WO2022243111A1 - New substituted pyridines as fungicides - Google Patents
New substituted pyridines as fungicides Download PDFInfo
- Publication number
- WO2022243111A1 WO2022243111A1 PCT/EP2022/062622 EP2022062622W WO2022243111A1 WO 2022243111 A1 WO2022243111 A1 WO 2022243111A1 EP 2022062622 W EP2022062622 W EP 2022062622W WO 2022243111 A1 WO2022243111 A1 WO 2022243111A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- compounds
- methyl
- phenyl
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P3/00—Fungicides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D267/00—Heterocyclic compounds containing rings of more than six members having one nitrogen atom and one oxygen atom as the only ring hetero atoms
- C07D267/02—Seven-membered rings
- C07D267/08—Seven-membered rings having the hetero atoms in positions 1 and 4
- C07D267/12—Seven-membered rings having the hetero atoms in positions 1 and 4 condensed with carbocyclic rings or ring systems
- C07D267/14—Seven-membered rings having the hetero atoms in positions 1 and 4 condensed with carbocyclic rings or ring systems condensed with one six-membered ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D498/10—Spiro-condensed systems
Definitions
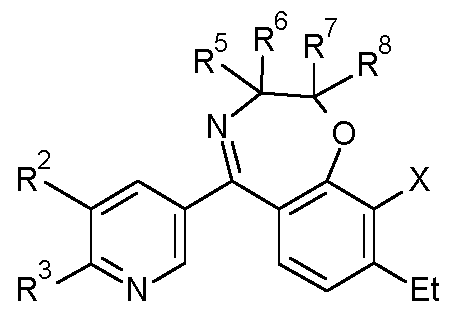
- the present invention relates to new pyridine compounds and the N-oxides and the salts thereof as fungicides as well to their use.
- the invention also relates to the composition comprising at least one compound I, to the method for combating phytopathogenic fungi and to the seed coated with at least one compound of the formula I.
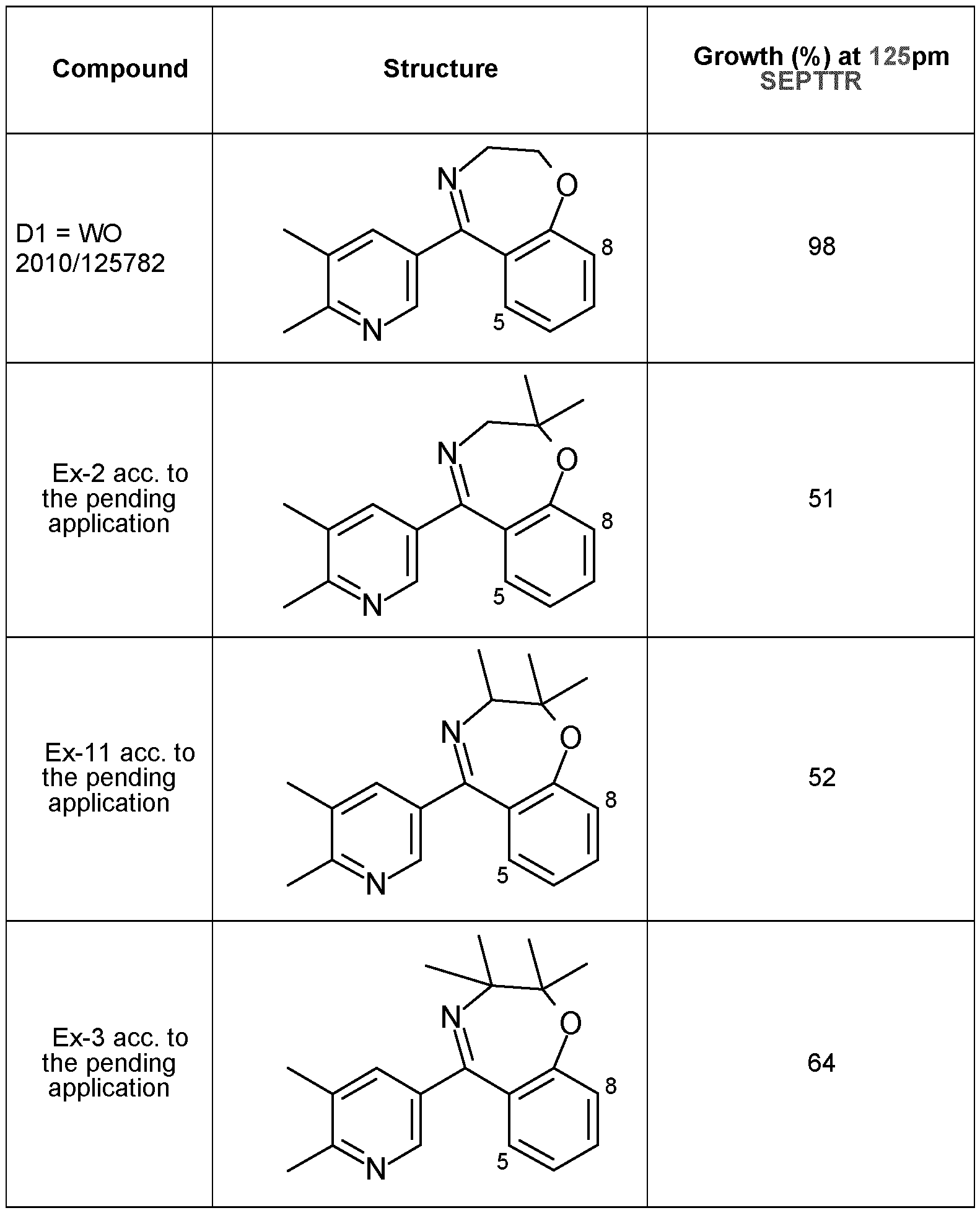
- W0201018686, WO201347441 disclose some pyridine compounds.
- the fungicidal activity of known compounds is unsatisfactory. Based on this, it was an objective of the present invention to provide compounds having improved activity and/or a broader activity spectrum against phytopathogenic fungi.
- Another object of the present invention is to provide fungicides with improved toxicological properties or with improved environmental fate properties.
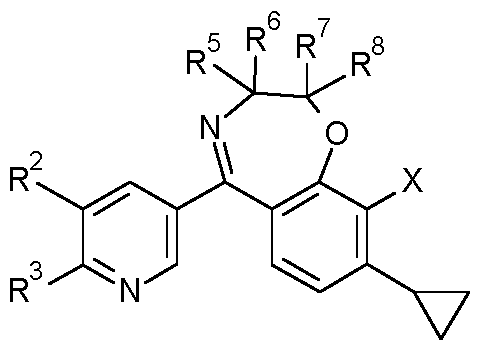
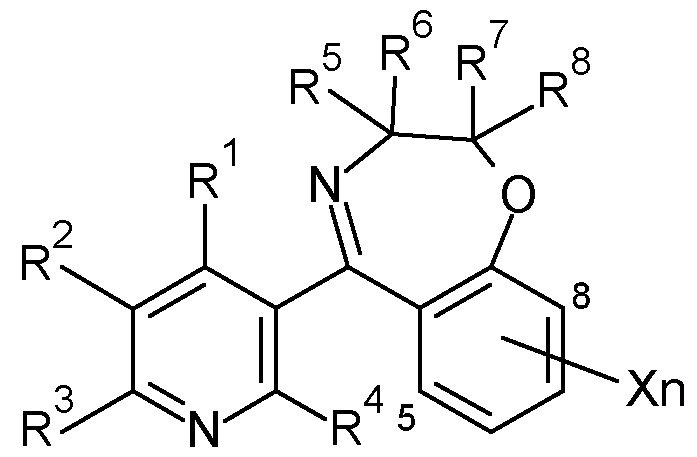
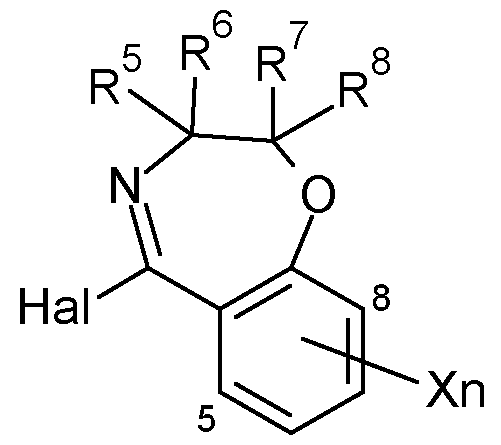
- R 1 is H
- R 2 is in each case independently selected from halogen, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, C 2 -C 6 -alkenyl, C 2 -C 6 -halogenalkenyl, C 2 -C 6 -alkynyl, 0-CrC 6 -alkyl, C3-C6-cycloalkyl;
- R 3 is in each case independently selected from CrC 6 -alkyl, CrC 6 -halogenalkyl, C 2 -C 6 - alkenyl, C 2 -C 6 -halogenalkenyl, C 2 -C 6 -alkynyl, 0-CrC 6 -alkyl, C3-C6-cycloalkyl;
- R 4 is H
- R 5 are in each case independently selected from H, F, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, C 2 -C 6 -alkenyl, phenyl, benzyl, wherein the moieties are unsubstituted or substituted by one to three groups R 5a , which independently of one another are selected from: halogen, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, 0-CrC 6 -alkyl;
- R 5 and R 6 form together with the C atoms to which they are bound a C3-C6-cycloalkyl or a a 3- to 6-membered saturated heterocycle which contains 1 , 2 or 3 heteroatoms from the group consisting of O and S; wherein the cycloalkyl or heterocycle can be unsubsituted or substitued by halogene, CrC 6 -alkyl, CrC 6 -halogenalkyl;
- R 7 are in each case independently selected from H, F, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, C 2 -C 6 -alkenyl, phenyl, benzyl, wherein the moieties are unsubstituted or substituted by one to three groups R 7a , which independently of one another are selected from: halogen, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, 0-CrC 6 -alkyl;
- R 8 are in each case independently selected from H, F, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, C 2 -C 6 -alkenyl, phenyl, benzyl, wherein the moieties are unsubstituted or substituted by one to three groups R 8a , which independently of one another are selected from: halogen, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, 0-CrC 6 -alkyl; or
- R 7 and R 8 form together with the C atoms to which they are bound a C3-C6-cycloalkyl or a a 3- to 6-membered saturated heterocycle which contains 1 , 2 or 3 heteroatoms from the group consisting of O and S;
- X is in each case independently selected from halogen, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, 0-CrC 6 -alkyl, 0-CrC 6 -halogenalkyl, C3-C6-cycloalkyl, C 2 -C 6 -alkenyl, C 2 -C 6 -alkynyl; n is 0, 1, 2 or 3, with the proviso that
- R 5 , R 6 , R 7 , R 8 can not be all H; and the N-oxides and the agriculturally acceptable salts thereof as fungicides.
- the N-oxides may be prepared from the inventive compounds according to conventional oxidation methods, e. g. by treating compounds I with an organic peracid such as metachloroperbenzoic acid (cf. WO 03/64572 or J. Med. Chem. 38(11), 1892-903, 1995); or with inorganic oxidizing agents such as hydrogen peroxide (cf. J. Heterocyc. Chem. 18(7), 1305-8, 1981) or oxone (cf. J. Am. Chem. Soc. 123(25), 5962-5973, 2001).
- the oxidation may lead to pure mono-N-oxides or to a mixture of different N-oxides, which can be separated by conventional methods such as chromatography.
- Agriculturally acceptable salts of the compounds of the formula I encompass especially the salts of those cations or the acid addition salts of those acids whose cations and anions, respectively, have no adverse effect on the fungicidal action of the compounds I.
- Suitable cations are thus in particular the ions of the alkali metals, preferably sodium and potassium, of the alkaline earth metals, preferably calcium, magnesium and barium, of the transition metals, preferably manganese, copper, zinc and iron, and also the ammonium ion which, if desired, may be substituted with one to four CrC 4 -alkyl substituents and/or one phenyl or benzyl substituent, preferably diisopropylammonium, tetramethylammonium, tetrabutylammonium, trimethylbenzylammonium, furthermore phosphonium ions, sulfonium ions, preferably tri(Ci-C 4 - alkyl)sulfonium,
- Anions of acceptable acid addition salts are primarily chloride, bromide, fluoride, hydrogensulfate, sulfate, dihydrogenphosphate, hydrogenphosphate, phosphate, nitrate, bicarbonate, carbonate, hexafluorosilicate, hexafluorophosphate, benzoate, and the anions of Ci-C 4 -alkanoic acids, preferably formate, acetate, propionate and butyrate. They can be formed by reacting a compound I with an acid of the corresponding anion, preferably of hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid or nitric acid.
- Stereoisomers of the formula I can exist as one or more stereoisomers.
- the various stereoisomers include enantiomers, diastereomers, atropisomers arising from restricted rotation about a single bond of asymmetric groups and geometric isomers. They also form part of the subject matter of the present invention.
- one stereoisomer may be more active and/or may exhibit beneficial effects when enriched relative to the other stereoisomer(s) or when separated from the other stereoisomer(s). Additionally, the skilled artisan knows how to separate, enrich, and/or to selectively prepare said stereoisomers.
- the compounds of the invention may be present as a mixture of stereoisomers, e.g. a racemate, individual stereoisomers, or as an optically active form.
- the embodiments of the intermediates obtained during preparation of compounds I correspond to the embodiments of the compounds of formula I.
- the term “compounds I” refers to compounds of the formula I.
- C n -C m indicates the number of carbon atoms possible in each case in the substituent or substituent moiety in question.
- halogen refers to fluorine, chlorine, bromine and iodine.
- CrC 6 -alkyl refers to a straight-chained or branched saturated hydrocarbon group having 1 to 6 carbon atoms, e.g. methyl, ethyl, propyl, 1-methylethyl, butyl, 1-methylpropyl, 2- methylpropyl, 1 , 1 -di methylethyl , pentyl, 1-methylbutyl, 2-methylbutyl, 3-methylbutyl,
- C2-C4-alkyl refers to a straight-chained or branched alkyl group having 2 to 4 carbon atoms, such as ethyl, propyl (n-propyl), 1-methylethyl (iso-propoyl), butyl, 1-methylpropyl (sec.-butyl), 2-methylpropyl (iso-butyl), 1 , 1 -dimethylethyl (tert.-butyl).
- C 1 -C 6 -halogenalkyl refers to an alkyl group having 1 or 6 carbon atoms as defined above, wherein some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as mentioned above.
- Ci-C2-halogenalkyl groups such as chloromethyl, bromomethyl, dichloromethyl, trichloromethyl, fluoromethyl, difluoromethyl, trifluoromethyl, chlorofluoromethyl, dichlorofluoromethyl, chlorodifluoromethyl, 1-chloroethyl, 1-bromoethyl, 1- fluoroethyl, 2-fluoroethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, 2-chloro-2-fluoroethyl, 2-chloro-
- CrC 6 -alkoxy refers to a straight-chain or branched alkyl group having 1 to 6 carbon atoms which is bonded via an oxygen, at any position in the alkyl group.
- Examples are "C 1 -C 4 - alkoxy” groups, such as methoxy, ethoxy, n-propoxy, 1-methylethoxy, butoxy, 1-methyhprop- oxy, 2-methylpropoxy or 1,1-dimethylethoxy.
- CrC 6 -halogenalkoxy refers to a CrC 6 -alkoxy radical as defined above, wherein some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as mentioned above.
- Examples are "CrC4-halogenalkoxy” groups, such as OCH 2 F, OCHF2, OCF 3 , OCH2CI, OCHCI2, OCCI 3 , chlorofluoromethoxy, dichlorofluoromethoxy, chlorodifluoromethoxy, 2-fluoroethoxy, 2-chlorothoxy, 2-bromoethoxy, 2-iodoethoxy, 2,2-difluoroethoxy, 2,2,2- trifluoroethoxy, 2-chloro-2-fluoroethoxy, 2-chloro-2,2-difluoroethoxy, 2,2-dichloro-2-fluoroethoxy,
- C 2 -C 6 -alkenyl refers to a straight-chain or branched unsaturated hydrocarbon radical having 2 to 6 carbon atoms and a double bond in any position.
- Examples are "C2-C4-alkenyl” groups, such as ethenyl, 1-propenyl, 2-propenyl (allyl), 1-methylethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1 -methyl-1 -propenyl, 2-methyl-1-propenyl, 1-methyl-2-propenyl, 2-methyl-2-propenyl.
- C 2 -C 6 -halogenalkenyl refers to an alkyl group having 2 or 6 carbon atoms as defined above, wherein some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as mentioned above.
- C 2 -C 6 -alkenyloxy refers to a straight-chain or branched alkenyl group having 2 to 6 carbon atoms which is bonded via an oxygen, at any position in the alkenyl group. Examples are “C2-C4-alkenyloxy” groups.
- C 2 -C 6 -alkynyl refers to a straight-chain or branched unsaturated hydrocarbon radical having 2 to 6 carbon atoms and containing at least one triple bond.
- Examples are "C2-C4- alkynyl” groups, such as ethynyl, prop-1-ynyl, prop-2-ynyl (propargyl), but-1-ynyl, but-2-ynyl, but-3-ynyl , 1 -methyl-prop-2-ynyl .
- C 2 -C 6 -halogenalkynyl refers to an alkyl group having 2 or 6 carbon atoms as defined above, wherein some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as mentioned above.
- C 2 -C 6 -alkynyloxy refers to a straight-chain or branched alkynyl group having 2 to 6 carbon atoms which is bonded via an oxygen, at any position in the alkynyl group. Examples are “C2-C4-alkynyloxy” groups.
- C3-C6-cycloalkyl refers to monocyclic saturated hydrocarbon radicals having 3 to 6 carbon ring members, such as cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl. Accordingly, a saturated three-, four-, five-, six-, seven-, eight-, nine or ten-membered carbocyclyl or carbo- cycle is a "C3-Cio-cycloalkyl".
- C3-C6-cycloalkenyl refers to a monocyclic partially unsaturated 3-, 4- 5- or 6- membered carbocycle having 3 to 6 carbon ring members and at least one double bond, such as cyclopentenyl, cyclopentadienyl, cyclohexadienyl. Accordingly, a partially unsaturated three-, four-, five-, six-, seven-, eight-, nine or ten-membered carbocyclyl or carbocycle is a "C3-C10- cycloalkenyl".
- C3-C8-cycloalkyl-Ci-C4-alkyl refers to alkyl having 1 to 4 carbon atoms (as defined above), whereAccording to one hydrogen atom of the alkyl radical is replaced by a cycloalkyl radical having 3 to 8 carbon atoms (as defined above).
- saturated or partially unsaturated three-, four-, five-, six-, seven-, eight-, nine or ten- membered heterocyclyl or heterocycle, wherein the heterocyclyl or heterocycle contains 1 , 2, 3 or 4 heteroatoms selected from N, O and S is to be understood as meaning both saturated and partially unsaturated heterocycles, wherein the ring member atoms of the heterocycle include besides carbon atoms 1 , 2, 3 or 4 heteroatoms independently selected from the group of O, N and S.
- a 3- or 4-membered saturated heterocycle which contains 1 or 2 heteroatoms from the group consisting of O, N and S as ring members such as oxirane, aziridine, thiirane, oxetane, azetidine, thiethane, [1,2]dioxetane, [1 ,2]dithietane, [1,2]diazetidine; and a 5- or 6-membered saturated or partially unsaturated heterocycle which contains 1, 2 or 3 heteroatoms from the group consisting of O, N and S as ring members such as 2- tetrahydrofuranyl, 3-tetrahydrofuranyl, 2-tetrahydrothienyl, 3-tetrahydrothienyl, 2-pyrrolidinyl, 3- pyrrolidinyl, 3-isoxazolidinyl, 4-isoxazolidinyl, 5-isoxazolidinyl, 3-isothiazolidinyl,
- substituted refers to substitued with 1 , 2, 3 or up to the maximum possible number of substituents.
- 5-or 6-membered heteroaryl or “5-or 6-membered heteroaromatic” refers to aromatic ring systems incuding besides carbon atoms, 1, 2, 3 or 4 heteroatoms independently selected from the group consisting of N, O and S, for example, a 5-membered heteroaryl such as pyrrol-1 -yl, pyrrol-2-yl, pyrrol-3-yl, thien-2-yl, thien-3-yl, furan- 2-yl, furan-3-yl, pyrazol-1-yl, pyrazol-3-yl, pyrazol-4-yl, pyrazol-5-yl, imidazol-1-yl, imidazol-2-yl, imidazol-4-yl, imidazol-5-yl, oxazol-2-yl, oxazol-4-yl, oxazol-5-yl, isoxazol-3-yl, isoxazol-4
- R 1 is H.
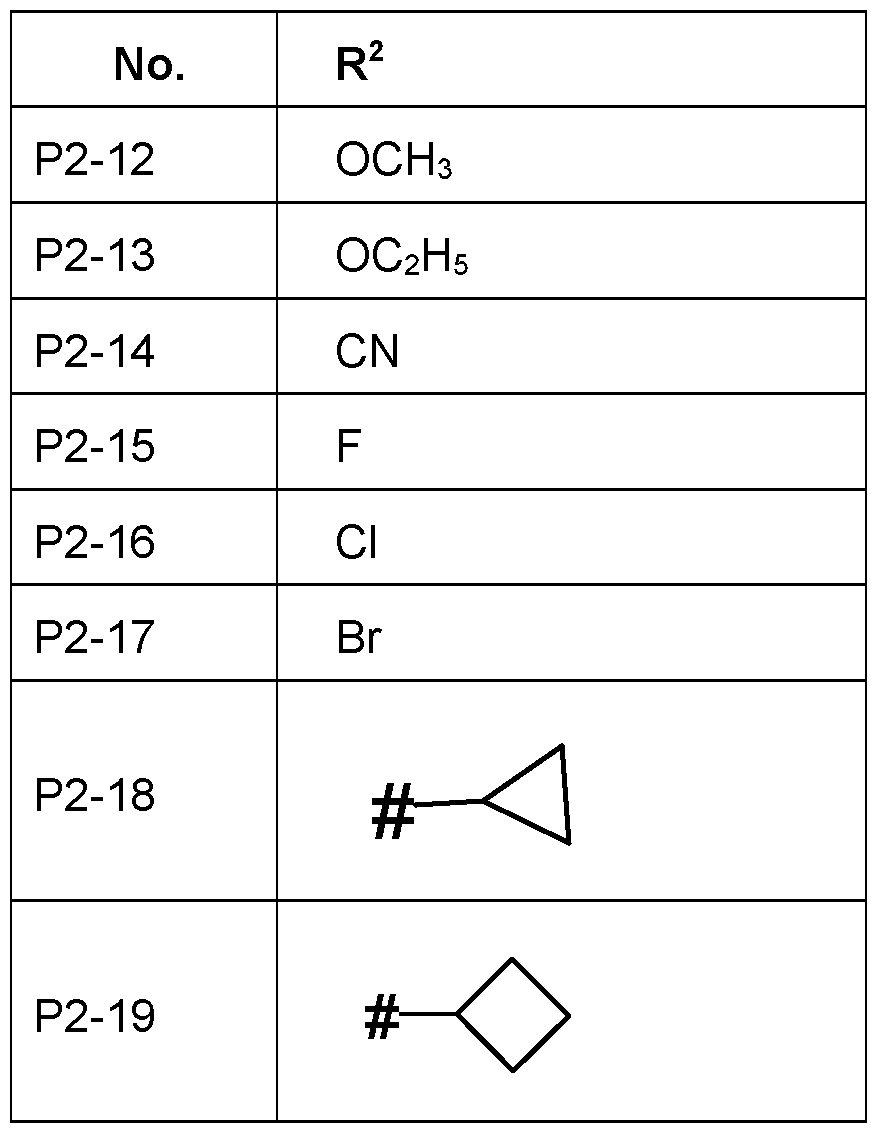
- R 2 is selected from halogen, CN, C 1 -C 6 -alkyl, C 1 -C 6 -halogenalkyl, C 2 -C 6 -alkenyl, C 2 -C 6 -halogenalkenyl, C 2 -C 6 -alkynyl, O-C 1 -C 6 - alkyl, C3-C6-cycloalkyl.
- R 2 is halogen, in particular F, Cl, Br or I, more specifically F, Cl or Br, in particular F or Cl.
- R 2 is F.
- R 2 is Cl
- R 2 is Br
- R 2 is CN
- R 2 is C 1 -C 6 -alkyl, in particular CrC4-alkyl, such as CH 3 or C 2 H 5 , in particular CH 3 or CH 2 CH 3 .
- R 2 is C 1 -C 6 -halogenalkyl, in particular C 1 -C 4 - halogenalkyl, such as CF 3 .
- R 2 is 0-C 1 -C 6 -alkyl, in particular C 1 -C 4 - alkyl, more specifically Ci-C 2 -alkoxy.
- R 2 is such as OCH 3 or OCH 2 CH 3 .
- R 2 is C3-C6-cycloalkyl, in particular cyclopropyl or cyclobutyl.
- R 2 Particularly preferred embodiments of R 2 according to the invention are in Table P2 below, wherein each line of lines P2-1 to P2-19 corresponds to one particular embodiment of the invention, wherein P2-1 to P2-19 are also in any combination with one another a preferred embodiment of the present invention.
- the connection point to the carbon atom, to which R 2 is bound is marked with “#” in the drawings.
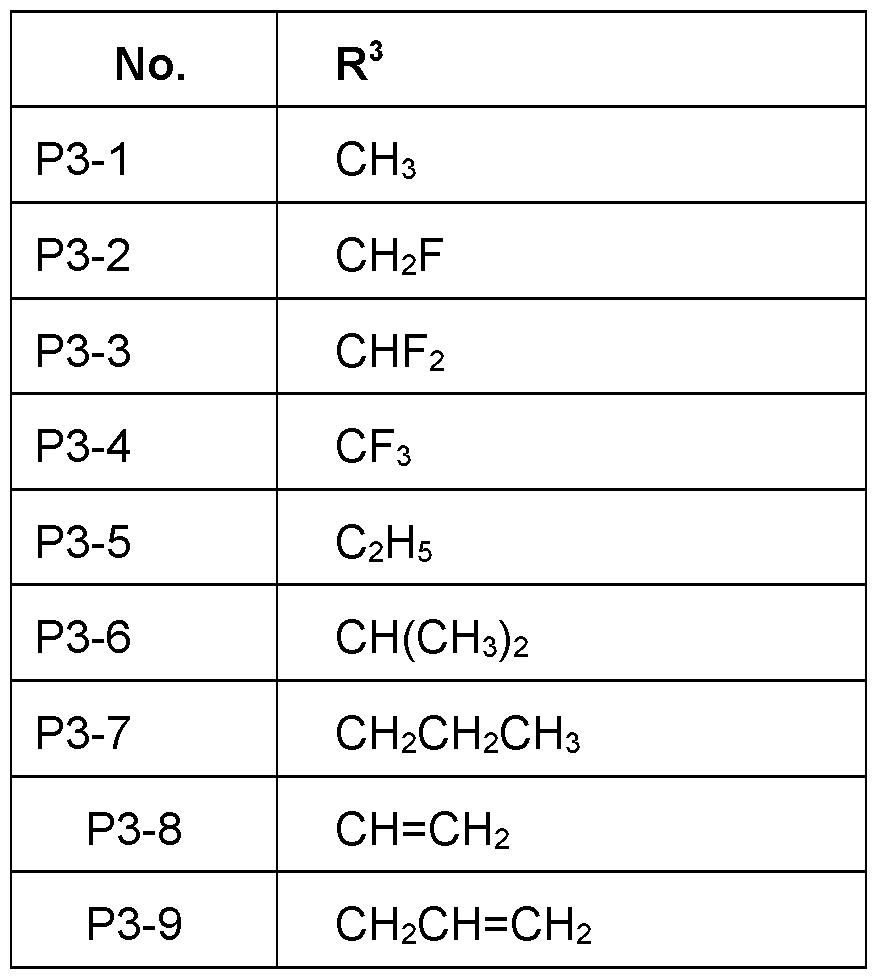
- R 3 is selected from the group consisting of C1-C 6 - alkyl, CrC 6 -halogenalkyl, in particular CH 3 , C 2 H 5 , CF 3 , CH 2 F, CHF2, more specifically CH 3 , CH 2 F, CF 2 H, CF 3 , cyclopropyl, cyclobutyl most preferred CH 3 , CF 3 , CF2H.
