WO2023152063A1 - Materialien für organische elektrolumineszenzvorrichtungen - Google Patents
Materialien für organische elektrolumineszenzvorrichtungen Download PDFInfo
- Publication number
- WO2023152063A1 WO2023152063A1 PCT/EP2023/052756 EP2023052756W WO2023152063A1 WO 2023152063 A1 WO2023152063 A1 WO 2023152063A1 EP 2023052756 W EP2023052756 W EP 2023052756W WO 2023152063 A1 WO2023152063 A1 WO 2023152063A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- radicals
- substituted
- aromatic
- aromatic ring
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B59/00—Introduction of isotopes of elements into organic compounds ; Labelled organic compounds per se
- C07B59/004—Acyclic, carbocyclic or heterocyclic compounds containing elements other than carbon, hydrogen, halogen, oxygen, nitrogen, sulfur, selenium or tellurium
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings
- C07D409/10—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing two hetero rings linked by a carbon chain containing aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/02—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains two hetero rings
- C07D491/04—Ortho-condensed systems
- C07D491/044—Ortho-condensed systems with only one oxygen atom as ring hetero atom in the oxygen-containing ring
- C07D491/048—Ortho-condensed systems with only one oxygen atom as ring hetero atom in the oxygen-containing ring the oxygen-containing ring being five-membered
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/622—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing four rings, e.g. pyrene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/615—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene
- H10K85/624—Polycyclic condensed aromatic hydrocarbons, e.g. anthracene containing six or more rings
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/654—Aromatic compounds comprising a hetero atom comprising only nitrogen as heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6574—Polycyclic condensed heteroaromatic hydrocarbons comprising only oxygen in the heteroaromatic polycondensed ring system, e.g. cumarine dyes
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6576—Polycyclic condensed heteroaromatic hydrocarbons comprising only sulfur in the heteroaromatic polycondensed ring system, e.g. benzothiophene
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07B—GENERAL METHODS OF ORGANIC CHEMISTRY; APPARATUS THEREFOR
- C07B2200/00—Indexing scheme relating to specific properties of organic compounds
- C07B2200/05—Isotopically modified compounds, e.g. labelled
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/15—Hole transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/16—Electron transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/18—Carrier blocking layers
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02E—REDUCTION OF GREENHOUSE GAS [GHG] EMISSIONS, RELATED TO ENERGY GENERATION, TRANSMISSION OR DISTRIBUTION
- Y02E10/00—Energy generation through renewable energy sources
- Y02E10/50—Photovoltaic [PV] energy
- Y02E10/549—Organic PV cells
Definitions
- the present invention describes connections, in particular for use in electronic devices.
- the invention also relates to a method for producing the compounds according to the invention and electronic devices containing these compounds.
- organic electroluminescent devices in which organic semiconductors are used as functional materials is described, for example, in US Pat. No. 4,539,507, US Pat.
- Organometallic complexes that show phosphorescence are often used as emitting materials.
- organometallic compounds as phosphorescence emitters.
- electroluminescent devices in particular also in electroluminescent devices which exhibit phosphorescence, for example with regard to efficiency, operating voltage and service life.
- organic electroluminescence devices are known which comprise fluorescent emitters or emitters which exhibit TADF (thermally activated delayed fluorescence).
- dibenzothiophene dioxides according to CN110790756 or KR2015/0031396 as well as according to WO2019/016430 are used as matrix materials for phosphorescent emitters or as electron transport materials.
- organic electroluminescent devices are not only determined by the emitters used.
- the other materials used are also here, such as matrix materials, hole-blocking materials, electron-transport materials, hole-transport materials and electron or exciton-blocking materials really important. Improvements in these materials can lead to significant improvements in electroluminescent devices.
- OLEDs containing the compounds should have high color purity.
- An object of the present invention is to provide compounds which are suitable for use in an organic electronic device, in particular in an organic electroluminescent device, as matrix materials or charge transport materials and which lead to good device properties when used in this device, and the provision the corresponding electronic device.
- a further object of the present invention can be seen as providing compounds which are suitable for use in a phosphorescent or fluorescent electroluminescent device, in particular as a matrix material.
- R* is a group of the following formula (2), where the dashed bond represents the bond to the backbone of formula (1),
- X is the same or different on each occurrence CR a or N or two adjacent groups X represent a group of the following formula
- Y is the same or different on each occurrence CR a or N;
- A is the same or different on each occurrence NR a , 0, S or CR a 2
- X 1 is the same or different on each occurrence of CR b or N with the
- a maximum of two groups X 1 represent N, and further provided that X 1 represents C when the group R* is attached to this X 1 ;
- X 2 is the same or different on each occurrence CR b or N with the proviso that a maximum of two X 2 groups are N;
- L is a single bond or an aromatic or heteroaromatic ring system having 5 to 24 aromatic ring atoms, which may be substituted with one or more R radicals;
- R 2 is selected identically or differently on each occurrence from the group consisting of H, D, F, CN, an aliphatic hydrocarbon radical having 1 to 20 carbon atoms or an aromatic or heteroaromatic ring system having 5 to 30 aromatic ring atoms, in which one or more H atoms can be replaced by D, F, CI, Br, I or CN and which can be substituted by one or more alkyl groups each having 1 to 4 carbon atoms, two or more, preferably adjacent, substituents R 2 together form a ring system.
- the present compounds can be used as an active compound in electronic devices.
- Active compounds are generally the organic or inorganic materials which are introduced, for example, in an organic electronic device, in particular in an organic electroluminescent device between anode and cathode, for example charge injection, charge transport or charge blocking materials, but in particular matrix materials.
- Organic materials are preferred here.
- Neighboring carbon atoms within the meaning of the present invention are carbon atoms which are linked directly to one another. Furthermore, in the definition of groups, "adjacent groups” means that these groups are attached to the same carbon atom or to adjacent carbon atoms are. These definitions apply accordingly, inter alia, to the terms “adjacent groups” and “adjacent substituents”.
- the above formulation should also be understood to mean that if one of the two radicals is hydrogen, the second radical binds to the position to which the hydrogen atom was bonded, forming a ring. This should be illustrated by the following scheme:
- a fused aryl group, a fused aromatic ring system or a fused heteroaromatic ring system in the context of the present invention is a group in which two or more aromatic groups are fused to one another via a common edge, ie fused, so that, for example, two carbon atoms form the belong to at least two aromatic or heteroaromatic rings, such as in naphthalene.
- fluorene for example, is not a fused aryl group in the context of the present invention, since the two aromatic groups in fluorene do not have a common edge.
- Corresponding definitions apply to heteroaryl groups as well as to fused ring systems, which can also contain heteroatoms, but do not have to.
- An electron-rich heteroaryl group within the meaning of the invention contains 5 to 30 aromatic ring atoms, preferably 5 to 24 aromatic ring atoms, very particularly preferably 5 to 14 aromatic ring atoms and is a group that conducts holes, preferably selected from the group of dibenzofurans, dibenzothiophenes, phenoxazines, Phenothiazines, carbazoles, bridged carbazoles, biscarbazoles, benzcarbazoles, indenocarbazoles, indolocarbazoles, benzofurocarbazoles, benzothioenocarbazoles, dihydroacridines, dihydrophenazines, dibenzodioxins, thianthrenes, phenoxathiines.
- radicals R, R a , R b and/or R 2 form a ring system with one another, a monocyclic or polycyclic, aliphatic, heteroaliphatic, aromatic or heteroaromatic ring system can result. If two radicals R 1 together form a ring system, a monocyclic or polycyclic aliphatic or heteroaliphatic ring system can result.
- An aryl group within the meaning of this invention contains 6 to 60 carbon atoms, preferably 6 to 40 carbon atoms, particularly preferably 6 to 30 carbon atoms;
- a heteroaryl group within the meaning of this invention contains 2 to 60 carbon atoms, preferably 2 to 40 carbon atoms, particularly preferably 2 to 30 carbon atoms and at least one heteroatom, with the proviso that the sum of carbon atoms and heteroatoms is at least 5 results.
- the heteroatoms are preferably selected from N, O and/or S.
- An aryl group or heteroaryl group is either a simple aromatic cycle, i.e. benzene, or a simple heteroaromatic cycle, for example pyridine, pyrimidine, thiophene, etc.
- An aromatic ring system within the meaning of this invention contains 6 to 60 carbon atoms, preferably 6 to 40 carbon atoms, particularly preferably 6 to 30 carbon atoms in the ring system.
- a heteroaromatic ring system within the meaning of this invention contains 1 to 60 C, preferably 1 to 40 C atoms, particularly preferably 1 to 30 C atoms and at least one heteroatom in the ring system, with the proviso that the sum of C atoms and heteroatoms is at least 5 results.
- the heteroatoms are preferably selected from N, O and/or S.
- An aromatic or heteroaromatic ring system in the context of this invention is to be understood as meaning a system which does not necessarily only contain aryl or heteroaryl groups, but also in which several aryl or heteroaryl groups a non-aromatic moiety (preferably less than 10% of the non-H atoms), such as e.g. B. a C, N or O atom or a carbonyl group can be interrupted.
- aryl or heteroaryl groups a non-aromatic moiety (preferably less than 10% of the non-H atoms), such as e.g. B. a C, N or O atom or a carbonyl group can be interrupted.
- systems such as 9,9 -spirobifluorene, 9,9-diarylfluorene, triarylamine, diaryl ether, stilbene, etc.
- aromatic ring systems in the context of this invention, as well as systems in which two or more aryl groups are replaced, for example, by a linear or cyclic alkyl group or are interrupted by a silyl group.
- systems in which two or more aryl or heteroaryl groups are bonded directly to each other such as.
- biphenyl, terphenyl, quaterphenyl or bipyridine also be understood as an aromatic or heteroaromatic ring system.
- a cyclic alkyl, alkoxy or thioalkoxy group in the context of this invention is understood as meaning a monocyclic, a bicyclic or a polycyclic group.
- a C1 to C20 alkyl group in which individual H atoms or CH2 groups can also be substituted by the groups mentioned above, for example the radicals methyl, ethyl, n-propyl, i-propyl, cyclopropyl, n-butyl, i-butyl, s-butyl, t-butyl, cyclobutyl, 2-methylbutyl, n-pentyl, s-pentyl, t-pentyl, 2-pentyl, neo-pentyl, cyclopentyl, n-hexyl, s-hexyl, t-hexyl, 2-hexyl, 3-hexyl, neo-hexyl, cyclohexyl, 1-methylcyclopentyl, 2-methylpentyl, n-heptyl, 2-heptyl, 3-heptyl, 4-h
- alkenyl group is understood to mean, for example, ethenyl, propenyl, butenyl, pentenyl, cyclopentenyl, hexenyl, cyclohexenyl, heptenyl, cycloheptenyl, octenyl, cyclooctenyl or cyclooctadienyl.
- An alkynyl group is understood to mean, for example, ethynyl, propynyl, butynyl, pentynyl, hexynyl, heptynyl or octynyl.
- a C1- to C40-alkoxy group is understood as meaning, for example, methoxy, trifluoromethoxy, ethoxy, n-propoxy, i-propoxy, n-butoxy, i-butoxy, s-butoxy, t-butoxy or 2-methylbutoxy.
- aromatic or heteroaromatic ring system with 5 to 60, preferably 5-40 aromatic ring atoms, particularly preferably 5 to 30 aromatic ring atoms, which can be substituted in each case with the abovementioned radicals and which can be linked via any positions on the aromatic or heteroaromatic , are understood, for example, groups derived from benzene, naphthalene, anthracene, benzanthracene, phenanthrene, benzophenanthrene, pyrene, chrysene, perylene, fluoranthene, benzfluoranthene, naphthacene, pentacene, benzopyrene, biphenyl, biphenylene, terphenyl, terphenylene, fluorene, spirobifluorene, dihydrophenanthrene, dihydropyrene, tetrahydro- pyrene, cis or trans indenofluorene, cis or trans mono
- Preferred compounds for the purposes of the invention are compounds of the formula (1) in which a maximum of two of the symbols X 1 and X 2 are N.
