CN1201657C - Methoxy methyl acrylate compounds as bactericidal agent - Google Patents
Methoxy methyl acrylate compounds as bactericidal agent Download PDFInfo
- Publication number
- CN1201657C CN1201657C CN 03120882 CN03120882A CN1201657C CN 1201657 C CN1201657 C CN 1201657C CN 03120882 CN03120882 CN 03120882 CN 03120882 A CN03120882 A CN 03120882A CN 1201657 C CN1201657 C CN 1201657C
- Authority
- CN
- China
- Prior art keywords
- compound
- composition
- powdery mildew
- mildew
- preparation
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C69/00—Esters of carboxylic acids; Esters of carbonic or haloformic acids
- C07C69/66—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety
- C07C69/73—Esters of carboxylic acids having esterified carboxylic groups bound to acyclic carbon atoms and having any of the groups OH, O—metal, —CHO, keto, ether, acyloxy, groups, groups, or in the acid moiety of unsaturated acids
- C07C69/734—Ethers
- C07C69/736—Ethers the hydroxy group of the ester being etherified with a hydroxy compound having the hydroxy group bound to a carbon atom of a six-membered aromatic ring
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/36—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing at least one carboxylic group or a thio analogue, or a derivative thereof, and a singly bound oxygen or sulfur atom attached to the same carbon skeleton, this oxygen or sulfur atom not being a member of a carboxylic group or of a thio analogue, or of a derivative thereof, e.g. hydroxy-carboxylic acids
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C67/00—Preparation of carboxylic acid esters
- C07C67/30—Preparation of carboxylic acid esters by modifying the acid moiety of the ester, such modification not being an introduction of an ester group
- C07C67/31—Preparation of carboxylic acid esters by modifying the acid moiety of the ester, such modification not being an introduction of an ester group by introduction of functional groups containing oxygen only in singly bound form
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C67/00—Preparation of carboxylic acid esters
- C07C67/30—Preparation of carboxylic acid esters by modifying the acid moiety of the ester, such modification not being an introduction of an ester group
- C07C67/333—Preparation of carboxylic acid esters by modifying the acid moiety of the ester, such modification not being an introduction of an ester group by isomerisation; by change of size of the carbon skeleton
- C07C67/343—Preparation of carboxylic acid esters by modifying the acid moiety of the ester, such modification not being an introduction of an ester group by isomerisation; by change of size of the carbon skeleton by increase in the number of carbon atoms
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
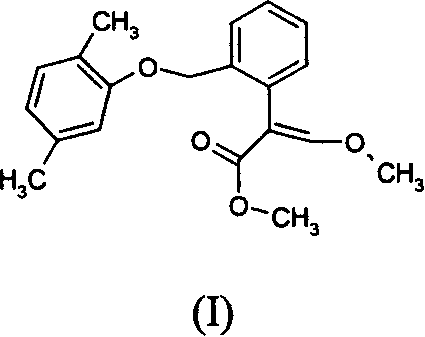
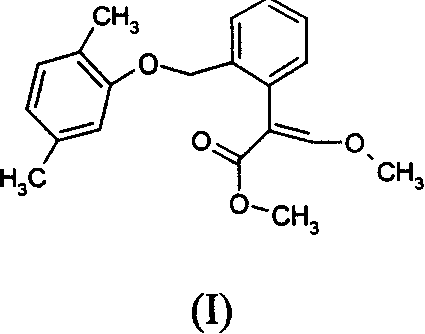
本发明涉及含有结构式I所示2-(2-((2,5-二甲基苯氧基)甲基苯基))-3-甲氧基丙烯酸甲酯及其立体异构体的杀真菌化合物的组合物,其使用方法,以及上述活性成分的化合物及其制备方法。生物活性测定表明:化合物I对防治农业上真菌产生的病害有很高的生物活性,对防治白粉病、霜霉病、炭咀病有特效。
The invention relates to a fungicide containing 2-(2-((2,5-dimethylphenoxy)methylphenyl))-3-methoxymethyl acrylate and its stereoisomers shown in structural formula I Compositions of compounds, methods of use thereof, and compounds of the above active ingredients and methods of preparation thereof. The biological activity test shows that the compound I has high biological activity for the prevention and treatment of diseases produced by fungi in agriculture, and has special effects for the prevention and treatment of powdery mildew, downy mildew and charcoal gnat.
Description
本发明技术领域Technical field of the present invention
本发明涉及一种甲氧基丙烯酸甲酯类农用杀菌剂。本发明进一步涉及一种具有高效杀菌效果的甲氧基丙烯酸甲酯化合物,其制备方法,其杀菌剂组合物及其用途。The invention relates to a methyl methoxyacrylate agricultural fungicide. The present invention further relates to a methyl methoxyacrylate compound with high-efficiency bactericidal effect, its preparation method, its bactericide composition and its application.
为了获得在很小的剂量下就可以控制各种真菌病害化合物,本发明发明者们合成了其中的一些化合物,这些化合物具有广谱杀菌活性可用于防治作物上的由藻菌纲、卵菌纲、子囊菌纲和半知菌纲等多种真菌引起的病害,而且由于这一化合物具有很高的生物活性使得在很低的剂量下就可以获得很好防治效果。本类化合物对白粉病、霜霉病、炭疽病表现出很好的生物活性外,对葡萄的黑痘病、葡萄白锈病也有防治作用。In order to obtain compounds that can control various fungal diseases at a very small dose, the inventors of the present invention have synthesized some of these compounds. , Ascomycetes and Deuteromycetes and other fungi, and because this compound has high biological activity, it can obtain good control effect at a very low dose. This kind of compound not only shows good biological activity against powdery mildew, downy mildew and anthracnose, but also has control effect on grape black pox and grape white rust.
本发明提供的化合物不但具有很高的生物活性,而且对环境非常友好,对环境没有污染。本发明提供化合物不但具有很高的生物活性,而且同世界类似产品比较生产成本低,是一个非常有前途的农用杀菌剂新品种。The compound provided by the invention not only has high biological activity, but also is very friendly to the environment and has no pollution to the environment. The compound provided by the invention not only has high biological activity, but also has low production cost compared with similar products in the world, and is a very promising new variety of agricultural fungicide.
背景技术 Background technique
在US 4,914,128号专利申请中公开了一些甲氧基丙烯酸甲酯类化合物,并公开了其中的化合物具有杀菌活性。Some methyl methoxyacrylate compounds are disclosed in US 4,914,128 patent application, and it is disclosed that the compounds have fungicidal activity.
本申请人意外地发现,其通式结构中的一些未具体公开的化合物,具有优异的杀真菌活性,并由此完成了本发明。The applicant unexpectedly found that some undisclosed compounds in the general structure have excellent fungicidal activity, and thus completed the present invention.
甲氧基丙烯酸甲酯类杀菌剂是从天然化合物通过结构改造而来的高效农用杀真菌剂。它们特点是杀菌广、对其它生物毒性低、环境相容性好。世界上有许多农药大公司进行这方面研究,发现了几个有很好市场前景的甲氧基丙烯酸甲酯类化合物。该类杀菌剂市场占有比例越来越大,将可能取代三唑类杀菌剂成为农用杀菌剂第一大品种。Methyl methoxyacrylate fungicides are high-efficiency agricultural fungicides derived from natural compounds through structural modification. They are characterized by wide sterilization, low toxicity to other organisms, and good environmental compatibility. Many large pesticide companies in the world have conducted research in this area and discovered several methyl methoxyacrylate compounds with good market prospects. The market share of this type of fungicide is increasing, and it may replace triazole fungicides to become the largest variety of agricultural fungicides.
本发明概述Summary of the invention
本发明提供了新的甲氧基丙烯酸甲酯类化合物和其制备方法,及其杀菌剂组合物和用途。本发明甲氧基丙烯酸甲酯杀菌剂化合物的化学名称为:The invention provides a new methyl methoxyacrylate compound, a preparation method thereof, and a fungicide composition and use thereof. The chemical name of methyl methoxyacrylate bactericide compound of the present invention is:
(E)-2-(2-((2,5-二甲基苯氧基)甲基苯基))-3-甲氧基丙烯酸甲酯(E)-2-(2-((2,5-dimethylphenoxy)methylphenyl))-3-methoxymethyl acrylate
结构式为:The structural formula is:
本发明的化合物可以通过以下方法制备:Compounds of the present invention can be prepared by the following methods:
方法一 反应如下:Method 1 reacts as follows:
化合物I可以这样来制备:在适当的溶剂中,加入式II的2,5-二甲基苯酚和式III的2-(2-卤代甲基苯基)-3-甲氧基丙烯酸甲酯,再加入适当的碱,在适当的温度下和适当时间内反应制得。其中III中的X离去基团如卤素,优选为氯、溴或碘。Compound I can be prepared by adding 2,5-dimethylphenol of formula II and methyl 2-(2-halomethylphenyl)-3-methoxyacrylate of formula III in a suitable solvent , then add an appropriate base, react at an appropriate temperature and within an appropriate time. Wherein the leaving group X in III is such as halogen, preferably chlorine, bromine or iodine.
优选溶剂为丙酮或四氢呋喃。Preferred solvents are acetone or tetrahydrofuran.
其中碱包括有机碱和无机碱:有机碱包括三乙胺、吡啶等;无机碱包括氢氧化钠、氢氧化钾、氢化钠、碳酸钠、碳酸钾等。本发明优选碱为碳酸钠或碳酸钾。The bases include organic bases and inorganic bases: organic bases include triethylamine, pyridine, etc.; inorganic bases include sodium hydroxide, potassium hydroxide, sodium hydride, sodium carbonate, potassium carbonate, etc. The preferred base of the present invention is sodium carbonate or potassium carbonate.
优选温度为0~100℃。The preferred temperature is 0 to 100°C.