- R 3 is CrC 6 -alkyl, in particular C 1 C 4 -alkyl, such as CH 3 or C2H5, in particular CH 3 or CH2CH 3 .
- R 3 is CrC 6 -halogenalkyl, in particular C 1 -C 4 - halogenalkyl, such as CF 3 , FCH 2 , F 2 CH, CF 3 CH2.
- R 3 is 0-CrC 6 -alkyl, in particular C 1 -C 4 - alkyl, more specifically C 1 -C 2 -alkoxy.
- R 3 is such as OCH 3 or OCH 2 CH 3 .
- R 3 is C 3 -C 6 -cycloalkyl, in particular cyclopropyl, cyclobutyl.
- R 3 Particularly preferred embodiments of R 3 according to the invention are in Table P3 below, wherein each line of lines P3-1 to P3-15 corresponds to one particular embodiment of the invention, wherein P3-1 to P3-15 are also in any combination with one another a preferred embodiment of the present invention.
- the connection point to the carbon atom, to which R 3 is bound is marked with “#” in the drawings.
- R 4 is H.
- R 5 is in each case independently selected from H, F, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, C2- C 6 -alkenyl, phenyl, benzyl, wherein the are unsubstituted or substituted by one to three groups R 5a , which independently of one another are selected from: halogen, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, 0-C 1 -C 6 -alkyl.
- R 5 is in each case independently selected from CrC 6 -alkyl (embodiment 5.1), CrC 6 -halogenalkyl (embodiment 5.2), H (embodiment 5.3), phenyl, CH 2 -phenyl (embodiment 5.4), wherein phenyl and CH 2 -phenyl is unsubstituted or substituted by one or two halogen.
- R 5 is CH 3 or CF 3 .
- R 5 is CH 3 .
- R 5 is H.
- R 5 is H, CH 2 CH 3 ,
- R 5 is phenyl, 2-F-phenyl, 4- F-phenyl, 2,4-F 2 -phenyl, 2-CI-phenyl, 4-CI-phenyl, CH 2 -phenyl, CH 2 -2-F-phenyl, CH 2 -4-F- phenyl.
- R 6 is in each case independently selected from are in each case independently selected from H, F, CN, CrC 6 -alkyl, C 1 -C 6 - halogenalkyl, C 2 -C 6 -alkenyl, phenyl, benzyl, wherein the moieties are unsubstituted or substituted by one to three groups R 6a , which independently of one another are selected from: halogen, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, 0-C 1 -C 6 -alkyl.
- R 6 is in each case independently selected from CrC 6 -alkyl (embodiment 6.1), H (embodiment 6.2).
- R 6 is H, CH 3 , CH 2 CH 3 , CH(CH 3 )2, CH(CH 3 )CH 2 CH 3 , C(CH 3 )3, CH 2 -CH(CH 3 )2, CH 2 -C(CH 3 )3, CH 2 -CH(CH 3 )-C(CH 3 )3, CH 2 -CH 2 -C(CH 3 )3.
- R 5 and R 6 form C 3 -C 6 - cycloalkyl (embodiment 6.4).
- R 5 and R 6 form 3- to 6- membered saturated heterocycle which contains 1, 2 or 3 heteroatoms from the group consisting of O and S.
- R 5 and R 6 form 3- to 6- membered saturated heterocycle which contains one O (embodiment 6.5).
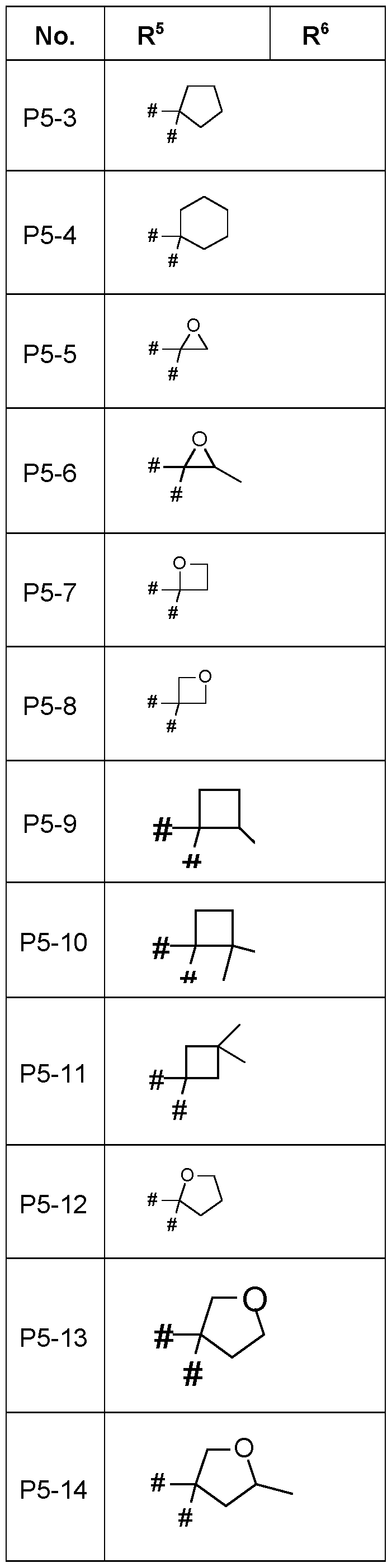
- R 5 , R 6 are in Table P5 below, wherein each line of lines P5-1 to P5-19 corresponds to one particular embodiment of the invention, wherein P5-1 to P5-19 are also in any combination with one another a preferred embodiment of the present invention.
- the connection point to the carbon atom, to which R 5 and R 6 is bound is marked with “#” in the drawings.
- R 7 is in each case independently selected from H, F, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, C2- C 6 -alkenyl, C 1 -C 6 -alkyl-0-CrC 6 -alkyl, phenyl, benzyl, wherein the moieties are unsubstituted or substituted by one to three groups R 7a , which independently of one another are selected from: halogen, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, 0-C 1 -C 6 -alkyl.
- R 7 is in each case independently selected from CrC 6 -alkyl (embodiment 7.1), CrC 6 -halogenalkyl (embodiment 7.2), CrC 6 -alkyl- 0-CrC 6 -alkyl (embodiment 7.3), phenyl, CH 2 -phenyl (embodiment 7.4), wherein phenyl and CH 2 -phenyl is unsubstituted or substituted by one or two halogen.
- R 7 is CH 3 or CF 3 .
- R 7 is CH 3 . According to one further embodiment of the compound of formula I, R 7 is H.
- R 7 is CH 2 CH 3 , CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , C(CH 3 )3, CH 2 -CH(CH 3 )2, CH 2 -C(CH 3 )3, CH2-O-CH3.
- R 7 is phenyl, 2-F-phenyl, 4- F-phenyl, 2,4-F 2 -phenyl, 2-CI-phenyl, 4-CI-phenyl, CH 2 -phenyl, CH 2 -2-F-phenyl, CH 2 -4-F- phenyl.
- R 8 is in each case independently selected from are in each case independently selected from H, F, CN, CrC 6 -alkyl, C 1 -C 6 - halogenalkyl, C 2 -C 6 -alkenyl, C 1 -C 6 -alkyl-0-CrC 6 -alkyl, phenyl, benzyl, CrC 6 -alkyl-0-phenyl, wherein the moieties are unsubstituted or substituted by one to three groups R 6a , which independently of one another are selected from: halogen, CN, CrC 6 -alkyl, CrC 6 -halogenalkyl, 0-C 1 -C 6 -alkyl.
- R 8 is in each case independently selected from CrC 6 -alkyl (embodiment 8.1), CrC 6 -alkyl-0-phenyl (embodiment 8.2), C 1 -C 6 - alkyl-0-C 1 -C 6 -alkyl (embodiment 8.3).
- R 8 is CH 2 CH 3 , CH(CH 3 ) 2 , CH(CH 3 )CH 2 CH 3 , C(CH 3 )3, CH 2 -CH(CH 3 )2, CH 2 -C(CH 3 )3, CH 2 -CH(CH 3 )-C(CH 3 )3, CH2-CH2- C(CH3) 3 , CH2-O-CH3, CH 2 -0-(CH 3 )3, CH 2 -0-phenyl.
- R 7 and R 8 form together with the C atoms to which they are bound a C3-C6-cycloalkyl or a a 3- to 6-membered saturated heterocycle which contains 1 , 2 or 3 heteroatoms from the group consisting of O and S.
- R 7 and R 8 form C3-C6- cycloalkyl (embodiment 8.4).
- R 7 and R 8 form 3- to 6- membered saturated heterocycle which contains 1 , 2 or 3 heteroatoms from the group consisting of O and S.
- R 7 and R 8 form 3- to 6- membered saturated heterocycle which contains one O (embodiment 8.5).
- R 7 and R 8 are in Table P5 below, wherein each line of lines P5-1 to P5-18 corresponds to one particular embodiment of the invention, wherein P5-1 to P5-18 are also in any combination with one another a preferred embodiment of the present invention.
- the connection point to the carbon atom, to which R 5 and R 6 is bound is marked with “#” in the drawings.
- X is in each case independently selected from halogen (embodiment X.1), CN, CrC 6 -alkyl (embodiment X.2), C 1 -C 6 - halogenalkyl (embodiment X.3), 0-CrC 6 -alkyl (embodiment X.4), 0-CrC 6 -halogenalkyl (embodiment X.5).
- X is in each case independently selected from halogen, 0-C 1 -C 6 -alkyl.
- X is in each case independently selected from F or Cl.
- Xn is as defined below: and X is F.
- Xn is as defined below: and X is F.
- n 0.
- n 1
- n is 2.
- the compounds of the formula I comprise one or more chiral center and are generally obtained in the form of a racemate.
- the R- and S-enantiomers can be separated and isolated in pure form with methods known by the skilled person, e.g. by using chiral HPLC.
- the compound of the formula I can be used in form of
- the compound of the formula I is present as racemic composition of the (R)-enantiomer and (S)-enantiomer, but the (R)- enantiomer and the (S)-enantiomer may also be present in any other proportion, for example the pure enantiomer (R) or the pure enantiomer (S) of the compound of the formula I.
- the compound of the formula I is provided and used as (R)-enantiomer with an enantiomeric excess (e.e.) of at least 40%, for example, at least 50%, 60%, 70% or 80%, preferably at least 90%, more preferably at least 95%, yet more preferably at least 98% and most preferably at least 99%.
- an enantiomeric excess e.e.
- the compound of the formula I is provided and used as (S)-enantiomer with an enantiomeric excess (e.e.) of at least 40%, for example, at least 50%, 60%, 70% or 80%, preferably at least 90%, more preferably at least 95%, yet more preferably at least 98% and most preferably at least 99%.
- an enantiomeric excess e.e.
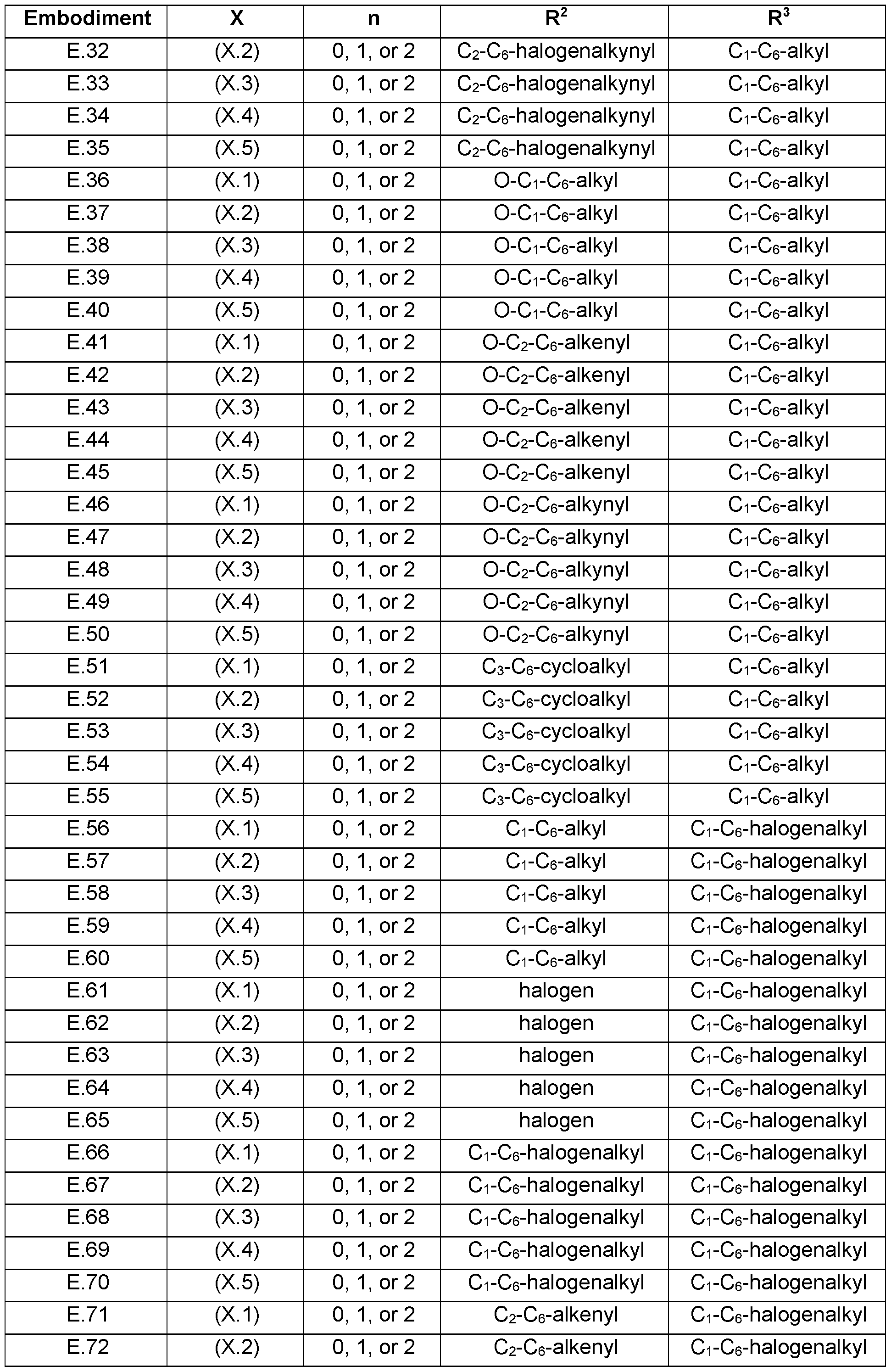
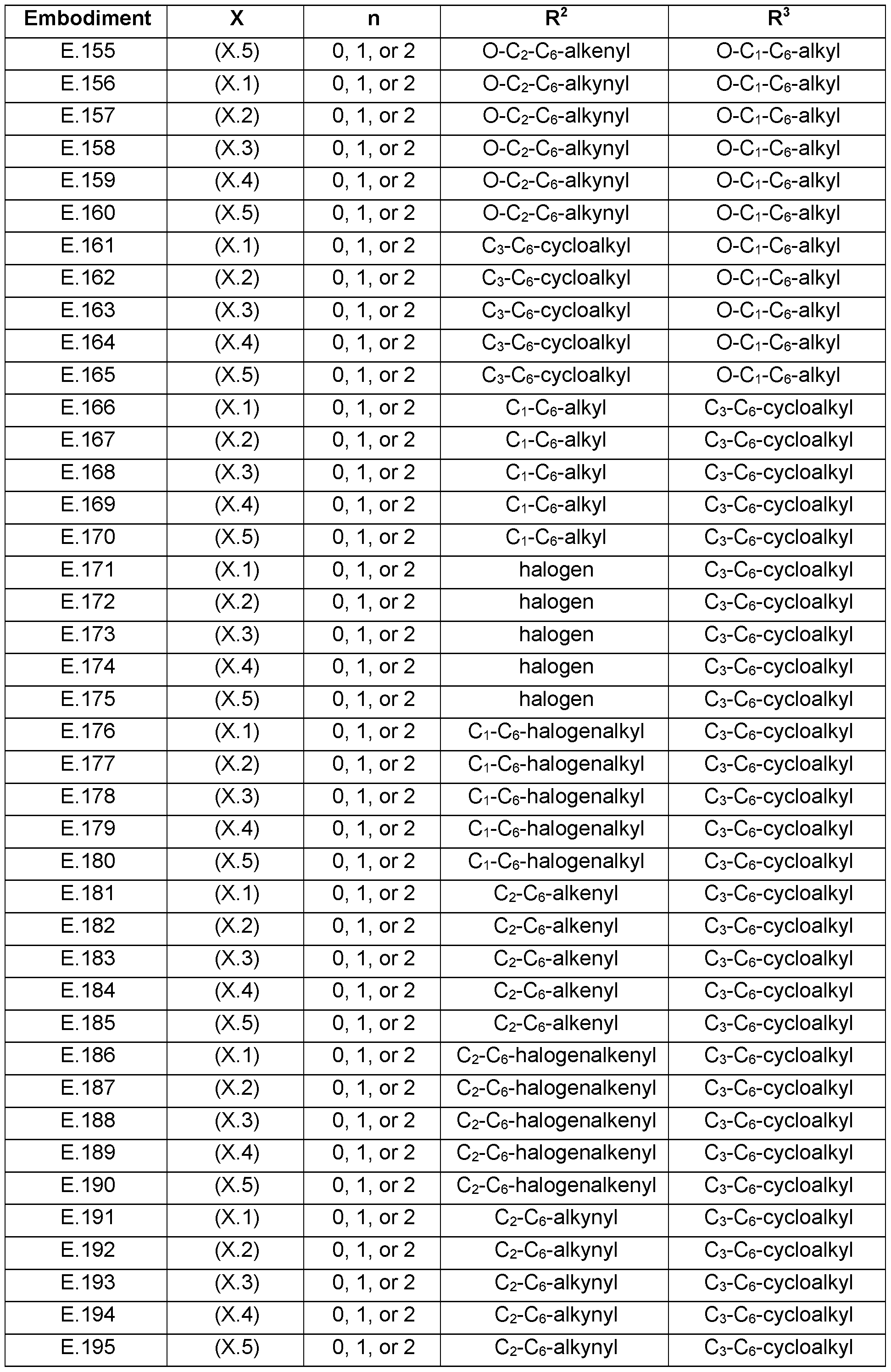
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, which represent preferred combinations of embodiments that are defined above for each of the variables R 2 , R 3 and X (represented by embodiments X.1 to X.6), n in compounds of formula s defined below.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.1 and R 6 is represented by embodiment 6.1. In further aspects the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.2 and R 6 is represented by embodiment 6.1.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.3 and R 6 is represented by embodiment 6.1.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.4 and R 6 is represented by embodiment 6.1.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.1 and R 6 is represented by embodiment 6.2.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.2 and R 6 is represented by embodiment 6.2.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.3 and R 6 is represented by embodiment 6.2.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.4 and R 6 is represented by embodiment 6.2.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.1 and R 6 is represented by embodiment 6.3.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.2 and R 6 is represented by embodiment 6.3.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.3 and R 6 is represented by embodiment 6.3.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 is represented by embodiment 5.4 and R 6 is represented by embodiment 6.3.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 and R 6 arerepresented by embodiment 6.4.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 5 and R 6 arerepresented by embodiment 6.5.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 7 is represented by embodiment 7.1 and R 8 is represented by embodiment 8.1.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 7 is represented by embodiment 7.2 and R 8 is represented by embodiment 8.1.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 7 is represented by embodiment 7.3 and R 8 is represented by embodiment 8.1.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 7 is represented by embodiment 7.4 and R 8 is represented by embodiment 8.1.
- the present invention relates to the embodiments E.1 to E.275 listed in Table E, wherein R 7 is represented by embodiment 7.1 and R 8 is represented by embodiment 8.2.
- the present invention relates to the embodiments E.1 to E.277 listed in Table E, wherein R 7 is represented by embodiment 7.2 and R 8 is represented by embodiment 8.2.
- the present invention relates to the embodiments E.1 to E.277 listed in Table E, wherein R 7 is represented by embodiment 7.3 and R 8 is represented by embodiment 8.2.
- the present invention relates to the embodiments E.1 to E.277 listed in Table E, wherein R 7 is represented by embodiment 7.4 and R 8 is represented by embodiment 8.2.
- the present invention relates to the embodiments E.1 to E.277 listed in Table E, wherein R 7 is represented by embodiment 7.1 and R 8 is represented by embodiment 8.3.
- the present invention relates to the embodiments E.1 to E.277 listed in Table E, wherein R 7 is represented by embodiment 7.2 and R 8 is represented by embodiment 8.3.
- the present invention relates to the embodiments E.1 to E.277 listed in Table E, wherein R 7 is represented by embodiment 7.3 and R 8 is represented by embodiment 8.3.
- the present invention relates to the embodiments E.1 to E.277 listed in Table E, wherein R 7 is represented by embodiment 7.4 and R 8 is represented by embodiment 8.3.
- the present invention relates to the embodiments E.1 to E.277 listed in Table E, wherein R 7 and R 8 arerepresented by embodiment 8.4.
- the present invention relates to the embodiments E.1 to E.277 listed in Table E, wherein R 7 and R 8 arerepresented by embodiment 8.7.
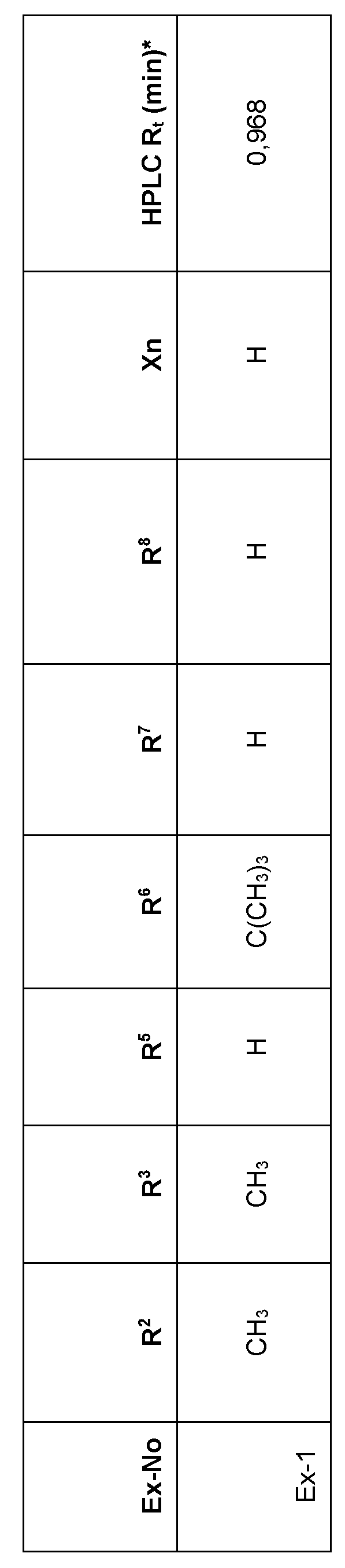
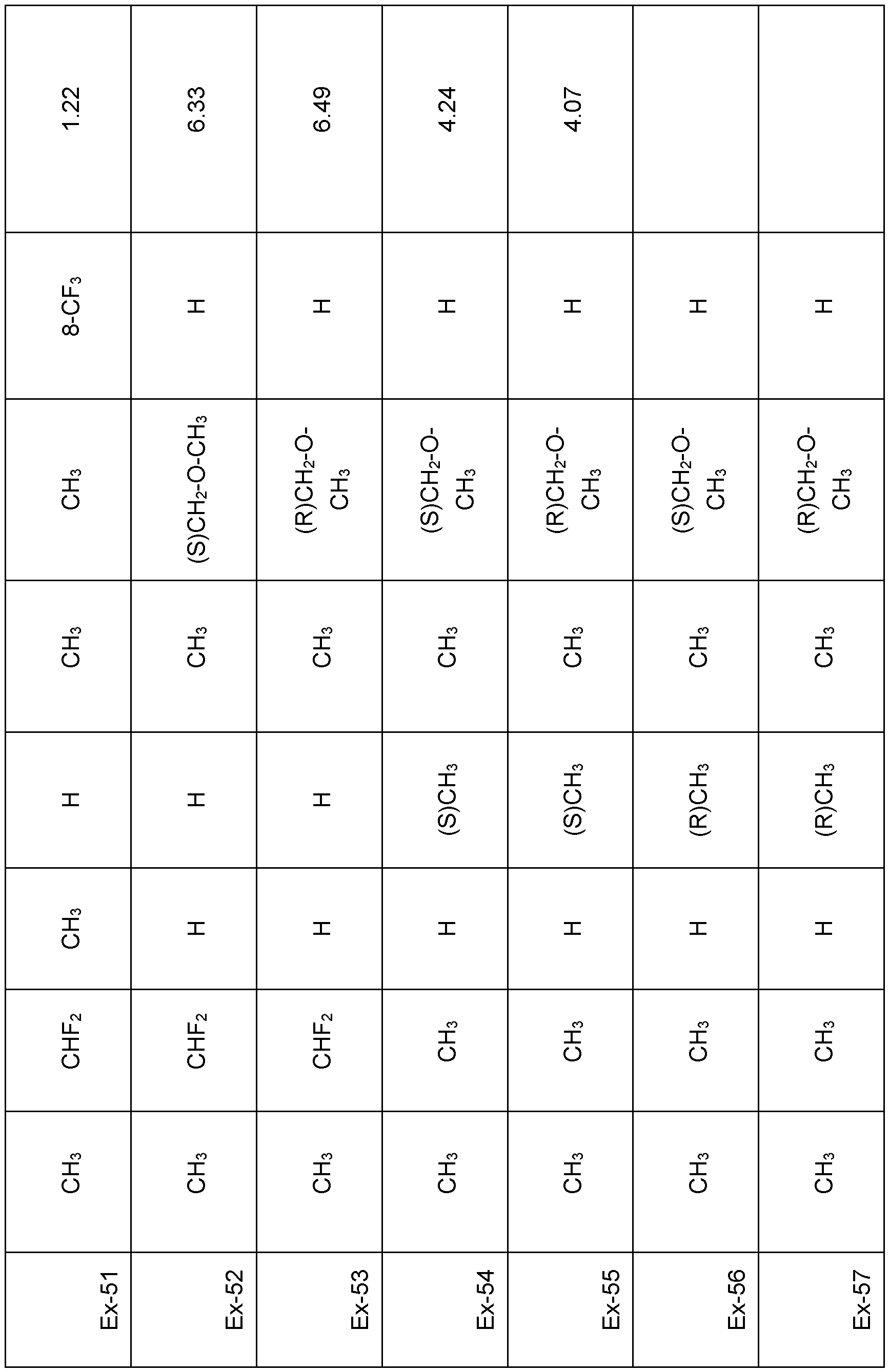
- Preferred embodiments of the present invention are the following compounds I.A-1 , I.A-2, I.A-3, I.A-4.
