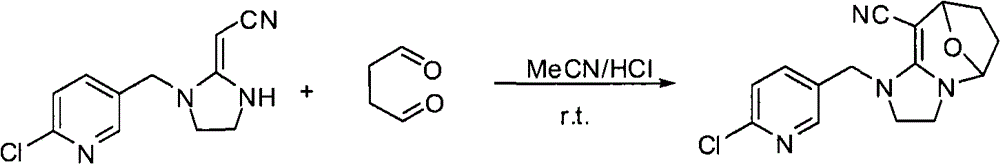CN101747320B - 二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 - Google Patents
二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 Download PDFInfo
- Publication number
- CN101747320B CN101747320B CN2008102073552A CN200810207355A CN101747320B CN 101747320 B CN101747320 B CN 101747320B CN 2008102073552 A CN2008102073552 A CN 2008102073552A CN 200810207355 A CN200810207355 A CN 200810207355A CN 101747320 B CN101747320 B CN 101747320B
- Authority
- CN
- China
- Prior art keywords
- alkyl
- carbonyl
- compound
- group
- saturated
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 0 *CN(*)C(N*)=C* Chemical compound *CN(*)C(N*)=C* 0.000 description 2
- CFRPSFYHXJZSBI-DHZHZOJOSA-N CCN(Cc(cn1)ccc1Cl)/C(/NC)=C/[N+]([O-])=O Chemical compound CCN(Cc(cn1)ccc1Cl)/C(/NC)=C/[N+]([O-])=O CFRPSFYHXJZSBI-DHZHZOJOSA-N 0.000 description 1
- WBWJDFFVJWYQPH-FLIBITNWSA-N CCN(Cc(cn1)ccc1Cl)/C(/SC)=C/[N+]([O-])=O Chemical compound CCN(Cc(cn1)ccc1Cl)/C(/SC)=C/[N+]([O-])=O WBWJDFFVJWYQPH-FLIBITNWSA-N 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/90—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having two or more relevant hetero rings, condensed among themselves or with a common carbocyclic ring system
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N33/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic nitrogen compounds
- A01N33/02—Amines; Quaternary ammonium compounds
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/34—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom
- A01N43/40—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with one nitrogen atom as the only ring hetero atom six-membered rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/50—1,3-Diazoles; Hydrogenated 1,3-diazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/54—1,3-Diazines; Hydrogenated 1,3-diazines
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/62—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms three- or four-membered rings or rings with more than six members
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/74—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,3
- A01N43/78—1,3-Thiazoles; Hydrogenated 1,3-thiazoles
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D213/00—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members
- C07D213/02—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members
- C07D213/04—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom
- C07D213/60—Heterocyclic compounds containing six-membered rings, not condensed with other rings, with one nitrogen atom as the only ring hetero atom and three or more double bonds between ring members or between ring members and non-ring members having three double bonds between ring members or between ring members and non-ring members having no bond between the ring nitrogen atom and a non-ring member or having only hydrogen or carbon atoms directly attached to the ring nitrogen atom with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D213/61—Halogen atoms or nitro radicals
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/02—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings
- C07D401/12—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/12—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains three hetero rings
- C07D471/14—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/12—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains three hetero rings
- C07D491/18—Bridged systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D498/08—Bridged systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D498/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D498/12—Heterocyclic compounds containing in the condensed system at least one hetero ring having nitrogen and oxygen atoms as the only ring hetero atoms in which the condensed system contains three hetero rings
- C07D498/18—Bridged systems
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Dentistry (AREA)
- Wood Science & Technology (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Agronomy & Crop Science (AREA)
- General Health & Medical Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Plural Heterocyclic Compounds (AREA)
- Pyridine Compounds (AREA)
- Nitrogen And Oxygen Or Sulfur-Condensed Heterocyclic Ring Systems (AREA)
- Low-Molecular Organic Synthesis Reactions Using Catalysts (AREA)
Abstract
本发明涉及二醛构建的具有杀虫活性的含氮或氧杂环化合物的制备及用途,提供了具有式(A)、(B)、(C)或(D)所示结构的化合物、或者所述化合物的光学异构体、顺反异构体或农药学上可接受的盐:
Description
技术领域
本发明涉及新型新烟碱类的杀虫剂、及其制备方法和应用。
背景技术
以吡虫啉为代表的新烟碱类杀虫剂因杀虫活性高,杀虫谱广,对哺乳动物和水生动物毒性低,且有良好的系统物性及适当的田间稳定性和环境友好性,成为新农药创制的重要热点领域。后来又相继开发出噻虫啉、噻虫胺、噻虫嗪、啶虫脒、烯啶虫胺、呋虫胺等一系列烟碱类杀虫剂(参见欧洲专利247477、296453、685477、235725、235725、315826、192060、244777、0386565、580553和1031566,日本专利62292765、8259568、8291171和7242633)。
但是由于吡虫啉过量频繁使用造成较为严重的抗性问题以及由于结构相似性带来的新烟碱杀虫剂之间的交互抗性,在一定程度上限制了该类化合物的应用,制约了此类化合物发展,同时新烟碱类杀虫剂主要对同翅目和鞘翅目害虫高效,其相对较窄的杀虫谱也限制了虫害防治方面的用药选择性。
因此,如何对具有高活性的硝基亚甲基化合物进行结构改造,以产生新的、更有效的杀虫剂,解决新烟碱类杀虫剂的抗性问题,扩大杀虫谱,使其应用于杀虫剂组合物就成为本领域迫切需要解决的技术问题。
发明内容
本发明提供了新的、更有效的杀虫剂,解决了新烟碱类杀虫剂的抗性问题,扩大了杀虫谱,解决了现有技术中存在的问题。
本发明目的在于,提供一类高效防治害虫的化合物及其制备方法。
本发明的另一个目的是为生长中的和收获的作物不受昆虫攻击和侵扰而提供保护。
一方面,本发明提供了一种具有式(A)、(B)、(C)或(D)所示结构的化合物、或者所述化合物的光学异构体、顺反异构体或农药学上可接受的盐:
式中:
R1为含氮、氧和/或硫的五元或六元杂环基,卤代的含氮、氧和/或硫的五元或六元杂环基,或者取代或未取代的苯基,其中,所述取代基为选自下组中的一个或多个:卤素,C1-4卤代烷基或C1-4氯代烷氧基;
R2为H,C1-C8饱和或不饱和烷基,卤代的C1-8饱和或不饱和烷基,-CH2CH2OCH2CH3,-CH2CH2OCH3,C1-8饱和或不饱和烷氧基,苯基,苄基,C1-4烷基-羰基或C1-4烷基-磺酰基;
R3和R4各自独立地为H,C1-6烷基,烯丙基,苄基,C1-4烷氧基-C1-4烷基,C1-4烷氧基-羰基,苯氧羰基,C2-6炔基-羰基,C2-3烯基-羰基,C3-6环烷基-羰基,苯甲酰基,或者被一个或多个选自卤原子、C1-4烷基、C1-4卤代烷基、C1-4烷氧基和C1-4烷基-羰基的取代基所取代的苯甲酰基,呋喃羰基或N,N-二甲基羰基,或者R3和R4共同构成-CH2-CH2-,-CH2-CH2-CH2-或CH2-XR-CH2-,式中X为N、O、S等杂原子,R是杂原子上的取代基,选自H,C1-6烷基,烯丙基,苄基,苯基,C1-4烷氧基-C1-4烷基,C1-4烷氧基-羰基,苯氧羰基,C2-6炔基-羰基,C2-3烯基-羰基,C3-6环烷基-羰基,苯甲酰基,或者被一个或多个选自卤原子、C1-4卤代烷基、C1-8饱和或不饱和烷基或烷氧基和C1-4烷基-羰基的取代基所取代的苯甲酰基,呋喃羰基或N,N-二甲基羰基;
R5,R6,R7,R8和R9为H,饱和或不饱和C1-4烷基,卤素,C1-8饱和或不饱和烷氧基,卤代C1-4饱和或不饱和烷氧基,C1-4烷基-羰基,C1-8烷基-酯基,C1-4烷基-磺酸酯基,苯基或苄基;
Y为硝基,氰基,三氟甲基,三氟乙酰基或三氟甲磺酰基。
在一个优选的实施方式中,R1选自:吡啶基、噻唑基、嘧啶基、四氢呋喃基、噁唑基或其卤代物。
在一个优选例中,优选R1为卤代的吡啶基、卤代的噻唑基、卤代的嘧啶基、卤代的四氢呋喃基、或卤代的噁唑基,更优选所述卤代物为氯代物。
在另一个优选的实施方式中,R2为H,C1-4饱和或不饱和烷基,卤代的C1-C4饱和或不饱和烷基,C1-4烷基-羰基,取代或未取代的苄基,其中,所述取代基为选自下组中的一个或多个:卤素,C1-4卤代烷基或C1-4氯代烷氧基。
在一个优选例中,R2为氢或C1-3烷基,优选氢或甲基。
在另一个优选的实施方式中,R3和R4为氢或C1-6烷基,或者R3和R4共同构成-CH2-CH2-或-CH2-CH2-CH2-。
在一个优选例中,R3和R4为氢或C1-3烷基,优选氢、甲基或乙基,或者R3和R4共同构成-CH2-CH2-或-CH2-CH2-CH2-。
