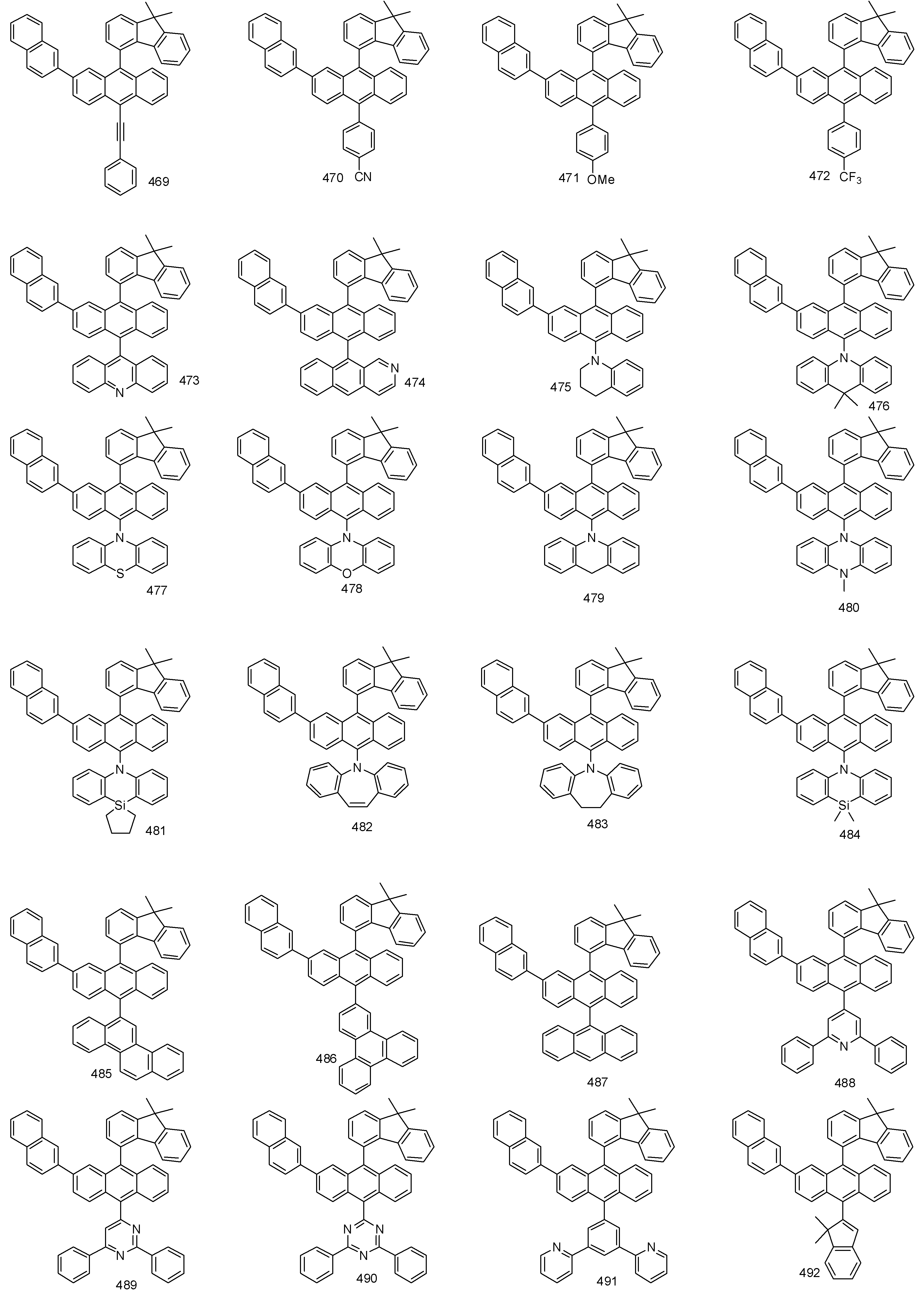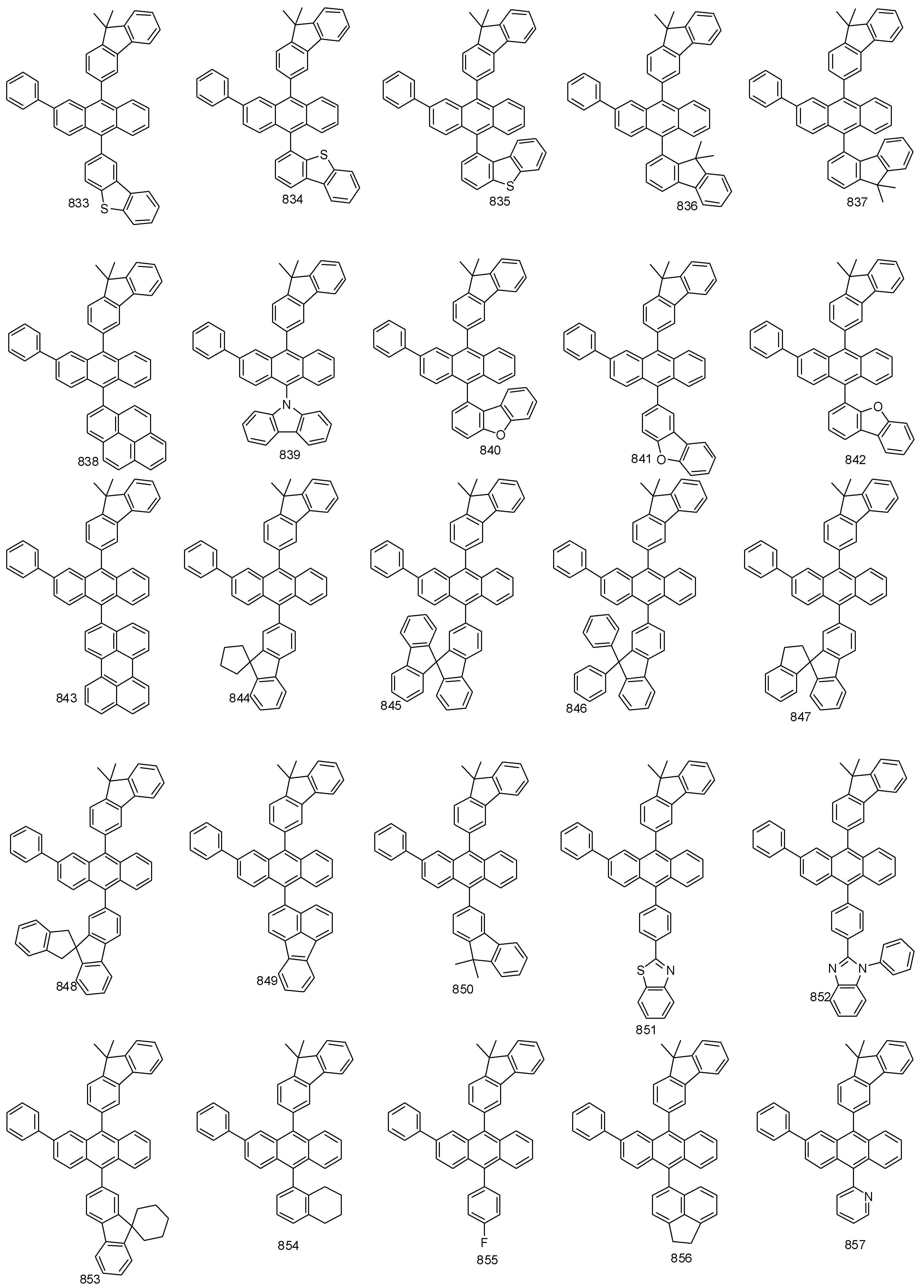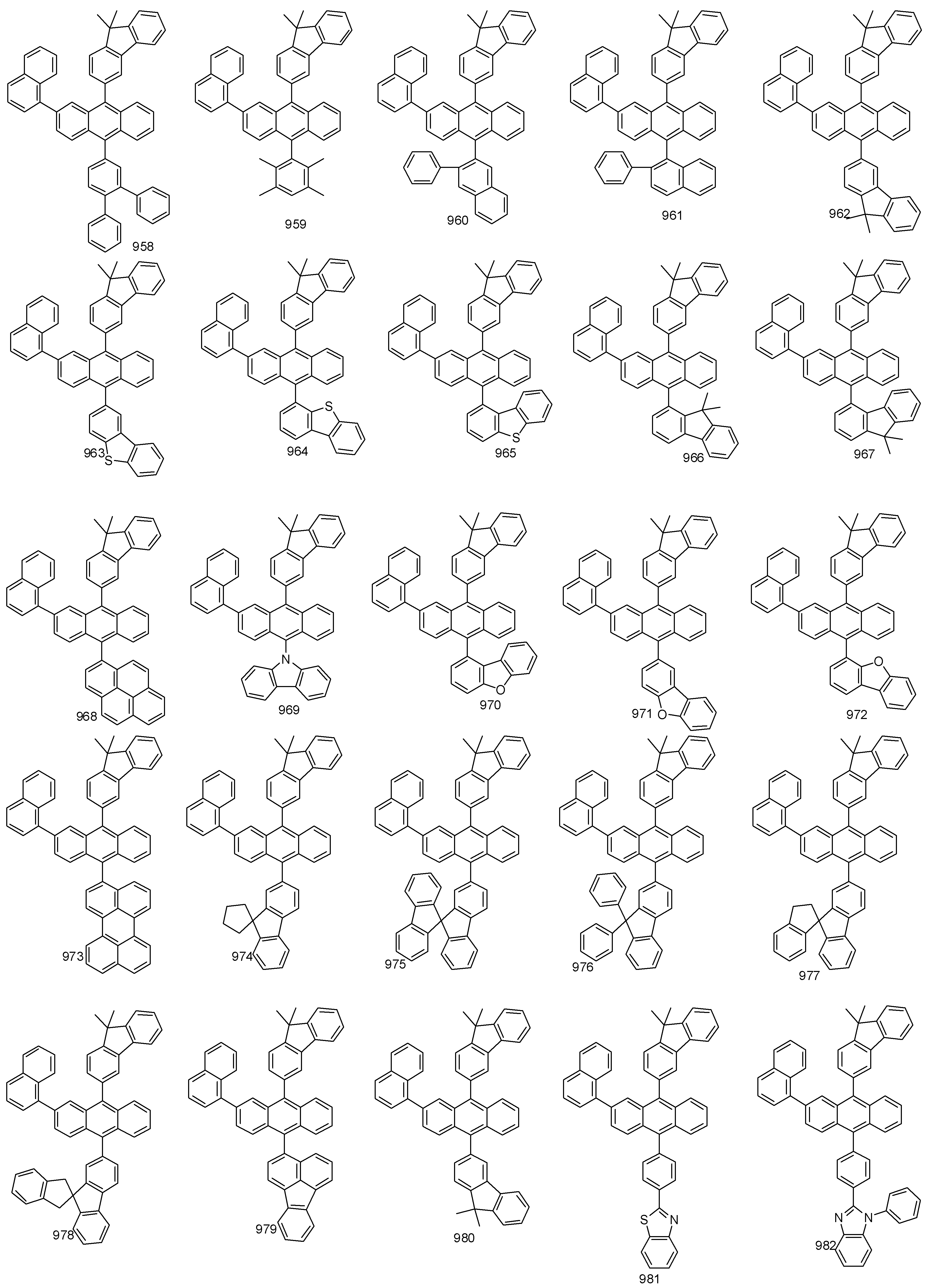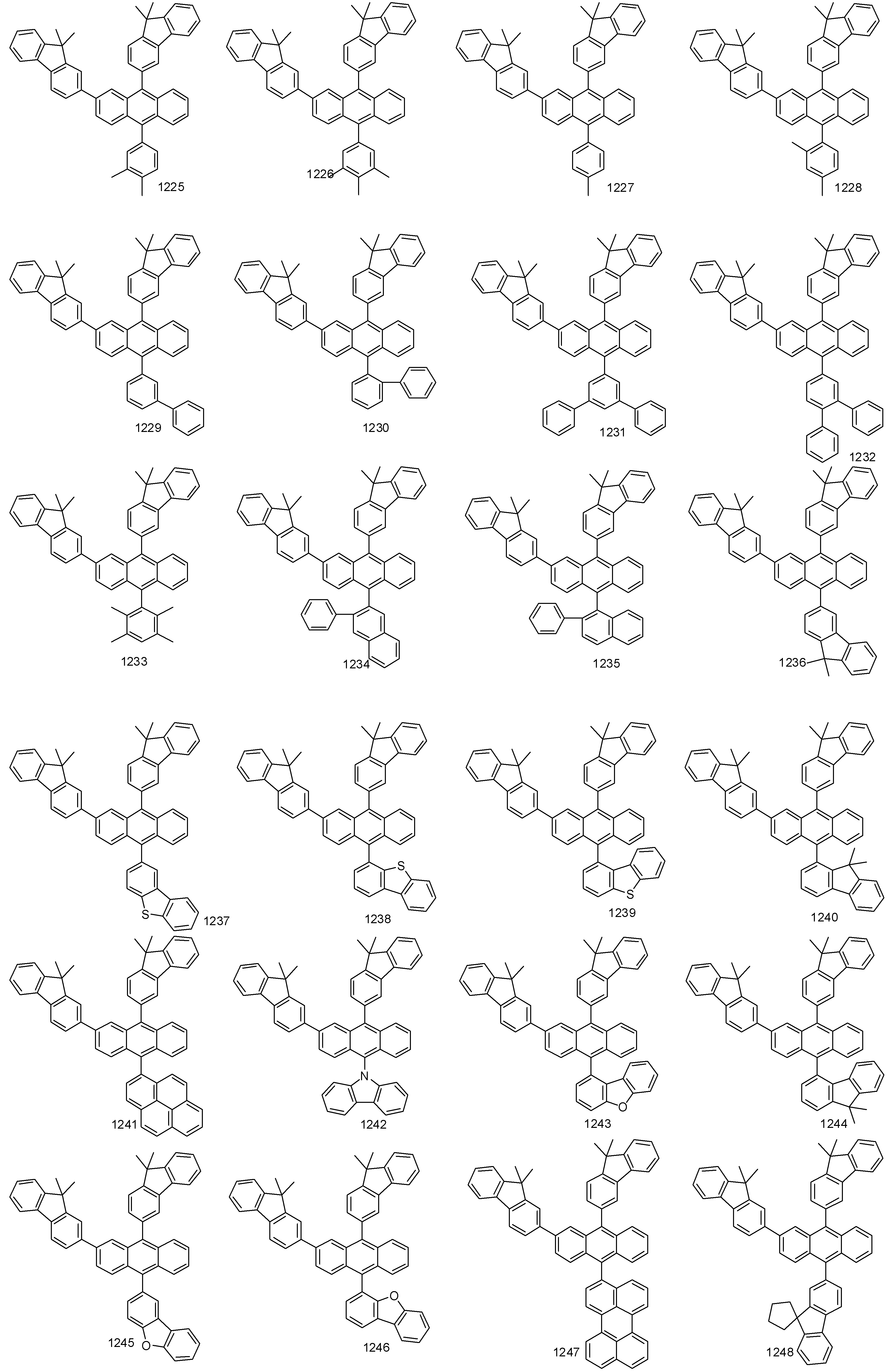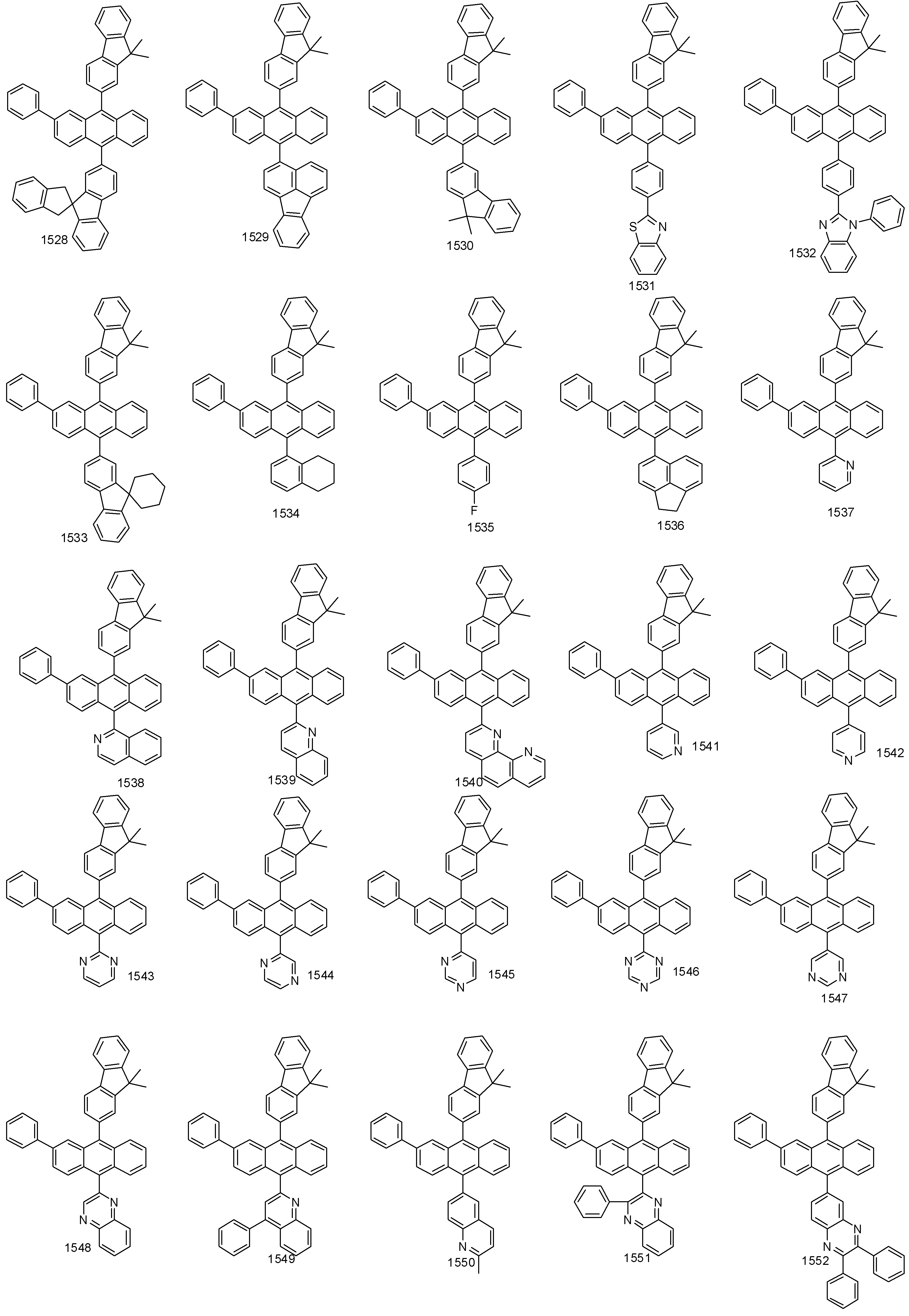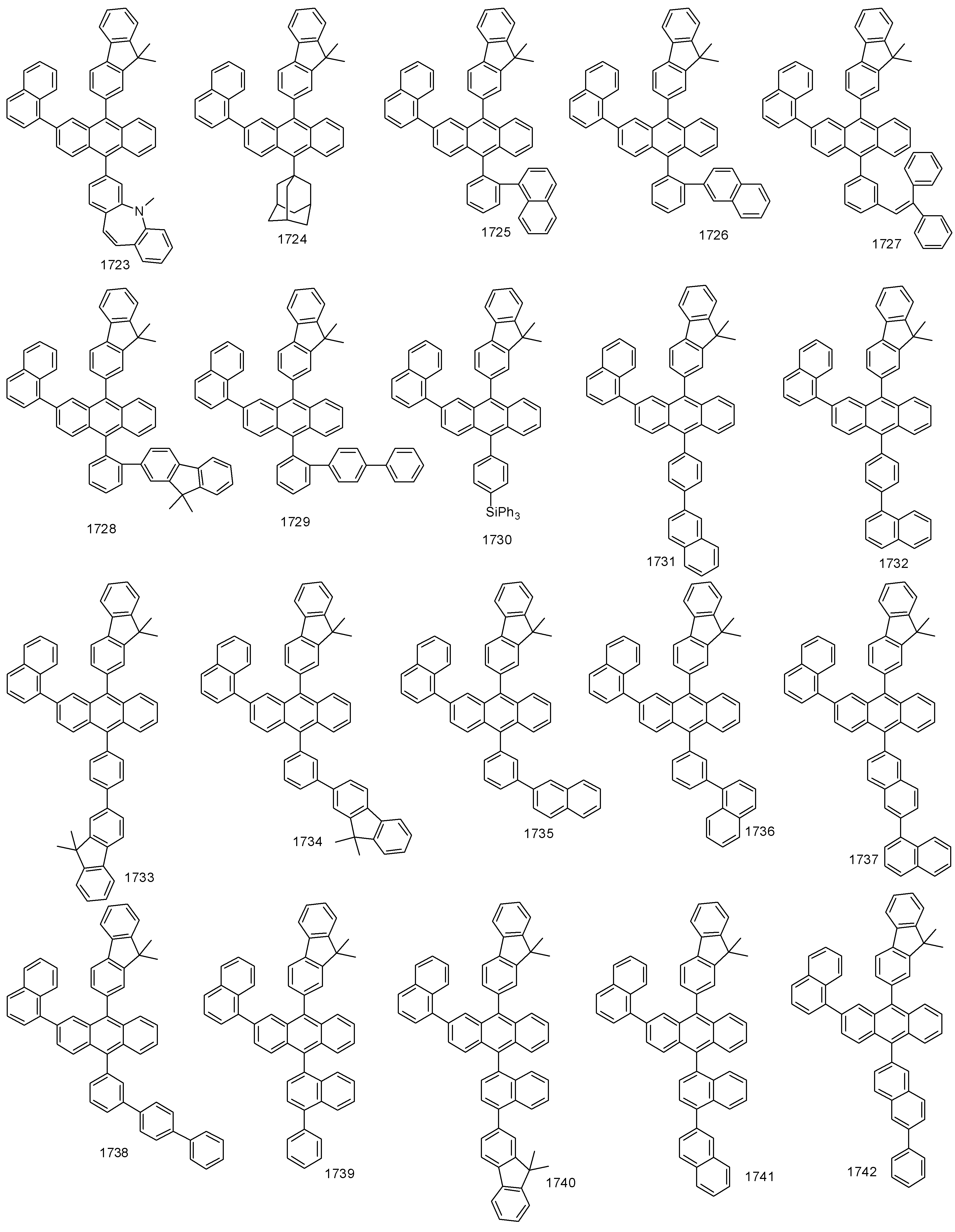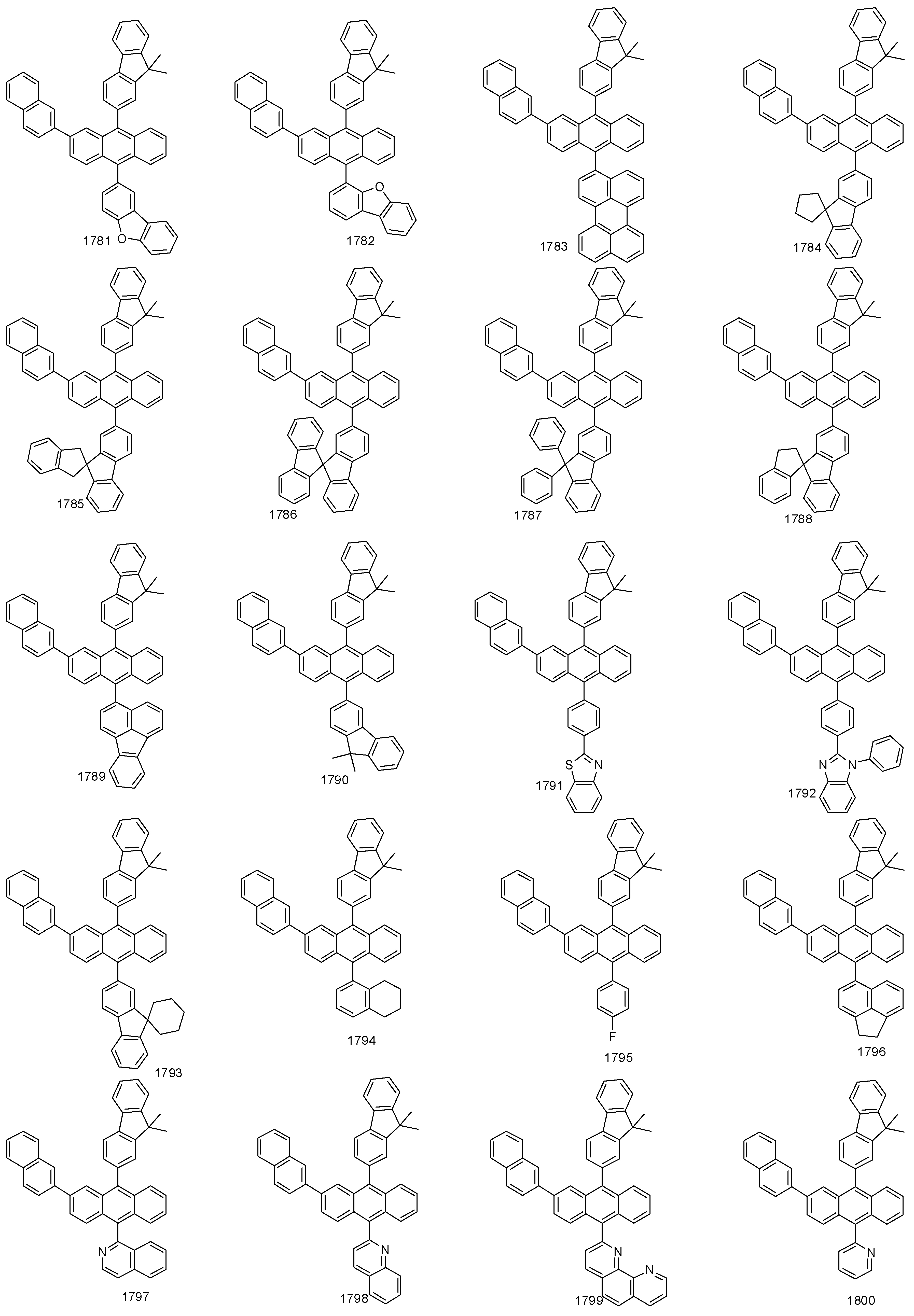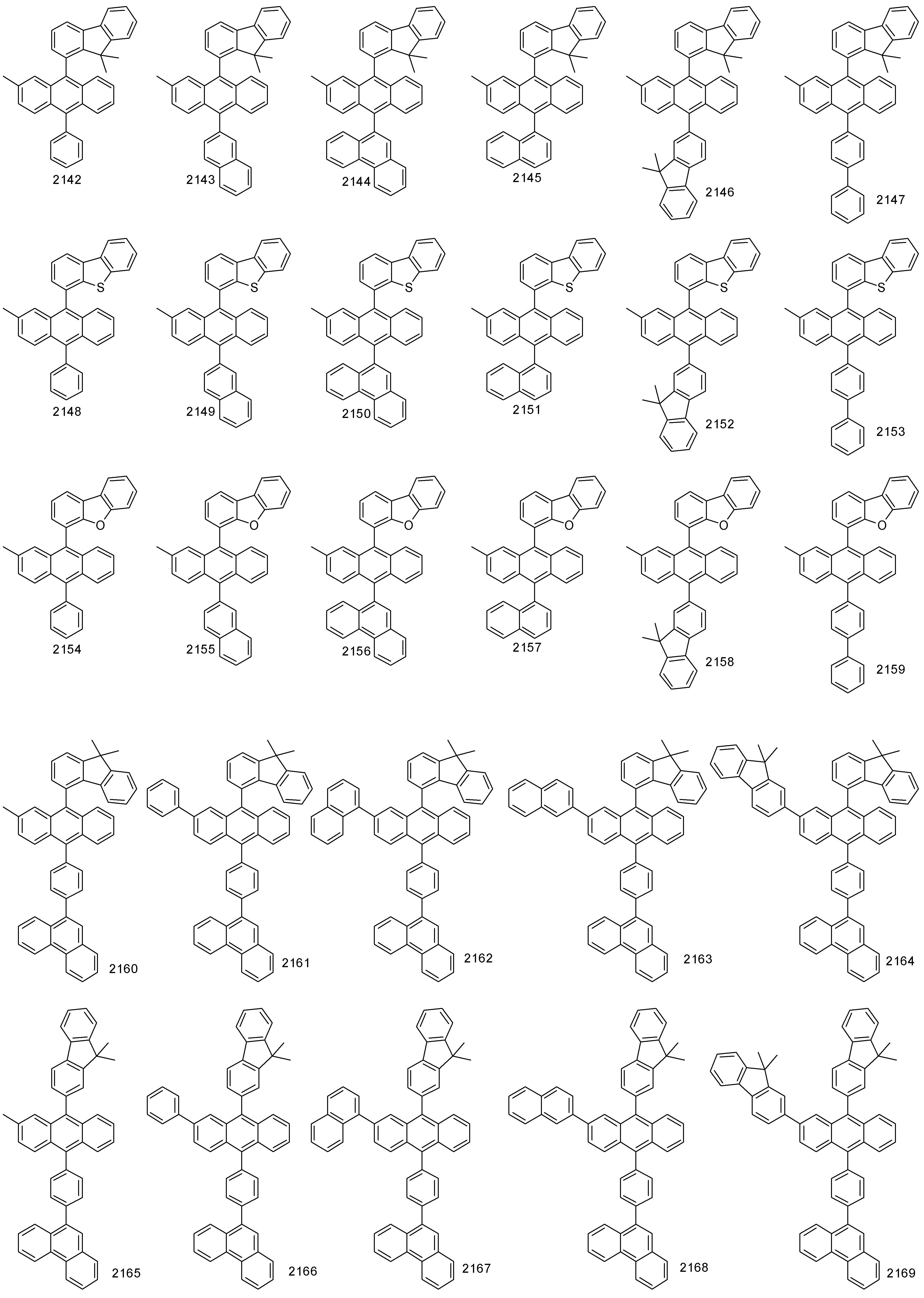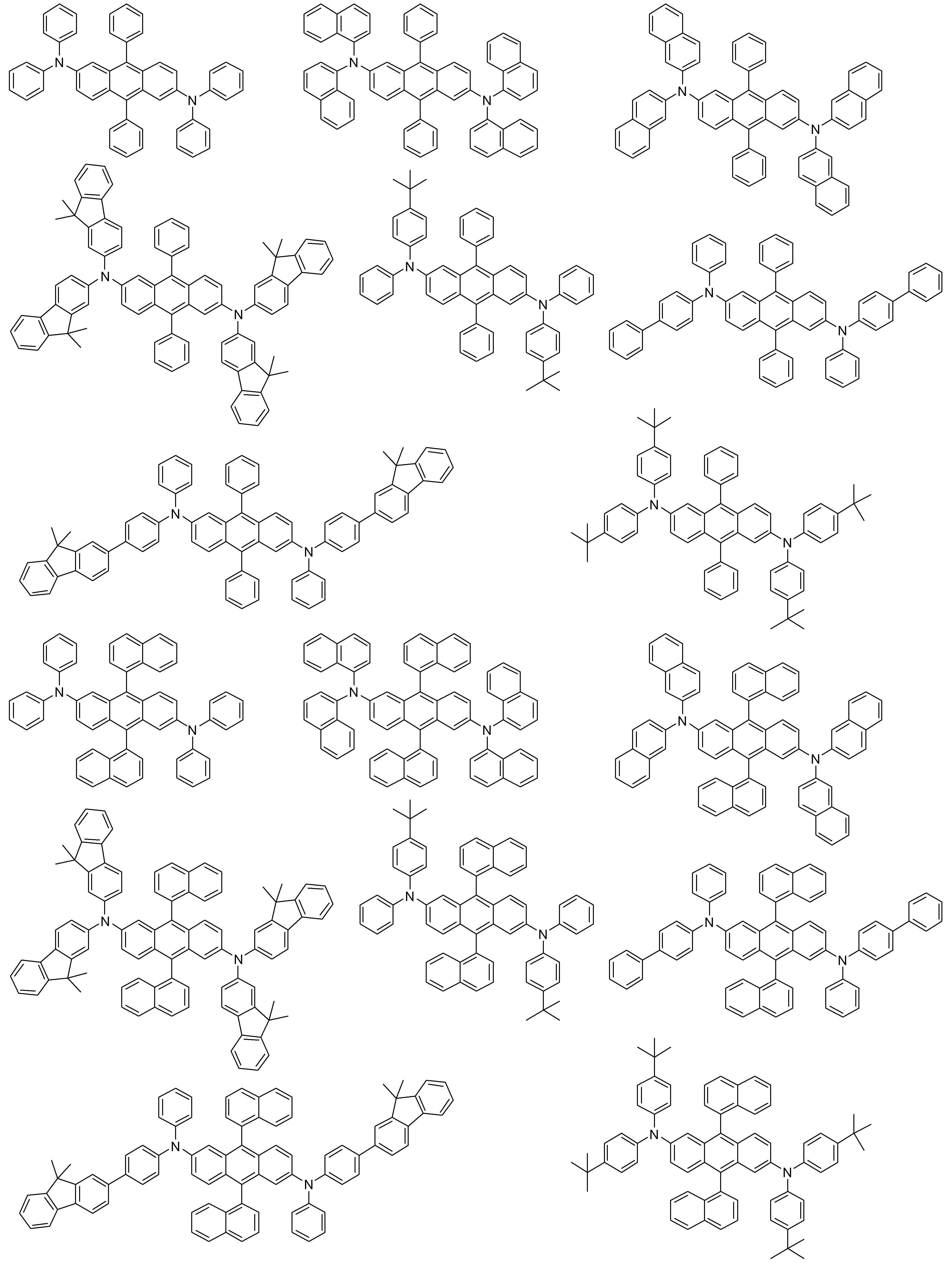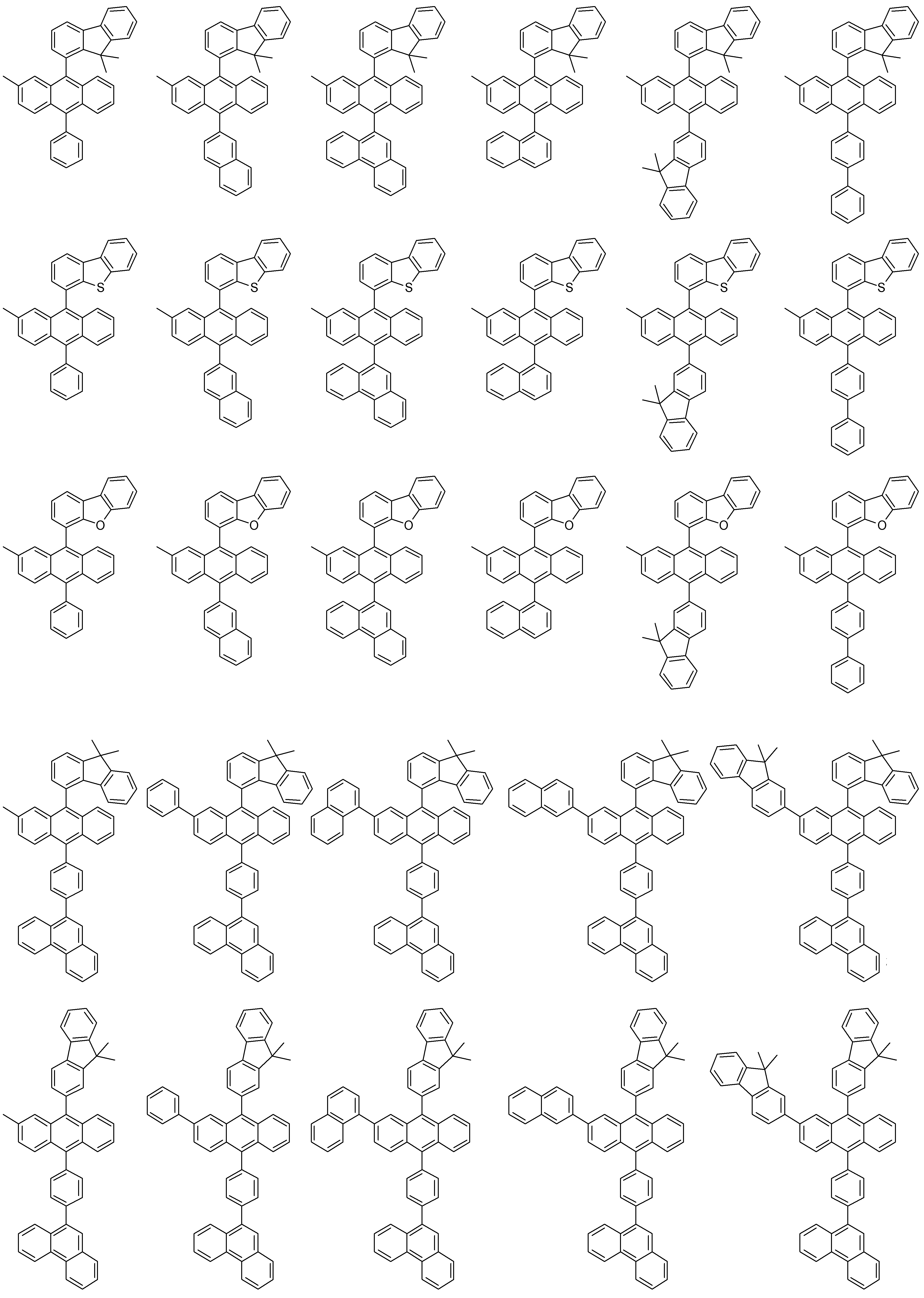KR100910150B1 - 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 발광 소자 - Google Patents
신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 발광 소자 Download PDFInfo
- Publication number
- KR100910150B1 KR100910150B1 KR1020080030645A KR20080030645A KR100910150B1 KR 100910150 B1 KR100910150 B1 KR 100910150B1 KR 1020080030645 A KR1020080030645 A KR 1020080030645A KR 20080030645 A KR20080030645 A KR 20080030645A KR 100910150 B1 KR100910150 B1 KR 100910150B1
- Authority
- KR
- South Korea
- Prior art keywords
- alkyl
- tri
- arylsilyl
- aryl
- light emitting
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 0 CC1(C)c2cc(-c3c(cccc4)c4c(-c(cc4)c(cccc5)c5c4-c4cc(C(*5CC5)=*=C)ccc4)c(cc4)c3cc4-c3ccccc3)ccc2-c2ccccc12 Chemical compound CC1(C)c2cc(-c3c(cccc4)c4c(-c(cc4)c(cccc5)c5c4-c4cc(C(*5CC5)=*=C)ccc4)c(cc4)c3cc4-c3ccccc3)ccc2-c2ccccc12 0.000 description 4
- ZTGPSUCDPFELFH-UHFFFAOYSA-N CC(C)(CCN1c2c3C(C)(C)CC1)c2cc1c3OCC(c2nc3ccccc3[s]2)=C1 Chemical compound CC(C)(CCN1c2c3C(C)(C)CC1)c2cc1c3OCC(c2nc3ccccc3[s]2)=C1 ZTGPSUCDPFELFH-UHFFFAOYSA-N 0.000 description 1
- OBCQHLPDJJDFOX-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2ccc(C)c1)c(cccc1)c1c2-c1nc2c3ncccc3ccc2cc1 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2ccc(C)c1)c(cccc1)c1c2-c1nc2c3ncccc3ccc2cc1 OBCQHLPDJJDFOX-UHFFFAOYSA-N 0.000 description 1
- IJYGDEHMYNAIKD-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2ccc(C)c1)c(cccc1)c1c2-c1nc2ccccc2nc1 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2ccc(C)c1)c(cccc1)c1c2-c1nc2ccccc2nc1 IJYGDEHMYNAIKD-UHFFFAOYSA-N 0.000 description 1
- PULYIPGULBLMAJ-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2-c1ccncn1 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2-c1ccncn1 PULYIPGULBLMAJ-UHFFFAOYSA-N 0.000 description 1
- RTMMRPQKEILAPI-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2-c1cnccn1 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2-c1cnccn1 RTMMRPQKEILAPI-UHFFFAOYSA-N 0.000 description 1
- FQWGUMPRVBUVDL-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2-c1nc2ccccc2nc1-c1ccccc1 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2-c1nc2ccccc2nc1-c1ccccc1 FQWGUMPRVBUVDL-UHFFFAOYSA-N 0.000 description 1
- MDISIMWTHSXLRW-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2-c1ncccn1 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2-c1ncccn1 MDISIMWTHSXLRW-UHFFFAOYSA-N 0.000 description 1
- QHPZNHXGPYERQO-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2-c1ncncn1 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2-c1ncncn1 QHPZNHXGPYERQO-UHFFFAOYSA-N 0.000 description 1
- ACSOTLCLYIUFMX-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2N1c2ccccc2Oc2c1cccc2 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2N1c2ccccc2Oc2c1cccc2 ACSOTLCLYIUFMX-UHFFFAOYSA-N 0.000 description 1
- SAXRZDVPGGWBGX-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2N1c2ccccc2Sc2c1cccc2 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c(c1c2cccc1)c(cc(C)cc1)c1c2N1c2ccccc2Sc2c1cccc2 SAXRZDVPGGWBGX-UHFFFAOYSA-N 0.000 description 1
- SGTUVORIDRRNQD-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c1c(cc(C)cc2)c2c(-c(cc2)ccc2OC)c2c1cccc2 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c1c(cc(C)cc2)c2c(-c(cc2)ccc2OC)c2c1cccc2 SGTUVORIDRRNQD-UHFFFAOYSA-N 0.000 description 1
- AIHOAMZQTVNSLT-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c1c(cc(C)cc2)c2c(-c2ccc(C(F)(F)F)cc2)c2c1cccc2 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c1c(cc(C)cc2)c2c(-c2ccc(C(F)(F)F)cc2)c2c1cccc2 AIHOAMZQTVNSLT-UHFFFAOYSA-N 0.000 description 1
- IKMXXCPJHBCRBK-UHFFFAOYSA-N CC(C)(c1ccccc1-c1c2)c1ccc2-c1c(cc(C)cc2)c2c(-c2cncnc2)c2c1cccc2 Chemical compound CC(C)(c1ccccc1-c1c2)c1ccc2-c1c(cc(C)cc2)c2c(-c2cncnc2)c2c1cccc2 IKMXXCPJHBCRBK-UHFFFAOYSA-N 0.000 description 1
- DDLCDCPOXYNHLR-UHFFFAOYSA-N CC(CC=C1)c2c1c(-c1cc3c(-c4ccc5-c6cc(-c(cc7)ccc7-c7nc(ccc(-c8c(ccc(-c9cccc%10c9cccc%10)c9)c9c(-c9ccc(C%10C=CC=CC%10(C)C%10(C)C)c%10c9)c9c8cccc9)c8)c8nc7-c7ccccc7)ccc6C(C)(C)c5c4)c(cccc4)c4c(-c4ccc(C(F)(F)F)cc4)c3cc1)ccc2 Chemical compound CC(CC=C1)c2c1c(-c1cc3c(-c4ccc5-c6cc(-c(cc7)ccc7-c7nc(ccc(-c8c(ccc(-c9cccc%10c9cccc%10)c9)c9c(-c9ccc(C%10C=CC=CC%10(C)C%10(C)C)c%10c9)c9c8cccc9)c8)c8nc7-c7ccccc7)ccc6C(C)(C)c5c4)c(cccc4)c4c(-c4ccc(C(F)(F)F)cc4)c3cc1)ccc2 DDLCDCPOXYNHLR-UHFFFAOYSA-N 0.000 description 1
- SRUGTFXBVJZRRC-UHFFFAOYSA-N CC1(C)c(cccc2)c2C(C=C2)=C1CC2C(c1cc(C(CC2)=CC3=C2c2ccccc2C3(C)C)ccc11)=C2C=CC=CC2C1c1nc2c3ncccc3ccc2cc1 Chemical compound CC1(C)c(cccc2)c2C(C=C2)=C1CC2C(c1cc(C(CC2)=CC3=C2c2ccccc2C3(C)C)ccc11)=C2C=CC=CC2C1c1nc2c3ncccc3ccc2cc1 SRUGTFXBVJZRRC-UHFFFAOYSA-N 0.000 description 1
- CQIOVHDKOFWHJU-UHFFFAOYSA-N CC1(C)c(cccc2)c2C(C=C2)=C1CC2c(c1c2cccc1)c(cc(cc1)-c3ccc4-c5ccccc5C(C)(C)c4c3)c1c2-c1ncncn1 Chemical compound CC1(C)c(cccc2)c2C(C=C2)=C1CC2c(c1c2cccc1)c(cc(cc1)-c3ccc4-c5ccccc5C(C)(C)c4c3)c1c2-c1ncncn1 CQIOVHDKOFWHJU-UHFFFAOYSA-N 0.000 description 1
- ZLUKJDNOBPTJEQ-UHFFFAOYSA-N CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5c(cccc6)c6ccc5)c3c4-c3ccncn3)ccc2-c2ccccc12 Chemical compound CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5c(cccc6)c6ccc5)c3c4-c3ccncn3)ccc2-c2ccccc12 ZLUKJDNOBPTJEQ-UHFFFAOYSA-N 0.000 description 1
- FETWUKMTECEKAX-UHFFFAOYSA-N CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5c(cccc6)c6ccc5)c3c4N3c4ccccc4Cc4c3cccc4)ccc2-c2ccccc12 Chemical compound CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5c(cccc6)c6ccc5)c3c4N3c4ccccc4Cc4c3cccc4)ccc2-c2ccccc12 FETWUKMTECEKAX-UHFFFAOYSA-N 0.000 description 1
- VDHMSSHNJXZHNN-UHFFFAOYSA-N CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4-c3cnc(cccc4)c4n3)ccc2-c2ccccc12 Chemical compound CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4-c3cnc(cccc4)c4n3)ccc2-c2ccccc12 VDHMSSHNJXZHNN-UHFFFAOYSA-N 0.000 description 1
- RYZJPSQXSWCLMF-UHFFFAOYSA-N CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4-c3cnccn3)ccc2-c2ccccc12 Chemical compound CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4-c3cnccn3)ccc2-c2ccccc12 RYZJPSQXSWCLMF-UHFFFAOYSA-N 0.000 description 1
- CCOWUEXLRBAACE-UHFFFAOYSA-N CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4-c3ncccn3)ccc2-c2ccccc12 Chemical compound CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4-c3ncccn3)ccc2-c2ccccc12 CCOWUEXLRBAACE-UHFFFAOYSA-N 0.000 description 1