- the substituents R 5 , R 6 , R 7 and R 8 and Xn are independently as defined above or preferably defined herein:
- Table 1a Compounds of the formula I.A-1, I.A-2, I.A-3, I.A-4 in which Xn is H and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I.A-1.1a.B-1 to I.A-1.1a.B-25, I.A-2.1a.B-1 to I.A-2.1a.B-25, I.A- 3.1a.B-1 to I.A-3.1a.B-25, I.A-4.1a.B-1 to I.A-4.1a.B-25).
- Table 2a Compounds of the formula I.A-1, I.A-2, I.A-3, I.A-4; in which Xn is 8-F and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I.A-1.2a. B-1 to I.A-1.2a. B-25, I.A-2.2a.B-1 to I.A- 2.2a. B-25, I.A-3.2a.B-1 to I.A-3.2a.B-25, I.A-4.2a.B-1 to I.A-4.2a.B-25).
- Table 3a Compounds of the formula I.A-1, I.A-2, I.A-3, I.A-4; in which Xn is 8-CI and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I.A-1.3a. B-1 to I.A-1.3a. B-25, I.A-2.3a. B-1 to I.A- 2.3a. B-25, I.A-3.3a.B-1 to I.A-3.3a.B-25, I.A-4.3a.B-1 to I.A-4.3a.B-25)
- Table 4a Compounds of the formula I.A-1, I.A-2, I.A-3, I.A-4; in which Xn is 7,8-F 2 and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I.A-1.4a. B-1 to I.A-1.4a. B-25, I.A-2.4a. B-1 to I.A- 2.4a. B-25, I.A-3.4a.B-1 to I.A-3.4a.B-25, I.A-4.4a.B-1 to I.A-4.4a.B-25).
- Table 5a Compounds of the formula I.A-1, I.A-2, I.A-3, I.A-4; in which Xn is 5,8-F 2 and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I.A-1.5a. B-1 to I.A-1.5a. B-25, I.A-2.5a. B-1 to I.A- 2.5a. B-25, I.A-3.5a.B-1 to I.A-3.5a.B-25, I.A-4.5a.B-1 to I.A-4.5a.B-25)
- Table 6a Compounds of the formula I.A-1, I.A-2, I.A-3, I.A-4; in which Xn is 7-OCH 3 and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I.A-1.6a. B-1 to I.A-1.6a. B-25, I.A-2.6a. B-1 to I.A- 2.6a. B-25, I.A-3.6a.B-1 to I.A-3.6a.B-25, I.A-4.6a.B-1 to I.A-4.6a.B-25
- Table 7a Compounds of the formula I.A-1, I.A-2, I.A-3, I.A-4; in which Xn is 6,8-F 2 and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I.A-1.7a. B-1 to I.A-1.7a. B-25, I.A-2.7a. B-1 to I.A- 2.7a. B-25, I.A-3.7a.B-1 to I.A-3.7a.B-25, I.A-4.7a.B-1 to I.A-4.7a.B-25)
- Preferred embodiments of the present invention are the following compounds I.B-1 , I.B-2, I.B-3, I.B-4.
- the substituents R 5 , R 6 , R 7 and R 8 and Xn are independently as defined above or preferably defined herein:
- Table 1b Compounds of the formula I.B-1 , I.B-2, I.B-3, I.B-4 in which Xn is H and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I.B-1.1b.B-1 to I. B-1.1 b. B-25, I.B-2.1b.B-1 to I.B-2.1b.B-25, I.B- 3.1b.B-1 to I.B-3.1b. B-25, I.B-4.1b.B-1 to I.B-4.1b.B-25).
- Table 2b Compounds of the formula I.B-1 , I.B-2, I.B-3, I.B-4; in which Xn is 8-F and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I. B-1.2b. B-1 to I. B-1.2b. B-25, I.B-2.2b. B-1 to I.B- 2.2b. B-25, I.B-3.2b. B-1 to I.B-3.2b.B-25, I.B-4.2b.B-1 to I.B-4.2b.B-25).
- Table 3b Compounds of the formula I.B-1 , I.B-2, I.B-3, I.B-4; in which Xn is 8-CI and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I. B-1.3b. B-1 to I. B-1.3b. B-25, I.B-2.3b. B-1 to I.B- 2.3b. B-25, I.B-3.3b. B-1 to I.B-3.3b.B-25, I.B-4.3b.B-1 to I.B-4.3b.B-25)
- Table 4b Compounds of the formula I.B-1 , I.B-2, I.B-3, I.B-4; in which Xn is 7,8-F 2 and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I. B-1.4b. B-1 to I. B-1.4b. B-25, I.B-2.4b. B-1 to I.B- 2.4b. B-25, I.B-3.4b. B-1 to I.B-3.4b.B-25, I.B-4.4b.B-1 to I.B-4.4b.B-25).
- Table 5b Compounds of the formula I.B-1 , I.B-2, I.B-3, I.B-4; in which Xn is 5,8-F 2 and the meaning for the combination of R 5 and R 6 for each individual compound corresponds in each case to one line of Table B (compounds I. B-1.5b. B-1 to I. B-1.5b. B-25, I.B-2.5b. B-1 to I.B- 2.5b. B-25, I.B-3.5b.
- compounds I can be prepared by a by palladium catalyzed Suzuki coupling reaction between a boronic acid derivative represented by formula (2) and an imidoyl halide derivative represented by formula (6) using a palladium complex in an organic solvent. It is preferred to conduct the reaction at elevated temperature, preferably between 60 and 160 °C, and using 1-3 equivalents of boronic acid derivative represented by formula 3 per 1 imidoyl halide derivative (6), as described in W02009119089A1.
- An imidoyl compound represented by formula (6) can be prepared by a method in which a cyclic amide represented by formula (7) is reacting in the presence of an suitable halogenating agent such as triphenylphosphine and a carbon tetrahalide, triphenylphosphine dichloride, phosgene, oxalyl chloride or thionyl chloride as described in US 2011/0136782 A1.
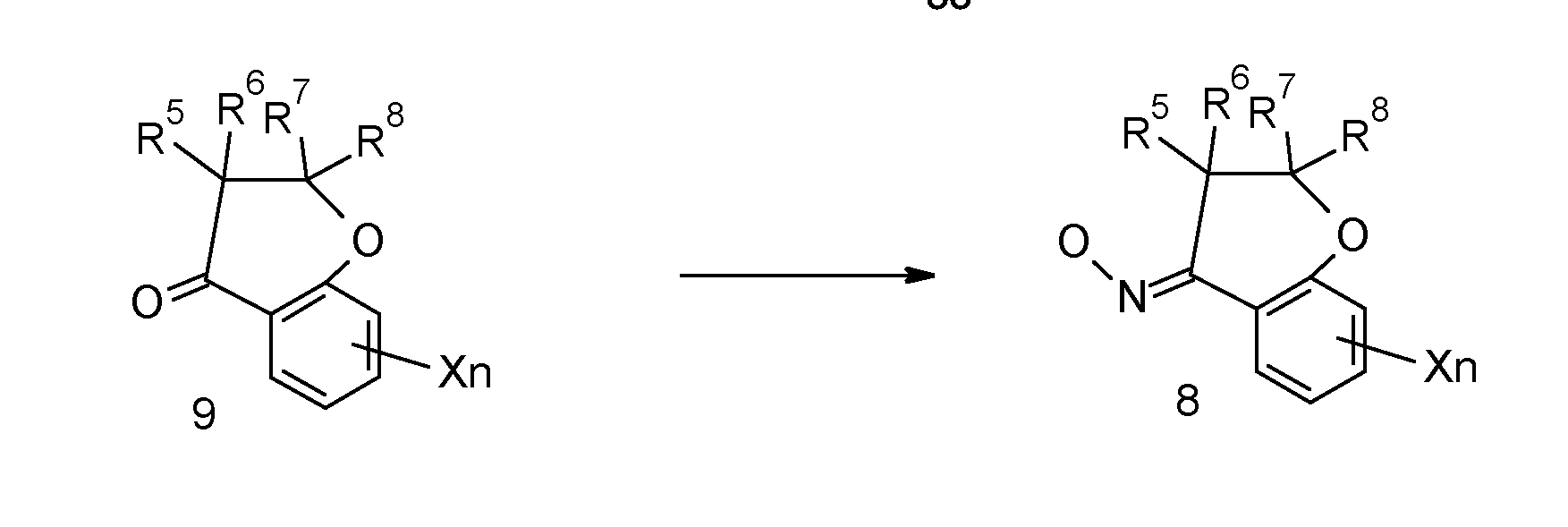
- a compound represented by formula (7) can be prepared by deriving a cyclic acetophenone derivative represented by formula (9) to an oxime by a Schmidt reaction, and then carrying out a Beckmann rearrangement. Various variations have been reported for both reactions.
- the Schmidt reaction can be carried out by, for example, reacting a ketone in sodium azide and a strong acid, such as concentrated hydrochloric acid, sulfuric acid, trifluoroacetic acid or methane sulfonic acid, and in the absence of a solvent or in a solvent such as acetonitrile, chloroform or methylene chloride.
- a strong acid such as concentrated hydrochloric acid, sulfuric acid, trifluoroacetic acid or methane sulfonic acid
- an oxime of a carbonyl compound is reacted with polyphosphoric acid or a trimethylsilyl ester thereof, or reacting at a high temperature with a Lewis acid such as aluminum triiodide or iron (III) chloride-impregnated montmorillonite or with thionyl chloride in the absence of solvent or in the presence of a solvent such as acetonitrile.
- a Lewis acid such as aluminum triiodide or iron (III) chloride-impregnated montmorillonite or with thionyl chloride in the absence of solvent or in the presence of a solvent such as acetonitrile.
- it can also be prepared by forming a mesylate or tosylate of an oxime followed by treating with a base such as aqueous sodium hydroxide solution or treating with a Lewis acid such as diethyl aluminum chloride, as described in Heterocycles (1994), 38(2), 305-18; US 2011/0136782 A1.
- a base such as aqueous sodium hydroxide solution
- a Lewis acid such as diethyl aluminum chloride
- the compound represented by formula (7) can be prepared in an one pot by copper(ll)-catalyzed Beckmann rearrangement of ketones (9) under mild reaction conditions using hydroxylamine-O-sulfonic acid as aminating agent, as described in Synthesis 2019, 51(19), 3709-3714.
- the oxime (8) can be prepared using a known method by reacting with hydroxylamine hydrochloride in a solvent such as ethanol followed by adding a base such as pyridine or sodium acetate or aqueous sodium hydroxide solution as necessary at a temperature up to the boiling point of the solvent, as described in Bioorganic & Medicinal Chemistry (2008), 16(11), 6124-6130; Heterocyclic Communications (1998), 4(6), 547-557.
- cyclic acetophenone derivatives represented by formula (9) are commercially available or can be accessed starting from 2-hydroxyacetophenone via a classical ring closure reaction using the corresponding ketone in the presence of pyrrolidine, as described in Bioorganic & Medicinal Chemistry (2008), 16(11), 6124-6130; Journal of the Chemical Society, Perkin Transactions 1 : Organic and Bio-Organic Chemistry (1995).
- the pending invention relates further to a process for preparing compounds of formula I, comprising a reaction of the compounf of the formula Y: wherein R 5 , R 6 , R 7 , R 8 and Xn are as defined above.
- the pending invention relates further to compounds of the formula Y wherein
- R 5 is selected from the group consisting of H, C 1 -C 6 -alkyl
- R 6 is selected from the group consisting of H, C 1 -C 6 -alkyl, or R 5 and R 6 form together with the C atoms to which they are bound a C3-C6-cycloalkyl;
- R 7 is selected from the group consisting of H, C 1 -C 6 -alkyl,
- R 8 is selected from the group consisting of H, C 1 -C 6 -alkyl, or
- R 7 and R 8 form together with the C atoms to which they are bound a C 3 -C 6 -cycloalkyl
- X is halogene, C 1 -C 6 -alkyl, n is 0, 1 or 2.
- R 5 is selected from the group consisting of H, CH 3,
- R 6 is selected from the group consisting of H, CH3, or R 5 and R 6 form together with the C atoms to which they are bound a cyclopropyl;
- R 7 is selected from the group consisting of H, CH 3 ,
- R 8 is selected from the group consisting of H, CH 3 , or
- R 7 and R 8 form together with the C atoms to which they are bound a cyclopropyl
- X is Cl, F or CH 3 , n is 0, 1 or 2.
- the compounds I and the compositions thereof, respectively, are suitable as fungicides effective against a broad spectrum of phytopathogenic fungi, including soil-borne fungi, in particular from the classes of Plasmodiophoromycetes, Peronosporomycetes (syn. Oomycetes), Chytridiomycetes, Zygomycetes, Ascomycetes, Basidiomycetes, and Deuteromycetes (syn. Fungi imperfecti). They can be used in crop protection as foliar fungicides, fungicides for seed dressing, and soil fungicides.
- the compounds I and the compositions thereof are preferably useful in the control of phytopathogenic fungi on various cultivated plants, such as cereals, e. g. wheat, rye, barley, triticale, oats, or rice; beet, e. g. sugar beet or fodder beet; fruits, e. g. pomes (apples, pears, etc.), stone fruits (e.g. plums, peaches, almonds, cherries), or soft fruits, also called berries (strawberries, raspberries, blackberries, gooseberries, etc.); leguminous plants, e. g. lentils, peas, alfalfa, or soybeans; oil plants, e. g.
- cereals e. g. wheat, rye, barley, triticale, oats, or rice
- beet e. g. sugar beet or fodder beet
- fruits e. g. pomes (apples, pears,
- oilseed rape mustard, olives, sunflowers, coconut, cocoa beans, castor oil plants, oil palms, ground nuts, or soybeans; cucurbits, e. g. squashes, cucumber, or melons; fiber plants, e. g. cotton, flax, hemp, or jute; citrus fruits, e. g. oranges, lemons, grapefruits, or mandarins; vegetables, e. g. spinach, lettuce, asparagus, cabbages, carrots, onions, tomatoes, potatoes, cucurbits, or paprika; lauraceous plants, e. g. avocados, cinnamon, or camphor; energy and raw material plants, e. g.
- corn, soybean, oilseed rape, sugar cane, or oil palm corn; tobacco; nuts; coffee; tea; bananas; vines (table grapes and grape juice grape vines); hop; turf; sweet leaf (also called Stevia); natural rubber plants; or ornamental and forestry plants, e. g. flowers, shrubs, broad-leaved trees, or evergreens (conifers, eucalypts, etc.); on the plant propagation material, such as seeds; and on the crop material of these plants.
- compounds I and compositions thereof, respectively are used for controlling fungi on field crops, such as potatoes, sugar beets, tobacco, wheat, rye, barley, oats, rice, corn, cotton, soybeans, oilseed rape, legumes, sunflowers, coffee or sugar cane; fruits; vines; ornamentals; or vegetables, such as cucumbers, tomatoes, beans or squashes.
- field crops such as potatoes, sugar beets, tobacco, wheat, rye, barley, oats, rice, corn, cotton, soybeans, oilseed rape, legumes, sunflowers, coffee or sugar cane; fruits; vines; ornamentals; or vegetables, such as cucumbers, tomatoes, beans or squashes.
- plant propagation material is to be understood to denote all the generative parts of the plant, such as seeds; and vegetative plant materials, such as cuttings and tubers (e. g. potatoes), which can be used for the multiplication of the plant. This includes seeds, roots, fruits, tubers, bulbs, rhizomes, shoots, sprouts and other parts of plants; including seedlings and young plants to be transplanted after germination or after emergence from soil.
- treatment of plant propagation materials with compounds I and compositions thereof, respectively, is used for controlling fungi on cereals, such as wheat, rye, barley and oats; rice, corn, cotton and soybeans.
- all of the above cultivated plants are understood to comprise all species, subspecies, variants, varieties and/or hybrids which belong to the respective cultivated plants, including but not limited to winter and spring varieties, in particular in cereals such as wheat and barley, as well as oilseed rape, e.g. winter wheat, spring wheat, winter barley etc.
- Corn is also known as Indian corn or maize (Zea mays) which comprises all kinds of corn such as field corn and sweet corn.
- all maize or corn subspecies and/or varieties are comprised, in particular flour corn (Zea mays var. amylacea), popcorn (Zea mays var. everta), dent corn (Zea mays var. indentata), flint corn (Zea mays var. indurata), sweet corn (Zea mays var. saccharata and var. rugosa ), waxy corn (Zea mays var. ceratina), amylomaize (high amylose Zea mays varieties), pod corn or wild maize (Zea mays var. tunicata) and striped maize (Zea mays var. japonica ).
- soybean cultivars are classifiable into indeterminate and determinate growth habit, whereas Glycine soja, the wild progenitor of soybean, is indeterminate (PNAS 2010, 107 (19) 8563-856).
- the indeterminate growth habit (Maturity Group, MG 00 to MG 4.9) is characterized by a continuation of vegetative growth after flowering begins whereas determinate soybean varieties (MG 5 to MG 8) characteristically have finished most of their vegetative growth when flowering begins.
- all soybean cultivars or varieties are comprised, in particular indeterminate and determinate cultivars or varieties.
- cultivagenesis includes random mutagenesis using X-rays or mutagenic chemicals, but also targeted mutagenesis to create mutations at a specific locus of a plant genome.
- Targeted mutagenesis frequently uses oligonucleotides or proteins like CRISPR/Cas, zinc-finger nucleases, TALENs or meganucleases.
- Genetic engineering usually uses recom binant DNA techniques to create modifications in a plant genome which under natural circum stances cannot readily be obtained by cross breeding, mutagenesis or natural recombination.
- one or more genes are integrated into the genome of a plant to add a trait or improve or modify a trait. These integrated genes are also referred to as transgenes, while plant com prising such transgenes are referred to as transgenic plants.
- the process of plant transforma tion usually produces several transformation events, wich differ in the genomic locus in which a transgene has been integrated. Plants comprising a specific transgene on a specific genomic locus are usually described as comprising a specific “event”, which is referred to by a specific event name. Traits which have been introduced in plants or have been modified include herbici de tolerance, insect resistance, increased yield and tolerance to abiotic conditions, like drought.
- Herbicide tolerance has been created by using mutagenesis and genetic engineering. Plants which have been rendered tolerant to acetolactate synthase (ALS) inhibitor herbicides by mutagenesis and breeding are e.g. available under the name Clearfield®. Herbicide tolerance to glyphosate, glufosinate, 2,4-D, dicamba, oxynil herbicides, like bromoxynil and ioxynil, sulfonylurea herbicides, ALS inhibitors and 4-hydroxyphenylpyruvate dioxygenase (HPPD) inhibitors, like isoxaflutole and mesotrione, has been created via the use of transgenes.
- HPPD 4-hydroxyphenylpyruvate dioxygenase
- Transgenes to provide herbicide tolerance traits comprise: for tolerance to glyphosate: cp4 epsps, epsps grg23ace5, mepsps, 2mepsps, gat4601, gat4621, goxv247; for tolerance to glufosinate: pat and bar, for tolerance to 2,4-D: aad-1 , aad-12; for tolerance to dicamba: dmo; for tolerance to oxynil herbicies: bxn; for tolerance to sulfonylurea herbicides: zm-hra, csr1-2, gm-hra, S4-HrA; for tolerance to ALS inhibitors: csr1-2; and for tolerance to HPPD inhibitors: hppdPF, W336, avhppd-03.
- Transgenic corn events comprising herbicide tolerance genes include, but are not limited to, DAS40278, MON801, MON802, MON809, MON810, MON832, MON87411, MON87419, MON87427, MON88017, MON89034, NK603, GA21, MZHG0JG, HCEM485, VCO-01981-5, 676, 678, 680, 33121, 4114, 59122, 98140, Bt10, Bt176, CBH-351, DBT418, DLL25, MS3,
- Transgenic soybean events comprising herbicide tolerance genes include, but are not limited to, GTS 40-3-2, MON87705, MON87708, MON87712, MON87769, MON89788, A2704-12, A2704-21, A5547-127, A5547-35, DP356043, DAS44406-6, DAS68416-4, DAS-81419-2, GU262, SYHT0H2, W62, W98, FG72 and CV127.
- Transgenic cotton events comprising herbicide tolerance genes include, but are not limited to, 19-51 a, 31707, 42317, 81910, 281-24-236, 3006-210-23, BXN10211, BXN10215, BXN10222, BXN 10224, MON1445, MON1698, MON88701, MON88913, GHB119, GHB614, LLCotton25, T303-3 and T304-40.
- Transgenic canola events comprising herbicide tolerance genes are for example, but not excluding others, MON88302, HCR-1, HCN10, HCN28, HCN92, MS1, MS8, PHY14, PHY23, PHY35, PHY36, RF1, RF2 and RF3.
- Transgenes to provide insect resistance preferably are toxin genes of Bacillus spp. and synthetic variants thereof, like cry1A, crylAb, cry1Ab-Ac, crylAc, cry1A.105, cry1F, cry1Fa2, cry2Ab2, cry2Ae, mcry3A, ecry3.1Ab, cry3Bb1, cry34Ab1, cry35Ab1, cry9C, vip3A(a), vip3Aa20.
- transgenes of plant origin such as genes coding for protease inhibitors, like CpTI and pinll, can be used.
- a further approach uses transgenes such as dvsnf7 to produce double-stranded RNA in plants.
- Transgenic corn events comprising genes for insecticidal proteins or double stranded RNA include, but are not limited to, Bt10, Bt11, Bt176, MON801, MON802, MON809, MON810, MON863, MON87411 , MON88017, MON89034, 33121, 4114, 5307, 59122, TC1507, TC6275, CBH-351, MIR162, DBT418 and MZIR098.
- Transgenic soybean events comprising genes for insecticidal proteins include, but are not limited to, MON87701, MON87751 and DAS-81419.
- Transgenic cotton events comprising genes for insecticidal proteins include, but are not limited to, SGK321 , MON531 , MON757, MON1076, MON15985, 31707, 3253, 3257, 3258, 42317,
- Cultivated plants with increased yield have been created by using the transgene athb17 (e.g. corn event MON87403), or bbx32 (e.g. soybean event MON87712).
- athb17 e.g. corn event MON87403
- bbx32 e.g. soybean event MON87712
- Cultivated plants comprising a modified oil content have been created by using the transgenes: gm-fad2-1, Pj.D6D, Nc.Fad3, fad2-1A and fatb1-A (e.g. soybean events 260-05, MON87705 and MON87769).
- Preferred combinations of traits are combinations of herbicide tolerance traits to different groups of herbicides, combinations of insect tolerance to different kind of insects, in particular tolerance to lepidopteran and coleopteran insects, combinations of herbicide tolerance with one or several types of insect resistance, combinations of herbicide tolerance with increased yield as well as combinations of herbicide tolerance and tolerance to abiotic conditions.
- Plants comprising singular or stacked traits as well as the genes and events providing these traits are well known in the art.
- detailed information as to the mutagenized or integrated genes and the respective events are available from websites of the organizations “International Service for the Acquisition of Agri-biotech Applications (ISAAA)” (http://www.isaaa.org/gmapprovaldatabase) and the “Center for Environmental Risk Assessment (CERA)” (http://cera-gmc.org/GMCropDatabase). Further information on specific events and methods to detect them can be found for canola events MS1, MS8, RF3, GT73, MON88302, KK179 in W001/031042, W001/041558, W001/041558, W002/036831,
- GHB614, T304-40 GHB119, MON88701, 81910 in WO02/034946, W002/100163, W002/100163, WO03/013224, WO04/072235, WO04/039986, WO05/103266, WO05/103266, WO06/128573, W007/017186, W008/122406, W008/151780, WO12/134808, W013/112527; for corn events GA21, MON810, DLL25, TC1507, MON863, MIR604, LY038, MON88017, 3272, 59122, NK603, MIR162, MON89034, 98140, 32138, MON87460, 5307, 4114, MON87427, DAS40278, MON87411, 33121, MON87403, MON87419 in W098/044140, US02/102582, US03/126634, WO04/099447,
- effects which are specific to a cultivated plant comprising a certain transgene or event may result in effects which are specific to a cultivated plant comprising a certain transgene or event. These effects might involve changes in growth behavior or changed resistance to biotic or abiotic stress factors. Such effects may in particular comprise enhanced yield, enhanced resistance or tolerance to insects, nematodes, fungal, bacterial, mycoplasma, viral or viroid pathogens as well as early vigour, early or delayed ripening, cold or heat tolerance as well as changed amino acid or fatty acid spectrum or content.
- the compounds I and compositions thereof, respectively, are particularly suitable for controlling the following causal agents of plant diseases:
- Albugo spp. white rust on ornamentals, vegetables (e. g. A. Candida ) and sunflowers (e. g. A. tragopogonis ); Alternaria spp. (Alternaria leaf spot) on vegetables (e.g. A. dauci or A. porn), oilseed rape (A. brassicicola or brassicae), sugar beets (A. tenuis), fruits (e.g. A. grandis), rice, soybeans, potatoes and tomatoes (e. g. A. solani, A. grandis or A. alternata), tomatoes (e. g. A. solani or A. alternata) and wheat (e.g. A. triticina) ⁇ , Aphanomyces spp.
- Ascochyta spp. on cereals and vegetables e. g. A. tritici (anthracnose) on wheat and A. hordei on barley; Aureobasidium zeae (syn. Kapatiella zeae) on corn; Bipolaris and Drechslera spp. (teleomorph: Cochliobolus spp.), e. g. Southern leaf blight (D. maydis) or Northern leaf blight ( B . zeicola) on corn, e. g. spot blotch ( B . sorokiniana) on cereals and e. g.
- Botrytis cinerea teleomorph: Botryotinia fuckeliana grey mold) on fruits and berries (e. g. strawberries), vegetables (e. g. lettuce, carrots, celery and cabbages); B. squamosa
- beans e. g. C. lindemuthianum
- soybeans e. g. C. truncatum or C. gloeosporioides
- vegetables e.g. C. lagenarium or C. capsici
- fruits e.g. C. acutatum
- coffee e.g. C. coffeanum or C. kahawae
- Corticium spp. e. g. C.
