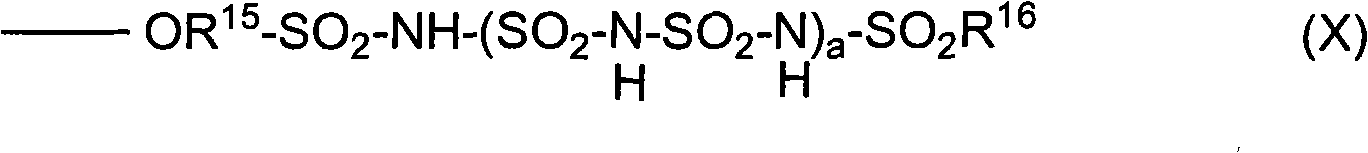CN101688052A - Aqueous dispersion of conductive polymer comprising inorganic nanoparticles - Google Patents
Aqueous dispersion of conductive polymer comprising inorganic nanoparticles Download PDFInfo
- Publication number
- CN101688052A CN101688052A CN200880023383A CN200880023383A CN101688052A CN 101688052 A CN101688052 A CN 101688052A CN 200880023383 A CN200880023383 A CN 200880023383A CN 200880023383 A CN200880023383 A CN 200880023383A CN 101688052 A CN101688052 A CN 101688052A
- Authority
- CN
- China
- Prior art keywords
- composition
- layer
- highly fluorinated
- poly
- ester
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Images
Classifications
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y20/00—Nanooptics, e.g. quantum optics or photonic crystals
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B82—NANOTECHNOLOGY
- B82Y—SPECIFIC USES OR APPLICATIONS OF NANOSTRUCTURES; MEASUREMENT OR ANALYSIS OF NANOSTRUCTURES; MANUFACTURE OR TREATMENT OF NANOSTRUCTURES
- B82Y30/00—Nanotechnology for materials or surface science, e.g. nanocomposites
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08L—COMPOSITIONS OF MACROMOLECULAR COMPOUNDS
- C08L65/00—Compositions of macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain; Compositions of derivatives of such polymers
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B1/00—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors
- H01B1/06—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors mainly consisting of other non-metallic substances
- H01B1/12—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors mainly consisting of other non-metallic substances organic substances
- H01B1/124—Intrinsically conductive polymers
- H01B1/127—Intrinsically conductive polymers comprising five-membered aromatic rings in the main chain, e.g. polypyrroles, polythiophenes
-
- H—ELECTRICITY
- H01—ELECTRIC ELEMENTS
- H01B—CABLES; CONDUCTORS; INSULATORS; SELECTION OF MATERIALS FOR THEIR CONDUCTIVE, INSULATING OR DIELECTRIC PROPERTIES
- H01B1/00—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors
- H01B1/06—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors mainly consisting of other non-metallic substances
- H01B1/12—Conductors or conductive bodies characterised by the conductive materials; Selection of materials as conductors mainly consisting of other non-metallic substances organic substances
- H01B1/124—Intrinsically conductive polymers
- H01B1/128—Intrinsically conductive polymers comprising six-membered aromatic rings in the main chain, e.g. polyanilines, polyphenylenes
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/10—Organic polymers or oligomers
- H10K85/111—Organic polymers or oligomers comprising aromatic, heteroaromatic, or aryl chains, e.g. polyaniline, polyphenylene or polyphenylene vinylene
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08K—Use of inorganic or non-macromolecular organic substances as compounding ingredients
- C08K3/00—Use of inorganic substances as compounding ingredients
- C08K3/18—Oxygen-containing compounds, e.g. metal carbonyls
- C08K3/20—Oxides; Hydroxides
- C08K3/22—Oxides; Hydroxides of metals
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2102/00—Constructional details relating to the organic devices covered by this subclass
- H10K2102/301—Details of OLEDs
- H10K2102/331—Nanoparticles used in non-emissive layers, e.g. in packaging layer
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/17—Carrier injection layers
Landscapes
- Chemical & Material Sciences (AREA)
- Engineering & Computer Science (AREA)
- Nanotechnology (AREA)
- Physics & Mathematics (AREA)
- Crystallography & Structural Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biophysics (AREA)
- Optics & Photonics (AREA)
- Materials Engineering (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Composite Materials (AREA)
- Condensed Matter Physics & Semiconductors (AREA)
- General Physics & Mathematics (AREA)
- Health & Medical Sciences (AREA)
- Compositions Of Macromolecular Compounds (AREA)
- Polyoxymethylene Polymers And Polymers With Carbon-To-Carbon Bonds (AREA)
- Manufacture Of Macromolecular Shaped Articles (AREA)
Abstract
本发明涉及导电聚合物组合物及其在电子器件中的应用。所述组合物包含掺入有至少一种高度氟化酸聚合物的至少一种导电聚合物的含水分散体,以及无机纳米颗粒。
This invention relates to conductive polymer compositions and their application in electronic devices. The compositions comprise an aqueous dispersion of at least one conductive polymer incorporated with at least one highly fluorinated acid polymer, and inorganic nanoparticles.
Description
发明背景Background of the invention
发明领域 field of invention
本发明整体涉及包含无机纳米颗粒的导电聚合物组合物及其在电子器件中的应用。The present invention generally relates to conductive polymer compositions comprising inorganic nanoparticles and their use in electronic devices.
背景技术 Background technique
电子器件限定了一类包括活性层的产品。有机电子器件具有至少一个有机活性层。此类器件可将电能转换成辐射(例如发光二极管)、通过电子方法探测信号、将辐射转换成电能(例如光伏电池),或包括一个或多个有机半导体层。Electronic devices define a class of products that include an active layer. Organic electronic devices have at least one organic active layer. Such devices may convert electrical energy to radiation (eg, light emitting diodes), detect signals electronically, convert radiation into electrical energy (eg, photovoltaic cells), or include one or more organic semiconductor layers.
有机发光二极管(OLED)为有机电子器件,其具有能够电致发光的有机层。包含导电聚合物的OLED可具有以下构造:Organic light emitting diodes (OLEDs) are organic electronic devices having organic layers capable of electroluminescence. OLEDs comprising conductive polymers can have the following configurations:
阳极/缓冲层/EL材料/阴极Anode/buffer layer/EL material/cathode
并在电极之间具有附加层。阳极通常为能够在EL材料中注入空穴的任何材料,例如氧化铟锡(ITO)。可任选地在玻璃或塑料基底上支撑阳极。EL材料包括荧光化合物、荧光和磷光金属络合物、共轭聚合物、以及它们的混合物。阴极通常为能够在EL材料中注入电子的任何材料(例如Ca或Ba)。具有在10-3至10-7S/cm范围内的低导电率的导电聚合物通常用作与导电无机氧化物阳极(例如ITO)直接接触的缓冲层。and have additional layers between the electrodes. The anode is typically any material capable of injecting holes into the EL material, such as indium tin oxide (ITO). The anode can optionally be supported on a glass or plastic substrate. EL materials include fluorescent compounds, fluorescent and phosphorescent metal complexes, conjugated polymers, and mixtures thereof. The cathode is generally any material (eg Ca or Ba) capable of injecting electrons into the EL material. Conductive polymers with low electrical conductivity in the range of 10 −3 to 10 −7 S/cm are generally used as buffer layers in direct contact with conductive inorganic oxide anodes such as ITO.
改善的缓冲层材料是一直需要的。There is a continuing need for improved cushioning layer materials.
发明概述Summary of the invention
本发明提供了一种组合物,该组合物包含掺杂了至少一种高度氟化酸聚合物的至少一种导电聚合物的含水分散体,并具有分散在其中的无机纳米颗粒。The present invention provides a composition comprising an aqueous dispersion of at least one conductive polymer doped with at least one highly fluorinated acid polymer and having inorganic nanoparticles dispersed therein.
在另一个实施方案中,提供了一种由上述组合物形成的薄膜。In another embodiment, there is provided a film formed from the composition described above.
在另一个实施方案中,提供了电子器件,这些电子器件具有至少一个包含上述薄膜的层。In another embodiment, electronic devices are provided having at least one layer comprising the above-described thin film.
附图简述Brief description of the drawings
附图以举例的方式示出本发明,但附图对本发明不构成任何限制。The accompanying drawings show the present invention by way of example, but the accompanying drawings do not constitute any limitation to the present invention.
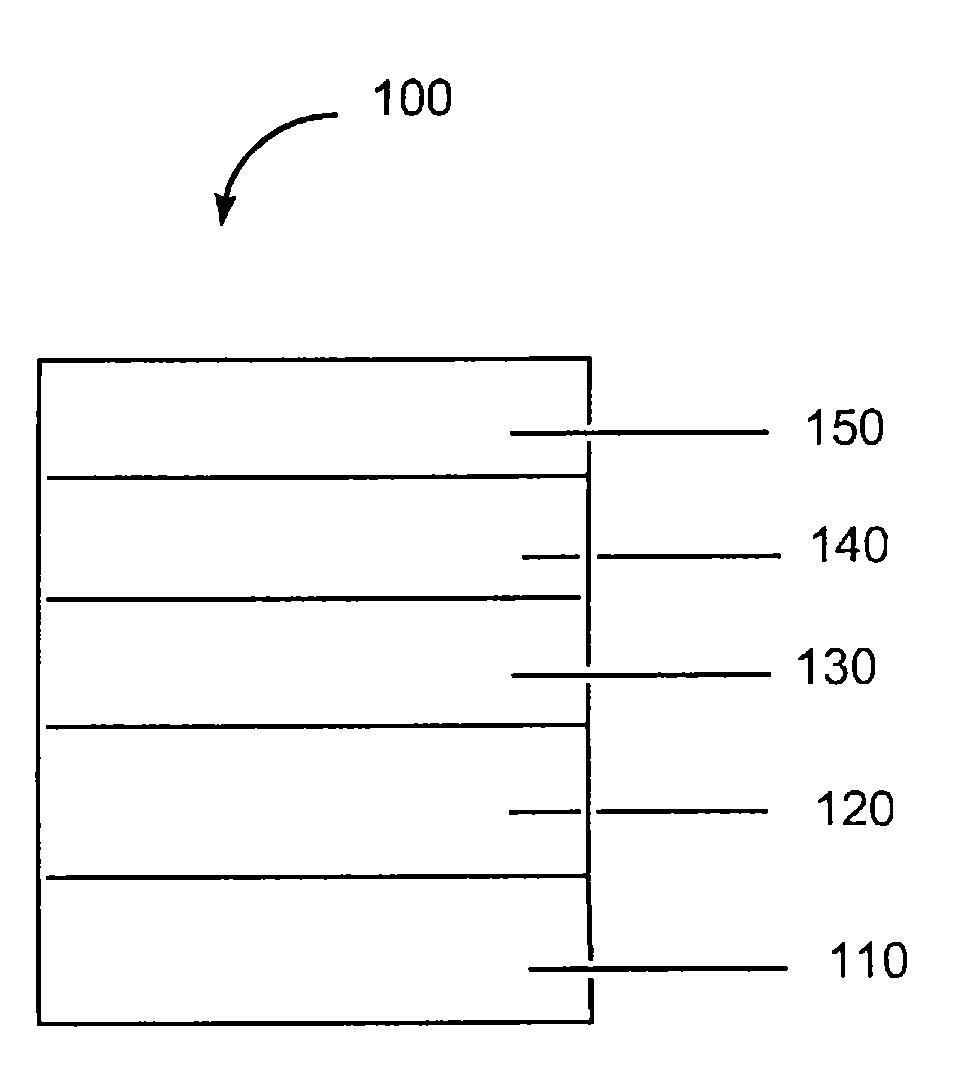
图1为有机电子器件的示意图。Figure 1 is a schematic diagram of an organic electronic device.
技术人员将会知道,附图中的物体以简洁明了的方式示出,并不一定按比例绘制。例如,图中一些物体的尺寸相对于其他物体可能有所放大以便于更好地理解实施方案。Skilled artisans will appreciate that objects in the drawings are shown for simplicity and clarity and have not necessarily been drawn to scale. For example, the dimensions of some of the objects in the figures may be exaggerated relative to other objects to provide a better understanding of the embodiments.
发明详述Detailed description of the invention
本文描述了许多方面和实施方案,它们仅为示例性的而非限制性的。在阅读本说明书后,技术人员将会知道,在不脱离本发明范围的前提下其他方面和实施方案也是可能的。There are many aspects and embodiments described herein which are illustrative only and not limiting. After reading this specification, skilled artisans will appreciate that other aspects and embodiments are possible without departing from the scope of the invention.
根据以下发明详述和权利要求,任何一个或多个实施方案的其他特征和有益效果将显而易见。发明详述首先提出了术语的定义和说明,然后介绍导电聚合物、高度氟化酸聚合物、无机纳米颗粒、掺杂型导电聚合物组合物的制备、缓冲层、电子器件,最后为实施例。Other features and benefits of any one or more embodiments will be apparent from the following detailed description and claims. The Detailed Description of the Invention first presents definitions and clarifications of terms, then presents conductive polymers, highly fluorinated acid polymers, inorganic nanoparticles, preparation of doped conductive polymer compositions, buffer layers, electronic devices, and finally examples .
1.说明书和权利要求中所用术语的定义和说明1. Definitions and clarifications of terms used in the specification and claims
在介绍下述实施方案详情之前,先定义或阐明一些术语。Before introducing the details of the embodiments described below, some terms are defined or clarified.
术语“导体”及其变体旨在指具有电性质的层材料、构件或结构,此电性质使得电流在无电势骤降的情况下能够流经此类层材料、构件或结构。该术语旨在包括半导体。在一些实施方案中,导体将形成具有至少10-7S/cm导电率的层。The term "conductor" and variations thereof are intended to refer to layer materials, components or structures having electrical properties that enable electrical current to flow through such layer materials, components or structures without a dip in potential. The term is intended to include semiconductors. In some embodiments, the conductor will form a layer having a conductivity of at least 10 −7 S/cm.
术语“导电”在涉及材料时,旨在表示在不添加炭黑或导电金属颗粒的情况下本身地或固有地能够导电的材料。The term "conductive" when referring to a material is intended to mean a material which is inherently or inherently capable of conducting electricity without the addition of carbon black or conductive metal particles.
术语“聚合物”旨在表示具有至少一种重复单体单元的材料。该术语包括只有一种或一类单体单元的均聚物以及具有两种或更多种不同单体单元的共聚物,包括不同物质的单体单元形成的共聚物。The term "polymer" is intended to mean a material having at least one repeating monomer unit. The term includes homopolymers of only one or one type of monomeric unit as well as copolymers of two or more different monomeric units, including copolymers of monomeric units of different species.
术语“酸聚合物”是指具有酸性基团的聚合物。The term "acid polymer" refers to a polymer having acidic groups.
术语“酸性基团”是指能够离子化以向布朗斯台德碱提供氢离子的基团。The term "acidic group" refers to a group capable of ionizing to donate a hydrogen ion to a Bronsted base.
术语“高度氟化的”是指其中至少90%的与碳键合的可用氢已被氟置换的化合物。The term "highly fluorinated" refers to compounds in which at least 90% of the available carbon-bonded hydrogens have been replaced by fluorine.
术语“完全氟化的”和“全氟化的”可交替使用,并指与碳键合的所有可用氢都已被氟置换的化合物。The terms "fully fluorinated" and "perfluorinated" are used interchangeably and refer to a compound in which all available hydrogens bonded to carbon have been replaced with fluorine.
组合物可包含一种或多种不同的导电聚合物以及一种或多种不同的高度氟化酸聚合物。The composition may comprise one or more different conductive polymers and one or more different highly fluorinated acid polymers.
术语“掺杂”在涉及导电聚合物时,旨在表示具有聚合抗衡离子以平衡导电聚合物上的电荷的导电聚合物。The term "doped" when referring to a conductive polymer is intended to mean a conductive polymer having polymeric counterions to balance the charge on the conductive polymer.
术语“掺杂型导电聚合物”旨在表示导电聚合物以及与其相关的聚合抗衡离子。The term "doped conducting polymer" is intended to mean conducting polymers and polymeric counterions associated therewith.
术语“层”与术语“膜”可交替使用,是指覆盖所需区域的涂层。该术语不受尺寸的限制。所述区域可以大如整个器件,也可以小如特定功能区例如实际可视显示器,或者小如单个子像素。层和薄膜可由任何常规的沉积技术形成,包括气相沉积、液相沉积(连续和不连续技术)、以及热转移。The term "layer" is used interchangeably with the term "film" and refers to a coating covering a desired area. The term is not limited by size. The area can be as large as an entire device, or as small as a specific functional area such as the actual visual display, or as small as a single sub-pixel. Layers and films can be formed by any conventional deposition technique, including vapor deposition, liquid deposition (continuous and discontinuous techniques), and thermal transfer.
术语“纳米颗粒”是指具有小于100nm的粒度的材料。在一些实施方案中,粒度小于10nm。在一些实施方案中,粒度小于5nm。The term "nanoparticle" refers to a material having a particle size of less than 100 nm. In some embodiments, the particle size is less than 10 nm. In some embodiments, the particle size is less than 5 nm.
术语“含水的”是指具有很大一部分水的液体,并在一个实施例中含至少约40重量%的水;在一些实施方案中,含至少约60重量%的水。The term "aqueous" refers to a liquid having a substantial portion of water, and in one embodiment at least about 40% by weight water; in some embodiments, at least about 60% by weight water.
术语“空穴传输”在涉及层、材料、构件或结构时,旨在表示此类层、材料、构件或结构有利于正电荷以相对高的效率和较小的电荷损失穿过所述层、材料、构件或结构的厚度进行迁移。The term "hole transport" when referring to a layer, material, member or structure is intended to mean that such layer, material, member or structure facilitates the passage of positive charges through said layer with relatively high efficiency and with little charge loss, The thickness of a material, member or structure is migrated.
术语“电子传输”在涉及层、材料、构件或结构时,表示此类层、材料、构件或结构可促进或有利于负电荷通过所述层、材料、构件或结构迁移到另一层、材料、构件或结构。The term "electron transport" when referring to a layer, material, member or structure means that such layer, material, member or structure facilitates or facilitates the migration of negative charges through said layer, material, member or structure to another layer, material , component or structure.
术语“有机电子器件”旨在表示包括一层或多层半导体层或材料的器件。有机电子器件包括但不限于:(1)将电能转换成辐射的器件(例如发光二极管、发光二极管显示器、二极管激光器、或发光面板);(2)通过电子方法探测信号的器件(例如光电探测器、光电导管、光敏电阻器、光控开关、光电晶体管、光电管、红外线(“IR”)探测器、或生物传感器);(3)将辐射转换成电能的器件(例如光伏器件或太阳能电池);以及(4)包括一个或多个电子元件,电子元件继而包括一个或多个有机半导体层的器件(例如晶体管或二极管)。The term "organic electronic device" is intended to mean a device comprising one or more semiconducting layers or materials. Organic electronic devices include, but are not limited to: (1) devices that convert electrical energy into radiation (such as light-emitting diodes, light-emitting diode displays, diode lasers, or light-emitting panels); (2) devices that detect signals electronically (such as photodetectors , photoconductors, photoresistors, photoswitches, phototransistors, photocells, infrared ("IR") detectors, or biosensors); (3) devices that convert radiation into electrical energy (such as photovoltaic devices or solar cells) and (4) a device (such as a transistor or a diode) comprising one or more electronic components which in turn comprise one or more organic semiconductor layers.
如本文所用,术语“包含”、“包括”、“具有”或它们的任何其他变型均旨在涵盖非排他性的包括。例如,包括要素列表的工艺、方法、制品或设备不必仅限于那些要素,而是可以包括未明确列出的或该工艺、方法、制品或设备所固有的其他要素。此外,除非另外明确说明,“或”是指包含性的“或”,而不是指排他性的“或”。例如,以下任何一种情况均满足条件“A或B”:A是真实的(或存在的)且B是虚假的(或不存在的),A是虚假的(或不存在的)且B是真实的(或存在的),以及A和B都是真实的(或存在的)。As used herein, the terms "comprises," "including," "having," or any other variation thereof, are intended to cover a non-exclusive inclusion. For example, a process, method, article, or apparatus that includes a list of elements is not necessarily limited to only those elements, but may include other elements not expressly listed or inherent to the process, method, article, or apparatus. Furthermore, unless expressly stated otherwise, "or" means an inclusive "or", not an exclusive "or". For example, the condition "A or B" is satisfied by any of the following: A is true (or exists) and B is false (or does not exist), A is false (or does not exist) and B is is real (or exists), and both A and B are real (or exist).
同样,使用“一个”或“一种”来描述本文所描述的要素和组分。这样做仅仅是为了方便,并且对本发明的范围提供一般性的意义。这种描述应被理解为包括一个或至少一个,并且该单数也包括复数,除非很明显地另指他意。Likewise, use of "a" or "an" is used to describe elements and components described herein. This is done merely for convenience and to give a general sense of the scope of the invention. This description should be read to include one or at least one and the singular also includes the plural unless it is obvious that it is meant otherwise.
与元素周期表内的列相对应的族序号使用如在“CRC Handbook ofChemistry and Physics”,81,(2000-2001)中所述的“新命名法”公约。Group numbers corresponding to columns within the Periodic Table use the "New Nomenclature" convention as described in "CRC Handbook of Chemistry and Physics", 81, (2000-2001).
除非另有定义,本文所用的所有技术和科学术语的含义均与本发明所属领域的普通技术人员通常理解的一样。在化学式中,字母Q、R、T、W、X、Y和Z用于指代其中限定的原子或基团。其他所有字母用于指代常规的原子符号。与元素周期表内的列相对应的族序号使用如在“CRC Handbook of Chemistry andPhysics”,81,(2000)中所述的“新命名法”公约。Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this invention belongs. In the chemical formulae, the letters Q, R, T, W, X, Y and Z are used to designate the atoms or groups defined therein. All other letters are used to refer to conventional atomic symbols. Group numbers corresponding to columns within the Periodic Table of the Elements use the "new nomenclature" convention as described in "CRC Handbook of Chemistry and Physics", 81, (2000).
本文未描述的有关特定材料、加工方法和电路的许多细节均是常规的,并且可以在有机发光二极管显示器、光源、光电探测器、光伏和半导体构件领域的教科书和其他来源中找到。Many details about specific materials, processing methods, and circuits not described herein are conventional and can be found in textbooks and other sources in the fields of OLED displays, light sources, photodetectors, photovoltaics, and semiconductor components.
2.导电聚合物2. Conductive polymer
任何导电聚合物均可用于本新型组合物中。在一些实施方案中,导电聚合物将形成具有大于10-7S/cm导电率的薄膜。Any conductive polymer can be used in the novel compositions. In some embodiments, the conductive polymer will form a film with a conductivity greater than 10 −7 S/cm.