- WZEWYXVPUOZNBU-UHFFFAOYSA-N CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4-c3ncncn3)ccc2-c2c1cccc2 Chemical compound CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4-c3ncncn3)ccc2-c2c1cccc2 WZEWYXVPUOZNBU-UHFFFAOYSA-N 0.000 description 1
- BIRMQNDSOHNJHT-UHFFFAOYSA-N CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4N(CCC3)c4c3cccc4)ccc2-c2ccccc12 Chemical compound CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4N(CCC3)c4c3cccc4)ccc2-c2ccccc12 BIRMQNDSOHNJHT-UHFFFAOYSA-N 0.000 description 1
- SNRACGBNBYMNQX-UHFFFAOYSA-N CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4N3c(cccc4)c4Oc4c3cccc4)ccc2-c2ccccc12 Chemical compound CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4N3c(cccc4)c4Oc4c3cccc4)ccc2-c2ccccc12 SNRACGBNBYMNQX-UHFFFAOYSA-N 0.000 description 1
- YJKZTCSUIRMEJO-UHFFFAOYSA-N CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4N3c4ccccc4Sc4c3cccc4)ccc2-c2c1cccc2 Chemical compound CC1(C)c2cc(-c(c3c4cccc3)c(cc(cc3)-c5cccc6c5cccc6)c3c4N3c4ccccc4Sc4c3cccc4)ccc2-c2c1cccc2 YJKZTCSUIRMEJO-UHFFFAOYSA-N 0.000 description 1
- NUTRBGOABWWSGV-UHFFFAOYSA-N CC1(C)c2cc(-c3c(cc(cc4)-c5c(cccc6)c6ccc5)c4c(-c4cncnc4)c4c3cccc4)ccc2-c2ccccc12 Chemical compound CC1(C)c2cc(-c3c(cc(cc4)-c5c(cccc6)c6ccc5)c4c(-c4cncnc4)c4c3cccc4)ccc2-c2ccccc12 NUTRBGOABWWSGV-UHFFFAOYSA-N 0.000 description 1
- TWJSEBOFSMWGKK-UHFFFAOYSA-N CC1(C)c2cc(-c3c(cc(cc4)-c5cccc6c5cccc6)c4c(-c(cc4)cc5c4nc(C)cc5)c4c3cccc4)ccc2-c2ccccc12 Chemical compound CC1(C)c2cc(-c3c(cc(cc4)-c5cccc6c5cccc6)c4c(-c(cc4)cc5c4nc(C)cc5)c4c3cccc4)ccc2-c2ccccc12 TWJSEBOFSMWGKK-UHFFFAOYSA-N 0.000 description 1
- WGLVHOJBBNOKKP-UHFFFAOYSA-N CC1(C)c2cc(-c3c(cc(cc4)-c5cccc6c5cccc6)c4c(-c(cc4)ccc4OC)c4c3cccc4)ccc2-c2ccccc12 Chemical compound CC1(C)c2cc(-c3c(cc(cc4)-c5cccc6c5cccc6)c4c(-c(cc4)ccc4OC)c4c3cccc4)ccc2-c2ccccc12 WGLVHOJBBNOKKP-UHFFFAOYSA-N 0.000 description 1
- DANQPALPCPKJJA-UHFFFAOYSA-N CC1C(c(c(C2(C)C)c3)ccc3-c3cc4c(C(C5)C=CC6=C5C(C)(C)c5c6cccc5)c(CCC=C5)c5c(-c5cnccn5)c4cc3)=C2C=CC1 Chemical compound CC1C(c(c(C2(C)C)c3)ccc3-c3cc4c(C(C5)C=CC6=C5C(C)(C)c5c6cccc5)c(CCC=C5)c5c(-c5cnccn5)c4cc3)=C2C=CC1 DANQPALPCPKJJA-UHFFFAOYSA-N 0.000 description 1
- GIZCIFKXNWIZSR-UHFFFAOYSA-N CC1C(c(c(C2(C)C)c3)ccc3-c3cc4c(C(C5)C=CC6=C5C(C)(C)c5c6cccc5)c(cccc5)c5c(-c5ccncn5)c4cc3)=C2C=CC1 Chemical compound CC1C(c(c(C2(C)C)c3)ccc3-c3cc4c(C(C5)C=CC6=C5C(C)(C)c5c6cccc5)c(cccc5)c5c(-c5ccncn5)c4cc3)=C2C=CC1 GIZCIFKXNWIZSR-UHFFFAOYSA-N 0.000 description 1
- HGZIZCPJTDJLLS-UHFFFAOYSA-N Cc(cc1)cc2c1c(-c1c(cccc3)c3ccc1)c(cccc1)c1c2-c1cccc2c1c(cccc1)c1[s]2 Chemical compound Cc(cc1)cc2c1c(-c1c(cccc3)c3ccc1)c(cccc1)c1c2-c1cccc2c1c(cccc1)c1[s]2 HGZIZCPJTDJLLS-UHFFFAOYSA-N 0.000 description 1
- OVYRQHVPLGYYCL-UHFFFAOYSA-N Cc1cc2c(-c3ccc4[s]c(cccc5)c5c4c3)c(cccc3)c3c(-c3cc4ccccc4cc3)c2cc1 Chemical compound Cc1cc2c(-c3ccc4[s]c(cccc5)c5c4c3)c(cccc3)c3c(-c3cc4ccccc4cc3)c2cc1 OVYRQHVPLGYYCL-UHFFFAOYSA-N 0.000 description 1
- UNFBVDQXZKVOGP-UHFFFAOYSA-N Cc1cc2c(-c3ccc4[s]c(cccc5)c5c4c3)c(cccc3)c3c(-c3ccccc3)c2cc1 Chemical compound Cc1cc2c(-c3ccc4[s]c(cccc5)c5c4c3)c(cccc3)c3c(-c3ccccc3)c2cc1 UNFBVDQXZKVOGP-UHFFFAOYSA-N 0.000 description 1
- ODGDTACYMBDFRO-UHFFFAOYSA-N Cc1cc2c(-c3ccc4[s]c5ccccc5c4c3)c(cccc3)c3c(-c3cccc4ccccc34)c2cc1 Chemical compound Cc1cc2c(-c3ccc4[s]c5ccccc5c4c3)c(cccc3)c3c(-c3cccc4ccccc34)c2cc1 ODGDTACYMBDFRO-UHFFFAOYSA-N 0.000 description 1
- YZGQDYVYDZCHOO-UHFFFAOYSA-N Cc1cc2c(-c3cccc4c3c(cccc3)c3[o]4)c(cccc3)c3c(-c3cccc4ccccc34)c2cc1 Chemical compound Cc1cc2c(-c3cccc4c3c(cccc3)c3[o]4)c(cccc3)c3c(-c3cccc4ccccc34)c2cc1 YZGQDYVYDZCHOO-UHFFFAOYSA-N 0.000 description 1
- VCXCMDIJTOVAHL-UHFFFAOYSA-N Cc1cc2c(-c3cccc4c3c3ccccc3[o]4)c(cccc3)c3c(-c3cc4ccccc4c4c3cccc4)c2cc1 Chemical compound Cc1cc2c(-c3cccc4c3c3ccccc3[o]4)c(cccc3)c3c(-c3cc4ccccc4c4c3cccc4)c2cc1 VCXCMDIJTOVAHL-UHFFFAOYSA-N 0.000 description 1
- ITXYOOMHVVFEKI-UHFFFAOYSA-N Cc1cc2c(-c3cccc4c3c3ccccc3[o]4)c(cccc3)c3c(-c3cc4ccccc4cc3)c2cc1 Chemical compound Cc1cc2c(-c3cccc4c3c3ccccc3[o]4)c(cccc3)c3c(-c3cc4ccccc4cc3)c2cc1 ITXYOOMHVVFEKI-UHFFFAOYSA-N 0.000 description 1
- GBQDZVNQDSURNZ-UHFFFAOYSA-N Cc1cc2c(-c3cccc4c3c3ccccc3[o]4)c(cccc3)c3c(-c3ccccc3)c2cc1 Chemical compound Cc1cc2c(-c3cccc4c3c3ccccc3[o]4)c(cccc3)c3c(-c3ccccc3)c2cc1 GBQDZVNQDSURNZ-UHFFFAOYSA-N 0.000 description 1
- BWVKLRMRXJOSAZ-UHFFFAOYSA-N Cc1cc2c(-c3cccc4c3c3ccccc3[s]4)c(cccc3)c3c(-c3cc4ccccc4c4c3cccc4)c2cc1 Chemical compound Cc1cc2c(-c3cccc4c3c3ccccc3[s]4)c(cccc3)c3c(-c3cc4ccccc4c4c3cccc4)c2cc1 BWVKLRMRXJOSAZ-UHFFFAOYSA-N 0.000 description 1
- LSYHMRROXSNTHJ-UHFFFAOYSA-N Cc1cc2c(-c3cccc4c3c3ccccc3[s]4)c(cccc3)c3c(-c3cc4ccccc4cc3)c2cc1 Chemical compound Cc1cc2c(-c3cccc4c3c3ccccc3[s]4)c(cccc3)c3c(-c3cc4ccccc4cc3)c2cc1 LSYHMRROXSNTHJ-UHFFFAOYSA-N 0.000 description 1
- AMNPDNFSYCKRTP-UHFFFAOYSA-N c(cc1)ccc1-c(cc1)cc2c1c(-c1ccc-3c4c1cccc4-c1cccc4c1c-3ccc4-c1ccc(-c(cc3)c45)c6c1cccc6-c4cccc5c3-c1c(cccc3)c3c(-c(cc3)c(cccc4-5)c4c3-c3c4c-5cccc4ccc3)c(cc3)c1cc3-c1cc(cccc3)c3cc1)c(cccc1)c1c2-c(cc1)c(cccc2-3)c2c1-c1cccc2c1c-3ccc2 Chemical compound c(cc1)ccc1-c(cc1)cc2c1c(-c1ccc-3c4c1cccc4-c1cccc4c1c-3ccc4-c1ccc(-c(cc3)c45)c6c1cccc6-c4cccc5c3-c1c(cccc3)c3c(-c(cc3)c(cccc4-5)c4c3-c3c4c-5cccc4ccc3)c(cc3)c1cc3-c1cc(cccc3)c3cc1)c(cccc1)c1c2-c(cc1)c(cccc2-3)c2c1-c1cccc2c1c-3ccc2 AMNPDNFSYCKRTP-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/54—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with three condensed rings
- C07C13/547—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with three condensed rings at least one ring not being six-membered, the other rings being at the most six-membered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/54—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with three condensed rings
- C07C13/547—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with three condensed rings at least one ring not being six-membered, the other rings being at the most six-membered
- C07C13/567—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with three condensed rings at least one ring not being six-membered, the other rings being at the most six-membered with a fluorene or hydrogenated fluorene ring system
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/54—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with three condensed rings
- C07C13/605—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with three condensed rings with a bridged ring system
- C07C13/615—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with three condensed rings with a bridged ring system with an adamantane ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/62—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with more than three condensed rings
- C07C13/66—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with more than three condensed rings the condensed ring system contains only four rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/72—Spiro hydrocarbons
