WO2024184050A1 - Cyclische stickstoffverbindungen für organische elektrolumineszenzvorrichtungen - Google Patents
Cyclische stickstoffverbindungen für organische elektrolumineszenzvorrichtungen Download PDFInfo
- Publication number
- WO2024184050A1 WO2024184050A1 PCT/EP2024/054151 EP2024054151W WO2024184050A1 WO 2024184050 A1 WO2024184050 A1 WO 2024184050A1 EP 2024054151 W EP2024054151 W EP 2024054151W WO 2024184050 A1 WO2024184050 A1 WO 2024184050A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- atoms
- group
- radicals
- substituted
- aromatic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/10—Spiro-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/22—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains four or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/02—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains two hetero rings
- C07D491/04—Ortho-condensed systems
- C07D491/044—Ortho-condensed systems with only one oxygen atom as ring hetero atom in the oxygen-containing ring
- C07D491/048—Ortho-condensed systems with only one oxygen atom as ring hetero atom in the oxygen-containing ring the oxygen-containing ring being five-membered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/12—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains three hetero rings
- C07D491/14—Ortho-condensed systems
- C07D491/147—Ortho-condensed systems the condensed system containing one ring with oxygen as ring hetero atom and two rings with nitrogen as ring hetero atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/22—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains four or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/12—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains three hetero rings
- C07D495/14—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/0803—Compounds with Si-C or Si-Si linkages
- C07F7/081—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te
- C07F7/0812—Compounds with Si-C or Si-Si linkages comprising at least one atom selected from the elements N, O, halogen, S, Se or Te comprising a heterocyclic ring
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/02—Silicon compounds
- C07F7/08—Compounds having one or more C—Si linkages
- C07F7/10—Compounds having one or more C—Si linkages containing nitrogen having a Si-N linkage
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F7/00—Compounds containing elements of Groups 4 or 14 of the Periodic Table
- C07F7/30—Germanium compounds
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/624—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing six or more rings
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/654—Aromatic compounds comprising a hetero atom comprising only nitrogen as heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6574—Polycyclic condensed heteroaromatic hydrocarbons comprising only oxygen in the heteroaromatic polycondensed ring system, e.g. cumarine dyes
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6576—Polycyclic condensed heteroaromatic hydrocarbons comprising only sulfur in the heteroaromatic polycondensed ring system, e.g. benzothiophene
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F1/00—Compounds containing elements of Groups 1 or 11 of the Periodic Table
- C07F1/02—Lithium compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F15/00—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table
- C07F15/0006—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table compounds of the platinum group
- C07F15/0033—Iridium compounds
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/17—Carrier injection layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/18—Carrier blocking layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/633—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising polycyclic condensed aromatic hydrocarbons as substituents on the nitrogen atom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/658—Organoboranes
Definitions
- the present invention relates to cyclic nitrogen compounds for use in electronic devices, in particular in organic electroluminescent devices, and to electronic devices, in particular organic electroluminescent devices, containing these materials.
- Electronic devices containing organic compounds are widely known and commercially available. These devices can, for example, each comprise one or more hole injection layers, hole transport layers, hole blocking layers, electron transport layers, electron injection layers, exciton blocking layers, electron blocking layers and/or charge generation layers.
- these devices can, for example, each comprise one or more hole injection layers, hole transport layers, hole blocking layers, electron transport layers, electron injection layers, exciton blocking layers, electron blocking layers and/or charge generation layers.
- there is a need for improvement with regard to the properties of these devices with the compounds used in the layers described above in particular having a major influence on the properties of the devices.
- phosphorescent organometallic complexes are often used as emitting materials in organic electroluminescent devices. For quantum mechanical reasons, up to four times the energy and power efficiency is possible when organometallic compounds are used as phosphorescent emitters. In general, there is still room for improvement in electroluminescent devices, especially in electroluminescent devices that exhibit triplet emission (phosphorescence). The properties of phosphorescent electroluminescent devices are not only determined by the triplet emitters used. The other materials used, such as matrix materials, are also of particular importance here. Improvements to these materials can therefore also lead to significant improvements in the properties of the electroluminescent devices. Similar statements also apply to organic electroluminescent devices based on fluorescent emitters or emitters exhibiting TADF (thermally activated delayed fluorescence).
- TADF thermalally activated delayed fluorescence
- heterocycles are known that can be used in organic electroluminescent devices.
- Compounds according to the present invention that can be used as matrix materials or as hole transport materials are not disclosed.
- the object of the present invention is therefore to provide compounds which are suitable for use in an organic electronic device, in particular in an organic electroluminescent device, and which lead to good device properties when used in this device, as well as to provide the corresponding electronic device.
- the object of the present invention is to provide compounds that lead to a long service life, good efficiency and low operating voltage.
- the properties of the matrix materials in particular have a significant influence on the service life and efficiency of the organic electroluminescent device.
- a further object of the present invention can be seen in providing compounds which are suitable for use in a phosphorescent or fluorescent electroluminescent device, in particular as a matrix material.
- the compounds especially when used as matrix materials or as hole transport materials in organic electroluminescent devices, should lead to devices that exhibit excellent lifetime and efficiency.
- the electronic devices should be able to be used or adapted for many purposes.
- the performance of the electronic devices should be maintained over a wide temperature range.
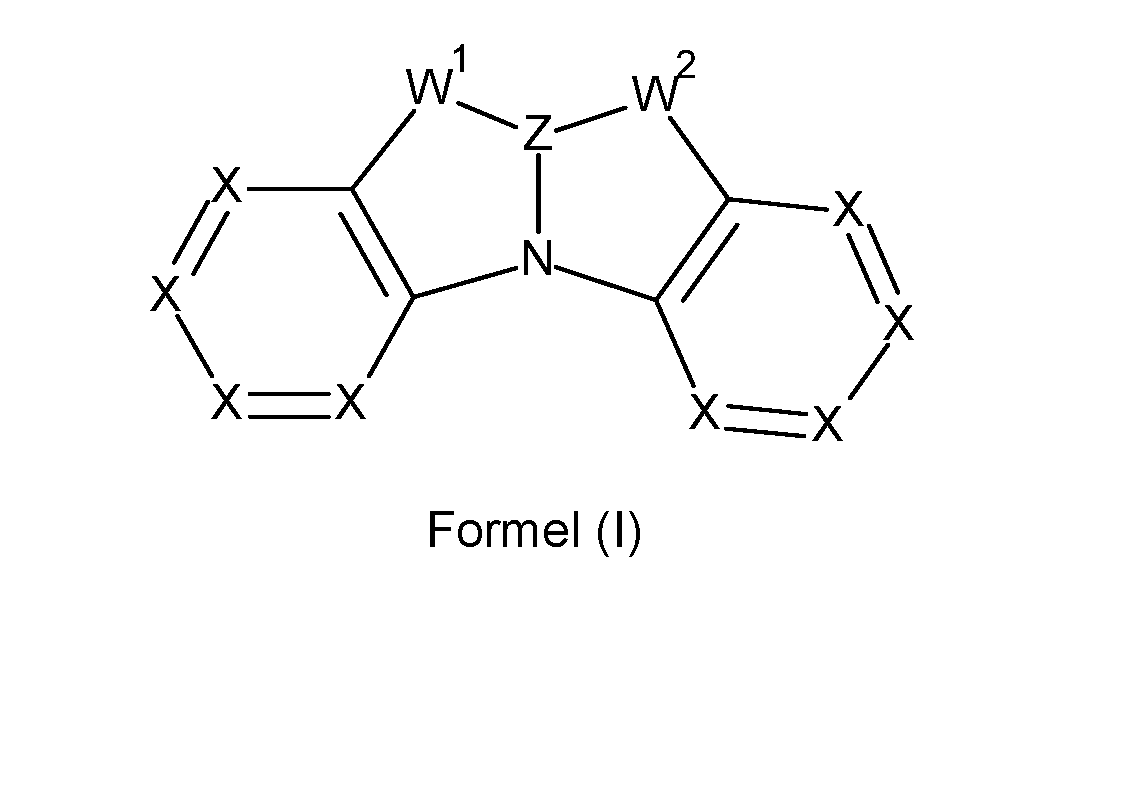
- the present invention relates to a compound comprising at least one structure of formula (I), preferably a compound according to formula (I),
- Z stands at each occurrence, identically or differently, for C(R a ), Si(R b ) or Ge(R b ), preferably for C(R a ) or Si(R b ) and particularly preferably for C(R a );
- W 1 stands at each occurrence, identically or differently, for a group - C(R C ) 2-, -C(R C ) 2-C(R C ) 2 - or -C(R C ) 2 -C(R c ) 2 -C(R c ) 2 -;
- W 2 stands at each occurrence, identically or differently, for a group -C(R d ) 2-, -C(R d ) 2-C(R d ) 2 - or -C(R d ) 2 -C(R c ) 2 -C(R d ) 2 -;
- X stands at each occurrence, identically or differently, for N or C(R), preferably for C(R), with the proviso that not more than two of the X groups in a cycle stand for N;
- the compound does not comprise a boron atom. Furthermore, it can be provided that the compound is not ionically charged and/or does not represent a salt.
- An aryl group in the sense of this invention contains 6 to 40 C atoms; a heteroaryl group in the sense of this invention contains 3 to 40 C atoms and at least one heteroatom, with the proviso that the sum of C atoms and heteroatoms is at least 5.
- the heteroatoms are preferably selected from N, O and/or S.
- An aryl group or heteroaryl group is understood to be either a simple aromatic cycle, i.e.
- benzene or a simple heteroaromatic cycle, for example pyridine, pyrimidine, thiophene, etc., or a condensed (fused) aryl or heteroaryl group, for example naphthalene, anthracene, phenanthrene, quinoline, isoquinoline, etc.
- Aromatics linked to one another by a single bond, such as biphenyl, are not referred to as aryl or heteroaryl groups, but as aromatic ring systems.
- An electron-poor heteroaryl group in the sense of the present invention is a heteroaryl group which has at least one heteroaromatic six-membered ring with at least one nitrogen atom. Further aromatic or heteroaromatic five-membered rings or six-membered rings can be fused to this six-membered ring. Examples of electron-poor heteroaryl groups are pyridine, pyrimidine, pyrazine, pyridazine, triazine, quinoline, quinazoline or quinoxaline.
- An aromatic ring system in the sense of this invention contains 6 to 60 C atoms in the ring system.
- a heteroaromatic ring system in the sense of this invention contains 3 to 60 C atoms and at least one heteroatom in the ring system, with the proviso that the sum of C atoms and heteroatoms is at least 5.
- the heteroatoms are preferably selected from N, O and/or S.
- An aromatic or heteroaromatic ring system in the sense of this invention is to be understood as a system which does not necessarily only contain aryl or heteroaryl groups, but in which several aryl or heteroaryl groups can also be connected by a non-aromatic unit, such as a C, N or O atom.
- systems such as fluorene, 9,9'-spirobifluorene, 9,9-diarylfluorene, triarylamine, diaryl ether, stilbene, etc. are to be understood as aromatic ring systems in the sense of this invention, as are systems in which two or more aryl groups are linked, for example, by a short alkyl group.
- the aromatic ring system is preferably selected from fluorene, 9,9'-spirobifluorene, 9,9-diarylamine or groups in which two or more aryl and/or heteroaryl groups are linked to one another by single bonds.
- an aliphatic hydrocarbon radical or an alkyl group or an alkenyl or alkynyl group which can contain 1 to 20 C atoms and in which individual H atoms or CH2 groups can also be substituted by the abovementioned groups, preferably the radicals methyl, ethyl, n-propyl, i-propyl, n-butyl, i-butyl, s-butyl, t-butyl, 2-methylbutyl, n-pentyl, s-pentyl, neo-pentyl, cyclopentyl, n-hexyl, neo-hexyl, cyclohexyl, n-heptyl, cycloheptyl, n-octyl, cyclooctyl, 2-ethylhexyl, trifluoromethyl, pentafluoroethyl, 2,2,
- An alkoxy group with 1 to 40 carbon atoms is preferably methoxy, trifluoromethoxy, ethoxy, n-propoxy, i-propoxy, n-butoxy, i-butoxy, s-butoxy, t-butoxy, n-pentoxy, s-pentoxy, 2-methylbutoxy, n-hexoxy, cyclohexyloxy, n-heptoxy, cycloheptyloxy, n -octyloxy, cyclo- octyloxy, 2-ethylhexyloxy, pentafluoroethoxy and 2,2,2-trifluoroethoxy.
- a thioalkyl group with 1 to 40 C atoms is understood to mean in particular methylthio, ethylthio, n-propylthio, i-propylthio, n-butylthio,
- 2-ethylhexylthio trifluoromethylthio, pentafluoroethylthio, 2,2,2-trifluoroethylthio, ethenylthio, propenylthio, butenylthio, pentenylthio, cyclopentenylthio, hexenylthio, cyclohexenylthio, heptenylthio, cycloheptenylthio, octenylthio, cyclooctenylthio, ethynylthio, propynylthio, butynylthio, pentynylthio, hexynylthio, heptynylthio or octynylthio.
- alkyl, alkoxy or thioalkyl groups according to the present invention can be straight-chain, branched or cyclic, where one or more non-adjacent CH2 groups can be replaced by the above-mentioned groups;
- one or more H atoms can be replaced by D, F, CI, Br, I, CN or NO2, preferably F, CI or CN, more preferably F or CN, particularly preferably CN.
- An aromatic or heteroaromatic ring system with 5 - 60 or 5 to 40 aromatic ring atoms, which can be substituted with the above-mentioned radicals and which can be linked to the aromatic or heteroaromatic via any position, is understood to mean in particular groups which are derived from benzene, naphthalene, anthracene, benzanthracene, phenanthrene, pyrene, chrysene, perylene, fluoranthene, naphthacene, pentacene, benzopyrene, biphenyl, biphenylene, terphenyl, triphenylene, fluorene, spirobifluorene, dihydrophenanthrene, dihydropyrene, tetrahydropyrene, cis- or trans-indenofluorene, cis- or trans-indenocarbazole, cis- or trans-indolocarbazole, truxene, is
- two or more radicals can form a ring is understood in the context of the present description to mean, among other things, that the two radicals are linked to one another by a chemical bond with formal elimination of two hydrogen atoms. This is illustrated by the following scheme.
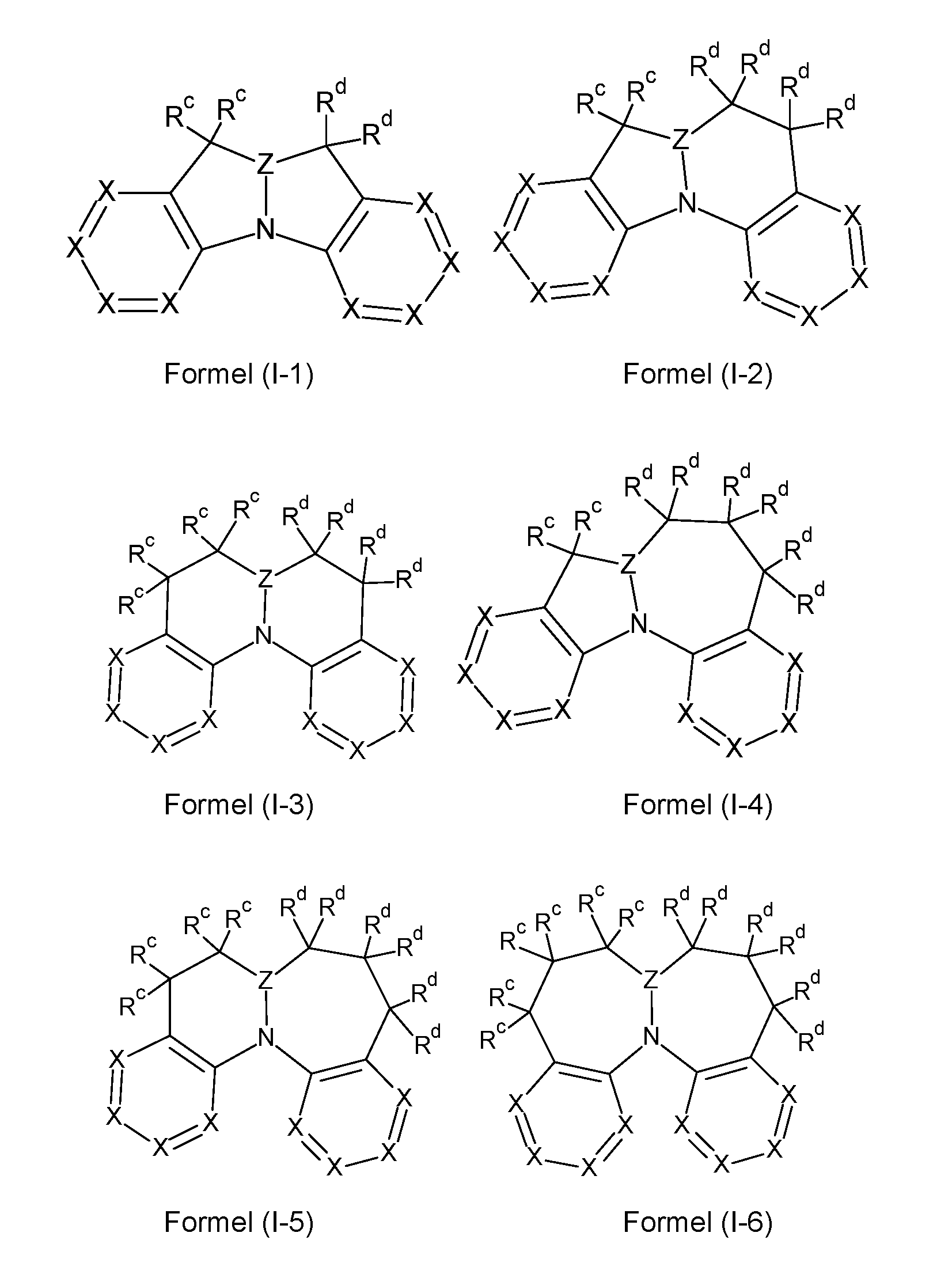
- the compounds according to the invention can preferably comprise at least one structure of the formulas (I-1) to (I-6), and are particularly preferably selected from the compounds of the formulas (I-1) to (I-6),
- Structures/compounds of formulas (I-1) to (I-4) are preferred and structures/compounds of formulas (I-1) to (I-3) are particularly preferred and structures/compounds of the formula (1-1 ) are particularly preferred.
- the radical Ar or at least one radical R represents an aromatic or heteroaromatic ring system having 5 to 18, preferably 5 to 13, particularly preferably 6 to 13 aromatic ring atoms, which may be substituted by one or more radicals R e .
- the radical Ar is selected, identically or differently on each occurrence, from phenyl, biphenyl, terphenyl, quaterphenyl, fluorene, spirobifluorene, naphthalene, indole, benzofuran, benzothiophene, carbazole, dibenzofuran, dibenzothiophene, indenocarbazole, indolocarbazole, pyridine, pyrimidine, pyrazine, pyridazine, triazine, quinoline, isoquinoline, quinazoline, quinoxaline, phenanthrene or triphenylene, which can each be substituted by one or more radicals R e , preferably phenyl, biphenyl, fluorene, dibenzofuran, triphenylene, carbazole, indolocarbazole.
- At least one radical R is selected from phenyl, biphenyl, terphenyl, quaterphenyl, fluorene, spirobifluorene, naphthalene, indole, benzofuran, benzothiophene, carbazole, dibenzofuran, dibenzothiophene, indenocarbazole, indolocarbazole, pyridine, pyrimidine, pyrazine, pyridazine, triazine, quinoline, isoquinoline, quinazoline, quinoxaline, phenanthrene or triphenylene, which can each be substituted by one or more radicals R e , preferably phenyl, biphenyl, fluorene, dibenzofuran, triphenylene, carbazole, indolocarbazole.
- the radical Ar or R can preferably represent a phenyl group which is substituted by at least one radical R e , where the substituent is in the ortho, meta or para position relative to the bonding site to the nitrogen atom.
- R e is a phenyl group
- an ortho, meta or para biphenyl group can be formed.
- the radical Ar or R represents a triazine group
- the triazine group has two radicals R e which are not H or D, where the two radicals R e are preferably an aromatic or heteroaromatic ring system having 5 to 60, preferably 6 to 30 aromatic ring atoms, which can each be substituted by one or more radicals R 1 .
- the radical Ar or R can preferably represent a phenyl group which is substituted by at least one radical R e , where the substituents together with the phenyl group represented by the group Ar are a fluorene radical which can be linked via the 1-, 2-, 3- or 4-position, a spirobifluorene radical which can be linked via the 1-, 2-, 3- or 4-position, an indole radical, a benzofuran radical, a benzothiophene radical, a carbazole radical which can be linked via the 1-, 2-, 3- or 4-position, a dibenzofuran radical which can be linked via the 1-, 2-, 3- or 4-position, a dibenzothiophene radical which can be linked via the 1-, 2-, 3- or 4-position, a Indenocarbazole residue or an indolocarbazole residue is formed.
- the structure/compound according to the present invention comprises at least one electron transport group, preferably a triazine group.
- Electron transport groups are widely known in the art and promote the ability of compounds to transport and/or conduct electrons. Examples of electron transport groups are pyridine, pyrimidine, pyrazine, pyridazine, triazine, quinazoline, quinoxaline, quinoline, isoquinoline, imidazole and/or benzimidazole groups, with triazine groups being particularly preferred.
- the structure/compound according to the present invention does not comprise a phosphine oxide residue, preferably no electron-withdrawing residue.
- the structure/compound according to the present invention has at least one hole transport group.
- Hole transport groups are also known in the art, and these preferably comprise triarylamine or carbazole groups.
- the present compounds are particularly suitable as host material for emitters, preferably as host material for singlet, triplet and TADF emitters, electron transport material, electron injection material, hole conductor material, hole injection material, electron blocking material, hole blocking material in an electronic device.
- the specific properties of the compounds are depending on the type and number of the respective functional groups.
- Compounds which comprise one, two or more electron transport groups but no hole transport group are particularly suitable as host material.
- Compounds which comprise one, two or more hole transport groups but no electron transport group are particularly suitable as host material, hole conductor material, hole injection material and/or electron blocking material.
- Compounds which comprise one, two or more hole transport groups and one, two or more electron transport groups and/or electron-withdrawing residues are particularly suitable as host material.
- radicals R, R c , R d , R e are not equal to H, preferably not equal to H, D, OH, NO2, F, CI, Br, I. Furthermore, it can be provided that none of the radicals R, R a , R b , R c , R d , R e is/are equal to OH, NO2, F, CI, Br, I.
