WO2022238157A1 - Fungicidal mixtures comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles - Google Patents
Fungicidal mixtures comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles Download PDFInfo
- Publication number
- WO2022238157A1 WO2022238157A1 PCT/EP2022/061683 EP2022061683W WO2022238157A1 WO 2022238157 A1 WO2022238157 A1 WO 2022238157A1 EP 2022061683 W EP2022061683 W EP 2022061683W WO 2022238157 A1 WO2022238157 A1 WO 2022238157A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- methyl
- phenyl
- compound
- mixtures
- trifluoromethyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/82—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with three ring hetero atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N37/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids
- A01N37/52—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds containing a carbon atom having three bonds to hetero atoms with at the most two bonds to halogen, e.g. carboxylic acids containing groups, e.g. carboxylic acid amidines
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P3/00—Fungicides
Definitions
- Fungicidal mixtures comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles
- the present invention relates to fungicidal mixtures comprising at least one substituted 3- phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles ( compounds I) and compound II; to a method for controlling phytopathogenic harmful fungi, comprising treating the fungi, their habitat or the seed, the soil or the plants to be protected against fungal attack with an effective amount of mixtures comprising compounds I and compound II; to the non-therapeutic use of mixtures comprising compounds I and compound II for controlling phytopathogenic harmful fungi; to agrochemical compositions comprising these mixtures; and to agrochemical compositions further comprising seed.
- the present invention relates to a fungicidal mixture
- a fungicidal mixture comprising as active component 1) at least one compound, or an /V-oxide, or an agriculturally useful salt thereof, selected from the group consisting of
- the compounds 1-1 and I-2 defined above for component 1) is herein collectively referred to as “compounds of the formula I” or “compounds I”.
- the compounds for component 3) selected from groups A) to O), as defined herein below, is herein collectively referred to as “compounds of the formula IN” or “compounds III”.
- oxadiazole fungicides 1-1 and I-2 their fungicidal action and methods for preparing them are known e.g. from WO 2015/185485, WO 2017/211649 and WO 2013/008162.
- Compound II its preparation and use as a fungicide is known from: WO 2016202742, WO 2018109002, WO 2018108977, WO 202169704, WO 202169706, WO 202169707 and WO 2021069702.
- Practical agricultural experience has shown that the repeated and exclusive application of an individual active compound in the control of harmful fungi leads in many cases to a rapid selection of those fungus strains which have developed natural or adapted resistance against the active compound in question. Effective control of these fungi with the active compound in question is then no longer possible.
- mixtures of different active compounds are conventionally employed for controlling harmful fungi.
- active compounds having different mechanisms of action it is possible to ensure successful control over a relatively long period of time.
- compositions comprising at least one compound I and compound II.
- the term “mixture” in the context of the present invention shall mean a physical mixture of the components prior to application or the result of a simultaneous, i.e. joint or separate, application of a compound I and of a compound II on the fungi, their habitat or the seed, the soil or the plants to be protected against fungal attack.
- Compounds I and/or the compounds II can be present in different crystal modifications, which may differ in biological activity.
- Agriculturally acceptable salts of the active compounds I, II and III encompass especially the salts of those cations or the acid addition salts of those acids whose cations and anions, respectively, have no adverse effect on the fungicidal action of the active compounds.
- Suitable cations are thus in particular the ions of the alkali metals, preferably sodium and potassium, of the alkaline earth metals, preferably calcium, magnesium and barium, of the transition metals, preferably manganese, copper, zinc and iron, and also the ammonium ion which, if desired, may carry 1 to 4 CrC4-alkyl substituents and/or one phenyl or benzyl substituent, preferably diisopropylammonium, tetramethylammonium, tetrabutylammonium, trimethylbenzylammonium, furthermore phosphonium ions, sulfonium ions, preferably tri(Ci-C4-alkyl)sulfonium, and s
- Anions of useful acid addition salts are primarily chloride, bromide, fluoride, hydrogensulfate, sulfate, dihydrogenphosphate, hydrogen- phosphate, phosphate, nitrate, bicarbonate, carbonate, hexafluorosilicate, hexafluorophosphate, benzoate, and the anions of CrC4-alkanoic acids, preferably formate, acetate, propionate and butyrate. They can be formed by reacting a compound I with an acid of the corresponding anion, preferably of hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid or nitric acid.
- the present invention relates to mixtures, wherein active component 1) comprises compound 1-1. In another embodiment the present invention relates to mixtures, wherein active component 1) is compound 1-1 and active component 2) is compound II. In one embodiment the present invention relates to mixtures, wherein active component 1) comprises compound I-2. In another embodiment the present invention relates to mixtures, wherein active component 1) is compound I-2 and active component 2) is compound II.
- the ratio by weight of component 1) and compound II in binary mixtures is from 500:1 to 1 :500, 100:1 to 1:100, 1:1 to 1 :100, or 100:1 to 1:1 ; preferably from 50:1 to 1:50; more preferably from 20:1 to 1 :20, 1:1 to 1 :20, or 20:1 to 1 :1 , most preferably from 5:1 to 1 :5, 1:1 to 1:5, 5:1 to 1 :1 , 3:1 to 1:3, 1 :1 to 1.3, 3:1 to 1:1 , 2:1 to 1 :2, or 1:1 to 1 :2.
- the scope of the present invention includes mixtures of the (R)- and (S)-isomers and the racemates of compounds having one or more chiral centers.
- atrope isomers of active compounds I, II and/or III may be present. They also form part of the subject matter of the invention.
- the present invention relates to fungicidal mixtures M.1 to M.30 that are each identified in one row of table M below, wherein component 1) and component 2) are identified together with their relative amounts (weight ratio).
- the amount of compound 1-1 in mixtures M.1 to M.15 is in the range between 80 and 150 g/ha, preferably 100 g/ha.
- the amount of compound 1-1 in mixtures M.1 to M.15 is in the range between 70 and 150 g/ha, preferably 100 g/ha.
- the amount of compound I-2 in mixtures M.16 to M.30 is in the range between 70 and 100 g/ha, preferably 85 g/ha.
- the binary mixtures above comprise, besides at least one compound I as component 1) and at least one compound II as component 2), additionally at least one further active compound III as component 3) resulting in a ternary mixture.
- one embodiment of the invention relates to ternary mixtures comprising, as active component 1) at least one active compound selected from the group consisting of 1-1 and I-2 as defined herein above, or an /V-oxide, or an agriculturally useful salt thereof; and as active component 2) compound II, or an /V-oxide, or an agriculturally useful salt thereof; and as active component 3) at least one active compound III selected from the groups A) to O), or an N-oxide, or an agriculturally useful salt thereof:
- Inhibitors of complex III at Q 0 site azoxystrobin (A.1.1), coumethoxystrobin (A.1.2), coumoxystrobin (A.1.3), dimoxystrobin (A.1.4), enestroburin (A.1.5), fenaminstrobin (A.1.6), fenoxystrobin/flufenoxystrobin (A.1.7), fluoxastrobin (A.1.8), kresoxim-methyl (A.1.9), mandestrobin (A.1.10), metominostrobin (A.1.11), orysastrobin (A.1.12), picoxystrobin (A.1.13), pyraclostrobin (A.1.14), pyrametostrobin (A.1.15), pyraoxystrobin (A.1.16), trifloxystrobin (A.1.17), 2-(2-(3-(2,6-dichlorophenyl)-1-methyl-allylideneaminooxymethyl)-phenyl)- 2-methoxyi
- Delta 14- reductase inhibitors aldimorph (B.2.1), dodemorph (B.2.2), dodemorph-acetate (B.2.3), fenpropimorph (B.2.4), tridemorph (B.2.5), fenpropidin (B.2.6), piperalin (B.2.7), spiroxamine (B.2.8);
- Nucleic acid synthesis inhibitors phenylamides or acyl amino acid fungicides benalaxyl (C.1.1), benalaxyl-M (C.1.2), kiralaxyl (C.1.3), metalaxyl (C.1.4), metalaxyl-M (C.1.5), ofurace (C.1.6), oxadixyl (C.1.7); other nucleic acid synthesis inhibitors: hymexazole (C.2.1), octhilinone (C.2.2), oxolinic acid (C.2.3), bupirimate (C.2.4), 5-fluorocytosine (C.2.5), 5-fluoro-2-(p-tolylmethoxy)pyrimidin- 4-amine (C.2.6), 5-fluoro-2-(4-fluorophenylmethoxy)pyrimidin-4-amine (C.2.7), 5-fluoro-
- M Growth regulators abscisic acid (M.1.1), amidochlor, ancymidol, 6-benzylaminopurine, brassinolide, butralin, chlormequat, chlormequat chloride, choline chloride, cyclanilide, daminozide, dikegulac, dimethipin, 2,6-dimethylpuridine, ethephon, flumetralin, flurprimidol, fluthiacet, forchlorfenuron, gibberellic acid, inabenfide, indole-3-acetic acid , maleic hydrazide, mefluidide, mepiquat, mepiquat chloride, naphthaleneacetic acid, A/-6-benzyladenine, paclobutrazol, prohexadione, prohexadione-calcium, prohydrojasmon, thidiazuron, triapenthenol, tributyl phosphorotrithioate
