WO2018039508A1 - Method for reducing unwanted effects in parasiticidal treatments - Google Patents
Method for reducing unwanted effects in parasiticidal treatments Download PDFInfo
- Publication number
- WO2018039508A1 WO2018039508A1 PCT/US2017/048527 US2017048527W WO2018039508A1 WO 2018039508 A1 WO2018039508 A1 WO 2018039508A1 US 2017048527 W US2017048527 W US 2017048527W WO 2018039508 A1 WO2018039508 A1 WO 2018039508A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- alkyl
- ring
- optionally substituted
- parasiticidal
- alkoxy
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 Cc1cc(C(C2)=NO[C@]2(C(F)(F)F)c2cc(*)c(*)c(*)c2)ccc1C(NCC(NCC(F)(F)F)=O)=O Chemical compound Cc1cc(C(C2)=NO[C@]2(C(F)(F)F)c2cc(*)c(*)c(*)c2)ccc1C(NCC(NCC(F)(F)F)=O)=O 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N25/00—Biocides, pest repellants or attractants, or plant growth regulators, characterised by their forms, or by their non-active ingredients or by their methods of application, e.g. seed treatment or sequential application; Substances for reducing the noxious effect of the active ingredients to organisms other than pests
- A01N25/32—Ingredients for reducing the noxious effect of the active substances to organisms other than pests, e.g. toxicity reducing compositions, self-destructing compositions
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
- A61K31/42—Oxazoles
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/10—Alcohols; Phenols; Salts thereof, e.g. glycerol; Polyethylene glycols [PEG]; Poloxamers; PEG/POE alkyl ethers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K9/00—Medicinal preparations characterised by special physical form
- A61K9/0012—Galenical forms characterised by the site of application
- A61K9/0019—Injectable compositions; Intramuscular, intravenous, arterial, subcutaneous administration; Compositions to be administered through the skin in an invasive manner
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P33/00—Antiparasitic agents
- A61P33/14—Ectoparasiticides, e.g. scabicides
Definitions
- the present invention provides for a method for reducing the withdrawal period when treating food-producing animals, including livestock, for parasitic infestations or infections (including ectoparasites (e.g., fleas or ticks) and/or endoparasites) with a parasiticidal composition comprising an isoxazoline compound enriched in the ( ⁇ -enantiomer, or a pharmaceutically acceptable salt thereof, and an inert carrier or formulation diluent.
- the present invention also provides for methods for reducing at least one unwanted effect while treating an animal to control or prevent a parasitic infestation with a parasiticidal composition comprising an isoxazoline compound or a pharmaceutically acceptable salt thereof and an inert carrier or formulation diluent.
- ectoparasites such as fleas, ticks and parasitic flies
- endoparasites such as nematodes and other worms.
- domesticated animals such as cats and dogs, are often infested with one or more of the following ectoparasites:
- - fleas e.g. Ctenocephalides spp., such as Ctenocephalides felis and the like;
- ticks e.g. Rhipicephalus spp., Ixodes spp., Dermacentor spp., Amblyomma spp., and the like
- Rhipicephalus spp. Ixodes spp.
- Dermacentor spp. Dermacentor spp.
- Amblyomma spp. and the like
- - lice e.g. Trichodectes spp., Cheyletiella spp., Linognathus spp. and the like
- Trichodectes spp. Cheyletiella spp., Linognathus spp. and the like
- Fleas are a particular problem because not only do they adversely affect the health of the animal or human, but they also cause a great deal of psychological stress. Moreover, fleas may also transmit pathogenic agents to animals and humans, such as tapeworm ⁇ Dipylidium caninum).
- ticks are also harmful to the physical and psychological health of the animal or human.
- Major diseases which may be transmitted by ticks include borreliosis (Lyme disease caused by Borrelia burgdorferi), babesiosis (or piroplasmosis caused by Babesia spp.) and rickettsioses (e.g. Rocky Mountain spotted fever).
- Ticks also release toxins which cause inflammation or paralysis in the host. Occasionally, these toxins are fatal to the host.
- Mosquitoes act as vectors for a number of viral and protozoal infections in the world. It has been reported that mosquitoes carry diseases that potentially compromise the health of one- eighth of the world's population and impact the health and economy of hundreds of millions of people per year. In addition to clinically significant diseases like malaria and yellow fever, mosquitoes are also implicated in the transmission of many arboviruses, infections with which cause symptoms ranging from nonspecific flu-like illnesses to encephalitis that may result in death. In addition to humans, domesticated animals such as dogs, cats, horses, cattle, sheep, pigs, as well as wild animals and birds may become infected and experience clinical disease as a result of that infection.
- arboviruses that infect humans and other mammals are members of one of three virus families, Togaviridae, Flavividae, and Bunyaviridae, and include the agent responsible for Eastern Equine Encephalitis (EEE), Western Equine Encephalitis (WEE), St. Louis Encephalitis (SLE), La Crosse Encephalitis (LCE), and infection caused by Dengue Virus, and West Nile Virus (WNV).
- EEE Eastern Equine Encephalitis
- WEE Western Equine Encephalitis
- SLE St. Louis Encephalitis
- LCE La Crosse Encephalitis
- WNV West Nile Virus
- arboviruses of medical importance include viruses responsible for Japanese Encephalitis (JE) and Venezuelan Equine Encephalitis.
- farm animals are also susceptible to parasite infestations.
- cattle are affected by a large number of parasites.
- a parasite which is prevalent among cattle in some regions is the tick of the genus Rhipicephalus, especially those of the species microplus (cattle tick), decoloratus and annulatus.
- Ticks such as Rhipicephalus microplus ( ormerly Boophilus microplus) are difficult to control because they lay eggs in the pasture where farm animals graze.
- This species of ticks is considered a one-host tick and spends immature and adult stages on one animal before the female engorges and falls off the host to lay eggs in the environment. The life cycle of the tick is approximately three to four weeks.
- Rhipicephalus microplus may infest buffalo, horses, donkeys, goats, sheep, deer, pigs, and dogs.
- a heavy tick burden on animals can decrease production and damage hides as well as transmit diseases such as babesiosis ("cattle fever") and anaplasmosis.
- a factor when treating food producing animals, such as cattle or sheep is the time period between the last drug treatment and when the animals are slaughtered to enter the human food chain ("withdrawal period").
- the FDA establishes withdrawal periods as part of the approval process to specify the required time needed after the last administration of a bioactive agent or agents to assure that drug residues in the marketable edible tissues, milk or eggs of food producing animals are below a predetermined and regulated residue tolerance level. Hence, reducing the withdrawal period has advantageous economic advantages for the rancher and farmer.
- helminthiasis which are caused by of parasitic worms categorized as cestodes (tapeworm), nematodes (roundworm) and trematodes (flatworm or flukes). These parasites adversely affect the nutrition of the animal and cause severe economic losses in pigs, sheep, horses, and cattle as well as affecting companion animals and poultry.
- parasites which occur in the gastrointestinal tract of animals and humans include those from the genus Ancylostoma, Necator, Ascaris, Strongyloides, Trichinella, Capillaria, Toxocara, Toxascaris, Trichuris, Enterobius and parasites which are found in the blood or other tissues and organs such as filarial worms and the extra intestinal stages of Strongyloides, Toxocara and Trichinella.
- WO 2013/039948 Al provides for topical veterinary compositions comprising at least one isoxazoline active agent and WO 2013/119442 Al provides for oral veterinary compositions such as a soft chew, which comprise at least one isoxazoline active agent. All the patent documents referred to above are incorporated herein by reference in their entirety.
- active agents in addition to topical and oral dosage forms, it is sometimes possible to formulate active agents as long acting injectable formulations or as extended release injectable formulations, depending upon, for example, the physiochemical properties of the individual active agent; these properties include, for example, solubility, bioavailability, etc.
- long acting injectable formulations comprising a bioactive agent are described in US 6,733,767 and US 8,362,086, which provide for long acting injectable formulations comprising, for example an avermectin or milbemycin and a biological acceptable polymer.
- Application PCT/US2016/19688, published as WO 2016/138339 provides for long acting injectable formulations wherein the bioactive agent is an isoxazoline compound.
- extended release injectable formulations are described, for example in Application PCT/US2016/026253, which is published as WO 2016/164487. Both these applications are incorporated by reference.
- Isoxazoline compounds achieve their parasiticidal effect by inhibiting the GABA-gated chloride-channels of insects, thereby causing hyper-excitation, which leads to death. Isoxazoline compounds are also known potentially to interact with GABA-gated chloride-channels and other off-target receptors in mammals, which may, in turn, lead to unwanted health effects. These off- target sites are typically screened for in rodent and human in vitro pharmacological assays.
- Afoxolaner for example, was shown to interact with calcium channel L-type receptors, Na channel site 2 receptors, norepinephrine cellular transport receptors, dopamine cellular transport receptors, CB-1 receptors, adenosine a3 receptors, leukotriene, cysteinyl Cys T receptors, Histamine 2 receptors, sodium channel site 2 receptors. Histamine H2 receptor binding has been associated with neurological disorders such as seizures, convulsions, restlessness, muscle tremors, insomnia, nervousness, delirium, depression, mydriasis, hyperesthesia. CB-1 receptor binding affects food intake. Gomez, et al, J. Neurosci., 22(21), 9612-9617 (2002).
- the safety of residues of veterinary drugs in human food is most commonly addressed through the conduct of toxicology studies in laboratory animal species that provide for the determination of a no-observed-adverse-effect level (NOAEL) and an acceptable daily intake (ADI) by application of appropriate safety/uncertainty factors.
- NOAEL no-observed-adverse-effect level
- ADI acceptable daily intake
- the ADI generally expressed as microgram ⁇ g) or milligram (mg)/kg body weight per day, is defined as the daily intake of drug residue which, for up to an entire lifetime, appears to be without adverse effects or harm to the health of the consumer.
- the ADI is then used to determine safe concentrations of drug residue in edible tissues and tissue tolerance levels. When combined with tissue residue depletion data, an appropriate product withdrawal period can be determined.
- compositions comprising isoxazoline bioactive agents alone or in combination with other bioactive agents there is a need to reduce the withdrawal period when treating food producing animals for parasitic infestations by administering parasiticidal compositions comprising at least one isoxazoline bioactive agent to said animals.
- parasiticidal compositions comprising at least one isoxazoline bioactive agent to reduce the unwanted effects that can be observed when treating animals for a parasitic infestation by administering parasiticidal compositions comprising at least one isoxazoline bioactive agent to said animals.
- parasiticidal compositions comprising at least one isoxazoline bioactive agent there is also a need to reduce unwanted effects when treating humans to control or protect them from parasites that act as vectors for viral, bacterial and protozoal infections (e.g. mosquitoes and ticks, among others).
- the present invention provides for a method for the treatment or prevention of a parasitic infestation in an animal with a parasiticidal pesticidal isoxazoline compounds that results in an improved toxicological/safety profile for the animal treated, and/or a lower toxicological residue, and/or when the animal treated is a livestock or milk-producing animal including cattle, a shorter withdrawal period, relative to the effect observed with administration of the racemic compound or the (R)-enantiomer of the compound.
- the present invention provides for a method for reducing at least one unwanted effect while treating an animal for parasitic infestations, which comprises administering an effective amount of an ( ⁇ -enantiomer of an isoxazoline compound, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S)- enantiomer of the isoxazoline compound or salt and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)-enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
- the unwanted effect is an unfavorable injection site reaction related to the injection of a composition comprising a parasiticidal isoxazoline compound to an animal.
- the present invention provides for, inter alia, a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an (S)-enantiomer of an isoxazoline compound, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the ( ⁇ -enantiomer of the isoxazoline compound or salt thereof and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)-enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
- the present invention provides a method for reducing at least one unwanted effect while treating an animal for parasitic infestations with an isoxazoline compound, or a pharmaceutically acceptable salt thereof, by reducing the binding of the isoxazoline compound to an off-target receptor, which comprises administering to said animal an effective amount of an (S) enantiomer of the isoxazoline compound, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising an effective amount of the ( ⁇ -enantiomer of the isoxazoline compound, or a pharmaceutically acceptable salt thereof, and an inert carrier or formulation diluent wherein the composition contains less than about 10% by weight of the (R) enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
- the present invention provides for a method for reducing a withdrawal period when treating food-producing animals, including livestock, for parasitic infestations, which comprises administering an effective amount of the ( ⁇ -enantiomer of an isoxazoline compound, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S) enantiomer of the isoxazoline compound, or a pharmaceutically acceptable salt thereof, and an inert carrier or formulation diluent, wherein the composition contains less than about 10% by weight of the (R) enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
- the present invention further provides for a method for treating or preventing an ectoparasitic infestation in an animal in need thereof while reducing at least one unwanted effect, which comprises administering an effective amount of an ( ⁇ -enantiomer of an isoxazoline compound, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S) enantiomer of an isoxazoline compound, or a pharmaceutically acceptable salt thereof, and an inert carrier or formulation diluent, wherein the composition contains less than about 10% by weight of the (R) enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof; and wherein said parasiticidal is effective against about provides methods for treating or preventing ectoparasitic infestations with an efficacy of about at least 90% against said ectoparasites for at least 2 months, at least 3 months, at least 4 moths, at least 5 months, or 6 months.
- administering a parasiticidal composition comprising an ( ⁇ -enantiomer of a parasiticidal isoxazoline compound in which the unwanted effect resides in the (R)-enantiomer and an inert carrier or formulation diluent wherein the composition contains less than about 10% by weight of the corresponding (R)-enantiomer of the isoxazoline compound, or a pharmaceutically acceptable salt thereof, will increase the safety profile of the parasiticidal compositions by reducing at least one unwanted effect by, for example, reducing off-target receptor binding, thereby, increasing the NOAEL and ADI levels and allowing one to reduce the withdrawal period.
- the methods of the invention allow for the reduction of unwanted effects at the injection site when the composition is administered by injection.
- Figure 1 depicts the mean body weight on female rats for 14 days following the administration of Formulation A (100 mg of the (R) enantiomer compound A dissolved in the carrier), Formulation B (200 mg of the racemic mixture of compound A dissolved the carrier); Formulation C (500 mg of the (S) enantiomer of the compound A dissolved in the carrier); and Control (carrier).
- Formulation A 100 mg of the (R) enantiomer compound A dissolved in the carrier
- Formulation B 200 mg of the racemic mixture of compound A dissolved the carrier
- Formulation C 500 mg of the (S) enantiomer of the compound A dissolved in the carrier
- Control carrier
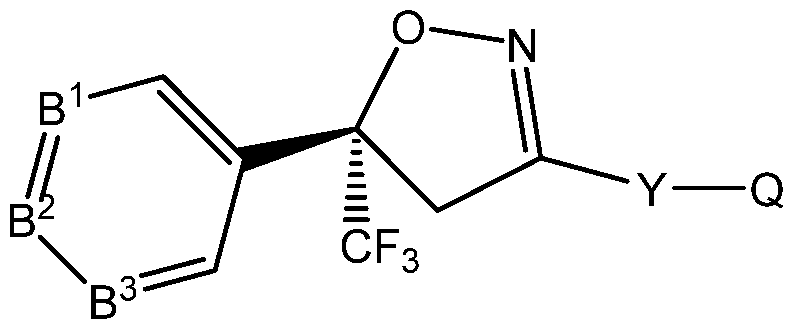
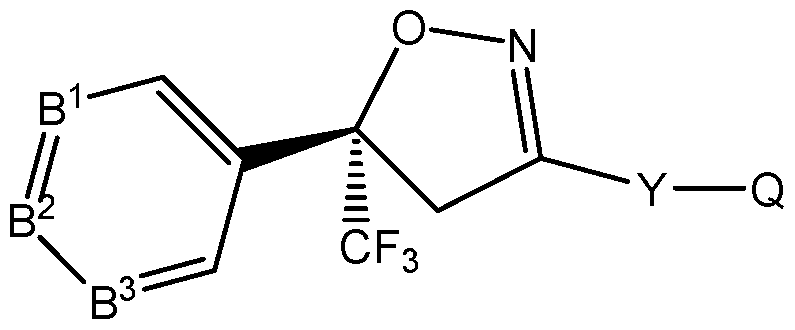
- An embodiment of the present invention provides for a method for reducing a withdrawal period when treating food-producing animals, including livestock, for parasitic infestations, which comprises administering to the animal an effective amount of an (S) enantiomer of an isoxazoline compound of the formula (S)-l :
- B 1 , B 2 , B 3 are each independently C-R or N; each R is independently H, halogen, cyano, -N0 2 , alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl;
- Q is X- R 2 R 3 , the group (-CH 2 -)(-CH 2 -)N-R 3 , OH, H 2 , alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
- R 2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
- R 3 is H, OR 7 , R 8 R 9 or Q 1 ; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R 4 ; or
- R 2 and R 3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N0 2 and alkoxy;
- each R 6 is independently halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN,— N0 2 , phenyl or pyridinyl;
- R 7 is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
- R 8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
- R 9 is H; Q 3 ; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R 4 ; or
- R 8 and R 9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N0 2 and alkoxy;
- Q 1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R 5 ;
- Q 2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 6 ;
- Q 3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 6 ;
- n 0, 1 or 2; and an inert carrier or formulation adjuvant;
- Another embodiment of the present invention is a method for reducing at least one unwanted effect while treating an animal for a parasitic infestation, which comprises administering to the animal an effective amount of an (S) enantiomer of an isoxazoline compound of formula (S)-l, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S) enantiomer of the isoxazoline compound of the formula (S)-l or pharmaceutically acceptable salt thereof,
- B 1 , B 2 , B 3 are each independently C-R or N; each R is independently H, halogen, cyano, -N0 2 , alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl;
- Q is X- R 2 R 3 , the group (-CH 2 -)(-CH 2 -)N-R 3 , OH, H 2 , alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
- R 2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
- R 3 is H, OR 7 , R 8 R 9 or Q 1 ; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R 4 ; or
- R 2 and R 3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— ⁇ 0 2 and alkoxy; each R 4 is independently halogen; alkyl, cycloalkyl, alkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, haloalkylamino, dialkylamino, dihaloalkylamino, cycloalkylamino, alkylcarbonyl, alkoxycarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, hal
- R 7 is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
- R 8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
- R 9 is H; Q 3 ; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R 4 ; or R 8 and R 9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N0 2 and alkoxy;
- Q 1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one
- Q 2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 6 ;
- Q 3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 6 ; and n is 0, 1 or 2; and an inert carrier or formulation diluent, wherein the compound or composition contains less than about 10% by weight (e.g., less than about 5% by weight, less than about 2% by weight, or less than about 1% by weight) of the (R) enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
- the present invention provides a method for reducing at least one unwanted effect while treating an animal (including a human) to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an ( ⁇ -enantiomer of an isoxazoline compound of formula (S)-l, as described above, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the ( ⁇ -enantiomer of the isoxazoline compound of formula (S)-l, or pharmaceutically acceptable salt thereof, and an inert carrier or a formulation diluent, wherein the compound or composition contains less than about 10% by weight of the (R)-enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
- Another embodiment of the present invention is a method for reducing at least one unwanted effect while treating an animal for a parasitic infestation with an isoxazoline compound, or a pharmaceutically acceptable salt thereof, by reducing the binding of the isoxazoline compound to an off-target receptor, which comprises administering to said animal the ( ⁇ -enantiomer of the isoxazoline compound of formula (S)-l, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising an effective amount of the (S)- enantiomer of an isoxazoline compound of the formula (S)-l, or pharmaceutically acceptable salt thereof:
- B 1 , B 2 , B 3 are each independently C-R or N; each R is independently H, halogen, cyano, -N0 2 , alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl;
- Q is X- R 2 R 3 , the group (-CH 2 -)(-CH 2 -)N-R 3 , OH, H 2 , alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
- R 2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
- R 3 is H, OR 7 , R 8 R 9 or Q 1 ; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R 4 ; or
- R 2 and R 3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— ⁇ 0 2 and alkoxy; each R 4 is independently halogen; alkyl, cycloalkyl, alkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, haloalkylamino, dialkylamino, dihaloalkylamino, cycloalkylamino, alkylcarbonyl, alkoxycarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, hal
- R 7 is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
- R 8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
- R 9 is H; Q 3 ; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R 4 ; or R 8 and R 9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N0 2 and alkoxy;
- Q 1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one
- Q 2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 6 ;
- Q 3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 6 ;
- n 0, 1 or 2;
- the parasiticidal compound or composition contains less than about 10% by weight (e.g., less than about 5% by weight, less than about 2% by weight, or less than about 1% by weight) of an (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof.
- Another embodiment of the present invention provides for a method for treating or preventing a parasitic infestation in an animal in need thereof while reducing at least one unwanted effect, which comprises administering an effective amount of an (S) enantiomer of an isoxazoline compound of formula (S)-l, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S) enantiomer of an isoxazoline compound of the formula (S)-l, or a pharmaceutically acceptable salt thereof:
- B 1 , B 2 , B 3 are each independently C-R or N; each R is independently H, halogen, cyano, -N0 2 , alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl;
- Q is X- R 2 R 3 , the group (-CH 2 -)(-CH 2 -)N-R 3 , OH, H 2 , alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
- R 2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
- R 3 is H, OR 7 , R 8 R 9 or Q 1 ; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R 4 ; or
- R 2 and R 3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N0 2 and alkoxy; each R 4 is independently halogen; alkyl, cycloalkyl, alkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, haloalkylamino, dialkylamino, dihaloalkylamino, cycloalkylamino, alkylcarbonyl, alkoxycarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, halo
- R 7 is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
- R 8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
- R 9 is H; Q 3 ; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R 4 ; or
- R 8 and R 9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N0 2 and alkoxy;
- Q 1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R 5 ;
- Q 2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 6 ;
- Q is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 6 ; and
- n is 0, 1 or 2; and an inert carrier or formulation adjuvant; wherein the compound or composition contains less than about 10% by weight (e.g., less than about 5% by weight, less than about 2% by weight, or less than about 1% by weight) of the (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof; and wherein said parasiticidal composition is effective against at least 90 % of the fleas and the ticks (e.g., 95%, 96%, 97%, 98%, 99% or 100%)) or for at least for 3 to 6 months (e.g.
- Another embodiment of the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of an arthropod- or mosquito-borne disease which comprises administering an effective amount of an (S) enantiomer of an isoxazoline compound of formula (S)-l to the animal, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S) enantiomer of an isoxazoline compound of the formula (S)-l, or a pharmaceutically acceptable salt thereof:
- B 1 , B 2 , B 3 are each independently C-R or N; each R is independently H, halogen, cyano, -N0 2 , alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl; Y is an optionally substituted phenylene, naphthylene, indanylene, a 5- or 6-membered heteroarylene or an 8-10-membered fused heterobicyclylene, wherein the optional substituents are selected from the group consisting of halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy,
- Q is X- R 2 R 3 , the group (-CH 2 -)(-CH 2 -)N-R 3 , OH, H 2 , alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
- R 2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxy carbonyl;
- R 3 is H, OR 7 , R 8 R 9 or Q 1 ; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R 4 ; or
- R 2 and R 3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N0 2 and alkoxy; each R 4 is independently halogen; alkyl, cycloalkyl, alkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, haloalkylamino, dialkylamino, dihaloalkylamino, cycloalkylamino, alkylcarbonyl, alkoxycarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, halo
- R 7 is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
- R 8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
- R 9 is H; Q 3 ; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R 4 ; or
- R 8 and R 9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N0 2 and alkoxy;
- Q 1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R 5 ;
- Q 2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 6 ;
- Q 3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 6 ; and n is 0, 1 or 2; and an inert carrier or formulation adjuvant; wherein the compound or composition contains less than about 10% by weight (e.g., less than about 5% by weight, less than about 2% by weight, or less than about 1% by weight) of the (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof; and wherein said parasiticidal composition is effective against at least 90 % of the fleas and the ticks (e.g., 95%, 96%, 97%, 98%, 99% or 100%)) or for at least for 3 to 6 months (e.g., at least 3, 4, 5 or 6 months).
- the compound or composition contains less than about 10% by weight (e.g., less than about 5% by weight, less than about 2% by weight, or less than about 1%
- the invention provides for any of the methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an antiparasitic isoxazoline compound of formula (S -II
- a , A z , A , A", A and A are independently selected from the group consisting of CR and N, provided that at most 3 of A 1 , A 2 , A 3 , A 4 , A 5 and A 6 are N;
- B 1 , B 2 and B 3 are independently selected from the group consisting of CR 2 and N;
- W is O or S
- each R 2 is independently H, halogen, Ci-C 6 alkyl, Ci-C 6 haloalkyl, Ci-C 6 alkoxy, Ci-C 6 haloalkoxy, Ci-C 6 alkylthio, Ci-C 6 haloalkylthio, Ci-C 6 alkylsulfinyl, Ci-C 6 haloalkylsulfinyl, Ci- C 6 alkylsulfonyl, Ci-C 6 haloalkylsulfonyl, Ci-C 6 alkylamino, C 2 -C 6 dialkylamino, C 2 -C 4 alkoxycarbonyl,— CN or— N0 2 ;
- each R 3 is independently H, halogen, Ci-C 6 alkyl, Ci-C 6 haloalkyl, C 3 -C 6 cycloalkyl, C 3 - C 6 halocycloalkyl, Ci-C 6 alkoxy, Ci-C 6 haloalkoxy, Ci-C 6 alkylthio, Ci-C 6 haloalkylthio, Ci-C 6 alkylsulfinyl, Ci-C 6 haloalkylsulfinyl, Ci-C 6 alkylsulfonyl, Ci-C 6 haloalkylsulfonyl, Ci-C 6 alkylamino, C 2 -C6 dialkylamino,— CN or— N0 2 ;
- R 4 is H, Ci-C 6 alkyl, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, C 3 -C 6 cycloalkyl, C 4 -C 7 alkylcycloalkyl, C 4 -C 7 cycloalkylalkyl, C 2 -C 7 alkylcarbonyl or C 2 -C 7 alkoxycarbonyl;
- R 5 is H, OR 10 , R U R 12 or Q 1 ; or Ci-C 6 alkyl, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, C 3 -C 6 cycloalkyl, C 4 -C 7 alkylcycloalkyl or C 4 -C 7 cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R 7 ; or
- R 4 and R 5 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of Ci-C 2 alkyl, halogen,— CN,— N0 2 and Ci-C 2 alkoxy; each R 7 is independently halogen; Ci-C 6 alkyl, C 3 -C 6 cycloalkyl, Ci-C 6 alkoxy, Ci-C 6 alkylthio, Ci-C 6 alkylsulfinyl, Ci-C 6 alkylsulfonyl, Ci-C 6 alkylamino, C 2 -C 8 dialkylamino, C 3 -C 6 cycloalkylamino, C 2 -C 7 alkylcarbonyl, C 2 -C 7 alkoxy carbonyl, C 2 -C 7 alkylaminocarbon
- each R 8 is independently halogen, Ci-C 6 alkoxy, Ci-C 6 haloalkoxy, Ci-C 6 alkylthio, Ci-C 6 haloalkylthio, Ci-C 6 alkylsulfinyl, Ci-C 6 haloalkylsulfinyl, Ci-C 6 alkylsulfonyl, Ci-C 6 haloalkylsulfonyl, Ci-C 6 alkylamino, C 2 -C 6 dialkylamino, C 2 -C 4 alkoxycarbonyl,— CN or— N0 2 ;
- each R 9 is independently halogen, Ci-C 6 alkyl, Ci-C 6 haloalkyl, C 3 -C 6 cycloalkyl, C 3 -C 6 halocycloalkyl, Ci-C 6 alkoxy, Ci-C 6 haloalkoxy, Ci-C 6 alkylthio, Ci-C 6 haloalkylthio, Ci-C 6 alkylsulfinyl, Ci-C 6 haloalkylsulfinyl, Ci-C 6 alkylsulfonyl, Ci-C 6 haloalkylsulfonyl, Ci-C 6 alkylamino, C 2 -C 6 dialkylamino,— CN,— N0 2 , phenyl or pyridinyl;
- R 10 is H; or Ci-C 6 alkyl, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, C 3 -C 6 cycloalkyl, C 4 -C 7 alkylcycloalkyl or C 4 -C 7 cycloalkylalkyl, each optionally substituted with one of more halogen;
- R 11 is H, Ci-C 6 alkyl, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, C 3 -C 6 cycloalkyl, C 4 -C 7 alkylcycloalkyl, C 4 -C 7 cycloalkylalkyl, C 2 -C 7 alkylcarbonyl or C 2 -C 7 alkoxycarbonyl;
- R 12 is H; Q 3 ; or Ci-C 6 alkyl, C 2 -C 6 alkenyl, C 2 -C 6 alkynyl, C 3 -C 6 cycloalkyl, C 4 -C 7 alkylcycloalkyl or C 4 -C 7 cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R 7 ; or
- R 11 and R 12 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of C 1 -C 2 alkyl, halogen,— CN,— ⁇ 0 2 and C 1 -C 2 alkoxy;
- Q 1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R 8 ;
- each Q 2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 9 ;
- Q 3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R 9 ;
- n 0, 1 or 2; or a pharmaceutically acceptable salt thereof, or
- the parasiticidal composition comprises a compound of formula (S)-II and an inert carrier or formulation adjuvant.
- the invention provides for any of the methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an antiparasitic isoxazoline compound of formula ( ⁇ -IIA:
- X 1 , X 2 and X 3 are each independently H, bromo, chloro, fluoro, iodo or CF 3 ; or wherein the parasiticidal composition comprises a compound of formula (S)-IIA and an inert carrier or formulation adjuvant.
- the parasiticidal compound is an isoxazoline compound of formula ( ⁇ -IIA where X 1 is CI, X 2 is H and X 3 is CF 3 ("Compound A").
- the parasiticidal compound is a compound of (5)-IIA where X 1 is CI, X 2 is F and X 3 is CF 3 .
- the parasiticidal compound is a compound of formula ( ⁇ -IIA where X 1 is CI, X 2 is H and X 3 is CI.
- the parasiticidal compound is a compound of formula ( ⁇ -IIA where X 1 is CI, X 2 is F and X 3 is CI.
- the invention provides for any of the inventive methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an antiparasitic isoxazoline compound of formula (S)-III:
- (S)-m wherein X 1 , X 2 and X 3 are each independently H, bromo, chloro, fluoro, iodo or CF 3 , or wherein the parasiticidal composition comprises a compound of formula (S)-III and an inert carrier or formulation adjuvant.
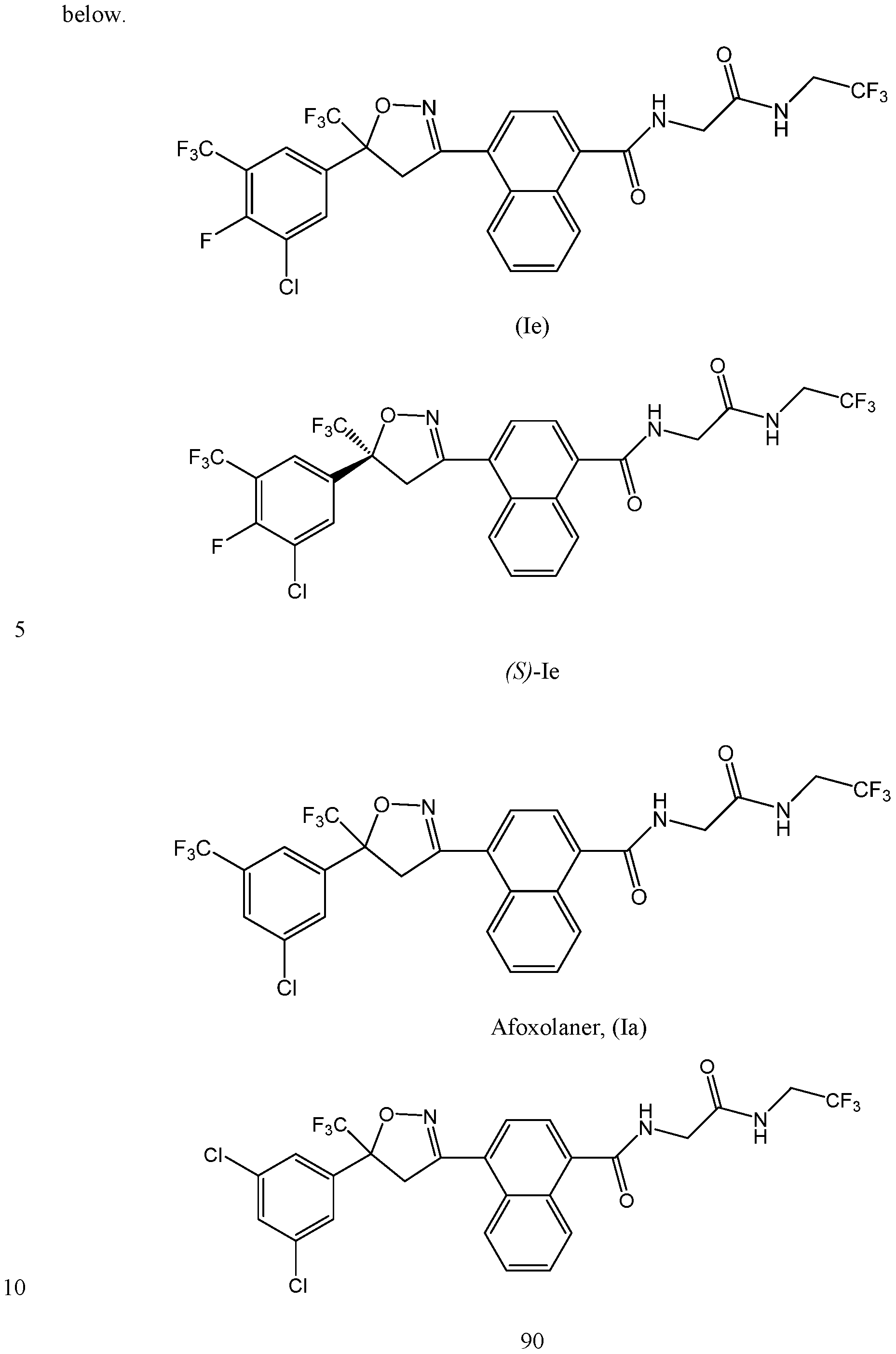
- the racemic compound corresponding to formula (S)-III wherein X 1 and X 3 are chloro and X 2 is hydrogen has the international non-proprietary name (INN) fluralaner.
- Fluralaner is the active ingredient in the product BRAVECTOTM flavored chews for dogs and topical solution for cats. BRAVECTOTM is indicated for the treatment and prevention of flea infestations and certain tick infestations.
- EPMAR European Medicines Agency published a European public MRL assessment report (EPMAR) for the use of the racemic compound fluralaner for use in poultry for the treatment and control of red poultry mite infestations.
- Fluralaner is a racemic compound composed of equal amounts of the (R)- and ( ⁇ -enantiomer of the compound.
- the parasiticidal composition comprises an isoxazoline compound of formula ( ⁇ -III where X 1 is CI, X 2 is H and X 3 is CF 3 .
- the composition comprises a compound of formula ( ⁇ -III where X 1 is CI, X 2 is F and X 3 is CF 3 .
- the composition comprises a compound of formula ( ⁇ -III where X 1 is CI, X 2 is H and X 3 is CI.
- the composition comprises a compound of formula ( ⁇ -III where X 1 is CI, X 2 is F and X 3 is CI.
- the invention provides a method for reducing a withdrawal period when treating livestock for parasitic infestations, which comprises administering an effective amount of a compound of formula (5)- ⁇ , wherein X 1 is CI, X 2 is H and X 3 is CI, or a parasiticidal composition comprising the compound.
- the invention provides a method for reducing a withdrawal period when treating cattle for parasitic infestations, which comprises administering an effective amount of a compound of formula (S)-III, wherein X 1 is CI, X 2 is H and X 3 is CI, or a parasiticidal composition comprising the compound.
- in another embodiment of the present invention is a method for reducing at least one unwanted effect while treating an animal for a parasitic infestation, which comprises administering an effective amount of a compound of formula (5)- ⁇ , wherein X 1 is CI, X 2 is H and X 3 is CI, or a parasiticidal composition comprising the compound.
- a method for reducing at least one unwanted effect while treating an animal for a parasitic infestation with an isoxazoline compound or a salt thereof by reducing the binding of an isoxazoline compound to an off-target receptor which comprises administering to said non-human animal an effective amount of a compound of formula (5)- ⁇ , wherein X 1 is CI, X 2 is H and X 3 is CI, or a parasiticidal composition comprising an effective amount of the compound.
- the present invention provides a method of treating or controlling red poultry mites in chickens comprising administering to the chickens an effective amount of the compound of formula (S)-III, wherein X 1 is CI, X 2 is H and X 3 is CI, in drinking water.
- the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an ( ⁇ -enantiomer of an isoxazoline compound of formula (5)- ⁇ , wherein X 1 is CI, X 2 is H or F and X 3 is CI or CF 3 , or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the ( ⁇ -enantiomer of the isoxazoline compound or salt and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)- enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
- the invention provides for any of the inventive methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an antiparasitic isoxazoline compound of formula I:
- X 1 , X 2 and X 3 are each independently H, bromo, chloro, fluoro, iodo or CF 3 ;
- G is G-l or G-2;
- Bi, B 2 , B 3 , B 4 and B 5 are independently N or C-R9;
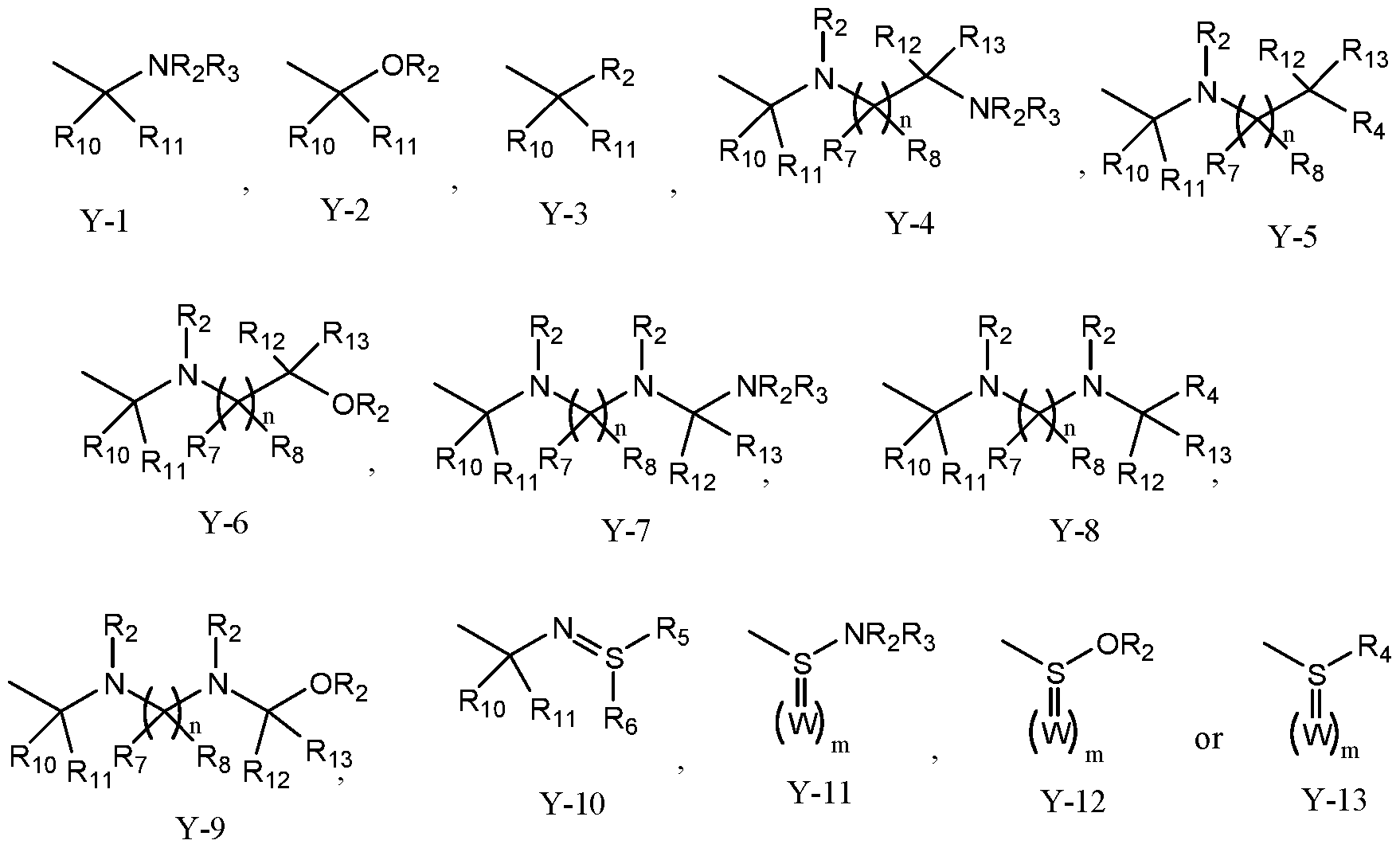
- Y is Y-1, Y-2, Y-3, Y-4, Y-5, Y-6, Y-7, Y-8, Y-9, Y-10, Y-1 1, Y-12 or Y-13;
- R 2 , R-3 are independently hydrogen, alkyl, haloalkyl, thioalkyl, alkylthioalkyl, hydroxyalkyl, alkoxylakyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, Ri 0 S(O)-, RioS(0) 2 -, RioC(O)-, RioC(S)-, Ri 0 RnNC(O)-, Ri 0 RiiNC(S)- RioOC(O)-;
- R4, R5 and R 6 are independently hydrogen, alkyl, haloalkyl, thioalkyl, alkylthioalkyl, hydroxyalkyl, alkoxylakyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, aryl or heteroaryl;
- R 7 and R 8 are independently hydrogen, alkyl, haloalkyl, thioalkyl, alkylthioalkyl, hydroxyalkyl, alkoxylakyl, alkenyl, haloalkenyl, alkynyl or haloalkynyl;
- R9 is hydrogen, halogen, -CN, or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, halocycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each which is unsubstituted or substituted with one or more of halogen, hydroxy, amino, alkyl- or di(alkyl)amino, alkyl, cycloalkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, R 7 S(0)-, R 7 S(0) 2 -, R 7 C(0)-, R 7 R 8 NC(0)-, R 7 OC(0)-, R 7 C(0)0-, R 7 C(0) R 8 -, -CN
- W is O, S or R 2 ;
- n 1-4;
- n 0, 1 or 2; or a pharmaceutically acceptable salt thereof, or wherein the parasiticidal composition comprises a compound of formula (,S)-IV and an inert carrier or formulation adjuvant.
- the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an ( ⁇ -enantiomer of an isoxazoline compound of formula (,S)-IV described above, wherein X 1 is CI, X 2 is H, CI or F and X 3 is CI or CF 3 , or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (,S)-enantiomer of the isoxazoline compound, or a pharmaceutically acceptable salt thereof, and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)-enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
- the parasiticidal compound or composition comprises an isoxazoline compound of formula (,S)-IV where X 1 is CI, X 2 is H and X 3 is CF 3 .
- the compound or composition comprises a compound of formula (,S)-IV where X 1 is CI, X 2 is F and X 3 is CF 3 .
- the compound or composition comprises a compound of formula (,S)-IV where X 1 is CI, X 2 is H and X 3 is CI.
- the compound or composition comprises a compound of formula (,S)-IV where X 1 is CI, X 2 is F and X 3 is CI.
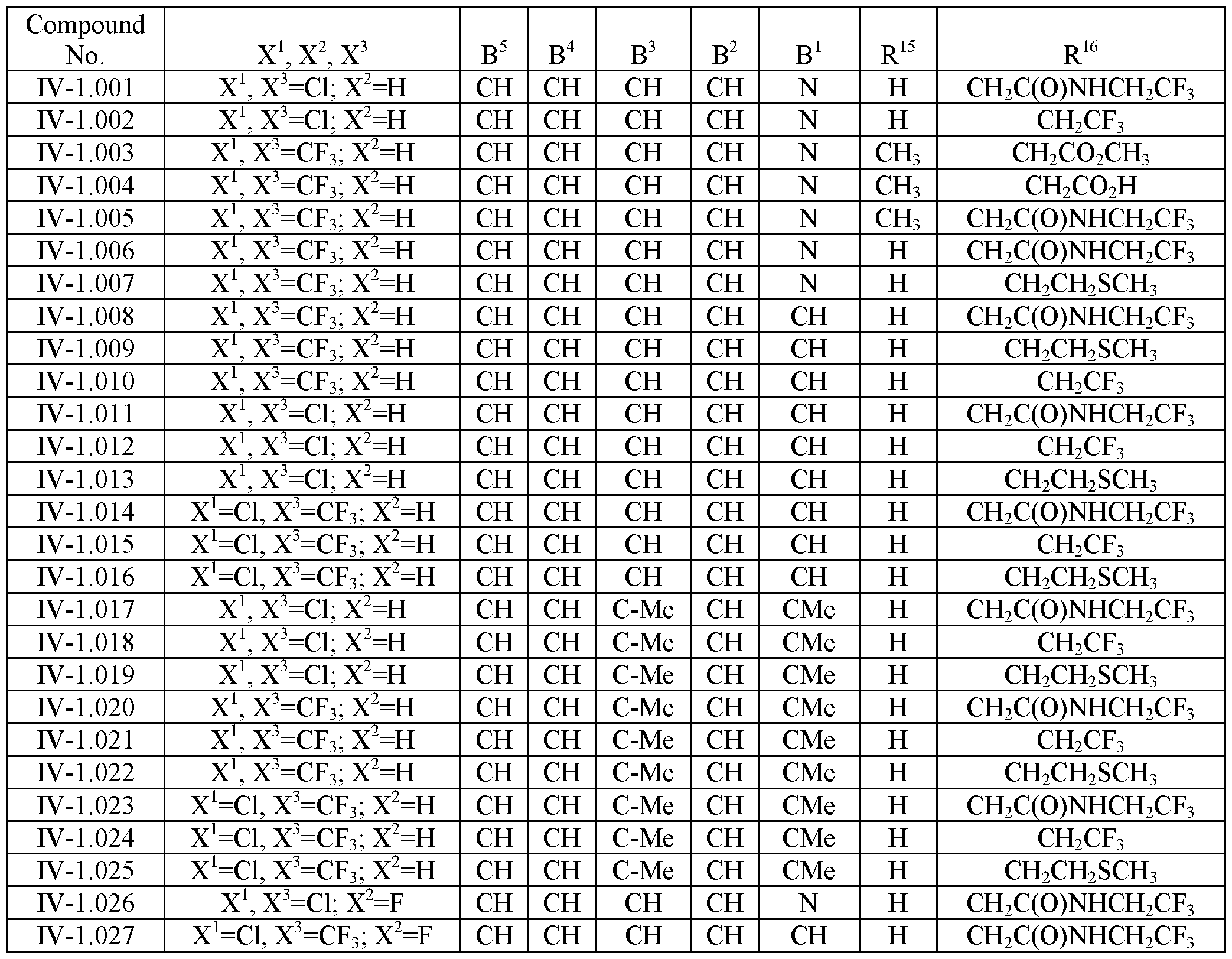
- the invention provides for any of the inventive methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an antiparasitic isoxazoline compound of formula IV-1.001 to IV-1.027 or IV-2.001 to IV-2.020:
- the parasiticidal composition comprises a compound of formula IV-1.001 to
- the invention provides for any of the inventive methods provided for above wherein the parasiticidal compound is an (S) enantiomer of antiparasitic isoxazoline compound of formula ( ⁇ S)-V:
- the parasiticidal composition comprises an isoxazoline compound of formula (S)-V where X 1 is CI, X 2 is H and X 3 is CF 3 .
- the composition comprises a compound of formula (S)-V where X 1 is CI, X 2 is F and X 3 is CF 3 .
- the composition comprises a compound of formula (S)-V where X 1 is CI, X 2 is H and X 3 is CI.
- the composition comprises a compound of formula (S)-V where X 1 is CI, X 2 is F and X 3 is CI.
- the composition comprises a compound of formula (S)-V where X 1 , X 2 and X 3 are each CI.
- the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an ( ⁇ -enantiomer of an isoxazoline compound of formula (S)-V, wherein X 1 is CI, X 2 is H, CI or F and X 3 is CI or CF 3 , or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the ( ⁇ -enantiomer of the isoxazoline compound or salt and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)- enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
- the invention provides for any of the embodiments of the methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an isoxazoline compound of the formula
- X 1 , X 2 and X 3 are each independently H, bromo, chloro, fluoro, iodo or CF 3 , or wherein the parasiticidal composition comprises a compound of formula (5)- VI and an inert carrier or formulation adjuvant.
- the parasiticidal composition comprises an isoxazoline compound of formula (5)- VI where X 1 is CI, X 2 is H and X 3 is CF 3 .
- the composition comprises a compound of formula (5)- VI where X 1 is CI, X 2 is F and X 3 is CF 3 .
- the composition comprises a compound of formula (5)- VI where X 1 is CI, X 2 is H and X 3 is CI.
- the composition comprises a compound of formula (5)- VI where X 1 is CI, X 2 is F and X 3 is CI.
- the composition comprises a compound of formula (S)-VI where each of X 1 , X 2 and X 3 is CI.
- the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an ( ⁇ -enantiomer of an isoxazoline compound of formula (S)-VL, wherein X 1 is CI, X 2 is H, CI or F and X 3 is CI or CF 3 , or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the ( ⁇ -enantiomer of the isoxazoline compound or salt and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)- enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
- the invention provides for any of the embodiments of the inventive methods provided for above wherein the compound is an (S) enantiomer of an isoxazoline compound of the formula
- composition comprises a compound of formula (,S)-VII and an inert carrier or formulation adjuvant.
- the parasiticidal composition comprises an isoxazoline compound of formula (,S)-VII where X 1 is CI, X 2 is H and X 3 is CF 3 .
- the compositions comprise a compound of formula (,S)-VII where X 1 is CI, X 2 is F and X 3 is CF 3 .
- the composition comprises a compound of formula (,S)-VII where X 1 is CI, X 2 is H and X 3 is CI.
- the composition comprises a compound of formula (,S)-VII where X 1 is CI, X 2 is F and X 3 is CI. In yet another embodiment of the inventive methods the composition comprises a compound of formula (S)- VII, X 1 , X 2 and X 3 are each CI.