- sasakii sheath blight
- Corynespora cassiicola leaf spots
- Cycloconium spp. e. g. C. oleaginum on olive trees
- ampelina anthracnose
- Entyloma oryzae leaf smut
- Epicoccum spp. black mold
- Erysiphe spp. potowdery mildew
- sugar beets E. betae
- vegetables e. g. E. pisi
- cucurbits e. g. E. cichoracearum
- cabbages oilseed rape (e. g. E. cruciferarum)]
- Eutypa lata (Eutypa canker or dieback, anamorph: Cytosporina lata, syn.
- G. sabinae rust on pears
- Helminthosporium spp. syn. Drechslera, teleomorph: Cochliobolus
- Hemileia spp. e. g. H. vastatrix (coffee leaf rust) on coffee
- Isariopsis clavispora syn. Cladosporium vitis
- Macrophomina phaseolina syn. phaseoli
- root and stem rot on soybeans and cotton
- Microdochium syn. Fusarium
- nivale pink snow mold
- Microsphaera diffusa (powdery mildew) on soybeans; Monilinia spp., e. g. M. laxa, M. fructicola and M. fructigena (syn. Monilia spp.: bloom and twig blight, brown rot) on stone fruits and other rosaceous plants; Mycosphaerella spp. on cereals, bananas, soft fruits and ground nuts, such as e. g. M. graminicola (anamorph: Zymoseptoria tritici formerly Septoria triticr. Septoria blotch) on wheat or M. fijiensis (syn. Pseudocercospora fijiensis ⁇ .
- meibomiae (soybean rust) on soybeans; Phialophora spp. e. g. on vines (e. g. P. tracheiphila and P. tetraspora) and soybeans (e. g. P. gregata : stem rot); Phoma lingam (syn. Leptosphaeria biglobosa and L maculans. root and stem rot) on oilseed rape and cabbage, P. betae (root rot, leaf spot and damping-off) on sugar beets and P. zeae-maydis (syn. Phyllostica zeae) on corn; Phomopsis spp. on sunflowers, vines (e.
- soybeans e. g. P. viticoia ⁇ can and leaf spot
- soybeans e. g. stem rot: P. phaseoli, teleomorph: Diaporthe phaseolorum
- Physoderma maydis brown spots
- Phytophthora spp. wilt, root, leaf, fruit and stem root
- various plants such as paprika and cucurbits (e. g. P. capsici), soybeans (e. g. P. megasperma, syn. P. sojae), potatoes and tomatoes (e. g. P. infestans : late blight) and broad-leaved trees (e. g. P.
- Plasmodiophora brassicae club root
- Plasmopara spp. e. g. P. viticola (grapevine downy mildew) on vines and P. halstedii on sunflowers
- Podosphaera spp. powdery mildew) on rosaceous plants, hop, pome and soft fruits (e. g. P. leucotricha on apples) and curcurbits (P. xanthii );
- Polymyxa spp. e. g. on cereals, such as barley and wheat (P. graminis) and sugar beets (P. betae) and thereby transmitted viral diseases; Pseudocercosporella herpotrichoides (syn. Oculimacula yallundae,
- O. acuformis eyespot, teleomorph: Tapesia yallundae) on cereals, e. g. wheat or barley; Pseudoperonospora (downy mildew) on various plants, e. g. P. cubensis on cucurbits or P. humili on hop; Pseudopezicula tracheiphila (red fire disease or .rotbrenner’, anamorph: Phialophora ) on vines; Puccinia spp. (rusts) on various plants, e. g. P. triticina (brown or leaf rust), P. striiformis (stripe or yellow rust), P.
- hordei dwarf rust
- P. graminis stem or black rust
- P. recondita brown or leaf rust
- cereals such as e. g. wheat, barley or rye
- P. kuehnii range rust
- sugar cane and P. asparagi on asparagus
- Pyrenopeziza spp. e.g. P. brassicae on oilseed rape
- Pyrenophora anamorph: Drechslera ) tritici-repentis (tan spot) on wheat or P. teres (net blotch) on barley
- Pyricularia spp. e. g. P.
- oryzae (teleomorph: Magnaporthe grisea ⁇ rice blast) on rice and P. grisea on turf and cereals; Pythium spp. (damping-off) on turf, rice, corn, wheat, cotton, oilseed rape, sunflowers, soybeans, sugar beets, vegetables and various other plants (e. g. P. ultimum or P. aphanidermatum ) and P. oligandrum on mushrooms; Ramularia spp., e. g. R. collo-cygni (Ramularia leaf spots, Physiological leaf spots) on barley, R. areola (teleomorph: Mycosphaerella areola ) on cotton and R.
- Rhizoctonia spp. on cotton, rice, potatoes, turf, corn, oilseed rape, potatoes, sugar beets, vegetables and various other plants, e. g. R. solani (root and stem rot) on soybeans, R. solani (sheath blight) on rice or R. cerealis (Rhizoctonia spring blight) on wheat or barley; Rhizopus stolonifer (black mold, soft rot) on strawberries, carrots, cabbage, vines and tomatoes; Rhynchosporium secalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and
- Attenuatum (sheath rot) on rice; Sclerotinia spp. (stem rot or white mold) on vegetables (S. minor and S. sclerotiorum ) and field crops, such as oilseed rape, sunflowers (e. g. S. sclerotiorum) and soybeans, S. rolfsii (syn. Athelia rolfsii) on soybeans, peanut, vegetables, corn, cereals and ornamentals; Septoria spp. on various plants, e. g. S. glycines (brown spot) on soybeans, S. tritici (syn. Zymoseptoria tritici, Septoria blotch) on wheat and S.
- Podosphaera xanthii. powdery mildew on cucurbits
- Spongospora subterranea powdery scab
- Stagonospora spp. on cereals, e. g. S. nodorum (Stagonospora blotch, teleomorph: Leptosphaeria [syn. Phaeosphaeria] nodorum, syn. Septoria nodorum) on wheat
- Synchytrium endobioticum on potatoes potato wart disease
- Taphrina spp. e. g. T. deformans (leaf curl disease) on peaches and T.
- pruni plum pocket
- Thielaviopsis spp. black root rot
- tobacco, pome fruits, vegetables, soybeans and cotton e. g. T. basicola (syn. Chalara elegans) Tilletia spp.
- the compounds I and compositions thereof, respectively, are particularly suitable for controlling the following causal agents of plant diseases: rusts on soybean and cereals (e.g. Phakopsora pachyrhizi and P. meibomiae on soy; Puccinia tritici and P. striiformis on wheat); molds on specialty crops, soybean, oil seed rape and sunflowers (e.g. Botrytis cinerea on strawberries and vines, Sclerotinia sclerotiorum, S. minor and S. rolfsii on oil seed rape, sunflowers and soybean); Fusarium diseases on cereals (e.g. Fusarium culmorum and F.
- rusts on soybean and cereals e.g. Phakopsora pachyrhizi and P. meibomiae on soy; Puccinia tritici and P. striiformis on wheat
- molds on specialty crops soybean, oil seed rape and sunflowers (e.g. Botryt
- compounds I.A-1.1a.B-1 to I.A-1.1a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-2.1a.B-1 to I.A-2.1a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-3.1a.B-1 to I.A-3.1a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-4.1a.B-1 to I.A-4.1a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-1.2a.B-1 to I.A-1.2a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-2.2a.B-1 to I.A-2.2a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-3.2a.B-1 to I.A-3.2a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-4.2a.B-1 to I.A-4.2a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-1.3a.B-1 to I.A-1.3a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-2.3a.B-1 to I.A-2.3a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-3.3a.B-1 to I.A-3.3a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-4.3a.B-1 to I.A-4.3a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-1.4a.B-1 to I.A-1.4a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-2.4a.B-1 to I.A-2.4a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-3.4a.B-1 to I.A-3.4a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-4.4a.B-1 to I.A-4.4a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-1.5a.B-1 to I.A-1.5a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-2.5a.B-1 to I.A-2.5a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-3.5a.B-1 to I.A-3.5a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-4.5a.B-1 to I.A-4.5a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-1.6a.B-1 to I.A-1.6a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-2.6a.B-1 to I.A-2.6a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-3.6a.B-1 to I.A-3.6a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-4.6a.B-1 to I.A-4.6a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-1.7a.B-1 to I.A-1.7a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-2.7a.B-1 to I.A-2.7a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-3.7a.B-1 to I.A-3.7a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.A-4.7a.B-1 to I.A-4.7a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-1.1a.B-1 to I.B-1.1a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-2.1a.B-1 to I.B-2.1a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-3.1a.B-1 to I.B-3.1a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-4.1a.B-1 to I.B-4.1a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-1.2a. B-1 to I.B-1.2a. B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-2.2a.B-1 to I.B-2.2a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-3.2a.B-1 to I.B-3.2a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-4.2a.B-1 to I.B-4.2a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I. B-1.3a. B-1 to I. B-1.3a. B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-2.3a.B-1 to I.B-2.3a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-3.3a.B-1 to I.B-3.3a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-4.3a.B-1 to I.B-4.3a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I. B-1.4a. B-1 to I. B-1.4a. B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-2.4a.B-1 to I.B-2.4a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-3.4a.B-1 to I.B-3.4a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-4.4a.B-1 to I.B-4.4a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I. B-1.5a. B-1 to I. B-1.5a. B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-2.5a.B-1 to I.B-2.5a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-3.5a.B-1 to I.B-3.5a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-4.5a.B-1 to I.B-4.5a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-1.6a.B-1 to I.B-1.6a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-2.6a.B-1 to I.B-2.6a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-3.6a.B-1 to I.B-3.6a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-4.6a.B-1 to I.B-4.6a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-1.7a.B-1 to I.B-1.7a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-2.7a.B-1 to I.B-2.7a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-3.7a.B-1 to I.B-3.7a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds I.B-4.7a.B-1 to I.B-4.7a.B-25 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- compounds Ex-1 to Ex-101 are particularly suitable for controlling the causal agents of plant diseases according to the list Z.
- Albugo spp. white rust on ornamentals, vegetables (e. g. A. Candida ) and sunflowers (e. g. A. tragopogonis ); Alternaria spp. (Alternaria leaf spot) on vegetables (e.g. A. dauci or A. porn), oilseed rape (A. brassicicola or brassicae), sugar beets (A. tenuis), fruits (e.g. A. grandis), rice, soybeans, potatoes and tomatoes (e. g. A. solani, A. grandis or A. alternata), tomatoes (e. g. A. solani or A. alternata) and wheat (e.g. A. triticina) ⁇ , Aphanomyces spp.
- Ascochyta spp. on cereals and vegetables e. g. A. tritici (anthracnose) on wheat and A. hordei on barley; Aureobasidium zeae (syn. Kapatiella zeae) on corn; Bipolaris and Drechslera spp. (teleomorph: Cochliobolus spp.), e. g. Southern leaf blight (D. maydis) or Northern leaf blight ( B . zeicola) on corn, e. g. spot blotch ( B . sorokiniana) on cereals and e. g.
- Botrytis cinerea teleomorph: Botryotinia fuckeliana grey mold) on fruits and berries (e. g. strawberries), vegetables (e. g. lettuce, carrots, celery and cabbages); B. squamosa
- miyabeanus anamorph: H. oryzae
- Colletotrichum teleomorph: Glomerella
- spp. anthracnose
- cotton e. g. C. gossypii
- corn e. g. C. graminicola: Anthracnose stalk rot
- soft fruits e. g.
- C. coccodes. black dot beans (e. g. C. lindemuthianum), soybeans (e. g. C. truncatum or C. gloeosporioides), vegetables (e.g. C. lagenarium or C. capsici), fruits (e.g. C. acutatum), coffee (e.g. C. coffeanum or C. kahawae) and C. gloeosporioides on various crops; Corticium spp., e. g. C.
- sasakii sheath blight
- Corynespora cassiicola leaf spots
- Cycloconium spp. e. g. C. oleaginum on olive trees
- ampelina anthracnose
- Entyloma oryzae leaf smut
- Epicoccum spp. black mold
- Erysiphe spp. potowdery mildew
- sugar beets E. betae
- vegetables e. g. E. pisi
- cucurbits e. g. E. cichoracearum
- cabbages oilseed rape (e. g. E. cruciferarum) ⁇
- Eutypa lata Eutypa canker or dieback, anamorph: Cytosporina lata, syn.
- G. sabinae rust on pears
- Helminthosporium spp. syn. Drechslera, teleomorph: Cochliobolus
- Hemileia spp. e. g. H. vastatrix (coffee leaf rust) on coffee
- Isariopsis clavispora syn. Cladosporium vitis
- Macrophomina phaseolina syn. phaseoli
- root and stem rot on soybeans and cotton
- Microdochium syn. Fusarium
- nivale pink snow mold
- Microsphaera diffusa (powdery mildew) on soybeans; Monilinia spp., e. g. M. laxa, M. fructicola and M. fructigena (syn. Monilia spp.: bloom and twig blight, brown rot) on stone fruits and other rosaceous plants; Mycosphaerella spp. on cereals, bananas, soft fruits and ground nuts, such as e. g. M. graminicola (anamorph: Zymoseptoria tritici formerly Septoria triticr. Septoria blotch) on wheat or M. fijiensis (syn. Pseudocercospora fijiensis ⁇ .
- meibomiae (soybean rust) on soybeans; Phialophora spp. e. g. on vines (e. g. P. tracheiphila and P. tetraspora) and soybeans (e. g. P. gregata : stem rot); Phoma lingam (syn. Leptosphaeria biglobosa and L. maculans. root and stem rot) on oilseed rape and cabbage, P. betae (root rot, leaf spot and damping-off) on sugar beets and P. zeae-maydis (syn. Phyllostica zeae) on corn; Phomopsis spp.
- soybeans e. g. stem rot: P. phaseoli, teleomorph: Diaporthe phaseolorum
- Physoderma maydis brown spots
- Phytophthora spp. wilt, root, leaf, fruit and stem root
- paprika and cucurbits e. g. P. capsici
- soybeans e. g. P. megasperma, syn. P. sojae
- potatoes and tomatoes e. g. P. infestans. late blight
- broad-leaved trees e. g. P. ramorurrr.
- Plasmodiophora brassicae club root
- Plasmopara spp. e. g. P. viticola (grapevine downy mildew) on vines and P. halstedii on sunflowers
- Podosphaera spp. powdery mildew) on rosaceous plants, hop, pome and soft fruits (e. g. P. leucotricha on apples) and curcurbits (P. xanthii );
- Polymyxa spp. e. g. on cereals, such as barley and wheat (P. graminis) and sugar beets (P. betae) and thereby transmitted viral diseases; Pseudocercosporella herpotrichoides (syn. Oculimacula yallundae,
- O. acuformis eyespot, teleomorph: Tapesia yallundae) on cereals, e. g. wheat or barley; Pseudoperonospora (downy mildew) on various plants, e. g. P. cubensis on cucurbits or P. humili on hop; Pseudopezicula tracheiphila (red fire disease or .rotbrenner’, anamorph: Phialophora) on vines; Puccinia spp. (rusts) on various plants, e. g. P. triticina (brown or leaf rust), P. striiformis (stripe or yellow rust), P.
- rusts rusts
- hordei dwarf rust
- P. graminis stem or black rust
- P. recondita brown or leaf rust
- cereals such as e. g. wheat, barley or rye
- P. kuehnii range rust
- sugar cane and P. asparagi on asparagus
- Pyrenopeziza spp. e.g. P. brassicae on oilseed rape
- Pyrenophora anamorph: Drechslera) tritici-repentis (tan spot) on wheat or P. teres (net blotch) on barley
- Pyricularia spp. e. g. P.
- oryzae (teleomorph: Magnaporthe grisea ⁇ rice blast) on rice and P. grisea on turf and cereals; Pythium spp. (damping-off) on turf, rice, corn, wheat, cotton, oilseed rape, sunflowers, soybeans, sugar beets, vegetables and various other plants (e. g. P. ultimum or P. aphanidermatum) and P. oligandrum on mushrooms; Ramularia spp., e. g. R. collo-cygni (Ramularia leaf spots, Physiological leaf spots) on barley, R. areola (teleomorph: Mycosphaerella areola) on cotton and R.
- Rhizoctonia spp. on cotton, rice, potatoes, turf, corn, oilseed rape, potatoes, sugar beets, vegetables and various other plants, e. g. R. solani (root and stem rot) on soybeans, R. solani (sheath blight) on rice or R. cerealis (Rhizoctonia spring blight) on wheat or barley; Rhizopus stolonifer (black mold, soft rot) on strawberries, carrots, cabbage, vines and tomatoes; Rhynchosporium secalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and R. sescalis and
- Attenuatum (sheath rot) on rice; Sclerotinia spp. (stem rot or white mold) on vegetables (S. minor and S. sclerotiorum) and field crops, such as oilseed rape, sunflowers (e. g. S. sclerotiorum) and soybeans, S. rolfsii (syn. Athelia rolfsii) on soybeans, peanut, vegetables, corn, cereals and ornamentals; Septoria spp. on various plants, e. g. S. glycines (brown spot) on soybeans, S. tritici (syn. Zymoseptoria tritici, Septoria blotch) on wheat and S.
- Podosphaera xanthir. powdery mildew on cucurbits
- Spongospora subterranea pausing potato
- Spongospora subterranea pausing potato
- Stagonospora spp. on cereals, e. g. S. nodorum (Stagonospora blotch, teleomorph: Leptosphaeria [syn. Phaeosphaeria ] nodorum, syn. Septoria nodorum ) on wheat
- Taphrina spp. e. g. T. deformans (leaf curl disease) on peaches and T.
- pruni plum pocket
- Thielaviopsis spp. black root rot
- tobacco, pome fruits, vegetables, soybeans and cotton e. g. T. basicola (syn. Chalara elegans ); Tilletia spp.
- the compounds I and compositions thereof, respectively, are also suitable for controlling harmful microorganisms in the protection of stored products or harvest, and in the protection of materials.
- stored products or harvest is understood to denote natural substances of plant or animal origin and their processed forms for which long-term protection is desired.
- Stored products of plant origin for example stalks, leafs, tubers, seeds, fruits or grains, can be protected in the freshly harvested state or in processed form, such as pre-dried, moistened, comminuted, ground, pressed or roasted, which process is also known as post-harvest treatment.
- timber whether in the form of crude timber, such as construction timber, electricity pylons and barriers, or in the form of finished articles, such as furniture or objects made from wood.
- Stored products of animal origin are hides, leather, furs, hairs and alike.
- stored products is understood to denote natural substances of plant origin and their processed forms, more preferably fruits and their processed forms, such as pomes, stone fruits, soft fruits and citrus fruits and their processed forms, where application of compounds I and compositions thereof can also prevent disadvantageous effects such as decay, discoloration or mold.
- protection of materials is to be understood to denote the protection of technical and non-living materials, such as adhesives, glues, wood, paper, paperboard, textiles, leather, paint dispersions, plastics, cooling lubricants, fiber, or fabrics against the infestation and destruction by harmful microorganisms, such as fungi and bacteria.
- the amount of active substance applied depends on the kind of application area and on the desired effect. Amounts customarily applied in the protection of materials are 0.001 g to 2 kg, preferably 0.005 g to 1 kg, of active substance per cubic meter of treated material.
- the compounds I and compositions thereof, respectively, may be used for improving the health of a plant.
- the invention also relates to a method for improving plant health by treating a plant, its propagation material, and/or the locus where the plant is growing or is to grow with an effective amount of compounds I and compositions thereof, respectively.
- plant health is to be understood to denote a condition of the plant and/or its products which is determined by several indicators alone or in combination with each other, such as yield (e. g. increased biomass and/or increased content of valuable ingredients), plant vigor (e. g. improved plant growth and/or greener leaves (“greening effect”)), quality (e. g. improved content or composition of certain ingredients), and tolerance to abiotic and/or biotic stress.
- yield e. g. increased biomass and/or increased content of valuable ingredients
- plant vigor e. g. improved plant growth and/or greener leaves (“greening effect”)
- quality e. g. improved content or composition of certain ingredients
- tolerance to abiotic and/or biotic stress e. g. improved content or composition of certain ingredients
- the compounds I are employed as such or in form of compositions by treating the fungi, the plants, plant propagation materials, such as seeds; soil, surfaces, materials, or rooms to be protected from fungal attack with a fungicidally effective amount of the active substances.
- the application can be carried out both before and after the infection of the plants, plant propagation materials, such as seeds; soil, surfaces, materials or rooms by the fungi.
- An agrochemical composition comprises a fungicidally effective amount of a compound I.
- fungicidally effective amount denotes an amount of the composition or of the compounds I, which is sufficient for controlling harmful fungi on cultivated plants or in the protection of stored products or harvest or of materials and which does not result in a substantial damage to the treated plants, the treated stored products or harvest, or to the treated materials. Such an amount can vary in a broad range and is dependent on various factors, such as the fungal species to be controlled, the treated cultivated plant, stored product, harvest or material, the climatic conditions and the specific compound I used.
- Plant propagation materials may be treated with compounds I as such or a composition com prising at least one compound I prophylactically either at or before planting or transplanting.
- the amounts of active substances applied are, depending on the kind of effect desired, from 0.001 to 2 kg per ha, preferably from 0.005 to 2 kg per ha, more preferably from 0.05 to 0.9 kg per ha, and in particular from 0.1 to 0.75 kg per ha.
- agrochemical composition In treatment of plant propagation materials, such as seeds, e. g. by dusting, coating, or drenching, amounts of active substance of generally from 0.1 to 1000 g, preferably from 1 to 1000 g, more preferably from 1 to 100 g and most preferably from 5 to 100 g, per 100 kg of plant propagation material (preferably seeds) are required.
- the user applies the agrochemical composition usually from a predosage device, a knapsack sprayer, a spray tank, a spray plane, or an irrigation system.
- the agrochemical composition is made up with water, buffer, and/or further auxiliaries to the desired application concentration and the ready-to-use spray liquor or the agrochemical composition according to the invention is thus obtained.
- 20 to 2000 liters, preferably 50 to 400 liters, of the ready-to-use spray liquor are applied per hectare of agricultural useful area.
- compositions e. g. solutions, emulsions, suspensions, dusts, powders, pastes, granules, pressings, capsules, and mixtures thereof.
- composition types see also “Catalogue of pesticide formulation types and international coding system”, Technical Monograph No. 2, 6 th Ed. May 2008, CropLife International) are suspensions (e. g. SC, OD,
- FS emulsifiable concentrates
- emulsions e. g. EW, EO, ES, ME
- capsules e. g.
- CS, ZC pastes, pastilles, wettable powders or dusts (e. g. WP, SP, WS, DP, DS), pressings (e. g. BR, TB, DT), granules (e. g. WG, SG, GR, FG, GG, MG), insecticidal articles (e. g. LN), as well as gel formulations for the treatment of plant propagation materials, such as seeds (e. g. GF).
- WP wettable powders or dusts
- pressings e. g. BR, TB, DT
- granules e. g. WG, SG, GR, FG, GG, MG
- insecticidal articles e. g. LN
- gel formulations for the treatment of plant propagation materials such as seeds (e. g. GF).
- compositions are prepared in a known manner, such as described by Mollet and Grubemann, Formulation technology, Wiley VCH, Weinheim, 2001; or by Knowles, New developments in crop protection product formulation, Agrow Reports DS243, T&F Informa, London, 2005.
- the invention also relates to agrochemical compositions comprising an auxiliary and at least one compound I.
- auxiliaries are solvents, liquid carriers, solid carriers or fillers, surfactants, dispersants, emulsifiers, wetters, adjuvants, solubilizers, penetration enhancers, protective colloids, adhesion agents, thickeners, humectants, repellents, attractants, feeding stimulants, compatibilizers, bactericides, anti-freezing agents, anti-foaming agents, colorants, tackifiers, and binders.
- Suitable solvents and liquid carriers are water and organic solvents, such as mineral oil fractions of medium to high boiling point, e. g. kerosene, diesel oil; oils of vegetable or animal origin; aliphatic, cyclic and aromatic hydrocarbons, e. g. toluene, paraffin, tetrahydronaphtha- lene, and alkylated naphthalenes; alcohols, e. g. ethanol, propanol, butanol, benzyl alcohol, cyclohexanol, glycols; DMSO; ketones, e. g. cyclohexanone; esters, e. g. lactates, carbonates, fatty acid esters, gamma-butyrolactone; fatty acids; phosphonates; amines; amides, e. g.
- N- methyl pyrrolidone N- methyl pyrrolidone, fatty acid dimethyl amides; and mixtures thereof.
- Suitable solid carriers or fillers are mineral earths, e. g. silicates, silica gels, talc, kaolins, limestone, lime, chalk, clays, dolomite, diatomaceous earth, bentonite, calcium sulfate, magnesium sulfate, magnesium oxide; polysaccharides, e. g. cellulose, starch; fertilizers, e. g. ammonium sulfate, ammonium phosphate, ammonium nitrate, ureas; products of vegetable origin, e. g. cereal meal, tree bark meal, wood meal, nutshell meal, and mixtures thereof.
- mineral earths e. g. silicates, silica gels, talc, kaolins, limestone, lime, chalk, clays, dolomite, diatomaceous earth, bentonite, calcium sulfate, magnesium sulfate, magnesium oxide
- polysaccharides e. g. cellulose, star
- Suitable surfactants are surface-active compounds, such as anionic, cationic, nonionic and amphoteric surfactants, block polymers, polyelectrolytes, and mixtures thereof. Such surfactants can be used as emulsifier, dispersant, solubilizer, wetter, penetration enhancer, protective colloid, or adjuvant. Examples of surfactants are listed in McCutcheon’s, Vol.1 : Emulsifiers & Detergents, McCutcheon’s Directories, Glen Rock, USA, 2008 (International Ed. or North American Ed.).
- Suitable anionic surfactants are alkali, alkaline earth or ammonium salts of sulfonates, sulfates, phosphates, carboxylates, and mixtures thereof.
- sulfonates are alkylaryl sulfonates, diphenyl sulfonates, alpha-olefin sulfonates, lignin sulfonates, sulfonates of fatty acids and oils, sulfonates of ethoxylated alkylphenols, sulfonates of alkoxylated arylphenols, sulfonates of condensed naphthalenes, sulfonates of dodecyl- and tridecylbenzenes, sulfonates of naphthalenes and of alkyl naphthalenes, sulfosuccinates, or sulfosuccinamates.
- Examples of sulfates are sulfates of fatty acids, of oils, of ethoxylated alkylphenols, of alcohols, of ethoxy lated alcohols, or of fatty acid esters.
- Examples of phosphates are phosphate esters.
- Examples of carboxylates are alkyl carboxylates, and carboxylated alcohol or alkylphenol ethoxylates.
- Suitable nonionic surfactants are alkoxylates, /V-substituted fatty acid amides, amine oxides, esters, sugar-based surfactants, polymeric surfactants, and mixtures thereof.