- Lipid biosynthesis inhibitors alloxydim, alloxydim-sodium, butroxydim, clethodim, clodinafop, clodinafop-propargyl, cycloxydim, cyhalofop, cyhalofop-butyl, diclofop, diclofop- methyl, fenoxaprop, fenoxaprop-ethyl, fenoxaprop-P, fenoxaprop-P-ethyl, fluazifop, fluazifop- butyl, fluazifop-P, fluazifop-P-butyl, haloxyfop, haloxyfop-methyl, haloxyfop-P, haloxyfop-P- methyl, metamifop, pinoxaden, profoxydim, propaquizafop, quizalofop, quizalofop-e
- N.2 ALS inhibitors amidosulfuron, azimsulfuron, bensulfuron, bensulfuron-methyl, chlorimuron, chlorimuron-ethyl, chlorsulfuron, cinosulfuron, cyclosulfamuron, ethametsulfuron, ethamet- sulfuron-methyl, ethoxysulfuron, flazasulfuron, flucetosulfuron, flupyrsulfuron, flupyrsulfuron- methyl-sodium, foramsulfuron, halosulfuron, halosulfuron-methyl, imazosulfuron, iodosulfuron, iodosulfuron-methyl-sodium, iofensulfuron, iofensulfuron-sodium, mesosulfuron, metazo- sulfuron, metsulfuron, metsulfuron-methyl, nicosulfuron, orthos
- Photosynthesis inhibitors amicarbazone; chlorotriazine; ametryn, atrazine, chloridazone, cyanazine, desmetryn, dimethametryn.hexazinone, metribuzin, prometon, prometryn, pro- pazine, simazine, simetryn, terbumeton, terbuthylazin, terbutryn, trietazin; chlorobromuron, chlorotoluron, chloroxuron, dimefuron, diuron, fluometuron, isoproturon, isouron, linuron, metamitron, methabenzthiazuron, metobenzuron, metoxuron, monolinuron, neburon, siduron, tebuthiuron, thiadiazuron, desmedipham, karbutilat, phenmedipham, phenmedipham-ethyl, bromofenoxim, bromoxynil and
- N.4 protoporphyrinogen-IX oxidase inhibitors acifluorfen, acifluorfen-sodium, azafenidin, ben- carbazone, benzfendizone, bifenox, butafenacil, carfentrazone, carfentrazone-ethyl, chlor- methoxyfen, cinidon-ethyl, fluazolate, flufenpyr, flufenpyr-ethyl, flumiclorac, flumiclorac-pentyl, flumioxazin, fluoroglycofen, fluoroglycofen-ethyl, fluthiacet, fluthiacet-methyl, fomesafen, halosafen, lactofen, oxadiargyl, oxadiazon, oxyfluorfen, pentoxazone, profluazol, pyraclonil, pyraflufen, pyraflufen-e
- N.5 Bleacher herbicides beflubutamid, diflufenican, fluridone, flurochloridone, flurtamone, norflurazon, picolinafen, 4-(3-trifluoromethyhphenoxy)-2-(4-trifluoromethylphenyl)pyrimidine (180608-33-7); benzobicyclon, benzofenap, bicyclopyrone, clomazone, fenquintrione, isoxaflutole, mesotrione, pyrasulfotole, pyrazolynate, pyrazoxyfen, sulcotrione, tefuryltrione, tembotrione, tolpyralate, topramezone; aclonifen, amitrole, flumeturon;
- N.6 EPSP synthase inhibitors glyphosate, glyphosate-isopropylammonium, glyposate- potassium, glyphosate-trimesium (sulfosate);
- Glutamine synthase inhibitors bilanaphos (bialaphos), bilanaphos-sodium, glufosinate, glufosinate-P, glufosinate-ammonium;
- Mitosis inhibitors benfluralin, butralin, dinitramine, ethalfluralin, fluchloralin, oryzalin, pen- dimethalin, prodiamine, trifluralin; amiprophos, amiprophos-methyl, butamiphos; chlorthal, chlorthal-dimethyl, dithiopyr, thiazopyr, propyzamide, tebutam; carbetamide, chlorpropham, flamprop, flamprop-isopropyl, flamprop-methyl, flamprop-M-isopropyl, flamprop-M-methyl, propham;
- N.10 VLCFA inhibitors acetochlor, alachlor, butachlor, dimethachlor, dimethenamid, dimethen- amid-P, metazachlor, metolachlor, metolachlor-S, pethoxamid, pretilachlor, propachlor, prop- isochlor, thenylchlor, flufenacet, mefenacet, diphenamid, naproanilide, napropamide, napro- pamide-M, fentrazamide, anilofos, cafenstrole, fenoxasulfone, ipfencarbazone, piperophos, pyroxasulfone, isoxazoline compounds of the formulae 11.1, II.2, II.3, II.4, II.5, II.6, II.7, II.8 and II.9
- N.11 Cellulose biosynthesis inhibitors chlorthiamid, dichlobenil, flupoxam, indaziflam, isoxaben, triaziflam, 1-cyclohexyl-5-pentafluorphenyloxy-14-[1 ,2,4,6]thiatriazin-3-ylamine (175899-01-1);
- N.12 Decoupler herbicides dinoseb, dinoterb, DNOC and its salts;
- N.13 Auxinic herbicides 2,4-D and its salts and esters, clacyfos, 2,4-DB and its salts and esters, aminocyclopyrachlor and its salts and esters, aminopyralid and its salts such as aminopyralid-dimethylammonium, aminopyralid-tris(2-hydroxypropyl)ammonium and its esters, benazolin, benazolin-ethyl, chloramben and its salts and esters, clomeprop, clopyralid and its salts and esters, dicamba and its salts and esters, dichlorprop and its salts and esters, dichlorprop-P and its salts and esters, fluroxypyr, fluroxypyr-butometyl, fluroxypyr-meptyl, halauxifen and its salts and esters (943832-60-8); MCPA and its salts and esters, MCPA- thioethyl, MCPB and
- N.14 Auxin transport inhibitors diflufenzopyr, diflufenzopyr-sodium, naptalam, naptalam- sodium;
- Acetylcholine esterase (AChE) inhibitors aldicarb (0.1.1), alanycarb (0.1.2), bendiocarb (0.1.3), benfuracarb (0.1.4), butocarboxim (0.1.5), butoxycarboxim (0.1.6), carbaryl (0.1.7), carbofuran (0.1.8), carbosulfan (0.1.9), ethiofencarb (0.1.10), fenobucarb (0.1.11), formetanate (0.1.12), furathiocarb (0.1.13), isoprocarb (0.1.14), methiocarb (0.1.15), methomyl (0.1.16), metolcarb (0.1.17), oxamyl (0.1.18), pirimicarb (0.1.19), propoxur (0.1.20), thiodicarb (0.1.21), thiofanox (0.1.22), trimethacarb (0.1.23), XMC (0.1.24), xylylcarb
- GABA-gated chloride channel antagonists endosulfan (0.2.1), chlordane (0.2.2), ethiprole (0.2.3), fipronil (0.2.4), flufiprole (0.2.5), pyrafluprole (0.2.6), pyriprole (0.2.7);
- Nicotinic acetylcholine receptor agonists acetamiprid (0.4.1), clothianidin (0.4.2), cycloxaprid (0.4.3), dinotefuran (0.4.4), imidacloprid (0.4.5), nitenpyram (0.4.6), thiacloprid (0.4.7), thiamethoxam (0.4.8), 4,5-dihydro-/V-nitro-1-(2-oxiranylmethyl)-1/-/-imidazol-2-amine (0.4.9), (2E)-1-[(6-chloropyridin-3-yl)methyl]-/V’-nitro-2-pentylidenehydrazinecarboximidamide (0.4.10), 1-[(6-chloropyridin-3-yl)methyl]-7-methyl-8-nitro-5-propoxy-1,2,3,5,6,7- hexahydroimidazo[1,2-a]pyridine (0.4.11), nicotine (0.4.12), s
- Nicotinic acetylcholine receptor allosteric activators spinosad (0.5.1), spinetoram (0.5.2); 0.6 Chloride channel activators: abamectin (0.6.1), emamectin benzoate (0.6.2), ivermectin (0.6.3), lepimectin (0.6.4), milbemectin (0.6.5);
- 0.8 miscellaneous non-specific (multi-site) inhibitors methyl bromide (0.8.1) and other alkyl halides, chloropicrin (0.8.2), sulfuryl fluoride (0.8.3), borax (0.8.4), tartar emetic (0.8.5);
- 0.9 Chordotonal organ TRPV channel modulators pymetrozine (0.9.1), pyrifluquinazon (0.9.2), flonicamid (0.9.3);
- Mite growth inhibitors clofentezine (0.10.1), hexythiazox (0.10.2), diflovidazin (0.10.3), etoxazole (0.10.4);
- Bacillus thuringiensis Bacillus sphaericus and the insecticdal proteins they produce: Bacillus thuringiensis subsp. Israelensis (0.11.1), Bacillus sphaericus (0.11.2), Bacillus thuringiensis subsp. aizawai (0.11.3), Bacillus thuringiensis subsp. kurstaki (0.11.4), Bacillus thuringiensis subsp.
- Inhibitors of mitochondrial ATP synthase diafenthiuron (0.12.1), azocyclotin (0.12.2), cyhexatin (0.12.3), fenbutatin oxide (0.12.4), propargite (0.12.5), tetradifon (0.12.6);
- Nicotinic acetylcholine receptor (nAChR) channel blockers bensultap (0.14.1), cartap hydrochloride (0.14.2), thiocyclam (0.14.3), thiosultap sodium (0.14.4);
- Inhibitors of the chitin biosynthesis type 0 bistrifluron (0.15.1), chlorfluazuron (0.15.2), diflubenzuron (0.15.3), flucycloxuron (0.15.4), flufenoxuron (0.15.5), hexaflumuron (0.15.6), lufenuron (0.15.7), novaluron (0.15.8), noviflumuron (0.15.9), teflubenzuron (0.15.10), triflumuron (0.15.11);
- Inhibitors of the chitin biosynthesis type 1 buprofezin (0.16.1);
- Ecdyson receptor agonists methoxyfenozide (0.18.1), tebufenozide (0.18.2), halofenozide (0.18.3), fufenozide (0.18.4), chromafenozide (0.18.5);
- Octopamin receptor agonists amitraz (0.19.1);
- Mitochondrial complex III electron transport inhibitors hydramethylnon (0.20.1), acequinocyl (0.20.2), fluacrypyrim (0.20.3), bifenazate (0.20.4);
- Mitochondrial complex I electron transport inhibitors fenazaquin (0.21.1), fenpyroximate (0.21.2), pyrimidifen (0.21.3), pyridaben (0.21.4), tebufenpyrad (0.21.5), tolfenpyrad (0.21.6), rotenone (0.21.7);
- indoxacarb (0.22.1), metaflumizonev (0.22.2), 2-[2-(4-cyanophenyl)-1-[3-(trifluoromethyl)phenyl]ethylidene]-/ ⁇ /-[4- (difluoromethoxy)phenyl]-hydrazinecarboxamide (0.22.3), A/-(3-chloro-2-methylphenyl)-2-[(4- chlorophenyl)-[4-[methyl(methylsulfonyl)amino]phenyl]methylene]-hydrazinecarboxamide (0.22.4);
- Inhibitors of the of acetyl CoA carboxylase spirodiclofen (0.23.1), spiromesifen (0.23.2), spirotetramat (0.23.3), spiropidion (0.23.4);
- Mitochondrial complex IV electron transport inhibitors aluminium phosphide (0.24.1), calcium phosphide (0.24.2), phosphine (0.24.3), zinc phosphide (0.24.4), cyanide (0.24.5); 0.25 Mitochondrial complex II electron transport inhibitors: cyenopyrafen (0.25.1), cyflumetofen (0.25.2);
- insecticidal active compounds of unknown or uncertain mode of action afidopyropen (0.28.1), afoxolaner (0.28.2), azadirachtin (0.28.3), amidoflumet (0.28.4), benzoximate (0.28.5), broflanilide (0.28.6), bromopropylate (0.28.7), chinomethionat (0.28.8), cryolite (0.28.9), dicloromezotiaz (0.28.10), dicofol (0.28.11), flufenerim (0.28.12), flometoquin (0.28.13), fluensulfone (0.28.14), fluhexafon (0.28.15), fluopyram (0.28.16), fluralaner (0.28.17), metoxadiazone (0.28.18), piperonyl butoxide (0.28.19), pyflubumide (0.28.20), pyridalyl (0.28.21), tioxazaf
- component 3 The active substances referred to as component 3), their preparation and their activity e. g. against harmful fungi is known (cf.: http://www.alanwood.net/pesticides/); these substances are commercially available.
- the compounds described by IUPAC nomenclature, their preparation and their pesticidal activity are also known (cf. Can. J.
- T1-1 to T1-738 listed in Table T1 below relate to ternary mixtures comprising as active components binary mixture M.1 of table M, and one compound III selected from the groups A) to O) as defined herein (component 3), for example, being (A.1.1) or azoxystrobin, in embodiment T1-1).