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C211/00—Compounds containing amino groups bound to a carbon skeleton
- C07C211/43—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
- C07C211/57—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings being part of condensed ring systems of the carbon skeleton
- C07C211/58—Naphthylamines; N-substituted derivatives thereof
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C211/00—Compounds containing amino groups bound to a carbon skeleton
- C07C211/43—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
- C07C211/57—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings being part of condensed ring systems of the carbon skeleton
- C07C211/61—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings being part of condensed ring systems of the carbon skeleton with at least one of the condensed ring systems formed by three or more rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C22/00—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom
- C07C22/02—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom having unsaturation in the rings
- C07C22/04—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom having unsaturation in the rings containing six-membered aromatic rings
- C07C22/08—Cyclic compounds containing halogen atoms bound to an acyclic carbon atom having unsaturation in the rings containing six-membered aromatic rings containing fluorine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C25/00—Compounds containing at least one halogen atom bound to a six-membered aromatic ring
- C07C25/18—Polycyclic aromatic halogenated hydrocarbons
- C07C25/22—Polycyclic aromatic halogenated hydrocarbons with condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C255/00—Carboxylic acid nitriles
- C07C255/49—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton
- C07C255/50—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton to carbon atoms of non-condensed six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C43/00—Ethers; Compounds having groups, groups or groups
- C07C43/02—Ethers
- C07C43/20—Ethers having an ether-oxygen atom bound to a carbon atom of a six-membered aromatic ring
- C07C43/21—Ethers having an ether-oxygen atom bound to a carbon atom of a six-membered aromatic ring containing rings other than six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D209/00—Heterocyclic compounds containing five-membered rings, condensed with other rings, with one nitrogen atom as the only ring hetero atom
- C07D209/56—Ring systems containing three or more rings
- C07D209/80—[b, c]- or [b, d]-condensed
- C07D209/82—Carbazoles; Hydrogenated carbazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/06—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom containing only hydrogen and carbon atoms in addition to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D215/00—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems
- C07D215/02—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom
- C07D215/04—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, directly attached to the ring carbon atoms
- C07D215/06—Heterocyclic compounds containing quinoline or hydrogenated quinoline ring systems having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen atoms or carbon atoms directly attached to the ring nitrogen atom with only hydrogen atoms or radicals containing only hydrogen and carbon atoms, directly attached to the ring carbon atoms having only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D217/00—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems
- C07D217/02—Heterocyclic compounds containing isoquinoline or hydrogenated isoquinoline ring systems with only hydrogen atoms or radicals containing only carbon and hydrogen atoms, directly attached to carbon atoms of the nitrogen-containing ring; Alkylene-bis-isoquinolines
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D223/00—Heterocyclic compounds containing seven-membered rings having one nitrogen atom as the only ring hetero atom
- C07D223/14—Heterocyclic compounds containing seven-membered rings having one nitrogen atom as the only ring hetero atom condensed with carbocyclic rings or ring systems
- C07D223/18—Dibenzazepines; Hydrogenated dibenzazepines
- C07D223/22—Dibenz [b, f] azepines; Hydrogenated dibenz [b, f] azepines
- C07D223/24—Dibenz [b, f] azepines; Hydrogenated dibenz [b, f] azepines with hydrocarbon radicals, substituted by nitrogen atoms, attached to the ring nitrogen atom
- C07D223/26—Dibenz [b, f] azepines; Hydrogenated dibenz [b, f] azepines with hydrocarbon radicals, substituted by nitrogen atoms, attached to the ring nitrogen atom having a double bond between positions 10 and 11
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D223/00—Heterocyclic compounds containing seven-membered rings having one nitrogen atom as the only ring hetero atom
- C07D223/14—Heterocyclic compounds containing seven-membered rings having one nitrogen atom as the only ring hetero atom condensed with carbocyclic rings or ring systems
- C07D223/18—Dibenzazepines; Hydrogenated dibenzazepines
- C07D223/22—Dibenz [b, f] azepines; Hydrogenated dibenz [b, f] azepines
- C07D223/24—Dibenz [b, f] azepines; Hydrogenated dibenz [b, f] azepines with hydrocarbon radicals, substituted by nitrogen atoms, attached to the ring nitrogen atom
- C07D223/28—Dibenz [b, f] azepines; Hydrogenated dibenz [b, f] azepines with hydrocarbon radicals, substituted by nitrogen atoms, attached to the ring nitrogen atom having a single bond between positions 10 and 11
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D235/00—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, condensed with other rings
- C07D235/02—Heterocyclic compounds containing 1,3-diazole or hydrogenated 1,3-diazole rings, condensed with other rings condensed with carbocyclic rings or ring systems
- C07D235/04—Benzimidazoles; Hydrogenated benzimidazoles
- C07D235/18—Benzimidazoles; Hydrogenated benzimidazoles with aryl radicals directly attached in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D239/00—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings
- C07D239/02—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings
- C07D239/24—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members
- C07D239/26—Heterocyclic compounds containing 1,3-diazine or hydrogenated 1,3-diazine rings not condensed with other rings having three or more double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/02—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings
- C07D241/10—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members