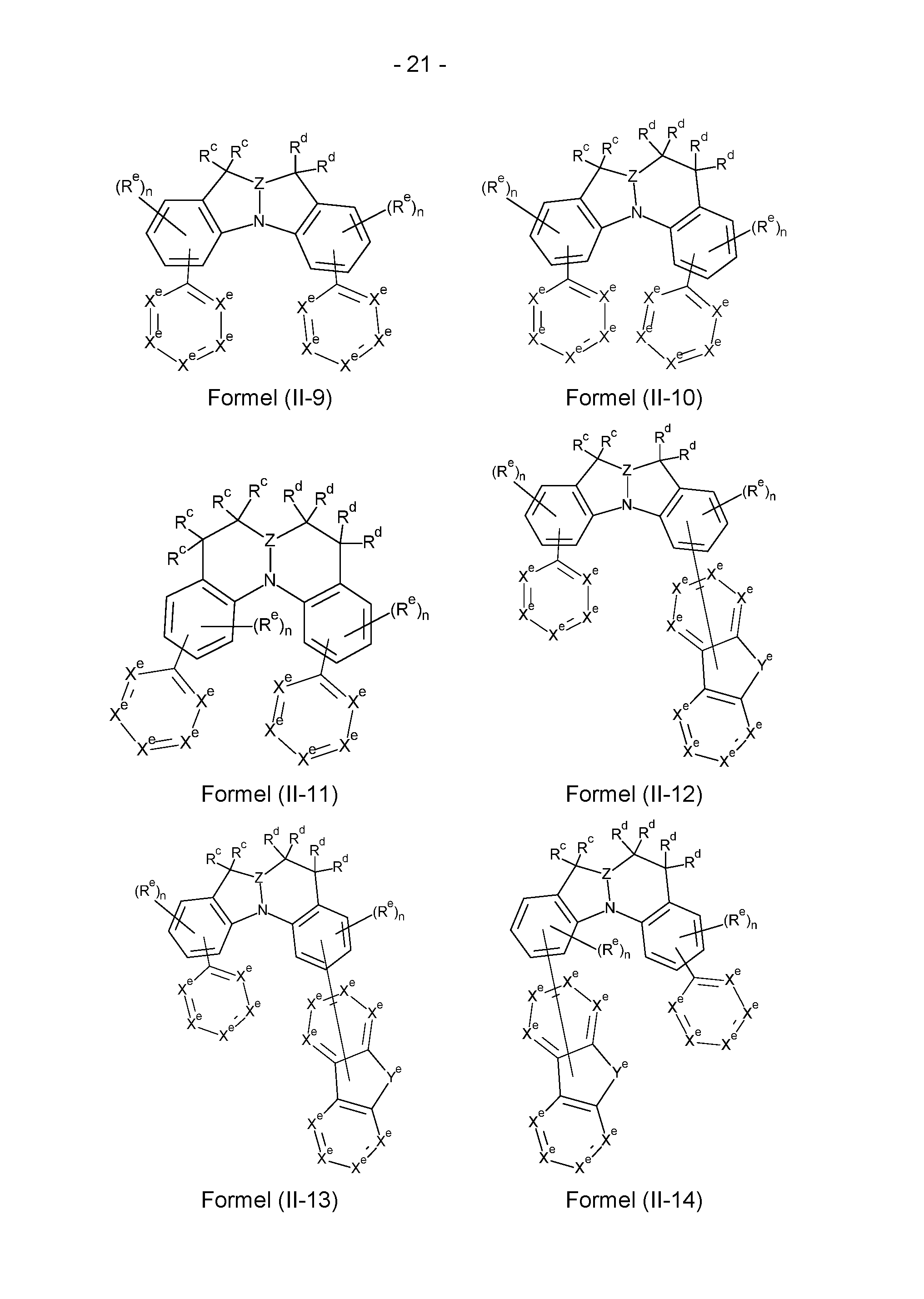
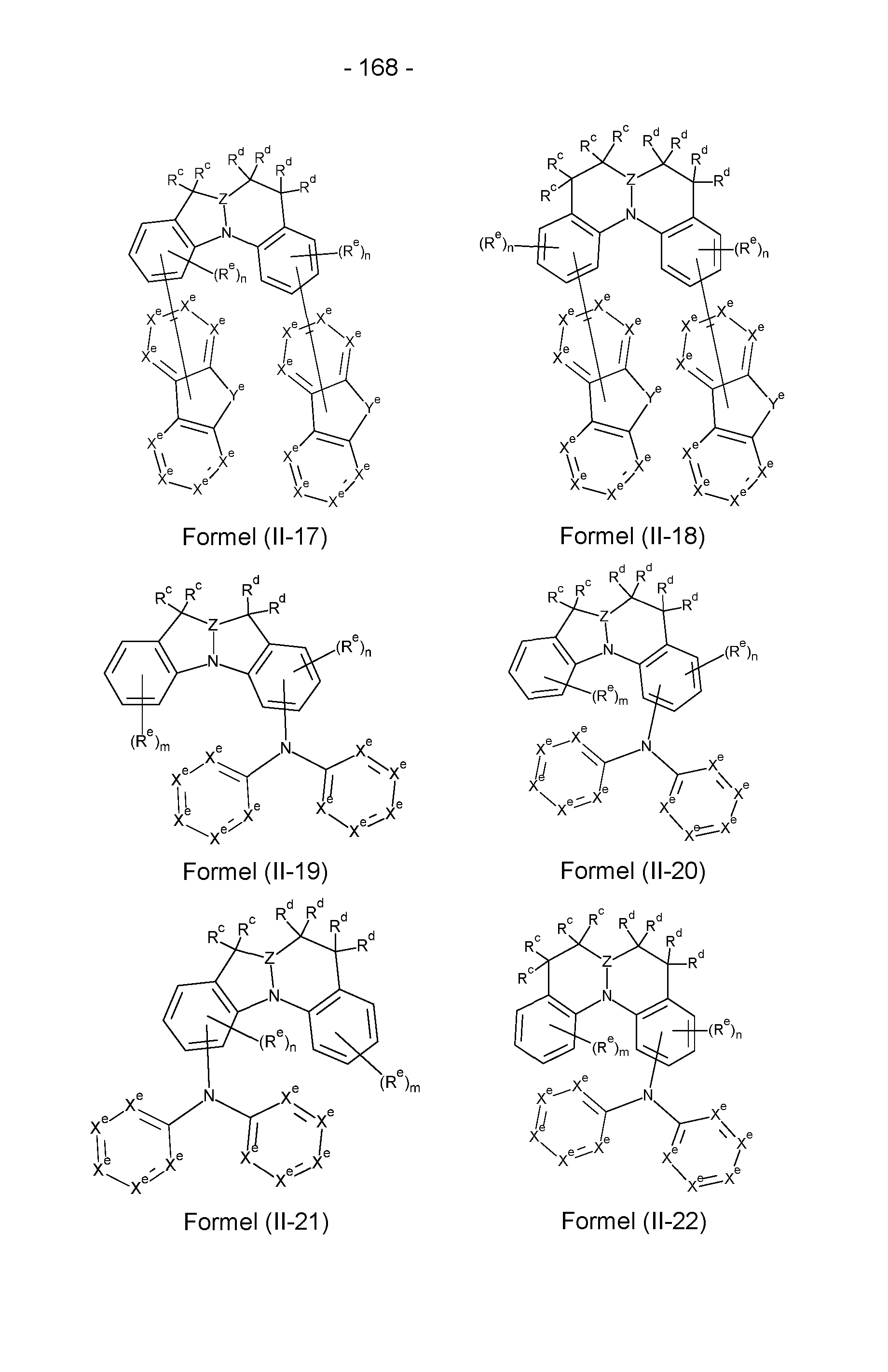
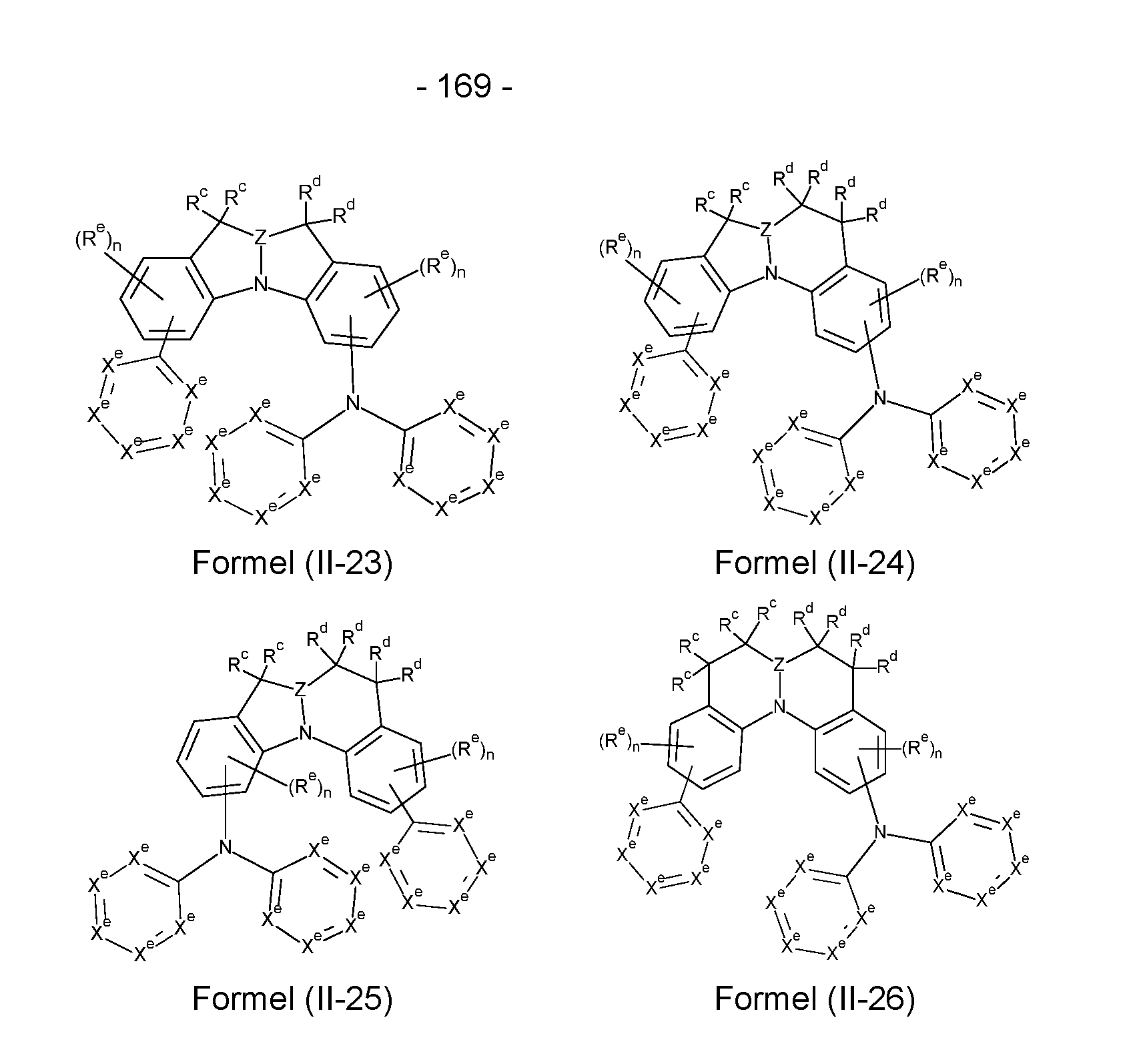
- the compounds according to the invention have a structure of the formulas (II-1) to
- X e stands on each occurrence, identically or differently, for N, CR e or C, in the case that a group binds to the structure, preferably for CR e or C, with the proviso that not more than three of the groups X e in a cycle stand for N, where R e has the meaning given in claim 1; n is 0, 1, 2 or 3, preferably 0, 1 or 2, m is 0, 1, 2, 3 or 4, preferably 0, 1 or 2.
- Structures/compounds of the formulae (II-1) to (II-5), (II-9), (11-11), (11-12), (11-16), (11-19) and (II-23) are preferred and structures/compounds of the formulae (II-1), (II-5), (II-9), (11-12) and (11-16) are particularly preferred.
- X e stands for C if a group binds to the respective structure.
- This group is in particular the ring structure shown in formulae (II-5) to (II-8) and formulae (11-12) to (11-18) and provided with a radical Y e .
- This ring structure can bind here via a radical X e or via Y e , where in the latter case Y e stands for N-, C(R e )-, Si(R e )- or Ge(R e )-.
- At least one, preferably at least two and particularly preferably three groups X e per ring represent N, whereby these groups are preferably not adjacent.
- These structures/compounds preferably comprise Electron transport groups and are therefore particularly suitable as matrix materials.
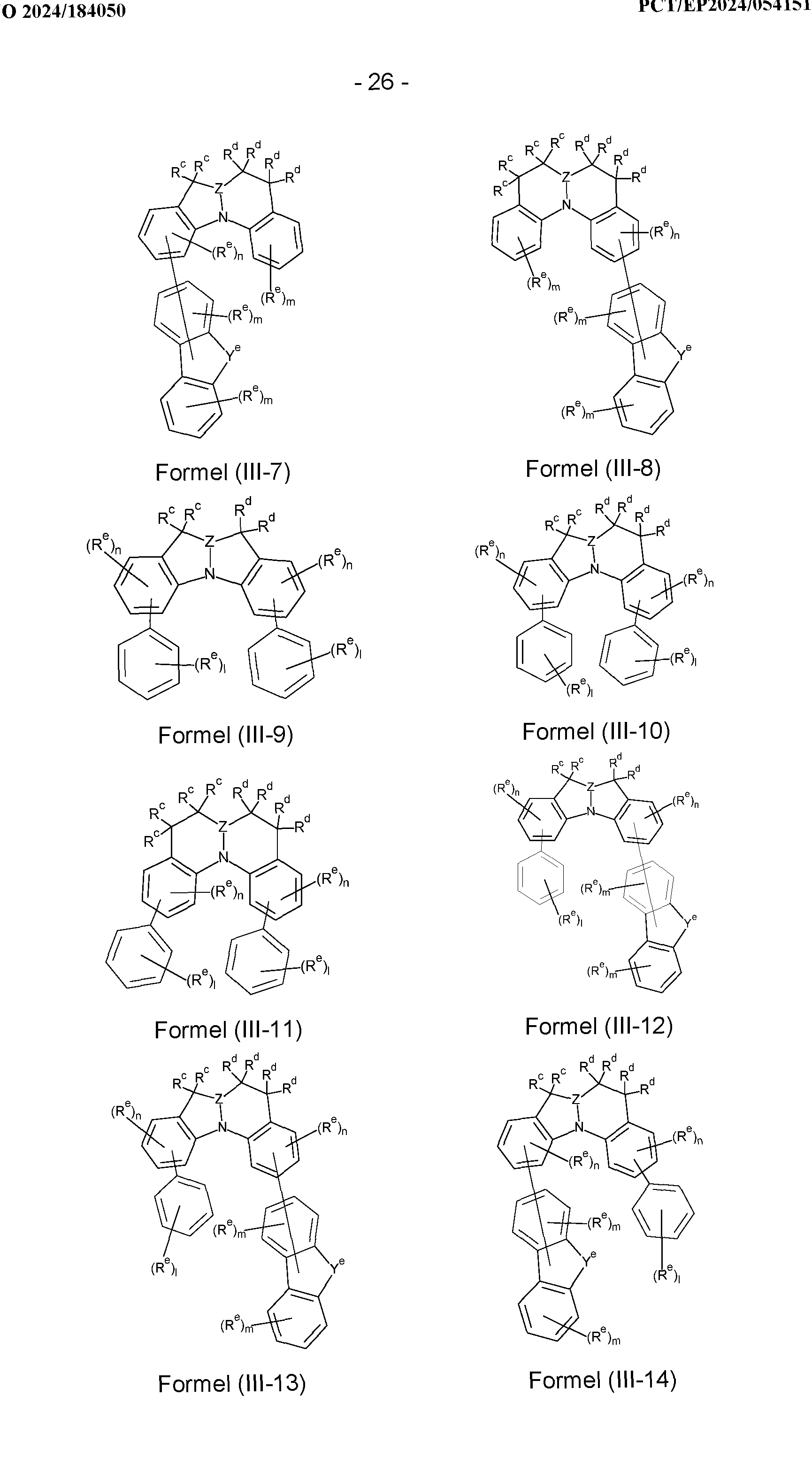
- the compounds according to the invention comprise a structure of the formulae (III-1) to (III-34), wherein the compounds according to the invention can particularly preferably be selected from the compounds of the formulae (III-1) to (III-34),
- a group is bonded to the structure, is N-, C(R e )-, Si(R e )- or Ge(R e )-; j is 0, 1 or 2; n is 0, 1, 2 or 3, preferably 0, 1 or 2; m is 0, 1, 2, 3 or 4, preferably 0, 1 or 2; l is 0, 1, 2, 3, 4 or 5, preferably 0, 1 or 2.
- Structures/compounds of the formulae (III-1) to (III-5), (III-9), (III-11), (III-12), (III-16), (III-19) and (III-23) are preferred and structures/compounds of the formulae (III-1) to (III-5), (III-9), (III-11), (III-12), (III-16) and (III-19) are particularly preferred and structures/compounds of the formulae (III-9), (III-12) and (III-19) are very particularly preferred.
- the sum of the indices j, m, n and l in structures/compounds of the formulas (III-1) to (III-34) is preferably at most 6, particularly preferably at most 4 and particularly preferably at most 2.
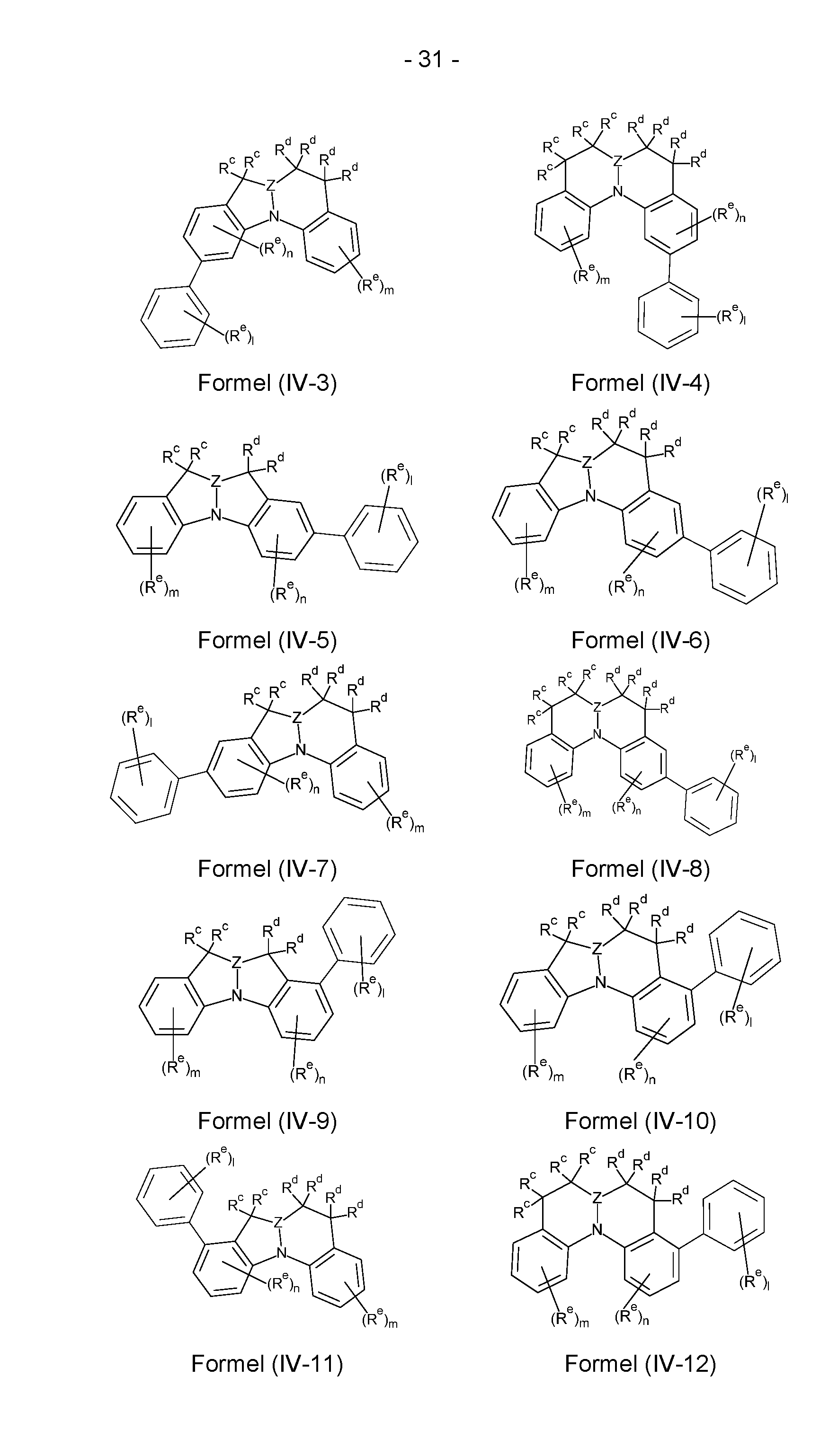
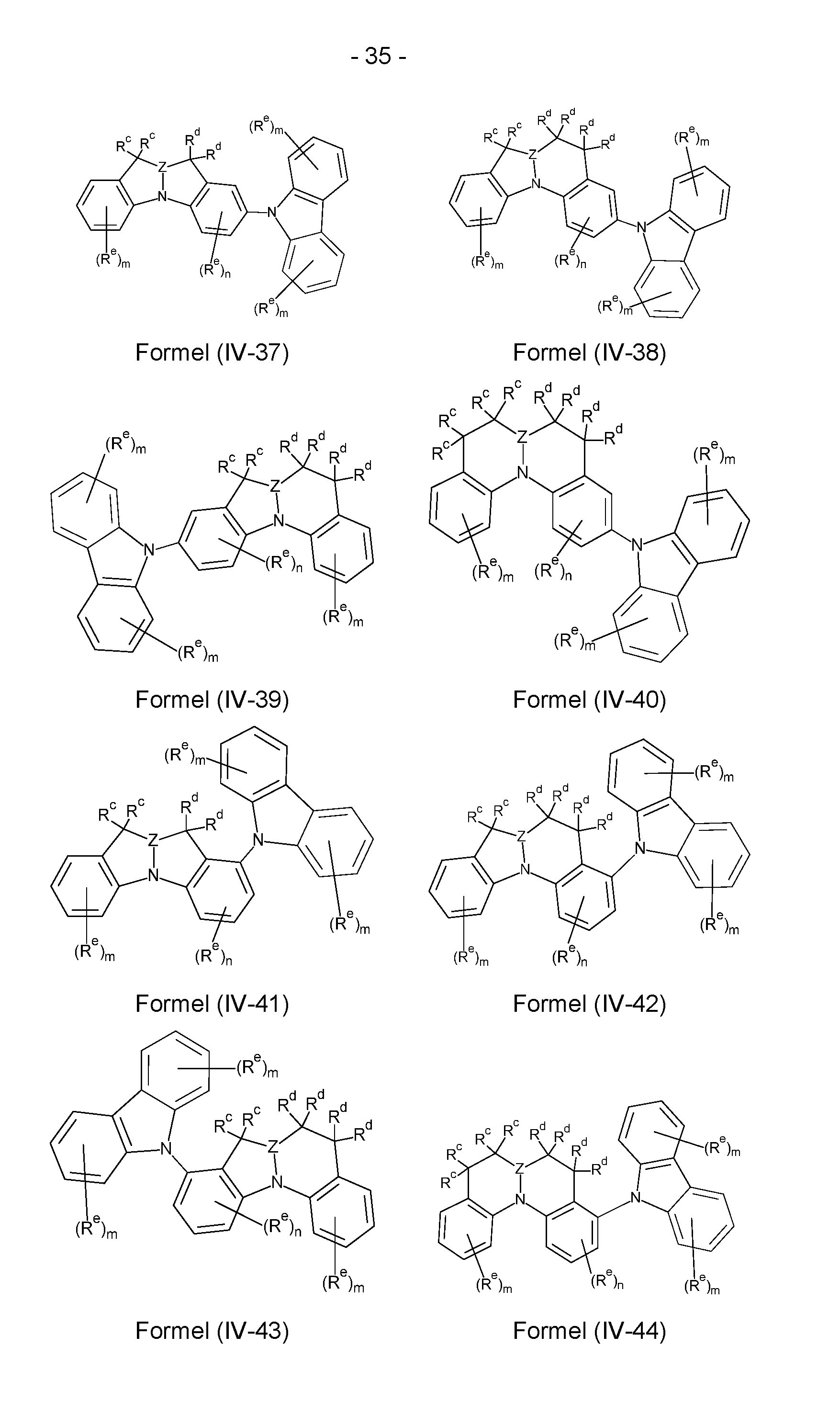
- the compounds according to the invention comprise a structure of the formulas (IV-1) to (IV-48), where the compounds according to the invention can particularly preferably be selected from the compounds of the formulas (IV-1) to (IV-48),
- Structures/compounds of the formulas (IV-1) to (IV-8), (IV-17) to (IV-24) and (IV-33) to (IV-40) are preferred, structures/compounds of the formulas (IV-1), (IV-4), (IV-5), (IV-8), (IV-17), (IV-20), (IV-21), (IV-24), (IV-33), (IV-36), (IV-37) and (IV-40) are particularly preferred and structures/compounds of the formulas IV-1), (IV-5), (IV-17), (IV-21), (IV-33) and (IV-37) are very particularly preferred.
- the sum of the indices m, n and I in structures/compounds of the formulas (IV-1) to (IV-48) is preferably at most 6, particularly preferably at most 4 and particularly preferably at most 2.
- radicals which can be selected in particular from R, R e , R 1 and/or R 2 , form a ring system with one another, this can be mono- or polycyclic, aliphatic, heteroaliphatic, aromatic or heteroaromatic.
- radicals with a group R a , R b , R c and/or R d form a ring system, this can be mono- or polycyclic, aliphatic or heteroaliphatic.
- two radicals R c and/or R d can represent an aromatic or heteroaromatic ring system which is linked, for example two radicals R c and/or R d can form a fluorene group or a phenanthrenyl group, etc.
- the radicals which form a ring system with one another can be adjacent, ie these radicals are bonded to the same carbon atom or to carbon atoms which are directly bonded to one another, or they can be further apart from one another.
- At least two, preferably adjacent radicals R c , R d , R e form a condensed ring with the other groups to which the two radicals R c , R d , R e bind.
- ring structures are formed which are described in the publication WO 2022/079068 A1 , filed on October 13, 2021 with the European Patent Office under the application number PCT7EP2021/078240, wherein for disclosure purposes the description of the condensed ring structures set out in these publications, which are described by the ring elements of the formulas (RA-1 ) to (RA-12), (RA-1 a) to (RA-4f) and/or (RB) on pages 37 to 40 of the publication WO 2022/079068 A1, is incorporated by reference into the ring structures set out above and described in more detail in the publication WO 2022/079068 A1, which preferably comprise the ring elements of the formulas (RA-1) to (RA-12) and (RA-1 a) to (RA-4f), lead in particular to structures/compounds according to the invention which have a surprisingly low refractive index.
- the substituents R, R a , R b , R c , R d , R e , R 1 and R 2 do not form a condensed aromatic or heteroaromatic ring system, particularly preferably no ring system, with the ring atoms of the ring system to which the substituents R, R a , R b , R c , R d , R e , R 1 and R 2 are bonded.