方法二 反应如下:Method 2 The reaction is as follows:
方法二制备化合物I过程为:在适当的溶剂中,加入式II的2,5-二甲基苯酚和式IV化合物,再加入适当的碱,在适当的温度下和适当时间内反应制得V。其中IV中的X离去基团如卤素,优选氯、溴或碘。V在适当的溶剂中,再加入适当的碱,加入适量的甲酸甲酯,在适当的温度下和适当时间内反应制得VI。VI在适当的溶剂中,加入甲基化试剂,如硫酸二甲酯、碘甲烷等,再加入适当的碱,在适当的温度下和适当时间内反应制得化合物I。Method 2 The process of preparing compound I is: in an appropriate solvent, add 2,5-dimethylphenol of formula II and compound of formula IV, then add an appropriate base, and react at an appropriate temperature and within an appropriate time to obtain V . Wherein the leaving group X in IV is such as halogen, preferably chlorine, bromine or iodine. V is in an appropriate solvent, then add an appropriate base, add an appropriate amount of methyl formate, and react at an appropriate temperature and within an appropriate time to obtain VI. VI In an appropriate solvent, add a methylating agent, such as dimethyl sulfate, methyl iodide, etc., and then add an appropriate base, and react at an appropriate temperature and for an appropriate time to obtain compound I.
其中在制备V时:优选溶剂为丙酮或四氢呋喃。Wherein when preparing V: the preferred solvent is acetone or tetrahydrofuran.
其中碱包括有机碱和无机碱:有机碱包括三乙胺、吡啶等;无机碱包括氢氧化钠、氢氧化钾、氢化钠、碳酸钠、碳酸钾等。本发明优选碱为碳酸钠或碳酸钾。The bases include organic bases and inorganic bases: organic bases include triethylamine, pyridine, etc.; inorganic bases include sodium hydroxide, potassium hydroxide, sodium hydride, sodium carbonate, potassium carbonate, etc. The preferred base of the present invention is sodium carbonate or potassium carbonate.
优选温度为0~100℃。The preferred temperature is 0 to 100°C.
在制备VI时优选碱为甲醇钠或氢化钠等;优选溶剂为乙醚或甲醇。优选温度为零下5~100℃。When preparing VI, the preferred base is sodium methoxide or sodium hydride, etc.; the preferred solvent is ether or methanol. The preferred temperature is minus 5 to 100°C.
其中在制备I时:优选溶剂为丙酮或四氢呋喃。Wherein when preparing I: the preferred solvent is acetone or tetrahydrofuran.
其中碱包括有机碱和无机碱:有机碱包括三乙胺、吡啶等;无机碱包括氢氧化钠、氢氧化钾、氢化钠、碳酸钠、碳酸钾等。本发明优选碱为碳酸钠或碳酸钾。The bases include organic bases and inorganic bases: organic bases include triethylamine, pyridine, etc.; inorganic bases include sodium hydroxide, potassium hydroxide, sodium hydride, sodium carbonate, potassium carbonate, etc. The preferred base of the present invention is sodium carbonate or potassium carbonate.
优选温度为0~100℃。The preferred temperature is 0 to 100°C.
本发明的化合物I是有效的杀真菌剂。特别适合于防治下列植物病害:The compounds I according to the invention are effective fungicides. Especially suitable for the control of the following plant diseases:
大麦和小麦白粉病、蔬菜白粉病、瓜类白粉病、果树白粉病、葡萄白粉病、草莓白粉病及花卉白粉病;小麦和大麦条锈病、叶锈病和其它锈病。Barley and wheat powdery mildew, vegetable powdery mildew, melon powdery mildew, fruit tree powdery mildew, grape powdery mildew, strawberry powdery mildew and flower powdery mildew; wheat and barley stripe rust, leaf rust and other rust diseases.
黄瓜霜霉病,葡萄霜霉病;瓜类上的炭疽病等病害。Cucumber downy mildew, grape downy mildew; anthracnose and other diseases on melons.
本发明还提供了如上所定义的化合物的杀菌组合物及其制备方法。The present invention also provides a fungicidal composition of the compound as defined above and a preparation method thereof.
本发明的组合物,优选含从0.1~99.0%重量的式(I)的活性成份。需要时可在组合物中加入合适的载体和表面活性剂成分。The composition of the present invention preferably contains from 0.1 to 99.0% by weight of the active ingredient of formula (I). Suitable carriers and surfactant ingredients can be added to the composition as desired.
本发明组合物的制备方法是将化合物I和至少一种载体混合。这种组合物还可以含有其它的农药活性成分。The composition of the invention is prepared by mixing compound I and at least one carrier. Such compositions may also contain other pesticidally active ingredients.
本发明的载体系满足下述条件的物质:它与活性成分配制后便于施用于待处理的位点,例如可以是植物、种子或土壤;或者有利于贮存、运输或操作。载体可以是固体或液体,和可使用任何通常在配制杀虫、杀菌组合物中所用的载体。The carrier system of the present invention is a substance that satisfies the following conditions: it can be conveniently applied to the site to be treated after being formulated with active ingredients, such as plants, seeds or soil; or it is convenient for storage, transportation or handling. The carrier can be solid or liquid, and any carrier commonly used in formulating insecticidal and fungicidal compositions can be used.
合适的固体载体包括天然和合成的硅酸盐、例如硅藻土、滑石、硅镁土、硅酸铝(高岭土)、蒙脱石和云母;碳酸钙、硫酸钙、硫酸铵;合成的氧化硅酸钙或硅酸铝;元素如碳和硫;天然的和合成的树脂如苯并呋喃树脂,聚氯乙烯和苯乙烯聚合物和共聚物;固体多氯苯酚;沥青;蜡如蜂蜡、石腊。Suitable solid carriers include natural and synthetic silicates such as diatomaceous earth, talc, attapulgite, aluminum silicates (kaolins), montmorillonite and mica; calcium carbonate, calcium sulfate, ammonium sulfate; synthetic oxidized silicic acid Calcium or aluminum silicates; elements such as carbon and sulfur; natural and synthetic resins such as benzofuran resins, polyvinyl chloride and styrene polymers and copolymers; solid polychlorinated phenols; bitumen; waxes such as beeswax, paraffin.
合适的液体载体包括水;醇如异丙醇和乙醇;酮如丙酮、甲基乙基酮、甲基异丁基酮和环己酮;醚;芳烃如苯、甲苯和二甲苯;石油馏分如煤油和矿物油;氯代烃如四氯化碳、全氯乙烯和三氯乙烷,通常,这些液体的混合物也是合适的。Suitable liquid carriers include water; alcohols such as isopropanol and ethanol; ketones such as acetone, methyl ethyl ketone, methyl isobutyl ketone, and cyclohexanone; ethers; aromatic hydrocarbons such as benzene, toluene, and xylene; and mineral oil; chlorinated hydrocarbons such as carbon tetrachloride, perchlorethylene, and trichloroethane; often, mixtures of these liquids are also suitable.
杀虫、杀菌组合物通常加工成浓缩物的形式并以此用于运输,在施用之前由使用者将其稀释。少量的表面活性剂载体的存在有助于稀释过程。这样,按照本发明的组合物中,优选含有表面活性剂。Insecticidal and fungicidal compositions are usually processed and transported in the form of concentrates, which are diluted by the user before application. The presence of a small amount of surfactant carrier aids in the dilution process. Thus, in the composition according to the present invention, a surfactant is preferably contained.
表面活性剂可以是乳化剂、分散剂或润湿剂:它可以是离子表面活性剂或非离子表面活性剂。如:聚丙烯酸和木质素磺酸的钠盐或钙盐;分子中含至少12个碳原子的脂肪酸或脂肪胺或酰胺;环氧乙烷和或环氧丙烷的缩合物;甘醇,山梨醇、蔗糖或季戊四醇脂肪酸脂及这些酯与环氧乙烷和或环氧丙环的缩合物;脂肪醇或烷基苯酚如对辛基苯酚或对辛基苯酚与环氧乙烷和或环氧丙烷的缩合物;这些缩合产物的硫酸盐或磺酸盐;在分子中含有至少10个碳原子的硫酸或磺酸酯的碱金属或碱土金属盐,优选钠盐,例如硫酸月桂酯钠,硫酸仲烷基酯钠,磺化蓖麻油钠盐,磺酸烷基芳基酯钠如十二烷基苯磺酸钠盐。A surfactant can be an emulsifying agent, a dispersing agent or a wetting agent: it can be an ionic or a nonionic surfactant. Such as: sodium or calcium salts of polyacrylic acid and lignosulfonic acid; fatty acids or fatty amines or amides with at least 12 carbon atoms in the molecule; condensates of ethylene oxide and or propylene oxide; glycol, sorbitol , sucrose or pentaerythritol fatty acid esters and condensation products of these esters with ethylene oxide and or propylene oxide; fatty alcohols or alkylphenols such as p-octylphenol or p-octylphenol with ethylene oxide and or propylene oxide condensates; sulfate or sulfonate salts of these condensation products; alkali metal or alkaline earth metal salts, preferably sodium salts, of sulfuric acid or sulfonate containing at least 10 carbon atoms in the molecule, for example sodium lauryl sulfate, secondary sulfate Sodium alkyl esters, sodium sulfonated castor oil, sodium alkylaryl sulfonates such as sodium dodecylbenzenesulfonate.