- C07D241/12—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to ring carbon atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/36—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings condensed with carbocyclic rings or ring systems
- C07D241/38—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings condensed with carbocyclic rings or ring systems with only hydrogen or carbon atoms directly attached to the ring nitrogen atoms
- C07D241/40—Benzopyrazines
- C07D241/42—Benzopyrazines with only hydrogen atoms, hydrocarbon or substituted hydrocarbon radicals, directly attached to carbon atoms of the hetero ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D241/00—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings
- C07D241/36—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings condensed with carbocyclic rings or ring systems
- C07D241/38—Heterocyclic compounds containing 1,4-diazine or hydrogenated 1,4-diazine rings condensed with carbocyclic rings or ring systems with only hydrogen or carbon atoms directly attached to the ring nitrogen atoms
- C07D241/46—Phenazines
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D251/00—Heterocyclic compounds containing 1,3,5-triazine rings
- C07D251/02—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings
- C07D251/12—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members
- C07D251/14—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hydrogen or carbon atoms directly attached to at least one ring carbon atom
- C07D251/16—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hydrogen or carbon atoms directly attached to at least one ring carbon atom to only one ring carbon atom
- C07D251/20—Heterocyclic compounds containing 1,3,5-triazine rings not condensed with other rings having three double bonds between ring members or between ring members and non-ring members with hydrogen or carbon atoms directly attached to at least one ring carbon atom to only one ring carbon atom with no nitrogen atoms directly attached to a ring carbon atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D265/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one oxygen atom as the only ring hetero atoms
- C07D265/28—1,4-Oxazines; Hydrogenated 1,4-oxazines
- C07D265/34—1,4-Oxazines; Hydrogenated 1,4-oxazines condensed with carbocyclic rings
- C07D265/38—[b, e]-condensed with two six-membered rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D277/00—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings
- C07D277/60—Heterocyclic compounds containing 1,3-thiazole or hydrogenated 1,3-thiazole rings condensed with carbocyclic rings or ring systems
- C07D277/62—Benzothiazoles
- C07D277/64—Benzothiazoles with only hydrocarbon or substituted hydrocarbon radicals attached in position 2
- C07D277/66—Benzothiazoles with only hydrocarbon or substituted hydrocarbon radicals attached in position 2 with aromatic rings or ring systems directly attached in position 2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D279/00—Heterocyclic compounds containing six-membered rings having one nitrogen atom and one sulfur atom as the only ring hetero atoms
- C07D279/10—1,4-Thiazines; Hydrogenated 1,4-thiazines
- C07D279/14—1,4-Thiazines; Hydrogenated 1,4-thiazines condensed with carbocyclic rings or ring systems
- C07D279/18—[b, e]-condensed with two six-membered rings
- C07D279/22—[b, e]-condensed with two six-membered rings with carbon atoms directly attached to the ring nitrogen atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D311/00—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings
- C07D311/02—Heterocyclic compounds containing six-membered rings having one oxygen atom as the only hetero atom, condensed with other rings ortho- or peri-condensed with carbocyclic rings or ring systems
- C07D311/78—Ring systems having three or more relevant rings
- C07D311/80—Dibenzopyrans; Hydrogenated dibenzopyrans
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D333/00—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom
- C07D333/50—Heterocyclic compounds containing five-membered rings having one sulfur atom as the only ring hetero atom condensed with carbocyclic rings or ring systems
- C07D333/76—Dibenzothiophenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/0805—Compounds with Si-C or Si-Si linkages comprising only Si, C or H atoms
- C07F7/0807—Compounds with Si-C or Si-Si linkages comprising only Si, C or H atoms comprising Si as a ring atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/081—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te
- C07F7/0812—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring
- C07F7/0816—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring said ring comprising Si as a ring atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6564—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having phosphorus atoms, with or without nitrogen, oxygen, sulfur, selenium or tellurium atoms, as ring hetero atoms
- C07F9/6568—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having phosphorus atoms, with or without nitrogen, oxygen, sulfur, selenium or tellurium atoms, as ring hetero atoms having phosphorus atoms as the only ring hetero atoms
- C07F9/65683—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having phosphorus atoms, with or without nitrogen, oxygen, sulfur, selenium or tellurium atoms, as ring hetero atoms having phosphorus atoms as the only ring hetero atoms the ring phosphorus atom being part of a phosphine
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/12—Light sources with substantially two-dimensional radiating surfaces
- H05B33/14—Light sources with substantially two-dimensional radiating surfaces characterised by the chemical or physical composition or the arrangement of the electroluminescent material, or by the simultaneous addition of the electroluminescent material in or onto the light source
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/626—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing more than one polycyclic condensed aromatic rings, e.g. bis-anthracene
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2602/00—Systems containing two condensed rings
- C07C2602/02—Systems containing two condensed rings the rings having only two atoms in common
- C07C2602/04—One of the condensed rings being a six-membered aromatic ring
- C07C2602/08—One of the condensed rings being a six-membered aromatic ring the other ring being five-membered, e.g. indane
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2602/00—Systems containing two condensed rings
- C07C2602/02—Systems containing two condensed rings the rings having only two atoms in common
- C07C2602/04—One of the condensed rings being a six-membered aromatic ring
- C07C2602/10—One of the condensed rings being a six-membered aromatic ring the other ring being six-membered, e.g. tetraline