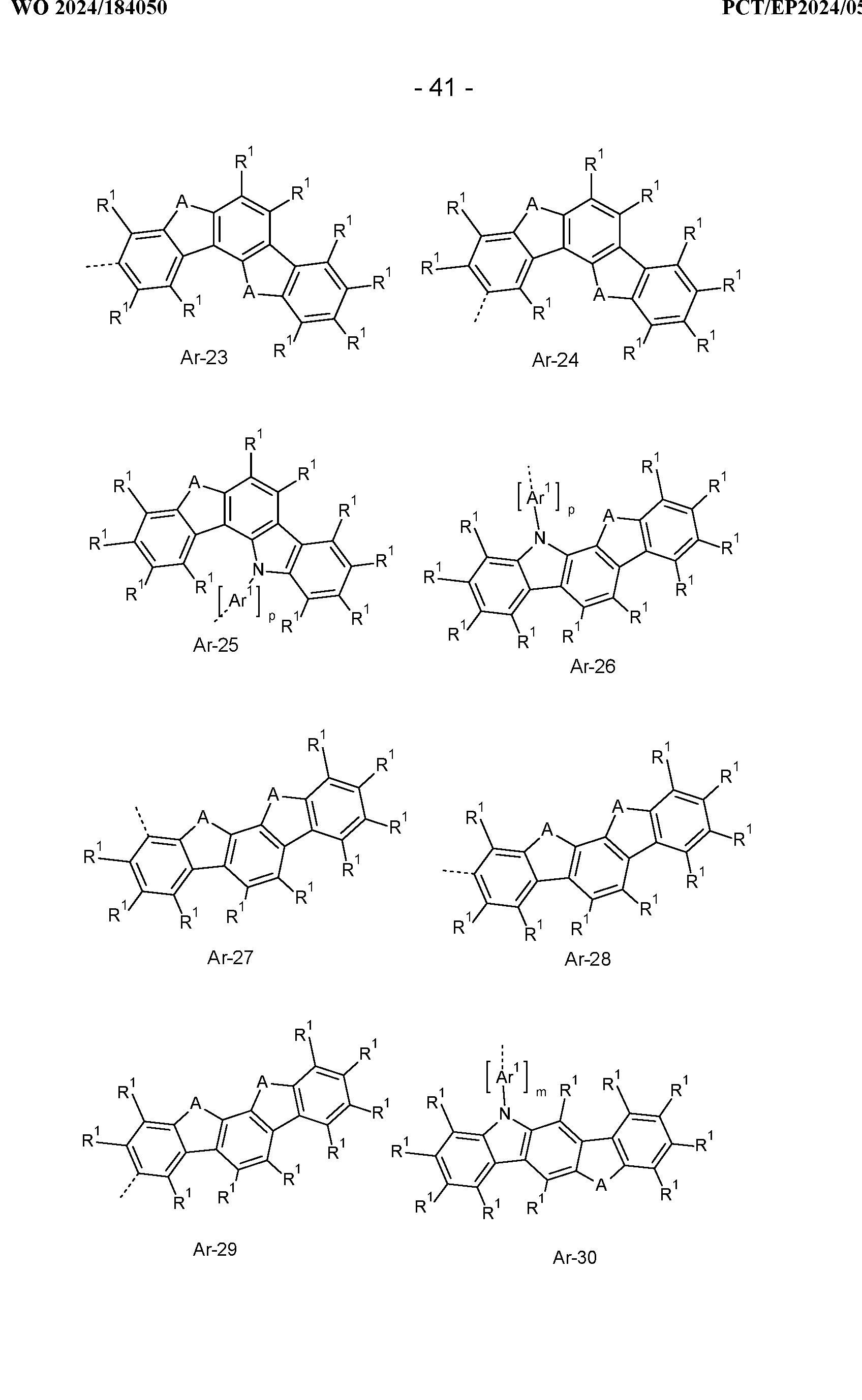
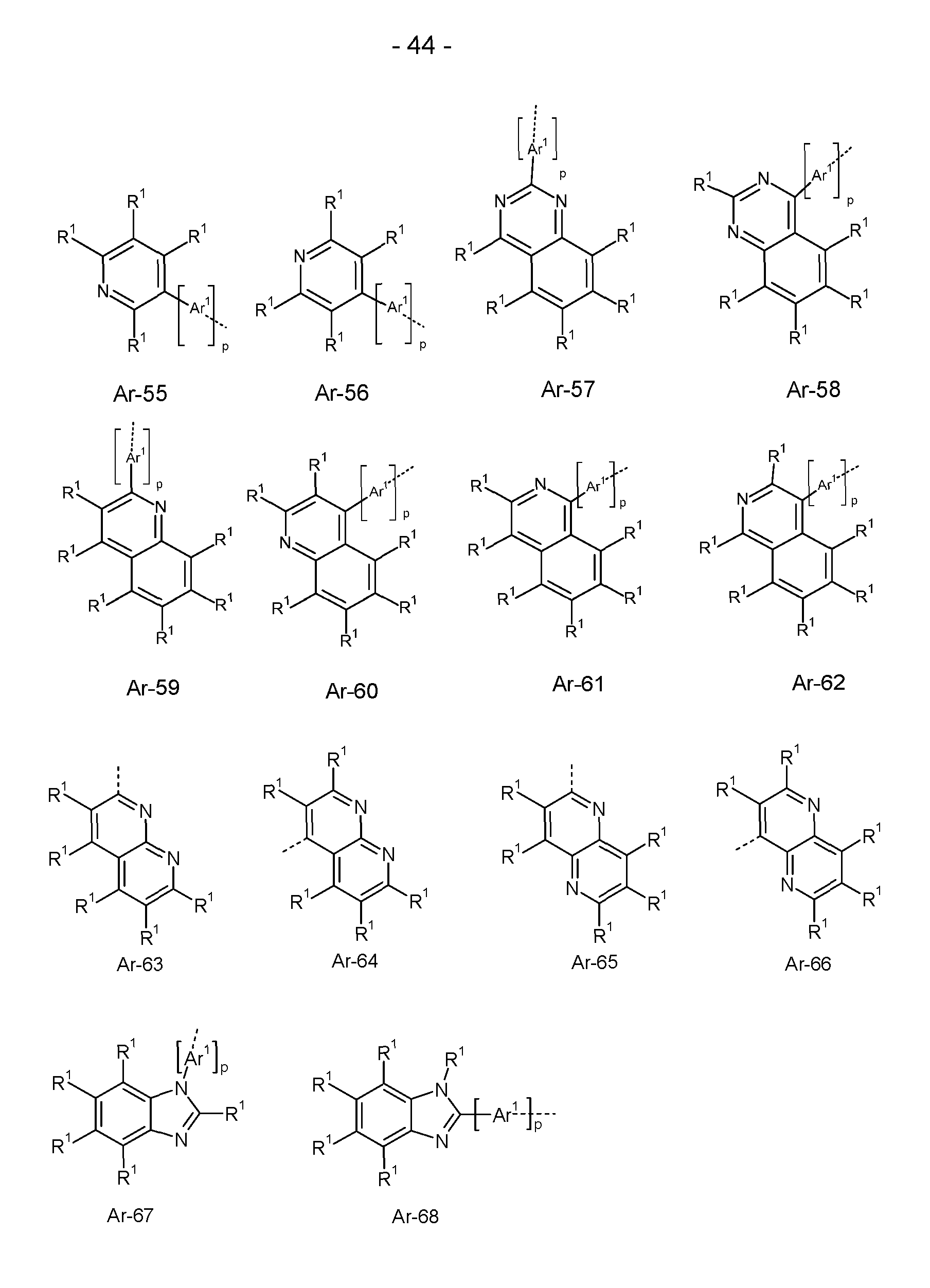
- At least one radical R a , R b , R c , R d , R e is selected, identically or differently on each occurrence, from the group consisting of H, D, a branched or cyclic alkyl, alkoxy or thioalkoxy group having 3 to 20 C atoms or an aromatic or heteroaromatic ring system selected from the groups of the following formulas Ar-1 to Ar-76, preferably the radical R e is selected, identically or differently on each occurrence, from the group consisting of H, D or an aromatic or heteroaromatic ring system selected from the groups of the following formulas Ar-1 to Ar-76, and/or the group Ar' is selected, identically or differently on each occurrence, from the groups of the following formulas Ar-1 to Ar-76,
- Ar 1 is, identically or differently at each occurrence, a bivalent aromatic or heteroaromatic ring system having 6 to 18 aromatic ring atoms, which may each be substituted by one or more radicals R 1 ;
- the substituent R 1 which is bonded to the nitrogen atom preferably represents an aromatic or heteroaromatic ring system having 5 to 24 aromatic ring atoms, which may also be substituted by one or more radicals R 2.
- this substituent R 1 identical or different on each occurrence, represents an aromatic or heteroaromatic ring system having 6 to 24 aromatic ring atoms, in particular having 6 to 18 aromatic ring atoms, which has no condensed aryl groups and which has no condensed heteroaryl groups in which two or more aromatic or heteroaromatic 6-ring groups are directly condensed to one another, and which may in each case also be substituted by one or more radicals R 2.
- Phenyl, biphenyl, terphenyl and quaterphenyl are preferred. Preference is also given to triazine, pyrimidine and quinazoline, as listed above for Ar-47 to Ar-50, Ar-57 and Ar-58, where these structures can be substituted by one or more radicals R 2 instead of R 1 .
- the substituents R 1 bonded to this carbon atom are preferably identical or different each occurrence represents a linear alkyl group having 1 to 10 C atoms or a branched or cyclic alkyl group having 3 to 10 C atoms or an aromatic or heteroaromatic ring system having 5 to 24 aromatic ring atoms, which may also be substituted by one or more radicals R 2 .
- R 1 most preferably represents a methyl group or a phenyl group.
- the radicals R 1 may also form a ring system with one another, resulting in a spiro system.
- the group Z can be C(R a ) and the group R a can be selected from H, D, a straight-chain alkyl group having 1 to 10 C atoms, a branched or cyclic alkyl group having 3 to 15 C atoms, where the alkyl groups can each be substituted by one or more radicals R 1 or an aromatic or heteroaromatic ring system having 5 to 20 aromatic ring atoms, each of which can be substituted by one or more radicals R 1 , preferably H, D, a straight-chain alkyl group having 1 to 10 C atoms, a branched or cyclic alkyl group having 3 to 15 C atoms or a phenyl group, each of which can be substituted by one or more radicals R 1 , preferably deuterated.
- the group Z stands for Si(R b ) or Ge(R b ), preferably for Si(R b ) and the group R b is selected from a straight-chain alkyl group with 1 to 10 C atoms, a branched or cyclic alkyl group with 3 to 15 C atoms, where the alkyl groups can each be substituted by one or more radicals R 1 or an aromatic or heteroaromatic ring system with 5 to 20 aromatic ring atoms, which can each be substituted by one or more radicals R 1 , preferably a straight-chain alkyl group with 1 to 10 C atoms, a branched or cyclic alkyl group with 3 to 15 C atoms or a phenyl group, which can each be substituted by one or more radicals R 1 , preferably can be deuterated.
- R c and R d are described below.
- the groups R c bonded to a C atom can be identical.
- the groups R c , R d can be H or D.
- the groups R c bonded to a C atom or the groups R d bonded to a C atom are selected from straight-chain alkyl groups having 1 to 10 C atoms or branched or cyclic alkyl groups having 3 to 10 C atoms, which can each be substituted by one or more radicals R 1 , preferably can be deuterated, whereby two or more, preferably adjacent substituents R c , R d can form a ring system with one another.
- the groups R c , R d bonded to a C atom are selected from aromatic or heteroaromatic ring systems having 5 to 20 aromatic ring atoms, each of which can be substituted by one or more radicals R 1 , preferably representing phenyl groups, each of which can be substituted by one or more radicals R 1 , preferably deuterated, where two or more, preferably adjacent, substituents R c , R d can form a ring system with one another.
- R c and R d are the same or different on each occurrence and are selected from the group consisting of H, D, a straight-chain alkyl group having 1 to 20 C atoms or a branched or cyclic alkyl group having 3 to 20 C atoms, where the alkyl group may in each case be substituted by one or more radicals R 1 , or an aromatic or heteroaromatic ring system having 5 to 60 aromatic ring atoms, preferably having 5 to 40 aromatic ring atoms, which may in each case be substituted by one or more radicals R 1 .
- At least one radical R c and/or R d preferably a substituent R c and/or R d is selected, identically or differently on each occurrence, from the group consisting of H, D, an aromatic or heteroaromatic ring system having 6 to 30 aromatic ring atoms, which may be substituted by one or more radicals R 1 , or a group N(Ar') 2 , particularly preferably at least one substituent R c and/or R d is selected, identically or differently on each occurrence, from the group consisting of an aromatic or heteroaromatic ring system having 6 to 30 aromatic ring atoms, which may be substituted by one or more radicals R 1 .
- the substituents R c and/or R d either form a condensed ring or the radical R c and/or R d is the same or different on each occurrence and is selected from the group consisting of H, D, an aromatic or heteroaromatic ring system having 6 to 30 aromatic ring atoms, which may be substituted by one or more radicals R 1 , or a group N(Ar')2.
- the radical R c and/or R d preferably the substituent R c and/or R d , is the same or different on each occurrence and is selected from the group consisting of H, D or an aromatic or heteroaromatic ring system having 6 to 24 aromatic ring atoms, preferably having 6 to 18 aromatic ring atoms, particularly preferably having 6 to 13 aromatic ring atoms, which may each be substituted by one or more radicals R 1 .
- R e is the same or different on each occurrence and is selected from the group consisting of H, D, F, CN, Si(R 1 ) s, Ge(R 1 ) s, a straight-chain alkyl group having 1 to 20 C atoms or a branched or cyclic alkyl group having 3 to 20 C atoms, where the alkyl group may in each case be substituted by one or more radicals R 1 , or an aromatic or heteroaromatic ring system having 5 to 60 aromatic ring atoms, preferably having 5 to 40 aromatic ring atoms, which may in each case be substituted by one or more radicals R 1 .
- radical R e is the same or different on each occurrence and is selected from the group consisting of H, D, F, a straight-chain alkyl group having 1 to 20 C atoms or a branched or cyclic alkyl group having 3 to 20 C atoms, where the alkyl group may in each case be substituted by one or more radicals R 1 , or an aromatic or heteroaromatic ring system having 5 to 60 aromatic ring atoms, preferably having 5 to 40 aromatic ring atoms, which may in each case be substituted by one or more radicals R 1 .
- At least one radical R e preferably a substituent R e
- R e is selected identically or differently on each occurrence from the group consisting of H, D, an aromatic or heteroaromatic ring system with 6 to 30 aromatic ring atoms, which can be substituted with one or more radicals R 1 , or a group N(Ar')2, particularly preferably at least one substituent R e is selected identically or differently on each occurrence from the group consisting of an aromatic or heteroaromatic ring system with 6 to 30 aromatic ring atoms, which can be substituted with one or more radicals R 1 .
- the substituents R e either form a condensed ring or the radical R e is selected identically or differently on each occurrence from the group consisting of H, D, an aromatic or heteroaromatic ring system with 6 to 30 aromatic ring atoms, which can be substituted with a or more radicals R 1 , or a group N(Ar')2.
- the radical R e preferably the substituent R e
- the radical R e is selected identically or differently on each occurrence from the group consisting of H or an aromatic or heteroaromatic ring system having 6 to 24 aromatic ring atoms, preferably having 6 to 18 aromatic ring atoms, particularly preferably having 6 to 13 aromatic ring atoms, which may each be substituted by one or more radicals R 1 .
- At least one radical R c , R d and/or R e preferably at least one radical R e , represents an aromatic or heteroaromatic ring system having 5 to 13 aromatic ring atoms, which can be substituted by one or more radicals R 1 .
- At least one radical preferably a substituent R c , R d and/or R e , particularly preferably at least one radical R e , is selected from phenyl, biphenyl, terphenyl, quaterphenyl, fluorene, spirobifluorene, naphthalene, indole, benzofuran, benzothiophene, carbazole, dibenzofuran, dibenzothiophene, indenocarbazole, indolocarbazole, pyridine, pyrimidine, pyrazine, pyridazine, triazine, quinoline, isoquinoline, quinazoline, quinoxaline, phenanthrene or triphenylene, which can each be substituted by one or more radicals R 1 .
- substituent means in particular that R c , R d and/or R e are not H, preferably not H and not D. Furthermore, the substituents R c , R d and/or R e may be the same or different if two or more substituents are present which are selected from the above-mentioned aromatic or heteroaromatic groups.
- Preferred aromatic or heteroaromatic ring systems which are represented by the radicals R, R a , R b , R c , R d and R e or Ar or Ar', are selected from phenyl, biphenyl, in particular ortho-, meta- or para-biphenyl, terphenyl, in particular ortho-, meta-, para- or branched terphenyl, quaterphenyl, in particular ortho-, meta-, para- or branched quaterphenyl, fluorene, which can be linked via the 1-, 2-, 3- or 4- position, spirobifluorene, which can be linked via the 1-, 2-, 3- or 4-position, naphthalene, in particular 1- or -linked naphthalene, indole, benzofuran, benzothiophene, carbazole, which can be linked via the 1-, 2-, 3- or 4-position, dibenzofuran, which can be linked via the 1-, 2-, 3- or 4-position, di
- Particularly preferred aromatic or heteroaromatic ring systems which can be represented by the radicals R, R a , R b , R c , R d and R e or Ar or Ar' are the structures (Ar-1 ) to (Ar-76) listed above, with structures of the formulae (Ar-1 ), (Ar-2), (Ar-3), (Ar-12), (Ar-13), (Ar-14), (Ar- 15), (Ar-16), (Ar-69), (Ar-70), (Ar-76) being preferred and structures of the formulas (Ar-1 ), (Ar-2), (Ar-3), (Ar-12), (Ar-13), (Ar-14), (Ar-15), (Ar-16) being particularly preferred.
- R, R a , R b , R c , R d and R e are groups of the formula -Ar 4 -N(Ar 2 )(Ar 3 ), where Ar 2 , Ar 3 and Ar 4 , identical or different on each occurrence, represent an aromatic or heteroaromatic ring system having 5 to 24 aromatic ring atoms, each of which may be substituted by one or more radicals R 1 .
- the total number of aromatic ring atoms of Ar 2 , Ar 3 and Ar 4 is a maximum of 60 and preferably a maximum of 40.
- Ar 4 and Ar 2 can be connected to one another and/or Ar 2 and Ar 3 can also be connected to one another by a single bond or a group selected from C(R 1 )2, NR 1 , O or S.
- Ar 4 and Ar 2 are connected to one another or Ar 2 and Ar 3 are connected to one another ortho to the position of the connection to the nitrogen atom.
- none of the groups Ar 2 , Ar 3 or Ar 4 are connected to one another.
- Ar 4 is an aromatic or heteroaromatic ring system having 6 to 24 aromatic ring atoms, preferably having 6 to 12 aromatic ring atoms, each of which may be substituted by one or more radicals R 1 .
- Ar 4 is selected from the group consisting of ortho-, meta- or para-phenylene or ortho-, meta- or para-biphenyl, each of which may be substituted by one or more radicals R 1 , but is preferably unsubstituted. Most preferably, Ar 4 is an unsubstituted phenylene group.
- Ar 2 and Ar 3 are the same or different on each occurrence and are an aromatic or heteroaromatic ring system having 6 to 24 aromatic ring atoms, which may each be substituted by one or more radicals R 1 .
- Particularly preferred groups Ar 2 and Ar 3 are the same or different on each occurrence and are selected from the group consisting of benzene, ortho-, meta- or para-biphenyl, ortho-, meta-, para- or branched terphenyl, ortho-, meta-, para- or branched quaterphenyl, 1-, 2-, 3- or 4-fluorenyl, 1-, 2-, 3- or 4-spiro-bifluorenyl, 1- or 2-naphthyl, indole, benzofuran, benzothiophene, 1-, 2-
- Ar 2 and Ar 3 are very particularly preferably selected, identically or differently on each occurrence, from the group consisting of benzene, biphenyl, in particular ortho-, meta- or para-biphenyl, terphenyl, in particular ortho-, meta-, para- or branched terphenyl, quaterphenyl, in particular ortho-, meta-, para- or branched quaterphenyl, fluorene, in particular 1-, 2-, 3- or 4-fluorene, or spirobifluorene, in particular 1-, 2-, 3- or 4-spirobifluorene.
- the preferences set out for the radicals R e also apply to the radicals R a , R b , R c and R d .
- R 1 is the same or different on each occurrence and is selected from the group consisting of H, D, F, CN, a straight-chain alkyl group having 1 to 10 C atoms or a branched or cyclic alkyl group having 3 to 10 C atoms, where the alkyl group may be substituted in each case by one or more radicals R 2 , or an aromatic or heteroaromatic ring system having 6 to 24 aromatic ring atoms, which may be substituted in each case by one or more radicals R 2 .
- R 1 is the same or different on each occurrence and is selected from the group consisting of H, a straight-chain alkyl group having 1 to 6 C atoms, in particular having 1, 2, 3 or 4 C atoms, or a branched or cyclic alkyl group having 3 to 6 C atoms, where the alkyl group may be substituted by one or more radicals R 2 , but is preferably unsubstituted, or an aromatic or heteroaromatic ring system having 6 to 13 aromatic ring atoms, each of which may be substituted by one or more radicals R 2 , but is preferably unsubstituted.
- R 2 is the same or different on each occurrence and is H, an alkyl group having 1 to 4 C atoms or an aryl group having 6 to 10 C atoms, which may be substituted by an alkyl group having 1 to 4 C atoms, but is preferably unsubstituted.
- the alkyl groups preferably have no more than five C atoms, particularly preferably no more than 4 C atoms, very particularly preferably no more than 1 C atom.
- compounds which are substituted with alkyl groups, in particular branched alkyl groups, with up to 10 C atoms or which are substituted with oligoarylene groups, for example ortho-, meta-, para- or branched terphenyl or quaterphenyl groups, are also suitable.
- the structures/compounds according to the invention have a high degree of deuteration.
- the degree of deuteration can be at least 50%, preferably at least 80%, especially preferably at least 90% and very particularly preferably at least 95%.
- the degree of deuteration is determined from the numerical ratio of deuterium to the sum of deuterium and 1 H hydrogen (D/(D+H)*100).
- the compounds are particularly preferably fully deuterated.
- the compounds according to the invention are particularly suitable for use in blue-emitting electroluminescent devices. Depending on the layer, these require materials with a high triplet level. However, many substituents with condensed aromatic or heteroaromatic groups can lead to a reduction in the triplet level.
- naphthyl structures are preferred over anthracene structures.
- fluorenyl, spirobifluorenyl, dibenzofuranyl and/or dibenzothienyl structures are preferred over naphthyl structures.
- radical Ar or R does not comprise an anthracene group, preferably none of the radicals Ar, R, R a , R b , R c , R d , R e comprises an anthracene group.
- the radical Ar or R does not comprise an aromatic or heteroaromatic ring system which has three linearly condensed aromatic 6 rings, wherein preferably none of the radicals Ar, R, R a , R b , R c , R d , R e comprises an aromatic or heteroaromatic ring system which has three linearly condensed aromatic 6 rings.
- the compound according to the invention is substituted by aromatic or heteroaromatic groups R a , R b , R c , R d , R e , R 1 or R 2 , it is preferred if these do not contain any aryl or heteroaryl groups with more than two aromatic six-membered rings fused directly to one another.
- the Substituents have no aryl or heteroaryl groups with six-membered rings directly fused to one another. This preference is due to the low triplet energy of such structures.
- Condensed aryl groups with more than two aromatic six-membered rings directly fused to one another, which are nevertheless also very suitable for the invention, are phenanthrene and triphenylene, since these also have a high triplet level.
- none of the radicals Ar, R, R a , R b , R c , R d and R e preferably none of the radicals Ar, R, R a , R b , R c , R d , R e , R 1 and R 2 comprises or forms a fluorenone group.
- a fluorenone comprises a 5-membered ring with a CO group to which two aromatic 6-membered rings are condensed.
- the compounds of formula (I) or the preferred embodiments are used as matrix material for a phosphorescent emitter or in a layer that directly borders a phosphorescent layer, it is further preferred if the compound does not contain any condensed aryl or heteroaryl groups in which more than two six-membered rings are directly condensed to one another.
- An exception to this are phenanthrene and triphenylene, which can be preferred due to their high triplet energy despite the presence of condensed aromatic six-membered rings.
- the compound according to the invention has an energy of the lowest triplet state Ti of at least 2.7 eV, preferably at least 2.9 eV, particularly preferably at least 3.1 eV.
- Molecular orbitals in particular the highest occupied molecular orbital (HOMO) and the lowest unoccupied molecular orbital (LUMO), their energy levels and the energy of the lowest triplet state Ti or the lowest excited singlet state Si of the materials are determined using quantum chemical calculations.
- a geometry optimization is first carried out using the The method “Ground State/Semi-empirical/Default Spin/AM1 /Charge O/Spin Singlet” is used.
- An energy calculation is then carried out based on the optimized geometry.
- the method “TD-SCF/DFT/Default Spin/B3PW91” is used with the basis set "6-31 G(d)" (Charge 0, Spin Singlet).
- the energy calculation yields the HOMO energy level HEh or LUMO energy level LEh in Hartree units. From this, the HOMO and LUMO energy levels calibrated using cyclic voltammetry measurements are determined in electron volts as follows:
- these values are to be regarded as HOMO or LUMO energy levels of the materials.
- the lowest triplet state Ti is defined as the energy of the triplet state with the lowest energy resulting from the quantum chemical calculation described.
- the lowest excited singlet state Si is defined as the energy of the excited singlet state with the lowest energy resulting from the quantum chemical calculation described.
- the compound comprises exactly two or exactly three structures according to formula (I), (1-1) to (I-6), (11-1) to (II-26), (111-1) to (III-34) and/or (IV-1) to (IV-48).
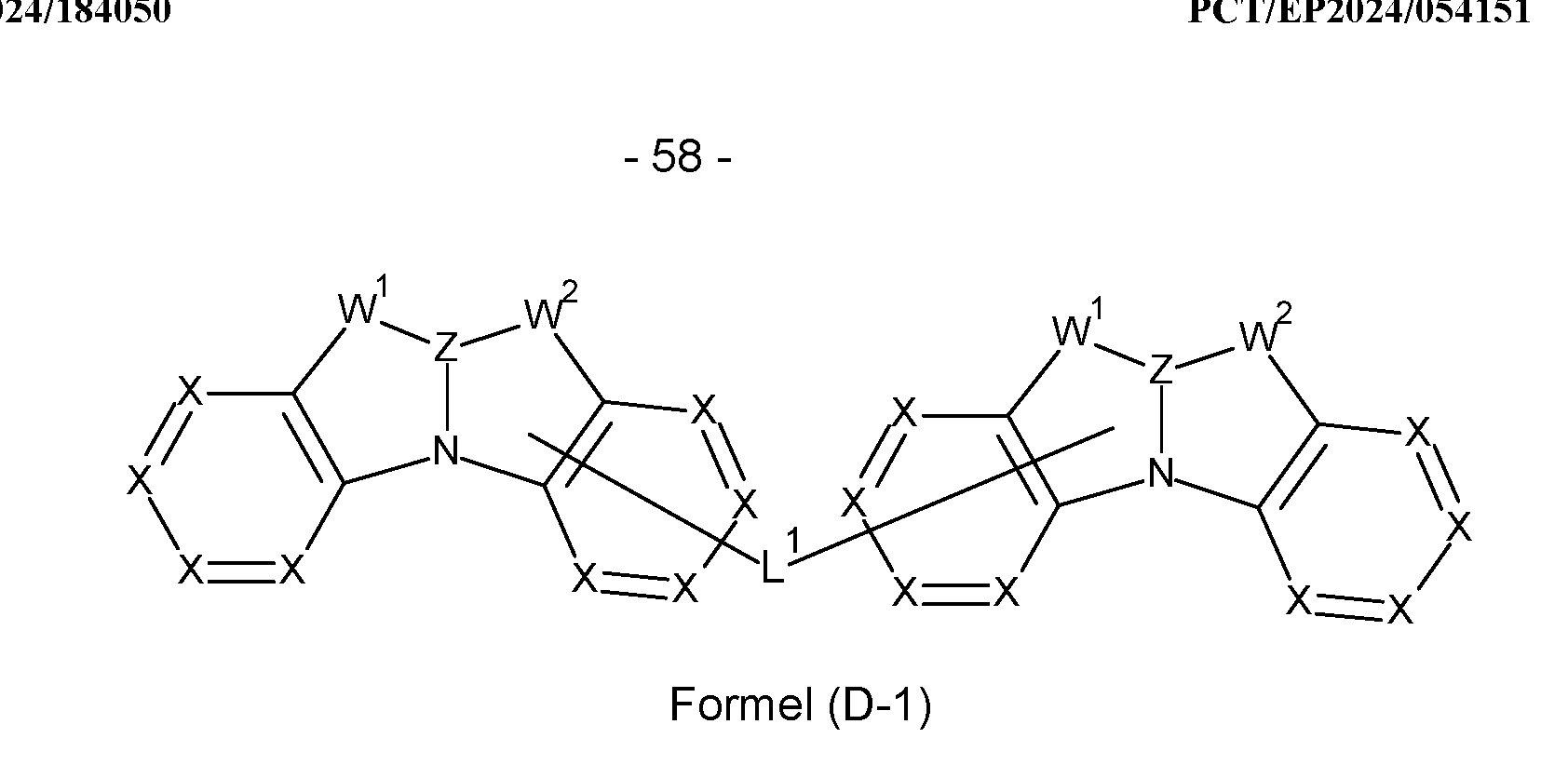
- the compounds are selected from compounds of the formula (D-1),
- group L 1 represents a connecting group, preferably a bond or an aromatic or heteroaromatic ring system having 5 to 40, preferably 5 to 30 aromatic ring atoms, which may be substituted by one or more radicals R e , and the other symbols and indices used have the meanings given in claim 1, where the group L 1 forms a bond to the basic structure instead of a hydrogen atom or a substituent, preferably the group L 1 binds to the radicals X.