- alkoxylates are compounds such as alcohols, alkylphenols, amines, amides, arylphenols, fatty acids or fatty acid esters which have been alkoxylated with 1 to 50 equivalents.
- Ethylene oxide and/or propylene oxide may be employed for the alkoxylation, preferably ethylene oxide.
- Examples of /V-substituted fatty acid amides are fatty acid glucamides or fatty acid alkanolamides.
- esters are fatty acid esters, glycerol esters, or monoglycerides.
- sugar-based surfactants are sorbitans, ethoxylated sorbitans, sucrose and glucose esters, or alkylpolyglucosides.
- polymeric surfactants are home- or copolymers of vinyl pyrrolidone, vinyl alcohols, or vinyl acetate.
- Suitable cationic surfactants are quaternary surfactants, for example quaternary ammonium compounds with one or two hydrophobic groups, or salts of long-chain primary amines.
- Suitable amphoteric surfactants are alkylbetains and imidazolines.
- Suitable block polymers are block polymers of the A-B or A-B-A type comprising blocks of polyethylene oxide and polypropylene oxide, or of the A-B-C type comprising alkanol, polyethylene oxide, and polypropylene oxide.
- Suitable polyelectrolytes are polyacids or polybases. Examples of polyacids are alkali salts of polyacrylic acid or polyacid comb polymers. Examples of polybases are polyvinyl amines or polyethylene amines.
- Suitable adjuvants are compounds, which have a negligible or even no pesticidal activity themselves, and which improve the biological performance of the compound I on the target.
- examples are surfactants, mineral or vegetable oils, and other auxiliaries. Further examples are listed by Knowles, Adjuvants and additives, Agrow Reports DS256, T&F Informa UK, 2006, chapter 5.
- Suitable thickeners are polysaccharides (e. g. xanthan gum, carboxymethyl cellulose), inorganic clays (organically modified or unmodified), polycarboxylates, and silicates.
- Suitable bactericides are bronopol and isothiazolinone derivatives, such as alkyliso- thiazolinones and benzisothiazolinones.
- Suitable anti-freezing agents are ethylene glycol, propylene glycol, urea and glycerin.
- Suitable anti-foaming agents are silicones, long chain alcohols, and salts of fatty acids.
- Suitable colorants are pigments of low water solubility and water-soluble dyes.
- examples are inorganic colorants (e. g. iron oxide, titan oxide, iron hexacyanoferrate) and organic colorants (e. g. alizarin-, azo- and phthalocyanine colorants).
- Suitable tackifiers or binders are polyvinyl pyrrolidones, polyvinyl acetates, polyvinyl alcohols, polyacrylates, biological or synthetic waxes, and cellulose ethers.
- the agrochemical compositions generally comprise between 0.01 and 95 %, preferably between 0.1 and 90 %, more preferably between 1 and 70 %, and in particular between 10 and 60 %, by weight of active substances (e.g. at least one compound I).
- the agrochemical compositions generally comprise between 5 and 99.9 %, preferably between 10 and 99.9 %, more preferably between 30 and 99 %, and in particular between 40 and 90 %, by weight of at least one auxiliary.
- the active substances (e.g. compounds I) are employed in a purity of from 90 % to 100 %, preferably from 95-% to 100 % (according to NMR spectrum).
- compositions in question give, after two-to-tenfold dilution, active substance concentrations of from 0.01 to 60 % by weight, preferably from 0.1 to 40 %, in the ready-to-use preparations. Application can be carried out before or during sowing.
- Methods for applying compound I and compositions thereof, respectively, onto plant propagation material, especially seeds include dressing, coating, pelleting, dusting, soaking, as well as in-furrow application methods.
- compound I or the compositions thereof, respectively are applied on to the plant propagation material by a method such that germination is not induced, e. g. by seed dressing, pelleting, coating, and dusting.
- oils, wetters, adjuvants, fertilizers, or micronutrients, and further pesticides may be added to the compounds I or the compositions thereof as premix, or, not until immediately prior to use (tank mix).
- pesticides e. g. fungicides, growth regulators, herbicides, insecticides, safeners
- These agents can be admixed with the compositions according to the invention in a weight ratio of 1 : 100 to 100: 1 , preferably 1 : 10 to 10: 1.
- a pesticide is generally a chemical or biological agent (such as pestidal active ingredient, compound, composition, virus, bacterium, antimicrobial, or disinfectant) that through its effect deters, incapacitates, kills or otherwise discourages pests.
- Target pests can include insects, plant pathogens, weeds, mollusks, birds, mammals, fish, nematodes (roundworms), and microbes that destroy property, cause nuisance, spread disease or are vectors for disease.
- pesticide includes also plant growth regulators that alter the expected growth, flowering, or reproduction rate of plants; defoliants that cause leaves or other foliage to drop from a plant, usually to facilitate harvest; desiccants that promote drying of living tissues, such as unwanted plant tops; plant activators that activate plant physiology for defense of against certain pests; safeners that reduce unwanted herbicidal action of pesticides on crop plants; and plant growth promoters that affect plant physiology e.g. to increase plant growth, biomass, yield or any other quality parameter of the harvestable goods of a crop plant.
- Biopesticides have been defined as a form of pesticides based on microorganisms (bacteria, fungi, viruses, nematodes, etc.) or natural products (compounds, such as metabolites, proteins, or extracts from biological or other natural sources) (U.S. Environmental Protection Agency: http://www.epa.gov/pesticides/biopesticides/). Biopesticides fall into two major classes, microbial and biochemical pesticides:
- Microbial pesticides consist of bacteria, fungi or viruses (and often include the metabolites that bacteria and fungi produce). Entomopathogenic nematodes are also classified as microbial pesticides, even though they are multi-cellular.
- Biochemical pesticides are naturally occurring substances that control pests or provide other crop protection uses as defined below, but are relatively non-toxic to mammals.
- Inhibitors of complex III at Q 0 site azoxystrobin (A.1.1), coumethoxystrobin (A.1.2), coumoxystrobin (A.1.3), dimoxystrobin (A.1.4), enestroburin (A.1.5), fenaminstrobin (A.1.6), fenoxystrobin/flufenoxystrobin (A.1.7), fluoxastrobin (A.1.8), kresoxim-methyl (A.1.9), mandestrobin (A.1.10), metominostrobin (A.1.11), orysastrobin (A.1.12), picoxystrobin (A.1.13), pyraclostrobin (A.1.14), pyrametostrobin (A.1.15), pyraoxystrobin (A.1.16), trifloxy- strobin (A.1.17) , 2-(2-(3-(2,6-dichlorophenyl)-1 -methyl-allylideneaminooxymethyl)-phenyl)- 2-
- respiration inhibitors diflumetorim (A.4.1); nitrophenyl derivates: binapacryl (A.4.2), dinobuton (A.4.3), dinocap (A.4.4), fluazinam (A.4.5), meptyldinocap (A.4.6), ferimzone (A.4.7); organometal compounds: fentin salts, e. g. fentin-acetate (A.4.8), fentin chloride (A.4.9) or fentin hydroxide (A.4.10); ametoctradin (A.4.11); silthiofam (A.4.12);
- - C14 demethylase inhibitors triazoles: azaconazole (B.1.1), bitertanol (B.1.2), bromu- conazole (B.1.3), cyproconazole (B.1.4), difenoconazole (B.1.5), diniconazole (B.1.6), diniconazole-M (B.1.7), epoxiconazole (B.1.8), fenbuconazole (B.1.9), fluquinconazole (B.1.10), flusilazole (B.1.11), flutriafol (B.1.12), hexaconazole (B.1.13), imibenconazole (B.1.14), ipconazole (B.1.15), metconazole (B.1.17), myclobutanil (B.1.18), oxpoconazole (B.1.19), paclobutrazole (B.1.20), penconazole (B.1.21), propiconazole (B
- Sterol biosynthesis inhibitors chlorphenomizole (B.4.1); C) Nucleic acid synthesis inhibitors
- benalaxyl (C.1.1), benalaxyl-M (C.1.2), kiralaxyl (C.1.3), metalaxyl (C.1.4), metalaxyl-M (C.1.5), ofurace (C.1.6), oxadixyl (C.1.7);
- nucleic acid synthesis inhibitors hymexazole (C.2.1), octhilinone (C.2.2), oxolinic acid (C.2.3), bupirimate (C.2.4), 5-fluorocytosine (C.2.5), 5-fluoro-2-(p-tolylmethoxy)pyrimidin- 4-amine (C.2.6), 5-fluoro-2-(4-fluorophenylmethoxy)pyrimidin-4-amine (C.2.7), 5-fluoro- 2-(4-chlorophenylmethoxy)pyrimidin-4 amine (C.2.8);
- tubulin inhibitors benomyl (D.1.1), carbendazim (D.1.2), fuberidazole (D1.3), thiabendazole (D.1.4), thiophanate-methyl (D.1.5), pyridachlometyl (D.1.6), A/-ethyl-2-[(3-ethynyl-8-methyl- 6-quinolyl)oxy]butanamide (D.1.8), A/-ethyl-2-[(3-ethynyl-8-methyl-6-quinolyl)oxy]-2-methyl- sulfanyl-acetamide (D.1.9), 2-[(3-ethynyl-8-methyl-6-quinolyl)oxy]-/ ⁇ /-(2-fluoroethyl)butan- amide (D.1.10), 2-[(3-ethynyl-8-methyl-6-quinolyl)oxy]-/ ⁇ /-(2-fluoroethyl)-2-methoxy-acet- amide
- diethofencarb (D.2.1), ethaboxam (D.2.2), pencycuron (D.2.3), fluopicolide (D.2.4), zoxamide (D.2.5), metrafenone (D.2.6), pyriofenone (D.2.7), phenamacril (D.2.8);
- cyprodinil E.1.1
- mepanipyrim E.1.2
- pyrimethanil E.1.3
- blasticidin-S (E.2.1), kasugamycin (E.2.2), kasugamycin hydro- chloride-hydrate (E.2.3), mildiomycin (E.2.4), streptomycin (E.2.5), oxytetracyclin (E.2.6);
- fluoroimid F.1.1
- iprodione F.1.2
- procymidone F.1.3
- vinclozolin F.1.4
- fludioxonil F.1.5
- quinoxyfen F.2.1
- edifenphos (G .1.1), iprobenfos (G.1.2), pyrazophos (G.1.3), isoprothiolane (G.1.4);
- dicloran G.2.1
- quintozene G.2.2
- tecnazene G.2.3
- tolclofos-methyl G.2.4
- biphenyl G.2.5
- chloroneb G.2.6
- etridiazole G.2.7
- zinc thiazole G.2.8
- dimethomorph G.3.1
- flumorph G.3.2
- mandipropamid G.3.3
- pyrimorph G.3.4
- benthiavalicarb G.3.5
- iprovalicarb G.3.6
- valifenalate G.3.7
- ferbam H.2.1
- mancozeb H.2.2
- maneb H.2.3
- metam H.2.4
- metiram H.2.5
- propineb H.2.6
- thiram H.2.7
- zineb H.2.8
- ziram H.2.9
- organochlorine compounds anilazine (H.3.1), chlorothalonil (H.3.2), captafol (H.3.3), captan (H.3.4), folpet (H.3.5), dichlofluanid (H.3.6), dichlorophen (H.3.7), hexachlorobenzene (H.3.8), pentachlorphenole (H.3.9) and its salts, phthalide (H.3.10), tolylfluanid (H.3.11);
- guanidine H.4.1
- dodine H.4.2
- dodine free base H.4.3
- guazatine H.4.4
- guazatine-acetate H.4.5
- iminoctadine H.4.6
- iminoctadine-triacetate H.4.7
- iminoctadine-tris(albesilate) H.4.8
- dithianon H.4.9
- 2,6-dimethyl-1H,5H-[1,4]di- thiino[2,3-c:5,6-c']dipyrrole-1,3,5,7(2H,6H)-tetraone H.4.10)
- - melanin synthesis inhibitors pyroquilon (1.2.1), tricyclazole (1.2.2), carpropamid (1.2.3), dicyclomet (1.2.4), fenoxanil (1.2.5);
- B. amyloliquefaciens B. amyloliquefaciens ssp. plantarum (also referred to as B. velezensis), B. megaterium, B. mojavensis, B. mycoides, B. pumilus, B. simplex, B. solisalsi, B. subtilis, B. subtilis var. amyloliquefaciens, B. velezensis, Candida oleo- phila, C.
- Biochemical pesticides with fungicidal, bactericidal, viricidal and/or plant defense activator activity harpin protein, Reynoutria sachalinensis extract;
- Microbial pesticides with insecticidal, acaricidal, molluscidal and/or nematicidal activity Agrobacterium radiobacter, Bacillus cereus, B. firmus, B. thuringiensis, B. thuringiensis ssp. aizawai, B. t. ssp. israelensis, B. t. ssp. galleriae, B. t. ssp. kurstaki, B. t. ssp. tene- brionis, Beauveria bassiana, B.
- Agrobacterium radiobacter Bacillus cereus, B. firmus, B. thuringiensis, B. thuringiensis ssp. aizawai, B. t. ssp. israelensis, B. t. ssp. galleriae, B. t. ssp. kurstaki, B. t. ssp.
- brongniartii Burkholderia spp., Chromobacterium sub- tsugae, Cydia pomonella granulovirus (CpGV), Cryptophlebia leucotreta granulovirus (CrleGV), Flavobacterium spp., Helicoverpa armigera nucleopolyhedrovirus (HearNPV), Helicoverpa zea nucleopolyhedrovirus (HzNPV), Helicoverpa zea single capsid nucleopolyhedrovirus (HzSNPV), Heterorhabditis bacteriophora, Isaria fumoso- rosea, Lecanicillium longisporum, L.
- HearNPV Helicoverpa armigera nucleopolyhedrovirus
- HzNPV Helicoverpa zea nucleopolyhedrovirus
- HzSNPV Helicoverpa zea single capsid nucleo
- Microbial pesticides with plant stress reducing, plant growth regulator, plant growth promoting and/or yield enhancing activity Azospirillum amazonense, A. brasilense, A. lipoferum, A. irakense, A. halopraeferens, Bradyrhizobium spp., B. elkanii, B.japoni- cum, B. liaoningense, B. lupini, Delftia acidovorans, Glomus intraradices, Mesorhizo- bium spp., Rhizobium leguminosarum bv. phaseoli, R. I. bv. trifolii, R. I. bv. viciae, R. tropici, Sinorhizobium melilotr,
- Acetylcholine esterase (AChE) inhibitors aldicarb, alanycarb, bendiocarb, benfuracarb, butocarboxim, butoxycarboxim, carbaryl, carbofuran, carbosulfan, ethiofencarb, fenobucarb, formetanate, furathiocarb, isoprocarb, methiocarb, methomyl, metolcarb, oxamyl, pirimicarb, propoxur, thiodicarb, thiofanox, trimethacarb, XMC, xylylcarb, triazamate; acephate, aza- methiphos, azinphos-ethyl, azinphosmethyl, cadusafos, chlorethoxyfos, chlorfenvinphos, chlormephos, chlorpyrifos, chlorpyrifos-methyl, coumaphos, cyanophos,
- GABA-gated chloride channel antagonists endosulfan, chlordane; ethiprole, fipronil, flufiprole, pyrafluprole, pyriprole;
- Sodium channel modulators acrinathrin, allethrin, d-cis-trans allethrin, d-trans allethrin, bifenthrin, kappa-bifenthrin, bioallethrin, bioallethrin S-cylclopentenyl, bioresmethrin, cycloprothrin, cyfluthrin, beta-cyfluthrin, cyhalothrin, lambda-cyhalothrin, gamma-cyhalothrin, cypermethrin, alpha-cypermethrin, beta-cypermethrin, theta-cypermethrin, zeta-cyper- methrin, cyphenothrin, deltamethrin, empenthrin, esfenvalerate, etofenprox, fenpropathrin, f
- Nicotinic acetylcholine receptor (nAChR) agonists acetamiprid, clothianidin, cycloxaprid, dinotefuran, imidacloprid, nitenpyram, thiacloprid, thiamethoxam; 4,5-dihydro-/V-nitro-
- Nicotinic acetylcholine receptor allosteric activators spinosad, spinetoram;
- Chloride channel activators abamectin, emamectin benzoate, ivermectin, lepimectin, milbemectin;
- Juvenile hormone mimics hydroprene, kinoprene, methoprene; fenoxycarb, pyriproxyfen;
- miscellaneous non-specific (multi-site) inhibitors methyl bromide and other alkyl halides; chloropicrin, sulfuryl fluoride, borax, tartar emetic; O.9 Chordotonal organ TRPV channel modulators: pymetrozine, pyrifluquinazon; O.10 Mite growth inhibitors: clofentezine, hexythiazox, diflovidazin; etoxazole; O.11 Microbial disruptors of insect midgut membranes: Bacillus thuringiensis, Bacillus sphaericus and the insecticdal proteins they produce: Bacillus thuringiensis subsp.
- insecticidal compounds of unknown or uncertain mode of action afidopyropen, afoxolaner, azadirachtin, amidoflumet, benzoximate, broflanilide, bromopropylate, chino- methionat, cryolite, cyproflanilide, dicloromezotiaz, dicofol, flufenerim, flometoquin, fluensulfone, fluhexafon, fluopyram, fluralaner, metoxadiazone, piperonyl butoxide, pyflu- bumide, pyridalyl, tioxazafen, 11-(4-chloro-2,6-dimethylphenyl)-12-hydroxy-1,4-dioxa-9- azadispiro[4.2.4.2]-tetradec-11-en-10-one, 3-(4’-fluoro-2,4-dimethylbiphenyl-3-yl)-4
- component 2 The active substances referred to as component 2, their preparation and their activity e. g. against harmful fungi is known (cf. : http://www.alanwood.net/pesticides/); these substances are commercially available.
- the compounds described by lUPAC nomenclature, their preparation and their pesticidal activity are also known (cf. Can. J. Plant Sci. 48(6), 587-94, 1968;
- the solid material (dry matter) of the biopesticides (with the exception of oils such as Neem oil) are considered as active components (e. g. to be obtained after drying or evaporation of the extraction or suspension medium in case of liquid formulations of the microbial pesticides).
- the weight ratios and percentages used for a biological extract such as Quillay extract are based on the total weight of the dry content (solid material) of the respective extract(s).
- the total weight ratios of compositions comprising at least one microbial pesticide in the form of viable microbial cells including dormant forms can be determined using the amount of CFU of the respective microorganism to calculate the total weight of the respective active component with the following equation that 1 x 10 10 CFU equals one gram of total weight of the respective active component.
- Colony forming unit is measure of viable microbial cells.
- CFU may also be understood as the number of (juvenile) individual nematodes in case of nematode biopesticides, such as Steinernema feltiae.
- the weight ratio of the component 1) and the component 2) generally depends from the properties of the components used, usually it is in the range of from 1:10,000 to 10,000:1, often from 1 :100 to 100:1, regularly from 1:50 to 50:1 , preferably from 1 :20 to 20:1, more preferably from 1 :10 to 10:1 , even more preferably from 1:4 to 4:1 and in particular from 1:2 to 2:1.
- the weight ratio of the component 1) and the component 2) usually is in the range of from 1000:1 to 1 :1, often from 100: 1 to 1:1 , regularly from 50:1 to 1 :1, preferably from 20:1 to 1:1 , more preferably from 10:1 to 1 :1, even more preferably from 4: 1 to 1 : 1 and in particular from 2: 1 to 1 : 1.
- the weight ratio of the component 1) and the component 2) usually is in the range of from 20,000:1 to 1 :10, often from 10,000:1 to 1:1, regularly from 5,000:1 to 5:1, preferably from 5,000:1 to 10:1, more preferably from 2,000:1 to 30:1 , even more preferably from 2,000:1 to 100:1 and in particular from 1,000:1 to 100:1.
- the weight ratio of the component 1) and the component 2) usually is in the range of from 1 :1 to 1:1000, often from 1 :1 to 1:100, regularly from 1:1 to 1 :50, preferably from 1:1 to 1 :20, more preferably from 1 :1 to 1:10, even more preferably from 1:1 to 1 :4 and in particular from 1 :1 to 1 :2.
- the weight ratio of the component 1) and the component 2) usually is in the range of from 10:1 to 1:20,000, often from 1 :1 to 1:10,000, regularly from 1:5 to 1 :5,000, preferably from 1 :10 to 1 :5,000, more preferably from 1:30 to 1:2,000, even more preferably from 1 :100 to 1:2,000 to and in particular from 1:100 to 1 :1 ,000.
- the weight ratio of component 1) and component 2) depends from the properties of the active substances used, usually it is in the range of from 1 :100 to 100:1, regularly from 1:50 to 50:1, preferably from 1:20 to 20:1, more preferably from 1 : 10 to 10: 1 and in particular from 1 :4 to 4: 1 , and the weight ratio of component 1 ) and component 3) usually it is in the range of from 1 :100 to 100:1 , regularly from 1:50 to 50:1, preferably from 1 :20 to 20: 1 , more preferably from 1 : 10 to 10:1 and in particular from 1 :4 to 4: 1. Any further active components are, if desired, added in a ratio of from 20:1 to 1 :20 to the component 1). These ratios are also suitable for mixtures applied by seed treatment.
- the application rates range from 1 x 10 6 to 5 x 10 16 (or more) CFU/ha, preferably from 1 x 10 8 to 1 x 10 13 CFU/ha, and even more preferably from 1 x 10 9 to 5 x 10 15 CFU/ha and in particular from 1 x 10 12 to 5 x 10 14 CFU/ha.
- the application rates regularly range from 1 x 10 5 to 1 x 10 12 (or more), preferably from 1 x 10 8 to 1 x 10 11 , more preferably from 5 x 10 8 to 1 x 10 10 individuals (e. g. in the form of eggs, juvenile or any other live stages, preferably in an infetive juvenile stage) per ha.
- the application rates generally range from 1 x 10 6 to 1 x 10 12 (or more) CFU/seed, preferably from 1 x 10 6 to 1 x 10 9 CFU/seed. Furthermore, the application rates with respect to seed treatment generally range from 1 x 10 7 to 1 x 10 14 (or more) CFU per 100 kg of seed, preferably from 1 x 10 9 to 1 x 10 12 CFU per 100 kg of seed.
- mixtures comprising as component 2) at least one active substance selected from inhibitors of complex III at Q 0 site in group A), more preferably selected from compounds (A.1.1), (A.1.4), (A.1.8), (A.1.9), (A.1.10), (A.1.12), (A.1.13), (A.1.14), (A.1.17),
- mixtures comprising as component 2) at least one active substance selected from inhibitors of complex III at Q, site in group A), more preferably selected from compounds (A.2.1), (A.2.3), (A.2.4) and (A.2.6); particularly selected from (A.2.3), (A.2.4) and (A.2.6).
- mixtures comprising as component 2) at least one active substance selected from inhibitors of complex II in group A), more preferably selected from compounds (A.3.2), (A.3.3), (A.3.4), (A.3.7), (A.3.9), (A.3.11), (A.3.12), (A.3.15), (A.3.16), (A.3.17), (A.3.18), (A.3.19), (A.3.20), (A.3.21), (A.3.22), (A.3.23), (A.3.24), (A.3.28), (A.3.31), (A.3.32), (A.3.33), (A.3.34), (A.3.35), (A.3.36), (A.3.37), (A.3.38) and (A.3.39); particularly selected from (A.3.2), (A.3.3), (A.3.4), (A.3.7), (A.3.9), (A.3.12), (A.3.15), (A.3.17), (A.3.19), (A.3.22), (A.3.23)
- mixtures comprising as component 2) at least one active substance selected from other respiration inhibitors in group A), more preferably selected from compounds (A.4.5) and (A.4.11); in particular (A.4.11).
- mixtures comprising as component 2) at least one active substance selected from C14 demethylase inhibitors in group B), more preferably selected from compounds (B.1.4), (B.1.5), (B.1.8), (B.1.10), (B.1.11), (B.1.12), (B.1.13), (B.1.17), (B.1.18),
- mixtures comprising as component 2) at least one active substance selected from Deltal 4-reductase inhibitors in group B), more preferably selected from compounds (B.2.4), (B.2.5), (B.2.6) and (B.2.8); in particular (B.2.4).
- mixtures comprising as component 2) at least one active substance selected from phenylamides and acyl amino acid fungicides in group C), more preferably selected from compounds (C.1.1), (C.1.2), (C.1.4) and (C.1.5); particularly selected from (C.1.1) and (C.1.4).
- mixtures comprising as component 2) at least one active substance selected from group E), more preferably selected from compounds (E.1.1), (E.1.3), (E.2.2) and (E.2.3); in particular (E.1.3).
- mixtures comprising as component 2) at least one active substance selected from group F), more preferably selected from compounds (F.1.2), (F.1.4) and (F.1.5).
- mixtures comprising as component 2) at least one active substance selected from group G), more preferably selected from compounds (G.3.1), (G.3.3), (G.3.6), (G.5.1), (G.5.3), (G.5.4), (G.5.5), G.5.6), G.5.7), (G.5.8), (G.5.9), (G.5.10) and (G.5.11); particularly selected from (G.3.1), (G.5.1) and (G.5.3).
- active substance selected from group G more preferably selected from compounds (G.3.1), (G.3.3), (G.3.6), (G.5.1), (G.5.3), (G.5.4), (G.5.5), G.5.6), G.5.7), (G.5.8), (G.5.9), (G.5.10) and (G.5.11); particularly selected from (G.3.1), (G.5.1) and (G.5.3).
- mixtures comprising as component 2) at least one active substance selected from group H), more preferably selected from compounds (H.2.2), (H.2.3), (H.2.5), (H.2.7), (H.2.8), (H.3.2), (H.3.4), (H.3.5), (H.4.9) and (H.4.10); particularly selected from (H.2.2), (H.2.5), (H.3.2), (H.4.9) and (H.4.10).
- mixtures comprising as component 2) at least one active substance selected from group I), more preferably selected from compounds (1.2.2) and (1.2.5).
- mixtures comprising as component 2) at least one active substance selected from group J), more preferably selected from compounds (J.1.2), (J.1.5), (J.1.8), (J.1.11) and (J.1.12); in particular (J.1.5).
- mixtures comprising as component 2) at least one active substance selected from group K), more preferably selected from compounds (K.1.41), (K.1.42), (K.1.44), (K.1.47), (K.1.57), (K.1.58) and (K.1.59); particularly selected from (K.1.41), (K.1.44), (K.1.47), (K.1.57), (K.1.58) and (K.1.59).
- the biopesticides from group L1) and/or L2) may also have insecticidal, acaricidal, molluscidal, pheromone, nematicidal, plant stress reducing, plant growth regulator, plant growth promoting and/or yield enhancing activity.
- the biopesticides from group L3) and/or L4) may also have fungicidal, bactericidal, viricidal, plant defense activator, plant stress reducing, plant growth regulator, plant growth promoting and/or yield enhancing activity.