在另一个优选的实施方式中,R5,R6,R7,R8和R9为H,饱和或不饱和C1-2烃基,卤素,C1-4饱和或不饱和烷氧基,卤代C1-2饱和或不饱和烷氧基,C1-4烷基-酯基(RCOO-),C1-2烷基-磺酰酯基或三氟甲磺酰酯。
在一个优选例中,R5,R6,R7,R8和R9为氢,甲基,氯,溴,甲氧基或乙氧基,优选氢,甲基,甲氧基。
在另一个优选的实施方式中,Y为硝基或氰基。
在一个优选例中,Y优选硝基。
另一方面,本发明提供了一种农用组合物,它包含:
(a)0.001-99.99重量%的上述化合物、其光学异构体、顺反异构体或农药学上可接受的盐、或者它们的组合;以及
(b)农药学上可接受的载体和/或赋形剂。
在一个优选例中,组分(a)占所述农用组合物的0.01-99.9重量%,优选0.05-90重量%。
在一个优选的实施方式中,所述农用组合物用于杀灭或预防选自下组的害虫:鞘翅目、鳞翅目、半翅目、直翅目、等翅目或双翅目昆虫。
在一个优选例中,所述害虫具有刺吸式或锉吸式口器。
在另一优选例中,所述害虫为蚜虫、飞虱、粉虱、叶蝉、蓟马、棉铃虫、菜青虫、小菜蛾、斜纹夜蛾、或粘虫。
在另一优选例中,所述农用组合物还包含其它活性物质,所述其它活性物质选自:杀虫剂、饵剂、杀菌剂、杀螨剂、杀线虫剂、杀真菌剂或生长控制剂。
另一方面,本发明涉及所述农用组合物的用途,用于杀灭或预防农业害虫、卫生害虫和危害动物健康的害虫;或用作用于杀灭或预防农业害虫、卫生害虫和危害动物健康的杀虫剂组合物。
另一方面,本发明提供了一种杀虫和/或防虫方法,所述方法包括将上述农用组合物施加于遭受或可能遭受虫害的植物体、其周围的土壤或环境中。
另一方面,本发明涉及上述化合物、其光学异构体、顺反异构体或农药学上可接受的盐、或它们的组合在制备杀虫剂组合物中的用途。
再一方面,本发明提供了上述化合物、其光学异构体、顺反异构体或农药学上可接受的盐的制备方法,所述方法包括步骤:
在催化量酸的存在下,于0-60℃下,使式(a)化合物与式(b)、(c)或(d)化合物反应,从而制得具有式(A)、(B)、(C)或(D)的化合物,式中R2为氢,
式中,R1、R3、R4、R5、R6,R7,R8,R9和Y如上文中所定义,n为0或1。
在一个优选例中,所述反应温度为15-45℃,优选20-30℃。
在另一优选例中,所用的溶剂选自:乙腈或乙醇,优选乙腈。
在另一优选例中,所用的催化量的酸选自:浓盐酸、浓硫酸或苯甲酸,优选浓盐酸。
在一个优选的实施方式中,所述方法包括步骤:
在乙腈中,于催化量的酸存在下,于20-30℃下,进行如下反应2-24小时,从而获得式(A)的化合物,式中R2为氢:
在乙腈中,于催化量的酸存在下,于20-30℃下,进行如下反应2-24小时,从而获得式(B)的化合物:
在乙腈中,于催化量的酸存在下,于10-50℃下,进行如下反应2-24小时,从而获得式(C)的化合物:
在乙腈中,于催化量的酸存在下,于10-50℃下,进行如下反应2-24小时,从而获得式(D)的化合物:
具体实施方式
本发明的发明人通过长期而深入的研究,基于现有的硝基亚甲基类新烟碱杀虫剂的硝基亚甲基结构,通过二醛与硝基亚甲基类化合物反应,合成了一种新型的新烟碱化合物,该化合物的杀虫活性显著提高,并具有扩大的杀虫谱。在此基础上,发明人完成了本发明。
基团定义
如本文所用,术语“C1-6烷基”指具有1-6个碳原子的直链或支链烷基,例如甲基、乙基、丙基、异丙基、丁基、异丁基、仲丁基、叔丁基、或类似基团。
术语“C1-6烷氧基”指具有1-6个碳原子的直链或支链烷氧基,例如甲氧基、乙氧基、丙氧基、异丙氧基、丁氧基、异丁氧基、仲丁氧基、叔丁氧基、或类似基团。
术语“卤素”指氟、氯、溴、或碘。术语“卤代的”指被相同或不同的一个或多个上述卤原子取代的基团,例如三氟甲基、五氟乙基、或类似基团。
术语“五元或六元杂环基”指含一个或多个选自氮、氧或硫的杂原子的五元或六元环,例如吡啶基、噻唑基、嘧啶基、四氢呋喃基、或噁唑基等。
本发明化合物的制备方法
本发明化合物可通过上文所描述的反应步骤合成。本领域技术人员可按照现有技术文献合成反应步骤中的式(a)化合物,例如可参照WO 2006056108A1,WO 2007101369A1和PCT/CN2008/071115。
在本发明的一个具体实施方式中,式(A)化合物的合成方法如下:
在本发明的一个具体实施方式中,式(B)化合物的合成方法如下:
在本发明的一个具体实施方式中,式(C)化合物的合成方法如下:
在本发明的一个具体实施方式中,式(C)化合物的合成方法如下:
在本发明的一个实施方式中,式(A)的化合物可通过下列反应制备:
(1)将2-氯-5-氯甲基吡啶的乙腈溶液滴加到5-10倍摩尔量的二胺溶液中,在0-50℃之间,反应5-10小时。后处理将二胺在减压条件下除去,用乙酸乙酯溶解,旋干溶剂,得到产品:N*1*-(6-氯-3-甲基吡啶基)-二胺;
(2)N*1*-(6-氯-3-甲基吡啶基)-二胺与1,1-二硫甲基-2-硝基乙烯,以乙醇作溶剂,回流4-8个小时,得到产品:硝基亚甲基类化合物;
(3)硝基亚甲基类化合物与乙二醛在酸(盐酸、硫酸、杂多酸等)催化条件下,得到所述通式的化合物(A)。
在另一个实施方式中,式(B)的化合物可通过下列反应制备:
(1)乙胺水溶液中加入适量乙腈,在冰浴下滴加6-氯-3-氯甲基吡啶的乙腈溶解液,TLC跟踪反应进程,反应结束后,向反应混合液中加入大量的水,二氯甲烷萃取,干燥,抽滤,蒸去溶剂,得到油状液体N-(6-氯吡啶-3-亚甲基)乙胺;
(2)N-(6-氯吡啶-3-亚甲基)乙胺与1,1-二硫甲基-2-硝基乙烯,以乙醇作溶剂,回流4-8个小时,浓缩,柱层析分离得到产物N-(6-氯吡啶-3-亚甲基)-N-乙基-1-硫甲基-2-硝基亚乙烯基胺;
(3)N-(6-氯吡啶-3-亚甲基)-N-乙基-1-硫甲基-2-硝基亚乙烯基胺与甲胺醇溶液,以乙醇作溶剂,在冰浴下反应4-8个小时,浓缩,柱层析分离得到N-(6-氯吡啶-3-亚甲基)-N-乙基-N’-甲基-2-硝基亚乙烯基二胺;
(4)N-(6-氯吡啶-3-亚甲基)-N-乙基-N’-甲基-2-硝基亚乙烯基二胺与丙二醛在酸(盐酸、硫酸、杂多酸等)催化条件下,得到所述式的化合物(B)。
在另一个实施方式中,式(C)的化合物可通过下列反应制备:
(1)将2-氯-5-氯甲基吡啶的乙腈溶液滴加到5-10倍摩尔量的二胺溶液中,在0-50℃之间,反应5-10小时;后处理将二胺在减压条件下除去,用乙酸乙酯溶解,旋干溶剂,得到产品:N*1*-(6-氯-3-甲基吡啶基)-二胺;
(2)N*1*-(6-氯-3-甲基吡啶基)-二胺与1,1-二硫甲基-2-硝基乙烯,以乙醇作溶剂,回流4-8个小时,得到产品:硝基亚甲基类化合物;
(3)硝基亚甲基类化合物(2-氯-5-(2-硝基亚甲基-咪唑烷-1-基甲基)-吡啶)与丁二醛在酸(盐酸、硫酸、杂多酸等)催化条件下,得到所述通式的化合物(C)。
在另一个实施方式中,式(D)的化合物可通过下列反应制备:
(1)将2-氯-5-氯甲基吡啶的乙腈溶液滴加到5-10倍摩尔量的二胺溶液中,在0-50℃之间,反应5-10小时。后处理将二胺在减压条件下除去,用乙酸乙酯溶解,旋干溶剂,得到产品N*1*-(6-氯-3-甲基吡啶基)-二胺;
(2)N*1*-(6-氯-3-甲基吡啶基)-二胺与1,1-二硫甲基-2-硝基乙烯,以乙醇作溶剂,回流4-8个小时,得到产品:硝基亚甲基类化合物;
(3)硝基亚甲基类化合物(2-氯-5-(2-硝基亚甲基-咪唑烷-1-基甲基)-吡啶)与戊二醛在酸(盐酸、硫酸、杂多酸等)催化条件下,得到所述通式的化合物(D)。
本发明活性物质的杀虫活性
术语“本发明的活性物质”或“本发明的活性化合物”是指本发明化合物、其光学异构体、顺反异构体或农药学上可接受的盐,其具有显著提高的杀虫活性,以及扩大的杀虫谱。
术语“农药学上可接受的盐”意指该盐的阴离子在形成杀虫剂药学上可接受的盐时为已了解的和可接受的。该盐较好的为水溶性的。合适的、由式(A),(B),(C)和(D)化合物形成的酸加成盐包括有无机酸形成的盐,例如盐酸盐、磷酸盐、硫酸盐、硝酸盐;及包括有机酸形成的盐,如醋酸盐,苯甲酸盐。
本发明的活性物质能用作控制和消灭广泛的农林植物害虫、贮藏谷类的害虫、公共卫生害虫以及危害动物健康的害虫等。在本说明书中,“杀虫剂”是具有防治上述提到的所有害虫的作用的物质的统称。害虫的例子包括但不限于:鞘翅目昆虫:玉米象(Sitophilus zeamais),赤拟谷盗(Tribolium castaneum),马铃薯瓢虫(Henosepilachna vigintioctomaculata),二十八星瓢虫(Henosepilachnasparsa),细胸叩头虫(Agriotes fuscicollis),红脚绿金龟(Anomala cupripes),四纹丽金龟(Popillia quadriguttata),马铃薯叶甲(Monolepta hieroglyphica),松天牛(Monochamus alternatus),稻根象(Echinocnemus squameus),泡桐叶甲(Basiprionota bisignata),星天牛(Anoplophora chinensis),桑天牛(Apriponagermari),脐腹小蠹(Scolytus schevy),或细胸金针虫(Agriotes fuscicollis);鳞翅目昆虫:舞毒娥(Lymantria dispar),天幕毛虫(Malacosoma neustria testacea),黄杨绢野螟(Diaphania perspectalis),大袋蛾(Clania variegata),黄刺蛾(Cnidocampa flauescens),马尾松毛虫(Dendrolimus punctatus),古毒蛾(Orgyiagonostigma),白杨透翅蛾(Paranthrene tabaniformis),斜纹夜蛾(Spodopteralitura),二化螟(Chilo suppressalis),玉米螟(Ostrinia nubilalis),粉斑螟(Ephestiacautella),棉卷蛾(Adoxophyes orana),栗子小卷蛾(laspyresia splendana),小地老虎(Agrotis fucosa),大蜡螟(Galleria mellonella),菜蛾(Plutella xylostella),桔潜蛾(Phyllocnistis citrella),或东方粘虫(Mythimna separata);同翅目昆虫:黑尾叶蝉(Nephotettix cincticeps),稻褐飞虱(Nilaparvata lugens),康氏粉蚧(Pseudococcus comstocki),矢尖蚧(Unaspis yanonensis),桃蚜(Myzus persicae),棉蚜(Aphis gossydii),萝卜蚜(Lipaphis erysimi pseudobrassicae),梨班网蝽(Stephanitis nashi),或粉虱(Bemisia tabaci);直翅目昆虫:德国小蠊(Blattellagermanica),美国大蠊(Periplaneta american),非洲蝼蛄(Gryllotalpa africana),或亚洲飞蝗(Locus migratoria);等翅目昆虫:入侵红火蚁(Solenopsis invicta),或家白蚁(Coptotermes formosanus);双翅目昆虫:家蝇(Musca domestica),埃及伊蚊(Aedes aegypti),种蝇(Delia platura),库蚊(Culex sp.),或中华按蚊(Anopheles sinensis)。危害动物健康的害虫包括微小牛蜱(Boophilus microplus),长角血蜱(Haemaphysalis longicornis),小亚璃眼蜱(Hyalomma anatolicum),牛皮蝇(Hypoderma spp.),肝片吸虫(Fasciola hepatica),贝氏莫尼茨绦虫(Moniezia blanchard),奥斯特线虫(Ostertagia spp.),原虫Trypanosoma enansi,Babesia bigemina等。
本发明涉及的化合物尤其对刺吸式、锉吸式口器害虫,如蚜虫、叶蝉、飞虱、蓟马、粉虱等农林害虫有特效。
含本发明活性物质的杀虫剂组合物
可将本发明的活性物质以常规的方法制备成杀虫剂组合物。这些活性化合物可做成常规的制剂,例如溶液剂,乳剂,混悬剂,粉剂,泡沫剂,糊剂,颗粒剂;气雾剂,用活性物质浸渍的天然的和合成的材料,在多聚物中的微胶囊,用于种子的包衣复方,和与燃烧装置一起使用的制剂,例如烟熏药筒,烟熏罐和烟熏盘,以及ULV冷雾(Cold mist)和热雾(Warm mist)制剂。
这些制剂可用已知的方法生产,例如,将活性化合物与扩充剂混合,这些扩充剂就是液体的或液化气的或固体的稀释剂或载体,并可任意选用表面活性剂即乳化剂和/或分散剂和/或泡沫形成剂。例如在用水作扩充剂时,有机溶剂也可用作助剂。
用液体溶剂作稀释剂或载体时基本上是合适的,如:芳香烃类,例如二甲苯,甲苯或烷基萘;氯化的芳香或氯化的脂肪烃类,例如氯苯,氯乙烯或二氯甲烷;脂肪烃类,例如环己烷或石蜡,例如矿物油馏分;醇类,例如乙醇或乙二醇以及它们的醚和脂类;酮类,例如丙酮,甲乙酮,甲基异丁基酮或环己酮;或不常用的极性溶剂,例如二甲基甲酰胺和二甲基亚砜,以及水。液化气的稀释剂或载体是指在常温常压下将成为气体的液体,例如气溶胶推进剂,如卤化的烃类以及丁烷,丙烷,氮气和二氧化碳。
固体载体可用地面天然的矿物质,例如高岭土,粘土,滑石,石英,活性白土,蒙脱土,或硅藻土,和地面合成的矿物质,例如高度分散的硅酸,氧化铝和硅酸盐。供颗粒用的固体载体是碾碎的和分级的天然告石,例如方解石,大理石,浮石,海泡石和白云石,以及无机和有机粗粉合成的颗粒,和有机材料例如锯木屑,椰子壳,玉米棒子和烟草梗的颗粒等。
非离子的和阴离子的乳化列可用作乳化剂和/或泡沫形成剂。例如聚氧乙烯-脂肪酸酯类,聚氧乙烯-脂肪醇醚类,例如烷芳基聚乙二醇醚类,烷基磺酸酯类,烷基硫酸酯类,芳基磺酸酯类以及白蛋白水解产物。分散剂包括,例如木质素亚硫酸盐废液和甲基纤维素。
在制剂中可以用粘合剂,例如羧甲基纤维素和以粉末,颗粒或乳液形式的天然和合成的多聚物,例如阿拉伯胶,聚乙烯基醇和聚乙烯醋酸酯。可以用着色剂例如无机染料,如氧化铁,氧化钻和普鲁士蓝;有机染料,如有机染料,如偶氯染料或金属钛菁染料;和用痕量营养剂,如铁,猛,硼,铜,钴,铝和锌的盐等。
本发明的这些活性化合物可与其它活性化合物制成混合物存在于商品制剂中或从这些制剂制备的使用剂型中,所述其它的活性化合物包括但不限于:杀虫剂,合饵,杀菌剂,杀螨剂,杀线,杀真菌剂,生长控制剂等。杀虫剂包括,例如磷酸酯类,氨基甲酸酯类,除虫菊酯类,氯化烃类,苯甲酰脲类,沙蚕毒素类以及由微生物产生的物质,如阿维菌素。