适于该新型组合物的导电聚合物由至少一种单体制成,该单体在单独聚合时形成导电均聚物。此类单体在本文中称为“导电前驱单体”。在单独聚合时形成不导电的均聚物的单体称为“非导电前驱单体”。导电聚合物可以为均聚物或共聚物。适用于该新型组合物的导电共聚物可由两种或更多种导电前驱单体制成,或者由一种或多种导电前驱单体和一种或多种非导电前驱单体的组合制成。Conductive polymers suitable for the novel composition are made from at least one monomer which, when polymerized alone, forms a conductive homopolymer. Such monomers are referred to herein as "conductive precursor monomers". Monomers that form nonconductive homopolymers when polymerized alone are referred to as "nonconductive precursor monomers." The conductive polymer can be a homopolymer or a copolymer. Conductive copolymers suitable for use in the novel compositions can be made from two or more conductive precursor monomers, or from a combination of one or more conductive precursor monomers and one or more non-conductive precursor monomers .
在一些实施方案中,导电聚合物由选自噻吩、吡咯、苯胺和多环芳族化合物中的至少一种导电前驱单体制成。术语“多环芳族化合物”是指具有一个以上芳环的化合物。所述环可通过一个或多个键接合,或者它们可稠合到一起。术语“芳环”旨在包括杂芳环。“多环杂芳族”化合物具有至少一个杂芳环。In some embodiments, the conductive polymer is made from at least one conductive precursor monomer selected from thiophene, pyrrole, aniline, and polycyclic aromatic compounds. The term "polycyclic aromatic compound" refers to a compound having more than one aromatic ring. The rings may be joined by one or more bonds, or they may be fused together. The term "aromatic ring" is intended to include heteroaromatic rings. A "polycyclic heteroaromatic" compound has at least one heteroaromatic ring.
在一些实施方案中,导电聚合物由至少一种选自噻吩、硒吩、碲吩、吡咯、苯胺、以及多环芳族化合物的前驱单体制成。由这些单体制成的聚合物在本文中分别称为聚噻吩、聚(硒吩)、聚(碲吩)、聚吡咯、聚苯胺、以及多环芳族聚合物。术语“多环芳族化合物”是指具有一个以上芳环的化合物。所述环可通过一个或多个键接合,或者它们可稠合到一起。术语“芳环”旨在包括杂芳环。“多环杂芳族”化合物具有至少一个杂芳环。在一些实施方案中,所述多环芳族聚合物为聚噻吩并噻吩。In some embodiments, the conductive polymer is made from at least one precursor monomer selected from the group consisting of thiophene, selenophene, tellurophene, pyrrole, aniline, and polycyclic aromatic compounds. Polymers made from these monomers are referred to herein as polythiophenes, poly(selenophenes), poly(tellophenes), polypyrroles, polyanilines, and polycyclic aromatic polymers, respectively. The term "polycyclic aromatic compound" refers to a compound having more than one aromatic ring. The rings may be joined by one or more bonds, or they may be fused together. The term "aromatic ring" is intended to include heteroaromatic rings. A "polycyclic heteroaromatic" compound has at least one heteroaromatic ring. In some embodiments, the polycyclic aromatic polymer is polythienothiophene.
在一些实施方案中,设想用于形成新型组合物中的导电聚合物的单体包括下式I:In some embodiments, monomers contemplated for use in forming the conductive polymers in the novel compositions include Formula I below:
其中:in:
Q选自S、Se和Te;Q is selected from S, Se and Te;
R1独立地选择使得在各出现处相同或不同,并选自氢、烷基、烯基、烷氧基、烷酰基、烷硫基、芳氧基、烷硫基烷基、烷基芳基、芳烷基、氨基、烷基氨基、二烷基氨基、芳基、烷基亚磺酰基、烷氧基烷基、烷基磺酰基、芳硫基、芳基亚磺酰基、烷氧羰基、芳基磺酰基、丙烯酸、磷酸、膦酸、卤素、硝基、氰基、羟基、环氧化物、硅烷、硅氧烷、醇、苄基、羧酸盐(酯)、醚、醚羧酸盐(酯)、酰胺基磺酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷;或者两个R1基团一起可形成亚烷基或亚烯基链,从而得到3、4、5、6、或7元芳环或脂环,该环可任选地包含一个或多个二价氮、硒、碲、硫或氧原子。R are independently selected such that they are the same or different at each occurrence and are selected from the group consisting of hydrogen, alkyl, alkenyl, alkoxy, alkanoyl, alkylthio, aryloxy, alkylthioalkyl, alkylaryl , aralkyl, amino, alkylamino, dialkylamino, aryl, alkylsulfinyl, alkoxyalkyl, alkylsulfonyl, arylthio, arylsulfinyl, alkoxycarbonyl, Arylsulfonyl, acrylic acid, phosphoric acid, phosphonic acid, halogen, nitro, cyano, hydroxyl, epoxy, silane, siloxane, alcohol, benzyl, carboxylate (ester), ether, ether carboxylate (ester), amidosulfonate (ester), ethersulfonate (ester), estersulfonate (ester), and urethane; or two R groups together may form an alkylene or alkenylene chain , thereby obtaining a 3, 4, 5, 6, or 7-membered aromatic or aliphatic ring, which may optionally contain one or more divalent nitrogen, selenium, tellurium, sulfur, or oxygen atoms.
如本文所用,术语“烷基”是指衍生自脂族烃的基团,其包括未取代的或取代的直链、支链和环状基团。术语“杂烷基”旨在表示其中烷基的一个或多个碳原子被另外的原子(例如氮、氧、硫等)取代的烷基。术语“亚烷基”是指具有两个连接点的烷基。As used herein, the term "alkyl" refers to a group derived from an aliphatic hydrocarbon, which includes unsubstituted or substituted straight chain, branched chain and cyclic groups. The term "heteroalkyl" is intended to mean an alkyl group in which one or more carbon atoms of the alkyl group are replaced by another atom (eg, nitrogen, oxygen, sulfur, etc.). The term "alkylene" refers to an alkyl group having two points of attachment.
如本文所用,术语“烯基”是指衍生自具有至少一个碳碳双键的脂族烃的基团,其包括可为未取代的或取代的直链、支链和环状基团。术语“杂烯基”旨在表示其中烯基的一个或多个碳原子被另外的原子(例如氮、氧、硫等)取代的烯基。术语“亚烯基”是指具有两个连接点的烯基。As used herein, the term "alkenyl" refers to a group derived from an aliphatic hydrocarbon having at least one carbon-carbon double bond, and includes straight chain, branched chain and cyclic groups which may be unsubstituted or substituted. The term "heteroalkenyl" is intended to mean an alkenyl group in which one or more carbon atoms of the alkenyl group are replaced by another atom (eg, nitrogen, oxygen, sulfur, etc.). The term "alkenylene" refers to an alkenyl group having two points of attachment.
如本文所用,用于取代基的下列术语是指下面给出的化学式:As used herein, the following terms for substituents refer to the chemical formula given below:
“醇” -R3-OH"Alcohol" -R 3 -OH
“酰胺基” -R3-C(O)N(R6)R6 "Amido" -R 3 -C(O)N(R 6 )R 6
“酰胺基磺酸盐(酯)” -R3-C(O)N(R6)R4-SO3Z"Amidosulfonate" -R 3 -C(O)N(R 6 )R 4 -SO 3 Z
“苄基” -CH2-C6H5 "Benzyl" -CH 2 -C 6 H 5
“羧酸盐(酯)” -R3-C(O)O-Z或-R3-O-C(O)-Z"Carboxylate (ester)" -R 3 -C(O)OZ or -R 3 -OC(O)-Z
“醚” -R3-(O-R5)p-O-R5 "Ether" -R 3 -(OR 5 ) p -OR 5
“醚羧酸盐(酯)” -R3-O-R4-C(O)O-Z或-R3-O-R4-O-C(O)-Z"Ether carboxylate (ester)" -R 3 -OR 4 -C(O)OZ or -R 3 -OR 4 -OC(O)-Z
“醚磺酸盐(酯)” -R3-O-R4-SO3Z"Ether sulfonate (ester)" -R 3 -OR 4 -SO 3 Z
“酯磺酸盐(酯)” -R3-O-C(O)-R4-SO3Z"Ester sulfonate (ester)" -R 3 -OC(O)-R 4 -SO 3 Z
“磺酰亚胺” -R3-SO2-NH-SO2-R5 "Sulfonimide" -R 3 -SO 2 -NH-SO 2 -R 5
“尿烷” -R3-O-C(O)-N(R6)2 "Urethane" -R 3 -OC(O)-N(R 6 ) 2
其中所有“R”基团在各出现处相同或不同,并且:wherein all "R" groups are the same or different at each occurrence, and:
R3为单键或亚烷基R 3 is a single bond or an alkylene group
R4为亚烷基R 4 is alkylene
R5为烷基R 5 is alkyl
R6为氢或烷基 R6 is hydrogen or alkyl
p为0或1至20的整数p is 0 or an integer from 1 to 20
Z为H、碱金属、碱土金属、N(R5)4或R5 Z is H, alkali metal, alkaline earth metal, N(R 5 ) 4 or R 5
上述基团中的任何一个还可以为未取代的或取代的,并且任何基团均可具有取代一个或多个氢的F,包括全氟化基团。在一些实施方案中,烷基和亚烷基具有1至20个碳原子。Any of the above groups may also be unsubstituted or substituted, and any group may have F in place of one or more hydrogens, including perfluorinated groups. In some embodiments, the alkyl and alkylene groups have 1 to 20 carbon atoms.
在一些实施方案中,单体中两个R1一起形成-W-(CY1Y2)m-W-,其中m为2或3,W为O、S、Se、PO、NR6,Y1在各出现处相同或不同并为氢或氟,Y2在各出现处相同或不同并选自氢、卤素、烷基、醇、酰胺基磺酸盐(酯)、苄基、羧酸盐(酯)、醚、醚羧酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷,其中Y基团可为部分氟化或完全氟化的。在一些实施方案中,所有Y均为氢。在一些实施方案中,聚合物为聚(3,4-乙撑二氧噻吩)。在一些实施方案中,至少一个Y基团不为氢。在一些实施方案中,至少一个Y基团为具有F的取代基,其中所述F取代至少一个氢。在一些实施方案中,至少一个Y基团为全氟化的。In some embodiments, two R 1 in the monomer together form -W-(CY 1 Y 2 ) m -W-, wherein m is 2 or 3, W is O, S, Se, PO, NR 6 , Y 1 is the same or different at each occurrence and is hydrogen or fluorine, Y 2 is the same or different at each occurrence and is selected from hydrogen, halogen, alkyl, alcohol, amidosulfonate (ester), benzyl, carboxylate (ester), ether, ether carboxylate (ester), ether sulfonate (ester), ester sulfonate (ester), and urethane, where the Y group can be partially or fully fluorinated. In some embodiments, all Y are hydrogen. In some embodiments, the polymer is poly(3,4-ethylenedioxythiophene). In some embodiments, at least one Y group is not hydrogen. In some embodiments, at least one Y group is a substituent having F, wherein the F replaces at least one hydrogen. In some embodiments, at least one Y group is perfluorinated.
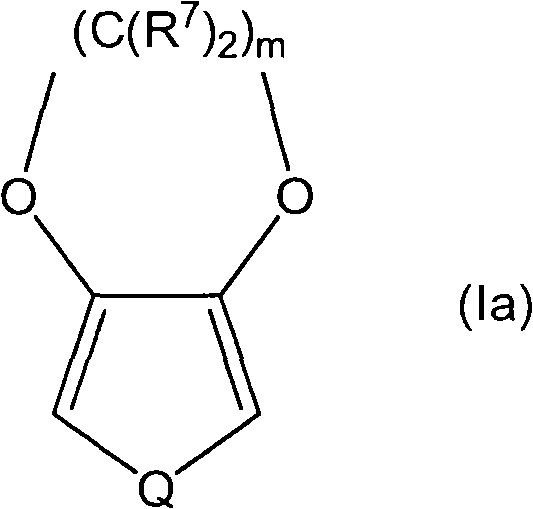
在一些实施方案中,单体具有式I(a):In some embodiments, the monomer has Formula I(a):
其中:in:
Q选自S、Se和Te;Q is selected from S, Se and Te;
R7在各出现处相同或不同,并选自氢、烷基、杂烷基、烯基、杂烯基、醇、酰胺基磺酸盐(酯)、苄基、羧酸盐(酯)、醚、醚羧酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷,前提条件是至少一个R7不为氢,并且 R is the same or different at each occurrence, and is selected from hydrogen, alkyl, heteroalkyl, alkenyl, heteroalkenyl, alcohol, amidosulfonate, benzyl, carboxylate, Ether, ether carboxylate (ester), ether sulfonate (ester), ester sulfonate (ester) and urethane, with the proviso that at least one R is not hydrogen, and
m为2或3。m is 2 or 3.
在式I(a)的一些实施方案中,m为2,其中一个R7为多于5个碳原子的烷基,而所有其他R7均为氢。In some embodiments of Formula I(a), m is 2, one R 7 is alkyl of more than 5 carbon atoms, and all other R 7 are hydrogen.
在式I(a)的一些实施方案中,至少一个R7基团为氟化的。在一些实施方案中,至少一个R7基团具有至少一个氟取代基。在一些实施方案中,R7基团为完全氟化的。In some embodiments of Formula I(a), at least one R group is fluorinated. In some embodiments, at least one R group has at least one fluorine substituent. In some embodiments, the R group is fully fluorinated.
在式I(a)的一些实施方案中,单体稠合脂环上的R7取代基可提供改善的单体水溶解性,并有利于在氟化酸聚合物的存在下发生聚合。In some embodiments of formula I(a), the R7 substituent on the fused alicyclic ring of the monomer can provide improved aqueous solubility of the monomer and facilitate polymerization in the presence of the fluorinated acid polymer.
在式I(a)的一些实施方案中,m为2,其中一个R7为磺酸-亚丙基-醚-亚甲基,而所有其他R7均为氢。在一些实施方案中,m为2,其中一个R7为丙基-醚-乙烯,而所有其他R7均为氢。在一些实施方案中,m为2,其中一个R7为甲氧基,而所有其他R7均为氢。在一些实施方案中,其中一个R7为磺酸二氟亚甲基酯亚甲基(-CH2-O-C(O)-CF2-SO3H),而所有其他R7均为氢。In some embodiments of Formula I(a), m is 2, one R 7 is sulfonic acid-propylene-ether-methylene, and all other R 7 are hydrogen. In some embodiments, m is 2, one R 7 is propyl-ether-ethylene, and all other R 7 are hydrogen. In some embodiments, m is 2, one R 7 is methoxy, and all other R 7 are hydrogen. In some embodiments, one R 7 is difluoromethylene sulfonate methylene (—CH 2 —OC(O)—CF 2 —SO 3 H), and all other R 7 are hydrogen.
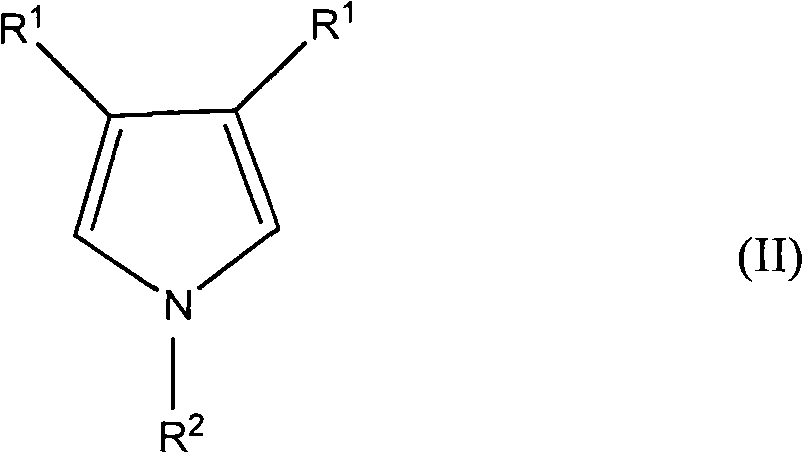
在一些实施方案中,设想用于形成新型组合物中的导电聚合物的吡咯单体包含下式II。In some embodiments, the pyrrole monomers contemplated for use in forming the conductive polymers in the novel compositions comprise Formula II below.
其中在式II中:Wherein in formula II:
独立地选择R1使得其在各出现处相同或不同,其选自氢、烷基、烯基、烷氧基、烷酰基、烷硫基、芳氧基、烷硫基烷基、烷基芳基、芳烷基、氨基、烷基氨基、二烷基氨基、芳基、烷基亚磺酰基、烷氧基烷基、烷基磺酰基、芳硫基、芳基亚磺酰基、烷氧羰基、芳基磺酰基、丙烯酸、磷酸、膦酸、卤素、硝基、氰基、羟基、环氧化物、硅烷、硅氧烷、醇、苄基、羧酸盐(酯)、醚、酰胺基磺酸盐(酯)、醚羧酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷;或者两个R1基团一起可形成亚烷基或亚烯基链,从而得到3、4、5、6、或7元芳环或脂环,该环可任选地包含一个或多个二价氮、硫、硒、碲、或氧原子;以及 R is independently selected such that it is the same or different at each occurrence and is selected from the group consisting of hydrogen, alkyl, alkenyl, alkoxy, alkanoyl, alkylthio, aryloxy, alkylthioalkyl, alkylaryl radical, aralkyl, amino, alkylamino, dialkylamino, aryl, alkylsulfinyl, alkoxyalkyl, alkylsulfonyl, arylthio, arylsulfinyl, alkoxycarbonyl , arylsulfonyl, acrylic acid, phosphoric acid, phosphonic acid, halogen, nitro, cyano, hydroxyl, epoxy, silane, siloxane, alcohol, benzyl, carboxylate (ester), ether, amidosulfonate salt (ester), ether carboxylate (ester), ether sulfonate (ester), ester sulfonate (ester), and urethane; or two R groups together can form an alkylene or alkenylene chain, thereby obtaining 3, 4, 5, 6, or 7 membered aromatic rings or alicyclic rings, which rings may optionally contain one or more divalent nitrogen, sulfur, selenium, tellurium, or oxygen atoms; and
独立地选择R2使得其在各出现处相同或不同,其选自氢、烷基、烯基、芳基、烷酰基、烷硫基烷基、烷基芳基、芳基烷基、氨基、环氧化物、硅烷、硅氧烷、醇、苄基、羧酸盐(酯)、醚、醚羧酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷。 R is independently selected such that it is the same or different at each occurrence and is selected from the group consisting of hydrogen, alkyl, alkenyl, aryl, alkanoyl, alkylthioalkyl, alkylaryl, arylalkyl, amino, Epoxides, silanes, siloxanes, alcohols, benzyls, carboxylates, ethers, ether carboxylates, ether sulfonates, ester sulfonates and urethanes .
在一些实施方案中,R1在各出现处相同或不同,并独立地选自氢、烷基、烯基、烷氧基、环烷基、环烯基、醇、苄基、羧酸盐(酯)、醚、酰胺基磺酸盐(酯)、醚羧酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)、尿烷、环氧化物、硅烷、硅氧烷,以及被磺酸、羧酸、丙烯酸、磷酸、膦酸、卤素、硝基、氰基、羟基、环氧化物、硅烷、或硅氧烷部分中的一个或多个取代的烷基。In some embodiments, R is the same or different at each occurrence and is independently selected from hydrogen, alkyl, alkenyl, alkoxy, cycloalkyl, cycloalkenyl, alcohol, benzyl, carboxylate ( ester), ether, amido sulfonate (ester), ether carboxylate (ester), ether sulfonate (ester), ester sulfonate (ester), urethane, epoxy, silane, siloxane , and alkyl substituted with one or more of sulfonic acid, carboxylic acid, acrylic acid, phosphoric acid, phosphonic acid, halogen, nitro, cyano, hydroxyl, epoxy, silane, or siloxane moieties.
在一些实施方案中,R2选自氢、烷基,以及被磺酸、羧酸、丙烯酸、磷酸、膦酸、卤素、氰基、羟基、环氧化物、硅烷、或硅氧烷部分中的一个或多个取代的烷基。In some embodiments, R is selected from hydrogen, alkyl, and sulfonic acid, carboxylic acid, acrylic acid, phosphoric acid, phosphonic acid, halogen, cyano, hydroxyl, epoxy, silane, or siloxane moieties. One or more substituted alkyl groups.
在一些实施方案中,吡咯单体为未取代的并且R1和R2均为氢。In some embodiments, the pyrrole monomer is unsubstituted and R and R are both hydrogen.
在一些实施方案中,两个R1一起形成6或7元脂环,其进一步被选自烷基、杂烷基、醇、苄基、羧酸盐(酯)、醚、醚羧酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷的基团取代。这些基团可改善单体和所得聚合物的溶解性。在一些实施方案中,两个R1一起形成6或7元脂环,其进一步被烷基取代。在一些实施方案中,两个R1一起形成6或7元脂环,其进一步被具有至少1个碳原子的烷基取代。In some embodiments, two R together form a 6- or 7-membered alicyclic ring further selected from the group consisting of alkyl, heteroalkyl, alcohol, benzyl, carboxylate, ether, ether carboxylate ( Ester), ether sulfonate (ester), ester sulfonate (ester) and urethane group substitution. These groups improve the solubility of the monomer and the resulting polymer. In some embodiments, two R 1 are taken together to form a 6- or 7-membered alicyclic ring, which is further substituted with an alkyl group. In some embodiments, two R 1 together form a 6- or 7-membered alicyclic ring, which is further substituted with an alkyl group having at least 1 carbon atom.
在一些实施方案中,两个R1一起形成-O-(CHY)m-O-,其中m为2或3,Y在各出现处相同或不同并选自氢、烷基、醇、苄基、羧酸盐(酯)、酰胺基磺酸盐(酯)、醚、醚羧酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷。在一些实施方案中,至少一个Y基团不为氢。在一些实施方案中,至少一个Y基团为具有F的取代基,其中所述F取代至少一个氢。在一些实施方案中,至少一个Y基团为全氟化的。In some embodiments, two R 's are taken together to form -O-(CHY) m -O-, where m is 2 or 3 and Y is the same or different at each occurrence and is selected from hydrogen, alkyl, alcohol, benzyl , carboxylate (ester), amidosulfonate (ester), ether, ether carboxylate (ester), ether sulfonate (ester), ester sulfonate (ester) and urethane. In some embodiments, at least one Y group is not hydrogen. In some embodiments, at least one Y group is a substituent having F, wherein the F replaces at least one hydrogen. In some embodiments, at least one Y group is perfluorinated.
