WO2025096494A1 - Ubiquitin specific processing protease 1 (usp1) compounds - Google Patents
Ubiquitin specific processing protease 1 (usp1) compounds Download PDFInfo
- Publication number
- WO2025096494A1 WO2025096494A1 PCT/US2024/053499 US2024053499W WO2025096494A1 WO 2025096494 A1 WO2025096494 A1 WO 2025096494A1 US 2024053499 W US2024053499 W US 2024053499W WO 2025096494 A1 WO2025096494 A1 WO 2025096494A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- mmol
- cancer
- alkyl
- imidazol
- trifluoromethyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/02—Antineoplastic agents specific for leukemia
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
Definitions
- This invention relates to compounds which are inhibitors of ubiquitin-specific- processing protease 1 (USP1) useful for treating diseases including, among others, cancer, autoimmune and inflammatory disorders.
- USP1 ubiquitin-specific- processing protease 1
- the invention further pertains to pharmaceutical compositions containing at least one compound according to the invention that are useful for the treatment of conditions related to the inhibition of USP1 in a mammal.
- Ubiquitination is important in the regulation of many cellular functions and cellular homeostasis.
- the conjugation of ubiquitin to a target protein is a multistep process involving the sequential action of a ubiquitin activating enzyme (El), a ubiquitin- conjugating enzyme (E2), and a ubiquitin protein-ligase (E3).
- El ubiquitin activating enzyme
- E2 ubiquitin- conjugating enzyme
- E3 ubiquitin protein-ligase
- the ubiquitin tags can mediate non-covalent interactions of the ubiquitinated substrate with other proteins bearing different types of ubiquitin-binding motifs.
- a family of enzymes, termed deubiquitinases act on ubiquitinated substrates to catalyze the removal of ubiquitin moieties.
- USP1 ubiquitin-specific protease 1
- USP1 is a regulator of several important steps in the DNA damage response, particularly in the Fanconi anemia pathway, and in the process of translesion synthesis. USP1 has also been reported to contribute to the repair of double-strand DNA breaks through homologous recombination. In addition, USP1 has been reported to deubiquitinate and stabilize members of the family of inhibitors of DNA binding (ID) proteins, ID1, ID2 and ID3.
- ID DNA binding
- the present disclosure provides compounds that modulate the expression or activity of USP1.
- the disclosure also provides compositions, including pharmaceutical compositions, kits that include the compounds, and methods of using (or administering) and making the compounds.
- the compounds provided herein are useful in treating diseases, disorders, or conditions that are mediated by USP1.
- the disclosure also provides compounds for use in therapy.
- the disclosure further provides compounds for use in a method of treating a disease, disorder, or condition that is mediated by USP1.
- the disclosure provides uses of the compounds in the manufacture of a medicament for the treatment of a disease, disorder or condition that is mediated by (or mediated, at least in part, by) USP1.
- G 1 is selected from -Ce aryl-, 5-6 membered heteroaryl, C3-8 cycloalkyl and 5-6 membered heterocyclyl; wherein each Ce aryl, 5-6 membered heteroaryl, C3-8 cycloalkyl and 5-6 membered heterocyclyl is optionally substituted with one to four R 100 ;
- compositions comprising a pharmaceutically acceptable carrier and at least one of the compounds disclosed herein.
- the present application also provides methods for the inhibition of USP1 comprising administering a therapeutically effective amount of at least one of Formula I.
- the present application also provides a method for treating proliferative, metabolic, allergic, autoimmune and inflammatory diseases, comprising administering to a host in need of such treatment a therapeutically effective amount of at least one of the compounds disclosed herein.
- the compounds of Formula I or a pharmaceutically acceptable salt thereof may be used to treat cancers that are mediated by, dependent on or associated with USP1 activity.
- the disease is a solid tumor.
- R 1 is selected from Ce aryl and 5-6 membered heteroaryl, optionally substituted with one to four halo, hydroxy, amino, -C(O)R a , -C(O)OR b , -C(O)NR a R b , -N(R a )C(O)R b , -S(O)NR a R b , -S(O) 2 NR a R b , -S(O)R g , -S(O) 2 R g , -NR a R b , -OR a , -SR b , Ci-6 alkyl, C 2 -6 alkenyl, C2-6 alkynyl and C3-8 cycloalkyl; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl and C3-8 cycloalkyl is optionally substituted with one to four R 100 ;
- R 2 is selected from absent, hydrogen, halo, hydroxy, amino, -CN, -C(O)R a , -C(O)OR b , - C(O)NR a R b , -N(R a )C(O)R b , -N(R a )C(O)NR a R b , -N(R a )SO 2 NR a R b , -S(O)NR a R b , - S(O) 2 NR a R b , -N(R a )S(O) 2 R b , -S(O)R g , -S(O) 2 R g , -NR a R b , -OR a , -SR b , -OC(O)R a , - OC(O)NR a R b C1-6 alkyl, C2-6 alkenyl, C2-6
- Y is absent or -O-
- Each W 1 , W 2 , W 3 and W 4 is independently selected from -N(R a )-, -C(O)- and -C(R a )-;
- W 5 is selected from -N- and -C(R a )-; wherein at least one of W 1 , W 2 , W 3 and W 4 is -C(R a )- or -C(O)-;
- G 1 is selected from -Ce aryl-, 5-6 membered heteroaryl, C3-8 cycloalkyl and 5-6 membered heterocyclyl; wherein each Ce aryl, 5-6 membered heteroaryl, C3-8 cycloalkyl and 5-6 membered heterocyclyl is optionally substituted with one to four R 100 ;
- G 2 is a 5 or 6 membered heteroaryl or 5-6 membered heterocyclyl optionally substituted with one to four R 100 ; each R a and R b is independently selected from absent, hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-6 cycloalkyl and 4-6 membered heterocyclyl; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-6 cycloalkyl and 4-6 membered heterocyclyl is optionally substituted with one to four R 200 ; each R 100 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)R C , -C(O)OR C , -C(O)NR c R d , -N(R c )C(O)R d , -S(O)NR c R d , -
- Formula (Illa) Formula (Illb) or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof.
- Formula (Via) Formula (VIb) or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof; wherein R 5 is Ci-6 alkyl.
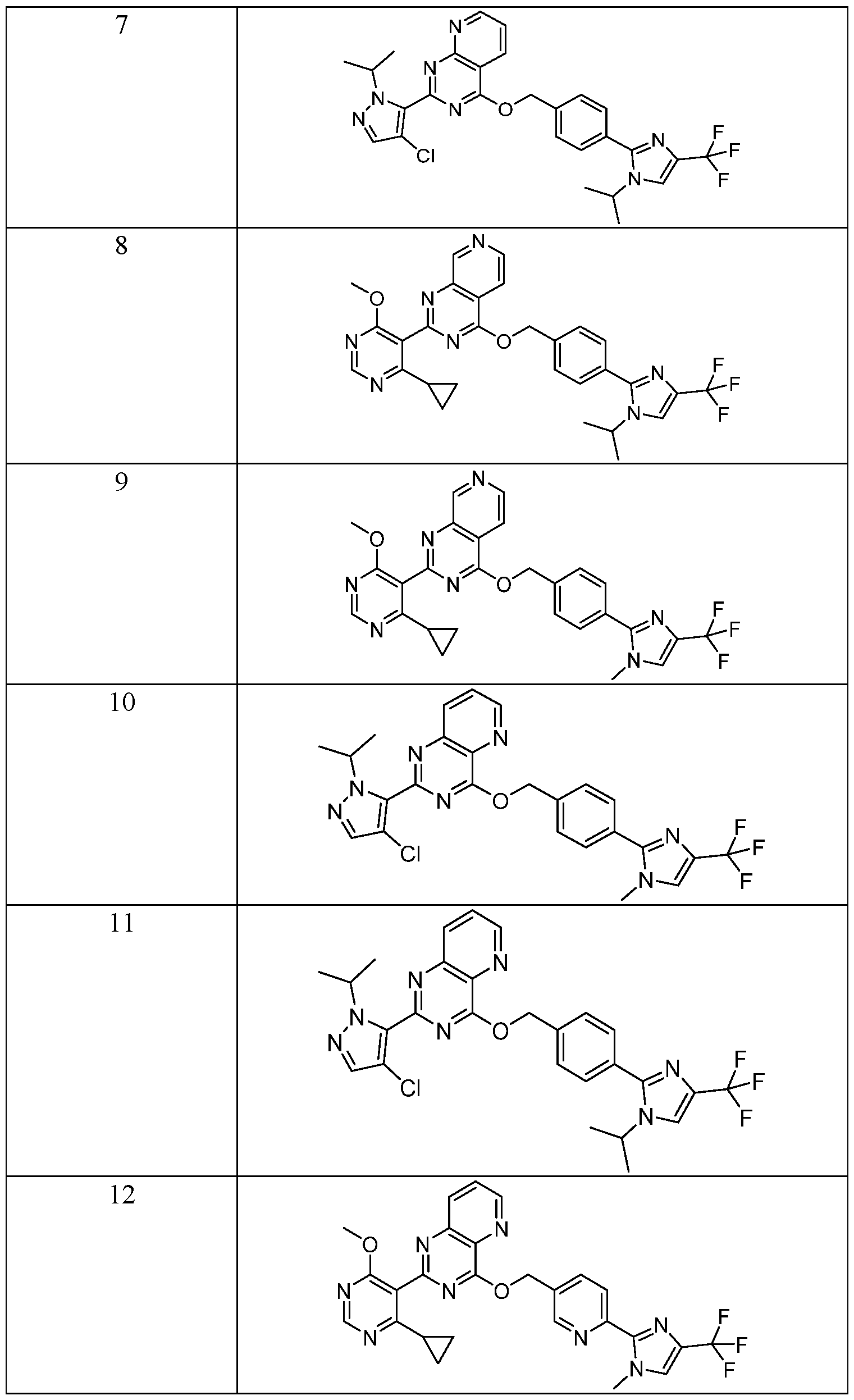
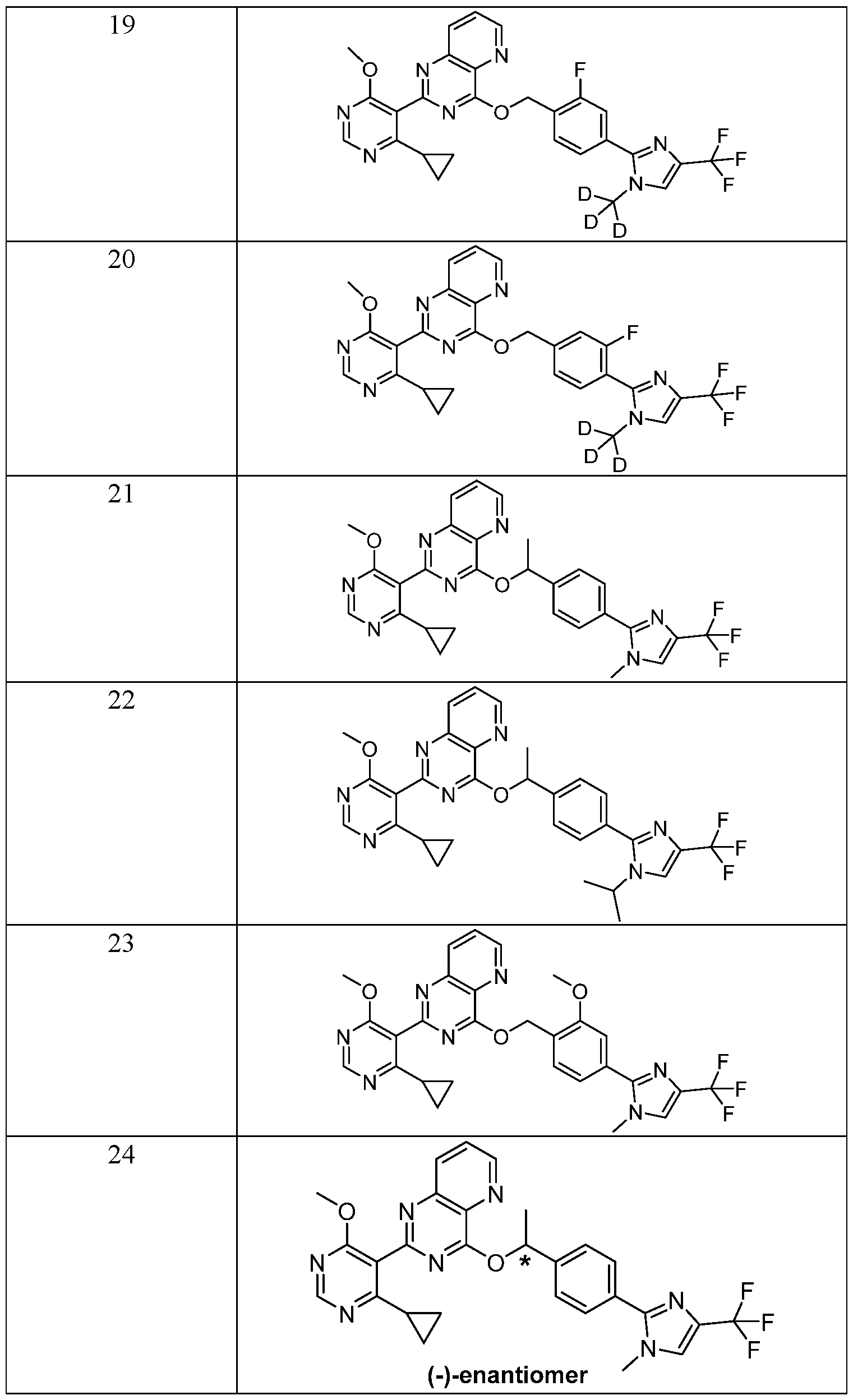
- R 1 is selected from: or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof.
- R 2 is selected from:
- G 2 is selected from: or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers, or deuterated analog thereof.
- the compounds described herein may be used to treat cancers that are mediated by, dependent on or associated with USP1 activity.
- the disease is a solid tumor.
- the solid tumor is selected from prostate cancer, pancreatic cancer, bladder cancer, colorectal cancer, breast cancer, renal cancer, hepatocellular cancer, lung cancer, ovarian cancer, cervical cancer, gastric cancer, esophageal cancer, head and neck cancer, melanoma, neuroendocrine cancers, CNS cancers, brain tumors (e.g., glioma, anaplastic oligodendroglioma, adult glioblastoma multiforme, and adult anaplastic astrocytoma), bone cancer, or soft tissue sarcoma.
- the solid tumor is from non-small cell lung cancer or small-cell lung cancer.
- nitrogen atoms e.g., amines
- these can be converted to N-oxides by treatment with an oxidizing agent (e.g., MCPBA and/or hydrogen peroxides) to afford other compounds of this invention.
- an oxidizing agent e.g., MCPBA and/or hydrogen peroxides
- a dash that is not between two letters or symbols is used to indicate a point of attachment for a substituent. For example, -CONH2 is attached through the carbon atom.
- a dash at the front or end of a chemical group is a matter of convenience; chemical groups may be depicted with or without one or more dashes without losing their ordinary meaning.
- a wavy line drawn through a line in a structure indicates a point of attachment of a group. Unless chemically or structurally required, no directionality is indicated or implied by the order in which a chemical group is written or named.
- optionally substituted in reference to a particular moiety of the compound of Formula I (e.g., an optionally substituted heteroaryl group) refers to a moiety having 0, 1, 2, or more substituents.
- optionally substituted alkyl encompasses both “alkyl” and “substituted alkyl” as defined below. It will be understood by those skilled in the art, with respect to any group containing one or more substituents, that such groups are not intended to introduce any substitution or substitution patterns that are sterically impractical, synthetically non-feasible and/or inherently unstable.
- Cu-v indicates that the following group has from u to v carbon atoms.
- Ci-6 alkyl indicates that the alkyl group has from 1 to 6 carbon atoms.
- alkyl or “alkylene” is intended to include both branched and straight-chain saturated aliphatic hydrocarbon groups having the specified number of carbon atoms.
- “Ci-io alkyl” (or alkylene), is intended to include Ci, C2, C3, C4, Cs, Ce, C7, Cs, C9, and C10 alkyl groups.
- “Ci-Ce alkyl” denotes alkyl having 1 to 6 carbon atoms.
- Alkyl groups can be unsubstituted or substituted so that one or more of its hydrogens are replaced by another chemical group.
- Example alkyl groups include, but are not limited to, methyl (Me), ethyl (Et), propyl (e.g., n-propyl and isopropyl), butyl (e.g., n-butyl, isobutyl, /-butyl), pentyl (e.g., n-pentyl, isopentyl, neopentyl), and the like.
- alkenyl or “alkenylene” is intended to include hydrocarbon chains of either straight or branched configuration and having one or more double carbon-carbon bonds that may occur in any stable point along the chain.
- C2-6 alkenyl (or alkenylene) is intended to include C2, C3, C4, Cs, and Ce alkenyl groups.
- alkenyl examples include, but are not limited to, ethenyl, 1 -propenyl, 2-propenyl, 2-butenyl, 3-butenyl, 2-pentenyl, 3- pentenyl, 4-pentenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-hexenyl, 2-methyl-2-propenyl, 4- methyl-3 -pentenyl, and the like.
- Alkynyl or “alkynylene” is intended to include hydrocarbon chains of either straight or branched configuration and having one or more triple carbon-carbon bonds that may occur in any stable point along the chain.
- C2-6 alkynyl (or alkynylene), is intended to include C2, C3, C4, Cs, and Ce alkynyl groups; such as ethynyl, propynyl, butynyl, pentynyl, hexynyl and the like.
- alkyl When the term “alkyl” is used together with another group, such as in “arylalkyl”, this conjunction defines with more specificity at least one of the substituents that the substituted alkyl will contain.
- arylalkyl refers to a substituted alkyl group as defined above where at least one of the substituents is an aryl, such as benzyl.
- aryl(Co-4)alkyl includes a substituted lower alkyl having at least one aryl substituent and also includes an aryl directly bonded to another group, z.e., aryl(Co)alkyl.
- heteroarylalkyl refers to a substituted alkyl group as defined above where at least one of the substituents is a heteroaryl.
- substituted alkenyl, alkynyl, alkylene, alkenylene, or alkynylene group these groups are substituted with one to three substituents as defined above for substituted alkyl groups.
- alkoxy refers to an oxygen atom substituted by alkyl or substituted alkyl, as defined herein.
- alkoxy includes the group -O-Ci-ealkyl such as methoxy, ethoxy, propoxy, isopropoxy, n-butoxy, sec-butoxy, te/7-butoxy, pentoxy, 2-pentyloxy, isopentoxy, neopentoxy, hexoxy, 2-hexoxy, 3-hexoxy, 3- methylpentoxy, and the like.
- “Lower alkoxy” refers to alkoxy groups having one to four carbons.
- cycloalkyl refers to cyclized alkyl groups, including mono-, bi- or polycyclic ring systems.
- C3-7 cycloalkyl is intended to include C3, C4, Cs, Ce, and C7 cycloalkyl groups.
- Example cycloalkyl groups include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, norbornyl, and the like.
- carrier or “carbocyclic residue” is intended to mean any stable 3-, 4-, 5-, 6-, or 7-membered monocyclic or bicyclic or 7-, 8-, 9-, 10-, 11-, 12-, or 13-membered bicyclic or tricyclic ring, any of which may be saturated, partially unsaturated, unsaturated or aromatic.
- carbocycles include, but are not limited to, cyclopropyl, cyclobutyl, cyclobutenyl, cyclopentyl, cyclopentenyl, cyclohexyl, cycloheptenyl, cycloheptyl, cycloheptenyl, adamantyl, cyclooctyl, cyclooctenyl, cyclooctadienyl, [3.3.0]bicyclooctane, [4.3.0]bicyclononane, [4.4.0]bicyclodecane, [2.2.2]bicyclooctane, fluorenyl, phenyl, naphthyl, indanyl, adamantyl, anthracenyl, and tetrahydronaphthyl (tetralin).
- bridged rings are also included in the definition of carbocycle (e.g., [2.2.2]bicyclooctane).
- carbocycles e.g., [2.2.2]bicyclooctane
- Preferred carbocycles are cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, and phenyl.
- carbocycle When the term “carbocycle” is used, it is intended to include “aryl”.
- a bridged ring occurs when one or more carbon atoms link two non-adjacent carbon atoms.
- Preferred bridges are one or two carbon atoms. It is noted that a bridge always converts a monocyclic ring into a bicyclic ring. When a ring is bridged, the substituents recited for the ring may also be present on the bridge.
- aryl refers to monocyclic or bicyclic aromatic hydrocarbon groups having 6 to 12 carbon atoms in the ring portion, such as phenyl, and naphthyl groups, each of which may be substituted.
- cycloalkyl includes cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, bicyclooctyl, etc., as well as the following ring systems: and the like, which optionally may be substituted at any available atoms of the ring(s).
- halo or halogen refers to chloro, bromo, fluoro and iodo.
- haloalkyl means a substituted alkyl having one or more halo substituents.
- haloalkyl includes mono, bi, and trifluoromethyl.
- haloalkoxy means an alkoxy group having one or more halo substituents.
- haloalkoxy includes OCF3.
- heterocycle refers to substituted and unsubstituted 3- to 7-membered monocyclic groups, 7- to 11 -membered bicyclic groups, and 10- to 15- membered tricyclic groups, in which at least one of the rings has at least one heteroatom (O, S or N), said heteroatom containing ring preferably having 1, 2, or 3 heteroatoms selected from O, S, and N.
- Each ring of such a group containing a heteroatom can contain one or two oxygen or sulfur atoms and/or from one to four nitrogen atoms provided that the total number of heteroatoms in each ring is four or less, and further provided that the ring contains at least one carbon atom.
- the nitrogen and sulfur atoms may optionally be oxidized and the nitrogen atoms may optionally be quaternized.
- the fused rings completing the bicyclic and tricyclic groups may contain only carbon atoms and may be saturated, partially saturated, or fully unsaturated.
- the heterocyclo group may be attached at any available nitrogen or carbon atom.
- the terms "heterocycle”, “heterocycloalkyl”, “heterocyclo”, “heterocyclic”, and “heterocyclyl” include “heteroaryl” groups, as defined below.
- exemplary monocyclic heterocyclyl groups include azetidinyl, pyrrolidinyl, oxetanyl, imidazolinyl, oxazolidinyl, isoxazolinyl, thiazolidinyl, isothiazolidinyl, tetrahydrofuranyl, piperidyl, piperazinyl, 2- oxopiperazinyl, 2-oxopiperidyl, 2-oxopyrrolodinyl, 2-oxoazepinyl, azepinyl, 1-pyridonyl, 4-piperidonyl, tetrahydropyranyl, morpholinyl, thiamorpholinyl, thiamorpholinyl sulfoxide, thiamorpholinyl sulfone, 1,3-dioxolane and tetrahydro- 1,1 -di oxothienyl and
- heteroaryl refers to substituted and unsubstituted aromatic 5- or 6- membered monocyclic groups, 9- or 10-membered bicyclic groups, and 11- to 14- membered tricyclic groups which have at least one heteroatom (O, S or N) in at least one of the rings, said heteroatom-containing ring preferably having 1, 2, or 3 heteroatoms selected from O, S, and N.
- Each ring of the heteroaryl group containing a heteroatom can contain one or two oxygen or sulfur atoms and/or from one to four nitrogen atoms provided that the total number of heteroatoms in each ring is four or less and each ring has at least one carbon atom.
- the fused rings completing the bicyclic and tricyclic groups may contain only carbon atoms and may be saturated, partially saturated, or unsaturated.
- the nitrogen and sulfur atoms may optionally be oxidized and the nitrogen atoms may optionally be quaternized.
- Heteroaryl groups which are bicyclic or tricyclic must include at least one fully aromatic ring but the other fused ring or rings may be aromatic or nonaromatic.
- Exemplary monocyclic heteroaryl groups include pyrrolyl, pyrazolyl, pyrazolinyl, imidazolyl, oxazolyl, isoxazolyl, thiazolyl, thiadiazolyl, isothiazolyl, furanyl, thienyl, oxadiazolyl, pyridyl, pyrazinyl, pyrimidinyl, pyridazinyl, triazinyl and the like.
- Exemplary bicyclic heteroaryl groups include indolyl, benzothiazolyl, benzodi oxolyl, benzoxazolyl, benzothienyl, quinolinyl, tetrahydroisoquinolinyl, isoquinolinyl, benzimidazolyl, benzopyranyl, indolizinyl, benzofuranyl, chromonyl, coumarinyl, benzopyranyl, cinnolinyl, quinoxalinyl, indazolyl, pyrrol opyridyl, furopyridyl, dihydroisoindolyl, tetrahydroquinolinyl and the like.
- Exemplary tricyclic heteroaryl groups include carbazolyl, benzindolyl, phenanthrollinyl, acridinyl, phenanthridinyl, xanthenyl and the like.
- preferred heteroaryl groups include: be substituted at any available carbon or nitrogen atom.
- aryl e.g., phenyl
- cycloalkyl e.g., cyclohexyl
- heterocyclo e.g., pyrrolidinyl, piperidinyl, and morpholinyl
- heteroaryl e.g, tetrazolyl, imidazolyl, pyrazolyl, triazolyl, thiazolyl, and furyl
- the reference is intended to include rings having 0 to 3, preferably 0 to 2, substituents selected from those recited above for the aryl, cycloalkyl, heterocyclo and/or heteroaryl groups, as appropriate.
- Carbocyclyl or “carbocyclic” refers to a saturated or unsaturated monocyclic or bicyclic ring in which all atoms of all rings are carbon. Thus, the term includes cycloalkyl and aryl rings.
- Monocyclic carbocycles have 3 to 6 ring atoms, still more typically 5 or 6 ring atoms.
- Bicyclic carbocycles have 7 to 12 ring atoms, e.g, arranged as a bicyclo [4,5], [5,5], [5,6] or [6,6] system, or 9 or 10 ring atoms arranged as a bicyclo [5,6] or [6,6] system.
- Examples of mono- and bicyclic carbocycles include cyclopropyl, cyclobutyl, cyclopentyl, 1 -cyclopent- 1-enyl, l-cyclopent-2-enyl, 1- cy cl opent-3 -enyl, cyclohexyl, 1 -cyclohex- 1-enyl, l-cyclohex-2-enyl, 1 -cyclohex-3 -enyl, phenyl and naphthyl.
- the carbocyclic ring may be substituted in which case the substituents are selected from those recited above for cycloalkyl and aryl groups.
- alkylthio refers to the group “alkyl-S-”.
- acyl refers to a group -C(O)R, wherein R is hydrogen, alkyl, cycloalkyl, heterocyclyl, aryl, heteroalkyl, or heteroaryl; each of which may be optionally substituted, as defined herein.
- R is hydrogen, alkyl, cycloalkyl, heterocyclyl, aryl, heteroalkyl, or heteroaryl; each of which may be optionally substituted, as defined herein.
- examples of acyl include formyl, acetyl, cylcohexylcarbonyl, cyclohexylmethyl-carbonyl, and benzoyl.
- amido refers to both a "C-amido” group which refers to the group — C(O)NR g R h and an "N-amido” group which refers to the group -NR g C(O)R h , wherein R g and R h are independently selected from hydrogen, alkyl, aryl, haloalkyl, or heteroaryl; each of which may be optionally substituted.
- amino refers to the group -NR g R h wherein R g and R h are independently selected from hydrogen, alkyl, haloalkyl, aryl, or heteroaryl; each of which may be optionally substituted.
- carbamoyl refers to both an "O-carbamoyl” group which refers to the group -O-C(O)NR'R' and an "N-carbamoyl” group which refers to the group - NR 1 C(O)OR i , wherein R 1 and R> are independently selected from hydrogen, alkyl, aryl, haloalkyl, or heteroaryl; each of which may be optionally substituted.
- Carboxyl ester refers to both -OC(O)R and -C(O)OR g , wherein R g is hydrogen, alkyl, cycloalkyl, heterocyclyl, aryl, heteroalkyl, or heteroaryl; each of which may be optionally substituted, as defined herein.
- cyano or "carbonitrile” refers to the group -CN.
- cycloalkyl refers to a saturated or partially unsaturated cyclic alkyl group having a single ring or multiple rings including fused, bridged, and spiro ring systems.
- cycloalkyl includes cycloalkenyl groups (i.e. the cyclic group having at least one double bond).
- cycloalkyl has from 3 to 20 ring carbon atoms (i.e., C. sub.3-20 cycloalkyl), 3 to 12 ring carbon atoms (i.e., C. sub.3-12 cycloalkyl), 3 to 10 ring carbon atoms (i.e., C.
- cycloalkyl groups include cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl.
- heteroatoms shall include oxygen, sulfur and nitrogen.
- substituted means that any one or more hydrogens on the designated atom or group is replaced with a selection from the indicated group, provided that the designated atom's normal valence is not exceeded.
- Keto substituents are not present on aromatic moieties.
- substituents are named into the core structure. For example, it is to be understood that when (cycloalkyl)alkyl is listed as a possible substituent, the point of attachment of this substituent to the core structure is in the alkyl portion.
- a stable compound or stable structure is meant to imply a compound that is sufficiently robust to survive isolation from a reaction mixture to a useful degree of purity, and subsequent formulation into an efficacious therapeutic agent. It is preferred that the presently recited compounds do not contain a N-halo, S(0)2H, or S(O)H group.
- salts denotes acidic and/or basic salts formed with inorganic and/or organic acids and bases.
- salt(s) may include zwitterions (inner salts), e.g., when a compound of formula I, contains both a basic moiety, such as an amine or a pyridine or imidazole ring, and an acidic moiety, such as a carboxylic acid.
- salts are preferred, such as, for example, acceptable metal and amine salts in which the cation does not contribute significantly to the toxicity or biological activity of the salt.
- other salts may be useful, e.g., in isolation or purification steps which may be employed during preparation, and thus, are contemplated within the scope of the invention.
- Salts of the compounds of herein may be formed, for example, by reacting a compound described herein with an amount of acid or base, such as an equivalent amount, in a medium such as one in which the salt precipitates or in an aqueous medium followed by lyophilization.
- Exemplary acid addition salts include acetates (such as those formed with acetic acid or trihaloacetic acid, for example, trifluoroacetic acid), adipates, alginates, ascorbates, aspartates, benzoates, benzenesulfonates, bisulfates, borates, butyrates, citrates, camphorates, camphorsulfonates, cyclopentanepropionates, digluconates, dodecylsulfates, ethanesulfonates, fumarates, glucoheptanoates, glycerophosphates, hemisulfates, heptanoates, hexanoates, hydrochlorides (formed with hydrochloric acid), hydrobromides (formed with hydrogen bromide), hydroiodides, 2- hydroxy ethanesulfonates, lactates, maleates (formed with maleic acid), methanesulfonates (formed with methanes
- Exemplary basic salts include ammonium salts, alkali metal salts such as sodium, lithium, and potassium salts; alkaline earth metal salts such as calcium and magnesium salts; barium, zinc, and aluminum salts; salts with organic bases (for example, organic amines) such as trialkylamines such as triethylamine, procaine, dibenzylamine, N-benzyl- P-phenethylamine, 1 -ephenamine, 7V,7V'-dibenzylethylene-diamine, dehydroabietylamine, N-ethylpiperidine, benzylamine, dicyclohexylamine or similar pharmaceutically acceptable amines and salts with amino acids such as arginine, lysine and the like.
- organic bases for example, organic amines
- trialkylamines such as triethylamine, procaine, dibenzylamine, N-benzyl- P-phenethylamine, 1 -ephenamine
- Basic nitrogen-containing groups may be quaternized with agents such as lower alkyl halides (e.g., methyl, ethyl, propyl, and butyl chlorides, bromides and iodides), dialkyl sulfates (e.g., dimethyl, diethyl, dibutyl, and diamyl sulfates), long chain halides (e.g., decyl, lauryl, myristyl and stearyl chlorides, bromides and iodides), aralkyl halides (e.g., benzyl and phenethyl bromides), and others.
- Preferred salts include monohydrochloride, hydrogensulfate, methanesulfonate, phosphate or nitrate salts.
- solvates e.g., hydrates
- the term “solvate” means a physical association of a compound with one or more solvent molecules, whether organic or inorganic. This physical association includes hydrogen bonding. In certain instances, the solvate will be capable of isolation, for example when one or more solvent molecules are incorporated in the crystal lattice of the crystalline solid.
- “Solvate” encompasses both solution-phase and isolable solvates. Exemplary solvates include hydrates, ethanolates, methanolates, isopropanolates, acetonitrile solvates, and ethyl acetate solvates. Methods of solvation are known in the art.
- compositions described herein subsequent to their preparation can be isolated and purified to obtain a composition containing an amount by weight equal to or greater than 99% of a compound (“substantially pure”), which is then used or formulated as described herein.
- substantially pure compounds described herein are also contemplated herein as part of the present invention.
- phrases "pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
- pharmaceutically acceptable salts refer to derivatives of the disclosed compounds wherein the parent compound is modified by making acid or base salts thereof.
- pharmaceutically-acceptable salts include, but are not limited to, mineral or organic acid salts of basic groups such as amines; and alkali or organic salts of acidic groups such as carboxylic acids.
- the pharmaceutically-acceptable salts include the conventional non-toxic salts or the quaternary ammonium salts of the parent compound formed, for example, from non-toxic inorganic or organic acids.
- such conventional non-toxic salts include those derived from inorganic acids such as hydrochloric, hydrobromic, sulfuric, sulfamic, phosphoric, and nitric; and the salts prepared from organic acids such as acetic, propionic, succinic, glycolic, stearic, lactic, malic, tartaric, citric, ascorbic, pamoic, maleic, hydroxymaleic, phenylacetic, glutamic, benzoic, salicylic, sulfanilic, 2-acetoxybenzoic, fumaric, toluenesulfonic, methanesulfonic, ethane disulfonic, oxalic, and isethionic, and the like.
- inorganic acids such as hydrochloric, hydrobromic, sulfuric, sulfamic, phosphoric, and nitric
- organic acids such as acetic, propionic, succinic, glycolic, stearic, lactic, malic,
- the pharmaceutically acceptable salts of the present invention can be synthesized from the parent compound which contains a basic or acidic moiety by conventional chemical methods.
- such salts can be prepared by reacting the free acid or base forms of these compounds with a stoichiometric amount of the appropriate base or acid in water or in an organic solvent, or in a mixture of the two; generally, nonaqueous media like ether, ethyl acetate, ethanol, isopropanol, or acetonitrile are preferred.
- nonaqueous media like ether, ethyl acetate, ethanol, isopropanol, or acetonitrile are preferred.
- Lists of suitable salts are found in Remington's Pharmaceutical Sciences, 18th Edition, Mack Publishing Company, Easton, PA (1990), the disclosure of which is hereby incorporated by reference.
- “Stable compound” and “stable structure” are meant to indicate a compound that is sufficiently robust to survive isolation to a useful degree of purity from a reaction mixture, and formulation into an efficacious therapeutic agent.
- the present invention is intended to embody stable compounds.
- “Therapeutically effective amount” is intended to include an amount of a compound of the present invention alone or an amount of the combination of compounds claimed or an amount of a compound of the present invention in combination with other active ingredients effective to act as an inhibitor of USP1, or effective to treat or prevent proliferative disorders, such as cancer.
- treating cover the treatment of a disease-state in a mammal, particularly in a human, and include: (a) preventing the disease-state from occurring in a mammal, in particular, when such mammal is predisposed to the diseasestate but has not yet been diagnosed as having it; (b) inhibiting the disease-state, i.e., arresting its development; and/or (c) relieving the disease-state, i.e., causing regression of the disease state.
- Stereoisomers may include compounds which are optical isomers through possession of one or more chiral atoms, as well as compounds which are optical isomers by virtue of limited rotation about one or more bonds (atropisomers).
- the definition of compounds according to the invention embraces all the possible stereoisomers and their mixtures. It very particularly embraces the racemic forms and the isolated optical isomers having the specified activity.
- the racemic forms can be resolved by physical methods, such as, for example, fractional crystallization, separation or crystallization of diastereomeric derivatives or separation by chiral column chromatography.
- the individual optical isomers can be obtained from the racemates from the conventional methods, such as, for example, salt formation with an optically active acid followed by crystallization.
- the present invention is intended to include all isotopes of atoms occurring in the present compounds.
- Isotopes include those atoms having the same atomic number but different mass numbers.
- isotopes of hydrogen include deuterium and tritium.
- Isotopes of carbon include 13 C and 14 C.
- Isotopically-labeled compounds of the invention can generally be prepared by conventional techniques known to those skilled in the art or by processes analogous to those described herein, using an appropriate isotopically-labeled reagent in place of the non-labeled reagent otherwise employed.
- Prodrugs and solvates of the inventive compounds are also contemplated.
- the term "prodrug” denotes a compound which, upon administration to a subject, undergoes chemical conversion by metabolic or chemical processes to yield a compound of the formula I, and/or a salt and/or solvate thereof. Any compound that will be converted in vivo to provide the bioactive agent (ie., the compound for formula I) is a prodrug within the scope and spirit of the invention.
- compounds containing a carboxy group can form physiologically hydrolyzable esters which serve as prodrugs by being hydrolyzed in the body to yield formula I compounds per se.
- Such prodrugs are preferably administered orally since hydrolysis in many instances occurs principally under the influence of the digestive enzymes.
- esters of compounds of formula I include Ci-ealkylbenzyl, 4-methoxybenzyl, indanyl, phthalyl, methoxymethyl, Ci-ealkanoyloxy-Ci-ealkyl, e.g., acetoxymethyl, pivaloyloxymethyl or propionyloxymethyl, Ci-ealkoxycarbonyloxy-Ci-ealkyl, e.g., methoxycarbonyl-oxymethyl or ethoxycarbonyloxymethyl, glycyloxymethyl, phenylglycyloxymethyl, (5-methyl-2- oxo- 1,3 -di oxolen-4-yl)-m ethyl and other well known physiologically hydrolyzable esters used, for example, in the penicillin and cephalosporin arts. Such esters may
- the disclosure herein further relates to compounds described herein, the tautomers and stereoisomeric forms thereof, and the pharmaceutically acceptable addition salts, and the solvates thereof, for use as a medicament. Furthermore, the disclosure herein relates to the use of a compound described herein, a tautomer or a stereoisomeric form thereof, or a pharmaceutically acceptable addition salt, or a solvate thereof, or a pharmaceutical composition according to the invention, for the manufacture of a medicament.
- inventive compositions may contain other therapeutic agents as described above and may be formulated, for example, by employing conventional solid or liquid vehicles or diluents, as well as pharmaceutical additives of a type appropriate to the mode of desired administration (e.g., excipients, binders, preservatives, stabilizers, flavors, etc.) according to techniques such as those well known in the art of pharmaceutical formulation.
- pharmaceutical additives e.g., excipients, binders, preservatives, stabilizers, flavors, etc.
- the present invention further includes compositions comprising one or more compounds described herein and a pharmaceutically acceptable carrier.
- a “pharmaceutically acceptable carrier” refers to media generally accepted in the art for the delivery of biologically active agents to animals, in particular, mammals.
- Pharmaceutically acceptable carriers are formulated according to a number of factors well within the purview of those of ordinary skill in the art. These include without limitation the type and nature of the active agent being formulated; the subject to which the agentcontaining composition is to be administered; the intended route of administration of the composition; and, the therapeutic indication being targeted.
- Pharmaceutically acceptable carriers include both aqueous and non-aqueous liquid media, as well as a variety of solid and semi-solid dosage forms.
- Such carriers can include a number of different ingredients and additives in addition to the active agent, such additional ingredients being included in the formulation for a variety of reasons, e.g., stabilization of the active agent, binders, etc., well known to those of ordinary skill in the art.
- suitable pharmaceutically acceptable carriers, and factors involved in their selection are found in a variety of readily available sources such as, for example, Remington's Pharmaceutical Sciences, 17th Edition (1985), which is incorporated herein by reference in its entirety.
- the compounds described herein may be administered by any means suitable for the condition to be treated, which may depend on the need for site-specific treatment or quantity of drug to be delivered. Topical administration is generally preferred for skin- related diseases, and systematic treatment preferred for cancerous or pre-cancerous conditions, although other modes of delivery are contemplated.
- the compounds may be delivered orally, such as in the form of tablets, capsules, granules, powders, or liquid formulations including syrups; topically, such as in the form of solutions, suspensions, gels or ointments; sublingually; bucally; parenterally, such as by subcutaneous, intravenous, intramuscular or intrasternal injection or infusion techniques (e.g., as sterile injectable aq. or non-aq.
- Dosage unit formulations containing non-toxic, pharmaceutically acceptable vehicles or diluents may be administered.
- the compounds may be administered in a form suitable for immediate release or extended release. Immediate release or extended release may be achieved with suitable pharmaceutical compositions or, particularly in the case of extended release, with devices such as subcutaneous implants or osmotic pumps.
- compositions for oral administration include suspensions which may contain, for example, microcrystalline cellulose for imparting bulk, alginic acid or sodium alginate as a suspending agent, methylcellulose as a viscosity enhancer, and sweeteners or flavoring agents such as those known in the art; and immediate release tablets which may contain, for example, microcrystalline cellulose, dicalcium phosphate, starch, magnesium stearate and/or lactose and/or other excipients, binders, extenders, disintegrants, diluents and lubricants such as those known in the art.
- the inventive compounds may also be orally delivered by sublingual and/or buccal administration, e.g., with molded, compressed, or freeze-dried tablets.
- compositions may include fast-dissolving diluents such as mannitol, lactose, sucrose, and/or cyclodextrins.
- fast-dissolving diluents such as mannitol, lactose, sucrose, and/or cyclodextrins.
- high molecular weight excipients such as celluloses (AVICEL®) or polyethylene glycols (PEG); an excipient to aid mucosal adhesion such as hydroxypropyl cellulose (HPC), hydroxypropyl methyl cellulose (HPMC), sodium carboxymethyl cellulose (SCMC), and/or maleic anhydride copolymer (e.g., GANTREZ®); and agents to control release such as polyacrylic copolymer (e.g., CARBOPOL 934®).
- Lubricants, glidants, flavors, coloring agents and stabilizers may also be added for ease of fabrication and use.
- Formulations for parenteral administration may be in the form of aqueous or nonaqueous isotonic sterile injection solutions or suspensions. These solutions and suspensions may be prepared from sterile powders or granules using one or more of the carriers or diluents mentioned for use in the formulations for oral administration or by using other suitable dispersing or wetting agents and suspending agents.
- the compounds may be dissolved in water, polyethylene glycol, propylene glycol, ethanol, com oil, cottonseed oil, peanut oil, sesame oil, benzyl alcohol, sodium chloride, tragacanth gum, and/or various buffers. Other adjuvants and modes of administration are well and widely known in the pharmaceutical art.
- the active ingredient may also be administered by injection as a composition with suitable carriers including saline, dextrose, or water, or with cyclodextrin (i.e. Captisol), cosolvent solubilization (i.e. propylene glycol) or micellar solubilization (i.e. Tween 80).
