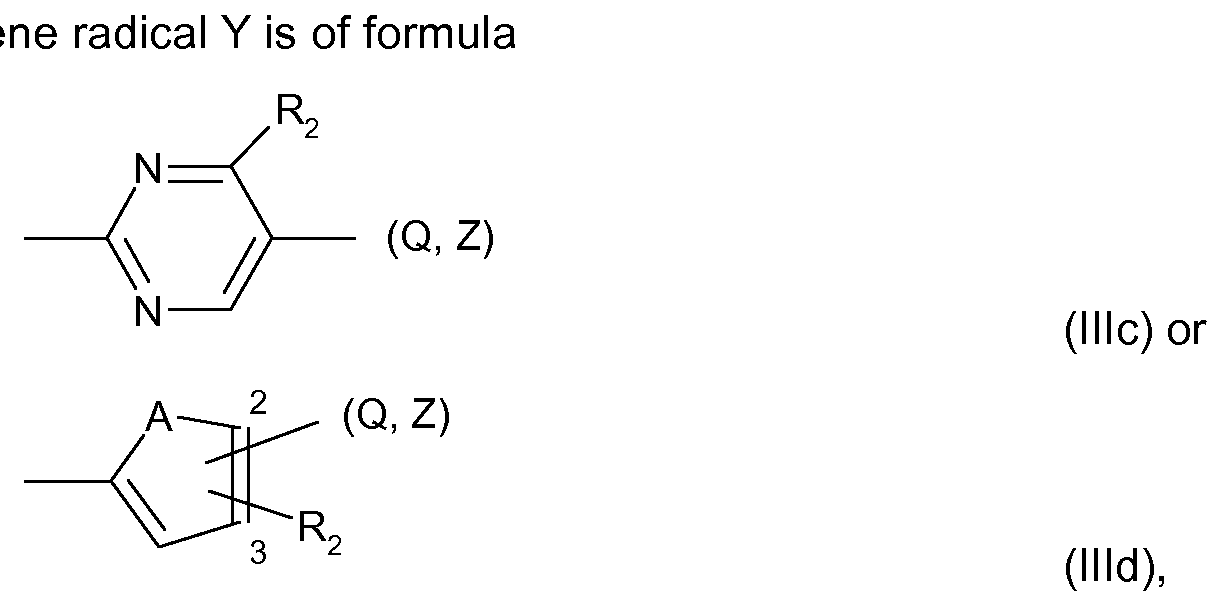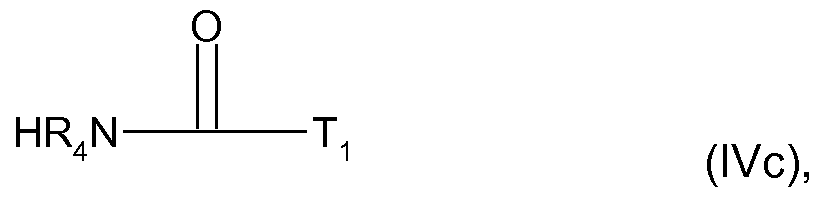WO2014090918A1 - Process for the enantiomeric enrichment of diaryloxazoline derivatives - Google Patents
Process for the enantiomeric enrichment of diaryloxazoline derivatives Download PDFInfo
- Publication number
- WO2014090918A1 WO2014090918A1 PCT/EP2013/076321 EP2013076321W WO2014090918A1 WO 2014090918 A1 WO2014090918 A1 WO 2014090918A1 EP 2013076321 W EP2013076321 W EP 2013076321W WO 2014090918 A1 WO2014090918 A1 WO 2014090918A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- enantiomer
- formula
- crc
- alkyl
- halogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 CC1c2ccc(C)cc2CC1* Chemical compound CC1c2ccc(C)cc2CC1* 0.000 description 3
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/04—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings directly linked by a ring-member-to-ring-member bond
Definitions
- the present invention relates to a process for the manufacture of pure diaryloxazoline enantiomers and their use for combating invertebrate pests in agriculture or in the veterinary field.
- aryl isoxazoline compounds has attracted a lot of attention in the agrochemical and also veterinary field.
- WO2005/085216 discloses the efficacy of said class of compounds as agrochemical pest control agents.
- the active ingredients have at least one asymmetrical C-atom, they are in general present as a racemic mixture of two enantiomers. No separation of the enantiomers is usually performed.
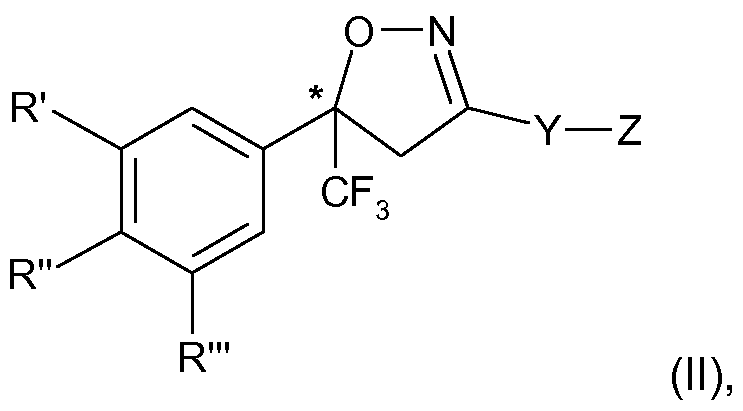
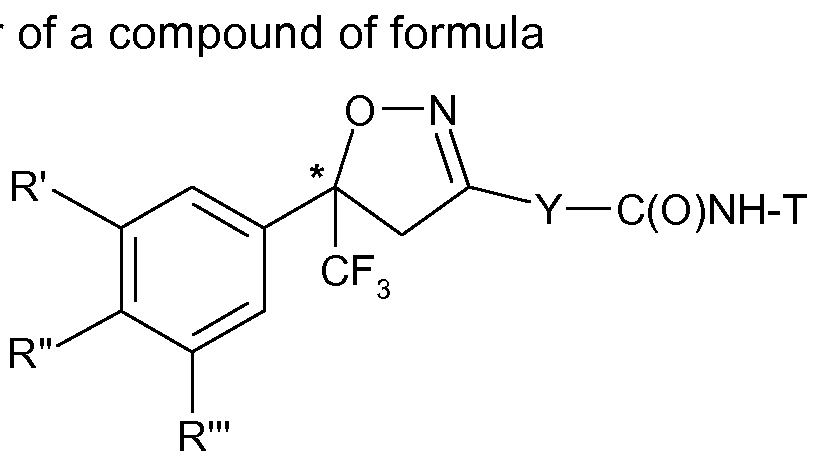
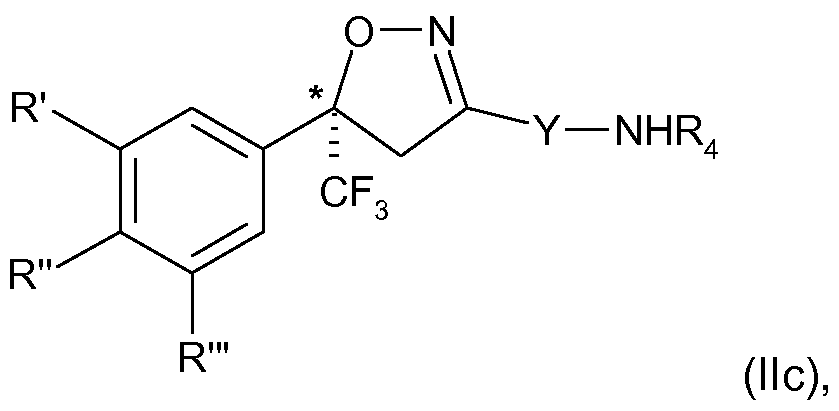
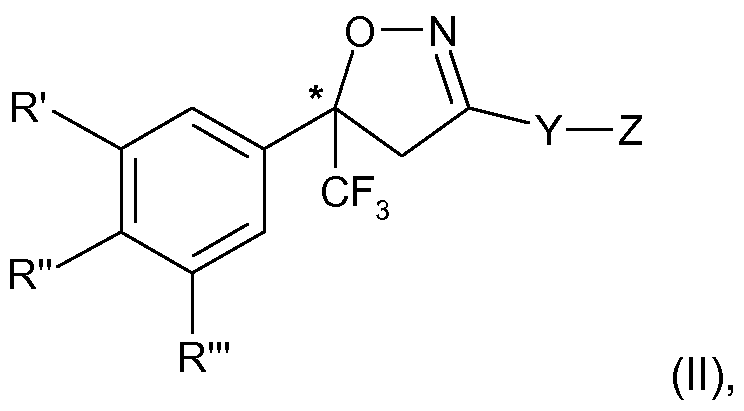
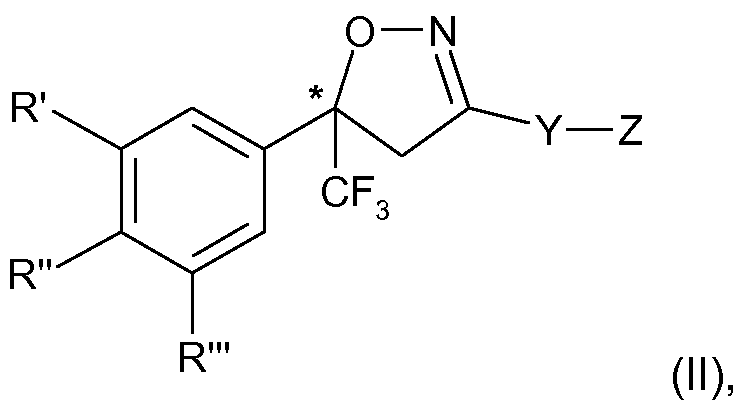
- the present invention therefore relates to a process for the manufacture of an enantiomer, preferably of the S-enantiomer, of a compound of formula
- R ⁇ R" and R'" are each independently hydrogen, halogen, cyano, CrC 2 -alkyl, halo-Ci-C 2 - alkyl, CrC 2 -alkoxy or CrC 2 -haloalkoxy, subject to the proviso that at least one of R', R" and R'" is not hydrogen;
- Y is phenylene, naphthylene, indanylene, 5- or 6-membered heteroarylene or 8-10- membered fused heterobicyclylene, which is each unsubstituted or substituted by Ci-C 2 - alkyl, Ci-C 2 -haloalkyl, halogen, nitro or cyano, and,
- Y is phenylene, naphthylene, heteroarylene or fused heterobicyclylene, Q is
- a 5- or 6-membered heteroaromatic ring comprising 1 to 3 same or different heteroatoms selected from the group consisting of O, S and N which is further unsubstituted or substituted; or is
- Y is indanylene
- Q is a group -N(R 4 )-C(0)-T 2 , wherein T 2 independently has the meaning of T above and R 4 is as defined above;
- R', R"and R'" and Y are as defined above and Z is Q; or is halogen, amino, carboxy
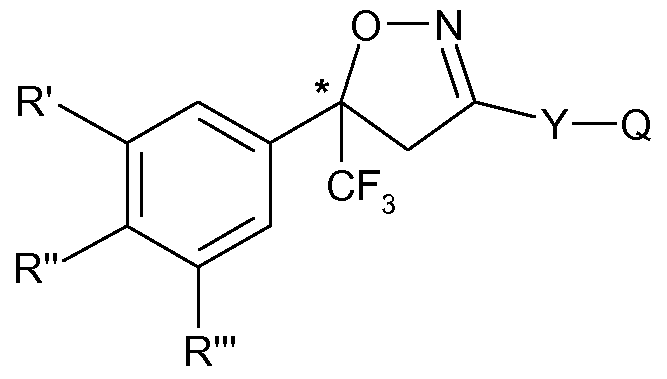
- step (B) if Z is other than Q, converting the desired enantiomer, preferably the S-enantiomer, of formula (II) from step (A) to the desired enantiomer, preferably to the S-enantiomer, of formula (I);
- step (C) racemizing the undesired enantiomer, preferably the R-enantiomer, of formula (II) from step (A) in a reaction medium comprising a base and an aprotic organic solvent at a temperature of from 30°C to 150°C, and,
- step (D) subjecting the racemate from step (C) to a further manufacturing cycle comprising step (A) and optionally (B).
- Z is Q, that is, a racemic compound of formula (I) is prepared and subjected to process steps (A), (C) and (D) in a repetitive cycle.
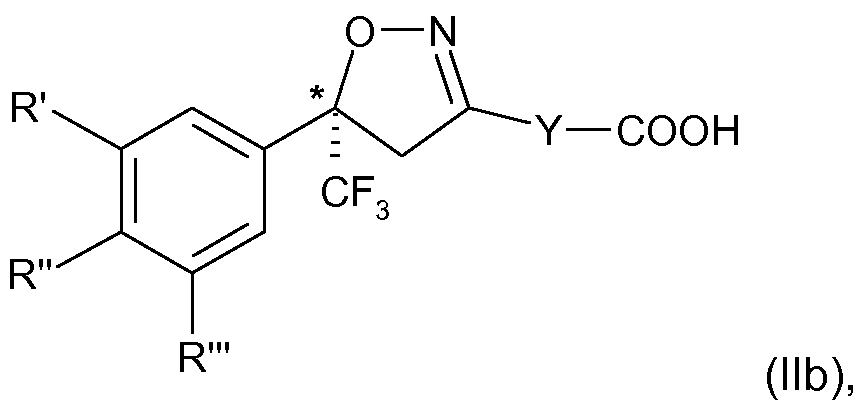
- Z is halogen, amino, carboxy (-COOH) or a salt or derivative thereof, in particular carboxy or a salt thereof, that is, a racemic intermediate compound of formula (II) is prepared and subjected to process steps (A), (B), (C) and (D) in a repetitive cycle.
- a preferred embodiment of the invention comprises a cyclic batch process, wherein one or more batches of a racemic compound of formula (II) are subjected to process steps (A) and optionally (B), the undesired enantiomer, in particular the R-enantiomer, obtained from the enantiomer separation of the one or more batches is subjected to racemization step (C), and the racemate from step (C) is subjected to a further enantiomer separation step (D).
- the undesired enantiomer, in particular the R-enantiomer, obtained from step (A) of a first batch process is racemized according to step (C) above, and the racemate obtained is added to a subsequent batch of racemic compound of formula (II) to be separated into the enantiomers according to the present invention. No compound of formula (II) is thus lost, as the undesired enantiomer of an enantiomer separation step (A) is always recovered according to step (C), and the racemate from step (C) is phased in a subsequent enantiomer separation step (A).
- R', R" and R'" are each independently of the other preferably H, halogen, CF 3 or cyano, and in particular H, CI, F or CF 3 , subject to the proviso that at least one of R', R" and R'" is not H.
- One preferred embodiment of the invention concerns compounds of formula (I), wherein R', R" and R'" are each independently of the other H, chlorine or fluorine, subject to the proviso that at least one of R', R" and R'" is not H.
- R', R" and R'" are each independently of the other H, chlorine or fluorine, subject to the proviso that at least one of R', R" and R'" is not H.
- R' and R'" are each halogen, for example chlorine or fluorine, in particular chlorine, and R" is H, chlorine or fluorine, in particular H or chlorine and especially chlorine.
- R' and R'" are CF 3 and the other one is halogen, for example chlorine or fluorine, in particular chlorine, and R" is H.
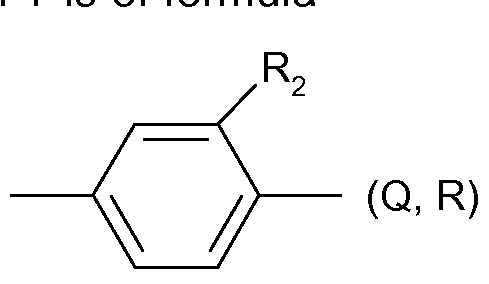
- a preferred phenylene radical Y is of formula
- R 2 is H, CrC 2 -alkyl, CrC 2 -haloalkyl, halogen, nitro or cyano, preferably H, methyl, chlorine, nitro, cyano or CF 3 , more preferably H, methyl, chlorine CF 3 or cyano, in particular methyl, chlorine CF 3 or cyano, and especially methyl.
- a preferred naphthylene radical Y is of formula
- a preferred indanylene radical Y is of formula
- R 2 ' is H, methyl, chlorine, nitro, cyano or CF 3 , in particular H.
- R 2 each the above given meanings and preferences apply, and A is O or S, in particular S.
- a particularly preferred embodiment of the heteroarylene radical Y is a radical of formula (llld) above, wherein A is S, Q or Z is located in the 2-position, and R 2 is methyl and is located in the 3-position.
- Embodiments of a fused heterobicyclylene emcompass for example, a radical of formula
- a suitable heterocyclic ring Q is, for example, a 5- or 6-membered heteroaromatic ring having from 1 to 4, preferably from 1 to 3 same or different heteroatoms selected from the group consisting of N, O and S, which is further unsubstituted or substituted by one or more substituents selected from the group consisting of halogen, cyano, nitro, CrC 4 -alkyl, Ci-C 4 -haloalkyl, hydroxy, Ci-C 4 -alkoxy, Ci-C 4 -haloalkoxy, Ci-C 4 - alkylthio, Ci-C 4 -haloalkylthio, CrC 4 -alkylsulfinyl, CrC 4 -haloalkylsulfinyl, CrC 4 -alkylsulfon
- heteroaromatic ring Q is preferably unsubstituted or substituted by 1 to 3, in particular 1 or 2, same or different substituents selected from the group consisting of halogen, cyano, nitro, CrC 2 -alkyl, CrC 2 -haloalkyl, Ci-C 2 -alkoxy, Ci-C 2 -haloalkoxy, Ci-C 2 -haloalkylthio, d- C 4 -alkoxycarbonyl, C 2 -C 3 -alkanoyl, aminocarbonyl, N-mono- or N,N-di-CrC 3 - alkylaminocarbonyl and C(S)NH 2 .