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/04—Ortho- or ortho- and peri-condensed systems containing three rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/04—Ortho- or ortho- and peri-condensed systems containing three rings
- C07C2603/06—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members
- C07C2603/10—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members containing five-membered rings
- C07C2603/12—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members containing five-membered rings only one five-membered ring
- C07C2603/18—Fluorenes; Hydrogenated fluorenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/04—Ortho- or ortho- and peri-condensed systems containing three rings
- C07C2603/06—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members
- C07C2603/10—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members containing five-membered rings
- C07C2603/12—Ortho- or ortho- and peri-condensed systems containing three rings containing at least one ring with less than six ring members containing five-membered rings only one five-membered ring
- C07C2603/20—Acenaphthenes; Hydrogenated acenaphthenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/04—Ortho- or ortho- and peri-condensed systems containing three rings
- C07C2603/22—Ortho- or ortho- and peri-condensed systems containing three rings containing only six-membered rings
- C07C2603/24—Anthracenes; Hydrogenated anthracenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/04—Ortho- or ortho- and peri-condensed systems containing three rings
- C07C2603/22—Ortho- or ortho- and peri-condensed systems containing three rings containing only six-membered rings
- C07C2603/26—Phenanthrenes; Hydrogenated phenanthrenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/40—Ortho- or ortho- and peri-condensed systems containing four condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/40—Ortho- or ortho- and peri-condensed systems containing four condensed rings
- C07C2603/42—Ortho- or ortho- and peri-condensed systems containing four condensed rings containing only six-membered rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/40—Ortho- or ortho- and peri-condensed systems containing four condensed rings
- C07C2603/42—Ortho- or ortho- and peri-condensed systems containing four condensed rings containing only six-membered rings
- C07C2603/48—Chrysenes; Hydrogenated chrysenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/40—Ortho- or ortho- and peri-condensed systems containing four condensed rings
- C07C2603/42—Ortho- or ortho- and peri-condensed systems containing four condensed rings containing only six-membered rings
- C07C2603/50—Pyrenes; Hydrogenated pyrenes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/52—Ortho- or ortho- and peri-condensed systems containing five condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/56—Ring systems containing bridged rings
- C07C2603/58—Ring systems containing bridged rings containing three rings
- C07C2603/70—Ring systems containing bridged rings containing three rings containing only six-membered rings
- C07C2603/74—Adamantanes
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/93—Spiro compounds
- C07C2603/94—Spiro compounds containing "free" spiro atoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1011—Condensed systems
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/549—Organic PV cells
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biochemistry (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Physics & Mathematics (AREA)
- Health & Medical Sciences (AREA)
- Electroluminescent Light Sources (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Indole Compounds (AREA)
- Other In-Based Heterocyclic Compounds (AREA)
- Furan Compounds (AREA)
- Pyridine Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
- Quinoline Compounds (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
- Thiazole And Isothizaole Compounds (AREA)
Abstract
Description
Claims (19)
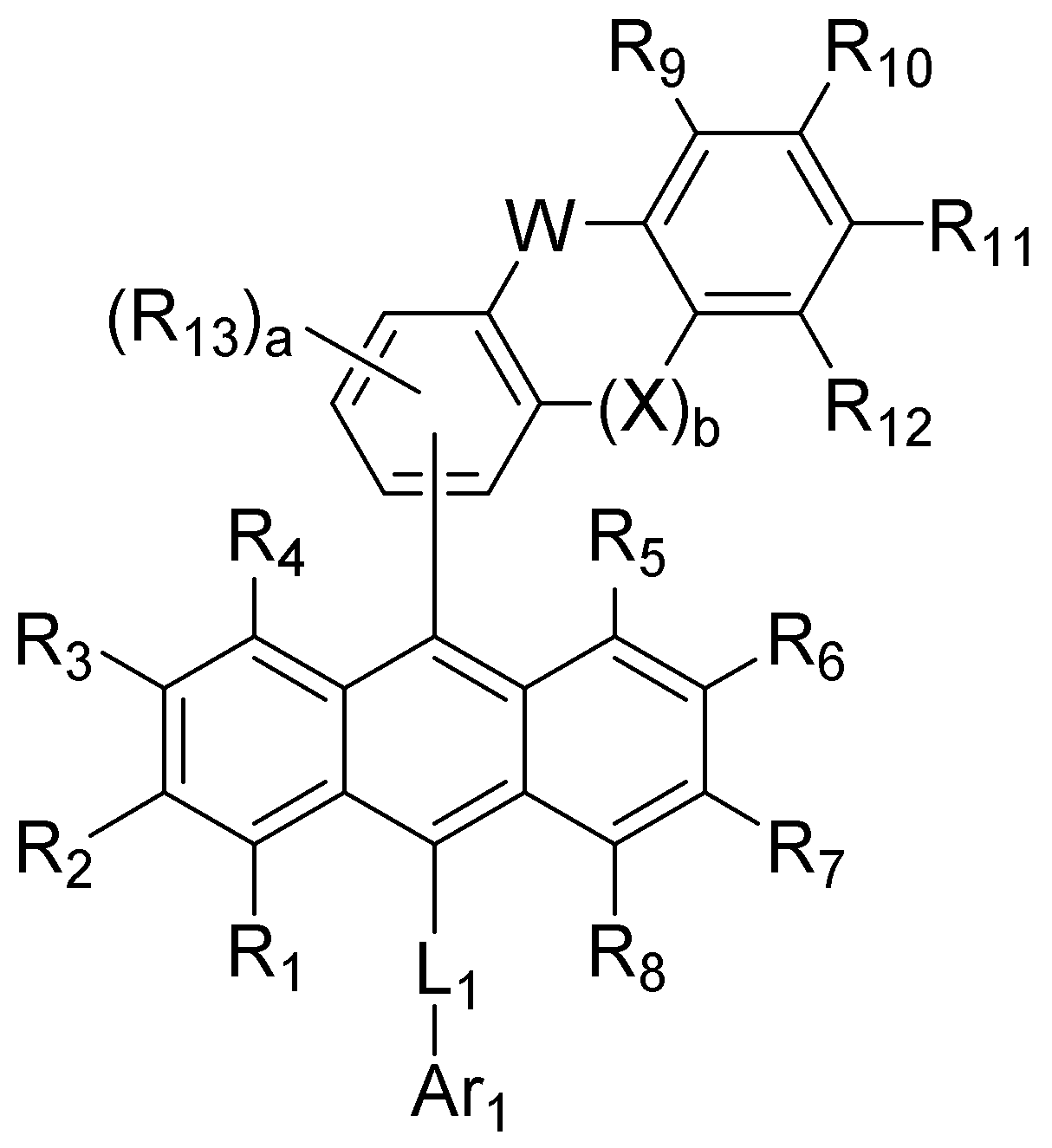
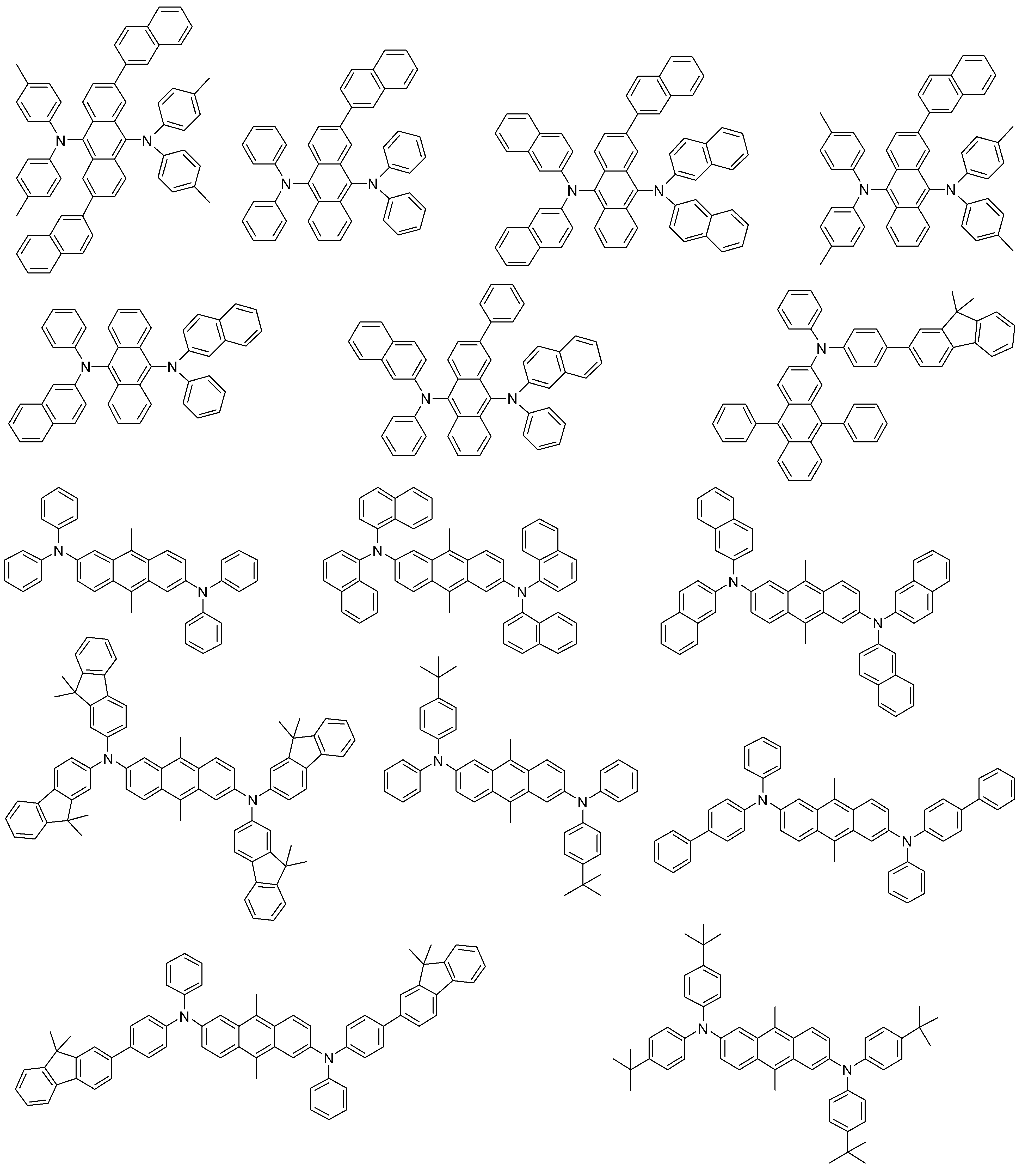
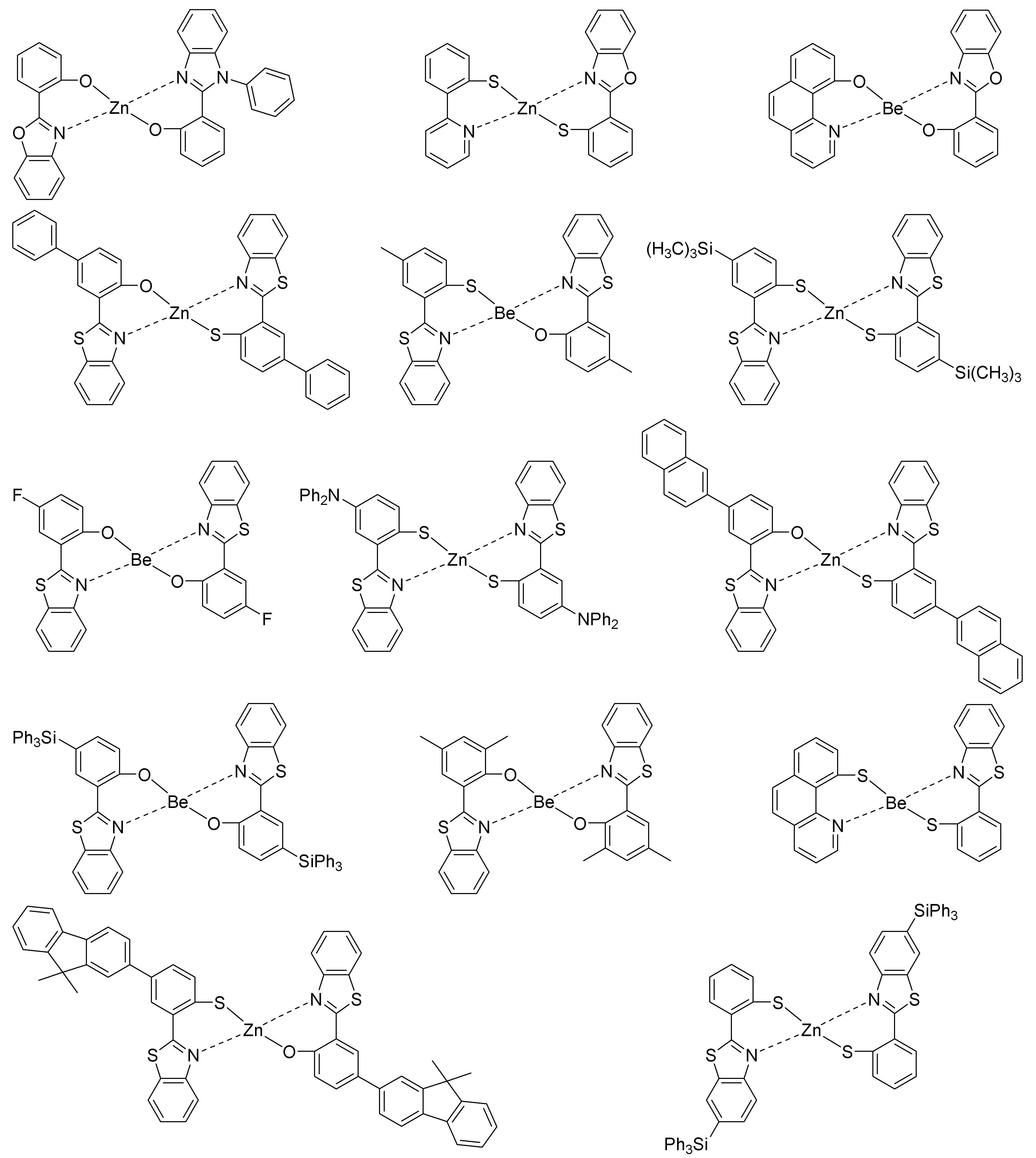
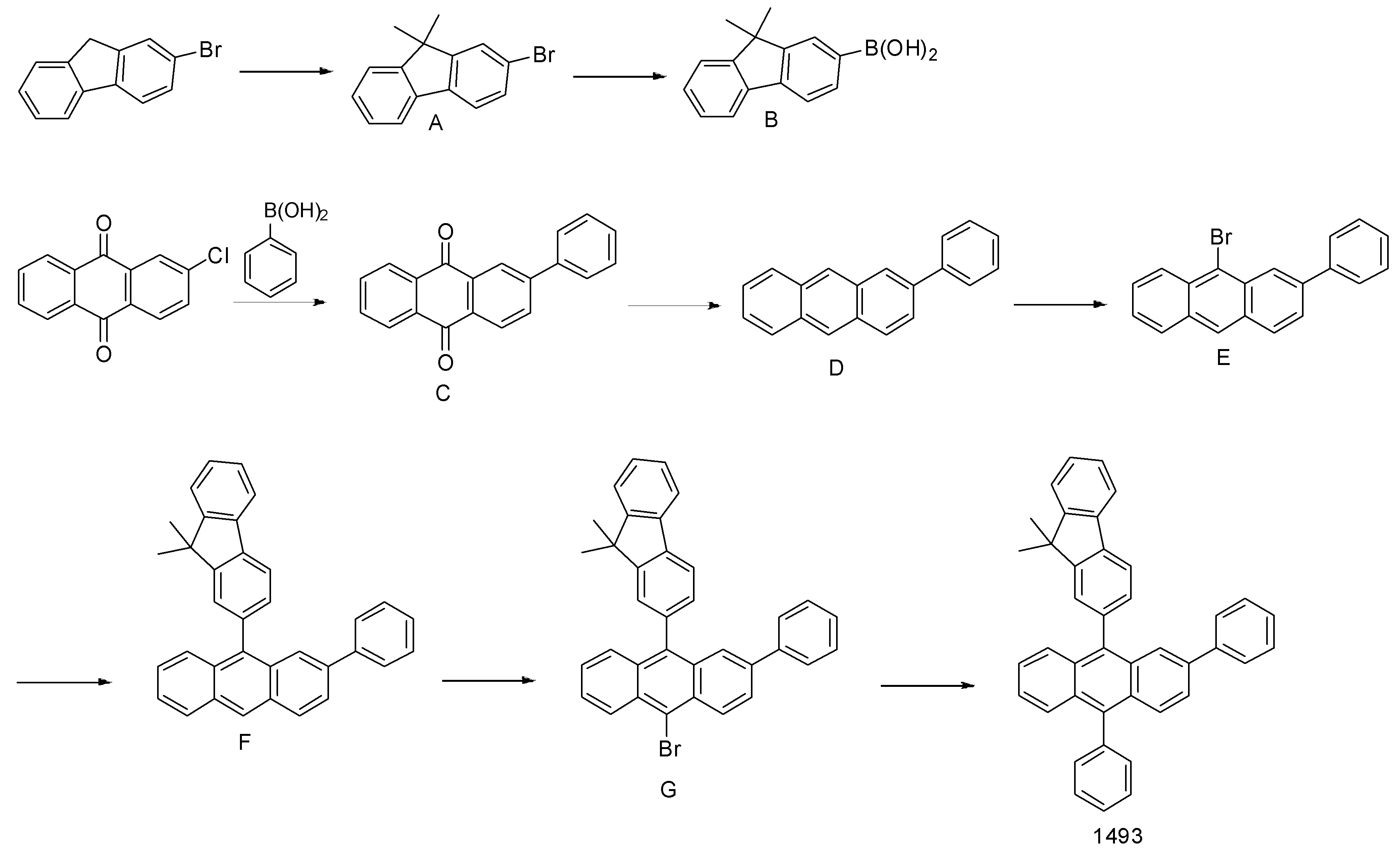
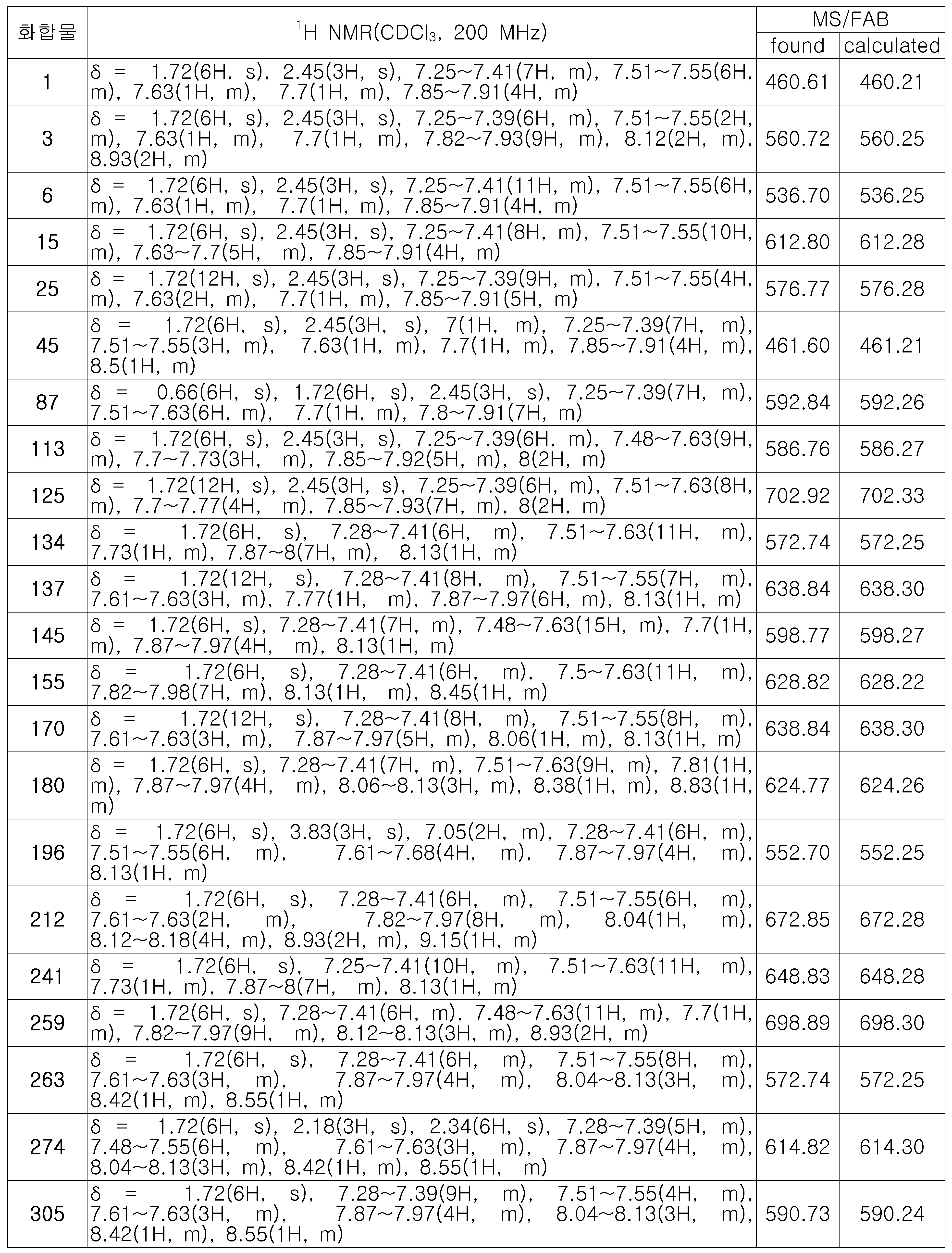
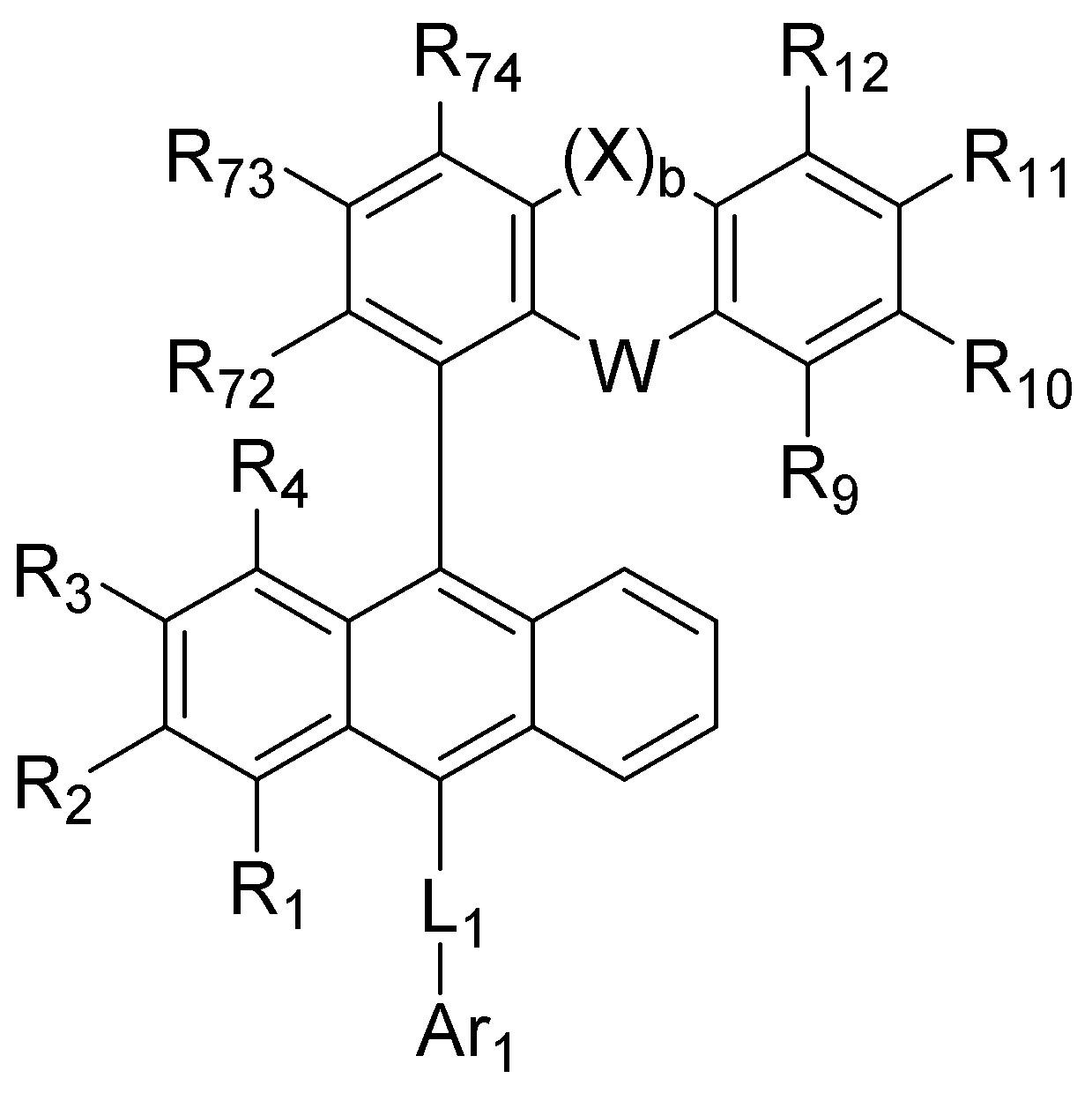
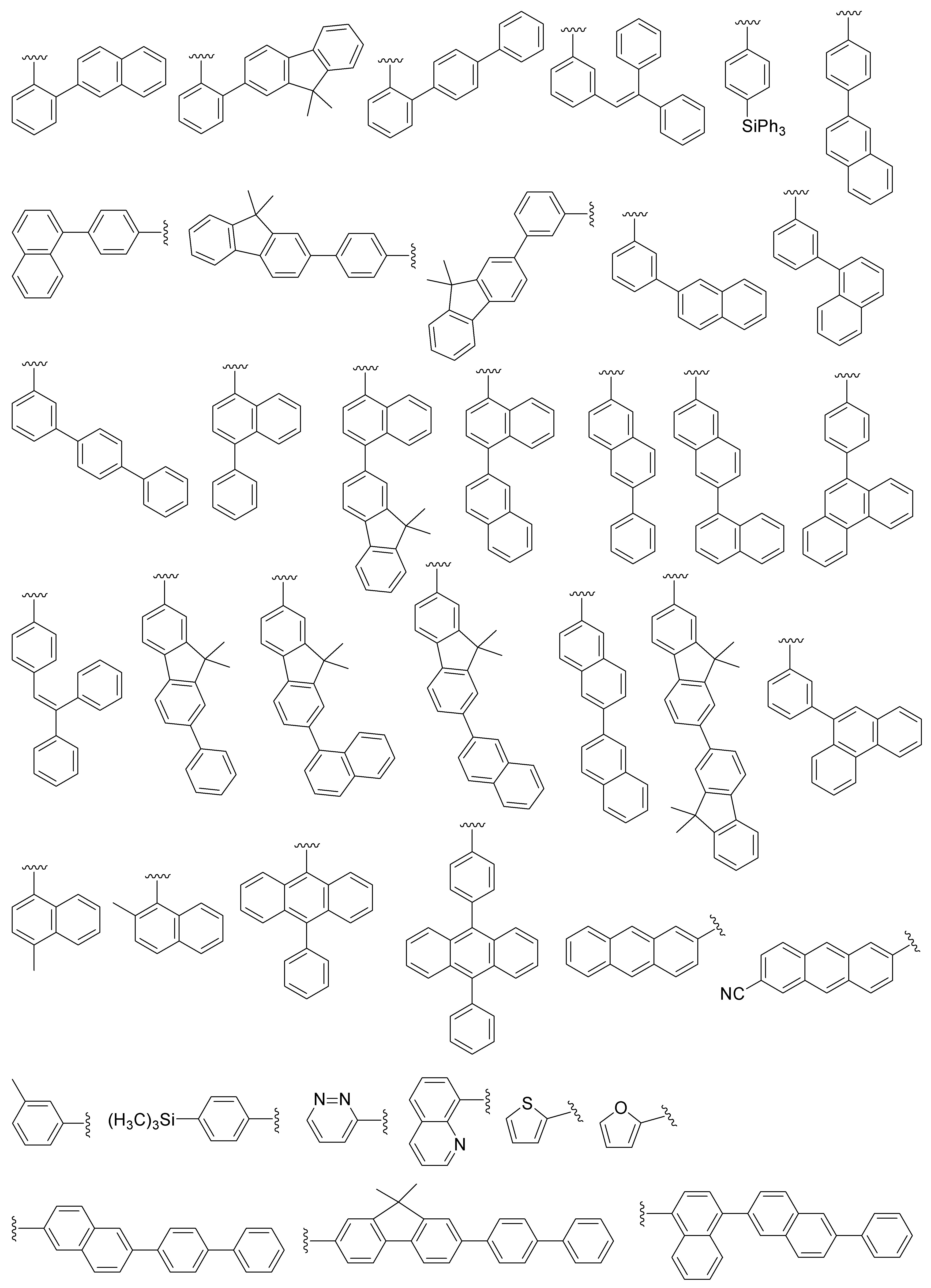
- 하기 화학식 1로 표시되는 유기 발광 화합물.[화학식 1][상기 화학식 1에서,R1 내지 R8은 서로 독립적으로 수소, 할로겐, (C1-C30)알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸이거나, R1 내지 R8는 인접한 치환체와 융합고리를 포함하거나 포함하지 않는 (C3-C30)알킬렌 또는 (C3-C30)알케닐렌으로 연결되어 지환족 고리 및 단일환 또는 다환의 방향족 고리를 형성할 수 있고, 단 R1 내지 R8은 동시에 수소가 아니며;R9 내지 R12는 서로 독립적으로 수소, 할로겐, (C1-C30)알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸, 시아노 또는 나이트로이거나, R9 내지 R12는 인접한 치환체와 융합고리를 포함하거나 포함하지 않는 (C3-C30)알킬렌 또는 (C3-C30)알케닐렌으로 연결되어 지환족 고리 및 단일환 또는 다환의 방향족 고리를 형성할 수 있고,R13은 할로겐, (C1-C30)알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, (C3-C30)시클로알킬, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸이며;W 및 X는 서로 독립적으로 화학결합이거나, CR14R15, NR16, S, O, SiR17R18 또는 PR19이며;R14 내지 R19는 서로 독립적으로 수소, (C1-C30)알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸이거나, R14와 R15, R17와 R18은 융합고리를 포함하거나 포함하지 않는 (C3-C30)알킬렌 또는 (C3-C30)알케닐렌으로 연결되어 지환족 고리 및 단일환 또는 다환의 방향족 고리를 형성할 수 있으며;L1은 화학결합이거나, (C6-C30)아릴렌 또는 (C3-C30)헤테로아릴렌이고, 상기 L1의 아릴렌 또는 헤테로아릴렌은 (C1-C30)알킬, 할로겐, 시아노, (C1-C30)알콕시, (C3-C30)시클로알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, 아다만틸, (C7-C30)바이시클로알킬, 시아노, (C1-C30)알킬아미노, (C6-C30)아릴아미노, (C6-C30)아르(C1-C30)알킬, (C6-C30)아릴옥시, (C6-C30)아릴티오, (C1-C30)알콕시카보닐, 카복실산, 나이트로, 하이드록시, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴 또는 트리(C6-C30)아릴실릴로부터 선택된 하나 이상이 더 치환될 수 있으며;Ar1은 (C1-C30)알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, N, O 및 S로부터 선택된 하나 이상을 포함하는 5원 내지 6원의 헤테로시클로알킬, (C3-C30)시클로알킬, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸, (C7-C30)바이시클로알킬, (C2-C30)알케닐, (C2-C30)알키닐, (C1-C30)알콕시, 시아노, (C1-C30)알킬아미노, (C6-C30)아릴아미노, (C6-C30)아르(C1-C30)알킬, (C6-C30)아릴옥시, (C6-C30)아릴티오, (C1-C30)알콕시카보닐, 카복실산, 나이트로 또는 하이드록시이거나, 하기 구조에서 선택되는 치환기이고,상기 R31 내지 R43는 서로 독립적으로 수소, 할로겐, (C1-C30)알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸이거나, R31 내지 R43는 인접한 치환체와 융합고리를 포함하거나 포함하지 않는 (C3-C30)알킬렌 또는 (C3-C30)알케닐렌으로 연결되어 지환족 고리 및 단일환 또는 다환의 방향족 고리를 형성할 수 있으며;Y 및 Z는 서로 독립적으로 화학결합이거나, CR51R52, NR53, S, O, SiR54R55 또는 PR56이며;R51 내지 R56은 서로 독립적으로 수소, (C1-C30)알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸이거나, R51와 R52, R54와 R55는 융합고리를 포함하거나 포함하지 않는 (C3-C30)알킬렌 또는 (C3-C30)알케닐렌으로 연결되어 지환족 고리 및 단일환 또는 다환의 방향족 고리를 형성할 수 있으며;Ar1, R1 내지 R19의 알킬, 아릴, 헤테로아릴, 헤테로시클로알킬, 시클로알킬, 트리알킬실릴, 디알킬아릴실릴, 트리아릴실릴, 아다만틸, 바이시클로알킬, 알케닐, 알키닐, 알킬아미노 또는 아릴아미노는 할로겐, (C1-C30)알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸로 더 치환될 수 있으며;a는 0 내지 3의 정수이고;b 및 c는 서로 독립적으로 1 내지 4 의 정수이다.]
- 제 1항에 있어서,하기 화학식 2 내지 화학식 5로부터 선택되는 것을 특징으로 하는 유기 발광 화합물.[화학식 2][화학식 3][화학식 4][화학식 5][상기 화학식 2 내지 화학식 5에서, L1, Ar1, R9-R12, X, W 및 b는 상기 청구항 제1항의 화학식 1의 정의와 동일하고;R1 내지 R4는 서로 독립적으로 수소, 할로겐, (C1-C30)알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸이고, 단 R1 내지 R4는 동시에 수소가 아니며;R71 내지 R74는 서로 독립적으로 수소, 할로겐, (C1-C30)알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸, (C1-C30)알콕시, 시아노, 카복실산, 나이트로 또는 하이드록시이며;상기 R1 내지 R4 및 R71 내지 R74의 알킬, 아릴, 헤테로아릴, 트리알킬실릴, 디알킬아릴실릴, 트리아릴실릴, 아다만틸은 할로겐, (C1-C30)알킬, (C6-C30)아릴, (C3-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸로 더 치환될 수 있다.]
- 제 2항에 있어서,상기 R1 내지 R4는 서로 독립적으로 수소, 클로로, 플루오르, 메틸, 에틸, n-프로필, i-프로필, n-부틸, i-부틸, t-부틸, n-펜틸, i-펜틸, n-헥실, n-헵틸, n-옥틸, 2-에틸헥실, n-노닐, 데실, 도데실, 헥사데실, 벤질, 트리플루오르메틸, 퍼플루오르에틸, 트리플루오르에틸, 퍼플루오르프로필, 퍼플루오르부틸, 페닐, 나프틸, 비페닐, 플루오레닐, 페난트릴, 안트릴, 플루오란텐일, 트리페닐렌일, 피렌일, 크라이세닐, 나프타세닐, 페릴렌일, 스피로바이플루오레닐, 피리딜, 피롤릴, 퓨란일, 티오펜일, 이미다졸릴, 벤조이미다졸릴, 피라진일, 피리미딘일, 피리다진일, 퀴놀릴, 트리아진일, 벤조퓨란일, 벤조티오펜일, 피라졸릴, 인돌릴, 카바졸릴, 티아졸릴, 옥사졸릴, 벤조티아졸릴, 벤조옥사졸릴, 페난트롤린일, 트리메틸실릴, 트리에틸실릴, 트리프로필실릴, 트리(t-부틸)실릴, t-부틸디메틸실릴, 디메틸페닐실릴, 트리페닐실릴, 아다만틸이고, 단 R1 내지 R4는 동시에 수소가 아닌 것을 특징으로 하는 유기 발광 화합물.
- 제 2항에 있어서,[R81 내지 R97은 서로 독립적으로 수소, 할로겐, (C1-C60)알킬, (C6-C60)아릴, (C3-C60)헤테로아릴, N, O 및 S로부터 선택된 하나 이상을 포함하는 5원 내지 6원의 헤테로시클로알킬, (C3-C60)시클로알킬, 트리(C1-C60)알킬실릴, 디(C1-C60) 알킬(C6-C60)아릴실릴, 트리(C6-C60)아릴실릴, 아다만틸, (C7-C60)바이시클로알킬, (C2-C60)알케닐, (C2-C60)알키닐, (C1-C60)알콕시, 시아노, (C1-C60)알킬아미노, (C6-C60)아릴아미노, (C6-C60)아르(C1-C60)알킬, (C6-C60)아릴옥시, (C6-C60)아릴티오, (C1-C60)알콕시카보닐, 카복실산, 나이트로 또는 하이드록시이며, 상기 R81 내지 R97의 알킬, 아릴, 헤테로아릴, 헤테로시클로알킬, 시클로알킬, 트리알킬실릴, 디알킬아릴실릴, 트리아릴실릴, 아다만틸, 바이시클로알킬, 알케닐, 알키닐, 알킬아미노 또는아릴아미노는 할로겐, (C1-C60)알킬, (C6-C60)아릴, (C3-C60)헤테로아릴, N, O 및 S로부터 선택된 하나 이상을 포함하는 5원 내지 6원의 헤테로시클로알킬, (C3-C60)시클로알킬, 트리(C1-C60)알킬실릴, 디(C1-C60)알킬(C6-C60)아릴실릴, 트리(C6-C60)아릴실릴, 아다만틸, (C7-C60)바이시클로알킬, (C2-C60)알케닐, (C2-C60)알키닐, (C1-C60)알콕시, 시아노, (C1-C60)알킬아미노, (C6-C60)아릴아미노, (C6-C60)아르(C1-C60)알킬, (C6-C60)아릴옥시, (C6-C60)아릴티오, (C1-C60)알콕시카보닐, 카복실산, 나이트로 또는 하이드록시로 더 치환될 수 있으며;L2 및 L3는 서로 독립적으로 화학결합이거나, (C6-C60)아릴렌 또는 (C3-C60)헤테로아릴렌이고, 상기 L2 및 L3의 아릴렌 또는 헤테로아릴렌은 (C1-C60)알킬, 할로겐, 시아노, (C1-C60)알콕시, (C3-C60)시클로알킬, (C6-C60)아릴, (C3-C60)헤테로아릴, 아다만틸, (C7-C60)바이시클로알킬, 시아노, (C1-C60)알킬아미노, (C6-C60)아릴아미노, (C6-C60)아르(C1-C60)알킬, (C6-C60)아릴옥시, (C6-C60)아릴티오, (C1-C60)알콕시카보닐, 카복실산, 나이트로, 하이드록시, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴 또는 트리(C6-C30)아릴실릴로부터 선택된 하나 이상이 더 치환될 수 있으며;A 및 B는 서로 독립적으로 화학결합이거나, CR101R102, NR103, S, O, SiR104R105, PR106, CO, BR107, InR108, Se, GeR109R110, SnR111R112 또는 GaR113이며;R101 내지 R113는 서로 독립적으로 수소, 할로겐, (C1-C60)알킬, (C6-C60)아릴, (C3-C60)헤테로아릴, N, O 및 S로부터 선택된 하나 이상을 포함하는 5원 내지 6원의 헤테로시클로알킬, (C3-C60)시클로알킬, 트리(C1-C60)알킬실릴, 디(C1-C60)알킬(C6-C60)아릴실릴, 트리(C6-C60)아릴실릴, 아다만틸, (C7-C60)바이시클로알킬, (C2-C60)알케닐, (C2-C60)알키닐, (C1-C60)알콕시, 시아노, (C1-C60)알킬아미노, (C6-C60)아릴아미노, (C6-C60)아르(C1-C60)알킬, (C6-C60)아릴옥시, (C6-C60)아릴티오, (C1-C60)알콕시카보닐, 카복실산, 나이트로 또는 하이드록시이거나, R101와 R102, R104와 R105, R109와 R110 및 R111와 R112는 융합고리를 포함하거나 포함하지 않는 (C3-C60)알킬렌 또는 (C3-C60)알케닐렌으로 연결되어 지환족 고리 및 단일환 또는 다환의 방향족 고리를 형성할 수 있으며;d는 1 내지 5의 정수이고;e는 1 내지 4의 정수이다.]
- 제 1항 내지 제 6항에서 선택되는 어느 한 항에 따른 유기 발광 화합물을 포함하는 것을 특징으로 하는 유기 발광 소자.
- 제 7항에 있어서,상기 유기 발광 화합물은 발광층의 호스트 물질로 사용되는 것을 특징으로 하는 유기 발광 소자.
- 제1전극;제2전극; 및상기 제1전극 및 제2전극 사이에 개재되는 1층 이상의 유기물층으로 이루어진 유기 발광 소자에 있어서,상기 유기물층은 제 1항 내지 제 6항에서 선택되는 어느 한 항에 따른 유기 발광 화합물을 하나 이상 포함하는 것을 특징으로 하는 유기 발광 소자.
- 제 9항에 있어서,상기 유기물층은 발광층을 포함하며, 상기 발광층은 상기 유기 발광 화합물 하나 이상과 도판트 하나 이상을 포함하는 것을 특징으로 하는 유기 발광 소자.
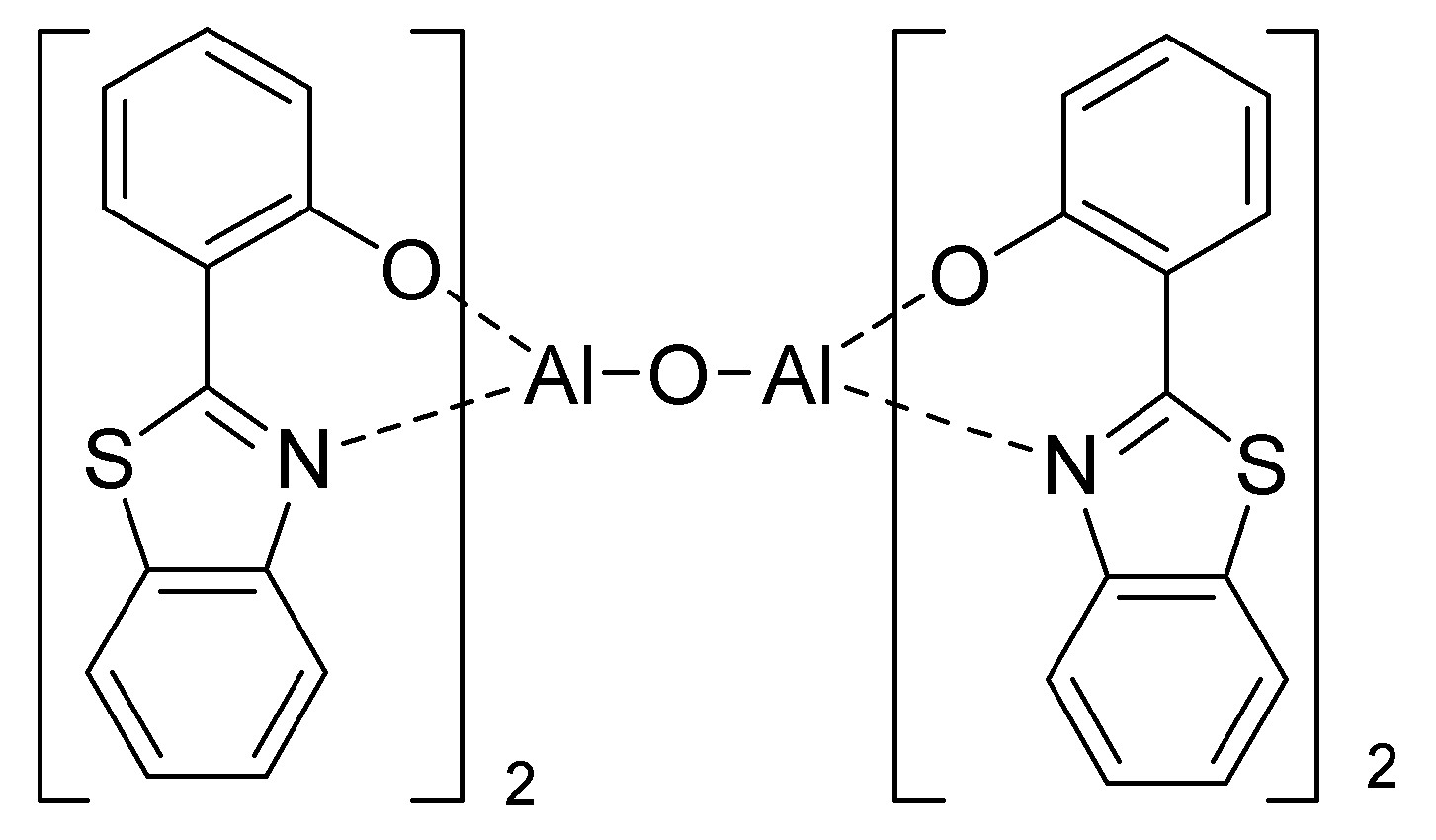
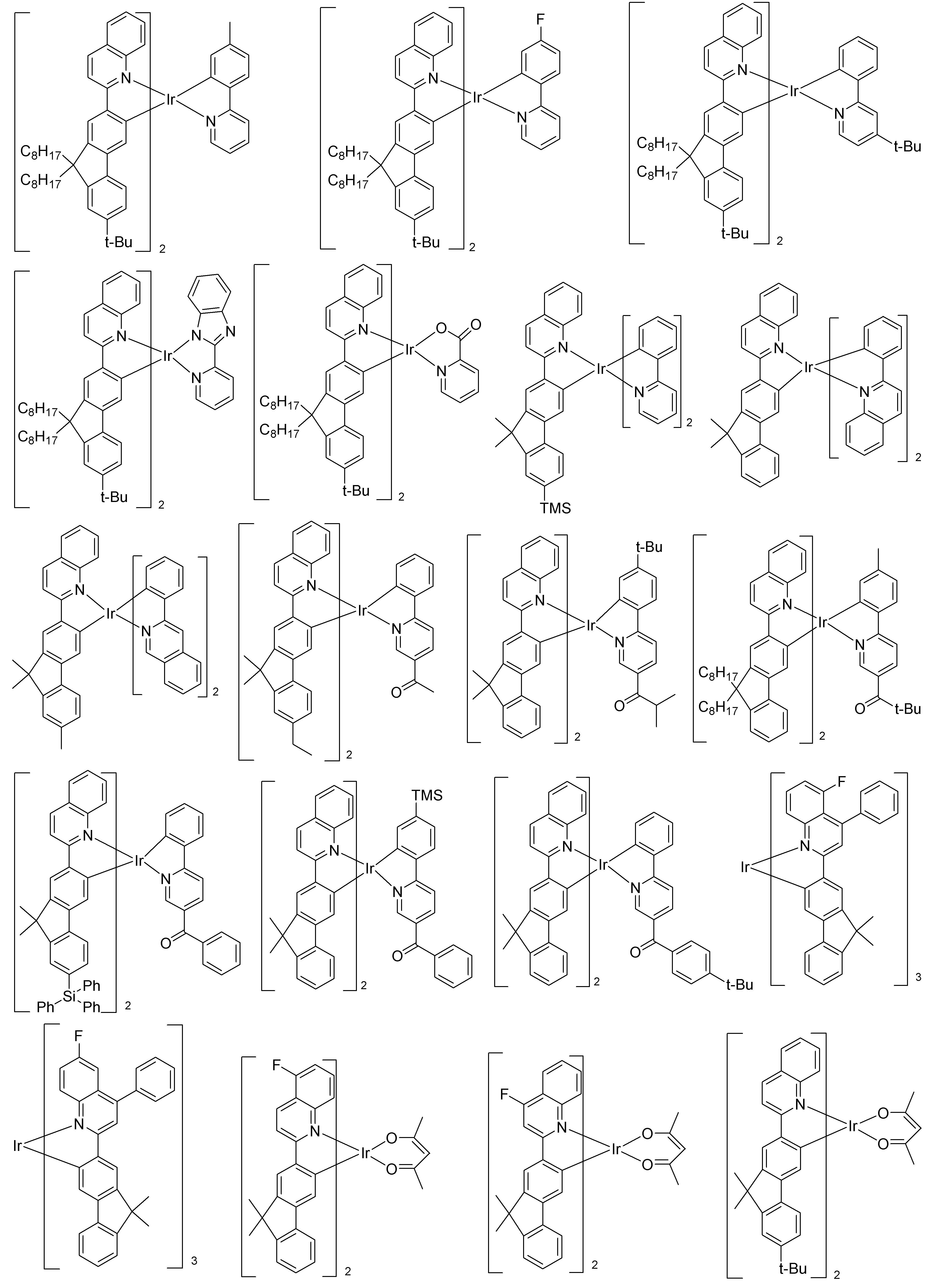
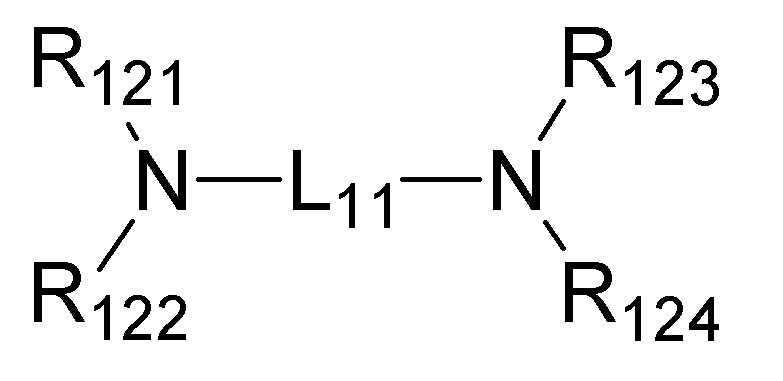
- 제 10항에 있어서,도판트는 하기 화학식 6 또는 화학식 7의 화합물에서 선택되는 것을 특징으로 하는 유기 발광 소자.[화학식 6][화학식 7][상기 화학식 7에서,L11은 할로겐, (C1-C30)알킬, (C6-C30)아릴, (C4-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸, 시아노, (C1-C30)알킬아미노, (C6-C30)아릴아미노로 이루어진 군으로부터 선택된 하나 이상의 치환기가 치환되거나 치환되지 않은 (C6-C30)아릴렌이고, 상기 아릴렌에 치환되는 치환기인 알킬, 아릴, 헤테로아릴, 알킬아미노 및 아릴아미노는 할로겐, (C1-C30)알킬, (C6-C30)아릴, (C4-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸, 시아노, (C1-C30)알킬아미노, (C6-C30)아릴아미노로부터 선택된 하나 이상이 더 치환될 수 있으며;R121 내지 R124는 서로 독립적으로 (C1-C30)알킬, (C6-C30)아릴, (C4-C30)헤테로아릴, (C6-C30)아릴아미노, (C1-C30)알킬아미노, (C3-C30)시클로알킬이거나, R121 내지 R124는 인접한 치환체와 융합고리를 포함하거나 포함하지 않는 (C3-C30)알킬렌 또는 (C3-C30)알케닐렌으로 연결되어 지환족 고리 및 단일환 또는 다환의 방향족 고리를 형성할 수 있고,상기 R121 내지 R124의 알킬, 아릴, 헤테로아릴, 아릴아미노, 알킬아미노, 시클로알킬은 할로겐, (C1-C30)알킬, (C6-C30)아릴, (C4-C30)헤테로아릴, 트리(C1-C30)알킬실릴, 디(C1-C30)알킬(C6-C30)아릴실릴, 트리(C6-C30)아릴실릴, 아다만틸, (C1-C30)알콕시, 시아노, (C1-C30)알킬아미노, (C6-C30)아릴아미노로부터 선택된 하나 이상이 더 치환될 수 있다.]