- L 1 is a bond or an aromatic or heteroaromatic ring system having 5 to 14 aromatic or heteroaromatic ring atoms, preferably an aromatic ring system having 6 to 12 carbon atoms, which may be substituted by one or more radicals R e , but is preferably unsubstituted, where R e may have the meaning given above, in particular for formula (I).
- L 1 particularly preferably stands for an aromatic ring system having 6 to 10 aromatic ring atoms or a heteroaromatic ring system having 6 to 13 heteroaromatic ring atoms, each of which may be substituted by one or more radicals R 1 , but is preferably unsubstituted, where R 1 may have the meaning given above, in particular for formula (I).
- the symbol L 1 set out, inter alia, in formula (D1 ) preferably represents, identically or differently on each occurrence, a bond or an aryl or heteroaryl radical having 5 to 24 ring atoms, preferably 6 to 13 ring atoms, particularly preferably 6 to 10 ring atoms, so that an aromatic or heteroaromatic group of an aromatic or heteroaromatic ring system is directly bonded, i.e. via an atom of the aromatic or heteroaromatic group, to the respective atom of the other group.
- the group L 1 shown in formula (D1) comprises an aromatic ring system with at most four, preferably at most three, particularly preferably at most two fused aromatic and/or heteroaromatic 6-membered rings, preferably no fused aromatic or heteroaromatic ring system.
- Suitable aromatic or heteroaromatic ring systems L 1 are selected from the group consisting of ortho-, meta- or para-phenylene, ortho-, meta- or para-biphenylene, terphenylene, in particular branched terphenylene, quaterphenylene, in particular branched quaterphenylene, fluorenylene, spirobifluorenylene, dibenzofuranylene, dibenzothienylene and carbazolylene, which may each be substituted by one or more radicals R 1 , but are preferably unsubstituted.
- a compound according to the invention can be represented by at least one of the structures according to formulas (I), (1-1) to (I-6), (11-1) to (II-26), (111-1) to (III-34) and/or (IV-1) to (IV-48).
- compounds according to the invention preferably comprising structures according to formulas (I), (1-1) to (I-6), (11-1) to (II-26), (111-1) to (III-34) and/or (IV-1) to (IV-48) have a molecular weight of less than or equal to 5000 g/mol, preferably less than or equal to 4000 g/mol, particularly preferably less than or equal to 3000 g/mol, especially preferably less than or equal to 2000 g/mol, more especially preferably less than or equal to 1200 g/mol and very particularly preferably less than or equal to 900 g/mol.