- suitable carriers including saline, dextrose, or water, or with cyclodextrin (i.e. Captisol), cosolvent solubilization (i.e. propylene glycol) or micellar solubilization (i.e. Tween 80).
- compositions for parenteral administration include injectable solutions or suspensions which may contain, for example, suitable non-toxic, parenterally acceptable diluents or solvents, such as mannitol, 1,3 -butanediol, water, Ringer's solution, an isotonic sodium chloride solution, or other suitable dispersing or wetting and suspending agents, including synthetic mono- or diglycerides, and fatty acids, including oleic acid.
- suitable non-toxic, parenterally acceptable diluents or solvents such as mannitol, 1,3 -butanediol, water, Ringer's solution, an isotonic sodium chloride solution, or other suitable dispersing or wetting and suspending agents, including synthetic mono- or diglycerides, and fatty acids, including oleic acid.
- the sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally acceptable diluent or solvent, for example as a solution in 1,3 -butanediol.
- a non-toxic parenterally acceptable diluent or solvent for example as a solution in 1,3 -butanediol.
- acceptable vehicles and solvents that may be employed are water, Ringer’s solution, and isotonic sodium chloride solution.
- sterile, fixed oils are conventionally employed as a solvent or suspending medium.
- any bland fixed oil may be employed, including synthetic mono- or diglycerides.
- fatty acids such as oleic acid find use in the preparation of injectables.
- a sterile injectable oil-in-water microemulsion can, for example, be prepared by 1) dissolving at least one compound described herein in an oily phase, such as, for example, a mixture of soybean oil and lecithin; 2) combining the compound-containing oil phase with a water and glycerol mixture; and 3) processing the combination to form a microemulsion.
- an oily phase such as, for example, a mixture of soybean oil and lecithin
- combining the compound-containing oil phase with a water and glycerol mixture and 3) processing the combination to form a microemulsion.
- a sterile aqueous or oleaginous suspension can be prepared in accordance with methods already known in the art.
- a sterile aqueous solution or suspension can be prepared with a non-toxic parenterally-acceptable diluent or solvent, such as, for example, 1,3-butane diol; and a sterile oleaginous suspension can be prepared with a sterile non-toxic acceptable solvent or suspending medium, such as, for example, sterile fixed oils, e.g., synthetic mono- or diglycerides; and fatty acids, such as, for example, oleic acid.
- compositions for nasal aerosol or inhalation administration include solutions which may contain, for example, benzyl alcohol or other suitable preservatives, absorption promoters to enhance absorption and/or bioavailability, and/or other solubilizing or dispersing agents such as those known in the art.
- Dispersible powders and granules can, for example, be prepared by admixing at least one compound described herein or a pharmaceutically acceptable salt thereof, with at least one dispersing and/or wetting agent; at least one suspending agent; and/or at least one preservative.
- exemplary preservatives include, but are not limited to, for example, anti-oxidants, e.g., ascorbic acid.
- dispersible powders and granules can also contain at least one excipient, including, but not limited to, for example, sweetening agents; flavoring agents; and coloring agents.
- compositions for rectal administration include suppositories which may contain, for example, suitable non-irritating excipients, such as cocoa butter, synthetic glyceride esters or polyethylene glycols, which are solid at ordinary temperatures but liquefy and/or dissolve in the rectal cavity to release the drug.
- suitable non-irritating excipients such as cocoa butter, synthetic glyceride esters or polyethylene glycols, which are solid at ordinary temperatures but liquefy and/or dissolve in the rectal cavity to release the drug.
- the therapeutically-effective amount of a compound of the present invention may be determined by one of ordinary skill in the art, and includes exemplary dosage amounts for a mammal of from about 0.05 to 1000 mg/kg; 1-1000 mg/kg; 1-50 mg/kg; 5-250 mg/kg; 250-1000 mg/kg of body weight of active compound per day, which may be administered in a single dose or in the form of individual divided doses, such as from 1 to 4 times per day.
- the specific dose level and frequency of dosage for any particular subject may be varied and will depend upon a variety of factors, including the activity of the specific compound employed, the metabolic stability and length of action of that compound, the species, age, body weight, general health, sex and diet of the subject, the mode and time of administration, rate of excretion, drug combination, and severity of the particular condition.
- Preferred subjects for treatment include animals, most preferably mammalian species such as humans, and domestic animals such as dogs, cats, horses, and the like.
- this term is intended to include all subjects, most preferably mammalian species that are affected by modulation of USP1 -mediated functions.
- the present application provides a combined preparation of a compound described herein and/or a pharmaceutically acceptable salt thereof, a stereoisomer thereof or a tautomer thereof, and additional therapeutic agent(s) for simultaneous, separate or sequential use in the treatment and/or prophylaxis of multiple diseases or disorders associated with USP1.
- the application provides a method of treating a patient suffering from or susceptible to a medical condition that is associated with USP1.
- a number of medical conditions can be treated.
- the method comprises administering to the patient a therapeutically effective amount of a composition comprising a compound described herien and/or a pharmaceutically acceptable salt thereof, a stereoisomer thereof or a tautomer thereof.
- the compounds described herein may be used to treat or proliferative diseases such as cancer, immunological disorders or inflammatory disorders.
- the compounds described herein are useful for the treatment of cancer.
- the compounds described herein may be used to treat cancers that are mediated by, dependent on or associated with USP1 activity.
- the disease is a solid tumor.
- the solid tumor is selected from prostate cancer, pancreatic cancer, bladder cancer, colorectal cancer, breast cancer, renal cancer, hepatocellular cancer, lung cancer, ovarian cancer, cervical cancer, gastric cancer, esophageal cancer, head and neck cancer, melanoma, neuroendocrine cancers, CNS cancers, brain tumors (e.g., glioma, anaplastic oligodendroglioma, adult glioblastoma multiforme, and adult anaplastic astrocytoma), bone cancer, or soft tissue sarcoma.
- the solid tumor is from non-small cell lung cancer or small-cell lung cancer.
- the compounds herein can be useful in the treatment of haematological malignancies.
- hematological malignancy is selected from multiple myeloma, non-Hodgkin's lymphoma, Hodgkin lymphoma, T-cell leukaemia, mucosa-associated lymphoid tissue lymphoma, diffuse large B-cell lymphoma and mantle cell lymphoma.
- solid tumor is selected from pancreatic cancer, breast cancer, melanoma and non-small cell lung cancer.
- cancer is selected from a carcinoma, preferably a carcinoma of the bladder, breast, colon (including colorectal carcinomas, such as colon adenocarcinoma and colon adenoma), kidney, urothelial, uterus, epidermis, liver, lung (including adenocarcinoma, small cell lung cancer, non-small cell lung carcinomas and squamous lung cancer), oesophagus, head and neck, gall bladder, ovary, pancreas (including exocrine pancreatic carcinoma), stomach, gastrointestinal cancer (including gastrointestinal stromal tumors), cervix, endometrium, thyroid, prostate and skin.
- a carcinoma preferably a carcinoma of the bladder, breast, colon (including colorectal carcinomas, such as colon adenocarcinoma and colon adenoma), kidney, urothelial, uterus, epidermis, liver, lung (including adenocarcinoma, small cell lung cancer, non-small cell lung carcinomas and
- the cancer is selected from pituitary cancer, a hematopoietic tumor of lymphoid lineage, for example leukemia, acute lymphocytic leukemia, chronic lymphocytic leukemia, B-cell lymphoma (e.g.
- B-cell lymphoma diffuse large B-cell lymphoma, mantle cell lymphoma), T-cell leukaemia/lymphoma, Hodgkin's lymphoma, non-Hodgkin's lymphoma, hairy cell lymphoma, or Burkett's lymphoma; a hematopoietic tumor of myeloid lineage, for example leukemias, acute and chronic myelogenous leukemias, chronic myelomonocytic leukemia (CMML), myeloproliferative disorder, myeloproliferative syndrome, myelodysplastic syndrome, or promyelocytic leukemia; multiple myeloma; thyroid follicular cancer; hepatocellular cancer, a tumor of mesenchymal origin (e.g.
- Ewing's sarcoma for example fibrosarcoma or rhabdomyosarcoma; a tumor of the central or peripheral nervous system, for example astrocytoma, neuroblastoma, glioma (such as glioblastoma multiforme) or schwannoma; melanoma; seminoma; teratocarcinoma; osteosarcoma; xeroderma pigmentosum; keratoctanthoma; thyroid follicular cancer; or Kaposi's sarcoma.
- astrocytoma neuroblastoma
- glioma such as glioblastoma multiforme
- schwannoma schwannoma
- melanoma seminoma
- osteosarcoma xeroderma pigmentosum
- keratoctanthoma thyroid follicular cancer
- Kaposi's sarcoma Kaposi'sarcoma.
- the compounds described herein may be used to treat cancers that are mediated by, dependent on or associated with USP1 activity.
- the disease is a solid tumor.
- the solid tumor is from prostate cancer, pancreatic cancer, bladder cancer, colorectal cancer, breast cancer, renal cancer, hepatocellular cancer, lung cancer, ovarian cancer, cervical cancer, gastric cancer, esophageal cancer, head and neck cancer, melanoma, neuroendocrine cancers, CNS cancers, brain tumors (e.g., glioma, anaplastic oligodendroglioma, adult glioblastoma multiforme, and adult anaplastic astrocytoma), bone cancer, or soft tissue sarcoma.
- the solid tumor is from non-small cell lung cancer or small-cell lung cancer.
- the disease is a hematologic malignancy.
- the disease is lymphoma, multiple myeloma, or leukemia.
- the hematologic malignancy is leukemia or lymphoma.
- the disease is acute lymphocytic leukemia (ALL), acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma (SLL), myelodysplastic syndrome (MDS), myeloproliferative disease (MPD), chronic myeloid leukemia (CML), juvenile myelomonocytic leukemia (JMML), multiple myeloma (MM), Hodgkin lymphoma, indolent non-Hodgkin's lymphoma (iNHL), refractory iNHL, nonHodgkin's lymphoma (NHL), mantle cell lymphoma (MCL), follicular lymphoma, Waldenstrom’s macroglobulinemia (WM), minimal residual disease (MRD), T-cell lymphoma, B-cell lymphoma, diffuse large B-cell lymphoma (DLBCL), T-cell acute lymphoblastic leuk
- ALL
- the disease is T-cell acute lymphoblastic leukemia (T-ALL), or B-cell acute lymphoblastic leukemia (B-ALL).
- non-Hodgkin lymphoma can be indolent B-cell diseases including follicular lymphoma, lymphoplasmacytic lymphoma, Waldenstrom macroglobulinemia, and marginal zone lymphoma, as well as the aggressive lymphomas that include, for example, Burkitt lymphoma, diffuse large B- cell lymphoma (DLBCL) and mantle cell lymphoma (MCL).
- T-ALL T-cell acute lymphoblastic leukemia
- B-ALL B-cell acute lymphoblastic leukemia
- non-Hodgkin lymphoma can be indolent B-cell diseases including follicular lymphoma, lymphoplasmacytic lymphoma, Waldenstrom macroglobulinemia, and marginal zone lymphoma, as well as the aggressive lymphomas that include, for example, Burkitt lympho
- the cancer is selected from hematological cancer, a lymphatic cancer.
- the cancer comprises cancer cells with DNA damage repair pathway deficiency.
- the cancer is a homologous recombination deficient cancer.
- the cancer comprises cancer cells with a mutation in a gene encoding p53.
- the mutation in a gene encoding p53 is a germline or somatic mutation.
- the cancer comprises with cancer cells with loss of function mutation in a gene encoding p53.
- the cancer is a BRCA1 and/or BRCA2 deficient cancer.
- the cancer is a somatic or germline BRCA1 and/or BRCA2 mutant cancer.
- the cancer is a Poly (ADP -ribose) polymerase (“PARP”) inhibitor refractory or resistant cancer.
- PARP Poly (ADP -ribose) polymerase
- the cancer is a PARP inhibitor resistant or refractory BRCA1 and/or BRCA2 deficient cancer.
- the cancer cell has a germline or somatic mutation in a gene encoding ataxia telangiectasia mutated (ATM) protein kinase or ATM deficiency.
- ATM telangiectasia mutated
- the cancer has a mutation in the gene encoding more than two of p53, BRCA1, BRCA2, ATM.
- the disease is an autoimmune or inflammatory disease or disorder.
- autoimmune or inflammatory diseases or conditions may be chronic or acute and include, but are not limited to, inflammatory pelvic disease, urethritis, skin sunburn, sinusitis, pneumonitis, encephalitis, meningitis, myocarditis, pericarditis, nephritis including lupus nephritis, osteomyelitis, myositis, eczema, hepatitis, gastritis, enteritis, dermatitis, gingivitis, appendicitis, pancreatitis, primary biliary cirrhosis, cholecystitis, sclerosing cholangitis, agammaglobulinemia, psoriasis, allergy, Crohn's disease, irritable bowel syndrome, ulcerative colitis, Sjogren's disease, tissue graft rejection such as acute graft-versus-host disease,
- the autoimmune and inflammatory diseases and conditions may also include systemic or tissue inflammation, inflammatory responses to hypoxia, cellular activation and proliferation, lipid metabolism, fibrosis, infections with bacteria, infections with viruses (e.g., herpes virus, human papilloma virus, adenovirus, poxvirus and other DNA viruses), fungi, parasites or their toxins, such as sepsis, sepsis syndrome, septic shock, endotoxaemia, systemic inflammatory response syndrome (SIRS), multi-organ dysfunction syndrome, toxic shock syndrome, acute lung injury, ARDS (adult respiratory distress syndrome), acute renal failure, fulminant hepatitis, burns, acute pancreatitis, post-surgical syndromes, sarcoidosis, Herxheimer reactions, encephalitis, myelitis, meningitis, malaria and SIRS associated with viral infections such as influenza, herpes zoster, herpes simplex and coronavirus.
- viruses e.g., herpes virus
- the compounds described herein may be administered in conjunction with standard of care, e.g., surgery, radiation, and/or chemotherapy.
- the compounds may be administered in conjunction with a chemotherapeutic agent.
- the compounds may be administered in conjunction with one or more of carboplatin, cisplatin, paclitaxel, nab-paclitaxel, gemcitabine or FOLFOX.
- the compounds may be administered in conjunction with carboplatin or nab-paclitaxel.
- the compounds may be administered in conjunction with carboplatin and paclitaxel.
- the compounds may be administered in conjunction with cisplatin and pemetrexed.
- the compounds may be administered in conjunction with cisplatin and gemcitabine. In some embodiments, the compounds may be administered in conjunction with FOLFOX. In some embodiments, the compounds may be administered in conjunction with FOLFIRI. In one embodiment, the compounds may be administered in combination with decarbazine for the treatment of melanoma. In some embodiments, cisplatin is intravenously administered as a 100 mg/ml dose once every four weeks. In some embodiments, the compounds may be administered in conjunction with doxorubicin (adriamycin), cisplatin bleomycin sulfate, carmustine, chlorambucil, dacarbazine and/or cyclophosphamide hydroxyurea. In some embodiments, adriamycin is intravenously administered as a 60 mg/ml to 75 mg/ml dose once every 21 days.
- the compounds of the present application may be used in combination with one or more additional therapeutic agent that are being used and/or developed to treat cancers or inflammatory disorders.
- the one or more additional therapeutic agent may be an inhibitor to Janus kinase (JAK) such as JAK1, JAK2 and/or JAK3, Tyroansine kinase (TYK), K-Ras, Mitogen activated protein kinases (MAPK), Bruton's tyrosine kinase (BTK), bromodomain containing protein inhibitor (BRD) such as BRD4, a lysyl oxidase protein (LOX), lysyl oxidase-like protein (LOXL) such as LOXL1-5, matrix metalloprotease (MMP) such as MMP 1-10, adenosine A2B receptor (A2B), isocitrate dehydrogenase (IDH) such as IDH1, apoptosis signal -regulating kinase (ASK) such as ASK1, serine/threonine kinase TPL2, discoidin domain receptor (DDR) such as DDR
- the compounds of the present application may be used in combination with additional chemotherapeutic agent, an immunotherapeutic agent, a radiotherapeutic agent, an anti-neoplastic agent, an anti-cancer agent, an anti- fibrotic agent, an anti -angiogenic agent, a therapeutic antibody, or any combination thereof.
- Chemotherapeutic agents may be categorized by their mechanism of action into, for example, the following groups: anti-metabolites/anti-cancer agents, such as pyrimidine analogs (floxuridine, capecitabine, and cytarabine); purine analogs, folate antagonists and related inhibitors antiproliferative/antimitotic agents including natural products such as vinca alkaloid (vinblastine, vincristine) and microtubule such as taxane (paclitaxel, docetaxel), vinblastin, nocodazole, epothilones and navelbine, epidipodophyllotoxins (etoposide, teniposide); DNA damaging agents (actinomycin, amsacrine, busulfan, carboplatin, chlorambucil, cisplatin, cyclophosphamide, Cytoxan, dactinomycin, daunorubicin, doxorubicin, epirubicin, iphosphamide,
- chemotherapeutic agents include alkylating agents such as thiotepa and cyclophosphamide; alkyl sulfonates such as busulfan, improsulfan and piposulfan; aziridines such as benzodopa, carboquone, meturedopa, and uredopa; emylerumines and memylamelamines including alfretamine, triemylenemelamine, triethylenephosphoramide, triethylenethiophosphoramide and trimemylolomelamine; acetogenins (especially bullatacin and bullatacinone); a camptothecin (including synthetic analogue topotecan); bryostatin; callystatin; CC-1065 (including its adozelesin, carzelesin and bizelesin synthetic analogues); cryptophycins (articularly cryptophycin 1 and cryptophy cin 8); dolastatin; duocarmycin (including the
- Chemotherapeutic agents may also include, for example, anti-hormonal agents that act to regulate or inhibit hormone action on tumors such as anti-estrogens and selective estrogen receptor modulators (SERMs), including, for example, tamoxifen, raloxifene, droloxifene, 4-hydroxytamoxifen, trioxifene, keoxifene, onapristone, and toremifene; inhibitors of the enzyme aromatase, which regulates estrogen production in the adrenal glands, such as, for example, 4(5)-imidazoles, aminoglutethimide, megestrol acetate, exemestane, formestane, fadrozole, vorozole letrozole and anastrozole; and antiandrogens such as flutamide, nilutamide, bicalutamide, leuprohde, and goserelin; and pharmaceutically acceptable salts thereof.
- SERMs selective estrogen receptor modulators
- anti-angiogenesis agents include antibodies, preferably monoclonal antibodies against these angiogenic growth factors: beta-FGF, alpha-FGF, FGF-5, VEGF isoforms, VEGF- C, HGF/SF and Ang-l/Ang-2.
- the application also provides a method for treating a subject who is undergoing one or more standard therapies, such as chemotherapy, radiotherapy, immunotherapy, surgery, or combination thereof. Accordingly, one or more therapeutic agent or inhibitors may be administered before, during, or after administration of chemotherapy, radiotherapy, immunotherapy, surgery or combination thereof.
- the subject may be a human who is (i) substantially refractory to at least one chemotherapy treatment, or (ii) in relapse after treatment with chemotherapy, or both (i) and (ii). In some of embodiments, the subject is refractory to at least two, at least three, or at least four chemotherapy treatments (including standard or experimental chemotherapies).
- the subject is refractory to at least one, at least two, at least three, or at least four chemotherapy treatment (including standard or experimental chemotherapy) selected from fludarabine, rituximab, obinutuzumab, alkylating agents, alemtuzumab and other chemotherapy treatments such as CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone); R-CHOP (rituximab-CHOP); hyperCVAD (hyperfractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone, methotrexate, cytarabine); R-hyperCVAD (rituximab-hyperCVAD); FCM (fludarabine, cyclophosphamide, mitoxantrone); R-FCM (rituximab, fludarabine, cyclophosphamide, mitoxantrone); bortezom
- chemotherapy treatment
- immunotherapeutic agents treating lymphoma or leukemia include, but are not limited to, rituximab (such as Rituxan), alemtuzumab (such as Campath, MabCampath), anti-CD19 antibodies, anti-CD20 antibodies, anti -MN- 14 antibodies, anti- TRAIL, Anti-TRAIL DR4 and DR5 antibodies, anti-CD74 antibodies, apolizumab, bevacizumab, CHIR-12.12, epratuzumab (hLL2-anti-CD22 humanized antibody), galiximab, ha20, ibritumomab tiuxetan, lumiliximab, milatuzumab, ofatumumab, PRO131921, SGN-40, WT-1 analog peptide vaccine, WT1 126
- the compounds of the application can be used in combination with additional therapeutic procedures.
- Other therapeutic procedures include peripheral blood stem cell transplantation, autologous hematopoietic stem cell transplantation, autologous bone marrow transplantation, antibody therapy, biological therapy, enzyme inhibitor therapy, total body irradiation, infusion of stem cells, bone marrow ablation with stem cell support, in vitro-treated peripheral blood stem cell transplantation, umbilical cord blood transplantation, immunoenzyme technique, pharmacological study, low-LET cobalt-60 gamma ray therapy, bleomycin, conventional surgery, radiation therapy, and nonmyeloablative allogeneic hematopoietic stem cell transplantation.
- the compounds of the application can be used in combination with anti-fibrotic agents.
- the anti-fibrotic agents include, but are not limited to, emylenemamine, hydrazine, phenylhydrazine, and their derivatives, semicarbazide, and urea derivatives, aminonitriles, such as beta-aminopropionitrile (BAPN), or 2-nitroethylamine, unsaturated or saturated haloamines, such as 2-bromo-ethylamine, 2-chloroethylamine, 2- trifluoroethylamine, 3 -bromopropylamine, p-halobenzylamines, selenohomocysteine lactone.
- BAPN beta-aminopropionitrile
- 2-nitroethylamine unsaturated or saturated haloamines, such as 2-bromo-ethylamine, 2-chloroethylamine, 2- trifluoroethylamine, 3 -bro
- the anti-fibrotic agents are copper chelating agents, penetrating or not penetrating the cells.
- Exemplary compounds include indirect inhibitors such compounds blocking the aldehyde derivatives originating from the oxidative deamination of the lysyl and hydroxylysyl residues by the lysyl oxidases, such as the thiolamines, in particular D- penicillamine, or its analogues such as 2-amino-5-mercapto-5-methylhexanoic acid, D-2- amino-3-methyl-3-((2-acetamidoethyl)dithio)butanoic acid, p-2-amino-3-methyl-3-((2- aminoethyl)dithio)butanoic acid, sodium -4-((p-l -dimethyl-2-amino-2- carboxyethyl)dithio)butane sulphurate, 2-acetamidoethyl-2-acetamidoe
- the compounds of the application can be used in combination with immunotherapeutic and anti-inflammatory treatments.
- the immunotherapeutic agents include and are not limited to therapeutic antibodies suitable for treating patients; such as abagovomab, adecatumumab, afutuzumab, alemtuzumab, altumomab, amatuximab, anatumomab, arcitumomab, bavituximab, bectumomab, bevacizumab, bivatuzumab, blinatumomab, brentuximab, cantuzumab, catumaxomab, cetuximab, citatuzumab, cixutumumab, clivatuzumab, conatumumab, daratumumab, drozitumab, duligotumab, dusigitumab, detumomab, dacetuzumab, da
- the immuno-oncology agent is (i) an agonist of a stimulatory (including a co-stimulatory) receptor or (ii) an antagonist of an inhibitory (including a co- inhibitory) signal on T cells, both of which result in amplifying antigen-specific T cell responses (often referred to as immune checkpoint regulators).
- Certain of the stimulatory and inhibitory molecules are members of the immunoglobulin super family (IgSF).
- IgSF immunoglobulin super family
- B7 family which includes B7- 1, B7-2, B7-H1 (PD-L1), B7-DC (PD-L2), B7-H2 (ICOS-L), B7-H3, B7-H4, B7-H5 (VISTA), and B7-H6.
- TNF family of molecules that bind to cognate TNF receptor family members which includes CD40 and CD40L, OX-40, OX-40L, CD70, CD27L, CD30, CD30L, 4-1BBL, CD137 (4-1BB), TRAIL/Apo2-L, TRAILR1/DR4, TRAILR2/DR5, TRAILR3, TRAILR4, OPG, RANK, RANKL, TWEAKR/Fnl4, TWEAK, BAFFR, ED AR, XEDAR, TACI, APRIL, BCMA, LTpR, LIGHT, DcR3, HVEM, VEGI/TL1A, TRAMP/DR3, ED AR, EDAI, XEDAR, EDA2, TNFR1, Lymphotoxin a/TNFp, TNFR2, TNFa, LTpR, Lymphotoxin a ip2,
- T cell responses can be stimulated by a combination of a compound of Formula (I) and one or more of (i) an antagonist of a protein that inhibits T cell activation (e.g., immune checkpoint inhibitors) such as CTLA-4, PD-1, PD-L1, PD-L2, LAG-3, TIM-3, Galectin 9, CEACAM-1, BTLA, CD69, Galectin-1, TIGIT, CD113, GPR56, VISTA, 2B4, CD48, GARP, PD1H, LAIR1, TIM-1, and TIM-4, and (ii) an agonist of a protein that stimulates T cell activation such as B7-1, B7-2, CD28, 4-1BB (CD137), 4-1BBL, ICOS, ICOS-L, 0X40, OX40L, GITR, GITRL, CD70, CD27, CD40, DR3 and CD28H.
- an antagonist of a protein that inhibits T cell activation e.g., immune
- agents that can be combined with compounds described herein for the treatment of cancer include antagonists of inhibitory receptors on NK cells or agonists of activating receptors on NK cells.
- compounds described herien can be combined with antagonists of KIR, such as lirilumab.
- agents for combination therapies include agents that inhibit or deplete macrophages or monocytes, including but not limited to CSF-1R antagonists such as CSF-1R antagonist antibodies including RG7155 (WO11/70024, WO11/107553, WO1 1/131407, WO13/87699, WO13/119716, WO13/132044) or FPA-008 (WO11/140249; WO13169264; WO14/036357).
- CSF-1R antagonists such as CSF-1R antagonist antibodies including RG7155 (WO11/70024, WO11/107553, WO1 1/131407, WO13/87699, WO13/119716, WO13/132044) or FPA-008 (WO11/140249; WO13169264; WO14/036357).
- compounds of the present application can be used with one or more of agonistic agents that ligate positive costimulatory receptors, blocking agents that attenuate signaling through inhibitory receptors, antagonists, and one or more agents that increase systemically the frequency of anti-tumor T cells, agents that overcome distinct immune suppressive pathways within the tumor microenvironment (e.g., block inhibitory receptor engagement (e.g., PD-L1/PD-1 interactions), deplete or inhibit Tregs (e.g., using an anti-CD25 monoclonal antibody (e.g., daclizumab) or by ex vivo anti-CD25 bead depletion), inhibit metabolic enzymes such as IDO, or rever se/prevent T cell anergy or exhaustion) and agents that trigger innate immune activation and/or inflammation at tumor sites.
- agonistic agents that ligate positive costimulatory receptors e.g., blocking agents that attenuate signaling through inhibitory receptors, antagonists, and one or more agents that increase systemically
- the immuno-oncology agent is a CTLA-4 antagonist, such as an antagonistic CTLA-4 antibody.
- CTLA-4 antibodies include, for example, YERVOY (ipilimumab) or tremelimumab.
- the immuno-oncology agent is a PD-1 antagonist, such as an antagonistic PD-1 antibody.
- Suitable PD-1 antibodies include, for example, OPDIVO (nivolumab), KEYTRUDA (pembrolizumab), or MEDL0680 (AMP-514; WO2012/145493).
- the immuno-oncology agent may also include pidilizumab (CT-011), though its specificity for PD-1 binding has been questioned.
- Another approach to target the PD-1 receptor is the recombinant protein composed of the extracellular domain of PD-L2 (B7-DC) fused to the Fc portion of IgGl, called AMP-224
- the immuno-oncology agent is a PD-L1 antagonist, such as an antagonistic PD-L1 antibody.
- Suitable PD-L1 antibodies include, for example, MPDL3280A (RG7446; WO2010/077634), durvalumab (MEDI4736), BMS-936559 (W02007/005874), and MSB0010718C (WO2013/79174).
- the immuno-oncology agent is a LAG-3 antagonist, such as an antagonistic LAG-3 antibody.
- LAG3 antibodies include, for example, BMS- 986016 (W010/19570, WO14/08218), or IMP-731 or IMP-321 (W008/132601, WO09/44273).
- the immuno-oncology agent is a CD137 (4-1BB) agonist, such as an agonistic CD137 antibody.
- Suitable CD137 antibodies include, for example, urelumab and PF-05082566 (WO12/32433).
- the immuno-oncology agent is a GITR agonist, such as an agonistic GITR antibody.
- Suitable GITR antibodies include, for example, BMS-986153, BMS-986156, TRX-518 (WO06/105021, W009/009116) and MK-4166 (WO 11/028683).
- the immuno-oncology agent is an IDO antagonist.
- IDO antagonists include, for example, INCB-024360 (W02006/122150, WO07/75598, WO08/36653, WO08/36642), indoximod, BMS-986205, or NLG-919 (W009/73620, WO09/1156652, WO11/56652, WO12/142237).
- the immuno-oncology agent is an 0X40 agonist, such as an agonistic 0X40 antibody.
- Suitable 0X40 antibodies include, for example, MEDI-6383 or MEDI-6469.
- the immuno-oncology agent is an OX40L antagonist, such as an antagonistic 0X40 antibody.
- OX40L antagonists include, for example, RG-7888 (WO06/029879).
- the immuno-oncology agent is a CD40 agonist, such as an agonistic CD40 antibody.
- the immuno-oncology agent is a CD40 antagonist, such as an antagonistic CD40 antibody.
- Suitable CD40 antibodies include, for example, lucatumumab or dacetuzumab.
- the immuno-oncology agent is a CD47 antagonist, such as a CD47 antagonist selected from the group MIAP301, MIAP410, TTI-621, CV1, Hu5F9- G4, CC-90002, B6H12 and 2D3.
- a CD47 antagonist selected from the group MIAP301, MIAP410, TTI-621, CV1, Hu5F9- G4, CC-90002, B6H12 and 2D3.
- the immuno-oncology agent is a CD27 agonist, such as an agonistic CD27 antibody.
- Suitable CD27 antibodies include, for example, varlilumab.
- the immuno-oncology agent is MGA271 (to B7H3) (WO 11/109400).
- the present invention also provides the compounds of the present invention for use in therapy.
- the compounds of the present invention can be prepared in a number of ways well known to one skilled in the art of organic synthesis.
- the compounds of the present invention can be synthesized using the methods described below, together with synthetic methods known in the art of synthetic organic chemistry, or variations thereon as appreciated by those skilled in the art. Preferred methods include, but are not limited to, those described below. All references cited herein are hereby incorporated in their entirety by reference.
- the compounds of this invention may be prepared using the reactions and techniques described in this section.
- the reactions are performed in solvents appropriate to the reagents and materials employed and are suitable for the transformations being effected.
- all proposed reaction conditions including choice of solvent, reaction atmosphere, reaction temperature, duration of the experiment and work up procedures, are chosen to be the conditions standard for that reaction, which should be readily recognized by one skilled in the art. It is understood by one skilled in the art of organic synthesis that the functionality present on various portions of the molecule must be compatible with the reagents and reactions proposed. Such restrictions to the substituents that are compatible with the reaction conditions will be readily apparent to one skilled in the art and alternate methods must then be used.
- Preparation of compounds of Formula I and intermediates used in the preparation of compounds of Formula I can be prepared using procedures shown in the following Examples and related procedures. The methods and conditions used in these examples, and the actual compounds prepared in these Examples, are not meant to be limiting, but are meant to demonstrate how the compounds of Formula I can be prepared. Starting materials and reagents used in these examples, when not prepared by a procedure described herein, are generally either commercially available, or are reported in the chemical literature, or may be prepared by using procedures described in the chemical literature.
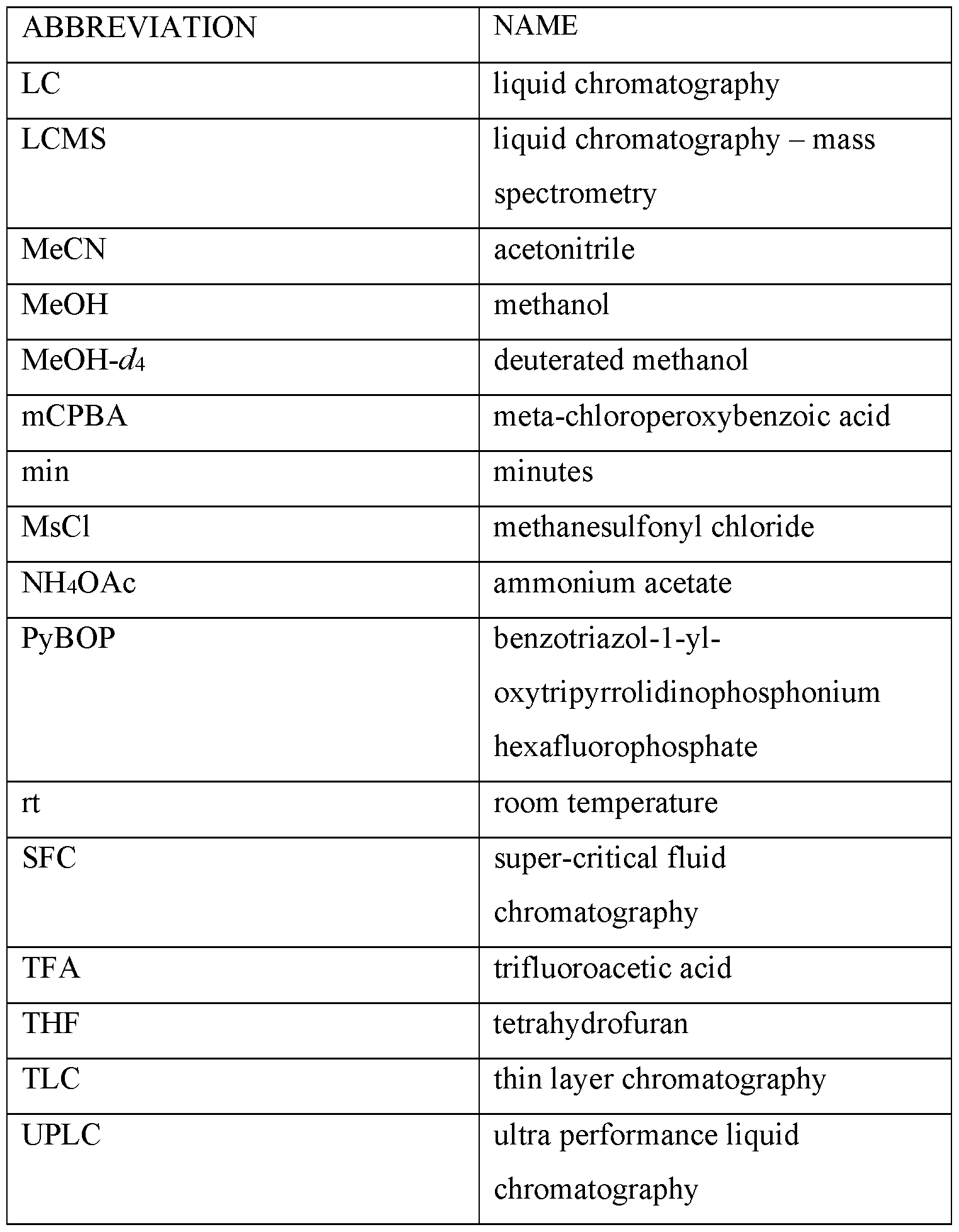
- Mobile phase A 5mm Ammonium formate pH 3.3: Acetonitrile (98:2)
- Mobile phase B ACN :5mm Ammonium formate pH 3.3 (98:2)
- Mobile phase B ACN :5mm Ammonium formate pH 3.3 (98:2)
- Mobile phase B ACN :5mm Ammonium formate pH 3.3 (98:2)
- Mobile phase B ACN :5mm Ammonium formate pH 3.3 (98:2)
- GRADIENT 0%B to 100%B over 1 min, stop time 1.5 min.
- GRADIENT 0-100 %B (0-3 min), 100 %B (3-3.5 min).
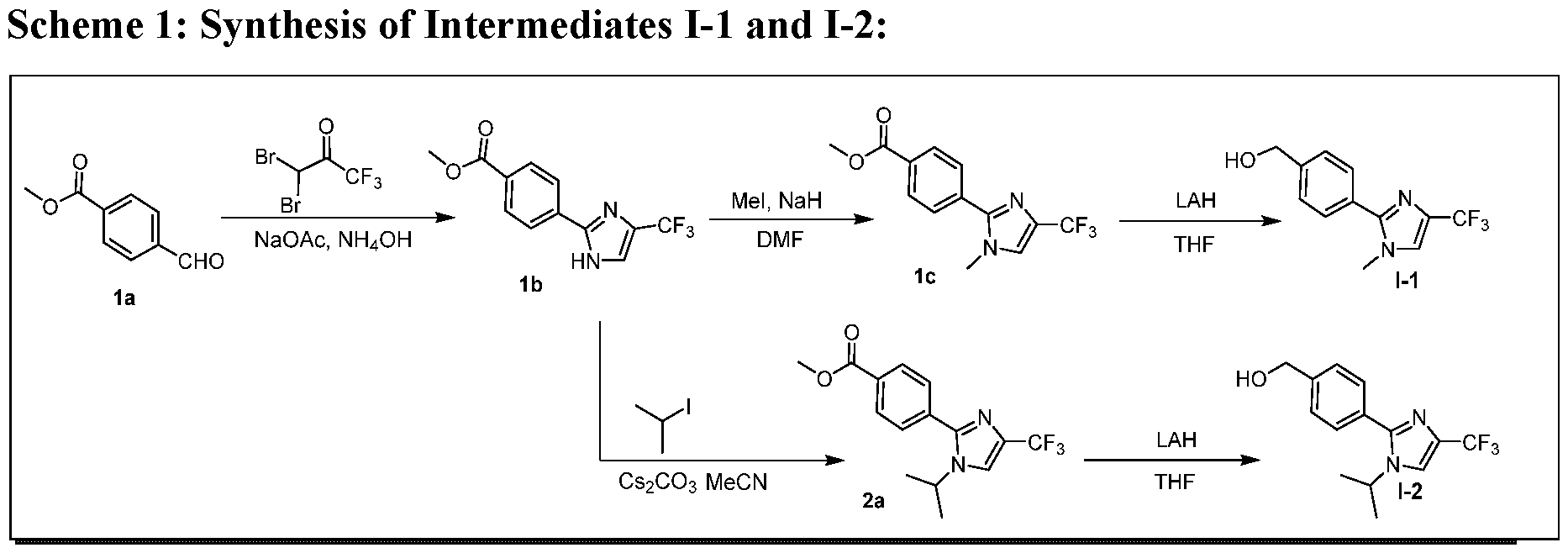
- Step-1 Methyl 4-(4-(trifluoromethyl)-LH-imidazol-2-yl )benzoate:
- Step-2 Methyl 4-(l-methyl-4-(trifluoromethyl)-lZ7-imidazol-2-yl)benzoate:
- Step-3 Synthesis of (4-(l-Methyl-4-(trifluoromethyl)-LH-imidazol-2- yl)phenyl)methanol (1-1):
- Step-1 Methyl 4-( l-isopropyl-4-(trifluoromethyl)-lZ/-imidazol-2-yl)benzoate: To a stirred solution of methyl 4-(4-(trifluoromethyl)- l7/-imidazol-2-yl)benzoate (2.0 g, 7.40 mmol) in acetonitrile (30 mL), CS2CO3 (4.82 g, 14.80 mmol) was added followed by addition of 2-iodopropane (1.110 mL, 11.10 mmol) at 0 °C.
- Step-2 Synthesis of (4-( l-Isopropyl-4-(trifluoromethyl)-lZ/-imidazol-2- yl)phenyl)methanol (1-2):
- Step-1 4-chloro-l-isopropyl-lH-pyrazole: To a stirred solution of 4-chloro-lH-pyrazole (5.0 g, 48.8 mmol) in acetonitrile (60 mL), CS2CO3 (31.8 g, 98 mmol) was added followed by addition of 2-iodopropane (5.69 mL, 58.5 mmol) and the contents heated at 80 °C for 2 h. The reaction mixture was cooled to ambient temperature, diluted with water and extracted with diethyl ether.
- Step-2 Synthesis of 4-chloro-l-isopropyl-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan- 2-yl)-lH-pyrazole (1-4):
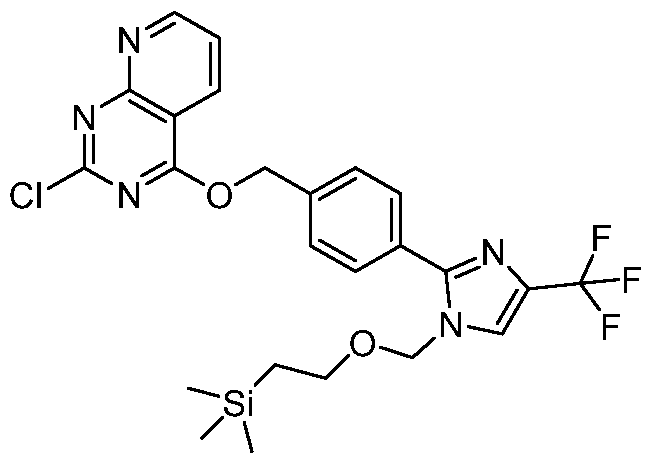
- Step-1 Synthesis of 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)pyrido [3, 2-d] pyrimidine: To a stirred solution of 2,4-dichloropyrido[3,2-d]pyrimidine (0.200 g, 1.00 mmol) in acetonitrile (10 mL), CS2CO3 (0.489 g, 1.50 mmol) and (4-(l-methyl-4-(trifluoromethyl)- lH-imidazol-2-yl)phenyl)methanol (0.256 g, 1.00 mmol) were added and the mixture was heated at 60 °C for 16 h.