- T1-1 M.1 + (A.1.1), T1-2: M.1 + (A.1.2), T1-3: M.1 + (A.1.3), T1-4: M.1 + (A.1.4), T1-5: M.1 + (A.1.5), T1-6: M.1 + (A.1.6), T1-7: M.1 + (A.1.7), T1-8: M.1 + (A.1.8), T1-9: M.1 + (A.1.9), T1-10: M.1 + (A.1.10), T1-11: M.1 + (A.1.11), T1-12: M.1 + (A.1.12), T1-13: M.1 + (A.1.13), T1-14: M.1 + (A.1.14), T1-15: M.1 + (A.1.15), T1-16: M.1 + (A.1.16), T1-17: M.1 + (A.1.17), T1-18: M.1 + (A.1.18), T1-19: M.1 + (A.1.1
- T1-56 M.1 + (A.3.22)
- T1-57 M.1 + (A.3.23)
- T1-58 M.1 + (A.3.24)
- T1-59 M.1 +
- T1-60 M.1 + (A.3.26)
- T1-61 M.1 + (A.3.27)
- T1-62 M.1 + (A.3.28)
- T1-63 M.1 +
- T1-64 M.1 + (A.3.31)
- T1-65 M.1 + (A.3.32)
- T1-66 M.1 + (A.3.33)
- T1-67 M.1 +
- T1-68 M.1 + (A.3.35)
- T1-69 M.1 + (A.3.36)
- T1-70 M.1 + (A.3.37)
- T1-71 M.1 +
- T1-72 M.1 + (A.3.39)
- T1-73 M.1 + (A.4.1)
- T1-74 M.1 + (A.4.2)
- T1-75 M.1 + (A.4.3)
- T1-76 M.1 + (A.4.4)
- T1-77 M.1 + (A.4.5)
- T1-78 M.1 + (A.4.6)
- T1-79 M.1 + (A.4.7)
- T1-80
- T1-94 M.1 + (B.1.9), T1-94: M.1 + (B.1.10), T1-95: M.1 + (B.1.11), T1-96: M.1 + (B.1.12), T1-97: M.1 + (B.1.13), T1-98: M.1 + (B.1.14), T1-99: M.1 + (B.1.15), T1-100: M.1 + (B.1.16), T1-101: M.1 + (B.1.17), T1-102: M.1 + (B.1.18), T1-103: M.1 + (B.1.19), T1-104: M.1 + (B.1.20), T1-105: M.1 +
- T1-106 M.1 + (B.1.22)
- T1-107 M.1 + (B.1.23)
- T1-108 M.1 + (B.1.24)
- T1-109 M.1 +
- T1-110 M.1 + (B.1.26)
- T1-111 M.1 + (B.1.27)
- T1-112 M.1 + (B.1.28)
- T1-113 M.1 +
- T1-114 M.1 + (B.1.30), T1-115: M.1 + (B.1.31), T1-116: M.1 + (B.1.32), T1-117: M.1 +
- T1-118 M.1 + (B.1.38)
- T1-119 M.1 + (B.1.39)
- T1-120 M.1 + (B.1.40)
- T1-121 M.1 +
- T1-122 M.1 + (B.1.42)
- T1-123 M.1 + (B.1.43)
- T1-124 M.1 + (B.1.44)
- T1-125 M.1 +
- T1-130 M.1 + (B.1.50)
- T1-131 M.1 + (B.1.51)
- T1-132 M.1 + (B.1.52)
- T1-133 M.1 +
- T 1 - 134 M.1 + (B.2.2), T1-135: M.1 + (B.2.3), T1-136: M.1 + (B.2.4), T1-137: M.1 + (B.2.5), T1-138: M.1 + (B.2.6), T1-139: M.1 + (B.2.7), T1-140: M.1 + (B.2.8), T1-141: M.1 + (B.3.1), T 1 - 142 : M.1 + (B.4.1), T1-143: M.1 + (C.1.1), T1-144: M.1 + (C.1.2), T1-145: M.1 + (C.1.3), T1-146: M.1 + (C.1.4), T1-147: M.1 + (C.1.5), T1-148: M.1 + (C.1.6), T1-149: M.1 + (C.1.7), T1-150: M.1 + (C.2.1), T1-151: M.1 + (B.
- T1-303 M.1 + (K.1.24)
- T1-304 M.1 + (K.1.25)
- T1-305 M.1 + (K.1.26)
- T1-306 M.1 +
- T1-307 M.1 + (K.1.28)
- T1-308 M.1 + (K.1.29)
- T1-309 M.1 + (K.1.30)
- T1-310 M.1 +
- T1-311 M.1 + (K.1.32)
- T1-312 M.1 + (K.1.33)
- T1-313 M.1 + (K.1.34)
- T1-314 M.1 +
- T1-319 M.1 + (K.1.40)
- T1-320 M.1 + (K.1.41)
- T1-321 M.1 + (K.1.42)
- T1-322 M.1 +
- T1-323 M.1 + (K.1.44)
- T1-324 M.1 + (K.1.45)
- T1-325 M.1 + (K.1.46)
- T1-326 M.1 +
- T 1-362 M.1 + (0.1.29), T1-363: M.1 + (0.1.30), T1-364: M.1 + (0.1.31), T1-365: M.1 + (0.1.32), T1-366: M.1 + (0.1.33), T1-367: M.1 + (0.1.34), T1-368: M.1 + (0.1.35), T1-369: M.1 + (0.1.36), T1-370: M.1 + (0.1.37), T1-371 : M.1 + (0.1.38), T1-372: M.1 + (0.1.39), T1-373:
- T1-412 M.1 + (0.1.78), T1-412: M.1 + (0.1.79), T1-413: M.1 + (0.1.80), T1-414: M.1 + (0.1.81), T1- 415: M.1 + (0.1.82), T1-416: M.1 + (0.1.83), T1-417: M.1 + (0.1.84), T1-418: M.1 + (0.1.85), T1-419: M.1 + (0.1.86), T1-420: M.1 + (0.1.87), T1-421 : M.1 + (0.1.88), T1-422: M.1 + (0.1.89), T1-423: M.1 + (0.1.90), T1-424: M.1 + (0.1.91), T1-425: M.1 + (0.2.1), T1-426: M.1 + (0.2.2), T1-427: M.1 + (0.2.3), T1-428: M.1 + (0.2.4), T1-429: M.1 + (0.2.5), T1-430: M.1 + (0.
- T1-451 M.1 + (0.3.20)
- T1-452 M.1 + (0.3.21)
- T1-453 M.1 + (0.3.22)
- T1- 454 M.1 + (0.3.23)
- T1-455 M.1 + (0.3.24)
- T1-456 M.1 + (0.3.25)
- T1-457 M.1 + (0.3.26)
- T1-458 M.1 + (0.3.27)
- T1-459 M.1 + (0.3.28)
- T1-460 M.1 + (0.3.29)
- T1-461 M.1 +
- T 1-462 M.1 + (0.3.31)
- T1-463 M.1 + (0.3.32)
- T1-464 M.1 + (0.3.33)
- T1-465 M.1 + (0.3.34)
- T 1-466 M.1 + (0.3.35)
- T1-467 M.1 + (0.3.36)
- T1-468 M.1 + (0.3.37)
- T1-469 T1-469:
- T1-470 M.1 + (0.3.38), T1-470: M.1 + (0.3.39), T1-471: M.1 + (0.3.40), T1-472: M.1 + (0.3.41), T1- 473: M.1 + (0.3.42), T1-474: M.1 + (0.3.43), T1-475: M.1 + (0.3.44), T1-476: M.1 + (0.3.45), T1-477: M.1 + (0.3.46), T1-478: M.1 + (0.3.47), T1-479: M.1 + (0.3.48), T1-480: M.1 +
- T1-481 M.1 + (0.3.50), T1-482: M.1 + (0.3.51), T1-483: M.1 + (0.3.52), T1-484: M.1 + (0.4.1), T1-485: M.1 + (0.4.2), T1-486: M.1 + (0.4.3), T1-487: M.1 + (0.4.4), T1-488: M.1 + (0.4.5), T1-489: M.1 + (0.4.6), T1-490: M.1 + (0.4.7), T1-491 : M.1 + (0.4.8), T1-492: M.1 + (0.4.9), T1-493: M.1 + (0.4.10), T1-494: M.1 + (0.4.11), T1-495: M.1 + (0.4.12), T1-496: M.1 + (0.4.13), T1-497: M.1 + (0.4.14), T1-498: M.1 + (0.4.15), T1-499: M.1 + (0.5.1), T1-500: M.1 + (0.5.