- the heteroaromatic ring Q is most preferably
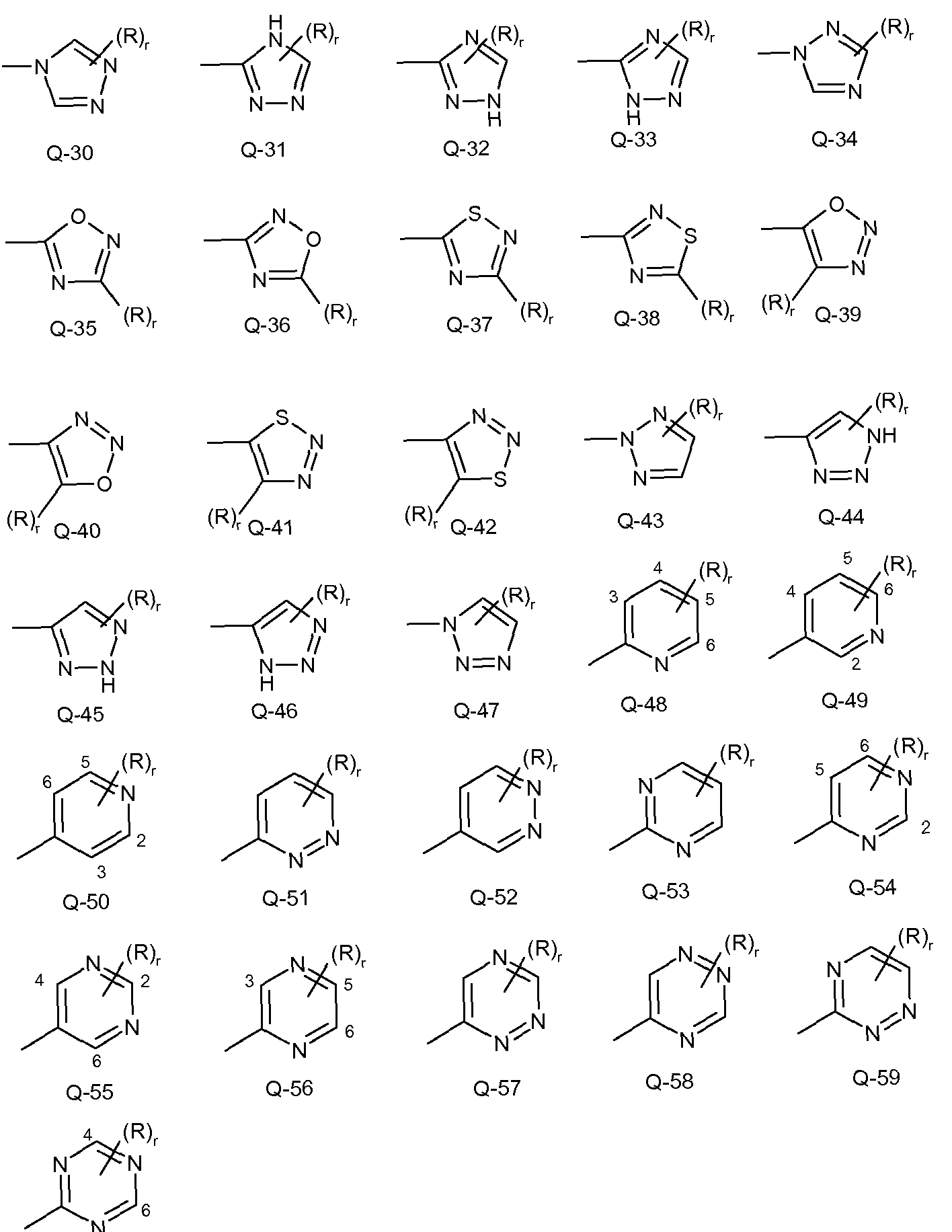
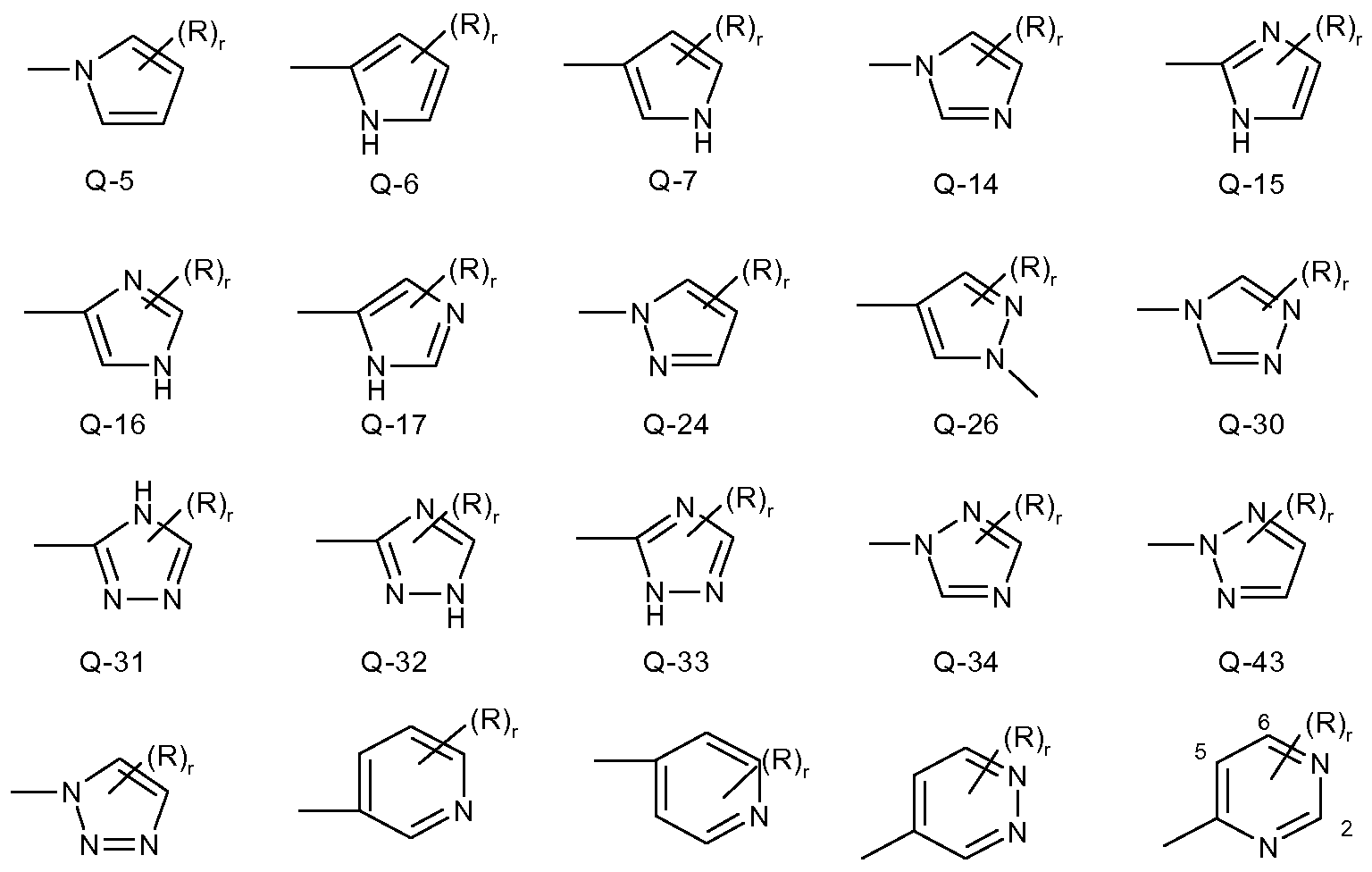
- Examples of a 5- or 6-membered heteroaromatic rings optionally substituted with from one or more substituents include the rings Q-1 through Q-60 illustrated in Exhibit 1 wherein R is any substituent as defined before including the preferences given, and r is an integer from 0 to 4, limited by the number of available positions on each Q group.
- R is any substituent as defined before including the preferences given
- r is an integer from 0 to 4, limited by the number of available positions on each Q group.
- Q-28,- Q-29, Q-35, Q-36, Q-37, Q-38, Q-39, Q-40, Q-41 and Q-42 have only one available position, for these Q groups r is limited to the integers 0 or 1 , and r being 0 means that the Q group is
- a preferred heterocyclic ring Q is of formula
- r is an integer from 0 to 3 and R is independently selected from the group given before for the heteroaromatic ring including the preferences.
- Q is particularly preferred the unsubstituted radical Q-14, Q-24, Q-34, Q-43 or Q-47, wherein r is 0 in each case.
- Q is especially preferred a radical Q-14, Q-34 or Q-47, wherein r is 0.
- Q is a group -C(0)N(Ri)-T (embodiment (ii)), is preferably H, methyl, ethyl or acetyl and in particular H.
- T as alkyl is preferably CrC 4 -alkyl, more preferably CrC 2 -alkyl and particularly preferably Ci-alkyl, which is each unsubstituted or substituted as defined above.
- the alkyl radical T is preferably unsubstituted or substituted by halogen; CrC 4 -alkoxy; C C 4 -alkoxycarbonyl; N-Ci-C 6 -alkylaminocarbonyl which is unsubstituted or substituted in the alkyl portion by halogen, cyano, ethenyl or ethynyl; or 5- to 6-membered heterocyclyl which is in turn unsubstituted or substituted by halogen-, CrC 2 -alkyl- or CrC 2 -haloalkyl.
- a preferred N-alkylaminocarbonyl substituent of the alkyl radical T is N-Ci-C 2 - alkylaminocarbonyl, which is unsubstituted or further substituted in the alkyl moiety by halogen, cyano, ethenyl or ethynyl.
- N-alkylaminocarbonyl-substituted alkyl is preferably N-ethylaminocarbonylmethyl, or a radical -CH 2 -C(0)NH-CH 2 CF 3 , -CH 2 -C(0)NH-CH 2 CN or-CH 2 -C(0)NH-CH 2 C ⁇ CH.
- T is heterocyclyl-substituted alkyl
- preferred meanings of heterocyclyl include pyridyl, pyrimidinyl, thiazolyl, oxazolyl, tetrahydrofuranyl, thietanyl, thietanyl-1 ,1 -dioxide or oxetanyl.
- Preferred heterocyclyl-substituted alkyl radicals T are in particular 2-pyridylmethyl, 2- tetrahydrofuranylmethyl.
- T as heterocyclyl preferably denotes as 4- to 6-membered ring comprising 1 to 3 same or different heteroatoms selected from the group consisting of O, S, S(O), S(0 2 ) and N, which is each unsubstituted or substituted by halogen, Ci-C 2 -alkyl or Ci-C 2 -haloalkyl.
- T is 4- to 6-membered heterocyclyl
- preferred meanings of heterocyclyl include pyridyl, pyrimidyl, thiazolyl, oxazolyl, tetrahydrofuranyl, thietanyl, thietanyl-1 -oxide, thietanyl-1 ,1 - dioxide or oxetanyl and in particular 2- 3- or 4- pyridyl, 3- 4- or 5- pyrimidyl, 2- or 3- tetrahydrofuranyl, thietan-3-yl or oxetan-3-yl and even more preferred 5-CI-pyrimid-3-yl, 3- tetrahydrofuranyl, thietan-3-yl, thietan-3-yl-1 -oxide, thietan-3-yl-1 ,1 -dioxide or oxetan-3-yl.
- Q is a group -C(0)N(R 1 )-T, is preferably H, methyl, ethyl or acetyl and T is Ci-C 2 -alkyl; CrC 2 -haloalkyl; Ci-C 2 -alkoxycarbonyl-CrC 2 -alkyl; Ci-C 2 -alkyl which is substituted by pyridyl, pyrimidinyl, thiazolyl, oxazolyl or tetrahydrofuranyl; N-Ci-C 2 -alkylaminocarbonyl-Cr C 2 -alkyl which is unsubstituted or substituted in the N-alkyl moiety by halogen, cyano, ethenyl or ethynyl; or is pyridyl; pyrimidyl; thiazolyl; oxazolyl; tetrahydrofuranyl; thietanyl; thi
- Q is a group -C(0)N(R 1 )-T, is most preferably H, methyl or ethyl, and T is Ci-C 2 -alkyl; CrC 2 -haloalkyl; methyl which is substituted by pyridyl, pyrimidinyl, thiazolyl, oxazolyl or tetrahydrofuranyl; methyl which is substituted by N-Ci-C 2 -alkylaminocarbonyl or by N-Ci-C 2 - alkylaminocarbonyl substituted in the alkyl moiety by halogen, cyano, ethenyl or ethynyl; or is pyridyl; pyrimidyl; tetrahydrofuranyl; thietanyl; thietanyl-1 -oxide; thietanyl-1 ,1 -dioxide; or oxetanyl.
- Q is a group -C(0)N(R 1 )-T, is particularly preferably H, and T is CrC 2 -alkyl; a radical -CH2CF 3; N-ethylaminocarbonylmethyl; a radical -CH2-C(0)NH-CH2CF 3!
- R 3 is preferably H or C C 2 -alkyl or cyano, more preferably H or methyl, and in particular H.
- R 4 is preferably H or CrC 2 -alkyl, in particular H.
- R 4 is preferably H or CrC 2 -alkyl, in particular H.
- T-i as optionally substituted alkyl is preferably straight-chain or branched CrC 4 -alkyl, which is each unsubstituted or substituted by C 3 -C 6 -cycloalkyl, halogen, cyano, CrC 4 -alkoxy, d- C 2 -haloalkoxy, CrC 4 -alkylthio, CrC 2 -haloalkylthio, CrC 4 -alkylsulfinyl, CrC 4 -haloalkylsulfinyl, CrC 4 -alkylsulfonyl, CrC 4 -haloalkylsulfonyl, Ci-C 2 -alkylcarbonylamino, CrC 2 -haloalkyl- carbonylamino or 4- to 6-membered heterocyclyl.
- Especially preferred alkyl radicals ⁇ are straight-chain or branched CrC 4 -alkyl or CrC 4 -alkyl which is substituted by cyclopropyl, halogen, cyano, CrC 2 -alkoxy, CrC 2 -haloalkoxy, CrC 2 -alkylthio, CrC 2 -alkylsulfinyl, CrC 2 - alkylsulfonyl, Ci-C 2 -haloalkylcarbonylamino, pyridyl, pyrimidyl, thiazolyl, oxazolyl, thietanyl, oxetanyl, dioxolanyl, methyldioxolanyl, dioxanyl or tetrahydrofuryl.
- T-i as alkyl is especially preferred straight-chain or branched CrC 4 -alkyl, Ci-C 3 -haloalkyl, cyclopropylmethyl, cyano-CrC 2 -alkyl, Ci-C2-alkoxy-Ci-C 2 -alkyl, Ci-C 2 -alkylthio-Ci-C 2 -alkyl, Ci-C2-alkylsulfinyl-Ci-C 2 -alkyl, Ci-C2-alkylsulfonyl-Ci-C 2 -alkyl, or methyl which is substituted by 1 ,3-dioxolan-2-yl, 2-methyl-1 ,3-dioxolan-2-yl or tetrahydrofuran-2- or -3-yl.
- alkyl radicals ⁇ are straight-chain or branched CrC 4 -alkyl; CrC 2 - alkyl which is substituted by halogen, cyano, CrC 2 -alkoxy, CrC 2 -alkylthio or CrC 2 -alkylsulfonyl; or 2-methyl-1 ,3-dioxolan-2-yl-methyl.
- Ti is C 3 -C 6 -cycloalkyl
- said cycloalkyl is preferably cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl, in particular cyclopropyl.
- T-i is 4- to 6-membered heterocyclyl
- said heterocyclyl is, for example, a 4-6-membered heteroaromatic ring, preferably a thienyl, furyl, oxazolyl, thiazolyl, pyridyl or pyrimidinyl radical, which is each unsubstituted or substituted by CrC 2 -alkyl, CrC 2 -haloalkyl or Ci-C 4 - alkoxycarbonyl.
- Especially preferred heteroaromatic radicals ⁇ are 2-, 3- or 4-pyridyl, 2- or 4-pyrimidinyl, 2-thiazolyl, 2-furyl or 2-thienyl.
- a further preferred heterocyclic radical ⁇ is, for example, a 4- to 6-membered
- heteroaliphatic ring selected from the group of thietanyl, for example thietan-3-yl, oxothietanyl, for example thietan-3-yl-1 -oxide or thiethan-3-yl-1 ,1 -dioxide, oxetanyl, for example oxetan-3-yl, azetidinyl, pyrrolidinyl, tetrahydrofuranyl, tetrahydrothiophenyl, piperidinyl, piperazinyl, morpholinyl, tetrahydropyranyl and thianyl which is each
- heteroaliphatic ring radicals ⁇ include pyrrolidinyl, tetrahydrofuranyl, tetrahydrothiophenyl, piperidinyl, piperazinyl, morpholinyl, thietanyl, thietanyl oxide or thietanyl dioxide which are each unsubstituted or substituted by CrC 2 -alkyl, Ci-C 2 -haloalkyl or Ci-C 4 -alkoxycarbonyl, and in particular pyrrolidine-1 -yl, tetrahydrofuran-2-yl,
- Q as a group -CH(R3)-N(R4)-C(0)-Ti is most preferably a radical -CH 2 -NH-C(0)-C C 2 -alkyl, -CH 2 -NH-C(0)-cyclopropyl, -CH 2 -N H-C(0)-(CH 2 ) 1-2 -0-Ci-C 2 -alkyl,
- Y is phenylene, naphthylene heteroarylene or fused heterobicyclylene, particular preferred meanings of Q are a radical
- One embodiment of the invention relates to the manufacture of the S-enantiomer of a compound of the formula (I) above wherein R' and R'" are each independently of the other halogen, for example chlorine or fluorine, in particular chlorine, R" is H or halogen, preferably H or chlorine and in particular chlorine, Y is a radical of formula (llld) above, wherein A is S, Q is a radical (q2) to (q25) as mentioned above in the 2-position, and R 2 is methyl and is located in the 3-position.
- a further embodiment of the present invention comprises a process for the manufacture of the S-enantiomer
- R', R" and R'" and Y are as defined and Z carboxy (-COOH) or a salt thereof; into the enantiomers and collecting the S-enantiomer;
- Veterinary acceptable salts of the compounds of formula (I) as well as salts of the compounds of formula (II) include acid-addition salts with inorganic or organic acids; just by way of example, the following acids may be mentioned: hydrobromic, hydrochloric, nitric, phosphoric, sulfuric, acetic, butyric, fumaric, lactic, maleic, malonic, oxalic, propionic, salicylic, tartaric, 4-toluenesulfonic or valeric acid.
- Enantiomer separation of a compound of formula (II) according to step (A) may be performed in a manner known per se, for example by simple recrystallization, kinetic resolution, by means of biotechnological methods (whole cells, enzymes, etc.), by chromatographic methods, or by methods that are based on the difference of
- a compound of formula (II) may be separated into its enantiomers by chiral column chromatography, in particular by chromatography on chiral adsorbents using a Simulated Moving Bed (SMB) system.
- SMB Simulated Moving Bed
- a liquid mobile phase carrying the racemic compound of formula (II) to be separated is contacted with a chiral stationary phase in a system that is constituted of a number of chromatographic columns in a circular flow arrangement, separated by ports where inlet and outlet streams can be fed or collected.
- a suitable chiral stationary phase is, for example, composed of a chiral polysaccharide (for example Chiralpak® AS-V, AD).
- the liquid mobile phase typically comprises one or more alcohols, for example methanol and/or ethanol , optionally in admixture with an organic acid, for example, formic acid.
- a suitable SMB system is described, for example, in
- a racemic compound of formula (II) which is an acid or amine is separated into the enantiomers by diastereomeric recrystallization.
- an optically pure reagent which is an amine or acid, is reacted with a racemic compound of formula (II), wherein Z is amino, carboxy or a salt thereof, thereby forming pairs of diastereomeric salts, which can be separated by conventional techniques in physical chemistry, i.e. by crystallization.
- a typical optical pure reagent is an organic acid, for example, (+)-D-di-0-benzoyl-tartaric acid, (-)-L- di-O-benzoyltartaric acid, (-)-di-0,0'-p-tolyl-L-tartaric acid, (+)- di-0,0'-p-tolyi-D-tartaric acid, R(+)-malic acid, S-(-)-malic acid, (+)-camphanic acid, (-)-camphanic acid, R(-)-1 ,V- binaphthalene-2,2'-diyl hydrogen phosphate, S(+)-1 ,1 '-binaphthalene-2,2'-diyl hydrogen phosphate, (+)-camphoric acid, (-)-camphoric acid, S(+)-2-phenylpropionic acid, R(-
- a typical optical pure reagent is an optical pure amine, for example (R)-(+)-1 -(4-methylphenyl)ethylamine, (S )-(-)- 1 -(4-methylphenyl)ethylamine, (R)-(+)-a-methylbenzylamin, (S)-(-)-a-methylbenzylamin, (R)- (-)-aminoindane, (S)-(+)-aminoindane, (R)-(+)-N,a-dimethylbenzylamine, (S)-(-)-N,a- dimethylbenzylamine, (R)-(+)-N,N-dimethyl-1 -phenyletyhlamine, (S)-(-)-N,N-dimethyl-1 - phenyletyhlamine, (R)-(-)-1 -cyclohexy
- a racemic compound of formula (II), wherein Z is carboxy or a salt thereof is separated into its enantiomers by reaction with an optical active amine, in particular (R)-(+)-1 -(4-methylphenyl)ethylamine.