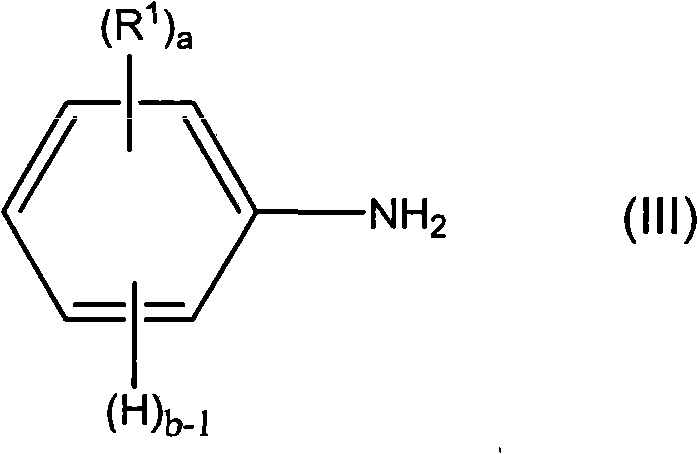
在一些实施方案中,设想用于形成新型组合物中的导电聚合物的苯胺单体包括下式III。In some embodiments, aniline monomers contemplated for use in forming the conductive polymers in the novel compositions include Formula III below.
其中:in:
a为0或1至4的整数;a is 0 or an integer from 1 to 4;
b为1至5的整数,前提条件是a+b=5;以及b is an integer from 1 to 5, provided that a+b=5; and
R1独立地选择使得在各出现处相同或不同,其选自氢、烷基、烯基、烷氧基、烷酰基、烷硫基、芳氧基、烷硫基烷基、烷基芳基、芳烷基、氨基、烷基氨基、二烷基氨基、芳基、烷基亚磺酰基、烷氧基烷基、烷基磺酰基、芳硫基、芳基亚磺酰基、烷氧羰基、芳基磺酰基、丙烯酸、磷酸、膦酸、卤素、硝基、氰基、羟基、环氧化物、硅烷、硅氧烷、醇、苄基、羧酸盐(酯)、醚、醚羧酸盐(酯)、酰胺基磺酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷;或者两个R1基团一起可形成亚烷基或亚烯基链,从而得到3、4、5、6、或7元芳环或脂环,该环可任选地包含一个或多个二价氮、硫或氧原子。 R independently selected such that the same or different at each occurrence is selected from hydrogen, alkyl, alkenyl, alkoxy, alkanoyl, alkylthio, aryloxy, alkylthioalkyl, alkylaryl , aralkyl, amino, alkylamino, dialkylamino, aryl, alkylsulfinyl, alkoxyalkyl, alkylsulfonyl, arylthio, arylsulfinyl, alkoxycarbonyl, Arylsulfonyl, acrylic acid, phosphoric acid, phosphonic acid, halogen, nitro, cyano, hydroxyl, epoxy, silane, siloxane, alcohol, benzyl, carboxylate (ester), ether, ether carboxylate (ester), amidosulfonate (ester), ethersulfonate (ester), estersulfonate (ester), and urethane; or two R groups together may form an alkylene or alkenylene chain , thereby obtaining a 3, 4, 5, 6, or 7 membered aromatic or aliphatic ring, which may optionally contain one or more divalent nitrogen, sulfur, or oxygen atoms.
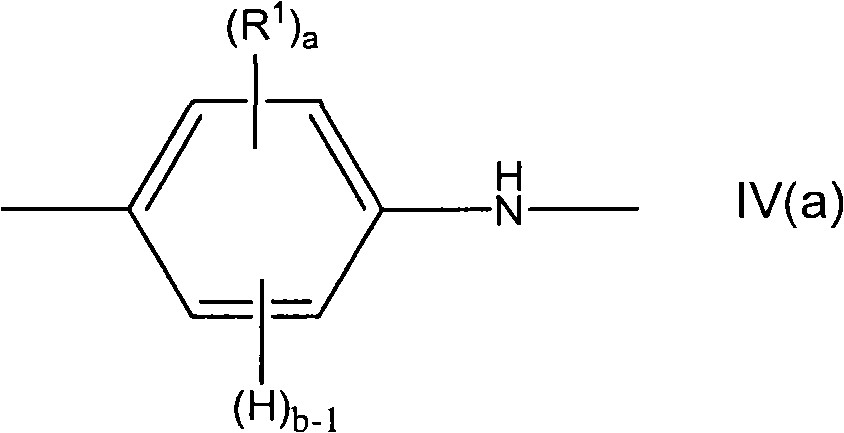
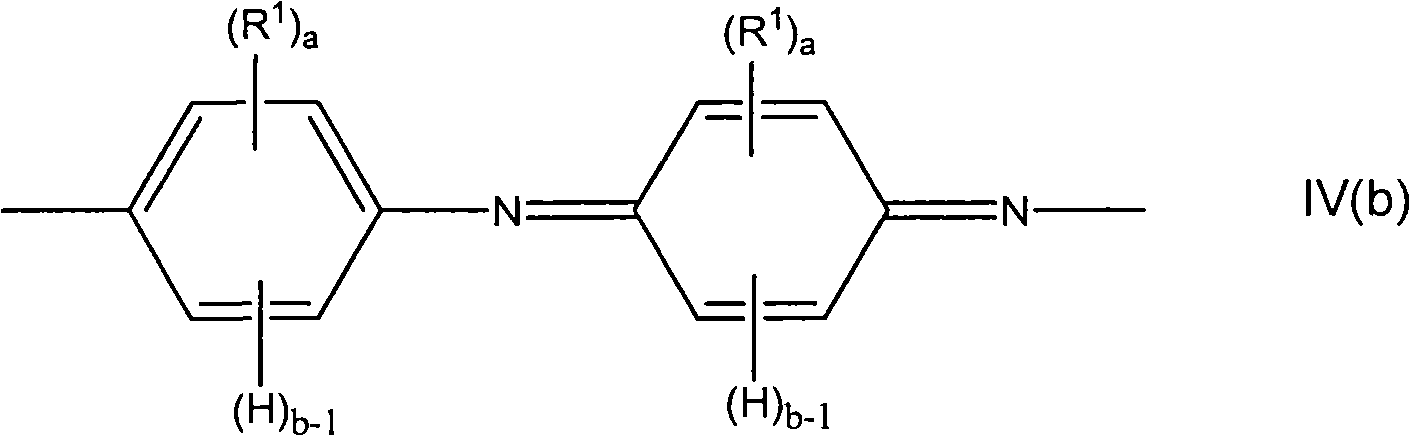
在聚合时,苯胺单体单元可具有如下所示的式IV(a)或式IV(b),或两式的组合。When polymerized, the aniline monomeric unit may have formula IV(a) or formula IV(b), as shown below, or a combination of the two formulas.
其中a、b和R1如上所定义。wherein a, b and R1 are as defined above.
在一些实施方案中,苯胺单体为未取代的并且a=0。In some embodiments, the aniline monomer is unsubstituted and a=0.
在一些实施方案中,a不为0并且至少一个R1为氟化的。在一些实施方案中,至少一个R1为全氟化的。In some embodiments, a is not 0 and at least one R 1 is fluorinated. In some embodiments, at least one R 1 is perfluorinated.
在一些实施方案中,设想用于形成新型组合物中的导电聚合物的稠合多环杂芳族单体具有两个或更多个稠合芳环,其中至少一个为杂芳环。在一些实施方案中,稠合多环杂芳族单体具有式V:In some embodiments, the fused polycyclic heteroaromatic monomers contemplated for use in forming the conductive polymers in the novel compositions have two or more fused aromatic rings, at least one of which is a heteroaromatic ring. In some embodiments, the fused polycyclic heteroaromatic monomer has the formula V:
其中:in:
Q为S、Se、Te、或NR6;Q is S, Se, Te, or NR 6 ;
R6为氢或烷基;R 6 is hydrogen or alkyl;
独立地选择R8、R9、R10和R11使得其在各出现处相同或不同,它们选自氢、烷基、烯基、烷氧基、烷酰基、烷硫基、芳氧基、烷硫基烷基、烷基芳基、芳烷基、氨基、烷基氨基、二烷基氨基、芳基、烷基亚磺酰基、烷氧基烷基、烷基磺酰基、芳硫基、芳基亚磺酰基、烷氧羰基、芳基磺酰基、丙烯酸、磷酸、膦酸、卤素、硝基、腈、氰基、羟基、环氧化物、硅烷、硅氧烷、醇、苄基、羧酸盐(酯)、醚、醚羧酸盐(酯)、酰胺基磺酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷;以及R 8 , R 9 , R 10 and R 11 are independently selected such that they are the same or different at each occurrence and are selected from the group consisting of hydrogen, alkyl, alkenyl, alkoxy, alkanoyl, alkylthio, aryloxy, Alkylthioalkyl, alkylaryl, aralkyl, amino, alkylamino, dialkylamino, aryl, alkylsulfinyl, alkoxyalkyl, alkylsulfonyl, arylthio, Arylsulfinyl, alkoxycarbonyl, arylsulfonyl, acrylic acid, phosphoric acid, phosphonic acid, halogen, nitro, nitrile, cyano, hydroxyl, epoxy, silane, siloxane, alcohol, benzyl, carboxyl acid salts, ethers, ether carboxylates, amido sulfonates, ether sulfonates, ester sulfonates and urethanes; and
R8与R9、R9与R10、以及R10与R11中的至少一对一起形成亚烯基链,从而得到5或6元芳环,该环可任选地包含一个或多个二价氮、硫、硒、碲、或氧原子。At least one pair of R 8 and R 9 , R 9 and R 10 , and R 10 and R 11 together form an alkenylene chain, resulting in a 5- or 6-membered aromatic ring, which may optionally contain one or more Divalent nitrogen, sulfur, selenium, tellurium, or oxygen atoms.
在一些实施方案中,稠合多环杂芳族单体具有的化学式选自V(a)、V(b)、V(c)、V(d)、V(e)、V(f)、V(g)、V(h)、V(i)、V(j)和V(k):In some embodiments, the fused polycyclic heteroaromatic monomer has a formula selected from the group consisting of V(a), V(b), V(c), V(d), V(e), V(f), V(g), V(h), V(i), V(j) and V(k):
其中:in:
Q为S、Se、Te、或NH;以及Q is S, Se, Te, or NH; and
T在各出现处相同或不同,并选自S、NR6、O、SiR6 2、Se、Te和PR6;T is the same or different at each occurrence and is selected from S, NR 6 , O, SiR 6 2 , Se, Te and PR 6 ;
Y为N;以及Y is N; and
R6为氢或烷基。R 6 is hydrogen or alkyl.
稠合多环杂芳族单体可进一步被选自烷基、杂烷基、醇、苄基、羧酸盐(酯)、醚、醚羧酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷的基团取代。在一些实施方案中,取代基为氟化的。在一些实施方案中,取代基为完全氟化的。The fused polycyclic heteroaromatic monomer can be further selected from alkyl, heteroalkyl, alcohol, benzyl, carboxylate, ether, ether carboxylate, ether sulfonate , ester sulfonate (ester) and urethane group substitution. In some embodiments, the substituents are fluorinated. In some embodiments, the substituents are fully fluorinated.
在一些实施方案中,稠合多环杂芳族单体为噻吩并噻吩。此类化合物已在例如Macromolecules,34,5746-5747(2001)和Macromolecules,35,7281-7286(2002)中有所讨论。在一些实施方案中,噻吩并噻吩选自噻吩并(2,3-b)噻吩、噻吩并(3,2-b)噻吩和噻吩并(3,4-b)噻吩。在一些实施方案中,噻吩并噻吩单体进一步被选自烷基、杂烷基、醇、苄基、羧酸盐(酯)、醚、醚羧酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷的至少一个基团取代。在一些实施方案中,取代基为氟化的。在一些实施方案中,取代基为完全氟化的。In some embodiments, the fused polycyclic heteroaromatic monomer is thienothiophene. Such compounds have been discussed eg in Macromolecules, 34, 5746-5747 (2001) and Macromolecules, 35, 7281-7286 (2002). In some embodiments, the thienothiophene is selected from thieno(2,3-b)thiophene, thieno(3,2-b)thiophene, and thieno(3,4-b)thiophene. In some embodiments, the thienothiophene monomer is further selected from the group consisting of alkyl, heteroalkyl, alcohol, benzyl, carboxylate, ether, ether carboxylate, ether sulfonate ), ester sulfonate (ester) and at least one group substitution of urethane. In some embodiments, the substituents are fluorinated. In some embodiments, the substituents are fully fluorinated.
在一些实施方案中,设想用于形成新型组合物中的聚合物的多环杂芳族单体包括式VI:In some embodiments, polycyclic heteroaromatic monomers contemplated for use in forming polymers in the novel compositions include Formula VI:
其中:in:
Q为S、Se、Te、或NR6;Q is S, Se, Te, or NR 6 ;
T选自S、NR6、O、SiR6 2、Se、Te和PR6;T is selected from S, NR 6 , O, SiR 6 2 , Se, Te and PR 6 ;
E选自亚烯基、亚芳基和杂亚芳基;E is selected from alkenylene, arylene and heteroarylene;
R6为氢或烷基;R 6 is hydrogen or alkyl;
R12在各出现处相同或不同,其选自氢、烷基、烯基、烷氧基、烷酰基、烷硫基、芳氧基、烷硫基烷基、烷基芳基、芳烷基、氨基、烷基氨基、二烷基氨基、芳基、烷基亚磺酰基、烷氧基烷基、烷基磺酰基、芳硫基、芳基亚磺酰基、烷氧羰基、芳基磺酰基、丙烯酸、磷酸、膦酸、卤素、硝基、腈、氰基、羟基、环氧化物、硅烷、硅氧烷、醇、苄基、羧酸盐(酯)、醚、醚羧酸盐(酯)、酰胺基磺酸盐(酯)、醚磺酸盐(酯)、酯磺酸盐(酯)和尿烷;或者两个R12基团一起可形成亚烷基或亚烯基链,从而得到3、4、5、6、或7元芳环或脂环,该环可任选地包含一个或多个二价氮、硫、硒、碲、或氧原子。R 12 is the same or different at each occurrence and is selected from hydrogen, alkyl, alkenyl, alkoxy, alkanoyl, alkylthio, aryloxy, alkylthioalkyl, alkylaryl, aralkyl , amino, alkylamino, dialkylamino, aryl, alkylsulfinyl, alkoxyalkyl, alkylsulfonyl, arylthio, arylsulfinyl, alkoxycarbonyl, arylsulfonyl , acrylic acid, phosphoric acid, phosphonic acid, halogen, nitro, nitrile, cyano, hydroxyl, epoxy, silane, siloxane, alcohol, benzyl, carboxylate (ester), ether, ether carboxylate (ester) ), amidosulfonate (ester), ethersulfonate (ester), estersulfonate (ester) and urethane; or two R groups together can form an alkylene or alkenylene chain, thereby A 3, 4, 5, 6, or 7 membered aromatic or aliphatic ring is obtained, which ring may optionally contain one or more divalent nitrogen, sulfur, selenium, tellurium, or oxygen atoms.
在一些实施方案中,导电聚合物为前驱单体和至少一种第二单体的共聚物。只要其不对共聚物的所需性能造成不利影响,可使用任何类型的第二单体。在一些实施方案中,基于单体单元的总数,第二单体在聚合物中不超过50%。在一些实施方案中,基于单体单元的总数,第二单体在聚合物中不超过30%。在一些实施方案中,基于单体单元的总数,第二单体在聚合物中不超过10%。In some embodiments, the conductive polymer is a copolymer of a precursor monomer and at least one second monomer. Any type of second monomer can be used as long as it does not adversely affect the desired properties of the copolymer. In some embodiments, the second monomer makes up no more than 50% of the polymer based on the total number of monomer units. In some embodiments, the second monomer is no more than 30% of the polymer based on the total number of monomer units. In some embodiments, the second monomer is no more than 10% of the polymer based on the total number of monomer units.
第二单体的示例性类型包括但不限于烯基、炔基、亚芳基和杂亚芳基。第二单体的实例包括但不限于芴、噁二唑、噻二唑、苯并噻二唑、苯撑乙烯、苯撑乙炔、吡啶、二嗪和三嗪,它们均可被进一步取代。Exemplary types of second monomers include, but are not limited to, alkenyl, alkynyl, arylene, and heteroarylene. Examples of the second monomer include, but are not limited to, fluorene, oxadiazole, thiadiazole, benzothiadiazole, phenylene vinylene, phenylene acetylene, pyridine, diazine, and triazine, all of which can be further substituted.
在一些实施方案中,通过首先形成具有结构A-B-C的中间前驱单体来制备共聚物,其中A和C表示前驱单体,它们可相同或不同,而B表示第二单体。可使用标准的有机合成技术制备A-B-C中间前驱单体,这些技术例如Yamamoto、Stille、Grignard复分解反应,Suzuki和Negishi偶合反应。然后仅通过该中间前驱单体,或与一种或多种其他前驱单体发生氧化聚合,从而形成共聚物。In some embodiments, the copolymer is prepared by first forming an intermediate precursor monomer having the structure A-B-C, where A and C represent precursor monomers, which may be the same or different, and B represents a second monomer. A-B-C intermediate precursor monomers can be prepared using standard organic synthesis techniques such as Yamamoto, Stille, Grignard metathesis, Suzuki and Negishi coupling reactions. A copolymer is then formed by oxidative polymerization of this intermediate precursor monomer alone, or with one or more other precursor monomers.
在一些实施方案中,导电聚合物选自聚噻吩、聚吡咯、稠合多环杂芳族聚合物、它们的共聚物、以及它们的组合。In some embodiments, the conductive polymer is selected from polythiophenes, polypyrroles, fused polycyclic heteroaromatic polymers, copolymers thereof, and combinations thereof.
在一些实施方案中,导电聚合物选自聚(3,4-乙撑二氧噻吩)、未取代的聚吡咯、聚(噻吩并(2,3-b)噻吩)、聚(噻吩并(3,2-b)噻吩)、以及聚(噻吩并(3,4-b)噻吩)。In some embodiments, the conductive polymer is selected from poly(3,4-ethylenedioxythiophene), unsubstituted polypyrrole, poly(thieno(2,3-b)thiophene), poly(thieno(3 , 2-b)thiophene), and poly(thieno(3,4-b)thiophene).
3.高度氟化酸聚合物3. Highly Fluorinated Acid Polymers
高度氟化酸聚合物(“HFAP”)可以为高度氟化的并具有带酸性质子的酸性基团的任何聚合物。所述酸性基团提供可电离的质子。在一些实施方案中,所述酸性质子具有小于3的pKa值。在一些实施方案中,所述酸性质子具有小于0的pKa值。在一些实施方案中,所述酸性质子具有小于-5的pKa值。所述酸性基团可以直接连接到聚合物主链上,或者它可以连接到聚合物主链的侧链上。酸性基团的实例包括但不限于羧酸基团、磺酸基团、磺酰亚胺基团、磷酸基团、膦酸基团、以及它们的组合。所述酸性基团可以全部相同,或者聚合物可具有一种以上类型的酸性基团。在一些实施方案中,酸性基团选自磺酸基团、磺酰胺基团、以及它们的组合。A highly fluorinated acid polymer ("HFAP") can be any polymer that is highly fluorinated and has acidic groups bearing acidic protons. The acidic groups donate ionizable protons. In some embodiments, the acidic proton has a pKa value of less than 3. In some embodiments, the acidic proton has a pKa value less than zero. In some embodiments, the acidic proton has a pKa value of less than -5. The acid group can be attached directly to the polymer backbone, or it can be attached to a side chain of the polymer backbone. Examples of acidic groups include, but are not limited to, carboxylic acid groups, sulfonic acid groups, sulfonimide groups, phosphoric acid groups, phosphonic acid groups, and combinations thereof. The acid groups may all be the same, or the polymer may have more than one type of acid group. In some embodiments, the acidic group is selected from sulfonic acid groups, sulfonamide groups, and combinations thereof.
在一些实施方案中,HFAP的氟化程度为至少95%;在一些实施方案中,其为完全氟化的。In some embodiments, the HFAP is at least 95% fluorinated; in some embodiments, it is fully fluorinated.
在一些实施方案中,HFAP是水溶性的。在一些实施方案中,HFAP是在水中可分散的。在一些实施方案中,HFAP是有机溶剂可润湿的。术语“有机溶剂可润湿的”是指材料在形成薄膜时与有机溶剂形成的接触角不大于60℃。在一些实施方案中,可润湿的材料形成的薄膜可被苯基己烷润湿,其接触角不大于55°。测量接触角的方法是熟知的。在一些实施方案中,可润湿的材料可由聚合酸制成,该聚合酸本身是不可润湿的,但是使用特选的添加剂可使其变成可润湿的。In some embodiments, the HFAP is water soluble. In some embodiments, the HFAP is dispersible in water. In some embodiments, the HFAP is organic solvent wettable. The term "organic solvent wettable" means that the contact angle formed by the material with an organic solvent when forming a thin film is not greater than 60°C. In some embodiments, the wettable material forms a film that is wettable by phenylhexane with a contact angle of no greater than 55°. Methods of measuring contact angles are well known. In some embodiments, wettable materials can be made from polymeric acids that are not inherently wettable but can be made wettable with selected additives.
适合的聚合物主链的实例包括但不限于:聚烯烃、聚丙烯酸酯、聚甲基丙烯酸酯、聚酰亚胺、聚酰胺、芳族聚酰胺、聚丙烯酰胺、聚苯乙烯以及它们的共聚物,所有这些均为高度氟化的;在一些实施方案中为完全氟化的。Examples of suitable polymer backbones include, but are not limited to: polyolefins, polyacrylates, polymethacrylates, polyimides, polyamides, aramids, polyacrylamides, polystyrene, and copolymers thereof compounds, all of which are highly fluorinated; in some embodiments fully fluorinated.
在一个实施方案中,酸性基团为磺酸基团或磺酰亚胺基团。磺酰亚胺基团具有下式:In one embodiment, the acidic group is a sulfonic acid group or a sulfonimide group. A sulfonimide group has the formula:
-SO2-NH-SO2-R-SO 2 -NH-SO 2 -R
其中R为烷基。wherein R is an alkyl group.
在一个实施方案中,酸性基团位于氟化侧链上。在一个实施方案中,氟化侧链选自烷基、烷氧基、酰胺基、醚基、以及它们的组合,所有这些均为完全氟化的。In one embodiment, the acidic group is located on the fluorinated side chain. In one embodiment, the fluorinated side chains are selected from the group consisting of alkyl, alkoxy, amide, ether, and combinations thereof, all of which are fully fluorinated.
