发明的实施方式并不限于以下内容。 制剂实施例 1: 水乳剂 An example of a preparation containing the compound of the present invention as an active ingredient, which is useful as an insecticide in the fields of agriculture, horticulture and flower cultivation, will be described below. However, embodiments of the invention are not limited to the following. Formulation Example 1: Water emulsion
将本发明化合物 20份、 甲苯 12份、 环氧乙垸-环氧丙垸嵌段共聚物 6份、 黄原胶 6份, 乙 二醇 /丙二醇复合防冻剂 8.5份, 有机硅 0.8份, 水 46.7份, 按水乳剂加工工艺得到活性成分 为 20%的水乳剂。 制剂实施例 2: 悬浮剂: 20 parts of the compound of the present invention, 12 parts of toluene, 6 parts of epoxy oxime-epoxypropene block copolymer, 6 parts of xanthan gum, 8.5 parts of ethylene glycol/propylene glycol composite antifreeze, 0.8 parts of silicone, water 46.7 parts, according to the water emulsion processing technology, an aqueous emulsion having an active ingredient of 20% was obtained. Formulation Example 2: Suspending agent:
将本发明化合物 25份、 润湿剂对甲基脂肪酰胺基苯磺酸钠 6份、 助悬剂垸基酚聚氧乙 烯醚甲醛缩合物 2份、 增粘剂羧甲基纤维素纳 6份、 防腐剂水杨酸钠 1份、 防冻剂丙二醇 2 份、 消泡剂硅油 1份, 水 57份, 按悬浮剂加工工艺得到活性成分为 25%的悬浮剂。 25 parts of the compound of the present invention, 6 parts of a wetting agent to sodium methyl fatty amide sulfonate, 2 parts of a suspending nonylphenol ethoxylate ether condensate, 6 parts of a tackifier carboxymethyl cellulose 1 part of preservative sodium salicylate, 2 parts of antifreeze propylene glycol, 1 part of antifoaming silicone oil, 57 parts of water, 25% suspension of active ingredient according to the suspension processing technology.
制剂实施例 3: 乳油 Formulation Example 3: Emulsifiable Oil
将本发明化合物 10 份、 二甲苯 40 份、 二甲基甲酰胺 35 份、 吐温 80乳化剂 15 份, 按浮油加工工艺得到活性成分为 10%的乳油。 制剂实施例 4: 水分散颗粒剂 10 parts of the compound of the present invention, 40 parts of xylene, 35 parts of dimethylformamide, and 15 parts of Tween 80 emulsifier were obtained by an oil slick processing technique to obtain an emulsifiable concentrate having an active ingredient of 10%. Formulation Example 4: Water-dispersible granules
将本发明化合物 80 份、 湿润剂 PO-EO嵌段聚醚 2份、 分散剂萘磺酸缩合物钠盐 10份、 崩解剂聚乙煅吡咯垸酮 1份、 硅藻土 7份, 按水分散颗粒剂加工工艺得到活性成份为 80%的 水分散颗粒剂。 下面将叙述以本发明化合物为活性成分的杀虫剂的试验例。但是, 本发明的实施方式 并不限于以下内容。 试验例 1: 对小菜蛾的杀虫效果 80 parts of the compound of the present invention, 2 parts of the wetting agent PO-EO block polyether, 10 parts of the dispersing agent naphthalenesulfonic acid condensate sodium salt, 1 part of the disintegrant polypyrrolidone, 7 parts of diatomaceous earth, The water-dispersible granule processing process gives an active ingredient of 80% Water-dispersible granules. Test examples of the insecticide using the compound of the present invention as an active ingredient will be described below. However, embodiments of the invention are not limited to the following. Test Example 1: Insecticidal effect on Plutella xylostella
选择 3龄幼虫, 采用浸叶饲喂法进行杀虫效果测试。 根据制剂实施例 3的组成方式, 将 本发明实施例的化合物分别制成杀虫剂。用纯净水将所得到的杀虫剂乳油进行稀释, 均匀 混合得到所需浓度的药液。选取苞菜, 清洗并晾干,用打孔器做成叶碟, 在药液中浸 10 秒 钟取出, 待自然晾干后装入培养皿内。 每皿接入小菜蛾 3龄幼虫 10头, 3次重复, 第 ld、 3d 调查死亡虫数, 统计死亡率, 评价其杀虫效果。 死亡率统计如下表。 The 3rd instar larvae were selected and tested for insecticidal effects using a leaf-fed feeding method. According to the constitution of Formulation Example 3, the compounds of the examples of the present invention were each made into an insecticide. The obtained insecticide emulsifiable concentrate is diluted with purified water and uniformly mixed to obtain a desired concentration of the chemical solution. Select amaranth, wash and dry, use a puncher to make a leaf dish, dip in the liquid for 10 seconds, and then dry it naturally and put it into the dish. Each dish was connected to P. xylostella 3 instar larvae 10 times, repeated 3 times, the number of dead insects was investigated in the first ld and 3d, the mortality was counted, and the insecticidal effect was evaluated. Mortality statistics are shown in the table below.
NO. 剂量 mg/L Id死亡率%> 2d死亡率%> 3d死亡率%> NO. dose mg/L Id mortality%> 2d mortality%> 3d mortality%>
1 100 0.00 10.00 20.001 100 0.00 10.00 20.00
2 100 0.00 40.00 100.002 100 0.00 40.00 100.00
3 100 19.05 76.19 90.483 100 19.05 76.19 90.48
4 100 0.00 65.00 100.004 100 0.00 65.00 100.00
5 100 100 100.00 100.005 100 100 100.00 100.00
6 100 4.76 80.95 100.006 100 4.76 80.95 100.00
7 100 5.26 68.42 94.747 100 5.26 68.42 94.74
8 100 0.00 80.95 47.628 100 0.00 80.95 47.62
9 100 0 0 50.009 100 0 0 50.00
10 100 31.82 72.73 100.0010 100 31.82 72.73 100.00
11 100 42.86 71.43 100.0011 100 42.86 71.43 100.00
12 100 9.52 85.71 100.0012 100 9.52 85.71 100.00
13 100 12.54 77.22 95.3313 100 12.54 77.22 95.33
14 100 7.00 53.25 82.3314 100 7.00 53.25 82.33
15 100 22.00 66.00 95.0015 100 22.00 66.00 95.00
16 100 18.13 57.32 90.1216 100 18.13 57.32 90.12
17 100 7.32 51.66 80.2317 100 7.32 51.66 80.23
18 100 9.44 60.23 87.6618 100 9.44 60.23 87.66
19 100 2.55 45.67 82.0019 100 2.55 45.67 82.00
20 100 4.88 55.45 88.22 21 100 8.53 32.55 80.1120 100 4.88 55.45 88.22 21 100 8.53 32.55 80.11
22 100 7.77 27.63 77.7522 100 7.77 27.63 77.75
23 100 18.55 46.23 95.1223 100 18.55 46.23 95.12
24 100 7.09 30.22 66.0624 100 7.09 30.22 66.06
25 100 5.22 37.73 64.2325 100 5.22 37.73 64.23
26 100 8.15 30.43 70.3226 100 8.15 30.43 70.32
27 100 21.01 57.66 96.3227 100 21.01 57.66 96.32
28 100 23.15 67.22 100.0028 100 23.15 67.22 100.00
29 100 12.53 55.11 82.2229 100 12.53 55.11 82.22
30 100 10.13 45. 66 80.7830 100 10.13 45. 66 80.78
31 100 5.34 34.53 66.0231 100 5.34 34.53 66.02
32 100 8.15 23.66 68.3432 100 8.15 23.66 68.34
33 100 15.17 33.33 78.6533 100 15.17 33.33 78.65
34 100 20.12 47.63 90.5534 100 20.12 47.63 90.55
35 100 18.45 57.72 92.5535 100 18.45 57.72 92.55
36 100 6.78 32.44 56.3036 100 6.78 32.44 56.30
37 100 8.78 27.83 45.0537 100 8.78 27.83 45.05
38 100 10.28 38.23 66.6838 100 10.28 38.23 66.68
39 100 4.55 27.52 70.6639 100 4.55 27.52 70.66
40 100 2.28 38.13 78.5440 100 2.28 38.13 78.54
41 100 7.52 18.78 67.0341 100 7.52 18.78 67.03
42 100 6.98 34.63 82.2442 100 6.98 34.63 82.24
43 100 11.66 29.15 87.8243 100 11.66 29.15 87.82
44 100 8.22 32.12 78.2344 100 8.22 32.12 78.23
45 100 5.52 17.66 48.9245 100 5.52 17.66 48.92
46 100 7.09 20.12 56.7346 100 7.09 20.12 56.73
47 100 9.55 28.44 73.1247 100 9.55 28.44 73.12
48 100 3.33 16.53 35.32 化合物 No.2、 No.4、 No.5、 No.6, No.l0、 No.l l、 No.12等在 lOOmg/L浓度下的第 3天 杀死率均达到 100% 。 试验例 2: 对蚕豆蚜的杀虫效果 选择室内连续饲养的蚕豆蚜 3日龄若虫, 采用浸渍法进行杀虫效果测试。 根据制剂实施例 3的组成方式, 将本发明的化合物分别制成杀虫剂。 用纯净水将所得 到的杀虫剂乳油进行稀释, 配制 400mg/L 的药液。 将 5头成蚜接种于 30~50mm高的蚕豆苗 上, 24h后去除成蚜, 每个蚕豆苗上约有 30头左右初产若蚜, 至第 3天得 3日龄若蚜, 沿茎 基部将蚕豆苗剪下, 记录若蚜基数。 将具若蚜的蚕豆苗在药液中浸 5s后取出, 插入装有 清水的小瓶中培养 (每一浓度重复三次)。 处理后移至观察室, 72h检查死活虫数, 统计 死亡率 (以蚜虫不能活动为死亡标准)。 化合物 No.2在 400mg/L浓度下第 3天的死亡率达 96.04%。 化合物?^0.35在40011¾/1^浓度下第3天的死亡率达95.16%。 48 100 3.33 16.53 35.32 Compound No.2, No.4, No.5, No.6, No.l0, No.ll, No.12, etc. The killing rate reached 100 at the 3rd day of 100mg/L. %. Test Example 2: Insecticidal effect on broad bean meal The 3 day old nymphs of Vicia faba, which were continuously raised in the room, were selected for the insecticidal effect test by the dipping method. According to the constitution of Formulation Example 3, the compounds of the present invention were each made into an insecticide. The obtained insecticide emulsifiable concentrate was diluted with purified water to prepare a 400 mg/L drug solution. Five scorpions were inoculated on 30~50mm high broad bean seedlings, and after 24h, they were removed into sorghum. About 30 heads of each of the broad bean seedlings were primilated, and the third day was 3 days old. The broad bean seedlings were cut at the base and the base number was recorded. The stalked broad bean seedlings were immersed in the liquid for 5 s, taken out, and inserted into a vial containing water (three times for each concentration). After treatment, it was moved to the observation room, and the number of dead and live insects was checked at 72 hours, and the mortality rate was counted (the aphid was unable to move as the death standard). Compound No. 2 had a mortality rate of 96.04% on day 3 at a concentration of 400 mg/L. The mortality of the compound ?^0.35 on the third day at a concentration of 400113⁄4/1 was 95.16%.
试验 3 : 对二化螟的杀螟效果 Trial 3: Killing effect on Chilo suppressalis
采用二化螟 3龄幼虫, 采用浸稻苗饲喂法进行杀虫效果测试。 根据制剂实施例 3的组成 方式, 将本发明实施例的化合物分别制成杀虫剂。用纯净水将所得到的杀虫剂乳油进行稀 释, 均匀混合得到所需浓度的药液。 选取稻苗, 分志 10株 /组, 在药液中浸 10 秒钟取出, 待自然晾干后装入指形管内。 每管接入二化螟 3龄幼虫 10头, 3次重复, 第 ld、 3d、 5d、 7d 调查死亡虫数, 统计死亡率, 评价其杀虫效果。 化合物 No. 11在处理后第 ld、 3d、 5d、 7d 调查死亡率分别为 16.67、 36.67、 43.33、 90.00%。 化合物 No. 34在处理后第 ld、 3d、 5d、 7d调查死亡率分别为 17.61、 34.61、 42.13、 91.01%。 The 3rd instar larvae of Chilo suppressalis were used, and the insecticidal effect test was carried out by using the rice seedling feeding method. According to the constitution of Formulation Example 3, the compounds of the examples of the present invention were each made into an insecticide. The obtained insecticide emulsifiable concentrate is diluted with purified water and uniformly mixed to obtain a desired concentration of the chemical solution. The rice seedlings were selected and divided into 10 strains/group, which were taken out in the liquid for 10 seconds, and then naturally dried to be placed in the finger tube. Each tube was connected to 10 larvae of the third instar larvae, and the larvae were counted three times. The number of dead insects was counted on the first ld, 3d, 5d, and 7d, and the mortality was evaluated to evaluate the insecticidal effect. Compound No. 11 had a mortality rate of 16.67, 36.67, 43.33, and 90.00% at the first ld, 3d, 5d, and 7d after treatment. Compound No. 34 had a mortality rate of 17.61, 34.61, 42.13, and 91.01% at ld, 3d, 5d, and 7d after treatment, respectively.