此外,本发明的这些活性化合物也可与增效剂制成一种混合物存在于它们的商品制剂中成从这些制剂制备的使用剂型中。增效剂是提高活性化合物作用的化合物,由于活性化合物本身有活性,也可不必加增效剂。
这些制剂通常含有占所述杀虫剂组合物0.001-99.99重量%,优选0.01-99.9重量%,更优选0.05-90重量%的本发明的活性化合物。从商品制剂制成使用剂型中的活性化合物的浓度可在广阔的范围内变动。使用剂型中的活性化合物的浓度可从0.0000001-100%(g/v),最好在0.0001与1%之间。
实施例
下面结合具体实施例,进一步阐述本发明。应理解,这些实施例仅用于说明本发明而不用于限制本发明的范围。下列实施例中未注明具体条件的实验方法,通常按照常规条件,或按照制造厂商所建议的条件。除非另外说明,否则百分比和份数按重量计算。其中,r.t.代表室温。
实施例1:4-(1-((6-氯吡啶-3-基)甲基)-4,5-二氢-1H-咪唑烷-2-基)-1-(1-((6-氯吡啶-3-基)甲基)咪唑烷-2-基)-1,4-二硝基-3-丁烯-2-醇(化合物13)的合成
以0.03mol的2-氯-5-氯甲基吡啶为起始原料,按照WO 2006056108A1和WO 2007101369A1所述的方法制备2-氯-5-(2-硝基亚甲基-咪唑烷-1-基甲基)-吡啶,产率为56%;Rf=0.46(石油醚∶乙酸乙酯=1∶1);mp=156.9℃-161.8℃。GC MS(m/s)220(25),126(100),90(9)。
4-(1-((6-氯吡啶-3-基)甲基)-4,5-二氢-1H-咪唑烷-2-基)-1-(1-((6-氯吡啶-3-基)
甲基)咪唑烷-2-基)-1,4-二硝基-3-丁烯-2-醇的合成
将1.27g(0.005mol)的2-氯-5-(2-硝基亚甲基-咪唑烷-1-基甲基)-吡啶,30ml的无水乙腈,3ml 30%的乙二醛水溶液加入50ml的圆底烧瓶中,搅拌半个小时,然后加入催化量的浓盐酸催化,继续搅拌,TLC跟踪反应至原料全部反应完全,然后抽滤得白色粉末状固体。重结晶得到化合物13的纯品,为白色粉末状固体1.05g,产率约76%。
mp=164.6-165.3℃;1H NMR(400Mz,DMSO-d6):δ9.01(s,1H),8.41(d,J=2.0Hz,1H),8.38(d,J=2.0Hz,1H),7.80-7.86(m,2H),7.51-7.54(m,2H),6.50(d,J=7.2Hz,1H),5.34(d,J=15.2Hz,1H),5.18(d,J=15.2Hz,1H),4.84(dd,J1=2.4Hz,J2=7.2Hz,1H),4.77(d,J=16.8Hz,1H),4.67(d,J=16.8Hz,1H),3.98(d,J=2.4Hz,1H),3.86-3.95(m,2H),3.61-3.80(m,5H),3.40-3.47(m,1H)ppm;13C NMR(100Mz,DMSO-d6):δ162.7,158.7,148.3,148.2,148.0,147.7,138.1,137.7,130.9,130.2,123.1,123.0,102.5,101.4,81.4,53.8,52.6,49.4,48.8,46.4,41.2,41.0ppm;HRMS(ES+)计算值C22H23N8O5 35Cl2(M+H)+,549.1168;测得值,549.1178;计算值C22H23N8O5 35Cl37Cl(M+H)+,551.1139;测得值,551.1152;计算值C22H23N8O5 37Cl2(M+H)+,553.1109;测得值,553.1108。
实施例2:2-氯-5-((-2-(-4-(1-((6-氯吡啶-3-基)甲基)-4,5-二氢-1H-咪唑烷-2-基)-2-甲氧基-1,4-二硝基-3-丁烯基)咪唑烷-1-基)甲基)吡啶(化合物14)的合成
将0.549g(0.001mol)的化合物1加入到50ml的圆底烧瓶中,再加入10ml的甲醇,50ml二氯甲烷和催化量的浓盐酸,回流,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到黄色粉末状纯品,产率为62%。
mp=151.6-153.1℃;1H NMR(400Mz,DMSO-d6):δ9.03(s,1H),8.38(d,J=2.0Hz,1H),8.36(d,J=2.0Hz,1H),7.81-7.85(m,2H),7.49-7.51(m,2H),6.50(d,J=7.2Hz,1H),5.35(d,J=15.2Hz,1H),5.19(d,J=15.2Hz,1H),4.80(d,J1=7.2Hz,1H),4.77(d,J=16.8Hz,1H),4.69(d,J=16.8Hz,1H),3.68(s,3H),3.88-3.95(m,2H),3.61-3.85(m,5H),3.38-3.41(m,1H)ppm;13C NMR(100Mz,DMSO-d6):δ162.6,158.7,148.9,148.3,148.1,147.6,138.1,137.8,130.9,129.9,122.8,123.1,102.2,101.6,81.6,58.7,53.8,52.6,49.6,48.9,46.4,41.3,41.0ppm;HRMS(ES+)计算值C23H25N8O5 35Cl2(M+H)+,563.1325;测得值,563.1311.计算值C23H25N8O5 35Cl37Cl(M+H)+,565.1295。
实施例3:N1,N7-二((6-氯吡啶-3-基)甲基)N1,N7-二乙基-N’1,N’7-二甲基-2,6-二硝基-2,5-庚二亚胺(化合物37)的合成
(1)N-(6-氯吡啶-3-亚甲基)乙胺的合成
在带有恒压滴液漏斗、温度计的300mL三口圆底烧瓶中,加入65-70%乙胺水溶液(70g,1mol),乙腈50mL,在冰浴下搅拌15分钟,使溶液温度稳定在0℃附近,在恒压滴液漏斗中加入6-氯-3-氯甲基吡啶(16.10g,0.10mol)的乙腈(25mL)溶液,控制滴加速度在3滴/分钟,滴加时间在3.5小时,反应结束后,加水,二氯甲烷萃取,收集有机相,得油状液体N-(6-氯吡啶-3-亚甲基)乙胺14g,收率70%。GC-MS:m/z(%)=170([M]+,20),155(80),126(100),114(10),90(12)。
(2)N-(6-氯吡啶-3-亚甲基)-N-乙基-1-硫甲基-2-硝基亚乙烯基胺的合成
在100毫升三口圆底烧瓶中,加入N-(6-氯吡啶-3-亚甲基)乙胺(17.0g,0.1mol),1,1-二硫甲基-2-硝基乙烯(15.0g,0.09mol),无水乙醇(50mL),回流。反应结束后,冷却至室温,减压浓缩得到浓稠状液体,柱层析分离得N-(6-氯吡啶-3-亚甲基)-N-乙基-1-硫甲基-2-硝基亚乙烯基胺5.3g,收率18.5%。GC-MS:m/z(%)=242([M]+-46,53),227(15),213(100),169(45),155(28),141(29),126(91),90(12)。
(3)N-(6-氯吡啶-3-亚甲基)-N-乙基-N’-甲基-2-硝基亚乙烯基二胺的合成
向100mL单口圆底烧瓶中,加入N-(6-氯吡啶-3-亚甲基)-N-乙基-1-硫甲基-2-硝基亚乙烯基胺(5g,0.017mol),甲胺醇溶液(1.8g,0.017mol甲胺),无水乙醇(30mL),在冰浴下搅拌使温度降至0℃,继续反应直至结束。再减压旋转蒸去溶剂,浓缩得浆状物,将此浆状物用适量二氯甲烷溶解,以硅胶为填料,用二氯甲烷∶乙醇=25∶1为展开剂进行柱分,收集产品,浓缩得到N-(6-氯吡啶-3-亚甲基)-N-乙基-N’-甲基-2-硝基亚乙烯基二胺0.9g,收率19.1%。TLC:二氯甲烷∶丙酮=5∶1,Rf=0.23.熔点:78-80oC.(lit[67]79-81℃)GC-MS:m/z(%)=236([M]+-34,32),207(49),169(52),126(49),110(20),90(16),67(100).16.65
(4)N1,N7-二((6-氯吡啶-3-基)甲基)N1,N7-二乙基-N’1,N’7-二甲基-2,6-二
硝基-2,5-庚二亚胺(化合物37)的合成
将1.35g(0.005mol)的N-(6-氯吡啶-3-亚甲基)-N-乙基-N’-甲基-2-硝基亚乙烯基二胺,30ml的无水乙腈,0.72g(0.01mol)的丙二醛,催化量的HCl置于50ml的圆底烧瓶中。常温下搅拌,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到淡黄色粉末状纯品,产率为56%。
mp=117.3-118.7℃;1H NMR(400Mz,DMSO-d6):δ8.36(d,J=2.4Hz,2H),7.88(dd,J1=2.4Hz,J2=8.4Hz,2H),7.51(d,J=8.4Hz,2H),4.97(t,J=2.8Hz,2H),4.86(d,J=15.2Hz,2H),4.49(d,J=15.2Hz,2H),3.95-3.99(m,4H),3.66-3.78(m,6H),3.12-3.21(m,2H),1.91-1.93(m,6H)ppm;13C NMR(100Mz,DMSO-d6):δ156.3,148.5,148.1,137.3,131.9,122.5,104.8,49.2,48.9,48.0,48.5,28.1,20.2ppm;HRMS(EI+)计算值C25H30N8O4 35Cl2(M+),576.1767;测得值,576.1751。
实施例4:1,5-二(1-((6-氯吡啶-3-基)甲基)-4,5-二氢-1H-咪唑烷-2-基)-1,5-二硝基-1,4-戊二烯(化合物39)的合成
将1.27g(0.005mol)的2-氯-5-(2-硝基亚甲基-咪唑烷-1-基甲基)-吡啶,30ml的无水乙腈,0.720g(0.01mol)的丙二醛,催化量的HCl置于50ml的圆底烧瓶中。常温下搅拌,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到淡黄色粉末状纯品,产率为52%。
mp=136.5-137.8℃;1H NMR(400Mz,DMSO-d6):δ8.34(d,J=2.4Hz,2H),7.82(dd,J1=2.4Hz,J2=8.4Hz,2H),7.47(d,J=8.4Hz,2H),4.96(t,J=2.8Hz,2H),4.81(d,J=15.8Hz,2H),4.44(d,J=15.8Hz,2H),3.92-3.97(m,4H),3.65-3.72(m,2H),3.49-3.56(m,2H),1.92-1.93(m,2H)ppm;13C NMR(100Mz,DMSO-d6):δ155.3,147.9,147.8,138.0,130.9,122.7,104.8,50.2,48.9,48.5,48.5,28.1ppm;HRMS(ES+)计算值C23H23N8O4 35Cl2(M+H)+,545.1219;测得值,545.1201。计算值C23H23N8O4 35Cl37Cl(M+H)+,547.1190;测得值,547.1178.计算值C23H23N8O4 37Cl2(M+H)+,549.1160;测得值,549.1181。
实施例5:1,5-二(1-((2-氯噻唑-5-基)甲基)-4,5-二氢-1H-咪唑烷-2-基)-1,5-二硝基-1,4-戊二烯(化合物41)的合成
按照实施例1所述的方法,以0.03mol 2-氯-5-氯甲基噻唑代替2-氯-5-氯甲基吡啶作为起始原料制备1-((2-氯噻唑-5-基)甲基)-2-(硝基亚甲基)-1-咪唑烷,产率为56%;GC MS(m/s)226(24),132(100),77(9)。
1,5-二(1-((2-氯噻唑-5-基)甲基)-4,5-二氢-1H-咪唑烷-2-基)-1,5-二硝基-1,4-戊
二烯的合成
将1.30g(0.005mol)的1-((2-氯噻唑-5-基)甲基)-2-(硝基亚甲基)-1-咪唑烷,30ml的无水乙腈,0.720g(0.01mol)的丙二醛,催化量的HCl置于50ml的圆底烧瓶中。常温下搅拌,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到淡黄色粉末状纯品,产率为44%。
mp=138.6-139.9℃;1H NMR(400Mz,DMSO-d6):δ7.63(s,1H),4.98(t,J=2.8Hz,2H),4.85(d,J=15.8Hz,2H),4.43(d,J=15.8Hz,2H),3.96-3.99(m,4H),3.67-3.71(m,2H),3.51-3.56(m,2H),1.95-1.97(m,2H)ppm;13C NMR(100Mz,DMSO-d6):δ157.6,149.3,138.3,105.8,50.6,48.9,48.4,48.1,29.1ppm;HRMS(ES+)计算值C19H19N8O4S2 35Cl2(M+H)+,557.0348;测得值,557.0363.计算值C19H19N8O4S2 35Cl37Cl(M+H)+,559.0318;测得值,559.0620。
实施例6:1,5-二(1-((6-氯吡啶-3-基)甲基)-1,4,5,6-四氢嘧啶-2-基)-1,5-二硝基-1,4-戊二烯(化合物43)的合成
以2.42g(0.015mol)的对氯吡啶为起始原料,按照WO 2006056108A1和WO2007101369A1所述的方法制备1-(6-氯-3-甲基吡啶基)-2-硝基亚甲基六氢嘧啶,产率为56%;Rf=0.19(乙醇∶二氯甲烷=1∶1);mp=175.7-182.6℃。GC MS(m/s)225(100),196(9),154(10),139(11),126(31),113(10),90(31)。
1,5-二(1-((6-氯吡啶-3-基)甲基)-4,5-二氢-1H-咪唑烷2-基)-1,5-二硝基-1,4-戊
二烯的合成
将1.34g(0.005mol)的1-(6-氯-3-甲基吡啶基)-2-硝基亚甲基六氢嘧啶,30ml的无水乙腈,0.720g(0.01mol)的丙二醛,催化量的HCl置于50ml的圆底烧瓶中。常温下搅拌,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到淡黄色粉末状纯品,产率为55%。