本发明的组合物的实例是可湿性粉剂、粉剂、颗粒剂、可乳化的浓缩剂、乳剂、悬浮浓缩剂、气雾剂和烟雾剂。可湿性粉剂通常含20-80%重量活性成分,且通常除固体惰性载体之外,还含有2~10%重量分散剂,且若需要或加入0~10%重量稳定剂和或其它添加剂如渗透剂或粘着剂。粉剂通常可成型为具有可湿性粉剂相似的组成但有分散剂的粉剂浓缩剂,在地里进一步用固体载体稀释,得到通常含0.5~10%重量活性成分的组合物。粒剂通常制成具有10和100目大小,且可用成团或注入技术制备。通常,粒剂含0.1~80%重量的活性成分和0~10%重量添加剂如稳定剂、表面活性剂、缓释剂等。所谓的“可流动干粉”由具有相对高浓度活性成分的相对小的颗粒组成。可乳化浓缩剂除溶剂外,当需要时通常含有共溶剂,1~50%W/V活性成分,2~20%W/V乳化剂和0~20%W/V其他添加剂如稳定剂、渗透剂和腐蚀抑制剂。悬浮浓缩齐通常含有10~80%重量的活性成分、0.5~15%重量的分散剂、0.1~10%重量的其它添加剂如消泡剂、腐蚀抑制剂、稳定剂、渗透剂和粘着剂。Examples of compositions according to the invention are wettable powders, dusts, granules, emulsifiable concentrates, emulsions, suspension concentrates, aerosols and aerosols. Wettable powders usually contain 20-80% by weight of active ingredients, and usually in addition to solid inert carriers, also contain 2-10% by weight of dispersants, and if necessary, or add 0-10% by weight of stabilizers and or other additives such as osmotic agent or adhesive. Dusts can generally be formed as powder concentrates having a composition similar to wettable powders but with a dispersing agent, which can be further diluted in the field with a solid carrier to give a composition generally containing 0.5 to 10% by weight of active ingredient. Granules are usually produced in 10 and 100 mesh sizes and can be prepared by agglomeration or injection techniques. Usually, granules contain 0.1-80% by weight of active ingredients and 0-10% by weight of additives such as stabilizers, surfactants, slow-release agents and the like. So-called "dry flowable powders" consist of relatively small granules having a relatively high concentration of active ingredient. Emulsifiable concentrates usually contain co-solvents, 1-50% W/V active ingredients, 2-20% W/V emulsifiers and 0-20% W/V other additives such as stabilizers, osmotic agents and corrosion inhibitors. Suspension concentrates usually contain 10-80% by weight of active ingredients, 0.5-15% by weight of dispersants, and 0.1-10% by weight of other additives such as defoamers, corrosion inhibitors, stabilizers, penetrating agents and adhesives.
水分散剂和乳化剂,例如通过用水稀释按照本发明的可湿性粉剂或浓缩物得到的组合物,也列入本发明范围。所说的乳剂可具有油包水或水包油两个类型。Aqueous dispersants and emulsifiers, such as compositions obtained by diluting the wettable powders or concentrates according to the invention with water, are also within the scope of the invention. Said emulsions may be of the water-in-oil or oil-in-water type.
通过在组合物中加入其他一种或多种杀真菌剂,使其能比单独的化合物(I)具有更广的活性。此外,其他杀真菌剂可对化合物(I)的杀菌活性具有增效作用。可以包含在本发明组合物中的杀真菌剂化合物的实例中:克菌丹、灭菌丹、嘧霉胺、十三吗啉、氯苯嘧啶醇、咯菌腈、氧化亚酮、恶醚唑、代森锌、代森锰锌、福美双、敌菌丹、异菌脲、甲基菌核利、甲乙菌核利、己唑醇、腈菌唑、戊唑醇、戊菌隆、霜脲氰、双胍辛醋酸盐、硫菌威、环菌胺、氟吗啉、氟哇唑、SSF-129,picoxystrobin,metrafenone、pyraclostrobin、resveratro、dimoxystrobin、UBF-307、BASF490F、ICIA5504、TH-164、RH7592、酯菌脲、烯唑醇、氟酰脲、多菌灵、苯菌灵、三唑酮、环唑醇、乙霉威、甲基托布津、土菌消、丁苯吗啉、霜霉威、甲霜灵、呋霜灵、苯霜灵、恶酰胺、磺菌威、啶斑肟、苯锈啶、嘧菌胺、烯酰吗啉、拌种咯、拌种强、丙环唑、百菌清、二嗪哝、硫酸铜、抑菌灵、乙磷铝、恶霉灵、等。By adding one or more other fungicides to the composition, it can have wider activity than compound (I) alone. In addition, other fungicides may have a synergistic effect on the fungicidal activity of compound (I). Among the examples of fungicide compounds that may be included in the composition of the present invention: Captan, Folpet, Pyrimethanil, Tridemorph, Chlorpyrimidol, Fludioxonil, Oxidone, Fefeconazole , Zinc, mancozeb, thiram, captafol, iprodione, proclozolid, proclozolid, hexaconazole, myclobutanil, tebuconazole, penbicolon, cyprodione Cyanide, biguanide octyl acetate, thiocarb, cycloheximide, flumorph, fluoxazole, SSF-129, picoxystrobin, metrafenone, pyraclostrobin, resveratro, dimoxystrobin, UBF-307, BASF490F, ICIA5504, TH-164, RH7592, Diprodicarb, Diniconazole, Fluoride, Carbendazim, Benomyl, Triadimefon, Cycloconazole, Dimethocarb, Thioprazine-methyl, Tufenxiao, Penfenmorph, Downy mildew Carboxyl, metalaxyl, furalaxyl, benalaxyl, oxamide, sulfonylcarb, pymetroxime, fenpropidin, azoxystrobin, dimethomorph, seed dressing, seed dressing strong, propiconazole, Chlorothalonil, diazine, copper sulfate, bacteriostat, fosfo-aluminum, hymexazol, etc.
可以与本发明的化合物混合形成组合物的杀虫剂有:杀虫剂溴螨酯、三氯杀螨醇、甲基1605、1605、杀螟松、地亚农、丁硫克百威、毒死蜱、锐尽特、除尽、灭多威、杀虫单、杀虫双、巴丹、阿维菌素、氯菊酯、氯氰菊酯、胺菊酯、七氟菊酯、氟氯氰菊酯、氰戊菊酯、氟虫脲、杀虫脲、定虫脲、唑螨酯、吡虫啉、双氧威、三唑磷、喹螨酯、杀螨隆、哒螨灵、四螨嗪等。Insecticides that can be mixed with the compounds of the present invention to form compositions include: insecticides bromofen, dicofol, methyl 1605, 1605, fenitrothion, diananon, carbosulfan, chlorpyrifos, and regent . .
具体实施方式 Detailed ways
通过以下具体的实例来对本发明进行详细的说明,除非另外指明本申请中的 份数一重 量计:The present invention is described in detail by the following specific examples, unless otherwise specified parts in the application are by weight :
合成实施例:Synthetic Example:
在室温下将12.2g2,5-二甲基苯酚加到500ml盛有150ml干燥丙酮的三口瓶中,然后加入13.8g碳酸钾,室温搅拌20分钟,然后慢慢加入28.5g(E)-2-(2-(溴甲基)苯基)-3-甲氧基丙烯酸甲酯。然后回流反应3小时结束,过滤,浓缩,得粗品。用乙酸乙酯和石油醚的混合液(1∶4)为洗脱液进行柱层析得到化合物I 26.08g,为白色固体,收率为80%。熔点:108~110℃At room temperature, add 12.2g of 2,5-xylenol to a 500ml three-neck flask filled with 150ml of dry acetone, then add 13.8g of potassium carbonate, stir at room temperature for 20 minutes, then slowly add 28.5g of (E)-2- Methyl (2-(bromomethyl)phenyl)-3-methoxyacrylate. Then the reflux reaction was completed for 3 hours, filtered and concentrated to obtain a crude product. Column chromatography was performed using a mixture of ethyl acetate and petroleum ether (1:4) as the eluent to obtain 26.08 g of compound I as a white solid with a yield of 80%. Melting point: 108~110℃
1HNMR:2.235(3H,s),2.279(3H,s),3.706(3H,s),3.835(3H,s),4.944(2H,s),6.584(1H,s)6.655-6.673(1H,d),7.013-7.032(1H,d),7.164-7.168(2H,d),7.300-7.7.382(2H,m),7.596(1H,s),7.617(1H,s) 1 H NMR: 2.235 (3H, s), 2.279 (3H, s), 3.706 (3H, s), 3.835 (3H, s), 4.944 (2H, s), 6.584 (1H, s) 6.655-6.673 (1H, d), 7.013-7.032 (1H, d), 7.164-7.168 (2H, d), 7.300-7.7.382 (2H, m), 7.596 (1H, s), 7.617 (1H, s)
注:1HNMR谱是用CDCl3溶剂记录。在实验中所用的缩写如下:Note: 1 HNMR spectra were recorded with CDCl 3 solvent. The abbreviations used in the experiments are as follows:
NMR=核磁共振s=单峰d=双峰m=多峰NMR = nuclear magnetic resonance s = singlet d = doublet m = multimodal
制剂实施例Formulation example
制剂实施例1Formulation Example 1
本发明的化合物40公斤、硅藻土53公斤、C12-20醇硫酸酯4公斤、十二烷基苯磺酸钠3公斤,将以上成分均匀混合,粉碎,得含有效成分40%的可湿性粉剂。40 kilograms of compounds of the present invention, 53 kilograms of diatomite, 4 kilograms of C12-20 alcohol sulfate, 3 kilograms of sodium dodecylbenzenesulfonate, the above ingredients are uniformly mixed, pulverized, and a wettable compound containing 40% of active ingredients is obtained. powder.
制剂实施例2Formulation Example 2
本发明的化物30公斤、二甲苯33公斤、二甲基甲酰胺30公斤、聚氧乙烯烷基丙醚7公斤,将以上成分均匀混合、溶解、得含有效成分30%的乳剂。30 kg of compound of the present invention, 33 kg of xylene, 30 kg of dimethylformamide, and 7 kg of polyoxyethylene alkyl propyl ether are uniformly mixed and dissolved to obtain an emulsion containing 30% active ingredient.
制剂实施例3Formulation Example 3
本发明的化合物10公斤、滑石粉89公斤、聚氧乙烯烷基丙醚公斤,将以上成分均匀混合,粉碎,得含有效成分10%的粉剂。10 kg of the compound of the present invention, 89 kg of talcum powder, and kg of polyoxyethylene alkyl propyl ether were uniformly mixed and pulverized to obtain a powder containing 10% of the active ingredient.
制剂实施例4Formulation Example 4
本发明的化合物5公斤、粘土73公斤、膨润土20公斤、二辛基硫代丁二酸钠1公斤、磷酸钠1公斤,将以上成分均匀混合,充分粉碎后,加入适量水。再充分混合,造粒,干燥后,得含有效成分5%的颗粒剂。5 kg of the compound of the present invention, 73 kg of clay, 20 kg of bentonite, 1 kg of sodium dioctyl thiosuccinate, and 1 kg of sodium phosphate. The above ingredients are uniformly mixed, fully pulverized, and an appropriate amount of water is added. Then fully mix, granulate, and dry to obtain granules containing 5% active ingredient.