Claims
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201010018397.9 | 2010-01-15 | ||
| CN201010018397A CN101747325A (en) | 2010-01-15 | 2010-01-15 | Neighboring Heterocyclic formanilide compound and synthesis method and application thereof |
| CN201010178282.6 | 2010-05-19 | ||
| CN201010178282 | 2010-05-19 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2011085575A1 true WO2011085575A1 (en) | 2011-07-21 |
Family
ID=44303818
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2010/073303 Ceased WO2011085575A1 (en) | 2010-01-15 | 2010-05-27 | Ortho-heterocyclyl formanilide compounds, their synthesis methods and use |
| PCT/CN2011/070267 Ceased WO2011085684A1 (en) | 2010-01-15 | 2011-01-14 | O-heterocyclyl formanilide compounds, their synthesis methods and use |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2011/070267 Ceased WO2011085684A1 (en) | 2010-01-15 | 2011-01-14 | O-heterocyclyl formanilide compounds, their synthesis methods and use |
Country Status (1)
| Country | Link |
|---|---|
| WO (2) | WO2011085575A1 (en) |
Cited By (385)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013136073A1 (en) * | 2012-03-13 | 2013-09-19 | Redx Pharma Limited | Agricultural chemicals |
| WO2014053405A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Pesticidally active mixtures comprising anthranilamide compounds |
| WO2014053395A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Use of n-thio-anthranilamide compounds on cultivated plants |
| WO2014053403A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of controlling insecticide resistant insects |
| WO2014053404A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Pesticidally active mixtures comprising anthranilamide compounds |
| WO2014053401A2 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of improving plant health |
| WO2014053407A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2014053406A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of controlling ryanodine-modulator insecticide resistant insects |
| WO2014079764A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079813A1 (en) | 2012-11-23 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079774A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079752A1 (en) | 2012-11-23 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079772A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079773A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079770A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079766A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079814A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079820A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Use of anthranilamide compounds for reducing insect-vectored viral infections |
| WO2014079804A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079841A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014090700A1 (en) | 2012-12-14 | 2014-06-19 | Basf Se | Malononitrile compounds for controlling animal pests |
| WO2014102244A1 (en) | 2012-12-27 | 2014-07-03 | Basf Se | 2-(pyridin-3-yl)-5-hetaryl-thiazole compounds carrying an imine or imine-derived substituent for combating invertebrate pests |
| WO2014170300A1 (en) | 2013-04-19 | 2014-10-23 | Basf Se | N-substituted acyl-imino-pyridine compounds and derivatives for combating animal pests |
| WO2014202751A1 (en) | 2013-06-21 | 2014-12-24 | Basf Se | Methods for controlling pests in soybean |
| WO2015007682A1 (en) | 2013-07-15 | 2015-01-22 | Basf Se | Pesticide compounds |
| WO2015040116A1 (en) | 2013-09-19 | 2015-03-26 | Basf Se | N-acylimino heterocyclic compounds |
| WO2015055497A1 (en) | 2013-10-16 | 2015-04-23 | Basf Se | Substituted pesticidal pyrazole compounds |
| WO2015055757A1 (en) | 2013-10-18 | 2015-04-23 | Basf Se | Use of pesticidal active carboxamide derivative in soil and seed application and treatment methods |
| WO2015091649A1 (en) | 2013-12-18 | 2015-06-25 | Basf Se | N-substituted imino heterocyclic compounds |
| WO2015091645A1 (en) | 2013-12-18 | 2015-06-25 | Basf Se | Azole compounds carrying an imine-derived substituent |
| WO2015104422A1 (en) | 2014-01-13 | 2015-07-16 | Basf Se | Dihydrothiophene compounds for controlling invertebrate pests |
| WO2015162260A1 (en) * | 2014-04-25 | 2015-10-29 | Basf Se | Process for preparing anthranilamide esters and derivatives |
| WO2016071499A1 (en) | 2014-11-06 | 2016-05-12 | Basf Se | 3-pyridyl heterobicyclic compound for controlling invertebrate pests |
| WO2016128261A2 (en) | 2015-02-11 | 2016-08-18 | Basf Se | Pesticidal mixture comprising a pyrazole compound, an insecticide and a fungicide |
| WO2016162371A1 (en) | 2015-04-07 | 2016-10-13 | Basf Agrochemical Products B.V. | Use of an insecticidal carboxamide compound against pests on cultivated plants |
| WO2016198611A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino heterocyclic compounds |
| WO2016198613A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino compounds |
| WO2017016883A1 (en) | 2015-07-24 | 2017-02-02 | Basf Se | Process for preparation of cyclopentene compounds |
| WO2017072039A1 (en) | 2015-10-26 | 2017-05-04 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093163A1 (en) | 2015-11-30 | 2017-06-08 | Basf Se | Mixtures of cis-jasmone and bacillus amyloliquefaciens |
| WO2017093180A1 (en) | 2015-12-01 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093214A1 (en) | 2015-12-03 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Mesoionic halogenated 3-(acetyl)-1-[(1,3-thiazol-5-yl)methyl]-1h-imidazo[1,2-a]pyridin-4-ium-2-olate derivatives and related compounds as insecticides |
| WO2017137338A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituierted 2-(het)aryl-imidazolyl-carboxyamides as pest control agents |
| WO2017137339A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituted 2-oxyimidazolyl-carboxamides as pest control agents |
| EP3210468A1 (en) | 2016-02-26 | 2017-08-30 | Bayer CropScience Aktiengesellschaft | Solvent-free formulations of low-melting point agents |
| WO2017144341A1 (en) | 2016-02-23 | 2017-08-31 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017153217A1 (en) | 2016-03-09 | 2017-09-14 | Basf Se | Spirocyclic derivatives |
| WO2017153218A1 (en) | 2016-03-11 | 2017-09-14 | Basf Se | Method for controlling pests of plants |
| WO2017157885A1 (en) | 2016-03-16 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | N-(cyanobenzyl)-6-(cyclopropyl-carbonylamino)-4-(phenyl)-pyridine-2-carboxamide derivatives and related compounds as pesticides and plant protection agents |
| WO2017157735A1 (en) | 2016-03-15 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2017167832A1 (en) | 2016-04-01 | 2017-10-05 | Basf Se | Bicyclic compounds |
| WO2017174414A1 (en) | 2016-04-05 | 2017-10-12 | Bayer Cropscience Aktiengesellschaft | Naphthaline-derivatives as pest control agents |
| WO2017178416A1 (en) | 2016-04-15 | 2017-10-19 | Bayer Animal Health Gmbh | Pyrazolopyrimidine derivatives |
| WO2017186536A1 (en) | 2016-04-25 | 2017-11-02 | Bayer Cropscience Aktiengesellschaft | Substituted 2-alkylimidazolyl-carboxamides as pest control agents |
| EP3241830A1 (en) | 2016-05-04 | 2017-11-08 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pesticides |
| EP3245865A1 (en) | 2016-05-17 | 2017-11-22 | Bayer CropScience Aktiengesellschaft | Method for increasing yield in brassicaceae |
| WO2017198588A1 (en) | 2016-05-18 | 2017-11-23 | Basf Se | Capsules comprising benzylpropargylethers for use as nitrification inhibitors |
| WO2017198452A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in soybean |
| WO2017198454A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in cotton |
| WO2017198450A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in maize |
| WO2017198451A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in small grain cereals such as wheat and rice |
| WO2017198449A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in brassicaceae |
| WO2017198453A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in potato, tomato or alfalfa |
| WO2017198455A2 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in beta spp. plants |
| WO2018015289A1 (en) | 2016-07-19 | 2018-01-25 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018019937A1 (en) | 2016-07-29 | 2018-02-01 | Bayer Cropscience Aktiengesellschaft | Formulation comprising a beneficial p. bilaii strain and talc for use in seed treatment |
| WO2018029102A1 (en) | 2016-08-10 | 2018-02-15 | Bayer Cropscience Aktiengesellschaft | Substituted 2-heterocyclyl imidazolyl-carboxamides as pest control agents |
| EP3284739A1 (en) | 2017-07-19 | 2018-02-21 | Bayer CropScience Aktiengesellschaft | Substituted (het) aryl compounds as pesticides |
| WO2018033455A1 (en) | 2016-08-15 | 2018-02-22 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018050825A1 (en) | 2016-09-19 | 2018-03-22 | Bayer Cropscience Aktiengesellschaft | Pyrazolo [1,5-a]pyridine derivatives and their use as pesticides |
| EP3305786A2 (en) | 2018-01-22 | 2018-04-11 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pesticides |
| WO2018065292A1 (en) | 2016-10-06 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018065288A1 (en) | 2016-10-07 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-[2-phenyl-1-(sulfonyl-methyl)-vinyl]-imidazo-[4,5-b] pyridine derivatives and related compounds as pesticides in plant protection |
| WO2018083288A1 (en) | 2016-11-07 | 2018-05-11 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2018087036A1 (en) | 2016-11-11 | 2018-05-17 | Bayer Animal Health Gmbh | New anthelmintic quinoline-3-carboxamide derivatives |
| WO2018095953A1 (en) | 2016-11-23 | 2018-05-31 | Bayer Cropscience Aktiengesellschaft | 2-[3-(alkylsulfonyl)-2h-indazol-2-yl]-3h-imidazo[4,5-b]pyridine derivatives and similar compounds as pesticides |
| WO2018104500A1 (en) | 2016-12-09 | 2018-06-14 | Bayer Cropscience Aktiengesellschaft | Plant health effect of purpureocillium lilacinum |
| WO2018108730A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| WO2018108671A1 (en) | 2016-12-16 | 2018-06-21 | Basf Se | Pesticidal compounds |
| WO2018108791A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Thiadiazole derivatives as pesticides |
| WO2018130443A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018130437A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018138050A1 (en) | 2017-01-26 | 2018-08-02 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclene derivatives as pest control agents |
| WO2018141954A1 (en) | 2017-02-06 | 2018-08-09 | Bayer Aktiengesellschaft | Aryl or heteroaryl-substituted imidazo pyridine derivatives and their use as pesticides |
| EP3369320A1 (en) | 2017-03-02 | 2018-09-05 | Bayer CropScience Aktiengesellschaft | Agent for controlling bugs |
| WO2018162312A1 (en) | 2017-03-10 | 2018-09-13 | Basf Se | Spirocyclic derivatives |
| WO2018166855A1 (en) | 2017-03-16 | 2018-09-20 | Basf Se | Heterobicyclic substituted dihydroisoxazoles |
| WO2018177781A1 (en) | 2017-03-28 | 2018-10-04 | Basf Se | Pesticidal compounds |
| WO2018177970A1 (en) | 2017-03-31 | 2018-10-04 | Basf Se | Process for preparing chiral 2,3-dihydrothiazolo[3,2-a]pyrimidin-4-ium compounds |
| WO2018189077A1 (en) | 2017-04-12 | 2018-10-18 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| WO2018192872A1 (en) | 2017-04-21 | 2018-10-25 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2018192793A1 (en) | 2017-04-20 | 2018-10-25 | Basf Se | Substituted rhodanine derivatives |
| WO2018197692A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Aktiengesellschaft | Heteroarylphenylaminoquinolines and analogues |
| WO2018197466A1 (en) | 2017-04-26 | 2018-11-01 | Basf Se | Substituted succinimide derivatives as pesticides |
| WO2018197257A1 (en) | 2017-04-24 | 2018-11-01 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic-compound derivatives as pest control agents |
| WO2018197401A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Animal Health Gmbh | New bicyclic pyrazole derivatives |
| WO2018202524A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | 2-{[2-(phenyloxymethyl)pyridin-5-yl]oxy}-ethanamin-derivatives and related compounds as pest-control agents e.g. for the protection of plants |
| WO2018202501A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202494A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202712A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylphenoxyquinolines and analogues |
| WO2018202706A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylheteroaryloxyquinolines and analogues |
| WO2018202715A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylbenzylbenzimidazoles and analogues |
| WO2018202525A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | Phenoxyethanamine derivatives for controlling pests |
| WO2018206479A1 (en) | 2017-05-10 | 2018-11-15 | Basf Se | Bicyclic pesticidal compounds |
| US10149477B2 (en) | 2014-10-06 | 2018-12-11 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| WO2018224455A1 (en) | 2017-06-07 | 2018-12-13 | Basf Se | Substituted cyclopropyl derivatives |
| WO2018229202A1 (en) | 2017-06-16 | 2018-12-20 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2018234202A1 (en) | 2017-06-19 | 2018-12-27 | Basf Se | SUBSTITUTED PYRIMIDINIUM COMPOUNDS AND DERIVATIVES FOR CONTROLLING HARMFUL ANIMALS |
| WO2018234488A1 (en) | 2017-06-23 | 2018-12-27 | Basf Se | SUBSTITUTED CYCLOPROPYL DERIVATIVES |
| WO2019002132A1 (en) | 2017-06-30 | 2019-01-03 | Bayer Animal Health Gmbh | New azaquinoline derivatives |
| WO2019007887A1 (en) | 2017-07-06 | 2019-01-10 | Bayer Aktiengesellschaft | INSECTICIDES AND FUNGICIDES ACTIVE COMPOUNDS |
| WO2019025341A1 (en) | 2017-08-04 | 2019-02-07 | Bayer Animal Health Gmbh | Quinoline derivatives for treating infections with helminths |
| WO2019035881A1 (en) | 2017-08-17 | 2019-02-21 | Bayer Cropscience Lp | Liquid fertilizer-dispersible compositions and methods thereof |
| WO2019038195A1 (en) | 2017-08-22 | 2019-02-28 | Bayer Aktiengesellschaft | HETEROCYCLEN DERIVATIVES AS PESTICIDES |
| WO2019043183A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| EP3453706A1 (en) | 2017-09-08 | 2019-03-13 | Basf Se | Pesticidal imidazole compounds |
| WO2019059412A1 (en) | 2017-09-20 | 2019-03-28 | Mitsui Chemicals Agro, Inc. | Prolonged ectoparasite-controlling agent for animal |
| WO2019068572A1 (en) | 2017-10-04 | 2019-04-11 | Bayer Aktiengesellschaft | HETEROCYCLEN DERIVATIVES AS PESTICIDES |
