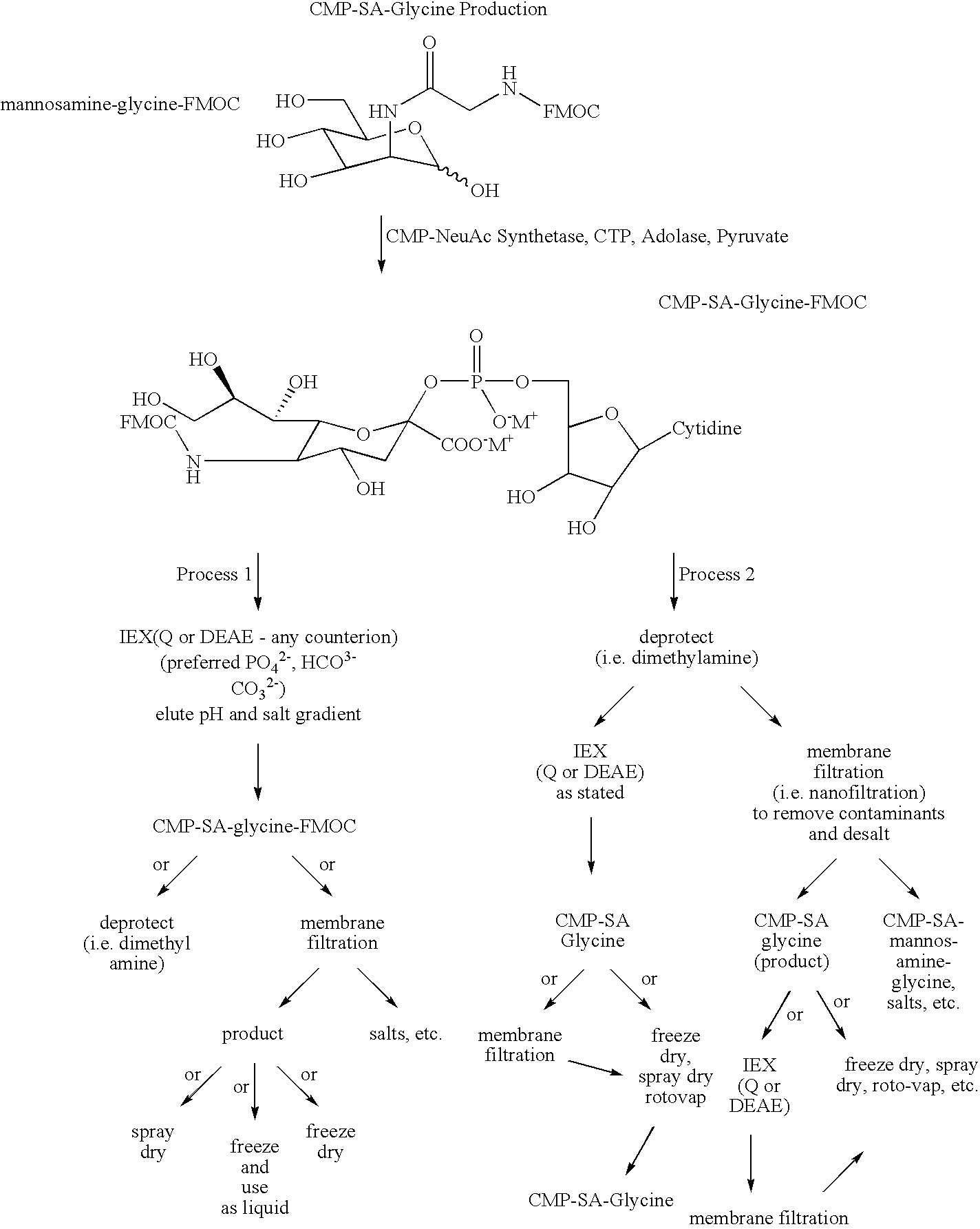
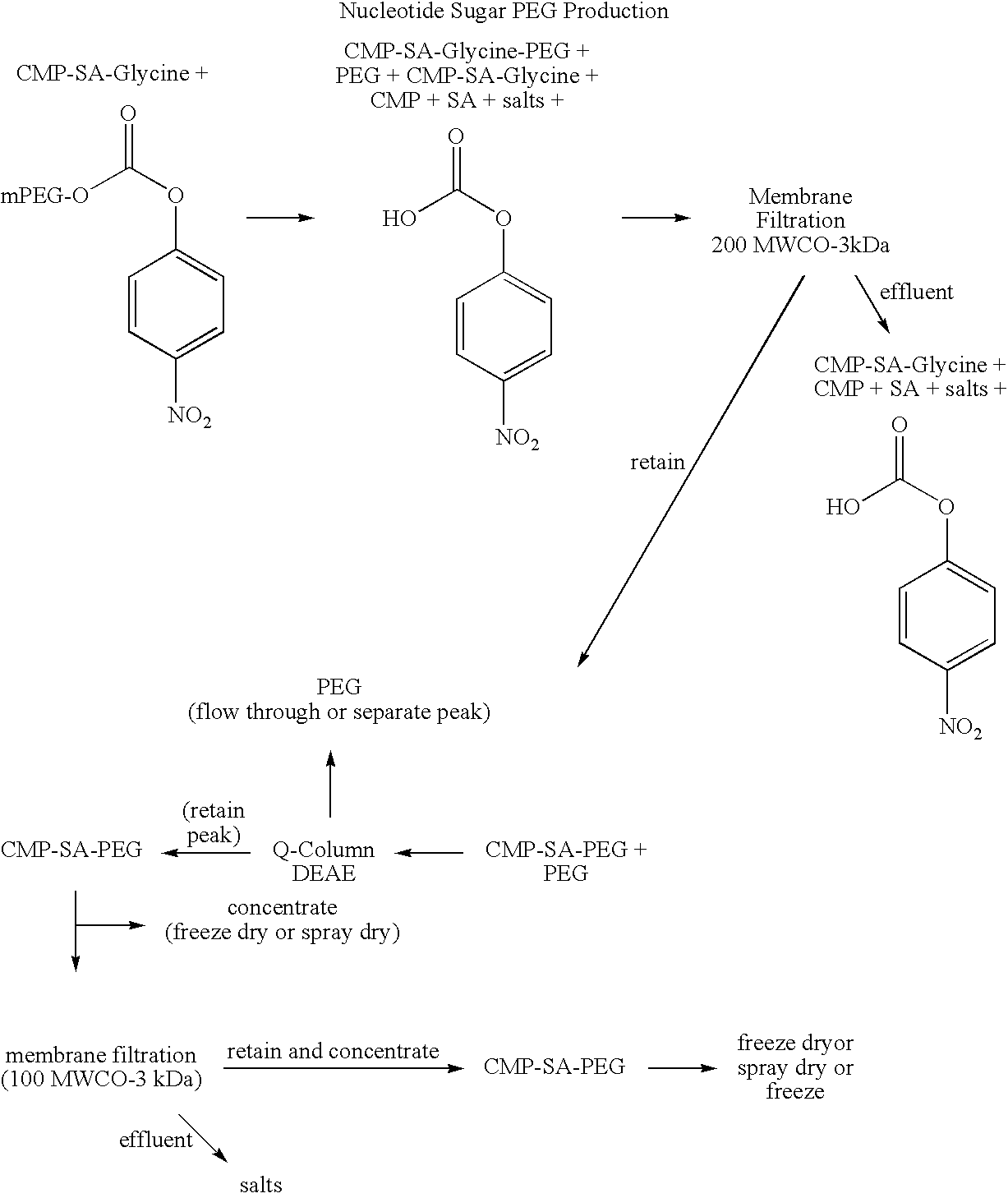
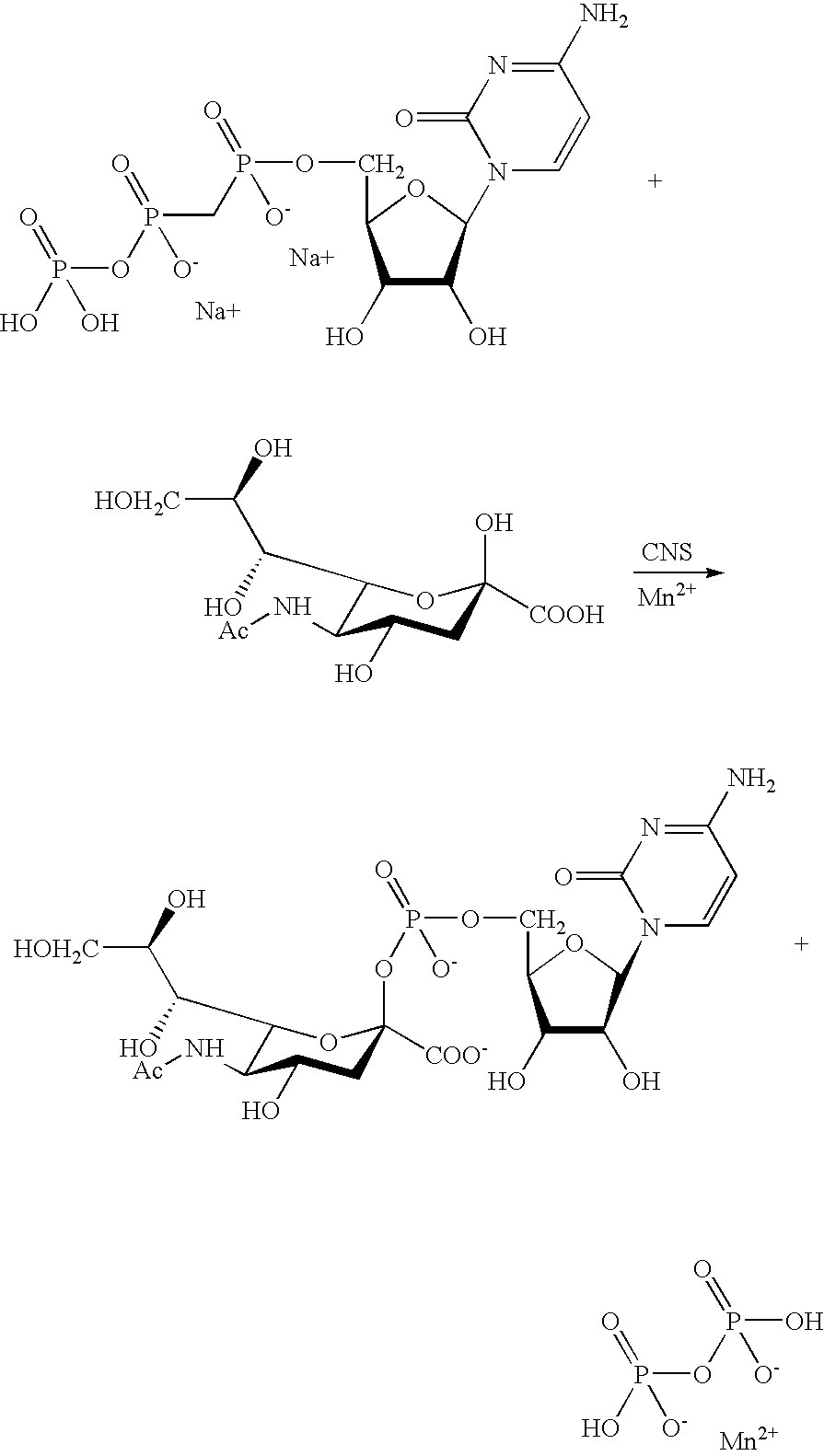
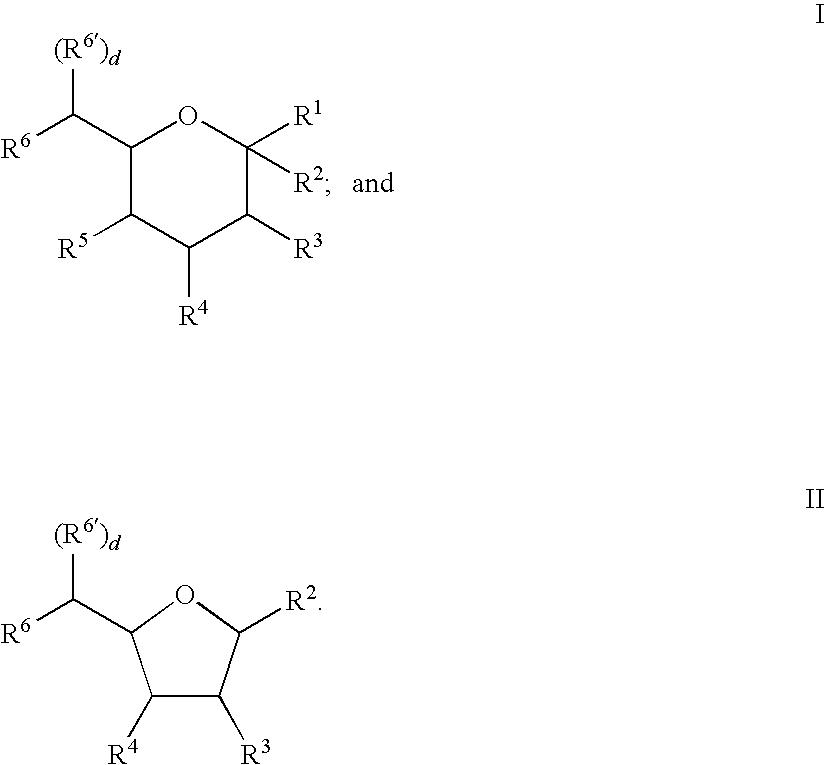
US20090048440A1 - Nucleotide Sugar Purification Using Membranes - Google Patents
Nucleotide Sugar Purification Using Membranes Download PDFInfo
- Publication number
- US20090048440A1 US20090048440A1 US12/092,563 US9256306A US2009048440A1 US 20090048440 A1 US20090048440 A1 US 20090048440A1 US 9256306 A US9256306 A US 9256306A US 2009048440 A1 US2009048440 A1 US 2009048440A1
- Authority
- US
- United States
- Prior art keywords
- substituted
- membrane
- nucleotide
- unsubstituted
- desired product
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 239000002773 nucleotide Substances 0.000 title claims abstract description 152
- 235000000346 sugar Nutrition 0.000 title claims abstract description 141
- 239000012528 membrane Substances 0.000 title claims description 200
- 125000003729 nucleotide group Chemical group 0.000 title claims description 136
- 238000000746 purification Methods 0.000 title description 82
- -1 nucleotide sugars Chemical class 0.000 claims abstract description 169
- 238000000034 method Methods 0.000 claims abstract description 158
- 239000000356 contaminant Substances 0.000 claims abstract description 62
- 239000000203 mixture Substances 0.000 claims abstract description 45
- 150000003839 salts Chemical class 0.000 claims abstract description 23
- OIZGSVFYNBZVIK-FHHHURIISA-N 3'-sialyllactose Chemical compound O1[C@@H]([C@H](O)[C@H](O)CO)[C@H](NC(=O)C)[C@@H](O)C[C@@]1(C(O)=O)O[C@@H]1[C@@H](O)[C@H](O[C@H]([C@H](O)CO)[C@H](O)[C@@H](O)C=O)O[C@H](CO)[C@@H]1O OIZGSVFYNBZVIK-FHHHURIISA-N 0.000 claims abstract description 11
- 229930186217 Glycolipid Natural products 0.000 claims abstract description 4
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 claims abstract 6
- DAEPDZWVDSPTHF-UHFFFAOYSA-M sodium pyruvate Chemical compound [Na+].CC(=O)C([O-])=O DAEPDZWVDSPTHF-UHFFFAOYSA-M 0.000 claims abstract 6
- 229910052943 magnesium sulfate Inorganic materials 0.000 claims abstract 3
- 235000019341 magnesium sulphate Nutrition 0.000 claims abstract 3
- FQENQNTWSFEDLI-UHFFFAOYSA-J sodium diphosphate Chemical group [Na+].[Na+].[Na+].[Na+].[O-]P([O-])(=O)OP([O-])([O-])=O FQENQNTWSFEDLI-UHFFFAOYSA-J 0.000 claims abstract 3
- 229940054269 sodium pyruvate Drugs 0.000 claims abstract 3
- SQVRNKJHWKZAKO-UHFFFAOYSA-N beta-N-Acetyl-D-neuraminic acid Natural products CC(=O)NC1C(O)CC(O)(C(O)=O)OC1C(O)C(O)CO SQVRNKJHWKZAKO-UHFFFAOYSA-N 0.000 claims description 59
- 238000001728 nano-filtration Methods 0.000 claims description 53
- 230000008569 process Effects 0.000 claims description 51
- 125000000217 alkyl group Chemical group 0.000 claims description 46
- SQVRNKJHWKZAKO-OQPLDHBCSA-N sialic acid Chemical compound CC(=O)N[C@@H]1[C@@H](O)C[C@@](O)(C(O)=O)OC1[C@H](O)[C@H](O)CO SQVRNKJHWKZAKO-OQPLDHBCSA-N 0.000 claims description 38
- 125000003118 aryl group Chemical group 0.000 claims description 28
- 125000004404 heteroalkyl group Chemical group 0.000 claims description 27
- IERHLVCPSMICTF-XVFCMESISA-N CMP group Chemical group P(=O)(O)(O)OC[C@@H]1[C@H]([C@H]([C@@H](O1)N1C(=O)N=C(N)C=C1)O)O IERHLVCPSMICTF-XVFCMESISA-N 0.000 claims description 25
- 229910019142 PO4 Inorganic materials 0.000 claims description 25
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 claims description 23
- 239000010452 phosphate Substances 0.000 claims description 22
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 claims description 19
- 125000001072 heteroaryl group Chemical group 0.000 claims description 16
- 239000011780 sodium chloride Substances 0.000 claims description 12
- HSCJRCZFDFQWRP-UHFFFAOYSA-N Uridindiphosphoglukose Natural products OC1C(O)C(O)C(CO)OC1OP(O)(=O)OP(O)(=O)OCC1C(O)C(O)C(N2C(NC(=O)C=C2)=O)O1 HSCJRCZFDFQWRP-UHFFFAOYSA-N 0.000 claims description 11
- 229910000162 sodium phosphate Inorganic materials 0.000 claims description 11
- 125000000592 heterocycloalkyl group Chemical group 0.000 claims description 10
- IEQCXFNWPAHHQR-UHFFFAOYSA-N lacto-N-neotetraose Natural products OCC1OC(OC2C(C(OC3C(OC(O)C(O)C3O)CO)OC(CO)C2O)O)C(NC(=O)C)C(O)C1OC1OC(CO)C(O)C(O)C1O IEQCXFNWPAHHQR-UHFFFAOYSA-N 0.000 claims description 10
- 230000000717 retained effect Effects 0.000 claims description 10
- 239000001488 sodium phosphate Substances 0.000 claims description 10
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 claims description 10
- OVRNDRQMDRJTHS-UHFFFAOYSA-N N-acelyl-D-glucosamine Natural products CC(=O)NC1C(O)OC(CO)C(O)C1O OVRNDRQMDRJTHS-UHFFFAOYSA-N 0.000 claims description 9
- OVRNDRQMDRJTHS-RTRLPJTCSA-N N-acetyl-D-glucosamine Chemical compound CC(=O)N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O OVRNDRQMDRJTHS-RTRLPJTCSA-N 0.000 claims description 9
- MBLBDJOUHNCFQT-LXGUWJNJSA-N N-acetylglucosamine Natural products CC(=O)N[C@@H](C=O)[C@@H](O)[C@H](O)[C@H](O)CO MBLBDJOUHNCFQT-LXGUWJNJSA-N 0.000 claims description 9
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 9
- 230000007935 neutral effect Effects 0.000 claims description 9
- 229910021380 Manganese Chloride Inorganic materials 0.000 claims description 8
- GLFNIEUTAYBVOC-UHFFFAOYSA-L Manganese chloride Chemical compound Cl[Mn]Cl GLFNIEUTAYBVOC-UHFFFAOYSA-L 0.000 claims description 8
- LFTYTUAZOPRMMI-UHFFFAOYSA-N UNPD164450 Natural products O1C(CO)C(O)C(O)C(NC(=O)C)C1OP(O)(=O)OP(O)(=O)OCC1C(O)C(O)C(N2C(NC(=O)C=C2)=O)O1 LFTYTUAZOPRMMI-UHFFFAOYSA-N 0.000 claims description 8
- LQEBEXMHBLQMDB-UHFFFAOYSA-N [[5-(2-amino-6-oxo-3h-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl] (3,4,5-trihydroxy-6-methyloxan-2-yl) hydrogen phosphate Chemical compound OC1C(O)C(O)C(C)OC1OP(O)(=O)OP(O)(=O)OCC1C(O)C(O)C(N2C3=C(C(N=C(N)N3)=O)N=C2)O1 LQEBEXMHBLQMDB-UHFFFAOYSA-N 0.000 claims description 8
- 235000011180 diphosphates Nutrition 0.000 claims description 8
- 239000011565 manganese chloride Substances 0.000 claims description 8
- 102000003960 Ligases Human genes 0.000 claims description 7
- 108090000364 Ligases Proteins 0.000 claims description 7
- 239000008101 lactose Substances 0.000 claims description 7
- HSCJRCZFDFQWRP-JZMIEXBBSA-N UDP-alpha-D-glucose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1OP(O)(=O)OP(O)(=O)OC[C@@H]1[C@@H](O)[C@@H](O)[C@H](N2C(NC(=O)C=C2)=O)O1 HSCJRCZFDFQWRP-JZMIEXBBSA-N 0.000 claims description 6
- 239000001226 triphosphate Substances 0.000 claims description 6
- 235000011178 triphosphate Nutrition 0.000 claims description 6
- MVMSCBBUIHUTGJ-UHFFFAOYSA-N 10108-97-1 Natural products C1=2NC(N)=NC(=O)C=2N=CN1C(C(C1O)O)OC1COP(O)(=O)OP(O)(=O)OC1OC(CO)C(O)C(O)C1O MVMSCBBUIHUTGJ-UHFFFAOYSA-N 0.000 claims description 5
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 claims description 5
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 claims description 5
- LFTYTUAZOPRMMI-NESSUJCYSA-N UDP-N-acetyl-alpha-D-galactosamine Chemical compound O1[C@H](CO)[C@H](O)[C@H](O)[C@@H](NC(=O)C)[C@H]1O[P@](O)(=O)O[P@](O)(=O)OC[C@@H]1[C@@H](O)[C@@H](O)[C@H](N2C(NC(=O)C=C2)=O)O1 LFTYTUAZOPRMMI-NESSUJCYSA-N 0.000 claims description 5
- HSCJRCZFDFQWRP-ABVWGUQPSA-N UDP-alpha-D-galactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1OP(O)(=O)OP(O)(=O)OC[C@@H]1[C@@H](O)[C@@H](O)[C@H](N2C(NC(=O)C=C2)=O)O1 HSCJRCZFDFQWRP-ABVWGUQPSA-N 0.000 claims description 5
- 239000001177 diphosphate Substances 0.000 claims description 5
- 235000002867 manganese chloride Nutrition 0.000 claims description 4
- 229940099607 manganese chloride Drugs 0.000 claims description 4
- XPPKVPWEQAFLFU-UHFFFAOYSA-N diphosphoric acid Chemical compound OP(O)(=O)OP(O)(O)=O XPPKVPWEQAFLFU-UHFFFAOYSA-N 0.000 claims description 3
- WPYMKLBDIGXBTP-UHFFFAOYSA-N benzoic acid Chemical compound OC(=O)C1=CC=CC=C1 WPYMKLBDIGXBTP-UHFFFAOYSA-N 0.000 claims 4
- IEQCXFNWPAHHQR-YKLSGRGUSA-N beta-D-Gal-(1->4)-beta-D-GlcNAc-(1->3)-beta-D-Gal-(1->4)-D-Glc Chemical compound O([C@H]1[C@H](O)[C@H]([C@@H](O[C@@H]1CO)O[C@@H]1[C@H]([C@H](O[C@@H]2[C@H](OC(O)[C@H](O)[C@H]2O)CO)O[C@H](CO)[C@@H]1O)O)NC(=O)C)[C@@H]1O[C@H](CO)[C@H](O)[C@H](O)[C@H]1O IEQCXFNWPAHHQR-YKLSGRGUSA-N 0.000 claims 4
- 102100039847 Globoside alpha-1,3-N-acetylgalactosaminyltransferase 1 Human genes 0.000 claims 3
- 101000887519 Homo sapiens Globoside alpha-1,3-N-acetylgalactosaminyltransferase 1 Proteins 0.000 claims 3
- XCCTYIAWTASOJW-UHFFFAOYSA-N UDP-Glc Natural products OC1C(O)C(COP(O)(=O)OP(O)(O)=O)OC1N1C(=O)NC(=O)C=C1 XCCTYIAWTASOJW-UHFFFAOYSA-N 0.000 claims 3
- LFTYTUAZOPRMMI-CFRASDGPSA-N UDP-N-acetyl-alpha-D-glucosamine Chemical compound O1[C@H](CO)[C@@H](O)[C@H](O)[C@@H](NC(=O)C)[C@H]1OP(O)(=O)OP(O)(=O)OC[C@@H]1[C@@H](O)[C@@H](O)[C@H](N2C(NC(=O)C=C2)=O)O1 LFTYTUAZOPRMMI-CFRASDGPSA-N 0.000 claims 3
- HDYANYHVCAPMJV-UHFFFAOYSA-N Uridine diphospho-D-glucuronic acid Natural products O1C(N2C(NC(=O)C=C2)=O)C(O)C(O)C1COP(O)(=O)OP(O)(=O)OC1OC(C(O)=O)C(O)C(O)C1O HDYANYHVCAPMJV-UHFFFAOYSA-N 0.000 claims 3
- DQQDLYVHOTZLOR-UHFFFAOYSA-N [[5-(2,4-dioxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl] (3,4,5-trihydroxyoxan-2-yl) hydrogen phosphate Chemical group O1C(N2C(NC(=O)C=C2)=O)C(O)C(O)C1COP(O)(=O)OP(O)(=O)OC1OCC(O)C(O)C1O DQQDLYVHOTZLOR-UHFFFAOYSA-N 0.000 claims 3
- WMWKTCPGFOEPBD-YGIWDPDDSA-N azane;(2s,3s,4s,5r,6r)-6-[[[(2r,3s,4r,5r)-5-(2,4-dioxopyrimidin-1-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-hydroxyphosphoryl]oxy-hydroxyphosphoryl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound N.C([C@@H]1[C@H]([C@H]([C@@H](O1)N1C(NC(=O)C=C1)=O)O)O)OP(O)(=O)OP(O)(=O)O[C@H]1O[C@H](C(O)=O)[C@@H](O)[C@H](O)[C@H]1O WMWKTCPGFOEPBD-YGIWDPDDSA-N 0.000 claims 3
- XOAGKSFNHBWACO-RAUZPKMFSA-L disodium;[[(2r,3s,4r,5r)-5-(2-amino-6-oxo-3h-purin-9-yl)-3,4-dihydroxyoxolan-2-yl]methoxy-oxidophosphoryl] [(2r,3s,4s,5s,6r)-3,4,5-trihydroxy-6-(hydroxymethyl)oxan-2-yl] phosphate Chemical compound [Na+].[Na+].C([C@H]1O[C@H]([C@@H]([C@@H]1O)O)N1C=NC=2C(=O)N=C(NC=21)N)OP([O-])(=O)OP([O-])(=O)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@@H]1O XOAGKSFNHBWACO-RAUZPKMFSA-L 0.000 claims 3
- 239000012634 fragment Substances 0.000 claims 3
- UHDGCWIWMRVCDJ-UHFFFAOYSA-N 1-beta-D-Xylofuranosyl-NH-Cytosine Natural products O=C1N=C(N)C=CN1C1C(O)C(O)C(CO)O1 UHDGCWIWMRVCDJ-UHFFFAOYSA-N 0.000 claims 2
- HVCOBJNICQPDBP-UHFFFAOYSA-N 3-[3-[3,5-dihydroxy-6-methyl-4-(3,4,5-trihydroxy-6-methyloxan-2-yl)oxyoxan-2-yl]oxydecanoyloxy]decanoic acid;hydrate Chemical compound O.OC1C(OC(CC(=O)OC(CCCCCCC)CC(O)=O)CCCCCCC)OC(C)C(O)C1OC1C(O)C(O)C(O)C(C)O1 HVCOBJNICQPDBP-UHFFFAOYSA-N 0.000 claims 2
- 239000005711 Benzoic acid Substances 0.000 claims 2
- UHDGCWIWMRVCDJ-PSQAKQOGSA-N Cytidine Natural products O=C1N=C(N)C=CN1[C@@H]1[C@@H](O)[C@@H](O)[C@H](CO)O1 UHDGCWIWMRVCDJ-PSQAKQOGSA-N 0.000 claims 2
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 claims 2
- 235000010233 benzoic acid Nutrition 0.000 claims 2
- UHDGCWIWMRVCDJ-ZAKLUEHWSA-N cytidine Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O1 UHDGCWIWMRVCDJ-ZAKLUEHWSA-N 0.000 claims 2
- XPPKVPWEQAFLFU-UHFFFAOYSA-J diphosphate(4-) Chemical compound [O-]P([O-])(=O)OP([O-])([O-])=O XPPKVPWEQAFLFU-UHFFFAOYSA-J 0.000 claims 2
- 150000002270 gangliosides Chemical class 0.000 claims 2
- WVDDGKGOMKODPV-ZQBYOMGUSA-N phenyl(114C)methanol Chemical compound O[14CH2]C1=CC=CC=C1 WVDDGKGOMKODPV-ZQBYOMGUSA-N 0.000 claims 2
- 239000011574 phosphorus Substances 0.000 claims 2
- 229910052698 phosphorus Inorganic materials 0.000 claims 2
- 229960003339 sodium phosphate Drugs 0.000 claims 2
- 235000011008 sodium phosphates Nutrition 0.000 claims 2
- 235000019818 tetrasodium diphosphate Nutrition 0.000 claims 2
- UNXRWKVEANCORM-UHFFFAOYSA-N triphosphoric acid Chemical compound OP(O)(=O)OP(O)(=O)OP(O)(O)=O UNXRWKVEANCORM-UHFFFAOYSA-N 0.000 claims 2
- 125000002467 phosphate group Chemical group [H]OP(=O)(O[H])O[*] 0.000 abstract description 3
- 238000010979 pH adjustment Methods 0.000 abstract description 2
- 239000000243 solution Substances 0.000 description 155
- 108700023372 Glycosyltransferases Proteins 0.000 description 94
- 102000051366 Glycosyltransferases Human genes 0.000 description 87
- 229920001223 polyethylene glycol Polymers 0.000 description 82
- 238000003786 synthesis reaction Methods 0.000 description 63
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 60
- 230000015572 biosynthetic process Effects 0.000 description 58
- 150000001720 carbohydrates Chemical class 0.000 description 58
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 54
- 238000006243 chemical reaction Methods 0.000 description 50
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 50
- 238000004587 chromatography analysis Methods 0.000 description 46
- 150000001875 compounds Chemical class 0.000 description 44
- 238000001914 filtration Methods 0.000 description 44
- TXCIAUNLDRJGJZ-BILDWYJOSA-N CMP-N-acetyl-beta-neuraminic acid Chemical compound O1[C@@H]([C@H](O)[C@H](O)CO)[C@H](NC(=O)C)[C@@H](O)C[C@]1(C(O)=O)OP(O)(=O)OC[C@@H]1[C@@H](O)[C@@H](O)[C@H](N2C(N=C(N)C=C2)=O)O1 TXCIAUNLDRJGJZ-BILDWYJOSA-N 0.000 description 43
- 238000000108 ultra-filtration Methods 0.000 description 40
- 235000014633 carbohydrates Nutrition 0.000 description 39
- 239000000047 product Substances 0.000 description 39
- 238000001223 reverse osmosis Methods 0.000 description 39
- 229920001542 oligosaccharide Polymers 0.000 description 30
- 150000002482 oligosaccharides Polymers 0.000 description 30
- 102000004169 proteins and genes Human genes 0.000 description 30
- 108090000623 proteins and genes Proteins 0.000 description 30
- 125000001424 substituent group Chemical group 0.000 description 25
- 238000011084 recovery Methods 0.000 description 24
- IERHLVCPSMICTF-UHFFFAOYSA-N cytidine monophosphate Natural products O=C1N=C(N)C=CN1C1C(O)C(O)C(COP(O)(O)=O)O1 IERHLVCPSMICTF-UHFFFAOYSA-N 0.000 description 23
- 239000012510 hollow fiber Substances 0.000 description 22
- PCDQPRRSZKQHHS-UHFFFAOYSA-N Cytidine 5'-triphosphate Natural products O=C1N=C(N)C=CN1C1C(O)C(O)C(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)O1 PCDQPRRSZKQHHS-UHFFFAOYSA-N 0.000 description 21
- 102000004190 Enzymes Human genes 0.000 description 21
- 108090000790 Enzymes Proteins 0.000 description 21
- PCDQPRRSZKQHHS-ZAKLUEHWSA-N cytidine-5'-triphosphate Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@H](O)[C@@H](O)[C@H](CO[P@](O)(=O)O[P@@](O)(=O)OP(O)(O)=O)O1 PCDQPRRSZKQHHS-ZAKLUEHWSA-N 0.000 description 21
- 229940088598 enzyme Drugs 0.000 description 21
- 239000012527 feed solution Substances 0.000 description 20
- 239000012466 permeate Substances 0.000 description 20
- 229920000642 polymer Polymers 0.000 description 20
- 239000012465 retentate Substances 0.000 description 20
- 238000009295 crossflow filtration Methods 0.000 description 19
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 18
- 229910052588 hydroxylapatite Inorganic materials 0.000 description 17
- 239000000463 material Substances 0.000 description 17
- 229940060155 neuac Drugs 0.000 description 17
- XYJRXVWERLGGKC-UHFFFAOYSA-D pentacalcium;hydroxide;triphosphate Chemical compound [OH-].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O XYJRXVWERLGGKC-UHFFFAOYSA-D 0.000 description 17
- SQVRNKJHWKZAKO-PFQGKNLYSA-N N-acetyl-beta-neuraminic acid Chemical group CC(=O)N[C@@H]1[C@@H](O)C[C@@](O)(C(O)=O)O[C@H]1[C@H](O)[C@H](O)CO SQVRNKJHWKZAKO-PFQGKNLYSA-N 0.000 description 16
- 239000011541 reaction mixture Substances 0.000 description 16
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 15
- WQZGKKKJIJFFOK-FPRJBGLDSA-N beta-D-galactose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-FPRJBGLDSA-N 0.000 description 14
- 239000001257 hydrogen Substances 0.000 description 14
- 229910052739 hydrogen Inorganic materials 0.000 description 14
- 238000000926 separation method Methods 0.000 description 14
- ROSDSFDQCJNGOL-UHFFFAOYSA-N Dimethylamine Chemical compound CNC ROSDSFDQCJNGOL-UHFFFAOYSA-N 0.000 description 13
- LCTONWCANYUPML-UHFFFAOYSA-M Pyruvate Chemical compound CC(=O)C([O-])=O LCTONWCANYUPML-UHFFFAOYSA-M 0.000 description 13
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 13
- 239000002777 nucleoside Substances 0.000 description 13
- 239000004471 Glycine Substances 0.000 description 12
- 102000003838 Sialyltransferases Human genes 0.000 description 12
- 108090000141 Sialyltransferases Proteins 0.000 description 12
- MSWZFWKMSRAUBD-UHFFFAOYSA-N beta-D-galactosamine Natural products NC1C(O)OC(CO)C(O)C1O MSWZFWKMSRAUBD-UHFFFAOYSA-N 0.000 description 12
- 239000003795 chemical substances by application Substances 0.000 description 12
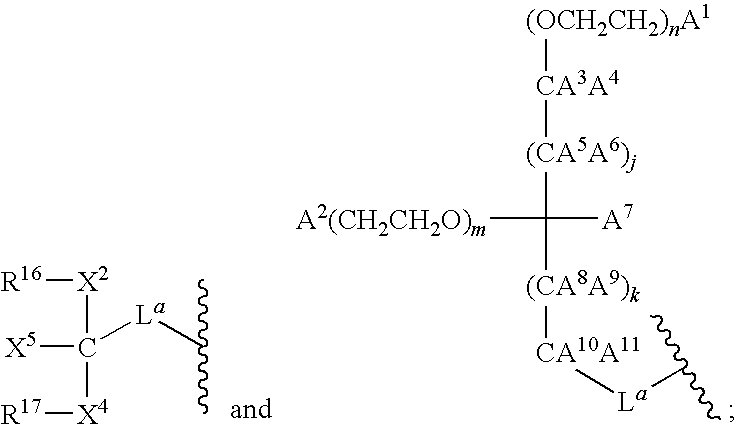
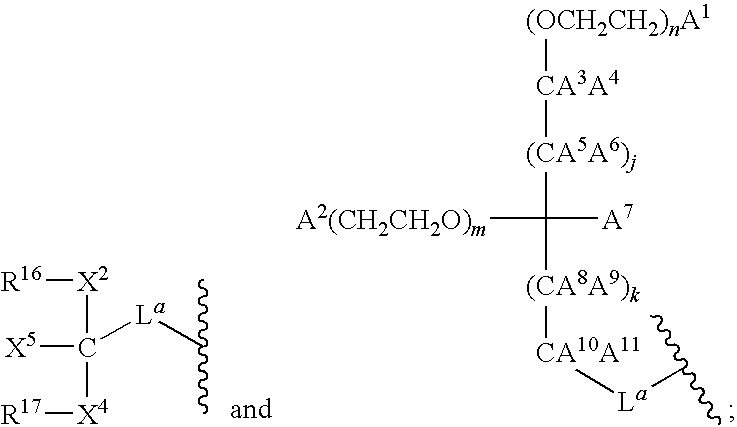
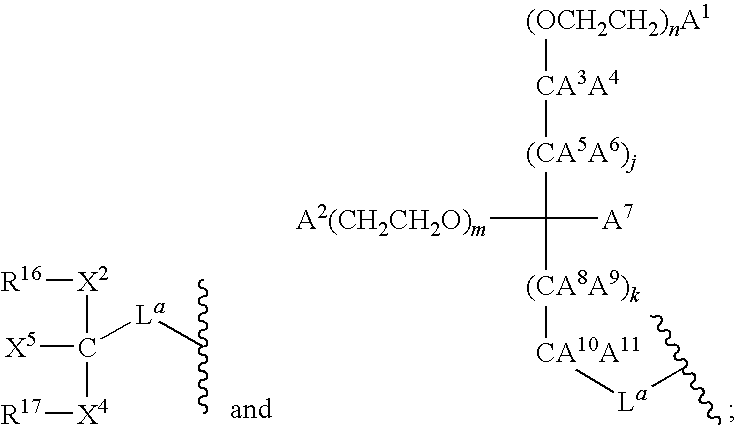
- 125000005647 linker group Chemical group 0.000 description 12
- 239000000843 powder Substances 0.000 description 12
- 150000003254 radicals Chemical class 0.000 description 12
- 239000011347 resin Substances 0.000 description 12
- 229920005989 resin Polymers 0.000 description 12
- OKTJSMMVPCPJKN-UHFFFAOYSA-N Carbon Chemical compound [C] OKTJSMMVPCPJKN-UHFFFAOYSA-N 0.000 description 11
- 230000002255 enzymatic effect Effects 0.000 description 11
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 11
- SQVRNKJHWKZAKO-LUWBGTNYSA-N N-acetylneuraminic acid Chemical compound CC(=O)N[C@@H]1[C@@H](O)CC(O)(C(O)=O)O[C@H]1[C@H](O)[C@H](O)CO SQVRNKJHWKZAKO-LUWBGTNYSA-N 0.000 description 10
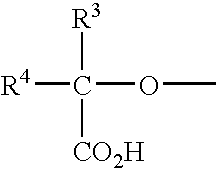
- 0 [3*]C([4*])(OC)C(=O)O Chemical compound [3*]C([4*])(OC)C(=O)O 0.000 description 10
- 229910052799 carbon Inorganic materials 0.000 description 10
- 238000003306 harvesting Methods 0.000 description 10
- 238000004366 reverse phase liquid chromatography Methods 0.000 description 10
- ZWIADYZPOWUWEW-XVFCMESISA-N CDP Chemical compound O=C1N=C(N)C=CN1[C@H]1[C@H](O)[C@H](O)[C@@H](COP(O)(=O)OP(O)(O)=O)O1 ZWIADYZPOWUWEW-XVFCMESISA-N 0.000 description 9
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 9
- 238000007792 addition Methods 0.000 description 9
- 125000003545 alkoxy group Chemical group 0.000 description 9
- 125000002947 alkylene group Chemical group 0.000 description 9
- 125000004432 carbon atom Chemical group C* 0.000 description 9
- 239000002158 endotoxin Substances 0.000 description 9
- CERZMXAJYMMUDR-UHFFFAOYSA-N neuraminic acid Natural products NC1C(O)CC(O)(C(O)=O)OC1C(O)C(O)CO CERZMXAJYMMUDR-UHFFFAOYSA-N 0.000 description 9
- 229910052757 nitrogen Inorganic materials 0.000 description 9
- 238000001556 precipitation Methods 0.000 description 9
- 238000002360 preparation method Methods 0.000 description 9
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 9
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 8
- 239000000499 gel Substances 0.000 description 8
- 125000005842 heteroatom Chemical group 0.000 description 8
- 238000005374 membrane filtration Methods 0.000 description 8
- 239000000126 substance Substances 0.000 description 8
- 150000008163 sugars Chemical class 0.000 description 8
- TXCIAUNLDRJGJZ-UHFFFAOYSA-N CMP-N-acetyl neuraminic acid Natural products O1C(C(O)C(O)CO)C(NC(=O)C)C(O)CC1(C(O)=O)OP(O)(=O)OCC1C(O)C(O)C(N2C(N=C(N)C=C2)=O)O1 TXCIAUNLDRJGJZ-UHFFFAOYSA-N 0.000 description 7
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 7
- 102000001390 Fructose-Bisphosphate Aldolase Human genes 0.000 description 7
- 108010068561 Fructose-Bisphosphate Aldolase Proteins 0.000 description 7
- 239000000370 acceptor Substances 0.000 description 7
- 238000005571 anion exchange chromatography Methods 0.000 description 7
- 150000001721 carbon Chemical group 0.000 description 7
- 238000005277 cation exchange chromatography Methods 0.000 description 7
- 210000004027 cell Anatomy 0.000 description 7
- 238000005352 clarification Methods 0.000 description 7
- 238000010511 deprotection reaction Methods 0.000 description 7
- 238000005516 engineering process Methods 0.000 description 7
- 102000045442 glycosyltransferase activity proteins Human genes 0.000 description 7
- 108700014210 glycosyltransferase activity proteins Proteins 0.000 description 7
- 239000012535 impurity Substances 0.000 description 7
- 150000002500 ions Chemical class 0.000 description 7
- 239000002244 precipitate Substances 0.000 description 7
- 239000008213 purified water Substances 0.000 description 7
- 239000000377 silicon dioxide Substances 0.000 description 7
- 230000001225 therapeutic effect Effects 0.000 description 7
- 125000004209 (C1-C8) alkyl group Chemical group 0.000 description 6
- MSWZFWKMSRAUBD-IVMDWMLBSA-N 2-amino-2-deoxy-D-glucopyranose Chemical compound N[C@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O MSWZFWKMSRAUBD-IVMDWMLBSA-N 0.000 description 6
- ZKHQWZAMYRWXGA-KQYNXXCUSA-J ATP(4-) Chemical compound C1=NC=2C(N)=NC=NC=2N1[C@@H]1O[C@H](COP([O-])(=O)OP([O-])(=O)OP([O-])([O-])=O)[C@@H](O)[C@H]1O ZKHQWZAMYRWXGA-KQYNXXCUSA-J 0.000 description 6
- ZKHQWZAMYRWXGA-UHFFFAOYSA-N Adenosine triphosphate Natural products C1=NC=2C(N)=NC=NC=2N1C1OC(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)C(O)C1O ZKHQWZAMYRWXGA-UHFFFAOYSA-N 0.000 description 6
- FZHXIRIBWMQPQF-UHFFFAOYSA-N Glc-NH2 Natural products O=CC(N)C(O)C(O)C(O)CO FZHXIRIBWMQPQF-UHFFFAOYSA-N 0.000 description 6
- WQDUMFSSJAZKTM-UHFFFAOYSA-N Sodium methoxide Chemical compound [Na+].[O-]C WQDUMFSSJAZKTM-UHFFFAOYSA-N 0.000 description 6
- 239000007864 aqueous solution Substances 0.000 description 6
- 238000011097 chromatography purification Methods 0.000 description 6
- 239000012141 concentrate Substances 0.000 description 6
- 238000011026 diafiltration Methods 0.000 description 6
- 229910052736 halogen Inorganic materials 0.000 description 6
- 238000004128 high performance liquid chromatography Methods 0.000 description 6
- 238000004191 hydrophobic interaction chromatography Methods 0.000 description 6
- 229940062780 lacto-n-neotetraose Drugs 0.000 description 6
- 230000014759 maintenance of location Effects 0.000 description 6
- RBMYDHMFFAVMMM-PLQWBNBWSA-N neolactotetraose Chemical compound O([C@H]1[C@H](O)[C@H]([C@@H](O[C@@H]1CO)O[C@@H]1[C@H]([C@H](O[C@H]([C@H](O)CO)[C@H](O)[C@@H](O)C=O)O[C@H](CO)[C@@H]1O)O)NC(=O)C)[C@@H]1O[C@H](CO)[C@H](O)[C@H](O)[C@H]1O RBMYDHMFFAVMMM-PLQWBNBWSA-N 0.000 description 6
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 6
- 230000002285 radioactive effect Effects 0.000 description 6
- 230000002441 reversible effect Effects 0.000 description 6
- 125000005630 sialyl group Chemical group 0.000 description 6
- 241000894007 species Species 0.000 description 6
- 229910052717 sulfur Inorganic materials 0.000 description 6
- DKVBOUDTNWVDEP-NJCHZNEYSA-N teicoplanin aglycone Chemical group N([C@H](C(N[C@@H](C1=CC(O)=CC(O)=C1C=1C(O)=CC=C2C=1)C(O)=O)=O)[C@H](O)C1=CC=C(C(=C1)Cl)OC=1C=C3C=C(C=1O)OC1=CC=C(C=C1Cl)C[C@H](C(=O)N1)NC([C@H](N)C=4C=C(O5)C(O)=CC=4)=O)C(=O)[C@@H]2NC(=O)[C@@H]3NC(=O)[C@@H]1C1=CC5=CC(O)=C1 DKVBOUDTNWVDEP-NJCHZNEYSA-N 0.000 description 6
- 229920003169 water-soluble polymer Polymers 0.000 description 6
- 125000004178 (C1-C4) alkyl group Chemical group 0.000 description 5
- YPJMOVVQKBFRNH-UHFFFAOYSA-N 1-(9-ethylcarbazol-3-yl)-n-(pyridin-2-ylmethyl)methanamine Chemical compound C=1C=C2N(CC)C3=CC=CC=C3C2=CC=1CNCC1=CC=CC=N1 YPJMOVVQKBFRNH-UHFFFAOYSA-N 0.000 description 5
- 102000030902 Galactosyltransferase Human genes 0.000 description 5
- 108060003306 Galactosyltransferase Proteins 0.000 description 5
- OVRNDRQMDRJTHS-ZTVVOAFPSA-N N-acetyl-D-mannosamine Chemical compound CC(=O)N[C@@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O OVRNDRQMDRJTHS-ZTVVOAFPSA-N 0.000 description 5
- 102000048245 N-acetylneuraminate lyases Human genes 0.000 description 5
- 108700023220 N-acetylneuraminate lyases Proteins 0.000 description 5
- 108091000080 Phosphotransferase Proteins 0.000 description 5
- 229920002684 Sepharose Polymers 0.000 description 5
- 125000003277 amino group Chemical group 0.000 description 5
- 238000004458 analytical method Methods 0.000 description 5
- 238000005349 anion exchange Methods 0.000 description 5
- 239000000872 buffer Substances 0.000 description 5
- 230000015556 catabolic process Effects 0.000 description 5
- 238000011210 chromatographic step Methods 0.000 description 5
- 238000004042 decolorization Methods 0.000 description 5
- 125000004474 heteroalkylene group Chemical group 0.000 description 5
- 125000004356 hydroxy functional group Chemical group O* 0.000 description 5
- 125000003835 nucleoside group Chemical group 0.000 description 5
- 102000020233 phosphotransferase Human genes 0.000 description 5
- 108090000765 processed proteins & peptides Proteins 0.000 description 5
- 239000000376 reactant Substances 0.000 description 5
- 125000005629 sialic acid group Chemical class 0.000 description 5
- 238000002415 sodium dodecyl sulfate polyacrylamide gel electrophoresis Methods 0.000 description 5
- 239000002904 solvent Substances 0.000 description 5
- 230000008685 targeting Effects 0.000 description 5
- LAQPKDLYOBZWBT-NYLDSJSYSA-N (2s,4s,5r,6r)-5-acetamido-2-{[(2s,3r,4s,5s,6r)-2-{[(2r,3r,4r,5r)-5-acetamido-1,2-dihydroxy-6-oxo-4-{[(2s,3s,4r,5s,6s)-3,4,5-trihydroxy-6-methyloxan-2-yl]oxy}hexan-3-yl]oxy}-3,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy}-4-hydroxy-6-[(1r,2r)-1,2,3-trihydrox Chemical compound O[C@H]1[C@H](O)[C@H](O)[C@H](C)O[C@H]1O[C@H]([C@@H](NC(C)=O)C=O)[C@@H]([C@H](O)CO)O[C@H]1[C@H](O)[C@@H](O[C@]2(O[C@H]([C@H](NC(C)=O)[C@@H](O)C2)[C@H](O)[C@H](O)CO)C(O)=O)[C@@H](O)[C@@H](CO)O1 LAQPKDLYOBZWBT-NYLDSJSYSA-N 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical group CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 4
- DGAQECJNVWCQMB-PUAWFVPOSA-M Ilexoside XXIX Chemical compound C[C@@H]1CC[C@@]2(CC[C@@]3(C(=CC[C@H]4[C@]3(CC[C@@H]5[C@@]4(CC[C@@H](C5(C)C)OS(=O)(=O)[O-])C)C)[C@@H]2[C@]1(C)O)C)C(=O)O[C@H]6[C@@H]([C@H]([C@@H]([C@H](O6)CO)O)O)O.[Na+] DGAQECJNVWCQMB-PUAWFVPOSA-M 0.000 description 4
- 239000012614 Q-Sepharose Substances 0.000 description 4
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical compound [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 4
- 125000002252 acyl group Chemical group 0.000 description 4
- 229910002092 carbon dioxide Inorganic materials 0.000 description 4
- 238000005341 cation exchange Methods 0.000 description 4
- 239000000919 ceramic Substances 0.000 description 4
- 229920001577 copolymer Polymers 0.000 description 4
- 125000004122 cyclic group Chemical group 0.000 description 4
- 231100000599 cytotoxic agent Toxicity 0.000 description 4
- 238000010586 diagram Methods 0.000 description 4
- 238000010828 elution Methods 0.000 description 4
- 125000005843 halogen group Chemical group 0.000 description 4
- 150000002367 halogens Chemical class 0.000 description 4
- 229910052740 iodine Inorganic materials 0.000 description 4
- 238000004255 ion exchange chromatography Methods 0.000 description 4
- 238000004519 manufacturing process Methods 0.000 description 4
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 4
- 229910052760 oxygen Inorganic materials 0.000 description 4
- 239000011148 porous material Substances 0.000 description 4
- 102000004196 processed proteins & peptides Human genes 0.000 description 4
- 239000011734 sodium Substances 0.000 description 4
- 229910052708 sodium Inorganic materials 0.000 description 4
- 238000003860 storage Methods 0.000 description 4
- 239000000758 substrate Substances 0.000 description 4
- 238000012360 testing method Methods 0.000 description 4
- 125000002023 trifluoromethyl group Chemical group FC(F)(F)* 0.000 description 4
- 125000006823 (C1-C6) acyl group Chemical group 0.000 description 3
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 3
- 125000003088 (fluoren-9-ylmethoxy)carbonyl group Chemical group 0.000 description 3
- 238000005160 1H NMR spectroscopy Methods 0.000 description 3
- AXAVXPMQTGXXJZ-UHFFFAOYSA-N 2-aminoacetic acid;2-amino-2-(hydroxymethyl)propane-1,3-diol Chemical compound NCC(O)=O.OCC(N)(CO)CO AXAVXPMQTGXXJZ-UHFFFAOYSA-N 0.000 description 3
- ZCYVEMRRCGMTRW-UHFFFAOYSA-N 7553-56-2 Chemical group [I] ZCYVEMRRCGMTRW-UHFFFAOYSA-N 0.000 description 3
- UXVMQQNJUSDDNG-UHFFFAOYSA-L Calcium chloride Chemical compound [Cl-].[Cl-].[Ca+2] UXVMQQNJUSDDNG-UHFFFAOYSA-L 0.000 description 3
- BVKZGUZCCUSVTD-UHFFFAOYSA-L Carbonate Chemical compound [O-]C([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-L 0.000 description 3
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 3
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 3
- 102000003951 Erythropoietin Human genes 0.000 description 3
- 108090000394 Erythropoietin Proteins 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- 102000004269 Granulocyte Colony-Stimulating Factor Human genes 0.000 description 3
- 108010017080 Granulocyte Colony-Stimulating Factor Proteins 0.000 description 3
- 102000004457 Granulocyte-Macrophage Colony-Stimulating Factor Human genes 0.000 description 3
- 108010017213 Granulocyte-Macrophage Colony-Stimulating Factor Proteins 0.000 description 3
- PWHULOQIROXLJO-UHFFFAOYSA-N Manganese Chemical compound [Mn] PWHULOQIROXLJO-UHFFFAOYSA-N 0.000 description 3
- 108010081778 N-acylneuraminate cytidylyltransferase Proteins 0.000 description 3
- FDJKUWYYUZCUJX-KVNVFURPSA-N N-glycolylneuraminic acid Chemical group OC[C@H](O)[C@H](O)[C@@H]1O[C@](O)(C(O)=O)C[C@H](O)[C@H]1NC(=O)CO FDJKUWYYUZCUJX-KVNVFURPSA-N 0.000 description 3
- DTBNBXWJWCWCIK-UHFFFAOYSA-N Phosphoenolpyruvic acid Natural products OC(=O)C(=C)OP(O)(O)=O DTBNBXWJWCWCIK-UHFFFAOYSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 102000003800 Selectins Human genes 0.000 description 3
- 108090000184 Selectins Proteins 0.000 description 3
- 208000027418 Wounds and injury Diseases 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 125000003282 alkyl amino group Chemical group 0.000 description 3
- 125000004414 alkyl thio group Chemical group 0.000 description 3
- 239000002246 antineoplastic agent Substances 0.000 description 3
- 229940041181 antineoplastic drug Drugs 0.000 description 3
- 125000003710 aryl alkyl group Chemical group 0.000 description 3
- 125000004429 atom Chemical group 0.000 description 3
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 3
- 239000006227 byproduct Substances 0.000 description 3
- 239000001110 calcium chloride Substances 0.000 description 3
- 229910001628 calcium chloride Inorganic materials 0.000 description 3
- 125000002915 carbonyl group Chemical group [*:2]C([*:1])=O 0.000 description 3
- 230000003197 catalytic effect Effects 0.000 description 3
- 150000001768 cations Chemical class 0.000 description 3
- 239000003153 chemical reaction reagent Substances 0.000 description 3
- 238000004140 cleaning Methods 0.000 description 3
- 239000002619 cytotoxin Substances 0.000 description 3
- 238000010612 desalination reaction Methods 0.000 description 3
- 150000002016 disaccharides Chemical class 0.000 description 3
- 229940105423 erythropoietin Drugs 0.000 description 3
- 239000012467 final product Substances 0.000 description 3
- 238000003818 flash chromatography Methods 0.000 description 3
- 239000003456 ion exchange resin Substances 0.000 description 3
- 229920003303 ion-exchange polymer Polymers 0.000 description 3
- 239000007788 liquid Substances 0.000 description 3
- 229920002521 macromolecule Polymers 0.000 description 3
- 229910052748 manganese Inorganic materials 0.000 description 3
- 239000011572 manganese Substances 0.000 description 3
- 229910052751 metal Inorganic materials 0.000 description 3
- 239000002184 metal Substances 0.000 description 3
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 3
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 description 3
- 125000004433 nitrogen atom Chemical group N* 0.000 description 3
- 230000003204 osmotic effect Effects 0.000 description 3
- 239000002245 particle Substances 0.000 description 3
- OXCMYAYHXIHQOA-UHFFFAOYSA-N potassium;[2-butyl-5-chloro-3-[[4-[2-(1,2,4-triaza-3-azanidacyclopenta-1,4-dien-5-yl)phenyl]phenyl]methyl]imidazol-4-yl]methanol Chemical compound [K+].CCCCC1=NC(Cl)=C(CO)N1CC1=CC=C(C=2C(=CC=CC=2)C2=N[N-]N=N2)C=C1 OXCMYAYHXIHQOA-UHFFFAOYSA-N 0.000 description 3
- 239000002994 raw material Substances 0.000 description 3
- 239000012429 reaction media Substances 0.000 description 3
- 238000004064 recycling Methods 0.000 description 3
- 230000002829 reductive effect Effects 0.000 description 3
- 229910052710 silicon Inorganic materials 0.000 description 3
- 125000003808 silyl group Chemical group [H][Si]([H])([H])[*] 0.000 description 3
- 239000000600 sorbitol Substances 0.000 description 3
- 239000007921 spray Substances 0.000 description 3
- 239000007858 starting material Substances 0.000 description 3
- 125000000547 substituted alkyl group Chemical group 0.000 description 3
- 125000004434 sulfur atom Chemical group 0.000 description 3
- 230000002194 synthesizing effect Effects 0.000 description 3
- 239000003053 toxin Substances 0.000 description 3
- 231100000765 toxin Toxicity 0.000 description 3
- 108700012359 toxins Proteins 0.000 description 3
- 238000012546 transfer Methods 0.000 description 3
- CBOJBBMQJBVCMW-MVNLRXSJSA-N (2s,3r,4s,5r)-2-amino-3,4,5,6-tetrahydroxyhexanal;hydrochloride Chemical compound Cl.O=C[C@@H](N)[C@@H](O)[C@H](O)[C@H](O)CO CBOJBBMQJBVCMW-MVNLRXSJSA-N 0.000 description 2
- MZOFCQQQCNRIBI-VMXHOPILSA-N (3s)-4-[[(2s)-1-[[(2s)-1-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-methyl-1-oxopentan-2-yl]amino]-5-(diaminomethylideneamino)-1-oxopentan-2-yl]amino]-3-[[2-[[(2s)-2,6-diaminohexanoyl]amino]acetyl]amino]-4-oxobutanoic acid Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCN=C(N)N)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@@H](N)CCCCN MZOFCQQQCNRIBI-VMXHOPILSA-N 0.000 description 2
- 125000006702 (C1-C18) alkyl group Chemical group 0.000 description 2
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 2
- MSWZFWKMSRAUBD-CBPJZXOFSA-N 2-amino-2-deoxy-D-mannopyranose Chemical compound N[C@@H]1C(O)O[C@H](CO)[C@@H](O)[C@@H]1O MSWZFWKMSRAUBD-CBPJZXOFSA-N 0.000 description 2
- BFSVOASYOCHEOV-UHFFFAOYSA-N 2-diethylaminoethanol Chemical compound CCN(CC)CCO BFSVOASYOCHEOV-UHFFFAOYSA-N 0.000 description 2
- INZOTETZQBPBCE-NYLDSJSYSA-N 3-sialyl lewis Chemical compound O[C@H]1[C@H](O)[C@H](O)[C@H](C)O[C@H]1O[C@H]([C@H](O)CO)[C@@H]([C@@H](NC(C)=O)C=O)O[C@H]1[C@H](O)[C@@H](O[C@]2(O[C@H]([C@H](NC(C)=O)[C@@H](O)C2)[C@H](O)[C@H](O)CO)C(O)=O)[C@@H](O)[C@@H](CO)O1 INZOTETZQBPBCE-NYLDSJSYSA-N 0.000 description 2
- OTLNPYWUJOZPPA-UHFFFAOYSA-N 4-nitrobenzoic acid Chemical compound OC(=O)C1=CC=C([N+]([O-])=O)C=C1 OTLNPYWUJOZPPA-UHFFFAOYSA-N 0.000 description 2
- 241000894006 Bacteria Species 0.000 description 2
- 102000004506 Blood Proteins Human genes 0.000 description 2
- 108010017384 Blood Proteins Proteins 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- 102000011022 Chorionic Gonadotropin Human genes 0.000 description 2
- 108010062540 Chorionic Gonadotropin Proteins 0.000 description 2
- AOJJSUZBOXZQNB-TZSSRYMLSA-N Doxorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(=O)CO)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 AOJJSUZBOXZQNB-TZSSRYMLSA-N 0.000 description 2
- 102000015689 E-Selectin Human genes 0.000 description 2
- 108010024212 E-Selectin Proteins 0.000 description 2
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 2
- 241000588724 Escherichia coli Species 0.000 description 2
- 102000012673 Follicle Stimulating Hormone Human genes 0.000 description 2
- 108010079345 Follicle Stimulating Hormone Proteins 0.000 description 2
- MVMSCBBUIHUTGJ-GDJBGNAASA-N GDP-alpha-D-mannose Chemical compound C([C@H]1O[C@H]([C@@H]([C@@H]1O)O)N1C=2N=C(NC(=O)C=2N=C1)N)OP(O)(=O)OP(O)(=O)O[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@@H]1O MVMSCBBUIHUTGJ-GDJBGNAASA-N 0.000 description 2
- LQEBEXMHBLQMDB-JGQUBWHWSA-N GDP-beta-L-fucose Chemical compound O[C@H]1[C@H](O)[C@H](O)[C@H](C)O[C@@H]1OP(O)(=O)OP(O)(=O)OC[C@@H]1[C@@H](O)[C@@H](O)[C@H](N2C3=C(C(NC(N)=N3)=O)N=C2)O1 LQEBEXMHBLQMDB-JGQUBWHWSA-N 0.000 description 2
- 102000003886 Glycoproteins Human genes 0.000 description 2
- 108090000288 Glycoproteins Proteins 0.000 description 2
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 description 2
- 102000014150 Interferons Human genes 0.000 description 2
- 108010050904 Interferons Proteins 0.000 description 2
- CLRLHXKNIYJWAW-UHFFFAOYSA-N KDN Natural products OCC(O)C(O)C1OC(O)(C(O)=O)CC(O)C1O CLRLHXKNIYJWAW-UHFFFAOYSA-N 0.000 description 2
- FDJKUWYYUZCUJX-UHFFFAOYSA-N N-glycolyl-beta-neuraminic acid Natural products OCC(O)C(O)C1OC(O)(C(O)=O)CC(O)C1NC(=O)CO FDJKUWYYUZCUJX-UHFFFAOYSA-N 0.000 description 2
- 241000588650 Neisseria meningitidis Species 0.000 description 2
- 206010028980 Neoplasm Diseases 0.000 description 2
- 239000004952 Polyamide Substances 0.000 description 2
- 239000002202 Polyethylene glycol Substances 0.000 description 2
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 2
- PPBRXRYQALVLMV-UHFFFAOYSA-N Styrene Chemical compound C=CC1=CC=CC=C1 PPBRXRYQALVLMV-UHFFFAOYSA-N 0.000 description 2
- NINIDFKCEFEMDL-UHFFFAOYSA-N Sulfur Chemical compound [S] NINIDFKCEFEMDL-UHFFFAOYSA-N 0.000 description 2
- 108060008682 Tumor Necrosis Factor Proteins 0.000 description 2
- 108060008683 Tumor Necrosis Factor Receptor Proteins 0.000 description 2
- 102000000852 Tumor Necrosis Factor-alpha Human genes 0.000 description 2
- 229910052770 Uranium Inorganic materials 0.000 description 2
- 238000002835 absorbance Methods 0.000 description 2
- 125000000738 acetamido group Chemical group [H]C([H])([H])C(=O)N([H])[*] 0.000 description 2
- RJURFGZVJUQBHK-UHFFFAOYSA-N actinomycin D Natural products CC1OC(=O)C(C(C)C)N(C)C(=O)CN(C)C(=O)C2CCCN2C(=O)C(C(C)C)NC(=O)C1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)NC4C(=O)NC(C(N5CCCC5C(=O)N(C)CC(=O)N(C)C(C(C)C)C(=O)OC4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-UHFFFAOYSA-N 0.000 description 2
- WQZGKKKJIJFFOK-PHYPRBDBSA-N alpha-D-galactose Chemical compound OC[C@H]1O[C@H](O)[C@H](O)[C@@H](O)[C@H]1O WQZGKKKJIJFFOK-PHYPRBDBSA-N 0.000 description 2
- HXXFSFRBOHSIMQ-VFUOTHLCSA-N alpha-D-glucose 1-phosphate Chemical compound OC[C@H]1O[C@H](OP(O)(O)=O)[C@H](O)[C@@H](O)[C@@H]1O HXXFSFRBOHSIMQ-VFUOTHLCSA-N 0.000 description 2
- 150000001412 amines Chemical class 0.000 description 2
- 150000001413 amino acids Chemical class 0.000 description 2
- 239000003242 anti bacterial agent Substances 0.000 description 2
- 229940088710 antibiotic agent Drugs 0.000 description 2
- WDIHJSXYQDMJHN-UHFFFAOYSA-L barium chloride Chemical compound [Cl-].[Cl-].[Ba+2] WDIHJSXYQDMJHN-UHFFFAOYSA-L 0.000 description 2
- 229910001626 barium chloride Inorganic materials 0.000 description 2
- 239000003637 basic solution Substances 0.000 description 2
- 239000011324 bead Substances 0.000 description 2
- 230000008901 benefit Effects 0.000 description 2
- 125000000043 benzamido group Chemical group [H]N([*])C(=O)C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 2
- GDTBXPJZTBHREO-UHFFFAOYSA-N bromine Chemical compound BrBr GDTBXPJZTBHREO-UHFFFAOYSA-N 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 238000006555 catalytic reaction Methods 0.000 description 2
- 239000002738 chelating agent Substances 0.000 description 2
- HGAZMNJKRQFZKS-UHFFFAOYSA-N chloroethene;ethenyl acetate Chemical compound ClC=C.CC(=O)OC=C HGAZMNJKRQFZKS-UHFFFAOYSA-N 0.000 description 2
- 239000012539 chromatography resin Substances 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- 229940126214 compound 3 Drugs 0.000 description 2
- 150000003983 crown ethers Chemical class 0.000 description 2
- 125000000113 cyclohexyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 2
- 238000006731 degradation reaction Methods 0.000 description 2
- 238000011033 desalting Methods 0.000 description 2
- 238000000502 dialysis Methods 0.000 description 2
- 238000001035 drying Methods 0.000 description 2
- 230000007515 enzymatic degradation Effects 0.000 description 2
- 238000006911 enzymatic reaction Methods 0.000 description 2
- 239000000835 fiber Substances 0.000 description 2
- 125000001153 fluoro group Chemical group F* 0.000 description 2
- 238000011010 flushing procedure Methods 0.000 description 2
- 230000004907 flux Effects 0.000 description 2
- 229940028334 follicle stimulating hormone Drugs 0.000 description 2
- 238000004108 freeze drying Methods 0.000 description 2
- 108020001507 fusion proteins Proteins 0.000 description 2
- 102000037865 fusion proteins Human genes 0.000 description 2
- 239000007789 gas Substances 0.000 description 2
- 229950010772 glucose-1-phosphate Drugs 0.000 description 2
- 229930182470 glycoside Natural products 0.000 description 2
- 150000002338 glycosides Chemical class 0.000 description 2
- 125000001188 haloalkyl group Chemical group 0.000 description 2
- 229940084986 human chorionic gonadotropin Drugs 0.000 description 2
- 230000002209 hydrophobic effect Effects 0.000 description 2
- AMWRITDGCCNYAT-UHFFFAOYSA-L hydroxy(oxo)manganese;manganese Chemical compound [Mn].O[Mn]=O.O[Mn]=O AMWRITDGCCNYAT-UHFFFAOYSA-L 0.000 description 2
- 230000003993 interaction Effects 0.000 description 2
- 239000011630 iodine Substances 0.000 description 2
- 238000005342 ion exchange Methods 0.000 description 2
- 210000000265 leukocyte Anatomy 0.000 description 2
- 239000003446 ligand Substances 0.000 description 2
- 229920001427 mPEG Polymers 0.000 description 2
- 229910021645 metal ion Inorganic materials 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 125000004573 morpholin-4-yl group Chemical group N1(CCOCC1)* 0.000 description 2
- 238000005457 optimization Methods 0.000 description 2
- 125000004430 oxygen atom Chemical group O* 0.000 description 2
- 230000037361 pathway Effects 0.000 description 2
- WXZMFSXDPGVJKK-UHFFFAOYSA-N pentaerythritol Chemical compound OCC(CO)(CO)CO WXZMFSXDPGVJKK-UHFFFAOYSA-N 0.000 description 2
- 230000035699 permeability Effects 0.000 description 2
- 229930029653 phosphoenolpyruvate Natural products 0.000 description 2
- 229920002492 poly(sulfone) Polymers 0.000 description 2
- 229920002401 polyacrylamide Polymers 0.000 description 2
- 229920002647 polyamide Polymers 0.000 description 2
- 229920000570 polyether Polymers 0.000 description 2
- 229920001451 polypropylene glycol Polymers 0.000 description 2
- 229920002451 polyvinyl alcohol Polymers 0.000 description 2
- AAEVYOVXGOFMJO-UHFFFAOYSA-N prometryn Chemical compound CSC1=NC(NC(C)C)=NC(NC(C)C)=N1 AAEVYOVXGOFMJO-UHFFFAOYSA-N 0.000 description 2
- AQHHHDLHHXJYJD-UHFFFAOYSA-N propranolol Chemical compound C1=CC=C2C(OCC(O)CNC(C)C)=CC=CC2=C1 AQHHHDLHHXJYJD-UHFFFAOYSA-N 0.000 description 2
- RXWNCPJZOCPEPQ-NVWDDTSBSA-N puromycin Chemical compound C1=CC(OC)=CC=C1C[C@H](N)C(=O)N[C@H]1[C@@H](O)[C@H](N2C3=NC=NC(=C3N=C2)N(C)C)O[C@@H]1CO RXWNCPJZOCPEPQ-NVWDDTSBSA-N 0.000 description 2
- 230000001172 regenerating effect Effects 0.000 description 2
- 238000012552 review Methods 0.000 description 2
- 239000012723 sample buffer Substances 0.000 description 2
- 229920006395 saturated elastomer Polymers 0.000 description 2
- 230000009450 sialylation Effects 0.000 description 2
- 235000017557 sodium bicarbonate Nutrition 0.000 description 2
- 229910000030 sodium bicarbonate Inorganic materials 0.000 description 2
- 239000007787 solid Substances 0.000 description 2
- 239000002594 sorbent Substances 0.000 description 2
- 238000001228 spectrum Methods 0.000 description 2
- 239000011593 sulfur Substances 0.000 description 2
- 125000005309 thioalkoxy group Chemical group 0.000 description 2
- 150000004043 trisaccharides Chemical class 0.000 description 2
- 102000003298 tumor necrosis factor receptor Human genes 0.000 description 2
- 125000004417 unsaturated alkyl group Chemical group 0.000 description 2
- 125000000229 (C1-C4)alkoxy group Chemical group 0.000 description 1
- IAKHMKGGTNLKSZ-INIZCTEOSA-N (S)-colchicine Chemical compound C1([C@@H](NC(C)=O)CC2)=CC(=O)C(OC)=CC=C1C1=C2C=C(OC)C(OC)=C1OC IAKHMKGGTNLKSZ-INIZCTEOSA-N 0.000 description 1
- 125000001637 1-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C(*)=C([H])C([H])=C([H])C2=C1[H] 0.000 description 1
- VSNHCAURESNICA-NJFSPNSNSA-N 1-oxidanylurea Chemical compound N[14C](=O)NO VSNHCAURESNICA-NJFSPNSNSA-N 0.000 description 1
- 125000004214 1-pyrrolidinyl group Chemical group [H]C1([H])N(*)C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000001462 1-pyrrolyl group Chemical group [*]N1C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 125000004206 2,2,2-trifluoroethyl group Chemical group [H]C([H])(*)C(F)(F)F 0.000 description 1
- YGDVXSDNEFDTGV-UHFFFAOYSA-N 2-[6-[bis(carboxymethyl)amino]hexyl-(carboxymethyl)amino]acetic acid Chemical compound OC(=O)CN(CC(O)=O)CCCCCCN(CC(O)=O)CC(O)=O YGDVXSDNEFDTGV-UHFFFAOYSA-N 0.000 description 1
- QKNYBSVHEMOAJP-UHFFFAOYSA-N 2-amino-2-(hydroxymethyl)propane-1,3-diol;hydron;chloride Chemical compound Cl.OCC(N)(CO)CO QKNYBSVHEMOAJP-UHFFFAOYSA-N 0.000 description 1
- 125000004174 2-benzimidazolyl group Chemical group [H]N1C(*)=NC2=C([H])C([H])=C([H])C([H])=C12 0.000 description 1
- 125000002941 2-furyl group Chemical group O1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- 125000001622 2-naphthyl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C(*)C([H])=C([H])C2=C1[H] 0.000 description 1
- 125000000094 2-phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- 125000004105 2-pyridyl group Chemical group N1=C([*])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- 125000000389 2-pyrrolyl group Chemical group [H]N1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- 125000000175 2-thienyl group Chemical group S1C([*])=C([H])C([H])=C1[H] 0.000 description 1
- ZSLUVFAKFWKJRC-IGMARMGPSA-N 232Th Chemical compound [232Th] ZSLUVFAKFWKJRC-IGMARMGPSA-N 0.000 description 1
- ZOOGRGPOEVQQDX-UUOKFMHZSA-N 3',5'-cyclic GMP Chemical compound C([C@H]1O2)OP(O)(=O)O[C@H]1[C@@H](O)[C@@H]2N1C(N=C(NC2=O)N)=C2N=C1 ZOOGRGPOEVQQDX-UUOKFMHZSA-N 0.000 description 1
- 125000000474 3-butynyl group Chemical group [H]C#CC([H])([H])C([H])([H])* 0.000 description 1
- 125000003682 3-furyl group Chemical group O1C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 125000003349 3-pyridyl group Chemical group N1=C([H])C([*])=C([H])C([H])=C1[H] 0.000 description 1
- 125000001397 3-pyrrolyl group Chemical group [H]N1C([H])=C([*])C([H])=C1[H] 0.000 description 1
- 125000001541 3-thienyl group Chemical group S1C([H])=C([*])C([H])=C1[H] 0.000 description 1
- BTJIUGUIPKRLHP-UHFFFAOYSA-N 4-nitrophenol Chemical compound OC1=CC=C([N+]([O-])=O)C=C1 BTJIUGUIPKRLHP-UHFFFAOYSA-N 0.000 description 1
- 125000000339 4-pyridyl group Chemical group N1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 1
- KDDQRKBRJSGMQE-UHFFFAOYSA-N 4-thiazolyl Chemical group [C]1=CSC=N1 KDDQRKBRJSGMQE-UHFFFAOYSA-N 0.000 description 1
- CWDWFSXUQODZGW-UHFFFAOYSA-N 5-thiazolyl Chemical group [C]1=CN=CS1 CWDWFSXUQODZGW-UHFFFAOYSA-N 0.000 description 1
- STQGQHZAVUOBTE-UHFFFAOYSA-N 7-Cyan-hept-2t-en-4,6-diinsaeure Natural products C1=2C(O)=C3C(=O)C=4C(OC)=CC=CC=4C(=O)C3=C(O)C=2CC(O)(C(C)=O)CC1OC1CC(N)C(O)C(C)O1 STQGQHZAVUOBTE-UHFFFAOYSA-N 0.000 description 1
- 108010092060 Acetate kinase Proteins 0.000 description 1
- 102000002281 Adenylate kinase Human genes 0.000 description 1
- 108020000543 Adenylate kinase Proteins 0.000 description 1
- 102000007698 Alcohol dehydrogenase Human genes 0.000 description 1
- 108010021809 Alcohol dehydrogenase Proteins 0.000 description 1
- 108010039627 Aprotinin Proteins 0.000 description 1
- 108010024976 Asparaginase Proteins 0.000 description 1
- 102000015790 Asparaginase Human genes 0.000 description 1
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 1
- 208000023275 Autoimmune disease Diseases 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-M Bicarbonate Chemical compound OC([O-])=O BVKZGUZCCUSVTD-UHFFFAOYSA-M 0.000 description 1
- 102000015081 Blood Coagulation Factors Human genes 0.000 description 1
- 108010039209 Blood Coagulation Factors Proteins 0.000 description 1
- IXLHQKVQARGGLL-PGDFZCNESA-H C.CC(=O)N[C@H]1C([C@H](O)[C@H](O)CO)O[C@](O)(C(=O)O)C[C@H]1O.CC(=O)N[C@H]1C([C@H](O)[C@H](O)CO)O[C@](OP(=O)([O-])OC[C@H]2OC(N3C=CC(N)=NC3=O)[C@@H](O)[C@H]2O)(C(=O)[O-])C[C@H]1O.NC1=NC(=O)N([C@@H]2O[C@H](COP(=O)([O-])CP(=O)([O-])OP(=O)(O)O)[C@H](O)C2O)C=C1.O=P([O-])(O)OP(=O)([O-])O.[Mn+2].[Mn+2].[Na+].[Na+] Chemical compound C.CC(=O)N[C@H]1C([C@H](O)[C@H](O)CO)O[C@](O)(C(=O)O)C[C@H]1O.CC(=O)N[C@H]1C([C@H](O)[C@H](O)CO)O[C@](OP(=O)([O-])OC[C@H]2OC(N3C=CC(N)=NC3=O)[C@@H](O)[C@H]2O)(C(=O)[O-])C[C@H]1O.NC1=NC(=O)N([C@@H]2O[C@H](COP(=O)([O-])CP(=O)([O-])OP(=O)(O)O)[C@H](O)C2O)C=C1.O=P([O-])(O)OP(=O)([O-])O.[Mn+2].[Mn+2].[Na+].[Na+] IXLHQKVQARGGLL-PGDFZCNESA-H 0.000 description 1
- VCYWMMQVJDUUDE-UHFFFAOYSA-N CC(=O)OC1=CC=C([N+](=O)[O-])C=C1.O=C(O)OC1=CC=C([N+](=O)[O-])C=C1.O=C(O)OC1=CC=C([N+](=O)[O-])C=C1 Chemical compound CC(=O)OC1=CC=C([N+](=O)[O-])C=C1.O=C(O)OC1=CC=C([N+](=O)[O-])C=C1.O=C(O)OC1=CC=C([N+](=O)[O-])C=C1 VCYWMMQVJDUUDE-UHFFFAOYSA-N 0.000 description 1
- PNCOXDHQFHBYOH-QMAKXQNDSA-M CC1=NC(=O)N([C@@H]2O[C@H](COP(=O)([O-])O[C@@]3(C(=O)[O-])C[C@@H](O)[C@@H](NC(=O)C[NH3+])C([C@H](O)[C@H](O)CO)O3)[C@H](O)C2O)C=C1.[Na+] Chemical compound CC1=NC(=O)N([C@@H]2O[C@H](COP(=O)([O-])O[C@@]3(C(=O)[O-])C[C@@H](O)[C@@H](NC(=O)C[NH3+])C([C@H](O)[C@H](O)CO)O3)[C@H](O)C2O)C=C1.[Na+] PNCOXDHQFHBYOH-QMAKXQNDSA-M 0.000 description 1
- 102100029962 CMP-N-acetylneuraminate-beta-1,4-galactoside alpha-2,3-sialyltransferase Human genes 0.000 description 1
- 102100031973 CMP-N-acetylneuraminate-beta-galactosamide-alpha-2,3-sialyltransferase 2 Human genes 0.000 description 1
- AMGOTCBFONZEIZ-LHRBTPEVSA-N CNC1C(O)CC(C)(OP(C)(=O)OCC2OC(C)C(O)C2O)OC1[C@H](O)[C@H](O)CO.CNCC(=O)NC1C(O)OC(CO)C(O)C1O Chemical compound CNC1C(O)CC(C)(OP(C)(=O)OCC2OC(C)C(O)C2O)OC1[C@H](O)[C@H](O)CO.CNCC(=O)NC1C(O)OC(CO)C(O)C1O AMGOTCBFONZEIZ-LHRBTPEVSA-N 0.000 description 1
- ASISZYYTTQEYFE-JLWMXSIHSA-N COCCOCCOC(=O)NCC(=O)NC1C(O)CC(C)(C(=O)O)OC1[C@H](O)C(O)CO.COCCOCCOC(=O)OC1=CC=C([N+](=O)[O-])C=C1.Cl.FC=S.NC1(CO)OC(O)CC(O)C1O.NC1=NC(=O)N([C@@H]2OC(CO=[PH](=O)(O)OC3(C(=O)O)CC(O)C(NC(=O)CNC(=O)OCC4C5=C(C=CC=C5)C5=C4C=CC=C5)C([C@H](O)[C@H](O)CO)O3)[C@@H](O)[C@H]2O)C=C1.NCC(=O)NC1C(O)CC(O[PH](=O)(O)=OCC2O[C@@H](N3C=CC(N)=NC3=O)[C@H](O)[C@@H]2O)(C(=O)O)OC1[C@H](O)[C@H](O)CO.O=C(CNC(=O)OCC1C2=C(C=CC=C2)C2=C1C=CC=C2)NC1(CO)OC(O)CC(O)C1O.O=C(CNC(=O)OCC1C2=C(C=CC=C2)C2=C1C=CC=C2)NC1C(O)CC(O)(C(=O)O)OC1[C@H](O)[C@H](O)CO.O=C(CNC(=O)OCC1C2=CC=CC=C2C2=C1C=CC=C2)ON1C(=O)CCC1=O Chemical compound COCCOCCOC(=O)NCC(=O)NC1C(O)CC(C)(C(=O)O)OC1[C@H](O)C(O)CO.COCCOCCOC(=O)OC1=CC=C([N+](=O)[O-])C=C1.Cl.FC=S.NC1(CO)OC(O)CC(O)C1O.NC1=NC(=O)N([C@@H]2OC(CO=[PH](=O)(O)OC3(C(=O)O)CC(O)C(NC(=O)CNC(=O)OCC4C5=C(C=CC=C5)C5=C4C=CC=C5)C([C@H](O)[C@H](O)CO)O3)[C@@H](O)[C@H]2O)C=C1.NCC(=O)NC1C(O)CC(O[PH](=O)(O)=OCC2O[C@@H](N3C=CC(N)=NC3=O)[C@H](O)[C@@H]2O)(C(=O)O)OC1[C@H](O)[C@H](O)CO.O=C(CNC(=O)OCC1C2=C(C=CC=C2)C2=C1C=CC=C2)NC1(CO)OC(O)CC(O)C1O.O=C(CNC(=O)OCC1C2=C(C=CC=C2)C2=C1C=CC=C2)NC1C(O)CC(O)(C(=O)O)OC1[C@H](O)[C@H](O)CO.O=C(CNC(=O)OCC1C2=CC=CC=C2C2=C1C=CC=C2)ON1C(=O)CCC1=O ASISZYYTTQEYFE-JLWMXSIHSA-N 0.000 description 1
- GIZNRQRZWVPOKN-RQILIBJBSA-L COCCOCCOC(=O)NCC(=O)N[C@H]1C([C@H](O)[C@H](O)CO)O[C@](OP(=O)([O-2])OCC2O[C@@H](N3C=CC(C)=NC3=O)[C@H](O)[C@@H]2O)(C(=O)[O-])C[C@H]1O.COCCOCCOC(=O)OC1=CC=C([N+](=O)[O-])C=C1.NCC(=O)N[C@H]1C([C@H](O)[C@H](O)CO)O[C@](OP(=O)([O-2])OCC2O[C@@H](N3C=CC(N)=NC3=O)[C@H](O)[C@@H]2O)(C(=O)[O-])C[C@H]1O.[Na+].[Na+].[NaH].[NaH] Chemical compound COCCOCCOC(=O)NCC(=O)N[C@H]1C([C@H](O)[C@H](O)CO)O[C@](OP(=O)([O-2])OCC2O[C@@H](N3C=CC(C)=NC3=O)[C@H](O)[C@@H]2O)(C(=O)[O-])C[C@H]1O.COCCOCCOC(=O)OC1=CC=C([N+](=O)[O-])C=C1.NCC(=O)N[C@H]1C([C@H](O)[C@H](O)CO)O[C@](OP(=O)([O-2])OCC2O[C@@H](N3C=CC(N)=NC3=O)[C@H](O)[C@@H]2O)(C(=O)[O-])C[C@H]1O.[Na+].[Na+].[NaH].[NaH] GIZNRQRZWVPOKN-RQILIBJBSA-L 0.000 description 1
- KXDHJXZQYSOELW-UHFFFAOYSA-M Carbamate Chemical compound NC([O-])=O KXDHJXZQYSOELW-UHFFFAOYSA-M 0.000 description 1
- 102000003846 Carbonic anhydrases Human genes 0.000 description 1
- 108090000209 Carbonic anhydrases Proteins 0.000 description 1
- KZBUYRJDOAKODT-UHFFFAOYSA-N Chlorine Chemical compound ClCl KZBUYRJDOAKODT-UHFFFAOYSA-N 0.000 description 1
- GUTLYIVDDKVIGB-OUBTZVSYSA-N Cobalt-60 Chemical compound [60Co] GUTLYIVDDKVIGB-OUBTZVSYSA-N 0.000 description 1
- 229920000858 Cyclodextrin Polymers 0.000 description 1
- 101710112752 Cytotoxin Proteins 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- 108010092160 Dactinomycin Proteins 0.000 description 1
- 101710088194 Dehydrogenase Proteins 0.000 description 1
- MBYXEBXZARTUSS-QLWBXOBMSA-N Emetamine Natural products O(C)c1c(OC)cc2c(c(C[C@@H]3[C@H](CC)CN4[C@H](c5c(cc(OC)c(OC)c5)CC4)C3)ncc2)c1 MBYXEBXZARTUSS-QLWBXOBMSA-N 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 description 1
- PXGOKWXKJXAPGV-UHFFFAOYSA-N Fluorine Chemical compound FF PXGOKWXKJXAPGV-UHFFFAOYSA-N 0.000 description 1
- 108010026389 Gramicidin Proteins 0.000 description 1
- 238000010268 HPLC based assay Methods 0.000 description 1
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 1
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 1
- 102000004877 Insulin Human genes 0.000 description 1
- 108090001061 Insulin Proteins 0.000 description 1
- 102000006992 Interferon-alpha Human genes 0.000 description 1
- 108010047761 Interferon-alpha Proteins 0.000 description 1
- 102000000588 Interleukin-2 Human genes 0.000 description 1
- 108010002350 Interleukin-2 Proteins 0.000 description 1
- 102000015696 Interleukins Human genes 0.000 description 1
- 108010063738 Interleukins Proteins 0.000 description 1
- 108090000862 Ion Channels Proteins 0.000 description 1
- 102000004310 Ion Channels Human genes 0.000 description 1
- 206010065973 Iron Overload Diseases 0.000 description 1
- PNIWLNAGKUGXDO-UHFFFAOYSA-N Lactosamine Natural products OC1C(N)C(O)OC(CO)C1OC1C(O)C(O)C(O)C(CO)O1 PNIWLNAGKUGXDO-UHFFFAOYSA-N 0.000 description 1
- NNJVILVZKWQKPM-UHFFFAOYSA-N Lidocaine Chemical compound CCN(CC)CC(=O)NC1=C(C)C=CC=C1C NNJVILVZKWQKPM-UHFFFAOYSA-N 0.000 description 1
- 239000004472 Lysine Substances 0.000 description 1
- KDXKERNSBIXSRK-UHFFFAOYSA-N Lysine Natural products NCCCCC(N)C(O)=O KDXKERNSBIXSRK-UHFFFAOYSA-N 0.000 description 1
- 102000007651 Macrophage Colony-Stimulating Factor Human genes 0.000 description 1
- 108010046938 Macrophage Colony-Stimulating Factor Proteins 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- 229930192392 Mitomycin Natural products 0.000 description 1
- 102000016943 Muramidase Human genes 0.000 description 1
- 108010014251 Muramidase Proteins 0.000 description 1
- 102000003505 Myosin Human genes 0.000 description 1
- 108060008487 Myosin Proteins 0.000 description 1
- NWIBSHFKIJFRCO-WUDYKRTCSA-N Mytomycin Chemical compound C1N2C(C(C(C)=C(N)C3=O)=O)=C3[C@@H](COC(N)=O)[C@@]2(OC)[C@@H]2[C@H]1N2 NWIBSHFKIJFRCO-WUDYKRTCSA-N 0.000 description 1
- QPCDCPDFJACHGM-UHFFFAOYSA-N N,N-bis{2-[bis(carboxymethyl)amino]ethyl}glycine Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(=O)O)CCN(CC(O)=O)CC(O)=O QPCDCPDFJACHGM-UHFFFAOYSA-N 0.000 description 1
- 108010062010 N-Acetylmuramoyl-L-alanine Amidase Proteins 0.000 description 1
- 125000003047 N-acetyl group Chemical group 0.000 description 1
- OVRNDRQMDRJTHS-KEWYIRBNSA-N N-acetyl-D-galactosamine Chemical compound CC(=O)N[C@H]1C(O)O[C@H](CO)[C@H](O)[C@@H]1O OVRNDRQMDRJTHS-KEWYIRBNSA-N 0.000 description 1
- MBLBDJOUHNCFQT-UHFFFAOYSA-N N-acetyl-D-galactosamine Natural products CC(=O)NC(C=O)C(O)C(O)C(O)CO MBLBDJOUHNCFQT-UHFFFAOYSA-N 0.000 description 1
- OVRNDRQMDRJTHS-OZRXBMAMSA-N N-acetyl-beta-D-mannosamine Chemical compound CC(=O)N[C@@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O OVRNDRQMDRJTHS-OZRXBMAMSA-N 0.000 description 1
- 108010015197 N-acetyllactosaminide alpha-2,3-sialyltransferase Proteins 0.000 description 1
- FDJKUWYYUZCUJX-AJKRCSPLSA-N N-glycoloyl-beta-neuraminic acid Chemical compound OC[C@@H](O)[C@@H](O)[C@@H]1O[C@](O)(C(O)=O)C[C@H](O)[C@H]1NC(=O)CO FDJKUWYYUZCUJX-AJKRCSPLSA-N 0.000 description 1
- SUHQNCLNRUAGOO-UHFFFAOYSA-N N-glycoloyl-neuraminic acid Natural products OCC(O)C(O)C(O)C(NC(=O)CO)C(O)CC(=O)C(O)=O SUHQNCLNRUAGOO-UHFFFAOYSA-N 0.000 description 1
- 238000005481 NMR spectroscopy Methods 0.000 description 1
- 241000588653 Neisseria Species 0.000 description 1
- 229910003849 O-Si Inorganic materials 0.000 description 1
- 229910003872 O—Si Inorganic materials 0.000 description 1
- 102000008212 P-Selectin Human genes 0.000 description 1
- 108010035766 P-Selectin Proteins 0.000 description 1
- 101710137390 P-selectin glycoprotein ligand 1 Proteins 0.000 description 1
- 102100034925 P-selectin glycoprotein ligand 1 Human genes 0.000 description 1
- 229930012538 Paclitaxel Natural products 0.000 description 1
- 229920002145 PharMed Polymers 0.000 description 1
- 102000004160 Phosphoric Monoester Hydrolases Human genes 0.000 description 1
- 108090000608 Phosphoric Monoester Hydrolases Proteins 0.000 description 1
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical class OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 1
- 102000009097 Phosphorylases Human genes 0.000 description 1
- 108010073135 Phosphorylases Proteins 0.000 description 1
- 239000004695 Polyether sulfone Substances 0.000 description 1
- 229920002873 Polyethylenimine Polymers 0.000 description 1
- 239000004743 Polypropylene Substances 0.000 description 1
- 229920001213 Polysorbate 20 Polymers 0.000 description 1
- 239000004793 Polystyrene Substances 0.000 description 1
- 239000004372 Polyvinyl alcohol Substances 0.000 description 1
- 102000013009 Pyruvate Kinase Human genes 0.000 description 1
- 108020005115 Pyruvate Kinase Proteins 0.000 description 1
- MUPFEKGTMRGPLJ-RMMQSMQOSA-N Raffinose Natural products O(C[C@H]1[C@@H](O)[C@H](O)[C@@H](O)[C@@H](O[C@@]2(CO)[C@H](O)[C@@H](O)[C@@H](CO)O2)O1)[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 MUPFEKGTMRGPLJ-RMMQSMQOSA-N 0.000 description 1
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 1
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 1
- 108010039491 Ricin Proteins 0.000 description 1
- AUVVAXYIELKVAI-UHFFFAOYSA-N SJ000285215 Natural products N1CCC2=CC(OC)=C(OC)C=C2C1CC1CC2C3=CC(OC)=C(OC)C=C3CCN2CC1CC AUVVAXYIELKVAI-UHFFFAOYSA-N 0.000 description 1
- 241000607142 Salmonella Species 0.000 description 1
- 229920005654 Sephadex Polymers 0.000 description 1
- 239000012507 Sephadex™ Substances 0.000 description 1
- 229910007161 Si(CH3)3 Inorganic materials 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- BQCADISMDOOEFD-UHFFFAOYSA-N Silver Chemical compound [Ag] BQCADISMDOOEFD-UHFFFAOYSA-N 0.000 description 1
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 1
- UQZIYBXSHAGNOE-USOSMYMVSA-N Stachyose Natural products O(C[C@H]1[C@@H](O)[C@H](O)[C@H](O)[C@@H](O[C@@]2(CO)[C@H](O)[C@@H](O)[C@@H](CO)O2)O1)[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@H](CO[C@@H]2[C@@H](O)[C@@H](O)[C@@H](O)[C@H](CO)O2)O1 UQZIYBXSHAGNOE-USOSMYMVSA-N 0.000 description 1
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 description 1
- 229930006000 Sucrose Natural products 0.000 description 1
- NSOXQYCFHDMMGV-UHFFFAOYSA-N Tetrakis(2-hydroxypropyl)ethylenediamine Chemical compound CC(O)CN(CC(C)O)CCN(CC(C)O)CC(C)O NSOXQYCFHDMMGV-UHFFFAOYSA-N 0.000 description 1
- WDLRUFUQRNWCPK-UHFFFAOYSA-N Tetraxetan Chemical compound OC(=O)CN1CCN(CC(O)=O)CCN(CC(O)=O)CCN(CC(O)=O)CC1 WDLRUFUQRNWCPK-UHFFFAOYSA-N 0.000 description 1
- 229910052776 Thorium Inorganic materials 0.000 description 1
- 102000004338 Transferrin Human genes 0.000 description 1
- 108090000901 Transferrin Proteins 0.000 description 1
- 102100021436 UDP-glucose 4-epimerase Human genes 0.000 description 1
- 108010075202 UDP-glucose 4-epimerase Proteins 0.000 description 1
- GBOGMAARMMDZGR-UHFFFAOYSA-N UNPD149280 Natural products N1C(=O)C23OC(=O)C=CC(O)CCCC(C)CC=CC3C(O)C(=C)C(C)C2C1CC1=CC=CC=C1 GBOGMAARMMDZGR-UHFFFAOYSA-N 0.000 description 1
- MUPFEKGTMRGPLJ-UHFFFAOYSA-N UNPD196149 Natural products OC1C(O)C(CO)OC1(CO)OC1C(O)C(O)C(O)C(COC2C(C(O)C(O)C(CO)O2)O)O1 MUPFEKGTMRGPLJ-UHFFFAOYSA-N 0.000 description 1
- AXQLFFDZXPOFPO-UHFFFAOYSA-N UNPD216 Natural products O1C(CO)C(O)C(OC2C(C(O)C(O)C(CO)O2)O)C(NC(=O)C)C1OC(C1O)C(O)C(CO)OC1OC1C(O)C(O)C(O)OC1CO AXQLFFDZXPOFPO-UHFFFAOYSA-N 0.000 description 1
- 208000025865 Ulcer Diseases 0.000 description 1
- 241000607626 Vibrio cholerae Species 0.000 description 1
- JXLYSJRDGCGARV-WWYNWVTFSA-N Vinblastine Natural products O=C(O[C@H]1[C@](O)(C(=O)OC)[C@@H]2N(C)c3c(cc(c(OC)c3)[C@]3(C(=O)OC)c4[nH]c5c(c4CCN4C[C@](O)(CC)C[C@H](C3)C4)cccc5)[C@@]32[C@H]2[C@@]1(CC)C=CCN2CC3)C JXLYSJRDGCGARV-WWYNWVTFSA-N 0.000 description 1
- LIPOUNRJVLNBCD-UHFFFAOYSA-N acetyl dihydrogen phosphate Chemical compound CC(=O)OP(O)(O)=O LIPOUNRJVLNBCD-UHFFFAOYSA-N 0.000 description 1
- 230000002378 acidificating effect Effects 0.000 description 1
- RJURFGZVJUQBHK-IIXSONLDSA-N actinomycin D Chemical compound C[C@H]1OC(=O)[C@H](C(C)C)N(C)C(=O)CN(C)C(=O)[C@@H]2CCCN2C(=O)[C@@H](C(C)C)NC(=O)[C@H]1NC(=O)C1=C(N)C(=O)C(C)=C2OC(C(C)=CC=C3C(=O)N[C@@H]4C(=O)N[C@@H](C(N5CCC[C@H]5C(=O)N(C)CC(=O)N(C)[C@@H](C(C)C)C(=O)O[C@@H]4C)=O)C(C)C)=C3N=C21 RJURFGZVJUQBHK-IIXSONLDSA-N 0.000 description 1
- 125000004423 acyloxy group Chemical group 0.000 description 1
- 230000002730 additional effect Effects 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- GFFGJBXGBJISGV-UHFFFAOYSA-N adenyl group Chemical class N1=CN=C2N=CNC2=C1N GFFGJBXGBJISGV-UHFFFAOYSA-N 0.000 description 1
- 238000013019 agitation Methods 0.000 description 1
- 150000001335 aliphatic alkanes Chemical class 0.000 description 1
- 125000003342 alkenyl group Chemical group 0.000 description 1
- 125000003302 alkenyloxy group Chemical group 0.000 description 1
- 239000002168 alkylating agent Substances 0.000 description 1
- 229940100198 alkylating agent Drugs 0.000 description 1
- 125000005237 alkyleneamino group Chemical group 0.000 description 1
- 125000005238 alkylenediamino group Chemical group 0.000 description 1
- 125000005530 alkylenedioxy group Chemical group 0.000 description 1
- 125000005529 alkyleneoxy group Chemical group 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- ODDPRQJTYDIWJU-HLICEBNFSA-N alpha-D-Gal-(1->3)-beta-D-Gal-(1->4)-D-Glc Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@@H](O)[C@H](O[C@@H]2[C@H](OC(O)[C@H](O)[C@H]2O)CO)O[C@H](CO)[C@@H]1O ODDPRQJTYDIWJU-HLICEBNFSA-N 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- BFNBIHQBYMNNAN-UHFFFAOYSA-N ammonium sulfate Chemical compound N.N.OS(O)(=O)=O BFNBIHQBYMNNAN-UHFFFAOYSA-N 0.000 description 1
- 229910052921 ammonium sulfate Inorganic materials 0.000 description 1
- 235000011130 ammonium sulphate Nutrition 0.000 description 1
- 239000003957 anion exchange resin Substances 0.000 description 1
- 150000001450 anions Chemical class 0.000 description 1
- 229940045799 anthracyclines and related substance Drugs 0.000 description 1
- 229940121363 anti-inflammatory agent Drugs 0.000 description 1
- 239000002260 anti-inflammatory agent Substances 0.000 description 1
- 230000000340 anti-metabolite Effects 0.000 description 1
- 230000000259 anti-tumor effect Effects 0.000 description 1
- 229940100197 antimetabolite Drugs 0.000 description 1
- 239000002256 antimetabolite Substances 0.000 description 1
- 239000003080 antimitotic agent Substances 0.000 description 1
- 229960004405 aprotinin Drugs 0.000 description 1
- 206010003246 arthritis Diseases 0.000 description 1
- 125000005165 aryl thioxy group Chemical group 0.000 description 1
- 125000004104 aryloxy group Chemical group 0.000 description 1
- 229960003272 asparaginase Drugs 0.000 description 1
- DCXYFEDJOCDNAF-UHFFFAOYSA-M asparaginate Chemical compound [O-]C(=O)C(N)CC(N)=O DCXYFEDJOCDNAF-UHFFFAOYSA-M 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- 125000000852 azido group Chemical group *N=[N+]=[N-] 0.000 description 1
- 239000002585 base Substances 0.000 description 1
- 125000000440 benzylamino group Chemical group [H]N(*)C([H])([H])C1=C([H])C([H])=C([H])C([H])=C1[H] 0.000 description 1
- AXQLFFDZXPOFPO-UNTPKZLMSA-N beta-D-Galp-(1->3)-beta-D-GlcpNAc-(1->3)-beta-D-Galp-(1->4)-beta-D-Glcp Chemical compound O([C@@H]1O[C@H](CO)[C@H](O)[C@@H]([C@H]1O)O[C@H]1[C@@H]([C@H]([C@H](O)[C@@H](CO)O1)O[C@H]1[C@@H]([C@@H](O)[C@@H](O)[C@@H](CO)O1)O)NC(=O)C)[C@H]1[C@H](O)[C@@H](O)[C@H](O)O[C@@H]1CO AXQLFFDZXPOFPO-UNTPKZLMSA-N 0.000 description 1
- KFEUJDWYNGMDBV-RPHKZZMBSA-N beta-D-Galp-(1->4)-D-GlcpNAc Chemical compound O[C@@H]1[C@@H](NC(=O)C)C(O)O[C@H](CO)[C@H]1O[C@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 KFEUJDWYNGMDBV-RPHKZZMBSA-N 0.000 description 1
- 108010057005 beta-galactoside alpha-2,3-sialyltransferase Proteins 0.000 description 1
- DLRVVLDZNNYCBX-ZZFZYMBESA-N beta-melibiose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1OC[C@@H]1[C@@H](O)[C@H](O)[C@@H](O)[C@H](O)O1 DLRVVLDZNNYCBX-ZZFZYMBESA-N 0.000 description 1
- 239000012867 bioactive agent Substances 0.000 description 1
- 125000000319 biphenyl-4-yl group Chemical group [H]C1=C([H])C([H])=C([H])C([H])=C1C1=C([H])C([H])=C([*])C([H])=C1[H] 0.000 description 1
- OWMVSZAMULFTJU-UHFFFAOYSA-N bis-tris Chemical compound OCCN(CCO)C(CO)(CO)CO OWMVSZAMULFTJU-UHFFFAOYSA-N 0.000 description 1
- 239000003114 blood coagulation factor Substances 0.000 description 1
- RSIHSRDYCUFFLA-DYKIIFRCSA-N boldenone Chemical compound O=C1C=C[C@]2(C)[C@H]3CC[C@](C)([C@H](CC4)O)[C@@H]4[C@@H]3CCC2=C1 RSIHSRDYCUFFLA-DYKIIFRCSA-N 0.000 description 1
- 229910052794 bromium Inorganic materials 0.000 description 1
- MTAZNLWOLGHBHU-UHFFFAOYSA-N butadiene-styrene rubber Chemical compound C=CC=C.C=CC1=CC=CC=C1 MTAZNLWOLGHBHU-UHFFFAOYSA-N 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 201000011510 cancer Diseases 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- BVKZGUZCCUSVTD-UHFFFAOYSA-N carbonic acid Chemical group OC(O)=O BVKZGUZCCUSVTD-UHFFFAOYSA-N 0.000 description 1
- 239000003054 catalyst Substances 0.000 description 1
- 239000003518 caustics Substances 0.000 description 1
- 238000004113 cell culture Methods 0.000 description 1
- 210000002421 cell wall Anatomy 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 229920002301 cellulose acetate Polymers 0.000 description 1
- 230000008859 change Effects 0.000 description 1
- NDAYQJDHGXTBJL-MWWSRJDJSA-N chembl557217 Chemical compound C1=CC=C2C(C[C@H](NC(=O)[C@@H](CC(C)C)NC(=O)[C@H](CC=3C4=CC=CC=C4NC=3)NC(=O)[C@@H](CC(C)C)NC(=O)[C@H](CC=3C4=CC=CC=C4NC=3)NC(=O)[C@@H](CC(C)C)NC(=O)[C@H](CC=3C4=CC=CC=C4NC=3)NC(=O)[C@@H](C(C)C)NC(=O)[C@H](C(C)C)NC(=O)[C@@H](C(C)C)NC(=O)[C@H](C)NC(=O)[C@H](NC(=O)CNC(=O)[C@@H](NC=O)C(C)C)CC(C)C)C(=O)NCCO)=CNC2=C1 NDAYQJDHGXTBJL-MWWSRJDJSA-N 0.000 description 1
- 238000003889 chemical engineering Methods 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 229910052801 chlorine Inorganic materials 0.000 description 1
- 239000000460 chlorine Substances 0.000 description 1
- 125000002668 chloroacetyl group Chemical group ClCC(=O)* 0.000 description 1
- 239000012459 cleaning agent Substances 0.000 description 1
- 239000002642 cobra venom Substances 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 229940125782 compound 2 Drugs 0.000 description 1
- 238000010276 construction Methods 0.000 description 1
- 239000003246 corticosteroid Substances 0.000 description 1
- 229960001334 corticosteroids Drugs 0.000 description 1
- 239000006184 cosolvent Substances 0.000 description 1
- 230000008878 coupling Effects 0.000 description 1
- 238000010168 coupling process Methods 0.000 description 1
- 238000005859 coupling reaction Methods 0.000 description 1
- 239000012043 crude product Substances 0.000 description 1
- 239000002739 cryptand Substances 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 125000000392 cycloalkenyl group Chemical group 0.000 description 1
- 229940097362 cyclodextrins Drugs 0.000 description 1
- 125000000582 cycloheptyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])([H])C([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 125000001511 cyclopentyl group Chemical group [H]C1([H])C([H])([H])C([H])([H])C([H])(*)C1([H])[H] 0.000 description 1
- 125000004186 cyclopropylmethyl group Chemical group [H]C([H])(*)C1([H])C([H])([H])C1([H])[H] 0.000 description 1
- GBOGMAARMMDZGR-TYHYBEHESA-N cytochalasin B Chemical compound C([C@H]1[C@@H]2[C@@H](C([C@@H](O)[C@@H]3/C=C/C[C@H](C)CCC[C@@H](O)/C=C/C(=O)O[C@@]23C(=O)N1)=C)C)C1=CC=CC=C1 GBOGMAARMMDZGR-TYHYBEHESA-N 0.000 description 1
- GBOGMAARMMDZGR-JREHFAHYSA-N cytochalasin B Natural products C[C@H]1CCC[C@@H](O)C=CC(=O)O[C@@]23[C@H](C=CC1)[C@H](O)C(=C)[C@@H](C)[C@@H]2[C@H](Cc4ccccc4)NC3=O GBOGMAARMMDZGR-JREHFAHYSA-N 0.000 description 1
- 231100000433 cytotoxic Toxicity 0.000 description 1
- 229940127089 cytotoxic agent Drugs 0.000 description 1
- 239000002254 cytotoxic agent Substances 0.000 description 1
- 230000001472 cytotoxic effect Effects 0.000 description 1
- 229960000640 dactinomycin Drugs 0.000 description 1
- 229960000975 daunorubicin Drugs 0.000 description 1
- STQGQHZAVUOBTE-VGBVRHCVSA-N daunorubicin Chemical compound O([C@H]1C[C@@](O)(CC=2C(O)=C3C(=O)C=4C=CC=C(C=4C(=O)C3=C(O)C=21)OC)C(C)=O)[C@H]1C[C@H](N)[C@H](O)[C@H](C)O1 STQGQHZAVUOBTE-VGBVRHCVSA-N 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- RSIHSRDYCUFFLA-UHFFFAOYSA-N dehydrotestosterone Natural products O=C1C=CC2(C)C3CCC(C)(C(CC4)O)C4C3CCC2=C1 RSIHSRDYCUFFLA-UHFFFAOYSA-N 0.000 description 1
- CFCUWKMKBJTWLW-UHFFFAOYSA-N deoliosyl-3C-alpha-L-digitoxosyl-MTM Natural products CC=1C(O)=C2C(O)=C3C(=O)C(OC4OC(C)C(O)C(OC5OC(C)C(O)C(OC6OC(C)C(O)C(C)(O)C6)C5)C4)C(C(OC)C(=O)C(O)C(C)O)CC3=CC2=CC=1OC(OC(C)C1O)CC1OC1CC(O)C(O)C(C)O1 CFCUWKMKBJTWLW-UHFFFAOYSA-N 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 238000001514 detection method Methods 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 150000004656 dimethylamines Chemical class 0.000 description 1
- 238000011143 downstream manufacturing Methods 0.000 description 1
- 229960004679 doxorubicin Drugs 0.000 description 1
- 229960005501 duocarmycin Drugs 0.000 description 1
- 229930184221 duocarmycin Natural products 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 239000003480 eluent Substances 0.000 description 1
- AUVVAXYIELKVAI-CKBKHPSWSA-N emetine Chemical compound N1CCC2=CC(OC)=C(OC)C=C2[C@H]1C[C@H]1C[C@H]2C3=CC(OC)=C(OC)C=C3CCN2C[C@@H]1CC AUVVAXYIELKVAI-CKBKHPSWSA-N 0.000 description 1
- 229960002694 emetine Drugs 0.000 description 1
- AUVVAXYIELKVAI-UWBTVBNJSA-N emetine Natural products N1CCC2=CC(OC)=C(OC)C=C2[C@H]1C[C@H]1C[C@H]2C3=CC(OC)=C(OC)C=C3CCN2C[C@H]1CC AUVVAXYIELKVAI-UWBTVBNJSA-N 0.000 description 1
- 239000000147 enterotoxin Substances 0.000 description 1
- 231100000655 enterotoxin Toxicity 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- ZMMJGEGLRURXTF-UHFFFAOYSA-N ethidium bromide Chemical compound [Br-].C12=CC(N)=CC=C2C2=CC=C(N)C=C2[N+](CC)=C1C1=CC=CC=C1 ZMMJGEGLRURXTF-UHFFFAOYSA-N 0.000 description 1
- 229960005542 ethidium bromide Drugs 0.000 description 1
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- VJJPUSNTGOMMGY-MRVIYFEKSA-N etoposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@H](C)OC[C@H]4O3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 VJJPUSNTGOMMGY-MRVIYFEKSA-N 0.000 description 1
- 229960005420 etoposide Drugs 0.000 description 1
- 238000001704 evaporation Methods 0.000 description 1
- 230000008020 evaporation Effects 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 239000012065 filter cake Substances 0.000 description 1
- 239000000706 filtrate Substances 0.000 description 1
- 238000013100 final test Methods 0.000 description 1
- 229910052731 fluorine Inorganic materials 0.000 description 1
- 239000011737 fluorine Substances 0.000 description 1
- 235000013305 food Nutrition 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 238000007306 functionalization reaction Methods 0.000 description 1
- 125000002519 galactosyl group Chemical group C1([C@H](O)[C@@H](O)[C@@H](O)[C@H](O1)CO)* 0.000 description 1
- 150000008195 galaktosides Chemical class 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 239000003862 glucocorticoid Substances 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- 229910001385 heavy metal Inorganic materials 0.000 description 1
- 125000000623 heterocyclic group Chemical group 0.000 description 1
- 125000004366 heterocycloalkenyl group Chemical group 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- 125000001183 hydrocarbyl group Chemical group 0.000 description 1
- CPSYWNLKRDURMG-UHFFFAOYSA-L hydron;manganese(2+);phosphate Chemical compound [Mn+2].OP([O-])([O-])=O CPSYWNLKRDURMG-UHFFFAOYSA-L 0.000 description 1
- OUUQCZGPVNCOIJ-UHFFFAOYSA-N hydroperoxyl Chemical group O[O] OUUQCZGPVNCOIJ-UHFFFAOYSA-N 0.000 description 1
- 125000004029 hydroxymethyl group Chemical group [H]OC([H])([H])* 0.000 description 1
- 238000001727 in vivo Methods 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 229940055742 indium-111 Drugs 0.000 description 1
- APFVFJFRJDLVQX-AHCXROLUSA-N indium-111 Chemical compound [111In] APFVFJFRJDLVQX-AHCXROLUSA-N 0.000 description 1
- ZPNFWUPYTFPOJU-LPYSRVMUSA-N iniprol Chemical compound C([C@H]1C(=O)NCC(=O)NCC(=O)N[C@H]2CSSC[C@H]3C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@H](C(N[C@H](C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=4C=CC(O)=CC=4)C(=O)N[C@@H](CC=4C=CC=CC=4)C(=O)N[C@@H](CC=4C=CC(O)=CC=4)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](C)C(=O)NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CSSC[C@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@H](CO)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CC=4C=CC=CC=4)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=N)NC2=O)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CSSC[C@H](NC(=O)[C@H](CC=2C=CC=CC=2)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H]2N(CCC2)C(=O)[C@@H](N)CCCNC(N)=N)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N2[C@@H](CCC2)C(=O)N2[C@@H](CCC2)C(=O)N[C@@H](CC=2C=CC(O)=CC=2)C(=O)N[C@@H]([C@@H](C)O)C(=O)NCC(=O)N2[C@@H](CCC2)C(=O)N3)C(=O)NCC(=O)NCC(=O)N[C@@H](C)C(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@H](C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@H](C(=O)N1)C(C)C)[C@@H](C)O)[C@@H](C)CC)=O)[C@@H](C)CC)C1=CC=C(O)C=C1 ZPNFWUPYTFPOJU-LPYSRVMUSA-N 0.000 description 1
- 229910001410 inorganic ion Inorganic materials 0.000 description 1
- 229910010272 inorganic material Inorganic materials 0.000 description 1
- 239000011147 inorganic material Substances 0.000 description 1
- 229940079322 interferon Drugs 0.000 description 1
- 229940047124 interferons Drugs 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 238000006317 isomerization reaction Methods 0.000 description 1
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- USIPEGYTBGEPJN-UHFFFAOYSA-N lacto-N-tetraose Natural products O1C(CO)C(O)C(OC2C(C(O)C(O)C(CO)O2)O)C(NC(=O)C)C1OC1C(O)C(CO)OC(OC(C(O)CO)C(O)C(O)C=O)C1O USIPEGYTBGEPJN-UHFFFAOYSA-N 0.000 description 1
- 150000002605 large molecules Chemical class 0.000 description 1
- 229960004194 lidocaine Drugs 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 229920006008 lipopolysaccharide Polymers 0.000 description 1
- 230000004807 localization Effects 0.000 description 1
- 239000004325 lysozyme Substances 0.000 description 1
- 229960000274 lysozyme Drugs 0.000 description 1
- 235000010335 lysozyme Nutrition 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 239000011159 matrix material Substances 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 230000001404 mediated effect Effects 0.000 description 1
- 230000004060 metabolic process Effects 0.000 description 1
- 150000002739 metals Chemical class 0.000 description 1
- HRDXJKGNWSUIBT-UHFFFAOYSA-N methoxybenzene Chemical group [CH2]OC1=CC=CC=C1 HRDXJKGNWSUIBT-UHFFFAOYSA-N 0.000 description 1
- 238000001471 micro-filtration Methods 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- CFCUWKMKBJTWLW-BKHRDMLASA-N mithramycin Chemical compound O([C@@H]1C[C@@H](O[C@H](C)[C@H]1O)OC=1C=C2C=C3C[C@H]([C@@H](C(=O)C3=C(O)C2=C(O)C=1C)O[C@@H]1O[C@H](C)[C@@H](O)[C@H](O[C@@H]2O[C@H](C)[C@H](O)[C@H](O[C@@H]3O[C@H](C)[C@@H](O)[C@@](C)(O)C3)C2)C1)[C@H](OC)C(=O)[C@@H](O)[C@@H](C)O)[C@H]1C[C@@H](O)[C@H](O)[C@@H](C)O1 CFCUWKMKBJTWLW-BKHRDMLASA-N 0.000 description 1
- 229960004857 mitomycin Drugs 0.000 description 1
- 229960001156 mitoxantrone Drugs 0.000 description 1
- KKZJGLLVHKMTCM-UHFFFAOYSA-N mitoxantrone Chemical compound O=C1C2=C(O)C=CC(O)=C2C(=O)C2=C1C(NCCNCCO)=CC=C2NCCNCCO KKZJGLLVHKMTCM-UHFFFAOYSA-N 0.000 description 1
- HPEUEJRPDGMIMY-IFQPEPLCSA-N molybdopterin Chemical compound O([C@H]1N2)[C@H](COP(O)(O)=O)C(S)=C(S)[C@@H]1NC1=C2N=C(N)NC1=O HPEUEJRPDGMIMY-IFQPEPLCSA-N 0.000 description 1
- 125000006682 monohaloalkyl group Chemical group 0.000 description 1
- 125000001483 monosaccharide substituent group Chemical group 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 125000004572 morpholin-3-yl group Chemical group N1C(COCC1)* 0.000 description 1
- 125000004108 n-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000003136 n-heptyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000001280 n-hexyl group Chemical group C(CCCCC)* 0.000 description 1
- 125000000740 n-pentyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000004123 n-propyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 229930014626 natural product Natural products 0.000 description 1
- 239000006199 nebulizer Substances 0.000 description 1
- QJGQUHMNIGDVPM-UHFFFAOYSA-N nitrogen group Chemical group [N] QJGQUHMNIGDVPM-UHFFFAOYSA-N 0.000 description 1
- 238000000655 nuclear magnetic resonance spectrum Methods 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- 238000005580 one pot reaction Methods 0.000 description 1
- 239000011368 organic material Substances 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229960001592 paclitaxel Drugs 0.000 description 1
- 244000052769 pathogen Species 0.000 description 1
- 230000002572 peristaltic effect Effects 0.000 description 1
- 239000008177 pharmaceutical agent Substances 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 230000000144 pharmacologic effect Effects 0.000 description 1
- 238000009520 phase I clinical trial Methods 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 125000000587 piperidin-1-yl group Chemical group [H]C1([H])N(*)C([H])([H])C([H])([H])C([H])([H])C1([H])[H] 0.000 description 1
- 125000004483 piperidin-3-yl group Chemical group N1CC(CCC1)* 0.000 description 1
- 229960003171 plicamycin Drugs 0.000 description 1
- 229920000765 poly(2-oxazolines) Polymers 0.000 description 1
- 229920001583 poly(oxyethylated polyols) Polymers 0.000 description 1
- 229920002627 poly(phosphazenes) Polymers 0.000 description 1
- 229920001515 polyalkylene glycol Polymers 0.000 description 1
- 229920000768 polyamine Polymers 0.000 description 1
- 239000004417 polycarbonate Substances 0.000 description 1
- 229920000515 polycarbonate Polymers 0.000 description 1
- 229920006393 polyether sulfone Polymers 0.000 description 1
- 229920000139 polyethylene terephthalate Polymers 0.000 description 1
- 239000005020 polyethylene terephthalate Substances 0.000 description 1
- 125000006684 polyhaloalkyl group Polymers 0.000 description 1
- 229920000656 polylysine Polymers 0.000 description 1
- 229920005862 polyol Polymers 0.000 description 1
- 150000003077 polyols Chemical class 0.000 description 1
- 239000000256 polyoxyethylene sorbitan monolaurate Substances 0.000 description 1
- 235000010486 polyoxyethylene sorbitan monolaurate Nutrition 0.000 description 1
- 229920001184 polypeptide Polymers 0.000 description 1
- 229920001155 polypropylene Polymers 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 229920002223 polystyrene Polymers 0.000 description 1
- 229920000915 polyvinyl chloride Polymers 0.000 description 1
- 239000004800 polyvinyl chloride Substances 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 229960004919 procaine Drugs 0.000 description 1
- MFDFERRIHVXMIY-UHFFFAOYSA-N procaine Chemical compound CCN(CC)CCOC(=O)C1=CC=C(N)C=C1 MFDFERRIHVXMIY-UHFFFAOYSA-N 0.000 description 1
- 229960000624 procarbazine Drugs 0.000 description 1
- CPTBDICYNRMXFX-UHFFFAOYSA-N procarbazine Chemical compound CNNCC1=CC=C(C(=O)NC(C)C)C=C1 CPTBDICYNRMXFX-UHFFFAOYSA-N 0.000 description 1
- 238000011165 process development Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 229940002612 prodrug Drugs 0.000 description 1
- 239000000651 prodrug Substances 0.000 description 1
- SCUZVMOVTVSBLE-UHFFFAOYSA-N prop-2-enenitrile;styrene Chemical compound C=CC#N.C=CC1=CC=CC=C1 SCUZVMOVTVSBLE-UHFFFAOYSA-N 0.000 description 1
- 229960003712 propranolol Drugs 0.000 description 1
- 238000011403 purification operation Methods 0.000 description 1
- 239000013014 purified material Substances 0.000 description 1
- 239000012264 purified product Substances 0.000 description 1
- 125000000561 purinyl group Chemical group N1=C(N=C2N=CNC2=C1)* 0.000 description 1
- 229950010131 puromycin Drugs 0.000 description 1
- 125000003373 pyrazinyl group Chemical group 0.000 description 1
- 125000005344 pyridylmethyl group Chemical group [H]C1=C([H])C([H])=C([H])C(=N1)C([H])([H])* 0.000 description 1
- XKMLYUALXHKNFT-UHFFFAOYSA-N rGTP Natural products C1=2NC(N)=NC(=O)C=2N=CN1C1OC(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)C(O)C1O XKMLYUALXHKNFT-UHFFFAOYSA-N 0.000 description 1
- 229910052705 radium Inorganic materials 0.000 description 1
- HCWPIIXVSYCSAN-UHFFFAOYSA-N radium atom Chemical compound [Ra] HCWPIIXVSYCSAN-UHFFFAOYSA-N 0.000 description 1
- MUPFEKGTMRGPLJ-ZQSKZDJDSA-N raffinose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO[C@@H]2[C@@H]([C@@H](O)[C@@H](O)[C@@H](CO)O2)O)O1 MUPFEKGTMRGPLJ-ZQSKZDJDSA-N 0.000 description 1
- 102000005962 receptors Human genes 0.000 description 1
- 108020003175 receptors Proteins 0.000 description 1
- 239000004627 regenerated cellulose Substances 0.000 description 1
- 230000008929 regeneration Effects 0.000 description 1
- 238000011069 regeneration method Methods 0.000 description 1
- 238000012958 reprocessing Methods 0.000 description 1
- 238000002390 rotary evaporation Methods 0.000 description 1
- 239000012146 running buffer Substances 0.000 description 1
- 108010038196 saccharide-binding proteins Proteins 0.000 description 1
- 239000000523 sample Substances 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 238000010898 silica gel chromatography Methods 0.000 description 1
- 239000010703 silicon Substances 0.000 description 1
- 229910052709 silver Inorganic materials 0.000 description 1
- 239000004332 silver Substances 0.000 description 1
- 238000001542 size-exclusion chromatography Methods 0.000 description 1
- 239000003998 snake venom Substances 0.000 description 1
- 239000001632 sodium acetate Substances 0.000 description 1
- 235000017281 sodium acetate Nutrition 0.000 description 1
- AJPJDKMHJJGVTQ-UHFFFAOYSA-M sodium dihydrogen phosphate Chemical compound [Na+].OP(O)([O-])=O AJPJDKMHJJGVTQ-UHFFFAOYSA-M 0.000 description 1
- 239000012064 sodium phosphate buffer Substances 0.000 description 1
- JBJWASZNUJCEKT-UHFFFAOYSA-M sodium;hydroxide;hydrate Chemical compound O.[OH-].[Na+] JBJWASZNUJCEKT-UHFFFAOYSA-M 0.000 description 1
- UQZIYBXSHAGNOE-XNSRJBNMSA-N stachyose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO[C@@H]2[C@@H]([C@@H](O)[C@@H](O)[C@@H](CO[C@@H]3[C@@H]([C@@H](O)[C@@H](O)[C@@H](CO)O3)O)O2)O)O1 UQZIYBXSHAGNOE-XNSRJBNMSA-N 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 238000003756 stirring Methods 0.000 description 1
- 125000003107 substituted aryl group Chemical group 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 239000005720 sucrose Substances 0.000 description 1
- 239000004094 surface-active agent Substances 0.000 description 1
- 229940037128 systemic glucocorticoids Drugs 0.000 description 1
- RCINICONZNJXQF-MZXODVADSA-N taxol Chemical compound O([C@@H]1[C@@]2(C[C@@H](C(C)=C(C2(C)C)[C@H](C([C@]2(C)[C@@H](O)C[C@H]3OC[C@]3([C@H]21)OC(C)=O)=O)OC(=O)C)OC(=O)[C@H](O)[C@@H](NC(=O)C=1C=CC=CC=1)C=1C=CC=CC=1)O)C(=O)C1=CC=CC=C1 RCINICONZNJXQF-MZXODVADSA-N 0.000 description 1
- NRUKOCRGYNPUPR-QBPJDGROSA-N teniposide Chemical compound COC1=C(O)C(OC)=CC([C@@H]2C3=CC=4OCOC=4C=C3[C@@H](O[C@H]3[C@@H]([C@@H](O)[C@@H]4O[C@@H](OC[C@H]4O3)C=3SC=CC=3)O)[C@@H]3[C@@H]2C(OC3)=O)=C1 NRUKOCRGYNPUPR-QBPJDGROSA-N 0.000 description 1
- 229960001278 teniposide Drugs 0.000 description 1
- 229920001897 terpolymer Polymers 0.000 description 1
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 description 1
- 229960002372 tetracaine Drugs 0.000 description 1
- GKCBAIGFKIBETG-UHFFFAOYSA-N tetracaine Chemical compound CCCCNC1=CC=C(C(=O)OCCN(C)C)C=C1 GKCBAIGFKIBETG-UHFFFAOYSA-N 0.000 description 1
- 125000004192 tetrahydrofuran-2-yl group Chemical group [H]C1([H])OC([H])(*)C([H])([H])C1([H])[H] 0.000 description 1
- 150000004044 tetrasaccharides Chemical class 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- 150000003573 thiols Chemical group 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 239000012581 transferrin Substances 0.000 description 1
- 125000002264 triphosphate group Chemical group [H]OP(=O)(O[H])OP(=O)(O[H])OP(=O)(O[H])O* 0.000 description 1
- 231100000397 ulcer Toxicity 0.000 description 1
- DNYWZCXLKNTFFI-UHFFFAOYSA-N uranium Chemical compound [U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U][U] DNYWZCXLKNTFFI-UHFFFAOYSA-N 0.000 description 1
- PGAVKCOVUIYSFO-UHFFFAOYSA-N uridine-triphosphate Natural products OC1C(O)C(COP(O)(=O)OP(O)(=O)OP(O)(O)=O)OC1N1C(=O)NC(=O)C=C1 PGAVKCOVUIYSFO-UHFFFAOYSA-N 0.000 description 1
- 229940118696 vibrio cholerae Drugs 0.000 description 1
- 229960003048 vinblastine Drugs 0.000 description 1
- JXLYSJRDGCGARV-XQKSVPLYSA-N vincaleukoblastine Chemical compound C([C@@H](C[C@]1(C(=O)OC)C=2C(=CC3=C([C@]45[C@H]([C@@]([C@H](OC(C)=O)[C@]6(CC)C=CCN([C@H]56)CC4)(O)C(=O)OC)N3C)C=2)OC)C[C@@](C2)(O)CC)N2CCC2=C1NC1=CC=CC=C21 JXLYSJRDGCGARV-XQKSVPLYSA-N 0.000 description 1
- OGWKCGZFUXNPDA-XQKSVPLYSA-N vincristine Chemical compound C([N@]1C[C@@H](C[C@]2(C(=O)OC)C=3C(=CC4=C([C@]56[C@H]([C@@]([C@H](OC(C)=O)[C@]7(CC)C=CCN([C@H]67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)C[C@@](C1)(O)CC)CC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-XQKSVPLYSA-N 0.000 description 1
- 229960004528 vincristine Drugs 0.000 description 1
- OGWKCGZFUXNPDA-UHFFFAOYSA-N vincristine Natural products C1C(CC)(O)CC(CC2(C(=O)OC)C=3C(=CC4=C(C56C(C(C(OC(C)=O)C7(CC)C=CCN(C67)CC5)(O)C(=O)OC)N4C=O)C=3)OC)CN1CCC1=C2NC2=CC=CC=C12 OGWKCGZFUXNPDA-UHFFFAOYSA-N 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- 229920002554 vinyl polymer Polymers 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12P—FERMENTATION OR ENZYME-USING PROCESSES TO SYNTHESISE A DESIRED CHEMICAL COMPOUND OR COMPOSITION OR TO SEPARATE OPTICAL ISOMERS FROM A RACEMIC MIXTURE
- C12P19/00—Preparation of compounds containing saccharide radicals
- C12P19/26—Preparation of nitrogen-containing carbohydrates
- C12P19/28—N-glycosides
- C12P19/30—Nucleotides
- C12P19/305—Pyrimidine nucleotides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07H—SUGARS; DERIVATIVES THEREOF; NUCLEOSIDES; NUCLEOTIDES; NUCLEIC ACIDS
- C07H19/00—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof
- C07H19/02—Compounds containing a hetero ring sharing one ring hetero atom with a saccharide radical; Nucleosides; Mononucleotides; Anhydro-derivatives thereof sharing nitrogen
- C07H19/04—Heterocyclic radicals containing only nitrogen atoms as ring hetero atom
- C07H19/06—Pyrimidine radicals
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/582—Recycling of unreacted starting or intermediate materials
Definitions
- sialyl lactose is thought to inhibit or disrupt the degree of occupancy of the Fc carbohydrate binding site on IgG, and thus prevent the formation of immune complexes (see, U.S. Pat. No. 5,164,374).
- sialyl- ⁇ (2,3)galactosides, sialyl lactose and sialyl lactosamine have been proposed for the treatment of ulcers, and Phase I clinical trials have begun for the use of the former compound in this capacity. See,Parken et al., FEMS Immunology and Medical Microbiology 7:29 (1993) and BioWorld Today, p. 5, Apr. 4, 1995.
- compounds comprising the sialyl Lewis ligands, sialyl Lewis x and sialyl Lewis a are present in leukocyte and non-leukocyte cell lines that bind to receptors such as the ELAM-1 and GMP 140 receptors.
- receptors such as the ELAM-1 and GMP 140 receptors.
- Polley et al., Proc. Natl. Acad. Sci., USA, 88:6224 (1991) and Phillips et al., Science, 250:1130 (1990) see, also, U.S. Ser. No. 08/063,181.
- glycosyltransferases Because of interest in making desired carbohydrate structures, glycosyltransferases and their role in enzyme-catalyzed synthesis of carbohydrates are presently being extensively studied.
- the use of glycosyltransferases for enzymatic synthesis of carbohydrate offers advantages over chemical methods due to the virtually complete stereoselectivity and linkage specificity offered by the enzymes (Ito et al., Pure Appl. Chem., 65:753 (1993) U.S. Pat. Nos. 5,352,670, and 5,374,541). Consequently, glycosyltransferases are increasingly used as enzymatic catalysts in synthesis of a number of carbohydrates used for therapeutic and other purposes.
- Carbohydrate compounds produced by enzymatic synthesis or by other methods are often obtained in the form of complex mixtures that include not only the desired compound but also contaminants such as unreacted sugars, salts, pyruvate, phosphate, PEP, nucleosides, nucleotides, and proteins, among others. The presence of these contaminants is undesirable for many applications for which the carbohydrate compounds are useful.
- Previously used methods for purifying oligosaccharides, such as chromatography, i.e., ion exchange and size exclusion chromatography have several disadvantages. For example, chromatographic purification methods are not amenable to large-scale purifications, thus precluding their use for commercial production of saccharides. Moreover, chromatographic purification methods are expensive. Therefore, a need exists for purification methods that are faster, more efficient, and less expensive than previously used methods. The present invention fulfills this and other needs.
- the present invention provides methods of purifying a carbohydrate compound from a feed solution containing a contaminant.
- the methods involve contacting the feed solution with a nanofiltration or reverse osmosis membrane under conditions such that the membrane retains the desired carbohydrate compound while a majority of the contaminant passes through the membrane.
- the invention provides methods of purifying a carbohydrate compound from a feed solution comprising a reaction mixture used to synthesize the carbohydrate compound.
- the synthesis can be enzymatic or chemical, or a combination thereof.
- the methods involve removing any proteins present in the feed solution by contacting the feed solution with an ultrafiltration membrane so that proteins are retained the membrane while the carbohydrate compound passes through the membrane as a permeate.
- the permeate from the ultrafiltration step is then contacted with a nanofiltration or reverse osmosis membrane under conditions such that the nanofiltration or reverse osmosis membrane retains the carbohydrate compound while a majority of an undesired contaminant passes through the membrane.
- Another embodiment of the invention provides methods for purifying nucleotides, nucleosides, and nucleotide sugars by contacting a feed solution containing the nucleotide or related compound with a nanofiltration or reverse osmosis membrane under conditions such that the membrane retains the nucleotide or related compound while a majority of the contaminant passes through the membrane.
- the present invention also provides methods for removing one or more contaminants from a solution that contains a carbohydrate of interest.
- the methods involve contacting the solution with a first side of a semipermeable membrane having rejection coefficients so as to retain the carbohydrate while allowing the contaminant to pass through the membrane.
- the membrane is selected from the group consisting of an ultrafiltration membrane, a nanofiltration membrane, and a reverse osmosis membrane, depending on the size and charge of the carbohydrate of interest relative to those of the contaminants.
- the membrane separates a feed solution containing a carbohydrate into a retentate portion and a permeate portion.
- the rejection coefficient of the membrane is greater for the carbohydrate than for the contaminant, the retentate portion will have a lower concentration of the contaminant relative to the contaminant concentration in the feed solution, and generally also a higher ratio of the carbohydrate to the undesired contaminant.
- a membrane having a rejection coefficient for the carbohydrate that is lesser than that for the contaminant will effect a separation wherein the concentration of the contaminant is lower in the permeate than in the feed solution, and the permeate will have a higher ratio of carbohydrate to contaminant than the feed solution.
- the fraction containing the carbohydrate can be recycled through the membrane system for further purification.
- contaminants that can be removed from solutions containing the compound of interest using the methods of the invention include, but are not limited to, unreacted sugars, inorganic ions, pyruvate, phosphate, phosphoenolpyruvate, and proteins.
- FIG. 1 is a diagram of an exemplary purification of a nucleotide sugar.
- FIG. 2 is a chromatogram recorded for an exemplary purification of CMP-SA-PEG-30 kDa using Q Sepharose chromatography as described in Example 6.
- CMP-SA-PEG-30 kDa was collected in two fractions. Fraction 1 contained pure product and Fraction 2 contained residual CMP-SA-Gly reagent and was reprocessed.
- FIG. 3 is a chromatogram recorded for an exemplary separation of CMP-SA-PEG 30 kDa from CMP-SA-Glycine using Q-Sepharose chromatography, showing a typical baseline separation.
- FIG. 4 is a diagram outlining an exemplary process for the preparation of an exemplary GSC (CMP-5′-Glycyl-Sialic Acid).
- FIG. 5 is a diagram outlining an exemplary process for the preparation of an exemplary PSC (CMP-SA-PEG).
- FIG. 6 is a diagram outlining an exemplary process for the purification of a glycosyltransferase.
- FIG. 7 is a table of exemplary sialyltransferases.
- sialic acid refers to any member of a family of nine-carbon carboxylated sugars.
- the most common member of the sialic acid family is N-acetyl-neuraminic acid (2-keto-5-acetamido-3,5-dideoxy-D-glycero-D-galactononulopyranos-1-onic acid (often abbreviated as Neu5Ac, NeuAc, or NANA).
- a second member of the family is N-glycolyl-neuraminic acid (Neu5Gc or NeuGc), in which the N-acetyl group of NeuAc is hydroxylated.
- a third sialic acid family member is 2-keto-3-deoxy-nonulosonic acid (KDN) (Nadano et al. (1986) J. Biol. Chem. 261: 11550-11557; Kanamori et al., J. Biol. Chem. 265: 21811-21819 (1990)). Also included are 9-substituted sialic acids such as a 9-O—C 1 -C 6 acyl-Neu5Ac like 9-O-lactyl-Neu5Ac or 9-O-acetyl-Neu5Ac, 9-deoxy-9-fluoro-Neu5Ac and 9-azido-9-deoxy-Neu5Ac.
- KDN 2-keto-3-deoxy-nonulosonic acid
- 9-substituted sialic acids such as a 9-O—C 1 -C 6 acyl-Neu5Ac like 9-O-lactyl-Neu5Ac or 9-O-
- sialic acid family see, e.g., Varki, Glycobiology 2: 25-40 (1992); Sialic Acids: Chemistry, Metabolism and Function , R. Schauer, Ed. (Springer-Verlag, New York (1992)).
- the synthesis and use of sialic acid compounds in a sialylation procedure is disclosed in international application WO 92/16640, published Oct. 1, 1992.
- modified sugar refers to a naturally- or non-naturally-occurring carbohydrate.
- the modified sugar is preferably selected from a number of enzyme substrates including, but not limited to sugar nucleotides (mono-, di-, and tri-phosphates), activated sugars (e.g., glycosyl halides, glycosyl mesylates) and sugars that are neither activated nor nucleotides.
- the “modified sugar” is covalently functionalized with a “modifying group.”
- Useful modifying groups include, but are not limited to, water-soluble polymers, targeting moieties therapeutic moieties, diagnostic moieties, radioactive moieties, cytotoxic moieties, biomolecules and the like.
- the modifying group is preferably not a naturally occurring, or an unmodified carbohydrate.
- the locus of functionalization with the modifying group is preferably selected such that it does not prevent the “modified sugar” from being added enzymatically to a peptide.
- water-soluble refers to moieties that have some detectable degree of solubility in water. Methods to detect and/or quantify water solubility are well known in the art.
- Exemplary water-soluble polymers include peptides, saccharides, poly(ethers), poly(amines), poly(carboxylic acids) and the like. Peptides can have mixed sequences of be composed of a single amino acid, e.g., poly(lysine).
- An exemplary polysaccharide is poly(sialic acid).
- An exemplary poly(ether) is poly(ethylene glycol), e.g., m-PEG.
- Poly(ethylene imine) is an exemplary polyamine
- poly(acrylic) acid is a representative poly(carboxylic acid).
- the polymer backbone of the water-soluble polymer can be poly(ethylene glycol) (i.e. PEG).
- PEG poly(ethylene glycol)
- other related polymers are also suitable for use in the practice of this invention and that the use of the term PEG or poly(ethylene glycol) is intended to be inclusive and not exclusive in this respect.
- PEG includes poly(ethylene glycol) in any of its forms, including alkoxy PEG, difunctional PEG, multiarmed PEG, forked PEG, branched PEG, pendent PEG (i.e. PEG or related polymers having one or more functional groups pendent to the polymer backbone), or PEG with degradable linkages therein.
- the polymer backbone can be linear or branched.
- Branched polymer backbones are generally known in the art.
- a branched polymer has a central branch core moiety and a plurality of linear polymer chains linked to the central branch core.
- PEG is commonly used in branched forms that can be prepared by addition of ethylene oxide to various polyols, such as glycerol, pentaerythritol and sorbitol.
- the central branch moiety can also be derived from several amino acids, such as lysine.
- the branched poly(ethylene glycol) can be represented in general form as R(-PEG-OH) m in which R represents the core moiety, such as glycerol or pentaerythritol, and m represents the number of arms.
- R represents the core moiety, such as glycerol or pentaerythritol
- m represents the number of arms.
- Multi-armed PEG molecules such as those described in U.S. Pat. No. 5,932,462, which is incorporated by reference herein in its entirety, can also be used as the polymer backbone.
- polymers are also suitable for the invention.
- suitable polymers include, but are not limited to, other poly(alkylene glycols), such as poly(propylene glycol) (“PPG”), copolymers of ethylene glycol and propylene glycol and the like, poly(oxyethylated polyol), poly(olefinic alcohol), poly(vinylpyrrolidone), poly(hydroxypropylmethacrylamide), poly( ⁇ -hydroxy acid), poly(vinyl alcohol), polyphosphazene, polyoxazoline, poly(N-acryloylmorpholine), such as described in U.S. Pat.
- PPG poly(propylene glycol)
- PPG poly(propylene glycol)
- copolymers of ethylene glycol and propylene glycol and the like poly(oxyethylated polyol), poly(olefinic alcohol),
- targeting moiety refers to species that will selectively localize in a particular tissue or region of the body. The localization is mediated by specific recognition of molecular determinants, molecular size of the targeting agent or conjugate, ionic interactions, hydrophobic interactions and the like. Other mechanisms of targeting an agent to a particular tissue or region are known to those of skill in the art.
- exemplary targeting moieties include antibodies, antibody fragments, transferrin, HS-glycoprotein, coagulation factors, serum proteins, ⁇ -glycoprotein, G-CSF, GM-CSF, M-CSF, EPO and the like.
- therapeutic moiety means any agent useful for therapy including, but not limited to, antibiotics, anti-inflammatory agents, anti-tumor drugs, cytotoxins, and radioactive agents.
- therapeutic moiety includes prodrugs of bioactive agents, constructs in which more than one therapeutic moiety is bound to a carrier, e.g, multivalent agents.
- Therapeutic moiety also includes proteins and constructs that include proteins.
- Exemplary proteins include, but are not limited to, Erythropoietin (EPO), Granulocyte Colony Stimulating Factor (GCSF), Granulocyte Macrophage Colony Stimulating Factor (GMCSF), Interferon (e.g., Interferon- ⁇ , - ⁇ , - ⁇ ), Interleukin (e.g., Interleukin II), serum proteins (e.g., Factors VII, VIa, VIII, IX, and X), Human Chorionic Gonadotropin (HCG), Follicle Stimulating Hormone (FSH) and Lutenizing Hormone (LH) and antibody fusion proteins (e.g. Tumor Necrosis Factor Receptor ((TNFR)/Fc domain fusion protein)).
- EPO Erythropoietin
- GCSF Granulocyte Colony Stimulating Factor
- GMCSF Granulocyte Macrophage Colony Stimulating Factor
- Interferon
- anti-tumor drug means any agent useful to combat cancer including, but not limited to, cytotoxins and agents such as antimetabolites, alkylating agents, anthracyclines, antibiotics, antimitotic agents, procarbazine, hydroxyurea, asparaginase, corticosteroids, interferons and radioactive agents.
- conjugates of peptides with anti-tumor activity e.g. TNF- ⁇ . Conjugates include, but are not limited to those formed between a therapeutic protein and a glycoprotein of the invention. A representative conjugate is that formed between PSGL-1 and TNF- ⁇ .
- a cytotoxin or cytotoxic agent means any agent that is detrimental to cells. Examples include taxol, cytochalasin B, gramicidin D, ethidium bromide, emetine, mitomycin, etoposide, tenoposide, vincristine, vinblastine, colchicin, doxorubicin, daunorubicin, dihydroxy anthracinedione, mitoxantrone, mithramycin, actinomycin D, 1-dehydrotestosterone, glucocorticoids, procaine, tetracaine, lidocaine, propranolol, and puromycin and analogs or homologs thereof.
- Other toxins include, for example, ricin, CC-1065 and analogues, the duocarmycins. Still other toxins include diptheria toxin, and snake venom (e.g., cobra venom).
- a radioactive agent includes any radioisotope that is effective in diagnosing or destroying a tumor. Examples include, but are not limited to, indium-111, cobalt-60. Additionally, naturally occurring radioactive elements such as uranium, radium, and thorium, which typically represent mixtures of radioisotopes, are suitable examples of a radioactive agent. The metal ions are typically chelated with an organic chelating moiety.
- a compound is “substantially purified” from an undesired component in a solution if the concentration of the undesired component after purification is no greater than about 40% of the concentration of the component prior to purification.
- the post-purification concentration of the undesired component will be less than about 20% by weight, and more preferably less than about 10%, and still more preferably less than about 5% of the pre-purification concentration.
- pharmaceutically pure refers to a compound that is sufficiently purified from undesired contaminants that the compound is suitable for administration as a pharmaceutical agent.
- the compound is purified such that the undesired contaminant is present after purification in an amount that is about 5% by weight or less of the pre-purification concentration of the contaminant in the feed solution.
- the post-purification concentration of the contaminant is about 1% or less of the pre-purification contaminant concentration, and most preferably about 0.5% or less of the pre-purification concentration of contaminant.
- a “feed solution” refers to any solution that contains a compound to be purified.
- a reaction mixture used to synthesize an oligosaccharide can be used as a feed solution from which the desired reaction product is purified using the methods of the invention.
- substituent groups are specified by their conventional chemical formulae, written from left to right, they equally encompass the chemically identical substituents, which would result from writing the structure from right to left, e.g., —CH 2 O— is intended to also recite —OCH 2 —.
- alkyl by itself or as part of another substituent means, unless otherwise stated, a straight or branched chain, or cyclic hydrocarbon radical, or combination thereof, which may be fully saturated, mono- or polyunsaturated and can include di- and multivalent radicals, having the number of carbon atoms designated (i.e. C 1 -C 10 means one to ten carbons).
- saturated hydrocarbon radicals include, but are not limited to, groups such as methyl, ethyl, n-propyl, isopropyl, n-butyl, t-butyl, isobutyl, sec-butyl, cyclohexyl, (cyclohexyl)methyl, cyclopropylmethyl, homologs and isomers of, for example, n-pentyl, n-hexyl, n-heptyl, n-octyl, and the like.
- An unsaturated alkyl group is one having one or more double bonds or triple bonds.
- alkyl groups examples include, but are not limited to, vinyl, 2-propenyl, crotyl, 2-isopentenyl, 2-(butadienyl), 2,4-pentadienyl, 3-(1,4-pentadienyl), ethynyl, 1- and 3-propynyl, 3-butynyl, and the higher homologs and isomers.
- alkyl unless otherwise noted, is also meant to include those derivatives of alkyl defined in more detail below, such as “heteroalkyl.” Alkyl groups that are limited to hydrocarbon groups are termed “homoalkyl”.
- alkylene by itself or as part of another substituent means a divalent radical derived from an alkane, as exemplified, but not limited, by —CH 2 CH 2 CH 2 CH 2 —, and further includes those groups described below as “heteroalkylene.”
- an alkyl (or alkylene) group will have from 1 to 24 carbon atoms, with those groups having 10 or fewer carbon atoms being preferred in the present invention.
- a “lower alkyl” or “lower alkylene” is a shorter chain alkyl or alkylene group, generally having eight or fewer carbon atoms.
- alkoxy alkylamino and “alkylthio” (or thioalkoxy) are used in their conventional sense, and refer to those alkyl groups attached to the remainder of the molecule via an oxygen atom, an amino group, or a sulfur atom, respectively.
- heteroalkyl by itself or in combination with another term, means, unless otherwise stated, a stable straight or branched chain, or cyclic hydrocarbon radical, or combinations thereof, consisting of the stated number of carbon atoms and at least one heteroatom selected from the group consisting of O, N, Si and S, and wherein the nitrogen and sulfur atoms may optionally be oxidized and the nitrogen heteroatom may optionally be quaternized.
- the heteroatom(s) O, N and S and Si may be placed at any interior position of the heteroalkyl group or at the position at which the alkyl group is attached to the remainder of the molecule.
- Examples include, but are not limited to, —CH 2 —CH 2 —O—CH 3 , —CH 2 —CH 2 —NH—CH 3 , —CH 2 —CH 2 —N(CH 3 )—CH 3 , —CH 2 —S—CH 2 —CH 3 , —CH 2 —CH 2 , —S(O)—CH 3 , —CH 2 —CH 2 —S(O) 2 —CH 3 , —CH ⁇ CH—O—CH 3 , —Si(CH 3 ) 3 , —CH 2 —CH ⁇ N—OCH 3 , and —CH ⁇ CH—N(CH 3 )—CH 3 .
- heteroalkylene by itself or as part of another substituent means a divalent radical derived from heteroalkyl, as exemplified, but not limited by, —CH 2 —CH 2 —S—CH 2 —CH 2 — and —CH 2 —S—CH 2 —CH 2 —NH—CH 2 —.
- heteroatoms can also occupy either or both of the chain termini (e.g., alkyleneoxy, alkylenedioxy, alkyleneamino, alkylenediamino, and the like). Still further, for alkylene and heteroalkylene linking groups, no orientation of the linking group is implied by the direction in which the formula of the linking group is written. For example, the formula —C(O) 2 R′— represents both —C(O) 2 R′— and —R′C(O) 2 —.
- cycloalkyl and “heterocycloalkyl”, by themselves or in combination with other terms, represent, unless otherwise stated, cyclic versions of “alkyl” and “heteroalkyl”, respectively. Additionally, for heterocycloalkyl, a heteroatom can occupy the position at which the heterocycle is attached to the remainder of the molecule. Examples of cycloalkyl include, but are not limited to, cyclopentyl, cyclohexyl, 1-cyclohexenyl, 3-cyclohexenyl, cycloheptyl, and the like.
- heterocycloalkyl examples include, but are not limited to, 1-(1,2,5,6-tetrahydropyridyl), 1-piperidinyl, 2-piperidinyl, 3-piperidinyl, 4-morpholinyl, 3-morpholinyl, tetrahydrofuran-2-yl, tetrahydrofuran-3-yl, tetrahydrothien-2-yl, tetrahydrothien-3-yl, 1-piperazinyl, 2-piperazinyl, and the like.
- halo or “halogen,” by themselves or as part of another substituent, mean, unless otherwise stated, a fluorine, chlorine, bromine, or iodine atom. Additionally, terms such as “haloalkyl,” are meant to include monohaloalkyl and polyhaloalkyl.
- halo(C 1 -C 4 )alkyl is mean to include, but not be limited to, trifluoromethyl, 2,2,2-trifluoroethyl, 4-chlorobutyl, 3-bromopropyl, and the like.
- aryl means, unless otherwise stated, a polyunsaturated, aromatic, substituent that can be a single ring or multiple rings (preferably from 1 to 3 rings), which are fused together or linked covalently.
- heteroaryl refers to aryl groups (or rings) that contain from one to four heteroatoms selected from N, O, and S, wherein the nitrogen and sulfur atoms are optionally oxidized, and the nitrogen atom(s) are optionally quaternized.
- a heteroaryl group can be attached to the remainder of the molecule through a heteroatom.
- Non-limiting examples of aryl and heteroaryl groups include phenyl, 1-naphthyl, 2-naphthyl, 4-biphenyl, 1-pyrrolyl, 2-pyrrolyl, 3-pyrrolyl, 3-pyrazolyl, 2-imidazolyl, 4-imidazolyl, pyrazinyl, 2-oxazolyl, 4-oxazolyl, 2-phenyl-4-oxazolyl, 5-oxazolyl, 3-isoxazolyl, 4-isoxazolyl, 5-isoxazolyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, 2-furyl, 3-furyl, 2-thienyl, 3-thienyl, 2-pyridyl, 3-pyridyl, 4-pyridyl, 2-pyrimidyl, 4-pyrimidyl, 5-benzothiazolyl, purinyl, 2-benzimidazolyl, 5-indolyl, 1-isoquinoly
- aryl when used in combination with other terms (e.g., aryloxy, arylthioxy, arylalkyl) includes both aryl and heteroaryl rings as defined above.
- arylalkyl is meant to include those radicals in which an aryl group is attached to an alkyl group (e.g., benzyl, phenethyl, pyridylmethyl and the like) including those alkyl groups in which a carbon atom (e.g., a methylene group) has been replaced by, for example, an oxygen atom (e.g., phenoxymethyl, 2-pyridyloxymethyl, 3-(1-naphthyloxy)propyl, and the like).
- alkyl group e.g., benzyl, phenethyl, pyridylmethyl and the like
- an oxygen atom e.g., phenoxymethyl, 2-pyridyloxymethyl, 3-(1-naph
- alkyl e.g., “alkyl,” “heteroalkyl,” “aryl” and “heteroaryl” is meant to include both substituted and unsubstituted forms of the indicated radical.
- Preferred substituents for each type of radical are provided below.
- alkyl and heteroalkyl radicals are generically referred to as “alkyl group substituents,” and they can be one or more of a variety of groups selected from, but not limited to: —OR′, ⁇ O, ⁇ NR′, ⁇ N—OR′, —NR′R′′, —SR′, -halogen, —SiR′R′′R′′′, —OC(O)R′, —C(O)R′, —CO 2 R′, —CONR′R′′, —OC(O)NR′R′′, —NR′′C(O)R′, —NR′—C(O)NR′′R′′′, —NR′′C(O) 2 R′, ——OR′, ⁇ O, ⁇ NR′, ⁇ N—OR′, —NR′R′′, —SR′, -halogen, —SiR′R′′R′′′, —OC(O)R′, —C(O)R′, —CO 2 R′
- R′, R′′, R′′′ and R′′′′ each preferably independently refer to hydrogen, substituted or unsubstituted heteroalkyl, substituted or unsubstituted aryl, e.g., aryl substituted with 1-3 halogens, substituted or unsubstituted alkyl, alkoxy or thioalkoxy groups, or arylalkyl groups.
- each of the R groups is independently selected as are each R′, R′′, R′′′ and R′′′′ groups when more than one of these groups is present.
- R′ and R′′ are attached to the same nitrogen atom, they can be combined with the nitrogen atom to form a 5-, 6-, or 7-membered ring.
- —NR′R′′ is meant to include, but not be limited to, 1-pyrrolidinyl and 4-morpholinyl.
- alkyl is meant to include groups including carbon atoms bound to groups other than hydrogen groups, such as haloalkyl (e.g., —CF 3 and —CH 2 CF 3 ) and acyl (e.g., —C(O)CH 3 , —C(O)CF 3 , —C(O)CH 2 OCH 3 , and the like).
- haloalkyl e.g., —CF 3 and —CH 2 CF 3
- acyl e.g., —C(O)CH 3 , —C(O)CF 3 , —C(O)CH 2 OCH 3 , and the like.
- substituents for the aryl and heteroaryl groups are generically referred to as “aryl group substituents.”
- the substituents are selected from, for example: halogen, —OR′, ⁇ O, ⁇ NR′, ⁇ N—OR′, —NR′R′′, —SR′, -halogen, —SiR′R′′R′′′, —OC(O)R′, —C(O)R′, —CO 2 R′, —CONR′R′′, —OC(O)NR′R′′, —NR′′C(O)R′, —NR′—C(O)NR′′R′′′, —NR′′C(O) 2 R′, —NR—C(NR′R′′R′′′) ⁇ NR′′′′, —NR—C(NR′R′′) ⁇ NR′′′, —S(O)R′, —S(O) 2 R′, —S(O) 2 NR′R′′
- each of the R groups is independently selected as are each R′, R′′, R′′′ and R′′′′ groups when more than one of these groups is present.
- the symbol X represents “R” as described above.
- Two of the substituents on adjacent atoms of the aryl or heteroaryl ring may optionally be replaced with a substituent of the formula -T-C(O)—(CRR′) q —U—, wherein T and U are independently —NR—, —O—, —CRR′— or a single bond, and q is an integer of from 0 to 3.
- two of the substituents on adjacent atoms of the aryl or heteroaryl ring may optionally be replaced with a substituent of the formula -A-(CH 2 ) r —B—, wherein A and B are independently —CRR′—, —O—, —NR—, —S—, —S(O)—, —S(O) 2 —, —S(O) 2 NR′— or a single bond, and r is an integer of from 1 to 4.
- One of the single bonds of the new ring so formed may optionally be replaced with a double bond.
- two of the substituents on adjacent atoms of the aryl or heteroaryl ring may optionally be replaced with a substituent of the formula —(CRR′) s —X—(CR′′R′′′) d —, where s and d are independently integers of from 0 to 3, and X is —O—, —NR′—, —S—, —S(O)—, —S(O) 2 —, or —S(O) 2 NR′—.
- the substituents R, R′, R′′ and R′′′ are preferably independently selected from hydrogen or substituted or unsubstituted (C 1 -C 6 )alkyl.
- heteroatom is meant to include oxygen (O), nitrogen (N), sulfur (S) and silicon (Si).
- the present invention provides methods for rapidly and efficiently purifying specific carbohydrate and oligosaccharide structures to a high degree of purity using semipermeable membranes such as reverse osmosis and/or nanofiltration membranes.
- the methods are particularly useful for separating desired oligosaccharide compounds from reactants and other contaminants that remain in a reaction mixture after synthesis or breakdown of the oligosaccharides.
- the invention provides methods for separating oligosaccharides from enzymes and/or other components of reaction mixtures used for enzymatic synthesis or enzymatic degradation of oligosaccharides, nucleotide sugars, glycolipids, liposaccharides, nucleotides, nucleosides, and other saccharide-containing compounds.
- the saccharides e.g., sialyl lactose, SLe x , and many others
- the purification methods of the invention are more efficient, rapid, and amenable to large-scale purifications than previously known carbohydrate purification methods.
- the invention provides single-step methods for purifying saccharide-containing compounds.
- a membrane is selected that is appropriate for separating the desired carbohydrate from the undesired components (contaminants) of the solution from which the carbohydrate is to be purified.
- the goal in selecting a membrane is to optimize for a particular application the molecular weight cutoff (MWCO), membrane composition, permeability, and rejection characteristics, that is, the membrane's total capacity to retain specific molecules while allowing other species, e.g., salts and other, generally smaller or opposite charged molecules, to pass through.
- MWCO molecular weight cutoff
- the percent retention of a component is also called the retention characteristic or the membrane rejection coefficient.
- a membrane is chosen that has a high rejection ratio for the saccharide of interest relative to the rejection ratio for compounds from which separation is desired. If a membrane has a high rejection ratio for a first compound relative to a second compound, the concentration of the first compound in the permeate solution which passes through the membrane is decreased relative to that of the second compound. Conversely, the concentration of the first compound increases relative to the concentration of the second compound in the retentate. If a membrane does not reject a compound, the concentration of the compound in both the permeate and the reject portions will remain essentially the same as in the feed solution.
- a membrane it is also possible for a membrane to have a negative rejection rate for a compound if the compound's concentration in the permeate becomes greater than the compound's concentration in the feed solution.
- a general review of membrane technology is found in “Membranes and Membrane Separation Processes,” in Ullmann's Encyclopedia of Industrial Chemistry (VCH, 1990); see also, Noble and Stem, Membrane Separations Technology: Principles and Applications (Elsevier, 1995).
- a membrane having a molecular weight cut-off (MWCO, which is often related to membrane pore size) that is expected to retain the desired compounds while allowing an undesired compound present in the feed stream to pass through the membrane.
- the desired MWCO is generally less than the molecular weight of the compound being purified, and is typically greater than the molecular weight of the undesired contaminant that is to be removed from the solution containing the compound being purified.
- MWCO molecular weight cut-off
- UF ultrafiltration
- NF nanofiltration
- RO reverse osmosis
- RO membranes typically have a nominal MWCO of less than about 200 Da and reject most ions
- NF membranes generally have a nominal MWCO of between about 150 Da and about 5 kDa
- UF membranes generally have a nominal MWCO of between about 1 kDa and about 300 kDa (these MWCO ranges assume a saccharide-like molecule).
- a second parameter that is considered in choosing an appropriate membrane for a particular separation is the polymer type of the membrane.
- Exemplary membranes of use in the invention are made of conventional membrane material whether inorganic, organic, or mixed inorganic and organic. Typical inorganic materials include glasses, ceramics, cermets, metals and the like. Ceramic membranes, which are preferred for the UF zone, may be made, for example, as described in U.S. Pat. No. 4,692,354 to Asaeda et al, U.S. Pat. No. 4,562,021 to Alary et al., and others.
- the organic materials which are preferred for the NF and RO applications are typically polymers, whether isotropic, or anisotropic with a thin layer or “skin” on either the bore side or the shell side of the fibers.
- Preferred materials for fibers are polyamides, polybenzamides, polysulfones (including sulfonated polysulfone and sulfonated polyether sulfone, among others), polystyrenes, including styrene-containing copolymers such as acrylo-nitrile-styrene, butadiene-styrene and styrene-vinylbenzylhalide copolymers, polycarbonates, cellulosic polymers including cellulose acetate, polypropylene, poly(vinyl chloride), poly(ethylene terephthalate), polyvinyl alcohol, fluorocarbons, and the like, such as those disclosed in U.S. Pat. Nos. 4,230,463, 4,806,244,
- a membrane surface charge is selected that has a surface charge that is appropriate for the ionic charge of the carbohydrate and that of the contaminants. While MWCO for a particular membrane is generally invariable, changing the pH of the feed solution can affect separation properties of a membrane by altering the membrane surface charge. For example, a membrane that has a net negative surface charge at neutral pH can be adjusted to have a net neutral charge simply by lowering the pH of the solution. An additional effect of adjusting solution pH is to modulate the ionic charge on the contaminants and on the carbohydrate of interest.
- a suitable membrane polymer type and pH By choosing a suitable membrane polymer type and pH, one can obtain a system in which both the contaminant and the membrane are neutral, facilitating pass-through of the contaminant.
- a contaminant is negatively charged at neutral pH, it is often desirable to lower the pH of the feed solution to protonate the contaminant. For example, removal of phosphate is facilitated by lowering the pH of the solution to about 3, which protonates the phosphate anion, allowing passage through a membrane.
- the pH will generally between about pH 1 and about pH 7.
- the pH of the feed solution can be adjusted to between about pH 7 and about pH 14.
- one aspect of the invention involves modulating a separation by adjusting the pH of a solution in contact with the membrane; this can change the ionic charge of a contaminant and can also affect the surface charge of the membrane, thus facilitating purification if the desired carbohydrate.
- the manufacturer's instructions must be followed as to acceptable pH range for a particular membrane to avoid damage to the membrane.
- a mixture is first subjected to nanofiltration or reverse osmosis at one pH, after which the retentate containing the saccharide of interest is adjusted to a different pH and subjected to an additional round of membrane purification.
- filtration of a reaction mixture used to synthesize sialyl lactose through an Osmonics MX07 membrane (a nanofiltration membrane having a MWCO of about 500 Da) at pH 3.0 will retain the sialyl lactose and remove most phosphate, pyruvate, salt and manganese from the solution, while also removing some of the GlcNAc, lactose, and sialic acid.
- a saccharide is to be purified from a mixture that contains proteins, such as enzymes used to synthesize a desired oligosaccharide or nucleotide sugar, it is often desirable to remove the proteins as a first step of the purification procedure.
- proteins such as enzymes used to synthesize a desired oligosaccharide or nucleotide sugar
- this separation is accomplished by choosing a membrane that has an MWCO which is less than the molecular mass of the protein or other macromolecule to be removed from the solution, but is greater than the molecular mass of the oligosaccharide being purified (i.e., the rejection ratio in this case is higher for the protein than for the desired saccharide).
- UF membranes that are suitable for use in the methods of the invention are available from several commercial manufacturers, including Millipore Corp. (Bedford, Mass.), Osmonics, Inc. (Minnetonka, Minn.), Filmtec (Minneapolis, Minn.), UOP, Desalination Systems, Advanced Membrane Technologies, and Nitto.
- the invention also provides methods for removing salts and other low molecular weight components from a mixture containing a saccharide of interest by using a nanofiltration (NF) or a reverse osmosis (RO) membrane.
- Nanofiltration membranes are a class of membranes for which separation is based both on molecular weight and ionic charge. These membranes typically fall between reverse osmosis and ultrafiltration membranes in terms of the size of species that will pass through the membrane.
- Nanofiltration membranes typically have micropores or openings between chains in a swollen polymer network. Molecular weight cut-offs for non-ionized molecules are typically in the range from 100-20,000 Daltons.
- a nanofiltration membrane useful in the methods of the invention will typically have a retention characteristic for the saccharide of interest of from about 40% to about 100%, preferably from about 70% to about 100%, more preferably from about 90% to about 100%.
- the nanofilter membranes used in the invention can be any one of the conventional nanofilter membranes, with polyamide membranes being particularly suitable.
- Several commercial manufacturers including Millipore Corp. (Bedford, Mass.), Osmonics, Inc. (Minnetonka, Minn.), Filmtec, UOP, Advanced Membrane Technologies, Desalination Systems, and Nitto, among others, distribute nanofiltration membranes that are suitable for use in the methods of the invention.
- suitable membranes include the Osmonics MX07, YK, GH (G-10), GE (G-5), and HL membranes, among others.
- RO membranes also allow a variety of aqueous solutes to pass through them while retaining selected molecules.
- osmosis refers to a process whereby a pure liquid (usually water) passes through a semipermeable membrane into a solution (usually sugar or salt and water) to dilute the solution and achieve osmotic equilibrium between the two liquids.
- reverse osmosis is a pressure driven membrane process wherein the application of external pressure to the membrane system results in a reverse flux with the water molecules passing from a saline or sugar solution compartment into the pure water compartment of the membrane system.
- a RO membrane which is semipermeable and non-porous, requires an aqueous feed to be pumped to it at a pressure above the osmotic pressure of the substances dissolved in the water.
- An RO membrane can effectively remove low molecular weight molecules ( ⁇ 200 Daltons) and also ions from water.
- the reverse osmosis membrane will have a retention characteristic for the saccharide of interest of from about 40% to about 100%, preferably from about 70% to about 100%, and more preferably from about 90% to about 100%.
- Suitable RO membranes include, but are not limited to, the Filmtec BW-30, Filmtec SW-30, Filmtec SW-30HR, UOP RO membranes, Desal RO membranes, Osmonics RO membranes, Advanced Membrane Technologies RO membranes, and the Nitto RO membranes, among others.
- a suitable RO membrane is Millipore Cat. No. CDRN500 60 (Millipore Corp., Bedford Mass.).
- the membranes used in the invention may be employed in any of the known membrane constructions.
- the membranes can be flat, plate and frame, tubular, spiral wound, hollow fiber, and the like.
- the membrane is spiral wound.
- the membranes can be employed in any suitable configuration, including either a cross-flow or a depth configuration.
- cross-flow which is preferred for ultrafiltration, nanofiltration and reverse osmosis purifications according to the invention
- the “feed” or solution from which the carbohydrate of interest is to be purified flows through membrane channels, either parallel or tangential to the membrane surface, and is separated into a retentate (also called recycle or concentrate) stream and a permeate stream.
- retentate also called recycle or concentrate
- the feed stream should flow, at a sufficiently high velocity, parallel to the membrane surface to create shear forces and/or turbulence to sweep away accumulating particles rejected by the membrane.
- Cross-flow filtration thus entails the flow of three streams—feed, permeate and retentate.
- a “dead end” or “depth” filter has only two streams—feed and filtrate (or permeate).
- the recycle or retentate stream which retains all the particles and large molecules rejected by the membrane, can be entirely recycled to the membrane module in which the recycle stream is generated, or can be partially removed from the system.
- the methods of the invention are used to purify saccharides from lower molecular weight components, for example, the desired saccharides are contained in the retentate stream (or feed stream, for a depth filter), while the permeate stream contains the removed contaminants.
- the purification methods of the invention can be further optimized by adjusting the pressure, flow rate, and temperature at which the filtration is carried out.
- UF, NF, and RO generally require increasing pressures above ambient to overcome the osmotic pressure of the solution being passed through the membrane.
- the membrane manufacturers' instructions as to maximum and recommended operating pressures can be followed, with further optimization possible by making incremental adjustments.
- the recommended pressure for UF will generally be between about 25 and about 100 psi, for NF between about 50 psi and about 1500 psi, and for RO between about 100 and about 1500 psi.
- Flow rates of both the concentrate (feed solution) and the permeate can also be adjusted to optimize the desired purification.
- Typical flow rates for the concentrate (P c ) will be between about 1 and about 15 gallons per minute (GPM), and more preferably between about 3 and about 7 GPM.
- flow rates (Pf) of between about 0.05 GPM and about 10 GPM are typical, with flow rates between about 0.2 and about 1 GPM being preferred.
- the temperature at which the purification is carried out can also influence the efficiency and speed of the purification. Temperatures of between about 0 and about 100° C. are typical, with temperatures between about 20 and 40° C. being preferred for most applications. Higher temperatures can, for some membranes, result in an increase in membrane pore size, thus providing an additional parameter that one can adjust to optimize a purification.
- the filtration is performed in a membrane purification machine which provides a means for automating control of flow rate, pressure, temperature, and other parameters that can affect purification.
- a membrane purification machine which provides a means for automating control of flow rate, pressure, temperature, and other parameters that can affect purification.
- the Osmonics 213T membrane purification machine is suitable for use in the methods of the invention, as are machines manufactured by other companies listed above.
- the membranes can be readily cleaned either after use or after the permeability of the membrane diminishes. Cleaning can be effected at a slightly elevated temperature if so desired, by rinsing with water or a caustic solution. If the streams contain small amounts of enzyme, rinsing in the presence of small amounts of surfactant, for instance ULTRASIL, is useful. Also, one can use prefilters (100-200 ⁇ m) to protect the more expensive nanofiltration membranes. Other cleaning agents can, if desired, be used. The choice of cleaning method will depend on the membrane being cleaned, and the membrane manufacturer's instructions should be consulted. The cleaning can be accomplished with a forward flushing or a backward flushing.
- the purification methods of the invention can be used alone or in combination with other methods for purifying carbohydrates.
- an ion exchange resin can be used to remove particular ions from a mixture containing a saccharide of interest, either before or after nanofiltration/reverse osmosis, or both before and after filtration. Ion exchange is particularly desirable if it is desired to remove ions such as phosphate and nucleotides that remain after a first round of nanofiltration or reverse osmosis.
- this can be accomplished, for example, by adding an anion exchange resin such as AG1X-8 (acetate form, BioRad; see, e.g., BioRad catalog for other ion exchange resins) to a retentate that is at about pH 3.0 or lower until the phosphate concentration is reduced as desired.
- AG1X-8 acetate form, BioRad; see, e.g., BioRad catalog for other ion exchange resins
- acetic acid is released, so one may wish to follow the ion exchange with an additional purification through the nanofiltration or reverse osmosis system.
- one can circulate the pH 3.0 or lower solution through an Osmonics MX07 or similar membrane until the conductivity of the permeate is low and stabilized.
- the pH of the solution can then be raised to 7.4 with NaOH and the solution recirculated through the same membrane to remove remaining sodium acetate and salt. Cations can be removed in a similar manner; for example, to remove Mn 2+ , an acidic ion exchange resin can be used, such as AG50WX8 (He) (BioRad).
- the purification methods of the invention are particularly useful for purifying oligosaccharides, modified saccharides, nucleotide sugars and modified nucleotide sugars prepared using enzymatic synthesis.
- Enzymatic synthesis using glycosyltransferases provides a powerful method for preparing oligosaccharides; for some applications it is desirable to purify the oligosaccharide from the enzymes and other reactants in the enzymatic synthesis reaction mixture.
- Preferred methods for producing many oligosaccharides involve glycosyl transferase cycles, which produce at least one mole of inorganic pyrophosphate for each mole of product formed and are typically carried out in the presence of a divalent metal ion.
- glycosyltransferase cycles are the sialyltransferase cycles, which use one or more enzymes as well as other reactants. See, e.g., U.S. Pat. No. 5,374,541 WO 9425615 A, PCT/US96/04790, and PCT/US96/04824.
- a reaction used for synthesis of sialylated oligosaccharides can contain a sialyltransferase ( FIG. 7 ), a CMP-sialic acid synthetase, a sialic acid, an acceptor for the sialyltransferase, CTP, and a soluble divalent metal cation.
- ⁇ (2,3)sialtransferase (EC 2.4.99.6) transfers sialic acid to the non-reducing terminal Gal of a Gal ⁇ 163Glc disaccharide or glycoside. See, Van den Eijnden et al., J. Biol. Chem., 256:3159 (1981), Weinstein et al., J. Biol. Chem., 257:13845 (1982) and Wen et al, J. Biol. Chem., 267:21011 (1992).
- Another exemplary ⁇ 2,3-sialyltransferase (EC 2.4.99.4) transfers sialic acid to the non-reducing terminal Gal of the disaccharide or glycoside. See, Rearick et al., J. Biol. Chem., 254:4444 (1979) and Gillespie et al., J. Biol. Chem., 267:21004 (1992). Further exemplary enzymes include Gal- ⁇ -1,4-GlcNAc ⁇ -2,6 sialyltransferase (See, Kurosawa et al. Eur. J. Biochem. 219: 375-381 (1994)).
- the reaction mixture will also contain an acceptor for the sialyltransferase, preferably having a galactosyl unit.
- Suitable acceptors include, for example, Gal ⁇ 1 ⁇ 3 GalNAc, lacto-N-tetraose, Gal ⁇ 1 ⁇ 3 GlcNAc, Gal ⁇ 1 ⁇ 3Ara, Gal ⁇ 1 ⁇ 6GlcNAc, Gal ⁇ 1 ⁇ 4Glc (lactose), Gal ⁇ 1 ⁇ 4Glc ⁇ 1-OCH 2 CH 3 , Gal ⁇ 1 ⁇ 4Glc ⁇ 1-OCH 2 CH 2 CH 3 , Gal ⁇ 1 ⁇ 4Glc ⁇ 1-OCH 2 C 6 H 5 , Gal ⁇ 1 ⁇ 4GlcNAc, Gal ⁇ 1-OCH 3 , melibiose, raffinose, stachyose, and lacto-N-neotetraose (LNnT).
- LNnT lacto-N-neotetraose
- the sialic acid present in the reaction mixture can include not only sialic acid itself (5-N-acetylneuraminic acid; 5-N-acetylamino-3,5-dideoxy-D-glycero-D-galacto-2-nonulosonic acid; NeuAc, and sometimes also abbreviated AcNeu or NANA), but also 9-substituted sialic acids such as a 9-O—C 1 -C 6 acyl-NeuAc like 9-O-lactyl-NeuAc or 9-O-acetyl-NeuAc, 9-deoxy-9-fluoro-NeuAc and 9-azido-9-deoxy-NeuAc.
- the synthesis and use of these compounds in a sialylation procedure is described in international application WO 92/16640, published Oct. 1, 1992.
- the reaction medium can further comprise a CMP-sialic acid recycling system comprising at least 2 moles of phosphate donor per each mole of sialic acid, and catalytic amounts of an adenine nucleotide, a kinase capable of transferring phosphate from the phosphate donor to nucleoside diphosphates, and a nucleoside monophosphate kinase capable of transferring the terminal phosphate from a nucleoside triphosphate to CMP.
- a CMP-sialic acid recycling system comprising at least 2 moles of phosphate donor per each mole of sialic acid, and catalytic amounts of an adenine nucleotide, a kinase capable of transferring phosphate from the phosphate donor to nucleoside diphosphates, and a nucleoside monophosphate kinase capable of transferring the terminal phosphate from a nucleoside triphosphate to CMP.
- a suitable CMP-sialic acid regenerating system comprises cytidine monophosphate (CMP), a nucleoside triphosphate (for example adenosine triphosphate (ATP), a phosphate donor (for example, phosphoenolpyruvate or acetyl phosphate), a kinase (for example, pyruvate kinase or acetate kinase) capable of transferring phosphate from the phosphate donor to nucleoside diphosphates and a nucleoside monophosphate kinase (for example, myokinase) capable of transferring the terminal phosphate from a nucleoside triphosphate to CMP.
- CMP cytidine monophosphate
- a nucleoside triphosphate for example adenosine triphosphate (ATP)
- a phosphate donor for example, phosphoenolpyruvate or acetyl phosphate
- reaction medium will preferably further comprise a phosphatase.
- Pyruvate is a byproduct of the sialyltransferase cycle and can be made use of in another reaction in which N-acetylmannosamine (ManNAc) and pyruvate are reacted in the presence of NeuAc aldolase (EC 4.1.3.3) to form sialic acid.
- ManNAc N-acetylmannosamine
- NeuAc aldolase EC 4.1.3.3
- advantage can be taken of the isomerization of GlcNAc to ManNAc, and the less expensive GlcNAc can be used as the starting material for sialic acid generation.
- the sialic acid can be replaced by ManNAc (or GlcNAc) and a catalytic amount of NeuAc aldolase.
- NeuAc aldolase also catalyzes the reverse reaction (NeuAc to ManNAc and pyruvate)
- the produced NeuAc is irreversibly incorporated into the reaction cycle via CMP-NeuAc catalyzed by CMP-sialic acid synthetase.
- the starting material, ManNAc can also be made by the chemical conversion of GlcNAc using methods known in the art (see, e.g., Simon et al., J. Am. Chem. Soc. 110:7159 (1988).
- the reaction medium will preferably contain, in addition to a galactosyltransferase, donor substrate, acceptor sugar and divalent metal cation, a donor substrate recycling system comprising at least 1 mole of glucose-1-phosphate per each mole of acceptor sugar, a phosphate donor, a kinase capable of transferring phosphate from the phosphate donor to nucleoside diphosphates, and a pyrophosphorylase capable of forming UDP-glucose from UTP and glucose-1-phosphate and catalytic amounts of UDP and a UDP-galactose-4-epimerase.
- a donor substrate recycling system comprising at least 1 mole of glucose-1-phosphate per each mole of acceptor sugar, a phosphate donor, a kinase capable of transferring phosphate from the phosphate donor to nucleoside diphosphates, and a pyrophosphorylase capable of forming UDP-glucose from UTP and glucose-1-phosphat
- Exemplary galactosyltransferases include ⁇ (1,3) galactosyltransferase (E.C. No. 2.4.1.151, see, e.g., Dabkowski et al., Transplant Proc. 25: 2921 (1993) and Yamamoto et al., Nature 345:229-233 (1990)) and ⁇ (1,4) galactosyltransferase (E.C. No. 2.4.1.38).
- Oligosaccharides synthesized by other enzymatic methods can also be purified by the methods of the invention.
- the methods are useful for purifying oligosaccharides produced in non-cyclic or partially cyclic reactions such as simple incubation of an activated saccharide and an appropriate acceptor molecule with a glycosyltransferase under conditions effective to transfer and covalently bond the saccharide to the acceptor molecule.
- Glycosyltransferases which include those described in, e.g., U.S. Pat. No. 5,180,674, and International Patent Publication Nos.
- glycosyltransferases encoded by the los locus of Neisseria can be bound to a cell surface or unbound. Oligosaccharides that can be obtained using these glycosyltransferases include, for example,
- sialic acid and any sugar having a sialic acid moiety are sialic acid and any sugar having a sialic acid moiety.
- exemplary species include sialic acid species modified with a linker (e.g., glycyl sialic acid) and with a polymer (e.g., poly(ethylene glycol).
- linker e.g., glycyl sialic acid
- polymer e.g., poly(ethylene glycol
- Other compounds include sialyl galactosides, including the sialyl lactosides, as well as compounds having the formula:
- R′ is alkyl or acyl from 1-18 carbons, 5,6,7,8-tetrahydro-2-naphthamido; benzamido; 2-naphthamido; 4-aminobenzamido; or 4-nitrobenzamido.
- R is a hydrogen, a alkyl C 1 -C 6 , a saccharide, an oligosaccharide or an aglycon group having at least one carbon atom.
- A represents an alkylene group of from 1 to 18 carbon atoms optionally substituted with halogen, thiol, hydroxy, oxygen, sulfur, amino, imino, or alkoxy
- Z is hydrogen, —OH, —SH, —NH 2 , —NHR 1 , —N(R 1 ) 2 , —CO 2 H, —CO 2 R 1 , —CONH 2 , —CONHR 1 , —CON(R 1 ) 2 , —CONHNH 2 , or —OR 1 wherein each R 1 is independently alkyl of from 1 to 5 carbon atoms.
- R can be:
- R can also be 3-(3,4,5-trimethoxyphenyl)propyl.
- the present invention is also useful for purifying a variety of compounds that comprise selectin-binding carbohydrate moieties.
- selectin-binding moieties have the general formula:
- R 0 is (C 1 -C 8 alkyl)carbonyl, (C 1 -C 8 alkoxy)carbonyl, or (C 2 -C 9 alkenyloxy)carbonyl
- R 1 is an oligosaccharide or a group having the formula:
- R 3 and R 4 may be the same or different and may be H, C 1 -C 8 alkyl, hydroxy-(C 1 -C 8 alkyl), aryl-(C 1 -C 8 alkyl), or (C 1 -C 8 alkoxy)-(C 1 -C 8 alkyl), substituted or unsubstituted.
- R 2 may be H, C 1 -C 8 alkyl, hydroxy-(C 1 -C 8 alkyl), aryl-(C 1 -C 8 -alkyl), (C 1 -C 8 alkyl)-aryl, alkylthio, ⁇ 1,2Man, ⁇ 1,6GalNAc, ⁇ 1,3Gal ⁇ 1,4Glc, ⁇ 1,2Man-R 8 , ⁇ 1,6GalNAc-R 8 , and ⁇ 1,3Gal-R 8 .
- R 8 may be H, C 1 -C 8 alkyl, C 1 -C 8 alkoxy, hydroxy-(C 1 -C 8 alkyl), aryl-(C 1 -C 8 alkyl), (C 1 -C 8 alkyl)-aryl, or alkylthio.
- m and n are integers and may be either 3 or 4; p may be zero or 1.
- substituted groups mentioned above may be substituted by hydroxy, hydroxy(C 1 -C 4 alkyl), polyhydroxy(C 1 -C 4 alkyl), alkanoamido, or hydroxyalknoamido substituents.
- Preferred substituents include hydroxy, polyhydroxy(C 3 alkyl), acetamido and hydroxyacetamido.
- a substituted radical may have more than one substitution, which may be the same or different.
- the oligosaccharide is preferably a trisaccharide.
- Preferred trisaccharides include NeuAc ⁇ 2,3Gal ⁇ 1,4GlcNAc ⁇ 1,3 or NeuGc ⁇ 2,3Gal ⁇ 1,4GlcNAc ⁇ 1,3.
- R 1 is the group having the formula
- R 3 and R 4 preferably form a single radical having the formula
- R 5 is C 3 -C 7 divalent alkyl, substituted or unsubstituted
- R 6 and R 7 are the same or different and are C 1 -C 6 divalent alkyl, substituted or unsubstituted.
- q and r are integers which may be the same or different and are either zero or 1. The sum of q and r is always at least 1.
- a more preferred structure for a single radical formed by R 3 and R 4 is one having the formula
- R 6 is C 3 -C 4 divalent alkyl, substituted or unsubstituted.
- R 6 may have the formula —CH 2 —CH 2 —CH 2 —CH 2 —, preferably substituted.
- the radical can be substituted with hydroxy, polyhydroxy(C 3 alkyl), and substituted or unsubstituted alkanoamido groups, such as acetamido or hydroxyacetamido.
- the substituted structure will typically form a monosaccharide, preferably a sialic acid such as NeuAc or NeuGc linked ⁇ 2,3 to the Gal residue.
- both m and n are integers and can be either 3 or 4.
- Gal is linked ⁇ 1,4 and Fuc is linked ⁇ 1,3 to GlcNAc.
- This formula includes the SLe x tetrasaccharide.
- SLe x has the formula NeuAc ⁇ 2,3Gal ⁇ 1,4(Fuc ⁇ 1,3)GlcNAc ⁇ 1-. This structure is selectively recognized by LECCAM-bearing cells.
- Other compounds that one can purify using the methods include those described in U.S. Pat. No. 5,604,207 having the formula:
- Z is hydrogen, C 1 -C 6 acyl or
- R 1 is selected from the group consisting of C(O), SO 2 , HNC(O), OC(O) and SC(O).
- R 1 is selected from the group consisting of an aryl, a substituted aryl and a phenyl C 1 -C 3 alkylene group, wherein said aryl substitutent is selected from the group consisting of a halo, trifluoromethyl, nitro, C 1 -C 18 alkyl, C 1 -C 18 alkoxy, amino, mono-C 1 -C 18 alkylamino, di-C 1 -C 18 alkylamino, benzylamino, C 1 -C 18 alkylbenzylamino, C 1 -C 18 thioaklyl and C 1 -C 18 alkyl carboxamido groups, or R 1 Y is allyloxycarbonyl or chloroacetyl.
- monosaccharide including
- R 5 is selected from the group consisting of hydrogen, benzyl, methoxybenzyl, dimethoxybenzyl and C 1 -C 6 acyl.
- R 7 is methyl or hydroxymethyl.
- X is selected from the group consisting of C 1 -C 6 acyloxy, C 2 -C 6 hydroxylacyloxy, hydroxy, halo and azido.
- a related set of structures included in the general formula are those in which Gal is linked ⁇ 1,3 and Fuc is linked ⁇ 1,4.
- the tetrasaccharide, NeuAc ⁇ 2,3Gal ⁇ 1,3(Fuc ⁇ 1,4)GlcNAc ⁇ 1- termed here SLe a
- selectin receptors See, Berg et al., J. Biol. Chem., 266:14869-14872 (1991).
- Berg et al. showed that cells transformed with E-selectin cDNA selectively bound neoglycoproteins comprising SLe a .
- compounds that can be purified according to the invention are lacto-N-neotetraose (LNnT), GlcNAc ⁇ 1,3Gal ⁇ 1,4Glc (LNT-2), sialyl( ⁇ 2,3)-lactose, and sialyl( ⁇ 2,6)-lactose.
- a modified sialic acid has the following structure:
- R 1 , R 2 , R 3 and R 4 are each independently selected from H, OR 5 , NR 6 R 7 , substituted alkyl, unsubstituted alkyl, substituted heteroalkyl and unsubstituted heteroalkyl.
- R 5 is H, substituted alkyl, unsubstituted alkyl, substituted heteroalkyl or unsubstituted heteroalkyl.
- the symbols R 6 and R 7 independently represent H, substituted alkyl, unsubstituted alkyl, substituted heteroalkyl and unsubstituted heteroalkyl.
- B is a nucleoside. Exemplary nucleosides include AMP, UMP, GMP, CMP, TMP, ADP, UDP, GDP, CDP, TDP, ATP, UTP, GTP, CTP, TTP, cAMP and cGMP.
- the sialic acid is modified with a linker group.
- Preferred sites for such modification are R 1 or R 2 .
- at least one of R 1 and R 2 includes a linker.
- An exemplary linker is a glycyl linker.
- the modified sialic acid has the following structure:
- a modifying group is attached to the sialic acid through the linker.
- An exemplary species according to this description includes a modifying group attached through the free amine moiety of the linker in the figure above.
- a presently preferred modifying group is a water-soluble polymer. Poly(ethylene glycol) is a preferred water-soluble polymer.
- the methods of the invention are useful not only for purifying carbohydrates (and modified carbohydrates and nucleotide sugars) that that are newly synthesized, but also those that are the products of degradation, e.g., enzymatic degradation. See, e.g., Sinnott, M. L., Chem. Rev. 90: 1171-1202 (1990) for examples of enzymes that catalyze degradation of oligosaccharides.
- the invention also provides methods for purifying nucleotides, nucleotide sugars, and related compounds.
- a nucleotide sugar such as GDP-fucose, GDP-mannose, CMP-NeuAc, UDP-glucose, UDP-galactose, UDP-N-acetylgalactosamine, and the like, can be purified by the methods described herein.
- the methods are also useful for purifying nucleotides and nucleotides in various states of phosphorylation (e.g., CMP, CDP, CTP, GMP, GDP, GTP, TMP, TDP, TTP, AMP, ADP, ATP, UMP, UDP, UTP), as well as the deoxy forms of these and other nucleotides, including modified nucleotides.
- states of phosphorylation e.g., CMP, CDP, CTP, GMP, GDP, GTP, TMP, TDP, TTP, AMP, ADP, ATP, UMP, UDP, UTP
- the method of the invention can be used to prepare and purify nucleotide sugars to a high degree of purity on a multi-kilogram scale (e.g., at least about 1 kg, preferably at least about 1.5 kg, more preferably at least about 2 kg, and even more preferably, at least about 3 kg of purified sugar nucleotide per synthesis/purification run).
- a multi-kilogram scale e.g., at least about 1 kg, preferably at least about 1.5 kg, more preferably at least about 2 kg, and even more preferably, at least about 3 kg of purified sugar nucleotide per synthesis/purification run.
- nucleotide sugars e.g., those bearing a linker arm (e.g., a glycl linker arm), a modifying group (e.g., a water-soluble polymer (e.g., PEG)), or a modifying group attached to the linker arm (e.g., PEG attached to the sugar through a glycyl linker).
- a linker arm e.g., a glycl linker arm
- a modifying group e.g., a water-soluble polymer (e.g., PEG)
- a modifying group attached to the linker arm e.g., PEG attached to the sugar through a glycyl linker
- the process of the invention routinely provides nucleotide sugars, e.g., CMP-NAN, in recovered yields of purified materials in greater that 40%, e.g., of from about 40% to about 80%.
- the yield of isolated CMP-NAN is from about 50% to about 70% of the theoretical synthesis yield.
- the process of the invention provides nucleotide sugars that are at least 80% pure, preferably at least 85% pure, more preferably, at least 90% pure and still more preferably, at least 95% pure.
- the nucleotide sugar is a CMP-sialic acid, e.g., CMP-NAN (N-acetylneuraminic acid).
- CMP-NAN N-acetylneuraminic acid
- a membrane-based methodology is utilized to purify the nucleotide sugar from reaction components.
- exemplary reaction components include cytidine monophosphate and its active analogues, and cytidine diphosphate, unreacted sialic acid, salts (e.g., PO 4 3 ⁇ , Mn 2+ ).
- the amount of CMP, CDP and/or CTP of the product is less than about 20%, preferably, less than about 15%, more preferably, less than about 10% and still more preferably less than 5%.
- the content of unreacted sialic acid, e.g., NAN, in the final product is less than about than about 20%, preferably, less than about 15%, more preferably, less than about 10%, still more preferably, less than about 5% and even more preferably, less than about 2%.
- the phosphate content of the final product is less than about 5%, preferably, less than about 2%, and more preferably, about 0%.
- the invention also provides methods for synthesizing and purifying nucleotide sugars.
- the nucleotide sugar is enzymatically synthesized from a nucleotide and a sugar in the presence of an enzyme. After the nucleotide sugar is synthesized, the nucleotide sugar is purified according to a method of the invention.
- a nucleotide sugar solution is optionally clarified by filtration.
- the nucleotide sugar solution passes through a membrane bag filter in which contaminating salts and other undesired contaminants are filtered out of the nucleotide sugar solution.
- the clarification step can be incorporated at any step of the process.
- the nucleotide sugar solution is clarified after synthesis of the nucleotide sugar.
- the nucleotide sugar solution may be clarified one or more times.
- the nucleotide sugar solution is purified using hollow fiber filtration.
- Hollow fiber filtration removes proteins introduced by the enzyme preparation of the nucleotide sugar.
- the hollow fiber membrane retains proteins from the enzyme preparation while allowing for passage of the nucleotide sugar solution through the membrane.
- the hollow fiber membrane comprises a hollow fiber membrane with a tangential filtration skid.
- the hollow fiber filtration step can be incorporated at any step of the process.
- the nucleotide sugar solution goes through hollow fiber filtration after clarification.
- the nucleotide sugar solution goes through hollow fiber filtration after synthesis of the nucleotide sugar.
- the nucleotide sugar solution may be filtered one or more times using hollow fiber filtration.
- the nucleotide sugar solution is purified using nanofiltration.
- Nanofiltration removes salts and other low molecular weight components from a mixture.
- Nanofiltration membranes separate molecules based on molecular weight and ionic charge. Molecular weight-cutoffs for non-ionized molecules are typically in the range from 100-20,000 daltons.
- saccharides of interest will be retained by the nanofiltration membrane and contaminating salts and other undesired components will pass through.
- the nanofiltration step can be incorporated at any step of the process.
- the nucleotide sugar solution goes through hollow-fiber filtration first and then nanofiltration.
- the nucleotide sugar solution goes through nanofiltration first and then hollow fiber filtration.
- the nucleotide sugar solution may be purified using either hollow-fiber filtration or nanofiltration.
- the nucleotide sugar solution goes through nanofiltration after clarification.
- the nucleotide sugar solution goes through nanofiltration after synthesis of the nucleotide sugar.
- the nucleotide sugar solution may be filtered one or more times using nanofiltration. After nanofiltration, the purified nucleotide sugar solution may generally be stored or may undergo further purification.
- the nucleotide sugar solution may optionally be decolorized (e.g., by passing the solution over activate carbon).
- decolorization involves passing the nucleotide sugar solution over a pre-packed column of activated carbon attached to a chromatography system. Decolorization can be incorporated at any step of the process.
- the nucleotide sugar solution is decolorized after nanofiltration.
- the nucleotide sugar solution is decolorized after hollow-fiber filtration.
- the nucleotide sugar solution is decolorized after clarification. The nucleotide sugar solution may be decolorized one or more times.
- the nucleotide sugar solution is purified using a charged depth media filter.
- the charged depth media filter removes endotoxins from the nucleotide sugar solution. Endotoxins are toxic, natural compounds such as lipopolysaccharides found inside pathogens on the outer cell wall of bacteria. Purification by a charged depth media filter can be incorporated at any step of the process.
- the nucleotide sugar solution is filtered after decolorization.
- nucleotide sugar solution is purified by a charged depth media filter after nanofiltration. In yet another embodiment, the nucleotide sugar solution is purified by a charged depth media filter after hollow-fiber filtration. In another embodiment, the nucleotide sugar solution is purified by a charged depth media filter after clarification. In another embodiment, the nucleotide sugar solution is purified by a charged depth media filter after synthesis of the nucleotide sugar. The nucleotide sugar solution may be filtered one or more times using a charged depth media filter.
- the nucleotide sugar solution is purified using a sterile filter.
- the sterile filter removes contaminating salts and other undesired contaminants from the nucleotide sugar solution.
- the sterile filter is pre-packaged and sterilized with a bag manifold system for final filtration and storage. Purification by a sterile filter can be incorporated at any step of the process.
- the nucleotide sugar solution is filtered by a sterile filter after purification by a charged depth media filter.
- the nucleotide sugar solution is purified by a sterile filter after decolorization.
- the nucleotide sugar solution is purified by a sterile filter after nanofiltration. In another embodiment, the nucleotide sugar solution is purified by a sterile filter after hollow fiber filtration. In another embodiment, the nucleotide sugar solution is purified by a sterile filter after clarification. In another embodiment, the nucleotide sugar solution is purified by a sterile filter after synthesis of the nucleotide sugar. The nucleotide sugar solution may be filtered one or more times using a sterile filter.
- the nucleotide sugar is first synthesized (1) from a nucleotide and a sugar in the presence of an enzyme.
- the nucleotide sugar is clarified by filtration (2) and transferred into a mobile tank (3).
- the clarified nucleotide sugar solution is concentrated using a hollow fiber filtration unit with a tangential flow filtration skid and diafiltered with purified water (4) and (5).
- the nucleotide sugar solution is transferred to a mobile tank (6) and the pH is adjusted.
- the purified solution is concentrated again and diafiltered with purified water using a nanofiltration system (7) and (8).
- the nucleotide sugar solution is then decolorized in which the color is removed from the nucleotide sugar solution (9).
- the nucleotide sugar solution subsequently undergoes two filtrations.
- the nucleotide sugar solution is filtered using a charged depth media filter in which endotoxins are removed (10).
- the charged depth media filter is a CUNO Zeta Plus 60 ZA filter or an equivalent.
- the nucleotide sugar solution is optionally filtered using a 0.2 ⁇ m sterile filter (11).
- the sterile filter is a CUNO LifeASSURE 0.2 ⁇ m sterile filter or an equivalent.
- the purified nucleotide sugar solution is appropriate for storage.
- nucleotide sugars that can be purified by the method described above include, but are not limited to, CMP-NAN, GDP-fucose, GDP-mannose, CMP-NeuAc, UDP-Glucose, UDP-galactose and UDP-N-acetylgalactosamine, and modified analogues thereof.
- the nucleotide sugar is CMP-NAN or a modified CMP-NAN.
- the invention also provides methods for synthesizing and purification of a nucleotide-Glycyl Sialic Acid (“nucleotide-GSC”).
- the synthesis of the nucleotide-GSC begins with the synthesis of the protected Fmoc-glycyl-mannosamine (“FGM”).
- FGM Fmoc-glycyl-mannosamine
- mannosamine and Fmoc-Glycyl-OSU are reacted in an aqueous solution under basic conditions.
- the aqueous solution may contain a base, e.g., sodium methoxide and an organic cosolvent, e.g., methanol, to facilitate the reaction.
- the FGM is optionally purified.
- FGM is purified by chromatography, e.g., silica gel chromatography. FGM may be chromatographed one or more times.
- FGM converted to the corresponding sialic acid analogue by reaction with pyruvate to form Fmoc-glycyl-sialic acid (“FSC”).
- FSC Fmoc-glycyl-sialic acid
- This reaction is efficiently catalyzed by a sialic acid aldolase.
- Appropriate sialic acid aldolases are commercially available.
- the reaction mixture includes at least the nucleotide, the aldolase, MnCl 2 and water.
- the resulting FSC is then coupled to a nucleotide in the presence of an enzyme to form the resulting product, nucleotide-FSC.
- the nucleotide-FSC is optionally purified.
- the nucleotide-FSC is purified by chromatography, e.g., reverse phase chromatography.
- the reverse phase chromatography is C18 reverse phase chromatography.
- the nucleotide FSC can be filtered (e.g., 0.22 filter) prior to and/or after chromatography.
- the nucleotide-FSC is preferably deprotected to produce the free amine analogue of the linker-nucleotide sugar construct.
- deprotection is effected with methanol:water:dimethylamine. Deprotection results in a modified sialic acid, nucleotide-Glycyl-Sialic Acid (“GSC”).
- GSC nucleotide-Glycyl-Sialic Acid
- the GSC is optionally purified, filtered and/or lyophilized.
- FIG. 4 An exemplary process for nucleotide-GSC synthesis and purification is described in FIG. 4 .
- nucleotide-GSC The synthesis of nucleotide-GSC begins with the synthesis and concentration of FGM (12). FGM is then purified using silica flash column chromatography (13). FGM is reacted with pyruvate to form FSC (14). The resulting FSC is then coupled to a nucleotide in the presence of an enzyme to form the resulting product, nucleotide-FSC (14). The nucleotide-FSC is then purified (15). The nucleotide-FSC is deprotected (16) thus cleaving off the Fmoc group from the nucleotide-FSC. The resultant product is a nucleotide-GSC. The nucleotide-GSC is further purified and concentrated (16). The nucleotide-GSC is then lyophilized (17) and subsequently released for testing (18).
- An exemplary nucleotide that can be used for the synthesis and purification of a nucleotide-GSC includes, but is not limited to, CMP, CDP, CTP, GMP, GDP, GTP, TMP, TDP, TTP, AMP, ADP, ATP, UMP, UDP, UTP, as well as the deoxy forms of these and other nucleotides.
- the nucleotide is CMP.
- the invention provides also methods for synthesizing and purification of a nucleotide-Sialic Acid (“SA”)-PEG.
- SA nucleotide-Sialic Acid
- the synthesis of the nucleotide-SA-PEG begins with Fmoc-glycyl-mannosamine (“FGM”). Synthesis and purification of this starting material is discussed above.
- FGM Fmoc-glycyl-mannosamine
- GSC and methoxy-paranitrophenyl-carbomate-polyethylene glycol (“mPEG-pNP”) are combined in under conditions suitable to allow formation of a conjugate between the PEG and the free amine of the glycyl linker.
- the synthesis reaction is performed in a 80% THF: 20% H 2 O solution.
- nucleotide-SA-PEG is purified. In a preferred embodiment, the nucleotide-SA-PEG is purified by reverse phase chromatography.
- FIG. 5 An exemplary process of nucleotide-SA-PEG (“PSC”) synthesis and purification is described in FIG. 5 .
- nucleotide-SA-PEG The synthesis of nucleotide-SA-PEG begins with the synthesis of FGM (19). FGM is rotovapped (20). FGM is then purified using silica column chromatography (21). FGM is detected by a UV light (22) and rotovapped (23). FGM is reacted with pyruvate to form FSC (24). The resulting FSC is then coupled to a nucleotide in the presence of an enzyme to form the resulting product, nucleotide-FSC (24). The nucleotide-FSC is then purified using a 0.2 ⁇ m filter (25) and a C18 reverse phase chromatography (26). The solution is then detected by a UV ray (27).
- the nucleotide-FSC is deprotected (28) thus cleaving off the Fmoc group from the nucleotide-FSC.
- the resultant product is a nucleotide-GSC.
- the nucleotide-GSC is rotovapped (29).
- the nucleotide-GSC is further purified (30) by a 0.2 ⁇ m filter.
- the nucleotide-GSC is then lyophilized (31).
- the nucleotide-GSC is reacted with a mPEG-pNP to form nucleotide-SA-PEG (“PSC”) (32).
- the PSC is purified by reverse phase chromatography (33).
- the PSC is detected by UV light (34) and rotovapped (35).
- the PSC is then lyophilized (36).
- An exemplary nucleotide that can be used for the synthesis and purification of a nucleotide-SA-PEG includes, but is not limited to, CMP, CDP, CTP, GMP, GDP, GTP, TMP, TDP, TTP, AMP, ADP, ATP, UMP, UDP, UTP, as well as the deoxy forms of these and other nucleotides.
- the nucleotide is CMP.
- the invention further provides methods for purifying a glycosyltransferase.
- a glycosyltransferase solution is harvested, isolating the enzyme from cell culture and other debris to produce a suitable feed material for subsequent purification steps.
- the harvesting reaction occurs at about pH 6.
- the harvesting step can be incorporated at any step of the process.
- the glycosyltransferase is optionally precipitated from the solution.
- the glycosyltransferase is precipitated by addition of calcium chloride (“CaCl 2 ”) to the solution.
- the pH of the solution can be adjusted as appropriate.
- the pH of the precipitation reaction is adjusted to about 7.5.
- the mixture can be incubated for any suitable time under appropriate conditions.
- the precipitation step lasts for about 30 minutes at about 4° C.
- Other exemplary additives include EDTA.
- the recovery of glycosyltransferase after precipitation is about 80% to about 100%, preferably from about 90% to about 100%, even more preferably about 100%.
- the glycosyltransferase solution is subjected to membrane filtration.
- Exemplary membrane filters have a pore size of about 0.1 ⁇ m to about 0.5 ⁇ m, preferably about 0.1 ⁇ m to about 0.3 ⁇ m, and more preferably about 0.20 ⁇ m to about 0.25 ⁇ m.
- the filtration step can be incorporated at any step of the process.
- the glycosyltransferase solution is ultrafiltrated.
- the ultrafiltration membrane has a molecular weight cut-off (MWCO) between about 5 kDa and about 200 kDa.
- the glycosyltransferase solution is equilibrated with 15 mM sodium phosphate buffer, pH 7.5 and conductivity is 2 ms/cm. In another embodiment, the glycosyltransferase solution is equilibrated with 15 mM sodium phosphate, pH 7.5. In another embodiment, the glycosyltransferase solution is equilibrated with 15 mM sodium phosphate, 0.01M NaCl, pH 7.5. In another embodiment, the glycosyltransferase is equilibrated with 15 mM sodium phosphate, 0.05M NaCl, pH 7.5.
- the glycosyltransferase solution is equilibrated with 15 mM sodium phosphate, 0.10M NaCl, pH 7.5.
- the pH range of the sodium phosphate may range from about 5.5 to about 8.5.
- the pH range of the glycosyltransferase solution may range from about 6.5 to about 7.5.
- the test excipient includes, but is not limited to, glycerol, mannitol, sorbitol, sucrose and Tween-20.
- the percent recovery of glycosyltransferase after ultrafiltration is about 45% to about 85%.
- the percent recovery of glycosyltransferase after ultrafiltration is about 55% to about 75%. In a more preferred embodiment, the percent recovery of glycosyltransferase after ultrafiltration is about 65%.
- the ultrafiltration step can be incorporated at any step of the process. In a preferred embodiment, the glycosyltransferase solution is ultrafiltrated after the filtration step. In another preferred embodiment, the glycosyltransferase solution is ultrafiltrated after the precipitation step.
- the glycosyltransferase solution is ultrafiltrated after the harvesting step.
- the glycosyltransferase solution may be ultrafiltrated one or more times.
- the glycosyltransferase is purified by chromatography, e.g., ion exchange chromatography.
- the glycosyltransferase is purified using an anion exchange column.
- the anion exchange column is a Mustang Q or an equivalent.
- the recovery of glycosyltransferase after anion exchange chromatography is preferably from about 80% to about 100%.
- the recovery of glycosyltransferase after chromatography is about 90% to about 100%.
- the recovery of glycosyltransferase after chromatography is about 100%.
- the chromatography step can be incorporated at any step of the process.
- the glycosyltransferase is purified by chromatography after an ultrafiltration step. In a preferred embodiment, the glycosyltransferase is purified by chromatography after the filtration step. In another preferred embodiment, the glycosyltransferase is purified by chromatography after the precipitation step. In another preferred embodiment, the glycosyltransferase is purified by chromatography after the harvesting step.
- the glycosyltransferase solution purified using a cation exchange column In another exemplary embodiment, the glycosyltransferase solution purified using a cation exchange column.
- An exemplary cation exchange chromatography protocol utilizes a Unosphere S resin or an equivalent.
- the cation exchange column is a 30S or an equivalent.
- the cation exchange column is eluted with at least one buffer.
- the first buffer comprises a 15 mM sodium phosphate, pH 7.5.
- the column is further eluted with a second buffer.
- An exemplary second buffer includes 1M NaCl, 15 mM sodium phosphate, pH 7.5.
- Elution of the column is optionally performed while developing a eluant gradient.
- the added elution step has a rate of about 5 ms/cm to about 10 ms/cm.
- the recovery of glycosyltransferase after cation exchange chromatography is about 35% to about 75%.
- the recovery of glycosyltransferase after chromatography is about 45% to about 65%.
- the percent recovery of glycosyltransferase after chromatography is about 55%.
- the chromatography step can be incorporated at any step of the process.
- the glycosyltransferase is purified by cation exchange chromatography after the anion exchange chromatography step described in the immediately preceding paragraph. In a preferred embodiment, the glycosyltransferase is purified by anion or cation exchange chromatography after the ultrafiltration step. In another preferred embodiment, the glycosyltransferase is purified by ion exchange chromatography after the filtration step. In yet another preferred embodiment, the glycosyltransferase is purified by chromatography after the precipitation step. In another preferred embodiment, the glycosyltransferase solution is purified by chromatography after the harvesting step. The glycosyltransferase solution may be purified by chromatography one or more times.
- the glycosyltransferase is purified by hydroxyapatite (HA) chromatography.
- HA hydroxyapatite
- Exemplary hydroxyapatite sorbents are selected from ceramic and crystalline hydroxyapatite materials.
- the particle size of the ceramic hydroxyapatite sorbent is between about 20 ⁇ m and about 180 ⁇ m, preferably about 60 to about 100 ⁇ m, and, more preferably about 80 ⁇ m.
- the recovery of glycosyltransferase after HA chromatography is about 50% to about 90%, preferably from about 60% to about 90%.
- the percent recovery of glycosyltransferase after chromatography is about 72%.
- HA chromatography can be incorporated at any step of the process.
- the glycosyltransferase is purified by HA chromatography after the cation chromatography step described above in the immediately preceding paragraph.
- the glycosyltransferase is purified by HA chromatography after the anion exchange chromatography step described above.
- the glycosyltransferase is purified by HA chromatography after the ultrafiltration step.
- the glycosyltransferase is purified by HA chromatography after the filtration step.
- the glycosyltransferase is purified by HA chromatography after the precipitation step.
- the glycosyltransferase is purified by HA chromatography after the harvesting step.
- the glycosyltransferase solution may be purified by HA chromatography one or more times.
- the glycosyltransferase is purified by hydrophobic interaction chromatography (“HIC”).
- HIC hydrophobic interaction chromatography
- the hydrophobic moieties of the column matrix are selected from, but are not limited to, alkyl groups, aromatic groups and ethers.
- the HIC column is packed with a phenyl 650M resin, butyl 650M resin, phenyl HP resin.
- the glycosyltransferase solution is equilibrated with 0.5M ammonium sulfate, 20 mM sodium phosphate, pH 7.
- the glycosyltransferase is purified by HIC chromatography after purification by the HA chromatography step described above in the immediately preceding paragraph.
- the glycosyltransferase is purified by chromatography after the cation exchange chromatography step described above.
- the glycosyltransferase is purified by chromatography after the anion exchange chromatography step described above.
- the glycosyltransferase solution is purified by chromatography after the ultrafiltration step.
- the glycosyltransferase solution is purified by chromatography after the filtration step.
- the glycosyltransferase solution is purified by chromatography after the precipitation step. In another preferred embodiment, the glycosyltransferase solution is purified by chromatography after the harvesting step. The glycosyltransferase solution may be purified by chromatography one or more times.
- the glycosyltransferase can be purified by ultrafiltration.
- the glycosyltransferase solution is equilibrated with 5 mM Bis-Tris, 0.1M NaCl, 5% sorbitol, pH 6.5. It is generally preferred that the recovery of glycosyltransferase after ultrafiltration is about 80% to about 100%. In a preferred embodiment, the percent recovery of glycosyltransferase after chromatography is about 90% to about 100%.
- Ultrafiltration can be incorporated at any step of the process.
- the glycosyltransferase is purified by ultrafiltration after the HIC chromatography step in the immediately preceding paragraph described above.
- the glycosyltransferase is purified by ultrafiltration after purification by the HA chromatography step described in the preceding paragraph above.
- the glycosyltransferase is purified by ultrafiltration after the cation exchange chromatography step described above.
- the glycosyltransferase is purified by ultrafiltration after the anion exchange chromatography step described above.
- the glycosyltransferase is purified by a second ultrafiltration step after the first ultrafiltration step described above.
- the glycosyltransferase can be purified by ultrafiltration after the filtration step or precipitation steps.
- the glycosyltransferase is purified by ultrafiltration after the harvesting step. The glycosyltransferase may be purified by ultrafiltration one or more times.
- the total percent recovery of glycosyltransferase from the purification process is about 5% to about 45%. In a preferred embodiment, the percent recovery of glycosyltransferase from the purification process is about 15% to about 35%.
- the glycosyltransferase is first harvested (37). In an exemplary embodiment, the harvesting reaction takes place at pH 6. After harvesting, the glycosyltransferase is precipitated using calcium chloride (38). The glycosyltransferase is then filtered (39). After filtration, the glycosyltransferase undergoes ultrafiltration (40). The glycosyltransferase passes through an anion exchange column such as Mustang Q (41) or an equivalent. After passing through the anion exchange column, the glycosyltransferase passes through a cation exchange column, such as UNOsphere S (42) or an equivalent. The glycosyltransferase is then passed through a HA Type I chromatography column (43). The glycosyltransferase then undergoes ultrafiltration (44).
- an anion exchange column such as Mustang Q (41) or an equivalent.
- a cation exchange column such as UNOsphere S (42) or an equivalent.
- the glycosyltransferase is a sialyltransferase.
- the sialyltranferase includes, but is not limited to, ST6GalNac, ST3Gal3, ⁇ (2,3)-sialyltransferase, ⁇ (2,6)-sialyltransferase and ⁇ (2,8)-sialyltransferase.
- the sialyltransferase includes, but is not limited to, a sialyltranferase listed in the table in FIG. 7 .
- the sialyltransferase is ST6GalNac.
- FGM Fmoc-glycyl-mannosamine
- the FGM solution was loaded onto the column in a 50:50 CHCl 3 :CH 3 OH solution.
- the silica column was then washed with 18 column volumes (CV) of 3% CHCl 3 /97% CH 3 OH.
- FGM was eluted from the column using 14 CV of 15% CHCl 3 /85% CH 3 OH.
- Fractions containing material were pooled and then rotovapped (20° C.) to dryness and stored at 4° C. The average recovery for this step from the consistency batches was 73.8%.
- FGM Fmoc-Glycyl-Mannosamine
- the silica column was a Biotage 75M Silica column.
- the column volume for a 75M column was 0.5 L.
- CMP-Sialic Acid-PEG PSC
- GSC CMP-Glycyl-Sialic Acid
- the first step in the synthesis of GSC was the reaction of mannosamine with Fmoc-Gly-OSu in methanol under basic conditions.
- the resulting Fmoc-glycyl-mannosamine was purified on a silica flash chromatography column.
- the purified Fmoc-glycyl-mannosamine then entered a two step enzymatic reaction.
- Fmoc-glycyl-mannosamine (FGM) was reacted with pyruvate to convert to Fmoc-glycyl-sialic acid. This reaction was catalyzed by a commercially available sialic acid aldolase.
- Fmoc-glycyl-sialic acid was then coupled to cytidine-5′-monophosphate through a CMP-NAN synthetase catalyzed reaction with cytidine-5′-triphosphate.
- the resulting product, CMP-Fmoc-glycyl-sialic acid was purified on a C18 reverse phase column.
- CMP-SA-10K PEG and CMP-SA-20K PEG were synthesized in a single step reaction of GSC with the appropriately sized mPEG-pNP. This reaction was performed in an 80% THF:20% H 2 O solution. Purification of the final product, CMP-SA-PEG (PSC), was performed by reverse phase chromatography. CMP-SA-10K PEG was purified using a C8 reverse phase chromatography resin, and CMP-SA-10K PEG was purified using a C4 reverse phase chromatography resin. The eluent from the reverse phase column was dried by lyophilization, yielding a white powder of purified CMP-SA-PEG.
- the process was scaled to produce approximately 10 g of the GSC intermediate. Approximately 5.5 g of CMP-SA-20K was produced from 0.5 g of GSC, while approximately 5.0 g of CMP-SA-10K PEG was produced from 1.0 g of GSC. The final CMP-SA-PEG products were approximately 90% pure with the major impurities being CMP and sialic acid-PEG, the primary breakdown products of CMP-SA-PEG.
- FSC CMP-Fmoc-glycyl-sialic acid
- FGM Fmoc-glycyl-mannosamine
- Fmoc-glycyl-sialic acid was then coupled to cytidine-5′-monophosphate through a CMP-NAN synthetase catalyzed reaction with cytidine-5′-triphosphate.
- the pH of the reaction was controlled at 7.5 ⁇ 0.5. Temperature was controlled at 30 ⁇ 2° C., and the reaction was continuously agitated. The reaction reached completion in 6-24 hrs. and the extent-of-reaction was determined as a function of the FSC concentration ( ⁇ 14.0 g/L, 95% of theoretical conversion). The resulting product solution can be stored at 4° C. until the purification is executed or for up to 72 h. The average recovery for the step during consistency batches was 91.2%.
- CMP-Fmoc-glycyl-sialic acid consists of two steps: the clarification of the reaction solution by 0.2 ⁇ m filtration and the reverse phase chromatographic purification of the FSC.
- the solution was simply pumped through a Millipore Millipak 0.2 ⁇ m filter cartridge. Little backpressure was generated during this filtration. After the filtration was complete, the filter cake and filter were rinsed with 500 mL of purified water.
- the chromatographic purification was performed using a Biotage pre-packed C18 reverse phase column.
- the FSC was loaded in an aqueous solution.
- the FSC binds to the column, and the column was washed with six column volumes of purified water.
- the FSC was then eluted in 10% methanol in purified water.
- the purification step was performed using the Biotage chromatography system (Z-1405).
- CMP-Fmoc-Glycyl-Sialic Acid occurred in a 10% methanol:water solution in a reaction with dimethylamine.
- Dimethylamine 40 wt % in water
- the Fmoc group was cleaved off, resulting in key intermediate, CMP-5′-Glycyl-Sialic Acid (GSC).
- GSC CMP-5′-Glycyl-Sialic Acid
- the reaction was complete when the peak area ratio of GSC:FSC was greater than 20, as measured by HPLC.
- the resulting GSC solution was then rotovapped (30° C.) to about 35% of the original volume.
- the free Fmoc-derivative formed a white precipitate that needed to be removed from the GSC solution.
- This material was filtered through a Millipore Millipak-200 Filter Unit (0.22 ⁇ m). The filter was then rinsed with RO water.
- This GSC solution was stored at 4° C. or frozen at ⁇ 20° C. until lyophilization. The average recovery for this step from the consistency batches was 86.1%.
- Consistency batches were performed for 10K and 20K CMP-Sialic Acid-PEG after development of the synthesis and purification operations. These batches demonstrated that a reproducible process had been developed to produce high-purity CMP-SA-PEG with very low contaminant levels, suitable for the glycopegylation projects.
- 10K CMP-SA-PEG was produced at greater than 80% purity at overall process yields of approximately 60%, and 20K CMP-SA-PEG was produced at greater than 70% purity at overall process yields of approximately 50%.
- the products were low in endotoxin, bioburden, and protein, and NMR has shown that the balance of the material was nearly all mPEG-OH, a by-product of the synthesis process.
- CMP-SA-PEG was produced in a reaction of CMP-Glycyl-Sialic Acid (GSC) with paranitrophenyl-carbomate-polyethylene glycol (pNP-PEG).
- GSC CMP-Glycyl-Sialic Acid
- pNP-PEG paranitrophenyl-carbomate-polyethylene glycol
- a molar ratio of PSC to GSC (product to reactant) of greater than 7:1 is found for 10K PSC and greater than 5:1 for 20K PSC.
- reaction solution were then rotovapped at ⁇ 30° C. to less than 80% of its original volume to remove the THF.
- the remaining aqueous solution was then diluted to five times the original reaction volume (diluted to 5 L for 10K PSC and 2.5 L for 20K PSC) using RO water.
- the diluted solution was then adjusted to pH 9.5 ⁇ 0.1 with 1M NaOH and allowed to stir for at least 1 hr.
- This elevated pH caused breakdown of residual pNP-PEG to free paranitrophenol, carbon dioxide, and methoxyPEG-OH (mPEG-OH).
- the solution is adjusted to pH 8.0 ⁇ 0.1 using 1M HCl.
- This concentrated fraction pool was then freeze dried on a Labconco flask-style freeze-drier. Final testing was performed on the resulting powder from this drying.
- the syntheses was performed in 2 L and 5 L sealed bottles, temperature controlled in a shaker incubator, SI-0017, B. Braun Certomat BS1.
- Chromatographic purification was performed on the Biotage Flash Chromatography System, Z-1405. Drying was performed on a lab-scale Labconco freeze dryer.
- a silver stain SDS-PAGE gel was run to analyze for residual CNS and aldolase. None was detectable in either the 10K or 20K PSC batches.
- the ratio of the methyl singlet (3.39 ppm) to the unique PSC resonances ( ⁇ 8.00, 2.50, & 1.55 ppm) is ⁇ 1 ⁇ 3, which would indicate relatively low levels of mPEG-OH impurities.
- the current method discusses PEG coupling with a nucleotide sugar, such as CMP-SA-glycine.
- the nucleotide sugar-PEG product can be separated from the reaction mixture by first desalting the reaction mixture using dialysis. Membrane filtration (reverse osmosis, nanofiltration, etc.) or size exclusion techniques (i.e. polyacrylamide) resin, Sephadex resin, Sepharose resin) can be used next to further purify the mixture. After purification, the reaction mixture containing the nucleotide sugar-PEG product undergoes further purification involving ion exchange chromatography. For most reaction mixtures, a DEAE or Q-resin can be used to remove the unreacted PEG from the product.
- the Q-Sepharose resin is currently preferred, although any polymer with a quaternary amine will also work.
- the Q-Sepharose resin can include ions such as —OH, Cl ⁇ , HCO 3 ⁇ , CO 3 2 ⁇ , PO 4 2 ⁇ , SO 3 ⁇ , Br ⁇ , BO 3 2 ⁇ or the like.
- the product can be loaded and eluted using known methods in the art such as changes in pH or ion strength (NaCl, KCl, etc.).
- the product may be eluted by a step-wise process or a gradient process.
- the nucleotide sugar-PEG product eluted from the column can be collected and desalted again using dialysis, membrane filtration, or size-exclusion techniques. The process would look similar to the figure below but can be varied dependent on processing results and desires.
- CMP-SA-glycine and the p-nitrophenyl-carbamate-mPEG are mixed together to obtain a reaction mixture comprising the product CMP-SA-glycine-PEG, as well as PEG, p-nitrophenyl-carboxylic acid, salts, CMP-SA-glycine, CMP and sialic acid (SA).
- the reaction mixture underwent membrane filtration wherein the membrane has MWCO of 200 (units)-300 kDa.
- CMP-SA-PEG and PEG were retained while p-nitrophenyl-carboxylic acid, salts, CMP-SA-glycine, CMP and SA passed through the membrane.
- the retained products underwent further purification with a Q-column or DEAE column in which PEG flows through the membrane and CMP-SA-PEG is retained and concentrated.
- the CMP-SA-PEG may be freeze-dried or spray dried.
- the CMP-SA-PEG underwent membrane filtration in which salts pass through and while CMP-SA-PEG was retained and concentrated.
- the membrane has a MWCO of 100 MW-3 kDa. This sample may be freeze dried, spray dried, or frozen.
- Any membrane size can be used from reverse osmosis (RO) pore sizes (molecular weight cut-offs) to microfiltration MWCOs depending on the separation desired.
- RO reverse osmosis
- the pH can be varied between 2 and 12, more specifically between 5-10 and more specifically between 7-9 for CMP-SA-PEG.
- the membrane filtration step can be used to remove impurities by such techniques as diafiltration and can be used to concentrate the process streams.
- Cytidine 5′-monophospho N-acetylneuraminic acid (CMP-NAN) is enzymatically synthesized from cytidine 5′-triphosphate (CTP) and N-acetylneuraminic acid (NAN) in the presence of E. coli - expressed CMP-NAN Synthetase (CNS).
- FIG. 1 illustrates the reaction.
- the primary by-product of the synthesis reaction was the formation of a manganese phosphate precipitate. Approximate 30% of the final volume of the reaction consisted of this heavy, brown precipitate. In order to remove this precipitate, the solution was filtered. The cake that formed on the filter was washed with water, and the final solution was then filtered to remove any fine particulates that were not held back by the first filtration.
- the CMP-NAN solution was permeated through a hollow fiber filter.
- the membrane successfully retains proteins from the enzyme preparation while allowing the passage of CMP-NAN.
- Residual salts were then removed from the solution by concentration and diafiltration of the CMP-NAN using a nanofiltration membrane.
- This membrane retains CMP-NAN during concentration and diafiltration while allowing passage of salts that remain in solution, primarily Tris HCl (the buffer from the enzyme preparation) and NaCl (from the synthesis reaction).
- the concentrated CMP-NAN was subsequently decolorized by passing the solution over a pre-packed column of activated carbon.
- the color is likely from manganese oxide formed during the addition of sodium hydroxide to control the pH during synthesis.
- Cytidine 5′-monophospho N-Acetylneuraminic acid (CMP-NAN) was synthesized in a temperature and pH controlled vessel from Cytidine 5′-triphosphate (CTP) and sialic acid (NAN) using CMP-NAN Synthetase (CNS) in the presence of MnCl 2 .
- the CNS is a recombinant protein cloned from N. meningitidis and was expressed in JM109 E. coli cells. The reaction proceeded for approximately 1.5 hours with sufficient agitation to keep the precipitate from settling.
- the resultant CMP-NAN solution was chilled to ⁇ 20° C., clarified by filtration, and transferred into a mobile tank.
- the clarified CMP-NAN solution was concentrated 10 times using a hollow fiber filtration unit and diafiltered with 5 volumes of USP purified water.
- the CMP-NAN was transferred to a mobile tank and the pH was adjusted. This solution was concentrated 10 times and diafiltered with 6 volumes of USP purified water using a nanofiltration system.
- Decolorization was achieved by running the CMP-NAN through an activated carbon column and endotoxin was removed using a CUNO Zeta Plus 60ZA filter.
- the purified CMP-NAN solution was then aseptically filtered (CUNO LifeASSURE 0.2 ⁇ m) and aliquoted into MITOS Sugar Nucleotide Bags and stored at ⁇ 20° C.
- This example describes the preparation and purification of CMP-SA-PEG 30 kDa (compound 3, below).
- the mPEG-p-nitrophenyl carbonate-30 kDa (compound 1, below) was reacted with sodium CMP-SA-Glycine (compound 2, below) in a mixture of THF/Water.
- the crude product was desalted by Tangential Flow Filtration (TFF), purified by Q Sepharose chromatography (IEX), and again desalted by TFF to provide 13.5 g of CMP-SA-PEG-30 kDa product (compound 3, below). Reprocessing of mixed IEX fractions afforded an additional 8.2 grams of 3 (overall yield 72.3%).
- CMP-SA-Glycine (dimethyl amine salt form, 1.35 g, 2.0 mmole) was dissolved in 20 mL H 2 O, and the pH was adjusted to 10.5 with 0.1 N NaOH ( ⁇ 20 mL). The basic solution was degassed under reduced pressure (vacuum 30 min), frozen and lyophilized to dryness. The resulting sodium CMP-SA-Glycine was redissolved in water (80 mL). The pH was measured and found to be 8.5. The pH was adjusted to 7.8 by addition of 0.2 N NaH 2 PO4 ( ⁇ 1.0 mL) and the resulting solution was diluted with THF (200 mL).
- the mPEG-p-nitrophenyl carbonate-30 kDa (30.0 g, 1 mmol) was added to the CMP-SA-Glycine solution in small portions over 3 hr at room temperature. The reaction mixture was stirred at room temperature for 43 hrs. The THF was then removed by rotary evaporation at reduced pressure without heating (water bath temperature at or below 30° C.). The aqueous residue (80 mL) was diluted with water to 600 mL, and adjusted to pH 9.5 with 1.0 N NaOH (about 1.0 mL).
- a Watson-Marlow peristaltic pump (505S) was connected through Tygon tubing (1 ⁇ 4′′ ID) to a Millipore Pellicon-2 Mini Holder equipped with two Millipore 1K Pellicon 2 “MINI” filter (PLAC-V 1K Regenerated Cellulose Membrane; Screen Type: V; 0.1 m2) ( FIG. 1 ).
- the crude aqueous product solution 500 mL, pH 9.5
- the product solution was fed onto the Pellicon Mini filter through Tygon tubing (1 ⁇ 4′′ ID) for diafiltration with a pump speed of 90 rpm (Cross flow rate: 430 mL/min; Flux rate: 20 mL/min; Pressure 13 psi).
- the retentate solution was returned to the bottle containing the bulk chilled product solution (PharMed tubing, 1 ⁇ 4′′ ID) which was maintained at a constant volume (600 mL) by addition of cold DI water (4° C.).
- the permeate solution was collected in 2 L fractions.
- the pH and conductivity values of the retentate/product solution were measured and recorded over time as shown in the table below.
- the pH of the retentate/product solution was maintained above pH 7.5 by the dropwise addition of 1.0 N NaOH, as needed.
- the retentate/product solution was diafiltered until the conductivity dropped below 0.8 mS, and then the retentate/product solution was allowed to concentrate to a volume of 500 mL.
- the concentrated retentate was then purified by anion exchange chromatography as described below.
- the permeate fractions were checked for product break-through by SDS-PAGE as described below:
- TFF permeate fractions 0.5 mL were concentrated to dryness under a stream of N 2 gas, resuspended in 10 ⁇ L water and mixed with 10 ⁇ L Tris-Gly SDS-PAGE sample buffer and loaded onto 4-20% polyacrylamide Tris-Gly SDS-PAGE gels. See Blue Plus2 protein standard was also loaded as a marker. Gels were run at a constant voltage of 125 V for 1 hr 50 min. After electrophoresis, the gels were washed with water (100 mL) for 10 min, and then incubated with a 5% barium chloride aqueous solution for 10 min.
- Iodine solution (0.1 N, 4.0 mL) was added to visualize any mPEG present.
- the staining process was stopped by washing the gels with water.
- the proteins used as a standard were a mix of myosin (250 kDa), phosphorylase (148 kDa), BSA (98 kDa, glutamic dehydrogenase (64 kDa), alcohol dehydrogenase (50 kDa), carbonic anhydrase (36 kDa), lysozyme (22 kDa), aprotinin (6 kDa), and insulin B-chain (4 kDa).
- the gels were visualized and scanned with an HP Scanjet 7400C, and the image of the gel was optimized with the HP Precision Scan Program.
- the Q Sepharose Big Beads (4.0 L) were treated with 1.0 M NaOH (8.0 L), and then with saturated aqueous sodium bicarbonate (8.0 L) to generate the bicarbonate form of the resin.
- the newly generated resin was packed in a 17 ⁇ 18 cm (ID) column which was connected to an HPLC system equipped with a UV (274 nm) and an ELS detector (Evaporation temp: 120° C.; Nebulizer temp: 90° C.; Gas flow rate: 1.85 SLM).
- the TFF retentate from above was slowly loaded on the Q column (60 mL/min.).
- Fraction 1 The first fraction (Fraction 1) of the ion-exchange-purified product was desalted using the TFF procedure described above. Upon reaching a constant conductivity, the retentate/product solution was allowed to concentrate to 400 mL. The final retentate (400 mL) was freeze-dried to yield 13.5 g of white solid.
- Cytidine-monophospho 5′-N-acetylneuraminic acid (CMP-NAN) is enzymatically synthesized from cytidine 5′-triphosphate (CTP) and N-acetylneuraminic acid (NAN) in the presence of E. coli - expressed CMP-NAN synthetase (CNS).
- the reactants (CTP and NAN) are incubated (30° C., pH 8.5-9.5) in a reactor (20 or 40 L Synthesis Reactor, jacketed temperature control is ⁇ 10° C. to 30° C., manual or automatic pH control is 8.5-9.5, and agitated) with CNS and manganese chloride (MnCl 2 ).
- CTP and NAN CNS and manganese chloride
- MnCl 2 manganese chloride
- the reaction proceeds to almost 100% conversion of NAN to the CMP-NAN product in under two hours.
- the solution is chilled to ⁇ 10.0° C. Once chilled the process stream is filtered through a 0.5 ⁇ m membrane bag filter (CUNO Polynet bag filter with CUNO bag filter housing and pressure-fed (nitrogen) filtration).
- the process stream is next permeated through a 10K hollow fiber membrane with a tangential flow filtration (TFF) skid (Amersham Bioscience (formerly AG Technology) 10K MWCO hollow fiber cartridge, temperature control is ⁇ 10° C., and inlet, outlet and permeate pressure indication is 0-60 psig).
- TFF tangential flow filtration
- the 10K membrane successfully retains proteins from the enzyme preparation while allowing for passage of the CMP-NAN. The result is a protein-free CMP-NAN solution.
- Residual salts are then removed from solution by concentration and diafiltration of the CMP-NAN using a nanofiltration membrane with a nanofiltration TFF skid (Millipore Nanomax-50 Helicon RO4 spiral wound membrane cartridge installed on a Millipore Prolab II skid, temperature control is ⁇ 10° C. and pressure indication is 0-600 psig).
- This membrane retains CMP-NAN during concentration and diafiltration while allowing passage of salts that remain in solution from the synthesis reaction and hollow fiber filtration.
- This concentrated CMP-NAN stream is subsequently de-colored by passing the solution over a pre-packed column of activated carbon attached to a Biotage chromatography system (75M activated chromatography column and Biotage skid).
- the activated carbon efficiently removes the color from the solution in one or two passes of the CMP-NAN stream.
- the process is completed by two filtrations.
- the CMP-NAN solution is filtered using a charged media depth filter (CUNO Zeta Plus 60ZA, 1 ft 2 ).
- the solution is sent through a final 0.2 ⁇ m filter that is pre-packaged and sterilized with a bag manifold system (provided by Mitos Technologies) for final filtration and storage.
- the product can be stored as a frozen solution or dried as a white powder for refrigerated storage.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Wood Science & Technology (AREA)
- Biochemistry (AREA)
- Genetics & Genomics (AREA)
- General Health & Medical Sciences (AREA)
- Biotechnology (AREA)
- Molecular Biology (AREA)
- Zoology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Bioinformatics & Cheminformatics (AREA)
- General Engineering & Computer Science (AREA)
- Microbiology (AREA)
- General Chemical & Material Sciences (AREA)
- Saccharide Compounds (AREA)
- Separation Using Semi-Permeable Membranes (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
- The present application is a U.S. national phase application of PCT/US2006/043048 filed Nov. 3, 2006, which claims priority under 35 U.S.C. §119(e) to U.S. Provisional Patent Application No. 60/829,242, filed Oct. 12, 2006, U.S. Provisional Application No. 60/823,538, filed Aug. 25, 2006, U.S. Provisional Application No. 60/746,754, filed May 8, 2006, U.S. Provisional Application No. 60/796,281, filed Apr. 28, 2006, and U.S. Provisional Application No. 60/733,975, filed Nov. 3, 2005, the disclosures of which are incorporated herein by reference for all purposes.
- Increased understanding of the role of carbohydrates as recognition elements on the surface of cells has led to increased interest in the production of carbohydrate molecules of defined structure. For instance, compounds comprising the oligosaccharide moiety, sialyl lactose, have been of interest as neutralizers for enterotoxins from bacteria such as Vibrio cholerae, Escherichia coli, and Salmonella (see, e.g., U.S. Pat. No. 5,330,975). Sialyl lactose has also been investigated for the treatment of arthritis and related autoimmune diseases. In particular, sialyl lactose is thought to inhibit or disrupt the degree of occupancy of the Fc carbohydrate binding site on IgG, and thus prevent the formation of immune complexes (see, U.S. Pat. No. 5,164,374). Recently, sialyl-α(2,3)galactosides, sialyl lactose and sialyl lactosamine have been proposed for the treatment of ulcers, and Phase I clinical trials have begun for the use of the former compound in this capacity. See, Balkonen et al., FEMS Immunology and Medical Microbiology 7:29 (1993) and BioWorld Today, p. 5, Apr. 4, 1995. As another example, compounds comprising the sialyl Lewis ligands, sialyl Lewisx and sialyl Lewisa are present in leukocyte and non-leukocyte cell lines that bind to receptors such as the ELAM-1 and GMP 140 receptors. Polley et al., Proc. Natl. Acad. Sci., USA, 88:6224 (1991) and Phillips et al., Science, 250:1130 (1990), see, also, U.S. Ser. No. 08/063,181.
- Because of interest in making desired carbohydrate structures, glycosyltransferases and their role in enzyme-catalyzed synthesis of carbohydrates are presently being extensively studied. The use of glycosyltransferases for enzymatic synthesis of carbohydrate offers advantages over chemical methods due to the virtually complete stereoselectivity and linkage specificity offered by the enzymes (Ito et al., Pure Appl. Chem., 65:753 (1993) U.S. Pat. Nos. 5,352,670, and 5,374,541). Consequently, glycosyltransferases are increasingly used as enzymatic catalysts in synthesis of a number of carbohydrates used for therapeutic and other purposes.
- Carbohydrate compounds produced by enzymatic synthesis or by other methods are often obtained in the form of complex mixtures that include not only the desired compound but also contaminants such as unreacted sugars, salts, pyruvate, phosphate, PEP, nucleosides, nucleotides, and proteins, among others. The presence of these contaminants is undesirable for many applications for which the carbohydrate compounds are useful. Previously used methods for purifying oligosaccharides, such as chromatography, i.e., ion exchange and size exclusion chromatography, have several disadvantages. For example, chromatographic purification methods are not amenable to large-scale purifications, thus precluding their use for commercial production of saccharides. Moreover, chromatographic purification methods are expensive. Therefore, a need exists for purification methods that are faster, more efficient, and less expensive than previously used methods. The present invention fulfills this and other needs.
- The present invention provides methods of purifying a carbohydrate compound from a feed solution containing a contaminant. The methods involve contacting the feed solution with a nanofiltration or reverse osmosis membrane under conditions such that the membrane retains the desired carbohydrate compound while a majority of the contaminant passes through the membrane. The invention provides methods for purifying carbohydrate compounds such as sialyl lactosides, sialic acid, lacto-N-neotetraose (LNnT) and GlcNAcβ1,3Galβ1,4Glc (LNT-2), NeuAcα(2→3)Galβ(1→4)(Fucα1→3)Glc(R1)β1-OR2, wherein R1 is OH or NAc; R2 is a hydrogen, an alkoxy, a saccharide, an oligosaccharide or an aglycon group having at least one carbon atom; and Galα(1→3)Galβ(1→4)Glc(R1)β-O—R3, wherein R1 is OH or NAc; R3 is —(CH2)n—COX, with X=OH, OR4, —NHNH2, R4 being a hydrogen, a saccharide, an oligosaccharide or an aglycon group having at least one carbon atom, and n=an integer from 2 to 18.
- Also provided are methods for purifying carbohydrate compounds having a formula NeuAcα(2→3)Galβ(1→4)GlcN(R1)β-OR2, NeuAcα(2→3)Galβ(1→4)GlcN(R1)β(1→3)Galβ-OR2, NeuAcα(2→3)Galβ(1→4) (Fucα1→3)GlcN(R1)β-OR2, or NeuAcα(2→3)Galβ(1→4) (Fucα1→3)GlcN(R1)β(1→3)Galβ-OR2, wherein R1 is alkyl or acyl from 1-18 carbons, 5,6,7,8-tetrahydro-2-naphthamido; benzamido; 2-naphthamido; 4-aminobenzamido; or 4-nitrobenzamido, and R2 is a hydrogen, a saccharide, an oligosaccharide or an aglycon group having at least one carbon atom.
- In another embodiment, the invention provides methods of purifying a carbohydrate compound from a feed solution comprising a reaction mixture used to synthesize the carbohydrate compound. The synthesis can be enzymatic or chemical, or a combination thereof. The methods involve removing any proteins present in the feed solution by contacting the feed solution with an ultrafiltration membrane so that proteins are retained the membrane while the carbohydrate compound passes through the membrane as a permeate. The permeate from the ultrafiltration step is then contacted with a nanofiltration or reverse osmosis membrane under conditions such that the nanofiltration or reverse osmosis membrane retains the carbohydrate compound while a majority of an undesired contaminant passes through the membrane.
- Another embodiment of the invention provides methods for purifying nucleotides, nucleosides, and nucleotide sugars by contacting a feed solution containing the nucleotide or related compound with a nanofiltration or reverse osmosis membrane under conditions such that the membrane retains the nucleotide or related compound while a majority of the contaminant passes through the membrane.
- The present invention also provides methods for removing one or more contaminants from a solution that contains a carbohydrate of interest. The methods involve contacting the solution with a first side of a semipermeable membrane having rejection coefficients so as to retain the carbohydrate while allowing the contaminant to pass through the membrane. The membrane is selected from the group consisting of an ultrafiltration membrane, a nanofiltration membrane, and a reverse osmosis membrane, depending on the size and charge of the carbohydrate of interest relative to those of the contaminants. The membrane separates a feed solution containing a carbohydrate into a retentate portion and a permeate portion. If the rejection coefficient of the membrane is greater for the carbohydrate than for the contaminant, the retentate portion will have a lower concentration of the contaminant relative to the contaminant concentration in the feed solution, and generally also a higher ratio of the carbohydrate to the undesired contaminant. Conversely, a membrane having a rejection coefficient for the carbohydrate that is lesser than that for the contaminant will effect a separation wherein the concentration of the contaminant is lower in the permeate than in the feed solution, and the permeate will have a higher ratio of carbohydrate to contaminant than the feed solution. If desired, the fraction containing the carbohydrate can be recycled through the membrane system for further purification.
- Examples of contaminants that can be removed from solutions containing the compound of interest using the methods of the invention include, but are not limited to, unreacted sugars, inorganic ions, pyruvate, phosphate, phosphoenolpyruvate, and proteins.
-
FIG. 1 is a diagram of an exemplary purification of a nucleotide sugar. -
FIG. 2 is a chromatogram recorded for an exemplary purification of CMP-SA-PEG-30 kDa using Q Sepharose chromatography as described in Example 6. CMP-SA-PEG-30 kDa was collected in two fractions.Fraction 1 contained pure product andFraction 2 contained residual CMP-SA-Gly reagent and was reprocessed. -
FIG. 3 is a chromatogram recorded for an exemplary separation of CMP-SA-PEG 30 kDa from CMP-SA-Glycine using Q-Sepharose chromatography, showing a typical baseline separation. -
FIG. 4 is a diagram outlining an exemplary process for the preparation of an exemplary GSC (CMP-5′-Glycyl-Sialic Acid). -
FIG. 5 is a diagram outlining an exemplary process for the preparation of an exemplary PSC (CMP-SA-PEG). -
FIG. 6 is a diagram outlining an exemplary process for the purification of a glycosyltransferase. -
FIG. 7 is a table of exemplary sialyltransferases. - The term “sialic acid” refers to any member of a family of nine-carbon carboxylated sugars. The most common member of the sialic acid family is N-acetyl-neuraminic acid (2-keto-5-acetamido-3,5-dideoxy-D-glycero-D-galactononulopyranos-1-onic acid (often abbreviated as Neu5Ac, NeuAc, or NANA). A second member of the family is N-glycolyl-neuraminic acid (Neu5Gc or NeuGc), in which the N-acetyl group of NeuAc is hydroxylated. A third sialic acid family member is 2-keto-3-deoxy-nonulosonic acid (KDN) (Nadano et al. (1986) J. Biol. Chem. 261: 11550-11557; Kanamori et al., J. Biol. Chem. 265: 21811-21819 (1990)). Also included are 9-substituted sialic acids such as a 9-O—C1-C6 acyl-Neu5Ac like 9-O-lactyl-Neu5Ac or 9-O-acetyl-Neu5Ac, 9-deoxy-9-fluoro-Neu5Ac and 9-azido-9-deoxy-Neu5Ac. For review of the sialic acid family, see, e.g., Varki, Glycobiology 2: 25-40 (1992); Sialic Acids: Chemistry, Metabolism and Function, R. Schauer, Ed. (Springer-Verlag, New York (1992)). The synthesis and use of sialic acid compounds in a sialylation procedure is disclosed in international application WO 92/16640, published Oct. 1, 1992.
- As used herein, the term “modified sugar,” refers to a naturally- or non-naturally-occurring carbohydrate. The modified sugar is preferably selected from a number of enzyme substrates including, but not limited to sugar nucleotides (mono-, di-, and tri-phosphates), activated sugars (e.g., glycosyl halides, glycosyl mesylates) and sugars that are neither activated nor nucleotides. The “modified sugar” is covalently functionalized with a “modifying group.” Useful modifying groups include, but are not limited to, water-soluble polymers, targeting moieties therapeutic moieties, diagnostic moieties, radioactive moieties, cytotoxic moieties, biomolecules and the like. The modifying group is preferably not a naturally occurring, or an unmodified carbohydrate. The locus of functionalization with the modifying group is preferably selected such that it does not prevent the “modified sugar” from being added enzymatically to a peptide.
- The term “water-soluble” refers to moieties that have some detectable degree of solubility in water. Methods to detect and/or quantify water solubility are well known in the art. Exemplary water-soluble polymers include peptides, saccharides, poly(ethers), poly(amines), poly(carboxylic acids) and the like. Peptides can have mixed sequences of be composed of a single amino acid, e.g., poly(lysine). An exemplary polysaccharide is poly(sialic acid). An exemplary poly(ether) is poly(ethylene glycol), e.g., m-PEG. Poly(ethylene imine) is an exemplary polyamine, and poly(acrylic) acid is a representative poly(carboxylic acid).
- The polymer backbone of the water-soluble polymer can be poly(ethylene glycol) (i.e. PEG). However, it should be understood that other related polymers are also suitable for use in the practice of this invention and that the use of the term PEG or poly(ethylene glycol) is intended to be inclusive and not exclusive in this respect. The term PEG includes poly(ethylene glycol) in any of its forms, including alkoxy PEG, difunctional PEG, multiarmed PEG, forked PEG, branched PEG, pendent PEG (i.e. PEG or related polymers having one or more functional groups pendent to the polymer backbone), or PEG with degradable linkages therein.
- The polymer backbone can be linear or branched. Branched polymer backbones are generally known in the art. Typically, a branched polymer has a central branch core moiety and a plurality of linear polymer chains linked to the central branch core. PEG is commonly used in branched forms that can be prepared by addition of ethylene oxide to various polyols, such as glycerol, pentaerythritol and sorbitol. The central branch moiety can also be derived from several amino acids, such as lysine. The branched poly(ethylene glycol) can be represented in general form as R(-PEG-OH)m in which R represents the core moiety, such as glycerol or pentaerythritol, and m represents the number of arms. Multi-armed PEG molecules, such as those described in U.S. Pat. No. 5,932,462, which is incorporated by reference herein in its entirety, can also be used as the polymer backbone.
- Many other polymers are also suitable for the invention. Polymer backbones that are non-peptidic and water-soluble, with from 2 to about 300 termini, are particularly useful in the invention. Examples of suitable polymers include, but are not limited to, other poly(alkylene glycols), such as poly(propylene glycol) (“PPG”), copolymers of ethylene glycol and propylene glycol and the like, poly(oxyethylated polyol), poly(olefinic alcohol), poly(vinylpyrrolidone), poly(hydroxypropylmethacrylamide), poly(α-hydroxy acid), poly(vinyl alcohol), polyphosphazene, polyoxazoline, poly(N-acryloylmorpholine), such as described in U.S. Pat. No. 5,629,384, which is incorporated by reference herein in its entirety, and copolymers, terpolymers, and mixtures thereof. Although the molecular weight of each chain of the polymer backbone can vary, it is typically in the range of from about 100 Da to about 100,000 Da, often from about 6,000 Da to about 80,000 Da.
- The term “targeting moiety,” as used herein, refers to species that will selectively localize in a particular tissue or region of the body. The localization is mediated by specific recognition of molecular determinants, molecular size of the targeting agent or conjugate, ionic interactions, hydrophobic interactions and the like. Other mechanisms of targeting an agent to a particular tissue or region are known to those of skill in the art. Exemplary targeting moieties include antibodies, antibody fragments, transferrin, HS-glycoprotein, coagulation factors, serum proteins, β-glycoprotein, G-CSF, GM-CSF, M-CSF, EPO and the like.
- As used herein, “therapeutic moiety” means any agent useful for therapy including, but not limited to, antibiotics, anti-inflammatory agents, anti-tumor drugs, cytotoxins, and radioactive agents. “Therapeutic moiety” includes prodrugs of bioactive agents, constructs in which more than one therapeutic moiety is bound to a carrier, e.g, multivalent agents. Therapeutic moiety also includes proteins and constructs that include proteins. Exemplary proteins include, but are not limited to, Erythropoietin (EPO), Granulocyte Colony Stimulating Factor (GCSF), Granulocyte Macrophage Colony Stimulating Factor (GMCSF), Interferon (e.g., Interferon-α, -β, -γ), Interleukin (e.g., Interleukin II), serum proteins (e.g., Factors VII, VIa, VIII, IX, and X), Human Chorionic Gonadotropin (HCG), Follicle Stimulating Hormone (FSH) and Lutenizing Hormone (LH) and antibody fusion proteins (e.g. Tumor Necrosis Factor Receptor ((TNFR)/Fc domain fusion protein)).
- As used herein, “anti-tumor drug” means any agent useful to combat cancer including, but not limited to, cytotoxins and agents such as antimetabolites, alkylating agents, anthracyclines, antibiotics, antimitotic agents, procarbazine, hydroxyurea, asparaginase, corticosteroids, interferons and radioactive agents. Also encompassed within the scope of the term “anti-tumor drug,” are conjugates of peptides with anti-tumor activity, e.g. TNF-α. Conjugates include, but are not limited to those formed between a therapeutic protein and a glycoprotein of the invention. A representative conjugate is that formed between PSGL-1 and TNF-α.
- As used herein, “a cytotoxin or cytotoxic agent” means any agent that is detrimental to cells. Examples include taxol, cytochalasin B, gramicidin D, ethidium bromide, emetine, mitomycin, etoposide, tenoposide, vincristine, vinblastine, colchicin, doxorubicin, daunorubicin, dihydroxy anthracinedione, mitoxantrone, mithramycin, actinomycin D, 1-dehydrotestosterone, glucocorticoids, procaine, tetracaine, lidocaine, propranolol, and puromycin and analogs or homologs thereof. Other toxins include, for example, ricin, CC-1065 and analogues, the duocarmycins. Still other toxins include diptheria toxin, and snake venom (e.g., cobra venom).
- As used herein, “a radioactive agent” includes any radioisotope that is effective in diagnosing or destroying a tumor. Examples include, but are not limited to, indium-111, cobalt-60. Additionally, naturally occurring radioactive elements such as uranium, radium, and thorium, which typically represent mixtures of radioisotopes, are suitable examples of a radioactive agent. The metal ions are typically chelated with an organic chelating moiety.
- Many useful chelating groups, crown ethers, cryptands and the like are known in the art and can be incorporated into the compounds of the invention (e.g., EDTA, DTPA, DOTA, NTA, HDTA, etc. and their phosphonate analogs such as DTPP, EDTP, HDTP, NTP, etc). See, for example, Pitt et al., “The Design of Chelating Agents for the Treatment of Iron Overload,” In, I
NORGANIC CHEMISTRY IN BIOLOGY AND MEDICINE ; Martell, Ed.; American Chemical Society, Washington, D.C., 1980, pp. 279-312; Lindoy, THE CHEMISTRY OF MACROCYCLIC LIGAND COMPLEXES ; Cambridge University Press, Cambridge, 1989; Dugas, BIOORGANIC CHEMISTRY ; Springer-Verlag, New York, 1989, and references contained therein. - Additionally, a manifold of routes allowing the attachment of chelating agents, crown ethers and cyclodextrins to other molecules is available to those of skill in the art. See, for example, Meares et al., “Properties of In Vivo Chelate-Tagged Proteins and Polypeptides.” In, M
ODIFICATION OF PROTEINS : FOOD , NUTRITIONAL, AND PHARMACOLOGICAL ASPECTS ;” Feeney, et al., Eds., American Chemical Society, Washington, D.C., 1982, pp. 370-387; Kasina et al., Bioconjugate Chem., 9: 108-117 (1998); Song et al., Bioconjugate Chem., 8: 249-255 (1997). - A compound is “substantially purified” from an undesired component in a solution if the concentration of the undesired component after purification is no greater than about 40% of the concentration of the component prior to purification. Preferably, the post-purification concentration of the undesired component will be less than about 20% by weight, and more preferably less than about 10%, and still more preferably less than about 5% of the pre-purification concentration.
- The term “pharmaceutically pure,” as used herein, refers to a compound that is sufficiently purified from undesired contaminants that the compound is suitable for administration as a pharmaceutical agent. Preferably, the compound is purified such that the undesired contaminant is present after purification in an amount that is about 5% by weight or less of the pre-purification concentration of the contaminant in the feed solution. More preferably, the post-purification concentration of the contaminant is about 1% or less of the pre-purification contaminant concentration, and most preferably about 0.5% or less of the pre-purification concentration of contaminant.
- A “feed solution” refers to any solution that contains a compound to be purified. For example, a reaction mixture used to synthesize an oligosaccharide can be used as a feed solution from which the desired reaction product is purified using the methods of the invention.
- Where substituent groups are specified by their conventional chemical formulae, written from left to right, they equally encompass the chemically identical substituents, which would result from writing the structure from right to left, e.g., —CH2O— is intended to also recite —OCH2—.
- The term “alkyl,” by itself or as part of another substituent means, unless otherwise stated, a straight or branched chain, or cyclic hydrocarbon radical, or combination thereof, which may be fully saturated, mono- or polyunsaturated and can include di- and multivalent radicals, having the number of carbon atoms designated (i.e. C1-C10 means one to ten carbons). Examples of saturated hydrocarbon radicals include, but are not limited to, groups such as methyl, ethyl, n-propyl, isopropyl, n-butyl, t-butyl, isobutyl, sec-butyl, cyclohexyl, (cyclohexyl)methyl, cyclopropylmethyl, homologs and isomers of, for example, n-pentyl, n-hexyl, n-heptyl, n-octyl, and the like. An unsaturated alkyl group is one having one or more double bonds or triple bonds. Examples of unsaturated alkyl groups include, but are not limited to, vinyl, 2-propenyl, crotyl, 2-isopentenyl, 2-(butadienyl), 2,4-pentadienyl, 3-(1,4-pentadienyl), ethynyl, 1- and 3-propynyl, 3-butynyl, and the higher homologs and isomers. The term “alkyl,” unless otherwise noted, is also meant to include those derivatives of alkyl defined in more detail below, such as “heteroalkyl.” Alkyl groups that are limited to hydrocarbon groups are termed “homoalkyl”.
- The term “alkylene” by itself or as part of another substituent means a divalent radical derived from an alkane, as exemplified, but not limited, by —CH2CH2CH2CH2—, and further includes those groups described below as “heteroalkylene.” Typically, an alkyl (or alkylene) group will have from 1 to 24 carbon atoms, with those groups having 10 or fewer carbon atoms being preferred in the present invention. A “lower alkyl” or “lower alkylene” is a shorter chain alkyl or alkylene group, generally having eight or fewer carbon atoms.
- The terms “alkoxy,” “alkylamino” and “alkylthio” (or thioalkoxy) are used in their conventional sense, and refer to those alkyl groups attached to the remainder of the molecule via an oxygen atom, an amino group, or a sulfur atom, respectively.
- The term “heteroalkyl,” by itself or in combination with another term, means, unless otherwise stated, a stable straight or branched chain, or cyclic hydrocarbon radical, or combinations thereof, consisting of the stated number of carbon atoms and at least one heteroatom selected from the group consisting of O, N, Si and S, and wherein the nitrogen and sulfur atoms may optionally be oxidized and the nitrogen heteroatom may optionally be quaternized. The heteroatom(s) O, N and S and Si may be placed at any interior position of the heteroalkyl group or at the position at which the alkyl group is attached to the remainder of the molecule. Examples include, but are not limited to, —CH2—CH2—O—CH3, —CH2—CH2—NH—CH3, —CH2—CH2—N(CH3)—CH3, —CH2—S—CH2—CH3, —CH2—CH2, —S(O)—CH3, —CH2—CH2—S(O)2—CH3, —CH═CH—O—CH3, —Si(CH3)3, —CH2—CH═N—OCH3, and —CH═CH—N(CH3)—CH3. Up to two heteroatoms may be consecutive, such as, for example, —CH2—NH—OCH3 and —CH2—O—Si(CH3)3. Similarly, the term “heteroalkylene” by itself or as part of another substituent means a divalent radical derived from heteroalkyl, as exemplified, but not limited by, —CH2—CH2—S—CH2—CH2— and —CH2—S—CH2—CH2—NH—CH2—. For heteroalkylene groups, heteroatoms can also occupy either or both of the chain termini (e.g., alkyleneoxy, alkylenedioxy, alkyleneamino, alkylenediamino, and the like). Still further, for alkylene and heteroalkylene linking groups, no orientation of the linking group is implied by the direction in which the formula of the linking group is written. For example, the formula —C(O)2R′— represents both —C(O)2R′— and —R′C(O)2—.
- The terms “cycloalkyl” and “heterocycloalkyl”, by themselves or in combination with other terms, represent, unless otherwise stated, cyclic versions of “alkyl” and “heteroalkyl”, respectively. Additionally, for heterocycloalkyl, a heteroatom can occupy the position at which the heterocycle is attached to the remainder of the molecule. Examples of cycloalkyl include, but are not limited to, cyclopentyl, cyclohexyl, 1-cyclohexenyl, 3-cyclohexenyl, cycloheptyl, and the like. Examples of heterocycloalkyl include, but are not limited to, 1-(1,2,5,6-tetrahydropyridyl), 1-piperidinyl, 2-piperidinyl, 3-piperidinyl, 4-morpholinyl, 3-morpholinyl, tetrahydrofuran-2-yl, tetrahydrofuran-3-yl, tetrahydrothien-2-yl, tetrahydrothien-3-yl, 1-piperazinyl, 2-piperazinyl, and the like.
- The terms “halo” or “halogen,” by themselves or as part of another substituent, mean, unless otherwise stated, a fluorine, chlorine, bromine, or iodine atom. Additionally, terms such as “haloalkyl,” are meant to include monohaloalkyl and polyhaloalkyl. For example, the term “halo(C1-C4)alkyl” is mean to include, but not be limited to, trifluoromethyl, 2,2,2-trifluoroethyl, 4-chlorobutyl, 3-bromopropyl, and the like.
- The term “aryl” means, unless otherwise stated, a polyunsaturated, aromatic, substituent that can be a single ring or multiple rings (preferably from 1 to 3 rings), which are fused together or linked covalently. The term “heteroaryl” refers to aryl groups (or rings) that contain from one to four heteroatoms selected from N, O, and S, wherein the nitrogen and sulfur atoms are optionally oxidized, and the nitrogen atom(s) are optionally quaternized. A heteroaryl group can be attached to the remainder of the molecule through a heteroatom. Non-limiting examples of aryl and heteroaryl groups include phenyl, 1-naphthyl, 2-naphthyl, 4-biphenyl, 1-pyrrolyl, 2-pyrrolyl, 3-pyrrolyl, 3-pyrazolyl, 2-imidazolyl, 4-imidazolyl, pyrazinyl, 2-oxazolyl, 4-oxazolyl, 2-phenyl-4-oxazolyl, 5-oxazolyl, 3-isoxazolyl, 4-isoxazolyl, 5-isoxazolyl, 2-thiazolyl, 4-thiazolyl, 5-thiazolyl, 2-furyl, 3-furyl, 2-thienyl, 3-thienyl, 2-pyridyl, 3-pyridyl, 4-pyridyl, 2-pyrimidyl, 4-pyrimidyl, 5-benzothiazolyl, purinyl, 2-benzimidazolyl, 5-indolyl, 1-isoquinolyl, 5-isoquinolyl, 2-quinoxalinyl, 5-quinoxalinyl, 3-quinolyl, tetrazolyl, benzo[b]furanyl, benzo[b]thienyl, 2,3-dihydrobenzo[1,4]dioxin-6-yl, benzo[1,3]dioxol-5-yl and 6-quinolyl. Substituents for each of the above noted aryl and heteroaryl ring systems are selected from the group of acceptable substituents described below.
- For brevity, the term “aryl” when used in combination with other terms (e.g., aryloxy, arylthioxy, arylalkyl) includes both aryl and heteroaryl rings as defined above. Thus, the term “arylalkyl” is meant to include those radicals in which an aryl group is attached to an alkyl group (e.g., benzyl, phenethyl, pyridylmethyl and the like) including those alkyl groups in which a carbon atom (e.g., a methylene group) has been replaced by, for example, an oxygen atom (e.g., phenoxymethyl, 2-pyridyloxymethyl, 3-(1-naphthyloxy)propyl, and the like).
- Each of the above terms (e.g., “alkyl,” “heteroalkyl,” “aryl” and “heteroaryl”) is meant to include both substituted and unsubstituted forms of the indicated radical. Preferred substituents for each type of radical are provided below.
- Substituents for the alkyl and heteroalkyl radicals (including those groups often referred to as alkylene, alkenyl, heteroalkylene, heteroalkenyl, alkynyl, cycloalkyl, heterocycloalkyl, cycloalkenyl, and heterocycloalkenyl) are generically referred to as “alkyl group substituents,” and they can be one or more of a variety of groups selected from, but not limited to: —OR′, ═O, ═NR′, ═N—OR′, —NR′R″, —SR′, -halogen, —SiR′R″R′″, —OC(O)R′, —C(O)R′, —CO2R′, —CONR′R″, —OC(O)NR′R″, —NR″C(O)R′, —NR′—C(O)NR″R′″, —NR″C(O)2R′, —NR—C(NR′R″R′″)═NR″″, —NR—C(NR′R″)═NR′″, —S(O)R′, —S(O)2R′, —S(O)2NR′R″, —NRSO2R′, —CN and —NO2 in a number ranging from zero to (2m′+1), where m′ is the total number of carbon atoms in such radical. R′, R″, R′″ and R″″ each preferably independently refer to hydrogen, substituted or unsubstituted heteroalkyl, substituted or unsubstituted aryl, e.g., aryl substituted with 1-3 halogens, substituted or unsubstituted alkyl, alkoxy or thioalkoxy groups, or arylalkyl groups. When a compound of the invention includes more than one R group, for example, each of the R groups is independently selected as are each R′, R″, R′″ and R″″ groups when more than one of these groups is present. When R′ and R″ are attached to the same nitrogen atom, they can be combined with the nitrogen atom to form a 5-, 6-, or 7-membered ring. For example, —NR′R″ is meant to include, but not be limited to, 1-pyrrolidinyl and 4-morpholinyl. From the above discussion of substituents, one of skill in the art will understand that the term “alkyl” is meant to include groups including carbon atoms bound to groups other than hydrogen groups, such as haloalkyl (e.g., —CF3 and —CH2CF3) and acyl (e.g., —C(O)CH3, —C(O)CF3, —C(O)CH2OCH3, and the like).
- Similar to the substituents described for the alkyl radical, substituents for the aryl and heteroaryl groups are generically referred to as “aryl group substituents.” The substituents are selected from, for example: halogen, —OR′, ═O, ═NR′, ═N—OR′, —NR′R″, —SR′, -halogen, —SiR′R″R′″, —OC(O)R′, —C(O)R′, —CO2R′, —CONR′R″, —OC(O)NR′R″, —NR″C(O)R′, —NR′—C(O)NR″R′″, —NR″C(O)2R′, —NR—C(NR′R″R′″)═NR″″, —NR—C(NR′R″)═NR′″, —S(O)R′, —S(O)2R′, —S(O)2NR′R″, —NRSO2R′, —CN and —NO2, —R′, —N3, —CH(Ph)2, fluoro(C1-C4)alkoxy, and fluoro(C1-C4)alkyl, in a number ranging from zero to the total number of open valences on the aromatic ring system; and where R′, R″, R′″ and R″″ are preferably independently selected from hydrogen, substituted or unsubstituted alkyl, substituted or unsubstituted heteroalkyl, substituted or unsubstituted aryl and substituted or unsubstituted heteroaryl. When a compound of the invention includes more than one R group, for example, each of the R groups is independently selected as are each R′, R″, R′″ and R″″ groups when more than one of these groups is present. In the schemes that follow, the symbol X represents “R” as described above.
- Two of the substituents on adjacent atoms of the aryl or heteroaryl ring may optionally be replaced with a substituent of the formula -T-C(O)—(CRR′)q—U—, wherein T and U are independently —NR—, —O—, —CRR′— or a single bond, and q is an integer of from 0 to 3. Alternatively, two of the substituents on adjacent atoms of the aryl or heteroaryl ring may optionally be replaced with a substituent of the formula -A-(CH2)r—B—, wherein A and B are independently —CRR′—, —O—, —NR—, —S—, —S(O)—, —S(O)2—, —S(O)2NR′— or a single bond, and r is an integer of from 1 to 4. One of the single bonds of the new ring so formed may optionally be replaced with a double bond. Alternatively, two of the substituents on adjacent atoms of the aryl or heteroaryl ring may optionally be replaced with a substituent of the formula —(CRR′)s—X—(CR″R′″)d—, where s and d are independently integers of from 0 to 3, and X is —O—, —NR′—, —S—, —S(O)—, —S(O)2—, or —S(O)2NR′—. The substituents R, R′, R″ and R′″ are preferably independently selected from hydrogen or substituted or unsubstituted (C1-C6)alkyl.
- As used herein, the term “heteroatom” is meant to include oxygen (O), nitrogen (N), sulfur (S) and silicon (Si).
- The present invention provides methods for rapidly and efficiently purifying specific carbohydrate and oligosaccharide structures to a high degree of purity using semipermeable membranes such as reverse osmosis and/or nanofiltration membranes. The methods are particularly useful for separating desired oligosaccharide compounds from reactants and other contaminants that remain in a reaction mixture after synthesis or breakdown of the oligosaccharides. For example, the invention provides methods for separating oligosaccharides from enzymes and/or other components of reaction mixtures used for enzymatic synthesis or enzymatic degradation of oligosaccharides, nucleotide sugars, glycolipids, liposaccharides, nucleotides, nucleosides, and other saccharide-containing compounds. Also provided are methods for removing salts, sugars and other components from feed solutions using ultrafiltration, nanofiltration or reverse osmosis. Using these techniques, the saccharides (e.g., sialyl lactose, SLex, and many others) can be produced at up to essentially 100% purity. Moreover, the purification methods of the invention are more efficient, rapid, and amenable to large-scale purifications than previously known carbohydrate purification methods.
- Often, a desired purification can be effected in a single step; additional purification steps such as crystallization and the like are generally not required. Accordingly, the invention provides single-step methods for purifying saccharide-containing compounds.
- To purify saccharides according to the methods of the invention, a membrane is selected that is appropriate for separating the desired carbohydrate from the undesired components (contaminants) of the solution from which the carbohydrate is to be purified. The goal in selecting a membrane is to optimize for a particular application the molecular weight cutoff (MWCO), membrane composition, permeability, and rejection characteristics, that is, the membrane's total capacity to retain specific molecules while allowing other species, e.g., salts and other, generally smaller or opposite charged molecules, to pass through. The percent retention of a component i (Ri) is given by the formula Ri=(1−Cip/Cir)×100%, wherein Cip is the concentration of component i in the permeate and Cir is the concentration of component i in the retentate, both expressed in weight percent. The percent retention of a component is also called the retention characteristic or the membrane rejection coefficient.
- In and exemplary embodiment, a membrane is chosen that has a high rejection ratio for the saccharide of interest relative to the rejection ratio for compounds from which separation is desired. If a membrane has a high rejection ratio for a first compound relative to a second compound, the concentration of the first compound in the permeate solution which passes through the membrane is decreased relative to that of the second compound. Conversely, the concentration of the first compound increases relative to the concentration of the second compound in the retentate. If a membrane does not reject a compound, the concentration of the compound in both the permeate and the reject portions will remain essentially the same as in the feed solution. It is also possible for a membrane to have a negative rejection rate for a compound if the compound's concentration in the permeate becomes greater than the compound's concentration in the feed solution. A general review of membrane technology is found in “Membranes and Membrane Separation Processes,” in Ullmann's Encyclopedia of Industrial Chemistry (VCH, 1990); see also, Noble and Stem, Membrane Separations Technology: Principles and Applications (Elsevier, 1995).
- As a starting point, one will generally choose a membrane having a molecular weight cut-off (MWCO, which is often related to membrane pore size) that is expected to retain the desired compounds while allowing an undesired compound present in the feed stream to pass through the membrane. The desired MWCO is generally less than the molecular weight of the compound being purified, and is typically greater than the molecular weight of the undesired contaminant that is to be removed from the solution containing the compound being purified. For example, to purify a compound having a molecular weight of 200 Da, one would choose a membrane that has a MWCO of less than about 200 Da. A membrane with a MWCO of 100 Da, for example, would also be a suitable candidate. The membranes that find use in the present invention are classified in part on the basis of their MWCO as ultrafiltration (UF) membranes, nanofiltration (NF) membranes, or reverse osmosis (RO) membranes, depending on the desired separation. For purposes of this invention, UF, NF, and RO membranes are classified as defined in the Pure Water Handbook, Osmonics, Inc. (Minnetonka Minn.). RO membranes typically have a nominal MWCO of less than about 200 Da and reject most ions, NF membranes generally have a nominal MWCO of between about 150 Da and about 5 kDa, and UF membranes generally have a nominal MWCO of between about 1 kDa and about 300 kDa (these MWCO ranges assume a saccharide-like molecule).
- A second parameter that is considered in choosing an appropriate membrane for a particular separation is the polymer type of the membrane. Exemplary membranes of use in the invention are made of conventional membrane material whether inorganic, organic, or mixed inorganic and organic. Typical inorganic materials include glasses, ceramics, cermets, metals and the like. Ceramic membranes, which are preferred for the UF zone, may be made, for example, as described in U.S. Pat. No. 4,692,354 to Asaeda et al, U.S. Pat. No. 4,562,021 to Alary et al., and others. The organic materials which are preferred for the NF and RO applications, are typically polymers, whether isotropic, or anisotropic with a thin layer or “skin” on either the bore side or the shell side of the fibers. Preferred materials for fibers are polyamides, polybenzamides, polysulfones (including sulfonated polysulfone and sulfonated polyether sulfone, among others), polystyrenes, including styrene-containing copolymers such as acrylo-nitrile-styrene, butadiene-styrene and styrene-vinylbenzylhalide copolymers, polycarbonates, cellulosic polymers including cellulose acetate, polypropylene, poly(vinyl chloride), poly(ethylene terephthalate), polyvinyl alcohol, fluorocarbons, and the like, such as those disclosed in U.S. Pat. Nos. 4,230,463, 4,806,244, and 4,259,183. The NF and RO membranes often consist of a porous support substrate in addition to the polymeric discrimination layer.
- Of particular importance in selecting a suitable membrane composition is the membrane surface charge. Within the required MWCO range, a membrane is selected that has a surface charge that is appropriate for the ionic charge of the carbohydrate and that of the contaminants. While MWCO for a particular membrane is generally invariable, changing the pH of the feed solution can affect separation properties of a membrane by altering the membrane surface charge. For example, a membrane that has a net negative surface charge at neutral pH can be adjusted to have a net neutral charge simply by lowering the pH of the solution. An additional effect of adjusting solution pH is to modulate the ionic charge on the contaminants and on the carbohydrate of interest. Therefore, by choosing a suitable membrane polymer type and pH, one can obtain a system in which both the contaminant and the membrane are neutral, facilitating pass-through of the contaminant. If, for instance, a contaminant is negatively charged at neutral pH, it is often desirable to lower the pH of the feed solution to protonate the contaminant. For example, removal of phosphate is facilitated by lowering the pH of the solution to about 3, which protonates the phosphate anion, allowing passage through a membrane. For purification of an anionic carbohydrate, the pH will generally between about
pH 1 and aboutpH 7. Conversely, if contaminant has a positive surface charge, the pH of the feed solution can be adjusted to between aboutpH 7 and aboutpH 14. For example, increasing the pH of a solution containing a contaminant having an amino group (—NH3 +) will make the amino group neutral, thus facilitating its passage through the membrane. Thus, one aspect of the invention involves modulating a separation by adjusting the pH of a solution in contact with the membrane; this can change the ionic charge of a contaminant and can also affect the surface charge of the membrane, thus facilitating purification if the desired carbohydrate. Of course, the manufacturer's instructions must be followed as to acceptable pH range for a particular membrane to avoid damage to the membrane. - For some applications, a mixture is first subjected to nanofiltration or reverse osmosis at one pH, after which the retentate containing the saccharide of interest is adjusted to a different pH and subjected to an additional round of membrane purification. For example, filtration of a reaction mixture used to synthesize sialyl lactose through an Osmonics MX07 membrane (a nanofiltration membrane having a MWCO of about 500 Da) at pH 3.0 will retain the sialyl lactose and remove most phosphate, pyruvate, salt and manganese from the solution, while also removing some of the GlcNAc, lactose, and sialic acid. Further recirculation through the MXO7 membrane after adjusting the pH of the retentate to 7.4 will remove most of the remaining phosphate, all of the pyruvate, all of the lactose, some of the sialic acid, and substantial amounts of the remaining manganese.
- If a saccharide is to be purified from a mixture that contains proteins, such as enzymes used to synthesize a desired oligosaccharide or nucleotide sugar, it is often desirable to remove the proteins as a first step of the purification procedure. For a saccharide that is smaller than the proteins, this separation is accomplished by choosing a membrane that has an MWCO which is less than the molecular mass of the protein or other macromolecule to be removed from the solution, but is greater than the molecular mass of the oligosaccharide being purified (i.e., the rejection ratio in this case is higher for the protein than for the desired saccharide). Proteins and other macromolecules that have a molecular mass greater than the MWCO will thus be rejected by the membrane, while the saccharide will pass through the membrane. Conversely, if an oligosaccharide or nucleotide sugar is to be purified from proteins that are smaller than the oligosaccharide or nucleotide sugar, a membrane is used that has a MWCO that is larger than the molecular mass of the protein but smaller than that of the oligosaccharide or nucleotide sugar. Generally, separation of proteins from carbohydrates will employ membranes that are commonly referred to as ultrafiltration (UF) membranes. UF membranes that are suitable for use in the methods of the invention are available from several commercial manufacturers, including Millipore Corp. (Bedford, Mass.), Osmonics, Inc. (Minnetonka, Minn.), Filmtec (Minneapolis, Minn.), UOP, Desalination Systems, Advanced Membrane Technologies, and Nitto.
- The invention also provides methods for removing salts and other low molecular weight components from a mixture containing a saccharide of interest by using a nanofiltration (NF) or a reverse osmosis (RO) membrane. Nanofiltration membranes are a class of membranes for which separation is based both on molecular weight and ionic charge. These membranes typically fall between reverse osmosis and ultrafiltration membranes in terms of the size of species that will pass through the membrane. Nanofiltration membranes typically have micropores or openings between chains in a swollen polymer network. Molecular weight cut-offs for non-ionized molecules are typically in the range from 100-20,000 Daltons. For ions of the same molecular weight, membrane rejections (retentions) will increase progressively for ionic charges of 0, 1, 2, 3 etc. for a particular membrane because of increasing charge density (see, e.g., Eriksson, P., “Nanofiltration Extends the Range of Membrane Filtration,” Environmental Progress, 7: 58-59 (1988)). Nanofiltration is also described in Chemical Engineering Progress, pp. 68-74 (March 1994), Rautenbach et al., Desalination 77: 73 (1990), and U.S. Pat. No. 4,806,244). In a typical application, saccharides of interest will be retained by the nanofiltration membrane and contaminating salts and other undesired components will pass through. A nanofiltration membrane useful in the methods of the invention will typically have a retention characteristic for the saccharide of interest of from about 40% to about 100%, preferably from about 70% to about 100%, more preferably from about 90% to about 100%. The nanofilter membranes used in the invention can be any one of the conventional nanofilter membranes, with polyamide membranes being particularly suitable. Several commercial manufacturers, including Millipore Corp. (Bedford, Mass.), Osmonics, Inc. (Minnetonka, Minn.), Filmtec, UOP, Advanced Membrane Technologies, Desalination Systems, and Nitto, among others, distribute nanofiltration membranes that are suitable for use in the methods of the invention. For example, suitable membranes include the Osmonics MX07, YK, GH (G-10), GE (G-5), and HL membranes, among others.
- Reverse osmosis (RO) membranes also allow a variety of aqueous solutes to pass through them while retaining selected molecules. Generally, osmosis refers to a process whereby a pure liquid (usually water) passes through a semipermeable membrane into a solution (usually sugar or salt and water) to dilute the solution and achieve osmotic equilibrium between the two liquids. In contrast, reverse osmosis is a pressure driven membrane process wherein the application of external pressure to the membrane system results in a reverse flux with the water molecules passing from a saline or sugar solution compartment into the pure water compartment of the membrane system. A RO membrane, which is semipermeable and non-porous, requires an aqueous feed to be pumped to it at a pressure above the osmotic pressure of the substances dissolved in the water. An RO membrane can effectively remove low molecular weight molecules (<200 Daltons) and also ions from water. Preferably, the reverse osmosis membrane will have a retention characteristic for the saccharide of interest of from about 40% to about 100%, preferably from about 70% to about 100%, and more preferably from about 90% to about 100%. Suitable RO membranes include, but are not limited to, the Filmtec BW-30, Filmtec SW-30, Filmtec SW-30HR, UOP RO membranes, Desal RO membranes, Osmonics RO membranes, Advanced Membrane Technologies RO membranes, and the Nitto RO membranes, among others. One example of a suitable RO membrane is Millipore Cat. No. CDRN500 60 (Millipore Corp., Bedford Mass.).
- The membranes used in the invention may be employed in any of the known membrane constructions. For example, the membranes can be flat, plate and frame, tubular, spiral wound, hollow fiber, and the like. In a preferred embodiment, the membrane is spiral wound. The membranes can be employed in any suitable configuration, including either a cross-flow or a depth configuration. In “cross-flow” filtration, which is preferred for ultrafiltration, nanofiltration and reverse osmosis purifications according to the invention, the “feed” or solution from which the carbohydrate of interest is to be purified flows through membrane channels, either parallel or tangential to the membrane surface, and is separated into a retentate (also called recycle or concentrate) stream and a permeate stream. To maintain an efficient membrane, the feed stream should flow, at a sufficiently high velocity, parallel to the membrane surface to create shear forces and/or turbulence to sweep away accumulating particles rejected by the membrane. Cross-flow filtration thus entails the flow of three streams—feed, permeate and retentate. In contrast, a “dead end” or “depth” filter has only two streams—feed and filtrate (or permeate). The recycle or retentate stream, which retains all the particles and large molecules rejected by the membrane, can be entirely recycled to the membrane module in which the recycle stream is generated, or can be partially removed from the system. When the methods of the invention are used to purify saccharides from lower molecular weight components, for example, the desired saccharides are contained in the retentate stream (or feed stream, for a depth filter), while the permeate stream contains the removed contaminants.
- The purification methods of the invention can be further optimized by adjusting the pressure, flow rate, and temperature at which the filtration is carried out. UF, NF, and RO generally require increasing pressures above ambient to overcome the osmotic pressure of the solution being passed through the membrane. The membrane manufacturers' instructions as to maximum and recommended operating pressures can be followed, with further optimization possible by making incremental adjustments. For example, the recommended pressure for UF will generally be between about 25 and about 100 psi, for NF between about 50 psi and about 1500 psi, and for RO between about 100 and about 1500 psi. Flow rates of both the concentrate (feed solution) and the permeate can also be adjusted to optimize the desired purification. Again, the manufacturers' recommendations for a particular membrane serve as a starting point from which to begin the optimization process by making incremental adjustments. Typical flow rates for the concentrate (Pc) will be between about 1 and about 15 gallons per minute (GPM), and more preferably between about 3 and about 7 GPM. For the permeate, flow rates (Pf) of between about 0.05 GPM and about 10 GPM are typical, with flow rates between about 0.2 and about 1 GPM being preferred. The temperature at which the purification is carried out can also influence the efficiency and speed of the purification. Temperatures of between about 0 and about 100° C. are typical, with temperatures between about 20 and 40° C. being preferred for most applications. Higher temperatures can, for some membranes, result in an increase in membrane pore size, thus providing an additional parameter that one can adjust to optimize a purification.
- In a preferred embodiment, the filtration is performed in a membrane purification machine which provides a means for automating control of flow rate, pressure, temperature, and other parameters that can affect purification. For example, the Osmonics 213T membrane purification machine is suitable for use in the methods of the invention, as are machines manufactured by other companies listed above.
- The membranes can be readily cleaned either after use or after the permeability of the membrane diminishes. Cleaning can be effected at a slightly elevated temperature if so desired, by rinsing with water or a caustic solution. If the streams contain small amounts of enzyme, rinsing in the presence of small amounts of surfactant, for instance ULTRASIL, is useful. Also, one can use prefilters (100-200 μm) to protect the more expensive nanofiltration membranes. Other cleaning agents can, if desired, be used. The choice of cleaning method will depend on the membrane being cleaned, and the membrane manufacturer's instructions should be consulted. The cleaning can be accomplished with a forward flushing or a backward flushing.
- The purification methods of the invention can be used alone or in combination with other methods for purifying carbohydrates. For example, an ion exchange resin can be used to remove particular ions from a mixture containing a saccharide of interest, either before or after nanofiltration/reverse osmosis, or both before and after filtration. Ion exchange is particularly desirable if it is desired to remove ions such as phosphate and nucleotides that remain after a first round of nanofiltration or reverse osmosis. In the case of sialyl lactose synthesis as discussed above, this can be accomplished, for example, by adding an anion exchange resin such as AG1X-8 (acetate form, BioRad; see, e.g., BioRad catalog for other ion exchange resins) to a retentate that is at about pH 3.0 or lower until the phosphate concentration is reduced as desired. In this process, acetic acid is released, so one may wish to follow the ion exchange with an additional purification through the nanofiltration or reverse osmosis system. For example, one can circulate the pH 3.0 or lower solution through an Osmonics MX07 or similar membrane until the conductivity of the permeate is low and stabilized. The pH of the solution can then be raised to 7.4 with NaOH and the solution recirculated through the same membrane to remove remaining sodium acetate and salt. Cations can be removed in a similar manner; for example, to remove Mn2+, an acidic ion exchange resin can be used, such as AG50WX8 (He) (BioRad).
- The purification methods of the invention are particularly useful for purifying oligosaccharides, modified saccharides, nucleotide sugars and modified nucleotide sugars prepared using enzymatic synthesis. Enzymatic synthesis using glycosyltransferases provides a powerful method for preparing oligosaccharides; for some applications it is desirable to purify the oligosaccharide from the enzymes and other reactants in the enzymatic synthesis reaction mixture. Preferred methods for producing many oligosaccharides involve glycosyl transferase cycles, which produce at least one mole of inorganic pyrophosphate for each mole of product formed and are typically carried out in the presence of a divalent metal ion. Examples of glycosyltransferase cycles are the sialyltransferase cycles, which use one or more enzymes as well as other reactants. See, e.g., U.S. Pat. No. 5,374,541 WO 9425615 A, PCT/US96/04790, and PCT/US96/04824. For example, a reaction used for synthesis of sialylated oligosaccharides can contain a sialyltransferase (
FIG. 7 ), a CMP-sialic acid synthetase, a sialic acid, an acceptor for the sialyltransferase, CTP, and a soluble divalent metal cation. An exemplary α(2,3)sialyltransferase referred to as α(2,3)sialtransferase (EC 2.4.99.6) transfers sialic acid to the non-reducing terminal Gal of a Galβ163Glc disaccharide or glycoside. See, Van den Eijnden et al., J. Biol. Chem., 256:3159 (1981), Weinstein et al., J. Biol. Chem., 257:13845 (1982) and Wen et al, J. Biol. Chem., 267:21011 (1992). Another exemplary α2,3-sialyltransferase (EC 2.4.99.4) transfers sialic acid to the non-reducing terminal Gal of the disaccharide or glycoside. See, Rearick et al., J. Biol. Chem., 254:4444 (1979) and Gillespie et al., J. Biol. Chem., 267:21004 (1992). Further exemplary enzymes include Gal-β-1,4-GlcNAc α-2,6 sialyltransferase (See, Kurosawa et al. Eur. J. Biochem. 219: 375-381 (1994)). The reaction mixture will also contain an acceptor for the sialyltransferase, preferably having a galactosyl unit. Suitable acceptors, include, for example, Galβ1→3 GalNAc, lacto-N-tetraose, Galβ1→3 GlcNAc, Galβ1→3Ara, Galβ1→6GlcNAc, Galβ1→4Glc (lactose), Galβ1→4Glcβ1-OCH2CH3, Galβ1→4Glcβ1-OCH2CH2CH3, Galβ1→4Glcβ1-OCH2C6H5, Galβ1→4GlcNAc, Galβ1-OCH3, melibiose, raffinose, stachyose, and lacto-N-neotetraose (LNnT). The sialic acid present in the reaction mixture can include not only sialic acid itself (5-N-acetylneuraminic acid; 5-N-acetylamino-3,5-dideoxy-D-glycero-D-galacto-2-nonulosonic acid; NeuAc, and sometimes also abbreviated AcNeu or NANA), but also 9-substituted sialic acids such as a 9-O—C1-C6 acyl-NeuAc like 9-O-lactyl-NeuAc or 9-O-acetyl-NeuAc, 9-deoxy-9-fluoro-NeuAc and 9-azido-9-deoxy-NeuAc. The synthesis and use of these compounds in a sialylation procedure is described in international application WO 92/16640, published Oct. 1, 1992. - In preferred embodiments the reaction medium can further comprise a CMP-sialic acid recycling system comprising at least 2 moles of phosphate donor per each mole of sialic acid, and catalytic amounts of an adenine nucleotide, a kinase capable of transferring phosphate from the phosphate donor to nucleoside diphosphates, and a nucleoside monophosphate kinase capable of transferring the terminal phosphate from a nucleoside triphosphate to CMP. For example, a suitable CMP-sialic acid regenerating system comprises cytidine monophosphate (CMP), a nucleoside triphosphate (for example adenosine triphosphate (ATP), a phosphate donor (for example, phosphoenolpyruvate or acetyl phosphate), a kinase (for example, pyruvate kinase or acetate kinase) capable of transferring phosphate from the phosphate donor to nucleoside diphosphates and a nucleoside monophosphate kinase (for example, myokinase) capable of transferring the terminal phosphate from a nucleoside triphosphate to CMP. The previously discussed α(2,3)sialyltransferase and CMP-sialic acid synthetase can also be formally viewed as part of the CMP-sialic acid regenerating system. For those embodiments in which a CMP-sialic acid recycling system is not used, the reaction medium will preferably further comprise a phosphatase.
- Pyruvate is a byproduct of the sialyltransferase cycle and can be made use of in another reaction in which N-acetylmannosamine (ManNAc) and pyruvate are reacted in the presence of NeuAc aldolase (EC 4.1.3.3) to form sialic acid. Alternatively, advantage can be taken of the isomerization of GlcNAc to ManNAc, and the less expensive GlcNAc can be used as the starting material for sialic acid generation. Thus, the sialic acid can be replaced by ManNAc (or GlcNAc) and a catalytic amount of NeuAc aldolase. Although NeuAc aldolase also catalyzes the reverse reaction (NeuAc to ManNAc and pyruvate), the produced NeuAc is irreversibly incorporated into the reaction cycle via CMP-NeuAc catalyzed by CMP-sialic acid synthetase. In addition, the starting material, ManNAc, can also be made by the chemical conversion of GlcNAc using methods known in the art (see, e.g., Simon et al., J. Am. Chem. Soc. 110:7159 (1988). The enzymatic synthesis of sialic acid and its 9-substituted derivatives and the use of a resulting sialic acid in a different sialylating reaction scheme is disclosed in International application WO 92/16640, published on Oct. 1, 1992, and incorporated herein by reference.
- When a galactosyltransferase is used for enzymatic synthesis of an oligosaccharide, the reaction medium will preferably contain, in addition to a galactosyltransferase, donor substrate, acceptor sugar and divalent metal cation, a donor substrate recycling system comprising at least 1 mole of glucose-1-phosphate per each mole of acceptor sugar, a phosphate donor, a kinase capable of transferring phosphate from the phosphate donor to nucleoside diphosphates, and a pyrophosphorylase capable of forming UDP-glucose from UTP and glucose-1-phosphate and catalytic amounts of UDP and a UDP-galactose-4-epimerase. Exemplary galactosyltransferases include α(1,3) galactosyltransferase (E.C. No. 2.4.1.151, see, e.g., Dabkowski et al., Transplant Proc. 25: 2921 (1993) and Yamamoto et al., Nature 345:229-233 (1990)) and β(1,4) galactosyltransferase (E.C. No. 2.4.1.38).
- Oligosaccharides synthesized by other enzymatic methods can also be purified by the methods of the invention. For example, the methods are useful for purifying oligosaccharides produced in non-cyclic or partially cyclic reactions such as simple incubation of an activated saccharide and an appropriate acceptor molecule with a glycosyltransferase under conditions effective to transfer and covalently bond the saccharide to the acceptor molecule. Glycosyltransferases, which include those described in, e.g., U.S. Pat. No. 5,180,674, and International Patent Publication Nos. WO 93/13198 and WO 95/02683, as well the glycosyltransferases encoded by the los locus of Neisseria (see, U.S. Pat. No. 5,545,553), can be bound to a cell surface or unbound. Oligosaccharides that can be obtained using these glycosyltransferases include, for example,
- GalNAcβ(1→3)Galβ(1→4)GlcNAcβ(1→3) Galβ(1→4)Glc, among many others.
- Among the compounds that one can purify using the described methods are sialic acid and any sugar having a sialic acid moiety. Exemplary species include sialic acid species modified with a linker (e.g., glycyl sialic acid) and with a polymer (e.g., poly(ethylene glycol). Other compounds include sialyl galactosides, including the sialyl lactosides, as well as compounds having the formula:
-
NeuAcα(2→3)Galβ(1→4)GlcN(R′)β-OR or -
NeuAcα(2→3)Galβ(3→4)GlcN(R′)β(1→3)Galβ-OR. - In these formulae, R′ is alkyl or acyl from 1-18 carbons, 5,6,7,8-tetrahydro-2-naphthamido; benzamido; 2-naphthamido; 4-aminobenzamido; or 4-nitrobenzamido. R is a hydrogen, a alkyl C1-C6, a saccharide, an oligosaccharide or an aglycon group having at least one carbon atom. The term “aglycon group having at least one carbon atom” refers to a group -A-Z, in which A represents an alkylene group of from 1 to 18 carbon atoms optionally substituted with halogen, thiol, hydroxy, oxygen, sulfur, amino, imino, or alkoxy; and Z is hydrogen, —OH, —SH, —NH2, —NHR1, —N(R1)2, —CO2H, —CO2R1, —CONH2, —CONHR1, —CON(R1)2, —CONHNH2, or —OR1 wherein each R1 is independently alkyl of from 1 to 5 carbon atoms. In addition, R can be:
-
(CH2)nCH(CH2)mCH3 - where n, m, and o are independently selected from the integers 1-18; (CH2)n—R2 (in which n=0-18), wherein R2 is a variously substituted aromatic ring, preferably, a phenyl group, being substituted with one or more alkoxy groups, preferably methoxy or O(CH2)mCH3, (in which m=0-18), or a combination thereof. R can also be 3-(3,4,5-trimethoxyphenyl)propyl.
- The present invention is also useful for purifying a variety of compounds that comprise selectin-binding carbohydrate moieties. These selectin-binding moieties have the general formula:
-
R1Galβ1,m(Fucα1,n)GlcNR0(R2)p— - in which R0 is (C1-C8 alkyl)carbonyl, (C1-C8 alkoxy)carbonyl, or (C2-C9 alkenyloxy)carbonyl, R1 is an oligosaccharide or a group having the formula:
- R3 and R4 may be the same or different and may be H, C1-C8 alkyl, hydroxy-(C1-C8 alkyl), aryl-(C1-C8 alkyl), or (C1-C8 alkoxy)-(C1-C8 alkyl), substituted or unsubstituted. R2 may be H, C1-C8 alkyl, hydroxy-(C1-C8 alkyl), aryl-(C1-C8-alkyl), (C1-C8 alkyl)-aryl, alkylthio, α1,2Man, α1,6GalNAc, β1,3Galβ1,4Glc, α1,2Man-R8, α1,6GalNAc-R8, and β1,3Gal-R8. R8 may be H, C1-C8 alkyl, C1-C8 alkoxy, hydroxy-(C1-C8 alkyl), aryl-(C1-C8 alkyl), (C1-C8 alkyl)-aryl, or alkylthio. In the formula, m and n are integers and may be either 3 or 4; p may be zero or 1.
- The substituted groups mentioned above may be substituted by hydroxy, hydroxy(C1-C4 alkyl), polyhydroxy(C1-C4 alkyl), alkanoamido, or hydroxyalknoamido substituents. Preferred substituents include hydroxy, polyhydroxy(C3 alkyl), acetamido and hydroxyacetamido. A substituted radical may have more than one substitution, which may be the same or different.
- For embodiments in which R1 is an oligosaccharide, the oligosaccharide is preferably a trisaccharide. Preferred trisaccharides include NeuAcα2,3Galβ1,4GlcNAcβ1,3 or NeuGcα2,3Galβ1,4GlcNAcβ1,3.
- For embodiments in which R1 is the group having the formula
- R3 and R4 preferably form a single radical having the formula
-
—R5— or —(R6)q—O—(R7)r— - in which R5 is C3-C7 divalent alkyl, substituted or unsubstituted, R6 and R7 are the same or different and are C1-C6 divalent alkyl, substituted or unsubstituted. In the formula, q and r are integers which may be the same or different and are either zero or 1. The sum of q and r is always at least 1.
- A more preferred structure for a single radical formed by R3 and R4 is one having the formula
-
—(R6)—O— - in which R6 is C3-C4 divalent alkyl, substituted or unsubstituted. For instance, R6 may have the formula —CH2—CH2—CH2—CH2—, preferably substituted. The radical can be substituted with hydroxy, polyhydroxy(C3 alkyl), and substituted or unsubstituted alkanoamido groups, such as acetamido or hydroxyacetamido. The substituted structure will typically form a monosaccharide, preferably a sialic acid such as NeuAc or NeuGc linked α2,3 to the Gal residue.
- In the general formula, above, both m and n are integers and can be either 3 or 4. Thus, in one set of structures Gal is linked β1,4 and Fuc is linked α1,3 to GlcNAc. This formula includes the SLex tetrasaccharide. SLex has the formula NeuAcα2,3Galβ1,4(Fucα1,3)GlcNAcβ1-. This structure is selectively recognized by LECCAM-bearing cells. SLex compounds that can be purified using the methods of the invention include NeuAcα2,3 Galβ1,4(Fucα1,3)GlcNAcβ1-Gal-OEt, NeuAcα2,3Galβ1,4(Fucα1,3)GlcNAcβ1,4Galβ1-OEt, and others that are described in international application WO 91/19502. Other compounds that one can purify using the methods include those described in U.S. Pat. No. 5,604,207 having the formula:
- wherein Z is hydrogen, C1-C6 acyl or
- Y is selected from the group consisting of C(O), SO2, HNC(O), OC(O) and SC(O). R1 is selected from the group consisting of an aryl, a substituted aryl and a phenyl C1-C3 alkylene group, wherein said aryl substitutent is selected from the group consisting of a halo, trifluoromethyl, nitro, C1-C18 alkyl, C1-C18 alkoxy, amino, mono-C1-C18 alkylamino, di-C1-C18 alkylamino, benzylamino, C1-C18 alkylbenzylamino, C1-C18 thioaklyl and C1-C18 alkyl carboxamido groups, or R1Y is allyloxycarbonyl or chloroacetyl. R2 is selected from the group consisting of monosaccharide (including β1,3Gal-OR, where R=H, alkyl, aryl or acyl), disaccharide, hydrogen, C1-C18 straight chain, branched chain or cyclic hydrocarbyl, C1-C6 alkyl, 3-(3,4,5-trimethoxyphenyl)propyl, C1-C5 alkylene-carboxylate, trisubstituted silyl C2-C4 alkylene wherein said trisubstituted silyl is a silyl group having three substituents independently selected from the group consisting of C1-C4 alkyl, phenyl, or OR2 together form a C1-C18 straight chain, branched chain or cyclic hydrocarbyl carbamate; R3 is hydrogen or C1-C6 acyl; R4 is hydrogen, C1-C6 alkyl or benzyl. R5 is selected from the group consisting of hydrogen, benzyl, methoxybenzyl, dimethoxybenzyl and C1-C6 acyl. R7 is methyl or hydroxymethyl. X is selected from the group consisting of C1-C6 acyloxy, C2-C6 hydroxylacyloxy, hydroxy, halo and azido.
- A related set of structures included in the general formula are those in which Gal is linked β1,3 and Fuc is linked α1,4. For instance, the tetrasaccharide, NeuAcα2,3Galβ1,3(Fucα1,4)GlcNAcβ1-, termed here SLea, is recognized by selectin receptors. See, Berg et al., J. Biol. Chem., 266:14869-14872 (1991). In particular, Berg et al. showed that cells transformed with E-selectin cDNA selectively bound neoglycoproteins comprising SLea.
- The methods of the invention are also useful for purifying oligosaccharide compounds having the general formula Galα1,3Gal-, including Galα1,3Galβ1,4Glc(R)β-O—R1, wherein R1 is —(CH2)n—COX, with X=OH, OR2, —NHNH2, R═OH or NAc, and R2 is a hydrogen, a saccharide, an oligosaccharide or an aglycon group having at least one carbon atom, and n=an integer from 2 to 18, more preferably from 2 to 10. For example, one can purify a compound having the formula Galα1,3Galβ1,4GlcNAcβ-O—(CH2)5—COOH using procedures such as those described in Examples 7-8. Also among the compounds that can be purified according to the invention are lacto-N-neotetraose (LNnT), GlcNAcβ1,3Galβ1,4Glc (LNT-2), sialyl(α2,3)-lactose, and sialyl(α2,6)-lactose.
- In one embodiment, a modified sialic acid has the following structure:
- wherein, R1, R2, R3 and R4 are each independently selected from H, OR5, NR6R7, substituted alkyl, unsubstituted alkyl, substituted heteroalkyl and unsubstituted heteroalkyl. R5 is H, substituted alkyl, unsubstituted alkyl, substituted heteroalkyl or unsubstituted heteroalkyl. The symbols R6 and R7 independently represent H, substituted alkyl, unsubstituted alkyl, substituted heteroalkyl and unsubstituted heteroalkyl. B is a nucleoside. Exemplary nucleosides include AMP, UMP, GMP, CMP, TMP, ADP, UDP, GDP, CDP, TDP, ATP, UTP, GTP, CTP, TTP, cAMP and cGMP.
- In a preferred embodiment, the sialic acid is modified with a linker group. Preferred sites for such modification are R1 or R2. Thus, in a preferred embodiment, at least one of R1 and R2 includes a linker. An exemplary linker is a glycyl linker.
- In a preferred embodiment, the modified sialic acid has the following structure:
- In yet another preferred embodiment, a modifying group is attached to the sialic acid through the linker. An exemplary species according to this description includes a modifying group attached through the free amine moiety of the linker in the figure above. A presently preferred modifying group is a water-soluble polymer. Poly(ethylene glycol) is a preferred water-soluble polymer.
- The methods of the invention are useful not only for purifying carbohydrates (and modified carbohydrates and nucleotide sugars) that that are newly synthesized, but also those that are the products of degradation, e.g., enzymatic degradation. See, e.g., Sinnott, M. L., Chem. Rev. 90: 1171-1202 (1990) for examples of enzymes that catalyze degradation of oligosaccharides.
- The invention also provides methods for purifying nucleotides, nucleotide sugars, and related compounds. For example, a nucleotide sugar such as GDP-fucose, GDP-mannose, CMP-NeuAc, UDP-glucose, UDP-galactose, UDP-N-acetylgalactosamine, and the like, can be purified by the methods described herein. The methods are also useful for purifying nucleotides and nucleotides in various states of phosphorylation (e.g., CMP, CDP, CTP, GMP, GDP, GTP, TMP, TDP, TTP, AMP, ADP, ATP, UMP, UDP, UTP), as well as the deoxy forms of these and other nucleotides, including modified nucleotides. The method of the invention can be used to prepare and purify nucleotide sugars to a high degree of purity on a multi-kilogram scale (e.g., at least about 1 kg, preferably at least about 1.5 kg, more preferably at least about 2 kg, and even more preferably, at least about 3 kg of purified sugar nucleotide per synthesis/purification run). An exemplary process flow chart is set forth in
FIG. 1 . - In the discussion that follows, focus is placed on the purification of nucleotide sugars. The methods set forth hereinbelow are equally applicable to the purification of sugars, modified sugars and modified nucleotides sugars (e.g., those bearing a linker arm (e.g., a glycl linker arm), a modifying group (e.g., a water-soluble polymer (e.g., PEG)), or a modifying group attached to the linker arm (e.g., PEG attached to the sugar through a glycyl linker).
- The process of the invention routinely provides nucleotide sugars, e.g., CMP-NAN, in recovered yields of purified materials in greater that 40%, e.g., of from about 40% to about 80%. In a preferred embodiment, the yield of isolated CMP-NAN is from about 50% to about 70% of the theoretical synthesis yield.
- In general, the process of the invention provides nucleotide sugars that are at least 80% pure, preferably at least 85% pure, more preferably, at least 90% pure and still more preferably, at least 95% pure.
- In a representative embodiment, the nucleotide sugar is a CMP-sialic acid, e.g., CMP-NAN (N-acetylneuraminic acid). The generic process for purification of nucleotide sugars is exemplified in the context of CMP-sialic acid, however, this focus is for clarity of illustration and should not be construed as limiting the process to practice with CMP-sialic acids.
- In a preferred embodiment, a membrane-based methodology is utilized to purify the nucleotide sugar from reaction components. In the case of CMP-sialic acid, exemplary reaction components include cytidine monophosphate and its active analogues, and cytidine diphosphate, unreacted sialic acid, salts (e.g., PO4 3−, Mn2+).
- In a preferred embodiment, the amount of CMP, CDP and/or CTP of the product is less than about 20%, preferably, less than about 15%, more preferably, less than about 10% and still more preferably less than 5%.
- In another preferred embodiment, the content of unreacted sialic acid, e.g., NAN, in the final product is less than about than about 20%, preferably, less than about 15%, more preferably, less than about 10%, still more preferably, less than about 5% and even more preferably, less than about 2%.
- In yet another preferred embodiment, the phosphate content of the final product is less than about 5%, preferably, less than about 2%, and more preferably, about 0%.
- The invention also provides methods for synthesizing and purifying nucleotide sugars. The nucleotide sugar is enzymatically synthesized from a nucleotide and a sugar in the presence of an enzyme. After the nucleotide sugar is synthesized, the nucleotide sugar is purified according to a method of the invention.
- In one embodiment, following synthesis, a nucleotide sugar solution is optionally clarified by filtration. The nucleotide sugar solution passes through a membrane bag filter in which contaminating salts and other undesired contaminants are filtered out of the nucleotide sugar solution. The clarification step can be incorporated at any step of the process. In a preferred embodiment, the nucleotide sugar solution is clarified after synthesis of the nucleotide sugar. The nucleotide sugar solution may be clarified one or more times.
- In another embodiment, the nucleotide sugar solution is purified using hollow fiber filtration. Hollow fiber filtration removes proteins introduced by the enzyme preparation of the nucleotide sugar. The hollow fiber membrane retains proteins from the enzyme preparation while allowing for passage of the nucleotide sugar solution through the membrane. In an exemplary embodiment, the hollow fiber membrane comprises a hollow fiber membrane with a tangential filtration skid. The hollow fiber filtration step can be incorporated at any step of the process. In one embodiment, the nucleotide sugar solution goes through hollow fiber filtration after clarification. In another embodiment, the nucleotide sugar solution goes through hollow fiber filtration after synthesis of the nucleotide sugar. The nucleotide sugar solution may be filtered one or more times using hollow fiber filtration.
- In another embodiment, the nucleotide sugar solution is purified using nanofiltration. Nanofiltration removes salts and other low molecular weight components from a mixture. Nanofiltration membranes separate molecules based on molecular weight and ionic charge. Molecular weight-cutoffs for non-ionized molecules are typically in the range from 100-20,000 daltons. In an exemplary application, saccharides of interest will be retained by the nanofiltration membrane and contaminating salts and other undesired components will pass through. The nanofiltration step can be incorporated at any step of the process. In one embodiment, the nucleotide sugar solution goes through hollow-fiber filtration first and then nanofiltration. In another embodiment, the nucleotide sugar solution goes through nanofiltration first and then hollow fiber filtration. In the alternative, the nucleotide sugar solution may be purified using either hollow-fiber filtration or nanofiltration. In another embodiment, the nucleotide sugar solution goes through nanofiltration after clarification. In yet another embodiment, the nucleotide sugar solution goes through nanofiltration after synthesis of the nucleotide sugar. The nucleotide sugar solution may be filtered one or more times using nanofiltration. After nanofiltration, the purified nucleotide sugar solution may generally be stored or may undergo further purification.
- In another embodiment, the nucleotide sugar solution may optionally be decolorized (e.g., by passing the solution over activate carbon). In a preferred embodiment, decolorization involves passing the nucleotide sugar solution over a pre-packed column of activated carbon attached to a chromatography system. Decolorization can be incorporated at any step of the process. In one embodiment, the nucleotide sugar solution is decolorized after nanofiltration. In another embodiment, the nucleotide sugar solution is decolorized after hollow-fiber filtration. In yet another embodiment, the nucleotide sugar solution is decolorized after clarification. The nucleotide sugar solution may be decolorized one or more times.
- In another embodiment, the nucleotide sugar solution is purified using a charged depth media filter. The charged depth media filter removes endotoxins from the nucleotide sugar solution. Endotoxins are toxic, natural compounds such as lipopolysaccharides found inside pathogens on the outer cell wall of bacteria. Purification by a charged depth media filter can be incorporated at any step of the process. In one embodiment, the nucleotide sugar solution is filtered after decolorization.
- In another embodiment, the nucleotide sugar solution is purified by a charged depth media filter after nanofiltration. In yet another embodiment, the nucleotide sugar solution is purified by a charged depth media filter after hollow-fiber filtration. In another embodiment, the nucleotide sugar solution is purified by a charged depth media filter after clarification. In another embodiment, the nucleotide sugar solution is purified by a charged depth media filter after synthesis of the nucleotide sugar. The nucleotide sugar solution may be filtered one or more times using a charged depth media filter.
- In another embodiment, the nucleotide sugar solution is purified using a sterile filter. The sterile filter removes contaminating salts and other undesired contaminants from the nucleotide sugar solution. In a more preferred embodiment, the sterile filter is pre-packaged and sterilized with a bag manifold system for final filtration and storage. Purification by a sterile filter can be incorporated at any step of the process. In one embodiment, the nucleotide sugar solution is filtered by a sterile filter after purification by a charged depth media filter. In another embodiment, the nucleotide sugar solution is purified by a sterile filter after decolorization. In yet another embodiment, the nucleotide sugar solution is purified by a sterile filter after nanofiltration. In another embodiment, the nucleotide sugar solution is purified by a sterile filter after hollow fiber filtration. In another embodiment, the nucleotide sugar solution is purified by a sterile filter after clarification. In another embodiment, the nucleotide sugar solution is purified by a sterile filter after synthesis of the nucleotide sugar. The nucleotide sugar solution may be filtered one or more times using a sterile filter.
- An exemplary process of nucleotide sugar purification is described in
FIG. 1 . The nucleotide sugar is first synthesized (1) from a nucleotide and a sugar in the presence of an enzyme. - After the nucleotide sugar is synthesized, the nucleotide sugar is clarified by filtration (2) and transferred into a mobile tank (3). The clarified nucleotide sugar solution is concentrated using a hollow fiber filtration unit with a tangential flow filtration skid and diafiltered with purified water (4) and (5). The nucleotide sugar solution is transferred to a mobile tank (6) and the pH is adjusted.
- The purified solution is concentrated again and diafiltered with purified water using a nanofiltration system (7) and (8). The nucleotide sugar solution is then decolorized in which the color is removed from the nucleotide sugar solution (9).
- The nucleotide sugar solution subsequently undergoes two filtrations. First, the nucleotide sugar solution is filtered using a charged depth media filter in which endotoxins are removed (10). In an exemplary embodiment, the charged depth media filter is a CUNO Zeta Plus 60 ZA filter or an equivalent. After the nucleotide sugar solution is filtered through the charged depth media filter, the nucleotide sugar solution is optionally filtered using a 0.2 μm sterile filter (11). In an exemplary embodiment, the sterile filter is a CUNO LifeASSURE 0.2 μm sterile filter or an equivalent. The purified nucleotide sugar solution is appropriate for storage.
- Exemplary nucleotide sugars that can be purified by the method described above include, but are not limited to, CMP-NAN, GDP-fucose, GDP-mannose, CMP-NeuAc, UDP-Glucose, UDP-galactose and UDP-N-acetylgalactosamine, and modified analogues thereof. In a preferred embodiment, the nucleotide sugar is CMP-NAN or a modified CMP-NAN.
- The invention also provides methods for synthesizing and purification of a nucleotide-Glycyl Sialic Acid (“nucleotide-GSC”).
- In one embodiment, the synthesis of the nucleotide-GSC begins with the synthesis of the protected Fmoc-glycyl-mannosamine (“FGM”). In an exemplary embodiment, mannosamine and Fmoc-Glycyl-OSU are reacted in an aqueous solution under basic conditions. The aqueous solution may contain a base, e.g., sodium methoxide and an organic cosolvent, e.g., methanol, to facilitate the reaction.
- The FGM is optionally purified. In an exemplary embodiment, FGM is purified by chromatography, e.g., silica gel chromatography. FGM may be chromatographed one or more times.
- In another embodiment, FGM converted to the corresponding sialic acid analogue by reaction with pyruvate to form Fmoc-glycyl-sialic acid (“FSC”). This reaction is efficiently catalyzed by a sialic acid aldolase. Appropriate sialic acid aldolases are commercially available. In an exemplary embodiment, the reaction mixture includes at least the nucleotide, the aldolase, MnCl2 and water. The resulting FSC is then coupled to a nucleotide in the presence of an enzyme to form the resulting product, nucleotide-FSC.
- The nucleotide-FSC is optionally purified. In an exemplary embodiment, the nucleotide-FSC is purified by chromatography, e.g., reverse phase chromatography. In an exemplary embodiment, the reverse phase chromatography is C18 reverse phase chromatography. The nucleotide FSC can be filtered (e.g., 0.22 filter) prior to and/or after chromatography.
- The nucleotide-FSC is preferably deprotected to produce the free amine analogue of the linker-nucleotide sugar construct. In an exemplary embodiment, deprotection is effected with methanol:water:dimethylamine. Deprotection results in a modified sialic acid, nucleotide-Glycyl-Sialic Acid (“GSC”).
- The GSC is optionally purified, filtered and/or lyophilized.
- An exemplary process for nucleotide-GSC synthesis and purification is described in
FIG. 4 . - The synthesis of nucleotide-GSC begins with the synthesis and concentration of FGM (12). FGM is then purified using silica flash column chromatography (13). FGM is reacted with pyruvate to form FSC (14). The resulting FSC is then coupled to a nucleotide in the presence of an enzyme to form the resulting product, nucleotide-FSC (14). The nucleotide-FSC is then purified (15). The nucleotide-FSC is deprotected (16) thus cleaving off the Fmoc group from the nucleotide-FSC. The resultant product is a nucleotide-GSC. The nucleotide-GSC is further purified and concentrated (16). The nucleotide-GSC is then lyophilized (17) and subsequently released for testing (18).
- An exemplary nucleotide that can be used for the synthesis and purification of a nucleotide-GSC includes, but is not limited to, CMP, CDP, CTP, GMP, GDP, GTP, TMP, TDP, TTP, AMP, ADP, ATP, UMP, UDP, UTP, as well as the deoxy forms of these and other nucleotides. In a preferred embodiment, the nucleotide is CMP.
- The invention provides also methods for synthesizing and purification of a nucleotide-Sialic Acid (“SA”)-PEG. In one embodiment, the synthesis of the nucleotide-SA-PEG begins with Fmoc-glycyl-mannosamine (“FGM”). Synthesis and purification of this starting material is discussed above. GSC and methoxy-paranitrophenyl-carbomate-polyethylene glycol (“mPEG-pNP”) are combined in under conditions suitable to allow formation of a conjugate between the PEG and the free amine of the glycyl linker. In a preferred embodiment, the synthesis reaction is performed in a 80% THF: 20% H2O solution.
- In another embodiment, the nucleotide-SA-PEG is purified. In a preferred embodiment, the nucleotide-SA-PEG is purified by reverse phase chromatography.
- An exemplary process of nucleotide-SA-PEG (“PSC”) synthesis and purification is described in
FIG. 5 . - The synthesis of nucleotide-SA-PEG begins with the synthesis of FGM (19). FGM is rotovapped (20). FGM is then purified using silica column chromatography (21). FGM is detected by a UV light (22) and rotovapped (23). FGM is reacted with pyruvate to form FSC (24). The resulting FSC is then coupled to a nucleotide in the presence of an enzyme to form the resulting product, nucleotide-FSC (24). The nucleotide-FSC is then purified using a 0.2 μm filter (25) and a C18 reverse phase chromatography (26). The solution is then detected by a UV ray (27). The nucleotide-FSC is deprotected (28) thus cleaving off the Fmoc group from the nucleotide-FSC. The resultant product is a nucleotide-GSC. The nucleotide-GSC is rotovapped (29). The nucleotide-GSC is further purified (30) by a 0.2 μm filter. The nucleotide-GSC is then lyophilized (31). The nucleotide-GSC is reacted with a mPEG-pNP to form nucleotide-SA-PEG (“PSC”) (32). The PSC is purified by reverse phase chromatography (33). The PSC is detected by UV light (34) and rotovapped (35). The PSC is then lyophilized (36).
- An exemplary nucleotide that can be used for the synthesis and purification of a nucleotide-SA-PEG includes, but is not limited to, CMP, CDP, CTP, GMP, GDP, GTP, TMP, TDP, TTP, AMP, ADP, ATP, UMP, UDP, UTP, as well as the deoxy forms of these and other nucleotides. In a preferred embodiment, the nucleotide is CMP.
- The invention further provides methods for purifying a glycosyltransferase. In one embodiment, a glycosyltransferase solution is harvested, isolating the enzyme from cell culture and other debris to produce a suitable feed material for subsequent purification steps. In an exemplary embodiment, the harvesting reaction occurs at about
pH 6. The harvesting step can be incorporated at any step of the process. - The glycosyltransferase is optionally precipitated from the solution. In an exemplary embodiment, the glycosyltransferase is precipitated by addition of calcium chloride (“CaCl2”) to the solution. The pH of the solution can be adjusted as appropriate. In an exemplary embodiment, the pH of the precipitation reaction is adjusted to about 7.5. The mixture can be incubated for any suitable time under appropriate conditions. In an exemplary embodiment, the precipitation step lasts for about 30 minutes at about 4° C. Other exemplary additives include EDTA.
- The recovery of glycosyltransferase after precipitation is about 80% to about 100%, preferably from about 90% to about 100%, even more preferably about 100%.
- In another embodiment, the glycosyltransferase solution is subjected to membrane filtration. Exemplary membrane filters have a pore size of about 0.1 μm to about 0.5 μm, preferably about 0.1 μm to about 0.3 μm, and more preferably about 0.20 μm to about 0.25 μm. The filtration step can be incorporated at any step of the process.
- In another embodiment, the glycosyltransferase solution is ultrafiltrated. In ultrafiltration, products of high molecular weight are retained on the membrane, while low molecular weight solutes pass through the membrane. In an exemplary embodiment, the ultrafiltration membrane has a molecular weight cut-off (MWCO) between about 5 kDa and about 200 kDa.
- In an exemplary embodiment, the glycosyltransferase solution is equilibrated with 15 mM sodium phosphate buffer, pH 7.5 and conductivity is 2 ms/cm. In another embodiment, the glycosyltransferase solution is equilibrated with 15 mM sodium phosphate, pH 7.5. In another embodiment, the glycosyltransferase solution is equilibrated with 15 mM sodium phosphate, 0.01M NaCl, pH 7.5. In another embodiment, the glycosyltransferase is equilibrated with 15 mM sodium phosphate, 0.05M NaCl, pH 7.5. In another embodiment, the glycosyltransferase solution is equilibrated with 15 mM sodium phosphate, 0.10M NaCl, pH 7.5. In another embodiment, the pH range of the sodium phosphate may range from about 5.5 to about 8.5. In a preferred embodiment, the pH range of the glycosyltransferase solution may range from about 6.5 to about 7.5. In another embodiment, the test excipient includes, but is not limited to, glycerol, mannitol, sorbitol, sucrose and Tween-20. In one embodiment, the percent recovery of glycosyltransferase after ultrafiltration is about 45% to about 85%. In a preferred embodiment, the percent recovery of glycosyltransferase after ultrafiltration is about 55% to about 75%. In a more preferred embodiment, the percent recovery of glycosyltransferase after ultrafiltration is about 65%. The ultrafiltration step can be incorporated at any step of the process. In a preferred embodiment, the glycosyltransferase solution is ultrafiltrated after the filtration step. In another preferred embodiment, the glycosyltransferase solution is ultrafiltrated after the precipitation step.
- In another preferred embodiment, the glycosyltransferase solution is ultrafiltrated after the harvesting step. The glycosyltransferase solution may be ultrafiltrated one or more times.