制剂实施例5Formulation Example 5
本发明的化合物10公斤,木质素磺酸钠4公斤,十二烷基苯磺酸钠1公斤,黄原酸1公斤,水84公斤,将以上成分均匀混合,湿式研磨到粒度为1微米以下,得到含有效成分为10%的胶悬剂。10 kg of compound of the present invention, 4 kg of sodium lignin sulfonate, 1 kg of sodium dodecylbenzene sulfonate, 1 kg of xanthic acid, 84 kg of water, uniformly mix the above ingredients, and wet grind until the particle size is below 1 micron , to obtain a suspension containing 10% active ingredient.
制剂实施例6Formulation Example 6
本发明化合物8公斤、代森锰锌50公斤、高岭土30公斤、十二烷基苯磺酸钠4公斤、木质素磺酸钠8公斤,将上述成分混合,充分粉碎后,得混剂含量为58%的可湿性粉剂。8 kilograms of compounds of the present invention, 50 kilograms of mancozeb, 30 kilograms of kaolin, 4 kilograms of sodium dodecylbenzenesulfonate, and 8 kilograms of sodium lignosulfonate are mixed with the above-mentioned components, and after fully pulverizing, the content of the mixed agent is: 58% wettable powder.
制剂实施例7Formulation Example 7
本发明化合物1公斤、三唑酮20公斤、高岭土64公斤、十二烷基苯磺酸钠6公斤、木质素磺酸钠9公斤,将以上成分均匀混合,充分粉碎后,得混合含量为21%的可湿性粉剂。1 kilogram of compound of the present invention, 20 kilograms of triadimefon, 64 kilograms of kaolin, 6 kilograms of sodium dodecylbenzenesulfonate, 9 kilograms of sodium lignosulfonate, the above ingredients are uniformly mixed, after fully pulverizing, the mixed content is 21 % wettable powder.
药效实施例:Medicinal effect embodiment:
本发明的化合物,同现有杀菌剂比具有佷好生物活性,可以在很低的剂量下很好地防治由真菌引起的病害,对白粉病、锈病、霜霉病、炭疽病有特别好的效果。如小麦、大麦的白粉病、黄瓜白粉、南瓜白粉、草莓白粉病、葡萄白粉病,小麦、大麦锈病,葡萄霜霉病、黄瓜霜霉病,黄瓜炭疽病。另位对葡萄黑痘病、白锈病也有一定的抑制作用。Compared with the existing fungicides, the compound of the present invention has good biological activity, can prevent and treat diseases caused by fungi well at very low doses, and is particularly effective against powdery mildew, rust, downy mildew and anthracnose Effect. Such as wheat, barley powdery mildew, cucumber powdery mildew, pumpkin powdery mildew, strawberry powdery mildew, grape powdery mildew, wheat, barley rust, grape downy mildew, cucumber downy mildew, cucumber anthracnose. The other also has a certain inhibitory effect on grape black pox and white rust.
药效实施例1:防治黄瓜白粉病保护与治疗作用试验(室内)Efficacy Example 1: Prevention and Treatment of Cucumber Powdery Mildew Protection and Therapeutic Effect Test (Indoor)
试验药剂为本发明化合物I(含量5%的乳油)设10、5、2.5、1.25、0.625、0.3125ppm六个浓度。对照ZA-1963(即先正达公司picoxystrobin,即相似专利中商品化的化合物)(含量5%的乳油)设10、5、2.5、1.25、0.625、0.3125ppm六个浓度。对照药剂20%三唑酮乳油,设100、50、25ppm。选择生长至2叶一心期的盆栽两片真叶平展的黄瓜健苗喷药,预防试验喷药后24小时接种,治疗试验接种后24小时喷药。接种后黄瓜幼移置人工气候箱内保湿培养,7天后调查防效。结果见表1:The test agent is compound I of the present invention (5% emulsifiable concentrate) with six concentrations of 10, 5, 2.5, 1.25, 0.625, and 0.3125 ppm. Control ZA-1963 (namely picoxystrobin from Syngenta Company, which is a commercially available compound in a similar patent) (5% emulsifiable concentrate) has six concentrations of 10, 5, 2.5, 1.25, 0.625, 0.3125 ppm. The control drug 20% triadimefon EC was set at 100, 50, and 25 ppm. Select the healthy cucumber seedlings with two flat true leaves growing to the stage of two leaves and one heart for spraying, inoculate 24 hours after the spraying for the prevention test, and spray for 24 hours after the inoculation for the treatment test. After inoculation, the young cucumbers were placed in an artificial climate box for moisturizing cultivation, and the control effect was investigated after 7 days. The results are shown in Table 1:
表1黄瓜白粉病保护与治疗作用试验结果:
保护及治疗试验结果表明:化合物I预防略优于治疗。0.625ppm预防为85.31%,治疗为64.81%。同时发现化合物(I)的预防和治疗效果的明显活性优于ZA-1963,特别是治疗作用。化合物I的生物活性远远好于三唑酮。The results of the protection and treatment test showed that the prevention of compound I was slightly better than the treatment. 0.625ppm is 85.31% for prevention and 64.81% for treatment. At the same time, it was found that compound (I) had better preventive and therapeutic effects than ZA-1963, especially the therapeutic effect. The biological activity of compound I is far better than that of triadimefon.
药效实施例2防治小麦白粉病保护与治疗作用试验(室内)Efficacy Example 2 Prevention and Treatment of Wheat Powdery Mildew Protection and Therapeutic Effect Test (Indoor)
试验药剂为本发明化合物I(含量5%的乳油)设25、12.5、6.25、3.125、1.563、0.781ppm六个浓度。对照ZA-1963(即先正达公司picoxystrobin,即相似专利中商品化的化合物)(含量5%的乳油)设25、12.5、6.25、3.125、1.563、0.781ppm六个浓度。对照药剂20%三唑酮乳油,设100、50、25ppm。选择生长至2叶一心期的盆栽小麦喷药,预防试验喷药后24小时接种,治疗试验接种后24小时喷药。接种后小麦移置人工气候箱内保湿培养,7天后调查防效。结果见表2The test agent is compound I of the present invention (5% emulsifiable concentrate) with six concentrations of 25, 12.5, 6.25, 3.125, 1.563, and 0.781 ppm. Control ZA-1963 (namely picoxystrobin from Syngenta Company, which is commercialized compound in similar patent) (5% emulsifiable concentrate) has six concentrations of 25, 12.5, 6.25, 3.125, 1.563, 0.781ppm. The control drug 20% triadimefon EC was set at 100, 50, and 25 ppm. Potted wheat grown to the 2-leaf one-heart stage was selected for spraying, inoculated 24 hours after spraying in the prevention test, and sprayed 24 hours after inoculation in the treatment test. After inoculation, the wheat was transferred to an artificial climate box for moisture cultivation, and the control effect was investigated 7 days later. The results are shown in Table 2
表2小表白粉病保护与治疗作用试验结果
保护及治疗试验结果表明:发现化合物I的预防和治疗效果的明显活性优于ZA-1963,治疗作用更加明显。化合物I的生物活性远远好于三唑酮。The results of the protection and treatment test showed that the prevention and treatment effect of compound I was found to be better than that of ZA-1963, and the treatment effect was more obvious. The biological activity of compound I is far better than that of triadimefon.
药效实施例3对小麦白粉病模拟田间试验Efficacy Example 3 Simulated Field Test on Wheat Powdery Mildew
化合物(I)处理浓度为50,25,12.5,6.25,3.125ppm,对照药剂ZA-1963的处理浓度与化合物I相同,三唑酮的处理浓度为100,50,25ppm。将盆栽小麦培养至4-5叶期,任其自然发病至中期,按上述所设浓度进行叶面喷雾处理,10天后调查防效。结果见表3The treatment concentration of compound (I) was 50, 25, 12.5, 6.25, 3.125ppm, the treatment concentration of the control agent ZA-1963 was the same as that of compound I, and the treatment concentration of triadimefon was 100, 50, 25ppm. Cultivate the potted wheat to the 4-5 leaf stage, let it naturally develop to the middle stage, spray the leaves according to the concentration set above, and investigate the control effect after 10 days. The results are shown in Table 3
表3对小麦白粉病模拟田间试验
模拟田间试验结果表明,化合物I对盆栽小麦自然发病至中期时施药,仍表现出很高的防治作用,并且防效优于对照药剂ZA-1963和三唑酮。The simulated field test results showed that compound I still exhibited a high control effect on potted wheat when it was applied at the mid-stage of natural onset, and the control effect was better than that of the control agents ZA-1963 and triadimefon.
药效实施例4持效期试验Drug effect embodiment 4 duration test
化合物I和对照药剂ZA-1963的处理浓度为50,25mg/l,测试对象为小麦白粉病。在盆栽小麦两片真叶平展时喷药,喷药后24小时接种小麦白粉菌,接种后的小麦移至温室内保湿培养,处理后7、10、15、20、25、30天调查防效。结果见表4The treatment concentrations of compound I and control agent ZA-1963 were 50 and 25 mg/l, and the test object was wheat powdery mildew. Spray when the two true leaves of potted wheat are flat, inoculate powdery mildew of wheat 24 hours after spraying, move the inoculated wheat to the greenhouse for moisture cultivation, and investigate the control effect 7, 10, 15, 20, 25, and 30 days after treatment . The results are shown in Table 4
表4持效期试验结果
持效期试验表明:在温室人工接种的条件下,化合物I的持效期比ZA-1963略长。The persistence period test showed that: under the conditions of artificial inoculation in the greenhouse, the persistence period of compound I was slightly longer than that of ZA-1963.
药效实施例5对南瓜白粉病的防效试验(田间)Control effect test (field) to pumpkin powdery mildew of drug effect embodiment 5
田间防效南瓜白粉病试验2002年6月在浙江绍兴市现代农业园区的大棚内进行。The field test of pumpkin powdery mildew control effect was carried out in June 2002 in the greenhouse of Modern Agricultural Park in Shaoxing City, Zhejiang Province.
