JP2022022419A - CFTR mRNA組成物ならびに関連する方法及び使用 - Google Patents
CFTR mRNA組成物ならびに関連する方法及び使用 Download PDFInfo
- Publication number
- JP2022022419A JP2022022419A JP2021200634A JP2021200634A JP2022022419A JP 2022022419 A JP2022022419 A JP 2022022419A JP 2021200634 A JP2021200634 A JP 2021200634A JP 2021200634 A JP2021200634 A JP 2021200634A JP 2022022419 A JP2022022419 A JP 2022022419A
- Authority
- JP
- Japan
- Prior art keywords
- cftr
- mrna
- seq
- expression
- lung
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/54—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being an organic compound
- A61K47/543—Lipids, e.g. triglycerides; Polyamines, e.g. spermine or spermidine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/713—Double-stranded nucleic acids or oligonucleotides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/7105—Natural ribonucleic acids, i.e. containing only riboses attached to adenine, guanine, cytosine or uracil and having 3'-5' phosphodiester links
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/70—Carbohydrates; Sugars; Derivatives thereof
- A61K31/7088—Compounds having three or more nucleosides or nucleotides
- A61K31/7115—Nucleic acids or oligonucleotides having modified bases, i.e. other than adenine, guanine, cytosine, uracil or thymine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/69—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit
- A61K47/6921—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a particulate, a powder, an adsorbate, a bead or a sphere
- A61K47/6927—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a particulate, a powder, an adsorbate, a bead or a sphere the form being a solid microparticle having no hollow or gas-filled cores
- A61K47/6929—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a particulate, a powder, an adsorbate, a bead or a sphere the form being a solid microparticle having no hollow or gas-filled cores the form being a nanoparticle, e.g. an immuno-nanoparticle
- A61K47/6931—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a particulate, a powder, an adsorbate, a bead or a sphere the form being a solid microparticle having no hollow or gas-filled cores the form being a nanoparticle, e.g. an immuno-nanoparticle the material constituting the nanoparticle being a polymer
- A61K47/6935—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the conjugate being characterised by physical or galenical forms, e.g. emulsion, particle, inclusion complex, stent or kit the form being a particulate, a powder, an adsorbate, a bead or a sphere the form being a solid microparticle having no hollow or gas-filled cores the form being a nanoparticle, e.g. an immuno-nanoparticle the material constituting the nanoparticle being a polymer the polymer being obtained otherwise than by reactions involving carbon to carbon unsaturated bonds, e.g. polyesters, polyamides or polyglycerol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
- A61K48/005—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy characterised by an aspect of the 'active' part of the composition delivered, i.e. the nucleic acid delivered
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/007—Pulmonary tract; Aromatherapy
- A61K9/0073—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/007—Pulmonary tract; Aromatherapy
- A61K9/0073—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy
- A61K9/0078—Sprays or powders for inhalation; Aerolised or nebulised preparations generated by other means than thermal energy for inhalation via a nebulizer such as a jet nebulizer, ultrasonic nebulizer, e.g. in the form of aqueous drug solutions or dispersions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Synthetic bilayered vehicles, e.g. liposomes or liposomes with cholesterol as the only non-phosphatidyl surfactant
- A61K9/1271—Non-conventional liposomes, e.g. PEGylated liposomes or liposomes coated or grafted with polymers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/48—Preparations in capsules, e.g. of gelatin, of chocolate
- A61K9/50—Microcapsules having a gas, liquid or semi-solid filling; Solid microparticles or pellets surrounded by a distinct coating layer, e.g. coated microspheres, coated drug crystals
- A61K9/51—Nanocapsules; Nanoparticles
- A61K9/5107—Excipients; Inactive ingredients
- A61K9/5123—Organic compounds, e.g. fats, sugars
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P11/00—Drugs for disorders of the respiratory system
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H21/00—Compounds containing two or more mononucleotide units having separate phosphate or polyphosphate groups linked by saccharide radicals of nucleoside groups, e.g. nucleic acids
- C07H21/02—Compounds containing two or more mononucleotide units having separate phosphate or polyphosphate groups linked by saccharide radicals of nucleoside groups, e.g. nucleic acids with ribosyl as saccharide radical
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/46—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates
- C07K14/47—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals
- C07K14/4701—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans from vertebrates from mammals not used
- C07K14/4712—Cystic fibrosis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/87—Introduction of foreign genetic material using processes not otherwise provided for, e.g. co-transformation
- C12N15/88—Introduction of foreign genetic material using processes not otherwise provided for, e.g. co-transformation using microencapsulation, e.g. using amphiphile liposome vesicle
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N9/00—Enzymes; Proenzymes; Compositions thereof; Processes for preparing, activating, inhibiting, separating or purifying enzymes
- C12N9/14—Hydrolases (3)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12Y—ENZYMES
- C12Y306/00—Hydrolases acting on acid anhydrides (3.6)
- C12Y306/03—Hydrolases acting on acid anhydrides (3.6) acting on acid anhydrides; catalysing transmembrane movement of substances (3.6.3)
- C12Y306/03049—Channel-conductance-controlling ATPase (3.6.3.49)
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/10—Dispersions; Emulsions
- A61K9/127—Synthetic bilayered vehicles, e.g. liposomes or liposomes with cholesterol as the only non-phosphatidyl surfactant
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H21/00—Compounds containing two or more mononucleotide units having separate phosphate or polyphosphate groups linked by saccharide radicals of nucleoside groups, e.g. nucleic acids
- C07H21/04—Compounds containing two or more mononucleotide units having separate phosphate or polyphosphate groups linked by saccharide radicals of nucleoside groups, e.g. nucleic acids with deoxyribosyl as saccharide radical
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Genetics & Genomics (AREA)
- Organic Chemistry (AREA)
- Epidemiology (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Zoology (AREA)
- Biotechnology (AREA)
- Biomedical Technology (AREA)
- Wood Science & Technology (AREA)
- Nanotechnology (AREA)
- General Engineering & Computer Science (AREA)
- Biophysics (AREA)
- Pulmonology (AREA)
- Immunology (AREA)
- Physics & Mathematics (AREA)
- Dispersion Chemistry (AREA)
- Microbiology (AREA)
- Otolaryngology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Toxicology (AREA)
- Gastroenterology & Hepatology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Cell Biology (AREA)
- Optics & Photonics (AREA)
- Plant Pathology (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
Description
本出願は、2013年3月14日出願の米国仮出願第61/783,663号の優先権を主張するものであり、その開示は、参照により本明細書に組み込まれる。
PCT特許公開第WO2007/024708号ならびに米国特許公開第2010/0203627号及び同第2011/0035819号は、CFTR mRNAの治療的投与を記述するが、CFTR mRNAの投与後の肺における機能的CFTRの産生の実践に対する実証された低減、または、インビトロ転写CFTR mRNAを使用する肺におけるCFTR発現の誘導に関連する難点を克服するための十分な助言のいずれも提供しない。これらは、mRNAのインビトロ合成の達成に伴う難点、及びmRNA組成物と肺特異性物質との相互作用に特異的な難点を含み、上記のAndriesらなどの研究者は、対応するDNAベースの組成物がいくらかのレベルの発現を提供したにも関わらず、これらの難点によりmRNA組成物が発現の誘導に無効なものとなっていることを見出している。
したがって、嚢胞性線維症の治療のための、哺乳動物の肺における誘導を含む、CFTR発現の誘導のためのCFTR mRNAの改善された材料、製剤、産生方法、及び送達方法が必要とされている。
mRNAの作製方法を提供し、該単離ポリヌクレオチド及びRNAポリメラーゼは、細胞中に含有されず、該単離ポリヌクレオチドは、該RNAポリメラーゼのための鋳型であり、該単離ポリヌクレオチドは、鋳型配列に作動可能に連結されたプロモーターを含み、該RNAポリメラーゼは、配列番号1をコードするコード配列を含むmRNAを合成する。
本発明は、例えば、以下の項目も提供する。
(項目1)
哺乳動物の肺における上皮細胞中のCFTR発現の誘導方法であって、
前記方法が、前記哺乳動物の前記肺における前記上皮細胞を組成物と接触させることを含み、
前記組成物が、インビトロ転写mRNAを含む薬学的組成物であり、
前記インビトロ転写mRNAが、配列番号1をコードするコード配列を含む、前記方法。
(項目2)
哺乳動物の標的細胞中のCFTR発現の誘導方法であって、
前記方法が、前記哺乳動物の標的細胞を組成物と接触させることを含み、
前記組成物が、配列番号1のアミノ酸配列をコードするインビトロ転写mRNAを含む、前記方法。
(項目3)
(a)前記インビトロ転写mRNA配列が、配列番号2よりも少ないクリプティックプロモーター(cryptic promoter)の補体を含む、
(b)前記インビトロ転写mRNA配列が、配列番号2よりも少ない直列反復及び/もしくは逆方向反復を含む、
(c)前記コード配列が、配列番号2よりも少ない不利なコドンを含む、かつ/または、
(d)前記コード配列のGC含量が、配列番号2のGC含量よりも低い、項目1または項目2に記載の方法。
(項目4)
コード配列、5’-UTR、及び3’-UTRを含み、前記コード配列が、配列番号1のアミノ酸配列をコードし、前記コード配列が、配列番号3と少なくとも80%同一である、非自然発生的mRNA分子。
(項目5)
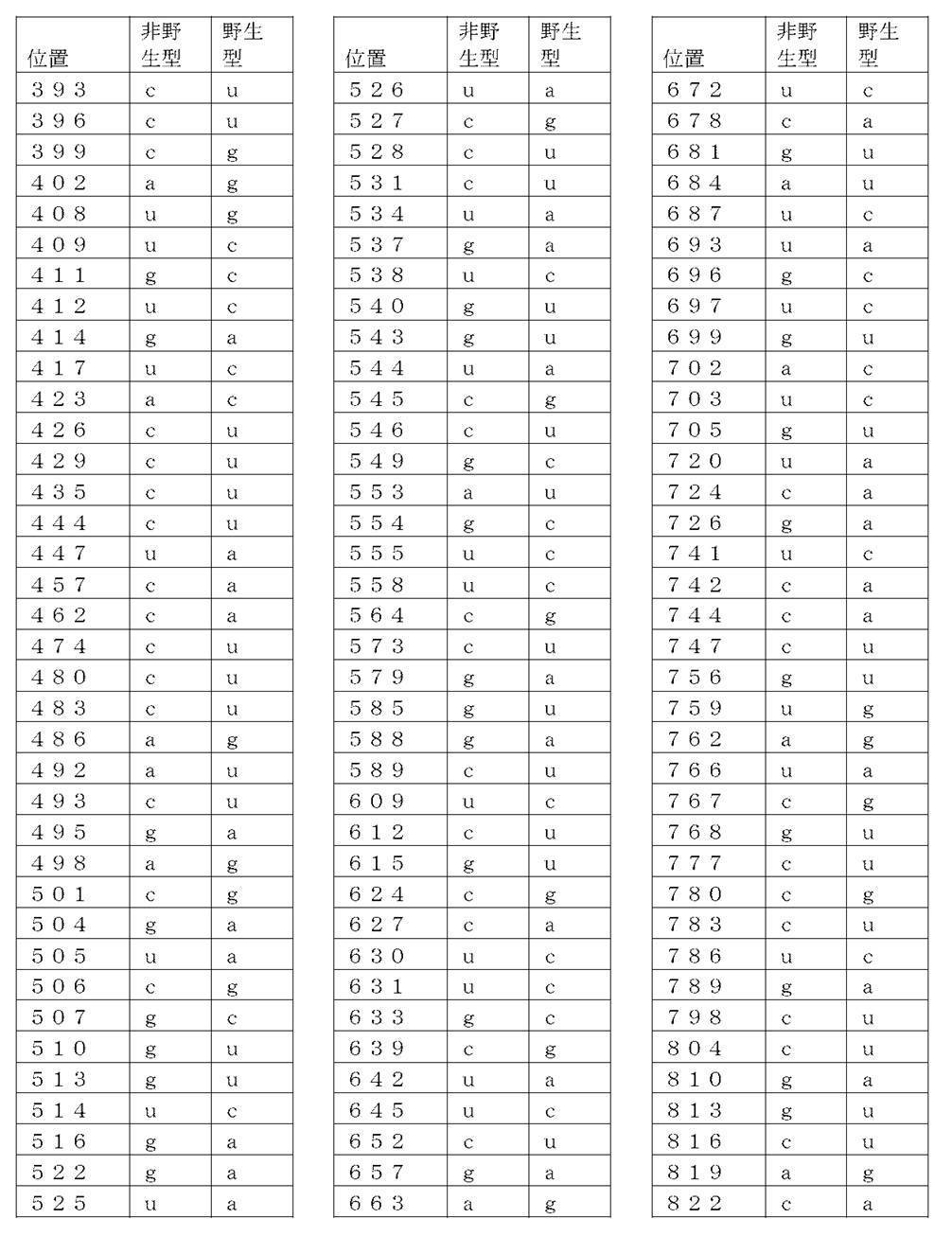
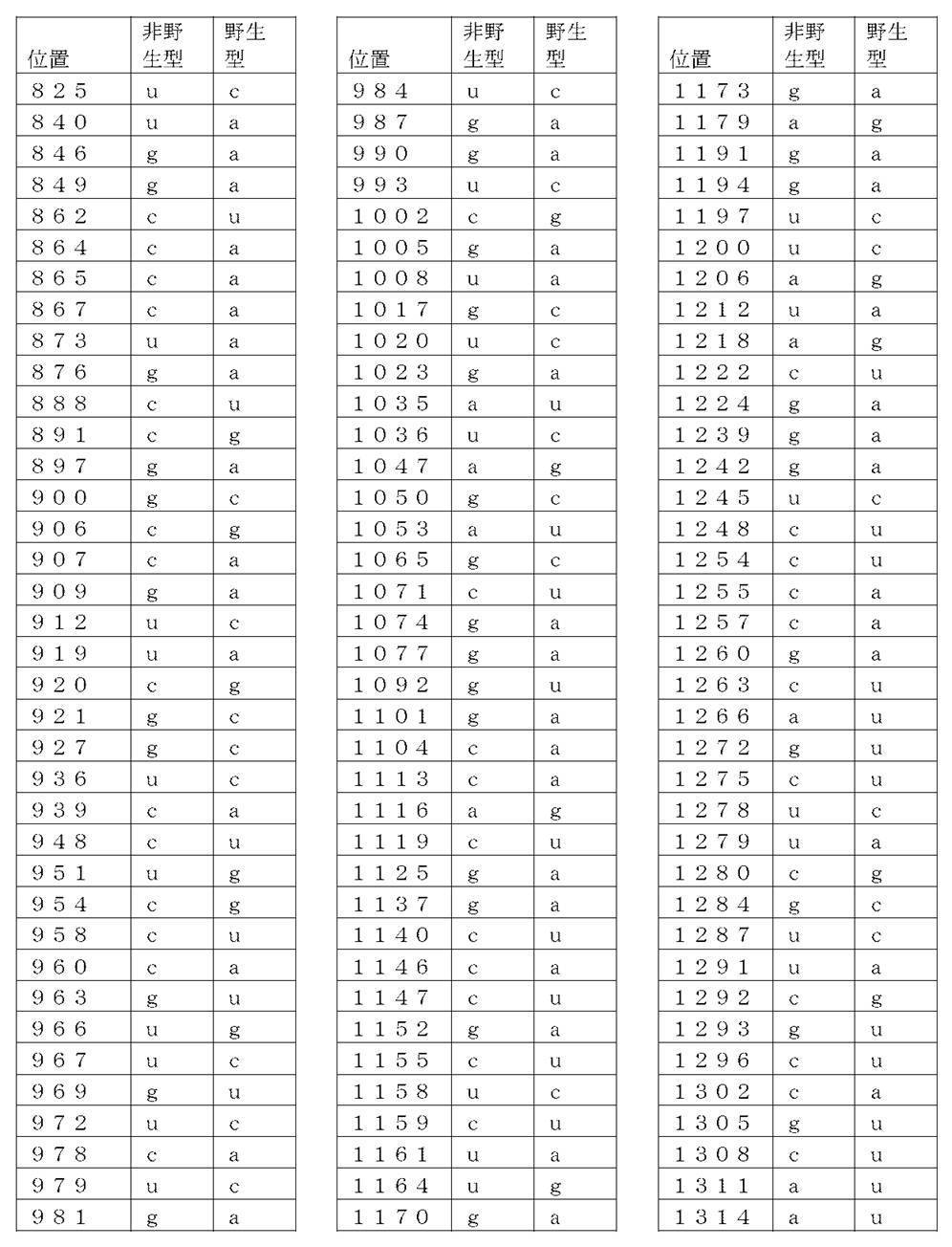
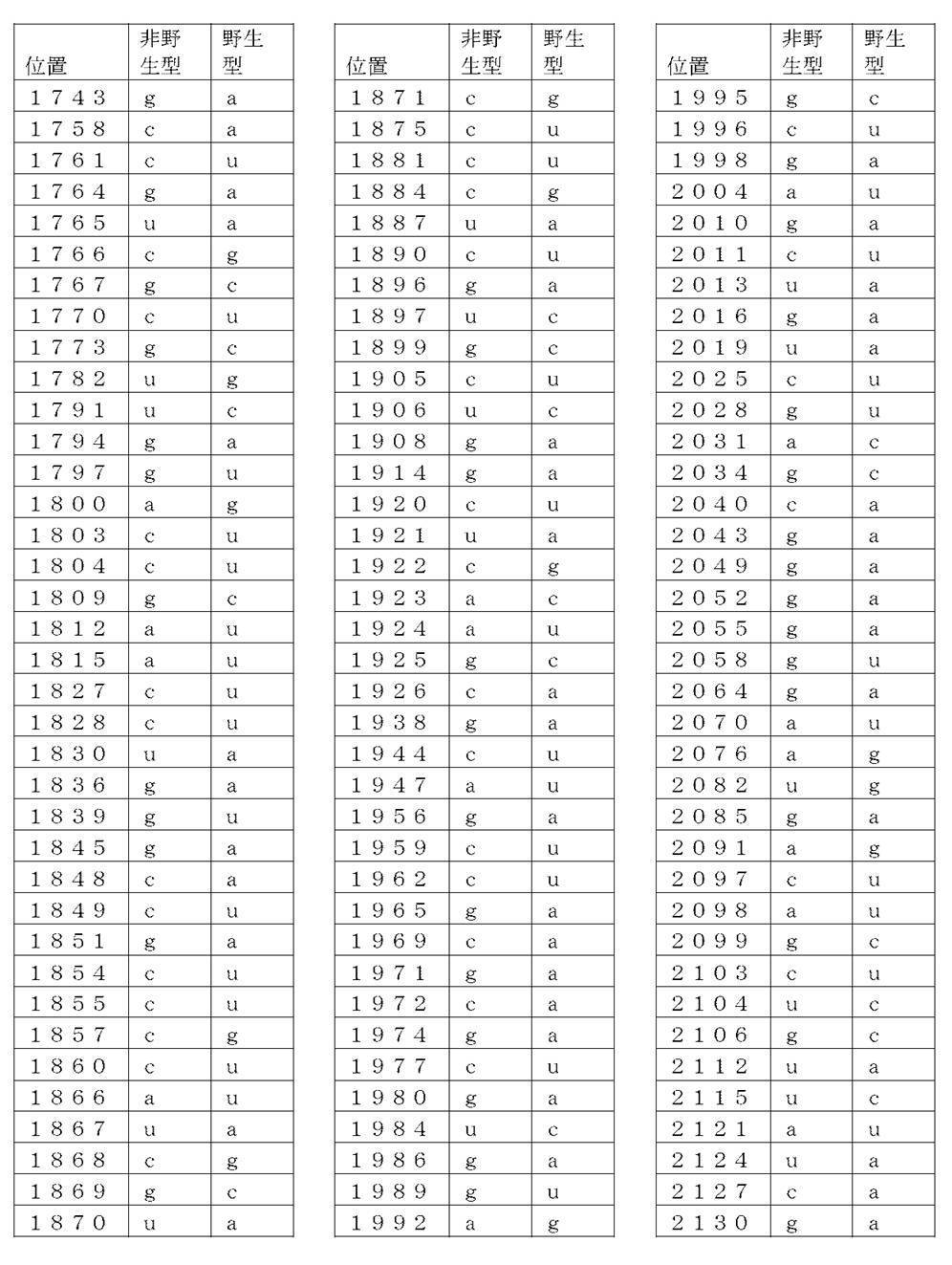
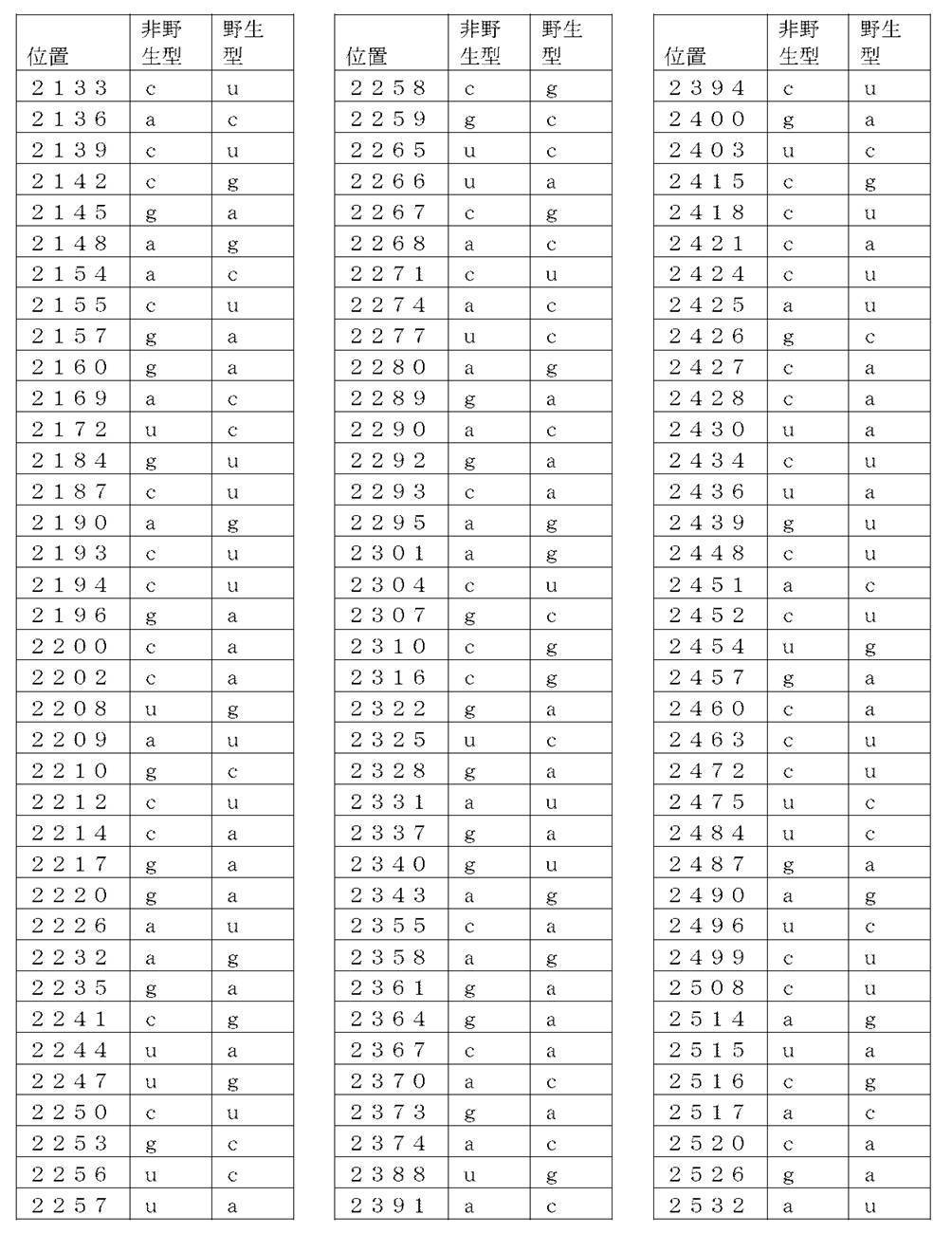
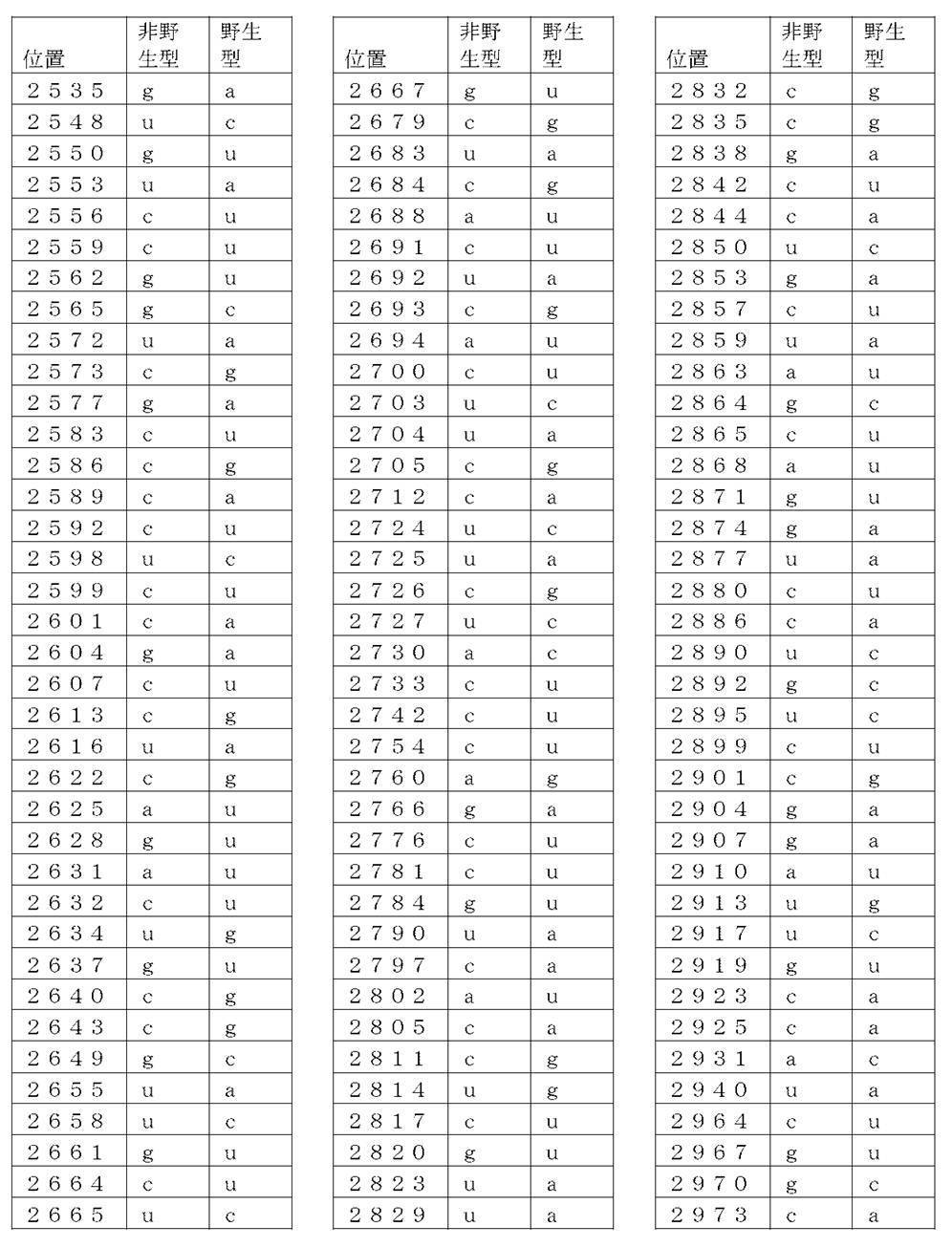
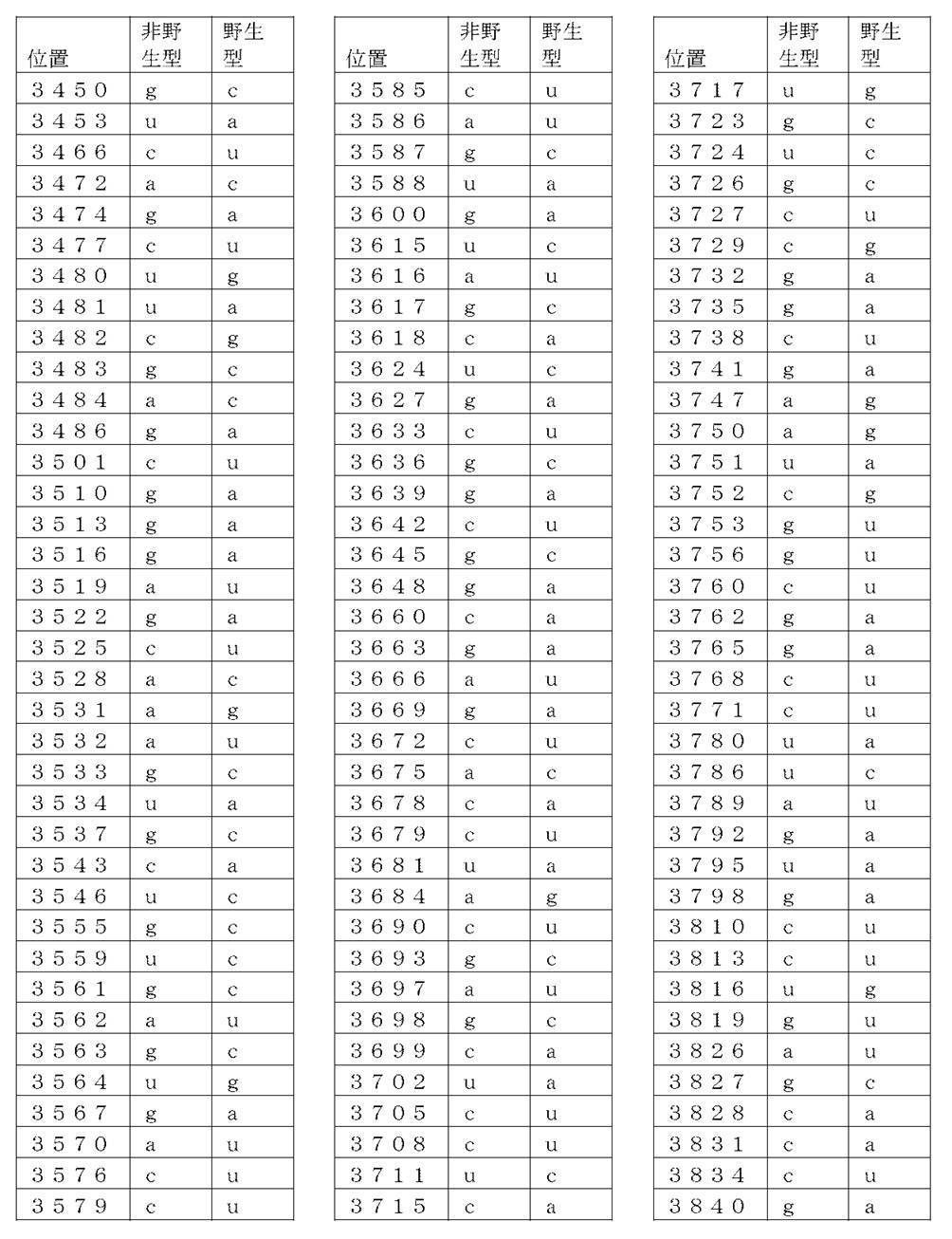
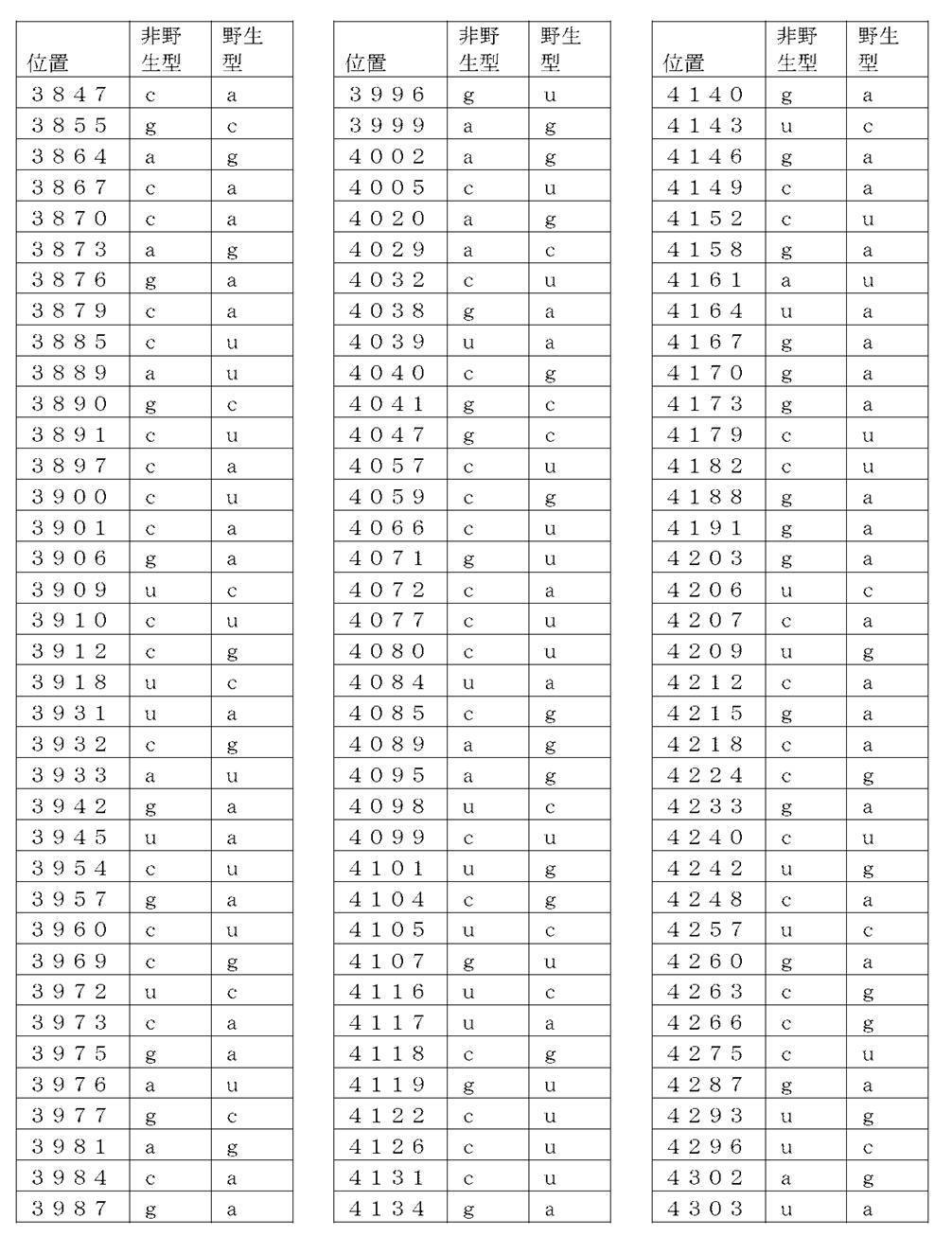
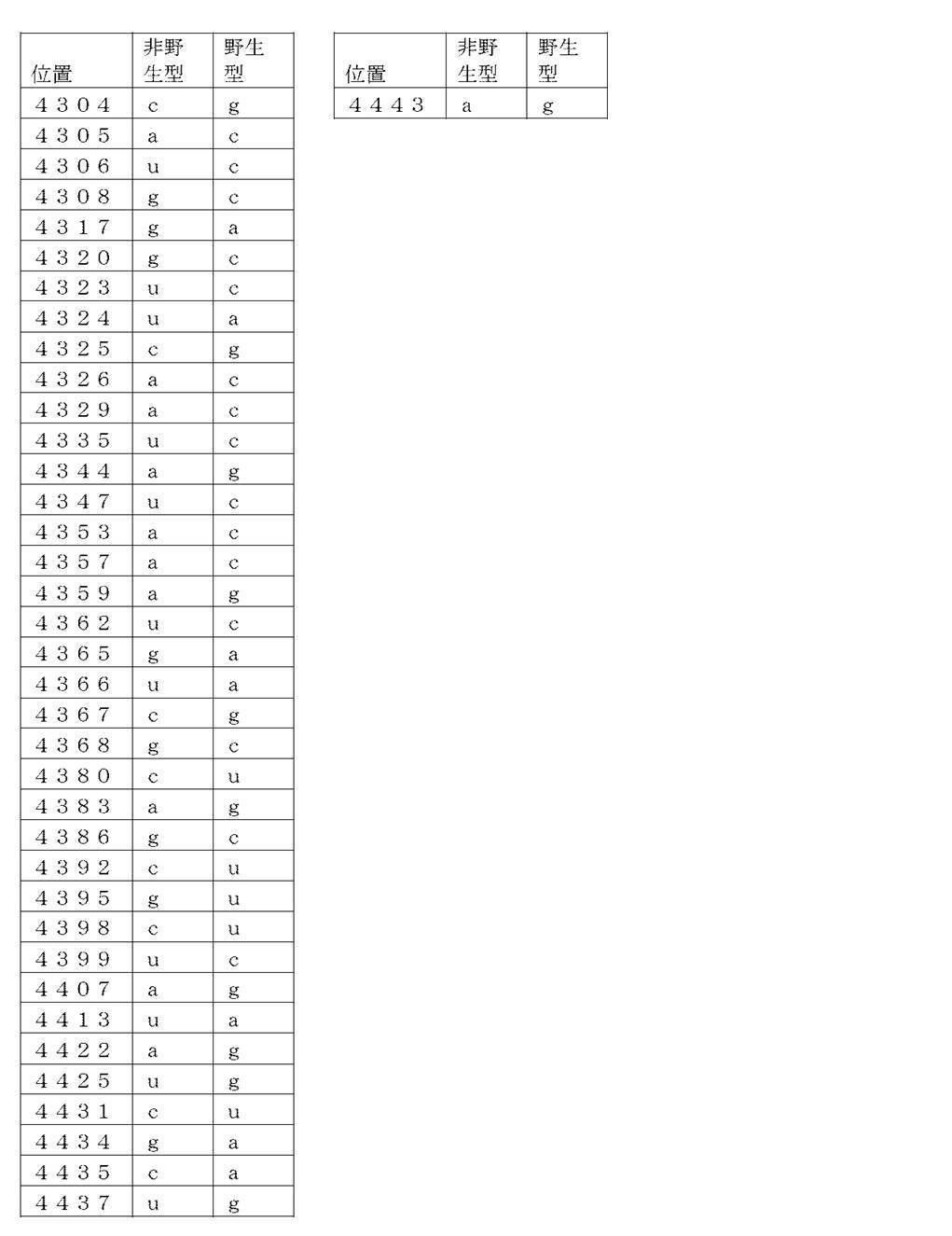
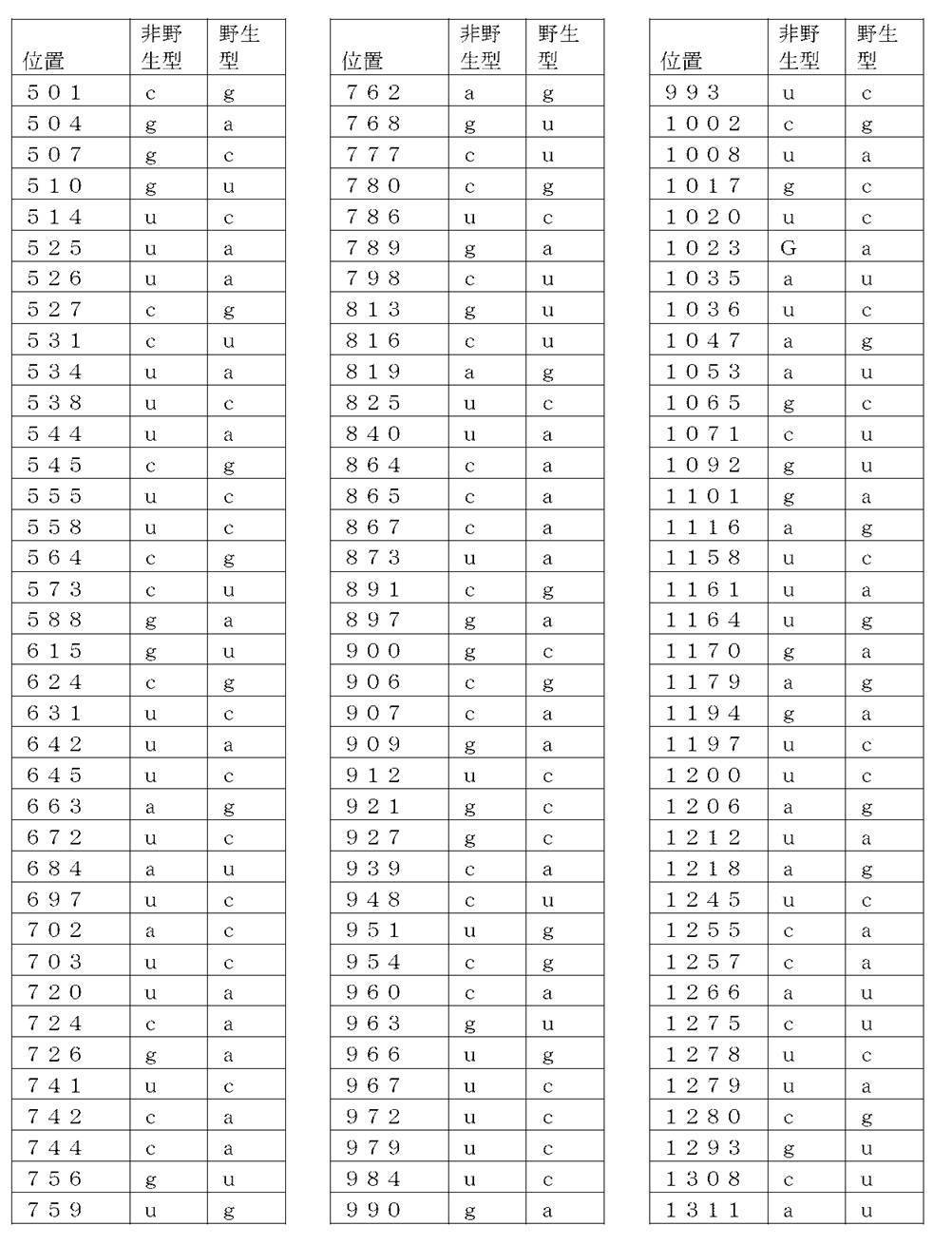
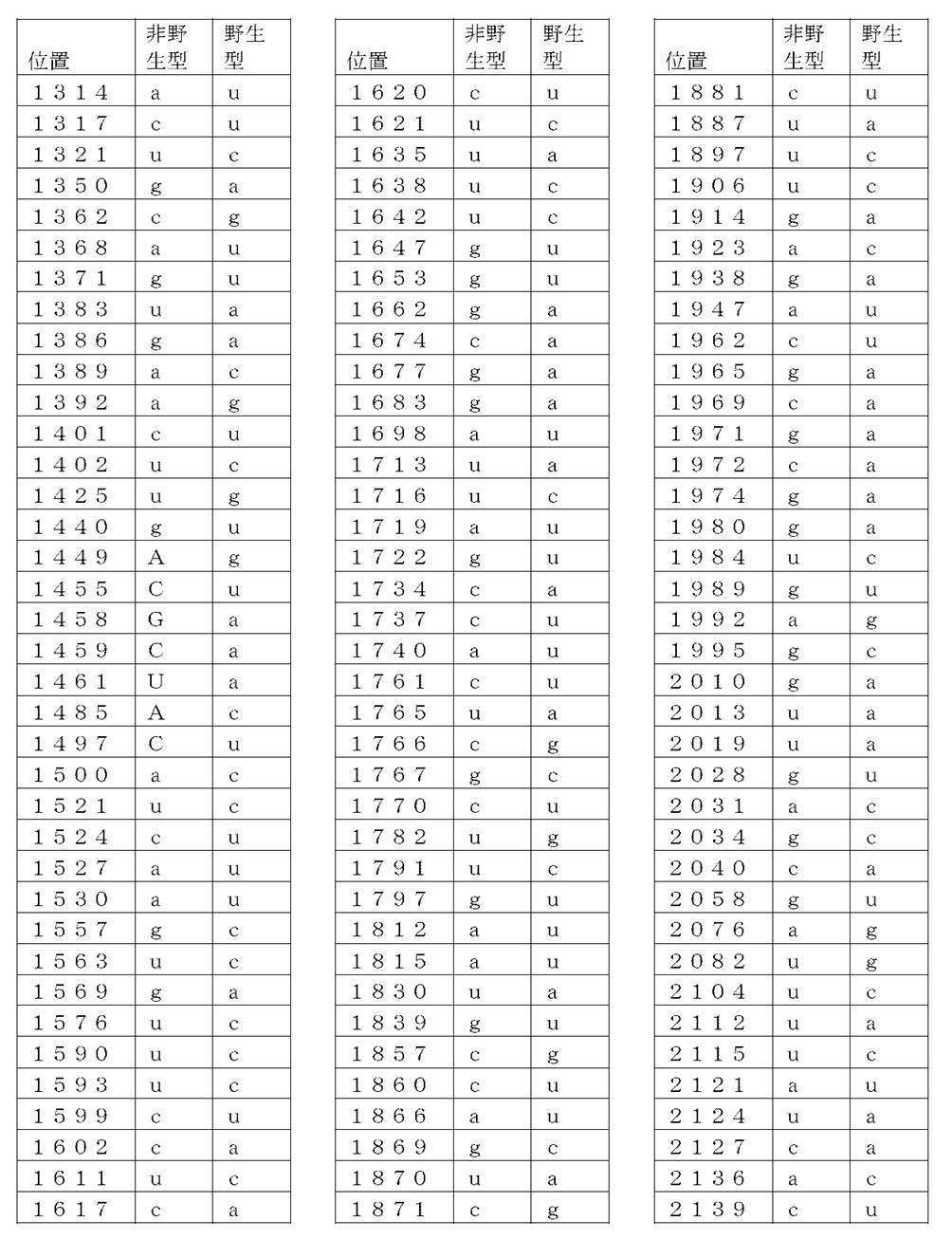
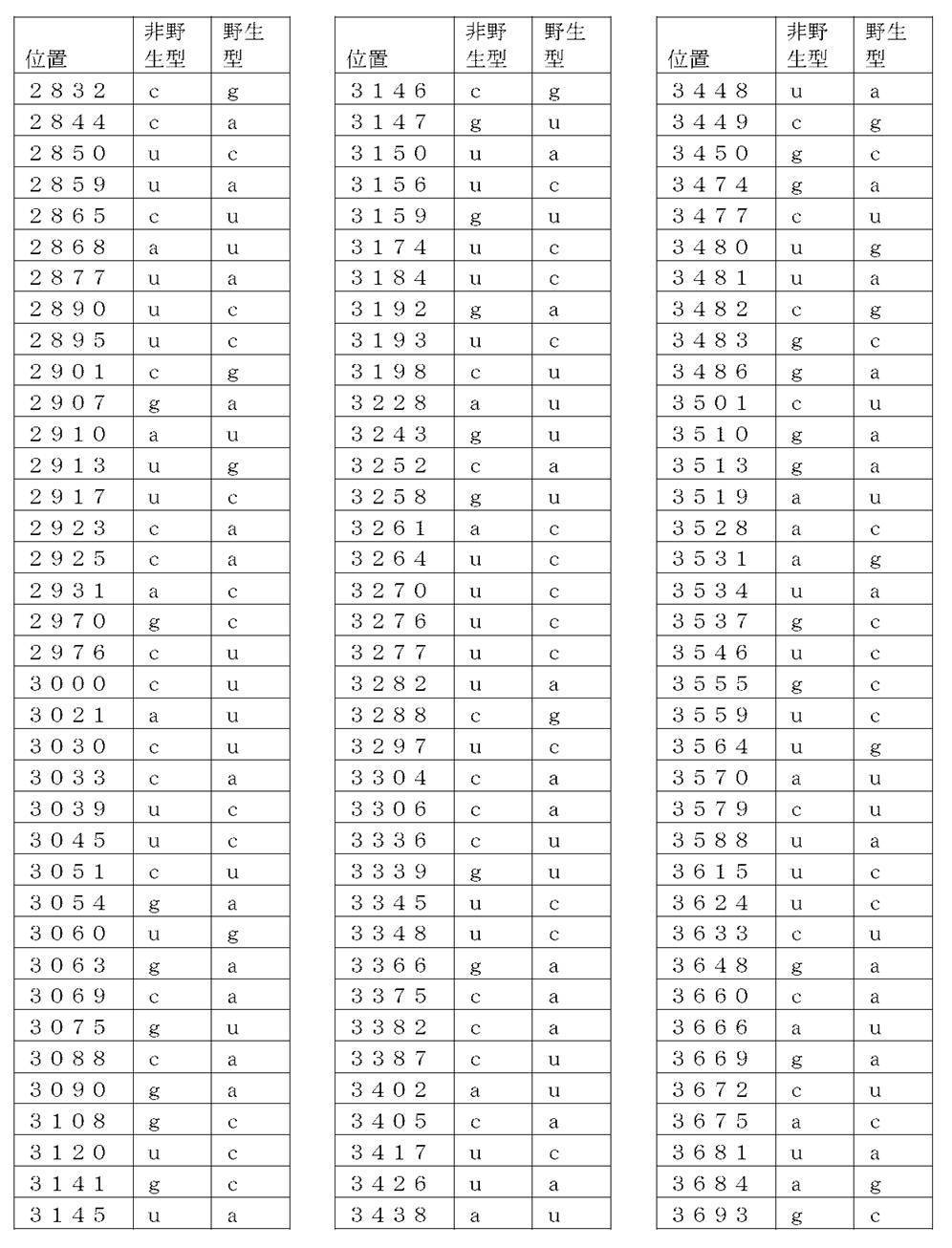
コード配列、5’-UTR、及び3’-UTRを含み、前記コード配列が、配列番号1のアミノ酸配列をコードし、前記コード配列が、配列番号2の野生型コード配列と比べて、少なくとも50%、少なくとも60%、少なくとも70%、少なくとも80%、少なくとも90%、または少なくとも95%の表1に列記される非野生型塩基を、表1に列記されるコード配列の位置において含む、非自然発生的mRNA分子。
(項目6)
コード配列、5’-UTR、及び3’-UTRを含み、前記コード配列が、配列番号1のアミノ酸配列をコードし、前記コード配列が、配列番号2の前記野生型コード配列と比べて、少なくとも50%、少なくとも60%、少なくとも70%、少なくとも80%、少なくとも90%、または少なくとも95%の表2に列記される非野生型塩基を、表2に列記されるコード配列の対応する位置において含む、非自然発生的mRNA分子。
(項目7)
前記コード配列が、配列番号3と少なくとも85%、少なくとも90%、少なくとも95%、少なくとも98%、または少なくとも99%同一である、項目4~6のいずれか1項に記載のmRNA分子。
(項目8)
前記コード配列が配列番号3と同一である、項目4に記載のmRNA分子。
(項目9)
前記5’-UTRが、配列番号4を含む、かつ/または前記3’-UTRが、配列番号5を含む、項目4~8のいずれか1項に記載のmRNA分子。
(項目10)
少なくとも70、100、120、150、200、または250残基長のポリAテールをさらに含む、項目4~9のいずれか1項に記載のmRNA分子。
(項目11)
5’キャップをさらに含む、項目4~10のいずれか1項に記載のmRNA分子。
(項目12)
少なくとも1つの構成ヌクレオチドが、ロックド核酸残基である、項目4~11のいずれか1項に記載のmRNA分子。
(項目13)
前記mRNAが、少なくとも1つの非標準核酸塩基を含む、項目4~12のいずれか1項に記載のmRNA分子。
(項目14)
前記非標準核酸塩基が、5-メチル-シチジン、プソイドウリジン、及び2-チオ-ウリジンのうちの1つ以上から選択される、項目13に記載のmRNA分子。
(項目15)
哺乳動物または哺乳動物の細胞における機能的CFTR発現を誘導するのに使用するための、項目4~14のいずれか1項に記載のmRNA分子。
(項目16)
項目4~15のいずれか1項に記載のmRNAの前記配列に相補的な配列を含む、ポリヌクレオチド。
(項目17)
前記ポリヌクレオチドが、デオキシリボヌクレオチド残基を含む線状または環状のポリヌクレオチドである、項目16に記載のポリヌクレオチド。
(項目18)
項目16または項目17に記載のポリヌクレオチド、RNAポリメラーゼ、及びヌクレオシド三リン酸塩を含む、組成物。
(項目19)
項目4~15のいずれか1項に記載のmRNAを含む、薬学的組成物。
(項目20)
ポリエチレンイミン(PEI)、プロタミン、PEG化プロタミン、PLL、PEG化PLL、またはカチオン性脂質から選択される有機カチオンをさらに含み、前記有機カチオンが、前記mRNAと非共有結合的に複合される、項目19に記載の薬学的組成物。
(項目21)
前記有機カチオンがカチオン性脂質であり、前記組成物が、中性脂質、PEG化脂質、及び/またはコレステロールをさらに含む、項目20に記載の薬学的組成物。
(項目22)
前記カチオン性脂質が、DODAP(1,2-ジオレイル-3-ジメチルアンモニウムプロパン)、DLinDMA、DLin-KC2-DMA、C12-200、HGT4003、HGT5000、HGT5001、MC3、ICE、ジアルキルアミノ部分を含むカチオン性脂質、イミダゾール部分を含むカチオン性脂質、及びグアニジウム部分を含むカチオン性脂質から選択される、項目21に記載の薬学的組成物。
(項目23)
前記中性脂質が、前記組成物中に存在し、かつ、DSPC(1,2-ジステアロイル-sn-グリセロ-3-ホスホコリン)、DPPC(1,2-ジパルミトイル-sn-グリセロ-3-ホスホコリン)、DOPE(1,2-ジオレイル-sn-グリセロ-3-ホスホエタノールアミン)、DPPE(1,2-ジパルミトイル-sn-グリセロ-3-ホスホエタノールアミン)、DMPE(1,2-ジミリストイル-sn-グリセロ-3-ホスホエタノールアミン)、及びDOPG(,2-ジオレオイル-sn-グリセロ-3-ホスホ-(1’-rac-グリセロール))から選択される、項目21または22のいずれか1項に記載の薬学的組成物。
(項目24)
前記PEG化脂質が、前記組成物中に存在し、かつ、長さが最大5kDaのポリ(エチレン)グリコール鎖に共有結合された、C6~C20長の1つ以上のアルキル鎖(複数可)を含む、項目21~23のいずれか1項に記載の薬学的組成物。
(項目25)
前記有機カチオンが、10kDa~40kDaの範囲の分子量を有する分岐状PEIである、項目20に記載の薬学的組成物。
(項目26)
項目19~25のいずれか1項に記載の薬学的組成物が装填された、噴霧またはエアロゾル化装置。
(項目27)
項目4~15のいずれか1項に記載のmRNAと、前記mRNAから発現される機能的CFTRと、を含む、培養細胞。
(項目28)
野生型ヒトCFTRをコードするゲノムDNAまたは野生型ヒトCFTRをコードするcDNAを含まない、項目27に記載の培養細胞。
(項目29)
機能的CFTRの発現の誘導のための、項目19~25のいずれか1項に記載の薬学的組成物の使用。
(項目30)
哺乳動物の肺における上皮細胞中のCFTR発現の誘導方法であって、
前記方法が、前記上皮細胞を組成物と接触させることを含み、
前記組成物が、項目4~15のいずれか1項に記載のmRNAを含む薬学的組成物である、前記方法。
(項目31)
哺乳動物の標的細胞中のCFTR発現の誘導方法であって、
前記方法が、前記哺乳動物の標的細胞を組成物と接触させることを含み、
前記組成物が、項目4~15のいずれか1項に記載のmRNAを含む、前記方法。
(項目32)
前記組成物が、ポリエチレンイミン(PEI)またはカチオン性脂質から選択される有機カチオンをさらに含み、前記有機カチオンが、前記インビトロ転写mRNAと非共有結合的に複合される、項目1~3または29~31のいずれか1項に記載の方法。
(項目33)
前記組成物が粘液溶解剤を含まない、項目1~3または29~32のいずれか1項に記載の方法。
(項目34)
前記有機カチオンがカチオン性脂質であり、前記組成物が、中性脂質、PEG化脂質、及び/またはコレステロールをさらに含む、項目1~3または29~33のいずれか1項に記載の方法。
(項目35)
前記カチオン性脂質が、DOTAP(1,2-ジオレイル-3-トリメチルアンモニウムプロパン)、DODAP(1,2-ジオレイル-3-ジメチルアンモニウムプロパン)、DOTMA(1,2-ジ-O-オクタデセニル-3-トリメチルアンモニウムプロパン)、DLinDMA、DLin-KC2-DMA、C12-200、HGT4003、HGT5000、HGT5001、MC3、ICE、ジアルキルアミノ部分を含むカチオン性脂質、イミダゾール部分を含むカチオン性脂質、及びグアニジウム部分を含むカチオン性脂質から選択される、項目1~3または29~34のいずれか1項に記載の方法。
(項目36)
前記中性脂質が、前記組成物中に存在し、かつ、DSPC(1,2-ジステアロイル-sn-グリセロ-3-ホスホコリン)、DPPC(1,2-ジパルミトイル-sn-グリセロ-3-ホスホコリン)、DOPE(1,2-ジオレイル-sn-グリセロ-3-ホスホエタノールアミン)、DPPE(1,2-ジパルミトイル-sn-グリセロ-3-ホスホエタノールアミン)、DMPE(1,2-ジミリストイル-sn-グリセロ-3-ホスホエタノールアミン)、及びDOPG(,2-ジオレオイル-sn-グリセロ-3-ホスホ-(1’-rac-グリセロール))から選択される、項目1~3または29~35のいずれか1項に記載の方法。
(項目37)
前記PEG化脂質が、前記組成物中に存在し、かつ、長さが最大5kDaのポリ(エチレン)グリコール鎖に共有結合された、C6~C20長の1つ以上のアルキル鎖(複数可)を含む、項目1~3または29~36のいずれか1項に記載の方法。
(項目38)
前記有機カチオンが、10kDa~40kDaの範囲の分子量を有する分岐状PEIである、項目1~3または29~37のいずれか1項に記載の方法。
(項目39)
前記組成物が、噴霧またはエアロゾル化を介して投与される、項目1~3または29~38のいずれか1項に記載の方法。
(項目40)
インビトロのCFTR mRNAの作製方法であって、ヌクレオシド三リン酸塩の存在下で、単離ポリヌクレオチドをRNAポリメラーゼと接触させることを含み、
前記単離ポリヌクレオチド及びRNAポリメラーゼが、細胞中に含有されず、
前記単離ポリヌクレオチドが、前記RNAポリメラーゼのための鋳型であり、
前記単離ポリヌクレオチドが、鋳型配列に作動可能に連結されたプロモーターを含み、
前記鋳型配列が、配列番号1をコードする配列に相補的であるコード配列補体を含み、
(a)前記鋳型配列が、配列番号2の補体よりも少ないクリプティックプロモーターを含むか、(b)前記鋳型配列が、配列番号2よりも少ない直列反復及び/もしくは逆方向反復を含むか、(c)前記鋳型配列が、配列番号2よりも少ない不利なコドンの補体を含むか、または(d)前記コード配列補体のGC含量が、配列番号2のGC含量よりも低い、前記方法。
(項目41)
インビトロのCFTR mRNAの作製方法であって、ヌクレオシド三リン酸塩の存在下で、項目16または17のいずれか1項に記載の単離ポリヌクレオチドをRNAポリメラーゼと接触させることを含み、
前記単離ポリヌクレオチド及びRNAポリメラーゼが、細胞中に含有されず、
前記単離ポリヌクレオチドが、前記RNAポリメラーゼのための鋳型であり、
前記単離ポリヌクレオチドが、鋳型配列に作動可能に連結されたプロモーターを含み、
前記RNAポリメラーゼが、配列番号1をコードするコード配列を含むmRNAを合成する、前記方法。
(項目42)
前記RNAポリメラーゼがT7 RNAポリメラーゼである、項目40または41のいずれか1項に記載の方法。
(項目43)
前記ヌクレオシド三リン酸塩が、プソイドウリジン三リン酸塩、5-メチル-シチジン三リン酸塩、及び2-チオ-ウリジン三リン酸塩のうちの1つ以上を含む、項目40~42のいずれか1項に記載の方法。
(項目44)
配列番号1をコードするコード配列を含む前記mRNAを単離することをさらに含む、項目40~43のいずれか1項に記載の方法。
(項目45)
5’キャップを前記単離mRNAに付加することをさらに含む、項目44に記載の方法。
(項目46)
前記キャップ形成された単離mRNAを、1つ以上の有機カチオンを含む1つ以上の薬学的に許容される担体と接触させることによって、薬学的組成物を製剤化することをさらに含む、項目45に記載の方法。
(項目47)
前記1つ以上の有機カチオンが、ポリエチレンイミン(PEI)、プロタミン、PEG化プロタミン、PLL、PEG化PLL、またはカチオン性脂質を含む、項目46に記載の方法。
本明細書において使用される場合、用語「ポリヌクレオチド」は、一般的に、核酸(例えば、DNAまたはRNA)を指すために使用される。ポリヌクレオチド、核酸、DNA、RNA、及びmRNAという用語は、標準的または無修飾の残基、非標準的または修飾された残基、ならびに標準的残基及び非標準的残基の混合物からなる分子を含む。
自然発生的または野生型のCFTR mRNA(及びそのmRNAを含む組成物)を使用するインビボの機能的CFTRの産生方法を提供することに加えて、本発明は、CFTRタンパク質(例えば、配列番号1)をコードする非自然発生的CFTR mRNAも提供する。いくつかの実施形態では、非自然発生的CFTR mRNAは、精製または単離される。
mRNAを合成しなかった、かつ/または、非自然発生的CFTR mRNA及び/もしくは機能的CFTR遺伝子に相補的なDNAを含まず、この細胞は、不活性CFTR遺伝子、例えば、遺伝子の発現産物を非機能性にする、ナンセンス変異、ミスセンス変異、フレームシフト変異、挿入変異、または欠失変異を有するCFTR遺伝子などを任意に含み得る。いくつかの実施形態では、非自然発生的CFTR mRNAを含む細胞は、非自然発生的CFTR mRNAから翻訳された機能的CFTRタンパク質をさらに含む。該細胞は、例えば、肺上皮細胞、肝細胞、または腎細胞であり得る。いくつかの実施形態では、該細胞は細胞培養物中にある。
いくつかの実施形態では、本発明に従うCFTR mRNAは、配列番号2(すなわち、野生型ヒトCFTRのコード配列)よりも少ないクリプティックプロモーターの補体、配列番号2よりも少ない直列反復及び/もしくは逆方向反復、配列番号2よりも少ない不利なコドンを有するコード配列を含む、かつ/または、該コード配列のGC含量が、配列番号2のGC含量よりも低い。
いくつかの実施形態では、CFTRタンパク質をコードするmRNAは、シグナルペプチドをコードするヌクレオチド配列を組み込む。本明細書において使用される場合、用語「シグナルペプチド」は、タンパク質の標的を分泌経路に定め得る、新たに合成されたタンパク質において存在するペプチドを指す。いくつかの実施形態では、シグナルペプチドは、mRNAの翻訳に続く小胞体内への移行後に切断される。シグナルペプチドは、シグナル配列、リーダー配列、またはリーダーペプチドとも称される。典型的に、シグナルペプチドは短い(例えば、5~30、5~25、5~20、5~15、または5~10アミノ酸長の)ペプチドである。シグナルペプチドは、新たに合成されたタンパク質のN末端に存在し得る。いかなる特定の理論にも制限されることを望むものではないが、CFTRをコードするmRNAへのシグナルペプチドをコードする配列の組み込みは、インビボのCFTRタンパク質の分泌及び/または産生を促進し得る。
5’ヒト成長ホルモン(hGH)配列(配列番号18):
AUGGCCACUGGAUCAAGAACCUCACUGCUGCUCGCUUUUGGACUGCUUUGCCUGCCCUGGUUGCAAGAAGGAUCGGCUUUCCCGACCAUCCCACUCUCC
代替的な5’ヒト成長ホルモン(hGH)配列(配列番号19):
AUGGCAACUGGAUCAAGAACCUCCCUCCUGCUCGCAUUCGGCCUGCUCUGUCUCCCAUGGCUCCAAGAAGGAAGCGCGUUCCCCACUAUCCCCCUCUCG
いくつかの実施形態では、本mRNAは、その5’-UTRにおいて、配列番号4と同一であるか、または配列番号4と少なくとも50%、少なくとも55%、少なくとも60%、少なくとも65%、少なくとも70%、少なくとも75%、少なくとも80%、少なくとも85%、少なくとも90%、少なくとも95%、少なくとも98%、もしくは少なくとも99%同一である配列を含む。
ある特定の実施形態では、本発明のmRNA分子は、裸または未包装のmRNAとして投与され得る。いくつかの実施形態では、本発明の組成物中のmRNAの投与は、好適な担体の包含によって促進され得る。ある特定の実施形態では、該担体は、標的細胞への1つ以上のmRNAのトランスフェクションを促進するその能力に基づいて選択される。
ある特定の実施形態では、本mRNAは、標的細胞への送達を促進するために脂質ナノ粒子と複合される。ある特定の実施形態では、本発明の組成物は、1つ以上のカチオン性脂質、非カチオン性脂質(ヘルパー脂質とも称される)などの追加の脂質、コレステロール系脂質、及び/またはmRNA被包のためのPEG化脂質を用いる多構成要素の脂質混合物と組み合わされる場合がある。
いくつかの実施形態では、好適な脂質ナノ粒子は、カチオン性脂質を含有する。本明細書において使用される場合、語句「カチオン性脂質」は、選択されたpH、例えば生理的pHにおいて正味の正電荷を有する、ある数の脂質種のうちのいずれかを指す。いくつかのカチオン性脂質、特に、滴定可能またはpH滴定可能なカチオン性脂質として知られるものが、mRNAを送達するに当たって特に有効である。いくつかのカチオン性(例えば、滴定可能)脂質が文献に記載されており、その多くは市販されている。本発明の組成物及び方法において使用するために特に好適なカチオン性脂質としては、国際特許公開第WO2010/053572号(そして特に、段落[00225]に記載されるC12-200)ならびに国際公開第WO2012/170930号に記載のものを含み、この両方が参照により本明細書に組み込まれる。いくつかの実施形態では、カチオン性脂質cKK-E12が使用され(国際公開第WO2013/063468号に開示される)、その教示は、参照によりその全体が本明細書に組み込まれる。いくつかの実施形態では、カチオン性脂質N-[1-(2,3-ジオレイルオキシ)プロピル]-N,N,N-塩化トリメチルアンモニウム、すなわち「DOTMA」が使用される。(Feigner et al.(Proc.Nat’l Acad.Sci.84,7413(1987)、米国特許第4,897,355号)。DOTMAは、単独で、または中性脂質、ジオレオイルホスファチジル-エタノールアミンもしくは「DOPE」、または他のカチオン性もしくは非カチオン性脂質と組み合わせて、リポソーム移動ビヒクルまたは脂質ナノ粒子へと製剤化され得、かかるリポソームは、標的細胞への核酸の送達を向上させるために使用され得る。他の好適なカチオン性脂質としては、例えば、5-カルボキシスペルミルグリシンジオクタデシルアミド、すなわち「DOGS」、2,3-ジオレイルオキシ-N-[2(スペルミン-カルボアミド)エチル]-N,N-ジメチル-1-プロパンアミニウム、すなわち「DOSPA」(Behr et al.Proc.Nat.’l Acad.Sci.86,6982(1989)、米国特許第5,171,678号、米国特許第5,334,761号)、1,2-ジオレオイル-3-ジメチルアンモニウム-プロパン、すなわち「DODAP」、1,2-ジオレオイル-3-トリメチルアンモニウム-プロパン、すなわち「DOTAP」が挙げられる。企図されるカチオン性脂質としては、1,2-ジステアリルオキシ-N,N-ジメチル-3-アミノプロパン、すなわち「DSDMA」、1,2-ジオレイルオキシ-N,N-ジメチル-3-アミノプロパン、すなわち「DODMA」、1,2-ジリノレイルオキシ-N,N-ジメチル-3-アミノプロパン、すなわち「DLinDMA」、1,2-ジリノレニルオキシ-N,N-ジメチル-3-アミノプロパン、すなわち「DLenDMA」、N-ジオレイル-N,N-ジメチルアンモニウム塩化物、すなわち「DODAC」、N,N-ジステアリル-N,N-ジメチルアンモニウム臭化物、すなわち「DDAB」、N-(1,2-ジミリスチルオキシプロプ-3-イル)-N,N-ジメチル-N-ヒドロキシエチルアンモニウム臭化物、すなわち「DMRIE」、3-ジメチルアミノ-2-(コレスト-5-エン-3-β-オキシブタン-4-オキシ)-1-(シス,シス-9,12-オクタデカジエンオキシ)プロパン、すなわち「CLinDMA」、2-[5’-(コレスト-5-エン-3-β-オキシ)-3’-オキサペントキシ)-3-ジメチル1-1-(シス,シス-9’,1-2’-オクタデカジエンオキシ)プロパン、すなわち「CpLinDMA」、N,N-ジメチル-3,4-ジオレイルオキシベンジルアミン、すなわち「DMOBA」、1,2-N,N’-ジオレイルカルバミル-3-ジメチルアミノプロパン、すなわち「DOcarbDAP」、2,3-ジリノレオイルオキシ-N,N-ジメチルプロピルアミン、すなわち「DLinDAP」、1,2-N,N’-ジリノレイルカルバミル-3-ジメチルアミノプロパン、すなわち「DLincarbDAP」、1,2-ジリノレオイルカルバミル-3-ジメチルアミノプロパン、すなわち「DLinCDAP」、2,2-ジリノレイル-4-ジメチルアミノメチル-[1,3]-ジオキソラン、すなわち「DLin--DMA」、2,2-ジリノレイル-4-ジメチルアミノエチル-[1,3]-ジオキソラン、すなわち「DLin-K-XTC2-DMA」、及び2-(2,2-ジ((9Z,12Z)-オクタデカ-9,12-ジエン-1-イル)-1,3-ジオキソラン-4-イル)-N,N-ジメチルエタンアミン(DLin-KC2-DMA))(国際公開第WO2010/042877号、Semple et al.,Nature Biotech.28:172-176(2010)を参照されたい)、またはこれらの混合物も挙げられる。(Heyes,J.,et al.,J Controlled Release 107:276-287(2005)、Morrissey,DV.,et al.,Nat.Biotechnol.23(8):1003-1007(2005)、PCT公開第WO2005/121348A1号)。
いくつかの実施形態では、好適な脂質ナノ粒子は、1つ以上の非カチオン性(「ヘルパー」)脂質を含有する。本明細書において使用される場合、語句「非カチオン性脂質」は、中性脂質、双性イオン性、またはアニオン性脂質のいずれかを指す。本明細書において使用される場合、語句「アニオン性脂質」は、選択されたpH、例えば生理的pHにおいて正味の負電荷を帯びる、ある数の脂質種のうちのいずれかを指す。いくつかの実施形態では、非カチオン性脂質は、中性脂質、すなわち、本組成物が製剤化及び/または投与される条件下で正味電荷を帯びない脂質である。非カチオン性脂質としては、ジステアロイルホスファチジルコリン(DSPC)、ジオレオイルホスファチジルコリン(DOPC)、ジパルミトイルホスファチジルコリン(DPPC)、ジオレオイルホスファチジルグリセロール(DOPG)、ジパルミトイルホスファチジルグリセロール(DPPG)、ジオレオイルホスファチジルエタノールアミン(DOPE)、パルミトイルオレオイルホスファチジルコリン(POPC)、パルミトイルオレオイル-ホスファチジルエタノールアミン(POPE)、ジオレオイル-ホスファチジルエタノールアミン4-(N-マレイミドメチル)-シクロヘキサン-1-カルボキシレート(DOPE-mal)、ジパルミトイルホスファチジルエタノールアミン(DPPE)、ジミリストイルホスホエタノールアミン(DMPE)、ジステアロイル-ホスファチジル-エタノールアミン(DSPE)、16-O-モノメチルPE、16-O-ジメチルPE、18-1-トランスPE、1-ステアロイル-2-オレオイル-ホスファチジエタノールアミン(SOPE)、またはこれらの混合物が挙げられるが、これらに限定されない。