- T1-505 M.1 + (0.6.5)
- T1-506 M.1 + (0.7.1)
- T1-507 M.1 + (0.7.2)
- T1-508 M.1 +
- T1-509 M.1 + (0.7.4)
- T1-510 M.1 + (0.7.5)
- T1-511 M.1 + (0.8.1)
- T1-512 M.1 +
- T1-513 M.1 + (0.8.3)
- T1-514 M.1 + (0.8.4)
- T1-515 M.1 + (0.8.5)
- T1-516 M.1 +
- T1-517 M.1 + (0.9.2)
- T1-518 M.1 + (0.9.3)
- T1-519 M.1 + (0.10.1)
- T1-520 M.1 + (0.10.2)
- T1-524 M.1 + (0.11.2)
- T1-526 M.1 + (0.11.4)
- T1-527 M.1 + (0.11.5)
- T1-529 M.1 + (0.11.7), T1-530: M.1 + (0.11.8), T1-531: M.1 + (0.11.9), T1- 532: M.1 + (0.11.10), T1-533: M.1 + (0.11.11), T1-534: M.1 + (0.11.12), T1-535: M.1 + (0.11.13), T1-536: M.1 + (0.12.1), T1-537: M.1 + (0.12.2), T1-538: M.1 + (0.12.3), T1-539: M.1 + (0.12.4), T1-540: M.1 + (0.12.5), T1-541 : M.1 + (0.12.6), T1-542: M.1 + (0.13.1), T1-543:
- T1-544 M.1 + (0.13.3), T1-545: M.1 + (0.14.1), T1-546: M.1 + (0.14.2), T1- 547: M.1 + (0.14.3), T1-548: M.1 + (0.14.4), T1-549: M.1 + (0.15.1), T1-550: M.1 + (0.15.2), T1-551 : M.1 + (0.15.3), T1-552: M.1 + (0.15.4), T1-553: M.1 + (0.15.5), T1-554: M.1 +
- T1-555 M.1 + (0.15.7)
- T1-556 M.1 + (0.15.8)
- T1-557 M.1 + (0.15.9)
- T1-558 M.1 + (0.15.10)
- T1-559 M.1 + (0.15.11)
- T1-560 M.1 + (0.16.1)
- T1-561 M.1 + (0.17.1)
- T1-562 M.1 + (0.18.1)
- T1-563 M.1 + (0.18.2)
- T1-564 M.1 + (0.18.3)
- T1- 566 M.1 + (0.18.5)
- T1-568 M.1 + (0.20.1)
- T1-569 M.1 + (0.20.2)
- T1-570 M.1 + (0.20.3)
- T1-571 M.1 + (0.20.4)
- T1-572 M.1 +
- T1-574 M.1 + (0.21.3)
- T1-575 M.1 + (0.21.4)
- T1-576 M.1 + (0.21.5)
- T1-577 M.1 + (0.21.6)
- T1-578 M.1 + (0.21.7)
- T1-579 M.1 + (0.22.1)
- T1-580 M.1 + (0.22.2)
- T1-581 M.1 + (0.21.2)
- T1-582 M.1 + (0.22.4)
- T1-583 M.1 + (0.23.1)
- T1-584 M.1 + (0.23.2)
- T1- 585 M.1 + (0.23.3)
- T1-586 M.1 + (0.23.4)
- T1-588 M.1 + (0.24.2)
- T1-589 M.1 + (0.24.3)
- T1-591 M.1 + (0.24.5)
- T1-592 M.1 +
- T1-593 M.1 + (0.25.2), T1-594: M.1 + (0.26.1), T1-595: M.1 + (0.26.2), T1-596: M.1 + (0.26.3), T1-597: M.1 + (0.26.4), T1-598: M.1 + (0.26.5), T1-599: M.1 + (0.26.6), T1-600:
- T 1-652 M.1 + (0.28.51)
- T1-653 M.1 + (0.28.52)
- T1-654 M.1 + (0.28.53)
- T1-655 M.1 + (0.28.54)
- T 1-656 M.1 + (0.28.55)
- T1-657 M.1 + (0.28.56)
- T1-658 M.1 + (0.28.57)
- T1-659 M.1 + (0.28.58)
- T 1-660 M.1 + (0.28.59)
- T1-661 M.1 + (0.28.60
- T1-662 M.1 + (0.28.61)
- T1-701 M.1 + (K.1.67)
- T1-702 M.1 + (K.1.68)
- T1-703 M.1 + (K.1.69)
- T1-704 M.1 +
- T1-705 M.1 + (K.1.71)
- T1-706 M.1 + (K.1.72)
- T1-707 M.1 + (K.1.73)
- T1-708 M.1 +
- T1-709 M.1 + (K.1.75)
- T1-710 M.1 + (K.1.76)
- T1-711 M.1 + (K.1.77)
- T1-712 M.1 +
- T1-713 M.1 + (K.1.79)
- T1-714 M.1 + (K.1.80)
- T1-715 M.1 + (K.1.81)
- T1-716 M.1 +
- T1-717 M.1 + (K.1.83)
- T1-718 M.1 + (K.1.84)
- T1-719 M.1 + (K.1.85)
- T1-720 M.1 +
- T1-721 M.1 + (K.1.87)
- T1-722 M.1 + (K.1.88)
- T1-723 M.1 + (K.1.89)
- T1-724 M.1 +
- T1-725 M.1 + (K.1.91)
- T1-726 M.1 + (K.1.92)
- T1-727 M.1 + (K.1.93)
- T1-728 M.1 +
- T1-729 M.1 + (K.1.95)
- T1-730 M.1 + (K.1.96)
- T1-731 M.1 + (K.1.97)
- T1-732 M.1 +
- T1-733 M.1 + (K.1.99)
- T1-734 M.1 + (K.1.100)
- T1-735 M.1 + (K.1.101)
- T1-736 M.1 + (K.1.101)
- the present invention relates to ternary mixtures depicted in tabes T2 to T30: Table T2: mixtures T2-1 to T2-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.2.
- Table T3 mixtures T3-1 to T3-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.3.
- Table T4 mixtures T4-1 to T4-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.4.
- Table T5 mixtures T5-1 to T5-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.5.
- Table T6 mixtures T6-1 to T6-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.6.
- Table T7 mixtures T7-1 to T7-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.7.
- Table T8 mixtures T8-1 to T8-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.8.
- Table T9 mixtures T9-1 to T9-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.9.
- Table T10 mixtures T10-1 to T10-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.10.
- Table T11 mixtures T11-1 to T11-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.11.
- Table T12 mixtures T12-1 to T12-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.12.
- Table T13 mixtures T13-1 to T13-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.13.
- Table T14 mixtures T14-1 to T14-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.14.
- Table T15 mixtures T15-1 to T15-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.15.
- Table T16 mixtures T16-1 to T16-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.16.
- Table T17 mixtures T17-1 to T17-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.17.
- Table T18 mixtures T18-1 to T18-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.18.
- Table T19 mixtures T19-1 to T19-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.19.
- Table T20 mixtures T20-1 to T20-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.20.
- Table T21 mixtures T21-1 to T21-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.21.
- Table T23 mixtures T23-1 to T23-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.23.
- Table T24 mixtures T24-1 to T24-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.24.
- Table T25 mixtures T25-1 to T25-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.25.
- Table T26 mixtures T26-1 to T26-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.26.
- Table T27 mixtures T27-1 to T27-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.27.
- Table T28 mixtures T28-1 to T28-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.28.
- Table T29 mixtures T29-1 to T29-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.29.
- Table T30 mixtures T30-1 to T30-738, which are identical with mixtures T1-1 to T1-738 in Table T1, with the exception that the binary mixture M.1 of table M is replaced by mixture M.30.
- the amount of compound 1-1 in mixtures of Tables T1 to T30 is in the range between 70 and 150 g/ha, preferably 100 g/ha.
- the amount of compound I-2 in mixtures of Tables T 1 to T30 is in the range between 70 and 100 g/ha, preferably 85 g/ha.
- the mixtures described in Tables T1 to T30 comprise the active components 1), 2) and 3) in a synergistically effective amount.
- the weight ratio of compound I and compound III in ternary mixtures is from 500:1 to 1:500, 100:1 to 1:100, 1:1 to 1:100, or 100:1 to 1:1; preferably from 50:1 to 1:50; more preferably from 20:1 to 1:20, 1:1 to 1:20, or 20:1 to 1:1, most preferably from 5:1 to 1:5, 1:1 to 1:5, 5:1 to 1:1, 3:1 to 1:3, 1:1 to 1.3, 3:1 to 1:1, 2:1 to 1:2, or 1:1 to 1:2.
- the binary and ternary mixtures and agrochemical compositions thereof according to the invention can, in the use form as fungicides, also be present together with further active substances, e. g. with herbicides, insecticides, growth regulators, fungicides or else with fertilizers, as pre-mix or, if appropriate, not until immediately prior to use (tank mix).
- further active substances e. g. with herbicides, insecticides, growth regulators, fungicides or else with fertilizers, as pre-mix or, if appropriate, not until immediately prior to use (tank mix).
- the binary and ternary mixtures and agrochemical compositions thereof according to the invention are suitable as fungicides. They are distinguished by an outstanding effectiveness against a broad spectrum of phytopathogenic fungi, including soil-borne fungi, which derive especially from the following classes or are closely related to any of them: Ascomycota (Ascomycetes) , for example, but not limited to the genus Cocholiobolus, Colletotrichum, Fusarium, Microdochium, Penicillium, Phoma, Magnaporte, Zymoseptoria, and Pseudocercosporella; Basdiomycota (Basidiomycetes), for example, but not limited to the genus Phakospora, Puccinia, Rhizoctonia, Sphacelotheca, Tilletia, Typhula, and Ustilago; Chytridiomycota (Chytridiomycetes), for example, but not limited to the genus Chytridiales,
- Fungi imperfecti for example, but not limited to the genus Ascochyta, Diplodia, Erysiphe, Fusarium, Phomopsis, and Pyrenophora; Peronosporomycetes (syn. Oomycetes), for example but not limited to the genus Peronospora, Pythium,
- Plasmodiophoromycetes for example but not limited to the genus Plasmodiophora
- Zygomycetes for example, but not limited to the genus Rhizopus.
- Some of the binary and ternary mixtures and agrochemical compositions thereof according to the invention are systemically effective and they can be used in crop protection as foliar fungicides, fungicides for seed dressing and soil fungicides. Moreover, they are suitable for controlling harmful fungi, which inter alia occur in wood or roots of plants.
- the binary and ternary mixtures and agrochemical compositions thereof according to the invention are particularly important in the control of a multitude of phytopathogenic fungi on various cultivated plants, such as cereals, e. g. wheat, rye, barley, triticale, oats or rice; beet, e. g. sugar beet or fodder beet; fruits, such as pomes, stone fruits or soft fruits, e. g.
- plant propagation material is to be understood to denote all the generative parts of the plant such as seeds and vegetative plant material such as cuttings and tubers (e. g. potatoes), which can be used for the multiplication of the plant. This includes seeds, roots, fruits, tubers, bulbs, rhizomes, shoots, sprouts and other parts of plants, including seedlings and young plants, which are to be transplanted after germination or after emergence from soil. These young plants may also be protected before transplantation by a total or partial treatment by immersion or pouring.
- treatment of plant propagation materials with binary and ternary mixtures and agrochemical compositions thereof according to the invention is used for controlling a multitude of fungi on cereals, such as wheat, rye, barley and oats; rice, corn, cotton and soybeans.
- the term "cultivated plants” is to be understood as including plants which have been modified by breeding, mutagenesis or genetic engineering including but not limiting to agricultural biotech products on the market or in development (cf. http://cera-gmc.org/, see GM crop database therein).
- the binary and ternary mixtures and agrochemical compositions thereof according to the invention are particularly suitable for controlling the following plant diseases: rusts on soybean and cereals (e.g.
- powdery mildews on specialty crops and cereals e.g. Uncinula necator on vines, Erysiphe spp. on various specialty crops, Blumeria graminis on cereals
- leaf spots on cereals, soybean and corn e.g. Septoria tritici and S. nodorum on cereals, S. glycines on soybean, Cercospora spp. on corn and soybean.
- the mixtures and compositions as defined herein are used against Phakopsora pachyrhizi and P. meibomiae on soybeans and/or against Puccinia triticina, P. striiformis, P. hordei, P. graminis or P. recondita on wheat, barley or rye; P. coronata e.g. on oats, P. sorghi und P. polysora on corn; Puccinia spp. on other crops, e.g. P. heliathi on sunflower, P.
- the binary and ternary mixtures and agrochemical compositions thereof according to the invention are employed as such or in form of compositions by treating the fungi or the plants, plant propagation materials, such as seeds, soil, surfaces, materials or rooms to be protected from fungal attack with a fungicidally effective amount of the active substances.
- the application can be carried out both before and after the infection of the plants, plant propagation materials, such as seeds, soil, surfaces, materials or rooms by the fungi.
- Plant propagation materials may be treated with binary and ternary mixtures and agrochemical compositions thereof according to the invention as such or a composition comprising at least one compound I prophylactically either at or before planting or transplanting.
- An agrochemical composition comprises a fungicidally effective amount of a binary and ternary mixtures according to the invention.
- effective amount denotes an amount of the composition or of the compounds I, II and III, which is sufficient for controlling harmful fungi on cultivated plants or in the protection of materials and which does not result in a substantial damage to the treated plants. Such an amount can vary in a broad range and is dependent on various factors, such as the fungal species to be controlled, the treated cultivated plant or material, the climatic conditions and the specific binary or ternary mixtures used.
