CN1309897A - Unsaturated oximino ether bactericide - Google Patents
Unsaturated oximino ether bactericide Download PDFInfo
- Publication number
- CN1309897A CN1309897A CN 00110143 CN00110143A CN1309897A CN 1309897 A CN1309897 A CN 1309897A CN 00110143 CN00110143 CN 00110143 CN 00110143 A CN00110143 A CN 00110143A CN 1309897 A CN1309897 A CN 1309897A
- Authority
- CN
- China
- Prior art keywords
- compound
- methyl
- formula
- comparative compound
- halogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Landscapes
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
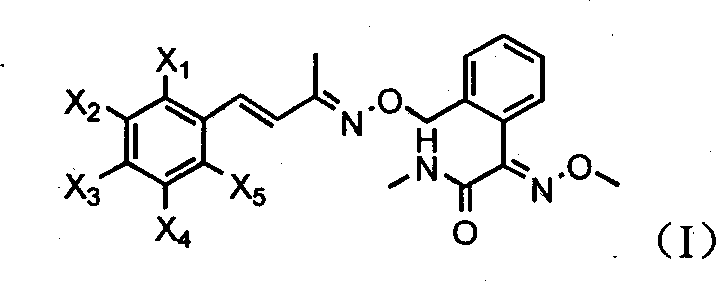
本发明涉及通式(Ⅰ)的不饱和肟醚结构及其立体异构体的杀菌化合物。式中X1-X5选自氢,卤素,烷基,烷氧基,硝基,卤烷基。与现有的不饱和肟醚类杀菌剂相比,它们具有更优良的杀菌活性。
The present invention relates to the bactericidal compound of the unsaturated oxime ether structure of the general formula (I) and its stereoisomer. In the formula, X 1 -X 5 are selected from hydrogen, halogen, alkyl, alkoxy, nitro, haloalkyl. Compared with the existing unsaturated oxime ether fungicides, they have better fungicidal activity.
Description
本发明属农用杀菌剂。The invention belongs to agricultural fungicide.
在中国专利公开CN1191670A中报导了具有不饱和肟醚结构的杀菌剂。这些不饱和肟醚结构的化合物具有广谱活性——可用于防治在各种作物上的由藻菌纲、卵菌纲、子囊菌纲和半知菌纲等多种真菌引起的病害。但是,现代农业和园艺上的发展要求我们不断创新,开发出使用剂量更小、药效更突出的农用杀菌剂。A fungicide with an unsaturated oxime ether structure is reported in Chinese patent publication CN1191670A. These compounds with unsaturated oxime ether structure have broad-spectrum activity—it can be used to prevent and control diseases caused by various fungi such as Phycomycetes, Oomycetes, Ascomycetes and Deuteromycetes on various crops. However, the development of modern agriculture and horticulture requires us to continue to innovate and develop agricultural fungicides with smaller doses and more outstanding efficacy.
为了获得在更小的剂量下就可以控制各种真菌病害的有效化合物,本发明者们在前述发明的基础上继续进行了大量的研究工作,合成、筛选出具有通式(Ⅰ)的不饱和肟醚结构的化合物。这些化合物具有广谱的杀菌活性,与现有技术中所公开的化合物相比,它们具有更高的杀菌活性,在更低的剂量下具有良好的防效,从而完成了本发明。In order to obtain effective compounds that can control various fungal diseases at a smaller dose, the inventors have continued to carry out a large amount of research work on the basis of the aforementioned inventions, and synthesized and screened out unsaturated compounds with general formula (I). Compounds with an oxime ether structure. These compounds have broad-spectrum bactericidal activity, and compared with the compounds disclosed in the prior art, they have higher bactericidal activity and good control effect at a lower dosage, thus completing the present invention.
本发明涉及可用作杀菌剂的不饱和肟醚的衍生物、它们的制备方法和含有这些化合物的组合物。The present invention relates to derivatives of unsaturated oxime ethers useful as fungicides, processes for their preparation and compositions containing these compounds.
本发明提供具有通式(Ⅰ)的不饱和肟醚化合物及其立体异构体: The present invention provides unsaturated oxime ether compounds with general formula (I) and stereoisomers thereof:
式中X1-X5选自氢,卤素,烷基,烷氧基,硝基,卤烷基。In the formula, X 1 -X 5 are selected from hydrogen, halogen, alkyl, alkoxy, nitro, haloalkyl.
卤素包括氟、氯、溴和碘。Halogen includes fluorine, chlorine, bromine and iodine.
在本发明的化合物中,由于碳-碳双键和碳-氮双键连接不同的取代基而可以形成几何异构体(分别以E和Z来表示不同的构型),本发明包括E型异构体和Z型异构体及其任何比例的混合物。In the compounds of the present invention, due to the carbon-carbon double bond and the carbon-nitrogen double bond connecting different substituents can form geometric isomers (respectively use E and Z to represent different configurations), and the present invention includes E-type isomers and Z-isomers and mixtures thereof in any proportion.
可以用下面表(Ⅰ)中的化合物来说明本发明,但并不限定本发明。 The present invention can be illustrated by the compounds in Table (I) below, but the present invention is not limited.
表(Ⅰ)
本发明通式(Ⅰ)的化合物可以通过下述反应式所示的步骤来制备。其中X的定义同上,Z是离去基团,如卤素(氯、溴或碘)。 The compound of the general formula (I) of the present invention can be prepared by the steps shown in the following reaction formula. wherein X is as defined above and Z is a leaving group such as halogen (chloro, bromo or iodo).
通式(Ⅳ)的化合物可以这样来制备:在适当的溶剂中如二甲基甲酰胺、四氢呋喃、丙酮或丁酮,用适当的碱如氢氧化钠、甲醇钠或氢化钠处理通式(Ⅲ)所示的肟,然后加入通式(Ⅱ)的化合物。Compounds of general formula (IV) can be prepared by treating general formula (III ) shown in the oxime, and then add the compound of general formula (II).
通式(Ⅲ)的肟可以由相应的α、β不饱和酮与盐酸羟胺在适当的溶剂(如甲醇或乙醇)中,并有适当的碱(如氢氧化钠或碳酸钾)存在下反应来获得。The oxime of general formula (Ⅲ) can be by corresponding α, β unsaturated ketone and hydroxylamine hydrochloride in suitable solvent (such as methanol or ethanol), and there is the reaction under the existence of suitable base (such as sodium hydroxide or potassium carbonate) get.
欧洲专利申请EP254426中叙述了Z是溴的(E)构型的通式(Ⅱ)的化合物。European patent application EP254426 describes compounds of general formula (II) in (E) configuration in which Z is bromine.
通式(Ⅰ)的化合物可以由通式(Ⅳ)的化合物与甲胺水溶液反应很容易的获得。The compound of the general formula (I) can be easily obtained by reacting the compound of the general formula (IV) with an aqueous solution of methylamine.
本发明的化合物是更有效的杀菌剂。它们具有内吸活性并可用作叶面和土壤杀菌剂。特别适合于防治下列植物病害:The compounds of the present invention are more effective fungicides. They are systemically active and useful as foliar and soil fungicides. Especially suitable for the control of the following plant diseases:
水稻稻瘟病(Pyricularia oryzae);小麦条锈病(Puccinia striiformis)、叶锈病(Puccinia recondita)和其它锈病;大麦条锈病(Puccinia striiformis)、叶锈病(Puccinia recandita)和其它锈病;大麦和小麦白粉病、黄瓜白粉病、苹果白粉病和葡萄白粉病。Rice blast (Pyricularia oryzae); wheat stripe rust (Puccinia striiformis), leaf rust (Puccinia recondita) and other rusts; barley stripe rust (Puccinia striiformis), leaf rust (Puccinia recandita) and other rusts; barley and wheat powdery mildew, Cucumber powdery mildew, apple powdery mildew and grape powdery mildew.
小麦纹枯病和颖枯病(Septoria nodorum)。谷物上的长蠕孢、嘴孢霉、壳针孢属病、核球壳菌属病、Pseudocercosporella herpotrichoides和Gaeumannomyces graminis。花生Cercospora arachidicola和Cercosporidiumpersonata甜菜、大豆和稻谷上的其它尾孢霉属病。番茄、黄瓜、葡萄灰霉病(Botrytic cinerea)。蔬菜(如黄瓜)上的铰链孢属病。黄瓜上的炭疽病,苹果黑星病,黄瓜霜霉病,葡萄霜霉病,马铃薯和番茄上的疫霉,稻谷上的Thanatephorus cucumeris以及其它宿主如小麦和大麦、蔬菜上的其它丝核菌。Wheat sheath blight and glume blight (Septoria nodorum). Helminthias elongatum, Cephalosporium, Septoria, Sclerotinia, Pseudocercosporella herpotrichoides and Gaeumannomyces graminis on cereals. Cercospora arachidicola on peanuts and Cercosporidium personata on sugar beets, soybeans and other Cercospora spp. Tomatoes, cucumbers, Botrytic cinerea. Artichokes on vegetables such as cucumbers. Anthracnose on cucumber, apple scab, downy mildew on cucumber, downy mildew on grape, Phytophthora on potato and tomato, Thanatephorus cucumeris on rice and other Rhizoctonia on other hosts such as wheat and barley, vegetables.
本发明还提供了如上所定义的组合物的制备方法,方法是将通式(Ⅰ)的化合物和至少一种载体混合。这种组合物可以含本发明的单一化合物或几种化合物的混合物。The invention also provides a process for the preparation of a composition as defined above by mixing a compound of general formula (I) and at least one carrier. Such compositions may contain a single compound of the invention or a mixture of several compounds.
按照本发明的组合物,优选含从1-99%重量的活性成分。本发明的载体为满足下述条件的物质:它与活性成分配制后便于施用于待处理的位点,例如可以是植物、种子或土壤;或者有利于贮存、运输或操作。载体可以是固体或液体,包括通常为气体但已压缩成液体的物质和可使用任何通常在配制杀虫、杀菌组合物中所用的载体。The compositions according to the invention preferably contain from 1 to 99% by weight of active ingredient. The carrier of the present invention is a substance that satisfies the following conditions: it can be conveniently applied to the locus to be treated after being formulated with the active ingredient, for example, it can be a plant, seed or soil; or it can be convenient for storage, transportation or handling. The carrier may be solid or liquid, including substances that are usually gaseous but compressed into liquid, and any carrier commonly used in formulating insecticidal and fungicidal compositions may be used.
合适的固体载体包括天然和合成的黏土和硅酸盐,例如硅藻土、滑石、硅镁土、硅酸铝(高岭土)、蒙脱石和云母、碳酸钙、硫酸钙、硫酸铵;合成的氧化硅和合成硅酸钙或硅酸铝;元素如碳和硫;天然的和合成的树脂如苯并呋喃树脂,聚氯乙烯和苯乙烯聚合物和共聚物;固体多氯苯酚;沥青;蜡如蜂蜡,石蜡。Suitable solid carriers include natural and synthetic clays and silicates such as diatomaceous earth, talc, attapulgite, aluminum silicate (kaolin), montmorillonite and mica, calcium carbonate, calcium sulfate, ammonium sulfate; Silicon and synthetic calcium or aluminum silicates; elements such as carbon and sulfur; natural and synthetic resins such as benzofuran resins, polyvinyl chloride and styrene polymers and copolymers; solid polychlorinated phenols; bitumen; waxes such as Beeswax, paraffin.
合适的液体载体包括水;醇如异丙醇和乙醇;酮如丙酮、甲基乙基酮、甲基异丁基酮和环己基酮;醚;芳烃如苯、甲苯和二甲苯;石油馏分如煤油和矿物油;氯代烃如四氯化碳、全氯乙烯和三氯乙烷。通常,这些液体的混合物也是合适的。Suitable liquid carriers include water; alcohols such as isopropanol and ethanol; ketones such as acetone, methyl ethyl ketone, methyl isobutyl ketone, and cyclohexyl ketone; ethers; aromatic hydrocarbons such as benzene, toluene, and xylene; and mineral oil; chlorinated hydrocarbons such as carbon tetrachloride, perchlorethylene and trichloroethane. Often, mixtures of these liquids are also suitable.
杀虫、杀菌组合物通常在施用之前将其稀释。少量的表面活性剂载体的存在有助于稀释过程。在本发明的组合物中,至少含有一种载体,优选以表面活性剂作载体。例如组合物可含有至少两种载体,其中至少一种是表面活性剂。Insecticidal and fungicidal compositions are usually diluted prior to application. The presence of a small amount of surfactant carrier aids in the dilution process. In the composition of the present invention, at least one carrier is contained, preferably a surfactant is used as the carrier. For example the composition may contain at least two carriers, at least one of which is a surfactant.