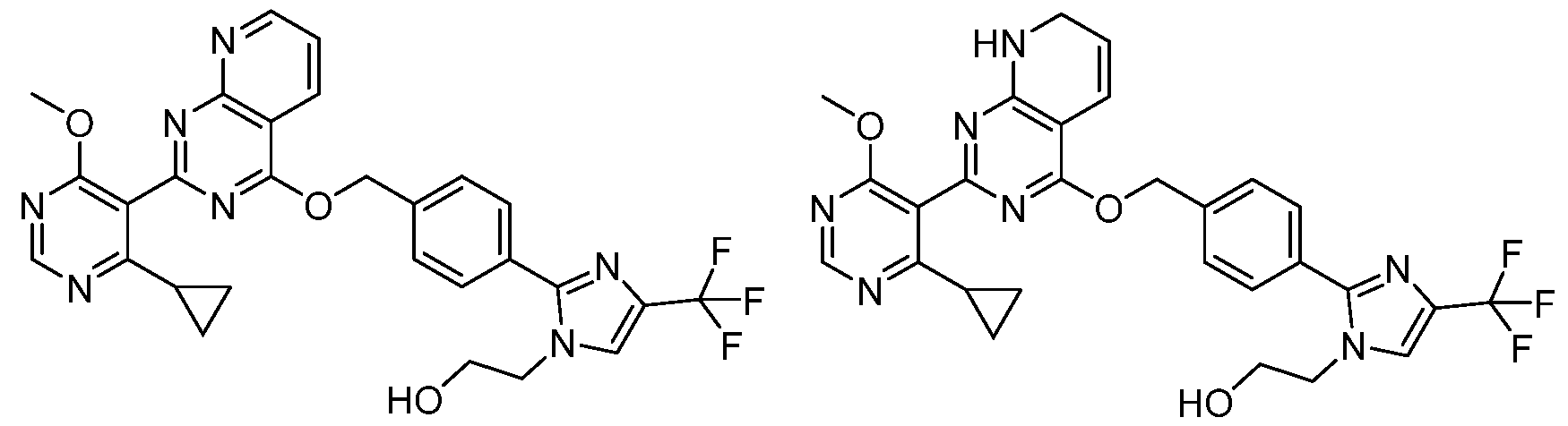
- Example 26 and 27 Chiral resolution of example 22:
- reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate.
- reaction was stirred at 62 °C under N2 for 10 min, then treated with a solution of 2,4- dichloropyrido[3,2-d]pyrimidine (500 mg, 2.500 mmol) in THF (5 mL) and tetrakis (289 mg, 0.250 mmol). The mixture was stirred at 65 °C for 2 h. After completion of reaction (monitored by UPLC), the reaction mixture was filtered through celite bed and Concentrated under reduced pressure. The crude was purified by column chromatography using 60-120 silica, eluted with 0-50% ethylacetate in petether.
- Example 31 and 32 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)pyrido [3,2-d] pyrimidine and (2-(4- cyclopropyl-6-methoxypyrimidin-5-yl)pyrido[3,2-d]pyrimidin-4-yl)(4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)phenyl)methanone:
- reaction mixture was heated at 90 °C for 4 h. After completion of reaction (monitored by UPLC), the reaction mixture was concentrated by using rota evaporator and extracted with ethylacetate (2X 50 mL). The organic layer was separated and dried over Na2SO4 and concentated by using rota evaporator. The crude was purified by column chromatography using 60-120 silica, eluted with 0-50% ethylacetate in petether.
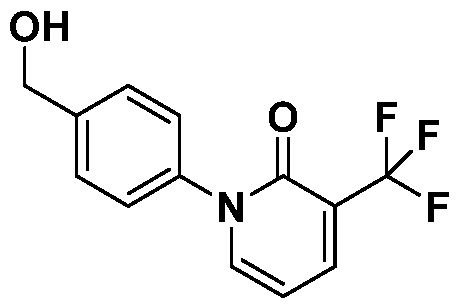
- Step B l-(4-(hydroxymethyl)phenyl)-3-(trifluoromethyl)pyridin-2(lH)-one
- Step A methyl 2-(4-(hydroxymethyl)phenyl)-l-methyl-lH-imidazole-4-carboxylate
- Step B 2-(2-(4-(hydroxymethyl)phenyl)- 1 -methyl- lH-imidazol-4-yl)propan-2-ol :
- Example 50 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)pyrido[3,2- d]pyrimidin-4-yl)oxy)methyl)phenyl)-l-methyl-lH-imidazol-4-yl)propan-2-ol:
- Step A 2-(2-(4-(((2-chloropyrido[3,2-d]pyrimidin-4-yl)oxy)methyl)phenyl)-l -methyl- lH-imidazol-4-yl)propan-2-ol:
- Step B 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)pyrido[3,2-d]pyrimidin-4- yl)oxy)methyl)phenyl)- 1 -methyl- lH-imidazol-4-yl)propan-2-ol :
- Procedure B To a stirred solution of methyl 4-(4-(trifluoromethyl)-17/-imidazol-2- yl)benzoate (2.0 g, 7.40 mmol) in acetonitrile (30 mL), CS2CO3 (4.82 g, 14.80 mmol) was added followed by addition of 2-iodopropane (1.110 mL, 11.10 mmol) at 0 °C and the mixture was stirred at 25 °C for 16 h. After that, reaction mixture was heated at 50 °C for additional 16 h. After completion of the reaction (monitored by TLC), volatiles were removed under reduced pressure, diluted with ethyl acetate and washed with water.
- reaction mixture was again cooled to 0 °C, chilled saturated ammonium chloride solution was added and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure.
- reaction mixture was heated at 120 °C for 2 h under MW irradiation. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate.
- reaction mixture was heated at 120 °C for 2 h under MW irradiation. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate.
- reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by column chromatography on silica gel, 230-400 mesh, to obtain 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(4- (tri fluoromethyl)- l-((2-(trimethylsilyl)ethoxy)methyl)-lH-imidazol-2- yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (300 mg, 0.462 mmol, 70.7 % yield) as yellow gum.
- Example 65 Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(l-(4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)phenyl)ethoxy)pyrido[2,3- djpyrimidine:
- Example 67 Synthesis of methyl 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5- yl)pyrido[2,3-d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH-imidazol- l-yl)acetate: To a stirred solution of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (150 mg, 0.289 mmol) in acetonitrile (10 mL), cesium carbonate (188 mg, 0.577 mmol) and methyl 2- bromoacetate (0.041 mL, 0.433 mmol) were added and reaction mixture was heated at 80 °C for 16 h.
- Example 70 Synthesis of 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5- yl)pyrido[2,3-d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH-imidazol- l-yl)-N,N-dimethylacetamide:
- Example 71 and 72 Synthesis of 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5- yl)pyrido[2,3-d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH-imidazol- l-yl)ethan-l-ol and 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-7,8- dihydropyrido[2,3-d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH- imidazol-l-yl)ethan-l-ol:
- Example 73 Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)quinazoline: To a stirred solution of 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)quinazoline (120 mg, 0.287 mmol) in a mixture of 1,4-dioxane and water (10 mL, 9: 1), potassium phosphate tribasic (122 mg, 0.573 mmol), 4-cyclopropyl-6- methoxy-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)pyrimidine (87 mg, 0.315 mmol) and tetrakis(triphenylphosphine)palladium
- Example 80 Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-6-fluoro-4-((4- (l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)quinazoline:
- reaction mixture was quenched with NH4Q and extracted with ethyl acetate.
- the combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure.
- Example 81 Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-7,8-dihydropyrido[4,3- d]pyrimidin-5(6H)-one:
- tert-butyl 2,4-dichloro-5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)- carboxylate 300 mg, 0.943 mmol
- tetrakis(triphenylphosphine)palladium(0) 109 mg, 0.094 mmol
- the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate.
- reaction mixture was heated at 110 °C for 16 h. After completion of reaction (monitored by UPLC-MS and TLC), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate.
- Example 82 Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(4-(l-methyl- 4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)-7,8-dihydropyrido[4,3-d]pyrimidin- 5(6H)-one:
- Example 83 Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)-7,8-dihydropyrido[4,3- d]pyrimidin-5(6H)-one:
- reaction mixture was purged with nitrogen gas and then stirred at 45 °C for 4 h under nitrogen atmosphere. After completion of reaction (monitored by UPLC-MS and TLC), the reaction mixture was cooled to ambient temperature, mixed with another batch of the same reaction (with 0.200 g of 2-chloro-6-((4-(l-methyl-4-(trifluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)-5-nitropyrimidin-4-amine), chilled water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure.
- Example 84 Synthesis of 2-(4-chloro-l-isopropyl-lH-pyrazol-5-yl)-4-((4-(l-methyl- 4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pteridin-7(8H)-one:
- USP1/UAF1 ubiquitin-rhodamine 110 hydrolysis assays were performed at room temperature in black, low-volume 384 well plates (Corning 3821). 100X solutions of compounds in DMSO were prepared by three-fold serial dilutions starting from a 10 mM stock. 2X solutions of His6-USPl/His6-UAFl (200 pM, Internally Produced) and ubiquitin-rhodamine 110 (10 pM, South Bay Bio SBB-PS0001) were prepared in assay buffer (50 mM Tris pH 7.5, 100 mM NaCl, 1 mM EDTA, 1 mM TCEP, 100 ng/pL BSA).
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Oncology (AREA)
- Hematology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Compounds having the following formula I or a stereoisomer or pharmaceutically acceptable salt thereof, where all substituents are as defined herein, are inhibitors of USP1 useful for treating diseases including, among others, treating proliferative, metabolic, allergic, autoimmune and inflammatory diseases.
Description
UBIQUITIN SPECIFIC PROCESSING PROTEASE 1 (USP1) COMPOUNDS
CROSS REFERENCE TO RELATED APPLICATIONS
This application claims the benefit of U.S. Provisional Application No. 63/594,458, filed, October 31, 2023, the entire content of which is hereby incorporated herein by reference.
FIELD
This invention relates to compounds which are inhibitors of ubiquitin-specific- processing protease 1 (USP1) useful for treating diseases including, among others, cancer, autoimmune and inflammatory disorders. The invention further pertains to pharmaceutical compositions containing at least one compound according to the invention that are useful for the treatment of conditions related to the inhibition of USP1 in a mammal.
BACKGROUND OF THE INVENTION
Ubiquitination is important in the regulation of many cellular functions and cellular homeostasis. The conjugation of ubiquitin to a target protein is a multistep process involving the sequential action of a ubiquitin activating enzyme (El), a ubiquitin- conjugating enzyme (E2), and a ubiquitin protein-ligase (E3). The ubiquitin tags can mediate non-covalent interactions of the ubiquitinated substrate with other proteins bearing different types of ubiquitin-binding motifs. A family of enzymes, termed deubiquitinases act on ubiquitinated substrates to catalyze the removal of ubiquitin moieties. One such enzyme is ubiquitin-specific protease 1 (USP1) which plays an important role in the regulation of DNA repair processes. USP1 is a regulator of several important steps in the DNA damage response, particularly in the Fanconi anemia pathway, and in the process of translesion synthesis. USP1 has also been reported to contribute to the repair of double-strand DNA breaks through homologous recombination. In addition, USP1 has been reported to deubiquitinate and stabilize members of the family of inhibitors of DNA binding (ID) proteins, ID1, ID2 and ID3. Garcia-Santisteban, I.,
Peters, G.J, Giovannetti, E. et al. Mol Cancer 12, 91 (2013); US Patent Nos. 7,754,463, 10,653,676, 9,518,032.
SUMMARY
The present disclosure provides compounds that modulate the expression or activity of USP1. The disclosure also provides compositions, including pharmaceutical compositions, kits that include the compounds, and methods of using (or administering) and making the compounds. The compounds provided herein are useful in treating diseases, disorders, or conditions that are mediated by USP1. The disclosure also provides compounds for use in therapy. The disclosure further provides compounds for use in a method of treating a disease, disorder, or condition that is mediated by USP1. Moreover, the disclosure provides uses of the compounds in the manufacture of a medicament for the treatment of a disease, disorder or condition that is mediated by (or mediated, at least in part, by) USP1.
Formula (I) or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof;
Wherein, R1 is selected from Ce aryl and 5-6 membered heteroaryl, optionally substituted with one to four halo, hydroxy, amino, -C(O)Ra, -C(O)ORb, -C(O)NRaRb, -N(Ra)C(O)Rb, -S(O)NRaRb, -S(O)2NRaRb, -S(O)Rg, -S(O)2Rg, -NRaRb, -ORa, -SRb, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl and C3-8 cycloalkyl; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl and C3-8 cycloalkyl is optionally substituted with one to four R100;
R2 is selected from absent, hydrogen, halo, hydroxy, amino, -CN, -C(O)Ra, -C(O)ORb, - C(O)NRaRb, -N(Ra)C(O)Rb, -N(Ra)C(O)NRaRb, -N(Ra)SO2NRaRb, -S(O)NRaRb, - S(O)2NRaRb, -N(Ra)S(O)2Rb, -S(O)Rg, -S(O)2Rg, -NRaRb, -ORa, -SRb, -OC(O)Ra, - OC(O)NRaRb Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S, and C3-8 cycloalkyl is optionally substituted with one to four R100;
X is selected from absent and C1-6 alkyl, wherein C1-6 alkyl is optionally substituted with one to four R100;
Y is absent or -O-;
Each W1, W2, W3 and W4 is independently selected from -N(Ra)-, -C(O)- and -C(Ra)-;
W5 is selected from -N- and -C(Ra)-; wherein at least one of W1, W2, W3 and W4 is -C(Ra)- or -C(O)-;
G1 is selected from -Ce aryl-, 5-6 membered heteroaryl, C3-8 cycloalkyl and 5-6 membered heterocyclyl; wherein each Ce aryl, 5-6 membered heteroaryl, C3-8 cycloalkyl and 5-6 membered heterocyclyl is optionally substituted with one to four R100;
G2 is a 5 or 6 membered heteroaryl or 5-6 membered heterocyclyl optionally substituted with one to four R100; each Ra and Rb is independently selected from absent, hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-6 cycloalkyl and 4-6 membered heterocyclyl ; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-6 cycloalkyl and 4-6 membered heterocyclyl is optionally substituted with one to four R200; each R100 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)RC, -C(O)ORC, -C(O)NRcRd, -N(Rc)C(O)Rd, -S(O)NRcRd, - S(O)2NRcRd, -S(O)Rh, -S(O)2Rh, -NRcRd, -ORC, -SRC, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl and 4-10 membered heterocyclyl is optionally substituted with one to four R201;
each Rc and Rd is independently selected from hydrogen, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; each R200 and R201 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)Re, -C(O)ORe, -C(O)NReRf, -N(Re)C(O)Rf, -S(O)NReRf, - S(O)2NReRf, -S(O)R‘, -8(0)2^, -NReRf, -ORe, -SRe, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl and 4-10 membered heterocyclyl is optionally substituted with one to four R300; each Rg, Rh and R1 is independently selected from C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, is optionally substituted with one to four R300; wherein each R300 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)Re, -C(O)ORe, -C(O)NReRf, -N(Re)C(O)Rf, -S(O)NReRf, - S(O)2NReRf, -NReRf, S(O)Re, -S(O)2Re, -NReRf, -ORe, -SRe, C1-6 alkyl, C2-6 alkenyl and C2-6 alkynyl; each Re and Rf is independently selected from hydrogen, C1-6 alkyl, C2-6 alkenyl and C2-6 alkynyl; C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl and 4-10 membered heterocyclyl is optionally substituted with one to four R400; each R400 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)Rk, -C(O)ORk, -C(O)NRkR1, -N(Rk)C(O)R*, -S(O)NRkR1, - S(O)2NRkR1, -NRkR‘, S(O)Rk, -S(O)2Rk, -NRkR‘, -ORk, -SRk, C1-6 alkyl, C2-6 alkenyl and C2-6 alkynyl; each Rk and R1 is independently selected from hydrogen, C1-6 alkyl, C2-6 alkenyl and C2-6 alkynyl; C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4
heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S.
In one aspect, provided are processes and intermediates for making the compounds of Formula I.
In one aspect, provided are pharmaceutical compositions comprising a pharmaceutically acceptable carrier and at least one of the compounds disclosed herein.
The present application also provides methods for the inhibition of USP1 comprising administering a therapeutically effective amount of at least one of Formula I.
The present application also provides a method for treating proliferative, metabolic, allergic, autoimmune and inflammatory diseases, comprising administering to a host in need of such treatment a therapeutically effective amount of at least one of the compounds disclosed herein. The compounds of Formula I or a pharmaceutically acceptable salt thereof, may be used to treat cancers that are mediated by, dependent on or associated with USP1 activity. In certain embodiments, the disease is a solid tumor.
DETAILED DESCRIPTION
In a first aspect, provided are compounds of formula (I) that function as inhibitors ofUSPl :
or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof;
Wherein, R1 is selected from Ce aryl and 5-6 membered heteroaryl, optionally substituted with one to four halo, hydroxy, amino, -C(O)Ra, -C(O)ORb, -C(O)NRaRb, -N(Ra)C(O)Rb, -S(O)NRaRb, -S(O)2NRaRb, -S(O)Rg, -S(O)2Rg, -NRaRb, -ORa, -SRb, Ci-6 alkyl, C2-6
alkenyl, C2-6 alkynyl and C3-8 cycloalkyl; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl and C3-8 cycloalkyl is optionally substituted with one to four R100;
R2 is selected from absent, hydrogen, halo, hydroxy, amino, -CN, -C(O)Ra, -C(O)ORb, - C(O)NRaRb, -N(Ra)C(O)Rb, -N(Ra)C(O)NRaRb, -N(Ra)SO2NRaRb, -S(O)NRaRb, - S(O)2NRaRb, -N(Ra)S(O)2Rb, -S(O)Rg, -S(O)2Rg, -NRaRb, -ORa, -SRb, -OC(O)Ra, - OC(O)NRaRb C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S, and C3-8 cycloalkyl is optionally substituted with one to four R100;
X is selected from absent and C1-6 alkyl, wherein C1-6 alkyl is optionally substituted with one to four R100;
Y is absent or -O-;
Each W1, W2, W3 and W4 is independently selected from -N(Ra)-, -C(O)- and -C(Ra)-;
W5 is selected from -N- and -C(Ra)-; wherein at least one of W1, W2, W3 and W4 is -C(Ra)- or -C(O)-;
G1 is selected from -Ce aryl-, 5-6 membered heteroaryl, C3-8 cycloalkyl and 5-6 membered heterocyclyl; wherein each Ce aryl, 5-6 membered heteroaryl, C3-8 cycloalkyl and 5-6 membered heterocyclyl is optionally substituted with one to four R100;
G2 is a 5 or 6 membered heteroaryl or 5-6 membered heterocyclyl optionally substituted with one to four R100; each Ra and Rb is independently selected from absent, hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-6 cycloalkyl and 4-6 membered heterocyclyl; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-6 cycloalkyl and 4-6 membered heterocyclyl is optionally substituted with one to four R200; each R100 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)RC, -C(O)ORC, -C(O)NRcRd, -N(Rc)C(O)Rd, -S(O)NRcRd, - S(O)2NRcRd, -S(O)Rh, -S(O)2Rh, -NRcRd, -ORC, -SRC, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl,
C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl and 4-10 membered heterocyclyl is optionally substituted with one to four R201; each Rc and Rd is independently selected from hydrogen, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; each R200 and R201 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)Re, -C(O)ORe, -C(O)NReRf, -N(Re)C(O)Rf, -S(O)NReRf, - S(O)2NReRf, -S(O)R‘, -S(O)2Ri, -NReRf, -ORe, -SRe, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl and 4-10 membered heterocyclyl is optionally substituted with one to four R300; each Rg, Rh and R1 is independently selected from C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, is optionally substituted with one to four R300; wherein each R300 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)Re, -C(O)ORe, -C(O)NReRf, -N(Re)C(O)Rf, -S(O)NReRf, - S(O)2NReRf, -NReRf, S(O)Re, -S(O)2Re, -NReRf, -ORe, -SRe, C1-6 alkyl, C2-6 alkenyl and C2-6 alkynyl; each Re and Rf is independently selected from hydrogen, C1-6 alkyl, C2-6 alkenyl and C2-6 alkynyl; C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl and 4-10 membered heterocyclyl is optionally substituted with one to four R400; each R400 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)Rk, -C(O)ORk, -C(O)NRkR1, -N(Rk)C(O)R*, -S(O)NRkR1, - S(O)2NRkR1, -NRkR‘, S(O)Rk, -S(O)2Rk, -NRkR‘, -ORk, -SRk, C1-6 alkyl, C2-6 alkenyl and C2-6 alkynyl;
each Rk and R1 is independently selected from hydrogen, Ci-6 alkyl, C2-6 alkenyl and C2-6 alkynyl; C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S.
Formula (II) or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof.
Formula (Illa) Formula (Illb)
or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof.
In one embodiment, provided are compounds of Formula (IVa), (IVb) or (IVc):
or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof; wherein R5 is Ci-6 alkyl.
In one embodiment, provided are compounds of Formula (Va), (Vb) or (Vc):
or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof; wherein R5 is Ci-6 alkyl.
Formula (Via) Formula (VIb) or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof; wherein R5 is Ci-6 alkyl.
Formula (Vile)
In one embodiment, provided are compounds of Formula (Villa) (Vlllb) or (VIIIc):
or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof; wherein R5 is Ci-6 alkyl.
In one embodiment, provided are compounds wherein R1 is selected from:
or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof. In one embodiment, provided are compounds wherein R2 is selected from:
-OCH3, -H, -SCH3, -S(O)2CH3, -S(O)2CH3, -C(O)OCH3, -C(O)OCH2CH3, and -C(O)NH2. or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers, or deuterated analog thereof. In one embodiment, provided are compounds wherein G2 is selected from:
or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers, or deuterated analog thereof.
In one embodiment, provided are compounds of Table A or a pharmaceutically acceptable salt thereof;
TABLE A
The compounds described herein (e.g., a compound of Formula I, II, Illa, Illb, inc, Illd, me, IVa, IVb, IVc, Va, Vb, Vc, Via, VIb, Vila, Vllb, Vile, Villa, Vlllb, VIIIc or Table A) or a pharmaceutically acceptable salt thereof, may be used to treat cancers that are mediated by, dependent on or associated with USP1 activity. In certain embodiments, the disease is a solid tumor. In particular embodiments, the solid tumor is selected from prostate cancer, pancreatic cancer, bladder cancer, colorectal cancer, breast cancer, renal cancer, hepatocellular cancer, lung cancer, ovarian cancer, cervical cancer, gastric cancer, esophageal cancer, head and neck cancer, melanoma, neuroendocrine cancers, CNS cancers, brain tumors (e.g., glioma, anaplastic oligodendroglioma, adult glioblastoma multiforme, and adult anaplastic astrocytoma), bone cancer, or soft tissue sarcoma. In some embodiments, the solid tumor is from non-small cell lung cancer or small-cell lung cancer.
The following are definitions of terms used in this specification and appended claims. The initial definition provided for a group or term herein applies to that group or term throughout the specification and claims, individually or as part of another group, unless otherwise indicated.
As used in the present specification, the following words, phrases and symbols are generally intended to have the meanings as set forth below, except to the extent that the context in which they are used indicates otherwise.
Compounds of this invention may have one or more asymmetric centers. Unless otherwise indicated, all chiral (enantiomeric and diastereomeric) and racemic forms of compounds of the present invention are included in the present invention. Many geometric isomers of olefins, C=N double bonds, and the like can also be present in the compounds, and all such stable isomers are contemplated in the present invention. Cis- and /ra//.s-geometric isomers of the compounds of the present invention are described and may be isolated as a mixture of isomers or as separated isomeric forms. The present compounds can be isolated in optically active or racemic forms. It is well known in the art how to prepare optically active forms, such as by resolution of racemic forms or by synthesis from optically active starting materials. All chiral, (enantiomeric and diastereomeric) and racemic forms and all geometric isomeric forms of a structure are intended, unless the specific stereochemistry or isomer form is specifically indicated.
When any variable (e.g., R3) occurs more than one time in any constituent or formula for a compound, its definition at each occurrence is independent of its definition at every other occurrence. Thus, for example, if a group is shown to be substituted with 0-2 R3, then said group may optionally be substituted with up to two R3 groups and R3 at each occurrence is selected independently from the definition of R3. Also, combinations of substituents and/or variables are permissible only if such combinations result in stable compounds.
When a bond to a substituent is shown to cross a bond connecting two atoms in a ring, then such substituent may be bonded to any atom on the ring. When a substituent is listed without indicating the atom via which such substituent is bonded to the rest of the compound of a given formula, then such substituent may be bonded via any atom in such substituent. Combinations of substituents and/or variables are permissible only if such combinations result in stable compounds.
In cases wherein there are nitrogen atoms (e.g., amines) on compounds of the present invention, these can be converted to N-oxides by treatment with an oxidizing agent (e.g., MCPBA and/or hydrogen peroxides) to afford other compounds of this invention. Thus, all shown and claimed nitrogen atoms are considered to cover both the shown nitrogen and its N-oxide (N— >0) derivative.
In accordance with a convention used in the art,
is used in structural formulas herein to depict the bond that is the point of attachment of the moiety or substituent to the core or backbone structure.
A dash that is not between two letters or symbols is used to indicate a point of attachment for a substituent. For example, -CONH2 is attached through the carbon atom. A dash at the front or end of a chemical group is a matter of convenience; chemical groups may be depicted with or without one or more dashes without losing their ordinary meaning. A wavy line drawn through a line in a structure indicates a point of attachment of a group. Unless chemically or structurally required, no directionality is indicated or implied by the order in which a chemical group is written or named.
The term "optionally substituted" in reference to a particular moiety of the compound of Formula I (e.g., an optionally substituted heteroaryl group) refers to a moiety having 0, 1, 2, or more substituents. For example, "optionally substituted alkyl" encompasses both "alkyl" and "substituted alkyl" as defined below. It will be understood by those skilled in the art, with respect to any group containing one or more substituents,
that such groups are not intended to introduce any substitution or substitution patterns that are sterically impractical, synthetically non-feasible and/or inherently unstable.
As used herein, the term "at least one chemical entity" is interchangeable with the term "a compound".
The prefix "Cu-v" indicates that the following group has from u to v carbon atoms. For example, "Ci-6 alkyl" indicates that the alkyl group has from 1 to 6 carbon atoms.
As used herein, the term "alkyl" or "alkylene" is intended to include both branched and straight-chain saturated aliphatic hydrocarbon groups having the specified number of carbon atoms. For example, "Ci-io alkyl" (or alkylene), is intended to include Ci, C2, C3, C4, Cs, Ce, C7, Cs, C9, and C10 alkyl groups. Additionally, for example, "Ci-Ce alkyl" denotes alkyl having 1 to 6 carbon atoms. Alkyl groups can be unsubstituted or substituted so that one or more of its hydrogens are replaced by another chemical group. Example alkyl groups include, but are not limited to, methyl (Me), ethyl (Et), propyl (e.g., n-propyl and isopropyl), butyl (e.g., n-butyl, isobutyl, /-butyl), pentyl (e.g., n-pentyl, isopentyl, neopentyl), and the like.
"Alkenyl" or "alkenylene" is intended to include hydrocarbon chains of either straight or branched configuration and having one or more double carbon-carbon bonds that may occur in any stable point along the chain. For example, "C2-6 alkenyl" (or alkenylene), is intended to include C2, C3, C4, Cs, and Ce alkenyl groups. Examples of alkenyl include, but are not limited to, ethenyl, 1 -propenyl, 2-propenyl, 2-butenyl, 3-butenyl, 2-pentenyl, 3- pentenyl, 4-pentenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-hexenyl, 2-methyl-2-propenyl, 4- methyl-3 -pentenyl, and the like.
"Alkynyl" or "alkynylene" is intended to include hydrocarbon chains of either straight or branched configuration and having one or more triple carbon-carbon bonds that may occur in any stable point along the chain. For example, "C2-6 alkynyl" (or alkynylene), is intended to include C2, C3, C4, Cs, and Ce alkynyl groups; such as ethynyl, propynyl, butynyl, pentynyl, hexynyl and the like.
One skilled in the field will understand that, when the designation "CO2" is used o II herein, this is intended to refer to the group c o
When the term "alkyl" is used together with another group, such as in "arylalkyl", this conjunction defines with more specificity at least one of the substituents that the substituted alkyl will contain. For example, "arylalkyl" refers to a substituted alkyl group
as defined above where at least one of the substituents is an aryl, such as benzyl. Thus, the term aryl(Co-4)alkyl includes a substituted lower alkyl having at least one aryl substituent and also includes an aryl directly bonded to another group, z.e., aryl(Co)alkyl. The term "heteroarylalkyl" refers to a substituted alkyl group as defined above where at least one of the substituents is a heteroaryl.
When reference is made to a substituted alkenyl, alkynyl, alkylene, alkenylene, or alkynylene group, these groups are substituted with one to three substituents as defined above for substituted alkyl groups.
The term "alkoxy" refers to an oxygen atom substituted by alkyl or substituted alkyl, as defined herein. For example, the term "alkoxy" includes the group -O-Ci-ealkyl such as methoxy, ethoxy, propoxy, isopropoxy, n-butoxy, sec-butoxy, te/7-butoxy, pentoxy, 2-pentyloxy, isopentoxy, neopentoxy, hexoxy, 2-hexoxy, 3-hexoxy, 3- methylpentoxy, and the like. "Lower alkoxy" refers to alkoxy groups having one to four carbons.
The term "cycloalkyl" refers to cyclized alkyl groups, including mono-, bi- or polycyclic ring systems. C3-7 cycloalkyl is intended to include C3, C4, Cs, Ce, and C7 cycloalkyl groups. Example cycloalkyl groups include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, norbornyl, and the like. As used herein, "carbocycle" or "carbocyclic residue" is intended to mean any stable 3-, 4-, 5-, 6-, or 7-membered monocyclic or bicyclic or 7-, 8-, 9-, 10-, 11-, 12-, or 13-membered bicyclic or tricyclic ring, any of which may be saturated, partially unsaturated, unsaturated or aromatic. Examples of such carbocycles include, but are not limited to, cyclopropyl, cyclobutyl, cyclobutenyl, cyclopentyl, cyclopentenyl, cyclohexyl, cycloheptenyl, cycloheptyl, cycloheptenyl, adamantyl, cyclooctyl, cyclooctenyl, cyclooctadienyl, [3.3.0]bicyclooctane, [4.3.0]bicyclononane, [4.4.0]bicyclodecane, [2.2.2]bicyclooctane, fluorenyl, phenyl, naphthyl, indanyl, adamantyl, anthracenyl, and tetrahydronaphthyl (tetralin). As shown above, bridged rings are also included in the definition of carbocycle (e.g., [2.2.2]bicyclooctane). Preferred carbocycles, unless otherwise specified, are cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, and phenyl. When the term "carbocycle" is used, it is intended to include "aryl". A bridged ring occurs when one or more carbon atoms link two non-adjacent carbon atoms. Preferred bridges are one or two carbon atoms. It is noted that a
bridge always converts a monocyclic ring into a bicyclic ring. When a ring is bridged, the substituents recited for the ring may also be present on the bridge.
The term "aryl" refers to monocyclic or bicyclic aromatic hydrocarbon groups having 6 to 12 carbon atoms in the ring portion, such as phenyl, and naphthyl groups, each of which may be substituted.
Accordingly, in compounds of formula I, the term "cycloalkyl" includes cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, bicyclooctyl, etc., as well as the following ring systems:
and the like, which optionally may be substituted at any available atoms of the ring(s).
The term "halo" or "halogen" refers to chloro, bromo, fluoro and iodo.
The term "haloalkyl" means a substituted alkyl having one or more halo substituents. For example, "haloalkyl" includes mono, bi, and trifluoromethyl.
The term "haloalkoxy" means an alkoxy group having one or more halo substituents. For example, "haloalkoxy" includes OCF3.
The terms "heterocycle", "heterocycloalkyl", "heterocyclo", "heterocyclic", or "heterocyclyl" may be used interchangeably and refer to substituted and unsubstituted 3- to 7-membered monocyclic groups, 7- to 11 -membered bicyclic groups, and 10- to 15- membered tricyclic groups, in which at least one of the rings has at least one heteroatom (O, S or N), said heteroatom containing ring preferably having 1, 2, or 3 heteroatoms selected from O, S, and N. Each ring of such a group containing a heteroatom can contain one or two oxygen or sulfur atoms and/or from one to four nitrogen atoms provided that the total number of heteroatoms in each ring is four or less, and further provided that the ring contains at least one carbon atom. The nitrogen and sulfur atoms may optionally be oxidized and the nitrogen atoms may optionally be quaternized. The fused rings completing the bicyclic and tricyclic groups may contain only carbon atoms and may be saturated, partially saturated, or fully unsaturated. The heterocyclo group may be attached at any available nitrogen or carbon atom. As used herein the terms
"heterocycle", "heterocycloalkyl", "heterocyclo", "heterocyclic", and "heterocyclyl" include "heteroaryl" groups, as defined below.
In addition to the heteroaryl groups described below, exemplary monocyclic heterocyclyl groups include azetidinyl, pyrrolidinyl, oxetanyl, imidazolinyl, oxazolidinyl, isoxazolinyl, thiazolidinyl, isothiazolidinyl, tetrahydrofuranyl, piperidyl, piperazinyl, 2- oxopiperazinyl, 2-oxopiperidyl, 2-oxopyrrolodinyl, 2-oxoazepinyl, azepinyl, 1-pyridonyl, 4-piperidonyl, tetrahydropyranyl, morpholinyl, thiamorpholinyl, thiamorpholinyl sulfoxide, thiamorpholinyl sulfone, 1,3-dioxolane and tetrahydro- 1,1 -di oxothienyl and the like. Exemplary bicyclic heterocyclo groups include quinuclidinyl.
The term "heteroaryl" refers to substituted and unsubstituted aromatic 5- or 6- membered monocyclic groups, 9- or 10-membered bicyclic groups, and 11- to 14- membered tricyclic groups which have at least one heteroatom (O, S or N) in at least one of the rings, said heteroatom-containing ring preferably having 1, 2, or 3 heteroatoms selected from O, S, and N. Each ring of the heteroaryl group containing a heteroatom can contain one or two oxygen or sulfur atoms and/or from one to four nitrogen atoms provided that the total number of heteroatoms in each ring is four or less and each ring has at least one carbon atom. The fused rings completing the bicyclic and tricyclic groups may contain only carbon atoms and may be saturated, partially saturated, or unsaturated. The nitrogen and sulfur atoms may optionally be oxidized and the nitrogen atoms may optionally be quaternized. Heteroaryl groups which are bicyclic or tricyclic must include at least one fully aromatic ring but the other fused ring or rings may be aromatic or nonaromatic. The heteroaryl group may be attached at any available nitrogen or carbon atom of any ring. As valence allows, if said further ring is cycloalkyl or heterocyclo it is additionally optionally substituted with =0 (oxo).
Exemplary monocyclic heteroaryl groups include pyrrolyl, pyrazolyl, pyrazolinyl, imidazolyl, oxazolyl, isoxazolyl, thiazolyl, thiadiazolyl, isothiazolyl, furanyl, thienyl, oxadiazolyl, pyridyl, pyrazinyl, pyrimidinyl, pyridazinyl, triazinyl and the like.
Exemplary bicyclic heteroaryl groups include indolyl, benzothiazolyl, benzodi oxolyl, benzoxazolyl, benzothienyl, quinolinyl, tetrahydroisoquinolinyl, isoquinolinyl, benzimidazolyl, benzopyranyl, indolizinyl, benzofuranyl, chromonyl, coumarinyl, benzopyranyl, cinnolinyl, quinoxalinyl, indazolyl, pyrrol opyridyl, furopyridyl, dihydroisoindolyl, tetrahydroquinolinyl and the like.
Exemplary tricyclic heteroaryl groups include carbazolyl, benzindolyl, phenanthrollinyl, acridinyl, phenanthridinyl, xanthenyl and the like.
In compounds of formula I, preferred heteroaryl groups include:
be substituted at any available carbon or nitrogen atom.
Unless otherwise indicated, when reference is made to a specifically-named aryl (e.g., phenyl), cycloalkyl (e.g., cyclohexyl), heterocyclo (e.g., pyrrolidinyl, piperidinyl, and morpholinyl) or heteroaryl (e.g, tetrazolyl, imidazolyl, pyrazolyl, triazolyl, thiazolyl, and furyl) the reference is intended to include rings having 0 to 3, preferably 0 to 2, substituents selected from those recited above for the aryl, cycloalkyl, heterocyclo and/or heteroaryl groups, as appropriate.
The term "carbocyclyl" or "carbocyclic" refers to a saturated or unsaturated monocyclic or bicyclic ring in which all atoms of all rings are carbon. Thus, the term includes cycloalkyl and aryl rings. Monocyclic carbocycles have 3 to 6 ring atoms, still more typically 5 or 6 ring atoms. Bicyclic carbocycles have 7 to 12 ring atoms, e.g, arranged as a bicyclo [4,5], [5,5], [5,6] or [6,6] system, or 9 or 10 ring atoms arranged as a bicyclo [5,6] or [6,6] system. Examples of mono- and bicyclic carbocycles include cyclopropyl, cyclobutyl, cyclopentyl, 1 -cyclopent- 1-enyl, l-cyclopent-2-enyl, 1- cy cl opent-3 -enyl, cyclohexyl, 1 -cyclohex- 1-enyl, l-cyclohex-2-enyl, 1 -cyclohex-3 -enyl, phenyl and naphthyl. The carbocyclic ring may be substituted in which case the substituents are selected from those recited above for cycloalkyl and aryl groups.
The term "alkylthio" refers to the group "alkyl-S-".
The term "acyl" refers to a group -C(O)R, wherein R is hydrogen, alkyl, cycloalkyl, heterocyclyl, aryl, heteroalkyl, or heteroaryl; each of which may be optionally
substituted, as defined herein. Examples of acyl include formyl, acetyl, cylcohexylcarbonyl, cyclohexylmethyl-carbonyl, and benzoyl.
The term "amido" refers to both a "C-amido" group which refers to the group — C(O)NRgRh and an "N-amido" group which refers to the group -NRgC(O)Rh, wherein Rg and Rh are independently selected from hydrogen, alkyl, aryl, haloalkyl, or heteroaryl; each of which may be optionally substituted.
The term "amino" refers to the group -NRg Rh wherein Rg and Rh are independently selected from hydrogen, alkyl, haloalkyl, aryl, or heteroaryl; each of which may be optionally substituted.
The term "azido" refers to -N3.
The term "carbamoyl" refers to both an "O-carbamoyl" group which refers to the group -O-C(O)NR'R' and an "N-carbamoyl" group which refers to the group - NR1C(O)ORi, wherein R1 and R> are independently selected from hydrogen, alkyl, aryl, haloalkyl, or heteroaryl; each of which may be optionally substituted.
The term "carboxyl" refers to -C(O)OH.
The term "carboxyl ester" refers to both -OC(O)R and -C(O)ORg, wherein Rg is hydrogen, alkyl, cycloalkyl, heterocyclyl, aryl, heteroalkyl, or heteroaryl; each of which may be optionally substituted, as defined herein.
The term "cyano" or "carbonitrile" refers to the group -CN.
The term "cycloalkyl" refers to a saturated or partially unsaturated cyclic alkyl group having a single ring or multiple rings including fused, bridged, and spiro ring systems. The term "cycloalkyl" includes cycloalkenyl groups (i.e. the cyclic group having at least one double bond). As used herein, cycloalkyl has from 3 to 20 ring carbon atoms (i.e., C. sub.3-20 cycloalkyl), 3 to 12 ring carbon atoms (i.e., C. sub.3-12 cycloalkyl), 3 to 10 ring carbon atoms (i.e., C. sub.3-10 cycloalkyl), 3 to 8 ring carbon atoms (i.e., C.sub.3- 8 cycloalkyl), or 3 to 6 ring carbon atoms (i.e., C. sub.3-6 cycloalkyl). Examples of cycloalkyl groups include cyclopropyl, cyclobutyl, cyclopentyl, and cyclohexyl.
The term "heteroatoms" shall include oxygen, sulfur and nitrogen.
When the term "unsaturated" is used herein to refer to a ring or group, the ring or group may be fully unsaturated or partially unsaturated.
Throughout the specification, groups and substituents thereof may be chosen by one skilled in the field to provide stable moieties and compounds and compounds useful
as pharmaceutically-acceptable compounds and/or intermediate compounds useful in making pharmaceutically-acceptable compounds.
It should be understood that the selections for all groups, including for example, alkoxy, thioalkyl, and aminoalkyl, will be made by one skilled in the field to provide stable compounds.
The term "substituted", as used herein, means that any one or more hydrogens on the designated atom or group is replaced with a selection from the indicated group, provided that the designated atom's normal valence is not exceeded. When a substituent is oxo, or keto, (z.e., =0) then 2 hydrogens on the atom are replaced. Keto substituents are not present on aromatic moieties. Unless otherwise specified, substituents are named into the core structure. For example, it is to be understood that when (cycloalkyl)alkyl is listed as a possible substituent, the point of attachment of this substituent to the core structure is in the alkyl portion. Ring double bonds, as used herein, are double bonds that are formed between two adjacent ring atoms (e.g., C=C, C=N, or N=N).
Combinations of substituents and/or variables are permissible only if such combinations result in stable compounds or useful synthetic intermediates. A stable compound or stable structure is meant to imply a compound that is sufficiently robust to survive isolation from a reaction mixture to a useful degree of purity, and subsequent formulation into an efficacious therapeutic agent. It is preferred that the presently recited compounds do not contain a N-halo, S(0)2H, or S(O)H group.
The compounds described herein may exist in a free form (with no ionization) or can form salts which are also within the scope of this invention. Unless otherwise indicated, reference to an inventive compound is understood to include reference to the free form and to salts thereof. The term "salt(s)" denotes acidic and/or basic salts formed with inorganic and/or organic acids and bases. In addition, the term "salt(s)" may include zwitterions (inner salts), e.g., when a compound of formula I, contains both a basic moiety, such as an amine or a pyridine or imidazole ring, and an acidic moiety, such as a carboxylic acid. Pharmaceutically acceptable (i.e., non-toxic, physiologically acceptable) salts are preferred, such as, for example, acceptable metal and amine salts in which the cation does not contribute significantly to the toxicity or biological activity of the salt. However, other salts may be useful, e.g., in isolation or purification steps which may be employed during preparation, and thus, are contemplated within the scope of the
invention. Salts of the compounds of herein may be formed, for example, by reacting a compound described herein with an amount of acid or base, such as an equivalent amount, in a medium such as one in which the salt precipitates or in an aqueous medium followed by lyophilization.
Exemplary acid addition salts include acetates (such as those formed with acetic acid or trihaloacetic acid, for example, trifluoroacetic acid), adipates, alginates, ascorbates, aspartates, benzoates, benzenesulfonates, bisulfates, borates, butyrates, citrates, camphorates, camphorsulfonates, cyclopentanepropionates, digluconates, dodecylsulfates, ethanesulfonates, fumarates, glucoheptanoates, glycerophosphates, hemisulfates, heptanoates, hexanoates, hydrochlorides (formed with hydrochloric acid), hydrobromides (formed with hydrogen bromide), hydroiodides, 2- hydroxy ethanesulfonates, lactates, maleates (formed with maleic acid), methanesulfonates (formed with methanesulfonic acid), 2-naphthalenesulfonates, nicotinates, nitrates, oxalates, pectinates, persulfates, 3 -phenylpropionates, phosphates, picrates, pivalates, propionates, salicylates, succinates, sulfates (such as those formed with sulfuric acid), sulfonates (such as those mentioned herein), tartrates, thiocyanates, toluenesulfonates such as tosylates, undecanoates, and the like.