| WO2019072906A1 (en) | 2017-10-13 | 2019-04-18 | Basf Se | Imidazolidine pyrimidinium compounds for combating animal pests |
| EP3473103A1 (en) | 2017-10-17 | 2019-04-24 | Bayer AG | Aqueous suspension concentrates based on 2- [(2,4-dichlorophenyl) -methyl] -4,4 '-dimethyl-3-isoxazolidinone |
| EP3473100A1 (en) | 2017-10-18 | 2019-04-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076754A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076751A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076752A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076749A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076750A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019092086A1 (en) | 2017-11-13 | 2019-05-16 | Bayer Aktiengesellschaft | Tetrazolylpropyl derivatives and their use as fungicides |
| WO2019105871A1 (en) | 2017-11-29 | 2019-06-06 | Bayer Aktiengesellschaft | Nitrogenous heterocycles as a pesticide |
| WO2019105875A1 (en) | 2017-11-28 | 2019-06-06 | Bayer Aktiengesellschaft | Heterocyclic compounds as pesticides |
| WO2019122319A1 (en) | 2017-12-21 | 2019-06-27 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylheteroaryloxyquinolines and analogues |
| WO2019121143A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Substituted cyclopropyl derivatives |
| WO2019121159A1 (en) | 2017-12-21 | 2019-06-27 | Basf Se | Pesticidal compounds |
| WO2019137995A1 (en) | 2018-01-11 | 2019-07-18 | Basf Se | Novel pyridazine compounds for controlling invertebrate pests |
| WO2019145140A1 (en) | 2018-01-09 | 2019-08-01 | Basf Se | Silylethynyl hetaryl compounds as nitrification inhibitors |
| WO2019155066A1 (en) | 2018-02-12 | 2019-08-15 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2019162174A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019162228A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | 1-(5-substituted imidazol-1-yl)but-3-en derivatives and their use as fungicides |
| WO2019166561A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of alkoxypyrazoles as nitrification inhibitors |
| WO2019166560A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of n-functionalized alkoxy pyrazole compounds as nitrification inhibitors |
| WO2019166558A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of pyrazole propargyl ethers as nitrification inhibitors |
| WO2019170626A1 (en) | 2018-03-08 | 2019-09-12 | Bayer Aktiengesellschaft | Use of heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides in plant protection |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019175046A1 (en) | 2018-03-12 | 2019-09-19 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| EP3545764A1 (en) | 2019-02-12 | 2019-10-02 | Bayer AG | Crystal form of 2-({2-fluoro-4-methyl-5-[(r)-(2,2,2-trifluoroethyl)sulfinyl]phenyl}imino)-3-(2,2,2- trifluoroethyl)-1,3-thiazolidin-4-one |
| WO2019185413A1 (en) | 2018-03-27 | 2019-10-03 | Basf Se | Pesticidal substituted cyclopropyl derivatives |
| WO2019197623A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, fungicidal and acaricidal properties |
| WO2019197371A1 (en) | 2018-04-10 | 2019-10-17 | Bayer Aktiengesellschaft | Oxadiazoline derivatives |
| WO2019197468A1 (en) | 2018-04-12 | 2019-10-17 | Bayer Aktiengesellschaft | N-(cyclopropylmethyl)-5-(methylsulfonyl)-n-{1-[1-(pyrimidin-2-yl)-1h-1,2,4-triazol-5-yl]ethyl}benzamide derivatives and the corresponding pyridine-carboxamide derivatives as pesticides |
| WO2019197615A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with fungicides, insecticides and acaricidal properties |
| WO2019201921A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2019201835A1 (en) | 2018-04-17 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019202077A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019206799A1 (en) | 2018-04-25 | 2019-10-31 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| EP3564225A1 (en) | 2019-03-21 | 2019-11-06 | Bayer Aktiengesellschaft | Crystalline form of spiromesifen |
| WO2019215182A1 (en) | 2018-05-09 | 2019-11-14 | Bayer Animal Health Gmbh | New quinoline derivatives |
| WO2019219529A1 (en) | 2018-05-15 | 2019-11-21 | Basf Se | Mixtures comprising benzpyrimoxan and oxazosulfyl and uses and methods of applying them |
| WO2019224092A1 (en) | 2018-05-22 | 2019-11-28 | Basf Se | Pesticidally active c15-derivatives of ginkgolides |
| WO2019224143A1 (en) | 2018-05-24 | 2019-11-28 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, nematicidal and acaricidal properties |
| EP3586630A1 (en) | 2018-06-28 | 2020-01-01 | Bayer AG | Active compound combinations having insecticidal/acaricidal properties |
| WO2020005678A1 (en) | 2018-06-25 | 2020-01-02 | Bayer Cropscience Lp | Seed treatment method |
| WO2020002472A1 (en) | 2018-06-28 | 2020-01-02 | Basf Se | Use of alkynylthiophenes as nitrification inhibitors |
| WO2020002189A1 (en) | 2018-06-27 | 2020-01-02 | Bayer Aktiengesellschaft | Active substance combinations |
| WO2020007902A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020020816A1 (en) | 2018-07-26 | 2020-01-30 | Bayer Aktiengesellschaft | Novel triazole derivatives |
| WO2020020777A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of substituted 2-thiazolines as nitrification inhibitors |
| WO2020020813A1 (en) | 2018-07-25 | 2020-01-30 | Bayer Aktiengesellschaft | Fungicidal active compound combinations |
| WO2020021082A1 (en) | 2018-07-27 | 2020-01-30 | Bayer Aktiengesellschaft | Controlled release formulations for agrochemicals |
| WO2020020765A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of a substituted thiazolidine compound as nitrification inhibitor |
| WO2020025650A1 (en) | 2018-07-31 | 2020-02-06 | Bayer Aktiengesellschaft | Controlled release formulations with lignin for agrochemicals |
| US10556844B2 (en) | 2015-02-06 | 2020-02-11 | Basf Se | Pyrazole compounds as nitrification inhibitors |
| EP3608311A1 (en) | 2019-06-28 | 2020-02-12 | Bayer AG | Crystalline form a of n-[4-chloro-3-[(1-cyanocyclopropyl)carbamoyl]phenyl]-2-methyl-4-methylsulfonyl-5-(1,1,2,2,2-pentafluoroethyl)pyrazole-3-carboxamide |
| EP3613736A1 (en) | 2018-08-22 | 2020-02-26 | Basf Se | Substituted glutarimide derivatives |
| WO2020043650A1 (en) | 2018-08-29 | 2020-03-05 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3620052A1 (en) | 2018-12-12 | 2020-03-11 | Bayer Aktiengesellschaft | Use of phenoxypyridinyl-substituted (1h-1,2,4-triazol-1-yl)alcohols for controlling fungicidal diseases in maize |
| WO2020053282A1 (en) | 2018-09-13 | 2020-03-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2020057939A1 (en) | 2018-09-17 | 2020-03-26 | Bayer Aktiengesellschaft | Use of the fungicide isoflucypram for controlling claviceps purpurea and reducing sclerotia in cereals |
| EP3628158A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| EP3628156A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method for controlling pests of sugarcane, citrus, rapeseed, and potato plants |
| EP3628157A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| WO2020064492A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling pests by seed treatment application of a mesoionic compound or mixture thereof |
| WO2020070050A1 (en) | 2018-10-01 | 2020-04-09 | Bayer Aktiengesellschaft | Fungicidal 5-substituted imidazol-1-yl carbinol derivatives |
| EP3636644A1 (en) | 2018-10-11 | 2020-04-15 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2020079173A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Pyridylphenylaminoquinolines and analogues |
| WO2020079232A1 (en) | 2018-10-20 | 2020-04-23 | Bayer Aktiengesellschaft | Oxetanylphenoxyquinolines and analogues |
| WO2020079167A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Heteroarylaminoquinolines and analogues |
| WO2020078839A1 (en) | 2018-10-16 | 2020-04-23 | Bayer Aktiengesellschaft | Active substance combinations |
| EP3643705A1 (en) | 2018-10-24 | 2020-04-29 | Basf Se | Pesticidal compounds |
| EP3643711A1 (en) | 2018-10-24 | 2020-04-29 | Bayer Animal Health GmbH | New anthelmintic compounds |
| WO2020109391A1 (en) | 2018-11-28 | 2020-06-04 | Bayer Aktiengesellschaft | Pyridazine (thio)amides as fungicidal compounds |
| WO2020109039A1 (en) | 2018-11-28 | 2020-06-04 | Basf Se | Pesticidal compounds |
| WO2020114932A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2020114934A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicide compositions |
| EP3669652A1 (en) | 2018-12-21 | 2020-06-24 | Bayer AG | Active compound combination |
| WO2020127780A1 (en) | 2018-12-20 | 2020-06-25 | Bayer Aktiengesellschaft | Heterocyclyl pyridazine as fungicidal compounds |
| WO2020126980A1 (en) | 2018-12-18 | 2020-06-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020127974A1 (en) | 2018-12-21 | 2020-06-25 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as new antifungal agents |
| WO2020126591A1 (en) | 2018-12-18 | 2020-06-25 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| EP3679791A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679792A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679790A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679793A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679789A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3696177A1 (en) | 2019-02-12 | 2020-08-19 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP3701796A1 (en) | 2019-08-08 | 2020-09-02 | Bayer AG | Active compound combinations |
| WO2020173860A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Fused bicyclic heterocycle derivatives as pesticides |
| WO2020173861A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2020178067A1 (en) | 2019-03-01 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020178307A1 (en) | 2019-03-05 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combination |
| EP3708565A1 (en) | 2020-03-04 | 2020-09-16 | Bayer AG | Pyrimidinyloxyphenylamidines and the use thereof as fungicides |
| WO2020182929A1 (en) | 2019-03-13 | 2020-09-17 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| WO2020187656A1 (en) | 2019-03-15 | 2020-09-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3725788A1 (en) | 2019-04-15 | 2020-10-21 | Bayer AG | Novel heteroaryl-substituted aminoalkyl azole compounds as pesticides |
| WO2020225438A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High uptake and rainfastness ulv formulations |
| WO2020225242A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Active compound combination |
| WO2020229398A1 (en) | 2019-05-14 | 2020-11-19 | Bayer Aktiengesellschaft | (1-alkenyl)-substituted pyrazoles and triazoles as pest control agents |
| WO2020231751A1 (en) | 2019-05-10 | 2020-11-19 | Bayer Cropscience Lp | Active compound combinations |
| WO2020239517A1 (en) | 2019-05-29 | 2020-12-03 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| EP3750888A1 (en) | 2019-06-12 | 2020-12-16 | Bayer Aktiengesellschaft | Crystalline form a of 1,4-dimethyl-2-[2-(pyridin-3-yl)-2h-indazol-5-yl]-1,2,4-triazolidine-3,5-dione |
| WO2020254492A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254490A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Phenoxyphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020254489A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Benzylphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020254486A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254494A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2020254487A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254493A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Thienylhydroxyisoxazolines and derivatives thereof |
| WO2020254488A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and use thereof as fungicides |
| WO2020263812A1 (en) | 2019-06-24 | 2020-12-30 | Auburn University | A bacillus strain and methods of its use for plant growth promotion |
| WO2021001331A1 (en) | 2019-07-03 | 2021-01-07 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides and derivatives thereof as microbicides |
| WO2021001273A1 (en) | 2019-07-04 | 2021-01-07 | Bayer Aktiengesellschaft | Herbicidal compositions |
| EP3766879A1 (en) | 2019-07-19 | 2021-01-20 | Basf Se | Pesticidal pyrazole derivatives |
| EP3769623A1 (en) | 2019-07-22 | 2021-01-27 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2021013719A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021013720A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021013721A1 (en) | 2019-07-22 | 2021-01-28 | Bayer Aktiengesellschaft | 5-amino substituted pyrazoles and triazoles as pest control agents |
| EP3771714A1 (en) | 2019-07-30 | 2021-02-03 | Bayer AG | Nitrogen-containing heterocycles as pesticides |
| WO2021018839A1 (en) | 2019-07-30 | 2021-02-04 | Bayer Animal Health Gmbh | Isoquinoline derivatives and their use for the treatment of parasitic infections |
| WO2021048188A1 (en) | 2019-09-11 | 2021-03-18 | Bayer Aktiengesellschaft | Highly effective formulations on the basis of 2-[(2,4-dichlorphenyl)-methyl]-4,4'-dimethyl-3-isoxazolidinones and preemergence herbicides |
| WO2021058659A1 (en) | 2019-09-26 | 2021-04-01 | Bayer Aktiengesellschaft | Rnai-mediated pest control |
| WO2021069569A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021069567A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021069575A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Animal Health Gmbh | Heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021089673A1 (en) | 2019-11-07 | 2021-05-14 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2021097162A1 (en) | 2019-11-13 | 2021-05-20 | Bayer Cropscience Lp | Beneficial combinations with paenibacillus |
| WO2021099303A1 (en) | 2019-11-18 | 2021-05-27 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021105091A1 (en) | 2019-11-25 | 2021-06-03 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021123051A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides, thiophene carboxylic acids and derivatives thereof |
| WO2021122986A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Thienyloxazolones and analogues |
| WO2021130143A1 (en) | 2019-12-23 | 2021-07-01 | Basf Se | Enzyme enhanced root uptake of agrochemical active compound |
| US11053175B2 (en) | 2015-05-12 | 2021-07-06 | Basf Se | Thioether compounds as nitrification inhibitors |
| EP3845304A1 (en) | 2019-12-30 | 2021-07-07 | Bayer AG | Capsule suspension concentrates based on polyisocyanates and biodegradable amine based cross-linker |