- the formation of the diastereomeric salt may be performed in a polar solvent or a solvent mixture comprising at least one polar solvent.
- Suitable solvents are for example, halogenated hydrocarbons, for example chlorobenzene, dichlorobenzene, bromobenzene, dichloromethane, trichloromethane, tetrachloromethane, dichloroethane, trichloroethene or tetrachloroethene; alcohols, such as methanol, ethanol, propanol, isopropanol, butanol, isobutanol or tert.
- ethers such as diethyl ether, dipropyl ether, diisopropyl ether, dibutyl ether, tert. -butyl methyl ether, ethylene glycol monomethyl ether, ethylene glycol monoethyl ether, ethylene glycol dimethyl ether, dimethoxydiethyl ether, tetrahydrofuran or dioxane; ketones, such as acetone, methyl ethyl ketone or methyl isobutyl ketone; amides, such as N,N-dimethylformamide, ⁇ , ⁇ -diethylformamide, N,N-dimethylacetamide, N- methylpyrrolidone or hexamethylphosphoramide; esters, such as methyl acetate, ethylacetate; nitriles, such as acetonitrile or propionitrile; sulfoxides, such as dimethyl sulfoxide; or
- Preferred solvents for the formation and crystallization of the diastereomeric salts according to the invention are alcohols, in particular ethanol, isopropanol, n-, sec. or tert. -butanol, nitriles, in particular acetonitrile, ethers, in particular tert. - butyl methylether, esters, in particular methylacetate, or mixtures therof, or a mixture one of or more of the above mentioned solvents with water.
- a particularly preferred reaction medium for the formation of the diastereomeric salts is a mixture of water, a C 2 -C 4 -alcanol and a dipolar aprotic solvent such as acetonitrile or the like.
- the formation of the diastereomeric salt may be performed at a temperatu re ranging from about 1 0° C to the reflux point of the solvent or solvent mixture. Salt formation is carried out preferably at a temperature ranging from room temperature to about 100°C.
- the mixture of the racemate to be separated, the optical active reagent and the solvent(s) is heated to an elevated temperature, for example to 30 to 100°C or preferably to 50 to 80°C, kept at this temperature for some time and then cooled, for example, to room temperature in order to affect crystallization of one of the diastereomeric salts.
- the reaction time is not critical; in general, a time period of 0.5 minutes to 6 hours, preferably 1 to 4 hours, and in particular from 1 .5 to 3 hours at an elevated temperature is sufficient for the formation of the diastereomeric salts.
- one of the diastereomeric salts preferably the one covering the S-enantiomer, usually crystallizes and may be isolated by filtration.
- the crystallized diastereomeric salt may be purified to the desired diastereomeric purity, for example by recrystallization in a suitable solvent or solvent mixture.
- the purified diastereomeric salt is then dissociated in basic or acidic medium in a suitable solvent or solvent mixture, and the desired (S)-enantiomer is recovered from the racemic mixture of the compound of formula (II).
- the desired S-enantiomer of the compound of formula (II) is collected and optionally further purified by processes known in the art, for example by recrystallization.
- the optical purity of the S-enantiomer as collected is for example ⁇ 90%, preferably ⁇ 95% and in particular ⁇ 98%.
- the S-enantiomer of formula (II), wherein Z is different from Q, is converted to a S- enantiomer of formula (I) according to step (B) in a manner known per se, for example from WO2010/070068 or WO201 1/157748.
- R', R", R'" and Yare as defined above and Hal is halogen, for example, fluoro, chloro or bromo, may be reacted with a compound of formula
- Typical catalysts are, for example copper iodide, a palladium catalyst or an inorganic base such as sodium or potassium carbonate.
- R', R", R'" and Y are as defined above, may be first converted to the respective acid halide which is reacted with a compound of formula
- R-i and T are as defined above, optionally in the presence of a base, to yield the respective S-enantiomer of formula (I), wherein Q is a group -C(0)N(R 1 )-T.
- An S-enantiomer of formula (l ib) above may also be first converted to the respective aldehyde, optionally via a respective ester, which is subjected to a triethylsilane-promoted reductive amination with a compound of formula
- R 4 and ⁇ is as defined above, to yield the S-enantiomer of formula (I), wherein Q is a group -CH(R 3 )-N(R 4 )-C(0)-T 1 .
- T 2 and Hal is as defined above, optionally in the presence of a base, to yield the respective S-enantiomer of formula (I), wherein Q is a group -NR 4 -C(0)T 2 .
- Suitable organic solvents in racemization step (C) are, for example aromatic, aliphatic (cyclic and alicyclic) and halogenated hydrocarbons, such as benzene, toluene, xylene, mesitylene, tetralin, chlorobenzene, dichlorobenzene, bromobenzene, petroleum ether, hexane, cyclohexane, dichloromethane, trichloromethane, tetrachloromethane, dichloro- ethane, trichloroethene or tetrachloroethene; alcohols, such as methanol, ethanol, propanol, isopropanol, butanol, isobutanol or tert.
- aromatic, aliphatic (cyclic and alicyclic) and halogenated hydrocarbons such as benzene, toluene, xylene, mesitylene, te
- ethers such as diethyl ether, dipropyl ether, diisopropyl ether, dibutyl ether, tert. -butyl methyl ether, ethylene glycol monomethyl ether, ethylene glycol monoethyl ether, ethylene glycol dimethyl ether, dimethoxydiethyl ether, tetrahydrofuran or dioxane; esters, such as methyl acetate, ethyl acetate or tert.
- ketones such as acetone, methyl ethyl ketone or methyl isobutyl ketone
- amides such as N,N-dimethylformamide, ⁇ , ⁇ -diethylformamide, N,N-dimethylacetamide, N- methylpyrrolidone or hexamethylphosphoramide
- nitriles such as acetonitrile or propionitrile
- sulfoxides such as dimethyl sulfoxide (DMSO).
- Preferred groups of organic solvents applied in step (C) according to the process of the invention are aromatic hydrocarbons or halogenated aromatic hydrocarbons, preferably benzene, toluene, xylene, mesitylene, or tetralin, especially toluene or o-, m- oder p-xylene, and in particular toluene; alcohols, for example ethanol, nitriles, for example acetonitrile; or DMSO.
- the amount of organic solvent being present in step (C) may be, for example, from 30 to 90% (w/w), in particular from 50 to 90% (w/w) based on the entire reaction mixture.
- reaction medium of step (C) may comprise water, in particular, if an alkali metal or alkaline earth metal hydroxide is used as the base.
- the amount of water being present in the reaction mixture, if present, may be, for example, from 1 to 20% (w/w), preferably from 1 .5 to 10% (w/w), based on the entire reaction mixture.
- Suitable bases for racemization step (C) are, for example e.g. alkali metal or alkaline earth metal hydroxides, for example lithium, sodium or potassium hydroxide; ammonium hydroxide; alkali metal or alkaline earth metal hydrides, for example sodium hydride, calcium hydride; alkali metal or alkaline earth metal alkanolates, for example sodium methanolate sodium ethanolate, sodium butanolate, potassium tert.-butanolate; alkali metal carbonates, for example potassium carbonate or cesium carbonate; amines, for example triethylamine, N,N-diisopropylethylamine; carbocyclic amines, for example 2,2,6,6- tetramethylpiperidine, 1 ,5-diazabicyclo[5.4.0]undec-5-ene (DBU) or 1 ,5- diazabicyclo[5.4.0]undec-7-ene (DBA); lithium diisopropylamide
- a preferred base according to the present invention is an alkali metal hydroxide, in particular sodium hydroxide.
- the base is in general present in molar excess relative to the amount of enantiomer to be racemized.
- a preferred reaction medium of step (C) comprises an aromatic hydrocarbon, in particular toluene, water and an alkali metal hydroxide, in particular sodium hydroxide.
- phase transfer catalysts are, for example, quaternary ammonium or phosphonium salts, preferably quaternary ammonium salts, in particular N-methyl-N,N-dioctyloctan-1 - ammonium chloride, benzyl trimethylammonium chloride, tetrabutylammonium bromide, or tributyl methylammonium chloride.
- the amount of phase transfer catalyst, if present in step (C) may be, for example, from 0.1 to 10% (w/w), preferably from 0.5 to 5% (w/w), and in particular from 1 to 4% (w/w) based on the entire reaction mixture.
- a further preferred reaction medium of step (C) according to the invention comprises an aromatic hydrocarbon, in particular toluene, water, a phase transfer catalyst, in particular a quaternary ammonium salt such as tributyl methylammonium chloride and an alkali metal hydroxide, in particular sodium hydroxide.
- a suitable reaction mixture according to step (C) comprises
- a preferred reaction mixture according to step (C) comprises
- phase transfer catalyst 0.5 to 5% (w/w) quaternary ammonium salt as phase transfer catalyst
- a particularly preferred reaction mixture according to step (C) comprises
- alkali metal hydroxide selected from sodium and potassium hydroxide
- phase transfer catalyst selected from tetrabutyl ammonium bromide and tributyl methylammonium chloride
- ad 100% (w/w) aromatic hydrocarbon selected from toluene or o-, m- oder p-xylene.
- the racemization reaction according to step (C) advantageously takes place in a temperature range of about 50°C to about 120°C , preferably from ca. 50°C to ca. 100°C, and in particular from 60°C to 90°C.
- an R-enantiomer of formula (II) is racemized in a reaction medium comprising an aromatic hydrocarbon, preferably toluene, water, a phase transfer catalyst and a base which is an alkali metal hydroxide at a temperature from about 50°C to about 100°C, preferably from 60 to 90°C.
- the reaction time may vary within wide limits, for example between 1 and 100 hours, preferably between 2 and 72 hours, more preferably between 2 and 24 hours and in particular between 3 and 12 hours.
- a particularly preferred reaction time is from 4 to 8 hours.
- R', R" and R'" are each independently of the other preferably H, CI, F or CF 3 , subject to the proviso that at least one of R', R" and R' is not H;
- Y is a radical
- R', R" and R'" and Y are as defined above and Z carboxy (-COOH) or a salt thereof; into the enantiomers and collecting the S-enantiomer; (B) converting the S-enantiomer of formula (II) from step (A) to the S-enantiomer of formula
- step (C) racemizing the R-enantiomer of formula (II) from step (A) in a reaction medium comprising a base and an aprotic organic solvent at a temperature of from 50 to 120°C, and,
- step (D) subjecting the racemate from step (C) to a further manufacturing cycle comprising steps (A) and (B).
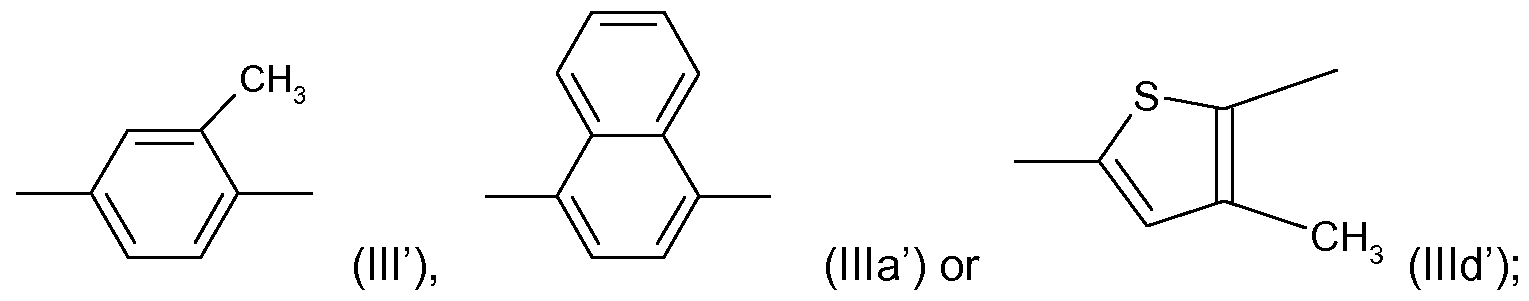
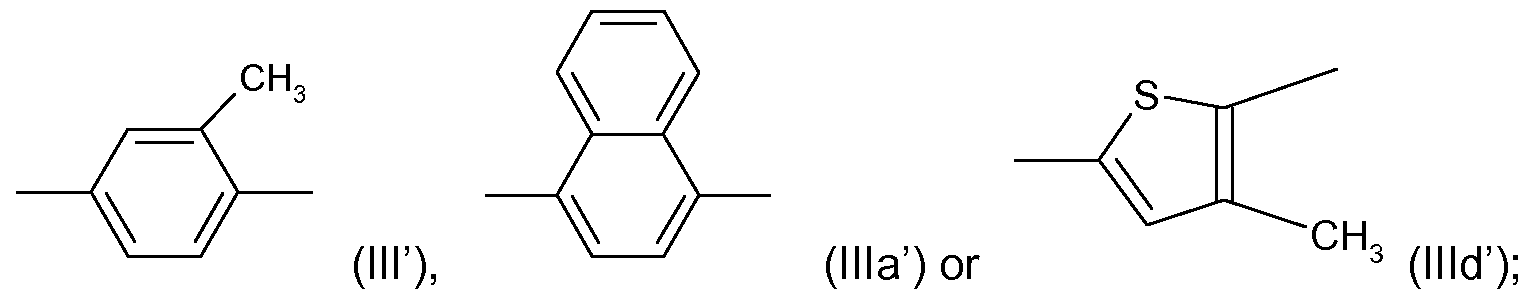
- R' and R'" are each independently CI or CF 3 , R" is H or CI; and Y is a radical ( ⁇ ), (Ilia') or (llld') as shown above;
- R', R" and R'" and Y are as defined above and Z is carboxy (-COOH) or a salt thereof; into the enantiomers, preferably by diastereomeric recrystallization, and collecting the S-enantiomer;
- step (C) racemizing the R-enantiomer of formula (II) from step (A) in a reaction medium comprising an aromatic hydrocarbon, preferably toluene, water, a phase transfer catalyst and a base which is an alkali metal hydroxide at a temperature from about 50°C to about 100°C, preferably from 60 to 90°C; and (D) subjecting the racemate from step (C) to a further manufacturing cycle comprising steps (A) and (B).
- a reaction medium comprising an aromatic hydrocarbon, preferably toluene, water, a phase transfer catalyst and a base which is an alkali metal hydroxide at a temperature from about 50°C to about 100°C, preferably from 60 to 90°C
- step (D) subjecting the racemate from step (C) to a further manufacturing cycle comprising steps (A) and (B).
- step (ii) 90 kg of the butanone bromothiophene from step (i) and 1000 kg ethanol are cooled below 5°C, and 13 kg hydroxylammonium chloride are added to the reaction mixture.
- Example 2 Enantiomer separation of racemic isoxazoline thiophene carboxylic acid of formula (II * ) and collection of the S-enantiomer
- the chiral purity of the product may be further increased by a further recrystallization step with acetonitrile/water (9:1 ) at 60°C and subsequent cooling, (chiral purity >98%).
- Example 3 Racemization of (R)-isoxazoline thiophene carboxylic acid
- step (i) The dichloromethane solution of step (i) is added to a mixture of 5.1 kg 2-amino-2',2',2'- trifluoroethyl-acetamide hydrochloride and 9.8 kg of triethylamine in 93 kg of dichloromethane at 5°C.
- the reaction mixture is stirred for additional 5 hours and is then extracted with 4% hydrochloric acid, 8% sodium hydrogen carbonate and water. Most of the organic layer is then removed under vacuum, and 50 kg of toluene are added.
- the reaction mixture is kept at 40°C, and 290 kg of heptane are added slowly to precipitate the product.
- the suspension obtained is cooled to below 5°C, and the crude product is isolated by filtration and is washed with 25 kg of heptane. Crystallization of the product may be performed as appropriate.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
The present invention concerns a process for the manufacture of the S-enantiomer of a compound of formula (I) wherein the variables are as defined in the specification and claims, comprising the steps of (A) separating a racemic compound of formula (II) wherein R', R"and R'" and Y are as defined above and Z is Q; or is halogen, amino, carboxy (-COOH); into the enantiomers and collecting the S-enantiomer; (B) if Z is other than Q, converting the S-enantiomer of formula (II) from step (A) to a S- enantiomer of formula (I); (C) racemizing the R-enantiomer of formula (II) from step (A) in a reaction medium comprising a base and an aprotic organic solvent at a temperature of from 30°C to 150°C, and (D) subjecting the racemate from step (C) to a further manufacturing cycle comprising step (A) and optionally (B). Accordingly, a cyclic process is provided, wherein the undesired enantiomer of a first enantiomer separation step is racemized and then phased in a further enantiomer separation step.
Description
PROCESS FOR THE ENANTIOMERIC ENRICHMENT OF DIARYLOXAZOLINE
DERIVATIVES
The present invention relates to a process for the manufacture of pure diaryloxazoline enantiomers and their use for combating invertebrate pests in agriculture or in the veterinary field.
The chemical class of aryl isoxazoline compounds has attracted a lot of attention in the agrochemical and also veterinary field. For example, WO2005/085216 discloses the efficacy of said class of compounds as agrochemical pest control agents. As the active ingredients have at least one asymmetrical C-atom, they are in general present as a racemic mixture of two enantiomers. No separation of the enantiomers is usually performed.
It has now been found out that the antiparasiticidal activity of the two enantiomers of a given diarylisoxazoline is very different; in particular, the respective activity of the S- enantiomer in general exceeds that of the corresponding R-enantiomer by orders of magnitude, and the R-enantiomer often does not exhibit any antiparasiticidal activity at all when applied in a commonly used concentration. Therefore it would be desirable to develop a product containing only the active S-enantiomer and few or no R-enantiomer at all.
Manufacturing of pure enantiomers is usually expensive and time-consuming; following enantiomer separation only 50% or even less of the original product have the desired configuration and the other 50% or more product with the "wrong" configuration have to be discarded. Accordingly, there is a need for recovering the undesired R-enantiomer in order to make manufacturing feasible on an industrial scale. Surprisingly, a synthetic route now has been found to racemize diaryloxazoline enantiomers. This opens the opportunity for a cyclic process, wherein the undesired enantiomer of a first enantiomer separation step is racemized and then phased in a further enantiomer separation step.