- a maximum of one of the symbols X 1 and X 2 is particularly preferably N, very particular preference is given to compounds of the formula (6) in which all the symbols X 1 and X 2 are CR b ,
- a preferred embodiment of the invention are compounds of the formulas (6-1) to (6-4). Compounds of the formula (6-1), (6-2) and (6-3) are particularly preferred. Very particular preference is given to compounds of the formula (6-1) and of the formula (6-3).
- a maximum of two radicals R b are a group different from H or D. Particularly preferably, a maximum of one radical R b or none of the radicals R b is a group different from H or D.
- the compounds are preferred selected from compounds of the formula (6-1a) to (6-4f), particularly preferably from the compounds of the formula (6-1a) to (6-3e) and very particularly preferably from the compounds of the formula (6-1 a) to (6-1 e) and the formula (6-3a) to (6-3e),
- all Xs identically or differently, represent CR or N, with the proviso that at least one X and at most three Xs represent N.
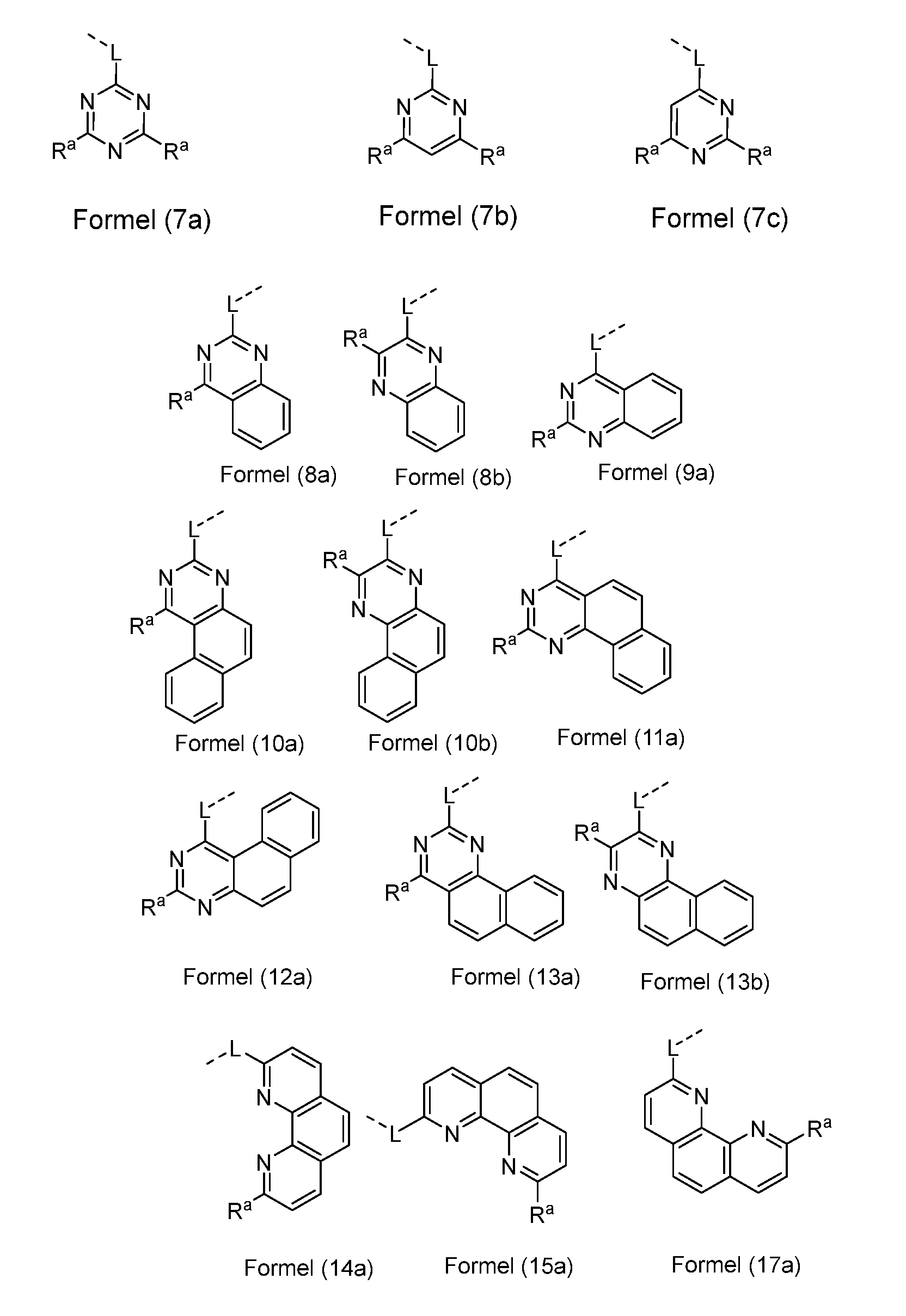
- These are preferably structures of the following formula (7), where the symbols used have the meanings given above, 1, 2 or 3 X stand for N and R a is preferably identical or different on each occurrence for an aromatic or heteroaromatic ring system having 5 to 40 aromatic ring atoms which is linked to one or more radicals R 1 may be substituted.
- Preferred embodiments of the formula (7) are the groups of the following formulas (7a), (7b) and (7c), the groups of the formula (7a) being particularly preferred where the symbols used have the meanings given above and R a is preferably identical or different on each occurrence for an aromatic or heteroaromatic ring system having 5 to 40 aromatic ring atoms which can be substituted by one or more R 1 radicals.
- R a is preferably identical or different on each occurrence for an aromatic or heteroaromatic ring system having 5 to 40 aromatic ring atoms which can be substituted by one or more R 1 radicals.
- two adjacent Xs stand for a group of the formula (3) or (4), where Y is identical or different for CR a , and of the remaining Xs exactly two Xs stand for N and that third X for CR a , so that it is a structure according to one of the following formulas (8) to (13),
- two adjacent Xs stand for a group of the formula (4), where exactly one group Y stands for N and the remaining Y stands for CR a , and exactly one group X stands for N and the remaining X are CR a , so that it is a structure according to one of the formulas (14) to (17), wherein the Symbols have the meanings listed above and exactly one group X and exactly one group Y is N.
- two adjacent Xs are a group of the formula (5), where Y is identical or different for CR a , and of the remaining Xs exactly two Xs are N and the third X is CR a , so that it is a structure according to one of the following formulas (18) to (21), where the symbols have the meanings listed above and exactly two groups X are N.
- Preferred embodiments of the formulas (8) to (21) are the structures of the following formulas (8a) to (21a),
- R a is preferably identical or different on each occurrence for H, D or for an aromatic or heteroaromatic ring system with 5 bis 40 aromatic ring atoms, which may be substituted by one or more R 1 radicals
- R a is identical or different on each occurrence and is H, D or an aromatic or heteroaromatic ring system having 6 to 30 aromatic ring atoms , which may be substituted by one or more R 1 radicals.
- R a is particularly preferably the same or different on each occurrence, H, D or an aromatic or heteroaromatic ring system having 6 to 24 aromatic ring atoms, in particular having 6 to 13 aromatic ring atoms, which is substituted by one or more, preferably non-aromatic, R 1 radicals may be substituted.
- R a is very particularly preferably selected from H, D, phenyl, ds-phenyl, meta- or para-biphenyl, dibenzofuran or carbazole, these groups each being replaced by one or more radicals R 1 can be substituted, but are preferably unsubstituted.
- A is NR a , 0 or S, in particular 0 or NR a .
- the group L represents a single bond or a bivalent aromatic or heteroaromatic ring system having 6 to 18 aromatic ring atoms, which can each be substituted by one or more R radicals.
- L particularly preferably represents a single bond or an aromatic ring system having 6 to 12 aromatic ring atoms which can be substituted by one or more R radicals, or a dibenzofuran or dibenzothiophene group which can be substituted by one or more R radicals.
- L is a single bond, a meta- or para-linked phenylene group which may be substituted by one or more R radicals, or a dibenzofuran or dibenzothiophene group, each of which may be substituted by one or more R radicals.
- the dibenzofuran or dibenzothiophene group is preferably linked via the 1,3, 1,6, 1,7, 1,8, 3,6, 3,8 or 3,9 position.
- L can be a dibenzofuran or dibenzothiophene group applies in particular when the heteroaryl group of the radical R* is a group of formula (7).
- L is a single bond or a meta- or para-linked phenylene group or a dibenzofuran group, each of which can be substituted by one or more R radicals, where the R group is preferably H or D, and the R group is very particularly preferably for H. If L stands for an aromatic or heteroaromatic ring system, this is preferably selected from the structures of the following formulas (L-1) to (L-34),
- L particularly preferably represents a single bond, an optionally substituted phenylene or dibenzofuran group, i.e. a group of the formula (L-1) to (L-3) or (L19) to (L26), in particular (L-1), (L -2) or (L-19) to (L26).
- R, R a , R b , R 1 and R 2 on the compounds according to the invention are described below.
- the preferences given below for R, R a , R b , R 1 and R 2 occur simultaneously and apply to the structures of the formula (1) and to all preferred embodiments.
- R is selected identically or differently on each occurrence from the group consisting of H, D, F, CN, OR 1 , a straight-chain alkyl group having 1 to 10 carbon atoms or an alkenyl group having 2 to 10 C atoms or a branched or cyclic alkyl group with 3 to 10 C atoms, it being possible for the alkyl or alkenyl group to be substituted with one or more radicals R 1 in each case, but it is preferably unsubstituted, and one or more non-adjacent CH 2 -groups can be replaced by 0, or an aroma- matic ring system having 6 to 30 aromatic ring atoms, each of which may be substituted by one or more R 1 radicals, or an electron-rich heteroaryl group having 5 to 30 aromatic ring atoms, each of which may be substituted by one or more R 1 radicals; two radicals R can also form an aliphatic, aromatic or heteroaromatic ring system with one another.
- R is particularly preferably selected identically or differently on each occurrence from the group consisting of H, D, a straight-chain alkyl group having 1 to 6 carbon atoms, in particular having 1, 2, 3 or 4 carbon atoms, or a branched or cyclic alkyl group having 3 to 6 carbon atoms, where each alkyl group may be substituted by one or more radicals R 1 , but is preferably unsubstituted, or an aromatic ring system having 6 to 24 aromatic ring atoms, each of which is substituted by one or more radicals R 1 , preferably non-aromatic radicals R 1 , can be substituted or an electron-rich heteroaryl group having 5 to 24 aromatic ring atoms, which can each be substituted by one or more radicals R 1 , preferably non-aromatic radicals R 1 .
- R is very particularly preferably selected on each occurrence, identically or differently, from the group consisting of H, D or an aromatic ring system having 6 to 14 aromatic ring atoms, each of which is substituted by one or more radicals R1, preferably non-aromatic radicals R1 or an electron-rich heteroaryl group with 5 to 24 aromatic ring atoms, which can each be substituted by one or more radicals R 1 , preferably non-aromatic radicals R 1 .
- R is particularly preferably selected on each occurrence, identically or differently, from the group consisting of H, D or an aromatic ring system having 6 to 14 aromatic ring atoms, which is substituted in each case by one or more radicals R 1 , preferably non-aromatic radicals R 1 can be, or an electron-rich heteroaryl group having 5 to 14 aromatic ring atoms, each of which can be substituted by one or more radicals R 1 , preferably non-aromatic radicals R 1 .
- R a is selected identically or differently on each occurrence from the group consisting of H, D, F, CN, OR 1 , a straight-chain alkyl group having 1 to 10 carbon atoms or an alkenyl group having 2 to 10 carbon atoms or a branched or cyclic alkyl group having 3 to 10 carbon atoms, where the alkyl or alkenyl group may be substituted by one or more radicals R 1 , but is preferably unsubstituted, and where one or more non-adjacent CH2 groups can be replaced by O, or an aromatic or heteroaromatic ring system having 6 to 30 aromatic ring atoms, each of which can be substituted by one or more radicals R 1 ; two radicals R a can also form an aliphatic, aromatic or heteroaromatic ring system with one another.