いくつかの実施形態では、好適な脂質ナノ粒子は、1つ以上のコレステロールベースの脂質を含む。例えば、好適なコレステロールベースのカチオン性脂質としては、例えば、コレステロール、PEG化コレステロール、DC-Choi(N,N-ジメチル-N-エチルカルボアミドコレステロール)、1,4-ビス(3-N-オレイルアミノ-プロピル)ピペラジン(Gao,et al.Biochem.Biophys.Res.Comm.179,280(1991)、Wolf et al.BioTechniques
23,139(1997)、米国特許第5,744,335号)、またはICEが挙げられる。
いくつかの実施形態では、好適な脂質ナノ粒子は、1つ以上のPEG化脂質を含む。例えば、ポリエチレングリコール(PEG)修飾リン脂質、及びN-オクタノイル-スフィンゴシン-1-[サクシニル(メトキシポリエチレングリコール)-2000](C8 PEG-2000セラミド)を含む誘導体化セラミド(PEG-CER)などの誘導体化脂質の使用は、カチオン性脂質のうちの1つ以上、そしていくつかの実施形態では他の脂質との組み合わせで、本発明によって企図される。いくつかの実施形態では、好適なPEG化脂質は、より短いアシル鎖(例えば、C14またはC18)を有するPEG-セラミドを含む。いくつかの実施形態では、PEG化脂質DSPE-PEG-マレイミド-レクチンが使用され得る。他の企図されるPEG修飾脂質としては、C6~C20長のアルキル鎖(複数可)を有する脂質に共有結合された、長さが最大5kDaのポリエチレングリコール鎖が挙げられるが、これに限定されない。特定の理論に制限されることを望むものではないが、PEG化脂質の付加は、複合体の凝集を防止し、循環の寿命を増加させ、リポソームで被包されたmRNAの標的細胞への送達を促進し得ることが企図される。
C12-200、DOPE、コレステロール、DMG-PEG2K、
DODAP、DOPE、コレステロール、DMG-PEG2K、
HGT5000、DOPE、コレステロール、DMG-PEG2K、
HGT5001、DOPE、コレステロール、DMG-PEG2K、
XTC、DSPC、コレステロール、PEG-DMG、
MC3、DSPC、コレステロール、PEG-DMG、
ALNY-100、DSPC、コレステロール、PEG-DSG、
cKK-E12、DOPE、Chol、PEGDMG2K。
いくつかの実施形態では、非自然発生的CFTR mRNAを含むカチオン性脂質ベースまたはPEIベースの組成物などの、本発明に従う薬学的組成物は、対象の呼吸器系への投与のための装置において提供される。本装置は、例えば、滴下、エアロゾル化、または噴霧装置であり得る。好適な装置としては、例えば、PARI Boyジェット噴霧器、Aeroneb(登録商標)実験室噴霧器、MicroSprayer(登録商標)、またはEFlowメッシュ噴霧器が挙げられる。あるいは、携帯型吸入器などの乾燥粉末吸入器またはエアロゾル化装置が使用され得る。
本発明に従う使用及び方法のためのmRNA
とりわけ、本発明は、特に哺乳動物の肺における、CFTRタンパク質のインビボの産生方法を提供する。いくつかの実施形態では、本発明は、哺乳動物の肺における上皮細胞中のCFTR発現の誘導方法であって、該上皮細胞をインビトロ転写mRNAを含む薬学的組成物と接触させることを含む、方法を提供し、該インビトロ転写mRNAは、配列番号1(野生型ヒトCFTRのアミノ酸配列)をコードするコード配列を含む。本発明はまた、哺乳動物の肺における上皮細胞中のCFTR発現の誘導のための、インビトロ転写mRNAを含む薬学的組成物の使用を提供し、該インビトロ転写mRNAは、配列番号1をコードするコード配列を含む。
本発明に従う使用のための薬学的組成物は、前項で記述された本発明に従う使用及び方法のためのmRNA、ならびにCFTR mRNAを含む組成物に関する上記の項で記述された追加の成分を含み得る。したがって、上述の担体のいずれかを含む薬学的組成物の使用及び/または投与が企図される。
哺乳動物の肺におけるCFTR発現の誘導のための方法及び使用のいくつかの実施形態では、上述の薬学的組成物は、気管内滴下、噴霧、及びエアロゾル化から選択される経路によって投与される。本組成物を投与するための装置は、薬学的組成物が装填された装置に関する上記の項に列記される装置から選択され得る。
とりわけ、本発明は、嚢胞性線維症を治療するために使用され得る。いくつかの実施形態では、本発明は、治療を必要とする対象に、本明細書に記載のCFTRタンパク質をコードするmRNA、または本mRNAを含有する薬学的組成物を投与することによる、嚢胞性線維症の治療方法を提供する。本mRNAまたは本mRNAを含有する薬学的組成物は、対象の肺に直接投与され得る。肺送達のための様々な投与経路が使用され得る。いくつかの実施形態では、本明細書に記載のmRNAまたはmRNAを含有する組成物は、吸入、噴霧、またはエアロゾル化によって投与される。様々な実施形態では、本mRNAの投与は、対象の肺(例えば、肺の上皮細胞)におけるCFTRの発現をもたらす。
以下の具体的な実施例は単なる例示であり、本開示の残部をいかようにも制限するものではないと解釈されるべきである。さらなる詳細を伴わずに、当業者であれば、本明細書の説明に基づいて、本発明を最大限に利用することができると考えられる。
伝令RNA合成。ヒト嚢胞性線維症膜貫通コンダクタンス制御因子(CFTR)mRNA、及びホタルルシフェラーゼ(FFL)mRNAを、遺伝子をコードするプラスミドDNA鋳型からインビトロ転写によって合成し、続いて、5’キャップ構造(キャップ1)(Fechter,P.;Brownlee,G.G.“Recognition of
mRNA cap structures by viral and cellular proteins”J.Gen.Virology 2005,86,1239-1249)、及び、ゲル電気泳動によって決定されたときに長さがおよそ200のヌクレオチドの3’ポリ(A)テールを付加した。5’及び3’の非翻訳領域が各mRNA産物中に存在した。
この実施例は、細胞に送達される合成ヒトCFTR mRNAから完全に機能的なCFTRタンパク質が発現されることを実証する。
この実施例は、CFTRタンパク質が、肺内投与によって送達されるCFTRをコードするmRNAから、インビボで有効に発現されることを実証する。
マウスの肺へのヒトCFTR伝令RNAの送達は、直接吸入ならびに噴霧のいずれかを介して達成され得る。インサイツのハイブリダイゼーション方法を使用すると、ヒトCFTR mRNAが充填されたナノ粒子をマウスに気管内投与した後、ヒトCFTR mRNAを成功裏に検出することができる。投与は、脂質ベースのナノ粒子(例えば、C12-200)ならびにポリマーナノ粒子(例えば、ポリエチレンイミン、PEI)を用いて達成され得る。
脂質ベースのナノ担体を使用するCFTR mRNAの投与。上述の通り、ヒトCFTR mRNAの成功裏の肺送達は、脂質ナノ粒子ベースの送達ビヒクルを介して達成され得る。カチオン性脂質構成要素としてC12-200を利用する、hCFTR mRNAが充填されたカチオン性脂質ナノ粒子の実施例がここに開示される。
マウス、ブタ、及び培養細胞におけるヒトCFTRタンパク質検出のための抗体検証。hCFTRタンパク質に特異的であり、マウス及びブタ類似体と交差反応せず、かつ将来の実験のために十分な供給が利用可能である抗体を同定するために、実験を実施した。手短に言えば、学術的及び商業的供給源からの様々な抗hCFTR抗体の試験は、マウスまたはブタCFTRのいずれかに対する交差反応性を伴わずに、免疫沈降及びウェスタンブロッティング(IP/WB)後のヒトCFTRタンパク質を検出することができた抗hCFTR抗体の組み合わせの同定につながった。したがって、マウスまたはブタCFTRのいずれかに対する交差反応性を伴わないhCFTRタンパク質の検出のための好適な抗hCFTR抗体が、IP/WB結果に基づいて同定された。
analysis of Mutant CFTR in Lung explants,Cell Physiol.Biochem.30,587-595(2012))によって公開されたプロトコル及び抗体を使用して、Burkhard Tummler
教授(Medizinsche Hochschule Hannover)と共同で、初期IP実験を実施した。hCFTRを過剰発現させるヒト結腸癌細胞(T84)を、IP実験のための陽性対照として使用した。
ブタの肺への被包されたmRNAエアロゾルの送達の確立。ブタ肺へのホタルルシフェラーゼ(FFL)SNIM RNAのエアロゾル投与を、段階的な実験手順によって確立した。第1のステップでは、制御呼吸の間に麻酔されたブタにFFL SNIM RNA製剤を噴霧した。第2のステップでは、エアロゾル投与が完了した直後に肺を切除し、肺検体を細胞培養培地中で一晩インキュベートし、その後BLIによって肺検体にエクスビボのルシフェラーゼ測定を実施した。
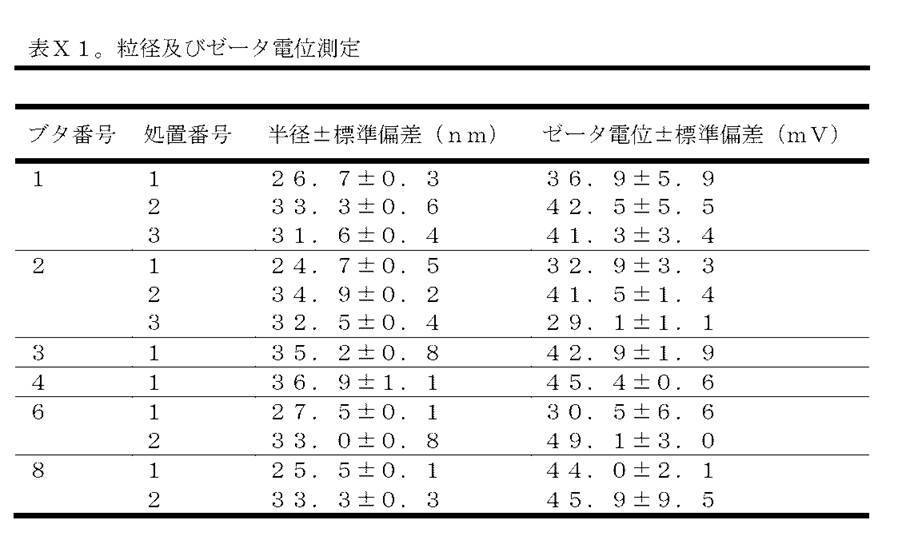
RNAを含む8ミリリットルの実施例6のPEI製剤を、WP5に記載の通り調製し、2回に分けて4mlずつの分量で相次いで噴霧した。細胞培養培地における一晩のインキュベーション後、様々な肺領域の切除された肺検体の組織ホモジネート中で、ルシフェラーゼ測定を実施した。肺検体の起点に従って、発現値をマッピングした(図21)。
治験を実施し、ブタにおける週1回のエアロゾル適用の実用性を評価した。肺疾患の誘導を伴わず(等級2より高い有害事象の存在なし)に1週間の間隔で修飾mRNAの3回のエアロゾル適用を実施することとして、実用性を定義した。追加の目的は、i)動物の苦痛の等級、ii)ブタの実験的または臨床的査定の間に生じる有害事象、ならびにiii)誘導されるタンパク質(ルシフェラーゼ及びhCFTR)の測定を評価するためであった。
SNIM RNAを用いて、弱く間隔で1回、2回、または3回処置した。2頭の無処置ブタを対照とした。処置の24時間後に肺を切除し、BLIによって単離された肺検体中のエクスビボのルシフェラーゼ活性を測定した。IP/WBを使用してhCFTRタンパク質の発現を分析した。細胞レベルでのルシフェラーゼ発現の検出のため、免疫組織化学的検査(IHC)を実施した。血清中の炎症性サイトカイン及び血液化学の測定によって、毒性を調査した。肺試料に病理組織診断を実施した。研究プロトコル「パイロットプロジェクト:ブタにおける嚢胞性線維症のエアロゾル療法のための動物モデルを確立するための修飾mRNAの繰り返しの適用(Pilot project: Repeated application of modified mRNA to establish an animal model for aerosol therapy of cystic fibrosis in pigs)」は、実験の開始前に地方当局によって承認された(動物実験許諾番号0-045-12)。
0群-処置なしの対照群
I群-1日目に実施例6のPEI製剤中の1mgのFFL SNIM RNA及び1mgのhCFTR SNIM RNAのエアロゾル投与。
II群-1日目に実施例6のPEI製剤中の2mgのhCFTR SNIM RNAのエアロゾル投与、かつ8日目に実施例6のPEI製剤中の1mgのFFL SNIM RNA及び1mgのhCFTR SNIM RNAのエアロゾル投与。
III群-1日目及び8日目に実施例6のPEI製剤中の2mgのhCFTR SNIM RNA(6379-186)のエアロゾル投与、15日目に実施例6のPEI製剤中の1mgのFFL SNIM RNA及び1mgのhCFTR SNIM RNAのエアロゾル投与。
al.,Cell Physiol Biochem.30,587-95(2012)によって説明されるプロトコルに従って、hCFTRのIP/WBを行った(図31)。成熟型複合グリコシル化hCFTRが、分散したいわゆるCバンドとして現れる。マンノースが豊富なhCFTRが、より高密度ないわゆるBバンドとして現れる。明らかに、hCFTR発現が、T84陽性対照細胞、及び実施例6のPEI製剤中のhCFTR SNIM RNAで処置されたブタ1番の肺組織中で観察される。hCFTRタンパク質の発現は、無処置ブタでは観察されなかった。同一のプロトコル(van Barneveld Aら、上記参照)を使用する公開された研究によるヒト肺組織中のhCFTRタンパク質発現の比較は、hCFTR SNIMエアロゾル処置後のブタ肺組織中のhCFTRの発現が、健常なヒトの肺におけるhCFTR発現と同様であったことを示唆した。
(1)アレルギー性/免疫性事象、(2)肺性/呼吸性、(3)体質的臨床兆候、(4)皮膚科学的/皮膚、(5)胃腸管系、及び(6)肺性/呼吸性。
mRNAで処置されたブタの肺において成功裏に実証された。
この実施例は、CFTRタンパク質が、シグナルペプチドをコードする配列を有するCFTRをコードするmRNAから有効に発現され得ることを実証する。
気管内投与されたmRNAが充填されたナノ粒子を介して産生されたヒトCFTRタンパク質の分析。CFTR KOマウスを使用して、すべての研究を実施した。PARI Boyジェット噴霧器を使用して、CFTR mRNA製剤またはビヒクル対照を導入した。マウスを屠殺し、所定期間後、mRNAからのタンパク質発現を可能にするため、生理食塩水で灌流した。
ブタの肺へのPEIで被包されたmRNAのエアロゾル送達の用量増大。ホタルルシフェラーゼ(FFL)SNIM RNAと、コドン最適化ヒトCFTR(CO-CFTR)SNIM RNAとの組み合わせの、様々な濃度でのブタ肺へのエアロゾル投与を、段階的な実験手順によって確立した。第1のステップでは、制御呼吸の間に麻酔されたブタにFFL/CO-CFTR SNIM RNA製剤を噴霧した。第2のステップでは、エアロゾル投与が完了した24時間後の鎮静後に、外側耳静脈を介するペントバルビタール(100mg/体重kg)及び塩化カリウムのボーラス注入によって、動物を屠殺した。肺を切除し、およそ1cm厚の組織検体にスライスした。ルシフェラーゼ活性の測定のため、組織検体を、D-ルシフェリン基質を含む培地浴中でインキュベートし、エクスビボのルシフェラーゼBLIを行った。BLIの後、病理組織診断、免疫組織化学的検査、ならびにインサイツのハイブリダイゼーションのために、ルシフェラーゼ陽性領域及びルシフェラーゼ陰性領域からの試料を取った。残留する検体を液体窒素中でショック凍結し、その後、IP/WB及び酵素結合免疫吸着検定法による分析まで-80℃で保管した。
RNAを、標準的な方法を使用してプラスミドDNA鋳型からインビトロ転写によって合成した。
of Pennsylvania,PA,USAから得た3つの異なる抗体(抗体217、432、596)の3重の組み合わせを使用するCFTRのウェスタンブロット免疫検出を行った。異なる動物間の群内の可変性、及びCFTR発現の可変性を制御するため、150kDaに対応するタンパク質の大きさの標準におけるマーカーバンドを基準として設定し、異なる群のバンド強度をこの値に正規化した。図39に示す通り、コホート1の対照ブタから分析された組織試料の16%のみが、基線を超えるCFTR発現レベルをもたらした。対照的に、それぞれ5mg及び10mgの処置群を表すコホート3及び4は、それぞれ、それらの肺組織試料の30%超が、基線よりも高いCFTR発現レベルに陽性の試験結果をもたらした(図39)。さらに、コホート3及び4において観察されたCFTR発現の増加は、対照のものよりもほぼ2倍高かった。
この実施例は、mRNAが充填されたナノ粒子のエアロゾル送達後の肺における成功裏のインビボの発現を実証する。Technical University Munich,Weihenstephan,Germanyから取得したGerman Landraceのブタを使用して、すべての研究を実施した。ブタは35~90kgの範囲の体重を有した。Pariジェット噴霧器を使用して、FFL/CO-CFTR-C-His10 mRNA製剤またはビヒクル対照を導入した。ブタを屠殺し、所定期間後、mRNAからのタンパク質発現を可能にするため、生理食塩水で灌流した。
配列番号1。野生型CFTRアミノ酸配列。
配列番号2。野生型CFTR mRNAコード配列。
配列番号3。非自然発生的CFTR mRNAコード配列番号1。
配列番号4。CFTR mRNA 5’-UTR。
配列番号5。CFTR mRNA 3’-UTR番号1。
配列番号6。FFL 5’UTR。
配列番号7。FFLコード配列。
配列番号8。FFL 3’UTR。
配列番号9。非自然発生的CFTR mRNAコード配列番号2。
配列番号10。非自然発生的CFTR mRNAコード配列番号3。
配列番号11。非自然発生的CFTR mRNAコード配列番号4。
配列番号12。非自然発生的CFTR mRNAコード配列番号5。
配列番号13。非自然発生的CFTR mRNAコード配列番号6。
配列番号14。非自然発生的CFTR mRNAコード配列番号7。
配列番号15。コドン最適化ヒトCFTR C末端His10融合mRNAコード配列。配列番号16。成長ホルモンリーダー配列を有するコドン最適化ヒトCFTR mRNAコード配列。
配列番号17。コドン最適化ヒトCFTR mRNA
配列番号18。mRNAリーダー配列番号1
配列番号19。mRNAリーダー配列番号2
配列番号20。CFTR mRNA 3’-UTR番号2。
配列番号1
MQRSPLEKASVVSKLFFSWTRPILRKGYRQRLELSDIYQIPSVDSADNLSEKLEREWDRELASKKNPKLINALRRCFFWRFMFYGIFLYLGEVTKAVQPLLLGRIIASYDPDNKEERSIAIYLGIGLCLLFIVRTLLLHPAIFGLHHIGMQMRIAMFSLIYKKTLKLSSRVLDKISIGQLVSLLSNNLNKFDEGLALAHFVWIAPLQVALLMGLIWELLQASAFCGLGFLIVLALFQAGLGRMMMKYRDQRAGKISERLVITSEMIENIQSVKAYCWEEAMEKMIENLRQTELKLTRKAAYVRYFNSSAFFFSGFFVVFLSVLPYALIKGIILRKIFTTISFCIVLRMAVTRQFPWAVQTWYDSLGAINKIQDFLQKQEYKTLEYNLTTTEVVMENVTAFWEEGFGELFEKAKQNNNNRKTSNGDDSLFFSNFSLLGTPVLKDINFKIERGQLLAVAGSTGAGKTSLLMVIMGELEPSEGKIKHSGRISFCSQFSWIMPGTIKENIIFGVSYDEYRYRSVIKACQLEEDISKFAEKDNIVLGEGGITLSGGQRARISLARAVYKDADLYLLDSPFGYLDVLTEKEIFESCVCKLMANKTRILVTSKMEHLKKADKILILHEGSSYFYGTFSELQNLQPDFSSKLMGCDSFDQFSAERRNSILTETLHRFSLEGDAPVSWTETKKQSFKQTGEFGEKRKNSILNPINSIRKFSIVQKTPLQMNGIEEDSDEPLERRLSLVPDSEQGEAILPRISVISTGPTLQARRRQSVLNLMTHSVNQGQNIHRKTTASTRKVSLAPQANLTELDIYSRRLSQETGLEISEEINEEDLKECFFDDMESIPAVTTWNTYLRYITVHKSLIFVLIWCLVIFLAEVAASLVVLWLLGNTPLQDKGNSTHSRNNSYAVIITSTSSYYVFYIYVGVADTLLAMGFFRGLPLVHTLITVSKILHHKMLHSVLQAPMSTLNTLKAGGILNRFSKDIAILDDLLPLTIFDFIQLLLIVIGAIAVVAVLQPYIFVATVPVIVAFIMLRAYFLQTSQQLKQLESEGRSPIFTHLVTSLKGLWTLRAFGRQPYFETLFHKALNLHTANWFLYLSTLRWFQMRIEMIFVIFFIAVTFISILTTGEGEGRVGIILTLAMNIMSTLQWAVNSSIDVDSLMRSVSRVFKFIDMPTEGKPTKSTKPYKNGQLSKVMIIENSHVKKDDIWPSGGQMTVKDLTAKYTEGGNAILENISFSISPGQRVGLLGRTGSGKSTLLSAFLRLLNTEGEIQIDGVSWDSITLQQWRKAFGVIPQKVFIFSGTFRKNLDPYEQWSDQEIWKVADEVGLRSVIEQFPGKLDFVLVDGGCVLSHGHKQLMCLARSVLSKAKILLLDEPSAHLDPVTYQIIRRTLKQAFADCTVILCEHRIEAMLECQQFLVIEENKVRQYDSIQKLLNERSLFRQAISPSDRVKLFPHRNSSKCKSKPQIAALKEETEEEVQDTRL(配列番号1)
配列番号2
AUGCAGAGGUCGCCUCUGGAAAAGGCCAGCGUUGUCUCCAAACUUUUUUUCAGCUGGACCAGACCAAUUUUGAGGAAAGGAUACAGACAGCGCCUGGAAUUGUCAGACAUAUACCAAAUCCCUUCUGUUGAUUCUGCUGACAAUCUAUCUGAAAAAUUGGAAAGAGAAUGGGAUAGAGAGCUGGCUUCAAAGAAAAAUCCUAAACUCAUUAAUGCCCUUCGGCGAUGUUUUUUCUGGAGAUUUAUGUUCUAUGGAAUCUUUUUAUAUUUAGGGGAAGUCACCAAAGCAGUACAGCCUCUCUUACUGGGAAGAAUCAUAGCUUCCUAUGACCCGGAUAACAAGGAGGAACGCUCUAUCGCGAUUUAUCUAGGCAUAGGCUUAUGCCUUCUCUUUAUUGUGAGGACACUGCUCCUACACCCAGCCAUUUUUGGCCUUCAUCACAUUGGAAUGCAGAUGAGAAUAGCUAUGUUUAGUUUGAUUUAUAAGAAGACUUUAAAGCUGUCAAGCCGUGUUCUAGAUAAAAUAAGUAUUGGACAACUUGUUAGUCUCCUUUCCAACAACCUGAACAAAUUUGAUGAAGGACUUGCAUUGGCACAUUUCGUGUGGAUCGCUCCUUUGCAAGUGGCACUCCUCAUGGGGCUAAUCUGGGAGUUGUUACAGGCGUCUGCCUUCUGUGGACUUGGUUUCCUGAUAGUCCUUGCCCUUUUUCAGGCUGGGCUAGGGAGAAUGAUGAUGAAGUACAGAGAUCAGAGAGCUGGGAAGAUCAGUGAAAGACUUGUGAUUACCUCAGAAAUGAUUGAAAAUAUCCAAUCUGUUAAGGCAUACUGCUGGGAAGAAGCAAUGGAAAAAAUGAUUGAAAACUUAAGACAAACAGAACUGAAACUGACUCGGAAGGCAGCCUAUGUGAGAUACUUCAAUAGCUCAGCCUUCUUCUUCUCAGGGUUCUUUGUGGUGUUUUUAUCUGUGCUUCCCUAUGCACUAAUCAAAGGAAUCAUCCUCCGGAAAAUAUUCACCACCAUCUCAUUCUGCAUUGUUCUGCGCAUGGCGGUCACUCGGCAAUUUCCCUGGGCUGUACAAACAUGGUAUGACUCUCUUGGAGCAAUAAACAAAAUACAGGAUUUCUUACAAAAGCAAGAAUAUAAGACAUUGGAAUAUAACUUAACGACUACAGAAGUAGUGAUGGAGAAUGUAACAGCCUUCUGGGAGGAGGGAUUUGGGGAAUUAUUUGAGAAAGCAAAACAAAACAAUAACAAUAGAAAAACUUCUAAUGGUGAUGACAGCCUCUUCUUCAGUAAUUUCUCACUUCUUGGUACUCCUGUCCUGAAAGAUAUUAAUUUCAAGAUAGAAAGAGGACAGUUGUUGGCGGUUGCUGGAUCCACUGGAGCAGGCAAGACUUCACUUCUAAUGAUGAUUAUGGGAGAACUGGAGCCUUCAGAGGGUAAAAUUAAGCACAGUGGAAGAAUUUCAUUCUGUUCUCAGUUUUCCUGGAUUAUGCCUGGCACCAUUAAAGAAAAUAUCAUCUUUGGUGUUUCCUAUGAUGAAUAUAGAUACAGAAGCGUCAUCAAAGCAUGCCAACUAGAAGAGGACAUCUCCAAGUUUGCAGAGAAAGACAAUAUAGUUCUUGGAGAAGGUGGAAUCACACUGAGUGGAGGUCAACGAGCAAGAAUUUCUUUAGCAAGAGCAGUAUACAAAGAUGCUGAUUUGUAUUUAUUAGACUCUCCUUUUGGAUACCUAGAUGUUUUAACAGAAAAAGAAAUAUUUGAAAGCUGUGUCUGUAAACUGAUGGCUAACAAAACUAGGAUUUUGGUCACUUCUAAAAUGGAACAUUUAAAGAAAGCUGACAAAAUAUUAAUUUUGAAUGAAGGUAGCAGCUAUUUUUAUGGGACAUUUUCAGAACUCCAAAAUCUACAGCCAGACUUUAGCUCAAAACUCAUGGGAUGUGAUUCUUUCGACCAAUUUAGUGCAGAAAGAAGAAAUUCAAUCCUAACUGAGACCUUACACCGUUUCUCAUUAGAAGGAGAUGCUCCUGUCUCCUGGACAGAAACAAAAAAACAAUCUUUUAAACAGACUGGAGAGUUUGGGGAAAAAAGGAAGAAUUCUAUUCUCAAUCCAAUCAACUCUAUACGAAAAUUUUCCAUUGUGCAAAAGACUCCCUUACAAAUGAAUGGCAUCGAAGAGGAUUCUGAUGAGCCUUUAGAGAGAAGGCUGUCCUUAGUACCAGAUUCUGAGCAGGGAGAGGCGAUACUGCCUCGCAUCAGCGUGAUCAGCACUGGCCCCACGCUUCAGGCACGAAGGAGGCAGUCUGUCCUGAACCUGAUGACACACUCAGUUAACCAAGGUCAGAACAUUCACCGAAAGACAACAGCAUCCACACGAAAAGUGUCACUGGCCCCUCAGGCAAACUUGACUGAACUGGAUAUAUAUUCAAGAAGGUUAUCUCAAGAAACUGGCUUGGAAAUAAGUGAAGAAAUUAACGAAGAAGACUUAAAGGAGUGCCUUUUUGAUGAUAUGGAGAGCAUACCAGCAGUGACUACAUGGAACACAUACCUUCGAUAUAUUACUGUCCACAAGAGCUUAAUUUUUGUGCUAAUUUGGUGCUUAGUAAUUUUUCUGGCAGAGGUGGCUGCUUCUUUGGUUGUGCUGUGGCUCCUUGGAAACACUCCUCUUCAAGACAAAGGGAAUAGUACUCAUAGUAGAAAUAACAGCUAUGCAGUGAUUAUCACCAGCACCAGUUCGUAUUAUGUGUUUUACAUUUACGUGGGAGUAGCCGACACUUUGCUUGCUAUGGGAUUCUUCAGAGGUCUACCACUGGUGCAUACUCUAAUCACAGUGUCGAAAAUUUUACACCACAAAAUGUUACAUUCUGUUCUUCAAGCACCUAUGUCAACCCUCAACACGUUGAAAGCAGGUGGGAUUCUUAAUAGAUUCUCCAAAGAUAUAGCAAUUUUGGAUGACCUUCUGCCUCUUACCAUAUUUGACUUCAUCCAGUUGUUAUUAAUUGUGAUUGGAGCUAUAGCAGUUGUCGCAGUUUUACAACCCUACAUCUUUGUUGCAACAGUGCCAGUGAUAGUGGCUUUUAUUAUGUUGAGAGCAUAUUUCCUCCAAACCUCACAGCAACUCAAACAACUGGAAUCUGAAGGCAGGAGUCCAAUUUUCACUCAUCUUGUUACAAGCUUAAAAGGACUAUGGACACUUCGUGCCUUCGGACGGCAGCCUUACUUUGAAACUCUGUUCCACAAAGCUCUGAAUUUACAUACUGCCAACUGGUUCUUGUACCUGUCAACACUGCGCUGGUUCCAAAUGAGAAUAGAAAUGAUUUUUGUCAUCUUCUUCAUUGCUGUUACCUUCAUUUCCAUUUUAACAACAGGAGAAGGAGAAGGAAGAGUUGGUAUUAUCCUGACUUUAGCCAUGAAUAUCAUGAGUACAUUGCAGUGGGCUGUAAACUCCAGCAUAGAUGUGGAUAGCUUGAUGCGAUCUGUGAGCCGAGUCUUUAAGUUCAUUGACAUGCCAACAGAAGGUAAACCUACCAAGUCAACCAAACCAUACAAGAAUGGCCAACUCUCGAAAGUUAUGAUUAUUGAGAAUUCACACGUGAAGAAAGAUGACAUCUGGCCCUCAGGGGGCCAAAUGACUGUCAAAGAUCUCACAGCAAAAUACACAGAAGGUGGAAAUGCCAUAUUAGAGAACAUUUCCUUCUCAAUAAGUCCUGGCCAGAGGGUGGGCCUCUUGGGAAGAACUGGAUCAGGGAAGAGUACUUUGUUAUCAGCUUUUUUGAGACUACUGAACACUGAAGGAGAAAUCCAGAUCGAUGGUGUGUCUUGGGAUUCAAUAACUUUGCAACAGUGGAGGAAAGCCUUUGGAGUGAUACCACAGAAAGUAUUUAUUUUUUCUGGAACAUUUAGAAAAAACUUGGAUCCCUAUGAACAGUGGAGUGAUCAAGAAAUAUGGAAAGUUGCAGAUGAGGUUGGGCUCAGAUCUGUGAUAGAACAGUUUCCUGGGAAGCUUGACUUUGUCCUUGUGGAUGGGGGCUGUGUCCUAAGCCAUGGCCACAAGCAGUUGAUGUGCUUGGCUAGAUCUGUUCUCAGUAAGGCGAAGAUCUUGCUGCUUGAUGAACCCAGUGCUCAUUUGGAUCCAGUAACAUACCAAAUAAUUAGAAGAACUCUAAAACAAGCAUUUGCUGAUUGCACAGUAAUUCUCUGUGAACACAGGAUAGAAGCAAUGCUGGAAUGCCAACAAUUUUUGGUCAUAGAAGAGAACAAAGUGCGGCAGUACGAUUCCAUCCAGAAACUGCUGAACGAGAGGAGCCUCUUCCGGCAAGCCAUCAGCCCCUCCGACAGGGUGAAGCUCUUUCCCCACCGGAACUCAAGCAAGUGCAAGUCUAAGCCCCAGAUUGCUGCUCUGAAAGAGGAGACAGAAGAAGAGGUGCAAGAUACAAGGCUUUAG(配列番号2)
配列番号3
AUGCAGCGGUCCCCGCUCGAAAAGGCCAGUGUCGUGUCCAAACUCUUCUUCUCAUGGACUCGGCCUAUCCUUAGAAAGGGGUAUCGGCAGAGGCUUGAGUUGUCUGACAUCUACCAGAUCCCCUCGGUAGAUUCGGCGGAUAACCUCUCGGAGAAGCUCGAACGGGAAUGGGACCGCGAACUCGCGUCUAAGAAAAACCCGAAGCUCAUCAACGCACUGAGAAGGUGCUUCUUCUGGCGGUUCAUGUUCUACGGUAUCUUCUUGUAUCUCGGGGAGGUCACAAAAGCAGUCCAACCCCUGUUGUUGGGUCGCAUUAUCGCCUCGUACGACCCCGAUAACAAAGAAGAACGGAGCAUCGCGAUCUACCUCGGGAUCGGACUGUGUUUGCUUUUCAUCGUCAGAACACUUUUGUUGCAUCCAGCAAUCUUCGGCCUCCAUCACAUCGGUAUGCAGAUGCGAAUCGCUAUGUUUAGCUUGAUCUACAAAAAGACACUGAAACUCUCGUCGCGGGUGUUGGAUAAGAUUUCCAUCGGUCAGUUGGUGUCCCUGCUUAGUAAUAACCUCAACAAAUUCGAUGAGGGACUGGCGCUGGCACAUUUCGUGUGGAUUGCCCCGUUGCAAGUCGCCCUUUUGAUGGGCCUUAUUUGGGAGCUGUUGCAGGCAUCUGCCUUUUGUGGCCUGGGAUUUCUGAUUGUGUUGGCAUUGUUUCAGGCUGGGCUUGGGCGGAUGAUGAUGAAGUAUCGCGACCAGAGAGCGGGUAAAAUCUCGGAAAGACUCGUCAUCACUUCGGAAAUGAUCGAAAACAUCCAGUCGGUCAAAGCCUAUUGCUGGGAAGAAGCUAUGGAGAAGAUGAUUGAAAACCUCCGCCAAACUGAGCUGAAACUGACCCGCAAGGCGGCGUAUGUCCGGUAUUUCAAUUCGUCAGCGUUCUUCUUUUCCGGGUUCUUCGUUGUCUUUCUCUCGGUUUUGCCUUAUGCCUUGAUUAAGGGGAUUAUCCUCCGCAAGAUUUUCACCACGAUUUCGUUCUGCAUUGUAUUGCGCAUGGCAGUGACACGGCAAUUUCCGUGGGCCGUGCAGACAUGGUAUGACUCGCUUGGAGCGAUCAACAAAAUCCAAGACUUCUUGCAAAAGCAAGAGUACAAGACCCUGGAGUACAAUCUUACUACUACGGAGGUAGUAAUGGAGAAUGUGACGGCUUUUUGGGAAGAGGGUUUUGGAGAACUGUUUGAGAAAGCAAAGCAGAAUAACAACAACCGCAAGACCUCAAAUGGGGACGAUUCCCUGUUUUUCUCGAACUUCUCCCUGCUCGGAACACCCGUGUUGAAGGACAUCAAUUUCAAGAUUGAGAGGGGACAGCUUCUCGCGGUAGCGGGAAGCACUGGUGCGGGAAAAACUAGCCUCUUGAUGGUGAUUAUGGGGGAGCUUGAGCCCAGCGAGGGGAAGAUUAAACACUCCGGGCGUAUCUCAUUCUGUAGCCAGUUUUCAUGGAUCAUGCCCGGAACCAUUAAAGAGAACAUCAUUUUCGGAGUAUCCUAUGAUGAGUACCGAUACAGAUCGGUCAUUAAGGCGUGCCAGUUGGAAGAGGACAUUUCUAAGUUCGCCGAGAAGGAUAACAUCGUCUUGGGAGAAGGGGGUAUUACAUUGUCGGGAGGGCAGCGAGCGCGGAUCAGCCUCGCGAGAGCGGUAUACAAAGAUGCAGAUUUGUAUCUGCUUGAUUCACCGUUUGGAUACCUCGACGUAUUGACAGAAAAAGAAAUCUUCGAGUCGUGCGUGUGUAAACUUAUGGCUAAUAAGACGAGAAUCCUGGUGACAUCAAAAAUGGAACACCUUAAGAAGGCGGACAAGAUCCUGAUCCUCCACGAAGGAUCGUCCUACUUUUACGGCACUUUCUCAGAGUUGCAAAACUUGCAGCCGGACUUCUCAAGCAAACUCAUGGGGUGUGACUCAUUCGACCAGUUCAGCGCGGAACGGCGGAACUCGAUCUUGACGGAAACGCUGCACCGAUUCUCGCUUGAGGGUGAUGCCCCGGUAUCGUGGACCGAGACAAAGAAGCAGUCGUUUAAGCAGACAGGAGAAUUUGGUGAGAAAAGAAAGAACAGUAUCUUGAAUCCUAUUAACUCAAUUCGCAAGUUCUCAAUCGUCCAGAAAACUCCACUGCAGAUGAAUGGAAUUGAAGAGGAUUCGGACGAACCCCUGGAGCGCAGGCUUAGCCUCGUGCCGGAUUCAGAGCAAGGGGAGGCCAUUCUUCCCCGGAUUUCGGUGAUUUCAACCGGACCUACACUUCAGGCGAGGCGAAGGCAAUCCGUGCUCAACCUCAUGACGCAUUCGGUAAACCAGGGGCAAAACAUUCACCGCAAAACGACGGCCUCAACGAGAAAAGUGUCACUUGCACCCCAGGCGAAUUUGACUGAACUCGACAUCUACAGCCGUAGGCUUUCGCAAGAAACCGGACUUGAGAUCAGCGAAGAAAUCAAUGAAGAAGAUUUGAAAGAGUGUUUCUUUGAUGACAUGGAAUCAAUCCCAGCGGUGACAACGUGGAACACAUACUUGCGUUACAUCACGGUGCACAAGUCCUUGAUUUUCGUCCUCAUCUGGUGUCUCGUGAUCUUUCUCGCUGAGGUCGCAGCGUCACUUGUGGUCCUCUGGCUGCUUGGUAAUACGCCCUUGCAAGACAAAGGCAAUUCUACACACUCAAGAAACAAUUCCUAUGCCGUGAUUAUCACUUCUACAAGCUCGUAUUACGUGUUUUACAUCUACGUAGGAGUGGCCGACACUCUGCUCGCGAUGGGUUUCUUCCGAGGACUCCCACUCGUUCACACGCUUAUCACUGUCUCCAAGAUUCUCCACCAUAAGAUGCUUCAUAGCGUACUGCAGGCUCCCAUGUCCACCUUGAAUACGCUCAAGGCGGGAGGUAUUUUGAAUCGCUUCUCAAAAGAUAUUGCAAUUUUGGAUGACCUUCUGCCCCUGACGAUCUUCGACUUCAUCCAGUUGUUGCUGAUCGUGAUUGGGGCUAUUGCAGUAGUCGCUGUCCUCCAGCCUUACAUUUUUGUCGCGACCGUUCCGGUGAUCGUGGCGUUUAUCAUGCUGCGGGCCUAUUUCUUGCAGACGUCACAGCAGCUUAAGCAACUGGAGUCUGAAGGGAGGUCGCCUAUCUUUACGCAUCUUGUGACCAGUUUGAAGGGAUUGUGGACGUUGCGCGCCUUUGGCAGGCAGCCCUACUUUGAAACACUGUUCCACAAAGCGCUGAAUCUCCAUACGGCAAAUUGGUUUUUGUAUUUGAGUACCCUCCGAUGGUUUCAGAUGCGCAUUGAGAUGAUUUUUGUGAUCUUCUUUAUCGCGGUGACUUUUAUCUCCAUCUUGACCACGGGAGAGGGCGAGGGACGGGUCGGUAUUAUCCUGACACUCGCCAUGAACAUUAUGAGCACUUUGCAGUGGGCAGUGAACAGCUCGAUUGAUGUGGAUAGCCUGAUGAGGUCCGUUUCGAGGGUCUUUAAGUUCAUCGACAUGCCGACGGAGGGAAAGCCCACAAAAAGUACGAAACCCUAUAAGAAUGGGCAAUUGAGUAAGGUAAUGAUCAUCGAGAACAGUCACGUGAAGAAGGAUGACAUCUGGCCUAGCGGGGGUCAGAUGACCGUGAAGGACCUGACGGCAAAAUACACCGAGGGAGGGAACGCAAUCCUUGAAAACAUCUCGUUCAGCAUUAGCCCCGGUCAGCGUGUGGGGUUGCUCGGGAGGACCGGGUCAGGAAAAUCGACGUUGCUGUCGGCCUUCUUGAGACUUCUGAAUACAGAGGGUGAGAUCCAGAUCGACGGCGUUUCGUGGGAUAGCAUCACCUUGCAGCAGUGGCGGAAAGCGUUUGGAGUAAUCCCCCAAAAGGUCUUUAUCUUUAGCGGAACCUUCCGAAAGAAUCUCGAUCCUUAUGAACAGUGGUCAGAUCAAGAGAUUUGGAAAGUCGCGGACGAGGUUGGCCUUCGGAGUGUAAUCGAGCAGUUUCCGGGAAAACUCGACUUUGUCCUUGUAGAUGGGGGAUGCGUCCUGUCGCAUGGGCACAAGCAGCUCAUGUGCCUGGCGCGAUCCGUCCUCUCUAAAGCGAAAAUUCUUCUCUUGGAUGAACCUUCGGCCCAUCUGGACCCGGUAACGUAUCAGAUCAUCAGAAGGACACUUAAGCAGGCGUUUGCCGACUGCACGGUGAUUCUCUGUGAGCAUCGUAUCGAGGCCAUGCUCGAAUGCCAGCAAUUUCUUGUCAUCGAAGAGAAUAAGGUCCGCCAGUACGACUCCAUCCAGAAGCUGCUUAAUGAGAGAUCAUUGUUCCGGCAGGCGAUUUCACCAUCCGAUAGGGUGAAACUUUUUCCACACAGAAAUUCGUCGAAGUGCAAGUCCAAACCGCAGAUCGCGGCCUUGAAAGAAGAGACUGAAGAAGAAGUUCAAGACACGCGUCUUUAA(配列番号3)
配列番号4
GGACAGAUCGCCUGGAGACGCCAUCCACGCUGUUUUGACCUCCAUAGAAGACACCGGGACCGAUCCAGCCUCCGCGGCCGGGAACGGUGCAUUGGAACGCGGAUUCCCCGUGCCAAGAGUGACUCACCGUCCUUGACACG(配列番号4)
配列番号5
CGGGUGGCAUCCCUGUGACCCCUCCCCAGUGCCUCUCCUGGCCCUGGAAGUUGCCACUCCAGUGCCCACCAGCCUUGUCCUAAUAAAAUUAAGUUGCAUC(配列番号5)
配列番号6
GGGAUCCUACC(配列番号6)
配列番号7
AUGGAAGAUGCCAAAAACAUUAAGAAGGGCCCAGCGCCAUUCUACCCACUCGAAGACGGGACCGCCGGCGAGCAGCUGCACAAAGCCAUGAAGCGCUACGCCCUGGUGCCCGGCACCAUCGCCUUUACCGACGCACAUAUCGAGGUGGACAUUACCUACGCCGAGUACUUCGAGAUGAGCGUUCGGCUGGCAGAAGCUAUGAAGCGCUAUGGGCUGAAUACAAACCAUCGGAUCGUGGUGUGCAGCGAGAAUAGCUUGCAGUUCUUCAUGCCCGUGUUGGGUGCCCUGUUCAUCGGUGUGGCUGUGGCCCCAGCUAACGACAUCUACAACGAGCGCGAGCUGCUGAACAGCAUGGGCAUCAGCCAGCCCACCGUCGUAUUCGUGAGCAAGAAAGGGCUGCAAAAGAUCCUCAACGUGCAAAAGAAGCUACCGAUCAUACAAAAGAUCAUCAUCAUGGAUAGCAAGACCGACUACCAGGGCUUCCAAAGCAUGUACACCUUCGUGACUUCCCAUUUGCCACCCGGCUUCAACGAGUACGACUUCGUGCCCGAGAGCUUCGACCGGGACAAAACCAUCGCCCUGAUCAUGAACAGUAGUGGCAGUACCGGAUUGCCCAAGGGCGUAGCCCUACCGCACCGCACCGCUUGUGUCCGAUUCAGUCAUGCCCGCGACCCCAUCUUCGGCAACCAGAUCAUCCCCGACACCGCUAUCCUCAGCGUGGUGCCAUUUCACCACGGCUUCGGCAUGUUCACCACGCUGGGCUACUUGAUCUGCGGCUUUCGGGUCGUGCUCAUGUACCGCUUCGAGGAGGAGCUAUUCUUGCGCAGCUUGCAAGACUAUAAGAUUCAAUCUGCCCUGCUGGUGCCCACACUAUUUAGCUUCUUCGCUAAGAGCACUCUCAUCGACAAGUACGACCUAAGCAACUUGCACGAGAUCGCCAGCGGCGGGGCGCCGCUCAGCAAGGAGGUAGGUGAGGCCGUGGCCAAACGCUUCCACCUACCAGGCAUCCGCCAGGGCUACGGCCUGACAGAAACAACCAGCGCCAUUCUGAUCACCCCCGAAGGGGACGACAAGCCUGGCGCAGUAGGCAAGGUGGUGCCCUUCUUCGAGGCUAAGGUGGUGGACUUGGACACCGGUAAGACACUGGGUGUGAACCAGCGCGGCGAGCUGUGCGUCCGUGGCCCCAUGAUCAUGAGCGGCUACGUUAACAACCCCGAGGCUACAAACGCUCUCAUCGACAAGGACGGCUGGCUGCACAGCGGCGACAUCGCCUACUGGGACGAGGACGAGCACUUCUUCAUCGUGGACCGGCUGAAGAGCCUGAUCAAAUACAAGGGCUACCAGGUAGCCCCAGCCGAACUGGAGAGCAUCCUGCUGCAACACCCCAACAUCUUCGACGCCGGGGUCGCCGGCCUGCCCGACGACGAUGCCGGCGAGCUGCCCGCCGCAGUCGUCGUGCUGGAACACGGUAAAACCAUGACCGAGAAGGAGAUCGUGGACUAUGUGGCCAGCCAGGUUACAACCGCCAAGAAGCUGCGCGGUGGUGUUGUGUUCGUGGACGAGGUGCCUAAAGGACUGACCGGCAAGUUGGACGCCCGCAAGAUCCGCGAGAUUCUCAUUAAGGCCAAGAAGGGCGGCAAGAUCGCCGUGUA(配列番号7)