| EP3868207A1 (en) | 2020-02-24 | 2021-08-25 | Bayer Aktiengesellschaft | Encapsulated pyrethroids with improved activity in soil and leaf applications |
| WO2021165195A1 (en) | 2020-02-18 | 2021-08-26 | Bayer Aktiengesellschaft | Heteroaryl-triazole compounds as pesticides |
| WO2021170463A1 (en) | 2020-02-28 | 2021-09-02 | BASF Agro B.V. | Methods and uses of a mixture comprising alpha-cypermethrin and dinotefuran for controlling invertebrate pests in turf |
| US11142514B2 (en) | 2015-10-02 | 2021-10-12 | Basf Se | Imino compounds with a 2-chloropyrimidin-5-yl substituent as pest-control agents |
| WO2021204930A1 (en) | 2020-04-09 | 2021-10-14 | Bayer Animal Health Gmbh | Substituted condensed azines as anthelmintic compounds |
| WO2021209366A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209368A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209490A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Cyclaminephenylaminoquinolines as fungicides |
| WO2021209363A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209364A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209365A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021213978A1 (en) | 2020-04-21 | 2021-10-28 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocyclic derivatives as pest control agents |
| WO2021219513A1 (en) | 2020-04-28 | 2021-11-04 | Basf Se | Pesticidal compounds |
| WO2021224323A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021224220A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Pyridine (thio)amides as fungicidal compounds |
| EP3909950A1 (en) | 2020-05-13 | 2021-11-17 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2021228734A1 (en) | 2020-05-12 | 2021-11-18 | Bayer Aktiengesellschaft | Triazine and pyrimidine (thio)amides as fungicidal compounds |
| WO2021233861A1 (en) | 2020-05-19 | 2021-11-25 | Bayer Aktiengesellschaft | Azabicyclic(thio)amides as fungicidal compounds |
| EP3915371A1 (en) | 2020-11-04 | 2021-12-01 | Bayer AG | Active compound combinations and fungicide compositions comprising those |
| EP3915971A1 (en) | 2020-12-16 | 2021-12-01 | Bayer Aktiengesellschaft | Phenyl-s(o)n-phenylamidines and the use thereof as fungicides |
| WO2021245087A1 (en) | 2020-06-04 | 2021-12-09 | Bayer Aktiengesellschaft | Heterocyclyl pyrimidines and triazines as novel fungicides |
| WO2021249995A1 (en) | 2020-06-10 | 2021-12-16 | Bayer Aktiengesellschaft | Azabicyclyl-substituted heterocycles as fungicides |
| WO2021255170A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| WO2021255169A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| WO2021255091A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as fungicides |
| WO2021255089A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines and 1,3,4-oxadiazole pyridines as fungicides |
| WO2021255071A1 (en) | 2020-06-18 | 2021-12-23 | Bayer Aktiengesellschaft | 3-(pyridazin-4-yl)-5,6-dihydro-4h-1,2,4-oxadiazine derivatives as fungicides for crop protection |
| EP3929189A1 (en) | 2020-06-25 | 2021-12-29 | Bayer Animal Health GmbH | Novel heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021260017A1 (en) | 2020-06-26 | 2021-12-30 | Bayer Aktiengesellschaft | Aqueous capsule suspension concentrates comprising biodegradable ester groups |
| WO2022002818A1 (en) | 2020-07-02 | 2022-01-06 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2022033991A1 (en) | 2020-08-13 | 2022-02-17 | Bayer Aktiengesellschaft | 5-amino substituted triazoles as pest control agents |
| WO2022053453A1 (en) | 2020-09-09 | 2022-03-17 | Bayer Aktiengesellschaft | Azole carboxamide as pest control agents |
| WO2022058327A1 (en) | 2020-09-15 | 2022-03-24 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| EP3974414A1 (en) | 2020-09-25 | 2022-03-30 | Bayer AG | 5-amino substituted pyrazoles and triazoles as pesticides |
| EP3994988A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and rainfastness properties |
| EP3994985A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift properties |
| EP3994989A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, rainfastness and uptake properties |
| EP3994991A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading, uptake and rainfastness properties |
| EP3994987A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift and uptake properties |
| EP3994986A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and spreading properties |
| EP3994990A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and uptake properties |
| EP3994994A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| EP3994993A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading and ulv tank mix adjuvant formulation |
| EP3994992A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Low drift, rainfastness, high uptake and ulv tank mix adjuvant formulation |
| EP3994995A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| WO2022129196A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | Heterobicycle substituted 1,2,4-oxadiazoles as fungicides |
| WO2022129188A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | 1,2,4-oxadiazol-3-yl pyrimidines as fungicides |
| WO2022129190A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | (hetero)aryl substituted 1,2,4-oxadiazoles as fungicides |
| WO2022152728A1 (en) | 2021-01-15 | 2022-07-21 | Bayer Aktiengesellschaft | Herbicidal compositions |
| EP4036083A1 (en) | 2021-02-02 | 2022-08-03 | Bayer Aktiengesellschaft | 5-oxy substituted heterocycles as pesticides |
| WO2022167488A1 (en) | 2021-02-02 | 2022-08-11 | Basf Se | Synergistic action of dcd and alkoxypyrazoles as nitrification inhibitors |
| EP4043444A1 (en) | 2021-02-11 | 2022-08-17 | Basf Se | Substituted isoxazoline derivatives |
| WO2022207496A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2022207494A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| US11492346B2 (en) | 2019-06-18 | 2022-11-08 | Pfizer Inc. | Benzisoxazole sulfonamide derivatives |
| WO2022233777A1 (en) | 2021-05-06 | 2022-11-10 | Bayer Aktiengesellschaft | Alkylamide substituted, annulated imidazoles and use thereof as insecticides |
| WO2022238391A1 (en) | 2021-05-12 | 2022-11-17 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocycle derivatives as pest control agents |
| WO2022238194A1 (en) | 2021-05-10 | 2022-11-17 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2022243521A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of ethynylpyridine compounds as nitrification inhibitors |
| WO2022243523A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of an n-functionalized alkoxy pyrazole compound as nitrification inhibitor |
| WO2022268810A1 (en) | 2021-06-21 | 2022-12-29 | Basf Se | Metal-organic frameworks with pyrazole-based building blocks |
| EP4119547A1 (en) | 2021-07-12 | 2023-01-18 | Basf Se | Triazole compounds for the control of invertebrate pests |
| WO2023017120A1 (en) | 2021-08-13 | 2023-02-16 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| EP4140986A1 (en) | 2021-08-23 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4140995A1 (en) | 2021-08-27 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| WO2023025682A1 (en) | 2021-08-25 | 2023-03-02 | Bayer Aktiengesellschaft | Novel pyrazinyl-triazole compounds as pesticides |
| EP4144739A1 (en) | 2021-09-02 | 2023-03-08 | Bayer Aktiengesellschaft | Anellated pyrazoles as parasiticides |
| EP4148052A1 (en) | 2021-09-09 | 2023-03-15 | Bayer Animal Health GmbH | New quinoline derivatives |
| EP4151631A1 (en) | 2021-09-20 | 2023-03-22 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2023078915A1 (en) | 2021-11-03 | 2023-05-11 | Bayer Aktiengesellschaft | Bis(hetero)aryl thioether (thio)amides as fungicidal compounds |
| WO2023092050A1 (en) | 2021-11-20 | 2023-05-25 | Bayer Cropscience Lp | Beneficial combinations with recombinant bacillus cells expressing a serine protease |
| WO2023099445A1 (en) | 2021-11-30 | 2023-06-08 | Bayer Aktiengesellschaft | Bis(hetero)aryl thioether oxadiazines as fungicidal compounds |
| EP4194453A1 (en) | 2021-12-08 | 2023-06-14 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4198023A1 (en) | 2021-12-16 | 2023-06-21 | Basf Se | Pesticidally active thiosemicarbazone compounds |
| EP4198033A1 (en) | 2021-12-14 | 2023-06-21 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2023110656A1 (en) | 2021-12-15 | 2023-06-22 | Bayer Aktiengesellschaft | Spectroscopic solution for non-destructive quantification of one or more chemical substances in a matrix comprising coating and bulk material in a sample, such as coated seeds, using multivariate data analysis |
| EP4238971A1 (en) | 2022-03-02 | 2023-09-06 | Basf Se | Substituted isoxazoline derivatives |
| EP4265110A1 (en) | 2022-04-20 | 2023-10-25 | Bayer AG | Water dispersible granules with low melting active ingredients prepared by extrusion |
| WO2023203066A1 (en) | 2022-04-21 | 2023-10-26 | Basf Se | Synergistic action as nitrification inhibitors of dcd oligomers with alkoxypyrazole and its oligomers |
| WO2023205602A1 (en) | 2022-04-18 | 2023-10-26 | Basf Corporation | High-load agricultural formulations and methods of making same |
| WO2023208447A1 (en) | 2022-04-25 | 2023-11-02 | Basf Se | An emulsifiable concentrate having a (substituted) benzaldehyde-based solvent system |
| WO2023213626A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Use of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine for controlling unwanted microorganisms |
| WO2023213670A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Crystalline forms of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine |
| WO2023217619A1 (en) | 2022-05-07 | 2023-11-16 | Bayer Aktiengesellschaft | Low drift aqueous liquid formulations for low, medium, and high spray volume application |
| WO2023237444A1 (en) | 2022-06-06 | 2023-12-14 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| EP4295688A1 (en) | 2022-09-28 | 2023-12-27 | Bayer Aktiengesellschaft | Active compound combination |
| EP4295683A1 (en) | 2022-06-21 | 2023-12-27 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| WO2024013015A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024013016A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024028243A1 (en) | 2022-08-02 | 2024-02-08 | Basf Se | Pyrazolo pesticidal compounds |
| US11911372B2 (en) | 2018-06-28 | 2024-02-27 | Ctxt Pty Ltd | Compounds |
| EP4342885A1 (en) | 2022-09-20 | 2024-03-27 | Basf Se | N-(3-(aminomethyl)-phenyl)-5-(4-phenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-amine derivatives and similar compounds as pesticides |
| WO2024068517A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068519A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068473A1 (en) | 2022-09-27 | 2024-04-04 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2024068520A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068518A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-heteroaryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| EP4353082A1 (en) | 2022-10-14 | 2024-04-17 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024104643A1 (en) | 2022-11-17 | 2024-05-23 | Bayer Aktiengesellschaft | Use of isotianil for controlling plasmodiophora brassica |
| EP4389210A1 (en) | 2022-12-21 | 2024-06-26 | Basf Se | Heteroaryl compounds for the control of invertebrate pests |
| WO2024170472A1 (en) | 2023-02-16 | 2024-08-22 | Bayer Aktiengesellschaft | Herbicidal mixtures |
| WO2024213752A1 (en) | 2023-04-14 | 2024-10-17 | Elanco Animal Health Gmbh | Long-term prevention and/or treatment of a disease by slo-1 inhibitors |
| EP4455137A1 (en) | 2023-04-24 | 2024-10-30 | Basf Se | Pyrimidine compounds for the control of invertebrate pests |
| EP4467535A1 (en) | 2023-05-25 | 2024-11-27 | Basf Se | Lactam pesticidal compounds |
| EP4488269A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488273A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488270A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| WO2025026738A1 (en) | 2023-07-31 | 2025-02-06 | Bayer Aktiengesellschaft | 6-[5-(ethylsulfonyl)-1-methyl-1h-imidazol-4-yl]-7-methyl-3-(pentafluoroethyl)-7h-imidazo[4,5-c]pyridazine derivatives as pesticides |
| WO2025026787A1 (en) | 2023-08-01 | 2025-02-06 | Globachem Nv | Plant defense elicitors |
| WO2025026815A1 (en) | 2023-08-01 | 2025-02-06 | Globachem Nv | Insecticidal mixtures |
| WO2025031668A1 (en) | 2023-08-09 | 2025-02-13 | Bayer Aktiengesellschaft | Azaheterobiaryl-substituted 4,5-dihydro-1h-2,4,5-oxadiazines as novel fungicides |
| WO2025032038A1 (en) | 2023-08-09 | 2025-02-13 | Bayer Aktiengesellschaft | Pyridazin-4-yloxadiazines as novel fungicides |
| WO2025040301A1 (en) | 2023-08-21 | 2025-02-27 | Bayer Aktiengesellschaft | Herbicide/safener combinations based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy] acetic acids and herbicides from the class of substituted cyclic diones and the salts thereof |
| WO2025040520A1 (en) | 2023-08-21 | 2025-02-27 | Bayer Aktiengesellschaft | Herbicide/safener combinations based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy] acetic acids and herbicides from the class of substituted cyclic diones and the salts thereof |
| WO2025049555A1 (en) | 2023-08-31 | 2025-03-06 | Oerth Bio Llc | Compositions and methods for targeted inhibition and degradation of proteins in an insect cell |
| WO2025078128A1 (en) | 2023-10-11 | 2025-04-17 | Bayer Aktiengesellschaft | Pyridazin-3-one-4-yloxadiazines as novel fungicides |
| WO2025098876A1 (en) | 2023-11-10 | 2025-05-15 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2025098874A1 (en) | 2023-11-10 | 2025-05-15 | Bayer Aktiengesellschaft | Active compound combinations having fungicidal/insecticidal/acaricidal properties |
| WO2025098875A1 (en) | 2023-11-10 | 2025-05-15 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2025108865A1 (en) | 2023-11-23 | 2025-05-30 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2025114238A1 (en) | 2023-12-01 | 2025-06-05 | Bayer Aktiengesellschaft | Solid agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2- difluoroethyl)amino]furan-2(5h)-one |
| WO2025117659A1 (en) | 2023-11-29 | 2025-06-05 | Basf Corporation | Insecticide delivery by nanocarriers |
| WO2025119860A1 (en) | 2023-12-05 | 2025-06-12 | Bayer Aktiengesellschaft | Liquid agrochemical formulations comprising flupyradifurone |