- R a is particularly preferably selected identically or differently on each occurrence from the group consisting of H, D, a straight-chain alkyl group having 1 to 6 carbon atoms, in particular having 1, 2, 3 or 4 carbon atoms, or a branched one or cyclic alkyl group having 3 to 6 carbon atoms, where each alkyl group may be substituted by one or more radicals R 1 , but is preferably unsubstituted, or an aromatic or heteroaromatic ring system having 6 to 24 aromatic ring atoms, each of which is substituted by one or more R 1 radicals, preferably non-aromatic R 1 radicals, may be substituted.
- R a is selected identically or differently on each occurrence from the group consisting of H, D or an aromatic or heteroaromatic ring system having 6 to 24 aromatic ring atoms, each of which is substituted by one or more radicals R 1 , preferably non-aromatic radicals R 1 may be substituted.
- R a is particularly preferably selected identically or differently on each occurrence from the group consisting of H, D or an aromatic or heteroaromatic ring system having 6 to 14 aromatic ring atoms, each of which is substituted by one or more radicals R 1 , preferably non-aromatic radicals R 1 may be substituted.
- the substituents R b on the dibenzothiophene dioxide base are preferably selected identically or differently on each occurrence from a group consisting of H, D, F, CN, OR 1 , NR 1 , a straight-chain alkyl group having 1 to 10 carbon atoms or an alkenyl group having 2 up to 10 carbon atoms or a branched or cyclic alkyl group having 3 to 10 carbon atoms, wherein the alkyl or alkenyl group may be substituted with one or more radicals R 1 , but is preferably unsubstituted, and one or more non-adjacent CH2 groups can be replaced by 0, or an aromatic ring system having 6 to 16 aromatic ring atoms, each of which can be substituted by one or more radicals R 1 , or a heteroaryl group having 5 to 30 aromatic ring atoms, which has a C Atom is bonded to the dibenzothiophendioxide backbone, each of which may be substituted by one or more R 1
- R b is particularly preferably selected identically or differently on each occurrence from the group consisting of H, D, a straight-chain alkyl group having 1 to 6 carbon atoms, in particular having 1, 2, 3 or 4 carbon atoms, or a branched one or cyclic alkyl group with 3 to 6 carbon atoms, where the alkyl group can be substituted by one or more radicals R 1 , but is preferably unsubstituted, or an aromatic ring system with 6 to 16 aromatic ring atoms, each of which is substituted by one or more radicals R 1 , preferably non-aromatic radicals R 1 , may be substituted, or a heteroaryl group having 5 to 24 aromatic ring atoms, each of which may be substituted by one or more radicals R 1 , preferably non-aromatic radicals R 1 , wherein the heteroaromatic ring system is bound to the dibenzothiophene dioxide backbone via a carbon atom.
- R b is very particularly preferably selected on each occurrence, identically or differently, from the group consisting of H, D or an aromatic ring system having 6 to 16 aromatic ring atoms, each of which is replaced by one or more radicals R 1 , preferably non-aromatic radicals R 1 , may be substituted, or a heteroaryl group having 5 to 24 aromatic ring atoms, each of which may be substituted by one or more radicals R 1 , preferably non-aromatic radicals R 1 , where the heteroaryl group is bonded to the basic structure of dibenzothiophene dioxide via a carbon atom.
- the non-aromatic radicals R 1 are particularly preferably H or D.
- Suitable aromatic or heteroaromatic ring systems R are selected from phenyl, biphenyl, in particular ortho-, meta- or para-biphenyl, terphenyl, in particular ortho-, meta-, para- or branched terphenyl, quaterphenyl, in particular ortho-, meta- , Para- or branched quaterphenyl, fluorene, which can be linked via the 1-, 2-, 3- or 4-position, spirobifluorene, which can be linked via the 1-, 2-, 3- or 4-position , naphthalene, which can be linked via the 1- or 2-position, indole, benzofuran, benzothiophene, carbazole, which can be linked via the 1-, 2-, 3- or 4-position or the N atom, di- benzofuran, which can be linked via the 1-, 2-, 3- or 4-position, dibenzothiophene, which can be linked via the 1-, 2-, 3- or 4-position, indenocarbazole, ind
- Suitable aromatic or heteroaromatic ring systems R a are selected from phenyl, biphenyl, in particular ortho-, meta- or para-biphenyl, terphenyl, in particular ortho-, meta-, para- or branched terphenyl, quaterphenyl, in particular ortho-, meta -, para- or branched quaterphenyl, fluorene, which can be linked via the 1-, 2-, 3- or 4-position, spirobifluorene, which can be linked via the 1-, 2-, 3- or 4-position can, naphthalene, which can be linked via the 1- or 2-position, indole, benzofuran, benzothiophene, carbazole, which can be linked via the 1-, 2-, 3- or 4-position or the N atom, di - benzofuran, which can be linked via the 1-, 2-, 3- or 4-position, dibenzothiophene, which can be linked via the 1-, 2-, 3- or 4-position, indenocarbazole
- Suitable aromatic or heteroaromatic ring systems R b are selected from phenyl, biphenyl, in particular ortho-, meta- or para-biphenyl, fluorene, which can be linked via the 1-, 2-, 3- or 4-position, and naphthalene Which can be linked via the 1 - or 2-position, indole, benzofuran, benzothiophene, carbazole, which can be linked via a carbon atom to the dibenzothiophene dioxide base, dibenzofuran, which via the 1-, 2-, 3- or 4-position can be linked, dibenzothiophene, which can be linked via the 1-, 2-, 3- or 4-position, indenocarbazole, indolocarbazole which are bonded to the dibenzothiophene dioxide base structure via a C atom, pyridine, pyrimidine, Pyrazine, pyridazine, triazine, quinoline, quinazoline, benzimidazole, phenanthrene
- the groups R a in the formulas (7) to (21a) are preferably selected from the groups of the formulas R1 to R83 if they represent an aromatic or heteroaromatic ring system.
- the groups R if they represent an aromatic or heteroaromatic ring system, are preferably selected from the groups of the formulas R-1 to R-46 and R-67 to R-75.
- the groups R b if they represent an aromatic or heteroaromatic ring system, are preferably selected from the groups of the formulas R-1 to R-4, R12 to R-42, R-47 to R74 and R-76 to R- 83
- R 1 has the meanings given above, the dashed bond represents the position of the bond of the group and the following also applies:
- Ar is the same or different on each occurrence and is a divalent aromatic or heteroaromatic ring system having 6 to 18 aromatic ring atoms, which can be substituted by one or more R 1 radicals;
- groups R-1 to R-83 mentioned above have several groups A 1 , then all combinations from the definition of A 1 are suitable for this. Preferred embodiments are then those in which one group A 1 is NR 1 and the other group A 1 is C(R 1 ) 2 or in which both groups A 1 are NR 1 or in which both groups A 1 are 0 . In a particularly preferred embodiment of the invention, in groups R-1 to R-83 which have several groups A 1 , at least one group A 1 is 0 or NR 1 .
- the substituent R 1 which is bonded to the nitrogen atom is preferably an aromatic or heteroaromatic ring system having 5 to 24 aromatic ring atoms, which can also be substituted by one or more R 2 radicals.
- this substituent R 1 is identical or different on each occurrence for an aromatic or heteroaromatic ring system having 6 to 24 aromatic ring atoms, preferably having 6 to 12 aromatic ring atoms, which has no fused aryl groups or heteroaryl groups in which two or more aromatic or heteroaromatic 6-ring groups are fused directly to one another, and which can each also be substituted by one or more R 2 radicals.
- phenyl, biphenyl, terphenyl and quaterphenyl with linkage patterns as above for R-1 to R-11 listed these structures may be substituted by one or more radicals R 2 , but are preferably unsubstituted.
- a 1 is C(R 1 ) 2
- the substituents R 1 which are bonded to this carbon atom are preferably identical or different on each occurrence for a linear alkyl group having 1 to 10 carbon atoms or for a branched or cyclic alkyl group having 3 to 10 carbon atoms or an aromatic or heteroaromatic ring system having 5 to 24 aromatic ring atoms, which can also be substituted by one or more R 2 radicals.
- R 1 is very particularly preferably a methyl group or a phenyl group.
- the radicals R 1 can also form a ring system with one another, which leads to a spiro system.
- R 1 is the same or different on each occurrence selected from the group consisting of H, D, F, CN, OR 2 , a straight-chain alkyl group having 1 to 10 carbon atoms or an alkenyl group having 2 to 10 C atoms or a branched or cyclic alkyl group having 3 to 10 C atoms, it being possible for the alkyl or alkenyl group to be substituted in each case with one or more R 2 radicals and for one or more non-adjacent CH 2 groups to be replaced by O can be, or an aromatic or heteroaromatic ring system having 6 to 30 aromatic ring atoms, each of which can be substituted by one or more radicals R 2 ; two or more R 2 radicals can form an aliphatic, heteroaliphatic, aromatic or heteroaromatic ring system with one another.
- R 1 is identical or different on each occurrence selected from the group consisting of H, D, a straight-chain alkyl group having 1 to 6 carbon atoms, in particular having 1, 2, 3 or 4 carbon atoms, or a branched or cyclic alkyl group having 3 to 6 carbon atoms, where the alkyl group can be substituted with one or more radicals R 2 , but is preferably unsubstituted, or an aromatic or heteroaromatic ring system having 6 to 24 aromatic ring atoms, preferably having 6 to 13 aromatic ring atoms, which may each be substituted by one or more R 2 radicals, but is preferably unsubstituted.
- R 1 represents an aromatic or heteroaromatic ring system, it is preferably selected from the structures (R-1) to (R-83) shown above, these structures then being substituted by R 2 instead of R 1 .
- R 2 is identical or different on each occurrence of H, D, F, an alkyl group having 1 to 4 carbon atoms or an aryl group having 6 to 10 carbon atoms, which is bonded to an alkyl group having 1 to 4 carbon atoms C atoms may be substituted, but is preferably unsubstituted.
- the alkyl groups in compounds according to the invention which are processed by vacuum evaporation preferably have no more than five carbon atoms, particularly preferably no more than 4 carbon atoms, very particularly preferably no more than 1 carbon atom.
- the compounds of the formula (1) or the preferred embodiments are used as matrix material for a phosphorescent emitter or in a layer which is directly adjacent to a phosphorescent layer, it is also preferred if the compound does not contain any condensed aryl or Contains heteroaryl groups in which more than two six-membered rings are fused directly to one another.
- the groups R, R a , R b , R 1 and R 2 do not contain any fused aryl or heteroaryl groups in which two or more six-membered rings are fused directly to one another. Exceptions to this are phenanthrene, triphenylene, quinazoline and quinoxaline, which due to their higher triplet energy may be preferred despite the presence of fused aromatic six-membered rings.
- a further subject of the present invention is therefore a process for the preparation of the compounds according to the invention, characterized by the following steps:
- formulations are compounds according to the invention required.
- a further subject of the present invention are therefore formulations containing at least one compound of the formula (1) or the preferred embodiments and at least one solvent.
- These formulations can be, for example, solutions, dispersions or emulsions. It may be preferable to use mixtures of two or more solvents for this.
- Suitable and preferred solvents are, for example, toluene, anisole, o-, m- or p-xylene, methyl benzoate, mesitylene, tetralin, veratrol, THF, methyl THF, THP, chlorobenzene, dioxane, phenoxytoluene, in particular 3-phenoxytoluene, ( -)-fenchone, 1,2,3,5-tetramethylbenzene, 1,2,4,5-tetramethylbenzene, 1-methyl-naphthalene, 2-methylbenzothiazole, 2-phenoxyethanol, 2-pyrrolidinone, 3-methylanisole, 4-methylanisole , 3,4-dimethylanisole, 3,5-dimethylanisole, acetophenone, terpineol, benzothiazole, butyl benzoate, cumene, cyclohexanol, cyclohexanone, cyclohexylbenzene, decalin, do
- the compounds of the formula (1) or of the preferred embodiments listed above are used according to the invention in an electronic device, in particular in an organic electroluminescent device.