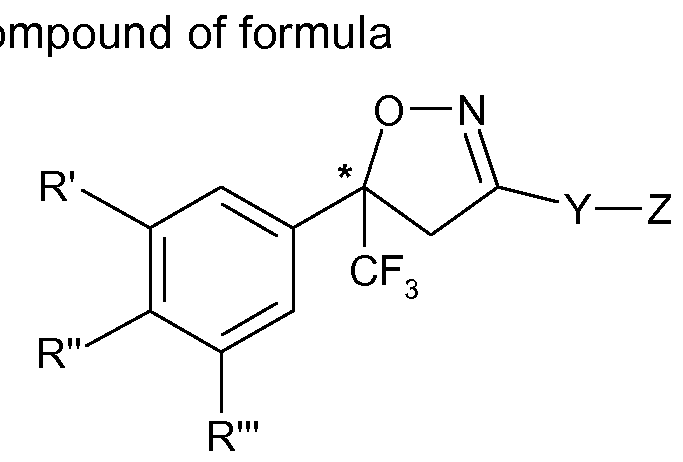
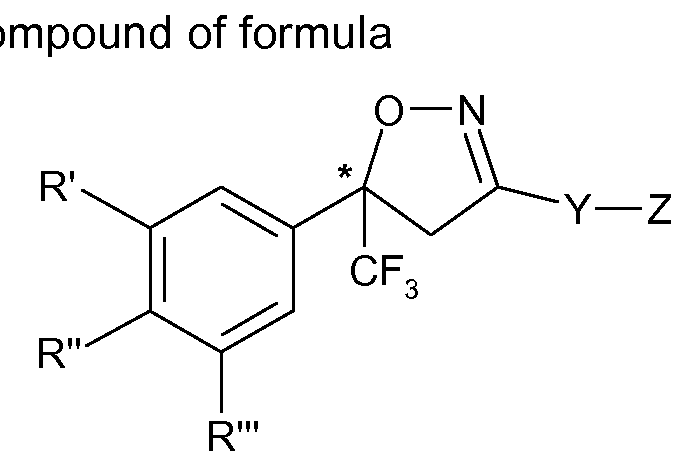
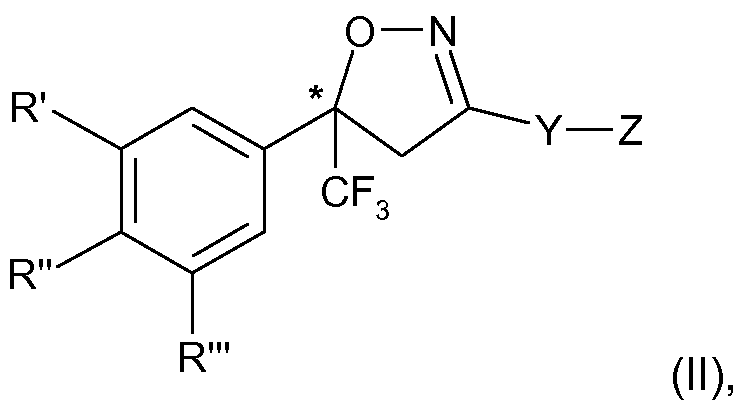
The present invention therefore relates to a process for the manufacture of an enantiomer, preferably of the S-enantiomer, of a compound of formula
(I)
or a veterinary acceptable salt thereof, wherein
R\ R" and R'" are each independently hydrogen, halogen, cyano, CrC2-alkyl, halo-Ci-C2- alkyl, CrC2-alkoxy or CrC2-haloalkoxy, subject to the proviso that at least one of R', R" and R'" is not hydrogen;
Y is phenylene, naphthylene, indanylene, 5- or 6-membered heteroarylene or 8-10- membered fused heterobicyclylene, which is each unsubstituted or substituted by Ci-C2- alkyl, Ci-C2-haloalkyl, halogen, nitro or cyano, and,
provided that Y is phenylene, naphthylene, heteroarylene or fused heterobicyclylene, Q is
(i) a 5- or 6-membered heteroaromatic ring comprising 1 to 3 same or different heteroatoms selected from the group consisting of O, S and N which is further unsubstituted or substituted; or is
(ii) a group -C(0)N(R1)-T, wherein is H, C C4-alkyl, C2-C4-alkylcarbonyl or C2-C4- alkoxycarbonyl and T is CrC6-alkyl which is unsubstituted or substituted by C3-C6-cycloalkyl, halogen, cyano, nitro, amino, hydroxy, CrC6-alkoxy, CrC6-haloalkoxy, CrC6-alkylthio, d- C6-haloalkylthio, CrC6-alkylsulfinyl, CrC6-haloalkylsulfinyl, CrC6-alkylsulfonyl, CrC6- haloalkylsulfonyl, carboxy, carbamoyl, Ci-C6-alkylcarbonylamino, CrC6-haloalkyl- carbonylamino, CrC6-alkoxycarbonyl, sulfonamido, N-mono- or N,N, di-CrC4-alkylsulfon- amido, C2-C6-alkanoyl, unsubstituted or in the alkyl portion by halogen, cyano, ethenyl or ethynyl substituted N-Ci-C6-alkylaminocarbonyl, or unsubstituted or halogen-, Ci-C2-alkyl-, CrC2-haloalkyl or cyano-substituted 4- to 6-membered heterocyclyl; or T is C3-C6-cycloalkyl or 4- to 6-membered heterocyclyl, which is each unsubstituted or substituted by halogen, Ci-C2-alkyl, Ci-C2-haloalkyl or cyano; or is
(iii) a radical -C(0)NH-CH=N-0-C C2-alkyl, a radical -C(0)N=CH-N-di-C C2-alkyl or a radical -C(0)N=C(NH2)-0-C C2-alkyl; or is
(iv) a group -CH(R3)-N(R4)-C(0)-T1, wherein R3 is H, C C6-alkyl, CrC6-haloalkyl, halogen or cyano, R4 is H; CrC4-alkyl C2-C4-alkylcarbonyl or C2-C4-alkoxycarbonyl, and Ti is
independently defined as T above; or,
provided that Y is indanylene, Q is a group -N(R4)-C(0)-T2, wherein T2 independently has the meaning of T above and R4 is as defined above;
comprising the steps of
wherein R', R"and R'" and Y are as defined above and Z is Q; or is halogen, amino, carboxy
(-COOH) or a salt thereof; into the enantiomers and collecting the desired enantiomer, preferably the S-enantiomer;
(B) if Z is other than Q, converting the desired enantiomer, preferably the S-enantiomer, of formula (II) from step (A) to the desired enantiomer, preferably to the S-enantiomer, of formula (I);
(C) racemizing the undesired enantiomer, preferably the R-enantiomer, of formula (II) from step (A) in a reaction medium comprising a base and an aprotic organic solvent at a temperature of from 30°C to 150°C, and,
(D) subjecting the racemate from step (C) to a further manufacturing cycle comprising step (A) and optionally (B).
According to one embodiment of the present invention Z is Q, that is, a racemic compound of formula (I) is prepared and subjected to process steps (A), (C) and (D) in a repetitive cycle.
According to a further embodiment of the invention, Z is halogen, amino, carboxy (-COOH) or a salt or derivative thereof, in particular carboxy or a salt thereof, that is, a racemic intermediate compound of formula (II) is prepared and subjected to process steps (A), (B), (C) and (D) in a repetitive cycle.
A preferred embodiment of the invention comprises a cyclic batch process, wherein one or more batches of a racemic compound of formula (II) are subjected to process steps (A) and optionally (B), the undesired enantiomer, in particular the R-enantiomer, obtained from the enantiomer separation of the one or more batches is subjected to racemization step (C), and the racemate from step (C) is subjected to a further enantiomer separation step (D). Conveniently, the undesired enantiomer, in particular the R-enantiomer, obtained from step
(A) of a first batch process is racemized according to step (C) above, and the racemate obtained is added to a subsequent batch of racemic compound of formula (II) to be separated into the enantiomers according to the present invention. No compound of formula (II) is thus lost, as the undesired enantiomer of an enantiomer separation step (A) is always recovered according to step (C), and the racemate from step (C) is phased in a subsequent enantiomer separation step (A).
Regarding the variables in formulae (I) and (II) the following preferences apply:
R', R" and R'" are each independently of the other preferably H, halogen, CF3 or cyano, and in particular H, CI, F or CF3, subject to the proviso that at least one of R', R" and R'" is not H. One preferred embodiment of the invention concerns compounds of formula (I), wherein R', R" and R'" are each independently of the other H, chlorine or fluorine, subject to the proviso that at least one of R', R" and R'" is not H. One especially preferred
embodiment concerns a compound of formula (I), wherein R' and R'" are each halogen, for example chlorine or fluorine, in particular chlorine, and R" is H, chlorine or fluorine, in particular H or chlorine and especially chlorine. Another preferred embodiment concerns a compound of formula (I), wherein one of R' and R'" is CF3 and the other one is halogen, for example chlorine or fluorine, in particular chlorine, and R" is H.
(Hi),
wherein R2 is H, CrC2-alkyl, CrC2-haloalkyl, halogen, nitro or cyano, preferably H, methyl, chlorine, nitro, cyano or CF3, more preferably H, methyl, chlorine CF3 or cyano, in particular methyl, chlorine CF3 or cyano, and especially methyl.
A preferred naphthylene radical Y is of formula
wherein R2' is H, methyl, chlorine, nitro, cyano or CF3, in particular H.
A preferred heteroaryle
wherein for R2 each the above given meanings and preferences apply, and A is O or S, in particular S.
A particularly preferred embodiment of the heteroarylene radical Y is a radical of formula (llld) above, wherein A is S, Q or Z is located in the 2-position, and R2 is methyl and is located in the 3-position.
Further embodiments of the heteroarylene radical Y relate to
- a radical of formula (llld) wherein A is O, Q or Z is located in the 2-position, and R2 is located in the 3-position;
- a compound of formula (llld) wherein A is S, Q or Z is located in the 3-position, and R2 is located in the 2-position; or
- a compound of formula (llld) wherein A is O, Q or Z is located in the 3-position, and R2 is located in the 2-position.
Embodiments of a fused heterobicyclylene emcompass, for example, a radical of formula
wherein B-i is CH, C(CH3) or N, one of B2 and B3 is CH and the other one is CH or N.
A suitable heterocyclic ring Q (embodiment (i)) is, for example, a 5- or 6-membered heteroaromatic ring having from 1 to 4, preferably from 1 to 3 same or different heteroatoms selected from the group consisting of N, O and S, which is further unsubstituted or substituted by one or more substituents selected from the group consisting of halogen, cyano, nitro, CrC4-alkyl, Ci-C4-haloalkyl, hydroxy, Ci-C4-alkoxy, Ci-C4-haloalkoxy, Ci-C4- alkylthio, Ci-C4-haloalkylthio, CrC4-alkylsulfinyl, CrC4-haloalkylsulfinyl, CrC4-alkylsulfonyl, CrC4-haloalkylsulfonyl, -COOH, Ci-C4-alkoxycarbonyl, sulfonamido, C2-C3-alkanoyl, aminocarbonyl, N-mono- or N,N-di-Ci-C4-alkylaminocarbonyl and C(S)NH2. The
heteroaromatic ring Q is preferably unsubstituted or substituted by 1 to 3, in particular 1 or 2, same or different substituents selected from the group consisting of halogen, cyano, nitro, CrC2-alkyl, CrC2-haloalkyl, Ci-C2-alkoxy, Ci-C2-haloalkoxy, Ci-C2-haloalkylthio, d- C4-alkoxycarbonyl, C2-C3-alkanoyl, aminocarbonyl, N-mono- or N,N-di-CrC3- alkylaminocarbonyl and C(S)NH2. The heteroaromatic ring Q is most preferably
unsubstituted or substituted by 1 substituent selected from the group consisting of halogen, cyano, CrC2-alkoxycarbonyl, aminocarbonyl, N-mono- or N,N-di-Ci-C2-alkylaminocarbonyl and C(S)NH2.
Examples of a 5- or 6-membered heteroaromatic rings optionally substituted with from one or more substituents include the rings Q-1 through Q-60 illustrated in Exhibit 1 wherein R is any substituent as defined before including the preferences given, and r is an integer from 0 to 4, limited by the number of available positions on each Q group. As Q-28,- Q-29, Q-35, Q-36, Q-37, Q-38, Q-39, Q-40, Q-41 and Q-42 have only one available position, for these Q groups r is limited to the integers 0 or 1 , and r being 0 means that the Q group is
unsubstituted and a hydrogen is present at the position indicated by (R)r.
Q-5 Q-6 Q-7 Q-8 Q-9
-7-
Q-60
A preferred heterocyclic ring Q is of formula
wherein r is an integer from 0 to 3 and R is independently selected from the group given before for the heteroaromatic ring including the preferences. Q is particularly preferred the unsubstituted radical Q-14, Q-24, Q-34, Q-43 or Q-47, wherein r is 0 in each case. Q is especially preferred a radical Q-14, Q-34 or Q-47, wherein r is 0.
If Q is a group -C(0)N(Ri)-T (embodiment (ii)), is preferably H, methyl, ethyl or acetyl and in particular H.
T as alkyl is preferably CrC4-alkyl, more preferably CrC2-alkyl and particularly preferably Ci-alkyl, which is each unsubstituted or substituted as defined above.
The alkyl radical T is preferably unsubstituted or substituted by halogen; CrC4-alkoxy; C C4-alkoxycarbonyl; N-Ci-C6-alkylaminocarbonyl which is unsubstituted or substituted in the alkyl portion by halogen, cyano, ethenyl or ethynyl; or 5- to 6-membered heterocyclyl which is in turn unsubstituted or substituted by halogen-, CrC2-alkyl- or CrC2-haloalkyl.
A preferred N-alkylaminocarbonyl substituent of the alkyl radical T is N-Ci-C2- alkylaminocarbonyl, which is unsubstituted or further substituted in the alkyl moiety by halogen, cyano, ethenyl or ethynyl. Especially preferred N-alkylaminocarbonyl substituents of the alkyl radical T are N-ethylaminocarbonyl or a radical -C(Q)NH-CH2CF3,
-C(0)NH-CH2CN, -C(0)NH-CH2CH=CH2 or -C(0)NH-CH2C≡CH.
T as N-alkylaminocarbonyl-substituted alkyl is preferably N-ethylaminocarbonylmethyl, or a radical -CH2-C(0)NH-CH2CF3, -CH2-C(0)NH-CH2CN or-CH2-C(0)NH-CH2C≡CH.
If T is heterocyclyl-substituted alkyl, preferred meanings of heterocyclyl include pyridyl, pyrimidinyl, thiazolyl, oxazolyl, tetrahydrofuranyl, thietanyl, thietanyl-1 ,1 -dioxide or oxetanyl. Preferred heterocyclyl-substituted alkyl radicals T are in particular 2-pyridylmethyl, 2- tetrahydrofuranylmethyl.
T as heterocyclyl preferably denotes as 4- to 6-membered ring comprising 1 to 3 same or different heteroatoms selected from the group consisting of O, S, S(O), S(02) and N, which is each unsubstituted or substituted by halogen, Ci-C2-alkyl or Ci-C2-haloalkyl.
If T is 4- to 6-membered heterocyclyl, preferred meanings of heterocyclyl include pyridyl, pyrimidyl, thiazolyl, oxazolyl, tetrahydrofuranyl, thietanyl, thietanyl-1 -oxide, thietanyl-1 ,1 - dioxide or oxetanyl and in particular 2- 3- or 4- pyridyl, 3- 4- or 5- pyrimidyl, 2- or 3- tetrahydrofuranyl, thietan-3-yl or oxetan-3-yl and even more preferred 5-CI-pyrimid-3-yl, 3- tetrahydrofuranyl, thietan-3-yl, thietan-3-yl-1 -oxide, thietan-3-yl-1 ,1 -dioxide or oxetan-3-yl.
If Q is a group -C(0)N(R1)-T, is preferably H, methyl, ethyl or acetyl and T is Ci-C2-alkyl; CrC2-haloalkyl; Ci-C2-alkoxycarbonyl-CrC2-alkyl; Ci-C2-alkyl which is substituted by pyridyl, pyrimidinyl, thiazolyl, oxazolyl or tetrahydrofuranyl; N-Ci-C2-alkylaminocarbonyl-Cr C2-alkyl which is unsubstituted or substituted in the N-alkyl moiety by halogen, cyano, ethenyl or ethynyl; or is pyridyl; pyrimidyl; thiazolyl; oxazolyl; tetrahydrofuranyl; thietanyl; thietanyl-1 -oxide; thietanyl-1 ,1 -dioxide; or oxetanyl.
If Q is a group -C(0)N(R1)-T, is most preferably H, methyl or ethyl, and T is Ci-C2-alkyl; CrC2-haloalkyl; methyl which is substituted by pyridyl, pyrimidinyl, thiazolyl, oxazolyl or tetrahydrofuranyl; methyl which is substituted by N-Ci-C2-alkylaminocarbonyl or by N-Ci-C2- alkylaminocarbonyl substituted in the alkyl moiety by halogen, cyano, ethenyl or ethynyl; or is pyridyl; pyrimidyl; tetrahydrofuranyl; thietanyl; thietanyl-1 -oxide; thietanyl-1 ,1 -dioxide; or oxetanyl.
If Q is a group -C(0)N(R1)-T, is particularly preferably H, and T is CrC2-alkyl; a radical -CH2CF3; N-ethylaminocarbonylmethyl; a radical -CH2-C(0)NH-CH2CF3!
-CH2-C(0)NH-CH2CN or -CH2-C(0)NH-CH2C≡CH; 2- pyridylmethyl; 5-CI-pyrimid-3-yl; 3- tetrahydrofuranyl; thietan-3-yl; thietan-3-yl-1 -oxide; thietan-3-yl-1 ,1 -dioxide; or oxetan-3-yl.
Preferred radicals Q of embodiment (iii) are a radical -C(0)NH-CH=N-0-CH3, a radical -C(0)N=CH-N(CH3)2 or a radical -C(0)N=C(NH2)-0-CH3.
If Q is a group -CH(R3)-N(R4)-C(0)-T1 (embodiment (iv)), R3 is preferably H or C C2-alkyl or cyano, more preferably H or methyl, and in particular H. R4 is preferably H or CrC2-alkyl, in particular H.
R4 is preferably H or CrC2-alkyl, in particular H.
T-i as optionally substituted alkyl is preferably straight-chain or branched CrC4-alkyl, which is each unsubstituted or substituted by C3-C6-cycloalkyl, halogen, cyano, CrC4-alkoxy, d- C2-haloalkoxy, CrC4-alkylthio, CrC2-haloalkylthio, CrC4-alkylsulfinyl, CrC4-haloalkylsulfinyl, CrC4-alkylsulfonyl, CrC4-haloalkylsulfonyl, Ci-C2-alkylcarbonylamino, CrC2-haloalkyl- carbonylamino or 4- to 6-membered heterocyclyl. Especially preferred alkyl radicals ΤΊ are straight-chain or branched CrC4-alkyl or CrC4-alkyl which is substituted by cyclopropyl, halogen, cyano, CrC2-alkoxy, CrC2-haloalkoxy, CrC2-alkylthio, CrC2-alkylsulfinyl, CrC2- alkylsulfonyl, Ci-C2-haloalkylcarbonylamino, pyridyl, pyrimidyl, thiazolyl, oxazolyl, thietanyl, oxetanyl, dioxolanyl, methyldioxolanyl, dioxanyl or tetrahydrofuryl.
T-i as alkyl is especially preferred straight-chain or branched CrC4-alkyl, Ci-C3-haloalkyl, cyclopropylmethyl, cyano-CrC2-alkyl, Ci-C2-alkoxy-Ci-C2-alkyl, Ci-C2-alkylthio-Ci-C2-alkyl, Ci-C2-alkylsulfinyl-Ci-C2-alkyl, Ci-C2-alkylsulfonyl-Ci-C2-alkyl, or methyl which is substituted by 1 ,3-dioxolan-2-yl, 2-methyl-1 ,3-dioxolan-2-yl or tetrahydrofuran-2- or -3-yl.
Particularly preferred alkyl radicals ΤΊ are straight-chain or branched CrC4-alkyl; CrC2- alkyl which is substituted by halogen, cyano, CrC2-alkoxy, CrC2-alkylthio or CrC2-alkylsulfonyl; or 2-methyl-1 ,3-dioxolan-2-yl-methyl.
If Ti is C3-C6-cycloalkyl, said cycloalkyl is preferably cyclopropyl, cyclobutyl, cyclopentyl or cyclohexyl, in particular cyclopropyl.