表面活性剂可以是乳化剂、分散剂或润湿剂;它可以是非离子的或离子的表面活性剂。合适的表面活性剂包括聚丙烯酸和木质素磺酸的钠盐或钙盐;分子中含至少12个碳原子的脂肪酸或脂肪胺或酰胺与环氧乙烷和/或环氧丙烷的缩合物;甘醇,山梨醇,蔗糖或季戊四醇脂肪酸酯及这些酯与环氧乙烷和/或环氧丙烷的缩合物;脂肪醇或烷基苯酚如对辛基苯酚或对辛基甲苯酚与环氧乙烷和/或环氧丙烷的缩合物;这些缩合产物的硫酸盐或磺酸盐;在分子中含有至少10个碳原子的硫酸或磺酸酯的碱金属或碱土金属盐,优选钠盐;例如硫酸月桂酯钠,硫酸仲烷基酯钠,磺化蓖麻油钠盐,磺酸烷基芳基酯钠如十二烷基苯磺酸钠盐。A surfactant can be an emulsifying agent, a dispersing agent or a wetting agent; it can be a nonionic or an ionic surfactant. Suitable surfactants include sodium or calcium salts of polyacrylic acid and lignosulfonic acid; condensation products of fatty acids or fatty amines or amides with at least 12 carbon atoms in the molecule and ethylene oxide and/or propylene oxide; Glycol, sorbitol, sucrose or pentaerythritol fatty acid esters and condensates of these esters with ethylene oxide and/or propylene oxide; fatty alcohols or alkylphenols such as p-octylphenol or p-octylcresol with epoxy Condensates of ethane and/or propylene oxide; sulfates or sulfonates of these condensation products; alkali metal or alkaline earth metal salts, preferably sodium salts, of sulfuric acid or sulfonates containing at least 10 carbon atoms in the molecule; For example sodium lauryl sulfate, sodium secondary alkyl sulfate, sodium sulfonated castor oil, sodium alkylaryl sulfonate such as sodium dodecylbenzenesulfonate.
本发明的组合物的实例是可湿性粉剂、粉剂、颗粒剂和溶液,可乳化的浓缩剂、乳剂、悬浮浓缩剂、气雾剂和烟雾剂。可湿性粉剂通常含25,50或75%(重量)活性成分,且通常除固体惰性载体之外,还含有3-10%(重量)的分散剂,且若需要可加入0-10%(重量)稳定剂和/或其它添加剂如渗透剂或粘着剂。粉剂通常可成型为具有与可湿性粉剂相似的组成但没有分散剂的粉剂浓缩剂,在地里进一步用固体载体稀释,得到通常含0.5-10%(重量)活性成分的组合物。粒剂通常制备成具有10和100目(1.676-0.152mm)大小,且可用成团或注入技术制备。通常,粒剂含0.5-75%(重量)的活性成分和0-10%(重量)添加剂如稳定剂、表面活性剂、缓释改良剂。所谓的“可流动干粉”由具有相对高浓度活性成分的相对小的颗粒组成。可乳化浓缩剂除溶剂外,当需要时通常含有共溶剂,1-50%W/V活性成分,2-20%W/V乳化剂和0-20%W/V其它添加剂如稳定剂、渗透剂和腐蚀抑制剂,悬浮浓缩剂通常含有10-75%(重量)的活性成分、0.5-15%(重量)的分散剂、0.1-10%(重量)的其它添加剂如消泡剂、腐蚀抑制剂、稳定剂、渗透剂和粘着剂。Examples of compositions according to the invention are wettable powders, powders, granules and solutions, emulsifiable concentrates, emulsions, suspension concentrates, aerosols and aerosols. Wettable powders usually contain 25, 50 or 75% (by weight) of active ingredient, and usually contain 3-10% (by weight) of dispersant in addition to solid inert carrier, and 0-10% (by weight) can be added if necessary. ) stabilizers and/or other additives such as penetrants or adhesives. Dusts are usually formulated as powder concentrates having a composition similar to wettable powders but without dispersants, which are further diluted in the field with solid carriers to give compositions usually containing 0.5-10% by weight of active ingredient. Granules are typically prepared in sizes of 10 and 100 mesh (1.676-0.152 mm), and may be prepared by agglomeration or injection techniques. Typically, granules contain 0.5-75% by weight of active ingredient and 0-10% by weight of additives such as stabilizers, surfactants, slow-release modifiers. So-called "dry flowable powders" consist of relatively small granules having a relatively high concentration of active ingredient. Emulsifiable concentrates usually contain, in addition to solvents, co-solvents when required, 1-50% W/V active ingredients, 2-20% W/V emulsifiers and 0-20% W/V other additives such as stabilizers, penetrants Agents and corrosion inhibitors, suspension concentrates usually contain 10-75% (weight) of active ingredients, 0.5-15% (weight) of dispersants, 0.1-10% (weight) of other additives such as antifoaming agents, corrosion inhibitors agent, stabilizer, penetrant and adhesive.
水分散剂和乳剂,例如通过用水稀释按照本发明的可湿性粉剂或浓缩物得到的组合物,也列入本发明范围。所说的乳剂可具有油包水或水包油两个类型。Aqueous dispersions and emulsions, such as compositions obtained by diluting the wettable powders or concentrates according to the invention with water, are also within the scope of the invention. Said emulsions may be of the water-in-oil or oil-in-water type.
通过在组合物中加入其它的一种或多种杀菌剂,使其能比单独的通式(Ⅰ)化合物具有更广谱的活性。此外,其它杀菌剂可对通式(Ⅰ)化合物的杀菌活性具有增效作用。可以包含在本发明组合物中的杀菌剂化合物的实例有:腈菌唑、粉锈宁、苯菌灵、多菌灵、百菌清、王铜、波尔多液、麦穗宁、双胍辛盐、土菌消、富士一号、春雷霉素、代森锰锌、代森锰、代森锌、多氧霉素、甲基代森锌、甲基托布津、福美双、十三吗啉、烯酰吗啉等。By adding one or more other fungicides to the composition, it can have a wider spectrum of activity than the compound of general formula (I) alone. In addition, other fungicides may have a synergistic effect on the fungicidal activity of the compounds of general formula (I). Examples of fungicide compounds that may be included in the compositions of the present invention are: myclobutanil, triclofenac, benomyl, carbendazim, chlorothalonil, king copper, Bordeaux mixture, fennel, biguanide octyl salt, soil fungus Xiao, Fuji No. 1, Kasugamycin, Mancozeb, Maneb, Zinc, Doxymycin, Zinc Methyl, Thioprazine Methyl, Thiram, Tridemorph, Dienoyl phylloline etc.
可以与本发明的化合物混合形成组合物的合适杀虫剂包括乐果、氧化乐果、毒死蜱、甲基毒死蜱、甲胺磷、久效磷、倍硫磷、辛硫磷、唑蚜威、吡虫啉和拟除虫菊酯类如氰戊菊酯、甲氰菊酯、氟胺氰菊酯、氯氟氰菊酯等。Suitable insecticides which may be mixed with the compounds of the present invention to form compositions include dimethoate, omethoate, chlorpyrifos, chlorpyrifos-methyl, methamidophos, monocrotophos, fenthion, phoxim, pyraflucarb, imidacloprid And pyrethroids such as fenvalerate, fenpropathrin, fluvalinate, cyhalothrin, etc.
本发明的化合物与现有技术中所公开的不饱和肟醚类杀菌剂相比具有更好的杀菌活性。中国专利公开CN1191670A中的化合物对各种真菌病害的防效试验其待测化合物浓度为100mg/L时是有效的;而本发明所提供的化合物当其浓度降为对照化合物的1/4甚至1/16时,其杀菌活性仍大大优于对照化合物。这种优异的杀菌效果使本发明的发明者达到了发明一种在更小的剂量下就可以控制各种真菌病害的有效化合物的目的,在不饱和肟醚类杀菌剂领域中完成了又一项重要发明。Compared with the unsaturated oxime ether fungicides disclosed in the prior art, the compound of the present invention has better fungicidal activity. The compounds in the Chinese Patent Publication CN1191670A are effective when the concentration of the compound to be tested is 100 mg/L in the control effect test of various fungal diseases; and the compounds provided by the present invention are 1/4 or even 1 /16, its bactericidal activity is still much better than the reference compound. This excellent bactericidal effect has enabled the inventors of the present invention to achieve the purpose of inventing an effective compound that can control various fungal diseases at a smaller dose, and has completed another step in the field of unsaturated oxime ether fungicides. an important invention.
下面以具体的实施例来说明本发明,但并不限定本发明。The present invention is illustrated below with specific examples, but the present invention is not limited.
合成实施例Synthetic example
实例1 化合物1的合成Synthesis of Example 1 Compound 1
17.5克(0.1摩尔)2,6-二氯苯甲醛溶于100毫升丙酮中,向其中滴加5克10%的氢氧化钠水溶液,滴加过程中保持温度不超过40℃。滴加完继续搅拌一小时。混合物倒入水中,用乙酸乙酯提取,再用水洗涤提取物三次,干燥浓缩得浅黄色油状物即2,6-二氯苯乙烯基甲基酮20.4克,收率94.9%。17.5 g (0.1 mole) of 2,6-dichlorobenzaldehyde was dissolved in 100 ml of acetone, and 5 g of 10% aqueous sodium hydroxide solution was added dropwise thereto, and the temperature was kept not exceeding 40° C. during the dropwise addition. Stirring was continued for one hour after the dropwise addition. The mixture was poured into water, extracted with ethyl acetate, washed with water three times, dried and concentrated to obtain 20.4 g of a light yellow oily substance, namely 2,6-dichlorostyryl methyl ketone, with a yield of 94.9%.
将10克(0.0465摩尔)2,6-二氯苯乙烯基甲基酮、7.77克盐酸羟胺(0.093摩尔)和12.9克(0.093摩尔)碳酸钾在150毫升乙醇中回流3小时。将反应混合物浓缩,用水稀释并用乙酸乙酯提取。合并的提取液干燥浓缩,得2,6-二氯苯乙烯基甲基酮肟9.3克,为浅黄色固体。收率86.93%。10 g (0.0465 mol) of 2,6-dichlorostyryl methyl ketone, 7.77 g of hydroxylamine hydrochloride (0.093 mol) and 12.9 g (0.093 mol) of potassium carbonate were refluxed in 150 ml of ethanol for 3 hours. The reaction mixture was concentrated, diluted with water and extracted with ethyl acetate. The combined extracts were dried and concentrated to obtain 9.3 g of 2,6-dichlorostyryl methyl ketone oxime as a light yellow solid. Yield 86.93%.
在室温下将2.3克(0.01摩尔)2,6-二氯苯乙烯基甲基酮肟的N,N-二甲基甲酰胺溶液(10毫升)滴加到含有0.4克(0.01摩尔)氢氧化钠的N,N-二甲基甲酰胺悬浮液(10毫升)中。半小时后,将2.86克(0.01摩尔)(E)-2-(溴甲基)苯酰基甲酸甲酯-O-甲基肟的N,N-二甲基甲酰胺溶液(10毫升)加到反应混合物中,在室温下继续搅拌2小时。将混合物倒入水中,用乙酸乙酯提取2次。合并的提取液用水洗涤3次,然后干燥、浓缩,得粗产品。用乙酸乙酯和石油醚的混合液(1∶5)为洗脱液进行柱层析得到浅黄色油状物1.28克,收率29.43%。At room temperature, 2.3 g (0.01 mol) of 2,6-dichlorostyryl methyl ketoxime in N,N-dimethylformamide (10 ml) was added dropwise to 0.4 g (0.01 mol) of Sodium N,N-dimethylformamide suspension (10 mL). Half an hour later, a solution of 2.86 g (0.01 mol) of (E)-methyl 2-(bromomethyl)benzoylformate-O-methyloxime in N,N-dimethylformamide (10 ml) was added to Stirring of the reaction mixture was continued at room temperature for 2 hours. The mixture was poured into water and extracted twice with ethyl acetate. The combined extracts were washed 3 times with water, then dried and concentrated to obtain a crude product. Column chromatography was performed using a mixture of ethyl acetate and petroleum ether (1:5) as the eluent to obtain 1.28 g of light yellow oil, with a yield of 29.43%.
将0.75克(0.00172摩尔)上述化合物与3倍摩尔比的甲胺40%水溶液在50毫升甲醇中加热回流过夜,浓缩后用乙酸乙酯提取2次,合并的提取物用水洗涤3次,然后干燥,浓缩,得到标题化合物0.55克,为白色固体,收率73.3%。熔点131-133℃0.75 g (0.00172 moles) of the above compound and 40% aqueous solution of methylamine in a molar ratio of 3 times were heated to reflux overnight in 50 ml of methanol, extracted twice with ethyl acetate after concentration, the combined extracts were washed with water three times, and then dried , and concentrated to obtain 0.55 g of the title compound as a white solid with a yield of 73.3%. Melting point 131-133°C
核磁共振数据:2.03(3H单峰)2.82-2.87(3H双峰)3.87(3H单峰)4.97(2H单峰)6.5-6.6(1H多峰)6.71(2H单峰)6.80-7.50(7H多峰)NMR data: 2.03 (3H unimodal) 2.82-2.87 (3H bimodal) 3.87 (3H unimodal) 4.97 (2H unimodal) 6.5-6.6 (1H multimodal) 6.71 (2H unimodal) 6.80-7.50 (7H multimodal peak)
实例2 化合物2的合成Example 2 Synthesis of compound 2
17.5克(0.1摩尔)2,3-二氯苯甲醛溶在100毫升丙酮中,向内滴加5克10%的氢氧化钠水溶液,在滴加过程中保持温度不超过40℃,之后继续搅拌一小时。混合物倒入水中,加入乙酸乙酯提取,用水洗涤三次,干燥浓缩得浅黄色油状物2,3-二氯苯乙烯基甲基酮20.2克,收率93.9%。17.5 grams (0.1 moles) of 2,3-dichlorobenzaldehyde were dissolved in 100 milliliters of acetone, and 5 grams of 10% sodium hydroxide aqueous solution was added dropwise, and the temperature was kept no more than 40 ° C during the dropping process, and then continued to stir One hour. The mixture was poured into water, extracted with ethyl acetate, washed with water three times, dried and concentrated to obtain 20.2 g of light yellow oily 2,3-dichlorostyryl methyl ketone, with a yield of 93.9%.