Exemplary basic salts include ammonium salts, alkali metal salts such as sodium, lithium, and potassium salts; alkaline earth metal salts such as calcium and magnesium salts; barium, zinc, and aluminum salts; salts with organic bases (for example, organic amines) such as trialkylamines such as triethylamine, procaine, dibenzylamine, N-benzyl- P-phenethylamine, 1 -ephenamine, 7V,7V'-dibenzylethylene-diamine, dehydroabietylamine, N-ethylpiperidine, benzylamine, dicyclohexylamine or similar pharmaceutically acceptable amines and salts with amino acids such as arginine, lysine and the like. Basic nitrogen-containing groups may be quaternized with agents such as lower alkyl halides (e.g., methyl, ethyl, propyl, and butyl chlorides, bromides and iodides), dialkyl sulfates (e.g., dimethyl, diethyl, dibutyl, and diamyl sulfates), long chain halides (e.g., decyl, lauryl, myristyl and stearyl chlorides, bromides and iodides), aralkyl halides (e.g., benzyl and phenethyl bromides), and others. Preferred salts include monohydrochloride, hydrogensulfate, methanesulfonate, phosphate or nitrate salts.
The compounds described herein can be provided as amorphous solids or crystalline solids. Lyophilization can be employed to provide the compounds as a solid.
It should further be understood that solvates (e.g., hydrates) of the compounds described herein are also within the scope of the present invention. The term “solvate” means a physical association of a compound with one or more solvent molecules, whether organic or inorganic. This physical association includes hydrogen bonding. In certain instances, the solvate will be capable of isolation, for example when one or more solvent molecules are incorporated in the crystal lattice of the crystalline solid. “Solvate” encompasses both solution-phase and isolable solvates. Exemplary solvates include hydrates, ethanolates, methanolates, isopropanolates, acetonitrile solvates, and ethyl acetate solvates. Methods of solvation are known in the art.
In addition, compounds described herein subsequent to their preparation, can be isolated and purified to obtain a composition containing an amount by weight equal to or greater than 99% of a compound (“substantially pure”), which is then used or formulated as described herein. Such “substantially pure” compounds described herein are also contemplated herein as part of the present invention.
The phrase "pharmaceutically acceptable" is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
As used herein, "pharmaceutically acceptable salts" refer to derivatives of the disclosed compounds wherein the parent compound is modified by making acid or base salts thereof. Examples of pharmaceutically-acceptable salts include, but are not limited to, mineral or organic acid salts of basic groups such as amines; and alkali or organic salts of acidic groups such as carboxylic acids. The pharmaceutically-acceptable salts include the conventional non-toxic salts or the quaternary ammonium salts of the parent compound formed, for example, from non-toxic inorganic or organic acids. For example, such conventional non-toxic salts include those derived from inorganic acids such as hydrochloric, hydrobromic, sulfuric, sulfamic, phosphoric, and nitric; and the salts prepared from organic acids such as acetic, propionic, succinic, glycolic, stearic, lactic, malic, tartaric, citric, ascorbic, pamoic, maleic, hydroxymaleic, phenylacetic, glutamic, benzoic, salicylic, sulfanilic, 2-acetoxybenzoic, fumaric, toluenesulfonic, methanesulfonic, ethane disulfonic, oxalic, and isethionic, and the like.
The pharmaceutically acceptable salts of the present invention can be synthesized from the parent compound which contains a basic or acidic moiety by conventional chemical methods. Generally, such salts can be prepared by reacting the free acid or base forms of these compounds with a stoichiometric amount of the appropriate base or acid in water or in an organic solvent, or in a mixture of the two; generally, nonaqueous media like ether, ethyl acetate, ethanol, isopropanol, or acetonitrile are preferred. Lists of suitable salts are found in Remington's Pharmaceutical Sciences, 18th Edition, Mack Publishing Company, Easton, PA (1990), the disclosure of which is hereby incorporated by reference.
“Stable compound” and “stable structure” are meant to indicate a compound that is sufficiently robust to survive isolation to a useful degree of purity from a reaction mixture, and formulation into an efficacious therapeutic agent. The present invention is intended to embody stable compounds.
“Therapeutically effective amount” is intended to include an amount of a compound of the present invention alone or an amount of the combination of compounds claimed or an amount of a compound of the present invention in combination with other active ingredients effective to act as an inhibitor of USP1, or effective to treat or prevent proliferative disorders, such as cancer.
As used herein, “treating” or “treatment” cover the treatment of a disease-state in a mammal, particularly in a human, and include: (a) preventing the disease-state from occurring in a mammal, in particular, when such mammal is predisposed to the diseasestate but has not yet been diagnosed as having it; (b) inhibiting the disease-state, i.e., arresting its development; and/or (c) relieving the disease-state, i.e., causing regression of the disease state.
All stereoisomers of the compounds of the instant invention are contemplated, either in admixture or in pure or substantially pure form. Stereoisomers may include compounds which are optical isomers through possession of one or more chiral atoms, as well as compounds which are optical isomers by virtue of limited rotation about one or more bonds (atropisomers). The definition of compounds according to the invention embraces all the possible stereoisomers and their mixtures. It very particularly embraces the racemic forms and the isolated optical isomers having the specified activity. The racemic forms can be resolved by physical methods, such as, for example, fractional crystallization, separation or
crystallization of diastereomeric derivatives or separation by chiral column chromatography. The individual optical isomers can be obtained from the racemates from the conventional methods, such as, for example, salt formation with an optically active acid followed by crystallization.
The present invention is intended to include all isotopes of atoms occurring in the present compounds. Isotopes include those atoms having the same atomic number but different mass numbers. By way of general example and without limitation, isotopes of hydrogen include deuterium and tritium. Isotopes of carbon include 13C and 14C. Isotopically-labeled compounds of the invention can generally be prepared by conventional techniques known to those skilled in the art or by processes analogous to those described herein, using an appropriate isotopically-labeled reagent in place of the non-labeled reagent otherwise employed.
Prodrugs and solvates of the inventive compounds are also contemplated. The term "prodrug" denotes a compound which, upon administration to a subject, undergoes chemical conversion by metabolic or chemical processes to yield a compound of the formula I, and/or a salt and/or solvate thereof. Any compound that will be converted in vivo to provide the bioactive agent (ie., the compound for formula I) is a prodrug within the scope and spirit of the invention. For example, compounds containing a carboxy group can form physiologically hydrolyzable esters which serve as prodrugs by being hydrolyzed in the body to yield formula I compounds per se. Such prodrugs are preferably administered orally since hydrolysis in many instances occurs principally under the influence of the digestive enzymes. Parenteral administration may be used where the ester per se is active, or in those instances where hydrolysis occurs in the blood. Examples of physiologically hydrolyzable esters of compounds of formula I include Ci-ealkylbenzyl, 4-methoxybenzyl, indanyl, phthalyl, methoxymethyl, Ci-ealkanoyloxy-Ci-ealkyl, e.g., acetoxymethyl, pivaloyloxymethyl or propionyloxymethyl, Ci-ealkoxycarbonyloxy-Ci-ealkyl, e.g., methoxycarbonyl-oxymethyl or ethoxycarbonyloxymethyl, glycyloxymethyl, phenylglycyloxymethyl, (5-methyl-2- oxo- 1,3 -di oxolen-4-yl)-m ethyl and other well known physiologically hydrolyzable esters used, for example, in the penicillin and cephalosporin arts. Such esters may be prepared by conventional techniques known in the art.
Various forms of prodrugs are well known in the art and are described in Rautio, J. et al., Nature Review Drug Discovery, 17, 559-587 (2018).
Compounds described in the application and their salts may exist in their tautomeric form, in which hydrogen atoms are transposed to other parts of the molecules and the chemical bonds between the atoms of the molecules are consequently rearranged. It should be understood that the all tautomeric forms, insofar as they may exist, are included within the invention. Additionally, inventive compounds may have trans- and cis-\ somers.
The disclosure herein further relates to compounds described herein, the tautomers and stereoisomeric forms thereof, and the pharmaceutically acceptable addition salts, and the solvates thereof, for use as a medicament. Furthermore, the disclosure herein relates to the use of a compound described herein, a tautomer or a stereoisomeric form thereof, or a pharmaceutically acceptable addition salt, or a solvate thereof, or a pharmaceutical composition according to the invention, for the manufacture of a medicament.
The inventive compositions may contain other therapeutic agents as described above and may be formulated, for example, by employing conventional solid or liquid vehicles or diluents, as well as pharmaceutical additives of a type appropriate to the mode of desired administration (e.g., excipients, binders, preservatives, stabilizers, flavors, etc.) according to techniques such as those well known in the art of pharmaceutical formulation.
Accordingly, the present invention further includes compositions comprising one or more compounds described herein and a pharmaceutically acceptable carrier.
A "pharmaceutically acceptable carrier" refers to media generally accepted in the art for the delivery of biologically active agents to animals, in particular, mammals. Pharmaceutically acceptable carriers are formulated according to a number of factors well within the purview of those of ordinary skill in the art. These include without limitation the type and nature of the active agent being formulated; the subject to which the agentcontaining composition is to be administered; the intended route of administration of the composition; and, the therapeutic indication being targeted. Pharmaceutically acceptable carriers include both aqueous and non-aqueous liquid media, as well as a variety of solid and semi-solid dosage forms. Such carriers can include a number of different ingredients
and additives in addition to the active agent, such additional ingredients being included in the formulation for a variety of reasons, e.g., stabilization of the active agent, binders, etc., well known to those of ordinary skill in the art. Descriptions of suitable pharmaceutically acceptable carriers, and factors involved in their selection, are found in a variety of readily available sources such as, for example, Remington's Pharmaceutical Sciences, 17th Edition (1985), which is incorporated herein by reference in its entirety.
The compounds described herein may be administered by any means suitable for the condition to be treated, which may depend on the need for site-specific treatment or quantity of drug to be delivered. Topical administration is generally preferred for skin- related diseases, and systematic treatment preferred for cancerous or pre-cancerous conditions, although other modes of delivery are contemplated. For example, the compounds may be delivered orally, such as in the form of tablets, capsules, granules, powders, or liquid formulations including syrups; topically, such as in the form of solutions, suspensions, gels or ointments; sublingually; bucally; parenterally, such as by subcutaneous, intravenous, intramuscular or intrasternal injection or infusion techniques (e.g., as sterile injectable aq. or non-aq. solutions or suspensions); nasally such as by inhalation spray; topically, such as in the form of a cream or ointment; rectally such as in the form of suppositories; or liposomally. Dosage unit formulations containing non-toxic, pharmaceutically acceptable vehicles or diluents may be administered. The compounds may be administered in a form suitable for immediate release or extended release. Immediate release or extended release may be achieved with suitable pharmaceutical compositions or, particularly in the case of extended release, with devices such as subcutaneous implants or osmotic pumps.
Exemplary compositions for oral administration include suspensions which may contain, for example, microcrystalline cellulose for imparting bulk, alginic acid or sodium alginate as a suspending agent, methylcellulose as a viscosity enhancer, and sweeteners or flavoring agents such as those known in the art; and immediate release tablets which may contain, for example, microcrystalline cellulose, dicalcium phosphate, starch, magnesium stearate and/or lactose and/or other excipients, binders, extenders, disintegrants, diluents and lubricants such as those known in the art. The inventive compounds may also be orally delivered by sublingual and/or buccal administration, e.g., with molded, compressed, or freeze-dried tablets. Exemplary compositions may include fast-dissolving
diluents such as mannitol, lactose, sucrose, and/or cyclodextrins. Also included in such formulations may be high molecular weight excipients such as celluloses (AVICEL®) or polyethylene glycols (PEG); an excipient to aid mucosal adhesion such as hydroxypropyl cellulose (HPC), hydroxypropyl methyl cellulose (HPMC), sodium carboxymethyl cellulose (SCMC), and/or maleic anhydride copolymer (e.g., GANTREZ®); and agents to control release such as polyacrylic copolymer (e.g., CARBOPOL 934®). Lubricants, glidants, flavors, coloring agents and stabilizers may also be added for ease of fabrication and use.
Formulations for parenteral administration may be in the form of aqueous or nonaqueous isotonic sterile injection solutions or suspensions. These solutions and suspensions may be prepared from sterile powders or granules using one or more of the carriers or diluents mentioned for use in the formulations for oral administration or by using other suitable dispersing or wetting agents and suspending agents. The compounds may be dissolved in water, polyethylene glycol, propylene glycol, ethanol, com oil, cottonseed oil, peanut oil, sesame oil, benzyl alcohol, sodium chloride, tragacanth gum, and/or various buffers. Other adjuvants and modes of administration are well and widely known in the pharmaceutical art. The active ingredient may also be administered by injection as a composition with suitable carriers including saline, dextrose, or water, or with cyclodextrin (i.e. Captisol), cosolvent solubilization (i.e. propylene glycol) or micellar solubilization (i.e. Tween 80).
Exemplary compositions for parenteral administration include injectable solutions or suspensions which may contain, for example, suitable non-toxic, parenterally acceptable diluents or solvents, such as mannitol, 1,3 -butanediol, water, Ringer's solution, an isotonic sodium chloride solution, or other suitable dispersing or wetting and suspending agents, including synthetic mono- or diglycerides, and fatty acids, including oleic acid.
The sterile injectable preparation may also be a sterile injectable solution or suspension in a non-toxic parenterally acceptable diluent or solvent, for example as a solution in 1,3 -butanediol. Among the acceptable vehicles and solvents that may be employed are water, Ringer’s solution, and isotonic sodium chloride solution. In addition, sterile, fixed oils are conventionally employed as a solvent or suspending medium. For this purpose any bland fixed oil may be employed, including synthetic
mono- or diglycerides. In addition, fatty acids such as oleic acid find use in the preparation of injectables.
A sterile injectable oil-in-water microemulsion can, for example, be prepared by 1) dissolving at least one compound described herein in an oily phase, such as, for example, a mixture of soybean oil and lecithin; 2) combining the compound-containing oil phase with a water and glycerol mixture; and 3) processing the combination to form a microemulsion.
A sterile aqueous or oleaginous suspension can be prepared in accordance with methods already known in the art. For example, a sterile aqueous solution or suspension can be prepared with a non-toxic parenterally-acceptable diluent or solvent, such as, for example, 1,3-butane diol; and a sterile oleaginous suspension can be prepared with a sterile non-toxic acceptable solvent or suspending medium, such as, for example, sterile fixed oils, e.g., synthetic mono- or diglycerides; and fatty acids, such as, for example, oleic acid.
Exemplary compositions for nasal aerosol or inhalation administration include solutions which may contain, for example, benzyl alcohol or other suitable preservatives, absorption promoters to enhance absorption and/or bioavailability, and/or other solubilizing or dispersing agents such as those known in the art.
Dispersible powders and granules can, for example, be prepared by admixing at least one compound described herein or a pharmaceutically acceptable salt thereof, with at least one dispersing and/or wetting agent; at least one suspending agent; and/or at least one preservative. Exemplary preservatives include, but are not limited to, for example, anti-oxidants, e.g., ascorbic acid. In addition, dispersible powders and granules can also contain at least one excipient, including, but not limited to, for example, sweetening agents; flavoring agents; and coloring agents.
Exemplary compositions for rectal administration include suppositories which may contain, for example, suitable non-irritating excipients, such as cocoa butter, synthetic glyceride esters or polyethylene glycols, which are solid at ordinary temperatures but liquefy and/or dissolve in the rectal cavity to release the drug.
The therapeutically-effective amount of a compound of the present invention may be determined by one of ordinary skill in the art, and includes exemplary dosage amounts for a mammal of from about 0.05 to 1000 mg/kg; 1-1000 mg/kg; 1-50 mg/kg; 5-250
mg/kg; 250-1000 mg/kg of body weight of active compound per day, which may be administered in a single dose or in the form of individual divided doses, such as from 1 to 4 times per day. It will be understood that the specific dose level and frequency of dosage for any particular subject may be varied and will depend upon a variety of factors, including the activity of the specific compound employed, the metabolic stability and length of action of that compound, the species, age, body weight, general health, sex and diet of the subject, the mode and time of administration, rate of excretion, drug combination, and severity of the particular condition. Preferred subjects for treatment include animals, most preferably mammalian species such as humans, and domestic animals such as dogs, cats, horses, and the like. Thus, when the term "patient" is used herein, this term is intended to include all subjects, most preferably mammalian species that are affected by modulation of USP1 -mediated functions.
In one embodiment, the present application provides a combined preparation of a compound described herein and/or a pharmaceutically acceptable salt thereof, a stereoisomer thereof or a tautomer thereof, and additional therapeutic agent(s) for simultaneous, separate or sequential use in the treatment and/or prophylaxis of multiple diseases or disorders associated with USP1.
In another aspect, the application provides a method of treating a patient suffering from or susceptible to a medical condition that is associated with USP1. A number of medical conditions can be treated. The method comprises administering to the patient a therapeutically effective amount of a composition comprising a compound described herien and/or a pharmaceutically acceptable salt thereof, a stereoisomer thereof or a tautomer thereof. For example, the compounds described herein may be used to treat or proliferative diseases such as cancer, immunological disorders or inflammatory disorders.
The compounds described herein are useful for the treatment of cancer. In other embodiments, the compounds described herein may be used to treat cancers that are mediated by, dependent on or associated with USP1 activity. In certain embodiments, the disease is a solid tumor. In particular embodiments, the solid tumor is selected from prostate cancer, pancreatic cancer, bladder cancer, colorectal cancer, breast cancer, renal cancer, hepatocellular cancer, lung cancer, ovarian cancer, cervical cancer, gastric cancer, esophageal cancer, head and neck cancer, melanoma, neuroendocrine cancers, CNS cancers, brain tumors (e.g., glioma, anaplastic oligodendroglioma, adult glioblastoma
multiforme, and adult anaplastic astrocytoma), bone cancer, or soft tissue sarcoma. In some embodiments, the solid tumor is from non-small cell lung cancer or small-cell lung cancer.
In one embodiment, the compounds herein can be useful in the treatment of haematological malignancies. In one embodiment, hematological malignancy is selected from multiple myeloma, non-Hodgkin's lymphoma, Hodgkin lymphoma, T-cell leukaemia, mucosa-associated lymphoid tissue lymphoma, diffuse large B-cell lymphoma and mantle cell lymphoma. In one embodiment solid tumor is selected from pancreatic cancer, breast cancer, melanoma and non-small cell lung cancer.
In one embodiment, cancer is selected from a carcinoma, preferably a carcinoma of the bladder, breast, colon (including colorectal carcinomas, such as colon adenocarcinoma and colon adenoma), kidney, urothelial, uterus, epidermis, liver, lung (including adenocarcinoma, small cell lung cancer, non-small cell lung carcinomas and squamous lung cancer), oesophagus, head and neck, gall bladder, ovary, pancreas (including exocrine pancreatic carcinoma), stomach, gastrointestinal cancer (including gastrointestinal stromal tumors), cervix, endometrium, thyroid, prostate and skin.
In one embodiment, the cancer is selected from pituitary cancer, a hematopoietic tumor of lymphoid lineage, for example leukemia, acute lymphocytic leukemia, chronic lymphocytic leukemia, B-cell lymphoma (e.g. diffuse large B-cell lymphoma, mantle cell lymphoma), T-cell leukaemia/lymphoma, Hodgkin's lymphoma, non-Hodgkin's lymphoma, hairy cell lymphoma, or Burkett's lymphoma; a hematopoietic tumor of myeloid lineage, for example leukemias, acute and chronic myelogenous leukemias, chronic myelomonocytic leukemia (CMML), myeloproliferative disorder, myeloproliferative syndrome, myelodysplastic syndrome, or promyelocytic leukemia; multiple myeloma; thyroid follicular cancer; hepatocellular cancer, a tumor of mesenchymal origin (e.g. Ewing's sarcoma), for example fibrosarcoma or rhabdomyosarcoma; a tumor of the central or peripheral nervous system, for example astrocytoma, neuroblastoma, glioma (such as glioblastoma multiforme) or schwannoma; melanoma; seminoma; teratocarcinoma; osteosarcoma; xeroderma pigmentosum; keratoctanthoma; thyroid follicular cancer; or Kaposi's sarcoma.
In other embodiments, the compounds described herein may be used to treat cancers that are mediated by, dependent on or associated with USP1 activity. In certain
embodiments, the disease is a solid tumor. In particular embodiments, the solid tumor is from prostate cancer, pancreatic cancer, bladder cancer, colorectal cancer, breast cancer, renal cancer, hepatocellular cancer, lung cancer, ovarian cancer, cervical cancer, gastric cancer, esophageal cancer, head and neck cancer, melanoma, neuroendocrine cancers, CNS cancers, brain tumors (e.g., glioma, anaplastic oligodendroglioma, adult glioblastoma multiforme, and adult anaplastic astrocytoma), bone cancer, or soft tissue sarcoma. In some embodiments, the solid tumor is from non-small cell lung cancer or small-cell lung cancer.
In other embodiments, the disease is a hematologic malignancy. In certain embodiments, the disease is lymphoma, multiple myeloma, or leukemia. In certain embodiments, the hematologic malignancy is leukemia or lymphoma. In specific embodiments, the disease is acute lymphocytic leukemia (ALL), acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma (SLL), myelodysplastic syndrome (MDS), myeloproliferative disease (MPD), chronic myeloid leukemia (CML), juvenile myelomonocytic leukemia (JMML), multiple myeloma (MM), Hodgkin lymphoma, indolent non-Hodgkin's lymphoma (iNHL), refractory iNHL, nonHodgkin's lymphoma (NHL), mantle cell lymphoma (MCL), follicular lymphoma, Waldenstrom’s macroglobulinemia (WM), minimal residual disease (MRD), T-cell lymphoma, B-cell lymphoma, diffuse large B-cell lymphoma (DLBCL), T-cell acute lymphoblastic leukemia (T-ALL), B-cell acute lymphoblastic leukemia (B-ALL), lymphoplasmacytic lymphoma, marginal zone lymphoma, or Burkitt lymphoma. In one embodiment, the disease is T-cell acute lymphoblastic leukemia (T-ALL), or B-cell acute lymphoblastic leukemia (B-ALL). In some embodiments, non-Hodgkin lymphoma can be indolent B-cell diseases including follicular lymphoma, lymphoplasmacytic lymphoma, Waldenstrom macroglobulinemia, and marginal zone lymphoma, as well as the aggressive lymphomas that include, for example, Burkitt lymphoma, diffuse large B- cell lymphoma (DLBCL) and mantle cell lymphoma (MCL).
In some embodiments, the cancer is selected from hematological cancer, a lymphatic cancer. In some embodiments, the cancer comprises cancer cells with DNA damage repair pathway deficiency. In some embodiments, the cancer is a homologous recombination deficient cancer. In some embodiments, the cancer comprises cancer cells with a mutation in a gene encoding p53. In some embodiments, the mutation in a gene
encoding p53 is a germline or somatic mutation. In some embodiments, the cancer comprises with cancer cells with loss of function mutation in a gene encoding p53. In some embodiments, the cancer is a BRCA1 and/or BRCA2 deficient cancer. In some embodiments, the cancer is a somatic or germline BRCA1 and/or BRCA2 mutant cancer. In some embodiments, the cancer is a Poly (ADP -ribose) polymerase (“PARP”) inhibitor refractory or resistant cancer. In some embodiments, the cancer is a PARP inhibitor resistant or refractory BRCA1 and/or BRCA2 deficient cancer. In some embodiments, the cancer cell has a germline or somatic mutation in a gene encoding ataxia telangiectasia mutated (ATM) protein kinase or ATM deficiency. In some embodiments, the cancer has a mutation in the gene encoding more than two of p53, BRCA1, BRCA2, ATM.
In some embodiments, the disease is an autoimmune or inflammatory disease or disorder. These autoimmune or inflammatory diseases or conditions may be chronic or acute and include, but are not limited to, inflammatory pelvic disease, urethritis, skin sunburn, sinusitis, pneumonitis, encephalitis, meningitis, myocarditis, pericarditis, nephritis including lupus nephritis, osteomyelitis, myositis, eczema, hepatitis, gastritis, enteritis, dermatitis, gingivitis, appendicitis, pancreatitis, primary biliary cirrhosis, cholecystitis, sclerosing cholangitis, agammaglobulinemia, psoriasis, allergy, Crohn's disease, irritable bowel syndrome, ulcerative colitis, Sjogren's disease, tissue graft rejection such as acute graft-versus-host disease, hyperacute rejection of transplanted organs, asthma, chronic obstructive airways disease, allergic rhinitis, chronic obstructive pulmonary disease (COPD), autoimmune polyglandular disease (also known as autoimmune polyglandular syndrome), autoimmune alopecia, pernicious anemia, vasculitis, glomerulonephritis, giant cell arteritis, Wegener's granulomatosis, Polyarteritis nodosa, dermatomyositis, multiple sclerosis, scleroderma, autoimmune hemolytic and thrombocytopenic states, Goodpasture's syndrome, atherosclerosis, Addison's disease, hypophysitis, Parkinson's disease, Alzheimer's disease, Kawasaki disease, Takayasu's Arteritis, depression, retinitis, uveitis, scleritis, Type I diabetes, septic shock, systemic lupus erythematosus (SLE), rheumatoid arthritis, psoriatic arthritis, juvenile arthritis, osteoarthritis, gout, chronic idiopathic thrombocytopenic purpura, Waldenstrom macroglobulinemia, myasthenia gravis, Hashimoto's thyroiditis, atopic dermatitis, degenerative joint disease, vitiligo, bullous skin diseases, autoimmune hypopituitarism, Guillain-Barre syndrome, Behcet's disease, scleracierma, mycosis fungoides, acute
inflammatory responses (such as acute respiratory distress syndrome and ischemia/reperfusion injury), and Graves' disease. In some embodiments, the autoimmune and inflammatory diseases and conditions may also include systemic or tissue inflammation, inflammatory responses to hypoxia, cellular activation and proliferation, lipid metabolism, fibrosis, infections with bacteria, infections with viruses (e.g., herpes virus, human papilloma virus, adenovirus, poxvirus and other DNA viruses), fungi, parasites or their toxins, such as sepsis, sepsis syndrome, septic shock, endotoxaemia, systemic inflammatory response syndrome (SIRS), multi-organ dysfunction syndrome, toxic shock syndrome, acute lung injury, ARDS (adult respiratory distress syndrome), acute renal failure, fulminant hepatitis, burns, acute pancreatitis, post-surgical syndromes, sarcoidosis, Herxheimer reactions, encephalitis, myelitis, meningitis, malaria and SIRS associated with viral infections such as influenza, herpes zoster, herpes simplex and coronavirus.
Combination Therapies
In some embodiments, the compounds described herein may be administered in conjunction with standard of care, e.g., surgery, radiation, and/or chemotherapy. In some embodiments, the compounds may be administered in conjunction with a chemotherapeutic agent. In some embodiments, the compounds may be administered in conjunction with one or more of carboplatin, cisplatin, paclitaxel, nab-paclitaxel, gemcitabine or FOLFOX. In some embodiment, the compounds may be administered in conjunction with carboplatin or nab-paclitaxel. In some embodiments, the compounds may be administered in conjunction with carboplatin and paclitaxel. In some embodiments, the compounds may be administered in conjunction with cisplatin and pemetrexed. In some embodiments, the compounds may be administered in conjunction with cisplatin and gemcitabine. In some embodiments, the compounds may be administered in conjunction with FOLFOX. In some embodiments, the compounds may be administered in conjunction with FOLFIRI. In one embodiment, the compounds may be administered in combination with decarbazine for the treatment of melanoma. In some embodiments, cisplatin is intravenously administered as a 100 mg/ml dose once every four weeks. In some embodiments, the compounds may be administered in conjunction with doxorubicin (adriamycin), cisplatin bleomycin sulfate, carmustine, chlorambucil,
dacarbazine and/or cyclophosphamide hydroxyurea. In some embodiments, adriamycin is intravenously administered as a 60 mg/ml to 75 mg/ml dose once every 21 days.
In one embodiment, the compounds of the present application (e.g., a compound described herein or a pharmaceutically acceptable salt, prodrug, or solvate thereof) may be used in combination with one or more additional therapeutic agent that are being used and/or developed to treat cancers or inflammatory disorders. The one or more additional therapeutic agent may be an inhibitor to Janus kinase (JAK) such as JAK1, JAK2 and/or JAK3, Tyroansine kinase (TYK), K-Ras, Mitogen activated protein kinases (MAPK), Bruton's tyrosine kinase (BTK), bromodomain containing protein inhibitor (BRD) such as BRD4, a lysyl oxidase protein (LOX), lysyl oxidase-like protein (LOXL) such as LOXL1-5, matrix metalloprotease (MMP) such as MMP 1-10, adenosine A2B receptor (A2B), isocitrate dehydrogenase (IDH) such as IDH1, apoptosis signal -regulating kinase (ASK) such as ASK1, serine/threonine kinase TPL2, discoidin domain receptor (DDR) such as DDR1 and DDR2, histone deacetylase (HD AC), protein kinase C (PKC), or any combination thereof.
In one embodiment, the compounds of the present application (e.g., a compound described herein or a pharmaceutically acceptable salt, prodrug, or solvate thereof) may be used in combination with additional chemotherapeutic agent, an immunotherapeutic agent, a radiotherapeutic agent, an anti-neoplastic agent, an anti-cancer agent, an anti- fibrotic agent, an anti -angiogenic agent, a therapeutic antibody, or any combination thereof.
Chemotherapeutic agents may be categorized by their mechanism of action into, for example, the following groups: anti-metabolites/anti-cancer agents, such as pyrimidine analogs (floxuridine, capecitabine, and cytarabine); purine analogs, folate antagonists and related inhibitors antiproliferative/antimitotic agents including natural products such as vinca alkaloid (vinblastine, vincristine) and microtubule such as taxane (paclitaxel, docetaxel), vinblastin, nocodazole, epothilones and navelbine, epidipodophyllotoxins (etoposide, teniposide); DNA damaging agents (actinomycin, amsacrine, busulfan, carboplatin, chlorambucil, cisplatin, cyclophosphamide, Cytoxan, dactinomycin, daunorubicin, doxorubicin, epirubicin, iphosphamide, melphalan, merchlorehtamine, mitomycin, mitoxantrone, nitrosourea, procarbazine, taxol, taxotere, teniposide, etoposide, triethylenethiophosphoramide); antibiotics such as dactinomycin
(actinomycin D), daunorubicin, doxorubicin (adriamycin), idarubicin, anthracyclines, mitoxantrone, bleomycins, plicamycin (mithramycin) and mitomycin; enzymes (L- asparaginase which systemically metabolizes L-asparagine and deprives cells which do not have the capacity to synthesize their own asparagine); antiplatelet agents; antiproliferative/antimitotic alkylating agents such as nitrogen mustards cyclophosphamide and analogs, melphalan, chlorambucil), and (hexamethylmelamine and thiotepa), alkyl nitrosoureas (BCNU) and analogs, streptozocin), trazenes-dacarbazinine (DTIC); antiproliferative/antimitotic antimetabolites such as folic acid analogs (methotrexate); platinum coordination complexes (cisplatin, oxiloplatinim, carboplatin), procarbazine, hydroxyurea, mitotane, aminoglutethimide; hormones, hormone analogs (estrogen, tamoxifen, goserelin, bicalutamide, nilutamide) and aromatase inhibitors (letrozole, anastrozole); anticoagulants (heparin, synthetic heparin salts and other inhibitors of thrombin); fibrinolytic agents (such as tissue plasminogen activator, streptokinase and urokinase), aspirin, dipyridamole, ticlopidine, clopidogrel; antimigratory agents; antisecretory agents (breveldin); immunosuppressives tacrolimus sirolimus azathioprine, mycophenolate; compounds (TNP-470, genistein) and growth factor inhibitors (vascular endothelial growth factor inhibitors, fibroblast growth factor inhibitors); angiotensin receptor blocker, nitric oxide donors; anti-sense oligonucleotides; antibodies (trastuzumab, rituximab); cell cycle inhibitors and differentiation inducers (tretinoin); inhibitors, topoisomerase inhibitors (doxorubicin (adriamycin), daunorubicin, dactinomycin, eniposide, epirubicin, etoposide, idarubicin, irinotecan and mitoxantrone, topotecan, irinotecan, camptothesin), corticosteroids (cortisone, dexamethasone, hydrocortisone, methylpednisolone, prednisone, and prenisolone); growth factor signal transduction kinase inhibitors; dysfunction inducers, toxins such as Cholera toxin, ricin, Pseudomonas exotoxin, Bordetella pertussis adenylate cyclase toxin, or diphtheria toxin, and caspase activators; and chromatin.
Examples of chemotherapeutic agents include alkylating agents such as thiotepa and cyclophosphamide; alkyl sulfonates such as busulfan, improsulfan and piposulfan; aziridines such as benzodopa, carboquone, meturedopa, and uredopa; emylerumines and memylamelamines including alfretamine, triemylenemelamine, triethylenephosphoramide, triethylenethiophosphoramide and trimemylolomelamine; acetogenins (especially bullatacin and bullatacinone); a camptothecin (including synthetic
analogue topotecan); bryostatin; callystatin; CC-1065 (including its adozelesin, carzelesin and bizelesin synthetic analogues); cryptophycins (articularly cryptophycin 1 and cryptophy cin 8); dolastatin; duocarmycin (including the synthetic analogues, KW-2189 and CBI-TMI); eleutherobin; pancrati statin; a sarcodictyin; spongistatin; nitrogen mustards such as chlorambucil, chlornaphazine, cholophosphamide, estramustine, ifosfamide, mechlorethamine, mechlorethamine oxide hydrochloride, melphalan, novembichin, phenesterine, prednimustine, trofosfamide, uracil mustard; nitrosoureas such as carmustine, chlorozotocin, foremustine, lomustine, nimustine, ranimustine; antibiotics such as the enediyne antibiotics (e.g., calicheamicin, calicheamicin gammall, dynemicin, dynemicin A; bisphosphonates, such as clodronate; an esperamicin; as well as neocarzinostatin chromophore and related chromoprotein enediyne antibiotic chromomophores), aclacinomysins, actinomycin, authramycin, azaserine, bleomycins, cactinomycin, carabicin, carrninomycin, carzinophilin, chromomycins, dactinomycin, daunorubicin, detorubicin, 6-diazo-5-oxo-L-norleucine, doxorubicin (including morpholino-doxorubicin, cyanomorpholino-doxorubicin, 2-pyrrolino-doxorubicin and deoxydoxorubicin), epirubicin, esorubicin, idarubicin, marcellomycin, mitomycins such as mitomycin C, mycophenolic acid, nogalamycin, olivomycins, peplomycin, potfiromycin, puromycin, quelamycin, rodorubicin, streptonigrin, streptozocin, tubercidin, ubenimex, zinostatin, zorubicin; anti-metabolites such as methotrexate and 5- fluorouracil (5-FU); folic acid analogues such as demopterin, methotrexate, pteropterin, trimetrexate; purine analogs such as fludarabine, 6-mercaptopurine, thiamiprine, thioguanine; pyrimidine analogues such as ancitabine, azacitidine, 6-azauridine, carmofur, cytarabine, dideoxyuridine, doxifluridine, enocitabine, floxuridine; androgens such as calusterone, dromostanolone propionate, epitiostanol, mepitiostane, testolactone; anti-adrenals such as aminoglutethimide, mitotane, trilostane; folic acid replinisher such as frolinic acid; aceglatone; aldophosphamide glycoside; aminolevulinic acid; eniluracil; amsacrine; hestrabucil; bisantrene; edatraxate; defofamine; demecolcine; diaziquone; elformthine; elliptinium acetate; an epothilone; etoglucid; gallium nitrate; hydroxyurea; lentinan; leucovorin; lonidamine; maytansinoids such as maytansine and ansamitocins; mitoguazone; mitoxantrone; mopidamol; nitracrine; pentostatin; phenamet; pirarubicin; losoxantrone; fluoropyrimidine; folinic acid; podophyllinic acid; 2-ethylhydrazide; procarbazine; razoxane; rhizoxin; sizofiran; spirogermanium; tenuazonic acid;
triaziquone; 2,2',2"-tricUorotriemylamine; trichothecenes (especially T-2 toxin, verracurin A, roridin A and anguidine); urethane; vindesine; dacarbazine; mannomustine; mitobronitol; mitolactol; pipobroman; gacytosine; arabinoside ("Ara-C"); cyclophosphamide; thiopeta; taxoids, e.g., paclitaxel and docetaxel, chlorambucil; gemcitabine; 6-thioguanine; mercaptopurine; methotrexate; platinum analogs such as cisplatin and carboplatin; vinblastine; platinum; etoposide; ifosfamide; mitroxantrone; vancristine; vinorelbine; novantrone; teniposide; edatrexate; daunomycin; aminopterin; xeoloda; ibandronate; CPT-11; topoisomerase inhibitor RFS 2000; difluoromethylomithine (DMFO); retinoids such as retinoic acid; capecitabine; FOLFIRI (fluorouracil, leucovorin, and irinotecan) and pharmaceutically acceptable salts, acids or derivatives of any of the above. One or more chemotherapeutic agent is used or included in the present application.
Chemotherapeutic agents may also include, for example, anti-hormonal agents that act to regulate or inhibit hormone action on tumors such as anti-estrogens and selective estrogen receptor modulators (SERMs), including, for example, tamoxifen, raloxifene, droloxifene, 4-hydroxytamoxifen, trioxifene, keoxifene, onapristone, and toremifene; inhibitors of the enzyme aromatase, which regulates estrogen production in the adrenal glands, such as, for example, 4(5)-imidazoles, aminoglutethimide, megestrol acetate, exemestane, formestane, fadrozole, vorozole letrozole and anastrozole; and antiandrogens such as flutamide, nilutamide, bicalutamide, leuprohde, and goserelin; and pharmaceutically acceptable salts thereof.
The anti-angiogenic agents include, but are not limited to, retinoid acid and derivatives thereof, 2-methoxyestradiol, suramin, squalamine, tissue inhibitor of metalloproteinase- 1, tissue inhibitor of metalloproternase-2, plasminogen activator inhibitor- 1, plasminogen activator inbibitor-2, cartilage-derived inhibitor, paclitaxel (nab- paclitaxel), platelet factor 4, protamine sulphate (clupeine), sulphated chitin derivatives (prepared from queen crab shells), sulphated polysaccharide peptidoglycan complex (sp- pg), staurosporine, modulators of matrix metabolism, including for example, proline analogs ((l-azetidine-2-carboxylic acid (LACA), cishydroxyproline, d, 1-3,4- dehydroproline, thiaproline, .alpha. -di pyridyl, beta-aminopropionitrile fumarate, 4- propyl-5-(4-pyridinyl)-2(3h)-oxazolone; methotrexate, mitoxantrone, heparin, interferons, 2 macroglobulin-serum, chimp-3, chymostatin, beta-cyclodextrin tetradecasulfate,
eponemycin; fumagillin, gold sodium thiomalate, d-penicillamine (CDPT), beta-1- anticollagenase-serum, alpba-2-antiplasmin, bisantrene, lobenzarit disodium, n-2- carboxyphenyl-4-chloroanthronilic acid disodium or "CCA", thalidomide; angiostatic steroid, cargboxynaminolmidazole; metalloproteinase inhibitors such as BB94. Other anti-angiogenesis agents include antibodies, preferably monoclonal antibodies against these angiogenic growth factors: beta-FGF, alpha-FGF, FGF-5, VEGF isoforms, VEGF- C, HGF/SF and Ang-l/Ang-2.
The application also provides a method for treating a subject who is undergoing one or more standard therapies, such as chemotherapy, radiotherapy, immunotherapy, surgery, or combination thereof. Accordingly, one or more therapeutic agent or inhibitors may be administered before, during, or after administration of chemotherapy, radiotherapy, immunotherapy, surgery or combination thereof.
In certain embodiments, the subject may be a human who is (i) substantially refractory to at least one chemotherapy treatment, or (ii) in relapse after treatment with chemotherapy, or both (i) and (ii). In some of embodiments, the subject is refractory to at least two, at least three, or at least four chemotherapy treatments (including standard or experimental chemotherapies).
In certain embodiments, the subject is refractory to at least one, at least two, at least three, or at least four chemotherapy treatment (including standard or experimental chemotherapy) selected from fludarabine, rituximab, obinutuzumab, alkylating agents, alemtuzumab and other chemotherapy treatments such as CHOP (cyclophosphamide, doxorubicin, vincristine, prednisone); R-CHOP (rituximab-CHOP); hyperCVAD (hyperfractionated cyclophosphamide, vincristine, doxorubicin, dexamethasone, methotrexate, cytarabine); R-hyperCVAD (rituximab-hyperCVAD); FCM (fludarabine, cyclophosphamide, mitoxantrone); R-FCM (rituximab, fludarabine, cyclophosphamide, mitoxantrone); bortezomib and rituximab; temsirolimus and rituximab; temsirolimus and Velcade.RTM.; Iodine-131 tositumomab (Bexxar.RTM.) and CHOP; CVP (cyclophosphamide, vincristine, prednisone); R-CVP (rituximab-CVP); ICE (iphosphamide, carboplatin, etoposide); R-ICE (rituximab-ICE); FCR (fludarabine, cyclophosphamide, rituximab); FR (fludarabine, rituximab); and D.T. PACE (dexamethasone, thalidomide, cisplatin, Adriamycin.RTM., cyclophosphamide, etoposide).
Examples of immunotherapeutic agents treating lymphoma or leukemia include, but are not limited to, rituximab (such as Rituxan), alemtuzumab (such as Campath, MabCampath), anti-CD19 antibodies, anti-CD20 antibodies, anti -MN- 14 antibodies, anti- TRAIL, Anti-TRAIL DR4 and DR5 antibodies, anti-CD74 antibodies, apolizumab, bevacizumab, CHIR-12.12, epratuzumab (hLL2-anti-CD22 humanized antibody), galiximab, ha20, ibritumomab tiuxetan, lumiliximab, milatuzumab, ofatumumab, PRO131921, SGN-40, WT-1 analog peptide vaccine, WT1 126-134 peptide vaccine, tositumomab, autologous human tumor-derived HSPPC-96, and veltuzumab. Additional immunotherapy agents includes using cancer vaccines based upon the genetic makeup of an individual patient's tumor, such as lymphoma vaccine GTOP-99.
The therapeutic treatments can be supplemented or combined with any of the abovementioned therapies with stem cell transplantation or treatment. One example of modified approach is radioimmunotherapy, wherein a monoclonal antibody is combined with a radioisotope particle, such as indium In-111, yttrium Y-90, iodine 1-131. Examples of combination therapies include, but are not limited to, Iodine-131 tositumomab, Yttrium-90 ibritumomab tiuxetan with CHOP.
The compounds of the application can be used in combination with additional therapeutic procedures. Other therapeutic procedures include peripheral blood stem cell transplantation, autologous hematopoietic stem cell transplantation, autologous bone marrow transplantation, antibody therapy, biological therapy, enzyme inhibitor therapy, total body irradiation, infusion of stem cells, bone marrow ablation with stem cell support, in vitro-treated peripheral blood stem cell transplantation, umbilical cord blood transplantation, immunoenzyme technique, pharmacological study, low-LET cobalt-60 gamma ray therapy, bleomycin, conventional surgery, radiation therapy, and nonmyeloablative allogeneic hematopoietic stem cell transplantation.
The compounds of the application can be used in combination with anti-fibrotic agents. The anti-fibrotic agents include, but are not limited to, emylenemamine, hydrazine, phenylhydrazine, and their derivatives, semicarbazide, and urea derivatives, aminonitriles, such as beta-aminopropionitrile (BAPN), or 2-nitroethylamine, unsaturated or saturated haloamines, such as 2-bromo-ethylamine, 2-chloroethylamine, 2- trifluoroethylamine, 3 -bromopropylamine, p-halobenzylamines, selenohomocysteine lactone. Also, the anti-fibrotic agents are copper chelating agents, penetrating or not
penetrating the cells. Exemplary compounds include indirect inhibitors such compounds blocking the aldehyde derivatives originating from the oxidative deamination of the lysyl and hydroxylysyl residues by the lysyl oxidases, such as the thiolamines, in particular D- penicillamine, or its analogues such as 2-amino-5-mercapto-5-methylhexanoic acid, D-2- amino-3-methyl-3-((2-acetamidoethyl)dithio)butanoic acid, p-2-amino-3-methyl-3-((2- aminoethyl)dithio)butanoic acid, sodium -4-((p-l -dimethyl-2-amino-2- carboxyethyl)dithio)butane sulphurate, 2-acetamidoethyl-2-acetamidoethanethiol sulphanate, sodium-4-mercaptobutanesulphinate trihydrate.
The compounds of the application can be used in combination with immunotherapeutic and anti-inflammatory treatments. The immunotherapeutic agents include and are not limited to therapeutic antibodies suitable for treating patients; such as abagovomab, adecatumumab, afutuzumab, alemtuzumab, altumomab, amatuximab, anatumomab, arcitumomab, bavituximab, bectumomab, bevacizumab, bivatuzumab, blinatumomab, brentuximab, cantuzumab, catumaxomab, cetuximab, citatuzumab, cixutumumab, clivatuzumab, conatumumab, daratumumab, drozitumab, duligotumab, dusigitumab, detumomab, dacetuzumab, dalotuzumab, ecromeximab, elotuzumab, ensituximab, ertumaxomab, etaracizumab, farietuzumab, ficlatuzumab, figitumumab, flanvotumab, futuximab, ganitumab, gemtuzumab, girentuximab, glembatumumab, ibritumomab, igovomab, imgatuzumab, indatuximab, inotuzumab, intetumumab, ipilimumab, iratumumab, labetuzumab, lexatumumab, lintuzumab, lorvotuzumab, lucatumumab, mapatumumab, matuzumab, milatuzumab, minretumomab, mitumomab, moxetumomab, narnatumab, naptumomab, necitumumab, nimotuzumab, nofetumomabn, ocaratuzumab, ofatumumab, olaratumab, onartuzumab, oportuzumab, oregovomab, panitumumab, parsatuzumab, patritumab, pemtumomab, pertuzumab, pintumomab, pritumumab, racotumomab, radretumab, rilotumumab, rituximab, robatumumab, satumomab, sibrotuzumab, siltuximab, simtuzumab, solitomab, tacatuzumab, taplitumomab, tenatumomab, teprotumumab, tigatuzumab, tositumomab, trastuzumab, tucotuzumab, ublituximab, veltuzumab, vorsetuzumab, votumumab, zalutumumab, CC49 and 3F8. The exemplified therapeutic antibodies may be further labeled or combined with a radioisotope particle, such as indium In-111, yttrium Y-90, iodine 1-131.
In one aspect, the immuno-oncology agent is (i) an agonist of a stimulatory (including a co-stimulatory) receptor or (ii) an antagonist of an inhibitory (including a co-
inhibitory) signal on T cells, both of which result in amplifying antigen-specific T cell responses (often referred to as immune checkpoint regulators).
Certain of the stimulatory and inhibitory molecules are members of the immunoglobulin super family (IgSF). One important family of membrane-bound ligands that bind to co-stimulatory or co-inhibitory receptors is the B7 family, which includes B7- 1, B7-2, B7-H1 (PD-L1), B7-DC (PD-L2), B7-H2 (ICOS-L), B7-H3, B7-H4, B7-H5 (VISTA), and B7-H6. Another family of membrane bound ligands that bind to costimulatory or co-inhibitory receptors is the TNF family of molecules that bind to cognate TNF receptor family members, which includes CD40 and CD40L, OX-40, OX-40L, CD70, CD27L, CD30, CD30L, 4-1BBL, CD137 (4-1BB), TRAIL/Apo2-L, TRAILR1/DR4, TRAILR2/DR5, TRAILR3, TRAILR4, OPG, RANK, RANKL, TWEAKR/Fnl4, TWEAK, BAFFR, ED AR, XEDAR, TACI, APRIL, BCMA, LTpR, LIGHT, DcR3, HVEM, VEGI/TL1A, TRAMP/DR3, ED AR, EDAI, XEDAR, EDA2, TNFR1, Lymphotoxin a/TNFp, TNFR2, TNFa, LTpR, Lymphotoxin a ip2, FAS, FASL, RELT, DR6, TROY, NGFR.
In one aspect, T cell responses can be stimulated by a combination of a compound of Formula (I) and one or more of (i) an antagonist of a protein that inhibits T cell activation (e.g., immune checkpoint inhibitors) such as CTLA-4, PD-1, PD-L1, PD-L2, LAG-3, TIM-3, Galectin 9, CEACAM-1, BTLA, CD69, Galectin-1, TIGIT, CD113, GPR56, VISTA, 2B4, CD48, GARP, PD1H, LAIR1, TIM-1, and TIM-4, and (ii) an agonist of a protein that stimulates T cell activation such as B7-1, B7-2, CD28, 4-1BB (CD137), 4-1BBL, ICOS, ICOS-L, 0X40, OX40L, GITR, GITRL, CD70, CD27, CD40, DR3 and CD28H.
Other agents that can be combined with compounds described herein for the treatment of cancer include antagonists of inhibitory receptors on NK cells or agonists of activating receptors on NK cells. For example, compounds described herien can be combined with antagonists of KIR, such as lirilumab.
Yet other agents for combination therapies include agents that inhibit or deplete macrophages or monocytes, including but not limited to CSF-1R antagonists such as CSF-1R antagonist antibodies including RG7155 (WO11/70024, WO11/107553, WO1 1/131407, WO13/87699, WO13/119716, WO13/132044) or FPA-008 (WO11/140249; WO13169264; WO14/036357).
In another aspect, compounds of the present application can be used with one or more of agonistic agents that ligate positive costimulatory receptors, blocking agents that attenuate signaling through inhibitory receptors, antagonists, and one or more agents that increase systemically the frequency of anti-tumor T cells, agents that overcome distinct immune suppressive pathways within the tumor microenvironment (e.g., block inhibitory receptor engagement (e.g., PD-L1/PD-1 interactions), deplete or inhibit Tregs (e.g., using an anti-CD25 monoclonal antibody (e.g., daclizumab) or by ex vivo anti-CD25 bead depletion), inhibit metabolic enzymes such as IDO, or rever se/prevent T cell anergy or exhaustion) and agents that trigger innate immune activation and/or inflammation at tumor sites.
In one aspect, the immuno-oncology agent is a CTLA-4 antagonist, such as an antagonistic CTLA-4 antibody. Suitable CTLA-4 antibodies include, for example, YERVOY (ipilimumab) or tremelimumab.
In another aspect, the immuno-oncology agent is a PD-1 antagonist, such as an antagonistic PD-1 antibody. Suitable PD-1 antibodies include, for example, OPDIVO (nivolumab), KEYTRUDA (pembrolizumab), or MEDL0680 (AMP-514; WO2012/145493). The immuno-oncology agent may also include pidilizumab (CT-011), though its specificity for PD-1 binding has been questioned. Another approach to target the PD-1 receptor is the recombinant protein composed of the extracellular domain of PD-L2 (B7-DC) fused to the Fc portion of IgGl, called AMP-224
In another aspect, the immuno-oncology agent is a PD-L1 antagonist, such as an antagonistic PD-L1 antibody. Suitable PD-L1 antibodies include, for example, MPDL3280A (RG7446; WO2010/077634), durvalumab (MEDI4736), BMS-936559 (W02007/005874), and MSB0010718C (WO2013/79174).
In another aspect, the immuno-oncology agent is a LAG-3 antagonist, such as an antagonistic LAG-3 antibody. Suitable LAG3 antibodies include, for example, BMS- 986016 (W010/19570, WO14/08218), or IMP-731 or IMP-321 (W008/132601, WO09/44273).
In another aspect, the immuno-oncology agent is a CD137 (4-1BB) agonist, such as an agonistic CD137 antibody. Suitable CD137 antibodies include, for example, urelumab and PF-05082566 (WO12/32433).
In another aspect, the immuno-oncology agent is a GITR agonist, such as an agonistic GITR antibody. Suitable GITR antibodies include, for example, BMS-986153, BMS-986156, TRX-518 (WO06/105021, W009/009116) and MK-4166 (WO 11/028683).
In another aspect, the immuno-oncology agent is an IDO antagonist. Suitable IDO antagonists include, for example, INCB-024360 (W02006/122150, WO07/75598, WO08/36653, WO08/36642), indoximod, BMS-986205, or NLG-919 (W009/73620, WO09/1156652, WO11/56652, WO12/142237).
In another aspect, the immuno-oncology agent is an 0X40 agonist, such as an agonistic 0X40 antibody. Suitable 0X40 antibodies include, for example, MEDI-6383 or MEDI-6469.
In another aspect, the immuno-oncology agent is an OX40L antagonist, such as an antagonistic 0X40 antibody. Suitable OX40L antagonists include, for example, RG-7888 (WO06/029879).
In another aspect, the immuno-oncology agent is a CD40 agonist, such as an agonistic CD40 antibody. In yet another embodiment, the immuno-oncology agent is a CD40 antagonist, such as an antagonistic CD40 antibody. Suitable CD40 antibodies include, for example, lucatumumab or dacetuzumab.
In another aspect, the immuno-oncology agent is a CD47 antagonist, such as a CD47 antagonist selected from the group MIAP301, MIAP410, TTI-621, CV1, Hu5F9- G4, CC-90002, B6H12 and 2D3.
In another aspect, the immuno-oncology agent is a CD27 agonist, such as an agonistic CD27 antibody. Suitable CD27 antibodies include, for example, varlilumab.
In another aspect, the immuno-oncology agent is MGA271 (to B7H3) (WO 11/109400).
The combination therapy is intended to embrace administration of these therapeutic agents in a sequential manner, that is, wherein each therapeutic agent is administered at a different time, as well as administration of these therapeutic agents, or at least two of the therapeutic agents, in a substantially simultaneous manner. Substantially simultaneous administration can be accomplished, for example, by administering to the subject a single dosage form having a fixed ratio of each therapeutic agent or in multiple, single dosage forms for each of the therapeutic agents. Sequential or substantially simultaneous administration of each therapeutic agent can be effected by
any appropriate route including, but not limited to, oral routes, intravenous routes, intramuscular routes, and direct absorption through mucous membrane tissues. The therapeutic agents can be administered by the same route or by different routes. For example, a first therapeutic agent of the combination selected may be administered by intravenous injection while the other therapeutic agents of the combination may be administered orally. Alternatively, for example, all therapeutic agents may be administered orally or all therapeutic agents may be administered by intravenous injection. Combination therapy also can embrace the administration of the therapeutic agents as described above in further combination with other biologically active ingredients and non-drug therapies (e.g., surgery or radiation treatment.) Where thwe combination therapy further comprises a non drug treatment, the non drug treatment may be conducted at any suitable time so long as a beneficial effect from the co-action of the combination of the therapeutic agents and non-drug treatment is achieved. For example, in appropriate cases, the beneficial effect is still achieved when the non drug treatment is temporally removed from the administration of the therapeutic agents, perhaps by days or even weeks.
The present invention also provides the compounds of the present invention for use in therapy.
In another embodiment, compounds of Formula I are selected from exemplified compounds or combinations of exemplified compounds or other embodiments herein.
In another embodiment are compounds having an IC50 < 1000 nM in at least one of the assays described below.
The present invention may be embodied in other specific forms without departing from the spirit or essential attributes thereof. This invention encompasses all combinations of preferred aspects and/or embodiments of the invention noted herein. It is understood that any and all embodiments of the present invention may be taken in conjunction with any other embodiment or embodiments to describe additional more preferred embodiments. It is also to be understood that each individual element of the preferred embodiments is its own independent preferred embodiment. Furthermore, any element of an embodiment is meant to be combined with any and all other elements from any embodiment to describe an additional embodiment.
METHODS OF PREPARATION
The compounds of the present invention can be prepared in a number of ways well known to one skilled in the art of organic synthesis. The compounds of the present invention can be synthesized using the methods described below, together with synthetic methods known in the art of synthetic organic chemistry, or variations thereon as appreciated by those skilled in the art. Preferred methods include, but are not limited to, those described below. All references cited herein are hereby incorporated in their entirety by reference.
The compounds of this invention may be prepared using the reactions and techniques described in this section. The reactions are performed in solvents appropriate to the reagents and materials employed and are suitable for the transformations being effected. Also, in the description of the synthetic methods described below, it is to be understood that all proposed reaction conditions, including choice of solvent, reaction atmosphere, reaction temperature, duration of the experiment and work up procedures, are chosen to be the conditions standard for that reaction, which should be readily recognized by one skilled in the art. It is understood by one skilled in the art of organic synthesis that the functionality present on various portions of the molecule must be compatible with the reagents and reactions proposed. Such restrictions to the substituents that are compatible with the reaction conditions will be readily apparent to one skilled in the art and alternate methods must then be used. This will sometimes require a judgment to modify the order of the synthetic steps or to select one particular process scheme over another in order to obtain a desired compound of the invention. It will also be recognized that another major consideration in the planning of any synthetic route in this field is the judicious choice of the protecting group used for protection of the reactive functional groups present in the compounds described in this invention. An authoritative account describing the many alternatives to the trained practitioner is Greene and Wuts (Protective Groups In Organic Synthesis, Third Edition, Wiley and Sons, 1999).
EXAMPLES
Preparation of compounds of Formula I and intermediates used in the preparation of compounds of Formula I can be prepared using procedures shown in the following Examples and related procedures. The methods and conditions used in these examples,
and the actual compounds prepared in these Examples, are not meant to be limiting, but are meant to demonstrate how the compounds of Formula I can be prepared. Starting materials and reagents used in these examples, when not prepared by a procedure described herein, are generally either commercially available, or are reported in the chemical literature, or may be prepared by using procedures described in the chemical literature.
The following examples illustrate the particular and preferred embodiments of the present invention and do not limit the scope of the present invention. Chemical abbreviations and symbols as well as scientific abbreviations and symbols have their usual and customary meanings unless otherwise specified. Common intermediates are generally useful for the preparation of more than one Example as shown in the Tables.
Chemical names were determined using ChemB ioDraw Ultra, version 14.0.0.126 (CambridgeSoft).
LCMS-Method 1:
COLUMN: Kinetex XB-C 18(75X3.0) mm, 2.6um
Mobile phase A: 5mm Ammonium formate pH 3.3: Acetonitrile (98:2) Mobile phase B: ACN :5mm Ammonium formate pH 3.3 (98:2)
Flow rate: 1 ml/min
Temperature: 25° C
GRADIENT: 20%B to 100%B over 4min, 1.5 ml/min flow at 0.5 min hold at 100%B
MS and UV: 220 & 254
LCMS-Method 2:
COLUMN: Kinetex -C18(75X3.0) mm, 2.6um
Mobile phase A: 0.1% TFA in water
Mobile phase B: 0.1% TFA in Acetonitrile
Flow rate: 1 ml/min
Temperature: 25° C
GRADIENT: 05%B to 95%B over 2.5min, 1.5 ml\min flow at 1.49min hold at 95%B
MS and UV: 220 & 254 nm
LCMS-Method 3:
COLUMN: Acquity BEH Cl 8(50X3.0) mm, 1.7um
Mobile phase A: 5mm Ammonium formate pH 3.3: Acetonitrile (98:2)
Mobile phase B: ACN :5mm Ammonium formate pH 3.3 (98:2)
Flow rate: 0.7 ml/min
Temperature: 25° C
GRADIENT: 20%B to 98%B over 1.5min, 0.5 min hold at 98%B
MS and UV: 220 nm
LCMS-Method 4:
COLUMN: Acquity Uplc BEH Cl 8(50X3.0) mm, 1.7um
Mobile phase A: 5mm Ammonium formate pH 3.3: Acetonitrile (98:2)
Mobile phase B: ACN :5mm Ammonium formate pH 3.3 (98:2)
Flow rate: 0.7 ml/min
Temperature: 25° C
GRADIENT: 20%B to 98%B over 1.5min, 0.5 min hold at 98%B
MS and UV: 220 nm
LCMS-Method 5:
COLUMN: Kinetex XB-C 18(75X3.0) mm, 2.6um
Mobile phase A: 0.1% TFA in water
Mobile phase B: 0.1% TFA in Acetonitrile
Flow rate: 1 ml/min
Temperature: 25° C
GRADIENT: 05%B to 95%B over 2.5min, 1.5 ml\min flow at 1.49 min hold at 95%B
MS and UV: 220 & 254 nm
LCMS-Method 6:
COLUMN: Kinetex -C18(75X3.0) mm, 2.6um
Mobile phase A: 5mm Ammonium formate pH 3.3: Acetonitrile (98:2)
Mobile phase B: ACN :5mm Ammonium formate pH 3.3 (98:2)
Flow rate: 0.7 ml/min
Temperature: 25° C
GRADIENT: 20%B to 100%B over 4min, 1.5 ml/min flow at 0.5 min hold at 100%B
MS and UV: 220 & 254
LCMS-Method 7:
COLUMN: Acquity BEH Cl 8(50X3.0) mm, 1.7um
Mobile phase A: 0.1% TFA in water
Mobile phase B: 0.1% TFA in Acetonitrile
Flow rate: 0.7 ml/min
Temperature: 25° C
GRADIENT: 20%B to 98%B over 1.5min, 0.5 min hold at 98%B
MS and UV: 220 nm
LCMS-Method 8:
COLUMN: X-bridge C8(50X4.5) mm, 5um
Mobile phase A: 0.1% TFA in water
Mobile phase B: 0.1% TFA in Acetonitrile
Flow rate: 1 ml/min
Temperature: Ambient
GRADIENT: 5%B to 95%B over 2.5min, 1.5ml/min flow at 1.5 min hold at 95%B
MS and UV: 220
LCMS-Method 9:
COLUMN: Waters Acquity BEH C18 1.7um 2.1 x 50 mm
Mobile phase A: 0.05% TFA in CH3CN:Water (5:95)
Mobile phase B: 0.05% TFA in CH3CN:Water (95:5)
Flow rate: 1 ml/min
Temperature: 50° C
GRADIENT: 0%B to 100%B over 1 min, stop time 1.5 min.
MS and UV: 220, 254
LCMS-Method 10:
COLUMN: XBridge C18, 2.1 mm x 50 mm, 1.7 pm particles
Mobile phase A: 0.05% TFA in CH3CN:Water (5:95)
Mobile phase B: 0.05% TFA in CH3CN:Water (95:5)
Flow rate: 1 ml/min
Temperature: 50° C
GRADIENT: 0-100 %B (0-3 min), 100 %B (3-3.5 min).
MS and UV: 220
A mixture of 3, 3 -dibromo- 1,1,1 -trifhioropropan-2-one (49.3 g, 183 mmol) and sodium acetate (19.99 g, 244 mmol) in water (100 mL) was stirred at 95 °C for 30 min. The reaction mixture was cooled to 0 °C and a cold solution of methyl 4-formylbenzoate (20.00 g, 122.00 mmol) in a mixture of NH4OH (28% aq, 100 mL) and MeOH (300 mL) was added. The resulting mixture was stirred at 25 °C for 16 h, Volatiles were evaporated, the residue obtained was diluted with ethyl acetate and washed with water. The organic extract was dried over anhydrous Na2SO4, filtered and concentrated. The residue obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain methyl 4-(4- (trifluorornethyl)-lJ/-imidazol-2-yl)benzoate (19.00 g, 60.5 mmol, 49.7%) as yellow solid. LCMS (ESI) m/z: 271.0 [M+H]+, LC retention time: 1.206 min (LCMS Method 3).
To a stirred solution of methyl 4-(4-(trifluoromethyl)-lH-imidazol-2-yl)benzoate (2.50 g, 9.25 mmol) in DMF (20 mL), sodium hydride (60% dispersion in mineral oil, 0.740 g, 18.50 mmol) was added followed by addition of iodomethane (0.868 mL, 13.88 mmol) at 0 °C. The reaction mixture was warmed to RT, stirred at RT for 3 h, quenched with chilled water and extracted with ethyl acetate. The organic extract was dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was
purified by flash chromatography on silica gel, 230-400 mesh to obtain methyl 4-(l- methyl-4-(trifluoromethyl)-lJ/-imidazol-2-yl)benzoate (1.40 g , 4.87 mmol, 52.7 %) as white solid. LCMS (ESI) m/z: 285.0 [M+H]+, LC retention time: 1.280 min (LCMS Method 3).
To a stirred solution of methyl 4-(l-methyl-4-(trifluorom ethyl)- lJT-imidazol-2- yl)benzoate (1.20 g, 4.22 mmol) in THF (25 mL), lithium aluminium hydride (2 M in THF, 4.22 mL, 8.44 mmol) was added at 0 °C and the mixture was stirred at 25 °C for 3 h. The reaction was quenched with saturated aqueous NH4Q and extracted with ethyl acetate. The organic extract was dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography on silica gel, 230-400 mesh, to yield (4-(l-methyl-4-(trifluoromethyl)-17/-imidazol-2- yl)phenyl)methanol (0.900 g, 3.42 mmol, 81%) as white solid. LCMS (ESI) m/z: 257.0 [M+H]+, LC retention time: 0.940 min (LCMS Method 3).
Synthesis of 1-2
Step-1:: Methyl 4-( l-isopropyl-4-(trifluoromethyl)-lZ/-imidazol-2-yl)benzoate:
To a stirred solution of methyl 4-(4-(trifluoromethyl)- l7/-imidazol-2-yl)benzoate (2.0 g, 7.40 mmol) in acetonitrile (30 mL), CS2CO3 (4.82 g, 14.80 mmol) was added followed by addition of 2-iodopropane (1.110 mL, 11.10 mmol) at 0 °C. The reaction mixture was stirred at 25 °C for 16 h and heated at 50 °C for an additional 16 h, cooled to RT, concentrated under reduced pressure, diluted with ethyl acetate and washed with water. The ethyl acetate layer was dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography on silica gel, 230- 400 mesh, to obtain methyl 4-(l-isopropyl-4-(trifluoromethyl)-lJ/-imidazol-2-yl)benzoate (0.800 g, 2.229 mmol, 30.1%) as white solid. LCMS (ESI) m/z: 313.2 [M+H]+, LC retention time: 1.434 min (LCMS Method 3).
To a stirred solution of methyl 4-(l-isopropyl-4-(trifluoromethyl)-17/-imidazol-2- yl)benzoate (0.70 g, 2.241 mmol) in THF (10 mL), lithium aluminium hydride (2 M in THF, 2.241 mL, 4.48 mmol) was added at 0 °C and the mixture was stirred at 25 °C for 3 h. The reaction was quenched with saturated aqueous NH4Q and extracted with ethyl acetate. The organic extract was dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure. The residue was purified by flash chromatography on silica gel, 230-400 mesh, to yield (4-(l-isopropyl-4-(trifluorornethyl)-lJ/-imidazol-2- yl)phenyl)methanol (0.650 g, 2.242 mmol, 100%) as white solid. LCMS (ESI) m/z: 285.0 [M+H]+, LC retention time: 1.127 min (LCMS Method 3).
Scheme 2: Synthesis of Intermediate 3: (4-(5-methyl-3-(trifluoromethyl)-lH-pyrazol- l-yl)phenyl)methanol:
To a stirred solution of methyl 4-(5-methyl-3-(trifluoromethyl)-lH-pyrazol-l-yl)benzoate (5.0 g, 17.59 mmol) in tetrahydrofuran (20 mL), DIBA1-H (1.2 M in Toluene, 44.0 mL, 52.8 mmol) was added at 0 °C and the mixture was stirred at ambient temperature for 3 h. The reaction mixture was slowly poured into chilled water containing crushed ice and filtered through a celite bed. The filtrate was extracted with ethyl acetate and solvents evaporated under reduced pressure to yield (4-(5-methyl-3-(trifluoromethyl)-lH-pyrazol- l-yl)phenyl)methanol (3.8 g, 14.24 mmol, 81%) as pale yellow liquid. LCMS (ESI) m/z: 257.0 [M+H]+, LC retention time: 1.224 min. (LCMS Method 3).
Step-1: 4-chloro-l-isopropyl-lH-pyrazole:
To a stirred solution of 4-chloro-lH-pyrazole (5.0 g, 48.8 mmol) in acetonitrile (60 mL), CS2CO3 (31.8 g, 98 mmol) was added followed by addition of 2-iodopropane (5.69 mL, 58.5 mmol) and the contents heated at 80 °C for 2 h. The reaction mixture was cooled to ambient temperature, diluted with water and extracted with diethyl ether. The organic extract was dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure, at ~35 °C, to obtain crude 4-chloro-l-isopropyl-lH-pyrazole (6.0 g, 39.4 mmol, 81%) as pale yellow oil, which was used as such for the next step. LCMS (ESI) m/z: 145.0 [M+H]+, LC retention time: 1.187 min. (LCMS Method 7).
Step-2: Synthesis of 4-chloro-l-isopropyl-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan- 2-yl)-lH-pyrazole (1-4):
To a stirred solution of 4-chloro-l-isopropyl-lH-pyrazole (5.50 g, 38.0 mmol) in tetrahydrofuran (50 mL), n-butyllithium (1.6 M in hexanes, 28.5 mL, 45.6 mmol) was added at 0 °C and the reaction mixture was stirred at ambient temperature for 1 h. The reaction mixture was cooled to -78 °C, 2-isopropoxy-4,4,5,5-tetramethyl-l,3,2- dioxaborolane (8.49 g, 45.6 mmol) was added and stirred for 2 h. Saturated aqueous NH4Q was added to the reaction mixture and the contents extracted with ethyl acetate. The organic extract was dried over anhydrous Na2SO4, filtered and concentrated under reduced pressure to obtained crude 4-chloro-l-isopropyl-5-(4,4,5,5-tetramethyl-l,3,2- dioxaborolan-2-yl)-lH-pyrazole (6.00 g, 14.41 mmol, 37.9%) as dark brown oil, which was used as such for the subsequent step. LCMS (ESI) m/z: 271.0 [M+H]+, LC retention time: 1.327 min. (LCMS Method 4).
Scheme 4: General Synthetic sequence for final targets. Representative example shown below
Step-1: Synthesis of 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)pyrido [3, 2-d] pyrimidine:
To a stirred solution of 2,4-dichloropyrido[3,2-d]pyrimidine (0.200 g, 1.00 mmol) in acetonitrile (10 mL), CS2CO3 (0.489 g, 1.50 mmol) and (4-(l-methyl-4-(trifluoromethyl)- lH-imidazol-2-yl)phenyl)methanol (0.256 g, 1.00 mmol) were added and the mixture was heated at 60 °C for 16 h. The reaction mixture was cooled to ambient temperature, chilled water was added and the contents extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered and concentrated under reduced
pressure. The residue obtained was purified by flash chromatography on silica gel using RediSep cartridgre to yield 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)pyrido[3,2-d]pyrimidine (0.214 g, 0.484 mmol, 48.4%) as light brown solid. LCMS (ESI) m/z: 420.0 [M+H]+,LC retention time: 2.65 min (LCMS method 1).
Intermediate in Table 1 were prepared according to procedure described in step-1 above.
Step-2 : Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido [3,2-d] pyrimidine (Example 1):
To a stirred solution of 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)pyrido[3,2-d]pyrimidine (200 mg, 0.476 mmol) (0.200 g, 0.476 mmol) in a mixture of 1,4-dioxane and water (4 mL, 10 : 1), tripotassium phosphate (0.202 g, 0.953 mmol), 4-cyclopropyl-6-methoxy-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2- yl)pyrimidine (0.145 g, 0.524 mmol) and tetrakis(triphenylphosphine)palladium(0) (0.0551 g, 0.048 mmol) were added under nitrogen atmosphere and the mixture was heated at 80 °C for 16 h. The reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was washed with ethyl acetate. The filtrate was concentrated under reduced pressure and the residue was purified by reverse phase preparative HPLC [Method: Diluent: THF : WATER:MeCN (40:10:40); Column: XSelect- C181 (250 xl9) mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium Formate in Water, Mobile phase B: Acetonitrile; Flow : 15mL/min; Time/Grad : 0/50,7/80,10/80] to yield 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4- (1 -methyl-4-(tri fluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)pyrido[3,2-d]pyrimidine (0.060 g, 0.112 mmol, 23.52%) as white solid. LCMS (ESI) m/z: 534.0 [M+H]+, LC retention time: 2.69 min. (LCMS Method 1); 'H-NMR: (400 MHz,DMSO-dd) 3 = 9.07 (q, J= 2.8 Hz & 1.6 Hz, 1H), 8.72 (s, 1H), 8.42 (dd, J= 7.2 Hz & 1.6 Hz, 1H), 8.02 (dd, J = 4.4 Hz & 4.4 Hz, 1H), 7.95 (d, J= 1.2 Hz, 1H), 7.78 (d, J= 6.4 Hz, 2H), 7.70 (d, J= 8.0 Hz, 2H), 5.74 (s, 2H), 3.88 (s, 3H), 3.81 (s, 3H), 1.89-1.81 (m, 1H), 1.12-1.05 (m, 2H), 0.93-0.85 (m, 2H).
Examples in Table 2 were prepared according to procedure described in step-2 above.
Intermediate in Table 4 were prepared according to procedure described in step-1 above.
Examples in Table 5 were prepared according to procedure described in step-2 above.
Enantiomers of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(l-(4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)phenyl)ethoxy)pyrido[3,2-d]pyrimidine:
Compound 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(l-(4-(l-methyl-4-
(trifluoromethyl)-lH-imidazol-2-yl)phenyl)ethoxy)pyrido[3,2-d]pyrimidine was submitted to Prep SFC for chiral enantiomers separation and enantiomer- 1 and enantiomer- 2 were obtained.
SFC Method:
Column/dimensions: Luxcellulose-4(250 X 30)mm, 5 pm; % CO2: 73%; % Co solvent: 27% of MeOH; Total Flow: 70.0 g/min; Back Pressure: 100 bar; Temperature : 40 °C; UV: 220 nm. SFC retention time for enantiomer peak 1 : 6.8 min., and enantiomer peak 2: 8.0 min.
Example 24: Enantiomer-1 [SFC retention time for enantiomer (peak) 1: 6.8 min.] [a]D 25 = -12.0 (c = 0.05 in MeOH). HPLC: 99.9% ee (Cellulose-4(250X4.6) mm, 5 m_Methanol_30_2.1cd, flow rate =3.0 mL/min, = 220 nm, Co-Solvent :30.0 %, Oven- A Temperature : 40 °C, Oven A Column Position : 1, BPR Pressure : 100.0 bar, BPR Temperature : 50 °C), HPLC retention time : = 3.936 min (major), 4.533 min (minor). LC- MS (ESI) m/z: 548.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 9.10 (dd, J= 1.6, 4.2 Hz, 1H), 8.70 (s, 1H), 8.40 (dd, J= 1.5, 8.5 Hz, 1H), 8.03 (dd, J= 4.2, 8.6 Hz, 1H), 7.94 (d, J= 1.1 Hz, 1H), 7.74 (d, J= 8.4 Hz, 2H), 7.70 - 7.65 (m, 2H), 6.59 (q, J= 6.3 Hz, 1H), 3.81 (s, 3H), 3.78 (s, 3H), 1.79 (d, J= 6.5 Hz, 3H), 1.76 - 1.69 (m, 1H), 1.09 - 0.97 (m, 2H), 0.89 - 0.73 (m, 2H).
Example 25: Enantiomer-2 [SFC retention time for enantiomer (peak) 2: 8.0 min.] [a]D 25 = +12.0 (c = 0.05 in MeOH). HPLC: 98.4% ee (Cellulose-4(250X4.6) mm, 5 m_Methanol_30_2.1cd, flow rate =3.0 mL/min, = 220 nm, Co-Solvent :30.0 %, Oven- A Temperature : 40 °C, Oven A Column Position : 1, BPR Pressure : 100.0 bar, BPR
Temperature : 50 °C), HPLC retention time : = 4.501 min (major), 3.936 min (minor). LC- MS (ESI) m/z: 548.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 9.09 (dd, J= 1.6, 4.2 Hz, 1H), 8.70 (s, 1H), 8.39 (dd, J= 1.6, 8.5 Hz, 1H), 8.02 (dd, J= 4.1, 8.5 Hz, 1H), 7.93 (d, J= 1.1 Hz, 1H), 7.75 (d, J= 8.4 Hz, 2H), 7.68 - 7.63 (m, 2H), 6.58 (q, J= 6.5 Hz, 1H), 3.81 (s, 3H), 3.78 (s, 3H), 1.78 (d, J= 6.6 Hz, 3H), 1.75 - 1.69 (m, 1H), 1.09 - 0.96 (m, 2H), 0.90 - 0.72 (m, 2H).
Enantiomers of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(l-(4-(l-isopropyl-4- (trifluoromethyl)-lH-imidazol-2-yl)phenyl)ethoxy)pyrido[3,2-d]pyrimidine:
Compound 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(l-(4-(l-isopropyl-4-
(trifluoromethyl)-lH-imidazol-2-yl)phenyl)ethoxy)pyrido[3,2-d]pyrimidine was submitted to Prep SFC for chiral enantiomers separation and enantiomer- 1 and enantiomer- 2 were obtained.
SFC Method:
Column/dimensions: Luxcellulose-4(250 X 30)mm, 5 pm; % CO2: 75%; % Co solvent: 25% of MeOH; Total Flow: 100.0 g/min; Back Pressure: 100 bar; Temperature : 40 °C; UV: 220 nm. SFC retention time for enantiomer peak 1 : 4.5 min., and enantiomer peak 2: 5.5 min.
Example 26: Enantiomer-1 [SFC retention time for enantiomer (peak) 1: 4.5 min.] [a]D 25 = -12.0 (c = 0.1 in MeOH). HPLC: >99.9% ee (Cellulose-4(250X4.6) mm, 5 m_Methanol_30_3.1cd, flow rate =3.0 mL/min, = 220 nm, Co-Solvent :30.0 %, Oven- A Temperature : 40 °C, Oven A Column Position : 1, BPR Pressure : 100.0 bar, BPR Temperature : 50 °C), LC-MS (ESI) m/z: 576.2 [M+H]+. 'HNMR (400 MHz, DMSO-de) 5 = 9.10 (dd, J= 1.6, 4.3 Hz, 1H), 8.70 (s, 1H), 8.40 (dd, J= 1.6, 8.6 Hz, 1H), 8.18 (d, J =
1.1 Hz, 1H), 8.03 (dd, J= 4.2, 8.6 Hz, 1H), 7.73 - 7.64 (m, 2H), 7.61 - 7.57 (m, 2H), 6.59 (q, J= 6.5 Hz, 1H), 4.47 (spt, J= 6.7 Hz, 1H), 3.81 (s, 3H), 1.79 (d, J= 6.5 Hz, 3H), 1.76 - 1.69 (m, 1H), 1.41 (dd, J= 3.1, 6.5 Hz, 6H), 1.05 - 1.01 (m, 2H), 0.91 - 0.70 (m, 2H).
Example 27: Enantiomer-2 [SEC retention time for enantiomer (peak) 2: 5.5 min.] [a]D 25 = +14.0 (c = 1.0 in MeOH). HPLC: 99.4% ee (Cellulose-4(250X4.6) mm, 5p.m_Methanol_30_3.lcd, flow rate = 3.0 mL/min, X = 220 nm, Co-Solvent : 30.0 %, Oven-A Temperature : 40 °C, Oven A Column Position : 1, BPR Pressure : 100.0 bar, BPR Temperature : 50 °C), LC-MS (ESI) m/z: 576.2 [M+H]+. 'HNMR (400 MHz, DMSO-de) 5 = 9.09 (dd, J= 1.6, 4.1 Hz, 1H), 8.70 (s, 1H), 8.40 (dd, J= 1.6, 8.6 Hz, 1H), 8.17 (d, J= 1.3 Hz, 1H), 8.02 (dd, J= 4.3, 8.5 Hz, 1H), 7.72 - 7.65 (m, 2H), 7.61 - 7.50 (m, 2H), 6.59 (q, J= 6.4 Hz, 1H), 4.46 (spt, J= 6.7 Hz, 1H), 3.81 (s, 3H), 1.78 (d, J= 6.5 Hz, 3H), 1.76 - 1.68 (m, 1H), 1.40 (dd, J= 3.1, 6.6 Hz, 6H), 1.07 - 0.99 (m, 2H), 0.90 - 0.69 (m, 2H).
Intermediate 24: 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenoxy)pyrido[3,2-d]pyrimidine
To a stirred solution of 2,4-dichloropyrido[4,3-d]pyrimidine (300 mg, 1.500 mmol) in acetonitrile (10 mL), CS2CO3 (977 mg, 3.00 mmol) was added followed by addition of 4-
(l-methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)phenol (291 mg, 1.200 mmol) at 25 °C and the mixture was stirred at 80 °C for 16 h. After completion of the reaction (monitored by TLC), volatiles were removed under reduced pressure, diluted with ethyl acetate and washed with water. The organic extract was dried over anhydrous Na2SO4, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, using 0-30% gradient of ethyl acetate in petroleum ether to obtain 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-
imidazol-2-yl)phenoxy)pyrido[3,2-d]pyrimidine (160 mg, 0.371 mmol, 24.71 % yield) as off-white solid. LC-MS (ESI) m/z: 406.0 [M+H]+.
Intermediate in Table 6 were prepared according to the procedure described above.
Example 28: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(4-(l-methyl-
4-(trifluoromethyl)-lH-imidazol-2-yl)phenoxy)pyrido[3,2-d]pyrimidine:
To a stirred solution of 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenoxy)pyrido[3,2-d]pyrimidine (160 mg, 0.394 mmol) in a mixture of 1,4-dioxane and water (10 mL, 9: 1), potassium phosphate tribasic (167 mg, 0.789 mmol), 4- cyclopropyl-6-methoxy-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)pyrimidine (120 mg, 0.434 mmol) and tetrakis(triphenylphosphine)palladium(0) (45.6 mg, 0.039 mmol) were added under nitrogen atmosphere and the mixture was heated at 120 °C for 2 h under microwave irradiation. After completion of the reaction (monitored by UPLC-MS and TLC), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC [Method: Diluent: THF:WATER:MeCN (40:20:40); Column: X-select C18 (150 X 19) mm, 5 micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium Formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/60, 12/90] to obtain 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(4-(l-methyl-4-(trifluoromethyl)-lH- imidazol-2-yl)phenoxy)pyrido[3,2-d]pyrimidine (85 mg, 0.160 mmol, 40.6 % yield) as white solid. LC-MS (ESI) m/z: 520.1 [M+H]+. 'H-NMR: (400 MHz, DMSO-d6) 3 = 9.19 (dd, J = 1.6, 4.2 Hz, 1H), 8.64 (s, 1H), 8.51 (dd, J = 1.2, 8.6 Hz, 1H), 8.12 (dd, J = 4.4, 8.6 Hz, 1H), 7.96 (q, J = 1.2 Hz, 1H), 7.85 (d, J = 2.0, 6.4 Hz, 2H), 7.56 (d, J = 6.8 Hz, 2H), 3.85 (s, 3H), 3.83 (s, 3H), 2.02-1.96 (m, 1H), 1.04-1.01 (m, 2H), 0.94-0.92 (m, 2H).
Examples in Table 7 were prepared according to the procedure described above.
To a stirred solution of (4-(l-methyl-4-(trifluorom ethyl)- lJT-imidazol-2- yl)phenyl)methanol (2.40 g, 9.40 mmol) in DCM (50 mL) were added triphenylphosphine (3.69 g, 14.1 mmol) and carbon tetrabromide (6.21 g, 18.7 mmol) at 0 °C. The reaction mixture was stirred at RT for 16 hrs. The reaction was diluted with DCM, washed with
water, the organic layer dried over Na2SO4, filtered and concentrated. The crude product was purified by column chromatography using 230-400 silica to afford 2-(4-(- (bromomethyl)phenyl)- l-methyl-4-(trifluorom ethyl)- Uf-imidazole (2.00 g, 5.3 mmol, 57 % yield) as a white solid: LC-MS (ESI) m/z: 319.0 [M+H]+. 'H-NMR: (400 MHZ,CDC13) 3 = 7.63-7.61 (m, 2H), 7.53-7.51 (m, 2H), 7.34 (s, IH), 4.55 (s, 2H),3.81 (s, 3H).
Intermediate in Table 8 were prepared according to procedure described above.
Intermediate 29: 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)pyrido[3,2-d]pyrimidine:
To an oven dried flask flushed with N2 at RT, were added zinc (327 mg, 5.00 mmol) and Tetrahydrofuran (10 mL), followed by 1,2-dibromoethane (0.022 mL, 0.250 mmol) and TMS-C1 (0.032 mL, 0.250 mmol). Contents heated to 62 °C under a N2 atmosphere. After 60 min, a solution of 2-(4-(bromomethyl)phenyl)-l-methyl-4- (trifluoromethyl)-lH-imidazole (877 mg, 2.75 mmol) in THF (5 mL) was added. The reaction was stirred at 62 °C under N2 for 10 min, then treated with a solution of 2,4- dichloropyrido[3,2-d]pyrimidine (500 mg, 2.500 mmol) in THF (5 mL) and tetrakis (289 mg, 0.250 mmol). The mixture was stirred at 65 °C for 2 h. After completion of reaction
(monitored by UPLC), the reaction mixture was filtered through celite bed and Concentrated under reduced pressure. The crude was purified by column chromatography using 60-120 silica, eluted with 0-50% ethylacetate in petether. Yield 2- chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)pyrido[3,2- d]pyrimidine (150 mg, 0.334 mmol, 13.37 % yield) as pale yellow gummy solid. LC-MS (ESI) m/z: 404.0 [M+H]+
Intermediate in Table 9 were prepared according to the procedure described above. Table 9
Scheme : Synthesis of example 31 and 32
Example 31 and 32: 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)pyrido [3,2-d] pyrimidine and (2-(4- cyclopropyl-6-methoxypyrimidin-5-yl)pyrido[3,2-d]pyrimidin-4-yl)(4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)phenyl)methanone:
To a stirred solution of 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)pyrido[3,2-d]pyrimidine (150 mg, 0.371 mmol)) and (4-cyclopropyl-6- methoxypyrimidin-5-yl)boronic acid (108 mg, 0.557 mmol) in 1,4-Dioxane (10 mL) were added Potassium phosphate tribasic (237 mg, 1.114 mmol) and purged with N2, then PdC12(dppf)-CH2C12adduct (30.3 mg, 0.037 mmol) added . The reaction mixture was heated at 90 °C for 4 h. After completion of reaction (monitored by UPLC), the reaction mixture was concentrated by using rota evaporator and extracted with ethylacetate (2X 50 mL). The organic layer was separated and dried over Na2SO4 and concentated by using rota evaporator. The crude was purified by column chromatography using 60-120 silica, eluted with 0-50% ethylacetate in petether. Yield 2-(4-cyclopropyl-6- methoxypyrimidin-5-yl)-4-(4-(l-methyl-4-(tri fluoromethyl)- IH-imidazol -2- yl)benzyl)pyrido[3,2-d]pyrimidine (42 mg, 0.079 mmol, 21.32 % yield) as yellow solid. LC-MS (ESI), m/z: 518.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 9.24 (dd, J= 1.5, 4.0 Hz, 1H), 8.73 (s, 1H), 8.52 (dd, J= 1.5, 8.5 Hz, 1H), 8.11 - 8.06 (m, 1H), 7.90 (d, J= 1.5 Hz, 1H), 7.64 (d, J= 8.0 Hz, 2H), 7.57 - 7.53 (m, 2H), 4.93 (s, 2H), 3.86 (s, 3H), 3.75 (s, 3H), 1.76 - 1.63 (m, 1H), 1.12 - 1.01 (m, 2H), 0.92 - 0.81 (m, 2H).
Examples in Table 10 were prepared according to the procedure described above.
Intermediate 38: 1 -(4-(hy droxymethyl)phenyl)-3 -(trifluoromethyl)pyridin-2( 1 H)-one
Step A: 4-(2-oxo-3-(trifluoromethyl)pyridin-l(2H)-yl)benzaldehyde
A mixture of 4-Fluorobenzaldehyde (560 mg, 4.51 mmol), cesium carbonate (1470 mg, 4.51 mmol) and 3-(Trifluoromethyl)pyridin-2-ol (736 mg, 4.51 mmol) on DMA (10 mL) was stirred at 140 °C for 18 hours. The mixture was diluted with EtOAc (25 mL) and was washed with a solution of aqueous 10% Lithium chloride solution (3 x 15 mL). The ethyl acetate layer was dried over sodium sulfate and concentrated. The crude product was subjected to ISCO flash chromatography (silica gel/hexane-EtOAc 100:0 to 0: 100 gradient). Yield 4-(2-oxo-3-(trifluoromethyl)pyridin-l(2H)-yl)benzaldehyde (800 mg, 2.99 mmol, 66.4 % yield). LC-MS (ESI) m/z: 267.9 [M+H]+.
To a solution of 4-(2-oxo-3-(trifluoromethyl)pyridin-l(2H)-yl)benzaldehyde (409 mg, 1.531 mmol) in THF (5.0 mL) and MeOH (5.0 mL) was added sodium borohydride (57.9 mg, 1.531 mmol), the mixture was stirred at RT for 1 hour. The mixture was concentrated. The crude product was subjected to ISCO flash chromatography (silica gel/hexane-EtOAc 100:0 to 0:100 gradient). Yield l-(4-(hydroxymethyl)phenyl)-3- (trifluoromethyl)pyridin-2(lH)-one (294 mg, 1.092 mmol, 71.3 % yield) as off-white solid. LC-MS (ESI) m/z: 269.9 [M+H]+.
Intermediate in Table 11 were prepared according to the procedure described above.
Scheme 7: Synthesis of intermediate 40
Intermediate 40: l-(4-(((2-chloropyrido[3,2-d]pyrimidin-4-yl)oxy)methyl)phenyl)-3- (trifluoromethyl)pyridin-2(lH)-one
To a solution of l-(4-(hydroxymethyl)phenyl)-3-(trifluoromethyl)pyridin-2(lH)-one (60 mg, 0.223 mmol) and 2,4-Dichloropyrido[3,2-d]pyrimidine (44.6 mg, 0.223 mmol) in anhydrous THF (2.0 mL) under nitrogen was added a solution of 1.0 M Sodium bis(trimethylsilyl)amide solution in THF (223 pl, 0.223 mmol), the mixture was stirred at RT for 18 hours. The mixture was diluted with EtOAc (5 mL) and was washed with a solution of aqueous 10% Lithium chloride solution (3 x 15 mL). The ethyl acetate layer was dried over sodium sulfate and concentrated. The crude product was subjected to ISCO flash chromatography (silica gel/hexane-EtOAc 100:0 to 0: 100 gradient). Yield 1- (4-(((2-chloropyrido[3,2-d]pyrimidin-4-yl)oxy)methyl)phenyl)-3- (trifluoromethyl)pyridin-2(lH)-one (41 mg, 0.095 mmol, 42.5 % yield) as white solid. LC-MS (ESI) m/z: 432.8 [M+H]+.
Intermediate in Table 12 were prepared according to the procedure described above.
Example 45: l-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)pyrido[3,2- d]pyrimidin-4-yl)oxy)methyl)phenyl)-3-(trifluoromethyl)pyridin-2(lH)-one
A mixture of l-(4-(((2-chloropyrido[3,2-d]pyrimidin-4-yl)oxy)methyl)phenyl)-3- (trifluoromethyl)pyridin-2(lH)-one (41 mg, 0.095 mmol), (4-cyclopropyl-6- methoxypyrimidin-5-yl)boronic acid (18.38 mg, 0.095 mmol), 2.0 M potassium phosphate tribasic (118 pl, 0.237 mmol) and Diehl oro[l,l'-bis(di -t- butylphosphino)ferrocene]palladium(II) (3.09 mg, 4.74 pmol) in dioxane (3 mL) under nitrogen was stirred at 60 °C for 18 hours. The mixtures was diluted with EtOAc (2 mL) and the ethyl acetate layer was dried over sodium sulfate and concentrated. The crude product was subjected to Prep HPLC purification. Column: XBridge Cl 8, 19 mm x 200 mm, 5 pm particles; Flow Rate: 20 mL/min; Column Temperature: 25 °C. Mobile Phase A (ACN/H2O (5:95) with 10 mM AA); Mobile Phase B (ACN/H2O (95:5) with 10 mM AA); Gradient 0% B to 100% B in 20 min. Yield l-(4-(((2-(4-cyclopropyl-6- methoxypyrimidin-5-yl)pyrido[3,2-d]pyrimidin-4-yl)oxy)methyl)phenyl)-3- (trifhioromethyl)pyridin-2(lH)-one (10.30 mg, 0.018 mmol, 19.24 % yield). LC-MS (ESI) m/z: 547.2 [M+H]+. 'H NMR (500 MHz, DMSO-t/e) 5 ppm 0.91 (br dd, J=7.86, 3.05 Hz, 2 H) 1.04 - 1.10 (m, 2 H) 1.83 (br dd, J=8.09, 3.59 Hz, 1 H) 3.87 (s, 3 H) 5.73 (s, 2 H) 6.49 (t, .7=7.00 Hz, 1 H) 7.51 (br d, J=7.39 Hz, 2 H) 7.72 (br d, J=8.39 Hz, 2 H) 7.97 - 8.08 (m, 3 H) 8.41 (d, J=8.36 Hz, 1 H) 8.71 (s, 1 H) 9.06 (dd, J=4.04, 1.53 Hz, 1 H).
Examples in Table 13 were prepared according to the procedure described above.
Table 13
Scheme 9: Synthesis of intermediate 44
A mixture of methyl 2-bromo-l-methyl-lH-imidazole-4-carboxylate (200 mg, 0.913 mmol), (4-(hydroxymethyl)phenyl)boronic acid (166 mg, 1.096 mmol), 1, l'-Bis(di-tert- butylphosphino) ferrocene palladium chloride (29.8 mg, 0.046 mmol) and 2.0 M potassium phosphate tribasic (1141 pl, 2.283 mmol) in dioxane (6.0 mL) under nitrogen was stirred at 60 °C for 18 hours. The mixture was diluted with EtOAc (5 mL) and was washed with a solution of aqueous 10% Lithium chloride solution (3 x 15 mL). The ethyl acetate layer was dried over sodium sulfate and concentrated. The crude product was subjected to ISCO flash chromatography (silica gel/hexane-EtOAc 100:0 to 0: 100 gradient). Yield methyl 2-(4-(hydroxymethyl)phenyl)-l -methyl- lH-imidazole-4-
carboxylate (210 mg, 0.853 mmol, 93 % yield) as brown gum. ). LC-MS (ESI) m/z:
246.9 [M+H]+.
To a solution of methyl 2-(4-(hydroxymethyl)phenyl)-l -methyl- lH-imidazole-4- carboxylate (210 mg, 0.853 mmol) in anhydrous THF (10 mL) under nitrogen was added a solution of 3.0 M Methylmagnesium bromide solution in diethyl ether (853 pl, 2.56 mmol), the mixture was stirred at RT for 18 hours. The reaction mixture was quenched with a solution of saturated NEUCl (10 mL) and the mixture was extracted with EtOAc (2 x 25 mL). The ethyl acetate layer was dried over sodium sulfate and concentrated. The crude product was subjected to ISCO flash chromatography (silica gel/hexane-EtOAc 100:0 to 0: 100 gradient). Yield 2-(2-(4-(hydroxymethyl)phenyl)-l-methyl-lH-imidazol- 4-yl)propan-2-ol (142 mg, 0.548 mmol, 64.2 % yield) as white gum. LC-MS (ESI) m/z: 247.0 [M+H]+. 'H NMR (500 MHz, CHLOROFORM-tZ) 5 ppm 1.58 - 1.63 (m, 6 H) 3.69 (s, 3 H) 4.75 (s, 2 H) 6.83 (s, 1 H) 7.38 (d, J=8.46 Hz, 2 H) 7.53 (d, J=7.53 Hz, 2 H).
Example 50: 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)pyrido[3,2- d]pyrimidin-4-yl)oxy)methyl)phenyl)-l-methyl-lH-imidazol-4-yl)propan-2-ol:
- I l l -
Step A: 2-(2-(4-(((2-chloropyrido[3,2-d]pyrimidin-4-yl)oxy)methyl)phenyl)-l -methyl- lH-imidazol-4-yl)propan-2-ol:
A mixture of 2-(2-(4-(hydroxymethyl)phenyl)-l -methyl- lH-imidazol-4-yl)propan-2-ol (42 mg, 0.171 mmol), cesium carbonate (55.6 mg, 0.171 mmol) and 2,4- Dichloropyrido[3,2-d]pyrimidine (34.1 mg, 0.171 mmol) in ACN (2.0 mL) was stirred at 80 °C for 18 hour. Ppt was filtered off and the filtrate was concentrated. The crude product was subjected to ISCO flash chromatography (silica gel/hexane-EtOAc 100:0 to 0: 100 gradient). Yield 2-(2-(4-(((2-chloropyrido[3,2-d]pyrimidin-4- yl)oxy)methyl)phenyl)-l-methyl-lH-imidazol-4-yl)propan-2-ol (30 mg, 0.073 mmol, 42.9 % yield) as white foam. LC-MS (ESI) m/z: 409.8 [M+H]+.
Step B: 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)pyrido[3,2-d]pyrimidin-4- yl)oxy)methyl)phenyl)- 1 -methyl- lH-imidazol-4-yl)propan-2-ol :
A mixture of 2-(2-(4-(((2-chloropyrido[3,2-d]pyrimidin-4-yl)oxy)methyl)phenyl)-l- methyl-lH-imidazol-4-yl)propan-2-ol (30 mg, 0.073 mmol), (4-Cyclopropyl-6- methoxypyrimidin-5-yl)boronic acid (14.20 mg, 0.073 mmol), 1, l'-Bis(di-tert- butylphosphino) ferrocene palladium chloride (47.7 mg, 0.073 mmol) and 2.0 M potassium phosphate tribasic (36.6 pl, 0.073 mmol) in dioxane (3.0 mL) under nitrogen
was stirred at 80 °C for 18 hours. The mixture was diluted with EtOAc (5 mL) and the ethyl acetate layer was dried over sodium sulfate and concentrated. The crude product was subjected to Prep HPLC purification. Column: XB ridge Cl 8, 19 mm x 200 mm, 5 pm particles; Flow Rate: 20 mL/min; Column Temperature: 25 °C. Mobile Phase A (ACN/H2O (5:95) with 10 mM AA); Mobile Phase B (ACN/H2O (95:5) with 10 mM AA); Gradient 0% B to 100% B in 20 min. Yield 2-(2-(4-(((2-(4-cyclopropyl-6- methoxypyrimidin-5-yl)pyrido[3,2-d]pyrimidin-4-yl)oxy)methyl)phenyl)-l -methyl- 1H- imidazol-4-yl)propan-2-ol (4.90 mg, 9.36 pmol, 12.79 % yield). LC-MS (ESI) m/z: 523.9 [M+H]+. 'H NMR (500 MHz, DMSO-t/e) 5 ppm 0.84 - 0.95 (m, 2 H) 1.03 - 1.13 (m, 2 H) 1.42 (s, 6 H) 1.77 - 1.89 (m, 1 H) 3.71 (s, 3 H) 3.88 (s, 3 H) 5.71 (s, 2 H) 7.03 (s,
1 H) 7.64 (m, J=8.24 Hz, 2 H) 7.71 (m, J=8.09 Hz, 2 H) 8.02 (dd, J=8.54, 4.20 Hz, 1 H) 8.41 (d, .7=8.33 Hz, 1 H) 8.72 (s, 1 H) 9.06 (dd, J=4.12, 1.53 Hz, 1 H).
Scheme 22: Syntheses of intermediates 91 to intermediate 109
Intermediate 91 : Synthesis of Methyl 4-(4-(trifluoromethyl)-lJ/-imidazol-2-yl)benzoate:
A mixture of 3,3-dibromo-l,l,l-trifluoropropan-2-one (49.3 g, 183 mmol) and sodium acetate (19.99 g, 244 mmol) in water (100 mL) was stirred at 95 °C for 30 min. After that, the mixture was cooled to 0 °C and a cold solution of methyl 4-formylbenzoate (20.00 g, 122.00 mmol) in a mixture of NH4OH (28% aq, 100 mL) and MeOH (300 mL) was added to that. The resulting mixture was stirred at 25 °C for 16 h. After completion of reaction (monitored by TLC), volatiles were evaporated, the residue obtained was diluted with ethyl acetate and washed with water. The organic extract was dried over anhydrous Na2SO4, filtered and solvents evaporated from the filtrate. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain methyl 4-(4-(trifluorom ethyl)- 1H- imidazol-2-yl)benzoate (19.00 g, 60.5 mmol, 49.7%) as yellow solid. LCMS (ESI) m/z: 271.0 [M+H]+.
To a stirred solution of methyl 4-(4-(trifluoromethyl)-lH-imidazol-2-yl)benzoate (2.50 g, 9.25 mmol) in DMF (20 mL), sodium hydride (60% dispersion in mineral oil, 0.740 g, 18.50 mmol) was added followed by addition of iodomethane (0.868 mL, 13.88 mmol) at 0 °C and the mixture was stirred at 25 °C for 3 h. After completion of reaction (monitored by TLC), chilled water was added and extracted with ethyl acetate. The organic extract was dried over anhydrous Na2SO4, filtered and solvents evaporated from the filtrate under
reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh to obtain methyl 4-(l-methyl-4-(trifluorom ethyl)- lJT-imidazol-2- yl)benzoate (1.40 g , 4.87 mmol, 52.7 %) as white solid. LCMS (ESI) m/z: 285.0 [M+H]+.
Intermediate in Table 14 was prepared according to procedure described above.
Intermediate 94: Synthesis of (4-(l-Methyl-4-(trifluorom ethyl)- IT/-imidazol-2- yl)phenyl)m ethanol :
To a stirred solution of methyl 4-(l-methyl-4-(trifluoromethyl)-17/-imidazol-2-yl)benzoate (1.20 g, 4.22 mmol) in THF (25 mL), lithium aluminium hydride (2 M in THF, 4.22 mL, 8.44 mmol) was added at 0 °C and the mixture was stirred at 25 °C for 3 h. After completion of the reaction (monitored by TLC), saturated aqueous NFUCl was added and extracted with ethyl acetate. The organic extract was dried over anhydrous Na2SO4, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain (4-(l-methyl-4-
(trifluoromethyl)- 17/-imidazol-2-yl)phenyl)methanol (0.900 g, 3.42 mmol, 81%) as white solid. LCMS (ESI) m/z: 257.0 [M+H]+.
Intermediate in Table 15 was prepared according to procedure described above.
Intermediate 96: Synthesis of Methyl 4-(l-isopropyl-4-(trifluoromethyl)-17/-imidazol-2- yl)benzoate:
Procedure B: To a stirred solution of methyl 4-(4-(trifluoromethyl)-17/-imidazol-2- yl)benzoate (2.0 g, 7.40 mmol) in acetonitrile (30 mL), CS2CO3 (4.82 g, 14.80 mmol) was added followed by addition of 2-iodopropane (1.110 mL, 11.10 mmol) at 0 °C and the mixture was stirred at 25 °C for 16 h. After that, reaction mixture was heated at 50 °C for additional 16 h. After completion of the reaction (monitored by TLC), volatiles were removed under reduced pressure, diluted with ethyl acetate and washed with water. The organic extract was dried over anhydrous Na2SO4, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash
chromatography on silica gel, 230-400 mesh, to obtain methyl 4-(l-isopropyl-4- (trifluoromethyl)-lJ/-imidazol-2-yl)benzoate (0.800 g, 2.229 mmol, 30.1%) as white solid. LCMS (ESI) m/z: 313.2 [M+H]+. Intermediate in Table 16 were prepared according to procedure described above.
Table 16
Intermediate 99: Synthesis of (4-( l -Isopropyl-4-(trifluoromethyl)- IT/-imidazol-2- yl)phenyl)m ethanol :
To a stirred solution of methyl 4-(l-isopropyl-4-(trifluoromethyl)-17/-imidazol-2- yl)benzoate (0.70 g, 2.241 mmol) in THF (10 mL), lithium aluminium hydride (2 M in THF, 2.241 mL, 4.48 mmol) at 0 °C and the mixture was stirred at 25 °C for 3 h. After completion of the reaction (monitored by TLC), saturated aqueous NH4CI was added and extracted with ethyl acetate. The organic extract was dried over anhydrous Na2SO4, filtered
and solvents evaporated from the filtrate. The crude was purified by flash chromatography on silica gel, 230-400 mesh, to obtain (4-(l-isopropyl-4-(trifluorornethyl)-lJ/-imidazol-2- yl)phenyl)methanol (0.650 g, 2.242 mmol, 100%) as white solid. LCMS (ESI) m/z: 285.0 [M+H]+.
Intermediate in Table 17 was prepared according to procedure described above.
Intermediate 102: Synthesis of 2-(4-(bromomethyl)phenyl)-l-methyl-4-(trifluoromethyl)- IH-imidazole:
To a stirred solution of (4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)methanol (2 g, 7.81 mmol) in dichloromethane (40 mL), triphenylphosphine (3.07 g, 11.71 mmol) and carbon tetrabromide (5.18 g, 15.61 mmol) were added at 25 °C and the mixture was stirred at 25 °C for 16 h. After completion of the reaction (monitored
by TLC), water was added and extracted with dichloromethane. The organic extract was dried over anhydrous Na2SO4, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain 2-(4-(bromomethyl)phenyl)-l-methyl-4-(trifluoromethyl)-lH- imidazole (2 g, 6.27 mmol, 80 % yield) as off-white solid. LCMS (ESI) m/z: 318.9 [M+H]+.
Intermediate in Table 18 was prepared according to procedure described above.
Intermediate 104: 4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzaldehyde:
In a 250 mL round bottom flask, (4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)methanol (500.0 mg, 1.717 mmol) was dissolved in DCM (10 mL) at 25 °C, then cooled to 0 °C, Dess-Martin periodinane (1.457 g, 3.43 mmol) was added portion wise at 0 °C. The reaction mixture was stirred at 25 °C for 3 h. After completion of the reaction (monitored by TLC), saturated aqueous NELCl was added and extracted with ethyl acetate. The organic extract was dried over anhydrous Na2SO4, filtered and solvents evaporated from the filtrate. The crude was purified by flash chromatography on silica gel, 230-400
mesh, to obtain (4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzaldehyde (430.0 mg, 1.600 mmol, 93 % yield)) as semi solid. LCMS (ESI) m/z: 255.1 [M-H]+.
Intermediate in Table 19 was prepared according to procedure described above.
In a 10 mL round bottom flask, 4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzaldehyde (330.0 mg, 1.228 mmol) was dissolved in diethyl ether (5.0 mL) at 25 °C and cooled to 0 °C. methyl-magnesium bromide in diethyl ether (1.0 M, 0.818 mL, 2.455 mmol) was added dropwise at 0 °C. The reaction mixture was stirred at 25 °C for 3 h. After completion of the reaction (monitored by TLC), saturated ammonium chloride solution was added, diluted with ethyl acetate and washed with water. The organic extract was dried over anhydrous Na2SO4, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain (l-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)phenyl)ethan-l-ol (330.0 mg, 1.220 mmol, 94 % yield)) as semi solid. LCMS (ESI) m/z: 271.0 [M-H]+.
Intermediate in Table 20 was prepared according to procedure described above.
Intermediate 108: Synthesis of l-methyl-4-(trifluoromethyl)-2-(4- ((trimethylstannyl)methyl)phenyl)-lH-imidazole:
In a stirred solution of 2-(4-(bromomethyl)phenyl)-l-methyl-4-(trifluoromethyl)-lH- imidazole (2.0 g, 6.27 mmol) in toluene (15 mL), hexamethylditin (2.60 mL, 12.53 mmol) and bis(triphenylphosphine)palladium(II) dichloride (0.880 g, 1.253 mmol) were added at 25 °C under the nitrogen atmosphere. The reaction mixture was heated to 90 °C for 2 h. After completion of reaction (monitored by UPLC-MS and TLC), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by column chromatography on silica gel, 60-120 mesh, to obtain l-methyl-4-(trifluoromethyl)-2-(4-((trimethylstannyl)methyl)phenyl)-lH-imidazole (1.7 g, 3.92 mmol, 62.6 % yield) as off-white solid. LCMS (ESI) m/z: 405.0 [M-H]+.
Intermediate 109: Synthesis of l-isopropyl-4-(trifluoromethyl)-2-(4- ((trimethylstannyl)methyl)phenyl)-lH-imidazole:
In a stirred solution of 2-(4-(bromomethyl)phenyl)-l-isopropyl-4-(trifluoromethyl)-lH- imidazole (200 mg, 0.576 mmol) in toluene (5 mL), hexamethyl ditin (0.179 mL, 0.864 mmol) and bis(triphenylphosphine)palladium(II) di chloride (81 mg, 0.115 mmol) were added at 25 °C under the nitrogen atmosphere. The reaction mixture was heated to 90 °C for 16 h. After completion of reaction (monitored by UPLC-MS and TLC), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by column chromatography on silica gel, 60-120 mesh, to obtain l-isopropyl-4-(trifluoromethyl)-2-(4-((trimethylstannyl)methyl)phenyl)-lH-imidazole (150 mg, 0.344 mmol, 59.8 % yield) as off-white solid. LCMS (ESI) m/z: 433.0 [M-H]+.
Intermediate 110: Synthesis of 2-methoxy-4-(4-(trifluoromethyl)-lH-imidazol-2- yl)benzonitrile:
A mixture of 3, 3 -dibromo- 1,1,1 -trifluoropropan-2-one (9.21 g, 34.100 mmol) and sodium acetate (5.09 g, 62.100 mmol) in water (20 mL) was stirred at 95 °C for 30 min. After that, the mixture was cooled to 0 °C and a cold solution of methyl 4-formyl-2- methoxybenzonitrile (5.0 g, 31.000 mmol) in a mixture of NH4OH (28% aq, 24 mL) and MeOH (100 mL) was added to that. The resulting mixture was stirred at 25 °C for 16 h. After completion of reaction (monitored by TLC), volatiles were evaporated, the residue obtained was diluted with ethyl acetate and washed with water. The organic extract was dried over anhydrous Na2SO4, filtered and solvents evaporated from the filtrate to obtain 2-methoxy-4-(4-(trifluoromethyl)-lH-imidazol-2-yl)benzonitrile (4.5 g, 12.29 mmol, 39.6% yield) as pale brown solid. Desired product was not ionized in LCMS, it was confirmed in next step.
Intermediate 111 : synthesis of 2-methoxy-4-(l-methyl-4-(trifluoromethyl)-lH-imidazol- 2-yl)benzonitrile:
To a stirred solution of 2-methoxy-4-(4-(trifluoromethyl)-lH-imidazol-2-yl)benzonitrile (1.5 g, 5.61 mmol) in DMF (15 mL) was added sodium hydride (60% in mineral oil, 0.674 g, 11.230 mmol) was added portion wise at 0 °C and reaction mixture was stirred at room temperature for 30 minutes. After 30 minutes, iodomethane (0.702 mL, 11.230 mmol) was added dropwise at 0 °C and reaction mixture was stirred at room temperature for 16 h. After completion of reaction (monitored by UPLC/TLC), reaction mixture was quenched with saturated NH4Q solution and extracted with ethyl acetate (2 x 50 mL). Combined organic extract was passed through Na2SO4, concentrated under reduced pressure to get crude product which is further purified by flash column chromatography on silica 230-400 mesh, using 15% ethyl acetate in pet ether gradient to obtain 2-methoxy-4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)benzonitrile (1.4 g, 4.430 mmol, 79% yield) as pale brown solid. LCMS (ESI) m/z: 282.0 [M+H] +.
Intermediate 112: Synthesis of 4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)-2- methoxybenzonitrile:
To a stirred solution of 2-methoxy-4-(4-(trifluoromethyl)-lH-imidazol-2-yl)benzonitrile (1.3 g, 4.870 mmol) in acetonitrile (15 mL) were added cesium carbonate (3.17 g, 9.730 mmol) and 2-iodopropane (0.730 mL, 7.300 mmol) and reaction mixture was heated at 80 °C for 16 h. After completion of reaction (monitored by UPLC/TLC), the reaction mixture was filtered through celite bed and filtrate was concentrated under reduced pressure to get crude product which is further purified by flash column chromatography on silica 230-400 mesh, using 35% ethyl acetate in pet ether gradient to obtain 4-(l-isopropyl-4- (trifluoromethyl)-lH-imidazol-2-yl)-2-methoxybenzonitrile (1.2 g, 3.260 mmol, 67.0% yield) as pale brown solid. LCMS (ESI) m/z: 310.2 [M+H] +.
Intermediate 113: synthesis of 2-methoxy-4-(l-methyl-4-(trifluoromethyl)-lH-imidazol- 2-yl)benzoic acid:
To a stirred solution of 2-methoxy-4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzonitrile (750 mg, 2.670 mmol) in ethanol (5 mL) was added aqueous solution NaOH (213 mg, 5.330 mmol) in H2O (5 mL) at room temperature and reaction mixture was heated at 80 °C for 16 h. After completion of reaction (monitored by UPLC/TLC), reaction mixture was concentrated and acidified with citric acid (to pH = 4) and extracted with ethyl acetate (2 x 25 mL). Combined organic extract was passed through Na2SO4, concentrated under
reduced pressure to get crude product which is further purified by flash column chromatography on silica 230-400 mesh, using 50% ethyl acetate in pet ether gradient to obtain 2-methoxy-4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzoic acid (700 mg, 2.173 mmol, 81% yield) as orange solid. LCMS (ESI) m/z: 301.0 [M+H] +.
Intermediate in Table 21 was prepared according to procedure described above.
Intermediate 115: Synthesis of (2-methoxy-4-(l-methyl-4-(trifluoromethyl)-lH-imidazol- 2-yl)phenyl)methanol :
To a stirred solution of 2-methoxy-4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzoic acid (800 mg, 2.660 mmol) in THF (15 mL) was added lithium aluminiumhydride (1 M in THF, 13.32 mL, 13.320 mmol) at 0 °C and reaction mixture was stirred at 0 °C for 4 h. After completion of reaction (monitored by UPLC/TLC), reaction mixture was quenched with saturated NH4Q solution and extracted with ethyl acetate (2 x 50 mL). Combined organic extract was dried over Na2SO4, concentrated under reduced pressure to get crude product which is further purified by flash column chromatography on silica 230- 400 mesh, using 40-50% ethyl acetate in pet ether gradient to obtain (2-methoxy-4-(l- methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)phenyl)methanol (600 mg, 79.0% yield) as colorless liquid. LCMS (ESI) m/z: 287.0 [M+H] +.
Intermediate in Table 22 was prepared according to procedure described above.
Scheme 20: Syntheses of example 60 to example73
Intermediate 60: Synthesis of 2-chloro-4-((2-methoxy-4-(l-methyl-4-(trifluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine:
To a stirred solution of (2-methoxy-4-(l-methyl-4-(trifluorornethyl)-lH-imidazol-2- yl)phenyl)methanol (300 mg, 1.048 mmol) in acetonitrile (10 mL), cesium carbonate (683 mg, 2.096 mmol) and 2,4-dichloropyrido[2,3-d]pyrimidine (210 mg, 1.048 mmol) were added and reaction mixture was heated at 80 °C for 16 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain 2- chloro-4-((2-methoxy-4-(l-methyl-4-(tri fluoromethyl)- IH-imidazol -2- yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (130 mg, 0.254 mmol, 24.27% yield) as brown solid. LCMS (ESI) m/z: 450.0 [M+H]+.
Intermediate 61 : Synthesis of 2-chloro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH- imidazol-2-yl)-2-methoxybenzyl)oxy)pyrido[2,3-d]pyrimidine:
To a stirred solution of 4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)-2- methoxyphenyl)methanol (400 mg, 1.273 mmol) and 2,4-dichloropyrido[2,3-d]pyrimidine (305 mg, 1.527 mmol) in THF (10 mL), lithium bis(trimethylsilyl)amide (LiHMDS) (1 M solution in THF, 2.55 mL, 2.55 mmol) was added at 0 °C and the mixture was stirred at 25 °C for 1 h. After completion of reaction (monitored by UPLC-MS and TLC), the reaction mixture was again cooled to 0 °C, chilled saturated ammonium chloride solution was added and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by column chromatography on silica gel, 60-120 mesh, to obtain 2- chloro-4-((4-(l-isopropyl-4-(tri fluoromethyl)- IH-imidazol -2 -yl)-2- methoxybenzyl)oxy)pyrido[2,3-d]pyrimidine (100 mg, 0.209 mmol, 16.44 % yield) as pale yellow gum. LCMS (ESI) m/z: 478.2 [M+H]+.
Intermediate 62: Synthesis of 2-chloro-4-((4-(l-ethyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)pyrido[2,3-d]pyrimidine:
To a stirred solution of (4-(l-ethyl-4-(trifluoromethyl)-lH-imidazol-2-yl)phenyl)methanol (300 mg, 1.068 mmol) in acetonitrile (5 mL), cesium carbonate (696 mg, 2.136 mmol) and 2,4-dichloropyrido[2,3-d]pyrimidine (256 mg, 1.281 mmol) were added and reaction mixture was heated at 60 °C for 4 h. After completion of the reaction (monitored by UPLC- MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain 2-chloro-4-((4-(l-ethyl-4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (200 mg, 0.403 mmol, 37.8 % yield) as off-white solid. LCMS (ESI) m/z: 434.2 [M+H]+,LC retention time: 2.59 min (LCMS Method 9).
Intermediate 63: Synthesis of 2-chloro-4-((4-(l-isobutyl-4-(trifluoromethyl)-lH-imidazol- 2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine:
To a stirred solution of (4-(l-isobutyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)methanol (200 mg, 0.610 mmol) in acetonitrile (5 mL), cesium carbonate (397 mg, 1.220 mmol) and 2,4-dichloropyrido[2,3-d]pyrimidine (146 mg, 0.732 mmol) were added and reaction mixture was heated at 60 °C for 6 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain 2- chloro-4-((4-(l-isobutyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3- d]pyrimidine (150 mg, 0.288 mmol, 47.2 % yield) as off-white solid. LCMS (ESI) m/z: 462.2 [M+H]+.
Intermediate 64: Synthesis of 2-chloro-4-((4-(4-(trifluoromethyl)-l-((2- (trimethylsilyl)ethoxy)methyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine:
To a stirred solution of (4-(4-(trifluorom ethyl)- l-((2-(trimethyl silyl)ethoxy)methyl)- 1H- imidazol-2-yl)phenyl)methanol (1.5 g, 4.03 mmol) in acetonitrile (25 mL), cesium carbonate (2.62 g, 8.05 mmol) and 2,4-dichloropyrido[2,3-d]pyrimidine (0.806 g, 4.03 mmol) were added and reaction mixture was heated at RT for 16 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain 2-chloro-4-((4-(4-(trifluorom ethyl)- l-((2-(trimethyl silyl)ethoxy)m ethyl)- 1H- imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (850 mg, 1.507 mmol, 37.4 % yield) as off-white solid. LCMS (ESI) m/z: 536.3 [M+H]+.
Intermediate 65: Synthesis of 2-chloro-4-(l-(4-(l-methyl-4-(trifluoromethyl)-lH- imidazol-2-yl)phenyl)ethoxy)pyrido[2,3-d]pyrimidine:
To a stirred solution of 2,4-dichloropyrido[2,3-d]pyrimidine (100 mg, 0.500 mmol) and 1- (4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)phenyl)ethan-l-ol (135 mg, 0.500 mmol) in anhydrous tetrahydrofuran (14 mL), NaHMDS (1 M solution in THF, 0.550 mL, 0.550 mmol) was added at -35 °C and the mixture was stirred at -35 °C for 20 min. After completion of reaction (monitored by UPLC-MS and TLC), chilled saturated ammonium chloride solution was added and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by column chromatography on silica gel, 60-120 mesh, to obtain 2-chloro-4-(l-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol- 2-yl)phenyl)ethoxy)pyrido[2,3-d]pyrimidine (90 mg, 0.191 mmol, 38.2 % yield) as off white solid. LCMS (ESI) m/z: 434.0 [M+H]+.
Intermediate 66: Synthesis of 2-chloro-4-(l-(4-(l-isopropyl-4-(trifluoromethyl)-lH- imidazol-2-yl)phenyl)ethoxy)pyrido[2,3-d]pyrimidine:
To a stirred solution of 2,4-dichloropyrido[2,3-d]pyrimidine (100 mg, 0.500 mmol) and 1- (4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)phenyl)ethan-l-ol (149 mg, 0.500 mmol) in anhydrous tetrahydrofuran (14 mL), NaHMDS (1 M solution in THF, 0.500 mL, 0.500 mmol) was added at -35 °C and the mixture was stirred at -35 °C for 20 min. After completion of reaction (monitored by UPLC-MS and TLC), chilled saturated ammonium chloride solution was added and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by column chromatography on silica gel, 60-120 mesh, to obtain 2-chloro-4-(l-(4-(l-isopropyl-4-(trifluoromethyl)-lH- imidazol-2-yl)phenyl)ethoxy)pyrido[2,3-d]pyrimidine (75 mg, 0.155 mmol, 31.1 % yield) as off-white solid. LCMS (ESI) m/z: 462.0 [M+H]+.
Intermediate 67: Synthesis of 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol- 2-yl)benzyl)pyrido[2,3-d]pyrimidine:
To a stirred solution of 2,4-dichloropyrido[2,3-d]pyrimidine (600 mg, 3.00 mmol) in 1,4- dioxane (8 mL), lithium chloride (191 mg, 4.50 mmol) and l-methyl-4-(trifluoromethyl)- 2-(4-((trimethylstannyl)methyl)phenyl)-lH-imidazole (1088 mg, 2.70 mmol) were added
and purged with N2 gas for 10 min. Then bis(tri-tert-butylphosphine)palladium(0) (153 mg, 0.300 mmol) was added at 25 °C and reaction mixture was heated at 100 °C for 2 h. After completion of reaction (monitored by UPLC-MS and TLC), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by column chromatography on silica gel, 60-120 mesh, to obtain 2-chloro-4-(4-(l -methyl-4-(tri fluoromethyl)- IH-imidazol -2 -yl)benzyl)pyrido[2, 3- d]pyrimidine (80 mg, 0.143 mmol, 4.76 % yield) as pale yellow gum. LCMS (ESI) m/z: 404.0 [M+H]+.
Example 60: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((2-methoxy- 4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3- djpyrimidine:
To a stirred solution of 2-chloro-4-((2-methoxy-4-(l-methyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (120 mg, 0.267 mmol) in 1,4-dioxane (10 mL), potassium phosphate tribasic (113 mg, 0.534 mmol), (4-cyclopropyl-6- methoxypyrimidin-5-yl)boronic acid (104 mg, 0.534 mmol) were added and purged with N2 gas for 5 min. then tetrakis(triphenylphosphine)palladium(0) (30.8 mg, 0.028 mmol) was added and reaction mixture was heated at 120 °C for 2 h under MW irradiation. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC (HPLC method: Diluent:
THF:WATER:ACN(30:30:40); Column: XBridge Prep C8 OBD (19x150mm) 5p; Temperature: Ambient; Mobile phase A: 5mM Ammonium Formate; Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/30, 15/70, 17/70) to obtain 2-(4- cyclopropyl-6-methoxypyrimidin-5-yl)-4-((2 -methoxy -4-(l -methyl-4-(trifluorom ethyl)- lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (9 mg, 0.016 mmol, 5.96% yield) as pale yellow solid. LCMS (ESI) m/z: 564.2 [M+H]+. 1 H NMR (400 MHz, DMSO-de) 5 = 9.24 (dd, J = 4.4, 2.0 Hz, 1H), 8.72-8.69 (m, 2H), 7.96 (d, J = 1.2 Hz, 1H), 7.77 (dd, J = 8.0, 4.4 Hz, 1H), 7.66 (d, J = 7.6 Hz, 1H), 7.35-7.31 (m, 2H), 5.70 (s, 2H), 3.88 (s, 3H), 3.87 (s, 3H), 3.81 (s, 3H), 1.89-1.82 (m, 1H), 1.11-1.07 (m, 2H), 0.91-0.89 (m, 2H).
Example 61: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)-2-methoxybenzyl)oxy)pyrido[2,3- djpyrimidine:
To a stirred solution of 2-chloro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)- 2-methoxybenzyl)oxy)pyrido[2,3-d]pyrimidine (90 mg, 0.188 mmol) in 1,4-dioxane (9 mL), potassium phosphate tribasic (80 mg, 0.377 mmol), (4-cyclopropyl-6- methoxypyrimidin-5-yl)boronic acid (73.1 mg, 0.377 mmol) were added and purged with N2 gas for 5 min. then tetrakis(triphenylphosphine)palladium(0) (21.76 mg, 0.019 mmol) was added and reaction mixture was heated at 120 °C for 2 h under MW irradiation. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC (HPLC method: Diluent: THF:WATER:ACN(30:30:40); Column: XBridge Prep C8 OBD (19x150mm) 5p;
Temperature: Ambient; Mobile phase A: 5mM Ammonium Formate; Mobile phase B: Acetonitrile; Flow :15 mL/min; Time/Grad : 0/40, 8/80, 12/80) to obtain 2-(4-cyclopropyl- 6-methoxypyrimidin-5-yl)-4-((4-(l -isopropyl-4-(tri fluoromethyl)- lH-imidazol-2-yl)-2- methoxybenzyl)oxy)pyrido[2,3-d]pyrimidine (25 mg, 0.042 mmol, 22.17 % yield) as off- white solid. LCMS (ESI) m/z: 592.2 [M+H]+. (400 MHz, DMSO-dd) 3 = 9.25 (dd, J = 4.4, 2.0 Hz, 1H), 8.74-8.71 (m, 2H), 8.20 (d, J = 1.2 Hz, 1H), 7.78 (dd, J = 8.0, 4.4 Hz, 1H), 7.68 (d, J = 7.6 Hz, 1H), 7.22 (d, J = 1.6 Hz, 1H), 7.17 (dd, J = 8.0, 2.0 Hz, 1H), 5.71 (s, 2H), 4.56-4.50 (m, 1H), 3.889 (s, 3H), 3.885 (s, 3H), 1.89-1.82 (m, 1H), 1.43 (d, J = 6.8 Hz, 6H), 1.11-1.08 (m, 2H), 0.90-0.87 (m, 2H).
Example 62: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l-ethyl- 4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine:
To a stirred solution of 2-chloro-4-((4-(l-ethyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (100 mg, 0.202 mmol) in 1,4-dioxane (10 mL), potassium phosphate tribasic (86 mg, 0.403 mmol), (4-cyclopropyl-6-methoxypyrimidin- 5-yl)boronic acid (46.9 mg, 0.242 mmol) and tetrakis(triphenylphosphine)palladium(0) (23.29 mg, 0.020 mmol) were added under nitrogen atmosphere, and reaction mixture was heated at 60 °C for 1 h under MW irradiation. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (30:30:40); X-Bridge C8 (250 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/60, 7/90, 10/90] to obtain
2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l-ethyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (32 mg, 29 % yield) as off-white solid. LCMS (ESI) m/z: 548.2 [M+H]+. *HNMR (400 MHz, DMSO-de) 5 = 9.26 (dd, J= 2.0, 4.4 Hz, 1H), 8.74 (dd, J= 2.1, 8.2 Hz, 1H), 8.73 (s, 1H), 8.05 (d, J= 1.3 Hz, 1H), 7.79 (dd, J = 4.4, 8.3 Hz, 1H), 7.74 - 7.67 (m, 4H), 5.76 (s, 2H), 4.11 (q, J= 13 Hz, 2H), 3.88 (s, 3H), 1.91 - 1.82 (m, 1H), 1.34 (t, J= 7.3 Hz, 3H), 1.13 - 1.06 (m, 2H), 0.94 - 0.84 (m, 2H).
Example 63: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- isobutyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine:
To a stirred solution of 2-chloro-4-((4-(l-isobutyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (100 mg, 0.192 mmol) in 1,4-dioxane (5 mL), potassium phosphate tribasic (81 mg, 0.384 mmol), (4-cyclopropyl-6-methoxypyrimidin- 5-yl)boronic acid (44.7 mg, 0.230 mmol) and tetrakis(triphenylphosphine)palladium(0) (22.18 mg, 0.019 mmol) were added under nitrogen atmosphere, and reaction mixture was heated at 60 °C for 1 h under MW irradiation. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (20:40:40); X-Select C18 (150 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/60, 15/90] to obtain 2-(4- cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l-isobutyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (15 mg, 14% yield) as off-white solid. LCMS (ESI) m/z: 576.2 [M+H]+. *HNMR (400 MHz, DMSO-de) 5 = 9.25 (dd, J= 2.0, 4.4 Hz, 1H), 8.74 (dd, J= 2.1, 8.2 Hz, 1H), 8.72 (s, 1H), 8.00 (d, J= 1.3 Hz, 1H), 7.79 (dd, J
= 4.4, 8.1 Hz, 1H), 7.73 - 7.65 (m, 4H), 5.76 (s, 2H), 4.00 - 3.91 (m, 2H), 3.87 (s, 3H), 1.99
- 1.80 (m, 2H), 1.12 - 1.02 (m, 2H), 0.93 - 0.83 (m, 2H), 0.71 (d, J= 6.6 Hz, 6H).
Intermediate 68: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(4-
(tri fluoromethyl)- l-((2-(trimethylsilyl)ethoxy)methyl)-lH-imidazol-2- yl)benzyl)oxy)pyrido[2,3-d]pyrimidine:
To a stirred solution of 2-chloro-4-((4-(4-(trifluoromethyl)-l-((2- (trimethylsilyl)ethoxy)methyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (350 mg, 0.653 mmol) in 1,4-dioxane (15 mL), potassium phosphate tribasic (277 mg, 1.306 mmol), (4-cyclopropyl-6-methoxypyrimidin-5-yl)boronic acid (152 mg, 0.784 mmol) and Pd(dppf)C12 (96 mg, 0.131 mmol) were added under nitrogen atmosphere and reaction mixture was heated at 60 °C for 16 h. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by column chromatography on silica gel, 230-400 mesh, to obtain 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(4- (tri fluoromethyl)- l-((2-(trimethylsilyl)ethoxy)methyl)-lH-imidazol-2- yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (300 mg, 0.462 mmol, 70.7 % yield) as yellow gum. LCMS (ESI) m/z: 650.2 [M+H]+.
Example 64: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(l-(4-(l- methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)phenyl)ethoxy)pyrido[2,3- djpyrimidine:
To a stirred solution of 2-chloro-4-(l-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)ethoxy)pyrido[2,3-d]pyrimidine ( 80 mg, 0.170 mmol) in a mixture of 1,4- dioxane and water (4 mL, 9:1), potassium phosphate tribasic (72.0 mg, 0.339 mmol), 4- cyclopropyl-6-methoxy-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)pyrimidine (65.6 mg, 0.238 mmol) and tetrakis(triphenylphosphine)palladium(0) (19.61 mg, 0.017 mmol) were added under nitrogen atmosphere, and reaction mixture was heated at 120 °C for 2 h under MW irradiation. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (30:30:40); X-Bridge C8 (250 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/40, 15/80] to obtain 2-(4-cyclopropyl-6- methoxypyrimidin-5-yl)-4-(l-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)ethoxy)pyrido[2,3-d]pyrimidine (44 mg, 0.075 mmol, 44.2 % yield) as off-white solid. LCMS (ESI) m/z: 548.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 9.25 (dd, J = 2.0, 4.4 Hz, 1H), 8.80 (dd, J= 2.0, 8.3 Hz, 1H), 8.71 (s, 1H), 7.94 (d, J= 1.1 Hz, 1H), 7.81 (dd, J= 4.4, 8.1 Hz, 1H), 7.76 - 7.68 (m, 4H), 6.57 (q, J= 6.4 Hz, 1H), 3.82 (s, 3H), 3.78 (s, 3H), 1.77 (d, J= 6.5 Hz, 3H), 1.81 - 1.73 (m, 1H), 1.10 - 0.99 (m, 2H), 0.92 - 0.75 (m, 2H).
Example 65: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(l-(4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)phenyl)ethoxy)pyrido[2,3- djpyrimidine:
To a stirred solution of 2-chloro-4-(l-(4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)ethoxy)pyrido[2,3-d]pyrimidine (85 mg, 0.177 mmol) in a mixture of 1,4- dioxane and water (4 mL, 9: 1), potassium phosphate tribasic (75 mg, 0.353 mmol), 4- cyclopropyl-6-methoxy-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)pyrimidine (68.3 mg, 0.247 mmol) and tetrakis(triphenylphosphine)palladium(0) (20.42 mg, 0.018 mmol) were added under nitrogen atmosphere, and reaction mixture was heated at 120 °C for 2 h under MW irradiation. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase flash purification (method: Diluent: THF:WATER:ACN(50:20:30); Column: Redisep 40gm C18, 20-40 micron; Temperature: Ambient; Mobile phase A: Water; Mobile phase B: Acetonitrile; Compound elution (%) : 52% Acetonitrile/ water, Flow :30 mL/min) to obtain 2-(4-cyclopropyl-6- methoxypyrimidin-5-yl)-4-(l-(4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)ethoxy)pyrido[2,3-d]pyrimidine (30 mg, 0.049 mmol, 27.7 % yield) as off-white solid. LCMS (ESI) m/z: 576.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 9.25 (dd, J = 2.0, 4.4 Hz, 1H), 8.81 (dd, J= 2.0, 8.3 Hz, 1H), 8.71 (s, 1H), 8.18 (d, J= 1.3 Hz, 1H), 7.81 (dd, J= 4.4, 8.1 Hz, 1H), 7.71 (d, J= 8.3 Hz, 2H), 7.58 (d, J= 8.4 Hz, 2H), 6.58 (q, J= 6.4 Hz, 1H), 4.52 - 4.41 (m, 1H), 3.82 (s, 3H), 1.78 (d, J = 6.5 Hz, 3H), 1.80 - 1.72 (m, 1H), 1.48 - 1.35 (m, 8H), 1.05 (dd, J= 2.6, 4.4 Hz, 2H).
Example 66: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(4-(l-methyl- 4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)pyrido[2,3-d]pyrimidine:
To a stirred solution of 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)pyrido[2,3-d]pyrimidine (80 mg, 0.198 mmol) in 1,4-dioxane (4 mL), potassium phosphate tribasic (126 mg, 0.594 mmol), (4-cyclopropyl-6-methoxypyrimidin-5- yl)boronic acid (57.7 mg, 0.297 mmol) and tetrakis(triphenylphosphine)palladium(0) (22.89 mg, 0.020 mmol) were added under nitrogen atmosphere and reaction mixture was heated at 120 °C for 3 h under MW irradiation. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (30:30:40); X-Bridge Prep C8 (250 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/30, 15/70] to obtain 2-(4- cy cl opropyl-6-methoxypyrimidin-5-yl)-4-(4-(l-methyl-4-(tri fluoromethyl)- IH-imidazol- 2-yl)benzyl)pyrido[2,3-d]pyrimidine (28 mg, 0.051 mmol, 25.8 % yield) as pale yellow solid. LCMS (ESI) m/z: 518.0 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 9.30 (dd, J = 1.9, 4.4 Hz, 1H), 9.05 (dd, J= 1.9, 8.4 Hz, 1H), 8.73 (s, 1H), 7.90 (d, J= 1.3 Hz, 1H), 7.85 (dd, J= 4.4, 8.4 Hz, 1H), 7.68 - 7.63 (m, 2H), 7.54 (d, J= 8.4 Hz, 2H), 4.85 (s, 2H), 3.88 (s, 3H), 3.74 (s, 3H), 1.83 - 1.69 (m, 1H), 1.12 - 1.06 (m, 2H), 0.93 - 0.86 (m, 2H).
Intermediate 69: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine:
A solution of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(4-(trifluoromethyl)-l- ((2-(trimethylsilyl)ethoxy)methyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (300 mg, 0.462 mmol) in trifluoroacetic acid (10 mL) was stirred at 0 °C (in ice bath) for 1 h, during which the temperature of the solution was allowed to rise to ambient temperature and continued stirring at 25 °C for 4 h. After completion of the reaction (monitored by TLC and UPLC-MS), saturated aqueous sodium bicarbonate was added to the reaction mixture at 0 °C and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure and the crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (300 mg, 0.577 mmol) as yellow gum. LCMS (ESI) m/z: 520.0 [M+H]+.
Example 67: Synthesis of methyl 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5- yl)pyrido[2,3-d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH-imidazol- l-yl)acetate:
To a stirred solution of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (150 mg, 0.289 mmol) in acetonitrile (10 mL), cesium carbonate (188 mg, 0.577 mmol) and methyl 2- bromoacetate (0.041 mL, 0.433 mmol) were added and reaction mixture was heated at 80 °C for 16 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (20:40:40); X-Bridge C18 (150 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/40, 10/80] to obtain methyl 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)pyrido[2,3- d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH-imidazol-l-yl)acetate (18 mg, 0.029 mmol, 10.04 % yield) as off-white solid. LCMS (ESI) m/z: 592.2 [M+H]+. 1H- NMR (400 MHz, DMSO-d6): 5 9.25 (dd, J = 2.0, 4.4 Hz, 1H), 8.74 (d, J = 2.0 Hz, 1H), 8.72 (d, J = 2.0 Hz, 1H), 7.99 (q, J = 1.2 Hz, 1H), 7.79 (dd, J = 4.4, 8.0 Hz, 1H), 7.70 (d, J = 8.4 Hz, 2H), 7.61 (d, J = 8.4 Hz, 2H), 5.75 (s, 2H), 5.09 (s, 2H), 3.87 (s, 3H), 3.64 (s, 3H), 1.85-1.87 (m, 1H), 1.07-1.10 (m, 2H), 0.86-0.91 (m, 2H).
Example 68: Synthesis of 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5- yl)pyrido[2,3-d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH-imidazol- l-yl)acetamide:
To a stirred solution of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(4- (trifhioromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (80 mg, 0.113 mmol) in acetonitrile (2 mL), potassium carbonate (38.9 mg, 0.281 mmol) and 2-
chloroacetamide (11.58 mg, 0.124 mmol) were added and reaction mixture was heated at 80 °C for 3 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (20:50:30); Agilent C18 (50 x21.2)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/20, 12/60] to obtain 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)pyrido[2,3-d]pyrimidin-4- yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH-imidazol-l-yl)acetamide (33 mg, 50 % yield) as off-white solid. LCMS (ESI) m/z: 577.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 9.25 (dd, J= 1.9, 4.4 Hz, 1H), 8.76 - 8.72 (m, 2H), 7.92 (d, J= 0.9 Hz, 1H), 7.79 (dd, J= 4.4, 8.1 Hz, 1H), 7.74 - 7.68 (m, 3H), 7.65 - 7.60 (m, 2H), 7.37 (s, 1H), 5.75 (s, 2H), 4.76 (s, 2H), 3.88 (s, 3H), 1.92 - 1.82 (m, 1H), 1.14 - 1.05 (m, 2H), 0.94 - 0.86 (m, 2H).
Example 69: Synthesis of 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5- yl)pyrido[2,3-d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH-imidazol- l-yl)-N-methylacetamide:
To a stirred solution of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (150 mg, 0.289 mmol) in acetonitrile (10 mL), cesium carbonate (188 mg, 0.577 mmol) and 2-chloro-N- methylacetamide (0.041 mL, 0.433 mmol) were added and reaction mixture was heated at 80 °C for 16 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined
organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (20:40:40); X-Bridge C18 (150 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/25, 10/65] to obtain 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)pyrido[2,3- d]pyrimidin-4-yl)oxy )methyl)phenyl)-4-(trifluorom ethyl)- IH-imidazol-l -yl)-N- methylacetamide (5 mg, 8.34 pmol, 2.89 % yield) as white solid. LCMS (ESI) m/z: 591.2 [M+H]+. 1H-NMR (400 MHz, DMSO-d6): 5 9.25 (dd, J = 2.0, 4.4 Hz, 1H), 8.75 (d, J = 2.0 Hz, 1H), 8.73 (d, J = 2.0 Hz, 1H), 8.17 (q, J = 4.8 Hz, 1H), 7.92 (q, J = 1.2 Hz, 1H), 7.79 (q, J = 4.4 Hz, 1H), 7.71 (d, J = 8.4 Hz, 2H), 7.63 (d, J = 8.4 Hz, 2H), 5.75 (s, 2H), 4.75 (s, 2H), 3.88 (s, 3H), 2.62 (d, J = 4.80 Hz, 3H), 1.85-1.89 (m, 1H), 1.08-1.11 (m, 2H), 0.87- 0.92 (m, 2H).
Example 70: Synthesis of 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5- yl)pyrido[2,3-d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH-imidazol- l-yl)-N,N-dimethylacetamide:
To a stirred solution of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(4- (trifhioromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrido[2,3-d]pyrimidine (100 mg, 0.141 mmol) in acetonitrile (5 mL), potassium carbonate (48.6 mg, 0.352 mmol) and 2-chloro- N,N-dimethylacetamide (18.81 mg, 0.155 mmol) were added and reaction mixture was heated at 80 °C for 16 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents
evaporated from the filtrate under reduced pressure. The crude obtained was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (30:30:40); X- Bridge C8 (250 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/40, 10/80] to obtain 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)pyrido[2,3- d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH-imidazol-l-yl)-N,N- dimethylacetamide (33 mg, 38 % yield) as off-white solid. LCMS (ESI) m/z: 605.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 9.25 (dd, J= 2.0, 4.4 Hz, 1H), 8.73 (s, 1H), 8.73 (dd, J= 2.0, 8.1 Hz, 1H), 7.84 (d, J= 1.3 Hz, 1H), 7.81 - 7.77 (m, 1H), 7.71 - 7.68 (m, 2H), 7.60 (d, J= 8.4 Hz, 2H), 5.74 (s, 2H), 5.09 (s, 2H), 3.88 (s, 3H), 2.98 (s, 3H), 2.86 (s, 3H), 1.92 - 1.80 (m, 1H), 1.14 - 1.05 (m, 2H), 0.93 - 0.85 (m, 2H).
Example 71 and 72: Synthesis of 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5- yl)pyrido[2,3-d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH-imidazol- l-yl)ethan-l-ol and 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-7,8- dihydropyrido[2,3-d]pyrimidin-4-yl)oxy)methyl)phenyl)-4-(trifluoromethyl)-lH- imidazol-l-yl)ethan-l-ol:
To a stirred solution of methyl 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5- yl)pyrido[2, 3 -d]pyrimidin-4-yl)oxy )methyl)phenyl)-4-(tri fluoromethyl)- IH-imidazol-l- yl)acetate (100 mg, 0.169 mmol) in THF (10 mL), lithium aluminium hydride (2 M in THF, 0.169 mL, 0.338 mmol) was added at 0 °C and the mixture was stirred at 25 °C for 2 h. After completion of the reaction (monitored by TLC), saturated aqueous NH4CI was added and extracted with ethyl acetate. The organic extract was dried over anhydrous Na2SO4, filtered and solvents evaporated from the filtrate. The crude obtained was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (20:50:30); X-
Select C18 (150 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/30, 15/70] to obtain example 71 2-(2-(4-(((2-(4-cyclopropyl-6-methoxypyrimidin-5- yl)pyrido[2, 3 -d]pyrimidin-4-yl)oxy )methyl)phenyl)-4-(tri fluoromethyl)- IH-imidazol-l- yl)ethan-l-ol (20 mg, 0.035 mmol, 20.77 % yield) as white solid. LCMS (ESI) m/z: 564.2 [M+H]+. 'HNMR (400 MHz, DMSO-de) 5 = 9.25 (dd, J= 2.0, 4.4 Hz, 1H), 8.76 - 8.72 (m, 2H), 8.00 (d, J= 1.3 Hz, 1H), 7.78 (dd, J = 4.4, 8.2 Hz, 1H), 7.76 - 7.73 (m, 2H), 7.72 - 7.69 (m, 2H), 5.75 (s, 2H), 5.09 (t, J= 4.9 Hz, 1H), 4.11 (t, J= 5.4 Hz, 2H), 3.87 (s, 3H), 3.72 (q, J= 5.0 Hz, 2H), 1.95 - 1.79 (m, 1H), 1.14 - 1.05 (m, 2H), 0.94 - 0.84 (m, 2H). The over reduced compound example 72: 2-(2-(4-(((2-(4-cyclopropyl-6- methoxypyrimidin-5-yl)-7,8-dihydropyrido[2,3-d]pyrimidin-4-yl)oxy)methyl)phenyl)-4- (trifluoromethyl)-lH-imidazol-l-yl)ethan-l-ol (15 mg, 0.026 mmol, 15.29 % yield) was also obtained as white solid. LCMS (ESI) m/z: 566.2 [M+H]+. 'HNMR (400 MHz, DMSO- d6) 5 = 8.60 (s, 1H), 7.99 (s, 1H), 7.70 (d, J = 7.0 Hz, 2H), 7.53 (d, J = 7.5 Hz, 2H), 7.30 (s, 1H), 6.38 (d, J= 10.0 Hz, 1H), 5.58 (d, J= 9.8 Hz, 1H), 5.39 (s, 2H), 5.09 (s, 1H), 4.36
(s, 2H), 4.10 (s, 2H), 3.86 (s, 3H), 3.72 (s, 2H), 1.85 - 1.73 (m, 1H), 1.03 - 0.97 (m, 2H), 0.93 - 0.84 (m, 2H).
Scheme 21: Syntheses of example 73 to example 80
Intermediate 70: Synthesis of 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol- 2-yl)benzyl)oxy)quinazoline:
To a stirred solution of (4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)methanol (566 mg, 2.211 mmol) in acetonitrile (10 mL), cesium carbonate (1.310 g, 4.02 mmol) and 2,4-dichloroquinazoline (400 mg, 2.010 mmol) were added and reaction mixture was heated at 80 °C for 16 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain 2-chloro-4-((4-(l- methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)quinazoline (420 mg, 0.919 mmol, 45.7 % yield) as off-white solid. LCMS (ESI) m/z: 419.1 [M+H]+.
Intermediate 71 : Synthesis of 2-chloro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)oxy)quinazoline:
To a stirred solution of (4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)methanol (314 mg, 1.105 mmol) in acetonitrile (10 mL) was added cesium carbonate (655 mg, 2.010 mmol) and 2,4-dichloroquinazoline (200 mg, 1.005 mmol) and reaction mixture was heated at 80 °C for 16 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain 2-chloro-4-((4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)quinazoline (220 mg, 0.438 mmol, 43.6 % yield) as off-white solid. LCMS (ESI) m/z: 447.2 [M+H]+.
Intermediate 72: Synthesis of 2-chloro-4-((4-(5-methyl-3-(trifluoromethyl)-lH-pyrazol-l- yl)benzyl)oxy)quinazoline:
To a stirred solution of ((4-(5-methyl-3-(trifluoromethyl)-lH-pyrazol-l- yl)phenyl)methanol (354 mg, 1.382 mmol) in acetonitrile (15 mL) was added cesium carbonate (819 mg, 2.51 mmol) and 2,4-dichloroquinazoline (250 mg, 1.256 mmol) and reaction mixture was heated at 80 °C for 16 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain 2-chloro-4-((4-(5- methyl-3-(trifluoromethyl)-lH-pyrazol-l-yl)benzyl)oxy)quinazoline (160 mg, 0.382 mmol, 30.4 % yield) as white solid. LCMS (ESI) m/z: 419.2 [M+H]+.
Intermediate 73: Synthesis of 2-chloro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)oxy)-5-methoxyquinazoline:
To a stirred solution of (4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)methanol (186 mg, 0.655 mmol) in acetonitrile (4 mL) was added cesium carbonate (427 mg, 1.310 mmol) and 2,4-dichloro-5-methoxyquinazoline (150 mg, 0.655 mmol) and reaction mixture was heated at 80 °C for 3 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain 2- chloro-4-((4-(l -isopropyl-4-(tri fluoromethyl)- IH-imidazol -2 -yl)benzyl)oxy)-5- methoxyquinazoline (100 mg, 0.186 mmol, 28.3 % yield) as off-white solid. LCMS (ESI) m/z: 477.1 [M+H]+.
Intermediate 74: Synthesis of 2-chloro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)oxy)-6-methoxyquinazoline:
To a stirred solution of (4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)methanol (205 mg, 0.720 mmol) in acetonitrile (5 mL), cesium carbonate (427 mg, 1.310 mmol) and 2,4-dichloro-6-methoxyquinazoline (150 mg, 0.655 mmol) were added and reaction mixture was heated at 80 °C for 6 h. After completion of the reaction (monitored by UPLC-MS and TLC), solvents evaporated, water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain 2- chloro-4-((4-(l -isopropyl-4-(tri fluoromethyl)- IH-imidazol -2 -yl)benzyl)oxy)-6- methoxyquinazoline (180 mg, 0.245 mmol, 37.5 % yield) as pale yellow gum. LCMS (ESI) m/z: 477.2 [M+H]+.
Intermediate 75: Synthesis of 2-chloro-5-fluoro-4-((4-(l-isopropyl-4-(trifluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)quinazoline:
To a stirred solution of 2,4-dichloro-5-fluoroquinazoline (150 mg, 0.691 mmol) and (4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)phenyl)methanol (196 mg, 0.691 mmol) in anhydrous tetrahydrofuran (5 mL), sodium bis(trimethylsilyl)amide (NaHMDS) (1 M solution in THF, 1.037 mL, 1.037 mmol) was added at 0 °C and the mixture was stirred at 25 °C for 2 h. After completion of reaction (monitored by UPLC-MS and TLC), the reaction mixture was again cooled to 0 °C, chilled saturated ammonium chloride solution was added and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by column chromatography on silica gel, 60-120 mesh, to obtain 2- chloro-5-fluoro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2-
yl)benzyl)oxy)quinazoline (120 mg, 0.250 mmol, 36.2 % yield) as pale yellow gum. LCMS (ESI) m/z: 465.0 [M+H]+.
Intermediate 76: Synthesis of 2-chloro-6-fluoro-4-((4-(l-isopropyl-4-(trifluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)quinazoline:
To a stirred solution of 2,4-dichloro-6-fluoroquinazoline (150 mg, 0.691 mmol) and (4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)phenyl)methanol (196 mg, 0.691 mmol) in anhydrous tetrahydrofuran (4 mL), sodium bis(trimethylsilyl)amide (NaHMDS) (1 M solution in THF, 1.037 mL, 1.037 mmol) was added at 0 °C and the mixture was stirred at 25 °C for 2 h. After completion of reaction (monitored by UPLC-MS and TLC), the reaction mixture was again cooled to 0 °C, chilled saturated ammonium chloride solution was added and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by column chromatography on silica gel, 60-120 mesh, to obtain 2- chloro-6-fluoro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)quinazoline (80 mg, 0.143 mmol, 20.67 % yield) as pale yellow gum. LCMS (ESI) m/z: 465.0 [M+H]+.
Example 73: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)quinazoline:
To a stirred solution of 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)quinazoline (120 mg, 0.287 mmol) in a mixture of 1,4-dioxane and water (10 mL, 9: 1), potassium phosphate tribasic (122 mg, 0.573 mmol), 4-cyclopropyl-6- methoxy-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)pyrimidine (87 mg, 0.315 mmol) and tetrakis(triphenylphosphine)palladium(0) (33.1 mg, 0.029 mmol) were added under nitrogen atmosphere, and reaction mixture was heated at 120 °C for 2 h under MW irradiation. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (20:40:40); Sunfire C18 (250 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 13 mL/min; Time/Grad : 0/60, 12/90] to obtain 2-(4-cyclopropyl-6-methoxypyrimidin-5- yl)-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)quinazoline (30 mg, 0.056 mmol, 19.56 % yield) as white solid. LCMS (ESI) m/z: 533.2 [M+H]+. (400 MHz, DMSO-dd) 3 = 8.71 (s, 1H), 8.30 (d, J= 8.0 Hz, 1H), 8.08-8.00 (m, 2H), 7.95 (d, J= 1.2 Hz, 1H), 7.81-7.75 (m, 3H), 7.70 (d, J = 8.4 Hz, 2H), 5.74 (s, 2H), 3.87 (s, 3H), 3.80 (s, 3H), 1.82-1.74 (m, 1H), 1.10-1.05 (m, 2H), 0.91-0.85 (m, 2H).
Example 74: Synthesis of 2-(4-chloro-l-isopropyl-lH-pyrazol-5-yl)-4-((4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)quinazoline:
To a stirred solution of 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)quinazoline (200 mg, 0.478 mmol) in a mixture of 1,4-dioxane and water
(10 mL, 9: 1), potassium phosphate tribasic (203 mg, 0.955 mmol), 4-chloro-l-isopropyl- 5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)-lH-pyrazole (155 mg, 0.573 mmol) and tetrakis(triphenylphosphine)palladium(0) (55.2 mg, 0.048 mmol) were added under nitrogen atmosphere, and reaction mixture was heated at 80 °C for 16 h. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (20:40:40); Sunfire C18 (150 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 13 mL/min; Time/Grad
0/60, 7/90] to obtain 2-(4-chloro-l-isopropyl-lH-pyrazol-5-yl)-4-((4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)quinazoline (25 mg, 0.047 mmol, 9.88 % yield) as off-white solid. LCMS (ESI) m/z: 527.0 [M+H]+. (400 MHz, DMSO-dd) 3 8.30 (dt, J= 8.4 Hz & 0.8 Hz, 1H), 8.09-8.00 (m, 2H), 7.95 (d, J= 1.2 Hz, 1H), 7.82-7.75 (m, 4H), 7.71 (d, .7= 8.4 Hz, 2H), 5.84 (s, 2H), 5.45-5.34 (m, 1H), 3.81 (s, 3H), 1.43 (d, = 6.8 Hz, 6H).
Example 75: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)quinazoline:
To a stirred solution of 2-chloro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)quinazoline (200 mg, 0.448 mmol) in a mixture of 1,4-di oxane and water (10 mL, 9: 1), potassium phosphate tribasic (190 mg, 0.895 mmol), 4-cyclopropyl-6- methoxy-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)pyrimidine (136 mg, 0.492 mmol) and tetrakis(triphenylphosphine)palladium(0) (51.7 mg, 0.045 mmol) were added
under nitrogen atmosphere, and reaction mixture was heated at 120 °C for 2 h under MW irradiation. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (20:40:40); Sunfire C18 (250 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 13 mL/min; Time/Grad 0/60, 5/90, 10/90] to obtain 2-(4-cyclopropyl-6- methoxypyrimidin-5-yl)-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)quinazoline (30 mg, 0.053 mmol, 11.94 % yield) as white solid. LCMS (ESI) m/z: 561.2 [M+H]+. (400 MHz, DMSO-d6): 8.71 (s, 1H), 8.33-8.29 (m, 1H), 8.19 (d, J = 1.2 Hz, 1H), 8.04-7.98 (m, 2H), 7.80-7.75 (m, 1H), 7.71 (d, J= 8.4 Hz, 2H), 7.71 (d, J = 8.4 Hz, 2H), 5.74 (s, 2H), 4.52-4.40 (m, 1H), 3.86 (s, 3H), 1.79-1.71 (m, 1H), 1.41 (d, J = 6.8 Hz, 6H), 1.10-1.01 (m, 2H), 0.89-0.84 (m, 2H).
Example 76: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(5- methyl-3-(trifluoromethyl)-lH-pyrazol-l-yl)benzyl)oxy)quinazoline:
To a stirred solution of 2-chloro-4-((4-(5-methyl-3-(trifluoromethyl)-lH-pyrazol-l- yl)benzyl)oxy)quinazoline (150 mg, 0.358 mmol) in a mixture of 1,4-dioxane and water (10 mL, 9: 1), potassium phosphate tribasic (152 mg, 0.716 mmol), 4-cyclopropyl-6- methoxy-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)pyrimidine (109 mg, 0.394 mmol) and tetrakis(triphenylphosphine)palladium(0) (83 mg, 0.072 mmol) were added under nitrogen atmosphere, and reaction mixture was heated at 90 °C for 16 h. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed
with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (60: 10:30); X-Bridge C18 (150 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/60, 5/90, 7/80] to obtain 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4- (5-methyl-3-(trifluoromethyl)-lH-pyrazol-l-yl)benzyl)oxy)quinazoline (32 mg, 0.060 mmol, 16.65 % yield) as off-white solid. LCMS (ESI) m/z: 533.2 [M+H]+. (400 MHz, DMSO-d6): 5 = 8.71 (s, 1H), 8.31 (d, J= 8.4 Hz, 1H), 8.06-7.98 (m, 2H), 7.82-7.73 (m, 3H), 7.62 (d, J = 8.4 Hz, 2H), 6.78 (s, 1H), 5.75 (s, 2H), 3.87 (s, 3H), 2.37 (s, 3H), 1.83- 1.72 (m, 1H), 1.09-1.01 (m, 2H), 0.91-0.86 (m, 2H).
Example 77: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-5-methoxyquinazoline:
To a stirred solution of 2-chloro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)-5-methoxyquinazoline (90 mg, 0.167 mmol) in 1,4-dioxane (4 mL), potassium phosphate tribasic (70.9 mg, 0.334 mmol), (4-cyclopropyl-6-methoxypyrimidin- 5-yl)boronic acid (48.6 mg, 0.251 mmol) and tetrakis(triphenylphosphine)palladium(0) (19.30 mg, 0.017 mmol) were added under nitrogen atmosphere and reaction mixture was heated at 90 °C for 12 h. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC (HPLC method: Diluent: THF:WATER:ACN(40:20:40); Column: X Select CSH C18
(19x250mm) 5p; Temperature: Ambient; Mobile phase A: 5mM Ammonium Formate in water; Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/40, 10/80) to obtain 2-(4-cy cl opropyl-6-methoxypyrimidin-5-yl)-4-((4-(l-isopropyl-4-(tri fluoromethyl)- 1H- imidazol-2-yl)benzyl)oxy)-5-methoxyquinazoline (60 mg, 0.095 mmol, 57.1 % yield) as off-white solid. LCMS (ESI) m/z: 591.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 8.70 (s, 1H), 8.19 (d, J= 1.3 Hz, 1H), 7.91 (t, J= 8.3 Hz, 1H), 7.75 - 7.71 (m, 2H), 7.65 - 7.61 (m, 2H), 7.52 (dd, J= 0.8, 8.3 Hz, 1H), 7.25 (d, J= 7.6 Hz, 1H), 5.70 (s, 2H), 4.51 (spt, J = 6.6 Hz, 1H), 4.04 (s, 3H), 3.86 (s, 3H), 1.82 - 1.73 (m, 1H), 1.42 (d, = 6.6 Hz, 6H), 1.08 - 1.03 (m, 2H), 0.90 - 0.84 (m, 2H).
Example 78: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-5-fluoro-4-((4- (l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)quinazoline:
To a stirred solution of 2-chloro-5-fluoro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)oxy)quinazoline (120 mg, 0.250 mmol) in 1,4-dioxane (4 mL), potassium phosphate tribasic (106 mg, 0.501 mmol), (4-cyclopropyl-6-methoxypyrimidin- 5-yl)boronic acid (72.8 mg, 0.375 mmol) and tetrakis(triphenylphosphine)palladium(0) (28.9 mg, 0.025 mmol) were added under nitrogen atmosphere and reaction mixture was heated at 90 °C for 8 h. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase flash purification (method: Diluent: THF:WATER:ACN(50:20:30); Column: Redisep 40gm C18, 20-40 micron; Temperature: Ambient; Mobile phase A: Water; Mobile phase B: Acetonitrile; Compound elution (%) : 72% Acetonitrile/ water, Flow :30 mL/min) to obtain 2-(4-cyclopropyl-6-
methoxypyrimidin-5-yl)-5-fluoro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)quinazoline (68 mg, 0.117 mmol, 46.9 % yield) as off-white solid. LCMS (ESI) m/z: 579.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 8.72 (s, 1H), 8.19 (d, J = 1.3 Hz, 1H), 8.01 (dt, J= 5.6, 8.2 Hz, 1H), 7.85 (dd, J= 0.7, 8.4 Hz, 1H), 7.72 - 7.68 (m, 2H), 7.64 - 7.60 (m, 2H), 7.56 (ddd, J= 0.8, 8.1, 10.9 Hz, 1H), 5.75 (s, 2H), 4.50 (spt, J= 6.6 Hz, 1H), 3.87 (s, 3H), 1.86 - 1.78 (m, 1H), 1.42 (d, = 6.6 Hz, 6H), 1.10 - 1.05 (m, 2H), 0.91 - 0.85 (m, 2H).
Example 79: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-6-methoxyquinazoline:
To a stirred solution of 2-chloro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)-6-methoxyquinazoline (180 mg, 0.249 mmol) in 1,4-dioxane (4 mL), potassium phosphate tribasic (106 mg, 0.498 mmol), (4-cyclopropyl-6-methoxypyrimidin- 5-yl)boronic acid (72.5 mg, 0.374 mmol) and tetrakis(triphenylphosphine)palladium(0) (28.8 mg, 0.025 mmol) were added under nitrogen atmosphere and reaction mixture was heated at 90 °C for 12 h. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase flash purification (method: Diluent: THF:WATER:ACN(50:20:30); Column: Redisep 40gm C18, 20-40 micron; Temperature: Ambient; Mobile phase A: Water; Mobile phase B: Acetonitrile; Compound elution (%) : 72% Acetonitrile/ water, Flow :30 mL/min) to obtain 2-(4-cyclopropyl-6- methoxypyrimidin-5-yl)-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)-6-methoxyquinazoline (70 mg, 0.118 mmol, 47.4 % yield) as off-white
solid. LCMS (ESI) m/z: 591.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 8.69 (s, 1H), 8.19 (d, J= 1.3 Hz, 1H), 7.95 (d, J= 9.1 Hz, 1H), 7.72 - 7.69 (m, 2H), 7.66 (dd, J= 2.9, 9.1 Hz, 1H), 7.63 - 7.59 (m, 2H), 7.55 (d, J = 2.9 Hz, 1H), 5.76 (s, 2H), 4.49 (spt, J = 6.6 Hz, 1H), 3.96 (s, 3H), 3.85 (s, 3H), 1.80 - 1.68 (m, 1H), 1.42 (d, J = 6.6 Hz, 6H), 1.08 - 1.01 (m, 2H), 0.88 - 0.81 (m, 2H).
Example 80: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-6-fluoro-4-((4- (l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)quinazoline:
To a stirred solution of 2-chloro-6-fluoro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)oxy)quinazoline (80 mg, 0.143 mmol) in 1,4-dioxane (4 mL), potassium phosphate tribasic (60.6 mg, 0.286 mmol), (4-cyclopropyl-6-methoxypyrimidin- 5-yl)boronic acid (41.6 mg, 0.214 mmol) and tetrakis(triphenylphosphine)palladium(0) (16.51 mg, 0.014 mmol) were added under nitrogen atmosphere and reaction mixture was heated at 90 °C for 12 h. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (30:30:40); X-Bridge C8 (250 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/60, 7/90, 10/90] to obtain 2-(4- cyclopropyl-6-methoxypyrimidin-5-yl)-6-fluoro-4-((4-(l-isopropyl-4-(trifluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)quinazoline (11 mg, 0.019 mmol, 13.17 % yield) as off- white solid. LCMS (ESI) m/z: 579.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 8.71 (s,
1H), 8.19 (d, = 1.3 Hz, 1H), 8.11 (dd, J= 5.1, 9.2 Hz, 1H), 8.01 (dd, = 2.7, 8.4 Hz, 1H), 7.98 - 7.92 (m, 1H), 7.72 (d, J = 8.3 Hz, 2H), 7.61 (d, J= 8.4 Hz, 2H), 5.74 (s, 2H), 4.49 (spt, J= 6.6 Hz, 1H), 3.85 (s, 3H), 1.81 - 1.72 (m, 1H), 1.41 (d, J = 6.6 Hz, 6H), 1.09 - 1.04 (m, 2H), 0.89 - 0.83 (m, 2H).
Intermediate 77: Synthesis of tert-butyl 2,4-dichloro-5-oxo-7,8-dihydropyrido[4,3- d]pyrimidine-6(5H)-carboxylate:
To a stirred solution of tert-butyl 2,4-dichloro-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)- carboxylate (1.0 g, 3.290 mmol) in ethyl acetate (1 mL) and water (1 mL), sodium (meta)periodate (3.38 g, 15.780 mmol) and Ruthenium(III) chloride hydrate (0.136 g, 0.658 mmol) were added and the reaction mixture was stirred at room temperature fori h. After completion of the reaction (monitored by UPLC-MS and TLC), water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure.
The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, using 25-30% gradient of ethyl acetate in petroleum ether to obtain tert-butyl 2,4-dichloro- 5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate (700 mg, 2.115 mmol, 64.3% yield) as white solid, LCMS (ESI) m/z: 316.0 [M-H]+.
Intermediate 78: Synthesis of tert-butyl 2-chloro-4-((4-(l-isopropyl-4-(trifluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate:
To a stirred solution of (4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)phenyl)methanol (1.028 g, 3.620 mmol), tert-butyl 2,4-dichloro-7,8-dihydropyrido[4,3- d]pyrimidine-6(5H)-carboxylate (1.0 g, 3.290 mmol) in THF (10 mL), lithium bis(trimethylsilyl)amide solution (1 M in THF, 4.93 mL, 4.930 mmol) was added and the reaction mixture was stirred at room temperature for 16 h. After completion of the reaction (monitored by UPLC-MS and TLC), reaction mixture was quenched with NH4Q and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, using 30-40% gradient of ethyl acetate in petroleum ether to obtain tert-butyl 2-chloro-4- ((4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-7,8- dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate (1.1 g, 1.878 mmol, 57.1% yield) as white solid, LCMS (ESI) m/z: 552.2 [M+H]+.
Intermediate 79: Synthesis of tert-butyl 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4- ((4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-7,8- dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate:
To a stirred solution of tert-butyl 2-chloro-4-((4-(l-isopropyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)oxy)-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate (1.0 g, 1.707 mmol) and (4-cyclopropyl-6-methoxypyrimidin-5-yl)boronic acid (0.397 g, 2.048 mmol) in 1,4-dioxane (10 mL), tripotassium phosphate (0.725 g, 3.410 mmol) and tetrakis(triphenylphosphine)palladium(0) (0.197 g, 0.171 mmol) were added and reaction mixture was purged with nitrogen gas for 2 min and the reaction mixture was stirred at 80 °C for 2 h. After completion of the reaction (monitored by UPLC-MS and TLC), water was added and extracted with ethyl acetate. Organic extract was dried over anhydrous sodium sulfate and concentrated under reduced pressure to get crude product which is purified by flash chromatography on silica gel, 230-400 mesh, using 35% gradient of ethyl acetate in petroleum ether to obtain tert-butyl 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-7,8-dihydropyrido[4,3- d]pyrimidine-6(5H)-carboxylate (800 mg, 1.175 mmol, 68.8% yield) as off-white solid. LCMS (ESI) m/z: 666.6 [M+H]+.
Intermediate 80: Synthesis of tert-butyl 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4- ((4-(l -isopropyl-4-(tri fluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)-5-oxo-7, 8- dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate:
To a stirred solution of tert-butyl 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-7,8-dihydropyrido[4,3- d]pyrimidine-6(5H)-carboxylate (200 mg, 0.294 mmol) in 1,2-di chloroethane (5 mL) were added potassium permanganate (93 mg, 0.588 mmol) and 18-Crown-6 (7.76 mg, 0.029 mmol) at 0 °C and the reaction mixture was stirred at room temperature for 2 h. After completion of reaction (monitored by UPLC-MS), water was added to the reaction mixture and extracted with ethyl acetate. Organic extract was concentrated under reduced pressure to obtain crude product which purified flash chromatography on silica gel, 230-400 mesh, using 38% gradient of ethyl acetate in petroleum ether to obtain tert-butyl 2-(4-cyclopropyl- 6-methoxypyrimidin-5-yl)-4-((4-(l -isopropyl-4-(tri fluoromethyl)- lH-imidazol-2- yl)benzyl)oxy)-5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate (51 mg, 0.061 mmol, 20.75% yield) as off white solid. LCMS (ESI) m/z: 680.3 [M+H]+.
Example 81: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-7,8-dihydropyrido[4,3- d]pyrimidin-5(6H)-one:
To a stirred solution of tert-butyl 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- i sopropyl-4-(tri fluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)-5-oxo-7, 8- dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate (50 mg, 0.060 mmol) in dichloromethane (2 mL), trifluoroacetic acid (0.023 mL, 0.299 mmol) was added at 0 °C and the reaction mixture was stirred at room temperature for 30 min. After completion of reaction (monitored by UPLC-MS), water was added to the reaction mixture and neutralized with aqueous solution of sodium bicarbonate and extracted with ethyl acetate. Organic extract was concentrated under reduced pressure to obtain crude product, which was purified by prep HPLC (method: Diluent : THF:WATER:ACN(40: 10:40); Column : XBridge-C8(250 xl9)mm, 5micron; Temperature : Ambient; Mobile phase A: 5mM Ammonium Formate; Mobile phase B: Acetonitrile; Flow :15 mL/min; Time/Grad : 0/40, 7/80, 9/80) to obtain 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l-isopropyl-4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-7,8-dihydropyrido[4,3-d]pyrimidin- 5(6H)-one (12 mg, 33.9% yield) as off white solid. LCMS (ESI) m/z: 580.8 [M+H]+. JH NMR (400 MHz, DMSO-de) 5 = 8.69 (s, 1H), 8.18 (d, J = 1.2 Hz, 1H), 8.04 (bs, 1H), 7.67 (d, J = 8.4 Hz, 2H), 7.58 (d, J = 8.4 Hz, 2H), 5.60 (s, 2H), 4.52-4.45 (m, 1H), 3.87 (s, 3H), 3.46-3.42 (m, 2H), 3.03 (t, J = 6.8 Hz, 2H), 1.80-1.74 (m, 1H), 1.42 (d, J = 6.8 Hz, 6H), 1.06-1.04 (m, 2H), 0.91-0.89 (m, 2H).
Scheme 22: Syntheses of example 82 and 83
Intermediate 81 : Synthesis of tert-butyl 2,4-dichloro-5-oxo-7,8-dihydropyrido[4,3- d]pyrimidine-6(5H)-carboxylate:
To a stirred solution of tert-butyl 2,4-dichloro-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)- carboxylate (1.0 g, 3.290 mmol) in ethyl acetate (1 mL) and water (1 mL), sodium (meta)periodate (3.38 g, 15.780 mmol) and Ruthenium(III) chloride hydrate (0.136 g, 0.658 mmol) were added and the reaction mixture was stirred at room temperature fori h. After completion of the reaction (monitored by UPLC-MS and TLC), water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure.
The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, to obtain tert-butyl 2,4-dichloro-5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)- carboxylate (700 mg, 2.115 mmol, 64.3% yield) as white solid, LCMS (ESI) m/z: 316.0 [M-H]+.
Intermediate 82: Synthesis of tert-butyl 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)-5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate:
To a stirred solution of zinc (123 mg, 1.886 mmol) and 1,2-dibromoethane (8.13 pl, 0.094 mmol) in THF (10 mL), trimethylsilyl chloride (0.012 mL, 0.094 mmol) was added under nitrogen atmosphere and the mixture was heated at 65 °C for 1 h. Then 2-(4- (bromomethyl)phenyl)-l-methyl-4-(trifluoromethyl)-lH-imidazole (331 mg, 1.037 mmol) in THF (2 mL) was added to the reaction mixture and continued stirring at 65 °C for 10 min. After that, tert-butyl 2,4-dichloro-5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)- carboxylate (300 mg, 0.943 mmol) and tetrakis(triphenylphosphine)palladium(0) (109 mg, 0.094 mmol) were added under nitrogen atmosphere and the mixture was heated at 65 °C for 2 h. After completion of the reaction (monitored by UPLC-MS and TLC), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude. The crude obtained was purified by flash chromatography on silica gel, 230- 400 mesh, to obtain tert-butyl 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)-5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate (110 mg, 0.211 mmol, 22.35 % yield) as pale yellow gum, LCMS (ESI) m/z: 520.0 [M-H]+.
Intermediate 83: Synthesis of tert-butyl 2-chloro-4-(4-(l-isopropyl-4-(trifluoromethyl)- lH-imidazol-2-yl)benzyl)-5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate:
To a stirred solution of l-isopropyl-4-(trifluoromethyl)-2-(4- ((trimethylstannyl)methyl)phenyl)-lH-imidazole (134 mg, 0.311 mmol) in 1,4-dioxane (10 mL), tert-butyl 2,4-dichloro-5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)- carboxylate (90 mg, 0.283 mmol) was added and purged with N2 gas for 10 min. Then XPhos Pd G3 (47.9 mg, 0.057 mmol) was added at 25 °C and reaction mixture was heated at 110 °C for 16 h. After completion of reaction (monitored by UPLC-MS and TLC), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by column chromatography on silica gel, 60- 120 mesh, to obtain tert-butyl 2-chloro-4-(4-(l-isopropyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)-5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate (30 mg, 0.055 mmol, 19.28 % yield) as pale yellow gum. LCMS (ESI) m/z: 550.0 [M+H]+.
Intermediate 84: Synthesis of 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol- 2-yl)benzyl)-7,8-dihydropyrido[4,3-d]pyrimidin-5(6H)-one:
A solution of tert-butyl 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)-5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate (110 mg, 0.211 mmol) in trifluoroacetic acid (3 mL) was stirred at 0 °C (in ice bath) for 1 h, during which the temperature of the solution was allowed to rise to ambient temperature and continued stirring at 25 °C for 1 h. After completion of the reaction (monitored by TLC and UPLC- MS), saturated aqueous sodium bicarbonate was added to the reaction mixture at 0 °C and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure and the crude obtained was purified by flash chromatography on silica gel, 60-120 mesh, to obtain 2- chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)-7,8- dihydropyrido[4,3-d]pyrimidin-5(6H)-one (70 mg, 0.166 mmol, 79 % yield) as pale yellow gum. LCMS (ESI) m/z: 422.0 [M+H]+.
Intermediate 85: Synthesis of 2-chloro-4-(4-(l-isopropyl-4-(trifluoromethyl)-lH- imidazol-2-yl)benzyl)-7,8-dihydropyrido[4,3-d]pyrimidin-5(6H)-one:
A solution of tert-butyl 2-chloro-4-(4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)-5-oxo-7,8-dihydropyrido[4,3-d]pyrimidine-6(5H)-carboxylate (30 mg, 0.055 mmol) in trifluoroacetic acid (1 mL) was stirred at 0 °C (in ice bath) for 1 h, during which the temperature of the solution was allowed to rise to ambient temperature and continued stirring at 25 °C for 30 min. After completion of the reaction (monitored by TLC and UPLC-MS), saturated aqueous sodium bicarbonate was added to the reaction mixture at 0 °C and extracted with ethyl acetate. The organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure and the crude obtained was purified by flash chromatography on silica gel, 60-120 mesh, to obtain
2-chloro-4-(4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)-7,8- dihydropyrido[4,3-d]pyrimidin-5(6H)-one (20 mg, 0.044 mmol, 82 % yield) as brown gum. LCMS (ESI) m/z: 450.0 [M+H]+.
Example 82: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(4-(l-methyl- 4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)-7,8-dihydropyrido[4,3-d]pyrimidin- 5(6H)-one:
To a stirred solution of 2-chloro-4-(4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)-7,8-dihydropyrido[4,3-d]pyrimidin-5(6H)-one (100 mg, 0.237 mmol) in a mixture of 1,4-dioxane and water (5 mL, 9: 1), potassium phosphate tribasic (101 mg, 0.474 mmol), 4-cyclopropyl-6-methoxy-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2- yl)pyrimidine (72.0 mg, 0.261 mmol) and tetrakis(triphenylphosphine)palladium(0) (27.4 mg, 0.024 mmol) were added under nitrogen atmosphere at 25 °C and the reaction mixture was heated at 90 °C for 16 h. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC (HPLC method: Diluent: THF:WATER:ACN(40:20:40); Column: X-Select C8 OBD (19x150mm) 5p; Temperature: Ambient; Mobile phase A: 5mM Ammonium Formate in Water; Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/30, 12/70) to obtain 2-(4- cy cl opropyl-6-methoxypyrimidin-5-yl)-4-(4-(l-methyl-4-(tri fluoromethyl)- IH-imidazol- 2-yl)benzyl)-7,8-dihydropyrido[4,3-d]pyrimidin-5(6H)-one (32 mg, 0.060 mmol, 25.2 % yield) as white solid. LCMS (ESI) m/z: 536.3 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 8.68 (s, 1H), 8.35 (s, 1H), 7.90 (d, J= 1.1 Hz, 1H), 7.60 (d, J= 8.3 Hz, 2H), 7.44 (d, J=
8.4 Hz, 2H), 4.74 (s, 2H), 3.85 (s, 3H), 3.75 (s, 3H), 3.46 (dt, J= 3.2, 6.5 Hz, 2H), 3.09 (t, J= 6.6 Hz, 2H), 1.70 - 1.60 (m, 1H), 1.08 - 0.99 (m, 2H), 0.88 - 0.82 (m, 2H).
Example 83: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-(4-(l- isopropyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)-7,8-dihydropyrido[4,3- d]pyrimidin-5(6H)-one:
To a stirred solution of 2-chloro-4-(4-(l-isopropyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)-7,8-dihydropyrido[4,3-d]pyrimidin-5(6H)-one (60 mg, 0.133 mmol) in a mixture of 1,4-dioxane and water (5 mL, 9: 1), potassium phosphate tribasic (56.6 mg, 0.267 mmol), 4-cyclopropyl-6-methoxy-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2- yl)pyrimidine (40.5 mg, 0.147 mmol) and tetrakis(triphenylphosphine)palladium(0) (15.41 mg, 0.013 mmol) were added under nitrogen atmosphere at 25 °C and the reaction mixture was heated at 90 °C for 16 h. After completion of reaction (monitored by UPLC-MS), the reaction mixture was cooled to ambient temperature and filtered through a celite bed. The celite bed was further washed with ethyl acetate and the washings were mixed with the filtrate. Solvents evaporated from the mixture of filtrate and washings under reduced pressure to obtain a crude, which was purified by reverse phase preparative HPLC (HPLC method: Diluent: THF: ACN(50:50); Column: X-Bridge C18 (21.2x250mm) 5p; Temperature: Ambient; Mobile phase A: 0.1% TFA in Water; Mobile phase B: Acetonitrile; Flow : 15 mL/min; Time/Grad : 0/50, 12.75/85.80) to obtain 2-(4-cyclopropyl- 6-methoxypyrimidin-5-yl)-4-(4-(l -isopropyl -4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)-7,8-dihydropyrido[4,3-d]pyrimidin-5(6H)-one (15 mg, 0.025 mmol, 18.86 % yield) as white solid. LCMS (ESI) m/z: 564.2 [M+H]+. 'H NMR (400 MHz, DMSO-de) 5 = 8.68 (s, 1H), 8.36 (s, 1H), 8.15 (d, J = 1.3 Hz, 1H), 7.46 (s, 4H), 4.75 (s, 2H), 4.44 (spt,
J= 6.7 Hz, 1H), 3.86 (s, 3H), 3.47 (dt, J= 3.1, 6.5 Hz, 2H), 3.10 (t, J= 6.6 Hz, 2H), 1.69 - 1.61 (m, 1H), 1.39 (d, J= 6.6 Hz, 6H), 1.06 - 1.01 (m, 2H), 0.87 - 0.81 (m, 2H).
To a solution of 2,4,6-trichloro-5-nitropyrimidine (3.0 g, 13.13 mmol) in THF (40 mL), ammonia in methanol (7 M, 1.876 mL, 13.13 mmol) was added at -78 °C and the mixture was allowed to stir at -78 °C for 30 min. After completion of reaction (monitored by UPLC- MS and TLC), the reaction mixture was allowed to warm to ambient temperature, filtered, washed with ethyl acetate and filtrate was concentrated under reduced pressure. The residue obtained was washed with petroleum ether and dried to obtain crude 2,6-dichloro-5- nitropyrimidin-4-amine (2.8 g, 13.40 mmol, 100%) as pale yellow solid, which was used as such for the next reaction. LCMS (ESI) m/z: 208.9 [M+H]+.
Intermediate 87: Synthesis of 2-chloro-6-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol- 2-yl)benzyl)oxy)-5-nitropyrimidin-4-amine:
To a stirred solution of 2,6-dichloro-5-nitropyrimidin-4-amine (1.0 g, 4.79 mmol) in tetrahydrofuran (15 mL), NaHMDS (1 M in THF, 7.18 mL, 7.18 mmol) and (4-(l-methyl-
4-(trifluoromethyl)-lH-imidazol-2-yl)phenyl)methanol (1.349 g, 5.26 mmol) were added at ambient temperature and the mixture was stirred for 3 h. Progress of the reaction was monitored by UPLC-MS. After 3 h, saturated aqueous ammonium chloride (10 mL) was added to the reaction mixture and extracted with di chloromethane. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on silica gel, 230-400 mesh, using 53-54% gradient of ethyl acetate in petroleum ether to obtain pure 2-chloro-6-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-
5-nitropyrimidin-4-amine (0.650 g, 1.213 mmol, 25.3%) as pale brown gum. LCMS (ESI) m/z: 429.0 [M+H]+.
Intermediate 88: Synthesis of 2-(4-chloro-l-isopropyl-lH-pyrazol-5-yl)-6-((4-(l-methyl- 4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-5-nitropyrimidin-4-amine:
To a stirred solution of 2-chloro-6-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)-5-nitropyrimidin-4-amine (0.650 g, 1.52 mmol) in 1,4-dioxane (2 mL), 4-
chloro-l-isopropyl-5-(4,4,5,5-tetramethyl-l,3,2-dioxaborolan-2-yl)-lH-pyrazole (0.451 g, 1.67 mmol), potassium phosphate tribasic (0.644 g, 3.03 mmol), water (0.133 mL) and tetrakis(triphenylphosphine)palladium(0) (0.350 g, 0.303 mmol) were added at ambient temperature. The reaction mixture was purged with nitrogen gas and then stirred at 45 °C for 4 h under nitrogen atmosphere. After completion of reaction (monitored by UPLC-MS and TLC), the reaction mixture was cooled to ambient temperature, mixed with another batch of the same reaction (with 0.200 g of 2-chloro-6-((4-(l-methyl-4-(trifluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)-5-nitropyrimidin-4-amine), chilled water was added and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by flash chromatography on isolara chromatography system, using ethy acetate and pet ether (3 : 7) as eluent to obtain 2-(4-chloro-l-isopropyl- lH-pyrazol-5-yl)-6-((4-(l-methyl-4-(tri fluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)-5- nitropyrimidin-4-amine (0.450 g, 0.84 mmol, 42.3%) as yellow solid. LCMS (ESI) m/z: 537.2 [M+H]+.
Intermediate 89: Synthesis of 2-(4-chl oro-1 -isopropyl- lH-pyrazol-5-yl)-6-((4-(l-methyl- 4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrimidine-4,5-diamine:
To a stirred solution of 2-(4-chloro-l-isopropyl-lH-pyrazol-5-yl)-6-((4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)-5-nitropyrimidin-4-amine (0.450 g, 0.84 mmol) in acetonitrile (15 mL), ammonium formate (0.528 g, 8.38 mmol) and zinc (0.548 g, 8.38 mmol) were added at 25 °C and stirring was continued for 2 h. After completion of reaction (monitored by UPLC-MS), the reaction mixture was filtered and solvents evaporated from the filtrate to obtain crude 2-(4-chloro-l-isopropyl-lH-pyrazol-5-yl)-6- ((4-(l -methyl-4-(tri fluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)pyrimidine-4,5-diamine
(0.400 g, 0.79 mmol, 94% yield) as yellow gum, which was used as such in the next step reaction. LCMS (ESI) m/z: 507.2 [M+H]+.
Example 84: Synthesis of 2-(4-chloro-l-isopropyl-lH-pyrazol-5-yl)-4-((4-(l-methyl- 4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pteridin-7(8H)-one:
To a stirred solution of 2-(4-chloro-l-isopropyl-lH-pyrazol-5-yl)-6-((4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pyrimidine-4,5-diamine (0.400 g, 0.79 mmol) in ethanol (20 mL), a solution of methyl 2-oxoacetate in toluene (0.208 g, 1.18 mmol) and acetic acid (4.52 pl, 0.08 mmol) were added at ambient temperature and the mixture was heated at 90 °C for 4 h. The product 2-(4-chloro-l -isopropyl- lH-pyrazol-5- yl)-4-((4-(l-methyl-4-(tri fluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)pteridin-7(8H)-one (10 mg, 0.016 mmol, 2.080 % yield) mass was observed by UPLC-MS. After completion of reaction (monitored by UPLC-MS), chilled water was added to the reaction mixture, and extracted with ethyl acetate. The combined organic extract was dried over anhydrous sodium sulfate, filtered and solvents evaporated from the filtrate under reduced pressure. The crude obtained was purified by reverse phase preparative HPLC [Method: Diluent: WATER: THF: MeCN (30:30:40); XBridge C8 (250 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15mL/min; Time/Grad : 0/40, 7/80, 9/80] to obtain 2-(4-chloro-l- isopropyl-lH-pyrazol-5-yl)-4-((4-(l-methyl-4-(tri fluoromethyl)- IH-imidazol -2- yl)benzyl)oxy)pteridin-7(8H)-one (0.010 g, 0.016 mmol, 2.08%) as off-white solid. LCMS (ESI) m/z: 545.0 [M+H]+. 'H-NMR: (400 MHz, DMSO-dd) 3 = 13.26 (s, 1H), 8.17 (s, 1H), 7.95 (m, 1H), 7.76-7.79 (m, 3H), 7.60-7.66 (m, 2H), 5.75 (s, 2H), 5.44-5.48 (m, 1H), 3.80 (s, 3H), 1.41 (d, J = 6.40 Hz, 6H).
Intermediate 90: Synthesis of 2-chloro-6-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol- 2-yl)benzyl)oxy)pyrimidine-4,5-diamine:
To a stirred solution of 2-chloro-6-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)-5-nitropyrimidin-4- amine (0.400 g, 0.68 mmol) in mixture of ethanol (4 mL) and acetic acid (4 mL), iron powder (0.228 g, 4.09 mmol) was added at 25 °C and mixture was heated at 60 °C for 6 h. After completion of reaction (monitored by TLC and UPLC-MS), the reaction mixture was cooled to ambient temperature, water was added and extracted with ethyl acetate. The combined organic extract was washed with brine, dried over anhydrous sodium sulphate, filtered and solvents evaporated from the filtrated under reduced pressure. The crude 2-chloro-6-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol2- yl)benzyl)oxy)pyrimidine-4,5-diamine (0.700 g) obtained as a brown solid, was washed with diethyl ether, dried and used in the next step without further purification. LCMS (ESI) m/z: 399.0 [M+H]+.
Intermediate 91 : Synthesis of 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-
2-yl)benzyl)oxy)pteridin-6(5H)-one and / or 2-chloro-4-((4-(l-methyl-4- (trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pteridin-7(8H)-one:
To a stirred solution of 2-chloro-6-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)pyrimidine-4,5-diamine (0.700 g crude, considered as 1.47 mmol) in ethanol (10 mL), methyl 2-oxoacetate (0.204 mL, 1.475 mmol) and acetic acid (8.44 pL, 0.147 mmol) were added at ambient temperature and the mixture was heated at 60 °C for 3 h. Progress of the reaction was monitored by UPLC-MS. After 3 h, the reaction mixture was cooled to ambient temperature, the precipitated solid was filtered and washed with diethyl ether to obtain 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)pteridin-6(5H)-one and / or 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)- lH-imidazol-2-yl)benzyl)oxy)pteridin-7(8H)-one (0.350 g, 0.745 mmol, 50.5 %) as off- white solid, which was used in the next step as such without further purification. LCMS (ESI) m/z: 437.0 [M+H]+.
Example 85: Synthesis of 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l- methyl-4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pteridin-7(8H)-one:
To a stirred solution of 2-chloro-4-((4-(l-methyl-4-(trifluoromethyl)-lH-imidazol-2- yl)benzyl)oxy)pteridin-7(8H)-one (0.100 g, 0.213 mmol) in 1,4-dioxane (2 mL), (4- cyclopropyl-6-methoxypyrimidin-5-yl)boronic acid (0.0496 g, 0.256 mmol), cesium carbonate (0.173 g, 0.532 mmol) and Xphos-Pd-G2 (0.0168 g, 0.021 mmol) were added at 25 °C under nitrogen atmosphere and the mixture was heated at 80 °C for 16 h. Progress of the reaction was monitored by TLC and UPLC-MS. After 16 h, the reaction mixture was cooled to ambient temperature, water was added and extracted with ethyl acetate. The combined organic extract was washed with brine, dried over anhydrous sodium sulfate, filtered and solvent evaporated from the filtrate under reduced pressure. The crude obtained
was purified by preparative HPLC [Method: Diluent: THF : MeCN : WATER (10:30:60); X-SELECT-C18 (150 xl9)mm, 5micron; Temperature: Ambient; Mobile phase A: 5mM Ammonium formate in Water, Mobile phase B: Acetonitrile; Flow : 15mL/min; Time/Grad : 0/20, 3/20, 15/70] to obtain 2-(4-cyclopropyl-6-methoxypyrimidin-5-yl)-4-((4-(l-methyl- 4-(trifluoromethyl)-lH-imidazol-2-yl)benzyl)oxy)pteridin-7(8H)-one (0.0018 g, 3.05 pmol, 1.43%) as off white solid. LCMS (ESI) m/z: 551.2 [M+H]+. 'H-NMR: (400 MHz,DMSO-dd) 3 = 10.59 (s, 1H), 8.68 (d, J= 3.2 Hz, 1H), 7.93 (d, J = 1.2 Hz, 1H), 7.73 - 7.65 (m, 4H), 6.84 (d, J= 6.4 Hz, 1H), 5.51 (s, 2H), 3.84 (s, 3H), 3.79 (s, 3H), 1.72- 1.66 (m, 1H), 1.05-0.98 (m, 2H), 0.88-0.83 (m, 2H).
USP1-UAF1 Rhodamine assay
USP1/UAF1 ubiquitin-rhodamine 110 hydrolysis assays were performed at room temperature in black, low-volume 384 well plates (Corning 3821). 100X solutions of compounds in DMSO were prepared by three-fold serial dilutions starting from a 10 mM stock. 2X solutions of His6-USPl/His6-UAFl (200 pM, Internally Produced) and ubiquitin-rhodamine 110 (10 pM, South Bay Bio SBB-PS0001) were prepared in assay buffer (50 mM Tris pH 7.5, 100 mM NaCl, 1 mM EDTA, 1 mM TCEP, 100 ng/pL BSA). Serially diluted compounds in DMSO were transferred to the assay plate by acoustic dispensing (100 nL per well). 5 pL of assay buffer was added to column 1 of the plates, and 5 pL of 2X USP1/UAF1 solution was added to columns 2-24 and incubated with compounds for 3 hrs. Reactions were initiated by addition of 5 pL of 2X ubiquitin- rhodamine 110 solution to each well for final concentrations of 100 pM USP1/UAF1 and 5 pM ubiquitin-rhodamine 110. Fluorescence was read at the minimum kinetic interval for 1 h using a BioTek Synergy HTX plate reader (Agilent Technologies) with excitation at 485 nm and emission at 528 nm. Initial rates were calculated by fitting the linear range of the plot of fluorescence vs time to a linear equation. IC50 values were calculated from dose-response curves.
ICso values for compounds of the invention in the USP1-UAF1 Rhodamine assay are shown below.
Table 3: USP1-UAF1 Rhodamine Assay 3-hour Incubation Results
Claims
1. A compound having the structure of formula (I):
or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof;
R1 is selected from Ce aryl and 5-6 membered heteroaryl, optionally substituted with one to four halo, hydroxy, amino, -C(O)Ra, -C(O)ORb, -C(O)NRaRb, -N(Ra)C(O)Rb, - S(O)NRaRb, -S(O)2NRaRb, -S(O)Rg, -S(O)2Rg, -NRaRb, -ORa, -SRb, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl and C3-8 cycloalkyl; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl and C3-8 cycloalkyl is optionally substituted with one to four R100;
R2 is selected from absent, hydrogen, halo, hydroxy, amino, -CN, -C(O)Ra, -C(O)ORb, - C(O)NRaRb, -N(Ra)C(O)Rb, -N(Ra)C(O)NRaRb, -N(Ra)SO2NRaRb, -S(O)NRaRb, - S(O)2NRaRb, -N(Ra)S(O)2Rb, -S(O)Rg, -S(O)2Rg, -NRaRb, -ORa, -SRb, -OC(O)Ra, - OC(O)NRaRb C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S, and C3-8 cycloalkyl is optionally substituted with one to four R100;
X is selected from absent and C1-6 alkyl, wherein C1-6 alkyl is optionally substituted with one to four R100;
Y is absent or -O-;
Each W1, W2, W3 and W4 is independently selected from -N(Ra)-, -C(O)- and -C(Ra)-; W5 is selected from -N- and -C(Ra)-; wherein at least one of W1, W2, W3 and W4 is -C(Ra)- or -C(O)-;
G1 is selected from -Ce aryl-, 5-6 membered heteroaryl, C3-8 cycloalkyl and 5-6 membered heterocyclyl; wherein each Ce aryl, 5-6 membered heteroaryl, C3-8 cycloalkyl and 5-6 membered heterocyclyl is optionally substituted with one to four R100;
G2 is a 5 or 6 membered heteroaryl or 5-6 membered heterocyclyl optionally substituted with one to four R100; each Ra and Rb is independently selected from absent, hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-6 cycloalkyl and 4-6 membered heterocyclyl; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-6 cycloalkyl and 4-6 membered heterocyclyl is optionally substituted with one to four R200; each R100 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)RC, -C(O)ORC, -C(O)NRcRd, -N(Rc)C(O)Rd, -S(O)NRcRd, - S(O)2NRcRd, -S(O)Rh, -S(O)2Rh, -NRcRd, -ORC, -SRC, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl and 4-10 membered heterocyclyl is optionally substituted with one to four R201; each Rc and Rd is independently selected from hydrogen, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; each R200 and R201 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)Re, -C(O)ORe, -C(O)NReRf, -N(Re)C(O)Rf, -S(O)NReRf, - S(O)2NReRf, -S(O)R‘, -8(0)2^, -NReRf, -ORe, -SRe, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl and 4-10 membered heterocyclyl is optionally substituted with one to four R300; each Rg, Rh and R1 is independently selected from C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, is optionally substituted with one to four R300;
wherein each R300 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, -C(O)Re, -C(O)ORe, -C(0)NReRf, -N(Re)C(0)Rf, -S(0)NReRf, - S(0)2NReRf, -NReRf, S(O)Re, -S(O)2Re, -NReRf, -0Re, -SRe, Ci-6 alkyl, C2-6 alkenyl and C2-6 alkynyl; each Re and Rf is independently selected from hydrogen, Ci-6 alkyl, C2-6 alkenyl and C2-6 alkynyl; C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S; wherein each C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl and 4-10 membered heterocyclyl is optionally substituted with one to four R400; each R400 is independently selected from hydrogen, halo, cyano, hydroxy, amino, oxo, thioxo, vinyl, S(O)NRkR1, - S(O)2NRkR‘, -
6 alkyl, C2-6 alkenyl and C2-6 alkynyl; each Rk and R1 is independently selected from hydrogen, C1-6 alkyl, C2-6 alkenyl and C2-6 alkynyl; C3-8 cycloalkyl, Ce-io aryl, 5-10 membered heteroaryl containing 1 to 4 heteroatoms selected from N, O, and S, and 4-10 membered heterocyclyl containing 1 to 4 heteroatoms selected from N, O, and S.
Formula (II); or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof.
4. A compound of claim 1, having the structure of Formula (IVa) or (IVb):
Formula (Vile)
or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof; wherein R5 is Ci-6 alkyl.
Formula (VIIIc) or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers thereof; wherein R5 is Ci-6 alkyl.
10. A compound according to any of claims 1-9 wherein R2 is selected from:
-OCH3, -H, -SCH3, -S(O)2CH3, -S(O)2CH3, -C(O)OCH3, -C(O)OCH2CH3, and -C(O)NH2. or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers, or deuterated analog thereof.
11. A compound according to any of claims 1-3 wherein G2 is selected from:
12. A compound according to any of claims 1-11 wherein Y is selected from absent and O, or a pharmaceutically acceptable salt, stereoisomer, mixture of stereoisomers, or deuterated analog thereof.
14. A pharmaceutical composition comprising one or more compounds according to any of the above claims and a pharmaceutically acceptable carrier or diluent.
15. A method of treating or preventing a disease or disorder associated with the inhibition of ubiquitin specific protease 1 (USP1) comprising, administering to a patient in need thereof an effective amount of a compound account to any of the above claims.
16. A method for treating cancer comprising administering a therapeutically effective amount of a compound according to claims 1-13 or a pharmaceutically acceptable salt thereof, to a patient in need thereof.
17. The method according to claim 16 wherein said disease or condition is a solid tumor selected from pancreatic cancer, bladder cancer, colorectal cancer, breast cancer, prostate cancer, renal cancer, hepatocellular cancer, lung cancer, ovarian cancer, cervical cancer, gastric cancer, esophageal cancer, head and neck cancer, melanoma, neuroendocrine cancers, CNS cancers, brain tumors (e.g., glioma, anaplastic oligodendroglioma, adult glioblastoma multiforme, and adult anaplastic astrocytoma), bone cancer, and soft tissue sarcoma.
18. The method according to claim 16, wherein the cancer is pancreatic cancer, bladder cancer, colorectal cancer, breast cancer, prostate cancer, renal cancer, hepatocellular cancer, lung cancer, ovarian cancer, cervical cancer, gastric cancer, esophageal cancer, head and neck cancer, melanoma, neuroendocrine cancer, CNS cancer, brain cancer, bone cancer, soft tissue sarcoma, non-small cell lung cancer, small-cell lung cancer or colon cancer.
19. The method according to claim 16, wherein the cancer is acute lymphocytic leukemia (ALL), acute myeloid leukemia (AML), chronic lymphocytic leukemia (CLL), small lymphocytic lymphoma (SLL), myelodysplastic syndrome (MDS), myeloproliferative disease (MPD), chronic myeloid leukemia (CML), multiple myeloma (MM), non-Hodgkin's lymphoma (NHL), mantle cell lymphoma (MCL), follicular lymphoma, Waldenstrom’s macroglobulinemia (WM), T-cell lymphoma, B-cell lymphoma or diffuse large B-cell lymphoma (DLBCL).
20. The method according to any of claims 15-19, further comprising administering at least one additional anti cancer agent or therapy.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363594458P | 2023-10-31 | 2023-10-31 | |
| US63/594,458 | 2023-10-31 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025096494A1 true WO2025096494A1 (en) | 2025-05-08 |
Family
ID=93462984
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/053499 Pending WO2025096494A1 (en) | 2023-10-31 | 2024-10-30 | Ubiquitin specific processing protease 1 (usp1) compounds |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025096494A1 (en) |
Citations (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006029879A2 (en) | 2004-09-17 | 2006-03-23 | F.Hoffmann-La Roche Ag | Anti-ox40l antibodies |
| WO2006105021A2 (en) | 2005-03-25 | 2006-10-05 | Tolerrx, Inc. | Gitr binding molecules and uses therefor |
| WO2006122150A1 (en) | 2005-05-10 | 2006-11-16 | Incyte Corporation | Modulators of indoleamine 2,3-dioxygenase and methods of using the same |
| WO2007005874A2 (en) | 2005-07-01 | 2007-01-11 | Medarex, Inc. | Human monoclonal antibodies to programmed death ligand 1 (pd-l1) |
| WO2007075598A2 (en) | 2005-12-20 | 2007-07-05 | Incyte Corporation | N-hydroxyamidinoheterocycles as modulators of indoleamine 2,3-dioxygenase |
| WO2008036642A2 (en) | 2006-09-19 | 2008-03-27 | Incyte Corporation | N-hydroxyamidinoheterocycles as modulators of indoleamine 2,3-dioxygenase |
| WO2008036653A2 (en) | 2006-09-19 | 2008-03-27 | Incyte Corporation | N-hydroxyamidinoheterocycles as modulators of indoleamine 2,3-dioxygenase |
| WO2008132601A1 (en) | 2007-04-30 | 2008-11-06 | Immutep | Cytotoxic anti-lag-3 monoclonal antibody and its use in the treatment or prevention of organ transplant rejection and autoimmune disease |
| WO2009009116A2 (en) | 2007-07-12 | 2009-01-15 | Tolerx, Inc. | Combination therapies employing gitr binding molecules |
| WO2009044273A2 (en) | 2007-10-05 | 2009-04-09 | Immutep | Use of recombinant lag-3 or the derivatives thereof for eliciting monocyte immune response |
| WO2009073620A2 (en) | 2007-11-30 | 2009-06-11 | Newlink Genetics | Ido inhibitors |
| WO2010019570A2 (en) | 2008-08-11 | 2010-02-18 | Medarex, Inc. | Human antibodies that bind lymphocyte activation gene-3 (lag-3), and uses thereof |
| WO2010077634A1 (en) | 2008-12-09 | 2010-07-08 | Genentech, Inc. | Anti-pd-l1 antibodies and their use to enhance t-cell function |
| WO2011028683A1 (en) | 2009-09-03 | 2011-03-10 | Schering Corporation | Anti-gitr antibodies |
| WO2011056652A1 (en) | 2009-10-28 | 2011-05-12 | Newlink Genetics | Imidazole derivatives as ido inhibitors |
| WO2011070024A1 (en) | 2009-12-10 | 2011-06-16 | F. Hoffmann-La Roche Ag | Antibodies binding preferentially human csf1r extracellular domain 4 and their use |
| WO2011109400A2 (en) | 2010-03-04 | 2011-09-09 | Macrogenics,Inc. | Antibodies reactive with b7-h3, immunologically active fragments thereof and uses thereof |
| WO2011107553A1 (en) | 2010-03-05 | 2011-09-09 | F. Hoffmann-La Roche Ag | Antibodies against human csf-1r and uses thereof |
| WO2011131407A1 (en) | 2010-03-05 | 2011-10-27 | F. Hoffmann-La Roche Ag | Antibodies against human csf-1r and uses thereof |
| WO2011140249A2 (en) | 2010-05-04 | 2011-11-10 | Five Prime Therapeutics, Inc. | Antibodies that bind csf1r |
| WO2012032433A1 (en) | 2010-09-09 | 2012-03-15 | Pfizer Inc. | 4-1bb binding molecules |
| WO2012142237A1 (en) | 2011-04-15 | 2012-10-18 | Newlink Geneticks Corporation | Fused imidazole derivatives useful as ido inhibitors |
| WO2012145493A1 (en) | 2011-04-20 | 2012-10-26 | Amplimmune, Inc. | Antibodies and other molecules that bind b7-h1 and pd-1 |
| WO2013079174A1 (en) | 2011-11-28 | 2013-06-06 | Merck Patent Gmbh | Anti-pd-l1 antibodies and uses thereof |
| WO2013087699A1 (en) | 2011-12-15 | 2013-06-20 | F. Hoffmann-La Roche Ag | Antibodies against human csf-1r and uses thereof |
| WO2013119716A1 (en) | 2012-02-06 | 2013-08-15 | Genentech, Inc. | Compositions and methods for using csf1r inhibitors |
| WO2013132044A1 (en) | 2012-03-08 | 2013-09-12 | F. Hoffmann-La Roche Ag | Combination therapy of antibodies against human csf-1r and uses thereof |
| WO2013169264A1 (en) | 2012-05-11 | 2013-11-14 | Five Prime Therapeutics, Inc. | Methods of treating conditions with antibodies that bind colony stimulating factor 1 receptor (csf1r) |
| WO2014008218A1 (en) | 2012-07-02 | 2014-01-09 | Bristol-Myers Squibb Company | Optimization of antibodies that bind lymphocyte activation gene-3 (lag-3), and uses thereof |
| WO2014036357A1 (en) | 2012-08-31 | 2014-03-06 | Five Prime Therapeutics, Inc. | Methods of treating conditions with antibodies that bind colony stimulating factor 1 receptor (csf1r) |
| WO2023148643A1 (en) * | 2022-02-03 | 2023-08-10 | Aurigene Oncology Limited | Fused bicyclic heterocyclyl compounds as usp1 inhibitors |
| WO2024006879A1 (en) * | 2022-06-29 | 2024-01-04 | Zentaur Therapeutics Usa Inc. | Usp1 inhibitors and uses thereof |
| WO2024208292A1 (en) * | 2023-04-04 | 2024-10-10 | 江苏亚虹医药科技股份有限公司 | Ubiquitin-specific protease 1 inhibitor, preparation method therefor, and medical use thereof |
| WO2024233605A1 (en) * | 2023-05-08 | 2024-11-14 | Tango Therapeutics, Inc. | Compounds and their use against cancer |
-
2024
- 2024-10-30 WO PCT/US2024/053499 patent/WO2025096494A1/en active Pending
Patent Citations (34)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2006029879A2 (en) | 2004-09-17 | 2006-03-23 | F.Hoffmann-La Roche Ag | Anti-ox40l antibodies |
| WO2006105021A2 (en) | 2005-03-25 | 2006-10-05 | Tolerrx, Inc. | Gitr binding molecules and uses therefor |
| WO2006122150A1 (en) | 2005-05-10 | 2006-11-16 | Incyte Corporation | Modulators of indoleamine 2,3-dioxygenase and methods of using the same |
| WO2007005874A2 (en) | 2005-07-01 | 2007-01-11 | Medarex, Inc. | Human monoclonal antibodies to programmed death ligand 1 (pd-l1) |
| WO2007075598A2 (en) | 2005-12-20 | 2007-07-05 | Incyte Corporation | N-hydroxyamidinoheterocycles as modulators of indoleamine 2,3-dioxygenase |
| WO2008036642A2 (en) | 2006-09-19 | 2008-03-27 | Incyte Corporation | N-hydroxyamidinoheterocycles as modulators of indoleamine 2,3-dioxygenase |
| WO2008036653A2 (en) | 2006-09-19 | 2008-03-27 | Incyte Corporation | N-hydroxyamidinoheterocycles as modulators of indoleamine 2,3-dioxygenase |
| WO2008132601A1 (en) | 2007-04-30 | 2008-11-06 | Immutep | Cytotoxic anti-lag-3 monoclonal antibody and its use in the treatment or prevention of organ transplant rejection and autoimmune disease |
| WO2009009116A2 (en) | 2007-07-12 | 2009-01-15 | Tolerx, Inc. | Combination therapies employing gitr binding molecules |
| WO2009044273A2 (en) | 2007-10-05 | 2009-04-09 | Immutep | Use of recombinant lag-3 or the derivatives thereof for eliciting monocyte immune response |
| WO2009073620A2 (en) | 2007-11-30 | 2009-06-11 | Newlink Genetics | Ido inhibitors |
| WO2010019570A2 (en) | 2008-08-11 | 2010-02-18 | Medarex, Inc. | Human antibodies that bind lymphocyte activation gene-3 (lag-3), and uses thereof |
| WO2010077634A1 (en) | 2008-12-09 | 2010-07-08 | Genentech, Inc. | Anti-pd-l1 antibodies and their use to enhance t-cell function |
| WO2011028683A1 (en) | 2009-09-03 | 2011-03-10 | Schering Corporation | Anti-gitr antibodies |
| WO2011056652A1 (en) | 2009-10-28 | 2011-05-12 | Newlink Genetics | Imidazole derivatives as ido inhibitors |
| WO2011070024A1 (en) | 2009-12-10 | 2011-06-16 | F. Hoffmann-La Roche Ag | Antibodies binding preferentially human csf1r extracellular domain 4 and their use |
| WO2011109400A2 (en) | 2010-03-04 | 2011-09-09 | Macrogenics,Inc. | Antibodies reactive with b7-h3, immunologically active fragments thereof and uses thereof |
| WO2011107553A1 (en) | 2010-03-05 | 2011-09-09 | F. Hoffmann-La Roche Ag | Antibodies against human csf-1r and uses thereof |
| WO2011131407A1 (en) | 2010-03-05 | 2011-10-27 | F. Hoffmann-La Roche Ag | Antibodies against human csf-1r and uses thereof |
| WO2011140249A2 (en) | 2010-05-04 | 2011-11-10 | Five Prime Therapeutics, Inc. | Antibodies that bind csf1r |
| WO2012032433A1 (en) | 2010-09-09 | 2012-03-15 | Pfizer Inc. | 4-1bb binding molecules |
| WO2012142237A1 (en) | 2011-04-15 | 2012-10-18 | Newlink Geneticks Corporation | Fused imidazole derivatives useful as ido inhibitors |
| WO2012145493A1 (en) | 2011-04-20 | 2012-10-26 | Amplimmune, Inc. | Antibodies and other molecules that bind b7-h1 and pd-1 |
| WO2013079174A1 (en) | 2011-11-28 | 2013-06-06 | Merck Patent Gmbh | Anti-pd-l1 antibodies and uses thereof |
| WO2013087699A1 (en) | 2011-12-15 | 2013-06-20 | F. Hoffmann-La Roche Ag | Antibodies against human csf-1r and uses thereof |
| WO2013119716A1 (en) | 2012-02-06 | 2013-08-15 | Genentech, Inc. | Compositions and methods for using csf1r inhibitors |
| WO2013132044A1 (en) | 2012-03-08 | 2013-09-12 | F. Hoffmann-La Roche Ag | Combination therapy of antibodies against human csf-1r and uses thereof |
| WO2013169264A1 (en) | 2012-05-11 | 2013-11-14 | Five Prime Therapeutics, Inc. | Methods of treating conditions with antibodies that bind colony stimulating factor 1 receptor (csf1r) |
| WO2014008218A1 (en) | 2012-07-02 | 2014-01-09 | Bristol-Myers Squibb Company | Optimization of antibodies that bind lymphocyte activation gene-3 (lag-3), and uses thereof |
| WO2014036357A1 (en) | 2012-08-31 | 2014-03-06 | Five Prime Therapeutics, Inc. | Methods of treating conditions with antibodies that bind colony stimulating factor 1 receptor (csf1r) |
| WO2023148643A1 (en) * | 2022-02-03 | 2023-08-10 | Aurigene Oncology Limited | Fused bicyclic heterocyclyl compounds as usp1 inhibitors |
| WO2024006879A1 (en) * | 2022-06-29 | 2024-01-04 | Zentaur Therapeutics Usa Inc. | Usp1 inhibitors and uses thereof |
| WO2024208292A1 (en) * | 2023-04-04 | 2024-10-10 | 江苏亚虹医药科技股份有限公司 | Ubiquitin-specific protease 1 inhibitor, preparation method therefor, and medical use thereof |
| WO2024233605A1 (en) * | 2023-05-08 | 2024-11-14 | Tango Therapeutics, Inc. | Compounds and their use against cancer |
Non-Patent Citations (4)
| Title |
|---|
| "Remington's Pharmaceutical Sciences", 1990, MACK PUBLISHING COMPANY |
| GREENEWUTS: "Protective Groups In Organic Synthesis", 1999, WILEY AND SONS |
| RAUTIO, J ET AL., NATURE REVIEW DRUG DISCOVERY, vol. 17, 2018, pages 559 - 587 |
| REMINGTON'S PHARMACEUTICAL SCIENCES, 1985 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TWI824405B (en) | Tetracyclic oxazepine compounds and uses thereof | |
| US20240239788A1 (en) | Small molecule inhibitors of kras g12d mutant | |
| JP6165977B2 (en) | Heteroarylpyridone and aza-pyridone amide compounds | |
| EP3154958B1 (en) | Phosphatidylinositol 3-kinase inhibitors | |
| AU2015274696B2 (en) | Phosphatidylinositol 3-kinase inhibitors | |
| TW202202504A (en) | Cd73 compounds | |
| EP3154961B1 (en) | Quinazolinone derivatives as phosphatidylinositol 3-kinase inhibitors | |
| TW201737913A (en) | TANK-binding kinase inhibitor compounds | |
| CN112823005A (en) | Pyrrolopyrimidine ITK inhibitors | |
| EP3154962B1 (en) | Phosphatidylinositol 3-kinase inhibitors | |
| EP4137489A1 (en) | Heterocyclic compounds as immunomodulators | |
| TW200526226A (en) | Pyrrolotriazine compounds as kinase inhibitors | |
| CN106660994A (en) | Phosphatidylinositol 3-kinase inhibitors | |
| WO2018156901A1 (en) | Inhibitors of bruton's tyrosine kinase | |
| JP7049519B2 (en) | Therapeutic heterocyclic compounds | |
| JP2021529210A (en) | Therapeutic heterocyclic compounds | |
| JP2022505872A (en) | 5-Azindazole derivative as an adenosine receptor antagonist | |
| KR102779693B1 (en) | MLL1 inhibitors and anticancer agents | |
| WO2025076284A1 (en) | Compounds, pharmaceutical compositions thereof, and methods of using the same | |
| WO2025096494A1 (en) | Ubiquitin specific processing protease 1 (usp1) compounds | |
| WO2025096539A1 (en) | Ubiquitin specific processing protease 1 (usp1) compounds | |
| WO2025096488A1 (en) | Ubiquitin specific processing protease 1 (usp1) compounds | |
| WO2025096489A1 (en) | Ubiquitin specific processing protease 1 (usp1) compounds | |
| WO2025096487A1 (en) | Ubiquitin specific processing protease 1 (usp1) compounds | |
| WO2025096490A1 (en) | Ubiquitin specific processing protease 1 (usp1) compounds |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24805356 Country of ref document: EP Kind code of ref document: A1 |