- a further subject matter of the present invention is therefore the use of the compounds of the formula (1) or the preferred embodiments in an electronic device, in particular in an OLED.
- Yet another subject matter of the present invention is an electronic device, in particular an organic electroluminescent device containing at least one compound according to the invention.
- An electronic device within the meaning of the present invention is a device which contains at least one layer which contains at least one organic compound. In this case, the component can also contain inorganic materials or also layers which are made up entirely of inorganic materials.
- the electronic device is preferably selected from the group consisting of organic electroluminescent devices (OLEDs), organic integrated circuits (O-ICs), organic field effect transistors (O-FETs), organic thin film transistors (0 TFTs), organic light emitting transistors (0 LETs), organic solar cells (0 SCs), dye-sensitized organic solar cells (DSSCs), organic optical detectors, organic photoreceptors, organic field quench devices (0 FQDs), light-emitting electrochemical cells (LECs). ), organic laser diodes (0 lasers) and organic plasmon emitting devices, but preferably organic electroluminescent devices (OLEDs), particularly preferably phosphorescent OLEDs.
- OLEDs organic electroluminescent devices
- O-ICs organic integrated circuits
- O-FETs organic field effect transistors
- TFTs organic thin film transistors
- organic light emitting transistors (0 LETs
- organic solar cells (0 SCs), dye-sensitized organic solar cells (DSSCs), organic optical detectors,
- the organic electroluminescent device contains cathode, anode and at least one emitting layer. In addition to these layers, it can also contain further layers, for example one or more hole-injection layers, hole-transport layers, hole-blocking layers, electron-transport layers, electron-injection layers, exciton-blocking layers, electron-blocking layers and/or charge-generation layers. Likewise, interlayers can be introduced between two emitting layers, which have an exciton-blocking function, for example. However, it should be pointed out that each of these layers does not necessarily have to be present. In this case, the organic electroluminescent device contain an emissive layer, or it may contain multiple emissive layers.
- a plurality of emission layers are present, these preferably have a total of a plurality of emission maxima between 380 nm and 750 nm, resulting in white emission overall, ie different emitting compounds which can fluoresce or phosphorescence are used in the emitting layers.
- Systems with three emitting layers are particularly preferred, with the three layers showing blue, green and orange or red emission.
- the organic electroluminescence device according to the invention can also be a tandem OLED, in particular for white-emitting OLEDs.
- connection according to the embodiments listed above can be used in different layers, depending on the precise structure. Preference is given to an organic electroluminescent device containing a compound of the formula (1) or the preferred embodiments outlined above in an emitting layer as matrix material for phosphorescent or fluorescent emitters or for emitters which show TADF (thermally activated delayed fluorescence), in particular special as a matrix material for phosphorescent emitters.
- the organic electroluminescent device can contain an emitting layer or it can contain a plurality of emitting layers, with at least one emitting layer containing at least one compound according to the invention as matrix material.
- the compound according to the invention can also be used in an electron transport layer and/or in a hole-blocking layer.
- the compound is used as matrix material for a phosphorescent compound in an emitting layer, it is preferably used in combination with one or more phosphorescent materials (triplet emitters).
- phosphorescent materials triplet emitters.
- all luminescent complexes with transition metals or lanthanides, in particular all indium, platinum and copper complexes are to be regarded as phosphorescent compounds.
- the mixture of the compound of the formula (1) or the preferred embodiments and the emitting compound contains between 99 and 1% by volume, preferably between 98 and 10% by volume, particularly preferably between 97 and 60% by volume, in particular between 95 and 80% by volume of the compound of the formula (1) or of the preferred embodiments, based on the total mixture of emitter and matrix material.
- the mixture contains between 1 and 99% by volume, preferably between 2 and 90% by volume, particularly preferably between 3 and 40% by volume, in particular between 5 and 20% by volume, of the emitter, based on the total mixture emitter and matrix material.
- a further preferred embodiment of the present invention is the use of the compound of the formula (1) or the preferred embodiments as matrix material for a phosphorescent emitter in combination with a further matrix material.
- Suitable matrix materials which can be used in combination with the compounds according to the invention are aromatic ketones, aromatic phosphine oxides or aromatic sulfoxides or sulfones, e.g. B. according to WO 2004/013080, WO 2004/093207, WO 2006/005627 or WO 2010/006680, triarylamines, carbazole derivatives, z. B.
- CBP N, N-bis-carbazolylbiphenyl
- WO 2005/039246 US 2005/0069729, JP 2004/288381
- EP 1205527 WO 2008/086851 or WO 2013/041176, indolocarbazole derivatives, z. B. according to WO 2007/063754 or WO 2008/056746, indenocarbazole derivatives, z. B. according to WO 2010/136109, WO 2011/000455, WO 2013/041176 or WO 2013/056776, azacarbazole derivatives, z. B. according to EP 1617710, EP 1617711, EP 1731584, JP 2005/347160, bipolar matrix materials, z. B.
- silanes e.g. B. according to WO 2005/111172, azaboroles or boron esters, z. B. according to WO 2006/117052, triazine derivatives, z. according to WO 2007/063754, WO 2008/056746, WO 2010/015306, WO 2011/057706, WO 2011/060859 or WO 2011/060877, zinc complexes, e.g. B. according to EP 652273 or WO 2009/062578, diazasilol or tetraazasilol derivatives, z. B. according to WO 2010/054729, diazaphosphole derivatives, z. B.
- WO 2010/054730 bridged carbazole derivatives, z. B. according to WO 2011/042107, WO 2011/060867, WO 2011/088877 and WO 2012/143080, triphenylene derivatives, z. B. according to WO 2012/048781, or dibenzofuran derivatives, z. according to WO 2015/169412, WO 2016/015810, WO 2016/023608, WO 2017/148564 or WO 2017/148565.
- another phosphorescent emitter which emits at a shorter wavelength than the actual emitter, can be present as a co-host in the mixture, or a compound that does not participate, or does not participate to a significant extent, in charge transport, as described, for example, in WO 2010/108579.
- the materials are used in combination with another matrix material.
- the compounds of the formula (1) or the preferred embodiments are electron-poor compounds.
- Preferred co-matrix materials are therefore hole-transporting compounds, which are preferably selected from the group of arylamine or carbazole derivatives.
- Preferred biscarbazoles are the structures of the following formulas (22) to (28),
- a 1 has the meanings given above and Ar 1 is selected identically or differently on each occurrence from an aromatic or heteroaromatic ring system having 5 to 40 aromatic ring atoms, which may be substituted by one or more R 1 radicals.
- a 1 is NR 1 or C(R 1 ) 2 .
- Preferred embodiments of R 1 are the embodiments for R 1 mentioned above in the definition of A 1 .
- Preferred embodiments of Ar 1 are the preferred structures listed above for aromatic or heteroaromatic radicals R, in particular the groups (R-1) to (R-83).
- Preferred embodiments of the compounds of the formulas (22) to (28) are the compounds of the following formulas (22a) to (28a),
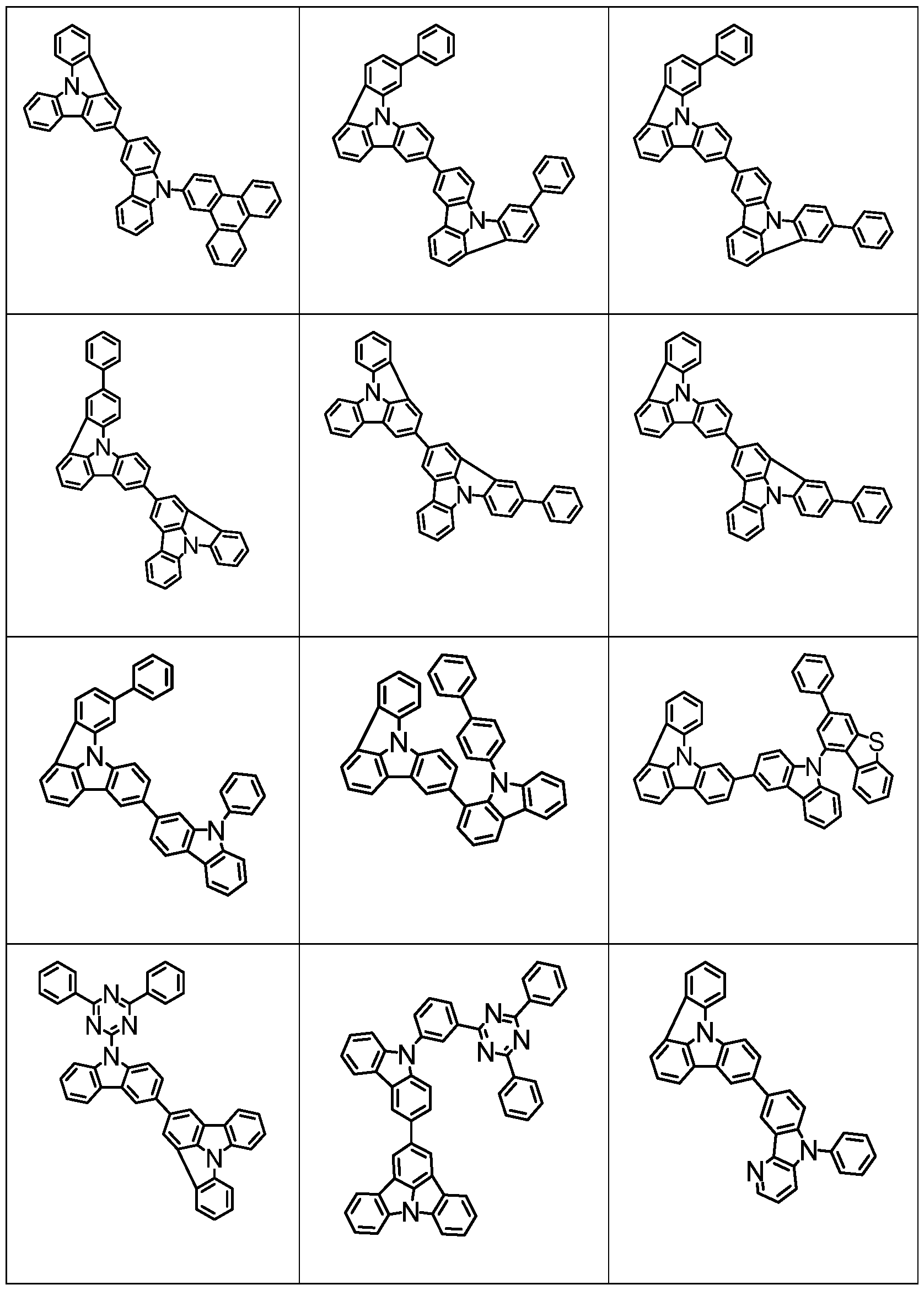
- Examples of suitable compounds of the formulas (22) to (28) are the compounds shown below.
- Preferred bridged carbazoles are the structures of the following formula (29), where A 1 and R have the meanings given above and A 1 is preferably selected identically or differently on each occurrence from the group consisting of NR 1 , 0, S or C(R 1 ) 2 where R 1 is an aromatic or heteroaromatic ring system 5 to 24 aromatic ring atoms, which may be substituted by one or more R 2 radicals.
- Preferred dibenzofuran derivatives are the compounds of the following
- L, R and Ar 1 have the meanings given above.
- the two groups Ar 1 which bind to the same nitrogen atom, or one group Ar 1 and one group L, which bind to the same nitrogen atom, can also be connected to one another, for example to form a carbazole.
- Examples of suitable dibenzofuran derivatives are the compounds shown below.
- Preferred carbazolamines are the structures of the following formulas (31), (32) and (33),
- Examples of suitable carbazolamine derivatives are the compounds shown below.