将10克(0.0465摩尔)2,3-二氯苯乙烯基甲基酮、7.77克盐酸羟胺(0.093摩尔)和12.9克(0.093摩尔)碳酸钾在150毫升乙醇中回流3小时。将反应混合物浓缩,用水稀释并用乙酸乙酯提取。合并的提取液干燥浓缩,得2,3-二氯苯乙烯基甲基酮肟9.5克,为浅黄色固体,收率88.8%。10 g (0.0465 mol) of 2,3-dichlorostyryl methyl ketone, 7.77 g of hydroxylamine hydrochloride (0.093 mol) and 12.9 g (0.093 mol) of potassium carbonate were refluxed in 150 ml of ethanol for 3 hours. The reaction mixture was concentrated, diluted with water and extracted with ethyl acetate. The combined extracts were dried and concentrated to obtain 9.5 g of 2,3-dichlorostyryl methyl ketone oxime as a light yellow solid with a yield of 88.8%.
在室温下将2.3克(0.01摩尔)2,3-二氯苯乙烯基甲基酮肟的N,N-二甲基甲酰胺溶液(10毫升)滴加到含有0.4克(0.01摩尔)氢氧化钠的N,N-二甲基甲酰胺悬浮液(10毫升)中。半小时后,将2.86克(0.01摩尔)(E)-2-(溴甲基)苯酰基甲酸甲酯-O-甲基肟的N,N-二甲基甲酰胺溶液(10毫升)加到反应混合物中,在室温下继续搅拌2小时。将混合物倒入水中,用乙酸乙酯提取2次。合并的提取液用水洗涤3次,然后干燥、浓缩,得粗产品。用乙酸乙酯和石油醚的混合液(1∶5)为洗脱液进行柱层析得到浅黄色油状物1.38克,收率31.73%。At room temperature, 2.3 g (0.01 mol) of 2,3-dichlorostyryl methyl ketoxime in N,N-dimethylformamide (10 ml) was added dropwise to 0.4 g (0.01 mol) of hydroxide Sodium N,N-dimethylformamide suspension (10 mL). Half an hour later, a solution of 2.86 g (0.01 mol) of (E)-methyl 2-(bromomethyl)benzoylformate-O-methyloxime in N,N-dimethylformamide (10 ml) was added to Stirring of the reaction mixture was continued at room temperature for 2 hours. The mixture was poured into water and extracted twice with ethyl acetate. The combined extracts were washed 3 times with water, then dried and concentrated to obtain a crude product. Column chromatography was performed using a mixture of ethyl acetate and petroleum ether (1:5) as the eluent to obtain 1.38 g of light yellow oil, with a yield of 31.73%.
将0.75克(0.00172摩尔)上述化合物与3倍摩尔比的甲胺40%水溶液于50毫升甲醇中加热回流过夜,浓缩后用乙酸乙酯提取2次,合并的提取物用水洗涤3次,然后干燥、浓缩,得到标题化合物0.65克,为粘稠油状物,收率86.63%。0.75 g (0.00172 moles) of the above compound and 40% aqueous solution of methylamine in a 3-fold molar ratio were heated to reflux overnight in 50 ml of methanol, extracted twice with ethyl acetate after concentration, and the combined extracts were washed three times with water, and then dried , and concentrated to obtain 0.65 g of the title compound as viscous oil with a yield of 86.63%.
核磁共振数据:2.04(3H单峰)2.82-2.87(3H双峰)3.89(3H单峰)5.01(2H单峰)6.5-6.6(1H多峰)6.70(2H单峰)6.80-7.50(7H多峰)NMR data: 2.04 (3H unimodal) 2.82-2.87 (3H bimodal) 3.89 (3H unimodal) 5.01 (2H unimodal) 6.5-6.6 (1H multimodal) 6.70 (2H unimodal) 6.80-7.50 (7H multimodal peak)
实例3 化合物3的合成Example 3 Synthesis of compound 3
17.5克(0.1摩尔)2,4-二氯苯甲醛溶于100毫升丙酮中,向其中滴加5克10%的氢氧化钠水溶液,滴加过程中保持温度不超过40℃,之后继续搅拌一小时。混合物倒入水中,加入乙酸乙酯提取,再用水洗涤三次,干燥浓缩得浅黄色油状物2,4-二氯苯乙烯基甲基酮20.6克,收率95.8%。17.5 grams (0.1 moles) of 2,4-dichlorobenzaldehyde were dissolved in 100 milliliters of acetone, and 5 grams of 10% sodium hydroxide aqueous solution was added dropwise thereto, and the temperature was kept no more than 40°C during the dropping process, and then continued to stir for a while. Hour. The mixture was poured into water, extracted with ethyl acetate, washed with water three times, dried and concentrated to obtain 20.6 g of light yellow oily 2,4-dichlorostyryl methyl ketone with a yield of 95.8%.
将10克(0.0465摩尔)2,4-二氯苯乙烯基甲基酮、7.77克盐酸羟胺(0.093摩尔)和12.9克(0.093摩尔)碳酸钾在150毫升乙醇中回流3小时。将反应混合物浓缩,用水稀释并用乙酸乙酯提取。合并的提取液干燥浓缩,得2,4-二氯苯乙烯基甲基酮肟9.1克,为浅黄色固体,收率85.06%。10 g (0.0465 mol) of 2,4-dichlorostyryl methyl ketone, 7.77 g of hydroxylamine hydrochloride (0.093 mol) and 12.9 g (0.093 mol) of potassium carbonate were refluxed in 150 ml of ethanol for 3 hours. The reaction mixture was concentrated, diluted with water and extracted with ethyl acetate. The combined extracts were dried and concentrated to obtain 9.1 g of 2,4-dichlorostyryl methyl ketone oxime as a light yellow solid with a yield of 85.06%.
在室温下将2.3克(0.01摩尔)2,4-二氯苯乙烯基甲基酮肟的N,N-二甲基甲酰胺溶液(10毫升)滴加到含有0.4克(0.01摩尔)氢氧化钠的N,N-二甲基甲酰胺悬浮液(10毫升)中。半小时后,将2.86克(0.01摩尔)(E)-2-(溴甲基)苯酰基甲酸甲酯-O-甲基肟的N,N-二甲基甲酰胺溶液(10毫升)加到反应混合物中,在室温下继续搅拌2小时。将混合物倒入水中,用乙酸乙酯提取2次。合并的提取液用水洗涤3次,然后干燥、浓缩,得粗产品。用乙酸乙酯和石油醚的混合液(1∶5)为洗脱液进行柱层析得到浅黄色油状物1.35克,收率31.04%。At room temperature, 2.3 g (0.01 mol) of 2,4-dichlorostyryl methyl ketoxime in N,N-dimethylformamide solution (10 ml) was added dropwise to 0.4 g (0.01 mol) of hydroxide Sodium N,N-dimethylformamide suspension (10 mL). Half an hour later, a solution of 2.86 g (0.01 mol) of (E)-methyl 2-(bromomethyl)benzoylformate-O-methyloxime in N,N-dimethylformamide (10 ml) was added to Stirring of the reaction mixture was continued at room temperature for 2 hours. The mixture was poured into water and extracted twice with ethyl acetate. The combined extracts were washed 3 times with water, then dried and concentrated to obtain a crude product. Column chromatography was performed using a mixture of ethyl acetate and petroleum ether (1:5) as the eluent to obtain 1.35 g of light yellow oil, with a yield of 31.04%.
将0.75克(0.00172摩尔)上述化合物与3倍摩尔比的甲胺40%水溶液在50毫升甲醇中加热回流过夜,浓缩后用乙酸乙酯提取2次,合并的提取物用水洗涤3次,然后干燥、浓缩,得到标题化合物0.63克,为粘稠油状物,收率83.96%。0.75 g (0.00172 moles) of the above compound and 40% aqueous solution of methylamine in a molar ratio of 3 times were heated to reflux overnight in 50 ml of methanol, extracted twice with ethyl acetate after concentration, the combined extracts were washed with water three times, and then dried , and concentrated to obtain 0.63 g of the title compound as viscous oil with a yield of 83.96%.
核磁共振数据:2.03(3H单峰)2.82-2.87(3H双峰)3.88(3H单峰)5.01(2H单峰)6.4-6.6(1H多峰)6.68(2H单峰)6.75-7.50(7H多峰)NMR data: 2.03 (3H unimodal) 2.82-2.87 (3H bimodal) 3.88 (3H unimodal) 5.01 (2H unimodal) 6.4-6.6 (1H multimodal) 6.68 (2H unimodal) 6.75-7.50 (7H multimodal peak)
实例4 化合物17的合成Example 4 Synthesis of compound 17
14.2克(0.1摩尔)2,6-二氟苯甲醛溶在100毫升丙酮中,向内滴加5克10%的氢氧化钠水溶液,在滴加过程中要保持温度不超过40℃,继续搅拌一小时。混合物倒入水中,加入乙酸乙酯提取,用水洗涤三次,干燥浓缩得浅黄色油状物2,6-二氟苯乙烯基甲基酮15.7克,收率86.26%。14.2 grams (0.1 moles) of 2,6-difluorobenzaldehyde were dissolved in 100 milliliters of acetone, and 5 grams of 10% sodium hydroxide aqueous solution was added dropwise, and the temperature should be kept no more than 40°C during the dropping process, and the stirring was continued One hour. The mixture was poured into water, extracted with ethyl acetate, washed with water three times, dried and concentrated to obtain 15.7 g of light yellow oily 2,6-difluorostyryl methyl ketone, with a yield of 86.26%.
将8.46克(0.0465摩尔)2,6-二氟苯乙烯基甲基酮、7.77克盐酸羟胺(0.093摩尔)和12.9克(0.093摩尔)碳酸钾在150毫升乙醇中回流3小时。将反应混合物浓缩,用水稀释并用乙酸乙酯提取。合并的提取液干燥浓缩,得2,6-二氟苯乙烯基甲基酮肟7.91克,为浅黄色固体。收率86.37%。8.46 g (0.0465 mol) of 2,6-difluorostyryl methyl ketone, 7.77 g of hydroxylamine hydrochloride (0.093 mol) and 12.9 g (0.093 mol) of potassium carbonate were refluxed in 150 ml of ethanol for 3 hours. The reaction mixture was concentrated, diluted with water and extracted with ethyl acetate. The combined extracts were dried and concentrated to obtain 7.91 g of 2,6-difluorostyryl methyl ketone oxime as a light yellow solid. Yield 86.37%.
在室温下将1.97克(0.01摩尔)2,6-二氟苯乙烯基甲基酮肟的N,N-二甲基甲酰胺溶液(10毫升)滴加到含有0.4克(0.01摩尔)氢氧化钠的N,N-二甲基甲酰胺悬浮液(10毫升)中。半小时后,将2.86克(0.01摩尔)(E)-2-(溴甲基)苯酰基甲酸甲酯-O-甲基肟的N,N-二甲基甲酰胺溶液(10毫升)加到反应混合物中,在室温下继续搅拌2小时。将混合物倒入水中,用乙酸乙酯提取2次。合并的提取液用水洗涤3次,然后干燥,浓缩,得粗产品。用乙酸乙酯和石油醚的混合液(1∶5)为洗脱液进行柱层析得到浅黄色油状物1.18克,收率29.35%。At room temperature, 1.97 g (0.01 mol) of 2,6-difluorostyryl methyl ketoxime in N,N-dimethylformamide solution (10 ml) was added dropwise to 0.4 g (0.01 mol) of hydroxide Sodium N,N-dimethylformamide suspension (10 mL). Half an hour later, a solution of 2.86 g (0.01 mol) of (E)-methyl 2-(bromomethyl)benzoylformate-O-methyloxime in N,N-dimethylformamide (10 ml) was added to Stirring of the reaction mixture was continued at room temperature for 2 hours. The mixture was poured into water and extracted twice with ethyl acetate. The combined extracts were washed 3 times with water, then dried and concentrated to obtain a crude product. Column chromatography was performed using a mixture of ethyl acetate and petroleum ether (1:5) as the eluent to obtain 1.18 g of light yellow oil, with a yield of 29.35%.
将0.75克(0.00186摩尔)上述化合物与3倍摩尔比的甲胺40%水溶液在50毫升甲醇中加热回流过夜,浓缩后用乙酸乙酯提取2次,合并的提取物用水洗涤3次,然后干燥、浓缩,得到标题化合物0.51克,为粘稠油状物,收率68.17%。0.75 g (0.00186 moles) of the above compound and 40% aqueous solution of methylamine in a 3-fold molar ratio were heated to reflux overnight in 50 ml of methanol, extracted twice with ethyl acetate after concentration, and the combined extracts were washed with water three times, then dried , and concentrated to obtain 0.51 g of the title compound as viscous oil with a yield of 68.17%.
核磁共振数据:2.01(3H单峰)2.82-2.88(3H双峰)3.87(3H单峰)4.97(2H单峰)6.6-7.40(10H多峰)NMR data: 2.01 (3H unimodal) 2.82-2.88 (3H bimodal) 3.87 (3H unimodal) 4.97 (2H unimodal) 6.6-7.40 (10H multimodal)
实例5 化合物39的合成Example 5 Synthesis of compound 39
15.8克(0.1摩尔)2-氯-6-氟苯甲醛溶于100毫升丙酮中,向其中滴加5克10%的氢氧化钠水溶液,滴加过程中保持温度不超过40℃,之后继续搅拌一小时。混合物倒入水中,加入乙酸乙酯提取,用水洗涤三次,干燥、浓缩得浅黄色油状物2-氯-6-氟苯乙烯基甲基酮15.7克,收率79.34%。15.8 grams (0.1 moles) of 2-chloro-6-fluorobenzaldehyde were dissolved in 100 milliliters of acetone, and 5 grams of 10% aqueous sodium hydroxide solution was added dropwise thereto, and the temperature was kept no more than 40°C during the dropping process, and then continued to stir One hour. The mixture was poured into water, extracted with ethyl acetate, washed with water three times, dried and concentrated to obtain 15.7 g of light yellow oily 2-chloro-6-fluorostyryl methyl ketone, with a yield of 79.34%.
将9.23克(0.0465摩尔)2-氯-6-氟苯乙烯基甲基酮、7.77克盐酸羟胺(0.093摩尔)和12.9克(0.093摩尔)碳酸钾在150毫升乙醇中回流3小时。将反应混合物浓缩,用水稀释并用乙酸乙酯提取。合并的提取液干燥浓缩,得2-氯-6-氟苯乙烯基甲基酮肟8.38克,为浅黄色固体。收率84.41%。9.23 g (0.0465 mol) of 2-chloro-6-fluorostyryl methyl ketone, 7.77 g of hydroxylamine hydrochloride (0.093 mol) and 12.9 g (0.093 mol) of potassium carbonate were refluxed in 150 ml of ethanol for 3 hours. The reaction mixture was concentrated, diluted with water and extracted with ethyl acetate. The combined extracts were dried and concentrated to obtain 8.38 g of 2-chloro-6-fluorostyryl methyl ketone oxime as a pale yellow solid. Yield 84.41%.
在室温下将2.14克(0.01摩尔)2-氯-6-氟苯乙烯基甲基酮肟的N,N-二甲基甲酰胺溶液(10毫升)滴加到含有0.4克(0.01摩尔)氢氧化钠的N,N-二甲基甲酰胺悬浮液(10毫升)中。半小时后,将2.86克(0.01摩尔)(E)-2-(溴甲基)苯酰基甲酸甲酯-O-甲基肟的N,N-二甲基甲酰胺溶液(10毫升)加到反应混合物中,室温下继续搅拌2小时。将混合物倒入水中,用乙酸乙酯提取2次。合并的提取液用水洗涤3次,然后干燥、浓缩,得粗产品。用乙酸乙酯和石油醚的混合液(1∶5)为洗脱液进行柱层析得到浅黄色油状物1.24克,收率29.63%。At room temperature, 2.14 g (0.01 mol) of 2-chloro-6-fluorostyryl methyl ketoxime in N,N-dimethylformamide solution (10 ml) was added dropwise to 0.4 g (0.01 mol) hydrogen Sodium oxide in N,N-dimethylformamide suspension (10 mL). Half an hour later, a solution of 2.86 g (0.01 mol) of (E)-methyl 2-(bromomethyl)benzoylformate-O-methyloxime in N,N-dimethylformamide (10 ml) was added to The reaction mixture was stirred at room temperature for 2 hours. The mixture was poured into water and extracted twice with ethyl acetate. The combined extracts were washed 3 times with water, then dried and concentrated to obtain a crude product. Column chromatography was performed using a mixture of ethyl acetate and petroleum ether (1:5) as the eluent to obtain 1.24 g of light yellow oil, with a yield of 29.63%.
将0.75克(0.00179摩尔)上述化合物与3倍摩尔比的甲胺40%水溶液在50毫升甲醇中加热回流过夜,浓缩后用乙酸乙酯提取2次,合并的提取物用水洗涤3次,然后干燥、浓缩,得到标题化合物0.59克,为粘稠油状物,收率78.85%。0.75 g (0.00179 moles) of the above compound and 40% aqueous solution of methylamine in a 3-fold molar ratio were heated to reflux overnight in 50 ml of methanol, extracted twice with ethyl acetate after concentration, and the combined extracts were washed three times with water, then dried , and concentrated to obtain 0.59 g of the title compound as viscous oil with a yield of 78.85%.
核磁共振数据:2.02(3H单峰)2.83-2.88(3H双峰)3.87(3H单峰)4.97(2H单峰)6.4-7.40(10H多峰)NMR data: 2.02 (3H unimodal) 2.83-2.88 (3H bimodal) 3.87 (3H unimodal) 4.97 (2H unimodal) 6.4-7.40 (10H multimodal)
配方实施例recipe example
实例6Example 6
将10份(重量)的化合物1溶解在下述混合物中,该混合物含90份(重量)二甲苯、6份(重量)的壬基酚聚氧乙烯醚、2份(重量)的十二烷基苯磺酸钙和2份(重量)的40摩尔环氧乙烷与1摩尔蓖麻油的加成产物来制备乳油(活性化合物含量为9%)。10 parts (weight) of compound 1 were dissolved in the following mixture containing 90 parts (weight) of xylene, 6 parts (weight) of nonylphenol ethoxylates, 2 parts (weight) of dodecyl An emulsifiable concentrate was prepared from calcium benzenesulfonate and 2 parts by weight of the addition product of 40 moles of ethylene oxide and 1 mole of castor oil (active compound content 9%).
生测实施例Bioassay example
实例7Example 7
部分化合物进行了杀菌活性试验。方法如下:待测化合物溶于1∶1丙酮和甲醇混合物,再以2∶1∶1水、丙酮和甲醇混合物(体积比)稀释至所需浓度。此溶液喷到待测植物上晾干2小时,一天后进行病菌接种。活性相对于空白对照以百分比计,分为A、B、C、D四级,控制率90%-100%为A级,控制率75%-90%为B级,控制率50%-75%为C级,控制率0%-50%为D级Some compounds were tested for bactericidal activity. The method is as follows: the test compound is dissolved in a 1:1 mixture of acetone and methanol, and then diluted to a desired concentration with a 2:1:1 mixture of water, acetone and methanol (volume ratio). This solution was sprayed on the plants to be tested and allowed to dry for 2 hours, and inoculated with pathogens one day later. The activity is calculated as a percentage relative to the blank control, and is divided into four grades: A, B, C, and D. The control rate is 90%-100% for grade A, the control rate for 75%-90% is grade B, and the control rate is 50%-75%. It is grade C, and the control rate is 0%-50% is grade D
黄瓜白粉病Cucumber powdery mildew
施药一天后接种。接种后在温室中放置7天左右调查。测试结果见表(2)Inoculate one day after application. Place them in the greenhouse for about 7 days after inoculation for investigation. The test results are shown in Table (2)
表(2)
*对照化合物为CN1191670A中的化合物,括号内为该专利化合物表中的编号。*The reference compound is the compound in CN1191670A, and the number in the patent compound table is in the brackets.
在25mg/l浓度下,本发明的化合物控制率为A级,而对照化合物中只有化合物5的控制率为C级,其余都为D级;在6mg/l浓度下,本发明的化合物控制率为A级,所有的对照化合物基本无效。从测试结果可以看出,本发明的化合物对黄瓜白粉病具有良好的防效,明显优于对照化合物。At a concentration of 25 mg/l, the control rate of the compound of the present invention is A grade, and only the control rate of compound 5 is C grade in the reference compound, and all the others are D grade; at a concentration of 6 mg/l, the control rate of the compound of the present invention Class A, all control compounds are basically ineffective. It can be seen from the test results that the compound of the invention has good control effect on cucumber powdery mildew, which is obviously better than that of the reference compound.
水稻稻瘟病rice blast
施药一天后接种。接种后在保湿箱中保湿36小时以利于侵染,移入温室,7天左右后调查。测试结果见表(3)Inoculate one day after application. After inoculation, keep it moist in the humidity box for 36 hours to facilitate infection, move it into the greenhouse, and investigate after about 7 days. The test results are shown in Table (3)
表(3)
*对照化合物为CN1191670A中的化合物,括号内为该专利化合物表中的编号。*The reference compound is the compound in CN1191670A, and the number in the patent compound table is in the brackets.
在25mg/l浓度下,本发明的化合物控制率为A级,而所有的对照化合物控制率都在B级以下;在6mg/l浓度下,本发明的化合物控制率为A级,所有的对照化合物基本无效。从测试结果可以看出,本发明的化合物对水稻稻瘟病具有良好的防效,明显优于对照化合物。Under the 25mg/l concentration, the control rate of the compound of the present invention is A grade, and all contrast compound control rates are all below B grade; Under the 6mg/l concentration, the compound control rate of the present invention is A grade, and all contrast Compounds are largely ineffective. It can be seen from the test results that the compound of the invention has good control effect on rice blast, which is obviously better than that of the reference compound.
黄瓜灰霉病Cucumber Botrytis
施药一天后接种。接种后在恒温恒湿箱中放置5天左右后调查。测试结果见表(4)Inoculate one day after application. After inoculation, place it in a constant temperature and humidity box for about 5 days and then investigate. The test results are shown in Table (4)
表(4)
*对照化合物为CN1191670A中的化合物,括号内为该专利化合物表中的编号。*The reference compound is the compound in CN1191670A, and the number in the patent compound table is in the brackets.
在25mg/l浓度下,本发明的化合物控制率为A级,而所有的对照化合物控制率都在B级以下;在6mg/l浓度下,本发明的化合物控制率为A级,所有的对照化合物都在C级以下。从测试结果可以看出,本发明的化合物对黄瓜灰霉病的防效明显优于对照化合物。小麦叶锈病Under the 25mg/l concentration, the control rate of the compound of the present invention is A grade, and all contrast compound control rates are all below B grade; Under the 6mg/l concentration, the compound control rate of the present invention is A grade, and all contrast The compounds are all below grade C. It can be seen from the test results that the control effect of the compound of the invention on cucumber gray mold is obviously better than that of the reference compound. wheat leaf rust
施药一天后接种。接种后在温室中放置10天左右后调查。测试结果见表(5)Inoculate one day after application. After inoculation, it was placed in the greenhouse for about 10 days and then investigated. The test results are shown in Table (5)
表(5)
*对照化合物为CN1191670A中的化合物,括号内为该专利化合物表中的编号。*The reference compound is the compound in CN1191670A, and the number in the patent compound table is in the brackets.
在25mg/l浓度下,本发明的化合物控制率为A级,而所有的对照化合物控制率都在B级以下;在6mg/l浓度下,本发明的化合物控制率为A级,所有的对照化合物基本无效。从测试结果可以看出,本发明的化合物对小麦叶锈病的防效明显优于对照化合物。小麦白粉病Under the 25mg/l concentration, the control rate of the compound of the present invention is A grade, and all contrast compound control rates are all below B grade; Under the 6mg/l concentration, the compound control rate of the present invention is A grade, and all contrast Compounds are largely ineffective. It can be seen from the test results that the control effect of the compound of the invention on wheat leaf rust is obviously better than that of the reference compound. wheat powdery mildew
施药一天后接种。接种后在温室中放置7天左右后调查。测试结果见表(6)Inoculate one day after application. After inoculation, they were placed in the greenhouse for about 7 days and then investigated. The test results are shown in Table (6)
表(6)
*对照化合物为CN1191670A中的化合物,括号内为该专利化合物表中的编号。*The reference compound is the compound in CN1191670A, and the number in the patent compound table is in the brackets.
在25mg/l浓度下,本发明的化合物控制率为A级,而所有的对照化合物控制率都在B级以下;在6mg/l浓度下,本发明的化合物控制率为A级,所有的对照化合物基本无效。从测试结果可以看出,本发明的化合物对小麦白粉病具有良好的防效,明显优于对照化合物。黄瓜炭疽病Under the 25mg/l concentration, the control rate of the compound of the present invention is A grade, and all contrast compound control rates are all below B grade; Under the 6mg/l concentration, the compound control rate of the present invention is A grade, and all contrast Compounds are largely ineffective. It can be seen from the test results that the compound of the invention has good control effect on wheat powdery mildew, which is obviously better than that of the reference compound. Cucumber Anthracnose
施药一天后接种。接种后在温室中放置7天左右后调查。测试结果见表(7)Inoculate one day after application. After inoculation, they were placed in the greenhouse for about 7 days and then investigated. The test results are shown in Table (7)
表(7)
*对照化合物为CN1191670A中的化合物,括号内为该专利化合物表中的编号。*The reference compound is the compound in CN1191670A, and the number in the patent compound table is in the brackets.