mp=133.7-134.9℃;1H NMR(400Mz,DMSO-d6):δ8.32(d,J=2.4Hz,2H),7.81(dd,J1=2.4Hz,J2=8.4Hz,2H),7.49(d,J=8.4Hz,2H),4.93(t,J=2.8Hz,2H),4.78(d,J=15.8Hz,2H),4.40(d,J=15.8Hz,2H),3.91-3.96(m,4H),3.63-3.71(m,2H),3.49-3.53(m,2H),2.32-2.37(m,2H),1.92-1.93(m,2H)ppm;13C NMR(100Mz,DMSO-d6):δ155.6,148.3,147.9,138.0,130.7,122.7,105.1,50.2,48.8,48.5,48.1,36.2,28.0ppm;HRMS(ES+)计算值C25H27N8O4 35Cl2(M+H)+,572.1454;测得值,572.1468。计算值C25H27N8O4 35Cl37Cl(M+H)+,574.1425;测得值,574.1416。
实施例7:N-((6-氯吡啶-3-基)甲基)-N-乙基-2-甲基-4-硝基-8-氧杂-2-氮杂双环[3,2,1]辛-3-烯-3-胺(化合物46)的合成
将1.35g(0.005mol)的N-(6-氯吡啶-3-亚甲基)-N-乙基-N’-甲基-2-硝基亚乙烯基二胺,30ml的无水乙腈,0.860g(0.01mol)的丁二醛,催化量的HCl置于50ml的圆底烧瓶中。常温下搅拌,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到淡黄色粉末状纯品,产率为40%。
mp=125.3-125.7℃;1H NMR(400Mz,DMSO-d6):δ8.72(s,1H),8.27(d,J=2.4Hz,1H),7.75(dd,J1=2.4Hz,J2=8.4Hz,1H),7.58(d,J=8.4Hz,1H),5.25-5.30(s,2H),4.41-4.50(m,2H),2.96-3.26(m,2H),2.86(s,3H),2.36-2.41(m,2H),1.81-1.5(m,4H),1.16-1.26(m,3H)ppm;13C NMR(100Mz,DMSO-d6):δ158.5,154.4,151.6,150.8,148.1,139.3,137.1,107.8,89.5,65.8,49.8,46.9,40.6,21.9,20.3ppm;HRMS(EI+)计算值C15H21N4O3 35Cl(M+),339.1224;测得值,339.1257。计算值C15H21N4O3 37Cl(M+),341.1194;测得值,341.1213。
实施例8:9-((6-氯吡啶-3-基)甲基)-4-硝基-8-氧杂-10,11-二氢咪唑并[2,3-a]双环[3,2,1]辛-3-烯(化合物52)的合成
将1.27g(0.005mol)的2-氯-5-(2-硝基亚甲基-咪唑烷-1-基甲基)-吡啶,30ml的无水乙腈,0.860g(0.01mol)的丁二醛,催化量的HCl置于50ml的圆底烧瓶中。常温下搅拌,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到淡黄色粉末状纯品,产率为71%。
mp=149.0-150.0℃;1H NMR(400Mz,DMSO-d6):δ8.35(d,J=2.4Hz,1H),7.81(dd,J1=2.4Hz,J2=8.4Hz,1H),7.51(d,J=8.4Hz,1H),5.36-5.39(s,2H),5.00(d,J=15.6Hz,1H),4.68(d,J=15.6Hz,1H),3.57-3.73(m,4H),1.94-2.04(m,4H)ppm;13C NMR(100Mz,DMSO-d6):δ155.6,149.7,149.6,139.7,132.6,124.5,109.6,87.0,75.1,51.2,50.3,46.6,31.9,31.7ppm;HRMS(ES+)计算值C14H16N4O3 35Cl(M+H)+,323.0911;测得值,323.0912。计算值C14H16N4O3 37Cl(M+H)+,325.0811;测得值,325.0895。计算值C14H15N4O3 35ClNa(M+Na)+,345.0730;测得值,345.0722。计算值C14H15N4O3 37ClNa(M+Na)++,347.0701;测得值,347.0692。
实施例9:9-((2-氯噻唑-5-基)甲基)-4-硝基-8-氧杂-10,11-二氢咪唑并[2,3-a]双环[3,2,1]辛-3-烯(化合物53)的合成
将1.30g(0.005mol)的2-氯-5-(2-硝基亚甲基-咪唑烷-1-基甲基)-吡啶,30ml的无水乙腈,0.860g(0.01mol)的丁二醛,催化量的HCl置于50ml的圆底烧瓶中。常温下搅拌,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到淡黄色粉末状纯品,产率为63%。
mp=151.7-152.1℃;1H NMR(400Mz,DMSO-d6):δ7.65(s,1H),5.33-5.37(s,2H),5.01(d,J=15.6Hz,1H),4.69(d,J=15.6Hz,1H),3.52-3.70(m,4H),1.90-2.01(m,4H)ppm;13C NMR(100Mz,DMSO-d6):δ155.2,149.6,139.1,124.5,110.6,87.1,75.6,51.3,50.6,46.9,31.9,31.2ppm;HRMS(ES+)计算值C12H15N4O3S35Cl(M+H)+,329.0475;测得值,329.0412.计算值C12H15N4O3S37Cl(M+H)+,331.0446;测得值,331.0423。
实施例10:9-((6-氯吡啶-3-基)甲基)-4-硝基-8-氧杂-10,11,12-三氢嘧啶并[2,3-a]双环[3,2,1]辛-3-烯(化合物61)的合成
将1.34g(0.005mol)的1-(6-氯-3-甲基吡啶基)-2-硝基亚甲基六氢嘧啶,30ml的无水乙腈,0.860g(0.01mol)的丁二醛,催化量的HCl置于50ml的圆底烧瓶中。常温下搅拌,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到淡黄色粉末状纯品,产率为38%。
mp=143.2-144.9℃;1H NMR(400Mz,DMSO-d6):δ8.33(d,J=2.4Hz,1H),7.80(dd,J1=2.4Hz,J2=8.4Hz,1H),7.49(d,J=8.4Hz,1H),5.32-5.35(s,2H),5.00(d,J=15.6Hz,1H),4.66(d,J=15.6Hz,1H),3.51-3.68(m,4H),2.33-2.41(m,2H),1.89-2.00(m,4H)ppm;13C NMR(100Mz,DMSO-d6):δ155.5,149.6,149.3,139.7,132.6,124.1,109.3,86.6,75.1,51.2,50.7,46.6,32.1,31.7,26.9ppm;HRMS(ES+)计算值C15H19N4O3 35Cl(M+H)+,337.1067;测得值,337.1015。计算值C15H19N4O3 37Cl(M+H)+,339.1038;测得值,339.0995。
实施例11:9-((6-氯吡啶-3-基)甲基)-4-氰基-8-氧杂-10,11-二氢咪唑并[2,3-a]双环[3,2,1]辛-3-烯(化合物64)的合成
将1.17g(0.005mol)的2-氯-5-(2-氰基亚甲基-咪唑烷-1-基甲基)-吡啶,30ml的无水乙腈,0.860g(0.01mol)的丁二醛,催化量的HCl置于50ml的圆底烧瓶中。常温下搅拌,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到淡黄色粉末状纯品,产率为66%。
mp=125.1-126.8℃;1H NMR(400Mz,DMSO-d6):δ8.34(d,J=2.4Hz,1H),7.82(dd,J1=2.4Hz,J2=8.4Hz,1H),7.55(d,J=8.4Hz,1H),5.33(s,1H),5.25(s,1H),5.00(d,J=15.6Hz,1H),4.78(d,J=15.6Hz,1H),3.56-3.78(m,4H),1.91-2.00(m,4H)ppm;13C NMR(100Mz,DMSO-d6):δ153.6,149.0,148.6,139.7,132.3,121.5,99.6,87.3,75.7,51.3,50.2,46.6,31.5,29.7ppm;HRMS(ES+)计算值C15H16N4O35Cl(M+H)+,303.1013;测得值,303.0992。计算值C15H16N4O37Cl(M+H)+,305.0983;测得值,305.0957。
实施例12:10-((6-氯吡啶-3-基)甲基)-4-硝基-9-氧杂-11,12-二氢咪唑并[2,3-a]双环[3,3,1]壬-3-烯(化合物77)的合成
将1.27g(0.005mol)的2-氯-5-(2-硝基亚甲基-咪唑烷-1-基甲基)-吡啶,30ml的无水乙腈,3ml 25%戊二醛水溶液,催化量的HCl置于50ml的圆底烧瓶中。常温下搅拌,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到淡黄色粉末状纯品,产率为86%。
mp=174.7-175.4℃;1H NMR(400Mz,DMSO-d6):δ8.38(dd,J1=0.6Hz,J2=2.4Hz,1H),7.84(dd,J1=2.4Hz,J2=8.4Hz,1H),7.52(dd,J1=0.6Hz,J2=8.4Hz,1H),5.12(s,1H),5.04-5.05(m,1H),4.97(d,J=15.6Hz,1H),4.71(d,J=15.6Hz,1H),3.62-3.74(m,4H),1.66-1.81(m,4H),1.51-1.55(m,1H),1.32-1.44(m,1H)ppm;13C NMR(100Mz,DMSO-d6):δ156.6,149.7,149.6,139.7,132.9,124.5,105.8,81.7,68.9,51.7,50.0,46.3,28.8,27.2,14.8ppm;HRMS(EI+)计算值C15H17N4O3 35Cl(M+),336.0989;测得值,336.0988。计算值C15H17N4O3 37Cl(M+),338.0960;测得值,338.0968。
实施例13:10-((四氢呋喃-3-基)甲基)-4-硝基-9-氧杂-11,12-二氢咪唑并[2,3-a]双环[3,3,1]壬-3-烯(化合物80)的合成
按照实施例1所述的方法,以0.2mol 3-氯甲基四氢呋喃代替2-氯-5-氯甲基吡啶作为起始原料制备1-((四氢呋喃-3-基)甲基)-2-(-硝基亚甲基)-1-咪唑啉,产率为51%;GC MS(m/s)177(29),99(100),56(9)。
10-((四氢呋喃-3-基)甲基)-4-硝基-9-氧杂-11,12-二氢咪唑并[2,3-a]双环[3,3,1]
壬-3-烯
将1.065g(0.005mol)的1-((四氢呋喃-3-基)甲基)-2-(-硝基亚甲基)-1-咪唑啉,3ml 25%戊二醛水溶液,催化量的HCl置于50ml的圆底烧瓶中。常温下搅拌,TLC跟踪反应。待反应结束后,除去溶剂,柱层析分离得到淡黄色粉末状纯品,产率为36%。
mp=115.3-116.9℃;1H NMR(400Mz,DMSO-d6):δ5.11(s,1H),5.00-5.03(m,1H),4.18(d,J=3.2Hz,2H),4.05-4.25(m,2H),3.85-3.96(m,4H),2.25(m,1H),1.66-1.81(m,4H),1.63-1.64(m,2H),1.57-1.59(m,2H),1.51-1.55(m,1H),1.32-1.44(m,1H)ppm;13C NMR(100Mz,DMSO-d6):δ81.7,80.6,78.5,68.9,50.0,49.7,46.9,44.6,36.8,33.9,28.8,27.2,17.8,14.8ppm;HRMS(EI+)计算值C14H21N3O4(M+),295.1532;测得值,295.1598。
实施例14:本发明化合物的杀虫活性测试
(1)对蚜虫的杀虫活性
蚜虫属于同翅目害虫,具有刺吸口器,是一种常见的农作物害虫。以豆蚜(Aphis craccivora)为测试对象,采用浸渍法测试。
操作过程:准确称量各种样品,分别加入N,N-二甲基甲酰胺配制成10g/L母液,实验时用含0.2mL/L Triton X-100的水溶液将其稀释至500ppm的浓度。待无翅成蚜在豆芽上稳定吸食后,连同豆芽一起浸入浓度为500ppm的药液中,5秒后取出,用吸水纸吸去多余药液,移入干净器皿中于23℃恒温饲养。每浓度设3次重复,对照组为含0.2mL/L Triton X-100的水溶液。处理24小时后,统计试蚜的死亡虫数,并根据公式计算死亡率(%):死亡率(%)=(对照活虫数-处理活虫数)/对照活虫数×100%。结果见下表1-4。
(2)对飞虱的杀虫活性
飞虱属于同翅目害虫,具有刺吸口器,是一种常见的农作物害虫。以褐飞虱(Nilaparvata lugens)为测试对象,采用喷雾法测试。
操作过程:将待测化合物以丙酮为溶剂准确配制成500、250、100、50、25、12.5、6.25、3.13、1.57、0.79ppm溶液,并以丙酮水溶液处理作空白对照。每个处理重复3杯(即重复3次)。用小型手动喷雾器每杯均匀喷雾2ml。施药前6小时每盆接稻飞虱10头。先后共进行了3批次试验。处理24小时后,统计试虫的死亡虫数,并计算死亡率(%)(公式同上)。结果见下表1-4。
(3)对粘虫的杀虫活性
采用浸叶饲喂法。将待测化合物以丙酮为溶剂准确配制成500、250、100、50、25、12.5、6.25、3.13、1.57ppm溶液,并以丙酮水溶液处理作空白对照。将新鲜的玉米叶片在溶液中浸渍3秒,然后在室温下凉干,供试虫取食,24h后检查并计算试虫的死亡率(%,公式同上),每处理使用10试虫,设3次重复。。结果见表1-4。
(4)对小菜蛾的杀虫活性
采用浸叶饲喂法。将新鲜的卷心菜叶片在上述溶液中浸渍3秒,然后在室温下凉干,供试虫取食,24小时后检查并计算试虫的死亡率(%,公式同上),每处理使用10试虫,设3次重复。以清水处理作空白对照。结果见下表1-4。
表1:式(A)化合物的杀虫活性
表2:式(B)化合物的杀虫活性
表3:式(C)化合物的杀虫活性
表4:式(D)化合物的杀虫活性
实施例15:含有本发明化合物的杀虫剂组合物的制备
(a)油状悬浮液
按比例准备以下组分:25%(重量百分比,下同)化合物1-73中任一种化合物;5%聚氧乙烯山梨醇六油酸酯;70%高级脂肪族烃油。将各组分在沙磨中一起研磨,直到固体颗粒降至约5微米以下为止。所得的粘稠悬浮液可直接使用,但也可在水中乳化后使用。