| EP4574819A1 (en) | 2023-12-22 | 2025-06-25 | Basf Se | Diazinone compounds for the control of invertebrate pests |
| WO2025132148A1 (en) | 2023-12-21 | 2025-06-26 | Bayer Aktiengesellschaft | Adjuvant compositions for agrochemical applications |
| US12365655B2 (en) | 2018-06-20 | 2025-07-22 | Ctxt Pty Ltd | Compounds |
| WO2025168620A1 (en) | 2024-02-07 | 2025-08-14 | Bayer Aktiengesellschaft | Heteroaryl-substituted 4,5-dihydro-1h-2,4,5-oxadiazines as novel fungicides |
| WO2025168777A1 (en) | 2024-02-08 | 2025-08-14 | Bayer Aktiengesellschaft | Use of isoxazolinecarboxamide for selective weed control in pineapple |
| WO2025186065A1 (en) | 2024-03-05 | 2025-09-12 | Bayer Aktiengesellschaft | Heteroaryl-substituted (aza)quinoxaline derivatives as pesticides |
| WO2025190927A1 (en) | 2024-03-14 | 2025-09-18 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2025242699A1 (en) | 2024-05-22 | 2025-11-27 | Basf Se | Method for improving rainfastness of an agrochemical active ingredient |
| WO2025257122A1 (en) | 2024-06-12 | 2025-12-18 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2025257242A1 (en) | 2024-06-14 | 2025-12-18 | Bayer Aktiengesellschaft | Heterocyclyl-substituted aryl- and heteroarylpyrazines and salts thereof, and the use thereof as herbicidal active substances |
| WO2025257121A1 (en) | 2024-06-12 | 2025-12-18 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997030047A1 (en) * | 1996-02-17 | 1997-08-21 | Agrevo Uk Limited | Fungicidal 1,2,4-oxadiazoles and analogues |
| JP2001064268A (en) * | 1999-06-25 | 2001-03-13 | Nippon Nohyaku Co Ltd | Benzamide derivatives, agricultural and horticultural insecticides and methods of using the same |
| CN1543460A (en) * | 2001-08-15 | 2004-11-03 | ��Ļ���Ű˾ | Ortho-heterocyclic substituted aryl amides for controlling invertebrate pests |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101747325A (en) * | 2010-01-15 | 2010-06-23 | 江苏省农药研究所股份有限公司 | Neighboring Heterocyclic formanilide compound and synthesis method and application thereof |
-
2010
- 2010-05-27 WO PCT/CN2010/073303 patent/WO2011085575A1/en not_active Ceased
-
2011
- 2011-01-14 WO PCT/CN2011/070267 patent/WO2011085684A1/en not_active Ceased
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1997030047A1 (en) * | 1996-02-17 | 1997-08-21 | Agrevo Uk Limited | Fungicidal 1,2,4-oxadiazoles and analogues |
| JP2001064268A (en) * | 1999-06-25 | 2001-03-13 | Nippon Nohyaku Co Ltd | Benzamide derivatives, agricultural and horticultural insecticides and methods of using the same |
| CN1543460A (en) * | 2001-08-15 | 2004-11-03 | ��Ļ���Ű˾ | Ortho-heterocyclic substituted aryl amides for controlling invertebrate pests |
Cited By (439)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2013136073A1 (en) * | 2012-03-13 | 2013-09-19 | Redx Pharma Limited | Agricultural chemicals |
| WO2014053405A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Pesticidally active mixtures comprising anthranilamide compounds |
| WO2014053395A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Use of n-thio-anthranilamide compounds on cultivated plants |
| WO2014053403A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of controlling insecticide resistant insects |
| WO2014053404A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Pesticidally active mixtures comprising anthranilamide compounds |
| WO2014053401A2 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of improving plant health |
| WO2014053407A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2014053406A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of controlling ryanodine-modulator insecticide resistant insects |
| WO2014079814A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079766A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079774A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079841A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079772A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079773A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079770A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079804A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079764A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079820A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Use of anthranilamide compounds for reducing insect-vectored viral infections |
| WO2014079752A1 (en) | 2012-11-23 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079813A1 (en) | 2012-11-23 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| US10117430B2 (en) | 2012-12-14 | 2018-11-06 | Basf Se | Malononitrile compounds for controlling animal pests |
| WO2014090700A1 (en) | 2012-12-14 | 2014-06-19 | Basf Se | Malononitrile compounds for controlling animal pests |
| WO2014102244A1 (en) | 2012-12-27 | 2014-07-03 | Basf Se | 2-(pyridin-3-yl)-5-hetaryl-thiazole compounds carrying an imine or imine-derived substituent for combating invertebrate pests |
| WO2014170300A1 (en) | 2013-04-19 | 2014-10-23 | Basf Se | N-substituted acyl-imino-pyridine compounds and derivatives for combating animal pests |
| WO2014202751A1 (en) | 2013-06-21 | 2014-12-24 | Basf Se | Methods for controlling pests in soybean |
| WO2015007682A1 (en) | 2013-07-15 | 2015-01-22 | Basf Se | Pesticide compounds |
| WO2015040116A1 (en) | 2013-09-19 | 2015-03-26 | Basf Se | N-acylimino heterocyclic compounds |
| WO2015055497A1 (en) | 2013-10-16 | 2015-04-23 | Basf Se | Substituted pesticidal pyrazole compounds |
| WO2015055757A1 (en) | 2013-10-18 | 2015-04-23 | Basf Se | Use of pesticidal active carboxamide derivative in soil and seed application and treatment methods |
| EP3456201A1 (en) | 2013-10-18 | 2019-03-20 | BASF Agrochemical Products B.V. | Use of pesticidal active carboxamide derivative in soil and seed application and treatment meth-ods |
| WO2015091649A1 (en) | 2013-12-18 | 2015-06-25 | Basf Se | N-substituted imino heterocyclic compounds |
| WO2015091645A1 (en) | 2013-12-18 | 2015-06-25 | Basf Se | Azole compounds carrying an imine-derived substituent |
| WO2015104422A1 (en) | 2014-01-13 | 2015-07-16 | Basf Se | Dihydrothiophene compounds for controlling invertebrate pests |
| WO2015162260A1 (en) * | 2014-04-25 | 2015-10-29 | Basf Se | Process for preparing anthranilamide esters and derivatives |
| US10149477B2 (en) | 2014-10-06 | 2018-12-11 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| WO2016071499A1 (en) | 2014-11-06 | 2016-05-12 | Basf Se | 3-pyridyl heterobicyclic compound for controlling invertebrate pests |
| US10556844B2 (en) | 2015-02-06 | 2020-02-11 | Basf Se | Pyrazole compounds as nitrification inhibitors |
| US10701937B2 (en) | 2015-02-11 | 2020-07-07 | Basf Se | Pesticidal mixture comprising a pyrazole compound, an insecticide and a fungicide |
| WO2016128261A2 (en) | 2015-02-11 | 2016-08-18 | Basf Se | Pesticidal mixture comprising a pyrazole compound, an insecticide and a fungicide |
| WO2016162371A1 (en) | 2015-04-07 | 2016-10-13 | Basf Agrochemical Products B.V. | Use of an insecticidal carboxamide compound against pests on cultivated plants |
| US11053175B2 (en) | 2015-05-12 | 2021-07-06 | Basf Se | Thioether compounds as nitrification inhibitors |
| WO2016198613A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino compounds |
| WO2016198611A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino heterocyclic compounds |
| WO2017016883A1 (en) | 2015-07-24 | 2017-02-02 | Basf Se | Process for preparation of cyclopentene compounds |
| US11142514B2 (en) | 2015-10-02 | 2021-10-12 | Basf Se | Imino compounds with a 2-chloropyrimidin-5-yl substituent as pest-control agents |
| WO2017072039A1 (en) | 2015-10-26 | 2017-05-04 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093163A1 (en) | 2015-11-30 | 2017-06-08 | Basf Se | Mixtures of cis-jasmone and bacillus amyloliquefaciens |
| WO2017093180A1 (en) | 2015-12-01 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2017093214A1 (en) | 2015-12-03 | 2017-06-08 | Bayer Cropscience Aktiengesellschaft | Mesoionic halogenated 3-(acetyl)-1-[(1,3-thiazol-5-yl)methyl]-1h-imidazo[1,2-a]pyridin-4-ium-2-olate derivatives and related compounds as insecticides |
| WO2017137339A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituted 2-oxyimidazolyl-carboxamides as pest control agents |
| WO2017137338A1 (en) | 2016-02-11 | 2017-08-17 | Bayer Cropscience Aktiengesellschaft | Substituierted 2-(het)aryl-imidazolyl-carboxyamides as pest control agents |
| WO2017144341A1 (en) | 2016-02-23 | 2017-08-31 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| EP3210468A1 (en) | 2016-02-26 | 2017-08-30 | Bayer CropScience Aktiengesellschaft | Solvent-free formulations of low-melting point agents |
| WO2017144497A1 (en) | 2016-02-26 | 2017-08-31 | Bayer Cropscience Aktiengesellschaft | Solvent-free formulations of low-melting active substances |
| WO2017153217A1 (en) | 2016-03-09 | 2017-09-14 | Basf Se | Spirocyclic derivatives |
| WO2017153218A1 (en) | 2016-03-11 | 2017-09-14 | Basf Se | Method for controlling pests of plants |
| WO2017157735A1 (en) | 2016-03-15 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2017157885A1 (en) | 2016-03-16 | 2017-09-21 | Bayer Cropscience Aktiengesellschaft | N-(cyanobenzyl)-6-(cyclopropyl-carbonylamino)-4-(phenyl)-pyridine-2-carboxamide derivatives and related compounds as pesticides and plant protection agents |
| WO2017167832A1 (en) | 2016-04-01 | 2017-10-05 | Basf Se | Bicyclic compounds |
| WO2017174414A1 (en) | 2016-04-05 | 2017-10-12 | Bayer Cropscience Aktiengesellschaft | Naphthaline-derivatives as pest control agents |
| WO2017178416A1 (en) | 2016-04-15 | 2017-10-19 | Bayer Animal Health Gmbh | Pyrazolopyrimidine derivatives |
| WO2017186536A1 (en) | 2016-04-25 | 2017-11-02 | Bayer Cropscience Aktiengesellschaft | Substituted 2-alkylimidazolyl-carboxamides as pest control agents |
| EP3241830A1 (en) | 2016-05-04 | 2017-11-08 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pesticides |
| WO2017198450A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in maize |
| WO2017198449A1 (en) | 2016-05-15 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in brassicaceae |
| WO2017198453A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in potato, tomato or alfalfa |
| WO2017198452A1 (en) | 2016-05-16 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in soybean |
| WO2017198451A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in small grain cereals such as wheat and rice |
| EP3245865A1 (en) | 2016-05-17 | 2017-11-22 | Bayer CropScience Aktiengesellschaft | Method for increasing yield in brassicaceae |
| WO2017198455A2 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in beta spp. plants |
| WO2017198454A1 (en) | 2016-05-17 | 2017-11-23 | Bayer Cropscience Nv | Method for increasing yield in cotton |
| WO2017198588A1 (en) | 2016-05-18 | 2017-11-23 | Basf Se | Capsules comprising benzylpropargylethers for use as nitrification inhibitors |
| WO2018015289A1 (en) | 2016-07-19 | 2018-01-25 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018019937A1 (en) | 2016-07-29 | 2018-02-01 | Bayer Cropscience Aktiengesellschaft | Formulation comprising a beneficial p. bilaii strain and talc for use in seed treatment |
| WO2018029102A1 (en) | 2016-08-10 | 2018-02-15 | Bayer Cropscience Aktiengesellschaft | Substituted 2-heterocyclyl imidazolyl-carboxamides as pest control agents |
| WO2018033455A1 (en) | 2016-08-15 | 2018-02-22 | Bayer Cropscience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018050825A1 (en) | 2016-09-19 | 2018-03-22 | Bayer Cropscience Aktiengesellschaft | Pyrazolo [1,5-a]pyridine derivatives and their use as pesticides |
| WO2018065292A1 (en) | 2016-10-06 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocycle derivatives as pest control agents |
| WO2018065288A1 (en) | 2016-10-07 | 2018-04-12 | Bayer Cropscience Aktiengesellschaft | 2-[2-phenyl-1-(sulfonyl-methyl)-vinyl]-imidazo-[4,5-b] pyridine derivatives and related compounds as pesticides in plant protection |
| WO2018083288A1 (en) | 2016-11-07 | 2018-05-11 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2018087036A1 (en) | 2016-11-11 | 2018-05-17 | Bayer Animal Health Gmbh | New anthelmintic quinoline-3-carboxamide derivatives |
| US10889573B2 (en) | 2016-11-11 | 2021-01-12 | Bayer Animal Health Gmbh | Anthelmintic quinoline-3-carboxamide derivatives |
| US11505545B2 (en) | 2016-11-11 | 2022-11-22 | Bayer Animal Health Gmbh | Anthelmintic quinoline-3-carboxamide derivatives |
| WO2018095953A1 (en) | 2016-11-23 | 2018-05-31 | Bayer Cropscience Aktiengesellschaft | 2-[3-(alkylsulfonyl)-2h-indazol-2-yl]-3h-imidazo[4,5-b]pyridine derivatives and similar compounds as pesticides |
| WO2018104500A1 (en) | 2016-12-09 | 2018-06-14 | Bayer Cropscience Aktiengesellschaft | Plant health effect of purpureocillium lilacinum |
| WO2018108791A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Thiadiazole derivatives as pesticides |
| WO2018108730A1 (en) | 2016-12-16 | 2018-06-21 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| WO2018108671A1 (en) | 2016-12-16 | 2018-06-21 | Basf Se | Pesticidal compounds |
| WO2018130437A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018130443A1 (en) | 2017-01-10 | 2018-07-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2018138050A1 (en) | 2017-01-26 | 2018-08-02 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclene derivatives as pest control agents |
| WO2018141954A1 (en) | 2017-02-06 | 2018-08-09 | Bayer Aktiengesellschaft | Aryl or heteroaryl-substituted imidazo pyridine derivatives and their use as pesticides |
| EP3369320A1 (en) | 2017-03-02 | 2018-09-05 | Bayer CropScience Aktiengesellschaft | Agent for controlling bugs |
| WO2018162312A1 (en) | 2017-03-10 | 2018-09-13 | Basf Se | Spirocyclic derivatives |
| WO2018166855A1 (en) | 2017-03-16 | 2018-09-20 | Basf Se | Heterobicyclic substituted dihydroisoxazoles |
| WO2018177781A1 (en) | 2017-03-28 | 2018-10-04 | Basf Se | Pesticidal compounds |
| WO2018177970A1 (en) | 2017-03-31 | 2018-10-04 | Basf Se | Process for preparing chiral 2,3-dihydrothiazolo[3,2-a]pyrimidin-4-ium compounds |
| EP3978504A1 (en) | 2017-03-31 | 2022-04-06 | Basf Se | Chiral 2,3-dihydrothiazolo[3,2-a]pyrimidine derivatives for combating animal pests |
| WO2018189077A1 (en) | 2017-04-12 | 2018-10-18 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines for use as insecticides |