化合物I(10%水悬剂)设50,25,12.5,6.25ppm四个浓度,对照药剂为20%粉锈宁EC设浓度为100ppm。另设清水对照。每处理4次重复。施药7天后调查。结果见表5Compound I (10% aqueous suspension) was set at four concentrations of 50, 25, 12.5, and 6.25 ppm, and the control drug was 20% fenrustin EC at a concentration of 100 ppm. In addition, clear water was used as a control. Each treatment was repeated 4 times. Survey after 7 days of application. The results are shown in Table 5
表5对南瓜白粉病的防效试验(田间)结果
注:CK防效栏为病指增加值。Note: The CK prevention effect column is the added value of the disease index.
从表5中可以看出:化合物在6.25ppm对南瓜白粉有很高的活性,明显优于对照药剂粉锈宁。It can be seen from Table 5 that the compound has a high activity on pumpkin white powder at 6.25ppm, which is obviously better than that of the contrast agent Fenxiening.
药效实施例6对黄瓜白粉病的防效试验(田间)Control effect test (field) to cucumber powdery mildew of drug effect embodiment 6
田间防治黄瓜白粉病试验2002年6月在浙江绍兴市现代农业园区的大棚内进行。The field test of controlling cucumber powdery mildew was carried out in June 2002 in a greenhouse in the modern agricultural park of Shaoxing City, Zhejiang Province.
化合物I(10%水悬剂)设50,25,12.5,6.25ppm四个浓度,对照药剂为20%粉锈宁EC设浓度为100ppm。另设清水对照。每处理4次重复。施药7天后调查。结果见表6Compound I (10% aqueous suspension) was set at four concentrations of 50, 25, 12.5, and 6.25 ppm, and the control drug was 20% fenrustin EC at a concentration of 100 ppm. In addition, clear water was used as a control. Each treatment was repeated 4 times. Survey after 7 days of application. The results are shown in Table 6
表6对黄瓜白粉病的防效试验(田间)结果
注:CK防效栏为病指增加值。Note: The CK prevention effect column is the added value of the disease index.
从表6中可以看出:化合物I在6.25ppm对黄瓜白粉有很高的活性,明显优于对照药剂粉锈宁。As can be seen from Table 6: compound I has very high activity to cucumber white powder at 6.25ppm, which is obviously better than the contrast agent fenxining.
药效实施例7化合物(I)对黄瓜霜霉病防效试验(室内)Drug effect embodiment 7 compound (I) is to cucumber downy mildew control effect test (indoor)
化合物I处理浓度为100,50,25,12.5,6.25ppm,对照药剂ZA-1963的处理浓度与化合物I相同,甲霜灵处理浓度为500ppm。选择生长至2叶一心期的盆栽两片真叶平展的黄瓜健苗喷药,预防试验喷药后24小时接种,治疗试验接种后24小时喷药。接种后黄瓜幼移置人工气候箱内保湿培养,7天后调查防效,结果见表7The treatment concentration of compound I was 100, 50, 25, 12.5, 6.25 ppm, the treatment concentration of the control agent ZA-1963 was the same as that of compound I, and the treatment concentration of metalaxyl was 500 ppm. Select the healthy cucumber seedlings with two flat true leaves growing to the stage of two leaves and one heart for spraying, inoculate 24 hours after the spraying for the prevention test, and spray for 24 hours after the inoculation for the treatment test. After inoculation, the young cucumbers were transplanted into an artificial climate box for moisturizing cultivation, and the control effect was investigated after 7 days. The results are shown in Table 7
表7对黄瓜霜霉病的防效试验(室内)结果
在温室条件下化合物I对黄瓜霜霉病的防效在相同浓度下明显优于对照药剂ZA-1963,明显优于甲霜灵。Under greenhouse conditions, the control effect of compound I on cucumber downy mildew was significantly better than that of the control agent ZA-1963 at the same concentration, and significantly better than metalaxyl.
药效实施例8对葡萄霜霉病的防效试验(田间)Control effect test (field) to grape downy mildew of drug effect embodiment 8
田间防治葡萄霜霉病试验2002年7月在安微省马鞍山市当涂县石桥乡农业科技示范场进行。The field test of grape downy mildew control was carried out in July 2002 in the agricultural science and technology demonstration field of Shiqiao Township, Dangtu County, Ma'anshan City, Anhui Province.
化合物I(5%乳油)设200,100,50,25,12.5ppm五个浓度,对照药剂72%杜邦克露可湿性粉剂设1000ppm浓度。另设空白对照,每处理重复4次。喷药7天后调查。结果如表8Five concentrations of compound I (5% emulsifiable concentrate) were set at 200, 100, 50, 25, and 12.5 ppm, and the concentration of the control drug 72% Dupont Clew WP was set at 1000 ppm. A blank control was also set up, and each treatment was repeated 4 times. Survey after 7 days of spraying. The results are shown in Table 8
表8对葡萄霜霉病的防效试验(田间)结果
从结果可以看出,化合物I在防治葡萄霜霉在200ppm与对照药克露1000ppm相当。药效实施例9对黄瓜霜霉病的防效试验(田间)As can be seen from the results, compound I is comparable to the control drug Kelu 1000ppm at 200ppm in the prevention and treatment of grape downy mildew. Control effect test (field) to cucumber downy mildew of drug effect embodiment 9
田间防治黄瓜霜霉病试验2002年7月在安微省马鞍山市当涂县石桥乡农业科技示范场进行。The field experiment of controlling cucumber downy mildew was carried out in July 2002 in the agricultural science and technology demonstration field of Shiqiao Township, Dangtu County, Ma'anshan City, Anhui Province.
化合物I(5%乳油)设200,100,50,25,12.5ppm五个浓度,对照药剂72%杜邦克露可湿性粉剂设1000ppm。另设空白对照,每处理重复4次。喷药7天后调查。结果如表9Compound I (5% emulsifiable concentrate) was set at five concentrations of 200, 100, 50, 25, and 12.5 ppm, and the control drug 72% DuPont Kelu WP was set at 1000 ppm. A blank control was also set up, and each treatment was repeated 4 times. Survey after 7 days of spraying. The results are shown in Table 9
表9对黄瓜霜霉病的防效试验(田间)结果
从结果可以看出,化合物I在防治黄瓜霜霉在200ppm与对照药克露1000ppm相当。As can be seen from the results, Compound I is comparable to the control drug Kelu 1000ppm at 200ppm in the control of cucumber downy mildew.
药效实施例10对黄瓜炭疽病防效果试验(室内)Efficacy Example 10 Anti-effect Test on Cucumber Anthracnose (Indoor)
化合物I和对照药剂ZA-1963的处理浓度为500,250,125,62.5,31.25ppm。测试对象为黄瓜炭疽病,盆栽黄瓜苗生长至一张真叶期时喷药,喷药24小时后接种炭疽病孢子悬浮液,接种后移入人工气候箱内,保持湿度100%,温度为20度,接种24小时后保持相对湿度为85%。7天后进行调查。结果见表10The treatment concentrations of compound I and control agent ZA-1963 were 500, 250, 125, 62.5, 31.25 ppm. The test object is cucumber anthracnose. Potted cucumber seedlings are sprayed when they grow to a true leaf stage. 24 hours after spraying, they are inoculated with anthracnose spore suspension. After inoculation, they are moved into an artificial climate box, and the humidity is kept at 100% and the temperature is 20 degrees. , maintain a relative humidity of 85% 24 hours after inoculation. Survey after 7 days. The results are shown in Table 10
表10对黄瓜炭疽病试验结果。(室内)
在温室条件下化合物I对黄瓜炭疽病的防效在相同浓度下略优于对照药剂ZA-1963The control effect of compound I on cucumber anthracnose was slightly better than that of the control agent ZA-1963 at the same concentration under greenhouse conditions
Claims (9)
Priority Applications (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 03120882 CN1201657C (en) | 2003-03-25 | 2003-03-25 | Methoxy methyl acrylate compounds as bactericidal agent |
| PCT/CN2004/000226 WO2004084632A1 (en) | 2003-03-25 | 2004-03-19 | Fungicides containing methoxy acrylic acid methyl ester compound |
| BRPI0409037-3A BRPI0409037A (en) | 2003-03-25 | 2004-03-19 | fungicides containing methoxy acrylic acid methyl ester compound |
| AU2004224838A AU2004224838A1 (en) | 2003-03-25 | 2004-03-19 | Fungicides containing methoxy acrylic acid methyl ester compound |
| TR2005/03847T TR200503847T1 (en) | 2003-03-25 | 2004-03-19 | Fungicides containing methoxy acrylic acid methyl ester compound. |
| ZA200508026A ZA200508026B (en) | 2003-03-25 | 2005-10-04 | Fungicides containing methoxy acrylic acid methyl ester compound |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN 03120882 CN1201657C (en) | 2003-03-25 | 2003-03-25 | Methoxy methyl acrylate compounds as bactericidal agent |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN1456054A CN1456054A (en) | 2003-11-19 |
| CN1201657C true CN1201657C (en) | 2005-05-18 |
Family
ID=29411572
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN 03120882 Expired - Lifetime CN1201657C (en) | 2003-03-25 | 2003-03-25 | Methoxy methyl acrylate compounds as bactericidal agent |
Country Status (6)
| Country | Link |
|---|---|
| CN (1) | CN1201657C (en) |
| AU (1) | AU2004224838A1 (en) |
| BR (1) | BRPI0409037A (en) |
| TR (1) | TR200503847T1 (en) |
| WO (1) | WO2004084632A1 (en) |
| ZA (1) | ZA200508026B (en) |
Families Citing this family (180)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101524076B (en) * | 2005-08-17 | 2012-11-07 | 浙江省化工研究院有限公司 | Fungicide composition |
| CN100401889C (en) * | 2005-08-17 | 2008-07-16 | 浙江化工科技集团有限公司 | Fungicide composition |
| CN101642119B (en) * | 2009-08-24 | 2013-06-05 | 陕西上格之路生物科学有限公司 | Agricultural sterilizing composition containing kresoxim-methyl benzene |
| WO2011132967A2 (en) * | 2010-04-21 | 2011-10-27 | 주식회사 오스코텍 | Alpha-arylmethoxyacrylate derivative, preparation method thereof, and pharmaceutical composition containing same |
| CN102115458B (en) * | 2010-11-25 | 2019-12-13 | 大连九信精细化工有限公司 | Synthetic method of 3-methoxy-2-aryl methyl acrylate compound |
| CN102919240B (en) * | 2010-12-08 | 2014-08-13 | 陕西美邦农药有限公司 | Tridemorph-containing sterilizing composition |
| CN102835399B (en) * | 2012-09-27 | 2014-04-30 | 陕西上格之路生物科学有限公司 | Metrafenone-containing bactericidal composition |
| CN103975927B (en) * | 2013-04-07 | 2016-06-08 | 海南正业中农高科股份有限公司 | A kind of bactericidal composition containing cyflufenamid and pyraoxystrobin or ZJ0712 and application |