(b)水悬浮液
按比例准备以下组分:25%化合物1-73中任一种化合物;3%水合硅镁土(hydrate attapulgit);10%木质素磺酸钙;0.5%磷酸二氢钠;61.5%水。将各组分在球磨机中一起研磨,直到固体颗粒降至约10微米以下为止。该水悬浮液可直接使用。
(c)饵剂
按比例准备以下组分:0.1-10%化合物1-73中任一种化合物;80%小麦面粉;19.9-10%糖蜜。将这些组分完全混合,按需要形成饵形状。可食用饵可以分散到卫生害虫所侵染的场所,例如家居或工业场所,诸如厨房、医院或商店或户外区域,以通过口服摄入来防治害虫。
本发明提及的所有文献都在本申请中引用作为参考,就如同每一篇文献被单独引用作为参考那样。此外应理解,在阅读了本发明的上述讲授内容之后,本领域技术人员可以对本发明作各种改动或修改,这些等价形式同样落于本申请所附权利要求书所限定的范围。
Claims (9)
1.具有式(C)所示结构的化合物、或者所述化合物的农药学上可接受的盐:
式中:
R1为含氮、氧和/或硫的五元或六元杂环基,卤代的含氮、氧和/或硫的五元或六元杂环基,或者取代或未取代的苯基,其中,所述取代基为选自下组中的一个或多个:卤素,C1–4卤代烷基或C1–4氯代烷氧基;
R3和R4各自独立地为H,C1-6烷基,烯丙基,苄基,C1–4烷氧基-C1–4烷基,C1–4烷氧基-羰基,苯氧羰基,C2–6炔基-羰基,C2–3烯基-羰基,C3–6环烷基-羰基,苯甲酰基,或者被一个或多个选自卤原子、C1–4烷基、C1–4卤代烷基、C1–4烷氧基和C1–4烷基-羰基的取代基所取代的苯甲酰基,呋喃羰基或N,N-二甲基羰基,或者R3和R4共同构成-CH2-CH2-,-CH2-CH2-CH2-或-CH2-XR-CH2-,式中X为杂原子,R为杂原子上的取代基,选自H,C1-6烷基,烯丙基,苄基,苯基,C1–4烷氧基-C1–4烷基,C1–4烷氧基-羰基,苯氧羰基,C2–6炔基-羰基,C2–3烯基-羰基,C3–6环烷基-羰基,苯甲酰基,或者被一个或多个选自卤原子、C1–4卤代烷基、C1-8饱和或不饱和烷基或烷氧基和C1–4烷基-羰基的取代基所取代的苯甲酰基,呋喃羰基或N,N-二甲基羰基;
R5,R6为H,饱和或不饱和C1-4烷基,卤素,C1-8饱和或不饱和烷氧基,卤代C1-4饱和或不饱和烷氧基,C1–4烷基-羰基,C1–8烷基-酯基,C1–4烷基-磺酸酯基,苯基或苄基;
Y为硝基,氰基,三氟甲基,三氟乙酰基或三氟甲磺酰基。
2.如权利要求1所述的化合物、或者所述化合物的农药学上可接受的盐,其特征在于,R1选自:吡啶基、噻唑基、嘧啶基、四氢呋喃基、噁唑基或其卤代物。
3.如权利要求1所述的化合物、或者所述化合物的农药学上可接受的盐,其特征在于,R3和R4为氢或C1-6烷基,或者R3和R4共同构成-CH2-CH2-或-CH2-CH2-CH2-。
4.如权利要求1所述的化合物、或者所述化合物的农药学上可接受的盐,其特征在于,R5,R6为H,饱和或不饱和C1-2烃基,卤素,C1-4饱和或不饱和烷氧基,卤代C1-2饱和或不饱和烷氧基,C1–4烷基-酯基,C1–2烷基-磺酰酯基或三氟甲磺酰酯。
5.如权利要求1所述的化合物、或者所述化合物的农药学上可接受的盐,其特征在于,Y为硝基或氰基。
6.一种农用组合物,它包含:
(a)0.001-99.99重量%的权利要求1-5中任一项所述的化合物或其农药学上可接受的盐、或者它们的组合;以及
(b)农药学上可接受的载体和/或赋形剂。
7.权利要求6所述的农用组合物的用途,其特征在于,用于杀灭或预防农业害虫、卫生害虫和危害动物健康的害虫;或用作用于杀灭或预防农业害虫、卫生害虫和危害动物健康的杀虫剂组合物。
8.权利要求1-5中任一项所述的化合物或其农药学上可接受的盐的制备方法,所述方法包括步骤:
在催化量酸的存在下,于0-60℃下,使式(a)化合物与式(c)化合物反应,从而制得具有式(C)的化合物,
式中,
R1为含氮、氧和/或硫的五元或六元杂环基,卤代的含氮、氧和/或硫的五元或六元杂环基,或者取代或未取代的苯基,其中,所述取代基为选自下组中的一个或多个:卤素,C1–4卤代烷基或C1–4氯代烷氧基;
R3和R4各自独立地为H,C1-6烷基,烯丙基,苄基,C1–4烷氧基-C1–4烷基,C1–4烷氧基-羰基,苯氧羰基,C2–6炔基-羰基,C2–3烯基-羰基,C3–6环烷基-羰基,苯甲酰基,或者被一个或多个选自卤原子、C1–4烷基、C1–4卤代烷基、C1–4烷氧基和C1–4烷基-羰基的取代基所取代的苯甲酰基,呋喃羰基或N,N-二甲基羰基,或者R3和R4共同构成-CH2-CH2-,-CH2-CH2-CH2-或-CH2-XR-CH2-,式中X为杂原子,R为杂原子上的取代基,选自H,C1-6烷基,烯丙基,苄基,苯基,C1–4烷氧基-C1–4烷基,C1–4烷氧基-羰基,苯氧羰基,C2–6炔基-羰基,C2–3烯基-羰基,C3–6环烷基-羰基,苯甲酰基,或者被一个或多个选自卤原子、C1–4卤代烷基、C1-8饱和或不饱和烷基或烷氧基和C1–4烷基-羰基的取代基所取代的苯甲酰基,呋喃羰基或N,N-二甲基羰基;
R5,R6为H,饱和或不饱和C1-4烷基,卤素,C1-8饱和或不饱和烷氧基,卤代C1-4饱和或不饱和烷氧基,C1–4烷基-羰基,C1–8烷基-酯基,C1–4烷基-磺酸酯基,苯基或苄基;
Y为硝基,氰基,三氟甲基,三氟乙酰基或三氟甲磺酰基。
9.如权利要求8所述的方法,其特征在于,所述方法包括步骤:
在乙腈中,于催化量的酸存在下,于10-50℃下,进行如下反应2-24小时,从而获得式(C)的化合物:
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201410690772.2A CN104557963B (zh) | 2008-12-19 | 2008-12-19 | 二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 |
| CN2008102073552A CN101747320B (zh) | 2008-12-19 | 2008-12-19 | 二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 |
| EP09832948.5A EP2377845B1 (en) | 2008-12-19 | 2009-12-18 | Heterocyclic nitrogenous or oxygenous compounds with insecticidal activity formed from dialdehydes and their preparation and uses thereof |
| PCT/CN2009/075693 WO2010069266A1 (zh) | 2008-12-19 | 2009-12-18 | 二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 |
| JP2011541073A JP5771150B2 (ja) | 2008-12-19 | 2009-12-18 | ジアルデヒドを用いて形成された殺虫活性を有する窒素含有複素環式化合物または酸素含有複素環式化合物およびその製造方法 |
| US13/140,968 US8563546B2 (en) | 2008-12-19 | 2009-12-18 | Heterocyclic nitrogenous or oxygenous compounds with insecticidal activity formed from dialdehydes and their preparation and uses thereof |
| BRPI0918359A BRPI0918359B1 (pt) | 2008-12-19 | 2009-12-18 | composto, composição agroquímica, uso da composição agroquímica e método para preparação do composto |
| RU2011129408/04A RU2495023C2 (ru) | 2008-12-19 | 2009-12-18 | Гетероциклические азотсодержащие или кислородсодержащие соединения с инсектицидной активностью, образованные из диальдегидов, и их получение и применения |
| KR1020117016887A KR101392296B1 (ko) | 2008-12-19 | 2009-12-18 | 디알데히드로 구축된 살충활성을 가진 질소 또는 산소를 포함한 헤테로화합물 및 그 제조방법 |
| AU2009328851A AU2009328851C1 (en) | 2008-12-19 | 2009-12-18 | Heterocyclic nitrogenous or oxygenous compounds with insecticidal activity formed from dialdehydes and their preparation and uses thereof |
| IL213656A IL213656A (en) | 2008-12-19 | 2011-06-19 | Nitrogenic or oxidative heterocyclic compounds formed from dialdehydes with insecticide activity, manner of preparation and use |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2008102073552A CN101747320B (zh) | 2008-12-19 | 2008-12-19 | 二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 |
Related Child Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201310398752.3A Division CN103518745B (zh) | 2008-12-19 | 2008-12-19 | 二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 |
| CN201410690772.2A Division CN104557963B (zh) | 2008-12-19 | 2008-12-19 | 二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN101747320A CN101747320A (zh) | 2010-06-23 |
| CN101747320B true CN101747320B (zh) | 2013-10-16 |
Family
ID=42268331
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2008102073552A Active CN101747320B (zh) | 2008-12-19 | 2008-12-19 | 二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 |
Country Status (10)
| Country | Link |
|---|---|
| US (1) | US8563546B2 (zh) |
| EP (1) | EP2377845B1 (zh) |
| JP (1) | JP5771150B2 (zh) |
| KR (1) | KR101392296B1 (zh) |
| CN (1) | CN101747320B (zh) |
| AU (1) | AU2009328851C1 (zh) |
| BR (1) | BRPI0918359B1 (zh) |
| IL (1) | IL213656A (zh) |
| RU (1) | RU2495023C2 (zh) |
| WO (1) | WO2010069266A1 (zh) |
Families Citing this family (129)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102093389B (zh) * | 2009-12-09 | 2014-11-19 | 华东理工大学 | 双联和氧桥杂环新烟碱化合物及其制备方法 |
| CN102197808B (zh) * | 2011-03-31 | 2013-10-30 | 上海生农生化制品有限公司 | 环氧虫啶水分散粒剂及其制备方法 |
| CN102197804A (zh) * | 2011-03-31 | 2011-09-28 | 上海生农生化制品有限公司 | 环氧虫啶悬浮剂及其制备方法 |
| CN102197807A (zh) * | 2011-03-31 | 2011-09-28 | 上海生农生化制品有限公司 | 环氧虫啶可湿性粉剂及其制备方法 |
| CN102326583B (zh) * | 2011-07-19 | 2013-09-18 | 陕西上格之路生物科学有限公司 | 一种含环氧虫啶和有机磷类杀虫剂的杀虫组合物 |
| CN102293208B (zh) * | 2011-08-29 | 2013-05-15 | 陕西上格之路生物科学有限公司 | 一种含环氧虫啶和噻嗪酮的杀虫组合物 |
| CN102396472A (zh) * | 2011-11-26 | 2012-04-04 | 陕西美邦农药有限公司 | 一种含环氧虫啶和激素类的杀虫组合物 |
| CN102487952B (zh) * | 2011-12-04 | 2013-10-23 | 陕西上格之路生物科学有限公司 | 一种含环氧虫啶和生物源杀虫剂的杀虫组合物 |
| CN103125481A (zh) * | 2011-12-05 | 2013-06-05 | 上海生农生化制品有限公司 | 环氧虫啶油悬剂及其制备方法 |
| CN102428949A (zh) * | 2011-12-21 | 2012-05-02 | 陕西美邦农药有限公司 | 一种含环氧虫啶和新烟碱类的杀虫组合物 |