| WO2018192793A1 (en) | 2017-04-20 | 2018-10-25 | Basf Se | Substituted rhodanine derivatives |
| WO2018192872A1 (en) | 2017-04-21 | 2018-10-25 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2018197257A1 (en) | 2017-04-24 | 2018-11-01 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic-compound derivatives as pest control agents |
| WO2018197466A1 (en) | 2017-04-26 | 2018-11-01 | Basf Se | Substituted succinimide derivatives as pesticides |
| WO2018197401A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Animal Health Gmbh | New bicyclic pyrazole derivatives |
| US11130768B2 (en) | 2017-04-27 | 2021-09-28 | Bayer Animal Health Gmbh | Bicyclic pyrazole derivatives |
| WO2018197692A1 (en) | 2017-04-27 | 2018-11-01 | Bayer Aktiengesellschaft | Heteroarylphenylaminoquinolines and analogues |
| WO2018202494A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202501A1 (en) | 2017-05-02 | 2018-11-08 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2018202706A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylheteroaryloxyquinolines and analogues |
| WO2018202715A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylbenzylbenzimidazoles and analogues |
| WO2018202712A1 (en) | 2017-05-03 | 2018-11-08 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylphenoxyquinolines and analogues |
| WO2018202524A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | 2-{[2-(phenyloxymethyl)pyridin-5-yl]oxy}-ethanamin-derivatives and related compounds as pest-control agents e.g. for the protection of plants |
| US11827616B2 (en) | 2017-05-04 | 2023-11-28 | Discovery Purchaser Corporation | Heterocyclic compounds as pesticides |
| WO2018202525A1 (en) | 2017-05-04 | 2018-11-08 | Bayer Cropscience Aktiengesellschaft | Phenoxyethanamine derivatives for controlling pests |
| WO2018206479A1 (en) | 2017-05-10 | 2018-11-15 | Basf Se | Bicyclic pesticidal compounds |
| EP3400801A1 (en) | 2017-05-10 | 2018-11-14 | Bayer CropScience Aktiengesellschaft | Plant health effect of purpureocillium lilacinum |
| WO2018224455A1 (en) | 2017-06-07 | 2018-12-13 | Basf Se | Substituted cyclopropyl derivatives |
| WO2018229202A1 (en) | 2017-06-16 | 2018-12-20 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2018234202A1 (en) | 2017-06-19 | 2018-12-27 | Basf Se | SUBSTITUTED PYRIMIDINIUM COMPOUNDS AND DERIVATIVES FOR CONTROLLING HARMFUL ANIMALS |
| WO2018234488A1 (en) | 2017-06-23 | 2018-12-27 | Basf Se | SUBSTITUTED CYCLOPROPYL DERIVATIVES |
| WO2019002132A1 (en) | 2017-06-30 | 2019-01-03 | Bayer Animal Health Gmbh | New azaquinoline derivatives |
| WO2019007887A1 (en) | 2017-07-06 | 2019-01-10 | Bayer Aktiengesellschaft | INSECTICIDES AND FUNGICIDES ACTIVE COMPOUNDS |
| EP3284739A1 (en) | 2017-07-19 | 2018-02-21 | Bayer CropScience Aktiengesellschaft | Substituted (het) aryl compounds as pesticides |
| WO2019025341A1 (en) | 2017-08-04 | 2019-02-07 | Bayer Animal Health Gmbh | Quinoline derivatives for treating infections with helminths |
| WO2019035881A1 (en) | 2017-08-17 | 2019-02-21 | Bayer Cropscience Lp | Liquid fertilizer-dispersible compositions and methods thereof |
| WO2019038195A1 (en) | 2017-08-22 | 2019-02-28 | Bayer Aktiengesellschaft | HETEROCYCLEN DERIVATIVES AS PESTICIDES |
| WO2019043183A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| WO2019042932A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| EP3453706A1 (en) | 2017-09-08 | 2019-03-13 | Basf Se | Pesticidal imidazole compounds |
| WO2019059412A1 (en) | 2017-09-20 | 2019-03-28 | Mitsui Chemicals Agro, Inc. | Prolonged ectoparasite-controlling agent for animal |
| WO2019068572A1 (en) | 2017-10-04 | 2019-04-11 | Bayer Aktiengesellschaft | HETEROCYCLEN DERIVATIVES AS PESTICIDES |
| WO2019072906A1 (en) | 2017-10-13 | 2019-04-18 | Basf Se | Imidazolidine pyrimidinium compounds for combating animal pests |
| EP3473103A1 (en) | 2017-10-17 | 2019-04-24 | Bayer AG | Aqueous suspension concentrates based on 2- [(2,4-dichlorophenyl) -methyl] -4,4 '-dimethyl-3-isoxazolidinone |
| WO2019076744A1 (en) | 2017-10-17 | 2019-04-25 | Bayer Aktiengesellschaft | AQUEOUS SUSPENSION CONCENTRATES BASED ON 2 - [(2,4-DICHLORPHENYL) -METHYL] -4,4'-DIMETHYL-3-ISOXAZOLIDINONE |
| WO2019076754A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3473100A1 (en) | 2017-10-18 | 2019-04-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076750A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076749A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076752A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019076751A1 (en) | 2017-10-18 | 2019-04-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2019092086A1 (en) | 2017-11-13 | 2019-05-16 | Bayer Aktiengesellschaft | Tetrazolylpropyl derivatives and their use as fungicides |
| WO2019105875A1 (en) | 2017-11-28 | 2019-06-06 | Bayer Aktiengesellschaft | Heterocyclic compounds as pesticides |
| WO2019105871A1 (en) | 2017-11-29 | 2019-06-06 | Bayer Aktiengesellschaft | Nitrogenous heterocycles as a pesticide |
| WO2019121143A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Substituted cyclopropyl derivatives |
| WO2019122319A1 (en) | 2017-12-21 | 2019-06-27 | Bayer Aktiengesellschaft | Trisubstitutedsilylmethylheteroaryloxyquinolines and analogues |
| WO2019121159A1 (en) | 2017-12-21 | 2019-06-27 | Basf Se | Pesticidal compounds |
| WO2019145140A1 (en) | 2018-01-09 | 2019-08-01 | Basf Se | Silylethynyl hetaryl compounds as nitrification inhibitors |
| WO2019137995A1 (en) | 2018-01-11 | 2019-07-18 | Basf Se | Novel pyridazine compounds for controlling invertebrate pests |
| EP3305786A2 (en) | 2018-01-22 | 2018-04-11 | Bayer CropScience Aktiengesellschaft | Condensed bicyclic heterocycle derivatives as pesticides |
| WO2019155066A1 (en) | 2018-02-12 | 2019-08-15 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2019162174A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019162228A1 (en) | 2018-02-21 | 2019-08-29 | Bayer Aktiengesellschaft | 1-(5-substituted imidazol-1-yl)but-3-en derivatives and their use as fungicides |
| WO2019166561A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of alkoxypyrazoles as nitrification inhibitors |
| WO2019166558A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of pyrazole propargyl ethers as nitrification inhibitors |
| WO2019166560A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of n-functionalized alkoxy pyrazole compounds as nitrification inhibitors |
| WO2019170626A1 (en) | 2018-03-08 | 2019-09-12 | Bayer Aktiengesellschaft | Use of heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides in plant protection |
| WO2019175045A1 (en) | 2018-03-12 | 2019-09-19 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019175046A1 (en) | 2018-03-12 | 2019-09-19 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019185413A1 (en) | 2018-03-27 | 2019-10-03 | Basf Se | Pesticidal substituted cyclopropyl derivatives |
| WO2019197371A1 (en) | 2018-04-10 | 2019-10-17 | Bayer Aktiengesellschaft | Oxadiazoline derivatives |
| WO2019197468A1 (en) | 2018-04-12 | 2019-10-17 | Bayer Aktiengesellschaft | N-(cyclopropylmethyl)-5-(methylsulfonyl)-n-{1-[1-(pyrimidin-2-yl)-1h-1,2,4-triazol-5-yl]ethyl}benzamide derivatives and the corresponding pyridine-carboxamide derivatives as pesticides |
| EP3904350A1 (en) | 2018-04-12 | 2021-11-03 | Bayer Aktiengesellschaft | N-(cyclopropylmethyl)-5-(methylsulfonyl)-n-{1-[1-(pyrimidin-2-yl)-1h-1,2,4-triazol-5-yl]ethyl}benzamide derivatives and the corresponding pyridine-carboxamide derivatives as pesticides |
| EP3904349A2 (en) | 2018-04-12 | 2021-11-03 | Bayer Aktiengesellschaft | N-(cyclopropylmethyl)-5-(methylsulfonyl)-n-{1-[1-(pyrimidin-2-yl)-1h-1,2,4-triazol-5-yl]ethyl}heterocyclyl amide derivatives and similar compounds as pesticides |
| WO2019197615A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with fungicides, insecticides and acaricidal properties |
| WO2019197623A1 (en) | 2018-04-13 | 2019-10-17 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, fungicidal and acaricidal properties |
| EP4670505A2 (en) | 2018-04-13 | 2025-12-31 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, nematicidal and acaricidal properties |
| WO2019201835A1 (en) | 2018-04-17 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019201921A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2019202077A1 (en) | 2018-04-20 | 2019-10-24 | Bayer Aktiengesellschaft | Heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| EP3919486A1 (en) | 2018-04-25 | 2021-12-08 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019206799A1 (en) | 2018-04-25 | 2019-10-31 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole and heteroaryl-tetrazole compounds as pesticides |
| WO2019215182A1 (en) | 2018-05-09 | 2019-11-14 | Bayer Animal Health Gmbh | New quinoline derivatives |
| WO2019219529A1 (en) | 2018-05-15 | 2019-11-21 | Basf Se | Mixtures comprising benzpyrimoxan and oxazosulfyl and uses and methods of applying them |
| WO2019224092A1 (en) | 2018-05-22 | 2019-11-28 | Basf Se | Pesticidally active c15-derivatives of ginkgolides |
| WO2019224143A1 (en) | 2018-05-24 | 2019-11-28 | Bayer Aktiengesellschaft | Active ingredient combinations with insecticidal, nematicidal and acaricidal properties |
| US12365655B2 (en) | 2018-06-20 | 2025-07-22 | Ctxt Pty Ltd | Compounds |
| WO2020005678A1 (en) | 2018-06-25 | 2020-01-02 | Bayer Cropscience Lp | Seed treatment method |
| WO2020002189A1 (en) | 2018-06-27 | 2020-01-02 | Bayer Aktiengesellschaft | Active substance combinations |
| WO2020002472A1 (en) | 2018-06-28 | 2020-01-02 | Basf Se | Use of alkynylthiophenes as nitrification inhibitors |
| EP3586630A1 (en) | 2018-06-28 | 2020-01-01 | Bayer AG | Active compound combinations having insecticidal/acaricidal properties |
| US11911372B2 (en) | 2018-06-28 | 2024-02-27 | Ctxt Pty Ltd | Compounds |
| US12516037B2 (en) | 2018-07-05 | 2026-01-06 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| US11884643B2 (en) | 2018-07-05 | 2024-01-30 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020007902A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| US11952359B2 (en) | 2018-07-05 | 2024-04-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020007905A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020007904A1 (en) | 2018-07-05 | 2020-01-09 | Bayer Aktiengesellschaft | Substituted thiophenecarboxamides and analogues as antibacterials agents |
| WO2020020765A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of a substituted thiazolidine compound as nitrification inhibitor |
| WO2020020777A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of substituted 2-thiazolines as nitrification inhibitors |
| WO2020020813A1 (en) | 2018-07-25 | 2020-01-30 | Bayer Aktiengesellschaft | Fungicidal active compound combinations |
| WO2020020816A1 (en) | 2018-07-26 | 2020-01-30 | Bayer Aktiengesellschaft | Novel triazole derivatives |
| WO2020021082A1 (en) | 2018-07-27 | 2020-01-30 | Bayer Aktiengesellschaft | Controlled release formulations for agrochemicals |
| WO2020025650A1 (en) | 2018-07-31 | 2020-02-06 | Bayer Aktiengesellschaft | Controlled release formulations with lignin for agrochemicals |
| EP3613736A1 (en) | 2018-08-22 | 2020-02-26 | Basf Se | Substituted glutarimide derivatives |
| WO2020043650A1 (en) | 2018-08-29 | 2020-03-05 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020053282A1 (en) | 2018-09-13 | 2020-03-19 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2020057939A1 (en) | 2018-09-17 | 2020-03-26 | Bayer Aktiengesellschaft | Use of the fungicide isoflucypram for controlling claviceps purpurea and reducing sclerotia in cereals |
| WO2020064492A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling pests by seed treatment application of a mesoionic compound or mixture thereof |
| WO2020064480A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| WO2020064408A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| EP3628158A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| EP3628156A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method for controlling pests of sugarcane, citrus, rapeseed, and potato plants |
| EP3628157A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| WO2020070050A1 (en) | 2018-10-01 | 2020-04-09 | Bayer Aktiengesellschaft | Fungicidal 5-substituted imidazol-1-yl carbinol derivatives |
| EP3636644A1 (en) | 2018-10-11 | 2020-04-15 | Bayer Aktiengesellschaft | Mesoionic imidazopyridines as insecticides |
| WO2020078839A1 (en) | 2018-10-16 | 2020-04-23 | Bayer Aktiengesellschaft | Active substance combinations |
| WO2020079167A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Heteroarylaminoquinolines and analogues |
| WO2020079173A1 (en) | 2018-10-18 | 2020-04-23 | Bayer Aktiengesellschaft | Pyridylphenylaminoquinolines and analogues |
| WO2020079232A1 (en) | 2018-10-20 | 2020-04-23 | Bayer Aktiengesellschaft | Oxetanylphenoxyquinolines and analogues |
| WO2020083971A2 (en) | 2018-10-24 | 2020-04-30 | Bayer Animal Health Gmbh | New anthelmintic compounds |
| EP3643705A1 (en) | 2018-10-24 | 2020-04-29 | Basf Se | Pesticidal compounds |
| EP3643711A1 (en) | 2018-10-24 | 2020-04-29 | Bayer Animal Health GmbH | New anthelmintic compounds |
| WO2020083733A1 (en) | 2018-10-24 | 2020-04-30 | Basf Se | Pesticidal compounds |
| US12435068B2 (en) | 2018-10-24 | 2025-10-07 | Elanco Animal Health Gmbh | Anthelmintic compounds |
| WO2020109391A1 (en) | 2018-11-28 | 2020-06-04 | Bayer Aktiengesellschaft | Pyridazine (thio)amides as fungicidal compounds |
| WO2020109039A1 (en) | 2018-11-28 | 2020-06-04 | Basf Se | Pesticidal compounds |
| WO2020114932A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2020114934A1 (en) | 2018-12-07 | 2020-06-11 | Bayer Aktiengesellschaft | Herbicide compositions |
| EP3620052A1 (en) | 2018-12-12 | 2020-03-11 | Bayer Aktiengesellschaft | Use of phenoxypyridinyl-substituted (1h-1,2,4-triazol-1-yl)alcohols for controlling fungicidal diseases in maize |
| WO2020126591A1 (en) | 2018-12-18 | 2020-06-25 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| WO2020126980A1 (en) | 2018-12-18 | 2020-06-25 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020127780A1 (en) | 2018-12-20 | 2020-06-25 | Bayer Aktiengesellschaft | Heterocyclyl pyridazine as fungicidal compounds |
| EP3669652A1 (en) | 2018-12-21 | 2020-06-24 | Bayer AG | Active compound combination |
| WO2020127974A1 (en) | 2018-12-21 | 2020-06-25 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as new antifungal agents |