- the biopesticides from group L5) may also have fungicidal, bactericidal, viricidal, plant defense activator, insecticidal, acaricidal, molluscidal, pheromone and/or nematicidal activity.
- the microbial pesticides in particular those from groups L1), L3) and L5), embrace not only the isolated, pure cultures of the respective microorganism as defined herein, but also its cell- free extract, its suspension in a whole broth culture and a metabolite-containing culture medium or a purified metabolite obtained from a whole broth culture of the microorganism.
- B. a. ssp. plantarum or B. velezensis FZB42 isolated from soil in Brandenburg, Germany (DSM 23117; J. Plant Dis. Prot. 105, 181-197, 1998; e. g. RhizoVital® 42 from AbiTEP GmbH, Germany), B. a. ssp. plantarum or B. velezensis MBI600 isolated from faba bean in Sutton Bonington, Nottinghamshire, U.K. at least before 1988 (also called 1430; NRRL B-50595;
- CNCM 1-1582 a variant of parental strain EIP-N1 (CNCM 1-1556) isolated from soil of central plain area of Israel (WO 2009/126473, US 6,406,690; e. g. Votivo® from Bayer CropScience LP, USA), B. pumilus GHA 25 isolated from apple tree rhizosphere in Mexico (IDAC 260707-01; e. g. PRO MIX® BX from Premier Horticulture, Quebec, Canada), B. pumilus INR-7 otherwise referred to as BU-F22 and BU-F33 isolated at least before 1993 from cucumber infested by Erwinia tracheiphila (NRRL B-50185, NRRL B-50153; US 8,445,255), B. pumilus KFP9F isolated from the rhizosphere of grasses in South Africa at least before 2008 (NRRL B-50754;
- B. pumilus QST 2808 was isolated from soil collected in Pohnpei, Federated States of Micronesia, in 1998 (NRRL B-30087; e. g. Sonata® or Ballad® Plus from Bayer Crop Science LP, USA), B. simplex ABU 288 (NRRL B-50304; US 8,445,255), B. subtilis FB17 also called UD 1022 or UD10-22 isolated from red beet roots in North America (ATCC PTA-11857; System. Appl. Microbiol.
- B. t. ssp. kurstaki SB4 isolated from E. saccharina larval cadavers (NRRL B-50753; e. g. Beta Pro® from BASF Agricultural Specialities (Pty) Ltd., South Africa), B. t. ssp. tenebrionis NB-176-1, a mutant of strain NB-125, a wild type strain isolated in 1982 from a dead pupa of the beetle Tenebrio molitor (DSM 5480; EP 585215 B1; e. g. Novodor® from Valent BioSciences, Switzerland), Beauveria bassiana GHA (ATCC 74250; e.
- B. bassiana JW-1 (ATCC 74040; e. g. Naturalis® from CBC (Europe) S.r.l., Italy)
- B. bassiana PPRI 5339 isolated from the larva of the tortoise beetle Conchyloctenia punctata (NRRL 50757; e. g.
- B. japonicum SEMIA 5080 obtained under lab condtions by Embrapa-Cerrados in Brazil and used in commercial inoculants since 1992, being a natural variant of SEMIA 586 (CB259) originally isolated in U.S.A. (CPAC 7; e. g. GELFIX 5 or ADHERE 60 from BASF Agricultural Specialties Ltd., Brazil); Burkholderia sp.
- HSSNPV single capsid nucleopolyhedrovirus
- ABA- NPV-U e. g. Heligen® from AgBiTech Pty Ltd., Queensland, Australia
- Heterorhabditis bacteriophora e. g.
- Met52® Novozymes Biologicals BioAg Group, Canada Metschnikowia fructicola 277 isolated from grapes in the central part of Israel (US 6,994,849; NRRL Y-30752; e. g. formerly Shemer® from Agrogreen, Israel), Paecilomyces ilacinus 251 isolated from infected nematode eggs in the Philippines (AGAL 89/030550; W01991/02051; Crop Protection 27, 352-361, 2008; e. g.
- PaenibacHlus alvei NAS6G6 isolated from the rhizosphere of grasses in South Africa at least before 2008 (WO 2014/029697; NRRL B-50755; e.g. BAC-UP from BASF Agricultural Specialities (Pty) Ltd., South Africa), PaenibacHlus strains isolated from soil samples from a variety of European locations including Germany: P. epiphyticus Lu17015 (WO 2016/020371; DSM 26971), P. polymyxa ssp. plantarum Lu16774 (WO 2016/020371; DSM 26969), P. p. ssp.
- the at least one pesticide II is selected from the groups L1) to L5):
- Microbial pesticides with fungicidal, bactericidal, viricidal and/or plant defense activator activity Aureobasidium pullulans DSM 14940 and DSM 14941 (L1.1), Bacillus amylolique- faciens A P-188 (L.1.2), B. amyloliquefaciens ssp. plantarum D747 (L.1.3), B. amylolique- faciens ssp. plantarum FZB24 (L.1.4), B. amyloliquefaciens ssp. plantarum FZB42 (L.1.5), B. amyloliquefaciens ssp. plantarum MBI600 (L.1.6), B.
- amyloliquefaciens ssp. plantarum GST-713 (L.1.7), B. amyloliquefaciens ssp. plantarum TJ1000 (L.1.8), B. pumilus GB34 (L.1.9), B. pumilus GHA 25 (L.1.10), B. pumilus INR-7 (L.1.11), B. pumilus KFP9F (L.1.12), B. pumilus CST 2808 (L.1.13), B. simplex ABU 288 (L.1.14), B.
- subtilis FB17 (L.1.15), Coniothyrium minitans CON/M/91-08 (L.1.16), Metschnikowia fructicola NRRL Y-30752 (L.1.17), Paenibacillus alvei NAS6G6 (L.1.18), P. epiphyticus Lu17015 (L.1.25), P. polymyxa ssp. plantarum Lu16774 (L.1.26), P. p. ssp. plantarum strain Lu17007 (L.1.27), Penicillium bilaiae ATCC 22348 (L.1.19), P.
- Microbial pesticides with insecticidal, acaricidal, molluscidal and/or nematicidal activity Bacillus firmus 1-1582 (L.3.1); B. thuringiensis ssp. aizawai ABTS-1857 (L.3.2), B. t. ssp. kurstaki ABTS-351 (L.3.3), B. t. ssp. kurstaki SB4 (L.3.4), B. t. ssp. tenebrionis NB-176-1 (L.3.5), Beauveria bassiana GHA (L.3.6), B. bassiana JW-1 (L.3.7), B.
- bassiana PPRI 5339 (L.3.8), Burkholderia sp. A396 (L.3.9), Helicoverpa armigera nucleopolyhedrovirus (HearNPV) (L.3.10), Helicoverpa zea nucleopolyhedrovirus (HzNPV) ABA-NPV-U (L.3.11), Helicoverpa zea single capsid nucleopolyhedrovirus (HzSNPV) (L.3.12), Heterohabditis bacteriophora (L.3.13), Isaria fumosorosea Apopka-97 (L.3.14), Metarhizium anisopliae var.
- HearNPV Helicoverpa armigera nucleopolyhedrovirus
- HzNPV Helicoverpa zea nucleopolyhedrovirus
- HzSNPV Helicoverpa zea single capsid nucleopolyhedrovirus
- anisopliae F52 (L.3.15), Paecilomyces lilacinus 251 (L.3.16), Pasteuria nishizawae Pn1 (L.3.17), Steinernema carpocapsae (L.3.18), S. feltiae (L.3.19);
- Microbial pesticides with plant stress reducing, plant growth regulator, plant growth promoting and/or yield enhancing activity Azospirillum brasilense Ab-V5 and Ab-V6 (L.5.1), A. brasilense Sp245 (L.5.2), Bradyrhizobium eikanii SEMIA 587 (L.5.3), B. eikanii SEMIA 5019 (L.5.4), B. japonicum 532c (L.5.5), B. japonicum E-109 (L.5.6), B. japonicum SEMIA 5079 (L.5.7), B. japonicum SEMIA 5080 (L.5.8).
- the present invention furthermore relates to agrochemical compositions comprising a mixture of at least one compound I (component 1) and at least one biopesticide selected from the group L) (component 2), in particular at least one biopesticide selected from the groups L1) and L2), as described above, and if desired at least one suitable auxiliary.
- the present invention furthermore relates to agrochemical compositions comprising a mixture of of at least one compound I (component 1) and at least one biopesticide selected from the group L) (component 2), in particular at least one biopesticide selected from the groups L3) and L4), as described above, and if desired at least one suitable auxiliary.
- mixtures comprising as pesticide II (component 2) a biopesticide selected from the groups L1), L3) and L5), preferably selected from strains denoted above as (L.1.2), (L.1.3), (L.1.4), (L.1.5), (L.1.6), (L.1.7), (L.1.8), (L.1.10), (L.1.11), (L.1.12), (L.1.13),
- mixtures comprising as pesticide II (component 2) a biopesticide selected from the groups L1), L3) and L5), preferably selected from strains denoted above as (L1.1), (L.1.2), (L.1.3), (L.1.6), (L.1.7), (L.1.9), (L.1.11), (L.1.12), (L.1.13), (L.1.14), (L.1.15), (L.1.17), (L.1.18), (L.1.22), (L.1.23), (L.1.24), (L.1.25), (L.1.26), (L.1.27), (L.2.2); (L.3.2), (L.3.3), (L.3.4), (L.3.5), (L.3.6), (L.3.7), (L.3.8), (L.3.10), (L.3.11), (L.3.12), (L.3.13), (L.3.14), (L.3.15), (L.3.18), (L.3.19); (L.4.2), even more preferably selected from (L1.1), (L
- compositions comprising mixtures of active ingredients can be prepared by usual means, e. g. by the means given for the compositions of compounds I.
- compositions When living microorganisms, such as pesticides II from groups L1), L3) and L5), form part of the compositions, such compositions can be prepared by usual means (e. g. H.D. Burges: For mulation of Microbial Biopesticides, Springer, 1998; WO 2008/002371 , US 6,955,912, US 5,422,107).
- 2,2,3-trimethylchroman-4-one oxime (66 g, 1 eq) was added to thionyl chloride (80, 3.5 eq) at a temperature below 30°C, and the reaction mixture was stirred at 50°C for 17 hours. After removal of thionyl chloride in vacuo the residue was poured into 1,4-dioxan (500 ml) and water (200 ml), and stirred for 1 h at 80°C. After removal of 1,4-dioxan in vacuo the resultant residue was extracted with ethyl acetate, washed successively with water and brine, and dried over anhydrous magnesium sulfate. Removal of solvent in vacuo the crude product was purified by flash chromatography on silica gel using heptane/MTBE as eluent to give the titled compound (25.4 g) as a with powder.
- Step 4 Preparation of 5-[6-(difluoromethoxy)-5-methyl-3-pyridyl]-2,2,3-trimethyl-3H-1,4- benzoxazepine [6-(difluoromethyl)-5-methyl-3-pyridyl]boronic acid (3.11 g, 1.2 eq), potassium carbonate (3.83 g, 2 eq), silver oxide (1.61 , 0.5 eq) and dichlorobis(triphenylphosphine)palladium(ll) (490 g, 0.05 eq) were added to a solution of 5-chloro-2,2,3-trimethyl-3H-1 ,4-benzoxazepine (3.1 g, 1 eq) in dry tetrahydrofuran (57 ml_), and the mixture was stirred under argon atmosphere at 80 °C for 18 hours.
- Table I Compounds Ex-1 to Ex-101 of formula I, wherein the meaning of R 2 , R 3 , R 5 , R 6 , R 7 , R 8 and Xn are as defined in each line.
- HPLC High Performance Liquid Chromatography; HPLC-column Kinetex XB C18 1,7m (50 x 2,1 mm); eluent: acetonitrile / water + 0.1% trifluoroacetic acid (gradient from 5:95 to 100 : 0 in 1.5 min at 60°C, flow gradient from 0.8 to 1.0 ml/min in 1.5 min).
- Rt retention time in minutes.
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- Example 1 Activity against the grey mold Botrytis cinerea in the microtiterplate test
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of Botrci cinerea in an aqueous biomalt or yeast-bactopeptone-sodiumacetate solution was then added.
- the plates were placed in a water vapor-saturated chamber at a temperature of 18°C. Using an absorption photometer, the MTPs were measured at 405 nm 7 days after the inoculation.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of Fusarium culmorum in an aqueous biomalt or yeast-bactopeptone-glycerine or DOB solution was then added.
- the plates were placed in a water vapor-saturated chamber at a temperature of 18°C. Using an absorption photometer, the MTPs were measured at 405 nm 7 days after the inoculation.
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of Botrci cinerea in an aqueous biomalt or yeast-bactopeptone-sodiumacetate solution was then added.
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of Botrci cinerea in an aqueous biomalt or yeast-bactopeptone-sodiumacetate solution was then added.
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of Fusarium culmorum in an aqueous biomalt yeast-bactopeptone-glycerine or DOB solution was then added.
- Example 6 Activity against the leaf blotch on wheat caused by Septoria tritici in the microtiterplate test
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of Septorion tritici in an aqueous biomalt or yeast-bactopeptone-glycerine or DOB solution was then added.
- Example 7 Activity against Microdochium nivale in the microtiterplate test
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of the Colletotrichum orbiculare isolates in a DOB media (ph 7) was then added.
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- Example 11 Activity against Monilinia laxa in the microtiterplate test
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- Example 15 Activity against Phytophthora infestans in the microtiterplate test
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of the Pyrenophora infenstans in a DOB media (ph 7) was then added.
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of the Mycosphorella fijiensis isolates in a DOB media (ph 7) was then added.
- Example 17 Activity against Corynespora cassiicola in the microtiterplate test
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- the measured parameters were compared to the growth of the active compound-free control variant (100%) and the fungus-free blank value to determine the relative growth in % of the pathogens in the respective active compounds.
- the compound was dissolved in a mixture of acetone and/or dimethylsulfoxide and the wetting agent/emulsifier Wettol, which is based on ethoxylated alkylphenoles, in a ratio (volume) solvent- emulsifier of 99 to 1 to give a total volume of 5 ml. Subsequently, water was added to total volume of 100 ml.
- Wettol which is based on ethoxylated alkylphenoles
- This stock solution was then diluted with the described solvent-emulsifier-water mixture to the final concentration given in the table below.
- Example 19 Preventative fungicidal control of Botrytis cinerea on leaves of green pepper
- Young seedlings of green pepper were grown in pots to the 4 to 5 leaf stage. These plants were sprayed to run-off with previously described spray solution, containing the concentration of active ingredient or mixture mentioned in the table below. The next day the plants were inoculated with an aqueous biomalt or DOB solution containing the spore suspension of Botrytis cinerea. Then the plants were immediately transferred to a humid chamber. After 5 days at 22 to 24°C and a saturated relative humidity, the extent of fungal attack on the leaves was visually assessed as % diseased leaf area.
- Example 20 Preventative fungicidal control of white mold on oilseed rape caused by Sclerotinia sclerotiorum
- Oilseed rapes were grown in pots to the 13 to 14 leaf stage. These plants were sprayed to run off with previously described spray solution, containing the concentration of active ingredient or their mixture mentioned in the table below. The plants could air-dry. The next day the applicated rape petals were fixed wit 25mI of 2.5% methylcellulose on leaf 1 and 2. 25 mI of a spore suspension of Sclerotinia sclerotiorum was pipetted on each fixed rape petal. After 14 days at 20°C and a relative humidity of 60 % the extent of fungal attack on the leaves was visually assessed as % diseased leaf area.
- Example 21 Preventative fungicidal control of Botrytis cinerea on leaves of green pepper
- Young seedlings of green pepper were grown in pots to the 4 to 5 leaf stage. These plants were sprayed to run-off with previously described spray solution, containing the concentration of active ingredient or mixture mentioned in the table below. The next day the plants were inoculated with an aqueous biomalt or DOB solution containing the spore suspension of Botrytis cinerea. Then the plants were immediately transferred to a humid chamber. After 5 days at 22 to 24°C and a saturated relative humidity, the extent of fungal attack on the leaves was visually assessed as % diseased leaf area.
- Young seedlings of green pepper were grown in pots to the 4 to 5 leaf stage. These plants were sprayed to run-off with previously described spray solution, containing the concentration of active ingredient or mixture mentioned in the table below. The plants were then cultivated in the greenhouse for 7 days and then inoculated with an aqueous biomalt or DOB solution containing the spore suspension of Botrytis cinerea. Then the plants were immediately transferred to a humid chamber. After 5 days at 22 to 24°C and a saturated relative humidity, the extent of fungal attack on the leaves was visually assessed as % diseased leaf area.
- Example 1 Activity against leaf blotch on wheat caused by Septoria tritici
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of Septoria tritici in an aqueous biomalt or yeast-bactopeptone-glycerine or DOB solution was then added.
- the plates were placed in a water vapor-saturated chamber at a temperature of 18°C. Using an absorption photometer, the MTPs were measured at 405 nm 7 days after the inoculation.
- Example 2 Activity against wheat leaf spots caused by Leptosphaeria nodorum
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide.
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of Leptosphaeria nodorum in an aqueous biomalt or yeast-bactopeptone-glycerine or DOB solution was then added.
- the plates were placed in a water vapor-saturated chamber at a temperature of 18°C. Using an absorption photometer, the MTPs were measured at 405 nm 7 days after the inoculation.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Environmental Sciences (AREA)
- Zoology (AREA)
- Engineering & Computer Science (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Wood Science & Technology (AREA)
- Agronomy & Crop Science (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Dentistry (AREA)
- Mycology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Microbiology (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Pyridine Compounds (AREA)
- Plural Heterocyclic Compounds (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
Abstract
Description
Claims
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| MX2023013721A MX2023013721A (en) | 2021-05-18 | 2022-05-10 | New substituted pyridines as fungicides. |
| US18/290,347 US20240270727A1 (en) | 2021-05-18 | 2022-05-10 | New substituted pyridines as fungicide |
| CA3218900A CA3218900A1 (en) | 2021-05-18 | 2022-05-10 | New substituted pyridines as fungicides |
| BR112023024005A BR112023024005A2 (en) | 2021-05-18 | 2022-05-10 | COMPOUNDS, COMPOSITION AND PROCESS FOR PREPARING COMPOUNDS OF FORMULA I |
| KR1020237039267A KR20240008856A (en) | 2021-05-18 | 2022-05-10 | Novel substituted pyridines as fungicides |
| JP2023571307A JP2024519815A (en) | 2021-05-18 | 2022-05-10 | New substituted pyridines as fungicides. |
| EP22728525.1A EP4341258B1 (en) | 2021-05-18 | 2022-05-10 | New substituted pyridines as fungicides |
| AU2022279357A AU2022279357A1 (en) | 2021-05-18 | 2022-05-10 | New substituted pyridines as fungicides |
| IL308529A IL308529A (en) | 2021-05-18 | 2022-05-10 | New substituted pyridines as fungicides |
| CN202280036430.1A CN117355519A (en) | 2021-05-18 | 2022-05-10 | New substituted pyridines for use as fungicides |
| CONC2023/0015529A CO2023015529A2 (en) | 2021-05-18 | 2023-11-17 | New substituted pyridines as fungicides |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP21174267 | 2021-05-18 | ||
| EP21174267.1 | 2021-05-18 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022243111A1 true WO2022243111A1 (en) | 2022-11-24 |
Family
ID=75977623
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2022/062622 Ceased WO2022243111A1 (en) | 2021-05-18 | 2022-05-10 | New substituted pyridines as fungicides |
Country Status (14)
| Country | Link |
|---|---|
| US (1) | US20240270727A1 (en) |
| EP (1) | EP4341258B1 (en) |
| JP (1) | JP2024519815A (en) |
| KR (1) | KR20240008856A (en) |
| CN (1) | CN117355519A (en) |
| AR (1) | AR125907A1 (en) |
| AU (1) | AU2022279357A1 (en) |
| BR (1) | BR112023024005A2 (en) |
| CA (1) | CA3218900A1 (en) |
| CL (1) | CL2023003418A1 (en) |
| CO (1) | CO2023015529A2 (en) |
| IL (1) | IL308529A (en) |
| MX (1) | MX2023013721A (en) |
| WO (1) | WO2022243111A1 (en) |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024104814A1 (en) * | 2022-11-16 | 2024-05-23 | Basf Se | Fungicidal mixture comprising substituted pyridines |
| WO2024165343A1 (en) * | 2023-02-08 | 2024-08-15 | Basf Se | New substituted quinoline compounds for combatitng phytopathogenic fungi |
| WO2024194038A1 (en) * | 2023-03-17 | 2024-09-26 | Basf Se | Substituted pyridyl/pyrazidyl dihydrobenzothiazepine compounds for combatting phytopathogenic fungi |
| WO2025031842A1 (en) | 2023-08-09 | 2025-02-13 | Basf Se | New substituted benzoxazepine picolinonitrile compounds for combatting phytopathogenic fungi |
| WO2025180964A1 (en) | 2024-03-01 | 2025-09-04 | Basf Se | New substituted benzoxazepine compounds for combatting phytopathogenic fungi |
Citations (150)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0141317A2 (en) | 1983-10-21 | 1985-05-15 | BASF Aktiengesellschaft | 7-Amino-azolo[1,5-a]pyrimidines and fungicides containing them |
| EP0152031A2 (en) | 1984-02-03 | 1985-08-21 | Shionogi & Co., Ltd. | Azolyl cycloalkanol derivatives and agricultural fungicides |
| EP0226917A1 (en) | 1985-12-20 | 1987-07-01 | BASF Aktiengesellschaft | Acrylic acid esters and fungicides containing these compounds |
| EP0243970A1 (en) | 1986-05-02 | 1987-11-04 | Stauffer Chemical Company | Fungicidal pyridyl imidates |
| EP0256503A2 (en) | 1986-08-12 | 1988-02-24 | Mitsubishi Kasei Corporation | Pyridinecarboxamide derivatives and their use as fungicide |
| EP0307510B1 (en) | 1987-09-17 | 1991-02-06 | BASF Aktiengesellschaft | Process for combating fungicides |
| WO1991002051A1 (en) | 1989-08-03 | 1991-02-21 | The Australian Technological Innovation Corporation | Myconematicide |
| EP0428941A1 (en) | 1989-11-10 | 1991-05-29 | Agro-Kanesho Co., Ltd. | Hexahydrotriazine compounds and insecticides |
| US5026417A (en) | 1987-03-17 | 1991-06-25 | Her Majesty The Queen In Right Of Canada, As Represented By The Minister Of Agriculture | Methods and compositions for increasing the amounts of phosphorus and/or micronutrients available for plant uptake from soils |
| EP0532022A1 (en) | 1991-09-13 | 1993-03-17 | Ube Industries, Ltd. | Acrylate compound, preparation process thereof and fungicide using the same |
| WO1994001546A1 (en) | 1992-07-01 | 1994-01-20 | Cornell Research Foundation, Inc. | Elicitor of the hypersensitive response in plants |
| US5422107A (en) | 1992-12-25 | 1995-06-06 | Hokkaido Green Kosan, Incorporated | Trichoderma harzianum SK-55 fungus, fungicide containing it, and method of manufacture of the same and its use |
| WO1995017806A1 (en) | 1993-12-29 | 1995-07-06 | Philom Bios Inc. | Methods and compositions for increasing the benefits of rhizobium inoculation to legume crop productivity |
| WO1996021358A1 (en) | 1995-01-14 | 1996-07-18 | Prophyta Biologischer Pflanzenschutz Gmbh | Fungus isolate, preparation for combatting plant-pathogenic fungi, process for producing it and its use |
| WO1998044140A1 (en) | 1997-04-03 | 1998-10-08 | Dekalb Genetics Corporation | Glyphosate resistant maize lines |
| EP0585215B1 (en) | 1989-11-17 | 1999-09-15 | Abbott Laboratories | Mutants or variants of bacillus thuringiensis producing high yields of delta endotoxin |
| WO2000026345A1 (en) | 1998-11-03 | 2000-05-11 | Aventis Cropscience N.V. | Glufosinate tolerant rice |
| WO2000026356A1 (en) | 1998-11-03 | 2000-05-11 | Aventis Cropscience N. V. | Glufosinate tolerant rice |
| EP1028125A1 (en) | 1998-11-30 | 2000-08-16 | Isagro Ricerca S.r.l. | Dipeptide compounds having fungicidal activity and their agronomic use |
| EP1035122A1 (en) | 1999-03-11 | 2000-09-13 | Rohm And Haas Company | Heterocyclic subsituted isoxazolidines and their use as fungicides |
| WO2001031042A2 (en) | 1999-10-29 | 2001-05-03 | Aventis Cropscience N.V. | Male-sterile brassica plants and methods for producing same |
| WO2001041558A1 (en) | 1999-12-08 | 2001-06-14 | Aventis Cropscience N.V. | Hybrid winter oilseed rape and methods for producing same |
| EP1122244A1 (en) | 2000-02-04 | 2001-08-08 | Sumitomo Chemical Company, Limited | Uracil compounds and their use |
| CN1309897A (en) | 2000-02-24 | 2001-08-29 | 沈阳化工研究院 | Unsaturated oximino ether bactericide |
| EP1201648A1 (en) | 1999-08-05 | 2002-05-02 | Kumiai Chemical Industry Co., Ltd. | Carbamate derivatives and agricultural/horticultural bactericides |
| WO2002034946A2 (en) | 2000-10-25 | 2002-05-02 | Monsanto Technology Llc | Cotton event pv-ghgt07(1445) and compositions and methods for detection thereof |