- the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an ( ⁇ -enantiomer of an isoxazoline compound of formula (S)-VII, wherein X 1 is CI, X 2 is H, CI or F and X 3 is CI or CF 3 , or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the ( ⁇ -enantiomer of the isoxazoline compound or salt and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)-enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
- the parasiticidal compositions is substantially free of an (R) enantiomer of the isoxazoline compound. In another embodiment of the inventive methods provided for above, the parasiticidal composition comprises no (R) enantiomer of an isoxazoline compound.
- the withdrawal period is reduced by about 30 to 70%, for example about 40-60%) or about 45-55%> (e.g., about 50%>) over a 40 to 60 day withdrawal period calculated for administration of the racemic isoxazoline compound.
- the withdrawal period is reduced by about 30 to 70%, for example about 40-60%> or about 45-55%> (e.g., about 50%>) over an 80 to 100 day withdrawal period calculated for administration of the racemic isoxazoline compound.
- the parasiticidal compositions are oral formulations, such as, for example, a tablet, a paste, a chewable tablet or a soft chew.
- the parasiticidal compositions are topical formulations, such as, for example, a spot-on or pour-on.
- the parasiticidal composition is dissolved in the drinking water.
- the parasiticidal composition is an injectable formulation, such as, for example an long acting injectable formulation or an extended release injectable formulation.
- the unwanted effects that may be reduced by the inventive methods include neurological disorders, such as seizures, convulsions, restlessness, muscle tremors, insomnia, nervousness, delirium, depression, mydriasis and hyperesthesia.
- the unwanted effect is weight loss in the animal during treatment (e.g., the animal does not lose more that 10%> of its body weight before treatment).
- the unwanted effects are vomiting, dry or flaky skin, diarrhea, lethargy or anorexia or a combination of the foregoing.
- the off-site receptors included by the inventive methods are: Sodium Channel, Site 2; Calcium Channel L-Type, Dihydropyridine; Transporter, Norepinephrine (NET);Transporter, Dopamine (DAT); Leukotriene, Cysteinyl CysLTl ; Dopamine Dl ; GABAA, Chloride Channel, TBOB; Adenosine A3; Cannabinoid CB 1 ; Calcium Channel L-Type, Benzothiazepine; Dopamine D3; Adrenergic alpha2A; Opiate mu (OP3, MOP); Serotonin (5- Hydroxytryptamine) 5-HT2B; Adrenergic betal ; Adrenergic alphalA; Prostanoid EP4; Glucocorticoid; Muscarinic M2; Potassium Channel hERG; Thyroid Hormone; Muscarinic Ml; Serotonin (5-Hydroxytryptamine) 5-HT1A; Melat
- the offsite targets included by the inventive methods include off- target receptors are Calcium Channel L-type receptors, Na Channel Site 2 receptors, Norepinephrine Cellular Transport receptors, Dopamine Cellular Transport receptors, CB-1 receptors, Adenosine a 3 receptors, Leukotriene, Cysteinyl Cys T receptors, Histamine 2 receptors, or Sodium Channel Site 2 receptors. Definitions
- the "withdrawal period” as used herein refers to the time interval that must pass between the last administration of a veterinary bioactive agent to a food producing animal and when the food producing animal can safely enter the food supply or its milk/eggs can be safely consumed.
- the withdrawal period is set be a governmental body such as the FDA in the United States or the EMA in Europe.
- treating means the administration of a parasiticidal composition used in the inventive methods to a non-human animal that has a parasitic infestation for the eradication of the parasite or the reduction of the number of the parasites infesting the animal undergoing treatment. It is noted that the parasiticidal compositions of the inventive methods may be used to prevent such a parasitic infestation.
- prevent means the administration of a parasiticidal composition used in the inventive methods to a non-human animal before the parasitic infection or infestation has occurred in order to keep said infection or infestation from occurring.
- reducing the withdrawal period means that the calculated withdrawal period using algorithms approved by relevant Regulatory authorities (e.g. FDA, EMA, etc.) has been reduced as compared to the withdrawal period when the racemic mixture of the isoxazoline compound or pharmaceutically acceptable salt thereof is administered to the non-human animal (at the approved dose).
- relevant Regulatory authorities e.g. FDA, EMA, etc.
- reducing at least one unwanted effect means that the occurrence of an unwanted or unintended biological effect, which is observed when the racemic mixture or the (R) enantiomer of the isoxazoline compound or pharmaceutically acceptable salt thereof is administered to the non-human animal, is considered to be reduced in severity and/or frequency by one of ordinary skill in this art.
- an effective amount it is intended a sufficient amount of a composition of the invention to eradicate or reduce the number of parasites infesting the animal.
- an effective amount of the active agent achieves at least 70% efficacy (% reduction vs. control) against the target parasite.
- an effective amount of the active agent achieves at least 80%, or at least 90% efficacy against the target pests.
- an effective amount of the bioactive agent will achieve at least 95%, at least 98% or 100% efficacy against the target parasites.
- organic moieties mentioned in the definitions of the variables of formula (S)-I- formula (,S)-VII are - like the term halogen - collective terms for individual listings of the individual group members.
- the prefix C n -C m indicates in each case the possible number of carbon atoms in the group.
- long acting or “long acting formulation” or “long acting composition” as used herein means a dosage form that is formulated in such a manner to make the bioactive agent(s) contained therein to be effective against the parasites of at least about 80% for two months, or longer.
- Long acting formulations are distinguished from “extended release” formulations in that they do not include pharmaceutically acceptable biodegradable polymers. For avoidance of doubt, polyethylene glycols (PEGs) and poloxamers are not considered biodegradable polymers.
- the long acting formulations are effective against the parasites of about 90% for about 3 months, or longer.
- the long acting formulations are effective against the parasites of about 95% for about 3 months, or longer.
- This definition is consistent with the use of the term known and accepted in the veterinary field as described in the article "Terminology Challenges: Defining Modified Release Dosage Forms in Veterinary Medicine” by Marilyn N. Martinez, Danielle Lindquist and Sanja Modric (Journal of Pharmaceutical Sciences, vol. 99, no. 8, August 2010).
- the extended release formulations according to the present invention would be understood to provide an efficacy of at least 90% against fleas and/or ticks for at least 3 months as described herein.
- non-human animal is used herein to include all mammals excluding humans, birds and fish and also include all vertebrate animals. Animals include, but are not limited to, cats, dogs, cattle, chickens, cows, deer, goats, horses, llamas, pigs, sheep and yaks. It also includes an individual animal in all stages of development, including embryonic and fetal stages.
- substantially pure is used herein to indicate that a compound or an enantiomer is at least about 90% (w/w) pure, at least about 95% (w/w), or at least about 98%> (w/w) pure, or higher.
- alkyl refers to saturated straight, branched, primary, secondary or tertiary hydrocarbons, including those having 1 to 20 atoms.
- alkyl groups will include C 1 -C 12 , Ci-Ci 0 , Ci-C 8 , Ci-C 6 or C 1 -C4 alkyl groups.
- Ci-Ci 0 alkyl examples include, but are not limited to, methyl, ethyl, propyl, 1-methylethyl, butyl, 1-methylpropyl, 2- methylpropyl, 1, 1-dimethylethyl, pentyl, 1-methylbutyl, 2-methylbutyl, 3-methylbutyl, 2,2- dimethylpropyl, 1-ethylpropyl, hexyl, 1,1-dimethylpropyl, 1,2-dimethylpropyl, 1-methylpentyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 1,3- dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3-dimethylbutyl, 1-ethylbutyl, 2- ethylbutyl, 1, 1,2-trimethylpropyl, 1,2,2-trimethylpropyl, 1
- Cyclic alkyl groups or "cycloalkyl” include those with 3 to 10 carbon atoms having single or multiple condensed rings.
- cycloalkyl groups include C 4 -C 7 or C 3 -C 4 cyclic alkyl groups.
- Non-limiting examples of cycloalkyl groups include adamantyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl and the like.
- alkyl groups described herein can be unsubstituted or substituted with one or more moieties selected from the group consisting of alkyl, halo, haloalkyl, hydroxyl, carboxyl, acyl, acyloxy, amino, alkyl- or dialkylamino, amido, arylamino, alkoxy, aryloxy, nitro, cyano, azido, thiol, imino, sulfonic acid, sulfate, sulfonyl, sulfanyl, sulfinyl, sulfamoyl, ester, phosphonyl, phosphinyl, phosphoryl, phosphine, thioester, thioether, acid halide, anhydride, oxime, hydrazine, carbamate, phosphoric acid, phosphate, phosphonate, or any other viable functional group that does not inhibit the biological activity of the compounds of the invention,
- alkyl such as “alkylcycloalkyl,” “cycloalkylalkyl,”
- alkylamino or “dialkylamino” will be understood to comprise an alkyl group as defined above linked to the other functional group, where the group is linked to the compound through the last group listed, as understood by those of skill in the art.
- alkenyl refers to both straight and branched carbon chains which have at least one carbon-carbon double bond.
- alkenyl groups may include C 2 -C 20 alkenyl groups.
- alkenyl includes C 2 -C 12 , C 2 -C 10 , C 2 -C8, C 2 -C6 or C 2 -C4 alkenyl groups.
- the number of double bonds is 1-3, in another embodiment of alkenyl, the number of double bonds is one or two. Other ranges of carbon- carbon double bonds and carbon numbers are also contemplated depending on the location of the alkenyl moiety on the molecule.
- C 2 -Cio-alkenyl groups may include more than one double bond in the chain. Examples include, but are not limited to, ethenyl, 1-propenyl, 2-propenyl, 1- methyl-ethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1 -methyl- 1-propenyl, 2-m ethyl- 1-propenyl, 1- methyl-2-propenyl, 2-methyl-2-propenyl; 1-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1- m ethyl- 1-butenyl, 2-m ethyl- 1-butenyl, 3 -methyl- 1-butenyl, l-methyl-2-butenyl, 2-methyl-2- butenyl, 3-methyl-2-butenyl, 1 -methyl -3-butenyl, 2-methyl-3-butenyl, 3-methyl-3-butenyl, 1,1- dimethyl-2
- alkynyl refers to both straight and branched carbon chains which have at least one carbon-carbon triple bond.
- the number of triple bonds is 1-3; in another embodiment of alkynyl, the number of triple bonds is one or two.
- alkynyl groups include from C 2 -C 20 alkynyl groups.
- alkynyl groups may include C 2 -C 12 , C 2 -C 10 , C 2 -C 8 , C 2 -C 6 or C 2 -C 4 alkynyl groups.
- Other ranges of carbon-carbon triple bonds and carbon numbers are also contemplated depending on the location of the alkenyl moiety on the molecule.
- C 2 -Cio-alkynyl refers to a straight-chain or branched unsaturated hydrocarbon group having 2 to 10 carbon atoms and containing at least one triple bond, such as ethynyl, prop-l-yn-l-yl, prop-2-yn-l-yl, n-but-l-yn- 1-yl, n-but-l-yn-3-yl, n-but-l-yn-4-yl, n-but-2-yn-l-yl, n-pent-l-yn-l-yl, n-pent-l-yn-3-yl, n- pent-l-yn-4-yl, n-pent-l-yn-5-yl, n-pent-2-yn-l-yl, n-pent-2-yn-4-yl, n-pent-2-yn-5-yl, 3- methyl
- haloalkyl refers to an alkyl group, as defined herein, which is substituted by one or more halogen atoms.
- Ci-C4-haloalkyl includes, but is not limited to, chloromethyl, bromomethyl, dichloromethyl, trichloromethyl, fluoromethyl, difluoromethyl, trifluoromethyl, chlorofluoromethyl, di chlorofluoromethyl, chlorodifluoromethyl, 1-chloroethyl, 1-bromoethyl, 1-fluoroethyl, 2-fluoroethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, 2-chloro-2- fluoroethyl, 2-chloro-2,2-difluoroethyl, 2,2-dichloro-2-fluoroethyl, 2,2,2-trichloroethyl, pentafluoroethyl and the like.
- haloalkenyl refers to an alkenyl group, as defined herein, which is substituted by one or more halogen atoms.
- haloalkynyl refers to an alkynyl group, as defined herein, which is substituted by one or more halogen atoms.
- Alkoxy refers to alkyl-O-, wherein alkyl is as defined above.
- alkenyloxy refers to alkyl-O-, wherein alkyl is as defined above.
- alkenyloxy refers to the groups alkenyl- 0-, alkynyl-O-, haloalkyl-O-, haloalkenyl-O-, haloalkynyl-O-, cycloalkyl-O-, cycloalkenyl-O-, halocycloalkyl-O-, and halocycloalkenyl-O-, respectively, wherein alkenyl, alkynyl, haloalkyl, haloalkenyl, haloalkynyl, cycloalkyl-O-, respectively, wherein alkenyl, alkynyl, haloalkyl, haloalkenyl, haloalkynyl, cycloalkyl-
- Ci-C 6 -alkoxy examples include, but are not limited to, methoxy, ethoxy, C 2 H 5 -CH 2 O-, (CH 3 ) 2 CHO-, n-butoxy, C 2 H 5 -CH(CH 3 )0-, (CH 3 ) 2 CH-CH 2 0- ; (CH 3 ) 3 CO-, n- pentoxy, 1-methylbutoxy, 2-methylbutoxy, 3-methylbutoxy, 1, 1-dimethylpropoxy, 1.2- dimethylpropoxy, 2,2-dimethyl-propoxy, 1-ethylpropoxy, n-hexoxy, 1 -methylpentoxy, 2- methylpentoxy, 3-methylpentoxy, 4-methylpentoxy, 1, 1-dimethylbutoxy, 1,2-dimethylbutoxy,
- alkylthio refers to alkyl-S-, wherein alkyl is as defined above.
- haloalkylthio refers to haloalkyl-S- and cycloalkyl-S- where haloalkyl and cycloalkyl are as defined above.
- alkyl sulfinyl refers to alkyl-S(O)-, wherein alkyl is as defined above.
- haloalkylsulfinyl refers to haloalkyl-S(O)- where haloalkyl is as defined above.
- alkyl sulfonyl refers to alkyl-S(0)2-, wherein alkyl is as defined above.
- haloalkylsulfonyl refers to haloalkyl-S(0)2- where haloalkyl is as defined above.
- alkylamino and dialkylamino refer to alkyl- H- and (alkyl) 2 N- where alkyl is as defined above.
- haloalkylamino refers to haloalkyl- H- where haloalkyl is as defined above.
- alkylcarbonyl alkoxycarbonyl
- alkylaminocarbonyl alkylaminocarbonyl
- dialkylaminocarbonyl refer to alkyl-C(O)-, alkoxy-C(O)-, alkylamino-C(O)- and dialkylamino-C(O)- where alkyl, alkoxy, alkylamino and dialkylamino are as defined above.
- haloalkylcarbonyl refers to the groups haloalkyl-C(O)-, haloalkoxy-C(O)-, haloalkylamino-C(O)- and dihaloalkylamino-C(O)- where haloalkyl, haloalkoxy, haloalkylamino and dihaloalkylamino are as defined above.
- Aryl refers to a monovalent aromatic carbocyclic group of from 6 to 14 carbon atoms having a single ring or multiple condensed rings.
- aryl groups include C 6 - Cio aryl groups.
- Aryl groups include, but are not limited to, phenyl, biphenyl, naphthyl, tetrahydronaphthyl, phenylcyclopropyl and indanyl.
- Aryl groups may be unsubstituted or substituted by one or more moieties selected from halogen, cyano, nitro, hydroxy, mercapto, amino, alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, haloalkyl, haloalkenyl, haloalkynyl, halocycloalkyl, halocycloalkenyl, alkoxy, alkenyloxy, alkynyloxy, haloalkoxy, haloalkenyloxy, haloalkynyloxy, cycloalkoxy, cycloalkenyloxy, halocycloalkoxy, halocycloalkenyloxy, alkylthio, haloalkylthio, cycloalkylthio, halocycloalkylthio, alkylsulfinyl, alkenylsulfinyl, alky
- aralkyl or "arylalkyl” refers to an aryl group that is bonded to the parent compound through a diradical alkylene bridge, (-CH 2 -) n , where n is 1-12 and where "aryl” is as defined above.
- Heteroaryl refers to a monovalent aromatic group of from 1 to 15 carbon atoms, preferably from 1 to 10 carbon atoms, having one or more oxygen, nitrogen, and sulfur heteroatoms within the ring, preferably 1 to 4 heteroatoms, or 1 to 3 heteroatoms.
- the nitrogen and sulfur heteroatoms may optionally be oxidized.
- Such heteroaryl groups can have a single ring (e.g., pyridyl or furyl) or multiple condensed rings provided that the point of attachment is through a heteroaryl ring atom.
- heteroaryls include pyridyl, piridazinyl, pyrimidinyl, pyrazinyl, triazinyl, pyrrolyl, indolyl, quinolinyl, isoquinolinyl, quinazolinyl, quinoxalinnyl, furanyl, thienyl, furyl, pyrrolyl, imidazolyl, oxazolyl, isoxazolyl, isothiazolyl, pyrazolyl benzofuranyl, and benzothienyl.
- Heteroaryl rings may be unsubstituted or substituted by one or more moieties as described for aryl above.
- the term "heteroarylene" (where the heteroaryl group is a bridging group) should be construed accordingly.
- Heterocyclyl refers to fully saturated or unsaturated, cyclic groups, for example, 3 to 7 membered monocyclic or 4 to 7 membered monocyclic; 7 to 11 membered bicyclic, or 10 to 15 membered tricyclic ring systems, which have one or more oxygen, sulfur or nitrogen heteroatoms in ring, preferably 1 to 4 or 1 to 3 heteroatoms.
- the nitrogen and sulfur heteroatoms may optionally be oxidized and the nitrogen heteroatoms may optionally be quaternized.
- the heterocyclic group may be attached at any heteroatom or carbon atom of the ring or ring system and may be unsubstituted or substituted by one or more moieties as described for aryl groups above.
- Exemplary monocyclic heterocyclic groups include, but are not limited to, pyrrolidinyl, pyrrolyl, pyrazolyl, oxetanyl, pyrazolinyl, imidazolyl, imidazolinyl, imidazolidinyl, oxazolyl, oxazolidinyl, isoxazolinyl, isoxazolyl, thiazolyl, thiadiazolyl, thiazolidinyl, isothiazolyl, isothiazolidinyl, furyl, tetrahydrofuryl, thienyl, oxadiazolyl, piperidinyl, piperazinyl, 2- oxopiperazinyl, 2-oxopiperidinyl, 2-oxopyrrolodinyl, 2-oxoazepinyl, azepinyl, 4-piperidonyl, pyridinyl, pyrazinyl, pyrimi
- bicyclic heterocyclic groups include, but are not limited to, indolyl, benzothiazolyl, benzoxazolyl, benzodioxolyl, benzothienyl, quinuclidinyl, quinolinyl, tetra- hydroisoquinolinyl, isoquinolinyl, benzimidazolyl, benzopyranyl, indolizinyl, benzofuryl, chromonyl, coumarinyl, benzopyranyl, cinnolinyl, quinoxalinyl, indazolyl, pyrrol opyridyl, furopyridinyl (such as furo[2,3-c]pyridinyl, furo[3,2-b]pyridinyl]or furo[2,3-b]pyridinyl), dihydroisoindolyl, dihydroquinazolinyl (such as 3,4-dihydro-4-oxo-
- Exemplary tricyclic heterocyclic groups include carbazolyl, benzidolyl, phenanthrolinyl, acridinyl, phenanthridinyl, xanthenyl, and the like.
- Halogen means the atoms fluorine, chlorine, bromine and iodine.
- the designation of "halo" (e.g. as illustrated in the term haloalkyl) refers to all degrees of substitutions from a single substitution to a perhalo substitution (e.g. as illustrated with methyl as chloromethyl (-CH 2 C1), dichloromethyl (-CHC1 2 ), trichloromethyl (-CC1 3 )).
- the compounds within the parasiticidal compositions of the inventive methods may exist as hydrates or solvates, in which a certain stoichiometric amount of water or a solvent is associated with the molecule in the crystalline form.
- the parasiticidal compositions may include hydrates and solvates of the active agents.
- the parasiticidal compositions of the inventive methods may include up to 15% (w/w), up to 20% (w/w), or up to 30%) (w/w) of a particular solid form.
- the term "acid salt” contemplates salts of the compounds with all pharmaceutically acceptable inorganic or organic acids.
- Inorganic acids include mineral acids such as hydrohalic acids such as hydrobromic acid and hydrochloric acid, sulfuric acid, phosphoric acids and nitric acid.
- Organic acids include all pharmaceutically acceptable aliphatic, alicyclic and aromatic carboxylic acids, dicarboxylic acids, tricarboxylic acids and fatty acids.
- the acids are straight chain or branched, saturated or unsaturated C1-C20 aliphatic carboxylic acids, which are optionally substituted by halogen or by hydroxyl groups, or C 6 -Ci2 aromatic carboxylic acids.
- acids are carbonic acid, formic acid, acetic acid, propionic acid, isopropionic acid, valeric acid, a-hydroxy acids such as glycolic acid and lactic acid, chloroacetic acid, benzoic acid, pamoic acid, methane sulfonic acid, and salicylic acid.
- dicarboxylic acids include oxalic acid, malic acid, succinic acid, tartaric acid, fumaric acid, and maleic acid.
- An example of a tricarboxylic acid is citric acid.
- Fatty acids include all pharmaceutically acceptable saturated or unsaturated aliphatic or aromatic carboxylic acids having 4 to 24 carbon atoms.
- Examples include butyric acid, isobutyric acid, sec-butyric acid, lauric acid, palmitic acid, stearic acid, oleic acid, linoleic acid, linolenic acid, and phenylsteric acid.
- Other acids include gluconic acid, glycoheptonic acid and lactobionic acid.
- base salt contemplates salts of the compounds with all pharmaceutically acceptable inorganic or organic bases, including hydroxides, carbonates or bicarbonates of alkali metal or alkaline earth metals. Salts formed with such bases include, for example, the alkali metal and alkaline earth metal salts, including, but not limited to, as the lithium, sodium, potassium, magnesium or calcium salts. Salts formed with organic bases include the common hydrocarbon and heterocyclic amine salts, which include, for example, ammonium salts ( H4 + ), alkyl- and dialkylammonium salts, and salts of cyclic amines such as the morpholine and piperidine salts.
- the parasiticidal compositions used in the inventive methods comprises a parasiticidal effective amount of at least one isoxazoline compound of formula (S)-l or a veterianarily acceptable salt thereof in combination with an inert carrier or formulation diluent and optionally other non-active excipients, wherein the composition contains less than about 10% by weight of an (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof.
- the parasiticidal compositions may be in a variety of solid and liquid forms which are suitable for application on or administration to an animal.
- the parasiticidal compositions employed in the inventive methods may be in formulations suitable for oral administration, injectable administration, including subcutaneous and parenteral administration, and topical administration (e.g. spot-on or pour-on), dermal or subdermal administration.
- the parasiticidal compositions used in the invention may be in a form suitable for oral use (see, e.g., U.S. Patent No. 4,564,631, which is hereby incorporated by reference in its entirety), dietary supplements, troches, lozenges, chewables, tablets, hard or soft capsules, bolus, emulsions, aqueous or oily suspensions, aqueous or oily solutions, oral drench formulations, dispersible powders or granules, premixes, syrups or elixirs, enteric formulations or pastes.
- compositions intended for oral use may be prepared according to any method known in the art for the manufacture of pharmaceutical compositions and such compositions may contain one or more sweetening agents, bittering agents, flavoring agents, coloring agents and preserving agents in order to provide pharmaceutically elegant and palatable preparations.
- Tablets may contain the active ingredient in admixture with non-toxic, pharmaceutically acceptable excipients which are suitable for the manufacture of tablets.
- excipients may be, for example, inert diluents, such as calcium carbonate, sodium carbonate, lactose, calcium phosphate or sodium phosphate; granulating and disintegrating agents, for example, corn starch, or alginic acid; binding agents, for example starch, gelatin or acacia, and lubricating agents, for example, magnesium stearate, stearic acid or talc.
- the tablets may be uncoated or they may be coated by known techniques to delay disintegration and absorption in the gastrointestinal tract and thereby provide a sustained action over a longer period.
- a time delay material such as glyceryl monostearate or glyceryl distearate may be employed. They may also be coated by the technique described in U.S. Patent Nos. 4,256, 108; 4,166,452; and 4,265,874 (all incorporated herein by reference in their entirety) to form osmotic therapeutic tablets for controlled release.
- Oral formulations include hard gelatin capsules, wherein the active ingredient is mixed with an inert solid diluent, for example, calcium carbonate, calcium phosphate or kaolin. Capsules may also be soft gelatin capsules, wherein the active ingredient is mixed with water or miscible solvents such as propylene glycol, polyethylene glycols (PEGs) and ethanol, or an oil medium, for example peanut oil, liquid paraffin, or olive oil.
- an inert solid diluent for example, calcium carbonate, calcium phosphate or kaolin.
- Capsules may also be soft gelatin capsules, wherein the active ingredient is mixed with water or miscible solvents such as propylene glycol, polyethylene glycols (PEGs) and ethanol, or an oil medium, for example peanut oil, liquid paraffin, or olive oil.
- PEGs polyethylene glycols
- oil medium for example peanut oil, liquid paraffin, or olive oil.
- the parasiticidal compositions may be chewable tablet compositions or soft chewable compositions ("soft chew") such as those described in US 9,233,100 B2, US 9,259,417, US 2010/0087492, US 2006/0222684, US 2004/0151759, US 7955632, US 2015/0057321, US 2015/0057239 and WO 2016/073347, all incorporated herein by reference in their entirety.
- soft chew such as those described in US 9,233,100 B2, US 9,259,417, US 2010/0087492, US 2006/0222684, US 2004/0151759, US 7955632, US 2015/0057321, US 2015/0057239 and WO 2016/073347, all incorporated herein by reference in their entirety.
- a soft chew is palatable and acceptable to the animal.
- the soft chews may include one or more of the following components: a solvent or mixture of solvents, one or more fillers, one or more binders, one or more surfactants, one or more humectants, one or more lubricants, one or more disintegrants, one or more colorants, one or more antimicrobial agents, one or more antioxidants, one or more pH modifiers and one or more flavoring agents.
- Solvents that may be used include, but are not limited to, various grades of liquid polyethylene glycol (PEG) including PEG 200, PEG 300, PEG 400 and PEG 540; propylene carbonate; propylene glycol; triglycerides including, but not limited to caprylic/capric triglyceride, caprylic/capric/linoleic triglyceride (e.g.
- MIGLYOL® 810 and 812 caprylic/capric/succinic triglyceride, propylene glycol dicaprylate/dicaprate, and the like; water, sorbitol solution, glycerol caprylate/caprate and polyglycolized glycerides (GELUCIRE ®), or a combination thereof.
- Fillers include, but are not limited to, corn starch, pre-gelatinized corn starch, soy protein fines, corn cob, and corn gluten meal, and the like. In some embodiments, a combination of two or more fillers may be used in the compositions.
- Binders that may be used in soft chewable compositions include, but are not limited to, polyvinylpyrrolidone (e.g. Povidone), cross-linked polyvinylpyrrolidone (Crospovidone), PEGs of various grades including PEG 3350, PEG 4000, PEG 6000, PEG 8000 and even PEG 20,000, and the like; co-polymers of vinylpyrrolidone and vinyl acetate (e.g.
- Copovidone such as the product sold by BASF by the tradename Kollidon® VA 64 and the like; starch such as potato starch, tapioca starch or corn starch; molasses, corn syrup, honey, maple syrup and sugars of various types; or a combination of two or more binders.
- Humectants that may be used in soft chewable compositions include, but are not limited to, glycerol (also referred to herein as glycerin), propylene glycol, cetyl alcohol and glycerol monostearate, and the like. Polyethylene glycols of various grades may also be used as humectants.
- Surfactants may be present in soft chewable composition to improve their solubility and absorption after ingestion. Surfactants are typically present in a concentration of about 1 to 10% (w/w), more typically about 1 to about 5% (w/w).
- surfactants examples include, but are not limited to, glyceryl monooleate, polyoxyethylene sorbitan fatty acid esters, sorbitan esters including sorbitan monooleate (Span® 20), polyvinyl alcohol, polysorbates including polysorbate 20 and polysorbate 80, ⁇ i-a-tocopheiyl polyethylene glycol 1000 succinate (TPGS), sodium lauryl sulfate, co-polymers of ethylene oxide and propylene oxide (e.g.
- poloxamers such as LUTROL® F87 and the like
- polyethylene glycol castor oil derivatives including polyoxyl 35 castor oil (Cremophor® EL), polyoxyl 40 hydrogenated castor oil (Cremophor® RH 40), polyoxyl 60 hydrogenated castor oil (Cremophor® RH60); propylene glycol monolaurate (LAUROGLYCOL®); glyceride esters including glycerol caprylate/caprate (CAPMUL® MCM), polyglycolized glycerides (GELUCIRE®), PEG 300 caprylic/capric glycerides (Softigen® 767), PEG 400 caprylic/capric glycerides (Labrasol®), PEG 300 oleic glycerides (Labrafil® M-1944CS), PEG 300 linoleic glycerides (Labrafil® M-2125CS); polyethylene glycol stearates and polyethylene glycol hydroxy stearates
- Polyethylene glycol stearates are mixtures of mono- and distearate esters of mixed polyoxyethylene polymers.
- Polyethylene glycol hydroxystearate is a mixture of mono- and diesters of hydroxystearic acid with polyethylene glycols.
- One polyethylene glycol hydroxystearate that may be used in the compositions is polyethylene glycol 12-hydroxy stearate.
- the compositions may include the surfactant polyethylene glycol 15 12-hydroxy stearate (Solutol® HS 15 from BASF), a mixture of mono- and diesters of 12-hydroxy stearic acid with 15 moles of ethylene oxide. Again, these compounds, as well as their amounts are well known in the art.
- the compositions may include polyoxyl 35 castor oil (Cremophor® EL) as a surfactant.
- the chewable compositions may include polyoxyl 40 hydrogenated castor oil (Cremophor® RH 40) or polyoxyl 60 hydrogenated castor oil (Cremophor® RH60) as surfactants.
- compositions of the invention may also include a combination of surfactants.
- Oral fomulations may contain other inert ingredients such as antioxidants, preservatives, or pH stabilizers. These compounds are well known in the formulation art.
- Antioxidants may be added to the compositions of the invention to inhibit degradation of the active agents. Suitable antioxidants include, but are not limited to, alpha tocopherol, ascorbic acid, ascrobyl palmitate, fumaric acid, malic acid, sodium ascorbate, sodium metabi sulfate, n-propyl gallate, butylated hydroxy anisole (BHA), BHT (butylated hydroxy toluene) monothioglycerol and the like.
- Oral formulations may also include one or more lubricants and/or processing aids.
- the lubricant/processing aid may also behave as a solvent, and accordingly, there some of the components of the parasiticidal compositions may have dual functions.
- Lubricants/processing aids include, but are not limited to polyethylene glycols of various molecular weight ranges including PEG 3350 (Dow Chemical) and PEG 4000, corn oil, mineral oil, hydrogenated vegetable oils (STEROTEX or LUBRITAB), peanut oil and/or castor oil.
- flavoring agents may be used in oral formulations to improve the palatability of the oral veterinary formulations.
- Preferred flavoring agents are those that are not derived from animal sources.
- flavoring components derived from fruit, meat including, but not limited to pork, beef, chicken, fish, poultry, and the like), vegetable, cheese, bacon, cheese-bacon and/or artificial flavorings may be used.
- a flavoring component is typically chosen based upon consideration related to the organism that will be ingesting the soft chew. For example, a horse may prefer an apple flavoring component, while a dog may prefer a meat flavoring component.
- flavoring components derived from non-animal sources are preferred, in some embodiments, natural flavors containing beef or liver extracts, etc., may be used such as braised beef flavor artificial powdered beef flavor, roast beef flavor and corned beef flavor among others.
- the parasiticidal composition may be administered via a drench, and is administered orally.
- Drench formulations are those in which the liquid-containing compositions of the invention are administered to the mouth or throat of the animal.
- the parasiticidal compositions used in the inventive methods may also be in the form of oil-in-water or water-in-oil emulsions.
- the oily phase maybe a vegetable oil, for example, olive oil or arachis oil, or a mineral oil, for example, liquid paraffin or mixtures of these.
- Suitable emulsifying agents include naturally-occurring phosphatides, for example, soy bean, lecithin, and esters or partial esters derived from fatty acids and hexitol anhydrides, for example, sorbitan monoleate, and condensation products of the said partial esters with ethylene oxide, for example, polyoxyethylene sorbitan monooleate.
- the emulsions may also contain sweetening agents, bittering agents, flavoring agents, and/or preservatives.
- the oral compositions may also be in the form of a microemulsion.
- Microemulsions are well suited as the liquid carrier vehicle. Microemulsions are quaternary systems comprising an aqueous phase, an oily phase, a surfactant and a co-surfactant. They are translucent and isotropic liquids.
- Microemulsions are composed of stable dispersions of microdroplets of the aqueous phase in the oily phase or conversely of microdroplets of the oily phase in the aqueous phase.
- the size of these microdroplets may be less than 200 nm (1000 to 100,000 nm for emulsions).
- the interfacial film may be composed of an alternation of surface-active (SA) and co-surface- active (Co-SA) molecules which, by lowering the interfacial tension, allows the microemulsion to be formed spontaneously.
- the oily phase may be formed from mineral or vegetable oils, from unsaturated polyglycosylated glycerides or from triglycerides, or alternatively from mixtures of such compounds.
- the oily phase may be comprised of triglycerides.
- the triglycerides may be medium-chain triglycerides, such as, for example C 8 -Ci 0 caprylic/capric triglyceride.
- the oily phase may represent a % v/v range of, for example, about 2 to about 15%; about 7 to about 10%; and about 8 to about 9% v/v of the microemulsion.
- the aqueous phase may include, for example water or glycol derivatives, such as propylene glycol, glycol ethers, polyethylene glycols or glycerol.
- the glycol may be propylene glycol, diethylene glycol monoethyl ether, dipropylene glycol monoethyl ether or mixtures thereof.
- the aqueous phase will represent a proportion from about 1 to about 4% v/v in the microemulsion.
- Surfactants for the microemulsion may include diethylene glycol monoethyl ether, dipropyelene glycol monomethyl ether, polyglycolyzed C 8 -Cio glycerides or polyglyceryl-6 dioleate.
- the co-surfactants may include short-chain alcohols, such as ethanol and propanol.
- the co-surfactant to surfactant ratio could be, for example, from about 1/7 to about 1/2.
- the amount of co-surfactant there will be from about 25 to about 75% v/v of surfactant and from about 10 to about 55% v/v of co-surfactant in the microemulsion.
- Oily suspensions may be formulated by suspending the active ingredient in a vegetable oil, for example, atachis oil, olive oil, sesame oil or coconut oil, or in mineral oil such as liquid paraffin.
- the oily suspensions may contain a thickening agent, for example, beeswax, hard paraffin or cetyl alcohol.
- Sweetening agents such as sucrose, saccharin or aspartame, bittering agents, and flavoring agents may be added to provide a palatable oral preparation.
- These compositions may be preserved by the addition of an anti-oxidant such as ascorbic acid, or other known preservatives.
- Aqueous suspensions may contain the active material in admixture with excipients suitable for the manufacture of aqueous suspensions.
- excipients include suspending agents, for example, sodium carboxymethylcellulose, methylcellulose, hydroxy-propylmethylcellulose, sodium alginate, polvinylpyrrolidone, gum tragacanth and gum acacia; dispersing or wetting agents include naturally-occurring phosphatide, for example, lecithin, or condensation products of an alkylene oxide with fatty acids, for example polyoxyethylene stearate, or condensation products of ethylene oxide with long chain aliphatic alcohols, for example, heptadecaethyleneoxycetanol, or condensation products of ethylene oxide with partial esters derived from fatty acids and a hexitol such as polyoxyethylene sorbitol monooleate, or condensation products of ethylene oxide, with partial esters derived from fatty acids and hexitol anhydrides, for example polyethylene sorbitan
- the aqueous suspensions may also contain one or more preservatives, for example ethyl, or n-propyl, p-hydroxybenzoate, one or more coloring agents, one or more flavoring agents, and one or more sweetening agents and/or bittering agents, such as those set forth above.
- preservatives for example ethyl, or n-propyl, p-hydroxybenzoate, one or more coloring agents, one or more flavoring agents, and one or more sweetening agents and/or bittering agents, such as those set forth above.
- Dispersible powders and granules suitable for preparation of an aqueous suspension by the addition of water may provide the active ingredient in admixture with a dispersing or wetting agent, suspending agent and one or more preservatives.
- a dispersing or wetting agent e.g., talc, kaolin, kaolin, kaolin, kaolin, kaolin, kaolin, kaolin, kaolin, kaolin, kaolin, kaolin, kaolin, kaolin, kaolin, kaolin, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, sorbitol, mannitol, mannitol, mannitol, mannitol, mannitol, mannitol, mannitol, mannitol
- Syrups and elixirs may be formulated with sweetening agents, for example, glycerol, propylene glycol, sorbitol or sucrose. Such formulations may also contain a demulcent, a preservative, flavoring agent(s) and/or coloring agent(s).
- sweetening agents for example, glycerol, propylene glycol, sorbitol or sucrose.
- Such formulations may also contain a demulcent, a preservative, flavoring agent(s) and/or coloring agent(s).
- the oral formulation may also be in paste form.
- paste form examples include, but are not limited to, those described in U.S. Patent Nos. 6,787,342 and 7,001,889 (each of which are incorporated herein by reference).
- the paste may further contain fumed silica; a viscosity modifier; a carrier (e.g., a hydrophilic carrier which is triacetin, a monoglyceride, a diglyceride, or a triglyceride); optionally, an absorbent; and optionally, a colorant, stabilizer, surfactant, or preservative.
- the paste may also include a viscosity modifier.
- Suitable viscosity modifiers include, but are not limited to, PEGs including, but not limited to, PEG 200, PEG 300, PEG 400, PEG 600; monoethanolamine, triethanolamine, glycerol, propylene glycol, polyoxyethylene (20) sorbitan mono-oleate (polysorbate 80 or Tween 80), or poloxamers (e.g., Pluronic L 81); an absorbent such as magnesium carbonate, calcium carbonate, starch, and cellulose and its derivatives; and a colorant including, but not limited to, titanium dioxide iron oxide, or FD&C Blue #1 Aluminum Lake.
- Topical, dermal and subdermal formulations may include, by way of non-limiting example, emulsions, creams, ointments, gels, pastes, powders, shampoos, pour-on formulations, ready-to-use formulations, spot-on solutions and suspensions, dips and sprays. Spot-on, spray-on or pour-on formulation allow the bioactive agent or agents to be absorbed through the skin to achieve systemic levels, distributed through the sebaceous glands or on the surface of the skin achieving levels throughout the coat.
- pour-on formulations are based on alcoholic solvents including isopropyl alcohol or ethyl alcohol, optionally in combination with other solvents.
- Organic solvents that can be used in the parasiticidal compositions include acetyltributyl citrate, fatty acid esters such as the dimethyl ester, diisobutyl adipate, acetone, acetonitrile, benzyl alcohol, ethyl alcohol, butyl diglycol, dimethylacetamide, dimethylformamide, dimethyl sulfoxide, dimethyl isosorbide, dipropylene glycol n-butyl ether, ethanol, isopropanol, methanol, ethylene glycol monoethyl ether, ethylene glycol monomethyl ether, monomethylacetamide, dipropylene glycol monomethyl ether, liquid polyoxyethylene glycols, propylene glycol, 2-pyrrolidone, N-methylpyrrolidone, diethylene glycol monoethyl ether, ethylene glycol, triacetin, Ci-Ci 0 esters of carboxylic acids such as butyl or
- the solvent will be used in proportion with the concentration of the active agent compound and its solubility in this solvent. It will be sought to have the lowest possible volume. The vehicle makes up the difference to 100%.
- a vehicle or diluent for the formulations may include dimethyl sulfoxide (DMSO), glycol derivatives such as, for example, propylene glycol, glycol ethers, polyethylene glycols or glycerol.
- DMSO dimethyl sulfoxide
- glycol derivatives such as, for example, propylene glycol, glycol ethers, polyethylene glycols or glycerol.
- plant oils such as, but not limited to soybean oil, groundnut oil, castor oil, corn oil, cotton oil, olive oil, rape seed oil, sunflower oil, etc.
- mineral oils such as, but not limited to, petrolatum, paraffin, silicone, etc.
- An emollient and/or spreading and/or film-forming agent may be added.
- Examples of a emollient and/or spreading and/or film-forming agent may be:
- polyvinylpyrrolidone polyvinyl alcohols, copolymers of vinyl acetate and vinylpyrrolidone, polyethylene glycols, benzyl alcohol, mannitol, glycerol, sorbitol, polyoxyethylenated sorbitan esters; lecithin, sodium carboxymethylcellulose, silicone oils, polydiorganosiloxane oils (such as polydimethylsiloxane (PDMS) oils), for example those containing silanol functionalities, or a 45 V2 oil,
- PDMS polydimethylsiloxane
- anionic surfactants such as alkaline stearates, sodium, potassium or ammonium stearates; calcium stearate, triethanolamine stearate; sodium abietate; alkyl sulphates (e.g. sodium lauryl sulphate and sodium cetyl sulphate); sodium dodecylbenzenesulphonate, sodium dioctylsulphosuccinate; fatty acids (e.g. those derived from coconut oil),
- cationic surfactants include water-soluble quaternary ammonium salts of formula N R'R"R"'R" ", Y " in which the radicals R are optionally hydroxylated hydrocarbon radicals and Y " is an anion of a strong acid such as the halide, sulphate and sulphonate anions; cetyltrimethylammonium bromide is among the cationic surfactants which can be used,
- nonionic surfactants such as sorbitan esters, which are optionally polyoxyethylenated (e.g. polysorbate 80), polyoxyethylenated alkyl ethers; polyoxypropylated fatty alcohols such as polyoxypropylene-styrol ether; polyethylene glycol stearate, polyoxyethylenated derivatives of castor oil, polyglycerol esters, polyoxyethylenated fatty alcohols, polyoxyethylenated fatty acids, copolymers of ethylene oxide and propylene oxide, (f) amphoteric surfactants such as the substituted lauryl compounds of betaine; or
- the amount of emollient that may be used may be in a proportion of from about 0.1 to 50% or 0.25 to 5%, by volume.
- the emollient used may be in a proportion of from about 0.1% to about 30%, about 1% to about 30%, about 1% to about 20%, or about 5%) to about 20% by volume.
- the parasiticidal composition may be in the form of a ready-to-use spot-on solution form as is described in U. S. Patent Nos. 6,395,765 and 9, 180, 121, both incorporated herein by reference.
- the ready-to-use solution may contain a crystallization inhibitor and an organic solvent or a mixture of organic solvents.
- water may be included with the organic solvent.
- the topical formulations may include a crystallization inhibitor in an amount of about 1 to about 50% (w/v) or about 5 to about 40% (w/v) based on the total weight of the formulation.
- the amount of crystallization inhibitor in the inventive formulations may be about 1% to about 30%, about 5% to about 20%, about 1% to about 15%, or about 1% to about 10%) (w/w).
- the type of crystallization inhibitor used in the topical formulations is not limited as long as it functions to inhibit crystallization of the active or inactive agents from the formulation.
- a solvent or co-solvent of the formulation may also function as a crystallization inhibitor if it sufficiently inhibits the formation of crystals from forming over time when the formulation is administered.
- Crystallization inhibitors which are useful include, but are not limited to:
- polyvinylpyrrolidone polyvinyl alcohols, copolymers of vinyl acetate and vinylpyrrolidone, polyethylene glycols, benzyl alcohol, dimethylformamide, dimethylacetamide, dimethylsulfoxide, 2-pyrrolidone, N-methylpyrrolidone, mannitol, glycerol, sorbitol or polyoxyethylenated esters of sorbitan; lecithin or sodium carboxymethylcellulose; or acrylic derivatives, such as acrylates or methacrylates or polymers or copolymers thereof, PEG or polymers containing polyethyleneglycols, such as glycofurol and the like, and others;
- anionic surfactants such as alkaline stearates (e.g. sodium, potassium or ammonium stearate); calcium stearate or triethanolamine stearate; sodium abietate; alkyl sulphates, which include but are not limited to sodium lauryl sulphate and sodium cetyl sulphate; sodium dodecylbenzenesulphonate or sodium dioctyl sulphosuccinate; or fatty acids (e.g. coconut oil);
- alkaline stearates e.g. sodium, potassium or ammonium stearate
- calcium stearate or triethanolamine stearate e.g. calcium stearate or triethanolamine stearate
- sodium abietate e.g. sodium stearate
- alkyl sulphates which include but are not limited to sodium lauryl sulphate and sodium cetyl sulphate; sodium dodecylbenzenesulphonate or sodium dioc
- cationic surfactants such as water-soluble quaternary ammonium salts of formula N R'R"R'"R" "Y ⁇ , in which the R radicals are identical or different optionally hydroxylated hydrocarbon radicals and Y is an anion of a strong acid, such as halide, sulphate and sulphonate anions; cetyltrimethylammonium bromide is one of the cationic surfactants which can be used;
- non-ionic surfactants such as optionally polyoxyethylenated esters of sorbitan, e.g. Polysorbate 80, or polyoxyethylenated alkyl ethers; polyethylene glycol stearate, polyoxyethylenated derivatives of castor oil, polyglycerol esters, polyoxyethylenated fatty alcohols, polyoxyethylenated fatty acids or copolymers of ethylene oxide and of propylene oxide;
- non-ionic surfactants such as optionally polyoxyethylenated esters of sorbitan, e.g. Polysorbate 80, or polyoxyethylenated alkyl ethers
- polyethylene glycol stearate polyoxyethylenated derivatives of castor oil, polyglycerol esters, polyoxyethylenated fatty alcohols, polyoxyethylenated fatty acids or copolymers of ethylene oxide and of propylene oxide
- amphoteric surfactants such as substituted lauryl compounds of betaine
- a crystallization inhibitor pair maybe used.
- Such pairs include, for example, the combination of a film-forming agent of polymeric type and of a surface-active agent. These agents will be selected from the compounds mentioned above as crystallization inhibitor.
- the organic solvent(s) may have a dielectric constant of between about 10 and about 35 or between about 20 and about 30. In other topical formulations, the organic solvent may have a dielectric constant of between about 10 and about 40 or between about 20 and about 30.
- the content of this organic solvent or mixture of solvents in the overall composition is not limited and will be present in an amount sufficient to dissolve the desired components to a desired concentration. As discussed above, the organic solvent may also function as a crystallization inhibitor in the formulation.
- one or more of the organic solvent(s) may have a boiling point of below about 100° C, or below about 80° C. In other topical formulations, the organic solvent(s) may have a boiling point of below about 300° C, below about 250° C, below about 230°C, below about 210° C or below about 200° C.
- the topical formulations may contain a mixture of solvents, i.e. a solvent and a co-solvent, the solvents may be present in the composition in a weight/weight (WAV) ratio of about 1/50 to about 1/1.
- WAV weight/weight
- the solvents will be in a ratio of about 1/30 to about 1/1, about 1/20 to about 1/1, or about 1/15 to about 1/1 by weight.
- the two solvents will be present in a weight/weight ratio of about 1/15 to about 1/2.
- at least one of the solvents present may act as to improve solubility of the active agent or as a drying promoter.
- at least one of the solvents will be miscible with water.
- the formulation may also comprise an antioxidizing agent intended to inhibit oxidation in air, this agent may be present in a proportion of about 0.005 to about 1% (w/v), about 0.01 to about 0.1%, or about 0.01 to about 0.05%.
- film-forming agent examples include agents are of the polymeric type which include but are not limited to the various grades of polyvinylpyrrolidone, polyvinyl alcohols, and copolymers of vinyl acetate and of vinylpyrrolidone.
- Surface-active agents may be included. These agents include but are not limited to those made of non-ionic surfactants; in another embodiment of the surface active agents, the agent is a polyoxyethylenated esters of sorbitan and in yet another embodiment of the surface-active agent, the agents include the various grades of polysorbate, for example Polysorbate 80.
- the film-forming agent and the surface-active agent may be incorporated in similar or identical amounts within the limit of the total amounts of crystallization inhibitor mentioned elsewhere.
- topical compositions include the compound crotamiton which may inhibit the crystallization of active agents from solution.
- the crystallization inhibitor inhibits the formation of crystals on the coat, and improves the maintenance of the cosmetic appearance of the skin or fur; that is to say without a tendency towards sticking or towards a sticky appearance, despite the high concentration of active material.
- Substances other than those mentioned herein may be used as crystallization inhibitors in the present invention.
- the effectiveness of the crystallization inhibitor may be demonstrated by a test according to which 0.3 mL of a solution comprising 10% (w/v) of the active agent in an appropriate solvent as defined above, and 10% (w/v) of the compound acting as a crystallization inhibitor are placed on a glass slide at 20° C for 24 hours, after which fewer than 10 crystals, preferably 0 crystals, are seen with the naked eye on the glass slide.
- Antioxidizing agents may be included. These agents are those conventional in the art and include but are not limited to BHA, butylated hydroxy toluene (BHT), ascorbic acid, sodium metabi sulphite, propyl gallate, sodium thiosulphate or a mixture of at least two compounds with antioxidant properties.
- BHA butylated hydroxy toluene
- ascorbic acid sodium metabi sulphite
- propyl gallate sodium thiosulphate
- sodium thiosulphate sodium thiosulphate
- composition adjuvants discussed above are well known to the practitioner in this art and may be obtained commercially or through known techniques. These concentrated compositions are generally prepared by simple mixing of the constituents as defined above; advantageously, the starting point is to mix the active material in the main solvent and then the other ingredients or adjuvants are added.
- the volume of the formulation applied will depend on the type of animal and the size of the animal as well as the strength of the formulation and the potency of the active agents. In one embodiment, an amount of about 0.3 to about 20 ml of the formulation may be applied to the animal. In other embodiment for the volume, the volume may be about 0.1 to about 10 ml, about 0.3 to about 5 ml, about 0.5 ml to about 10 ml, or about 0.3 to about 3 ml.
- application of a spot-on formulation according to the present invention may also provide long-lasting and broad-spectrum efficacy when the solution is applied to the mammal or bird.
- the spot-on formulations provide for topical administration of a concentrated solution, suspension, microemulsion or emulsion for intermittent application to a spot on the animal, generally between the two shoulders (solution of spot-on type).