- 제 9항에 있어서,상기 유기물층에 아릴아민계 화합물 또는 스티릴아릴아민계 화합물로 이루어진 군으로부터 선택된 하나 이상의 화합물을 포함하는 것을 특징으로 하는 유기 발광 소자.
- 제 9항에 있어서,상기 유기물층에 1족, 2족, 4주기, 5주기 전이금속, 란탄계열금속 및 d-전이원소의 유기금속으로 이루어진 군으로부터 선택되는 하나 이상의 금속을 더 포함하는 것을 특징으로 하는 유기 발광 소자.
- 제 9항에 있어서,상기 발광층을 포함하는 유기전기발광소자를 서브픽셀로 하고, Ir, Pt, Pd, Rh, Re, Os, Tl, Pb, Bi, In, Sn, Sb, Te, Au 및 Ag로 이루어진 군에서 선택되는 하나 이상의 금속화합물을 포함하는 서브픽셀 하나 이상을 동시에 병렬로 패터닝한 독립발광 방식의 픽셀구조를 가진 유기 발광 소자.
- 제 9항에 있어서,상기 발광층에 500nm이하의 파장을 발광피크로 갖는 화합물 또는 560nm이상의 파장을 발광피크로 갖는 화합물을 동시에 포함하는 것을 특징으로 하는 유기 발광 소자.
- 제 9항에 있어서,상기 유기물층은 발광층 이외에 전하생성층을 동시에 포함하는 것을 특징으로 하는 유기 발광 소자.
- 제 9항에 있어서,한 쌍의 전극중 하나 이상의 내측표면에 칼코제나이드(chalcogenide)층, 할 로겐화 금속층 및 금속 산화물층으로 구성되는 군으로부터 선택되는 하나 이상의 층이 배치되는 것을 특징으로 하는 유기 발광 소자.
- 제 9항에 있어서,한 쌍의 전극중 하나 이상의 내측표면에 환원성 도판트(dopant)와 유기물의 혼합 영역, 또는 산화성 도판트와 유기물의 혼합 영역이 배치되는 것을 특징으로 하는 유기 발광 소자.
- 삭제
Priority Applications (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020080030645A KR100910150B1 (ko) | 2008-04-02 | 2008-04-02 | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 발광 소자 |
| JP2009065946A JP5730468B2 (ja) | 2008-04-02 | 2009-03-18 | 新規な有機電界発光化合物及びこれを用いる有機電界発光素子 |
| TW098109327A TW200944575A (en) | 2008-04-02 | 2009-03-23 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| EP09250820A EP2108689A3 (en) | 2008-04-02 | 2009-03-24 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| US12/383,956 US20100045170A1 (en) | 2008-04-02 | 2009-03-31 | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| CN200910130579.2A CN101560136B (zh) | 2008-04-02 | 2009-04-01 | 有机电致发光化合物和使用该化合物的有机电致发光器件 |
| CN2013100666785A CN103214337A (zh) | 2008-04-02 | 2009-04-01 | 新颖的有机电致发光化合物和使用该化合物的有机电致发光器件 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020080030645A KR100910150B1 (ko) | 2008-04-02 | 2008-04-02 | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 발광 소자 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR100910150B1 true KR100910150B1 (ko) | 2009-08-03 |
Family
ID=40873417
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020080030645A Expired - Fee Related KR100910150B1 (ko) | 2008-04-02 | 2008-04-02 | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 발광 소자 |
Country Status (6)
| Country | Link |
|---|---|
| US (1) | US20100045170A1 (ko) |
| EP (1) | EP2108689A3 (ko) |
| JP (1) | JP5730468B2 (ko) |
| KR (1) | KR100910150B1 (ko) |
| CN (2) | CN101560136B (ko) |
| TW (1) | TW200944575A (ko) |
Cited By (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2010044607A1 (en) * | 2008-10-14 | 2010-04-22 | Cheil Industries Inc. | Benzimidazole compounds and organic photoelectric device with the same |
| WO2011081451A3 (ko) * | 2009-12-30 | 2011-12-01 | 주식회사 두산 | 트리페닐렌계 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| KR101127579B1 (ko) | 2009-08-28 | 2012-03-23 | 삼성모바일디스플레이주식회사 | 헤테로아릴아민 화합물 및 이를 이용한 유기 발광 소자 |
| KR101137385B1 (ko) | 2009-08-28 | 2012-04-20 | 삼성모바일디스플레이주식회사 | 헤테로아릴아민 화합물 및 이를 이용한 유기 발광 소자 |
| KR101298465B1 (ko) * | 2011-01-04 | 2013-08-23 | 주식회사 두산 | 페나진계 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| KR20140020208A (ko) * | 2012-08-07 | 2014-02-18 | 주식회사 동진쎄미켐 | 아크리딘 유도체를 포함하는 유기발광 화합물 및 이를 포함하는 유기발광소자 |
| WO2015047018A1 (ko) * | 2013-09-30 | 2015-04-02 | 주식회사 두산 | 유기 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| KR20150037119A (ko) * | 2013-09-30 | 2015-04-08 | 주식회사 두산 | 유기 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| KR101539730B1 (ko) * | 2014-08-11 | 2015-07-28 | 성균관대학교산학협력단 | 유기 발광 화합물, 이의 제조 방법, 및 이를 포함하는 유기 전계 발광 소자 |
| US9147847B2 (en) | 2009-05-29 | 2015-09-29 | Idemitsu Kosan Co., Ltd. | Anthracene derivative and organic electroluminescent element using the same |
| KR20160089693A (ko) | 2015-01-20 | 2016-07-28 | 에스에프씨 주식회사 | 유기발광소자용 화합물 및 이를 포함하는 유기 발광 소자 |
| KR20160112111A (ko) | 2015-03-18 | 2016-09-28 | 에스에프씨 주식회사 | 유기발광소자용 화합물 및 이를 포함하는 유기 발광 소자 |
| KR20160126873A (ko) | 2015-04-23 | 2016-11-02 | 에스에프씨 주식회사 | 유기 발광 소자용 화합물 및 이를 포함하는 유기발광소자 |
| WO2017010749A1 (ko) * | 2015-07-13 | 2017-01-19 | 에스에프씨 주식회사 | 고효율의 유기 발광 소자 |
| KR20180030914A (ko) * | 2015-07-29 | 2018-03-26 | 메르크 파텐트 게엠베하 | 플루오렌 구조를 갖는 화합물 |
| US10103338B1 (en) | 2017-08-14 | 2018-10-16 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device and electronic device |
| KR20190033504A (ko) | 2019-03-20 | 2019-03-29 | 에스에프씨 주식회사 | 유기 발광 소자용 화합물 및 이를 포함하는 유기발광소자 |
| KR20190127622A (ko) | 2015-01-20 | 2019-11-13 | 에스에프씨 주식회사 | 유기발광소자용 화합물 및 이를 포함하는 유기 발광 소자 |
| WO2019235902A1 (ko) * | 2018-06-08 | 2019-12-12 | 주식회사 엘지화학 | 다환 화합물 및 이를 포함하는 유기전자소자 |
| US10800796B2 (en) | 2017-09-21 | 2020-10-13 | Samsung Display Co., Ltd. | Aromatic compound and organic electroluminescence device including the same |
| US10818851B2 (en) | 2015-07-31 | 2020-10-27 | Samsung Display Co., Ltd. | Condensed cyclic compound and organic light-emitting device including the same |
| KR20200139113A (ko) | 2015-01-20 | 2020-12-11 | 에스에프씨 주식회사 | 유기발광소자용 화합물 및 이를 포함하는 유기 발광 소자 |
| WO2021086099A1 (ko) * | 2019-10-30 | 2021-05-06 | 주식회사 엘지화학 | 안트라센 화합물 및 이를 포함하는 유기 발광 소자 |
| US11251380B2 (en) | 2018-01-08 | 2022-02-15 | Samsung Display Co., Ltd. | Organic electroluminescence device and polycyclic compound for organic electroluminescence device |
| US12225815B2 (en) | 2016-10-07 | 2025-02-11 | Samsung Sdi Co., Ltd. | Composition for organic optoelectronic device, organic optoelectronic device, and display device |
Families Citing this family (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8623520B2 (en) | 2007-11-21 | 2014-01-07 | Idemitsu Kosan Co., Ltd. | Fused aromatic derivative and organic electroluminescence device using the same |
| EP2372803A4 (en) | 2008-12-26 | 2012-07-04 | Idemitsu Kosan Co | MATERIAL FOR AN ORGANIC ELECTROLUMINESCENCE ELEMENT AND ORGANIC ELECTROLUMINESCENE ELEMENT |
| EP2390249B1 (en) | 2009-01-20 | 2015-08-26 | LG Chem, Ltd. | Novel cycloalkene derivatives and organic electronic devices using the same |
| CN102239141A (zh) | 2009-12-16 | 2011-11-09 | 出光兴产株式会社 | 芳香族胺衍生物和使用其的有机电致发光元件 |
| DE102010013806B4 (de) * | 2010-04-03 | 2021-06-10 | Merck Patent Gmbh | Materialien für organische Elektrolumineszenzvorrichtungen |
| JP6007467B2 (ja) * | 2010-07-27 | 2016-10-12 | コニカミノルタ株式会社 | 有機エレクトロルミネッセンス素子材料、有機エレクトロルミネッセンス素子、 |
| JP5507381B2 (ja) * | 2010-07-30 | 2014-05-28 | ユー・ディー・シー アイルランド リミテッド | 有機電界発光素子及び化合物 |
| KR101950363B1 (ko) * | 2010-10-29 | 2019-02-20 | 가부시키가이샤 한도오따이 에네루기 켄큐쇼 | 페난트렌 화합물, 발광 소자, 발광 장치, 전자 기기, 및 조명 장치 |
| JP5984450B2 (ja) * | 2011-03-31 | 2016-09-06 | ユー・ディー・シー アイルランド リミテッド | 有機電界発光素子、並びに、該素子を用いた発光装置、表示装置、照明装置及び該素子用の化合物 |
| JP5659972B2 (ja) * | 2011-07-07 | 2015-01-28 | コニカミノルタ株式会社 | 有機エレクトロルミネッセンス素子および照明装置 |
| TWI774347B (zh) | 2011-08-25 | 2022-08-11 | 日商半導體能源研究所股份有限公司 | 發光元件,發光裝置,電子裝置,照明裝置以及新穎有機化合物 |
| CN103130724A (zh) * | 2011-11-23 | 2013-06-05 | 海洋王照明科技股份有限公司 | 含萘基蒽的有机半导体材料、其制备方法和应用 |
| CN103288730A (zh) * | 2012-02-27 | 2013-09-11 | 海洋王照明科技股份有限公司 | 含喹啉有机半导体材料、其制备方法及有机电致发光器件 |
| EP2876104B1 (en) * | 2012-07-19 | 2018-09-05 | LG Chem, Ltd. | Polycyclic compound and organic electronic device comprising same |
| US9312500B2 (en) | 2012-08-31 | 2016-04-12 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device |
| JP2015233024A (ja) * | 2012-09-03 | 2015-12-24 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| KR102240991B1 (ko) | 2012-09-12 | 2021-04-16 | 이데미쓰 고산 가부시키가이샤 | 신규 화합물, 유기 일렉트로 루미네선스 소자용 재료, 유기 일렉트로 루미네선스 소자 및 전자 기기 |
| KR102104357B1 (ko) * | 2012-12-24 | 2020-04-27 | 엘지디스플레이 주식회사 | 청색 형광 화합물 및 이를 사용한 유기전계발광소자 |
| JPWO2014129048A1 (ja) * | 2013-02-22 | 2017-02-02 | 出光興産株式会社 | アントラセン誘導体、有機エレクトロルミネッセンス素子用材料、有機エレクトロルミネッセンス素子、および電子機器 |
| WO2014141725A1 (ja) | 2013-03-15 | 2014-09-18 | 出光興産株式会社 | アントラセン誘導体及びそれを用いた有機エレクトロルミネッセンス素子 |
| JP6328890B2 (ja) * | 2013-07-09 | 2018-05-23 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子、有機エレクトロルミネッセンス素子用材料、および電子機器 |
| US20160181542A1 (en) * | 2013-09-06 | 2016-06-23 | Idemitsu Kosan Co., Ltd. | Anthracene derivative and organic electroluminescent element using same |
| US10497876B2 (en) * | 2014-12-24 | 2019-12-03 | Hodogaya Chemical Co., Ltd. | Organic electroluminescent device |
| EP3255692B1 (en) * | 2015-02-03 | 2023-07-05 | Hodogaya Chemical Co., Ltd. | Organic electroluminescent element |
| KR101974860B1 (ko) * | 2015-02-04 | 2019-09-05 | 에스에프씨주식회사 | 저전압구동이 가능하며 장수명을 갖는 유기 발광 소자 |
| CN106032350B (zh) * | 2015-03-09 | 2019-03-01 | 广东阿格蕾雅光电材料有限公司 | 有机电子材料 |
| CN104844587B (zh) * | 2015-04-29 | 2018-06-01 | 深圳市华星光电技术有限公司 | 含有吩恶噻结构的共轭化合物及其制备方法与有机电致发光二极管器件 |
| KR102002031B1 (ko) * | 2015-06-12 | 2019-07-23 | 에스에프씨주식회사 | 고효율을 갖는 유기 발광 소자 |
| CN107922359A (zh) | 2015-07-30 | 2018-04-17 | 默克专利有限公司 | 用于有机电致发光器件的材料 |
| KR102442614B1 (ko) * | 2015-08-07 | 2022-09-14 | 삼성디스플레이 주식회사 | 디벤조보롤계 화합물 및 이를 포함한 유기 발광 소자 |
| TWI745361B (zh) | 2016-03-17 | 2021-11-11 | 德商麥克專利有限公司 | 具有螺聯茀結構之化合物 |
| JP7073388B2 (ja) * | 2016-11-08 | 2022-05-23 | メルク パテント ゲーエムベーハー | 電子デバイスのための化合物 |
| CN108218860A (zh) * | 2018-01-18 | 2018-06-29 | 长春海谱润斯科技有限公司 | 一种杂蒽衍生物及其制备方法和有机发光器件 |
| EP3775110B1 (en) * | 2018-04-05 | 2023-04-19 | Dow Global Technologies, LLC | Substituted dibenzofurans as fuel markers |
| CN109384726A (zh) * | 2018-06-28 | 2019-02-26 | 吉林奥来德光电材料股份有限公司 | 一种有机发光化合物及其制备方法和有机电致发光器件 |
| CN108794404B (zh) * | 2018-06-28 | 2020-08-21 | 吉林奥来德光电材料股份有限公司 | 一种蒽类有机发光化合物及其制备方法以及有机电致发光器件 |
| CN109369598A (zh) * | 2018-10-29 | 2019-02-22 | 吉林奥来德光电材料股份有限公司 | 一种有机发光材料、及制法和含该材料的有机发光器件 |
| CN109456297A (zh) * | 2018-10-29 | 2019-03-12 | 吉林奥来德光电材料股份有限公司 | 一种有机发光材料及其制法和含该材料的有机电致发光器件 |
| CN109369660A (zh) * | 2018-10-29 | 2019-02-22 | 吉林奥来德光电材料股份有限公司 | 有机发光材料及其制法和含该材料的有机电致发光器件 |
| KR102290023B1 (ko) * | 2018-11-06 | 2021-08-13 | 주식회사 엘지화학 | 유기 발광 소자 |
| JP2022123150A (ja) * | 2019-04-26 | 2022-08-24 | 出光興産株式会社 | 化合物、有機エレクトロルミネッセンス素子及び電子機器 |
| JP2022123149A (ja) * | 2019-04-26 | 2022-08-24 | 出光興産株式会社 | 化合物、有機エレクトロルミネッセンス素子及び電子機器 |
| CN110330472B (zh) * | 2019-07-10 | 2022-02-01 | 吉林奥来德光电材料股份有限公司 | 一种蓝光材料及其制备方法和应用 |
| CN110845421A (zh) * | 2019-11-28 | 2020-02-28 | 吉林奥来德光电材料股份有限公司 | 一种电子传输化合物、其合成方法及有机电致发光器件 |
| WO2023078824A1 (en) * | 2021-11-04 | 2023-05-11 | Dottikon Es Holding Ag | Spiro-(indane-fluorene) type compounds and their use in organic electronics |
| CN120136892B (zh) * | 2025-05-16 | 2025-08-26 | 烟台九目化学股份有限公司 | 一种哒嗪并蒽类发光材料及应用 |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001176664A (ja) | 1999-12-15 | 2001-06-29 | Nec Corp | 有機エレクトロルミネッセント素子 |
| JP2001307885A (ja) | 2000-02-18 | 2001-11-02 | Nec Corp | 有機el素子及び有機elディスプレイ |
Family Cites Families (45)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5077142A (en) * | 1989-04-20 | 1991-12-31 | Ricoh Company, Ltd. | Electroluminescent devices |
| US5061569A (en) * | 1990-07-26 | 1991-10-29 | Eastman Kodak Company | Electroluminescent device with organic electroluminescent medium |
| JP3505257B2 (ja) * | 1995-02-24 | 2004-03-08 | 三洋電機株式会社 | 有機エレクトロルミネッセンス素子 |
| JP3712760B2 (ja) * | 1995-05-17 | 2005-11-02 | Tdk株式会社 | 有機el素子 |
| EP1146034A1 (en) * | 1995-09-25 | 2001-10-17 | Toyo Ink Manufacturing Co., Ltd. | Light-emitting material for organic electroluminescence device, and organic electroluminescence device for which the light-emitting material is adapted |
| US5989737A (en) * | 1997-02-27 | 1999-11-23 | Xerox Corporation | Organic electroluminescent devices |
| US5935721A (en) * | 1998-03-20 | 1999-08-10 | Eastman Kodak Company | Organic electroluminescent elements for stable electroluminescent |
| US6465115B2 (en) * | 1998-12-09 | 2002-10-15 | Eastman Kodak Company | Electroluminescent device with anthracene derivatives hole transport layer |
| EP1666561A1 (en) * | 1998-12-28 | 2006-06-07 | Idemitsu Kosan Company Limited | Organic electroluminescent element |
| KR100799799B1 (ko) * | 1999-09-21 | 2008-02-01 | 이데미쓰 고산 가부시키가이샤 | 유기 전자발광 소자 및 유기 발광 매체 |
| JP4094203B2 (ja) * | 2000-03-30 | 2008-06-04 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子及び有機発光媒体 |
| JP4996794B2 (ja) * | 2000-08-10 | 2012-08-08 | 三井化学株式会社 | 炭化水素化合物、有機電界発光素子用材料および有機電界発光素子 |
| TWI290546B (en) | 2000-08-10 | 2007-12-01 | Mitsui Chemicals Inc | Hydrocarbon compounds for organic electroluminescent elements and organic electroluminescent elements |
| JP3998903B2 (ja) * | 2000-09-05 | 2007-10-31 | 出光興産株式会社 | 新規アリールアミン化合物及び有機エレクトロルミネッセンス素子 |
| KR100377575B1 (ko) | 2000-10-17 | 2003-03-26 | 삼성에스디아이 주식회사 | 유기 전계 발광 소자용 청색 발광 화합물 및 이를 사용한유기 전계 발광 소자 |
| JP4220696B2 (ja) * | 2001-10-16 | 2009-02-04 | 三井化学株式会社 | 炭化水素化合物、有機電界発光素子用材料および有機電界発光素子 |
| EP2199361B2 (en) * | 2002-07-19 | 2016-06-15 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent devices and organic luminescent medium |
| JP4025137B2 (ja) * | 2002-08-02 | 2007-12-19 | 出光興産株式会社 | アントラセン誘導体及びそれを利用した有機エレクトロルミネッセンス素子 |
| KR100924462B1 (ko) * | 2002-08-23 | 2009-11-03 | 이데미쓰 고산 가부시키가이샤 | 유기 전기발광 소자 및 안트라센 유도체 |
| TW593624B (en) | 2002-10-16 | 2004-06-21 | Univ Tsinghua | Aromatic compounds and organic LED |
| WO2004053018A1 (en) * | 2002-12-06 | 2004-06-24 | Shuang Xie | Electroluminescent devices |
| JP4067414B2 (ja) * | 2003-01-22 | 2008-03-26 | 三井化学株式会社 | 非対称置換アントラセン化合物、および該非対称置換アントラセン化合物を含有する有機電界発光素子 |
| US7651787B2 (en) * | 2003-02-19 | 2010-01-26 | Lg Display Co., Ltd. | Organic electroluminescent device |
| JP4070676B2 (ja) * | 2003-07-25 | 2008-04-02 | 三井化学株式会社 | 非対称置換アントラセン化合物、および該非対称置換アントラセン化合物を含有する有機電界発光素子 |
| US7180089B2 (en) | 2003-08-19 | 2007-02-20 | National Taiwan University | Reconfigurable organic light-emitting device and display apparatus employing the same |