If T-i is 4- to 6-membered heterocyclyl, said heterocyclyl is, for example, a 4-6-membered heteroaromatic ring, preferably a thienyl, furyl, oxazolyl, thiazolyl, pyridyl or pyrimidinyl radical, which is each unsubstituted or substituted by CrC2-alkyl, CrC2-haloalkyl or Ci-C4- alkoxycarbonyl. Especially preferred heteroaromatic radicals ΤΊ are 2-, 3- or 4-pyridyl, 2- or 4-pyrimidinyl, 2-thiazolyl, 2-furyl or 2-thienyl.
A further preferred heterocyclic radical ΤΊ is, for example, a 4- to 6-membered
heteroaliphatic ring selected from the group of thietanyl, for example thietan-3-yl, oxothietanyl, for example thietan-3-yl-1 -oxide or thiethan-3-yl-1 ,1 -dioxide, oxetanyl, for example oxetan-3-yl, azetidinyl, pyrrolidinyl, tetrahydrofuranyl, tetrahydrothiophenyl, piperidinyl, piperazinyl, morpholinyl, tetrahydropyranyl and thianyl which is each
unsubstituted or substituted by CrC2-alkyl, CrC2-haloalkyl or Ci-C4-alkoxycarbonyl.
Especially preferred heteroaliphatic ring radicals ΤΊ include pyrrolidinyl, tetrahydrofuranyl, tetrahydrothiophenyl, piperidinyl, piperazinyl, morpholinyl, thietanyl, thietanyl oxide or thietanyl dioxide which are each unsubstituted or substituted by CrC2-alkyl, Ci-C2-haloalkyl or Ci-C4-alkoxycarbonyl, and in particular pyrrolidine-1 -yl, tetrahydrofuran-2-yl,
tetrahydrofuran-3-yl, piperidine-1 -yl, morpholine-4-yl or thietan-3-yl, thietan-3-yl-1 -oxide or thiethan-3-yl-1 ,1 -dioxide.
Q as a group -CH(R3)-N(R4)-C(0)-Ti is most preferably a radical -CH2-NH-C(0)-C C2-alkyl, -CH2-NH-C(0)-cyclopropyl, -CH2-N H-C(0)-(CH2)1-2-0-Ci-C2-alkyl,
-CH2-N H-C(0)-(CH2)1-2-S-Ci-C2-alkyl or -CH2-N H-C(0)-(CH2)1-2-S(0)2-Ci-C2-alkyl.
If Q is a group -N(R4)-C(0)-T2, for R4 each the above given meanings and preferences apply independently; in addition, for T2 each the meanings and preferences given above for Ti apply.
Provided that Y is phenylene, naphthylene heteroarylene or fused heterobicyclylene, particular preferred meanings of Q are a radical
2) -C 0)NH-CH2CF3
-C(0)-N- (q6)
-C(0)-N-
S=0
-C(0)-N-
-O
(q9)
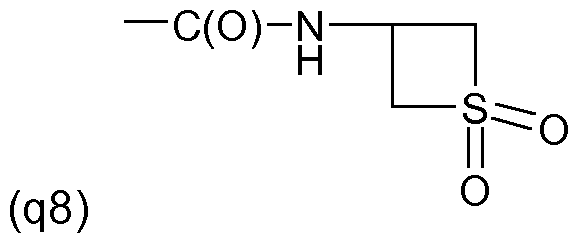
(q10) -C(0)NH-CH2-C(0)NH-CH2CF3 (q1 1 ) -C(0)NH-CH2-C(0)NH-CH2CN (q12) -C(0)NH-CH2-C(0)NH-CH2C≡CH (q13) -C(0)NH-C=N-0-CH3
(q14) -C(0)N=C-N(CH3)2
(q15) -C(0)N=C(NH2)-0-CH3
(q16) -CH2-NH-C(0)-CrC3-alkyl (q17) -CH2-NH-C(0)-cyclopropyl (q18) -CH2-NH-C(0)-cyclobutyl (q19) -CH2-NH-C(0)-CrC2-haloalkyl
(q20) -CH2-NH-C(0)-(CH2)1-2-S-Ci-C2-alkyl
(q21 ) -CH2-NH-C(0)- )-(CH2)1-2-S(0)2-Ci-C2-alkyl
(q22) -CH2-NH-C(0)-(CH2)1-2-0-Ci-C2-alkyl
23) .-CH2-NH-C(0)-(CH2)1-2-CN
Provided that Y is indanyl, particular preferred meanings of Q
(q26) -NH-C(0)-Ci-C3-alkyl,
(q27) -NH-C(0)-cyclopropyl,
(q28) -NH-C(0)-cyclobutyl,
(q29) -NH-C(0)-CrC2-haloalkyl,
(q30) -NH-C(O)-(CH2)1-2-S-Ci-C2-alkyl,
(q31 ) -NH-C(0)-(CH2)1-2-S(0)2-Ci-C2-alkyl,
(q32) -NH-C(0)-Ci-C3-alkyl-0-Ci-C2-alkyl,
33) -NH-C(0)-(CH2)1-2-CN,
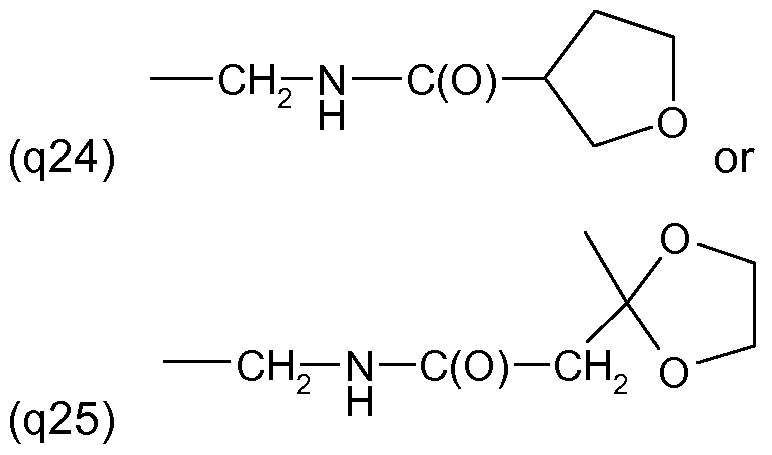
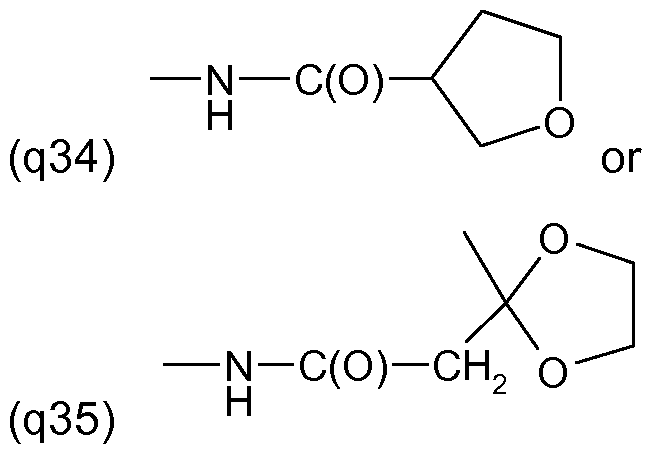
One embodiment of the invention relates to the manufacture of the S-enantiomer of a compound of the formula (I) above wherein R' and R'" are each independently of the other halogen, for example chlorine or fluorine, in particular chlorine, R" is H or halogen, preferably H or chlorine and in particular chlorine, Y is a radical of formula (llld) above, wherein A is S, Q is a radical (q2) to (q25) as mentioned above in the 2-position, and R2 is methyl and is located in the 3-position.
A further embodiment of the present invention comprises a process for the manufacture of the S-enantiomer
or a veterinary acceptable salt thereof, wherein for R', R", R'", Y and T each the above given meanings and preferences apply; comprising the steps of
(A) separating a racemic co
(II).
wherein R', R" and R'" and Y are as defined and Z carboxy (-COOH) or a salt thereof; into the enantiomers and collecting the S-enantiomer;
followed by steps (B), (C) and (D) as described above, and including the preferences given below.
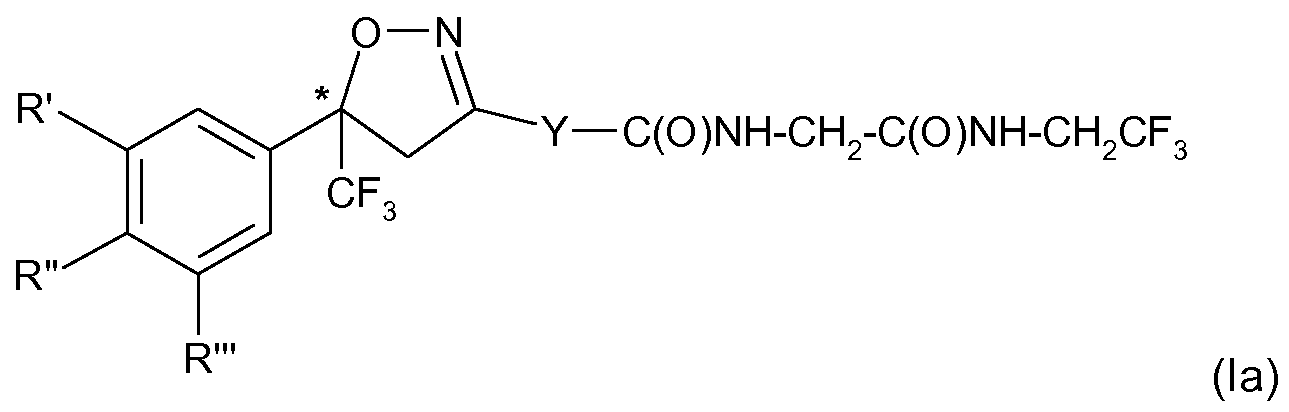
Specific examples of preferred compounds of formula (I) are the S-enantiomers of the compounds:
5-[5-(3,5-dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-3-methyl-thiophene-2- carboxylic acid [(2,2,2-trifluoro-ethylcarbamoyl)-methyl]-amide; or
5-[5-(3,4,5-trichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-3-methyl-thiophene- 2-carboxylic acid [(2,2,2-trifluoro-ethylcarbamoyl)-methyl]-amide;
4-[5-(3,5-dichlorophenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-2-methyl-N{-2-oxo-2- [(2,2,2-trifluoroethyl)amino]ethyl}-benzamide;
4-[5-[3-chloro-5-(trifluoromethyl)phenyl]-4,5-dihydro-5-(trifluoromethyl)-3-isoxazolyl]-N-[2- oxo-2-[(2,2,2-trifluoroethyl)amino]ethyl]-1 -naphthalenecarboxamide;
4-[5-[3,5-dichlorophenyl]-4,5-dihydro-5-(trifluoromethyl)-3-isoxazolyl]-N-[2-oxo-2-[(2,2,2- trifluoroethyl)amino]ethyl]-1 -naphthalenecarboxamide;
4-[5-(3,5-dichlorophenyl)-5-trifluorom
yl)-benzamide;
4-[5-(3,5-dichlorophenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-2-methyl-N(thie 1 -oxo)-benzamide;
4-[5-(3,5-dichlorophenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-2-methyl-N(thie 1 ,1 -dioxo)-benzamide;
4- [5-(3,5-dichlorophenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-2-cyano-1 -(1 ,2,4-triazo
1 - yl)-phenyl.
Specific examples of preferred racemic compounds of formula (II), wherein Z is different from Q, are
5- [5-(3,4,5-trichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-3-methyl-thiophene-
2- carboxylic acid;
5-[5-(3,5-dichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-3-methyl-thiophene-2- carboxylic acid;
4-[5-(3,5-dichlorophenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-2-methyl-benzoic acid; 4-[5-[3-chloro-5-(trifluoromethyl)phenyl]-4,5-dihydro-5-(trifluoromethyl)-3-isoxazolyl]-1 - naphthalenecarboxylic acid;
4-[5-[3,5-dichlorophenyl]-4,5-dihydro-5-(trifluoromethyl)-3-isoxazolyl]-1 -naphthalene- carboxylic acid.
Veterinary acceptable salts of the compounds of formula (I) as well as salts of the compounds of formula (II) include acid-addition salts with inorganic or organic acids; just by way of example, the following acids may be mentioned: hydrobromic, hydrochloric, nitric, phosphoric, sulfuric, acetic, butyric, fumaric, lactic, maleic, malonic, oxalic, propionic, salicylic, tartaric, 4-toluenesulfonic or valeric acid.
Enantiomer separation of a compound of formula (II) according to step (A) may be performed in a manner known per se, for example by simple recrystallization, kinetic resolution, by means of biotechnological methods (whole cells, enzymes, etc.), by chromatographic methods, or by methods that are based on the difference of
physicochemical properties of diastereomers prepared by reversible addition of an optically pure reagent.
For example, a compound of formula (II) may be separated into its enantiomers by chiral column chromatography, in particular by chromatography on chiral adsorbents using a Simulated Moving Bed (SMB) system. To this end, a liquid mobile phase carrying the racemic compound of formula (II) to be separated is contacted with a chiral stationary phase in a system that is constituted of a number of chromatographic columns in a circular flow arrangement, separated by ports where inlet and outlet streams can be fed or collected. A suitable chiral stationary phase is, for example, composed of a chiral polysaccharide (for example Chiralpak® AS-V, AD). The liquid mobile phase typically comprises one or more alcohols, for example methanol and/or ethanol , optionally in admixture with an organic acid, for example, formic acid. A suitable SMB system is described, for example, in
WO2006/050887.
According to a further embodiment, a racemic compound of formula (II) which is an acid or amine, is separated into the enantiomers by diastereomeric recrystallization. To this end, an optically pure reagent, which is an amine or acid, is reacted with a racemic compound of formula (II), wherein Z is amino, carboxy or a salt thereof, thereby forming pairs of diastereomeric salts, which can be separated by conventional techniques in physical chemistry, i.e. by crystallization.
In case of a racemic compound of formula (II) wherein Z is amino or a salt thereof, a typical optical pure reagent is an organic acid, for example, (+)-D-di-0-benzoyl-tartaric acid, (-)-L- di-O-benzoyltartaric acid, (-)-di-0,0'-p-tolyl-L-tartaric acid, (+)- di-0,0'-p-tolyi-D-tartaric acid, R(+)-malic acid, S-(-)-malic acid, (+)-camphanic acid, (-)-camphanic acid, R(-)-1 ,V- binaphthalene-2,2'-diyl hydrogen phosphate, S(+)-1 ,1 '-binaphthalene-2,2'-diyl hydrogen phosphate, (+)-camphoric acid, (-)-camphoric acid, S(+)-2-phenylpropionic acid, R(-)-2- phenylpropionic acid, D(-)- mandelic acid, L(+)-mandelic, acid, D-tartaric acid, L-tartaric acid, or a mixture thereof.
In case of a racemic compound of formula (II) which is an acid, a typical optical pure reagent is an optical pure amine, for example (R)-(+)-1 -(4-methylphenyl)ethylamine, (S )-(-)- 1 -(4-methylphenyl)ethylamine, (R)-(+)-a-methylbenzylamin, (S)-(-)-a-methylbenzylamin, (R)- (-)-aminoindane, (S)-(+)-aminoindane, (R)-(+)-N,a-dimethylbenzylamine, (S)-(-)-N,a- dimethylbenzylamine, (R)-(+)-N,N-dimethyl-1 -phenyletyhlamine, (S)-(-)-N,N-dimethyl-1 -
phenyletyhlamine, (R)-(-)-1 -cyclohexylethylamine, (S)-(+)-1 -cyclohexylethylamine, (R)-(-)- 1 ,2,3,4-tetrahydro-l -naphthylamine, (S )-(+)- 1 ,2,3,4-tetrahydro-l -naphthylamine, (-)-bis[(S)- 1 -phenylethyl]amine, (+)-bis[(R)-1 -phenylethyl]amine. Upon the reaction of the racemic compound with the optical pure enantiomer, diasteriometric salts are formed which may be separated, for example, by crystallization.
According to a preferred embodiment, a racemic compound of formula (II), wherein Z is carboxy or a salt thereof, is separated into its enantiomers by reaction with an optical active amine, in particular (R)-(+)-1 -(4-methylphenyl)ethylamine.
The formation of the diastereomeric salt may be performed in a polar solvent or a solvent mixture comprising at least one polar solvent. Suitable solvents are for example, halogenated hydrocarbons, for example chlorobenzene, dichlorobenzene, bromobenzene, dichloromethane, trichloromethane, tetrachloromethane, dichloroethane, trichloroethene or tetrachloroethene; alcohols, such as methanol, ethanol, propanol, isopropanol, butanol, isobutanol or tert. butanol; ethers, such as diethyl ether, dipropyl ether, diisopropyl ether, dibutyl ether, tert. -butyl methyl ether, ethylene glycol monomethyl ether, ethylene glycol monoethyl ether, ethylene glycol dimethyl ether, dimethoxydiethyl ether, tetrahydrofuran or dioxane; ketones, such as acetone, methyl ethyl ketone or methyl isobutyl ketone; amides, such as N,N-dimethylformamide, Ν,Ν-diethylformamide, N,N-dimethylacetamide, N- methylpyrrolidone or hexamethylphosphoramide; esters, such as methyl acetate, ethylacetate; nitriles, such as acetonitrile or propionitrile; sulfoxides, such as dimethyl sulfoxide; or water. Preferred solvents for the formation and crystallization of the diastereomeric salts according to the invention are alcohols, in particular ethanol, isopropanol, n-, sec. or tert. -butanol, nitriles, in particular acetonitrile, ethers, in particular tert. - butyl methylether, esters, in particular methylacetate, or mixtures therof, or a mixture one of or more of the above mentioned solvents with water. A particularly preferred reaction medium for the formation of the diastereomeric salts is a mixture of water, a C2-C4-alcanol and a dipolar aprotic solvent such as acetonitrile or the like.
The formation of the diastereomeric salt may be performed at a temperatu re ranging from about 1 0° C to the reflux point of the solvent or solvent mixture. Salt formation is carried out preferably at a temperature ranging from room temperature to about 100°C. For example, the mixture of the racemate to be separated, the optical active reagent and the
solvent(s) is heated to an elevated temperature, for example to 30 to 100°C or preferably to 50 to 80°C, kept at this temperature for some time and then cooled, for example, to room temperature in order to affect crystallization of one of the diastereomeric salts. The reaction time is not critical; in general, a time period of 0.5 minutes to 6 hours, preferably 1 to 4 hours, and in particular from 1 .5 to 3 hours at an elevated temperature is sufficient for the formation of the diastereomeric salts. Upon cooling of the reaction mixture, one of the diastereomeric salts, preferably the one covering the S-enantiomer, usually crystallizes and may be isolated by filtration.
Once the crystallized diastereomeric salt has been isolated, it may be purified to the desired diastereomeric purity, for example by recrystallization in a suitable solvent or solvent mixture. The purified diastereomeric salt is then dissociated in basic or acidic medium in a suitable solvent or solvent mixture, and the desired (S)-enantiomer is recovered from the racemic mixture of the compound of formula (II).
The desired S-enantiomer of the compound of formula (II) is collected and optionally further purified by processes known in the art, for example by recrystallization. The optical purity of the S-enantiomer as collected is for example≥90%, preferably≥95% and in particular ≥98%.
The S-enantiomer of formula (II), wherein Z is different from Q, is converted to a S- enantiomer of formula (I) according to step (B) in a manner known per se, for example from WO2010/070068 or WO201 1/157748.