- preferred compounds according to the invention are characterized in that they are sublimable. These compounds generally have a molecular weight of less than about 1200 g/mol. Furthermore, it can be provided that the compound comprising structures according to formula (I), preferably the compound according to formula (I) or a preferred embodiment of this structure/compound is not in direct contact with a metal atom, preferably does not represent a ligand for a metal complex.
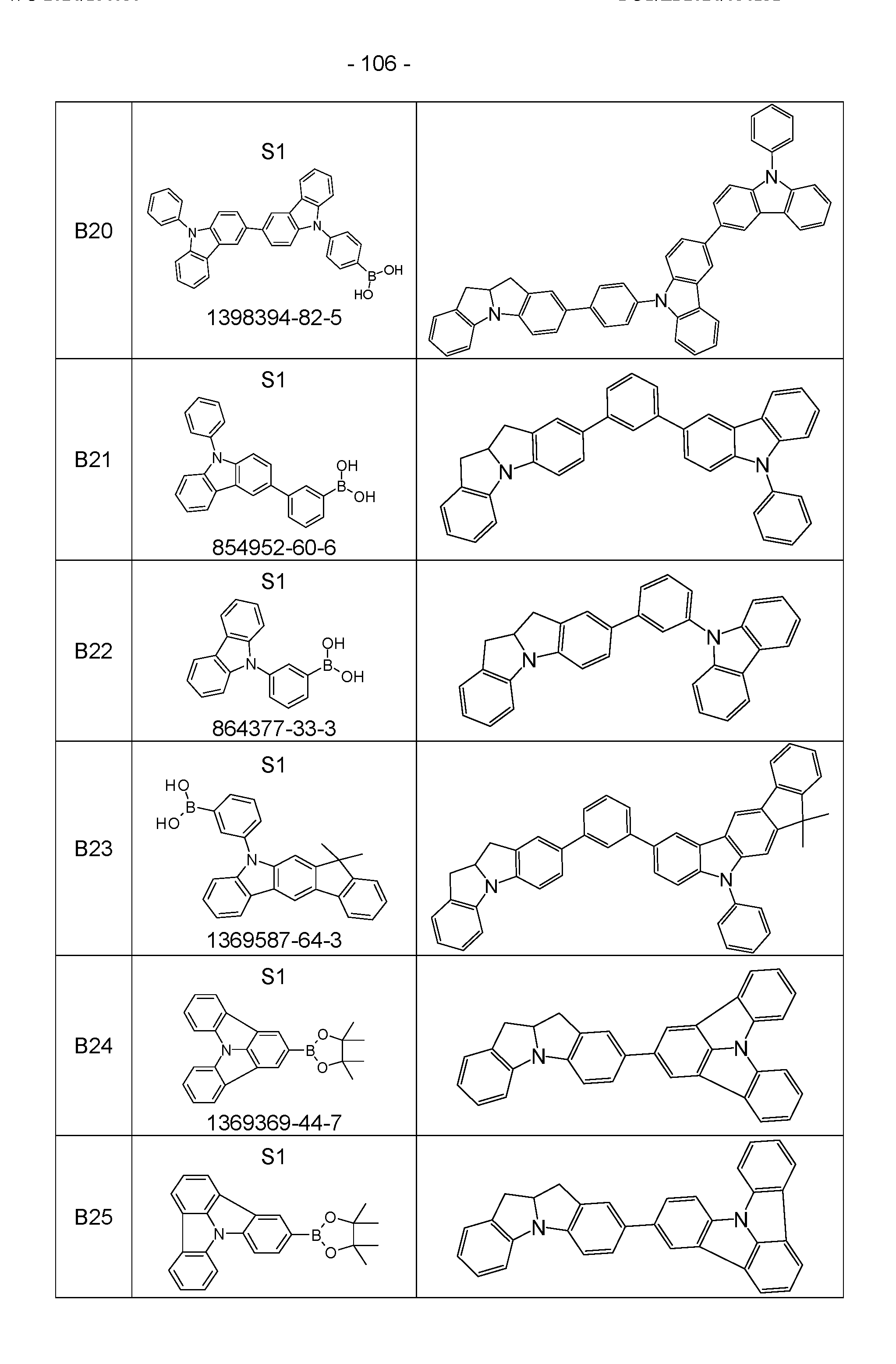
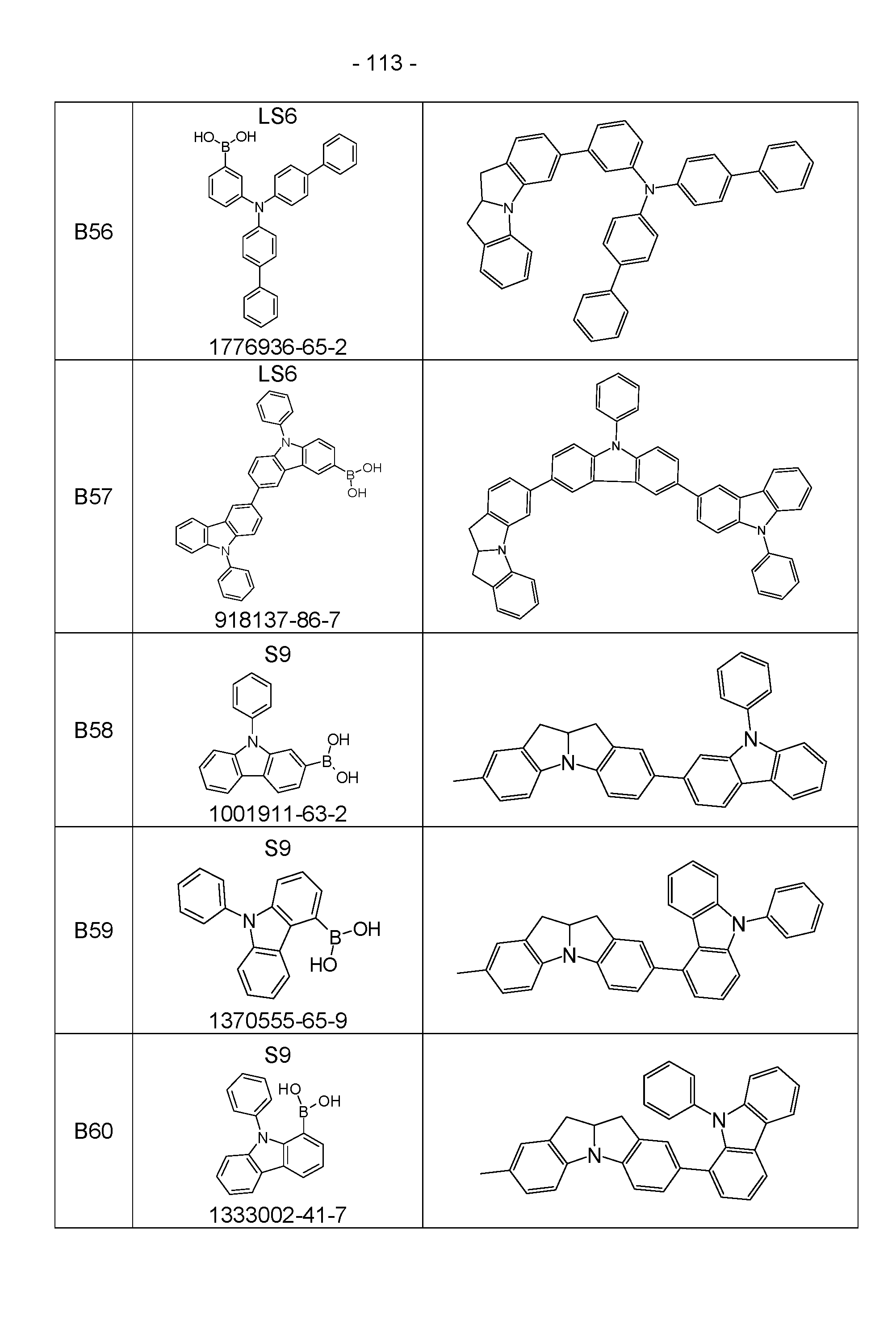
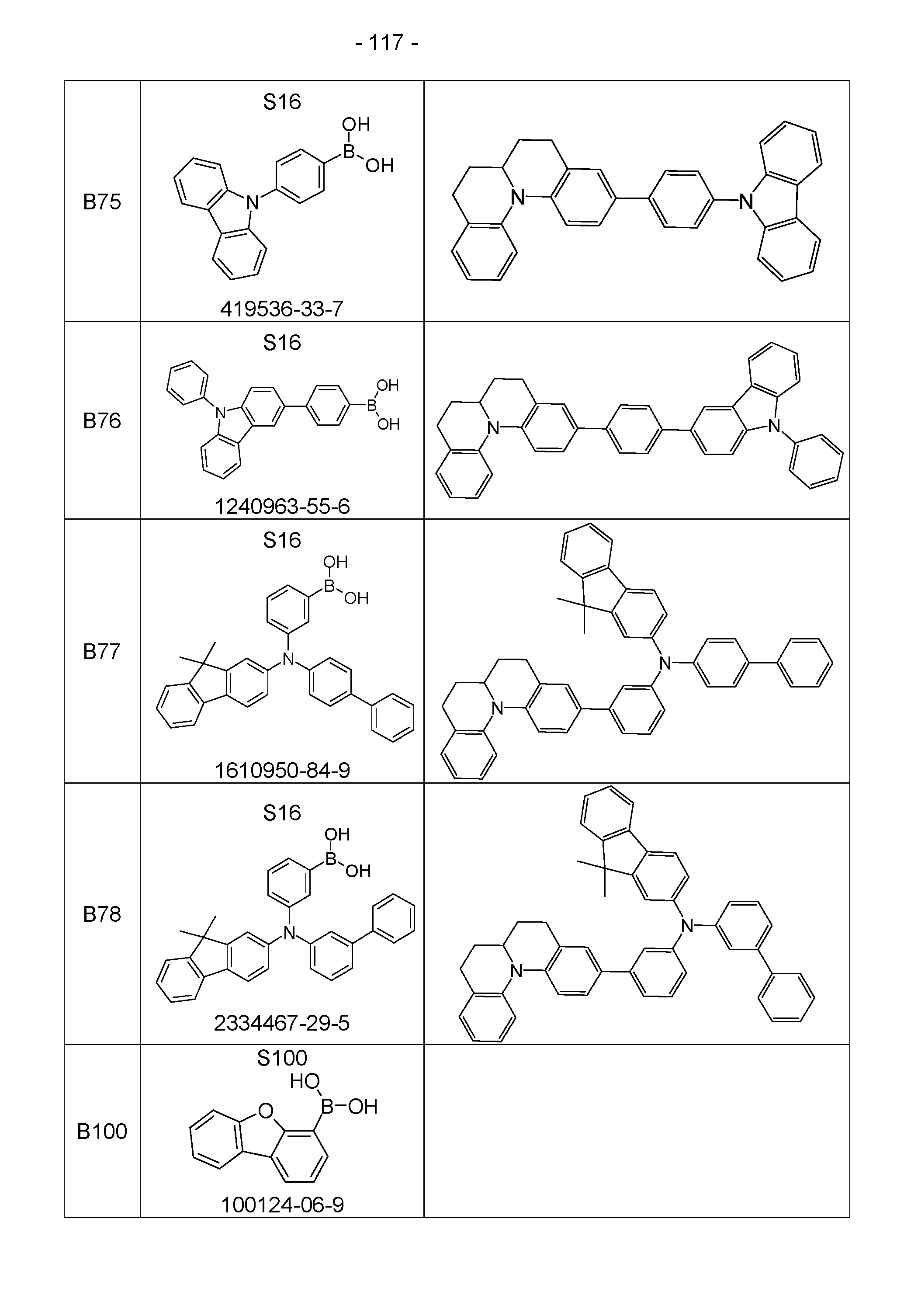
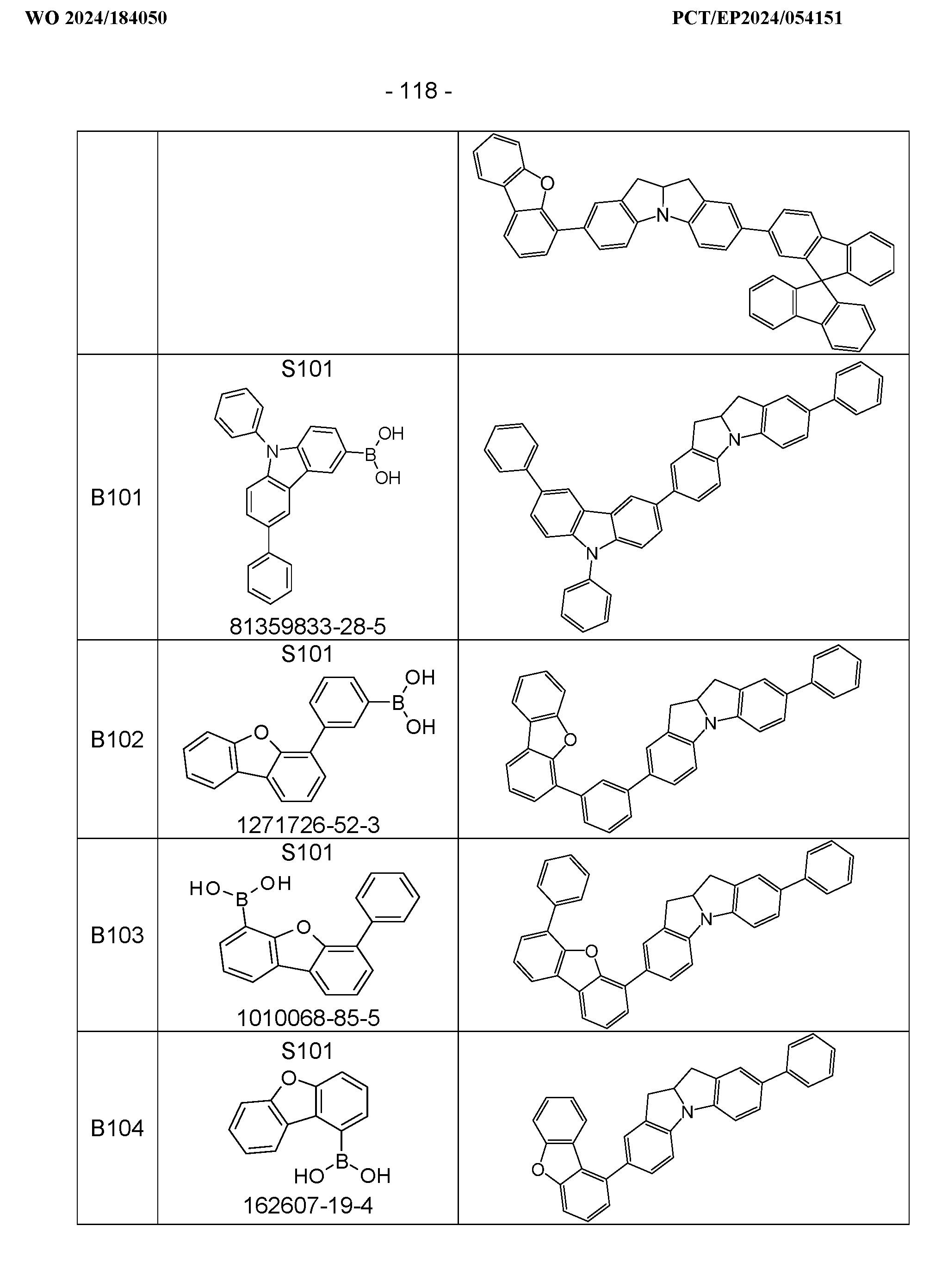
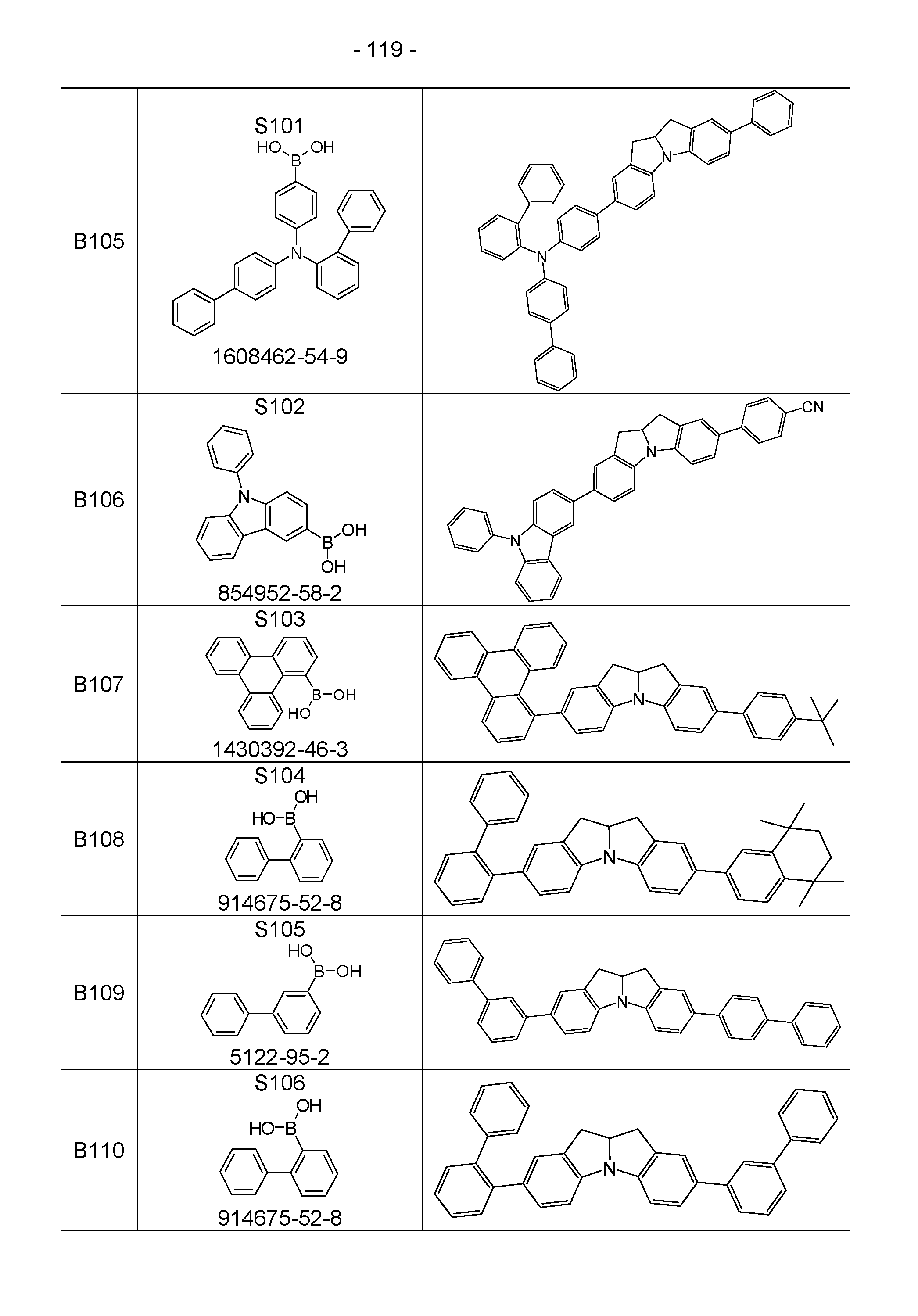
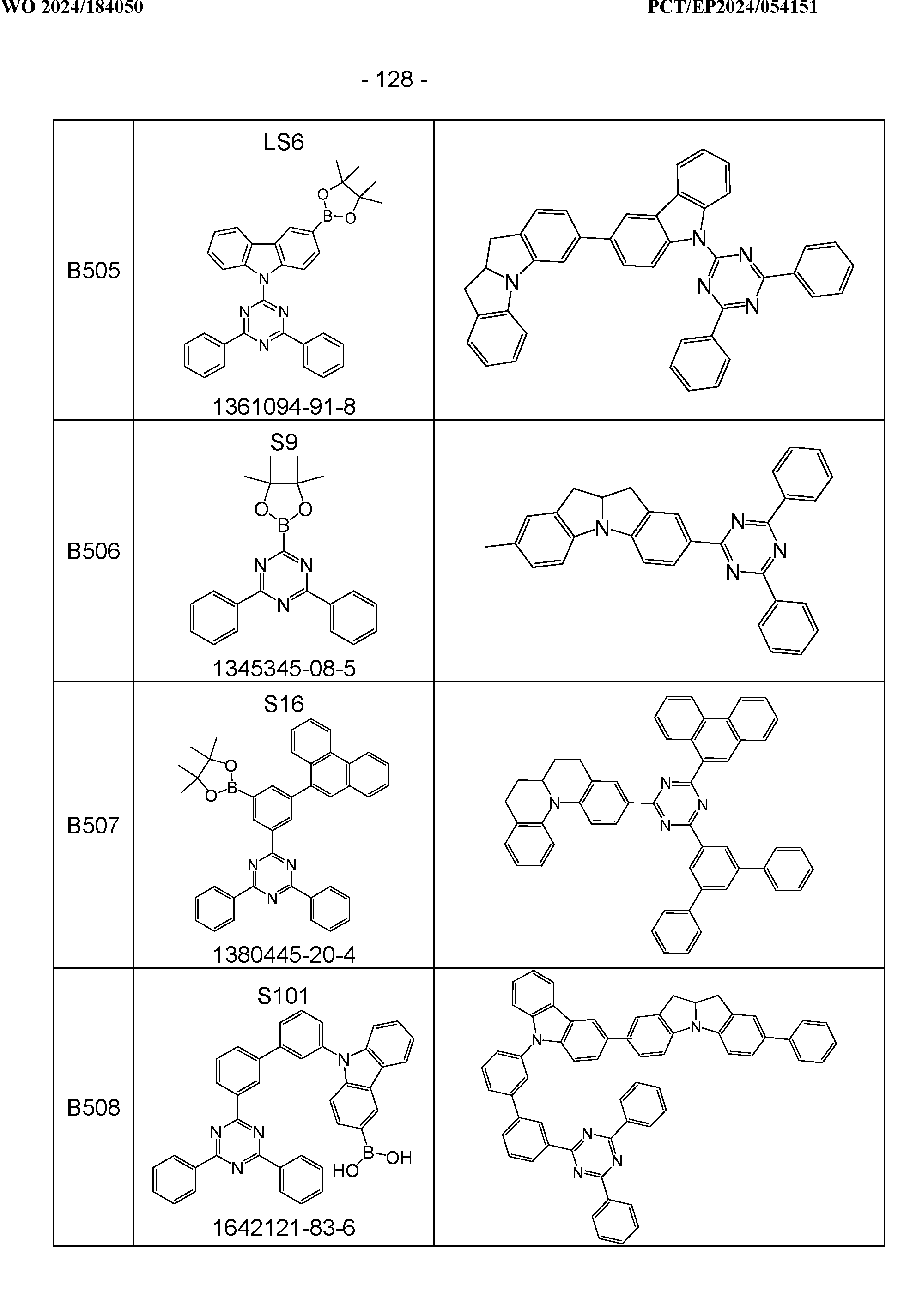
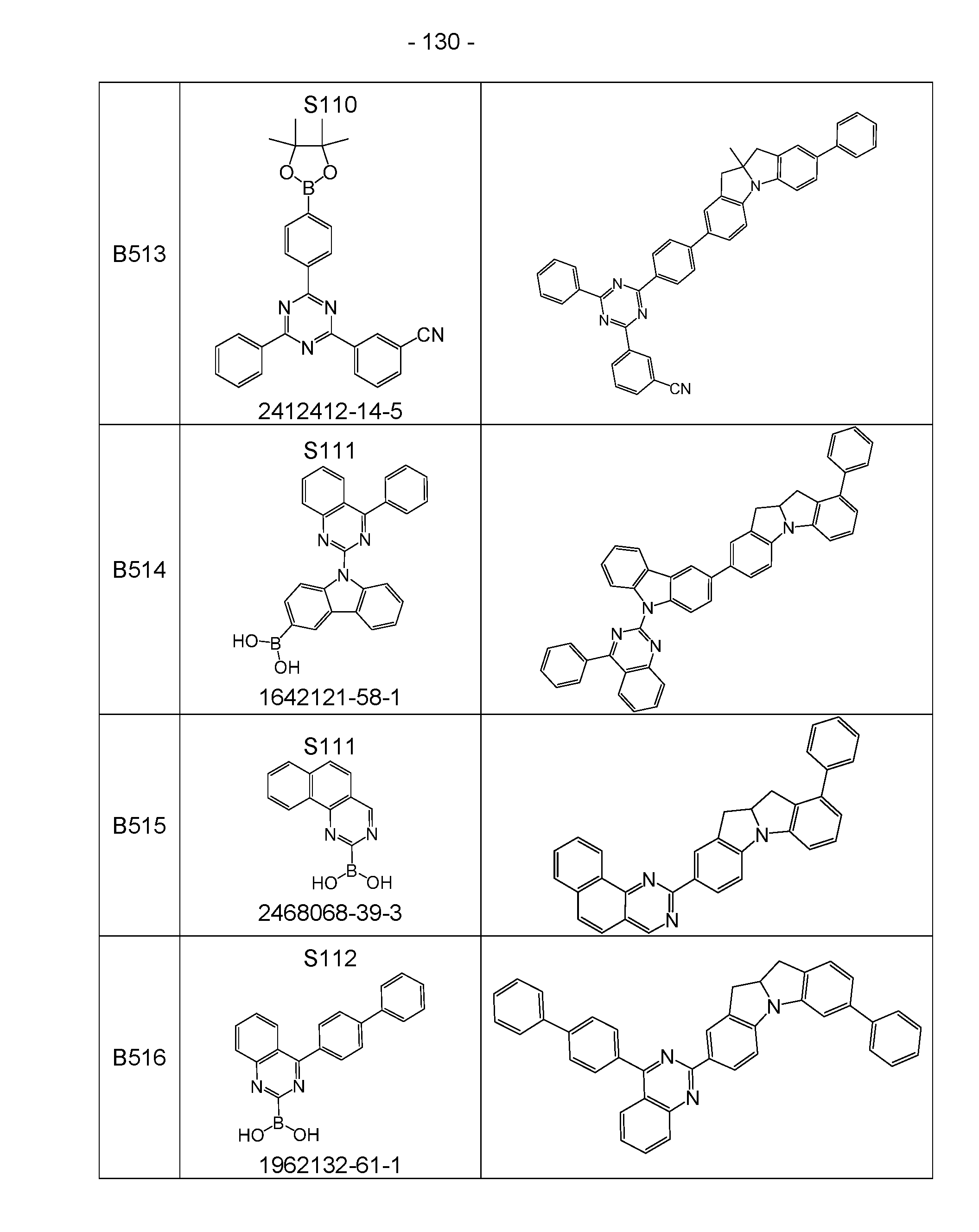
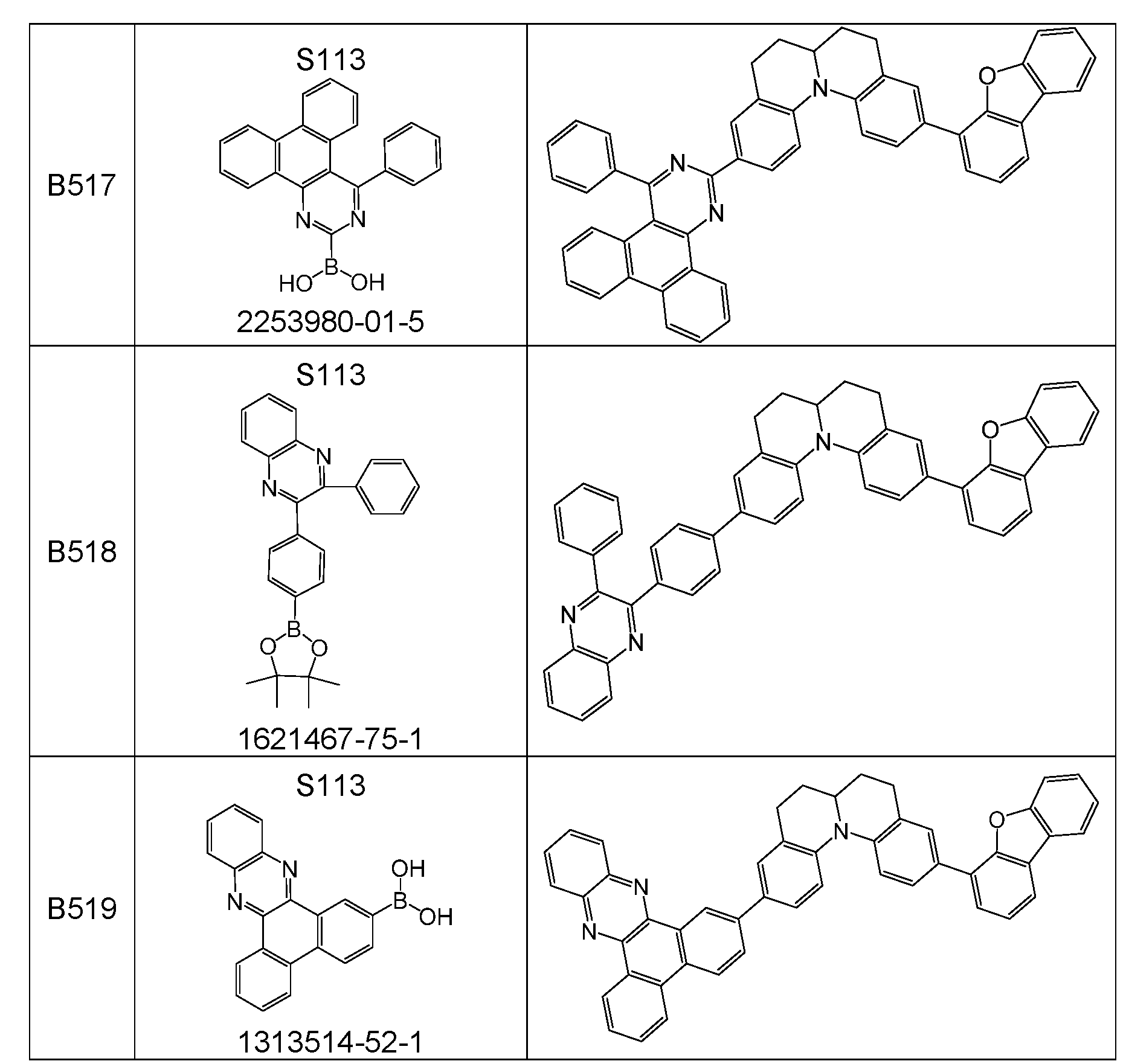
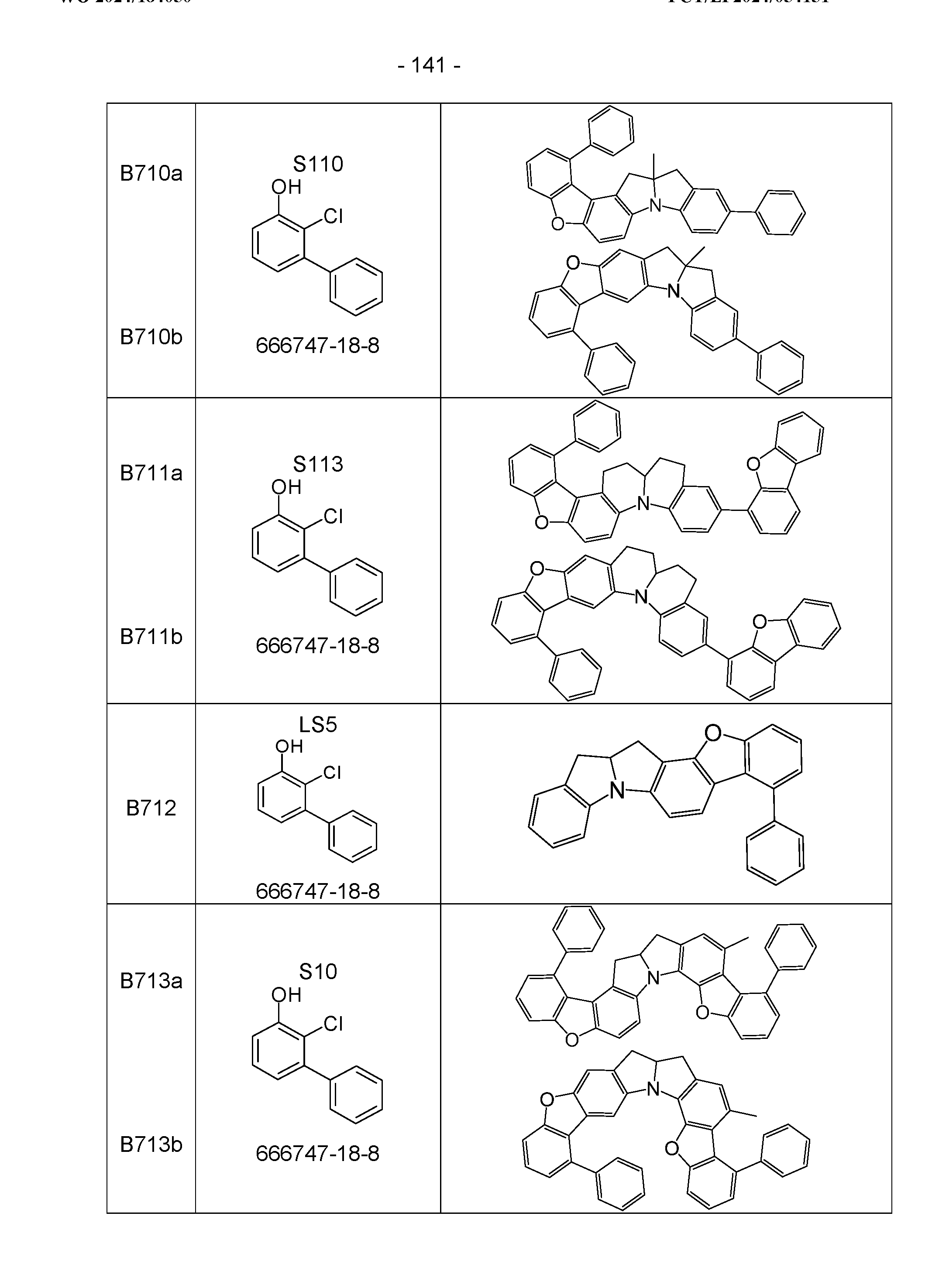
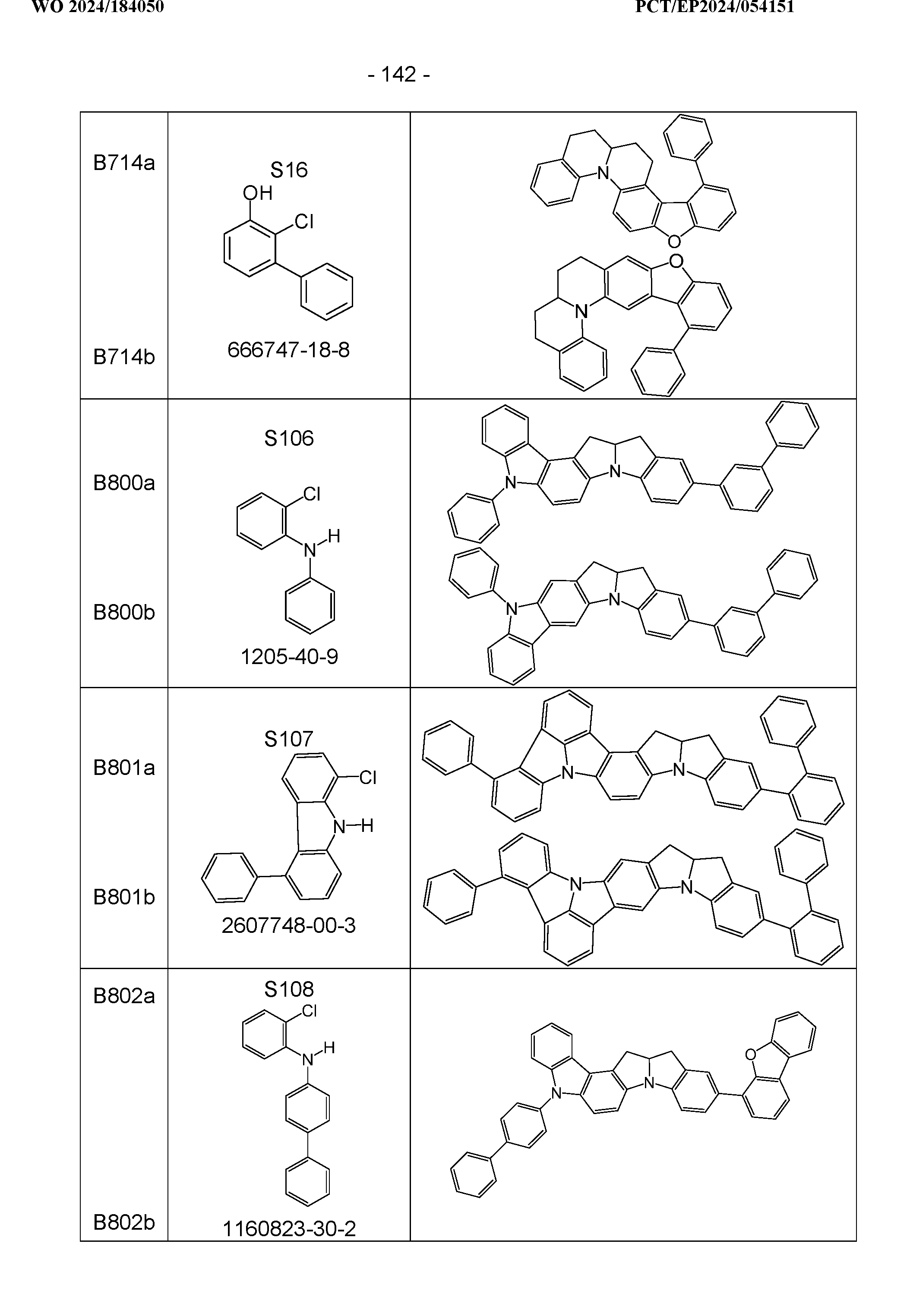
- the above-mentioned preferred embodiments can be combined with one another as desired within the restrictions defined in claim 1. In a particularly preferred embodiment of the invention, the above-mentioned preferences occur simultaneously. Examples of preferred compounds according to the above-mentioned embodiments are the compounds listed in the following table.
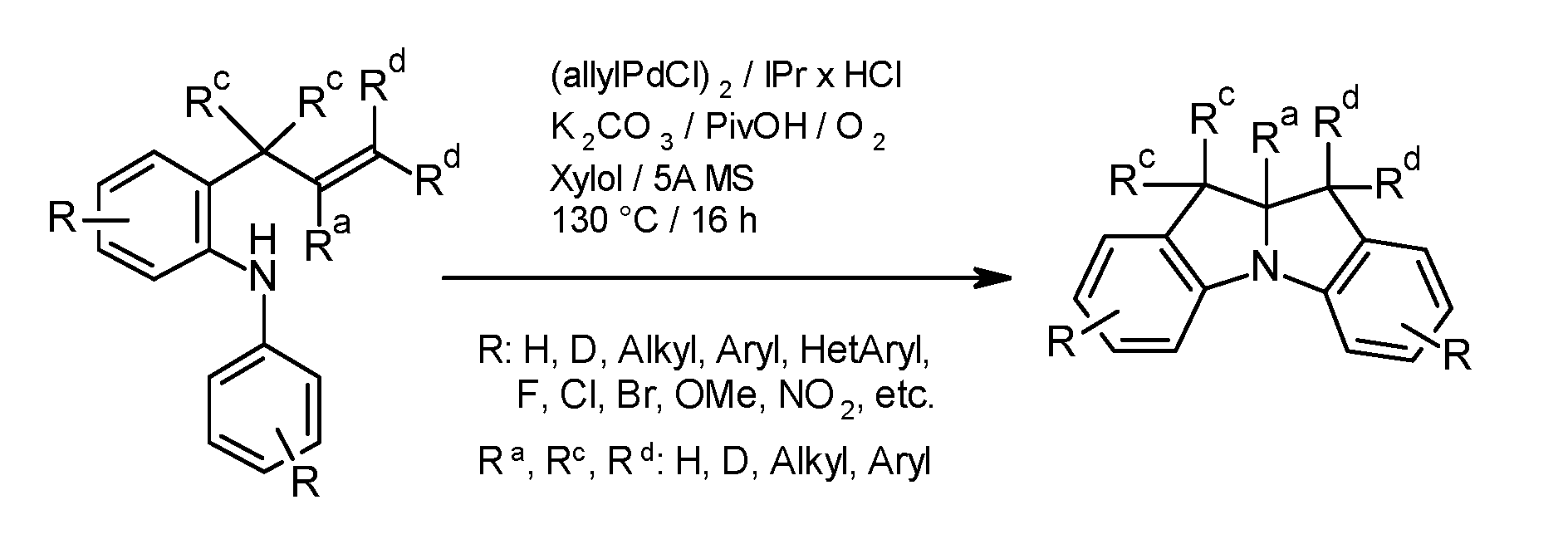
- the basic structure of the compounds according to the invention can be represented by the methods outlined in the following schemes.
- the individual synthesis steps such as coupling reactions that lead to C-C linkages and/or C-N linkages, are known in principle to the person skilled in the art. These include reactions according to BUCHWALD, SUZUKI, YAMAMOTO, STILLE, HECK, NEGISHI, SONOGASHIRA and HIYAMA. Further information on the synthesis of the compounds according to the invention can be found in the synthesis examples.
- the preparation of compounds according to the invention in particular of unsubstituted and aliphatically or aromatically substituted 10a, 11-dihydro-10H-indolo[1,2-a]indoles, can be carried out in accordance with processes described in detail in the literature (see, for example, ES Sherman et al., Adv. Synth. Catal. 2009, 351, 467 and W. Du et al., J. Am. Chem. Soc.
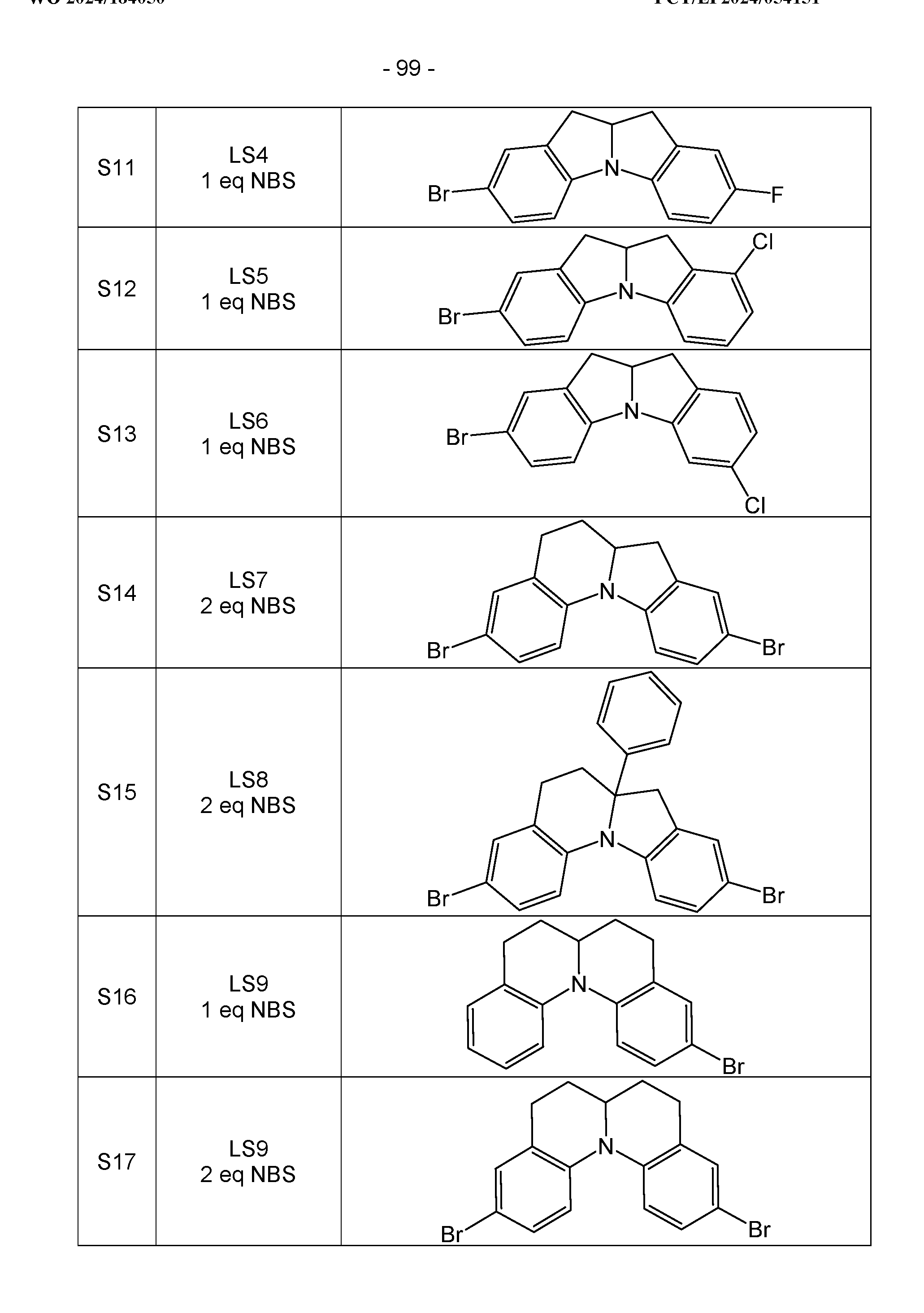
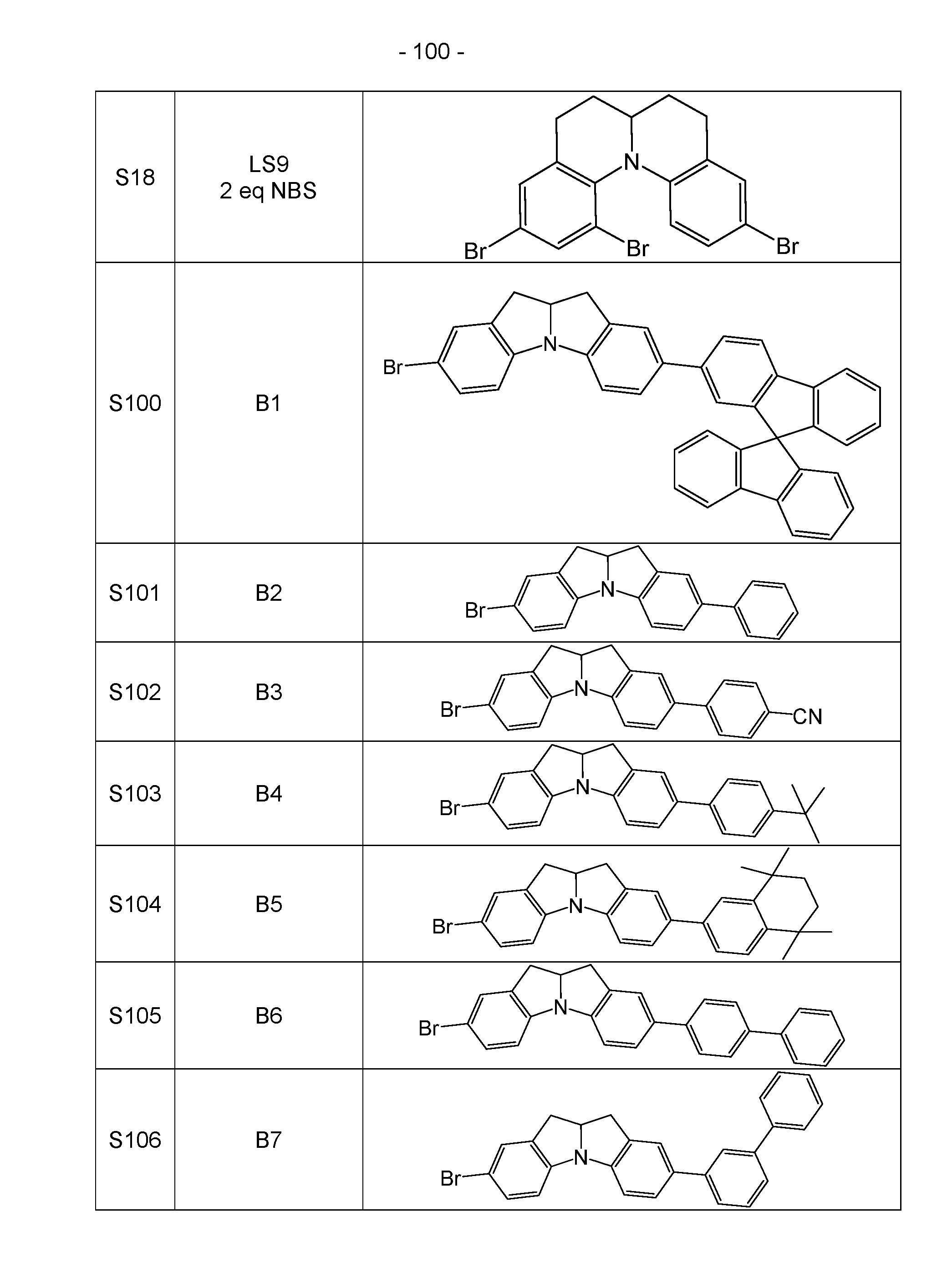
- halogen compounds obtained in this way can be further functionalized using common methods by CC coupling (Suzuki, Negishi, Sonogashira, Grignard-Cross, etc.) or CN coupling (Buchwald-Hartwig, Ullmann, etc.), as shown for the monobromide in Scheme 3.