| CN103242323B (zh) * | 2012-02-06 | 2017-05-10 | 华东理工大学 | 具有杀虫活性的含氮(硫)桥环化合物、制备及用途 |
| CN103238609A (zh) * | 2012-02-09 | 2013-08-14 | 陕西美邦农药有限公司 | 一种含环氧虫啶的杀虫组合物 |
| CN102440252B (zh) * | 2012-02-13 | 2013-09-18 | 陕西上格之路生物科学有限公司 | 一种含环氧虫啶和吡唑类杀虫剂的杀虫组合物 |
| WO2013156331A1 (en) | 2012-04-16 | 2013-10-24 | Basf Se | Synergistic compositions comprising pyraclostrobin and an insecticidal compound |
| CN102657189B (zh) * | 2012-05-08 | 2014-04-30 | 陕西上格之路生物科学有限公司 | 一种含环氧虫啶的杀虫组合物 |
| WO2014005982A1 (de) | 2012-07-05 | 2014-01-09 | Bayer Cropscience Ag | Insektizide und fungizide wirkstoffkombinationen |
| CN102771510B (zh) * | 2012-07-31 | 2014-06-04 | 陕西上格之路生物科学有限公司 | 一种含噻唑膦的杀虫组合物 |
| MX2015004175A (es) | 2012-10-01 | 2015-06-10 | Basf Se | Uso de compuestos de n-tio-antranilamida en plantas cultivadas. |
| US20150237858A1 (en) | 2012-10-01 | 2015-08-27 | Basf Se | Method of controlling ryanodine-modulator insecticide resistant insects |
| WO2014053403A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of controlling insecticide resistant insects |
| WO2014053401A2 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of improving plant health |
| US20150250175A1 (en) | 2012-10-01 | 2015-09-10 | Basf Se | Pesticidally active mixtures comprising anthranilamide compounds |
| WO2014053407A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| CN104768377A (zh) | 2012-10-01 | 2015-07-08 | 巴斯夫欧洲公司 | 包含邻氨基苯甲酰胺类化合物的农药活性混合物 |
| WO2014079820A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Use of anthranilamide compounds for reducing insect-vectored viral infections |
| CA2894023A1 (en) | 2012-12-14 | 2014-06-19 | Basf Se | Malononitrile compounds for controlling animal pests |
| BR112015015503A2 (pt) | 2012-12-27 | 2017-07-11 | Basf Se | composto substituído, composição veterinária, utilização de um composto, método para o controle de pragas de invertebrados e para o tratamento ou proteção de um animal e material de propagação do vegetal |
| CN104054726B (zh) * | 2013-03-20 | 2018-10-26 | 上海生农生化制品股份有限公司 | 苯氧威和环氧虫啶杀虫组合物 |
| WO2014170300A1 (en) | 2013-04-19 | 2014-10-23 | Basf Se | N-substituted acyl-imino-pyridine compounds and derivatives for combating animal pests |
| BR112015031439A2 (pt) | 2013-06-21 | 2017-07-25 | Basf Se | métodos para o combate ou controle das pragas, para o tratamento, prevenção e proteção de culturas de soja, para o controle e proteção do material de propagação dos vegetais de soja, para o combate ou controle das pragas e utilização de um composto de fórmula i |
| EP3022185B1 (en) | 2013-07-15 | 2017-09-06 | Basf Se | Pesticide compounds |
| CN103430952B (zh) * | 2013-07-18 | 2015-03-04 | 张强 | 一种水溶性甲基硫菌灵制剂及其生产工艺 |
| EA201600270A1 (ru) | 2013-09-19 | 2016-08-31 | Басф Се | N-ацилимино гетероциклические соединения |
| WO2015055554A1 (de) | 2013-10-14 | 2015-04-23 | Bayer Cropscience Ag | Wirkstoff für die saatgut- und bodenbehandlung |
| UY35772A (es) | 2013-10-14 | 2015-05-29 | Bayer Cropscience Ag | Nuevos compuestos plaguicidas |
| WO2015055497A1 (en) | 2013-10-16 | 2015-04-23 | Basf Se | Substituted pesticidal pyrazole compounds |
| CN106061254B (zh) | 2013-10-18 | 2019-04-05 | 巴斯夫农业化学品有限公司 | 农药活性羧酰胺衍生物在土壤和种子施用和处理方法中的用途 |
| JP6472444B2 (ja) | 2013-10-23 | 2019-02-20 | バイエル・クロップサイエンス・アクチェンゲゼルシャフト | 有害生物防除剤としての置換されているキノキサリン誘導体 |
| WO2015091645A1 (en) | 2013-12-18 | 2015-06-25 | Basf Se | Azole compounds carrying an imine-derived substituent |
| EP3083581A1 (en) | 2013-12-18 | 2016-10-26 | Basf Se | N-substituted imino heterocyclic compounds |
| CN103719115B (zh) * | 2013-12-25 | 2015-08-05 | 江苏龙灯化学有限公司 | 一种杀虫组合物 |
| CN104738052A (zh) * | 2013-12-25 | 2015-07-01 | 南京华洲药业有限公司 | 含三氟甲吡醚和环氧虫啶的复合杀虫组合物及其用途 |
| WO2015104422A1 (en) | 2014-01-13 | 2015-07-16 | Basf Se | Dihydrothiophene compounds for controlling invertebrate pests |
| TW201612161A (en) | 2014-01-20 | 2016-04-01 | Bayer Cropscience Ag | Quinoline derivatives as insecticides and acaricides |
| US10059706B2 (en) | 2014-05-08 | 2018-08-28 | Bayer Cropscience Aktiengesellschaft | Pyrazolopyridine sulfonamides as nematicides |
| CN106715424B (zh) | 2014-06-05 | 2020-07-14 | 拜耳作物科学股份公司 | 作为农药的双环化合物 |
| WO2016001119A1 (de) | 2014-07-01 | 2016-01-07 | Bayer Cropscience Aktiengesellschaft | Insektizide und fungizide wirkstoffkombinationen |
| US10208015B2 (en) | 2014-07-15 | 2019-02-19 | Bayer Animal Health Gmbh | Aryl-triazolyl pyridines as pest control agents |
| EP3204390B1 (en) | 2014-10-06 | 2019-06-05 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| WO2016055096A1 (en) | 2014-10-07 | 2016-04-14 | Bayer Cropscience Ag | Method for treating rice seed |
| CN105557709B (zh) * | 2014-10-11 | 2018-08-14 | 南京农业大学 | 八元氧桥杂环化合物作为杀虫剂增效剂的用途 |
| BR112017009513A2 (pt) | 2014-11-06 | 2018-02-06 | Basf Se | utilização de um composto heterobicíclico, utilização dos compostos i, compostos, composição agrícola ou veterinária, método para o combate ou controle de pragas, método para a proteção de culturas e sementes |
| EP3081085A1 (en) | 2015-04-14 | 2016-10-19 | Bayer CropScience AG | Method for improving earliness in cotton |
| CN107207443A (zh) | 2015-02-06 | 2017-09-26 | 巴斯夫欧洲公司 | 作为硝化抑制剂的吡唑化合物 |
| EP3255990B1 (en) | 2015-02-11 | 2020-06-24 | Basf Se | Pesticidal mixture comprising a pyrazole compound, an insecticide and a fungicide |
| WO2016162371A1 (en) | 2015-04-07 | 2016-10-13 | Basf Agrochemical Products B.V. | Use of an insecticidal carboxamide compound against pests on cultivated plants |
| EP2910126A1 (en) | 2015-05-05 | 2015-08-26 | Bayer CropScience AG | Active compound combinations having insecticidal properties |
| JP6806981B2 (ja) | 2015-05-12 | 2021-01-06 | ビーエイエスエフ・ソシエタス・エウロパエアBasf Se | 硝化阻害剤としてのチオエーテル化合物 |
| WO2016198611A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino heterocyclic compounds |
| WO2016198613A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino compounds |
| WO2017016883A1 (en) | 2015-07-24 | 2017-02-02 | Basf Se | Process for preparation of cyclopentene compounds |
| US11142514B2 (en) | 2015-10-02 | 2021-10-12 | Basf Se | Imino compounds with a 2-chloropyrimidin-5-yl substituent as pest-control agents |
| CN105330593A (zh) * | 2015-11-23 | 2016-02-17 | 上海晋景化学有限公司 | 一种制备烯啶虫胺的改进方法 |
| CN113303339A (zh) | 2015-11-30 | 2021-08-27 | 巴斯夫欧洲公司 | 顺式-茉莉酮和解淀粉芽孢杆菌的混合物 |