在一个实施方案中,HFAP具有高度氟化的烯烃主链,并具有高度氟化的烷基磺酸盐(酯)、高度氟化的醚磺酸盐(酯)、高度氟化的酯磺酸盐(酯)、或高度氟化的醚磺酰亚胺侧基。在一个实施方案中,HFAP是具有全氟-醚-磺酸侧链的全氟烯烃。在一个实施方案中,聚合物为1,1-二氟乙烯与2-(1,1-二氟-2-(三氟甲基)烯丙氧基)-1,1,2,2-四氟乙磺酸的共聚物。在一个实施方案中,聚合物为乙烯与2-(2-(1,2,2-三氟乙烯氧基)-1,1,2,3,3,3-六氟丙氧基)-1,1,2,2-四氟乙磺酸的共聚物。这些共聚物可被制成对应的磺酰氟聚合物,然后可再转化为磺酸形式。In one embodiment, the HFAP has a highly fluorinated olefin backbone and has highly fluorinated alkyl sulfonates, highly fluorinated ether sulfonates, highly fluorinated ester sulfonates salts (esters), or highly fluorinated ether sulfonimide side groups. In one embodiment, the HFAP is a perfluoroalkene with perfluoro-ether-sulfonic acid side chains. In one embodiment, the polymer is 1,1-difluoroethylene and 2-(1,1-difluoro-2-(trifluoromethyl)allyloxy)-1,1,2,2-tetrafluoroethylene Copolymers of fluoroethanesulfonic acid. In one embodiment, the polymer is ethylene and 2-(2-(1,2,2-trifluoroethyleneoxy)-1,1,2,3,3,3-hexafluoropropoxy)-1 , 1,2,2-copolymers of tetrafluoroethanesulfonic acid. These copolymers can be made into the corresponding sulfonyl fluoride polymers, which can then be converted to the sulfonic acid form.
在一个实施方案中,HFAP为氟化的和部分磺化的聚(亚芳基醚砜)的均聚物或共聚物。该共聚物可以是嵌段共聚物。In one embodiment, the HFAP is a homopolymer or copolymer of fluorinated and partially sulfonated poly(arylene ether sulfone). The copolymer may be a block copolymer.
在一个实施方案中,HFAP为具有式IX的磺酰亚胺聚合物:In one embodiment, HFAP is a sulfonimide polymer having formula IX:
其中:in:
Rf选自高度氟化的亚烷基、高度氟化的杂亚烷基、高度氟化的亚芳基、以及高度氟化的杂亚芳基,它们可由一个或多个醚氧取代;以及 R is selected from highly fluorinated alkylene, highly fluorinated heteroalkylene, highly fluorinated arylene, and highly fluorinated heteroarylene, which may be substituted by one or more ether oxygen; and
n为至少4。n is at least 4.
在式IX的一个实施方案中,Rf为全氟烷基。在一个实施方案中,Rf为全氟丁基。在一个实施方案中,Rf包含醚氧。在一个实施方案中,n大于10。In one embodiment of formula IX, Rf is perfluoroalkyl. In one embodiment, Rf is perfluorobutyl. In one embodiment, Rf comprises ether oxygen. In one embodiment, n is greater than 10.
在一个实施方案中,HFAP包含高度氟化的聚合物主链和具有式X的侧链:In one embodiment, the HFAP comprises a highly fluorinated polymer backbone and side chains having the formula X:
其中:in:
R15为高度氟化的亚烷基或高度氟化的杂亚烷基;R 15 is highly fluorinated alkylene or highly fluorinated heteroalkylene;
R16为高度氟化的烷基或高度氟化的芳基;以及R 16 is highly fluorinated alkyl or highly fluorinated aryl; and
a为0或1至4的整数。a is 0 or an integer of 1 to 4;
在一个实施方案中,HFAP具有式XI:In one embodiment, the HFAP has formula XI:
其中:in:
R16为高度氟化的烷基或高度氟化的芳基;R 16 is highly fluorinated alkyl or highly fluorinated aryl;
c独立地为0或1至3的整数;以及c is independently 0 or an integer from 1 to 3; and
n为至少4。n is at least 4.
HFAP的合成在例如A.Feiring等人,“J.Fluorine Chemistry”,2000,105,129-135;A.Feiring等人,“Macromolecules”,2000,33,9262-9271;D.D.Desmarteau,“J.Fluorine Chem.”,1995,72,203-208;A.J.Appleby等人,“J.Electrochem.Soc.”,1993,140(1),109-111;和Desmarteau的美国专利5,463,005中有所描述。The synthesis of HFAP is described, for example, in A. Feiring et al., "J. Fluorine Chemistry", 2000, 105, 129-135; A. Feiring et al., "Macromolecules", 2000, 33, 9262-9271; D.D. Desmarteau, "J. Fluorine Chem.", 1995, 72, 203-208; A.J. Appleby et al., "J. Electrochem. Soc.", 1993, 140(1), 109-111; and US Patent 5,463,005 to Desmarteau.
在一个实施方案中,HFAP还包含衍生自至少一种高度氟化的烯键不饱和化合物的重复单元。全氟烯烃包含2至20个碳原子。代表性全氟烯烃包括但不限于:四氟乙烯、六氟丙烯、全氟-(2,2-二甲基-1,3-二氧杂环戊烯)、全氟-(2-亚甲基-4-甲基-1,3-二氧杂环戊烷)、CF2=CFO(CF2)tCF=CF2(其中t为1或2)和Rf”OCF=CF2(其中Rf”为具有1至约10个碳原子的饱和全氟烷基)。在一个实施方案中,共聚单体为四氟乙烯。In one embodiment, the HFAP further comprises repeat units derived from at least one highly fluorinated ethylenically unsaturated compound. Perfluoroolefins contain 2 to 20 carbon atoms. Representative perfluoroalkenes include, but are not limited to: tetrafluoroethylene, hexafluoropropylene, perfluoro-(2,2-dimethyl-1,3-dioxole), perfluoro-(2-methylene base-4-methyl-1,3-dioxolane), CF 2 =CFO(CF 2 ) t CF=CF 2 (where t is 1 or 2) and R f "OCF=CF 2 (where Rf " is a saturated perfluoroalkyl group having 1 to about 10 carbon atoms). In one embodiment, the comonomer is tetrafluoroethylene.
在一个实施方案中,HFAP为成胶聚合酸。如本文所用,术语“成胶”是指不溶于水并且当分散到含水介质中时形成胶体的材料。成胶聚合酸通常具有在约10,000至约4,000,000范围内的分子量。在一个实施方案中,聚合酸具有约100,000至约2,000,000的分子量。胶体粒度通常在2纳米(nm)至约140nm的范围内。在一个实施方案中,胶体具有2nm至约30nm的粒度。可使用具有酸性质子的高度氟化的成胶聚合材料。上文所述的某些聚合物可以非酸形式形成,例如,以盐、酯、或磺酰氟的形式。它们将被转化为酸形式以用于制备下文所述的导电组合物。In one embodiment, the HFAP is a gel-forming polymeric acid. As used herein, the term "gel-forming" refers to a material that is insoluble in water and forms a colloid when dispersed in an aqueous medium. The gel-forming polymeric acid typically has a molecular weight in the range of about 10,000 to about 4,000,000. In one embodiment, the polymeric acid has a molecular weight of from about 100,000 to about 2,000,000. Colloidal particle sizes typically range from 2 nanometers (nm) to about 140 nm. In one embodiment, the colloid has a particle size of 2 nm to about 30 nm. Highly fluorinated gel-forming polymeric materials with acidic protons can be used. Certain polymers described above can be formed in non-acid form, for example, as salts, esters, or sulfonyl fluorides. They will be converted to the acid form for use in the preparation of the conductive compositions described below.
在一些实施方案中,HFAP包含高度氟化碳主链和由下式表示的侧链In some embodiments, the HFAP comprises a highly fluorinated carbon backbone and side chains represented by the formula
-(O-CF2CFRf 3)a-O-CF2CFRf 4SO3E5 -(O-CF 2 CFR f 3 ) a -O-CF 2 CFR f 4 SO 3 E 5
其中Rf 3和Rf 4独立地选自F、Cl或具有1至10个碳原子的高度氟化的烷基,a=0、1或2,以及E5。在某些情况下,E5可以是阳离子,例如Li、Na、或K,并且可转换成酸形式。wherein R f 3 and R f 4 are independently selected from F, Cl or a highly fluorinated alkyl group having 1 to 10 carbon atoms, a=0, 1 or 2, and E 5 . In some cases, E5 can be a cation, such as Li, Na, or K, and can be converted to the acid form.
在一些实施方案中,HFAP可以是美国专利3,282,875以及美国专利4,358,545和4,940,525中所公开的聚合物。在一些实施方案中,HFAP包含全氟化碳主链以及由下式表示的侧链In some embodiments, the HFAP may be a polymer disclosed in US Patent 3,282,875 and US Patents 4,358,545 and 4,940,525. In some embodiments, HFAP comprises a perfluorocarbon backbone with side chains represented by the formula
-O-CF2CF(CF3)-O-CF2CF2SO3E5 -O-CF 2 CF(CF 3 )-O-CF 2 CF 2 SO 3 E 5
其中E5如上文所定义。该类型的HFAP在美国专利3,282,875中有所公开,并可由四氟乙烯(TFE)和全氟化乙烯基醚CF2=CF-O-CF2CF(CF3)-O-CF2CF2SO2F、全氟(3,6-二氧杂-4-甲基-7-辛烯磺酰氟)(PDMOF)发生共聚,随后通过水解磺酰氟基团转化为磺酸根基团,并在必要时进行离子交换以将它们转化为所需的离子形式而制备。美国专利4,358,545和4,940,525中所公开的这种类型的聚合物的实例具有侧链-O-CF2CF2SO3E5,其中E5如上所定义。该聚合物可由四氟乙烯(TFE)和全氟乙烯基醚CF2=CF-O-CF2CF2SO2F、全氟(3-氧杂-4-戊烯磺酰氟)(POPF)发生共聚,随后进行水解,并根据需要进一步进行离子交换而制备。wherein E is as defined above. This type of HFAP is disclosed in US Patent 3,282,875 and can be prepared from tetrafluoroethylene (TFE) and perfluorinated vinyl ether CF2 =CF-O- CF2CF ( CF3 ) -O - CF2CF2SO 2 F. Copolymerization of perfluoro(3,6-dioxa-4-methyl-7-octenesulfonyl fluoride) (PDMOF), followed by conversion of sulfonyl fluoride groups to sulfonate groups by hydrolysis, and in They are prepared by ion exchange where necessary to convert them to the desired ionic form. Examples of polymers of this type disclosed in US Pat. Nos . 4,358,545 and 4,940,525 have side chains -O- CF2CF2SO3E5 , where E5 is as defined above. The polymer can be prepared from tetrafluoroethylene (TFE) and perfluorovinyl ether CF 2 =CF-O-CF 2 CF 2 SO 2 F, perfluoro(3-oxa-4-pentenesulfonyl fluoride) (POPF) It is prepared by copolymerization followed by hydrolysis and, if necessary, further ion exchange.
一种类型的HFAP可以含水分散体的形式从E.I.du Pont deNemours and Company(Wilmington,DE)商购获得。One type of HFAP can contain water Dispersion forms are commercially available from EI du Pont de Nemours and Company (Wilmington, DE).
4.无机纳米颗粒4. Inorganic nanoparticles
无机纳米颗粒可以是绝缘的或半导电的。Inorganic nanoparticles can be insulating or semiconducting.
在一些实施方案中,无机纳米颗粒为金属硫化物或金属氧化物。半导体金属氧化物的实例包括但不限于:混合价金属氧化物,例如亚锑酸锌;以及非化学计量金属氧化物,例如缺氧的三氧化钼、五氧化钒等。绝缘金属氧化物的实例包括但不限于二氧化钛、氧化锆、三氧化钼、氧化钒、以及氧化铝In some embodiments, the inorganic nanoparticles are metal sulfides or metal oxides. Examples of semiconducting metal oxides include, but are not limited to: mixed valence metal oxides, such as zinc antimonite; and non-stoichiometric metal oxides, such as oxygen-deficient molybdenum trioxide, vanadium pentoxide, and the like. Examples of insulating metal oxides include, but are not limited to, titanium dioxide, zirconia, molybdenum trioxide, vanadium oxide, and aluminum oxide
在一些实施方案中,使用偶联剂对纳米颗粒进行表面处理以与含水导电聚合物相容。表面改性剂的种类包括但不限于硅烷、钛酸盐、锆酸盐、铝酸盐以及聚合物分散剂。表面改性剂包含化学官能团,其实例包括但不限于腈、氨基、氰基、烷基氨基、烷基、芳基、烯基、烷氧基、芳氧基、磺酸、丙烯酸、磷酸,以及上述酸的碱金属盐、丙烯酸盐(酯)、磺酸盐(酯)、酰胺基磺酸盐(酯)、醚、醚磺酸盐(酯)、酯磺酸盐(酯)、烷硫基、以及芳硫基。在一个实施方案中,化学官能团可包括连接基团例如环氧基、烷基乙烯基和芳基乙烯基,以在接下来的上层中与纳米复合材料或空穴传输材料中的导电聚合物反应。在一个实施方案中,表面改性剂为氟化的或全氟化的,例如四氟-三氟乙基-乙烯基-醚-三乙氧基硅烷、全氟丁烷-三乙氧基硅烷、全氟辛基三乙氧基硅烷、双(三氟丙基)-四甲基二硅氮烷、以及双(3-三乙氧甲硅烷基)丙基四硫化物。In some embodiments, the nanoparticles are surface treated with a coupling agent to be compatible with the aqueous conductive polymer. Types of surface modifiers include, but are not limited to, silanes, titanates, zirconates, aluminates, and polymeric dispersants. Surface modifiers contain chemical functional groups, examples of which include, but are not limited to, nitrile, amino, cyano, alkylamino, alkyl, aryl, alkenyl, alkoxy, aryloxy, sulfonic acid, acrylic acid, phosphoric acid, and Alkali metal salts, acrylates (esters), sulfonates (esters), amidosulfonates (esters), ethers, ether sulfonates (esters), ester sulfonates (esters), alkylthio , and arylthio. In one embodiment, chemical functionalities may include linking groups such as epoxy, alkylvinyl, and arylvinyl to react with the conducting polymer in the nanocomposite or hole transport material in the next upper layer . In one embodiment, the surface modifier is fluorinated or perfluorinated, such as tetrafluoro-trifluoroethyl-vinyl-ether-triethoxysilane, perfluorobutane-triethoxysilane , perfluorooctyltriethoxysilane, bis(trifluoropropyl)-tetramethyldisilazane, and bis(3-triethoxysilyl)propyltetrasulfide.
5.制备掺杂型导电聚合物组合物5. Preparation of Doped Conductive Polymer Composition
在下列讨论中,将以单数形式论及导电聚合物、HFAP和无机纳米颗粒。然而,应当理解的是,可使用这些物质中的任意一种以上或全部。In the following discussion, conductive polymers, HFAP, and inorganic nanoparticles will be referred to in the singular. However, it should be understood that any more than one or all of these substances may be used.
新型导电聚合物组合物的制备方法为:首先形成掺杂型导电聚合物,然后添加无机纳米颗粒。The preparation method of the novel conductive polymer composition is as follows: first forming a doped conductive polymer, and then adding inorganic nanoparticles.
掺杂型导电聚合物的形成方法为:在含水介质中存在HFAP的情况下,让前驱单体发生氧化聚合反应。聚合反应在已公布的美国专利申请2004/0102577、2004/0127637和2005/205860中有所描述。The doped conductive polymer is formed by oxidative polymerization of precursor monomers in the presence of HFAP in an aqueous medium. Polymerization reactions are described in published US patent applications 2004/0102577, 2004/0127637 and 2005/205860.
可以固体形式直接将无机纳米颗粒添加到掺杂型导电聚合物分散体中。在一些实施方案中,将无机纳米颗粒分散在水溶液中,再将该分散体与掺杂型导电聚合物分散体混合。纳米颗粒与导电聚合物的重量比率在0.1至10.0的范围内。Inorganic nanoparticles can be added directly to the doped conductive polymer dispersion in solid form. In some embodiments, the inorganic nanoparticles are dispersed in an aqueous solution, and the dispersion is mixed with the doped conductive polymer dispersion. The weight ratio of nanoparticles to conductive polymer is in the range of 0.1 to 10.0.
在一些实施方案中,在添加无机颗粒之前或之后调高pH值。掺杂型导电聚合物和无机纳米颗粒的分散体可在这样形成的为约2至中性的pH值下保持稳定。pH值的调节可在添加纳米颗粒之前使用阳离子交换树脂进行处理。在一些实施方案中,通过添加碱性水溶液调节pH值。碱的阳离子可以为但不限于碱金属、碱土金属、铵和烷基铵。在一些实施方案中,碱金属要优于碱土金属阳离子。In some embodiments, the pH is adjusted up before or after adding the inorganic particles. The dispersion of doped conductive polymer and inorganic nanoparticles can be stable at the pH value thus formed of about 2 to neutral. Adjustment of the pH can be performed using a cation exchange resin prior to the addition of the nanoparticles. In some embodiments, the pH is adjusted by adding an aqueous alkaline solution. The cation of the base may be, but is not limited to, alkali metals, alkaline earth metals, ammonium and alkylammonium. In some embodiments, alkali metals are preferred over alkaline earth metal cations.
由本文所述的新型导电组合物制成的薄膜在下文中称为“本文所述的新型薄膜”。可使用任何液相沉积技术制备该薄膜,包括连续和不连续技术。连续沉积技术包括但不限于旋涂、凹版涂布、帘式涂布、浸涂、槽模涂布、喷涂和连续喷涂。不连续沉积技术包括但不限于喷墨印刷、凹版印刷、以及丝网印刷。Films made from the novel conductive compositions described herein are hereinafter referred to as "the novel films described herein". The films can be prepared using any liquid deposition technique, including continuous and discontinuous techniques. Continuous deposition techniques include, but are not limited to, spin coating, gravure coating, curtain coating, dip coating, slot die coating, spray coating, and continuous spray coating. Discontinuous deposition techniques include, but are not limited to, inkjet printing, gravure printing, and screen printing.
由此形成的薄膜为光滑的并相对透明,具有大于1.4的折射指数(在460nm的波长下),并且可具有在10-7至10-3S/cm范围内的导电率。The films thus formed are smooth and relatively transparent, have a refractive index (at a wavelength of 460 nm) greater than 1.4, and may have electrical conductivity in the range of 10 −7 to 10 −3 S/cm.
6.缓冲层6. Buffer layer
在本发明的另一个实施方案中,提供了由含有新型导电聚合物组合物的含水分散体沉积的缓冲层。术语“缓冲层”或“缓冲材料”旨在表示导电或半导电材料,并且可在有机电子器件中具有一种或多种功能,包括但不限于下面的层的平面化、电荷传输和/或电荷注入性能、清除杂质(如氧或金属离子),以及其他有利于或可改善有机电子器件性能的方面。术语“层”与术语“膜”可交替使用,是指覆盖所需区域的涂层。该术语不受尺寸的限制。所述区域可以大如整个器件,也可以小如特定功能区例如实际可视显示器,或者小如单个子像素。层和薄膜可以由任何常规的沉积技术形成,包括气相沉积、液相沉积(连续和不连续技术)、以及热转移。连续沉积技术包括但不限于旋涂、凹版涂布、帘式涂布、浸涂、槽模涂布、喷涂和连续喷涂。不连续沉积技术包括但不限于喷墨印刷、凹版印刷、以及丝网印刷。In another embodiment of the present invention there is provided a buffer layer deposited from an aqueous dispersion comprising the novel conductive polymer composition. The term "buffer layer" or "buffer material" is intended to mean a conductive or semiconductive material, and may serve one or more functions in an organic electronic device, including but not limited to planarization of underlying layers, charge transport and/or Charge injection properties, scavenging of impurities such as oxygen or metal ions, and other aspects that are beneficial or can improve the performance of organic electronic devices. The term "layer" is used interchangeably with the term "film" and refers to a coating covering a desired area. The term is not limited by size. The area can be as large as an entire device, or as small as a specific functional area such as the actual visual display, or as small as a single sub-pixel. Layers and films can be formed by any conventional deposition technique, including vapor deposition, liquid deposition (continuous and discontinuous techniques), and thermal transfer. Continuous deposition techniques include, but are not limited to, spin coating, gravure coating, curtain coating, dip coating, slot die coating, spray coating, and continuous spray coating. Discontinuous deposition techniques include, but are not limited to, inkjet printing, gravure printing, and screen printing.
新型导电聚合物组合物的干膜一般不能再分散于水中。因此可将缓冲层作为多个薄层施加。此外,缓冲层可被不同的水溶性或水分散性材料的层包覆而不会受到损坏。令人惊讶的是,据发现包含新型导电聚合物组合物的缓冲层具有改善的可润湿性。Dry films of the novel conductive polymer compositions are generally not redispersible in water. The buffer layer can thus be applied as several thin layers. Furthermore, the buffer layer can be coated with layers of different water-soluble or water-dispersible materials without damage. Surprisingly, it was found that the buffer layer comprising the novel conductive polymer composition has improved wettability.
在另一个实施方案中,提供了由含水分散体沉积的缓冲层,该含水分散体含有与其他水溶性或水分散性材料混合的新型导电聚合物组合物。可以添加的材料类型的实例包括但不限于聚合物、染料、涂布助剂、有机和无机导电油墨和糊剂、电荷传输材料、交联剂、以及它们的组合。其他水溶性或水分散性材料可以为简单分子或聚合物。适合的聚合物的实例包括但不限于导电聚合物,例如聚噻吩、聚苯胺、聚吡咯、聚乙炔、聚(噻吩并噻吩)、以及它们的组合。In another embodiment, there is provided a buffer layer deposited from an aqueous dispersion containing the novel conductive polymer composition in admixture with other water-soluble or water-dispersible materials. Examples of the types of materials that can be added include, but are not limited to, polymers, dyes, coating aids, organic and inorganic conductive inks and pastes, charge transport materials, crosslinkers, and combinations thereof. Other water soluble or dispersible materials can be simple molecules or polymers. Examples of suitable polymers include, but are not limited to, conductive polymers such as polythiophene, polyaniline, polypyrrole, polyacetylene, poly(thienothiophene), and combinations thereof.
7.电子器件7. Electronic devices
在本发明的另一个实施方案中,提供了包含至少一个电活性层的电子器件,该电活性层位于两个电接触层之间,其中该器件还包括新型缓冲层。术语“电活性”在涉及层或材料时旨在表示表现出电子特性或电辐射特性的层或材料。电活性层材料可发出辐射或在接受辐射时表现出电子-空穴对的浓度变化。In another embodiment of the present invention there is provided an electronic device comprising at least one electroactive layer positioned between two electrical contact layers, wherein the device further comprises a novel buffer layer. The term "electroactive" when referring to a layer or material is intended to mean a layer or material exhibiting electronic or electroradiative properties. Electroactive layer materials can emit radiation or exhibit a change in concentration of electron-hole pairs when receiving radiation.