在25mg/l浓度下,本发明的化合物控制率为A级,而所有的对照化合物控制率都在B级以下;在6mg/l浓度下,本发明的化合物控制率为A级,只有对照化合物14的控制率为B级,其它所有的对照化合物基本无效。从测试结果可以看出,本发明的化合物对黄瓜炭疽病具有良好的防效,明显优于对照化合物。At a concentration of 25 mg/l, the control rate of the compound of the present invention is A grade, and all control compounds are below the B grade; at a concentration of 6 mg/l, the control rate of the compound of the present invention is A grade, only the control compound The control rate of 14 was grade B, and all other control compounds were basically ineffective. It can be seen from the test results that the compound of the invention has good control effect on cucumber anthracnose, which is obviously better than that of the reference compound.
Claims (9)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 00110143 CN1114590C (en) | 2000-02-24 | 2000-02-24 | Unsaturated oximino ether bactericide |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 00110143 CN1114590C (en) | 2000-02-24 | 2000-02-24 | Unsaturated oximino ether bactericide |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1309897A true CN1309897A (en) | 2001-08-29 |
| CN1114590C CN1114590C (en) | 2003-07-16 |
Family
ID=4580157
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 00110143 Expired - Lifetime CN1114590C (en) | 2000-02-24 | 2000-02-24 | Unsaturated oximino ether bactericide |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN1114590C (en) |
Cited By (183)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100393212C (en) * | 2005-08-26 | 2008-06-11 | 沈阳化工研究院 | A kind of bactericidal, insecticidal composition |
| CN102630687A (en) * | 2012-04-01 | 2012-08-15 | 陕西上格之路生物科学有限公司 | Bactericidal composition containing fenamistrobin and metiram |
| CN102640761A (en) * | 2012-05-08 | 2012-08-22 | 陕西上格之路生物科学有限公司 | Sterilization composition containing SYP-1620 and oomycete prevention and treatment sterilization agents |
| CN102669164A (en) * | 2012-05-30 | 2012-09-19 | 陕西上格之路生物科学有限公司 | A kind of bactericidal composition containing enoxastrobin |
| CN102919241A (en) * | 2010-12-08 | 2013-02-13 | 陕西美邦农药有限公司 | Fungicide composition containing tridemorph |
| CN102972419A (en) * | 2012-12-28 | 2013-03-20 | 江苏龙灯化学有限公司 | Bactericidal composition containing thifluzamide and SYT (N-methyl 2-[2-((((4-(2, 6 dichloro-phenyl)-3-butenylidene-2-yl) amino) xy) methyl)-phenyl]-2-methoxyiminoacetamide) and application thereof |
| CN103070176A (en) * | 2013-02-25 | 2013-05-01 | 海利尔药业集团股份有限公司 | Sterilizing composition containing prothioconazole and syp-1620 |
| CN103081924A (en) * | 2013-02-27 | 2013-05-08 | 海利尔药业集团股份有限公司 | Sterilizing composition containing enestroburin and tiadinil |
| CN103125494A (en) * | 2013-03-16 | 2013-06-05 | 海利尔药业集团股份有限公司 | Bactericidal composition containing (E,E,E)-N-methyl-2-[((((1-methyl-3-(2,6-dichlorophenyl)-2-propenyl)imino)oxy)methyl)phenyl]-2-methoxyiminoacetamide and penthiopyrad |
| CN103271032A (en) * | 2012-05-31 | 2013-09-04 | 陕西上格之路生物科学有限公司 | Bactericide composition containing SYP-1620 |
| CN103271036A (en) * | 2012-05-31 | 2013-09-04 | 陕西上格之路生物科学有限公司 | Sterilization composition containing SYP-1620 |
| CN103271076A (en) * | 2012-05-31 | 2013-09-04 | 陕西上格之路生物科学有限公司 | Bactericide composition containing SYP-1620 |
| CN103704214A (en) * | 2013-12-25 | 2014-04-09 | 江苏龙灯化学有限公司 | Bactericide composition and application thereof |
| CN103951619A (en) * | 2014-05-16 | 2014-07-30 | 东南大学成贤学院 | Methoxyl imino acetamide compounds containing aryl pyrazole and use |
| CN104365633A (en) * | 2014-10-31 | 2015-02-25 | 海南江河农药化工厂有限公司 | Pesticide composition for preventing and controlling rubber trees from Corynesporacassiicola (Berk.Curt.)Wei and application thereof |
| WO2016174042A1 (en) | 2015-04-27 | 2016-11-03 | BASF Agro B.V. | Pesticidal compositions |
| WO2017024971A1 (en) * | 2015-08-12 | 2017-02-16 | 沈阳中化农药化工研发有限公司 | Unsaturated oximino ether compound and use thereof |
| WO2017076740A1 (en) | 2015-11-04 | 2017-05-11 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017076757A1 (en) | 2015-11-02 | 2017-05-11 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017081312A1 (en) | 2015-11-13 | 2017-05-18 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017081310A1 (en) | 2015-11-13 | 2017-05-18 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017085098A1 (en) | 2015-11-19 | 2017-05-26 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017085100A1 (en) | 2015-11-19 | 2017-05-26 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| CN106857551A (en) * | 2015-12-11 | 2017-06-20 | 沈阳中化农药化工研发有限公司 | One kind is for preventing and treating soil-borne disease bactericidal composition and its application |
| WO2017153200A1 (en) | 2016-03-10 | 2017-09-14 | Basf Se | Fungicidal mixtures iii comprising strobilurin-type fungicides |
| WO2017178245A1 (en) | 2016-04-11 | 2017-10-19 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018050421A1 (en) | 2016-09-13 | 2018-03-22 | Basf Se | Fungicidal mixtures i comprising quinoline fungicides |
| WO2018054711A1 (en) | 2016-09-26 | 2018-03-29 | Basf Se | Pyridine compounds for controlling phytopathogenic harmful fungi |
| WO2018054723A1 (en) | 2016-09-26 | 2018-03-29 | Basf Se | Pyridine compounds for controlling phytopathogenic harmful fungi |
| WO2018054721A1 (en) | 2016-09-26 | 2018-03-29 | Basf Se | Pyridine compounds for controlling phytopathogenic harmful fungi |
| WO2018065182A1 (en) | 2016-10-04 | 2018-04-12 | Basf Se | Reduced quinoline compounds as antifuni agents |
| WO2018073110A1 (en) | 2016-10-20 | 2018-04-26 | Basf Se | Quinoline compounds as fungicides |
| CN108191709A (en) * | 2016-12-08 | 2018-06-22 | 沈阳中化农药化工研发有限公司 | A kind of method for preparing unsaturated oximido ethers compound |
| EP3339297A1 (en) | 2016-12-20 | 2018-06-27 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| EP3338552A1 (en) | 2016-12-21 | 2018-06-27 | Basf Se | Use of a tetrazolinone fungicide on transgenic plants |
| WO2018114393A1 (en) | 2016-12-19 | 2018-06-28 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018134127A1 (en) | 2017-01-23 | 2018-07-26 | Basf Se | Fungicidal pyridine compounds |
| WO2018149754A1 (en) | 2017-02-16 | 2018-08-23 | Basf Se | Pyridine compounds |
| WO2018153730A1 (en) | 2017-02-21 | 2018-08-30 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018177970A1 (en) | 2017-03-31 | 2018-10-04 | Basf Se | Process for preparing chiral 2,3-dihydrothiazolo[3,2-a]pyrimidin-4-ium compounds |
| WO2018184970A1 (en) | 2017-04-07 | 2018-10-11 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018184882A1 (en) | 2017-04-06 | 2018-10-11 | Basf Se | Pyridine compounds |
| WO2018188962A1 (en) | 2017-04-11 | 2018-10-18 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018193385A1 (en) | 2017-04-20 | 2018-10-25 | Pi Industries Ltd. | Novel phenylamine compounds |
| WO2018202737A1 (en) | 2017-05-05 | 2018-11-08 | Basf Se | Fungicidal mixtures comprising triazole compounds |
| WO2018202491A1 (en) | 2017-05-04 | 2018-11-08 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2018202428A1 (en) | 2017-05-02 | 2018-11-08 | Basf Se | Fungicidal mixture comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| WO2018202487A1 (en) | 2017-05-04 | 2018-11-08 | Basf Se | Substituted 5-(haloalkyl)-5-hydroxy-isoxazoles for combating phytopathogenic fungi |
| WO2018210661A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2018210658A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2018210660A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2018210659A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2018219797A1 (en) | 2017-06-02 | 2018-12-06 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018219725A1 (en) | 2017-05-30 | 2018-12-06 | Basf Se | Pyridine and pyrazine compounds |
| WO2018234139A1 (en) | 2017-06-19 | 2018-12-27 | Basf Se | 2 - [[5- (TRIFLUOROMETHYL) -1,2,4-OXADIAZOL-3-YL] ARYLOXY] (THIO) ACETAMIDES FOR THE CONTROL OF PHYTOPATHOGENIC FUNGI |
| WO2019002158A1 (en) | 2017-06-30 | 2019-01-03 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019025250A1 (en) | 2017-08-04 | 2019-02-07 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019038042A1 (en) | 2017-08-21 | 2019-02-28 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019042800A1 (en) | 2017-08-29 | 2019-03-07 | Basf Se | Pesticidal mixtures |
| WO2019043183A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| EP3453706A1 (en) | 2017-09-08 | 2019-03-13 | Basf Se | Pesticidal imidazole compounds |
| WO2019052932A1 (en) | 2017-09-18 | 2019-03-21 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019057660A1 (en) | 2017-09-25 | 2019-03-28 | Basf Se | Indole and azaindole compounds with substituted 6-membered aryl and heteroaryl rings as agrochemical fungicides |
| WO2019072906A1 (en) | 2017-10-13 | 2019-04-18 | Basf Se | Imidazolidine pyrimidinium compounds for combating animal pests |
| WO2019101511A1 (en) | 2017-11-23 | 2019-05-31 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019115343A1 (en) | 2017-12-15 | 2019-06-20 | Basf Se | Fungicidal mixture comprising substituted pyridines |
| WO2019115511A1 (en) | 2017-12-14 | 2019-06-20 | Basf Se | Fungicidal mixture comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| WO2019123196A1 (en) | 2017-12-20 | 2019-06-27 | Pi Industries Ltd. | Fluoralkenyl compounds, process for preparation and use thereof |
| WO2019121143A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Substituted cyclopropyl derivatives |
| WO2019137995A1 (en) | 2018-01-11 | 2019-07-18 | Basf Se | Novel pyridazine compounds for controlling invertebrate pests |
| WO2019145140A1 (en) | 2018-01-09 | 2019-08-01 | Basf Se | Silylethynyl hetaryl compounds as nitrification inhibitors |
| WO2019150311A1 (en) | 2018-02-02 | 2019-08-08 | Pi Industries Ltd. | 1-3 dithiol compounds and their use for the protection of crops from phytopathogenic microorganisms |
| WO2019154663A1 (en) | 2018-02-07 | 2019-08-15 | Basf Se | New pyridine carboxamides |
| WO2019154665A1 (en) | 2018-02-07 | 2019-08-15 | Basf Se | New pyridine carboxamides |
| EP3530116A1 (en) | 2018-02-27 | 2019-08-28 | Basf Se | Fungicidal mixtures comprising xemium |
| EP3530118A1 (en) | 2018-02-26 | 2019-08-28 | Basf Se | Fungicidal mixtures |
| EP3533333A1 (en) | 2018-03-02 | 2019-09-04 | Basf Se | Fungicidal mixtures comprising pydiflumetofen |
| EP3533331A1 (en) | 2018-03-02 | 2019-09-04 | Basf Se | Fungicidal mixtures comprising pydiflumetofen |
| WO2019166560A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of n-functionalized alkoxy pyrazole compounds as nitrification inhibitors |
| WO2019166561A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of alkoxypyrazoles as nitrification inhibitors |
| WO2019166252A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Fungicidal mixtures comprising fenpropidin |
| WO2019166257A1 (en) | 2018-03-01 | 2019-09-06 | BASF Agro B.V. | Fungicidal compositions of mefentrifluconazole |
| WO2019166558A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of pyrazole propargyl ethers as nitrification inhibitors |
| EP3536150A1 (en) | 2018-03-06 | 2019-09-11 | Basf Se | Fungicidal mixtures comprising fluxapyroxad |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| CN110407681A (en) * | 2019-08-12 | 2019-11-05 | 海南大学 | A kind of dehydrogingerone derivative, its preparation method and application |
| WO2019219464A1 (en) | 2018-05-15 | 2019-11-21 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019219529A1 (en) | 2018-05-15 | 2019-11-21 | Basf Se | Mixtures comprising benzpyrimoxan and oxazosulfyl and uses and methods of applying them |
| WO2019224092A1 (en) | 2018-05-22 | 2019-11-28 | Basf Se | Pesticidally active c15-derivatives of ginkgolides |
| WO2020002472A1 (en) | 2018-06-28 | 2020-01-02 | Basf Se | Use of alkynylthiophenes as nitrification inhibitors |
| WO2020020777A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of substituted 2-thiazolines as nitrification inhibitors |
| WO2020020765A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of a substituted thiazolidine compound as nitrification inhibitor |
| WO2020035826A1 (en) | 2018-08-17 | 2020-02-20 | Pi Industries Ltd. | 1,2-dithiolone compounds and use thereof |
| EP3613736A1 (en) | 2018-08-22 | 2020-02-26 | Basf Se | Substituted glutarimide derivatives |
| EP3628158A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| EP3628157A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| EP3628156A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method for controlling pests of sugarcane, citrus, rapeseed, and potato plants |
| WO2020064492A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling pests by seed treatment application of a mesoionic compound or mixture thereof |
| EP3643705A1 (en) | 2018-10-24 | 2020-04-29 | Basf Se | Pesticidal compounds |
| WO2020095161A1 (en) | 2018-11-05 | 2020-05-14 | Pi Industries Ltd. | Nitrone compounds and use thereof |
| EP3670501A1 (en) | 2018-12-17 | 2020-06-24 | Basf Se | Substituted [1,2,4]triazole compounds as fungicides |
| EP3696177A1 (en) | 2019-02-12 | 2020-08-19 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP3730489A1 (en) | 2019-04-25 | 2020-10-28 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2020239517A1 (en) | 2019-05-29 | 2020-12-03 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2020244969A1 (en) | 2019-06-06 | 2020-12-10 | Basf Se | Pyridine derivatives and their use as fungicides |
| WO2020244968A1 (en) | 2019-06-06 | 2020-12-10 | Basf Se | Fungicidal n-(pyrid-3-yl)carboxamides |
| WO2020244970A1 (en) | 2019-06-06 | 2020-12-10 | Basf Se | New carbocyclic pyridine carboxamides |
| EP3766879A1 (en) | 2019-07-19 | 2021-01-20 | Basf Se | Pesticidal pyrazole derivatives |
| EP3769623A1 (en) | 2019-07-22 | 2021-01-27 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2021063736A1 (en) | 2019-10-02 | 2021-04-08 | Basf Se | Bicyclic pyridine derivatives |
| WO2021063735A1 (en) | 2019-10-02 | 2021-04-08 | Basf Se | New bicyclic pyridine derivatives |
| WO2021130143A1 (en) | 2019-12-23 | 2021-07-01 | Basf Se | Enzyme enhanced root uptake of agrochemical active compound |
| WO2021170463A1 (en) | 2020-02-28 | 2021-09-02 | BASF Agro B.V. | Methods and uses of a mixture comprising alpha-cypermethrin and dinotefuran for controlling invertebrate pests in turf |
| WO2021175669A1 (en) | 2020-03-04 | 2021-09-10 | Basf Se | Use of substituted 1,2,4-oxadiazoles for combating phytopathogenic fungi |
| WO2021209360A1 (en) | 2020-04-14 | 2021-10-21 | Basf Se | Fungicidal mixtures comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| EP3903581A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors i |
| EP3903584A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors iv |
| EP3903582A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors ii |
| EP3903583A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors iii |
| WO2021219513A1 (en) | 2020-04-28 | 2021-11-04 | Basf Se | Pesticidal compounds |
| EP3909950A1 (en) | 2020-05-13 | 2021-11-17 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2021249800A1 (en) | 2020-06-10 | 2021-12-16 | Basf Se | Substituted [1,2,4]triazole compounds as fungicides |
| EP3939961A1 (en) | 2020-07-16 | 2022-01-19 | Basf Se | Strobilurin type compounds and their use for combating phytopathogenic fungi |
| WO2022017836A1 (en) | 2020-07-20 | 2022-01-27 | BASF Agro B.V. | Fungicidal compositions comprising (r)-2-[4-(4-chlorophenoxy)-2-(trifluoromethyl)phenyl]-1- (1,2,4-triazol-1-yl)propan-2-ol |
| EP3945089A1 (en) | 2020-07-31 | 2022-02-02 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors v |
| EP3960727A1 (en) | 2020-08-28 | 2022-03-02 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors vi |
| EP3970494A1 (en) | 2020-09-21 | 2022-03-23 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors viii |
| WO2022064453A1 (en) | 2020-09-26 | 2022-03-31 | Pi Industries Ltd. | Nematocidal compounds and use thereof |
| WO2022090071A1 (en) | 2020-11-02 | 2022-05-05 | Basf Se | Use of mefenpyr-diethyl for controlling phytopathogenic fungi |
| WO2022089969A1 (en) | 2020-10-27 | 2022-05-05 | BASF Agro B.V. | Compositions comprising mefentrifluconazole |
| WO2022090069A1 (en) | 2020-11-02 | 2022-05-05 | Basf Se | Compositions comprising mefenpyr-diethyl |
| WO2022106304A1 (en) | 2020-11-23 | 2022-05-27 | BASF Agro B.V. | Compositions comprising mefentrifluconazole |
| EP4018830A1 (en) | 2020-12-23 | 2022-06-29 | Basf Se | Pesticidal mixtures |
| WO2022167488A1 (en) | 2021-02-02 | 2022-08-11 | Basf Se | Synergistic action of dcd and alkoxypyrazoles as nitrification inhibitors |
| EP4043444A1 (en) | 2021-02-11 | 2022-08-17 | Basf Se | Substituted isoxazoline derivatives |
| WO2022238157A1 (en) | 2021-05-11 | 2022-11-17 | Basf Se | Fungicidal mixtures comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| WO2022243109A1 (en) | 2021-05-18 | 2022-11-24 | Basf Se | New substituted quinolines as fungicides |
| WO2022243111A1 (en) | 2021-05-18 | 2022-11-24 | Basf Se | New substituted pyridines as fungicides |
| WO2022243521A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of ethynylpyridine compounds as nitrification inhibitors |
| WO2022243107A1 (en) | 2021-05-18 | 2022-11-24 | Basf Se | New substituted pyridines as fungicides |
| WO2022243523A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of an n-functionalized alkoxy pyrazole compound as nitrification inhibitor |
| EP4094579A1 (en) | 2021-05-28 | 2022-11-30 | Basf Se | Pesticidal mixtures comprising metyltetraprole |
| WO2022268810A1 (en) | 2021-06-21 | 2022-12-29 | Basf Se | Metal-organic frameworks with pyrazole-based building blocks |
| EP4119547A1 (en) | 2021-07-12 | 2023-01-18 | Basf Se | Triazole compounds for the control of invertebrate pests |
| WO2023011958A1 (en) | 2021-08-02 | 2023-02-09 | Basf Se | (3-pirydyl)-quinazoline |
| WO2023011957A1 (en) | 2021-08-02 | 2023-02-09 | Basf Se | (3-quinolyl)-quinazoline |
| EP4140986A1 (en) | 2021-08-23 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4140995A1 (en) | 2021-08-27 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4151631A1 (en) | 2021-09-20 | 2023-03-22 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| CN115956568A (en) * | 2023-01-05 | 2023-04-14 | 河南科技学院 | Flutriafol-containing bactericidal composition |
| WO2023072671A1 (en) | 2021-10-28 | 2023-05-04 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors ix |
| WO2023072670A1 (en) | 2021-10-28 | 2023-05-04 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors x |
| EP4194453A1 (en) | 2021-12-08 | 2023-06-14 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4198033A1 (en) | 2021-12-14 | 2023-06-21 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP4198023A1 (en) | 2021-12-16 | 2023-06-21 | Basf Se | Pesticidally active thiosemicarbazone compounds |
| EP4238971A1 (en) | 2022-03-02 | 2023-09-06 | Basf Se | Substituted isoxazoline derivatives |
| WO2023203066A1 (en) | 2022-04-21 | 2023-10-26 | Basf Se | Synergistic action as nitrification inhibitors of dcd oligomers with alkoxypyrazole and its oligomers |
| WO2024028243A1 (en) | 2022-08-02 | 2024-02-08 | Basf Se | Pyrazolo pesticidal compounds |
| EP4342885A1 (en) | 2022-09-20 | 2024-03-27 | Basf Se | N-(3-(aminomethyl)-phenyl)-5-(4-phenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-amine derivatives and similar compounds as pesticides |
| EP4361126A1 (en) | 2022-10-24 | 2024-05-01 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors xv |
| WO2024104818A1 (en) | 2022-11-16 | 2024-05-23 | Basf Se | Substituted benzodiazepines as fungicides |
| WO2024104823A1 (en) | 2022-11-16 | 2024-05-23 | Basf Se | New substituted tetrahydrobenzoxazepine |
| WO2024104814A1 (en) | 2022-11-16 | 2024-05-23 | Basf Se | Fungicidal mixture comprising substituted pyridines |
| WO2024104813A1 (en) | 2022-11-14 | 2024-05-23 | Basf Se | Fungicidal mixture comprising substituted pyridines |
| WO2024104815A1 (en) | 2022-11-16 | 2024-05-23 | Basf Se | Substituted benzodiazepines as fungicides |
| WO2024104822A1 (en) | 2022-11-16 | 2024-05-23 | Basf Se | Substituted tetrahydrobenzodiazepine as fungicides |
| EP4389210A1 (en) | 2022-12-21 | 2024-06-26 | Basf Se | Heteroaryl compounds for the control of invertebrate pests |
| WO2024165343A1 (en) | 2023-02-08 | 2024-08-15 | Basf Se | New substituted quinoline compounds for combatitng phytopathogenic fungi |
| WO2024194038A1 (en) | 2023-03-17 | 2024-09-26 | Basf Se | Substituted pyridyl/pyrazidyl dihydrobenzothiazepine compounds for combatting phytopathogenic fungi |
| EP4455137A1 (en) | 2023-04-24 | 2024-10-30 | Basf Se | Pyrimidine compounds for the control of invertebrate pests |
| WO2024223034A1 (en) | 2023-04-26 | 2024-10-31 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors xvi |
| EP4467535A1 (en) | 2023-05-25 | 2024-11-27 | Basf Se | Lactam pesticidal compounds |
| EP4488269A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488273A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488270A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4574819A1 (en) | 2023-12-22 | 2025-06-25 | Basf Se | Diazinone compounds for the control of invertebrate pests |
| EP4640052A1 (en) | 2024-04-24 | 2025-10-29 | Basf Se | Mixtures of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors with at least one further pesticide i |
| WO2025223904A1 (en) | 2024-04-24 | 2025-10-30 | Basf Se | Mixtures of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors with at least one further pesticide i |
| WO2026021910A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
| WO2026021912A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
| WO2026021911A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
| WO2026021909A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
-
2000
- 2000-02-24 CN CN 00110143 patent/CN1114590C/en not_active Expired - Lifetime
Cited By (200)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100393212C (en) * | 2005-08-26 | 2008-06-11 | 沈阳化工研究院 | A kind of bactericidal, insecticidal composition |
| CN102919241A (en) * | 2010-12-08 | 2013-02-13 | 陕西美邦农药有限公司 | Fungicide composition containing tridemorph |
| CN102919241B (en) * | 2010-12-08 | 2014-08-13 | 陕西美邦农药有限公司 | Fungicide composition containing tridemorph |
| CN102630687A (en) * | 2012-04-01 | 2012-08-15 | 陕西上格之路生物科学有限公司 | Bactericidal composition containing fenamistrobin and metiram |
| CN102640761A (en) * | 2012-05-08 | 2012-08-22 | 陕西上格之路生物科学有限公司 | Sterilization composition containing SYP-1620 and oomycete prevention and treatment sterilization agents |
| CN102669164A (en) * | 2012-05-30 | 2012-09-19 | 陕西上格之路生物科学有限公司 | A kind of bactericidal composition containing enoxastrobin |
| CN103271032B (en) * | 2012-05-31 | 2014-04-09 | 陕西上格之路生物科学有限公司 | Bactericide composition containing SYP-1620 |
| CN103271032A (en) * | 2012-05-31 | 2013-09-04 | 陕西上格之路生物科学有限公司 | Bactericide composition containing SYP-1620 |
| CN103271036A (en) * | 2012-05-31 | 2013-09-04 | 陕西上格之路生物科学有限公司 | Sterilization composition containing SYP-1620 |
| CN103271076A (en) * | 2012-05-31 | 2013-09-04 | 陕西上格之路生物科学有限公司 | Bactericide composition containing SYP-1620 |
| CN103271076B (en) * | 2012-05-31 | 2014-04-09 | 陕西上格之路生物科学有限公司 | Bactericide composition containing SYP-1620 |
| CN103271036B (en) * | 2012-05-31 | 2014-04-09 | 陕西上格之路生物科学有限公司 | Sterilization composition containing SYP-1620 |
| CN102972419B (en) * | 2012-12-28 | 2014-08-27 | 江苏龙灯化学有限公司 | Bactericidal composition containing thifluzamide and SYT (N-methyl 2-[2-((((4-(2, 6 dichloro-phenyl)-3-butenylidene-2-yl) amino) xy) methyl)-phenyl]-2-methoxyiminoacetamide) and application thereof |
| CN102972419A (en) * | 2012-12-28 | 2013-03-20 | 江苏龙灯化学有限公司 | Bactericidal composition containing thifluzamide and SYT (N-methyl 2-[2-((((4-(2, 6 dichloro-phenyl)-3-butenylidene-2-yl) amino) xy) methyl)-phenyl]-2-methoxyiminoacetamide) and application thereof |
| CN103070176A (en) * | 2013-02-25 | 2013-05-01 | 海利尔药业集团股份有限公司 | Sterilizing composition containing prothioconazole and syp-1620 |
| CN103081924A (en) * | 2013-02-27 | 2013-05-08 | 海利尔药业集团股份有限公司 | Sterilizing composition containing enestroburin and tiadinil |
| CN103125494A (en) * | 2013-03-16 | 2013-06-05 | 海利尔药业集团股份有限公司 | Bactericidal composition containing (E,E,E)-N-methyl-2-[((((1-methyl-3-(2,6-dichlorophenyl)-2-propenyl)imino)oxy)methyl)phenyl]-2-methoxyiminoacetamide and penthiopyrad |
| CN103704214B (en) * | 2013-12-25 | 2015-09-09 | 江苏龙灯化学有限公司 | A kind of fungicide composition and its application |
| CN103704214A (en) * | 2013-12-25 | 2014-04-09 | 江苏龙灯化学有限公司 | Bactericide composition and application thereof |
| CN103951619A (en) * | 2014-05-16 | 2014-07-30 | 东南大学成贤学院 | Methoxyl imino acetamide compounds containing aryl pyrazole and use |
| CN104365633A (en) * | 2014-10-31 | 2015-02-25 | 海南江河农药化工厂有限公司 | Pesticide composition for preventing and controlling rubber trees from Corynesporacassiicola (Berk.Curt.)Wei and application thereof |
| CN104365633B (en) * | 2014-10-31 | 2016-03-02 | 海南江河农药化工厂有限公司 | A kind of composition pesticide and application thereof preventing and treating the mould defoliation of bamboo grows rod spore |
| WO2016174042A1 (en) | 2015-04-27 | 2016-11-03 | BASF Agro B.V. | Pesticidal compositions |
| WO2017024971A1 (en) * | 2015-08-12 | 2017-02-16 | 沈阳中化农药化工研发有限公司 | Unsaturated oximino ether compound and use thereof |
| WO2017076757A1 (en) | 2015-11-02 | 2017-05-11 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017076740A1 (en) | 2015-11-04 | 2017-05-11 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017081312A1 (en) | 2015-11-13 | 2017-05-18 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017081310A1 (en) | 2015-11-13 | 2017-05-18 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017085098A1 (en) | 2015-11-19 | 2017-05-26 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017085100A1 (en) | 2015-11-19 | 2017-05-26 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| CN106857551A (en) * | 2015-12-11 | 2017-06-20 | 沈阳中化农药化工研发有限公司 | One kind is for preventing and treating soil-borne disease bactericidal composition and its application |
| WO2017153200A1 (en) | 2016-03-10 | 2017-09-14 | Basf Se | Fungicidal mixtures iii comprising strobilurin-type fungicides |
| WO2017178245A1 (en) | 2016-04-11 | 2017-10-19 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018050421A1 (en) | 2016-09-13 | 2018-03-22 | Basf Se | Fungicidal mixtures i comprising quinoline fungicides |
| WO2018054711A1 (en) | 2016-09-26 | 2018-03-29 | Basf Se | Pyridine compounds for controlling phytopathogenic harmful fungi |
| WO2018054723A1 (en) | 2016-09-26 | 2018-03-29 | Basf Se | Pyridine compounds for controlling phytopathogenic harmful fungi |
| WO2018054721A1 (en) | 2016-09-26 | 2018-03-29 | Basf Se | Pyridine compounds for controlling phytopathogenic harmful fungi |
| WO2018065182A1 (en) | 2016-10-04 | 2018-04-12 | Basf Se | Reduced quinoline compounds as antifuni agents |
| WO2018073110A1 (en) | 2016-10-20 | 2018-04-26 | Basf Se | Quinoline compounds as fungicides |
| CN108191709A (en) * | 2016-12-08 | 2018-06-22 | 沈阳中化农药化工研发有限公司 | A kind of method for preparing unsaturated oximido ethers compound |
| CN108191709B (en) * | 2016-12-08 | 2020-03-24 | 沈阳中化农药化工研发有限公司 | A kind of method for preparing unsaturated oxime ether compounds |
| WO2018114393A1 (en) | 2016-12-19 | 2018-06-28 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| EP3339297A1 (en) | 2016-12-20 | 2018-06-27 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| EP3338552A1 (en) | 2016-12-21 | 2018-06-27 | Basf Se | Use of a tetrazolinone fungicide on transgenic plants |
| WO2018134127A1 (en) | 2017-01-23 | 2018-07-26 | Basf Se | Fungicidal pyridine compounds |
| WO2018149754A1 (en) | 2017-02-16 | 2018-08-23 | Basf Se | Pyridine compounds |
| WO2018153730A1 (en) | 2017-02-21 | 2018-08-30 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018177970A1 (en) | 2017-03-31 | 2018-10-04 | Basf Se | Process for preparing chiral 2,3-dihydrothiazolo[3,2-a]pyrimidin-4-ium compounds |
| EP3978504A1 (en) | 2017-03-31 | 2022-04-06 | Basf Se | Chiral 2,3-dihydrothiazolo[3,2-a]pyrimidine derivatives for combating animal pests |
| WO2018184882A1 (en) | 2017-04-06 | 2018-10-11 | Basf Se | Pyridine compounds |
| WO2018184970A1 (en) | 2017-04-07 | 2018-10-11 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018188962A1 (en) | 2017-04-11 | 2018-10-18 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018193385A1 (en) | 2017-04-20 | 2018-10-25 | Pi Industries Ltd. | Novel phenylamine compounds |
| US11524934B2 (en) | 2017-04-20 | 2022-12-13 | Pi Industries Ltd | Phenylamine compounds |
| WO2018202428A1 (en) | 2017-05-02 | 2018-11-08 | Basf Se | Fungicidal mixture comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| WO2018202491A1 (en) | 2017-05-04 | 2018-11-08 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2018202487A1 (en) | 2017-05-04 | 2018-11-08 | Basf Se | Substituted 5-(haloalkyl)-5-hydroxy-isoxazoles for combating phytopathogenic fungi |
| WO2018202737A1 (en) | 2017-05-05 | 2018-11-08 | Basf Se | Fungicidal mixtures comprising triazole compounds |
| WO2018210661A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2018210658A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2018210660A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2018210659A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2018219725A1 (en) | 2017-05-30 | 2018-12-06 | Basf Se | Pyridine and pyrazine compounds |
| WO2018219797A1 (en) | 2017-06-02 | 2018-12-06 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018234139A1 (en) | 2017-06-19 | 2018-12-27 | Basf Se | 2 - [[5- (TRIFLUOROMETHYL) -1,2,4-OXADIAZOL-3-YL] ARYLOXY] (THIO) ACETAMIDES FOR THE CONTROL OF PHYTOPATHOGENIC FUNGI |
| WO2019002158A1 (en) | 2017-06-30 | 2019-01-03 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019025250A1 (en) | 2017-08-04 | 2019-02-07 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019038042A1 (en) | 2017-08-21 | 2019-02-28 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019042800A1 (en) | 2017-08-29 | 2019-03-07 | Basf Se | Pesticidal mixtures |
| EP3915379A1 (en) | 2017-08-29 | 2021-12-01 | Basf Se | Pesticidal mixtures |
| WO2019042932A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| WO2019043183A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| EP3453706A1 (en) | 2017-09-08 | 2019-03-13 | Basf Se | Pesticidal imidazole compounds |
| WO2019052932A1 (en) | 2017-09-18 | 2019-03-21 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019057660A1 (en) | 2017-09-25 | 2019-03-28 | Basf Se | Indole and azaindole compounds with substituted 6-membered aryl and heteroaryl rings as agrochemical fungicides |
| WO2019072906A1 (en) | 2017-10-13 | 2019-04-18 | Basf Se | Imidazolidine pyrimidinium compounds for combating animal pests |
| WO2019101511A1 (en) | 2017-11-23 | 2019-05-31 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019115511A1 (en) | 2017-12-14 | 2019-06-20 | Basf Se | Fungicidal mixture comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| WO2019115343A1 (en) | 2017-12-15 | 2019-06-20 | Basf Se | Fungicidal mixture comprising substituted pyridines |
| WO2019123196A1 (en) | 2017-12-20 | 2019-06-27 | Pi Industries Ltd. | Fluoralkenyl compounds, process for preparation and use thereof |
| WO2019121143A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Substituted cyclopropyl derivatives |
| WO2019145140A1 (en) | 2018-01-09 | 2019-08-01 | Basf Se | Silylethynyl hetaryl compounds as nitrification inhibitors |
| WO2019137995A1 (en) | 2018-01-11 | 2019-07-18 | Basf Se | Novel pyridazine compounds for controlling invertebrate pests |
| WO2019150311A1 (en) | 2018-02-02 | 2019-08-08 | Pi Industries Ltd. | 1-3 dithiol compounds and their use for the protection of crops from phytopathogenic microorganisms |
| WO2019154665A1 (en) | 2018-02-07 | 2019-08-15 | Basf Se | New pyridine carboxamides |
| WO2019154663A1 (en) | 2018-02-07 | 2019-08-15 | Basf Se | New pyridine carboxamides |
| EP3530118A1 (en) | 2018-02-26 | 2019-08-28 | Basf Se | Fungicidal mixtures |
| EP3530116A1 (en) | 2018-02-27 | 2019-08-28 | Basf Se | Fungicidal mixtures comprising xemium |
| WO2019166560A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of n-functionalized alkoxy pyrazole compounds as nitrification inhibitors |
| WO2019166561A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of alkoxypyrazoles as nitrification inhibitors |
| WO2019166252A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Fungicidal mixtures comprising fenpropidin |
| WO2019166558A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of pyrazole propargyl ethers as nitrification inhibitors |
| WO2019166257A1 (en) | 2018-03-01 | 2019-09-06 | BASF Agro B.V. | Fungicidal compositions of mefentrifluconazole |
| EP3533333A1 (en) | 2018-03-02 | 2019-09-04 | Basf Se | Fungicidal mixtures comprising pydiflumetofen |
| EP3533331A1 (en) | 2018-03-02 | 2019-09-04 | Basf Se | Fungicidal mixtures comprising pydiflumetofen |
| EP3536150A1 (en) | 2018-03-06 | 2019-09-11 | Basf Se | Fungicidal mixtures comprising fluxapyroxad |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019219529A1 (en) | 2018-05-15 | 2019-11-21 | Basf Se | Mixtures comprising benzpyrimoxan and oxazosulfyl and uses and methods of applying them |
| WO2019219464A1 (en) | 2018-05-15 | 2019-11-21 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019224092A1 (en) | 2018-05-22 | 2019-11-28 | Basf Se | Pesticidally active c15-derivatives of ginkgolides |
| WO2020002472A1 (en) | 2018-06-28 | 2020-01-02 | Basf Se | Use of alkynylthiophenes as nitrification inhibitors |
| WO2020020765A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of a substituted thiazolidine compound as nitrification inhibitor |
| WO2020020777A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of substituted 2-thiazolines as nitrification inhibitors |
| WO2020035826A1 (en) | 2018-08-17 | 2020-02-20 | Pi Industries Ltd. | 1,2-dithiolone compounds and use thereof |
| EP3613736A1 (en) | 2018-08-22 | 2020-02-26 | Basf Se | Substituted glutarimide derivatives |
| EP3628158A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| EP3628157A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| EP3628156A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method for controlling pests of sugarcane, citrus, rapeseed, and potato plants |
| WO2020064408A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| WO2020064492A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling pests by seed treatment application of a mesoionic compound or mixture thereof |
| WO2020064480A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| EP3643705A1 (en) | 2018-10-24 | 2020-04-29 | Basf Se | Pesticidal compounds |
| WO2020083733A1 (en) | 2018-10-24 | 2020-04-30 | Basf Se | Pesticidal compounds |
| WO2020095161A1 (en) | 2018-11-05 | 2020-05-14 | Pi Industries Ltd. | Nitrone compounds and use thereof |
| EP3670501A1 (en) | 2018-12-17 | 2020-06-24 | Basf Se | Substituted [1,2,4]triazole compounds as fungicides |
| EP3696177A1 (en) | 2019-02-12 | 2020-08-19 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP3730489A1 (en) | 2019-04-25 | 2020-10-28 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2020239517A1 (en) | 2019-05-29 | 2020-12-03 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2020244969A1 (en) | 2019-06-06 | 2020-12-10 | Basf Se | Pyridine derivatives and their use as fungicides |
| WO2020244968A1 (en) | 2019-06-06 | 2020-12-10 | Basf Se | Fungicidal n-(pyrid-3-yl)carboxamides |
| WO2020244970A1 (en) | 2019-06-06 | 2020-12-10 | Basf Se | New carbocyclic pyridine carboxamides |
| EP3766879A1 (en) | 2019-07-19 | 2021-01-20 | Basf Se | Pesticidal pyrazole derivatives |
| WO2021013561A1 (en) | 2019-07-19 | 2021-01-28 | Basf Se | Pesticidal pyrazole and triazole derivatives |
| EP3769623A1 (en) | 2019-07-22 | 2021-01-27 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| CN110407681A (en) * | 2019-08-12 | 2019-11-05 | 海南大学 | A kind of dehydrogingerone derivative, its preparation method and application |
| WO2021063736A1 (en) | 2019-10-02 | 2021-04-08 | Basf Se | Bicyclic pyridine derivatives |
| WO2021063735A1 (en) | 2019-10-02 | 2021-04-08 | Basf Se | New bicyclic pyridine derivatives |
| WO2021130143A1 (en) | 2019-12-23 | 2021-07-01 | Basf Se | Enzyme enhanced root uptake of agrochemical active compound |
| WO2021170463A1 (en) | 2020-02-28 | 2021-09-02 | BASF Agro B.V. | Methods and uses of a mixture comprising alpha-cypermethrin and dinotefuran for controlling invertebrate pests in turf |
| WO2021175669A1 (en) | 2020-03-04 | 2021-09-10 | Basf Se | Use of substituted 1,2,4-oxadiazoles for combating phytopathogenic fungi |
| WO2021209360A1 (en) | 2020-04-14 | 2021-10-21 | Basf Se | Fungicidal mixtures comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| EP3903581A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors i |
| EP3903584A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors iv |
| EP3903582A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors ii |
| EP3903583A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors iii |
| WO2021219513A1 (en) | 2020-04-28 | 2021-11-04 | Basf Se | Pesticidal compounds |
| EP3909950A1 (en) | 2020-05-13 | 2021-11-17 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2021249800A1 (en) | 2020-06-10 | 2021-12-16 | Basf Se | Substituted [1,2,4]triazole compounds as fungicides |
| EP3939961A1 (en) | 2020-07-16 | 2022-01-19 | Basf Se | Strobilurin type compounds and their use for combating phytopathogenic fungi |
| WO2022017836A1 (en) | 2020-07-20 | 2022-01-27 | BASF Agro B.V. | Fungicidal compositions comprising (r)-2-[4-(4-chlorophenoxy)-2-(trifluoromethyl)phenyl]-1- (1,2,4-triazol-1-yl)propan-2-ol |
| EP3945089A1 (en) | 2020-07-31 | 2022-02-02 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors v |
| EP3960727A1 (en) | 2020-08-28 | 2022-03-02 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors vi |
| EP3970494A1 (en) | 2020-09-21 | 2022-03-23 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors viii |
| WO2022064453A1 (en) | 2020-09-26 | 2022-03-31 | Pi Industries Ltd. | Nematocidal compounds and use thereof |
| WO2022089969A1 (en) | 2020-10-27 | 2022-05-05 | BASF Agro B.V. | Compositions comprising mefentrifluconazole |
| WO2022090071A1 (en) | 2020-11-02 | 2022-05-05 | Basf Se | Use of mefenpyr-diethyl for controlling phytopathogenic fungi |
| WO2022090069A1 (en) | 2020-11-02 | 2022-05-05 | Basf Se | Compositions comprising mefenpyr-diethyl |
| WO2022106304A1 (en) | 2020-11-23 | 2022-05-27 | BASF Agro B.V. | Compositions comprising mefentrifluconazole |
| EP4018830A1 (en) | 2020-12-23 | 2022-06-29 | Basf Se | Pesticidal mixtures |
| WO2022167488A1 (en) | 2021-02-02 | 2022-08-11 | Basf Se | Synergistic action of dcd and alkoxypyrazoles as nitrification inhibitors |
| EP4043444A1 (en) | 2021-02-11 | 2022-08-17 | Basf Se | Substituted isoxazoline derivatives |
| WO2022238157A1 (en) | 2021-05-11 | 2022-11-17 | Basf Se | Fungicidal mixtures comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| WO2022243111A1 (en) | 2021-05-18 | 2022-11-24 | Basf Se | New substituted pyridines as fungicides |
| WO2022243107A1 (en) | 2021-05-18 | 2022-11-24 | Basf Se | New substituted pyridines as fungicides |
| WO2022243109A1 (en) | 2021-05-18 | 2022-11-24 | Basf Se | New substituted quinolines as fungicides |
| WO2022243521A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of ethynylpyridine compounds as nitrification inhibitors |
| WO2022243523A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of an n-functionalized alkoxy pyrazole compound as nitrification inhibitor |
| EP4094579A1 (en) | 2021-05-28 | 2022-11-30 | Basf Se | Pesticidal mixtures comprising metyltetraprole |
| WO2022268810A1 (en) | 2021-06-21 | 2022-12-29 | Basf Se | Metal-organic frameworks with pyrazole-based building blocks |
| EP4119547A1 (en) | 2021-07-12 | 2023-01-18 | Basf Se | Triazole compounds for the control of invertebrate pests |
| WO2023011958A1 (en) | 2021-08-02 | 2023-02-09 | Basf Se | (3-pirydyl)-quinazoline |
| WO2023011957A1 (en) | 2021-08-02 | 2023-02-09 | Basf Se | (3-quinolyl)-quinazoline |
| EP4140986A1 (en) | 2021-08-23 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4140995A1 (en) | 2021-08-27 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4151631A1 (en) | 2021-09-20 | 2023-03-22 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2023072671A1 (en) | 2021-10-28 | 2023-05-04 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors ix |
| WO2023072670A1 (en) | 2021-10-28 | 2023-05-04 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors x |
| EP4194453A1 (en) | 2021-12-08 | 2023-06-14 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4198033A1 (en) | 2021-12-14 | 2023-06-21 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP4198023A1 (en) | 2021-12-16 | 2023-06-21 | Basf Se | Pesticidally active thiosemicarbazone compounds |
| EP4238971A1 (en) | 2022-03-02 | 2023-09-06 | Basf Se | Substituted isoxazoline derivatives |
| WO2023203066A1 (en) | 2022-04-21 | 2023-10-26 | Basf Se | Synergistic action as nitrification inhibitors of dcd oligomers with alkoxypyrazole and its oligomers |
| WO2024028243A1 (en) | 2022-08-02 | 2024-02-08 | Basf Se | Pyrazolo pesticidal compounds |
| EP4342885A1 (en) | 2022-09-20 | 2024-03-27 | Basf Se | N-(3-(aminomethyl)-phenyl)-5-(4-phenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-amine derivatives and similar compounds as pesticides |
| EP4361126A1 (en) | 2022-10-24 | 2024-05-01 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors xv |
| WO2024088792A1 (en) | 2022-10-24 | 2024-05-02 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors xv |
| WO2024104813A1 (en) | 2022-11-14 | 2024-05-23 | Basf Se | Fungicidal mixture comprising substituted pyridines |
| WO2024104818A1 (en) | 2022-11-16 | 2024-05-23 | Basf Se | Substituted benzodiazepines as fungicides |
| WO2024104823A1 (en) | 2022-11-16 | 2024-05-23 | Basf Se | New substituted tetrahydrobenzoxazepine |
| WO2024104814A1 (en) | 2022-11-16 | 2024-05-23 | Basf Se | Fungicidal mixture comprising substituted pyridines |
| WO2024104815A1 (en) | 2022-11-16 | 2024-05-23 | Basf Se | Substituted benzodiazepines as fungicides |
| WO2024104822A1 (en) | 2022-11-16 | 2024-05-23 | Basf Se | Substituted tetrahydrobenzodiazepine as fungicides |
| EP4389210A1 (en) | 2022-12-21 | 2024-06-26 | Basf Se | Heteroaryl compounds for the control of invertebrate pests |
| CN115956568A (en) * | 2023-01-05 | 2023-04-14 | 河南科技学院 | Flutriafol-containing bactericidal composition |
| WO2024165343A1 (en) | 2023-02-08 | 2024-08-15 | Basf Se | New substituted quinoline compounds for combatitng phytopathogenic fungi |
| WO2024194038A1 (en) | 2023-03-17 | 2024-09-26 | Basf Se | Substituted pyridyl/pyrazidyl dihydrobenzothiazepine compounds for combatting phytopathogenic fungi |
| EP4455137A1 (en) | 2023-04-24 | 2024-10-30 | Basf Se | Pyrimidine compounds for the control of invertebrate pests |
| WO2024223034A1 (en) | 2023-04-26 | 2024-10-31 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors xvi |
| EP4467535A1 (en) | 2023-05-25 | 2024-11-27 | Basf Se | Lactam pesticidal compounds |
| EP4488269A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488273A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488270A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4574819A1 (en) | 2023-12-22 | 2025-06-25 | Basf Se | Diazinone compounds for the control of invertebrate pests |
| EP4640052A1 (en) | 2024-04-24 | 2025-10-29 | Basf Se | Mixtures of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors with at least one further pesticide i |
| WO2025223904A1 (en) | 2024-04-24 | 2025-10-30 | Basf Se | Mixtures of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors with at least one further pesticide i |
| WO2026021910A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
| WO2026021912A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
| WO2026021911A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
| WO2026021909A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
Also Published As
| Publication number | Publication date |
|---|---|
| CN1114590C (en) | 2003-07-16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1114590C (en) | Unsaturated oximino ether bactericide | |
| CN1036305C (en) | Herbicidal composition and its application | |
| CN1310899C (en) | Benzopyrone compounds with pest killing and sterilizing activity and preparation and use | |
| CN1201657C (en) | Methoxy methyl acrylate compounds as bactericidal agent | |
| CN1305858C (en) | Substituted azole compounds and their preparation and application | |
| CN1060473C (en) | Microbicidal triazolyl derivative | |
| CN1094939C (en) | Microbiocidal benzotriazoles compounds | |
| CN1062711C (en) | Pesticide and fungicide of unsaturated oximido ethers | |
| CN1141295A (en) | Process for preparing 4-benzoyl isoxazole derivatives | |
| CN1022015C (en) | Fungicidal compositions containing thiazole derivatives | |
| CN1113062C (en) | Dihydrofuran carboxamides | |
| KR20220012866A (en) | Fungicide Aryl Amidine | |
| US20020013472A1 (en) | Oxyranyle-triazoline thiones and their use as microbicides | |
| CN1869032A (en) | Coumarin kind compound and its preparation and application | |
| CN1887847A (en) | Substituted p-trifluoromethyl phenyl ether compound and its prepn and application | |
| CN103539768A (en) | Benzofuran substituted oxime ether compounds and preparation method and applications thereof | |
| CN86106320A (en) | Imidazole compounds, their preparation and their use as fungicides | |
| CN87104156A (en) | Benzo buprofezin derivate | |
| CN101747263B (en) | Pyridyloxy phenoxyalkanoic acids compound and application | |
| CN87101273A (en) | Preparation method of fungicide azole-based derivatives | |
| CN1309898A (en) | Pesticide containing 0-benzyl oximeether derivative and its use | |
| CN1067650A (en) | biocidal compound | |
| CN1927847A (en) | Application of isoxazole compounds as bactericide | |
| CN1284768C (en) | Thioethers fungicides | |
| CN1258284A (en) | Fluoromethoxyimino compound |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| ASS | Succession or assignment of patent right |
Owner name: SINOCHEM CORPORATION; SHENYANG CHEMICAL ENGINEERI Free format text: FORMER OWNER: SHENYANG CHEMICAL ENGINEERING INST. Effective date: 20080711 |
|
| C41 | Transfer of patent application or patent right or utility model | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20080711 Address after: F6-F12, middle seat of Kai Chen World Trade Centre, 28 Fuxing Man Street, Xicheng District, Beijing, zip code: 100031 Co-patentee after: Shenyang Research Institute of Chemical Industry Patentee after: Sinochem Corp. Address before: No 8, Shen Liaodong Road, Tiexi District, Liaoning, Shenyang: 110021 Patentee before: Shenyang Research Institute of Chemical Industry |
|
| ASS | Succession or assignment of patent right |
Owner name: CHINA CHEMISTRY CO., LTD. Free format text: FORMER OWNER: SINOCHEM CORPORATION Effective date: 20091016 |
|
| C41 | Transfer of patent application or patent right or utility model | ||
| C56 | Change in the name or address of the patentee | ||
| CP03 | Change of name, title or address |
Address after: F6-F12, middle seat of Kai Chen World Trade Centre, 28 Fuxing Man Street, Xicheng District, Beijing, zip code: 100031 Co-patentee after: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Co.,Ltd. Patentee after: Sinochem Corp. Address before: F6-F12, middle seat of Kai Chen World Trade Centre, 28 Fuxing Man Street, Xicheng District, Beijing, zip code: 100031 Co-patentee before: Shenyang Research Institute of Chemical Industry Patentee before: Sinochem Corp. |
|
| TR01 | Transfer of patent right |
Effective date of registration: 20091016 Address after: 28, Fuxing Man Street, Xicheng District, Beijing, zip code: 100031 Co-patentee after: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Co.,Ltd. Patentee after: SINOCHEM Corp. Address before: F6-F12, middle seat of Kai Chen World Trade Centre, 28 Fuxing Man Street, Xicheng District, Beijing, zip code: 100031 Co-patentee before: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Co.,Ltd. Patentee before: Sinochem Corp. |
|
| C41 | Transfer of patent application or patent right or utility model | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20160122 Address after: 110021 Liaodong Road, Tiexi District, Liaoning, No. 8-1, No. Patentee after: SHENYANG SINOCHEM AGROCHEMICALS R&D Co.,Ltd. Address before: 100031 Beijing, Xicheng District, the door of the revitalization of the main street, No. 28 Patentee before: SINOCHEM Corp. Patentee before: SHENYANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Co.,Ltd. |
|
| CX01 | Expiry of patent term |
Granted publication date: 20030716 |
|
| CX01 | Expiry of patent term |