配列番号8
UUUGAAUU(配列番号8)
配列番号9
AUGCAGAGAAGCCCCCUGGAAAAGGCCAGCGUGGUGUCCAAGCUGUUCUUCAGCUGGACCAGACCCAUCCUGAGAAAGGGCUACAGACAGAGACUGGAACUGAGCGACAUCUACCAGAUCCCCAGCGUGGACAGCGCCGACAACCUGAGCGAGAAGCUGGAAAGAGAGUGGGACAGAGAGCUGGCUAGCAAGAAGAACCCCAAGCUGAUCAACGCCCUGAGGCGGUGCUUCUUCUGGCGGUUUAUGUUCUACGGCAUCUUCCUGUACCUGGGCGAAGUGACAAAGGCCGUGCAGCCCCUGCUCCUGGGCAGAAUCAUUGCCAGCUACGACCCCGACAACAAAGAGGAAAGAUCUAUCGCCAUCUACCUGGGCAUCGGCCUGUGCCUGCUGUUCAUCGUGCGGACACUGCUGCUGCACCCCGCCAUCUUCGGCCUGCACCACAUCGGCAUGCAGAUGAGAAUCGCCAUGUUCAGCCUGAUCUACAAGAAAACCCUGAAGCUGAGCAGCAGGGUGCUGGACAAGAUCAGCAUCGGACAGCUGGUGUCCCUGCUGAGCAACAACCUGAACAAGUUCGACGAGGGACUGGCCCUGGCUCACUUCGUGUGGAUCGCUCCACUGCAGGUCGCCCUGCUGAUGGGCCUGAUCUGGGAGCUGCUGCAGGCCAGCGCUUUCUGCGGCCUGGGCUUUCUGAUUGUGCUGGCCCUGUUUCAGGCUGGCCUGGGCAGGAUGAUGAUGAAGUACAGGGACCAGAGAGCCGGCAAGAUCAGCGAGAGACUGGUCAUCACCAGCGAGAUGAUCGAGAACAUCCAGAGCGUGAAGGCCUACUGCUGGGAAGAGGCCAUGGAAAAGAUGAUCGAAAACCUGAGACAGACCGAGCUGAAGCUGACCAGAAAGGCCGCCUACGUGCGGUACUUCAACAGCAGCGCCUUCUUCUUCUCCGGCUUCUUCGUGGUGUUCCUGUCCGUGCUGCCCUACGCCCUGAUCAAGGGCAUCAUCCUGAGGAAGAUCUUCACCACCAUUUCUUUCUGCAUCGUGCUGAGAAUGGCCGUGACCAGACAGUUCCCCUGGGCCGUGCAGACUUGGUACGACAGCCUGGGCGCCAUCAACAAGAUCCAGGACUUCCUGCAGAAGCAGGAGUACAAGACCCUCGAGUACAACCUGACCACCACCGAGGUGGUCAUGGAAAACGUGACCGCCUUCUGGGAGGAAGGCUUCGGCGAGCUGUUCGAGAAGGCCAAGCAGAACAACAACAACAGAAAGACCAGCAACGGCGACGACUCCCUGUUCUUCUCCAACUUCUCCCUGCUGGGCACCCCCGUGCUGAAGGACAUCAACUUCAAGAUCGAGAGAGGCCAGCUGCUCGCCGUGGCCGGCUCUACAGGCGCUGGCAAGACCUCUCUGCUGAUGGUCAUCAUGGGCGAGCUGGAACCCAGCGAGGGCAAGAUCAAGCACAGCGGCAGAAUCAGCUUCUGCAGCCAGUUCAGCUGGAUCAUGCCCGGCACCAUCAAAGAGAACAUCAUCUUCGGCGUGUCCUACGACGAGUACAGAUACAGAAGCGUGAUCAAGGCCUGCCAGCUGGAAGAGGACAUCAGCAAGUUCGCCGAGAAGGACAACAUCGUGCUGGGCGAGGGCGGCAUCACCCUGUCUGGCGGCCAGAGAGCCAGAAUCAGCCUGGCCAGAGCCGUGUACAAGGACGCCGACCUGUACCUGCUGGACAGCCCCUUCGGCUACCUGGACGUGCUGACCGAGAAAGAGAUCUUCGAGAGCUGCGUGUGCAAGCUGAUGGCCAACAAGACCAGAAUCCUGGUCACCAGCAAGAUGGAACACCUGAAGAAGGCCGACAAGAUCCUGAUCCUGCACGAGGGCAGCAGCUACUUCUACGGCACAUUCAGCGAGCUGCAGAACCUGCAGCCCGACUUCAGCAGCAAACUGAUGGGCUGCGACAGCUUCGACCAGUUCAGCGCCGAGAGAAGAAACAGCAUCCUGACCGAGACACUGCACAGAUUCAGCCUGGAAGGCGACGCCCCCGUGUCUUGGACCGAGACAAAGAAGCAGAGCUUCAAGCAGACCGGCGAGUUCGGCGAGAAGAGAAAGAACUCCAUCCUGAACCCCAUCAACAGCAUCCGGAAGUUCAGCAUCGUGCAGAAAACCCCCCUGCAGAUGAACGGCAUCGAAGAGGACAGCGACGAGCCCCUGGAAAGACGGCUGAGCCUGGUGCCUGACAGCGAGCAGGGCGAGGCCAUCCUGCCUAGAAUCAGCGUGAUCAGCACCGGCCCCACCCUGCAGGCUAGAAGGCGGCAGAGCGUGCUGAACCUGAUGACCCACAGCGUGAACCAGGGCCAGAACAUCCACCGCAAGACCACCGCCAGCACCAGAAAGGUGUCCCUGGCUCCUCAGGCCAACCUGACCGAGCUGGACAUCUACAGCAGAAGGCUGAGCCAGGAAACCGGCCUGGAAAUCAGCGAGGAAAUCAACGAAGAGGACCUGAAAGAGUGCUUCUUCGACGACAUGGAAUCCAUCCCCGCCGUGACCACCUGGAACACCUACCUGCGGUACAUCACCGUGCACAAGAGCCUGAUCUUCGUGCUGAUCUGGUGCCUGGUCAUCUUCCUGGCCGAGGUGGCCGCCAGCCUGGUGGUGCUGUGGCUCCUGGGAAACACCCCUCUGCAGGACAAGGGCAACAGCACCCACAGCAGAAACAACAGCUACGCCGUGAUCAUCACCUCCACCAGCUCCUACUACGUGUUCUACAUCUACGUGGGCGUGGCCGACACCCUGCUGGCUAUGGGCUUCUUCAGAGGCCUGCCCCUGGUGCACACCCUGAUCACCGUGUCCAAGAUCCUGCACCAUAAGAUGCUGCACAGCGUGCUGCAGGCUCCCAUGAGCACCCUGAACACACUGAAGGCUGGCGGCAUCCUGAACAGGUUCAGCAAGGAUAUCGCCAUCCUGGACGACCUGCUGCCUCUGACCAUCUUCGACUUCAUCCAGCUGCUGCUGAUCGUGAUCGGCGCUAUCGCCGUGGUGGCCGUGCUGCAGCCCUACAUCUUCGUGGCCACCGUGCCCGUGAUCGUGGCCUUCAUUAUGCUGAGAGCCUACUUUCUGCAGACCAGCCAGCAGCUGAAGCAGCUGGAAAGCGAGGGCAGAAGCCCCAUCUUCACCCACCUCGUGACCAGCCUGAAGGGCCUGUGGACCCUGAGAGCCUUCGGCAGACAGCCCUACUUCGAGACACUGUUCCACAAGGCCCUGAACCUGCACACCGCCAACUGGUUUCUGUACCUGUCCACCCUGAGAUGGUUCCAGAUGAGGAUCGAGAUGAUCUUCGUCAUCUUCUUUAUCGCCGUGACCUUCAUCUCUAUCCUGACCACCGGCGAGGGCGAGGGAAGAGUGGGAAUCAUCCUGACCCUGGCCAUGAACAUCAUGAGCACACUGCAGUGGGCCGUGAACAGCAGCAUCGACGUGGACAGCCUGAUGAGAAGCGUGUCCAGAGUGUUCAAGUUCAUCGACAUGCCUACCGAGGGCAAGCCCACCAAGAGCACCAAGCCCUACAAGAACGGCCAGCUGAGCAAAGUGAUGAUCAUCGAGAACAGCCACGUCAAGAAGGACGACAUCUGGCCCAGCGGCGGACAGAUGACCGUGAAGGACCUGACCGCCAAGUACACAGAGGGCGGCAACGCUAUCCUGGAAAACAUCAGCUUCAGCAUCAGCCCAGGCCAGAGAGUGGGCCUGCUGGGGAGAACAGGCAGCGGCAAGUCUACCCUGCUGUCCGCCUUCCUGAGACUGCUGAACACCGAGGGCGAGAUCCAGAUCGAUGGCGUGUCCUGGGACUCCAUCACCCUGCAGCAGUGGCGCAAGGCCUUCGGCGUGAUCCCCCAGAAGGUGUUCAUCUUCAGCGGCACCUUCAGAAAGAACCUGGACCCCUACGAGCAGUGGUCCGACCAGGAAAUCUGGAAGGUCGCCGAUGAAGUGGGCCUGAGAUCCGUGAUCGAGCAGUUCCCCGGCAAGCUGGACUUCGUGCUGGUGGACGGCGGCUGCGUGCUGAGCCACGGCCACAAGCAGCUGAUGUGUCUGGCCCGCUCCGUGCUGAGCAAGGCUAAGAUUCUGCUGCUGGACGAGCCUAGCGCCCACCUGGACCCUGUGACCUACCAGAUCAUCAGAAGGACCCUGAAGCAGGCCUUCGCCGACUGCACCGUGAUCCUGUGCGAGCACAGAAUCGAGGCCAUGCUGGAAUGCCAGCAGUUCCUGGUCAUCGAAGAGAACAAAGUGCGGCAGUACGACAGCAUCCAGAAGCUGCUGAACGAGAGAAGCCUGUUCAGACAGGCCAUCAGCCCCAGCGACAGAGUGAAGCUGUUCCCCCACCGCAACAGCAGCAAGUGCAAGAGCAAGCCCCAGAUCGCCGCCCUGAAAGAAGAGACUGAGGAAGAGGUGCAGGACACCAGACUGUGA(配列番号9)
配列番号10
AUGCAGCGGUCCCCGCUCGAAAAGGCCAGUGUCGUGUCCAAACUCUUCUUCUCAUGGACUCGGCCUAUCCUUAGAAAGGGGUAUCGGCAGAGGCUUGAGUUGUCUGACAUCUACCAGAUCCCCUCGGUAGAUUCGGCGGAUAACCUCUCGGAGAAGCUCGAACGGGAAUGGGACCGCGAACUCGCGUCUAAGAAAAACCCGAAGCUCAUCAACGCACUGAGAAGGUGCUUCUUCUGGCGGUUCAUGUUCUACGGUAUCUUCUUGUAUCUCGGGGAGGUCACAAAAGCAGUCCAACCCCUGUUGUUGGGUCGCAUUAUCGCCUCGUACGACCCCGAUAACAAAGAAGAACGGAGCAUCGCGAUCUACCUCGGGAUCGGACUGUGUUUGCUUUUCAUCGUCAGAACACUUUUGUUGCAUCCAGCAAUCUUCGGCCUCCAUCACAUCGGUAUGCAGAUGCGAAUCGCUAUGUUUAGCUUGAUCUACAAAAAGACACUGAAACUCUCGUCGCGGGUGUUGGAUAAGAUUUCCAUCGGUCAGUUGGUGUCCCUGCUUAGUAAUAACCUCAACAAAUUCGAUGAGGGACUGGCGCUGGCACAUUUCGUGUGGAUUGCCCCGCUGCAAGUCGCACUGCUUAUGGGACUGAUUUGGGAACUGUUGCAGGCCAGCGCCUUUUGCGGCCUGGGAUUUCUCAUUGUGCUUGCACUUUUCCAAGCAGGGCUCGGCAGAAUGAUGAUGAAGUACAGGGACCAGAGAGCCGGAAAGAUCUCAGAACGGCUCGUGAUUACUUCAGAAAUGAUCGAGAACAUUCAAUCGGUGAAAGCGUACUGCUGGGAAGAGGCGAUGGAAAAGAUGAUCGAAAACCUCAGACAGACCGAGUUGAAGCUGACCCGGAAGGCCGCGUACGUCAGAUACUUCAACAGCAGCGCUUUCUUCUUCUCGGGCUUCUUCGUCGUGUUCCUGUCGGUGCUGCCGUAUGCCCUCAUUAAGGGAAUUAUCUUGCGGAAGAUCUUUACUACUAUCUCAUUUUGCAUCGUCCUUCGGAUGGCGGUCACUCGGCAGUUCCCGUGGGCCGUGCAGACCUGGUACGACAGCCUCGGGGCCAUCAACAAGAUCCAAGACUUUCUCCAAAAGCAAGAGUACAAAACCCUCGAAUACAACCUCACCACUACUGAAGUGGUCAUGGAAAACGUGACCGCCUUUUGGGAAGAAGGCUUCGGAGAACUGUUCGAGAAGGCGAAGCAAAACAACAAUAAUCGCAAGACUAGCAACGGGGAUGACUCACUGUUCUUCAGCAAUUUCUCACUGCUCGGCACCCCGGUGCUUAAGGACAUCAACUUCAAGAUUGAACGCGGACAGCUCUUGGCGGUGGCCGGAUCCACCGGAGCAGGAAAGACUAGCCUGCUGAUGGUGAUCAUGGGUGAGCUGGAACCGUCCGAAGGCAAAAUCAAGCACUCCGGCAGAAUCAGCUUCUGCUCGCAGUUUUCGUGGAUCAUGCCAGGAACCAUCAAAGAGAACAUCAUCUUUGGAGUCUCAUACGAUGAGUACCGCUACAGAAGCGUGAUUAAGGCCUGCCAGCUUGAAGAGGACAUCUCCAAGUUCGCGGAAAAGGACAACAUCGUGCUGGGUGAGGGAGGGAUCACGUUGUCGGGCGGUCAGAGAGCCCGCAUUUCGCUGGCACGGGCUGUGUACAAGGAUGCGGAUCUUUACCUUCUGGACUCGCCAUUCGGUUACCUCGACGUGCUGACCGAAAAAGAAAUCUUCGAGAGCUGCGUGUGUAAGCUGAUGGCUAAUAAGACUAGAAUCCUCGUGACGUCCAAAAUGGAACAUCUUAAGAAGGCGGAUAAGAUUCUCAUUCUUCACGAGGGGUCGAGCUACUUCUACGGGACUUUUAGCGAGCUGCAGAAUUUGCAGCCGGACUUCAGCUCAAAGCUCAUGGGCUGCGACUCGUUCGAUCAGUUCAGCGCCGAACGGCGCAAUUCGAUCUUGACGGAAACCCUGCACAGAUUCUCGCUGGAGGGAGAUGCACCUGUCUCGUGGACCGAAACCAAGAAGCAGUCCUUCAAGCAGACGGGAGAGUUCGGAGAAAAGCGGAAGAACUCAAUCCUCAACCCAAUCAACUCCAUUCGCAAAUUCUCAAUCGUGCAGAAAACUCCACUGCAGAUGAACGGUAUCGAAGAGGAUUCGGACGAGCCACUUGAGCGGAGACUGUCGCUGGUGCCAGAUUCAGAACAGGGGGAGGCAAUCCUGCCGCGCAUUUCCGUGAUCAGCACUGGGCCGACCCUCCAAGCUAGACGCAGGCAAUCAGUGCUGAAUCUCAUGACCCACUCCGUCAACCAGGGACAGAAUAUCCACCGCAAGACCACCGCGUCGACUAGAAAGGUGUCAUUGGCACCGCAAGCAAAUUUGACUGAACUUGACAUCUACUCACGGCGCCUCUCCCAAGAAACCGGAUUGGAAAUCUCCGAAGAGAUUAACGAAGAAGAUUUGAAAGAGUGUUUCUUCGACGAUAUGGAGUCGAUCCCCGCAGUGACCACUUGGAAUACGUAUCUUCGGUACAUCACCGUGCACAAGAGCCUGAUCUUCGUCCUCAUCUGGUGCCUGGUGAUCUUUCUGGCCGAAGUCGCCGCUUCGCUGGUCGUGCUGUGGCUGCUCGGUAAUACCCCGCUCCAAGACAAAGGCAAUUCCACUCACUCGCGCAACAACAGCUACGCUGUGAUUAUCACGUCAACCUCGUCGUACUAUGUGUUCUACAUCUACGUGGGAGUCGCGGACACUCUGCUCGCUAUGGGCUUCUUUCGCGGACUGCCCCUGGUCCACACUCUCAUCACGGUGAGCAAGAUCCUCCAUCAUAAGAUGCUCCAUUCCGUGCUGCAGGCCCCGAUGAGCACUCUCAACACUCUGAAGGCGGGUGGAAUCUUGAACAGAUUUUCCAAAGACAUCGCGAUUCUGGACGAUCUGCUCCCACUCACUAUCUUCGACUUCAUCCAACUGCUGCUGAUCGUCAUCGGAGCUAUCGCCGUGGUGGCUGUCCUCCAGCCGUAUAUCUUCGUGGCCACUGUGCCGGUGAUUGUCGCUUUCAUCAUGUUGCGCGCGUACUUCUUGCAAACCUCGCAGCAACUCAAGCAACUGGAGUCCGAGGGCCGGAGCCCAAUCUUUACCCAUCUGGUGACUUCACUGAAAGGUCUGUGGACCCUCCGCGCCUUUGGUCGCCAGCCUUACUUCGAAACUCUCUUUCACAAAGCACUGAAUCUCCACACUGCAAACUGGUUCUUGUACCUGUCCACCCUGCGGUGGUUCCAAAUGCGGAUCGAGAUGAUCUUUGUCAUCUUCUUCAUCGCCGUGACUUUUAUCUCCAUCCUCACCACCGGCGAGGGAGAGGGGAGAGUGGGAAUCAUCCUGACGCUGGCGAUGAAUAUCAUGUCCACUUUGCAGUGGGCCGUCAAUUCGAGCAUCGACGUGGAUUCGCUGAUGCGCAGCGUGUCGCGCGUGUUCAAGUUCAUCGAUAUGCCCACCGAAGGUAAACCCACCAAGAGCACGAAGCCUUACAAGAACGGGCAGCUCUCAAAGGUGAUGAUUAUCGAGAACUCCCAUGUGAAGAAGGACGACAUCUGGCCAUCCGGAGGACAGAUGACCGUGAAGGACCUGACCGCCAAAUACACGGAGGGCGGAAAUGCAAUCCUCGAAAACAUCUCGUUCUCCAUCUCGCCUGGCCAAAGGGUGGGACUUUUGGGACGCACUGGAUCCGGAAAGAGCACCCUGCUUAGCGCCUUCUUGAGGCUCUUGAACACCGAGGGCGAAAUCCAGAUCGAUGGCGUGUCGUGGGAUUCGAUCACCCUGCAGCAGUGGAGAAAGGCCUUCGGGGUGAUCCCGCAAAAAGUGUUCAUCUUCUCCGGAACGUUUCGGAAAAACCUUGACCCAUACGAACAAUGGUCGGAUCAAGAGAUUUGGAAGGUCGCCGACGAAGUGGGGCUGCGCUCCGUGAUCGAGCAGUUUCCGGGAAAACUGGACUUCGUCUUGGUCGACGGCGGAUGCGUCCUGUCCCACGGACAUAAGCAGCUGAUGUGCCUGGCCCGCAGCGUCCUUUCAAAAGCUAAGAUCCUGCUGCUGGAUGAACCUUCAGCACACCUCGACCCGGUCACCUACCAGAUCAUCAGACGGACCCUGAAACAGGCCUUUGCGGAUUGUACUGUGAUCUUGUGUGAACACCGCAUUGAAGCCAUGCUGGAGUGCCAGCAGUUCCUGGUCAUCGAAGAGAACAAAGUGCGGCAGUACGAUUCCAUCCAAAAACUGCUCAAUGAGCGGUCCCUGUUCAGACAGGCAAUUAGCCCGAGCGACAGGGUCAAAUUGUUCCCCCAUAGAAAUUCGUCGAAAUGUAAGUCAAAGCCUCAGAUCGCGGCACUGAAAGAAGAAACUGAAGAAGAGGUGCAAGACACCAGACUGUGA(配列番号10)
配列番号11
AUGCAGAGAAGCCCACUGGAAAAGGCGUCGGUGGUGUCAAAGCUGUUCUUUAGCUGGACCAGACCUAUCUUGCGGAAGGGAUACCGCCAACGCCUGGAGCUGUCGGACAUCUACCAGAUUCCGUCAGUGGAUUCAGCAGACAAUCUCUCCGAAAAGCUGGAACGCGAAUGGGACAGAGAGUUGGCGUCAAAGAAGAACCCAAAGUUGAUCAAUGCCCUGCGCCGCUGCUUCUUCUGGCGGUUCAUGUUCUACGGAAUCUUUCUGUACCUCGGCGAAGUCACCAAGGCUGUGCAACCGCUUCUGCUGGGACGCAUCAUCGCCUCAUACGACCCGGACAACAAGGAAGAACGCUCCAUCGCAAUCUACCUCGGGAUCGGCCUCUGCCUGCUGUUUAUCGUGCGGACGCUGCUGCUCCAUCCAGCCAUUUUCGGACUGCACCACAUUGGCAUGCAAAUGCGGAUCGCCAUGUUCAGCCUGAUCUACAAAAAGACCCUGAAGUUGAGCUCACGGGUGUUGGAUAAGAUUUCGAUCGGACAGCUGGUGUCGCUGCUCUCCAACAACCUCAACAAGUUUGACGAAGGCCUGGCACUGGCCCACUUCGUGUGGAUUGCCCCGUUGCAAGUCGCCCUUUUGAUGGGCCUUAUUUGGGAGCUGUUGCAGGCAUCUGCCUUUUGUGGCCUGGGAUUUCUGAUUGUGUUGGCAUUGUUUCAGGCUGGGCUUGGGCGGAUGAUGAUGAAGUAUCGCGACCAGAGAGCGGGUAAAAUCUCGGAAAGACUCGUCAUCACUUCGGAAAUGAUCGAAAACAUCCAGUCGGUCAAAGCCUAUUGCUGGGAAGAAGCUAUGGAGAAGAUGAUUGAAAACCUCCGCCAAACUGAGCUGAAACUGACCCGCAAGGCGGCGUAUGUCCGGUAUUUCAAUUCGUCAGCGUUCUUCUUUUCCGGGUUCUUCGUUGUCUUUCUCUCGGUUUUGCCUUAUGCCUUGAUUAAGGGGAUUAUCCUCCGCAAGAUUUUCACCACGAUUUCGUUCUGCAUUGUAUUGCGCAUGGCAGUGACACGGCAAUUUCCGUGGGCCGUGCAGACAUGGUAUGACUCGCUUGGAGCGAUCAACAAAAUCCAAGACUUCUUGCAAAAGCAAGAGUACAAGACCCUGGAGUACAAUCUUACUACUACGGAGGUAGUAAUGGAGAAUGUGACGGCUUUUUGGGAGGAAGGAUUCGGCGAAUUGUUCGAAAAGGCUAAGCAGAACAACAACAAUCGGAAAACCUCCAAUGGGGACGAUUCGCUGUUCUUCUCGAAUUUCUCCCUGCUGGGAACGCCCGUGCUUAAAGACAUCAACUUCAAGAUCGAACGGGGCCAGCUGCUCGCGGUCGCGGGCAGCACUGGAGCGGGAAAGACUUCCCUGCUCAUGGUCAUCAUGGGAGAGCUGGAGCCCUCGGAGGGCAAAAUCAAGCACUCGGGGAGGAUCUCAUUUUGCAGCCAGUUCUCGUGGAUCAUGCCCGGUACUAUCAAAGAAAACAUCAUCUUUGGAGUCAGCUAUGACGAGUACCGCUACCGGUCGGUGAUCAAGGCCUGCCAGCUGGAAGAAGAUAUCUCCAAGUUCGCCGAAAAGGACAACAUUGUGCUGGGAGAAGGUGGAAUCACUCUCUCGGGAGGCCAGCGCGCACGGAUCUCACUCGCAAGGGCCGUGUACAAGGAUGCCGAUUUGUACCUGUUGGAUUCGCCGUUCGGUUAUCUUGAUGUCCUCACUGAGAAAGAGAUUUUUGAGUCGUGCGUCUGUAAGCUGAUGGCCAACAAAACCCGCAUCCUGGUGACCUCGAAGAUGGAGCACUUGAAGAAGGCCGACAAAAUCCUUAUCCUCCAUGAGGGUAGCUCAUACUUCUACGGCACCUUUUCGGAACUGCAGAAUCUGCAGCCCGACUUCUCAUCAAAACUGAUGGGAUGUGACUCGUUCGAUCAGUUCUCGGCGGAGCGGCGGAACUCGAUCCUCACCGAAACUCUCCACCGGUUCAGCCUCGAGGGAGAUGCCCCAGUCAGCUGGACCGAAACUAAGAAGCAGUCCUUCAAACAGACCGGAGAGUUCGGAGAAAAACGCAAGAACUCCAUCCUCAAUCCAAUCAACAGCAUCCGCAAGUUCAGCAUCGUGCAGAAAACUCCACUUCAGAUGAACGGAAUCGAAGAGGAUAGCGACGAGCCGCUUGAGCGGAGAUUGUCACUGGUGCCGGACAGCGAGCAAGGGGAAGCGAUUCUGCCGCGGAUCUCCGUGAUCUCGACUGGCCCUACCCUCCAAGCUCGCAGACGCCAGAGCGUGCUGAAUCUCAUGACCCACUCAGUCAACCAGGGACAAAACAUCCAUAGAAAGACCACCGCUUCAACCCGGAAAGUGUCACUUGCACCGCAGGCAAACCUGACCGAACUCGACAUCUACAGCAGACGGCUCUCACAAGAAACUGGAUUGGAGAUCAGCGAAGAGAUCAACGAAGAAGAUCUCAAAGAAUGCUUCUUCGACGAUAUGGAGUCCAUCCCAGCAGUCACUACGUGGAAUACCUACCUCCGCUACAUCACUGUGCACAAGAGCCUGAUUUUCGUGUUGAUCUGGUGCCUGGUCAUCUUCUUGGCCGAGGUGGCCGCGAGCCUCGUGGUCCUCUGGCUGCUCGGCAAUACGCCGCUGCAAGAUAAGGGAAAUUCCACGCAUAGCAGAAACAACUCAUACGCAGUGAUCAUCACUAGCACUUCAUCGUACUACGUGUUCUACAUCUACGUGGGGGUGGCCGAUACUCUGUUGGCAAUGGGAUUCUUUAGAGGGCUGCCUCUGGUGCAUACUCUGAUCACUGUGUCCAAGAUCCUCCACCACAAGAUGCUCCACUCCGUGCUUCAGGCCCCUAUGUCAACUCUCAACACCCUCAAGGCCGGAGGUAUUCUUAAUCGCUUUUCCAAGGACAUCGCCAUUCUCGAUGACUUGCUUCCCCUGACUAUCUUCGACUUUAUCCAGUUGCUGCUGAUUGUGAUCGGCGCUAUUGCCGUCGUCGCAGUGCUGCAACCGUACAUCUUUGUGGCUACCGUCCCAGUCAUUGUGGCCUUCAUCAUGCUCAGGGCAUACUUUCUCCAGACCAGCCAGCAGCUCAAGCAGCUCGAAUCCGAAGGCAGAUCGCCGAUCUUCACCCACCUCGUCACUUCGCUCAAGGGCCUCUGGACCCUGCGCGCCUUCGGUCGCCAGCCGUAUUUCGAAACCCUGUUCCAUAAAGCACUGAACCUCCAUACUGCGAACUGGUUUCUCUACCUUUCAACCCUGAGGUGGUUCCAGAUGAGAAUCGAGAUGAUCUUUGUGAUCUUCUUUAUCGCUGUGACGUUCAUCUCCAUUCUCACUACCGGCGAGGGAGAGGGCAGAGUGGGGAUUAUCCUCACGCUGGCCAUGAAUAUCAUGAGCACGCUGCAGUGGGCCGUCAAUAGCAGCAUCGACGUGGACUCCCUGAUGCGGUCCGUGUCGAGAGUGUUUAAGUUCAUCGAUAUGCCUACUGAAGGGAAACCGACCAAGUCGACCAAGCCGUACAAGAAUGGGCAGCUGAGCAAGGUGAUGAUUAUUGAGAACUCCCAUGUGAAGAAGGACGACAUCUGGCCCAGCGGAGGCCAGAUGACCGUGAAGGACUUGACCGCUAAGUACACUGAGGGUGGAAAUGCCAUUCUUGAGAAUAUCAGCUUCUCGAUCUCGCCGGGACAACGCGUGGGAUUGCUCGGGCGCACUGGCAGCGGCAAAUCCACCCUGCUUAGCGCUUUUCUGAGGCUGCUGAACACUGAAGGUGAAAUUCAAAUCGAUGGAGUGUCGUGGGAUAGCAUCACCCUUCAACAGUGGCGCAAGGCCUUCGGCGUGAUCCCUCAAAAGGUCUUUAUCUUCUCGGGGACGUUCCGGAAAAAUCUCGACCCCUACGAACAGUGGUCAGACCAAGAGAUUUGGAAAGUCGCAGAUGAGGUCGGACUGCGCUCAGUGAUCGAACAGUUUCCGGGUAAACUUGACUUCGUGCUCGUCGAUGGAGGUUGCGUCCUGUCCCACGGACAUAAGCAGCUGAUGUGUCUGGCGCGCUCGGUCCUCUCCAAAGCGAAGAUCCUGCUGCUCGAUGAACCGUCCGCCCACCUUGAUCCAGUGACCUAUCAGAUCAUUCGGAGAACUUUGAAGCAAGCCUUCGCUGACUGCACCGUCAUCCUCUGCGAACACCGGAUCGAGGCAAUGCUGGAGUGCCAACAGUUUCUGGUCAUCGAAGAAAACAAAGUGCGCCAGUAUGACUCGAUCCAAAAACUUCUGAACGAGCGCUCCCUCUUCCGGCAGGCAAUCAGCCCAUCCGACCGCGUGAAGUUGUUCCCUCAUCGGAAUAGCUCCAAAUGCAAAUCGAAGCCGCAGAUCGCUGCCUUGAAAGAAGAAACCGAAGAAGAAGUCCAAGACACUAGGUUGUAG(配列番号11)
配列番号12
AUGCAGCGGUCCCCUCUGGAGAAGGCUUCCGUGGUCAGCAAGCUGUUCUUCUCGUGGACCAGACCUAUCCUCCGCAAGGGAUACCGCCAGCGCCUGGAGCUGUCAGAUAUCUACCAGAUCCCAAGCGUGGACUCAGCCGACAAUCUGAGCGAAAAGCUGGAACGGGAGUGGGACCGGGAGCUCGCCUCCAAGAAGAAUCCGAAGUUGAUCAAUGCGCUGCGCAGAUGCUUCUUCUGGCGGUUUAUGUUUUACGGCAUCUUUCUGUAUCUCGGAGAAGUGACCAAAGCCGUGCAGCCGCUGCUCUUGGGUAGGAUCAUUGCUUCGUACGACCCGGACAACAAAGAAGAACGCUCCAUCGCCAUCUACCUCGGAAUCGGUCUGUGCCUGCUCUUUAUCGUGCGCACUCUCCUGCUGCAUCCGGCGAUCUUCGGACUGCACCACAUCGGCAUGCAAAUGCGGAUCGCAAUGUUCUCACUGAUCUACAAAAAGACUCUGAAGCUCAGCUCCAGAGUGCUGGAUAAGAUCUCGAUCGGGCAACUCGUCAGCCUGCUGUCGAACAAUCUGAAUAAGUUCGACGAAGGGUUGGCCCUCGCACAUUUCGUGUGGAUCGCACCGCUGCAAGUGGCGCUCCUGAUGGGACUCAUUUGGGAACUGCUCCAAGCCAGCGCGUUUUGCGGACUCGGAUUCCUGAUCGUGCUCGCCCUGUUCCAAGCCGGACUGGGGCGCAUGAUGAUGAAGUACCGCGAUCAGCGGGCAGGAAAGAUCUCCGAGCGGUUGGUGAUCACUUCCGAAAUGAUCGAGAAUAUUCAGUCCGUGAAGGCCUACUGCUGGGAAGAAGCUAUGGAAAAGAUGAUUGAAAACUUGCGGCAAACUGAGCUGAAAUUGACUCGCAAAGCGGCAUACGUCCGCUACUUCAAUAGCAGCGCCUUCUUCUUUUCGGGCUUUUUCGUGGUGUUUCUGAGCGUGCUGCCCUACGCUCUGAUCAAGGGAAUCAUCCUCCGGAAAAUCUUCACCACCAUUUCGUUCUGUAUCGUGUUGCGCAUGGCCGUGACUCGCCAGUUCCCCUGGGCGGUGCAGACCUGGUACGACAGCUUGGGGGCAAUCAAUAAGAUUCAAGACUUCUUGCAAAAGCAGGAGUACAAGACUCUGGAGUACAACCUGACCACCACUGAAGUCGUGAUGGAGAACGUGACCGCCUUUUGGGAAGAGGGUUUUGGAGAACUGUUUGAGAAAGCAAAGCAGAAUAACAACAACCGCAAGACCUCAAAUGGGGACGAUUCCCUGUUUUUCUCGAACUUCUCCCUGCUCGGAACACCCGUGUUGAAGGACAUCAAUUUCAAGAUUGAGAGGGGACAGCUUCUCGCGGUAGCGGGAAGCACUGGUGCGGGAAAAACUAGCCUCUUGAUGGUGAUUAUGGGGGAGCUUGAGCCCAGCGAGGGGAAGAUUAAACACUCCGGGCGUAUCUCAUUCUGUAGCCAGUUUUCAUGGAUCAUGCCCGGAACCAUUAAAGAGAACAUCAUUUUCGGAGUAUCCUAUGAUGAGUACCGAUACAGAUCGGUCAUUAAGGCGUGCCAGUUGGAAGAGGACAUUUCUAAGUUCGCCGAGAAGGAUAACAUCGUCUUGGGAGAAGGGGGUAUUACAUUGUCGGGAGGGCAGCGAGCGCGGAUCAGCCUCGCGAGAGCGGUAUACAAAGAUGCAGAUUUGUAUCUGCUUGAUUCACCGUUUGGAUACCUCGACGUAUUGACAGAAAAAGAAAUCUUCGAGUCGUGCGUGUGUAAACUUAUGGCUAAUAAGACGAGAAUCCUGGUGACUUCCAAAAUGGAGCAUCUCAAGAAGGCGGACAAGAUCCUGAUUCUGCAUGAGGGAUCAAGCUAUUUCUACGGAACUUUUUCCGAGCUGCAGAACCUCCAGCCGGAUUUUAGCUCCAAGCUGAUGGGUUGCGACUCAUUCGACCAAUUCUCGGCUGAGCGGCGGAACUCAAUCCUGACCGAAACCCUGCAUCGCUUCUCCCUUGAGGGAGAUGCCCCGGUGUCGUGGACUGAGACUAAAAAGCAGUCGUUUAAGCAAACUGGCGAAUUCGGCGAAAAGCGGAAGAAUAGCAUCCUCAACCCAAUCAACAGCAUUCGGAAGUUCAGCAUCGUCCAAAAGACCCCGCUCCAGAUGAACGGCAUUGAAGAGGACUCAGACGAGCCAUUGGAAAGACGCCUGUCACUGGUCCCAGAUUCGGAGCAGGGUGAAGCAAUUCUGCCUCGGAUCUCGGUCAUCUCGACUGGCCCCACUCUCCAAGCUCGGCGGAGACAGAGCGUGCUUAACUUGAUGACCCACUCCGUGAACCAGGGUCAGAACAUCCACCGCAAAACCACCGCCUCCACCAGGAAGGUGUCACUGGCCCCUCAAGCCAAUCUGACUGAGUUGGAUAUCUACUCCAGAAGGCUCAGCCAGGAAACCGGACUGGAAAUCUCGGAAGAGAUCAACGAAGAGGAUCUCAAAGAGUGUUUCUUCGACGACAUGGAAUCAAUCCCUGCUGUCACUACUUGGAACACCUAUCUCCGCUACAUUACCGUGCACAAGUCACUCAUCUUCGUCCUGAUCUGGUGCCUCGUGAUCUUCCUGGCCGAGGUCGCAGCAUCGCUGGUCGUGCUGUGGCUGCUCGGCAACACCCCACUCCAAGACAAAGGCAACAGCACCCAUUCCCGCAACAACUCCUACGCGGUGAUCAUCACUUCAACUUCGUCCUACUACGUCUUUUACAUCUACGUGGGCGUGGCGGACACGCUCCUGGCUAUGGGGUUCUUUCGCGGGCUGCCUCUUGUCCACACGCUCAUCACUGUGUCAAAGAUUCUCCACCACAAAAUGCUGCACUCCGUGCUCCAGGCCCCUAUGUCGACUUUGAACACGCUUAAGGCCGGAGGCAUCCUUAACAGAUUCUCGAAAGAUAUCGCGAUCUUGGACGAUCUUCUGCCGCUGACUAUCUUUGACUUCAUCCAACUCCUGCUGAUCGUCAUCGGUGCCAUCGCAGUGGUCGCGGUGCUCCAACCGUACAUUUUCGUGGCGACUGUGCCGGUGAUCGUGGCGUUCAUCAUGCUGCGGGCUUACUUUCUUCAGACCUCACAGCAGCUGAAGCAACUCGAAUCGGAGGGUAGAUCACCAAUCUUUACCCACCUCGUCACCUCGCUGAAGGGACUCUGGACCCUGCGCGCAUUUGGACGGCAACCGUACUUCGAGACUCUCUUCCAUAAGGCCCUGAAUCUGCAUACGGCGAAUUGGUUUCUUUACCUCUCGACGCUCCGCUGGUUCCAGAUGCGCAUUGAGAUGAUUUUCGUCAUCUUUUUCAUCGCGGUGACCUUCAUCUCCAUCCUCACCACGGGUGAGGGAGAGGGCAGAGUCGGAAUUAUCCUCACUCUGGCCAUGAACAUCAUGUCCACUCUGCAGUGGGCCGUCAACUCAUCCAUUGACGUGGACUCGCUGAUGCGCUCCGUGUCGAGAGUGUUCAAGUUCAUCGAUAUGCCGACCGAGGGAAAGCCAACUAAGUCGACCAAGCCGUACAAAAACGGACAGCUGAGCAAGGUCAUGAUCAUCGAAAACUCCCACGUGAAAAAGGAUGACAUCUGGCCGUCCGGUGGACAGAUGACGGUGAAGGAUCUGACUGCGAAGUACACUGAGGGAGGGAAUGCCAUCCUCGAAAACAUCUCAUUCUCAAUCUCCCCUGGACAGAGGGUCGGGCUGCUGGGCCGCACUGGCUCGGGGAAGUCGACUCUUCUUUCGGCAUUUCUGCGCUUGCUCAAUACCGAGGGAGAAAUCCAGAUCGAUGGAGUGUCAUGGGACUCGAUCACCCUGCAGCAGUGGCGCAAGGCUUUUGGCGUCAUCCCGCAAAAGGUGUUCAUCUUCUCGGGCACUUUUAGAAAGAAUCUGGAUCCCUACGAACAGUGGUCAGAUCAAGAGAUUUGGAAAGUCGCAGACGAAGUGGGCCUCCGGUCCGUGAUUGAACAGUUUCCGGGAAAGCUCGACUUCGUGCUUGUGGACGGAGGAUGUGUGCUGAGCCACGGCCACAAACAGCUCAUGUGCCUGGCUCGGUCGGUCCUGUCGAAAGCAAAGAUCCUGCUGCUGGACGAACCGUCGGCACACCUCGAUCCAGUGACGUACCAGAUCAUCCGGCGGACCCUGAAGCAGGCCUUCGCAGACUGCACUGUCAUUUUGUGUGAACACAGAAUCGAAGCUAUGUUGGAGUGCCAGCAGUUCCUGGUCAUCGAAGAAAACAAAGUCCGCCAGUACGAUUCGAUUCAGAAGCUGCUGAACGAACGGAGCCUCUUCAGACAGGCGAUCAGCCCCAGCGAUCGGGUCAAGUUGUUCCCGCAUCGGAACAGCAGCAAGUGUAAGUCAAAGCCUCAGAUCGCUGCACUCAAAGAAGAGACUGAAGAAGAAGUGCAAGACACCAGACUCUGA(配列番号12)
配列番号13