- the carrier may be a liquid carrier vehicle as described in U.S. Patent Nos. 6,426,333; 6,395,765 (incorporated herein by reference), which in may comprise a solvent or mixture of solvents including, but not limited to, acetone, an aliphatic alcohol such as methanol, ethanol, propanol, butanol, isopropanol, pentanol, hexanol, heptanol, octanol, nonanol, cyclopentanol, cyclohexanol, ethylene glycol, propylene glycol and the like; an aromatic alcohol such as phenol, cresol, naphthol, benzyl alcohol and the like; acetonitrile, butyl diglycol, an organic amide such as dimethylacetamide, dimethylformamide, monomethylacetamide, 2- pyrrolidone, N-methylpyrrolidone, vinylpyrrolidone and the like; propylene
- Solvents which may be used for the spot-on or pour-on compositions of the invention include those described in US 9, 180,121 (incorporated by reference). These solvents include, but are not limited to, carboxylic acid esters, diesters of dicarboxylic acids, fatty acid esters or diesters of fatty diacids, or a combination thereof, including, but not limited to, isopropyl palmitate, isostearyl lactate, diisopropyl adipate, dibutyl adipate, diethyl sebacate, dibutyl sebacate, octyl palmitate, polyethyleneglycol (PEG) stearate and cetearyl octanoate; oils including, but not limited to, mineral oil, diglycerides, triglycerides, jojoba oil, lecithin and castor oil, or a combination thereof; long chain aliphatic alcohols such as isostearyl alcohol and the like; fatty alcohols and their esters,
- Excipients that may promote the containment of the active agent in the skin for longer periods of time and may be included in the compositions of the invention include, but are not limited to, mixed esters of sucrose and carboxylic acids including sucrose acetate isobutyrate (SAIB) and the like; low temperature melting waxes, hydrogenated vegetable oils, caprylic/capric glycerides; glycerol esters, including for example, triacetin, glycerol monooleate, glycerol monolinoleate, glycerol stearate, glyceryl distearate and the like; triglycerides, including for example, caprylic, capric/ myristic/ stearic triglyceride; thermoreversible polymers, such as Pluronic and poloxamers, including for example, Lutrol F127 by itself or in mixture with other poloxamers; or a combination thereof.
- SAIB sucrose acetate isobutyrate
- the pharmaceutically acceptable carrier for the topical compositions comprise a mixture of a diester of a dicarboxylic acid alone or in combination with one or more of additional solvents listed above, and/or an "oily" lipophilic substance, including a liquid or low melting lipophilic active agent such as ( ⁇ -methoprene, pyriproxyfen and/or permethrin; and/or a mixed ester of sucrose and carboxylic acids including a mixed ester of sucrose and acetic and isobutyric acids such as sucrose acetate isobutyrate (SAIB), and/or low melting waxes and/or hard fats.
- SAIB sucrose acetate isobutyrate
- Diethyl sebacate or diisopropyl adipate are examples for a diester of a dicarboxylic acid.
- the blends of solvents comprising a dicarboxylic acid ester comprises a glycol or polyglycol, or a glycol or polyglycol ether or ester including, but not limited to, ethylene glycol (EG), propylene glycol (PG), liquid PEGs of various grades including PEG 400, EG or PG monocaprylate, EG or PG caprylate, EG or PG monolaurate, EG or PG dicaprylate/dicaprate, diethyleneglycol monoethyl ether (DGME, Transcutol ® ), butyl diglycol, dipropylene glycol n- butyl ether, ethyleneglycol monoethyl ether, ethyleneglycol monomethyl ether, dipropylene glycol monomethyl ether, propylene glycol monomethyl ether, propylene glycol mono
- the carrier for the topical compositions may comprise a dialkyl ester of a dicarboxylic acid, such as diethyl sebacate, diisopropyl sebacate, diisopropyl adipate, dibutyl adipate, or a combination thereof, alone or in combination with solvents selected from:
- PG ester including PG monocaprylate, PG caprylate, PG monolaurate, PG dicaprylate / dicaprate, or a combination thereof;
- an ether solvent including dimethyl isosorbide, diethylene glycol monoethyl ether (also known as DGME or Transcutol ® ), or a combination thereof;
- carboxylic acid ester including, but not limited to, triacetin, lauryl lactate, isopropyl palmitate, diisopropyl sebacate, or a combination thereof; and other "oily" or lipophilic organic solvents including glycerol formal and the like.
- the amount the additional solvents combined with the carboxylic acid ester or diester of a dicarboxylic acid are present in an amount of at least about 1% (v/v), at least about 5% (v/v), at least about 9.0% (v/v), at least about 13% (v/v), at least about 17% (v/v) or at least about 20% (v/v).
- the additional solvents will be in an amount of at least about 9% (v/v).
- the additional solvents will be present in an amount of about 5-70% (v/v), about 10-60% (v/v), about 10-50% (v/v), about 15-60% (v/v) or about 15-50% (v/v).
- the additional solvents will be present in an amount of about 20-70% (v/v), about 20-60% (v/v), about 20-50% (v/v) or about 25-50% (v/v).
- the liquid carrier vehicle may optionally contain a crystallization inhibitor including, but not limited to, those described in (a) to (h) above, or a compound that may act both as a solvent and a crystallization inhibitor (as defined above), or a mixture of these crystallization inhibitors.
- Spot-on formulations may be prepared by dissolving the active ingredients into the pharmaceutically or veterinary acceptable vehicle. Alternatively, the spot-on formulation may be prepared by encapsulation of the active ingredient to leave a residue of the therapeutic agent on the surface of the animal. These formulations will vary with regard to the weight of the therapeutic agent in the combination depending on the species of host animal to be treated, the severity and type of infection and the body weight of the host.
- Dosage forms may typically contain from about 0.1 mg to about 5 g. In other embodiments, the dosage form may contain about 0.5 mg to about 5 g of an active agent. In one embodiment of the dosage form, the dosage may contain from about 1 mg to about 500 mg of an active agent, typically about 25 mg, about 50 mg, about 100 mg, about 200 mg, about 300 mg, about 400 mg, about 500 mg, about 600 mg, about 800 mg, or about 1000 mg.
- the bioactive agent may be present in the formulation at a concentration of about 0.05 to about 10% weight/volume.
- the bioactive agent may be present in the formulation as a concentration from about 0.1 to about 2% weight/volume or the bioactive agent may be present in the formulation as a concentration from about 0.25 to about 1.5% weight/volume.
- the bioactive agent may be present in the formulation as a concentration about 1% weight/volume.
- the parasiticidal formulations used in the inventive methods may include long-acting injectable formulations. These formulations comprise at least one bioactive agent (e.g., an isoxazoline compound) and optionally one or more bioactive agent(s), at least one pharmaceutically acceptable non-biodegradable polymer and a co-solvent.
- Pharmaceutically acceptable non-biodegradable polymers include, but are not limited to, PEGs and poloxamers.
- Long-acting injectable formulations are active for an extended period of time; e.g., a period from at least two (2) (e.g., three (3), four (4), five (5)) to six (6) months.
- a particularly advantageous long acting injectable formulation is used.
- This formulation comprises an isoxazoline bioactive agent, at least one liquid PEG and a co-solvent and excludes other bioactive polymers, such as those identified above.
- the formulations have the advantage of exhibiting favorable properties with respect to the injection site, while being effective for an extended period of time; they are described in U.S. Provisional Patent Application No. 62/299,333, herein incorporated by reference.
- Liquid PEGs as provided in these formulations are those polyethylene glycols that are liquid at room temperature (20 - 30 °C). Polyethylene glycols have the following structural formula:
- Non-limiting examples for n in the above formula are those compounds when n is from 1 to about 10,000 (e.g., from about 4 to about 25).
- Liquid PEGs include combinations of different polyethylene glycols.
- Non-limiting examples of liquid PEGs include PEG 200, PEG 300, PEG 400, PEG 600, and PEG 1000 or combinations thereof.
- the invention provides long acting injectable compositions that comprise an isoxazoline active agent in a carrier that comprises a poloxamer and optionally a co-solvent.
- Poloxamers are a family of synthetic block copolymers of ethylene oxide and propylene oxide. Poloxamers may be liquid, a milky white paste or a powder and are represented by the following structure:
- Poloxamer are available from commercial sources such as BASF and Croda.
- An example of a poloxamer is P-124 which is a solid at room temperature.
- the amount of poloxamer may be, for example, from about 0.5% to about 20 (w/w); from about 1% to about 20% (w/w); from about 1%) to about 10% (w/w); or from about 1 to about 5% (w/w).
- the amount of poloxamer, when present is from about 1 % to about 3 % (w/w).
- the co-solvents used in the long acting injectable compositions may be a single or a blend of co-solvents.
- the co-solvents used in the long-acting injectable formulations of the present invention include polar solvents that are miscible in water.
- Non-limiting examples of these co-solvents include ethanol, isopropanol, glycol ethers (e.g., including, but limited to, diethyleneglycol monoethyl ether (DGME, Transcutol ® ), butyl diglycol, dipropylene glycol n- butyl ether, ethyleneglycol monoethyl ether, ethyleneglycol monomethyl ether, dipropylene glycol monomethyl ether, propylene glycol monomethyl ether, propylene glycol monoethyl ether, and the like), propylene glycol, carbonates (e.g., propylene carbonate), 2-pyrrolidone, N- methylpyrrolidone, dimethyl isosorbide (DMI), Dimethylacetamide, glycerol formal or a mixture of at least two of these solvents.
- glycol ethers e.g., including, but limited to, diethyleneglycol monoethyl ether (DGME, Trans
- the long acting formulations may comprise a polar protic solvent including, but not limited to, an alcohol such as ethanol, isopropanol or a glycol or glycol ether.
- the long-acting injectable formulations may comprise a polar aprotic solvent such as N-methylpyrrolidone, dimethyl isosorbide, dimethylacetamide or propylene carbonate.
- a polar aprotic solvent such as N-methylpyrrolidone, dimethyl isosorbide, dimethylacetamide or propylene carbonate.
- the long-acting injectable formulations may include non-water miscible co- solvents or solvents with only partial solubility in water.
- these co- solvents include benzyl alcohol, triacetin, lipids, triglycerides including medium chain triglycerides such C 8 -Cio triglycerides such as capric/caprilic triglycerides, propylene glycol derivatives (e.g. propylene glycol monolaurate), caprylocaproyl polyoxyl-8 glycerides (Labrasol) (non-ionic water dispersible surfactant, isopropyl myristate, or a mixture of at least two of these co-solvents.
- the long acting injectable formations may include neutral oils as a co-solvent.
- Neutral oils are triglycerides of fractionated plant fatty acids with chain lengths of C 8 to C 10 .
- Two commercially available products are known as MIGLYOL ® 810 and MIGLYOL ® 812.
- the neutral oil is a triglyceride of fractionated plant fatty acids with chain lengths of C 8 and C 10 combined with linoleic acid (about 4-5 %).
- a commercially available product is known as MIGLYOL ® 818.
- the neutral oil is a glycerin ester of fractionated plant fatty acids with chain lengths of C 8 and C 10 combined with succinic acid.
- a commercially available product is known as MIGLYOL ® 829.
- the neutral oil may be a propylene glycol diester of saturated plant fatty acids with chain lengths of C 8 and C 10 combined with succinic acid.
- a commercially available product is known as MIGLYOL ® 840 (propylene glycol dicaprylate/dicaprate).
- the co-solvent may be a mixture of two or more neutral oils.
- the long acting injectable formulations may contain other inert ingredients such as antioxidants, preservatives, or pH stabilizers. These compounds are well known in the formulation art.
- Antioxidants such as vitamin E, alpha tocopherol, ascorbic acid, ascorbyl palmitate, citric acid, fumaric acid, malic acid, sodium ascorbate, sodium metabi sulfate, sodium metabi sulfite, n-propyl gallate, BHA, BHT, BHA and citric acid, monothioglycerol, tert-butyl hydroquinone (TBHQ), and the like, may be added to the present formulation.
- the antioxidants are generally added to the formulation in amounts of from about 0.01 to about 2.0%, based upon total weight of the formulation, with about 0.05 to about 1.0% being especially preferred.
- Preservatives such as the parabens (methylparaben and/or propylparaben), are suitably used in the formulation in amounts ranging from about 0.01 to about 2.0%>, with about 0.05 to about 1.0% being especially preferred.
- Other preservatives include benzalkonium chloride, benzethonium chloride, benzoic acid, benzyl alcohol, bronopol, butylparaben, cetrimide, chlorhexidine, chlorobutanol, chlorocresol, cresol, ethylparaben, imidurea, methylparaben, phenol, phenoxyethanol, phenylethyl alcohol, phenylmercuric acetate, phenylmercuric borate, phenylmercuric nitrate, potassium sorbate, sodium benzoate, sodium propionate, sorbic acid, thimerosal, and the like. Preferred ranges for these compounds include from about 0.01 to about 5%.
- Buffering systems include, for example, systems selected from the group consisting of acetic acid/acetate, malic acid/malate, citric acid/citrate, tartaric acid/tartrate, lactic acid/lactate, phosphoric acid/phosphate, glycine/glycimate, tris, glutamic acid/glutamates and sodium carbonate.
- the parasiticidal compositions may be extended release injectable formulations.
- Extended release injectable formulations comprise a bioactive agent or agents, a pharmaceutically acceptable polymer (e.g., PLGA polymer) and at least one solvent; optional ingredients include a surfactant, a pharmaceutically acceptable additive or excipient.
- Extended- release injectable formulations comprising an isoxazoline compound as a bioactive agent are described, for example in application PCT/US2016/026253, herein incorporated by reference.
- the pharmaceutically acceptable polymers in the extended release injectable formulations include, but are not limited to, polylactides, polyglycolides, polycaprolactones, polyanhydrides, polyamides, polyurethanes, polyesteramides, polyorthoesters, polydioxanones, polyacetals, polyketals, polycarbonates, polyorthocarbonates, polyphosphazenes, pseudo poly(amides), polyhydroxyalcanoates, polyhydroxybutyrates, polyhydroxyvalerates, polyalkylene oxalates, polyalkylene succinates, poly(malic acid), poly(amino acids), poly(methyl vinyl ether), poly(maleic anhydride), chitin, chitosan, and copolymers, terpolymers, or combinations or mixtures therein including copolymers of polylactides, polycaprolactones, polyglycolides (e.g., poly(lactide-co-glycolide) and copolymers of poly
- the extended release compositions may also include pharmaceutically acceptable polymers that are not biodegradable such as PEGs and poloxamers. Also included are derivatives of pharmaceutically acceptable polymers such as hydroxylated derivatives including polycaprolactone diols and the like.
- the pharmaceutically acceptable polymer may be, for example, a biodegradable polymer and might have one or more or all of the following characteristics: be bioerodible by cellular action, biodegradable by action of non-living body fluid components, soften when exposed to heat but return to the original state when cooled and are capable of substantially dissolving or dispersing in a water-miscible carrier or solvent to form a solution or dispersion.
- a biodegradable polymer Upon contact with an aqueous fluid the polymer is capable of assisting in the formation of the film coated or encapsulated liquid or solid (which will contain the active agent in the present invention).
- the kinds of polymers suitable for the present composition generally include any having the foregoing characteristics.
- biodegradable polymers include, but are not limited to, polylactides, polycaprolactones, polyglycolides, polyorthoesters, polyurethanes, polyphasphazenes, pseudo poly(amides), and copolymers thereof.
- the molecular weight of a polymer is not a discreet number but can be presented in a molecular weight range.
- the average molecular weight of a polymer may be found by techniques familiar to persons of skill in the art, for example, size exclusion chromatography with molecular weight standards, or the like.
- the molecular weight range of a polymer can impact the physical characteristics of the material and the way that it interacts with the active agent. Accordingly the molecular weight range of the polymer may impact the characteristics of the extended release compositions of the invention.
- the weight average molecular weight range is from about 5 to about 20 kDa (kilo daltons) or from about 7 to about 15 kDa.
- a later release of the isoxazoline active agent may be observed when the weight average molecular weight is between about 30 to about 70 kDa (e.g., about 40 to about 70 kDa or about 45 to about 60 kDa) or from about 90 to about 200 kDa (e.g., about 100 to about 150 or about 105 to about 130 kDa).
- a combination of polymers having different average molecular weights may provide a release rate that combines the effect of the different polymers used.
- Inherent viscosity (IV) in polymer chemistry is a viscometric method for measuring molecular weight. It is defined as the ratio of the natural logarithm of the relative viscosity to the mass concentration of the polymer and is based on the flow time of a polymer solution through a narrow capillary.
- the term “low molecular weight” refers to polymer with an inherent viscosity in the range of 0.05-0.29 dL/g; the term “medium molecular weight” refers to a polymer with an inherent viscosity in the range of 0.3-0.55dL/g; and high molecular weight refers to a polymer with an inherent viscosity in the range of 0.55-1.0 dL/g.
- the pharmaceutically acceptable polymer will have an inherent viscosity of about 0.10-0.20 dL/g.
- the pharmaceutically acceptable polymer will have an inherent viscosity of about 0.35-0.50.
- the extended release injectable formulations may comprise, for example, polylactides, polycaprolactones, polyglycolides and copolymers thereof.
- the compositions include a poly(lactide-co-glycolide) copolymer (PLGA).
- PLGA copolymers may have different molecular weight ranges and may also have different weigh weight ratios of lactide to glycolide. This ratio, may affect the properties of the copolymer and the way that it interacts with the active agent.
- a lactide group contains an additional methyl group in the sidechain compared with a glycolide, this change may affect the conformation of the polymer and change the way in which the polymer interacts with the isoxazoline active agent (and/or other active agent combined with the isoxazoline).
- extended-release injectable formulations having a higher lactide to glycolide ratio result in an increase in hydrogen bonding between the active agent and the polymer, leading to better solubility of the active agent in vivo. This effect improves the injection site reaction and allows for the extendable release injectable compositions to include a higher amount of the active agent, which will translate to a longer duration of efficacy.
- the ratio of lactide to glycolide is about 30:70 to about 99: 1. In other formulations, where the pharmaceutically acceptable polymer is PLGA, the ratio of lactide to glycolide is about 40:60 to about 80: 15. In other formulations, where the pharmaceutically acceptable polymer is PLGA, the ratio of lactide to glycolide is about 40:60 to about 60:40. In another embodiment of the formulations, where the pharmaceutically acceptable polymer is PLGA, the ratio of lactide to glycolide is about 70:30 to about 80:20. In another embodiment, where the pharmaceutically acceptable polymer is PLGA, the ratio of lactide to glycolide is about 50:50. In another embodiment, where the pharmaceutically acceptable polymer is PLGA, the ratio of lactide to glycolide is about 75:25.
- the amount of PLGA contained in the extended release injectable formulation is about 1% to about 30 % (w/w), In another embodiment, the compositions comprise about 1 to about 20% (w/w) of PLGA. In another embodiment, the compositions comprise about 5 to about 20%, about 8% to about 20% (w/w) or about 10 to about 20%) (w/w). In another embodiment, the formulations comprise about 5 to about 15% (w/w) of PLGA. In other embodiments, the amount of PLGA contained in the extended release injectable formulations is from about 3% to about 15% (w/w) or is from about 10% to about 15% (w/w). In yet another embodiment, the formulations comprise about 7 to about 13% or about 8 to about 15% (w/w) of PLGA.
- the weigh weight ratio of PLGA to the isoxazoline active agent is greater than or equal to about 1 : 1, for example, from about 1.1 : 1 to about 20: 1; e.g., about 1 : 1 to about 10: 1, about 1.1 : 1 to about 10: 1 or about 2: 1 to about 5: 1.
- the weigh weight ratio of PLGA to the isoxazoline active agent is about 1.2: 1 to about 5: 1.
- the weigh weight ratio of PLGA to the isoxazoline active agent is about 1.2: 1 to about 2: 1.
- the weight: weight ratio of PLGA to the isoxazoline active agent is about 1.2: 1 to about 1.3 : 1.
- the weightweight ratio of PLGA to the isoxazoline active agent is about 1.5: 1 to about 1 : 1.5. In other formulations, the ratio of the isoxazoline active agent to PLGA is from about 1.25: 1 to about 1 : 1 :25.
- the solvents used in theses extended-release injectable formulations may be a single or a blend of solvents.
- Non-limiting examples of these solvents include alcohols such as ethanol, 1- propanol, isopropanol, glycol ethers (e.g., including, but limited to, diethyleneglycol monoethyl ether (DGME, Transcutol ® ), butyl diglycol, dipropylene glycol n-butyl ether, ethyleneglycol monoethyl ether, ethyleneglycol monomethyl ether, dipropylene glycol monomethyl ether, propylene glycol monomethyl ether, propylene glycol monoethyl ether, and the like), liquid polyethylene glycols (PEGs) including, but not limited to, PEG 200, PEG 300 and PEG 400; propylene glycol, glycerol, glycerol esters including glycerol triacetate (triacetin), cyclic carbonates (e.
- the extended-release injectable formulations may include one or more poloxamers as a solvent or surfactant.
- the extended release injectable formulations may comprise a solvent or mixture of solvents that is miscible with water.
- Solvents that are miscible with water are well known and include certain alcohols, liquid PEGs, certain poloxamers, glycols and glycol ethers and polar aprotic solvents.
- Alcohols that are miscible with water include, but are not limited to ethanol, isopropanol, «-propanol, Solketal (isopropylidene glycerol) or glycerol formal.
- Polar aprotic solvents include, but are not limited to, amides such as dimethylacetamide, dimethylformamide, 2-pyrrolidone, N-alkylpyrrolidones such as N-methylpyrrolidone and N-octylpyrrolidone, dimethylisosorbide, dimethylsulfoxide, cyclic carbonates including propylene carbonate and ethylene carbonate, and certain ketones such as acetone and the like.
- Glycol ethers include, but are not limited to, diethyleneglycol monoethyl ether (DGME, Transcutol ® ), butyl diglycol, dipropylene glycol n-butyl ether, ethyleneglycol monoethyl ether, ethyleneglycol monomethyl ether, dipropylene glycol monomethyl ether, propylene glycol monomethyl ether, propylene glycol monoethyl ether, and the like.
- DGME diethyleneglycol monoethyl ether
- Transcutol ® diethyleneglycol monoethyl ether
- butyl diglycol dipropylene glycol n-butyl ether
- ethyleneglycol monoethyl ether ethyleneglycol monomethyl ether
- dipropylene glycol monomethyl ether dipropylene glycol monomethyl ether
- propylene glycol monomethyl ether propylene glycol monoethyl ether
- the extended release formulations may comprise, for example, a polar protic solvent including, but not limited to, an alcohol such as ethanol, isopropanol or a glycol or glycol ether.
- a polar protic solvent including, but not limited to, an alcohol such as ethanol, isopropanol or a glycol or glycol ether.
- the formulation may comprise, for example, a polar aprotic solvent such as N-methylpyrrolidone, dimethyl isosorbide, dimethylacetamide or propylene carbonate.
- a polar aprotic solvent such as N-methylpyrrolidone, dimethyl isosorbide, dimethylacetamide or propylene carbonate.
- the extended release injectable formulations may include non-water miscible solvents
- Non- limiting examples of these solvents include 1-butanol, 2-butanol, 1-pentanol, 3-pentanol, benzyl alcohol, methylethylketone (MEK), triacetin, lipids, triglycerides including medium chain triglycerides such C 8 -Cio triglycerides such as capric/caprilic triglycerides, propylene glycol derivatives (e.g.
- propylene glycol monolaurate caprylocaproyl polyoxyl-8 glycerides (Labrasol) (non-ionic water dispersible surfactant, isopropyl myristate, oils such as castor oil, soybean oil or other vegetable oils or derivatives thereof such as epoxidized or hydrogenated vegetable oils such as epoxidized soybean oil or hydrogenated castor oil, or a mixture of at least two of these solvents.
- Labrasol non-ionic water dispersible surfactant, isopropyl myristate, oils such as castor oil, soybean oil or other vegetable oils or derivatives thereof such as epoxidized or hydrogenated vegetable oils such as epoxidized soybean oil or hydrogenated castor oil, or a mixture of at least two of these solvents.
- the extend release formulations may include neutral oils as a solvent.
- Neutral oils are triglycerides of fractionated plant fatty acids with chain lengths of C 8 to Cio.
- Two commercially available products are known as MIGLYOL® 810 and MIGLYOL®812.
- the neutral oil is a triglyceride of fractionated plant fatty acids with chain lengths of C 8 and Ci 0 combined with linoleic acid (about 4-5 %).
- a commercially available product is known as MIGLYOL® 818.
- the neutral oil is a glycerin ester of fractionated plant fatty acids with chain lengths of C 8 and Cio combined with succinic acid.
- a commercially available product is known as MIGLYOL® 829.
- the neutral oil is a propylene glycol fatty acid ester.
- the neutral oil may be a propylene glycol diester of saturated plant fatty acids with chain lengths of C 8 and Cio.
- a commercially available product is known as MIGLYOL® 840 (propylene glycol dicaprylate/dicaprate).
- the solvent may be a mixture of two or more neutral oils.
- blends of solvents may be used as the solvent of the extended release injectable formulations.
- the compositions of the invention may contain a blend of a water-miscible solvent with a solvent that is not water miscible.
- the solvent may be a mixture of a cyclic carbonate such as propylene carbonate with triacetin.
- a water-miscible solvent and a non-water miscible solvent are possible.
- the water-miscible solvent in the solvent blend may be a water-miscible alcohol such as ethanol or isopropanol, glycerol formal or Solketal, an amide such as 2-pyrrolidone, N-methylpyrrolidone, dimethylisosorbide or dimethylacetamide, a glycol such as propylene glycol, glycerol or a glycol ether.
- a water-miscible alcohol such as ethanol or isopropanol, glycerol formal or Solketal
- an amide such as 2-pyrrolidone, N-methylpyrrolidone, dimethylisosorbide or dimethylacetamide
- a glycol such as propylene glycol, glycerol or a glycol ether.
- the non-water miscible solvent in the solvent blend may be, for example, triacetin, benzyl alcohol, a triglyceride including C 8 -Cio triglycerides such as capric/caprilic triglycerides, propylene glycol derivatives (e.g. propylene glycol monolaurate), caprylocaproyl polyoxyl-8 glycerides (Labrasol); a propylene glycol fatty acid diester, and the like.
- triacetin benzyl alcohol
- a triglyceride including C 8 -Cio triglycerides such as capric/caprilic triglycerides
- propylene glycol derivatives e.g. propylene glycol monolaurate
- caprylocaproyl polyoxyl-8 glycerides Labrasol
- a propylene glycol fatty acid diester and the like.
- the solvent in the extended release formulations in the inventive methods may be a blend of a water-miscible solvent and a non-water miscible solvent in a weight: weight ratio of between about 10 to 1 to about 1 to 10, water-miscible solvent to non- water miscible solvent.
- the weigh weight ratio of the water-miscible solvent to non-water miscible solvent may be from about 5 to 1 to about 1 to 1.
- the weigh weight ratio of the water-miscible solvent to non-water miscible solvent may be from about 3 to 1 to about 1 to 1.
- the weight: weight ratio of the water-miscible solvent to non-water miscible solvent may be from about 3 to 1 to about 2 to 1 or about 2 to 1 to about 1 to 1.
- the solvent in the extended release formulations in the inventive methods may be a blend of a water-miscible solvent and a non-water miscible solvent in a weightweight ratio of about 1 to 2 or about 1 to 3, water-miscible solvent to non-water miscible solvent.
- the weightweight ratio of the water-miscible solvent to non-water miscible solvent may be from about 1 to about 5, 1 to about 7.
- the solvent in the extended release formulations in the inventive methods may be a blend of cyclic carbonate (e.g., propylene carbonate) and glycerol ester (e.g., triacetin) in a weight: weight ratio of between about 10 to 1 to about 1 to 1, cyclic carbonate (e.g., propylene carbonate) to glycerol ester (e.g., triacetin).
- the solvent may be a blend of cyclic carbonate (e.g., propylene carbonate) and glycerol ester (e.g., triacetin) in a weight: weight ratio of between about 5 to 1 to about 1 to 1, cyclic carbonate (e.g.
- the solvent may be a blend of cyclic carbonate (e.g., propylene carbonate) and glycerol ester (e.g., triacetin) in a weight: weight ratio of between about 3 to 1 to about 1 to 1, cyclic carbonate (e.g. propylene carbonate) to glycerol ester (e.g., triacetin).
- the solvent may be a blend of cyclic carbonate (e.g., propylene carbonate) and glycerol ester (e.g., triacetin) in a weight: weight ratio of between about 2 to 1 to about 1 to 1 or about 3 : 1 to about 2: 1, cyclic carbonate (e.g. propylene carbonate) to glycerol ester (e.g., triacetin).
- the range for the weigh weight ratio of cyclic carbonate (e.g. propylene carbonate) to glycerol ester (e.g., triacetin) is 1.5: 1 to about 15: 1 or from about 2: 1 to about 6: 1.
- the extended release injectable formulations may include surfactants, which may be present at concentrations of about 0.1 % to about 10% (w/w), about 1 % to about 10% (w/w) or about 5% to about 10% (w/w). More typically, surfactants may be present at concentrations of about 0.1 % to about 5% (w/w) or about 1 to about 5% (w/w).
- surfactants examples include, but are not limited to, glyceryl monooleate, poly oxy ethylene sorbitan fatty acid esters, sorbitan esters 30 including sorbitan monooleate (Span ® 20), polyvinyl alcohol, polysorbates including polysorbate 20 and polysorbate 80, d-a- tocopheryl polyethylene glycol 1000 succinate (TPGS), sodium lauryl sulfate, co-polymers of ethylene oxide and propylene oxide (e.g.
- poloxamers such as LUTROL ® F87 and the like
- polyethylene glycol castor oil derivatives including polyoxyl 35 castor oil (Cremophor ® EL), polyoxyl 40 hydrogenated castor oil (Cremophor ® RH 40), polyoxyl 60 hydrogenated castor oil (Cremophor ® RH60); propylene glycol monolaurate (LAUROGLYCOL ® ); glyceride esters including glycerol caprylate/caprate (CAPMUL ® MCM), polyglycolized glycerides (GELUCIRE ® ), PEG 300 caprylic/capric glycerides (Softigen ® 767), PEG 400 caprylic/capric glycerides (Labrasol ® ), PEG 300 oleic glycerides (Labrafil® M-1944CS), PEG 300 linoleic glycerides (Labrafil ® M-2125CS); polyethylene glycol stearate
- Polyethylene glycol stearates are mixtures of mono- and distearate esters of mixed polyoxyethylene polymers.
- Polyethylene glycol hydroxystearate is a mixture of mono- and diesters of hydroxystearic acid with polyethylene glycols.
- One polyethylene glycol hydroxystearate that may be used in the compositions is polyethylene glycol 12-hydroxy stearate.
- the formulations may include the surfactant polyethylene glycol 15 12- hydroxystearate (Kolliphor ® HS 15 from BASF), a mixture of mono- and diesters of 12- hydroxystearic acid with 15 moles of ethylene oxide. Again, these compounds, as well as their amounts are well known in the art.
- the inventive formulations may include polyoxyl 35 castor oil (Kolliphor ® EL) as a surfactant.
- the inventive formulations may include polyoxyl 40 hydrogenated castor oil (Kolliphor ® RH 40) or polyoxyl 60 hydrogenated castor oil as surfactants.
- the formulations of the invention may also include a combination of surfactants.
- the extended release injectable formulations may also include other inert ingredients such as antioxidants, preservatives, or pH stabilizers. These compounds are well known in the formulation art. Antioxidants such as vitamin E, alpha tocopherol, ascorbic acid, ascorbyl palmitate, citric acid, fumaric acid, malic acid, sodium ascorbate, sodium metabi sulfate, sodium metabi sulfite, n-propyl gallate, BHA, BHT, BHA and citric acid, monothioglycerol, tert-butyl hydroquinone (TBHQ), and the like, may be added.
- antioxidants such as antioxidants, preservatives, or pH stabilizers.
- Antioxidants such as vitamin E, alpha tocopherol, ascorbic acid, ascorbyl palmitate, citric acid, fumaric acid, malic acid, sodium ascorbate, sodium metabi sulfate, sodium metabi sulfite, n-propyl gallate, B
- the antioxidants are generally added to the formulation in amounts of from about 0.01 to about 5.0%, based upon total weight of the formulation, with about 0.05 to about 2.0% being especially preferred.
- the formulation preferably contains about 0.05 to about 1.0% (w/w) of an antioxidant.
- Preservatives such as the parabens (methylparaben, ethylparaben, butylparaben and/or propylparaben), are suitably used in the extended release injectable formulations in amounts ranging from about 0.01 to about 2.0%, with about 0.05 to about 1.0% being especially preferred.
- preservatives include benzalkonium chloride, benzethonium chloride, benzoic acid, benzyl alcohol, bronopol, cetrimide, chlorhexidine, chlorobutanol, chlorocresol, cresol, imidurea, phenol, phenoxyethanol, phenylethyl alcohol, phenylmercuric acetate, phenylmercuric borate, phenylmercuric nitrate, potassium sorbate, sodium benzoate, sodium propionate, sorbic acid, thimerosal, and the like. Preferred ranges for these compounds include from about 0.01 to about 5%.
- Buffering systems include, for example, systems selected from the group consisting of acetic acid/acetate, malic acid/malate, citric acid/citrate, tartaric acid/tartrate, lactic acid/lactate, phosphoric acid/phosphate, glycine/glycimate, tris, glutamic acid/glutamates and sodium carbonate
- the extended release injectable formulations used in the inventive methods may comprise, for example, about 1 to about 40% (w/w) of a pharmaceutically acceptable polymer, including a biodegradable polymer.
- the formulations may comprise about 1 to about 30%) (w/w) or about 1 to about 20% (w/w) of a pharmaceutically acceptable polymer.
- the formulations may comprise about 1 to about 15%> (w/w) or about 1 to about 10%) (w/w) of a pharmaceutically acceptable polymer.
- the formulations may comprise about 5 to about 20% (w/w) or about 5 to about 15%> (w/w) of a pharmaceutically acceptable polymer.
- the formulations may comprise about 10 to about 20% (w/w) or about 10 to about 15%> (w/w) of a pharmaceutically acceptable polymer. In another embodiment, the formulations may comprise about 7 to about 13%> (w/w) or about 8 to about 15%> (w/w) of a pharmaceutically acceptable polymer. In yet another embodiment, the formulations may comprise about 1 to about 7% (w/w), about 1 to about 5% (w/w) or about 3 to about 7% (w/w) of a pharmaceutically acceptable polymer.
- the extended release injectable formulations used in the inventive methods may comprise, for example, about 30% to about 90% (w/w) of a solvent or mixture of solvents.
- formulations may comprise about 40% to about 90% (w/w) of a solvent or mixture of solvents.
- the formulations may comprise about 40% to about 80% (w/w), about 50% to about 80% (w/w) or about 45% to about 80% (w/w) of a solvent or a mixture of solvents.
- the formulations may comprise about 60% to about 80%) (w/w) or about 65% to about 80% (w/w) of a solvent or a mixture of solvents.
- the formulations may comprise about 65% to about 75% (w/w) or about 70%) to about 80% (w/w) of a solvent or a mixture of solvents.
- the extended release injectable formulations used in the inventive methods may comprise, for example, about 0.01% to about 10% (w/w) of a pharmaceutically acceptable additive, excipient or mixtures thereof.
- the formulations may comprise about 0.01% to about 5% (w/w), about 0.1% to about 10% (w/w) or about 0.1% to about 5% (w/w) of a pharmaceutically acceptable additive, excipient or mixtures thereof.
- the extended release injectable formulations may comprise, for example, about 0.01% to about 5%) (w/w) of an antioxidant. In other embodiments, the formulations may comprise about 0.01%) to about 3%) (w/w) or about 0.01 to about 2% (w/w) of an antioxidant.
- the long acting and extended release injectable formulations used in the inventive methods may comprise, for example, from about 1 to about 50% (w/w) of an isoxazoline bioactive agent,
- the extended release formulations may include lower concentrations of the compound such as about 1 to about 10%) (w/w) or about 1 to about 5% (w/w).
- the formulations may comprise about 5 to about 30%> (w/w) of the isoxazoline bioactive agent.
- the formulations may include about 5 to about 20% (w/w) or about 5 to about 15%> (w/w) of the isoxazoline bioactive agent.
- the formulations may comprise about 10 to about 40% (w/w) or 10 to about 30%> (w/w) of an isoxazoline bioactive agent.
- the formulations may comprise about 10 to about 20% of an isoxazoline bioactive agent.
- the formulations may comprise about 15% to about 40%) (w/w), about 15% to about 35% (w/w) or about 15% to about 30% (w/w) of an isoxazoline compound.
- the formulations may comprise about 20 to about 30% (w/w), about 20 to about 25% (w/w) or about 25 to about 30% (w/w) of the isoxazoline bioactive agent.
- Dosage forms may contain from about 0.5 mg to about 5 g of a single or combination of bioactive agent(s) in the long acting and extended release formulations used in the methods of the invention. More typically, the amount of active agent(s) in the long acting and extended release formulations used in the inventive methods will be from about 1 mg to about 3 g. In another embodiment, the amount of bioactive agent(s) in the compositions will be from about 20 mg to about 3 g. In another embodiment, the amount of bioactive agent(s) present in the long acting and extended release formulations will be from about 20 mg to about 2 g, about 20 mg to about 1.5 g or about 20 mg to about 1 g.
- the amount of bioactive agent(s) in the compositions will be from about 20 mg to about 500 mg, about 30 mg to about 200 mg or about 50 mg to about 200 mg. In still another embodiment, the amount of bioactive agent(s) present in the compositions will be from about 50 mg to about 2 g, about 50 mg to about 1 g or about 50 mg to about 500 mg. In yet another embodiment of the invention, the about of bioactive agent(s) present will be from about 100 mg to about 2 g, about 100 mg to about 1 g or about 100 mg to about 500 mg.
- Additional veterinary/pharmaceutical bioactive active agents may be used with the compositions of the invention.
- the additional active agents may include, but are not limited to, acaricides, anthelmintics, anti-parasitics and insecticides.
- Anti-parasitic agents can include both ectoparasiticidal and/or endoparasiticidal agents.
- Veterinary pharmaceutical agents that may be included in the compositions of the invention are well-known in the art (see e.g. Plumb 's Veterinary Drug Handbook, 5 th Edition, ed. Donald C. Plumb, Blackwell Publishing, (2005) or The Merck Veterinary Manual, 9 th Edition, (January 2005)) and include but are not limited to acarbose, acepromazine maleate, acetaminophen, acetazol amide, acetazolamide sodium, acetic acid, acetohydroxamic acid, acetylcysteine, acitretin, acyclovir, albendazole, albuterol sulfate, alfentanil, allopurinol, alprazolam, altrenogest, amantadine, amikacin sulfate, aminocaproic acid, aminopentamide hydrogen sulfate, aminophylline/theophylline, amiodarone, amitript
- the isoxazoline compound in the parasiticidal compositions of the present methods may be combined with arylpyrazole compounds, such as phenylpyrazoles, known in the art.
- arylpyrazole compounds include but are not limited to fipronil, pyriprole, ethiprole and those described in U.S. Patent Nos. 6,001,384; 6,010,710; 6,083,519; 6,096,329; 6, 174,540; 6,685,954 and 6,998, 131 (all of which are incorporated herein by reference).
- the isoxazoline compound in the parasiticidal compositions of the present methods may combined also be combined with one or more macrocyclic lactones or lactams, which act as an acaricide, anthelmintic agent and/or insecticide.
- the macrocyclic lactones include, but are not limited to, avermectins such as abamectin, dimadectin, doramectin, emamectin, eprinomectin, ivermectin, latidectin, lepimectin, selamectin and ML-1, 694,554, and milbemycins such as milbemectin, milbemycin D, milbemycin oxime, moxidectin and nemadectin. Also included are the 5-oxo and 5-oxime derivatives of said avermectins and milbemycins.
- the macrocyclic lactone compounds are known in the art and can easily be obtained commercially or through synthesis techniques known in the art. Reference is made to the widely available technical and commercial literature.
- avermectins ivermectin and abamectin
- doramectin "Veterinary Parasitology", vol. 49, No.
- milbemycins reference may be made, inter alia, to Davies H.G. et al., 1986, "Avermectins and Milbemycins", Nat. Prod. Rep., 3, 87-121, Mrozik H. et al., 1983, Synthesis of Milbemycins from Avermectins, Tetrahedron Lett., 24, 5333-5336, U.S. Patent No. 4,134,973 and EP 0 677 054.
- Macrocyclic lactones are either natural products or are semi-synthetic derivatives thereof.
- the structure of the avermectins and milbemycins are closely related, e.g., by sharing a complex 16-membered macrocyclic lactone ring.
- the natural product avermectins are disclosed in U.S. Patent No. 4,310,519 and the 22,23-dihydro avermectin compounds are disclosed in U.S. Patent No. 4, 199,569. Mention is also made of U.S. Patent Nos. 4,468,390, 5,824,653, EP 0 007 812 Al, U.K. Patent Specification 1 390 336, EP 0 002 916, and New Zealand Patent No.
- the parasiticidal compositions comprise an isoxazoline compound in combination with systemically-acting compounds from a class of acaricides or insecticides known as insect growth regulators (IGRs).
- IGRs insect growth regulators
- Compounds belonging to this group are well known to the practitioner and represent a wide range of different chemical classes. These compounds all act by interfering with the development or growth of the insect pests.
- Insect growth regulators are described, for example, in U.S. Patent Nos. 3,748,356, 3,818,047, 4,225,598, 4,798,837, 4,751,225, EP 0 179 022 or U.K. 2 140 010 as well as U.S. Patent Nos. 6,096,329 and 6,685,954 (all incorporated herein by reference).
- the IGR is a compound that mimics juvenile hormone.
- juvenile hormone mimics include azadirachtin, diofenolan, fenoxycarb, hydroprene, kinoprene, methoprene, pyriproxyfen, tetrahydroazadirachtin and 4-chloro-2(2-chloro-2-methyl-propyl)-5- (6-iodo-3-pyridylmethoxy)pyridazine-3(2H)-one.
- the IGR is methoprene or pyriproxyfen.
- the IGR compound is a chitin synthesis inhibitor.
- Chi tin synthesis inhibitors include chlorofluazuron, cyromazine, diflubenzuron, fluazuron, flucycloxuron, flufenoxuron, hexaflumoron, lufenuron, tebufenozide, teflubenzuron, triflumoron, novaluron, l-(2,6-difluorobenzoyl)-3-(2-fluoro-4-(trifluoromethyl)phenylurea, l-(2,6-difluoro- benzoyl)-3-(2-fluoro-4-(l,l,2,2-tetrafluoroethoxy)-phenylurea and l-(2,6-difluorobenzoyl)-3-(2- fluoro-4-trifluoromethyl)phenylurea.
- adulticide insecticides and acaricides can also be added to the extended release formulations of the present invention.
- pyrethrins which include cinerin I, cinerin II, jasmolin I, jasmolin II, pyrethrin I, pyrethrin II and mixtures thereof
- carbamates including, but are not limited to, benomyl, carbanolate, carbaryl, carbofuran, meththiocarb, metolcarb, promacyl, propoxur, aldicarb, butocarboxim, oxamyl, thiocarboxime and thiofanox.
- the parasiticidal compositions comprise an isoxazoline compound combined with permethrin.
- the parasiticidal compositions of the inventive methods may include one or more antinematodal agents including, but not limited to, active agents in the benzimidazoles, imidazothiazoles, tetrahydropyrimidines, and organophosphate class of compounds.
- benzimidazoles including, but not limited to, thiabendazole, cambendazole, parbendazole, oxibendazole, mebendazole, flubendazole, fenbendazole, oxfendazole, albendazole, cyclobendazole, febantel, thiophanate and its ⁇ , ⁇ -dimethyl analogue may be included in the compositions.
- the parasiticidal compositions of the inventive methods may include an imidazothiazole compounds including, but not limited to, tetramisole, levamisole and butamisole.
- the extended release formulations of the present invention may include tetrahydropyrimidine active agents including, but not limited to, pyrantel, oxantel, and morantel.
- Suitable organophosphate active agents include, but are not limited to, coumaphos, trichlorfon, haloxon, naftalofos and dichlorvos, heptenophos, mevinphos, monocrotophos, TEPP, and tetrachlorvinphos.
- the parasiticidal compositions of the inventive methods may include the antinematodal compounds phenothiazine and piperazine as the neutral compound or in various salt forms, diethylcarbamazine, phenols such as disophenol, arsenicals such as arsenamide, ethanolamines such as bephenium, thenium closylate, and methyridine; cyanine dyes including pyrvinium chloride, pyrvinium pamoate and dithiazanine iodide; isothiocyanates including bitoscanate, suramin sodium, phthalofyne, and various natural products including, but not limited to, hygromycin B, a-santonin and kainic acid.
- the parasiticidal compositions of the inventive methods may include antitrematodal agents.
- Suitable antitrematodal agents include, but are not limited to, the miracils such as miracil D and mirasan; praziquantel, epsiprantel, clonazepam and its 3-methyl derivative, oltipraz, lucanthone, hycanthone, oxamniquine, amoscanate, niridazole, nitroxynil, various bisphenol compounds known in the art including hexachlorophene, bithionol, bithionol sulfoxide and menichlopholan; various salicylanilide compounds including tribromsalan, oxyclozanide, clioxanide, rafoxanide, brotianide, bromoxanide and closantel; triclabendazole, diamfenetide, clorsulon, hetolin and
- Anticestodal compounds may also be advantageously used in parasiticidal compositions of the inventive methods including, but not limited to, praziquantel, epsiprantel, and arecoline in various salt forms, bunamidine, niclosamide, nitroscanate, paromomycin and paromomycin II.
- parasiticidal compositions of the inventive methods may include other active agents that are effective against arthropod parasites.
- Suitable active agents include, but are not limited to, bromocyclen, chlordane, DDT, endosulfan, lindane, methoxychlor, toxaphene, bromophos, bromophos-ethyl, carbophenothion, chlorfenvinphos, chlorpyrifos, crotoxyphos, cythioate, diazinon, dichlorenthion, diemthoate, dioxathion, ethion, famphur, fenitrothion, fenthion, fospirate, iodofenphos, malathion, naled, phosalone, phosmet, phoxim, propetamphos, ronnel, stirofos, allethrin, cyhalothrin, cypermethrin,
- An antiparasitic agent that can be combined with an isoxazoline compounds in the parasiticidal formulations of the inventive methods can be a biologically active peptide or protein including, but not limited to, depsipeptides, which act at the neuromuscular junction by stimulating presynaptic receptors belonging to the secretin receptor family resulting in the paralysis and death of parasites.
- the depsipeptide is emodepside (see Willson et al., Parasitology, Jan. 2003, 126(Pt l):79-86).
- the depsipeptide is PF1022A or a derivative thereof.
- the isoxazoline compounds may be combined with a cyclic depsipeptide described in WO 2016/187534 Al or WO 2017/116702 Al, both incorporated herein by reference in their entirety.
- the parasiticidal compositions of the inventive methods may comprise an active agent from the neonicotinoid class of pesticides.
- the neonicotinoids bind and inhibit insect specific nicotinic acetylcholine receptors.
- the neonicotinoid insecticidal agent that can be combined with an isoxazoline compound to form an extended release injectable formulation of the invention is imidacloprid.
- Imidacloprid is a well-known neonicotinoid active agent and is the key active ingredient in the topical parasiticide products Advantage ® , Advantage ® II, K9 Advantix ® , and K9 Advantix ® II sold by Bayer Animal Health and the oral soft-chewable formulation AdvantusTM from Piedmont Animal Health. Agents of this class are described, for example, in U.S. Patent No. 4,742,060 or in EP 0 892 060.
- parasiticidal compositions of the inventive methods may comprise nitenpyram, another active agent of the neonicotinoid class of pesticides in combination with the isoxazoline compound.
- Nitenpyram is the active ingredient in the oral product CAPSTARTM Tablets sold by Novartis Animal Health.
- the parasiticidal composition comprises an insecticidal agent that is a semicarbazone, such as metaflumizone.
- the parasitical compositions of the inventive methods may advantageously include a combination of a second isoxazoline compounds known in the art.
- active agents are described in WO 2007/079162, WO 2007/075459 and US 2009/0133319, WO 2007/070606 and US 2009/0143410, WO 2009/003075, WO 2009/002809, WO 2009/024541, WO 2005/085216 and US 2007/0066617 and WO 2008/122375, all of which are incorporated herein by reference in their entirety.
- nodulisporic acid and its derivatives may be added to the parasiticidal compositions of the present methods.
- These compounds are used to treat or prevent infections in humans and animals and are described, for example, in U.S. Patent No. 5,399,582, 5,962,499, 6,221,894 and 6,399,786, all of which are hereby incorporated by reference in their entirety.
- the formulations may include one or more of the known nodulisporic acid derivatives in the art, including all stereoisomers, such as those described in the patents cited above.
- anthelmintic compounds of the amino acetonitrile class (AAD) of compounds such as monepantel (ZOLVIX), and the like may be added to the parasiticidal compositions of the inventive methods.
- AAD amino acetonitrile class
- ZOLVIX monepantel
- the parasiticidal compositions of the present methods may also include aryloazol-2-yl cyanoethylamino compounds such as those described in US Patent No. 8,088,801 to Soil et al., which is incorporated herein in its entirety, and thioamide derivatives of these compounds, as described in U.S. Patent No. 7,964,621, which is incorporated herein by reference.
- the parasiticidal compositions of the inventive methods may also be combined with paraherquamide compounds and derivatives of these compounds, including derquantel (see Ostlind et al., Research in Veterinary Science, 1990, 48, 260-61; and Ostlind et al., Medical and Veterinary Entomology, 1997, 11, 407-408).
- the paraherquamide family of compounds is a known class of compounds that include a spirodioxepino indole core with activity against certain parasites (see Tet. Lett. 1981, 22, 135; J. Antibiotics 1990, 43, 1380, and J. Antibiotics 1991, 44, 492).
- marcfortines A-C structurally related marcfortine family of compounds, such as marcfortines A-C, are also known and may be combined with the formulations of the invention (see J. Chem. Soc. - Chem. Comm. 1980, 601 and Tet. Lett. 1981, 22, 1977). Further references to the paraherquamide derivatives can be found, for example, in WO 91/09961, WO 92/22555, WO 97/03988, WO 01/076370, WO 09/004432, U.S. Patent 5,703,078 and U.S. Patent 5,750,695, all of which are hereby incorporated by reference in their entirety.
- the parasiticidal compositions may include a spinosyn active agent produced by the soil actinomycete Saccharopolyspora spinosa (see, for example Salgado V.L. and Sparks T.C., "The Spinosyns: Chemistry, Biochemistry, Mode of Action, and Resistance, ' " in Comprehensive Molecular Insect Science, vol. 6, pp. 137-173, 2005) or a semi-synthetic spinosoid active agent.