- Particularly suitable phosphorescent compounds are compounds which, when suitably excited, emit light, preferably in the visible range, and also at least one atom with an atomic number greater than 20, preferably greater than 38 and less than 84, particularly preferably greater than 56 and less than 80 included, in particular a metal with this atomic number.
- Compounds containing copper, molybdenum, tungsten, rhenium, ruthenium, osmium, rhodium, indium, palladium, platinum, silver, gold or europium are preferably used as phosphorescence emitters, in particular compounds containing indium or platinum.
- Examples of the emitter described above can be registered where 00/70655, where 2002/02714, WO 2002/15645, EP 1191612, EP 1191614, WO 05/019373, US 2005/ 0258742, WO 2009/146770, WO 2010/015307, WO 2010/031485, WO 2010/054731, WO 2010/054728, WO 2010/086089, WO 2010/099852, WO 2010/102709, WO 2011/032626, WO 2011/ 066898, WO 2011/157339, WO 2012/007086, WO 2014/008982, WO 2014/023377, WO 2014/094961, WO 2014/094960, WO 2015/036074, WO 2015/104045, WO 2015/117718, WO 2016/01 5815, WO 2016/124304, WO 2017/032439, WO 2018/011186 and WO 2018/041769, WO 2019/020538, WO 2018
- Examples of phosphorescent dopants are listed below.
- organic electroluminescent device In the further layers of the organic electroluminescent device according to the invention it is possible to use all the materials which are customarily used in accordance with the prior art. The person skilled in the art can therefore use all materials known for organic electroluminescent devices in combination with the compounds of the formula (1) or the preferred embodiments described above without any inventive step. Also preferred is an organic electroluminescent device, characterized in that one or more layers are coated using a sublimation process. The materials are vapour-deposited in vacuum sublimation systems at an initial pressure of less than 10 -5 mbar, preferably less than 10 -6 mbar. However, it is also possible for the initial pressure to be even lower, for example less than 10 -7 mbar.
- An organic electroluminescent device is also preferred, characterized in that one or more layers are coated using the OVPD (organic vapor phase deposition) method or with the aid of carrier gas sublimation.
- the materials are applied at a pressure between 10 -5 mbar and 1 bar.
- OVPD organic vapor phase deposition
- a special case of this process is the OVJP (Organic Vapor Jet Printing) process, in which the materials are applied directly through a nozzle and thus structured.
- an organic electroluminescent device characterized in that one or more layers of solution, such as. B. by spin coating, or with any printing method, such as. B. screen printing, flexographic printing, offset printing, LITI (Light Induced Thermal Imaging, thermal transfer printing), ink-jet printing (ink jet printing) or nozzle printing.
- any printing method such as. B. screen printing, flexographic printing, offset printing, LITI (Light Induced Thermal Imaging, thermal transfer printing), ink-jet printing (ink jet printing) or nozzle printing.
- Hybrid processes are also possible, in which, for example, one or more layers are applied from solution and one or more further layers are vapor-deposited.
- OLEDs containing the compounds of the formula (1) lead to high efficiencies. This applies in particular when the compounds are used as matrix material for a phosphorescent emitter. In particular, the compounds show improved efficiency compared to OLEDs with matrix materials that have a dibenzothiophene in the backbone instead of a dibenzothiophene dioxide.
- OLEDs containing the compounds of the formula (1) lead to low operating voltages. This applies in particular when the compounds are used as matrix material for a phosphorescent emitter. In particular, the compounds show a lower operating voltage compared to OLEDs with matrix materials that have a dibenzothiophene in the basic structure instead of a dibenzothiophene dioxide.
- OLEDs containing the compounds of the formula (1) as matrix material for phosphorescent emitters lead to long lifetimes. This applies in particular when the compounds are used as matrix material for a phosphorescent emitter.
- the compounds according to the invention can also be used with very good properties in an electron transport layer, also in combination with a fluorescent emission layer, or in a hole-blocking layer.
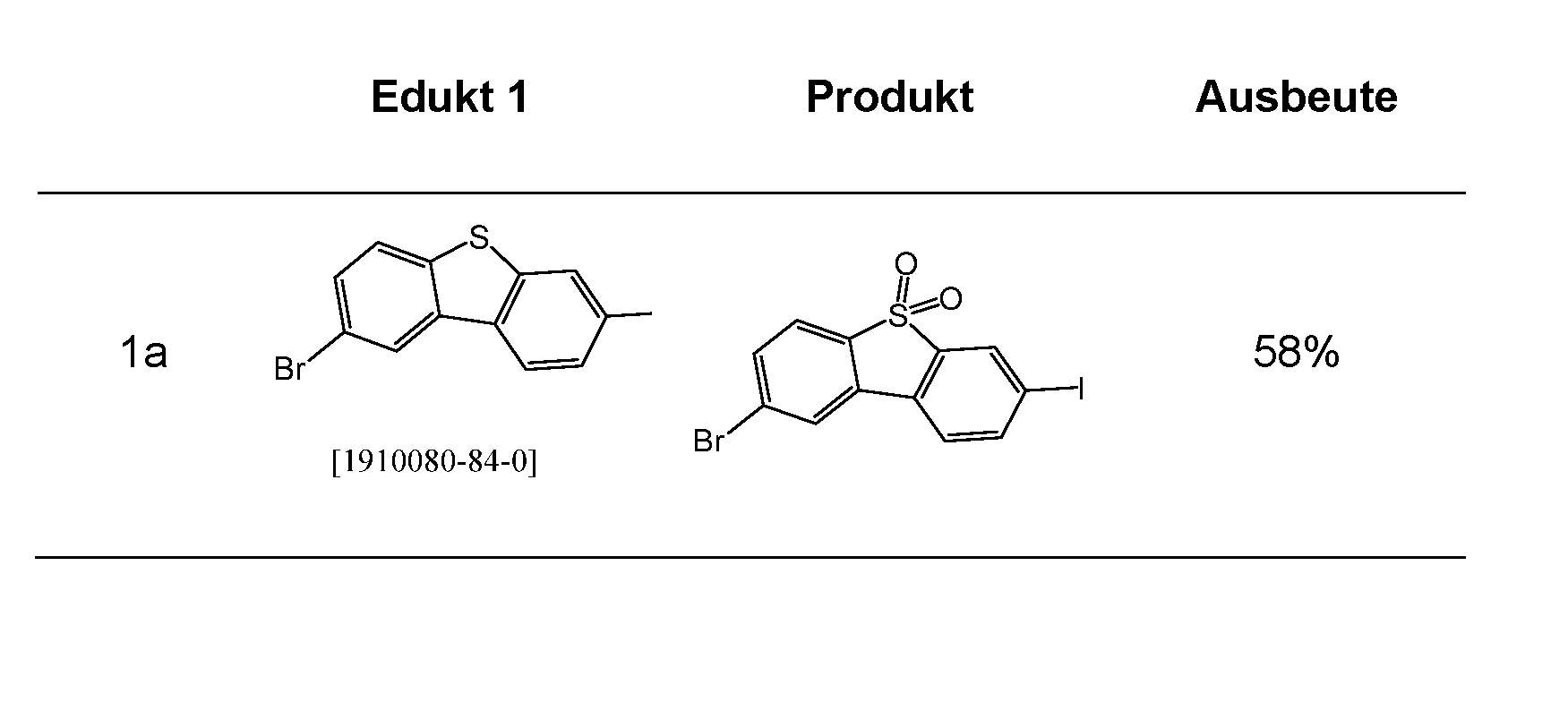
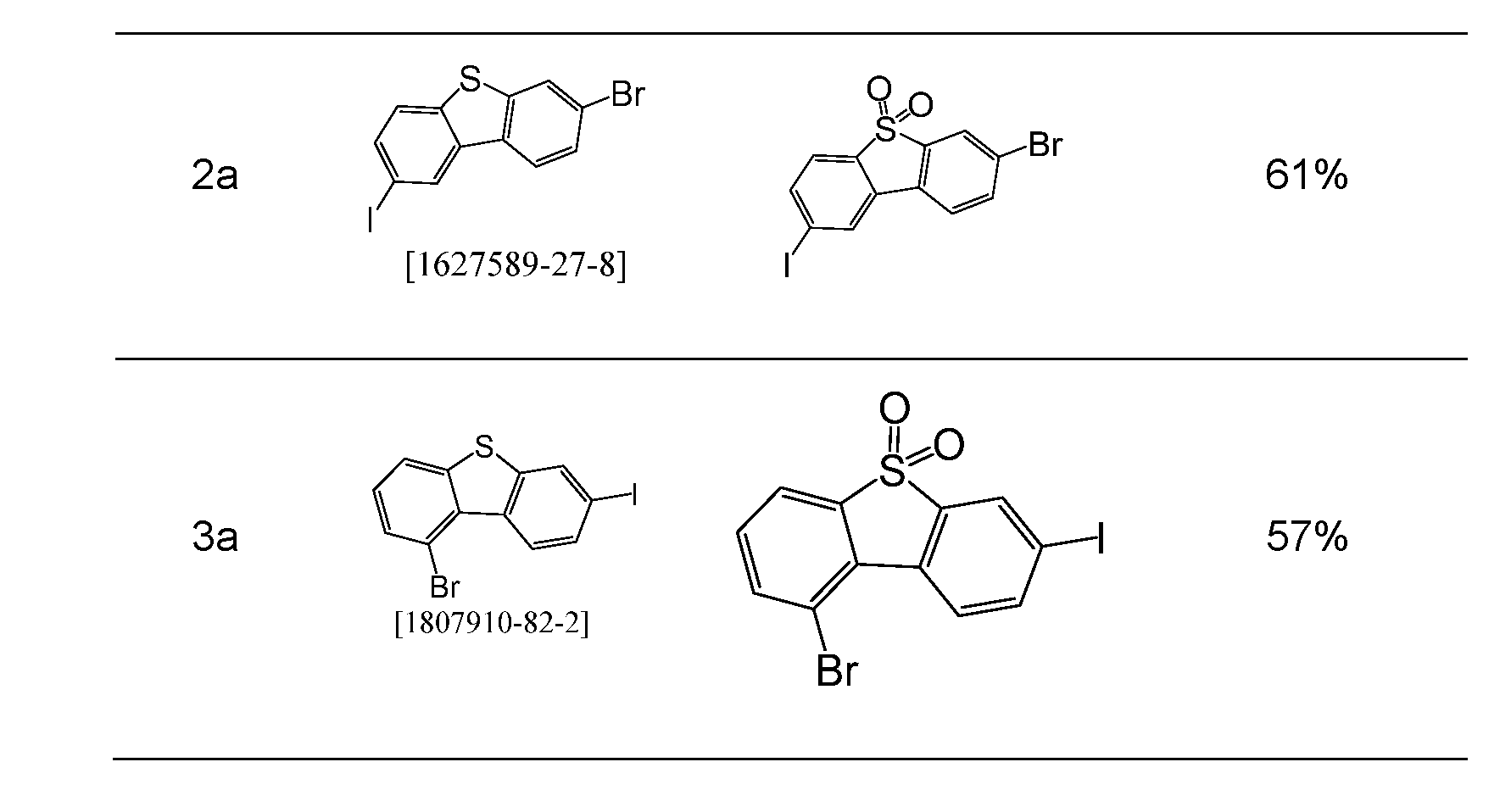
- reaction mixture After 24 hours, the reaction mixture is allowed to cool to room temperature and the volume of the mixture is reduced to one third under reduced pressure, water is added and the precipitated solid is filtered off, washed with water, ethanol and heptane and further purified by filtration through a silica gel-packed filter Column (THF as eluent). The solvent is then removed on the Rotavap.
- THF silica gel-packed filter Column
- OLEDs have the following layer structure: substrate / hole injection layer (HIL) / hole transport layer (HTL) / electron blocking layer (EBL) / emission layer (EML) / optional hole blocking layer (HBL) / electron transport layer (ETL) / optional electron injection layer (EIL) and finally a cathode.
- the cathode is formed by a 100 nm thick aluminum layer.