| EP3679791A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679789A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679793A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679790A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3679792A1 (en) | 2019-01-08 | 2020-07-15 | Bayer AG | Active compound combinations |
| EP3696177A1 (en) | 2019-02-12 | 2020-08-19 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP3545764A1 (en) | 2019-02-12 | 2019-10-02 | Bayer AG | Crystal form of 2-({2-fluoro-4-methyl-5-[(r)-(2,2,2-trifluoroethyl)sulfinyl]phenyl}imino)-3-(2,2,2- trifluoroethyl)-1,3-thiazolidin-4-one |
| WO2020173860A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Fused bicyclic heterocycle derivatives as pesticides |
| WO2020173861A1 (en) | 2019-02-26 | 2020-09-03 | Bayer Aktiengesellschaft | Condensed bicyclic heterocyclic derivatives as pest control agents |
| WO2020178067A1 (en) | 2019-03-01 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2020178307A1 (en) | 2019-03-05 | 2020-09-10 | Bayer Aktiengesellschaft | Active compound combination |
| WO2020182929A1 (en) | 2019-03-13 | 2020-09-17 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| WO2020187656A1 (en) | 2019-03-15 | 2020-09-24 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| EP3564225A1 (en) | 2019-03-21 | 2019-11-06 | Bayer Aktiengesellschaft | Crystalline form of spiromesifen |
| EP3725788A1 (en) | 2019-04-15 | 2020-10-21 | Bayer AG | Novel heteroaryl-substituted aminoalkyl azole compounds as pesticides |
| WO2020212235A1 (en) | 2019-04-15 | 2020-10-22 | Bayer Animal Health Gmbh | Novel heteroaryl-substituted aminoalkyl azole compounds as pesticides |
| WO2020225434A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading ulv formulations for agrochemical compounds ii |
| WO2020225435A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading and uptake ulv formulations |
| WO2020225439A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Ulv formulations with enhanced rainfastness |
| WO2020225438A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High uptake and rainfastness ulv formulations |
| WO2020225437A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Ulv formulations with enhanced uptake |
| WO2020225428A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading ulv formulations for insecticides |
| WO2020225242A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | Active compound combination |
| WO2020225436A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading, uptake and rainfastness ulv formulations |
| WO2020225440A1 (en) | 2019-05-08 | 2020-11-12 | Bayer Aktiengesellschaft | High spreading and rainfastness ulv formulations |
| WO2020231751A1 (en) | 2019-05-10 | 2020-11-19 | Bayer Cropscience Lp | Active compound combinations |
| WO2020229398A1 (en) | 2019-05-14 | 2020-11-19 | Bayer Aktiengesellschaft | (1-alkenyl)-substituted pyrazoles and triazoles as pest control agents |
| WO2020239517A1 (en) | 2019-05-29 | 2020-12-03 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| EP3750888A1 (en) | 2019-06-12 | 2020-12-16 | Bayer Aktiengesellschaft | Crystalline form a of 1,4-dimethyl-2-[2-(pyridin-3-yl)-2h-indazol-5-yl]-1,2,4-triazolidine-3,5-dione |
| US11492346B2 (en) | 2019-06-18 | 2022-11-08 | Pfizer Inc. | Benzisoxazole sulfonamide derivatives |
| US12371425B2 (en) | 2019-06-18 | 2025-07-29 | Pfizer Inc. | Benzisoxazole sulfonamide derivatives |
| WO2020254489A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Benzylphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020254493A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Thienylhydroxyisoxazolines and derivatives thereof |
| WO2020254492A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254490A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Phenoxyphenyl hydroxyisoxazolines and analogues as new antifungal agents |
| WO2020254486A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254494A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Fungicidal oxadiazoles |
| WO2020254487A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and derivatives thereof |
| WO2020254488A1 (en) | 2019-06-21 | 2020-12-24 | Bayer Aktiengesellschaft | Hydroxyisoxazolines and use thereof as fungicides |
| WO2020263812A1 (en) | 2019-06-24 | 2020-12-30 | Auburn University | A bacillus strain and methods of its use for plant growth promotion |
| EP3608311A1 (en) | 2019-06-28 | 2020-02-12 | Bayer AG | Crystalline form a of n-[4-chloro-3-[(1-cyanocyclopropyl)carbamoyl]phenyl]-2-methyl-4-methylsulfonyl-5-(1,1,2,2,2-pentafluoroethyl)pyrazole-3-carboxamide |
| WO2021001331A1 (en) | 2019-07-03 | 2021-01-07 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides and derivatives thereof as microbicides |
| EP4378314A2 (en) | 2019-07-04 | 2024-06-05 | Bayer AG | Herbicidal compositions |
| WO2021001273A1 (en) | 2019-07-04 | 2021-01-07 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2021013561A1 (en) | 2019-07-19 | 2021-01-28 | Basf Se | Pesticidal pyrazole and triazole derivatives |
| EP3766879A1 (en) | 2019-07-19 | 2021-01-20 | Basf Se | Pesticidal pyrazole derivatives |
| WO2021013721A1 (en) | 2019-07-22 | 2021-01-28 | Bayer Aktiengesellschaft | 5-amino substituted pyrazoles and triazoles as pest control agents |
| EP3769623A1 (en) | 2019-07-22 | 2021-01-27 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2021013720A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021013719A1 (en) | 2019-07-23 | 2021-01-28 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021018839A1 (en) | 2019-07-30 | 2021-02-04 | Bayer Animal Health Gmbh | Isoquinoline derivatives and their use for the treatment of parasitic infections |
| EP3771714A1 (en) | 2019-07-30 | 2021-02-03 | Bayer AG | Nitrogen-containing heterocycles as pesticides |
| EP3701796A1 (en) | 2019-08-08 | 2020-09-02 | Bayer AG | Active compound combinations |
| WO2021048188A1 (en) | 2019-09-11 | 2021-03-18 | Bayer Aktiengesellschaft | Highly effective formulations on the basis of 2-[(2,4-dichlorphenyl)-methyl]-4,4'-dimethyl-3-isoxazolidinones and preemergence herbicides |
| WO2021058659A1 (en) | 2019-09-26 | 2021-04-01 | Bayer Aktiengesellschaft | Rnai-mediated pest control |
| WO2021069569A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021069567A1 (en) | 2019-10-09 | 2021-04-15 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021069575A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Animal Health Gmbh | Heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021089673A1 (en) | 2019-11-07 | 2021-05-14 | Bayer Aktiengesellschaft | Substituted sulfonyl amides for controlling animal pests |
| WO2021097162A1 (en) | 2019-11-13 | 2021-05-20 | Bayer Cropscience Lp | Beneficial combinations with paenibacillus |
| WO2021099303A1 (en) | 2019-11-18 | 2021-05-27 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021105091A1 (en) | 2019-11-25 | 2021-06-03 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021123051A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides, thiophene carboxylic acids and derivatives thereof |
| WO2021122986A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Thienyloxazolones and analogues |
| WO2021130143A1 (en) | 2019-12-23 | 2021-07-01 | Basf Se | Enzyme enhanced root uptake of agrochemical active compound |
| WO2021136758A1 (en) | 2019-12-30 | 2021-07-08 | Bayer Aktiengesellschaft | Aqueous capsule suspension concentrates based on polyurea shell material containing polyfunctional aminocarboxylic esters |
| EP3845304A1 (en) | 2019-12-30 | 2021-07-07 | Bayer AG | Capsule suspension concentrates based on polyisocyanates and biodegradable amine based cross-linker |
| WO2021165195A1 (en) | 2020-02-18 | 2021-08-26 | Bayer Aktiengesellschaft | Heteroaryl-triazole compounds as pesticides |
| WO2021170527A1 (en) | 2020-02-24 | 2021-09-02 | Bayer Aktiengesellschaft | Encapsulated pyrethroids with improved effictiveness in soil and leaf applications |
| EP3868207A1 (en) | 2020-02-24 | 2021-08-25 | Bayer Aktiengesellschaft | Encapsulated pyrethroids with improved activity in soil and leaf applications |
| WO2021170463A1 (en) | 2020-02-28 | 2021-09-02 | BASF Agro B.V. | Methods and uses of a mixture comprising alpha-cypermethrin and dinotefuran for controlling invertebrate pests in turf |
| EP3708565A1 (en) | 2020-03-04 | 2020-09-16 | Bayer AG | Pyrimidinyloxyphenylamidines and the use thereof as fungicides |
| WO2021204930A1 (en) | 2020-04-09 | 2021-10-14 | Bayer Animal Health Gmbh | Substituted condensed azines as anthelmintic compounds |
| WO2021209363A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209364A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209365A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209490A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Cyclaminephenylaminoquinolines as fungicides |
| WO2021209366A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021209368A1 (en) | 2020-04-16 | 2021-10-21 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| WO2021213978A1 (en) | 2020-04-21 | 2021-10-28 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocyclic derivatives as pest control agents |
| WO2021219513A1 (en) | 2020-04-28 | 2021-11-04 | Basf Se | Pesticidal compounds |
| WO2021224323A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Novel heteroaryl-triazole compounds as pesticides |
| WO2021224220A1 (en) | 2020-05-06 | 2021-11-11 | Bayer Aktiengesellschaft | Pyridine (thio)amides as fungicidal compounds |
| WO2021228734A1 (en) | 2020-05-12 | 2021-11-18 | Bayer Aktiengesellschaft | Triazine and pyrimidine (thio)amides as fungicidal compounds |
| EP3909950A1 (en) | 2020-05-13 | 2021-11-17 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2021233861A1 (en) | 2020-05-19 | 2021-11-25 | Bayer Aktiengesellschaft | Azabicyclic(thio)amides as fungicidal compounds |
| WO2021245087A1 (en) | 2020-06-04 | 2021-12-09 | Bayer Aktiengesellschaft | Heterocyclyl pyrimidines and triazines as novel fungicides |
| WO2021249995A1 (en) | 2020-06-10 | 2021-12-16 | Bayer Aktiengesellschaft | Azabicyclyl-substituted heterocycles as fungicides |
| WO2021255071A1 (en) | 2020-06-18 | 2021-12-23 | Bayer Aktiengesellschaft | 3-(pyridazin-4-yl)-5,6-dihydro-4h-1,2,4-oxadiazine derivatives as fungicides for crop protection |
| WO2021255089A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines and 1,3,4-oxadiazole pyridines as fungicides |
| WO2021255170A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| WO2021255169A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazole pyrimidines as fungicides |
| WO2021255091A1 (en) | 2020-06-19 | 2021-12-23 | Bayer Aktiengesellschaft | 1,3,4-oxadiazoles and their derivatives as fungicides |
| WO2021259997A1 (en) | 2020-06-25 | 2021-12-30 | Bayer Animal Health Gmbh | Novel heteroaryl-substituted pyrazine derivatives as pesticides |
| EP3929189A1 (en) | 2020-06-25 | 2021-12-29 | Bayer Animal Health GmbH | Novel heteroaryl-substituted pyrazine derivatives as pesticides |
| WO2021260017A1 (en) | 2020-06-26 | 2021-12-30 | Bayer Aktiengesellschaft | Aqueous capsule suspension concentrates comprising biodegradable ester groups |
| WO2022002818A1 (en) | 2020-07-02 | 2022-01-06 | Bayer Aktiengesellschaft | Heterocyclene derivatives as pest control agents |
| WO2022033991A1 (en) | 2020-08-13 | 2022-02-17 | Bayer Aktiengesellschaft | 5-amino substituted triazoles as pest control agents |
| WO2022053453A1 (en) | 2020-09-09 | 2022-03-17 | Bayer Aktiengesellschaft | Azole carboxamide as pest control agents |
| WO2022058327A1 (en) | 2020-09-15 | 2022-03-24 | Bayer Aktiengesellschaft | Substituted ureas and derivatives as new antifungal agents |
| EP3974414A1 (en) | 2020-09-25 | 2022-03-30 | Bayer AG | 5-amino substituted pyrazoles and triazoles as pesticides |
| EP3915371A1 (en) | 2020-11-04 | 2021-12-01 | Bayer AG | Active compound combinations and fungicide compositions comprising those |
| WO2022096694A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading and ulv tank mix adjuvant formulation |
| EP3994993A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading and ulv tank mix adjuvant formulation |
| WO2022096688A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading and rainfastness properties |
| WO2022096691A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, uptake and rainfastness properties |
| WO2022096696A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| WO2022096685A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and spreading properties |
| WO2022096686A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift properties |
| WO2022096692A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading, uptake and rainfastness properties |
| WO2022096687A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and uptake properties |
| WO2022096693A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high uptake and ulv tank mix adjuvant formulation |
| WO2022096695A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| EP3994992A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Low drift, rainfastness, high uptake and ulv tank mix adjuvant formulation |
| EP3994987A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift and uptake properties |
| WO2022096690A1 (en) | 2020-11-08 | 2022-05-12 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading and uptake properties |
| EP3994988A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and rainfastness properties |
| EP3994985A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift properties |
| EP3994994A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| EP3994989A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, rainfastness and uptake properties |
| EP3994990A1 (en) | 2020-11-08 | 2022-05-11 | Bayer AG | Agrochemical composition with improved drift, spreading and uptake properties |
| EP3994991A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift, spreading, uptake and rainfastness properties |
| EP3994995A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Low drift, rainfastness, high spreading, high uptake and ulv tank mix adjuvant formulation |
| EP3994986A1 (en) | 2020-11-08 | 2022-05-11 | Bayer Aktiengesellschaft | Agrochemical composition with improved drift and spreading properties |
| EP3915971A1 (en) | 2020-12-16 | 2021-12-01 | Bayer Aktiengesellschaft | Phenyl-s(o)n-phenylamidines and the use thereof as fungicides |