如图1所示,典型的器件100具有阳极层110、缓冲层120、电活性层130和阴极层150。邻近阴极层150的是任选的电子注入/传输层140。As shown in FIG. 1 , a
器件可包括载体或基板(未示出),其可以邻近阳极层110或阴极层150。最常见的是,载体邻近阳极层110。载体可以是柔性的或刚性的、有机的或无机的。载体材料的实例包括但不限于玻璃、陶瓷、金属和塑料薄膜。The device may include a carrier or substrate (not shown), which may be adjacent to the
阳极层110为与阴极层150相比对注入空穴更有效的电极。阳极可包含包括金属、混合金属、合金、金属氧化物或混合氧化物的材料。合适的材料包括2族元素(即铍、镁、钙、锶、钡、镭)、11族元素、4、5和6族元素、以及8至10族过渡元素的混合氧化物。如果阳极层110要为透光的,则可以使用12、13和14族元素的混合氧化物,例如氧化铟锡。如本文所用,短语“混合氧化物”是指具有选自2族元素或12、13或14族元素的两种或更多种不同阳离子的氧化物。用于阳极层110的材料的一些非限制性具体实例包括但不限于氧化铟锡(“ITO”)、氧化铟锌、氧化铝锡、金、银、铜、以及镍。阳极也可包含有机材料,尤其是导电聚合物,例如聚苯胺,包括“Flexible light-emitting diodesmade from soluble conducting polymer”,“Nature”第357卷,第477至479页(1992年6月11日)中所述的示例性材料。阳极和阴极中的至少一个应当是至少部分透明的,以使产生的光线能够被观察到。The
可以通过化学或物理气相沉积方法或旋铸法来形成阳极层110。化学气相沉积可以等离子体增强化学气相沉积(“PECVD”)或金属有机化学气相沉积(“MOCVD”)的形式实施。物理气相沉积可包括所有的溅射(包括离子束溅射),以及电子束蒸发和电阻蒸发形式。物理气相沉积的具体形式包括射频磁控溅射和电感耦合等离子体物理气相沉积(“IMP-PVD”)。这些沉积技术在半导体制制造领域是熟知的。The
在一个实施方案中,阳极层110在平版印刷操作期间被图案化。图案可以根据需要变化。层可通过以下方法形成图案,例如:在施加第一电接触层材料之前,将图案化掩模或抗蚀剂定位在第一柔性复合材料屏障结构上。或者作为另一种选择,可以整体层(也称为毯状沉积)的形式施加所述层,随后使用例如图案化抗蚀剂层和湿化学或干蚀刻技术将其图案化。还可以使用本领域熟知的其他图案化方法。In one embodiment, the
缓冲层120包含本文所述的新型导电组合物。由掺入有HFAP的导电聚合物制成的缓冲层一般是不可被有机溶剂润湿的并具有低于1.4的折射指数(在460nm的波长下)。本文所述的缓冲层可具有更高的可润湿性,因此更易由非极性有机溶剂涂覆下一层。本文所述的缓冲层还可具有大于1.4的折射指数(在460nm下)。通常使用本领域技术人员熟知的多种技术将缓冲层沉积到基底上。如上所述,典型的沉积技术包括气相沉积、液相沉积(连续和不连续技术)、以及热转移。
任选层(未示出)可存在于缓冲层120和电活性层130之间。该层可以包含空穴传输材料。空穴传输材料的实例例如由Y.Wang在“Kirk-OthmerEncyclopedia of Chemical Technology”,第四版,第18卷,第837至860页,1996中有所综述。空穴传输分子和空穴传输聚合物均可使用。常用的空穴传输分子包括但不仅限于:4,4’,4”-三(N,N-二苯基-氨基)-三苯胺(TDATA);4,4’,4”-三(N-3-甲基苯基-N-苯基-氨基)-三苯胺(MTDATA);N,N’-二苯基-N,N’-双(3-甲基苯基)-[1,1’-联苯基]-4,4’-二胺(TPD);1,1-双[(二-4-甲苯基氨基)苯基]环己烷(TAPC);N,N’-双(4-甲基苯基)-N,N’-双(4-乙基苯基)-[1,1’-(3,3’-二甲基)联苯基]-4,4’-二胺(ETPD);四-(3-甲基苯基)-N,N,N’,N’-2,5-苯二胺(PDA);α-苯基-4-N,N-二苯基氨基苯乙烯(TPS);对-(二乙基氨基)苯甲醛二苯腙(DEH);三苯胺(TPA);双[4-(N,N-二乙基氨基)-2-甲基苯基](4-甲基苯基)甲烷(MPMP);1-苯基-3-[对-(二乙基氨基)苯乙烯基]-5-[对-(二乙基氨基)苯基]吡唑啉(PPR或DEASP);1,2-反式-双(9H-咔唑-9-基)环丁烷(DCZB);N,N,N’,N’-四(4-甲基苯基)-(1,1’-联苯基)-4,4’-二胺(TTB);N,N’-双(萘-1-基)-N,N’-双-(苯基)对二氨基联苯(α-NPB);以及卟啉化合物例如铜酞菁。常用的空穴传输聚合物包括但不限于聚乙烯咔唑、(苯基甲基)聚硅烷、聚(二氧噻吩)、聚苯胺、以及聚吡咯。还可通过将空穴传输分子诸如上述那些掺入到聚合物诸如聚苯乙烯和聚碳酸酯中,来获得空穴传输聚合物。An optional layer (not shown) may be present between
根据器件的应用,电活性层130可以是通过外加电压激活的发光层(例如在发光二极管或发光电化学电池中),或是可响应辐射能并在有或无外加偏压下产生信号的材料层(例如在光电探测器中)。在一个实施方案中,电活性材料为有机电致发光(“EL”)材料。任何EL材料均可用于器件中,包括但不限于小分子有机荧光化合物、荧光和磷光金属络合物、共轭聚合物、以及它们的混合物。荧光化合物的实例包括但不限于嵌二萘、二萘嵌苯、红荧烯、香豆素、它们的衍生物、以及它们的混合物。金属络合物的实例包括但不限于金属螯合8-羟基喹啉化合物,例如三(8-羟基喹啉)铝(Alq3);环金属铱和铂电致发光化合物,例如在Petrov等人的美国专利6,670,645以及已公布的PCT专利申请WO 03/063555和WO 2004/016710中所公开的铱与苯基吡啶、苯基喹啉、或苯基嘧啶配体的络合物,以及在例如已公布的PCT专利申请WO 03/008424、WO 03/091688、以及WO03/040257中所述的有机金属络合物,以及它们的混合物。Thompson等人在美国专利6,303,238中,以及Burrows和Thompson在已公布的PCT专利申请WO00/70655和WO 01/41512中已描述了包含带电基质材料和金属络合物的电致发光发射层。共轭聚合物的实例包括但不限于聚(苯撑乙烯)、聚芴、聚(螺二芴)、聚噻吩、聚(对亚苯基)、它们的共聚物,以及它们的混合物。Depending on the application of the device, the
任选的层140可同时起到促进电子注入/传输的作用,还可用作限制层以防止在层界面上发生猝灭反应。更具体地讲,如果层130和层150以其他方式直接接触,则层140可以促进电子迁移率并减小猝灭反应的可能性。任选层140的材料的实例包括但不限于金属螯合8-羟基喹啉化合物,例如双(2-甲基-8-羟基喹啉)(对苯基-酚氧基)铝(III)(BAlQ)以及三(8-羟基喹啉)铝(Alq3);四(8-羟基喹啉)锆;唑化合物,例如2-(4-联苯基)-5-(4-叔丁基苯基)-1,3,4-噁二唑(PBD)、3-(4-联苯基)-4-苯基-5-(4-叔丁基苯基)-1,2,4-三唑(TAZ)和1,3,5-三(苯基-2-苯并咪唑)苯(TPBI);喹喔啉衍生物,例如2,3-双(4-氟代苯基)喹喔啉;菲咯啉衍生物,例如9,10-二苯基菲咯啉(DPA)和2,9-二甲基-4,7-二苯基-1,10-菲咯啉(DDPA);以及它们的任何一种或多种的组合。作为另外一种选择,任选层140可以是无机的,并包含BaO、LiF、Li2O等。
阴极层150是对注入电子或负载流子特别有效的电极。阴极层150可为比第一电接触层(在这种情况下为阳极层110)功函更低的任何金属或非金属。如本文所用,术语“更低的功函”旨在表示具有不大于约4.4eV功函的材料。如本文所用,“更高的功函”旨在表示具有至少大约4.4eV功函的材料。The
用于阴极层的材料可选自1族碱金属(例如锂、钠、钾、铷、铯)、2族金属(例如镁、钙、钡等)、12族金属、镧系元素(例如铈、钐、铕等)和锕系元素(例如钍、铀等)。还可以使用诸如铝、铟、钇、以及它们的组合的材料。用于阴极层150的材料的具体非限制性实例包括但不限于钡、锂、铈、铯、铕、铷、钇、镁、钐、以及它们的合金和组合。Materials for the cathode layer may be selected from group 1 alkali metals (e.g. lithium, sodium, potassium, rubidium, cesium), group 2 metals (e.g. magnesium, calcium, barium, etc.), group 12 metals, lanthanides (e.g. cerium, samarium, europium, etc.) and actinides (such as thorium, uranium, etc.). Materials such as aluminum, indium, yttrium, and combinations thereof may also be used. Specific non-limiting examples of materials for
通常通过化学或物理气相沉积方法形成阴极层150。在一些实施方案中,阴极层将被图案化,如上文涉及阳极层110所论述的那样。
器件中的其他层可根据这些层所要提供的功能由已知在这些层中可用的任何材料制成。Other layers in the device may be made of any material known to be useful in these layers, depending on the function these layers are intended to provide.
在一些实施方案中,在接触层150上沉积封装层(未示出),以防止不可取的组分例如水和氧进入器件100。这些组分会对有机层130造成不良影响。在一个实施方案中,封装层为阻挡层或薄膜。在一个实施方案中,封装层为玻璃盖。In some embodiments, an encapsulation layer (not shown) is deposited over
尽管未示出,但应当理解的是器件100可包括附加层。可使用本领域已知或未知的其他层。此外,任何上述层均可包括两个或更多个亚层,或者可形成层状结构。作为另外一种选择,可对阳极层110、空穴传输层120、电子传输层140、阴极层150、以及其他层的某些或全部进行处理,尤其是表面处理,以增强载荷子的传输效率或器件的其他物理特性。对各组件层材料的选择优选地通过平衡以下目标来确定:在为器件提供高器件效率的同时考虑器件使用寿命、制造时间和复杂性因素,以及本领域技术人员所理解的其他因素。应当理解,对本领域的普通技术人员而言,确定最佳组件、组件配置和组成特性均是常规的。Although not shown, it should be understood that
在一个实施方案中,不同的层具有下列厚度范围:阳极110,500至,在一个实施方案中为1000至;缓冲层120,50至,在一个实施方案中为200至;光敏层130,10至,在一个实施方案中为100至;任选的电子传输层140,50至,在一个实施方案中为100至;阴极150,200至,在一个实施方案中为300至。器件内电子-空穴复合区域的位置可受到每层相对厚度的影响,继而影响器件的发射光谱。因此,选择电子传输层的厚度时,应使得电子-空穴复合区域位于发光层中。所需的各层厚度的比率将取决于所用材料的确切性质。In one embodiment, the different layers have the following thickness ranges:
在操作中,将由合适电源(未示出)产生的电压施加到器件100上。因此电流流经器件100的层。电子进入有机聚合物层,释放光子。在被称为有源矩阵OLED显示器的某些OLED中,光敏有机膜的各沉积物可独立地被电流通路激发,从而导致各像素发光。在被称为无源矩阵OLED显示器的某些OLED中,光敏有机膜的沉积物可被电接触层的行和列激发。In operation, a voltage generated by a suitable power supply (not shown) is applied across
虽然本文所述的那些方法和材料的相似者或等同者均可用于本发明的实践或检验,但是合适的方法和材料是如下文所述的那些。本文提及的所有出版物、专利申请、专利、以及其他参考均全文以引用方式并入本文。如发生矛盾,以本说明书(包括定义)为准。此外,材料、方法和实施例仅是例证性的并且不旨在进行限制。Although methods and materials similar or equivalent to those described herein can be used in the practice or testing of the present invention, suitable methods and materials are those described below. All publications, patent applications, patents, and other references mentioned herein are hereby incorporated by reference in their entirety. In case of conflict, the present specification, including definitions, will control. In addition, the materials, methods, and examples are illustrative only and not intended to be limiting.
应当理解,为清楚起见,在参照不同实施方案的上文和下文中所描述的本发明的那些特点也可在单个实施方案中以组合的方式给出。反之,为简化起见在参照单个实施方案中描述的本发明的多个特点也可以分别给出,或以任何子组合给出。此外,范围内提出的相关数值包括所述范围内的每个值。It is to be understood that, for clarity, those features of the invention which are described above and below with reference to different embodiments may also be presented in combination in a single embodiment. Conversely, various features of the invention which are, for brevity, described with reference to a single embodiment, may also be presented separately or in any subcombination. Further, reference to numerical values stated in ranges includes each and every value within that range.
实施例 Example
比较实施例AComparative Example A
该比较实施例示出了PAni/(聚(四氟乙烯)/全氟醚磺酸)薄膜在不添加无机纳米颗粒的情况下的低导电率和不可润湿性。This comparative example shows that PAni/ (Low conductivity and non-wettability of poly(tetrafluoroethylene)/perfluoroethersulfonic acid) thin films without the addition of inorganic nanoparticles.
用于该实施例的PAni/分散体采用EW(酸当量)为1000的含水胶态分散体制备。采用与美国专利6,150,426实施例1第2部分相似的方法制备25%(w/w)的分散体,不同的是温度为约270℃,然后用水将其稀释成12.0%(w/w)的分散体以用于聚合反应。The PAni/ The dispersion adopts water containing water with EW (acid equivalent weight) of 1000 Colloidal Dispersion Preparation. 25% (w/w) of The dispersion, except that the temperature was about 270°C, was then diluted with water to a 12.0% (w/w) dispersion for polymerization.
在500mL的反应釜中加入96.4g固体含量为12%的含水分散体(11.57mmol SO3H基团)和103g水。使用安装有双级螺旋桨叶的顶置式搅拌器,以300RPM的转速搅拌稀释的。向该稀释的分散体中迅速加入:溶于15mL水的1.21g(5.09mmol)过硫酸钠(Na2S2O8),以及溶于266微升(9.28mmol)HCl和20mL水的422微升(4.63mmol)苯胺。聚合反应的液体变得不透明并且非常粘稠,但在5分钟内无可见的颜色变化。加入约20mg硫酸铁,但仍无可见变化。然而,聚合反应的液体在30分钟后开始变蓝并在之后变为绿色。大约8小时后,向聚合反应混合物中加入Dowex M31和Dowex M43离子交换树脂各25g,以及100g去离子水。将混合物过夜搅拌,然后用滤纸过滤。向滤液中加入100g去离子水以降低粘度。将滤液分成五等份。In a 500mL reactor, add 96.4g of water containing 12% solids Dispersion (11.57mmol SO3H groups) and 103g water. Using an overhead stirrer equipped with two-stage propeller blades, stir the diluted . to the diluted The dispersion was quickly added: 1.21 g (5.09 mmol) of sodium persulfate (Na 2 S 2 O 8 ) dissolved in 15 mL of water, and 422 µl (4.63 mmol) of 266 µl (9.28 mmol) of HCl and 20 mL of water )aniline. The polymerized liquid became opaque and very viscous, but there was no visible color change within 5 minutes. About 20 mg of ferric sulfate was added, but still no visible change. However, the polymerized liquid started to turn blue after 30 minutes and turned green thereafter. After about 8 hours, 25 grams each of Dowex M31 and Dowex M43 ion exchange resins, and 100 grams of deionized water were added to the polymerization mixture. The mixture was stirred overnight, then filtered through filter paper. 100 g of deionized water was added to the filtrate to reduce the viscosity. The filtrate was divided into five equal portions.
一部分保持原样,不加碱。经测定该部分的pH值为2,并含2.88%(w/w)的PAni/。由该PAni/制备薄膜,然后在130℃下于空气中烘烤。经测定薄膜的室温导电率为1.2×10-8S/cm,其也在表1中示出。将一小滴甲苯滴在一片薄膜上,但是甲苯迅速从薄膜上滚落,这表明该薄膜表面不能被非极性有机溶剂润湿。非极性溶剂通常用于发光聚合物以及发光小分子。A part is left as is without adding alkali. The pH value of this part was determined to be 2, and contained 2.88% (w/w) of PAni/ . by the PAni/ Films were prepared and then baked at 130°C in air. The room temperature conductivity of the film was measured to be 1.2×10 −8 S/cm, which is also shown in Table 1. A small drop of toluene was dropped on a piece of film, but the toluene rolled off the film quickly, which indicated that the surface of the film was not wettable by non-polar organic solvents. Nonpolar solvents are commonly used for light-emitting polymers as well as light-emitting small molecules.
向第二部分pH值为2的PAni/中加入0.1M NaOH水溶液,将pH值调至5.0。经测定,该部分含Na+分散体包含2.89%(w/w)的PAni/。由pH值为5.0的PAni/制成的薄膜的导电率经测定为3.8×10-8S/cm,其也在表1中示出。经测试,该PAni/薄膜不能被甲苯润湿。To the second part of PAni/ with a pH value of 2 0.1M NaOH aqueous solution was added to adjust the pH value to 5.0. It was determined that this portion of the Na + -containing dispersion contained 2.89% (w/w) of PAni/ . From PAni/ with a pH value of 5.0 The electrical conductivity of the produced thin film was determined to be 3.8×10 −8 S/cm, which is also shown in Table 1. After testing, the PAni/ The film cannot be wetted by toluene.
实施例1Example 1
该实施例示出了半导体纳米颗粒在增强PAni/(聚(四氟乙烯)/全氟醚磺酸)薄膜的导电率和可润湿性方面的影响。This example shows the role of semiconductor nanoparticles in enhancing PAni/ (Poly(tetrafluoroethylene)/perfluoroether sulfonic acid) film conductivity and wettability.
将比较实施例1中制备的pH值为2和pH值为5.0的PAni/分散体用于说明本发明的实施方案。向5.0166g pH值为2的PAni/分散体中加入1.1313g Celnax CX-Z300H-(一种含水亚锑酸锌分散体,得自NissanChemical Industries,Ltd.Houston,Texas,USA)。CX-Z300H-F2具有为约7的pH值,并含有26.47%(w/w)的亚锑酸锌颗粒,其粒度小于20nm。配方中PAni/聚合物与亚锑酸锌的重量比率为约0.47。混合物形成了稳定的分散体,历经至少五个月也无颗粒沉淀的迹象。在烘干水分后,其还形成了光滑、透明的薄膜。数据清楚地表明Celnax CX-Z300H-中的特定亚锑酸锡颗粒与PAni/相容。然而,为了改善表面光滑度,使粗糙度低于至少5nm,该方法需要通过更耗能的方法加以改善,而不是简单地将两种组分加在一起。经测定,含有PAni/和亚锑酸锌的分散体在室温下的薄膜导电率为6.6×10- 4S/cm(两个薄膜样本的平均值),其也在表1中示出。导电率被提高了四个数量级以上。让一片薄膜与一滴甲苯接触。甲苯容易地在薄膜表面蔓延,这表明该薄膜可被常用的非极性有机溶剂润湿。The pH value prepared in Comparative Example 1 is 2 and the PAni/pH value of 5.0 Dispersions are used to illustrate embodiments of the invention. To 5.0166g PAni/ with a pH value of 2 To the dispersion was added 1.1313g Celnax CX-Z300H- (an aqueous zinc antimonite dispersion available from Nissan Chemical Industries, Ltd. Houston, Texas, USA). CX-Z300H-F2 has a pH of about 7 and contains 26.47% (w/w) zinc antimonite particles with a particle size of less than 20 nm. PAni/ The weight ratio of polymer to zinc antimonite was about 0.47. The mixture formed a stable dispersion with no evidence of particle settling for at least five months. It also forms a smooth, clear film after drying off the moisture. The data clearly show that the Celnax CX-Z300H- Specific tin antimonite particles in PAni/ compatible. However, to improve surface smoothness to a roughness below at least 5nm, this approach needs to be improved by more energy-intensive methods than simply adding the two components together. After determination, containing PAni/ The film conductivity of the dispersion with zinc antimonite at room temperature is 6.6× 10 −4 S /cm (average value of two film samples), which is also shown in Table 1. The conductivity is improved by more than four orders of magnitude. Expose a piece of film to a drop of toluene. Toluene spread easily on the surface of the film, which indicated that the film was wettable by commonly used non-polar organic solvents.
也向pH值为5.0的PAni/中加入CX-Z300H-F2,以确定其对导电率和可润湿性的影响。向5.0666g pH值为5.0的PAni/分散体中加入1.1450g Celnax CX-Z300H-。配方中PAni/聚合物与亚锑酸锌的重量比率为约0.47。混合物形成了稳定的分散体,并无颗粒沉淀的迹象。在烘干水分后,它还形成了光滑、透明的薄膜。数据清楚地表明Celnax CX-Z300H-中的特定亚锑酸锡颗粒与PAni/相容。然而,为了改善表面光滑度,使粗糙度低于至少5nm,该方法需要通过更耗能的方法来加以改善,而不是简单地将两种组分加在一起。经测定,含PAni/和亚锑酸锌的分散体在室温下的薄膜导电率为9.3×10-4S/cm(两个薄膜样本的平均值),其也在表1中示出。导电率被提高了四个数量级以上。让一片薄膜与一滴甲苯接触。甲苯容易地在薄膜表面延展,这表明薄膜可被常用的非极性有机溶剂润湿。Also to PAni/ at pH 5.0 CX-Z300H-F2 was added to determine its effect on conductivity and wettability. To 5.0666g PAni/ with a pH value of 5.0 Add 1.1450g Celnax CX-Z300H- . PAni/ The weight ratio of polymer to zinc antimonite was about 0.47. The mixture formed a stable dispersion with no evidence of particle settling. It also forms a smooth, clear film after drying off the moisture. The data clearly show that the Celnax CX-Z300H- Specific tin antimonite particles in PAni/ compatible. However, to improve surface smoothness down to a roughness below at least 5nm, this approach needs to be improved by a more energy-intensive approach than simply adding the two components together. After determination, containing PAni/ and zinc antimonite dispersion at room temperature had a film conductivity of 9.3×10 −4 S/cm (average of two film samples), which is also shown in Table 1. The conductivity is improved by more than four orders of magnitude. Expose a piece of film to a drop of toluene. Toluene spreads easily on the surface of the film, which indicates that the film is wettable by commonly used non-polar organic solvents.
表1Table 1
CX-Z300H-F2对导电率的影响Effect of CX-Z300H-F2 on conductivity
应注意的是,并不是在所有的上文一般性描述或实施例中所描述的行为都是必须的,一部分具体行为不是必需的,并且除了所描述的那些以外,还可实施一个或多个其他行为。此外,所列的行为的顺序不必是实施它们的顺序。It should be noted that not all of the acts described above in the general description or in the examples are required, that some of the specific acts are not required, and that one or more of the acts may be implemented in addition to those described other behavior. Furthermore, the order in which the acts are listed is not necessarily the order in which they are performed.
