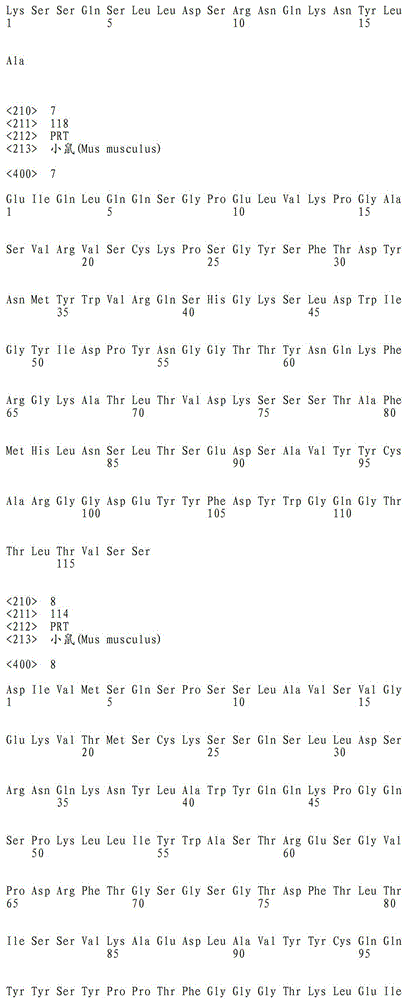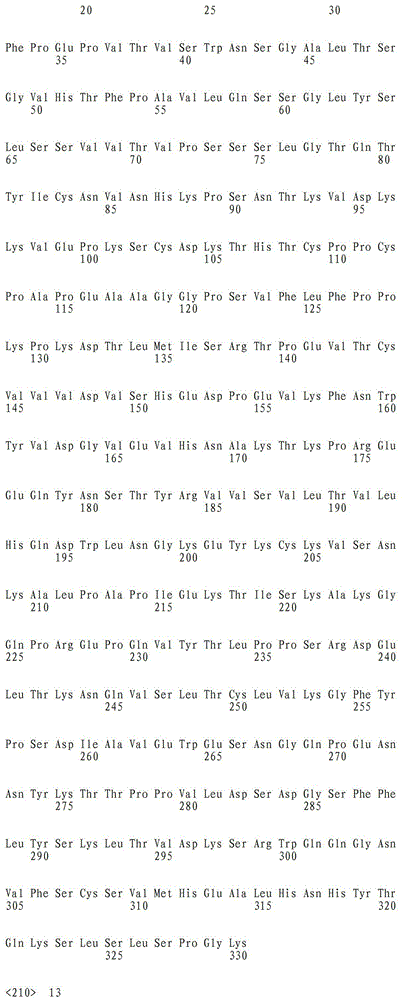CN102918061A - Antibodies against human CSF-1R and uses thereof - Google Patents
Antibodies against human CSF-1R and uses thereof Download PDFInfo
- Publication number
- CN102918061A CN102918061A CN2011800125069A CN201180012506A CN102918061A CN 102918061 A CN102918061 A CN 102918061A CN 2011800125069 A CN2011800125069 A CN 2011800125069A CN 201180012506 A CN201180012506 A CN 201180012506A CN 102918061 A CN102918061 A CN 102918061A
- Authority
- CN
- China
- Prior art keywords
- antibody
- csf
- seq
- amino acid
- region
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2866—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against receptors for cytokines, lymphokines, interferons
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K39/395—Antibodies; Immunoglobulins; Immune serum, e.g. antilymphocytic serum
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/08—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease
- A61P19/10—Drugs for skeletal disorders for bone diseases, e.g. rachitism, Paget's disease for osteoporosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/11—DNA or RNA fragments; Modified forms thereof; Non-coding nucleic acids having a biological activity
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/73—Inducing cell death, e.g. apoptosis, necrosis or inhibition of cell proliferation
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/70—Immunoglobulins specific features characterized by effect upon binding to a cell or to an antigen
- C07K2317/76—Antagonist effect on antigen, e.g. neutralization or inhibition of binding
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Immunology (AREA)
- Genetics & Genomics (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Engineering & Computer Science (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Molecular Biology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biophysics (AREA)
- Biochemistry (AREA)
- Physical Education & Sports Medicine (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Rheumatology (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Biomedical Technology (AREA)
- Microbiology (AREA)
- Zoology (AREA)
- Oncology (AREA)
- Wood Science & Technology (AREA)
- Biotechnology (AREA)
- General Engineering & Computer Science (AREA)
- Physics & Mathematics (AREA)
- Plant Pathology (AREA)
- Pain & Pain Management (AREA)
- Hematology (AREA)
- Mycology (AREA)
- Epidemiology (AREA)
- Peptides Or Proteins (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
发明领域 field of invention
本发明涉及针对人CSF-1R的抗体(CSF-1R抗体)、其生成方法、含有所述抗体的药物组合物、及其用途。The present invention relates to an antibody against human CSF-1R (CSF-1R antibody), a production method thereof, a pharmaceutical composition containing the antibody, and uses thereof.
发明背景Background of the invention
CSF-1受体(CSF-1R;同义词:M-CSF受体;巨噬细胞集落刺激因子1受体、EC 2.7.10.1、Fms原癌基因、c-fms、Swiss Prot P07333、CD115)自1986年以来是已知的(Coussens L.等,Nature 320(1986)277-280)。CSF-1R是一种生长因子,并且由c-fms原癌基因编码(综述见例如Roth,P.和Stanley,E.R.,Curr.Top.Microbiol.Immunol.181(1992)141-67)。CSF-1 receptor (CSF-1R; synonyms: M-CSF receptor; macrophage colony-
CSF-1R是M-CSF(巨噬细胞集落刺激因子,又称作CSF-1)的受体,并且介导此细胞因子的生物学效应(Sherr,C.J.等,Cell 41(1985)665-676)。集落刺激因子-1受体(又称作c-fms)的克隆第一次记载于Roussel,M.F.等,Nature325(1987)549-552。在该出版物中,显示了CSF-1R具有转化潜力,其依赖于蛋白质的C端尾部的变化,包含抑制性酪氨酸969磷酸化的丧失,其结合Cbl,并且由此调节受体下调(Lee,P.S.等,Embo J.18(1999)3616-3628)。CSF-1R is a receptor for M-CSF (macrophage colony-stimulating factor, also known as CSF-1), and mediates the biological effects of this cytokine (Sherr, C.J. et al., Cell 41(1985) 665-676 ). The cloning of the colony-stimulating factor-1 receptor (also known as c-fms) was first described by Roussel, M.F. et al., Nature 325 (1987) 549-552. In this publication, it was shown that CSF-1R has a transforming potential that is dependent on changes in the C-terminal tail of the protein, including loss of phosphorylation of the inhibitory tyrosine 969, which binds Cbl and thereby regulates receptor downregulation ( Lee, P.S. et al., Embo J. 18(1999) 3616-3628).
CSF-1R是一种单链、跨膜受体酪氨酸激酶(RTK)和以受体的胞外部分中的重复Ig域为特征的含有免疫球蛋白(Ig)基序的RTK家族的一名成员。胞内蛋白质酪氨酸激酶域以独特插入域中断,所述独特插入域也存在于包括血小板衍生的生长因子受体(PDGFR)、干细胞生长因子受体(c-Kit)和fins样细胞因子受体(FLT3)的其它相关RTK III类家族成员中。尽管此生长因子受体家族间有结构同源性,但是它们具有截然不同的组织特异性功能。CSF-1R主要在单核细胞谱系的细胞上及在女性生殖道和胎盘中表达。另外,已经在皮肤中的朗格汉斯细胞,即一种平滑肌细胞子集(Inaba,T.等,J.Biol.Chem.267(1992)5693-5699)、B细胞(Baker,A.H.等,Oncogene 8(1993)371-378)和小胶质(Sawada,M.等,Brain Res.509(1990)119-124)中报告了CSF-1R的表达。CSF-1R is a single-chain, transmembrane receptor tyrosine kinase (RTK) and member of the immunoglobulin (Ig) motif-containing RTK family characterized by repeated Ig domains in the extracellular portion of the receptor members. Intracellular protein tyrosine kinase domains are interrupted by unique insertion domains also present in receptors including platelet-derived growth factor receptor (PDGFR), stem cell growth factor receptor (c-Kit), and fins-like cytokine receptors. among other related RTK class III family members of the FLT3. Despite the structural homology between this family of growth factor receptors, they have distinct tissue-specific functions. CSF-1R is mainly expressed on cells of the monocyte lineage and in the female reproductive tract and placenta. In addition, Langerhans cells, a subset of smooth muscle cells already in the skin (Inaba, T. et al., J. Biol. Chem. 267 (1992) 5693-5699), B cells (Baker, A.H. et al., Expression of CSF-1R has been reported in Oncogene 8 (1993) 371-378) and in microglia (Sawada, M. et al., Brain Res. 509 (1990) 119-124).
CSF-1R信号传导的主要生物学效应是造血前体细胞向巨噬细胞谱系(包括破骨细胞)的分化、增殖、迁移、和存活。CSF-1R的激活由其配体M-CSF介导。M-CSF对CSF-1R的结合诱导同二聚体的形成和通过酪氨酸磷酸化实现的激酶活化(Stanley,E.R.等,Mol.Reprod.Dev.46(1997)4-10)。别的信号传导由分别连接PI3K/AKT和Ras/MAPK途径的PI3K的p85亚基和Grb2介导。这两种重要的信号传导途径可以调节增殖、存活和凋亡。结合CSF-1R的磷酸化的胞内域的其它信号传导分子包括STAT1、STAT3、PLCy、和Cbl(Bourette,R.P.和Rohrschneider,L.R.,Growth Factors 17(2000)155-166)。The major biological effects of CSF-1R signaling are the differentiation, proliferation, migration, and survival of hematopoietic precursor cells to the macrophage lineage, including osteoclasts. Activation of CSF-1R is mediated by its ligand M-CSF. M-CSF binding to CSF-IR induces homodimer formation and kinase activation by tyrosine phosphorylation (Stanley, E.R. et al., Mol. Reprod. Dev. 46 (1997) 4-10). Additional signaling is mediated by the p85 subunit of PI3K and Grb2 linking the PI3K/AKT and Ras/MAPK pathways, respectively. These two important signaling pathways regulate proliferation, survival and apoptosis. Other signaling molecules that bind the phosphorylated intracellular domain of CSF-1R include STAT1, STAT3, PLCy, and Cbl (Bourette, R.P. and Rohrschneider, L.R., Growth Factors 17 (2000) 155-166).
CSF-1R信号传导在免疫应答中、在骨重建(bone remodeling)中及在生殖系统中具有生理学作用。已经显示了M-CSF-1(Pollard,J.W.,Mol.Reprod.Dev.46(1997)54-61)或CSF-1R(Dai,X.M.等,Blood 99(2002)111-120)的敲除动物具有与CSF-1R在相应细胞类型中的作用一致的骨硬化(osteopetrotic)、造血、组织巨噬细胞、和生殖表型。CSF-1R signaling has physiological roles in the immune response, in bone remodeling and in the reproductive system. Knockout animals of M-CSF-1 (Pollard, J.W., Mol. Reprod. Dev. 46 (1997) 54-61) or CSF-1R (Dai, X.M. et al., Blood 99 (2002) 111-120) have been shown Osteopetrotic, hematopoietic, tissue macrophage, and reproductive phenotypes consistent with CSF-IR roles in the corresponding cell types.
Sherr,C.J.等,Blood 73(1989)1786-1793描述了涉及抑制CSF-1活性的一些针对CSF-1R的抗体(见Sherr,C.J.等,Blood 73(1989)1786-1793)。Ashum,R.A.等,Blood 73(1989)827-837涉及CSF-1R抗体。Lenda,D.M.等,Journal of immunology 170(2003)3254-3262涉及CSF-1缺陷型小鼠中降低的巨噬细胞募集、增殖、和活化导致肾炎症期间降低的肾小管凋亡。Kitaura,H.等,Journal of dental research 87(2008)396-400提及一种抑制口腔正畸牙移动的抗CSF-1抗体。WO 2001/030381在仅披露CSF-1反义核苷酸的情况中提及包括反义核苷酸和抗体的CSF-1活性抑制剂。WO 2004/045532涉及通过M-CSF拮抗剂(仅披露抗CSF-1抗体作为拮抗剂)对转移癌的转移和骨损失预防和治疗。WO 2005/046657涉及通过抗CSF-1抗体治疗炎性肠病。US2002/0141994涉及集落刺激因子的抑制剂。WO 2006/096489涉及通过抗CSF-1抗体治疗类风湿性关节炎。Sherr, C.J. et al., Blood 73 (1989) 1786-1793 describe some antibodies against CSF-1R involved in the inhibition of CSF-1 activity (see Sherr, C.J. et al., Blood 73 (1989) 1786-1793). Ashum, R.A. et al., Blood 73 (1989) 827-837 relates to CSF-1R antibodies. Lenda, D.M. et al., Journal of immunology 170 (2003) 3254-3262 implicates reduced macrophage recruitment, proliferation, and activation in CSF-1 deficient mice leading to reduced tubular apoptosis during renal inflammation. Kitaura, H. et al., Journal of dental research 87 (2008) 396-400 mention an anti-CSF-1 antibody that inhibits orthodontic tooth movement. WO 2001/030381 mentions CSF-1 activity inhibitors including antisense nucleotides and antibodies in the context of disclosing only CSF-1 antisense nucleotides. WO 2004/045532 relates to metastasis and bone loss prevention and treatment of metastatic cancer by M-CSF antagonists (only anti-CSF-1 antibodies are disclosed as antagonists). WO 2005/046657 relates to the treatment of inflammatory bowel disease by anti-CSF-1 antibodies. US2002/0141994 relates to inhibitors of colony stimulating factor. WO 2006/096489 relates to the treatment of rheumatoid arthritis by anti-CSF-1 antibodies.
WO 2009/026303和WO 2009/112245涉及抗CSF-1R抗体。WO 2009/026303 and WO 2009/112245 relate to anti-CSF-1R antibodies.
发明概述Summary of the invention
本发明包括一种结合人CSF-1R的抗体,其特征在于与保藏抗体DSMACC2921结合相同表位。The present invention includes an antibody binding to human CSF-1R, which is characterized in that it binds to the same epitope as the deposited antibody DSMACC2921.
在一个实施方案中,所述抗体特征在于作为重链可变域CDR3区包含SEQ ID NO:1的CDR3区。In one embodiment, the antibody is characterized in comprising the CDR3 region of SEQ ID NO: 1 as the heavy chain variable domain CDR3 region.
在一个实施方案中,所述抗体特征在于:In one embodiment, the antibody is characterized by:
a)重链可变域包含SEQ ID NO:1的CDR3区、SEQ ID NO:2的CDR2区、和SEQ ID NO:3的CDR1区,且轻链可变域包含SEQ ID NO:4的CDR3区、SEQID NO:5的CDR2区、和SEQ ID NO:6的CDR1区;或a) The heavy chain variable domain comprises the CDR3 region of SEQ ID NO:1, the CDR2 region of SEQ ID NO:2, and the CDR1 region of SEQ ID NO:3, and the light chain variable domain comprises the CDR3 of SEQ ID NO:4 region, the CDR2 region of SEQ ID NO:5, and the CDR1 region of SEQ ID NO:6; or
b)a)所述抗体的CDR嫁接的、人源化的或T细胞表位消减的抗体变体。b) A CDR-grafted, humanized or T-cell epitope-depleted antibody variant of the antibody of a).
在一个实施方案中,所述抗体特征在于包含:In one embodiment, the antibody is characterized in comprising:
a)重链可变域的氨基酸序列是SEQ ID NO:7,且轻链可变域的氨基酸序列是SEQ ID NO:8;或a) the amino acid sequence of the heavy chain variable domain is SEQ ID NO: 7, and the amino acid sequence of the light chain variable domain is SEQ ID NO: 8; or
b)a)所述抗体的CDR嫁接的、人源化的或T细胞表位消减的抗体变体。b) A CDR-grafted, humanized or T-cell epitope-depleted antibody variant of the antibody of a).
在一个实施方案中,所述抗体结合人CSF-1R,并且特征在于上文所提及的氨基酸序列和氨基酸序列片段是人IgG1亚类的或者是人IgG4亚类的。In one embodiment, said antibody binds human CSF-1R and is characterized in that the above-mentioned amino acid sequences and amino acid sequence fragments are of human IgG1 subclass or of human IgG4 subclass.
本发明的又一个实施方案是包含依照本发明的抗体的药物组合物。Yet another embodiment of the invention is a pharmaceutical composition comprising an antibody according to the invention.
本发明进一步包括以包含结合人CSF-1R的抗体为特征的药物组合物,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文所提及的氨基酸序列和氨基酸序列片段。The invention further includes pharmaceutical compositions characterized by comprising an antibody binding to human CSF-1R characterized by the above-mentioned epitope binding properties or alternatively by the above-mentioned Amino acid sequences and amino acid sequence fragments.
本发明进一步包括以包含结合人CSF-1R的抗体为特征的抗体用于制造药物组合物的用途,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文所提及的氨基酸序列和氨基酸序列片段。The invention further comprises the use of an antibody characterized by comprising an antibody binding to human CSF-1R characterized by the above mentioned epitope binding properties or alternatively for the manufacture of a pharmaceutical composition Amino acid sequences and amino acid sequence fragments mentioned above.
本发明进一步包括以包含结合人CSF-1R的抗体为特征的抗体用于治疗CSF-1R介导的疾病的用途,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文所提及的氨基酸序列和氨基酸序列片段。The present invention further comprises the use of an antibody characterized by comprising an antibody binding to human CSF-1R characterized by the above-mentioned epitope binding for the treatment of a CSF-1R mediated disease Properties or alternatively the above mentioned amino acid sequences and amino acid sequence fragments.
本发明进一步包括以包含结合人CSF-1R的抗体为特征的抗体用于治疗癌症的用途,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文所提及的氨基酸序列和氨基酸序列片段。The invention further comprises the use of an antibody characterized by comprising an antibody binding to human CSF-1R characterized by the above-mentioned epitope binding properties or alternatively the above-mentioned Amino acid sequences and amino acid sequence fragments mentioned.
本发明进一步包括以包含结合人CSF-1R的抗体为特征的抗体用于治疗骨损失的用途,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文所提及的氨基酸序列和氨基酸序列片段。The invention further includes the use of an antibody characterized by comprising an antibody binding to human CSF-1R characterized by the above-mentioned epitope binding properties or alternatively by the above-mentioned Amino acid sequences and amino acid sequence fragments mentioned herein.
本发明进一步包括以包含结合人CSF-1R的抗体为特征的抗体用于预防或治疗转移的用途,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文所提及的氨基酸序列和氨基酸序列片段。The invention further includes the use of an antibody characterized by comprising an antibody binding to human CSF-1R characterized by the above mentioned epitope binding properties or alternatively for the prevention or treatment of metastasis Amino acid sequences and amino acid sequence fragments mentioned above.
本发明进一步包括以包含结合人CSF-1R的抗体为特征的抗体用于治疗炎性疾病的用途,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文所提及的氨基酸序列和氨基酸序列片段。The invention further includes the use of an antibody characterized by comprising an antibody binding to human CSF-1R characterized by the above-mentioned epitope binding properties or alternatively, for the treatment of an inflammatory disease Amino acid sequences and amino acid sequence fragments mentioned above.
本发明的一方面是结合人CSF-1R的抗体,其特征在于作为重链可变域CDR3区包含SEQ ID NO:1的CDR3区。One aspect of the invention is an antibody that binds human CSF-1R, characterized in comprising the CDR3 region of SEQ ID NO: 1 as the heavy chain variable domain CDR3 region.
本发明的另一方面是结合人CSF-1R的抗体,其特征在于Another aspect of the invention is an antibody that binds to human CSF-1R, characterized in that
a)重链可变域包含SEQ ID NO:1的CDR3区、SEQ ID NO:2的CDR2区、和SEQ ID NO:3的CDR1区,且轻链可变域包含SEQ ID NO:4的CDR3区、SEQID NO:5的CDR2区、和SEQ ID NO:6的CDR1区;或a) The heavy chain variable domain comprises the CDR3 region of SEQ ID NO:1, the CDR2 region of SEQ ID NO:2, and the CDR1 region of SEQ ID NO:3, and the light chain variable domain comprises the CDR3 of SEQ ID NO:4 region, the CDR2 region of SEQ ID NO:5, and the CDR1 region of SEQ ID NO:6; or
b)a)所述抗体的CDR嫁接的、人源化的或T细胞表位消减的抗体变体。b) A CDR-grafted, humanized or T-cell epitope-depleted antibody variant of the antibody of a).
在一个实施方案中,所述抗体特征在于包含In one embodiment, the antibody is characterized in comprising
a)重链可变域的氨基酸序列是SEQ ID NO:7,且轻链可变域的氨基酸序列是SEQ ID NO:8;或a) the amino acid sequence of the heavy chain variable domain is SEQ ID NO: 7, and the amino acid sequence of the light chain variable domain is SEQ ID NO: 8; or
b)a)所述抗体的CDR嫁接的、人源化的或T细胞表位消减的抗体变体。b) A CDR-grafted, humanized or T-cell epitope-depleted antibody variant of the antibody of a).
在本发明的一方面,依照本发明的抗体以至少10-8mol/l至10-12mol/l的亲和力结合人CSF-1R。In one aspect of the invention, an antibody according to the invention binds human CSF-1R with an affinity of at least 10 −8 mol/l to 10 −12 mol/l.
在本发明的一方面,依照本发明的抗体是人源化抗体。In one aspect of the invention, the antibody according to the invention is a humanized antibody.
本发明的又一个实施方案是编码依照本发明的抗体的重链可变域和/或轻链可变域的核酸。优选地,所述核酸编码结合人CSF-1R的抗体的重链,其特征在于作为重链CDR3区包含SEQ ID NO:1的CDR3区。Yet another embodiment of the invention is a nucleic acid encoding a heavy chain variable domain and/or a light chain variable domain of an antibody according to the invention. Preferably, said nucleic acid encodes a heavy chain of an antibody that binds to human CSF-1R, characterized in that it comprises the CDR3 region of SEQ ID NO: 1 as the heavy chain CDR3 region.
本发明的又一个实施方案是编码依照本发明的抗体的核酸,所述抗体特征在于:Yet another embodiment of the invention is a nucleic acid encoding an antibody according to the invention, said antibody being characterized in that:
a)重链可变域包含SEQ ID NO:1的CDR3区、SEQ ID NO:2的CDR2区、和SEQ ID NO:3的CDR1区,且轻链可变域包含SEQ ID NO:4的CDR3区、SEQID NO:5的CDR2区、和SEQ ID NO:6的CDR1区;或a) The heavy chain variable domain comprises the CDR3 region of SEQ ID NO:1, the CDR2 region of SEQ ID NO:2, and the CDR1 region of SEQ ID NO:3, and the light chain variable domain comprises the CDR3 of SEQ ID NO:4 region, the CDR2 region of SEQ ID NO:5, and the CDR1 region of SEQ ID NO:6; or
b)a)所述抗体的CDR嫁接的、人源化的或T细胞表位消减的抗体变体。b) A CDR-grafted, humanized or T-cell epitope-depleted antibody variant of the antibody of a).
本发明进一步提供了含有依照本发明的核酸、能够在原核或真核宿主细胞中表达所述核酸的表达载体,和含有此类载体以重组生成此类抗体的宿主细胞。The invention further provides expression vectors containing nucleic acids according to the invention, capable of expressing said nucleic acids in prokaryotic or eukaryotic host cells, and host cells containing such vectors for the recombinant production of such antibodies.
本发明进一步包括包含依照本发明的载体的原核或真核宿主细胞。The invention further includes prokaryotic or eukaryotic host cells comprising a vector according to the invention.
本发明进一步包括用于生成依照本发明的重组人源化抗体的方法,其特征在于在原核或真核宿主细胞中表达依照本发明的核酸,并自所述细胞或细胞培养物上清液回收所述抗体。本发明进一步包括通过此类重组方法可获得的抗体。The invention further comprises a method for producing a recombinant humanized antibody according to the invention, characterized in that the nucleic acid according to the invention is expressed in a prokaryotic or eukaryotic host cell and recovered from said cells or cell culture supernatant the antibody. The invention further includes antibodies obtainable by such recombinant methods.
依照本发明的抗体对需要CSF-1R靶向疗法的患者显示益处。依照本发明的抗体具有对患有肿瘤疾病,尤其是患有癌症的患者引起益处的、具有新颖的且创造性的特性。Antibodies according to the invention show benefit for patients in need of CSF-1R targeted therapy. The antibodies according to the invention have novel and inventive properties which are of benefit to patients suffering from neoplastic diseases, especially cancer.
本发明进一步提供了用于治疗患有癌症的患者的方法,包括对诊断为具有此类疾病(并且因此需要此类疗法)的患者施用有效量的依照本发明的结合人CSF-1R的抗体。优选地,在药物组合物中施用抗体。The invention further provides a method for treating a patient with cancer comprising administering to a patient diagnosed with such disease (and therefore in need of such therapy) an effective amount of an antibody that binds human CSF-1R according to the invention. Preferably, the antibody is administered in a pharmaceutical composition.
本发明的又一个实施方案是用于治疗患有癌症的患者的方法,其特征在于对患者施用依照本发明的抗体。Yet another embodiment of the invention is a method for the treatment of a patient suffering from cancer, characterized in that the antibody according to the invention is administered to the patient.
本发明进一步包括依照本发明的抗体用于治疗患有癌症的患者及用于制造依照本发明的药物组合物的用途。另外,本发明包括用于制造依照本发明的药物组合物的方法。The invention further comprises the use of an antibody according to the invention for the treatment of a patient suffering from cancer and for the manufacture of a pharmaceutical composition according to the invention. Additionally, the present invention includes methods for the manufacture of pharmaceutical compositions according to the present invention.
本发明进一步包括包含依照本发明的抗体,任选地以及出于药用目的可用于配制抗体的缓冲液和/或佐剂的药物组合物。The invention further comprises pharmaceutical compositions comprising an antibody according to the invention, optionally and buffers and/or adjuvants useful for formulating the antibody for pharmaceutical purposes.
本发明进一步提供了包含药学可接受载体中的依照本发明的抗体的药物组合物。在一个实施方案中,药物组合物可以包含在制品或试剂盒中。The invention further provides a pharmaceutical composition comprising an antibody according to the invention in a pharmaceutically acceptable carrier. In one embodiment, the pharmaceutical compositions may be included in an article of manufacture or a kit.
附图简述Brief description of the drawings
图1:在用10μg/ml浓度的不同抗CSF-1R单克隆抗体处理下对3D培养中BeWo肿瘤细胞的生长抑制。Figure 1 : Growth inhibition of BeWo tumor cells in 3D culture under treatment with different anti-CSF-1R monoclonal antibodies at a concentration of 10 μg/ml.
X轴:与细胞的ATP含量对应的存活力均值相对光单位(RLU)(CellTiterGlo测定法)。X-axis: mean relative light units (RLU) of viability corresponding to the ATP content of the cells (CellTiterGlo assay).
Y轴:测试探针:极限培养基(0.5%FBS)、小鼠IgG1(mIgG1,10μg/ml)、小鼠IgG2a(mIgG2a 10μg/ml)、仅CSF-1、<CSF-1R>7G5.3B6、和SC-02、克隆2-4A5。Y axis: Test probes: minimal medium (0.5%FBS), mouse IgG1 (mIgG1, 10μg/ml), mouse IgG2a (mIgG2a 10μg/ml), CSF-1 only, <CSF-1R>7G5.3B6 , and SC-02, clone 2-4A5.
用依照本发明的抗CSF-1R抗体观察到对CSF-1诱导的生长的最高抑制。The highest inhibition of CSF-1 induced growth was observed with anti-CSF-1R antibodies according to the invention.
发明详述Detailed description of the invention
I.定义I. Definition
术语“抗体”涵盖各种形式的抗体,包括但不限于全抗体、抗体片段、人源化抗体、嵌合抗体、T细胞表位消减的抗体、和别的遗传工程化抗体,只要依照本发明的特征性特性得到保留。The term "antibody" encompasses various forms of antibodies, including but not limited to whole antibodies, antibody fragments, humanized antibodies, chimeric antibodies, T-cell epitope-depleted antibodies, and other genetically engineered antibodies, as long as they are in accordance with the present invention characteristic properties are preserved.
“抗体片段”包含全长抗体的一部分,优选地,其可变域,或至少其抗原结合位点。抗体片段的例子包括双抗体、单链抗体分子、和自抗体片段形成的多特异性抗体。例如,scFv抗体记载于Huston,J.S.,Methods in Enzymol.203(1991)46-88。另外,抗体片段包含具有结合CSF-1R的VH域或结合CSF-1R的VL域的特征(即能够与VL域或VH域一起装配成功能性抗原结合位点,并且由此提供该特性)的单链多肽。An "antibody fragment" comprises a portion of a full-length antibody, preferably, its variable domain, or at least its antigen-binding site. Examples of antibody fragments include diabodies, single chain antibody molecules, and multispecific antibodies formed from antibody fragments. For example, scFv antibodies are described in Huston, JS, Methods in Enzymol. 203 (1991) 46-88. In addition, antibody fragments comprise the characteristic of having a CSF-1R-binding VH domain or a CSF-1R-binding VL domain (i.e., capable of assembling together with a VL or VH domain into a functional antigen-binding site, and thereby providing This characteristic) single-chain polypeptide.
如本文中所使用的,术语“单克隆抗体”或“单克隆抗体组合物”指单一氨基酸组成的抗体分子的制备物。As used herein, the term "monoclonal antibody" or "monoclonal antibody composition" refers to a preparation of antibody molecules composed of a single amino acid.
术语“嵌合抗体”指通常通过重组DNA技术制备的,包含来自小鼠的可变区,即结合区和自不同来源或物种衍生的恒定区的至少一部分的单克隆抗体。包含小鼠可变区和人恒定区的嵌合抗体是特别优选的。此类大鼠/人嵌合抗体是包含编码大鼠免疫球蛋白可变区的DNA区段和编码人免疫球蛋白恒定区的DNA区段的所表达免疫球蛋白基因的产物。本发明所涵盖的“嵌合抗体”的其它形式是那些其中类或亚类已经自初始抗体的类或亚类修饰或改变的。此类“嵌合”抗体又称为“类转换抗体”。用于生成嵌合抗体的方法牵涉本领域现在公知的常规重组DNA和基因转染技术。参见例如Morrison,S.L.等,Proc.Natl.Acad Sci.USA 81(1984)6851-6855;US 5,202,238和US5,204,244。The term "chimeric antibody" refers to a monoclonal antibody comprising at least a portion of a mouse-derived variable region, ie, a binding region, and a constant region derived from a different source or species, usually produced by recombinant DNA techniques. Chimeric antibodies comprising mouse variable regions and human constant regions are particularly preferred. Such rat/human chimeric antibodies are the product of expressed immunoglobulin genes comprising DNA segments encoding rat immunoglobulin variable regions and DNA segments encoding human immunoglobulin constant regions. Other forms of "chimeric antibodies" encompassed by the invention are those in which the class or subclass has been modified or changed from that of the original antibody. Such "chimeric" antibodies are also known as "class switched antibodies". Methods for generating chimeric antibodies involve conventional recombinant DNA and gene transfection techniques now well known in the art. See eg Morrison, S.L. et al., Proc. Natl. Acad Sci. USA 81 (1984) 6851-6855; US 5,202,238 and US 5,204,244.