- the binary and ternary mixtures according to the invention can be converted into customary types of agrochemical compositions, e. g. solutions, emulsions, suspensions, dusts, powders, pastes, granules, pressings, capsules, and mixtures thereof.
- composition types are suspensions (e. g. SC, OD, FS), emulsifiable concentrates (e. g. EC), emulsions (e. g. EW, EO, ES, ME), capsules (e. g. CS, ZC), pastes, pastilles, wettable powders or dusts (e. g. WP, SP, WS, DP, DS), pressings (e. g.
- compositions types are defined in the “Catalogue of pesticide formulation types and international coding system”, Technical Monograph No. 2, 6 th Ed. May 2008, CropLife International.
- compositions are prepared in a known manner, such as described by Mollet and Grubemann, Formulation technology, Wiley VCH, Weinheim, 2001; or Knowles, New developments in crop protection product formulation, Agrow Reports DS243, T&F Informa, London, 2005.
- the binary or ternary mixtures of active compounds described herein can be prepared as compositions comprising besides the active ingredients at least one inert ingredient (auxiliary) by usual means, e. g. by the means given herein for the compositions of compounds I.
- the amounts of active substances applied are, depending on the kind of effect desired, from 0.001 to 2 kg per ha, preferably from 0.005 to 2 kg per ha, more preferably from 0.05 to 0.9 kg per ha, and in particular from 0.1 to 0.75 kg per ha.
- amounts of active compound of from 0.1 to 1000 g, preferably from 1 to 1000 g, more preferably from 1 to 100 g and most preferably from 5 to 100 g, per 100 kilogram of plant propagation material (preferably seeds) are generally required.
- the amount of active compound applied depends on the kind of application area and on the desired effect. Amounts customarily applied in the protection of materials are 0.001 g to 2 kg, preferably 0.005 g to 1 kg, of active compound per cubic meter of treated material.
- oils, wetters, adjuvants, fertilizer, or micronutrients, and further pesticides may be added to the active substances or the compositions comprising them as premix or, if appropriate not until immediately prior to use (tank mix).
- pesticides e. g. herbicides, insecticides, fungicides, growth regulators, safeners, biopesticides
- These agents can be admixed with the compositions according to the invention in a weight ratio of 1:100 to 100:1, preferably 1:10 to 10:1.
- a synergistic effect can be obtained, i.e. more than simple addition of the individual effects is obtained (synergistic mixtures).
- This can be obtained by applying the binary or ternary mixture according to the invention either jointly (e. g. as tank-mix) or separately, or in succession, wherein the time interval between the individual applications is selected to ensure that the active compound applied first still occurs at the site of action in a sufficient amount at the time of application of the further active substance(s).
- the order of application is not essential for working of the present invention.
- the time between both applications may vary e. g. between 2 hours to 7 days.
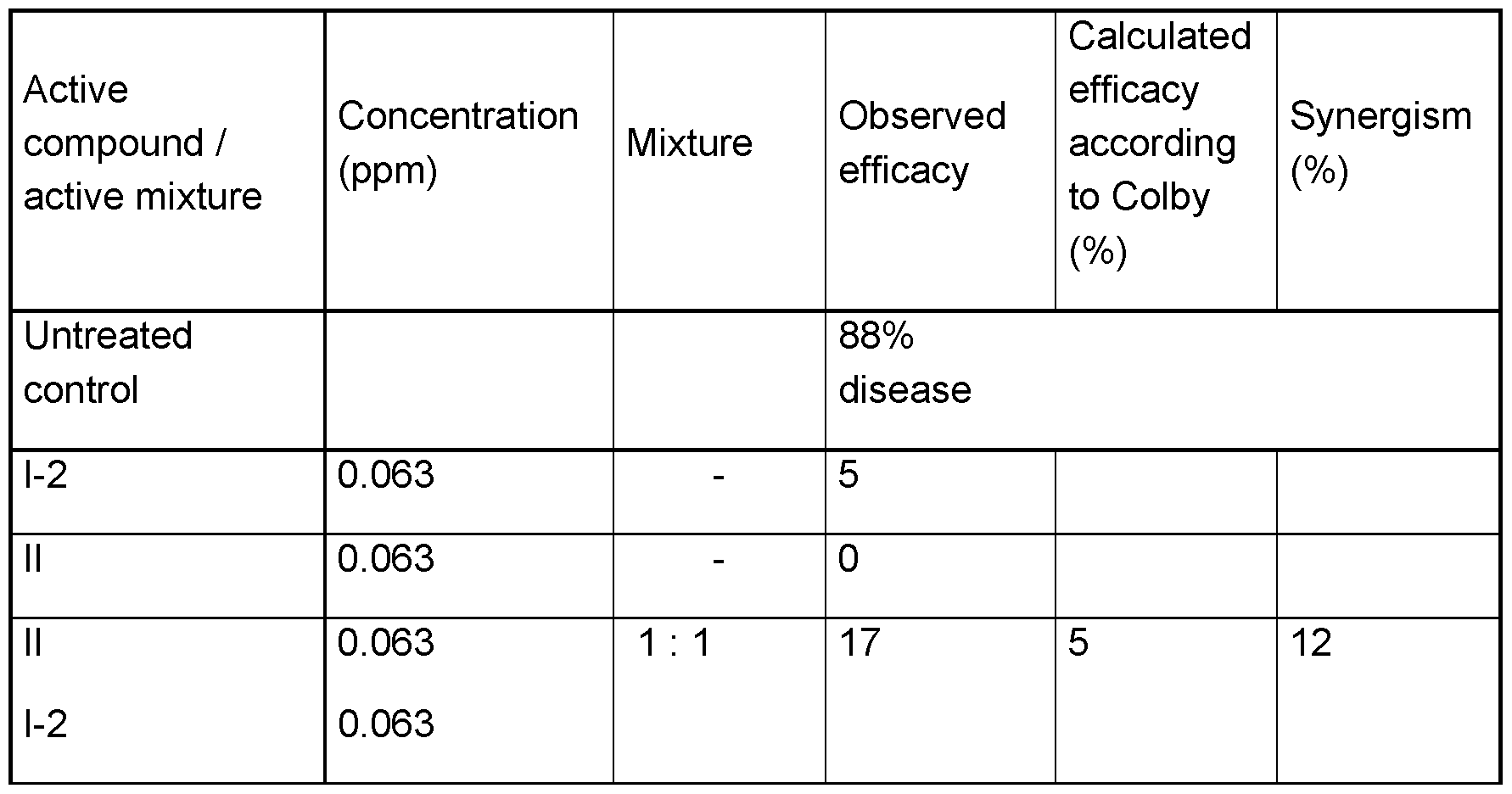
- the expected efficacies of active compound combinations may be determined using Colby's formula (Colby, S.R. "Calculating synergistic and antagonistic responses of herbicide combinations", Weeds, 15, pp.20-22, 1967) and compared with the observed efficacies.
- E x+y - x ⁇ y/100 E expected efficacy, expressed in % of the untreated control, when using the mixture of the active compounds A and B at the concentrations a and b x efficacy, expressed in % of the untreated control, when using the active compound A at the concentration a y efficacy, expressed in % of the untreated control, when using the active compound B at the concentration b.
- the active compounds were formulated separately as a stock solution having a concentration of 10000 ppm in dimethyl sulfoxide. Activity against anthracnose caused by Colletotrichum orbiculare in the microtiterplate test (COLLLA).
- the stock solutions were mixed according to the ratio, pipetted onto a micro titer plate (MTP) and diluted with water to the stated concentrations.
- MTP micro titer plate
- a spore suspension of Colletotrichum orbiculare in an aqueous bio malt solution was then added.
- the plates were placed in a water vapor-saturated chamber at a temperature of 18°C.
- the MTPs were measured at 405 nm 7 days after the inoculation.
- the measured parameters were compared to the growth of the active compound-free control variant (100%) and the fungus-free blank value to determine the relative growth in % of the pathogens in the respective active compounds. These percentages were converted into efficacies.
- An efficacy of 0 means that the growth level of the pathogens corresponds to that of the untreated control; an efficacy of 100 means that the pathogens were not growing.
- the compound was dissolved in a mixture of acetone and/or dimethylsulfoxide and the wetting agent/emulsifier Wettol, which is based on ethoxylated alkylphenoles, in a ratio (volume) solvent-emulsifier of 99 to 1 to give a total volume of 5 ml. Subsequently, water was added to total volume of 100 ml.
- Wettol which is based on ethoxylated alkylphenoles
- This stock solution was then diluted with the described solvent-emulsifier-water mixture to the final concentration given in the table below.
- Leaves of potted soybean seedlings were inoculated with spores of Phakopsora pachyrhizi. To ensure the success of the artificial inoculation, the plants were transferred to a humid chamber with a relative humidity of about 95% and 20°C to 24°C for 24 hours. The next day the plants were cultivated for 2 days in a greenhouse chamber at 23°C to 27 ° C and a relative humidity between 60% and 80%. Then the plants were sprayed to run-off with the previously described spray solution, containing the concentration of active ingredient or their mixture as described below. The plants were allowed to air-dry. Then the trial plants were cultivated for 14 days in a greenhouse chamber at 23°C to 27 ° C and a relative humidity between 60% and 80%. The extent of fungal attack on the leaves was visually assessed as % diseased leaf area.
- Leaves of potted soybean seedlings were sprayed to run-off with the previously described spray solution, containing the concentration of active ingredient or their mixture as described below.
- the plants were allowed to air-dry.
- the trial plants were cultivated for 2 days in a greenhouse chamber at 23°C to 27°C and a relative humidity between 60% and 80 %.
- the plants were inoculated with spores of Phakopsora pachyrhizi.
- the plants were transferred to a humid chamber with a relative humidity of about 95 % and 20°C to 24°C for 24 hours.
- the trial plants were cultivated for fourteen days in a greenhouse chamber at 23°C to 27 ° C and a relative humidity between 60% and 80 %.
- the extent of fungal attack on the leaves was visually assessed as % diseased leaf area.
- the disease ratings (or diseased leaf area) were converted into efficacies.