- a spinosyn active agent produced by the soil actinomycete Saccharopolyspora spinosa (see, for example Salgado V.L. and Sparks T.C., "The Spinosyns: Chemistry, Biochemistry, Mode of Action, and Resistance, ' " in Comprehensive Molecular Insect Science, vol. 6, pp. 137-173, 2005) or a semi-synthetic spinosoid active agent.
- the spinosyns are typically referred to as factors or components A, B, C, D, E, F, G, H, J, K, L, M, N, O, P, Q, R, S, T, U, V, W, or Y, and any of these components, or a combination thereof, may be used in the compositions of the invention.
- the spinosyn compound may be a 5,6,5-tricylic ring system, fused to a 12-membered macro cyclic lactone, a neutral sugar (rhamnose), and an amino sugar (forosamine).
- spinosyn compounds including 21-butenyl spinosyn produced by Saccharopolyspora pagona, which may be used in the parasiticidal compositions of the inventive methods, may be produced via fermentation by conventional techniques known in the art.
- Other spinosyn compounds that may be used in the compositions of the invention are disclosed in U.S. Patent Nos. 5,496,931; 5,670,364; 5,591,606; 5,571,901; 5,202,242; 5,767,253; 5,840,861; 5,670,486; 5,631, 155 and 6,001,981, all incorporated by reference herein in their entirety.
- the spinosyn compounds may include, but are not limited to, spinosyn A, spinosyn D, spinosad, spinetoram, or combinations thereof.
- Spinosad is a combination of spinosyn A and spinosyn D
- spinetoram is a combination of 3'-ethoxy-5,6-dihydro spinosyn J and 3'-ethoxy spinosyn L.
- the additional bioactive agent is included in the parasiticidal compositions of the inventive methods is in an amount of between about 0.1 ⁇ g and about 1000 mg. More typically, the additional bioactive agent may be included in an amount of about 10 ⁇ g to about 500 mg, about 1 mg to about 300 mg, about 10 mg to about 200 mg or about 10 mg to about 100 mg.
- the additional bioactive agent may be included in the composition to deliver a dose of about 5 ⁇ g/kg to about 50 mg/kg per weight of the animal. In other embodiments, the additional bioactive agent may be present in an amount sufficient to deliver a dose of about 0.01 mg/kg to about 30 mg/kg, about 0.1 mg/kg to about 20 mg/kg, or about 0.1 mg/kg to about 10 mg/kg of weight of animal. In other embodiments, the additional bioactive agent may be present in a dose of about 5 ⁇ g/kg to about 200 ⁇ g/kg or about 0.1 mg/kg to about 1 mg/kg of weight of animal. In still another embodiment of the invention, the additional bioactive agent is included in a dose between about 0.5 mg/kg to about 50 mg/kg. Parasitic Infestations and Infections
- the parasitic infestations and infections of the inventive methods are caused by ectoparasites and endoparasites.
- the methods of the invention treat or prevent parasitic infestations and infections (when including an endoparasiticidal agent) while reducing unwanted side effects.
- Ectoparasites include, but are not limited to, fleas, ticks, mites, mosquitoes, parasitic flies (e.g., horn fly, stable fly, blowfly) and lice.
- Endoparasites include, but are not limited to, cestodes, nematodes, hookworms and roundworms of the digestive tract of non-human animal and filarial worms such as Dirofilaria immitis.
- ectoparasites include one or more insect or arachnid including those of the genera Ctenocephalides, Rhipicephalus, Dermacentor, Ixodes, Amblyomma, Haemaphysalis, Hyalomma, Sarcoptes, Psoroptes, Otodectes, Chorioptes, Hypoderma, Damalinia, Linognathus, Haematopinus, Solenoptes, Trichodectes, and Felicola.
- Specific examples include, but are not limited to, cat and dog fleas ⁇ Ctenocephalides sp.
- Ctenocephalides felis, Ctenocephalides canis, and the like ticks (Rhipicephalus sp., Ixodes sp., Dermacentor sp., Amblyomma sp. and the like), and mites (Demodex sp., Sarcoptes sp., Otodectes sp. and the like), lice (Trichodectes sp., Cheyletiella sp., Linognathus sp., and the like), mosquitoes (Aedes sp., Culex sp., Anopheles sp., and the like) and parasitic flies (Haematobia sp. including Haematobia irritans, Musca sp., Stomoxys sp. including Stomoxys calcitrans, Dermatobia sp., Cochliomyia sp., and the like).
- ticks Rosicephalus
- ectoparasites include but are not limited to the tick genus Rhipicephalus, especially those of the species microplus (cattle tick), decoloratus and annulatus; myiasis such as Dermatobia hominis (known as Berne in Brazil) and Cochliomyia hominivorax (greenbottle); sheep myiasis such as Lucilia sericata, Lucilia cuprina (known as blowfly strike in Australia, New Zealand and South Africa).
- Flies proper namely those whose adult constitutes the parasite, such as Haematobia irritans (horn fly) and Stomoxys calcitrans (stable fly); lice such as Linognathus vituli, etc.; and mites such as Sarcoptes scabiei and Psoroptes ovis.
- Haematobia irritans horn fly
- Stomoxys calcitrans stable fly
- lice such as Linognathus vituli, etc.
- mites such as Sarcoptes scabiei and Psoroptes ovis.
- the above list is not exhaustive and other ectoparasites are well known in the art to be harmful to animals and humans. These include, for example migrating dipterous larvae.
- Endoparasites include helminths selected from the group consisting of Anaplocephala, Ancylostoma, Necator, Ascaris, Capillaria, Cooperia, Dipylidium, Dirofilaria, Echinococcus, Enterobius, Fasciola, Haemonchus, Oesophagostomum, Ostertagia, Toxocara, Strongyloides, Toxascaris, Trichinella, Trichuris, Angiostrongylus and Trichostrongylus, among others.
- the methods of the invention will prevent the occurrence of Heartworm disease by killing immature stages of Dirofilaria immitis before they can mature to adult worms.
- non-human animals which parasiticidal infections occurs include livestock and companion animals, such as cats, dogs, horses, birds including chickens, sheep, goats, pigs, deer, turkeys and cattle.
- livestock and companion animals such as cats, dogs, horses, birds including chickens, sheep, goats, pigs, deer, turkeys and cattle.
- the formulations of the invention are administered in parasiticidally effective amounts which are which are suitable to control the parasite in question to the desired extent, as described below.
- the compounds and compositions of the invention can be applied against a single pest or combinations thereof.
- a dose of from about 0.001 to about 100 mg per kg of body weight given as a single dose or in divided doses for a period of from 1 to 5 days will be satisfactory but, of course, there can be instances where higher or lower dosage ranges are indicated, and such are within the scope of this invention. It is well within the routine skill of the practitioner to determine a particular dosing regimen for a specific host and parasite.
- the dose of the isoxazoline bioactive agent administered is between about 0.1 to about 50 mg per kg of body weight. More typically the dose of the isoxazoline bioactive agent administered is about 0.5 to about 40 mg/kg or about 0.5 to about 30 mg/kg body weight. In another embodiment, the dose of the isoxazoline bioactive agent administered is about 10 to about 40 mg/kg, about 15 to about 35 mg/kg or about 20 to about 30 mg/kg of body weight. In another embodiment, the dose of the isoxazoline active bioagent will be about 20 to about 25 mg/kg of body weight.
- the dose administered may be lower depending on the animal and the isoxazoline administered.
- the dose is from about 0.1 to about 30 mg/kg of body weight.
- the dose may be from about 0.1 to about 20 mg/kg or about 0.1 to about 10 mg/kg of body weight.
- the dose may be from about 1 to about 20 mg/kg of body weight, about 1 to about 10 mg/kg or about 1 to about 5 mg/kg.
- the dose may be from about 0.1 to about 2 mg/kg or from about 0.5 to about 2 mg/kg.
- the dose may be from about 5 to about 20 mg/kg, about 5 to about 15 mg/kg or about 10 to about 20 mg/kg of body weight.
- the dose may be from about 10 to about 30 mg/kg of body weight.
- doses of the isoxazoline bioactive agent administered may be about 0.1 to about 40 mg/kg of body weight. More typically the doses administered will be about 1 to about 30 mg/kg, about 1 to about 20 mg/kg or about 1 to about 10 mg/kg of body weight. In yet another embodiment, the dose may be from about 10 to about 25 mg/kg, about 15 to about 30 mg/kg of body weight or about 20 to about 30 mg/kg of body weight. In another embodiment, the dose for livestock animals may be from about 0.1 mg/kg to about 3 mg/kg, from about 0.1 mg/kg to about 2 mg/kg or from about 0.1 mg/kg to about 1 mg/kg of body weight.
- the parasiticidal compounds and compositions of the inventive methods may exhibit an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least about 90% or higher for about 1 month, or longer.
- the parasiticidal compounds and compositions of the inventive methods will exhibit an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least about 90% for about 2 months or about 3 months.
- These compositions may provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least 95.0% or higher for about, 3 months or longer.
- parasiticidal compositions in the inventive methods may provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least 90% or higher for about 6 months or longer.
- parasiticidal formulations of the inventive methods may provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least 95% or higher for about 6 months or longer.
- the parasiticidal provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least 90% or higher for about 9 months or longer.
- the parasiticidal compositions provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least 90% or higher for about 12 months or longer. In another embodiment, the parasiticidal compositions provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites in cats and dogs of at least about 90% for two months, or longer. In another embodiment, the efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites in cats and dogs of about 95% for about 3 months, or longer. In still another embodiment, the parasiticidal compositions provide an efficacy of about 95% for about 5 months or longer.
- the parasiticidal compositions provide an efficacy against
- the parasiticidal compositions provide an efficacy against Rhipicephalus microplus in cattle of at least about 90% for one month, or longer.
- the parasiticidal compositions provide an efficacy against Rhipicephalus microplus in cattle of at least about 90% for two month, or longer.
- the parasiticidal compositions provide an efficacy against Rhipicephalus microplus in cattle of at least about 90% for three month, or longer.
- Example 1 An experiment was performed to measure the effect the enantiomers and the racemic mixture of an isoxazoline compound (Compound A) has on the weight of laboratory mice for 14 days following the administration of the isoxazoline compound.
- the weight of the rats was then monitored for 14 days.
- Fig. 1 depicts the change in weight of the rats over the 14 day period. Rats that were administered the formulation containing the (S) enantiomer (Formulation C) experienced approximately the same weight gain as those rats that were administered the control. Rats that were administered the formulations containing either (R) enantiomer or the racemic mixture experienced weight loss, thereby indicating that the unwanted effect (weight loss) resided in the (R) enantiomer.
- Example 2 Efficacy and Acceptability of Injectable Formulation Against Rhipicephalus (Boophilus) microplus ticks.
- compositions of the invention comprising the compounds of formula (Ie) and (S)-le shown below, against Rhipicephalus microplus ticks on cattle was determined against an untreated control group.
- the efficacy of compositions comprising the compounds (Ie) and (S)-e were also compared with injectable compositions comprising afoxolaner (formula la, 4-[5-[3-chloro-5-(trifluoromethyl)phenyl]-4,5- dihydro-5-(trifluoromethyl)-3-isoxazolyl]-N-[2-oxo-2-[(2,2,2-trifluoroethyl)amino]ethyl]-l- naphthalanecarboxamide), and comparative compounds of formulae (If) and (S)-If shown (If)
- Each compound was formulated in an injectable composition at a concentration of 10% (w/v) in a carrier comprising 8% (w/v) ethanol and Q.S. with polyethylene glycol 400.
- the racemic compounds afoxolaner and (If) were dosed at 1.0 mg/kg body weight and the (S)- enantiomers ⁇ (S)-le and (S)-If) were dosed at 0.5 mg/kg body weight.
- Six healthy head of cattle were used in each study group. Cattle in Group 1 were untreated (control).
- Cattle in Groups 2, 3, 4, 5 and 6 were treated on Day 0 subcutaneously with injectable compositions comprising the compounds of formula Ie, (S)-If, (S)-Ie, afoxolaner (la) and (If), respectively.
- injectable compositions comprising the compounds of formula Ie, (S)-If, (S)-Ie, afoxolaner (la) and (If), respectively.
- Several weeks before treatment cattle were infested three times a week with approximately 2500 Rhipicephalus microplus larvae to establish ongoing infestations. Each animal was challenged by infestation with approximately 5000 R. microplus larvae on Days 7 and 21 and every 14 days thereafter. Ticks dropping from each animal in the previous 24 hours were collected daily and counted from Day 1 until the end of the study. The cattle in study Groups 3, 5 and 6 were not infested further when the efficacy of the treatment dropped significantly.
- Tables 1A, IB and 1C below show the total tick count % efficacy of injectable compositions comprising compounds of the invention ((Ie) and (S)-Ie) against R. microplus for selected days through Day 110 post treatment compared with the untreated control group and the comparison isoxazoline compounds.
- the compositions comprising the compounds (Ie) and (S)-le of the invention dosed at 1.0 mg/kg and 0.5 mg/kg, respectively provide superior efficacy against Rhipicephalus microplus ticks for an extended period of time compared with compositions comprising afoxolaner, (If) or (SJ- ⁇ .
- the efficacy of compounds (Ie) and (S)-le was also found to be faster-acting than compositions comprising the other isoxazoline compounds.
- Table 1A Tick Count Efficacy Against Rhipicephalus microplus
- Group 2 (Ie) 100 100 100 100 100 100 100 100 100 100 100 100
- Table 1C Tick Count Efficacy against Rhipicephalus microplus
- Group 2 (Ie) 100 100 93 82 Group 3 ((S)-If) - -
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Engineering & Computer Science (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Agronomy & Crop Science (AREA)
- Plant Pathology (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Tropical Medicine & Parasitology (AREA)
- Dermatology (AREA)
- Organic Chemistry (AREA)
- Pest Control & Pesticides (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Toxicology (AREA)
- Dentistry (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present intention provides for, inter alia, a method for reducing a withdrawal period when treating livestock for parasitic infestations, which comprises administering an effective amount of a parasiticidal composition comprising an (S) enantiomer of an isoxazoline compound of the formula (S)-1 or a pharmaceutically acceptable salt thereof, wherein the variables are defined herein, and an inert carrier or formulation adjuvant; wherein the composition contains less than 10% by weight of an (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof. Moreover, the present invention further provides for method for reducing at least one unwanted effect while treating a non-human animal for a parasitic infestation by administering an effective amount of a parasiticidal composition comprising an (S) enantiomer of an isoxazoline compound of the formula (S)-l or a pharmaceutically acceptable salt thereof and an inert carrier or formulation adjuvant, wherein the composition contains less than 10% by weight of an (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof.
Description
TITLE OF THE INVENTION
METHOD FOR REDUCING UNWANTED EFFECTS IN PARA SITICID AL TREATMENTS
CROSS REFERENCE TO RELATED APPLICATIONS
This application claims the benefit of U.S. Provisional Application No. 62/379,345 filed on August 25, 2016, which is herein incorporated by reference in its entirety.
FIELD OF THE INVENTION
The present invention provides for a method for reducing the withdrawal period when treating food-producing animals, including livestock, for parasitic infestations or infections (including ectoparasites (e.g., fleas or ticks) and/or endoparasites) with a parasiticidal composition comprising an isoxazoline compound enriched in the (^-enantiomer, or a pharmaceutically acceptable salt thereof, and an inert carrier or formulation diluent. The present invention also provides for methods for reducing at least one unwanted effect while treating an animal to control or prevent a parasitic infestation with a parasiticidal composition comprising an isoxazoline compound or a pharmaceutically acceptable salt thereof and an inert carrier or formulation diluent.
BACKGROUND OF THE INVENTION
Animals such as mammals and birds are often susceptible to parasite infestations/infections. These parasites may be ectoparasites, such as fleas, ticks and parasitic flies, and endoparasites such as nematodes and other worms. Domesticated animals, such as cats and dogs, are often infested with one or more of the following ectoparasites:
- fleas (e.g. Ctenocephalides spp., such as Ctenocephalides felis and the like);
- ticks (e.g. Rhipicephalus spp., Ixodes spp., Dermacentor spp., Amblyomma spp., and the like);
- mites (e.g. Demodex spp., Sarcoptes spp., Otodectes spp., and the like);
- lice (e.g. Trichodectes spp., Cheyletiella spp., Linognathus spp. and the like);
- mosquitoes (Aedes spp., Culex spp., Anopheles spp. and the like); and
- flies (Haematobia spp., Musca spp., Stomoxys spp., Dermatobia spp., Cochliomyia spp. and the like).
Fleas are a particular problem because not only do they adversely affect the health of the
animal or human, but they also cause a great deal of psychological stress. Moreover, fleas may also transmit pathogenic agents to animals and humans, such as tapeworm {Dipylidium caninum).
Similarly, ticks are also harmful to the physical and psychological health of the animal or human. However, the most serious problem associated with ticks is that they are vectors of pathogenic agents in both humans and animals. Major diseases which may be transmitted by ticks include borreliosis (Lyme disease caused by Borrelia burgdorferi), babesiosis (or piroplasmosis caused by Babesia spp.) and rickettsioses (e.g. Rocky Mountain spotted fever). Ticks also release toxins which cause inflammation or paralysis in the host. Occasionally, these toxins are fatal to the host.
Mosquitoes act as vectors for a number of viral and protozoal infections in the world. It has been reported that mosquitoes carry diseases that potentially compromise the health of one- eighth of the world's population and impact the health and economy of hundreds of millions of people per year. In addition to clinically significant diseases like malaria and yellow fever, mosquitoes are also implicated in the transmission of many arboviruses, infections with which cause symptoms ranging from nonspecific flu-like illnesses to encephalitis that may result in death. In addition to humans, domesticated animals such as dogs, cats, horses, cattle, sheep, pigs, as well as wild animals and birds may become infected and experience clinical disease as a result of that infection. For example, arboviruses that infect humans and other mammals are members of one of three virus families, Togaviridae, Flavividae, and Bunyaviridae, and include the agent responsible for Eastern Equine Encephalitis (EEE), Western Equine Encephalitis (WEE), St. Louis Encephalitis (SLE), La Crosse Encephalitis (LCE), and infection caused by Dengue Virus, and West Nile Virus (WNV). Worldwide, other arboviruses of medical importance include viruses responsible for Japanese Encephalitis (JE) and Venezuelan Equine Encephalitis.
Likewise, farm animals are also susceptible to parasite infestations. For example, cattle are affected by a large number of parasites. A parasite which is prevalent among cattle in some regions is the tick of the genus Rhipicephalus, especially those of the species microplus (cattle tick), decoloratus and annulatus. Ticks such as Rhipicephalus microplus ( ormerly Boophilus microplus) are difficult to control because they lay eggs in the pasture where farm animals graze. This species of ticks is considered a one-host tick and spends immature and adult stages on one
animal before the female engorges and falls off the host to lay eggs in the environment. The life cycle of the tick is approximately three to four weeks. In addition to cattle, Rhipicephalus microplus may infest buffalo, horses, donkeys, goats, sheep, deer, pigs, and dogs. A heavy tick burden on animals can decrease production and damage hides as well as transmit diseases such as babesiosis ("cattle fever") and anaplasmosis. A factor when treating food producing animals, such as cattle or sheep, is the time period between the last drug treatment and when the animals are slaughtered to enter the human food chain ("withdrawal period"). The FDA establishes withdrawal periods as part of the approval process to specify the required time needed after the last administration of a bioactive agent or agents to assure that drug residues in the marketable edible tissues, milk or eggs of food producing animals are below a predetermined and regulated residue tolerance level. Hence, reducing the withdrawal period has advantageous economic advantages for the rancher and farmer.
Animals and humans also suffer from endoparasitic infections including, for example, helminthiasis, which are caused by of parasitic worms categorized as cestodes (tapeworm), nematodes (roundworm) and trematodes (flatworm or flukes). These parasites adversely affect the nutrition of the animal and cause severe economic losses in pigs, sheep, horses, and cattle as well as affecting companion animals and poultry. Other parasites which occur in the gastrointestinal tract of animals and humans include those from the genus Ancylostoma, Necator, Ascaris, Strongyloides, Trichinella, Capillaria, Toxocara, Toxascaris, Trichuris, Enterobius and parasites which are found in the blood or other tissues and organs such as filarial worms and the extra intestinal stages of Strongyloides, Toxocara and Trichinella.
Recently, isoxazole and isoxazoline-containing compounds have been demonstrated to be effective against parasites that harm animals. For example, US 7,964,204 (to DuPont, incorporated by reference herein in its entirety) discloses isoxazoline compounds according to Formula (I) below, which are active against ectoparasites and/or endoparasites.
In addition, published patent application nos. US 2010/0254960 Al, WO 2007/070606 A2, WO 2007/123855 A2, WO 2010/003923 Al, US 7,951,828 & US 7,662,972, US 2010/0137372 Al, US 2010/0179194 A2, US 2011/0086886 A2, US 2011/0059988 Al, US 2010/0179195 Al and WO 2007/075459 A2 and U.S. Patent Nos. 7,951,828 and 7,662,972 describe various other parasiticidal isoxazoline compounds. Other published patent applications that describe various other parasiticidal isoxazoline compounds and formulations comprising the same include WO 2007/079162 Al, WO 2008/154528 Al, WO 2009/002809 A2, WO 2011/149749 Al, WO 2014/439475 Al, US 8,466,115, WO 2012/120399, WO 2014/039484, WO 2014/189837, (Zoetis) and WO2012 120135A1 (Novartis). WO 2012/089623 describes topical localized isoxazoline formulations comprising glycofurol. WO 2013/039948 Al provides for topical veterinary compositions comprising at least one isoxazoline active agent and WO 2013/119442 Al provides for oral veterinary compositions such as a soft chew, which comprise at least one isoxazoline active agent. All the patent documents referred to above are incorporated herein by reference in their entirety.
In addition to topical and oral dosage forms, it is sometimes possible to formulate active agents as long acting injectable formulations or as extended release injectable formulations, depending upon, for example, the physiochemical properties of the individual active agent; these properties include, for example, solubility, bioavailability, etc. Examples of long acting injectable formulations comprising a bioactive agent are described in US 6,733,767 and US 8,362,086, which provide for long acting injectable formulations comprising, for example an avermectin or milbemycin and a biological acceptable polymer. Application PCT/US2016/19688, published as WO 2016/138339, provides for long acting injectable formulations wherein the bioactive agent is an isoxazoline compound. Examples of extended release injectable formulations are described, for example in Application PCT/US2016/026253, which is published as WO 2016/164487. Both these applications are incorporated by reference.
Isoxazoline compounds achieve their parasiticidal effect by inhibiting the GABA-gated chloride-channels of insects, thereby causing hyper-excitation, which leads to death. Isoxazoline compounds are also known potentially to interact with GABA-gated chloride-channels and other off-target receptors in mammals, which may, in turn, lead to unwanted health effects. These off- target sites are typically screened for in rodent and human in vitro pharmacological assays. Afoxolaner, for example, was shown to interact with calcium channel L-type receptors, Na
channel site 2 receptors, norepinephrine cellular transport receptors, dopamine cellular transport receptors, CB-1 receptors, adenosine a3 receptors, leukotriene, cysteinyl Cys T receptors, Histamine 2 receptors, sodium channel site 2 receptors. Histamine H2 receptor binding has been associated with neurological disorders such as seizures, convulsions, restlessness, muscle tremors, insomnia, nervousness, delirium, depression, mydriasis, hyperesthesia. CB-1 receptor binding affects food intake. Gomez, et al, J. Neurosci., 22(21), 9612-9617 (2002).
The safety of residues of veterinary drugs in human food is most commonly addressed through the conduct of toxicology studies in laboratory animal species that provide for the determination of a no-observed-adverse-effect level (NOAEL) and an acceptable daily intake (ADI) by application of appropriate safety/uncertainty factors. The ADI, generally expressed as microgram ^g) or milligram (mg)/kg body weight per day, is defined as the daily intake of drug residue which, for up to an entire lifetime, appears to be without adverse effects or harm to the health of the consumer. The ADI is then used to determine safe concentrations of drug residue in edible tissues and tissue tolerance levels. When combined with tissue residue depletion data, an appropriate product withdrawal period can be determined.
Notwithstanding the compositions comprising isoxazoline bioactive agents alone or in combination with other bioactive agents, there is a need to reduce the withdrawal period when treating food producing animals for parasitic infestations by administering parasiticidal compositions comprising at least one isoxazoline bioactive agent to said animals. Moreover, there is a need to reduce the unwanted effects that can be observed when treating animals for a parasitic infestation by administering parasiticidal compositions comprising at least one isoxazoline bioactive agent to said animals. There is also a need to reduce unwanted effects when treating humans to control or protect them from parasites that act as vectors for viral, bacterial and protozoal infections (e.g. mosquitoes and ticks, among others). Further, there is the need to reduce withdraw period or to r reduce unwanted health effects when treating parasitic infestation in nonhuman animals wherein the parasiticidal composition exhibits superior bioavailability and pharmacokinetics, thereby increasing the efficacy of the product for a longer duration of time. This invention addresses these needs.
INCORPORATION BY REFERENCE Any foregoing applications, and all documents cited therein or during their prosecution
("application cited documents") and all documents cited or referenced in the application cited documents, and all documents cited or referenced herein ("herein cited documents"), and all documents cited or referenced in herein cited documents, together with any manufacturer's instructions, descriptions, product specifications, and product sheets for any products mentioned herein or in any document incorporated by reference herein, are hereby incorporated herein by reference, and may be employed in the practice of the invention.
Citation or identification of any document in this application is not an admission that such document is available as prior art to the present invention.
SUMMARY OF THE INVENTION The present invention provides for a method for the treatment or prevention of a parasitic infestation in an animal with a parasiticidal pesticidal isoxazoline compounds that results in an improved toxicological/safety profile for the animal treated, and/or a lower toxicological residue, and/or when the animal treated is a livestock or milk-producing animal including cattle, a shorter withdrawal period, relative to the effect observed with administration of the racemic compound or the (R)-enantiomer of the compound.
In one embodiment, the present invention provides for a method for reducing at least one unwanted effect while treating an animal for parasitic infestations, which comprises administering an effective amount of an (^-enantiomer of an isoxazoline compound, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S)- enantiomer of the isoxazoline compound or salt and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)-enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof. In one embodiment, the unwanted effect is an unfavorable injection site reaction related to the injection of a composition comprising a parasiticidal isoxazoline compound to an animal. While not wishing to be bound by theory, most or possibly all of the pesticidal activity resides in (^-enantiomer. Hence, one may be able to administer less of the active compound (e.g. approximately half the dose) to the animal to achieve the intended effect against parasites, thereby significantly reducing unfavorable effects at the site of the injection.
In another embodiment, the present invention provides for, inter alia, a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the
transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an (S)-enantiomer of an isoxazoline compound, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (^-enantiomer of the isoxazoline compound or salt thereof and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)-enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
Further, the present invention provides a method for reducing at least one unwanted effect while treating an animal for parasitic infestations with an isoxazoline compound, or a pharmaceutically acceptable salt thereof, by reducing the binding of the isoxazoline compound to an off-target receptor, which comprises administering to said animal an effective amount of an (S) enantiomer of the isoxazoline compound, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising an effective amount of the (^-enantiomer of the isoxazoline compound, or a pharmaceutically acceptable salt thereof, and an inert carrier or formulation diluent wherein the composition contains less than about 10% by weight of the (R) enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
The present invention provides for a method for reducing a withdrawal period when treating food-producing animals, including livestock, for parasitic infestations, which comprises administering an effective amount of the (^-enantiomer of an isoxazoline compound, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S) enantiomer of the isoxazoline compound, or a pharmaceutically acceptable salt thereof, and an inert carrier or formulation diluent, wherein the composition contains less than about 10% by weight of the (R) enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
Moreover, the present invention further provides for a method for treating or preventing an ectoparasitic infestation in an animal in need thereof while reducing at least one unwanted effect, which comprises administering an effective amount of an (^-enantiomer of an isoxazoline compound, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S) enantiomer of an isoxazoline compound, or a pharmaceutically acceptable salt thereof, and an inert carrier or formulation diluent, wherein the composition contains less than about 10% by weight of the (R) enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof; and wherein said parasiticidal is effective against about
provides methods for treating or preventing ectoparasitic infestations with an efficacy of about at least 90% against said ectoparasites for at least 2 months, at least 3 months, at least 4 moths, at least 5 months, or 6 months.
In accordance with the present invention, it has been discovered that one or more of the unwanted toxicological effects in rats elicited by the racemic isoxazoline compound afoxolaner surprisingly reside in one enantiomer (viz., the (R)-enantiomer), which is typically not the situation. Hence, administering a parasiticidal composition comprising an (^-enantiomer of a parasiticidal isoxazoline compound in which the unwanted effect resides in the (R)-enantiomer and an inert carrier or formulation diluent wherein the composition contains less than about 10% by weight of the corresponding (R)-enantiomer of the isoxazoline compound, or a pharmaceutically acceptable salt thereof, will increase the safety profile of the parasiticidal compositions by reducing at least one unwanted effect by, for example, reducing off-target receptor binding, thereby, increasing the NOAEL and ADI levels and allowing one to reduce the withdrawal period. In another embodiment, the methods of the invention allow for the reduction of unwanted effects at the injection site when the composition is administered by injection.
Accordingly, it is an object of the invention to not encompass within the invention any previously known product, process of making the product, or method of using the product such that Applicants reserve the right and hereby disclose a disclaimer of any previously known product, process, or method. It is further noted that the invention does not intend to encompass within the scope of the invention any product, process, or making of the product or method of using the product, which does not meet the written description and enablement requirements of the USPTO (35 U.S.C. §112, first paragraph) or the EPO (Article 83 of the EPC), such that Applicants reserve the right and hereby disclose a disclaimer of any previously described product, process of making the product, or method of using the product.
It is noted that in this disclosure and particularly in the claims and/or paragraphs, terms such as "comprises", "comprised", "comprising" and the like can have the meaning attributed to it in U.S. Patent law; e.g., they can mean "includes", "included", "including", and the like; and that terms such as "consisting essentially of and "consists essentially of have the meaning ascribed to them in U.S. Patent law, e.g., they allow for elements not explicitly recited, but
exclude elements that are found in the prior art or that affect a basic or novel characteristic of the invention.
These and other embodiments are disclosed or are obvious from and encompassed by, the following Detailed Description.
BRIEF DESCRIPTION OF THE DRAWINGS
The following detailed description, given by way of example, but not intended to limit the invention solely to the specific embodiments described, may best be understood in conjunction with the accompanying drawings.
Figure 1 depicts the mean body weight on female rats for 14 days following the administration of Formulation A (100 mg of the (R) enantiomer compound A dissolved in the carrier), Formulation B (200 mg of the racemic mixture of compound A dissolved the carrier); Formulation C (500 mg of the (S) enantiomer of the compound A dissolved in the carrier); and Control (carrier).
DETAILED DESCRIPTION
An embodiment of the present invention provides for a method for reducing a withdrawal period when treating food-producing animals, including livestock, for parasitic infestations, which comprises administering to the animal an effective amount of an (S) enantiomer of an isoxazoline compound of the formula (S)-l :
(5 1 or a pharmaceutically acceptable salt thereof or a parasiticidal composition compri effective amount of the compound or pharmaceutically acceptable salt thereof, wherein:
B1, B2, B3, are each independently C-R or N;
each R is independently H, halogen, cyano, -N02, alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl;
Y is an optionally substituted phenylene, naphthylene, indanylene, a 5- or 6-membered heteroarylene or an 8-10-membered fused heterobicyclylene, wherein the optional substituents are selected from the group consisting of halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino, — CN or — Ν02 and NH2- C(=S)-;
Q is X- R2R3, the group (-CH2-)(-CH2-)N-R3, OH, H2, alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
X is (CH2)n, CH(CH3), CH(CN), C(=0) or C(=S);
R2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R3 is H, OR7, R8R9 or Q1; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R4; or
R2 and R3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N02 and alkoxy;
each R4 is independently halogen; alkyl, cycloalkyl, alkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, haloalkylamino, dialkylamino, dihaloalkylamino, cycloalkylamino, alkylcarbonyl, alkoxycarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, haloalkylcarbonyl, haloalkoxycarbonyl, haloalkylaminocarbonyl, dihaloalkylaminocarbonyl, hydroxy,— NH2,— CN or— N02; or Q2;
each R5 is independently halogen, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino, alkoxycarbonyl,— CN or— N02;
each R6 is independently halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN,— N02, phenyl or pyridinyl;
R7 is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
R8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R9 is H; Q3; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R4; or
R8 and R9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N02 and alkoxy;
Q1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R5;
Q2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6;
Q3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6; and
n is 0, 1 or 2; and an inert carrier or formulation adjuvant;
wherein the compound or composition contains less than about 10% by weight (e.g., less than about 5% by weight, less than about 2% by weight, or less than about 1% by weight) of the (R) enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
Another embodiment of the present invention is a method for reducing at least one unwanted effect while treating an animal for a parasitic infestation, which comprises administering to the animal an effective amount of an (S) enantiomer of an isoxazoline compound of formula (S)-l, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S) enantiomer of the isoxazoline compound of the formula (S)-l or pharmaceutically acceptable salt thereof,
(S)-l wherein:
B1, B2, B3, are each independently C-R or N; each R is independently H, halogen, cyano, -N02, alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl;
Y is an optionally substituted phenylene, naphthylene, indanylene, a 5- or 6-membered heteroarylene or an 8-10-membered fused heterobicyclylene, wherein the optional substituents are selected from the group consisting of halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN or— N02 and H2-C(=S)-;
Q is X- R2R3, the group (-CH2-)(-CH2-)N-R3, OH, H2, alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
X is (CH2)n, CH(CH3), CH(CN), C(=0) or C(=S);
R2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R3 is H, OR7, R8R9 or Q1; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R4; or
R2 and R3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— Ν02 and alkoxy; each R4 is independently halogen; alkyl, cycloalkyl, alkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, haloalkylamino, dialkylamino, dihaloalkylamino, cycloalkylamino, alkylcarbonyl, alkoxycarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, haloalkylcarbonyl, haloalkoxycarbonyl, haloalkylaminocarbonyl, dihaloalkylaminocarbonyl, hydroxy,— NH2,— CN or— N02; or Q2; each R5 is independently halogen, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino, alkoxycarbonyl, — CN or— N02; each R6 is independently halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN,— N02, phenyl or pyridinyl;
R7 is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
R8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R9 is H; Q3; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R4; or
R8 and R9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N02 and alkoxy; Q1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R5;
Q2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6;
Q3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6; and n is 0, 1 or 2; and an inert carrier or formulation diluent, wherein the compound or composition contains less than about 10% by weight (e.g., less than about 5% by weight, less than about 2% by weight, or less than about 1% by weight) of the (R) enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
In one embodiment, the present invention provides a method for reducing at least one unwanted effect while treating an animal (including a human) to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an (^-enantiomer of an isoxazoline compound of formula (S)-l, as described above, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (^-enantiomer of the isoxazoline compound of formula (S)-l, or pharmaceutically acceptable salt thereof, and an inert carrier or a formulation diluent, wherein the compound or composition contains less than about 10% by weight of the (R)-enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
Another embodiment of the present invention is a method for reducing at least one unwanted effect while treating an animal for a parasitic infestation with an isoxazoline
compound, or a pharmaceutically acceptable salt thereof, by reducing the binding of the isoxazoline compound to an off-target receptor, which comprises administering to said animal the (^-enantiomer of the isoxazoline compound of formula (S)-l, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising an effective amount of the (S)- enantiomer of an isoxazoline compound of the formula (S)-l, or pharmaceutically acceptable salt thereof:
(S)-l wherein:
B1, B2, B3, are each independently C-R or N; each R is independently H, halogen, cyano, -N02, alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl;
Y is an optionally substituted phenylene, naphthylene, indanylene, a 5- or 6-membered heteroarylene or an 8-10-membered fused heterobicyclylene, wherein the optional substituents are selected from the group consisting of halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN or— N02 and H2-C(=S)-;
Q is X- R2R3, the group (-CH2-)(-CH2-)N-R3, OH, H2, alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
X is (CH2)n, CH(CH3), CH(CN), C(=0) or C(=S);
R2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R3 is H, OR7, R8R9 or Q1; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R4; or
R2 and R3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— Ν02 and alkoxy; each R4 is independently halogen; alkyl, cycloalkyl, alkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, haloalkylamino, dialkylamino, dihaloalkylamino, cycloalkylamino, alkylcarbonyl, alkoxycarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, haloalkylcarbonyl, haloalkoxycarbonyl, haloalkylaminocarbonyl, dihaloalkylaminocarbonyl, hydroxy,— NH2,— CN or— N02; or Q2; each R5 is independently halogen, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino, alkoxycarbonyl, — CN or— N02; each R6 is independently halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN,— N02, phenyl or pyridinyl;
R7 is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
R8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R9 is H; Q3; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R4; or
R8 and R9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N02 and alkoxy; Q1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R5;
Q2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6;
Q3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6; and
n is 0, 1 or 2;
and an inert carrier or formulation adjuvant;
wherein the parasiticidal compound or composition contains less than about 10% by weight (e.g., less than about 5% by weight, less than about 2% by weight, or less than about 1% by weight) of an (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof.
Another embodiment of the present invention provides for a method for treating or preventing a parasitic infestation in an animal in need thereof while reducing at least one unwanted effect, which comprises administering an effective amount of an (S) enantiomer of an isoxazoline compound of formula (S)-l, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S) enantiomer of an isoxazoline compound of the formula (S)-l, or a pharmaceutically acceptable salt thereof:
(S)-l
wherein:
B1, B2, B3, are each independently C-R or N; each R is independently H, halogen, cyano, -N02, alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl;
Y is an optionally substituted phenylene, naphthylene, indanylene, a 5- or 6-membered heteroarylene or an 8-10-membered fused heterobicyclylene, wherein the optional substituents are selected from the group consisting of halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN or— Ν02 and H2-C(=S)-;
Q is X- R2R3, the group (-CH2-)(-CH2-)N-R3, OH, H2, alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
X is (CH2)n, CH(CH3), CH(CN), C(=0) or C(=S);
R2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R3 is H, OR7, R8R9 or Q1; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R4; or
R2 and R3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N02 and alkoxy;
each R4 is independently halogen; alkyl, cycloalkyl, alkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, haloalkylamino, dialkylamino, dihaloalkylamino, cycloalkylamino, alkylcarbonyl, alkoxycarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, haloalkylcarbonyl, haloalkoxycarbonyl, haloalkylaminocarbonyl, dihaloalkylaminocarbonyl, hydroxy,— NH2,— CN or— N02; or Q2; each R5 is independently halogen, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino, alkoxycarbonyl, — CN or— N02; each R6 is independently halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN,— N02, phenyl or pyridinyl;
R7 is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
R8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R9 is H; Q3; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R4; or
R8 and R9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N02 and alkoxy;
Q1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R5;
Q2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6;
Q is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6; and n is 0, 1 or 2; and an inert carrier or formulation adjuvant; wherein the compound or composition contains less than about 10% by weight (e.g., less than about 5% by weight, less than about 2% by weight, or less than about 1% by weight) of the (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof; and wherein said parasiticidal composition is effective against at least 90 % of the fleas and the ticks (e.g., 95%, 96%, 97%, 98%, 99% or 100%)) or for at least for 3 to 6 months (e.g., at least 3, 4, 5 or 6 months).
Another embodiment of the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of an arthropod- or mosquito-borne disease which comprises administering an effective amount of an (S) enantiomer of an isoxazoline compound of formula (S)-l to the animal, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (S) enantiomer of an isoxazoline compound of the formula (S)-l, or a pharmaceutically acceptable salt thereof:
(S)-l wherein: B1, B2, B3, are each independently C-R or N; each R is independently H, halogen, cyano, -N02, alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl;
Y is an optionally substituted phenylene, naphthylene, indanylene, a 5- or 6-membered heteroarylene or an 8-10-membered fused heterobicyclylene, wherein the optional substituents are selected from the group consisting of halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN or— Ν02 and NH2-C(=S)-;
Q is X- R2R3, the group (-CH2-)(-CH2-)N-R3, OH, H2, alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
X is (CH2)n, CH(CH3), CH(CN), C(=0) or C(=S);
R2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxy carbonyl;
R3 is H, OR7, R8R9 or Q1; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R4; or
R2 and R3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N02 and alkoxy; each R4 is independently halogen; alkyl, cycloalkyl, alkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, haloalkylamino, dialkylamino, dihaloalkylamino, cycloalkylamino, alkylcarbonyl, alkoxycarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, haloalkylcarbonyl, haloalkoxycarbonyl, haloalkylaminocarbonyl, dihaloalkylaminocarbonyl, hydroxy,— NH2,— CN or— N02; or Q2;
each R5 is independently halogen, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino, alkoxycarbonyl, — CN or— N02; each R6 is independently halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN,— N02, phenyl or pyridinyl;
R7 is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
R8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R9 is H; Q3; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R4; or
R8 and R9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N02 and alkoxy;
Q1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R5;
Q2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6;
Q3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6; and n is 0, 1 or 2;
and an inert carrier or formulation adjuvant; wherein the compound or composition contains less than about 10% by weight (e.g., less than about 5% by weight, less than about 2% by weight, or less than about 1% by weight) of the (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof; and wherein said parasiticidal composition is effective against at least 90 % of the fleas and the ticks (e.g., 95%, 96%, 97%, 98%, 99% or 100%)) or for at least for 3 to 6 months (e.g., at least 3, 4, 5 or 6 months).
In another embodiment, the invention provides for any of the methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an antiparasitic isoxazoline compound of formula (S -II
(S)-II
wherein:
1 2 3 4 5 6 3
A , Az, A , A", A and A are independently selected from the group consisting of CR and N, provided that at most 3 of A1, A2, A3, A4, A5 and A6 are N;
B1, B2 and B3 are independently selected from the group consisting of CR2 and N;
W is O or S;
each R2 is independently H, halogen, Ci-C6 alkyl, Ci-C6 haloalkyl, Ci-C6 alkoxy, Ci-C6 haloalkoxy, Ci-C6 alkylthio, Ci-C6 haloalkylthio, Ci-C6 alkylsulfinyl, Ci-C6 haloalkylsulfinyl, Ci- C6 alkylsulfonyl, Ci-C6 haloalkylsulfonyl, Ci-C6 alkylamino, C2-C6 dialkylamino, C2-C4 alkoxycarbonyl,— CN or— N02;
each R3 is independently H, halogen, Ci-C6 alkyl, Ci-C6 haloalkyl, C3-C6 cycloalkyl, C3- C6 halocycloalkyl, Ci-C6 alkoxy, Ci-C6 haloalkoxy, Ci-C6 alkylthio, Ci-C6 haloalkylthio, Ci-C6 alkylsulfinyl, Ci-C6 haloalkylsulfinyl, Ci-C6 alkylsulfonyl, Ci-C6 haloalkylsulfonyl, Ci-C6 alkylamino, C2-C6 dialkylamino,— CN or— N02;
R4 is H, Ci-C6 alkyl, C2-C6 alkenyl, C2-C6 alkynyl, C3-C6 cycloalkyl, C4-C7 alkylcycloalkyl, C4-C7 cycloalkylalkyl, C2-C7 alkylcarbonyl or C2-C7 alkoxycarbonyl;
R5 is H, OR10, RUR12 or Q1; or Ci-C6 alkyl, C2-C6 alkenyl, C2-C6 alkynyl, C3-C6 cycloalkyl, C4-C7 alkylcycloalkyl or C4-C7 cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R7; or
R4 and R5 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of Ci-C2 alkyl, halogen,— CN,— N02 and Ci-C2 alkoxy; each R7 is independently halogen; Ci-C6 alkyl, C3-C6 cycloalkyl, Ci-C6 alkoxy, Ci-C6 alkylthio, Ci-C6 alkylsulfinyl, Ci-C6 alkylsulfonyl, Ci-C6 alkylamino, C2-C8 dialkylamino, C3-C6 cycloalkylamino, C2-C7 alkylcarbonyl, C2-C7 alkoxy carbonyl, C2-C7 alkylaminocarbonyl, C3-C9 dialkylaminocarbonyl, C2-C7 haloalkylcarbonyl, C2-C7 haloalkoxycarbonyl, C2-C7 haloalkylaminocarbonyl, C3-Cg dihaloalkylaminocarbonyl, hydroxy,— NH2,— CN or— N02; or
Q2;
each R8 is independently halogen, Ci-C6 alkoxy, Ci-C6 haloalkoxy, Ci-C6 alkylthio, Ci-C6 haloalkylthio, Ci-C6 alkylsulfinyl, Ci-C6 haloalkylsulfinyl, Ci-C6 alkylsulfonyl, Ci-C6 haloalkylsulfonyl, Ci-C6 alkylamino, C2-C6 dialkylamino, C2-C4 alkoxycarbonyl,— CN or— N02;
each R9 is independently halogen, Ci-C6 alkyl, Ci-C6 haloalkyl, C3-C6 cycloalkyl, C3-C6 halocycloalkyl, Ci-C6 alkoxy, Ci-C6 haloalkoxy, Ci-C6 alkylthio, Ci-C6 haloalkylthio, Ci-C6 alkylsulfinyl, Ci-C6 haloalkylsulfinyl, Ci-C6 alkylsulfonyl, Ci-C6 haloalkylsulfonyl, Ci-C6 alkylamino, C2-C6 dialkylamino,— CN,— N02, phenyl or pyridinyl;
R10 is H; or Ci-C6 alkyl, C2-C6 alkenyl, C2-C6 alkynyl, C3-C6 cycloalkyl, C4-C7 alkylcycloalkyl or C4-C7 cycloalkylalkyl, each optionally substituted with one of more halogen;
R11 is H, Ci-C6 alkyl, C2-C6 alkenyl, C2-C6 alkynyl, C3-C6 cycloalkyl, C4-C7 alkylcycloalkyl, C4-C7 cycloalkylalkyl, C2-C7 alkylcarbonyl or C2-C7 alkoxycarbonyl;
R12 is H; Q3; or Ci-C6 alkyl, C2-C6 alkenyl, C2-C6 alkynyl, C3-C6 cycloalkyl, C4-C7 alkylcycloalkyl or C4-C7 cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R7; or
R11 and R12 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group
consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of C1-C2 alkyl, halogen,— CN,— Ν02 and C1-C2 alkoxy;
Q1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R8;
each Q2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R9;
Q3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R9; and
n is 0, 1 or 2; or a pharmaceutically acceptable salt thereof, or
wherein the parasiticidal composition comprises a compound of formula (S)-II and an inert carrier or formulation adjuvant.
In another embodiment, the invention provides for any of the methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an antiparasitic isoxazoline compound of formula (^-IIA:
(S)-llA,
wherein X1, X2 and X3 are each independently H, bromo, chloro, fluoro, iodo or CF3; or wherein the parasiticidal composition comprises a compound of formula (S)-IIA and an inert carrier or formulation adjuvant.
In other embodiments for any of the inventive methods provided for above, the parasiticidal compound is an isoxazoline compound of formula (^-IIA where X1 is CI, X2 is H and X3 is CF3 ("Compound A"). In another embodiment of the inventive methods, the parasiticidal compound is a compound of (5)-IIA where X1 is CI, X2 is F and X3 is CF3. In another embodiment of the inventive methods, the parasiticidal compound is a compound of
formula (^-IIA where X1 is CI, X2 is H and X3 is CI. In yet another embodiment of the inventive methods the parasiticidal compound is a compound of formula (^-IIA where X1 is CI, X2 is F and X3 is CI.
In another embodiment, the invention provides for any of the inventive methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an antiparasitic isoxazoline compound of formula (S)-III:
(S)-m; wherein X1, X2 and X3 are each independently H, bromo, chloro, fluoro, iodo or CF3, or wherein the parasiticidal composition comprises a compound of formula (S)-III and an inert carrier or formulation adjuvant.
The racemic compound corresponding to formula (S)-III wherein X1 and X3 are chloro and X2 is hydrogen has the international non-proprietary name (INN) fluralaner. Fluralaner is the active ingredient in the product BRAVECTO™ flavored chews for dogs and topical solution for cats. BRAVECTO™ is indicated for the treatment and prevention of flea infestations and certain tick infestations. Recently, the European Medicines Agency published a European public MRL assessment report (EPMAR) for the use of the racemic compound fluralaner for use in poultry for the treatment and control of red poultry mite infestations. Fluralaner is a racemic compound composed of equal amounts of the (R)- and (^-enantiomer of the compound. It is believed that the activity of fluralaner against arthropod pests resides in the (^-enantiomer as with other compounds in this class. Therefore, the methods of the invention described above directed to the treatment of food-producing animals are particularly relevant for the use of the (S)-enantiomer of fluralaner. Use of the (S)-enantiomer of fluralaner would reduce the dose required to control ectoparasites in food-producing animals and thereby reduce the amount of drug residue and/or result in an increased ADI that results in a shorter withdrawal period before the marketable
edible tissues, milk or eggs of food producing animals can be introduced into the human food chain.
In one embodiment of the inventive methods provided for above, the parasiticidal composition comprises an isoxazoline compound of formula (^-III where X1 is CI, X2 is H and X3 is CF3. In another embodiment of the methods, the composition comprises a compound of formula (^-III where X1 is CI, X2 is F and X3 is CF3. In another embodiment of the inventive methods, the composition comprises a compound of formula (^-III where X1 is CI, X2 is H and X3 is CI. In yet another embodiment of the inventive methods, the composition comprises a compound of formula (^-III where X1 is CI, X2 is F and X3 is CI.
In another embodiment, the invention provides a method for reducing a withdrawal period when treating livestock for parasitic infestations, which comprises administering an effective amount of a compound of formula (5)-ΙΠ, wherein X1 is CI, X2 is H and X3 is CI, or a parasiticidal composition comprising the compound.
In another embodiment, the invention provides a method for reducing a withdrawal period when treating cattle for parasitic infestations, which comprises administering an effective amount of a compound of formula (S)-III, wherein X1 is CI, X2 is H and X3 is CI, or a parasiticidal composition comprising the compound.
In another embodiment of the present invention is a method for reducing at least one unwanted effect while treating an animal for a parasitic infestation, which comprises administering an effective amount of a compound of formula (5)-ΠΙ, wherein X1 is CI, X2 is H and X3 is CI, or a parasiticidal composition comprising the compound.
In another embodiment of the present invention, a method for reducing at least one unwanted effect while treating an animal for a parasitic infestation with an isoxazoline compound or a salt thereof by reducing the binding of an isoxazoline compound to an off-target receptor, which comprises administering to said non-human animal an effective amount of a compound of formula (5)-ΙΠ, wherein X1 is CI, X2 is H and X3 is CI, or a parasiticidal composition comprising an effective amount of the compound.
In another embodiment, the present invention provides a method of treating or controlling red poultry mites in chickens comprising administering to the chickens an effective amount of the compound of formula (S)-III, wherein X1 is CI, X2 is H and X3 is CI, in drinking water.
In another embodiment, the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an (^-enantiomer of an isoxazoline compound of formula (5)-ΠΙ, wherein X1 is CI, X2 is H or F and X3 is CI or CF3, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (^-enantiomer of the isoxazoline compound or salt and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)- enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
In another embodiment, the invention provides for any of the inventive methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an antiparasitic isoxazoline compound of formula I:
(5 IV
wherein:
X1, X2 and X3 are each independently H, bromo, chloro, fluoro, iodo or CF3;
G is G-l or G-2;
G-l G-2
Bi, B2, B3, B4 and B5 are independently N or C-R9;
Y is Y-1, Y-2, Y-3, Y-4, Y-5, Y-6, Y-7, Y-8, Y-9, Y-10, Y-1 1, Y-12 or Y-13;
R2, R-3 are independently hydrogen, alkyl, haloalkyl, thioalkyl, alkylthioalkyl, hydroxyalkyl, alkoxylakyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, Ri0S(O)-, RioS(0)2-, RioC(O)-, RioC(S)-, Ri0RnNC(O)-, Ri0RiiNC(S)- RioOC(O)-;
R4, R5 and R6 are independently hydrogen, alkyl, haloalkyl, thioalkyl, alkylthioalkyl, hydroxyalkyl, alkoxylakyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, aryl or heteroaryl;
R7 and R8 are independently hydrogen, alkyl, haloalkyl, thioalkyl, alkylthioalkyl, hydroxyalkyl, alkoxylakyl, alkenyl, haloalkenyl, alkynyl or haloalkynyl;
R9 is hydrogen, halogen, -CN, or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, halocycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each which is unsubstituted or substituted with one or more of halogen, hydroxy, amino, alkyl- or di(alkyl)amino, alkyl, cycloalkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, R7S(0)-, R7S(0)2-, R7C(0)-, R7R8NC(0)-, R7OC(0)-, R7C(0)0-, R7C(0) R8-, -CN or -N02;
Rio, R11, Ri2 and Ri3 are each independently hydrogen, alkyl, haloalkyl, thioalkyl, alkylthioalkyl, hydroxyalkyl, alkoxylakyl, alkenyl, haloalkenyl, alkynyl or haloalkynyl; or
Rio together with Rn form =0, =S or = R2; or
Ri2 together with Ri3 form =0, =S or = R2;
W is O, S or R2;
n is 1-4; and
m is 0, 1 or 2; or a pharmaceutically acceptable salt thereof, or wherein the parasiticidal composition comprises a compound of formula (,S)-IV and an inert carrier or formulation adjuvant.
In another embodiment, the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an (^-enantiomer of an isoxazoline compound of formula (,S)-IV described above, wherein X1 is CI, X2 is H, CI or F and X3 is CI or CF3, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (,S)-enantiomer of the isoxazoline compound, or a pharmaceutically acceptable salt thereof, and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)-enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
In one embodiment of the inventive methods provided for above, the parasiticidal compound or composition comprises an isoxazoline compound of formula (,S)-IV where X1 is CI, X2 is H and X3 is CF3. In another embodiment of the inventive methods, the compound or composition comprises a compound of formula (,S)-IV where X1 is CI, X2 is F and X3 is CF3. In another embodiment of the inventive methods, the compound or composition comprises a compound of formula (,S)-IV where X1 is CI, X2 is H and X3 is CI. In yet another embodiment of the inventive methods, the compound or composition comprises a compound of formula (,S)-IV where X1 is CI, X2 is F and X3 is CI.
In another embodiment, the invention provides for any of the inventive methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an antiparasitic isoxazoline compound of formula IV-1.001 to IV-1.027 or IV-2.001 to IV-2.020:
IV-1.001 to IV-1.027
wherein X1, X2, X3, B1, B2, B3, B4, B5, R15 and R16 are as shown in the table below:
Compounds IV-2.001 to IV-2.020
IV-1.027 or IV-2.001 to IV-2.020 and an inert carrier or formulation adjuvant.
In another embodiment, the invention provides for any of the inventive methods provided for above wherein the parasiticidal compound is an (S) enantiomer of antiparasitic isoxazoline compound of formula (<S)-V:
(S)-V wherein X1, X2 and X3 are each independently H, bromo, chloro, fluoro, iodo or CF3, or a composition comprising a compound of formula (^-Vand an inert carrier or formuation adjuvant.
In one embodiment of the inventive methods provided for above, the parasiticidal composition comprises an isoxazoline compound of formula (S)-V where X1 is CI, X2 is H and X3 is CF3. In another embodiment of the inventive methods, the composition comprises a compound of formula (S)-V where X1 is CI, X2 is F and X3 is CF3. In another embodiment of the inventive methods, the composition comprises a compound of formula (S)-V where X1 is CI, X2 is H and X3 is CI. In yet another embodiment of the inventive methods the composition comprises a compound of formula (S)-V where X1 is CI, X2 is F and X3 is CI. In yet another embodiment of the method, the composition comprises a compound of formula (S)-V where X1, X2 and X3 are each CI.
In another embodiment, the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an (^-enantiomer of an isoxazoline compound of formula (S)-V, wherein X1 is CI, X2 is H, CI or F and X3 is CI or CF3, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (^-enantiomer of the isoxazoline compound or salt and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)- enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
In another embodiment, the invention provides for any of the embodiments of the methods provided for above wherein the parasiticidal compound is an (S) enantiomer of an isoxazoline compound of the formula
(5 VI wherein X1, X2 and X3 are each independently H, bromo, chloro, fluoro, iodo or CF3, or wherein the parasiticidal composition comprises a compound of formula (5)- VI and an inert carrier or formulation adjuvant.
In one embodiment of the inventive methods provided for above, the parasiticidal composition comprises an isoxazoline compound of formula (5)- VI where X1 is CI, X2 is H and X3 is CF3. In another embodiment of the inventive methods, the composition comprises a compound of formula (5)- VI where X1 is CI, X2 is F and X3 is CF3. In another embodiment of the inventive methods, the composition comprises a compound of formula (5)- VI where X1 is CI, X2 is H and X3 is CI. In yet another embodiment of the inventive methods, the composition comprises a compound of formula (5)- VI where X1 is CI, X2 is F and X3 is CI. In still another embodiment of the inventive methods, the composition comprises a compound of formula (S)-VI where each of X1, X2 and X3 is CI.
In another embodiment, the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an (^-enantiomer of an isoxazoline compound of formula (S)-VL, wherein X1 is CI, X2 is H, CI or F and X3 is CI or CF3, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (^-enantiomer of the isoxazoline compound or salt and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)- enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
In another embodiment, the invention provides for any of the embodiments of the inventive methods provided for above wherein the compound is an (S) enantiomer of an isoxazoline compound of the formula
(S)- VII wherein X1, X2 and X3 are each independently H, bromo, chloro, fluoro, iodo or CF3, or wherein the composition comprises a compound of formula (,S)-VII and an inert carrier or formulation adjuvant.
In one embodiment of the inventive methods provided for above, the parasiticidal composition comprises an isoxazoline compound of formula (,S)-VII where X1 is CI, X2 is H and X3 is CF3. In another embodiment of the inventive methods, the compositions comprise a compound of formula (,S)-VII where X1 is CI, X2 is F and X3 is CF3. In another embodiment of the inventive methods, the composition comprises a compound of formula (,S)-VII where X1 is CI, X2 is H and X3 is CI. In yet another embodiment of the inventive methods, the composition comprises a compound of formula (,S)-VII where X1 is CI, X2 is F and X3 is CI. In yet another embodiment of the inventive methods the composition comprises a compound of formula (S)- VII, X1, X2 and X3 are each CI.
In another embodiment, the present invention provides for a method for reducing at least one unwanted effect while treating an animal to prevent or interrupt the transmission of arthropod- or mosquito-borne diseases which comprises administering an effective amount of an (^-enantiomer of an isoxazoline compound of formula (S)-VII, wherein X1 is CI, X2 is H, CI or F and X3 is CI or CF3, or a pharmaceutically acceptable salt thereof, or a parasiticidal composition comprising the (^-enantiomer of the isoxazoline compound or salt and an inert carrier or a formulation diluent, wherein the composition contains less than about 10% by weight of the (R)-enantiomer of the isoxazoline compound or a pharmaceutically acceptable salt thereof.
In one embodiment of the inventive methods provided for above, the parasiticidal compositions is substantially free of an (R) enantiomer of the isoxazoline compound.
In another embodiment of the inventive methods provided for above, the parasiticidal composition comprises no (R) enantiomer of an isoxazoline compound.
In one embodiment the withdrawal period is reduced by about 30 to 70%, for example about 40-60%) or about 45-55%> (e.g., about 50%>) over a 40 to 60 day withdrawal period calculated for administration of the racemic isoxazoline compound.
In another embodiment the withdrawal period is reduced by about 30 to 70%, for example about 40-60%> or about 45-55%> (e.g., about 50%>) over an 80 to 100 day withdrawal period calculated for administration of the racemic isoxazoline compound.
In one embodiment of the inventive methods provided for above, the parasiticidal compositions are oral formulations, such as, for example, a tablet, a paste, a chewable tablet or a soft chew.
In other embodiments of the inventive methods provided for above, the parasiticidal compositions are topical formulations, such as, for example, a spot-on or pour-on.
In other embodiments of the inventive methods provided for above, the parasiticidal composition is dissolved in the drinking water.
In a further embodiment of the inventive methods provided for above, the parasiticidal composition is an injectable formulation, such as, for example an long acting injectable formulation or an extended release injectable formulation.
In one embodiment the unwanted effects that may be reduced by the inventive methods include neurological disorders, such as seizures, convulsions, restlessness, muscle tremors, insomnia, nervousness, delirium, depression, mydriasis and hyperesthesia. In another embodiment, the unwanted effect is weight loss in the animal during treatment (e.g., the animal does not lose more that 10%> of its body weight before treatment). Another embodiment the unwanted effects are vomiting, dry or flaky skin, diarrhea, lethargy or anorexia or a combination of the foregoing.
In one embodiment the off-site receptors included by the inventive methods are: Sodium Channel, Site 2; Calcium Channel L-Type, Dihydropyridine; Transporter, Norepinephrine (NET);Transporter, Dopamine (DAT); Leukotriene, Cysteinyl CysLTl ; Dopamine Dl ; GABAA, Chloride Channel, TBOB; Adenosine A3; Cannabinoid CB 1 ; Calcium Channel L-Type, Benzothiazepine; Dopamine D3; Adrenergic alpha2A; Opiate mu (OP3, MOP); Serotonin (5- Hydroxytryptamine) 5-HT2B; Adrenergic betal ; Adrenergic alphalA; Prostanoid EP4;
Glucocorticoid; Muscarinic M2; Potassium Channel hERG; Thyroid Hormone; Muscarinic Ml; Serotonin (5-Hydroxytryptamine) 5-HT1A; Melatonin MT1; Platelet Activating Factor (PAF); Muscarinic M3; Sigmal; Epidermal Growth Factor (EGF); Rolipram; Adenosine Al; Phorbol Ester; Purinergic P2Y; Androgen (Testosterone); Potassium Channel [SKCA]; Adrenergic alphalB; Glutamate, NMDA, Agonism; Histamine HI; Dopamine D2S; Estrogen ERalpha; Glutamate, NMDA, Polyamine; Purinergic P2X; Adenosine A2A; Bradykinin Bl; Dopamine D4.2; GABABIB; Adrenergic alphalD; GABAA, Flunitrazepam, Central; Opiate kappa (OP2, KOP); Potassium Channel [KATP]; Potassium Channel [KA]; Transporter, GAB A; GABAA, Muscimol, Central; Imidazoline 12, Central; Endothelin ETB; GABABIA; Opiate deltal (OP1, DOP); Thyrotropin Releasing Hormone (TRH); Glutamate, NMDA, Phencyclidine; Histamine H3; Tachykinin NK1; Adrenergic beta2; Glutamate, Kainate; Transporter, Serotonin (5- Hydroxytryptamine) (SERT); Retinoid X Receptor RXRalpha; Interleukin IL-1; Endothelin ETA; Glutamate, NMDA, Glycine; Neuropeptide Y Y2; Calcium Channel N-Type; Bradykinin B2; Nicotinic Acetylcholine; Histamine H2; Nicotinic Acetylcholine Alphal, Bungarotoxin; Serotonin (5-Hydroxytryptamine) 5-HT3; Glycine, Strychnine-Sensitive; Neuropeptide Y Yl; Glutamate, AMP A; APJ; Bombesin BB1; Calcitonin; Calcitonin Gene-Related Peptide CGRPl; Cholecystokinin CCK1 (CCKA); Cholecystokinin CCK2 (CCKB); Galanin GALl; Galanin GAL2; Glucagon-Like Peptide-1 (GLP-1); Glutamate, NMDA, Agonist; Glutamate, NMDA, MK-801; Growth Hormone Secretagogue (GHS, Ghrelin); Insulin; Interleukin IL-6; Leptin; Melanocortin MC3; Melanocortin MC4; Motilin; Neuromedin U NMU1; Neuromedin U NMU2; Orexin OX1; Orexin OX2; Oxytocin; Pituitary Adenylate Cyclase-Activating Polypeptide -Type I (PAC1); Serotonin (5-Hydroxytryptamine) 5-HT2C; Somatostatin sstl; Somatostatin sst2; Somatostatin sst3; Thyroid Receptor Alpha Coactivator
In another embodiment the offsite targets included by the inventive methods include off- target receptors are Calcium Channel L-type receptors, Na Channel Site 2 receptors, Norepinephrine Cellular Transport receptors, Dopamine Cellular Transport receptors, CB-1 receptors, Adenosine a3 receptors, Leukotriene, Cysteinyl Cys T receptors, Histamine 2 receptors, or Sodium Channel Site 2 receptors. Definitions
Terms used herein will have their customary meaning in the art unless specified
otherwise.
The "withdrawal period" as used herein refers to the time interval that must pass between the last administration of a veterinary bioactive agent to a food producing animal and when the food producing animal can safely enter the food supply or its milk/eggs can be safely consumed. The withdrawal period is set be a governmental body such as the FDA in the United States or the EMA in Europe.
The terms "treating" or "treat" or "treatment" as used herein means the administration of a parasiticidal composition used in the inventive methods to a non-human animal that has a parasitic infestation for the eradication of the parasite or the reduction of the number of the parasites infesting the animal undergoing treatment. It is noted that the parasiticidal compositions of the inventive methods may be used to prevent such a parasitic infestation.
The terms "prevent", "prevention" or "prophylaxis" as used herein means the administration of a parasiticidal composition used in the inventive methods to a non-human animal before the parasitic infection or infestation has occurred in order to keep said infection or infestation from occurring.
The term "reducing the withdrawal period" means that the calculated withdrawal period using algorithms approved by relevant Regulatory Authorities (e.g. FDA, EMA, etc.) has been reduced as compared to the withdrawal period when the racemic mixture of the isoxazoline compound or pharmaceutically acceptable salt thereof is administered to the non-human animal (at the approved dose).
The term "reducing at least one unwanted effect" as used herein means that the occurrence of an unwanted or unintended biological effect, which is observed when the racemic mixture or the (R) enantiomer of the isoxazoline compound or pharmaceutically acceptable salt thereof is administered to the non-human animal, is considered to be reduced in severity and/or frequency by one of ordinary skill in this art.
By "effective amount" it is intended a sufficient amount of a composition of the invention to eradicate or reduce the number of parasites infesting the animal. In some embodiments, an effective amount of the active agent achieves at least 70% efficacy (% reduction vs. control) against the target parasite. In other embodiments, an effective amount of the active agent achieves at least 80%, or at least 90% efficacy against the target pests. Preferably, an effective amount of the bioactive agent will achieve at least 95%, at least 98% or 100% efficacy against
the target parasites.
The organic moieties mentioned in the definitions of the variables of formula (S)-I- formula (,S)-VII are - like the term halogen - collective terms for individual listings of the individual group members. The prefix Cn-Cm indicates in each case the possible number of carbon atoms in the group.
The term "long acting" or "long acting formulation" or "long acting composition" as used herein means a dosage form that is formulated in such a manner to make the bioactive agent(s) contained therein to be effective against the parasites of at least about 80% for two months, or longer. Long acting formulations are distinguished from "extended release" formulations in that they do not include pharmaceutically acceptable biodegradable polymers. For avoidance of doubt, polyethylene glycols (PEGs) and poloxamers are not considered biodegradable polymers. In another embodiment the long acting formulations are effective against the parasites of about 90% for about 3 months, or longer. In still another embodiment, the In another embodiment the long acting formulations are effective against the parasites of about 95% for about 3 months, or longer.
The term "extended release" or "extended release formulation" or "extended release composition" as used herein means a dosage form that is formulated in such a manner to make the bioactive agent(s) contained therein to be available over an extended period of time due to the interaction of the formulation components in combination with the natural pharmacokinetic or pharmacodynamic characteristics of the active agent(s). This definition is consistent with the use of the term known and accepted in the veterinary field as described in the article "Terminology Challenges: Defining Modified Release Dosage Forms in Veterinary Medicine" by Marilyn N. Martinez, Danielle Lindquist and Sanja Modric (Journal of Pharmaceutical Sciences, vol. 99, no. 8, August 2010).
For example, the extended release formulations according to the present invention would be understood to provide an efficacy of at least 90% against fleas and/or ticks for at least 3 months as described herein.
The term "non-human animal" is used herein to include all mammals excluding humans, birds and fish and also include all vertebrate animals. Animals include, but are not limited to, cats, dogs, cattle, chickens, cows, deer, goats, horses, llamas, pigs, sheep and yaks. It also includes an individual animal in all stages of development, including embryonic and fetal stages.
The term "substantially pure" is used herein to indicate that a compound or an enantiomer is at least about 90% (w/w) pure, at least about 95% (w/w), or at least about 98%> (w/w) pure, or higher.
The term "alkyl" refers to saturated straight, branched, primary, secondary or tertiary hydrocarbons, including those having 1 to 20 atoms. In some embodiments, alkyl groups will include C1-C12, Ci-Ci0, Ci-C8, Ci-C6 or C1-C4 alkyl groups. Examples of Ci-Ci0 alkyl include, but are not limited to, methyl, ethyl, propyl, 1-methylethyl, butyl, 1-methylpropyl, 2- methylpropyl, 1, 1-dimethylethyl, pentyl, 1-methylbutyl, 2-methylbutyl, 3-methylbutyl, 2,2- dimethylpropyl, 1-ethylpropyl, hexyl, 1,1-dimethylpropyl, 1,2-dimethylpropyl, 1-methylpentyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 1,3- dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3-dimethylbutyl, 1-ethylbutyl, 2- ethylbutyl, 1, 1,2-trimethylpropyl, 1,2,2-trimethylpropyl, 1 -ethyl- 1-methylpropyl, l-ethyl-2- methylpropyl, heptyl, octyl, 2-ethylhexyl, nonyl and decyl and their isomers. Ci-C4-alkyl means for example methyl, ethyl, propyl, 1-methylethyl, butyl, 1-methylpropyl, 2-methylpropyl or 1,1- dimethylethyl.
Cyclic alkyl groups or "cycloalkyl" include those with 3 to 10 carbon atoms having single or multiple condensed rings. In some embodiments, cycloalkyl groups include C4-C7 or C3-C4 cyclic alkyl groups. Non-limiting examples of cycloalkyl groups include adamantyl, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, cyclooctyl and the like.
The alkyl groups described herein can be unsubstituted or substituted with one or more moieties selected from the group consisting of alkyl, halo, haloalkyl, hydroxyl, carboxyl, acyl, acyloxy, amino, alkyl- or dialkylamino, amido, arylamino, alkoxy, aryloxy, nitro, cyano, azido, thiol, imino, sulfonic acid, sulfate, sulfonyl, sulfanyl, sulfinyl, sulfamoyl, ester, phosphonyl, phosphinyl, phosphoryl, phosphine, thioester, thioether, acid halide, anhydride, oxime, hydrazine, carbamate, phosphoric acid, phosphate, phosphonate, or any other viable functional group that does not inhibit the biological activity of the compounds of the invention, either unprotected, or protected as necessary, as known to those skilled in the art, for example, as taught in Greene, et al., Protective Groups in Organic Synthesis, John Wiley and Sons, Third Edition, 1999, hereby incorporated by reference.
Terms including the term "alkyl" such as "alkylcycloalkyl," "cycloalkylalkyl,"
"alkylamino," or "dialkylamino" will be understood to comprise an alkyl group as defined above
linked to the other functional group, where the group is linked to the compound through the last group listed, as understood by those of skill in the art.
The term "alkenyl" refers to both straight and branched carbon chains which have at least one carbon-carbon double bond. In some embodiments, alkenyl groups may include C2-C20 alkenyl groups. In other embodiments, alkenyl includes C2-C12, C2-C10, C2-C8, C2-C6 or C2-C4 alkenyl groups. In one embodiment of alkenyl, the number of double bonds is 1-3, in another embodiment of alkenyl, the number of double bonds is one or two. Other ranges of carbon- carbon double bonds and carbon numbers are also contemplated depending on the location of the alkenyl moiety on the molecule. "C2-Cio-alkenyl" groups may include more than one double bond in the chain. Examples include, but are not limited to, ethenyl, 1-propenyl, 2-propenyl, 1- methyl-ethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1 -methyl- 1-propenyl, 2-m ethyl- 1-propenyl, 1- methyl-2-propenyl, 2-methyl-2-propenyl; 1-pentenyl, 2-pentenyl, 3-pentenyl, 4-pentenyl, 1- m ethyl- 1-butenyl, 2-m ethyl- 1-butenyl, 3 -methyl- 1-butenyl, l-methyl-2-butenyl, 2-methyl-2- butenyl, 3-methyl-2-butenyl, 1 -methyl -3-butenyl, 2-methyl-3-butenyl, 3-methyl-3-butenyl, 1,1- dimethyl-2-propenyl, 1,2-dimethyl- 1-propenyl, l,2-dimethyl-2-propenyl, 1 -ethyl- 1-propenyl, 1- ethyl-2-propenyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-hexenyl, 1 -methyl- 1-pentenyl, 2-methyl- 1-pentenyl, 3 -methyl- 1-pentenyl, 4-methyl- 1-pentenyl, l-methyl-2-pentenyl, 2-methyl- 2-pentenyl, 3-methyl-2-pentenyl, 4-methyl-2-pentenyl, l-methyl-3 -pentenyl, 2-methyl-3- pentenyl, 3 -methyl-3 -pentenyl, 4-methyl-3 -pentenyl, l-methyl-4-pentenyl, 2-methyl-4-pentenyl, 3-methyl-4-pentenyl, 4-methyl-4-pentenyl, l,l-dimethyl-2-butenyl, l,l-dimethyl-3-butenyl, 1,2- dimethyl- 1-butenyl, l,2-dimethyl-2-butenyl, l,2-dimethyl-3-butenyl, 1,3-dimethyl-l-butenyl, l,3-dimethyl-2-butenyl, l,3-dimethyl-3-butenyl, 2,2-dimethyl-3-butenyl, 2,3-dimethyl-l-butenyl, 2,3-dimethyl-2-butenyl, 2,3-dimethyl-3-butenyl, 3, 3 -dimethyl- 1-butenyl, 3,3-dimethyl-2-butenyl, 1 -ethyl- 1-butenyl, l-ethyl-2-butenyl, l-ethyl-3-butenyl, 2-ethyl- 1-butenyl, 2-ethyl-2-butenyl, 2- ethyl-3-butenyl, l,l,2-trimethyl-2-propenyl, 1 -ethyl- l-methyl-2-propenyl, l-ethyl-2-methyl-l- propenyl and l-ethyl-2-methyl-2-propenyl.
"Alkynyl" refers to both straight and branched carbon chains which have at least one carbon-carbon triple bond. In one embodiment of alkynyl, the number of triple bonds is 1-3; in another embodiment of alkynyl, the number of triple bonds is one or two. In some embodiments, alkynyl groups include from C2-C20 alkynyl groups. In other embodiments, alkynyl groups may include C2-C12, C2-C10, C2-C8, C2-C6 or C2-C4 alkynyl groups. Other ranges of carbon-carbon
triple bonds and carbon numbers are also contemplated depending on the location of the alkenyl moiety on the molecule. For example, the term "C2-Cio-alkynyl" as used herein refers to a straight-chain or branched unsaturated hydrocarbon group having 2 to 10 carbon atoms and containing at least one triple bond, such as ethynyl, prop-l-yn-l-yl, prop-2-yn-l-yl, n-but-l-yn- 1-yl, n-but-l-yn-3-yl, n-but-l-yn-4-yl, n-but-2-yn-l-yl, n-pent-l-yn-l-yl, n-pent-l-yn-3-yl, n- pent-l-yn-4-yl, n-pent-l-yn-5-yl, n-pent-2-yn-l-yl, n-pent-2-yn-4-yl, n-pent-2-yn-5-yl, 3- methylbut-l-yn-3-yl, 3-methylbut-l-yn-4-yl, n-hex-l-yn-l-yl, n-hex-l-yn-3-yl, n-hex-l-yn-4-yl, n-hex-l-yn-5-yl, n-hex-l-yn-6-yl, n-hex-2-yn-l-yl, n-hex-2-yn-4-yl, n-hex-2-yn-5-yl, n-hex-2- yn-6-yl, n-hex-3-yn-l-yl, n-hex-3-yn-2-yl, 3-methylpent-l-yn-l-yl, 3-methylpent-l-yn-3-yl, 3- methylpent-l-yn-4-yl, 3-methylpent-l-yn-5-yl, 4-methylpent-l-yn-l-yl, 4-methylpent-2-yn-4-yl or 4-methylpent-2-yn-5-yl and the like.
The term "haloalkyl" refers to an alkyl group, as defined herein, which is substituted by one or more halogen atoms. For example Ci-C4-haloalkyl includes, but is not limited to, chloromethyl, bromomethyl, dichloromethyl, trichloromethyl, fluoromethyl, difluoromethyl, trifluoromethyl, chlorofluoromethyl, di chlorofluoromethyl, chlorodifluoromethyl, 1-chloroethyl, 1-bromoethyl, 1-fluoroethyl, 2-fluoroethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, 2-chloro-2- fluoroethyl, 2-chloro-2,2-difluoroethyl, 2,2-dichloro-2-fluoroethyl, 2,2,2-trichloroethyl, pentafluoroethyl and the like.
The term "haloalkenyl" refers to an alkenyl group, as defined herein, which is substituted by one or more halogen atoms.
The term "haloalkynyl" refers to an alkynyl group, as defined herein, which is substituted by one or more halogen atoms.
"Alkoxy" refers to alkyl-O-, wherein alkyl is as defined above. Similarly, the terms "alkenyloxy," "alkynyloxy," "haloalkoxy," "haloalkenyloxy," "haloalkynyloxy," "cycloalkoxy," "cycloalkenyloxy," "halocycloalkoxy," and "halocycloalkenyloxy" refer to the groups alkenyl- 0-, alkynyl-O-, haloalkyl-O-, haloalkenyl-O-, haloalkynyl-O-, cycloalkyl-O-, cycloalkenyl-O-, halocycloalkyl-O-, and halocycloalkenyl-O-, respectively, wherein alkenyl, alkynyl, haloalkyl, haloalkenyl, haloalkynyl, cycloalkyl, cycloalkenyl, halocycloalkyl, and halocycloalkenyl are as defined above. Examples of Ci-C6-alkoxy include, but are not limited to, methoxy, ethoxy, C2H5-CH2O-, (CH3)2CHO-, n-butoxy, C2H5-CH(CH3)0-, (CH3)2CH-CH20-; (CH3)3CO-, n- pentoxy, 1-methylbutoxy, 2-methylbutoxy, 3-methylbutoxy, 1, 1-dimethylpropoxy,
1.2- dimethylpropoxy, 2,2-dimethyl-propoxy, 1-ethylpropoxy, n-hexoxy, 1 -methylpentoxy, 2- methylpentoxy, 3-methylpentoxy, 4-methylpentoxy, 1, 1-dimethylbutoxy, 1,2-dimethylbutoxy,
1.3- dimethylbutoxy, 2,2-dimethylbutoxy, 2,3-dimethylbutoxy, 3,3-dimethylbutoxy, 1- ethylbutoxy, 2-ethylbutoxy, 1, 1,2-trimethylpropoxy, 1,2,2-trimethylpropoxy, 1 -ethyl- 1- methylpropoxy, l-ethyl-2-methylpropoxy and the like.
The term "alkylthio" refers to alkyl-S-, wherein alkyl is as defined above. Similarly, the terms "haloalkylthio," "cycloalkylthio," and the like, refer to haloalkyl-S- and cycloalkyl-S- where haloalkyl and cycloalkyl are as defined above.
The term " alkyl sulfinyl" refers to alkyl-S(O)-, wherein alkyl is as defined above. Similarly, the term "haloalkylsulfinyl" refers to haloalkyl-S(O)- where haloalkyl is as defined above.
The term " alkyl sulfonyl" refers to alkyl-S(0)2-, wherein alkyl is as defined above. Similarly, the term "haloalkylsulfonyl" refers to haloalkyl-S(0)2- where haloalkyl is as defined above.
The term alkylamino and dialkylamino refer to alkyl- H- and (alkyl)2N- where alkyl is as defined above. Similarly, the terms "haloalkylamino" refers to haloalkyl- H- where haloalkyl is as defined above.
The terms "alkylcarbonyl," "alkoxycarbonyl," "alkylaminocarbonyl," and "dialkylaminocarbonyl" refer to alkyl-C(O)-, alkoxy-C(O)-, alkylamino-C(O)- and dialkylamino-C(O)- where alkyl, alkoxy, alkylamino and dialkylamino are as defined above. Similarly, the terms "haloalkylcarbonyl," "haloalkoxycarbonyl," "haloalkylaminocarbonyl," and "dihaloalkylaminocarbonyl" refer to the groups haloalkyl-C(O)-, haloalkoxy-C(O)-, haloalkylamino-C(O)- and dihaloalkylamino-C(O)- where haloalkyl, haloalkoxy, haloalkylamino and dihaloalkylamino are as defined above.
"Aryl" refers to a monovalent aromatic carbocyclic group of from 6 to 14 carbon atoms having a single ring or multiple condensed rings. In some embodiments, aryl groups include C6- Cio aryl groups. Aryl groups include, but are not limited to, phenyl, biphenyl, naphthyl, tetrahydronaphthyl, phenylcyclopropyl and indanyl. Aryl groups may be unsubstituted or substituted by one or more moieties selected from halogen, cyano, nitro, hydroxy, mercapto, amino, alkyl, alkenyl, alkynyl, cycloalkyl, cycloalkenyl, haloalkyl, haloalkenyl, haloalkynyl, halocycloalkyl, halocycloalkenyl, alkoxy, alkenyloxy, alkynyloxy, haloalkoxy, haloalkenyloxy,
haloalkynyloxy, cycloalkoxy, cycloalkenyloxy, halocycloalkoxy, halocycloalkenyloxy, alkylthio, haloalkylthio, cycloalkylthio, halocycloalkylthio, alkylsulfinyl, alkenylsulfinyl, alkynyl-sulfinyl, haloalkylsulfinyl, haloalkenylsulfinyl, haloalkynylsulfinyl, alkylsulfonyl, alkenylsulfonyl, alkynylsulfonyl, haloalkyl-sulfonyl, haloalkenylsulfonyl, haloalkynylsulfonyl, alkylamino, alkenylamino, alkynylamino, di(alkyl)amino, di(alkenyl)-amino, di(alkynyl)amino, or trialkylsilyl.
The terms "aralkyl" or "arylalkyl" refers to an aryl group that is bonded to the parent compound through a diradical alkylene bridge, (-CH2-)n, where n is 1-12 and where "aryl" is as defined above.
"Heteroaryl" refers to a monovalent aromatic group of from 1 to 15 carbon atoms, preferably from 1 to 10 carbon atoms, having one or more oxygen, nitrogen, and sulfur heteroatoms within the ring, preferably 1 to 4 heteroatoms, or 1 to 3 heteroatoms. The nitrogen and sulfur heteroatoms may optionally be oxidized. Such heteroaryl groups can have a single ring (e.g., pyridyl or furyl) or multiple condensed rings provided that the point of attachment is through a heteroaryl ring atom. Preferred heteroaryls include pyridyl, piridazinyl, pyrimidinyl, pyrazinyl, triazinyl, pyrrolyl, indolyl, quinolinyl, isoquinolinyl, quinazolinyl, quinoxalinnyl, furanyl, thienyl, furyl, pyrrolyl, imidazolyl, oxazolyl, isoxazolyl, isothiazolyl, pyrazolyl benzofuranyl, and benzothienyl. Heteroaryl rings may be unsubstituted or substituted by one or more moieties as described for aryl above. The term "heteroarylene" (where the heteroaryl group is a bridging group) should be construed accordingly.
"Heterocyclyl," "heterocyclic" or "heterocyclo" refer to fully saturated or unsaturated, cyclic groups, for example, 3 to 7 membered monocyclic or 4 to 7 membered monocyclic; 7 to 11 membered bicyclic, or 10 to 15 membered tricyclic ring systems, which have one or more oxygen, sulfur or nitrogen heteroatoms in ring, preferably 1 to 4 or 1 to 3 heteroatoms. The nitrogen and sulfur heteroatoms may optionally be oxidized and the nitrogen heteroatoms may optionally be quaternized. The heterocyclic group may be attached at any heteroatom or carbon atom of the ring or ring system and may be unsubstituted or substituted by one or more moieties as described for aryl groups above.
Exemplary monocyclic heterocyclic groups include, but are not limited to, pyrrolidinyl, pyrrolyl, pyrazolyl, oxetanyl, pyrazolinyl, imidazolyl, imidazolinyl, imidazolidinyl, oxazolyl, oxazolidinyl, isoxazolinyl, isoxazolyl, thiazolyl, thiadiazolyl, thiazolidinyl, isothiazolyl,
isothiazolidinyl, furyl, tetrahydrofuryl, thienyl, oxadiazolyl, piperidinyl, piperazinyl, 2- oxopiperazinyl, 2-oxopiperidinyl, 2-oxopyrrolodinyl, 2-oxoazepinyl, azepinyl, 4-piperidonyl, pyridinyl, pyrazinyl, pyrimidinyl, pyridazinyl, tetrahydropyranyl, morpholinyl, thiamorpholinyl, thiamorpholinyl sulfoxide, thiamorpholinyl sulfonyl, 1,3-dioxolane and tetrahydro-1,1- dioxothienyl, triazolyl, triazinyl, and the like.
Exemplary bicyclic heterocyclic groups include, but are not limited to, indolyl, benzothiazolyl, benzoxazolyl, benzodioxolyl, benzothienyl, quinuclidinyl, quinolinyl, tetra- hydroisoquinolinyl, isoquinolinyl, benzimidazolyl, benzopyranyl, indolizinyl, benzofuryl, chromonyl, coumarinyl, benzopyranyl, cinnolinyl, quinoxalinyl, indazolyl, pyrrol opyridyl, furopyridinyl (such as furo[2,3-c]pyridinyl, furo[3,2-b]pyridinyl]or furo[2,3-b]pyridinyl), dihydroisoindolyl, dihydroquinazolinyl (such as 3,4-dihydro-4-oxo-quinazolinyl), tetrahydroquinolinyl and the like. The term "heterobicyclylene" (where the bicyclic heterocyclic group is a bridging group) should be construed accordingly.
Exemplary tricyclic heterocyclic groups include carbazolyl, benzidolyl, phenanthrolinyl, acridinyl, phenanthridinyl, xanthenyl, and the like.
Halogen means the atoms fluorine, chlorine, bromine and iodine. The designation of "halo" (e.g. as illustrated in the term haloalkyl) refers to all degrees of substitutions from a single substitution to a perhalo substitution (e.g. as illustrated with methyl as chloromethyl (-CH2C1), dichloromethyl (-CHC12), trichloromethyl (-CC13)).
In addition, the compounds within the parasiticidal compositions of the inventive methods may exist as hydrates or solvates, in which a certain stoichiometric amount of water or a solvent is associated with the molecule in the crystalline form. The parasiticidal compositions may include hydrates and solvates of the active agents. In some embodiments, the parasiticidal compositions of the inventive methods may include up to 15% (w/w), up to 20% (w/w), or up to 30%) (w/w) of a particular solid form.
Salts
Also contemplated within the scope of the invention are acid or base salts, where applicable, of the compounds of the invention provided for herein.
The term "acid salt" contemplates salts of the compounds with all pharmaceutically acceptable inorganic or organic acids. Inorganic acids include mineral acids such as hydrohalic acids such as hydrobromic acid and hydrochloric acid, sulfuric acid, phosphoric acids and nitric
acid. Organic acids include all pharmaceutically acceptable aliphatic, alicyclic and aromatic carboxylic acids, dicarboxylic acids, tricarboxylic acids and fatty acids. In one embodiment of the acids, the acids are straight chain or branched, saturated or unsaturated C1-C20 aliphatic carboxylic acids, which are optionally substituted by halogen or by hydroxyl groups, or C6-Ci2 aromatic carboxylic acids. Examples of such acids are carbonic acid, formic acid, acetic acid, propionic acid, isopropionic acid, valeric acid, a-hydroxy acids such as glycolic acid and lactic acid, chloroacetic acid, benzoic acid, pamoic acid, methane sulfonic acid, and salicylic acid. Examples of dicarboxylic acids include oxalic acid, malic acid, succinic acid, tartaric acid, fumaric acid, and maleic acid. An example of a tricarboxylic acid is citric acid. Fatty acids include all pharmaceutically acceptable saturated or unsaturated aliphatic or aromatic carboxylic acids having 4 to 24 carbon atoms. Examples include butyric acid, isobutyric acid, sec-butyric acid, lauric acid, palmitic acid, stearic acid, oleic acid, linoleic acid, linolenic acid, and phenylsteric acid. Other acids include gluconic acid, glycoheptonic acid and lactobionic acid.
The term "base salt" contemplates salts of the compounds with all pharmaceutically acceptable inorganic or organic bases, including hydroxides, carbonates or bicarbonates of alkali metal or alkaline earth metals. Salts formed with such bases include, for example, the alkali metal and alkaline earth metal salts, including, but not limited to, as the lithium, sodium, potassium, magnesium or calcium salts. Salts formed with organic bases include the common hydrocarbon and heterocyclic amine salts, which include, for example, ammonium salts ( H4+), alkyl- and dialkylammonium salts, and salts of cyclic amines such as the morpholine and piperidine salts.
Parasiticidal Compositions
The parasiticidal compositions used in the inventive methods comprises a parasiticidal effective amount of at least one isoxazoline compound of formula (S)-l or a veterianarily acceptable salt thereof in combination with an inert carrier or formulation diluent and optionally other non-active excipients, wherein the composition contains less than about 10% by weight of an (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof. The parasiticidal compositions may be in a variety of solid and liquid forms which are suitable for application on or administration to an animal. For example, the parasiticidal compositions employed in the inventive methods may be in formulations suitable for oral administration, injectable administration, including subcutaneous and parenteral administration, and topical
administration (e.g. spot-on or pour-on), dermal or subdermal administration.
A. Oral Formulations
As discussed above, the parasiticidal compositions used in the invention may be in a form suitable for oral use (see, e.g., U.S. Patent No. 4,564,631, which is hereby incorporated by reference in its entirety), dietary supplements, troches, lozenges, chewables, tablets, hard or soft capsules, bolus, emulsions, aqueous or oily suspensions, aqueous or oily solutions, oral drench formulations, dispersible powders or granules, premixes, syrups or elixirs, enteric formulations or pastes. Compositions intended for oral use may be prepared according to any method known in the art for the manufacture of pharmaceutical compositions and such compositions may contain one or more sweetening agents, bittering agents, flavoring agents, coloring agents and preserving agents in order to provide pharmaceutically elegant and palatable preparations.
Tablets may contain the active ingredient in admixture with non-toxic, pharmaceutically acceptable excipients which are suitable for the manufacture of tablets. These excipients may be, for example, inert diluents, such as calcium carbonate, sodium carbonate, lactose, calcium phosphate or sodium phosphate; granulating and disintegrating agents, for example, corn starch, or alginic acid; binding agents, for example starch, gelatin or acacia, and lubricating agents, for example, magnesium stearate, stearic acid or talc. The tablets may be uncoated or they may be coated by known techniques to delay disintegration and absorption in the gastrointestinal tract and thereby provide a sustained action over a longer period. For example, a time delay material such as glyceryl monostearate or glyceryl distearate may be employed. They may also be coated by the technique described in U.S. Patent Nos. 4,256, 108; 4,166,452; and 4,265,874 (all incorporated herein by reference in their entirety) to form osmotic therapeutic tablets for controlled release.
Oral formulations include hard gelatin capsules, wherein the active ingredient is mixed with an inert solid diluent, for example, calcium carbonate, calcium phosphate or kaolin. Capsules may also be soft gelatin capsules, wherein the active ingredient is mixed with water or miscible solvents such as propylene glycol, polyethylene glycols (PEGs) and ethanol, or an oil medium, for example peanut oil, liquid paraffin, or olive oil.
The parasiticidal compositions may be chewable tablet compositions or soft chewable compositions ("soft chew") such as those described in US 9,233,100 B2, US 9,259,417, US
2010/0087492, US 2006/0222684, US 2004/0151759, US 7955632, US 2015/0057321, US 2015/0057239 and WO 2016/073347, all incorporated herein by reference in their entirety.
A soft chew is palatable and acceptable to the animal. In addition to the bioactive active agents(s), the soft chews may include one or more of the following components: a solvent or mixture of solvents, one or more fillers, one or more binders, one or more surfactants, one or more humectants, one or more lubricants, one or more disintegrants, one or more colorants, one or more antimicrobial agents, one or more antioxidants, one or more pH modifiers and one or more flavoring agents.
Solvents that may be used include, but are not limited to, various grades of liquid polyethylene glycol (PEG) including PEG 200, PEG 300, PEG 400 and PEG 540; propylene carbonate; propylene glycol; triglycerides including, but not limited to caprylic/capric triglyceride, caprylic/capric/linoleic triglyceride (e.g. MIGLYOL® 810 and 812, caprylic/capric/succinic triglyceride, propylene glycol dicaprylate/dicaprate, and the like; water, sorbitol solution, glycerol caprylate/caprate and polyglycolized glycerides (GELUCIRE ®), or a combination thereof.
Various fillers known in the art may be used in the soft chewable compositions. Fillers include, but are not limited to, corn starch, pre-gelatinized corn starch, soy protein fines, corn cob, and corn gluten meal, and the like. In some embodiments, a combination of two or more fillers may be used in the compositions.
Binders that may be used in soft chewable compositions include, but are not limited to, polyvinylpyrrolidone (e.g. Povidone), cross-linked polyvinylpyrrolidone (Crospovidone), PEGs of various grades including PEG 3350, PEG 4000, PEG 6000, PEG 8000 and even PEG 20,000, and the like; co-polymers of vinylpyrrolidone and vinyl acetate (e.g. Copovidone) such as the product sold by BASF by the tradename Kollidon® VA 64 and the like; starch such as potato starch, tapioca starch or corn starch; molasses, corn syrup, honey, maple syrup and sugars of various types; or a combination of two or more binders.
Humectants that may be used in soft chewable compositions include, but are not limited to, glycerol (also referred to herein as glycerin), propylene glycol, cetyl alcohol and glycerol monostearate, and the like. Polyethylene glycols of various grades may also be used as humectants.
Surfactants may be present in soft chewable composition to improve their solubility and absorption after ingestion. Surfactants are typically present in a concentration of about 1 to 10% (w/w), more typically about 1 to about 5% (w/w). Examples of surfactants that may be used in the compositions include, but are not limited to, glyceryl monooleate, polyoxyethylene sorbitan fatty acid esters, sorbitan esters including sorbitan monooleate (Span® 20), polyvinyl alcohol, polysorbates including polysorbate 20 and polysorbate 80, <i-a-tocopheiyl polyethylene glycol 1000 succinate (TPGS), sodium lauryl sulfate, co-polymers of ethylene oxide and propylene oxide (e.g. poloxamers such as LUTROL® F87 and the like), polyethylene glycol castor oil derivatives including polyoxyl 35 castor oil (Cremophor® EL), polyoxyl 40 hydrogenated castor oil (Cremophor® RH 40), polyoxyl 60 hydrogenated castor oil (Cremophor® RH60); propylene glycol monolaurate (LAUROGLYCOL®); glyceride esters including glycerol caprylate/caprate (CAPMUL® MCM), polyglycolized glycerides (GELUCIRE®), PEG 300 caprylic/capric glycerides (Softigen® 767), PEG 400 caprylic/capric glycerides (Labrasol®), PEG 300 oleic glycerides (Labrafil® M-1944CS), PEG 300 linoleic glycerides (Labrafil® M-2125CS); polyethylene glycol stearates and polyethylene glycol hydroxy stearates including polyoxyl 8 stearate (PEG 400 monostearate), polyoxyl 40 stearate (PEG 1750 monostearate), and the like. Polyethylene glycol stearates (synonyms include macrogol stearates, polyoxyl stearates, polyoxyethylene stearates, ethoxylated stearates; CAS No. 9004-99-3, 9005-08-7) are mixtures of mono- and distearate esters of mixed polyoxyethylene polymers. Polyethylene glycol hydroxystearate is a mixture of mono- and diesters of hydroxystearic acid with polyethylene glycols. One polyethylene glycol hydroxystearate that may be used in the compositions is polyethylene glycol 12-hydroxy stearate. In another embodiment, the compositions may include the surfactant polyethylene glycol 15 12-hydroxy stearate (Solutol® HS 15 from BASF), a mixture of mono- and diesters of 12-hydroxy stearic acid with 15 moles of ethylene oxide. Again, these compounds, as well as their amounts are well known in the art. In another embodiment of the invention, the compositions may include polyoxyl 35 castor oil (Cremophor® EL) as a surfactant. In other embodiments, the chewable compositions may include polyoxyl 40 hydrogenated castor oil (Cremophor® RH 40) or polyoxyl 60 hydrogenated castor oil (Cremophor® RH60) as surfactants. The compositions of the invention may also include a combination of surfactants.
Oral fomulations (including soft chewable formulations) may contain other inert ingredients such as antioxidants, preservatives, or pH stabilizers. These compounds are well known in the formulation art. Antioxidants may be added to the compositions of the invention to inhibit degradation of the active agents. Suitable antioxidants include, but are not limited to, alpha tocopherol, ascorbic acid, ascrobyl palmitate, fumaric acid, malic acid, sodium ascorbate, sodium metabi sulfate, n-propyl gallate, butylated hydroxy anisole (BHA), BHT (butylated hydroxy toluene) monothioglycerol and the like.
Oral formulations (including soft chewable formulations) may also include one or more lubricants and/or processing aids. In some cases, the lubricant/processing aid may also behave as a solvent, and accordingly, there some of the components of the parasiticidal compositions may have dual functions. Lubricants/processing aids include, but are not limited to polyethylene glycols of various molecular weight ranges including PEG 3350 (Dow Chemical) and PEG 4000, corn oil, mineral oil, hydrogenated vegetable oils (STEROTEX or LUBRITAB), peanut oil and/or castor oil.
Many flavoring agents may be used in oral formulations to improve the palatability of the oral veterinary formulations. Preferred flavoring agents are those that are not derived from animal sources. In various embodiments, flavoring components derived from fruit, meat (including, but not limited to pork, beef, chicken, fish, poultry, and the like), vegetable, cheese, bacon, cheese-bacon and/or artificial flavorings may be used. A flavoring component is typically chosen based upon consideration related to the organism that will be ingesting the soft chew. For example, a horse may prefer an apple flavoring component, while a dog may prefer a meat flavoring component. Although flavoring components derived from non-animal sources are preferred, in some embodiments, natural flavors containing beef or liver extracts, etc., may be used such as braised beef flavor artificial powdered beef flavor, roast beef flavor and corned beef flavor among others.
Alternatively, the parasiticidal composition may be administered via a drench, and is administered orally. Drench formulations are those in which the liquid-containing compositions of the invention are administered to the mouth or throat of the animal.
The parasiticidal compositions used in the inventive methods may also be in the form of oil-in-water or water-in-oil emulsions. The oily phase maybe a vegetable oil, for example, olive oil or arachis oil, or a mineral oil, for example, liquid paraffin or mixtures of these. Suitable
emulsifying agents include naturally-occurring phosphatides, for example, soy bean, lecithin, and esters or partial esters derived from fatty acids and hexitol anhydrides, for example, sorbitan monoleate, and condensation products of the said partial esters with ethylene oxide, for example, polyoxyethylene sorbitan monooleate. The emulsions may also contain sweetening agents, bittering agents, flavoring agents, and/or preservatives.
The oral compositions may also be in the form of a microemulsion. Microemulsions are well suited as the liquid carrier vehicle. Microemulsions are quaternary systems comprising an aqueous phase, an oily phase, a surfactant and a co-surfactant. They are translucent and isotropic liquids.
Microemulsions are composed of stable dispersions of microdroplets of the aqueous phase in the oily phase or conversely of microdroplets of the oily phase in the aqueous phase. The size of these microdroplets may be less than 200 nm (1000 to 100,000 nm for emulsions). The interfacial film may be composed of an alternation of surface-active (SA) and co-surface- active (Co-SA) molecules which, by lowering the interfacial tension, allows the microemulsion to be formed spontaneously.
The oily phase may be formed from mineral or vegetable oils, from unsaturated polyglycosylated glycerides or from triglycerides, or alternatively from mixtures of such compounds. For example, the oily phase may be comprised of triglycerides. Alternatively, the triglycerides may be medium-chain triglycerides, such as, for example C8-Ci0 caprylic/capric triglyceride. The oily phase may represent a % v/v range of, for example, about 2 to about 15%; about 7 to about 10%; and about 8 to about 9% v/v of the microemulsion.
The aqueous phase may include, for example water or glycol derivatives, such as propylene glycol, glycol ethers, polyethylene glycols or glycerol. In one embodiment, the glycol may be propylene glycol, diethylene glycol monoethyl ether, dipropylene glycol monoethyl ether or mixtures thereof. Generally, the aqueous phase will represent a proportion from about 1 to about 4% v/v in the microemulsion.
Surfactants for the microemulsion may include diethylene glycol monoethyl ether, dipropyelene glycol monomethyl ether, polyglycolyzed C8-Cio glycerides or polyglyceryl-6 dioleate. In addition to these surfactants, the co-surfactants may include short-chain alcohols, such as ethanol and propanol.
Some compounds are common to the three components discussed above, i.e., aqueous phase, surfactant and co-surfactant. However, it is well within the skill level of the practitioner to use different compounds for each component of the same formulation. For example, for the amount of surfactant/co-surfactant, the co-surfactant to surfactant ratio could be, for example, from about 1/7 to about 1/2. Generally, the amount of co-surfactant, there will be from about 25 to about 75% v/v of surfactant and from about 10 to about 55% v/v of co-surfactant in the microemulsion.
Oily suspensions may be formulated by suspending the active ingredient in a vegetable oil, for example, atachis oil, olive oil, sesame oil or coconut oil, or in mineral oil such as liquid paraffin. The oily suspensions may contain a thickening agent, for example, beeswax, hard paraffin or cetyl alcohol. Sweetening agents such as sucrose, saccharin or aspartame, bittering agents, and flavoring agents may be added to provide a palatable oral preparation. These compositions may be preserved by the addition of an anti-oxidant such as ascorbic acid, or other known preservatives.
Aqueous suspensions may contain the active material in admixture with excipients suitable for the manufacture of aqueous suspensions. Such excipients include suspending agents, for example, sodium carboxymethylcellulose, methylcellulose, hydroxy-propylmethylcellulose, sodium alginate, polvinylpyrrolidone, gum tragacanth and gum acacia; dispersing or wetting agents include naturally-occurring phosphatide, for example, lecithin, or condensation products of an alkylene oxide with fatty acids, for example polyoxyethylene stearate, or condensation products of ethylene oxide with long chain aliphatic alcohols, for example, heptadecaethyleneoxycetanol, or condensation products of ethylene oxide with partial esters derived from fatty acids and a hexitol such as polyoxyethylene sorbitol monooleate, or condensation products of ethylene oxide, with partial esters derived from fatty acids and hexitol anhydrides, for example polyethylene sorbitan monooleate. The aqueous suspensions may also contain one or more preservatives, for example ethyl, or n-propyl, p-hydroxybenzoate, one or more coloring agents, one or more flavoring agents, and one or more sweetening agents and/or bittering agents, such as those set forth above.
Dispersible powders and granules suitable for preparation of an aqueous suspension by the addition of water may provide the active ingredient in admixture with a dispersing or wetting agent, suspending agent and one or more preservatives. Suitable dispersing or wetting agents
and suspending agents are exemplified by those already mentioned above. Additional excipients, for example, sweetening, bittering, flavoring and coloring agents, may also be present.
Syrups and elixirs may be formulated with sweetening agents, for example, glycerol, propylene glycol, sorbitol or sucrose. Such formulations may also contain a demulcent, a preservative, flavoring agent(s) and/or coloring agent(s).
The oral formulation may also be in paste form. Examples of embodiments in a paste form include, but are not limited to, those described in U.S. Patent Nos. 6,787,342 and 7,001,889 (each of which are incorporated herein by reference). In addition to the compounds of the invention, the paste may further contain fumed silica; a viscosity modifier; a carrier (e.g., a hydrophilic carrier which is triacetin, a monoglyceride, a diglyceride, or a triglyceride); optionally, an absorbent; and optionally, a colorant, stabilizer, surfactant, or preservative.
The paste may also include a viscosity modifier. Suitable viscosity modifiers include, but are not limited to, PEGs including, but not limited to, PEG 200, PEG 300, PEG 400, PEG 600; monoethanolamine, triethanolamine, glycerol, propylene glycol, polyoxyethylene (20) sorbitan mono-oleate (polysorbate 80 or Tween 80), or poloxamers (e.g., Pluronic L 81); an absorbent such as magnesium carbonate, calcium carbonate, starch, and cellulose and its derivatives; and a colorant including, but not limited to, titanium dioxide iron oxide, or FD&C Blue #1 Aluminum Lake.
B. Topical Formulations
Alternatively, the parasiticidal compositions used in the inventive methods may be designed for topical administration. Topical, dermal and subdermal formulations may include, by way of non-limiting example, emulsions, creams, ointments, gels, pastes, powders, shampoos, pour-on formulations, ready-to-use formulations, spot-on solutions and suspensions, dips and sprays. Spot-on, spray-on or pour-on formulation allow the bioactive agent or agents to be absorbed through the skin to achieve systemic levels, distributed through the sebaceous glands or on the surface of the skin achieving levels throughout the coat. When the compound is distributed through the sebaceous glands, they may act as a reservoir, whereby there may be a long-lasting effect (up to several months) effect. Spot-on formulations are typically applied in a localized region which refers to an area other than the entire animal. In one embodiment, the location may be between the shoulders. In another embodiment it may be a stripe, e.g. a stripe from head to tail of the animal.
Pour-on formulations are described in U. S. Patent Nos. 6,010,710 and 9, 180, 121, both incorporated herein by reference. Pour-on formulations may be advantageously oily, and generally comprise a diluent or vehicle and also a solvent (e.g. an organic solvent) for the active ingredient if the latter is not soluble in the diluent. In another embodiment, pour-on formulations are based on alcoholic solvents including isopropyl alcohol or ethyl alcohol, optionally in combination with other solvents.
Organic solvents that can be used in the parasiticidal compositions, but are not limited to, include acetyltributyl citrate, fatty acid esters such as the dimethyl ester, diisobutyl adipate, acetone, acetonitrile, benzyl alcohol, ethyl alcohol, butyl diglycol, dimethylacetamide, dimethylformamide, dimethyl sulfoxide, dimethyl isosorbide, dipropylene glycol n-butyl ether, ethanol, isopropanol, methanol, ethylene glycol monoethyl ether, ethylene glycol monomethyl ether, monomethylacetamide, dipropylene glycol monomethyl ether, liquid polyoxyethylene glycols, propylene glycol, 2-pyrrolidone, N-methylpyrrolidone, diethylene glycol monoethyl ether, ethylene glycol, triacetin, Ci-Ci0 esters of carboxylic acids such as butyl or octyl acetate, glycerol formal, dialkyl esters of dicarboxylic acids including diethyl sebacate, diisopropyl sebacate, diisopropyl adipate and the like, and diethyl phthalate, or a mixture of at least two of these solvents.
The solvent will be used in proportion with the concentration of the active agent compound and its solubility in this solvent. It will be sought to have the lowest possible volume. The vehicle makes up the difference to 100%.
A vehicle or diluent for the formulations may include dimethyl sulfoxide (DMSO), glycol derivatives such as, for example, propylene glycol, glycol ethers, polyethylene glycols or glycerol. As vehicle or diluent, mention may also be made of plant oils such as, but not limited to soybean oil, groundnut oil, castor oil, corn oil, cotton oil, olive oil, rape seed oil, sunflower oil, etc.; mineral oils such as, but not limited to, petrolatum, paraffin, silicone, etc.; aliphatic or cyclic hydrocarbons or alternatively, for example, medium-chain (such as C8 to C12) triglycerides.
An emollient and/or spreading and/or film-forming agent may be added. Examples of a emollient and/or spreading and/or film-forming agent may be:
(a) polyvinylpyrrolidone, polyvinyl alcohols, copolymers of vinyl acetate and vinylpyrrolidone, polyethylene glycols, benzyl alcohol, mannitol, glycerol, sorbitol,
polyoxyethylenated sorbitan esters; lecithin, sodium carboxymethylcellulose, silicone oils, polydiorganosiloxane oils (such as polydimethylsiloxane (PDMS) oils), for example those containing silanol functionalities, or a 45 V2 oil,
(b) anionic surfactants such as alkaline stearates, sodium, potassium or ammonium stearates; calcium stearate, triethanolamine stearate; sodium abietate; alkyl sulphates (e.g. sodium lauryl sulphate and sodium cetyl sulphate); sodium dodecylbenzenesulphonate, sodium dioctylsulphosuccinate; fatty acids (e.g. those derived from coconut oil),
(c) cationic surfactants include water-soluble quaternary ammonium salts of formula N R'R"R"'R" ", Y" in which the radicals R are optionally hydroxylated hydrocarbon radicals and Y" is an anion of a strong acid such as the halide, sulphate and sulphonate anions; cetyltrimethylammonium bromide is among the cationic surfactants which can be used,
(d) amine salts of formula N+ HR'R"R" in which the radicals R are optionally hydroxylated hydrocarbon radicals; octadecylamine hydrochloride is among the cationic surfactants which can be used,
(e) nonionic surfactants such as sorbitan esters, which are optionally polyoxyethylenated (e.g. polysorbate 80), polyoxyethylenated alkyl ethers; polyoxypropylated fatty alcohols such as polyoxypropylene-styrol ether; polyethylene glycol stearate, polyoxyethylenated derivatives of castor oil, polyglycerol esters, polyoxyethylenated fatty alcohols, polyoxyethylenated fatty acids, copolymers of ethylene oxide and propylene oxide, (f) amphoteric surfactants such as the substituted lauryl compounds of betaine; or
(g) a mixture of at least two of these agents.
The amount of emollient that may be used, for example, may be in a proportion of from about 0.1 to 50% or 0.25 to 5%, by volume. Alternatively, the emollient used may be in a proportion of from about 0.1% to about 30%, about 1% to about 30%, about 1% to about 20%, or about 5%) to about 20% by volume.
Alternatively, the parasiticidal composition may be in the form of a ready-to-use spot-on solution form as is described in U. S. Patent Nos. 6,395,765 and 9, 180, 121, both incorporated herein by reference. In addition to the bioactive agent(s), the ready-to-use solution may contain a crystallization inhibitor and an organic solvent or a mixture of organic solvents. In some of these formulations, water may be included with the organic solvent.
The topical formulations may include a crystallization inhibitor in an amount of about 1 to about 50% (w/v) or about 5 to about 40% (w/v) based on the total weight of the formulation. In other embodiments, the amount of crystallization inhibitor in the inventive formulations may be about 1% to about 30%, about 5% to about 20%, about 1% to about 15%, or about 1% to about 10%) (w/w). The type of crystallization inhibitor used in the topical formulations is not limited as long as it functions to inhibit crystallization of the active or inactive agents from the formulation. For example, in certain embodiments of the invention, a solvent or co-solvent of the formulation may also function as a crystallization inhibitor if it sufficiently inhibits the formation of crystals from forming over time when the formulation is administered.
Crystallization inhibitors which are useful include, but are not limited to:
(a) polyvinylpyrrolidone, polyvinyl alcohols, copolymers of vinyl acetate and vinylpyrrolidone, polyethylene glycols, benzyl alcohol, dimethylformamide, dimethylacetamide, dimethylsulfoxide, 2-pyrrolidone, N-methylpyrrolidone, mannitol, glycerol, sorbitol or polyoxyethylenated esters of sorbitan; lecithin or sodium carboxymethylcellulose; or acrylic derivatives, such as acrylates or methacrylates or polymers or copolymers thereof, PEG or polymers containing polyethyleneglycols, such as glycofurol and the like, and others;
(b) anionic surfactants, such as alkaline stearates (e.g. sodium, potassium or ammonium stearate); calcium stearate or triethanolamine stearate; sodium abietate; alkyl sulphates, which include but are not limited to sodium lauryl sulphate and sodium cetyl sulphate; sodium dodecylbenzenesulphonate or sodium dioctyl sulphosuccinate; or fatty acids (e.g. coconut oil);
(c) cationic surfactants, such as water-soluble quaternary ammonium salts of formula N R'R"R'"R" "Y~, in which the R radicals are identical or different optionally hydroxylated hydrocarbon radicals and Y is an anion of a strong acid, such as halide, sulphate and sulphonate anions; cetyltrimethylammonium bromide is one of the cationic surfactants which can be used;
(d) amine salts of formula N+HR'R"R", in which the R radicals are identical or different optionally hydroxylated hydrocarbon radicals; octadecylamine hydrochloride is one of the cationic surfactants which can be used;
(e) non-ionic surfactants, such as optionally polyoxyethylenated esters of sorbitan, e.g. Polysorbate 80, or polyoxyethylenated alkyl ethers; polyethylene glycol stearate, polyoxyethylenated derivatives of castor oil, polyglycerol esters, polyoxyethylenated fatty
alcohols, polyoxyethylenated fatty acids or copolymers of ethylene oxide and of propylene oxide;
(f) amphoteric surfactants, such as substituted lauryl compounds of betaine;
(g) a mixture of at least two of the compounds listed in (a)-(f) above; or
(h) an organic solvent or mixture of solvents which inhibit the formation of crystals or amorphous solid after the formulation is administered.
Alternatively, a crystallization inhibitor pair maybe used. Such pairs include, for example, the combination of a film-forming agent of polymeric type and of a surface-active agent. These agents will be selected from the compounds mentioned above as crystallization inhibitor.
The organic solvent(s) may have a dielectric constant of between about 10 and about 35 or between about 20 and about 30. In other topical formulations, the organic solvent may have a dielectric constant of between about 10 and about 40 or between about 20 and about 30. The content of this organic solvent or mixture of solvents in the overall composition is not limited and will be present in an amount sufficient to dissolve the desired components to a desired concentration. As discussed above, the organic solvent may also function as a crystallization inhibitor in the formulation.
In some topical formulations, one or more of the organic solvent(s) may have a boiling point of below about 100° C, or below about 80° C. In other topical formulations, the organic solvent(s) may have a boiling point of below about 300° C, below about 250° C, below about 230°C, below about 210° C or below about 200° C.
Alternatively, the topical formulations may contain a mixture of solvents, i.e. a solvent and a co-solvent, the solvents may be present in the composition in a weight/weight (WAV) ratio of about 1/50 to about 1/1. Typically the solvents will be in a ratio of about 1/30 to about 1/1, about 1/20 to about 1/1, or about 1/15 to about 1/1 by weight. Preferably, the two solvents will be present in a weight/weight ratio of about 1/15 to about 1/2. In some topical formulations, at least one of the solvents present may act as to improve solubility of the active agent or as a drying promoter. In some of the topical formulations, at least one of the solvents will be miscible with water.
The formulation may also comprise an antioxidizing agent intended to inhibit oxidation in air, this agent may be present in a proportion of about 0.005 to about 1% (w/v), about 0.01 to about 0.1%, or about 0.01 to about 0.05%.
Examples of film-forming agent include agents are of the polymeric type which include but are not limited to the various grades of polyvinylpyrrolidone, polyvinyl alcohols, and copolymers of vinyl acetate and of vinylpyrrolidone.
Surface-active agents may be included. These agents include but are not limited to those made of non-ionic surfactants; in another embodiment of the surface active agents, the agent is a polyoxyethylenated esters of sorbitan and in yet another embodiment of the surface-active agent, the agents include the various grades of polysorbate, for example Polysorbate 80.
The film-forming agent and the surface-active agent may be incorporated in similar or identical amounts within the limit of the total amounts of crystallization inhibitor mentioned elsewhere.
Alternatively, the topical compositions include the compound crotamiton which may inhibit the crystallization of active agents from solution.
The crystallization inhibitor inhibits the formation of crystals on the coat, and improves the maintenance of the cosmetic appearance of the skin or fur; that is to say without a tendency towards sticking or towards a sticky appearance, despite the high concentration of active material. Substances other than those mentioned herein may be used as crystallization inhibitors in the present invention. The effectiveness of the crystallization inhibitor may be demonstrated by a test according to which 0.3 mL of a solution comprising 10% (w/v) of the active agent in an appropriate solvent as defined above, and 10% (w/v) of the compound acting as a crystallization inhibitor are placed on a glass slide at 20° C for 24 hours, after which fewer than 10 crystals, preferably 0 crystals, are seen with the naked eye on the glass slide.
Antioxidizing agents may be included. These agents are those conventional in the art and include but are not limited to BHA, butylated hydroxy toluene (BHT), ascorbic acid, sodium metabi sulphite, propyl gallate, sodium thiosulphate or a mixture of at least two compounds with antioxidant properties.
The formulation adjuvants discussed above are well known to the practitioner in this art and may be obtained commercially or through known techniques. These concentrated compositions are generally prepared by simple mixing of the constituents as defined above;
advantageously, the starting point is to mix the active material in the main solvent and then the other ingredients or adjuvants are added.
The volume of the formulation applied will depend on the type of animal and the size of the animal as well as the strength of the formulation and the potency of the active agents. In one embodiment, an amount of about 0.3 to about 20 ml of the formulation may be applied to the animal. In other embodiment for the volume, the volume may be about 0.1 to about 10 ml, about 0.3 to about 5 ml, about 0.5 ml to about 10 ml, or about 0.3 to about 3 ml.
In another embodiment of the inventive methods, application of a spot-on formulation according to the present invention may also provide long-lasting and broad-spectrum efficacy when the solution is applied to the mammal or bird. The spot-on formulations provide for topical administration of a concentrated solution, suspension, microemulsion or emulsion for intermittent application to a spot on the animal, generally between the two shoulders (solution of spot-on type).
For spot-on formulations, the carrier may be a liquid carrier vehicle as described in U.S. Patent Nos. 6,426,333; 6,395,765 (incorporated herein by reference), which in may comprise a solvent or mixture of solvents including, but not limited to, acetone, an aliphatic alcohol such as methanol, ethanol, propanol, butanol, isopropanol, pentanol, hexanol, heptanol, octanol, nonanol, cyclopentanol, cyclohexanol, ethylene glycol, propylene glycol and the like; an aromatic alcohol such as phenol, cresol, naphthol, benzyl alcohol and the like; acetonitrile, butyl diglycol, an organic amide such as dimethylacetamide, dimethylformamide, monomethylacetamide, 2- pyrrolidone, N-methylpyrrolidone, vinylpyrrolidone and the like; propylene or ethylene carbonate, dimethyl sulfoxide (DMSO), a glycol polymer or an ether thereof, such as polyethylene glycol (PEG) of various grades, polypropylene glycols of various grades, dipropylene glycol n-butyl ether, ethylene glycol monoethyl ether, ethylene glycol monomethyl ether, dipropylene glycol monomethyl ether, diethylene glycol monoethyl ether, ethylene glycol, diethyl phthalate fatty acid esters, such as the diethyl ester or diisobutyl adipate, or a mixture of at least two of these solvents.
Solvents which may be used for the spot-on or pour-on compositions of the invention include those described in US 9, 180,121 (incorporated by reference). These solvents include, but are not limited to, carboxylic acid esters, diesters of dicarboxylic acids, fatty acid esters or diesters of fatty diacids, or a combination thereof, including, but not limited to, isopropyl
palmitate, isostearyl lactate, diisopropyl adipate, dibutyl adipate, diethyl sebacate, dibutyl sebacate, octyl palmitate, polyethyleneglycol (PEG) stearate and cetearyl octanoate; oils including, but not limited to, mineral oil, diglycerides, triglycerides, jojoba oil, lecithin and castor oil, or a combination thereof; long chain aliphatic alcohols such as isostearyl alcohol and the like; fatty alcohols and their esters, including for example, cetyl alcohol, cetearyl alcohol and the like, or a combination thereof; polyethylene glycols of different molecular weight ranges including, but not limited to, PEG 300, PEG 400, PEG 600 and PEG 1000, or a combination thereof; and glycol ethers including, but not limited to, diethyleneglycol monoethyl ether (Transcutol®), butyl diglycol, propylene glycol monomethyl ether, propylene glycol monoethyl ether, dipropylene glycol n-butyl ether, ethylene glycol monoethyl ether, ethylene glycol monomethyl ether and dipropylene glycol monomethyl ether, or a combination thereof; or a combination of two or more of these solvents.
Excipients that may promote the containment of the active agent in the skin for longer periods of time and may be included in the compositions of the invention include, but are not limited to, mixed esters of sucrose and carboxylic acids including sucrose acetate isobutyrate (SAIB) and the like; low temperature melting waxes, hydrogenated vegetable oils, caprylic/capric glycerides; glycerol esters, including for example, triacetin, glycerol monooleate, glycerol monolinoleate, glycerol stearate, glyceryl distearate and the like; triglycerides, including for example, caprylic, capric/ myristic/ stearic triglyceride; thermoreversible polymers, such as Pluronic and poloxamers, including for example, Lutrol F127 by itself or in mixture with other poloxamers; or a combination thereof.
In some circumstance the pharmaceutically acceptable carrier for the topical compositions comprise a mixture of a diester of a dicarboxylic acid alone or in combination with one or more of additional solvents listed above, and/or an "oily" lipophilic substance, including a liquid or low melting lipophilic active agent such as (^-methoprene, pyriproxyfen and/or permethrin; and/or a mixed ester of sucrose and carboxylic acids including a mixed ester of sucrose and acetic and isobutyric acids such as sucrose acetate isobutyrate (SAIB), and/or low melting waxes and/or hard fats.
Diethyl sebacate or diisopropyl adipate are examples for a diester of a dicarboxylic acid. The blends of solvents comprising a dicarboxylic acid ester comprises a glycol or polyglycol, or a glycol or polyglycol ether or ester including, but not limited to, ethylene glycol (EG),
propylene glycol (PG), liquid PEGs of various grades including PEG 400, EG or PG monocaprylate, EG or PG caprylate, EG or PG monolaurate, EG or PG dicaprylate/dicaprate, diethyleneglycol monoethyl ether (DGME, Transcutol®), butyl diglycol, dipropylene glycol n- butyl ether, ethyleneglycol monoethyl ether, ethyleneglycol monomethyl ether, dipropylene glycol monomethyl ether, propylene glycol monomethyl ether, propylene glycol monoethyl ether, and the like, or a combination thereof; an ether including, but not limited to, dimethyl isosorbide; an ester or di-ester including, but not limited to, triacetin, lauryl lactate; and other solvents including glycerol formal, or mixtures thereof.
The carrier for the topical compositions may comprise a dialkyl ester of a dicarboxylic acid, such as diethyl sebacate, diisopropyl sebacate, diisopropyl adipate, dibutyl adipate, or a combination thereof, alone or in combination with solvents selected from:
a propylene glycol (PG) ester including PG monocaprylate, PG caprylate, PG monolaurate, PG dicaprylate / dicaprate, or a combination thereof;
an ether solvent including dimethyl isosorbide, diethylene glycol monoethyl ether (also known as DGME or Transcutol®), or a combination thereof;
a carboxylic acid ester including, but not limited to, triacetin, lauryl lactate, isopropyl palmitate, diisopropyl sebacate, or a combination thereof; and other "oily" or lipophilic organic solvents including glycerol formal and the like.
The amount the additional solvents combined with the carboxylic acid ester or diester of a dicarboxylic acid are present in an amount of at least about 1% (v/v), at least about 5% (v/v), at least about 9.0% (v/v), at least about 13% (v/v), at least about 17% (v/v) or at least about 20% (v/v). Preferably the additional solvents will be in an amount of at least about 9% (v/v).
The additional solvents will be present in an amount of about 5-70% (v/v), about 10-60% (v/v), about 10-50% (v/v), about 15-60% (v/v) or about 15-50% (v/v). Alternatively, the additional solvents will be present in an amount of about 20-70% (v/v), about 20-60% (v/v), about 20-50% (v/v) or about 25-50% (v/v).
The liquid carrier vehicle may optionally contain a crystallization inhibitor including, but not limited to, those described in (a) to (h) above, or a compound that may act both as a solvent and a crystallization inhibitor (as defined above), or a mixture of these crystallization inhibitors.
Spot-on formulations may be prepared by dissolving the active ingredients into the pharmaceutically or veterinary acceptable vehicle. Alternatively, the spot-on formulation may be prepared by encapsulation of the active ingredient to leave a residue of the therapeutic agent on the surface of the animal. These formulations will vary with regard to the weight of the therapeutic agent in the combination depending on the species of host animal to be treated, the severity and type of infection and the body weight of the host.
Dosage forms may typically contain from about 0.1 mg to about 5 g. In other embodiments, the dosage form may contain about 0.5 mg to about 5 g of an active agent. In one embodiment of the dosage form, the dosage may contain from about 1 mg to about 500 mg of an active agent, typically about 25 mg, about 50 mg, about 100 mg, about 200 mg, about 300 mg, about 400 mg, about 500 mg, about 600 mg, about 800 mg, or about 1000 mg.
For example, the bioactive agent may be present in the formulation at a concentration of about 0.05 to about 10% weight/volume. Alternatively, the bioactive agent may be present in the formulation as a concentration from about 0.1 to about 2% weight/volume or the bioactive agent may be present in the formulation as a concentration from about 0.25 to about 1.5% weight/volume. Alternatively, the bioactive agent may be present in the formulation as a concentration about 1% weight/volume.
C. Long Acting Injectable Formulations
The parasiticidal formulations used in the inventive methods may include long-acting injectable formulations. These formulations comprise at least one bioactive agent (e.g., an isoxazoline compound) and optionally one or more bioactive agent(s), at least one pharmaceutically acceptable non-biodegradable polymer and a co-solvent. Pharmaceutically acceptable non-biodegradable polymers include, but are not limited to, PEGs and poloxamers. Long-acting injectable formulations are active for an extended period of time; e.g., a period from at least two (2) (e.g., three (3), four (4), five (5)) to six (6) months.
In one embodiment of the invention, a particularly advantageous long acting injectable formulation is used. This formulation comprises an isoxazoline bioactive agent, at least one liquid PEG and a co-solvent and excludes other bioactive polymers, such as those identified above. The formulations have the advantage of exhibiting favorable properties with respect to the injection site, while being effective for an extended period of time; they are described in U.S. Provisional Patent Application No. 62/299,333, herein incorporated by reference.
Liquid PEGs as provided in these formulations are those polyethylene glycols that are liquid at room temperature (20 - 30 °C). Polyethylene glycols have the following structural formula:
H-(0-CH2-CH2)n-OH.
Non-limiting examples for n in the above formula are those compounds when n is from 1 to about 10,000 (e.g., from about 4 to about 25). Liquid PEGs include combinations of different polyethylene glycols. Non-limiting examples of liquid PEGs include PEG 200, PEG 300, PEG 400, PEG 600, and PEG 1000 or combinations thereof.
In another embodiment, the invention provides long acting injectable compositions that comprise an isoxazoline active agent in a carrier that comprises a poloxamer and optionally a co-solvent. Poloxamers are a family of synthetic block copolymers of ethylene oxide and propylene oxide. Poloxamers may be liquid, a milky white paste or a powder and are represented by the following structure:
where a is an integer between 2 and 130 and b is an integer between 15 and 67 (see, US 3,740,421). Poloxamer are available from commercial sources such as BASF and Croda. An example of a poloxamer is P-124 which is a solid at room temperature. In one embodiment, poloxamer P-124 has the values a = 12 and b = 20. Other poloxamers include P-128 (a = 38 and b = 29), P-181 (a = 3 and b = 30) P-188 (a = 80 and b =27), P-237 (a = 64 and b = 37), P338(a = 141 and b = 44,) and P407(a = 101 and b = 56,). When present, the amount of poloxamer, may be, for example, from about 0.5% to about 20 (w/w); from about 1% to about 20% (w/w); from about 1%) to about 10% (w/w); or from about 1 to about 5% (w/w). In other extended release injectable formulations, the amount of poloxamer, when present, is from about 1 % to about 3 % (w/w).
The co-solvents used in the long acting injectable compositions may be a single or a blend of co-solvents. The co-solvents used in the long-acting injectable formulations of the present invention include polar solvents that are miscible in water. Non-limiting examples of
these co-solvents include ethanol, isopropanol, glycol ethers (e.g., including, but limited to, diethyleneglycol monoethyl ether (DGME, Transcutol®), butyl diglycol, dipropylene glycol n- butyl ether, ethyleneglycol monoethyl ether, ethyleneglycol monomethyl ether, dipropylene glycol monomethyl ether, propylene glycol monomethyl ether, propylene glycol monoethyl ether, and the like), propylene glycol, carbonates (e.g., propylene carbonate), 2-pyrrolidone, N- methylpyrrolidone, dimethyl isosorbide (DMI), Dimethylacetamide, glycerol formal or a mixture of at least two of these solvents.
The long acting formulations may comprise a polar protic solvent including, but not limited to, an alcohol such as ethanol, isopropanol or a glycol or glycol ether.
Alternatively, the long-acting injectable formulations may comprise a polar aprotic solvent such as N-methylpyrrolidone, dimethyl isosorbide, dimethylacetamide or propylene carbonate.
Alternatively, the long-acting injectable formulations may include non-water miscible co- solvents or solvents with only partial solubility in water. Non-limiting examples of these co- solvents include benzyl alcohol, triacetin, lipids, triglycerides including medium chain triglycerides such C8-Cio triglycerides such as capric/caprilic triglycerides, propylene glycol derivatives (e.g. propylene glycol monolaurate), caprylocaproyl polyoxyl-8 glycerides (Labrasol) (non-ionic water dispersible surfactant, isopropyl myristate, or a mixture of at least two of these co-solvents.
Moreover, the long acting injectable formations may include neutral oils as a co-solvent.
Neutral oils are triglycerides of fractionated plant fatty acids with chain lengths of C8 to C10. Two commercially available products are known as MIGLYOL® 810 and MIGLYOL®812. In another embodiment, the neutral oil is a triglyceride of fractionated plant fatty acids with chain lengths of C8 and C10 combined with linoleic acid (about 4-5 %). A commercially available product is known as MIGLYOL® 818. In yet another long acting injectable formulation, the neutral oil is a glycerin ester of fractionated plant fatty acids with chain lengths of C8 and C10 combined with succinic acid. A commercially available product is known as MIGLYOL® 829. In another embodiment, the neutral oil may be a propylene glycol diester of saturated plant fatty acids with chain lengths of C8 and C10 combined with succinic acid. A commercially available product is known as MIGLYOL® 840 (propylene glycol dicaprylate/dicaprate). In yet another embodiment, the co-solvent may be a mixture of two or more neutral oils.
The long acting injectable formulations may contain other inert ingredients such as antioxidants, preservatives, or pH stabilizers. These compounds are well known in the formulation art. Antioxidants such as vitamin E, alpha tocopherol, ascorbic acid, ascorbyl palmitate, citric acid, fumaric acid, malic acid, sodium ascorbate, sodium metabi sulfate, sodium metabi sulfite, n-propyl gallate, BHA, BHT, BHA and citric acid, monothioglycerol, tert-butyl hydroquinone (TBHQ), and the like, may be added to the present formulation. The antioxidants are generally added to the formulation in amounts of from about 0.01 to about 2.0%, based upon total weight of the formulation, with about 0.05 to about 1.0% being especially preferred.
Preservatives, such as the parabens (methylparaben and/or propylparaben), are suitably used in the formulation in amounts ranging from about 0.01 to about 2.0%>, with about 0.05 to about 1.0% being especially preferred. Other preservatives include benzalkonium chloride, benzethonium chloride, benzoic acid, benzyl alcohol, bronopol, butylparaben, cetrimide, chlorhexidine, chlorobutanol, chlorocresol, cresol, ethylparaben, imidurea, methylparaben, phenol, phenoxyethanol, phenylethyl alcohol, phenylmercuric acetate, phenylmercuric borate, phenylmercuric nitrate, potassium sorbate, sodium benzoate, sodium propionate, sorbic acid, thimerosal, and the like. Preferred ranges for these compounds include from about 0.01 to about 5%.
Compounds which stabilize the pH of the formulation are also contemplated. Again, such compounds are well known to a practitioner in the art as well as how to use these compounds. Buffering systems include, for example, systems selected from the group consisting of acetic acid/acetate, malic acid/malate, citric acid/citrate, tartaric acid/tartrate, lactic acid/lactate, phosphoric acid/phosphate, glycine/glycimate, tris, glutamic acid/glutamates and sodium carbonate.
D. Extended Release Injectable Formulations
In other embodiments the parasiticidal compositions may be extended release injectable formulations. Extended release injectable formulations comprise a bioactive agent or agents, a pharmaceutically acceptable polymer (e.g., PLGA polymer) and at least one solvent; optional ingredients include a surfactant, a pharmaceutically acceptable additive or excipient. Extended- release injectable formulations comprising an isoxazoline compound as a bioactive agent are described, for example in application PCT/US2016/026253, herein incorporated by reference.
The pharmaceutically acceptable polymers in the extended release injectable
formulations, include, but are not limited to, polylactides, polyglycolides, polycaprolactones, polyanhydrides, polyamides, polyurethanes, polyesteramides, polyorthoesters, polydioxanones, polyacetals, polyketals, polycarbonates, polyorthocarbonates, polyphosphazenes, pseudo poly(amides), polyhydroxyalcanoates, polyhydroxybutyrates, polyhydroxyvalerates, polyalkylene oxalates, polyalkylene succinates, poly(malic acid), poly(amino acids), poly(methyl vinyl ether), poly(maleic anhydride), chitin, chitosan, and copolymers, terpolymers, or combinations or mixtures therein including copolymers of polylactides, polycaprolactones, polyglycolides (e.g., poly(lactide-co-glycolide) and copolymers of polyethylene glycol or methoxy polyethylene glycol with one or more of polycaprolactone, polylactide or any of the other polymers/polymer groups mentioned above. In addition to the pharmaceutically acceptable polymers noted above, the extended release compositions may also include pharmaceutically acceptable polymers that are not biodegradable such as PEGs and poloxamers. Also included are derivatives of pharmaceutically acceptable polymers such as hydroxylated derivatives including polycaprolactone diols and the like.
The pharmaceutically acceptable polymer may be, for example, a biodegradable polymer and might have one or more or all of the following characteristics: be bioerodible by cellular action, biodegradable by action of non-living body fluid components, soften when exposed to heat but return to the original state when cooled and are capable of substantially dissolving or dispersing in a water-miscible carrier or solvent to form a solution or dispersion. Upon contact with an aqueous fluid the polymer is capable of assisting in the formation of the film coated or encapsulated liquid or solid (which will contain the active agent in the present invention). The kinds of polymers suitable for the present composition generally include any having the foregoing characteristics. Examples of biodegradable polymers include, but are not limited to, polylactides, polycaprolactones, polyglycolides, polyorthoesters, polyurethanes, polyphasphazenes, pseudo poly(amides), and copolymers thereof.
It will be apparent to the skilled person that the molecular weight of a polymer is not a discreet number but can be presented in a molecular weight range. The average molecular weight of a polymer may be found by techniques familiar to persons of skill in the art, for example, size exclusion chromatography with molecular weight standards, or the like. The molecular weight range of a polymer can impact the physical characteristics of the material and the way that it interacts with the active agent. Accordingly the molecular weight range of the polymer may
impact the characteristics of the extended release compositions of the invention. For example, in some formulations, depending on the bioactive agent(s) and solvents included, one may see an earlier release of the isoxazoline active agent when the weight average molecular weight range is from about 5 to about 20 kDa (kilo daltons) or from about 7 to about 15 kDa. A later release of the isoxazoline active agent may be observed when the weight average molecular weight is between about 30 to about 70 kDa (e.g., about 40 to about 70 kDa or about 45 to about 60 kDa) or from about 90 to about 200 kDa (e.g., about 100 to about 150 or about 105 to about 130 kDa). In some formulations, a combination of polymers having different average molecular weights may provide a release rate that combines the effect of the different polymers used.
Inherent viscosity (IV) in polymer chemistry is a viscometric method for measuring molecular weight. It is defined as the ratio of the natural logarithm of the relative viscosity to the mass concentration of the polymer and is based on the flow time of a polymer solution through a narrow capillary. As used herein with respect to the molecular weight range of a pharmaceutically acceptable polymer, the term "low molecular weight" (LMW) refers to polymer with an inherent viscosity in the range of 0.05-0.29 dL/g; the term "medium molecular weight" refers to a polymer with an inherent viscosity in the range of 0.3-0.55dL/g; and high molecular weight refers to a polymer with an inherent viscosity in the range of 0.55-1.0 dL/g. In another formulation, the pharmaceutically acceptable polymer will have an inherent viscosity of about 0.10-0.20 dL/g. In another formulation, the pharmaceutically acceptable polymer will have an inherent viscosity of about 0.35-0.50.
The extended release injectable formulations may comprise, for example, polylactides, polycaprolactones, polyglycolides and copolymers thereof. In another embodiment, the compositions include a poly(lactide-co-glycolide) copolymer (PLGA). PLGA copolymers may have different molecular weight ranges and may also have different weigh weight ratios of lactide to glycolide. This ratio, may affect the properties of the copolymer and the way that it interacts with the active agent. Since a lactide group contains an additional methyl group in the sidechain compared with a glycolide, this change may affect the conformation of the polymer and change the way in which the polymer interacts with the isoxazoline active agent (and/or other active agent combined with the isoxazoline). Although not bound by theory, in one embodiment, extended-release injectable formulations having a higher lactide to glycolide ratio (e.g. 75 :25 compared with 50:50) result in an increase in hydrogen bonding between the active
agent and the polymer, leading to better solubility of the active agent in vivo. This effect improves the injection site reaction and allows for the extendable release injectable compositions to include a higher amount of the active agent, which will translate to a longer duration of efficacy.
In some extended release formulations, when the pharmaceutically acceptable polymer is
PLGA, the ratio of lactide to glycolide is about 30:70 to about 99: 1. In other formulations, where the pharmaceutically acceptable polymer is PLGA, the ratio of lactide to glycolide is about 40:60 to about 80: 15. In other formulations, where the pharmaceutically acceptable polymer is PLGA, the ratio of lactide to glycolide is about 40:60 to about 60:40. In another embodiment of the formulations, where the pharmaceutically acceptable polymer is PLGA, the ratio of lactide to glycolide is about 70:30 to about 80:20. In another embodiment, where the pharmaceutically acceptable polymer is PLGA, the ratio of lactide to glycolide is about 50:50. In another embodiment, where the pharmaceutically acceptable polymer is PLGA, the ratio of lactide to glycolide is about 75:25.
In some extended release formulations, the amount of PLGA contained in the extended release injectable formulation is about 1% to about 30 % (w/w), In another embodiment, the compositions comprise about 1 to about 20% (w/w) of PLGA. In another embodiment, the compositions comprise about 5 to about 20%, about 8% to about 20% (w/w) or about 10 to about 20%) (w/w). In another embodiment, the formulations comprise about 5 to about 15% (w/w) of PLGA. In other embodiments, the amount of PLGA contained in the extended release injectable formulations is from about 3% to about 15% (w/w) or is from about 10% to about 15% (w/w). In yet another embodiment, the formulations comprise about 7 to about 13% or about 8 to about 15% (w/w) of PLGA.
In some extended release formulations, the weigh weight ratio of PLGA to the isoxazoline active agent is greater than or equal to about 1 : 1, for example, from about 1.1 : 1 to about 20: 1; e.g., about 1 : 1 to about 10: 1, about 1.1 : 1 to about 10: 1 or about 2: 1 to about 5: 1. In other formulations, the weigh weight ratio of PLGA to the isoxazoline active agent is about 1.2: 1 to about 5: 1. In another formulation, the weigh weight ratio of PLGA to the isoxazoline active agent is about 1.2: 1 to about 2: 1. In yet another formulation, the weight: weight ratio of PLGA to the isoxazoline active agent is about 1.2: 1 to about 1.3 : 1.
In other extended release formulations the weightweight ratio of PLGA to the isoxazoline active agent is about 1.5: 1 to about 1 : 1.5. In other formulations, the ratio of the isoxazoline active agent to PLGA is from about 1.25: 1 to about 1 : 1 :25.
The solvents used in theses extended-release injectable formulations may be a single or a blend of solvents. Non-limiting examples of these solvents include alcohols such as ethanol, 1- propanol, isopropanol, glycol ethers (e.g., including, but limited to, diethyleneglycol monoethyl ether (DGME, Transcutol®), butyl diglycol, dipropylene glycol n-butyl ether, ethyleneglycol monoethyl ether, ethyleneglycol monomethyl ether, dipropylene glycol monomethyl ether, propylene glycol monomethyl ether, propylene glycol monoethyl ether, and the like), liquid polyethylene glycols (PEGs) including, but not limited to, PEG 200, PEG 300 and PEG 400; propylene glycol, glycerol, glycerol esters including glycerol triacetate (triacetin), cyclic carbonates (e.g., ethylene carbonate and propylene carbonate), 2-pyrrolidone, N- methylpyrrolidone, dimethyl isosorbide (DMI), dimethylacetamide (DMA), dimethyl formamide (DMF), caprolactam, glycerol formal, acetone, dimethylsulfoxide (DMSO), ethyl acetate, ethyl lactate, benzyl benzoate, or a mixture of at least two of these solvents.
The extended-release injectable formulations may include one or more poloxamers as a solvent or surfactant.
The extended release injectable formulations may comprise a solvent or mixture of solvents that is miscible with water. Solvents that are miscible with water are well known and include certain alcohols, liquid PEGs, certain poloxamers, glycols and glycol ethers and polar aprotic solvents. Alcohols that are miscible with water include, but are not limited to ethanol, isopropanol, «-propanol, Solketal (isopropylidene glycerol) or glycerol formal. Polar aprotic solvents include, but are not limited to, amides such as dimethylacetamide, dimethylformamide, 2-pyrrolidone, N-alkylpyrrolidones such as N-methylpyrrolidone and N-octylpyrrolidone, dimethylisosorbide, dimethylsulfoxide, cyclic carbonates including propylene carbonate and ethylene carbonate, and certain ketones such as acetone and the like. Glycol ethers include, but are not limited to, diethyleneglycol monoethyl ether (DGME, Transcutol®), butyl diglycol, dipropylene glycol n-butyl ether, ethyleneglycol monoethyl ether, ethyleneglycol monomethyl ether, dipropylene glycol monomethyl ether, propylene glycol monomethyl ether, propylene glycol monoethyl ether, and the like.
The extended release formulations may comprise, for example, a polar protic solvent
including, but not limited to, an alcohol such as ethanol, isopropanol or a glycol or glycol ether.
In another extended release injectable formulation, the formulation may comprise, for example, a polar aprotic solvent such as N-methylpyrrolidone, dimethyl isosorbide, dimethylacetamide or propylene carbonate.
The extended release injectable formulations may include non-water miscible solvents
(e.g. not completely miscible with water, although they may have some solubility in water). Non- limiting examples of these solvents include 1-butanol, 2-butanol, 1-pentanol, 3-pentanol, benzyl alcohol, methylethylketone (MEK), triacetin, lipids, triglycerides including medium chain triglycerides such C8-Cio triglycerides such as capric/caprilic triglycerides, propylene glycol derivatives (e.g. propylene glycol monolaurate), caprylocaproyl polyoxyl-8 glycerides (Labrasol) (non-ionic water dispersible surfactant, isopropyl myristate, oils such as castor oil, soybean oil or other vegetable oils or derivatives thereof such as epoxidized or hydrogenated vegetable oils such as epoxidized soybean oil or hydrogenated castor oil, or a mixture of at least two of these solvents.
Alternatively, the extend release formulations may include neutral oils as a solvent.
Neutral oils are triglycerides of fractionated plant fatty acids with chain lengths of C8 to Cio. Two commercially available products are known as MIGLYOL® 810 and MIGLYOL®812. In another embodiment, the neutral oil is a triglyceride of fractionated plant fatty acids with chain lengths of C8 and Ci0 combined with linoleic acid (about 4-5 %). A commercially available product is known as MIGLYOL® 818. In yet another extended release injectable formulation, the neutral oil is a glycerin ester of fractionated plant fatty acids with chain lengths of C8 and Cio combined with succinic acid. A commercially available product is known as MIGLYOL® 829. In yet another formulation, the neutral oil is a propylene glycol fatty acid ester. In one embodiment, the neutral oil may be a propylene glycol diester of saturated plant fatty acids with chain lengths of C8 and Cio. A commercially available product is known as MIGLYOL® 840 (propylene glycol dicaprylate/dicaprate). In yet another extended release injectable formulation, the solvent may be a mixture of two or more neutral oils.
It will be appreciated that blends of solvents may be used as the solvent of the extended release injectable formulations. In one embodiment, the compositions of the invention may contain a blend of a water-miscible solvent with a solvent that is not water miscible. For example, the solvent may be a mixture of a cyclic carbonate such as propylene carbonate with
triacetin. Of course, other blends of a water-miscible solvent and a non-water miscible solvent are possible. In one long acting injectable formulation, the water-miscible solvent in the solvent blend may be a water-miscible alcohol such as ethanol or isopropanol, glycerol formal or Solketal, an amide such as 2-pyrrolidone, N-methylpyrrolidone, dimethylisosorbide or dimethylacetamide, a glycol such as propylene glycol, glycerol or a glycol ether.
In another long acting injectable formulations, the non-water miscible solvent in the solvent blend may be, for example, triacetin, benzyl alcohol, a triglyceride including C8-Cio triglycerides such as capric/caprilic triglycerides, propylene glycol derivatives (e.g. propylene glycol monolaurate), caprylocaproyl polyoxyl-8 glycerides (Labrasol); a propylene glycol fatty acid diester, and the like.
In one embodiment, the solvent in the extended release formulations in the inventive methods may be a blend of a water-miscible solvent and a non-water miscible solvent in a weight: weight ratio of between about 10 to 1 to about 1 to 10, water-miscible solvent to non- water miscible solvent. In another embodiment, the weigh weight ratio of the water-miscible solvent to non-water miscible solvent may be from about 5 to 1 to about 1 to 1. In another embodiment, the weigh weight ratio of the water-miscible solvent to non-water miscible solvent may be from about 3 to 1 to about 1 to 1. In another embodiment, the weight: weight ratio of the water-miscible solvent to non-water miscible solvent may be from about 3 to 1 to about 2 to 1 or about 2 to 1 to about 1 to 1.
In yet another embodiment, the solvent in the extended release formulations in the inventive methods may be a blend of a water-miscible solvent and a non-water miscible solvent in a weightweight ratio of about 1 to 2 or about 1 to 3, water-miscible solvent to non-water miscible solvent. In another embodiment, the weightweight ratio of the water-miscible solvent to non-water miscible solvent may be from about 1 to about 5, 1 to about 7.
In one embodiment, the solvent in the extended release formulations in the inventive methods may be a blend of cyclic carbonate (e.g., propylene carbonate) and glycerol ester (e.g., triacetin) in a weight: weight ratio of between about 10 to 1 to about 1 to 1, cyclic carbonate (e.g., propylene carbonate) to glycerol ester (e.g., triacetin). In one embodiment, the solvent may be a blend of cyclic carbonate (e.g., propylene carbonate) and glycerol ester (e.g., triacetin) in a weight: weight ratio of between about 5 to 1 to about 1 to 1, cyclic carbonate (e.g. propylene carbonate) to glycerol ester (e.g., triacetin). In another embodiment, the solvent may be a blend
of cyclic carbonate (e.g., propylene carbonate) and glycerol ester (e.g., triacetin) in a weight: weight ratio of between about 3 to 1 to about 1 to 1, cyclic carbonate (e.g. propylene carbonate) to glycerol ester (e.g., triacetin). In yet another embodiment, the solvent may be a blend of cyclic carbonate (e.g., propylene carbonate) and glycerol ester (e.g., triacetin) in a weight: weight ratio of between about 2 to 1 to about 1 to 1 or about 3 : 1 to about 2: 1, cyclic carbonate (e.g. propylene carbonate) to glycerol ester (e.g., triacetin). In other embodiments, the range for the weigh weight ratio of cyclic carbonate (e.g. propylene carbonate) to glycerol ester (e.g., triacetin) is 1.5: 1 to about 15: 1 or from about 2: 1 to about 6: 1.
The extended release injectable formulations may include surfactants, which may be present at concentrations of about 0.1 % to about 10% (w/w), about 1 % to about 10% (w/w) or about 5% to about 10% (w/w). More typically, surfactants may be present at concentrations of about 0.1 % to about 5% (w/w) or about 1 to about 5% (w/w). Examples of surfactants that may be used in the compositions include, but are not limited to, glyceryl monooleate, poly oxy ethylene sorbitan fatty acid esters, sorbitan esters 30 including sorbitan monooleate (Span® 20), polyvinyl alcohol, polysorbates including polysorbate 20 and polysorbate 80, d-a- tocopheryl polyethylene glycol 1000 succinate (TPGS), sodium lauryl sulfate, co-polymers of ethylene oxide and propylene oxide (e.g. poloxamers such as LUTROL® F87 and the like), polyethylene glycol castor oil derivatives including polyoxyl 35 castor oil (Cremophor® EL), polyoxyl 40 hydrogenated castor oil (Cremophor® RH 40), polyoxyl 60 hydrogenated castor oil (Cremophor® RH60); propylene glycol monolaurate (LAUROGLYCOL®); glyceride esters including glycerol caprylate/caprate (CAPMUL® MCM), polyglycolized glycerides (GELUCIRE®), PEG 300 caprylic/capric glycerides (Softigen® 767), PEG 400 caprylic/capric glycerides (Labrasol®), PEG 300 oleic glycerides (Labrafil® M-1944CS), PEG 300 linoleic glycerides (Labrafil® M-2125CS); polyethylene glycol stearates and polyethylene glycol hydroxy stearates including polyoxyl 8 stearate (PEG 400 monostearate) , polyoxyl 40 stearate (PEG 1750 monostearate, and the like). Polyethylene glycol stearates (synonyms include macrogol stearates, polyoxyl stearates, polyoxyethylene stearates, ethoxylated stearates; CAS No. 9004-99-3, 9005-08-7) are mixtures of mono- and distearate esters of mixed polyoxyethylene polymers. Polyethylene glycol hydroxystearate is a mixture of mono- and diesters of hydroxystearic acid with polyethylene glycols. One polyethylene glycol hydroxystearate that may be used in the compositions is polyethylene glycol 12-hydroxy stearate. In another
embodiment, the formulations may include the surfactant polyethylene glycol 15 12- hydroxystearate (Kolliphor® HS 15 from BASF), a mixture of mono- and diesters of 12- hydroxystearic acid with 15 moles of ethylene oxide. Again, these compounds, as well as their amounts are well known in the art. In another embodiment of the invention, the inventive formulations may include polyoxyl 35 castor oil (Kolliphor® EL) as a surfactant. In other embodiments, the inventive formulations may include polyoxyl 40 hydrogenated castor oil (Kolliphor® RH 40) or polyoxyl 60 hydrogenated castor oil as surfactants. The formulations of the invention may also include a combination of surfactants.
The extended release injectable formulations may also include other inert ingredients such as antioxidants, preservatives, or pH stabilizers. These compounds are well known in the formulation art. Antioxidants such as vitamin E, alpha tocopherol, ascorbic acid, ascorbyl palmitate, citric acid, fumaric acid, malic acid, sodium ascorbate, sodium metabi sulfate, sodium metabi sulfite, n-propyl gallate, BHA, BHT, BHA and citric acid, monothioglycerol, tert-butyl hydroquinone (TBHQ), and the like, may be added. The antioxidants are generally added to the formulation in amounts of from about 0.01 to about 5.0%, based upon total weight of the formulation, with about 0.05 to about 2.0% being especially preferred. In another embodiment, the formulation preferably contains about 0.05 to about 1.0% (w/w) of an antioxidant.
Preservatives, such as the parabens (methylparaben, ethylparaben, butylparaben and/or propylparaben), are suitably used in the extended release injectable formulations in amounts ranging from about 0.01 to about 2.0%, with about 0.05 to about 1.0% being especially preferred. Other preservatives include benzalkonium chloride, benzethonium chloride, benzoic acid, benzyl alcohol, bronopol, cetrimide, chlorhexidine, chlorobutanol, chlorocresol, cresol, imidurea, phenol, phenoxyethanol, phenylethyl alcohol, phenylmercuric acetate, phenylmercuric borate, phenylmercuric nitrate, potassium sorbate, sodium benzoate, sodium propionate, sorbic acid, thimerosal, and the like. Preferred ranges for these compounds include from about 0.01 to about 5%.
Compounds which stabilize the pH of the formulation are also contemplated. Again, such compounds are well known to a practitioner in the art as well as how to use these compounds. Buffering systems include, for example, systems selected from the group consisting of acetic acid/acetate, malic acid/malate, citric acid/citrate, tartaric acid/tartrate, lactic acid/lactate, phosphoric acid/phosphate, glycine/glycimate, tris, glutamic acid/glutamates and sodium
carbonate
The extended release injectable formulations used in the inventive methods may comprise, for example, about 1 to about 40% (w/w) of a pharmaceutically acceptable polymer, including a biodegradable polymer. In other embodiments, the formulations may comprise about 1 to about 30%) (w/w) or about 1 to about 20% (w/w) of a pharmaceutically acceptable polymer. In another embodiment, the formulations may comprise about 1 to about 15%> (w/w) or about 1 to about 10%) (w/w) of a pharmaceutically acceptable polymer. In another embodiment, the formulations may comprise about 5 to about 20% (w/w) or about 5 to about 15%> (w/w) of a pharmaceutically acceptable polymer. In another embodiment, the formulations may comprise about 10 to about 20% (w/w) or about 10 to about 15%> (w/w) of a pharmaceutically acceptable polymer. In another embodiment, the formulations may comprise about 7 to about 13%> (w/w) or about 8 to about 15%> (w/w) of a pharmaceutically acceptable polymer. In yet another embodiment, the formulations may comprise about 1 to about 7% (w/w), about 1 to about 5% (w/w) or about 3 to about 7% (w/w) of a pharmaceutically acceptable polymer.
The extended release injectable formulations used in the inventive methods may comprise, for example, about 30% to about 90% (w/w) of a solvent or mixture of solvents. In another embodiment, formulations may comprise about 40% to about 90% (w/w) of a solvent or mixture of solvents. In yet another embodiment, the formulations may comprise about 40% to about 80% (w/w), about 50% to about 80% (w/w) or about 45% to about 80% (w/w) of a solvent or a mixture of solvents. In yet another embodiment, the formulations may comprise about 60% to about 80%) (w/w) or about 65% to about 80% (w/w) of a solvent or a mixture of solvents. In still another embodiment, the formulations may comprise about 65% to about 75% (w/w) or about 70%) to about 80% (w/w) of a solvent or a mixture of solvents.
The extended release injectable formulations used in the inventive methods may comprise, for example, about 0.01% to about 10% (w/w) of a pharmaceutically acceptable additive, excipient or mixtures thereof. In other embodiments, the formulations may comprise about 0.01% to about 5% (w/w), about 0.1% to about 10% (w/w) or about 0.1% to about 5% (w/w) of a pharmaceutically acceptable additive, excipient or mixtures thereof.
The extended release injectable formulations may comprise, for example, about 0.01% to about 5%) (w/w) of an antioxidant. In other embodiments, the formulations may comprise about 0.01%) to about 3%) (w/w) or about 0.01 to about 2% (w/w) of an antioxidant.
The long acting and extended release injectable formulations used in the inventive methods may comprise, for example, from about 1 to about 50% (w/w) of an isoxazoline bioactive agent, In some embodiments in which the formulations utilize the (S)-enantiomer of the isoxazoline active agent, which is significantly more potent than the racemic mixture, the extended release formulations may include lower concentrations of the compound such as about 1 to about 10%) (w/w) or about 1 to about 5% (w/w). In another embodiment, the formulations may comprise about 5 to about 30%> (w/w) of the isoxazoline bioactive agent. In yet other embodiments, the formulations may include about 5 to about 20% (w/w) or about 5 to about 15%> (w/w) of the isoxazoline bioactive agent. In another embodiment, the formulations may comprise about 10 to about 40% (w/w) or 10 to about 30%> (w/w) of an isoxazoline bioactive agent. In another embodiment, the formulations may comprise about 10 to about 20% of an isoxazoline bioactive agent. In yet another embodiment, the formulations may comprise about 15% to about 40%) (w/w), about 15% to about 35% (w/w) or about 15% to about 30% (w/w) of an isoxazoline compound. In yet another embodiment, the formulations may comprise about 20 to about 30% (w/w), about 20 to about 25% (w/w) or about 25 to about 30% (w/w) of the isoxazoline bioactive agent.
Dosage forms may contain from about 0.5 mg to about 5 g of a single or combination of bioactive agent(s) in the long acting and extended release formulations used in the methods of the invention. More typically, the amount of active agent(s) in the long acting and extended release formulations used in the inventive methods will be from about 1 mg to about 3 g. In another embodiment, the amount of bioactive agent(s) in the compositions will be from about 20 mg to about 3 g. In another embodiment, the amount of bioactive agent(s) present in the long acting and extended release formulations will be from about 20 mg to about 2 g, about 20 mg to about 1.5 g or about 20 mg to about 1 g. In other embodiments, the amount of bioactive agent(s) in the compositions will be from about 20 mg to about 500 mg, about 30 mg to about 200 mg or about 50 mg to about 200 mg. In still another embodiment, the amount of bioactive agent(s) present in the compositions will be from about 50 mg to about 2 g, about 50 mg to about 1 g or about 50 mg to about 500 mg. In yet another embodiment of the invention, the about of bioactive agent(s) present will be from about 100 mg to about 2 g, about 100 mg to about 1 g or about 100 mg to about 500 mg.
In another embodiment, the amount of bioactive agent(s) present in an amount of from
about 1 mg to about 500 mg of an active agent, about 1 mg to about 100 mg or about 1 mg to about 25 mg. In still other embodiments, the amount of the bioactive agent(s) present in the compositions is about 10 mg about 50 mg or about 10 mg to about 100 mg. In other embodiments, the amount of bioactive agent(s) present in the compositions is about 50 mg to about 200 mg, about 100 mg to about 300 mg, about 100 mg to about 400 mg, about 200 mg to about 500 mg, about 300 mg to about 600 mg, about 400 mg to about 800 mg, or about 500 mg to about 1000 mg.
Additional Bioactive Agents
Additional veterinary/pharmaceutical bioactive active agents may be used with the compositions of the invention. In some embodiments, the additional active agents may include, but are not limited to, acaricides, anthelmintics, anti-parasitics and insecticides. Anti-parasitic agents can include both ectoparasiticidal and/or endoparasiticidal agents.
Veterinary pharmaceutical agents that may be included in the compositions of the invention are well-known in the art (see e.g. Plumb 's Veterinary Drug Handbook, 5th Edition, ed. Donald C. Plumb, Blackwell Publishing, (2005) or The Merck Veterinary Manual, 9th Edition, (January 2005)) and include but are not limited to acarbose, acepromazine maleate, acetaminophen, acetazol amide, acetazolamide sodium, acetic acid, acetohydroxamic acid, acetylcysteine, acitretin, acyclovir, albendazole, albuterol sulfate, alfentanil, allopurinol, alprazolam, altrenogest, amantadine, amikacin sulfate, aminocaproic acid, aminopentamide hydrogen sulfate, aminophylline/theophylline, amiodarone, amitriptyline, amlodipine besylate, ammonium chloride, ammonium molybdenate, amoxicillin, clavulanate potassium, amphotericin B desoxycholate, amphotericin B lipid-based, ampicillin, amprolium, antacids (oral), antivenin, apomorphione, apramycin sulfate, ascorbic acid, asparaginase, aspiring, atenolol, atipamezole, atracurium besylate, atropine sulfate, aurnofin, aurothioglucose, azaperone, azathioprine, azithromycin, baclofen, barbituates, benazepril, betamethasone, bethanechol chloride, bisacodyl, bismuth subsalicylate, bleomycin sulfate, boldenone undecylenate, bromides, bromocriptine mesylate, budenoside, buprenorphine, buspirone, busulfan, butorphanol tartrate, cabergoline, calcitonin salmon, calcitrol, calcium salts, captopril, carbenicillin indanyl sodium, carbimazole, carboplatin, carnitine, carprofen, carvedilol, cefadroxil, cefazolin sodium, cefixime, clorsulon, cefoperazone sodium, cefotaxime sodium, cefotetan di sodium, cefoxitin sodium, cefpodoxime proxetil, ceftazidime, ceftiofur sodium, ceftiofur, ceftiaxone sodium, cephalexin, cephalosporins,
cephapirin, charcoal (activated), chlorambucil, chloramphenicol, chlordiazepoxide, chlordiazepoxide +/- clidinium bromide, chlorothiazide, chlorpheniramine maleate, chlorpromazine, chlorpropamide, chlortetracycline, chorionic gonadotropin (HCG), chromium, cimetidine, ciprofloxacin, cisapride, cisplatin, citrate salts, clarithromycin, clemastine fumarate, clenbuterol, clindamycin, clofazimine, clomipramine, claonazepam, clonidine, cloprostenol sodium, clorazepate dipotassium, clorsulon, cloxacillin, codeine phosphate, colchicine, corticotropin (ACTH), cosyntropin, cyclophosphamide, cyclosporine, cyproheptadine, cytarabine, dacarbazine, dactinomycin/actinomycin D, dalteparin sodium, danazol, dantrolene sodium, dapsone, decoquinate, deferoxamine mesylate, deracoxib, deslorelin acetate, desmopressin acetate, desoxycorticosterone pivalate, detomidine, dexamethasone, dexpanthenol, dexraazoxane, dextran, diazepam, diazoxide (oral), dichlorphenamide, diclofenac sodium, dicloxacillin, diethylcarbamazine citrate, di ethyl stilbestrol (DES), difloxacin, digoxin, dihydrotachysterol (DHT), diltiazem, dimenhydrinate, dimercaprol/BAL, dimethyl sulfoxide, dinoprost tromethamine, diphenylhydramine, disopyramide phosphate, dobutamine, docusate/DSS, dolasetron mesylate, domperidone, dopamine, doramectin, doxapram, doxepin, doxorubicin, doxycycline, edetate calcium disodium. calcium EDTA, edrophonium chloride, enalapril/enalaprilat, enoxaparin sodium, enrofloxacin, ephedrine sulfate, epinephrine, epoetin/erythropoietin, eprinomectin, epsiprantel, erythromycin, esmolol, estradiol cypionate, ethacrynic acid/ethacrynate sodium, ethanol (alcohol), etidronate sodium, etodolac, etomidate, euthanasia agents w/pentobarbital, famotidine, fatty acids (essential/omega), felbamate, fentanyl, ferrous sulfate, filgrastim, finasteride, fipronil, florfenicol, fluconazole, flucytosine, fludrocortisone acetate, flumazenil, flumethasone, flunixin meglumine, fluorouracil (5-FU), fluoxetine, fluticasone propionate, fluvoxamine maleate, fomepizole (4-MP), furazolidone, furosemide, gabapentin, gemcitabine, gentamicin sulfate, glimepiride, glipizide, glucagon, glucocorticoid agents, glucosamine/chondroitin sulfate, glutamine, glyburide, glycerine (oral), glycopyrrolate, gonadorelin, grisseofulvin, guaifenesin, halothane, hemoglobin glutamer-200 (OXYGLOBIN®), heparin, hetastarch, hyaluronate sodium, hydrazaline, hydrochlorothiazide, hydrocodone bitartrate, hydrocortisone, hydromorphone, hydroxyurea, hydroxyzine, ifosfamide, imidacloprid, imidocarb dipropinate, impenem-cilastatin sodium, imipramine, inamrinone lactate, insulin, interferon alfa-2a (human recombinant), iodide (sodium/potassium), ipecac (syrup), ipodate sodium, iron dextran, isoflurane, isoproterenol, isotretinoin, isoxsuprine,
itraconazole, ivermectin, kaolin/pectin, ketamine, ketoconazole, ketoprofen, ketorolac tromethamine, lactulose, leuprolide, levamisole, levetiracetam, levothyroxine sodium, lidocaine, lincomycin, liothyronine sodium, lisinopril, lomustine (CCNU), lufenuron, lysine, magnesium, mannitol, marbofloxacin, mechlorethamine, meclizine, meclofenamic acid, medetomidine, medium chain triglycerides, medroxyprogesterone acetate, megestrol acetate, melarsomine, melatonin, meloxican, melphalan, meperidine, mercaptopurine, meropenem, metformin, methadone, methazolamide, methenamine mandelate/hippurate, methimazole, methionine, methocarbamol, methohexital sodium, methotrexate, methoxyflurane, methylene blue, methylphenidate, methylprednisolone, metoclopramide, metoprolol, metronidaxole, mexiletine, mibolerlone, midazolam milbemycin oxime, mineral oil, minocycline, misoprostol, mitotane, mitoxantrone, morphine sulfate, moxidectin, naloxone, mandrolone decanoate, naproxen, narcotic (opiate) agonist analgesics, neomycin sulfate, neostigmine, niacinamide, nitazoxanide, nitenpyram, nitrofurantoin, nitroglycerin, nitroprusside sodium, nizatidine, novobiocin sodium, nystatin, octreotide acetate, olsalazine sodium, omeprozole, ondansetron, opiate antidiarrheals, orbifloxacin, oxacillin sodium, oxazepam, oxibutynin chloride, oxymorphone, oxytretracycline, oxytocin, pamidronate disodium, pancreplipase, pancuronium bromide, paromomycin sulfate, parozetine, pencillamine, general information penicillins, penicillin G, penicillin V potassium, pentazocine, pentobarbital sodium, pentosan polysulfate sodium, pentoxifylline, pergolide mesylate, phenobarbital, phenoxybenzamine, pheylbutazone, phenylephrine, phenypropanolamine, phenytoin sodium, pheromones, parenteral phosphate, phytonadi one/vitamin K-l, pimobendan, piperazine, pirlimycin, piroxicam, poly sulfated glycosaminoglycan, ponazuril, potassium chloride, pralidoxime chloride, prazosin, prednisolone/prednisone, primidone, procainamide, procarbazine, prochlorperazine, propantheline bromide, propionibacterium acnes injection, propofol, propranolol, protamine sulfate, pseudoephedrine, psyllium hydrophilic mucilloid, pyridostigmine bromide, pyrilamine maleate, pyrimethamine, quinacrine, quinidine, ranitidine, rifampin, s-adenosyl-methionine (SAMe), saline/hyperosmotic laxative, selamectin, selegiline /1-deprenyl, sertraline, sevelamer, sevoflurane, silymarin/milk thistle, sodium bicarbonate, sodium polystyrene sulfonate, sodium stibogluconate, sodium sulfate, sodium thiosulfate, somatotropin, sotalol, spectinomycin, spironolactone, stanozolol, streptokinase, streptozocin, succimer, succinylcholine chloride, sucralfate, sufentanil citrate, sulfachlorpyridazine sodium, sulfadiazine/trimethroprim,
sulfamethoxazole/trimethoprim, sulfadimentoxine, sulfadimethoxine/ormetoprim, sulfasalazine, taurine, tepoxaline, terbinafline, terbutaline sulfate, testosterone, tetracycline, thiacetarsamide sodium, thiamine, thioguanine, thiopental sodium, thiotepa, thyrotropin, tiamulin, ticarcilin disodium, tiletamine /zolazepam, tilmocsin, tiopronin, tobramycin sulfate, tocainide, tolazoline, telfenamic acid, topiramate, tramadol, trimcinolone acetonide, trientine, trilostane, trimepraxine tartrate w/prednisolone, tripelennamine, tylosin, urdosiol, valproic acid, vanadium, vancomycin, vasopressin, vecuronium bromide, verapamil, vinblastine sulfate, vincristine sulfate, vitamin E/selenium, warfarin sodium, xylazine, yohimbine, zafirlukast, zidovudine (AZT), zinc acetate/zinc sulfate, zonisamide and mixtures thereof.
The isoxazoline compound in the parasiticidal compositions of the present methods may be combined with arylpyrazole compounds, such as phenylpyrazoles, known in the art. Examples of such arylpyrazole compounds include but are not limited to fipronil, pyriprole, ethiprole and those described in U.S. Patent Nos. 6,001,384; 6,010,710; 6,083,519; 6,096,329; 6, 174,540; 6,685,954 and 6,998, 131 (all of which are incorporated herein by reference).
The isoxazoline compound in the parasiticidal compositions of the present methods may combined also be combined with one or more macrocyclic lactones or lactams, which act as an acaricide, anthelmintic agent and/or insecticide.
The macrocyclic lactones include, but are not limited to, avermectins such as abamectin, dimadectin, doramectin, emamectin, eprinomectin, ivermectin, latidectin, lepimectin, selamectin and ML-1, 694,554, and milbemycins such as milbemectin, milbemycin D, milbemycin oxime, moxidectin and nemadectin. Also included are the 5-oxo and 5-oxime derivatives of said avermectins and milbemycins.
The macrocyclic lactone compounds are known in the art and can easily be obtained commercially or through synthesis techniques known in the art. Reference is made to the widely available technical and commercial literature. For avermectins, ivermectin and abamectin, reference may be made, for example, to the work "Ivermectin and Abamectin", 1989, by M.H. Fischer and H. Mrozik, William C. Campbell, published by Springer Verlag., or Albers- Schonberg et al. (1981), "Avermectins Structure Determination", J. Am. Chem. Soc, 103, 4216- 4221. For doramectin, "Veterinary Parasitology", vol. 49, No. 1, July 1993, 5-15 may be consulted. For milbemycins, reference may be made, inter alia, to Davies H.G. et al., 1986, "Avermectins and Milbemycins", Nat. Prod. Rep., 3, 87-121, Mrozik H. et al., 1983, Synthesis
of Milbemycins from Avermectins, Tetrahedron Lett., 24, 5333-5336, U.S. Patent No. 4,134,973 and EP 0 677 054.
Macrocyclic lactones are either natural products or are semi-synthetic derivatives thereof. The structure of the avermectins and milbemycins are closely related, e.g., by sharing a complex 16-membered macrocyclic lactone ring. The natural product avermectins are disclosed in U.S. Patent No. 4,310,519 and the 22,23-dihydro avermectin compounds are disclosed in U.S. Patent No. 4, 199,569. Mention is also made of U.S. Patent Nos. 4,468,390, 5,824,653, EP 0 007 812 Al, U.K. Patent Specification 1 390 336, EP 0 002 916, and New Zealand Patent No. 237 086, inter alia. Naturally occurring milbemycins are described in U.S. Patent No. 3,950,360 as well as in the various references cited in "The Merck Index" 12th ed., S. Budavari, Ed., Merck & Co., Inc. Whitehouse Station, New Jersey (1996). Latidectin is described in the "International Nonproprietary Names for Pharmaceutical Substances (INN)", WHO Drug Information, vol. 17, no. 4, pp. 263- 286, (2003). Semisynthetic derivatives of these classes of compounds are well known in the art and are described, for example, in U.S. Patent Nos. 5,077,308, 4,859,657, 4,963,582, 4,855,317, 4,871,719, 4,874,749, 4,427,663, 4,310,519, 4, 199,569, 5,055,596, 4,973,711, 4,978,677, 4,920,148 and EP 0 667 054.
In another embodiment of the inventive methods, the parasiticidal compositions comprise an isoxazoline compound in combination with systemically-acting compounds from a class of acaricides or insecticides known as insect growth regulators (IGRs). Compounds belonging to this group are well known to the practitioner and represent a wide range of different chemical classes. These compounds all act by interfering with the development or growth of the insect pests. Insect growth regulators are described, for example, in U.S. Patent Nos. 3,748,356, 3,818,047, 4,225,598, 4,798,837, 4,751,225, EP 0 179 022 or U.K. 2 140 010 as well as U.S. Patent Nos. 6,096,329 and 6,685,954 (all incorporated herein by reference).
In one embodiment the IGR is a compound that mimics juvenile hormone. Examples of juvenile hormone mimics include azadirachtin, diofenolan, fenoxycarb, hydroprene, kinoprene, methoprene, pyriproxyfen, tetrahydroazadirachtin and 4-chloro-2(2-chloro-2-methyl-propyl)-5- (6-iodo-3-pyridylmethoxy)pyridazine-3(2H)-one. In another embodiment the IGR is methoprene or pyriproxyfen.
In another embodiment, the IGR compound is a chitin synthesis inhibitor. Chi tin synthesis inhibitors include chlorofluazuron, cyromazine, diflubenzuron, fluazuron,
flucycloxuron, flufenoxuron, hexaflumoron, lufenuron, tebufenozide, teflubenzuron, triflumoron, novaluron, l-(2,6-difluorobenzoyl)-3-(2-fluoro-4-(trifluoromethyl)phenylurea, l-(2,6-difluoro- benzoyl)-3-(2-fluoro-4-(l,l,2,2-tetrafluoroethoxy)-phenylurea and l-(2,6-difluorobenzoyl)-3-(2- fluoro-4-trifluoromethyl)phenylurea.
In yet another embodiment, adulticide insecticides and acaricides can also be added to the extended release formulations of the present invention. These include pyrethrins (which include cinerin I, cinerin II, jasmolin I, jasmolin II, pyrethrin I, pyrethrin II and mixtures thereof) and pyrethroids, and carbamates including, but are not limited to, benomyl, carbanolate, carbaryl, carbofuran, meththiocarb, metolcarb, promacyl, propoxur, aldicarb, butocarboxim, oxamyl, thiocarboxime and thiofanox. In one embodiment, the parasiticidal compositions comprise an isoxazoline compound combined with permethrin.
In some embodiments, the parasiticidal compositions of the inventive methods may include one or more antinematodal agents including, but not limited to, active agents in the benzimidazoles, imidazothiazoles, tetrahydropyrimidines, and organophosphate class of compounds. In some embodiments, benzimidazoles including, but not limited to, thiabendazole, cambendazole, parbendazole, oxibendazole, mebendazole, flubendazole, fenbendazole, oxfendazole, albendazole, cyclobendazole, febantel, thiophanate and its ο,ο-dimethyl analogue may be included in the compositions.
In other embodiments, the parasiticidal compositions of the inventive methods may include an imidazothiazole compounds including, but not limited to, tetramisole, levamisole and butamisole. In still other embodiments, the extended release formulations of the present invention may include tetrahydropyrimidine active agents including, but not limited to, pyrantel, oxantel, and morantel.
Suitable organophosphate active agents include, but are not limited to, coumaphos, trichlorfon, haloxon, naftalofos and dichlorvos, heptenophos, mevinphos, monocrotophos, TEPP, and tetrachlorvinphos.
In other embodiments, the parasiticidal compositions of the inventive methods may include the antinematodal compounds phenothiazine and piperazine as the neutral compound or in various salt forms, diethylcarbamazine, phenols such as disophenol, arsenicals such as arsenamide, ethanolamines such as bephenium, thenium closylate, and methyridine; cyanine dyes including pyrvinium chloride, pyrvinium pamoate and dithiazanine iodide; isothiocyanates
including bitoscanate, suramin sodium, phthalofyne, and various natural products including, but not limited to, hygromycin B, a-santonin and kainic acid.
In other embodiments, the parasiticidal compositions of the inventive methods may include antitrematodal agents. Suitable antitrematodal agents include, but are not limited to, the miracils such as miracil D and mirasan; praziquantel, epsiprantel, clonazepam and its 3-methyl derivative, oltipraz, lucanthone, hycanthone, oxamniquine, amoscanate, niridazole, nitroxynil, various bisphenol compounds known in the art including hexachlorophene, bithionol, bithionol sulfoxide and menichlopholan; various salicylanilide compounds including tribromsalan, oxyclozanide, clioxanide, rafoxanide, brotianide, bromoxanide and closantel; triclabendazole, diamfenetide, clorsulon, hetolin and emetine.
Anticestodal compounds may also be advantageously used in parasiticidal compositions of the inventive methods including, but not limited to, praziquantel, epsiprantel, and arecoline in various salt forms, bunamidine, niclosamide, nitroscanate, paromomycin and paromomycin II.
In yet other embodiments, parasiticidal compositions of the inventive methods may include other active agents that are effective against arthropod parasites. Suitable active agents include, but are not limited to, bromocyclen, chlordane, DDT, endosulfan, lindane, methoxychlor, toxaphene, bromophos, bromophos-ethyl, carbophenothion, chlorfenvinphos, chlorpyrifos, crotoxyphos, cythioate, diazinon, dichlorenthion, diemthoate, dioxathion, ethion, famphur, fenitrothion, fenthion, fospirate, iodofenphos, malathion, naled, phosalone, phosmet, phoxim, propetamphos, ronnel, stirofos, allethrin, cyhalothrin, cypermethrin, deltamethrin, fenvalerate, flucythrinate, permethrin, phenothrin, pyrethrins, resmethrin, benzyl benzoate, carbon disulfide, crotamiton, diflubenzuron, diphenylamine, disulfiram, isobornyl thiocyanato acetate, methoprene, monosulfiram, piperonylbutoxide, rotenone, triphenyltin acetate, triphenyltin hydroxide, deet, dimethyl phthalate, and the compounds l,5a,6,9,9a,9b-hexahydro- 4a(4H)-dibenzofurancarboxaldehyde (MGK-11), 2-(2-ethylhexyl)-3a,4,7,7a-tetrahydro-4,7- methano-lH-isoindole-l,3(2H)dione (MGK-264), dipropyl-2,5-pyridinedicarboxylate (MGK- 326) and 2-(octylthio)ethanol (MGK-874).
An antiparasitic agent that can be combined with an isoxazoline compounds in the parasiticidal formulations of the inventive methods can be a biologically active peptide or protein including, but not limited to, depsipeptides, which act at the neuromuscular junction by stimulating presynaptic receptors belonging to the secretin receptor family resulting in the
paralysis and death of parasites. In one embodiment of the depsipeptide, the depsipeptide is emodepside (see Willson et al., Parasitology, Jan. 2003, 126(Pt l):79-86). In another embodiment, the depsipeptide is PF1022A or a derivative thereof. In another embodiment, the isoxazoline compounds may be combined with a cyclic depsipeptide described in WO 2016/187534 Al or WO 2017/116702 Al, both incorporated herein by reference in their entirety.
In another embodiment, the parasiticidal compositions of the inventive methods may comprise an active agent from the neonicotinoid class of pesticides. The neonicotinoids bind and inhibit insect specific nicotinic acetylcholine receptors. In one embodiment, the neonicotinoid insecticidal agent that can be combined with an isoxazoline compound to form an extended release injectable formulation of the invention is imidacloprid. Imidacloprid is a well-known neonicotinoid active agent and is the key active ingredient in the topical parasiticide products Advantage®, Advantage® II, K9 Advantix®, and K9 Advantix® II sold by Bayer Animal Health and the oral soft-chewable formulation Advantus™ from Piedmont Animal Health. Agents of this class are described, for example, in U.S. Patent No. 4,742,060 or in EP 0 892 060.
In another embodiment, parasiticidal compositions of the inventive methods may comprise nitenpyram, another active agent of the neonicotinoid class of pesticides in combination with the isoxazoline compound. Nitenpyram is the active ingredient in the oral product CAPSTAR™ Tablets sold by Novartis Animal Health.
In certain embodiments, the parasiticidal composition comprises an insecticidal agent that is a semicarbazone, such as metaflumizone.
In another embodiment, the parasitical compositions of the inventive methods may advantageously include a combination of a second isoxazoline compounds known in the art. These active agents are described in WO 2007/079162, WO 2007/075459 and US 2009/0133319, WO 2007/070606 and US 2009/0143410, WO 2009/003075, WO 2009/002809, WO 2009/024541, WO 2005/085216 and US 2007/0066617 and WO 2008/122375, all of which are incorporated herein by reference in their entirety.
In another embodiment of the invention, nodulisporic acid and its derivatives (a class of known acaricidal, anthelmintic, anti-parasitic and insecticidal agents) may be added to the parasiticidal compositions of the present methods. These compounds are used to treat or prevent infections in humans and animals and are described, for example, in U.S. Patent No. 5,399,582, 5,962,499, 6,221,894 and 6,399,786, all of which are hereby incorporated by reference in their
entirety. The formulations may include one or more of the known nodulisporic acid derivatives in the art, including all stereoisomers, such as those described in the patents cited above.
In another embodiment, anthelmintic compounds of the amino acetonitrile class (AAD) of compounds such as monepantel (ZOLVIX), and the like, may be added to the parasiticidal compositions of the inventive methods. These compounds are described, for example, in WO 2004/024704 and U.S. Patent No. 7,084,280 (incorporated by reference); Sager et al., Veterinary Parasitology, 2009, 159, 49-54; Kaminsky et al., Nature vol. 452, 13 March 2008, 176-181.
The parasiticidal compositions of the present methods may also include aryloazol-2-yl cyanoethylamino compounds such as those described in US Patent No. 8,088,801 to Soil et al., which is incorporated herein in its entirety, and thioamide derivatives of these compounds, as described in U.S. Patent No. 7,964,621, which is incorporated herein by reference.
The parasiticidal compositions of the inventive methods may also be combined with paraherquamide compounds and derivatives of these compounds, including derquantel (see Ostlind et al., Research in Veterinary Science, 1990, 48, 260-61; and Ostlind et al., Medical and Veterinary Entomology, 1997, 11, 407-408). The paraherquamide family of compounds is a known class of compounds that include a spirodioxepino indole core with activity against certain parasites (see Tet. Lett. 1981, 22, 135; J. Antibiotics 1990, 43, 1380, and J. Antibiotics 1991, 44, 492). In addition, the structurally related marcfortine family of compounds, such as marcfortines A-C, are also known and may be combined with the formulations of the invention (see J. Chem. Soc. - Chem. Comm. 1980, 601 and Tet. Lett. 1981, 22, 1977). Further references to the paraherquamide derivatives can be found, for example, in WO 91/09961, WO 92/22555, WO 97/03988, WO 01/076370, WO 09/004432, U.S. Patent 5,703,078 and U.S. Patent 5,750,695, all of which are hereby incorporated by reference in their entirety.
In another embodiment of the invention, the parasiticidal compositions may include a spinosyn active agent produced by the soil actinomycete Saccharopolyspora spinosa (see, for example Salgado V.L. and Sparks T.C., "The Spinosyns: Chemistry, Biochemistry, Mode of Action, and Resistance,'" in Comprehensive Molecular Insect Science, vol. 6, pp. 137-173, 2005) or a semi-synthetic spinosoid active agent. The spinosyns are typically referred to as factors or components A, B, C, D, E, F, G, H, J, K, L, M, N, O, P, Q, R, S, T, U, V, W, or Y, and any of these components, or a combination thereof, may be used in the compositions of the invention. The spinosyn compound may be a 5,6,5-tricylic ring system, fused to a 12-membered macro
cyclic lactone, a neutral sugar (rhamnose), and an amino sugar (forosamine). These and other natural spinosyn compounds, including 21-butenyl spinosyn produced by Saccharopolyspora pagona, which may be used in the parasiticidal compositions of the inventive methods, may be produced via fermentation by conventional techniques known in the art. Other spinosyn compounds that may be used in the compositions of the invention are disclosed in U.S. Patent Nos. 5,496,931; 5,670,364; 5,591,606; 5,571,901; 5,202,242; 5,767,253; 5,840,861; 5,670,486; 5,631, 155 and 6,001,981, all incorporated by reference herein in their entirety. The spinosyn compounds may include, but are not limited to, spinosyn A, spinosyn D, spinosad, spinetoram, or combinations thereof. Spinosad is a combination of spinosyn A and spinosyn D, and spinetoram is a combination of 3'-ethoxy-5,6-dihydro spinosyn J and 3'-ethoxy spinosyn L.
In general, the additional bioactive agent is included in the parasiticidal compositions of the inventive methods is in an amount of between about 0.1 μg and about 1000 mg. More typically, the additional bioactive agent may be included in an amount of about 10 μg to about 500 mg, about 1 mg to about 300 mg, about 10 mg to about 200 mg or about 10 mg to about 100 mg.
In other embodiments of the parasiticidal compositions of the inventive methods, the additional bioactive agent may be included in the composition to deliver a dose of about 5 μg/kg to about 50 mg/kg per weight of the animal. In other embodiments, the additional bioactive agent may be present in an amount sufficient to deliver a dose of about 0.01 mg/kg to about 30 mg/kg, about 0.1 mg/kg to about 20 mg/kg, or about 0.1 mg/kg to about 10 mg/kg of weight of animal. In other embodiments, the additional bioactive agent may be present in a dose of about 5 μg/kg to about 200 μg/kg or about 0.1 mg/kg to about 1 mg/kg of weight of animal. In still another embodiment of the invention, the additional bioactive agent is included in a dose between about 0.5 mg/kg to about 50 mg/kg. Parasitic Infestations and Infections
The parasitic infestations and infections of the inventive methods are caused by ectoparasites and endoparasites. The methods of the invention treat or prevent parasitic infestations and infections (when including an endoparasiticidal agent) while reducing unwanted side effects. Ectoparasites include, but are not limited to, fleas, ticks, mites, mosquitoes, parasitic flies (e.g., horn fly, stable fly, blowfly) and lice. Endoparasites include, but are not limited to,
cestodes, nematodes, hookworms and roundworms of the digestive tract of non-human animal and filarial worms such as Dirofilaria immitis.
More specifically, ectoparasites include one or more insect or arachnid including those of the genera Ctenocephalides, Rhipicephalus, Dermacentor, Ixodes, Amblyomma, Haemaphysalis, Hyalomma, Sarcoptes, Psoroptes, Otodectes, Chorioptes, Hypoderma, Damalinia, Linognathus, Haematopinus, Solenoptes, Trichodectes, and Felicola. Specific examples include, but are not limited to, cat and dog fleas {Ctenocephalides sp. such as Ctenocephalides felis, Ctenocephalides canis, and the like), ticks (Rhipicephalus sp., Ixodes sp., Dermacentor sp., Amblyomma sp. and the like), and mites (Demodex sp., Sarcoptes sp., Otodectes sp. and the like), lice (Trichodectes sp., Cheyletiella sp., Linognathus sp., and the like), mosquitoes (Aedes sp., Culex sp., Anopheles sp., and the like) and parasitic flies (Haematobia sp. including Haematobia irritans, Musca sp., Stomoxys sp. including Stomoxys calcitrans, Dermatobia sp., Cochliomyia sp., and the like).
Additional examples of ectoparasites include but are not limited to the tick genus Rhipicephalus, especially those of the species microplus (cattle tick), decoloratus and annulatus; myiasis such as Dermatobia hominis (known as Berne in Brazil) and Cochliomyia hominivorax (greenbottle); sheep myiasis such as Lucilia sericata, Lucilia cuprina (known as blowfly strike in Australia, New Zealand and South Africa). Flies proper, namely those whose adult constitutes the parasite, such as Haematobia irritans (horn fly) and Stomoxys calcitrans (stable fly); lice such as Linognathus vituli, etc.; and mites such as Sarcoptes scabiei and Psoroptes ovis. The above list is not exhaustive and other ectoparasites are well known in the art to be harmful to animals and humans. These include, for example migrating dipterous larvae.
Endoparasites include helminths selected from the group consisting of Anaplocephala, Ancylostoma, Necator, Ascaris, Capillaria, Cooperia, Dipylidium, Dirofilaria, Echinococcus, Enterobius, Fasciola, Haemonchus, Oesophagostomum, Ostertagia, Toxocara, Strongyloides, Toxascaris, Trichinella, Trichuris, Angiostrongylus and Trichostrongylus, among others. In one embodiment, the methods of the invention will prevent the occurrence of Heartworm disease by killing immature stages of Dirofilaria immitis before they can mature to adult worms.
Examples of non-human animals which parasiticidal infections occurs include livestock and companion animals, such as cats, dogs, horses, birds including chickens, sheep, goats, pigs, deer, turkeys and cattle.
The formulations of the invention are administered in parasiticidally effective amounts which are which are suitable to control the parasite in question to the desired extent, as described below. In each aspect of the invention, the compounds and compositions of the invention can be applied against a single pest or combinations thereof.
Generally, a dose of from about 0.001 to about 100 mg per kg of body weight given as a single dose or in divided doses for a period of from 1 to 5 days will be satisfactory but, of course, there can be instances where higher or lower dosage ranges are indicated, and such are within the scope of this invention. It is well within the routine skill of the practitioner to determine a particular dosing regimen for a specific host and parasite.
In companion animals, the dose of the isoxazoline bioactive agent administered is between about 0.1 to about 50 mg per kg of body weight. More typically the dose of the isoxazoline bioactive agent administered is about 0.5 to about 40 mg/kg or about 0.5 to about 30 mg/kg body weight. In another embodiment, the dose of the isoxazoline bioactive agent administered is about 10 to about 40 mg/kg, about 15 to about 35 mg/kg or about 20 to about 30 mg/kg of body weight. In another embodiment, the dose of the isoxazoline active bioagent will be about 20 to about 25 mg/kg of body weight.
The dose administered may be lower depending on the animal and the isoxazoline administered. In some embodiments, the dose is from about 0.1 to about 30 mg/kg of body weight. In another embodiment, the dose may be from about 0.1 to about 20 mg/kg or about 0.1 to about 10 mg/kg of body weight. In other embodiments, the dose may be from about 1 to about 20 mg/kg of body weight, about 1 to about 10 mg/kg or about 1 to about 5 mg/kg. In another embodiment, the dose may be from about 0.1 to about 2 mg/kg or from about 0.5 to about 2 mg/kg. In yet another embodiment, the dose may be from about 5 to about 20 mg/kg, about 5 to about 15 mg/kg or about 10 to about 20 mg/kg of body weight. In another embodiment, the dose may be from about 10 to about 30 mg/kg of body weight.
For the treatment of livestock animals, such as cattle or sheep, doses of the isoxazoline bioactive agent administered may be about 0.1 to about 40 mg/kg of body weight. More typically the doses administered will be about 1 to about 30 mg/kg, about 1 to about 20 mg/kg or about 1 to about 10 mg/kg of body weight. In yet another embodiment, the dose may be from about 10 to about 25 mg/kg, about 15 to about 30 mg/kg of body weight or about 20 to about 30 mg/kg of body weight. In another embodiment, the dose for livestock animals may be from about 0.1
mg/kg to about 3 mg/kg, from about 0.1 mg/kg to about 2 mg/kg or from about 0.1 mg/kg to about 1 mg/kg of body weight.
In one embodiment, the parasiticidal compounds and compositions of the inventive methods may exhibit an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least about 90% or higher for about 1 month, or longer. In another embodiment, the parasiticidal compounds and compositions of the inventive methods will exhibit an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least about 90% for about 2 months or about 3 months. These compositions may provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least 95.0% or higher for about, 3 months or longer. Other parasiticidal compositions in the inventive methods may provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least 90% or higher for about 6 months or longer. In yet another embodiment, parasiticidal formulations of the inventive methods may provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least 95% or higher for about 6 months or longer. In another embodiment, the parasiticidal provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least 90% or higher for about 9 months or longer. In yet another embodiment, the parasiticidal compositions provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites of at least 90% or higher for about 12 months or longer. In another embodiment, the parasiticidal compositions provide an efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites in cats and dogs of at least about 90% for two months, or longer. In another embodiment, the efficacy against fleas and/or ticks and/or mosquitoes, or other ectoparasites in cats and dogs of about 95% for about 3 months, or longer. In still another embodiment, the parasiticidal compositions provide an efficacy of about 95% for about 5 months or longer.
In another embodiment, the parasiticidal compositions provide an efficacy against
Rhipicephalus microplus in cattle of at least about 90% for one month, or longer. In another embodiment, the parasiticidal compositions provide an efficacy against Rhipicephalus microplus in cattle of at least about 90% for two month, or longer. In another embodiment, the parasiticidal compositions provide an efficacy against Rhipicephalus microplus in cattle of at least about 90% for three month, or longer. In another embodiment, the efficacy against Rhipicephalus microplus
and/or other ectoparasites in cattle of about 95% for about one month, two months, three months or longer.
The present invention will be further illustrated in the following Examples which are given for illustration purposes only and are not intended to limit the invention in any way.
Examples
Example 1 : An experiment was performed to measure the effect the enantiomers and the racemic mixture of an isoxazoline compound (Compound A) has on the weight of laboratory mice for 14 days following the administration of the isoxazoline compound.
The following formulations were prepared and administered to female laboratory rats:
Formulation A (100 mg of the (R) enantiomer Compound A dissolved in the carrier),
Formulation B (200 mg of the racemic mixture of Compound A dissolved the carrier);
Formulation C (500 mg of the (S) enantiomer of the Compound A dissolved in the carrier); and
Control (carrier).
The weight of the rats was then monitored for 14 days.
Fig. 1 depicts the change in weight of the rats over the 14 day period. Rats that were administered the formulation containing the (S) enantiomer (Formulation C) experienced approximately the same weight gain as those rats that were administered the control. Rats that were administered the formulations containing either (R) enantiomer or the racemic mixture experienced weight loss, thereby indicating that the unwanted effect (weight loss) resided in the (R) enantiomer.
Example 2: Efficacy and Acceptability of Injectable Formulation Against Rhipicephalus (Boophilus) microplus ticks.
The efficacy and acceptability of long-acting injectable compositions of the invention comprising the compounds of formula (Ie) and (S)-le shown below, against Rhipicephalus microplus ticks on cattle was determined against an untreated control group. The efficacy of compositions comprising the compounds (Ie) and (S)-e were also compared with injectable compositions comprising afoxolaner (formula la, 4-[5-[3-chloro-5-(trifluoromethyl)phenyl]-4,5- dihydro-5-(trifluoromethyl)-3-isoxazolyl]-N-[2-oxo-2-[(2,2,2-trifluoroethyl)amino]ethyl]-l- naphthalanecarboxamide), and comparative compounds of formulae (If) and (S)-If shown
(If)
(S)-li
Each compound was formulated in an injectable composition at a concentration of 10% (w/v) in a carrier comprising 8% (w/v) ethanol and Q.S. with polyethylene glycol 400. The racemic compounds afoxolaner and (If) were dosed at 1.0 mg/kg body weight and the (S)- enantiomers {(S)-le and (S)-If) were dosed at 0.5 mg/kg body weight. Six healthy head of cattle were used in each study group. Cattle in Group 1 were untreated (control). Cattle in Groups 2, 3, 4, 5 and 6 were treated on Day 0 subcutaneously with injectable compositions comprising the compounds of formula Ie, (S)-If, (S)-Ie, afoxolaner (la) and (If), respectively. Several weeks before treatment, cattle were infested three times a week with approximately 2500 Rhipicephalus microplus larvae to establish ongoing infestations. Each animal was challenged by infestation with approximately 5000 R. microplus larvae on Days 7 and 21 and every 14 days thereafter. Ticks dropping from each animal in the previous 24 hours were collected daily and counted from Day 1 until the end of the study. The cattle in study Groups 3, 5 and 6 were not infested further when the efficacy of the treatment dropped significantly.
No abnormal health effect observations related to any of the treatments were seen following treatment with the formulations or at the daily general health observations that were conducted. Since the total dose of (S)-If, (S)-le was half of the dose of the dose of the racemic compounds, it is expected that the effect on the injection site would be significantly reduced.
Tables 1A, IB and 1C below show the total tick count % efficacy of injectable compositions comprising compounds of the invention ((Ie) and (S)-Ie) against R. microplus for selected days through Day 110 post treatment compared with the untreated control group and the comparison isoxazoline compounds. As evidenced from the data in Tables 1A, IB and 1C below, the compositions comprising the compounds (Ie) and (S)-le of the invention dosed at 1.0 mg/kg and 0.5 mg/kg, respectively, provide superior efficacy against Rhipicephalus microplus ticks for
an extended period of time compared with compositions comprising afoxolaner, (If) or (SJ-Ιΐ. Further, the efficacy of compounds (Ie) and (S)-le was also found to be faster-acting than compositions comprising the other isoxazoline compounds.
Table 1A: Tick Count Efficacy Against Rhipicephalus microplus
Average % Efficacy (Tick Count)
Treatment Group Day Day Day Day Day Day Day
5 10 20 30 40 49 55
Group 2 (Ie) 69 100 100 100 100 100 100
Group 3 ((S)-If) 39 27 33 86 100 36 0
Group 4 ((S)-Ie) 66 93 100 100 100 100 100
Group 5 afoxolaner 38 48 77 99 100 94 73
Group 6 (If) 70 62 57 84 100 70 14
Table IB : Tick Count Efficacy Against Rhipicephalus microplus
Average % Efficacy (Tick Count)
Treatment Group Day Day Day Day Day Day Day
60 65 70 75 80 85 88
Group 2 (Ie) 100 100 100 100 100 100 100
Group 3 ((S)-If) . . . . . . .
Group 4 ((S)-Ie) 100 100 100 100 100 90 88
Group 5 afoxolaner 71 48 3 0 - - -
Group 6 (If)
Table 1C: Tick Count Efficacy against Rhipicephalus microplus
Average % Efficacy
(Tick Count)
Treatment Group
Day Day Day Day
95 100 105 110
Group 2 (Ie) 100 100 93 82
Group 3 ((S)-If) - -
Group 4 ((S)-Ie) 92 88 76 65
Group 5 afoxolaner - -
Group 6 (If) - - - -
Claims
What is claimed is:
1. A method for reducing an unwanted effect when treating livestock for parasitic infestations, which comprises administering an effective amount of a parasiticidal composition comprising an (S) enantiomer of an isoxazoline compound of the formula
(S)-l wherein:
B1, B2, B3, are each independently C-R or N; each R is independently H, halogen, cyano, -N02, alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl;
Y is an optionally substituted phenylene, naphthylene, indanylene, a 5- or 6-membered heteroarylene or an 8-10-membered fused heterobicyclylene, wherein the optional substituents are selected from the group consisting of halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN or— N02 and H2-C(=S)-;
Q is X- R2R3, the group (-CH2-)(-CH2-)N-R3, OH, H2, alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
X is (CH2)n, CH(CH3), CH(CN), C(=0) or C(=S);
R2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R3 is H, OR7, R8R9 or Q1; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R4; or
R2 and R3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— Ν02 and alkoxy; each R4 is independently halogen; alkyl, cycloalkyl, alkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, haloalkylamino, dialkylamino, dihaloalkylamino, cycloalkylamino, alkylcarbonyl, alkoxycarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, haloalkylcarbonyl, haloalkoxycarbonyl, haloalkylaminocarbonyl, dihaloalkylaminocarbonyl, hydroxy,— NH2,— CN or— N02; or Q2; each R5 is independently halogen, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino, alkoxycarbonyl, — CN or— N02; each R6 is independently halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN,— N02, phenyl or pyridinyl;
R7 is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
R8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R9 is H; Q3; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R4; or
R8 and R9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N02 and alkoxy;
Q1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R5;
Q2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6;
Q3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6; and n is 0, 1 or 2; or a pharmaceutically acceptable salt thereof and an inert carrier or formulation adjuvant; wherein the composition contains less than 10% by weight of an (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof.
2. The method according to claim 1, wherein Y is
Y-l Y-2 Y-3
Y-4 Y-5 Υ-6
wherein Ζ is Ν or CH.
3. The method according to claim 1 or 2, wherein Q is -C(0) HCH2C(0) HCH2CF3, -C(0)CH2S(0)2CH3, -C(0) HCH2CH2SCH3 or (-CH2-)(-CH2-)N(CO)CH2S(0)2CH3.
4. The method according to claim 1, wherein the isoxazoline compound of formula (S)-I has the structure (,S)-IA below:
(5 IA
or a pharmaceutically acceptable salt thereof;
wherein X1, X2 and X3 are each independently H, bromo, chloro, fluoro, iodo or CF3.
5. The method of claim 4, wherein X1 is chloro or CF3, X2 is fluoro, and X3 is chloro.
6. The method of claim 5, wherein X1 is CF3 and X2 is fluoro.
7. The method of claim 1, wherein the isoxazoline compound of formula (S)-I has the structure (S)-TB below
(s m;
or a pharmaceutically acceptable salt thereof;
wherein X1, X2 and X3 are each independently H, bromo, chloro, fluoro, iodo or CF3.
8. The method of claim 7, wherein X1 is chloro or CF3, X2 is H or fluoro, and X3 is chloro.
9. The method of claim 8, wherein X1 is chloro and X2 is H.
10. The method of claim 8, wherein X1 is chloro and X2 is fluoro.
1 1. The method according to claim 1, wherein the parasiticidal composition is substantially free of an (R) enantiomer of the isoxazoline compound.
12. The method according to claim 1, wherein the parasiticidal composition is a topical composition.
13. The method according to claim 12, wherein the topical parasiticidal composition is a spot-on or pour-on.
14. The method according to claim 1, wherein the parasiticidal composition is an oral composition.
15. The method according to claim 14, wherein the parasiticidal composition is an oral drench.
16. The method according to claim 14, wherein the parasiticidal composition is dissolved in the drinking water.
17. The method according to claim 1, wherein the parasiticidal composition is an injectable composition.
18. The method according to claim 17, wherein the injectable composition is a long-acting injectable composition or an extended-release injectable composition.
19. The method according to claim 1, wherein the livestock are sheep or cattle.
20. A method for treating fleas and ticks in a non-human animal in need thereof while reducing at least one unwanted effect, which comprises administering an effective amount of a parasiticidal composition comprising an (S) enantiomer of an isoxazoline compound of the formula
(S)-l wherein:
B1, B2, B3, are each independently C-R or N; each R is independently H, halogen, cyano, -N02, alkyl, haloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino or alkoxycarbonyl;
Y is an optionally substituted phenylene, naphthylene, indanylene, a 5- or 6-membered heteroarylene or an 8-10-membered fused heterobicyclylene, wherein the optional substituents are selected from the group consisting of halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN or— Ν02 and H2-C(=S)-;
Q is X- R2R3, the group (-CH2-)(-CH2-)N-R3, OH, H2, alkoxy, haloalkoxy, alkylamino, haloalkylamino, dialkylamino, halodialkylamino, thiol, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, or an optionally substituted 5- or 6-membered carbocyclyl, heterocyclyl or heteroaryl ring;
X is (CH2)n, CH(CH3), CH(CN), C(=0) or C(=S);
R2 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxy carbonyl;
R3 is H, OR7, R8R9 or Q1; or alkyl, haloalkyl, alkenyl, haloalkenyl, alkynyl, haloalkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl, alkoxycarbonyl, aminocarbonyl, alkylaminocarbonyl or dialkylaminocarbonyl, each optionally substituted with one or more substituents independently selected from R4; or
R2 and R3 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— N02 and alkoxy; each R4 is independently halogen; alkyl, cycloalkyl, alkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, haloalkylamino, dialkylamino, dihaloalkylamino, cycloalkylamino, alkylcarbonyl, alkoxycarbonyl, alkylaminocarbonyl, dialkylaminocarbonyl, haloalkylcarbonyl, haloalkoxycarbonyl, haloalkylaminocarbonyl, dihaloalkylaminocarbonyl, hydroxy,— NH2,— CN or— N02; or Q2; each R5 is independently halogen, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino, alkoxycarbonyl, — CN or— N02; each R6 is independently halogen, alkyl, haloalkyl, cycloalkyl, halocycloalkyl, alkoxy, haloalkoxy, alkylthio, haloalkylthio, alkylsulfinyl, haloalkylsulfinyl, alkylsulfonyl, haloalkylsulfonyl, alkylamino, dialkylamino,— CN,— N02, phenyl or pyridinyl;
R is H; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one of more halogen;
R8 is H, alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl, cycloalkylalkyl, alkylcarbonyl or alkoxycarbonyl;
R9 is H; Q3; or alkyl, alkenyl, alkynyl, cycloalkyl, alkylcycloalkyl or cycloalkylalkyl, each optionally substituted with one or more substituents independently selected from R4; or
R8 and R9 are taken together with the nitrogen to which they are attached to form a ring containing 2 to 6 atoms of carbon and optionally one additional atom selected from the group consisting of N, S and O, said ring optionally substituted with 1 to 4 substituents independently selected from the group consisting of alkyl, halogen,— CN,— Ν02 and alkoxy;
Q1 is a phenyl ring, a 5- or 6-membered heterocyclic ring, or an 8-, 9- or 10-membered fused bicyclic ring system optionally containing one to three heteroatoms selected from up to 1 O, up to 1 S and up to 3 N, each ring or ring system optionally substituted with one or more substituents independently selected from R5;
Q2 is independently a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6;
Q3 is a phenyl ring or a 5- or 6-membered heterocyclic ring, each ring optionally substituted with one or more substituents independently selected from R6; and n is 0, 1 or 2; or a pharmaceutically acceptable salt thereof and an inert carrier or formulation diluent; wherein the composition contains less than about 10% by weight of an (R) enantiomer of an isoxazoline compound or a pharmaceutically acceptable salt thereof; and wherein said parasiticidal is effective against 90 % of the fleas and the ticks for at least for 3 to 6 months.
1. The method according to claim 20, wherein Y is
wherein Z is N or CH.
22. The method according to claim 20 or 21, wherein Q is -C(0) HCH2C(0) HCH2CF3, -C(0)CH2S(0)2CH3, -C(0) HCH2CH2SCH3 or (-CH2-)(-CH2-)N(CO)CH2S(0)2CH3.
23. The method according to claim 20, wherein the isoxazoline compound of formula (S)-I has the structure (S)-IA below:
(S)-IA
or a pharmaceutically acceptable salt thereof;
wherein X1, X2 and X3 are each independently H, bromo, chloro, fluoro, iodo
The method of claim 23, wherein X1 is chloro or CF3, X2 is H or fluoro, and X3 is chloro.
25. The method of claim 24, wherein X1 is CF3 and X2 is fluoro.
26. The method of claim 20, wherein the isoxazoline compound of formula (S)-I has the structure (S)-IB below
(s m;
or a pharmaceutically acceptable salt thereof;
wherein X1, X2 and X3 are each independently H, bromo, chloro, fluoro, iodo or CF3.
27. The method of claim 26, wherein X1 is chloro or CF3, X2 is H or fluoro, and X3 is chloro.
28. The method of claim 27, wherein X1 is chloro and X2 is H.
29. The method of claim 27, wherein X1 is chloro and X2 is fluoro.
30. The method according to claim 20, wherein the parasiticidal composition is substantially free of an (R) enantiomer of an isoxazoline compound.
31. The method according to claim 20, wherein the parasiticidal composition comprises no (R) enantiomer of an isoxazoline compound.
32. The method according to claim 20, wherein the parasiticidal composition is a topical composition.
33. The method according to claim 32, wherein the topical parasiticidal composition is a spot-on or pour-on.
34. The method according to claim 20, wherein the parasiticidal composition is an oral composition.
35. The method according to claim 34, wherein the oral parasiticidal composition is a paste, a chewable tablet, or a soft chew.
36. The method according to claim 20, wherein the parasiticidal composition is an injectable composition.
37. The method according to claim 36, wherein the injectable composition is a long acting injectable composition or an extended release injectable formulation.
38. The method according to claim 20, wherein the non-human animal is a dog or cat.
39. The method according to claim 20, wherein the unwanted effect is weight loss.
40. The method according to claim 20, wherein the unwanted effect is a neurological disorder.
41. The method according to claim 36, wherein the unwanted effect is an adverse effect at the injection site.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201662379345P | 2016-08-25 | 2016-08-25 | |
| US62/379,345 | 2016-08-25 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2018039508A1 true WO2018039508A1 (en) | 2018-03-01 |
Family
ID=59772815
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2017/048527 Ceased WO2018039508A1 (en) | 2016-08-25 | 2017-08-25 | Method for reducing unwanted effects in parasiticidal treatments |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2018039508A1 (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2020225143A1 (en) * | 2019-05-03 | 2020-11-12 | Intervet International B.V. | Injectable pharmaceutical compositions and uses thereof |
| EP2658542B1 (en) | 2010-12-27 | 2022-01-26 | Intervet International B.V. | Topical localized isoxazoline formulation |
| EP2658541B1 (en) | 2010-12-27 | 2022-01-26 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| US11576382B2 (en) | 2018-02-12 | 2023-02-14 | Fmc Corporation | Naphthalene isoxazoline compounds for controlling invertebrate pests |
| RU2829447C2 (en) * | 2019-05-03 | 2024-10-30 | Интервет Интернэшнл Б.В. | Pharmaceutical compositions for injections and use thereof |
| US12304903B2 (en) | 2020-07-24 | 2025-05-20 | Elanco Us Inc. | Process for making an isoxazoline compound and intermediate thereof |
| US12497370B2 (en) | 2019-12-18 | 2025-12-16 | Elanco Tiergesundheit Ag | Isoxazoline derivatives |
| WO2025257633A1 (en) * | 2024-06-12 | 2025-12-18 | Boehringer Ingelheim Vetmedica Gmbh | Long-acting castor oil-containing injectable formulations and methods of use thereof |
Citations (103)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3740421A (en) | 1966-09-19 | 1973-06-19 | Basf Wyandotte Corp | Polyoxyethylene-polyoxypropylene aqueous gels |
| US3748356A (en) | 1970-05-15 | 1973-07-24 | Philips Corp | Substituted benzoyl ureas |
| US3818047A (en) | 1972-08-07 | 1974-06-18 | C Henrick | Substituted pyrones |
| GB1390336A (en) | 1972-06-08 | 1975-04-09 | Sankyo Co | Antibiotic b-41 |
| US3950360A (en) | 1972-06-08 | 1976-04-13 | Sankyo Company Limited | Antibiotic substances |
| US4134973A (en) | 1977-04-11 | 1979-01-16 | Merck & Co., Inc. | Carbohydrate derivatives of milbemycin and processes therefor |
| EP0002916A2 (en) | 1977-12-19 | 1979-07-11 | Merck & Co. Inc. | A milbemycin compound or a mixture thereof for use in the treatment of helminthic infections and anthelmintic compositions containing such compounds |
| US4166452A (en) | 1976-05-03 | 1979-09-04 | Generales Constantine D J Jr | Apparatus for testing human responses to stimuli |
| EP0007812A1 (en) | 1978-08-02 | 1980-02-06 | Merck & Co. Inc. | Sugar derivatives of C-076 compounds, their preparation and their use as antiparasitic agents |
| US4199569A (en) | 1977-10-03 | 1980-04-22 | Merck & Co., Inc. | Selective hydrogenation products of C-076 compounds and derivatives thereof |
| US4225598A (en) | 1976-08-19 | 1980-09-30 | Ciba-Geigy Corporation | 2-Cyclopropylamino-4,6-diamino-s-triazine |
| US4256108A (en) | 1977-04-07 | 1981-03-17 | Alza Corporation | Microporous-semipermeable laminated osmotic system |
| US4265874A (en) | 1980-04-25 | 1981-05-05 | Alza Corporation | Method of delivering drug with aid of effervescent activity generated in environment of use |
| US4310519A (en) | 1976-04-19 | 1982-01-12 | Merck & Co., Inc. | Novel substances and process for their production |
| US4427663A (en) | 1982-03-16 | 1984-01-24 | Merck & Co., Inc. | 4"-Keto-and 4"-amino-4"-deoxy avermectin compounds and substituted amino derivatives thereof |
| US4468390A (en) | 1981-02-23 | 1984-08-28 | Sankyo Company, Limited | Anthelmintic composition and the use thereof |
| GB2140010A (en) | 1983-04-25 | 1984-11-21 | Sumitomo Chemical Co | Nitrogen-containing heterocyclic compounds, and their production and use |
| US4564631A (en) | 1982-09-29 | 1986-01-14 | Bayer Aktiengesellschaft | Baits for combating vermin |
| EP0179022A2 (en) | 1984-10-18 | 1986-04-23 | Ciba-Geigy Ag | Benzoylphenyl ureas |
| US4742060A (en) | 1985-02-04 | 1988-05-03 | Nihon Tokushu Noyaku Seizo K. K. | Heterocyclic compounds |
| US4855317A (en) | 1987-03-06 | 1989-08-08 | Ciba-Geigy Corporation | Insecticides and parasiticides |
| US4859657A (en) | 1986-07-02 | 1989-08-22 | Ciba-Geigy Corporation | Pesticides |
| US4871719A (en) | 1987-03-24 | 1989-10-03 | Ciba-Geigy Corporation | Composition for controlling parasites in productive livestock |
| US4874749A (en) | 1987-07-31 | 1989-10-17 | Merck & Co., Inc. | 4"-Deoxy-4-N-methylamino avermectin Bla/Blb |
| US4963582A (en) | 1986-03-25 | 1990-10-16 | Sankyo Company Limited | Macrolide compounds, their preparation and their use |
| US4978677A (en) | 1986-03-06 | 1990-12-18 | Ciba-Geigy Corporation | C(29)-carbonyloxymilbemycin derivatives for controlling parasitic pets of animals and plants |
| WO1991009961A2 (en) | 1989-12-21 | 1991-07-11 | Beecham Group Plc | Novel products |
| US5055596A (en) | 1987-11-03 | 1991-10-08 | Beecham Group P.L.C. | Derivatives of avermectin and milbemycin |
| US5077308A (en) | 1989-02-16 | 1991-12-31 | Merck & Co., Inc. | Avermectin ketal derivatives useful as antiparasitic |
| WO1992022555A1 (en) | 1991-06-17 | 1992-12-23 | Beecham Group Plc | Paraherquamide derivatives, precursor thereof, processes for their preparation, microorganism used and their use as antiparasitic agents |
| US5202242A (en) | 1991-11-08 | 1993-04-13 | Dowelanco | A83543 compounds and processes for production thereof |
| NZ237086A (en) | 1991-02-12 | 1993-07-27 | Ancare Distributors | Veterinary anthelminitic drench compositions containing praziquantel and at least one other anthelmintic compound; method of treatment |
| US5399582A (en) | 1993-11-01 | 1995-03-21 | Merck & Co., Inc. | Antiparasitic agents |
| EP0667054A1 (en) | 1992-10-30 | 1995-08-16 | Electric Power Research Institute, Inc | Harmonic controller for an active power line conditioner |
| EP0677054A1 (en) | 1993-01-18 | 1995-10-18 | Pfizer Inc. | New antiparasitic agents related to the milbemycins and avermectins |
| US5496931A (en) | 1988-12-19 | 1996-03-05 | Dowelanco | Insecticide and miticide A83543 compounds and their method of production by fermentation |
| US5591606A (en) | 1992-11-06 | 1997-01-07 | Dowelanco | Process for the production of A83543 compounds with Saccharopolyspora spinosa |
| WO1997003988A1 (en) | 1995-07-21 | 1997-02-06 | Pharmacia & Upjohn Company | Antiparasitic marcfortines and paraherquamides |
| US5670364A (en) | 1993-03-12 | 1997-09-23 | Dowelanco | A83543 compounds and process for production thereof |
| US5824653A (en) | 1994-11-28 | 1998-10-20 | Virbac S.A. | Anthelmintic compositions for equidae |
| EP0892060A2 (en) | 1997-07-18 | 1999-01-20 | Smithkline Beecham Corporation | The Chlamydia trachomatis Glutamyl-tRNAGln amidotransferase subunit ratA |
| US5962499A (en) | 1995-03-20 | 1999-10-05 | Merck & Co., Inc. | Nodulisporic acid derivatives |
| US6001384A (en) | 1996-08-20 | 1999-12-14 | Merial | Method for fighting against myiasis affecting cattle and sheep populations and compositions for implementing same |
| US6001981A (en) | 1996-06-13 | 1999-12-14 | Dow Agrosciences Llc | Synthetic modification of Spinosyn compounds |
| US6010710A (en) | 1996-03-29 | 2000-01-04 | Merial | Direct pour-on skin solution for antiparasitic use in cattle and sheep |
| US6083519A (en) | 1996-03-29 | 2000-07-04 | Merial | N-phenylpyrazole-based anti-flea and anti-tick collar for cats and dogs |
| US6096329A (en) | 1996-03-29 | 2000-08-01 | Merial | Insecticidal combination to control mammal fleas, in particular fleas on cats and dogs |
| US6174540B1 (en) | 1998-09-14 | 2001-01-16 | Merck & Co., Inc. | Long acting injectable formulations containing hydrogenated caster oil |
| US6221894B1 (en) | 1995-03-20 | 2001-04-24 | Merck & Co., Inc. | Nodulisporic acid derivatives |
| WO2001076370A2 (en) | 2000-04-07 | 2001-10-18 | Pharmacia & Upjohn Company | Anthelmintic combinations |
| US6395765B1 (en) | 1995-09-29 | 2002-05-28 | Merial | Antiparasitic composition for the treatment and protection of pets |
| US6399786B1 (en) | 2000-07-14 | 2002-06-04 | Merck & Co., Inc. | Nonacyclic nodulisporic acid derivatives |
| US6426333B1 (en) | 1996-09-19 | 2002-07-30 | Merial | Spot-on formulations for combating parasites |
| WO2004024704A1 (en) | 2002-09-11 | 2004-03-25 | Novartis Ag | Organic compounds |
| US6733767B2 (en) | 1998-03-19 | 2004-05-11 | Merck & Co., Inc. | Liquid polymeric compositions for controlled release of bioactive substances |
| US20040151759A1 (en) | 2002-08-16 | 2004-08-05 | Douglas Cleverly | Non-animal product containing veterinary formulations |
| US6787342B2 (en) | 2000-02-16 | 2004-09-07 | Merial Limited | Paste formulations |
| WO2005085216A1 (en) | 2004-03-05 | 2005-09-15 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and noxious organism control agent |
| US6998131B2 (en) | 1996-09-19 | 2006-02-14 | Merial Limited | Spot-on formulations for combating parasites |
| US7001889B2 (en) | 2002-06-21 | 2006-02-21 | Merial Limited | Anthelmintic oral homogeneous veterinary pastes |
| US20060222684A1 (en) | 2003-07-30 | 2006-10-05 | Ute Isele | Palatable ductile chewable veterinary composition |
| WO2007070606A2 (en) | 2005-12-14 | 2007-06-21 | E. I. Du Pont De Nemours And Company | Isoxazolines for controlling invertebrate pests |
| WO2007075459A2 (en) | 2005-12-16 | 2007-07-05 | E. I. Du Pont De Nemours And Company | 5-aryl isoxazolines for controlling invertebrate pests |
| WO2007079162A1 (en) | 2005-12-30 | 2007-07-12 | E. I. Du Pont De Nemours And Company | Isoxazolines for controlling invertebrate pests |
| WO2007123855A2 (en) | 2006-04-20 | 2007-11-01 | E. I. Du Pont De Nemours And Company | Pyrazolines for controlling invertebrate pests |
| WO2008122375A2 (en) | 2007-04-10 | 2008-10-16 | Bayer Cropscience Ag | Insecticidal aryl isoxazoline derivatives |
| WO2008154528A2 (en) | 2007-06-13 | 2008-12-18 | E. I. Du Pont De Nemours And Company | Isoxazoline insecticides |
| WO2009003075A1 (en) | 2007-06-27 | 2008-12-31 | E.I. Du Pont De Nemours And Company | Animal pest control method |
| WO2009002809A2 (en) | 2007-06-26 | 2008-12-31 | E. I. Du Pont De Nemours And Company | Naphthalene isoxazoline invertebrate pest control agents |
| WO2009004432A1 (en) | 2007-06-29 | 2009-01-08 | Pfizer Inc. | Anthelmintic combination |
| WO2009024541A2 (en) | 2007-08-17 | 2009-02-26 | Intervet International B.V. | Isoxazoline compositions and their use as antiparasitics |
| WO2009045999A1 (en) * | 2007-10-03 | 2009-04-09 | E. I. Du Pont De Nemours And Company | Naphthalene isoxazoline compounds for control of invertebrate pests |
| WO2010003923A1 (en) | 2008-07-09 | 2010-01-14 | Basf Se | Pestcidal active mixtures comprising isoxazoline compounds i |
| US20100087492A1 (en) | 2008-10-03 | 2010-04-08 | Triad Specialty Products Llc | Systemic treatment of blood-sucking and blood-consuming parasites by oral administration of a parasiticidal agent |
| US20100137372A1 (en) | 2007-03-29 | 2010-06-03 | Hideki Ihara | Isoxazoline compounds and their use in pest control |
| US20110086886A2 (en) | 2006-08-15 | 2011-04-14 | Bayer Cropscience Ag | Insecticidal isoxazolines |
| US7951828B1 (en) | 2005-09-02 | 2011-05-31 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and pesticide |
| US7955632B2 (en) | 2005-12-07 | 2011-06-07 | Bayer B.V. | Process for manufacturing chewable dosage forms for drug delivery and products thereof |
| WO2011067272A1 (en) * | 2009-12-01 | 2011-06-09 | Syngenta Participations Ag | Insecticidal compounds based on isoxazoline derivatives |
| US7964621B2 (en) | 2008-10-21 | 2011-06-21 | Merial Limited | Thioamide compounds, method of making and method of using thereof |
| WO2011149749A1 (en) | 2010-05-27 | 2011-12-01 | E.I. Du Pont De Nemours And Company | Crystalline form of 4- [5 - [3 -chloro-5 - (trifluoromethyl) phenyl] -4, 5 - dihydro - 5 - (trifluoromethyl) -3 - isoxazolyl] -n- [2-0x0-2- [ ( 2, 2, 2 - trifluoroethyl) amino] ethyl] -1- naphthalenecarboxamide |
| US8088801B2 (en) | 2007-05-15 | 2012-01-03 | Merial Limited | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| WO2012089623A1 (en) | 2010-12-27 | 2012-07-05 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| WO2012120399A1 (en) | 2011-03-10 | 2012-09-13 | Pfizer Inc. | Spirocyclic isoxazoline derivatives as antiparasitic agents |
| WO2012120135A1 (en) | 2011-03-10 | 2012-09-13 | Novartis Ag | Isoxazole derivatives |
| US8362086B2 (en) | 2005-08-19 | 2013-01-29 | Merial Limited | Long acting injectable formulations |
| WO2013026695A1 (en) * | 2011-08-25 | 2013-02-28 | Syngenta Participations Ag | Isoxazoline derivatives as insecticidal compounds |
| WO2013026930A1 (en) * | 2011-08-25 | 2013-02-28 | Syngenta Participations Ag | Process for the preparation of thietane derivatives |
| WO2013039948A1 (en) | 2011-09-12 | 2013-03-21 | Merial Limited | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| WO2013119442A1 (en) | 2012-02-06 | 2013-08-15 | Merial Limited | Parasiticidal oral veterinary compositions comprising systemically-acting active agents, methods and uses thereof |
| WO2014039475A1 (en) | 2012-09-07 | 2014-03-13 | Zoetis Llc | Spirocyclic isoxazoline parasiticidal combinations |
| WO2014039484A1 (en) | 2012-09-07 | 2014-03-13 | Zoetis Llc | Spirocyclic isoxazolines as antiparasitic agents |
| WO2014090918A1 (en) * | 2012-12-13 | 2014-06-19 | Novartis Ag | Process for the enantiomeric enrichment of diaryloxazoline derivatives |
| WO2014189837A1 (en) | 2013-05-20 | 2014-11-27 | Zoetis Llc | Long-acting spiro-isoxazoline antiparasitic compositions |
| US20150057239A1 (en) | 2012-04-04 | 2015-02-26 | Intervet Inc. | Solid oral pharmaceutical compositions for isoxazoline compounds |
| WO2015091898A1 (en) * | 2013-12-20 | 2015-06-25 | Intervet International B.V. | Isoxazoline compositions and use thereof in the prevention or treatment of parasite infestations in animals |
| WO2015091900A1 (en) * | 2013-12-20 | 2015-06-25 | Intervet International B.V. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| WO2016073347A1 (en) | 2014-11-03 | 2016-05-12 | Zoetis Services Llc | Palatable chewable veterinary composition |
| WO2016102437A1 (en) * | 2014-12-22 | 2016-06-30 | Intervet International B.V. | Use of isoxazoline compounds for treating demodicosis |
| WO2016138339A1 (en) | 2015-02-26 | 2016-09-01 | Merial, Inc. | Long-acting injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| WO2016164487A1 (en) | 2015-04-08 | 2016-10-13 | Merial, Inc. | Extended release injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| WO2016187534A1 (en) | 2015-05-20 | 2016-11-24 | Merial, Inc. | Anthelmintic depsipeptide compounds |
| WO2017116702A1 (en) | 2015-12-28 | 2017-07-06 | Merial, Inc. | Anthelmintic depsipeptide compounds |
-
2017
- 2017-08-25 WO PCT/US2017/048527 patent/WO2018039508A1/en not_active Ceased
Patent Citations (130)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3740421A (en) | 1966-09-19 | 1973-06-19 | Basf Wyandotte Corp | Polyoxyethylene-polyoxypropylene aqueous gels |
| US3748356A (en) | 1970-05-15 | 1973-07-24 | Philips Corp | Substituted benzoyl ureas |
| GB1390336A (en) | 1972-06-08 | 1975-04-09 | Sankyo Co | Antibiotic b-41 |
| US3950360A (en) | 1972-06-08 | 1976-04-13 | Sankyo Company Limited | Antibiotic substances |
| US3818047A (en) | 1972-08-07 | 1974-06-18 | C Henrick | Substituted pyrones |
| US4310519A (en) | 1976-04-19 | 1982-01-12 | Merck & Co., Inc. | Novel substances and process for their production |
| US4166452A (en) | 1976-05-03 | 1979-09-04 | Generales Constantine D J Jr | Apparatus for testing human responses to stimuli |
| US4225598A (en) | 1976-08-19 | 1980-09-30 | Ciba-Geigy Corporation | 2-Cyclopropylamino-4,6-diamino-s-triazine |
| US4256108A (en) | 1977-04-07 | 1981-03-17 | Alza Corporation | Microporous-semipermeable laminated osmotic system |
| US4134973A (en) | 1977-04-11 | 1979-01-16 | Merck & Co., Inc. | Carbohydrate derivatives of milbemycin and processes therefor |
| US4199569A (en) | 1977-10-03 | 1980-04-22 | Merck & Co., Inc. | Selective hydrogenation products of C-076 compounds and derivatives thereof |
| EP0002916A2 (en) | 1977-12-19 | 1979-07-11 | Merck & Co. Inc. | A milbemycin compound or a mixture thereof for use in the treatment of helminthic infections and anthelmintic compositions containing such compounds |
| EP0007812A1 (en) | 1978-08-02 | 1980-02-06 | Merck & Co. Inc. | Sugar derivatives of C-076 compounds, their preparation and their use as antiparasitic agents |
| US4265874A (en) | 1980-04-25 | 1981-05-05 | Alza Corporation | Method of delivering drug with aid of effervescent activity generated in environment of use |
| US4468390A (en) | 1981-02-23 | 1984-08-28 | Sankyo Company, Limited | Anthelmintic composition and the use thereof |
| US4427663A (en) | 1982-03-16 | 1984-01-24 | Merck & Co., Inc. | 4"-Keto-and 4"-amino-4"-deoxy avermectin compounds and substituted amino derivatives thereof |
| US4564631A (en) | 1982-09-29 | 1986-01-14 | Bayer Aktiengesellschaft | Baits for combating vermin |
| US4751225A (en) | 1983-04-25 | 1988-06-14 | Sumitomo Chemical Company, Limited | Certain 2-pyridyloxy-lower alkylene-oxy-phenoxy compounds, thio-and methylene analogues having insecticidal properties |
| GB2140010A (en) | 1983-04-25 | 1984-11-21 | Sumitomo Chemical Co | Nitrogen-containing heterocyclic compounds, and their production and use |
| EP0179022A2 (en) | 1984-10-18 | 1986-04-23 | Ciba-Geigy Ag | Benzoylphenyl ureas |
| US4798837A (en) | 1984-10-18 | 1989-01-17 | Ciba-Geigy Corporation | Benzoylphenylureas |
| US4742060A (en) | 1985-02-04 | 1988-05-03 | Nihon Tokushu Noyaku Seizo K. K. | Heterocyclic compounds |
| US4978677A (en) | 1986-03-06 | 1990-12-18 | Ciba-Geigy Corporation | C(29)-carbonyloxymilbemycin derivatives for controlling parasitic pets of animals and plants |
| US4963582A (en) | 1986-03-25 | 1990-10-16 | Sankyo Company Limited | Macrolide compounds, their preparation and their use |
| US4859657A (en) | 1986-07-02 | 1989-08-22 | Ciba-Geigy Corporation | Pesticides |
| US4855317A (en) | 1987-03-06 | 1989-08-08 | Ciba-Geigy Corporation | Insecticides and parasiticides |
| US4920148A (en) | 1987-03-06 | 1990-04-24 | Ciba-Geigy Corporation | Insecticides and parasiticides |
| US4973711A (en) | 1987-03-24 | 1990-11-27 | Ciba-Geigy Corporation | Composition for controlling parasites in productive livestock |
| US4871719A (en) | 1987-03-24 | 1989-10-03 | Ciba-Geigy Corporation | Composition for controlling parasites in productive livestock |
| US4874749A (en) | 1987-07-31 | 1989-10-17 | Merck & Co., Inc. | 4"-Deoxy-4-N-methylamino avermectin Bla/Blb |
| US5055596A (en) | 1987-11-03 | 1991-10-08 | Beecham Group P.L.C. | Derivatives of avermectin and milbemycin |
| US5496931A (en) | 1988-12-19 | 1996-03-05 | Dowelanco | Insecticide and miticide A83543 compounds and their method of production by fermentation |
| US5571901A (en) | 1988-12-19 | 1996-11-05 | Dowelanco | Insecticide and miticide compositions containing A83543 compounds |
| US5077308A (en) | 1989-02-16 | 1991-12-31 | Merck & Co., Inc. | Avermectin ketal derivatives useful as antiparasitic |
| WO1991009961A2 (en) | 1989-12-21 | 1991-07-11 | Beecham Group Plc | Novel products |
| NZ237086A (en) | 1991-02-12 | 1993-07-27 | Ancare Distributors | Veterinary anthelminitic drench compositions containing praziquantel and at least one other anthelmintic compound; method of treatment |
| WO1992022555A1 (en) | 1991-06-17 | 1992-12-23 | Beecham Group Plc | Paraherquamide derivatives, precursor thereof, processes for their preparation, microorganism used and their use as antiparasitic agents |
| US5202242A (en) | 1991-11-08 | 1993-04-13 | Dowelanco | A83543 compounds and processes for production thereof |
| EP0667054A1 (en) | 1992-10-30 | 1995-08-16 | Electric Power Research Institute, Inc | Harmonic controller for an active power line conditioner |
| US5631155A (en) | 1992-11-06 | 1997-05-20 | Dowelanco | Saccharopolyspora spinosa strain |
| US5591606A (en) | 1992-11-06 | 1997-01-07 | Dowelanco | Process for the production of A83543 compounds with Saccharopolyspora spinosa |
| US5767253A (en) | 1992-11-06 | 1998-06-16 | Dow Agrosciences Llc | A83543 Compounds; factors Q, R, S, and T |
| EP0677054A1 (en) | 1993-01-18 | 1995-10-18 | Pfizer Inc. | New antiparasitic agents related to the milbemycins and avermectins |
| US5670486A (en) | 1993-03-12 | 1997-09-23 | Dowelanco | A83543 compounds and process for production thereof |
| US5840861A (en) | 1993-03-12 | 1998-11-24 | Dowelanco | A83543 compounds and process for production thereof |
| US5670364A (en) | 1993-03-12 | 1997-09-23 | Dowelanco | A83543 compounds and process for production thereof |
| US5399582A (en) | 1993-11-01 | 1995-03-21 | Merck & Co., Inc. | Antiparasitic agents |
| US5824653A (en) | 1994-11-28 | 1998-10-20 | Virbac S.A. | Anthelmintic compositions for equidae |
| US6221894B1 (en) | 1995-03-20 | 2001-04-24 | Merck & Co., Inc. | Nodulisporic acid derivatives |
| US5962499A (en) | 1995-03-20 | 1999-10-05 | Merck & Co., Inc. | Nodulisporic acid derivatives |
| WO1997003988A1 (en) | 1995-07-21 | 1997-02-06 | Pharmacia & Upjohn Company | Antiparasitic marcfortines and paraherquamides |
| US5750695A (en) | 1995-07-21 | 1998-05-12 | Pharmacia & Upjohn Company | Antiparasitic paraherquamides |
| US5703078A (en) | 1995-07-21 | 1997-12-30 | Pharmacia & Upjohn Company | Antiparasitic marcfortines and paraherquamides |
| US6395765B1 (en) | 1995-09-29 | 2002-05-28 | Merial | Antiparasitic composition for the treatment and protection of pets |
| US6685954B2 (en) | 1996-03-29 | 2004-02-03 | Merial | Insecticidal combination to control mammal fleas, in particular fleas on cats and dogs |
| US6010710A (en) | 1996-03-29 | 2000-01-04 | Merial | Direct pour-on skin solution for antiparasitic use in cattle and sheep |
| US6083519A (en) | 1996-03-29 | 2000-07-04 | Merial | N-phenylpyrazole-based anti-flea and anti-tick collar for cats and dogs |
| US6096329A (en) | 1996-03-29 | 2000-08-01 | Merial | Insecticidal combination to control mammal fleas, in particular fleas on cats and dogs |
| US6001981A (en) | 1996-06-13 | 1999-12-14 | Dow Agrosciences Llc | Synthetic modification of Spinosyn compounds |
| US6001384A (en) | 1996-08-20 | 1999-12-14 | Merial | Method for fighting against myiasis affecting cattle and sheep populations and compositions for implementing same |
| US6426333B1 (en) | 1996-09-19 | 2002-07-30 | Merial | Spot-on formulations for combating parasites |
| US6998131B2 (en) | 1996-09-19 | 2006-02-14 | Merial Limited | Spot-on formulations for combating parasites |
| EP0892060A2 (en) | 1997-07-18 | 1999-01-20 | Smithkline Beecham Corporation | The Chlamydia trachomatis Glutamyl-tRNAGln amidotransferase subunit ratA |
| US6733767B2 (en) | 1998-03-19 | 2004-05-11 | Merck & Co., Inc. | Liquid polymeric compositions for controlled release of bioactive substances |
| US6174540B1 (en) | 1998-09-14 | 2001-01-16 | Merck & Co., Inc. | Long acting injectable formulations containing hydrogenated caster oil |
| US6787342B2 (en) | 2000-02-16 | 2004-09-07 | Merial Limited | Paste formulations |
| WO2001076370A2 (en) | 2000-04-07 | 2001-10-18 | Pharmacia & Upjohn Company | Anthelmintic combinations |
| US6399786B1 (en) | 2000-07-14 | 2002-06-04 | Merck & Co., Inc. | Nonacyclic nodulisporic acid derivatives |
| US7001889B2 (en) | 2002-06-21 | 2006-02-21 | Merial Limited | Anthelmintic oral homogeneous veterinary pastes |
| US20040151759A1 (en) | 2002-08-16 | 2004-08-05 | Douglas Cleverly | Non-animal product containing veterinary formulations |
| WO2004024704A1 (en) | 2002-09-11 | 2004-03-25 | Novartis Ag | Organic compounds |
| US7084280B2 (en) | 2002-09-11 | 2006-08-01 | Pierre Ducray | Benzotriazol-1-yl-aminoacetonitrile compounds and their use in the control of parasite disease |
| US20060222684A1 (en) | 2003-07-30 | 2006-10-05 | Ute Isele | Palatable ductile chewable veterinary composition |
| US20070066617A1 (en) | 2004-03-05 | 2007-03-22 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and pesticide |
| US7662972B2 (en) | 2004-03-05 | 2010-02-16 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and pesticide |
| WO2005085216A1 (en) | 2004-03-05 | 2005-09-15 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and noxious organism control agent |
| US8362086B2 (en) | 2005-08-19 | 2013-01-29 | Merial Limited | Long acting injectable formulations |
| US7951828B1 (en) | 2005-09-02 | 2011-05-31 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and pesticide |
| US7955632B2 (en) | 2005-12-07 | 2011-06-07 | Bayer B.V. | Process for manufacturing chewable dosage forms for drug delivery and products thereof |
| WO2007070606A2 (en) | 2005-12-14 | 2007-06-21 | E. I. Du Pont De Nemours And Company | Isoxazolines for controlling invertebrate pests |
| US20090143410A1 (en) | 2005-12-14 | 2009-06-04 | Kanu Maganbhai Patel | Isoxazolines for Controlling Invertebrate Pests |
| WO2007075459A2 (en) | 2005-12-16 | 2007-07-05 | E. I. Du Pont De Nemours And Company | 5-aryl isoxazolines for controlling invertebrate pests |
| US20090133319A1 (en) | 2005-12-16 | 2009-05-28 | E. I. Du Pont De Nemours And Company | 5-Aryl Isoxazolines for Controlling Invertebrate Pests |
| WO2007079162A1 (en) | 2005-12-30 | 2007-07-12 | E. I. Du Pont De Nemours And Company | Isoxazolines for controlling invertebrate pests |
| US7964204B2 (en) | 2005-12-30 | 2011-06-21 | E.I. Du Pont De Nemours And Company | Isoxazolines for controlling invertebrate pests |
| WO2007123855A2 (en) | 2006-04-20 | 2007-11-01 | E. I. Du Pont De Nemours And Company | Pyrazolines for controlling invertebrate pests |
| US20110086886A2 (en) | 2006-08-15 | 2011-04-14 | Bayer Cropscience Ag | Insecticidal isoxazolines |
| US20100137372A1 (en) | 2007-03-29 | 2010-06-03 | Hideki Ihara | Isoxazoline compounds and their use in pest control |
| WO2008122375A2 (en) | 2007-04-10 | 2008-10-16 | Bayer Cropscience Ag | Insecticidal aryl isoxazoline derivatives |
| US20100179194A1 (en) | 2007-04-10 | 2010-07-15 | Bayer Corpscience Ag | Insecticidal aryl isoxazoline derivatives |
| US8088801B2 (en) | 2007-05-15 | 2012-01-03 | Merial Limited | Aryloazol-2-yl cyanoethylamino compounds, method of making and method of using thereof |
| WO2008154528A2 (en) | 2007-06-13 | 2008-12-18 | E. I. Du Pont De Nemours And Company | Isoxazoline insecticides |
| US20100179195A1 (en) | 2007-06-13 | 2010-07-15 | E.I. Dupont De Nemours And Company | Isoxazoline insecticides |
| WO2009002809A2 (en) | 2007-06-26 | 2008-12-31 | E. I. Du Pont De Nemours And Company | Naphthalene isoxazoline invertebrate pest control agents |
| WO2009003075A1 (en) | 2007-06-27 | 2008-12-31 | E.I. Du Pont De Nemours And Company | Animal pest control method |
| WO2009004432A1 (en) | 2007-06-29 | 2009-01-08 | Pfizer Inc. | Anthelmintic combination |
| US20110059988A1 (en) | 2007-08-17 | 2011-03-10 | Intervet International B.V. | Isoxazoline compositions and their use as antiparasitics |
| WO2009024541A2 (en) | 2007-08-17 | 2009-02-26 | Intervet International B.V. | Isoxazoline compositions and their use as antiparasitics |
| WO2009045999A1 (en) * | 2007-10-03 | 2009-04-09 | E. I. Du Pont De Nemours And Company | Naphthalene isoxazoline compounds for control of invertebrate pests |
| US20100254960A1 (en) | 2007-10-03 | 2010-10-07 | E. I. Dupont Nemours And Company | Naphthalene isoxazoline compounds for control of invertebrate pests |
| WO2010003923A1 (en) | 2008-07-09 | 2010-01-14 | Basf Se | Pestcidal active mixtures comprising isoxazoline compounds i |
| US20100087492A1 (en) | 2008-10-03 | 2010-04-08 | Triad Specialty Products Llc | Systemic treatment of blood-sucking and blood-consuming parasites by oral administration of a parasiticidal agent |
| US7964621B2 (en) | 2008-10-21 | 2011-06-21 | Merial Limited | Thioamide compounds, method of making and method of using thereof |
| WO2011067272A1 (en) * | 2009-12-01 | 2011-06-09 | Syngenta Participations Ag | Insecticidal compounds based on isoxazoline derivatives |
| WO2011149749A1 (en) | 2010-05-27 | 2011-12-01 | E.I. Du Pont De Nemours And Company | Crystalline form of 4- [5 - [3 -chloro-5 - (trifluoromethyl) phenyl] -4, 5 - dihydro - 5 - (trifluoromethyl) -3 - isoxazolyl] -n- [2-0x0-2- [ ( 2, 2, 2 - trifluoroethyl) amino] ethyl] -1- naphthalenecarboxamide |
| WO2012089623A1 (en) | 2010-12-27 | 2012-07-05 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| WO2012120399A1 (en) | 2011-03-10 | 2012-09-13 | Pfizer Inc. | Spirocyclic isoxazoline derivatives as antiparasitic agents |
| WO2012120135A1 (en) | 2011-03-10 | 2012-09-13 | Novartis Ag | Isoxazole derivatives |
| US8466115B2 (en) | 2011-03-10 | 2013-06-18 | Zoetis Llc | Spirocyclic isoxazoline derivatives as antiparasitic agents |
| WO2013026695A1 (en) * | 2011-08-25 | 2013-02-28 | Syngenta Participations Ag | Isoxazoline derivatives as insecticidal compounds |
| WO2013026930A1 (en) * | 2011-08-25 | 2013-02-28 | Syngenta Participations Ag | Process for the preparation of thietane derivatives |
| US9180121B2 (en) | 2011-09-12 | 2015-11-10 | Merial, Inc. | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| WO2013039948A1 (en) | 2011-09-12 | 2013-03-21 | Merial Limited | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof |
| US9233100B2 (en) | 2012-02-06 | 2016-01-12 | Merial, Inc. | Parasiticidal oral veterinary compositions comprising systemically-acting active agents, methods and uses thereof |
| US9259417B2 (en) | 2012-02-06 | 2016-02-16 | Merial, Inc. | Parasiticidal oral veterinary compositions comprising systemically-acting active agents, methods and uses thereof |
| WO2013119442A1 (en) | 2012-02-06 | 2013-08-15 | Merial Limited | Parasiticidal oral veterinary compositions comprising systemically-acting active agents, methods and uses thereof |
| US20150057239A1 (en) | 2012-04-04 | 2015-02-26 | Intervet Inc. | Solid oral pharmaceutical compositions for isoxazoline compounds |
| US20150057321A1 (en) | 2012-04-04 | 2015-02-26 | Intervet Inc. | Soft chewable pharmaceutical products |
| WO2014039475A1 (en) | 2012-09-07 | 2014-03-13 | Zoetis Llc | Spirocyclic isoxazoline parasiticidal combinations |
| WO2014039484A1 (en) | 2012-09-07 | 2014-03-13 | Zoetis Llc | Spirocyclic isoxazolines as antiparasitic agents |
| WO2014090918A1 (en) * | 2012-12-13 | 2014-06-19 | Novartis Ag | Process for the enantiomeric enrichment of diaryloxazoline derivatives |
| WO2014189837A1 (en) | 2013-05-20 | 2014-11-27 | Zoetis Llc | Long-acting spiro-isoxazoline antiparasitic compositions |
| WO2015091900A1 (en) * | 2013-12-20 | 2015-06-25 | Intervet International B.V. | Use of isoxazoline derivatives for the treatment or prevention of arthropod infestations in poultry |
| WO2015091898A1 (en) * | 2013-12-20 | 2015-06-25 | Intervet International B.V. | Isoxazoline compositions and use thereof in the prevention or treatment of parasite infestations in animals |
| WO2016073347A1 (en) | 2014-11-03 | 2016-05-12 | Zoetis Services Llc | Palatable chewable veterinary composition |
| WO2016102437A1 (en) * | 2014-12-22 | 2016-06-30 | Intervet International B.V. | Use of isoxazoline compounds for treating demodicosis |
| WO2016138339A1 (en) | 2015-02-26 | 2016-09-01 | Merial, Inc. | Long-acting injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| WO2016164487A1 (en) | 2015-04-08 | 2016-10-13 | Merial, Inc. | Extended release injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| WO2016187534A1 (en) | 2015-05-20 | 2016-11-24 | Merial, Inc. | Anthelmintic depsipeptide compounds |
| WO2017116702A1 (en) | 2015-12-28 | 2017-07-06 | Merial, Inc. | Anthelmintic depsipeptide compounds |
Non-Patent Citations (23)
| Title |
|---|
| "International Nonproprietary Names for Pharmaceutical Substances (INN", WHO DRUG INFORMATION, vol. 17, no. 4, 2003, pages 263 - 286 |
| ALBERS-SCHONBERG ET AL.: "Avermectins Structure Determination", J. AM. CHEM. SOC., vol. 103, 1981, pages 4216 - 4221 |
| DAVIES H.G. ET AL.: "Avermectins and Milbemycins", NAT. PROD. REP., vol. 3, 1986, pages 87 - 121 |
| DONALD C. PLUMB,: "Plumb's Veterinary Drug Handbook, 5th edition", 2005, BLACKWELL PUBLISHING |
| GOMEZ ET AL., J. NEUROSCI., vol. 22, no. 21, 2002, pages 9612 - 9617 |
| GREENE ET AL.: "Protective Groups in Organic Synthesis, Third Edition,", 1999, JOHN WILEY AND SONS |
| J. ANTIBIOTICS, vol. 43, 1990, pages 1380 |
| J. ANTIBIOTICS, vol. 44, 1991, pages 492 |
| J. CHEM. SOC. - CHEM. COMM., vol. 601, 1980 |
| KAMINSKY ET AL., NATURE, vol. 452, 13 March 2008 (2008-03-13), pages 176 - 181 |
| M.H. FISCHER; H. MROZIK; WILLIAM C. CAMPBELL: "Ivermectin and Abamectin", 1989, SPRINGER VERLAG. |
| MARILYN N. MARTINEZ; DANIELLE LINDQUIST; SANJA MODRIC: "Terminology Challenges: Defining Modified Release Dosage Forms in Veterinary Medicine", JOURNAL OF PHARMACEUTICAL SCIENCES, vol. 99, no. 8, August 2010 (2010-08-01) |
| MERCK: "The Merck Veterinary Manual, 9th Edition,", January 2005 |
| MROZIK H. ET AL.: "Synthesis of Milbemycins from Avermectins", TETRAHEDRON LETT., vol. 24, 1983, pages 5333 - 5336 |
| OSTLIND ET AL., MEDICAL AND VETERINARY ENTOMOLOGY,, vol. 11, 1997, pages 407 - 408 |
| OSTLIND ET AL., RESEARCH IN VETERINARY SCIENCE, vol. 48, 1990, pages 260 - 61 |
| S. BUDAVARI,: "The Merck Index" 12th ed.,", 1996, MERCK & CO., INC. |
| SAGER ET AL., VETERINARY PARASITOLOGY, vol. 159, 2009, pages 49 - 54 |
| SALGADO V.L.; SPARKS T.C.: "The Spinosyns: Chemistry, Biochemistry, Mode of Action, and Resistance", COMPREHENSIVE MOLECULAR INSECT SCIENCE, vol. 6, 2005, pages 137 - 173 |
| TET. LETT., vol. 22, 1981, pages 135 |
| TET. LETT., vol. 22, 1981, pages 1977 |
| VETERINARY PARASITOLOGY, vol. 49, no. 1, July 1993 (1993-07-01), pages 5 - 15 |
| WILLSON ET AL., PARASITOLOGY, vol. 126, no. 1, January 2003 (2003-01-01), pages 79 - 86 |
Cited By (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP2658541B2 (en) † | 2010-12-27 | 2025-12-17 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| EP2658542B1 (en) | 2010-12-27 | 2022-01-26 | Intervet International B.V. | Topical localized isoxazoline formulation |
| EP2658541B1 (en) | 2010-12-27 | 2022-01-26 | Intervet International B.V. | Topical localized isoxazoline formulation comprising glycofurol |
| US11576382B2 (en) | 2018-02-12 | 2023-02-14 | Fmc Corporation | Naphthalene isoxazoline compounds for controlling invertebrate pests |
| US12137688B2 (en) | 2018-02-12 | 2024-11-12 | Fmc Corporation | Naphthalene isoxazoline compounds for controlling invertebrate pests |
| JP2024170642A (en) * | 2019-05-03 | 2024-12-10 | インターベット インターナショナル ベー. フェー. | Injectable pharmaceutical compositions and uses thereof |
| AU2020268618B2 (en) * | 2019-05-03 | 2025-12-04 | Intervet International B.V. | Injectable pharmaceutical compositions and uses thereof |
| RU2829447C2 (en) * | 2019-05-03 | 2024-10-30 | Интервет Интернэшнл Б.В. | Pharmaceutical compositions for injections and use thereof |
| CN113784702B (en) * | 2019-05-03 | 2024-11-05 | 英特维特国际股份有限公司 | Injectable pharmaceutical composition and use thereof |
| JP2022531590A (en) * | 2019-05-03 | 2022-07-07 | インターベット インターナショナル ベー. フェー. | Injectable pharmaceutical compositions and their use |
| WO2020225143A1 (en) * | 2019-05-03 | 2020-11-12 | Intervet International B.V. | Injectable pharmaceutical compositions and uses thereof |
| CN113784702A (en) * | 2019-05-03 | 2021-12-10 | 英特维特国际股份有限公司 | Injectable pharmaceutical composition and use thereof |
| JP7558197B2 (en) | 2019-05-03 | 2024-09-30 | インターベット インターナショナル ベー. フェー. | Injectable pharmaceutical compositions and uses thereof |
| JP7769070B2 (en) | 2019-05-03 | 2025-11-12 | インターベット インターナショナル ベー. フェー. | Injectable pharmaceutical compositions and uses thereof |
| US12497370B2 (en) | 2019-12-18 | 2025-12-16 | Elanco Tiergesundheit Ag | Isoxazoline derivatives |
| US12304903B2 (en) | 2020-07-24 | 2025-05-20 | Elanco Us Inc. | Process for making an isoxazoline compound and intermediate thereof |
| US12544336B2 (en) | 2021-09-02 | 2026-02-10 | Elanco Us Inc. | Palatable formulations |
| WO2025257633A1 (en) * | 2024-06-12 | 2025-12-18 | Boehringer Ingelheim Vetmedica Gmbh | Long-acting castor oil-containing injectable formulations and methods of use thereof |
| RU2842530C1 (en) * | 2024-12-26 | 2025-06-30 | Общество с ограниченной ответственностью "БиоФерон" | Complex anti-parasitic composition for cats |
| RU2850716C1 (en) * | 2024-12-30 | 2025-11-13 | Федеральное государственное бюджетное образовательное учреждение высшего образования "Московская государственная академия ветеринарной медицины и биотехнологии - МВА имени К.И. Скрябина" (ФГБОУ ВО МГАВМиБ - МВА имени К.И. Скрябина) | Method for changing effectiveness of antihelminthic preparation prazitel |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20230087923A1 (en) | Parasiticidal compositions comprising an isoxazoline active agent, methods and uses thereof | |
| KR102593069B1 (en) | Extended release injectable formulations comprising an isoxazoline active agent, methods and uses thereof | |
| AU2018205137B2 (en) | Long-acting injectable formulations comprising an isoxazoline active agent, methods and uses thereof | |
| DK179451B1 (en) | PARASITICIDAL SOFT CHEWABLE VETERINARY COMPOSITION COMPOSITION ISOXALINE ACTIVE AGENTS, AND USES THEREOF. | |
| TWI574623B (en) | Parasiticidal compositions comprising multiple active agents, methods and uses thereof | |
| WO2018039508A1 (en) | Method for reducing unwanted effects in parasiticidal treatments | |
| AU2015202084B2 (en) | Parasiticidal compositions comprising multiple active agents, methods and uses thereof | |
| AU2016247183B2 (en) | Parasiticidal compositions comprising multiple active agents, methods and uses thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 17761751 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 17761751 Country of ref document: EP Kind code of ref document: A1 |