如本申请内所使用的,术语“CDR嫁接的变体”意指通常通过重组DNA技术制备的包含来自一种来源或物种的互补决定区(CDR或高变区)和来自不同来源或物种的框架区(FR)的抗体可变域。包含鼠CDR和人FR的可变域的CDR嫁接变体是优选的。As used within this application, the term "CDR-grafted variant" means a variant comprising complementarity determining regions (CDRs or hypervariable regions) from one source or species and CDRs from a different source or species, usually prepared by recombinant DNA techniques. Antibody variable domains in the framework regions (FR). CDR-grafted variants comprising murine CDRs and variable domains of human FRs are preferred.
如本申请内所使用的,术语“T细胞表位消减的变体”意指通过除去人T细胞表位(可变域内具有结合MHC II类分子的能力的肽序列)修饰为消除或降低免疫原性的抗体可变域。通过此方法,鉴定可变域的氨基酸侧链与具有MHC II类结合沟的特定结合袋间的相互作用。将鉴定的免疫原性区突变以消除免疫原性。一般地,此类方法记载于例如WO 98/52976。As used within this application, the term "T-cell epitope-subtracted variant" means a variant modified to eliminate or reduce immunity by removing human T-cell epitopes (peptide sequences within variable domains that have the ability to bind MHC class II molecules). Antibody variable domains. By this method, the interactions between the amino acid side chains of the variable domains and specific binding pockets with the MHC class II binding groove are identified. The identified immunogenic regions were mutated to eliminate immunogenicity. Generally, such methods are described, for example, in WO 98/52976.
如本申请内所使用的,术语“人源化变体”意指自非人起源,例如非人物种的互补决定区(CDR),并且自人起源的框架区(FR)重建的,并且已经进一步修饰以还重建或改善初始非人可变域的结合亲和力和特异性的抗体可变域。此类人源化变体通常通过重组DNA技术来制备。亲本非人可变域的亲和力和特异性的重建是至关重要的步骤,对此目前使用不同方法。在一种方法中,测定在非人CDR中及在人FR中引入突变,即所谓的回复突变是否是有益的。可以例如通过序列或同源性分析,通过选择人框架(固定的框架方法;同源性匹配或最佳拟合),通过使用共有序列,通过自几种不同人单抗选择FR,或者通过用存在于人单抗中的最常见的残基替换三维表面上的非人残基(“表面重修”或“镶饰”)来鉴定此类回复突变的合适位置。As used within this application, the term "humanized variant" means a complementarity determining region (CDR) of non-human origin, e.g. a non-human species, and a framework region (FR) of human origin reconstituted, and has Antibody variable domains that are further modified to also re-establish or improve the binding affinity and specificity of the original non-human variable domains. Such humanized variants are usually prepared by recombinant DNA techniques. Reconstitution of the affinity and specificity of the parental non-human variable domains is a crucial step, for which different approaches are currently used. In one approach, it is determined whether introducing mutations, so called back mutations, in non-human CDRs as well as in human FRs is beneficial. It can be done, for example, by sequence or homology analysis, by selecting a human framework (fixed framework approach; homology matching or best fit), by using consensus sequences, by selecting FRs from several different human mAbs, or by using The most common residues present in human mAbs were substituted for non-human residues on the three-dimensional surface (“resurfacing” or “veneering”) to identify suitable locations for such backmutations.
另外,依照本发明的抗体包括具有“保守序列修饰”,即不影响或改变依照本发明的抗体的上文所提及的特征的核苷酸和氨基酸序列修饰的此类抗体。可以通过本领域中已知的标准技术,诸如定点诱变和PCR介导的诱变引入修饰。保守氨基酸替代包括用具有相似侧链的氨基酸残基替换氨基酸残基的。本领域中已经限定具有相似侧链的氨基酸残基的家族。这些家族包括具有碱性侧链(例如,赖氨酸、精氨酸、组氨酸)、酸性侧链(例如,天冬氨酸、谷氨酸)、不带电荷的极性侧链(例如,甘氨酸、天冬酰胺、谷氨酰胺、丝氨酸、苏氨酸、酪氨酸、半胱氨酸、色氨酸)、非极性侧链(例如,丙氨酸、缬氨酸、亮氨酸、异亮氨酸、脯氨酸、苯丙氨酸、甲硫氨酸)、β-分支的侧链(例如,苏氨酸、缬氨酸、异亮氨酸)和芳香族侧链(例如,酪氨酸、苯丙氨酸、色氨酸、组氨酸)的氨基酸。如此,优选地,可以将人抗CSF-1R抗体中预测的非必需氨基酸残基用来自同一侧链家族的另一种氨基酸残基替换。In addition, antibodies according to the present invention include such antibodies with "conservative sequence modifications", ie nucleotide and amino acid sequence modifications that do not affect or alter the above-mentioned characteristics of the antibodies according to the present invention. Modifications can be introduced by standard techniques known in the art, such as site-directed mutagenesis and PCR-mediated mutagenesis. Conservative amino acid substitutions include the replacement of amino acid residues with amino acid residues having similar side chains. Families of amino acid residues having similar side chains have been defined in the art. These families include those with basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., , glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine, tryptophan), nonpolar side chains (e.g., alanine, valine, leucine , isoleucine, proline, phenylalanine, methionine), β-branched side chains (e.g., threonine, valine, isoleucine) and aromatic side chains (e.g. , tyrosine, phenylalanine, tryptophan, histidine) amino acids. Thus, preferably, a predicted non-essential amino acid residue in a human anti-CSF-1R antibody may be replaced with another amino acid residue from the same side chain family.
可以基于分子建模通过诱变实施氨基酸替代,如Riechmann,L.等,Nature332(1988)323-327及Queen,C.等,Proc.Natl.Acad.Sci.USA 86(1989)10029-10033所描述的。Amino acid substitutions can be performed by mutagenesis based on molecular modeling as described by Riechmann, L. et al., Nature 332 (1988) 323-327 and Queen, C. et al., Proc. Natl. Acad. Sci. USA 86 (1989) 10029-10033 describe.
如本文中所使用的,术语“CSF-1R”指人CSF-1R (SEQ ID No:15)。CSF-1R(同义词:CSF-1受体;M-CSF受体;巨噬细胞集落刺激因子1受体、EC 2.7.10.1、Fms原癌基因、c-fms、Swiss Prot P07333、CD115)自1986以来是已知的(Coussens L.等,Nature 320(1986)277-280)。CSF-1R是一种生长因子,并且由c-fms原癌基因编码(综述见例如Roth,P.和Stanley,E.R.,Curr.Top.Microbiol.Immunol.181(1992)141-67)。As used herein, the term "CSF-1R" refers to human CSF-1R (SEQ ID No: 15). CSF-1R (synonyms: CSF-1 receptor; M-CSF receptor; macrophage colony-stimulating
CSF-1R是M-CSF(巨噬细胞集落刺激因子,又称作CSF-1)的受体,并且介导此细胞因子的生物学效应(Sherr,C.J.等,Cell 41(1985)665-676)。集落刺激因子-1受体(又称作c-fms)的克隆第一次记载于Roussel,M.F.等,Nature325(1987)549-552。在该出版物中,显示了CSF-1R具有转化潜力,其依赖于该蛋白质的C端尾部的变化,包括抑制性酪氨酸969磷酸化的丧失,其结合Cbl,并且由此调节受体下调(Lee,P.S.等,Embo J.18(1999)3616-3628)。CSF-1R is a receptor for M-CSF (macrophage colony-stimulating factor, also known as CSF-1), and mediates the biological effects of this cytokine (Sherr, C.J. et al., Cell 41(1985) 665-676 ). The cloning of the colony-stimulating factor-1 receptor (also known as c-fms) was first described by Roussel, M.F. et al., Nature 325 (1987) 549-552. In this publication, it was shown that CSF-1R has a transforming potential that is dependent on changes in the C-terminal tail of the protein, including loss of phosphorylation of the inhibitory tyrosine 969, which binds Cbl, and thereby regulates receptor downregulation (Lee, P.S. et al., Embo J. 18(1999) 3616-3628).
CSF-1R是一种单链、跨膜受体酪氨酸激酶(RTK)和以受体的胞外部分中的重复Ig域为特征的含有免疫球蛋白(Ig)基序的RTK家族的一名成员。胞内蛋白质酪氨酸激酶域以独特插入域中断,所述独特插入域也存在于包括血小板衍生的生长因子受体(PDGFR)、干细胞生长因子受体(c-Kit)和fins样细胞因子受体(FLT3)的其它相关RTK III类家族成员中。尽管此生长因子受体家族间有结构同源性,但是它们具有截然不同的组织特异性功能。CSF-1R主要在单核细胞谱系的细胞上及在女性生殖道和胎盘中存在。另外,已经在皮肤中的朗格汉斯细胞,即一种平滑肌细胞子集(Inaba,T.等,J.Biol.Chem.267(1992)5693-5699)、B细胞(Baker,A.H.等,Oncogene 8(1993)371-378)和小胶质(Sawada,M.等,Brain Res.509(1990)119-124)中报告了CSF-1R的表达。CSF-1R is a single-chain, transmembrane receptor tyrosine kinase (RTK) and member of the immunoglobulin (Ig) motif-containing RTK family characterized by repeated Ig domains in the extracellular portion of the receptor members. Intracellular protein tyrosine kinase domains are interrupted by unique insertion domains also present in receptors including platelet-derived growth factor receptor (PDGFR), stem cell growth factor receptor (c-Kit), and fins-like cytokine receptors. among other related RTK class III family members of the FLT3. Despite the structural homology between this family of growth factor receptors, they have distinct tissue-specific functions. CSF-1R is predominantly found on cells of the monocyte lineage and in the female reproductive tract and placenta. In addition, Langerhans cells, a subset of smooth muscle cells already in the skin (Inaba, T. et al., J. Biol. Chem. 267 (1992) 5693-5699), B cells (Baker, A.H. et al., Expression of CSF-1R has been reported in Oncogene 8 (1993) 371-378) and in microglia (Sawada, M. et al., Brain Res. 509 (1990) 119-124).
如本文中所使用的,术语“结合人CSF-1R”或“抗CSF-1R”可互换使用,并且指特异性结合人CSF-1R抗原的抗体。结合亲和力是于35℃的1.0x10-8mol/l或更低的KD值的,优选地,于35℃的1.0x10-9mol/l或更低的KD值的。用标准结合测定法,诸如表面等离振子共振技术(Biacore)测定结合亲和力(见实施例4)。As used herein, the terms "bind to human CSF-1R" or "anti-CSF-1R" are used interchangeably and refer to antibodies that specifically bind human CSF-1R antigens. The binding affinity is a KD value of 1.0x10 -8 mol/l or lower at 35°C, preferably a KD value of 1.0x10 -9 mol/l or lower at 35°C. Using standard binding assays such as surface plasmon resonance (Biacore ) to determine binding affinity (see Example 4).
术语“表位”意指能够特异性结合抗体的蛋白质决定簇。表位通常由分子的化学活性表面聚集诸如氨基酸或糖侧链组成,并且通常表位具有特定的三维结构特征及特定的电荷特征。构象性和非构象性表位的区别之处在于在存在变性溶剂的情况中丧失对前一种,而不是对后一种的结合。优选地,依照本发明的抗体特异性结合天然的,而不是变性的CSF-1R。The term "epitope" means a determinant of a protein capable of specifically binding an antibody. Epitopes usually consist of chemically active surface aggregations of molecules such as amino acids or sugar side chains, and usually epitopes have specific three-dimensional structural characteristics as well as specific charge characteristics. Conformational and non-conformational epitopes are distinguished by the loss of binding to the former, but not to the latter, in the presence of denaturing solvents. Preferably, the antibodies according to the invention specifically bind native, not denatured CSF-1R.
如本文中所使用的,术语“与保藏抗体DSM ACC2921结合相同表位”指结合CSF-1R上抗体<CSF-1R>7G5.3B6(保藏号DSM ACC2921)结合的相同表位的本发明的抗CSF-1R抗体。可以使用本领域中已知的技术来测定本发明的抗CSF-1R抗体的表位结合特性。于25℃在体外竞争性结合抑制测定法中通过表面等离振子共振(SPR)测定测试抗体抑制抗体<CSF-1R>7G5.3B6(保藏号DSM ACC2921)结合CSF-1R的能力来测量CSF-1R抗体。这可以通过BIAcore测定法(Pharmacia Biosensor AB,Uppsala,Sweden)调查,如例如在实施例5中。在实施例5中,与结合的抗体<CSF-1R>7G5.3B6(保藏号DSMACC2921)竞争的本发明的CSF-1R抗体的预期结合响应的百分比(%)通过“100*相对响应(大体_稳定性_早期)/rMax”计算,其中rMax通过“相对响应(大体_稳定性_晚期)*抗体分子量/抗原分子量”计算,如记载于Biacore测定法表位定位仪的。还自相同抗体1和2的对计算最小限度结合响应(见实施例5)。其获得的最大值+100%,优选地50%设置为显著竞争并且如此显著结合相同表位的阈值(见实施例5,对于抗体<CSF-1R>7G5.3B6,计算的阈值是3+3=6,优选地3+1.5=4.5)。如此,以“与<CSF-1R>7G5.3B6(保藏号DSM ACC2921)结合相同表位”为特征的结合人CSF-1R的抗体具有低于6,优选地4.5的预期结合响应的百分比(%)(小于6,优选地小于4.5的%预期结合响应)。As used herein, the term "binding to the same epitope as the deposited antibody DSM ACC2921" refers to the antibody of the present invention that binds to the same epitope as the antibody <CSF-1R>7G5.3B6 (deposit number DSM ACC2921) on CSF-1R. Antibody to CSF-1R. The epitope binding properties of the anti-CSF-1R antibodies of the invention can be determined using techniques known in the art. CSF- 1R antibody. This can be investigated by BIAcore assay (Pharmacia Biosensor AB, Uppsala, Sweden), as for example in Example 5. In Example 5, the percentage (%) of the expected binding response of the CSF-1R antibody of the invention competing with the bound antibody <CSF-1R>7G5.3B6 (Accession No. DSMACC2921) was calculated by "100*relative response (approximately_ Stability_early)/rMax", where rMax is calculated by "Relative Response (Mass_Stability_Late)*Antibody Molecular Weight/Antigen Molecular Weight" as described in the Biacore assay epitope localizer. Minimal binding responses were also calculated from pairs of the
在一方面,依照本发明的抗体与保藏抗体DSM ACC2921竞争对人CSF-1R的结合。可以使用本领域中已知的技术测定此类结合竞争。于25℃在体外竞争性结合抑制测定法中通过表面等离振子共振(SPR)测定测试抗体抑制抗体<CSF-1R>7G5.3B6(保藏号DSM ACC2921)结合人CSF-1R的能力来测量CSF-1R抗体。这可以通过BIAcore测定法(Pharmacia Biosensor AB,Uppsala,Sweden)调查,如例如在实施例5中。In one aspect, the antibody according to the invention competes with the deposited antibody DSM ACC2921 for binding to human CSF-1R. Such binding competition can be determined using techniques known in the art. CSF was measured by testing the ability of the antibody to inhibit the binding of antibody <CSF-1R>7G5.3B6 (Accession No. DSM ACC2921) to human CSF-1R by surface plasmon resonance (SPR) assay in an in vitro competitive binding inhibition assay at 25°C -1R antibody. This can be investigated by BIAcore assay (Pharmacia Biosensor AB, Uppsala, Sweden), as for example in Example 5.
如本文中所使用的,“可变域”(轻链可变域(VL)、重链可变域(VH))意为直接牵涉抗体结合抗原的轻和重链域对之每种。可变轻和重链域具有相同的一般结构,并且每个域包含通过三个“高变区”(或互补决定区,CDR)连接的其序列广泛保守的四个框架(FR)区。框架区采用β-片层构象,而CDR可以形成连接β-片层结构的环。每条链中的CDR通过框架区保持其三维结构,并且与来自另一条链的CDR一起形成抗原结合位点。抗体的重和轻链CDR3区在依照本发明的抗体的结合特异性/亲和力中发挥特别重要的作用,并且因此提供本发明的又一个目的。As used herein, "variable domain" (light chain variable domain (VL), heavy chain variable domain (VH)) means each of the pair of light and heavy chain domains that are directly involved in antibody binding to antigen. The variable light and heavy chain domains have the same general structure, and each domain comprises four framework (FR) regions whose sequences are widely conserved, connected by three "hypervariable regions" (or complementarity determining regions, CDRs). The framework regions adopt a β-sheet conformation, while the CDRs can form loops connecting the β-sheet structures. The CDRs in each chain maintain their three-dimensional structure through the framework regions and, together with the CDRs from the other chain, form an antigen-binding site. The heavy and light chain CDR3 regions of antibodies play a particularly important role in the binding specificity/affinity of antibodies according to the invention and thus provide a further object of the invention.
术语“抗体的抗原结合部分”在本文中使用时指抗体中负责抗原结合的氨基酸残基。抗体的抗原结合部分包含来自“互补决定区”或“CDR”的氨基酸残基。“框架”或“FR”区是除了如本文中所限定的高变区残基外的那些可变域区。因此,抗体的轻和重链可变域从N至C端包含域FR1、CDR1、FR2、CDR2、FR3、CDR3、和FR4。特别地,重链的CDR3是对抗原结合贡献最大并且限定抗体特性的区域。CDR和FR区依照Kabat,E.A.等,Sequencesof Proteins of Immunological Interest,第5版,Public Health Service,NationalInstitutes of Health,Bethesda,MD(1991)的标准定义和/或来自“高变环”的那些残基确定。The term "antigen-binding portion of an antibody" as used herein refers to the amino acid residues of an antibody that are responsible for antigen-binding. The antigen-binding portion of an antibody comprises amino acid residues from "complementarity determining regions" or "CDRs." "Framework" or "FR" regions are those variable domain regions other than the hypervariable region residues as defined herein. Thus, the light and heavy chain variable domains of an antibody comprise, from N to C-terminus, the domains FR1, CDR1, FR2, CDR2, FR3, CDR3, and FR4. In particular, CDR3 of the heavy chain is the region that contributes most to antigen binding and defines antibody properties. CDR and FR regions are defined according to the standard of Kabat, E.A. et al., Sequences of Proteins of Immunological Interest, 5th edition, Public Health Service, National Institutes of Health, Bethesda, MD (1991) and/or those residues from "hypervariable loops" Sure.
如本文中所使用的,术语“核酸”或“核酸分子”意图包括DNA分子和RNA分子。核酸分子可以是单链或双链,但是优选的是双链DNA。As used herein, the term "nucleic acid" or "nucleic acid molecule" is intended to include DNA molecules and RNA molecules. Nucleic acid molecules can be single-stranded or double-stranded, but are preferably double-stranded DNA.
如本申请内所使用的,术语“氨基酸”意指天然存在的羧基α-氨基酸的组,包括丙氨酸(三字母代码:ala,单字母代码:A)、精氨酸(arg,R)、天冬酰胺(asn,N)、天冬氨酸(asp,D)、半胱氨酸(cys,C)、谷氨酰胺(gln,Q)、谷氨酸(glu,E)、甘氨酸(gly,G)、组氨酸(his,H)、异亮氨酸(ile,I)、亮氨酸(leu,L)、赖氨酸(lys,K)、甲硫氨酸(met,M)、苯丙氨酸(phe,F)、脯氨酸(pro,P)、丝氨酸(ser,S)、苏氨酸(thr,T)、色氨酸(trp,W)、酪氨酸(tyr,Y)、和缬氨酸(val,V)。As used within this application, the term "amino acid" means the group of naturally occurring carboxy alpha-amino acids, including alanine (three-letter code: ala, one-letter code: A), arginine (arg, R) , asparagine (asn, N), aspartic acid (asp, D), cysteine (cys, C), glutamine (gln, Q), glutamic acid (glu, E), glycine ( gly, G), histidine (his, H), isoleucine (ile, I), leucine (leu, L), lysine (lys, K), methionine (met, M ), phenylalanine (phe, F), proline (pro, P), serine (ser, S), threonine (thr, T), tryptophan (trp, W), tyrosine ( tyr, Y), and valine (val, V).
“免疫缀合物”指与一种或多种异源分子,包括但不限于细胞毒剂缀合的抗体。"Immunoconjugate" refers to an antibody conjugated to one or more heterologous molecules, including but not limited to cytotoxic agents.
“个体”或“受试者”是哺乳动物。哺乳动物包括但不限于驯养的动物(例如,牛、绵羊、猫、犬、和马)、灵长类(例如,人和非人灵长类诸如猴)、家兔、和啮齿类(例如,小鼠和大鼠)。在某些实施方案中,个体或受试者是人。An "individual" or "subject" is a mammal. Mammals include, but are not limited to, domesticated animals (e.g., cattle, sheep, cats, dogs, and horses), primates (e.g., humans and non-human primates such as monkeys), rabbits, and rodents (e.g., mice and rats). In certain embodiments, the individual or subject is a human.
“分离的”抗体指已经与其天然环境的组分分开的抗体。在一些实施方案中,抗体纯化至大于95%或99%的纯度,如通过例如电泳(例如,SDS-PAGE、等电聚焦(IEF)、毛细管电泳)或层析(例如,离子交换或反相HPLC)测定的。关于用于评估抗体纯度的方法的综述,见例如Flatman等,J.Chromatogr.B 848:79-87(2007)。An "isolated" antibody refers to an antibody that has been separated from components of its natural environment. In some embodiments, antibodies are purified to greater than 95% or 99% purity, such as by, for example, electrophoresis (e.g., SDS-PAGE, isoelectric focusing (IEF), capillary electrophoresis) or chromatography (e.g., ion exchange or reverse phase determined by HPLC). For a review of methods for assessing antibody purity, see, eg, Flatman et al., J. Chromatogr. B 848:79-87 (2007).
“分离的”核酸指已经与其天然环境的组分分开的核酸分子。分离的核酸包括通常含有核酸分子的细胞中含有的核酸分子,但是核酸分子在染色体外或在与其天然染色体位置不同的染色体位置处存在。An "isolated" nucleic acid refers to a nucleic acid molecule that has been separated from components of its natural environment. An isolated nucleic acid includes a nucleic acid molecule contained in cells that normally contain the nucleic acid molecule, but the nucleic acid molecule is present extrachromosomally or at a chromosomal location different from its natural chromosomal location.
“编码抗CSF-1R抗体的分离的核酸”指编码抗体重和轻链(或其片段)的一种或多种核酸分子,包括单一载体或不同载体中的此类核酸分子,和存在于宿主细胞中一个或多个位置的此类核酸分子。"Isolated nucleic acid encoding an anti-CSF-1R antibody" refers to one or more nucleic acid molecules encoding the antibody heavy and light chains (or fragments thereof), including such nucleic acid molecules in a single vector or in different vectors, and present in a host Such nucleic acid molecules at one or more locations in a cell.
“天然抗体”指具有不同结构的天然存在的免疫球蛋白分子。例如,天然IgG抗体是约150,000道尔顿的异四聚糖蛋白,由二硫化物键合的两条相同轻链和两条相同重链构成。从N至C端,每条重链具有一个可变区(VH),又称作可变重域或重链可变域,接着是三个恒定域(CH1、CH2、和CH3)。类似地,从N至C端,每条轻链具有一个可变区(VL),又称作可变轻域或轻链可变域,接着是一个恒定轻(CL)域。根据其恒定域氨基酸序列,抗体轻链可归入两种型中的一种,称作卡帕(κ)和拉姆达(λ)。"Native antibody" refers to naturally occurring immunoglobulin molecules of varying structure. For example, native IgG antibodies are heterotetrameric glycoproteins of approximately 150,000 Daltons, composed of two identical light chains and two identical heavy chains disulfide-bonded. From N to C-terminus, each heavy chain has a variable region (VH), also called variable heavy domain or heavy chain variable domain, followed by three constant domains (CH1, CH2, and CH3). Similarly, from N to C-terminus, each light chain has a variable region (VL), also called variable light domain or light chain variable domain, followed by a constant light (CL) domain. Based on the amino acid sequence of their constant domains, antibody light chains can be assigned to one of two types, called kappa (κ) and lambda (λ).
术语“包装插页”用于指治疗性产品的商业包装中通常包含的用法说明书,其含有关于涉及此类治疗性产品应用的适应症、用法、剂量、施用、联合疗法、禁忌症和/或警告的信息。The term "package insert" is used to refer to instructions commonly included in commercial packages of therapeutic products that contain information on the indications, usage, dosage, administration, combination therapies, contraindications and/or warnings concerning the use of such therapeutic products Information.
关于参照多肽序列的“百分比(%)氨基酸序列同一性”定义为比对序列并在必要时引入缺口以获取最大百分比序列同一性后,且不将任何保守替代视为序列同一性的一部分时,候选序列中与参照多肽序列中的氨基酸残基相同的氨基酸残基的百分率。为测定百分比氨基酸序列同一性目的的对比可以以本领域技术范围内的多种方式进行,例如使用公众可得到的计算机软件,诸如BLAST、BLAST-2、ALIGN或Megalign(DNASTAR)软件。本领域技术人员可以决定用于比对序列的合适参数,包括对所比较序列全长获得最大对比所需的任何算法。然而,为了本发明的目的,%氨基酸序列同一性值是使用序列比较计算机程序ALIGN-2产生的。ALIGN-2序列比较计算机程序由Genentech,Inc.编写,并且源代码已经连同用户文档一起提交给美国版权局(US Copyright Office,Washington D.C.,20559),其中其以美国版权注册号TXU510087注册。公众自Genentech,Inc.,South San Francisco,California可获得ALIGN-2程序,或者可以从源代码编译。ALIGN2程序应当编译成在UNIX操作系统,包括数码UNIX V4.0D上使用。所有序列比较参数由ALIGN-2程序设定且不变。"Percent (%) amino acid sequence identity" with respect to a reference polypeptide sequence is defined as after aligning the sequences and introducing gaps, if necessary, to obtain the maximum percent sequence identity, and when any conservative substitutions are not considered part of the sequence identity, The percentage of amino acid residues in a candidate sequence that are identical to those in a reference polypeptide sequence. Alignment for purposes of determining percent amino acid sequence identity can be performed in various ways that are within the skill in the art, for example, using publicly available computer software such as BLAST, BLAST-2, ALIGN or Megalign (DNASTAR) software. Those skilled in the art can determine appropriate parameters for aligning sequences, including any algorithms needed to achieve maximal alignment over the full length of the sequences being compared. However, for the purposes of the present invention, % amino acid sequence identity values are generated using the sequence comparison computer program ALIGN-2. The ALIGN-2 sequence comparison computer program was written by Genentech, Inc., and the source code, along with user documentation, has been filed with the US Copyright Office, Washington D.C., 20559, where it is registered under US Copyright Registration No. TXU510087. The ALIGN-2 program is publicly available from Genentech, Inc., South San Francisco, California, or can be compiled from source. The ALIGN2 program should be compiled for use on UNIX operating systems, including Digital UNIX V4.0D. All sequence comparison parameters are set by the ALIGN-2 program and do not change.
在采用ALIGN-2来比较氨基酸序列的情况中,给定氨基酸序列A相对于(to)、与(with)、或针对(against)给定氨基酸序列B的%氨基酸序列同一性(或者可表述为具有或包含相对于、与、或针对给定氨基酸序列B的某一%氨基酸序列同一性的给定氨基酸序列A)如下计算:In the case of comparing amino acid sequences using ALIGN-2, the % amino acid sequence identity of a given amino acid sequence A relative to (to), with (with), or against (against) a given amino acid sequence B (or can be expressed as A given amino acid sequence A) having or comprising a certain % amino acid sequence identity to, with, or for a given amino acid sequence B) is calculated as follows:
分数X/Y乘100Fraction X/Y times 100
其中X是由序列比对程序ALIGN-2在该程序的A和B比对中评分为相同匹配的氨基酸残基数,且其中Y是B中的氨基酸残基总数。应当领会,若氨基酸序列A的长度与氨基酸序列B的长度不相等,则A相对于B的%氨基酸序列同一性将不等于B相对于A的%氨基酸序列同一性。除非另有明确说明,本文中所使用的所有%氨基酸序列同一性值都是依照上一段所述,使用ALIGN-2计算机程序获得的。where X is the number of amino acid residues scored as identical matches in the alignment of A and B by the sequence alignment program ALIGN-2, and where Y is the total number of amino acid residues in B. It will be appreciated that if the length of amino acid sequence A is not equal to the length of amino acid sequence B, the % amino acid sequence identity of A with respect to B will not be equal to the % amino acid sequence identity of B with respect to A. Unless expressly stated otherwise, all % amino acid sequence identity values used herein were obtained using the ALIGN-2 computer program as described in the preceding paragraph.
II.组合物和方法II. Compositions and Methods
在一方面,本发明部分基于与保藏抗体DSM ACC2921针对相同表位。本发明的抗体可用于例如诊断或治疗癌症、炎性疾病或骨损失;或者用于预防或治疗转移。In one aspect, the invention is based in part on the same epitope as the deposited antibody DSM ACC2921. Antibodies of the invention are useful, for example, in the diagnosis or treatment of cancer, inflammatory diseases, or bone loss; or in the prevention or treatment of metastasis.
例示性的抗CSF-1R抗体Exemplary anti-CSF-1R antibodies
在一方面,本发明提供了结合人CSF-1R的抗体。在某些实施方案中,抗CSF-1R抗体特征在于与保藏抗体DSMACC2921结合相同表位。In one aspect, the invention provides antibodies that bind human CSF-1R. In certain embodiments, the anti-CSF-1R antibody is characterized in that it binds to the same epitope as the deposited antibody DSMACC2921.
本发明的另一方面是结合人CSF-1R的抗体,其特征在于:Another aspect of the invention is an antibody that binds to human CSF-1R, characterized in that:
a)重链可变域包含SEQ ID NO:1的CDR3区、SEQ ID NO:2的CDR2区、和SEQ ID NO:3的CDR1区,且轻链可变域包含SEQ ID NO:4的CDR3区、SEQID NO:5的CDR2区、和SEQ ID NO:6的CDR1区;或a) The heavy chain variable domain comprises the CDR3 region of SEQ ID NO:1, the CDR2 region of SEQ ID NO:2, and the CDR1 region of SEQ ID NO:3, and the light chain variable domain comprises the CDR3 of SEQ ID NO:4 region, the CDR2 region of SEQ ID NO:5, and the CDR1 region of SEQ ID NO:6; or
b)a)所述抗体的CDR嫁接的、人源化的或T细胞表位消减的抗体变体。b) A CDR-grafted, humanized or T-cell epitope-depleted antibody variant of the antibody of a).