- An efficacy of 0 means that the infection level of the treated plants corresponds to that of the untreated control plants; an efficacy of 100 means that the treated plants were not infected.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Environmental Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Engineering & Computer Science (AREA)
- Zoology (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Dentistry (AREA)
- Agronomy & Crop Science (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
Description
Claims
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP22726714.3A EP4337012A1 (en) | 2021-05-11 | 2022-05-02 | Fungicidal mixtures comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| BR112023023592A BR112023023592A2 (en) | 2021-05-11 | 2022-05-02 | FUNGICIDAL MIXTURE, AGROCHEMICAL COMPOSITION, USE OF THE MIXTURE AND METHOD TO CONTROL HARMFUL PHYTOPATHOGENIC FUNGI |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP21173233.4 | 2021-05-11 | ||
| EP21173233 | 2021-05-11 | ||
| EP21173220.1 | 2021-05-11 | ||
| EP21173220 | 2021-05-11 | ||
| EP21203665 | 2021-10-20 | ||
| EP21203665.1 | 2021-10-20 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2022238157A1 true WO2022238157A1 (en) | 2022-11-17 |
Family
ID=81854600
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2022/061683 Ceased WO2022238157A1 (en) | 2021-05-11 | 2022-05-02 | Fungicidal mixtures comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
Country Status (3)
| Country | Link |
|---|---|
| EP (1) | EP4337012A1 (en) |
| BR (1) | BR112023023592A2 (en) |
| WO (1) | WO2022238157A1 (en) |
Citations (84)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0141317A2 (en) | 1983-10-21 | 1985-05-15 | BASF Aktiengesellschaft | 7-Amino-azolo[1,5-a]pyrimidines and fungicides containing them |
| EP0152031A2 (en) | 1984-02-03 | 1985-08-21 | Shionogi & Co., Ltd. | Azolyl cycloalkanol derivatives and agricultural fungicides |
| EP0226917A1 (en) | 1985-12-20 | 1987-07-01 | BASF Aktiengesellschaft | Acrylic acid esters and fungicides containing these compounds |
| EP0243970A1 (en) | 1986-05-02 | 1987-11-04 | Stauffer Chemical Company | Fungicidal pyridyl imidates |
| EP0256503A2 (en) | 1986-08-12 | 1988-02-24 | Mitsubishi Kasei Corporation | Pyridinecarboxamide derivatives and their use as fungicide |
| EP0428941A1 (en) | 1989-11-10 | 1991-05-29 | Agro-Kanesho Co., Ltd. | Hexahydrotriazine compounds and insecticides |
| EP0532022A1 (en) | 1991-09-13 | 1993-03-17 | Ube Industries, Ltd. | Acrylate compound, preparation process thereof and fungicide using the same |
| WO1994001546A1 (en) | 1992-07-01 | 1994-01-20 | Cornell Research Foundation, Inc. | Elicitor of the hypersensitive response in plants |
| WO2000046148A1 (en) | 1999-02-02 | 2000-08-10 | Sintokogio, Ltd. | Silica gel carrying titanium oxide photocatalyst in high concentration and method for preparation thereof |
| EP1028125A1 (en) | 1998-11-30 | 2000-08-16 | Isagro Ricerca S.r.l. | Dipeptide compounds having fungicidal activity and their agronomic use |
| EP1035122A1 (en) | 1999-03-11 | 2000-09-13 | Rohm And Haas Company | Heterocyclic subsituted isoxazolidines and their use as fungicides |
| WO2000065913A1 (en) | 1999-04-28 | 2000-11-09 | Takeda Chemical Industries, Ltd. | Sulfonamide derivatives |
| WO2001054501A2 (en) | 2000-01-25 | 2001-08-02 | Syngenta Participations Ag | Herbicidal composition |
| EP1122244A1 (en) | 2000-02-04 | 2001-08-08 | Sumitomo Chemical Company, Limited | Uracil compounds and their use |
| WO2001056358A2 (en) | 2000-01-28 | 2001-08-09 | Rohm And Haas Company | Enhanced propertied pesticides |
| CN1309897A (en) | 2000-02-24 | 2001-08-29 | 沈阳化工研究院 | Unsaturated oximino ether bactericide |
| WO2002022583A2 (en) | 2000-09-18 | 2002-03-21 | E. I. Du Pont De Nemours And Company | Pyridinyl amides and imides for use as fungicides |
| EP1201648A1 (en) | 1999-08-05 | 2002-05-02 | Kumiai Chemical Industry Co., Ltd. | Carbamate derivatives and agricultural/horticultural bactericides |
| WO2002040431A2 (en) | 2000-11-17 | 2002-05-23 | Dow Agrosciences Llc | Compounds having fungicidal activity and processes to make and use same |
| WO2003010149A1 (en) | 2001-07-25 | 2003-02-06 | Bayer Cropscience Ag | Pyrazolylcarboxanilides as fungicides |
| WO2003011853A1 (en) | 2001-07-30 | 2003-02-13 | Dow Agrosciences Llc | 6-aryl-4-aminopicolinates and their use as herbicides |
| WO2003014103A1 (en) | 2001-08-03 | 2003-02-20 | Bayer Cropscience S.A. | Iodobenzopyran-4-one derivatives having fungicidal activity |
| WO2003016286A1 (en) | 2001-08-17 | 2003-02-27 | Sankyo Agro Company, Limited | 3-phenoxy-4-pyridazinol derivative and herbicide composition containing the same |
| WO2003016303A1 (en) | 2001-08-20 | 2003-02-27 | Dainippon Ink And Chemicals, Inc. | Tetrazoyl oxime derivative and agricultural chemical containing the same as active ingredient |
| WO2003053145A1 (en) | 2001-12-21 | 2003-07-03 | Nissan Chemical Industries, Ltd. | Bactericidal composition |
| WO2003061388A1 (en) | 2002-01-18 | 2003-07-31 | Sumitomo Chemical Takeda Agro Company, Limited | Fused heterocyclic sulfonylurea compound, herbicide containing the same, and method of controlling weed with the same |
| WO2003066609A1 (en) | 2002-02-04 | 2003-08-14 | Bayer Cropscience Aktiengesellschaft | Disubstituted thiazolyl carboxanilides and their use as microbicides |
| WO2003074491A1 (en) | 2002-03-05 | 2003-09-12 | Syngenta Participations Ag | O-cyclopropyl-carboxanilides and their use as fungicides |
| CN1456054A (en) | 2003-03-25 | 2003-11-19 | 浙江省化工研究院 | Methoxy methyl acrylate compounds as bactericidal agent |
| WO2004049804A2 (en) | 2002-11-29 | 2004-06-17 | Syngenta Participations Ag | Fungicidal combinations for crop potection |
| WO2004083193A1 (en) | 2003-03-17 | 2004-09-30 | Sumitomo Chemical Company, Limited | Amide compound and bactericide composition containing the same |
| WO2005063721A1 (en) | 2003-12-19 | 2005-07-14 | E.I. Dupont De Nemours And Company | Herbicidal pyrimidines |
| WO2005087772A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| WO2005087773A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| WO2005120234A2 (en) | 2004-06-03 | 2005-12-22 | E.I. Dupont De Nemours And Company | Fungicidal mixtures of amidinylphenyl compounds |
| WO2005123690A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-difluoromethyl-pyrazol-4-carbonic acid-(ortho-phenyl)-anilides, and use thereof as a fungicide |
| WO2005123689A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-trifluoromethyl-pyrazole-4-carboxylic acid (ortho-phenyl)-anilides and to use thereof as fungicide |
| WO2006015866A1 (en) | 2004-08-12 | 2006-02-16 | Syngenta Participations Ag | Method for protecting useful plants or plant propagation material |
| WO2006087325A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | 5-alkoxyalkyl-6-alkyl-7-amino-azolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said substances |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| WO2007006670A1 (en) | 2005-07-07 | 2007-01-18 | Basf Aktiengesellschaft | N-thio-anthranilamid compounds and their use as pesticides |
| CN1907024A (en) | 2005-08-03 | 2007-02-07 | 浙江化工科技集团有限公司 | Methoxyl group displacement methyl acrylate compound bactericidal agent |
| WO2007082098A2 (en) | 2006-01-13 | 2007-07-19 | Dow Agrosciences Llc | 6-(poly-substituted aryl)-4-aminopicolinates and their use as herbicides |
| WO2007090624A2 (en) | 2006-02-09 | 2007-08-16 | Syngenta Participations Ag | A method of protecting a plant propagation material, a plant, and/or plant organs |
| WO2007129454A1 (en) | 2006-05-08 | 2007-11-15 | Kumiai Chemical Industry Co., Ltd. | 1,2-benzisothiazole derivative, and agricultural or horticultural plant disease-controlling agent |
| WO2008013622A2 (en) | 2006-07-27 | 2008-01-31 | E. I. Du Pont De Nemours And Company | Fungicidal azocyclic amides |
| WO2009090181A2 (en) | 2008-01-15 | 2009-07-23 | Bayer Cropscience Sa | Pesticide composition comprising a tetrazolyloxime derivative and a fungicide or an insecticide active substance |
| WO2009094442A2 (en) | 2008-01-22 | 2009-07-30 | Dow Agrosciences Llc | 5-fluoro pyrimidine derivatives |
| WO2010069882A1 (en) | 2008-12-17 | 2010-06-24 | Syngenta Participations Ag | Isoxazole derivatives for use as fungicides |
| WO2010139271A1 (en) | 2009-06-05 | 2010-12-09 | 中国中化股份有限公司 | E-type phenyl acrylic ester compounds containing substituted anilino pyrimidine group and uses thereof |
| WO2011028657A1 (en) | 2009-09-01 | 2011-03-10 | Dow Agrosciences Llc | Synergistic fungicidal compositions containing a 5-fluoropyrimidine derivative for fungal control in cereals |
| WO2011077514A1 (en) | 2009-12-22 | 2011-06-30 | 三井化学アグロ株式会社 | Plant disease control composition and method for controlling plant diseases by applying the composition |
| WO2011081174A1 (en) | 2010-01-04 | 2011-07-07 | 日本曹達株式会社 | Nitrogen-containing heterocyclic compound and agricultural/horticultural germicide |
| WO2011135833A1 (en) | 2010-04-28 | 2011-11-03 | Sumitomo Chemical Company, Limited | Plant disease control composition and its use |
| WO2012084812A1 (en) | 2010-12-20 | 2012-06-28 | Isagro Ricerca S.R.L. | Aminoindanes amides having a high fungicidal activity and their phytosanitary compositions |
| WO2012165511A1 (en) | 2011-05-31 | 2012-12-06 | クミアイ化学工業株式会社 | Method for controlling diseases in rice plant |
| WO2012168188A1 (en) | 2011-06-07 | 2012-12-13 | Bayer Intellectual Property Gmbh | Active compound combinations |
| WO2013007767A1 (en) | 2011-07-13 | 2013-01-17 | Basf Se | Fungicidal substituted 2-[2-halogenalkyl-4-(phenoxy)-phenyl]-1-[1,2,4]triazol-1-yl-ethanol compounds |
| WO2013008162A1 (en) | 2011-07-08 | 2013-01-17 | Novartis Ag | Novel trifluoromethyl-oxadiazole derivatives and their use in the treatment of disease |
| WO2013010862A1 (en) | 2011-07-15 | 2013-01-24 | Basf Se | Fungicidal alkyl-substituted 2-[2-chloro-4-(4-chloro-phenoxy)-phenyl]-1-[1,2,4]triazol-1-yl-ethanol compounds |
| WO2013024010A1 (en) | 2011-08-12 | 2013-02-21 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2013024009A1 (en) | 2011-08-12 | 2013-02-21 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2013047441A1 (en) | 2011-09-26 | 2013-04-04 | 日本曹達株式会社 | Agricultural and horticultural bactericide composition |
| WO2013047749A1 (en) | 2011-09-29 | 2013-04-04 | 三井化学アグロ株式会社 | Production method for 4, 4-difluoro-3,4-dihydroisoquinoline derivative |
| WO2013092224A1 (en) | 2011-12-21 | 2013-06-27 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi resistant to qo inhibitors |
| WO2013116251A2 (en) | 2012-02-01 | 2013-08-08 | E. I. Du Pont De Nemours And Company | Fungicidal pyrazole mixtures |
| WO2013127704A1 (en) | 2012-02-27 | 2013-09-06 | Bayer Intellectual Property Gmbh | Active compound combinations containing a thiazoylisoxazoline and a fungicide |
| WO2013162072A1 (en) | 2012-04-27 | 2013-10-31 | Sumitomo Chemical Company, Limited | Tetrazolinone compounds and its use as pesticides |
| CN103387541A (en) | 2012-05-10 | 2013-11-13 | 中国中化股份有限公司 | Preparation method of substituted pyrazolylether compound |
| WO2014060177A1 (en) | 2012-10-16 | 2014-04-24 | Syngenta Participations Ag | Fungicidal compositions |
| EP2865265A1 (en) | 2014-02-13 | 2015-04-29 | Bayer CropScience AG | Active compound combinations comprising phenylamidine compounds and biological control agents |
| WO2015065922A1 (en) | 2013-10-28 | 2015-05-07 | Dexcom, Inc. | Devices used in connection with continuous analyte monitoring that provide the user with one or more notifications, and related methods |
| WO2015185485A1 (en) | 2014-06-06 | 2015-12-10 | Basf Se | Use of substituted oxadiazoles for combating phytopathogenic fungi |
| WO2016156241A1 (en) | 2015-04-02 | 2016-10-06 | Syngenta Participations Ag | Herbicidal mixtures |
| WO2016162265A1 (en) | 2015-04-07 | 2016-10-13 | Syngenta Participations Ag | Herbicidal mixtures |
| WO2016202742A1 (en) | 2015-06-15 | 2016-12-22 | Bayer Cropscience Aktiengesellschaft | Halogen-substituted phenoxyphenylamidines and the use thereof as fungicides |
| WO2017211649A1 (en) | 2016-06-09 | 2017-12-14 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018109002A1 (en) | 2016-12-14 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Active compound combinations |
| WO2018108977A1 (en) | 2016-12-14 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Active compound combinations |
| WO2021069707A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Aktiengesellschaft | Active compound combinations |
| WO2021069704A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Aktiengesellschaft | Active compound combinations |
| WO2021069702A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Aktiengesellschaft | Active compound combinations |
| WO2021069706A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Aktiengesellschaft | Active compound combinations |
| WO2022017836A1 (en) * | 2020-07-20 | 2022-01-27 | BASF Agro B.V. | Fungicidal compositions comprising (r)-2-[4-(4-chlorophenoxy)-2-(trifluoromethyl)phenyl]-1- (1,2,4-triazol-1-yl)propan-2-ol |
-
2022
- 2022-05-02 BR BR112023023592A patent/BR112023023592A2/en not_active Application Discontinuation
- 2022-05-02 WO PCT/EP2022/061683 patent/WO2022238157A1/en not_active Ceased
- 2022-05-02 EP EP22726714.3A patent/EP4337012A1/en not_active Withdrawn
Patent Citations (84)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0141317A2 (en) | 1983-10-21 | 1985-05-15 | BASF Aktiengesellschaft | 7-Amino-azolo[1,5-a]pyrimidines and fungicides containing them |
| EP0152031A2 (en) | 1984-02-03 | 1985-08-21 | Shionogi & Co., Ltd. | Azolyl cycloalkanol derivatives and agricultural fungicides |
| EP0226917A1 (en) | 1985-12-20 | 1987-07-01 | BASF Aktiengesellschaft | Acrylic acid esters and fungicides containing these compounds |
| EP0243970A1 (en) | 1986-05-02 | 1987-11-04 | Stauffer Chemical Company | Fungicidal pyridyl imidates |
| EP0256503A2 (en) | 1986-08-12 | 1988-02-24 | Mitsubishi Kasei Corporation | Pyridinecarboxamide derivatives and their use as fungicide |
| EP0428941A1 (en) | 1989-11-10 | 1991-05-29 | Agro-Kanesho Co., Ltd. | Hexahydrotriazine compounds and insecticides |
| EP0532022A1 (en) | 1991-09-13 | 1993-03-17 | Ube Industries, Ltd. | Acrylate compound, preparation process thereof and fungicide using the same |
| WO1994001546A1 (en) | 1992-07-01 | 1994-01-20 | Cornell Research Foundation, Inc. | Elicitor of the hypersensitive response in plants |
| EP1028125A1 (en) | 1998-11-30 | 2000-08-16 | Isagro Ricerca S.r.l. | Dipeptide compounds having fungicidal activity and their agronomic use |
| WO2000046148A1 (en) | 1999-02-02 | 2000-08-10 | Sintokogio, Ltd. | Silica gel carrying titanium oxide photocatalyst in high concentration and method for preparation thereof |
| EP1035122A1 (en) | 1999-03-11 | 2000-09-13 | Rohm And Haas Company | Heterocyclic subsituted isoxazolidines and their use as fungicides |
| WO2000065913A1 (en) | 1999-04-28 | 2000-11-09 | Takeda Chemical Industries, Ltd. | Sulfonamide derivatives |
| EP1201648A1 (en) | 1999-08-05 | 2002-05-02 | Kumiai Chemical Industry Co., Ltd. | Carbamate derivatives and agricultural/horticultural bactericides |
| WO2001054501A2 (en) | 2000-01-25 | 2001-08-02 | Syngenta Participations Ag | Herbicidal composition |
| WO2001056358A2 (en) | 2000-01-28 | 2001-08-09 | Rohm And Haas Company | Enhanced propertied pesticides |
| EP1122244A1 (en) | 2000-02-04 | 2001-08-08 | Sumitomo Chemical Company, Limited | Uracil compounds and their use |
| CN1309897A (en) | 2000-02-24 | 2001-08-29 | 沈阳化工研究院 | Unsaturated oximino ether bactericide |
| WO2002022583A2 (en) | 2000-09-18 | 2002-03-21 | E. I. Du Pont De Nemours And Company | Pyridinyl amides and imides for use as fungicides |
| WO2002040431A2 (en) | 2000-11-17 | 2002-05-23 | Dow Agrosciences Llc | Compounds having fungicidal activity and processes to make and use same |
| WO2003010149A1 (en) | 2001-07-25 | 2003-02-06 | Bayer Cropscience Ag | Pyrazolylcarboxanilides as fungicides |
| WO2003011853A1 (en) | 2001-07-30 | 2003-02-13 | Dow Agrosciences Llc | 6-aryl-4-aminopicolinates and their use as herbicides |
| WO2003014103A1 (en) | 2001-08-03 | 2003-02-20 | Bayer Cropscience S.A. | Iodobenzopyran-4-one derivatives having fungicidal activity |
| WO2003016286A1 (en) | 2001-08-17 | 2003-02-27 | Sankyo Agro Company, Limited | 3-phenoxy-4-pyridazinol derivative and herbicide composition containing the same |
| WO2003016303A1 (en) | 2001-08-20 | 2003-02-27 | Dainippon Ink And Chemicals, Inc. | Tetrazoyl oxime derivative and agricultural chemical containing the same as active ingredient |
| WO2003053145A1 (en) | 2001-12-21 | 2003-07-03 | Nissan Chemical Industries, Ltd. | Bactericidal composition |
| WO2003061388A1 (en) | 2002-01-18 | 2003-07-31 | Sumitomo Chemical Takeda Agro Company, Limited | Fused heterocyclic sulfonylurea compound, herbicide containing the same, and method of controlling weed with the same |
| WO2003066609A1 (en) | 2002-02-04 | 2003-08-14 | Bayer Cropscience Aktiengesellschaft | Disubstituted thiazolyl carboxanilides and their use as microbicides |
| WO2003074491A1 (en) | 2002-03-05 | 2003-09-12 | Syngenta Participations Ag | O-cyclopropyl-carboxanilides and their use as fungicides |
| WO2004049804A2 (en) | 2002-11-29 | 2004-06-17 | Syngenta Participations Ag | Fungicidal combinations for crop potection |
| WO2004083193A1 (en) | 2003-03-17 | 2004-09-30 | Sumitomo Chemical Company, Limited | Amide compound and bactericide composition containing the same |
| CN1456054A (en) | 2003-03-25 | 2003-11-19 | 浙江省化工研究院 | Methoxy methyl acrylate compounds as bactericidal agent |
| WO2005063721A1 (en) | 2003-12-19 | 2005-07-14 | E.I. Dupont De Nemours And Company | Herbicidal pyrimidines |
| WO2005087772A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| WO2005087773A1 (en) | 2004-03-10 | 2005-09-22 | Basf Aktiengesellschaft | 5,6-dialkyl-7-amino-triazolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said compounds |
| WO2005120234A2 (en) | 2004-06-03 | 2005-12-22 | E.I. Dupont De Nemours And Company | Fungicidal mixtures of amidinylphenyl compounds |
| WO2005123690A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-difluoromethyl-pyrazol-4-carbonic acid-(ortho-phenyl)-anilides, and use thereof as a fungicide |
| WO2005123689A1 (en) | 2004-06-18 | 2005-12-29 | Basf Aktiengesellschaft | 1-methyl-3-trifluoromethyl-pyrazole-4-carboxylic acid (ortho-phenyl)-anilides and to use thereof as fungicide |
| WO2006015866A1 (en) | 2004-08-12 | 2006-02-16 | Syngenta Participations Ag | Method for protecting useful plants or plant propagation material |
| WO2006087325A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | 5-alkoxyalkyl-6-alkyl-7-amino-azolopyrimidines, method for their production, their use for controlling pathogenic fungi and agents containing said substances |
| WO2006087343A1 (en) | 2005-02-16 | 2006-08-24 | Basf Aktiengesellschaft | Pyrazole carboxylic acid anilides, method for the production thereof and agents containing them for controlling pathogenic fungi |
| WO2007006670A1 (en) | 2005-07-07 | 2007-01-18 | Basf Aktiengesellschaft | N-thio-anthranilamid compounds and their use as pesticides |
| CN1907024A (en) | 2005-08-03 | 2007-02-07 | 浙江化工科技集团有限公司 | Methoxyl group displacement methyl acrylate compound bactericidal agent |
| WO2007082098A2 (en) | 2006-01-13 | 2007-07-19 | Dow Agrosciences Llc | 6-(poly-substituted aryl)-4-aminopicolinates and their use as herbicides |
| WO2007090624A2 (en) | 2006-02-09 | 2007-08-16 | Syngenta Participations Ag | A method of protecting a plant propagation material, a plant, and/or plant organs |
| WO2007129454A1 (en) | 2006-05-08 | 2007-11-15 | Kumiai Chemical Industry Co., Ltd. | 1,2-benzisothiazole derivative, and agricultural or horticultural plant disease-controlling agent |
| WO2008013622A2 (en) | 2006-07-27 | 2008-01-31 | E. I. Du Pont De Nemours And Company | Fungicidal azocyclic amides |
| WO2009090181A2 (en) | 2008-01-15 | 2009-07-23 | Bayer Cropscience Sa | Pesticide composition comprising a tetrazolyloxime derivative and a fungicide or an insecticide active substance |
| WO2009094442A2 (en) | 2008-01-22 | 2009-07-30 | Dow Agrosciences Llc | 5-fluoro pyrimidine derivatives |
| WO2010069882A1 (en) | 2008-12-17 | 2010-06-24 | Syngenta Participations Ag | Isoxazole derivatives for use as fungicides |
| WO2010139271A1 (en) | 2009-06-05 | 2010-12-09 | 中国中化股份有限公司 | E-type phenyl acrylic ester compounds containing substituted anilino pyrimidine group and uses thereof |
| WO2011028657A1 (en) | 2009-09-01 | 2011-03-10 | Dow Agrosciences Llc | Synergistic fungicidal compositions containing a 5-fluoropyrimidine derivative for fungal control in cereals |
| WO2011077514A1 (en) | 2009-12-22 | 2011-06-30 | 三井化学アグロ株式会社 | Plant disease control composition and method for controlling plant diseases by applying the composition |
| WO2011081174A1 (en) | 2010-01-04 | 2011-07-07 | 日本曹達株式会社 | Nitrogen-containing heterocyclic compound and agricultural/horticultural germicide |
| WO2011135833A1 (en) | 2010-04-28 | 2011-11-03 | Sumitomo Chemical Company, Limited | Plant disease control composition and its use |
| WO2012084812A1 (en) | 2010-12-20 | 2012-06-28 | Isagro Ricerca S.R.L. | Aminoindanes amides having a high fungicidal activity and their phytosanitary compositions |
| WO2012165511A1 (en) | 2011-05-31 | 2012-12-06 | クミアイ化学工業株式会社 | Method for controlling diseases in rice plant |
| WO2012168188A1 (en) | 2011-06-07 | 2012-12-13 | Bayer Intellectual Property Gmbh | Active compound combinations |
| WO2013008162A1 (en) | 2011-07-08 | 2013-01-17 | Novartis Ag | Novel trifluoromethyl-oxadiazole derivatives and their use in the treatment of disease |
| WO2013007767A1 (en) | 2011-07-13 | 2013-01-17 | Basf Se | Fungicidal substituted 2-[2-halogenalkyl-4-(phenoxy)-phenyl]-1-[1,2,4]triazol-1-yl-ethanol compounds |
| WO2013010862A1 (en) | 2011-07-15 | 2013-01-24 | Basf Se | Fungicidal alkyl-substituted 2-[2-chloro-4-(4-chloro-phenoxy)-phenyl]-1-[1,2,4]triazol-1-yl-ethanol compounds |
| WO2013024010A1 (en) | 2011-08-12 | 2013-02-21 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2013024009A1 (en) | 2011-08-12 | 2013-02-21 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2013047441A1 (en) | 2011-09-26 | 2013-04-04 | 日本曹達株式会社 | Agricultural and horticultural bactericide composition |
| WO2013047749A1 (en) | 2011-09-29 | 2013-04-04 | 三井化学アグロ株式会社 | Production method for 4, 4-difluoro-3,4-dihydroisoquinoline derivative |
| WO2013092224A1 (en) | 2011-12-21 | 2013-06-27 | Basf Se | Use of strobilurin type compounds for combating phytopathogenic fungi resistant to qo inhibitors |
| WO2013116251A2 (en) | 2012-02-01 | 2013-08-08 | E. I. Du Pont De Nemours And Company | Fungicidal pyrazole mixtures |
| WO2013127704A1 (en) | 2012-02-27 | 2013-09-06 | Bayer Intellectual Property Gmbh | Active compound combinations containing a thiazoylisoxazoline and a fungicide |
| WO2013162072A1 (en) | 2012-04-27 | 2013-10-31 | Sumitomo Chemical Company, Limited | Tetrazolinone compounds and its use as pesticides |
| CN103387541A (en) | 2012-05-10 | 2013-11-13 | 中国中化股份有限公司 | Preparation method of substituted pyrazolylether compound |
| WO2014060177A1 (en) | 2012-10-16 | 2014-04-24 | Syngenta Participations Ag | Fungicidal compositions |
| WO2015065922A1 (en) | 2013-10-28 | 2015-05-07 | Dexcom, Inc. | Devices used in connection with continuous analyte monitoring that provide the user with one or more notifications, and related methods |
| EP2865265A1 (en) | 2014-02-13 | 2015-04-29 | Bayer CropScience AG | Active compound combinations comprising phenylamidine compounds and biological control agents |
| WO2015185485A1 (en) | 2014-06-06 | 2015-12-10 | Basf Se | Use of substituted oxadiazoles for combating phytopathogenic fungi |
| WO2016156241A1 (en) | 2015-04-02 | 2016-10-06 | Syngenta Participations Ag | Herbicidal mixtures |
| WO2016162265A1 (en) | 2015-04-07 | 2016-10-13 | Syngenta Participations Ag | Herbicidal mixtures |
| WO2016202742A1 (en) | 2015-06-15 | 2016-12-22 | Bayer Cropscience Aktiengesellschaft | Halogen-substituted phenoxyphenylamidines and the use thereof as fungicides |
| WO2017211649A1 (en) | 2016-06-09 | 2017-12-14 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018109002A1 (en) | 2016-12-14 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Active compound combinations |
| WO2018108977A1 (en) | 2016-12-14 | 2018-06-21 | Bayer Cropscience Aktiengesellschaft | Active compound combinations |
| WO2021069707A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Aktiengesellschaft | Active compound combinations |
| WO2021069704A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Aktiengesellschaft | Active compound combinations |
| WO2021069702A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Aktiengesellschaft | Active compound combinations |
| WO2021069706A1 (en) | 2019-10-11 | 2021-04-15 | Bayer Aktiengesellschaft | Active compound combinations |
| WO2022017836A1 (en) * | 2020-07-20 | 2022-01-27 | BASF Agro B.V. | Fungicidal compositions comprising (r)-2-[4-(4-chlorophenoxy)-2-(trifluoromethyl)phenyl]-1- (1,2,4-triazol-1-yl)propan-2-ol |
Non-Patent Citations (6)
| Title |
|---|
| "Method for the control of fungicide-resistant Phakopsora pachyrhizi ED - Darl Kuhn", IP.COM, IP.COM INC., WEST HENRIETTA, NY, US, 18 April 2019 (2019-04-18), XP013182938, ISSN: 1533-0001 * |
| "Technical Monograph", 2008, CROPLIFE INTERNATIONAL, article "Catalogue of pesticide formulation types and international coding system" |
| CAN. J. PLANT SCI., vol. 48, no. 6, 1968, pages 587 - 94 |
| COLBY, S.R.: "Calculating synergistic and antagonistic responses of herbicide combinations", WEEDS, vol. 15, 1967, pages 20 - 22, XP001112961 |
| KNOWLES: "Agrow Reports DS243", 2005, T&F INFORMA, article "New developments in crop protection product formulation" |
| MOLLETGRUBEMANN: "Formulation technology", 2001, WILEY VCH |
Also Published As
| Publication number | Publication date |
|---|---|
| BR112023023592A2 (en) | 2024-03-12 |
| EP4337012A1 (en) | 2024-03-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3383180B1 (en) | Substituted oxadiazoles for combating phytopathogenic fungi | |
| WO2017153200A1 (en) | Fungicidal mixtures iii comprising strobilurin-type fungicides | |
| RU2018131441A (en) | MIXTURES AND COMPOSITIONS CONTAINING PAENIBACILLUS OR FUSARICIDINES AND CHEMICAL PESTICIDES | |
| CN108347937A (en) | Qu Dai oxadiazoles for preventing and kill off plant pathogenic fungi | |
| JP7649254B2 (en) | Highly spreadable ULV formulation for pesticide compound II | |
| BR112018009539B1 (en) | Use of compounds of formula I, compounds, mixture, agrochemical composition, use of compounds and method to combat harmful phytopathogenic fungi | |
| EP3606912A1 (en) | Substituted oxadiazoles for combating phytopathogenic fungi | |
| ES3009092T3 (en) | Herbicidal compositions | |
| CN116669551A (en) | Agrochemical compositions with improved drift, absorption and rain resistance properties | |
| EP4240158A1 (en) | Low drift, rainfastness, high uptake and ulv tank mix adjuvant formulation | |
| WO2022238194A1 (en) | Herbicide/safener combination based on safeners from the class of substituted [(1,5-diphenyl-1h-1,2,4-triazol-3-yl)oxy]acetic acids and their salts | |
| CN111801014B (en) | Fungicidal compositions of cloroxen | |
| US11839214B2 (en) | Fungicidal mixture comprising substituted pyridines | |
| CN113993847A (en) | Fungicidal N- (pyridin-3-yl) carboxamides | |
| WO2019154665A1 (en) | New pyridine carboxamides | |
| WO2022238157A1 (en) | Fungicidal mixtures comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles | |
| CN121038608A (en) | Sophorolipids and their derivatives are used as tank-mixing additives and adjuvants in crop protection formulations. | |
| BR112019023117B1 (en) | FUNGICIDAL MIXTURE, AGROCHEMICAL COMPOSITION, USE OF THE FUNGICIDAL MIXTURE OR AGROCHEMICAL COMPOSITION AND METHOD FOR CONTROLLING HARMFUL PHYTOPATHOGENIC FUNGI | |
| BR122023006464B1 (en) | FUNGICIDAL MIXTURE, AGROCHEMICAL COMPOSITION, USE OF THE FUNGICIDAL MIXTURE AND AGROCHEMICAL COMPOSITION AND METHOD FOR CONTROLING HARMFUL PHYTOPATHOGENIC FUNGI | |
| BR122023006510B1 (en) | FUNGICIDAL MIXTURE, AGROCHEMICAL COMPOSITION, USE OF THE FUNGICIDAL MIXTURE AND AGROCHEMICAL COMPOSITION AND METHOD FOR CONTROLING HARMFUL PHYTOPATHOGENIC FUNGI | |
| BR122023004527B1 (en) | FUNGICIDAL COMPOSITIONS, METHODS FOR CONTROLLING HARMFUL PHYTOPATHOGENIC FUNGI, FOR IMPROVING PLANT HEALTH AND FOR PROTECTING PLANT PROPAGATION MATERIAL, COATED SEED AND USE OF A COMPOSITION | |
| BR112020016805B1 (en) | FUNGICIDAL COMPOSITIONS, METHODS FOR CONTROLLING HARMFUL PHYTOPATHOGENIC FUNGI, FOR IMPROVING PLANT HEALTH AND FOR PROTECTING PLANT PROPAGATION MATERIAL, COATED SEED AND USE OF A COMPOSITION |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 22726714 Country of ref document: EP Kind code of ref document: A1 |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112023023592 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2022726714 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2022726714 Country of ref document: EP Effective date: 20231211 |
|
| ENP | Entry into the national phase |
Ref document number: 112023023592 Country of ref document: BR Kind code of ref document: A2 Effective date: 20231110 |