| WO2002036831A2 (en) | 2000-10-30 | 2002-05-10 | Monsanto Technology Llc | Canola event pv-bngt04(rt73) and compositions and methods for detection thereof |
| US6406690B1 (en) | 1995-04-17 | 2002-06-18 | Minrav Industries Ltd. | Bacillus firmus CNCM I-1582 or Bacillus cereus CNCM I-1562 for controlling nematodes |
| US20020102582A1 (en) | 2000-09-13 | 2002-08-01 | Levine Elaine B. | Corn event MON810 and compositions and methods for detection thereof |
| WO2002100163A2 (en) | 2001-06-11 | 2002-12-19 | Monsanto Technology Llc | Cotton event moni5985 and compositions and methods for detection |
| WO2003013224A2 (en) | 2001-08-06 | 2003-02-20 | Bayer Bioscience N.V. | Herbicide tolerant cotton plants and methods for producing and identifying same |
| WO2003016303A1 (en) | 2001-08-20 | 2003-02-27 | Dainippon Ink And Chemicals, Inc. | Tetrazoyl oxime derivative and agricultural chemical containing the same as active ingredient |
| US20030126634A1 (en) | 1990-08-09 | 2003-07-03 | Dekalb Genetics Corporation | Methods and compositions for the increase of yield in plants |
| WO2003064572A1 (en) | 2002-01-31 | 2003-08-07 | Exxonmobil Research And Engineering Company | Lubricating oil compositions with improved friction properties |
| WO2003066609A1 (en) | 2002-02-04 | 2003-08-14 | Bayer Cropscience Aktiengesellschaft | Disubstituted thiazolyl carboxanilides and their use as microbicides |
| WO2003074491A1 (en) | 2002-03-05 | 2003-09-12 | Syngenta Participations Ag | O-cyclopropyl-carboxanilides and their use as fungicides |
| CN1456054A (en) | 2003-03-25 | 2003-11-19 | 浙江省化工研究院 | Methoxy methyl acrylate compounds as bactericidal agent |
| WO2004011601A2 (en) | 2002-07-29 | 2004-02-05 | Monsanto Technology, Llc | Corn event pv-zmir13 (mon863) plants and compositions and methods for detection thereof |
| WO2004039986A1 (en) | 2002-10-29 | 2004-05-13 | Syngenta Participations Ag | Cot102 insecticidal cotton |
| WO2004049804A2 (en) | 2002-11-29 | 2004-06-17 | Syngenta Participations Ag | Fungicidal combinations for crop potection |
| WO2004072235A2 (en) | 2003-02-12 | 2004-08-26 | Monsanto Technology Llc | Cotton event mon 88913 and compositions and methods for detection thereof |
| WO2004074492A1 (en) | 2003-02-20 | 2004-09-02 | Kws Saat Ag | Glyphosate tolerant sugar beet |
| WO2004083193A1 (en) | 2003-03-17 | 2004-09-30 | Sumitomo Chemical Company, Limited | Amide compound and bactericide composition containing the same |
| WO2004099447A2 (en) | 2003-05-02 | 2004-11-18 | Dow Agrosciences Llc | Corn event tc1507 and methods for detection thereof |
| WO2005059103A2 (en) | 2003-12-15 | 2005-06-30 | Monsanto Technology Llc | Corn plant mon88017 and compositions and methods for detection thereof |
| WO2005061720A2 (en) | 2003-12-11 | 2005-07-07 | Monsanto Technology Llc | High lysine maize compositions and methods for detection thereof |
| WO2005063721A1 (en) | 2003-12-19 | 2005-07-14 | E.I. Dupont De Nemours And Company | Herbicidal pyrimidines |
| WO2005087773A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| WO2005087772A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| US6955912B2 (en) | 2000-03-31 | 2005-10-18 | Yasuharu Sasaki | Process for producing Trichoderma harzianum ferm BP-4346 |
| WO2005103266A1 (en) | 2004-03-26 | 2005-11-03 | Dow Agrosciences Llc | Cry1f and cry1ac transgenic cotton lines and event-specific identification thereof |
| WO2005103301A2 (en) | 2004-03-25 | 2005-11-03 | Syngenta Participations Ag | Corn event mir604 |
| CA2471555A1 (en) | 2004-06-18 | 2005-12-18 | Thomas D. Johnson | Controlling plant pathogens with fungal/bacterial antagonist combinations comprising trichoderma virens and bacillus amyloliquefaciens |
| WO2005120234A2 (en) | 2004-06-03 | 2005-12-22 | E.I. Dupont De Nemours And Company | Fungicidal mixtures of amidinylphenyl compounds |
| WO2005123690A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-difluoromethyl-pyrazol-4-carbonic acid-(ortho-phenyl)-anilides, and use thereof as a fungicide |
| WO2005123689A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-trifluoromethyl-pyrazole-4-carboxylic acid (ortho-phenyl)-anilides and to use thereof as fungicide |
| US6994849B2 (en) | 2001-03-14 | 2006-02-07 | State Of Israel, Ministry Of Agriculture, Agricultural Research Organization | Yeast Metschnikowia fructicola NRRL Y-30752 for inhibiting deleterious microorganisms on plants |
| WO2006015866A1 (en) | 2004-08-12 | 2006-02-16 | Syngenta Participations Ag | Method for protecting useful plants or plant propagation material |
| WO2006039376A2 (en) | 2004-09-29 | 2006-04-13 | Pioneer Hi-Bred International, Inc. | Corn event das-59122-7 and methods for detection thereof |
| WO2006087325A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | 5-alkoxyalkyl-6-alkyl-7-amino-azolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said substances |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| WO2006098952A2 (en) | 2005-03-16 | 2006-09-21 | Syngenta Participations Ag | Corn event 3272 and methods of detection thereof |
| WO2006108675A2 (en) | 2005-04-11 | 2006-10-19 | Bayer Bioscience N.V. | Elite event a5547-127 and methods and kits for identifying such event in biological samples |
| WO2006108674A2 (en) | 2005-04-08 | 2006-10-19 | Bayer Bioscience N.V. | Elite event a2704-12 and methods and kits for identifying such event in biological samples |
| WO2006128573A2 (en) | 2005-06-02 | 2006-12-07 | Syngenta Participations Ag | Ce43- 67b, insecticidal transgenic cotton expressing cry1ab |
| WO2006130436A2 (en) | 2005-05-27 | 2006-12-07 | Monsanto Technology Llc | Soybean event mon89788 and methods for detection thereof |
| WO2007006670A1 (en) | 2005-07-07 | 2007-01-18 | Basf Aktiengesellschaft | N-thio-anthranilamid compounds and their use as pesticides |
| CN1907024A (en) | 2005-08-03 | 2007-02-07 | 浙江化工科技集团有限公司 | Methoxyl group displacement methyl acrylate compound bactericidal agent |
| WO2007017186A1 (en) | 2005-08-08 | 2007-02-15 | Bayer Bioscience N.V. | Herbicide tolerant cotton plants and methods for identifying same |
| WO2007082098A2 (en) | 2006-01-13 | 2007-07-19 | Dow Agrosciences Llc | 6-(poly-substituted aryl)-4-aminopicolinates and their use as herbicides |
| WO2007090624A2 (en) | 2006-02-09 | 2007-08-16 | Syngenta Participations Ag | A method of protecting a plant propagation material, a plant, and/or plant organs |
| WO2007129454A1 (en) | 2006-05-08 | 2007-11-15 | Kumiai Chemical Industry Co., Ltd. | 1,2-benzisothiazole derivative, and agricultural or horticultural plant disease-controlling agent |
| WO2007140256A1 (en) | 2006-05-26 | 2007-12-06 | Monsanto Technology, Llc | Corn plant and seed corresponding to transgenic event mon89034 and methods for detection and use thereof |
| WO2007142840A2 (en) | 2006-06-03 | 2007-12-13 | Syngenta Participations Ag | Corn event mir162 |
| US20070292854A1 (en) | 2000-06-22 | 2007-12-20 | Behr Carl F | Corn event PV-ZMGT32(nk603) and compositions and methods for detection thereof |
| WO2008002371A1 (en) | 2006-06-23 | 2008-01-03 | Becker Underwood Inc. | Improved shelf life and on seed stabilization of liquid bacterium inoculants |
| WO2008002872A2 (en) | 2006-06-28 | 2008-01-03 | Pioneer Hi-Bred International, Inc. | Soybean event 3560.4.3.5 and compositions and methods for the identification and/or detection thereof |
| WO2008013622A2 (en) | 2006-07-27 | 2008-01-31 | E. I. Du Pont De Nemours And Company | Fungicidal azocyclic amides |
| WO2008054747A2 (en) | 2006-10-31 | 2008-05-08 | E. I. Du Pont De Nemours And Company | Soybean event dp-305423-1 and compositions and methods for the identification and/or detection thereof |
| WO2008112019A2 (en) | 2006-10-30 | 2008-09-18 | Pioneer Hi-Bred International, Inc. | Maize event dp-098140-6 and compositions and methods for the identification and/or detection thereof |
| WO2008122406A1 (en) | 2007-04-05 | 2008-10-16 | Bayer Bioscience N.V. | Insect resistant cotton plants and methods for identifying same |
| WO2008151780A1 (en) | 2007-06-11 | 2008-12-18 | Bayer Bioscience N.V. | Insect resistant cotton plants comprising elite event ee-gh6 and methods for identifying same |
| WO2009064652A1 (en) | 2007-11-15 | 2009-05-22 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87701 and methods for detection thereof |
| WO2009090181A2 (en) | 2008-01-15 | 2009-07-23 | Bayer Cropscience Sa | Pesticide composition comprising a tetrazolyloxime derivative and a fungicide or an insecticide active substance |
| WO2009094442A2 (en) | 2008-01-22 | 2009-07-30 | Dow Agrosciences Llc | 5-fluoro pyrimidine derivatives |
| WO2009102873A1 (en) | 2008-02-15 | 2009-08-20 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87769 and methods for detection thereof |
| WO2009103049A2 (en) | 2008-02-14 | 2009-08-20 | Pioneer Hi-Bred International, Inc. | Plant genomic dna flanking spt event and methods for identifying spt event |
| WO2009111263A1 (en) | 2008-02-29 | 2009-09-11 | Monsanto Technology Llc | Corn plant event mon87460 and compositions and methods for detection thereof |
| WO2009119089A1 (en) | 2008-03-27 | 2009-10-01 | 日本曹達株式会社 | Nitrogen-containing heterocyclic compound and bactericide for agricultural and horticultural use |
| WO2009126473A1 (en) | 2008-04-07 | 2009-10-15 | Bayer Cropscience Lp | Stable aqueous spore-containing formulation |
| WO2010018686A1 (en) | 2008-08-12 | 2010-02-18 | 日本曹達株式会社 | Nitrogenated heterocyclic derivative, and agricultural or horticultural bactericidal agent |
| WO2010037016A1 (en) | 2008-09-29 | 2010-04-01 | Monsanto Technology Llc | Soybean transgenic event mon87705 and methods for detection thereof |
| WO2010069882A1 (en) | 2008-12-17 | 2010-06-24 | Syngenta Participations Ag | Isoxazole derivatives for use as fungicides |
| WO2010077816A1 (en) | 2008-12-16 | 2010-07-08 | Syngenta Participations Ag | Corn event 5307 |
| WO2010080829A1 (en) | 2009-01-07 | 2010-07-15 | Basf Agrochemical Products B.V. | Soybean event 127 and methods related thereto |
| US20100260735A1 (en) | 2009-04-13 | 2010-10-14 | University of Delawre | Methods for promoting plant health |
| WO2010139271A1 (en) | 2009-06-05 | 2010-12-09 | 中国中化股份有限公司 | E-type phenyl acrylic ester compounds containing substituted anilino pyrimidine group and uses thereof |
| WO2011022469A2 (en) | 2009-08-19 | 2011-02-24 | Dow Agrosciences Llc | Aad-1 event das-40278-9, related transgenic corn lines, and event-specific identification thereof |
| WO2011028657A1 (en) | 2009-09-01 | 2011-03-10 | Dow Agrosciences Llc | Synergistic fungicidal compositions containing a 5-fluoropyrimidine derivative for fungal control in cereals |
| WO2011034704A1 (en) | 2009-09-17 | 2011-03-24 | Monsanto Technology Llc | Soybean transgenic event mon 87708 and methods of use thereof |
| WO2011062904A1 (en) | 2009-11-23 | 2011-05-26 | Monsanto Technology Llc | Transgenic maize event mon 87427 and the relative development scale |
| WO2011066384A1 (en) | 2009-11-24 | 2011-06-03 | Dow Agrosciences Llc | Aad-12 event 416, related transgenic soybean lines, and event-specific identification thereof |
| WO2011077514A1 (en) | 2009-12-22 | 2011-06-30 | 三井化学アグロ株式会社 | Plant disease control composition and method for controlling plant diseases by applying the composition |
| WO2011081174A1 (en) | 2010-01-04 | 2011-07-07 | 日本曹達株式会社 | Nitrogen-containing heterocyclic compound and agricultural/horticultural germicide |
| WO2011084621A1 (en) | 2009-12-17 | 2011-07-14 | Pioneer Hi-Bred International, Inc. | Maize event dp-004114-3 and methods for detection thereof |
| WO2011109395A2 (en) | 2010-03-01 | 2011-09-09 | University Of Delaware | Compositions and methods for increasing biomass, iron concentration, and tolerance to pathogens in plants |
| WO2011135833A1 (en) | 2010-04-28 | 2011-11-03 | Sumitomo Chemical Company, Limited | Plant disease control composition and its use |
| WO2011153186A1 (en) | 2010-06-04 | 2011-12-08 | Monsanto Technology Llc | Transgenic brassica event mon 88302 and methods of use thereof |
| WO2012051199A2 (en) | 2010-10-12 | 2012-04-19 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87712 and methods for detection thereof |
| US20120149571A1 (en) | 2010-12-10 | 2012-06-14 | Auburn University | Inoculants Including Bacillus Bacteria for Inducing Production of Volatile Organic Compounds in Plants |
| WO2012082548A2 (en) | 2010-12-15 | 2012-06-21 | Syngenta Participations Ag | Soybean event syht0h2 and compositions and methods for detection thereof |
| WO2012084812A1 (en) | 2010-12-20 | 2012-06-28 | Isagro Ricerca S.R.L. | Aminoindanes amides having a high fungicidal activity and their phytosanitary compositions |
| WO2012134808A1 (en) | 2011-03-30 | 2012-10-04 | Monsanto Technology Llc | Cotton transgenic event mon 88701 and methods of use thereof |
| WO2012165511A1 (en) | 2011-05-31 | 2012-12-06 | クミアイ化学工業株式会社 | Method for controlling diseases in rice plant |
| WO2012168188A1 (en) | 2011-06-07 | 2012-12-13 | Bayer Intellectual Property Gmbh | Active compound combinations |
| WO2013003558A1 (en) | 2011-06-30 | 2013-01-03 | Monsanto Technology Llc | Alfalfa plant and seed corresponding to transgenic event kk 179-2 and methods for detection thereof |
| WO2013007767A1 (en) | 2011-07-13 | 2013-01-17 | Basf Se | Fungicidal substituted 2-[2-halogenalkyl-4-(phenoxy)-phenyl]-1-[1,2,4]triazol-1-yl-ethanol compounds |
| WO2013010862A1 (en) | 2011-07-15 | 2013-01-24 | Basf Se | Fungicidal alkyl-substituted 2-[2-chloro-4-(4-chloro-phenoxy)-phenyl]-1-[1,2,4]triazol-1-yl-ethanol compounds |
| WO2013016527A1 (en) | 2011-07-26 | 2013-01-31 | Dow Agrosciences Llc | Insect resistant and herbicide tolerant soybean event 9582.814.19.1 |
| WO2013024010A1 (en) | 2011-08-12 | 2013-02-21 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2013024009A1 (en) | 2011-08-12 | 2013-02-21 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2013032693A2 (en) | 2011-08-27 | 2013-03-07 | Marrone Bio Innovations, Inc. | Isolated bacterial strain of the genus burkholderia and pesticidal metabolites therefrom-formulations and uses |
| WO2013047749A1 (en) | 2011-09-29 | 2013-04-04 | 三井化学アグロ株式会社 | Production method for 4, 4-difluoro-3,4-dihydroisoquinoline derivative |
| WO2013047441A1 (en) | 2011-09-26 | 2013-04-04 | 日本曹達株式会社 | Agricultural and horticultural bactericide composition |
| WO2013092224A1 (en) | 2011-12-21 | 2013-06-27 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi resistant to qo inhibitors |
| WO2013112527A1 (en) | 2012-01-23 | 2013-08-01 | Dow Agrosciences Llc | Herbicide tolerant cotton event pdab4468.19.10.3 |
| WO2013116251A2 (en) | 2012-02-01 | 2013-08-08 | E. I. Du Pont De Nemours And Company | Fungicidal pyrazole mixtures |
| WO2013127704A1 (en) | 2012-02-27 | 2013-09-06 | Bayer Intellectual Property Gmbh | Active compound combinations containing a thiazoylisoxazoline and a fungicide |
| US20130236522A1 (en) | 2010-11-10 | 2013-09-12 | Kumiai Chemical Industry Co., Ltd. | Microbial pesticidal composition |
| WO2013162072A1 (en) | 2012-04-27 | 2013-10-31 | Sumitomo Chemical Company, Limited | Tetrazolinone compounds and its use as pesticides |
| CN103387541A (en) | 2012-05-10 | 2013-11-13 | 中国中化股份有限公司 | Preparation method of substituted pyrazolylether compound |
| WO2013169923A2 (en) | 2012-05-08 | 2013-11-14 | Monsanto Technology Llc | Corn event mon 87411 |
| WO2014029697A1 (en) | 2012-08-22 | 2014-02-27 | Basf Se | Fungicidal ternary mixtures comprising fluazinam |
| WO2014060177A1 (en) | 2012-10-16 | 2014-04-24 | Syngenta Participations Ag | Fungicidal compositions |
| WO2014116854A1 (en) | 2013-01-25 | 2014-07-31 | Pioneer Hi-Bred International, Inc. | Maize event dp-033121-3 and methods for detection thereof |
| WO2014124369A1 (en) | 2013-02-11 | 2014-08-14 | Bayer Cropscience Lp | Compositions comprising a streptomyces-based biological control agent and a fungicide |
| WO2014178913A1 (en) | 2013-05-02 | 2014-11-06 | J.R. Simplot Company | Potato cultivar f10 |
| WO2014201235A2 (en) | 2013-06-14 | 2014-12-18 | Monsanto Technology Llc | Soybean transgenic event mon87751 and methods for detection and use thereof |
| WO2015053998A1 (en) | 2013-10-09 | 2015-04-16 | Monsanto Technology Llc | Transgenic corn event mon87403 and methods for detection thereof |
| EP2865265A1 (en) | 2014-02-13 | 2015-04-29 | Bayer CropScience AG | Active compound combinations comprising phenylamidine compounds and biological control agents |
| WO2015065922A1 (en) | 2013-10-28 | 2015-05-07 | Dexcom, Inc. | Devices used in connection with continuous analyte monitoring that provide the user with one or more notifications, and related methods |
| WO2015142571A1 (en) | 2014-03-20 | 2015-09-24 | Monsanto Technology Llc | Transgenic maize event mon 87419 and methods of use thereof |
| WO2016020371A1 (en) | 2014-08-04 | 2016-02-11 | Basf Se | Antifungal paenibacillus strains, fusaricidin-type compounds, and their use |
| WO2016156241A1 (en) | 2015-04-02 | 2016-10-06 | Syngenta Participations Ag | Herbicidal mixtures |
| WO2016162265A1 (en) | 2015-04-07 | 2016-10-13 | Syngenta Participations Ag | Herbicidal mixtures |
| WO2016183445A1 (en) | 2015-05-14 | 2016-11-17 | J.R. Simplot Company | Potato cultivar v11 |
| WO2017062825A1 (en) | 2015-10-08 | 2017-04-13 | J.R. Simplot Company | Potato cultivar y9 |
| WO2017062831A1 (en) | 2015-10-08 | 2017-04-13 | J.R. Simplot Company | Potato cultivar x17 |
| WO2018177894A1 (en) | 2017-03-31 | 2018-10-04 | Syngenta Participations Ag | Fungicidal compositions |
| WO2020212513A1 (en) | 2019-04-18 | 2020-10-22 | Syngenta Crop Protection Ag | Process for the preparation of microbiocidal oxadiazole derivatives |
-
2022
- 2022-05-10 US US18/290,347 patent/US20240270727A1/en active Pending
- 2022-05-10 AU AU2022279357A patent/AU2022279357A1/en active Pending
- 2022-05-10 KR KR1020237039267A patent/KR20240008856A/en active Pending
- 2022-05-10 MX MX2023013721A patent/MX2023013721A/en unknown
- 2022-05-10 CA CA3218900A patent/CA3218900A1/en active Pending
- 2022-05-10 CN CN202280036430.1A patent/CN117355519A/en active Pending
- 2022-05-10 JP JP2023571307A patent/JP2024519815A/en active Pending
- 2022-05-10 BR BR112023024005A patent/BR112023024005A2/en unknown
- 2022-05-10 EP EP22728525.1A patent/EP4341258B1/en active Active
- 2022-05-10 IL IL308529A patent/IL308529A/en unknown
- 2022-05-10 WO PCT/EP2022/062622 patent/WO2022243111A1/en not_active Ceased
- 2022-05-17 AR ARP220101321A patent/AR125907A1/en unknown
-
2023
- 2023-11-16 CL CL2023003418A patent/CL2023003418A1/en unknown
- 2023-11-17 CO CONC2023/0015529A patent/CO2023015529A2/en unknown
Patent Citations (158)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0141317A2 (en) | 1983-10-21 | 1985-05-15 | BASF Aktiengesellschaft | 7-Amino-azolo[1,5-a]pyrimidines and fungicides containing them |
| EP0152031A2 (en) | 1984-02-03 | 1985-08-21 | Shionogi & Co., Ltd. | Azolyl cycloalkanol derivatives and agricultural fungicides |
| EP0226917A1 (en) | 1985-12-20 | 1987-07-01 | BASF Aktiengesellschaft | Acrylic acid esters and fungicides containing these compounds |
| EP0243970A1 (en) | 1986-05-02 | 1987-11-04 | Stauffer Chemical Company | Fungicidal pyridyl imidates |
| EP0256503A2 (en) | 1986-08-12 | 1988-02-24 | Mitsubishi Kasei Corporation | Pyridinecarboxamide derivatives and their use as fungicide |
| US5026417A (en) | 1987-03-17 | 1991-06-25 | Her Majesty The Queen In Right Of Canada, As Represented By The Minister Of Agriculture | Methods and compositions for increasing the amounts of phosphorus and/or micronutrients available for plant uptake from soils |
| EP0307510B1 (en) | 1987-09-17 | 1991-02-06 | BASF Aktiengesellschaft | Process for combating fungicides |
| WO1991002051A1 (en) | 1989-08-03 | 1991-02-21 | The Australian Technological Innovation Corporation | Myconematicide |
| EP0428941A1 (en) | 1989-11-10 | 1991-05-29 | Agro-Kanesho Co., Ltd. | Hexahydrotriazine compounds and insecticides |
| EP0585215B1 (en) | 1989-11-17 | 1999-09-15 | Abbott Laboratories | Mutants or variants of bacillus thuringiensis producing high yields of delta endotoxin |
| US20030126634A1 (en) | 1990-08-09 | 2003-07-03 | Dekalb Genetics Corporation | Methods and compositions for the increase of yield in plants |
| EP0532022A1 (en) | 1991-09-13 | 1993-03-17 | Ube Industries, Ltd. | Acrylate compound, preparation process thereof and fungicide using the same |
| WO1994001546A1 (en) | 1992-07-01 | 1994-01-20 | Cornell Research Foundation, Inc. | Elicitor of the hypersensitive response in plants |
| US5422107A (en) | 1992-12-25 | 1995-06-06 | Hokkaido Green Kosan, Incorporated | Trichoderma harzianum SK-55 fungus, fungicide containing it, and method of manufacture of the same and its use |
| WO1995017806A1 (en) | 1993-12-29 | 1995-07-06 | Philom Bios Inc. | Methods and compositions for increasing the benefits of rhizobium inoculation to legume crop productivity |
| WO1996021358A1 (en) | 1995-01-14 | 1996-07-18 | Prophyta Biologischer Pflanzenschutz Gmbh | Fungus isolate, preparation for combatting plant-pathogenic fungi, process for producing it and its use |
| US6406690B1 (en) | 1995-04-17 | 2002-06-18 | Minrav Industries Ltd. | Bacillus firmus CNCM I-1582 or Bacillus cereus CNCM I-1562 for controlling nematodes |
| WO1998044140A1 (en) | 1997-04-03 | 1998-10-08 | Dekalb Genetics Corporation | Glyphosate resistant maize lines |
| WO2000026345A1 (en) | 1998-11-03 | 2000-05-11 | Aventis Cropscience N.V. | Glufosinate tolerant rice |
| WO2000026356A1 (en) | 1998-11-03 | 2000-05-11 | Aventis Cropscience N. V. | Glufosinate tolerant rice |
| EP1028125A1 (en) | 1998-11-30 | 2000-08-16 | Isagro Ricerca S.r.l. | Dipeptide compounds having fungicidal activity and their agronomic use |
| EP1035122A1 (en) | 1999-03-11 | 2000-09-13 | Rohm And Haas Company | Heterocyclic subsituted isoxazolidines and their use as fungicides |
| EP1201648A1 (en) | 1999-08-05 | 2002-05-02 | Kumiai Chemical Industry Co., Ltd. | Carbamate derivatives and agricultural/horticultural bactericides |
| WO2001031042A2 (en) | 1999-10-29 | 2001-05-03 | Aventis Cropscience N.V. | Male-sterile brassica plants and methods for producing same |
| WO2001041558A1 (en) | 1999-12-08 | 2001-06-14 | Aventis Cropscience N.V. | Hybrid winter oilseed rape and methods for producing same |
| EP1122244A1 (en) | 2000-02-04 | 2001-08-08 | Sumitomo Chemical Company, Limited | Uracil compounds and their use |
| CN1309897A (en) | 2000-02-24 | 2001-08-29 | 沈阳化工研究院 | Unsaturated oximino ether bactericide |
| US6955912B2 (en) | 2000-03-31 | 2005-10-18 | Yasuharu Sasaki | Process for producing Trichoderma harzianum ferm BP-4346 |
| US20070292854A1 (en) | 2000-06-22 | 2007-12-20 | Behr Carl F | Corn event PV-ZMGT32(nk603) and compositions and methods for detection thereof |
| US20020102582A1 (en) | 2000-09-13 | 2002-08-01 | Levine Elaine B. | Corn event MON810 and compositions and methods for detection thereof |
| WO2002034946A2 (en) | 2000-10-25 | 2002-05-02 | Monsanto Technology Llc | Cotton event pv-ghgt07(1445) and compositions and methods for detection thereof |
| WO2002036831A2 (en) | 2000-10-30 | 2002-05-10 | Monsanto Technology Llc | Canola event pv-bngt04(rt73) and compositions and methods for detection thereof |
| US6994849B2 (en) | 2001-03-14 | 2006-02-07 | State Of Israel, Ministry Of Agriculture, Agricultural Research Organization | Yeast Metschnikowia fructicola NRRL Y-30752 for inhibiting deleterious microorganisms on plants |
| WO2002100163A2 (en) | 2001-06-11 | 2002-12-19 | Monsanto Technology Llc | Cotton event moni5985 and compositions and methods for detection |
| WO2003013224A2 (en) | 2001-08-06 | 2003-02-20 | Bayer Bioscience N.V. | Herbicide tolerant cotton plants and methods for producing and identifying same |
| WO2003016303A1 (en) | 2001-08-20 | 2003-02-27 | Dainippon Ink And Chemicals, Inc. | Tetrazoyl oxime derivative and agricultural chemical containing the same as active ingredient |