本发明的另一方面是结合人CSF-1R的抗体,其特征在于:Another aspect of the invention is an antibody that binds to human CSF-1R, characterized in that:
a)重链可变域包含SEQ ID NO:1的CDR3区、SEQ ID NO:2的CDR2区、和SEQ ID NO:3的CDR1区,且轻链可变域包含SEQ ID NO:4的CDR3区、SEQID NO:5的CDR2区、和SEQ ID NO:6的CDR1区;或a) The heavy chain variable domain comprises the CDR3 region of SEQ ID NO:1, the CDR2 region of SEQ ID NO:2, and the CDR1 region of SEQ ID NO:3, and the light chain variable domain comprises the CDR3 of SEQ ID NO:4 region, the CDR2 region of SEQ ID NO:5, and the CDR1 region of SEQ ID NO:6; or
b)a)所述抗体的CDR嫁接的、人源化的或T细胞表位消减的抗体变体,且b) a CDR-grafted, humanized or T-cell epitope-depleted antibody variant of said antibody, and
具有下列一项或多项特性(如记载于实施例2、3、4、6、7和8中的测定法中测定的):Has one or more of the following properties (as determined in the assays described in Examples 2, 3, 4, 6, 7 and 8):
-抗CSF-1R抗体以25ng/ml或更低的IC50,在一个实施方案中,以20ng/ml或更低的IC50抑制CSF-1结合CSF-1R;-An anti-CSF-1R antibody inhibits CSF-1 binding to CSF-1R with an IC50 of 25 ng/ml or lower, in one embodiment, with an IC50 of 20 ng/ml or lower;
-抗CSF-1R抗体以100ng/ml或更低的IC50,在一个实施方案中,以50ng/ml或更低的IC50抑制CSF-1诱导的CSF-1R磷酸化(在NIH3T3-CSF-1R重组细胞中);-An anti-CSF-1R antibody inhibits CSF-1-induced CSF-1R phosphorylation with an IC50 of 100 ng/ml or less, in one embodiment, with an IC50 of 50 ng/ml or less (in recombinant cells);
-抗CSF-1R抗体将表达人CSF-1R(SEQ ID No:15)的重组NIH3T3细胞的生长抑制80%或更多(与没有抗体相比),优选地90%或更多;-An anti-CSF-1R antibody inhibits the growth of recombinant NIH3T3 cells expressing human CSF-1R (SEQ ID No: 15) by 80% or more (compared to no antibody), preferably 90% or more;
-抗CSF-1R抗体将BeWo肿瘤细胞(ATCC CCL-98)的生长抑制80%或更多(在10μg/ml的抗体浓度;且与没有抗体相比),优选地90%或更多;- the anti-CSF-1R antibody inhibits the growth of BeWo tumor cells (ATCC CCL-98) by 80% or more (at an antibody concentration of 10 μg/ml; and compared with no antibody), preferably 90% or more;
-抗CSF-1R抗体抑制巨噬细胞分化(在一个实施方案中,抗CSF-1R抗体以1.5nM或更低的IC50,优选地以1.0nM或更低的IC50抑制单核细胞存活);或- the anti-CSF-1R antibody inhibits macrophage differentiation (in one embodiment, the anti-CSF-1R antibody inhibits monocyte survival with an IC50 of 1.5 nM or less, preferably with an IC50 of 1.0 nM or less); or
-抗CSF-1R抗体于35℃以KD=1.0x10-9mol/l或更低的结合亲和力结合人CSF-1R。- The anti-CSF-1R antibody binds to human CSF-1R at 35°C with a binding affinity of KD=1.0×10 −9 mol/l or lower.
在另一方面,依照本发明的抗CSF-1R抗体在重链可变域(VH)序列中包含a)具有与SEQ ID NO:3相同的氨基酸序列,或者相对于SEQ ID NO:3包含1、2、或3处氨基酸残基替代的CDR1H,b)具有与SEQ ID NO:2相同的氨基酸序列,或者相对于SEQ ID NO:2包含1、2、或3处氨基酸残基替代的CDR2H和c)具有与SEQ ID NO:1相同的氨基酸序列,或者相对于SEQ ID NO:1包含1、2、或3处氨基酸残基替代的CDR3H。In another aspect, the anti-CSF-1R antibody according to the present invention comprises in the heavy chain variable domain (VH) sequence a) an amino acid sequence identical to SEQ ID NO: 3, or comprises 1 relative to SEQ ID NO: 3 , CDR1H with 2, or 3 amino acid residue substitutions, b) having the same amino acid sequence as SEQ ID NO: 2, or CDR2H comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO: 2 and c) have the same amino acid sequence as SEQ ID NO: 1, or CDR3H comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO: 1.
在某些实施方案中,包含a)具有与SEQ ID NO:3相同的氨基酸序列,或者相对于SEQ ID NO:3包含1、2、或3处氨基酸残基替代的CDR1H,b)具有与SEQ ID NO:2相同的氨基酸序列,或者相对于SEQ ID NO:2包含1、2、或3处氨基酸残基替代的CDR2H,和c)具有与SEQ ID NO:1相同的氨基酸序列,或者相对于SEQ ID NO:1包含1、2、或3处氨基酸残基替代的CDR3H的重链可变域(VH)序列相对于参照序列含有替代(例如,保守替代)、插入、或删除,但是包含所述序列的抗CSF-1R抗体保留结合CSF-1R的能力。In certain embodiments, comprising a) having the same amino acid sequence as SEQ ID NO:3, or CDR1H comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO:3, b) having the same amino acid sequence as SEQ ID NO:3 ID NO:2 the same amino acid sequence, or CDR2H comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO:2, and c) having the same amino acid sequence as SEQ ID NO:1, or relative to The heavy chain variable domain (VH) sequence of SEQ ID NO: 1 comprising 1, 2, or 3 amino acid residue substitutions of CDR3H contains substitutions (e.g., conservative substitutions), insertions, or deletions relative to the reference sequence, but includes all Anti-CSF-1R antibodies of the above sequence retain the ability to bind CSF-1R.
在另一方面,依照本发明的抗CSF-1R抗体在轻链可变域(VL)序列中包含a)具有与SEQ ID NO:6相同的氨基酸序列,或者相对于SEQ ID NO:6包含1、2、或3处氨基酸残基替代的CDR1L,b)具有与SEQ ID NO:5相同的氨基酸序列,或者相对于SEQ ID NO:5包含1、2、或3处氨基酸残基替代的CDR2L,和c)具有与SEQ ID NO:4相同的氨基酸序列,或者相对于SEQ ID NO:4包含1、2、或3处氨基酸残基替代的CDR3L。In another aspect, the anti-CSF-1R antibody according to the present invention comprises in the light chain variable domain (VL) sequence a) an amino acid sequence identical to SEQ ID NO: 6, or comprises 1 relative to SEQ ID NO: 6 , CDR1L with 2, or 3 amino acid residue substitutions, b) having the same amino acid sequence as SEQ ID NO: 5, or CDR2L comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO: 5, and c) a CDR3L having the same amino acid sequence as SEQ ID NO:4, or comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO:4.
在某些实施方案中,包含a)具有与SEQ ID NO:6相同的氨基酸序列,或者相对于SEQ ID NO:6包含1、2、或3处氨基酸残基替代的CDR1L,b)具有与SEQ ID NO:5相同的氨基酸序列,或者相对于SEQ ID NO:5包含1、2、或3处氨基酸残基替代的CDR2L,和c)具有与SEQ ID NO:4相同的氨基酸序列,或者相对于SEQ ID NO:4包含1、2、或3处氨基酸残基替代的CDR3L的轻链可变域(VL)序列相对于参照序列含有替代(例如,保守替代)、插入、或删除,但是包含所述序列的抗CSF-1R抗体保留结合CSF-1R的能力。In certain embodiments, comprising a) a CDR1L having the same amino acid sequence as SEQ ID NO:6, or comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO:6, b) having the same amino acid sequence as SEQ ID NO:6 ID NO:5 the same amino acid sequence, or CDR2L comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO:5, and c) having the same amino acid sequence as SEQ ID NO:4, or relative to The light chain variable domain (VL) sequence of the CDR3L of SEQ ID NO:4 comprising 1, 2, or 3 amino acid residue substitutions contains substitutions (for example, conservative substitutions), insertions, or deletions relative to the reference sequence, but includes all Anti-CSF-1R antibodies of the above sequence retain the ability to bind CSF-1R.
在另一方面,依照本发明的抗CSF-1R抗体In another aspect, the anti-CSF-1R antibody according to the present invention
-在重链可变域(VH)序列中包含a)具有与SEQ ID NO:3相同的氨基酸序列,或者相对于SEQ ID NO:3包含1、2、或3处氨基酸残基替代的CDR1H,b)具有与SEQ ID NO:2相同的氨基酸序列,或者相对于SEQ ID NO:2包含1、2、或3处氨基酸残基替代的CDR2H,和c)具有与SEQ ID NO:1相同的氨基酸序列,或者相对于SEQ ID NO:1包含1、2、或3处氨基酸残基替代的CDR3H,且在轻链可变域(VL)序列中包含d)具有与SEQ ID NO:6相同的氨基酸序列,或者相对于SEQ ID NO:6包含1、2、或3处氨基酸残基替代的CDR1L,e)具有与SEQ ID NO:5相同的氨基酸序列,或者相对于SEQ ID NO:5包含1、2、或3处氨基酸残基替代的CDR2L,和f)具有与SEQ ID NO:4相同的氨基酸序列,或者相对于SEQ ID NO:4包含1、2、或3处氨基酸残基替代的CDR3L。- comprising in the heavy chain variable domain (VH) sequence a) a CDR1H having the same amino acid sequence as SEQ ID NO: 3, or comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO: 3, b) has the same amino acid sequence as SEQ ID NO:2, or a CDR2H comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO:2, and c) has the same amino acid sequence as SEQ ID NO:1 sequence, or CDR3H comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO: 1, and comprising d) the same amino acid as SEQ ID NO: 6 in the light chain variable domain (VL) sequence sequence, or CDR1L comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO:6, e) having the same amino acid sequence as SEQ ID NO:5, or comprising 1, 2, or 3 amino acid residues relative to SEQ ID NO:5 2, or CDR2L with 3 amino acid residue substitutions, and f) having the same amino acid sequence as SEQ ID NO: 4, or CDR3L comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO: 4.
在另一方面,依照本发明的抗CSF-1R抗体In another aspect, the anti-CSF-1R antibody according to the present invention
-在重链可变域(VH)序列中包含a)具有与SEQ ID NO:3相同的氨基酸序列,或者相对于SEQ ID NO:3包含1、2、或3处氨基酸残基替代的CDR1H,b)具有与SEQ ID NO:2相同的氨基酸序列,或者相对于SEQ ID NO:2包含1、2、或3处氨基酸残基替代的CDR2H,和c)具有与SEQ ID NO:1相同的氨基酸序列,或者相对于SEQ ID NO:1包含1、2、或3处氨基酸残基替代的CDR3H,且在轻链可变域(VL)序列中包含d)具有与SEQ ID NO:6相同的氨基酸序列,或者相对于SEQ ID NO:6包含1、2、或3处氨基酸残基替代的CDR1L,e)具有与SEQ ID NO:5相同的氨基酸序列,或者相对于SEQ ID NO:5包含1、2、或3处氨基酸残基替代的CDR2L,和f)具有与SEQ ID NO:4相同的氨基酸序列,或者相对于SEQ ID NO:4包含1、2、或3处氨基酸残基替代的CDR3L;且- comprising in the heavy chain variable domain (VH) sequence a) a CDR1H having the same amino acid sequence as SEQ ID NO: 3, or comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO: 3, b) has the same amino acid sequence as SEQ ID NO:2, or a CDR2H comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO:2, and c) has the same amino acid sequence as SEQ ID NO:1 sequence, or CDR3H comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO: 1, and comprising d) the same amino acid as SEQ ID NO: 6 in the light chain variable domain (VL) sequence sequence, or CDR1L comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO:6, e) having the same amino acid sequence as SEQ ID NO:5, or comprising 1, 2, or 3 amino acid residues relative to SEQ ID NO:5 2, or CDR2L with 3 amino acid residue substitutions, and f) a CDR3L having the same amino acid sequence as SEQ ID NO: 4, or comprising 1, 2, or 3 amino acid residue substitutions relative to SEQ ID NO: 4; and
抗CSF-1R抗体具有下列一项或多项特性(如记载于实施例2、3、4、6、7和8中的测定法中测定的):Anti-CSF-1R antibodies have one or more of the following properties (as determined in the assays described in Examples 2, 3, 4, 6, 7 and 8):
-抗CSF-1R抗体以25ng/ml或更低的IC50,在一个实施方案中,以20ng/ml或更低的IC50抑制CSF-1结合CSF-1R;-An anti-CSF-1R antibody inhibits CSF-1 binding to CSF-1R with an IC50 of 25 ng/ml or lower, in one embodiment, with an IC50 of 20 ng/ml or lower;
-抗CSF-1R抗体以100ng/ml或更低的IC50,在一个实施方案中,以50ng/ml或更低的IC50抑制CSF-1诱导的CSF-1R磷酸化(在NIH3T3-CSF-1R重组细胞中);-An anti-CSF-1R antibody inhibits CSF-1-induced CSF-1R phosphorylation with an IC50 of 100 ng/ml or less, in one embodiment, with an IC50 of 50 ng/ml or less (in recombinant cells);
-抗CSF-1R抗体将表达人CSF-1R(SEQ ID No:15)的重组NIH3T3细胞的生长抑制80%或更多(与没有抗体相比),优选地90%或更多;-An anti-CSF-1R antibody inhibits the growth of recombinant NIH3T3 cells expressing human CSF-1R (SEQ ID No: 15) by 80% or more (compared to no antibody), preferably 90% or more;
-抗CSF-1R抗体将BeWo肿瘤细胞(ATCC CCL-98)的生长抑制80%或更多(在10μg/ml的抗体浓度;且与没有抗体相比),优选地90%或更多;- the anti-CSF-1R antibody inhibits the growth of BeWo tumor cells (ATCC CCL-98) by 80% or more (at an antibody concentration of 10 μg/ml; and compared with no antibody), preferably 90% or more;
-抗CSF-1R抗体抑制巨噬细胞分化(在一个实施方案中,抗CSF-1R抗体以1.5nM或更低的IC50,优选地以1.0nM或更低的IC50抑制单核细胞存活);或- the anti-CSF-1R antibody inhibits macrophage differentiation (in one embodiment, the anti-CSF-1R antibody inhibits monocyte survival with an IC50 of 1.5 nM or less, preferably with an IC50 of 1.0 nM or less); or
-抗CSF-1R抗体于35℃以KD=1.0x10-9mol/l或更低的结合亲和力结合人CSF-1R。- The anti-CSF-1R antibody binds to human CSF-1R at 35°C with a binding affinity of KD=1.0×10 −9 mol/l or lower.
重组方法和组合物Recombinant methods and compositions
优选地,通过重组手段来生成依照本发明的抗体。此类方法是现有技术中普遍已知的,并且包括在原核和真核细胞中表达蛋白质,随后分离抗体多肽,并且通常纯化至药学可接受纯度。对于蛋白质表达,通过标准的方法将编码轻和重链或其片段的核酸插入表达载体中。在合适的原核或真核宿主细胞,如CHO细胞、NS0细胞、SP2/0细胞、HEK293细胞、COS细胞、酵母、或大肠杆菌细胞中实施表达,并且自细胞(自上清液或在细胞裂解后)回收抗体。Preferably, the antibodies according to the invention are produced by recombinant means. Such methods are generally known in the art and include expression of the protein in prokaryotic and eukaryotic cells followed by isolation of the antibody polypeptide and usually purification to a pharmaceutically acceptable purity. For protein expression, nucleic acids encoding the light and heavy chains or fragments thereof are inserted into expression vectors by standard methods. Expression is carried out in suitable prokaryotic or eukaryotic host cells, such as CHO cells, NSO cells, SP2/0 cells, HEK293 cells, COS cells, yeast, or E. coli cells, and from cells (from supernatant or in cell lysates) after) to recover the antibody.
抗体的重组生成是现有技术中公知的,并且例如记载于综述文章Makrides,S.C.,Protein Expr.Purif.17(1999)183-202;Geisse,S.等,ProteinExpr.Purif.8(1996)271-282;Kaufman,R.J.,Mol.Biotechnol.16(2000)151-161;Werner,R.G.,Drug Res.48(1998)870-880。Recombinant production of antibodies is well known in the prior art and is described, for example, in the review articles Makrides, S.C., Protein Expr. Purif. 17 (1999) 183-202; Geisse, S. et al., Protein Expr. -282; Kaufman, R.J., Mol. Biotechnol. 16 (2000) 151-161; Werner, R.G., Drug Res. 48 (1998) 870-880.
抗体可以存在于整个细胞中,在细胞溶胞物中,或者为部分纯化的或基本上纯的形式。通过标准的技术,包括碱/SDS处理、CsCl分带、柱层析、琼脂糖凝胶电泳、和本领域中公知的其它技术来实施纯化以消除其它细胞组分或其它污染物,例如其它细胞核酸或蛋白质。见Ausubel,F.等编CurrentProtocols in Molecular Biology,Greene Publishing and Wiley Interscience,NewYork(1987)。Antibodies may be present in whole cells, in cell lysates, or in partially purified or substantially pure form. Purification is performed by standard techniques, including base/SDS treatment, CsCl banding, column chromatography, agarose gel electrophoresis, and other techniques known in the art to eliminate other cellular components or other contaminants, such as other cells nucleic acid or protein. See Ausubel, F. et al., eds. Current Protocols in Molecular Biology, Greene Publishing and Wiley Interscience, New York (1987).
NS0细胞中的表达由例如Barnes,L.M.等,Cytotechnology 32(2000)109-123;Barnes,L.M.等,Biotech.Bioeng.73(2001)261-270描述。瞬时表达由例如Durocher,Y.等,Nucl.Acids.Res.30(2002)E9描述。可变域的克隆由Orlandi,R.等,Proc.Natl.Acad.Sci.USA 86(1989)3833-3837;Carter,P.等,Proc.Natl.Acad.Sci.USA 89(1992)4285-4289;Norderhaug,L.等,J.Immunol.Methods 204(1997)77-87描述。一种优选的瞬时表达系统(HEK 293)由Schlaeger,E.-J.,Christensen,K.,于Cytotechnology 30(1999)71-83,及由Schlaeger,E.-J.,于J.Immunol.Methods 194(1996)191-199描述。Expression in NSO cells is described eg by Barnes, L.M. et al., Cytotechnology 32 (2000) 109-123; Barnes, L.M. et al., Biotech. Bioeng. 73 (2001) 261-270. Transient expression is described eg by Durocher, Y. et al., Nucl. Acids. Res. 30 (2002) E9. The cloning of variable domain is by Orlandi, R. etc., Proc.Natl.Acad.Sci.USA 86 (1989) 3833-3837; Carter, P. etc., Proc.Natl.Acad.Sci.USA 89 (1992) 4285- 4289; described by Norderhaug, L. et al., J. Immunol. Methods 204 (1997) 77-87. A preferred transient expression system (HEK 293) is described by Schlaeger, E.-J., Christensen, K., in Cytotechnology 30 (1999) 71-83, and by Schlaeger, E.-J., in J. Immunol. Methods 194 (1996) 191-199 describe.
适合于原核生物的控制序列例如包括启动子,任选地操纵基因序列,和核糖体结合位点。已知真核细胞利用启动子、增强子和多腺苷酸化信号。Control sequences suitable for prokaryotes include, for example, a promoter, optionally an operator sequence, and a ribosome binding site. Eukaryotic cells are known to utilize promoters, enhancers and polyadenylation signals.
在将核酸放置入与另一种核酸序列的功能性关系中时,它是“可操作连接的”。例如,若前序列或分泌前导的DNA以参与多肽分泌的前蛋白表达,则它与多肽的DNA可操作连接;若启动子或增强子影响序列的转录,则它与编码序列可操作连接;或者若核糖体结合位点定位为使得便于翻译,则它与编码序列可操作连接。一般而言,“可操作连接的”意指所连接的DNA序列是连续的,并且在分泌前导的情况中,是连续的且在读码框中。然而,增强子不必是连续的。通过在方便的限制性位点处的连接来实现连接。若不存在此类位点,则依照常规的实践使用合成的寡核苷酸衔接头或接头。A nucleic acid is "operably linked" when it is placed into a functional relationship with another nucleic acid sequence. For example, the DNA of a presequence or secretory leader is operably linked to the DNA of a polypeptide if it expresses a preprotein involved in the secretion of the polypeptide; it is operably linked to a coding sequence if a promoter or enhancer affects the transcription of the sequence; or A ribosome binding site is operably linked to a coding sequence if it is positioned such that it facilitates translation. Generally, "operably linked" means that the DNA sequences being linked are contiguous, and, in the case of a secretory leader, contiguous and in reading frame. However, enhancers need not be contiguous. Linking is accomplished by ligation at convenient restriction sites. If no such sites exist, synthetic oligonucleotide adapters or linkers are used in accordance with conventional practice.
通过常规的免疫球蛋白纯化规程,诸如例如蛋白A-Sepharose、羟磷灰石层析、凝胶电泳、透析、或亲和层析自培养液适当地分离单克隆抗体。容易使用常规规程将编码单克隆抗体的DNA和RNA分离并测序。杂交瘤细胞可以充当此类DNA和RNA的来源。一旦分离,可以将DNA插入表达载体中,然后将所述表达载体转染入不另外生成免疫球蛋白蛋白质的宿主细胞诸如HEK 293细胞、CHO细胞、或骨髓瘤细胞中,以获得重组单克隆抗体在宿主细胞中的合成。Monoclonal antibodies are suitably isolated from culture broth by conventional immunoglobulin purification procedures such as, for example, Protein A-Sepharose, hydroxyapatite chromatography, gel electrophoresis, dialysis, or affinity chromatography. DNA and RNA encoding the monoclonal antibodies are readily isolated and sequenced using routine procedures. Hybridoma cells can serve as a source of such DNA and RNA. Once isolated, the DNA can be inserted into an expression vector that is then transfected into a host cell that does not otherwise produce immunoglobulin protein, such as HEK 293 cells, CHO cells, or myeloma cells, to obtain recombinant monoclonal antibodies Synthesis in host cells.
通过本领域中已知的多种方法来制备编码抗CSF-1R抗体的氨基酸序列变体的核酸分子。这些方法包括但不限于自天然来源分离(在天然存在的氨基酸序列变体的情况中)或者通过对较早制备的变体或非变体型式的人源化抗CSF-1R抗体进行寡核苷酸介导的(或定点)诱变、PCR诱变、和盒式诱变来制备。Nucleic acid molecules encoding amino acid sequence variants of anti-CSF-1R antibodies are prepared by a variety of methods known in the art. These methods include, but are not limited to, isolation from natural sources (in the case of naturally-occurring amino acid sequence variants) or by oligonucleotide processing of variant or non-variant versions of humanized anti-CSF-1R antibodies prepared earlier. acid-mediated (or site-directed) mutagenesis, PCR mutagenesis, and cassette mutagenesis.
将依照本发明的重和轻链可变域与启动子、翻译起始、恒定区、3’非翻译区、多腺苷酸化、和转录终止的序列组合以形成表达载体构建体。可以将重和轻链表达构建体组合入单一载体中,共转染、连续转染、或分开转染入宿主细胞中,然后,将所述宿主细胞融合以形成表达这两条链的单一宿主细胞。The heavy and light chain variable domains according to the invention are combined with sequences for promoter, translation initiation, constant region, 3' untranslated region, polyadenylation, and transcription termination to form an expression vector construct. The heavy and light chain expression constructs can be combined into a single vector, co-transfected, serially transfected, or separately transfected into host cells which are then fused to form a single host expressing both chains cell.
如本文中所使用的,表述“细胞”、“细胞系”、和“细胞培养物”可互换使用,并且所有此类名称包括后代。如此,词语“转化体”和“经转化的细胞”包括原代主题细胞和自其衍生的培养物,而不管传递的次数。还应当理解,由于有意或无意的突变,所有后代在DNA内容上可以不是正好相同的。包括具有如在初始转化细胞中筛选的相同功能或生物学活性的变体后代。As used herein, the expressions "cell," "cell line," and "cell culture" are used interchangeably, and all such designations include progeny. Thus, the words "transformant" and "transformed cell" include the primary subject cell and cultures derived therefrom, regardless of the number of passages. It should also be understood that all progeny may not be exactly identical in DNA content, due to deliberate or unintentional mutations. Variant progeny having the same function or biological activity as screened for in the originally transformed cell are included.
抗体的“Fc部分”不直接牵涉抗体对抗原的结合,但是展现出各种效应器功能。“抗体的Fc部分”是熟练技术人员公知的术语,并且基于木瓜蛋白酶对抗体的切割限定。根据其重链恒定区的氨基酸序列,抗体或免疫球蛋白分成类:IgA、IgD、IgE、IgG和IgM,并且这些中的数种可以进一步分成亚类(同种型),例如IgG1、IgG2、IgG3、和IgG4、IgA1、和IgA2。依照重链恒定区,免疫球蛋白的不同类分别称作α、δ、ε、γ、和μ。抗体的Fc部分直接牵涉ADCC(抗体依赖性细胞介导的细胞毒性)和CDC(补体依赖性细胞毒性),其基于补体激活、C1q结合和Fc受体结合。补体激活(CDC)通过补体因子C1q与大多数IgG抗体亚类的Fc部分的结合来启动。虽然抗体对补体系统的影响依赖于某些条件,但是对C1q的结合是由Fc部分中的限定结合位点引起的。此类结合位点是现有技术中已知的,并且例如记载于Boackle,R.J.等,Nature 282(1979)742-743,Lukas,T.J.等,J.Immunol.127(1981)2555-2560,Brunhouse,R.和Cebra,J.J.,Mol.Immunol.16(1979)907-917,Burton,D.R.等,Nature 288(1980)338-344,Thommesen,J.E.等,Mol.Immunol.37(2000)995-1004,Idusogie,E.E.等,J.Immunol.164(2000)4178-4184,Hezareh,M.等,J.Virology 75(2001)12161-12168,Morgan,A.等,Immunology 86(1995)319-324,EP 0307434。此类结合位点是例如L234、L235、D270、N297、E318、K320、K322、P331和P329(依照Kabat,E.A.的EU索引的编号方式,参见下文)。亚类IgG1、IgG2和IgG3的抗体通常显示补体激活及C1q和C3结合,而IgG4不激活补体系统,而且不结合C1q和C3。The "Fc portion" of an antibody is not directly involved in binding of the antibody to antigen, but exhibits various effector functions. "Fc portion of an antibody" is a term well known to the skilled artisan and is defined based on cleavage of the antibody by papain. Depending on the amino acid sequence of the constant region of their heavy chains, antibodies or immunoglobulins are divided into classes: IgA, IgD, IgE, IgG, and IgM, and several of these can be further divided into subclasses (isotypes), such as IgG1, IgG2, IgG3, and IgG4, IgAl, and IgA2. According to the heavy-chain constant regions, the different classes of immunoglobulins are called alpha, delta, epsilon, gamma, and mu, respectively. The Fc portion of an antibody is directly involved in ADCC (antibody-dependent cell-mediated cytotoxicity) and CDC (complement-dependent cytotoxicity), which are based on complement activation, Clq binding and Fc receptor binding. Complement activation (CDC) is initiated by the binding of complement factor C1q to the Fc portion of most IgG antibody subclasses. Binding to C1q is caused by a defined binding site in the Fc portion, although the effect of the antibody on the complement system is dependent on certain conditions. Such binding sites are known in the prior art and are described, for example, in Boackle, R.J. et al., Nature 282 (1979) 742-743, Lukas, T.J. et al., J. Immunol. 127 (1981) 2555-2560, Brunhouse , R. and Cebra, J.J., Mol. Immunol.16 (1979) 907-917, Burton, D.R. et al., Nature 288 (1980) 338-344, Thommesen, J.E. et al., Mol. Immunol.37 (2000) 995-1004 , Idusogie, E.E. et al., J.Immunol.164(2000) 4178-4184, Hezareh, M. et al., J.Virology 75(2001) 12161-12168, Morgan, A. et al., Immunology 86(1995) 319-324, EP 0307434. Such binding sites are eg L234, L235, D270, N297, E318, K320, K322, P331 and P329 (numbering according to the EU index of Kabat, E.A., see below). Antibodies of the subclasses IgG1, IgG2, and IgG3 usually show complement activation and C1q and C3 binding, whereas IgG4 does not activate the complement system and does not bind C1q and C3.
在一个实施方案中,依照本发明的抗体包含自人起源衍生的Fc部分,和优选地人恒定区的所有其它部分。如本文中所使用的,术语“自人起源衍生的Fc部分”意指如下的Fc部分,其作为IgG1、IgG2、IgG3或IgG4亚类的人抗体的Fc部分,优选地,来自人IgG1亚类的Fc部分、来自人IgG1亚类的突变Fc部分(优选地具有L234A+L235A方面的突变)、来自人IgG4亚类的Fc部分或来自人IgG4亚类的突变Fc部分(优选地具有S228P方面的突变)。通常优选的是SEQ ID NO:11(人IgG1亚类)、SEQ ID NO:12(具有突变L234A和L235A的人IgG1亚类)、SEQ ID NO:13(人IgG4亚类)、或SEQ ID NO:14(具有突变S228P的人IgG4亚类)的人重链恒定区。In one embodiment, an antibody according to the invention comprises an Fc part derived from human origin, and preferably all other parts of the human constant region. As used herein, the term "Fc portion derived from human origin" means an Fc portion as the Fc portion of a human antibody of the IgG1, IgG2, IgG3 or IgG4 subclass, preferably, from the human IgG1 subclass Fc part from human IgG1 subclass (preferably with mutations in L234A+L235A), Fc part from human IgG4 subclass or mutant Fc part from human IgG4 subclass (preferably with mutations in S228P mutation). Generally preferred are SEQ ID NO: 11 (human IgG1 subclass), SEQ ID NO: 12 (human IgG1 subclass with mutations L234A and L235A), SEQ ID NO: 13 (human IgG4 subclass), or SEQ ID NO Human heavy chain constant region of :14 (human IgG4 subclass with mutation S228P).