- Table 1 The precise structure of the OLEDs can be found in Table 1.
- the materials required to produce the OLEDs are shown in Table 2.
- the data of the OLEDs are listed in Table 3.
- the emission layer always consists of at least one matrix material (host material, host material) and an emitting dopant (dopant, emitter), which is added to the matrix material or matrix materials by co-evaporation in a certain proportion by volume.
- a specification such as EG1 :IC2:TER5 (55%:35%:10%) means that the material EG1 accounts for 55% by volume, IC2 for 35% by volume and TER5 for 10% by volume in the layer present.
- the electron transport layer can also consist of a mixture of two materials.
- the OLEDs are characterized by default.
- the electroluminescence spectra, the external quantum efficiency (EQE, measured in %) as a function of the luminance, calculated from current-voltage-luminance characteristics assuming a Lambertian emission characteristic, and the service life are determined.
- the electroluminescence spectra are determined at a luminance of 1000 cd/m 2 and the CIE 1931 x and y color coordinates are calculated therefrom.
- the specification U1000 in Table 3 designates the voltage that is required for a Luminance of 1000 cd/m 2 is required.
- EQE1000 designates the external quantum efficiency that can be achieved at 1000 cd/m 2 .
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Physics & Mathematics (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Oxygen Or Sulfur (AREA)
Abstract
Description
Claims
Priority Applications (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020247030073A KR20240146680A (ko) | 2022-02-09 | 2023-02-06 | 유기 전계 발광 디바이스용 재료 |
| CN202380020550.7A CN118660882A (zh) | 2022-02-09 | 2023-02-06 | 用于有机电致发光器件的材料 |
| US18/836,787 US20250163035A1 (en) | 2022-02-09 | 2023-02-06 | Materials for organic electroluminescent devices |
| EP23702824.6A EP4476215A1 (de) | 2022-02-09 | 2023-02-06 | Materialien für organische elektrolumineszenzvorrichtungen |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP22155956.0 | 2022-02-09 | ||
| EP22155956 | 2022-02-09 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2023152063A1 true WO2023152063A1 (de) | 2023-08-17 |
Family
ID=80447777
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2023/052756 Ceased WO2023152063A1 (de) | 2022-02-09 | 2023-02-06 | Materialien für organische elektrolumineszenzvorrichtungen |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US20250163035A1 (de) |
| EP (1) | EP4476215A1 (de) |
| KR (1) | KR20240146680A (de) |
| CN (1) | CN118660882A (de) |
| WO (1) | WO2023152063A1 (de) |
Citations (96)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4539507A (en) | 1983-03-25 | 1985-09-03 | Eastman Kodak Company | Organic electroluminescent devices having improved power conversion efficiencies |
| US5151629A (en) | 1991-08-01 | 1992-09-29 | Eastman Kodak Company | Blue emitting internal junction organic electroluminescent device (I) |
| WO1993014080A1 (en) * | 1992-01-15 | 1993-07-22 | E.I. Du Pont De Nemours And Company | Bridged heterocyclic fungicides |
| EP0652273A1 (de) | 1993-11-09 | 1995-05-10 | Shinko Electric Industries Co. Ltd. | Organisches Material für elektrolumineszente Vorrichtung und elektrolumineszente Vorrichtung |
| EP0676461A2 (de) | 1994-04-07 | 1995-10-11 | Hoechst Aktiengesellschaft | Spiroverbindungen und ihre Verwendung als Elektrolumineszenzmaterialien |
| WO1998027136A1 (de) | 1996-12-16 | 1998-06-25 | Aventis Research & Technologies Gmbh & Co Kg | ARYLSUBSTITUIERTE POLY(p-ARYLENVINYLENE), VERFAHREN ZUR HERSTELLUNG UND DEREN VERWENDUNG IN ELEKTROLUMINESZENZBAUELEMENTEN |
| WO2000070655A2 (en) | 1999-05-13 | 2000-11-23 | The Trustees Of Princeton University | Very high efficiency organic light emitting devices based on electrophosphorescence |
| WO2001041512A1 (en) | 1999-12-01 | 2001-06-07 | The Trustees Of Princeton University | Complexes of form l2mx as phosphorescent dopants for organic leds |
| WO2002002714A2 (en) | 2000-06-30 | 2002-01-10 | E.I. Du Pont De Nemours And Company | Electroluminescent iridium compounds with fluorinated phenylpyridines, phenylpyrimidines, and phenylquinolines and devices made with such compounds |
| WO2002015645A1 (en) | 2000-08-11 | 2002-02-21 | The Trustees Of Princeton University | Organometallic compounds and emission-shifting organic electrophosphorescence |
| EP1191614A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung und dafür verwendete Metallkoordinationsverbindung |
| EP1191613A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung, Bildanzeigevorrichtung und Metallkoordinationsverbindung |
| EP1191612A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung, Bildanzeigevorrichtung und Metallkoordinationsverbindung |
| EP1205527A1 (de) | 2000-03-27 | 2002-05-15 | Idemitsu Kosan Co., Ltd. | Organische elektrolumineszierende vorrichtung |
| WO2004013080A1 (en) | 2002-08-01 | 2004-02-12 | Covion Organic Semiconductors Gmbh | Spirobifluorene derivatives, their preparation and uses thereof |
| JP2004288381A (ja) | 2003-03-19 | 2004-10-14 | Konica Minolta Holdings Inc | 有機エレクトロルミネッセンス素子 |
| WO2004093207A2 (de) | 2003-04-15 | 2004-10-28 | Covion Organic Semiconductors Gmbh | Mischungen von organischen zur emission befähigten halbleitern und matrixmaterialien, deren verwendung und elektronikbauteile enthaltend diese mischungen |
| WO2005019373A2 (de) | 2003-08-19 | 2005-03-03 | Basf Aktiengesellschaft | Übergangsmetallkomplexe mit carbenliganden als emitter für organische licht-emittierende dioden (oleds) |
| US20050069729A1 (en) | 2003-09-30 | 2005-03-31 | Konica Minolta Holdings, Inc. | Organic electroluminescent element, illuminator, display and compound |
| WO2005033244A1 (de) | 2003-09-29 | 2005-04-14 | Covion Organic Semiconductors Gmbh | Metallkomplexe |
| US20050258742A1 (en) | 2004-05-18 | 2005-11-24 | Yui-Yi Tsai | Carbene containing metal complexes as OLEDs |
| WO2005111172A2 (de) | 2004-05-11 | 2005-11-24 | Merck Patent Gmbh | Neue materialmischungen für die elektrolumineszenz |
| JP2005347160A (ja) | 2004-06-04 | 2005-12-15 | Konica Minolta Holdings Inc | 有機エレクトロルミネッセンス素子、照明装置及び表示装置 |
| EP1617710A1 (de) | 2003-04-23 | 2006-01-18 | Konica Minolta Holdings, Inc. | Material für ein organisches elektrolumineszenzgerät, organisches elektrolumineszenzgerät, beleuchtungsvorrichtung und anzeige |
| WO2006005627A1 (en) | 2004-07-15 | 2006-01-19 | Merck Patent Gmbh | Oligomeric derivatives of spirobifluorene, their preparation and use |
| WO2006117052A1 (de) | 2005-05-03 | 2006-11-09 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtung und in deren herstellung verwendete boronsäure- und borinsäure-derivate |
| EP1731584A1 (de) | 2004-03-31 | 2006-12-13 | Konica Minolta Holdings, Inc. | Organischer elektrolumineszenzvorrichtungsstoff, organische elektrolumineszenzvorrichtung, display und beleuchtungsvorrichtung |
| WO2007063754A1 (ja) | 2005-12-01 | 2007-06-07 | Nippon Steel Chemical Co., Ltd. | 有機電界発光素子用化合物及び有機電界発光素子 |
| WO2007137725A1 (de) | 2006-05-31 | 2007-12-06 | Merck Patent Gmbh | Neue materialien für organische elektrolumineszenzvorrichtungen |
| WO2008056746A1 (en) | 2006-11-09 | 2008-05-15 | Nippon Steel Chemical Co., Ltd. | Compound for organic electroluminescent device and organic electroluminescent device |
| WO2008086851A1 (de) | 2007-01-18 | 2008-07-24 | Merck Patent Gmbh | Carbazol-derivate für organische elektrolumineszenzvorrichtungen |
| WO2009062578A1 (de) | 2007-11-12 | 2009-05-22 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtungen enthaltend azomethin-metall-komplexe |
| WO2009146770A2 (de) | 2008-06-05 | 2009-12-10 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe |
| WO2010006680A1 (de) | 2008-07-18 | 2010-01-21 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010015307A1 (de) | 2008-08-04 | 2010-02-11 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe mit isonitrilliganden |
| WO2010015306A1 (de) | 2008-08-08 | 2010-02-11 | Merck Patent Gmbh, | Organische elektrolumineszenzvorrichtung |
| WO2010031485A1 (de) | 2008-09-22 | 2010-03-25 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054731A1 (de) | 2008-11-13 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054728A1 (de) | 2008-11-13 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054729A2 (de) | 2008-11-11 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054730A1 (de) | 2008-11-11 | 2010-05-20 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtungen |
| WO2010086089A1 (de) | 2009-02-02 | 2010-08-05 | Merck Patent Gmbh | Metallkomplexe |
| WO2010099852A1 (de) | 2009-03-02 | 2010-09-10 | Merck Patent Gmbh | Metallkomplexe mit azaborol-liganden und elektronische vorrichtung damit |
| WO2010102709A1 (de) | 2009-03-13 | 2010-09-16 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010108579A1 (de) | 2009-03-23 | 2010-09-30 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtung |
| WO2010136109A1 (de) | 2009-05-29 | 2010-12-02 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010151006A1 (en) | 2009-06-22 | 2010-12-29 | Dow Advanced Display Materials, Ltd. | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| WO2011000455A1 (de) | 2009-06-30 | 2011-01-06 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011032626A1 (de) | 2009-09-16 | 2011-03-24 | Merck Patent Gmbh | Metallkomplexe |
| WO2011042107A2 (de) | 2009-10-08 | 2011-04-14 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011057706A2 (de) | 2009-11-14 | 2011-05-19 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2011060867A1 (de) | 2009-11-18 | 2011-05-26 | Merck Patent Gmbh | Stickstoffhaltige kondensierte heterozyklen für oleds |
| WO2011060877A2 (de) | 2009-11-17 | 2011-05-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011060859A1 (de) | 2009-11-17 | 2011-05-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011066898A1 (de) | 2009-12-05 | 2011-06-09 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe |
| WO2011088877A1 (de) | 2010-01-25 | 2011-07-28 | Merck Patent Gmbh | Verbindungen für elektronische vorrichtungen |
| DE102010019306A1 (de) * | 2010-05-04 | 2011-11-10 | Merck Patent Gmbh | Organische Elektrolumineszenzvorrichtungen |