在上述说明书中,已参考具体的实施方案描述了各概念。然而,本领域的普通技术人员认识到,在不脱离如下所附权利要求中所述的本发明范围的情况下可进行各种修改和变化。因此,说明书和附图应被认为是示例性而非限制性的,并且所有此类修改形式均旨在被包括在本发明的范围内。In the foregoing specification, concepts have been described with reference to specific embodiments. However, one of ordinary skill in the art recognizes that various modifications and changes can be made without departing from the scope of the present invention as set forth in the claims below. Accordingly, the specification and drawings are to be regarded as illustrative rather than restrictive, and all such modifications are intended to be included within the scope of the present invention.
上文已结合具体的实施方案描述了有益效果、其他优点以及问题的解决方案。然而,有益效果、优点、问题的解决方案、以及可致使任何有益效果、优点或解决方案产生或变得更显著的任何特征不可解释为是任何或所有权利要求的关键、必需或基本特征。Benefits, other advantages, and solutions to problems have been described above in conjunction with specific embodiments. However, benefits, advantages, solutions to problems, and any feature that may cause any benefit, advantage or solution to be produced or made more pronounced, are not to be construed as critical, required or essential features of any or all claims.
应当认识到,为清楚起见,在本文不同实施方案的上下文中所描述的某些特征也可在单个实施方案中以组合方式提供。反之,为简化起见在单个实施方案上下文中所描述的多个特征也可以分别提供,或以任何子组合的方式提供。It will be appreciated that, for clarity, certain features that are described herein in the context of different implementations can also be provided in combination in a single implementation. Conversely, various features which are, for brevity, described in the context of a single embodiment, may also be provided separately or in any subcombination.
本文规定的各种范围内的数值的使用均是以近似值的形式描述的,就如同所述范围内的最小值和最大值前均有“约”字一样。这样,在所述范围之上或之下的微小变化值均可用于获得与这些范围内的值基本上相同的结果。而且,这些范围的公开还旨在作为包括介于最小和最大平均值之间的每个值的连续范围,当一个值的某些组分与不同值的那些混合时,其包括可产生结果的部分值。然而,当公开更宽的和更窄的范围时,使来自一个范围的最小值与来自另一个范围的最大值匹配,反之亦然也在本发明的期望内。The use of numerical values in the various ranges stated herein are stated as approximations, as if the word "about" preceded the minimum and maximum values in the stated ranges. Thus, slight variations in values above and below the stated ranges can be used to obtain substantially the same results as values within these ranges. Moreover, the disclosure of these ranges is also intended to be a continuous range including each value between the minimum and maximum average values, which includes the resulting range when certain components of one value are mixed with those of different values. partial value. However, when wider and narrower ranges are disclosed, it is also within the contemplation of the invention that the minimum value from one range be matched with the maximum value from the other range, and vice versa.
Claims (15)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US95237207P | 2007-07-27 | 2007-07-27 | |
| US60/952,372 | 2007-07-27 | ||
| PCT/US2008/070718 WO2009018009A1 (en) | 2007-07-27 | 2008-07-22 | Aqueous dispersions of electrically conducting polymers containing inorganic nanoparticles |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| CN101688052A true CN101688052A (en) | 2010-03-31 |
Family
ID=39862953
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN200880023383A Pending CN101688052A (en) | 2007-07-27 | 2008-07-22 | Aqueous dispersion of conductive polymer comprising inorganic nanoparticles |
Country Status (6)
| Country | Link |
|---|---|
| EP (1) | EP2173811A1 (en) |
| JP (1) | JP2010534739A (en) |
| KR (1) | KR20100065302A (en) |
| CN (1) | CN101688052A (en) |
| TW (1) | TW200915350A (en) |
| WO (1) | WO2009018009A1 (en) |
Cited By (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN104520383A (en) * | 2012-07-31 | 2015-04-15 | 国际商业机器公司 | Water-dispersible electrically conductive fluorine-containing polyaniline compositions for lithography |
| CN106887522B (en) * | 2010-07-26 | 2018-09-18 | 默克专利有限公司 | Include the device of nanocrystal |
| CN109585830A (en) * | 2018-12-03 | 2019-04-05 | 江苏理工学院 | The sulphur selenium compound and its preparation of coated with conductive polymer and graphene oxide and application |
| CN114502638A (en) * | 2019-10-09 | 2022-05-13 | 大金工业株式会社 | Composition containing fluoropolymer and crosslinked product |
Families Citing this family (644)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100540628C (en) | 2002-09-24 | 2009-09-16 | E.I.内穆尔杜邦公司 | Conductive organic polymer/nanoparticle compositions and methods of application thereof |
| AU2003275203A1 (en) | 2002-09-24 | 2004-04-19 | E.I. Du Pont De Nemours And Company | Water dispersible polythiophenes made with polymeric acid colloids |
| US7351358B2 (en) | 2004-03-17 | 2008-04-01 | E.I. Du Pont De Nemours And Company | Water dispersible polypyrroles made with polymeric acid colloids for electronics applications |
| US9070884B2 (en) | 2005-04-13 | 2015-06-30 | Universal Display Corporation | Hybrid OLED having phosphorescent and fluorescent emitters |
| US8586204B2 (en) | 2007-12-28 | 2013-11-19 | Universal Display Corporation | Phosphorescent emitters and host materials with improved stability |
| US9051344B2 (en) | 2005-05-06 | 2015-06-09 | Universal Display Corporation | Stability OLED materials and devices |
| KR101600624B1 (en) | 2006-02-10 | 2016-03-21 | 유니버셜 디스플레이 코포레이션 | METAL COMPLEXES OF CYCLOMETALLATED IMIDAZO[1,2-f]PHENANTHRIDINE AND DIIMIDAZO[1,2-A:1',2'-C]QUINAZOLINE LIGANDS AND ISOELECTRONIC AND BENZANNULATED ANALOGS THEREOF |
| US8115378B2 (en) | 2006-12-28 | 2012-02-14 | E. I. Du Pont De Nemours And Company | Tetra-substituted chrysenes for luminescent applications |
| US20080191172A1 (en) | 2006-12-29 | 2008-08-14 | Che-Hsiung Hsu | High work-function and high conductivity compositions of electrically conducting polymers |
| US20130032785A1 (en) | 2011-08-01 | 2013-02-07 | Universal Display Corporation | Materials for organic light emitting diode |
| EP3719099B1 (en) | 2007-03-08 | 2022-09-14 | Universal Display Corporation | Phosphorescent materials |
| US9130177B2 (en) | 2011-01-13 | 2015-09-08 | Universal Display Corporation | 5-substituted 2 phenylquinoline complexes materials for light emitting diode |
| JP5401448B2 (en) | 2007-06-01 | 2014-01-29 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニー | Chrysene for deep blue light emitting applications |
| KR101554751B1 (en) | 2007-06-01 | 2015-09-22 | 이 아이 듀폰 디 네모아 앤드 캄파니 | Green luminous material |
| EP2185532B1 (en) | 2007-08-08 | 2016-11-09 | Universal Display Corporation | Benzo-fused thiophene compounds comprising a triphenylene group |
| EP3424918A1 (en) | 2007-08-08 | 2019-01-09 | Universal Display Corporation | Single triphenylene chromophores in phosphorescent light emitting diodes |
| WO2009073245A1 (en) | 2007-12-06 | 2009-06-11 | Universal Display Corporation | Light-emitting organometallic complexes |
| WO2009085344A2 (en) | 2007-12-28 | 2009-07-09 | Universal Display Corporation | Dibenzothiophene-containing materials in phosphorescent light emitting diodes |
| WO2009158555A2 (en) | 2008-06-26 | 2009-12-30 | E.I. Du Pont De Nemours And Company | Organic light-emitting diode luminaires |
| CN102099871A (en) * | 2008-07-22 | 2011-06-15 | E.I.内穆尔杜邦公司 | Aqueous dispersions of electrically conducting polymers containing inorganic nanoparticles |
| WO2010027583A1 (en) | 2008-09-03 | 2010-03-11 | Universal Display Corporation | Phosphorescent materials |
| US9034483B2 (en) | 2008-09-16 | 2015-05-19 | Universal Display Corporation | Phosphorescent materials |
| CN103094490B (en) | 2008-09-25 | 2016-03-09 | 通用显示公司 | Organic Selenium material and the purposes in organic light emitting apparatus thereof |
| US8815415B2 (en) | 2008-12-12 | 2014-08-26 | Universal Display Corporation | Blue emitter with high efficiency based on imidazo[1,2-f] phenanthridine iridium complexes |
| US8263973B2 (en) | 2008-12-19 | 2012-09-11 | E I Du Pont De Nemours And Company | Anthracene compounds for luminescent applications |
| US8932733B2 (en) | 2008-12-19 | 2015-01-13 | E I Du Pont De Nemours And Company | Chrysene derivative host materials |
| US8531100B2 (en) | 2008-12-22 | 2013-09-10 | E I Du Pont De Nemours And Company | Deuterated compounds for luminescent applications |
| US9067947B2 (en) | 2009-01-16 | 2015-06-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US8759818B2 (en) | 2009-02-27 | 2014-06-24 | E I Du Pont De Nemours And Company | Deuterated compounds for electronic applications |
| TW201100480A (en) * | 2009-03-12 | 2011-01-01 | Du Pont | Electrically conductive polymer compositions for coating applications |
| US11910700B2 (en) | 2009-03-23 | 2024-02-20 | Universal Display Corporation | Heteroleptic iridium complexes as dopants |
| US8722205B2 (en) | 2009-03-23 | 2014-05-13 | Universal Display Corporation | Heteroleptic iridium complex |
| US8709615B2 (en) | 2011-07-28 | 2014-04-29 | Universal Display Corporation | Heteroleptic iridium complexes as dopants |
| JP5567656B2 (en) | 2009-04-03 | 2014-08-06 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニー | Electroactive materials |
| JP5587980B2 (en) * | 2009-04-21 | 2014-09-10 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニー | Conductive polymer composition and film made therefrom |
| US8945427B2 (en) | 2009-04-24 | 2015-02-03 | E I Du Pont De Nemours And Company | Electrically conductive polymer compositions and films made therefrom |
| TWI687408B (en) | 2009-04-28 | 2020-03-11 | 美商環球展覽公司 | Iridium complex with methyl-D3 substitution |
| EP2432849B1 (en) | 2009-05-19 | 2015-02-25 | E. I. du Pont de Nemours and Company | Chrysene compounds for luminescent applications |
| US8586203B2 (en) | 2009-05-20 | 2013-11-19 | Universal Display Corporation | Metal complexes with boron-nitrogen heterocycle containing ligands |
| US9133095B2 (en) | 2009-07-01 | 2015-09-15 | E I Du Pont De Nemours And Company | Chrysene compounds for luminescent applications |
| JP5727478B2 (en) | 2009-07-27 | 2015-06-03 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニーE.I.Du Pont De Nemours And Company | Methods and materials for making confinement layers and devices made thereby |
| JP5567675B2 (en) | 2009-08-13 | 2014-08-06 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニー | Chrysene derivative material |
| KR20120055713A (en) | 2009-08-24 | 2012-05-31 | 이 아이 듀폰 디 네모아 앤드 캄파니 | Organic light-emitting diode luminaires |
| CN102484219A (en) | 2009-08-24 | 2012-05-30 | E.I.内穆尔杜邦公司 | Organic light-emitting diode lamp |
| JP5715142B2 (en) | 2009-09-29 | 2015-05-07 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニーE.I.Du Pont De Nemours And Company | Deuterium compounds for luminescence applications |
| CN102596893A (en) | 2009-10-19 | 2012-07-18 | E.I.内穆尔杜邦公司 | Triarylamine compounds for electronic applications |
| KR101782660B1 (en) | 2009-10-19 | 2017-09-27 | 이 아이 듀폰 디 네모아 앤드 캄파니 | Triarylamine compounds for electronic applications |
| US8674343B2 (en) | 2009-10-29 | 2014-03-18 | E I Du Pont De Nemours And Company | Organic light-emitting diodes having white light emission |
| US8716699B2 (en) | 2009-10-29 | 2014-05-06 | E I Du Pont De Nemours And Company | Organic light-emitting diodes having white light emission |
| US20110101312A1 (en) | 2009-10-29 | 2011-05-05 | E. I. Du Pont De Nemours And Company | Deuterated compounds for electronic applications |
| WO2011059789A2 (en) | 2009-10-29 | 2011-05-19 | E. I. Du Pont De Nemours And Company | Organic light-emitting diode luminaires |
| US8716700B2 (en) | 2009-10-29 | 2014-05-06 | E I Du Pont De Nemours And Company | Organic light-emitting diodes having white light emission |
| US8580394B2 (en) | 2009-11-19 | 2013-11-12 | Universal Display Corporation | 3-coordinate copper(I)-carbene complexes |
| US8617720B2 (en) | 2009-12-21 | 2013-12-31 | E I Du Pont De Nemours And Company | Electroactive composition and electronic device made with the composition |
| US8288187B2 (en) | 2010-01-20 | 2012-10-16 | Universal Display Corporation | Electroluminescent devices for lighting applications |
| US9156870B2 (en) | 2010-02-25 | 2015-10-13 | Universal Display Corporation | Phosphorescent emitters |
| US9175211B2 (en) | 2010-03-03 | 2015-11-03 | Universal Display Corporation | Phosphorescent materials |
| EP2550690B1 (en) | 2010-03-25 | 2018-12-26 | Universal Display Corporation | Solution processable doped triarylamine hole injection materials |
| US8968887B2 (en) | 2010-04-28 | 2015-03-03 | Universal Display Corporation | Triphenylene-benzofuran/benzothiophene/benzoselenophene compounds with substituents joining to form fused rings |
| US9040962B2 (en) | 2010-04-28 | 2015-05-26 | Universal Display Corporation | Depositing premixed materials |
| US8742657B2 (en) | 2010-06-11 | 2014-06-03 | Universal Display Corporation | Triplet-Triplet annihilation up conversion (TTA-UC) for display and lighting applications |
| US8673458B2 (en) | 2010-06-11 | 2014-03-18 | Universal Display Corporation | Delayed fluorescence OLED |
| TW201200975A (en) | 2010-06-17 | 2012-01-01 | Du Pont | Process and materials for making contained layers and devices made with same |
| TW201204687A (en) | 2010-06-17 | 2012-02-01 | Du Pont | Electroactive materials |
| JP6069708B2 (en) | 2010-07-02 | 2017-02-01 | 日産化学工業株式会社 | Hole transport composition and related devices and methods (I) |
| US9435021B2 (en) | 2010-07-29 | 2016-09-06 | University Of Southern California | Co-deposition methods for the fabrication of organic optoelectronic devices |
| CN103124711A (en) | 2010-08-11 | 2013-05-29 | E·I·内穆尔杜邦公司 | Electroactive compound and composition and electronic device made with the composition |
| WO2012023947A1 (en) | 2010-08-20 | 2012-02-23 | Universal Display Corporation | Bicarbazole compounds for oleds |
| JP5886858B2 (en) | 2010-08-24 | 2016-03-16 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニーE.I.Du Pont De Nemours And Company | PHOTOACTIVE COMPOSITION AND ELECTRONIC DEVICE PRODUCED BY USING THE COMPOSITION |
| US20120049168A1 (en) | 2010-08-31 | 2012-03-01 | Universal Display Corporation | Cross-Linked Charge Transport Layer Containing an Additive Compound |
| US8932734B2 (en) | 2010-10-08 | 2015-01-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US20120138906A1 (en) | 2010-12-07 | 2012-06-07 | The University of Southern California USC Stevens Institute for Innovation | Capture agents for unsaturated metal complexes |
| TW201229010A (en) | 2010-12-13 | 2012-07-16 | Du Pont | Electroactive materials |
| EP2651942B1 (en) | 2010-12-15 | 2016-03-16 | E. I. du Pont de Nemours and Company | Electroactive 1,7- and 4,10-diazachrysene derivatives and devices made with such materials |
| KR102158326B1 (en) | 2010-12-20 | 2020-09-21 | 주식회사 엘지화학 | Electroactive compositions for electronic applications |
| US20130248848A1 (en) | 2010-12-20 | 2013-09-26 | E.I. Du Pont Nemours And Company | Process and materials for making contained layers and devices made with same |
| WO2012087960A1 (en) | 2010-12-20 | 2012-06-28 | E. I. Du Pont De Nemours And Company | Triazine derivatives for electronic applications |
| WO2012087955A1 (en) | 2010-12-20 | 2012-06-28 | E. I. Du Pont De Nemours And Company | Compositions for electronic applications |
| JP2014504021A (en) | 2010-12-21 | 2014-02-13 | イー・アイ・デュポン・ドウ・ヌムール・アンド・カンパニー | Liquid compositions for the deposition of organic electroactive materials |
| CN103261172B (en) | 2010-12-21 | 2016-05-04 | E.I.内穆尔杜邦公司 | Comprise the electronic installation of pyrimidine compound |
| US10008677B2 (en) | 2011-01-13 | 2018-06-26 | Universal Display Corporation | Materials for organic light emitting diode |
| US9005772B2 (en) | 2011-02-23 | 2015-04-14 | Universal Display Corporation | Thioazole and oxazole carbene metal complexes as phosphorescent OLED materials |
| US8563737B2 (en) | 2011-02-23 | 2013-10-22 | Universal Display Corporation | Methods of making bis-tridentate carbene complexes of ruthenium and osmium |
| TWI560191B (en) | 2011-02-23 | 2016-12-01 | Universal Display Corp | Novel tetradentate platinum complexes |
| US8883322B2 (en) | 2011-03-08 | 2014-11-11 | Universal Display Corporation | Pyridyl carbene phosphorescent emitters |
| TW201245408A (en) | 2011-04-08 | 2012-11-16 | Du Pont | Electronic device |
| US8927308B2 (en) | 2011-05-12 | 2015-01-06 | Universal Display Corporation | Method of forming bus line designs for large-area OLED lighting |
| US8432095B2 (en) | 2011-05-11 | 2013-04-30 | Universal Display Corporation | Process for fabricating metal bus lines for OLED lighting panels |
| US9212197B2 (en) | 2011-05-19 | 2015-12-15 | Universal Display Corporation | Phosphorescent heteroleptic phenylbenzimidazole dopants |
| US8795850B2 (en) | 2011-05-19 | 2014-08-05 | Universal Display Corporation | Phosphorescent heteroleptic phenylbenzimidazole dopants and new synthetic methodology |
| US8748012B2 (en) | 2011-05-25 | 2014-06-10 | Universal Display Corporation | Host materials for OLED |
| US10158089B2 (en) | 2011-05-27 | 2018-12-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10079349B2 (en) | 2011-05-27 | 2018-09-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| KR102119353B1 (en) | 2011-06-08 | 2020-06-29 | 유니버셜 디스플레이 코포레이션 | Heteroleptic iridium carbene complexes and light emitting device using them |
| US8659036B2 (en) | 2011-06-17 | 2014-02-25 | Universal Display Corporation | Fine tuning of emission spectra by combination of multiple emitter spectra |
| US8884316B2 (en) | 2011-06-17 | 2014-11-11 | Universal Display Corporation | Non-common capping layer on an organic device |
| US9397310B2 (en) | 2011-07-14 | 2016-07-19 | Universal Display Corporation | Organice electroluminescent materials and devices |
| KR101965014B1 (en) | 2011-07-14 | 2019-04-02 | 유니버셜 디스플레이 코포레이션 | Inorganic hosts in oleds |
| US9023420B2 (en) | 2011-07-14 | 2015-05-05 | Universal Display Corporation | Composite organic/inorganic layer for organic light-emitting devices |
| US9783564B2 (en) | 2011-07-25 | 2017-10-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US8926119B2 (en) | 2011-08-04 | 2015-01-06 | Universal Display Corporation | Extendable light source with variable light emitting area |
| US9493698B2 (en) | 2011-08-31 | 2016-11-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| CN103875092A (en) | 2011-10-19 | 2014-06-18 | E.I.内穆尔杜邦公司 | Organic electronic device for lighting |
| US8652656B2 (en) | 2011-11-14 | 2014-02-18 | Universal Display Corporation | Triphenylene silane hosts |
| US9193745B2 (en) | 2011-11-15 | 2015-11-24 | Universal Display Corporation | Heteroleptic iridium complex |
| US9217004B2 (en) | 2011-11-21 | 2015-12-22 | Universal Display Corporation | Organic light emitting materials |
| US9512355B2 (en) | 2011-12-09 | 2016-12-06 | Universal Display Corporation | Organic light emitting materials |
| US20130146875A1 (en) | 2011-12-13 | 2013-06-13 | Universal Display Corporation | Split electrode for organic devices |
| US8987451B2 (en) | 2012-01-03 | 2015-03-24 | Universal Display Corporation | Synthesis of cyclometallated platinum(II) complexes |
| US9461254B2 (en) | 2012-01-03 | 2016-10-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9163174B2 (en) | 2012-01-04 | 2015-10-20 | Universal Display Corporation | Highly efficient phosphorescent materials |
| US8969592B2 (en) | 2012-01-10 | 2015-03-03 | Universal Display Corporation | Heterocyclic host materials |
| US10211413B2 (en) | 2012-01-17 | 2019-02-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9118017B2 (en) | 2012-02-27 | 2015-08-25 | Universal Display Corporation | Host compounds for red phosphorescent OLEDs |
| US9054323B2 (en) | 2012-03-15 | 2015-06-09 | Universal Display Corporation | Secondary hole transporting layer with diarylamino-phenyl-carbazole compounds |
| US9386657B2 (en) | 2012-03-15 | 2016-07-05 | Universal Display Corporation | Organic Electroluminescent materials and devices |
| WO2013142633A1 (en) | 2012-03-23 | 2013-09-26 | E. I. Du Pont De Nemours And Company | Green luminescent materials |
| US9593085B2 (en) | 2012-04-02 | 2017-03-14 | E I Du Pont De Nemours And Company | Blue luminescent compounds |
| WO2013163019A1 (en) | 2012-04-23 | 2013-10-31 | E. I. Du Pont De Nemours And Company | Blue luminescent compounds |
| US9184399B2 (en) | 2012-05-04 | 2015-11-10 | Universal Display Corporation | Asymmetric hosts with triaryl silane side chains |
| US9112157B2 (en) | 2012-05-15 | 2015-08-18 | Solvay Usa, Inc. | Hole transport materials including OLED applications |
| US9773985B2 (en) | 2012-05-21 | 2017-09-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9062993B2 (en) | 2012-05-22 | 2015-06-23 | E I Du Pont De Nemours And Company | Method and apparatus for liquid flow calibration check |
| CN104487831B (en) * | 2012-05-29 | 2016-09-07 | 3M创新有限公司 | Humidity sensor and sensor element |
| US9670404B2 (en) | 2012-06-06 | 2017-06-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9502672B2 (en) | 2012-06-21 | 2016-11-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9725476B2 (en) | 2012-07-09 | 2017-08-08 | Universal Display Corporation | Silylated metal complexes |
| US9231218B2 (en) | 2012-07-10 | 2016-01-05 | Universal Display Corporation | Phosphorescent emitters containing dibenzo[1,4]azaborinine structure |
| US9540329B2 (en) | 2012-07-19 | 2017-01-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9059412B2 (en) | 2012-07-19 | 2015-06-16 | Universal Display Corporation | Transition metal complexes containing substituted imidazole carbene as ligands and their application in OLEDs |
| US9663544B2 (en) | 2012-07-25 | 2017-05-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9318710B2 (en) | 2012-07-30 | 2016-04-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| EP2890221A4 (en) | 2012-08-24 | 2016-09-14 | Konica Minolta Inc | TRANSPARENT ELECTRODE, ELECTRONIC DEVICE, AND METHOD FOR MANUFACTURING TRANSPARENT ELECTRODE |
| US9978958B2 (en) | 2012-08-24 | 2018-05-22 | Universal Display Corporation | Phosphorescent emitters with phenylimidazole ligands |
| US8952362B2 (en) | 2012-08-31 | 2015-02-10 | The Regents Of The University Of Michigan | High efficiency and brightness fluorescent organic light emitting diode by triplet-triplet fusion |
| US10957870B2 (en) | 2012-09-07 | 2021-03-23 | Universal Display Corporation | Organic light emitting device |
| US9287513B2 (en) | 2012-09-24 | 2016-03-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9312505B2 (en) | 2012-09-25 | 2016-04-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9252363B2 (en) | 2012-10-04 | 2016-02-02 | Universal Display Corporation | Aryloxyalkylcarboxylate solvent compositions for inkjet printing of organic layers |
| US8692241B1 (en) | 2012-11-08 | 2014-04-08 | Universal Display Corporation | Transition metal complexes containing triazole and tetrazole carbene ligands |
| US9634264B2 (en) | 2012-11-09 | 2017-04-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US8946697B1 (en) | 2012-11-09 | 2015-02-03 | Universal Display Corporation | Iridium complexes with aza-benzo fused ligands |
| US9748500B2 (en) | 2015-01-15 | 2017-08-29 | Universal Display Corporation | Organic light emitting materials |
| US9685617B2 (en) | 2012-11-09 | 2017-06-20 | Universal Display Corporation | Organic electronuminescent materials and devices |
| US9190623B2 (en) | 2012-11-20 | 2015-11-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10069090B2 (en) | 2012-11-20 | 2018-09-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9512136B2 (en) | 2012-11-26 | 2016-12-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9166175B2 (en) | 2012-11-27 | 2015-10-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9196860B2 (en) | 2012-12-04 | 2015-11-24 | Universal Display Corporation | Compounds for triplet-triplet annihilation upconversion |
| US8716484B1 (en) | 2012-12-05 | 2014-05-06 | Universal Display Corporation | Hole transporting materials with twisted aryl groups |
| US9209411B2 (en) | 2012-12-07 | 2015-12-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9653691B2 (en) | 2012-12-12 | 2017-05-16 | Universal Display Corporation | Phosphorescence-sensitizing fluorescence material system |
| CN104838513B (en) | 2012-12-13 | 2018-03-13 | E.I.内穆尔杜邦公司 | For preparing the method and material and the equipment that is made from it of included layer |
| US9986742B2 (en) | 2012-12-20 | 2018-06-05 | Quick-Med Technologies, Inc. | Durable antimicrobial treatments for textiles and other substrates |
| PL2945653T3 (en) | 2012-12-20 | 2017-11-30 | Quick-Med Technologies, Inc. | Regeneration of antimicrobial coatings containing metal derivatives when exposed to aqueous hydrogen peroxide |
| US10400163B2 (en) | 2013-02-08 | 2019-09-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10367154B2 (en) | 2013-02-21 | 2019-07-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9748497B2 (en) | 2013-02-25 | 2017-08-29 | E I Du Pont De Nemours And Company | Electronic device including a diazachrysene derivative |
| US8927749B2 (en) | 2013-03-07 | 2015-01-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9419225B2 (en) | 2013-03-14 | 2016-08-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9997712B2 (en) | 2013-03-27 | 2018-06-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10135002B2 (en) | 2013-03-29 | 2018-11-20 | Konica Minolta, Inc. | Organic electroluminescent element, and lighting device and display device which are provided with same |
| JP6269655B2 (en) | 2013-03-29 | 2018-01-31 | コニカミノルタ株式会社 | ORGANIC ELECTROLUMINESCENT ELEMENT MATERIAL, ORGANIC ELECTROLUMINESCENT ELEMENT, DISPLAY DEVICE AND LIGHTING DEVICE |
| EP2980876B1 (en) | 2013-03-29 | 2019-05-08 | Konica Minolta, Inc. | Organic electroluminescent element, lighting device and display device |