| US20190059381A1 (en) | 2016-02-19 | 2019-02-28 | Basf Se | Method for controlling pests of soybean, corn, and cotton plants |
| BR112018068034A2 (pt) | 2016-03-09 | 2019-01-08 | Basf Se | compostos espiro da fórmula i, composição, composição agrícola para combater pragas animais, método de combate ou controle de pragas invertebradas, método de proteção de plantas, semente e uso dos compostos |
| WO2017153218A1 (en) | 2016-03-11 | 2017-09-14 | Basf Se | Method for controlling pests of plants |
| PE20181898A1 (es) | 2016-04-01 | 2018-12-11 | Basf Se | Compuestos biciclicos |
| WO2017198588A1 (en) | 2016-05-18 | 2017-11-23 | Basf Se | Capsules comprising benzylpropargylethers for use as nitrification inhibitors |
| US10750750B2 (en) | 2016-07-11 | 2020-08-25 | Covestro Llc | Aqueous compositions for treating seeds, seeds treated therewith, and methods for treating seeds |
| US10653135B2 (en) | 2016-07-11 | 2020-05-19 | Covestro Llc | Methods for treating seeds with an aqueous composition and seeds treated therewith |
| US10653136B2 (en) | 2016-07-11 | 2020-05-19 | Covestro Llc | Aqueous compositions for treating seeds, seeds treated therewith, and methods for treating seeds |
| CN106577694A (zh) * | 2016-10-24 | 2017-04-26 | 东莞市联洲知识产权运营管理有限公司 | 一种基于环氧虫啶和苦参碱的高效广谱增效农药组合物 |
| KR20190092539A (ko) | 2016-12-16 | 2019-08-07 | 바스프 에스이 | 살충 화합물 |
| WO2018162312A1 (en) | 2017-03-10 | 2018-09-13 | Basf Se | Spirocyclic derivatives |
| WO2018166855A1 (en) | 2017-03-16 | 2018-09-20 | Basf Se | Heterobicyclic substituted dihydroisoxazoles |
| KR102596592B1 (ko) | 2017-03-28 | 2023-10-31 | 바스프 에스이 | 살충 화합물 |
| CN110461854A (zh) | 2017-03-31 | 2019-11-15 | 巴斯夫欧洲公司 | 制备手性2,3-二氢噻唑并[3,2-a]嘧啶-4-鎓化合物的方法 |
| WO2018192793A1 (en) | 2017-04-20 | 2018-10-25 | Basf Se | Substituted rhodanine derivatives |
| EP3615530B1 (en) | 2017-04-26 | 2021-04-14 | Basf Se | Substituted succinimide derivatives as pesticides |
| CN110546152B (zh) | 2017-05-10 | 2023-09-08 | 巴斯夫欧洲公司 | 双环农药化合物 |
| WO2018224455A1 (en) | 2017-06-07 | 2018-12-13 | Basf Se | Substituted cyclopropyl derivatives |
| EP3638677A1 (en) | 2017-06-16 | 2020-04-22 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| EP3642203A1 (en) | 2017-06-19 | 2020-04-29 | Basf Se | Substituted pyrimidinium compounds and derivatives for combating animal pests |
| WO2018234488A1 (en) | 2017-06-23 | 2018-12-27 | Basf Se | Substituted cyclopropyl derivatives |
| WO2019007888A1 (de) | 2017-07-06 | 2019-01-10 | Bayer Aktiengesellschaft | Insektizide wirkstoffkombinationen |
| WO2019007891A1 (de) | 2017-07-06 | 2019-01-10 | Bayer Aktiengesellschaft | Insektizide wirkstoffkombinationen |
| WO2019042932A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | METHOD FOR CONTROLLING RICE PARASITES IN RICE |
| EP3453706A1 (en) | 2017-09-08 | 2019-03-13 | Basf Se | Pesticidal imidazole compounds |
| US11399543B2 (en) | 2017-10-13 | 2022-08-02 | Basf Se | Substituted 1,2,3,5-tetrahydroimidazo[1,2-a]pyrimidiniumolates for combating animal pests |
| JP6933413B2 (ja) | 2017-11-03 | 2021-09-08 | 華南農業大学 | 窒素含有縮合三環式化合物及び農業・林業殺虫剤としての使用 |
| WO2019121143A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Substituted cyclopropyl derivatives |
| UA127604C2 (uk) | 2017-12-21 | 2023-11-01 | Басф Се | Пестицидні сполуки |
| US20200397001A1 (en) | 2018-01-05 | 2020-12-24 | Basf Se | Control of pests of soybean plants with mesoionic compounds |
| AU2019211978B2 (en) | 2018-01-09 | 2024-08-22 | Basf Se | Silylethynyl hetaryl compounds as nitrification inhibitors |
| WO2019137995A1 (en) | 2018-01-11 | 2019-07-18 | Basf Se | Novel pyridazine compounds for controlling invertebrate pests |
| PE20211753A1 (es) | 2018-02-28 | 2021-09-06 | Basf Se | Uso de alcoxipirazoles como inhibidores de la nitrificacion |
| WO2019166558A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of pyrazole propargyl ethers as nitrification inhibitors |
| KR102730587B1 (ko) | 2018-02-28 | 2024-11-14 | 바스프 에스이 | 질화작용 저해제로서의 n-관능화 알콕시 피라졸 화합물의 용도 |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019185413A1 (en) | 2018-03-27 | 2019-10-03 | Basf Se | Pesticidal substituted cyclopropyl derivatives |
| WO2019211106A1 (en) | 2018-04-30 | 2019-11-07 | Basf Se | Control of pests of soybean plants with mesoionic compounds |
| KR102727205B1 (ko) | 2018-05-15 | 2024-11-06 | 바스프 에스이 | 벤즈피리목산 및 옥사조술필을 포함하는 혼합물 및 이의 용도 및 이의 적용 방법 |
| WO2019224092A1 (en) | 2018-05-22 | 2019-11-28 | Basf Se | Pesticidally active c15-derivatives of ginkgolides |
| WO2020002472A1 (en) | 2018-06-28 | 2020-01-02 | Basf Se | Use of alkynylthiophenes as nitrification inhibitors |
| PL3826983T3 (pl) | 2018-07-23 | 2024-09-09 | Basf Se | Zastosowanie podstawionych 2-tiazolin jako inhibitorów nitryfikacji |
| CN112424147B (zh) | 2018-07-23 | 2023-06-30 | 巴斯夫欧洲公司 | 取代噻唑烷化合物作为硝化抑制剂的用途 |
| EP3613736A1 (en) | 2018-08-22 | 2020-02-26 | Basf Se | Substituted glutarimide derivatives |
| EP3628158A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| EP3628157A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| EP3628156A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method for controlling pests of sugarcane, citrus, rapeseed, and potato plants |
| AU2019348280A1 (en) | 2018-09-28 | 2021-04-22 | Basf Se | Method of controlling pests by seed treatment application of a mesoionic compound or mixture thereof |
| EP3643705A1 (en) | 2018-10-24 | 2020-04-29 | Basf Se | Pesticidal compounds |
| US12459934B2 (en) | 2018-11-28 | 2025-11-04 | Basf Se | Pesticidal compounds |
| WO2020126591A1 (en) | 2018-12-18 | 2020-06-25 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| EP3696177A1 (en) | 2019-02-12 | 2020-08-19 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP3769623A1 (en) | 2019-07-22 | 2021-01-27 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| BR112021019416A2 (pt) | 2019-05-29 | 2021-12-07 | Basf Se | Compostos, composição, métodos de proteção de safras e de combate, controle, prevenção ou proteção contra infestações, método não terapêutico de tratamento de animais infestados, semente e uso |
| EP3766879A1 (en) | 2019-07-19 | 2021-01-20 | Basf Se | Pesticidal pyrazole derivatives |
| CN111165505A (zh) * | 2020-02-24 | 2020-05-19 | 华东理工大学 | 八元氧桥杂环化合物作为蜜蜂选择性杀虫剂增效剂的用途 |
| CN112010873A (zh) * | 2020-08-20 | 2020-12-01 | 上海生农生化制品股份有限公司 | 一种环氧虫啶的合成方法 |