在一个实施方案中,依照本发明的抗体的特征在于:恒定链是人起源的。此类恒定链是现有技术中公知的,并且例如由Kabat,E.A.(见例如Johnson,G.和Wu,T.T.,Nucleic Acids Res.28(2000)214-218)描述。例如,一种有用的人重链恒定区包含氨基酸序列SEQ ID NO:9。例如,一种有用的人轻链恒定区包含κ轻链恒定区氨基酸序列SEQ ID NO:10。进一步优选的是,抗体是小鼠起源的,并且包含依照Kabat的小鼠抗体的抗体可变序列框。In one embodiment, the antibody according to the invention is characterized in that the invariant chain is of human origin. Such invariant chains are well known in the prior art and are eg described by Kabat, E.A. (see eg Johnson, G. and Wu, T.T., Nucleic Acids Res. 28 (2000) 214-218). For example, one useful human heavy chain constant region comprises the amino acid sequence of SEQ ID NO:9. For example, a useful human light chain constant region comprises the amino acid sequence of a kappa light chain constant region of SEQ ID NO: 10. It is further preferred that the antibody is of mouse origin and comprises the antibody variable sequence frame of a mouse antibody according to Kabat.
免疫缀合物Immunoconjugate
本发明还提供了包含与一种或多种细胞毒剂,诸如化学治疗剂或药物、生长抑制剂、毒素(例如,蛋白质毒素、细菌、真菌、植物、或动物起源的酶活性毒素、或其片段)、或放射性同位素缀合的本文中的抗CSF-1R抗体的免疫缀合物。The present invention also provides compounds comprising and one or more cytotoxic agents, such as chemotherapeutics or drugs, growth inhibitors, toxins (e.g., protein toxins, enzymatically active toxins of bacterial, fungal, plant, or animal origin, or fragments thereof) ), or a radioisotope-conjugated immunoconjugate of the anti-CSF-1R antibody herein.
在一个实施方案中,免疫缀合物是抗体-药物缀合物(ADC),其中抗体与一种或多种药物缀合,包括但不限于美登木素生物碱(见美国专利No.5,208,020、5,416,064和欧洲专利EP 0 425 235B1);auristatin诸如单甲基auristatin药物模块DE和DF(MMAE和MMAF)(见美国专利No.5,635,483和5,780,588及7,498,298);多拉司他汀(dolastatin);加利车霉素(calicheamicin)或其衍生物(见美国专利No.5,712,374,5,714,586,5,739,116,5,767,285,5,770,701,5,770,710,5,773,001和5,877,296;Hinman等,Cancer Res.53:3336-3342(1993);及Lode等,Cancer Res.58:2925-2928(1998));蒽环类抗生素诸如道诺霉素(daunomycin)或多柔比星(doxorubicin)(见Kratz等,CurrentMed.Chem.13:477-523(2006);Jeffrey等,Bioorganic&Med.Chem.Letters16:358-362(2006);Torgov等,Bioconj.Chem.16:717-721(2005);Nagy等,Proc.Natl.Acad.Sci.USA 97:829-834(2000);Dubowchik等,Bioorg.&Med.Chem.Letters 12:1529-1532(2002);King等,J.Med.Chem.45:4336-4343(2002);及美国专利No.6,630,579);甲氨蝶呤;长春地辛(vindesine);紫杉烷(taxane)诸如多西他赛(docetaxel)、帕利他塞(paclitaxel)、larotaxel、tesetaxel、和ortataxel;单端孢霉素(trichothecene);和CC1065。In one embodiment, the immunoconjugate is an antibody-drug conjugate (ADC), wherein the antibody is conjugated to one or more drugs, including but not limited to maytansinoids (see U.S. Patent No. 5,208,020 , 5,416,064 and European Patent EP 0 425 235B1); auristatins such as monomethyl auristatin drug moieties DE and DF (MMAE and MMAF) (see U.S. Patent Nos. 5,635,483 and 5,780,588 and 7,498,298); dolastatin (dolastatin); Calicheamicin or its derivatives (see U.S. Patent Nos. 5,712,374, 5,714,586, 5,739,116, 5,767,285, 5,770,701, 5,770,710, 5,773,001 and 5,877,296; Hinman et al., Cancer Res. 53:3336-3342); and (1Lo93 , Cancer Res.58:2925-2928 (1998)); anthracyclines such as daunomycin (daunomycin) or doxorubicin (doxorubicin) (see Kratz et al., CurrentMed.Chem.13:477-523 (2006 ); Jeffrey et al, Bioorganic & Med.Chem.Letters 16:358-362 (2006); Torgov et al, Bioconj.Chem.16:717-721 (2005); Nagy et al, Proc.Natl.Acad.Sci.USA 97:829- 834 (2000); Dubowchik et al., Bioorg. & Med. Chem. Letters 12:1529-1532 (2002); King et al., J. Med. Chem. 45:4336-4343 (2002); and U.S. Patent No. 6,630,579); methotrexate; vindesine; taxanes such as docetaxel, paclitaxel, larotaxel, tesetaxel, and ortataxel; trichothecene; and CC1065.
在另一个实施方案中,免疫缀合物包含与酶活性毒素或其片段缀合的如本文中所描述的抗体,所述酶活性毒素包括但不限于白喉A链、白喉毒素的非结合活性片段、外毒素A链(来自铜绿假单胞菌(Pseudomonas aeruginosa))、蓖麻毒蛋白(ricin)A链、相思豆毒蛋白(abrin)A链、蒴莲根毒蛋白(modeccin)A链、α-帚曲霉素(sarcin)、油桐(Aleutites fordii)毒蛋白、香石竹(dianthin)毒蛋白、美洲商陆(Phytolaca americana)蛋白(PAPI、PAPII和PAP-S)、苦瓜(Momordica charantia)抑制物、麻疯树毒蛋白(curcin)、巴豆毒蛋白(crotin)、肥皂草(sapaonaria officinalis)抑制剂、白树毒蛋白(gelonin)、丝林霉素(mitogellin)、局限曲菌素(restrictocin)、酚霉素(phenomycin)、依诺霉素(enomycin)和单端孢菌素(trichothecenes)。In another embodiment, the immunoconjugate comprises an antibody as described herein conjugated to an enzymatically active toxin, or a fragment thereof, including but not limited to diphtheria A chain, non-binding active fragments of diphtheria toxin , exotoxin A chain (from Pseudomonas aeruginosa), ricin A chain, abrin A chain, modeccin A chain, α - Inhibition of sarcin, Aleutites fordii toxin, dianthin toxin, Phytolaca americana proteins (PAPI, PAPII and PAP-S), Momordica charantia Curcin, crotin, sapaonaria officinalis inhibitor, gelonin, mitogellin, restrictocin , phenomycin, enomycin and trichothecenes.
在另一个实施方案中,免疫缀合物包含与放射性原子缀合以形成放射性缀合物的如本文中所描述的抗体。多种放射性同位素可用于生成放射性缀合物。例子包括At211、I131、I125、Y90、Re186、Re188、Sm153、Bi212、P32、Pb212和Lu的放射性同位素。在使用放射性缀合物进行检测时,它可以包含供闪烁法研究用的放射性原子,例如tc99m或I123,或供核磁共振(NMR)成像(又称为磁共振成像,mri)用的自旋标记物,诸如再一次的碘-123、碘-131、铟-111、氟-19、碳-13、氮-15、氧-17、钆、锰或铁。In another embodiment, the immunoconjugate comprises an antibody as described herein conjugated to a radioactive atom to form a radioconjugate. A variety of radioisotopes are available for the generation of radioconjugates. Examples include At 211 , I 131 , I 125 , Y 90 , Re 186 , Re 188 , Sm 153 , Bi 212 , P 32 , Pb 212 , and radioactive isotopes of Lu. When radioconjugates are used for detection, it can contain radioactive atoms such as tc99m or I123 for scintillation studies, or spin labels for nuclear magnetic resonance (NMR) imaging (also known as magnetic resonance imaging, mri) substances such as again iodine-123, iodine-131, indium-111, fluorine-19, carbon-13, nitrogen-15, oxygen-17, gadolinium, manganese or iron.
可以使用多种双功能蛋白质偶联剂来生成抗体和细胞毒剂的缀合物,诸如N-琥珀酰亚氨基3-(2-吡啶基二硫代)丙酸酯(SPDP),琥珀酰亚氨基-4-(N-马来酰亚氨基甲基)环己烷-1-羧酸酯(SMCC),亚氨基硫烷(IT),亚氨酸酯(诸如盐酸己二酰亚氨酸二甲酯)、活性酯类(诸如辛二酸二琥珀酰亚氨基酯)、醛类(诸如戊二醛)、双叠氮化合物(诸如双(对-叠氮苯甲酰基)己二胺)、双重氮衍生物(诸如双(对-重氮苯甲酰基)-乙二胺)、二异氰酸酯(诸如甲苯2,6-二异氰酸酯)、和双活性氟化合物(诸如1,5-二氟-2,4-二硝基苯)的双功能衍生物。例如,可以如Vitetta等,Science 238:1098(1987)中所述制备蓖麻毒蛋白免疫毒素。碳-14标记的1-异硫氰酸苄基-3-甲基二亚乙基三胺五乙酸(MX-DTPA)是用于将放射性核苷酸与抗体偶联的例示性螯合剂。参见WO94/11026。接头可以是便于在细胞中释放细胞毒性药物的“可切割接头”。例如,可使用酸不稳定接头、肽酶敏感性接头、光不稳定接头、二甲基接头或含二硫化物接头(Chari等,Cancer Res 52:127-131(1992);美国专利No.5,208,020)。A variety of bifunctional protein coupling agents can be used to generate conjugates of antibodies and cytotoxic agents, such as N-succinimidyl 3-(2-pyridyldithio)propionate (SPDP), succinimidyl -4-(N-maleimidomethyl)cyclohexane-1-carboxylate (SMCC), iminothiolane (IT), imidate (such as dimethyl adipimidate hydrochloride esters), active esters (such as disuccinimidyl suberate), aldehydes (such as glutaraldehyde), bis-azides (such as bis(p-azidobenzoyl)hexamethylenediamine), double Nitrogen derivatives (such as bis(p-diazobenzoyl)-ethylenediamine), diisocyanates (such as toluene 2,6-diisocyanate), and bis-reactive fluorine compounds (such as 1,5-difluoro-2, 4-Dinitrobenzene) bifunctional derivatives. For example, a ricin immunotoxin can be prepared as described in Vitetta et al., Science 238:1098 (1987). Carbon-14 labeled 1-isothiocyanatobenzyl-3-methyldiethylenetriaminepentaacetic acid (MX-DTPA) is an exemplary chelating agent for conjugation of radionucleotides to antibodies. See WO94/11026. The linker may be a "cleavable linker" that facilitates release of the cytotoxic drug in the cell. For example, acid-labile linkers, peptidase-sensitive linkers, photolabile linkers, dimethyl linkers, or disulfide-containing linkers can be used (Chari et al., Cancer Res 52:127-131 (1992); U.S. Patent No. 5,208,020 ).
本文中的免疫缀合物或ADC明确涵盖,但不限于用交联试剂制备的此类缀合物,所述交联试剂包括但不限于BMPS、EMCS、GMBS、HBVS、LC-SMCC、MBS、MPBH、SBAP、SIA、SIAB、SMCC、SMPB、SMPH、sulfo-EMCS、sulfo-GMBS、sulfo-KMUS、sulfo-MBS、sulfo-SIAB、sulfo-SMCC、和sulfo-SMPB,及SVSB(琥珀酰亚氨基-(4-乙烯基砜)苯甲酸酯),它们是商品化的(例如,来自Pierce Biotechnology,Inc.,Rockford,IL.,U.S.A)。Immunoconjugates or ADCs herein expressly encompass, but are not limited to, such conjugates prepared with crosslinking reagents including, but not limited to, BMPS, EMCS, GMBS, HBVS, LC-SMCC, MBS, MPBH, SBAP, SIA, SIAB, SMCC, SMPB, SMPH, sulfo-EMCS, sulfo-GMBS, sulfo-KMUS, sulfo-MBS, sulfo-SIAB, sulfo-SMCC, and sulfo-SMPB, and SVSB (succinimidyl -(4-vinylsulfone)benzoate), which are commercially available (for example, from Pierce Biotechnology, Inc., Rockford, IL., U.S.A).
治疗性方法和组合物Therapeutic methods and compositions
本发明包括一种用于治疗需要治疗的患者的方法,其特征在于对患者施用治疗有效量的依照本发明的抗体。The invention includes a method for treating a patient in need thereof, characterized in that a therapeutically effective amount of an antibody according to the invention is administered to the patient.
本发明包括依照本发明的抗体用于治疗的用途。The invention includes the use of an antibody according to the invention for therapy.
本发明的一个优选的实施方案是在治疗“CSF-1R介导的疾病”中使用的本发明的CSF-1R抗体或用于制造在治疗“CSF-1R介导的疾病”(其可以如下描述)中的药物的本发明的CSF-1R抗体。A preferred embodiment of the present invention is the CSF-1R antibody of the present invention for use in the treatment of "CSF-1R-mediated diseases" or for the manufacture of "CSF-1R-mediated diseases" (which can be described as follows ) of the CSF-1R antibody of the invention as a drug.
存在着CSF-1R信号传导可能牵涉肿瘤生长和转移的3种独特的机制。第一种是已经在女性生殖系统(乳房、卵巢、子宫内膜、宫颈)中起源的肿瘤细胞中发现CSF配体和受体的表达(Scholl,S.M.等,J.Natl.Cancer Inst.86(1994)120-126;Kacinski,B.M.,Mol.Reprod.Dev.46(1997)71-74;Ngan,H.Y.等,Eur.J.Cancer 35(1999)1546-1550;Kirma,N.等,Cancer Res 67(2007)1918-1926),并且表达已经与乳腺癌异种移植物生长以及乳腺癌患者中不良预后联系起来。在一项研究中测试的约10-20%急性髓细胞性白血病、慢性髓细胞性白血病和脊髓发育不良患者中在CSF-1R中看到两处点突变,并且发现突变之一破坏受体周转(Ridge,S.A.等,Proc.Natl.Acad.Sci USA 87(1990)1377-1380)。然而,突变的发生在后来的研究中未能得到确认(Abu-Duhier,F.M.等,Br.J.Haematol.120(2003)464-470)。还在肝细胞癌(Yang,D.H.等,Hepatobiliary Pancreat.Dis.Int.3(2004)86-89)和特发性骨髓纤维化(Abu-Duhier,F.M.等,Br.J.Haematol.120(2003)464-470)的一些病例中发现突变。There are 3 unique mechanisms by which CSF-IR signaling may be involved in tumor growth and metastasis. The first is that the expression of CSF ligands and receptors has been found in tumor cells originating in the female reproductive system (breast, ovary, endometrium, cervix) (Scholl, S.M. et al., J. Natl. Cancer Inst. 86( 1994) 120-126; Kacinski, B.M., Mol.Reprod.Dev.46(1997) 71-74; Ngan, H.Y. et al., Eur.J.Cancer 35(1999) 1546-1550; Kirma, N. et al., Cancer Res 67 (2007) 1918-1926), and expression has been linked to breast cancer xenograft growth and poor prognosis in breast cancer patients. Two point mutations were seen in CSF-1R in approximately 10-20% of patients with acute myeloid leukemia, chronic myelogenous leukemia and myelodysplasia tested in one study, and one of the mutations was found to disrupt receptor turnover (Ridge, S.A. et al., Proc. Natl. Acad. Sci USA 87 (1990) 1377-1380). However, the occurrence of the mutation could not be confirmed in later studies (Abu-Duhier, F.M. et al., Br. J. Haematol. 120 (2003) 464-470). Also in hepatocellular carcinoma (Yang, D.H. et al., Hepatobiliary Pancreat. Dis. Int. 3 (2004) 86-89) and idiopathic myelofibrosis (Abu-Duhier, F.M. et al., Br.J.Haematol.120 (2003 )464-470) mutations were found in some cases.
色素沉着绒毛结节性滑膜炎(PVNS)和腱鞘滑膜巨大细胞肿瘤(TGCT)可以由于将M-CSF基因与胶原基因COL6A3融合,并导致M-CSF过表达的易位而发生(West,R.B.等,Proc.Natl.Acad.Sci.USA 103(2006)690-695)。提出景观效应(landscape effect)造成所得的肿瘤块,其由被表达M-CSF的细胞吸引的单核细胞组成。TGCT是可以自它们通常发生的指相对容易地除去的较小肿瘤。PVNS由于它可以在大的关节中复发而更具攻击性,而且不同样容易用手术控制。Pigmented villonodular synovitis (PVNS) and tenosynovial giant cell tumor (TGCT) can occur as a result of a translocation that fuses the M-CSF gene to the collagen gene COL6A3, leading to M-CSF overexpression (West, R.B. et al., Proc. Natl. Acad. Sci. USA 103 (2006) 690-695). A landscape effect is proposed to account for the resulting tumor mass consisting of monocytes attracted to M-CSF expressing cells. TGCTs are small tumors that can be removed relatively easily from where they usually arise. PVNS is more aggressive because it can recur in large joints and is not as easy to control surgically.
第二种机制基于阻断骨中转移部位经由M-CSF/CSF-1R的信号传导,其诱导破骨细胞发生(osteoclastogenesis)、骨吸收和溶骨性骨损伤。乳腺癌、多发性骨髓瘤和肺癌是已经发现转移至骨,并且引起溶骨性骨疾病,导致骨骼并发症的癌症的例子。由肿瘤细胞和基质释放的M-CSF与核因子κ-B配体的受体激活物RANKL协作诱导造血髓样单核细胞祖先分化成成熟的破骨细胞。在此过程期间,M-CSF通过对破骨细胞给予存活信号充当允许因子(Tanaka,S.等,J.Clin.Invest.91(1993)257-263)。用抗CSF-1R抗体对破骨细胞分化和成熟期间CSF-1R活性的抑制有可能阻止引起溶骨性疾病和转移性疾病中有关的骨骼相关事件的破骨细胞的不平衡活性。鉴于乳腺、肺癌和多发性骨髓瘤通常导致溶骨性损伤,前列腺癌中的骨转移最初具有成骨细胞出现,其中升高的骨形成活性导致“编织骨”,其与正常骨的典型薄片状结构不同。在疾病进展期间,骨损伤展现出显著的溶骨性组分及骨吸收的高血清水平,并且提示了抗吸收疗法可以是有用的。已经显示了二膦酸盐类抑制溶骨性损伤的形成,并且仅在具有激素不应性转移性前列腺癌的男性中减少骨骼相关事件的数目,但是在这点上,其对成骨细胞损伤的影响是有争议的,并且二膦酸盐类至今在预防骨转移或激素响应性前列腺癌中尚不是有益的。抗吸收剂在混合性溶骨性/成骨细胞性前列腺癌中的效果在临床中仍在研究(Choueiri,M.B.等,Cancer Metastasis Rev.25(2006)601-609;Vessella,R.L.和Corey,E.,Clin.Cancer Res.12(20Pt 2)(2006)6285s-6290s)。The second mechanism is based on blocking signaling via M-CSF/CSF-IR at metastatic sites in bone, which induces osteoclastogenesis, bone resorption and osteolytic bone damage. Breast cancer, multiple myeloma, and lung cancer are examples of cancers that have been found to metastasize to bone and cause osteolytic bone disease, leading to skeletal complications. M-CSF released by tumor cells and stroma cooperates with the receptor activator of nuclear factor kappa-B ligand RANKL to induce differentiation of hematopoietic myeloid mononuclear progenitors into mature osteoclasts. During this process, M-CSF acts as a permissive factor by imparting a survival signal to osteoclasts (Tanaka, S. et al., J. Clin. Invest. 91 (1993) 257-263). Inhibition of CSF-1R activity during osteoclast differentiation and maturation with anti-CSF-1R antibodies has the potential to prevent the unbalanced activity of osteoclasts responsible for bone-related events involved in osteolytic and metastatic disease. Whereas breast, lung cancer, and multiple myeloma often result in osteolytic lesions, bone metastases in prostate cancer initially have an osteoblastic appearance in which elevated bone-forming activity results in "woven bone" that differs from the typical lamellae of normal bone. The structure is different. During disease progression, bone lesions exhibit a marked osteolytic component and high serum levels of bone resorption, and suggest that antiresorptive therapy may be useful. Bisphosphonates have been shown to inhibit the formation of osteolytic lesions and reduce the number of bone-related events only in men with hormone-refractory metastatic prostate cancer, but at this point, their effect on osteoblast damage The effect of is controversial, and bisphosphonates have not been beneficial in preventing bone metastases or hormone-responsive prostate cancer to date. The effect of antiresorptive agents in mixed osteolytic/osteogenic prostate cancer is still under investigation in the clinic (Choueiri, M.B. et al., Cancer Metastasis Rev. 25 (2006) 601-609; Vessella, R.L. and Corey, E. ., Clin. Cancer Res. 12 (20Pt 2) (2006) 6285s-6290s).
第三种机制基于最近的观察结果,即存在于乳癌、前列腺、卵巢和宫颈癌的实体瘤中的肿瘤有关的巨噬细胞(TAM)与不良预后相关联(Bingle,L.等,J.Pathol.196(2002)254-265;Pollard,J.W.,Nat.Rev.Cancer 4(2004)71-78)。巨噬细胞通过M-CSF和其它趋化因子募集至肿瘤。然后,巨噬细胞可以经由分泌血管生成因子、蛋白酶和其它生长因子和细胞因子促成肿瘤进展,并且可以通过抑制CSF-1R信号传导而阻断。最近,Zins,K.等(Zins,K.等,CancerRes.67(2007)1038-1045)显示了肿瘤坏死因子α(TNFα)、M-CSF或这两者的组合的siRNA的表达会在相应siRNA的肿瘤内注射后将小鼠异种移植物模型中的肿瘤生长降低34%-50%。靶向由人SW620细胞分泌的TNFα的siRNA降低小鼠M-CSF水平,并且导致肿瘤中的巨噬细胞减少。另外,用针对M-CSF的抗原结合片段处理MCF7肿瘤异种移植物确实导致40%肿瘤生长抑制,反转对化疗剂的抗性,而且在与化疗剂组合给予时改善小鼠的存活(Paulus,P.等,Cancer Res.66(2006)4349-4356)。The third mechanism is based on recent observations that tumor-associated macrophages (TAMs) present in solid tumors of breast, prostate, ovary, and cervix are associated with poor prognosis (Bingle, L. et al., J. Pathol .196 (2002) 254-265; Pollard, J.W., Nat. Rev. Cancer 4 (2004) 71-78). Macrophages are recruited to tumors by M-CSF and other chemokines. Macrophages can then contribute to tumor progression via secretion of angiogenic factors, proteases, and other growth factors and cytokines, and can be blocked by inhibiting CSF-1R signaling. Recently, Zins, K. et al. (Zins, K. et al., CancerRes. 67 (2007) 1038-1045) showed that expression of siRNA for tumor necrosis factor alpha (TNFα), M-CSF, or a combination of both Intratumoral injection of siRNA reduces tumor growth in mouse xenograft models by 34%-50%. siRNA targeting TNFα secreted by human SW620 cells decreased M-CSF levels in mice and resulted in decreased macrophages in tumors. In addition, treatment of MCF7 tumor xenografts with antigen-binding fragments directed against M-CSF did result in 40% tumor growth inhibition, reversed resistance to chemotherapeutic agents, and improved survival of mice when given in combination with chemotherapeutic agents (Paulus, P. et al., Cancer Res. 66(2006) 4349-4356).
TAM是慢性炎症和癌症间新出现联系的唯一一个例子。炎症与癌症间的联系存在着别的证据,因为许多慢性疾病与升高的癌症风险有关,癌症在慢性炎症的部位出现,在许多癌症中找到炎症的化学介导物;删除炎症的细胞或化学介导物抑制实验性癌症的形成,并且抗炎剂的长期使用降低一些癌症的风险。那些幽门螺杆菌(H.pylori)诱导的胃炎(对于胃癌)、血吸虫病(对于膀胱癌)、HHVX(对于卡波西(Kaposi)肉瘤)、子宫内膜异位症(endometriosis)(对于卵巢癌)和前列腺炎(对于前列腺癌)中的许多炎性状况存在与癌症的联系(Balkwill,F.等,Cancer Cell 7(2005)211-217)。巨噬细胞是慢性炎症中的关键细胞,并且差别响应其微环境。存在着在功能性状态的连续区中被认为是极端的两类巨噬细胞:M1巨噬细胞牵涉1型反应。这些反应牵涉通过微生物产物的激活及随后杀死病原性微生物,这生成反应性氧中间体。极端的另一端是牵涉2型反应的M2巨噬细胞,所述2型反应促进细胞增殖、调整炎症和适应性免疫,而且促进组织重塑、血管发生和修复(Mantovani,A.等,Trends Immunol.25(2004)677-686)。导致建立的新生物形成的慢性炎症通常与M2巨噬细胞有关。介导炎性反应的一种枢要的细胞因子是TNF α,其名符其实在高剂量能刺激抗肿瘤免疫和出血性坏死,而且最近还已经发现了由肿瘤细胞表达,并且充当肿瘤促进物(Zins,K.等,CancerRes.67(2007)1038-1045;Balkwill,F.,Cancer Metastasis Rev.25(2006)409-416)。巨噬细胞就肿瘤而言的特定作用仍需要更好地了解,包括对其功能的潜在空间和时间依赖性和与特定肿瘤类型的相关性。TAMs are the only example of an emerging link between chronic inflammation and cancer. There is additional evidence for a link between inflammation and cancer, as many chronic diseases are associated with increased cancer risk, cancer arises at sites of chronic inflammation, and chemical mediators of inflammation are found in many cancers; cells or chemicals that delete inflammation The mediator inhibits the formation of experimental cancers, and long-term use of the anti-inflammatory reduces the risk of some cancers. Those with H. pylori-induced gastritis (for stomach cancer), schistosomiasis (for bladder cancer), HHVX (for Kaposi sarcoma), endometriosis (for ovarian cancer ) and prostatitis (for prostate cancer), there are many inflammatory conditions associated with cancer (Balkwill, F. et al., Cancer Cell 7 (2005) 211-217). Macrophages are key cells in chronic inflammation and respond differentially to their microenvironment. There are two classes of macrophages that are considered extreme on a continuum of functional states: M1 macrophages are involved in
如此,本发明的一个实施方案是在治疗癌症中使用的本发明的CSF-1R抗体。如本文中所使用的,术语“癌症”可以例如是肺癌、非小细胞肺(NSCL)癌、细支气管肺泡细胞肺癌、骨癌、胰腺癌、皮肤癌、头或颈癌、皮肤或眼内黑素瘤、子宫癌、卵巢癌、直肠癌、肛区癌、胃癌(stomach cancer)、胃癌(gastric cancer)、结肠癌、乳腺癌、子宫癌、输卵管癌、子宫内膜癌、宫颈癌、阴道癌、外阴癌、何杰金(Hodgkin)氏病、食管癌、小肠癌、内分泌系统癌、甲状腺癌、甲状旁腺癌、肾上腺癌、软组织肉瘤、尿道癌、阴茎癌、前列腺癌、膀胱癌、肾或输尿管癌、肾细胞癌、肾盂癌、间皮瘤、肝细胞癌、胆癌(biliary cancer)、中枢神经系统(CNS)新生物、脊柱轴肿瘤、脑干胶质瘤、多形性成胶质细胞瘤(glioblastoma multiforme)、星形细胞瘤、神经鞘瘤(schwanoma)、室鼓膜瘤(ependymona)、髓母细胞瘤、脑脊膜瘤、鳞状细胞癌、垂体腺瘤、淋巴瘤、淋巴细胞性白血病,包括任何上述癌症的顽固性型式、或一种或多种上述癌症的组合。优选地,此类癌症是乳腺癌、卵巢癌、宫颈癌、肺癌或前列腺癌。优选地,此类癌症的进一步特征在于CSF-1或CSF-1R表达或过表达。本发明的又一个实施方案是在原发性肿瘤和新的转移的同时治疗中使用的本发明的CSF-1R抗体。Thus, one embodiment of the invention is a CSF-IR antibody of the invention for use in the treatment of cancer. As used herein, the term "cancer" may be, for example, lung cancer, non-small cell lung (NSCL) cancer, bronchioloalveolar cell lung cancer, bone cancer, pancreatic cancer, skin cancer, head or neck cancer, melanoma of the skin or eye Tumor, uterine cancer, ovarian cancer, rectal cancer, anal region cancer, gastric cancer, gastric cancer, colon cancer, breast cancer, uterine cancer, fallopian tube cancer, endometrial cancer, cervical cancer, vaginal cancer , vulvar cancer, Hodgkin's disease, esophagus cancer, small intestine cancer, endocrine system cancer, thyroid cancer, parathyroid cancer, adrenal cancer, soft tissue sarcoma, urethral cancer, penile cancer, prostate cancer, bladder cancer, kidney or ureteral carcinoma, renal cell carcinoma, renal pelvis carcinoma, mesothelioma, hepatocellular carcinoma, biliary cancer, central nervous system (CNS) neoplasm, spinal axis tumor, brainstem glioma, glioma multiforme Glioblastoma multiforme, astrocytoma, schwanoma, ependymona, medulloblastoma, meningioma, squamous cell carcinoma, pituitary adenoma, lymphoma, lymphoma Leukemia, including refractory forms of any of the above cancers, or a combination of one or more of the above cancers. Preferably, such cancer is breast cancer, ovarian cancer, cervical cancer, lung cancer or prostate cancer. Preferably, such cancers are further characterized by CSF-1 or CSF-1R expression or overexpression. Yet another embodiment of the invention is the CSF-IR antibody of the invention for use in the simultaneous treatment of primary tumors and new metastases.
如此,本发明的另一个实施方案是在治疗牙周炎、组织细胞增多病X、骨质疏松、佩吉特(Paget)氏骨病(PDB)、由于癌症疗法所致的骨损失、假体周围骨质溶解、糖皮质素诱导的骨质疏松、类风湿性关节炎、银屑病关节炎(psiratic arthritis)、骨关节炎(osteoarthritis)、炎性关节炎(inflammatoryarthridities)、和炎症中使用的本发明的CSF-1R抗体。Thus, another embodiment of the present invention is in the treatment of periodontitis, histiocytosis X, osteoporosis, Paget's disease of bone (PDB), bone loss due to cancer therapy, prosthetic Peripheral osteolysis, glucocorticoid-induced osteoporosis, rheumatoid arthritis, psiratic arthritis, osteoarthritis, inflammatory arthritis, and inflammation CSF-1R antibodies of the invention.
Rabello,D.等,Biochem.Biophys.Res.Commun.347(2006)791-796已经表明CSF1基因中的SNP展现出与攻击性牙周炎:即一种由于牙槽骨的吸收引起牙损失的牙周组织炎性疾病的正关联。Rabello, D. et al., Biochem. Biophys. Res. Commun. 347 (2006) 791-796 have shown that a SNP in the CSF1 gene exhibits an association with aggressive periodontitis: a disease of tooth loss due to alveolar bone resorption. Positive association with periodontal inflammatory disease.