| US9537106B2 (en) | 2013-05-09 | 2017-01-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9735373B2 (en) | 2013-06-10 | 2017-08-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9673401B2 (en) | 2013-06-28 | 2017-06-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10199581B2 (en) | 2013-07-01 | 2019-02-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10121975B2 (en) | 2013-07-03 | 2018-11-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9761807B2 (en) | 2013-07-15 | 2017-09-12 | Universal Display Corporation | Organic light emitting diode materials |
| US9553274B2 (en) | 2013-07-16 | 2017-01-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9324949B2 (en) | 2013-07-16 | 2016-04-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9224958B2 (en) | 2013-07-19 | 2015-12-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US20150028290A1 (en) | 2013-07-25 | 2015-01-29 | Universal Display Corporation | Heteroleptic osmium complex and method of making the same |
| US10074806B2 (en) | 2013-08-20 | 2018-09-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9831437B2 (en) | 2013-08-20 | 2017-11-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9932359B2 (en) | 2013-08-30 | 2018-04-03 | University Of Southern California | Organic electroluminescent materials and devices |
| US10199582B2 (en) | 2013-09-03 | 2019-02-05 | University Of Southern California | Organic electroluminescent materials and devices |
| US9735378B2 (en) | 2013-09-09 | 2017-08-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9748503B2 (en) | 2013-09-13 | 2017-08-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10003034B2 (en) | 2013-09-30 | 2018-06-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| KR101457304B1 (en) * | 2013-10-07 | 2014-11-03 | 인하대학교 산학협력단 | Method of preparing high dielectric insulating material and insulating device using the same |
| US9831447B2 (en) | 2013-10-08 | 2017-11-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9293712B2 (en) | 2013-10-11 | 2016-03-22 | Universal Display Corporation | Disubstituted pyrene compounds with amino group containing ortho aryl group and devices containing the same |
| US9853229B2 (en) | 2013-10-23 | 2017-12-26 | University Of Southern California | Organic electroluminescent materials and devices |
| US20150115250A1 (en) | 2013-10-29 | 2015-04-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9306179B2 (en) | 2013-11-08 | 2016-04-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9647218B2 (en) | 2013-11-14 | 2017-05-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9905784B2 (en) | 2013-11-15 | 2018-02-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10056565B2 (en) | 2013-11-20 | 2018-08-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| FR3013719B1 (en) * | 2013-11-26 | 2018-01-12 | Commissariat A L'energie Atomique Et Aux Energies Alternatives | INK FOR FORMING P-LAYERS IN ORGANIC ELECTRONIC DEVICES |
| US10644251B2 (en) | 2013-12-04 | 2020-05-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9876173B2 (en) | 2013-12-09 | 2018-01-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10355227B2 (en) | 2013-12-16 | 2019-07-16 | Universal Display Corporation | Metal complex for phosphorescent OLED |
| US9847496B2 (en) | 2013-12-23 | 2017-12-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10135008B2 (en) | 2014-01-07 | 2018-11-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9978961B2 (en) | 2014-01-08 | 2018-05-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9755159B2 (en) | 2014-01-23 | 2017-09-05 | Universal Display Corporation | Organic materials for OLEDs |
| US10586933B2 (en) | 2014-01-23 | 2020-03-10 | Lg Chem, Ltd. | Electroactive metal complexes |
| US9935277B2 (en) | 2014-01-30 | 2018-04-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9590194B2 (en) | 2014-02-14 | 2017-03-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9847497B2 (en) | 2014-02-18 | 2017-12-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10003033B2 (en) | 2014-02-18 | 2018-06-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10707423B2 (en) | 2014-02-21 | 2020-07-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9647217B2 (en) | 2014-02-24 | 2017-05-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9502656B2 (en) | 2014-02-24 | 2016-11-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10403825B2 (en) | 2014-02-27 | 2019-09-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9590195B2 (en) | 2014-02-28 | 2017-03-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9673407B2 (en) | 2014-02-28 | 2017-06-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9181270B2 (en) | 2014-02-28 | 2015-11-10 | Universal Display Corporation | Method of making sulfide compounds |
| US9190620B2 (en) | 2014-03-01 | 2015-11-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9397309B2 (en) | 2014-03-13 | 2016-07-19 | Universal Display Corporation | Organic electroluminescent devices |
| US10208026B2 (en) | 2014-03-18 | 2019-02-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9748504B2 (en) | 2014-03-25 | 2017-08-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9929353B2 (en) | 2014-04-02 | 2018-03-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9691993B2 (en) | 2014-04-09 | 2017-06-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9905785B2 (en) | 2014-04-14 | 2018-02-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10008679B2 (en) | 2014-04-14 | 2018-06-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9450198B2 (en) | 2014-04-15 | 2016-09-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10256427B2 (en) | 2014-04-15 | 2019-04-09 | Universal Display Corporation | Efficient organic electroluminescent devices |
| US9741941B2 (en) | 2014-04-29 | 2017-08-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10457699B2 (en) | 2014-05-02 | 2019-10-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10403830B2 (en) | 2014-05-08 | 2019-09-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10636983B2 (en) | 2014-05-08 | 2020-04-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| WO2015171627A1 (en) | 2014-05-08 | 2015-11-12 | Universal Display Corporation | Stabilized imidazophenanthridine materials |
| US10301338B2 (en) | 2014-05-08 | 2019-05-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9997716B2 (en) | 2014-05-27 | 2018-06-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10461260B2 (en) | 2014-06-03 | 2019-10-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| CN104021838B (en) * | 2014-06-20 | 2016-09-07 | 上海赤龙科技有限公司 | A kind of polythiophene/mixed valence metal oxide works in coordination with electrocondution slurry and preparation method thereof |
| US9911931B2 (en) | 2014-06-26 | 2018-03-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10297762B2 (en) | 2014-07-09 | 2019-05-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10566546B2 (en) | 2014-07-14 | 2020-02-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9929357B2 (en) | 2014-07-22 | 2018-03-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11108000B2 (en) | 2014-08-07 | 2021-08-31 | Unniversal Display Corporation | Organic electroluminescent materials and devices |
| US10411200B2 (en) | 2014-08-07 | 2019-09-10 | Universal Display Corporation | Electroluminescent (2-phenylpyridine)iridium complexes and devices |
| US9843001B2 (en) | 2014-08-18 | 2017-12-12 | E I Du Pont De Nemours And Company | Blue luminescent compounds |
| US9944846B2 (en) | 2014-08-28 | 2018-04-17 | E I Du Pont De Nemours And Company | Compositions for electronic applications |
| US10135007B2 (en) | 2014-09-29 | 2018-11-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10749113B2 (en) | 2014-09-29 | 2020-08-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10361375B2 (en) | 2014-10-06 | 2019-07-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9397302B2 (en) | 2014-10-08 | 2016-07-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10854826B2 (en) | 2014-10-08 | 2020-12-01 | Universal Display Corporation | Organic electroluminescent compounds, compositions and devices |
| US10950803B2 (en) | 2014-10-13 | 2021-03-16 | Universal Display Corporation | Compounds and uses in devices |
| US10544123B2 (en) | 2014-10-20 | 2020-01-28 | Lg Chem, Ltd. | Blue luminescent compounds |
| US9484541B2 (en) | 2014-10-20 | 2016-11-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| WO2016069321A2 (en) | 2014-10-31 | 2016-05-06 | E. I. Du Pont De Nemours And Company | Electroactive materials |
| US10868261B2 (en) | 2014-11-10 | 2020-12-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10038151B2 (en) | 2014-11-12 | 2018-07-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10411201B2 (en) | 2014-11-12 | 2019-09-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9882151B2 (en) | 2014-11-14 | 2018-01-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9761814B2 (en) | 2014-11-18 | 2017-09-12 | Universal Display Corporation | Organic light-emitting materials and devices |
| CN107108861B (en) | 2014-11-20 | 2019-11-12 | 株式会社Lg化学 | hole transport material |
| US10381569B2 (en) | 2014-11-25 | 2019-08-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9444075B2 (en) | 2014-11-26 | 2016-09-13 | Universal Display Corporation | Emissive display with photo-switchable polarization |
| US9450195B2 (en) | 2014-12-17 | 2016-09-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10253252B2 (en) | 2014-12-30 | 2019-04-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10636978B2 (en) | 2014-12-30 | 2020-04-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9312499B1 (en) | 2015-01-05 | 2016-04-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9406892B2 (en) | 2015-01-07 | 2016-08-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10418569B2 (en) | 2015-01-25 | 2019-09-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9711730B2 (en) | 2015-01-25 | 2017-07-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10355222B2 (en) | 2015-02-06 | 2019-07-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10418562B2 (en) | 2015-02-06 | 2019-09-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10644247B2 (en) | 2015-02-06 | 2020-05-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10177316B2 (en) | 2015-02-09 | 2019-01-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10144867B2 (en) | 2015-02-13 | 2018-12-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| JP5831654B1 (en) | 2015-02-13 | 2015-12-09 | コニカミノルタ株式会社 | Aromatic heterocycle derivative, organic electroluminescence device using the same, illumination device and display device |
| US10680183B2 (en) | 2015-02-15 | 2020-06-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9929361B2 (en) | 2015-02-16 | 2018-03-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11056657B2 (en) | 2015-02-27 | 2021-07-06 | University Display Corporation | Organic electroluminescent materials and devices |
| US10600966B2 (en) | 2015-02-27 | 2020-03-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10686143B2 (en) | 2015-03-05 | 2020-06-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10270046B2 (en) | 2015-03-06 | 2019-04-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9780316B2 (en) | 2015-03-16 | 2017-10-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9911928B2 (en) | 2015-03-19 | 2018-03-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| JP6294254B2 (en) | 2015-03-23 | 2018-03-14 | 信越化学工業株式会社 | Conductive material and substrate |
| US9871214B2 (en) | 2015-03-23 | 2018-01-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10529931B2 (en) | 2015-03-24 | 2020-01-07 | Universal Display Corporation | Organic Electroluminescent materials and devices |
| US9972783B2 (en) | 2015-03-25 | 2018-05-15 | E I Du Pont De Nemours And Company | High energy triarylamine compounds for hole transport materials |
| US10297770B2 (en) | 2015-03-27 | 2019-05-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11818949B2 (en) | 2015-04-06 | 2023-11-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11495749B2 (en) | 2015-04-06 | 2022-11-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US20160293854A1 (en) | 2015-04-06 | 2016-10-06 | Universal Display Corporation | Organic Electroluminescent Materials and Devices |
| US9954174B2 (en) | 2015-05-06 | 2018-04-24 | E I Du Pont De Nemours And Company | Hole transport materials |
| US10403826B2 (en) | 2015-05-07 | 2019-09-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10777749B2 (en) | 2015-05-07 | 2020-09-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9478758B1 (en) | 2015-05-08 | 2016-10-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9859510B2 (en) | 2015-05-15 | 2018-01-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10109799B2 (en) | 2015-05-21 | 2018-10-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10804473B2 (en) | 2015-05-21 | 2020-10-13 | Lg Chem, Ltd. | Electron transport materials for electronic applications |
| US10256411B2 (en) | 2015-05-21 | 2019-04-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10033004B2 (en) | 2015-06-01 | 2018-07-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10418568B2 (en) | 2015-06-01 | 2019-09-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11925102B2 (en) | 2015-06-04 | 2024-03-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10818853B2 (en) | 2015-06-04 | 2020-10-27 | University Of Southern California | Organic electroluminescent materials and devices |
| US10825997B2 (en) | 2015-06-25 | 2020-11-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10873036B2 (en) | 2015-07-07 | 2020-12-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9978956B2 (en) | 2015-07-15 | 2018-05-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10700284B2 (en) | 2015-07-20 | 2020-06-30 | Lg Chem, Ltd. | Photoactive composition |
| US11127905B2 (en) | 2015-07-29 | 2021-09-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11018309B2 (en) | 2015-08-03 | 2021-05-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11522140B2 (en) | 2015-08-17 | 2022-12-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10522769B2 (en) | 2015-08-18 | 2019-12-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10181564B2 (en) | 2015-08-26 | 2019-01-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10672996B2 (en) | 2015-09-03 | 2020-06-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11706972B2 (en) | 2015-09-08 | 2023-07-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11302872B2 (en) | 2015-09-09 | 2022-04-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10770664B2 (en) | 2015-09-21 | 2020-09-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US20170092880A1 (en) | 2015-09-25 | 2017-03-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10847728B2 (en) | 2015-10-01 | 2020-11-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10593892B2 (en) | 2015-10-01 | 2020-03-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10991895B2 (en) | 2015-10-06 | 2021-04-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10388893B2 (en) | 2015-10-29 | 2019-08-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10388892B2 (en) | 2015-10-29 | 2019-08-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10177318B2 (en) | 2015-10-29 | 2019-01-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10998507B2 (en) | 2015-11-23 | 2021-05-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10476010B2 (en) | 2015-11-30 | 2019-11-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10957861B2 (en) | 2015-12-29 | 2021-03-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11024808B2 (en) | 2015-12-29 | 2021-06-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10135006B2 (en) | 2016-01-04 | 2018-11-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| JP6788314B2 (en) | 2016-01-06 | 2020-11-25 | コニカミノルタ株式会社 | Organic electroluminescence element, manufacturing method of organic electroluminescence element, display device and lighting device |
| US10457864B2 (en) | 2016-02-09 | 2019-10-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10707427B2 (en) | 2016-02-09 | 2020-07-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10600967B2 (en) | 2016-02-18 | 2020-03-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11094891B2 (en) | 2016-03-16 | 2021-08-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10276809B2 (en) | 2016-04-05 | 2019-04-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10236456B2 (en) | 2016-04-11 | 2019-03-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10566552B2 (en) | 2016-04-13 | 2020-02-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11081647B2 (en) | 2016-04-22 | 2021-08-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11228002B2 (en) | 2016-04-22 | 2022-01-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11228003B2 (en) | 2016-04-22 | 2022-01-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US20170324049A1 (en) | 2016-05-05 | 2017-11-09 | Universal Display Corporation | Organic Electroluminescent Materials and Devices |
| US10840458B2 (en) | 2016-05-25 | 2020-11-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9966542B2 (en) | 2016-06-02 | 2018-05-08 | E I Du Pont De Nemours And Company | Electroactive materials |
| US10468609B2 (en) | 2016-06-02 | 2019-11-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| CN109153684B (en) | 2016-06-03 | 2021-12-21 | 株式会社Lg化学 | Electroactive compounds |
| US10727423B2 (en) | 2016-06-20 | 2020-07-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10862054B2 (en) | 2016-06-20 | 2020-12-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10651403B2 (en) | 2016-06-20 | 2020-05-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10686140B2 (en) | 2016-06-20 | 2020-06-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11482683B2 (en) | 2016-06-20 | 2022-10-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10672997B2 (en) | 2016-06-20 | 2020-06-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10957866B2 (en) | 2016-06-30 | 2021-03-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9929360B2 (en) | 2016-07-08 | 2018-03-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10680184B2 (en) | 2016-07-11 | 2020-06-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10720587B2 (en) | 2016-07-19 | 2020-07-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10153443B2 (en) | 2016-07-19 | 2018-12-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11114621B2 (en) | 2016-07-20 | 2021-09-07 | Lg Chem, Ltd. | Electroactive materials |
| US10205105B2 (en) | 2016-08-15 | 2019-02-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10608186B2 (en) | 2016-09-14 | 2020-03-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10505127B2 (en) | 2016-09-19 | 2019-12-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10680187B2 (en) | 2016-09-23 | 2020-06-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11189804B2 (en) | 2016-10-03 | 2021-11-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11183642B2 (en) | 2016-10-03 | 2021-11-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11127906B2 (en) | 2016-10-03 | 2021-09-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11196010B2 (en) | 2016-10-03 | 2021-12-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11081658B2 (en) | 2016-10-03 | 2021-08-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11011709B2 (en) | 2016-10-07 | 2021-05-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11239432B2 (en) | 2016-10-14 | 2022-02-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10608185B2 (en) | 2016-10-17 | 2020-03-31 | Univeral Display Corporation | Organic electroluminescent materials and devices |
| US10236458B2 (en) | 2016-10-24 | 2019-03-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12317745B2 (en) | 2016-11-09 | 2025-05-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10340464B2 (en) | 2016-11-10 | 2019-07-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10680188B2 (en) | 2016-11-11 | 2020-06-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10897016B2 (en) | 2016-11-14 | 2021-01-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10964893B2 (en) | 2016-11-17 | 2021-03-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10662196B2 (en) | 2016-11-17 | 2020-05-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10833276B2 (en) | 2016-11-21 | 2020-11-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10153445B2 (en) | 2016-11-21 | 2018-12-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11239426B2 (en) | 2016-11-23 | 2022-02-01 | Lg Chem, Ltd. | Electroactive compounds |
| US11555048B2 (en) | 2016-12-01 | 2023-01-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10490753B2 (en) | 2016-12-15 | 2019-11-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11548905B2 (en) | 2016-12-15 | 2023-01-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11545636B2 (en) | 2016-12-15 | 2023-01-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10811618B2 (en) | 2016-12-19 | 2020-10-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11152579B2 (en) | 2016-12-28 | 2021-10-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US20180190915A1 (en) | 2017-01-03 | 2018-07-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11780865B2 (en) | 2017-01-09 | 2023-10-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11201298B2 (en) | 2017-01-09 | 2021-12-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10804475B2 (en) | 2017-01-11 | 2020-10-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11545637B2 (en) | 2017-01-13 | 2023-01-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10629820B2 (en) | 2017-01-18 | 2020-04-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11053268B2 (en) | 2017-01-20 | 2021-07-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10964904B2 (en) | 2017-01-20 | 2021-03-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11765968B2 (en) | 2017-01-23 | 2023-09-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11050028B2 (en) | 2017-01-24 | 2021-06-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12089486B2 (en) | 2017-02-08 | 2024-09-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10978647B2 (en) | 2017-02-15 | 2021-04-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10844084B2 (en) | 2017-02-22 | 2020-11-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10745431B2 (en) | 2017-03-08 | 2020-08-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10741780B2 (en) | 2017-03-10 | 2020-08-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10672998B2 (en) | 2017-03-23 | 2020-06-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10873037B2 (en) | 2017-03-28 | 2020-12-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10910577B2 (en) | 2017-03-28 | 2021-02-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10844085B2 (en) | 2017-03-29 | 2020-11-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11158820B2 (en) | 2017-03-29 | 2021-10-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11056658B2 (en) | 2017-03-29 | 2021-07-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10862046B2 (en) | 2017-03-30 | 2020-12-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11276829B2 (en) | 2017-03-31 | 2022-03-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11139443B2 (en) | 2017-03-31 | 2021-10-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11038117B2 (en) | 2017-04-11 | 2021-06-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10777754B2 (en) | 2017-04-11 | 2020-09-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11101434B2 (en) | 2017-04-21 | 2021-08-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10975113B2 (en) | 2017-04-21 | 2021-04-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11084838B2 (en) | 2017-04-21 | 2021-08-10 | Universal Display Corporation | Organic electroluminescent materials and device |
| US10910570B2 (en) | 2017-04-28 | 2021-02-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11038137B2 (en) | 2017-04-28 | 2021-06-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11117897B2 (en) | 2017-05-01 | 2021-09-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10941170B2 (en) | 2017-05-03 | 2021-03-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11201299B2 (en) | 2017-05-04 | 2021-12-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10862055B2 (en) | 2017-05-05 | 2020-12-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10870668B2 (en) | 2017-05-05 | 2020-12-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10930864B2 (en) | 2017-05-10 | 2021-02-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10944060B2 (en) | 2017-05-11 | 2021-03-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10822362B2 (en) | 2017-05-11 | 2020-11-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10790455B2 (en) | 2017-05-18 | 2020-09-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10840459B2 (en) | 2017-05-18 | 2020-11-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10944062B2 (en) | 2017-05-18 | 2021-03-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11038115B2 (en) | 2017-05-18 | 2021-06-15 | Universal Display Corporation | Organic electroluminescent materials and device |