AUGCAGCGCUCGCCUCUGGAGAAAGCCUCAGUCGUGUCAAAACUGUUCUUUAGCUGGACUCGCCCGAUUCUCCGGAAGGGUUAUAGACAGCGCUUGGAGCUCUCCGACAUCUACCAAAUCCCUUCCGUGGACUCCGCCGACAACCUGUCGGAGAAGCUCGAACGCGAGUGGGACCGGGAACUCGCGUCCAAAAAGAAUCCAAAACUCAUUAAUGCACUGCGCCGCUGCUUCUUCUGGCGCUUUAUGUUUUACGGUAUCUUUCUCUACCUGGGCGAGGUGACGAAAGCAGUGCAGCCGCUCCUGCUUGGCAGAAUUAUCGCCUCGUACGAUCCGGAUAACAAAGAAGAACGCUCAAUCGCUAUCUACCUCGGUAUCGGAUUGUGCCUGCUUUUCAUCGUGCGCACCCUGUUGCUGCACCCGGCGAUUUUCGGACUCCACCACAUCGGAAUGCAAAUGAGAAUUGCAAUGUUCUCAUUGAUCUACAAAAAGACCCUUAAACUGUCGUCCCGCGUCCUCGACAAGAUUUCAAUCGGCCAGCUGGUGUCGCUUCUUUCGAAUAAUCUUAACAAGUUCGAUGAAGGACUCGCGCUCGCCCAUUUCGUGUGGAUCGCACCACUUCAAGUCGCACUGCUCAUGGGACUGAUUUGGGAGUUGCUGCAGGCUUCCGCCUUUUGCGGCCUGGGAUUCCUGAUCGUCCUGGCUUUGUUCCAGGCUGGACUGGGCAGAAUGAUGAUGAAGUACCGGGACCAGCGGGCAGGAAAGAUCAGCGAAAGGCUCGUGAUCACUAGCGAAAUGAUCGAGAACAUCCAAUCCGUCAAGGCGUACUGCUGGGAAGAAGCGAUGGAGAAGAUGAUCGAAAAUCUUCGCCAGACCGAACUCAAACUCACUAGAAAGGCUGCCUACGUGCGCUACUUUAACAGCUCAGCAUUUUUCUUCUCCGGAUUUUUCGUGGUGUUCCUGUCGGUGCUGCCAUACGCCCUGAUCAAGGGGAUCAUUCUUCGCAAAAUCUUCACCACGAUCUCAUUCUGCAUUGUCCUCCGGAUGGCCGUGACGCGGCAGUUCCCUUGGGCAGUGCAAACUUGGUACGAUUCGCUGGGGGCCAUUAACAAGAUUCAAGAUUUUCUUCAAAAGCAGGAGUACAAAACCCUGGAGUACAAUCUGACCACUACGGAAGUCGUGAUGGAAAACGUGACUGCUUUUUGGGAGGAAGGCUUCGGCGAACUUUUUGAAAAGGCAAAGCAAAACAAUAACAACAGAAAGACGUCAAACGGCGAUGACUCGCUGUUCUUCUCCAAUUUCUCCCUGCUCGGCACCCCUGUGCUGAAGGACAUCAACUUCAAAAUUGAACGCGGACAGCUGCUGGCCGUGGCGGGAUCGACCGGGGCUGGGAAAACCUCGUUGUUGAUGGUGAUCAUGGGAGAACUCGAACCCUCGGAGGGAAAGAUUAAGCAUAGCGGACGGAUCAGCUUCUGUUCCCAGUUCUCGUGGAUCAUGCCGGGAACCAUUAAGGAAAACAUCAUCUUCGGCGUGUCCUACGACGAGUACCGGUAUAGGUCGGUGAUCAAGGCCUGCCAGUUGGAAGAGGACAUCUCCAAGUUCGCUGAGAAGGACAACAUCGUGCUCGGUGAAGGGGGCAUUACUCUGUCCGGUGGCCAGCGCGCGAGAAUUUCGCUGGCUCGCGCGGUGUACAAAGAUGCGGAUCUCUAUCUGCUGGAUUCGCCCUUCGGAUACCUCGAUGUCCUCACGGAGAAGGAGAUCUUCGAAUCGUGCGUGUGCAAGUUGAUGGCGAACAAGACUAGGAUCCUGGUCACUUCCAAGAUGGAGCACUUGAAGAAGGCCGAUAAGAUCUUGAUCCUCCAUGAAGGAUCGAGCUACUUUUACGGAACUUUCUCAGAGCUGCAGAACUUGCAGCCGGACUUCUCAAGCAAACUGAUGGGUUGCGACUCGUUCGACCAGUUUUCGGCAGAACGGCGGAACUCGAUCCUGACUGAGACUCUGCAUCGCUUUUCGCUGGAAGGCGAUGCCCCUGUGUCCUGGACUGAAACCAAGAAGCAAUCCUUCAAACAAACUGGAGAAUUCGGAGAAAAGCGGAAGAACUCCAUCCUUAACCCCAUCAAUAGCAUCCGGAAGUUCUCAAUCGUCCAAAAGACCCCGCUGCAGAUGAAUGGCAUCGAAGAAGAUAGCGACGAACCUCUUGAAAGACGGCUGUCCUUGGUGCCAGACUCAGAACAGGGAGAAGCUAUCCUGCCGCGGAUCUCCGUGAUCAGCACCGGACCGACUCUGCAGGCUCGCAGACGCCAGAGCGUGCUCAACCUGAUGACCCACUCCGUGAACCAGGGACAAAACAUCCAUAGAAAGACCACGGCCUCCACCAGAAAAGUCUCCCUGGCACCGCAAGCCAACCUGACUGAACUGGACAUCUACAGCAGAAGGCUCAGCCAAGAAACCGGACUGGAGAUUUCAGAAGAAAUCAACGAGGAAGAUCUUAAAGAGUGCUUCUUCGACGACAUGGAAUCGAUCCCAGCCGUGACCACUUGGAAUACCUAUCUGAGAUACAUCACCGUGCACAAAUCCCUGAUCUUCGUGCUGAUCUGGUGCCUGGUGAUCUUCCUGGCUGAGGUGGCCGCCUCACUGGUGGUGCUUUGGUUGCUGGGGAAUACGCCGCUCCAAGACAAGGGAAACUCCACGCACUCCAGAAACAACUCGUACGCCGUGAUCAUCACGUCGACUUCGUCGUACUACGUGUUCUACAUCUACGUCGGUGUGGCAGACACUCUCUUGGCGAUGGGCUUUUUCCGGGGACUGCCACUGGUCCACACCCUGAUCACCGUGUCCAAAAUCUUGCACCACAAGAUGCUCCACAGCGUGCUGCAAGCCCCGAUGAGCACCCUGAAUACCCUCAAAGCGGGAGGCAUCCUCAACAGAUUCAGCAAGGACAUCGCCAUCCUCGACGACCUGUUGCCCCUGACCAUCUUCGAUUUCAUCCAGCUUCUUCUCAUCGUGAUCGGGGCAAUCGCUGUCGUGGCGGUGCUGCAGCCGUACAUCUUCGUGGCGACUGUGCCAGUGAUCGUCGCCUUUAUCAUGCUGCGGGCCUACUUUCUCCAAACUUCCCAACAGCUGAAACAACUGGAGUCGGAGGGCCGCAGCCCUAUCUUCACCCAUCUGGUGACCAGCCUCAAAGGACUGUGGACUCUGAGGGCUUUCGGGAGGCAGCCAUACUUCGAGACUCUCUUUCACAAGGCCCUGAAUCUCCAUACGGCAAAUUGGUUUUUGUAUUUGAGUACCCUCCGAUGGUUUCAGAUGCGCAUUGAGAUGAUUUUUGUGAUCUUCUUUAUCGCGGUGACUUUUAUCUCCAUCUUGACCACGGGAGAGGGCGAGGGACGGGUCGGUAUUAUCCUGACACUCGCCAUGAACAUUAUGAGCACUUUGCAGUGGGCAGUGAACAGCUCGAUUGAUGUGGAUAGCCUGAUGAGGUCCGUUUCGAGGGUCUUUAAGUUCAUCGACAUGCCGACGGAGGGAAAGCCCACAAAAAGUACGAAACCCUAUAAGAAUGGGCAAUUGAGUAAGGUAAUGAUCAUCGAGAACAGUCACGUGAAGAAGGAUGACAUCUGGCCUAGCGGGGGUCAGAUGACCGUGAAGGACCUGACGGCAAAAUACACCGAGGGAGGGAACGCAAUCCUUGAAAACAUCUCGUUCAGCAUUAGCCCCGGUCAGCGUGUGGGGUUGCUCGGGAGGACCGGGUCAGGAAAAUCGACGUUGCUGUCGGCCUUCUUGAGACUUCUGAAUACAGAGGGUGAGAUCCAGAUCGACGGCGUUUCGUGGGAUAGCAUCACCUUGCAGCAGUGGCGCAAGGCGUUCGGAGUCAUUCCCCAAAAGGUGUUCAUCUUUUCGGGAACCUUCCGCAAGAAUCUGGAUCCGUACGAACAGUGGAGCGACCAAGAGAUUUGGAAAGUGGCAGAUGAAGUGGGAUUGCGGAGCGUCAUCGAACAGUUUCCGGGAAAGCUCGAUUUCGUCCUUGUGGACGGUGGAUGUGUGCUGUCGCACGGCCAUAAGCAGCUGAUGUGUCUCGCCCGCUCGGUGCUGUCAAAGGCGAAGAUCCUCUUGCUGGAUGAGCCAUCAGCCCAUCUGGACCCGGUGACGUACCAGAUCAUUAGACGGACGCUGAAACAGGCAUUCGCGGACUGCACUGUGAUCCUCUGUGAACAUCGGAUCGAGGCCAUGCUGGAGUGUCAACAAUUCUUGGUCAUCGAAGAGAACAAAGUGCGGCAGUACGACAGCAUCCAAAAGCUGCUGAACGAGAGGUCCCUCUUCCGCCAGGCCAUCUCCCCAUCCGACCGGGUCAAGCUGUUCCCUCACCGCAACAGCUCAAAGUGCAAAUCCAAACCCCAGAUCGCAGCGCUGAAAGAAGAAACUGAAGAAGAAGUGCAAGACACUAGACUGUGA(配列番号13)
配列番号14
AUGCAAAGGUCCCCAUUGGAGAAGGCCUCAGUGGUGUCGAAGCUGUUCUUCUCGUGGACCAGGCCUAUCCUCCGGAAGGGAUACAGACAGCGGCUGGAACUGUCCGAUAUCUACCAGAUCCCCAGCGUGGACAGCGCCGAUAAUCUCAGCGAAAAGCUGGAACGGGAAUGGGACCGCGAACUCGCUUCGAAGAAGAACCCGAAGCUGAUUAAUGCUCUGCGGAGAUGUUUCUUUUGGCGGUUCAUGUUUUACGGAAUCUUUCUGUACUUGGGAGAGGUCACGAAGGCUGUGCAGCCUCUGCUGCUGGGACGGAUUAUCGCGUCGUAUGACCCCGACAAUAAGGAAGAACGCAGCAUCGCAAUCUACCUGGGCAUCGGAUUGUGCCUGCUGUUCAUCGUGAGAACUCUCCUGCUGCAUCCAGCCAUCUUCGGACUCCACCACAUUGGAAUGCAGAUGAGAAUCGCAAUGUUCUCCCUGAUCUACAAGAAAACGCUCAAGCUCAGCAGCCGCGUGCUCGAUAAGAUCAGCAUCGGUCAAUUGGUGUCCCUGCUGUCGAAUAACCUCAACAAGUUCGACGAAGGGUUGGCCCUCGCUCACUUCGUGUGGAUCGCACCUCUGCAAGUGGCCCUGCUGAUGGGACUGAUUUGGGAGCUGCUGCAGGCUUCCGCUUUCUGCGGCCUGGGAUUUCUUAUCGUGCUUGCUCUGUUCCAGGCGGGACUGGGACGCAUGAUGAUGAAGUACCGGGACCAACGGGCUGGAAAGAUCAGCGAACGGCUGGUGAUCACUUCCGAAAUGAUUGAGAAUAUCCAGUCAGUCAAGGCGUACUGCUGGGAAGAGGCUAUGGAAAAGAUGAUUGAAAAUCUGAGACAAACCGAGCUGAAGCUGACUCGGAAAGCGGCCUACGUCAGAUACUUCAAUAGCUCAGCUUUCUUUUUCUCGGGGUUUUUCGUCGUGUUCCUGUCGGUGCUUCCCUAUGCCCUGAUUAAGGGCAUCAUUCUGCGCAAGAUCUUCACUACGAUCUCAUUCUGCAUCGUGCUGCGCAUGGCUGUGACCAGACAAUUCCCGUGGGCCGUGCAAACCUGGUACGAUUCACUGGGAGCCAUCAACAAGAUCCAAGACUUUCUCCAAAAACAGGAGUAUAAGACCCUGGAGUACAACCUGACUACUACCGAGGUGGUGAUGGAGAACGUGACUGCGUUUUGGGAAGAAGGGUUCGGCGAACUGUUUGAAAAGGCCAAGCAGAACAAUAACAACAGAAAGACUUCAAACGGAGAUGACUCGCUGUUCUUUUCGAACUUCAGCCUGCUGGGUACCCCAGUGUUGAAAGAUAUCAACUUCAAGAUUGAGAGAGGACAGCUGCUGGCUGUGGCGGGAUCCACCGGAGCAGGAAAAACUUCACUCCUGAUGGUGAUCAUGGGAGAACUCGAACCGUCAGAGGGGAAGAUUAAACACUCGGGAAGAAUCUCAUUUUGCUCCCAAUUUUCAUGGAUUAUGCCGGGAACCAUUAAAGAAAACAUUAUCUUCGGCGUGUCCUACGACGAGUACCGCUACAGAUCGGUGAUCAAAGCAUGCCAGCUGGAAGAGGACAUCUCGAAAUUCGCUGAAAAAGACAAUAUCGUGCUCGGGGAAGGCGGCAUCACCCUCAGCGGAGGACAACGGGCACGGAUUUCGCUCGCACGCGCAGUCUACAAAGACGCCGAUCUCUACCUCUUGGACAGCCCAUUCGGGUAUCUGGACGUGCUCACCGAGAAAGAGAUCUUCGAAAGCUGCGUCUGCAAGCUCAUGGCCAACAAGACCCGCAUCCUCGUGACGUCGAAGAUGGAACAUCUUAAGAAGGCUGACAAGAUUCUCAUUCUCCAUGAAGGGAGCUCAUACUUCUACGGCACCUUUUCCGAGCUCCAGAAUCUGCAACCGGACUUCUCGUCCAAGCUGAUGGGCUGCGAUUCGUUUGAUCAGUUCUCCGCCGAGCGGAGAAACAGCAUUCUGACGGAAACCCUGCACCGGUUCUCGCUGGAAGGCGAUGCACCGGUGUCGUGGACCGAAACUAAGAAGCAAUCGUUCAAGCAGACGGGAGAGUUUGGAGAGAAGCGGAAAAACUCCAUCCUCAACCCGAUCAACAGCAUCCGGAAGUUCAGCAUCGUGCAAAAGACCCCGCUCCAGAUGAAUGGCAUUGAAGAGGACUCCGACGAACCUUUGGAACGCAGACUGAGCCUCGUGCCGGAUUCAGAACAGGGAGAAGCCAUUCUGCCACGGAUCUCCGUGAUCAGCACUGGGCCAACUCUCCAAGCACGGCGGAGGCAGUCCGUGCUGAAUCUUAUGACGCACAGCGUGAACCAAGGGCAGAACAUCCAUAGAAAAACGACCGCUUCGACCAGGAAAGUCUCCCUCGCCCCACAAGCUAACCUCACGGAACUGGAUAUCUACUCCCGCAGACUGUCGCAAGAGACUGGCCUUGAGAUCUCCGAAGAGAUUAACGAAGAAGAUCUCAAAGAAUGUUUCUUCGAUGAUAUGGAAUCAAUCCCGGCAGUGACCACUUGGAACACCUACUUGCGCUAUAUCACUGUGCACAAAAGCCUUAUCUUCGUCCUCAUCUGGUGCCUCGUCAUCUUCCUGGCUGAGGUCGCAGCCUCGCUGGUCGUGCUCUGGUUGCUCGGAAACACUCCGCUGCAGGAUAAGGGGAAUUCGACUCACUCGCGGAACAAUUCGUACGCUGUCAUUAUCACCUCGACGUCGUCAUACUACGUGUUUUACAUCUACGUGGGAGUGGCUGACACUCUGUUGGCUAUGGGGUUCUUUCGCGGCCUGCCACUGGUCCAUACUCUCAUUACUGUGUCCAAAAUCCUUCAUCACAAGAUGUUGCAUUCAGUGCUGCAAGCACCGAUGUCCACCCUCAAUACCCUUAAGGCUGGCGGGAUUCUCAACCGCUUCUCGAAAGACAUCGCCAUCCUCGAUGAUCUUCUGCCUCUCACCAUCUUUGAUUUCAUCCAGCUGCUCCUGAUCGUGAUCGGAGCGAUUGCCGUGGUGGCAGUGUUGCAGCCGUACAUCUUUGUCGCAACUGUGCCGGUCAUCGUCGCCUUCAUCAUGCUGCGCGCCUACUUCUUGCAAACGUCACAGCAACUGAAGCAGCUUGAAUCCGAGGGAAGAUCACCUAUCUUCACCCACCUCGUGACUUCGCUGAAGGGGCUGUGGACGCUGCGCGCAUUUGGAAGGCAACCGUACUUCGAGACUUUGUUCCACAAGGCGCUCAAUCUUCACACUGCCAAUUGGUUCUUGUACCUGUCAACGCUGAGAUGGUUUCAGAUGCGGAUCGAAAUGAUCUUCGUGAUCUUCUUUAUCGCGGUGACUUUCAUCUCGAUCCUGACUACCGGAGAGGGAGAAGGACGGGUGGGUAUUAUCCUCACUCUGGCGAUGAACAUCAUGUCGACGCUUCAGUGGGCGGUGAAUAGCUCAAUCGAUGUCGACUCGCUGAUGCGCUCCGUGAGCCGGGUGUUUAAGUUCAUCGACAUGCCAACUGAAGGGAAGCCGACCAAGUCGACCAAACCGUACAAAAACGGACAGCUCUCCAAGGUGAUGAUUAUCGAGAAUUCCCACGUGAAAAAGGACGACAUCUGGCCAUCCGGUGGACAGAUGACCGUGAAGGACCUGACCGCGAAGUACACUGAGGGAGGCAACGCAAUCCUUGAGAACAUCAGCUUCUCCAUCUCGCCCGGUCAGAGGGUGGGCCUUCUUGGCCGGACCGGAUCGGGAAAGUCCACUCUUCUGUCGGCCUUUCUUCGCCUCUUGAAUACUGAAGGGGAAAUCCAGAUCGACGGAGUGUCGUGGGAUAGCAUCACUCUGCAGCAGUGGCGGAAAGCGUUUGGAGUAAUCCCCCAAAAGGUCUUUAUCUUUAGCGGAACCUUCCGAAAGAAUCUCGAUCCUUAUGAACAGUGGUCAGAUCAAGAGAUUUGGAAAGUCGCGGACGAGGUUGGCCUUCGGAGUGUAAUCGAGCAGUUUCCGGGAAAACUCGACUUUGUCCUUGUAGAUGGGGGAUGCGUCCUGUCGCAUGGGCACAAGCAGCUCAUGUGCCUGGCGCGAUCCGUCCUCUCUAAAGCGAAAAUUCUUCUCUUGGAUGAACCUUCGGCCCAUCUGGACCCGGUAACGUAUCAGAUCAUCAGAAGGACACUUAAGCAGGCGUUUGCCGACUGCACGGUGAUUCUCUGUGAGCAUCGUAUCGAGGCCAUGCUCGAAUGCCAGCAAUUUCUUGUCAUCGAAGAGAAUAAGGUCCGCCAGUACGACUCCAUCCAGAAGCUGCUUAAUGAGAGAUCAUUGUUCCGGCAGGCGAUUUCACCAUCCGAUAGGGUGAAACUUUUUCCACACAGAAAUUCGUCGAAGUGCAAGUCCAAACCGCAGAUCGCGGCCUUGAAAGAAGAGACUGAAGAAGAAGUUCAAGACACGCGUCUUUAA(配列番号14)
配列番号15
AUGCAGCGGUCCCCGCUCGAAAAGGCCAGUGUCGUGUCCAAACUCUUCUUCUCAUGGACUCGGCCUAUCCUUAGAAAGGGGUAUCGGCAGAGGCUUGAGUUGUCUGACAUCUACCAGAUCCCCUCGGUAGAUUCGGCGGAUAACCUCUCGGAGAAGCUCGAACGGGAAUGGGACCGCGAACUCGCGUCUAAGAAAAACCCGAAGCUCAUCAACGCACUGAGAAGGUGCUUCUUCUGGCGGUUCAUGUUCUACGGUAUCUUCUUGUAUCUCGGGGAGGUCACAAAAGCAGUCCAACCCCUGUUGUUGGGUCGCAUUAUCGCCUCGUACGACCCCGAUAACAAAGAAGAACGGAGCAUCGCGAUCUACCUCGGGAUCGGACUGUGUUUGCUUUUCAUCGUCAGAACACUUUUGUUGCAUCCAGCAAUCUUCGGCCUCCAUCACAUCGGUAUGCAGAUGCGAAUCGCUAUGUUUAGCUUGAUCUACAAAAAGACACUGAAACUCUCGUCGCGGGUGUUGGAUAAGAUUUCCAUCGGUCAGUUGGUGUCCCUGCUUAGUAAUAACCUCAACAAAUUCGAUGAGGGACUGGCGCUGGCACAUUUCGUGUGGAUUGCCCCGUUGCAAGUCGCCCUUUUGAUGGGCCUUAUUUGGGAGCUGUUGCAGGCAUCUGCCUUUUGUGGCCUGGGAUUUCUGAUUGUGUUGGCAUUGUUUCAGGCUGGGCUUGGGCGGAUGAUGAUGAAGUAUCGCGACCAGAGAGCGGGUAAAAUCUCGGAAAGACUCGUCAUCACUUCGGAAAUGAUCGAAAACAUCCAGUCGGUCAAAGCCUAUUGCUGGGAAGAAGCUAUGGAGAAGAUGAUUGAAAACCUCCGCCAAACUGAGCUGAAACUGACCCGCAAGGCGGCGUAUGUCCGGUAUUUCAAUUCGUCAGCGUUCUUCUUUUCCGGGUUCUUCGUUGUCUUUCUCUCGGUUUUGCCUUAUGCCUUGAUUAAGGGGAUUAUCCUCCGCAAGAUUUUCACCACGAUUUCGUUCUGCAUUGUAUUGCGCAUGGCAGUGACACGGCAAUUUCCGUGGGCCGUGCAGACAUGGUAUGACUCGCUUGGAGCGAUCAACAAAAUCCAAGACUUCUUGCAAAAGCAAGAGUACAAGACCCUGGAGUACAAUCUUACUACUACGGAGGUAGUAAUGGAGAAUGUGACGGCUUUUUGGGAAGAGGGUUUUGGAGAACUGUUUGAGAAAGCAAAGCAGAAUAACAACAACCGCAAGACCUCAAAUGGGGACGAUUCCCUGUUUUUCUCGAACUUCUCCCUGCUCGGAACACCCGUGUUGAAGGACAUCAAUUUCAAGAUUGAGAGGGGACAGCUUCUCGCGGUAGCGGGAAGCACUGGUGCGGGAAAAACUAGCCUCUUGAUGGUGAUUAUGGGGGAGCUUGAGCCCAGCGAGGGGAAGAUUAAACACUCCGGGCGUAUCUCAUUCUGUAGCCAGUUUUCAUGGAUCAUGCCCGGAACCAUUAAAGAGAACAUCAUUUUCGGAGUAUCCUAUGAUGAGUACCGAUACAGAUCGGUCAUUAAGGCGUGCCAGUUGGAAGAGGACAUUUCUAAGUUCGCCGAGAAGGAUAACAUCGUCUUGGGAGAAGGGGGUAUUACAUUGUCGGGAGGGCAGCGAGCGCGGAUCAGCCUCGCGAGAGCGGUAUACAAAGAUGCAGAUUUGUAUCUGCUUGAUUCACCGUUUGGAUACCUCGACGUAUUGACAGAAAAAGAAAUCUUCGAGUCGUGCGUGUGUAAACUUAUGGCUAAUAAGACGAGAAUCCUGGUGACAUCAAAAAUGGAACACCUUAAGAAGGCGGACAAGAUCCUGAUCCUCCACGAAGGAUCGUCCUACUUUUACGGCACUUUCUCAGAGUUGCAAAACUUGCAGCCGGACUUCUCAAGCAAACUCAUGGGGUGUGACUCAUUCGACCAGUUCAGCGCGGAACGGCGGAACUCGAUCUUGACGGAAACGCUGCACCGAUUCUCGCUUGAGGGUGAUGCCCCGGUAUCGUGGACCGAGACAAAGAAGCAGUCGUUUAAGCAGACAGGAGAAUUUGGUGAGAAAAGAAAGAACAGUAUCUUGAAUCCUAUUAACUCAAUUCGCAAGUUCUCAAUCGUCCAGAAAACUCCACUGCAGAUGAAUGGAAUUGAAGAGGAUUCGGACGAACCCCUGGAGCGCAGGCUUAGCCUCGUGCCGGAUUCAGAGCAAGGGGAGGCCAUUCUUCCCCGGAUUUCGGUGAUUUCAACCGGACCUACACUUCAGGCGAGGCGAAGGCAAUCCGUGCUCAACCUCAUGACGCAUUCGGUAAACCAGGGGCAAAACAUUCACCGCAAAACGACGGCCUCAACGAGAAAAGUGUCACUUGCACCCCAGGCGAAUUUGACUGAACUCGACAUCUACAGCCGUAGGCUUUCGCAAGAAACCGGACUUGAGAUCAGCGAAGAAAUCAAUGAAGAAGAUUUGAAAGAGUGUUUCUUUGAUGACAUGGAAUCAAUCCCAGCGGUGACAACGUGGAACACAUACUUGCGUUACAUCACGGUGCACAAGUCCUUGAUUUUCGUCCUCAUCUGGUGUCUCGUGAUCUUUCUCGCUGAGGUCGCAGCGUCACUUGUGGUCCUCUGGCUGCUUGGUAAUACGCCCUUGCAAGACAAAGGCAAUUCUACACACUCAAGAAACAAUUCCUAUGCCGUGAUUAUCACUUCUACAAGCUCGUAUUACGUGUUUUACAUCUACGUAGGAGUGGCCGACACUCUGCUCGCGAUGGGUUUCUUCCGAGGACUCCCACUCGUUCACACGCUUAUCACUGUCUCCAAGAUUCUCCACCAUAAGAUGCUUCAUAGCGUACUGCAGGCUCCCAUGUCCACCUUGAAUACGCUCAAGGCGGGAGGUAUUUUGAAUCGCUUCUCAAAAGAUAUUGCAAUUUUGGAUGACCUUCUGCCCCUGACGAUCUUCGACUUCAUCCAGUUGUUGCUGAUCGUGAUUGGGGCUAUUGCAGUAGUCGCUGUCCUCCAGCCUUACAUUUUUGUCGCGACCGUUCCGGUGAUCGUGGCGUUUAUCAUGCUGCGGGCCUAUUUCUUGCAGACGUCACAGCAGCUUAAGCAACUGGAGUCUGAAGGGAGGUCGCCUAUCUUUACGCAUCUUGUGACCAGUUUGAAGGGAUUGUGGACGUUGCGCGCCUUUGGCAGGCAGCCCUACUUUGAAACACUGUUCCACAAAGCGCUGAAUCUCCAUACGGCAAAUUGGUUUUUGUAUUUGAGUACCCUCCGAUGGUUUCAGAUGCGCAUUGAGAUGAUUUUUGUGAUCUUCUUUAUCGCGGUGACUUUUAUCUCCAUCUUGACCACGGGAGAGGGCGAGGGACGGGUCGGUAUUAUCCUGACACUCGCCAUGAACAUUAUGAGCACUUUGCAGUGGGCAGUGAACAGCUCGAUUGAUGUGGAUAGCCUGAUGAGGUCCGUUUCGAGGGUCUUUAAGUUCAUCGACAUGCCGACGGAGGGAAAGCCCACAAAAAGUACGAAACCCUAUAAGAAUGGGCAAUUGAGUAAGGUAAUGAUCAUCGAGAACAGUCACGUGAAGAAGGAUGACAUCUGGCCUAGCGGGGGUCAGAUGACCGUGAAGGACCUGACGGCAAAAUACACCGAGGGAGGGAACGCAAUCCUUGAAAACAUCUCGUUCAGCAUUAGCCCCGGUCAGCGUGUGGGGUUGCUCGGGAGGACCGGGUCAGGAAAAUCGACGUUGCUGUCGGCCUUCUUGAGACUUCUGAAUACAGAGGGUGAGAUCCAGAUCGACGGCGUUUCGUGGGAUAGCAUCACCUUGCAGCAGUGGCGGAAAGCGUUUGGAGUAAUCCCCCAAAAGGUCUUUAUCUUUAGCGGAACCUUCCGAAAGAAUCUCGAUCCUUAUGAACAGUGGUCAGAUCAAGAGAUUUGGAAAGUCGCGGACGAGGUUGGCCUUCGGAGUGUAAUCGAGCAGUUUCCGGGAAAACUCGACUUUGUCCUUGUAGAUGGGGGAUGCGUCCUGUCGCAUGGGCACAAGCAGCUCAUGUGCCUGGCGCGAUCCGUCCUCUCUAAAGCGAAAAUUCUUCUCUUGGAUGAACCUUCGGCCCAUCUGGACCCGGUAACGUAUCAGAUCAUCAGAAGGACACUUAAGCAGGCGUUUGCCGACUGCACGGUGAUUCUCUGUGAGCAUCGUAUCGAGGCCAUGCUCGAAUGCCAGCAAUUUCUUGUCAUCGAAGAGAAUAAGGUCCGCCAGUACGACUCCAUCCAGAAGCUGCUUAAUGAGAGAUCAUUGUUCCGGCAGGCGAUUUCACCAUCCGAUAGGGUGAAACUUUUUCCACACAGAAAUUCGUCGAAGUGCAAGUCCAAACCGCAGAUCGCGGCCUUGAAAGAAGAGACUGAAGAAGAAGUUCAAGACACGCGUCUUCACCAUCACCAUCACCAUCACCAUCACCAUUAA(配列番号15)
配列番号16
AUGGCCACUGGAUCAAGAACCUCACUGCUGCUCGCUUUUGGACUGCUUUGCCUGCCCUGGUUGCAAGAAGGAUCGGCUUUCCCGACCAUCCCACUCUCCAUGCAGCGGUCCCCGCUCGAAAAGGCCAGUGUCGUGUCCAAACUCUUCUUCUCAUGGACUCGGCCUAUCCUUAGAAAGGGGUAUCGGCAGAGGCUUGAGUUGUCUGACAUCUACCAGAUCCCCUCGGUAGAUUCGGCGGAUAACCUCUCGGAGAAGCUCGAACGGGAAUGGGACCGCGAACUCGCGUCUAAGAAAAACCCGAAGCUCAUCAACGCACUGAGAAGGUGCUUCUUCUGGCGGUUCAUGUUCUACGGUAUCUUCUUGUAUCUCGGGGAGGUCACAAAAGCAGUCCAACCCCUGUUGUUGGGUCGCAUUAUCGCCUCGUACGACCCCGAUAACAAAGAAGAACGGAGCAUCGCGAUCUACCUCGGGAUCGGACUGUGUUUGCUUUUCAUCGUCAGAACACUUUUGUUGCAUCCAGCAAUCUUCGGCCUCCAUCACAUCGGUAUGCAGAUGCGAAUCGCUAUGUUUAGCUUGAUCUACAAAAAGACACUGAAACUCUCGUCGCGGGUGUUGGAUAAGAUUUCCAUCGGUCAGUUGGUGUCCCUGCUUAGUAAUAACCUCAACAAAUUCGAUGAGGGACUGGCGCUGGCACAUUUCGUGUGGAUUGCCCCGUUGCAAGUCGCCCUUUUGAUGGGCCUUAUUUGGGAGCUGUUGCAGGCAUCUGCCUUUUGUGGCCUGGGAUUUCUGAUUGUGUUGGCAUUGUUUCAGGCUGGGCUUGGGCGGAUGAUGAUGAAGUAUCGCGACCAGAGAGCGGGUAAAAUCUCGGAAAGACUCGUCAUCACUUCGGAAAUGAUCGAAAACAUCCAGUCGGUCAAAGCCUAUUGCUGGGAAGAAGCUAUGGAGAAGAUGAUUGAAAACCUCCGCCAAACUGAGCUGAAACUGACCCGCAAGGCGGCGUAUGUCCGGUAUUUCAAUUCGUCAGCGUUCUUCUUUUCCGGGUUCUUCGUUGUCUUUCUCUCGGUUUUGCCUUAUGCCUUGAUUAAGGGGAUUAUCCUCCGCAAGAUUUUCACCACGAUUUCGUUCUGCAUUGUAUUGCGCAUGGCAGUGACACGGCAAUUUCCGUGGGCCGUGCAGACAUGGUAUGACUCGCUUGGAGCGAUCAACAAAAUCCAAGACUUCUUGCAAAAGCAAGAGUACAAGACCCUGGAGUACAAUCUUACUACUACGGAGGUAGUAAUGGAGAAUGUGACGGCUUUUUGGGAAGAGGGUUUUGGAGAACUGUUUGAGAAAGCAAAGCAGAAUAACAACAACCGCAAGACCUCAAAUGGGGACGAUUCCCUGUUUUUCUCGAACUUCUCCCUGCUCGGAACACCCGUGUUGAAGGACAUCAAUUUCAAGAUUGAGAGGGGACAGCUUCUCGCGGUAGCGGGAAGCACUGGUGCGGGAAAAACUAGCCUCUUGAUGGUGAUUAUGGGGGAGCUUGAGCCCAGCGAGGGGAAGAUUAAACACUCCGGGCGUAUCUCAUUCUGUAGCCAGUUUUCAUGGAUCAUGCCCGGAACCAUUAAAGAGAACAUCAUUUUCGGAGUAUCCUAUGAUGAGUACCGAUACAGAUCGGUCAUUAAGGCGUGCCAGUUGGAAGAGGACAUUUCUAAGUUCGCCGAGAAGGAUAACAUCGUCUUGGGAGAAGGGGGUAUUACAUUGUCGGGAGGGCAGCGAGCGCGGAUCAGCCUCGCGAGAGCGGUAUACAAAGAUGCAGAUUUGUAUCUGCUUGAUUCACCGUUUGGAUACCUCGACGUAUUGACAGAAAAAGAAAUCUUCGAGUCGUGCGUGUGUAAACUUAUGGCUAAUAAGACGAGAAUCCUGGUGACAUCAAAAAUGGAACACCUUAAGAAGGCGGACAAGAUCCUGAUCCUCCACGAAGGAUCGUCCUACUUUUACGGCACUUUCUCAGAGUUGCAAAACUUGCAGCCGGACUUCUCAAGCAAACUCAUGGGGUGUGACUCAUUCGACCAGUUCAGCGCGGAACGGCGGAACUCGAUCUUGACGGAAACGCUGCACCGAUUCUCGCUUGAGGGUGAUGCCCCGGUAUCGUGGACCGAGACAAAGAAGCAGUCGUUUAAGCAGACAGGAGAAUUUGGUGAGAAAAGAAAGAACAGUAUCUUGAAUCCUAUUAACUCAAUUCGCAAGUUCUCAAUCGUCCAGAAAACUCCACUGCAGAUGAAUGGAAUUGAAGAGGAUUCGGACGAACCCCUGGAGCGCAGGCUUAGCCUCGUGCCGGAUUCAGAGCAAGGGGAGGCCAUUCUUCCCCGGAUUUCGGUGAUUUCAACCGGACCUACACUUCAGGCGAGGCGAAGGCAAUCCGUGCUCAACCUCAUGACGCAUUCGGUAAACCAGGGGCAAAACAUUCACCGCAAAACGACGGCCUCAACGAGAAAAGUGUCACUUGCACCCCAGGCGAAUUUGACUGAACUCGACAUCUACAGCCGUAGGCUUUCGCAAGAAACCGGACUUGAGAUCAGCGAAGAAAUCAAUGAAGAAGAUUUGAAAGAGUGUUUCUUUGAUGACAUGGAAUCAAUCCCAGCGGUGACAACGUGGAACACAUACUUGCGUUACAUCACGGUGCACAAGUCCUUGAUUUUCGUCCUCAUCUGGUGUCUCGUGAUCUUUCUCGCUGAGGUCGCAGCGUCACUUGUGGUCCUCUGGCUGCUUGGUAAUACGCCCUUGCAAGACAAAGGCAAUUCUACACACUCAAGAAACAAUUCCUAUGCCGUGAUUAUCACUUCUACAAGCUCGUAUUACGUGUUUUACAUCUACGUAGGAGUGGCCGACACUCUGCUCGCGAUGGGUUUCUUCCGAGGACUCCCACUCGUUCACACGCUUAUCACUGUCUCCAAGAUUCUCCACCAUAAGAUGCUUCAUAGCGUACUGCAGGCUCCCAUGUCCACCUUGAAUACGCUCAAGGCGGGAGGUAUUUUGAAUCGCUUCUCAAAAGAUAUUGCAAUUUUGGAUGACCUUCUGCCCCUGACGAUCUUCGACUUCAUCCAGUUGUUGCUGAUCGUGAUUGGGGCUAUUGCAGUAGUCGCUGUCCUCCAGCCUUACAUUUUUGUCGCGACCGUUCCGGUGAUCGUGGCGUUUAUCAUGCUGCGGGCCUAUUUCUUGCAGACGUCACAGCAGCUUAAGCAACUGGAGUCUGAAGGGAGGUCGCCUAUCUUUACGCAUCUUGUGACCAGUUUGAAGGGAUUGUGGACGUUGCGCGCCUUUGGCAGGCAGCCCUACUUUGAAACACUGUUCCACAAAGCGCUGAAUCUCCAUACGGCAAAUUGGUUUUUGUAUUUGAGUACCCUCCGAUGGUUUCAGAUGCGCAUUGAGAUGAUUUUUGUGAUCUUCUUUAUCGCGGUGACUUUUAUCUCCAUCUUGACCACGGGAGAGGGCGAGGGACGGGUCGGUAUUAUCCUGACACUCGCCAUGAACAUUAUGAGCACUUUGCAGUGGGCAGUGAACAGCUCGAUUGAUGUGGAUAGCCUGAUGAGGUCCGUUUCGAGGGUCUUUAAGUUCAUCGACAUGCCGACGGAGGGAAAGCCCACAAAAAGUACGAAACCCUAUAAGAAUGGGCAAUUGAGUAAGGUAAUGAUCAUCGAGAACAGUCACGUGAAGAAGGAUGACAUCUGGCCUAGCGGGGGUCAGAUGACCGUGAAGGACCUGACGGCAAAAUACACCGAGGGAGGGAACGCAAUCCUUGAAAACAUCUCGUUCAGCAUUAGCCCCGGUCAGCGUGUGGGGUUGCUCGGGAGGACCGGGUCAGGAAAAUCGACGUUGCUGUCGGCCUUCUUGAGACUUCUGAAUACAGAGGGUGAGAUCCAGAUCGACGGCGUUUCGUGGGAUAGCAUCACCUUGCAGCAGUGGCGGAAAGCGUUUGGAGUAAUCCCCCAAAAGGUCUUUAUCUUUAGCGGAACCUUCCGAAAGAAUCUCGAUCCUUAUGAACAGUGGUCAGAUCAAGAGAUUUGGAAAGUCGCGGACGAGGUUGGCCUUCGGAGUGUAAUCGAGCAGUUUCCGGGAAAACUCGACUUUGUCCUUGUAGAUGGGGGAUGCGUCCUGUCGCAUGGGCACAAGCAGCUCAUGUGCCUGGCGCGAUCCGUCCUCUCUAAAGCGAAAAUUCUUCUCUUGGAUGAACCUUCGGCCCAUCUGGACCCGGUAACGUAUCAGAUCAUCAGAAGGACACUUAAGCAGGCGUUUGCCGACUGCACGGUGAUUCUCUGUGAGCAUCGUAUCGAGGCCAUGCUCGAAUGCCAGCAAUUUCUUGUCAUCGAAGAGAAUAAGGUCCGCCAGUACGACUCCAUCCAGAAGCUGCUUAAUGAGAGAUCAUUGUUCCGGCAGGCGAUUUCACCAUCCGAUAGGGUGAAACUUUUUCCACACAGAAAUUCGUCGAAGUGCAAGUCCAAACCGCAGAUCGCGGCCUUGAAAGAAGAGACUGAAGAAGAAGUUCAAGACACGCGUCUUUAA(配列番号16)
配列番号17