| EP4284171B1 (en) | 2021-01-28 | 2024-11-13 | Specialty Operations France | Method for treating rice seed with improved retention of agrochemical, micronutrient and colorant |
| EP4288398A1 (en) | 2021-02-02 | 2023-12-13 | Basf Se | Synergistic action of dcd and alkoxypyrazoles as nitrification inhibitors |
| US20240270658A1 (en) | 2021-05-21 | 2024-08-15 | Basf Se | Use of an N-Functionalized Alkoxy Pyrazole Compound as Nitrification Inhibitor |
| US20240351959A1 (en) | 2021-05-21 | 2024-10-24 | Basf Se | Use of ethynylpyridine compounds as nitrification inhibitors |
| CA3223077A1 (en) | 2021-06-21 | 2022-12-29 | Barbara Nave | Metal-organic frameworks with pyrazole-based building blocks |
| WO2023203066A1 (en) | 2022-04-21 | 2023-10-26 | Basf Se | Synergistic action as nitrification inhibitors of dcd oligomers with alkoxypyrazole and its oligomers |
| AU2023317620A1 (en) | 2022-08-02 | 2025-02-13 | Basf Se | Pyrazolo pesticidal compounds |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101016277A (zh) * | 2007-02-12 | 2007-08-15 | 江苏天容集团股份有限公司 | 第二代新烟碱类杀虫剂jt-l001及其化学合成方法 |
Family Cites Families (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS4819934B1 (zh) * | 1968-10-08 | 1973-06-18 | ||
| EP0192060B1 (de) | 1985-02-04 | 1991-09-18 | Nihon Bayer Agrochem K.K. | Heterocyclische Verbindungen |
| JPH0717621B2 (ja) | 1986-03-07 | 1995-03-01 | 日本バイエルアグロケム株式会社 | 新規ヘテロ環式化合物 |
| US4704563A (en) | 1986-05-09 | 1987-11-03 | General Electric Company | Fluorescent lamp operating circuit |
| DE3638121A1 (de) | 1986-05-30 | 1987-12-03 | Bayer Ag | 1,2,3,6-tetrahydro-5-nitro-pyrimidin-derivate |
| JPH0710865B2 (ja) | 1987-06-26 | 1995-02-08 | 日本バイエルアグロケム株式会社 | ニトロ置換ヘテロ環式化合物及び殺虫剤 |
| JP2583429B2 (ja) | 1987-11-06 | 1997-02-19 | 日本バイエルアグロケム株式会社 | イミダゾリン類及び殺虫剤 |
| JP2610988B2 (ja) | 1989-03-09 | 1997-05-14 | 日本バイエルアグロケム 株式会社 | 新規ヘテロ環式化合物及び殺虫剤 |
| TW240163B (en) | 1992-07-22 | 1995-02-11 | Syngenta Participations Ag | Oxadiazine derivatives |
| JPH07242633A (ja) | 1994-03-03 | 1995-09-19 | Ishihara Sangyo Kaisha Ltd | 5−アミノエチルアミノメチル−2−クロロピリジンの製造方法 |
| JP4136000B2 (ja) | 1994-06-03 | 2008-08-20 | 三井化学株式会社 | 殺虫性テトラヒドロフラン系化合物 |
| JPH08259568A (ja) | 1995-03-27 | 1996-10-08 | Mitsui Toatsu Chem Inc | 殺虫性テトラヒドロピリミジン誘導体 |
| JP3722512B2 (ja) | 1995-04-20 | 2005-11-30 | 三井化学株式会社 | 殺虫性5−{(テトラヒドロ−3−フラニル)メチル}−4−ニトロイミノパーヒドロ−1,3,5−オキサジアジン誘導体 |
| DE19908447A1 (de) | 1999-02-26 | 2000-08-31 | Bayer Ag | Verfahren zur Herstellung von 2-Chlor-5-chlormethylthiazol |
| EP1572654A4 (en) * | 2002-12-19 | 2006-08-09 | Dow Agrosciences Llc | AS PESTICIDES SUITABLE COMPOUNDS |
| UA79571C2 (en) * | 2003-12-04 | 2007-06-25 | Basf Ag | Metod for the protection of seeds from soil pests comprising |
| CN1295228C (zh) | 2004-11-23 | 2007-01-17 | 华东理工大学 | 硝基亚甲基衍生物及其用途 |
| WO2007101369A1 (fr) | 2006-03-09 | 2007-09-13 | East China University Of Science And Technology | Méthode de préparation et utilisation de composés présentant une action biocide |
| CN101492444B (zh) | 2008-01-23 | 2012-07-04 | 华东理工大学 | 具有杀虫活性的含氮杂环化合物、其制备及用途 |
| CN102093389B (zh) * | 2009-12-09 | 2014-11-19 | 华东理工大学 | 双联和氧桥杂环新烟碱化合物及其制备方法 |
-
2008
- 2008-12-19 CN CN2008102073552A patent/CN101747320B/zh active Active
-
2009
- 2009-12-18 EP EP09832948.5A patent/EP2377845B1/en active Active
- 2009-12-18 BR BRPI0918359A patent/BRPI0918359B1/pt active IP Right Grant
- 2009-12-18 RU RU2011129408/04A patent/RU2495023C2/ru active
- 2009-12-18 US US13/140,968 patent/US8563546B2/en not_active Expired - Fee Related
- 2009-12-18 JP JP2011541073A patent/JP5771150B2/ja active Active
- 2009-12-18 KR KR1020117016887A patent/KR101392296B1/ko active Active
- 2009-12-18 WO PCT/CN2009/075693 patent/WO2010069266A1/zh not_active Ceased
- 2009-12-18 AU AU2009328851A patent/AU2009328851C1/en active Active
-
2011
- 2011-06-19 IL IL213656A patent/IL213656A/en active IP Right Grant
Patent Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101016277A (zh) * | 2007-02-12 | 2007-08-15 | 江苏天容集团股份有限公司 | 第二代新烟碱类杀虫剂jt-l001及其化学合成方法 |
Also Published As
| Publication number | Publication date |
|---|---|
| AU2009328851A1 (en) | 2011-08-04 |
| JP5771150B2 (ja) | 2015-08-26 |
| EP2377845A4 (en) | 2012-12-26 |
| WO2010069266A1 (zh) | 2010-06-24 |
| KR101392296B1 (ko) | 2014-05-07 |
| CN101747320A (zh) | 2010-06-23 |
| US20110269751A1 (en) | 2011-11-03 |
| BRPI0918359B1 (pt) | 2018-11-06 |
| JP2012512191A (ja) | 2012-05-31 |
| KR20110097970A (ko) | 2011-08-31 |
| RU2011129408A (ru) | 2013-01-27 |
| BRPI0918359A2 (pt) | 2015-12-15 |
| IL213656A0 (en) | 2011-07-31 |
| US8563546B2 (en) | 2013-10-22 |
| EP2377845A1 (en) | 2011-10-19 |
| IL213656A (en) | 2015-11-30 |
| RU2495023C2 (ru) | 2013-10-10 |
| AU2009328851C1 (en) | 2012-12-06 |
| EP2377845B1 (en) | 2015-01-28 |
| AU2009328851B2 (en) | 2012-04-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101747320B (zh) | 二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 | |
| CN102093389B (zh) | 双联和氧桥杂环新烟碱化合物及其制备方法 | |
| CN101492444B (zh) | 具有杀虫活性的含氮杂环化合物、其制备及用途 | |
| CN102070607A (zh) | 具有杀虫活性的吡啶氮氧化物类新烟碱化合物及其用途 | |
| CN101768161B (zh) | 具有杀虫活性的含氮杂环化合物、其制备及用途 | |
| CN101906096B (zh) | 3,4-二氢吡啶-2-酮杂环类化合物及其用途 | |
| CN102464653A (zh) | 具有杀虫活性的偶氮杂环化合物、其制备及用途 | |
| CN102863446B (zh) | 具有杀虫活性的吡咯及二氢吡咯稠环化合物的制备与用途 | |
| CN101875653B (zh) | 1,2,3-3h吡啶杂环化合物的制备及用途 | |
| CN103242323B (zh) | 具有杀虫活性的含氮(硫)桥环化合物、制备及用途 | |
| CN104557963B (zh) | 二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 | |
| CN103518745B (zh) | 二醛构建的具有杀虫活性的含氮或氧杂环化合物及其制备方法 | |
| CN103570729B (zh) | 环烯酮构建的二环新烟碱化合物及其制备方法和应用 | |
| CN105461724B (zh) | 具有杀虫活性的含氮桥环化合物及其制备和用途 | |
| CN110256404A (zh) | 具有杀虫活性的双烯化合物、其制备及用途 | |
| CN106632300B (zh) | 具有杀虫活性的多取代异噁唑类化合物及其制备方法 | |
| WO2016055029A1 (zh) | 八元氧桥杂环化合物作为杀虫剂增效剂的用途 | |
| CN103864795A (zh) | 含二硫戊环的顺式新烟碱化合物及其制备方法和应用 | |
| CN105017257A (zh) | 四氢茚并吡咯并咪唑的衍生物及其制备方法和用途 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| EE01 | Entry into force of recordation of patent licensing contract |
Assignee: Shengnong Biological-Chemical Products Co., Ltd., Shanghai Assignor: East China University of Science and Technology Contract record no.: 2012310000083 Denomination of invention: Dialdehyde-built disinsection activity-having nitrogen or oxygen-containing heterocyclic compound and preparation method License type: Exclusive License Open date: 20100623 Record date: 20120611 |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant |