WO2013003977A1 - Compound of 2,5-disubstituted-3-nitroimino-1,2,4-triazoline and preparation method and use as pesticide thereof - Google Patents
Compound of 2,5-disubstituted-3-nitroimino-1,2,4-triazoline and preparation method and use as pesticide thereof Download PDFInfo
- Publication number
- WO2013003977A1 WO2013003977A1 PCT/CN2011/001096 CN2011001096W WO2013003977A1 WO 2013003977 A1 WO2013003977 A1 WO 2013003977A1 CN 2011001096 W CN2011001096 W CN 2011001096W WO 2013003977 A1 WO2013003977 A1 WO 2013003977A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- group
- substituted
- formula
- unsubstituted
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D249/00—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms
- C07D249/02—Heterocyclic compounds containing five-membered rings having three nitrogen atoms as the only ring hetero atoms not condensed with other rings
- C07D249/08—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
- C07D249/10—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles with hetero atoms or with carbon atoms having three bonds to hetero atoms with at the most one bond to halogen, e.g. ester or nitrile radicals, directly attached to ring carbon atoms
- C07D249/14—Nitrogen atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N51/00—Biocides, pest repellants or attractants, or plant growth regulators containing organic compounds having the sequences of atoms O—N—S, X—O—S, N—N—S, O—N—N or O-halogen, regardless of the number of bonds each atom has and with no atom of these sequences forming part of a heterocyclic ring
Definitions
- the present invention relates to 2,5-disubstituted-3-niminoimido-1,2,4-triazoline compounds and precursors thereof, nitroaminoamino compounds, processes for their preparation, and their use as insecticides Application of the agent.
- the imidacloprid developed by Bayer in the 1980s is an outstanding representative of neonicotinoid insecticides. It has excellent control effects on sucking pests, certain chewable pests, soil pests and pet fleas. A wide range of applications. Subsequently, it was found that dehydropyridinium, a metabolite of imidacloprid in the soil, has stronger insecticidal activity than it, which has caused great interest. However, the synthesis of dehydropyrimidin is not easy, and it has not been able to be industrialized on a large scale.
- the object of the present invention is to provide an insecticidal active compound 2,5-disubstituted-3-niminoimido-1,2,4-triazoline having a structure similar to dehydropyrimidin, and a precursor compound nitro group for preparing the same Aminoguanidine compounds.
- ⁇ is a C1 ⁇ C10 saturated or unsaturated aliphatic hydrocarbon group, a C1 ⁇ C10 saturated or unsaturated fluoroaliphatic hydrocarbon group, a substituted or unsubstituted phenyl group, a substituted or unsubstituted pyridyl group, a substituted or unsubstituted thiazolyl group, a substituted or Unsubstituted pyrazolyl, substituted or unsubstituted oxazolyl, substituted or unsubstituted isoxazolyl, or tetrahydrofuranyl; wherein, substituted phenyl, substituted pyridyl, substituted thiazolyl, substituted
- the substituents in the pyrazolyl, substituted oxazolyl and substituted isoxazolyl groups may be independently selected from any one or more of the following groups: halogen (specifically F, Cl, Br, I), amino group, hydroxyl
- the substituent in the phenyl group may be halogen, hydroxy, amino, C1 to C5 alkyl, C1 to C5 alkoxy, C1 to C5 fluoroalkyl or C1 to C5 fluoroalkoxy, aryl (e.g., phenyl, pyridyl, imidazolyl, oxazolyl, thiazolyl), aryloxy (e.g., phenoxy, pyridyloxy), etc.
- the substituent in the substituted pyridyl group may be halogen, C1 a C5 al
- the substituent in the substituted furyl group may be a halogen, a trifluoromethyl group or the like, and the substituent in the substituted naphthyl group may be a C1 to C5 alkyl group, a hydroxyl group, a nitro group, a halogen group, an amino group or the like.
- Preferred is (: 1 to (: 10 unsaturated aliphatic hydrocarbon group, halopy pyridyl group, halothiazolyl group, or tetrahydrofuranyl group; preferably R 2 is hydrogen, substituted phenyl group, or C1 to C7 saturated or unsaturated aliphatic hydrocarbon group) .
- ⁇ is vinyl, ethynyl, chloropyridyl, chlorothiazolyl, tetrahydrofuran-3-yl; more preferably R 2 is hydrogen, halophenyl or C3 to C7 saturated or unsaturated aliphatic hydrocarbon.
- the saturated or unsaturated aliphatic hydrocarbon group in the present invention may be a straight chain or a branched chain.
- a method for preparing a 2,5-disubstituted-3-niminoimido-1,2,4-triazoline of the formula I and a nitroamino hydrazine compound of the formula VI includes the following steps:
- nitroguanidine reacts with hydrazine hydrate to form nitroaminoguanidine of formula II;
- ⁇ is the same as in the formula I, X is Cl, Br, I, OTos (p-toluenesulfonyloxy) or OTf (trifluoromethanesulfonyloxy);
- reaction in the step 1) is carried out in a solvent
- the solvent may be water
- reaction temperature of the reaction is 45-70 ° C
- the molar ratio of nitroguanidine to hydrazine hydrate can be 1: (1-1.5).
- the reaction in the step 2) is carried out in a solvent, and the solvent may be anhydrous ethanol or methanol; the reaction temperature of the reaction may be 50-80 ° C; the acid used in the reaction may specifically be acetic acid or Toluenesulfonic acid.
- Step 2) The molar ratio of the nitroaminopurine to the carbonyl compound of the formula III in the reaction may be 1: (1-2).
- the reaction in the step 3) is carried out in a solvent, which may be DMF (; dimethylformamide) or DME (; dimethylacetamide;); the reaction temperature may be 0-50 ° C; The base used in the reaction may specifically be sodium hydride, sodium ethoxide, sodium methoxide or sodium amide.
- Step 3) The molar ratio of the compound of the formula IV to the compound of the formula V may be 1: (1.2-2.5).
- the reaction in the step 4) is carried out in a solvent, which may be water, methanol, ethanol, acetonitrile; the temperature of the reaction is 0-100 ° C; the weakly acidic medium may be dilute hydrochloric acid, dilute sulfuric acid, Phosphoric acid or acetic acid; the weakly basic medium may be an aqueous solution of sodium carbonate, an aqueous solution of sodium acetate, dilute sodium hydroxide, pyridine, or triethylamine.
- a solvent which may be water, methanol, ethanol, acetonitrile
- the temperature of the reaction is 0-100 ° C
- the weakly acidic medium may be dilute hydrochloric acid, dilute sulfuric acid, Phosphoric acid or acetic acid
- the weakly basic medium may be an aqueous solution of sodium carbonate, an aqueous solution of sodium acetate, dilute sodium hydroxide, pyridine, or triethylamine.
- Another object of the present invention is to provide a 2,5-disubstituted-1,2,4-triazoline-3-nitroimine compound of the formula I and a nitroamino hydrazine compound of the formula VI the use of.
- the 2,5-disubstituted-1,2,4-triazoline-3-niminoimine compound represented by the structural formula of the formula I provided by the present invention and the nitroaminoamino compound represented by the formula VI The use of the compound of formula I and VI or a pharmaceutically acceptable salt thereof or a pharmaceutical composition containing any of them Application in plant pesticides.
- Still another object of the present invention is to provide two plant insecticides and their formulations.
- the plant insecticidal drug or preparation provided by the invention wherein the active ingredient is a 2,5-disubstituted-1,2,4-triazoline-3-niminoimine compound represented by the formula I; A nitroaminoguanidine compound of the formula VI or a pharmaceutically acceptable salt thereof.
- the mass percentage of the active ingredient in the plant insecticide or preparation is from 0.01% to 99.99%.
- the insecticidal formulation can be processed into any acceptable dosage form as desired.
- it may be a suspending agent, an emulsion, an aerosol, a wettable powder, an emulsifiable concentrate, a granule, or the like.
- suspending agent The active ingredient content in the commonly used formula is 5%-35%.
- the water, the main drug, the water dispersing agent, the suspending agent and the antifreezing agent are added to the sander and ground to prepare a suspending agent.
- Preparation of wettable powder According to the formulation requirements, the original drug, various surfactants and solid diluents are thoroughly mixed, and after superfine pulverization, a predetermined content of the wettable powder product is obtained. In order to prepare a wettable powder suitable for spraying, a mixture of the original drug and a finely divided solid powder such as a clay, an inorganic silicate, a carbonate, and a wetting agent, a binder and/or a dispersing agent may also be used.
- a finely divided solid powder such as a clay, an inorganic silicate, a carbonate, and a wetting agent, a binder and/or a dispersing agent may also be used.
- emulsifiable concentrate It is prepared by dissolving the active ingredient in an organic solvent according to the formulation requirements, adding emulsifier and other auxiliary agents.
- the solvent may be toluene, xylene, methanol, etc., and if necessary, a cosolvent; other additives, including stabilizers, penetrants, and corrosion inhibitors.
- the compounds of formula I and formula VI provided by the present invention and insecticidal drugs or preparations using the same as active ingredients can control and eliminate a wide range of pests, including sucking insects (Sucking insects), mites (; Biting insects) ) and other plant pests, storage of cereal pests and health pests that cause health hazards.
- Homopteran pests include pests of the family Polygonaceae, Fanicidae, Polygonaceae, Hibiscus, Aphididae and Polygonaceae.
- the pests of the genus Polygonaceae may be cotton aphid, soybean meal, peach aphid, peach meal, radish or cabbage aphid; the pests of the plant may be brown planthopper or rice planthopper; the pests of the whitefly may be beech white;
- the pests of the genus Polygonaceae can be black-tailed spider mites; the pests of the genus Polygonaceae can be genus.
- Lepidopteran pests include the Noctuidae and the Plutella xylostella pests.
- the pest of the Noctuidae can be Beet armyworm, Spodoptera litura or Helicoverpa armigera; the pest of the genus Hydrangea can be Plutella xylostella.
- the true eyeworm includes the leafhopper pest.
- the leaf worm can specifically be cinnabar leafhopper.
- the plant insecticidal drugs or preparations provided by the present invention can be used for controlling plant pests. Especially for sucking pests, scraping mouthparts pests, such as various types of aphids, flying locusts, leaf mites, leaf mites, white mites, scorpion horses have special effects, and are highly effective against cotton bollworm and beet armyworm.
- the method for controlling plant pests provided by the present invention is to apply the plant insecticidal drug or preparation provided by the present invention to plant leaves and/or fruits and/or seeds, and the leaves and/or fruits and/or seeds of the plants are being In an environment in which growth or anticipation is to be grown; the active ingredient of the plant insecticide or formulation is applied at a concentration of from 1 to 600 mg/L, preferably at a concentration of from 3 to 50 mg/L.
- the structure characterization data is as follows: 1H NMR (CDCl 3 , 5 ppm): 0.93 (t, 3H), 1.28-1.38 (m, 2H) ; 1.48-1.58 (m, 2H), 2.36-2.42 (m, 2H), 5.25 ( s, 2H), 7.39 (t, lH), 7.42 (s, lH), 7.42 (brs, lH), 8.99 (brs, lH)
- Example 3 the insecticidal activity test of the compound of formula I and formula VI provided by the present invention
- Aphids belong to Homoptera pests and have a sucking mouth device, which is a common pest of common crops.
- ⁇ ⁇ ⁇ (radish ⁇ ) (z ⁇ op s er /m/) is the test object, tested by dipping method.
- the peach aphid was collected from the cabbage field in Haidian District, Beijing.
- the peach aphid was collected from Taoshu, Haidian District, Beijing.
- the cotton aphid was collected from Mudu Tree, Haidian District, Beijing.
- the vegetable tube was collected from the cabbage field in Haidian District, Beijing.
- the 3 day old nymph was measured.
- Triton X-100 The mass percentage of 0.5% mother liquor is used to make a mass percentage of 0.5% mother liquor, and the mass content is 0.1% Triton X-100.
- the aqueous solution is formulated into a measuring solution having a concentration of 0.06% (i.e., 600 ppm) (the solution can be diluted to the desired concentration when the concentration is low;).
- the criteria for death judgment are: Lightly touching the worm, and the individual who cannot crawl normally is regarded as a dead individual.
- Corrected mortality (%) (sample mortality - blank control mortality) I (1-blank control mortality) ⁇ 100%.
- the cinnabar is a female cockroach i Tetranychus cinnabarinus Boisduva, taken from the singular beta cotton field.
- the insecticidal activity is determined by the dipping method.
- the bean leaves transferred to the cinnabar leafhopper are immersed in the liquid for 5 s. After drying, the number of insects is recorded in a petri dish with moisturizing filter paper, and the petiole is moisturized.
- the criteria for death are: Light-touched worms, individuals who cannot crawl normally are considered dead individuals.
- Helicoverpa armigera is a pest of Lepidoptera, and has a chewing mouth.
- the sample of the compound is weighed in a one-tenth of a 10,000-week volume, and the mother liquor is prepared with dimethylformamide (DMF), and then 0.1% Triton
- Operation process Clean the cotton leaf, use a puncher to cut the clean leek leaf into a 2cm diameter leaf dish, soak it in the liquid for 10s, dry it, put the leaf dish into the 10-hole test box, and connect the cotton bollworm 2nd instar larvae, 1 per hole, add plastic wrap, cover and put in (27 ⁇ 1) °C light incubator. Check the results after 48 hours. Death occurs when the needle touches the insect body and the reaction is abnormal or non-responsive.
- DMF dimethylformamide
- Operation process Wash the leek leaves and use a puncher to cut the clean leek leaves into diameters.
- WP wettable powder
- SC suspending agent
- the present invention provides a 2,5-disubstituted-3-niminoimido-1,2,4-triazoline compound of the formula I and a precursor compound thereof, and a nitro group of the formula VI.
- Preparation methods and applications of terpenoids The above compounds are extensively investigated and rationally designed by the inventors of the present invention, and The screening of the quantitative compound has a high insecticidal activity, and the preparation method is simple, and provides a preferred process route, which has good safety and low cost, and the practical application value of the compound is greatly improved.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Dentistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Plant Pathology (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Agronomy & Crop Science (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
2,5-二取代 -3-硝亚胺基 -1,2,4-三唑啉类化合物及其制备方法与其作为 杀虫剂的应用 技术领域 2,5-disubstituted -3-nitroimino-1,2,4-triazoline compound and preparation method thereof and application thereof as insecticide
本发明涉及 2,5-二取代 -3-硝亚胺基 -1,2,4-三唑啉类化合物及其前体硝基 缩氨基胍类化合物与它们的制备方法, 以及其作为杀虫剂的应用。 The present invention relates to 2,5-disubstituted-3-niminoimido-1,2,4-triazoline compounds and precursors thereof, nitroaminoamino compounds, processes for their preparation, and their use as insecticides Application of the agent.
背景技术 Background technique
拜耳公司于 20世纪 80年代开发的吡虫啉是新烟碱类杀虫剂的杰出代 表, 对吮吸式害虫、 某些咀嚼性害虫、 土壤害虫及宠物跳蚤等具有优异的 防效, 在农业生产中取得了广泛的应用。 随后人们发现吡虫啉在土壤中的 代谢产物脱氢吡虫啉具有比其更强的杀虫活性, 从而引起了人们极大的兴 趣, 但脱氢吡虫啉合成不易, 目前尚无法大规模工业化生产。 The imidacloprid developed by Bayer in the 1980s is an outstanding representative of neonicotinoid insecticides. It has excellent control effects on sucking pests, certain chewable pests, soil pests and pet fleas. A wide range of applications. Subsequently, it was found that dehydropyridinium, a metabolite of imidacloprid in the soil, has stronger insecticidal activity than it, which has caused great interest. However, the synthesis of dehydropyrimidin is not easy, and it has not been able to be industrialized on a large scale.
发明公开 Invention disclosure
本发明的目的是提供结构与脱氢吡虫啉类似的杀虫活性化合物 2,5-二取 代 -3-硝亚胺基 -1,2,4-三唑啉, 以及制备它们的前体化合物硝基缩氨基胍类 化合物。 The object of the present invention is to provide an insecticidal active compound 2,5-disubstituted-3-niminoimido-1,2,4-triazoline having a structure similar to dehydropyrimidin, and a precursor compound nitro group for preparing the same Aminoguanidine compounds.
本发明所提供的 2,5-二取代 -3-硝亚胺基 -1,2,4-三唑啉类化合物的结构 通式如式 I所示, 其前体化合物硝基缩氨基胍类化合物的结构通式如式 VI 所示: The structural formula of the 2,5-disubstituted-3-niminoimido-1,2,4-triazoline compound provided by the present invention is as shown in Formula I, and the precursor compound thereof is nitroamino hydrazine. The structural formula of the compound is as shown in formula VI:
N'N02 N'N0 2
NN02 NN0 2
NH X R NH X R
N Η2Ν « 2 N Η 2 Ν « 2
Ri H Ri H
(;式 I) (;式 VI) (; Formula I) (; Formula VI)
式 I和式 VI中: In formula I and formula VI:
!^为 C1〜C10饱和或不饱和脂肪烃基、 C1〜C10饱和或不饱和氟代脂 肪烃基、 取代或未取代的苯基、 取代或未取代的吡啶基、 取代或未取代的 噻唑基、 取代或未取代的吡唑基、 取代或未取代的恶唑基、 取代或未取代 的异恶唑基、 或四氢呋喃基; 其中, 所述取代的苯基、 取代的吡啶基、 取 代的噻唑基、 取代的吡唑基、 取代的恶唑基和取代的异恶唑基中的取代基 可独立的选自下述基团中的任意一种或几种: 卤素 (具体可为 F、 Cl、 Br、 I) 、 氨基、 羟基、 C1〜C5的烷基、 C1〜C5的烷氧基、 C1〜C5的氟 代烷基和 C1〜C5的氟代烷氧基等; ! ^ is a C1~C10 saturated or unsaturated aliphatic hydrocarbon group, a C1~C10 saturated or unsaturated fluoroaliphatic hydrocarbon group, a substituted or unsubstituted phenyl group, a substituted or unsubstituted pyridyl group, a substituted or unsubstituted thiazolyl group, a substituted or Unsubstituted pyrazolyl, substituted or unsubstituted oxazolyl, substituted or unsubstituted isoxazolyl, or tetrahydrofuranyl; wherein, substituted phenyl, substituted pyridyl, substituted thiazolyl, substituted The substituents in the pyrazolyl, substituted oxazolyl and substituted isoxazolyl groups may be independently selected from any one or more of the following groups: halogen (specifically F, Cl, Br, I), amino group, hydroxyl group, C1~C5 alkyl group, C1~C5 alkoxy group, C1~C5 fluorine Alkanoyl group and a fluoroalkoxy group of C1 to C5;
为氢、 C1〜C10饱和或不饱和脂肪烃基、 取代或未取代的苯基、 取 代或未取代的吡啶基、 取代或未取代的呋喃基、 取代或未取代的萘基; 其 中, 所述取代苯基中的取代基可为卤素、 羟基、 氨基、 C1〜C5的烷基、 C1〜C5的烷氧基、 C1〜C5的氟代烷基或 C1〜C5的氟代烷氧基、 芳基 (如: 苯基、 吡啶基、 咪唑基、 恶唑基、 噻唑基) 、 芳氧基 (如: 苯氧 基、 吡啶氧基) 等, 所述取代吡啶基中的取代基可为卤素、 C1〜C5的烷 基、 C1〜C5的烷氧基、 C1〜C5的氟代烷基或 C1〜C5的氟代烷氧基、 芳 氧基 (如: 苯氧基、 吡啶氧基) 等, 所述取代呋喃基中的取代基可为卤 素、 三氟甲基等, 所述取代的萘基中的取代基可为 C1〜C5烷基、 羟基、 硝 基、 卤素、 氨基等。 Is a hydrogen, C1 to C10 saturated or unsaturated aliphatic hydrocarbon group, a substituted or unsubstituted phenyl group, a substituted or unsubstituted pyridyl group, a substituted or unsubstituted furyl group, a substituted or unsubstituted naphthyl group; wherein the substitution The substituent in the phenyl group may be halogen, hydroxy, amino, C1 to C5 alkyl, C1 to C5 alkoxy, C1 to C5 fluoroalkyl or C1 to C5 fluoroalkoxy, aryl (e.g., phenyl, pyridyl, imidazolyl, oxazolyl, thiazolyl), aryloxy (e.g., phenoxy, pyridyloxy), etc., the substituent in the substituted pyridyl group may be halogen, C1 a C5 alkyl group, a C1 to C5 alkoxy group, a C1 to C5 fluoroalkyl group, a C1 to C5 fluoroalkoxy group, an aryloxy group (e.g., a phenoxy group, a pyridyloxy group), etc. The substituent in the substituted furyl group may be a halogen, a trifluoromethyl group or the like, and the substituent in the substituted naphthyl group may be a C1 to C5 alkyl group, a hydroxyl group, a nitro group, a halogen group, an amino group or the like.
优选的 为(:1〜(:10不饱和脂肪烃基、 卤代吡啶基、 卤代噻唑基、 或四氢呋喃基; 优选的 R2为氢、 取代苯基、 或 C1〜C7饱和或不饱和脂肪 烃基。 Preferred is (: 1 to (: 10 unsaturated aliphatic hydrocarbon group, halopy pyridyl group, halothiazolyl group, or tetrahydrofuranyl group; preferably R 2 is hydrogen, substituted phenyl group, or C1 to C7 saturated or unsaturated aliphatic hydrocarbon group) .
更优选的!^为乙烯基、 乙炔基、 氯代吡啶基、 氯代噻唑基、 四氢呋 喃 -3-基; 更优选的 R2为氢、 卤代苯基或 C3〜C7饱和或不饱和脂肪烃基。 More preferred! ^ is vinyl, ethynyl, chloropyridyl, chlorothiazolyl, tetrahydrofuran-3-yl; more preferably R 2 is hydrogen, halophenyl or C3 to C7 saturated or unsaturated aliphatic hydrocarbon.
本发明中所述饱和或不饱和脂肪烃基可为直链或支链。 The saturated or unsaturated aliphatic hydrocarbon group in the present invention may be a straight chain or a branched chain.
本发明所提供的制备式 I所示的 2,5-二取代 -3-硝亚胺基 -1,2,4-三唑啉 和式 VI所示的硝基缩氨基胍类化合物的方法, 包括以下步骤: A method for preparing a 2,5-disubstituted-3-niminoimido-1,2,4-triazoline of the formula I and a nitroamino hydrazine compound of the formula VI, provided by the present invention, Includes the following steps:
1 ) 硝基胍与水合肼反应生成式 II所示的硝基氨基胍; 1) nitroguanidine reacts with hydrazine hydrate to form nitroaminoguanidine of formula II;
(式 II ) (Formula II)
2 ) 将式 II所示的硝基氨基胍与式 III所示的羰基化合物在酸催化作用下 反应, 生成式 IV所示的硝基缩氨基胍; 2) reacting the nitroaminopurine of the formula II with a carbonyl compound of the formula III under acid catalysis to form a nitroaminoguanidine of the formula IV;
0 NN02 0 NN0 2
人 H2N人 N'N丫 Human H 2 N human N'N丫
H R2 Η ΐ H R 2 Η ΐ
(式 III) (式 IV) (Formula III) (Formula IV)
3)将式 IV所示的化合物与式 V所示的化合物 代烃或磺酸酯;)在碱催 化作用下反应, 生成式 VI所示的化合物; NN02 3) reacting a compound of formula IV with a compound of formula V or a sulfonate; and reacting under base catalysis to form a compound of formula VI; NN0 2
H2N人 «2 H 2 N people « 2
R X ^ H R X ^ H
(式 V) (式 VI) (Formula V) (Formula VI)
4)将式 VI所示的化合物在弱酸性或弱碱性介质中反应关环, 生成式 I 结构通式所示的化合物; 4) reacting a compound represented by formula VI in a weakly acidic or weakly basic medium to form a compound of the formula I;
其中, 式 III和式 VI中 R2的定义与式 VI、 式 I中的 R2相同; Wherein formula III and formula VI wherein R 2 is as defined in Formula VI, Formula I is the same as R 2;
式 中!^的定义与式 I中相同, X为 Cl、 Br、 I、 OTos (对甲苯磺酰 氧基) 或 OTf (三氟甲磺酰氧基) ; In the middle! The definition of ^ is the same as in the formula I, X is Cl, Br, I, OTos (p-toluenesulfonyloxy) or OTf (trifluoromethanesulfonyloxy);
CH3~^G^S02°_ F3CS020— CH3 ~^G^ S02 ° _ F 3 CS0 2 0—
对甲苯磺酰氧基 三氟甲磺酸基 其中, 步骤 1) 中所述反应在溶剂中进行, 所述溶剂可为水; 所述反 应的反应温度为 45-70°C, 所述反应中硝基胍与水合肼的摩尔比可为 1: (1-1.5) 。 a p-toluenesulfonyloxytrifluoromethanesulfonate group, wherein the reaction in the step 1) is carried out in a solvent, the solvent may be water; the reaction temperature of the reaction is 45-70 ° C, in the reaction The molar ratio of nitroguanidine to hydrazine hydrate can be 1: (1-1.5).
步骤 2) 中所述反应在溶剂中进行, 所述溶剂可为无水乙醇或甲醇; 所述反应的反应温度可为 50-80°C; 所述反应中所用的酸具体可为醋酸或对 甲苯磺酸。 步骤 2) 所述反应中硝基氨基胍与式 III所示的羰基化合物的摩 尔比可为 1: (1-2) 。 The reaction in the step 2) is carried out in a solvent, and the solvent may be anhydrous ethanol or methanol; the reaction temperature of the reaction may be 50-80 ° C; the acid used in the reaction may specifically be acetic acid or Toluenesulfonic acid. Step 2) The molar ratio of the nitroaminopurine to the carbonyl compound of the formula III in the reaction may be 1: (1-2).
步骤 3) 中所述反应在溶剂中进行, 所述溶剂可为 DMF (;二甲基甲酰胺) 或 DME (;二甲基乙酰胺;); 所述反应的反应温度可为 0-50°C; 所述反应中所 用的碱具体可为氢化钠、 乙醇钠、 甲醇钠或氨基钠。 步骤 3) 中式 IV所示 化合物与式 V所示化合物的摩尔比可为 1: (1.2-2.5) 。 The reaction in the step 3) is carried out in a solvent, which may be DMF (; dimethylformamide) or DME (; dimethylacetamide;); the reaction temperature may be 0-50 ° C; The base used in the reaction may specifically be sodium hydride, sodium ethoxide, sodium methoxide or sodium amide. Step 3) The molar ratio of the compound of the formula IV to the compound of the formula V may be 1: (1.2-2.5).
步骤 4) 中所述反应在溶剂中进行, 所述溶剂可为水、 甲醇、 乙醇、 乙腈; 所述反应的温度为 0-100°C; 所述弱酸性介质可以为稀盐酸、 稀硫 酸、 磷酸或乙酸; 所述弱碱性介质可以为碳酸钠水溶液、 乙酸钠水溶液、 稀氢氧化钠、 吡啶、 三乙胺。 The reaction in the step 4) is carried out in a solvent, which may be water, methanol, ethanol, acetonitrile; the temperature of the reaction is 0-100 ° C; the weakly acidic medium may be dilute hydrochloric acid, dilute sulfuric acid, Phosphoric acid or acetic acid; the weakly basic medium may be an aqueous solution of sodium carbonate, an aqueous solution of sodium acetate, dilute sodium hydroxide, pyridine, or triethylamine.
本发明的另一个目的是提供式 I所示的 2,5-二取代 -1,2,4-三唑啉 -3-硝 亚胺类化合物以及式 VI所示的硝基缩氨基胍类化合物的用途。 Another object of the present invention is to provide a 2,5-disubstituted-1,2,4-triazoline-3-nitroimine compound of the formula I and a nitroamino hydrazine compound of the formula VI the use of.
本发明所提供的式 I结构通式所示的 2,5-二取代 -1,2,4-三唑啉 -3-硝亚 胺类化合物及式 VI所示的硝基缩氨基胍类化合物的用途是式 I及 VI所示 的化合物或其药学上可接受的盐或含有它们中任何一种的药物组合物在制 备植物杀虫剂中的应用。 The 2,5-disubstituted-1,2,4-triazoline-3-niminoimine compound represented by the structural formula of the formula I provided by the present invention and the nitroaminoamino compound represented by the formula VI The use of the compound of formula I and VI or a pharmaceutically acceptable salt thereof or a pharmaceutical composition containing any of them Application in plant pesticides.
本发明的再一个目的是提供两种植物杀虫药物以及它们的制剂。 Still another object of the present invention is to provide two plant insecticides and their formulations.
本发明所提供的植物杀虫药物或制剂, 它的活性成分为式 I结构通式 所示的 2,5-二取代 - 1,2,4-三唑啉 -3-硝亚胺类化合物及式 VI所示的硝基缩氨 基胍类化合物或其药学上可接受的盐。 The plant insecticidal drug or preparation provided by the invention, wherein the active ingredient is a 2,5-disubstituted-1,2,4-triazoline-3-niminoimine compound represented by the formula I; A nitroaminoguanidine compound of the formula VI or a pharmaceutically acceptable salt thereof.
所述植物杀虫药物或制剂中活性成分的质量百分含量为 0.01%- 99.99%。 The mass percentage of the active ingredient in the plant insecticide or preparation is from 0.01% to 99.99%.
所述杀虫制剂可以按需要加工成任何可接受的剂型。 例如可以是悬浮 剂、 乳剂、 气雾剂、 可湿性粉剂、 乳油、 颗粒剂等。 The insecticidal formulation can be processed into any acceptable dosage form as desired. For example, it may be a suspending agent, an emulsion, an aerosol, a wettable powder, an emulsifiable concentrate, a granule, or the like.
剂型的配制方法举例如下: Examples of preparation methods for the dosage form are as follows:
悬浮剂的配制: 常用配方中活性组分含量为 5%-35%。 以水为介质, 将原药、 水分散剂、 助悬剂和抗冻剂等加入磨砂机中, 进行研磨, 制成悬 浮剂。 Preparation of suspending agent: The active ingredient content in the commonly used formula is 5%-35%. The water, the main drug, the water dispersing agent, the suspending agent and the antifreezing agent are added to the sander and ground to prepare a suspending agent.
可湿性粉剂的配制: 按配方要求, 将原药、 各种表面活性剂及固体稀 释剂等充分混合, 经超细粉碎后, 即得到预定含量的可湿性粉剂产品。 为 制备适于喷洒用的可湿性粉剂, 也可将原药和研细的固体粉末如粘土、 无 机硅酸盐、 碳酸盐以及润湿剂、 粘合剂和 /或分散剂组成混合物。 Preparation of wettable powder: According to the formulation requirements, the original drug, various surfactants and solid diluents are thoroughly mixed, and after superfine pulverization, a predetermined content of the wettable powder product is obtained. In order to prepare a wettable powder suitable for spraying, a mixture of the original drug and a finely divided solid powder such as a clay, an inorganic silicate, a carbonate, and a wetting agent, a binder and/or a dispersing agent may also be used.
乳油的配制: 按配方要求将有效成分溶于有机溶剂中, 并添加乳化剂 和其它助剂加工制成。 溶剂可采用甲苯、 二甲苯、 甲醇等, 在需要时也含 有共溶剂; 其它助剂, 包括稳定剂、 渗透剂和腐蚀抑制剂等。 Preparation of emulsifiable concentrate: It is prepared by dissolving the active ingredient in an organic solvent according to the formulation requirements, adding emulsifier and other auxiliary agents. The solvent may be toluene, xylene, methanol, etc., and if necessary, a cosolvent; other additives, including stabilizers, penetrants, and corrosion inhibitors.
本发明所提供的式 I及式 VI所示化合物以及以它们为活性成分的杀虫 药物或制剂能控制和消灭广泛的害虫, 包括吸式虫 (; Sucking insects) , 剌式 虫 (; Biting insects)和其他植物害虫, 储藏谷类的害虫和引起健康危害的卫生 害虫等。 The compounds of formula I and formula VI provided by the present invention and insecticidal drugs or preparations using the same as active ingredients can control and eliminate a wide range of pests, including sucking insects (Sucking insects), mites (; Biting insects) ) and other plant pests, storage of cereal pests and health pests that cause health hazards.
所述害虫举例如下: Examples of the pests are as follows:
同翅目害虫包括蚜科、 粉虱科、 飞虱科、 木虱科、 叶蝉科和蚧科的害 虫。 蚜科害虫具体可为棉蚜、 豆蚜、 桃蚜、 桃粉蚜、 萝卜蚜或甘蓝蚜虫; 飞虱科害虫具体可为褐飞虱或稻飞虱; 粉虱科害虫具体可为烟粉虱; 叶蝉 科害虫具体可为黑尾叶蝉; 蚧科害虫具体可为矢尖蚧。 Homopteran pests include pests of the family Polygonaceae, Fanicidae, Polygonaceae, Hibiscus, Aphididae and Polygonaceae. The pests of the genus Polygonaceae may be cotton aphid, soybean meal, peach aphid, peach meal, radish or cabbage aphid; the pests of the plant may be brown planthopper or rice planthopper; the pests of the whitefly may be beech white; The pests of the genus Polygonaceae can be black-tailed spider mites; the pests of the genus Polygonaceae can be genus.
鳞翅目害虫包括夜蛾科和菜蛾科害虫。 夜蛾科害虫具体可为甜菜夜 蛾、 斜纹夜蛾或棉铃虫; 菜蛾科害虫具体可为小菜蛾。 真螨目害虫包括叶螨科害虫。 叶螨科害虫具体可为朱砂叶螨。 Lepidopteran pests include the Noctuidae and the Plutella xylostella pests. The pest of the Noctuidae can be Beet armyworm, Spodoptera litura or Helicoverpa armigera; the pest of the genus Hydrangea can be Plutella xylostella. The true eyeworm includes the leafhopper pest. The leaf worm can specifically be cinnabar leafhopper.
本发明所提供的植物杀虫药物或制剂可用于防治植物害虫。 尤其对剌 吸式害虫、 刮吸式口器害虫, 如各类蚜虫、 飞虱、 叶蝉、 叶螨、 粉虱、 蓟 马有特效, 对棉铃虫、 甜菜夜蛾高效。 The plant insecticidal drugs or preparations provided by the present invention can be used for controlling plant pests. Especially for sucking pests, scraping mouthparts pests, such as various types of aphids, flying locusts, leaf mites, leaf mites, white mites, scorpion horses have special effects, and are highly effective against cotton bollworm and beet armyworm.
本发明再一个目的是提供一种防治植物害虫的方法。 It is still another object of the present invention to provide a method of controlling plant pests.
本发明所提供的防治植物害虫的方法, 是将本发明所提供的植物杀虫 药物或制剂施用在植物叶子和 /或果实和 /或种子上, 以及植物叶子和 /或果 实和 /或种子正在生长或预期要生长的环境中; 所述植物杀虫药物或制剂的 活性成分的施用浓度为 l-600mg/L, 优选施用浓度为 3-50 mg/L。 The method for controlling plant pests provided by the present invention is to apply the plant insecticidal drug or preparation provided by the present invention to plant leaves and/or fruits and/or seeds, and the leaves and/or fruits and/or seeds of the plants are being In an environment in which growth or anticipation is to be grown; the active ingredient of the plant insecticide or formulation is applied at a concentration of from 1 to 600 mg/L, preferably at a concentration of from 3 to 50 mg/L.
实施发明的最佳方式 The best way to implement the invention
本发明 2,5-二取代 -3-硝亚胺基 -1,2,4-三唑啉类化合物和硝基缩氨基胍 类化合物可通过如下的反应步骤合成: The 2,5-disubstituted 3-nitroiminyl-1,2,4-triazoline compound and the nitroaminoamino compound of the present invention can be synthesized by the following reaction steps:
下面结合具体实施例, 进一步阐述本发明。 应理解, 这些实施例仅用 于说明本发明而不用于限制本发明的范围。 下列实施例中未注明具体实施 条件的实验方法, 通常按照常规条件, 或按照制造厂商所建议的条件。 除 非另外说明, 否则百分比和份数按质量计算。 The invention is further illustrated below in conjunction with specific embodiments. It is to be understood that the examples are not intended to limit the scope of the invention. The experimental methods in the following examples which are not specified in the specific examples are usually carried out according to conventional conditions or according to the conditions recommended by the manufacturer. The percentages and parts are calculated by mass unless otherwise stated.
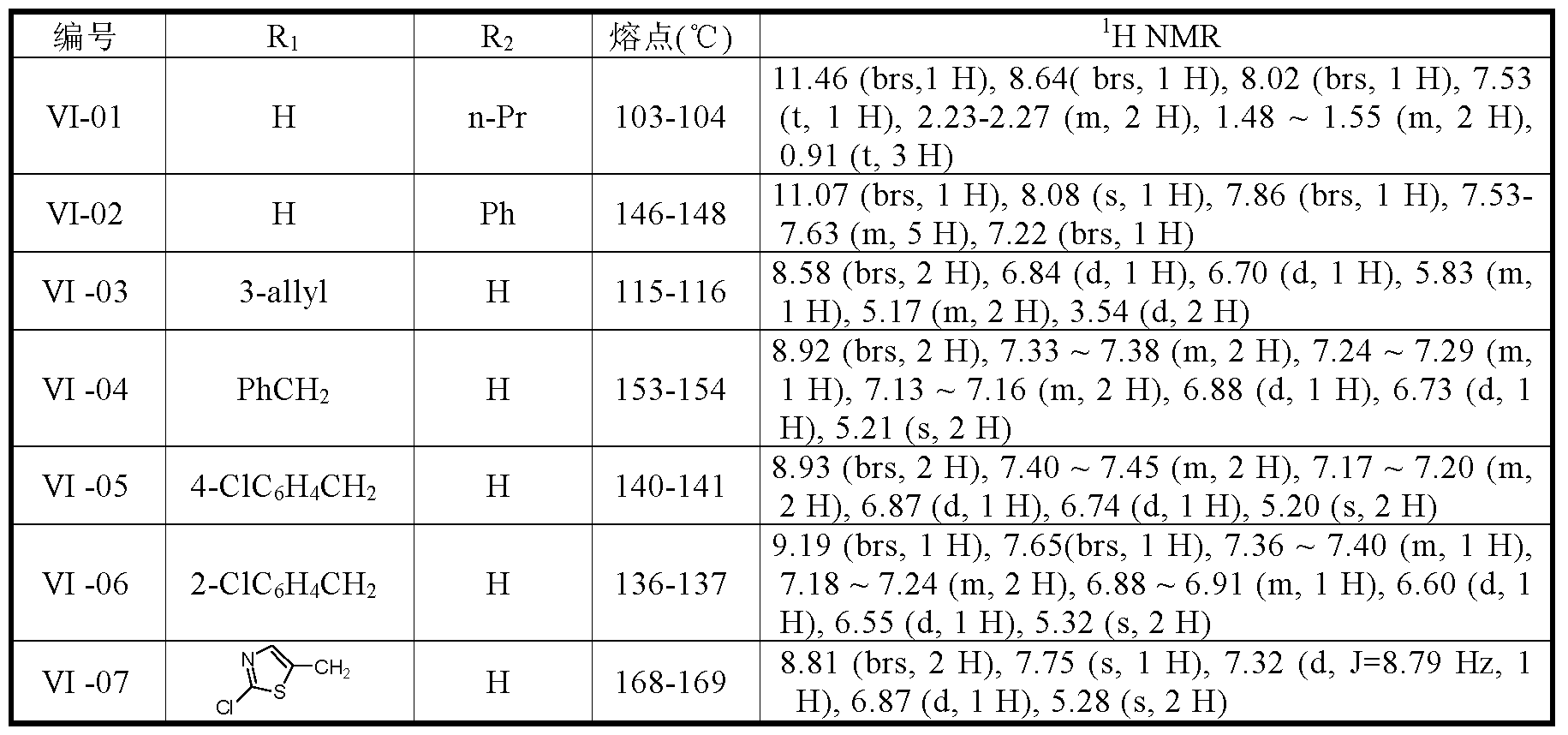
下面以 2-(2-氯吡啶 -5-甲基) -3-硝亚胺基 -1,2,4-三唑啉和 2-(2-氯噻唑 -5- 甲基) -5-丁基 -3-硝亚胺基 -1,2,4-三唑啉为例, 阐明本发明所提供的式 ( I ) 所示 2,5-二取代 -3-硝亚胺基 -1,2,4-三唑啉类化合物的制备方法。 2-(2-chloropyridine-5-methyl)-3-nitroimino-1,2,4-triazoline and 2-(2-chlorothiazol-5-methyl)-5-butyl The exemplified by the present invention, the 2,5-disubstituted-3-niminoimido-1,2 represented by the formula (I) provided by the present invention is exemplified. A method for preparing a 4-triazoline compound.
实施例 1、 2-(2-氯吡啶 -5-甲基) -3-硝亚胺基 -1,2,4-三唑啉 (式 I中 R1为 2-氯吡啶 -5-基, R2为氢)的合成: Example 1. 2-(2-Chloropyridin-5-methyl)-3-nitroimino-1,2,4-triazoline (R1 in the formula I is 2-chloropyridin-5-yl, R2 Synthesis of hydrogen):
(l) N'-硝基氨基胍的合成 (l) Synthesis of N'-nitroaminopurine
于 250mL三口瓶中, 依次投入硝基胍 5.0g (0.048mol)和水 70mL。 磁 搅拌下加热至 55 °C时, 从滴液漏斗中缓慢滴加质量百分含量为 85%的水合 肼的水溶液 (其中, 加入水合肼的质量为 3.5g (0.059mol) ) 。 保持物料温 度在 55〜60°C之间继续反应 20分钟。 当物料变为橙黄色澄清液时, 用冰 水浴快速冷却, 并缓慢滴加浓盐酸 (质量百分含量为 37%) 约 6mL至 pH 值为 5〜6; 继续冷却至 2〜3 °C并保持 1小时。 减压抽滤, 用少量冰水洗 涤, 用热水重结晶, 得浅黄色粉未 (Ν'-硝基氨基胍) 2.74g, 收率 48%, 熔点 191〜192°C。 In a 250 mL three-necked flask, 5.0 g (0.048 mol) of nitroguanidine and 70 mL of water were successively introduced. When heated to 55 ° C under magnetic stirring, an aqueous solution of hydrazine hydrate having a mass percentage of 85% was slowly added dropwise from the dropping funnel (wherein the mass of hydrazine hydrate added was 3.5 g (0.059 mol)). The reaction was continued for 20 minutes while maintaining the temperature of the material between 55 and 60 °C. When the material turns into an orange-yellow clear liquid, use ice The water bath was rapidly cooled, and concentrated hydrochloric acid (37% by mass) was slowly added dropwise to about 6 mL to a pH of 5 to 6; cooling was continued to 2 to 3 ° C for 1 hour. The mixture was filtered under reduced pressure, washed with a small portion of ice water, and then recrystallized from hot water to give a pale yellow powder (yield: y-nitroamino hydrazine) 2.74 g, yield 48%, melting point 191 to 192 °C.
结构表征数据如下: The structural characterization data is as follows:
1H NMR(DMSO-d6, 5ppm): 4.69(s,2H,-NHNH2),7.56(s, lH,-NHNH2), 8.27(s, lH,-NHNO2),9.33(s, lH,C=Ng)。 1H NMR (DMSO-d 6 , 5 ppm): 4.69 (s, 2H, -NHNH 2 ), 7.56 (s, lH, -NHNH 2 ), 8.27 (s, lH, -NHNO 2 ), 9.33 (s, lH, C =Ng).
(2) Ν'-硝基甲醛缩氨基胍的合成 (2) Synthesis of Ν'-nitroform acetal
于 250ml三口瓶中, 依次投入 N,-硝基氨基胍 2.0g(0.017mol), 无水甲 醇 30mL和冰醋酸 0.25mL, 磁力搅拌下加热至 50°C, 从恒压滴液漏斗中缓 慢滴加 40%甲醛 1.6g ( 0.02mol) , 滴加完毕, 加热至回流, 并回流 1.0小 时。 降温, 冷却, 有大量白色晶体析出, 得 2.1g, 收率 95%, 直接用于下 一步反应。 In a 250 ml three-necked flask, 2.0 g (0.017 mol) of N-nitroaminopurine, 30 mL of anhydrous methanol and 0.25 mL of glacial acetic acid were sequentially added, and heated to 50 ° C under magnetic stirring, and slowly dripped from a constant pressure dropping funnel. Add 40% formaldehyde 1.6 g (0.02 mol), add dropwise, heat to reflux, and reflux for 1.0 hour. The temperature was lowered, cooled, and a large amount of white crystals were precipitated to obtain 2.1 g, and the yield was 95%, which was directly used for the next reaction.
(3) Ν'-硝基 -N-(2-氯吡啶 -5-甲基)甲醛缩氨基胍的合成 (3) Synthesis of Ν'-nitro-N-(2-chloropyridine-5-methyl)formal amide
于 250mL 三口瓶中, 依次投入 N,-硝基甲醛缩氨基胍 2.6g (0.02 mol) , 无水 DMF 40mL和少量 KI, 安装干燥管, 开动磁搅拌, 并用冰水 浴降温。 降温至 10°C以下, 分 3 批加入质量含量为 70%的氢化钠 1.37 g (0.04mol), 并反应 1小时。 从滴液漏斗中缓慢滴加 2-氯 -5-氯甲基吡啶 4.8g (0.03mol)和无水 DMF 20mL的溶液。 滴加完毕, 撤去冰水浴, 自然升温至 室温。 在室温下反应 16小时后, 缓慢加入 200mL水中, 有深红色粘稠状 化合物出现, 柱层析 (石油醚: 乙酸乙酯 =2:3, v/v) , 得白色固体 (Ν'-硝 基 _Ν- ( 2-氯 -吡啶 -5-甲基) 甲醛缩氨基胍) l. lg, 收率 21%, 熔点 124〜 126 °C。 In a 250 mL three-necked flask, 2.6 g (0.02 mol) of N-nitroformaldehyde acetaminophen, 40 mL of anhydrous DMF and a small amount of KI were placed in this order, and a drying tube was installed, magnetic stirring was started, and the temperature was lowered with an ice water bath. The temperature was lowered to below 10 ° C, and 1.37 g (0.04 mol) of sodium hydride having a mass content of 70% was added in three portions, and reacted for 1 hour. A solution of 4.8 g (0.03 mol) of 2-chloro-5-chloromethylpyridine and 20 mL of anhydrous DMF was slowly added dropwise from the dropping funnel. After the addition is completed, the ice water bath is removed and the temperature is naturally raised to room temperature. After reacting for 16 hours at room temperature, slowly add 200 mL of water, a dark red viscous compound appeared, and column chromatography (petroleum ether: ethyl acetate = 2:3, v/v) gave a white solid (Ν'- Base _ Ν - (2-chloro-pyridin-5-methyl) formaldehyde acetaminophen) l. lg, yield 21%, m.p. 124~ 126 °C.
结构表征数据如下: The structural characterization data is as follows:
1HNMR(DMSO-d6,5ppm):6.88(d, 1H),7.13(d, lH),8.41(brs, lH),8.74(brs,1H NMR (DMSO-d 6 , 5 ppm): 6.88 (d, 1H), 7.13 (d, lH), 8.41 (brs, lH), 8.74 (brs,
1H), 11.67(S, 1H) 1H), 11.67(S, 1H)
(4) 2-(2-氯吡啶 -5-甲基) -3-硝亚胺基 -1,2,4-三唑啉的合成 (4) Synthesis of 2-(2-chloropyridine-5-methyl)-3-nitroimino--1,2,4-triazoline
于 250mL三口瓶中, 依次投入 3.0gN,-硝基 -N- (2-氯吡啶 -5-甲基) 甲 醛缩氨基胍和 80mL 5mol/L的醋酸水溶液, 加热至回流, 并保持 5小时。 冷却, 有红色油状物析出, 柱层析 (石油醚: 乙酸乙酯 =1 : 1, v/v) , 得白 色晶体 (2- ( 2-氯吡啶 -5-甲基) -3-硝亚胺基 -1,2,4-三唑啉) 1.2g, 收率 40%, 熔点: 113〜115°C。 Into a 250 mL three-necked flask, 3.0 g of N,-nitro-N-(2-chloropyridin-5-methyl)formalaminoguanidine and 80 mL of a 5 mol/L aqueous acetic acid solution were successively introduced, and the mixture was heated to reflux for 5 hours. Cooled, red oil precipitated, column chromatography (petroleum ether: ethyl acetate = 1: 1, v/v), white Color crystal (2-(2-chloropyridine-5-methyl)-3-nitroimino-1,2,4-triazoline) 1.2 g, yield 40%, m.p.: 113~115.
结构表征数据如下: The structural characterization data is as follows:
1H NMR(DMSO-d6,5ppm):5.27(s,2H),7.53(d, lH),7.79(q, lH),8.41(d, 1H), 8.53(s, lH), 14.05(brs, lH) 实施例 2、 2-(2-氯噻唑 -5-甲基) -5-丁基 -3-硝亚胺基 -1,2,4-三唑啉 (式 I 中 R1为 2-氯噻唑 -5-基, R2为丁基)的合成 1H NMR (DMSO-d 6 , 5 ppm): 5.27 (s, 2H), 7.53 (d, lH), 7.79 (q, lH), 8.41 (d, 1H), 8.53 (s, lH), 14.05 (brs, lH) Example 2, 2-(2-chlorothiazol-5-methyl)-5-butyl-3-nitroimino-1,2,4-triazoline (in the formula I, R1 is 2-chloro Synthesis of thiazol-5-yl, R2 is butyl)
(1) Ν'-硝基正戊醛缩氨基胍的合成 (1) Synthesis of Ν'-nitro-n-pentanal acetal
于 250ml三口瓶中, 依次投入 N,-硝基氨基胍 2.0g(0.017mol), 无水甲 醇 30mL和冰醋酸 0.25mL, 磁力搅拌下加热至 50°C, 从恒压滴液漏斗中缓 慢滴加正戊醛 1.7g (0.02mol) , 滴加完毕, 加热至回流反应 1.0小时。 降 温, 减压脱去溶剂, 将所得粗品用乙醇-石油醚 (体积比为 3 : 1 ) 重结晶, 得淡黄色粉末 (Ν'-硝基正戊醛缩氨基胍) 2.6g, 收率 83%, 熔点 94〜 95°C。 In a 250 ml three-necked flask, 2.0 g (0.017 mol) of N-nitroaminopurine, 30 mL of anhydrous methanol and 0.25 mL of glacial acetic acid were sequentially added, and heated to 50 ° C under magnetic stirring, and slowly dripped from a constant pressure dropping funnel. 1.7 g (0.02 mol) of n-pentanal was added, and the addition was completed, and the mixture was heated to reflux for 1.0 hour. The temperature was lowered, the solvent was evaporated under reduced pressure, and the obtained crude product was recrystallized from ethanol- petroleum ether (3:1 by volume) to give a pale yellow powder ( Ν'- nitro-n-pental acetal) 2.6 g, yield 83 %, melting point 94~95 °C.
(2) Ν'-硝基 -N-(2-氯噻唑 -5-甲基)正戊醛缩氨基胍的合成 (2) Synthesis of Ν'-nitro-N-(2-chlorothiazole-5-methyl)-n-pental acetal
于 250mL三口瓶中, 依次投入 N,-硝基正戊醛缩氨基胍 3.7g (0.02 mol) , 无水 DMF 40mL和少量 KI, 安装干燥管, 开动磁搅拌, 并用冰水 浴降温。 降温至 10°C以下, 分 3 批加入质量含量为 70%的氢化钠 1.37 g (0.04mol), 并反应 1小时。 从滴液漏斗中缓慢滴加 2-氯 -5氯甲基噻唑 5.1g (0.03mol)和无水 DMF 20mL的溶液。 滴加完毕, 撤去冰水浴, 自然升温至 室温。 在室温下反应 16 小时后, 缓慢加入 200mL水中, 析出固体。 静 置, 过滤, 水洗涤, 干燥, 得无色针状晶体 (Ν'-硝基 -N-2- (2-氯噻唑 -5- 甲基) 正戊醛缩氨基胍) 3.2g, 收率 51%。 用乙醇-石油醚 (体积比为 1 : 2) 重结晶, 熔点 60〜61 °C。 In a 250 mL three-necked flask, N,-nitro-n-pental acetal, 3.7 g (0.02 mol), anhydrous DMF 40 mL, and a small amount of KI were placed in this order. A dry tube was installed, magnetic stirring was started, and the temperature was lowered with an ice water bath. The temperature was lowered to below 10 ° C, and 1.37 g (0.04 mol) of sodium hydride having a mass content of 70% was added in three portions and reacted for 1 hour. A solution of 2-chloro-5 chloromethylthiazole 5.1 g (0.03 mol) and anhydrous DMF 20 mL was slowly added dropwise from the dropping funnel. After the addition is completed, the ice water bath is removed and the temperature is naturally raised to room temperature. After reacting at room temperature for 16 hours, it was slowly added to 200 mL of water to precipitate a solid. After standing, filtration, water washing, and drying, colorless needle crystals (Ν'-nitro-N-2-(2-chlorothiazol-5-methyl)-n-pental acetal) 3.2 g, yield 51%. Recrystallization from ethanol-petroleum ether (volume ratio 1: 2), melting point 60~61 °C.
结构表征数据如下: 1H NMR(CDCl3,5ppm):0.93(t,3H), 1.28-1.38(m,2H); 1.48-1.58(m,2H),2.36-2.42(m,2H),5.25(s,2H),7.39(t, lH),7.42(s, lH),7.42(brs, lH),8.99(brs, lH) The structure characterization data is as follows: 1H NMR (CDCl 3 , 5 ppm): 0.93 (t, 3H), 1.28-1.38 (m, 2H) ; 1.48-1.58 (m, 2H), 2.36-2.42 (m, 2H), 5.25 ( s, 2H), 7.39 (t, lH), 7.42 (s, lH), 7.42 (brs, lH), 8.99 (brs, lH)
(3) 2-(2-氯噻唑 -5-甲基) -5-丁基 -3-硝亚胺基 -1,2,4-三唑啉的合成 (3) Synthesis of 2-(2-chlorothiazole-5-methyl)-5-butyl-3-nitroimino--1,2,4-triazoline
于 250mL三口瓶中, 依次投入 3.0g N,-硝基 -N-2- (2-氯噻唑 -5-甲基) 正戊醛缩氨基胍和 80mL 5mol/L的醋酸水溶液, 加热至回流, 并保持 5小 时。 冷却, 有红色油状物析出, 柱层析 (石油醚: 乙酸乙酯 =2:3 , v/v) , 得白色晶体 (2- ( 2-氯吡啶 -5-甲基) -3-硝亚胺基 -1,2,4-三唑啉) 1.2g, 收 率 40%,熔点: 88~90°C o In a 250 mL three-necked flask, 3.0 g of N,-nitro-N-2-(2-chlorothiazole-5-methyl) was sequentially introduced. N-pentanal aminoguanidine and 80 mL of a 5 mol/L aqueous acetic acid solution were heated to reflux for 5 hours. Cooling, red oily precipitation, column chromatography (petroleum ether: ethyl acetate = 2:3, v/v) to give white crystals (2-(2-chloropyridine-5-methyl)-3-nit. Amino-1,2,4-triazoline) 1.2g, yield 40%, melting point: 88~90°C o
结构表征数据如下: 1H NMR (DMSO-d6,5ppm):0.89(t,3H), 1.26-The structural characterization data is as follows: 1H NMR (DMSO-d 6 , 5 ppm): 0.89 (t, 3H), 1.26-
1.36(m,2H), 1.58-1.67(m,2H),2.66-2.71(m,2H),5.39(s,2H),7.75(s, lH), 13.851.36 (m, 2H), 1.58-1.67 (m, 2H), 2.66-2.71 (m, 2H), 5.39 (s, 2H), 7.75 (s, lH), 13.85
(brs, lH) (brs, lH)
合成化合物结构见表 1和表 2。 The structures of the synthesized compounds are shown in Tables 1 and 2.
表 1.化合物 I的结构和分析数据 Table 1. Structure and analytical data of Compound I
N N02 N N0 2
表 2.化合物 VI的结构和分析数据 Table 2. Structure and analytical data for Compound VI
NN02 H NN0 2 H
(式 VI) (formula VI)
实施例 3、 本发明所提供式 I、 式 VI化合物的杀虫活性测试 Example 3, the insecticidal activity test of the compound of formula I and formula VI provided by the present invention
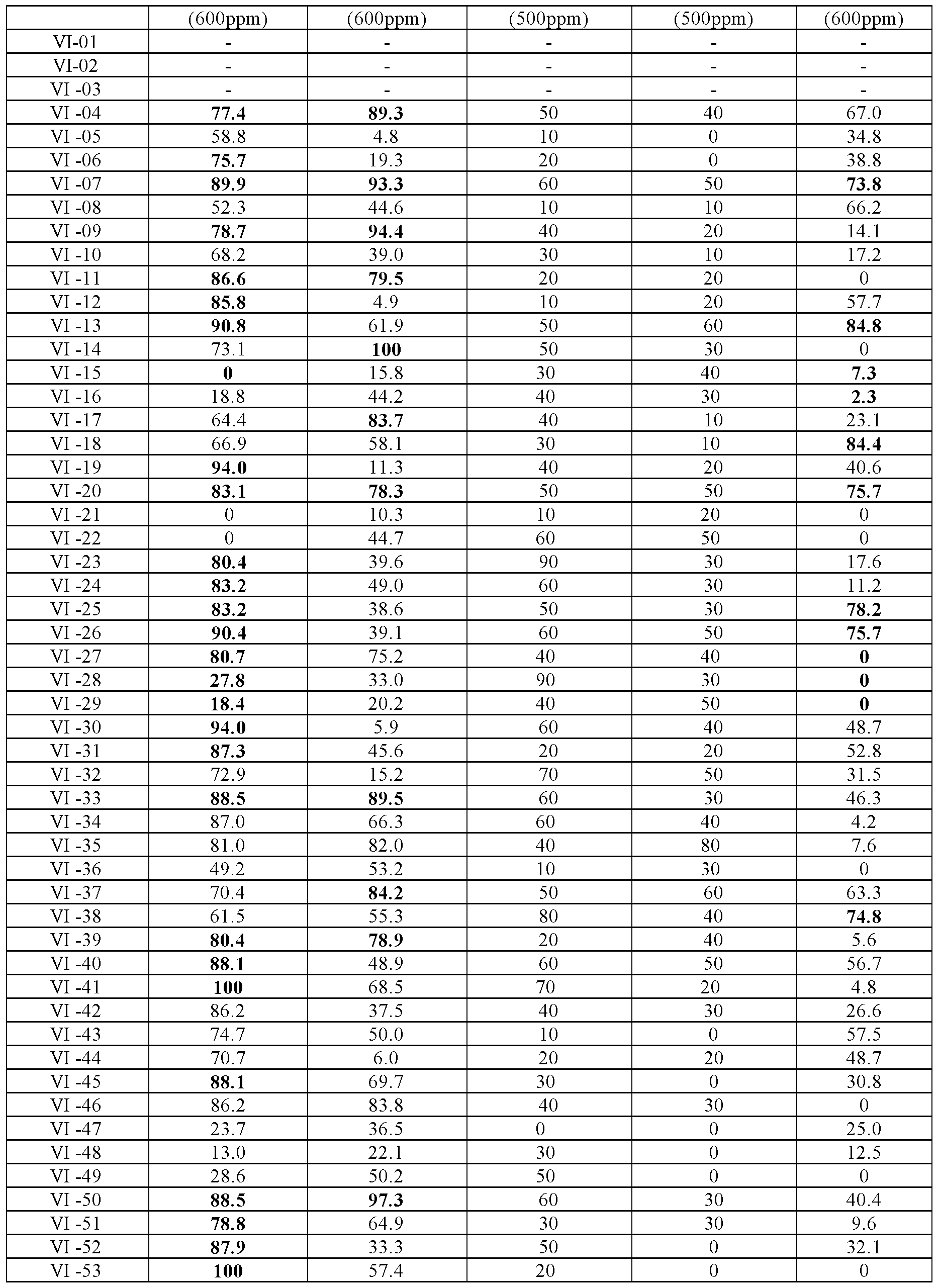
蚜虫属于同翅目害虫, 具有剌吸口器, 是一种常见的农作物主要害 虫。 以桃虫牙 Aphids belong to Homoptera pests and have a sucking mouth device, which is a common pest of common crops. Peach tooth
gossypii), 菜缢管蚜 (萝卜蚜) ( z^op s er /m/)为测试对象, 采用浸渍法 测试。 其中桃蚜采自北京市海淀区甘蓝田, 桃粉蚜采自北京市海淀区桃 树, 棉蚜采自北京市海淀区木槿树, 菜缢管蚜采自北京市海淀区甘蓝田, 均使用 3日龄若虫进行测定。 Gossypii), 缢 缢 蚜 (radish 蚜) (z^op s er /m/) is the test object, tested by dipping method. Among them, the peach aphid was collected from the cabbage field in Haidian District, Beijing. The peach aphid was collected from Taoshu, Haidian District, Beijing. The cotton aphid was collected from Mudu Tree, Haidian District, Beijing. The vegetable tube was collected from the cabbage field in Haidian District, Beijing. The 3 day old nymph was measured.
操作过程: 准确称取本发明所提供的化合物 20mg (;按 100%含量计 算;), 用 4mL丙酮配制成质量百分含量为 0.5%母液, 再用含有质量含量 0. 1%Triton X- 100的水溶液配制成浓度为 0.06% (即 600ppm) 的测定药液 (低浓度测定时以此溶液稀释至所需浓度即可;)。 选择带蚜叶片, 留下 3日龄 若蚜, 将带蚜叶片在药液中浸渍 5s, 晾干后记录虫数, 放入加有保湿滤纸 的培养皿中, 加盖后放入 (25±1)°C光照培养箱中, 每个药剂处理 30头以 上, 同时设空白对照。 5-48小时后检査结果。 O. Triton X-100. The mass percentage of 0.5% mother liquor is used to make a mass percentage of 0.5% mother liquor, and the mass content is 0.1% Triton X-100. The aqueous solution is formulated into a measuring solution having a concentration of 0.06% (i.e., 600 ppm) (the solution can be diluted to the desired concentration when the concentration is low;). Select the leaves with sputum, leave 3 days old, and immerse the leaves with sputum for 5s in the liquid. After drying, record the number of insects, put them into the culture dish with moisturizing filter paper, cover and put (25± 1) In the °C light incubator, each agent is treated with 30 heads. On the top, set a blank control. Check the results after 5-48 hours.
死亡判断标准为: 轻触虫体, 不能正常爬行的害虫个体视为死亡个 体。 The criteria for death judgment are: Lightly touching the worm, and the individual who cannot crawl normally is regarded as a dead individual.
校正死亡率 (%) = (样品死亡率 -空白对照死亡率) I ( 1-空白对照死 亡率) χ 100%。 Corrected mortality (%) = (sample mortality - blank control mortality) I (1-blank control mortality) χ 100%.
朱砂叶螨雌成螨 i Tetranychus cinnabarinus Boisduva , 采自甘单 β棉 田。 用万分之一天平于 20 mL称量瓶称取 12 mg化合物样品, 再用 1〜5 mL 移液枪取 2 mL丙酮 /甲醇 (1 : 1)混合溶剂加入称量瓶, 待其充分溶解后, 加 入 18 ml含有 0. 1%吐温 -80 的水溶液, 充分混匀, 得 600 igl h 的测定 液。 The cinnabar is a female cockroach i Tetranychus cinnabarinus Boisduva, taken from the singular beta cotton field. Weigh 12 mg of the compound sample in a 20 mL weighing bottle with a one-tenth of a balance, and then add 2 mL of acetone/methanol (1:1) mixed solvent to the weighing bottle with a 1~5 mL pipette, until it is fully dissolved. Thereafter, 18 ml of an aqueous solution containing 0.1% Tween-80 was added, and the mixture was thoroughly mixed to obtain a measuring solution of 600 μg h .
操作过程: 采用浸渍法进行杀虫活性测定, 将转接好朱砂叶螨的豆叶在药 液中浸渍 5 s, 晾干后记录虫数放入加有保湿滤纸的培养皿中, 叶柄用保湿 棉保湿, 加盖后放入 (25±1)°C光照培养箱中。 每个药剂处理 20头以上。 48 小时后检査结果。 死亡判断标准为: 轻触虫体, 不能正常爬行的害虫个体 视为死亡个体。 Operation process: The insecticidal activity is determined by the dipping method. The bean leaves transferred to the cinnabar leafhopper are immersed in the liquid for 5 s. After drying, the number of insects is recorded in a petri dish with moisturizing filter paper, and the petiole is moisturized. Cotton moisturizing, capped and placed in a (25 ± 1) ° C light incubator. Each drug is processed more than 20 times. Check the results after 48 hours. The criteria for death are: Light-touched worms, individuals who cannot crawl normally are considered dead individuals.
棉铃虫属鳞翅目夜蛾科害虫, 具有咀嚼口器。 用万分之一天平量瓶称 取化合物样品, 用二甲基甲酰胺 (DMF ) 配制成母液, 再用含 0. 1%曲拉通 Helicoverpa armigera is a pest of Lepidoptera, and has a chewing mouth. The sample of the compound is weighed in a one-tenth of a 10,000-week volume, and the mother liquor is prepared with dimethylformamide (DMF), and then 0.1% Triton
X- 100的水溶液配制测定药液。 An aqueous solution of X-100 was prepared to determine the drug solution.
操作过程: 将棉叶清洗干净, 用打孔器器将干净苋菜叶打成直径 2cm叶 碟, 在药液中浸渍 10s, 晾干后将叶碟放入 10孔试验盒中, 接入棉铃虫 2 龄幼虫, 每孔 1头, 加保鲜膜、 加盖后放入 (27±1)°C光照培养箱中。 48小 时后检査结果。 以拨针轻触虫体反应不正常或无反应者为死亡。 Operation process: Clean the cotton leaf, use a puncher to cut the clean leek leaf into a 2cm diameter leaf dish, soak it in the liquid for 10s, dry it, put the leaf dish into the 10-hole test box, and connect the cotton bollworm 2nd instar larvae, 1 per hole, add plastic wrap, cover and put in (27±1) °C light incubator. Check the results after 48 hours. Death occurs when the needle touches the insect body and the reaction is abnormal or non-responsive.
甜菜夜蛾属鳞翅目夜蛾科害虫, 具有咀嚼口器, 采用药剂配制: 用万 分之一天平量瓶称取化合物样品, 用二甲基甲酰胺 (DMF ) 配制成母液, 再用含 0. 1%曲拉通 X- 100的水溶液配制测定药液。 Beet armyworm, Lepidoptera, Lepidoptera, has a chewing mouth, and is formulated with a drug: a compound sample is weighed in a one-tenth of a milliliter bottle, and a mother liquor is prepared with dimethylformamide (DMF). 0. 1% Triton X-100 aqueous solution was prepared to determine the drug solution.
操作过程: 将苋菜叶清洗干净, 用打孔器器将干净苋菜叶打成直径 Operation process: Wash the leek leaves and use a puncher to cut the clean leek leaves into diameters.
2cm叶碟, 在药液中浸渍 10s, 晾干后将叶碟放入 10孔试验盒中, 接入甜 菜夜蛾 2龄幼虫, 每孔 1头, 加保鲜膜、 加盖后放入 (27±1)°C光照培养箱 中。 48小时后检査结果。 以拨针轻触虫体反应不正常或无反应者为死亡。 杀虫活性结果见表 3和表 4。 表 3.化合物 I对蚜虫杀灭活性 (^OOppm) 2cm leaf dish, immersed in the liquid for 10s, dry and put the leaf dish into the 10-hole test box, connect the 2nd instar larva of beet armyworm, one hole per hole, add plastic wrap, cover and put in (27 ±1) °C in the light incubator. The results were checked after 48 hours. Death occurs when the needle touches the insect body and the reaction is abnormal or non-responsive. The insecticidal activity results are shown in Tables 3 and 4. Table 3. Compound I killing activity against aphids (^OOppm)
化合物 校正死亡率(%) Compound corrected mortality (%)
桃蚜 (48hrs) 瓜 (棉)蚜 (24hrs) 桃粉蚜(24hrs) 菜缢管蚜 (24hrs;i 1-01 42.6 55.8 33.2 75.0Taoyuan (48hrs) melon (cotton) 蚜 (24hrs) peach 蚜 (24hrs) 菜缢管蚜 (24hrs;i 1-01 42.6 55.8 33.2 75.0
1-02 32.0 45.5 40.1 44.41-02 32.0 45.5 40.1 44.4
1-03 75.3 50.9 25.2 1001-03 75.3 50.9 25.2 100
1-04 32.3 41.7 27.6 60.31-04 32.3 41.7 27.6 60.3
1-05 55.5 50.0 46.3 62.31-05 55.5 50.0 46.3 62.3
1-06 26.7 54.3 15.3 51.81-06 26.7 54.3 15.3 51.8
1-07 30.0 47.5 43.2 75.21-07 30.0 47.5 43.2 75.2
1-08 100 32.6 94.7 1001-08 100 32.6 94.7 100
1-09 72.2 41.7 55.5 81.61-09 72.2 41.7 55.5 81.6
I -10 78.0 57.7 60.2 85.8I -10 78.0 57.7 60.2 85.8
1-11 52.3 14.3 40.2 77.31-11 52.3 14.3 40.2 77.3
1-12 46.9 34.4 23.8 63.81-12 46.9 34.4 23.8 63.8
1-13 85.2 60.4 79.6 88.21-13 85.2 60.4 79.6 88.2
1-14 93.5 100 97.8 1001-14 93.5 100 97.8 100
1-15 76.4 55.6 65.6 93.01-15 76.4 55.6 65.6 93.0
1-16 77.8 46.2 60.3 90.81-16 77.8 46.2 60.3 90.8
1-17 56.0 30.0 25.9 77.31-17 56.0 30.0 25.9 77.3
1-18 66.0 67.4 32.3 69.71-18 66.0 67.4 32.3 69.7
1-19 76.2 88.9 78.6 7.01-19 76.2 88.9 78.6 7.0
1-20 16.8 59.4 20.6 50.11-20 16.8 59.4 20.6 50.1
1-21 87.5 100 100 1001-21 87.5 100 100 100
1-22 13.2 55.0 25.2 7.31-22 13.2 55.0 25.2 7.3
1-23 56.6 57.1 75.3 62.31-23 56.6 57.1 75.3 62.3
1-24 60.8 27.7 26.1 36.21-24 60.8 27.7 26.1 36.2
1-25 89.9 46.7 90.5 1001-25 89.9 46.7 90.5 100
1-26 91.7 55.9 89.3 1001-26 91.7 55.9 89.3 100
1-27 100 100 98.0 1001-27 100 100 98.0 100
1-28 68.2 73.0 69.8 48.91-28 68.2 73.0 69.8 48.9
1-29 60.5 20.0 56.9 67.41-29 60.5 20.0 56.9 67.4
1-30 72.0 44.7 68.6 73.61-30 72.0 44.7 68.6 73.6
1-31 59.2 68.1 63.5 21.01-31 59.2 68.1 63.5 21.0
1-32 88.0 44.1 80.2 94.61-32 88.0 44.1 80.2 94.6
1-33 54.6 40.0 65.3 32.01-33 54.6 40.0 65.3 32.0
1-34 100 96.7 94.4 99.21-34 100 96.7 94.4 99.2
I 35 42.2 30.6 36.4 24.4I 35 42.2 30.6 36.4 24.4
1-36 68.5 56.0 59.0 46.81-36 68.5 56.0 59.0 46.8
1-37 88.7 60.6 92.2 86.51-37 88.7 60.6 92.2 86.5
1-38 56.4 65.1 64.3 32.61-38 56.4 65.1 64.3 32.6
1-39 90.3 73.8 88.8 95.71-39 90.3 73.8 88.8 95.7
1-40 83.3 40.3 86.4 78.91-40 83.3 40.3 86.4 78.9
1-41 100 96.9 66.7 95.41-41 100 96.9 66.7 95.4
1-42 86.5 31.3 78.8 1001-42 86.5 31.3 78.8 100
1-43 95.6 61.5 87.6 96.71-43 95.6 61.5 87.6 96.7
1-44 32.3 25.7 55.5 45.21-44 32.3 25.7 55.5 45.2
1-45 78.9 78.1 65.2 77.31-45 78.9 78.1 65.2 77.3
1-46 76.2 63.3 55.8 31.41-46 76.2 63.3 55.8 31.4
1-47 89.5 85.4 82.5 72.31-47 89.5 85.4 82.5 72.3
1-48 68.8 45.0 75.6 94.91-48 68.8 45.0 75.6 94.9
1-49 76.9 54.8 52.0 96.71-49 76.9 54.8 52.0 96.7
1-50 65.2 54.8 46.5 71.11-50 65.2 54.8 46.5 71.1
1-51 58.5 47.7 75.3 40.41-51 58.5 47.7 75.3 40.4
1-52 69.6 40.3 50.0 49.21-52 69.6 40.3 50.0 49.2
1-53 95.8 100 97.0 100 吡虫啉 100 62.3 100 62.3 表 4.化合物 VI的杀虫活性1-53 95.8 100 97.0 100 Imidacloprid 100 62.3 100 62.3 Table 4. Insecticidal activity of compound VI
部分化合物进一步筛选结果见表 5 Further screening results of some compounds are shown in Table 5.
表 5.部分化合物 I和 VI对蚜虫的杀虫活性 校正死亡率(%) Table 5. Insecticidal activity of some compounds I and VI against aphids Corrected mortality (%)
化合物 桃蚜 瓜!;棉;!蚜 菜缢管蚜 Compound peach 蚜 melon! ;棉;!蚜菜缢管蚜
lOppm 3ppm lOppm 3ppm lOppm 3ppm lOppm 3ppm lOppm 3ppm lOppm 3ppm
< 1 -08 98.3 92.5 45.5 13.2 88.7 65.4< 1 -08 98.3 92.5 45.5 13.2 88.7 65.4
1 -14 Ο 99.4 94.6 99.4 95.6 100 98.81 -14 Ο 99.4 94.6 99.4 95.6 100 98.8
1 -21 96.5 93.4 98.3 95.1 99.8 96.71 -21 96.5 93.4 98.3 95.1 99.8 96.7
1 -25 88.6 80.7 75.2 60.3 95.7 88.31 -25 88.6 80.7 75.2 60.3 95.7 88.3
1 -26 81.4 82.3 65.5 50.2 88.9 86.41 -26 81.4 82.3 65.5 50.2 88.9 86.4
1 -27 100 98.6 98.7 96.3 100 99.41 -27 100 98.6 98.7 96.3 100 99.4
1 -34 98.2 90.3 92.6 89.6 90.5 90.21 -34 98.2 90.3 92.6 89.6 90.5 90.2
1 -37 78.6 65.8 69.7 63.5 88.2 79.11 -37 78.6 65.8 69.7 63.5 88.2 79.1
1 -39 82.4 78.2 69.5 60.8 86.9 70.21 -39 82.4 78.2 69.5 60.8 86.9 70.2
1 -41 98.4 90.4 88.3 89.0 90.5 86.41 -41 98.4 90.4 88.3 89.0 90.5 86.4
1 -42 66.6 56.8 50.4 51.7 87.6 80.51 -42 66.6 56.8 50.4 51.7 87.6 80.5
1 -43 79.4 60.3 69.8 42.0 88.4 80.21 -43 79.4 60.3 69.8 42.0 88.4 80.2
1 -53 100 95.6 95.4 92.6 100 98.71 -53 100 95.6 95.4 92.6 100 98.7
VI -07 75.5 60.6 45.3 26.8 65.2 50.2 VI -07 75.5 60.6 45.3 26.8 65.2 50.2
61.2 46.9 79.8 72.5 59.4 46.3 61.2 46.9 79.8 72.5 59.4 46.3
VI -26 52.4 50.5 23.7 22.0 53.5 21.4VI -26 52.4 50.5 23.7 22.0 53.5 21.4
VI -33 66.8 61.5 77.3 54.6 44.1 23.5 VI -33 66.8 61.5 77.3 54.6 44.1 23.5
59.8 47.6 42.5 15.9 66.4 36.8 59.8 47.6 42.5 15.9 66.4 36.8
VI -41 100 99.8 92.3 90.6 100 99.6VI -41 100 99.8 92.3 90.6 100 99.6
VI -46 45.2 30.0 65.4 48.3 23.2 4.0 VI -46 45.2 30.0 65.4 48.3 23.2 4.0
76.6 62.5 78.5 62.0 80.4 75.1 76.6 62.5 78.5 62.0 80.4 75.1
VI -53 100 94.3 90.2 91.0 100 99.8 表 6.化合物 VI-41和 VI-53对蚜虫的田间小区防效 VI -53 100 94.3 90.2 91.0 100 99.8 Table 6. Compounds VI-41 and VI-53 for field control of aphids
"WP"代表可湿性粉剂; " SC"代表悬浮剂。 "WP" stands for wettable powder; "SC" stands for suspending agent.
表 7.化合物 VI-41和 VI-53对稻飞虱的田间小区防效 Table 7. Compound VI-41 and VI-53 for field control of rice planthopper
表中, "WP"代表可湿性粉剂; " SC"代表悬浮剂。 工业应用 In the table, "WP" stands for wettable powder; "SC" stands for suspending agent. Industrial application
本发明提供了结构通式如式 I所示 2,5-二取代 -3-硝亚胺基 -1,2,4-三唑 啉类化合物及其前体化合物式 VI所示硝基缩氨基胍类化合物的制备方法与 应用。 上述化合物是本发明的发明人经过广泛调研和合理设计, 通过对大 量化合物的筛选, 优选出的一类的杀虫活性高, 且制备方法简便, 并提供 了一条优选的工艺路线, 安全性好, 成本低廉, 使得该化合物的实际应用 价值大大提高。 The present invention provides a 2,5-disubstituted-3-niminoimido-1,2,4-triazoline compound of the formula I and a precursor compound thereof, and a nitro group of the formula VI. Preparation methods and applications of terpenoids. The above compounds are extensively investigated and rationally designed by the inventors of the present invention, and The screening of the quantitative compound has a high insecticidal activity, and the preparation method is simple, and provides a preferred process route, which has good safety and low cost, and the practical application value of the compound is greatly improved.
杀虫活性实验表明, 式 ( I ) 所示 2,5-二取代 -3-硝亚胺基 - 1,2,4-三唑 啉类化合物和式 (VI ) 所示硝基缩氨基胍类化合物对蚜虫、 飞虱、 叶螨、 棉铃虫、 甜菜夜蛾等农作物害虫具有很高的防效, 可用作植物杀虫剂使 用。 2 Insecticidal activity experiments showed that the 2,5-disubstituted 3-nitroimido-1,2,4-triazoline compounds of formula (I) and the nitroaminosamines of formula (VI) The compound has high control effect against crop pests such as aphids, planthoppers, leafhoppers, cotton bollworms, and beet armyworm, and can be used as a plant insecticide. 2
(式 I) (式 VI) (Formula I) (Formula VI)
Claims
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2011/001096 WO2013003977A1 (en) | 2011-07-01 | 2011-07-01 | Compound of 2,5-disubstituted-3-nitroimino-1,2,4-triazoline and preparation method and use as pesticide thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| PCT/CN2011/001096 WO2013003977A1 (en) | 2011-07-01 | 2011-07-01 | Compound of 2,5-disubstituted-3-nitroimino-1,2,4-triazoline and preparation method and use as pesticide thereof |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2013003977A1 true WO2013003977A1 (en) | 2013-01-10 |
Family
ID=47436429
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/CN2011/001096 Ceased WO2013003977A1 (en) | 2011-07-01 | 2011-07-01 | Compound of 2,5-disubstituted-3-nitroimino-1,2,4-triazoline and preparation method and use as pesticide thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2013003977A1 (en) |
Cited By (123)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN103314673A (en) * | 2013-06-04 | 2013-09-25 | 新疆生产建设兵团第十三师农业科学研究所 | Method of processing cotton seeds |
| WO2014053404A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Pesticidally active mixtures comprising anthranilamide compounds |
| WO2014053395A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Use of n-thio-anthranilamide compounds on cultivated plants |
| WO2014053406A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of controlling ryanodine-modulator insecticide resistant insects |
| WO2014053403A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of controlling insecticide resistant insects |
| WO2014053401A2 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of improving plant health |
| WO2014053405A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Pesticidally active mixtures comprising anthranilamide compounds |
| WO2014053407A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2014079841A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079804A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079814A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079752A1 (en) | 2012-11-23 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079813A1 (en) | 2012-11-23 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079772A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079773A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079820A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Use of anthranilamide compounds for reducing insect-vectored viral infections |
| WO2014079764A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079774A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079766A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014090700A1 (en) | 2012-12-14 | 2014-06-19 | Basf Se | Malononitrile compounds for controlling animal pests |
| WO2014102244A1 (en) | 2012-12-27 | 2014-07-03 | Basf Se | 2-(pyridin-3-yl)-5-hetaryl-thiazole compounds carrying an imine or imine-derived substituent for combating invertebrate pests |
| WO2014170300A1 (en) | 2013-04-19 | 2014-10-23 | Basf Se | N-substituted acyl-imino-pyridine compounds and derivatives for combating animal pests |
| WO2014202751A1 (en) | 2013-06-21 | 2014-12-24 | Basf Se | Methods for controlling pests in soybean |
| WO2015007682A1 (en) | 2013-07-15 | 2015-01-22 | Basf Se | Pesticide compounds |
| CN104447617A (en) * | 2014-12-17 | 2015-03-25 | 南开大学 | Synthesis and application of nitro arylideneamino guanidine compounds containing 1, 2, 3-thiadiazole |
| WO2015040116A1 (en) | 2013-09-19 | 2015-03-26 | Basf Se | N-acylimino heterocyclic compounds |
| WO2015055497A1 (en) | 2013-10-16 | 2015-04-23 | Basf Se | Substituted pesticidal pyrazole compounds |
| WO2015055757A1 (en) | 2013-10-18 | 2015-04-23 | Basf Se | Use of pesticidal active carboxamide derivative in soil and seed application and treatment methods |
| WO2015091645A1 (en) | 2013-12-18 | 2015-06-25 | Basf Se | Azole compounds carrying an imine-derived substituent |
| WO2015091649A1 (en) | 2013-12-18 | 2015-06-25 | Basf Se | N-substituted imino heterocyclic compounds |
| WO2015104422A1 (en) | 2014-01-13 | 2015-07-16 | Basf Se | Dihydrothiophene compounds for controlling invertebrate pests |
| WO2016071499A1 (en) | 2014-11-06 | 2016-05-12 | Basf Se | 3-pyridyl heterobicyclic compound for controlling invertebrate pests |
| WO2016128261A2 (en) | 2015-02-11 | 2016-08-18 | Basf Se | Pesticidal mixture comprising a pyrazole compound, an insecticide and a fungicide |
| WO2016162371A1 (en) | 2015-04-07 | 2016-10-13 | Basf Agrochemical Products B.V. | Use of an insecticidal carboxamide compound against pests on cultivated plants |
| WO2016198611A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino heterocyclic compounds |
| WO2016198613A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino compounds |
| WO2017016883A1 (en) | 2015-07-24 | 2017-02-02 | Basf Se | Process for preparation of cyclopentene compounds |
| WO2017093163A1 (en) | 2015-11-30 | 2017-06-08 | Basf Se | Mixtures of cis-jasmone and bacillus amyloliquefaciens |
| WO2017140614A1 (en) | 2016-02-19 | 2017-08-24 | Basf Se | Method for controlling pests of soybean, corn, and cotton plants |
| WO2017153217A1 (en) | 2016-03-09 | 2017-09-14 | Basf Se | Spirocyclic derivatives |
| WO2017153218A1 (en) | 2016-03-11 | 2017-09-14 | Basf Se | Method for controlling pests of plants |
| WO2017167832A1 (en) | 2016-04-01 | 2017-10-05 | Basf Se | Bicyclic compounds |
| WO2017198588A1 (en) | 2016-05-18 | 2017-11-23 | Basf Se | Capsules comprising benzylpropargylethers for use as nitrification inhibitors |
| WO2018108671A1 (en) | 2016-12-16 | 2018-06-21 | Basf Se | Pesticidal compounds |
| WO2018162312A1 (en) | 2017-03-10 | 2018-09-13 | Basf Se | Spirocyclic derivatives |
| WO2018166855A1 (en) | 2017-03-16 | 2018-09-20 | Basf Se | Heterobicyclic substituted dihydroisoxazoles |
| WO2018177781A1 (en) | 2017-03-28 | 2018-10-04 | Basf Se | Pesticidal compounds |
| WO2018177970A1 (en) | 2017-03-31 | 2018-10-04 | Basf Se | Process for preparing chiral 2,3-dihydrothiazolo[3,2-a]pyrimidin-4-ium compounds |
| WO2018192793A1 (en) | 2017-04-20 | 2018-10-25 | Basf Se | Substituted rhodanine derivatives |
| WO2018197466A1 (en) | 2017-04-26 | 2018-11-01 | Basf Se | Substituted succinimide derivatives as pesticides |
| WO2018206479A1 (en) | 2017-05-10 | 2018-11-15 | Basf Se | Bicyclic pesticidal compounds |
| US10149477B2 (en) | 2014-10-06 | 2018-12-11 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| WO2018224455A1 (en) | 2017-06-07 | 2018-12-13 | Basf Se | Substituted cyclopropyl derivatives |
| WO2018229202A1 (en) | 2017-06-16 | 2018-12-20 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2018234488A1 (en) | 2017-06-23 | 2018-12-27 | Basf Se | SUBSTITUTED CYCLOPROPYL DERIVATIVES |
| WO2018234202A1 (en) | 2017-06-19 | 2018-12-27 | Basf Se | SUBSTITUTED PYRIMIDINIUM COMPOUNDS AND DERIVATIVES FOR CONTROLLING HARMFUL ANIMALS |
| CN109180536A (en) * | 2018-10-09 | 2019-01-11 | 南通天泽化工有限公司 | A kind of preparation method of nitro-guanyl hydrazine |
| WO2019042932A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| EP3453706A1 (en) | 2017-09-08 | 2019-03-13 | Basf Se | Pesticidal imidazole compounds |
| WO2019072906A1 (en) | 2017-10-13 | 2019-04-18 | Basf Se | Imidazolidine pyrimidinium compounds for combating animal pests |
| WO2019121143A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Substituted cyclopropyl derivatives |
| WO2019121159A1 (en) | 2017-12-21 | 2019-06-27 | Basf Se | Pesticidal compounds |
| WO2019134840A1 (en) | 2018-01-05 | 2019-07-11 | Basf Se | Control of pests of soybean plants with mesoionic compounds |
| WO2019137995A1 (en) | 2018-01-11 | 2019-07-18 | Basf Se | Novel pyridazine compounds for controlling invertebrate pests |
| WO2019145140A1 (en) | 2018-01-09 | 2019-08-01 | Basf Se | Silylethynyl hetaryl compounds as nitrification inhibitors |
| WO2019166558A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of pyrazole propargyl ethers as nitrification inhibitors |
| WO2019166561A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of alkoxypyrazoles as nitrification inhibitors |
| WO2019166560A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of n-functionalized alkoxy pyrazole compounds as nitrification inhibitors |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| WO2019185413A1 (en) | 2018-03-27 | 2019-10-03 | Basf Se | Pesticidal substituted cyclopropyl derivatives |
| WO2019211106A1 (en) | 2018-04-30 | 2019-11-07 | Basf Se | Control of pests of soybean plants with mesoionic compounds |
| WO2019219529A1 (en) | 2018-05-15 | 2019-11-21 | Basf Se | Mixtures comprising benzpyrimoxan and oxazosulfyl and uses and methods of applying them |
| WO2019224092A1 (en) | 2018-05-22 | 2019-11-28 | Basf Se | Pesticidally active c15-derivatives of ginkgolides |
| WO2020002472A1 (en) | 2018-06-28 | 2020-01-02 | Basf Se | Use of alkynylthiophenes as nitrification inhibitors |
| WO2020020765A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of a substituted thiazolidine compound as nitrification inhibitor |
| WO2020020777A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of substituted 2-thiazolines as nitrification inhibitors |
| US10556844B2 (en) | 2015-02-06 | 2020-02-11 | Basf Se | Pyrazole compounds as nitrification inhibitors |
| EP3613736A1 (en) | 2018-08-22 | 2020-02-26 | Basf Se | Substituted glutarimide derivatives |
| EP3628156A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method for controlling pests of sugarcane, citrus, rapeseed, and potato plants |
| EP3628157A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| EP3628158A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| WO2020064492A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling pests by seed treatment application of a mesoionic compound or mixture thereof |
| EP3643705A1 (en) | 2018-10-24 | 2020-04-29 | Basf Se | Pesticidal compounds |
| WO2020109039A1 (en) | 2018-11-28 | 2020-06-04 | Basf Se | Pesticidal compounds |
| WO2020126591A1 (en) | 2018-12-18 | 2020-06-25 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| US10743535B2 (en) | 2017-08-18 | 2020-08-18 | H&K Solutions Llc | Insecticide for flight-capable pests |
| EP3696177A1 (en) | 2019-02-12 | 2020-08-19 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2020239517A1 (en) | 2019-05-29 | 2020-12-03 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| EP3766879A1 (en) | 2019-07-19 | 2021-01-20 | Basf Se | Pesticidal pyrazole derivatives |
| EP3769623A1 (en) | 2019-07-22 | 2021-01-27 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2021130143A1 (en) | 2019-12-23 | 2021-07-01 | Basf Se | Enzyme enhanced root uptake of agrochemical active compound |
| WO2021170463A1 (en) | 2020-02-28 | 2021-09-02 | BASF Agro B.V. | Methods and uses of a mixture comprising alpha-cypermethrin and dinotefuran for controlling invertebrate pests in turf |
| US11142514B2 (en) | 2015-10-02 | 2021-10-12 | Basf Se | Imino compounds with a 2-chloropyrimidin-5-yl substituent as pest-control agents |
| WO2021219513A1 (en) | 2020-04-28 | 2021-11-04 | Basf Se | Pesticidal compounds |
| EP3909950A1 (en) | 2020-05-13 | 2021-11-17 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| CN114634456A (en) * | 2022-03-22 | 2022-06-17 | 赣南师范大学 | A kind of 5-nitroimino-4H-1,2,4-triazole compound and its preparation method and application |
| WO2022167488A1 (en) | 2021-02-02 | 2022-08-11 | Basf Se | Synergistic action of dcd and alkoxypyrazoles as nitrification inhibitors |
| EP4043444A1 (en) | 2021-02-11 | 2022-08-17 | Basf Se | Substituted isoxazoline derivatives |
| WO2022243523A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of an n-functionalized alkoxy pyrazole compound as nitrification inhibitor |
| WO2022243521A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of ethynylpyridine compounds as nitrification inhibitors |
| WO2022268810A1 (en) | 2021-06-21 | 2022-12-29 | Basf Se | Metal-organic frameworks with pyrazole-based building blocks |
| EP4119547A1 (en) | 2021-07-12 | 2023-01-18 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4140986A1 (en) | 2021-08-23 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4140995A1 (en) | 2021-08-27 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4151631A1 (en) | 2021-09-20 | 2023-03-22 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP4194453A1 (en) | 2021-12-08 | 2023-06-14 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4198023A1 (en) | 2021-12-16 | 2023-06-21 | Basf Se | Pesticidally active thiosemicarbazone compounds |
| EP4198033A1 (en) | 2021-12-14 | 2023-06-21 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP4238971A1 (en) | 2022-03-02 | 2023-09-06 | Basf Se | Substituted isoxazoline derivatives |
| WO2023203066A1 (en) | 2022-04-21 | 2023-10-26 | Basf Se | Synergistic action as nitrification inhibitors of dcd oligomers with alkoxypyrazole and its oligomers |
| WO2023208447A1 (en) | 2022-04-25 | 2023-11-02 | Basf Se | An emulsifiable concentrate having a (substituted) benzaldehyde-based solvent system |
| WO2024028243A1 (en) | 2022-08-02 | 2024-02-08 | Basf Se | Pyrazolo pesticidal compounds |
| EP4342885A1 (en) | 2022-09-20 | 2024-03-27 | Basf Se | N-(3-(aminomethyl)-phenyl)-5-(4-phenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-amine derivatives and similar compounds as pesticides |
| EP4389210A1 (en) | 2022-12-21 | 2024-06-26 | Basf Se | Heteroaryl compounds for the control of invertebrate pests |
| EP4455137A1 (en) | 2023-04-24 | 2024-10-30 | Basf Se | Pyrimidine compounds for the control of invertebrate pests |
| EP4467535A1 (en) | 2023-05-25 | 2024-11-27 | Basf Se | Lactam pesticidal compounds |
| EP4488269A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488270A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488273A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| WO2025117659A1 (en) | 2023-11-29 | 2025-06-05 | Basf Corporation | Insecticide delivery by nanocarriers |
| EP4574819A1 (en) | 2023-12-22 | 2025-06-25 | Basf Se | Diazinone compounds for the control of invertebrate pests |
| WO2025242699A1 (en) | 2024-05-22 | 2025-11-27 | Basf Se | Method for improving rainfastness of an agrochemical active ingredient |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3636001A (en) * | 1968-04-16 | 1972-01-18 | Sandoz Ltd | Amine salts of nitraminotriazoles |
| CN1077843A (en) * | 1988-12-27 | 1993-11-03 | 武田药品工业株式会社 | Guanidine derivative insecticide composition |
-
2011
- 2011-07-01 WO PCT/CN2011/001096 patent/WO2013003977A1/en not_active Ceased
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US3636001A (en) * | 1968-04-16 | 1972-01-18 | Sandoz Ltd | Amine salts of nitraminotriazoles |
| CN1077843A (en) * | 1988-12-27 | 1993-11-03 | 武田药品工业株式会社 | Guanidine derivative insecticide composition |
Non-Patent Citations (4)
Cited By (133)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2014053407A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | N-thio-anthranilamide compounds and their use as pesticides |
| WO2014053404A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Pesticidally active mixtures comprising anthranilamide compounds |
| WO2014053395A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Use of n-thio-anthranilamide compounds on cultivated plants |
| WO2014053406A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of controlling ryanodine-modulator insecticide resistant insects |
| WO2014053403A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of controlling insecticide resistant insects |
| WO2014053401A2 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Method of improving plant health |
| WO2014053405A1 (en) | 2012-10-01 | 2014-04-10 | Basf Se | Pesticidally active mixtures comprising anthranilamide compounds |
| WO2014079764A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079841A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079804A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079814A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079772A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079773A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079820A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Use of anthranilamide compounds for reducing insect-vectored viral infections |
| WO2014079774A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079766A1 (en) | 2012-11-22 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079752A1 (en) | 2012-11-23 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| WO2014079813A1 (en) | 2012-11-23 | 2014-05-30 | Basf Se | Pesticidal mixtures |
| US10117430B2 (en) | 2012-12-14 | 2018-11-06 | Basf Se | Malononitrile compounds for controlling animal pests |
| WO2014090700A1 (en) | 2012-12-14 | 2014-06-19 | Basf Se | Malononitrile compounds for controlling animal pests |
| WO2014102244A1 (en) | 2012-12-27 | 2014-07-03 | Basf Se | 2-(pyridin-3-yl)-5-hetaryl-thiazole compounds carrying an imine or imine-derived substituent for combating invertebrate pests |
| WO2014170300A1 (en) | 2013-04-19 | 2014-10-23 | Basf Se | N-substituted acyl-imino-pyridine compounds and derivatives for combating animal pests |
| CN103314673A (en) * | 2013-06-04 | 2013-09-25 | 新疆生产建设兵团第十三师农业科学研究所 | Method of processing cotton seeds |
| WO2014202751A1 (en) | 2013-06-21 | 2014-12-24 | Basf Se | Methods for controlling pests in soybean |
| WO2015007682A1 (en) | 2013-07-15 | 2015-01-22 | Basf Se | Pesticide compounds |
| WO2015040116A1 (en) | 2013-09-19 | 2015-03-26 | Basf Se | N-acylimino heterocyclic compounds |
| WO2015055497A1 (en) | 2013-10-16 | 2015-04-23 | Basf Se | Substituted pesticidal pyrazole compounds |
| WO2015055757A1 (en) | 2013-10-18 | 2015-04-23 | Basf Se | Use of pesticidal active carboxamide derivative in soil and seed application and treatment methods |
| EP3456201A1 (en) | 2013-10-18 | 2019-03-20 | BASF Agrochemical Products B.V. | Use of pesticidal active carboxamide derivative in soil and seed application and treatment meth-ods |
| WO2015091645A1 (en) | 2013-12-18 | 2015-06-25 | Basf Se | Azole compounds carrying an imine-derived substituent |
| WO2015091649A1 (en) | 2013-12-18 | 2015-06-25 | Basf Se | N-substituted imino heterocyclic compounds |
| WO2015104422A1 (en) | 2014-01-13 | 2015-07-16 | Basf Se | Dihydrothiophene compounds for controlling invertebrate pests |
| US10149477B2 (en) | 2014-10-06 | 2018-12-11 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| WO2016071499A1 (en) | 2014-11-06 | 2016-05-12 | Basf Se | 3-pyridyl heterobicyclic compound for controlling invertebrate pests |
| CN104447617A (en) * | 2014-12-17 | 2015-03-25 | 南开大学 | Synthesis and application of nitro arylideneamino guanidine compounds containing 1, 2, 3-thiadiazole |
| US10556844B2 (en) | 2015-02-06 | 2020-02-11 | Basf Se | Pyrazole compounds as nitrification inhibitors |
| WO2016128261A2 (en) | 2015-02-11 | 2016-08-18 | Basf Se | Pesticidal mixture comprising a pyrazole compound, an insecticide and a fungicide |
| US10701937B2 (en) | 2015-02-11 | 2020-07-07 | Basf Se | Pesticidal mixture comprising a pyrazole compound, an insecticide and a fungicide |
| WO2016162371A1 (en) | 2015-04-07 | 2016-10-13 | Basf Agrochemical Products B.V. | Use of an insecticidal carboxamide compound against pests on cultivated plants |
| WO2016198611A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino heterocyclic compounds |
| WO2016198613A1 (en) | 2015-06-11 | 2016-12-15 | Basf Se | N-(thio)acylimino compounds |
| WO2017016883A1 (en) | 2015-07-24 | 2017-02-02 | Basf Se | Process for preparation of cyclopentene compounds |
| US11142514B2 (en) | 2015-10-02 | 2021-10-12 | Basf Se | Imino compounds with a 2-chloropyrimidin-5-yl substituent as pest-control agents |
| WO2017093163A1 (en) | 2015-11-30 | 2017-06-08 | Basf Se | Mixtures of cis-jasmone and bacillus amyloliquefaciens |
| WO2017140614A1 (en) | 2016-02-19 | 2017-08-24 | Basf Se | Method for controlling pests of soybean, corn, and cotton plants |
| WO2017153217A1 (en) | 2016-03-09 | 2017-09-14 | Basf Se | Spirocyclic derivatives |
| WO2017153218A1 (en) | 2016-03-11 | 2017-09-14 | Basf Se | Method for controlling pests of plants |
| WO2017167832A1 (en) | 2016-04-01 | 2017-10-05 | Basf Se | Bicyclic compounds |
| WO2017198588A1 (en) | 2016-05-18 | 2017-11-23 | Basf Se | Capsules comprising benzylpropargylethers for use as nitrification inhibitors |
| WO2018108671A1 (en) | 2016-12-16 | 2018-06-21 | Basf Se | Pesticidal compounds |
| WO2018162312A1 (en) | 2017-03-10 | 2018-09-13 | Basf Se | Spirocyclic derivatives |
| WO2018166855A1 (en) | 2017-03-16 | 2018-09-20 | Basf Se | Heterobicyclic substituted dihydroisoxazoles |
| WO2018177781A1 (en) | 2017-03-28 | 2018-10-04 | Basf Se | Pesticidal compounds |
| WO2018177970A1 (en) | 2017-03-31 | 2018-10-04 | Basf Se | Process for preparing chiral 2,3-dihydrothiazolo[3,2-a]pyrimidin-4-ium compounds |
| EP3978504A1 (en) | 2017-03-31 | 2022-04-06 | Basf Se | Chiral 2,3-dihydrothiazolo[3,2-a]pyrimidine derivatives for combating animal pests |
| WO2018192793A1 (en) | 2017-04-20 | 2018-10-25 | Basf Se | Substituted rhodanine derivatives |
| WO2018197466A1 (en) | 2017-04-26 | 2018-11-01 | Basf Se | Substituted succinimide derivatives as pesticides |
| WO2018206479A1 (en) | 2017-05-10 | 2018-11-15 | Basf Se | Bicyclic pesticidal compounds |
| WO2018224455A1 (en) | 2017-06-07 | 2018-12-13 | Basf Se | Substituted cyclopropyl derivatives |
| WO2018229202A1 (en) | 2017-06-16 | 2018-12-20 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2018234202A1 (en) | 2017-06-19 | 2018-12-27 | Basf Se | SUBSTITUTED PYRIMIDINIUM COMPOUNDS AND DERIVATIVES FOR CONTROLLING HARMFUL ANIMALS |
| WO2018234488A1 (en) | 2017-06-23 | 2018-12-27 | Basf Se | SUBSTITUTED CYCLOPROPYL DERIVATIVES |
| US10743535B2 (en) | 2017-08-18 | 2020-08-18 | H&K Solutions Llc | Insecticide for flight-capable pests |
| WO2019042932A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| WO2019043183A1 (en) | 2017-08-31 | 2019-03-07 | Basf Se | Method of controlling rice pests in rice |
| EP3453706A1 (en) | 2017-09-08 | 2019-03-13 | Basf Se | Pesticidal imidazole compounds |
| WO2019072906A1 (en) | 2017-10-13 | 2019-04-18 | Basf Se | Imidazolidine pyrimidinium compounds for combating animal pests |
| WO2019121143A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Substituted cyclopropyl derivatives |
| WO2019121159A1 (en) | 2017-12-21 | 2019-06-27 | Basf Se | Pesticidal compounds |
| WO2019134840A1 (en) | 2018-01-05 | 2019-07-11 | Basf Se | Control of pests of soybean plants with mesoionic compounds |
| WO2019145140A1 (en) | 2018-01-09 | 2019-08-01 | Basf Se | Silylethynyl hetaryl compounds as nitrification inhibitors |
| WO2019137995A1 (en) | 2018-01-11 | 2019-07-18 | Basf Se | Novel pyridazine compounds for controlling invertebrate pests |
| WO2019166561A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of alkoxypyrazoles as nitrification inhibitors |
| WO2019166560A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of n-functionalized alkoxy pyrazole compounds as nitrification inhibitors |
| WO2019166558A1 (en) | 2018-02-28 | 2019-09-06 | Basf Se | Use of pyrazole propargyl ethers as nitrification inhibitors |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019185413A1 (en) | 2018-03-27 | 2019-10-03 | Basf Se | Pesticidal substituted cyclopropyl derivatives |
| WO2019211106A1 (en) | 2018-04-30 | 2019-11-07 | Basf Se | Control of pests of soybean plants with mesoionic compounds |
| WO2019219529A1 (en) | 2018-05-15 | 2019-11-21 | Basf Se | Mixtures comprising benzpyrimoxan and oxazosulfyl and uses and methods of applying them |
| WO2019224092A1 (en) | 2018-05-22 | 2019-11-28 | Basf Se | Pesticidally active c15-derivatives of ginkgolides |
| WO2020002472A1 (en) | 2018-06-28 | 2020-01-02 | Basf Se | Use of alkynylthiophenes as nitrification inhibitors |
| WO2020020765A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of a substituted thiazolidine compound as nitrification inhibitor |
| WO2020020777A1 (en) | 2018-07-23 | 2020-01-30 | Basf Se | Use of substituted 2-thiazolines as nitrification inhibitors |
| EP3613736A1 (en) | 2018-08-22 | 2020-02-26 | Basf Se | Substituted glutarimide derivatives |
| EP3628158A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| WO2020064408A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| WO2020064480A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Pesticidal mixture comprising a mesoionic compound and a biopesticide |
| WO2020064492A1 (en) | 2018-09-28 | 2020-04-02 | Basf Se | Method of controlling pests by seed treatment application of a mesoionic compound or mixture thereof |
| EP3628157A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method of controlling insecticide resistant insects and virus transmission to plants |
| EP3628156A1 (en) | 2018-09-28 | 2020-04-01 | Basf Se | Method for controlling pests of sugarcane, citrus, rapeseed, and potato plants |
| CN109180536A (en) * | 2018-10-09 | 2019-01-11 | 南通天泽化工有限公司 | A kind of preparation method of nitro-guanyl hydrazine |
| EP3643705A1 (en) | 2018-10-24 | 2020-04-29 | Basf Se | Pesticidal compounds |
| WO2020083733A1 (en) | 2018-10-24 | 2020-04-30 | Basf Se | Pesticidal compounds |
| WO2020109039A1 (en) | 2018-11-28 | 2020-06-04 | Basf Se | Pesticidal compounds |
| WO2020126591A1 (en) | 2018-12-18 | 2020-06-25 | Basf Se | Substituted pyrimidinium compounds for combating animal pests |
| EP3696177A1 (en) | 2019-02-12 | 2020-08-19 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2020239517A1 (en) | 2019-05-29 | 2020-12-03 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| EP3766879A1 (en) | 2019-07-19 | 2021-01-20 | Basf Se | Pesticidal pyrazole derivatives |
| WO2021013561A1 (en) | 2019-07-19 | 2021-01-28 | Basf Se | Pesticidal pyrazole and triazole derivatives |
| EP3769623A1 (en) | 2019-07-22 | 2021-01-27 | Basf Se | Mesoionic imidazolium compounds and derivatives for combating animal pests |
| WO2021130143A1 (en) | 2019-12-23 | 2021-07-01 | Basf Se | Enzyme enhanced root uptake of agrochemical active compound |
| WO2021170463A1 (en) | 2020-02-28 | 2021-09-02 | BASF Agro B.V. | Methods and uses of a mixture comprising alpha-cypermethrin and dinotefuran for controlling invertebrate pests in turf |
| WO2021219513A1 (en) | 2020-04-28 | 2021-11-04 | Basf Se | Pesticidal compounds |
| EP3909950A1 (en) | 2020-05-13 | 2021-11-17 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| WO2022167488A1 (en) | 2021-02-02 | 2022-08-11 | Basf Se | Synergistic action of dcd and alkoxypyrazoles as nitrification inhibitors |
| EP4043444A1 (en) | 2021-02-11 | 2022-08-17 | Basf Se | Substituted isoxazoline derivatives |
| WO2022243523A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of an n-functionalized alkoxy pyrazole compound as nitrification inhibitor |
| WO2022243521A1 (en) | 2021-05-21 | 2022-11-24 | Basf Se | Use of ethynylpyridine compounds as nitrification inhibitors |
| WO2022268810A1 (en) | 2021-06-21 | 2022-12-29 | Basf Se | Metal-organic frameworks with pyrazole-based building blocks |
| EP4119547A1 (en) | 2021-07-12 | 2023-01-18 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4140986A1 (en) | 2021-08-23 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4140995A1 (en) | 2021-08-27 | 2023-03-01 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4151631A1 (en) | 2021-09-20 | 2023-03-22 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP4194453A1 (en) | 2021-12-08 | 2023-06-14 | Basf Se | Pyrazine compounds for the control of invertebrate pests |
| EP4198033A1 (en) | 2021-12-14 | 2023-06-21 | Basf Se | Heterocyclic compounds for the control of invertebrate pests |
| EP4198023A1 (en) | 2021-12-16 | 2023-06-21 | Basf Se | Pesticidally active thiosemicarbazone compounds |
| EP4238971A1 (en) | 2022-03-02 | 2023-09-06 | Basf Se | Substituted isoxazoline derivatives |
| CN114634456A (en) * | 2022-03-22 | 2022-06-17 | 赣南师范大学 | A kind of 5-nitroimino-4H-1,2,4-triazole compound and its preparation method and application |
| CN114634456B (en) * | 2022-03-22 | 2024-02-20 | 赣南师范大学 | 5-nitroimino-4H-1, 2, 4-triazole compound and preparation method and application thereof |
| WO2023203066A1 (en) | 2022-04-21 | 2023-10-26 | Basf Se | Synergistic action as nitrification inhibitors of dcd oligomers with alkoxypyrazole and its oligomers |
| WO2023208447A1 (en) | 2022-04-25 | 2023-11-02 | Basf Se | An emulsifiable concentrate having a (substituted) benzaldehyde-based solvent system |
| WO2024028243A1 (en) | 2022-08-02 | 2024-02-08 | Basf Se | Pyrazolo pesticidal compounds |
| EP4342885A1 (en) | 2022-09-20 | 2024-03-27 | Basf Se | N-(3-(aminomethyl)-phenyl)-5-(4-phenyl)-5-(trifluoromethyl)-4,5-dihydroisoxazol-3-amine derivatives and similar compounds as pesticides |
| EP4389210A1 (en) | 2022-12-21 | 2024-06-26 | Basf Se | Heteroaryl compounds for the control of invertebrate pests |
| EP4455137A1 (en) | 2023-04-24 | 2024-10-30 | Basf Se | Pyrimidine compounds for the control of invertebrate pests |
| EP4467535A1 (en) | 2023-05-25 | 2024-11-27 | Basf Se | Lactam pesticidal compounds |
| EP4488269A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488270A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| EP4488273A1 (en) | 2023-07-06 | 2025-01-08 | Basf Se | Triazole compounds for the control of invertebrate pests |
| WO2025117659A1 (en) | 2023-11-29 | 2025-06-05 | Basf Corporation | Insecticide delivery by nanocarriers |
| EP4574819A1 (en) | 2023-12-22 | 2025-06-25 | Basf Se | Diazinone compounds for the control of invertebrate pests |
| WO2025242699A1 (en) | 2024-05-22 | 2025-11-27 | Basf Se | Method for improving rainfastness of an agrochemical active ingredient |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2013003977A1 (en) | Compound of 2,5-disubstituted-3-nitroimino-1,2,4-triazoline and preparation method and use as pesticide thereof | |
| CN101821232B (en) | Condensed amino nitroguanidine compounds, preparation method and use as botanical insecticides thereof | |
| JPS6368567A (en) | Novel heterocyclic compound and insecticide | |
| DE2946524A1 (en) | AZOLYLOXY-CARBONIC ACID-N-OXY-AMIDES, METHOD FOR THE PRODUCTION THEREOF AND THEIR USE AS HERBICIDES | |
| WO2020177778A1 (en) | 1-pyridylpyrazole amide compound, the preparation method and application thereof | |
| US5134145A (en) | Pesticidal pyrimidinyl benzoic acids and esters | |
| US5084459A (en) | Tetrahydropyrimidines, their production and use | |
| EP2692723B1 (en) | Aryloxy dihalopropenyl ether compound and use thereof | |
| CN102690258A (en) | Nitrogenous heterocyclic-imide derivative and preparation method and purpose thereof | |
| TW200526123A (en) | Substituted nicotinoylcarbamates | |
| AU2020210116B2 (en) | 3-substituted phenylamidine compounds, preparation and use thereof | |
| CN103755700A (en) | Novel pyrazol amides compound and application thereof | |
| WO2012065568A1 (en) | Azo heterocyclic compound having insecticidal activity, and preparation and applications thereof | |
| CN102285965B (en) | N-(cyanoalkyl) benzamide compound and application thereof | |
| US5225423A (en) | Butenone compounds, their preparation and their use as pesticides | |
| US5486521A (en) | Pyrimidinyl aryl ketone oximes | |
| DE69301675T2 (en) | Derivatives of 7-ethynyl-alpha (methoxymethylene) 1-naphthylacetic acid, its production process and its use as pesticides | |
| CN103833639B (en) | pyrazolyl acrylonitrile compound and application thereof | |
| JPS61267575A (en) | Nitroimino derivative, production thereof and insecticide | |
| CN110256404B (en) | Diene compounds with insecticidal activity, preparation and use thereof | |
| DD234603A5 (en) | Antifungal Composition | |
| CN119661510B (en) | Isoxazoline substituted pyridine amide compound and preparation method and application thereof | |
| CN111518079A (en) | Preparation and Application of Pyrazole Derivatives Containing N-pyridyl-3-pyrazolephenylmethoxypyrazole Units | |
| CN116606263B (en) | A fluorine-containing benzothiazole diester compound and its use | |
| CN114591262B (en) | A kind of m-phenylaminoamide compound containing oxadiazole substituent, its preparation method and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 11869002 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 11869002 Country of ref document: EP Kind code of ref document: A1 |