| WO2011157339A1 (de) | 2010-06-15 | 2011-12-22 | Merck Patent Gmbh | Metallkomplexe |
| WO2012007086A1 (de) | 2010-07-16 | 2012-01-19 | Merck Patent Gmbh | Metallkomplexe |
| WO2012048781A1 (de) | 2010-10-15 | 2012-04-19 | Merck Patent Gmbh | Materialien auf basis von triphenylen für organische elektrolumineszenzvorrichtungen |
| WO2012143080A2 (de) | 2011-04-18 | 2012-10-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2013041176A1 (de) | 2011-09-21 | 2013-03-28 | Merck Patent Gmbh | Carbazolderivate für organische elektrolumineszenzvorrichtungen |
| WO2013056776A1 (de) | 2011-10-20 | 2013-04-25 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| EP2599771A1 (de) * | 2010-07-30 | 2013-06-05 | Daito Chemix Corporation | Naphthalinderivat |
| WO2014008982A1 (de) | 2012-07-13 | 2014-01-16 | Merck Patent Gmbh | Metallkomplexe |
| WO2014023377A2 (de) | 2012-08-07 | 2014-02-13 | Merck Patent Gmbh | Metallkomplexe |
| WO2014094961A1 (de) | 2012-12-21 | 2014-06-26 | Merck Patent Gmbh | Metallkomplexe |
| WO2014094960A1 (de) | 2012-12-21 | 2014-06-26 | Merck Patent Gmbh | Metallkomplexe |
| WO2014114007A1 (zh) * | 2013-01-25 | 2014-07-31 | 深圳市华星光电技术有限公司 | 含砜基的化合物、采用含砜基的化合物的有机电致发光器件及其制备方法 |
| WO2015036074A1 (de) | 2013-09-11 | 2015-03-19 | Merck Patent Gmbh | Metallkomplexe |
| KR20150031396A (ko) | 2013-09-13 | 2015-03-24 | (주)씨에스엘쏠라 | 신규한 유기발광화합물 및 이를 포함하는 유기전기발광소자 |
| WO2015104045A1 (de) | 2014-01-13 | 2015-07-16 | Merck Patent Gmbh | Metallkomplexe |
| WO2015117718A1 (de) | 2014-02-05 | 2015-08-13 | Merck Patent Gmbh | Metallkomplexe |
| WO2015169412A1 (de) | 2014-05-05 | 2015-11-12 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2016015810A1 (de) | 2014-07-29 | 2016-02-04 | Merck Patent Gmbh | Materialien f?r organische elektrolumineszenzvorrichtungen |
| WO2016015815A1 (de) | 2014-07-28 | 2016-02-04 | Merck Patent Gmbh | Metallkomplexe |
| WO2016023608A1 (de) | 2014-08-13 | 2016-02-18 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2016124304A1 (de) | 2015-02-03 | 2016-08-11 | Merck Patent Gmbh | Metallkomplexe |
| WO2017032439A1 (de) | 2015-08-25 | 2017-03-02 | Merck Patent Gmbh | Metallkomplexe |
| WO2017148564A1 (de) | 2016-03-03 | 2017-09-08 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2018011186A1 (de) | 2016-07-14 | 2018-01-18 | Merck Patent Gmbh | Metallkomplexe |
| WO2018041769A1 (de) | 2016-08-30 | 2018-03-08 | Merck Patent Gmbh | Bl- und trinukleare metallkomplexe aufgebaut aus zwei miteinander verknüpften tripodalen hexadentaten liganden zur verwendung in elektrolumineszenzvorrichtungen |
| CN108359445A (zh) * | 2018-05-05 | 2018-08-03 | 长春海谱润斯科技有限公司 | 一种双极性有机电致发光材料及其有机发光器件 |
| WO2018178001A1 (de) | 2017-03-29 | 2018-10-04 | Merck Patent Gmbh | Metallkomplexe |
| CN105384767B (zh) * | 2015-10-13 | 2019-01-15 | 华南理工大学 | 一类基于s,s-二氧-二苯并噻吩鎓盐的聚合物前躯体单体、聚合物及其制备方法与应用 |
| WO2019016430A1 (en) | 2017-07-21 | 2019-01-24 | Fomatec Oy | TRACK ASSEMBLIES FOR FIELD VEHICLES AND METHOD FOR REPAIRING TRACKS |
| WO2019020538A1 (de) | 2017-07-25 | 2019-01-31 | Merck Patent Gmbh | Metallkomplexe |
| US20190093009A1 (en) * | 2017-09-26 | 2019-03-28 | Luminescence Technology Corporation | Delayed fluorescence compound and organic electroluminescent device using the same |
| WO2019115423A1 (de) | 2017-12-13 | 2019-06-20 | Merck Patent Gmbh | Metallkomplexe |
| WO2019158453A1 (de) | 2018-02-13 | 2019-08-22 | Merck Patent Gmbh | Metallkomplexe |
| WO2019194594A1 (ko) * | 2018-04-05 | 2019-10-10 | 주식회사 엘지화학 | 화합물 및 이를 포함하는 유기 전자 소자 |
| CN110790756A (zh) | 2018-08-03 | 2020-02-14 | 北京绿人科技有限责任公司 | 有机化合物及其应用和一种有机电致发光器件 |
| CN108299514B (zh) * | 2018-04-02 | 2020-04-14 | 江西师范大学 | 含二苯并噻吩砜基的铱配合物发光材料及其应用 |
| CN111217832A (zh) * | 2018-11-23 | 2020-06-02 | 北京夏禾科技有限公司 | 一种有机化合物、包含其的电致发光器件及其用途 |
| WO2021045460A1 (ko) * | 2019-09-06 | 2021-03-11 | 엘티소재주식회사 | 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 |
| KR20210091558A (ko) * | 2020-01-14 | 2021-07-22 | 주식회사 엘지화학 | 신규한 화합물 및 이를 포함하는 유기발광 소자 |
-
2023
- 2023-02-06 US US18/836,787 patent/US20250163035A1/en active Pending
- 2023-02-06 KR KR1020247030073A patent/KR20240146680A/ko active Pending
- 2023-02-06 WO PCT/EP2023/052756 patent/WO2023152063A1/de not_active Ceased
- 2023-02-06 CN CN202380020550.7A patent/CN118660882A/zh active Pending
- 2023-02-06 EP EP23702824.6A patent/EP4476215A1/de active Pending
Patent Citations (99)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4539507A (en) | 1983-03-25 | 1985-09-03 | Eastman Kodak Company | Organic electroluminescent devices having improved power conversion efficiencies |
| US5151629A (en) | 1991-08-01 | 1992-09-29 | Eastman Kodak Company | Blue emitting internal junction organic electroluminescent device (I) |
| WO1993014080A1 (en) * | 1992-01-15 | 1993-07-22 | E.I. Du Pont De Nemours And Company | Bridged heterocyclic fungicides |
| EP0652273A1 (de) | 1993-11-09 | 1995-05-10 | Shinko Electric Industries Co. Ltd. | Organisches Material für elektrolumineszente Vorrichtung und elektrolumineszente Vorrichtung |
| EP0676461A2 (de) | 1994-04-07 | 1995-10-11 | Hoechst Aktiengesellschaft | Spiroverbindungen und ihre Verwendung als Elektrolumineszenzmaterialien |
| WO1998027136A1 (de) | 1996-12-16 | 1998-06-25 | Aventis Research & Technologies Gmbh & Co Kg | ARYLSUBSTITUIERTE POLY(p-ARYLENVINYLENE), VERFAHREN ZUR HERSTELLUNG UND DEREN VERWENDUNG IN ELEKTROLUMINESZENZBAUELEMENTEN |
| WO2000070655A2 (en) | 1999-05-13 | 2000-11-23 | The Trustees Of Princeton University | Very high efficiency organic light emitting devices based on electrophosphorescence |
| WO2001041512A1 (en) | 1999-12-01 | 2001-06-07 | The Trustees Of Princeton University | Complexes of form l2mx as phosphorescent dopants for organic leds |
| EP1205527A1 (de) | 2000-03-27 | 2002-05-15 | Idemitsu Kosan Co., Ltd. | Organische elektrolumineszierende vorrichtung |
| WO2002002714A2 (en) | 2000-06-30 | 2002-01-10 | E.I. Du Pont De Nemours And Company | Electroluminescent iridium compounds with fluorinated phenylpyridines, phenylpyrimidines, and phenylquinolines and devices made with such compounds |
| WO2002015645A1 (en) | 2000-08-11 | 2002-02-21 | The Trustees Of Princeton University | Organometallic compounds and emission-shifting organic electrophosphorescence |
| EP1191614A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung und dafür verwendete Metallkoordinationsverbindung |
| EP1191613A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung, Bildanzeigevorrichtung und Metallkoordinationsverbindung |
| EP1191612A2 (de) | 2000-09-26 | 2002-03-27 | Canon Kabushiki Kaisha | Lumineszente Vorrichtung, Bildanzeigevorrichtung und Metallkoordinationsverbindung |
| WO2004013080A1 (en) | 2002-08-01 | 2004-02-12 | Covion Organic Semiconductors Gmbh | Spirobifluorene derivatives, their preparation and uses thereof |
| JP2004288381A (ja) | 2003-03-19 | 2004-10-14 | Konica Minolta Holdings Inc | 有機エレクトロルミネッセンス素子 |
| WO2004093207A2 (de) | 2003-04-15 | 2004-10-28 | Covion Organic Semiconductors Gmbh | Mischungen von organischen zur emission befähigten halbleitern und matrixmaterialien, deren verwendung und elektronikbauteile enthaltend diese mischungen |
| EP1617710A1 (de) | 2003-04-23 | 2006-01-18 | Konica Minolta Holdings, Inc. | Material für ein organisches elektrolumineszenzgerät, organisches elektrolumineszenzgerät, beleuchtungsvorrichtung und anzeige |
| EP1617711A1 (de) | 2003-04-23 | 2006-01-18 | Konica Minolta Holdings, Inc. | Organisches elektrolumineszenzbauelement und anzeige |
| WO2005019373A2 (de) | 2003-08-19 | 2005-03-03 | Basf Aktiengesellschaft | Übergangsmetallkomplexe mit carbenliganden als emitter für organische licht-emittierende dioden (oleds) |
| WO2005033244A1 (de) | 2003-09-29 | 2005-04-14 | Covion Organic Semiconductors Gmbh | Metallkomplexe |
| WO2005039246A1 (ja) | 2003-09-30 | 2005-04-28 | Konica Minolta Holdings, Inc. | 有機エレクトロルミネッセンス素子、照明装置、表示装置 |
| US20050069729A1 (en) | 2003-09-30 | 2005-03-31 | Konica Minolta Holdings, Inc. | Organic electroluminescent element, illuminator, display and compound |
| EP1731584A1 (de) | 2004-03-31 | 2006-12-13 | Konica Minolta Holdings, Inc. | Organischer elektrolumineszenzvorrichtungsstoff, organische elektrolumineszenzvorrichtung, display und beleuchtungsvorrichtung |
| WO2005111172A2 (de) | 2004-05-11 | 2005-11-24 | Merck Patent Gmbh | Neue materialmischungen für die elektrolumineszenz |
| US20050258742A1 (en) | 2004-05-18 | 2005-11-24 | Yui-Yi Tsai | Carbene containing metal complexes as OLEDs |
| JP2005347160A (ja) | 2004-06-04 | 2005-12-15 | Konica Minolta Holdings Inc | 有機エレクトロルミネッセンス素子、照明装置及び表示装置 |
| WO2006005627A1 (en) | 2004-07-15 | 2006-01-19 | Merck Patent Gmbh | Oligomeric derivatives of spirobifluorene, their preparation and use |
| WO2006117052A1 (de) | 2005-05-03 | 2006-11-09 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtung und in deren herstellung verwendete boronsäure- und borinsäure-derivate |
| WO2007063754A1 (ja) | 2005-12-01 | 2007-06-07 | Nippon Steel Chemical Co., Ltd. | 有機電界発光素子用化合物及び有機電界発光素子 |
| WO2007137725A1 (de) | 2006-05-31 | 2007-12-06 | Merck Patent Gmbh | Neue materialien für organische elektrolumineszenzvorrichtungen |
| WO2008056746A1 (en) | 2006-11-09 | 2008-05-15 | Nippon Steel Chemical Co., Ltd. | Compound for organic electroluminescent device and organic electroluminescent device |
| WO2008086851A1 (de) | 2007-01-18 | 2008-07-24 | Merck Patent Gmbh | Carbazol-derivate für organische elektrolumineszenzvorrichtungen |
| WO2009062578A1 (de) | 2007-11-12 | 2009-05-22 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtungen enthaltend azomethin-metall-komplexe |
| WO2009146770A2 (de) | 2008-06-05 | 2009-12-10 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe |
| WO2010006680A1 (de) | 2008-07-18 | 2010-01-21 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010015307A1 (de) | 2008-08-04 | 2010-02-11 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe mit isonitrilliganden |
| WO2010015306A1 (de) | 2008-08-08 | 2010-02-11 | Merck Patent Gmbh, | Organische elektrolumineszenzvorrichtung |
| WO2010031485A1 (de) | 2008-09-22 | 2010-03-25 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054730A1 (de) | 2008-11-11 | 2010-05-20 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtungen |
| WO2010054729A2 (de) | 2008-11-11 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054728A1 (de) | 2008-11-13 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010054731A1 (de) | 2008-11-13 | 2010-05-20 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010086089A1 (de) | 2009-02-02 | 2010-08-05 | Merck Patent Gmbh | Metallkomplexe |
| WO2010099852A1 (de) | 2009-03-02 | 2010-09-10 | Merck Patent Gmbh | Metallkomplexe mit azaborol-liganden und elektronische vorrichtung damit |
| WO2010102709A1 (de) | 2009-03-13 | 2010-09-16 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010108579A1 (de) | 2009-03-23 | 2010-09-30 | Merck Patent Gmbh | Organische elektrolumineszenzvorrichtung |
| WO2010136109A1 (de) | 2009-05-29 | 2010-12-02 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2010151006A1 (en) | 2009-06-22 | 2010-12-29 | Dow Advanced Display Materials, Ltd. | Novel organic electroluminescent compounds and organic electroluminescent device using the same |
| WO2011000455A1 (de) | 2009-06-30 | 2011-01-06 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011032626A1 (de) | 2009-09-16 | 2011-03-24 | Merck Patent Gmbh | Metallkomplexe |
| WO2011042107A2 (de) | 2009-10-08 | 2011-04-14 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011057706A2 (de) | 2009-11-14 | 2011-05-19 | Merck Patent Gmbh | Materialien für elektronische vorrichtungen |
| WO2011060859A1 (de) | 2009-11-17 | 2011-05-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011060877A2 (de) | 2009-11-17 | 2011-05-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2011060867A1 (de) | 2009-11-18 | 2011-05-26 | Merck Patent Gmbh | Stickstoffhaltige kondensierte heterozyklen für oleds |
| WO2011066898A1 (de) | 2009-12-05 | 2011-06-09 | Merck Patent Gmbh | Elektronische vorrichtung enthaltend metallkomplexe |
| WO2011088877A1 (de) | 2010-01-25 | 2011-07-28 | Merck Patent Gmbh | Verbindungen für elektronische vorrichtungen |
| DE102010019306A1 (de) * | 2010-05-04 | 2011-11-10 | Merck Patent Gmbh | Organische Elektrolumineszenzvorrichtungen |
| WO2011157339A1 (de) | 2010-06-15 | 2011-12-22 | Merck Patent Gmbh | Metallkomplexe |
| WO2012007086A1 (de) | 2010-07-16 | 2012-01-19 | Merck Patent Gmbh | Metallkomplexe |
| EP2599771A1 (de) * | 2010-07-30 | 2013-06-05 | Daito Chemix Corporation | Naphthalinderivat |
| WO2012048781A1 (de) | 2010-10-15 | 2012-04-19 | Merck Patent Gmbh | Materialien auf basis von triphenylen für organische elektrolumineszenzvorrichtungen |
| WO2012143080A2 (de) | 2011-04-18 | 2012-10-26 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2013041176A1 (de) | 2011-09-21 | 2013-03-28 | Merck Patent Gmbh | Carbazolderivate für organische elektrolumineszenzvorrichtungen |
| WO2013056776A1 (de) | 2011-10-20 | 2013-04-25 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2014008982A1 (de) | 2012-07-13 | 2014-01-16 | Merck Patent Gmbh | Metallkomplexe |
| WO2014023377A2 (de) | 2012-08-07 | 2014-02-13 | Merck Patent Gmbh | Metallkomplexe |
| WO2014094961A1 (de) | 2012-12-21 | 2014-06-26 | Merck Patent Gmbh | Metallkomplexe |
| WO2014094960A1 (de) | 2012-12-21 | 2014-06-26 | Merck Patent Gmbh | Metallkomplexe |
| WO2014114007A1 (zh) * | 2013-01-25 | 2014-07-31 | 深圳市华星光电技术有限公司 | 含砜基的化合物、采用含砜基的化合物的有机电致发光器件及其制备方法 |
| WO2015036074A1 (de) | 2013-09-11 | 2015-03-19 | Merck Patent Gmbh | Metallkomplexe |
| KR20150031396A (ko) | 2013-09-13 | 2015-03-24 | (주)씨에스엘쏠라 | 신규한 유기발광화합물 및 이를 포함하는 유기전기발광소자 |
| WO2015104045A1 (de) | 2014-01-13 | 2015-07-16 | Merck Patent Gmbh | Metallkomplexe |
| WO2015117718A1 (de) | 2014-02-05 | 2015-08-13 | Merck Patent Gmbh | Metallkomplexe |
| WO2015169412A1 (de) | 2014-05-05 | 2015-11-12 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2016015815A1 (de) | 2014-07-28 | 2016-02-04 | Merck Patent Gmbh | Metallkomplexe |
| WO2016015810A1 (de) | 2014-07-29 | 2016-02-04 | Merck Patent Gmbh | Materialien f?r organische elektrolumineszenzvorrichtungen |
| WO2016023608A1 (de) | 2014-08-13 | 2016-02-18 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2016124304A1 (de) | 2015-02-03 | 2016-08-11 | Merck Patent Gmbh | Metallkomplexe |
| WO2017032439A1 (de) | 2015-08-25 | 2017-03-02 | Merck Patent Gmbh | Metallkomplexe |
| CN105384767B (zh) * | 2015-10-13 | 2019-01-15 | 华南理工大学 | 一类基于s,s-二氧-二苯并噻吩鎓盐的聚合物前躯体单体、聚合物及其制备方法与应用 |
| WO2017148564A1 (de) | 2016-03-03 | 2017-09-08 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2017148565A1 (de) | 2016-03-03 | 2017-09-08 | Merck Patent Gmbh | Materialien für organische elektrolumineszenzvorrichtungen |
| WO2018011186A1 (de) | 2016-07-14 | 2018-01-18 | Merck Patent Gmbh | Metallkomplexe |
| WO2018041769A1 (de) | 2016-08-30 | 2018-03-08 | Merck Patent Gmbh | Bl- und trinukleare metallkomplexe aufgebaut aus zwei miteinander verknüpften tripodalen hexadentaten liganden zur verwendung in elektrolumineszenzvorrichtungen |
| WO2018178001A1 (de) | 2017-03-29 | 2018-10-04 | Merck Patent Gmbh | Metallkomplexe |
| WO2019016430A1 (en) | 2017-07-21 | 2019-01-24 | Fomatec Oy | TRACK ASSEMBLIES FOR FIELD VEHICLES AND METHOD FOR REPAIRING TRACKS |
| WO2019020538A1 (de) | 2017-07-25 | 2019-01-31 | Merck Patent Gmbh | Metallkomplexe |
| US20190093009A1 (en) * | 2017-09-26 | 2019-03-28 | Luminescence Technology Corporation | Delayed fluorescence compound and organic electroluminescent device using the same |
| WO2019115423A1 (de) | 2017-12-13 | 2019-06-20 | Merck Patent Gmbh | Metallkomplexe |
| WO2019158453A1 (de) | 2018-02-13 | 2019-08-22 | Merck Patent Gmbh | Metallkomplexe |
| CN108299514B (zh) * | 2018-04-02 | 2020-04-14 | 江西师范大学 | 含二苯并噻吩砜基的铱配合物发光材料及其应用 |
| WO2019194594A1 (ko) * | 2018-04-05 | 2019-10-10 | 주식회사 엘지화학 | 화합물 및 이를 포함하는 유기 전자 소자 |
| CN108359445A (zh) * | 2018-05-05 | 2018-08-03 | 长春海谱润斯科技有限公司 | 一种双极性有机电致发光材料及其有机发光器件 |
| CN110790756A (zh) | 2018-08-03 | 2020-02-14 | 北京绿人科技有限责任公司 | 有机化合物及其应用和一种有机电致发光器件 |
| CN111217832A (zh) * | 2018-11-23 | 2020-06-02 | 北京夏禾科技有限公司 | 一种有机化合物、包含其的电致发光器件及其用途 |
| WO2021045460A1 (ko) * | 2019-09-06 | 2021-03-11 | 엘티소재주식회사 | 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 |
| KR20210091558A (ko) * | 2020-01-14 | 2021-07-22 | 주식회사 엘지화학 | 신규한 화합물 및 이를 포함하는 유기발광 소자 |
Non-Patent Citations (3)
| Title |
|---|
| CHEN GUITING ET AL: "Synthesis and optical and electrochemical properties of water-soluble cationic fluorophores based on bispyridinium and dibenzothiophene-S,S-dioxide", NEW JOURNAL OF CHEMISTRY, vol. 41, no. 4, 1 January 2017 (2017-01-01), GB, pages 1696 - 1703, XP093037404, ISSN: 1144-0546, DOI: 10.1039/C6NJ03108K * |
| LUO MING ET AL: "Novel yellow phosphorescent iridium complexes with cycolmetalated (pyridin-2-yl)dibenzothiophene-S,S-dioxide ligands for singly doped emissive layer hybrid white organic light-emitting diodes", OPTICAL MATERIALS, vol. 91, 2019, pages 439 - 446, XP085697249, ISSN: 0925-3467, DOI: 10.1016/J.OPTMAT.2019.04.006 * |
| SOLOMATINA ANASTASIA I. ET AL: "Dibenzothiophene-platinated complexes: probing the effect of ancillary ligands on the photophysical performance", DALTON TRANSACTIONS, vol. 46, no. 12, 1 January 2017 (2017-01-01), Cambridge, pages 3895 - 3905, XP093037307, ISSN: 1477-9226, DOI: 10.1039/C7DT00349H * |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20240146680A (ko) | 2024-10-08 |
| US20250163035A1 (en) | 2025-05-22 |
| CN118660882A (zh) | 2024-09-17 |
| EP4476215A1 (de) | 2024-12-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP3744155B1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| EP3371274B1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| EP3423543A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| DE102009053645A1 (de) | Materialien für organische Elektrolumineszenzvorrichtung | |
| EP3335254A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| EP3160954A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| WO2018166932A1 (de) | Verbindungen mit arylamin-strukturen | |
| EP3681890A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| EP3197982A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| EP4229064A1 (de) | Heterocyclische verbindungen für organische elektrolumineszenzvorrichtungen | |
| WO2018087022A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| EP4519383A1 (de) | Cyclische verbindungen für organische elektrolumineszenzvorrichtungen | |
| WO2022157343A1 (de) | Stickstoffhaltige verbindungen für organische elektrolumineszenzvorrichtungen | |
| WO2022194799A1 (de) | Heteroaromatische verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP3877373B1 (de) | 5,6-diphenyl-5,6-dihydro-dibenz[c,e][1,2]azaphosphorin- und 6-phenyl-6h-dibenzo[c,e][1,2]thiazin-5,5-dioxid-derivate und ähnliche verbindungen als organische elektrolumineszenzmaterialien für oleds | |
| WO2022200638A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| EP4244228A1 (de) | Schwefelhaltige verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP4192832A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| WO2023152063A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| WO2023110742A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| EP4301757A1 (de) | Verbindungen für organische elektrolumineszenzvorrichtungen | |
| EP4638439A1 (de) | Neue materialien für organische elektrolumineszenzvorrichtungen | |
| EP4274827A1 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| EP4259628A2 (de) | Materialien für organische elektrolumineszenzvorrichtungen | |
| EP4255905A1 (de) | Heterocyclische verbindungen für organische elektrolumineszenzvorrichtungen |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 23702824 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202380020550.7 Country of ref document: CN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 18836787 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: 20247030073 Country of ref document: KR Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020247030073 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023702824 Country of ref document: EP |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2023702824 Country of ref document: EP Effective date: 20240909 |
|
| WWP | Wipo information: published in national office |
Ref document number: 18836787 Country of ref document: US |