| US10934293B2 (en) | 2017-05-18 | 2021-03-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10930862B2 (en) | 2017-06-01 | 2021-02-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11678565B2 (en) | 2017-06-23 | 2023-06-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11758804B2 (en) | 2017-06-23 | 2023-09-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10968226B2 (en) | 2017-06-23 | 2021-04-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11495757B2 (en) | 2017-06-23 | 2022-11-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11814403B2 (en) | 2017-06-23 | 2023-11-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11552261B2 (en) | 2017-06-23 | 2023-01-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11608321B2 (en) | 2017-06-23 | 2023-03-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11174259B2 (en) | 2017-06-23 | 2021-11-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11832510B2 (en) | 2017-06-23 | 2023-11-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11802136B2 (en) | 2017-06-23 | 2023-10-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12098157B2 (en) | 2017-06-23 | 2024-09-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11725022B2 (en) | 2017-06-23 | 2023-08-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11469382B2 (en) | 2017-07-12 | 2022-10-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11917843B2 (en) | 2017-07-26 | 2024-02-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11765970B2 (en) | 2017-07-26 | 2023-09-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11322691B2 (en) | 2017-07-26 | 2022-05-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11228010B2 (en) | 2017-07-26 | 2022-01-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11239433B2 (en) | 2017-07-26 | 2022-02-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11968883B2 (en) | 2017-07-26 | 2024-04-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11744141B2 (en) | 2017-08-09 | 2023-08-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11508913B2 (en) | 2017-08-10 | 2022-11-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11349083B2 (en) | 2017-08-10 | 2022-05-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11744142B2 (en) | 2017-08-10 | 2023-08-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11910699B2 (en) | 2017-08-10 | 2024-02-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11462697B2 (en) | 2017-08-22 | 2022-10-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11723269B2 (en) | 2017-08-22 | 2023-08-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11437591B2 (en) | 2017-08-24 | 2022-09-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11605791B2 (en) | 2017-09-01 | 2023-03-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11444249B2 (en) | 2017-09-07 | 2022-09-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11424420B2 (en) | 2017-09-07 | 2022-08-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11696492B2 (en) | 2017-09-07 | 2023-07-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10608188B2 (en) | 2017-09-11 | 2020-03-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11778897B2 (en) | 2017-09-20 | 2023-10-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12389791B2 (en) | 2017-09-21 | 2025-08-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12459946B2 (en) | 2017-10-05 | 2025-11-04 | Universal Display Corporation | Organic host materials for electroluminescent devices |
| US11183646B2 (en) | 2017-11-07 | 2021-11-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11910702B2 (en) | 2017-11-07 | 2024-02-20 | Universal Display Corporation | Organic electroluminescent devices |
| US11214587B2 (en) | 2017-11-07 | 2022-01-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11168103B2 (en) | 2017-11-17 | 2021-11-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11825735B2 (en) | 2017-11-28 | 2023-11-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11937503B2 (en) | 2017-11-30 | 2024-03-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11233204B2 (en) | 2017-12-14 | 2022-01-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10971687B2 (en) | 2017-12-14 | 2021-04-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12075690B2 (en) | 2017-12-14 | 2024-08-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11233205B2 (en) | 2017-12-14 | 2022-01-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11081659B2 (en) | 2018-01-10 | 2021-08-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11700765B2 (en) | 2018-01-10 | 2023-07-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11515493B2 (en) | 2018-01-11 | 2022-11-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11271177B2 (en) | 2018-01-11 | 2022-03-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11845764B2 (en) | 2018-01-26 | 2023-12-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11367840B2 (en) | 2018-01-26 | 2022-06-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11542289B2 (en) | 2018-01-26 | 2023-01-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12029055B2 (en) | 2018-01-30 | 2024-07-02 | The University Of Southern California | OLED with hybrid emissive layer |
| US11180519B2 (en) | 2018-02-09 | 2021-11-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11239434B2 (en) | 2018-02-09 | 2022-02-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11957050B2 (en) | 2018-02-09 | 2024-04-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11342509B2 (en) | 2018-02-09 | 2022-05-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11217757B2 (en) | 2018-03-12 | 2022-01-04 | Universal Display Corporation | Host materials for electroluminescent devices |
| US11557733B2 (en) | 2018-03-12 | 2023-01-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11142538B2 (en) | 2018-03-12 | 2021-10-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11165028B2 (en) | 2018-03-12 | 2021-11-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11279722B2 (en) | 2018-03-12 | 2022-03-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11882759B2 (en) | 2018-04-13 | 2024-01-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11390639B2 (en) | 2018-04-13 | 2022-07-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12522608B2 (en) | 2018-04-13 | 2026-01-13 | Universal Display Corporation | Host materials for electroluminescent devices |
| US11616203B2 (en) | 2018-04-17 | 2023-03-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11515494B2 (en) | 2018-05-04 | 2022-11-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11342513B2 (en) | 2018-05-04 | 2022-05-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11753427B2 (en) | 2018-05-04 | 2023-09-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11793073B2 (en) | 2018-05-06 | 2023-10-17 | Universal Display Corporation | Host materials for electroluminescent devices |
| US11450822B2 (en) | 2018-05-25 | 2022-09-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11459349B2 (en) | 2018-05-25 | 2022-10-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11716900B2 (en) | 2018-05-30 | 2023-08-01 | Universal Display Corporation | Host materials for electroluminescent devices |
| US11296283B2 (en) | 2018-06-04 | 2022-04-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11925103B2 (en) | 2018-06-05 | 2024-03-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11339182B2 (en) | 2018-06-07 | 2022-05-24 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11228004B2 (en) | 2018-06-22 | 2022-01-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11261207B2 (en) | 2018-06-25 | 2022-03-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11753425B2 (en) | 2018-07-11 | 2023-09-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12378197B2 (en) | 2018-07-13 | 2025-08-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12453279B2 (en) | 2018-08-22 | 2025-10-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11233203B2 (en) | 2018-09-06 | 2022-01-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12171137B2 (en) | 2018-09-10 | 2024-12-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11485706B2 (en) | 2018-09-11 | 2022-11-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11718634B2 (en) | 2018-09-14 | 2023-08-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11903305B2 (en) | 2018-09-24 | 2024-02-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11469383B2 (en) | 2018-10-08 | 2022-10-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11495752B2 (en) | 2018-10-08 | 2022-11-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11476430B2 (en) | 2018-10-15 | 2022-10-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11515482B2 (en) | 2018-10-23 | 2022-11-29 | Universal Display Corporation | Deep HOMO (highest occupied molecular orbital) emitter device structures |
| US11469384B2 (en) | 2018-11-02 | 2022-10-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11825736B2 (en) | 2018-11-19 | 2023-11-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11963441B2 (en) | 2018-11-26 | 2024-04-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11889708B2 (en) | 2019-11-14 | 2024-01-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11716899B2 (en) | 2018-11-28 | 2023-08-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11672165B2 (en) | 2018-11-28 | 2023-06-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11672176B2 (en) | 2018-11-28 | 2023-06-06 | Universal Display Corporation | Host materials for electroluminescent devices |
| US11690285B2 (en) | 2018-11-28 | 2023-06-27 | Universal Display Corporation | Electroluminescent devices |
| US11515489B2 (en) | 2018-11-28 | 2022-11-29 | Universal Display Corporation | Host materials for electroluminescent devices |
| US11623936B2 (en) | 2018-12-11 | 2023-04-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12281129B2 (en) | 2018-12-12 | 2025-04-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11737349B2 (en) | 2018-12-12 | 2023-08-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11834459B2 (en) | 2018-12-12 | 2023-12-05 | Universal Display Corporation | Host materials for electroluminescent devices |
| US12167673B2 (en) | 2018-12-19 | 2024-12-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11780829B2 (en) | 2019-01-30 | 2023-10-10 | The University Of Southern California | Organic electroluminescent materials and devices |
| US11812624B2 (en) | 2019-01-30 | 2023-11-07 | The University Of Southern California | Organic electroluminescent materials and devices |
| US12477890B2 (en) | 2019-02-01 | 2025-11-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11325932B2 (en) | 2019-02-08 | 2022-05-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12137605B2 (en) | 2019-02-08 | 2024-11-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11370809B2 (en) | 2019-02-08 | 2022-06-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11773320B2 (en) | 2019-02-21 | 2023-10-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11557738B2 (en) | 2019-02-22 | 2023-01-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12250872B2 (en) | 2019-02-22 | 2025-03-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11871653B2 (en) | 2019-02-22 | 2024-01-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11512093B2 (en) | 2019-03-04 | 2022-11-29 | Universal Display Corporation | Compound used for organic light emitting device (OLED), consumer product and formulation |
| US11739081B2 (en) | 2019-03-11 | 2023-08-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11569480B2 (en) | 2019-03-12 | 2023-01-31 | Universal Display Corporation | Plasmonic OLEDs and vertical dipole emitters |
| US11637261B2 (en) | 2019-03-12 | 2023-04-25 | Universal Display Corporation | Nanopatch antenna outcoupling structure for use in OLEDs |
| US11963438B2 (en) | 2019-03-26 | 2024-04-16 | The University Of Southern California | Organic electroluminescent materials and devices |
| US12122793B2 (en) | 2019-03-27 | 2024-10-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12240865B2 (en) | 2019-03-27 | 2025-03-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11639363B2 (en) | 2019-04-22 | 2023-05-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12075691B2 (en) | 2019-04-30 | 2024-08-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11613550B2 (en) | 2019-04-30 | 2023-03-28 | Universal Display Corporation | Organic electroluminescent materials and devices comprising benzimidazole-containing metal complexes |
| US11495756B2 (en) | 2019-05-07 | 2022-11-08 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11827651B2 (en) | 2019-05-13 | 2023-11-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12103942B2 (en) | 2019-05-13 | 2024-10-01 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11634445B2 (en) | 2019-05-21 | 2023-04-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12010859B2 (en) | 2019-05-24 | 2024-06-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11647667B2 (en) | 2019-06-14 | 2023-05-09 | Universal Display Corporation | Organic electroluminescent compounds and organic light emitting devices using the same |
| US12077550B2 (en) | 2019-07-02 | 2024-09-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11920070B2 (en) | 2019-07-12 | 2024-03-05 | The University Of Southern California | Luminescent janus-type, two-coordinated metal complexes |
| US11685754B2 (en) | 2019-07-22 | 2023-06-27 | Universal Display Corporation | Heteroleptic organic electroluminescent materials |
| US11926638B2 (en) | 2019-07-22 | 2024-03-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12281128B2 (en) | 2019-07-30 | 2025-04-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12295251B2 (en) | 2019-08-01 | 2025-05-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11708355B2 (en) | 2019-08-01 | 2023-07-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11985888B2 (en) | 2019-08-12 | 2024-05-14 | The Regents Of The University Of Michigan | Organic electroluminescent device |
| US11374181B2 (en) | 2019-08-14 | 2022-06-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12227525B2 (en) | 2019-08-14 | 2025-02-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11930699B2 (en) | 2019-08-15 | 2024-03-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12139501B2 (en) | 2019-08-16 | 2024-11-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12234249B2 (en) | 2019-08-21 | 2025-02-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11925105B2 (en) | 2019-08-26 | 2024-03-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11937494B2 (en) | 2019-08-28 | 2024-03-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11600787B2 (en) | 2019-08-30 | 2023-03-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11820783B2 (en) | 2019-09-06 | 2023-11-21 | Universal Display Corporation | Organic electroluminescent materials and devices |
| CN112457349B (en) | 2019-09-06 | 2024-10-15 | 环球展览公司 | Organic electroluminescent material and device |
| US12144244B2 (en) | 2019-09-10 | 2024-11-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11999886B2 (en) | 2019-09-26 | 2024-06-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12331065B2 (en) | 2019-09-26 | 2025-06-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12486296B2 (en) | 2019-10-02 | 2025-12-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11864458B2 (en) | 2019-10-08 | 2024-01-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11950493B2 (en) | 2019-10-15 | 2024-04-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11697653B2 (en) | 2019-10-21 | 2023-07-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11919914B2 (en) | 2019-10-25 | 2024-03-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12522565B2 (en) | 2019-10-25 | 2026-01-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| CN112707923B (en) | 2019-10-25 | 2025-06-03 | 环球展览公司 | Organic electroluminescent materials and devices |
| US11765965B2 (en) | 2019-10-30 | 2023-09-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12538698B2 (en) | 2020-01-06 | 2026-01-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12187751B2 (en) | 2020-01-08 | 2025-01-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12201013B2 (en) | 2020-01-08 | 2025-01-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11778895B2 (en) | 2020-01-13 | 2023-10-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11917900B2 (en) | 2020-01-28 | 2024-02-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12426495B2 (en) | 2020-01-28 | 2025-09-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11932660B2 (en) | 2020-01-29 | 2024-03-19 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12245502B2 (en) | 2020-02-03 | 2025-03-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12156463B2 (en) | 2020-02-07 | 2024-11-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12084465B2 (en) | 2020-02-24 | 2024-09-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12324348B2 (en) | 2020-02-28 | 2025-06-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12234251B2 (en) | 2020-03-04 | 2025-02-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12018035B2 (en) | 2020-03-23 | 2024-06-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12129269B2 (en) | 2020-04-13 | 2024-10-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11970508B2 (en) | 2020-04-22 | 2024-04-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12279520B2 (en) | 2020-04-22 | 2025-04-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12466844B2 (en) | 2020-05-13 | 2025-11-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12286410B2 (en) | 2020-05-18 | 2025-04-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12035613B2 (en) | 2020-05-26 | 2024-07-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12275747B2 (en) | 2020-06-02 | 2025-04-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12247043B2 (en) | 2020-06-11 | 2025-03-11 | Universal Display Corporation | Organic electroluminescent material and formulations, and its various uses as hosts, fluorescent dopants, and acceptors in organic light emitting diodes |
| US12279516B2 (en) | 2020-06-17 | 2025-04-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12419188B2 (en) | 2020-06-30 | 2025-09-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12336427B2 (en) | 2020-07-09 | 2025-06-17 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12157748B2 (en) | 2020-07-30 | 2024-12-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12369487B2 (en) | 2020-08-05 | 2025-07-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12065451B2 (en) | 2020-08-19 | 2024-08-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12289986B2 (en) | 2020-08-25 | 2025-04-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12369488B2 (en) | 2020-09-09 | 2025-07-22 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12543492B2 (en) | 2020-09-09 | 2026-02-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12486295B2 (en) | 2020-09-14 | 2025-12-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12221455B2 (en) | 2020-09-24 | 2025-02-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12252499B2 (en) | 2020-09-29 | 2025-03-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12435102B2 (en) | 2020-10-02 | 2025-10-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12460128B2 (en) | 2020-10-02 | 2025-11-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12486451B2 (en) | 2020-10-02 | 2025-12-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12497420B2 (en) | 2020-10-02 | 2025-12-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12286447B2 (en) | 2020-10-06 | 2025-04-29 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12520713B2 (en) | 2020-10-08 | 2026-01-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12486288B2 (en) | 2020-10-12 | 2025-12-02 | University Of Southern California | Fast phosphors utilizing HP2H ligands |
| US12312365B2 (en) | 2020-10-12 | 2025-05-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12137606B2 (en) | 2020-10-20 | 2024-11-05 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12245504B2 (en) | 2020-10-26 | 2025-03-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12247042B2 (en) | 2020-10-26 | 2025-03-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12262631B2 (en) | 2020-11-10 | 2025-03-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12528833B2 (en) | 2020-11-18 | 2026-01-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12534485B2 (en) | 2020-11-24 | 2026-01-27 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12325717B2 (en) | 2020-11-24 | 2025-06-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12516078B2 (en) | 2020-11-24 | 2026-01-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12342716B2 (en) | 2020-12-09 | 2025-06-24 | University Of Southern California | Tandem-carbene phosphors |
| US12331064B2 (en) | 2020-12-09 | 2025-06-17 | University Of Southern California | Tandem carbene phosphors |
| US12466848B2 (en) | 2020-12-10 | 2025-11-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12398164B2 (en) | 2021-02-01 | 2025-08-26 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12459966B2 (en) | 2021-02-02 | 2025-11-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12497419B2 (en) | 2021-02-22 | 2025-12-16 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12484437B2 (en) | 2021-02-26 | 2025-11-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12421262B2 (en) | 2021-03-15 | 2025-09-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12473317B2 (en) | 2021-06-04 | 2025-11-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12495715B2 (en) | 2021-06-29 | 2025-12-09 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12520715B2 (en) | 2021-07-16 | 2026-01-06 | University Of Southern California | Organic electroluminescent materials and devices |
| US12507586B2 (en) | 2021-07-21 | 2025-12-23 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12514110B2 (en) | 2021-08-06 | 2025-12-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12509628B2 (en) | 2021-12-16 | 2025-12-30 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12414433B2 (en) | 2022-02-11 | 2025-09-09 | Universal Display Corporation | Organic electroluminescent devices |
Family Cites Families (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7317047B2 (en) * | 2002-09-24 | 2008-01-08 | E.I. Du Pont De Nemours And Company | Electrically conducting organic polymer/nanoparticle composites and methods for use thereof |
| US7390438B2 (en) * | 2003-04-22 | 2008-06-24 | E.I. Du Pont De Nemours And Company | Water dispersible substituted polydioxythiophenes made with fluorinated polymeric sulfonic acid colloids |
| US7351358B2 (en) * | 2004-03-17 | 2008-04-01 | E.I. Du Pont De Nemours And Company | Water dispersible polypyrroles made with polymeric acid colloids for electronics applications |
-
2008
- 2008-07-22 CN CN200880023383A patent/CN101688052A/en active Pending
- 2008-07-22 WO PCT/US2008/070718 patent/WO2009018009A1/en not_active Ceased
- 2008-07-22 KR KR1020107004335A patent/KR20100065302A/en not_active Withdrawn
- 2008-07-22 EP EP08796400A patent/EP2173811A1/en not_active Withdrawn
- 2008-07-22 JP JP2010518326A patent/JP2010534739A/en active Pending
- 2008-07-23 TW TW097127994A patent/TW200915350A/en unknown
Cited By (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106887522B (en) * | 2010-07-26 | 2018-09-18 | 默克专利有限公司 | Include the device of nanocrystal |
| CN104520383A (en) * | 2012-07-31 | 2015-04-15 | 国际商业机器公司 | Water-dispersible electrically conductive fluorine-containing polyaniline compositions for lithography |
| CN104520383B (en) * | 2012-07-31 | 2016-10-05 | 国际商业机器公司 | For photolithographic aqueous-dispersible conductive Fluorine-containing Polyaniline compositions |
| CN109585830A (en) * | 2018-12-03 | 2019-04-05 | 江苏理工学院 | The sulphur selenium compound and its preparation of coated with conductive polymer and graphene oxide and application |
| CN114502638A (en) * | 2019-10-09 | 2022-05-13 | 大金工业株式会社 | Composition containing fluoropolymer and crosslinked product |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2010534739A (en) | 2010-11-11 |
| WO2009018009A1 (en) | 2009-02-05 |
| KR20100065302A (en) | 2010-06-16 |
| EP2173811A1 (en) | 2010-04-14 |
| TW200915350A (en) | 2009-04-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101688052A (en) | Aqueous dispersion of conductive polymer comprising inorganic nanoparticles | |
| KR101387894B1 (en) | Stabilized compositions of conductive polymers and partially-fluorinated acid polymers | |
| CN102395627B (en) | Electrically conductive polymer compositions and films made therefrom | |
| CN102349115B (en) | Electrically conductive polymer compositions for coating applications | |
| CN101977985A (en) | Electrically conductive polymer compositions and films made therefrom | |
| US8461758B2 (en) | Buffer bilayers for electronic devices | |
| KR20110044240A (en) | Aqueous Dispersion of Electroconductive Polymer Containing Inorganic Nanoparticles | |
| KR20100114052A (en) | Buffer bilayers for electronic devices | |
| JP2009534831A (en) | High energy potential double layer composition | |
| JP5587980B2 (en) | Conductive polymer composition and film made therefrom | |
| KR20110102441A (en) | Electrically conductive polymer composition | |
| KR20080108619A (en) | Electrically conductive polymer compositions | |
| CN101351851B (en) | Conductive polymer composition, aqueous dispersion containing same, and electronic device | |
| US8785913B2 (en) | Buffer bilayers for electronic devices | |
| KR20110110154A (en) | Electrically conductive polymer composition | |
| US8278405B2 (en) | Vinylphenoxy polymers | |
| US8216685B2 (en) | Buffer bilayers for electronic devices |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C02 | Deemed withdrawal of patent application after publication (patent law 2001) | ||
| WD01 | Invention patent application deemed withdrawn after publication |
Open date: 20100331 |