AUGCAGCGGUCCCCGCUCGAAAAGGCCAGUGUCGUGUCCAAACUCUUCUUCUCAUGGACUCGGCCUAUCCUUAGAAAGGGGUAUCGGCAGAGGCUUGAGUUGUCUGACAUCUACCAGAUCCCCUCGGUAGAUUCGGCGGAUAACCUCUCGGAGAAGCUCGAACGGGAAUGGGACCGCGAACUCGCGUCUAAGAAAAACCCGAAGCUCAUCAACGCACUGAGAAGGUGCUUCUUCUGGCGGUUCAUGUUCUACGGUAUCUUCUUGUAUCUCGGGGAGGUCACAAAAGCAGUCCAACCCCUGUUGUUGGGUCGCAUUAUCGCCUCGUACGACCCCGAUAACAAAGAAGAACGGAGCAUCGCGAUCUACCUCGGGAUCGGACUGUGUUUGCUUUUCAUCGUCAGAACACUUUUGUUGCAUCCAGCAAUCUUCGGCCUCCAUCACAUCGGUAUGCAGAUGCGAAUCGCUAUGUUUAGCUUGAUCUACAAAAAGACACUGAAACUCUCGUCGCGGGUGUUGGAUAAGAUUUCCAUCGGUCAGUUGGUGUCCCUGCUUAGUAAUAACCUCAACAAAUUCGAUGAGGGACUGGCGCUGGCACAUUUCGUGUGGAUUGCCCCGUUGCAAGUCGCCCUUUUGAUGGGCCUUAUUUGGGAGCUGUUGCAGGCAUCUGCCUUUUGUGGCCUGGGAUUUCUGAUUGUGUUGGCAUUGUUUCAGGCUGGGCUUGGGCGGAUGAUGAUGAAGUAUCGCGACCAGAGAGCGGGUAAAAUCUCGGAAAGACUCGUCAUCACUUCGGAAAUGAUCGAAAACAUCCAGUCGGUCAAAGCCUAUUGCUGGGAAGAAGCUAUGGAGAAGAUGAUUGAAAACCUCCGCCAAACUGAGCUGAAACUGACCCGCAAGGCGGCGUAUGUCCGGUAUUUCAAUUCGUCAGCGUUCUUCUUUUCCGGGUUCUUCGUUGUCUUUCUCUCGGUUUUGCCUUAUGCCUUGAUUAAGGGGAUUAUCCUCCGCAAGAUUUUCACCACGAUUUCGUUCUGCAUUGUAUUGCGCAUGGCAGUGACACGGCAAUUUCCGUGGGCCGUGCAGACAUGGUAUGACUCGCUUGGAGCGAUCAACAAAAUCCAAGACUUCUUGCAAAAGCAAGAGUACAAGACCCUGGAGUACAAUCUUACUACUACGGAGGUAGUAAUGGAGAAUGUGACGGCUUUUUGGGAAGAGGGUUUUGGAGAACUGUUUGAGAAAGCAAAGCAGAAUAACAACAACCGCAAGACCUCAAAUGGGGACGAUUCCCUGUUUUUCUCGAACUUCUCCCUGCUCGGAACACCCGUGUUGAAGGACAUCAAUUUCAAGAUUGAGAGGGGACAGCUUCUCGCGGUAGCGGGAAGCACUGGUGCGGGAAAAACUAGCCUCUUGAUGGUGAUUAUGGGGGAGCUUGAGCCCAGCGAGGGGAAGAUUAAACACUCCGGGCGUAUCUCAUUCUGUAGCCAGUUUUCAUGGAUCAUGCCCGGAACCAUUAAAGAGAACAUCAUUUUCGGAGUAUCCUAUGAUGAGUACCGAUACAGAUCGGUCAUUAAGGCGUGCCAGUUGGAAGAGGACAUUUCUAAGUUCGCCGAGAAGGAUAACAUCGUCUUGGGAGAAGGGGGUAUUACAUUGUCGGGAGGGCAGCGAGCGCGGAUCAGCCUCGCGAGAGCGGUAUACAAAGAUGCAGAUUUGUAUCUGCUUGAUUCACCGUUUGGAUACCUCGACGUAUUGACAGAAAAAGAAAUCUUCGAGUCGUGCGUGUGUAAACUUAUGGCUAAUAAGACGAGAAUCCUGGUGACAUCAAAAAUGGAACACCUUAAGAAGGCGGACAAGAUCCUGAUCCUCCACGAAGGAUCGUCCUACUUUUACGGCACUUUCUCAGAGUUGCAAAACUUGCAGCCGGACUUCUCAAGCAAACUCAUGGGGUGUGACUCAUUCGACCAGUUCAGCGCGGAACGGCGGAACUCGAUCUUGACGGAAACGCUGCACCGAUUCUCGCUUGAGGGUGAUGCCCCGGUAUCGUGGACCGAGACAAAGAAGCAGUCGUUUAAGCAGACAGGAGAAUUUGGUGAGAAAAGAAAGAACAGUAUCUUGAAUCCUAUUAACUCAAUUCGCAAGUUCUCAAUCGUCCAGAAAACUCCACUGCAGAUGAAUGGAAUUGAAGAGGAUUCGGACGAACCCCUGGAGCGCAGGCUUAGCCUCGUGCCGGAUUCAGAGCAAGGGGAGGCCAUUCUUCCCCGGAUUUCGGUGAUUUCAACCGGACCUACACUUCAGGCGAGGCGAAGGCAAUCCGUGCUCAACCUCAUGACGCAUUCGGUAAACCAGGGGCAAAACAUUCACCGCAAAACGACGGCCUCAACGAGAAAAGUGUCACUUGCACCCCAGGCGAAUUUGACUGAACUCGACAUCUACAGCCGUAGGCUUUCGCAAGAAACCGGACUUGAGAUCAGCGAAGAAAUCAAUGAAGAAGAUUUGAAAGAGUGUUUCUUUGAUGACAUGGAAUCAAUCCCAGCGGUGACAACGUGGAACACAUACUUGCGUUACAUCACGGUGCACAAGUCCUUGAUUUUCGUCCUCAUCUGGUGUCUCGUGAUCUUUCUCGCUGAGGUCGCAGCGUCACUUGUGGUCCUCUGGCUGCUUGGUAAUACGCCCUUGCAAGACAAAGGCAAUUCUACACACUCAAGAAACAAUUCCUAUGCCGUGAUUAUCACUUCUACAAGCUCGUAUUACGUGUUUUACAUCUACGUAGGAGUGGCCGACACUCUGCUCGCGAUGGGUUUCUUCCGAGGACUCCCACUCGUUCACACGCUUAUCACUGUCUCCAAGAUUCUCCACCAUAAGAUGCUUCAUAGCGUACUGCAGGCUCCCAUGUCCACCUUGAAUACGCUCAAGGCGGGAGGUAUUUUGAAUCGCUUCUCAAAAGAUAUUGCAAUUUUGGAUGACCUUCUGCCCCUGACGAUCUUCGACUUCAUCCAGUUGUUGCUGAUCGUGAUUGGGGCUAUUGCAGUAGUCGCUGUCCUCCAGCCUUACAUUUUUGUCGCGACCGUUCCGGUGAUCGUGGCGUUUAUCAUGCUGCGGGCCUAUUUCUUGCAGACGUCACAGCAGCUUAAGCAACUGGAGUCUGAAGGGAGGUCGCCUAUCUUUACGCAUCUUGUGACCAGUUUGAAGGGAUUGUGGACGUUGCGCGCCUUUGGCAGGCAGCCCUACUUUGAAACACUGUUCCACAAAGCGCUGAAUCUCCAUACGGCAAAUUGGUUUUUGUAUUUGAGUACCCUCCGAUGGUUUCAGAUGCGCAUUGAGAUGAUUUUUGUGAUCUUCUUUAUCGCGGUGACUUUUAUCUCCAUCUUGACCACGGGAGAGGGCGAGGGACGGGUCGGUAUUAUCCUGACACUCGCCAUGAACAUUAUGAGCACUUUGCAGUGGGCAGUGAACAGCUCGAUUGAUGUGGAUAGCCUGAUGAGGUCCGUUUCGAGGGUCUUUAAGUUCAUCGACAUGCCGACGGAGGGAAAGCCCACAAAAAGUACGAAACCCUAUAAGAAUGGGCAAUUGAGUAAGGUAAUGAUCAUCGAGAACAGUCACGUGAAGAAGGAUGACAUCUGGCCUAGCGGGGGUCAGAUGACCGUGAAGGACCUGACGGCAAAAUACACCGAGGGAGGGAACGCAAUCCUUGAAAACAUCUCGUUCAGCAUUAGCCCCGGUCAGCGUGUGGGGUUGCUCGGGAGGACCGGGUCAGGAAAAUCGACGUUGCUGUCGGCCUUCUUGAGACUUCUGAAUACAGAGGGUGAGAUCCAGAUCGACGGCGUUUCGUGGGAUAGCAUCACCUUGCAGCAGUGGCGGAAAGCGUUUGGAGUAAUCCCCCAAAAGGUCUUUAUCUUUAGCGGAACCUUCCGAAAGAAUCUCGAUCCUUAUGAACAGUGGUCAGAUCAAGAGAUUUGGAAAGUCGCGGACGAGGUUGGCCUUCGGAGUGUAAUCGAGCAGUUUCCGGGAAAACUCGACUUUGUCCUUGUAGAUGGGGGAUGCGUCCUGUCGCAUGGGCACAAGCAGCUCAUGUGCCUGGCGCGAUCCGUCCUCUCUAAAGCGAAAAUUCUUCUCUUGGAUGAACCUUCGGCCCAUCUGGACCCGGUAACGUAUCAGAUCAUCAGAAGGACACUUAAGCAGGCGUUUGCCGACUGCACGGUGAUUCUCUGUGAGCAUCGUAUCGAGGCCAUGCUCGAAUGCCAGCAAUUUCUUGUCAUCGAAGAGAAUAAGGUCCGCCAGUACGACUCCAUCCAGAAGCUGCUUAAUGAGAGAUCAUUGUUCCGGCAGGCGAUUUCACCAUCCGAUAGGGUGAAACUUUUUCCACACAGAAAUUCGUCGAAGUGCAAGUCCAAACCGCAGAUCGCGGCCUUGAAAGAAGAGACUGAAGAAGAAGUUCAAGACACGCGUCUUUAA(配列番号17)
配列番号18
AUGGCCACUGGAUCAAGAACCUCACUGCUGCUCGCUUUUGGACUGCUUUGCCUGCCCUGGUUGCAAGAAGGAUCGGCUUUCCCGACCAUCCCACUCUCC(配列番号18)
配列番号19
AUGGCAACUGGAUCAAGAACCUCCCUCCUGCUCGCAUUCGGCCUGCUCUGUCUCCCAUGGCUCCAAGAAGGAAGCGCGUUCCCCACUAUCCCCCUCUCG(配列番号19)
配列番号20
CGGGUGGCAUCCCUGUGACCCCUCCCCAGUGCCUCUCCUGGCCCUGGAAGUUGCCACUCCAGUGCCCACCAGCCUUGUCCUAAUAAAAUUAAGUUGCAUCAAGCU(配列番号20)
本明細書は、本明細書中に引用される参考文献の教示を踏まえて最も完全に理解される。本明細書中の実施形態は、本発明の実施形態の例示を提供するものであり、本発明の範囲を制限するものと解釈されるべきではない。当業者は、多くの他の実施形態が本発明に包含されることを認識する。本開示において引用されるすべての刊行物及び特許は、参照によりその全体が組み込まれる。参照により組み込まれる材料が本明細書と矛盾するか、または一貫しない場合、本明細書はいかなるかかる材料にも優先する。本明細書におけるいかなる参考文献の引用も、かかる参考文献が本発明の先行技術であることを認めるものではない。
Claims (14)
- mRNAが充填されたナノ粒子を含む組成物であって、該ナノ粒子がリポソームであり、該mRNAが、インビトロ転写mRNAであり、配列番号3と少なくとも80%同一のコード配列を有し、該mRNAが、配列番号1のアミノ酸配列を含むヒトCFTRタンパク質をコードする、組成物。
- 前記mRNAが、配列番号3と少なくとも90%同一のコード配列を有する、請求項1に記載の組成物。
- 前記mRNAが、配列番号3と100%同一のコード配列を有する、請求項1に記載の組成物。
- 前記mRNAが、5’非翻訳領域(UTR)および/または3’UTRを含む、請求項1に記載の組成物。
- 前記5’UTRが配列番号4を含み、および/または前記3’UTRが配列番号5を含む、請求項4に記載の組成物。
- 前記mRNAがポリAテールをさらに含む、請求項4に記載の組成物。
- 前記ポリAテールが、少なくとも70、100、120、150、200、または250残基長である、請求項6に記載の組成物。
- 前記mRNAが5’キャップをさらに含む、請求項4に記載の組成物。
- 前記ヒトCFTRタンパク質が、肺の上皮細胞において発現される、請求項1に記載の組成物。
- 前記組成物が薬学的に許容される担体をさらに含む、請求項1に記載の組成物。
- 前記リポソームが1つ以上のPEG化脂質を含む、請求項1に記載の組成物。
- 前記リポソームが1つ以上のカチオン性脂質および1つ以上の非カチオン性脂質をさらに含む、請求項1に記載の組成物。
- 前記リポソームが、エアロゾル化により哺乳動物の肺に投与されることを特徴とする、請求項1に記載の組成物。
- 前記エアロゾル化が噴霧である、請求項13に記載の組成物。
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2023028264A JP2023054353A (ja) | 2013-03-14 | 2023-02-27 | CFTR mRNA組成物ならびに関連する方法及び使用 |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201361783663P | 2013-03-14 | 2013-03-14 | |
| US61/783,663 | 2013-03-14 | ||
| JP2020122566A JP2020169218A (ja) | 2013-03-14 | 2020-07-17 | CFTR mRNA組成物ならびに関連する方法及び使用 |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2020122566A Division JP2020169218A (ja) | 2013-03-14 | 2020-07-17 | CFTR mRNA組成物ならびに関連する方法及び使用 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2023028264A Division JP2023054353A (ja) | 2013-03-14 | 2023-02-27 | CFTR mRNA組成物ならびに関連する方法及び使用 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| JP2022022419A true JP2022022419A (ja) | 2022-02-03 |
| JP7236525B2 JP7236525B2 (ja) | 2023-03-09 |
Family
ID=50513499
Family Applications (5)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2016502924A Active JP6316930B2 (ja) | 2013-03-14 | 2014-03-14 | CFTRmRNA組成物ならびに関連する方法及び使用 |
| JP2018062010A Active JP6738366B2 (ja) | 2013-03-14 | 2018-03-28 | CFTR mRNA組成物ならびに関連する方法及び使用 |
| JP2020122566A Withdrawn JP2020169218A (ja) | 2013-03-14 | 2020-07-17 | CFTR mRNA組成物ならびに関連する方法及び使用 |
| JP2021200634A Active JP7236525B2 (ja) | 2013-03-14 | 2021-12-10 | CFTR mRNA組成物ならびに関連する方法及び使用 |
| JP2023028264A Pending JP2023054353A (ja) | 2013-03-14 | 2023-02-27 | CFTR mRNA組成物ならびに関連する方法及び使用 |
Family Applications Before (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2016502924A Active JP6316930B2 (ja) | 2013-03-14 | 2014-03-14 | CFTRmRNA組成物ならびに関連する方法及び使用 |
| JP2018062010A Active JP6738366B2 (ja) | 2013-03-14 | 2018-03-28 | CFTR mRNA組成物ならびに関連する方法及び使用 |
| JP2020122566A Withdrawn JP2020169218A (ja) | 2013-03-14 | 2020-07-17 | CFTR mRNA組成物ならびに関連する方法及び使用 |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| JP2023028264A Pending JP2023054353A (ja) | 2013-03-14 | 2023-02-27 | CFTR mRNA組成物ならびに関連する方法及び使用 |
Country Status (29)
| Country | Link |
|---|---|
| US (5) | US9181321B2 (ja) |
| EP (2) | EP2968586B1 (ja) |
| JP (5) | JP6316930B2 (ja) |
| KR (3) | KR20210122917A (ja) |
| CN (2) | CN115154620A (ja) |
| AU (3) | AU2014236305B2 (ja) |
| BR (1) | BR112015022868B1 (ja) |
| CA (1) | CA2904151C (ja) |
| CL (1) | CL2015002675A1 (ja) |
| CY (1) | CY1120790T1 (ja) |
| DK (1) | DK2968586T3 (ja) |
| EA (2) | EA037922B1 (ja) |
| ES (1) | ES2689523T3 (ja) |
| HK (1) | HK1218068A1 (ja) |
| HR (1) | HRP20181580T1 (ja) |
| HU (1) | HUE042640T2 (ja) |
| IL (5) | IL290953B2 (ja) |
| LT (1) | LT2968586T (ja) |
| MX (2) | MX393573B (ja) |
| NZ (1) | NZ751462A (ja) |
| PE (1) | PE20151773A1 (ja) |
| PL (1) | PL2968586T3 (ja) |
| PT (1) | PT2968586T (ja) |
| RS (1) | RS57739B1 (ja) |
| SG (2) | SG11201507391VA (ja) |
| SI (1) | SI2968586T1 (ja) |
| SM (1) | SMT201800546T1 (ja) |
| UA (1) | UA117008C2 (ja) |
| WO (1) | WO2014153052A2 (ja) |
Families Citing this family (65)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| ES2666559T3 (es) | 2009-12-01 | 2018-05-07 | Translate Bio, Inc. | Entrega del mrna para la aumentación de proteínas y enzimas en enfermedades genéticas humanas |
| EP2338520A1 (de) | 2009-12-21 | 2011-06-29 | Ludwig Maximilians Universität | Konjugat mit Zielfindungsligand und dessen Verwendung |
| WO2012075040A2 (en) * | 2010-11-30 | 2012-06-07 | Shire Human Genetic Therapies, Inc. | mRNA FOR USE IN TREATMENT OF HUMAN GENETIC DISEASES |
| ES2663360T3 (es) | 2011-06-08 | 2018-04-12 | Translate Bio, Inc. | Lípidos escindibles |
| BR112013031553A2 (pt) | 2011-06-08 | 2020-11-10 | Shire Human Genetic Therapies, Inc. | composições, mrna que codifica para uma hgla e seu uso, uso de pelo menos uma molécula de mrna e um veículo de transferência e uso de um mrna que codifica para proteína exógena |
| EP2859102A4 (en) | 2012-06-08 | 2016-05-11 | Shire Human Genetic Therapies | NUCLEASE RESISTANT POLYNUCLEOTIDES AND USES THEREOF |
| WO2014153052A2 (en) | 2013-03-14 | 2014-09-25 | Shire Human Genetic Therapies, Inc. | Cftr mrna compositions and related methods and uses |
| EP4446413A3 (en) | 2013-03-14 | 2024-12-18 | Translate Bio, Inc. | Methods for purification of messenger rna |
| CN105658242A (zh) | 2013-10-22 | 2016-06-08 | 夏尔人类遗传性治疗公司 | 用于苯丙酮尿症的mrna疗法 |
| EP4036241A1 (en) * | 2013-10-22 | 2022-08-03 | Translate Bio, Inc. | Cns delivery of mrna and uses thereof |
| ES2707966T3 (es) | 2013-10-22 | 2019-04-08 | Translate Bio Inc | Terapia de ARNm para la deficiencia en síntesis de argininosuccinato |
| AU2014340155B2 (en) | 2013-10-22 | 2018-11-01 | Massachusetts Institute Of Technology | Lipid formulations for delivery of messenger RNA |
| CN106164248B (zh) | 2014-04-25 | 2019-10-15 | 川斯勒佰尔公司 | 信使rna的纯化方法 |
| JP6895892B2 (ja) * | 2015-03-19 | 2021-06-30 | トランスレイト バイオ, インコーポレイテッド | ポンペ病のmRNA治療 |
| BR122023027721A2 (pt) | 2015-06-30 | 2024-01-30 | ethris GmbH | Composição |
| EP3405579A1 (en) | 2016-01-22 | 2018-11-28 | Modernatx, Inc. | Messenger ribonucleic acids for the production of intracellular binding polypeptides and methods of use thereof |
| SG11201808314QA (en) | 2016-04-08 | 2018-10-30 | Krystal Biotech Inc | Compositions and methods for the treatment of wounds, disorders, and diseases of the skin |
| CA3020343A1 (en) | 2016-04-08 | 2017-10-12 | Translate Bio, Inc. | Multimeric coding nucleic acid and uses thereof |
| US20190167811A1 (en) | 2016-04-13 | 2019-06-06 | Modernatx, Inc. | Lipid compositions and their uses for intratumoral polynucleotide delivery |
| EP3458108A4 (en) * | 2016-05-18 | 2020-04-22 | ModernaTX, Inc. | POLYNUCLEOTIDES ENCODING A CYSTIC FIBROSIS TRANSMEMBRANE CONDUCTANCE REGULATOR FOR THE TREATMENT OF CYSTIC FIBROSIS |
| SG11201809381XA (en) | 2016-05-18 | 2018-12-28 | Modernatx Inc | Polynucleotides encoding interleukin-12 (il12) and uses thereof |
| WO2017205739A1 (en) | 2016-05-26 | 2017-11-30 | University Of Iowa Research Foundation | cis AND trans REQUIREMENTS FOR TERMINAL RESOLUTION OF HUMAN BOCAVIRUS 1 |
| MX2019005470A (es) | 2016-11-10 | 2019-11-21 | Translate Bio Inc | Formulación de nanopartículas lipídicas basadas en ice mejorada para el suministro de arnm. |
| CN110225980B (zh) | 2016-11-21 | 2023-01-06 | 纳米线科技公司 | 化学组合物及其使用方法 |
| CN115845040A (zh) | 2017-01-27 | 2023-03-28 | 卫理公会医院 | 用于免疫疗法的核/壳结构平台 |
| HUE059594T2 (hu) | 2017-02-27 | 2022-11-28 | Translate Bio Inc | Hírvivõ RNS tisztítására szolgáló eljárások |
| MX2019010155A (es) | 2017-02-27 | 2020-12-10 | Translate Bio Inc | Arnm de cftr optimizado por codón novedoso. |
| EP3971291A1 (en) | 2017-02-27 | 2022-03-23 | Translate Bio, Inc. | Methods for purification of messenger rna |
| TW202428301A (zh) * | 2017-02-28 | 2024-07-16 | 法商賽諾菲公司 | 治療性rna |
| EP3600379B1 (en) | 2017-03-31 | 2022-12-21 | Accanis Biotech F&E GmbH & Co KG | Prevention and treatment of non-melanoma skin cancer (nmsc) |
| EP3398963B1 (en) * | 2017-05-04 | 2021-09-29 | Eberhard Karls Universität Tübingen Medizinische Fakultät | Chemically modified mrna for use in the treatment of a disease associated with the cftr gene |
| WO2018213476A1 (en) * | 2017-05-16 | 2018-11-22 | Translate Bio, Inc. | Treatment of cystic fibrosis by delivery of codon-optimized mrna encoding cftr |
| WO2018231990A2 (en) | 2017-06-14 | 2018-12-20 | Modernatx, Inc. | Polynucleotides encoding methylmalonyl-coa mutase |
| US10034951B1 (en) | 2017-06-21 | 2018-07-31 | New England Biolabs, Inc. | Use of thermostable RNA polymerases to produce RNAs having reduced immunogenicity |
| EP3437650A1 (en) | 2017-07-31 | 2019-02-06 | Accanis Biotech F&E GmbH & Co KG | Treatment of local skin hypotrophy conditions |
| SG11202000765PA (en) * | 2017-09-08 | 2020-03-30 | Generation Bio Co | Lipid nanoparticle formulations of non-viral, capsid-free dna vectors |
| EP3775236A1 (en) | 2018-04-12 | 2021-02-17 | Krystal Biotech, LLC | Compositions and methods for the treatment of autosomal recessive congenital ichthyosis |
| JP7602999B2 (ja) | 2018-04-27 | 2024-12-19 | クリスタル バイオテック インコーポレイテッド | 審美的用途のための美容用タンパク質をコードする組換え核酸 |
| EP3794146B1 (en) | 2018-05-14 | 2025-12-10 | Bruker Spatial Biology, Inc. | Method for identifying a predetermined nucleotide sequence |
| CA3107055A1 (en) * | 2018-07-23 | 2020-01-30 | Translate Bio, Inc. | Dry powder formulations for messenger rna |
| KR20210060480A (ko) | 2018-08-24 | 2021-05-26 | 트랜슬레이트 바이오 인코포레이티드 | 전령 rna의 정제 방법 |
| GB2617430B (en) * | 2018-09-04 | 2023-12-27 | Univ Texas | Compositions and methods for organ specific delivery of nucleic acids |
| WO2020056294A1 (en) * | 2018-09-14 | 2020-03-19 | Translate Bio, Inc. | Composition and methods for treatment of methylmalonic acidemia |
| JP7562515B2 (ja) | 2018-09-24 | 2024-10-07 | クリスタル バイオテック インコーポレイテッド | ネザートン症候群の治療のための組成物及び方法 |
| US11072808B2 (en) | 2018-10-04 | 2021-07-27 | New England Biolabs, Inc. | Methods and compositions for increasing capping efficiency of transcribed RNA |
| AU2019355177A1 (en) | 2018-10-04 | 2021-05-06 | New England Biolabs, Inc. | Methods and compositions for increasing capping efficiency of transcribed RNA |
| WO2020097509A1 (en) | 2018-11-08 | 2020-05-14 | Translate Bio, Inc. | Methods and compositions for messenger rna purification |
| US12280147B2 (en) | 2018-11-09 | 2025-04-22 | Translate Bio, Inc. | PEG lipidoid compounds |
| CA3120647A1 (en) | 2018-11-21 | 2020-05-28 | Translate Bio, Inc. | Treatment of cystic fibrosis by delivery of nebulized mrna encoding cftr |
| KR20210124277A (ko) | 2019-02-08 | 2021-10-14 | 크리스탈 바이오테크, 인크. | Cftr 폴리펩타이드를 전달하기 위한 조성물 및 방법 |
| CA3129663A1 (en) | 2019-02-11 | 2020-08-20 | Ethris Gmbh | Mrna purification by tangential flow filtration |
| AU2020257182A1 (en) * | 2019-04-15 | 2021-12-09 | Spirovant Sciences, Inc. | Methods and compositions for transgene expression |
| CN114641318A (zh) * | 2019-04-15 | 2022-06-17 | 衣阿华大学研究基金会 | 用于治疗囊性纤维化的组合物和方法 |
| MX2022003394A (es) * | 2019-09-20 | 2022-06-22 | Translate Bio Inc | Arnm que codifica cftr manipulado. |
| AU2021264465A1 (en) * | 2020-04-27 | 2022-12-15 | University Of Iowa Research Foundation | Compositions and methods for the treatment of cystic fibrosis |
| JP2023524071A (ja) | 2020-05-01 | 2023-06-08 | アークトゥラス・セラピューティクス・インコーポレイテッド | 嚢胞性線維症を治療するための核酸及び方法 |
| US20230056935A1 (en) | 2020-09-16 | 2023-02-23 | Lg Chem, Ltd. | Compound, Antimicrobial Deodorant Composition Comprising Same, and Method for Producing Same |
| US12458604B2 (en) | 2020-10-14 | 2025-11-04 | The Trustees Of The University Of Pennsylvania | Methods of lipid nanoparticle manufacture and compositions derived therefrom |
| MX2023011243A (es) * | 2021-03-23 | 2023-12-14 | Recode Therapeutics Inc | Composiciones de polinucleotidos, formulaciones relacionadas y metodos de uso de los mismos. |
| CA3213789A1 (en) | 2021-04-02 | 2022-10-06 | Krystal Biotech, Inc. | Viral vectors for cancer therapy |
| JP2024520834A (ja) * | 2021-06-09 | 2024-05-24 | レコード・セラピューティクス・インコーポレイテッド | ポリヌクレオチド組成物、関連する製剤及びその使用方法 |
| CN120693181A (zh) | 2022-12-08 | 2025-09-23 | 瑞科德治疗公司 | 脂质纳米颗粒组合物及其用途 |
| EP4434534A1 (en) | 2023-03-22 | 2024-09-25 | ADvantage Therapeutics, Inc. | Klotho mrna |
| WO2025036956A1 (en) * | 2023-08-14 | 2025-02-20 | Ethris Gmbh | Non-systemic mrna administration |
| US12364773B2 (en) | 2023-12-01 | 2025-07-22 | Recode Therapeutics, Inc. | Lipid nanoparticle compositions and uses thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010506838A (ja) * | 2006-10-12 | 2010-03-04 | コペルニクス セラピューティクス インコーポレイテッド | コドン最適化cftr |
| WO2011012316A2 (de) * | 2009-07-31 | 2011-02-03 | Ludwig-Maximilians-Universität | Rna mit einer kombination aus unmodifizierten und modifizierten nucleotiden zur proteinexpression |
Family Cites Families (437)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2647121A (en) | 1951-02-02 | 1953-07-28 | Ruth P Jacoby | Diamine-bis-acetamides |
| US2819718A (en) | 1953-07-16 | 1958-01-14 | Isidore H Goldman | Drainage tube |
| US2717909A (en) | 1953-09-24 | 1955-09-13 | Monsanto Chemicals | Hydroxyethyl-keryl-alkylene-ammonium compounds |
| US2844629A (en) | 1956-04-25 | 1958-07-22 | American Home Prod | Fatty acid amides and derivatives thereof |
| US3096560A (en) | 1958-11-21 | 1963-07-09 | William J Liebig | Process for synthetic vascular implants |
| GB1072118A (en) | 1962-12-01 | 1967-06-14 | Sandoz Ag | Amides of aminopropionic acid |
| JPS5141663B1 (ja) | 1966-03-12 | 1976-11-11 | ||
| JPS4822365B1 (ja) | 1968-10-25 | 1973-07-05 | ||
| NL143127B (nl) | 1969-02-04 | 1974-09-16 | Rhone Poulenc Sa | Versterkingsorgaan voor een defecte hartklep. |
| US3614954A (en) | 1970-02-09 | 1971-10-26 | Medtronic Inc | Electronic standby defibrillator |
| US3614955A (en) | 1970-02-09 | 1971-10-26 | Medtronic Inc | Standby defibrillator and method of operation |
| JPS5024216B1 (ja) | 1970-12-29 | 1975-08-14 | ||
| JPS5012146Y2 (ja) | 1971-07-27 | 1975-04-15 | ||
| US3945052A (en) | 1972-05-01 | 1976-03-23 | Meadox Medicals, Inc. | Synthetic vascular graft and method for manufacturing the same |
| US3805301A (en) | 1972-07-28 | 1974-04-23 | Meadox Medicals Inc | Tubular grafts having indicia thereon |
| JPS49127908A (ja) | 1973-04-20 | 1974-12-07 | ||
| JPS5624664B2 (ja) | 1973-06-28 | 1981-06-08 | ||
| US4013507A (en) | 1973-09-18 | 1977-03-22 | California Institute Of Technology | Ionene polymers for selectively inhibiting the vitro growth of malignant cells |
| JPS5123537A (ja) | 1974-04-26 | 1976-02-25 | Adeka Argus Chemical Co Ltd | Kasozaisoseibutsu |
| GB1527592A (en) | 1974-08-05 | 1978-10-04 | Ici Ltd | Wound dressing |
| US3995623A (en) | 1974-12-23 | 1976-12-07 | American Hospital Supply Corporation | Multipurpose flow-directed catheter |
| JPS5813576B2 (ja) | 1974-12-27 | 1983-03-14 | アデカ ア−ガスカガク カブシキガイシヤ | 安定化された合成高分子組成物 |
| US4281669A (en) | 1975-05-09 | 1981-08-04 | Macgregor David C | Pacemaker electrode with porous system |
| DE2520814A1 (de) | 1975-05-09 | 1976-11-18 | Bayer Ag | Lichtstabilisierung von polyurethanen |
| JPS5210847A (en) | 1975-07-16 | 1977-01-27 | Nippon Steel Corp | Pinch roll |
| US4096860A (en) | 1975-10-08 | 1978-06-27 | Mclaughlin William F | Dual flow encatheter |
| CA1069652A (en) | 1976-01-09 | 1980-01-15 | Alain F. Carpentier | Supported bioprosthetic heart valve with compliant orifice ring |
| US4134402A (en) | 1976-02-11 | 1979-01-16 | Mahurkar Sakharam D | Double lumen hemodialysis catheter |
| US4072146A (en) | 1976-09-08 | 1978-02-07 | Howes Randolph M | Venous catheter device |
| US4335723A (en) | 1976-11-26 | 1982-06-22 | The Kendall Company | Catheter having inflatable retention means |
| US4099528A (en) | 1977-02-17 | 1978-07-11 | Sorenson Research Co., Inc. | Double lumen cannula |
| US4140126A (en) | 1977-02-18 | 1979-02-20 | Choudhury M Hasan | Method for performing aneurysm repair |
| US4265745A (en) | 1977-05-25 | 1981-05-05 | Teijin Limited | Permselective membrane |
| US4182833A (en) | 1977-12-07 | 1980-01-08 | Celanese Polymer Specialties Company | Cationic epoxide-amine reaction products |
| US4180068A (en) | 1978-04-13 | 1979-12-25 | Motion Control, Incorporated | Bi-directional flow catheter with retractable trocar/valve structure |