For example, an S-enantiomer of formula
wherein R', R", R'" and Yare as defined above and Hal is halogen, for example, fluoro, chloro or bromo, may be reacted with a compound of formula
H - Q (IVa),
wherein Q is a 5- or 6-membered heteroaromatic ring, in the presence of a catalyst, to yield the respective S-enantiomer of formula (I), wherein Q is a heteroaromatic radical. Typical catalysts are, for example copper iodide, a palladium catalyst or an inorganic base such as sodium or potassium carbonate.
Moreover, an S-enantiomer of formula
or a salt thereof, wherein R', R", R'" and Y are as defined above, may be first converted to the respective acid halide which is reacted with a compound of formula
wherein R-i and T are as defined above, optionally in the presence of a base, to yield the respective S-enantiomer of formula (I), wherein Q is a group -C(0)N(R1)-T.
S-enantiomers of the formula (I), wherein Q is a radical -C(0)NH-CH=N-0-Ci-C2-alkyl, -C(0)N=CH-N-di-C C2-alkyl or -C(0)N=C(NH2)-0-Ci-C2-alkyl, may be obtained from a compound of formula (l ib) above via conversion to the respective amide.
An S-enantiomer of formula (l ib) above may also be first converted to the respective aldehyde, optionally via a respective ester, which is subjected to a triethylsilane-promoted reductive amination with a compound of formula
wherein R4 and ΤΊ is as defined above, to yield the S-enantiomer of formula (I), wherein Q is a group -CH(R3)-N(R4)-C(0)-T1.
or a salt thereof, wherein R', R", R'" and Y are as defined above, may be reacted with a compound of formula
Hal - C(0) - T2 (IVd),
wherein T2 and Hal is as defined above, optionally in the presence of a base, to yield the respective S-enantiomer of formula (I), wherein Q is a group -NR4-C(0)T2.
Suitable organic solvents in racemization step (C) are, for example aromatic, aliphatic (cyclic and alicyclic) and halogenated hydrocarbons, such as benzene, toluene, xylene, mesitylene, tetralin, chlorobenzene, dichlorobenzene, bromobenzene, petroleum ether, hexane, cyclohexane, dichloromethane, trichloromethane, tetrachloromethane, dichloro- ethane, trichloroethene or tetrachloroethene; alcohols, such as methanol, ethanol, propanol, isopropanol, butanol, isobutanol or tert. butanol; ethers, such as diethyl ether, dipropyl ether, diisopropyl ether, dibutyl ether, tert. -butyl methyl ether, ethylene glycol monomethyl ether, ethylene glycol monoethyl ether, ethylene glycol dimethyl ether, dimethoxydiethyl ether, tetrahydrofuran or dioxane; esters, such as methyl acetate, ethyl acetate or tert. -butyl acetate; ketones, such as acetone, methyl ethyl ketone or methyl isobutyl ketone; amides, such as N,N-dimethylformamide, Ν,Ν-diethylformamide, N,N-dimethylacetamide, N- methylpyrrolidone or hexamethylphosphoramide; nitriles, such as acetonitrile or propionitrile; and sulfoxides, such as dimethyl sulfoxide (DMSO).
Preferred groups of organic solvents applied in step (C) according to the process of the invention are aromatic hydrocarbons or halogenated aromatic hydrocarbons, preferably benzene, toluene, xylene, mesitylene, or tetralin, especially toluene or o-, m- oder p-xylene, and in particular toluene; alcohols, for example ethanol, nitriles, for example acetonitrile; or DMSO. The amount of organic solvent being present in step (C) may be, for example, from 30 to 90% (w/w), in particular from 50 to 90% (w/w) based on the entire reaction mixture.
In addition, the reaction medium of step (C) may comprise water, in particular, if an alkali metal or alkaline earth metal hydroxide is used as the base. The amount of water being present in the reaction mixture, if present, may be, for example, from 1 to 20% (w/w), preferably from 1 .5 to 10% (w/w), based on the entire reaction mixture.
Suitable bases for racemization step (C) are, for example e.g. alkali metal or alkaline earth metal hydroxides, for example lithium, sodium or potassium hydroxide; ammonium hydroxide; alkali metal or alkaline earth metal hydrides, for example sodium hydride, calcium hydride; alkali metal or alkaline earth metal alkanolates, for example sodium methanolate sodium ethanolate, sodium butanolate, potassium tert.-butanolate; alkali metal carbonates, for example potassium carbonate or cesium carbonate; amines, for example triethylamine, N,N-diisopropylethylamine; carbocyclic amines, for example 2,2,6,6- tetramethylpiperidine, 1 ,5-diazabicyclo[5.4.0]undec-5-ene (DBU) or 1 ,5- diazabicyclo[5.4.0]undec-7-ene (DBA); lithium diisopropylamide (LDA); lithium N- isopropylcyclohexylamide (LICA); lithium or potassium hexamethyldisilazide (LHMDS, KHMDS); lithium 2,2,6, 6-tetramethylpiperidide (LTMP); guanidine; n-, sec- or tert- butyllithium; phosphazenes, for example tert.-butylimino-tris(dimethylamino)phosphorane.
A preferred base according to the present invention is an alkali metal hydroxide, in particular sodium hydroxide. The base is in general present in molar excess relative to the amount of enantiomer to be racemized.
A preferred reaction medium of step (C) comprises an aromatic hydrocarbon, in particular toluene, water and an alkali metal hydroxide, in particular sodium hydroxide.
In case of a reaction medium comprising an organic solvent with low water solubility and water it has proven as valuable to add a phase transfer catalyst to the reaction mixture. Suitable phase transfer catalysts are, for example, quaternary ammonium or phosphonium salts, preferably quaternary ammonium salts, in particular N-methyl-N,N-dioctyloctan-1 - ammonium chloride, benzyl trimethylammonium chloride, tetrabutylammonium bromide, or tributyl methylammonium chloride. The amount of phase transfer catalyst, if present in step (C) may be, for example, from 0.1 to 10% (w/w), preferably from 0.5 to 5% (w/w), and in particular from 1 to 4% (w/w) based on the entire reaction mixture.
A further preferred reaction medium of step (C) according to the invention comprises an aromatic hydrocarbon, in particular toluene, water, a phase transfer catalyst, in particular a quaternary ammonium salt such as tributyl methylammonium chloride and an alkali metal hydroxide, in particular sodium hydroxide.
A suitable reaction mixture according to step (C) comprises
1 to 20 % (w/w) R-enantiomer of formula (II) to be racemized
1 to 20% (w/w) water
1 to 20% (w/w) base
0.1 to 10% (w/w) phase transfer catalyst, and
ad 100% (w/w) organic solvent.
A preferred reaction mixture according to step (C) comprises
1 to 20 % (w/w) R-enantiomer of formula (II) to be racemized, wherein the above
meanings and preferences apply;
1 to 10% (w/w) water
1 to 10% (w/w) alkali metal hydroxide
0.5 to 5% (w/w) quaternary ammonium salt as phase transfer catalyst, and
ad 100% (w/w) aromatic hydrocarbon as organic solvent.
A particularly preferred reaction mixture according to step (C) comprises
1 to 20 % (w/w) R-5-[5-(3,4,5-trichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3-yl]-
3- methyl-thiophene-2-carboxylic acid;
1 to 10% (w/w) water;
1 to 10% (w/w) alkali metal hydroxide selected from sodium and potassium hydroxide; 0.5 to 5% (w/w) phase transfer catalyst selected from tetrabutyl ammonium bromide and tributyl methylammonium chloride; and
ad 100% (w/w) aromatic hydrocarbon selected from toluene or o-, m- oder p-xylene.
The racemization reaction according to step (C) advantageously takes place in a temperature range of about 50°C to about 120°C , preferably from ca. 50°C to ca. 100°C, and in particular from 60°C to 90°C.
In a preferred embodiment, an R-enantiomer of formula (II) is racemized in a reaction medium comprising an aromatic hydrocarbon, preferably toluene, water, a phase transfer catalyst and a base which is an alkali metal hydroxide at a temperature from about 50°C to about 100°C, preferably from 60 to 90°C.
The reaction time may vary within wide limits, for example between 1 and 100 hours, preferably between 2 and 72 hours, more preferably between 2 and 24 hours and in particular between 3 and 12 hours. A particularly preferred reaction time is from 4 to 8 hours.
A preferred embodiment of the present invention comprises a process for the manufacture of the S-enantio
or a veterinary acceptable salt thereof, wherein R', R" and R'" are each independently of the other preferably H, CI, F or CF3, subject to the proviso that at least one of R', R" and R' is not H; Y is a radical
and for T the above given meanings and preferences apply; comprising the steps of (A) separating a racemic co
(II).
wherein R', R" and R'" and Y are as defined above and Z carboxy (-COOH) or a salt thereof; into the enantiomers and collecting the S-enantiomer;
(B) converting the S-enantiomer of formula (II) from step (A) to the S-enantiomer of formula
(I);
(C) racemizing the R-enantiomer of formula (II) from step (A) in a reaction medium comprising a base and an aprotic organic solvent at a temperature of from 50 to 120°C, and,
(D) subjecting the racemate from step (C) to a further manufacturing cycle comprising steps (A) and (B).
An especially preferred embodiment of the present invention comprises a process for the manufacture of the S-enantiomer of a compound of formula
or a veterinary acceptable salt thereof, wherein R' and R'" are each independently CI or CF3, R" is H or CI; and Y is a radical (ΙΙΓ), (Ilia') or (llld') as shown above;
comprising the steps of
(A) separating a racemic compound of formula
wherein R', R" and R'" and Y are as defined above and Z is carboxy (-COOH) or a salt thereof; into the enantiomers, preferably by diastereomeric recrystallization, and collecting the S-enantiomer;
(B) converting the S-enantiomer of formula (II) from step (A) to the S-enantiomer of formula
(i);
(C) racemizing the R-enantiomer of formula (II) from step (A) in a reaction medium comprising an aromatic hydrocarbon, preferably toluene, water, a phase transfer catalyst and a base which is an alkali metal hydroxide at a temperature from about 50°C to about 100°C, preferably from 60 to 90°C; and
(D) subjecting the racemate from step (C) to a further manufacturing cycle comprising steps (A) and (B).
While the above description concerns the collection of the S-enantiomer of a compound of formula (I), racemization of the R-enantiomer and the use of the resulting racemate in a further enantiomer separation step, the reverse process, that is collection of the R- enantiomer of a compound of formula (I), racemization of the S-enantiomer and use of the resulting racemate in a further enantiomer separation step, may be performed in the same manner including the above-given preferences and therefore constitutes a further object of the invention.
The Examples further illustrate the present invention.
Example 1 : Preparation of a racemic compound of formula (II)
(i) 89 kg 2-bromo-3-methyl-5-acetylthiophene and 1 13 kg trifluoro-3,4,5- trichloroacetophenone are heated in about 880 kg tert. -butyl methylether and 121 kg triethylamine for 2 hours at 57°C. The reaction mixture is then cooled to below 5°C, and 67 kg thionyl chloride are added slowly. The reaction mixture is then heated for 3 hours to 40°C and afterwards extracted with 10% sodium hydrogencarbonate solution and water, the combined aqueous phases are extracted twice with tert. -butyl methylether, and the combined organic phases are extracted again with water. Following the removal of the solvent (tert-butyl methylether) by distillation the product of formula
is crystallized from ethanol/water.
(ii) 90 kg of the butanone bromothiophene from step (i) and 1000 kg ethanol are cooled below 5°C, and 13 kg hydroxylammonium chloride are added to the reaction mixture.
Following the slow addition of 55 kg of 30% sodium hydroxide the suspension is stirred for 3 hours . 1650 kg water are then added, and the reaction mixture is stirred for additional 2
hours. The product of formula
is centrifuged, washed with water and dried in vacuum.
(iii) 18.5 kg of the isoxazolin bromothiophene from step (ii) are dissolved in 80 kg tetrahydrofuran (THF) at ambient temperature. 18.6 kg of ethyl magnesium chloride (25% in THF) are added slowly, followed by the slow addition of 5.8 kg of C02 below the surface. The reaction mixture is then stirred for approximately 30 minutes at ambient temperature. Thereafter a mixture of 8 kg NaCI, 64 kg water and 7.9 kg HCI 37% is added. Following phase separation the organic layer is distilled to a minimum and heptane is added.
Following the removal of the heptane by distillation, the residuwe is dissolved in about 10 kg ethyl acetate, and the product is precipitated by the slow addition of 240 kg heptane at 40°C. The prod nd the racemic product of formula
Example 2: Enantiomer separation of racemic isoxazoline thiophene carboxylic acid of formula (II*) and collection of the S-enantiomer
100 kg of of racemic isoxazoline thiophene carboxylic acid from Example 1 , 171 kg 2- butanol, 680 kg acetonitrile and 45 kg water are stirred at ambient temperature. To this reaction mixture a solution of 15 kg (R)-1 -(4-methylphenyl)ethylamine, 19 kg 2-butanol, 76 kg acetonitrile and 5 kg water are added, and the reaction mixture is then slowly heated to 60°C and kept for 2 hours at this temperature. Thereafter the reaction mixture is cooled slowly to room temperature; the (S)-isoxazoline thiophene carboxylic acid salt precipitates and is collected by filtration and washed with a mixture of acetonitrile/water (9:1 ). Chiral purity of the product >95%.
The chiral purity of the product may be further increased by a further recrystallization step with acetonitrile/water (9:1 ) at 60°C and subsequent cooling, (chiral purity >98%).
Example 3: Racemization of (R)-isoxazoline thiophene carboxylic acid
3800 kg of the mother liquid from the racemic resolution of Example 2 are charged to a reactor and the solvent is removed under vacuum. 1750 kg of toluene are added and distilled off again. 54 kg of tributyl methylammonium chloride (as a 75% solution in water) 129 kg 50% aqueous sodium hydroxide solution and 1750 kg of toluene are added, and the mixture is then stirred for 6 hours at 75°C.The reaction mixture is then cooled to room temperature, followed by the addition of about 1600 kg of toluene. The organic layer is extracted three times with 1 molar HCI solution, and the toluene is then removed under vacuum. The residue is dissolved in about 1 10 kg of ethyl acetate at 40°C, and about 2600 kg of heptane are added slowly. Following cooling to ambient temperature, the product is isolated by centrifugation, washed with heptane and dried under vacuum. Yield: 80% racemic isoxazoline thiophene carboxylic acid of formula (II*), S/R enantiomer ratio
49.9/50.1 .
Example 4: Preparation of an (S)-enantiomer of formula (I) from an(S)-isoxazoline thiophene carboxylic acid salt
(i) 15 kg of the (S)-isoxazoline thiophene carboxylic acid salt from Example 2 in 150 kg of toluene are heated to 60°C. the solution is extracted three times with 56 kg of 1 molar HCI solution. About 50% of the organic layer is then distilled off under vacuum. Following the addition of further 60 kg of toluene the mixture is heated to 1 10°C and 8 kg of thionyl chloride are dosed slowly to the reaction mixture. Following stirring for an additional hour 94 kg of toluene are removed under vacuum. The reaction mixture is cooled to ambient temperature, and 94 kg of dichloromethane are added.
(ii) The dichloromethane solution of step (i) is added to a mixture of 5.1 kg 2-amino-2',2',2'- trifluoroethyl-acetamide hydrochloride and 9.8 kg of triethylamine in 93 kg of dichloromethane at 5°C. The reaction mixture is stirred for additional 5 hours and is then extracted with 4% hydrochloric acid, 8% sodium hydrogen carbonate and water. Most of the organic layer is then removed under vacuum, and 50 kg of toluene are added. The reaction mixture is kept at 40°C, and 290 kg of heptane are added slowly to precipitate the product. The suspension obtained is cooled to below 5°C, and the crude product is isolated by filtration and is washed with 25 kg of heptane. Crystallization of the product may be performed as appropriate.
Claims
1 . Process for the manufacture of an enantiomer of a compound of formula
(I)
or a veterinary acceptable salt thereof, wherein
R\ R" and R'" are each independently hydrogen, halogen, cyano, CrC2-alkyl, halo-Ci-C2- alkyl, CrC2-alkoxy or CrC2-haloalkoxy, subject to the proviso that at least one of R', R" and R'" is not hydrogen;
Y is phenylene, naphthylene, indanylene, 5- or 6-membered heteroarylene or 8-10- membered fused heterobicyclylene, which is each unsubstituted or substituted by Ci-C2- alkyl, Ci-C2-haloalkyl, halogen, nitro or cyano, and,
provided that Y is phenylene, naphthylene, heteroarylene or fused heterobicycylene, Q is
(i) a 5- or 6-membered heteroaromatic ring comprising 1 to 3 same or different heteroatoms selected from the group consisting of O, S and N which is further unsubstituted or substituted; or is
(ii) a group -C(0)N(R1)-T, wherein is H, C C4-alkyl, C2-C4-alkylcarbonyl or C2-C4- alkoxycarbonyl and T is CrC6-alkyl which is unsubstituted or substituted by C3-C6-cycloalkyl, halogen, cyano, nitro, amino, hydroxy, CrC6-alkoxy, CrC6-haloalkoxy, CrC6-alkylthio, d- C6-haloalkylthio, CrC6-alkylsulfinyl, CrC6-haloalkylsulfinyl, CrC6-alkylsulfonyl, CrC6- haloalkylsulfonyl, carboxy, carbamoyl, Ci-C6-alkylcarbonylamino, CrC6-haloalkyl- carbonylamino, CrC6-alkoxycarbonyl, sulfonamido, N-mono- or N,N, di-CrC4-alkylsulfon- amido, C2-C6-alkanoyl, unsubstituted or in the alkyl portion by halogen, cyano, ethenyl or ethynyl substituted N-Ci-C6-alkylaminocarbonyl, or unsubstituted or halogen-, Ci-C2-alkyl-, CrC2-haloalkyl or cyano-substituted 4- to 6-membered heterocyclyl; or T is C3-C6-cycloalkyl or 4- to 6-membered heterocyclyl, which is each unsubstituted or substituted by halogen, Ci-C2-alkyl, Ci-C2-haloalkyl or cyano; or is
(iii) a radical -C(0)NH-CH=N-0-C C2-alkyl, a radical -C(0)N=CH-N-di-C C2-alkyl or a radical -C(0)N=C(NH2)-0-C C2-alkyl; or is
(iv) a group -CH(R3)-N(R4)-C(0)-T1, wherein R3 is H, CrC6-alkyl, CrC6-haloalkyl, halogen or cyano, R4 is H; d-C4-alkyl C2-C4-alkylcarbonyl or C2-C4-alkoxycarbonyl, and Ti is
independently defined as T above; or,
provided that Y is indanylene, Q is a group -N(R4)-C(0)-T2, wherein T2 independently has the meaning of T above and R4 is as defined above;
comprising the steps of
(A) separating a racemic compound of formula
wherein R', R"and R'" and Y are as defined above and Z is Q; or is halogen, amino, carboxy
(-COOH) or a salt or a derivative thereof; into the enantiomers and collecting the desired enantiomer;
(B) if Z is other than Q, converting the desired enantiomer of formula (II) from step (A) to the desired enantiomer of formula (I);
(C) racemizing the undesired enantiomer of formula (II) from step (A) in a reaction medium comprising a base and an aprotic organic solvent at a temperature of from 30°C to 150°C, and,
(D) subjecting the racemate from step (C) to a further manufacturing cycle comprising step (A) and optionally (B).