| US7326371B2 (en) * | 2004-03-25 | 2008-02-05 | Eastman Kodak Company | Electroluminescent device with anthracene derivative host |
| JP4384536B2 (ja) * | 2004-04-27 | 2009-12-16 | 三井化学株式会社 | アントラセン化合物、および該アントラセン化合物を含有する有機電界発光素子 |
| TWI373506B (en) * | 2004-05-21 | 2012-10-01 | Toray Industries | Light-emitting element material and light-emitting material |
| TWI327563B (en) * | 2004-05-24 | 2010-07-21 | Au Optronics Corp | Anthracene compound and organic electroluminescent device including the anthracene compound |
| JP4829486B2 (ja) * | 2004-08-04 | 2011-12-07 | Jnc株式会社 | 有機電界発光素子 |
| DE602005026533D1 (de) * | 2004-09-02 | 2011-04-07 | Lg Chemical Ltd | Anthracenderivate und deren verwendung als lichtemittierendes material in organischer lichtemittierender vorrichtung |
| JP4790260B2 (ja) * | 2004-12-22 | 2011-10-12 | 出光興産株式会社 | アントラセン誘導体を用いた有機エレクトロルミネッセンス素子 |
| US20060204783A1 (en) * | 2005-03-10 | 2006-09-14 | Conley Scott R | Organic electroluminescent device |
| US20060269782A1 (en) * | 2005-05-25 | 2006-11-30 | Eastman Kodak Company | OLED electron-transporting layer |
| US7479330B2 (en) * | 2005-05-26 | 2009-01-20 | Au Optronics Corporation | Anthracene derivatives for organic electroluminescent device |
| DE602006011734D1 (de) * | 2005-05-30 | 2010-03-04 | Basf Se | Elektrolumineszenz-gerät |
| KR100788254B1 (ko) * | 2005-08-16 | 2007-12-27 | (주)그라쎌 | 녹색 발광 화합물 및 이를 발광재료로서 채용하고 있는발광소자 |
| JP4807013B2 (ja) * | 2005-09-02 | 2011-11-02 | 東レ株式会社 | 発光素子材料および発光素子 |
| JP4726584B2 (ja) * | 2005-09-15 | 2011-07-20 | 三井化学株式会社 | 芳香族化合物、該芳香族化合物を含有する有機電界発光素子 |
| US8647753B2 (en) * | 2005-10-12 | 2014-02-11 | Lg Display Co., Ltd. | Organic electroluminescence device |
| US20070092759A1 (en) * | 2005-10-26 | 2007-04-26 | Begley William J | Organic element for low voltage electroluminescent devices |
| US20070152568A1 (en) * | 2005-12-29 | 2007-07-05 | Chun-Liang Lai | Compounds for an organic electroluminescent device and an organic electroluminescent device using the same |
| JP2007227717A (ja) * | 2006-02-24 | 2007-09-06 | Toyo Ink Mfg Co Ltd | 有機エレクトロルミネッセンス素子 |
| JP2008063240A (ja) * | 2006-09-05 | 2008-03-21 | Mitsui Chemicals Inc | アントラセン化合物の製造方法 |
| KR20090098585A (ko) * | 2008-03-14 | 2009-09-17 | (주)그라쎌 | 유기발광화합물을 발광재료로서 채용하고 있는유기전기발광소자 |
-
2008
- 2008-04-02 KR KR1020080030645A patent/KR100910150B1/ko not_active Expired - Fee Related
-
2009
- 2009-03-18 JP JP2009065946A patent/JP5730468B2/ja not_active Expired - Fee Related
- 2009-03-23 TW TW098109327A patent/TW200944575A/zh unknown
- 2009-03-24 EP EP09250820A patent/EP2108689A3/en not_active Withdrawn
- 2009-03-31 US US12/383,956 patent/US20100045170A1/en not_active Abandoned
- 2009-04-01 CN CN200910130579.2A patent/CN101560136B/zh not_active Expired - Fee Related
- 2009-04-01 CN CN2013100666785A patent/CN103214337A/zh active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2001176664A (ja) | 1999-12-15 | 2001-06-29 | Nec Corp | 有機エレクトロルミネッセント素子 |
| JP2001307885A (ja) | 2000-02-18 | 2001-11-02 | Nec Corp | 有機el素子及び有機elディスプレイ |
Cited By (54)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9530970B2 (en) | 2008-10-14 | 2016-12-27 | Cheil Industries, Inc. | Benzimidazole compound, organic photoelectric device including the same, and display element including the same |
| WO2010044607A1 (en) * | 2008-10-14 | 2010-04-22 | Cheil Industries Inc. | Benzimidazole compounds and organic photoelectric device with the same |
| US9147847B2 (en) | 2009-05-29 | 2015-09-29 | Idemitsu Kosan Co., Ltd. | Anthracene derivative and organic electroluminescent element using the same |
| US9373792B2 (en) | 2009-05-29 | 2016-06-21 | Idemitsu Kosan Co., Ltd. | Anthracene derivative and organic electroluminescent element using the same |
| KR101127579B1 (ko) | 2009-08-28 | 2012-03-23 | 삼성모바일디스플레이주식회사 | 헤테로아릴아민 화합물 및 이를 이용한 유기 발광 소자 |
| KR101137385B1 (ko) | 2009-08-28 | 2012-04-20 | 삼성모바일디스플레이주식회사 | 헤테로아릴아민 화합물 및 이를 이용한 유기 발광 소자 |
| US8399880B2 (en) | 2009-08-28 | 2013-03-19 | Samsung Display Co., Ltd. | Heteroarylamine compound and organic light-emitting device including the same |
| US8911885B2 (en) | 2009-08-28 | 2014-12-16 | Samsung Display Co., Ltd. | Heteroarylamine compound and organic luminescence device using the same |
| WO2011081451A3 (ko) * | 2009-12-30 | 2011-12-01 | 주식회사 두산 | 트리페닐렌계 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| KR101196142B1 (ko) | 2009-12-30 | 2012-10-30 | 주식회사 두산 | 트리페닐렌계 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| KR101298465B1 (ko) * | 2011-01-04 | 2013-08-23 | 주식회사 두산 | 페나진계 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| KR20140020208A (ko) * | 2012-08-07 | 2014-02-18 | 주식회사 동진쎄미켐 | 아크리딘 유도체를 포함하는 유기발광 화합물 및 이를 포함하는 유기발광소자 |
| KR102160946B1 (ko) * | 2012-08-07 | 2020-09-29 | 주식회사 동진쎄미켐 | 아크리딘 유도체를 포함하는 유기발광 화합물 및 이를 포함하는 유기발광소자 |
| KR20150037119A (ko) * | 2013-09-30 | 2015-04-08 | 주식회사 두산 | 유기 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| KR101603383B1 (ko) | 2013-09-30 | 2016-03-14 | 주식회사 두산 | 유기 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| WO2015047018A1 (ko) * | 2013-09-30 | 2015-04-02 | 주식회사 두산 | 유기 화합물 및 이를 포함하는 유기 전계 발광 소자 |
| KR101539730B1 (ko) * | 2014-08-11 | 2015-07-28 | 성균관대학교산학협력단 | 유기 발광 화합물, 이의 제조 방법, 및 이를 포함하는 유기 전계 발광 소자 |
| KR20190127622A (ko) | 2015-01-20 | 2019-11-13 | 에스에프씨 주식회사 | 유기발광소자용 화합물 및 이를 포함하는 유기 발광 소자 |
| KR20200139113A (ko) | 2015-01-20 | 2020-12-11 | 에스에프씨 주식회사 | 유기발광소자용 화합물 및 이를 포함하는 유기 발광 소자 |
| WO2016117848A1 (ko) * | 2015-01-20 | 2016-07-28 | 에스에프씨 주식회사 | 유기발광소자용 화합물 및 이를 포함하는 유기 발광 소자 |
| CN107108545B (zh) * | 2015-01-20 | 2020-10-13 | Sfc株式会社 | 有机发光元件用化合物及包括此的有机发光元件 |
| CN107108545A (zh) * | 2015-01-20 | 2017-08-29 | Sfc株式会社 | 有机发光元件用化合物及包括此的有机发光元件 |
| KR20160089693A (ko) | 2015-01-20 | 2016-07-28 | 에스에프씨 주식회사 | 유기발광소자용 화합물 및 이를 포함하는 유기 발광 소자 |
| US10562876B2 (en) | 2015-01-20 | 2020-02-18 | Sfc Co., Ltd. | Organic compounds for organic light-emitting diode and organic light-emitting diode including the same |
| KR20160112111A (ko) | 2015-03-18 | 2016-09-28 | 에스에프씨 주식회사 | 유기발광소자용 화합물 및 이를 포함하는 유기 발광 소자 |
| KR20200124636A (ko) | 2015-04-23 | 2020-11-03 | 에스에프씨 주식회사 | 유기 발광 소자용 화합물 및 이를 포함하는 유기발광소자 |
| KR20160126873A (ko) | 2015-04-23 | 2016-11-02 | 에스에프씨 주식회사 | 유기 발광 소자용 화합물 및 이를 포함하는 유기발광소자 |
| KR102176843B1 (ko) * | 2015-04-23 | 2020-11-10 | 에스에프씨주식회사 | 유기 발광 소자용 화합물 및 이를 포함하는 유기발광소자 |
| WO2016171429A3 (ko) * | 2015-04-23 | 2016-12-15 | 에스에프씨 주식회사 | 유기 발광 소자용 화합물 및 이를 포함하는 유기발광소자 |
| CN107531661A (zh) * | 2015-04-23 | 2018-01-02 | Sfc株式会社 | 有机发光元件用化合物以及包括该化合物的有机发光元件 |
| CN107531661B (zh) * | 2015-04-23 | 2024-05-28 | Sfc株式会社 | 有机发光元件用化合物以及包括该化合物的有机发光元件 |
| US10693084B2 (en) | 2015-04-23 | 2020-06-23 | Sfc Co., Ltd. | Compound for organic light-emitting device and organic light-emitting device including same |
| KR102393196B1 (ko) * | 2015-04-23 | 2022-05-02 | 에스에프씨주식회사 | 유기 발광 소자용 화합물 및 이를 포함하는 유기발광소자 |
| WO2017010749A1 (ko) * | 2015-07-13 | 2017-01-19 | 에스에프씨 주식회사 | 고효율의 유기 발광 소자 |
| KR102799374B1 (ko) * | 2015-07-29 | 2025-04-22 | 메르크 파텐트 게엠베하 | 플루오렌 구조를 갖는 화합물 |
| KR20180030914A (ko) * | 2015-07-29 | 2018-03-26 | 메르크 파텐트 게엠베하 | 플루오렌 구조를 갖는 화합물 |
| US10818851B2 (en) | 2015-07-31 | 2020-10-27 | Samsung Display Co., Ltd. | Condensed cyclic compound and organic light-emitting device including the same |
| US12225815B2 (en) | 2016-10-07 | 2025-02-11 | Samsung Sdi Co., Ltd. | Composition for organic optoelectronic device, organic optoelectronic device, and display device |
| US12289993B2 (en) | 2016-10-07 | 2025-04-29 | Samsung Sdi Co., Ltd. | Composition for organic optoelectronic device, organic optoelectronic device, and display device |
| US12310232B2 (en) | 2016-10-07 | 2025-05-20 | Samsung Sdi Co., Ltd. | Composition for organic optoelectronic device, organic optoelectronic device, and display device |
| KR20200040225A (ko) | 2017-08-14 | 2020-04-17 | 이데미쓰 고산 가부시키가이샤 | 유기 일렉트로루미네센스 소자 및 전자 기기 |
| US10103338B1 (en) | 2017-08-14 | 2018-10-16 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device and electronic device |
| US10109803B1 (en) | 2017-08-14 | 2018-10-23 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device and electronic device |
| WO2019035412A1 (ja) | 2017-08-14 | 2019-02-21 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子及び電子機器 |
| US10109804B1 (en) | 2017-08-14 | 2018-10-23 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device and electronic device |
| US11665962B2 (en) | 2017-08-14 | 2023-05-30 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence element and electronic device |
| US10800796B2 (en) | 2017-09-21 | 2020-10-13 | Samsung Display Co., Ltd. | Aromatic compound and organic electroluminescence device including the same |
| US11787822B2 (en) | 2017-09-21 | 2023-10-17 | Samsung Display Co., Ltd. | Aromatic compound and organic electroluminescence device including the same |
| US11251380B2 (en) | 2018-01-08 | 2022-02-15 | Samsung Display Co., Ltd. | Organic electroluminescence device and polycyclic compound for organic electroluminescence device |
| WO2019235902A1 (ko) * | 2018-06-08 | 2019-12-12 | 주식회사 엘지화학 | 다환 화합물 및 이를 포함하는 유기전자소자 |
| KR20190033504A (ko) | 2019-03-20 | 2019-03-29 | 에스에프씨 주식회사 | 유기 발광 소자용 화합물 및 이를 포함하는 유기발광소자 |
| KR102118011B1 (ko) * | 2019-03-20 | 2020-06-04 | 에스에프씨주식회사 | 유기 발광 소자용 화합물 및 이를 포함하는 유기발광소자 |
| WO2021086099A1 (ko) * | 2019-10-30 | 2021-05-06 | 주식회사 엘지화학 | 안트라센 화합물 및 이를 포함하는 유기 발광 소자 |
| US12453284B2 (en) | 2019-10-30 | 2025-10-21 | Lg Chem, Ltd. | Anthracene compound and organic light-emitting device comprising same |
Also Published As
| Publication number | Publication date |
|---|---|
| JP5730468B2 (ja) | 2015-06-10 |
| CN101560136A (zh) | 2009-10-21 |
| TW200944575A (en) | 2009-11-01 |
| US20100045170A1 (en) | 2010-02-25 |
| CN103214337A (zh) | 2013-07-24 |
| JP2009249378A (ja) | 2009-10-29 |
| EP2108689A3 (en) | 2010-03-17 |
| EP2108689A2 (en) | 2009-10-14 |
| CN101560136B (zh) | 2014-09-17 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR100910150B1 (ko) | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 발광 소자 | |
| KR100901887B1 (ko) | 신규한 유기 발광 화합물 및 이를 채용하고 있는 유기 발광소자 | |
| KR100946411B1 (ko) | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 발광 소자 | |
| KR101376530B1 (ko) | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고 있는 유기 발광 소자 | |
| KR101178219B1 (ko) | 전기발광화합물을 발광재료로서 채용하고 있는 전기발광소자 | |
| KR20100000121A (ko) | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 발광 소자 | |
| KR100989815B1 (ko) | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 발광 소자 | |
| KR20090111915A (ko) | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 발광 소자 | |
| KR101495547B1 (ko) | 신규한 전자 재료용 화합물 및 이를 포함하는 유기 전자소자 | |
| EP2281863A2 (en) | Fluorene-derivatives and organic electroluminescent device using the same | |
| KR20100048203A (ko) | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고 있는 유기 발광 소자 | |
| KR20090098585A (ko) | 유기발광화합물을 발광재료로서 채용하고 있는유기전기발광소자 | |
| KR20100048447A (ko) | 신규한 유기 전자재료용 화합물 및 이를 포함하는 유기 전자 소자 | |
| KR20100028168A (ko) | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고 있는 유기 발광 소자 | |
| JP2015120692A (ja) | 新規な有機電界発光化合物及びこれを使用している有機電界発光素子 | |
| KR20100048210A (ko) | 신규한 유기 발광 화합물 및 이를 포함하는 유기 발광 소자 | |
| KR20110121147A (ko) | 신규한 유기 발광 화합물 및 이를 포함하는 유기 발광 소자 | |
| KR20090105495A (ko) | 신규한 유기 발광 화합물 및 이를 발광재료로서 채용하고있는 유기 전기 발광 소자 | |
| KR20100130059A (ko) | 신규한 유기 발광 화합물 및 이를 포함하는 유기 발광 소자 | |
| KR100936400B1 (ko) | 신규한 유기 발광 화합물 및 이를 채용하고 있는 유기 발광소자 | |
| KR20100086972A (ko) | 유기발광화합물을 발광재료로서 채용하고 있는 유기전기발광소자 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0109 | Patent application |
St.27 status event code: A-0-1-A10-A12-nap-PA0109 |
|
| A201 | Request for examination | ||
| A302 | Request for accelerated examination | ||
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| PA0302 | Request for accelerated examination |
St.27 status event code: A-1-2-D10-D17-exm-PA0302 St.27 status event code: A-1-2-D10-D16-exm-PA0302 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| T11-X000 | Administrative time limit extension requested |
St.27 status event code: U-3-3-T10-T11-oth-X000 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U11-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R13-asn-PN2301 St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 4 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R11-asn-PN2301 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-5-5-R10-R14-asn-PN2301 |
|
| FPAY | Annual fee payment |
Payment date: 20130701 Year of fee payment: 5 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 5 |
|
| FPAY | Annual fee payment |
Payment date: 20140701 Year of fee payment: 6 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 6 |
|
| FPAY | Annual fee payment |
Payment date: 20150619 Year of fee payment: 7 |
|
| PR1001 | Payment of annual fee |
St.27 status event code: A-4-4-U10-U11-oth-PR1001 Fee payment year number: 7 |
|
| LAPS | Lapse due to unpaid annual fee | ||
| PC1903 | Unpaid annual fee |
St.27 status event code: A-4-4-U10-U13-oth-PC1903 Not in force date: 20160729 Payment event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: N-4-6-H10-H13-oth-PC1903 Ip right cessation event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE Not in force date: 20160729 |
|
| P22-X000 | Classification modified |
St.27 status event code: A-4-4-P10-P22-nap-X000 |