| DE2960875D1 (en) | 1978-04-19 | 1981-12-10 | Ici Plc | A method of preparing a tubular product by electrostatic spinning |
| US4284459A (en) | 1978-07-03 | 1981-08-18 | The Kendall Company | Method for making a molded catheter |
| US4227533A (en) | 1978-11-03 | 1980-10-14 | Bristol-Myers Company | Flushable urinary catheter |
| US4375817A (en) | 1979-07-19 | 1983-03-08 | Medtronic, Inc. | Implantable cardioverter |
| DE3010841A1 (de) | 1980-03-21 | 1981-10-08 | Ulrich Dr.med. 6936 Haag Uthmann | Katheder |
| US4308085A (en) | 1980-07-28 | 1981-12-29 | Jenoptik Jena Gmbh | Process for the preparation of high molecular thermoplastic epoxide-amine-polyadducts |
| US4339369A (en) | 1981-04-23 | 1982-07-13 | Celanese Corporation | Cationic epoxide-amine reaction products |
| US4406656A (en) | 1981-06-01 | 1983-09-27 | Brack Gillium Hattler | Venous catheter having collapsible multi-lumens |
| US4475972A (en) | 1981-10-01 | 1984-10-09 | Ontario Research Foundation | Implantable material |
| US4401472A (en) | 1982-02-26 | 1983-08-30 | Martin Marietta Corporation | Hydraulic cement mixes and processes for improving hydraulic cement mixes |
| US4568329A (en) | 1982-03-08 | 1986-02-04 | Mahurkar Sakharam D | Double lumen catheter |
| US4546499A (en) | 1982-12-13 | 1985-10-15 | Possis Medical, Inc. | Method of supplying blood to blood receiving vessels |
| US4530113A (en) | 1983-05-20 | 1985-07-23 | Intervascular, Inc. | Vascular grafts with cross-weave patterns |
| US4550447A (en) | 1983-08-03 | 1985-11-05 | Shiley Incorporated | Vascular graft prosthesis |
| US4647416A (en) | 1983-08-03 | 1987-03-03 | Shiley Incorporated | Method of preparing a vascular graft prosthesis |
| US5104399A (en) | 1986-12-10 | 1992-04-14 | Endovascular Technologies, Inc. | Artificial graft and implantation method |
| US4571241A (en) | 1983-12-16 | 1986-02-18 | Christopher T Graham | Urinary catheter with collapsible urethral tube |
| US4710169A (en) | 1983-12-16 | 1987-12-01 | Christopher T Graham | Urinary catheter with collapsible urethral tube |
| US4737518A (en) | 1984-04-03 | 1988-04-12 | Takeda Chemical Industries, Ltd. | Lipid derivatives, their production and use |
| US4562596A (en) | 1984-04-25 | 1986-01-07 | Elliot Kornberg | Aortic graft, device and method for performing an intraluminal abdominal aortic aneurysm repair |
| US4782836A (en) | 1984-05-24 | 1988-11-08 | Intermedics, Inc. | Rate adaptive cardiac pacemaker responsive to patient activity and temperature |
| US4897355A (en) | 1985-01-07 | 1990-01-30 | Syntex (U.S.A.) Inc. | N[ω,(ω-1)-dialkyloxy]- and N-[ω,(ω-1)-dialkenyloxy]-alk-1-yl-N,N,N-tetrasubstituted ammonium lipids and uses therefor |
| US4662382A (en) | 1985-01-16 | 1987-05-05 | Intermedics, Inc. | Pacemaker lead with enhanced sensitivity |
| US4762915A (en) | 1985-01-18 | 1988-08-09 | Liposome Technology, Inc. | Protein-liposome conjugates |
| US4860751A (en) | 1985-02-04 | 1989-08-29 | Cordis Corporation | Activity sensor for pacemaker control |
| US5223263A (en) | 1988-07-07 | 1993-06-29 | Vical, Inc. | Liponucleotide-containing liposomes |
| CA1320724C (en) | 1985-07-19 | 1993-07-27 | Koichi Kanehira | Terpene amino alcohols and medicinal uses thereof |
| US4701162A (en) | 1985-09-24 | 1987-10-20 | The Kendall Company | Foley catheter assembly |
| US4737323A (en) | 1986-02-13 | 1988-04-12 | Liposome Technology, Inc. | Liposome extrusion method |
| DE3616824A1 (de) | 1986-05-17 | 1987-11-19 | Schering Ag | Verwendung von haertbaren kunstharzmischungen fuer oberflaechenbeschichtungen und druckfarben und verfahren zu ihrer herstellung |
| DE3780374D1 (de) | 1986-07-31 | 1992-08-20 | Irnich Werner | Frequenzadaptierender herzschrittmacher. |
| US4960409A (en) | 1986-09-11 | 1990-10-02 | Catalano Marc L | Method of using bilumen peripheral venous catheter with adapter |
| JPH0829776B2 (ja) | 1986-10-29 | 1996-03-27 | 東燃化学株式会社 | 合成樹脂製容器及びその製造用金型 |
| US4720517A (en) | 1986-11-24 | 1988-01-19 | Ciba-Geigy Corporation | Compositions stabilized with N-hydroxyiminodiacetic and dipropionic acids and esters thereof |
| US4920016A (en) | 1986-12-24 | 1990-04-24 | Linear Technology, Inc. | Liposomes with enhanced circulation time |
| JPS63154788U (ja) | 1987-03-31 | 1988-10-11 | ||
| DE3728917A1 (de) | 1987-08-29 | 1989-03-09 | Roth Hermann J | Neue lipide mit unsymmetrisch substituierter disulfidbruecke |
| US4946683A (en) | 1987-11-18 | 1990-08-07 | Vestar, Inc. | Multiple step entrapment/loading procedure for preparing lipophilic drug-containing liposomes |
| US5047540A (en) | 1987-12-17 | 1991-09-10 | Shionogi & Co., Ltd. | Lipid derivatives |
| US5138067A (en) | 1987-12-17 | 1992-08-11 | Shionogi & Co. Ltd. | Lipid derivatives |
| US4892540A (en) | 1988-04-21 | 1990-01-09 | Sorin Biomedica S.P.A. | Two-leaflet prosthetic heart valve |
| US5176661A (en) | 1988-09-06 | 1993-01-05 | Advanced Cardiovascular Systems, Inc. | Composite vascular catheter |
| US5024671A (en) | 1988-09-19 | 1991-06-18 | Baxter International Inc. | Microporous vascular graft |
| US5200395A (en) | 1988-10-18 | 1993-04-06 | Ajinomoto Company, Inc. | Pharmaceutical composition of BUF-5 for treating anemia |
| CA2001401A1 (en) | 1988-10-25 | 1990-04-25 | Claude Piantadosi | Quaternary amine containing ether or ester lipid derivatives and therapeutic compositions |
| EP1026253B2 (en) | 1989-03-21 | 2012-12-19 | Vical Incorporated | Expression of exogenous polynucleotide sequences in a vertebrate |
| US6214804B1 (en) | 1989-03-21 | 2001-04-10 | Vical Incorporated | Induction of a protective immune response in a mammal by injecting a DNA sequence |
| US5703055A (en) | 1989-03-21 | 1997-12-30 | Wisconsin Alumni Research Foundation | Generation of antibodies through lipid mediated DNA delivery |
| FR2645866B1 (fr) | 1989-04-17 | 1991-07-05 | Centre Nat Rech Scient | Nouvelles lipopolyamines, leur preparation et leur emploi |
| US5194654A (en) | 1989-11-22 | 1993-03-16 | Vical, Inc. | Lipid derivatives of phosphonoacids for liposomal incorporation and method of use |
| US5279833A (en) | 1990-04-04 | 1994-01-18 | Yale University | Liposomal transfection of nucleic acids into animal cells |
| US5101824A (en) | 1990-04-16 | 1992-04-07 | Siemens-Pacesetter, Inc. | Rate-responsive pacemaker with circuitry for processing multiple sensor inputs |
| US5264618A (en) | 1990-04-19 | 1993-11-23 | Vical, Inc. | Cationic lipids for intracellular delivery of biologically active molecules |
| EP0549590A1 (en) | 1990-07-26 | 1993-07-07 | LANE, Rodney James | Self expanding vascular endoprosthesis for aneurysms |
| US5693338A (en) | 1994-09-29 | 1997-12-02 | Emisphere Technologies, Inc. | Diketopiperazine-based delivery systems |
| DE9117152U1 (de) | 1990-10-09 | 1996-07-11 | Cook Inc., Bloomington, Ind. | Stent |
| DE59008908D1 (de) | 1990-12-19 | 1995-05-18 | Osypka Peter | Herzschrittmacherleitung mit einem inneren Kanal und mit einem Elektrodenkopf. |
| US5116360A (en) | 1990-12-27 | 1992-05-26 | Corvita Corporation | Mesh composite graft |
| US5405363A (en) | 1991-03-15 | 1995-04-11 | Angelon Corporation | Implantable cardioverter defibrillator having a smaller displacement volume |
| US5330768A (en) | 1991-07-05 | 1994-07-19 | Massachusetts Institute Of Technology | Controlled drug delivery using polymer/pluronic blends |
| US5545449A (en) | 1991-10-02 | 1996-08-13 | Weyerhaeuser Company | Polyether-reinforced fiber-based materials |
| US6013638A (en) * | 1991-10-02 | 2000-01-11 | The United States Of America As Represented By The Department Of Health And Human Services | Adenovirus comprising deletions on the E1A, E1B and E3 regions for transfer of genes to the lung |
| US5151105A (en) | 1991-10-07 | 1992-09-29 | Kwan Gett Clifford | Collapsible vessel sleeve implant |
| JPH0753535B2 (ja) | 1992-02-14 | 1995-06-07 | 株式会社カワタ | 粉粒体の材料供給装置における弁装置 |
| US5284491A (en) | 1992-02-27 | 1994-02-08 | Medtronic, Inc. | Cardiac pacemaker with hysteresis behavior |
| US5352461A (en) | 1992-03-11 | 1994-10-04 | Pharmaceutical Discovery Corporation | Self assembling diketopiperazine drug delivery system |
| SE9200951D0 (sv) | 1992-03-27 | 1992-03-27 | Kabi Pharmacia Ab | Pharmaceutical composition containing a defined lipid system |
| EP0635019B1 (en) | 1992-04-06 | 1999-05-26 | Biosite Diagnostics Inc. | Opiate derivatives and protein and polypeptide opiate derivative conjugates and labels |
| US6670178B1 (en) | 1992-07-10 | 2003-12-30 | Transkaryotic Therapies, Inc. | In Vivo production and delivery of insulinotropin for gene therapy |
| KR950702628A (ko) | 1992-08-01 | 1995-07-29 | 치세이 라 | 항알레르기 조성물(Antiallergic agent) |
| US5334761A (en) | 1992-08-28 | 1994-08-02 | Life Technologies, Inc. | Cationic lipids |
| US5461223A (en) | 1992-10-09 | 1995-10-24 | Eastman Kodak Company | Bar code detecting circuitry |
| US5300022A (en) | 1992-11-12 | 1994-04-05 | Martin Klapper | Urinary catheter and bladder irrigation system |
| US5496362A (en) | 1992-11-24 | 1996-03-05 | Cardiac Pacemakers, Inc. | Implantable conformal coil patch electrode with multiple conductive elements for cardioversion and defibrillation |
| US5552155A (en) | 1992-12-04 | 1996-09-03 | The Liposome Company, Inc. | Fusogenic lipsomes and methods for making and using same |
| US5716395A (en) | 1992-12-11 | 1998-02-10 | W.L. Gore & Associates, Inc. | Prosthetic vascular graft |
| EP0685457B1 (en) | 1993-02-19 | 1999-12-15 | Nippon Shinyaku Company, Limited | Glycerol derivative, device and pharmaceutical composition |
| US5395619A (en) | 1993-03-03 | 1995-03-07 | Liposome Technology, Inc. | Lipid-polymer conjugates and liposomes |
| US5697953A (en) | 1993-03-13 | 1997-12-16 | Angeion Corporation | Implantable cardioverter defibrillator having a smaller displacement volume |
| US5624976A (en) | 1994-03-25 | 1997-04-29 | Dentsply Gmbh | Dental filling composition and method |
| US5314430A (en) | 1993-06-24 | 1994-05-24 | Medtronic, Inc. | Atrial defibrillator employing transvenous and subcutaneous electrodes and method of use |
| DE4325848A1 (de) | 1993-07-31 | 1995-02-02 | Basf Ag | Verfahren zur Herstellung von N-(2-Hydroxyethyl)-piperazin |
| EP0647462A1 (en) | 1993-10-06 | 1995-04-12 | The Kansai Electric Power Co., Inc. | Method for removing carbon dioxide from combustion exhaust gas |
| US5609624A (en) | 1993-10-08 | 1997-03-11 | Impra, Inc. | Reinforced vascular graft and method of making same |
| SE9303481L (sv) | 1993-10-22 | 1995-04-23 | Berol Nobel Ab | Hygienkomposition |
| WO1995013033A1 (en) | 1993-11-08 | 1995-05-18 | Lazarus Harrison M | Intraluminal vascular graft and method |
| KR960705798A (ko) | 1993-11-24 | 1996-11-08 | 벤자민 에프 맥그로우 | 피페라진의 양쪽친화성 유도체(amphiphilic derivatives of piperazine) |
| US5595756A (en) | 1993-12-22 | 1997-01-21 | Inex Pharmaceuticals Corporation | Liposomal compositions for enhanced retention of bioactive agents |
| US5464924A (en) | 1994-01-07 | 1995-11-07 | The Dow Chemical Company | Flexible poly(amino ethers) for barrier packaging |
| US5844107A (en) | 1994-03-23 | 1998-12-01 | Case Western Reserve University | Compacted nucleic acids and their delivery to cells |
| EP0758883B1 (en) | 1994-04-12 | 2003-03-19 | The Liposome Company, Inc. | Fusogenic liposomes and methods of making and using same |
| US6392118B1 (en) | 1994-07-20 | 2002-05-21 | Neurotech S.A. | Mx-1 conditionally immortalized cells |
| US5820873A (en) | 1994-09-30 | 1998-10-13 | The University Of British Columbia | Polyethylene glycol modified ceramide lipids and liposome uses thereof |
| US5885613A (en) | 1994-09-30 | 1999-03-23 | The University Of British Columbia | Bilayer stabilizing components and their use in forming programmable fusogenic liposomes |
| US5641665A (en) | 1994-11-28 | 1997-06-24 | Vical Incorporated | Plasmids suitable for IL-2 expression |
| US6071890A (en) | 1994-12-09 | 2000-06-06 | Genzyme Corporation | Organ-specific targeting of cationic amphiphile/DNA complexes for gene therapy |
| US5965434A (en) | 1994-12-29 | 1999-10-12 | Wolff; Jon A. | Amphipathic PH sensitive compounds and delivery systems for delivering biologically active compounds |
| US6485726B1 (en) | 1995-01-17 | 2002-11-26 | The Brigham And Women's Hospital, Inc. | Receptor specific transepithelial transport of therapeutics |
| US5830430A (en) | 1995-02-21 | 1998-11-03 | Imarx Pharmaceutical Corp. | Cationic lipids and the use thereof |
| JP2001523215A (ja) | 1995-04-17 | 2001-11-20 | イマークス ファーマシューティカル コーポレーション | ハイブリッド磁気共鳴造影剤 |
| US5772694A (en) | 1995-05-16 | 1998-06-30 | Medical Carbon Research Institute L.L.C. | Prosthetic heart valve with improved blood flow |
| US5783383A (en) | 1995-05-23 | 1998-07-21 | The Board Of Trustees Of The Leland Stanford Junior University | Method of detecting cytomegalovirus (CMV) |
| US7422902B1 (en) | 1995-06-07 | 2008-09-09 | The University Of British Columbia | Lipid-nucleic acid particles prepared via a hydrophobic lipid-nucleic acid complex intermediate and use for gene transfer |
| US5609629A (en) | 1995-06-07 | 1997-03-11 | Med Institute, Inc. | Coated implantable medical device |
| JP4335310B2 (ja) | 1995-06-07 | 2009-09-30 | ザ ユニバーシティ オブ ブリティッシュ コロンビア | 疎水性脂質−核酸複合中間体を通して調製される脂質−核酸粒子、及び遺伝子移送のための使用 |
| US5705385A (en) | 1995-06-07 | 1998-01-06 | Inex Pharmaceuticals Corporation | Lipid-nucleic acid particles prepared via a hydrophobic lipid-nucleic acid complex intermediate and use for gene transfer |
| US5981501A (en) | 1995-06-07 | 1999-11-09 | Inex Pharmaceuticals Corp. | Methods for encapsulating plasmids in lipid bilayers |
| US5607385A (en) | 1995-08-17 | 1997-03-04 | Medtronic, Inc. | Device and algorithm for a combined cardiomyostimulator and a cardiac pacer-carioverter-defibrillator |
| US5744335A (en) | 1995-09-19 | 1998-04-28 | Mirus Corporation | Process of transfecting a cell with a polynucleotide mixed with an amphipathic compound and a DNA-binding protein |
| FR2740978B1 (fr) | 1995-11-10 | 1998-01-02 | Ela Medical Sa | Dispositif medical actif du type defibrillateur/cardioverteur implantable |
| US5874105A (en) | 1996-01-31 | 1999-02-23 | Collaborative Laboratories, Inc. | Lipid vesicles formed with alkylammonium fatty acid salts |
| EP0910576B1 (en) | 1996-04-11 | 2004-08-11 | University Of British Columbia | Fusogenic liposomes |
| US5935936A (en) | 1996-06-03 | 1999-08-10 | Genzyme Corporation | Compositions comprising cationic amphiphiles and co-lipids for intracellular delivery of therapeutic molecules |
| US5913848A (en) | 1996-06-06 | 1999-06-22 | Luther Medical Products, Inc. | Hard tip over-the-needle catheter and method of manufacturing the same |
| US5677124A (en) | 1996-07-03 | 1997-10-14 | Ambion, Inc. | Ribonuclease resistant viral RNA standards |
| US5736573A (en) | 1996-07-31 | 1998-04-07 | Galat; Alexander | Lipid and water soluble derivatives of drugs |
| US7288266B2 (en) | 1996-08-19 | 2007-10-30 | United States Of America As Represented By The Secretary, Department Of Health And Human Services | Liposome complexes for increased systemic delivery |
| US6271208B1 (en) | 1996-08-26 | 2001-08-07 | Transgene S.A. | Process of making cationic lipid-nucleic acid complexes |
| CA2271388C (en) | 1996-09-13 | 2007-11-06 | The School Of Pharmacy, University Of London | Liposomes encapsulating polynucleotides operatively coding for immunogenic polypeptides |
| TW520297B (en) | 1996-10-11 | 2003-02-11 | Sequus Pharm Inc | Fusogenic liposome composition and method |
| CA2270396C (en) | 1996-11-04 | 2008-03-11 | Qiagen Gmbh | Cationic reagents for transfection |
| US6887665B2 (en) | 1996-11-14 | 2005-05-03 | Affymetrix, Inc. | Methods of array synthesis |
| US5985930A (en) | 1996-11-21 | 1999-11-16 | Pasinetti; Giulio M. | Treatment of neurodegenerative conditions with nimesulide |
| US6204297B1 (en) | 1996-11-26 | 2001-03-20 | Rhodia Inc. | Nonionic gemini surfactants |
| JPH10197978A (ja) | 1997-01-09 | 1998-07-31 | Mitsubishi Paper Mills Ltd | ハロゲン化銀写真感光材料 |
| EP0853123A1 (de) | 1997-01-10 | 1998-07-15 | Roche Diagnostics GmbH | Reinigung von DNA durch Cross-Flow-Filtration |
| FR2760193B1 (fr) | 1997-02-28 | 1999-05-28 | Transgene Sa | Lipides et complexes de lipides cationiques et de substances actives, notamment pour la transfection de cellules |
| US5837283A (en) | 1997-03-12 | 1998-11-17 | The Regents Of The University Of California | Cationic lipid compositions targeting angiogenic endothelial cells |
| US5945326A (en) | 1997-03-20 | 1999-08-31 | New England Biolabs, Inc. | Method for cloning and producing the Spel restriction endonuclease |
| AU733310C (en) | 1997-05-14 | 2001-11-29 | University Of British Columbia, The | High efficiency encapsulation of charged therapeutic agents in lipid vesicles |
| US20030104044A1 (en) | 1997-05-14 | 2003-06-05 | Semple Sean C. | Compositions for stimulating cytokine secretion and inducing an immune response |
| US6835395B1 (en) | 1997-05-14 | 2004-12-28 | The University Of British Columbia | Composition containing small multilamellar oligodeoxynucleotide-containing lipid vesicles |
| JPH115786A (ja) | 1997-06-13 | 1999-01-12 | Pola Chem Ind Inc | 新規アミノヒドロキシプロピルピペラジン誘導体 |
| US6067471A (en) | 1998-08-07 | 2000-05-23 | Cardiac Pacemakers, Inc. | Atrial and ventricular implantable cardioverter-defibrillator and lead system |
| JPH1180142A (ja) | 1997-09-05 | 1999-03-26 | Pola Chem Ind Inc | ジフェニルアルキル化合物の製造法 |
| AU9319398A (en) * | 1997-09-19 | 1999-04-05 | Sequitur, Inc. | Sense mrna therapy |
| US6165763A (en) | 1997-10-30 | 2000-12-26 | Smithkline Beecham Corporation | Ornithine carbamoyltransferase |
| US6096075A (en) | 1998-01-22 | 2000-08-01 | Medical Carbon Research Institute, Llc | Prosthetic heart valve |
| US6617171B2 (en) | 1998-02-27 | 2003-09-09 | The General Hospital Corporation | Methods for diagnosing and treating autoimmune disease |
| US6271209B1 (en) | 1998-04-03 | 2001-08-07 | Valentis, Inc. | Cationic lipid formulation delivering nucleic acid to peritoneal tumors |
| US6176877B1 (en) | 1998-04-20 | 2001-01-23 | St. Jude Medical, Inc. | Two piece prosthetic heart valve |
| DE19822602A1 (de) | 1998-05-20 | 1999-11-25 | Goldschmidt Ag Th | Verfahren zur Herstellung von Polyaminosäureestern durch Veresterung von sauren Polyaminosäuren oder Umesterung von Polyaminosäureestern |
| NO313244B1 (no) | 1998-07-08 | 2002-09-02 | Crew Dev Corp | Fremgangsmåte for isolering og produksjon av magnesitt eller magnesiumklorid |
| US6055454A (en) | 1998-07-27 | 2000-04-25 | Cardiac Pacemakers, Inc. | Cardiac pacemaker with automatic response optimization of a physiologic sensor based on a second sensor |
| US6312926B1 (en) | 1998-08-14 | 2001-11-06 | University Of Medicine & Dentistry Of New Jersey | mRNA capping enzymes and uses thereof |
| WO2000010622A1 (en) | 1998-08-20 | 2000-03-02 | Cook Incorporated | Coated implantable medical device |
| US6210892B1 (en) | 1998-10-07 | 2001-04-03 | Isis Pharmaceuticals, Inc. | Alteration of cellular behavior by antisense modulation of mRNA processing |
| US6468793B1 (en) * | 1998-10-23 | 2002-10-22 | Florida State University Research Foundation | CFTR genes and proteins for cystic fibrosis gene therapy |