| CN103598193B (en) * | 2013-10-23 | 2015-04-01 | 江苏丰登作物保护股份有限公司 | Picoxystrobin and thiram containing sterilization composition and application thereof |
| EP3214937B1 (en) | 2014-11-07 | 2024-07-03 | Basf Se | Pesticidal mixtures |
| WO2016174042A1 (en) | 2015-04-27 | 2016-11-03 | BASF Agro B.V. | Pesticidal compositions |
| EP3371177A1 (en) | 2015-11-02 | 2018-09-12 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| US20180317490A1 (en) | 2015-11-04 | 2018-11-08 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| BR112018009579A2 (en) | 2015-11-13 | 2018-11-06 | Basf Se | compound of formula i, mixture, agrochemical composition, compound use and fungal control method |
| WO2017081310A1 (en) | 2015-11-13 | 2017-05-18 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| MX2018006244A (en) | 2015-11-19 | 2018-11-09 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi. |
| CR20180332A (en) | 2015-11-19 | 2018-10-18 | Basf Se | OXADIAZOLS REPLACED TO FIGHT FITOPATHOGEN FUNGI |
| EP3202267A1 (en) | 2016-02-05 | 2017-08-09 | Basf Se | Pesticidal mixtures |
| EP3205208A1 (en) | 2016-02-09 | 2017-08-16 | Basf Se | Mixtures and compositions comprising paenibacillus strains or fusaricidins and chemical pesticides |
| WO2017153200A1 (en) | 2016-03-10 | 2017-09-14 | Basf Se | Fungicidal mixtures iii comprising strobilurin-type fungicides |
| US11425909B2 (en) | 2016-03-16 | 2022-08-30 | Basf Se | Use of tetrazolinones for combating resistant phytopathogenic fungi on fruits |
| US11241012B2 (en) | 2016-03-16 | 2022-02-08 | Basf Se | Use of tetrazolinones for combating resistant phytopathogenic fungi on soybean |
| UA125210C2 (en) | 2016-03-16 | 2022-02-02 | Басф Се | APPLICATION OF TETRAZOLINONS TO CONTROL RESISTANT PHYTOPATHOGENIC MUSHROOMS ON CEREALS |
| US10986839B2 (en) | 2016-04-11 | 2021-04-27 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| CN105766919A (en) * | 2016-04-22 | 2016-07-20 | 广东中迅农科股份有限公司 | Bactericidal composition containing triflumizole and (E)2-[2-(2, 5-dimethylphenoxytoluene)-phenyl]-3-methoxyacrylate |
| WO2018050421A1 (en) | 2016-09-13 | 2018-03-22 | Basf Se | Fungicidal mixtures i comprising quinoline fungicides |
| WO2018054711A1 (en) | 2016-09-26 | 2018-03-29 | Basf Se | Pyridine compounds for controlling phytopathogenic harmful fungi |
| WO2018054723A1 (en) | 2016-09-26 | 2018-03-29 | Basf Se | Pyridine compounds for controlling phytopathogenic harmful fungi |
| WO2018054721A1 (en) | 2016-09-26 | 2018-03-29 | Basf Se | Pyridine compounds for controlling phytopathogenic harmful fungi |
| WO2018065182A1 (en) | 2016-10-04 | 2018-04-12 | Basf Se | Reduced quinoline compounds as antifuni agents |
| WO2018073110A1 (en) | 2016-10-20 | 2018-04-26 | Basf Se | Quinoline compounds as fungicides |
| WO2018114393A1 (en) | 2016-12-19 | 2018-06-28 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| EP3339297A1 (en) | 2016-12-20 | 2018-06-27 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| EP3338552A1 (en) | 2016-12-21 | 2018-06-27 | Basf Se | Use of a tetrazolinone fungicide on transgenic plants |
| BR112019014061A2 (en) | 2017-01-23 | 2020-02-04 | Basf Se | compounds of formula i, intermediates b, intermediates c, intermediates ii and intermediates d, composition, use, method to combat phytopathogenic fungi, seed and process for the synthesis of the compounds of formula i |
| WO2018149754A1 (en) | 2017-02-16 | 2018-08-23 | Basf Se | Pyridine compounds |
| BR112019015338B1 (en) | 2017-02-21 | 2023-03-14 | Basf Se | COMPOUNDS OF FORMULA I, AGROCHEMICAL COMPOSITION, COATED SEED, USE OF THE COMPOUNDS AND METHOD TO COMBAT HARMFUL PHYTOPATHOGENIC FUNGI |
| CN110461854A (en) | 2017-03-31 | 2019-11-15 | 巴斯夫欧洲公司 | Process for the preparation of chiral 2,3-dihydrothiazolo[3,2-A]pyrimidine-4-onium compounds |
| BR112019020879A2 (en) | 2017-04-06 | 2020-04-28 | Basf Se | compounds, composition, use of a compound of formula i, method to combat phytopathogenic fungi, seed and intermediates |
| WO2018184970A1 (en) | 2017-04-07 | 2018-10-11 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018188962A1 (en) | 2017-04-11 | 2018-10-18 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| CA3059301A1 (en) | 2017-04-20 | 2018-10-25 | Pi Industries Ltd. | Novel phenylamine compounds |
| EP3618629A1 (en) | 2017-05-02 | 2020-03-11 | Basf Se | Fungicidal mixture comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| EP3619207B1 (en) | 2017-05-04 | 2021-06-23 | Basf Se | Substituted 5-(haloalkyl)-5-hydroxy-isoxazolines for combating phytopathogenic fungi |
| WO2018202491A1 (en) | 2017-05-04 | 2018-11-08 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2018202737A1 (en) | 2017-05-05 | 2018-11-08 | Basf Se | Fungicidal mixtures comprising triazole compounds |
| WO2018210661A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2018210660A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2018210658A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| WO2018210659A1 (en) | 2017-05-15 | 2018-11-22 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| IL270873B2 (en) | 2017-05-30 | 2023-04-01 | Basf Se | “derivatives of pyridine and pyrazine compounds, composition comprising them and their use as fungicides” |
| WO2018219797A1 (en) | 2017-06-02 | 2018-12-06 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018234139A1 (en) | 2017-06-19 | 2018-12-27 | Basf Se | 2 - [[5- (TRIFLUOROMETHYL) -1,2,4-OXADIAZOL-3-YL] ARYLOXY] (THIO) ACETAMIDES FOR THE CONTROL OF PHYTOPATHOGENIC FUNGI |
| WO2019002158A1 (en) | 2017-06-30 | 2019-01-03 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019025250A1 (en) | 2017-08-04 | 2019-02-07 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019038042A1 (en) | 2017-08-21 | 2019-02-28 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| EP3915379A1 (en) | 2017-08-29 | 2021-12-01 | Basf Se | Pesticidal mixtures |
| WO2019042932A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| EP3453706A1 (en) | 2017-09-08 | 2019-03-13 | Basf Se | Pesticidal imidazole compounds |
| US11076596B2 (en) | 2017-09-18 | 2021-08-03 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019057660A1 (en) | 2017-09-25 | 2019-03-28 | Basf Se | Indole and azaindole compounds with substituted 6-membered aryl and heteroaryl rings as agrochemical fungicides |
| US11399543B2 (en) | 2017-10-13 | 2022-08-02 | Basf Se | Substituted 1,2,3,5-tetrahydroimidazo[1,2-a]pyrimidiniumolates for combating animal pests |
| US11147275B2 (en) | 2017-11-23 | 2021-10-19 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| WO2019115511A1 (en) | 2017-12-14 | 2019-06-20 | Basf Se | Fungicidal mixture comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| EP3723485A1 (en) | 2017-12-15 | 2020-10-21 | Basf Se | Fungicidal mixture comprising substituted pyridines |
| CN111670180A (en) | 2017-12-20 | 2020-09-15 | Pi工业有限公司 | Fluoroalkenyl compound, preparation method and use thereof |
| WO2019121143A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Substituted cyclopropyl derivatives |
| AU2019211978B2 (en) | 2018-01-09 | 2024-08-22 | Basf Se | Silylethynyl hetaryl compounds as nitrification inhibitors |
| WO2019137995A1 (en) | 2018-01-11 | 2019-07-18 | Basf Se | Novel pyridazine compounds for controlling invertebrate pests |
| WO2019150311A1 (en) | 2018-02-02 | 2019-08-08 | Pi Industries Ltd. | 1-3 dithiol compounds and their use for the protection of crops from phytopathogenic microorganisms |
| WO2019154665A1 (en) | 2018-02-07 | 2019-08-15 | Basf Se | New pyridine carboxamides |
| CN111683933A (en) | 2018-02-07 | 2020-09-18 | 巴斯夫欧洲公司 | New pyridinecarboxamides |
| EP3530118A1 (en) | 2018-02-26 | 2019-08-28 | Basf Se | Fungicidal mixtures |
| EP3530116A1 (en) | 2018-02-27 | 2019-08-28 | Basf Se | Fungicidal mixtures comprising xemium |
| KR102730587B1 (en) | 2018-02-28 | 2024-11-14 | 바스프 에스이 | Use of N-functionalized alkoxy pyrazole compounds as nitrification inhibitors |
| WO2019166558A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of pyrazole propargyl ethers as nitrification inhibitors |
| WO2019166252A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Fungicidal mixtures comprising fenpropidin |
| PE20211753A1 (en) | 2018-02-28 | 2021-09-06 | Basf Se | USE OF ALCOXYPYRAZOLES AS NITRIFICATION INHIBITORS |
| EA202092018A1 (en) | 2018-03-01 | 2021-02-01 | Басф Агро Б.В. | FUNGICIDE COMPOSITIONS OF MEFENTRIFLUKONAZOLE |
| EP3533333A1 (en) | 2018-03-02 | 2019-09-04 | Basf Se | Fungicidal mixtures comprising pydiflumetofen |
| EP3533331A1 (en) | 2018-03-02 | 2019-09-04 | Basf Se | Fungicidal mixtures comprising pydiflumetofen |
| EP3536150A1 (en) | 2018-03-06 | 2019-09-11 | Basf Se | Fungicidal mixtures comprising fluxapyroxad |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019219464A1 (en) | 2018-05-15 | 2019-11-21 | Basf Se | Substituted trifluoromethyloxadiazoles for combating phytopathogenic fungi |
| KR102727205B1 (en) * | 2018-05-15 | 2024-11-06 | 바스프 에스이 | Mixtures containing benzpyrimoxane and oxazosulpirin, uses thereof and methods of application thereof |
| WO2019224092A1 (en) | 2018-05-22 | 2019-11-28 | Basf Se | Pesticidally active c15-derivatives of ginkgolides |
| WO2020002472A1 (en) | 2018-06-28 | 2020-01-02 | Basf Se | Use of alkynylthiophenes as nitrification inhibitors |
| PL3826983T3 (en) | 2018-07-23 | 2024-09-09 | Basf Se | Use of substituted 2-thiazolines as nitrification inhibitors |
| CN112424147B (en) | 2018-07-23 | 2023-06-30 | 巴斯夫欧洲公司 | Use of substituted thiazolidine compounds as nitrification inhibitors |
| AR115984A1 (en) | 2018-08-17 | 2021-03-17 | Pi Industries Ltd | 1,2-DITIOLONE COMPOUNDS AND THEIR USES |
| EP3613736A1 (en) | 2018-08-22 | 2020-02-26 | Basf Se | Substituted glutarimide derivatives |
| EP3628158A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| AU2019348280A1 (en) | 2018-09-28 | 2021-04-22 | Basf Se | Method of controlling pests by seed treatment application of a mesoionic compound or mixture thereof |
| EP3628156A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method for controlling pests of sugarcane, citrus, rapeseed, and potato plants |
| EP3628157A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| EP3643705A1 (en) | 2018-10-24 | 2020-04-29 | Basf Se | Pesticidal compounds |
| WO2020095161A1 (en) | 2018-11-05 | 2020-05-14 | Pi Industries Ltd. | Nitrone compounds and use thereof |
| EP3670501A1 (en) | 2018-12-17 | 2020-06-24 | Basf Se | Substituted [1,2,4]triazole compounds as fungicides |
| EP3696177A1 (en) | 2019-02-12 | 2020-08-19 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| KR20210131350A (en) | 2019-02-20 | 2021-11-02 | 바스프 에스이 | Pesticide Mixtures Containing Pyrazole Compounds |
| EP3730489A1 (en) | 2019-04-25 | 2020-10-28 | Basf Se | Heteroaryl compounds as agrochemical fungicides |
| EP3769623A1 (en) | 2019-07-22 | 2021-01-27 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| BR112021019416A2 (en) | 2019-05-29 | 2021-12-07 | Basf Se | Compounds, composition, methods of protecting crops and combating, controlling, preventing or protecting against infestations, non-therapeutic method of treating infested animals, seed and use |
| WO2020244970A1 (en) | 2019-06-06 | 2020-12-10 | Basf Se | New carbocyclic pyridine carboxamides |
| WO2020244969A1 (en) | 2019-06-06 | 2020-12-10 | Basf Se | Pyridine derivatives and their use as fungicides |
| WO2020244968A1 (en) | 2019-06-06 | 2020-12-10 | Basf Se | Fungicidal n-(pyrid-3-yl)carboxamides |
| EP3766879A1 (en) | 2019-07-19 | 2021-01-20 | Basf Se | Pesticidal pyrazole derivatives |
| WO2021063735A1 (en) | 2019-10-02 | 2021-04-08 | Basf Se | New bicyclic pyridine derivatives |
| WO2021063736A1 (en) | 2019-10-02 | 2021-04-08 | Basf Se | Bicyclic pyridine derivatives |
| JP7785003B2 (en) | 2019-12-23 | 2025-12-12 | ビーエーエスエフ ソシエタス・ヨーロピア | Enzyme-enhanced root uptake of pesticide compounds. |
| WO2021170463A1 (en) | 2020-02-28 | 2021-09-02 | BASF Agro B.V. | Methods and uses of a mixture comprising alpha-cypermethrin and dinotefuran for controlling invertebrate pests in turf |
| BR112022017563A2 (en) | 2020-03-04 | 2022-10-18 | Basf Se | USE OF COMPOUNDS, AGROCHEMICAL COMPOSITION AND METHOD TO FIGHT HARMFUL PHYTOPATOGENIC FUNGI |
| BR112022020612A2 (en) | 2020-04-14 | 2022-11-29 | Basf Se | FUNGICIDAL MIXTURE, AGROCHEMICAL COMPOSITION, NON-THERAPEUTIC USE OF THE MIXTURE AND METHOD TO CONTROL HARMFUL PHYTOPATHOGENIC FUNGI |
| EP3903582A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors ii |
| EP3903584A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors iv |
| BR112022021631A2 (en) | 2020-04-28 | 2022-12-06 | Basf Se | COMPOUNDS, COMPOSITION, METHODS TO COMBAT OR CONTROL INVERTEBRATE PEST, TO PROTECT GROWING PLANTS AND TO TREAT OR PROTECT AN ANIMAL, SEED AND USE OF A COMPOUND |
| EP3903583A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors iii |
| EP3903581A1 (en) | 2020-04-28 | 2021-11-03 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors i |
| EP3909950A1 (en) | 2020-05-13 | 2021-11-17 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP3945089A1 (en) | 2020-07-31 | 2022-02-02 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors v |
| WO2021249800A1 (en) | 2020-06-10 | 2021-12-16 | Basf Se | Substituted [1,2,4]triazole compounds as fungicides |
| EP3960727A1 (en) | 2020-08-28 | 2022-03-02 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors vi |
| EP3939961A1 (en) | 2020-07-16 | 2022-01-19 | Basf Se | Strobilurin type compounds and their use for combating phytopathogenic fungi |
| WO2022017836A1 (en) | 2020-07-20 | 2022-01-27 | BASF Agro B.V. | Fungicidal compositions comprising (r)-2-[4-(4-chlorophenoxy)-2-(trifluoromethyl)phenyl]-1- (1,2,4-triazol-1-yl)propan-2-ol |
| EP3970494A1 (en) | 2020-09-21 | 2022-03-23 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors viii |
| TW202229241A (en) | 2020-09-26 | 2022-08-01 | 印度商皮埃企業有限公司 | Nematocidal compounds and use thereof |
| US20230397607A1 (en) | 2020-10-27 | 2023-12-14 | BASF Agro B.V. | Compositions comprising mefentrifluconazole |
| WO2022090071A1 (en) | 2020-11-02 | 2022-05-05 | Basf Se | Use of mefenpyr-diethyl for controlling phytopathogenic fungi |
| WO2022090069A1 (en) | 2020-11-02 | 2022-05-05 | Basf Se | Compositions comprising mefenpyr-diethyl |
| WO2022106304A1 (en) | 2020-11-23 | 2022-05-27 | BASF Agro B.V. | Compositions comprising mefentrifluconazole |
| EP4018830A1 (en) | 2020-12-23 | 2022-06-29 | Basf Se | Pesticidal mixtures |
| EP4288398A1 (en) | 2021-02-02 | 2023-12-13 | Basf Se | Synergistic action of dcd and alkoxypyrazoles as nitrification inhibitors |
| EP4043444A1 (en) | 2021-02-11 | 2022-08-17 | Basf Se | Substituted isoxazoline derivatives |
| BR112023023592A2 (en) | 2021-05-11 | 2024-03-12 | Basf Se | FUNGICIDAL MIXTURE, AGROCHEMICAL COMPOSITION, USE OF THE MIXTURE AND METHOD TO CONTROL HARMFUL PHYTOPATHOGENIC FUNGI |
| WO2022243107A1 (en) | 2021-05-18 | 2022-11-24 | Basf Se | New substituted pyridines as fungicides |
| CN117355520A (en) | 2021-05-18 | 2024-01-05 | 巴斯夫欧洲公司 | New substituted quinolines for use as fungicides |
| US20240270727A1 (en) | 2021-05-18 | 2024-08-15 | Basf Se | New substituted pyridines as fungicide |
| US20240270658A1 (en) | 2021-05-21 | 2024-08-15 | Basf Se | Use of an N-Functionalized Alkoxy Pyrazole Compound as Nitrification Inhibitor |
| US20240351959A1 (en) | 2021-05-21 | 2024-10-24 | Basf Se | Use of ethynylpyridine compounds as nitrification inhibitors |
| EP4094579A1 (en) | 2021-05-28 | 2022-11-30 | Basf Se | Pesticidal mixtures comprising metyltetraprole |
| CA3223077A1 (en) | 2021-06-21 | 2022-12-29 | Barbara Nave | Metal-organic frameworks with pyrazole-based building blocks |
| EP4119547A1 (en) | 2021-07-12 | 2023-01-18 | Basf Se | Triazole compounds for the control of invertebrate pests |
| AU2022321882A1 (en) | 2021-08-02 | 2024-02-15 | Basf Se | (3-pirydyl)-quinazoline |
| US20250019361A1 (en) | 2021-08-02 | 2025-01-16 | Basf Se | (3-quinolyl)-quinazoline |
| EP4140986A1 (en) | 2021-08-23 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4140995A1 (en) | 2021-08-27 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4151631A1 (en) | 2021-09-20 | 2023-03-22 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2023072671A1 (en) | 2021-10-28 | 2023-05-04 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors ix |
| WO2023072670A1 (en) | 2021-10-28 | 2023-05-04 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors x |
| EP4194453A1 (en) | 2021-12-08 | 2023-06-14 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4198033A1 (en) | 2021-12-14 | 2023-06-21 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP4198023A1 (en) | 2021-12-16 | 2023-06-21 | Basf Se | Pesticidally active thiosemicarbazone compounds |
| EP4238971A1 (en) | 2022-03-02 | 2023-09-06 | Basf Se | Substituted isoxazoline derivatives |
| WO2023203066A1 (en) | 2022-04-21 | 2023-10-26 | Basf Se | Synergistic action as nitrification inhibitors of dcd oligomers with alkoxypyrazole and its oligomers |
| AU2023317620A1 (en) | 2022-08-02 | 2025-02-13 | Basf Se | Pyrazolo pesticidal compounds |
| EP4342885A1 (en) | 2022-09-20 | 2024-03-27 | Basf Se | N-(3-(aminomethyl)-phenyl)-5-(4-phenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-amine derivatives and similar compounds as pesticides |
| EP4361126A1 (en) | 2022-10-24 | 2024-05-01 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors xv |