- the di-, tri- and tetrahalides can be reacted analogously.
- the group Ar/HetAr stands for an aromatic or heteroaromatic ring system.
- CN coupling of the Buchwald-Hartwig type The groups Ar 1 /Ar 2 represent an aromatic or heteroaromatic ring system. Reactive leaving groups or their precursors, e.g.
- methoxy groups can be used in the synthesis of 10a,11-dihydro-10H-indolo[1,2-a]indole
- the mixture of regioisomers obtained during cyclization can be separated using conventional methods (chromatography, fractional crystallization) (see ES Sherman et al., Adv. Synth. Catal.2009, 351, 467).
- the methoxy groups can then be saponified using conventional methods, e.g. in the pyridinium hydrochloride melt, and the resulting bisphenols can be esterified with trifluoromethanesulfonic anhydride.
- the 10a,11-dihydro-1 Oa-sila-10H-indolo[1,2-a]indoles can be prepared starting from bis(2-alkylphenyl)amines and primary alkyl- or aryl-silanes according to Q. Li et al., Angew. Chem. Int. Ed. 2014, 53, 8471, see Scheme 7.
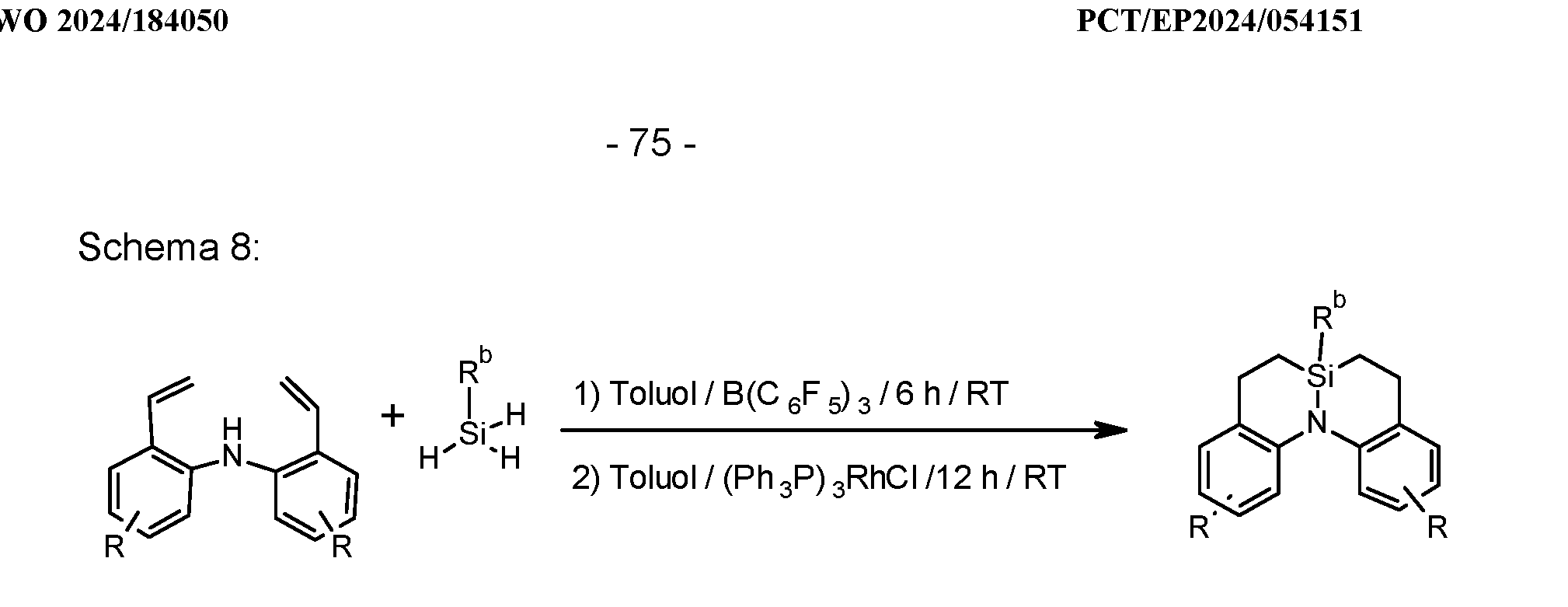
- the 6,6a,7,8-tetrahydro-5-sila-5H-dibenzo[c,f]quinolizines can be prepared starting from bis(2-alkenylphenyl)amines and primary alkyl- or arylsilanes according to Z.-Y. Liu et al, Angew. Chem. Int. Ed. 2017, 56, 5817 and H. Nagashima et al, Organometallics, 1989, 8, 10, see Scheme 8.
- Germanium compounds can be obtained in particular according to the processes shown in Schemes 7 and 8.
- a further object of the present invention is therefore a process for preparing a compound according to the invention, wherein a heterocyclic basic structure with an amino group is synthesized and at least one aromatic or heteroaromatic radical is introduced, preferably by means of a nucleophilic aromatic substitution reaction or a coupling reaction.
- the compounds according to the invention can be obtained in high purity, preferably more than 99% (determined by 1 H-NMR and/or HPLC).
- the compounds according to the invention can also be mixed with a polymer. It is also possible to incorporate these compounds covalently into a polymer. This is particularly possible with compounds which are substituted with reactive leaving groups, such as bromine, iodine, chlorine, boronic acid or boronic acid esters, or with reactive, polymerizable groups, such as olefins or oxetanes. These can be used as monomers to produce corresponding oligomers, dendrimers or polymers. The oligomerization or polymerization preferably takes place via the halogen functionality or the boronic acid functionality. or via the polymerizable group. It is also possible to crosslink the polymers via such groups. The compounds and polymers according to the invention can be used as a crosslinked or uncrosslinked layer.
- reactive leaving groups such as bromine, iodine, chlorine, boronic acid or boronic acid esters
- reactive, polymerizable groups such as olefins or oxetanes.
- the invention therefore further relates to oligomers, polymers or dendrimers containing one or more of the above-listed structures of the formula (I) and preferred embodiments of this formula or compounds according to the invention, where one or more bonds of the compounds according to the invention or the structures of the formula (I) and preferred embodiments of this formula to the polymer, oligomer or dendrimer are present.
- these therefore form a side chain of the oligomer or polymer or are linked in the main chain.
- the polymers, oligomers or dendrimers can be conjugated, partially conjugated or non-conjugated.
- the oligomers or polymers can be linear, branched or dendritic. The same preferences apply to the repeat units of the compounds according to the invention in oligomers, dendrimers and polymers as described above.
- the monomers according to the invention are homopolymerized or copolymerized with other monomers. Preference is given to copolymers in which the units according to formula (I) or the preferred embodiments set out above and below are present in amounts of 0.01 to 99.9 mol%, preferably 5 to 90 mol%, particularly preferably 20 to 80 mol%.
- Suitable and preferred comonomers which form the polymer backbone are selected from fluorenes (e.g. according to EP 842208 or WO 2000/022026), spirobifluorenes (e.g. according to EP 707020, EP 894107 or WO 2006/061181), para-phenylenes (e.g.
- WO 92/18552 carbazoles (e.g. according to WO 2004/070772 or WO 2004/113468), thiophenes (e.g. according to EP 1028136), dihydrophenanthrenes (e.g. according to WO 2005/014689), cis- and trans-indenofluorenes (e.g. according to WO 2004/041901 or WO 2004/113412), ketones (e.g. according to WO 2005/040302), phenanthrenes (e.g. according to WO 2005/104264 or WO 2007/017066) or several of these units.
- the polymers, oligomers and dendrimers can contain further units, for example hole transport units, in particular those based on triarylamines, and/or electron transport units.
- compounds according to the invention which are characterized by a high glass transition temperature.
- compounds according to the invention are particularly preferred which comprise structures according to the formula (I) or the preferred embodiments set out above and below which have a glass transition temperature of at least 70 °C, particularly preferably of at least 110 °C, very particularly preferably of at least 125 °C and especially preferably of at least 150 °C, determined according to DIN 51005 (version 2005-08).
- formulations of the compounds according to the invention are required. These formulations can be, for example, solutions, dispersions or emulsions. It may be preferable to use mixtures of two or more solvents for this purpose.
- Suitable and preferred solvents are, for example, toluene, anisole, o-, m- or p-xylene, methyl benzoate, mesitylene, tetralin, veratrole, THF, methyl-THF, THP, chlorobenzene, dioxane, phenoxytoluene, in particular 3-phenoxytoluene, (-)-fenchone, 1,2,3,5-tetramethylbenzene, 1,2,4,5-tetramethylbenzene, 1-methylnaphthalene, 2-methylbenzothiazole, 2-phenoxyethanol, 2-pyrrolidinone, 3-methylanisole, 4-methylanisole, 3,4-dimethylanisole, 3,5-dimethylanisole, acetophenone, a-terpineol, benzothiazole, butylbenzoate, cumene, cyclohexanol, cyclohexanone, Cyclohexylbenzene, decalin, do
- the present invention therefore further relates to a formulation or a composition comprising at least one compound according to the invention and at least one further compound.
- the further compound can be, for example, a solvent, in particular one of the abovementioned solvents or a mixture of these solvents. If the further compound comprises a solvent, this mixture is referred to herein as a formulation.
- the further compound can also be at least one further organic or inorganic compound which is also used in the electronic device, for example an emitter and/or a matrix material, whereby these compounds differ from the compounds according to the invention. Suitable emitters and matrix materials are listed below in connection with the organic electroluminescent device.
- the further compound can also be polymeric.
- compositions comprising at least one compound of formula (I) where the symbols used have the meanings given above, where the structure of formula (I) does not comprise a six-membered ring in which a boron atom is arranged in the para position to a nitrogen atom, preferably at least one inventive Compound or an oligomer, polymer or dendrimer comprising structures according to formula (I) and at least one further organic functional material.
- Functional materials are generally the organic or inorganic materials which are introduced between the anode and the cathode.
- the organic functional material is selected from the group consisting of fluorescent emitters, phosphorescent emitters, emitters which exhibit TADF (thermally activated delayed fluorescence), host materials, electron transport materials, electron injection materials, hole conductor materials, hole injection materials, electron blocking materials and hole blocking materials, preferably host materials.
- TADF thermally activated delayed fluorescence
- Another object of the present invention is the use of a compound according to formula (I) where the symbols used have the meanings given above, where the structure of formula (I) does not comprise a six-membered ring in which a boron atom is arranged in para position to a nitrogen atom, preferably a compound according to the invention or an oligomer, polymer or dendrimer comprising structures according to formula (I) in an electronic device, preferably in an organic electroluminescent device, preferably as host material, hole conductor material, hole injection material or electron blocking material.
- a compound according to formula (I), preferably a compound according to the invention is used as host material, hole conductor material, hole injection material or electron blocking material and that this compound according to the invention has at least one hole transport group, where preferred hole transport groups have been previously defined.
- a compound according to formula (I), preferably a compound according to the invention is used as host material and this compound according to the invention comprises both at least one hole transport group and at least one electron transport group, wherein preferred hole transport groups and/or electron transport groups have been defined previously.
- Yet another object of the present invention is an electronic device comprising at least one compound of formula (I) where the symbols used have the meanings given above, where the structure of formula (I) does not comprise a six-membered ring in which a boron atom is arranged in the para position to a nitrogen atom, preferably at least one compound according to the invention or an oligomer, polymer or dendrimer comprising structures according to formula (I).
- An electronic device in the sense of the present invention is a device which contains at least one layer which contains at least one organic compound. The component can also contain inorganic materials or layers which are made up entirely of inorganic materials.
- Particularly preferred electronic device is selected from the group consisting of organic electroluminescent devices (OLEDs, sOLED, PLEDs, LECs, etc.), preferably organic light-emitting diodes (OLEDs), organic light-emitting diodes based on small molecules (sOLEDs), organic light-emitting diodes based on polymers (PLEDs), light-emitting electrochemical cells (LECs), organic laser diodes (O-lasers), “organic plasmon emitting devices” (DM Koller et al., Nature Photonics 2008, 1- 4); organic integrated circuits (O-ICs), organic field-effect transistors (O-FETs), organic thin-film transistors (O-TFTs), organic light-emitting transistors (O-LETs), organic solar cells (O-SCs), organic optical detectors, organic photoreceptors, organic field quench devices (O-FQDs) and organic electrical sensors, preferably organic electroluminescent devices (OLEDs, sOL
- the organic electroluminescent device contains a cathode, an anode and at least one emitting layer. In addition to these layers, it can contain further layers, for example one or more hole injection layers, hole transport layers, hole blocking layers, electron transport layers, electron injection layers, exciton blocking layers, electron blocking layers and/or charge generation layers. Interlayers can also be introduced between two emitting layers, which, for example, have an exciton blocking function. It should be noted, however, that not all of these layers necessarily have to be present.
- the organic electroluminescent device can contain one emitting layer, or it can contain several emitting layers.
- the organic electroluminescent device according to the invention can also be a tandem electroluminescent device, in particular for white-emitting OLEDs.
- the compound according to formula (I), preferably the compound according to the invention, can be used in different layers, depending on the precise structure. Preference is given to an organic electroluminescent device containing a compound according to formula (I) or the preferred embodiments set out above in an emitting layer as host material for fluorescent emitters, phosphorescent emitters or for emitters that exhibit TADF (thermally activated delayed fluorescence), in particular for phosphorescent emitters. Furthermore, the compound according to the invention can also be used in a hole transport layer, hole injection layer and/or in an exciton blocking layer.
- the compound according to the invention is particularly preferably used as a matrix material for phosphorescent emitters, in particular for red, orange, green, yellow or blue phosphorescent emitters, in an emitting layer, as a hole transport or electron blocking material in a hole transport or electron blocking layer, as a hole injection material in a hole injection layer.
- phosphorescent emitters in particular for red, orange, green, yellow or blue phosphorescent emitters
- an emitting layer as a hole transport or electron blocking material in a hole transport or electron blocking layer, as a hole injection material in a hole injection layer.
- the compound according to the invention is used as a matrix material for a phosphorescent compound in an emitting layer, it is preferably used in combination with one or more phosphorescent materials (triplet emitters).
- Phosphorescence in the sense of this invention is understood to mean luminescence from an excited state with a higher spin multiplicity, i.e. a spin state > 1, in particular from an excited triplet state.
- a spin state > 1 in particular from an excited triplet state.
- all luminescent complexes with transition metals or lanthanides, in particular all Iridium, platinum and copper complexes are considered phosphorescent compounds.
- the mixture of the compound according to the invention and the emitting compound contains between 99 and 1 vol.%, preferably between 98 and 10 vol.%, particularly preferably between 97 and 60 vol.%, in particular between 95 and 80 vol.% of the compound according to the invention, based on the total mixture of emitter and matrix material. Accordingly, the mixture contains between 1 and 99 vol.%, preferably between 2 and 90 vol.%, particularly preferably between 3 and 40 vol.%, in particular between 5 and 20 vol.% of the emitter, based on the total mixture of emitter and matrix material.
- the compound according to the invention is used as the only matrix material (“single host”) for the phosphorescent emitter.
- a further embodiment of the present invention is the use of the compound according to the invention as a matrix material for a phosphorescent emitter in combination with a further matrix material.
- Suitable matrix materials which can be used in combination with the compounds according to the invention are aromatic ketones, aromatic phosphine oxides or aromatic sulfoxides or sulfones, e.g. according to WO 2004/013080, WO 2004/093207, WO 2006/005627 or WO 2010/006680, triarylamines, carbazole derivatives, e.g. B.
- CBP N,N-biscarbazolylbiphenyl
- CBP CBP (N,N-biscarbazolylbiphenyl) or those in WO 2005/039246, US 2005/0069729, JP 2004/288381, EP 1205527, WO 2008/086851 or WO 2013/041176, indolocarbazole derivatives, e.g. according to WO 2007/063754 or WO 2008/056746, indenocarbazole derivatives, e.g. according to WO 2010/136109, WO 2011/000455, WO 2013/041176 or WO 2013/056776, azacarbazole derivatives, e.g. B.
- EP 1617710, EP 1617711, EP 1731584, JP 2005/347160 bipolar matrix materials, e.g. according to WO 2007/137725, silanes, e.g. according to WO 2005/111172, azaboroles or boronate esters, e.g. according to WO 2006/117052, triazine derivatives, e.g. according to WO 2007/063754, WO 2008/056746, WO 2010/015306, WO 2011/057706, WO 2011/060859 or WO 2011/060877, zinc complexes, e.g.
- diazasilole or tetra-azasilole derivatives e.g. according to WO 2010/054729
- diazaphosphole derivatives e.g. according to WO 2010/054730
- bridged carbazole derivatives e.g. according to WO 2011/042107, WO 2011/060867, WO 2011/088877 and WO 2012/143080
- triphenylene derivatives e.g. according to WO 2012/048781
- dibenzofuran derivatives e.g. B. according to WO 2015/169412, WO 2016/015810, WO 2016/023608, WO 2017/148564 or WO 2017/148565 or biscarbazoles, e.g. according to JP 3139321 B2.
- a compound containing a structure/compound according to formula (I) or the preferred embodiments described above, which is used as host material is preferably used in combination with one or more phosphorescent materials (triplet emitters) and/or a compound which is a TADF host material (thermally activated delayed fluorescence).
- phosphorescent materials triplet emitters
- TADF host material thermalally activated delayed fluorescence
- a hyperfluorescence system as described in WO 2012/133188 and/or hyperphosphorescence system as in US 2017271611 is preferably formed. This combination represents a preferred composition according to the present invention.
- WO 2015/091716 A1 and WO 2016/193243 A1 disclose OLEDs that contain both a phosphorescent compound and a fluorescent emitter in the emission layer, with the energy being transferred from the phosphorescent compound to the fluorescent emitter (hyperphosphorescence).
- the phosphorescent compound therefore behaves like a host material.
- host materials have higher singlet and triplet energies compared to the emitters, so that the energy of the host material is transferred to the emitter as optimally as possible.
- the systems disclosed in the prior art have precisely such an energy relationship.
- Preferred emitters that can be used in combination with a compound according to the invention are described, inter alia, by Sungho Nam et al., Adv. Sci. 2021, 2100586 and Eungdo Kin et al., Sci. Adv. 2022, 8, eabq 1641.
- preferred triplet emitters or triplet emitter classes also called sensitizers in connection with hyperfluorescence systems, are described in EP 3 435 438 A2, wherein emitters 2 and 3 on page 21 are preferred; in CN 109111487, wherein the compounds set out on pages 76 and 77 are preferred; in US 2020/0140471, wherein the compounds set out on pages 166 to 175 are preferred; in KR2020108705, wherein the compounds set out on pages 8 to 14 are preferred; in US 2019/0119312, wherein the compounds set out on pages 114 to 121 are preferred; and in US 2020/0411775, wherein the compounds set out on pages 123 to 128 are preferred; Furthermore, preferred fluorescent emitters or classes of fluorescent emitters are described in WO 2021/090932, wherein the compounds set out on pages 129 to 133, 157 to 166, 171 to 187, 200 to 211, 222 to 227, 236 to 252, 255 are preferred; in WO 2020/054676, wherein the compounds set out on
- another phosphorescent emitter which emits at a shorter wavelength than the actual emitter, can be present in the mixture as a co-host. Particularly good results are achieved when a red phosphorescent emitter is used as the emitter and a yellow phosphorescent emitter is used as the co-host in combination with the compound according to the invention.
- a compound that does not participate or does not participate to a significant extent in charge transport can be used as a co-host, as described for example in WO 2010/108579.
- suitable co- Matrix material compounds which have a large band gap and do not participate themselves, or at least not to a significant extent, in the charge transport of the emitting layer.
- Such materials are preferably pure hydrocarbons. Examples of such materials can be found, for example, in WO 2009/124627 or in WO 2010/006680.
- compounds according to the invention without special functional groups, for example hole transport groups and/or electron transport groups have advantageous properties.
- phosphorescent compounds which emit light, preferably in the visible range, when suitably excited and which also contain at least one atom with an atomic number greater than 20, preferably greater than 38 and less than 84, particularly preferably greater than 56 and less than 80, in particular a metal with this atomic number.
- phosphorescent emitters are compounds which contain copper, molybdenum, tungsten, rhenium, ruthenium, osmium, rhodium, iridium, palladium, platinum, silver, gold or europium, in particular compounds which contain iridium or platinum.
- Examples of the emitters described above can be found in applications WO 00/70655, WO 2001/41512, WO 2002/02714, WO 2002/15645, EP 1191613, EP 1191612, EP 1191614, WO 05/033244, WO 05/019373, US 2005/0258742, WO 2009/146770, WO 2010/015307, WO 2010/031485, WO 2010/054731, WO 2010/054728, WO 2010/086089, WO 2010/099852, WO 2010/102709, WO 2011/032626, WO 2011/066898, WO 2011/157339, WO 2012/007086, WO 2014/008982, WO 2014/023377, WO 2014/094961, WO 2014/094960, WO 2015/036074, WO 2015/104045, WO 2015/117718, WO 2016/015815, WO 2016/124304, WO 2017/032439 and
- Examples of phosphorescent dopants are listed in the following table.
- the compounds according to the invention are also particularly suitable as matrix materials for phosphorescent emitters in organic electroluminescent devices, as described, for example, in WO 98/24271, US 2011/0248247 and US 2012/0223633.
- an additional blue emission layer is vapor-deposited over the entire surface of all pixels, including those with a color other than blue.
- the organic electroluminescent device according to the invention does not contain a separate hole injection layer and/or hole transport layer and/or hole blocking layer and/or electron transport layer, i.e. the emitting layer is directly adjacent to the hole injection layer or the anode, and/or the emitting layer is directly adjacent to the electron transport layer or the electron injection layer or the cathode, as described for example in WO 2005/053051.
- a metal complex that is the same or similar to the metal complex in the emitting layer directly adjacent to the emitting layer as a hole transport or hole injection material, as described for example in WO 2009/030981.
- organic electroluminescent device characterized in that one or more layers are coated using a sublimation process.
- the materials are vapor-deposited in vacuum sublimation systems at an initial pressure of less than 10' 5 mbar, preferably less than 10' 6 mbar. However, it is also possible for the initial pressure to be even lower, for example less than 10- 7 mbar.
- an organic electroluminescent device characterized in that one or more layers are coated using the OVPD (Organic Vapour Phase Deposition) method or with the aid of carrier gas sublimation.
- the materials are applied at a pressure between 10' 5 mbar and 1 bar.
- OVPD Organic Vapour Phase Deposition
- OVJP Organic Vapour Jet Printing
- an organic electroluminescent device characterized in that one or more layers are produced from solution, such as by spin coating, or using any printing method, such as screen printing, flexographic printing, offset printing, LITI (light induced thermal imaging, thermal transfer printing), ink-jet printing or nozzle printing. Soluble compounds are required for this, which are obtained, for example, by suitable substitution.
- Formulations for applying a compound according to formula (I) or the preferred embodiments thereof set out above are novel.
- a further subject matter of the present invention is therefore a formulation comprising at least one solvent and a compound according to formula (I) or the preferred embodiments thereof set out above.
- hybrid processes are possible in which, for example, one or more layers are applied from solution and one or more further layers are vapor deposited. These methods are generally known to the person skilled in the art and can be applied by him without inventive step to organic electroluminescent devices containing the compounds according to the invention.
- the compounds according to the invention and the organic electroluminescent devices according to the invention are distinguished from the prior art in particular by improved efficiency and/or operating voltage. Furthermore, these compounds and the organic electroluminescent devices obtainable from them have an improved service life. In a particular variant, the compounds according to the invention and the organic electroluminescent devices according to the invention are distinguished from the prior art in particular by a low refractive index (Refractive Index RI). Furthermore, preferred compounds according to the invention have a high triplet Ti level, so that these compounds are particularly suitable as host material for blue-emitting triplet emitters.
- Refractive Index RI refractive index
- preferred compounds according to the invention have a high triplet Ti level, so that these compounds are particularly suitable as host material for blue-emitting triplet emitters.