| WO2003064572A1 (en) | 2002-01-31 | 2003-08-07 | Exxonmobil Research And Engineering Company | Lubricating oil compositions with improved friction properties |
| WO2003066609A1 (en) | 2002-02-04 | 2003-08-14 | Bayer Cropscience Aktiengesellschaft | Disubstituted thiazolyl carboxanilides and their use as microbicides |
| WO2003074491A1 (en) | 2002-03-05 | 2003-09-12 | Syngenta Participations Ag | O-cyclopropyl-carboxanilides and their use as fungicides |
| WO2004011601A2 (en) | 2002-07-29 | 2004-02-05 | Monsanto Technology, Llc | Corn event pv-zmir13 (mon863) plants and compositions and methods for detection thereof |
| WO2004039986A1 (en) | 2002-10-29 | 2004-05-13 | Syngenta Participations Ag | Cot102 insecticidal cotton |
| WO2004049804A2 (en) | 2002-11-29 | 2004-06-17 | Syngenta Participations Ag | Fungicidal combinations for crop potection |
| WO2004072235A2 (en) | 2003-02-12 | 2004-08-26 | Monsanto Technology Llc | Cotton event mon 88913 and compositions and methods for detection thereof |
| WO2004074492A1 (en) | 2003-02-20 | 2004-09-02 | Kws Saat Ag | Glyphosate tolerant sugar beet |
| WO2004083193A1 (en) | 2003-03-17 | 2004-09-30 | Sumitomo Chemical Company, Limited | Amide compound and bactericide composition containing the same |
| CN1456054A (en) | 2003-03-25 | 2003-11-19 | 浙江省化工研究院 | Methoxy methyl acrylate compounds as bactericidal agent |
| WO2004099447A2 (en) | 2003-05-02 | 2004-11-18 | Dow Agrosciences Llc | Corn event tc1507 and methods for detection thereof |
| WO2005061720A2 (en) | 2003-12-11 | 2005-07-07 | Monsanto Technology Llc | High lysine maize compositions and methods for detection thereof |
| WO2005059103A2 (en) | 2003-12-15 | 2005-06-30 | Monsanto Technology Llc | Corn plant mon88017 and compositions and methods for detection thereof |
| WO2005063721A1 (en) | 2003-12-19 | 2005-07-14 | E.I. Dupont De Nemours And Company | Herbicidal pyrimidines |
| WO2005087772A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| WO2005087773A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| WO2005103301A2 (en) | 2004-03-25 | 2005-11-03 | Syngenta Participations Ag | Corn event mir604 |
| WO2005103266A1 (en) | 2004-03-26 | 2005-11-03 | Dow Agrosciences Llc | Cry1f and cry1ac transgenic cotton lines and event-specific identification thereof |
| WO2005120234A2 (en) | 2004-06-03 | 2005-12-22 | E.I. Dupont De Nemours And Company | Fungicidal mixtures of amidinylphenyl compounds |
| CA2471555A1 (en) | 2004-06-18 | 2005-12-18 | Thomas D. Johnson | Controlling plant pathogens with fungal/bacterial antagonist combinations comprising trichoderma virens and bacillus amyloliquefaciens |
| WO2005123690A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-difluoromethyl-pyrazol-4-carbonic acid-(ortho-phenyl)-anilides, and use thereof as a fungicide |
| WO2005123689A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-trifluoromethyl-pyrazole-4-carboxylic acid (ortho-phenyl)-anilides and to use thereof as fungicide |
| WO2006015866A1 (en) | 2004-08-12 | 2006-02-16 | Syngenta Participations Ag | Method for protecting useful plants or plant propagation material |
| WO2006039376A2 (en) | 2004-09-29 | 2006-04-13 | Pioneer Hi-Bred International, Inc. | Corn event das-59122-7 and methods for detection thereof |
| WO2006087325A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | 5-alkoxyalkyl-6-alkyl-7-amino-azolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said substances |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| WO2006098952A2 (en) | 2005-03-16 | 2006-09-21 | Syngenta Participations Ag | Corn event 3272 and methods of detection thereof |
| WO2006108674A2 (en) | 2005-04-08 | 2006-10-19 | Bayer Bioscience N.V. | Elite event a2704-12 and methods and kits for identifying such event in biological samples |
| WO2006108675A2 (en) | 2005-04-11 | 2006-10-19 | Bayer Bioscience N.V. | Elite event a5547-127 and methods and kits for identifying such event in biological samples |
| WO2006130436A2 (en) | 2005-05-27 | 2006-12-07 | Monsanto Technology Llc | Soybean event mon89788 and methods for detection thereof |
| WO2006128573A2 (en) | 2005-06-02 | 2006-12-07 | Syngenta Participations Ag | Ce43- 67b, insecticidal transgenic cotton expressing cry1ab |
| WO2007006670A1 (en) | 2005-07-07 | 2007-01-18 | Basf Aktiengesellschaft | N-thio-anthranilamid compounds and their use as pesticides |
| CN1907024A (en) | 2005-08-03 | 2007-02-07 | 浙江化工科技集团有限公司 | Methoxyl group displacement methyl acrylate compound bactericidal agent |
| WO2007017186A1 (en) | 2005-08-08 | 2007-02-15 | Bayer Bioscience N.V. | Herbicide tolerant cotton plants and methods for identifying same |
| WO2007082098A2 (en) | 2006-01-13 | 2007-07-19 | Dow Agrosciences Llc | 6-(poly-substituted aryl)-4-aminopicolinates and their use as herbicides |
| WO2007090624A2 (en) | 2006-02-09 | 2007-08-16 | Syngenta Participations Ag | A method of protecting a plant propagation material, a plant, and/or plant organs |
| WO2007129454A1 (en) | 2006-05-08 | 2007-11-15 | Kumiai Chemical Industry Co., Ltd. | 1,2-benzisothiazole derivative, and agricultural or horticultural plant disease-controlling agent |
| WO2007140256A1 (en) | 2006-05-26 | 2007-12-06 | Monsanto Technology, Llc | Corn plant and seed corresponding to transgenic event mon89034 and methods for detection and use thereof |
| WO2007142840A2 (en) | 2006-06-03 | 2007-12-13 | Syngenta Participations Ag | Corn event mir162 |
| WO2008002371A1 (en) | 2006-06-23 | 2008-01-03 | Becker Underwood Inc. | Improved shelf life and on seed stabilization of liquid bacterium inoculants |
| WO2008002872A2 (en) | 2006-06-28 | 2008-01-03 | Pioneer Hi-Bred International, Inc. | Soybean event 3560.4.3.5 and compositions and methods for the identification and/or detection thereof |
| WO2008013622A2 (en) | 2006-07-27 | 2008-01-31 | E. I. Du Pont De Nemours And Company | Fungicidal azocyclic amides |
| WO2008112019A2 (en) | 2006-10-30 | 2008-09-18 | Pioneer Hi-Bred International, Inc. | Maize event dp-098140-6 and compositions and methods for the identification and/or detection thereof |
| WO2008054747A2 (en) | 2006-10-31 | 2008-05-08 | E. I. Du Pont De Nemours And Company | Soybean event dp-305423-1 and compositions and methods for the identification and/or detection thereof |
| WO2008122406A1 (en) | 2007-04-05 | 2008-10-16 | Bayer Bioscience N.V. | Insect resistant cotton plants and methods for identifying same |
| WO2008151780A1 (en) | 2007-06-11 | 2008-12-18 | Bayer Bioscience N.V. | Insect resistant cotton plants comprising elite event ee-gh6 and methods for identifying same |
| WO2009064652A1 (en) | 2007-11-15 | 2009-05-22 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87701 and methods for detection thereof |
| WO2009090181A2 (en) | 2008-01-15 | 2009-07-23 | Bayer Cropscience Sa | Pesticide composition comprising a tetrazolyloxime derivative and a fungicide or an insecticide active substance |
| WO2009094442A2 (en) | 2008-01-22 | 2009-07-30 | Dow Agrosciences Llc | 5-fluoro pyrimidine derivatives |
| WO2009103049A2 (en) | 2008-02-14 | 2009-08-20 | Pioneer Hi-Bred International, Inc. | Plant genomic dna flanking spt event and methods for identifying spt event |
| WO2009102873A1 (en) | 2008-02-15 | 2009-08-20 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87769 and methods for detection thereof |
| WO2009111263A1 (en) | 2008-02-29 | 2009-09-11 | Monsanto Technology Llc | Corn plant event mon87460 and compositions and methods for detection thereof |
| WO2009119089A1 (en) | 2008-03-27 | 2009-10-01 | 日本曹達株式会社 | Nitrogen-containing heterocyclic compound and bactericide for agricultural and horticultural use |
| WO2009126473A1 (en) | 2008-04-07 | 2009-10-15 | Bayer Cropscience Lp | Stable aqueous spore-containing formulation |
| WO2010018686A1 (en) | 2008-08-12 | 2010-02-18 | 日本曹達株式会社 | Nitrogenated heterocyclic derivative, and agricultural or horticultural bactericidal agent |
| US20110136782A1 (en) | 2008-08-12 | 2011-06-09 | Nippon Soda Co., Ltd | Nitrogen-containing heterocyclic derivative and fungicide for agricultural and horticultural use |
| EP2314583A1 (en) * | 2008-08-12 | 2011-04-27 | Nippon Soda Co., Ltd. | Nitrogenated heterocyclic derivative, and agricultural or horticultural bactericidal agent |
| WO2010037016A1 (en) | 2008-09-29 | 2010-04-01 | Monsanto Technology Llc | Soybean transgenic event mon87705 and methods for detection thereof |
| WO2010077816A1 (en) | 2008-12-16 | 2010-07-08 | Syngenta Participations Ag | Corn event 5307 |
| WO2010069882A1 (en) | 2008-12-17 | 2010-06-24 | Syngenta Participations Ag | Isoxazole derivatives for use as fungicides |
| WO2010080829A1 (en) | 2009-01-07 | 2010-07-15 | Basf Agrochemical Products B.V. | Soybean event 127 and methods related thereto |
| US20100260735A1 (en) | 2009-04-13 | 2010-10-14 | University of Delawre | Methods for promoting plant health |
| WO2010139271A1 (en) | 2009-06-05 | 2010-12-09 | 中国中化股份有限公司 | E-type phenyl acrylic ester compounds containing substituted anilino pyrimidine group and uses thereof |
| WO2011022469A2 (en) | 2009-08-19 | 2011-02-24 | Dow Agrosciences Llc | Aad-1 event das-40278-9, related transgenic corn lines, and event-specific identification thereof |
| WO2011028657A1 (en) | 2009-09-01 | 2011-03-10 | Dow Agrosciences Llc | Synergistic fungicidal compositions containing a 5-fluoropyrimidine derivative for fungal control in cereals |
| WO2011034704A1 (en) | 2009-09-17 | 2011-03-24 | Monsanto Technology Llc | Soybean transgenic event mon 87708 and methods of use thereof |
| WO2011062904A1 (en) | 2009-11-23 | 2011-05-26 | Monsanto Technology Llc | Transgenic maize event mon 87427 and the relative development scale |
| WO2011066384A1 (en) | 2009-11-24 | 2011-06-03 | Dow Agrosciences Llc | Aad-12 event 416, related transgenic soybean lines, and event-specific identification thereof |
| WO2011084621A1 (en) | 2009-12-17 | 2011-07-14 | Pioneer Hi-Bred International, Inc. | Maize event dp-004114-3 and methods for detection thereof |
| WO2011077514A1 (en) | 2009-12-22 | 2011-06-30 | 三井化学アグロ株式会社 | Plant disease control composition and method for controlling plant diseases by applying the composition |
| WO2011081174A1 (en) | 2010-01-04 | 2011-07-07 | 日本曹達株式会社 | Nitrogen-containing heterocyclic compound and agricultural/horticultural germicide |
| WO2011109395A2 (en) | 2010-03-01 | 2011-09-09 | University Of Delaware | Compositions and methods for increasing biomass, iron concentration, and tolerance to pathogens in plants |
| WO2011135833A1 (en) | 2010-04-28 | 2011-11-03 | Sumitomo Chemical Company, Limited | Plant disease control composition and its use |
| WO2011153186A1 (en) | 2010-06-04 | 2011-12-08 | Monsanto Technology Llc | Transgenic brassica event mon 88302 and methods of use thereof |
| WO2012051199A2 (en) | 2010-10-12 | 2012-04-19 | Monsanto Technology Llc | Soybean plant and seed corresponding to transgenic event mon87712 and methods for detection thereof |
| US20130236522A1 (en) | 2010-11-10 | 2013-09-12 | Kumiai Chemical Industry Co., Ltd. | Microbial pesticidal composition |
| US20120149571A1 (en) | 2010-12-10 | 2012-06-14 | Auburn University | Inoculants Including Bacillus Bacteria for Inducing Production of Volatile Organic Compounds in Plants |
| US8445255B2 (en) | 2010-12-10 | 2013-05-21 | Auburn University | Inoculants including Bacillus bacteria for inducing production of volatile organic compounds in plants |
| WO2012082548A2 (en) | 2010-12-15 | 2012-06-21 | Syngenta Participations Ag | Soybean event syht0h2 and compositions and methods for detection thereof |
| WO2012084812A1 (en) | 2010-12-20 | 2012-06-28 | Isagro Ricerca S.R.L. | Aminoindanes amides having a high fungicidal activity and their phytosanitary compositions |
| WO2012134808A1 (en) | 2011-03-30 | 2012-10-04 | Monsanto Technology Llc | Cotton transgenic event mon 88701 and methods of use thereof |
| WO2012165511A1 (en) | 2011-05-31 | 2012-12-06 | クミアイ化学工業株式会社 | Method for controlling diseases in rice plant |
| WO2012168188A1 (en) | 2011-06-07 | 2012-12-13 | Bayer Intellectual Property Gmbh | Active compound combinations |
| WO2013003558A1 (en) | 2011-06-30 | 2013-01-03 | Monsanto Technology Llc | Alfalfa plant and seed corresponding to transgenic event kk 179-2 and methods for detection thereof |
| WO2013007767A1 (en) | 2011-07-13 | 2013-01-17 | Basf Se | Fungicidal substituted 2-[2-halogenalkyl-4-(phenoxy)-phenyl]-1-[1,2,4]triazol-1-yl-ethanol compounds |
| WO2013010862A1 (en) | 2011-07-15 | 2013-01-24 | Basf Se | Fungicidal alkyl-substituted 2-[2-chloro-4-(4-chloro-phenoxy)-phenyl]-1-[1,2,4]triazol-1-yl-ethanol compounds |
| WO2013016516A1 (en) | 2011-07-26 | 2013-01-31 | Dow Agrosciences Llc | Insect resistant and herbicide tolerant breeding stack of soybean event pdab9582.814.19.1 and pdab4468.04.16.1 |
| WO2013016527A1 (en) | 2011-07-26 | 2013-01-31 | Dow Agrosciences Llc | Insect resistant and herbicide tolerant soybean event 9582.814.19.1 |
| WO2013024010A1 (en) | 2011-08-12 | 2013-02-21 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2013024009A1 (en) | 2011-08-12 | 2013-02-21 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2013032693A2 (en) | 2011-08-27 | 2013-03-07 | Marrone Bio Innovations, Inc. | Isolated bacterial strain of the genus burkholderia and pesticidal metabolites therefrom-formulations and uses |
| WO2013047441A1 (en) | 2011-09-26 | 2013-04-04 | 日本曹達株式会社 | Agricultural and horticultural bactericide composition |
| EP2762002A1 (en) * | 2011-09-26 | 2014-08-06 | Nippon Soda Co., Ltd. | Agricultural and horticultural fungicidal composition |
| WO2013047749A1 (en) | 2011-09-29 | 2013-04-04 | 三井化学アグロ株式会社 | Production method for 4, 4-difluoro-3,4-dihydroisoquinoline derivative |
| WO2013092224A1 (en) | 2011-12-21 | 2013-06-27 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi resistant to qo inhibitors |
| WO2013112527A1 (en) | 2012-01-23 | 2013-08-01 | Dow Agrosciences Llc | Herbicide tolerant cotton event pdab4468.19.10.3 |
| WO2013116251A2 (en) | 2012-02-01 | 2013-08-08 | E. I. Du Pont De Nemours And Company | Fungicidal pyrazole mixtures |
| WO2013127704A1 (en) | 2012-02-27 | 2013-09-06 | Bayer Intellectual Property Gmbh | Active compound combinations containing a thiazoylisoxazoline and a fungicide |
| WO2013162072A1 (en) | 2012-04-27 | 2013-10-31 | Sumitomo Chemical Company, Limited | Tetrazolinone compounds and its use as pesticides |
| WO2013169923A2 (en) | 2012-05-08 | 2013-11-14 | Monsanto Technology Llc | Corn event mon 87411 |
| CN103387541A (en) | 2012-05-10 | 2013-11-13 | 中国中化股份有限公司 | Preparation method of substituted pyrazolylether compound |
| WO2014029697A1 (en) | 2012-08-22 | 2014-02-27 | Basf Se | Fungicidal ternary mixtures comprising fluazinam |
| WO2014060177A1 (en) | 2012-10-16 | 2014-04-24 | Syngenta Participations Ag | Fungicidal compositions |
| WO2014116854A1 (en) | 2013-01-25 | 2014-07-31 | Pioneer Hi-Bred International, Inc. | Maize event dp-033121-3 and methods for detection thereof |
| WO2014124369A1 (en) | 2013-02-11 | 2014-08-14 | Bayer Cropscience Lp | Compositions comprising a streptomyces-based biological control agent and a fungicide |
| WO2014178913A1 (en) | 2013-05-02 | 2014-11-06 | J.R. Simplot Company | Potato cultivar f10 |
| WO2014178910A1 (en) | 2013-05-02 | 2014-11-06 | J.R. Simplot Company | Potato cultivar e12 |
| WO2014178941A1 (en) | 2013-05-02 | 2014-11-06 | J.R. Simplot Company | Potato cultivar j3 |
| WO2014179276A1 (en) | 2013-05-02 | 2014-11-06 | J.R. Simplot Company | Potato cultivar j55 |
| WO2014201235A2 (en) | 2013-06-14 | 2014-12-18 | Monsanto Technology Llc | Soybean transgenic event mon87751 and methods for detection and use thereof |
| WO2015053998A1 (en) | 2013-10-09 | 2015-04-16 | Monsanto Technology Llc | Transgenic corn event mon87403 and methods for detection thereof |
| WO2015065922A1 (en) | 2013-10-28 | 2015-05-07 | Dexcom, Inc. | Devices used in connection with continuous analyte monitoring that provide the user with one or more notifications, and related methods |
| EP2865265A1 (en) | 2014-02-13 | 2015-04-29 | Bayer CropScience AG | Active compound combinations comprising phenylamidine compounds and biological control agents |
| WO2015142571A1 (en) | 2014-03-20 | 2015-09-24 | Monsanto Technology Llc | Transgenic maize event mon 87419 and methods of use thereof |
| WO2016020371A1 (en) | 2014-08-04 | 2016-02-11 | Basf Se | Antifungal paenibacillus strains, fusaricidin-type compounds, and their use |
| WO2016156241A1 (en) | 2015-04-02 | 2016-10-06 | Syngenta Participations Ag | Herbicidal mixtures |
| WO2016162265A1 (en) | 2015-04-07 | 2016-10-13 | Syngenta Participations Ag | Herbicidal mixtures |
| WO2016183445A1 (en) | 2015-05-14 | 2016-11-17 | J.R. Simplot Company | Potato cultivar v11 |
| WO2017062825A1 (en) | 2015-10-08 | 2017-04-13 | J.R. Simplot Company | Potato cultivar y9 |
| WO2017062831A1 (en) | 2015-10-08 | 2017-04-13 | J.R. Simplot Company | Potato cultivar x17 |
| WO2018177894A1 (en) | 2017-03-31 | 2018-10-04 | Syngenta Participations Ag | Fungicidal compositions |
| WO2020212513A1 (en) | 2019-04-18 | 2020-10-22 | Syngenta Crop Protection Ag | Process for the preparation of microbiocidal oxadiazole derivatives |
Non-Patent Citations (30)
| Title |
|---|
| "Perkin Transactions 1: Organic and Bio-Organic Chemistry", JOURNAL OF THE CHEMICAL SOCIETY, 1995 |
| "Technical Monograph No. 2", 2008, CROPLIFE INTERNATIONAL, article "Catalogue of pesticide formulation types and international coding system" |
| APPL. ENVIRON. MICROBIOL., vol. 73, no. 8, 2007, pages 2635 |
| BIOCONTROL SCIENCE TECHNOL., vol. 22, no. 7, 2012, pages 747 - 761 |
| BIOCONTROL, vol. 57, 2012, pages 687 - 696 |
| BIOL. FERTIL. SOILS, vol. 47, 2011, pages 81 - 89 |
| BIOORGANIC & MEDICINAL CHEMISTRY, vol. 16, no. 11, 2008, pages 6124 - 6130 |
| CAN. J. PLANT SCI., vol. 48, no. 6, 1968, pages 587 - 94 |
| CAN. J. PLANT. SCI., vol. 70, 1990, pages 661 - 666 |
| CROP PROTECTION, vol. 27, 2008, pages 352 - 361 |
| EUR. J. SOIL BIOL., vol. 45, 2009, pages 28 - 35 |
| FEDERAL REGISTER, vol. 76, no. 22, 2 February 2011 (2011-02-02), pages 5808 |
| FERTILIZER RES., vol. 39, 1994, pages 97 - 103 |
| HETEROCYCLES, vol. 38, no. 2, 1994, pages 305 - 18 |
| HETEROCYCLIC COMMUNICATIONS, vol. 4, no. 6, 1998, pages 547 - 557 |
| INT. J. SYST. EVOL. MICROBIOL., vol. 66, 2016, pages 1212 - 1217 |
| J. AM. CHEM. SOC., vol. 123, no. 25, 2001, pages 5962 - 5973 |
| J. HETEROCYC. CHEM., vol. 18, no. 7, 1981, pages 1305 - 8 |
| J. INVERTEBRATE PATHOL., vol. 107, 2011, pages 112 - 126 |
| J. MED. CHEM., vol. 38, no. 11, 1995, pages 1892 - 903 |
| J. PLANT DIS. PROT., vol. 105, 1998, pages 181 - 197 |
| KNOWLES: "Agrow Reports DS243", 2005, T&F INFORMA, article "New developments in crop protection product formulation" |
| KNOWLES: "Agrow Reports DS256", 2006, T&F INFORMA, article "Adjuvants and additives" |
| MOLLETGRUBEMANN: "Formulation technology", 2001, WILEY VCH |
| PLANT SOIL, vol. 331, 2010, pages 413 - 425 |
| PNAS, vol. 107, no. 19, 2010, pages 8563 - 856 |
| SCI., vol. 78, no. 1, 1998, pages 91 - 102 |
| SCIENCE, vol. 257, 1992, pages 85 - 88 |
| SYNTHESIS, vol. 51, no. 19, 2019, pages 3709 - 3714 |
| SYSTEM. APPL. MICROBIOL., vol. 27, 2004, pages 372 - 379 |
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2024104814A1 (en) * | 2022-11-16 | 2024-05-23 | Basf Se | Fungicidal mixture comprising substituted pyridines |
| WO2024165343A1 (en) * | 2023-02-08 | 2024-08-15 | Basf Se | New substituted quinoline compounds for combatitng phytopathogenic fungi |
| WO2024194038A1 (en) * | 2023-03-17 | 2024-09-26 | Basf Se | Substituted pyridyl/pyrazidyl dihydrobenzothiazepine compounds for combatting phytopathogenic fungi |
| WO2025031842A1 (en) | 2023-08-09 | 2025-02-13 | Basf Se | New substituted benzoxazepine picolinonitrile compounds for combatting phytopathogenic fungi |
| WO2025180964A1 (en) | 2024-03-01 | 2025-09-04 | Basf Se | New substituted benzoxazepine compounds for combatting phytopathogenic fungi |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2022279357A1 (en) | 2023-11-30 |
| CN117355519A (en) | 2024-01-05 |
| CO2023015529A2 (en) | 2023-11-30 |
| KR20240008856A (en) | 2024-01-19 |
| CA3218900A1 (en) | 2022-11-24 |
| AR125907A1 (en) | 2023-08-23 |
| JP2024519815A (en) | 2024-05-21 |
| EP4341258B1 (en) | 2025-12-03 |
| IL308529A (en) | 2024-01-01 |
| BR112023024005A2 (en) | 2024-01-30 |
| EP4341258A1 (en) | 2024-03-27 |
| CL2023003418A1 (en) | 2024-04-26 |
| MX2023013721A (en) | 2024-01-16 |
| US20240270727A1 (en) | 2024-08-15 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP4341258B1 (en) | New substituted pyridines as fungicides | |
| EP4341256A1 (en) | New substituted pyridines as fungicides | |
| EP4380927A1 (en) | (3-pirydyl)-quinazoline | |
| EP4380926A1 (en) | (3-quinolyl)-quinazoline | |
| WO2025031843A1 (en) | New substituted benzoxazine picolinonitrile compounds for combatting phytopathogenic fungi | |
| WO2024104823A1 (en) | New substituted tetrahydrobenzoxazepine | |
| WO2022243109A1 (en) | New substituted quinolines as fungicides | |
| WO2025031842A1 (en) | New substituted benzoxazepine picolinonitrile compounds for combatting phytopathogenic fungi | |
| WO2025008227A1 (en) | Substituted pyridyl/pyrazinyl dihydropyrrolotriazine compounds for combatting phytopath-ogenic fungi | |
| WO2025180964A1 (en) | New substituted benzoxazepine compounds for combatting phytopathogenic fungi | |
| WO2024104815A1 (en) | Substituted benzodiazepines as fungicides | |
| WO2026021910A1 (en) | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi | |
| WO2025008226A1 (en) | Substituted quinolyl/quinoxalyl dihydropyrrolotriazine compounds for combatting phyto-pathogenic fungi | |
| WO2026021911A1 (en) | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi | |
| EP4619393A1 (en) | Substituted benzodiazepines as fungicides | |
| WO2026021909A1 (en) | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi | |
| WO2026021912A1 (en) | New substituted benzothiazine pyridine compounds for combatting phytopathogenic fungi | |
| EP4619394A1 (en) | Substituted tetrahydrobenzodiazepine as fungicides | |
| KR20250156732A (en) | Substituted pyridyl/pyrazidyl dihydrobenzothiazepine compounds for combating phytopathogenic fungi | |
| EA049848B1 (en) | NEW SUBSTITUTED PYRIDINES AS FUNGICIDES |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22728525 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202317077037 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18290347 Country of ref document: US Ref document number: 3218900 Country of ref document: CA Ref document number: 2022279357 Country of ref document: AU Ref document number: AU2022279357 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 308529 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023571307 Country of ref document: JP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: NC2023/0015529 Country of ref document: CO Ref document number: MX/A/2023/013721 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202280036430.1 Country of ref document: CN |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112023024005 Country of ref document: BR |
|
| ENP | Entry into the national phase |
Ref document number: 2022279357 Country of ref document: AU Date of ref document: 20220510 Kind code of ref document: A |
|
| WWP | Wipo information: published in national office |
Ref document number: NC2023/0015529 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202393249 Country of ref document: EA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022728525 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2022728525 Country of ref document: EP Effective date: 20231218 |
|
| ENP | Entry into the national phase |
Ref document number: 112023024005 Country of ref document: BR Kind code of ref document: A2 Effective date: 20231116 |
|
| WWG | Wipo information: grant in national office |
Ref document number: 2022728525 Country of ref document: EP |