| AU1830200A (en) | 1998-11-25 | 2000-06-13 | Vanderbilt University | Cationic liposomes for gene transfer |
| US6248725B1 (en) | 1999-02-23 | 2001-06-19 | Amgen, Inc. | Combinations and methods for promoting in vivo liver cell proliferation and enhancing in vivo liver-directed gene transduction |
| US6379698B1 (en) | 1999-04-06 | 2002-04-30 | Isis Pharmaceuticals, Inc. | Fusogenic lipids and vesicles |
| EP1173600A2 (en) | 1999-04-20 | 2002-01-23 | The University Of British Columbia | Cationic peg-lipids and methods of use |
| MXPA01010750A (es) | 1999-04-23 | 2003-08-20 | Alza Corp | Conjugado que tiene un enlace dividible para utilizarse en un liposoma. |
| US6169923B1 (en) | 1999-04-23 | 2001-01-02 | Pacesetter, Inc. | Implantable cardioverter-defibrillator with automatic arrhythmia detection criteria adjustment |
| DE10081288D2 (de) * | 1999-05-17 | 2002-07-11 | Edscha Ag | Fronthaubenanordnung |
| CN1361793A (zh) | 1999-05-19 | 2002-07-31 | 利思进药品公司 | 干扰素-α蛋白作为Fc融合蛋白的表达和运输 |
| US6696424B1 (en) | 1999-05-28 | 2004-02-24 | Vical Incorporated | Cytofectin dimers and methods of use thereof |
| US6346382B1 (en) | 1999-06-01 | 2002-02-12 | Vanderbilt University | Human carbamyl phosphate synthetase I polymorphism and diagnostic methods related thereto |
| EP1202714A1 (en) | 1999-07-16 | 2002-05-08 | Purdue Research Foundation | Vinyl ether lipids with cleavable hydrophilic headgroups |
| PL364816A1 (en) | 1999-07-23 | 2004-12-13 | Genentech, Inc. | Method for rnase- and organic solvent-free plasmid dna purification using tangential flow filtration |
| US6358278B1 (en) | 1999-09-24 | 2002-03-19 | St. Jude Medical, Inc. | Heart valve prosthesis with rotatable cuff |
| US6371983B1 (en) | 1999-10-04 | 2002-04-16 | Ernest Lane | Bioprosthetic heart valve |
| US7060291B1 (en) | 1999-11-24 | 2006-06-13 | Transave, Inc. | Modular targeted liposomal delivery system |
| AU3366901A (en) | 1999-12-30 | 2001-07-16 | Novartis Ag | Novel colloid synthetic vectors for gene therapy |
| EP1261379A2 (en) | 2000-02-17 | 2002-12-04 | Genzyme Corporation | Genetic modification of the lung as a portal for gene delivery |
| US6370434B1 (en) | 2000-02-28 | 2002-04-09 | Cardiac Pacemakers, Inc. | Cardiac lead and method for lead implantation |
| US6565960B2 (en) | 2000-06-01 | 2003-05-20 | Shriners Hospital Of Children | Polymer composite compositions |
| EP1297169B1 (en) | 2000-06-26 | 2012-08-08 | Ethris Gmbh | Method for transfecting cells using a magnetic field |
| IL138474A0 (en) | 2000-09-14 | 2001-10-31 | Epox Ltd | Highly branched water-soluble polyamine oligomers, process for their preparation and applications thereof |
| US6998115B2 (en) | 2000-10-10 | 2006-02-14 | Massachusetts Institute Of Technology | Biodegradable poly(β-amino esters) and uses thereof |
| US7427394B2 (en) | 2000-10-10 | 2008-09-23 | Massachusetts Institute Of Technology | Biodegradable poly(β-amino esters) and uses thereof |
| USRE43612E1 (en) | 2000-10-10 | 2012-08-28 | Massachusetts Institute Of Technology | Biodegradable poly(β-amino esters) and uses thereof |
| AU2002214854A1 (en) | 2000-10-25 | 2002-05-06 | Inex Pharmaceuticals Corporation | Lipid formulations for target delivery |
| GB0028361D0 (en) | 2000-11-21 | 2001-01-03 | Glaxo Group Ltd | Method of separating extra chromosomal dna from other cellular components |
| US20020094528A1 (en) | 2000-11-29 | 2002-07-18 | Salafsky Joshua S. | Method and apparatus using a surface-selective nonlinear optical technique for detection of probe-target interations |
| JP2002167368A (ja) | 2000-12-01 | 2002-06-11 | Nitto Denko Corp | アルキル置換デンドリマーおよびその製造法 |
| US20050004058A1 (en) | 2000-12-07 | 2005-01-06 | Patrick Benoit | Sequences upstream of the carp gene, vectors containing them and uses thereof |
| DE10109897A1 (de) | 2001-02-21 | 2002-11-07 | Novosom Ag | Fakultativ kationische Liposomen und Verwendung dieser |
| US20020192721A1 (en) | 2001-03-28 | 2002-12-19 | Engeneos, Inc. | Modular molecular clasps and uses thereof |
| TW588032B (en) | 2001-04-23 | 2004-05-21 | Shinetsu Chemical Co | New tertiary amine compound having ester structure and method for producing the same |
| US6585410B1 (en) | 2001-05-03 | 2003-07-01 | The United States Of America As Represented By The Administrator Of The National Aeronautics And Space Administration | Radiant temperature nulling radiometer |
| ES2340499T3 (es) | 2001-06-05 | 2010-06-04 | Curevac Gmbh | Arnm de antigeno tumoral estabilizado con un contenido de g/c aumentado. |
| JP4371812B2 (ja) | 2001-09-28 | 2009-11-25 | マックス−プランク−ゲゼルシャフト・ツア・フェルデルング・デア・ヴィッセンシャフテン・エー・ファオ | マイクロrna分子 |
| AU2002340490A1 (en) | 2001-11-09 | 2003-05-19 | Bayer Healthcare Ag | Isotopically coded affinity markers 3 |
| DE10162480A1 (de) | 2001-12-19 | 2003-08-07 | Ingmar Hoerr | Die Applikation von mRNA für den Einsatz als Therapeutikum gegen Tumorerkrankungen |
| DE10207178A1 (de) | 2002-02-19 | 2003-09-04 | Novosom Ag | Komponenten für die Herstellung amphoterer Liposomen |
| DE10214983A1 (de) | 2002-04-04 | 2004-04-08 | TransMIT Gesellschaft für Technologietransfer mbH | Vernebelbare Liposomen und ihre Verwendung zur pulmonalen Applikation von Wirkstoffen |
| US20030215395A1 (en) | 2002-05-14 | 2003-11-20 | Lei Yu | Controllably degradable polymeric biomolecule or drug carrier and method of synthesizing said carrier |
| US7601367B2 (en) | 2002-05-28 | 2009-10-13 | Mirus Bio Llc | Compositions and processes using siRNA, amphipathic compounds and polycations |
| AU2003238791A1 (en) * | 2002-05-31 | 2003-12-19 | Children's Hospital Medical Center | CFTR modifier genes and expressed polypeptides useful in treating cystic fibrosis |
| EP2338478B1 (en) | 2002-06-28 | 2014-07-23 | Protiva Biotherapeutics Inc. | Method for producing liposomes |
| DE10229872A1 (de) | 2002-07-03 | 2004-01-29 | Curevac Gmbh | Immunstimulation durch chemisch modifizierte RNA |
| US20040028804A1 (en) | 2002-08-07 | 2004-02-12 | Anderson Daniel G. | Production of polymeric microarrays |
| JP2005536214A (ja) | 2002-08-22 | 2005-12-02 | セルトラン リミテッド | 細胞培養表面 |
| MXPA05004762A (es) | 2002-11-04 | 2005-08-03 | Ge Bayer Silicones Gmbh & Co | Copolimeros de poliamino y/o poliamonio polisiloxano i lineales. |
| EP1572669A2 (en) | 2002-11-22 | 2005-09-14 | Novo Nordisk A/S | 2,5-diketopiperazines for the treatment of obesity |
| US7169892B2 (en) | 2003-01-10 | 2007-01-30 | Astellas Pharma Inc. | Lipid-peptide-polymer conjugates for long blood circulation and tumor specific drug delivery systems |
| JP4408862B2 (ja) | 2003-01-20 | 2010-02-03 | 旭化成エレクトロニクス株式会社 | ポインティングデバイス |
| JP2006520611A (ja) | 2003-03-05 | 2006-09-14 | セネスコ テクノロジーズ,インコーポレイティド | eIF−5A1の発現を抑制するための、アンチセンス・オリゴヌクレオチド又はsiRNAの使用 |
| US20040224912A1 (en) | 2003-05-07 | 2004-11-11 | Isis Pharmaceuticals Inc. | Modulation of PAI-1 mRNA-binding protein expression |
| US7619017B2 (en) | 2003-05-19 | 2009-11-17 | Wacker Chemical Corporation | Polymer emulsions resistant to biodeterioration |
| US7507859B2 (en) | 2003-06-16 | 2009-03-24 | Fifth Base Llc | Functional synthetic molecules and macromolecules for gene delivery |
| WO2005028619A2 (en) | 2003-09-15 | 2005-03-31 | Massachusetts Institute Of Technology | Nanoliter-scale synthesis of arrayed biomaterials and screening thereof |
| US7803397B2 (en) | 2003-09-15 | 2010-09-28 | Protiva Biotherapeutics, Inc. | Polyethyleneglycol-modified lipid compounds and uses thereof |
| US20050069590A1 (en) | 2003-09-30 | 2005-03-31 | Buehler Gail K. | Stable suspensions for medicinal dosages |
| EA010056B1 (ru) | 2003-10-10 | 2008-06-30 | Паудерджект Вэксинс, Инк. | Конструкции нуклеиновых кислот |
| WO2005037226A2 (en) | 2003-10-17 | 2005-04-28 | Georgia Tech Research Corporation | Genetically engineered enteroendocrine cells for treating glucose-related metabolic disorders |
| CA2543237A1 (en) | 2003-11-10 | 2005-05-19 | Nippon Kayaku Kabushiki Kaisha | Diimonium salt compound and use thereof |
| US7022214B2 (en) | 2004-01-21 | 2006-04-04 | Bio-Rad Laboratories, Inc. | Carrier ampholytes of high pH range |
| US7556684B2 (en) | 2004-02-26 | 2009-07-07 | Construction Research & Technology Gmbh | Amine containing strength improvement admixture |
| US20060228404A1 (en) | 2004-03-04 | 2006-10-12 | Anderson Daniel G | Compositions and methods for treatment of hypertrophic tissues |
| WO2005118874A1 (en) | 2004-06-04 | 2005-12-15 | Wyeth | Enhancing protein expression |
| WO2005120152A2 (en) | 2004-06-07 | 2005-12-22 | Protiva Biotherapeutics, Inc. | Cationic lipids and methods of use |
| EP1766035B1 (en) | 2004-06-07 | 2011-12-07 | Protiva Biotherapeutics Inc. | Lipid encapsulated interfering rna |
| US7670595B2 (en) | 2004-06-28 | 2010-03-02 | Merck Patent Gmbh | Fc-interferon-beta fusion proteins |
| GB0418172D0 (en) | 2004-08-13 | 2004-09-15 | Ic Vec Ltd | Vector |
| DE102004042546A1 (de) | 2004-09-02 | 2006-03-09 | Curevac Gmbh | Kombinationstherapie zur Immunstimulation |
| DE102004043342A1 (de) | 2004-09-08 | 2006-03-09 | Bayer Materialscience Ag | Blockierte Polyurethan-Prepolymere als Klebstoffe |
| CA2586708A1 (en) | 2004-11-05 | 2006-05-11 | Novosom Ag | Improvements in or relating to pharmaceutical compositions comprising an oligonucleotide as an active agent |
| GB0502482D0 (en) | 2005-02-07 | 2005-03-16 | Glaxo Group Ltd | Novel compounds |
| CA2597724A1 (en) | 2005-02-14 | 2007-08-02 | Sirna Therapeutics, Inc. | Cationic lipids and formulated molecular compositions containing them |
| EP1869106B1 (en) | 2005-03-28 | 2014-06-25 | Dendritic Nanotechnologies, Inc. | Janus dendrimers and dendrons |
| EP1912679A4 (en) | 2005-06-15 | 2009-07-29 | Massachusetts Inst Technology | AMINOUS LIPIDS AND ITS USES |
| US9012219B2 (en) | 2005-08-23 | 2015-04-21 | The Trustees Of The University Of Pennsylvania | RNA preparations comprising purified modified RNA for reprogramming cells |
| EP4174179B1 (en) * | 2005-08-23 | 2025-05-07 | The Trustees of the University of Pennsylvania | Rna containing modified nucleosides and methods of use thereof |
| WO2007031091A2 (en) | 2005-09-15 | 2007-03-22 | Santaris Pharma A/S | Rna antagonist compounds for the modulation of p21 ras expression |
| CN101346393B (zh) | 2005-11-02 | 2015-07-22 | 普洛体维生物治疗公司 | 修饰的siRNA分子及其应用 |
| US7238791B1 (en) | 2005-12-16 | 2007-07-03 | Roche Diagnostics Operations, Inc. | 6-monoacetylmorphine derivatives useful in immunoassay |
| US20090221684A1 (en) | 2005-12-22 | 2009-09-03 | Trustees Of Boston University | Molecules for Gene Delivery and Gene Therapy, and Methods of Use Thereof |
| CN100569877C (zh) | 2005-12-30 | 2009-12-16 | 财团法人工业技术研究院 | 含多uv交联反应基的分枝状结构化合物及其应用 |
| CA2649325C (en) | 2006-04-14 | 2019-01-08 | Epicentre Technologies Corporation | Kits and methods for generating 5' capped rna |
| US9085778B2 (en) | 2006-05-03 | 2015-07-21 | VL27, Inc. | Exosome transfer of nucleic acids to cells |
| BRPI0711950A2 (pt) * | 2006-05-19 | 2011-12-13 | Scripps Research Inst | tratamento para o dobramento inadequado da proteìna |
| US20070275923A1 (en) | 2006-05-25 | 2007-11-29 | Nastech Pharmaceutical Company Inc. | CATIONIC PEPTIDES FOR siRNA INTRACELLULAR DELIVERY |
| EP2032652A4 (en) | 2006-06-05 | 2011-08-17 | Massachusetts Inst Technology | NETWORKED DEVELOPABLE POLYMERS AND ITS USE |
| US20090186805A1 (en) | 2006-07-06 | 2009-07-23 | Aaron Thomas Tabor | Compositions and Methods for Genetic Modification of Cells Having Cosmetic Function to Enhance Cosmetic Appearance |
| ES2293834B1 (es) | 2006-07-20 | 2009-02-16 | Consejo Superior Investig. Cientificas | Compuesto con actividad inhibidora de las interacciones ubc13-uev, composiciones farmaceuticas que lo comprenden y sus aplicaciones terapeuticas. |
| WO2008011561A2 (en) | 2006-07-21 | 2008-01-24 | Massachusetts Institute Of Technology | End-modified poly(beta-amino esters) and uses thereof |
| AU2007303205A1 (en) | 2006-10-03 | 2008-04-10 | Tekmira Pharmaceuticals Corporation | Lipid containing formulations |
| DE102006051516A1 (de) | 2006-10-31 | 2008-05-08 | Curevac Gmbh | (Basen-)modifizierte RNA zur Expressionssteigerung eines Proteins |
| EP2104739B1 (en) | 2006-12-21 | 2013-06-19 | Novozymes Inc. | Modified messenger rna stabilizing sequences for expressing genes in bacterial cells |
| DE102007001370A1 (de) | 2007-01-09 | 2008-07-10 | Curevac Gmbh | RNA-kodierte Antikörper |
| US8859229B2 (en) | 2007-02-02 | 2014-10-14 | Yale University | Transient transfection with RNA |
| EP2139461A2 (en) | 2007-03-20 | 2010-01-06 | Recepticon Aps | Amino derivatives to prevent nephrotoxicity and cancer |
| EP2141985B1 (en) * | 2007-03-28 | 2018-10-17 | University of Iowa Research Foundation | Transgenic pig models of cystic fibrosis |
| JP5186126B2 (ja) | 2007-03-29 | 2013-04-17 | 公益財団法人地球環境産業技術研究機構 | 新規トリアジン誘導体ならびにその製法およびそのガス分離膜としての用途 |
| JP5480804B2 (ja) | 2007-04-18 | 2014-04-23 | コーナーストーン ファーマシューティカルズ,インコーポレーテッド | リポ酸誘導体を含有する医薬製剤 |
| US8678686B2 (en) | 2007-05-01 | 2014-03-25 | Pgr-Solutions | Multi-chain lipophilic polyamines |
| US20090163705A1 (en) | 2007-05-21 | 2009-06-25 | Alnylam Pharmaceuticals, Inc. | Cationic lipids |
| WO2009030254A1 (en) | 2007-09-04 | 2009-03-12 | Curevac Gmbh | Complexes of rna and cationic peptides for transfection and for immunostimulation |
| US8389238B2 (en) | 2007-09-12 | 2013-03-05 | Copernicus Therapeutics, Inc. | Long-term in vivo transgene expression |
| WO2009046220A2 (en) | 2007-10-02 | 2009-04-09 | Mdrna, Inc. | Lipopeptides for delivery of nucleic acids |
| WO2009046739A1 (en) | 2007-10-09 | 2009-04-16 | Curevac Gmbh | Composition for treating prostate cancer (pca) |
| WO2009066758A1 (ja) | 2007-11-22 | 2009-05-28 | Japan Science And Technology Agency | 低分子rnaによる細胞または人工細胞モデルでの翻訳制御システム |
| JP5635412B2 (ja) | 2007-12-04 | 2014-12-03 | アルニラム ファーマスーティカルズ インコーポレイテッドAlnylam Pharmaceuticals, Inc. | 標的化脂質 |
| EP2238251B1 (en) | 2007-12-27 | 2015-02-11 | Protiva Biotherapeutics Inc. | Silencing of polo-like kinase expression using interfering rna |
| WO2009120247A2 (en) | 2007-12-27 | 2009-10-01 | The Ohio State University Research Foundation | Lipid nanoparticle compositions and methods of making and using the same |
| CA2711236A1 (en) | 2008-01-02 | 2009-07-16 | Alnylam Pharmaceuticals, Inc. | Screening method for selected amino lipid-containing compositions |
| EP2274425A2 (en) | 2008-04-11 | 2011-01-19 | Alnylam Pharmaceuticals Inc. | Site-specific delivery of nucleic acids by combining targeting ligands with endosomolytic components |
| AU2009238175C1 (en) | 2008-04-15 | 2023-11-30 | Arbutus Biopharma Corporation | Novel lipid formulations for nucleic acid delivery |
| WO2009127230A1 (en) | 2008-04-16 | 2009-10-22 | Curevac Gmbh | MODIFIED (m)RNA FOR SUPPRESSING OR AVOIDING AN IMMUNOSTIMULATORY RESPONSE AND IMMUNOSUPPRESSIVE COMPOSITION |
| US20090263407A1 (en) | 2008-04-16 | 2009-10-22 | Abbott Laboratories | Cationic Lipids and Uses Thereof |
| US8222221B2 (en) | 2008-06-04 | 2012-07-17 | The Board Of Regents Of The University Of Texas System | Modulation of gene expression through endogenous small RNA targeting of gene promoters |
| JP5024216B2 (ja) | 2008-07-23 | 2012-09-12 | トヨタ自動車株式会社 | 内燃機関の点火時期制御装置及び点火時期制御方法 |
| US20100035249A1 (en) | 2008-08-05 | 2010-02-11 | Kabushiki Kaisha Dnaform | Rna sequencing and analysis using solid support |
| US8404198B2 (en) | 2008-08-27 | 2013-03-26 | Life Technologies Corporation | Apparatus for and method of processing biological samples |
| WO2010037408A1 (en) | 2008-09-30 | 2010-04-08 | Curevac Gmbh | Composition comprising a complexed (m)rna and a naked mrna for providing or enhancing an immunostimulatory response in a mammal and uses thereof |
| EP2350043B9 (en) | 2008-10-09 | 2014-08-20 | TEKMIRA Pharmaceuticals Corporation | Improved amino lipids and methods for the delivery of nucleic acids |
| KR20110071017A (ko) | 2008-10-16 | 2011-06-27 | 마리나 바이오테크, 인크. | 유전자 침묵 치료제의 리포좀에 의한 효율적인 전달을 위한 프로세스 및 조성물 |
| US9080211B2 (en) | 2008-10-24 | 2015-07-14 | Epicentre Technologies Corporation | Transposon end compositions and methods for modifying nucleic acids |
| US20120009222A1 (en) | 2008-10-27 | 2012-01-12 | Massachusetts Institute Of Technology | Modulation of the immune response |
| MX353900B (es) | 2008-11-07 | 2018-02-01 | Massachusetts Inst Technology | Lipidoides de aminoalcohol y usos de los mismos. |
| CN111909020A (zh) | 2008-11-10 | 2020-11-10 | 阿布特斯生物制药公司 | 用于递送治疗剂的脂质和组合物 |
| EP2350296A4 (en) | 2008-11-17 | 2013-04-03 | Enzon Pharmaceuticals Inc | BRANCHED CATIONIC LIPIDS FOR THE SYSTEM FOR INTRODUCING NUCLEIC ACIDS |
| CA2750561C (en) | 2009-01-26 | 2017-10-10 | Protiva Biotherapeutics, Inc. | Compositions and methods for silencing apolipoprotein c-iii expression |
| US20100222489A1 (en) | 2009-02-27 | 2010-09-02 | Jiang Dayue D | Copolymer composition, membrane article, and methods thereof |
| CA2754043A1 (en) | 2009-03-12 | 2010-09-16 | Alnylam Pharmaceuticals, Inc. | Lipid formulated compositions and methods for inhibiting expression of eg5 and vegf genes |
| CN102334237B (zh) | 2009-04-02 | 2015-05-13 | 西蒙公司 | 电信接线板 |
| DK2419144T3 (da) | 2009-04-17 | 2019-10-21 | Univ Oxford Innovation Ltd | Sammensætning til levering af genetisk materiale |
| US9220682B2 (en) | 2009-04-22 | 2015-12-29 | Emory University | Nanocarrier therapy for treating invasive tumors |
| KR20240015723A (ko) | 2009-05-05 | 2024-02-05 | 알닐람 파마슈티칼스 인코포레이티드 | 지질 조성물 |
| CA2764609C (en) | 2009-06-10 | 2018-10-02 | Alnylam Pharmaceuticals, Inc. | Improved cationic lipid of formula i |
| WO2010147992A1 (en) | 2009-06-15 | 2010-12-23 | Alnylam Pharmaceuticals, Inc. | Methods for increasing efficacy of lipid formulated sirna |
| CN102458366B (zh) | 2009-06-15 | 2015-02-11 | 阿尔尼拉姆医药品有限公司 | 靶向pcsk9基因的脂质配制的dsrna |
| US9018187B2 (en) | 2009-07-01 | 2015-04-28 | Protiva Biotherapeutics, Inc. | Cationic lipids and methods for the delivery of therapeutic agents |
| EP2449106B1 (en) | 2009-07-01 | 2015-04-08 | Protiva Biotherapeutics Inc. | Compositions and methods for silencing apolipoprotein b |
| WO2011000106A1 (en) | 2009-07-01 | 2011-01-06 | Protiva Biotherapeutics, Inc. | Improved cationic lipids and methods for the delivery of therapeutic agents |
| US8716464B2 (en) | 2009-07-20 | 2014-05-06 | Thomas W. Geisbert | Compositions and methods for silencing Ebola virus gene expression |
| NZ598125A (en) | 2009-07-30 | 2014-05-30 | Salvat Lab Sa | Apaf-1 inhibitor compounds |
| DE102009043342A1 (de) | 2009-09-29 | 2011-03-31 | Bayer Technology Services Gmbh | Stoffe für selbstorganisierte Träger zur kontrollierten Freisetzung eines Wirkstoffs |
| ES2666559T3 (es) | 2009-12-01 | 2018-05-07 | Translate Bio, Inc. | Entrega del mrna para la aumentación de proteínas y enzimas en enfermedades genéticas humanas |
| KR102171849B1 (ko) | 2009-12-07 | 2020-10-30 | 더 트러스티스 오브 더 유니버시티 오브 펜실바니아 | 세포 리프로그래밍을 위한 정제된 변형 rna를 포함하는 rna 제제 |
| WO2011069529A1 (en) | 2009-12-09 | 2011-06-16 | Curevac Gmbh | Mannose-containing solution for lyophilization, transfection and/or injection of nucleic acids |
| WO2011075656A1 (en) | 2009-12-18 | 2011-06-23 | The University Of British Columbia | Methods and compositions for delivery of nucleic acids |
| EP2338520A1 (de) | 2009-12-21 | 2011-06-29 | Ludwig Maximilians Universität | Konjugat mit Zielfindungsligand und dessen Verwendung |
| KR101762466B1 (ko) | 2009-12-23 | 2017-07-27 | 노파르티스 아게 | 지질, 지질 조성물 및 이의 사용 방법 |
| WO2011141705A1 (en) | 2010-05-12 | 2011-11-17 | Protiva Biotherapeutics, Inc. | Novel cationic lipids and methods of use thereof |
| ES3053320T3 (en) | 2010-06-03 | 2026-01-21 | Alnylam Pharmaceuticals Inc | Biodegradable lipids for the delivery of active agents |
| CN101863544B (zh) | 2010-06-29 | 2011-09-28 | 湖南科技大学 | 一种氰尿酸基重金属螯合絮凝剂及其制备方法 |
| US9006417B2 (en) | 2010-06-30 | 2015-04-14 | Protiva Biotherapeutics, Inc. | Non-liposomal systems for nucleic acid delivery |
| WO2012016184A2 (en) | 2010-07-30 | 2012-02-02 | Alnylam Pharmaceuticals, Inc. | Methods and compositions for delivery of active agents |
| EP2600901B1 (en) | 2010-08-06 | 2019-03-27 | ModernaTX, Inc. | A pharmaceutical formulation comprising engineered nucleic acids and medical use thereof |
| WO2012019630A1 (en) | 2010-08-13 | 2012-02-16 | Curevac Gmbh | Nucleic acid comprising or coding for a histone stem-loop and a poly(a) sequence or a polyadenylation signal for increasing the expression of an encoded protein |
| EP2609135A4 (en) | 2010-08-26 | 2015-05-20 | Massachusetts Inst Technology | POLY (BETA AMINO ALCOHOLS), THEIR PREPARATION AND USES THEREOF |
| CN104531812A (zh) | 2010-10-01 | 2015-04-22 | 现代治疗公司 | 设计核酸及其使用方法 |
| WO2012075040A2 (en) | 2010-11-30 | 2012-06-07 | Shire Human Genetic Therapies, Inc. | mRNA FOR USE IN TREATMENT OF HUMAN GENETIC DISEASES |
| WO2012113413A1 (en) | 2011-02-21 | 2012-08-30 | Curevac Gmbh | Vaccine composition comprising complexed immunostimulatory nucleic acids and antigens packaged with disulfide-linked polyethyleneglycol/peptide conjugates |
| DK2691443T3 (da) | 2011-03-28 | 2021-05-03 | Massachusetts Inst Technology | Konjugerede lipomerer og anvendelser af disse |
| US8710200B2 (en) | 2011-03-31 | 2014-04-29 | Moderna Therapeutics, Inc. | Engineered nucleic acids encoding a modified erythropoietin and their expression |
| WO2012133737A1 (ja) | 2011-03-31 | 2012-10-04 | 公益財団法人地球環境産業技術研究機構 | 架橋性アミン化合物、該化合物を用いた高分子膜及びその製造方法 |
| WO2012151503A2 (en) | 2011-05-04 | 2012-11-08 | The Broad Institute, Inc. | Multiplexed genetic reporter assays and compositions |
| WO2012158736A1 (en) | 2011-05-17 | 2012-11-22 | modeRNA Therapeutics | Engineered nucleic acids and methods of use thereof for non-human vertebrates |
| EP2532649B1 (en) | 2011-06-07 | 2015-04-08 | Incella GmbH | Amino lipids, their synthesis and uses thereof |
| ES2663360T3 (es) | 2011-06-08 | 2018-04-12 | Translate Bio, Inc. | Lípidos escindibles |
| BR112013031553A2 (pt) | 2011-06-08 | 2020-11-10 | Shire Human Genetic Therapies, Inc. | composições, mrna que codifica para uma hgla e seu uso, uso de pelo menos uma molécula de mrna e um veículo de transferência e uso de um mrna que codifica para proteína exógena |
| WO2013003475A1 (en) | 2011-06-27 | 2013-01-03 | Cellscript, Inc. | Inhibition of innate immune response |
| US9464124B2 (en) | 2011-09-12 | 2016-10-11 | Moderna Therapeutics, Inc. | Engineered nucleic acids and methods of use thereof |
| EP2755986A4 (en) | 2011-09-12 | 2015-05-20 | Moderna Therapeutics Inc | MANIPULATED NUCLEIC ACIDS AND METHOD OF APPLICATION THEREFOR |
| WO2013039861A2 (en) | 2011-09-12 | 2013-03-21 | modeRNA Therapeutics | Engineered nucleic acids and methods of use thereof |
| BR112014007852B1 (pt) | 2011-10-03 | 2021-11-03 | Moderna Therapeutics, Inc | Polinucleotídeo isolado modificado e composição farmacêutica |
| CN104114571A (zh) | 2011-10-05 | 2014-10-22 | 普洛体维生物治疗公司 | 用于沉默醛脱氢酶的组合物和方法 |
| NZ747501A (en) | 2011-10-27 | 2020-05-29 | Massachusetts Inst Technology | Amino acid derivatives functionalized on the n-terminal capable of forming drug encapsulating microspheres |
| US20140343129A1 (en) | 2011-12-14 | 2014-11-20 | Moderna Therapeutics, Inc. | Modified nucleic acids, and acute care uses thereof |
| EP2791364A4 (en) | 2011-12-14 | 2015-11-11 | Moderna Therapeutics Inc | PROCESS FOR RESPONSE TO A BIOLOGICAL THREAT |
| PL2791160T3 (pl) | 2011-12-16 | 2022-06-20 | Modernatx, Inc. | Kompozycje zmodyfikowanego mrna |
| JP2015510495A (ja) | 2011-12-21 | 2015-04-09 | モデルナ セラピューティクス インコーポレイテッドModerna Therapeutics,Inc. | 器官または器官移植片の生存可能性または寿命を延長する方法 |
| EP2797634A4 (en) | 2011-12-29 | 2015-08-05 | Moderna Therapeutics Inc | MODIFIED MRNAS FOR THE CODING OF CELL-PENETRATING POLYPEPTIDES |
| EP2798064B1 (en) | 2011-12-30 | 2016-08-31 | Cellscript, Llc | Making and using in vitro-synthesized ssrna for introducing into mammalian cells to induce a biological or biochemical effect |
| WO2013120497A1 (en) | 2012-02-15 | 2013-08-22 | Curevac Gmbh | Nucleic acid comprising or coding for a histone stem-loop and a poly(a) sequence or a polyadenylation signal for increasing the expression of an encoded therapeutic protein |
| RS58077B1 (sr) | 2012-02-24 | 2019-02-28 | Arbutus Biopharma Corp | Trialkil katjonski lipidi i postupci njihove primene |
| BR112014024131A2 (pt) | 2012-03-29 | 2017-07-25 | Shire Human Genetic Therapies | lipídios catiônicos ionizáveis |
| JP6211054B2 (ja) | 2012-03-29 | 2017-10-11 | シャイアー ヒューマン ジェネティック セラピーズ インコーポレイテッド | 脂質誘導される中性ナノ粒子 |
| HK1206779A1 (en) | 2012-04-02 | 2016-01-15 | Modernatx, Inc. | Modified polynucleotides for the production of nuclear proteins |
| US10501512B2 (en) | 2012-04-02 | 2019-12-10 | Modernatx, Inc. | Modified polynucleotides |
| US20150050354A1 (en) | 2012-04-02 | 2015-02-19 | Moderna Therapeutics, Inc. | Modified polynucleotides for the treatment of otic diseases and conditions |
| US20140275229A1 (en) | 2012-04-02 | 2014-09-18 | Moderna Therapeutics, Inc. | Modified polynucleotides encoding udp glucuronosyltransferase 1 family, polypeptide a1 |
| AU2013243949A1 (en) | 2012-04-02 | 2014-10-30 | Moderna Therapeutics, Inc. | Modified polynucleotides for the production of biologics and proteins associated with human disease |
| US9283287B2 (en) | 2012-04-02 | 2016-03-15 | Moderna Therapeutics, Inc. | Modified polynucleotides for the production of nuclear proteins |
| US9572897B2 (en) | 2012-04-02 | 2017-02-21 | Modernatx, Inc. | Modified polynucleotides for the production of cytoplasmic and cytoskeletal proteins |
| CA2873274C (en) * | 2012-06-08 | 2021-06-01 | Ethris Gmbh | Pulmonary delivery of messenger rna |
| EP2859102A4 (en) | 2012-06-08 | 2016-05-11 | Shire Human Genetic Therapies | NUCLEASE RESISTANT POLYNUCLEOTIDES AND USES THEREOF |
| CA2876155C (en) * | 2012-06-08 | 2022-12-13 | Ethris Gmbh | Pulmonary delivery of mrna to non-lung target cells |
| CA2884870C (en) | 2012-08-13 | 2022-03-29 | Massachusetts Institute Of Technology | Amine-containing lipidoids and uses thereof |
| WO2014093924A1 (en) | 2012-12-13 | 2014-06-19 | Moderna Therapeutics, Inc. | Modified nucleic acid molecules and uses thereof |
| DK2922554T3 (en) | 2012-11-26 | 2022-05-23 | Modernatx Inc | Terminalt modificeret rna |
| EP2929035A1 (en) | 2012-12-07 | 2015-10-14 | Shire Human Genetic Therapies, Inc. | Lipidic nanoparticles for mrna delivering |
| US20150315541A1 (en) | 2012-12-13 | 2015-11-05 | Moderna Therapeutics, Inc. | Modified polynucleotides for altering cell phenotype |
| AU2013374345A1 (en) | 2013-01-17 | 2015-08-06 | Moderna Therapeutics, Inc. | Signal-sensor polynucleotides for the alteration of cellular phenotypes |
| EP2968397A4 (en) | 2013-03-12 | 2016-12-28 | Moderna Therapeutics Inc | DIAGNOSIS AND TREATMENT OF FIBROSIS |
| EP2968391A1 (en) | 2013-03-13 | 2016-01-20 | Moderna Therapeutics, Inc. | Long-lived polynucleotide molecules |
| EP3495505B1 (en) | 2013-03-14 | 2024-09-11 | Translate Bio, Inc. | Quantitative assessment for cap efficiency of messenger rna |
| HUE055044T2 (hu) | 2013-03-14 | 2021-10-28 | Translate Bio Inc | MRNS-kódolt ellenanyagok bejuttatására szolgáló eljárások és készítmények |
| EP2971010B1 (en) | 2013-03-14 | 2020-06-10 | ModernaTX, Inc. | Formulation and delivery of modified nucleoside, nucleotide, and nucleic acid compositions |
| EP4446413A3 (en) | 2013-03-14 | 2024-12-18 | Translate Bio, Inc. | Methods for purification of messenger rna |
| US20160184458A1 (en) | 2013-03-14 | 2016-06-30 | Shire Human Genetic Therapies, Inc. | Mrna therapeutic compositions and use to treat diseases and disorders |
| EP3301102B1 (en) | 2013-03-14 | 2020-04-15 | Translate Bio, Inc. | Ribonucleic acids with 4'-thio-modified nucleotides and related methods |
| CA2903487A1 (en) | 2013-03-14 | 2014-09-25 | Shire Human Genetic Therapies, Inc. | Quantitative assessment for cap efficiency of messenger rna |
| WO2014153052A2 (en) | 2013-03-14 | 2014-09-25 | Shire Human Genetic Therapies, Inc. | Cftr mrna compositions and related methods and uses |
| ES2795249T3 (es) | 2013-03-15 | 2020-11-23 | Translate Bio Inc | Mejora sinérgica de la administración de ácidos nucleicos a través de formulaciones mezcladas |
| US10077439B2 (en) | 2013-03-15 | 2018-09-18 | Modernatx, Inc. | Removal of DNA fragments in mRNA production process |
| US20160032273A1 (en) | 2013-03-15 | 2016-02-04 | Moderna Therapeutics, Inc. | Characterization of mrna molecules |
| WO2014144711A1 (en) | 2013-03-15 | 2014-09-18 | Moderna Therapeutics, Inc. | Analysis of mrna heterogeneity and stability |
| US11377470B2 (en) | 2013-03-15 | 2022-07-05 | Modernatx, Inc. | Ribonucleic acid purification |
| WO2014144767A1 (en) | 2013-03-15 | 2014-09-18 | Moderna Therapeutics, Inc. | Ion exchange purification of mrna |
| US10138507B2 (en) | 2013-03-15 | 2018-11-27 | Modernatx, Inc. | Manufacturing methods for production of RNA transcripts |
| US8980864B2 (en) | 2013-03-15 | 2015-03-17 | Moderna Therapeutics, Inc. | Compositions and methods of altering cholesterol levels |
| WO2014179562A1 (en) | 2013-05-01 | 2014-11-06 | Massachusetts Institute Of Technology | 1,3,5-triazinane-2,4,6-trione derivatives and uses thereof |
| WO2014210356A1 (en) | 2013-06-26 | 2014-12-31 | Massachusetts Institute Of Technology | Multi-tailed lipids and uses thereof |
| PT3019619T (pt) | 2013-07-11 | 2021-11-11 | Modernatx Inc | Composições que compreendem polinucleótidos sintéticos que codificam proteínas relacionadas com crispr e sgarn sintéticos e métodos de utilização |
| CN105555757A (zh) | 2013-07-23 | 2016-05-04 | 普洛体维生物治疗公司 | 用于递送信使rna的组合物和方法 |
| US20160194625A1 (en) | 2013-09-03 | 2016-07-07 | Moderna Therapeutics, Inc. | Chimeric polynucleotides |
| EP3041938A1 (en) | 2013-09-03 | 2016-07-13 | Moderna Therapeutics, Inc. | Circular polynucleotides |
| US10023626B2 (en) | 2013-09-30 | 2018-07-17 | Modernatx, Inc. | Polynucleotides encoding immune modulating polypeptides |
| EP3052479A4 (en) | 2013-10-02 | 2017-10-25 | Moderna Therapeutics, Inc. | Polynucleotide molecules and uses thereof |
| EP3052511A4 (en) | 2013-10-02 | 2017-05-31 | Moderna Therapeutics, Inc. | Polynucleotide molecules and uses thereof |
| CA2927393A1 (en) | 2013-10-18 | 2015-04-23 | Moderna Therapeutics, Inc. | Compositions and methods for tolerizing cellular systems |
| EP4036241A1 (en) | 2013-10-22 | 2022-08-03 | Translate Bio, Inc. | Cns delivery of mrna and uses thereof |
| CN105658242A (zh) | 2013-10-22 | 2016-06-08 | 夏尔人类遗传性治疗公司 | 用于苯丙酮尿症的mrna疗法 |
| ES2707966T3 (es) | 2013-10-22 | 2019-04-08 | Translate Bio Inc | Terapia de ARNm para la deficiencia en síntesis de argininosuccinato |
| WO2015085318A2 (en) | 2013-12-06 | 2015-06-11 | Moderna Therapeutics, Inc. | Targeted adaptive vaccines |
| EP3053585A1 (en) | 2013-12-13 | 2016-08-10 | Moderna Therapeutics, Inc. | Alternative nucleic acid molecules and uses thereof |
| KR102399799B1 (ko) | 2013-12-30 | 2022-05-18 | 큐어백 아게 | 인공 핵산 분자 |
| US20170002060A1 (en) | 2014-01-08 | 2017-01-05 | Moderna Therapeutics, Inc. | Polynucleotides for the in vivo production of antibodies |
| ES2754239T3 (es) | 2014-03-12 | 2020-04-16 | Curevac Ag | Combinación de vacunación y agonistas de OX40 |
| PL3981437T3 (pl) | 2014-04-23 | 2025-02-24 | Modernatx, Inc. | Szczepionki z kwasem nukleinowym |
| JP2017536092A (ja) | 2014-10-02 | 2017-12-07 | プロティバ バイオセラピューティクス インコーポレイテッド | B型肝炎ウイルスの遺伝子発現をサイレンシングするための組成物及び方法 |
| WO2016071857A1 (en) | 2014-11-07 | 2016-05-12 | Protiva Biotherapeutics, Inc. | Compositions and methods for silencing ebola virus expression |
| EP3461904A1 (en) | 2014-11-10 | 2019-04-03 | ModernaTX, Inc. | Alternative nucleic acid molecules containing reduced uracil content and uses thereof |
| WO2016077123A1 (en) | 2014-11-10 | 2016-05-19 | Moderna Therapeutics, Inc. | Multiparametric nucleic acid optimization |
| EP3247363A4 (en) | 2015-01-21 | 2018-10-03 | Moderna Therapeutics, Inc. | Lipid nanoparticle compositions |
| WO2016118725A1 (en) | 2015-01-23 | 2016-07-28 | Moderna Therapeutics, Inc. | Lipid nanoparticle compositions |
| WO2016154127A2 (en) | 2015-03-20 | 2016-09-29 | Protiva Biotherapeutics, Inc. | Compositions and methods for treating hypertriglyceridemia |
| WO2016164762A1 (en) | 2015-04-08 | 2016-10-13 | Moderna Therapeutics, Inc. | Polynucleotides encoding low density lipoprotein receptor egf-a and intracellular domain mutants and methods of using the same |
| WO2016183366A2 (en) | 2015-05-12 | 2016-11-17 | Protiva Biotherapeutics, Inc. | Compositions and methods for silencing expression of hepatitis d virus rna |
| WO2016197133A1 (en) | 2015-06-04 | 2016-12-08 | Protiva Biotherapeutics, Inc. | Delivering crispr therapeutics with lipid nanoparticles |
| WO2016197132A1 (en) | 2015-06-04 | 2016-12-08 | Protiva Biotherapeutics Inc. | Treating hepatitis b virus infection using crispr |
| WO2016201377A1 (en) | 2015-06-10 | 2016-12-15 | Moderna Therapeutics, Inc. | Targeted adaptive vaccines |
| CN108350455A (zh) | 2015-07-29 | 2018-07-31 | 阿布特斯生物制药公司 | 用于使b型肝炎病毒基因表达沉默的组合物和方法 |
| AU2016324463B2 (en) | 2015-09-17 | 2022-10-27 | Modernatx, Inc. | Polynucleotides containing a stabilizing tail region |
| WO2017049286A1 (en) | 2015-09-17 | 2017-03-23 | Moderna Therapeutics, Inc. | Polynucleotides containing a morpholino linker |
| US20190054112A1 (en) | 2015-09-18 | 2019-02-21 | Moderna Therapeutics, Inc. | Polynucleotide formulations for use in the treatment of renal diseases |
| KR20210021943A (ko) | 2018-04-25 | 2021-03-02 | 에트리스 게엠베하 | 미립자 제형용 동결 방지제 |
-
2014
- 2014-03-14 WO PCT/US2014/028849 patent/WO2014153052A2/en not_active Ceased
- 2014-03-14 UA UAA201508754A patent/UA117008C2/uk unknown
- 2014-03-14 EP EP14718284.4A patent/EP2968586B1/en active Active
- 2014-03-14 PL PL14718284T patent/PL2968586T3/pl unknown
- 2014-03-14 KR KR1020217031812A patent/KR20210122917A/ko not_active Ceased
- 2014-03-14 LT LTEP14718284.4T patent/LT2968586T/lt unknown
- 2014-03-14 HK HK16105865.3A patent/HK1218068A1/zh unknown
- 2014-03-14 PT PT14718284T patent/PT2968586T/pt unknown
- 2014-03-14 SM SM20180546T patent/SMT201800546T1/it unknown
- 2014-03-14 AU AU2014236305A patent/AU2014236305B2/en active Active
- 2014-03-14 CN CN202210265261.0A patent/CN115154620A/zh active Pending
- 2014-03-14 MX MX2019006294A patent/MX393573B/es unknown
- 2014-03-14 HU HUE14718284A patent/HUE042640T2/hu unknown
- 2014-03-14 HR HRP20181580TT patent/HRP20181580T1/hr unknown
- 2014-03-14 PE PE2015001986A patent/PE20151773A1/es active IP Right Grant
- 2014-03-14 KR KR1020157023977A patent/KR102248744B1/ko active Active
- 2014-03-14 ES ES14718284.4T patent/ES2689523T3/es active Active
- 2014-03-14 NZ NZ751462A patent/NZ751462A/en unknown
- 2014-03-14 IL IL290953A patent/IL290953B2/en unknown
- 2014-03-14 SI SI201430888T patent/SI2968586T1/sl unknown
- 2014-03-14 EA EA201591477A patent/EA037922B1/ru unknown
- 2014-03-14 EA EA202190410A patent/EA202190410A1/ru unknown
- 2014-03-14 CN CN201480015043.5A patent/CN105142676B/zh active Active
- 2014-03-14 CA CA2904151A patent/CA2904151C/en active Active
- 2014-03-14 IL IL305374A patent/IL305374A/en unknown
- 2014-03-14 DK DK14718284.4T patent/DK2968586T3/en active
- 2014-03-14 MX MX2015012333A patent/MX365409B/es active IP Right Grant
- 2014-03-14 BR BR112015022868-2A patent/BR112015022868B1/pt active IP Right Grant
- 2014-03-14 SG SG11201507391VA patent/SG11201507391VA/en unknown
- 2014-03-14 JP JP2016502924A patent/JP6316930B2/ja active Active
- 2014-03-14 EP EP18185286.4A patent/EP3446712A1/en active Pending
- 2014-03-14 RS RS20181158A patent/RS57739B1/sr unknown
- 2014-03-14 KR KR1020217012925A patent/KR102311614B1/ko active Active
- 2014-03-14 SG SG10201710253QA patent/SG10201710253QA/en unknown
- 2014-06-17 US US14/307,322 patent/US9181321B2/en active Active
-
2015
- 2015-09-01 IL IL240982A patent/IL240982B/en active IP Right Grant
- 2015-09-14 CL CL2015002675A patent/CL2015002675A1/es unknown
- 2015-10-06 US US14/876,071 patent/US9713626B2/en active Active
-
2017
- 2017-06-16 US US15/625,648 patent/US10420791B2/en active Active
-
2018
- 2018-03-28 JP JP2018062010A patent/JP6738366B2/ja active Active
- 2018-10-24 CY CY181101097T patent/CY1120790T1/el unknown
- 2018-11-13 IL IL262984A patent/IL262984B/en active IP Right Grant
-
2019
- 2019-04-15 AU AU2019202582A patent/AU2019202582B2/en active Active
- 2019-08-14 US US16/540,960 patent/US11510937B2/en active Active
-
2020
- 2020-07-17 JP JP2020122566A patent/JP2020169218A/ja not_active Withdrawn
-
2021
- 2021-04-20 IL IL282453A patent/IL282453B/en unknown
- 2021-08-09 AU AU2021215095A patent/AU2021215095B2/en active Active
- 2021-12-10 JP JP2021200634A patent/JP7236525B2/ja active Active
-
2022
- 2022-10-24 US US18/049,210 patent/US20230302039A1/en active Pending
-
2023
- 2023-02-27 JP JP2023028264A patent/JP2023054353A/ja active Pending
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2010506838A (ja) * | 2006-10-12 | 2010-03-04 | コペルニクス セラピューティクス インコーポレイテッド | コドン最適化cftr |
| WO2011012316A2 (de) * | 2009-07-31 | 2011-02-03 | Ludwig-Maximilians-Universität | Rna mit einer kombination aus unmodifizierten und modifizierten nucleotiden zur proteinexpression |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7236525B2 (ja) | CFTR mRNA組成物ならびに関連する方法及び使用 | |
| HK40005353A (en) | Cftr mrna compositions and related methods and uses | |
| HK1220137B (en) | Cftr mrna compositions and related methods and uses | |
| EA047691B1 (ru) | КОМПОЗИЦИИ mPHK CFTR И СВЯЗАННЫЕ С НИМИ СПОСОБЫ И ВАРИАНТЫ ПРИМЕНЕНИЯ | |
| NZ711657B2 (en) | Cftr mrna compositions and related methods and uses |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A621 | Written request for application examination |
Free format text: JAPANESE INTERMEDIATE CODE: A621 Effective date: 20211210 |
|
| TRDD | Decision of grant or rejection written | ||
| A01 | Written decision to grant a patent or to grant a registration (utility model) |
Free format text: JAPANESE INTERMEDIATE CODE: A01 Effective date: 20230126 |
|
| A61 | First payment of annual fees (during grant procedure) |
Free format text: JAPANESE INTERMEDIATE CODE: A61 Effective date: 20230227 |
|
| R150 | Certificate of patent or registration of utility model |
Ref document number: 7236525 Country of ref document: JP Free format text: JAPANESE INTERMEDIATE CODE: R150 |
|
| S111 | Request for change of ownership or part of ownership |
Free format text: JAPANESE INTERMEDIATE CODE: R313117 |
|
| R350 | Written notification of registration of transfer |
Free format text: JAPANESE INTERMEDIATE CODE: R350 |