2. The process according to claim 1 , wherein the desired enantiomer is the S-enantiomer, and the undesired enantiomer is the R-enantiomer.
3. The process according to claim 1 or 2, wherein Z is Q.
4. The process according to claim 1 or 2, wherein Z is halogen, amino, carboxy (-COOH) or a salt thereof, in particular carboxy or a salt thereof.
5. The process according to claim 1 for the manufacture of the S-enantiomer of a compound of formula
or a veterinary acceptable salt thereof, wherein for R', R", R"\ Y and T are each as defined in claim 1 ; comprising the step
(A) separating a racemic co
(II).
wherein R', R" and R'" and Y are as defined and Z carboxy (-COOH) or a salt thereof; into the enantiomers and collecting the S-enantiomer.
6. The process according to claim 5, for the manufacture of the S-enantiomer of a compound of formula
(la) or a veterinary acceptable salt thereof, wherein R' and R'" are each independently CI or CF3, R" is H or CI; and Y is a radical
comprising the step
wherein R', R" and R'" and Y are as defined above and Z carboxy (-COOH) or a salt thereof; into the enantiomers, and collecting the S-enantiomer.
7. The process according to any one of claims 1 to 6, wherein separation of the racemic compound of formula (II) into the enantiomers according to step (A) is performed by chiral column chromatography or by diastereomeric recrystallization.
8. The process according to claim 7, wherein a compound of formula (II), which is an amine or acid or a salt thereof, is reacted with an optical pure reagent which is an acid or amine, thereby forming a pair of diastereomeric salts which are then separated.
9. The process according to any one of claims 1 to 8, wherein the reaction medium in step (C) comprises an aromatic hydrocarbon and water.
10. Process according to claim 9, wherein the reaction medium comprises, in addition, a phase transfer catalyst.
1 1 . The process according to claim 9 or 10, wherein in step (C) the R-enantiomer of formula (II) is racemized in a reaction medium comprising an aromatic hydrocarbon, water, a phase transfer catalyst and a base which is an alkali metal hydroxide at a temperature from 50°C to 100°C.
12. The process according to claim 1 for the manufacture of the S-enantiomer of a compound of formula
or a veterinary acceptable salt thereof, wherein R' and R'" are each independently CI or CF R" is H or CI; and Y is a radical
(A) separating a racemic compound of formula
wherein R', R" and R'" and Y are as defined above and Z carboxy (-COOH) or a salt thereof; into the enantiomers by diastereomeric recrystallization, and collecting the S- enantiomer;
(B) converting the S-enantiomer of formula (II) from step (A) to the S-enantiomer of formula
(I);
(C) racemizing the R-enantiomer of formula (II) from step (A) in a reaction medium comprising an aromatic hydrocarbon, water, a phase transfer catalyst and a base which is an alkali metal hydroxide at a temperature from about 50°C to about 100°C; and
(D) subjecting the racemate from step (C) to a further manufacturing cycle comprising steps (A) and (B).
wherein R', R" and R'", Y and Z are as defined in claim 1 ,
comprising treating the enantiomer of formula (II) in a reaction medium comprising a base and an aprotic organic solvent at a temperature of from 30°C to 150°C.
14. The process according to claim 13, wherein the R-enantiomer of formula (II) is racemized in a reaction medium comprising an aromatic hydrocarbon, water, a phase transfer catalyst and a base which is an alkali metal hydroxide at a temperature from 50°C to 100°C.
15. The compound (S)-5-[5-(3,4,5-trichloro-phenyl)-5-trifluoromethyl-4,5-dihydro-isoxazol-3- yl]-3-methyl-thiophene-2-carboxylic acid [(2,2,2-trifluoro-ethylcarbamoyl)-methyl]-amide.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP12197067 | 2012-12-13 | ||
| EP12197067.7 | 2012-12-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2014090918A1 true WO2014090918A1 (en) | 2014-06-19 |
Family
ID=47355902
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2013/076321 Ceased WO2014090918A1 (en) | 2012-12-13 | 2013-12-12 | Process for the enantiomeric enrichment of diaryloxazoline derivatives |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2014090918A1 (en) |
Cited By (84)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3018129A1 (en) | 2014-11-10 | 2016-05-11 | Novartis Tiergesundheit AG | Diaryl isoxazoline compound |
| WO2016071499A1 (en) | 2014-11-06 | 2016-05-12 | Basf Se | 3-pyridyl heterobicyclic compound for controlling invertebrate pests |
| WO2016138339A1 (en) | 2015-02-26 | 2016-09-01 | Merial, Inc. | Long-acting injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| WO2016162371A1 (en) | 2015-04-07 | 2016-10-13 | Basf Agrochemical Products B.V. | Use of an insecticidal carboxamide compound against pests on cultivated plants |
| WO2016198611A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino heterocyclic compounds |
| WO2016198613A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino compounds |
| WO2017016883A1 (en) | 2015-07-24 | 2017-02-02 | Basf Se | Process for preparation of cyclopentene compounds |
| WO2017093163A1 (en) | 2015-11-30 | 2017-06-08 | Basf Se | Mixtures of cis-jasmone and bacillus amyloliquefaciens |
| WO2017147352A1 (en) | 2016-02-24 | 2017-08-31 | Merial, Inc. | Antiparasitic isoxazoline compounds, long-acting injectable formulations comprising them, methods and uses thereof |
| WO2017153217A1 (en) | 2016-03-09 | 2017-09-14 | Basf Se | Spirocyclic derivatives |
| WO2017153218A1 (en) | 2016-03-11 | 2017-09-14 | Basf Se | Method for controlling pests of plants |
| WO2017167832A1 (en) | 2016-04-01 | 2017-10-05 | Basf Se | Bicyclic compounds |
| WO2017176948A1 (en) * | 2016-04-06 | 2017-10-12 | Merial, Inc. | Process for the preparation of enantiomerically enriched isoxazoline compounds - crystalline toluene solvate of (s)-afoxolaner |
| WO2017196607A1 (en) * | 2016-05-10 | 2017-11-16 | Elanco Tiergesundheit Ag | Dihydroisoxazole compound for use in controlling sea lice |
| JP2017538767A (en) * | 2014-12-22 | 2017-12-28 | インターベット インターナショナル ベー. フェー. | Use of isoxazoline compounds for the treatment of folliculosis |
| WO2018039508A1 (en) * | 2016-08-25 | 2018-03-01 | Merial, Inc. | Method for reducing unwanted effects in parasiticidal treatments |
| WO2018071535A1 (en) | 2016-10-14 | 2018-04-19 | Merial, Inc. | Pesticidal and parasiticidal vinyl isoxazoline compounds |
| WO2018108671A1 (en) | 2016-12-16 | 2018-06-21 | Basf Se | Pesticidal compounds |
| WO2018162312A1 (en) | 2017-03-10 | 2018-09-13 | Basf Se | Spirocyclic derivatives |
| WO2018166855A1 (en) | 2017-03-16 | 2018-09-20 | Basf Se | Heterobicyclic substituted dihydroisoxazoles |
| WO2018177781A1 (en) | 2017-03-28 | 2018-10-04 | Basf Se | Pesticidal compounds |
| WO2018177970A1 (en) | 2017-03-31 | 2018-10-04 | Basf Se | Process for preparing chiral 2,3-dihydrothiazolo[3,2-a]pyrimidin-4-ium compounds |
| WO2018192793A1 (en) | 2017-04-20 | 2018-10-25 | Basf Se | Substituted rhodanine derivatives |
| WO2018197466A1 (en) | 2017-04-26 | 2018-11-01 | Basf Se | Substituted succinimide derivatives as pesticides |
| WO2018206479A1 (en) | 2017-05-10 | 2018-11-15 | Basf Se | Bicyclic pesticidal compounds |
| US10149477B2 (en) | 2014-10-06 | 2018-12-11 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| WO2018224455A1 (en) | 2017-06-07 | 2018-12-13 | Basf Se | Substituted cyclopropyl derivatives |
| WO2018229202A1 (en) | 2017-06-16 | 2018-12-20 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2018234202A1 (en) | 2017-06-19 | 2018-12-27 | Basf Se | SUBSTITUTED PYRIMIDINIUM COMPOUNDS AND DERIVATIVES FOR CONTROLLING HARMFUL ANIMALS |
| WO2018234488A1 (en) | 2017-06-23 | 2018-12-27 | Basf Se | SUBSTITUTED CYCLOPROPYL DERIVATIVES |
| WO2019036407A1 (en) | 2017-08-14 | 2019-02-21 | Merial, Inc. | Pesticidal and parasiticidal pyrazole-isoxazoline compounds |
| WO2019042932A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| EP3453706A1 (en) | 2017-09-08 | 2019-03-13 | Basf Se | Pesticidal imidazole compounds |
| WO2019072906A1 (en) | 2017-10-13 | 2019-04-18 | Basf Se | Imidazolidine pyrimidinium compounds for combating animal pests |
| WO2019121159A1 (en) | 2017-12-21 | 2019-06-27 | Basf Se | Pesticidal compounds |
| WO2019121143A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Substituted cyclopropyl derivatives |
| WO2019137995A1 (en) | 2018-01-11 | 2019-07-18 | Basf Se | Novel pyridazine compounds for controlling invertebrate pests |
| WO2019145140A1 (en) | 2018-01-09 | 2019-08-01 | Basf Se | Silylethynyl hetaryl compounds as nitrification inhibitors |
| WO2019166561A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of alkoxypyrazoles as nitrification inhibitors |
| WO2019166560A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of n-functionalized alkoxy pyrazole compounds as nitrification inhibitors |
| WO2019166558A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of pyrazole propargyl ethers as nitrification inhibitors |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019185413A1 (en) | 2018-03-27 | 2019-10-03 | Basf Se | Pesticidal substituted cyclopropyl derivatives |
| WO2019219529A1 (en) | 2018-05-15 | 2019-11-21 | Basf Se | Mixtures comprising benzpyrimoxan and oxazosulfyl and uses and methods of applying them |
| WO2019224092A1 (en) | 2018-05-22 | 2019-11-28 | Basf Se | Pesticidally active c15-derivatives of ginkgolides |
| WO2020002472A1 (en) | 2018-06-28 | 2020-01-02 | Basf Se | Use of alkynylthiophenes as nitrification inhibitors |
| WO2020020765A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of a substituted thiazolidine compound as nitrification inhibitor |
| WO2020020777A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of substituted 2-thiazolines as nitrification inhibitors |
| EP3613736A1 (en) | 2018-08-22 | 2020-02-26 | Basf Se | Substituted glutarimide derivatives |
| EP3628157A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| EP3628158A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| EP3628156A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method for controlling pests of sugarcane, citrus, rapeseed, and potato plants |
| WO2020064492A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling pests by seed treatment application of a mesoionic compound or mixture thereof |
| CN111032634A (en) * | 2017-04-05 | 2020-04-17 | 勃林格殷格翰动物保健美国公司 | The crystal form of (S)-Aflana |
| EP3643705A1 (en) | 2018-10-24 | 2020-04-29 | Basf Se | Pesticidal compounds |
| WO2020088949A1 (en) | 2018-10-29 | 2020-05-07 | Basf Se | Process for preparation of optically enriched aldol compounds |
| WO2020094434A1 (en) | 2018-11-06 | 2020-05-14 | Basf Se | Process for preparation of optically enriched isoxazolines |
| WO2020109039A1 (en) | 2018-11-28 | 2020-06-04 | Basf Se | Pesticidal compounds |
| WO2020126591A1 (en) | 2018-12-18 | 2020-06-25 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| WO2020127878A1 (en) | 2018-12-21 | 2020-06-25 | Intervet International B.V. | Method for producing (5s)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4h-isoxazol-3-yl]-2-methyl-benzoic acid |
| WO2020127944A1 (en) | 2018-12-21 | 2020-06-25 | Intervet International B.V. | Method for racemisation of (5r)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4h-isoxazol-3-yl]-2-methyl-benzoic acid |
| WO2020127935A1 (en) | 2018-12-21 | 2020-06-25 | Intervet International B.V. | Method for producing (5s)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4hisoxazol-3-yl]-2-methyl-benzoic acid |
| EP3696177A1 (en) | 2019-02-12 | 2020-08-19 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2020239517A1 (en) | 2019-05-29 | 2020-12-03 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| EP3766879A1 (en) | 2019-07-19 | 2021-01-20 | Basf Se | Pesticidal pyrazole derivatives |
| EP3769623A1 (en) | 2019-07-22 | 2021-01-27 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| EP3725782A4 (en) * | 2017-12-15 | 2021-04-14 | Kumiai Chemical Industry Co., Ltd. | NEW PROCESS FOR THE PRODUCTION OF 4,5-DIHYDROISOXAZOLE 5,5-DISUBSTITUTED |
| WO2021123051A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides, thiophene carboxylic acids and derivatives thereof |
| US11142514B2 (en) | 2015-10-02 | 2021-10-12 | Basf Se | Imino compounds with a 2-chloropyrimidin-5-yl substituent as pest-control agents |
| WO2022016490A1 (en) * | 2020-07-24 | 2022-01-27 | Elanco Us Inc. | Process for lotilaner intermediate |
| WO2022020585A1 (en) | 2020-07-24 | 2022-01-27 | Elanco Us Inc. | Process for making an isoxazoline compound and intermediate thereof |
| WO2022167488A1 (en) | 2021-02-02 | 2022-08-11 | Basf Se | Synergistic action of dcd and alkoxypyrazoles as nitrification inhibitors |
| WO2022243521A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of ethynylpyridine compounds as nitrification inhibitors |
| WO2022243523A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of an n-functionalized alkoxy pyrazole compound as nitrification inhibitor |
| RU2786390C1 (en) * | 2018-12-21 | 2022-12-20 | Интервет Интернэшнл Б.В. | Method for producing (5s)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4h-isoxazol-3-yl]-2-methylbenzoic acid |
| CN115504971A (en) * | 2022-11-09 | 2022-12-23 | 世华合创生物技术开发(山东)有限公司 | Synthetic method of lotilanide |
| WO2022268810A1 (en) | 2021-06-21 | 2022-12-29 | Basf Se | Metal-organic frameworks with pyrazole-based building blocks |
| WO2023018806A1 (en) | 2021-08-11 | 2023-02-16 | ELANCO US, Inc. | Process for making diaryl isoxazoline derivative |
| WO2023107660A1 (en) * | 2021-12-10 | 2023-06-15 | Teva Pharmaceuticals International Gmbh | Solid state forms of lotilaner and process for preparation thereof |
| WO2023203066A1 (en) | 2022-04-21 | 2023-10-26 | Basf Se | Synergistic action as nitrification inhibitors of dcd oligomers with alkoxypyrazole and its oligomers |
| WO2024028243A1 (en) | 2022-08-02 | 2024-02-08 | Basf Se | Pyrazolo pesticidal compounds |
| CN119798241A (en) * | 2025-03-17 | 2025-04-11 | 北京元延医药科技股份有限公司 | A simple method for preparing high-purity lotirana |
| US12497370B2 (en) | 2019-12-18 | 2025-12-16 | Elanco Tiergesundheit Ag | Isoxazoline derivatives |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005085216A1 (en) | 2004-03-05 | 2005-09-15 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and noxious organism control agent |
| WO2006050887A1 (en) | 2004-11-09 | 2006-05-18 | Novartis Ag | Process for the preparation of enantiomers of amidoacetonitrile compounds from their racemates |
| WO2010070068A2 (en) | 2008-12-19 | 2010-06-24 | Novartis Ag | Organic compounds |
| WO2011157748A1 (en) | 2010-06-17 | 2011-12-22 | Novartis Ag | 5-aryl isoxazolines for controlling pests |
-
2013
- 2013-12-12 WO PCT/EP2013/076321 patent/WO2014090918A1/en not_active Ceased
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2005085216A1 (en) | 2004-03-05 | 2005-09-15 | Nissan Chemical Industries, Ltd. | Isoxazoline-substituted benzamide compound and noxious organism control agent |
| WO2006050887A1 (en) | 2004-11-09 | 2006-05-18 | Novartis Ag | Process for the preparation of enantiomers of amidoacetonitrile compounds from their racemates |
| WO2010070068A2 (en) | 2008-12-19 | 2010-06-24 | Novartis Ag | Organic compounds |
| WO2011157748A1 (en) | 2010-06-17 | 2011-12-22 | Novartis Ag | 5-aryl isoxazolines for controlling pests |
Non-Patent Citations (3)
| Title |
|---|
| DATABASE REGISTRY [Online] CHEMICAL ABSTRACTS SERVICE, COLUMBUS, OHIO, US; 18 April 2012 (2012-04-18), XP002691588, Database accession no. 1369852-71-0 * |
| JAAN A. PESTI ET AL: "Reversible Michael Reaction-Enzymatic Hydrolysis: A New Variant of Dynamic Resolution", JOURNAL OF THE AMERICAN CHEMICAL SOCIETY, vol. 123, no. 44, 1 November 2001 (2001-11-01), pages 11075 - 11076, XP055052375, ISSN: 0002-7863, DOI: 10.1021/ja011811l * |
| OZOE Y ET AL: "The antiparasitic isoxazoline A1443 is a potent blocker of insect ligand-gated chloride channels", BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS, ACADEMIC PRESS INC. ORLANDO, FL, US, vol. 391, no. 1, 1 January 2010 (2010-01-01), pages 744 - 749, XP026907975, ISSN: 0006-291X, [retrieved on 20091126], DOI: 10.1016/J.BBRC.2009.11.131 * |
Cited By (122)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US10149477B2 (en) | 2014-10-06 | 2018-12-11 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| WO2016071499A1 (en) | 2014-11-06 | 2016-05-12 | Basf Se | 3-pyridyl heterobicyclic compound for controlling invertebrate pests |
| EP3018129A1 (en) | 2014-11-10 | 2016-05-11 | Novartis Tiergesundheit AG | Diaryl isoxazoline compound |
| WO2016077158A1 (en) | 2014-11-10 | 2016-05-19 | Novartis Tiergesundheit Ag | Diaryl isoxazoline compound |
| JP2017538767A (en) * | 2014-12-22 | 2017-12-28 | インターベット インターナショナル ベー. フェー. | Use of isoxazoline compounds for the treatment of folliculosis |
| WO2016138339A1 (en) | 2015-02-26 | 2016-09-01 | Merial, Inc. | Long-acting injectable formulations comprising an isoxazoline active agent, methods and uses thereof |
| WO2016162371A1 (en) | 2015-04-07 | 2016-10-13 | Basf Agrochemical Products B.V. | Use of an insecticidal carboxamide compound against pests on cultivated plants |
| WO2016198613A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino compounds |
| WO2016198611A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino heterocyclic compounds |
| WO2017016883A1 (en) | 2015-07-24 | 2017-02-02 | Basf Se | Process for preparation of cyclopentene compounds |
| US11142514B2 (en) | 2015-10-02 | 2021-10-12 | Basf Se | Imino compounds with a 2-chloropyrimidin-5-yl substituent as pest-control agents |
| WO2017093163A1 (en) | 2015-11-30 | 2017-06-08 | Basf Se | Mixtures of cis-jasmone and bacillus amyloliquefaciens |
| WO2017147352A1 (en) | 2016-02-24 | 2017-08-31 | Merial, Inc. | Antiparasitic isoxazoline compounds, long-acting injectable formulations comprising them, methods and uses thereof |
| EP3763211A1 (en) | 2016-02-24 | 2021-01-13 | Boehringer Ingelheim Animal Health USA Inc. | Antiparasitic isoxazoline compounds, long-acting injectable formulations comprising them, methods and uses thereof |
| WO2017153217A1 (en) | 2016-03-09 | 2017-09-14 | Basf Se | Spirocyclic derivatives |
| WO2017153218A1 (en) | 2016-03-11 | 2017-09-14 | Basf Se | Method for controlling pests of plants |
| WO2017167832A1 (en) | 2016-04-01 | 2017-10-05 | Basf Se | Bicyclic compounds |
| TWI753897B (en) * | 2016-04-06 | 2022-02-01 | 美商百靈佳殷格翰動物保健美國有限公司 | Process for the preparation of isoxazoline compounds |
| CN114805234A (en) * | 2016-04-06 | 2022-07-29 | 勃林格殷格翰动物保健美国公司 | Process for the preparation of crystalline toluene solvates of enantiomerically enriched isoxazoline compounds-(S)-afoxolaner |
| US11324220B2 (en) | 2016-04-06 | 2022-05-10 | Boehringer Ingelheim Animal Health USA Inc. | Process for the preparation of isoxazoline compounds |
| EP3978483A1 (en) * | 2016-04-06 | 2022-04-06 | Boehringer Ingelheim Animal Health USA Inc. | Crystalline toluene solvate of (s)-afoxolaner |
| WO2017176948A1 (en) * | 2016-04-06 | 2017-10-12 | Merial, Inc. | Process for the preparation of enantiomerically enriched isoxazoline compounds - crystalline toluene solvate of (s)-afoxolaner |
| US10433552B2 (en) | 2016-04-06 | 2019-10-08 | Merial, Inc. | Process for the preparation of isoxazoline compounds |
| US10750744B2 (en) | 2016-04-06 | 2020-08-25 | Boehringer Ingelheim Animal Health USA Inc. | Process for the preparation of isoxazoline compounds |
| US10537549B2 (en) | 2016-05-10 | 2020-01-21 | Elanco Tiergesundheit Ag | Dihydroisoxazole compound for use in controlling sea lice |
| WO2017196607A1 (en) * | 2016-05-10 | 2017-11-16 | Elanco Tiergesundheit Ag | Dihydroisoxazole compound for use in controlling sea lice |
| WO2018039508A1 (en) * | 2016-08-25 | 2018-03-01 | Merial, Inc. | Method for reducing unwanted effects in parasiticidal treatments |
| WO2018071535A1 (en) | 2016-10-14 | 2018-04-19 | Merial, Inc. | Pesticidal and parasiticidal vinyl isoxazoline compounds |
| WO2018108671A1 (en) | 2016-12-16 | 2018-06-21 | Basf Se | Pesticidal compounds |
| WO2018162312A1 (en) | 2017-03-10 | 2018-09-13 | Basf Se | Spirocyclic derivatives |
| WO2018166855A1 (en) | 2017-03-16 | 2018-09-20 | Basf Se | Heterobicyclic substituted dihydroisoxazoles |
| WO2018177781A1 (en) | 2017-03-28 | 2018-10-04 | Basf Se | Pesticidal compounds |
| EP3978504A1 (en) | 2017-03-31 | 2022-04-06 | Basf Se | Chiral 2,3-dihydrothiazolo[3,2-a]pyrimidine derivatives for combating animal pests |
| WO2018177970A1 (en) | 2017-03-31 | 2018-10-04 | Basf Se | Process for preparing chiral 2,3-dihydrothiazolo[3,2-a]pyrimidin-4-ium compounds |
| CN111032634A (en) * | 2017-04-05 | 2020-04-17 | 勃林格殷格翰动物保健美国公司 | The crystal form of (S)-Aflana |
| WO2018192793A1 (en) | 2017-04-20 | 2018-10-25 | Basf Se | Substituted rhodanine derivatives |
| WO2018197466A1 (en) | 2017-04-26 | 2018-11-01 | Basf Se | Substituted succinimide derivatives as pesticides |
| WO2018206479A1 (en) | 2017-05-10 | 2018-11-15 | Basf Se | Bicyclic pesticidal compounds |
| WO2018224455A1 (en) | 2017-06-07 | 2018-12-13 | Basf Se | Substituted cyclopropyl derivatives |
| WO2018229202A1 (en) | 2017-06-16 | 2018-12-20 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2018234202A1 (en) | 2017-06-19 | 2018-12-27 | Basf Se | SUBSTITUTED PYRIMIDINIUM COMPOUNDS AND DERIVATIVES FOR CONTROLLING HARMFUL ANIMALS |
| WO2018234488A1 (en) | 2017-06-23 | 2018-12-27 | Basf Se | SUBSTITUTED CYCLOPROPYL DERIVATIVES |
| WO2019036407A1 (en) | 2017-08-14 | 2019-02-21 | Merial, Inc. | Pesticidal and parasiticidal pyrazole-isoxazoline compounds |
| WO2019042932A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| WO2019043183A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| EP3453706A1 (en) | 2017-09-08 | 2019-03-13 | Basf Se | Pesticidal imidazole compounds |
| WO2019072906A1 (en) | 2017-10-13 | 2019-04-18 | Basf Se | Imidazolidine pyrimidinium compounds for combating animal pests |
| EP3725782A4 (en) * | 2017-12-15 | 2021-04-14 | Kumiai Chemical Industry Co., Ltd. | NEW PROCESS FOR THE PRODUCTION OF 4,5-DIHYDROISOXAZOLE 5,5-DISUBSTITUTED |
| IL275272B (en) * | 2017-12-15 | 2022-08-01 | Kumiai Chemical Industry Co | A method for the production of 5,5-di-converted-5,4- dihydroisoxazole |
| WO2019121143A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Substituted cyclopropyl derivatives |
| WO2019121159A1 (en) | 2017-12-21 | 2019-06-27 | Basf Se | Pesticidal compounds |
| WO2019145140A1 (en) | 2018-01-09 | 2019-08-01 | Basf Se | Silylethynyl hetaryl compounds as nitrification inhibitors |
| WO2019137995A1 (en) | 2018-01-11 | 2019-07-18 | Basf Se | Novel pyridazine compounds for controlling invertebrate pests |
| WO2019166561A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of alkoxypyrazoles as nitrification inhibitors |
| WO2019166558A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of pyrazole propargyl ethers as nitrification inhibitors |
| WO2019166560A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of n-functionalized alkoxy pyrazole compounds as nitrification inhibitors |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| WO2019185413A1 (en) | 2018-03-27 | 2019-10-03 | Basf Se | Pesticidal substituted cyclopropyl derivatives |
| WO2019219529A1 (en) | 2018-05-15 | 2019-11-21 | Basf Se | Mixtures comprising benzpyrimoxan and oxazosulfyl and uses and methods of applying them |
| WO2019224092A1 (en) | 2018-05-22 | 2019-11-28 | Basf Se | Pesticidally active c15-derivatives of ginkgolides |
| WO2020002472A1 (en) | 2018-06-28 | 2020-01-02 | Basf Se | Use of alkynylthiophenes as nitrification inhibitors |
| WO2020020777A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of substituted 2-thiazolines as nitrification inhibitors |
| WO2020020765A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of a substituted thiazolidine compound as nitrification inhibitor |
| EP3613736A1 (en) | 2018-08-22 | 2020-02-26 | Basf Se | Substituted glutarimide derivatives |
| WO2020064408A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| WO2020064480A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| WO2020064492A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling pests by seed treatment application of a mesoionic compound or mixture thereof |
| EP3628156A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method for controlling pests of sugarcane, citrus, rapeseed, and potato plants |
| EP3628158A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| EP3628157A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| WO2020083733A1 (en) | 2018-10-24 | 2020-04-30 | Basf Se | Pesticidal compounds |
| EP3643705A1 (en) | 2018-10-24 | 2020-04-29 | Basf Se | Pesticidal compounds |
| WO2020088949A1 (en) | 2018-10-29 | 2020-05-07 | Basf Se | Process for preparation of optically enriched aldol compounds |
| WO2020094434A1 (en) | 2018-11-06 | 2020-05-14 | Basf Se | Process for preparation of optically enriched isoxazolines |
| WO2020109039A1 (en) | 2018-11-28 | 2020-06-04 | Basf Se | Pesticidal compounds |
| WO2020126591A1 (en) | 2018-12-18 | 2020-06-25 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| CN113242854A (en) * | 2018-12-21 | 2021-08-10 | 英特维特国际股份有限公司 | Preparation method of (5S) -4- [5- (3, 5-dichlorophenyl) -5- (trifluoromethyl) -4H-isoxazol-3-yl ] -2-methylbenzoic acid |
| RU2809762C2 (en) * | 2018-12-21 | 2023-12-18 | Интервет Интернэшнл Б.В. | Method of producing (5s)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4h-isoxazol-3-yl]-2-methyl-benzoic acid |
| CN113195459A (en) * | 2018-12-21 | 2021-07-30 | 英特维特国际股份有限公司 | Process for the racemisation of (5R) -4- [5- (3, 5-dichlorophenyl) -5- (trifluoromethyl) -4H isoxazol-3-yl ] -2-methyl-benzoic acid |
| CN113227060A (en) * | 2018-12-21 | 2021-08-06 | 英特维特国际股份有限公司 | Preparation method of (5S) -4- [5- (3, 5-dichlorophenyl) -5- (trifluoromethyl) -4H-isoxazol-3-yl ] -2-methylbenzoic acid |
| AU2019408398B2 (en) * | 2018-12-21 | 2025-03-13 | Intervet International B.V. | Method for racemisation of (5R)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-2-methyl-benzoic acid |
| US11548859B2 (en) | 2018-12-21 | 2023-01-10 | Intervet Inc. | Method for producing (5,S)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4Hisoxazol-3-yl]-2-methyl-benzoic acid |
| CN113195459B (en) * | 2018-12-21 | 2024-11-01 | 英特维特国际股份有限公司 | Process for the racemization of (5R)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-2-methyl-benzoic acid |
| JP7668215B2 (en) | 2018-12-21 | 2025-04-24 | インターベット インターナショナル ベー. フェー. | Method for preparing (5S)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-2-methyl-benzoic acid |
| RU2786390C1 (en) * | 2018-12-21 | 2022-12-20 | Интервет Интернэшнл Б.В. | Method for producing (5s)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4h-isoxazol-3-yl]-2-methylbenzoic acid |
| JP2022514305A (en) * | 2018-12-21 | 2022-02-10 | インターベット インターナショナル ベー. フェー. | (5S) -4- [5- (3,5-dichlorophenyl) -5- (trifluoromethyl) -4H-isoxazole-3-yl] -2-methyl-benzoic acid production method |
| JP2022514306A (en) * | 2018-12-21 | 2022-02-10 | インターベット インターナショナル ベー. フェー. | (5S) -4- [5- (3,5-dichlorophenyl) -5- (trifluoromethyl) -4H-isoxazole-3-yl] -2-methyl-benzoic acid |
| JP2022514740A (en) * | 2018-12-21 | 2022-02-15 | インターベット インターナショナル ベー. フェー. | (5R) -4- [5- (3,5-dichlorophenyl) -5- (trifluoromethyl) -4H-isoxazole-3-yl] -2-methyl-benzoic acid racemization method |
| US11530187B2 (en) | 2018-12-21 | 2022-12-20 | Intervet Inc. | Method for producing (5S)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-2-methyl-benzoic acid |
| RU2805659C2 (en) * | 2018-12-21 | 2023-10-23 | Интервет Интернэшнл Б.В. | Racemization method of (5r)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4h-isoxazol-3-yl]-2-methylbenzoic acid |
| WO2020127935A1 (en) | 2018-12-21 | 2020-06-25 | Intervet International B.V. | Method for producing (5s)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4hisoxazol-3-yl]-2-methyl-benzoic acid |
| WO2020127944A1 (en) | 2018-12-21 | 2020-06-25 | Intervet International B.V. | Method for racemisation of (5r)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4h-isoxazol-3-yl]-2-methyl-benzoic acid |
| WO2020127878A1 (en) | 2018-12-21 | 2020-06-25 | Intervet International B.V. | Method for producing (5s)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4h-isoxazol-3-yl]-2-methyl-benzoic acid |
| US12077514B2 (en) | 2018-12-21 | 2024-09-03 | Intervet Inc. | Method for racemisation of (5R)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H¬isoxazol-3-yl]-2-methyl-benzoic acid |
| JP7668216B2 (en) | 2018-12-21 | 2025-04-24 | インターベット インターナショナル ベー. フェー. | Process for preparing (5S)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-2-methyl-benzoic acid |
| EP3696177A1 (en) | 2019-02-12 | 2020-08-19 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2020239517A1 (en) | 2019-05-29 | 2020-12-03 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| EP3766879A1 (en) | 2019-07-19 | 2021-01-20 | Basf Se | Pesticidal pyrazole derivatives |
| WO2021013561A1 (en) | 2019-07-19 | 2021-01-28 | Basf Se | Pesticidal pyrazole and triazole derivatives |
| EP3769623A1 (en) | 2019-07-22 | 2021-01-27 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| US12497370B2 (en) | 2019-12-18 | 2025-12-16 | Elanco Tiergesundheit Ag | Isoxazoline derivatives |
| WO2021123051A1 (en) | 2019-12-20 | 2021-06-24 | Bayer Aktiengesellschaft | Substituted thiophene carboxamides, thiophene carboxylic acids and derivatives thereof |
| TWI899292B (en) * | 2020-07-24 | 2025-10-01 | 美商益農美國公司 | Process for making an isoxazoline compound and intermediate thereof |
| JP7724845B2 (en) | 2020-07-24 | 2025-08-18 | エランコ・ユーエス・インコーポレイテッド | Methods for making isoxazoline compounds and intermediates therefor - Patent Application 20070122997 |
| JP2023536070A (en) * | 2020-07-24 | 2023-08-23 | エランコ・ユーエス・インコーポレイテッド | Method for making isoxazoline compounds and intermediates thereof |
| CN116194450A (en) * | 2020-07-24 | 2023-05-30 | 礼蓝美国公司 | Process for preparing isoxazoline compounds and intermediates thereof |
| US12304903B2 (en) | 2020-07-24 | 2025-05-20 | Elanco Us Inc. | Process for making an isoxazoline compound and intermediate thereof |
| WO2022020585A1 (en) | 2020-07-24 | 2022-01-27 | Elanco Us Inc. | Process for making an isoxazoline compound and intermediate thereof |
| WO2022016490A1 (en) * | 2020-07-24 | 2022-01-27 | Elanco Us Inc. | Process for lotilaner intermediate |
| WO2022167488A1 (en) | 2021-02-02 | 2022-08-11 | Basf Se | Synergistic action of dcd and alkoxypyrazoles as nitrification inhibitors |
| WO2022243523A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of an n-functionalized alkoxy pyrazole compound as nitrification inhibitor |
| WO2022243521A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of ethynylpyridine compounds as nitrification inhibitors |
| WO2022268810A1 (en) | 2021-06-21 | 2022-12-29 | Basf Se | Metal-organic frameworks with pyrazole-based building blocks |
| US20240308993A1 (en) * | 2021-08-11 | 2024-09-19 | Elanco Us Inc. | Process for making diaryl isoxazoline derivative |
| WO2023018806A1 (en) | 2021-08-11 | 2023-02-16 | ELANCO US, Inc. | Process for making diaryl isoxazoline derivative |
| WO2023107660A1 (en) * | 2021-12-10 | 2023-06-15 | Teva Pharmaceuticals International Gmbh | Solid state forms of lotilaner and process for preparation thereof |
| WO2023203066A1 (en) | 2022-04-21 | 2023-10-26 | Basf Se | Synergistic action as nitrification inhibitors of dcd oligomers with alkoxypyrazole and its oligomers |
| WO2024028243A1 (en) | 2022-08-02 | 2024-02-08 | Basf Se | Pyrazolo pesticidal compounds |
| CN115504971A (en) * | 2022-11-09 | 2022-12-23 | 世华合创生物技术开发(山东)有限公司 | Synthetic method of lotilanide |
| CN119798241A (en) * | 2025-03-17 | 2025-04-11 | 北京元延医药科技股份有限公司 | A simple method for preparing high-purity lotirana |
| CN119798241B (en) * | 2025-03-17 | 2025-06-06 | 北京元延医药科技股份有限公司 | Method for simply and conveniently preparing high-purity rotigorskite |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2014090918A1 (en) | Process for the enantiomeric enrichment of diaryloxazoline derivatives | |
| CN114698375B (en) | Process for preparing 2-cyanoethyl (4S) -4- (4-cyano-2-methoxyphenyl) -5-ethoxy-2, 8-dimethyl-1, 4-dihydro-1, 6-naphthyridine-3-carboxylate | |
| TW200936574A (en) | New process | |
| JP5654462B2 (en) | Process for producing crystalline polymorph of 2- (3-cyano-4-isobutyloxyphenyl) -4-methyl-5-thiazolecarboxylic acid by a poor solvent addition method | |
| WO2011042827A1 (en) | Process for the manufacture of enantiomerically pure antifungal azoles as ravuconazole and isavuconazole | |
| JP6911033B2 (en) | Chiral division method of N- [4- (1-aminoethyl) -phenyl] -sulfonamide derivative using polar aprotic solvent | |
| CN106660949A (en) | Chiral resolution method of n-[4-(1-aminoethyl)-phenyl]-sulfonamide derivatives | |
| EP3207041B1 (en) | An improved process for the preparation of lurasidone hydrochloride | |
| EP1773793A2 (en) | Process for the preparation of pramipexole by chiral chromatography | |
| CN101857559A (en) | Chiral α-(trichloromethyl)amine compound and preparation method thereof | |
| WO2012171248A1 (en) | 1,3-oxazolidine-2-one-like compound, preparation method and uses thereof | |
| CN107098866B (en) | The method for splitting of drug Lesinurad axial chirality enantiomer | |
| CN106967057A (en) | A kind of efficient preparation technology of Aprepitant | |
| JP2024167399A (en) | Method for preparing (5S)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-2-methyl-benzoic acid | |
| CN113242854B (en) | Preparation method of (5S)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4H-isoxazol-3-yl]-2-methylbenzoic acid | |
| JP2024530297A (en) | Process for preparing optically active isoxazoline compounds | |
| WO2020127944A1 (en) | Method for racemisation of (5r)-4-[5-(3,5-dichlorophenyl)-5-(trifluoromethyl)-4h-isoxazol-3-yl]-2-methyl-benzoic acid | |
| CN103396367B (en) | Pyrazole bisamide compound containing mandelic acid group, preparation method and use | |
| BR112021012221B1 (en) | METHODS FOR PREPARING (S)-4-[5-(3,5-DICHLOROPHENYL)-5-(TRIFLUOROMETHYL)-4H-ISOXAZOL-3-YL]-2-METHYL-BENZOIC ACID AND 4-[(5S)-5 -(3,5-DICHLOROPHENYL)-4,5-DIHYDRO-5-TRIFLUOROMETHYL-1,2-OXAZOL-3-YL]-N-[2-OXO-2-(2,2,2-TRIFLUOROETHYLAMINO)ETHYL] - O-TOLUAMIDE | |
| JPH10237038A (en) | Purification of nitroguanidine derivative | |
| WO2014139087A1 (en) | Racemization method of chiral 2-[(4,6-dimethylpyrimidine-2-yl)oxy]-3-methoxy-3,3-diphenyl propionic acid | |
| WO2015162622A1 (en) | Process for preparation of linezolid | |
| CN107021910A (en) | The method for preparing S-oxiracetam crystal formation II |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 13805853 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 13805853 Country of ref document: EP Kind code of ref document: A1 |