- the electronic devices according to the invention are characterized by one or more of the following surprising advantages over the prior art:
- Electronic devices in particular organic electroluminescent devices containing compounds according to formula (I) or the preferred embodiments set out above and below, in particular as matrix material or as hole-conducting materials, have excellent efficiency. In this case, compounds according to the invention according to formula (I) or the preferred embodiments set out above and below result in a low operating voltage when used in electronic devices. 2. Electronic devices, in particular organic electroluminescent devices containing compounds according to formula (I) or the preferred embodiments set out above and below, in particular as matrix material or as hole-conducting materials, have a very good service life. In this case, these compounds in particular cause a low roll-off, ie a small drop in the power efficiency of the device at high luminances.
- Electronic devices in particular organic electroluminescent devices containing compounds according to formula (I) or the preferred embodiments set out above and below, in particular as matrix material or as hole-conducting materials, have a high T1 level.
- optical loss channels can be avoided in electronic devices, in particular organic electroluminescent devices. As a result, these devices are characterized by a high PL and thus high EL efficiency of emitters or an excellent energy transfer from the matrices to dopants.
- the following syntheses are carried out under a protective gas atmosphere in dried solvents, unless otherwise stated.
- the metal complexes are also handled in the absence of light or under yellow light.
- the solvents and reagents can be obtained from Sigma-ALDRICH or ABCR, for example.
- the respective information in square brackets or the numbers given for individual compounds refer to the CAS numbers of the compounds known from the literature. For compounds that can have several enantiomeric, diastereomeric or tautomeric forms, one form is shown as a representative.
- LS5 and LS6 are obtained analogously to E. S. Sherman et al., Adv. Synth. Catal. 2009, 351 , 467, compound 4g and 4h, by using 3-chlorophenylboronic acid [63503-60-6].
- N-bromosuccinimide (NBS)
- BG N-bromosuccinimide
- Stir for 2 hours at 0 °C allow to warm to room temperature overnight, wash the reaction mixture three times with 300 ml of water each time, once with 300 ml of saturated sodium chloride solution and dry over magnesium sulfate.
- the drying agent is filtered off, the filtrate is evaporated to dryness and the residue is chromatographed (Torrent column machine from A. Semrau). Yield:
- the following compounds can be prepared analogously by adjusting the stoichiometry of the reactants, whereby when using chlorides such as LS5 or LS6 instead of 1 mmol palladium acetate and 2 mmol S-Phos.
- a solution of 20.7 g (100 mmol) S1 and 38.6 g (120 mmol) bis-p-biphenylamine [102113-98-4] in 500 ml toluene is treated with 4.0 ml (4 mmol) a tri-tert-butylphosphine solution 1.0 M in toluene, 449 mg (2 mmol) of palladium acetate and 15.4 g of sodium tert-butoxide (160 mmol) are added and the mixture is heated under reflux for 16 h.
- the reaction mixture is filtered while still warm through a Celite bed pre-slurried with toluene.
- the filtrate is concentrated in vacuo and the residue is stirred hot with 300 ml of ethanol.
- the crude product is purified by hot extraction crystallization (typical organic solvents, preferably acetonitrile or acetonitrile/dichloromethane mixtures 4:1 to 1:4 vv) or chromatography (Torrent column machine from A. Semrau) and by fractional sublimation in vacuo (p ⁇ 10 -5 mbar, T ⁇ 280 °C). Yield: 40.0 g (72 mmol) 72 %. Purity according to HPLC >99.9%.
- the mixture is filtered off while still hot through a Celite bed pre-slurried with DMA, the filtrate is largely concentrated, the residue is stirred hot with 300 ml of a methanol/water mixture (1:1 vv), the crude product is filtered off with suction, washed twice with 50 ml of methanol each time and dried in vacuo.
- the regioisomers B700a and B700b are separated by chromatography (Torrent column machine from A. Semrau).
- the individual regioisomers are further purified by repeated hot extraction crystallization (usual organic solvents or combinations thereof, preferably acetonitrile-DCM, 1:3 to 3:1 vv) and fractional sublimation or annealing in a high vacuum. Yield: B700a: 9.7 g (26 mmol) 26%; Purity: approx. 99.9% according to HPLC.
- step 1 coupling of 2.21 g (10 mmol) 2-ethenyl-N-(2-ethenylphenyl)benzenamine [2172900-84-2] with 1.84 g (10 mmol) biphenylsilane [51919-05-2], according to the procedure footnote 17, then step 2 intermolecular bis-hydrosilylation according to H. Nagashima et al, Organometallics, 1989, 8, 10, example 7. Purification is carried out in each case by chromatography and repeated hot extraction crystallization (usual organic solvents or combinations thereof, preferably acetonitrile-DCM, 1:3 to 3:1 vv) and fractional sublimation or.
- OLEDs according to the invention as well as OLEDs according to the prior art is carried out according to a general process according to WO 2004/058911, which is adapted to the conditions described here (layer thickness variation, materials used).
- the compounds B according to the invention can be used in the hole injection layer (HIL), hole transport layer (HTL) and in the electron blocking layer (EBL). All materials are thermally vapor-deposited in a vacuum chamber.
- the emission layer (EML) always consists of at least one matrix material (host material) SMB (see Table 1 ) and an emitting dopant (dopant, emitter) D, which is mixed into the matrix material or the matrix materials by co-evaporation in a certain volume proportion.
- a specification such as SMB:D (97:3%) means that the material SMB is mixed in a volume proportion of 97% and the dopant D is present in the layer in a proportion of 3%.
- the electron transport layer can also consist of a mixture of two materials, see Table 1. The materials used to produce the OLEDs are shown in Table 5 or refer to the synthesis examples presented above.
- the OLEDs are characterized as standard.
- the electroluminescence spectra, the current efficiency (measured in cd/A), the power efficiency (measured in Im/W) and the external quantum efficiency (EQE, measured in percent) are determined as a function of the luminance, calculated from current-voltage-luminance characteristics (IUL characteristics) assuming a Lambertian curve.
- the EQE in (%) and the voltage in (V) are specified at a luminance of 1000 cd/m 2 .
- the OLEDs have the following layer structure:
- Hole injection layer (HIL) made of HTM1 doped with 5% NDP-9 (commercially available from Novaled), 20 nm Hole transport layer (HTL), see Table 1 Electron blocking layer (EBL), see Table 1 Emission layer (EML), see Table 1 Electron transport layer (ETL), see Table 1 Electron injection layer (EIL) made of ETM2, 1 nm Cathode made of aluminum, 100 nm 1b) Phosphorescent OLED components:
- the compounds B according to the invention can be used in the hole injection layer (HIL), the hole transport layer (HTL), the electron blocking layer (EBL) and in the emission layer (EML) as matrix material (host material). For this purpose, all materials are thermally vapor-deposited in a vacuum chamber.
- the emission layer always consists of at least one or more matrix materials M and a phosphorescent dopant Ir, which is mixed into the matrix material or materials by co-evaporation in a certain volume proportion.
- a specification such as M1:M2:Ir (55%:35%:10%) means that the material M1 is present in the layer in a volume proportion of 55%, M2 in a volume proportion of 35% and Ir in a volume proportion of 10%.
- the electron transport layer can also consist of a mixture of two materials.
- the exact structure of the OLEDs can be found in Table 3.
- the materials used to manufacture the OLEDs are shown in Table 5 or refer to the synthesis examples presented above.
- the OLEDs are characterized as standard.
- the electroluminescence spectra, the current efficiency (measured in cd/A), the power efficiency (measured in lm/W) and the external quantum efficiency (EQE, measured in percent) are determined as a function of the luminance, calculated from current-voltage-luminance characteristics (IUL characteristics) assuming a Lambertian radiation characteristic.
- the EQE in (%) and the voltage in (V) are given at a luminance of 1000 cd/m 2 .
- the OLEDs have the following layer structure: Substrate Hole injection layer (HIL) made of HTM1 doped with 5% NDP-9 (commercially available from Novaled), 20 nm hole transport layer (HTL), see Table 3 Electron blocking layer (EBL), see Table 3 Emission layer (EML), see Table 3 Hole blocking layer (HBL), see Table 3 Electron transport layer (ETL), made of ETM1:ETM2 (50%:50%), 30 nm Electron injection layer (EIL) made of ETM2, 1 nm Cathode made of aluminum, 100 nm 15 Table 4: Results of phosphorescent OLED devices
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Optics & Photonics (AREA)
Abstract
Description
Claims
Priority Applications (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP24706691.3A EP4676924A1 (de) | 2023-03-07 | 2024-02-19 | Cyclische stickstoffverbindungen für organische elektrolumineszenzvorrichtungen |
| KR1020257033287A KR20250156170A (ko) | 2023-03-07 | 2024-02-19 | 유기 전계 발광 디바이스용 환형 질소 화합물 |
| CN202480015993.1A CN120787228A (zh) | 2023-03-07 | 2024-02-19 | 用于有机电致发光器件的环状氮化合物 |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP23160430.7 | 2023-03-07 | ||
| EP23160430 | 2023-03-07 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024184050A1 true WO2024184050A1 (de) | 2024-09-12 |
Family
ID=85510890
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2024/054151 Ceased WO2024184050A1 (de) | 2023-03-07 | 2024-02-19 | Cyclische stickstoffverbindungen für organische elektrolumineszenzvorrichtungen |
Country Status (4)
| Country | Link |
|---|---|
| EP (1) | EP4676924A1 (de) |
| KR (1) | KR20250156170A (de) |
| CN (1) | CN120787228A (de) |
| WO (1) | WO2024184050A1 (de) |
Citations (117)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1992018552A1 (de) | 1991-04-11 | 1992-10-29 | Wacker-Chemie Gmbh | Leiterpolymere mit konjugierten doppelbindungen |
| EP0652273A1 (de) | 1993-11-09 | 1995-05-10 | Shinko Electric Industries Co. Ltd. | Organisches Material für elektrolumineszente Vorrichtung und elektrolumineszente Vorrichtung |
| EP0707020A2 (de) | 1994-10-14 | 1996-04-17 | Hoechst Aktiengesellschaft | Konjugierte Polymere mit Spirozentren und ihre Verwendung als Elektrolumineszenzmaterialien |
| EP0842208A1 (de) | 1995-07-28 | 1998-05-20 | The Dow Chemical Company | 2,7-aryl-9-substituierte fluorene und 9-substituierte fluorenoligomere und polymere |
| WO1998024271A1 (en) | 1996-11-25 | 1998-06-04 | Seiko Epson Corporation | Method of producing organic el elements, organic el elements and organic el display device |
| EP0894107A1 (de) | 1996-04-17 | 1999-02-03 | Hoechst Research & Technology Deutschland GmbH & Co. KG | Polymere mit spiroatomen und ihre verwendung als elektrolumineszenzmaterialien |
| WO2000022026A1 (de) | 1998-10-10 | 2000-04-20 | Celanese Ventures Gmbh | Konjugierte polymere, enthaltend spezielle fluoren-bausteine mit verbesserten eigenschaften |
| EP1028136A2 (de) | 1999-02-10 | 2000-08-16 | Carnegie-Mellon University | Ein Verfahren zur Herstellung von Poly(3-substituierten)thiophenen |
| WO2000070655A2 (en) | 1999-05-13 | 2000-11-23 | The Trustees Of Princeton University | Very high efficiency organic light emitting devices based on electrophosphorescence |
| JP3139321B2 (ja) | 1994-03-31 | 2001-02-26 | 東レ株式会社 | 発光素子 |
| WO2001041512A1 (en) | 1999-12-01 | 2001-06-07 | The Trustees Of Princeton University | Complexes of form l2mx as phosphorescent dopants for organic leds |
| WO2002002714A2 (en) | 2000-06-30 | 2002-01-10 | E.I. Du Pont De Nemours And Company | Electroluminescent iridium compounds with fluorinated phenylpyridines, phenylpyrimidines, and phenylquinolines and devices made with such compounds |
| WO2002015645A1 (en) | 2000-08-11 | 2002-02-21 | The Trustees Of Princeton University | Organometallic compounds and emission-shifting organic electrophosphorescence |
| EP1191614A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung und dafür verwendete Metallkoordinationsverbindung |
| EP1191613A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung, Bildanzeigevorrichtung und Metallkoordinationsverbindung |
| EP1191612A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung, Bildanzeigevorrichtung und Metallkoordinationsverbindung |
| EP1205527A1 (de) | 2000-03-27 | 2002-05-15 | Idemitsu Kosan Co., Ltd. | Organische elektrolumineszierende vorrichtung |
| WO2004013080A1 (en) | 2002-08-01 | 2004-02-12 | Covion Organic Semiconductors Gmbh | Spirobifluorene derivatives, their preparation and uses thereof |
| WO2004041901A1 (en) | 2002-11-08 | 2004-05-21 | Covion Organic Semiconductors Gmbh | Aryl-substituted polyindenofluorenes for use in organic electroluminiscent devices |
| WO2004058911A2 (de) | 2002-12-23 | 2004-07-15 | Covion Organic Semiconductors Gmbh | Organisches elektrolumineszenzelement |
| WO2004070772A2 (de) | 2003-02-06 | 2004-08-19 | Covion Organic Semiconductors Gmbh | Carbazol-enthaltende konjugierte polymere und blends, deren darstellung und verwendung |
| JP2004288381A (ja) | 2003-03-19 | 2004-10-14 | Konica Minolta Holdings Inc | 有機エレクトロルミネッセンス素子 |
| WO2004093207A2 (de) | 2003-04-15 | 2004-10-28 | Covion Organic Semiconductors Gmbh | Mischungen von organischen zur emission befähigten halbleitern und matrixmaterialien, deren verwendung und elektronikbauteile enthaltend diese mischungen |
| WO2004113412A2 (en) | 2003-06-23 | 2004-12-29 | Covion Organic Semiconductors Gmbh | Polymer |
| WO2004113468A1 (de) | 2003-06-26 | 2004-12-29 | Covion Organic Semiconductors Gmbh | Neue materialien für die elektrolumineszenz |
| WO2005014689A2 (de) | 2003-08-12 | 2005-02-17 | Covion Organic Semiconductors Gmbh | Konjugierte polymere enthaltend dihydrophenanthren-einheiten und deren verwendung |
| WO2005019373A2 (de) | 2003-08-19 | 2005-03-03 | Basf Aktiengesellschaft | Übergangsmetallkomplexe mit carbenliganden als emitter für organische licht-emittierende dioden (oleds) |
| US20050069729A1 (en) | 2003-09-30 | 2005-03-31 | Konica Minolta Holdings, Inc. | Organic electroluminescent element, illuminator, display and compound |
| WO2005033244A1 (de) | 2003-09-29 | 2005-04-14 | Covion Organic Semiconductors Gmbh | Metallkomplexe |
| WO2005040302A1 (de) | 2003-10-22 | 2005-05-06 | Merck Patent Gmbh | Neue materialien für die elektrolumineszenz und deren verwendung |
| WO2005053051A1 (de) | 2003-11-25 | 2005-06-09 | Merck Patent Gmbh | Organisches elektrolumineszenzelement |
| WO2005104264A1 (de) | 2004-04-26 | 2005-11-03 | Merck Patent Gmbh | Elektrolumineszierende polymere und deren verwendung |
| US20050258742A1 (en) | 2004-05-18 | 2005-11-24 | Yui-Yi Tsai | Carbene containing metal complexes as OLEDs |
| WO2005111172A2 (de) | 2004-05-11 | 2005-11-24 | Merck Patent Gmbh | Neue materialmischungen für die elektrolumineszenz |
| JP2005347160A (ja) | 2004-06-04 | 2005-12-15 | Konica Minolta Holdings Inc | 有機エレクトロルミネッセンス素子、照明装置及び表示装置 |
| EP1617710A1 (de) | 2003-04-23 | 2006-01-18 | Konica Minolta Holdings, Inc. | Material für ein organisches elektrolumineszenzgerät, organisches elektrolumineszenzgerät, beleuchtungsvorrichtung und anzeige |
| WO2006005627A1 (en) | 2004-07-15 | 2006-01-19 | Merck Patent Gmbh | Oligomeric derivatives of spirobifluorene, their preparation and use |
| WO2006061181A1 (de) | 2004-12-06 | 2006-06-15 | Merck Patent Gmbh | Teilkonjugierte polymere, deren darstellung und verwendung |
| WO2006117052A1 (de) | 2005-05-03 | 2006-11-09 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtung und in deren herstellung verwendete boronsäure- und borinsäure-derivate |
| EP1731584A1 (de) | 2004-03-31 | 2006-12-13 | Konica Minolta Holdings, Inc. | Organischer elektrolumineszenzvorrichtungsstoff, organische elektrolumineszenzvorrichtung, display und beleuchtungsvorrichtung |
| WO2007017066A1 (de) | 2005-08-10 | 2007-02-15 | Merck Patent Gmbh | Elektrolumineszierende polymere und ihre verwendung |
| WO2007063754A1 (ja) | 2005-12-01 | 2007-06-07 | Nippon Steel Chemical Co., Ltd. | 有機電界発光素子用化合物及び有機電界発光素子 |
| WO2007137725A1 (de) | 2006-05-31 | 2007-12-06 | Merck Patent Gmbh | Neue materialien für organische elektrolumineszenzvorrichtungen |
| WO2008056746A1 (en) | 2006-11-09 | 2008-05-15 | Nippon Steel Chemical Co., Ltd. | Compound for organic electroluminescent device and organic electroluminescent device |
| WO2008086851A1 (de) | 2007-01-18 | 2008-07-24 | Merck Patent Gmbh | Carbazol-derivate für organische elektrolumineszenzvorrichtungen |
| WO2009030981A2 (en) | 2006-12-28 | 2009-03-12 | Universal Display Corporation | Long lifetime phosphorescent organic light emitting device (oled) structures |
| WO2009062578A1 (de) | 2007-11-12 | 2009-05-22 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtungen enthaltend azomethin-metall-komplexe |
| WO2009124627A1 (de) | 2008-04-07 | 2009-10-15 | Merck Patent Gmbh | Pluorenderivate für organische elektrolumineszenzvorrichtungen |
| WO2009146770A2 (de) | 2008-06-05 | 2009-12-10 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe |
| WO2010006680A1 (de) | 2008-07-18 | 2010-01-21 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010015306A1 (de) | 2008-08-08 | 2010-02-11 | Merck Patent Gmbh, | Organische elektrolumineszenzvorrichtung |
| WO2010015307A1 (de) | 2008-08-04 | 2010-02-11 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe mit isonitrilliganden |
| WO2010031485A1 (de) | 2008-09-22 | 2010-03-25 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054729A2 (de) | 2008-11-11 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054731A1 (de) | 2008-11-13 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054728A1 (de) | 2008-11-13 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054730A1 (de) | 2008-11-11 | 2010-05-20 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtungen |
| WO2010086089A1 (de) | 2009-02-02 | 2010-08-05 | Merck Patent Gmbh | Metallkomplexe |
| WO2010099852A1 (de) | 2009-03-02 | 2010-09-10 | Merck Patent Gmbh | Metallkomplexe mit azaborol-liganden und elektronische vorrichtung damit |
| WO2010102709A1 (de) | 2009-03-13 | 2010-09-16 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010108579A1 (de) | 2009-03-23 | 2010-09-30 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtung |
| WO2010136109A1 (de) | 2009-05-29 | 2010-12-02 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011000455A1 (de) | 2009-06-30 | 2011-01-06 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011032626A1 (de) | 2009-09-16 | 2011-03-24 | Merck Patent Gmbh | Metallkomplexe |
| WO2011042107A2 (de) | 2009-10-08 | 2011-04-14 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011057706A2 (de) | 2009-11-14 | 2011-05-19 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2011060859A1 (de) | 2009-11-17 | 2011-05-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011060867A1 (de) | 2009-11-18 | 2011-05-26 | Merck Patent Gmbh | Stickstoffhaltige kondensierte heterozyklen für oleds |
| WO2011060877A2 (de) | 2009-11-17 | 2011-05-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011066898A1 (de) | 2009-12-05 | 2011-06-09 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe |
| WO2011088877A1 (de) | 2010-01-25 | 2011-07-28 | Merck Patent Gmbh | Verbindungen für elektronische vorrichtungen |
| US20110248247A1 (en) | 2010-04-09 | 2011-10-13 | Sony Corporation | Organic el display unit, method of manufacturing the same, and solution used in method |
| WO2011157339A1 (de) | 2010-06-15 | 2011-12-22 | Merck Patent Gmbh | Metallkomplexe |
| WO2012007086A1 (de) | 2010-07-16 | 2012-01-19 | Merck Patent Gmbh | Metallkomplexe |
| WO2012048781A1 (de) | 2010-10-15 | 2012-04-19 | Merck Patent Gmbh | Materialien auf basis von triphenylen für organische elektrolumineszenzvorrichtungen |
| US20120223633A1 (en) | 2011-03-04 | 2012-09-06 | Sony Corporation | Organic el display device and method of manufacturing the same |
| WO2012133188A1 (ja) | 2011-03-25 | 2012-10-04 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2012139692A1 (de) * | 2011-04-13 | 2012-10-18 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2012143080A2 (de) | 2011-04-18 | 2012-10-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2013041176A1 (de) | 2011-09-21 | 2013-03-28 | Merck Patent Gmbh | Carbazolderivate für organische elektrolumineszenzvorrichtungen |
| WO2013056776A1 (de) | 2011-10-20 | 2013-04-25 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2014008982A1 (de) | 2012-07-13 | 2014-01-16 | Merck Patent Gmbh | Metallkomplexe |
| WO2014023377A2 (de) | 2012-08-07 | 2014-02-13 | Merck Patent Gmbh | Metallkomplexe |
| WO2014094961A1 (de) | 2012-12-21 | 2014-06-26 | Merck Patent Gmbh | Metallkomplexe |
| WO2014094960A1 (de) | 2012-12-21 | 2014-06-26 | Merck Patent Gmbh | Metallkomplexe |
| WO2015036074A1 (de) | 2013-09-11 | 2015-03-19 | Merck Patent Gmbh | Metallkomplexe |
| WO2015091716A1 (en) | 2013-12-20 | 2015-06-25 | Basf Se | Highly efficient oled devices with very short decay times |
| WO2015104045A1 (de) | 2014-01-13 | 2015-07-16 | Merck Patent Gmbh | Metallkomplexe |
| WO2015117718A1 (de) | 2014-02-05 | 2015-08-13 | Merck Patent Gmbh | Metallkomplexe |
| WO2015169412A1 (de) | 2014-05-05 | 2015-11-12 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2016015810A1 (de) | 2014-07-29 | 2016-02-04 | Merck Patent Gmbh | Materialien f?r organische elektrolumineszenzvorrichtungen |
| WO2016015815A1 (de) | 2014-07-28 | 2016-02-04 | Merck Patent Gmbh | Metallkomplexe |
| WO2016023608A1 (de) | 2014-08-13 | 2016-02-18 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2016124304A1 (de) | 2015-02-03 | 2016-08-11 | Merck Patent Gmbh | Metallkomplexe |
| WO2016193243A1 (en) | 2015-06-03 | 2016-12-08 | Udc Ireland Limited | Highly efficient oled devices with very short decay times |
| WO2017032439A1 (de) | 2015-08-25 | 2017-03-02 | Merck Patent Gmbh | Metallkomplexe |
| WO2017148564A1 (de) | 2016-03-03 | 2017-09-08 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| US20170271611A1 (en) | 2014-08-22 | 2017-09-21 | Jian Li | Organic light-emitting diodes with fluorescent and phosphorescent emitters |
| WO2018011186A1 (de) | 2016-07-14 | 2018-01-18 | Merck Patent Gmbh | Metallkomplexe |
| WO2018212169A1 (ja) | 2017-05-16 | 2018-11-22 | 学校法人関西学院 | 多環芳香族化合物 |
| CN109111487A (zh) | 2017-06-23 | 2019-01-01 | 环球展览公司 | 有机电致发光材料和装置 |
| EP3435438A2 (de) | 2017-07-26 | 2019-01-30 | Universal Display Corporation | Organische elektrolumineszente materialien und vorrichtungen |
| US10249832B1 (en) | 2017-12-06 | 2019-04-02 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device and novel compound |
| US20190119312A1 (en) | 2017-06-23 | 2019-04-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| WO2019235452A1 (ja) | 2018-06-06 | 2019-12-12 | 学校法人関西学院 | ターシャリーアルキル置換多環芳香族化合物 |
| WO2020017931A1 (ko) | 2018-07-19 | 2020-01-23 | 주식회사 엘지화학 | 다환 화합물 및 이를 포함하는 유기 발광 소자 |
| WO2020054676A1 (ja) | 2018-09-10 | 2020-03-19 | 学校法人関西学院 | 有機電界発光素子 |
| US20200140471A1 (en) | 2017-06-23 | 2020-05-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| KR20200108705A (ko) | 2019-03-11 | 2020-09-21 | 부산대학교 산학협력단 | 진청색 이리듐 착화합물 및 이를 포함하는 유기전계발광소자 |
| WO2020218079A1 (ja) | 2019-04-22 | 2020-10-29 | 学校法人関西学院 | シクロアルカン縮合多環芳香族化合物 |
| US20200411775A1 (en) | 2017-06-23 | 2020-12-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| WO2021014001A1 (en) | 2019-07-25 | 2021-01-28 | Cynora Gmbh | Organic molecules for optoelectronic devices |
| WO2021090932A1 (ja) | 2019-11-08 | 2021-05-14 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子及び電子機器 |
| EP3885350A1 (de) | 2018-11-19 | 2021-09-29 | SFC Co., Ltd. | Neuartige bor-verbindung und organische lichtemittierende vorrichtung damit |
| WO2021230653A1 (ko) | 2020-05-12 | 2021-11-18 | 에스에프씨 주식회사 | 유기발광 화합물 및 이를 포함하는 유기발광소자 |
| WO2022079068A1 (de) | 2020-10-16 | 2022-04-21 | Merck Patent Gmbh | Heterocyclische verbindungen für organische elektrolumineszenzvorrichtungen |
| EP4123735A1 (de) | 2020-03-18 | 2023-01-25 | SFC Co., Ltd. | Organoelektrolumineszente vorrichtung mit polycyclischen aromatischen verbindungen |
-
2024
- 2024-02-19 WO PCT/EP2024/054151 patent/WO2024184050A1/de not_active Ceased
- 2024-02-19 CN CN202480015993.1A patent/CN120787228A/zh active Pending
- 2024-02-19 EP EP24706691.3A patent/EP4676924A1/de active Pending
- 2024-02-19 KR KR1020257033287A patent/KR20250156170A/ko active Pending
Patent Citations (120)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO1992018552A1 (de) | 1991-04-11 | 1992-10-29 | Wacker-Chemie Gmbh | Leiterpolymere mit konjugierten doppelbindungen |
| EP0652273A1 (de) | 1993-11-09 | 1995-05-10 | Shinko Electric Industries Co. Ltd. | Organisches Material für elektrolumineszente Vorrichtung und elektrolumineszente Vorrichtung |
| JP3139321B2 (ja) | 1994-03-31 | 2001-02-26 | 東レ株式会社 | 発光素子 |
| EP0707020A2 (de) | 1994-10-14 | 1996-04-17 | Hoechst Aktiengesellschaft | Konjugierte Polymere mit Spirozentren und ihre Verwendung als Elektrolumineszenzmaterialien |
| EP0842208A1 (de) | 1995-07-28 | 1998-05-20 | The Dow Chemical Company | 2,7-aryl-9-substituierte fluorene und 9-substituierte fluorenoligomere und polymere |
| EP0894107A1 (de) | 1996-04-17 | 1999-02-03 | Hoechst Research & Technology Deutschland GmbH & Co. KG | Polymere mit spiroatomen und ihre verwendung als elektrolumineszenzmaterialien |
| WO1998024271A1 (en) | 1996-11-25 | 1998-06-04 | Seiko Epson Corporation | Method of producing organic el elements, organic el elements and organic el display device |
| WO2000022026A1 (de) | 1998-10-10 | 2000-04-20 | Celanese Ventures Gmbh | Konjugierte polymere, enthaltend spezielle fluoren-bausteine mit verbesserten eigenschaften |
| EP1028136A2 (de) | 1999-02-10 | 2000-08-16 | Carnegie-Mellon University | Ein Verfahren zur Herstellung von Poly(3-substituierten)thiophenen |
| WO2000070655A2 (en) | 1999-05-13 | 2000-11-23 | The Trustees Of Princeton University | Very high efficiency organic light emitting devices based on electrophosphorescence |
| WO2001041512A1 (en) | 1999-12-01 | 2001-06-07 | The Trustees Of Princeton University | Complexes of form l2mx as phosphorescent dopants for organic leds |
| EP1205527A1 (de) | 2000-03-27 | 2002-05-15 | Idemitsu Kosan Co., Ltd. | Organische elektrolumineszierende vorrichtung |
| WO2002002714A2 (en) | 2000-06-30 | 2002-01-10 | E.I. Du Pont De Nemours And Company | Electroluminescent iridium compounds with fluorinated phenylpyridines, phenylpyrimidines, and phenylquinolines and devices made with such compounds |
| WO2002015645A1 (en) | 2000-08-11 | 2002-02-21 | The Trustees Of Princeton University | Organometallic compounds and emission-shifting organic electrophosphorescence |
| EP1191614A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung und dafür verwendete Metallkoordinationsverbindung |
| EP1191613A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung, Bildanzeigevorrichtung und Metallkoordinationsverbindung |
| EP1191612A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung, Bildanzeigevorrichtung und Metallkoordinationsverbindung |
| WO2004013080A1 (en) | 2002-08-01 | 2004-02-12 | Covion Organic Semiconductors Gmbh | Spirobifluorene derivatives, their preparation and uses thereof |
| WO2004041901A1 (en) | 2002-11-08 | 2004-05-21 | Covion Organic Semiconductors Gmbh | Aryl-substituted polyindenofluorenes for use in organic electroluminiscent devices |
| WO2004058911A2 (de) | 2002-12-23 | 2004-07-15 | Covion Organic Semiconductors Gmbh | Organisches elektrolumineszenzelement |
| WO2004070772A2 (de) | 2003-02-06 | 2004-08-19 | Covion Organic Semiconductors Gmbh | Carbazol-enthaltende konjugierte polymere und blends, deren darstellung und verwendung |
| JP2004288381A (ja) | 2003-03-19 | 2004-10-14 | Konica Minolta Holdings Inc | 有機エレクトロルミネッセンス素子 |
| WO2004093207A2 (de) | 2003-04-15 | 2004-10-28 | Covion Organic Semiconductors Gmbh | Mischungen von organischen zur emission befähigten halbleitern und matrixmaterialien, deren verwendung und elektronikbauteile enthaltend diese mischungen |
| EP1617710A1 (de) | 2003-04-23 | 2006-01-18 | Konica Minolta Holdings, Inc. | Material für ein organisches elektrolumineszenzgerät, organisches elektrolumineszenzgerät, beleuchtungsvorrichtung und anzeige |
| EP1617711A1 (de) | 2003-04-23 | 2006-01-18 | Konica Minolta Holdings, Inc. | Organisches elektrolumineszenzbauelement und anzeige |
| WO2004113412A2 (en) | 2003-06-23 | 2004-12-29 | Covion Organic Semiconductors Gmbh | Polymer |
| WO2004113468A1 (de) | 2003-06-26 | 2004-12-29 | Covion Organic Semiconductors Gmbh | Neue materialien für die elektrolumineszenz |
| WO2005014689A2 (de) | 2003-08-12 | 2005-02-17 | Covion Organic Semiconductors Gmbh | Konjugierte polymere enthaltend dihydrophenanthren-einheiten und deren verwendung |
| WO2005019373A2 (de) | 2003-08-19 | 2005-03-03 | Basf Aktiengesellschaft | Übergangsmetallkomplexe mit carbenliganden als emitter für organische licht-emittierende dioden (oleds) |
| WO2005033244A1 (de) | 2003-09-29 | 2005-04-14 | Covion Organic Semiconductors Gmbh | Metallkomplexe |
| US20050069729A1 (en) | 2003-09-30 | 2005-03-31 | Konica Minolta Holdings, Inc. | Organic electroluminescent element, illuminator, display and compound |
| WO2005039246A1 (ja) | 2003-09-30 | 2005-04-28 | Konica Minolta Holdings, Inc. | 有機エレクトロルミネッセンス素子、照明装置、表示装置 |
| WO2005040302A1 (de) | 2003-10-22 | 2005-05-06 | Merck Patent Gmbh | Neue materialien für die elektrolumineszenz und deren verwendung |
| WO2005053051A1 (de) | 2003-11-25 | 2005-06-09 | Merck Patent Gmbh | Organisches elektrolumineszenzelement |
| EP1731584A1 (de) | 2004-03-31 | 2006-12-13 | Konica Minolta Holdings, Inc. | Organischer elektrolumineszenzvorrichtungsstoff, organische elektrolumineszenzvorrichtung, display und beleuchtungsvorrichtung |
| WO2005104264A1 (de) | 2004-04-26 | 2005-11-03 | Merck Patent Gmbh | Elektrolumineszierende polymere und deren verwendung |
| WO2005111172A2 (de) | 2004-05-11 | 2005-11-24 | Merck Patent Gmbh | Neue materialmischungen für die elektrolumineszenz |
| US20050258742A1 (en) | 2004-05-18 | 2005-11-24 | Yui-Yi Tsai | Carbene containing metal complexes as OLEDs |
| JP2005347160A (ja) | 2004-06-04 | 2005-12-15 | Konica Minolta Holdings Inc | 有機エレクトロルミネッセンス素子、照明装置及び表示装置 |
| WO2006005627A1 (en) | 2004-07-15 | 2006-01-19 | Merck Patent Gmbh | Oligomeric derivatives of spirobifluorene, their preparation and use |
| WO2006061181A1 (de) | 2004-12-06 | 2006-06-15 | Merck Patent Gmbh | Teilkonjugierte polymere, deren darstellung und verwendung |
| WO2006117052A1 (de) | 2005-05-03 | 2006-11-09 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtung und in deren herstellung verwendete boronsäure- und borinsäure-derivate |
| WO2007017066A1 (de) | 2005-08-10 | 2007-02-15 | Merck Patent Gmbh | Elektrolumineszierende polymere und ihre verwendung |
| WO2007063754A1 (ja) | 2005-12-01 | 2007-06-07 | Nippon Steel Chemical Co., Ltd. | 有機電界発光素子用化合物及び有機電界発光素子 |
| WO2007137725A1 (de) | 2006-05-31 | 2007-12-06 | Merck Patent Gmbh | Neue materialien für organische elektrolumineszenzvorrichtungen |
| WO2008056746A1 (en) | 2006-11-09 | 2008-05-15 | Nippon Steel Chemical Co., Ltd. | Compound for organic electroluminescent device and organic electroluminescent device |
| WO2009030981A2 (en) | 2006-12-28 | 2009-03-12 | Universal Display Corporation | Long lifetime phosphorescent organic light emitting device (oled) structures |
| WO2008086851A1 (de) | 2007-01-18 | 2008-07-24 | Merck Patent Gmbh | Carbazol-derivate für organische elektrolumineszenzvorrichtungen |
| WO2009062578A1 (de) | 2007-11-12 | 2009-05-22 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtungen enthaltend azomethin-metall-komplexe |
| WO2009124627A1 (de) | 2008-04-07 | 2009-10-15 | Merck Patent Gmbh | Pluorenderivate für organische elektrolumineszenzvorrichtungen |
| WO2009146770A2 (de) | 2008-06-05 | 2009-12-10 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe |
| WO2010006680A1 (de) | 2008-07-18 | 2010-01-21 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010015307A1 (de) | 2008-08-04 | 2010-02-11 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe mit isonitrilliganden |
| WO2010015306A1 (de) | 2008-08-08 | 2010-02-11 | Merck Patent Gmbh, | Organische elektrolumineszenzvorrichtung |
| WO2010031485A1 (de) | 2008-09-22 | 2010-03-25 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054729A2 (de) | 2008-11-11 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054730A1 (de) | 2008-11-11 | 2010-05-20 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtungen |
| WO2010054731A1 (de) | 2008-11-13 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054728A1 (de) | 2008-11-13 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010086089A1 (de) | 2009-02-02 | 2010-08-05 | Merck Patent Gmbh | Metallkomplexe |
| WO2010099852A1 (de) | 2009-03-02 | 2010-09-10 | Merck Patent Gmbh | Metallkomplexe mit azaborol-liganden und elektronische vorrichtung damit |
| WO2010102709A1 (de) | 2009-03-13 | 2010-09-16 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010108579A1 (de) | 2009-03-23 | 2010-09-30 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtung |
| WO2010136109A1 (de) | 2009-05-29 | 2010-12-02 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011000455A1 (de) | 2009-06-30 | 2011-01-06 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011032626A1 (de) | 2009-09-16 | 2011-03-24 | Merck Patent Gmbh | Metallkomplexe |
| WO2011042107A2 (de) | 2009-10-08 | 2011-04-14 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011057706A2 (de) | 2009-11-14 | 2011-05-19 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2011060859A1 (de) | 2009-11-17 | 2011-05-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011060877A2 (de) | 2009-11-17 | 2011-05-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011060867A1 (de) | 2009-11-18 | 2011-05-26 | Merck Patent Gmbh | Stickstoffhaltige kondensierte heterozyklen für oleds |
| WO2011066898A1 (de) | 2009-12-05 | 2011-06-09 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe |
| WO2011088877A1 (de) | 2010-01-25 | 2011-07-28 | Merck Patent Gmbh | Verbindungen für elektronische vorrichtungen |
| US20110248247A1 (en) | 2010-04-09 | 2011-10-13 | Sony Corporation | Organic el display unit, method of manufacturing the same, and solution used in method |
| WO2011157339A1 (de) | 2010-06-15 | 2011-12-22 | Merck Patent Gmbh | Metallkomplexe |
| WO2012007086A1 (de) | 2010-07-16 | 2012-01-19 | Merck Patent Gmbh | Metallkomplexe |
| WO2012048781A1 (de) | 2010-10-15 | 2012-04-19 | Merck Patent Gmbh | Materialien auf basis von triphenylen für organische elektrolumineszenzvorrichtungen |
| US20120223633A1 (en) | 2011-03-04 | 2012-09-06 | Sony Corporation | Organic el display device and method of manufacturing the same |
| WO2012133188A1 (ja) | 2011-03-25 | 2012-10-04 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子 |
| WO2012139692A1 (de) * | 2011-04-13 | 2012-10-18 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2012143080A2 (de) | 2011-04-18 | 2012-10-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2013041176A1 (de) | 2011-09-21 | 2013-03-28 | Merck Patent Gmbh | Carbazolderivate für organische elektrolumineszenzvorrichtungen |
| WO2013056776A1 (de) | 2011-10-20 | 2013-04-25 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2014008982A1 (de) | 2012-07-13 | 2014-01-16 | Merck Patent Gmbh | Metallkomplexe |
| WO2014023377A2 (de) | 2012-08-07 | 2014-02-13 | Merck Patent Gmbh | Metallkomplexe |
| WO2014094961A1 (de) | 2012-12-21 | 2014-06-26 | Merck Patent Gmbh | Metallkomplexe |
| WO2014094960A1 (de) | 2012-12-21 | 2014-06-26 | Merck Patent Gmbh | Metallkomplexe |
| WO2015036074A1 (de) | 2013-09-11 | 2015-03-19 | Merck Patent Gmbh | Metallkomplexe |
| WO2015091716A1 (en) | 2013-12-20 | 2015-06-25 | Basf Se | Highly efficient oled devices with very short decay times |
| WO2015104045A1 (de) | 2014-01-13 | 2015-07-16 | Merck Patent Gmbh | Metallkomplexe |
| WO2015117718A1 (de) | 2014-02-05 | 2015-08-13 | Merck Patent Gmbh | Metallkomplexe |
| WO2015169412A1 (de) | 2014-05-05 | 2015-11-12 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2016015815A1 (de) | 2014-07-28 | 2016-02-04 | Merck Patent Gmbh | Metallkomplexe |
| WO2016015810A1 (de) | 2014-07-29 | 2016-02-04 | Merck Patent Gmbh | Materialien f?r organische elektrolumineszenzvorrichtungen |
| WO2016023608A1 (de) | 2014-08-13 | 2016-02-18 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| US20170271611A1 (en) | 2014-08-22 | 2017-09-21 | Jian Li | Organic light-emitting diodes with fluorescent and phosphorescent emitters |
| WO2016124304A1 (de) | 2015-02-03 | 2016-08-11 | Merck Patent Gmbh | Metallkomplexe |
| WO2016193243A1 (en) | 2015-06-03 | 2016-12-08 | Udc Ireland Limited | Highly efficient oled devices with very short decay times |
| WO2017032439A1 (de) | 2015-08-25 | 2017-03-02 | Merck Patent Gmbh | Metallkomplexe |
| WO2017148564A1 (de) | 2016-03-03 | 2017-09-08 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2017148565A1 (de) | 2016-03-03 | 2017-09-08 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2018011186A1 (de) | 2016-07-14 | 2018-01-18 | Merck Patent Gmbh | Metallkomplexe |
| WO2018212169A1 (ja) | 2017-05-16 | 2018-11-22 | 学校法人関西学院 | 多環芳香族化合物 |
| US20190119312A1 (en) | 2017-06-23 | 2019-04-25 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US20200411775A1 (en) | 2017-06-23 | 2020-12-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US20200140471A1 (en) | 2017-06-23 | 2020-05-07 | Universal Display Corporation | Organic electroluminescent materials and devices |
| CN109111487A (zh) | 2017-06-23 | 2019-01-01 | 环球展览公司 | 有机电致发光材料和装置 |
| EP3435438A2 (de) | 2017-07-26 | 2019-01-30 | Universal Display Corporation | Organische elektrolumineszente materialien und vorrichtungen |
| US10249832B1 (en) | 2017-12-06 | 2019-04-02 | Idemitsu Kosan Co., Ltd. | Organic electroluminescence device and novel compound |
| WO2019235452A1 (ja) | 2018-06-06 | 2019-12-12 | 学校法人関西学院 | ターシャリーアルキル置換多環芳香族化合物 |
| WO2020017931A1 (ko) | 2018-07-19 | 2020-01-23 | 주식회사 엘지화학 | 다환 화합물 및 이를 포함하는 유기 발광 소자 |
| WO2020054676A1 (ja) | 2018-09-10 | 2020-03-19 | 学校法人関西学院 | 有機電界発光素子 |
| EP3885350A1 (de) | 2018-11-19 | 2021-09-29 | SFC Co., Ltd. | Neuartige bor-verbindung und organische lichtemittierende vorrichtung damit |
| KR20200108705A (ko) | 2019-03-11 | 2020-09-21 | 부산대학교 산학협력단 | 진청색 이리듐 착화합물 및 이를 포함하는 유기전계발광소자 |
| WO2020218079A1 (ja) | 2019-04-22 | 2020-10-29 | 学校法人関西学院 | シクロアルカン縮合多環芳香族化合物 |
| WO2021014001A1 (en) | 2019-07-25 | 2021-01-28 | Cynora Gmbh | Organic molecules for optoelectronic devices |
| WO2021090932A1 (ja) | 2019-11-08 | 2021-05-14 | 出光興産株式会社 | 有機エレクトロルミネッセンス素子及び電子機器 |
| EP4123735A1 (de) | 2020-03-18 | 2023-01-25 | SFC Co., Ltd. | Organoelektrolumineszente vorrichtung mit polycyclischen aromatischen verbindungen |
| WO2021230653A1 (ko) | 2020-05-12 | 2021-11-18 | 에스에프씨 주식회사 | 유기발광 화합물 및 이를 포함하는 유기발광소자 |
| WO2022079068A1 (de) | 2020-10-16 | 2022-04-21 | Merck Patent Gmbh | Heterocyclische verbindungen für organische elektrolumineszenzvorrichtungen |
Non-Patent Citations (22)
| Title |
|---|
| A. W. FREEMAN ET AL., J. ORG. CHEM., vol. 70, 2005, pages 5014 |
| B. E. S. SHERMAN ET AL., ADV. SYNTH. CATAL., vol. 351, no. 2429897-25-4, 2009, pages 467 |
| BUNESCU ET AL.: "Three-Component Povarov Reaction-Heteroannulation with Arynes: Synthesis of 5,6-Dihydroindolo[1,2- a ]quinolines", ORG. LETT., vol. 16, no. 6, 21 March 2014 (2014-03-21), pages 1756 - 1759, XP055141532, ISSN: 1523-7060, DOI: 10.1021/ol500447r * |
| CAMPEAU ET AL., J. AM. CHEM. SOC., vol. 128, 2006, pages 593 |
| D. M. KOLLER ET AL., NATURE PHOTONICS, 2008, pages 1 - 4 |
| DATABASE Reaxys [online] Elsevier Life Sciences IP Limited; 24 July 2018 (2018-07-24), LI ET AL.: "Chiral phosphoric acid-catalyzed direct asymmetric mannich reaction of cyclic: C -acylimines with simple ketones", XP093150666, Database accession no. 33251554, 33251557 * |
| EUNGDO KIN ET AL., SCI., vol. 8, 2022, pages 1641 |
| H. NAGASHIMA ET AL., ORGANOMETALLICS, vol. 8, 1989, pages 10 |
| HUANG ET AL.: "Intermolecular Dehydrative [4 + 2] Aza-Annulation of N -Arylamides with Alkenes: A Direct and Divergent Entrance to Aza-Heterocycles", ORG. LETT., vol. 21, no. 6, 26 February 2019 (2019-02-26), US, pages 1681 - 1685, XP093150655, ISSN: 1523-7060, DOI: 10.1021/acs.orglett.9b00233 * |
| I. ABDELLAH ET AL., CATALYSIS SCIENCE & TECHNOLOGY, vol. 8, no. 24, 2018, pages 6486 |
| J.-S. LI ET AL., CHEM. COMMUN., vol. 54, 2018, pages 9151 |
| K. SUZUKI ET AL., ADV. SYNTH. & CATAL., vol. 350, no. 5, 2008, pages 652 |
| L. ZHANG ET AL., ORGANIC PROCESS RESEARCH & DEVELOPMENT, vol. 24, no. 10, 2020, pages 2078 |
| LI ET AL.: "Electronic Supplementary Information (ESI) for: Chiral Phosphoric Acid-Catalyzed Direct Asymmetric Mannich Reaction of Cyclic C-Acylimines with Simple Ketones: Facile Access to C2-Quaternary Indolin-3-ones", CHEM. COMMUN. 2018,54, 9151-9154, 24 July 2018 (2018-07-24), pages 1 - 92, XP093150661, Retrieved from the Internet <URL:https://www.rsc.org/suppdata/c8/cc/c8cc05125a/c8cc05125a1.pdf> [retrieved on 20240411] * |
| M. WARSITZ ET AL., EUR. J. ORG. CHEM., 2020, pages 6997 |
| Q. LI ET AL., ANGEW. CHEM. INT. ED., vol. 53, 2014, pages 8471 |
| SCHULTZ ET AL.: "Supporting Information for: Synthesis of Polycyclic Nitrogen Heterocycles via Alkene Aminopalladation/Carbopalladation Cascade Reactions", ORG. LETT. 2010, 12, 1028-2031, vol. 12, no. 5, 8 February 2010 (2010-02-08), US, pages S1 - S25, XP093150716, ISSN: 1523-7060, DOI: 10.1021/ol100033s * |
| SHERMAN, CHEMLER: "Copper(II)-Catalyzed Aminooxygenation and Carboamination of N-Aryl-2-allylanilines", ADV. SYNTH. CATAL., vol. 351, no. 3, 2 February 2009 (2009-02-02), pages 467 - 471, XP072352369, ISSN: 1615-4150, DOI: 10.1002/ADSC.200800705 * |
| SUNGHO NAM ET AL., ADV. SCI., 2021, pages 2100586 |
| W. DU ET AL., J. AM. CHEM. SOC., vol. 137, 2015, pages 1130 |
| WANG ET AL.: "Unsymmetric N-heterocyclic carbene ligand enabled nickel-catalysed arylation of bulky primary and secondary amines", CHEM. SCI., vol. 14, no. 16, 28 March 2023 (2023-03-28), United Kingdom, pages 4390 - 4396, XP093150566, ISSN: 2041-6520, DOI: 10.1039/D3SC00492A * |
| Z.-Y. LIU ET AL., ANGEW. CHEM. INT. ED., vol. 56, 2017, pages 5817 |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20250156170A (ko) | 2025-10-31 |
| EP4676924A1 (de) | 2026-01-14 |
| CN120787228A (zh) | 2025-10-14 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3227269B1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| EP3016952B1 (de) | Spirokondensierte lactamverbindungen für organische elektrolumineszenzvorrichtungen | |
| WO2021122740A1 (de) | Polycyclische verbindungen für organische elektrolumineszenzvorrichtungen | |
| WO2022129114A1 (de) | Stickstoffhaltige verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP4229064A1 (de) | Heterocyclische verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP4077335A1 (de) | Aromatische verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP4122028A1 (de) | Heterocyclische verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP4519383A1 (de) | Cyclische verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP4165052A1 (de) | Heterocyclische verbindungen für organische elektrolumineszenzvorrichtungen | |
| WO2022002772A1 (de) | Heteroaromatische verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP4121432A1 (de) | Heteroaromatische verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP4229145B1 (de) | Verbindungen mit heteroatomen für organische elektrolumineszenzvorrichtungen | |
| EP4330257B1 (de) | Stickstoffhaltige, heterocyclische verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP4402221B1 (de) | Borhaltige, heterocyclische verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP4423209B1 (de) | Bor- und stickstoffhaltige, heterocyclische verbindungen für organische elektrolumineszenzvorrichtungen | |
| WO2024184050A1 (de) | Cyclische stickstoffverbindungen für organische elektrolumineszenzvorrichtungen | |
| WO2024094592A2 (de) | Stickstoffhaltige heterocyclen für organische elektrolumineszenzvorrichtungen | |
| WO2025181124A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| WO2025181044A1 (de) | Stickstoffenthaltende verbindungen für organische elektrolumineszenzvorrichtungen | |
| WO2024153568A1 (de) | Heterocyclen für organische elektrolumineszenzvorrichtungen | |
| WO2025181097A1 (de) | Stickstoffenthaltende heterocyclen für organische elektrolumineszenzvorrichtungen | |
| WO2025109056A1 (de) | Sauerstoffenthaltende heterocyclen für organische elektrolumineszenzvorrichtungen | |
| WO2024149694A1 (de) | Stickstoffhaltige heterocyclen für organische elektrolumineszenzvorrichtungen | |
| WO2025021855A1 (de) | Materialien für organische lichtemittierende vorrichtungen und organische sensoren | |
| EP4222790A1 (de) | Verbindungen zur strukturierung von funktionalen schichten organischer elektrolumineszenzvorrichtungen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24706691 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202480015993.1 Country of ref document: CN |
|
| ENP | Entry into the national phase |
Ref document number: 1020257033287 Country of ref document: KR Free format text: ST27 STATUS EVENT CODE: A-0-1-A10-A15-NAP-PA0105 (AS PROVIDED BY THE NATIONAL OFFICE) |
|
| WWE | Wipo information: entry into national phase |
Ref document number: KR1020257033287 Country of ref document: KR Ref document number: 1020257033287 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2024706691 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWP | Wipo information: published in national office |
Ref document number: 202480015993.1 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020257033287 Country of ref document: KR |
|
| ENP | Entry into the national phase |
Ref document number: 2024706691 Country of ref document: EP Effective date: 20251007 |
|
| ENP | Entry into the national phase |
Ref document number: 2024706691 Country of ref document: EP Effective date: 20251007 |
|
| ENP | Entry into the national phase |
Ref document number: 2024706691 Country of ref document: EP Effective date: 20251007 |
|
| ENP | Entry into the national phase |
Ref document number: 2024706691 Country of ref document: EP Effective date: 20251007 |
|
| WWP | Wipo information: published in national office |
Ref document number: 2024706691 Country of ref document: EP |