组织细胞增多病X(又称作朗格汉斯细胞组织细胞增多病,LCH)是一种表现为在骨和骨外LCH损伤中分化成破骨细胞的朗格汉斯树突细胞的增殖性疾病。朗格汉斯细胞自循环单核细胞衍生。发现在血清和损伤中测量到的升高的M-CSF水平与疾病严重性相关联(da Costa,C.E.等,J.Exp.Med.201(2005)687-693)。疾病主要在儿科患者群体中发生,并且在疾病变为系统性或者是复发性时不得不用化学疗法治疗。Histiocytosis X (also known as Langerhans cell histiocytosis, LCH) is a proliferative disorder of Langerhans dendritic cells that differentiate into osteoclasts in bony and extraskeletal LCH lesions. disease. Langerhans cells are derived from circulating monocytes. Elevated M-CSF levels measured in serum and lesions were found to correlate with disease severity (da Costa, C.E. et al., J. Exp. Med. 201 (2005) 687-693). The disease occurs mainly in the pediatric patient population and has to be treated with chemotherapy when the disease becomes systemic or recurrent.
骨质疏松的病理生理学是由形成骨的造骨细胞的损失和增加的破骨细胞依赖性骨吸收介导的。Cenci等已经描述了支持性数据,其显示了抗M-CSF抗体注射在切除卵巢的小鼠中保护骨密度并且抑制骨吸收(Cenci,S.等,J.Clin.Invest.105(2000)1279-1287)。最近,鉴定由于雌激素缺乏所致的绝经后骨损失间的潜在联系,并且发现了TNF α生成T细胞的存在影响骨代谢(Roggia,C.等,Minerva Med.95(2004)125-132)。一种可能的机制可能是TNFα在体内诱导M-CSF。通过阻断小鼠中TNFα诱导的骨质溶解的针对M-CSF的抗体的效果确认M-CSF在TNF-α诱导的破骨细胞发生中的重要作用,并且由此生成炎性关节炎的CSF-1R信号传导潜在的靶物的抑制剂(Kitaura,H.等,J.Clin.Invest.115(2005)3418-3427)。The pathophysiology of osteoporosis is mediated by loss of bone-forming osteoblasts and increased osteoclast-dependent bone resorption. Cenci et al. have described supporting data showing that anti-M-CSF antibody injections protect bone mineral density and inhibit bone resorption in ovariectomized mice (Cenci, S. et al., J. Clin. Invest. 105 (2000) 1279 -1287). Recently, a potential link between postmenopausal bone loss due to estrogen deficiency was identified and the presence of TNFα-producing T cells was found to affect bone metabolism (Roggia, C. et al., Minerva Med. 95 (2004) 125-132) . One possible mechanism could be that TNFα induces M-CSF in vivo. The essential role of M-CSF in TNF-α-induced osteoclastogenesis was confirmed by the effect of antibodies against M-CSF in blocking TNFα-induced osteolysis in mice, and thus generation of inflammatory arthritic CSF Inhibitors of potential targets of -1R signaling (Kitaura, H. et al., J. Clin. Invest. 115 (2005) 3418-3427).
佩吉特氏骨病(PDB)是骨质疏松后第二位最常见的骨代谢病症,其中骨周转升高的病灶性异常导致并发症诸如骨痛、畸形、病理性骨折和耳聋。已经鉴定出四种基因中的突变,其调节正常的破骨细胞功能,并且使个体有PDB及相关病症的素因:TNFRSF11A(其编码核因子(NF)κB的受体激活物(RANK),即破骨细胞功能的一种至关重要的调节物)中的插入突变、编码护骨蛋白(RANK配体的诱饵受体)的TNFRSF11B的失活突变、Sequestosome1基因(SQSTM1)(其编码NFκB途径中的重要支架蛋白)的突变和含有缬酪肽的蛋白质(VCP)基因中的突变。此基因编码VCP,其在使NFκB抑制剂靶向蛋白酶体所致降解中具有作用(Daroszewska,A.和Ralston,S.H.,Nat.Clin.Pract.Rheumatol.2(2006)270-277)。靶向性CSF-1R抑制剂提供一个间接阻断RANKL信号传导脱调节的机会,并且对目前使用的二膦酸盐类添加额外的治疗选项。Paget's disease of bone (PDB) is the second most common disorder of bone metabolism after osteoporosis, in which focal abnormalities of increased bone turnover lead to complications such as bone pain, deformity, pathological fractures and deafness. Mutations in four genes that regulate normal osteoclast function and predispose individuals to PDB and related disorders have been identified: TNFRSF11A (which encodes the receptor activator of nuclear factor (NF)κB (RANK), which a critical regulator of osteoclast function), inactivating mutations in TNFRSF11B encoding osteoprotegerin (a decoy receptor for RANK ligand), Sequestosome1 gene (SQSTM1) (which encodes important scaffold proteins) and mutations in the valoropeptide-containing protein (VCP) gene. This gene encodes a VCP that has a role in targeting NFKB inhibitors for degradation by the proteasome (Daroszewska, A. and Ralston, S.H., Nat. Clin. Pract. Rheumatol. 2 (2006) 270-277). Targeted CSF-1R inhibitors offer an opportunity to indirectly block deregulation of RANKL signaling and add additional therapeutic options to currently used bisphosphonates.
癌症疗法诱导的骨损失(尤其在乳腺和前列腺癌患者中)是一种别的适应症,其中靶向性CSF-1R抑制剂可以预防骨损失(Lester,J.E.等,Br.J.Cancer94(2006)30-35)。随着早期乳腺癌的预后改善,辅助疗法的长期后果变得更加重要,因为一些疗法,包括化学疗法、放射、芳香酶抑制剂和卵巢切除通过降低骨矿物密度影响骨代谢,导致骨质疏松及有关的骨折的风险升高(Lester,J.E.等,Br.J.Cancer 94(2006)30-35)。乳腺癌中的辅助芳香酶抑制剂疗法的等同方案是前列腺癌中的雄激素消融疗法,其导致骨矿物密度损失,而且显著增加骨质疏松相关骨折的风险(Stoch,S.A.等,J.Clin.Endocrinol.Metab.86(2001)2787-2791)。Cancer therapy-induced bone loss, especially in breast and prostate cancer patients, is an additional indication where targeted CSF-1R inhibitors can prevent bone loss (Lester, J.E. et al., Br.J.Cancer94 (2006 )30-35). As the prognosis of early breast cancer improves, the long-term consequences of adjuvant therapy become more important because some therapies, including chemotherapy, radiation, aromatase inhibitors, and ovariectomy affect bone metabolism by reducing bone mineral density, leading to osteoporosis and associated increased risk of fractures (Lester, J.E. et al., Br. J. Cancer 94 (2006) 30-35). The equivalent of adjuvant aromatase inhibitor therapy in breast cancer is androgen ablation therapy in prostate cancer, which results in loss of bone mineral density and significantly increases the risk of osteoporosis-related fractures (Stoch, S.A. et al., J. Clin. Endocrinol. Metab. 86 (2001) 2787-2791).
对CSF-1R信号传导的靶向性抑制在靶定的细胞类型包括破骨细胞和巨噬细胞时在其它适应症中也可能是有益的,例如治疗响应关节置换术(由于类风湿性关节炎)的特定并发症。由于假体周围骨损失及随后的假体松弛所致的植入失败是关节置换术的一个主要并发症,并且需要重复手术,对于个体患者和健康护理系统造成高的社会经济负担。至今,没有用于预防或抑制假体周围骨质溶解的得到批准的药物疗法(Drees,P.等,Nat.Clin.Pract.Rheumatol.3(2007)165-171)。Targeted inhibition of CSF-1R signaling may also be beneficial in other indications when targeting cell types including osteoclasts and macrophages, such as therapy in response to joint replacement (due to rheumatoid arthritis ) specific complications. Implant failure due to periprosthetic bone loss and subsequent prosthetic laxity is a major complication of joint replacement surgery and requires repeat surgery with a high socioeconomic burden on the individual patient and the healthcare system. To date, there is no approved drug therapy for preventing or inhibiting periprosthetic osteolysis (Drees, P. et al., Nat. Clin. Pract. Rheumatol. 3 (2007) 165-171).
糖皮质素诱导的骨质疏松(GIOP)是另一种适应症,其中CSF-1R抑制剂可以阻止由于那些慢性阻塞性肺疾病、哮喘和类风湿性关节炎的各种状况而给予的长期糖皮质类固醇使用后的骨损失(Guzman-Clark,J.R.等,ArthritisRheum.57(2007)140-146;Feldstein,A.C.等,Osteoporos.Int.16(2005)2168-2174)。Glucocorticoid-induced osteoporosis (GIOP) is another indication in which CSF-1R inhibitors can prevent long-term glucose given for various conditions of chronic obstructive pulmonary disease, asthma and rheumatoid arthritis Bone loss following corticosteroid use (Guzman-Clark, J.R. et al., Arthritis Rheum. 57 (2007) 140-146; Feldstein, A.C. et al., Osteoporos. Int. 16 (2005) 2168-2174).
类风湿性关节炎、银屑病关节炎和炎性关节炎本身是CSF-1R信号传导抑制剂的潜在适应症,因为它们由巨噬细胞组分和不同程度骨破坏组成(Ritchlin,C.T.等,J.Clin.Invest.111(2003)821-831)。骨关节炎和类风湿性关节炎是由巨噬细胞在结缔组织中积累和巨噬细胞浸润入滑液中(其由M-CSF至少部分介导)引起的炎性自身免疫性疾病。Campbell,I.K.等,J.Leukoc.Biol.68(2000)144-150表明人-关节组织细胞(软骨细胞、滑膜成纤维细胞)在体外生成M-CSF,并且其存在于类风湿性关节炎患者的滑液中,提示了它促成与疾病的发病机制有关的巨噬细胞浸润和滑膜组织增殖。对CSF-1R信号传导的抑制有可能控制关节中巨噬细胞的数目,并且减轻来自有关的骨破坏的疼痛。为了使不利影响最小化并且为了进一步了解CSF-1R信号传导在这些适应症中的影响,一种方法是特异性抑制CSF-1R,而不靶向无数其它激酶,诸如Raf激酶。Rheumatoid arthritis, psoriatic arthritis, and inflammatory arthritis themselves are potential indications for CSF-1R signaling inhibitors because they consist of a macrophage component and varying degrees of bone destruction (Ritchlin, C.T. et al., J. Clin. Invest. 111 (2003) 821-831). Osteoarthritis and rheumatoid arthritis are inflammatory autoimmune diseases caused by the accumulation of macrophages in connective tissue and the infiltration of macrophages into synovial fluid, which is at least partially mediated by M-CSF. Campbell, I.K. et al., J.Leukoc.Biol.68 (2000) 144-150 show that human-articular tissue cells (chondrocytes, synovial fibroblasts) produce M-CSF in vitro and that it is present in rheumatoid arthritis in the synovial fluid of patients, suggesting that it contributes to the infiltration of macrophages and proliferation of synovial tissue associated with the pathogenesis of the disease. Inhibition of CSF-1R signaling has the potential to control the number of macrophages in joints and alleviate pain from the associated bone destruction. To minimize adverse effects and to further understand the impact of CSF-1R signaling in these indications, one approach is to specifically inhibit CSF-1R without targeting myriad other kinases, such as Raf kinases.
最近的文献报告将增加的循环M-CSF与慢性冠状动脉疾病中的不良预后和动脉粥样硬化进展联系起来(Saitoh,T.等,J.Am.Coll.Cardiol.35(2000)655-665;Ikonomidis,I.等,Eur.Heart.J.26(2005)第1618-1624页);M-CSF通过帮助形成表达CSF-1R并且呈现初始斑块的泡沫细胞(具有摄取的氧化的LDL的巨噬细胞)影响动脉粥样硬化过程(Murayama,T.等,Circulation 99(1999)1740-1746)。Recent literature reports link increased circulating M-CSF to poor prognosis and progression of atherosclerosis in chronic coronary artery disease (Saitoh, T. et al., J. Am. Coll. Cardiol. 35 (2000) 655-665 ; Ikonomidis, I. et al., Eur.Heart.J.26 (2005) pp. 1618-1624); M-CSF acts by helping to form foam cells that express CSF-1R and present initial plaques (with uptake of oxidized LDL Macrophages) affect the atherosclerotic process (Murayama, T. et al., Circulation 99 (1999) 1740-1746).
在活化的小胶质中找到M-CSF和CSF-1R的表达和信号传导。小胶质(其是中枢神经系统的驻留巨噬细胞)可以通过多种损伤,包括感染和创伤性损伤活化。认为M-CSF是脑中炎性应答的一种关键调节物,并且M-CSF水平在HIV-1、脑炎、阿耳茨海默(Alzheimer)氏病(AD)和脑肿瘤中升高。由于M-CSF/CSF-1R的自分泌信号传导所致的小胶质细胞增生(microgliosis)导致诱导炎性细胞因子和一氧化氮释放,如通过例如使用实验性神经元损伤模型证明的(Hao,A.J.等,Neuroscience 112(2002)889-900;Murphy,G.M.,Jr.等,J.Biol.Chem.273(1998)20967-20971)。发现具有升高的CSF-1R表达的小胶质在AD中及在AD的淀粉样前体蛋白V717F转基因小鼠模型中围绕斑块(Murphy,G.M.,Jr.等,Am.J.Pathol.157(2000)895-904)。另一方面,与正常对照相比,在脑中具有较少小胶质的op/op小鼠导致A-β的原纤维沉积和神经元损失,提示了小胶质在op/op小鼠中在AD缺乏的形成中确实具有神经保护性功能(Kaku,M.等,Brain Res.Brain Res.Protoc.12(2003)104-108)。Expression and signaling of M-CSF and CSF-1R were found in activated microglia. Microglia, which are resident macrophages of the central nervous system, can be activated by a variety of insults, including infection and traumatic injury. M-CSF is thought to be a key regulator of inflammatory responses in the brain, and M-CSF levels are elevated in HIV-1, encephalitis, Alzheimer's disease (AD) and brain tumors. Microgliosis due to autocrine signaling of M-CSF/CSF-1R leads to induction of inflammatory cytokines and nitric oxide release, as demonstrated by, for example, using an experimental neuronal injury model (Hao , A.J. et al., Neuroscience 112 (2002) 889-900; Murphy, G.M., Jr. et al., J. Biol. Chem. 273 (1998) 20967-20971). Microglia with elevated CSF-1R expression were found to surround plaques in AD and in an amyloid precursor protein V717F transgenic mouse model of AD (Murphy, G.M., Jr. et al., Am. J. Pathol. 157 (2000) 895-904). On the other hand, op/op mice with fewer microglia in the brain resulted in fibril deposition of A-β and neuronal loss compared with normal controls, suggesting a role for microglia in op/op mice It does have a neuroprotective function in the development of AD deficiency (Kaku, M. et al., Brain Res. Brain Res. Protoc. 12 (2003) 104-108).
M-CSF和CSF-1R的表达和信号传导与炎性肠病(IBD)有关(WO2005/046657)。术语“炎性肠病”指以胃肠道中多个部位的慢性炎症为特征的严重的、慢性的肠道病症,并且明确包括溃疡性结肠炎(UC)和克罗恩(Crohn)氏病。The expression and signaling of M-CSF and CSF-IR are associated with inflammatory bowel disease (IBD) (WO2005/046657). The term "inflammatory bowel disease" refers to severe, chronic intestinal disorders characterized by chronic inflammation at multiple sites in the gastrointestinal tract, and specifically includes ulcerative colitis (UC) and Crohn's disease.
本发明包括用于治疗癌症的抗体,其特征在于包括结合人CSF-1R的抗体,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文提及的氨基酸序列和氨基酸序列片段。The present invention includes antibodies for use in the treatment of cancer characterized by comprising antibodies binding to human CSF-1R characterized by the above mentioned epitope binding properties or alternatively by the above mentioned Amino acid sequences and amino acid sequence fragments.
本发明包括用于治疗骨损失的抗体,其特征在于包括结合人CSF-1R的抗体,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文提及的氨基酸序列和氨基酸序列片段。The present invention includes antibodies for use in the treatment of bone loss characterized by comprising antibodies binding to human CSF-1R characterized by the above mentioned epitope binding properties or alternatively by the above mentioned And amino acid sequences and amino acid sequence fragments.
本发明包括用于预防或治疗转移的抗体,其特征在于包括结合人CSF-1R的抗体,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文提及的氨基酸序列和氨基酸序列片段。The present invention includes antibodies for the prevention or treatment of metastasis characterized by comprising antibodies binding to human CSF-1R characterized by the above mentioned epitope binding properties or alternatively by the above Amino acid sequences and amino acid sequence fragments mentioned.
本发明包括用于治疗炎性疾病的抗体,其特征在于包括结合人CSF-1R的抗体,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文提及的氨基酸序列和氨基酸序列片段。The present invention includes antibodies for use in the treatment of inflammatory diseases characterized by comprising antibodies binding to human CSF-1R characterized by the above mentioned epitope binding properties or alternatively by the above Amino acid sequences and amino acid sequence fragments mentioned.
本发明包括抗体用于治疗癌症或者备选地用于制造供治疗癌症用的药物的用途,所述抗体特征在于包括结合人CSF-1R的抗体,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文提及的氨基酸序列和氨基酸序列片段。The present invention includes the use of an antibody for the treatment of cancer or alternatively for the manufacture of a medicament for the treatment of cancer, said antibody being characterized in that it comprises an antibody that binds to human CSF-1R, said antibody that binds to human CSF-1R is characterized by the above The epitope binding properties mentioned herein or alternatively the amino acid sequences and amino acid sequence fragments mentioned above.
本发明包括抗体用于治疗骨损失或者备选地用于制造供治疗骨损失用的药物的用途,所述抗体特征在于包括结合人CSF-1R的抗体,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文提及的氨基酸序列和氨基酸序列片段。The present invention includes the use of an antibody for the treatment of bone loss or alternatively for the manufacture of a medicament for the treatment of bone loss, said antibody being characterized by comprising an antibody that binds to human CSF-1R, said antibody that binds to human CSF-1R is characterized by In the above mentioned epitope binding properties or alternatively in the above mentioned amino acid sequences and amino acid sequence fragments.
本发明包括抗体用于预防或治疗转移或者备选地用于制造供预防或治疗转移用的药物的用途,所述抗体特征在于包括结合人CSF-1R的抗体,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文提及的氨基酸序列和氨基酸序列片段。The present invention includes the use of an antibody for preventing or treating metastasis or alternatively for the manufacture of a medicament for preventing or treating metastasis, said antibody being characterized in that it comprises an antibody that binds to human CSF-1R, said human CSF-1R that binds Antibodies are characterized by the above mentioned epitope binding properties or alternatively by the above mentioned amino acid sequences and amino acid sequence fragments.
本发明包括抗体用于治疗炎性疾病或者备选地用于制造供治疗炎性疾病用的药物的用途,所述抗体特征在于包括结合人CSF-1R的抗体,所述结合人CSF-1R的抗体特征在于上文所提及的表位结合特性或者备选地上文提及的氨基酸序列和氨基酸序列片段。The present invention includes the use of an antibody for the treatment of an inflammatory disease or alternatively for the manufacture of a medicament for the treatment of an inflammatory disease, said antibody being characterized in that it comprises an antibody that binds to human CSF-1R, said human CSF-1R that binds Antibodies are characterized by the above mentioned epitope binding properties or alternatively by the above mentioned amino acid sequences and amino acid sequence fragments.
在一个实施方案中,依照本发明的抗体以25ng/ml或更低的IC50,优选地以20ng/ml或更低的IC50抑制CSF-1结合CSF-1R。可以如实施例2中所示测定抑制CSF-1结合CSF-1R的IC50。In one embodiment, the antibody according to the invention inhibits CSF-1 binding to CSF-1R with an IC50 of 25 ng/ml or lower, preferably with an IC50 of 20 ng/ml or lower. The IC50 for inhibition of CSF-1 binding to CSF-1R can be determined as shown in Example 2.
在一个实施方案中,依照本发明的抗体以100ng/ml或更低的IC50,优选地以50ng/ml或更低的IC50,更优选地以25ng/ml或更低的IC50抑制CSF-1诱导的CSF-1R磷酸化(在NIH3T3-CSF-1R重组细胞中)。可以如实施例3中所示测定CSF-1诱导的CSF-1R磷酸化的IC50。In one embodiment, the antibody according to the invention inhibits CSF-1 induction with an IC50 of 100 ng/ml or less, preferably with an IC50 of 50 ng/ml or less, more preferably with an IC50 of 25 ng/ml or less CSF-1R phosphorylation (in NIH3T3-CSF-1R recombinant cells). The IC50 of CSF-1-induced phosphorylation of CSF-1R can be determined as shown in Example 3.
在一个实施方案中,依照本发明的抗体将表达人CSF-1R(SEQ ID No:15)的重组NIH3T3细胞的生长抑制80%或更多(与没有抗体相比),优选地90%或更多。如在实施例6中所显示测定%生长抑制,其中测量%存活。自%存活,如下计算%生长抑制:%生长抑制=100-%存活。例如<CSF-1R>7G5.3B6显示100-0=100%的对表达wt人CSF-1R的NIH3T3细胞的生长抑制。In one embodiment, the antibody according to the present invention inhibits the growth of recombinant NIH3T3 cells expressing human CSF-1R (SEQ ID No: 15) by 80% or more (compared to no antibody), preferably 90% or more many. % growth inhibition was determined as shown in Example 6, where % survival was measured. From % survival, % growth inhibition was calculated as follows: % growth inhibition=100-% survival. For example <CSF-1R>7G5.3B6 showed 100-0=100% growth inhibition of NIH3T3 cells expressing wt human CSF-1R.
在一个实施方案中,依照本发明的抗体将表达人突变型CSF-1R L301SY969F(SEQ ID No:16)的重组NIH3T3细胞的生长刺激5%或更多(与没有抗体相比),优选地20%或更多。如在实施例6中所显示测定%生长刺激,其中测量%存活。自%存活,如下计算%生长刺激:%生长刺激=-(100-%存活)。例如,<CSF-1R>7G5.3B6显示-(100-0)=-(100-140)%=+40%的对表达突变型人CSF-1R的NIH3T3细胞的生长刺激。In one embodiment, the antibody according to the invention stimulates the growth of recombinant NIH3T3 cells expressing human mutant CSF-1R L301SY969F (SEQ ID No: 16) by 5% or more (compared to no antibody), preferably 20 %Or more. % growth stimulation was determined as shown in Example 6, where % survival was measured. From % Survival, % Growth Stimulation was calculated as follows: % Growth Stimulation = -(100-% Survival). For example, <CSF-1R>7G5.3B6 showed -(100-0)=-(100-140)%=+40% growth stimulation of NIH3T3 cells expressing mutant human CSF-1R.
在一个实施方案中,依照本发明的抗体将BeWo肿瘤细胞(ATCC CCL-98)的生长抑制80%或更多(抗体浓度为10μg/ml;且与没有抗体相比),优选地90%或更多。如在实施例7中所显示测定%生长抑制。例如<CSF-1R>7G5.3B6显示101%的对BeWo肿瘤细胞的生长抑制。In one embodiment, the antibody according to the invention inhibits the growth of BeWo tumor cells (ATCC CCL-98) by 80% or more (antibody concentration is 10 μg/ml; and compared to no antibody), preferably 90% or More. % growth inhibition was determined as shown in Example 7. For example <CSF-1R>7G5.3B6 showed 101% growth inhibition of BeWo tumor cells.
在一个实施方案中,依照本发明的抗体抑制巨噬细胞分化。在一个实施方案中,依照本发明的抗体以1.5nM或更低的IC50,优选地以1.0nM或更低的IC50抑制单核细胞存活。如实施例8中所显示测定对单核细胞存活的抑制。In one embodiment, an antibody according to the invention inhibits macrophage differentiation. In one embodiment, the antibody according to the invention inhibits monocyte survival with an IC50 of 1.5 nM or lower, preferably with an IC50 of 1.0 nM or lower. Inhibition of monocyte survival was determined as shown in Example 8.
本发明的又一个实施方案是用于生成针对CSF-1R的抗体的方法,其特征在于将编码依照本发明的结合人CSF-1R的人IgG1类抗体的重链的核酸(所述经修饰核酸)和编码所述抗体的轻链的核酸的序列插入表达载体中,在真核宿主细胞中插入所述载体,表达编码的蛋白质,并自宿主细胞或上清液回收。Yet another embodiment of the present invention is a method for producing antibodies against CSF-1R, characterized in that the nucleic acid encoding the heavy chain of a human IgG1 class antibody that binds to human CSF-1R according to the present invention (the modified nucleic acid ) and the nucleic acid sequence encoding the light chain of the antibody are inserted into an expression vector, the vector is inserted in a eukaryotic host cell, the encoded protein is expressed, and recovered from the host cell or supernatant.
药物配制剂pharmaceutical preparations
术语“药物配制剂”指如下的制剂,其处于使得容许其中含有的活性成分的生物学活性是有效的形式,且不含对会接受配制剂施用的受试者具有不可接受的毒性的别的组分。The term "pharmaceutical formulation" refers to a formulation which is in such a form as to allow the biological activity of the active ingredients contained therein to be effective and which contains no other substances which are unacceptably toxic to a subject to whom the formulation will be administered. components.
在另一方面,本发明提供了含有与药学可接受载体一起配制的一种或一组的本发明的单克隆抗体或其抗原结合部分的组合物,例如药物组合物。In another aspect, the invention provides compositions, eg, pharmaceutical compositions, comprising one or a panel of monoclonal antibodies or antigen-binding portions thereof of the invention formulated together with a pharmaceutically acceptable carrier.
“药学可接受载体”指药物配制剂中与活性成分不同的,且对受试者无毒的成分。药学可接受载体包括但不限于缓冲剂、赋形剂、稳定剂、或防腐剂。如本文中所使用的,“药学可接受载体”包括生理学相容的任何和所有溶剂、分散介质、涂层材料、抗细菌剂和抗真菌剂、等张剂和吸收延迟剂、等等。优选地,载体适合于注射或输注。"Pharmaceutically acceptable carrier" refers to an ingredient in a pharmaceutical formulation that is different from the active ingredient and is non-toxic to the subject. Pharmaceutically acceptable carriers include, but are not limited to, buffers, excipients, stabilizers, or preservatives. As used herein, "pharmaceutically acceptable carrier" includes any and all solvents, dispersion media, coating materials, antibacterial and antifungal agents, isotonic and absorption delaying agents, and the like that are physiologically compatible. Preferably, the carrier is suitable for injection or infusion.
可以通过本领域中已知的多种方法来施用本发明的组合物。如熟练技术人员会领会的,施用的路径和/或模式会随期望的结果而变化。Compositions of the invention can be administered by a variety of methods known in the art. As the skilled artisan will appreciate, the route and/or mode of administration will vary with the desired result.
药学可接受载体包括无菌水溶液或分散体和供制备无菌可注射溶液或分散体用的无菌粉剂。对药学活性物质使用此类介质和药剂是本领域中已知的。在水外,载体可以是例如等张缓冲盐水溶液。Pharmaceutically acceptable carriers include sterile aqueous solutions or dispersions and sterile powders for the preparation of sterile injectable solutions or dispersion. The use of such media and agents for pharmaceutically active substances is known in the art. Besides water, the carrier can be, for example, an isotonic buffered saline solution.
不管选择的施用路径,通过本领域技术人员已知的常规方法来将本发明的化合物(其可以以合适的水合形式使用)和/或本发明的药物组合物配制成药学可接受剂量形式。Irrespective of the chosen route of administration, the compounds of the invention (which may be used in suitably hydrated form) and/or the pharmaceutical compositions of the invention are formulated into pharmaceutically acceptable dosage forms by conventional methods known to those skilled in the art.
可以改变活性成分在本发明的药物组合物中的实际剂量水平,从而获得对于实现特定患者的期望的治疗响应、组成、和施用模式有效的,而对患者无毒的活性成分量(有效量)。选定的剂量水平会取决于多种药动学因素,包括所采用的本发明的特定组合物,或其酯、盐或酰胺的活性、施用路径、施用时间、所采用的特定化合物的排泄速率、与所采用的特定组合物组合使用的其它药物、化合物和/或材料、所治疗的患者的年龄、性别、重量、状况、一般健康和先前的医学史等医学领域公知的因素。Actual dosage levels of the active ingredients in the pharmaceutical compositions of the present invention can be varied to obtain an amount of the active ingredient that is effective to achieve a particular patient's desired therapeutic response, composition, and mode of administration, without being toxic to the patient (effective amount) . The selected dosage level will depend on a variety of pharmacokinetic factors, including the activity of the particular composition of the invention employed, or its ester, salt or amide, the route of administration, time of administration, the rate of excretion of the particular compound employed. , other drugs, compounds and/or materials used in combination with the particular composition employed, the age, sex, weight, condition, general health and prior medical history of the patient being treated are factors well known in the medical art.
本发明包括依照本发明的抗体用于治疗患有癌症,尤其是结肠、肺或胰腺癌的患者的用途。The present invention includes the use of an antibody according to the invention for the treatment of a patient suffering from cancer, especially colon, lung or pancreatic cancer.
本发明还包括用于治疗患有此类疾病的患者的方法。The invention also includes methods for treating patients suffering from such diseases.
本发明进一步提供了用于制造包含有效量的依照本发明的抗体及药学可接受载体的药物组合物的方法和依照本发明的抗体用于此类方法的用途。The present invention further provides methods for the manufacture of pharmaceutical compositions comprising an effective amount of an antibody according to the invention and a pharmaceutically acceptable carrier and the use of an antibody according to the invention for such methods.
本发明进一步提供了有效量的依照本发明的抗体用于制造药剂(优选地与药学可接受载体一起)以治疗患有癌症的患者的用途。The invention further provides the use of an effective amount of an antibody according to the invention for the manufacture of a medicament, preferably together with a pharmaceutically acceptable carrier, for the treatment of a patient suffering from cancer.
本发明还提供了有效量的依照本发明的抗体用于制造药剂(优选地与药学可接受载体一起)以治疗患有癌症的患者的用途。The present invention also provides the use of an effective amount of an antibody according to the present invention for the manufacture of a medicament, preferably together with a pharmaceutically acceptable carrier, for the treatment of a patient suffering from cancer.
制品products
在本发明的另一方面,提供了一种制品,其含有可用于治疗、预防和/或诊断上文所描述的病症的材料。制品包含容器和容器上或与容器联合的标签或包装插页。合适的容器包括例如瓶、管形瓶、注射器、IV溶液袋、等等。容器可以由多种材料诸如玻璃或塑料形成。容器容纳单独或与另一种组合物组合有效治疗、预防和/或诊断状况的组合物,并且可以具有无菌存取口(例如,容器可以是具有由皮下注射针可穿过的塞子的管形瓶或静脉内溶液袋)。组合物中的至少一种活性剂是本发明的抗体。标签或包装插页指示使用组合物来治疗选择的状况。此外,制品可以包含(a)具有其中含有的组合物的第一容器,其中组合物包含本发明的抗体;和(b)具有其中含有的组合物的第二容器,其中组合物包含别的细胞毒性或其它方面治疗性的药剂。在本发明的此实施方案中的制品可以进一步包含包装插页,其指示可以使用组合物来治疗特定的状况。或者/另外,制品可以进一步包含第二(或第三)容器,其包含药学可接受缓冲液,诸如抑菌性注射用水(BWFI)、磷酸盐缓冲盐水、Ringer氏溶液和右旋糖溶液。它可以进一步包含从商业和用户观点看期望的其它材料,包括其它缓冲液、稀释剂、滤器、针、和注射器。In another aspect of the invention there is provided an article of manufacture comprising materials useful for the treatment, prevention and/or diagnosis of the disorders described above. Articles of manufacture comprising containers and labels or package inserts on or associated with containers. Suitable containers include, for example, bottles, vials, syringes, IV solution bags, and the like. The container can be formed from a variety of materials such as glass or plastic. The container contains a composition effective to treat, prevent, and/or diagnose a condition, alone or in combination with another composition, and can have a sterile access opening (e.g., the container can be a tube with a stopper passable by a hypodermic needle vial or bag of intravenous solution). At least one active agent in the composition is an antibody of the invention. The label or package insert directs use of the composition to treat the condition of choice. Additionally, the article of manufacture may comprise (a) a first container having a composition contained therein, wherein the composition comprises an antibody of the invention; and (b) a second container having a composition contained therein, wherein the composition comprises additional cells Toxic or otherwise therapeutic agents. The article of manufacture in this embodiment of the invention may further comprise a package insert indicating that the composition may be used to treat a particular condition. Alternatively, or additionally, the article of manufacture may further comprise a second (or third) container comprising a pharmaceutically acceptable buffer, such as bacteriostatic water for injection (BWFI), phosphate buffered saline, Ringer's solution, and dextrose solution. It may further contain other materials desired from a commercial and user standpoint, including other buffers, diluents, filters, needles, and syringes.
应当理解,任何上述制品可以包含替换抗CSF-1R抗体或在CSF-1R抗体外的本发明的免疫缀合物。It will be appreciated that any of the foregoing preparations may comprise an immunoconjugate of the invention in place of or in addition to the anti-CSF-1R antibody.
提供以下实施例和序列表来帮助理解本发明,其实际范围在所附权利要求书中列出。应当理解,可以在不背离本发明精神的前提下在所列的规程中做出修改。The following examples and sequence listing are provided to aid in the understanding of the invention, the true scope of which is set forth in the appended claims. It should be understood that modifications may be made in the procedures set forth without departing from the spirit of the invention.
抗体保藏Antibody storage
序列的描述sequence description
提供以下实施例、序列表和图来帮助理解本发明,其实际范围在所附权利要求书中列出。应当理解,可以在不背离本发明精神的前提下在所列的规程中做出修改。The following examples, sequence listing and figures are provided to aid in the understanding of the present invention, the true scope of which is set forth in the appended claims. It should be understood that modifications may be made in the procedures set forth without departing from the spirit of the invention.
III.实施例III. Example
实施例1Example 1
生成抗CSF-1R抗体的杂交瘤细胞系的生成Generation of hybridoma cell lines producing anti-CSF-1R antibodies
NMRI小鼠的免疫规程Immunization protocol for NMRI mice
通过利用电穿孔用编码huCSF-1R的胞外域的表达载体pDisplayTM(Invitrogen,USA)免疫NMRI小鼠。用100μgDNA将每只小鼠免疫4次。在发现抗huCSF-1R的血清滴度足够时,在融合前4和3天用200μl PBS中50μg 1∶1混合物huCSF-1R ECD/huCSF-1R ECDhuFc嵌合物静脉内(i.v.)将小鼠额外加强一次。NMRI mice were immunized with the expression vector pDisplay ™ (Invitrogen, USA) encoding the extracellular domain of huCSF-1R by using electroporation. Each mouse was immunized 4 times with 100 μg DNA. When serum titers against huCSF-1R were found to be sufficient, mice were additionally treated intravenously (iv) with 50 μg of the 1:1 mixture huCSF-1R ECD/huCSF-1R ECDhuFc chimera in 200 μl PBS 4 and 3 days before fusion. Strengthen once.
抗原特异性ELISAantigen-specific ELISA
通过抗原特异性ELISA测定经免疫小鼠的血清中的抗CSF-1R滴度。Anti-CSF-1R titers in sera of immunized mice were determined by antigen-specific ELISA.
在具有0.1mg/ml生物素化的抗Fcγ(Jackson ImmunoResearch.,产品目录编号109-066-098)的链霉亲合素板(MaxiSorb;MicroCoat,DE,产品目录编号11974998/MC1099)上捕获0.3μg/ml huCSF 1R-huFc嵌合物(可溶性胞外域),并添加在PBS/0.05%Tween20/0.5%BSA中以1/800稀释的辣根过氧化物酶(HRP)缀合的F(ab’)2抗小鼠IgG(GE Healthcare,UK,产品目录编号NA9310V)。将来自所有抽液(tap)的血清在PBS/0.05%Tween20/0.5%BSA中以1/40稀释,并连续稀释,直至1/1638400。将稀释的血清添加至孔。抽液前血清作为阴性对照使用。使用自500ng/ml至0,25ng/ml的小鼠抗人CSF-IR单抗3291(R&D Systems,UK)的稀释系列作为阳性对照。将所有组分一起温育1,5小时。用PBST(PBS/0.2%Tween20)将孔清洗6次,并于RT用新鲜制备的ABTS溶液(1mg/ml)(ABTS:2,2’-连氮基二(3-乙基苯并噻唑啉-6-磺酸))显现测定法10分钟。于405nm测量吸光度。Capture 0.3 on Streptavidin plates (MaxiSorb; MicroCoat, DE, Cat. No. 11974998/MC1099) with 0.1 mg/ml biotinylated anti-Fcγ (Jackson ImmunoResearch., Cat. No. 109-066-098). μg/
杂交瘤生成hybridoma generation
可以将小鼠淋巴细胞分离,并使用基于PEG的标准方案与小鼠骨髓瘤细胞系融合以生成杂交瘤。然后,对所得的杂交瘤筛选抗原特异性抗体的生成。例如,用50%PEG将来自经免疫小鼠的脾衍生淋巴细胞的单细胞悬浮液与Ag8非分泌性小鼠骨髓瘤细胞P3X63Ag8.653(ATCC,CRL-1580)融合。将细胞以约104个在平底96孔微量滴定板中分配,接着在选择性培养基中温育约2周。然后,通过ELISA对个别孔筛选人抗CSF-1R单克隆IgM和IgG抗体。一旦发生广泛的杂交瘤生长,将分泌抗体的杂交瘤再分配,再次筛选,并且若在人IgG方面仍呈阳性,可以通过FACS亚克隆抗CSF-1R单克隆抗体。然后,在体外培养稳定的亚克隆以在组织培养液中生成抗体以进行表征。Mouse lymphocytes can be isolated and fused with mouse myeloma cell lines using standard PEG-based protocols to generate hybridomas. The resulting hybridomas are then screened for the production of antigen-specific antibodies. For example, single cell suspensions of spleen-derived lymphocytes from immunized mice were fused with Ag8 non-secreting mouse myeloma cells P3X63Ag8.653 (ATCC, CRL-1580) with 50% PEG. Cells were distributed in approximately 104 in flat-bottomed 96-well microtiter plates, followed by incubation in selective medium for approximately 2 weeks. Individual wells were then screened for human anti-CSF-IR monoclonal IgM and IgG antibodies by ELISA. Once extensive hybridoma growth has occurred, antibody-secreting hybridomas are redistributed, screened again, and if still positive for human IgG, anti-CSF-1R monoclonal antibodies can be subcloned by FACS. Stable subclones are then grown in vitro to produce antibodies in tissue culture for characterization.
杂交瘤的培养Hybridoma culture
将生成的鼠单抗杂交瘤在补充有2mM L-谷氨酰胺(GIBCO-产品目录编号35050-038)、1mM丙酮酸钠(GIBCO-产品目录编号11360-039)、1xNEAA(GIBCO-产品目录编号11140-035)、10%FCS(PAA-产品目录编号A15-649)、1x Pen Strep(Roche-产品目录编号1074440)、1x Nutridoma CS(Roche-产品目录编号1363743)、50μM巯基乙醇(GIBCO-产品目录编号31350-010)和50个U/ml IL 6小鼠(Roche-产品目录编号1444581)的RPMI1640(PAN-产品目录编号PO4-17500)中于37℃和5%CO2培养。The resulting murine mAb hybridomas were treated in the presence of 2 mM L-Glutamine (GIBCO - Catalog No. 35050-038), 1 mM Sodium Pyruvate (GIBCO - Catalog No. 11140-035), 10% FCS (PAA - Catalog No. A15-649), 1x Pen Strep (Roche - Catalog No. 1074440), 1x Nutridoma CS (Roche - Catalog No. 1363743), 50 μM Mercaptoethanol (GIBCO - Catalog No. Cat. No. 31350-010) and 50 U/ml IL 6 mice (Roche - Cat. No. 1444581) in RPMI1640 (PAN - Cat. No. PO4-17500) at 37 °C and 5% CO 2 .
实施例2Example 2
对CSF-1结合CSF-1R的抑制(ELISA)Inhibition of CSF-1 binding to CSF-1R (ELISA)
在384孔微量滴定板(MicroCoat,DE,产品目录编号464718)上于RT实施测试。在每个温育步骤后,用PBST将板清洗3次。Assays were performed at RT in 384-well microtiter plates (MicroCoat, DE, catalog number 464718). After each incubation step, the plates were washed 3 times with PBST.
开始时,用0.5mg/ml山羊F(ab’)2生物素化的抗Fcγ(JacksonImmunoResearch.,产品目录编号109-006-170)将板包被1小时。Initially, plates were coated with 0.5 mg/ml goat F(ab') 2 biotinylated anti-Fcγ (Jackson ImmunoResearch., Cat. No. 109-006-170) for 1 hour.
此后,用补充有0.2%Tween-20和2%BSA的PBS (Roche DiagnosticsGmbH,DE)将孔封闭0.5小时。将75ng/ml huCSF 1R-huFc嵌合物(可溶性胞外域)固定化于板达1小时。然后,将纯化的抗体在PBS/0.05%Tween20/0.5%BSA中的稀释液温育1小时。在添加3ng/ml CSF-1(Biomol,DE,产品目录编号60530)、50ng/ml生物素化的抗CSF-1克隆BAF216(R&D Systems,UK)和1:5000稀释的链霉亲合素HRP(Roche Diagnostics GmbH,DE,产品目录编号11089153001)的混合物达1小时后,用PBST将板清洗6次。使用抗CSF-IRSC-02,克隆2-4A5(Santa Cruz Biotechnology,US)(其抑制配体-受体相互作用)作为阳性对照。用新鲜制备的BM bluePOD底物溶液(BM blue3,3’-5,5’-四甲基联苯胺,Roche Diagnostics GmbH,DE,产品目录编号11484281001)将板于RT显现30分钟。于370nm测量吸光度。所有抗CSF-1R抗体都显示对CSF-1结合CSF-1R的显著抑制(见表1)。使用抗CSF-IR SC-02,克隆2-4A5(Santa Cruz Biotechnology,US)(其抑制配体-受体相互作用)作为参照对照。Thereafter, supplemented with 0.2%Tween Wells were blocked with 20 and 2% BSA in PBS (Roche Diagnostics GmbH, DE) for 0.5 hours. 75ng/
表1:抑制CSF-1/CSF-1R相互作用的计算IC50值Table 1: Calculated IC50 values for inhibition of CSF-1/CSF-1R interaction
实施例3Example 3
对NIH3T3-CSF-1R重组细胞中CSF-1诱导的CSF-1R磷酸化的抑制Inhibition of CSF-1-induced CSF-1R phosphorylation in NIH3T3-CSF-1R recombinant cells
将4.5x103个用全长CSF-1R的表达载体以逆转录病毒感染的NIH 3T3细胞在DMEM(PAA产品目录编号E15-011)、2mM L-谷氨酰胺(Sigma,产品目录编号G7513)、2mM丙酮酸钠、1x非必需氨基酸、10%FKS(PAA,产品目录编号A15-649)和100μg/ml PenStrep(Sigma,产品目录编号P4333[10mg/ml])中培养,直至它们达到汇合。此后,将细胞用补充有亚硒酸钠[5ng/ml](Sigma,产品目录编号S9133)、转铁蛋白[10μg/ml](Sigma,产品目录编号T8158)、BSA[400μg/ml](Roche Diagnostics GmbH,产品目录编号10735078)、4mM L-谷氨酰胺(Sigma,产品目录编号G7513)、2mM丙酮酸钠(Gibco,产品目录编号11360)、1x非必需氨基酸(Gibco,产品目录编号:11140-035)、2-巯基乙醇[0,05mM](Merck,产品目录编号M7522)、100μg/ml和PenStrep(Sigma,产品目录编号P4333)的无血清DMEM培养基(PAA产品目录编号E15-011)清洗,并在30μl相同培养基中温育16小时以容许受体上调。对细胞添加10μl稀释的抗CSR-1R抗体达1.5小时。然后,用10μl100ng/ml huM-CSF-1(Biomol产品目录编号60530)将细胞刺激5分钟。温育后,除去上清液,将细胞用80μl冰冷的PBS清洗两次,并添加每10ml缓冲液50μl新鲜制备的冰冷的裂解缓冲液(150mM NaCl/20mM Tris pH 7.5/1mMEDTA/1mM EGTA/1%Triton X-100/1片蛋白酶抑制剂片(Roche DiagnosticsGmbH产品目录编号1836170)/10μl/ml磷酸酶抑制剂混合物1(Sigma产品目录编号P-2850,100x储液)/10μl/ml蛋白酶抑制剂1(Sigma产品目录编号P-5726,100x储液)/10μl/ml 1M NaF)。在冰上30分钟后,将板在摇板仪上有力摇动3分钟,然后,以2200rpm离心10分钟(Heraeus Megafuge 10)。4.5x10 3 NIH 3T3 cells infected with the full-length CSF-1R expression vector with retrovirus in DMEM (PAA catalog number E15-011), 2mM L-glutamine (Sigma, catalog number G7513), 2 mM sodium pyruvate, 1x non-essential amino acids, 10% FKS (PAA, catalog number A15-649) and 100 μg/ml PenStrep (Sigma, catalog number P4333 [10 mg/ml]) until they reached confluency. Thereafter, the cells were treated with sodium selenite [5 ng/ml] (Sigma, catalog number S9133), transferrin [10 μg/ml] (Sigma, catalog number T8158), BSA [400 μg/ml] (Roche Diagnostics GmbH, catalog number 10735078), 4 mM L-glutamine (Sigma, catalog number G7513), 2 mM sodium pyruvate (Gibco, catalog number 11360), 1x non-essential amino acids (Gibco, catalog number: 11140- 035), 2-Mercaptoethanol [0,05 mM] (Merck, catalog number M7522), 100 μg/ml and PenStrep (Sigma, catalog number P4333) in serum-free DMEM medium (PAA catalog number E15-011) wash , and incubated in 30 μl of the same medium for 16 hours to allow receptor upregulation. 10 [mu]l of diluted anti-CSR-1R antibody was added to the cells for 1.5 hours. Cells were then stimulated with 10 μl of 100 ng/ml huM-CSF-1 (Biomol Cat# 60530) for 5 minutes. After incubation, the supernatant was removed, the cells were washed twice with 80 μl of ice-cold PBS, and 50 μl of freshly prepared ice-cold lysis buffer (150 mM NaCl/20 mM Tris pH 7.5/1 mM EDTA/1 mM EGTA/1 %Triton X-100/1 protease inhibitor tablet (Roche Diagnostics GmbH catalog number 1836170)/10 μl/ml Phosphatase inhibitor cocktail 1 (Sigma catalog number P-2850, 100x stock solution)/10 μl/ml protease inhibitor 1 (Sigma catalog number P-5726, 100x stock solution)/10 μl/ml 1M NaF). After 30 minutes on ice, the plate was shaken vigorously on a plate shaker for 3 minutes and then centrifuged at 2200 rpm for 10 minutes (Heraeus Megafuge 10).
用Elisa分析细胞裂解物中磷酸化的和总的CSF-1受体的存在。对于检测磷酸化的受体,依照供应商的用法说明书使用来自R&D Systems(产品目录编号DYC3268-2)的试剂盒。对于检测总的CSF-1R,通过使用试剂盒中含有的捕捉抗体将10μl裂解物固定化于板上。此后,添加1:750稀释的生物素化的抗CSF-1R抗体BAF329(R&D Systems)和1:1000稀释的链霉亲合素-HRP缀合物。在60分钟后,用新鲜制备的ABTS溶液将板显现,并检测吸光度。数据以没有抗体情况中的阳性对照%计算,并且表示比率值磷酸/总受体。在没有添加M-CSF-1的情况中限定阴性对照。使用抗CSF-1R SC-02,克隆2-4A5(Santa Cruz Biotechnology,US,还可见Sherr,C.J.等,Cell 41(1985)665-676)(其抑制配体-受体相互作用)作为参照对照。Cell lysates were analyzed by Elisa for the presence of phosphorylated and total CSF-1 receptors. For detection of phosphorylated receptors, a kit from R&D Systems (Catalogue No. DYC3268-2) was used according to the supplier's instructions. For detection of total CSF-1R, 10 μl of lysate were immobilized on the plate by using the capture antibody contained in the kit. Thereafter, a 1:750 dilution of biotinylated anti-CSF-1R antibody BAF329 (R&D Systems) and a 1:1000 dilution of streptavidin-HRP conjugate were added. After 60 minutes, with freshly prepared ABTS The solution visualizes the plate and the absorbance is measured. Data are calculated as % positive control in the absence of antibody and represent ratio values phospho/total receptor. A negative control was defined in the absence of M-CSF-1 addition. Anti-CSF-1R SC-02, clone 2-4A5 (Santa Cruz Biotechnology, US, see also Sherr, CJ et al., Cell 41 (1985) 665-676), which inhibits ligand-receptor interactions, was used as a reference control .
表2:抑制CSF-1受体磷酸化的计算IC50值Table 2: Calculated IC50 values for inhibition of CSF-1 receptor phosphorylation
实施例4Example 4
测定抗CSF-1R抗体对CSF-1R的亲和力Determination of the affinity of anti-CSF-1R antibodies for CSF-1R
仪器:BIACOREA100Instrument: BIACORE A100
芯片:CM5(Biacore BR-1006-68)Chip: CM5 (Biacore BR-1006-68)
偶联:胺偶联Coupling: amine coupling
缓冲液:PBS (Biacore BR-1006-72),pH 7.4,35℃Buffer: PBS (Biacore BR-1006-72), pH 7.4, 35°C
对于亲和力测量,将36μg/ml抗小鼠Fcγ抗体(来自山羊,Jackson ImmunoReasearch JIR115-005-071)与芯片表面偶联以捕捉针对CSF-1R的抗体。将CSF-1R ECD(R&D-Systems 329-MR或内部亚克隆的pCMV-presS-HisAvitag-hCSF-1R-ECD)在溶液中以多种浓度添加。通过于35℃注射CSF-1R 1.5分钟测量结合;通过于35°用缓冲液清洗芯片表面10分钟测量解离。使用抗CSF-IR SC-02,克隆2-4A5(Santa Cruz Biotechnology,US;还可见Sherr,C.J.等,Cell 41(1985)665-676)(其抑制配体-受体相互作用)作为参照对照。For affinity measurements, 36 μg/ml anti-mouse Fcγ antibody (from goat, Jackson ImmunoReasearch JIR115-005-071) was coupled to the chip surface to capture antibody against CSF-1R. CSF-1R ECD (R&D-Systems 329-MR or in-house subcloned pCMV-presS-HisAvitag-hCSF-1R-ECD) was added in solution at various concentrations. Binding was measured by injecting CSF-1R for 1.5 minutes at 35°C; dissociation was measured by washing the chip surface with buffer for 10 minutes at 35°C. Anti-CSF-IR SC-02, clone 2-4A5 (Santa Cruz Biotechnology, US; see also Sherr, C.J. et al., Cell 41 (1985) 665-676 ), which inhibits ligand-receptor interactions, was used as a reference control .
对于动力学参数的计算,使用朗缪尔(Langmuir)1∶1模型。For the calculation of kinetic parameters, the Langmuir 1:1 model was used.
表3:于35℃通过SPR(BIACOREA100)测量的亲和力数据Table 3: At 35°C by SPR (BIACORE A100) measured affinity data
实施例5Example 5
通过利用SPR基于交叉竞争对抗CSF-1R单克隆抗体的表位定位Epitope mapping of anti-CSF-1R monoclonal antibody based on cross-competition by utilizing SPR
仪器:BIACOREA100Instrument: BIACORE A100
芯片:CM5(Biacore BR-1006-68)Chip: CM5 (Biacore BR-1006-68)
偶联:胺偶联Coupling: amine coupling
缓冲液:PBS PBS(Biacore BR-1006-72),pH 7.4,25℃Buffer: PBS PBS (Biacore BR-1006-72), pH 7.4, 25°C
对于经由交叉竞争的表位定位测定法,将36μg/ml抗小鼠Fcγ抗体或抗大鼠Fcγ抗体(来自山羊,Jackson Immuno Research产品目录编号115-005-071和产品目录编号112-005-071)与传感器芯片表面偶联以呈现针对CSF-1R的抗体。在自5μg/ml抗CSF-1R单克隆抗体捕捉后,用250μg/ml小鼠或大鼠免疫球蛋白(Pierce产品目录编号31202和Pierce产品目录编号31233)封闭捕捉抗体的自由结合能力,接着注射12.5μg/ml CSF-1R(R&D-Systems产品目录编号329-MR)达2分钟。通过注射2分钟分析第二抗CSF-1R抗体的结合,通过用缓冲液清洗5分钟测量解离。于25℃进行测定法和测量。针对用相同芯片设置,但仅仅不注射CSF-1R的点比照第二抗CSF-1R抗体的特异性结合。已经以第二抗CSF-1R抗体的预期结合响应的百分比(%)计算交叉竞争数据。第二抗体结合的项“预期结合响应的百分比(%)”通过“100*相对响应(大体_稳定性_早期)/rMax”计算,其中rMax通过“相对响应(大体_稳定性_晚期)*抗体分子量/抗原分子量”计算,如记载于Biacore表位定位仪(对于BIACOREA100仪)的。For epitope mapping assays via cross-competition, 36 μg/ml anti-mouse Fcγ antibody or anti-rat Fcγ antibody (from goat, Jackson Immuno Research Cat. No. 115-005-071 and Cat. No. 112-005-071 ) coupled to the sensor chip surface to present antibodies against CSF-1R. After capture from 5 μg/ml anti-CSF-1R monoclonal antibody, block the free binding capacity of the capture antibody with 250 μg/ml mouse or rat immunoglobulin (Pierce Cat. No. 31202 and Pierce Cat. No. 31233), followed by injection 12.5 μg/ml CSF-1R (R&D-Systems catalog number 329-MR) for 2 minutes. Binding of the secondary anti-CSF-1R antibody was analyzed by injection for 2 minutes and dissociation was measured by washing with buffer for 5 minutes. Assays and measurements were performed at 25°C. Specific binding of the second anti-CSF-1R antibody was compared against spots with the same chip setup but only without CSF-1R injection. Cross-competition data have been calculated as a percentage (%) of the expected binding response of the second anti-CSF-1R antibody. The term "Percentage (%) of expected binding response" for secondary antibody binding is calculated by "100*relative response(mass_stability_early)/rMax", where rMax is calculated by "relative response(mass_stability_late)* Antibody Molecular Weight/Antigen Molecular Weight" calculation, as described in Biacore epitope localizer (for BIACORE A100 meter).
还自相同抗体1和2的对计算最小限度结合响应。其获得的最大值+100%,优选地50%设置为显著结合竞争的阈值(见表X,例如对于抗体<CSF-1R>7G5.3B6,计算的阈值是3+3=6,优选地3+1.5=4.5)。如此,“与<CSF-1R>7G5.3B6结合相同表位的抗CSF-1R抗体”具有小于6,优选地小于4.5的预期结合响应的百分比(%)。Minimal binding responses were also calculated from pairs of the
使用抗CSF-1R SC-02,克隆2-4A5(Santa Cruz Biotechnology,US,还可见Sherr,C.J.等,Cell 41(1985)665-676)(其抑制配体-受体相互作用)作为参照对照。Anti-CSF-1R SC-02, clone 2-4A5 (Santa Cruz Biotechnology, US, see also Sherr, C.J. et al., Cell 41 (1985) 665-676 ), which inhibits ligand-receptor interactions, was used as a reference control .
表4:抗CSF-1R抗体的经由交叉竞争数据的表位定位Table 4: Epitope mapping of anti-CSF-1R antibodies via cross-competition data
结果指示与<CSF-1R>7G5.3B6结合相同表位的抗体与SC-02,克隆2-4A5相比结合另一表位。The results indicate that the antibody that binds the same epitope as <CSF-1R>7G5.3B6 binds another epitope than SC-02, clone 2-4A5.
实施例6Example 6
抗CSF-1R单克隆抗体处理下对3D培养中NIH3T3-CSF-1R重组细胞的生长抑制(CellTiterGlo测定法)Growth inhibition of NIH3T3-CSF-1R recombinant cells in 3D culture by anti-CSF-1R monoclonal antibody treatment (CellTiterGlo assay)
将用全长野生型CSF-1R (SEQ ID No:15)或突变型CSF-1R L301S Y969F(SEQ ID No:16)的表达载体以逆转录病毒感染的NIH 3T3细胞在补充有2mML-谷氨酰胺、2mM丙酮酸钠和非必需氨基酸和10%胎牛血清(Sigma,Taufkirchen,Germany)的DMEM高葡萄糖培养基(PAA,Pasching,Austria)中在经聚HEMA(聚(2-羟乙基甲基丙烯酸酯))(Polysciences,Warrington,PA,USA))包被(以防止对塑料表面的粘着)的皿上培养。将细胞在用5ng/ml亚硒酸钠、10mg/ml转铁蛋白、400μg/ml BSA和0.05mM 2-巯基乙醇替换血清的培养基中接种。在用100ng/ml huCSF-1(Biomol,Hamburg,Germany)处理时,表达wtCSF-1R的细胞形成三维生长的密集球状体,即一种称作贴壁不依赖性的特性。这些球状体极其类似原位实体瘤的三维结构和组织。突变型CSF-1R重组细胞能够不依赖于CSF-1配体形成球状体。将球状体培养物在存在10μg/ml抗体的情况中温育3天。使用CellTiterGlo测定法来通过测量细胞的ATP含量检测细胞存活力。The NIH 3T3 cells infected with retrovirus will be supplemented with 2mML-glutamine with the expression vector of full-length wild type CSF-1R (SEQ ID No:15) or mutant CSF-1R L301S Y969F (SEQ ID No:16). Amide, 2 mM sodium pyruvate and non-essential amino acids and 10% fetal bovine serum (Sigma, Taufkirchen, Germany) in DMEM high glucose medium (PAA, Pasching, Austria) in polyHEMA (poly(2-hydroxyethylmethyl) (Polysciences, Warrington, PA, USA)) (Polysciences, Warrington, PA, USA)) coated (to prevent adhesion to plastic surfaces). Cells were seeded in medium replaced with serum with 5 ng/ml sodium selenite, 10 mg/ml transferrin, 400 μg/ml BSA and 0.05 mM 2-mercaptoethanol. Upon treatment with 100 ng/ml huCSF-1 (Biomol, Hamburg, Germany), cells expressing wtCSF-1R formed dense spheroids that grew three-dimensionally, a property termed anchorage-independent. These spheroids closely resemble the three-dimensional structure and organization of solid tumors in situ. Mutant CSF-1R recombinant cells were able to form spheroids independently of CSF-1 ligand. Spheroid cultures were incubated for 3 days in the presence of 10 μg/ml antibody. CellTiterGlo assay was used to detect cell viability by measuring the ATP content of cells.
表5:table 5:
**15个不同实验的平均值, ** Average of 15 different experiments,
***6个不同实验的平均值 *** Average of 6 different experiments
实施例7Example 7
抗CSF-1R单克隆抗体处理下对3D培养中BeWo肿瘤细胞的生长抑制(CellTiterGlo测定法)Growth Inhibition of BeWo Tumor Cells in 3D Culture by Anti-CSF-1R Monoclonal Antibody Treatment (CellTiterGlo Assay)
将BeWo绒毛膜癌细胞(ATCC CCL-98)在补充有10%FBS(Sigma)和2mML-谷氨酰胺的F12K培养基(Sigma,Steinheim,Germany)中培养。将5x104个细胞/孔在含有补充有0.5%FBS和5%BSA的F12K培养基的96孔经聚HEMA(聚(2-羟乙基甲基丙烯酸酯))包被的板中接种。伴随地,添加200ng/ml huCSF-1和10μg/ml不同抗CSF-1R单克隆抗体,并温育6天。使用CellTiterGlo测定法来通过以相对光单位(RLU)测量细胞的ATP含量检测细胞存活力。在用不同抗CSF-1R抗体(10μg/ml)处理BeWo球状体培养物时,观察到对CSF-1诱导的生长的抑制。为了计算抗体介导的抑制,自所有样品减去未刺激的BeWo细胞的均值RLU数值。将经CSF-1刺激的细胞的均值RLU数值任意设置成100%。用CSF-1刺激并用抗CSF-1R抗体处理的细胞的均值RLU数值以CSF-1刺激的RLU的%计算。表6显示了计算的数据;图1描绘了均值RLU数值。每个均值数值自一式三份导出。BeWo choriocarcinoma cells (ATCC CCL-98) were cultured in F12K medium (Sigma, Steinheim, Germany) supplemented with 10% FBS (Sigma) and 2 mM L-glutamine. 5x104 cells/well were seeded in 96-well polyHEMA (poly(2-hydroxyethylmethacrylate)) coated plates containing F12K medium supplemented with 0.5% FBS and 5% BSA. Concomitantly, 200 ng/ml huCSF-1 and 10 μg/ml different anti-CSF-1R monoclonal antibodies were added and incubated for 6 days. CellTiterGlo assay was used to detect cell viability by measuring the ATP content of cells in relative light units (RLU). Inhibition of CSF-1 -induced growth was observed when BeWo spheroid cultures were treated with different anti-CSF-1R antibodies (10 μg/ml). To calculate antibody-mediated inhibition, the mean RLU value of unstimulated BeWo cells was subtracted from all samples. The mean RLU value of CSF-1 stimulated cells was arbitrarily set to 100%. Mean RLU values of cells stimulated with CSF-1 and treated with anti-CSF-1R antibody were calculated as % of CSF-1 stimulated RLU. Table 6 shows the calculated data; Figure 1 depicts the mean RLU values. Each mean value was derived from triplicate.
表6:Table 6:
实施例8Example 8
抗CSF-1R单克隆抗体处理下对巨噬细胞分化/单核细胞存活的抑制(CellTiterGlo测定法)Inhibition of Macrophage Differentiation/Monocyte Survival by Anti-CSF-1R Monoclonal Antibody Treatment (CellTiterGlo Assay)
使用RosetteSepTM人单核细胞富集混合物(StemCell Tech.-产品目录编号15028)自外周血分离单核细胞。将富集的单核细胞群于37℃和5%CO2在补充有10FCS(GIBCO-产品目录编号011-090014M)、4mM L-谷氨酰胺(GIBCO-产品目录编号25030)和1xPenStrep(Roche产品目录编号1 074440)的100μl RPMI 1640(Gibco-产品目录编号31870)中接种入96孔微量滴定板(2.5x104个细胞/孔)中。在对培养基添加150ng/ml huCSF-1时,可以观察到清楚的分化成粘附的巨噬细胞。此分化可以通过添加抗CSF-1R抗体抑制。伴随地,单核细胞存活受到影响,并且可以通过CellTiterGlo(CTG)分析来分析。自抗体处理对单核细胞存活的浓度依赖性抑制,计算IC50(见表7)。Mononuclear cells were isolated from peripheral blood using RosetteSep ™ Human Monocyte Enrichment Cocktail (StemCell Tech. - Cat. No. 15028). Incubate the enriched monocyte population at 37°C and 5% CO in a solution supplemented with 10FCS (GIBCO - Cat. No. 011-090014M), 4 mM L-Glutamine (GIBCO - Cat. No. 25030) and 1xPenStrep (
表7:Table 7:
PCTPCT
由受理局填写To be completed by the receiving Office
由国际局填写To be completed by the International Bureau
PCT/RO/134表Form PCT/RO/134
Claims (16)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP10002269 | 2010-03-05 | ||
| EP10002269.8 | 2010-03-05 | ||
| PCT/EP2011/053213 WO2011131407A1 (en) | 2010-03-05 | 2011-03-03 | Antibodies against human csf-1r and uses thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102918061A true CN102918061A (en) | 2013-02-06 |
| CN102918061B CN102918061B (en) | 2016-06-08 |
Family
ID=43020630
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201180012506.9A Expired - Fee Related CN102918061B (en) | 2010-03-05 | 2011-03-03 | Antibodies against human CSF-1R and uses thereof |
Country Status (10)
| Country | Link |
|---|---|
| US (3) | US9221910B2 (en) |
| EP (1) | EP2542588A1 (en) |
| JP (1) | JP5989547B2 (en) |
| KR (1) | KR101647871B1 (en) |
| CN (1) | CN102918061B (en) |
| BR (1) | BR112012022046A2 (en) |
| CA (1) | CA2789071C (en) |
| MX (1) | MX336682B (en) |
| RU (1) | RU2617966C2 (en) |
| WO (1) | WO2011131407A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105658236A (en) * | 2013-08-30 | 2016-06-08 | Ucb生物制药私人有限公司 | Antibodies |
| CN105873949A (en) * | 2014-01-31 | 2016-08-17 | 勃林格殷格翰国际有限公司 | Novel anti-BAFF antibodies |
Families Citing this family (267)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7632929B2 (en) | 2000-04-20 | 2009-12-15 | The Board Of Trustees Of The University Of Arkansas | Methamphetamine-like hapten compounds, linkers, carriers and compositions and uses thereof |
| HUE044179T2 (en) | 2009-12-10 | 2019-10-28 | Hoffmann La Roche | Antibodies binding preferentially human csf1r extracellular domain 4 and their use |
| US9169323B2 (en) | 2010-03-05 | 2015-10-27 | Hoffmann-La Roche Inc. | Antibodies against human CSF-1R |
| EP2542588A1 (en) | 2010-03-05 | 2013-01-09 | F. Hoffmann-La Roche AG | Antibodies against human csf-1r and uses thereof |
| NZ603193A (en) | 2010-05-04 | 2014-07-25 | Five Prime Therapeutics Inc | Antibodies that bind csf1r |
| JP2014520873A (en) * | 2011-07-18 | 2014-08-25 | ザ ユニバーシティ オブ メルボルン | Use of c-Fms antagonists |
| BR112014012624A2 (en) | 2011-12-15 | 2018-10-09 | F Hoffmann-La Roche Ag | antibodies, pharmaceutical composition, nucleic acid, expression vectors, host cell, method for producing a recombinant antibody and use of the antibody |
| AR090263A1 (en) * | 2012-03-08 | 2014-10-29 | Hoffmann La Roche | COMBINED ANTIBODY THERAPY AGAINST HUMAN CSF-1R AND USES OF THE SAME |
| US20130302322A1 (en) | 2012-05-11 | 2013-11-14 | Five Prime Therapeutics, Inc. | Methods of treating conditions with antibodies that bind colony stimulating factor 1 receptor (csf1r) |
| KR102134088B1 (en) | 2012-08-24 | 2020-07-14 | 더 리젠츠 오브 더 유니버시티 오브 캘리포니아 | Antibodies and vaccines for use in treating ror1 cancers and inhibiting metastasis |
| HK1212214A1 (en) | 2012-08-31 | 2016-06-10 | 戊瑞治疗有限公司 | Methods of treating conditions with antibodies that bind colony stimulating factor 1 receptor (csf1r) |
| PL2914612T3 (en) | 2012-10-30 | 2019-04-30 | Hoffmann La Roche | Purification of polypeptides using dual stage tangential-flow ultrafiltration |
| US9023353B2 (en) * | 2013-03-13 | 2015-05-05 | The Board Of Trustees Of The University Of Arkansas | Anti-(+)—methamphetamine monoclonal antibodies |
| US9856316B2 (en) | 2013-04-12 | 2018-01-02 | Morphosys Ag | Antibodies targeting M-CSF |
| AR095882A1 (en) | 2013-04-22 | 2015-11-18 | Hoffmann La Roche | ANTIBODY COMBINATION THERAPY AGAINST HUMAN CSF-1R WITH A TLR9 AGONIST |
| AR097584A1 (en) | 2013-09-12 | 2016-03-23 | Hoffmann La Roche | ANTIBODY COMBINATION THERAPY AGAINST HUMAN CSF-1R AND ANTIBODIES AGAINST HUMAN PD-L1 |
| CA2934953C (en) | 2013-12-24 | 2022-09-20 | Bristol-Myers Squibb Company | Substituted pyrido[3,2-b]indoyl compounds and pharmaceutical compositions thereof as anticancer agents |
| RU2016149812A (en) | 2014-06-06 | 2018-07-17 | Флексус Байосайенсиз, Инк. | IMMUNO REGULATORY MEANS |
| PT3151921T (en) | 2014-06-06 | 2019-11-21 | Bristol Myers Squibb Co | ANTIBODIES AGAINST GLUCOCORTICOID-INDUCED TUMOR NECROSIS FACTOR RECEIVER AND USES OF THE SAME |
| BR112016029460A2 (en) | 2014-06-23 | 2017-10-24 | Five Prime Therapeutics Inc | Methods To Treat Rheumatoid Arthritis |
| EA036261B1 (en) | 2014-10-29 | 2020-10-20 | Файв Прайм Терапьютикс, Инк. | Combination therapy for cancer |
| HRP20210809T8 (en) | 2014-10-29 | 2021-09-17 | Bicyclerd Limited | Bicyclic peptide ligands specific for mt1-mmp |
| CA2964276A1 (en) | 2014-11-05 | 2016-05-12 | Flexus Biosciences, Inc. | Immunoregulatory agents |
| AR102537A1 (en) | 2014-11-05 | 2017-03-08 | Flexus Biosciences Inc | IMMUNOMODULATING AGENTS |
| UY36390A (en) | 2014-11-05 | 2016-06-01 | Flexus Biosciences Inc | MODULATING COMPOUNDS OF INDOLAMINE ENZYME 2,3-DIOXYGENASE (IDO), ITS SYNTHESIS METHODS AND PHARMACEUTICAL COMPOSITIONS CONTAINING THEM |
| DK3221363T3 (en) | 2014-11-21 | 2020-08-10 | Bristol Myers Squibb Co | ANTIBODIES TO CD73 AND USES THEREOF |
| TW201630907A (en) | 2014-12-22 | 2016-09-01 | 必治妥美雅史谷比公司 | TGF[beta]R antagonists |
| EP3237447B1 (en) | 2014-12-22 | 2020-12-02 | Five Prime Therapeutics, Inc. | Anti-csf1r antibodies for treating pvns |
| MA40662B1 (en) | 2014-12-23 | 2020-12-31 | Bristol Myers Squibb Co | Antibodies against tigit |
| ES2789331T3 (en) | 2015-03-02 | 2020-10-26 | Rigel Pharmaceuticals Inc | TGF-beta inhibitors |
| HK1247922A1 (en) | 2015-04-03 | 2018-10-05 | Bristol-Myers Squibb Company | Inhibitors of indoleamine 2,3-dioxygenase for the treatment of cancer |
| WO2016162505A1 (en) | 2015-04-08 | 2016-10-13 | F-Star Biotechnology Limited | Her2 binding agent therapies |
| PT3283527T (en) | 2015-04-13 | 2021-03-03 | Five Prime Therapeutics Inc | COMBINATION THERAPEUTIC FOR CANCER |
| US10722517B2 (en) | 2015-05-08 | 2020-07-28 | Memorial Sloan Kettering Cancer Center | Compositions and methods for treatment of glioma |
| ES2815683T3 (en) | 2015-05-11 | 2021-03-30 | Bristol Myers Squibb Co | Tricyclic compounds as antineoplastic agents |
| US9725449B2 (en) | 2015-05-12 | 2017-08-08 | Bristol-Myers Squibb Company | Tricyclic compounds as anticancer agents |
| WO2016183115A1 (en) | 2015-05-12 | 2016-11-17 | Bristol-Myers Squibb Company | 5h-pyrido[3,2-b]indole compounds as anticancer agents |
| BR112017024899A2 (en) | 2015-05-21 | 2018-11-13 | Harpoon Therapeutics, Inc. | trispecific binding proteins and methods of use. |
| EP3303390A1 (en) | 2015-05-27 | 2018-04-11 | UCB Biopharma SPRL | Method for the treatment of neurological disease |
| DK3303396T5 (en) | 2015-05-29 | 2024-10-07 | Bristol Myers Squibb Co | ANTIBODIES AGAINST OX40 AND USES THEREOF |
| KR20180021833A (en) | 2015-06-29 | 2018-03-05 | 더 락커펠러 유니버시티 | Antibodies to CD40 with enhanced agonist activity |
| WO2017019757A1 (en) | 2015-07-28 | 2017-02-02 | Bristol-Myers Squibb Company | Tgf beta receptor antagonists |
| JP2018525415A (en) | 2015-08-25 | 2018-09-06 | ブリストル−マイヤーズ スクイブ カンパニーBristol−Myers Squibb Company | TGF beta receptor antagonist |
| CN108884147B (en) | 2015-09-23 | 2024-02-27 | 百时美施贵宝公司 | Fibronectin-based scaffold molecules that bind glypican 3 |
| CN108738324B (en) | 2015-11-19 | 2022-06-21 | 百时美施贵宝公司 | Anti-glucocorticoid-induced tumor necrosis factor receptor (GITR) antibodies and uses thereof |
| WO2017106291A1 (en) | 2015-12-15 | 2017-06-22 | Bristol-Myers Squibb Company | Cxcr4 receptor antagonists |
| IL295230A (en) | 2016-03-04 | 2022-10-01 | Bristol Myers Squibb Co | Combination therapy with anti-cd73 antibodies |
| SG11201808821WA (en) | 2016-04-18 | 2018-11-29 | Celldex Therapeutics Inc | Agonistic antibodies that bind human cd40 and uses thereof |
| CN109414420A (en) | 2016-05-04 | 2019-03-01 | 百时美施贵宝公司 | The inhibitor and its application method of indole amine 2,3-dioxygenase |
| JP2019516682A (en) | 2016-05-04 | 2019-06-20 | ブリストル−マイヤーズ スクイブ カンパニーBristol−Myers Squibb Company | Indoleamine 2,3-dioxygenase inhibitors and methods of use thereof |
| KR20190004742A (en) | 2016-05-04 | 2019-01-14 | 브리스톨-마이어스 스큅 컴퍼니 | Inhibitors of indoleamine 2,3-dioxygenase and methods of use thereof |
| CN109415320A (en) | 2016-05-04 | 2019-03-01 | 百时美施贵宝公司 | The inhibitor and its application method of indole amine 2,3-dioxygenase |
| JP2019522627A (en) | 2016-05-04 | 2019-08-15 | ブリストル−マイヤーズ スクイブ カンパニーBristol−Myers Squibb Company | Indoleamine 2,3-dioxygenase inhibitor and method of use thereof |
| US11623958B2 (en) | 2016-05-20 | 2023-04-11 | Harpoon Therapeutics, Inc. | Single chain variable fragment CD3 binding proteins |
| WO2017210335A1 (en) | 2016-06-01 | 2017-12-07 | Bristol-Myers Squibb Company | Imaging methods using 18f-radiolabeled biologics |
| SG11201810509PA (en) | 2016-06-20 | 2018-12-28 | Kymab Ltd | Anti-pd-l1 antibodies |
| US10688181B2 (en) | 2016-06-27 | 2020-06-23 | The Regents Of The University Of California | Cancer treatment combinations |
| GB201611530D0 (en) * | 2016-07-01 | 2016-08-17 | Alligator Bioscience Ab | Novel polypeptides |
| TWI806832B (en) | 2016-07-12 | 2023-07-01 | 美商銳新醫藥公司 | 2,5-disubstituted 3-methyl pyrazines and 2,5,6-trisubstituted 3-methyl pyrazines as allosteric shp2 inhibitors |
| BR112019000431A2 (en) | 2016-07-14 | 2019-07-09 | Bristol-Myers Squibb Company | antibodies against tim3 and their uses |
| WO2018017633A1 (en) | 2016-07-21 | 2018-01-25 | Bristol-Myers Squibb Company | TGF Beta RECEPTOR ANTAGONISTS |
| JOP20170154B1 (en) * | 2016-08-01 | 2023-03-28 | Omeros Corp | Compositions and methods of inhibiting masp-3 for the treatment of various diseases and disorders |
| CN109790220A (en) | 2016-08-25 | 2019-05-21 | 豪夫迈·罗氏有限公司 | Intermittent dosing of anti-CSF-1R antibodies in combination with macrophage activators |
| JP2019528300A (en) | 2016-08-26 | 2019-10-10 | ブリストル−マイヤーズ スクイブ カンパニーBristol−Myers Squibb Company | Indoleamine 2,3-dioxygenase inhibitors and methods of use thereof |
| JP2019528301A (en) | 2016-08-26 | 2019-10-10 | ブリストル−マイヤーズ スクイブ カンパニーBristol−Myers Squibb Company | Indoleamine 2,3-dioxygenase inhibitors and methods of use thereof |
| US10660909B2 (en) | 2016-11-17 | 2020-05-26 | Syntrix Biosystems Inc. | Method for treating cancer using chemokine antagonists |
| EP3558360A1 (en) | 2016-12-22 | 2019-10-30 | F. Hoffmann-La Roche AG | Treatment of tumors with an anti-csf-1r antibody in combination with an anti-pd-l1 antibody after failure of anti-pd-l1/pd1 treatment |
| US10961239B2 (en) | 2017-01-05 | 2021-03-30 | Bristol-Myers Squibb Company | TGF beta receptor antagonists |
| CA3047600A1 (en) | 2017-01-20 | 2018-07-26 | Arcus Biosciences, Inc. | Azolopyrimidine for the treatment of cancer-related disorders |
| BR112019014527A2 (en) | 2017-01-23 | 2020-02-27 | Revolution Medicines, Inc. | PYRIDINE COMPOUNDS AS ALLOSTIC SHP2 INHIBITORS |
| AU2018210770B2 (en) | 2017-01-23 | 2022-03-24 | Revolution Medicines, Inc. | Bicyclic compounds as allosteric SHP2 inhibitors |
| WO2018183366A1 (en) | 2017-03-28 | 2018-10-04 | Syndax Pharmaceuticals, Inc. | Combination therapies of csf-1r or csf-1 antibodies and a t-cell engaging therapy |
| TWI788340B (en) | 2017-04-07 | 2023-01-01 | 美商必治妥美雅史谷比公司 | Anti-icos agonist antibodies and uses thereof |
| CA3058944A1 (en) | 2017-04-19 | 2018-10-25 | Elstar Therapeutics, Inc. | Multispecific molecules and uses thereof |
| CA3059939A1 (en) | 2017-04-21 | 2018-10-25 | Kyn Therapeutics | Indole ahr inhibitors and uses thereof |
| US11066392B2 (en) | 2017-05-12 | 2021-07-20 | Bristol-Myers Squibb Company | Inhibitors of indoleamine 2,3-dioxygenase and methods of their use |
| CA3063359A1 (en) | 2017-05-12 | 2018-11-15 | Harpoon Therapeutics, Inc. | Mesothelin binding proteins |
| EP3634417B1 (en) | 2017-05-17 | 2023-07-12 | Arcus Biosciences, Inc. | Quinazoline-pyrazole derivatives for the treatment of cancer-related disorders |
| WO2018213665A1 (en) | 2017-05-19 | 2018-11-22 | Syndax Pharmaceuticals, Inc. | Combination therapies |
| EP3644993B1 (en) | 2017-06-30 | 2023-08-02 | Bristol-Myers Squibb Company | Amorphous and crystalline forms of ido inhibitors |
| CN110997691B (en) | 2017-07-28 | 2023-11-07 | 百时美施贵宝公司 | Cyclic dinucleotides as anticancer agents |
| WO2019025811A1 (en) | 2017-08-04 | 2019-02-07 | Bicycletx Limited | Bicyclic peptide ligands specific for cd137 |
| WO2019036657A1 (en) | 2017-08-17 | 2019-02-21 | Kyn Therapeutics | Ahr inhibitors and uses thereof |
| MA49950A (en) | 2017-08-25 | 2020-07-01 | Five Prime Therapeutics Inc | ANTI-B7-H4 ANTIBODIES AND PROCEDURES FOR USE |
| ES2904317T3 (en) | 2017-08-31 | 2022-04-04 | Bristol Myers Squibb Co | Cyclic dinucleotides as anticancer agents |
| WO2019046498A1 (en) | 2017-08-31 | 2019-03-07 | Bristol-Myers Squibb Company | Cyclic dinucleotides as anticancer agents |
| US11667663B2 (en) | 2017-08-31 | 2023-06-06 | Bristol-Myers Squibb Company | Cyclic dinucleotides as anticancer agents |
| SG11202001282UA (en) | 2017-09-07 | 2020-03-30 | Revolution Medicines Inc | Shp2 inhibitor compositions and methods for treating cancer |
| JP2020535119A (en) | 2017-09-13 | 2020-12-03 | ファイヴ プライム セラピューティクス インク | Combination therapy of anti-CSF1R and anti-PD-1 antibody for pancreatic cancer |
| US11358948B2 (en) | 2017-09-22 | 2022-06-14 | Kymera Therapeutics, Inc. | CRBN ligands and uses thereof |
| IL273432B (en) | 2017-09-22 | 2022-09-01 | Kymera Therapeutics Inc | Protein degraders and uses thereof |
| WO2019074824A1 (en) | 2017-10-09 | 2019-04-18 | Bristol-Myers Squibb Company | Inhibitors of indoleamine 2,3-dioxygenase and methods of their use |
| WO2019074822A1 (en) | 2017-10-09 | 2019-04-18 | Bristol-Myers Squibb Company | Inhibitors of indoleamine 2,3-dioxygenase and methods of their use |
| US11230601B2 (en) | 2017-10-10 | 2022-01-25 | Tilos Therapeutics, Inc. | Methods of using anti-lap antibodies |
| WO2019074887A1 (en) | 2017-10-10 | 2019-04-18 | Bristol-Myers Squibb Company | Cyclic dinucleotides as anticancer agents |
| BR112020007058A2 (en) | 2017-10-12 | 2020-10-06 | Revolution Medicines, Inc. | pyridine, pyrazine, and triazine compounds as allosteric shp2 inhibitors |
| IL315737A (en) | 2017-10-13 | 2024-11-01 | Harpoon Therapeutics Inc | B cell maturation antigen binding proteins |
| KR102425983B1 (en) | 2017-10-13 | 2022-07-29 | 하푼 테라퓨틱스, 인크. | Trispecific Proteins and Methods of Use |
| CN111406063B (en) | 2017-10-16 | 2023-09-15 | 百时美施贵宝公司 | Cyclic dinucleotides as anticancer agents |
| EP3704159A1 (en) | 2017-11-01 | 2020-09-09 | Bristol-Myers Squibb Company | Immunostimulatory agonistic antibodies for use in treating cancer |
| EP3707138B1 (en) | 2017-11-06 | 2022-07-13 | Bristol-Myers Squibb Company | Isofuranone compounds useful as hpk1 inhibitors |
| WO2019113464A1 (en) | 2017-12-08 | 2019-06-13 | Elstar Therapeutics, Inc. | Multispecific molecules and uses thereof |
| WO2019118909A1 (en) | 2017-12-15 | 2019-06-20 | Revolution Medicines, Inc. | Polycyclic compounds as allosteric shp2 inhibitors |
| IL315310A (en) | 2017-12-26 | 2024-10-01 | Kymera Therapeutics Inc | IRAK joints and used in them |
| CN111788227B (en) | 2017-12-27 | 2025-02-25 | 百时美施贵宝公司 | Anti-CD40 antibodies and uses thereof |
| WO2019136112A1 (en) | 2018-01-05 | 2019-07-11 | Bristol-Myers Squibb Company | Inhibitors of indoleamine 2,3-dioxygenase and methods of their use |
| EP3737675A4 (en) | 2018-01-12 | 2022-01-05 | Kymera Therapeutics, Inc. | CRBN LIGANDS AND USES THEREOF |
| US12129297B2 (en) | 2018-01-12 | 2024-10-29 | Bristol-Myers Squibb Company | Antibodies against TIM3 and uses thereof |
| WO2019140380A1 (en) | 2018-01-12 | 2019-07-18 | Kymera Therapeutics, Inc. | Protein degraders and uses thereof |
| EP3743095A1 (en) | 2018-01-26 | 2020-12-02 | Celldex Therapeutics, Inc. | Methods of treating cancer with dendritic cell mobilizing agents |
| CN111867581B (en) | 2018-01-29 | 2023-12-26 | 默克专利股份有限公司 | GCN2 inhibitors and their uses |
| SG11202006701TA (en) | 2018-01-29 | 2020-08-28 | Merck Patent Gmbh | Gcn2 inhibitors and uses thereof |
| US10519187B2 (en) | 2018-02-13 | 2019-12-31 | Bristol-Myers Squibb Company | Cyclic dinucleotides as anticancer agents |
| WO2019161054A1 (en) | 2018-02-16 | 2019-08-22 | Arcus Biosciences, Inc. | Dosing with an azolopyrimidine compound |
| SG11202007821WA (en) | 2018-02-21 | 2020-09-29 | Five Prime Therapeutics Inc | B7-h4 antibody formulations |
| WO2019165315A1 (en) | 2018-02-23 | 2019-08-29 | Syntrix Biosystems Inc. | Method for treating cancer using chemokine antagonists alone or in combination |
| MX2020008791A (en) | 2018-02-23 | 2021-01-08 | Bicycletx Ltd | Multimeric bicyclic peptide ligands. |
| JP2021516051A (en) | 2018-03-02 | 2021-07-01 | ファイブ プライム セラピューティクス, インコーポレイテッド | B7-H4 antibody and how to use it |
| MX2020009290A (en) | 2018-03-05 | 2020-09-28 | Arcus Biosciences Inc | Arginase inhibitors. |
| WO2019173587A1 (en) | 2018-03-08 | 2019-09-12 | Bristol-Myers Squibb Company | Cyclic dinucleotides as anticancer agents |
| CA3092589A1 (en) | 2018-03-21 | 2019-09-26 | Five Prime Therapeutics, Inc. | Antibodies binding to vista at acidic ph |
| US11242393B2 (en) | 2018-03-23 | 2022-02-08 | Bristol-Myers Squibb Company | Antibodies against MICA and/or MICB and uses thereof |
| TWI853807B (en) | 2018-03-28 | 2024-09-01 | 美商必治妥美雅史谷比公司 | Interleukin-2/interleukin-2 receptor alpha fusion proteins and methods of use |
| JP2021521182A (en) | 2018-04-12 | 2021-08-26 | ブリストル−マイヤーズ スクイブ カンパニーBristol−Myers Squibb Company | Combination therapy of CD73 antagonist and PD-1 / PD-L1 axis antagonist |
| EP3781162A1 (en) | 2018-04-16 | 2021-02-24 | Arrys Therapeutics, Inc. | Ep4 inhibitors and use thereof |
| US12037323B2 (en) | 2018-05-03 | 2024-07-16 | Bristol-Myers Squibb Company | Uracil derivatives as Mer-AXL inhibitors |
| US11180531B2 (en) | 2018-06-22 | 2021-11-23 | Bicycletx Limited | Bicyclic peptide ligands specific for Nectin-4 |
| WO2020006018A1 (en) | 2018-06-27 | 2020-01-02 | Bristol-Myers Squibb Company | Substituted naphthyridinone compounds useful as t cell activators |
| US11866430B2 (en) | 2018-06-27 | 2024-01-09 | Bristol-Myers Squibb Company | Naphthyridinone compounds useful as T cell activators |
| EP3817822A4 (en) | 2018-07-06 | 2022-07-27 | Kymera Therapeutics, Inc. | PROTEIN DEGRADING AGENTS AND THEIR USES |
| WO2020010177A1 (en) | 2018-07-06 | 2020-01-09 | Kymera Therapeutics, Inc. | Tricyclic crbn ligands and uses thereof |
| SG11202100096XA (en) | 2018-07-09 | 2021-02-25 | Five Prime Therapeutics Inc | Antibodies binding to ilt4 |
| MX2021000213A (en) | 2018-07-11 | 2021-03-25 | Five Prime Therapeutics Inc | Antibodies binding to vista at acidic ph. |
| JP7522720B2 (en) | 2018-07-18 | 2024-07-25 | アーカス バイオサイエンシズ インコーポレイティド | Solid forms of azolopyrimidine compounds |
| US12059420B2 (en) | 2018-07-23 | 2024-08-13 | Bristol-Myers Squibb Company | Inhibitors of indoleamine 2,3-dioxygenase and methods of their use |
| US12145927B2 (en) | 2018-07-23 | 2024-11-19 | Bristol-Myers Squibb Company | Inhibitors of indoleamine 2,3-dioxygenase and methods of their use |
| US11253525B2 (en) | 2018-08-29 | 2022-02-22 | Bristol-Myers Squibb Company | Inhibitors of indoleamine 2,3-dioxygenase and methods of their use |
| US10959986B2 (en) | 2018-08-29 | 2021-03-30 | Bristol-Myers Squibb Company | Inhibitors of indoleamine 2,3-dioxygenase and methods of their use |
| EP3846793B1 (en) | 2018-09-07 | 2024-01-24 | PIC Therapeutics, Inc. | Eif4e inhibitors and uses thereof |
| US12195544B2 (en) | 2018-09-21 | 2025-01-14 | Harpoon Therapeutics, Inc. | EGFR binding proteins and methods of use |
| EP3856771A4 (en) | 2018-09-25 | 2022-06-29 | Harpoon Therapeutics, Inc. | Dll3 binding proteins and methods of use |
| EP3856350A1 (en) | 2018-09-27 | 2021-08-04 | Marengo Therapeutics, Inc. | Csf1r/ccr2 multispecific antibodies |
| TW202035445A (en) | 2018-10-10 | 2020-10-01 | 美商帝洛斯療法股份有限公司 | Anti-lap antibody variants and uses thereof |
| ES3034676T3 (en) | 2018-11-16 | 2025-08-21 | Arcus Biosciences Inc | Inhibitors of arg1 and/or arg2 |
| JP7520003B2 (en) | 2018-11-16 | 2024-07-22 | ブリストル-マイヤーズ スクイブ カンパニー | Anti-NKG2A Antibodies and Uses Thereof |
| CA3119773A1 (en) | 2018-11-30 | 2020-06-04 | Kymera Therapeutics, Inc. | Irak degraders and uses thereof |
| US20220016168A1 (en) | 2018-12-11 | 2022-01-20 | Celldex Therapeutics, Inc. | Methods of using cd27 antibodies as conditioning treatment for adoptive cell therapy |
| GB201820295D0 (en) | 2018-12-13 | 2019-01-30 | Bicyclerd Ltd | Bicyclic peptide ligands specific for MT1-MMP |
| EP3894014A4 (en) * | 2018-12-13 | 2022-04-20 | Development Center for Biotechnology | HUMAN ANTI-CSF-1R ANTIBODIES AND ITS USES |
| GB201820288D0 (en) | 2018-12-13 | 2019-01-30 | Bicycle Tx Ltd | Bicycle peptide ligaands specific for MT1-MMP |
| GB201820325D0 (en) | 2018-12-13 | 2019-01-30 | Bicyclerd Ltd | Bicyclic peptide ligands specific for psma |
| EP3670659A1 (en) | 2018-12-20 | 2020-06-24 | Abivax | Biomarkers, and uses in treatment of viral infections, inflammations, or cancer |
| EP3897849A1 (en) | 2018-12-21 | 2021-10-27 | BicycleTx Limited | Bicyclic peptide ligands specific for pd-l1 |
| US12453738B2 (en) | 2019-03-12 | 2025-10-28 | Arcus Biosciences, Inc. | Treatment of oncogene-driven cancers |
| CN113795264A (en) | 2019-03-19 | 2021-12-14 | 瓦尔希伯伦私人肿瘤研究基金会 | Combination therapy for cancer with Omomyc and antibodies that bind PD-1 or CTLA-4 |
| KR20210144821A (en) | 2019-03-29 | 2021-11-30 | 아르커스 바이오사이언시즈 인코포레이티드 | Cancer treatment method using the identified adenosine fingerprint |
| WO2020201753A1 (en) | 2019-04-02 | 2020-10-08 | Bicycletx Limited | Bicycle toxin conjugates and uses thereof |
| CN120574283A (en) | 2019-04-05 | 2025-09-02 | 凯麦拉医疗公司 | STAT degraders and their uses |
| US12012374B2 (en) | 2019-05-13 | 2024-06-18 | Bristol-Myers Squibb Company | Agonists of ROR GAMMAt |
| US20230295087A1 (en) | 2019-05-13 | 2023-09-21 | Bristol-Myers Squibb Company | AGONISTS OF ROR GAMMAt |
| TW202108559A (en) | 2019-05-31 | 2021-03-01 | 美商醫肯納腫瘤學公司 | Tead inhibitors and uses thereof |
| WO2020251972A1 (en) | 2019-06-10 | 2020-12-17 | Kymera Therapeutics, Inc. | Smarca degraders and uses thereof |
| WO2021024020A1 (en) | 2019-08-06 | 2021-02-11 | Astellas Pharma Inc. | Combination therapy involving antibodies against claudin 18.2 and immune checkpoint inhibitors for treatment of cancer |
| WO2021026179A1 (en) | 2019-08-06 | 2021-02-11 | Bristol-Myers Squibb Company | AGONISTS OF ROR GAMMAt |
| AR119821A1 (en) | 2019-08-28 | 2022-01-12 | Bristol Myers Squibb Co | SUBSTITUTED PYRIDOPYRIMIDINOL COMPOUNDS USEFUL AS T-CELL ACTIVATORS |
| WO2021050964A1 (en) | 2019-09-13 | 2021-03-18 | Nimbus Saturn, Inc. | Hpk1 antagonists and uses thereof |
| BR112022004831A2 (en) | 2019-09-19 | 2022-06-07 | Bristol Myers Squibb Co | Antibodies that bind to seen at acidic pH |
| BR112022009514A2 (en) | 2019-11-19 | 2022-08-16 | Bristol Myers Squibb Co | COMPOUNDS USEFUL AS HELIOS PROTEIN INHIBITORS |
| JP2023502531A (en) | 2019-11-26 | 2023-01-24 | イケナ オンコロジー, インコーポレイテッド | Polymorphic carbazole derivatives and uses thereof |
| CA3159071A1 (en) | 2019-11-26 | 2021-06-03 | Tamar I. ROSENBAUM | Salts/cocrystals of (r)-n-(4-chlorophenyl)-2-((1s,4s)-4-(6-fluoroquinolin-4-yl)cyclohexyl)propanamide |
| JP2023509366A (en) | 2019-12-17 | 2023-03-08 | カイメラ セラピューティクス, インコーポレイテッド | IRAK dissolving agents and their uses |
| EP4076524A4 (en) | 2019-12-17 | 2023-11-29 | Kymera Therapeutics, Inc. | IRAQ DEGRADATION AGENTS AND THEIR USES |
| MX2022007130A (en) | 2019-12-23 | 2022-07-11 | Bristol Myers Squibb Co | SUBSTITUTED PIPERAZINE DERIVATIVES USEFUL AS T CELL ACTIVATORS. |
| AU2020414688A1 (en) | 2019-12-23 | 2022-08-18 | Bristol-Myers Squibb Company | Substituted quinolinonyl piperazine compounds useful as T cell activators |
| CN115297931A (en) | 2019-12-23 | 2022-11-04 | 凯麦拉医疗公司 | SMARCA degrading agents and uses thereof |
| ES3038401T3 (en) | 2019-12-23 | 2025-10-13 | Bristol Myers Squibb Co | Substituted quinazolinyl compounds useful as t cell activators |
| JP7677976B2 (en) | 2019-12-23 | 2025-05-15 | ブリストル-マイヤーズ スクイブ カンパニー | Substituted Heteroaryl Compounds Useful as T Cell Activators - Patent application |
| AR120823A1 (en) | 2019-12-23 | 2022-03-23 | Bristol Myers Squibb Co | SUBSTITUTED BICYCLIC COMPOUNDS USEFUL AS T-CELL ACTIVATORS |
| AU2021206618A1 (en) | 2020-01-06 | 2022-08-18 | Hifibio, Inc. | Anti-TNFR2 antibody and uses thereof |
| BR112022013494A2 (en) | 2020-01-07 | 2022-09-13 | Hifibio Hk Ltd | ANTI-GALECTIN-9 ANTIBODY AND USES THEREOF |
| US20230140384A1 (en) | 2020-03-09 | 2023-05-04 | Bristol-Myers Squibb Company | Antibodies to cd40 with enhanced agonist activity |
| WO2021188769A1 (en) | 2020-03-19 | 2021-09-23 | Arcus Biosciences, Inc. | Tetralin and tetrahydroquinoline compounds as inhibitors of hif-2alpha |
| US12528785B2 (en) | 2020-03-19 | 2026-01-20 | Kymera Therapeutics, Inc. | MDM2 degraders and uses thereof |
| TW202140441A (en) | 2020-03-23 | 2021-11-01 | 美商必治妥美雅史谷比公司 | Substituted oxoisoindoline compounds |
| WO2021207449A1 (en) | 2020-04-09 | 2021-10-14 | Merck Sharp & Dohme Corp. | Affinity matured anti-lap antibodies and uses thereof |
| WO2021231732A1 (en) | 2020-05-15 | 2021-11-18 | Bristol-Myers Squibb Company | Antibodies to garp |
| CR20220611A (en) | 2020-06-02 | 2023-06-07 | Arcus Biosciences Inc | ANTI-TIGIT ANTIBODIES |
| TW202210483A (en) | 2020-06-03 | 2022-03-16 | 美商凱麥拉醫療公司 | Crystalline forms of irak degraders |
| US20230250110A1 (en) | 2020-06-03 | 2023-08-10 | Kymera Therapeutics, Inc. | Deuterated irak degraders and uses thereof |
| CN115942977A (en) | 2020-06-17 | 2023-04-07 | 艾库斯生物科学有限公司 | Crystalline forms of CD73 inhibitors and uses thereof |
| US20250262293A1 (en) | 2020-07-07 | 2025-08-21 | BioNTech SE | Therapeutic rna for hpv-positive cancer |
| EP4188374A4 (en) | 2020-07-30 | 2024-08-14 | Kymera Therapeutics, Inc. | Methods of treating mutant lymphomas |
| CA3189761A1 (en) | 2020-08-03 | 2022-02-10 | Gemma Mudd | Peptide-based linkers |
| MX2023001588A (en) | 2020-08-17 | 2023-05-03 | Bicycletx Ltd | Bicycle conjugates specific for nectin-4 and uses thereof. |
| RU2751249C1 (en) * | 2020-10-28 | 2021-07-12 | Закрытое Акционерное Общество "Биокад" | Monoclonal antibody that specifically binds to csf-1r |
| WO2022120353A1 (en) | 2020-12-02 | 2022-06-09 | Ikena Oncology, Inc. | Tead inhibitors and uses thereof |
| CN117015531A (en) | 2020-12-02 | 2023-11-07 | 医肯纳肿瘤学公司 | TEAD inhibitors and their uses |
| WO2022135666A1 (en) | 2020-12-21 | 2022-06-30 | BioNTech SE | Treatment schedule for cytokine proteins |
| TW202245808A (en) | 2020-12-21 | 2022-12-01 | 德商拜恩迪克公司 | Therapeutic rna for treating cancer |
| WO2022135667A1 (en) | 2020-12-21 | 2022-06-30 | BioNTech SE | Therapeutic rna for treating cancer |
| TW202241891A (en) | 2020-12-30 | 2022-11-01 | 美商凱麥拉醫療公司 | Irak degraders and uses thereof |
| US20250186539A2 (en) | 2021-01-11 | 2025-06-12 | Bicycletx Limited | Methods for treating cancer |
| BR112023015584A2 (en) | 2021-02-02 | 2023-10-24 | Liminal Biosciences Ltd | GPR84 ANTAGONISTS AND USES THEREOF |
| EP4288427A1 (en) | 2021-02-02 | 2023-12-13 | Liminal Biosciences Limited | Gpr84 antagonists and uses thereof |
| US20240109899A1 (en) | 2021-02-04 | 2024-04-04 | Bristol-Myers Squibb Company | Benzofuran compounds as sting agonists |
| CN117120090A (en) | 2021-02-12 | 2023-11-24 | 林伯士萨顿公司 | HPK1 antagonists and uses thereof |
| MX2023009178A (en) | 2021-02-15 | 2023-08-21 | Kymera Therapeutics Inc | Irak4 degraders and uses thereof. |
| US12171768B2 (en) | 2021-02-15 | 2024-12-24 | Kymera Therapeutics, Inc. | IRAK4 degraders and uses thereof |
| WO2022187856A1 (en) | 2021-03-05 | 2022-09-09 | Nimbus Saturn, Inc. | Hpk1 antagonists and uses thereof |
| WO2022197641A1 (en) | 2021-03-15 | 2022-09-22 | Rapt Therapeutics, Inc. | 1h-pyrazolo[3,4-d]pyrimidin-6-yl-amine derivatives as hematopoietic progenitor kinase 1 (hpk1) modulators and/or inhibitors for the treatment of cancer and other diseases |
| EP4313989A4 (en) | 2021-03-29 | 2025-03-05 | Nimbus Saturn, Inc. | HPK1 ANTAGONISTS AND THEIR USES |
| JP2024514530A (en) | 2021-04-02 | 2024-04-02 | ザ リージェンツ オブ ザ ユニバーシティ オブ カリフォルニア | Antibodies against truncated CDCP1 and uses thereof |
| AU2022253450A1 (en) | 2021-04-05 | 2023-11-16 | Bristol-Myers Squibb Company | Pyridinyl substituted oxoisoindoline compounds for the treatment of cancer |
| SMT202500281T1 (en) | 2021-04-06 | 2025-09-12 | Bristol Myers Squibb Co | Pyridinyl substituted oxoisoindoline compounds |
| KR20240007155A (en) | 2021-04-09 | 2024-01-16 | 님버스 클리오, 인코포레이티드 | CBL-B modulators and their uses |
| WO2022221866A1 (en) | 2021-04-16 | 2022-10-20 | Ikena Oncology, Inc. | Mek inhibitors and uses thereof |
| IL308314A (en) | 2021-05-07 | 2024-01-01 | Kymera Therapeutics Inc | Cdk2 degraders and uses thereof |
| JP2024521712A (en) | 2021-05-21 | 2024-06-04 | アーカス バイオサイエンシーズ,インコーポレーテッド | AXL Inhibitor Compounds |
| JP2024521706A (en) | 2021-05-21 | 2024-06-04 | アーカス バイオサイエンシーズ,インコーポレーテッド | AXL Compound |
| CA3225254A1 (en) | 2021-07-13 | 2023-01-19 | BioNTech SE | Multispecific binding agents against cd40 and cd137 in combination therapy for cancer |
| US12234231B2 (en) | 2021-08-25 | 2025-02-25 | PIC Therapeutics, Inc. | EIF4E inhibitors and uses thereof |
| JP2024532276A (en) | 2021-08-25 | 2024-09-05 | ピク セラピューティクス, インコーポレイテッド | eIF4E inhibitors and uses thereof |
| TW202333802A (en) | 2021-10-11 | 2023-09-01 | 德商拜恩迪克公司 | Therapeutic rna for lung cancer |
| IL312348A (en) | 2021-10-29 | 2024-06-01 | Arcus Biosciences Inc | Inhibitors of hif-2alpha and methods of use thereof |
| AR127505A1 (en) | 2021-10-29 | 2024-01-31 | Kymera Therapeutics Inc | IRAQ-4 DEGRADERS AND SYNTHESIS THEREOF |
| WO2023114984A1 (en) | 2021-12-17 | 2023-06-22 | Ikena Oncology, Inc. | Tead inhibitors and uses thereof |
| WO2023147594A2 (en) | 2022-01-31 | 2023-08-03 | Kymera Therapeutics, Inc. | Irak degraders and uses thereof |
| WO2023150186A1 (en) | 2022-02-01 | 2023-08-10 | Arvinas Operations, Inc. | Dgk targeting compounds and uses thereof |
| WO2023173057A1 (en) | 2022-03-10 | 2023-09-14 | Ikena Oncology, Inc. | Mek inhibitors and uses thereof |
| WO2023173053A1 (en) | 2022-03-10 | 2023-09-14 | Ikena Oncology, Inc. | Mek inhibitors and uses thereof |
| WO2023211889A1 (en) | 2022-04-25 | 2023-11-02 | Ikena Oncology, Inc. | Polymorphic compounds and uses thereof |
| US20250179176A1 (en) | 2022-05-02 | 2025-06-05 | Arcus Biosciences, Inc. | Anti-tigit antibodies and uses of the same |
| TW202404581A (en) | 2022-05-25 | 2024-02-01 | 美商醫肯納腫瘤學公司 | Mek inhibitors and uses thereof |
| EP4554680A1 (en) | 2022-07-15 | 2025-05-21 | Arcus Biosciences, Inc. | Inhibitors of hpk1 and methods of use thereof |
| US20260028336A1 (en) | 2022-07-20 | 2026-01-29 | Arcus Biosciences, Inc. | Cbl-b inhibitors and methods of use thereof |
| CN120019047A (en) | 2022-08-02 | 2025-05-16 | 里米诺生物科学有限公司 | Heteroaryl carboxamides and related GPR84 antagonists and their uses |
| KR20250057801A (en) | 2022-08-02 | 2025-04-29 | 리미널 바이오사이언시스 리미티드 | Aryl-triazolyl and related GPR84 antagonists and uses thereof |
| KR20250056924A (en) | 2022-08-02 | 2025-04-28 | 리미널 바이오사이언시스 리미티드 | Substituted pyridone GPR84 antagonists and uses thereof |
| EP4568950A1 (en) | 2022-08-08 | 2025-06-18 | Bristol-Myers Squibb Company | Substituted tetrazolyl compounds useful as t cell activators |
| EP4568963A1 (en) | 2022-08-09 | 2025-06-18 | Bristol-Myers Squibb Company | Tertiary amine substituted bicyclic compounds useful as t cell activators |
| WO2024081385A1 (en) | 2022-10-14 | 2024-04-18 | Arcus Biosciences, Inc. | Hpk1 inhibitors and methods of use thereof |
| EP4604920A1 (en) | 2022-10-20 | 2025-08-27 | Arcus Biosciences, Inc. | Lyophilized formulations of cd73 compounds |
| WO2024112894A1 (en) | 2022-11-22 | 2024-05-30 | PIC Therapeutics, Inc. | Eif4e inhibitors and uses thereof |
| IL321098A (en) | 2022-12-14 | 2025-07-01 | Astellas Pharma Europe Bv | Combination therapy involving bispecific binding agents binding to cldn18.2 and cd3 and immune checkpoint inhibitors |
| WO2024233360A1 (en) | 2023-05-05 | 2024-11-14 | Arcus Biosciences, Inc. | Cbl-b inhibitors and methods of use thereof |
| CN121057732A (en) | 2023-05-08 | 2025-12-02 | 百时美施贵宝公司 | Substituted phenyl oxazolone compounds |
| WO2024243502A1 (en) | 2023-05-25 | 2024-11-28 | Arcus Biosciences, Inc. | Cbl-b inhibitors and methods of use thereof |
| WO2024249540A1 (en) | 2023-05-31 | 2024-12-05 | Bristol-Myers Squibb Company | Substituted oxazolone compound for decreasing levels of ikzf1-4 proteins |
| AU2024285766A1 (en) | 2023-06-07 | 2026-01-08 | Bristol-Myers Squibb Company | Spirocyclic substituted oxoisoindolinyl piperidine-2,6-dione compound |
| WO2024263853A1 (en) | 2023-06-23 | 2024-12-26 | Bristol-Myers Squibb Company | Substituted oxoisoindolinyl piperidine-2,6-dione compound as anticancer agent |
| CN120865251A (en) | 2023-06-23 | 2025-10-31 | 凯麦拉医疗公司 | IRAK degradation agent and application thereof |
| WO2025030002A2 (en) | 2023-08-02 | 2025-02-06 | Arvinas Operations, Inc. | Dgk targeting compounds and uses thereof |
| WO2025064197A1 (en) | 2023-09-02 | 2025-03-27 | Bristol-Myers Squibb Company | Substituted azetidinyl oxoisoindolinyl piperidine-2,6-dione compounds |
| AR133696A1 (en) | 2023-09-02 | 2025-10-22 | Bristol Myers Squibb Co | SUBSTITUTED PHENYL OXOOXAZOLYL PIPERIDINADIONE COMPOUNDS |
| EP4587429A1 (en) | 2023-09-13 | 2025-07-23 | Bristol-Myers Squibb Company | Substituted oxoisoindolinyl piperidine-2,6-dione compounds |
| TW202517260A (en) | 2023-10-06 | 2025-05-01 | 美商阿克思生物科學有限公司 | Cbl-b inhibitors and methods of use thereof |
| WO2025096505A1 (en) | 2023-10-31 | 2025-05-08 | Bristol-Myers Squibb Company | Ubiquitin specific processing protease 1 (usp1) compounds |
| WO2025096489A1 (en) | 2023-10-31 | 2025-05-08 | Bristol-Myers Squibb Company | Ubiquitin specific processing protease 1 (usp1) compounds |
| WO2025096539A1 (en) | 2023-10-31 | 2025-05-08 | Bristol-Myers Squibb Company | Ubiquitin specific processing protease 1 (usp1) compounds |
| WO2025096487A1 (en) | 2023-10-31 | 2025-05-08 | Bristol-Myers Squibb Company | Ubiquitin specific processing protease 1 (usp1) compounds |
| WO2025096488A1 (en) | 2023-10-31 | 2025-05-08 | Bristol-Myers Squibb Company | Ubiquitin specific processing protease 1 (usp1) compounds |
| WO2025096494A1 (en) | 2023-10-31 | 2025-05-08 | Bristol-Myers Squibb Company | Ubiquitin specific processing protease 1 (usp1) compounds |
| WO2025120867A1 (en) | 2023-12-08 | 2025-06-12 | Astellas Pharma Inc. | Combination therapy involving bispecific binding agents binding to cldn18.2 and cd3 and anti-vegfr2 antibodies |
| TW202541837A (en) | 2023-12-08 | 2025-11-01 | 日商安斯泰來製藥公司 | Combination therapy involving bispecific binding agents binding to cldn18.2 and cd3 and agents stabilizing or increasing expression of cldn18.2 |
| WO2025120866A1 (en) | 2023-12-08 | 2025-06-12 | Astellas Pharma Inc. | Combination therapy involving bispecific binding agents binding to cldn18.2 and cd3 and agents stabilizing or increasing expression of cldn18.2 |
| WO2025226767A1 (en) | 2024-04-24 | 2025-10-30 | Bristol-Myers Squibb Company | Substituted 3-(5-(6-aminopyridin-2-yl)-4-fluoro-1-oxoisoindolin-2-yl)piperidine-2,6-dione compounds for use in the treatment of cancer |
| WO2026006759A1 (en) | 2024-06-28 | 2026-01-02 | Arcus Biosciences, Inc. | Pharmaceutical compositions, dosage forms, and methods of making and using same |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009026303A1 (en) * | 2007-08-21 | 2009-02-26 | Amgen Inc. | Human c-fms antigen binding proteins |
| WO2009112245A1 (en) * | 2008-03-14 | 2009-09-17 | Transgene S.A. | Antibody against the csf-1 r |
Family Cites Families (42)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1988007089A1 (en) | 1987-03-18 | 1988-09-22 | Medical Research Council | Altered antibodies |
| US5202238A (en) | 1987-10-27 | 1993-04-13 | Oncogen | Production of chimeric antibodies by homologous recombination |
| US5204244A (en) | 1987-10-27 | 1993-04-20 | Oncogen | Production of chimeric antibodies by homologous recombination |
| US5770701A (en) | 1987-10-30 | 1998-06-23 | American Cyanamid Company | Process for preparing targeted forms of methyltrithio antitumor agents |
| US5606040A (en) | 1987-10-30 | 1997-02-25 | American Cyanamid Company | Antitumor and antibacterial substituted disulfide derivatives prepared from compounds possessing a methyl-trithio group |
| US5208020A (en) | 1989-10-25 | 1993-05-04 | Immunogen Inc. | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| CA2026147C (en) | 1989-10-25 | 2006-02-07 | Ravi J. Chari | Cytotoxic agents comprising maytansinoids and their therapeutic use |
| WO1993025687A1 (en) | 1992-06-09 | 1993-12-23 | Chiron Corporation | Crystallization of m-csf |
| ATE139900T1 (en) | 1992-11-13 | 1996-07-15 | Idec Pharma Corp | THERAPEUTIC USE OF CHIMERIC AND LABELED ANTIBODIES AGAINST HUMAN B LYMPHOCYTE RESTRICTED DIFFERENTIATION ANTIGEN FOR THE TREATMENT OF B CELL LYMPHOMA |
| US5635483A (en) | 1992-12-03 | 1997-06-03 | Arizona Board Of Regents Acting On Behalf Of Arizona State University | Tumor inhibiting tetrapeptide bearing modified phenethyl amides |
| US5780588A (en) | 1993-01-26 | 1998-07-14 | Arizona Board Of Regents | Elucidation and synthesis of selected pentapeptides |
| US5773001A (en) | 1994-06-03 | 1998-06-30 | American Cyanamid Company | Conjugates of methyltrithio antitumor agents and intermediates for their synthesis |
| US5714586A (en) | 1995-06-07 | 1998-02-03 | American Cyanamid Company | Methods for the preparation of monomeric calicheamicin derivative/carrier conjugates |
| US5712374A (en) | 1995-06-07 | 1998-01-27 | American Cyanamid Company | Method for the preparation of substantiallly monomeric calicheamicin derivative/carrier conjugates |
| JPH0967400A (en) | 1995-08-31 | 1997-03-11 | Toray Ind Inc | Monoclonal antibody, hybridoma producing the same antibody and its utilization |
| ATE319745T1 (en) | 1997-05-21 | 2006-03-15 | Biovation Ltd | METHOD FOR PRODUCING NON-IMMUNOGENIC PROTEINS |
| CA2304354A1 (en) | 1997-10-02 | 1999-04-15 | Max-Planck-Gesellschaft Zur Forderung Der Wissenschaften E.V. | Methods for the modulation of neovascularization and/or the growth of collateral arteries and/or other arteries from preexisting arteriolar connections |
| EP1223980B1 (en) | 1999-10-28 | 2003-05-21 | Hofbauer, Reinhold | Use of csf-1 inhibitors |
| DK1242438T3 (en) | 1999-12-29 | 2007-02-12 | Immunogen Inc | Cytotoxic agents comprising modified doxorubicins and daunorubicins and their therapeutic use |
| US7108852B2 (en) | 2000-03-20 | 2006-09-19 | Warner-Lambert Company Llc | Methods of treating inflammation using antibodies to M-CSF |
| AU2003291002A1 (en) | 2002-11-15 | 2004-06-15 | Chiron Corporation | Methods for preventing and treating cancer metastasis and bone loss associated with cancer metastasis |
| GB0325836D0 (en) | 2003-11-05 | 2003-12-10 | Celltech R&D Ltd | Biological products |
| PT2489364E (en) | 2003-11-06 | 2015-04-16 | Seattle Genetics Inc | Monomethylvaline compounds conjugated to antibodies |
| AU2005267062C1 (en) | 2004-07-22 | 2013-01-17 | Five Prime Therapeutics, Inc. | Compositions and methods of use for MGD-CSF in disease treatment |
| AR054428A1 (en) | 2005-03-08 | 2007-06-27 | Pharmacia & Upjohn Co Llc | COMPOSITIONS OF ANTIBODIES ANTI FACTOR OF COLOGNE OF MACROFAGOS (ANTI-M-CSF) THAT HAVE LOWER LEVELS OF ENDOTOXIN |
| WO2007075326A2 (en) | 2005-12-09 | 2007-07-05 | Seattle Genetics, Inc. | Methods of using cd40 binding agents |
| EP1984024A2 (en) | 2006-01-05 | 2008-10-29 | Novartis AG | Methods for preventing and treating cancer metastasis and bone loss associated with cancer metastasis |
| TW200922234A (en) | 2006-10-11 | 2009-05-16 | Interdigital Tech Corp | Increasing a secret bit generation rate in wireless communication |
| US8470977B2 (en) | 2008-03-14 | 2013-06-25 | Transgene S.A. | Antibody against the CSF-1R |
| CN102046660A (en) | 2008-03-26 | 2011-05-04 | 塞勒兰特治疗公司 | Immunoglobulin and/or toll-like receptor proteins associated with myelogenous haematological proliferative disorders and uses thereof |
| HUE028537T2 (en) | 2009-04-20 | 2016-12-28 | Kyowa Hakko Kirin Co Ltd | Antibody containing igg2 having amino acid mutation introduced therein |
| HUE044179T2 (en) | 2009-12-10 | 2019-10-28 | Hoffmann La Roche | Antibodies binding preferentially human csf1r extracellular domain 4 and their use |
| US9169323B2 (en) | 2010-03-05 | 2015-10-27 | Hoffmann-La Roche Inc. | Antibodies against human CSF-1R |
| EP2542588A1 (en) | 2010-03-05 | 2013-01-09 | F. Hoffmann-La Roche AG | Antibodies against human csf-1r and uses thereof |
| TWI426920B (en) | 2010-03-26 | 2014-02-21 | Hoffmann La Roche | Bispecific, bivalent anti-VEGF/anti-ANG-2 antibody |
| AR080698A1 (en) | 2010-04-01 | 2012-05-02 | Imclone Llc | ANTIBODY OR FRAGMENT OF THE SAME THAT SPECIFICALLY LINKS THE VARIOUS OF HUMAN CSF -1R, PHARMACEUTICAL COMPOSITION THAT INCLUDES IT, ITS USE FOR THE MANUFACTURE OF A USEFUL MEDICINAL PRODUCT FOR THE TREATMENT OF CANCER AND METHOD TO DETERMINE IF A BASED SUBJECT MATTER AND |
| NZ603193A (en) | 2010-05-04 | 2014-07-25 | Five Prime Therapeutics Inc | Antibodies that bind csf1r |
| JP5798404B2 (en) | 2010-08-31 | 2015-10-21 | 日東電工株式会社 | Adhesive tape for electrode plate protection |
| BR112014012624A2 (en) | 2011-12-15 | 2018-10-09 | F Hoffmann-La Roche Ag | antibodies, pharmaceutical composition, nucleic acid, expression vectors, host cell, method for producing a recombinant antibody and use of the antibody |
| AR090263A1 (en) | 2012-03-08 | 2014-10-29 | Hoffmann La Roche | COMBINED ANTIBODY THERAPY AGAINST HUMAN CSF-1R AND USES OF THE SAME |
| AR095882A1 (en) | 2013-04-22 | 2015-11-18 | Hoffmann La Roche | ANTIBODY COMBINATION THERAPY AGAINST HUMAN CSF-1R WITH A TLR9 AGONIST |
| AR097584A1 (en) | 2013-09-12 | 2016-03-23 | Hoffmann La Roche | ANTIBODY COMBINATION THERAPY AGAINST HUMAN CSF-1R AND ANTIBODIES AGAINST HUMAN PD-L1 |
-
2011
- 2011-03-03 EP EP11723276A patent/EP2542588A1/en not_active Ceased
- 2011-03-03 BR BR112012022046A patent/BR112012022046A2/en not_active Application Discontinuation
- 2011-03-03 US US13/582,989 patent/US9221910B2/en not_active Expired - Fee Related
- 2011-03-03 CN CN201180012506.9A patent/CN102918061B/en not_active Expired - Fee Related
- 2011-03-03 MX MX2012010019A patent/MX336682B/en unknown
- 2011-03-03 KR KR1020127025956A patent/KR101647871B1/en not_active Expired - Fee Related
- 2011-03-03 WO PCT/EP2011/053213 patent/WO2011131407A1/en not_active Ceased
- 2011-03-03 CA CA2789071A patent/CA2789071C/en not_active Expired - Fee Related
- 2011-03-03 JP JP2012555429A patent/JP5989547B2/en not_active Expired - Fee Related
- 2011-03-03 RU RU2012142230A patent/RU2617966C2/en not_active IP Right Cessation
-
2015
- 2015-10-29 US US14/927,373 patent/US9624302B2/en not_active Expired - Fee Related
-
2017
- 2017-03-03 US US15/448,986 patent/US20170275368A1/en not_active Abandoned
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2009026303A1 (en) * | 2007-08-21 | 2009-02-26 | Amgen Inc. | Human c-fms antigen binding proteins |
| WO2009112245A1 (en) * | 2008-03-14 | 2009-09-17 | Transgene S.A. | Antibody against the csf-1 r |
Cited By (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105658236A (en) * | 2013-08-30 | 2016-06-08 | Ucb生物制药私人有限公司 | Antibodies |
| CN105658236B (en) * | 2013-08-30 | 2021-03-19 | Ucb生物制药私人有限公司 | Antibodies |
| CN105873949A (en) * | 2014-01-31 | 2016-08-17 | 勃林格殷格翰国际有限公司 | Novel anti-BAFF antibodies |
Also Published As
| Publication number | Publication date |
|---|---|
| US20170275368A1 (en) | 2017-09-28 |
| MX336682B (en) | 2016-01-27 |
| JP2013520978A (en) | 2013-06-10 |
| CA2789071A1 (en) | 2011-10-27 |
| MX2012010019A (en) | 2012-11-29 |
| RU2617966C2 (en) | 2017-04-28 |
| KR101647871B1 (en) | 2016-08-11 |
| JP5989547B2 (en) | 2016-09-07 |
| CN102918061B (en) | 2016-06-08 |
| US20130005949A1 (en) | 2013-01-03 |
| US20160053015A1 (en) | 2016-02-25 |
| US9221910B2 (en) | 2015-12-29 |
| CA2789071C (en) | 2018-03-27 |
| EP2542588A1 (en) | 2013-01-09 |
| BR112012022046A2 (en) | 2017-02-14 |
| US9624302B2 (en) | 2017-04-18 |
| KR20120134138A (en) | 2012-12-11 |
| WO2011131407A1 (en) | 2011-10-27 |
| RU2012142230A (en) | 2014-04-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102918061B (en) | Antibodies against human CSF-1R and uses thereof | |
| CN102918060B (en) | Anti-human CSF-1R antibody and use thereof | |
| US10336830B2 (en) | Antibodies against human CSF-1R and uses thereof | |
| HK1176947B (en) | Antibodies against human csf-1r and uses thereof | |
| HK1176947A (en) | Antibodies against human csf-1r and uses thereof | |
| HK1176948B (en) | Antibodies against human csf-1r and uses thereof | |
| HK1201282B (en) | Antibodies against human csf-1r and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: DE Ref document number: 1176947 Country of ref document: HK |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| REG | Reference to a national code |
Ref country code: HK Ref legal event code: GR Ref document number: 1176947 Country of ref document: HK |
|
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20160608 Termination date: 20190303 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |