KR20090021169A - R(+) 및 s(-) 프라미펙솔을 포함하는 조성물 및 이의 사용 방법 - Google Patents
R(+) 및 s(-) 프라미펙솔을 포함하는 조성물 및 이의 사용 방법 Download PDFInfo
- Publication number
- KR20090021169A KR20090021169A KR1020087030672A KR20087030672A KR20090021169A KR 20090021169 A KR20090021169 A KR 20090021169A KR 1020087030672 A KR1020087030672 A KR 1020087030672A KR 20087030672 A KR20087030672 A KR 20087030672A KR 20090021169 A KR20090021169 A KR 20090021169A
- Authority
- KR
- South Korea
- Prior art keywords
- pramipexole
- pharmaceutical composition
- component
- sustained release
- enantiomer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- FASDKYOPVNHBLU-ZETCQYMHSA-N pramipexole Chemical compound C1[C@@H](NCCC)CCC2=C1SC(N)=N2 FASDKYOPVNHBLU-ZETCQYMHSA-N 0.000 title claims abstract description 275
- 239000000203 mixture Substances 0.000 title claims abstract description 159
- 238000000034 method Methods 0.000 title claims abstract description 70
- FASDKYOPVNHBLU-SSDOTTSWSA-N dexpramipexole Chemical compound C1[C@H](NCCC)CCC2=C1SC(N)=N2 FASDKYOPVNHBLU-SSDOTTSWSA-N 0.000 claims abstract description 167
- 208000018737 Parkinson disease Diseases 0.000 claims abstract description 18
- 239000008194 pharmaceutical composition Substances 0.000 claims description 133
- 229960003089 pramipexole Drugs 0.000 claims description 94
- 238000013268 sustained release Methods 0.000 claims description 67
- 239000012730 sustained-release form Substances 0.000 claims description 67
- 239000003826 tablet Substances 0.000 claims description 59
- 239000002775 capsule Substances 0.000 claims description 45
- 238000009472 formulation Methods 0.000 claims description 38
- 230000000694 effects Effects 0.000 claims description 29
- 229910052717 sulfur Inorganic materials 0.000 claims description 21
- 239000000243 solution Substances 0.000 claims description 17
- 239000012729 immediate-release (IR) formulation Substances 0.000 claims description 15
- 208000011580 syndromic disease Diseases 0.000 claims description 14
- 239000011159 matrix material Substances 0.000 claims description 12
- 238000007911 parenteral administration Methods 0.000 claims description 9
- 238000007918 intramuscular administration Methods 0.000 claims description 6
- 239000007788 liquid Substances 0.000 claims description 5
- 238000007920 subcutaneous administration Methods 0.000 claims description 3
- 238000010254 subcutaneous injection Methods 0.000 claims 1
- 239000007929 subcutaneous injection Substances 0.000 claims 1
- 238000011200 topical administration Methods 0.000 claims 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 48
- 150000001875 compounds Chemical class 0.000 description 38
- 231100000062 no-observed-adverse-effect level Toxicity 0.000 description 36
- 150000003839 salts Chemical class 0.000 description 33
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 28
- -1 for example Chemical compound 0.000 description 28
- 241000282414 Homo sapiens Species 0.000 description 19
- 239000004480 active ingredient Substances 0.000 description 19
- 201000010099 disease Diseases 0.000 description 19
- 239000007787 solid Substances 0.000 description 15
- 230000001225 therapeutic effect Effects 0.000 description 14
- 241000282472 Canis lupus familiaris Species 0.000 description 13
- 102000015554 Dopamine receptor Human genes 0.000 description 13
- 108050004812 Dopamine receptor Proteins 0.000 description 13
- 238000006243 chemical reaction Methods 0.000 description 13
- 239000003814 drug Substances 0.000 description 13
- 238000000576 coating method Methods 0.000 description 12
- 239000000463 material Substances 0.000 description 12
- 239000003937 drug carrier Substances 0.000 description 11
- 102000005962 receptors Human genes 0.000 description 10
- 108020003175 receptors Proteins 0.000 description 10
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 9
- 208000035475 disorder Diseases 0.000 description 9
- 229940079593 drug Drugs 0.000 description 9
- 239000010410 layer Substances 0.000 description 9
- 239000000843 powder Substances 0.000 description 9
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 8
- 239000011248 coating agent Substances 0.000 description 8
- 239000000126 substance Substances 0.000 description 8
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 8
- 241001465754 Metazoa Species 0.000 description 7
- 238000002474 experimental method Methods 0.000 description 7
- 229920000159 gelatin Polymers 0.000 description 7
- 235000019322 gelatine Nutrition 0.000 description 7
- 229940101972 mirapex Drugs 0.000 description 7
- 230000000324 neuroprotective effect Effects 0.000 description 7
- 230000003287 optical effect Effects 0.000 description 7
- 239000003960 organic solvent Substances 0.000 description 7
- 239000000546 pharmaceutical excipient Substances 0.000 description 7
- 239000000375 suspending agent Substances 0.000 description 7
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 6
- 108010010803 Gelatin Proteins 0.000 description 6
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 6
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 6
- 239000002253 acid Substances 0.000 description 6
- 239000007894 caplet Substances 0.000 description 6
- 239000002270 dispersing agent Substances 0.000 description 6
- VYFYYTLLBUKUHU-UHFFFAOYSA-N dopamine Chemical compound NCCC1=CC=C(O)C(O)=C1 VYFYYTLLBUKUHU-UHFFFAOYSA-N 0.000 description 6
- 239000003136 dopamine receptor stimulating agent Substances 0.000 description 6
- 239000002552 dosage form Substances 0.000 description 6
- 239000000839 emulsion Substances 0.000 description 6
- 150000002148 esters Chemical class 0.000 description 6
- 235000019441 ethanol Nutrition 0.000 description 6
- 239000008273 gelatin Substances 0.000 description 6
- 229940014259 gelatin Drugs 0.000 description 6
- 235000011852 gelatine desserts Nutrition 0.000 description 6
- 239000008187 granular material Substances 0.000 description 6
- 238000004128 high performance liquid chromatography Methods 0.000 description 6
- 238000002347 injection Methods 0.000 description 6
- 239000007924 injection Substances 0.000 description 6
- 210000003470 mitochondria Anatomy 0.000 description 6
- 239000003921 oil Substances 0.000 description 6
- 235000019198 oils Nutrition 0.000 description 6
- 208000024891 symptom Diseases 0.000 description 6
- 229920002472 Starch Polymers 0.000 description 5
- 230000008901 benefit Effects 0.000 description 5
- 239000007859 condensation product Substances 0.000 description 5
- 150000004985 diamines Chemical class 0.000 description 5
- 235000014113 dietary fatty acids Nutrition 0.000 description 5
- 239000003085 diluting agent Substances 0.000 description 5
- 239000000194 fatty acid Substances 0.000 description 5
- 229930195729 fatty acid Natural products 0.000 description 5
- 150000004665 fatty acids Chemical class 0.000 description 5
- 239000000796 flavoring agent Substances 0.000 description 5
- 238000010348 incorporation Methods 0.000 description 5
- 239000003112 inhibitor Substances 0.000 description 5
- 230000007170 pathology Effects 0.000 description 5
- 239000008188 pellet Substances 0.000 description 5
- 229920001223 polyethylene glycol Polymers 0.000 description 5
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 5
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 5
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 5
- 239000003755 preservative agent Substances 0.000 description 5
- 230000002265 prevention Effects 0.000 description 5
- 239000000047 product Substances 0.000 description 5
- 239000000725 suspension Substances 0.000 description 5
- 239000003765 sweetening agent Substances 0.000 description 5
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 4
- VTYYLEPIZMXCLO-UHFFFAOYSA-L Calcium carbonate Chemical compound [Ca+2].[O-]C([O-])=O VTYYLEPIZMXCLO-UHFFFAOYSA-L 0.000 description 4
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 4
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 4
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 4
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 4
- JGFZNNIVVJXRND-UHFFFAOYSA-N N,N-Diisopropylethylamine (DIPEA) Chemical compound CCN(C(C)C)C(C)C JGFZNNIVVJXRND-UHFFFAOYSA-N 0.000 description 4
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 4
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 4
- 229930006000 Sucrose Natural products 0.000 description 4
- 229920001615 Tragacanth Polymers 0.000 description 4
- 230000003078 antioxidant effect Effects 0.000 description 4
- 239000001768 carboxy methyl cellulose Substances 0.000 description 4
- 230000000875 corresponding effect Effects 0.000 description 4
- 238000004090 dissolution Methods 0.000 description 4
- 239000008298 dragée Substances 0.000 description 4
- 239000000945 filler Substances 0.000 description 4
- 235000003599 food sweetener Nutrition 0.000 description 4
- 239000000499 gel Substances 0.000 description 4
- 238000001727 in vivo Methods 0.000 description 4
- 239000008101 lactose Substances 0.000 description 4
- 229940057995 liquid paraffin Drugs 0.000 description 4
- 239000000314 lubricant Substances 0.000 description 4
- 230000002438 mitochondrial effect Effects 0.000 description 4
- 230000004770 neurodegeneration Effects 0.000 description 4
- 208000015122 neurodegenerative disease Diseases 0.000 description 4
- 239000004090 neuroprotective agent Substances 0.000 description 4
- 239000004006 olive oil Substances 0.000 description 4
- 235000008390 olive oil Nutrition 0.000 description 4
- 230000036542 oxidative stress Effects 0.000 description 4
- 239000002244 precipitate Substances 0.000 description 4
- 238000000746 purification Methods 0.000 description 4
- 235000010356 sorbitol Nutrition 0.000 description 4
- 239000000600 sorbitol Substances 0.000 description 4
- 235000019698 starch Nutrition 0.000 description 4
- 239000005720 sucrose Substances 0.000 description 4
- 235000000346 sugar Nutrition 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- LNAZSHAWQACDHT-XIYTZBAFSA-N (2r,3r,4s,5r,6s)-4,5-dimethoxy-2-(methoxymethyl)-3-[(2s,3r,4s,5r,6r)-3,4,5-trimethoxy-6-(methoxymethyl)oxan-2-yl]oxy-6-[(2r,3r,4s,5r,6r)-4,5,6-trimethoxy-2-(methoxymethyl)oxan-3-yl]oxyoxane Chemical compound CO[C@@H]1[C@@H](OC)[C@H](OC)[C@@H](COC)O[C@H]1O[C@H]1[C@H](OC)[C@@H](OC)[C@H](O[C@H]2[C@@H]([C@@H](OC)[C@H](OC)O[C@@H]2COC)OC)O[C@@H]1COC LNAZSHAWQACDHT-XIYTZBAFSA-N 0.000 description 3
- ZORQXIQZAOLNGE-UHFFFAOYSA-N 1,1-difluorocyclohexane Chemical compound FC1(F)CCCCC1 ZORQXIQZAOLNGE-UHFFFAOYSA-N 0.000 description 3
- 241000282693 Cercopithecidae Species 0.000 description 3
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 3
- 229920000084 Gum arabic Polymers 0.000 description 3
- 239000002202 Polyethylene glycol Substances 0.000 description 3
- 235000010489 acacia gum Nutrition 0.000 description 3
- 239000000205 acacia gum Substances 0.000 description 3
- DPXJVFZANSGRMM-UHFFFAOYSA-N acetic acid;2,3,4,5,6-pentahydroxyhexanal;sodium Chemical compound [Na].CC(O)=O.OCC(O)C(O)C(O)C(O)C=O DPXJVFZANSGRMM-UHFFFAOYSA-N 0.000 description 3
- 206010002026 amyotrophic lateral sclerosis Diseases 0.000 description 3
- 239000003963 antioxidant agent Substances 0.000 description 3
- 235000006708 antioxidants Nutrition 0.000 description 3
- 239000000305 astragalus gummifer gum Substances 0.000 description 3
- 239000011324 bead Substances 0.000 description 3
- 239000011230 binding agent Substances 0.000 description 3
- 210000004027 cell Anatomy 0.000 description 3
- 235000010980 cellulose Nutrition 0.000 description 3
- 239000001913 cellulose Substances 0.000 description 3
- 239000007795 chemical reaction product Substances 0.000 description 3
- 239000003795 chemical substances by application Substances 0.000 description 3
- 239000003086 colorant Substances 0.000 description 3
- 238000013270 controlled release Methods 0.000 description 3
- 229960003638 dopamine Drugs 0.000 description 3
- 238000009505 enteric coating Methods 0.000 description 3
- 239000002702 enteric coating Substances 0.000 description 3
- 238000011156 evaluation Methods 0.000 description 3
- 235000013355 food flavoring agent Nutrition 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- 235000011187 glycerol Nutrition 0.000 description 3
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 3
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 3
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 3
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 239000003446 ligand Substances 0.000 description 3
- 230000000670 limiting effect Effects 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 231100000682 maximum tolerated dose Toxicity 0.000 description 3
- 229940126601 medicinal product Drugs 0.000 description 3
- 229920000609 methyl cellulose Polymers 0.000 description 3
- 235000010981 methylcellulose Nutrition 0.000 description 3
- 239000001923 methylcellulose Substances 0.000 description 3
- 210000002569 neuron Anatomy 0.000 description 3
- 239000008203 oral pharmaceutical composition Substances 0.000 description 3
- 239000001301 oxygen Substances 0.000 description 3
- 229910052760 oxygen Inorganic materials 0.000 description 3
- 230000036961 partial effect Effects 0.000 description 3
- 230000000144 pharmacologic effect Effects 0.000 description 3
- 229920003023 plastic Polymers 0.000 description 3
- 239000004033 plastic Substances 0.000 description 3
- 229920000642 polymer Polymers 0.000 description 3
- 238000001556 precipitation Methods 0.000 description 3
- 238000002360 preparation method Methods 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- KCXFHTAICRTXLI-UHFFFAOYSA-N propane-1-sulfonic acid Chemical compound CCCS(O)(=O)=O KCXFHTAICRTXLI-UHFFFAOYSA-N 0.000 description 3
- 239000000376 reactant Substances 0.000 description 3
- 239000011541 reaction mixture Substances 0.000 description 3
- 230000004044 response Effects 0.000 description 3
- 235000019812 sodium carboxymethyl cellulose Nutrition 0.000 description 3
- 229920001027 sodium carboxymethylcellulose Polymers 0.000 description 3
- 239000002904 solvent Substances 0.000 description 3
- 235000011069 sorbitan monooleate Nutrition 0.000 description 3
- 239000001593 sorbitan monooleate Substances 0.000 description 3
- 229940035049 sorbitan monooleate Drugs 0.000 description 3
- 239000003381 stabilizer Substances 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 150000008163 sugars Chemical class 0.000 description 3
- 239000000829 suppository Substances 0.000 description 3
- 239000000454 talc Substances 0.000 description 3
- 229910052623 talc Inorganic materials 0.000 description 3
- 235000012222 talc Nutrition 0.000 description 3
- 238000012360 testing method Methods 0.000 description 3
- 231100000419 toxicity Toxicity 0.000 description 3
- 230000001988 toxicity Effects 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- 239000000080 wetting agent Substances 0.000 description 3
- IXPNQXFRVYWDDI-UHFFFAOYSA-N 1-methyl-2,4-dioxo-1,3-diazinane-5-carboximidamide Chemical compound CN1CC(C(N)=N)C(=O)NC1=O IXPNQXFRVYWDDI-UHFFFAOYSA-N 0.000 description 2
- VBICKXHEKHSIBG-UHFFFAOYSA-N 1-monostearoylglycerol Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)CO VBICKXHEKHSIBG-UHFFFAOYSA-N 0.000 description 2
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 2
- IZHVBANLECCAGF-UHFFFAOYSA-N 2-hydroxy-3-(octadecanoyloxy)propyl octadecanoate Chemical compound CCCCCCCCCCCCCCCCCC(=O)OCC(O)COC(=O)CCCCCCCCCCCCCCCCC IZHVBANLECCAGF-UHFFFAOYSA-N 0.000 description 2
- 229920001817 Agar Polymers 0.000 description 2
- 208000024827 Alzheimer disease Diseases 0.000 description 2
- 235000003911 Arachis Nutrition 0.000 description 2
- 244000105624 Arachis hypogaea Species 0.000 description 2
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 2
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- 229920002261 Corn starch Polymers 0.000 description 2
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 2
- 208000012661 Dyskinesia Diseases 0.000 description 2
- 208000000059 Dyspnea Diseases 0.000 description 2
- 206010013975 Dyspnoeas Diseases 0.000 description 2
- 239000001856 Ethyl cellulose Substances 0.000 description 2
- ZZSNKZQZMQGXPY-UHFFFAOYSA-N Ethyl cellulose Chemical compound CCOCC1OC(OC)C(OCC)C(OCC)C1OC1C(O)C(O)C(OC)C(CO)O1 ZZSNKZQZMQGXPY-UHFFFAOYSA-N 0.000 description 2
- 241000282412 Homo Species 0.000 description 2
- 206010020751 Hypersensitivity Diseases 0.000 description 2
- 208000001953 Hypotension Diseases 0.000 description 2
- 241000124008 Mammalia Species 0.000 description 2
- 229930195725 Mannitol Natural products 0.000 description 2
- BZLVMXJERCGZMT-UHFFFAOYSA-N Methyl tert-butyl ether Chemical compound COC(C)(C)C BZLVMXJERCGZMT-UHFFFAOYSA-N 0.000 description 2
- IMNFDUFMRHMDMM-UHFFFAOYSA-N N-Heptane Chemical compound CCCCCCC IMNFDUFMRHMDMM-UHFFFAOYSA-N 0.000 description 2
- 235000019483 Peanut oil Nutrition 0.000 description 2
- 239000004698 Polyethylene Substances 0.000 description 2
- RJKFOVLPORLFTN-LEKSSAKUSA-N Progesterone Chemical compound C1CC2=CC(=O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H](C(=O)C)[C@@]1(C)CC2 RJKFOVLPORLFTN-LEKSSAKUSA-N 0.000 description 2
- 241000283984 Rodentia Species 0.000 description 2
- 206010041349 Somnolence Diseases 0.000 description 2
- GWEVSGVZZGPLCZ-UHFFFAOYSA-N Titan oxide Chemical compound O=[Ti]=O GWEVSGVZZGPLCZ-UHFFFAOYSA-N 0.000 description 2
- 206010044565 Tremor Diseases 0.000 description 2
- 206010047700 Vomiting Diseases 0.000 description 2
- 230000009471 action Effects 0.000 description 2
- 239000013543 active substance Substances 0.000 description 2
- 239000002671 adjuvant Substances 0.000 description 2
- 230000002411 adverse Effects 0.000 description 2
- 239000000443 aerosol Substances 0.000 description 2
- 239000008272 agar Substances 0.000 description 2
- 235000010419 agar Nutrition 0.000 description 2
- 239000000556 agonist Substances 0.000 description 2
- 235000010443 alginic acid Nutrition 0.000 description 2
- 229920000615 alginic acid Polymers 0.000 description 2
- 239000000783 alginic acid Substances 0.000 description 2
- 229960001126 alginic acid Drugs 0.000 description 2
- 150000004781 alginic acids Chemical class 0.000 description 2
- 239000005557 antagonist Substances 0.000 description 2
- 239000012736 aqueous medium Substances 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 229910000019 calcium carbonate Inorganic materials 0.000 description 2
- 239000001506 calcium phosphate Substances 0.000 description 2
- 229910000389 calcium phosphate Inorganic materials 0.000 description 2
- 235000011010 calcium phosphates Nutrition 0.000 description 2
- 239000000969 carrier Substances 0.000 description 2
- 150000001768 cations Chemical class 0.000 description 2
- 229920002678 cellulose Polymers 0.000 description 2
- 239000003153 chemical reaction reagent Substances 0.000 description 2
- 229940110456 cocoa butter Drugs 0.000 description 2
- 235000019868 cocoa butter Nutrition 0.000 description 2
- 238000007906 compression Methods 0.000 description 2
- 230000006835 compression Effects 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- 235000008504 concentrate Nutrition 0.000 description 2
- 238000001816 cooling Methods 0.000 description 2
- 239000008120 corn starch Substances 0.000 description 2
- 239000006071 cream Substances 0.000 description 2
- 230000003247 decreasing effect Effects 0.000 description 2
- 230000003111 delayed effect Effects 0.000 description 2
- 238000001784 detoxification Methods 0.000 description 2
- 208000002173 dizziness Diseases 0.000 description 2
- 238000012377 drug delivery Methods 0.000 description 2
- 239000013583 drug formulation Substances 0.000 description 2
- 239000003995 emulsifying agent Substances 0.000 description 2
- 235000019325 ethyl cellulose Nutrition 0.000 description 2
- 229920001249 ethyl cellulose Polymers 0.000 description 2
- MMXKVMNBHPAILY-UHFFFAOYSA-N ethyl laurate Chemical compound CCCCCCCCCCCC(=O)OCC MMXKVMNBHPAILY-UHFFFAOYSA-N 0.000 description 2
- 230000001747 exhibiting effect Effects 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 239000012530 fluid Substances 0.000 description 2
- 238000000227 grinding Methods 0.000 description 2
- BXWNKGSJHAJOGX-UHFFFAOYSA-N hexadecan-1-ol Chemical compound CCCCCCCCCCCCCCCCO BXWNKGSJHAJOGX-UHFFFAOYSA-N 0.000 description 2
- 230000002209 hydrophobic effect Effects 0.000 description 2
- 230000036543 hypotension Effects 0.000 description 2
- 239000007943 implant Substances 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 238000000338 in vitro Methods 0.000 description 2
- 238000001802 infusion Methods 0.000 description 2
- 230000003993 interaction Effects 0.000 description 2
- 238000007912 intraperitoneal administration Methods 0.000 description 2
- 238000001990 intravenous administration Methods 0.000 description 2
- 238000011835 investigation Methods 0.000 description 2
- 235000010445 lecithin Nutrition 0.000 description 2
- 239000000787 lecithin Substances 0.000 description 2
- 229940067606 lecithin Drugs 0.000 description 2
- 230000003859 lipid peroxidation Effects 0.000 description 2
- 239000007937 lozenge Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 235000010355 mannitol Nutrition 0.000 description 2
- 239000000594 mannitol Substances 0.000 description 2
- 230000007246 mechanism Effects 0.000 description 2
- 239000002480 mineral oil Substances 0.000 description 2
- 230000006676 mitochondrial damage Effects 0.000 description 2
- 230000004065 mitochondrial dysfunction Effects 0.000 description 2
- 230000004898 mitochondrial function Effects 0.000 description 2
- 239000001788 mono and diglycerides of fatty acids Substances 0.000 description 2
- 238000000465 moulding Methods 0.000 description 2
- 230000016273 neuron death Effects 0.000 description 2
- 231100000252 nontoxic Toxicity 0.000 description 2
- 230000003000 nontoxic effect Effects 0.000 description 2
- 238000010606 normalization Methods 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 239000000312 peanut oil Substances 0.000 description 2
- AQSJGOWTSHOLKH-UHFFFAOYSA-N phosphite(3-) Chemical class [O-]P([O-])[O-] AQSJGOWTSHOLKH-UHFFFAOYSA-N 0.000 description 2
- 239000006187 pill Substances 0.000 description 2
- 229920000573 polyethylene Polymers 0.000 description 2
- 229920001592 potato starch Polymers 0.000 description 2
- 230000003389 potentiating effect Effects 0.000 description 2
- 230000002335 preservative effect Effects 0.000 description 2
- 238000003825 pressing Methods 0.000 description 2
- 239000011241 protective layer Substances 0.000 description 2
- 230000002829 reductive effect Effects 0.000 description 2
- 239000008159 sesame oil Substances 0.000 description 2
- 235000011803 sesame oil Nutrition 0.000 description 2
- 235000010413 sodium alginate Nutrition 0.000 description 2
- 239000000661 sodium alginate Substances 0.000 description 2
- 229940005550 sodium alginate Drugs 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 210000000278 spinal cord Anatomy 0.000 description 2
- 239000008107 starch Substances 0.000 description 2
- 239000004094 surface-active agent Substances 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 230000002194 synthesizing effect Effects 0.000 description 2
- 239000006188 syrup Substances 0.000 description 2
- 235000020357 syrup Nutrition 0.000 description 2
- 239000002562 thickening agent Substances 0.000 description 2
- 230000000699 topical effect Effects 0.000 description 2
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 2
- 238000001291 vacuum drying Methods 0.000 description 2
- 235000015112 vegetable and seed oil Nutrition 0.000 description 2
- 239000008158 vegetable oil Substances 0.000 description 2
- 230000008673 vomiting Effects 0.000 description 2
- GHOKWGTUZJEAQD-ZETCQYMHSA-N (D)-(+)-Pantothenic acid Chemical compound OCC(C)(C)[C@@H](O)C(=O)NCCC(O)=O GHOKWGTUZJEAQD-ZETCQYMHSA-N 0.000 description 1
- DDMOUSALMHHKOS-UHFFFAOYSA-N 1,2-dichloro-1,1,2,2-tetrafluoroethane Chemical compound FC(F)(Cl)C(F)(F)Cl DDMOUSALMHHKOS-UHFFFAOYSA-N 0.000 description 1
- 238000001644 13C nuclear magnetic resonance spectroscopy Methods 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- ALUQMCBDQKDRAK-UHFFFAOYSA-N 2,3,3a,4-tetrahydro-1,3-benzothiazole Chemical compound C1C=CC=C2SCNC21 ALUQMCBDQKDRAK-UHFFFAOYSA-N 0.000 description 1
- UHGULLIUJBCTEF-UHFFFAOYSA-N 2-aminobenzothiazole Chemical class C1=CC=C2SC(N)=NC2=C1 UHGULLIUJBCTEF-UHFFFAOYSA-N 0.000 description 1
- HZLCGUXUOFWCCN-UHFFFAOYSA-N 2-hydroxynonadecane-1,2,3-tricarboxylic acid Chemical compound CCCCCCCCCCCCCCCCC(C(O)=O)C(O)(C(O)=O)CC(O)=O HZLCGUXUOFWCCN-UHFFFAOYSA-N 0.000 description 1
- FJKROLUGYXJWQN-UHFFFAOYSA-M 4-hydroxybenzoate Chemical compound OC1=CC=C(C([O-])=O)C=C1 FJKROLUGYXJWQN-UHFFFAOYSA-M 0.000 description 1
- 208000004998 Abdominal Pain Diseases 0.000 description 1
- 244000215068 Acacia senegal Species 0.000 description 1
- 239000005995 Aluminium silicate Substances 0.000 description 1
- 208000019901 Anxiety disease Diseases 0.000 description 1
- 241000416162 Astragalus gummifer Species 0.000 description 1
- 208000008035 Back Pain Diseases 0.000 description 1
- 229920002134 Carboxymethyl cellulose Polymers 0.000 description 1
- 229920000623 Cellulose acetate phthalate Polymers 0.000 description 1
- 206010008479 Chest Pain Diseases 0.000 description 1
- 206010010741 Conjunctivitis Diseases 0.000 description 1
- 206010010774 Constipation Diseases 0.000 description 1
- 206010011224 Cough Diseases 0.000 description 1
- 241000699802 Cricetulus griseus Species 0.000 description 1
- RGHNJXZEOKUKBD-SQOUGZDYSA-M D-gluconate Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@@H](O)C([O-])=O RGHNJXZEOKUKBD-SQOUGZDYSA-M 0.000 description 1
- FEWJPZIEWOKRBE-JCYAYHJZSA-N Dextrotartaric acid Chemical compound OC(=O)[C@H](O)[C@@H](O)C(O)=O FEWJPZIEWOKRBE-JCYAYHJZSA-N 0.000 description 1
- 206010012735 Diarrhoea Diseases 0.000 description 1
- 239000004338 Dichlorodifluoromethane Substances 0.000 description 1
- 208000003164 Diplopia Diseases 0.000 description 1
- 208000014094 Dystonic disease Diseases 0.000 description 1
- LVGKNOAMLMIIKO-UHFFFAOYSA-N Elaidinsaeure-aethylester Natural products CCCCCCCCC=CCCCCCCCC(=O)OCC LVGKNOAMLMIIKO-UHFFFAOYSA-N 0.000 description 1
- 241000792859 Enema Species 0.000 description 1
- 208000010201 Exanthema Diseases 0.000 description 1
- 208000027776 Extrapyramidal disease Diseases 0.000 description 1
- KRHYYFGTRYWZRS-UHFFFAOYSA-N Fluorane Chemical compound F KRHYYFGTRYWZRS-UHFFFAOYSA-N 0.000 description 1
- 238000005033 Fourier transform infrared spectroscopy Methods 0.000 description 1
- 206010017577 Gait disturbance Diseases 0.000 description 1
- 239000001828 Gelatine Substances 0.000 description 1
- 206010060891 General symptom Diseases 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 244000068988 Glycine max Species 0.000 description 1
- 235000010469 Glycine max Nutrition 0.000 description 1
- 208000004547 Hallucinations Diseases 0.000 description 1
- 206010019233 Headaches Diseases 0.000 description 1
- CPELXLSAUQHCOX-UHFFFAOYSA-N Hydrogen bromide Chemical compound Br CPELXLSAUQHCOX-UHFFFAOYSA-N 0.000 description 1
- 229920001479 Hydroxyethyl methyl cellulose Polymers 0.000 description 1
- 229920002153 Hydroxypropyl cellulose Polymers 0.000 description 1
- 208000008454 Hyperhidrosis Diseases 0.000 description 1
- 206010020772 Hypertension Diseases 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 description 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-M Lactate Chemical compound CC(O)C([O-])=O JVTAAEKCZFNVCJ-UHFFFAOYSA-M 0.000 description 1
- 239000000232 Lipid Bilayer Substances 0.000 description 1
- AFVFQIVMOAPDHO-UHFFFAOYSA-N Methanesulfonic acid Chemical class CS(O)(=O)=O AFVFQIVMOAPDHO-UHFFFAOYSA-N 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- 206010061296 Motor dysfunction Diseases 0.000 description 1
- 208000007101 Muscle Cramp Diseases 0.000 description 1
- 206010052904 Musculoskeletal stiffness Diseases 0.000 description 1
- 208000006550 Mydriasis Diseases 0.000 description 1
- GXCLVBGFBYZDAG-UHFFFAOYSA-N N-[2-(1H-indol-3-yl)ethyl]-N-methylprop-2-en-1-amine Chemical compound CN(CCC1=CNC2=C1C=CC=C2)CC=C GXCLVBGFBYZDAG-UHFFFAOYSA-N 0.000 description 1
- PQBAWAQIRZIWIV-UHFFFAOYSA-N N-methylpyridinium Chemical compound C[N+]1=CC=CC=C1 PQBAWAQIRZIWIV-UHFFFAOYSA-N 0.000 description 1
- FASDKYOPVNHBLU-UHFFFAOYSA-N N6-Propyl-4,5,6,7-tetrahydro-1,3-benzothiazole-2,6-diamine Chemical compound C1C(NCCC)CCC2=C1SC(N)=N2 FASDKYOPVNHBLU-UHFFFAOYSA-N 0.000 description 1
- 206010028813 Nausea Diseases 0.000 description 1
- 206010028836 Neck pain Diseases 0.000 description 1
- 206010029216 Nervousness Diseases 0.000 description 1
- 101710138657 Neurotoxin Proteins 0.000 description 1
- MUBZPKHOEPUJKR-UHFFFAOYSA-N Oxalic acid Chemical compound OC(=O)C(O)=O MUBZPKHOEPUJKR-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- 206010033799 Paralysis Diseases 0.000 description 1
- 206010034010 Parkinsonism Diseases 0.000 description 1
- 201000007100 Pharyngitis Diseases 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 229920003171 Poly (ethylene oxide) Polymers 0.000 description 1
- 241000288906 Primates Species 0.000 description 1
- 102000003946 Prolactin Human genes 0.000 description 1
- 108010057464 Prolactin Proteins 0.000 description 1
- 208000003251 Pruritus Diseases 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 208000005793 Restless legs syndrome Diseases 0.000 description 1
- 201000007737 Retinal degeneration Diseases 0.000 description 1
- 241000220010 Rhode Species 0.000 description 1
- GOTMKOSCLKVOGG-UHFFFAOYSA-N SCH 23390 Chemical compound C1N(C)CCC2=CC(Cl)=C(O)C=C2C1C1=CC=CC=C1 GOTMKOSCLKVOGG-UHFFFAOYSA-N 0.000 description 1
- 235000019485 Safflower oil Nutrition 0.000 description 1
- 208000032140 Sleepiness Diseases 0.000 description 1
- 229920002125 Sokalan® Polymers 0.000 description 1
- 206010050014 Sudden onset of sleep Diseases 0.000 description 1
- 208000001871 Tachycardia Diseases 0.000 description 1
- FEWJPZIEWOKRBE-UHFFFAOYSA-N Tartaric acid Natural products [H+].[H+].[O-]C(=O)C(O)C(O)C([O-])=O FEWJPZIEWOKRBE-UHFFFAOYSA-N 0.000 description 1
- 208000009205 Tinnitus Diseases 0.000 description 1
- 208000010641 Tooth disease Diseases 0.000 description 1
- 208000027418 Wounds and injury Diseases 0.000 description 1
- WERKSKAQRVDLDW-ANOHMWSOSA-N [(2s,3r,4r,5r)-2,3,4,5,6-pentahydroxyhexyl] (z)-octadec-9-enoate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO WERKSKAQRVDLDW-ANOHMWSOSA-N 0.000 description 1
- 230000005856 abnormality Effects 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 238000009825 accumulation Methods 0.000 description 1
- 150000001242 acetic acid derivatives Chemical class 0.000 description 1
- 239000012190 activator Substances 0.000 description 1
- 239000011149 active material Substances 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 230000000996 additive effect Effects 0.000 description 1
- 229940040563 agaric acid Drugs 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- WNROFYMDJYEPJX-UHFFFAOYSA-K aluminium hydroxide Chemical compound [OH-].[OH-].[OH-].[Al+3] WNROFYMDJYEPJX-UHFFFAOYSA-K 0.000 description 1
- 235000012211 aluminium silicate Nutrition 0.000 description 1
- 229940024606 amino acid Drugs 0.000 description 1
- 230000036506 anxiety Effects 0.000 description 1
- 239000007900 aqueous suspension Substances 0.000 description 1
- 229960005070 ascorbic acid Drugs 0.000 description 1
- 235000010323 ascorbic acid Nutrition 0.000 description 1
- 239000011668 ascorbic acid Substances 0.000 description 1
- 229940009098 aspartate Drugs 0.000 description 1
- 230000004888 barrier function Effects 0.000 description 1
- 235000013871 bee wax Nutrition 0.000 description 1
- 239000012166 beeswax Substances 0.000 description 1
- 230000003542 behavioural effect Effects 0.000 description 1
- 150000008107 benzenesulfonic acids Chemical class 0.000 description 1
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- 210000004556 brain Anatomy 0.000 description 1
- 210000004958 brain cell Anatomy 0.000 description 1
- DEGAKNSWVGKMLS-UHFFFAOYSA-N calcein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC(CN(CC(O)=O)CC(O)=O)=C(O)C=C1OC1=C2C=C(CN(CC(O)=O)CC(=O)O)C(O)=C1 DEGAKNSWVGKMLS-UHFFFAOYSA-N 0.000 description 1
- 235000010216 calcium carbonate Nutrition 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 229960004424 carbon dioxide Drugs 0.000 description 1
- 235000010948 carboxy methyl cellulose Nutrition 0.000 description 1
- 239000008112 carboxymethyl-cellulose Substances 0.000 description 1
- 230000030833 cell death Effects 0.000 description 1
- 230000006721 cell death pathway Effects 0.000 description 1
- 210000000170 cell membrane Anatomy 0.000 description 1
- 230000003833 cell viability Effects 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 229940081734 cellulose acetate phthalate Drugs 0.000 description 1
- 229920006184 cellulose methylcellulose Polymers 0.000 description 1
- 229960000541 cetyl alcohol Drugs 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- 238000002144 chemical decomposition reaction Methods 0.000 description 1
- 238000004296 chiral HPLC Methods 0.000 description 1
- 150000001860 citric acid derivatives Chemical class 0.000 description 1
- 239000003240 coconut oil Substances 0.000 description 1
- 235000019864 coconut oil Nutrition 0.000 description 1
- 239000007891 compressed tablet Substances 0.000 description 1
- 230000008602 contraction Effects 0.000 description 1
- 239000000599 controlled substance Substances 0.000 description 1
- 230000001276 controlling effect Effects 0.000 description 1
- 229920001577 copolymer Polymers 0.000 description 1
- 235000005687 corn oil Nutrition 0.000 description 1
- 239000002285 corn oil Substances 0.000 description 1
- 229940099112 cornstarch Drugs 0.000 description 1
- 230000002596 correlated effect Effects 0.000 description 1
- 235000012343 cottonseed oil Nutrition 0.000 description 1
- 239000002385 cottonseed oil Substances 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 230000034994 death Effects 0.000 description 1
- 230000007850 degeneration Effects 0.000 description 1
- 239000003405 delayed action preparation Substances 0.000 description 1
- 238000000151 deposition Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- PXBRQCKWGAHEHS-UHFFFAOYSA-N dichlorodifluoromethane Chemical compound FC(F)(Cl)Cl PXBRQCKWGAHEHS-UHFFFAOYSA-N 0.000 description 1
- 235000019404 dichlorodifluoromethane Nutrition 0.000 description 1
- 229940042935 dichlorodifluoromethane Drugs 0.000 description 1
- 229940087091 dichlorotetrafluoroethane Drugs 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 239000001177 diphosphate Substances 0.000 description 1
- XPPKVPWEQAFLFU-UHFFFAOYSA-J diphosphate(4-) Chemical compound [O-]P([O-])(=O)OP([O-])([O-])=O XPPKVPWEQAFLFU-UHFFFAOYSA-J 0.000 description 1
- 235000011180 diphosphates Nutrition 0.000 description 1
- 229940042399 direct acting antivirals protease inhibitors Drugs 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- VFNGKCDDZUSWLR-UHFFFAOYSA-L disulfate(2-) Chemical compound [O-]S(=O)(=O)OS([O-])(=O)=O VFNGKCDDZUSWLR-UHFFFAOYSA-L 0.000 description 1
- 239000000433 dopamine 1 receptor blocking agent Substances 0.000 description 1
- 229940052760 dopamine agonists Drugs 0.000 description 1
- 230000003291 dopaminomimetic effect Effects 0.000 description 1
- 206010013781 dry mouth Diseases 0.000 description 1
- 238000001035 drying Methods 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 201000006549 dyspepsia Diseases 0.000 description 1
- 208000010118 dystonia Diseases 0.000 description 1
- 230000002526 effect on cardiovascular system Effects 0.000 description 1
- 239000008393 encapsulating agent Substances 0.000 description 1
- 239000007920 enema Substances 0.000 description 1
- 229940095399 enema Drugs 0.000 description 1
- CCIVGXIOQKPBKL-UHFFFAOYSA-N ethanesulfonic acid Chemical class CCS(O)(=O)=O CCIVGXIOQKPBKL-UHFFFAOYSA-N 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- LVGKNOAMLMIIKO-QXMHVHEDSA-N ethyl oleate Chemical compound CCCCCCCC\C=C/CCCCCCCC(=O)OCC LVGKNOAMLMIIKO-QXMHVHEDSA-N 0.000 description 1
- 229940093471 ethyl oleate Drugs 0.000 description 1
- 201000005884 exanthem Diseases 0.000 description 1
- 208000019995 familial amyotrophic lateral sclerosis Diseases 0.000 description 1
- 239000007888 film coating Substances 0.000 description 1
- 238000009501 film coating Methods 0.000 description 1
- 239000006260 foam Substances 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 230000037406 food intake Effects 0.000 description 1
- VZCYOOQTPOCHFL-OWOJBTEDSA-L fumarate(2-) Chemical class [O-]C(=O)\C=C\C([O-])=O VZCYOOQTPOCHFL-OWOJBTEDSA-L 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 210000004051 gastric juice Anatomy 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 238000003304 gavage Methods 0.000 description 1
- 239000007903 gelatin capsule Substances 0.000 description 1
- 239000007897 gelcap Substances 0.000 description 1
- 229940050410 gluconate Drugs 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 229930195712 glutamate Natural products 0.000 description 1
- 125000005456 glyceride group Chemical group 0.000 description 1
- 229940074045 glyceryl distearate Drugs 0.000 description 1
- 229940075507 glyceryl monostearate Drugs 0.000 description 1
- 150000002334 glycols Chemical class 0.000 description 1
- 239000008202 granule composition Substances 0.000 description 1
- 150000004820 halides Chemical class 0.000 description 1
- 231100000869 headache Toxicity 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- FBPFZTCFMRRESA-UHFFFAOYSA-N hexane-1,2,3,4,5,6-hexol Chemical compound OCC(O)C(O)C(O)C(O)CO FBPFZTCFMRRESA-UHFFFAOYSA-N 0.000 description 1
- 239000003906 humectant Substances 0.000 description 1
- XMBWDFGMSWQBCA-UHFFFAOYSA-N hydrogen iodide Chemical compound I XMBWDFGMSWQBCA-UHFFFAOYSA-N 0.000 description 1
- 235000010977 hydroxypropyl cellulose Nutrition 0.000 description 1
- 239000001863 hydroxypropyl cellulose Substances 0.000 description 1
- 229920003132 hydroxypropyl methylcellulose phthalate Polymers 0.000 description 1
- 229940031704 hydroxypropyl methylcellulose phthalate Drugs 0.000 description 1
- 229920000639 hydroxypropylmethylcellulose acetate succinate Polymers 0.000 description 1
- 208000013403 hyperactivity Diseases 0.000 description 1
- 230000001771 impaired effect Effects 0.000 description 1
- 238000002513 implantation Methods 0.000 description 1
- 239000005414 inactive ingredient Substances 0.000 description 1
- 239000003701 inert diluent Substances 0.000 description 1
- 229940060367 inert ingredients Drugs 0.000 description 1
- 206010022000 influenza Diseases 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 239000007972 injectable composition Substances 0.000 description 1
- 208000014674 injury Diseases 0.000 description 1
- 230000000968 intestinal effect Effects 0.000 description 1
- 238000001361 intraarterial administration Methods 0.000 description 1
- 238000010255 intramuscular injection Methods 0.000 description 1
- 239000007927 intramuscular injection Substances 0.000 description 1
- 238000007914 intraventricular administration Methods 0.000 description 1
- 239000003456 ion exchange resin Substances 0.000 description 1
- 229920003303 ion-exchange polymer Polymers 0.000 description 1
- 230000007794 irritation Effects 0.000 description 1
- 230000007803 itching Effects 0.000 description 1
- 235000015110 jellies Nutrition 0.000 description 1
- 239000008274 jelly Substances 0.000 description 1
- NLYAJNPCOHFWQQ-UHFFFAOYSA-N kaolin Chemical compound O.O.O=[Al]O[Si](=O)O[Si](=O)O[Al]=O NLYAJNPCOHFWQQ-UHFFFAOYSA-N 0.000 description 1
- 210000003734 kidney Anatomy 0.000 description 1
- 239000004922 lacquer Substances 0.000 description 1
- 229940126707 lipid peroxidation inhibitor Drugs 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 238000009115 maintenance therapy Methods 0.000 description 1
- VZCYOOQTPOCHFL-UPHRSURJSA-N maleic acid Chemical compound OC(=O)\C=C/C(O)=O VZCYOOQTPOCHFL-UPHRSURJSA-N 0.000 description 1
- 150000002688 maleic acid derivatives Chemical class 0.000 description 1
- IWYDHOAUDWTVEP-UHFFFAOYSA-M mandelate Chemical compound [O-]C(=O)C(O)C1=CC=CC=C1 IWYDHOAUDWTVEP-UHFFFAOYSA-M 0.000 description 1
- 238000002483 medication Methods 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 239000000155 melt Substances 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 229920003145 methacrylic acid copolymer Polymers 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 230000027939 micturition Effects 0.000 description 1
- 235000010446 mineral oil Nutrition 0.000 description 1
- 244000309715 mini pig Species 0.000 description 1
- 210000001700 mitochondrial membrane Anatomy 0.000 description 1
- 238000002156 mixing Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 239000007932 molded tablet Substances 0.000 description 1
- 238000012544 monitoring process Methods 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- 208000016334 muscle symptom Diseases 0.000 description 1
- 210000004165 myocardium Anatomy 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 230000008693 nausea Effects 0.000 description 1
- 210000000944 nerve tissue Anatomy 0.000 description 1
- 230000001537 neural effect Effects 0.000 description 1
- 230000007658 neurological function Effects 0.000 description 1
- 239000002581 neurotoxin Substances 0.000 description 1
- 231100000618 neurotoxin Toxicity 0.000 description 1
- 239000002858 neurotransmitter agent Substances 0.000 description 1
- 150000002823 nitrates Chemical class 0.000 description 1
- 229910052757 nitrogen Inorganic materials 0.000 description 1
- 231100000706 no observed effect level Toxicity 0.000 description 1
- 238000010534 nucleophilic substitution reaction Methods 0.000 description 1
- 229960002378 oftasceine Drugs 0.000 description 1
- 239000002674 ointment Substances 0.000 description 1
- 239000008183 oral pharmaceutical preparation Substances 0.000 description 1
- 239000003791 organic solvent mixture Substances 0.000 description 1
- 210000001672 ovary Anatomy 0.000 description 1
- JOXIMZWYDAKGHI-UHFFFAOYSA-N p-toluenesulfonic acid Substances CC1=CC=C(S(O)(=O)=O)C=C1 JOXIMZWYDAKGHI-UHFFFAOYSA-N 0.000 description 1
- 229940014662 pantothenate Drugs 0.000 description 1
- 235000019161 pantothenic acid Nutrition 0.000 description 1
- 239000011713 pantothenic acid Substances 0.000 description 1
- 239000012188 paraffin wax Substances 0.000 description 1
- 239000006201 parenteral dosage form Substances 0.000 description 1
- 208000035824 paresthesia Diseases 0.000 description 1
- 239000006072 paste Substances 0.000 description 1
- 239000000137 peptide hydrolase inhibitor Substances 0.000 description 1
- VLTRZXGMWDSKGL-UHFFFAOYSA-N perchloric acid Chemical class OCl(=O)(=O)=O VLTRZXGMWDSKGL-UHFFFAOYSA-N 0.000 description 1
- 239000008177 pharmaceutical agent Substances 0.000 description 1
- 235000021317 phosphate Nutrition 0.000 description 1
- 239000008363 phosphate buffer Substances 0.000 description 1
- 150000003013 phosphoric acid derivatives Chemical class 0.000 description 1
- 229910052698 phosphorus Inorganic materials 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 238000006303 photolysis reaction Methods 0.000 description 1
- 230000015843 photosynthesis, light reaction Effects 0.000 description 1
- 239000000049 pigment Substances 0.000 description 1
- 239000011505 plaster Substances 0.000 description 1
- 239000004014 plasticizer Substances 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 229940100467 polyvinyl acetate phthalate Drugs 0.000 description 1
- 239000011148 porous material Substances 0.000 description 1
- 230000008092 positive effect Effects 0.000 description 1
- 229940116317 potato starch Drugs 0.000 description 1
- 229960002652 pramipexole dihydrochloride Drugs 0.000 description 1
- 239000002243 precursor Substances 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 239000000651 prodrug Substances 0.000 description 1
- 229940002612 prodrug Drugs 0.000 description 1
- 229960003387 progesterone Drugs 0.000 description 1
- 239000000186 progesterone Substances 0.000 description 1
- 229940097325 prolactin Drugs 0.000 description 1
- 239000003380 propellant Substances 0.000 description 1
- JTTWNTXHFYNETH-UHFFFAOYSA-N propyl 4-methylbenzenesulfonate Chemical compound CCCOS(=O)(=O)C1=CC=C(C)C=C1 JTTWNTXHFYNETH-UHFFFAOYSA-N 0.000 description 1
- 235000013772 propylene glycol Nutrition 0.000 description 1
- 239000003223 protective agent Substances 0.000 description 1
- 229940126409 proton pump inhibitor Drugs 0.000 description 1
- 239000000612 proton pump inhibitor Substances 0.000 description 1
- 230000005855 radiation Effects 0.000 description 1
- 239000002287 radioligand Substances 0.000 description 1
- 206010037844 rash Diseases 0.000 description 1
- 230000006950 reactive oxygen species formation Effects 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 239000006215 rectal suppository Substances 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 210000004994 reproductive system Anatomy 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 230000004258 retinal degeneration Effects 0.000 description 1
- 230000002207 retinal effect Effects 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 206010039083 rhinitis Diseases 0.000 description 1
- 229940100486 rice starch Drugs 0.000 description 1
- 230000000630 rising effect Effects 0.000 description 1
- CVHZOJJKTDOEJC-UHFFFAOYSA-N saccharin Chemical compound C1=CC=C2C(=O)NS(=O)(=O)C2=C1 CVHZOJJKTDOEJC-UHFFFAOYSA-N 0.000 description 1
- 229940081974 saccharin Drugs 0.000 description 1
- 235000019204 saccharin Nutrition 0.000 description 1
- 239000000901 saccharin and its Na,K and Ca salt Substances 0.000 description 1
- 235000005713 safflower oil Nutrition 0.000 description 1
- 239000003813 safflower oil Substances 0.000 description 1
- 210000003296 saliva Anatomy 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 238000000926 separation method Methods 0.000 description 1
- 231100000161 signs of toxicity Toxicity 0.000 description 1
- 201000009890 sinusitis Diseases 0.000 description 1
- 208000019116 sleep disease Diseases 0.000 description 1
- 208000022925 sleep disturbance Diseases 0.000 description 1
- 230000037321 sleepiness Effects 0.000 description 1
- 239000002002 slurry Substances 0.000 description 1
- 239000007901 soft capsule Substances 0.000 description 1
- 239000007909 solid dosage form Substances 0.000 description 1
- 239000012439 solid excipient Substances 0.000 description 1
- 239000003549 soybean oil Substances 0.000 description 1
- 235000012424 soybean oil Nutrition 0.000 description 1
- 241000894007 species Species 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 239000011550 stock solution Substances 0.000 description 1
- 210000002784 stomach Anatomy 0.000 description 1
- 230000035882 stress Effects 0.000 description 1
- 150000003890 succinate salts Chemical class 0.000 description 1
- 238000009495 sugar coating Methods 0.000 description 1
- 150000003871 sulfonates Chemical class 0.000 description 1
- 150000003467 sulfuric acid derivatives Chemical class 0.000 description 1
- 239000002511 suppository base Substances 0.000 description 1
- 230000035900 sweating Effects 0.000 description 1
- 230000009885 systemic effect Effects 0.000 description 1
- 239000007916 tablet composition Substances 0.000 description 1
- 230000006794 tachycardia Effects 0.000 description 1
- VQMNWIMYFHHFMC-UHFFFAOYSA-N tert-butyl 4-hydroxyindole-1-carboxylate Chemical compound C1=CC=C2N(C(=O)OC(C)(C)C)C=CC2=C1O VQMNWIMYFHHFMC-UHFFFAOYSA-N 0.000 description 1
- 238000011287 therapeutic dose Methods 0.000 description 1
- 239000010409 thin film Substances 0.000 description 1
- 231100000886 tinnitus Toxicity 0.000 description 1
- 239000004408 titanium dioxide Substances 0.000 description 1
- 239000006208 topical dosage form Substances 0.000 description 1
- 235000010487 tragacanth Nutrition 0.000 description 1
- 239000000196 tragacanth Substances 0.000 description 1
- 229940116362 tragacanth Drugs 0.000 description 1
- VZCYOOQTPOCHFL-UHFFFAOYSA-N trans-butenedioic acid Natural products OC(=O)C=CC(O)=O VZCYOOQTPOCHFL-UHFFFAOYSA-N 0.000 description 1
- 230000007704 transition Effects 0.000 description 1
- 230000005945 translocation Effects 0.000 description 1
- CYRMSUTZVYGINF-UHFFFAOYSA-N trichlorofluoromethane Chemical compound FC(Cl)(Cl)Cl CYRMSUTZVYGINF-UHFFFAOYSA-N 0.000 description 1
- 229940029284 trichlorofluoromethane Drugs 0.000 description 1
- ITMCEJHCFYSIIV-UHFFFAOYSA-N triflic acid Chemical compound OS(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-N 0.000 description 1
- 208000019206 urinary tract infection Diseases 0.000 description 1
- 239000000522 vaginal cream Substances 0.000 description 1
- 239000006213 vaginal ring Substances 0.000 description 1
- 230000024883 vasodilation Effects 0.000 description 1
- 239000011800 void material Substances 0.000 description 1
- 239000001993 wax Substances 0.000 description 1
- 230000004580 weight loss Effects 0.000 description 1
- 229940100445 wheat starch Drugs 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/425—Thiazoles
- A61K31/428—Thiazoles condensed with carbocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0014—Skin, i.e. galenical aspects of topical compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0053—Mouth and digestive tract, i.e. intraoral and peroral administration
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/06—Ointments; Bases therefor; Other semi-solid forms, e.g. creams, sticks, gels
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/20—Pills, tablets, discs, rods
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/4841—Filling excipients; Inactive ingredients
- A61K9/4866—Organic macromolecular compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P39/00—General protective or antinoxious agents
- A61P39/06—Free radical scavengers or antioxidants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Engineering & Computer Science (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- Physiology (AREA)
- Nutrition Science (AREA)
- Dermatology (AREA)
- Biochemistry (AREA)
- Psychiatry (AREA)
- Hospice & Palliative Care (AREA)
- Psychology (AREA)
- Toxicology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Tires In General (AREA)
- Plural Heterocyclic Compounds (AREA)
- Thiazole And Isothizaole Compounds (AREA)
Abstract
Description
Claims (77)
- R(+) 프라미펙솔을 포함하는 제1 성분, 및 S(-) 프라미펙솔을 포함하는 제2 성분을 포함하는, 다성분 약제학적 조성물.
- 제1항에 있어서, R(+) 프라미펙솔을 포함하는 상기 제1 성분이 속방형 제형이고 S(-) 프라미펙솔을 포함하는 상기 제2 성분이 속방형 제형인, 다성분 약제학적 조성물.
- 제1항에 있어서, R(+) 프라미펙솔을 포함하는 상기 제1 성분이 속방형 제형이고 S(-) 프라미펙솔을 포함하는 상기 제2 성분이 서방형 제형인, 다성분 약제학적 조성물.
- 제1항에 있어서, R(+) 프라미펙솔을 포함하는 상기 제1 성분이 서방형 제형이고 S(-) 프라미펙솔을 포함하는 상기 제2 성분이 속방형 제형인, 다성분 약제학적 조성물.
- 제1항에 있어서, R(+) 프라미펙솔을 포함하는 상기 제1 성분이 서방형 제형이고 S(-) 프라미펙솔을 포함하는 상기 제2 성분이 서방형 제형인, 다성분 약제학적 조성물.
- 제1항에 있어서, 장관내 투여에 적합한 다성분 약제학적 조성물.
- 제6항에 있어서, 상기 장관내 투여가 경구, 직장 또는 비내 투여로부터 선택되는, 다성분 약제학적 조성물.
- 제1항에 있어서, 경구 투여에 적합한 다성분 약제학적 조성물.
- 제6항에 있어서, 정제, 캡슐제 및 액제로부터 선택되는, 다성분 약제학적 조성물.
- 제1항에 있어서, 2중층 정제인 다성분 약제학적 조성물.
- 제1항에 있어서, 캡슐제인 다성분 약제학적 조성물.
- 제1항에 있어서, 국소 투여에 적합한 다성분 약제학적 조성물.
- 제1항에 있어서, 비경구 투여에 적합한 다성분 약제학적 조성물.
- 제13항에 있어서, 상기 비경구 투여가 혈관내 투여, 피하 주사 및 근육내 투 여로부터 선택되는, 다성분 약제학적 조성물.
- 제1항에 있어서, 상기 제1 성분이 치료학적 유효량의 R(+) 프라미펙솔을 포함하고 상기 제2 성분이 치료학적 유효량의 S(-) 프라미펙솔을 포함하는, 다성분 약제학적 조성물.
- 제15항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔이 약 100 내지 약 3,000mg 범위인, 다성분 약제학적 조성물.
- 제15항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔이 약 300 내지 약 1,500mg 범위인, 다성분 약제학적 조성물.
- 제15항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔이 약 500 내지 약 1,000mg 범위인, 다성분 약제학적 조성물.
- 제15항에 있어서, 상기 치료학적 유효량의 S(-) 프라미펙솔이 약 0.375 내지 약 4.5mg/day 범위인, 다성분 약제학적 조성물.
- 제1항에 있어서, R(+) 프라미펙솔을 포함하는 상기 제1 성분이 제1 속도로 방출되고, S(-) 프라미펙솔을 포함하는 상기 제2 성분이 제2 속도로 방출되는, 다 성분 약제학적 조성물.
- 제20항에 있어서, 상기 제1 속도가 상기 제2 속도보다 빠른, 다성분 약제학적 조성물.
- 제20항에 있어서, 상기 제1 속도가 상기 제2 속도보다 느린, 다성분 약제학적 조성물.
- 제1항에 있어서, 상기 제1 성분이, 프라미펙솔의 R(+) 에난티오머의 키랄 순도가 80% 이상인, 다성분 약제학적 조성물.
- 제1항에 있어서, 상기 제1 성분이, 프라미펙솔의 R(+) 에난티오머의 키랄 순도가 90% 이상인, 다성분 약제학적 조성물.
- 제1항에 있어서, 상기 제1 성분이, 프라미펙솔의 R(+) 에난티오머의 키랄 순도가 95% 이상인, 다성분 약제학적 조성물.
- 제1항에 있어서, 상기 제1 성분이, 프라미펙솔의 R(+) 에난티오머의 키랄 순도가 99% 이상인, 다성분 약제학적 조성물.
- 제1항에 있어서, 상기 제1 성분이, 프라미펙솔의 R(+) 에난티오머의 키랄 순도가 100% 이상인, 다성분 약제학적 조성물.
- 제1항에 있어서, 상기 제1 성분이, 프라미펙솔의 S(-) 에난티오머의 키랄 순도가 80% 이상인, 다성분 약제학적 조성물.
- 제1항에 있어서, 상기 제1 성분이, 프라미펙솔의 S(-) 에난티오머의 키랄 순도가 90% 이상인, 다성분 약제학적 조성물.
- 제1항에 있어서, 상기 제1 성분이, 프라미펙솔의 S(-) 에난티오머의 키랄 순도가 95% 이상인, 다성분 약제학적 조성물.
- 제1항에 있어서, 상기 제1 성분이, 프라미펙솔의 S(-) 에난티오머의 키랄 순도가 99% 이상인, 다성분 약제학적 조성물.
- 제1항에 있어서, 상기 제1 성분이, 프라미펙솔의 S(-) 에난티오머의 키랄 순도가 100% 이상인, 다성분 약제학적 조성물.
- S(-) 프라미펙솔과 R(+) 프라미펙솔을 서방형 매트릭스 내에 포함하는, 서방형 약제학적 조성물.
- 제33항에 있어서, 상기 R(+) 프라미펙솔이 제1 서방형 매트릭스에 존재하고 상기 S(-) 프라미펙솔이 제2 서방형 매트릭스에 존재하는, 서방형 약제학적 조성물.
- 제33항에 있어서, 장관내 투여에 적합한 서방형 약제학적 조성물.
- 제35항에 있어서, 상기 장관내 투여가 경구, 직장 또는 비내 투여로부터 선택되는, 서방형 약제학적 조성물.
- 제33항에 있어서, 경구 투여에 적합한 서방형 약제학적 조성물.
- 제37항에 있어서, 정제, 캡슐제 및 액제로부터 선택되는, 서방형 약제학적 조성물.
- 제33항에 있어서, 2중층 정제인 서방형 약제학적 조성물.
- 제33항에 있어서, 캡슐제인 서방형 약제학적 조성물.
- 제33항에 있어서, 비경구 투여에 적합한 서방형 약제학적 조성물.
- 제41항에 있어서, 상기 비경구 투여가 혈관내 투여, 피하 주사 및 근육내 투여로부터 선택되는, 서방형 약제학적 조성물.
- 제33항에 있어서, 치료학적 유효량의 R(+) 프라미펙솔을 포함하는, 서방형 약제학적 조성물.
- 제43항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔이 약 100 내지 약 3,000mg 범위인, 서방형 약제학적 조성물.
- 제43항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔이 약 300 내지 약 1,500mg 범위인, 서방형 약제학적 조성물.
- 제43항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔이 약 500 내지 약 1,000mg 범위인, 서방형 약제학적 조성물.
- 제33항에 있어서, 치료학적 유효량의 S(-) 프라미펙솔을 포함하는, 서방형 약제학적 조성물.
- 제47항에 있어서, 상기 치료학적 유효량의 S(-) 프라미펙솔이 약 0.375 내지 약 4.5mg/day 범위인, 서방형 약제학적 조성물.
- 제34항에 있어서, 상기 제1 서방형 매트릭스가 상기 제2 서방형 매트릭스와 상이한, 서방형 약제학적 조성물.
- 제34항에 있어서, 상기 R(+) 프라미펙솔이 제1 속도로 방출되고 상기 S(-) 프라미펙솔이 제2 속도로 방출되는, 서방형 약제학적 조성물.
- 제50항에 있어서, 상기 제1 속도가 상기 제2 속도보다 빠른, 서방형 약제학적 조성물.
- 제50항에 있어서, 상기 제1 속도가 상기 제2 속도보다 느린, 서방형 약제학적 조성물.
- 제33항에 있어서, 상기 R(+) 프라미펙솔이, 프라미펙솔의 R(+) 에난티오머의 키랄 순도가 80% 이상인, 서방형 약제학적 조성물.
- 제33항에 있어서, 상기 R(+) 프라미펙솔이, 프라미펙솔의 R(+) 에난티오머의 키랄 순도가 90% 이상인, 서방형 약제학적 조성물.
- 제33항에 있어서, 상기 R(+) 프라미펙솔이, 프라미펙솔의 R(+) 에난티오머의 키랄 순도가 95% 이상인, 서방형 약제학적 조성물.
- 제33항에 있어서, 상기 R(+) 프라미펙솔이, 프라미펙솔의 R(+) 에난티오머의 키랄 순도가 99% 이상인, 서방형 약제학적 조성물.
- 제33항에 있어서, 상기 R(+) 프라미펙솔이, 프라미펙솔의 R(+) 에난티오머의 키랄 순도가 100% 이상인, 서방형 약제학적 조성물.
- 제33항에 있어서, 상기 S(-) 프라미펙솔이, 프라미펙솔의 S(-) 에난티오머의 키랄 순도가 80% 이상인, 서방형 약제학적 조성물.
- 제33항에 있어서, 상기 S(-) 프라미펙솔이, 프라미펙솔의 S(-) 에난티오머의 키랄 순도가 90% 이상인, 서방형 약제학적 조성물.
- 제33항에 있어서, 상기 S(-) 프라미펙솔이, 프라미펙솔의 S(-) 에난티오머의 키랄 순도가 95% 이상인, 서방형 약제학적 조성물.
- 제33항에 있어서, 상기 S(-) 프라미펙솔이, 프라미펙솔의 S(-) 에난티오머의 키랄 순도가 99% 이상인, 서방형 약제학적 조성물.
- 제33항에 있어서, 상기 S(-) 프라미펙솔이, 프라미펙솔의 S(-) 에난티오머의 키랄 순도가 100% 이상인, 서방형 약제학적 조성물.
- 치료학적 유효량의 R(+) 프라미펙솔 및 특정량의 S(-) 프라미펙솔을 투여하는 것을 포함하는, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제63항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔이 약 100 내지 약 3,000mg 범위인, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제63항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔이 약 300 내지 약 1,500mg 범위인, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제63항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔이 약 500 내지 약 1,000mg 범위인, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제63항에 있어서, 상기 S(-) 프라미펙솔의 양이 치료학적 유효량인, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제67항에 있어서, 상기 치료학적 유효량의 S(-) 프라미펙솔이 약 0.375 내지 약 4.5mg/day 범위인, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제63항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔 및 상기한 양의 S(-) 프라미펙솔이 1일 1 내지 5회 투여되는, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제63항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔 및 상기한 양의 S(-) 프라미펙솔이 1일 3회 투여되는, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제63항에 있어서, 상기 치료학적 유효량의 R(+) 프라미펙솔 및 상기한 양의 S(-) 프라미펙솔이 단일의 약제학적 조성물로 투여되는, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제71항에 있어서, 상기 약제학적 조성물이, R(+) 에난티오머인 프라미펙솔의 키랄 순도가 80% 이상인, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제71항에 있어서, 상기 약제학적 조성물이, R(+) 에난티오머인 프라미펙솔의 키랄 순도가 90% 이상인, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제71항에 있어서, 상기 약제학적 조성물이, R(+) 에난티오머인 프라미펙솔의 키랄 순도가 95% 이상인, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제71항에 있어서, 상기 약제학적 조성물이, R(+) 에난티오머인 프라미펙솔의 키랄 순도가 99% 이상인, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
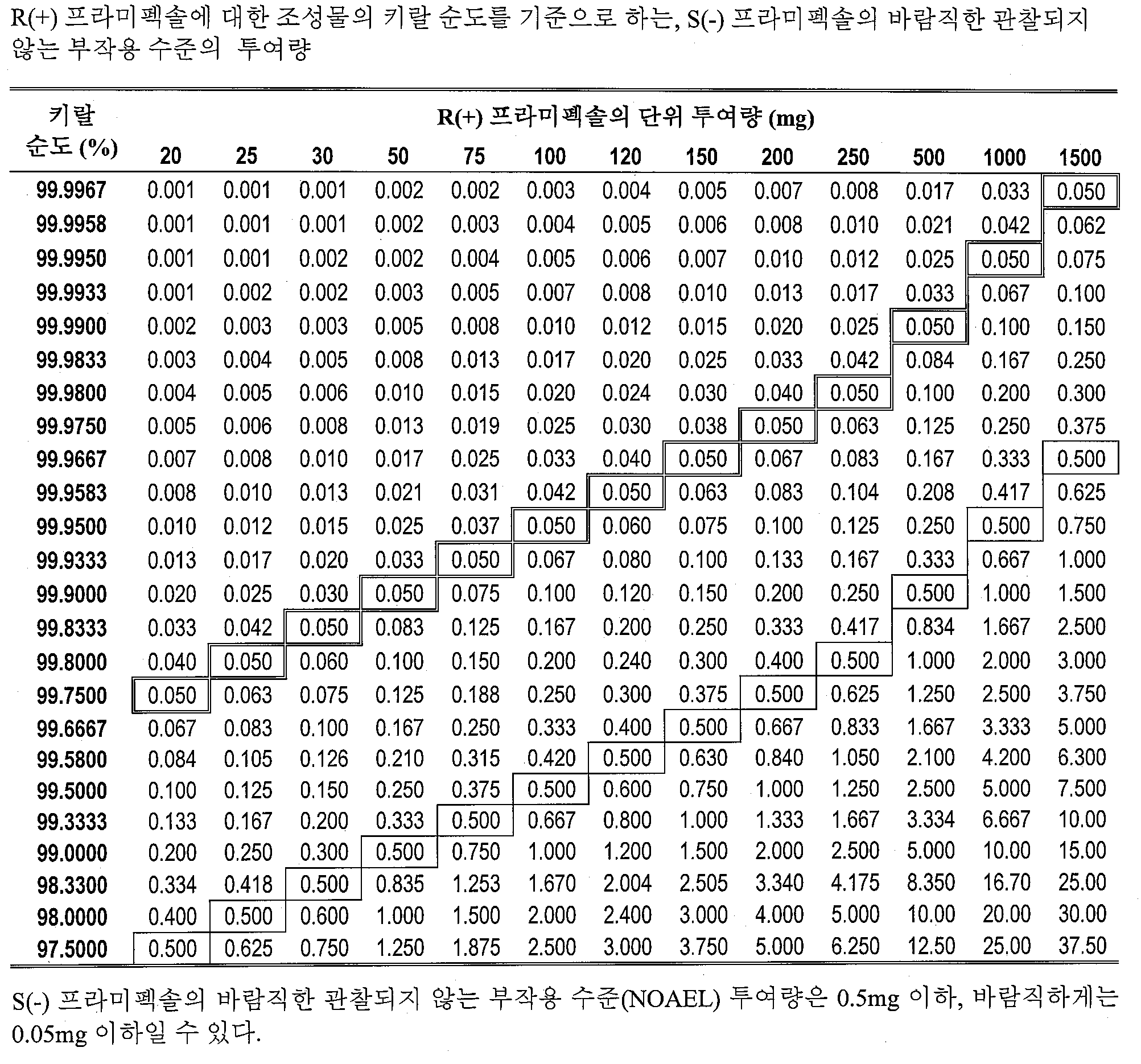
- 제63항에 있어서, 상기한 양의 S(-) 프라미펙솔의 양이 관찰되지 않는 부작용 수준의 S(-) 프라미펙솔 양인, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
- 제76항에 있어서, 상기 관찰되지 않는 부작용 수준의 S(-) 프라미펙솔 양이 1.5mg 이하인, 파킨슨병 또는 이의 증후군을 치료 또는 예방하는 방법.
Applications Claiming Priority (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US74732006P | 2006-05-16 | 2006-05-16 | |
| US60/747,320 | 2006-05-16 | ||
| US87000906P | 2006-12-14 | 2006-12-14 | |
| US60/870,009 | 2006-12-14 | ||
| US89479907P | 2007-03-14 | 2007-03-14 | |
| US89483507P | 2007-03-14 | 2007-03-14 | |
| US89482907P | 2007-03-14 | 2007-03-14 | |
| US60/894,835 | 2007-03-14 | ||
| US60/894,829 | 2007-03-14 | ||
| US60/894,799 | 2007-03-14 | ||
| US11/733,642 US20070259930A1 (en) | 2006-04-10 | 2007-04-10 | Compositions and methods of using r(+) pramipexole |
| US11/733,642 | 2007-04-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20090021169A true KR20090021169A (ko) | 2009-02-27 |
Family
ID=40134109
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020087030672A Ceased KR20090021169A (ko) | 2006-05-16 | 2007-05-16 | R(+) 및 s(-) 프라미펙솔을 포함하는 조성물 및 이의 사용 방법 |
Country Status (13)
| Country | Link |
|---|---|
| US (3) | US8017598B2 (ko) |
| EP (1) | EP2026803B1 (ko) |
| JP (1) | JP2009537565A (ko) |
| KR (1) | KR20090021169A (ko) |
| CN (2) | CN102160865A (ko) |
| AT (1) | ATE537826T1 (ko) |
| AU (1) | AU2007253899A1 (ko) |
| CA (1) | CA2652251A1 (ko) |
| DK (1) | DK2026803T3 (ko) |
| ES (1) | ES2379117T3 (ko) |
| IL (1) | IL195256A0 (ko) |
| PL (1) | PL2026803T3 (ko) |
| WO (1) | WO2007137071A2 (ko) |
Families Citing this family (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8518926B2 (en) | 2006-04-10 | 2013-08-27 | Knopp Neurosciences, Inc. | Compositions and methods of using (R)-pramipexole |
| WO2007121188A2 (en) * | 2006-04-10 | 2007-10-25 | Knopp Neurosciences, Inc. | Compositions and methods of using r(+) pramipexole |
| KR20090021169A (ko) | 2006-05-16 | 2009-02-27 | 크놉 뉴로사이언시스 인코포레이티드 | R(+) 및 s(-) 프라미펙솔을 포함하는 조성물 및 이의 사용 방법 |
| EP2497473A1 (en) * | 2006-05-16 | 2012-09-12 | Knopp Neurosciences, Inc. | Therapeutically effective amounts of R(+) and S(-) pramipexole for use in the treatment of Parkinson's disease and pharmaceutical compositions thereof |
| US8524695B2 (en) | 2006-12-14 | 2013-09-03 | Knopp Neurosciences, Inc. | Modified release formulations of (6R)-4,5,6,7-tetrahydro-N6-propyl-2,6-benzothiazole-diamine and methods of using the same |
| JP2010521496A (ja) | 2007-03-14 | 2010-06-24 | ノップ ニューロサイエンシーズ、インク. | キラル精製置換ベンゾチアゾールジアミンの合成 |
| AU2008224869A1 (en) * | 2007-03-14 | 2008-09-18 | Knopp Neurosciences, Inc. | Modified release formulations of (6R)-4,5,6,7-tetrahydro-N6-propyl-2,6-benzothiazole-diamine and methods of using the same |
| WO2009109990A2 (en) * | 2008-01-24 | 2009-09-11 | Sun Pharmaceutical Industries Ltd. | Pharmaceutical composition of pramipexole |
| NO2303330T3 (ko) | 2008-06-06 | 2018-04-14 | ||
| US20110190356A1 (en) * | 2008-08-19 | 2011-08-04 | Knopp Neurosciences Inc. | Compositions and Methods of Using (R)- Pramipexole |
| CA2749253A1 (en) * | 2009-04-09 | 2010-10-14 | Purdue Pharma | Once-daily oral ir/cr pramipexole formulation |
| WO2010148409A1 (en) * | 2009-06-19 | 2010-12-23 | Knopp Neurosciences, Inc. | Compositions and methods for treating amyotrophic lateral sclerosis |
| US20120059013A1 (en) * | 2010-03-09 | 2012-03-08 | Boehringer Ingelheim International Gmbh | Method of treating early morning akinesia in subjects having parkinson's disease |
| US9512096B2 (en) | 2011-12-22 | 2016-12-06 | Knopp Biosciences, LLP | Synthesis of amine substituted 4,5,6,7-tetrahydrobenzothiazole compounds |
| US9662313B2 (en) | 2013-02-28 | 2017-05-30 | Knopp Biosciences Llc | Compositions and methods for treating amyotrophic lateral sclerosis in responders |
| US9468630B2 (en) | 2013-07-12 | 2016-10-18 | Knopp Biosciences Llc | Compositions and methods for treating conditions related to increased eosinophils |
| ES3043076T3 (en) | 2013-07-12 | 2025-11-24 | Areteia Therapeutics Inc | Treating elevated levels of eosinophils and/or basophils |
| EP3038467B1 (en) | 2013-08-13 | 2020-07-29 | Knopp Biosciences LLC | Compositions and methods for treating plasma cell disorders and b-cell prolymphocytic disorders |
| US9763918B2 (en) | 2013-08-13 | 2017-09-19 | Knopp Biosciences Llc | Compositions and methods for treating chronic urticaria |
| EP2870965A1 (en) * | 2013-11-06 | 2015-05-13 | Sanovel Ilac Sanayi ve Ticaret A.S. | Oral liquid pharmaceutical formulations of pramipexole |
| EP3600291A4 (en) * | 2017-03-27 | 2020-12-23 | Chase Therapeutics Corporation | COMPOSITIONS AND METHODS OF TREATMENT OF SYNUCLEINOPATHIES |
| IL312486B2 (en) | 2017-04-10 | 2025-05-01 | Chase Therapeutics Corp | NK1 antagonist combination and method for treating synucleinopathies |
| EP3609578A4 (en) | 2017-04-13 | 2021-03-10 | Chase Therapeutics Corporation | PHARMACEUTICAL COMBINATION AND ITS USE FOR THE TREATMENT OF SYNUCLEINOPATHIES |
| CN111588692B (zh) * | 2020-05-23 | 2022-11-22 | 常州市第四制药厂有限公司 | 盐酸普拉克索口服溶液 |
| CN115260119B (zh) * | 2022-07-26 | 2023-02-10 | 山东京卫制药有限公司 | 一种普拉克索昔萘酸盐及其药物缓释制剂 |
Family Cites Families (153)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3797494A (en) * | 1969-04-01 | 1974-03-19 | Alza Corp | Bandage for the administration of drug by controlled metering through microporous materials |
| US3598122A (en) * | 1969-04-01 | 1971-08-10 | Alza Corp | Bandage for administering drugs |
| US3598123A (en) * | 1969-04-01 | 1971-08-10 | Alza Corp | Bandage for administering drugs |
| US3731683A (en) * | 1971-06-04 | 1973-05-08 | Alza Corp | Bandage for the controlled metering of topical drugs to the skin |
| US4144317A (en) * | 1975-05-30 | 1979-03-13 | Alza Corporation | Device consisting of copolymer having acetoxy groups for delivering drugs |
| US4031894A (en) * | 1975-12-08 | 1977-06-28 | Alza Corporation | Bandage for transdermally administering scopolamine to prevent nausea |
| US4201211A (en) * | 1977-07-12 | 1980-05-06 | Alza Corporation | Therapeutic system for administering clonidine transdermally |
| US4286592A (en) * | 1980-02-04 | 1981-09-01 | Alza Corporation | Therapeutic system for administering drugs to the skin |
| US4314557A (en) * | 1980-05-19 | 1982-02-09 | Alza Corporation | Dissolution controlled active agent dispenser |
| US4395859A (en) * | 1980-06-05 | 1983-08-02 | State Of Illinois, Department Of Transportation | Method and apparatus for securing an object to a support structure |
| US4327725A (en) * | 1980-11-25 | 1982-05-04 | Alza Corporation | Osmotic device with hydrogel driving member |
| US4379454A (en) * | 1981-02-17 | 1983-04-12 | Alza Corporation | Dosage for coadministering drug and percutaneous absorption enhancer |
| US4849226A (en) * | 1981-06-29 | 1989-07-18 | Alza Corporation | Method for increasing oxygen supply by administering vasodilator |
| US4725272A (en) * | 1981-06-29 | 1988-02-16 | Alza Corporation | Novel bandage for administering beneficial drug |
| US4435180A (en) * | 1982-05-25 | 1984-03-06 | Alza Corporation | Elastomeric active agent delivery system and method of use |
| US4559222A (en) | 1983-05-04 | 1985-12-17 | Alza Corporation | Matrix composition for transdermal therapeutic system |
| US4612008A (en) * | 1983-05-11 | 1986-09-16 | Alza Corporation | Osmotic device with dual thermodynamic activity |
| US5082668A (en) * | 1983-05-11 | 1992-01-21 | Alza Corporation | Controlled-release system with constant pushing source |
| US4783337A (en) * | 1983-05-11 | 1988-11-08 | Alza Corporation | Osmotic system comprising plurality of members for dispensing drug |
| US4704282A (en) * | 1984-06-29 | 1987-11-03 | Alza Corporation | Transdermal therapeutic system having improved delivery characteristics |
| US4588580B2 (en) * | 1984-07-23 | 1999-02-16 | Alaz Corp | Transdermal administration of fentanyl and device therefor |
| US4626539A (en) | 1984-08-10 | 1986-12-02 | E. I. Dupont De Nemours And Company | Trandermal delivery of opioids |
| US4573995A (en) * | 1984-10-09 | 1986-03-04 | Alza Corporation | Transdermal therapeutic systems for the administration of naloxone, naltrexone and nalbuphine |
| US4568343A (en) * | 1984-10-09 | 1986-02-04 | Alza Corporation | Skin permeation enhancer compositions |
| EP0186087B1 (de) | 1984-12-22 | 1989-08-23 | Dr. Karl Thomae GmbH | Tetrahydro-benzthiazole, deren Herstellung und deren Verwendung als Zwischenprodukte oder als Arnzneimittel |
| US4806341A (en) * | 1985-02-25 | 1989-02-21 | Rutgers, The State University Of New Jersey | Transdermal absorption dosage unit for narcotic analgesics and antagonists and process for administration |
| US4904475A (en) * | 1985-05-03 | 1990-02-27 | Alza Corporation | Transdermal delivery of drugs from an aqueous reservoir |
| US4645502A (en) * | 1985-05-03 | 1987-02-24 | Alza Corporation | Transdermal delivery of highly ionized fat insoluble drugs |
| US4698062A (en) * | 1985-10-30 | 1987-10-06 | Alza Corporation | Medical device for pulsatile transdermal delivery of biologically active agents |
| EP0249343B1 (en) * | 1986-06-13 | 1992-01-08 | Alza Corporation | Moisture activation of transdermal drug delivery system |
| US4938759A (en) * | 1986-09-02 | 1990-07-03 | Alza Corporation | Transdermal delivery device having a rate controlling adhesive |
| US4908027A (en) * | 1986-09-12 | 1990-03-13 | Alza Corporation | Subsaturated transdermal therapeutic system having improved release characteristics |
| US5344656A (en) * | 1986-09-12 | 1994-09-06 | Alza Corporation | Subsaturated transdermal therapeutic system having improved release characteristics |
| US4816258A (en) * | 1987-02-26 | 1989-03-28 | Alza Corporation | Transdermal contraceptive formulations |
| US4788062A (en) * | 1987-02-26 | 1988-11-29 | Alza Corporation | Transdermal administration of progesterone, estradiol esters, and mixtures thereof |
| US5071656A (en) | 1987-03-05 | 1991-12-10 | Alza Corporation | Delayed onset transdermal delivery device |
| US4943435A (en) * | 1987-10-05 | 1990-07-24 | Pharmetrix Corporation | Prolonged activity nicotine patch |
| US4917895A (en) * | 1987-11-02 | 1990-04-17 | Alza Corporation | Transdermal drug delivery device |
| US4781924A (en) * | 1987-11-09 | 1988-11-01 | Alza Corporation | Transdermal drug delivery device |
| US5004610A (en) * | 1988-06-14 | 1991-04-02 | Alza Corporation | Subsaturated nicotine transdermal therapeutic system |
| DE69009540T2 (de) * | 1989-03-15 | 1994-09-29 | Nitto Denko Corp | Arzneimittel enthaltendes Heftpflaster. |
| US5091190A (en) * | 1989-09-05 | 1992-02-25 | Alza Corporation | Delivery system for administration blood-glucose lowering drug |
| US5591454A (en) * | 1989-09-05 | 1997-01-07 | Alza Corporation | Method for lowering blood glucose |
| US5024843A (en) * | 1989-09-05 | 1991-06-18 | Alza Corporation | Oral hypoglycemic glipizide granulation |
| DE3937271A1 (de) * | 1989-11-09 | 1991-05-16 | Boehringer Ingelheim Kg | Transdermale applikation von 2-amino-6-n-propylamino-4,5,6,7-tetrahydrobenzothiazol |
| US5069909A (en) | 1990-06-20 | 1991-12-03 | Cygnus Therapeutic Systems | Transdermal administration of buprenorphine |
| US5122382A (en) * | 1990-10-29 | 1992-06-16 | Alza Corporation | Transdermal contraceptive formulations, methods and devices |
| US5314694A (en) * | 1990-10-29 | 1994-05-24 | Alza Corporation | Transdermal formulations, methods and devices |
| FR2688138B1 (fr) | 1992-03-06 | 1995-05-05 | Rhone Poulenc Rorer Sa | Application de l'amino-2 trifluoromethoxy-6 benzothiazole pour obtenir un medicament destine au traitement de la sclerose laterale amyotrophique. |
| KR950701516A (ko) * | 1992-05-13 | 1995-04-28 | 에드워드 엘. 만델 | 옥시부티닌의 경피 투여(Transdermal Administration of Oxybutynin) |
| US5792664A (en) | 1992-05-29 | 1998-08-11 | The Rockefeller University | Methods for producing and analyzing biopolymer ladders |
| NZ267842A (en) | 1993-05-28 | 1997-09-22 | Baylor College Medicine | Apparatus for measuring molecular mass (by mass spectrometry) in which the sample is ionised on the sample holder and desorbed therefrom by laser pulses |
| US5442117A (en) * | 1993-12-13 | 1995-08-15 | Albemarle Corporation | Enantiomeric resolution |
| US5635203A (en) * | 1994-09-29 | 1997-06-03 | Alza Corporation | Transdermal device having decreased delamination |
| US6156777A (en) | 1994-12-15 | 2000-12-05 | Pharmacia & Upjohn Company | Use of pramipexole as a neuroprotective agent |
| US5650420A (en) * | 1994-12-15 | 1997-07-22 | Pharmacia & Upjohn Company | Pramipexole as a neuroprotective agent |
| US5674895A (en) | 1995-05-22 | 1997-10-07 | Alza Corporation | Dosage form comprising oxybutynin |
| US6262115B1 (en) * | 1995-05-22 | 2001-07-17 | Alza Coporation | Method for the management of incontinence |
| US5912268A (en) * | 1995-05-22 | 1999-06-15 | Alza Corporation | Dosage form and method for treating incontinence |
| CN1104241C (zh) | 1995-10-26 | 2003-04-02 | 萨诺费合成实验室 | 1-(2-萘-2-基乙基)-4-(3-三氟甲基苯基)-1,2,3,6-四氢吡啶用于制备治疗肌萎缩性脊髓侧索硬化的药物的用途 |
| US6929801B2 (en) * | 1996-02-19 | 2005-08-16 | Acrux Dds Pty Ltd | Transdermal delivery of antiparkinson agents |
| US6919373B1 (en) * | 1996-11-12 | 2005-07-19 | Alza Corporation | Methods and devices for providing prolonged drug therapy |
| GB9705428D0 (en) * | 1997-03-15 | 1997-04-30 | Knoll Ag | Therapeutic agents |
| US5804215A (en) * | 1997-03-21 | 1998-09-08 | L. Perrigo Company | Transdermal patch disposal system and method |
| KR20010024251A (ko) | 1997-09-23 | 2001-03-26 | 사이퍼젠 바이오시스템스, 인코오포레이티드 | 비행 시간형 질량 분석계용 2차 이온 발생기 검출기 |
| US6197339B1 (en) | 1997-09-30 | 2001-03-06 | Pharmacia & Upjohn Company | Sustained release tablet formulation to treat Parkinson's disease |
| PE20000728A1 (es) | 1998-06-26 | 2000-08-21 | Cocensys Inc | Heterociclos 4-bencil piperidina alquilsulfoxido y su uso como antagonistas receptores subtipo-selectivo nmda |
| DE19830201A1 (de) | 1998-07-07 | 2000-01-13 | Boehringer Ingelheim Pharma | Mittel mit antidepressiver Wirkung |
| CA2301899C (en) | 1998-07-27 | 2008-11-18 | Boehringer Ingelheim Pharma Kg | Agent with an antidepressant activity |
| US6322819B1 (en) | 1998-10-21 | 2001-11-27 | Shire Laboratories, Inc. | Oral pulsed dose drug delivery system |
| CN100444830C (zh) | 1998-11-02 | 2008-12-24 | 伊兰公司,Plc | 多颗粒改进释放组合物 |
| US6776984B1 (en) | 1999-08-20 | 2004-08-17 | George R. Schwartz | Induced regeneration and repair of damaged neurons and nerve axon myelin |
| CA2375843A1 (en) * | 1999-06-04 | 2000-12-14 | Qi Zhao | Compositions and methods for inhibiting cell death |
| DE19938825A1 (de) | 1999-08-19 | 2001-04-26 | Boehringer Ingelheim Pharma | Wirkstoffkombination mit Clonidin |
| US6480820B1 (en) | 1999-09-20 | 2002-11-12 | Advanced Cochlear Systems, Inc. | Method of processing auditory data |
| US6750235B1 (en) * | 1999-09-30 | 2004-06-15 | The General Hospital Corporation | Pramipexole as a treatment for cocaine craving |
| AU7620600A (en) | 1999-09-30 | 2001-04-30 | General Hospital Corporation, The | Use of pramipexole as a treatment for cocaine craving |
| US6443976B1 (en) * | 1999-11-30 | 2002-09-03 | Akorn, Inc. | Methods for treating conditions and illnesses associated with abnormal vasculature |
| AU2001233156A1 (en) * | 2000-02-01 | 2001-08-14 | Human Genome Sciences, Inc. | Bcl-2-like polynucleotides, polypeptides, and antibodies |
| PE20011074A1 (es) | 2000-02-23 | 2001-10-04 | Upjohn Co | Uso de pramipexol en el tratamiento de trastornos de adiccion |
| CA2443806A1 (en) | 2000-04-14 | 2002-10-25 | Metabolon, Inc. | Methods for drug discovery, disease treatment, and diagnosis using metabolomics |
| US6955821B2 (en) * | 2000-04-28 | 2005-10-18 | Adams Laboratories, Inc. | Sustained release formulations of guaifenesin and additional drug ingredients |
| WO2002004409A2 (en) | 2000-07-06 | 2002-01-17 | Us Gov Health & Human Serv | Tetrahydrobenzothiazole analogues as neuroprotective agents |
| DE10037619A1 (de) | 2000-08-02 | 2002-02-14 | Daimler Chrysler Ag | Anordnung von Bedienelementen |
| ES2187249B1 (es) * | 2000-09-18 | 2004-09-16 | Synthon Bv | Procedimiento para la preparacion de 2-amino-6-(alquil)amino-4,5,6,7-tetrahidrobenzotiazoles. |
| AU2001296703A1 (en) | 2000-10-06 | 2002-04-15 | Xenoport, Inc. | Bile acid prodrugs of l-dopa and their use in the sustained treatment of parkinsonism |
| US6618138B2 (en) | 2000-10-12 | 2003-09-09 | Jed Khoury | Scanning fluorescent systems for various diagnostic |
| ATE540421T1 (de) | 2000-11-16 | 2012-01-15 | Bio Rad Laboratories | Methode zum analysieren von massenspektren |
| US20020177626A1 (en) | 2001-01-19 | 2002-11-28 | Cook Graham D. | Treatment of sleep disturbances |
| EP1379269B1 (en) * | 2001-04-09 | 2009-03-04 | Neurosearch A/S | Adenosine a2a receptor antagonists combined with neurotrophic activity compounds in the treatment of parkinson's disease |
| US20030013120A1 (en) | 2001-07-12 | 2003-01-16 | Patz Edward F. | System and method for differential protein expression and a diagnostic biomarker discovery system and method using same |
| GB0117618D0 (en) * | 2001-07-19 | 2001-09-12 | Phoqus Ltd | Pharmaceutical dosage form |
| DE10137082A1 (de) | 2001-07-28 | 2003-02-13 | Hexal Ag | Matrixkontrolliertes transdermales therapeutisches System zur Anwendung von Pramiprexol und Ropinirol |
| US20060281797A1 (en) | 2001-12-11 | 2006-12-14 | University Of Virginia Patent Foundation | Neurorestoration with R(+) Pramipexole |
| EP2305252A1 (en) | 2001-12-11 | 2011-04-06 | University Of Virginia Patent Foundation | Use of pramipexole to treat amyotrophic lateral sclerosis |
| TWI262083B (en) | 2001-12-28 | 2006-09-21 | Syngenta Participations Ag | Microbially-expressed thermotolerant phytase for animal feed |
| ES2298351T5 (es) * | 2002-01-16 | 2012-01-26 | BOEHRINGER INGELHEIM PHARMA GMBH & CO. KG | Método para producir un comprimido farmacéutico de dos capas que comprenden telmisartán e hidroclorotiazida. |
| US6927036B2 (en) | 2002-02-19 | 2005-08-09 | Xero Port, Inc. | Methods for synthesis of prodrugs from 1-acyl-alkyl derivatives and compositions thereof |
| AU2003225837B2 (en) * | 2002-03-15 | 2008-11-06 | Forest Laboratories Holdings Limited | NE and 5-HT reuptake inhibitors for treating visceral pain syndromes |
| WO2003087834A2 (en) | 2002-04-08 | 2003-10-23 | Affinium Pharmaceuticals, Inc. | High throughput analysis of recombinant proteins in multi-well plates |
| MY142204A (en) | 2002-07-25 | 2010-10-29 | Pharmacia Corp | Pramipexole once-daily dosage form |
| US20050226926A1 (en) * | 2002-07-25 | 2005-10-13 | Pfizer Inc | Sustained-release tablet composition of pramipexole |
| US20050074865A1 (en) * | 2002-08-27 | 2005-04-07 | Compound Therapeutics, Inc. | Adzymes and uses thereof |
| GB0221513D0 (en) * | 2002-09-17 | 2002-10-23 | Generics Uk Ltd | Novel compounds and processes |
| US20040121010A1 (en) * | 2002-10-25 | 2004-06-24 | Collegium Pharmaceutical, Inc. | Pulsatile release compositions of milnacipran |
| GB2394951A (en) | 2002-11-04 | 2004-05-12 | Cipla Ltd | One pot synthesis of 2,6-diamino-4,5,6,7-tetrahydro-benzothiazole |
| US20050202093A1 (en) | 2002-12-02 | 2005-09-15 | Kohane Daniel S. | Prolonged suppression of electrical activity in excitable tissues |
| SI1610791T1 (sl) * | 2003-03-31 | 2011-05-31 | Titan Pharmaceuticals Inc | Implantibilna polimerna naprava za zadržano sproščanje dopaminskega agonista |
| CA2529984C (en) | 2003-06-26 | 2012-09-25 | Isa Odidi | Oral multi-functional pharmaceutical capsule preparations of proton pump inhibitors |
| US7662987B2 (en) | 2003-07-15 | 2010-02-16 | Xenoport, Inc. | Methods for synthesis of acyloxyalkyl compounds |
| DE10333393A1 (de) | 2003-07-23 | 2005-02-24 | Lts Lohmann Therapie-Systeme Ag | Transdermales Therapeutisches System mit dem Wirkstoff Pramipexol |
| US7365086B2 (en) * | 2003-07-25 | 2008-04-29 | Synthon Ip Inc. | Pramipexole acid addition salts |
| US20080020028A1 (en) * | 2003-08-20 | 2008-01-24 | Euro-Celtique S.A. | Transdermal dosage form comprising an active agent and a salt and a free-base form of an adverse agent |
| US20050053649A1 (en) * | 2003-09-08 | 2005-03-10 | Anne-Marie Chalmers | Medication delivery device |
| AU2004286240A1 (en) | 2003-10-23 | 2005-05-12 | University Of Pittsburgh Of The Commonwealth System Of Higher Education | Biomarkers for amyotrophic lateral sclerosis |
| CA2560128A1 (en) | 2004-03-19 | 2005-10-06 | Dipharma S.P.A. | Intermediates for the preparation of pramipexole |
| US20050220877A1 (en) * | 2004-03-31 | 2005-10-06 | Patel Ashish A | Bilayer tablet comprising an antihistamine and a decongestant |
| US20080113003A1 (en) * | 2004-04-06 | 2008-05-15 | University Of Saskatchewan | Sinapic Acid Supplementation |
| US7539203B2 (en) | 2004-05-26 | 2009-05-26 | Intel Corporation | Multiple channel flow control with first-in-first-out (FIFO) read/write random access memory (RAM) |
| WO2006003471A2 (en) * | 2004-07-03 | 2006-01-12 | Generics [Uk] Limited | Process for the preparation of pramipexole by chiral chromatography |
| KR20070050081A (ko) | 2004-08-13 | 2007-05-14 | 베링거 인겔하임 인터내셔날 게엠베하 | 프라미펙솔 또는 약제학적으로 허용되는 이의 염을포함하는 연장 방출형 펠렛 제형, 이의 제조방법 및 이의용도 |
| RS52057B2 (sr) | 2004-08-13 | 2018-03-30 | Boehringer Ingelheim Int | Formulacija tablete sa produženim oslobađanjem koja sadrži pramipeksol ili njegovu farmaceutski prihvatljivu so |
| TW200616604A (en) | 2004-08-26 | 2006-06-01 | Nicholas Piramal India Ltd | Nitric oxide releasing prodrugs containing bio-cleavable linker |
| DE102004044578A1 (de) * | 2004-09-13 | 2006-03-30 | Lts Lohmann Therapie-Systeme Ag | Transdermales therapeutisches System mit einer Haftschicht, Verfahren zum Silikonisieren einer Rückschicht des Systems und Verwendung der Rückschicht |
| US20060069263A1 (en) * | 2004-09-30 | 2006-03-30 | Irina Gribun | Process for the reduction of (S)-2-amino-6-propionamido-4,5,6,7-tetrahydrobenzo-thiazole |
| EA013053B1 (ru) * | 2004-11-05 | 2010-02-26 | Бёрингер Ингельхайм Интернациональ Гмбх | Двуслойная таблетка, содержащая телмисартан и амлодипин |
| US7572596B2 (en) | 2004-12-02 | 2009-08-11 | University Of Pittsburgh Of The Commonwealth System Of Higher Education | Modulation of the neuroendoctrine system as a therapy for motor neuron disease |
| WO2006060799A2 (en) | 2004-12-02 | 2006-06-08 | University Of Pittsburgh - Of The Commonwealth System Of Higher Education | Protein biomarkers and therapeutic targets in an animal model for amyotrophic lateral sclerosis |
| WO2006070406A1 (en) * | 2004-12-29 | 2006-07-06 | J.B. Chemicals & Pharmaceuticals Ltd | Bilayer tablets of oxcarbazepine for controlled delivery and a process of preparation thereof |
| US20060148866A1 (en) * | 2004-12-30 | 2006-07-06 | Chemagis Ltd. | Novel process for preparing pramipexole and its optical isomeric mixture by reduction with sodium triacetoxyborohydride |
| US20060286167A1 (en) | 2005-05-02 | 2006-12-21 | Jane Staunton | Compositions and methods for the treatment of neurodegenerative diseases |
| ES2264378B1 (es) | 2005-05-09 | 2007-11-01 | Ragactives, S.L. | Procedimiento para la resolucion de 2-amino-6propilamino-4,5,6,7-tetrahidrobenzotiazol y compuestos intermedios. |
| JP2009504748A (ja) | 2005-08-15 | 2009-02-05 | ユニバーシティ オブ バージニア パテント ファウンデーション | R(+)プラミペキソールを用いた神経回復 |
| EP1931632A4 (en) * | 2005-08-18 | 2011-05-11 | Microbia Inc | USEFUL INDOOR CONNECTIONS |
| RU2008119454A (ru) * | 2005-10-18 | 2009-11-27 | Оно Фармасьютикал Ко., Лтд. (Jp) | Лекарственное средство для защиты моторного нейрона у пациентов с боковым амиотрофическим склерозом |
| PL378587A1 (pl) | 2005-12-29 | 2007-07-09 | Instytut Farmaceutyczny | Sposób wytwarzania (S)-(-)-2-amino-6-n-propyloamino-4,5,6,7-tetrahydrobenzotiazolu i/lub jego soli |
| WO2007090882A2 (en) * | 2006-02-10 | 2007-08-16 | Boehringer Ingelheim International Gmbh | Pharmaceutical extended release compositions comprising pramipexole |
| US8518926B2 (en) * | 2006-04-10 | 2013-08-27 | Knopp Neurosciences, Inc. | Compositions and methods of using (R)-pramipexole |
| WO2007121188A2 (en) | 2006-04-10 | 2007-10-25 | Knopp Neurosciences, Inc. | Compositions and methods of using r(+) pramipexole |
| KR20090021169A (ko) | 2006-05-16 | 2009-02-27 | 크놉 뉴로사이언시스 인코포레이티드 | R(+) 및 s(-) 프라미펙솔을 포함하는 조성물 및 이의 사용 방법 |
| EP2497473A1 (en) | 2006-05-16 | 2012-09-12 | Knopp Neurosciences, Inc. | Therapeutically effective amounts of R(+) and S(-) pramipexole for use in the treatment of Parkinson's disease and pharmaceutical compositions thereof |
| US7675228B2 (en) * | 2006-06-14 | 2010-03-09 | E.I. Du Pont De Nemours And Company | Electroluminescent iridium compounds with silylated, germanylated, and stannylated ligands, and devices made with such compounds |
| RU2009110253A (ru) | 2006-08-24 | 2010-09-27 | БЁРИНГЕР ИНГЕЛЬХАЙМ ФАРМА ГМБХ УНД Ко. КГ (DE) | Способ приготовления таблеток прамипексолдигидрохлорида, обладающих высокой стабильностью при хранении |
| WO2008042611A2 (en) * | 2006-09-29 | 2008-04-10 | Centocor, Inc. | Method of using il6 antagonists with mitoxantrone for prostate cancer |
| US8524695B2 (en) * | 2006-12-14 | 2013-09-03 | Knopp Neurosciences, Inc. | Modified release formulations of (6R)-4,5,6,7-tetrahydro-N6-propyl-2,6-benzothiazole-diamine and methods of using the same |
| EP2101766B1 (en) | 2006-12-14 | 2016-09-21 | Knopp Biosciences LLC | Compositions and methods of using (r)-pramipexole |
| WO2008104847A2 (en) | 2007-02-26 | 2008-09-04 | Wockhardt Research Centre | Processes for the preparation of pramipexole and salts thereof |
| AU2008224869A1 (en) | 2007-03-14 | 2008-09-18 | Knopp Neurosciences, Inc. | Modified release formulations of (6R)-4,5,6,7-tetrahydro-N6-propyl-2,6-benzothiazole-diamine and methods of using the same |
| JP2010521496A (ja) * | 2007-03-14 | 2010-06-24 | ノップ ニューロサイエンシーズ、インク. | キラル精製置換ベンゾチアゾールジアミンの合成 |
| RU2470635C2 (ru) | 2007-05-11 | 2012-12-27 | Сантен Фармасьютикал Ко., Лтд. | Профилактическое или терапевтическое средство, применяемое при заболеваниях заднего отдела глаза |
| US20110190356A1 (en) | 2008-08-19 | 2011-08-04 | Knopp Neurosciences Inc. | Compositions and Methods of Using (R)- Pramipexole |
| WO2010148409A1 (en) | 2009-06-19 | 2010-12-23 | Knopp Neurosciences, Inc. | Compositions and methods for treating amyotrophic lateral sclerosis |
| WO2011150221A2 (en) | 2010-05-26 | 2011-12-01 | Knopp Neurosciences, Inc. | Compounds and methods of modulating mitochondrial bioenergetic efficiency through an interaction with atp synthase (complex v) and its subunits |
-
2007
- 2007-05-16 KR KR1020087030672A patent/KR20090021169A/ko not_active Ceased
- 2007-05-16 JP JP2009511221A patent/JP2009537565A/ja active Pending
- 2007-05-16 ES ES07797504T patent/ES2379117T3/es active Active
- 2007-05-16 EP EP07797504A patent/EP2026803B1/en not_active Not-in-force
- 2007-05-16 CN CN2011100474822A patent/CN102160865A/zh active Pending
- 2007-05-16 US US11/749,497 patent/US8017598B2/en active Active
- 2007-05-16 AU AU2007253899A patent/AU2007253899A1/en not_active Abandoned
- 2007-05-16 PL PL07797504T patent/PL2026803T3/pl unknown
- 2007-05-16 AT AT07797504T patent/ATE537826T1/de active
- 2007-05-16 DK DK07797504.3T patent/DK2026803T3/da active
- 2007-05-16 WO PCT/US2007/069044 patent/WO2007137071A2/en not_active Ceased
- 2007-05-16 CA CA002652251A patent/CA2652251A1/en not_active Abandoned
- 2007-05-16 CN CN2007800179185A patent/CN101448498B/zh not_active Expired - Fee Related
-
2008
- 2008-11-12 IL IL195256A patent/IL195256A0/en unknown
-
2011
- 2011-08-05 US US13/204,351 patent/US8445474B2/en active Active
-
2013
- 2013-04-12 US US13/862,357 patent/US20130230569A1/en not_active Abandoned
Also Published As
| Publication number | Publication date |
|---|---|
| JP2009537565A (ja) | 2009-10-29 |
| CN102160865A (zh) | 2011-08-24 |
| ATE537826T1 (de) | 2012-01-15 |
| CN101448498A (zh) | 2009-06-03 |
| AU2007253899A1 (en) | 2007-11-29 |
| US8017598B2 (en) | 2011-09-13 |
| WO2007137071A2 (en) | 2007-11-29 |
| US20110293718A1 (en) | 2011-12-01 |
| IL195256A0 (en) | 2009-08-03 |
| CA2652251A1 (en) | 2007-11-29 |
| US20080014259A1 (en) | 2008-01-17 |
| US20130230569A1 (en) | 2013-09-05 |
| US8445474B2 (en) | 2013-05-21 |
| DK2026803T3 (da) | 2012-04-16 |
| CN101448498B (zh) | 2011-04-27 |
| EP2026803B1 (en) | 2011-12-21 |
| ES2379117T3 (es) | 2012-04-20 |
| WO2007137071A3 (en) | 2008-04-17 |
| EP2026803A2 (en) | 2009-02-25 |
| PL2026803T3 (pl) | 2012-05-31 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20090021169A (ko) | R(+) 및 s(-) 프라미펙솔을 포함하는 조성물 및 이의 사용 방법 | |
| EP2465500A1 (en) | Therapeutically effective amounts of R(+) and S(-) pramipexole for use in the treatment of parkinson's disease | |
| KR100849999B1 (ko) | 두통, 편두통, 구역질 및 구토를 위한 약학적 조성물 | |
| EP0687472A2 (en) | Potentiation of drug response by a serotonin 1A receptor antagonist | |
| CN102652135A (zh) | 脂肪酸酰胺水解酶的含氮杂环抑制剂 | |
| CA1157379A (en) | Method of preparation of pharmaceutical composition for treating peripheral orthostatic hypotention | |
| KR20070116632A (ko) | 프로스타글란딘 F2a 유도체를 유효 성분으로서 함유하는망막신경세포 보호제 | |
| NZ506020A (en) | Use of pharmaceutical combinations containing tramadol and an antiemetic | |
| US12403134B2 (en) | Method for improving or enhancing cognition | |
| JP2004508272A (ja) | アセチルコリンエンハンサー | |
| JP2011246478A (ja) | テルカゲパントカリウムの固形投与製剤 | |
| JP5376786B2 (ja) | 神経細胞賦活組成物 | |
| CN101433536A (zh) | 含有烟酸氨氯地平和沙坦类药物的治疗组合物 | |
| HK1170420A (en) | Therapeutically effective amounts of r(+) and s(-) pramipexole for use in the treatment of parkinson's disease | |
| EP0818198A1 (en) | Potentiation of drug response by increasing serotonin availability | |
| AU2006261296B2 (en) | Pharmaceutical composition comprising a 1-(3-chlorophenyl)-3-alkylpiperazine for treating apetite disorder | |
| JPWO2005000293A1 (ja) | 脳血管障害に伴う過活動膀胱治療剤 | |
| CN101420947A (zh) | 用于非恢复性睡眠的α-2-δ配体 | |
| JPH0827000A (ja) | 悪阻による諸症状の予防または治療薬 | |
| JP2003503452A (ja) | 治療剤 | |
| CA2497464A1 (en) | A therapeutic agent for improving hypogastric and/or perineal pains | |
| JPH0827001A (ja) | 術後嘔吐の予防または治療薬 | |
| HK1013797A (en) | Potentiation of drug response by a serotonin 1a receptor antagonist | |
| JP2001506642A (ja) | 糖尿病合併症の処置のための2−(2−モルホリノフェニル)グアニジン誘導体の使用 | |
| MXPA00012880A (en) | Agents with an antidepressive effect |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20081216 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20110211 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20130117 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20130329 Comment text: Decision to Refuse Application Patent event code: PE06012S01D Patent event date: 20130117 Comment text: Notification of reason for refusal Patent event code: PE06011S01I |