| WO2022129196A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | Heterobicycle substituted 1,2,4-oxadiazoles as fungicides |
| WO2022129188A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | 1,2,4-oxadiazol-3-yl pyrimidines as fungicides |
| WO2022129190A1 (en) | 2020-12-18 | 2022-06-23 | Bayer Aktiengesellschaft | (hetero)aryl substituted 1,2,4-oxadiazoles as fungicides |
| WO2022152728A1 (en) | 2021-01-15 | 2022-07-21 | Bayer Aktiengesellschaft | Herbicidal compositions |
| EP4036083A1 (en) | 2021-02-02 | 2022-08-03 | Bayer Aktiengesellschaft | 5-oxy substituted heterocycles as pesticides |
| WO2022167488A1 (en) | 2021-02-02 | 2022-08-11 | Basf Se | Synergistic action of dcd and alkoxypyrazoles as nitrification inhibitors |
| EP4043444A1 (en) | 2021-02-11 | 2022-08-17 | Basf Se | Substituted isoxazoline derivatives |
| WO2022207494A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2022207496A1 (en) | 2021-03-30 | 2022-10-06 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2022233777A1 (en) | 2021-05-06 | 2022-11-10 | Bayer Aktiengesellschaft | Alkylamide substituted, annulated imidazoles and use thereof as insecticides |
| WO2022238194A1 (en) | 2021-05-10 | 2022-11-17 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2022238391A1 (en) | 2021-05-12 | 2022-11-17 | Bayer Aktiengesellschaft | 2-(het)aryl-substituted condensed heterocycle derivatives as pest control agents |
| WO2022243523A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of an n-functionalized alkoxy pyrazole compound as nitrification inhibitor |
| WO2022243521A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of ethynylpyridine compounds as nitrification inhibitors |
| WO2022268810A1 (en) | 2021-06-21 | 2022-12-29 | Basf Se | Metal-organic frameworks with pyrazole-based building blocks |
| EP4119547A1 (en) | 2021-07-12 | 2023-01-18 | Basf Se | Triazole compounds for the control of invertebrate pests |
| WO2023017120A1 (en) | 2021-08-13 | 2023-02-16 | Bayer Aktiengesellschaft | Active compound combinations and fungicide compositions comprising those |
| EP4140986A1 (en) | 2021-08-23 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| WO2023025682A1 (en) | 2021-08-25 | 2023-03-02 | Bayer Aktiengesellschaft | Novel pyrazinyl-triazole compounds as pesticides |
| EP4140995A1 (en) | 2021-08-27 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4144739A1 (en) | 2021-09-02 | 2023-03-08 | Bayer Aktiengesellschaft | Anellated pyrazoles as parasiticides |
| WO2023036821A1 (en) | 2021-09-09 | 2023-03-16 | Bayer Animal Health Gmbh | New quinoline derivatives |
| EP4148052A1 (en) | 2021-09-09 | 2023-03-15 | Bayer Animal Health GmbH | New quinoline derivatives |
| EP4151631A1 (en) | 2021-09-20 | 2023-03-22 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2023078915A1 (en) | 2021-11-03 | 2023-05-11 | Bayer Aktiengesellschaft | Bis(hetero)aryl thioether (thio)amides as fungicidal compounds |
| WO2023092050A1 (en) | 2021-11-20 | 2023-05-25 | Bayer Cropscience Lp | Beneficial combinations with recombinant bacillus cells expressing a serine protease |
| WO2023099445A1 (en) | 2021-11-30 | 2023-06-08 | Bayer Aktiengesellschaft | Bis(hetero)aryl thioether oxadiazines as fungicidal compounds |
| EP4194453A1 (en) | 2021-12-08 | 2023-06-14 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4198033A1 (en) | 2021-12-14 | 2023-06-21 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2023110656A1 (en) | 2021-12-15 | 2023-06-22 | Bayer Aktiengesellschaft | Spectroscopic solution for non-destructive quantification of one or more chemical substances in a matrix comprising coating and bulk material in a sample, such as coated seeds, using multivariate data analysis |
| EP4198023A1 (en) | 2021-12-16 | 2023-06-21 | Basf Se | Pesticidally active thiosemicarbazone compounds |
| EP4238971A1 (en) | 2022-03-02 | 2023-09-06 | Basf Se | Substituted isoxazoline derivatives |
| WO2023205602A1 (en) | 2022-04-18 | 2023-10-26 | Basf Corporation | High-load agricultural formulations and methods of making same |
| WO2023203009A1 (en) | 2022-04-20 | 2023-10-26 | Bayer Aktiengesellschaft | Water dispersible granules with low melting active ingredients prepared by extrusion |
| EP4265110A1 (en) | 2022-04-20 | 2023-10-25 | Bayer AG | Water dispersible granules with low melting active ingredients prepared by extrusion |
| WO2023203066A1 (en) | 2022-04-21 | 2023-10-26 | Basf Se | Synergistic action as nitrification inhibitors of dcd oligomers with alkoxypyrazole and its oligomers |
| WO2023208447A1 (en) | 2022-04-25 | 2023-11-02 | Basf Se | An emulsifiable concentrate having a (substituted) benzaldehyde-based solvent system |
| WO2023213670A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Crystalline forms of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine |
| WO2023213626A1 (en) | 2022-05-03 | 2023-11-09 | Bayer Aktiengesellschaft | Use of (5s)-3-[3-(3-chloro-2-fluorophenoxy)-6-methylpyridazin-4-yl]-5-(2-chloro-4-methylbenzyl)-5,6-dihydro-4h-1,2,4-oxadiazine for controlling unwanted microorganisms |
| WO2023217619A1 (en) | 2022-05-07 | 2023-11-16 | Bayer Aktiengesellschaft | Low drift aqueous liquid formulations for low, medium, and high spray volume application |
| WO2023237444A1 (en) | 2022-06-06 | 2023-12-14 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| EP4295683A1 (en) | 2022-06-21 | 2023-12-27 | Bayer Aktiengesellschaft | Agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2-difluoroethyl)amino]furan-2(5h)-one |
| WO2024013016A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024013015A1 (en) | 2022-07-11 | 2024-01-18 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024028243A1 (en) | 2022-08-02 | 2024-02-08 | Basf Se | Pyrazolo pesticidal compounds |
| EP4342885A1 (en) | 2022-09-20 | 2024-03-27 | Basf Se | N-(3-(aminomethyl)-phenyl)-5-(4-phenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-amine derivatives and similar compounds as pesticides |
| WO2024068473A1 (en) | 2022-09-27 | 2024-04-04 | Bayer Aktiengesellschaft | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts |
| WO2024068520A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068518A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-heteroaryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068519A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| WO2024068517A1 (en) | 2022-09-28 | 2024-04-04 | Bayer Aktiengesellschaft | 3-(hetero)aryl-5-chlorodifluoromethyl-1,2,4-oxadiazole as fungicide |
| EP4295688A1 (en) | 2022-09-28 | 2023-12-27 | Bayer Aktiengesellschaft | Active compound combination |
| EP4353082A1 (en) | 2022-10-14 | 2024-04-17 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2024104643A1 (en) | 2022-11-17 | 2024-05-23 | Bayer Aktiengesellschaft | Use of isotianil for controlling plasmodiophora brassica |
| EP4389210A1 (en) | 2022-12-21 | 2024-06-26 | Basf Se | Heteroaryl compounds for the control of invertebrate pests |
| WO2024170472A1 (en) | 2023-02-16 | 2024-08-22 | Bayer Aktiengesellschaft | Herbicidal mixtures |
| WO2024213752A1 (en) | 2023-04-14 | 2024-10-17 | Elanco Animal Health Gmbh | Long-term prevention and/or treatment of a disease by slo-1 inhibitors |
| EP4455137A1 (en) | 2023-04-24 | 2024-10-30 | Basf Se | Pyrimidine compounds for the control of invertebrate pests |
| EP4467535A1 (en) | 2023-05-25 | 2024-11-27 | Basf Se | Lactam pesticidal compounds |
| EP4488273A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488269A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488270A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| WO2025026738A1 (en) | 2023-07-31 | 2025-02-06 | Bayer Aktiengesellschaft | 6-[5-(ethylsulfonyl)-1-methyl-1h-imidazol-4-yl]-7-methyl-3-(pentafluoroethyl)-7h-imidazo[4,5-c]pyridazine derivatives as pesticides |
| WO2025026787A1 (en) | 2023-08-01 | 2025-02-06 | Globachem Nv | Plant defense elicitors |
| WO2025026815A1 (en) | 2023-08-01 | 2025-02-06 | Globachem Nv | Insecticidal mixtures |
| WO2025031668A1 (en) | 2023-08-09 | 2025-02-13 | Bayer Aktiengesellschaft | Azaheterobiaryl-substituted 4,5-dihydro-1h-2,4,5-oxadiazines as novel fungicides |
| WO2025032038A1 (en) | 2023-08-09 | 2025-02-13 | Bayer Aktiengesellschaft | Pyridazin-4-yloxadiazines as novel fungicides |
| WO2025040301A1 (en) | 2023-08-21 | 2025-02-27 | Bayer Aktiengesellschaft | Herbicide/safener combinations based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy] acetic acids and herbicides from the class of substituted cyclic diones and the salts thereof |
| WO2025040520A1 (en) | 2023-08-21 | 2025-02-27 | Bayer Aktiengesellschaft | Herbicide/safener combinations based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy] acetic acids and herbicides from the class of substituted cyclic diones and the salts thereof |
| WO2025049555A1 (en) | 2023-08-31 | 2025-03-06 | Oerth Bio Llc | Compositions and methods for targeted inhibition and degradation of proteins in an insect cell |
| WO2025078128A1 (en) | 2023-10-11 | 2025-04-17 | Bayer Aktiengesellschaft | Pyridazin-3-one-4-yloxadiazines as novel fungicides |
| WO2025098875A1 (en) | 2023-11-10 | 2025-05-15 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2025098874A1 (en) | 2023-11-10 | 2025-05-15 | Bayer Aktiengesellschaft | Active compound combinations having fungicidal/insecticidal/acaricidal properties |
| WO2025098876A1 (en) | 2023-11-10 | 2025-05-15 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2025108865A1 (en) | 2023-11-23 | 2025-05-30 | Bayer Aktiengesellschaft | Herbicidal compositions |
| WO2025117659A1 (en) | 2023-11-29 | 2025-06-05 | Basf Corporation | Insecticide delivery by nanocarriers |
| WO2025114238A1 (en) | 2023-12-01 | 2025-06-05 | Bayer Aktiengesellschaft | Solid agrochemical formulations comprising crystalline form a of 4-[(6-chloro-3-pyridylmethyl)(2,2- difluoroethyl)amino]furan-2(5h)-one |
| WO2025119860A1 (en) | 2023-12-05 | 2025-06-12 | Bayer Aktiengesellschaft | Liquid agrochemical formulations comprising flupyradifurone |
| WO2025132148A1 (en) | 2023-12-21 | 2025-06-26 | Bayer Aktiengesellschaft | Adjuvant compositions for agrochemical applications |
| EP4574819A1 (en) | 2023-12-22 | 2025-06-25 | Basf Se | Diazinone compounds for the control of invertebrate pests |
| WO2025168620A1 (en) | 2024-02-07 | 2025-08-14 | Bayer Aktiengesellschaft | Heteroaryl-substituted 4,5-dihydro-1h-2,4,5-oxadiazines as novel fungicides |
| WO2025168777A1 (en) | 2024-02-08 | 2025-08-14 | Bayer Aktiengesellschaft | Use of isoxazolinecarboxamide for selective weed control in pineapple |
| WO2025186065A1 (en) | 2024-03-05 | 2025-09-12 | Bayer Aktiengesellschaft | Heteroaryl-substituted (aza)quinoxaline derivatives as pesticides |
| WO2025190927A1 (en) | 2024-03-14 | 2025-09-18 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2025242699A1 (en) | 2024-05-22 | 2025-11-27 | Basf Se | Method for improving rainfastness of an agrochemical active ingredient |
| WO2025257122A1 (en) | 2024-06-12 | 2025-12-18 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2025257121A1 (en) | 2024-06-12 | 2025-12-18 | Bayer Aktiengesellschaft | Active compound combinations having insecticidal/acaricidal properties |
| WO2025257242A1 (en) | 2024-06-14 | 2025-12-18 | Bayer Aktiengesellschaft | Heterocyclyl-substituted aryl- and heteroarylpyrazines and salts thereof, and the use thereof as herbicidal active substances |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2011085684A1 (en) | 2011-07-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2011085575A1 (en) | Ortho-heterocyclyl formanilide compounds, their synthesis methods and use | |
| CN110028423B (en) | Meta-diamide compound and preparation method and application thereof | |
| KR100890886B1 (en) | Novel haloalkylsulfonanilide derivative, herbicide, and method of use thereof | |
| CN1993328B (en) | Quinoline derivatives and insecticides containing them as active ingredients | |
| CA3150249A1 (en) | Piperonylic acid derivative and application thereof | |
| JP7138781B2 (en) | Benzamide compound and use thereof | |
| WO1998045274A1 (en) | Pyrazole derivatives, process for preparing the same, intermediates, and pest control agent containing the same as active ingredient | |
| WO2012034472A1 (en) | Cyano benzenedicarboxamide compounds, preparing methods and as agricultural insecticides uses thereof | |
| JPH0551582B2 (en) | ||
| CN101668748A (en) | Substituted pyrimidine ether compound and application thereof | |
| CN104961728A (en) | Preparation method and application of pyridinyl methoxybiphenyl structure-containing pyrazole oxime ester compound | |
| EP0329020B1 (en) | Pyrazole derivatives, processes for their manufacture and insecticidal, miticidal and fungicidal compositions containg the derivatives as active ingredients | |
| CN119059936A (en) | 3-Aminobenzoic acid bisamide compounds | |
| WO1991007392A1 (en) | Pesticidal pyrimidinyl benzoic acids and esters | |
| KR100647175B1 (en) | Diamine derivative, its manufacturing method, and bactericide using diamine derivative as an active ingredient | |
| JP2024507409A (en) | Benzimidazole compounds or their salts, their production methods and uses, and insecticides and acaricides and their uses | |
| EP0307801B1 (en) | Pyrazole derivative and insecticidal and miticidal composition containing the derivative as active ingredient | |
| WO2025130973A1 (en) | Substituted benzimidazole compound and/or salt thereof and preparation method therefor and use thereof, and insecticidal-miticidal agent | |
| CN117062800A (en) | Aryl sulfides and their preparation methods and applications | |
| JP2000016982A (en) | Quinoline derivatives and herbicides containing the same as active ingredients | |
| JP2888946B2 (en) | Oxazoline derivatives and insecticides and acaricides containing the same | |
| WO2008134970A1 (en) | Anthranilamide compounds and the use thereof | |
| EP0354297B1 (en) | Thiadiazines, process for production thereof, and insecticidal and acaricidal agents comprising the thiadiazines | |
| JPH0776577A (en) | Diphenyltriazole derivative and insecticide, acaricide | |
| KR102035091B1 (en) | Pyrazole carboxamide compounds containing organosulfur group and insecticide composition comprising same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 10842859 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 10842859 Country of ref document: EP Kind code of ref document: A1 |