| WO2024104813A1 (en) | 2022-11-14 | 2024-05-23 | Basf Se | Fungicidal mixture comprising substituted pyridines |
| EP4619399A1 (en) | 2022-11-16 | 2025-09-24 | Basf Se | New substituted tetrahydrobenzoxazepine |
| CN120202195A (en) | 2022-11-16 | 2025-06-24 | 巴斯夫欧洲公司 | Substituted benzodiazepines as fungicides |
| AU2023381249A1 (en) | 2022-11-16 | 2025-05-29 | Basf Se | Fungicidal mixture comprising substituted pyridines |
| WO2024104822A1 (en) | 2022-11-16 | 2024-05-23 | Basf Se | Substituted tetrahydrobenzodiazepine as fungicides |
| EP4619393A1 (en) | 2022-11-16 | 2025-09-24 | Basf Se | Substituted benzodiazepines as fungicides |
| EP4389210A1 (en) | 2022-12-21 | 2024-06-26 | Basf Se | Heteroaryl compounds for the control of invertebrate pests |
| WO2024165343A1 (en) | 2023-02-08 | 2024-08-15 | Basf Se | New substituted quinoline compounds for combatitng phytopathogenic fungi |
| AU2024238668A1 (en) | 2023-03-17 | 2025-09-25 | Basf Se | Substituted pyridyl/pyrazidyl dihydrobenzothiazepine compounds for combatting phytopathogenic fungi |
| EP4455137A1 (en) | 2023-04-24 | 2024-10-30 | Basf Se | Pyrimidine compounds for the control of invertebrate pests |
| CN121335882A (en) | 2023-04-26 | 2026-01-13 | 巴斯夫欧洲公司 | The use of umbelliferone-type compounds to combat plant pathogenic fungi containing amino acid substitutions in mitochondrial cytochrome b proteins that confer resistance to the Qo inhibitor XVI, specifically F129L. |
| EP4467535A1 (en) | 2023-05-25 | 2024-11-27 | Basf Se | Lactam pesticidal compounds |
| EP4488270A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488269A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488273A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4574819A1 (en) | 2023-12-22 | 2025-06-25 | Basf Se | Diazinone compounds for the control of invertebrate pests |
| EP4640052A1 (en) | 2024-04-24 | 2025-10-29 | Basf Se | Mixtures of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors with at least one further pesticide i |
| WO2025223904A1 (en) | 2024-04-24 | 2025-10-30 | Basf Se | Mixtures of strobilurin type compounds for combating phytopathogenic fungi containing an amino acid substitution f129l in the mitochondrial cytochrome b protein conferring resistance to qo inhibitors with at least one further pesticide i |
| WO2026021911A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
| WO2026021909A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
| WO2026021910A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
| WO2026021912A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE3545319A1 (en) * | 1985-12-20 | 1987-06-25 | Basf Ag | ACRYLIC ACID ESTERS AND FUNGICIDES THAT CONTAIN THESE COMPOUNDS |
-
2003
- 2003-03-25 CN CN 03120882 patent/CN1201657C/en not_active Expired - Lifetime
-
2004
- 2004-03-19 TR TR2005/03847T patent/TR200503847T1/en unknown
- 2004-03-19 WO PCT/CN2004/000226 patent/WO2004084632A1/en not_active Ceased
- 2004-03-19 BR BRPI0409037-3A patent/BRPI0409037A/en not_active Application Discontinuation
- 2004-03-19 AU AU2004224838A patent/AU2004224838A1/en not_active Abandoned
-
2005
- 2005-10-04 ZA ZA200508026A patent/ZA200508026B/en unknown
Also Published As
| Publication number | Publication date |
|---|---|
| BRPI0409037A (en) | 2006-03-28 |
| ZA200508026B (en) | 2006-10-25 |
| TR200503847T1 (en) | 2006-04-21 |
| WO2004084632A1 (en) | 2004-10-07 |
| AU2004224838A1 (en) | 2004-10-07 |
| CN1456054A (en) | 2003-11-19 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN1201657C (en) | Methoxy methyl acrylate compounds as bactericidal agent | |
| CN1553770A (en) | Fungicidal compositions based on aryl amidine derivatives and known fungicidal compounds | |
| CN1917760A (en) | Fungicidal compositions comprising pyridylethylbenzamide derivatives and compounds capable of inhibiting ergosterol biosynthesis | |
| KR20150100865A (en) | Synergistic fungicidal mixtures for fungal control in cereals | |
| CN101080170A (en) | Methods of controlling fungal diseases in leguminous plants | |
| CN101052302A (en) | Agricultural or horticultural bactericidal composition and method of controlling plant disease | |
| CN1078043C (en) | Combinations of a fungicide having an azole group with an insecticide having a pyrazole, pyrrole or phenylimidazole group | |
| TW200808183A (en) | Azolymethyloxiranes, their use for controlling phytopathogenic fungi, and compositions comprising them | |
| JP2003522100A (en) | Fungicide 6- (2-halo-4-alkoxyphenyl) -triazolopyrimidine | |
| CN100340167C (en) | synergistic composition | |
| CN1918125A (en) | Fungicidal composition comprising a pyridylethylbenzamide derivative and a compound capable of inhibiting the transport of electrons of the respiratory chain in phytopathogenic fungal organisms | |
| CN1066610C (en) | Microbicide | |
| CN1284456C (en) | Fungicidal composition based on pyridinemethylbenzamide derivatives and valamide derivatives | |
| CN1074251C (en) | Fungicidal compositions | |
| CN1250079C (en) | Fungicide compositions | |
| CN1250080C (en) | Fungicide compositions based on pyridylethylbenzamide and propamocarb derivative | |
| UA72459C2 (en) | 6-(2-halo-4-alkoxyphenyl)triazolepyrimidines, a method for preparation and a fungicide composition | |
| CN1968604A (en) | Herbicidal composition | |
| CN1307877C (en) | Fungicide composition comprising fenamidone and hymexazol | |
| CN1393134A (en) | Composition of fungicide and insecticide containing cyanamethanil | |
| CN1163143C (en) | Fungicide mixture | |
| CN1565182A (en) | Bactericidal compositions of flumorph and enostroburin and synergist | |
| CN1536962A (en) | Composition for controlling phytopathogens and method for controlling phytopathogens | |
| CN1711022A (en) | Fungicidal mixtures for controlling rice pathogens | |
| CN1212767C (en) | fungicidal composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C56 | Change in the name or address of the patentee |
Owner name: ZHEJIANG CHEM-TECH GROUP CO., LTD. Free format text: FORMER NAME: CHEMICAL INDUSTRY INST. OF ZHEJIANG PROV. Owner name: ZHEJIANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY C Free format text: FORMER NAME: ZHEJIANG CHEM-TECH GROUP CO., LTD. |
|
| CP01 | Change in the name or title of a patent holder |
Address after: 310023, Zhejiang, Hangzhou Province, leaving the camp door Patentee after: ZHEJIANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Ltd. Address before: 310023, Zhejiang, Hangzhou Province, leaving the camp door Patentee before: ZHEJIANG CHEM TECH GROUP Co.,Ltd. Address after: 310023, Zhejiang, Hangzhou Province, leaving the camp door Patentee after: ZHEJIANG CHEM TECH GROUP Co.,Ltd. Address before: 310023, Zhejiang, Hangzhou Province, leaving the camp door Patentee before: ZHEJIANG CHEMICAL INDUSTRY Research Institute |
|
| ASS | Succession or assignment of patent right |
Owner name: ZHEJIANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY, Effective date: 20121012 Owner name: SINOCHEM LANTIAN CO., LTD. Free format text: FORMER OWNER: ZHEJIANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY, LTD. Effective date: 20121012 |
|
| C41 | Transfer of patent application or patent right or utility model | ||
| COR | Change of bibliographic data |
Free format text: CORRECT: ADDRESS; FROM: 310023 HANGZHOU, ZHEJIANG PROVINCE TO: 310051 HANGZHOU, ZHEJIANG PROVINCE |
|
| TR01 | Transfer of patent right |
Effective date of registration: 20121012 Address after: 310051 Binjiang District Jiangnan Road, Zhejiang, No. 96, No. Patentee after: SINOCHEM LANTIAN Co.,Ltd. Patentee after: ZHEJIANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Ltd. Address before: 310023, Zhejiang, Hangzhou Province, leaving the camp door Patentee before: ZHEJIANG RESEARCH INSTITUTE OF CHEMICAL INDUSTRY Ltd. |
|
| PE01 | Entry into force of the registration of the contract for pledge of patent right |
Denomination of invention: Methyl Methoxyacrylate compound fungicide Effective date of registration: 20211011 Granted publication date: 20050518 Pledgee: Industrial and Commercial Bank of China Limited Hangzhou Zhaohui sub branch Pledgor: SINOCHEM LANTIAN Co.,Ltd. Registration number: Y2021330001830 |
|
| PE01 | Entry into force of the registration of the contract for pledge of patent right | ||
| PC01 | Cancellation of the registration of the contract for pledge of patent right |
Date of cancellation: 20220507 Granted publication date: 20050518 Pledgee: Industrial and Commercial Bank of China Limited Hangzhou Zhaohui sub branch Pledgor: SINOCHEM LANTIAN Co.,Ltd. Registration number: Y2021330001830 |
|
| PC01 | Cancellation of the registration of the contract for pledge of patent right | ||
| CX01 | Expiry of patent term |
Granted publication date: 20050518 |
|
| CX01 | Expiry of patent term |