- In another embodiment, the glycosyltransferase is purified by chromatography, e.g., ion exchange chromatography. In an exemplary embodiment, the glycosyltransferase is purified using an anion exchange column. In a further exemplary embodiment, the anion exchange column is a Mustang Q or an equivalent. The recovery of glycosyltransferase after anion exchange chromatography is preferably from about 80% to about 100%. In a preferred embodiment, the recovery of glycosyltransferase after chromatography is about 90% to about 100%. In a more preferred embodiment, the recovery of glycosyltransferase after chromatography is about 100%. The chromatography step can be incorporated at any step of the process. In a preferred embodiment, the glycosyltransferase is purified by chromatography after an ultrafiltration step. In a preferred embodiment, the glycosyltransferase is purified by chromatography after the filtration step. In another preferred embodiment, the glycosyltransferase is purified by chromatography after the precipitation step. In another preferred embodiment, the glycosyltransferase is purified by chromatography after the harvesting step.
- In another exemplary embodiment, the glycosyltransferase solution purified using a cation exchange column. An exemplary cation exchange chromatography protocol utilizes a Unosphere S resin or an equivalent. In a preferred embodiment, the cation exchange column is a 30S or an equivalent.
- The cation exchange column is eluted with at least one buffer. In one embodiment, the first buffer comprises a 15 mM sodium phosphate, pH 7.5. In another embodiment, the column is further eluted with a second buffer. An exemplary second buffer includes 1M NaCl, 15 mM sodium phosphate, pH 7.5.
- Elution of the column is optionally performed while developing a eluant gradient. In an exemplary embodiment, the added elution step has a rate of about 5 ms/cm to about 10 ms/cm. The recovery of glycosyltransferase after cation exchange chromatography is about 35% to about 75%. In a preferred embodiment, the recovery of glycosyltransferase after chromatography is about 45% to about 65%. In a more preferred embodiment, the percent recovery of glycosyltransferase after chromatography is about 55%. The chromatography step can be incorporated at any step of the process. In a preferred embodiment, the glycosyltransferase is purified by cation exchange chromatography after the anion exchange chromatography step described in the immediately preceding paragraph. In a preferred embodiment, the glycosyltransferase is purified by anion or cation exchange chromatography after the ultrafiltration step. In another preferred embodiment, the glycosyltransferase is purified by ion exchange chromatography after the filtration step. In yet another preferred embodiment, the glycosyltransferase is purified by chromatography after the precipitation step. In another preferred embodiment, the glycosyltransferase solution is purified by chromatography after the harvesting step. The glycosyltransferase solution may be purified by chromatography one or more times.
- In an exemplary embodiment, the glycosyltransferase is purified by hydroxyapatite (HA) chromatography. Exemplary hydroxyapatite sorbents are selected from ceramic and crystalline hydroxyapatite materials. In an exemplary embodiment the particle size of the ceramic hydroxyapatite sorbent is between about 20 μm and about 180 μm, preferably about 60 to about 100 μm, and, more preferably about 80 μm. In a preferred embodiment, the recovery of glycosyltransferase after HA chromatography is about 50% to about 90%, preferably from about 60% to about 90%. In a more preferred embodiment, the percent recovery of glycosyltransferase after chromatography is about 72%. HA chromatography can be incorporated at any step of the process. In a preferred embodiment, the glycosyltransferase is purified by HA chromatography after the cation chromatography step described above in the immediately preceding paragraph. In a preferred embodiment, the glycosyltransferase is purified by HA chromatography after the anion exchange chromatography step described above. In another preferred embodiment, the glycosyltransferase is purified by HA chromatography after the ultrafiltration step. In still another preferred embodiment, the glycosyltransferase is purified by HA chromatography after the filtration step. In another preferred embodiment, the glycosyltransferase is purified by HA chromatography after the precipitation step. In another preferred embodiment, the glycosyltransferase is purified by HA chromatography after the harvesting step. The glycosyltransferase solution may be purified by HA chromatography one or more times.
- In another exemplary embodiment, the glycosyltransferase is purified by hydrophobic interaction chromatography (“HIC”). In an exemplary embodiment, the hydrophobic moieties of the column matrix are selected from, but are not limited to, alkyl groups, aromatic groups and ethers. In an exemplary embodiment, the HIC column is packed with a phenyl 650M resin, butyl 650M resin, phenyl HP resin.
- In an exemplary embodiment, the glycosyltransferase solution is equilibrated with 0.5M ammonium sulfate, 20 mM sodium phosphate,
pH 7. - HIC chromatography can be incorporated at any step of the process. In a preferred embodiment, the glycosyltransferase is purified by HIC chromatography after purification by the HA chromatography step described above in the immediately preceding paragraph. In a preferred embodiment, the glycosyltransferase is purified by chromatography after the cation exchange chromatography step described above. In another preferred embodiment, the glycosyltransferase is purified by chromatography after the anion exchange chromatography step described above. In a preferred embodiment, the glycosyltransferase solution is purified by chromatography after the ultrafiltration step. In a preferred embodiment, the glycosyltransferase solution is purified by chromatography after the filtration step. In another preferred embodiment, the glycosyltransferase solution is purified by chromatography after the precipitation step. In another preferred embodiment, the glycosyltransferase solution is purified by chromatography after the harvesting step. The glycosyltransferase solution may be purified by chromatography one or more times.
- The glycosyltransferase can be purified by ultrafiltration. In an exemplary embodiment, the glycosyltransferase solution is equilibrated with 5 mM Bis-Tris, 0.1M NaCl, 5% sorbitol, pH 6.5. It is generally preferred that the recovery of glycosyltransferase after ultrafiltration is about 80% to about 100%. In a preferred embodiment, the percent recovery of glycosyltransferase after chromatography is about 90% to about 100%.
- Ultrafiltration can be incorporated at any step of the process. In a preferred embodiment, the glycosyltransferase is purified by ultrafiltration after the HIC chromatography step in the immediately preceding paragraph described above. In a preferred embodiment, the glycosyltransferase is purified by ultrafiltration after purification by the HA chromatography step described in the preceding paragraph above. In another preferred embodiment, the glycosyltransferase is purified by ultrafiltration after the cation exchange chromatography step described above. In still a further preferred embodiment, the glycosyltransferase is purified by ultrafiltration after the anion exchange chromatography step described above. In another embodiment, the glycosyltransferase is purified by a second ultrafiltration step after the first ultrafiltration step described above. The glycosyltransferase can be purified by ultrafiltration after the filtration step or precipitation steps. In still another embodiment, the glycosyltransferase is purified by ultrafiltration after the harvesting step. The glycosyltransferase may be purified by ultrafiltration one or more times.
- In another embodiment, the total percent recovery of glycosyltransferase from the purification process is about 5% to about 45%. In a preferred embodiment, the percent recovery of glycosyltransferase from the purification process is about 15% to about 35%.
- An exemplary process of a glycosyltransferase purification scheme is described in
FIG. 6 . The glycosyltransferase is first harvested (37). In an exemplary embodiment, the harvesting reaction takes place atpH 6. After harvesting, the glycosyltransferase is precipitated using calcium chloride (38). The glycosyltransferase is then filtered (39). After filtration, the glycosyltransferase undergoes ultrafiltration (40). The glycosyltransferase passes through an anion exchange column such as Mustang Q (41) or an equivalent. After passing through the anion exchange column, the glycosyltransferase passes through a cation exchange column, such as UNOsphere S (42) or an equivalent. The glycosyltransferase is then passed through a HA Type I chromatography column (43). The glycosyltransferase then undergoes ultrafiltration (44). - In one embodiment, the glycosyltransferase is a sialyltransferase. In a more preferred embodiment, the sialyltranferase includes, but is not limited to, ST6GalNac, ST3Gal3, α(2,3)-sialyltransferase, α(2,6)-sialyltransferase and α(2,8)-sialyltransferase. In another embodiment, the sialyltransferase includes, but is not limited to, a sialyltranferase listed in the table in
FIG. 7 . In a more preferred embodiment, the sialyltransferase is ST6GalNac. - The following examples are offered solely for the purposes of illustration, and are intended neither to limit nor to define the invention.
- The synthesis of Fmoc-glycyl-mannosamine (FGM) occurred in a non-aqueous solution involving two main compounds: D-Mannosamine HCl and Fmoc-Glycyl-OSu. Both materials were dry powders that were introduced into a system comprised of anhydrous methanol and sodium methoxide. The reaction was agitated at 25° C. for 1 hThe reaction was complete when the FGM concentration was greater than 15 mg/mL, determined by HPLC. The FGM synthesis was then rotovapped (20° C.) to about 8% of the initial volume. The chromatographic purification was performed using a Biotage pre-packed silica column. The FGM solution was loaded onto the column in a 50:50 CHCl3:CH3OH solution. The silica column was then washed with 18 column volumes (CV) of 3% CHCl3/97% CH3OH. Following the wash, FGM was eluted from the column using 14 CV of 15% CHCl3/85% CH3OH. Fractions containing material were pooled and then rotovapped (20° C.) to dryness and stored at 4° C. The average recovery for this step from the consistency batches was 73.8%.
- Raw Materials: The table below lists the materials used during the synthesis and purification of Fmoc-Glycyl-Mannosamine (FGM). The first four reagents in the table were used during the synthesis of FGM. The last two reagents were used during the purification of FGM.
-
D-Mannosamine HCl 9.0 g Fmoc-Glycyl-OSu 36.2 g Sodium Methoxide, 0.5 M 184 mL Anhydrous Methanol 726 mL Methanol, ACS 1.4 L Chloroform, ACS 16.2 L - The silica column was a Biotage 75M Silica column. The column volume for a 75M column was 0.5 L.
- The production of CMP-Sialic Acid-PEG (PSC) was performed in two segments. First, a key intermediate, CMP-Glycyl-Sialic Acid (GSC), was synthesized, purified, and dried, and second, this intermediate was PEGylated, purified, and dried. A synthetic pathway for CMP-SA-PEG is shown below.
- The first step in the synthesis of GSC was the reaction of mannosamine with Fmoc-Gly-OSu in methanol under basic conditions. The resulting Fmoc-glycyl-mannosamine was purified on a silica flash chromatography column. The purified Fmoc-glycyl-mannosamine then entered a two step enzymatic reaction. Fmoc-glycyl-mannosamine (FGM) was reacted with pyruvate to convert to Fmoc-glycyl-sialic acid. This reaction was catalyzed by a commercially available sialic acid aldolase. Fmoc-glycyl-sialic acid was then coupled to cytidine-5′-monophosphate through a CMP-NAN synthetase catalyzed reaction with cytidine-5′-triphosphate. The resulting product, CMP-Fmoc-glycyl-sialic acid, was purified on a C18 reverse phase column.
- Finally, the Fmoc-group was removed from the CMP-Fmoc-glycyl-sialic acid by deprotection with dimethylamine, forming a precipitate. The resulting precipitate was filtered out of solution using a 0.2 μm membrane and discarded. The resulting solution was dried in a freeze dryer, yielding a white powder of CMP-glycyl-sialic acid (GSC).
- Both CMP-SA-10K PEG and CMP-SA-20K PEG were synthesized in a single step reaction of GSC with the appropriately sized mPEG-pNP. This reaction was performed in an 80% THF:20% H2O solution. Purification of the final product, CMP-SA-PEG (PSC), was performed by reverse phase chromatography. CMP-SA-10K PEG was purified using a C8 reverse phase chromatography resin, and CMP-SA-10K PEG was purified using a C4 reverse phase chromatography resin. The eluent from the reverse phase column was dried by lyophilization, yielding a white powder of purified CMP-SA-PEG.
- The process was scaled to produce approximately 10 g of the GSC intermediate. Approximately 5.5 g of CMP-SA-20K was produced from 0.5 g of GSC, while approximately 5.0 g of CMP-SA-10K PEG was produced from 1.0 g of GSC. The final CMP-SA-PEG products were approximately 90% pure with the major impurities being CMP and sialic acid-PEG, the primary breakdown products of CMP-SA-PEG.
- The synthesis of CMP-Fmoc-glycyl-sialic acid (FSC) was a two-step enzymatic process performed simultaneously in one pot. Reacting with pyruvate, Fmoc-glycyl-mannosamine (FGM) was converted to Fmoc-glycyl-sialic acid, catalyzed by sialic acid-aldolase. Fmoc-glycyl-sialic acid was then coupled to cytidine-5′-monophosphate through a CMP-NAN synthetase catalyzed reaction with cytidine-5′-triphosphate.
- The pH of the reaction was controlled at 7.5±0.5. Temperature was controlled at 30±2° C., and the reaction was continuously agitated. The reaction reached completion in 6-24 hrs. and the extent-of-reaction was determined as a function of the FSC concentration (≧14.0 g/L, 95% of theoretical conversion). The resulting product solution can be stored at 4° C. until the purification is executed or for up to 72 h. The average recovery for the step during consistency batches was 91.2%.
- The quantities of materials required for the synthesis of CMP-Fmoc-glycyl-sialic acid varied with the quantity of Fmoc-glycyl-mannosamine generated from the purification step of the process. Exemplary quantities are as follows:
-
Fmoc-glycyl-mannosamine 11 g Cytidine-5′-triphosphate 15.8 g Manganese Chloride 9.5 g Pyruvate 26.4 g Sodium Hydroxide, 50% (w/w) 17 mL CMP-NAN-Synthetase 50,600 U (N. meningitidis) N-Acetyneuraminic acid Aldolase 1.19 g - The purification of CMP-Fmoc-glycyl-sialic acid (FSC) consists of two steps: the clarification of the reaction solution by 0.2 μm filtration and the reverse phase chromatographic purification of the FSC. To clarify the reaction, the solution was simply pumped through a Millipore Millipak 0.2 μm filter cartridge. Little backpressure was generated during this filtration. After the filtration was complete, the filter cake and filter were rinsed with 500 mL of purified water.
- The chromatographic purification was performed using a Biotage pre-packed C18 reverse phase column. The FSC was loaded in an aqueous solution. The FSC binds to the column, and the column was washed with six column volumes of purified water. The FSC was then eluted in 10% methanol in purified water.
- The purification step was performed using the Biotage chromatography system (Z-1405).
- The deprotection of CMP-Fmoc-Glycyl-Sialic Acid (FSC) occurred in a 10% methanol:water solution in a reaction with dimethylamine. Dimethylamine (40 wt % in water) is a solution that was added to the FSC C18 fraction pool. The Fmoc group was cleaved off, resulting in key intermediate, CMP-5′-Glycyl-Sialic Acid (GSC). The reaction was agitated at 25° C. for 75 minutes.
- The reaction was complete when the peak area ratio of GSC:FSC was greater than 20, as measured by HPLC. The resulting GSC solution was then rotovapped (30° C.) to about 35% of the original volume. The free Fmoc-derivative formed a white precipitate that needed to be removed from the GSC solution. This material was filtered through a Millipore Millipak-200 Filter Unit (0.22 μm). The filter was then rinsed with RO water. This GSC solution was stored at 4° C. or frozen at −20° C. until lyophilization. The average recovery for this step from the consistency batches was 86.1%.
- The table below lists the materials used during the deprotection of CMP-Fmoc-Glycyl-Sialic Acid (FSC) and filtration of CMP-5′-Glycyl-Sialic Acid (GSC).
-
Raw Materials for CMP-Glycyl-Sialic Acid (GSC) Deprotection and Filtration CMP-Fmoc-Glycyl-Sialic Acid Volume determined from P30 batch (FSC) C18 Fraction Pool record Dimethylamine (40 wt %) Volume calculated from P40 batch record See Section A, step 1RO H2O Volume calculated from P40 batch record See Section B, step 6 - Another method of producing and purifying CMP-SA-Glycine is provided below:
- Consistency batches were performed for 10K and 20K CMP-Sialic Acid-PEG after development of the synthesis and purification operations. These batches demonstrated that a reproducible process had been developed to produce high-purity CMP-SA-PEG with very low contaminant levels, suitable for the glycopegylation projects.
- 10K CMP-SA-PEG was produced at greater than 80% purity at overall process yields of approximately 60%, and 20K CMP-SA-PEG was produced at greater than 70% purity at overall process yields of approximately 50%. The products were low in endotoxin, bioburden, and protein, and NMR has shown that the balance of the material was nearly all mPEG-OH, a by-product of the synthesis process.
- CMP-SA-PEG (PSC) was produced in a reaction of CMP-Glycyl-Sialic Acid (GSC) with paranitrophenyl-carbomate-polyethylene glycol (pNP-PEG). The reaction conditions for the consistency batches of each size CMP-SA-PEG are summarized below.
-
CMP-SA-PEG Synthesis Conditions 10K PSC 20K PSC Raw Materials CMP-5′-Glycyl-Sialic Acid 1.0 g 0.5 g (GSC) 10K 20K 10K 20K pNP-PEG 24 g 10K pNP-PEG 24 g 20K pNP-PEG 10K 20K 10K 20K RO H2O 200 mL 100 mL Tetrahydrofuran (THF) 800 mL 400 mL unstabilized 10K 20K 10K 20K Reaction Conditions Length of Reaction 24 ± 1 hrs. 20 ± 1 hrs. pH adjustment to 17 ± 1 hrs. 17 ± 1 hrs. 8.9 ± 0.1 Additions of PEG 3 additions (90 min. 3 additions (90 min. between) each of between) each of 8 g of pNP-PEG 8 g of pNP-PEG - At the end of the reaction, a molar ratio of PSC to GSC (product to reactant) of greater than 7:1 is found for 10K PSC and greater than 5:1 for 20K PSC.
- The reaction solution were then rotovapped at <30° C. to less than 80% of its original volume to remove the THF. The remaining aqueous solution was then diluted to five times the original reaction volume (diluted to 5 L for 10K PSC and 2.5 L for 20K PSC) using RO water. The diluted solution was then adjusted to pH 9.5±0.1 with 1M NaOH and allowed to stir for at least 1 hr. This elevated pH caused breakdown of residual pNP-PEG to free paranitrophenol, carbon dioxide, and methoxyPEG-OH (mPEG-OH). After at least 1 hr. at the elevated pH, the solution is adjusted to pH 8.0±0.1 using 1M HCl.
- Both 10K and 20K PSC were then purified on a C4 reverse-phase chromatography column. The table below summarizes the purification conditions.
-
CMP-SA-PEG Purification Conditions 10K PSC 20K PSC Column Biotage C4 75 L (1 L bed Biotage C4 75 L (1 L bed volume, 7.5 cm diameter) volume, 7.5 cm diameter) Flowrate 100 mL/min (136 cm/hr) 100 mL/min (136 cm/hr) Wash 10 column volumes (CVs) 10 CVs 10% MeOH, 1mL Conditions 10% MeOH, 1 mL 1M 1M NaOH/L water NaOH/ L water Elution 4 CVs 40% MeOH, 1mL 4CVs 43% MeOH, 1 mL Conditions 1M NaOH/L water 1M NaOH/L water Regeneration 3CVs 100 % MeOH 3CVs 100% MeOH Conditions 2CVs 50% MeOH, 1 mL 2CVs 50% MeOH, 1 mL 1M NaOH/L water 1M NaOH/L water 2CVs 25% MeOH, 1 mL 2CVs 25% MeOH, 1 mL 1M NaOH/L water 1M NaOH/ L water 3CVs 100% RO water, 3CVs 100% RO water,1 mL 1M NaOH/ L water 1 mL 1M NaOH/L water - From the column, fractions during the elution that contain UV (27 nm) absorbance of at least 10% of the main peak absorbance were pooled, and the methanol from the pool was removed by rotovap.
- This concentrated fraction pool was then freeze dried on a Labconco flask-style freeze-drier. Final testing was performed on the resulting powder from this drying.
- The syntheses was performed in 2 L and 5 L sealed bottles, temperature controlled in a shaker incubator, SI-0017, B. Braun Certomat BS1.
- Chromatographic purification was performed on the Biotage Flash Chromatography System, Z-1405. Drying was performed on a lab-scale Labconco freeze dryer.
- Using the methods described above, two batches each of 10K and 20K PSC were synthesized, purified, and dried. The chart below summarizes the yield and recoveries from the synthesis and purification steps of the process. Note that the extent-of-reaction is noted as a ratio of peak areas of PSC to GSC from the HPLC assay. Quantitative yields from the syntheses are not available because the use of THF in the reaction disrupts the quantitative ability of the HPLC method.
-
CMP-SA-PEG Consistency Batch Yields and Recovery PSC Synthesis 10K PSC A 10K PSC B 20K PSC A 20K PSC B GSC content 60.66% 67.38% 67.38% 61.15% (wet weight) EOR 13.8:1 8.0:1 6.3:1 5.4:1 PSC Purification 10K PSC A 10K PSC B Average 20K PSC A 20K PSC B Average Quantity (g) 6.7 7.9 5.5 7.3 Yield PSC (g) 5.5 6.6 6.1 4.2 5.5 4.8 % Recovery 59.8% 64.0% 61.9% 42.4% 60.5% 51.4%
Results for the four batches are summarized below. -
PSC Consistency Batch Testing Results 10K PSC A 10K PSC B 20K PSC A 20K PSC B Identification (RP-LC) Match with Match with Match with Match with standard standard standard standard 10K PSC purity (g/100 g 82.62 83.01 76.30 74.56 of dry wt) CMP (g/100 g of dry wt) 0.02 0.02 0.02 0.02 Moisture content 0.60 0.49 0.52 0.48 (g/100 g of wet wt) Sodium % Na+ (g/100 g 0.45 0.50 0.22 0.14 of wet wt) Aldolase/CNS2 impurity None visible None visible None visible None visible Endotoxin (EU/mg of 0.011 0.007 0.0113 0.006 wet wt) Microbial limits testing *** *** *** *** Total aerobic counts <10 <10 <10 <10 (CFU/g of wet wt.) Total yeast and mold <10 <10 <10 <10 (CFU/g of wet wt.) - The HPLC traces of the 4 batches showed that the CMP-SA-PEG was >98% by peak area. CMP levels were also very low and much lower than seen in previous lots of PSC.
- A silver stain SDS-PAGE gel was run to analyze for residual CNS and aldolase. None was detectable in either the 10K or 20K PSC batches.
- Also, as requested in the materials specifications, 1H-NMR was performed on the batches.
- The 10K PSC samples from
Consistency Batch# 2 andConsistency Batch# 3, were submitted for 1H NMR analysis. The NMR spectra indicated that the PSC is reasonably clean, agreeing with the HPLC analysis. There are no observable resonances for sialic acid-PEG (expected at 2.22 & 1.83 ppm) or CMP (distinct resonance expected ˜8.11 ppm). A very small amount of PEG-carbamate-DMA (the product of the reaction of dimethylamine with pNP-PEG, 2.93 ppm) is seen inBatch # 2, while none is seen inBatch # 3. Although non-quantitative, the ratio of the methyl singlet (3.39 ppm) to the unique PSC resonances (˜8.00, 2.50, & 1.55 ppm) is ˜⅓, which would indicate relatively low levels of mPEG-OH impurities. - The 20K PSC samples from
Consistency Batch# 2 andConsistency Batch# 3, submitted for 1H NMR analysis showed that the PSC is also reasonably clean. There are no observable resonances for sialic acid-PEG (2.22 & 1.83 ppm), CMP (distinct resonance at ˜8.11 ppm), or PEG-carbamate-DMA (˜2.93 ppm). Although non-quantitative, the ratio of the methyl singlet (3.39 ppm) to the unique PSC resonances (˜8.00, 2.50, & 1.55 ppm) is ˜⅓, which would indicate relatively low levels of mPEG-OH impurities. - The current method discusses PEG coupling with a nucleotide sugar, such as CMP-SA-glycine. The nucleotide sugar-PEG product can be separated from the reaction mixture by first desalting the reaction mixture using dialysis. Membrane filtration (reverse osmosis, nanofiltration, etc.) or size exclusion techniques (i.e. polyacrylamide) resin, Sephadex resin, Sepharose resin) can be used next to further purify the mixture. After purification, the reaction mixture containing the nucleotide sugar-PEG product undergoes further purification involving ion exchange chromatography. For most reaction mixtures, a DEAE or Q-resin can be used to remove the unreacted PEG from the product. Q-Sepharose resin is currently preferred, although any polymer with a quaternary amine will also work. The Q-Sepharose resin can include ions such as —OH, Cl−, HCO3 −, CO3 2−, PO4 2−, SO3 −, Br−, BO3 2− or the like. The product can be loaded and eluted using known methods in the art such as changes in pH or ion strength (NaCl, KCl, etc.). The product may be eluted by a step-wise process or a gradient process. The nucleotide sugar-PEG product eluted from the column can be collected and desalted again using dialysis, membrane filtration, or size-exclusion techniques. The process would look similar to the figure below but can be varied dependent on processing results and desires.
- CMP-SA-glycine and the p-nitrophenyl-carbamate-mPEG are mixed together to obtain a reaction mixture comprising the product CMP-SA-glycine-PEG, as well as PEG, p-nitrophenyl-carboxylic acid, salts, CMP-SA-glycine, CMP and sialic acid (SA). The reaction mixture underwent membrane filtration wherein the membrane has MWCO of 200 (units)-300 kDa. CMP-SA-PEG and PEG were retained while p-nitrophenyl-carboxylic acid, salts, CMP-SA-glycine, CMP and SA passed through the membrane. The retained products underwent further purification with a Q-column or DEAE column in which PEG flows through the membrane and CMP-SA-PEG is retained and concentrated. At this step, the CMP-SA-PEG may be freeze-dried or spray dried. The CMP-SA-PEG underwent membrane filtration in which salts pass through and while CMP-SA-PEG was retained and concentrated. The membrane has a MWCO of 100 MW-3 kDa. This sample may be freeze dried, spray dried, or frozen.
- Any membrane size can be used from reverse osmosis (RO) pore sizes (molecular weight cut-offs) to microfiltration MWCOs depending on the separation desired. During membrane filtration, the pH can be varied between 2 and 12, more specifically between 5-10 and more specifically between 7-9 for CMP-SA-PEG.
- The membrane filtration step can be used to remove impurities by such techniques as diafiltration and can be used to concentrate the process streams.
-
Cytidine 5′-monophospho N-acetylneuraminic acid (CMP-NAN) is enzymatically synthesized fromcytidine 5′-triphosphate (CTP) and N-acetylneuraminic acid (NAN) in the presence of E. coli-expressed CMP-NAN Synthetase (CNS).FIG. 1 illustrates the reaction. -
- CTP and NAN were incubated in a reactor with CNS and MnCl2. Using a small excess of CTP, the reaction proceeded to almost 100% conversion of NAN to CMP-NAN in under two hours.
- The primary by-product of the synthesis reaction was the formation of a manganese phosphate precipitate. Approximate 30% of the final volume of the reaction consisted of this heavy, brown precipitate. In order to remove this precipitate, the solution was filtered. The cake that formed on the filter was washed with water, and the final solution was then filtered to remove any fine particulates that were not held back by the first filtration.
- In order to remove proteins introduced from the CNS, the CMP-NAN solution was permeated through a hollow fiber filter. The membrane successfully retains proteins from the enzyme preparation while allowing the passage of CMP-NAN.
- Residual salts were then removed from the solution by concentration and diafiltration of the CMP-NAN using a nanofiltration membrane. This membrane retains CMP-NAN during concentration and diafiltration while allowing passage of salts that remain in solution, primarily Tris HCl (the buffer from the enzyme preparation) and NaCl (from the synthesis reaction).
- The concentrated CMP-NAN was subsequently decolorized by passing the solution over a pre-packed column of activated carbon. The color is likely from manganese oxide formed during the addition of sodium hydroxide to control the pH during synthesis.
- Finally, endotoxin was removed using a charged media depth filter, and then the CMP-NAN solution was filtered through a 0.2 μm filter and aliquotted into sterile containers.
- An aliquot was spray dried and tested. The CMP-NAN purity was 83.3%. The remainder of the material was stored as a frozen liquid at −20° C. A portion of the frozen bulk was lyophilized. This material was analyzed and released per specification RM0300 with a purity of 82%.
-
Cytidine 5′-monophospho N-Acetylneuraminic acid (CMP-NAN) was synthesized in a temperature and pH controlled vessel fromCytidine 5′-triphosphate (CTP) and sialic acid (NAN) using CMP-NAN Synthetase (CNS) in the presence of MnCl2. The CNS is a recombinant protein cloned from N. meningitidis and was expressed in JM109 E. coli cells. The reaction proceeded for approximately 1.5 hours with sufficient agitation to keep the precipitate from settling. The resultant CMP-NAN solution was chilled to <20° C., clarified by filtration, and transferred into a mobile tank. - The clarified CMP-NAN solution was concentrated 10 times using a hollow fiber filtration unit and diafiltered with 5 volumes of USP purified water. The CMP-NAN was transferred to a mobile tank and the pH was adjusted. This solution was concentrated 10 times and diafiltered with 6 volumes of USP purified water using a nanofiltration system.
- Decolorization was achieved by running the CMP-NAN through an activated carbon column and endotoxin was removed using a CUNO Zeta Plus 60ZA filter.
- The purified CMP-NAN solution was then aseptically filtered (CUNO LifeASSURE 0.2 μm) and aliquoted into MITOS Sugar Nucleotide Bags and stored at −20° C.
- Three consistency runs were performed at the 20 L scale before the Engineering Trial took place. The tables below summarize the yields and recoveries and some of the analysis results.
-
Consistency Runs Summary Data Synthesis Theoretical Overall Process Purification Batch Yield Yield Yield Recovery 1 1339 g 98.1% 607.8 g 45.4% 2 1264 g 92.6% 793.6 g 62.7% 3 1373 g 100.6% 952.0 g 69.3% -
Analysis Results from Consistency Runs Chemical Component Batch # 1 Batch # 2Batch # 3CMP-NAN 75.0% 77.5% 82.5% CMP 5.6% 5.0% 5.0 % CDP 0% 0.5% 0% NAN 9.4% 8.5% 4.6% Moisture 7.5% 7.3% 7.7% Mass Balance 97.5% 98.8% 99.8% Other Impurities 0.2% 0.9% 0.4% PO4 -
Property Requirement Result Appearance White to White powder off-white powder Identification Compares Compares to reference to reference spectra spectra Moisture Content ≦5 % moisture 5% Purity ≧80% CMP-NAN 82% Percent CMP, CDP <10% CMP, CDP, 5% and CTP and CTP Manganese by ICP Report Results 120 ppm Sodium % by ICP Report Results 7.5% Heavy Metals Report Results <20 ppm (as Pb) Host Cell Protein Report Results Non detected Total Aerobic ≦10 cfu/mg <1 cfu/mg Count Yeast and Mold ≦10 cfu/mg <1 cfu/mg Endotoxin ≦0.5 EU/mg .00165 EU/mg - This example describes the preparation and purification of CMP-SA-
PEG 30 kDa (compound 3, below). The mPEG-p-nitrophenyl carbonate-30 kDa (compound 1, below) was reacted with sodium CMP-SA-Glycine (compound 2, below) in a mixture of THF/Water. The crude product was desalted by Tangential Flow Filtration (TFF), purified by Q Sepharose chromatography (IEX), and again desalted by TFF to provide 13.5 g of CMP-SA-PEG-30 kDa product (compound 3, below). Reprocessing of mixed IEX fractions afforded an additional 8.2 grams of 3 (overall yield 72.3%). -
-
Materials Name Unit Q Sepharose Big Beads Double 1K Pellicon-2 “MINI” PBLC 1K Regen. filters Cellulose Membrane; Screen Type: V; 0.1 m2 CMP-SA-Glycine (2) Powder Dimethylamine salt 30K mPEG-p-nitrophenyl Powder carbonate (1) (obtained from NOF America) NaCl Powder NaOH 1.0 N Tris-Glycine gel, 4-20% 1.0 mm, 10 wells SeeBlue- plus2 Standard 12 proteins Tris- Glycine Running Buffer 10× concentrated Tris-Glycine Sample Buffer 50 mL solution BaCl2 Powder Iodine Solution N/10 Sodium Bicarbonate Powder - CMP-SA-Glycine (dimethyl amine salt form, 1.35 g, 2.0 mmole) was dissolved in 20 mL H2O, and the pH was adjusted to 10.5 with 0.1 N NaOH (˜20 mL). The basic solution was degassed under reduced pressure (vacuum 30 min), frozen and lyophilized to dryness. The resulting sodium CMP-SA-Glycine was redissolved in water (80 mL). The pH was measured and found to be 8.5. The pH was adjusted to 7.8 by addition of 0.2 N NaH2PO4 (<1.0 mL) and the resulting solution was diluted with THF (200 mL). The mPEG-p-nitrophenyl carbonate-30 kDa (30.0 g, 1 mmol) was added to the CMP-SA-Glycine solution in small portions over 3 hr at room temperature. The reaction mixture was stirred at room temperature for 43 hrs. The THF was then removed by rotary evaporation at reduced pressure without heating (water bath temperature at or below 30° C.). The aqueous residue (80 mL) was diluted with water to 600 mL, and adjusted to pH 9.5 with 1.0 N NaOH (about 1.0 mL). The basic solution was stirred at room temperature for 1 hour to hydrolyze any unreacted mPEG-p-nitrophenyl carbonate-30 kDa, and then purified by Tangential Flow Filtration (TFF), Q-Sepharose chromatography (IEX), and then TFF as described below. The final purified fractions were freeze-dried to afford 21.7 g (72.3%) of a white solid (3).
- A Watson-Marlow peristaltic pump (505S) was connected through Tygon tubing (¼″ ID) to a Millipore Pellicon-2 Mini Holder equipped with two
Millipore 1K Pellicon 2 “MINI” filter (PLAC-V 1K Regenerated Cellulose Membrane; Screen Type: V; 0.1 m2) (FIG. 1 ). The crude aqueous product solution (500 mL, pH 9.5) from above was transferred to a 1000 mL bottle immersed in an ice bath, equipped with a conductivity meter and a pH meter. The product solution was fed onto the Pellicon Mini filter through Tygon tubing (¼″ ID) for diafiltration with a pump speed of 90 rpm (Cross flow rate: 430 mL/min; Flux rate: 20 mL/min;Pressure 13 psi). The retentate solution was returned to the bottle containing the bulk chilled product solution (PharMed tubing, ¼″ ID) which was maintained at a constant volume (600 mL) by addition of cold DI water (4° C.). The permeate solution was collected in 2 L fractions. The pH and conductivity values of the retentate/product solution were measured and recorded over time as shown in the table below. The pH of the retentate/product solution was maintained above pH 7.5 by the dropwise addition of 1.0 N NaOH, as needed. The retentate/product solution was diafiltered until the conductivity dropped below 0.8 mS, and then the retentate/product solution was allowed to concentrate to a volume of 500 mL. The concentrated retentate was then purified by anion exchange chromatography as described below. The permeate fractions were checked for product break-through by SDS-PAGE as described below: -
Time Conductivity pH Total (min.) (mS) Value Vol (mL) 0 1.93 9.58 600 30 1.59 9.50 600 60 1.10 9.07 600 90 0.899 8.69 600 120 0.818 8.47 600 150 0.743 8.35 600 180 0.755 8.56 500 - Samples of the TFF permeate fractions (0.5 mL) were concentrated to dryness under a stream of N2 gas, resuspended in 10 μL water and mixed with 10 μL Tris-Gly SDS-PAGE sample buffer and loaded onto 4-20% polyacrylamide Tris-Gly SDS-PAGE gels. See Blue Plus2 protein standard was also loaded as a marker. Gels were run at a constant voltage of 125 V for 1 hr 50 min. After electrophoresis, the gels were washed with water (100 mL) for 10 min, and then incubated with a 5% barium chloride aqueous solution for 10 min. Iodine solution (0.1 N, 4.0 mL) was added to visualize any mPEG present. The staining process was stopped by washing the gels with water. The proteins used as a standard were a mix of myosin (250 kDa), phosphorylase (148 kDa), BSA (98 kDa, glutamic dehydrogenase (64 kDa), alcohol dehydrogenase (50 kDa), carbonic anhydrase (36 kDa), lysozyme (22 kDa), aprotinin (6 kDa), and insulin B-chain (4 kDa). The gels were visualized and scanned with an HP Scanjet 7400C, and the image of the gel was optimized with the HP Precision Scan Program.
- The Q Sepharose Big Beads (4.0 L) were treated with 1.0 M NaOH (8.0 L), and then with saturated aqueous sodium bicarbonate (8.0 L) to generate the bicarbonate form of the resin. The newly generated resin was packed in a 17×18 cm (ID) column which was connected to an HPLC system equipped with a UV (274 nm) and an ELS detector (Evaporation temp: 120° C.; Nebulizer temp: 90° C.; Gas flow rate: 1.85 SLM). The TFF retentate from above was slowly loaded on the Q column (60 mL/min.). The column was then washed with solvent A (cold DI water, 8.0 L) at a flow rate of 125 mL/min until ELS detection indicated that all non-binding impurities (mPEG-OH) had been washed from the column. The product was then eluted with a gradient of 90% Solvent A/10% Solvent B (1.0 N NaCl) to 20% Solvent A/80% Solvent B over 100 min, and then to 100% B for 5 minutes at a flow rate of 125 ml/min. Product-containing fractions were collected (detected by UV, 274 nM) as shown in
FIG. 2 .Fraction 1 was desalted by TFF as described below.Fraction 2 was reprocessed (data not shown). - The first fraction (Fraction 1) of the ion-exchange-purified product was desalted using the TFF procedure described above. Upon reaching a constant conductivity, the retentate/product solution was allowed to concentrate to 400 mL. The final retentate (400 mL) was freeze-dried to yield 13.5 g of white solid.
-
Time Conductivity pH Total (min.) (mS) Value Vol (mL) 0 23.9 7.79 600 30 23.1 7.81 600 120 10.8 7.91 600 150 8.46 7.94 600 180 5.59 8.14 600 210 2.68 8.30 600 240 1.14 8.46 600 270 0.706 8.68 600 300 0.489 8.81 600 330 0.403 8.90 500 360 0.389 8.53 400 - Cytidine-
monophospho 5′-N-acetylneuraminic acid (CMP-NAN) is enzymatically synthesized fromcytidine 5′-triphosphate (CTP) and N-acetylneuraminic acid (NAN) in the presence of E. coli-expressed CMP-NAN synthetase (CNS). - The reactants (CTP and NAN) are incubated (30° C., pH 8.5-9.5) in a reactor (20 or 40 L Synthesis Reactor, jacketed temperature control is <10° C. to 30° C., manual or automatic pH control is 8.5-9.5, and agitated) with CNS and manganese chloride (MnCl2). Using a small excess of CTP, the reaction proceeds to almost 100% conversion of NAN to the CMP-NAN product in under two hours. When the reaction is complete, the solution is chilled to <10.0° C. Once chilled the process stream is filtered through a 0.5 μm membrane bag filter (CUNO Polynet bag filter with CUNO bag filter housing and pressure-fed (nitrogen) filtration).
- In order to remove proteins introduced by the enzyme preparation, the process stream is next permeated through a 10K hollow fiber membrane with a tangential flow filtration (TFF) skid (Amersham Bioscience (formerly AG Technology) 10K MWCO hollow fiber cartridge, temperature control is <10° C., and inlet, outlet and permeate pressure indication is 0-60 psig). The 10K membrane successfully retains proteins from the enzyme preparation while allowing for passage of the CMP-NAN. The result is a protein-free CMP-NAN solution.
- Residual salts are then removed from solution by concentration and diafiltration of the CMP-NAN using a nanofiltration membrane with a nanofiltration TFF skid (Millipore Nanomax-50 Helicon RO4 spiral wound membrane cartridge installed on a Millipore Prolab II skid, temperature control is <10° C. and pressure indication is 0-600 psig). This membrane retains CMP-NAN during concentration and diafiltration while allowing passage of salts that remain in solution from the synthesis reaction and hollow fiber filtration.
- This concentrated CMP-NAN stream is subsequently de-colored by passing the solution over a pre-packed column of activated carbon attached to a Biotage chromatography system (75M activated chromatography column and Biotage skid). The activated carbon efficiently removes the color from the solution in one or two passes of the CMP-NAN stream.
- The process is completed by two filtrations. First, the CMP-NAN solution is filtered using a charged media depth filter (CUNO Zeta Plus 60ZA, 1 ft2). Second, the solution is sent through a final 0.2 μm filter that is pre-packaged and sterilized with a bag manifold system (provided by Mitos Technologies) for final filtration and storage. The product can be stored as a frozen solution or dried as a white powder for refrigerated storage.
- All publications, patents and patent applications mentioned in this specification are herein incorporated by reference into the specification to the same extent as if each individual publication, patent or patent application was specifically and individually indicated to be incorporated herein by reference.
Claims (30)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US12/092,563 US20090048440A1 (en) | 2005-11-03 | 2006-11-03 | Nucleotide Sugar Purification Using Membranes |
Applications Claiming Priority (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US73397505P | 2005-11-03 | 2005-11-03 | |
| US79628106P | 2006-04-28 | 2006-04-28 | |
| US74675406P | 2006-05-08 | 2006-05-08 | |
| US82353806P | 2006-08-25 | 2006-08-25 | |
| US82924206P | 2006-10-12 | 2006-10-12 | |
| US12/092,563 US20090048440A1 (en) | 2005-11-03 | 2006-11-03 | Nucleotide Sugar Purification Using Membranes |
| PCT/US2006/043048 WO2007056191A2 (en) | 2005-11-03 | 2006-11-03 | Nucleotide sugar purification using membranes |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2006/043048 A-371-Of-International WO2007056191A2 (en) | 2005-11-03 | 2006-11-03 | Nucleotide sugar purification using membranes |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/215,439 Division US8841439B2 (en) | 2005-11-03 | 2011-08-23 | Nucleotide sugar purification using membranes |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20090048440A1 true US20090048440A1 (en) | 2009-02-19 |
Family
ID=38023848
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/092,563 Abandoned US20090048440A1 (en) | 2005-11-03 | 2006-11-03 | Nucleotide Sugar Purification Using Membranes |
| US13/215,439 Expired - Fee Related US8841439B2 (en) | 2005-11-03 | 2011-08-23 | Nucleotide sugar purification using membranes |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US13/215,439 Expired - Fee Related US8841439B2 (en) | 2005-11-03 | 2011-08-23 | Nucleotide sugar purification using membranes |
Country Status (2)
| Country | Link |
|---|---|
| US (2) | US20090048440A1 (en) |
| WO (1) | WO2007056191A2 (en) |
Cited By (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20040063911A1 (en) * | 2001-10-10 | 2004-04-01 | Neose Technologies, Inc. | Protein remodeling methods and proteins/peptides produced by the methods |
| US20060111279A1 (en) * | 2003-11-24 | 2006-05-25 | Neose Technologies, Inc. | Glycopegylated erythropoietin |
| US20060287224A1 (en) * | 2003-11-24 | 2006-12-21 | Neose Technologies, Inc. | Glycopegylated erythropoietin formulations |
| US20070032405A1 (en) * | 2003-03-14 | 2007-02-08 | Neose Technologies, Inc. | Branched water-soluble polymers and their conjugates |
| US20070059275A1 (en) * | 2003-07-25 | 2007-03-15 | Defrees Shawn | Antibody toxin conjugates |
| US20070254836A1 (en) * | 2003-12-03 | 2007-11-01 | Defrees Shawn | Glycopegylated Granulocyte Colony Stimulating Factor |
| US20080050772A1 (en) * | 2001-10-10 | 2008-02-28 | Neose Technologies, Inc. | Granulocyte colony stimulating factor: remodeling and glycoconjugation of G-CSF |
| US20080102083A1 (en) * | 2003-05-09 | 2008-05-01 | Neose Technologies, Inc. | Compositions and Methods for the Preparation of Human Growth Hormone Glycosylation Mutants |
| US20080146782A1 (en) * | 2006-10-04 | 2008-06-19 | Neose Technologies, Inc. | Glycerol linked pegylated sugars and glycopeptides |
| US20080176790A1 (en) * | 2004-10-29 | 2008-07-24 | Defrees Shawn | Remodeling and Glycopegylation of Fibroblast Growth Factor (Fgf) |
| US20080242846A1 (en) * | 2004-01-08 | 2008-10-02 | Neose Technologies, Inc. | O-linked glycosylation of peptides |
| US20080255040A1 (en) * | 2006-07-21 | 2008-10-16 | Neose Technologies, Inc. | Glycosylation of peptides via o-linked glycosylation sequences |
| US20080300175A1 (en) * | 2003-11-24 | 2008-12-04 | Neose Technologies, Inc. | Glycopegylated erythropoietin |
| US20080305992A1 (en) * | 2003-11-24 | 2008-12-11 | Neose Technologies, Inc. | Glycopegylated erythropoietin |
| US20090000924A1 (en) * | 2007-06-29 | 2009-01-01 | Harley-Davidson Motor Company Group, Inc. | Integrated ignition and key switch |
| US20090028822A1 (en) * | 2004-09-10 | 2009-01-29 | Neose Technologies, Inc. | Glycopegylated Interferon Alpha |
| US20090143292A1 (en) * | 2007-08-29 | 2009-06-04 | Neose Technologies, Inc. | Liquid Formulation of G-CSF Conjugate |
| US20090203579A1 (en) * | 2005-01-10 | 2009-08-13 | Defrees Shawn | Glycopegylated Granulocyte Colony Stimulating Factor |
| US20090227504A1 (en) * | 2002-06-21 | 2009-09-10 | Novo Nordisk A/S | Pegylated Factor VII Glycoforms |
| US20090292110A1 (en) * | 2004-07-23 | 2009-11-26 | Defrees Shawn | Enzymatic modification of glycopeptides |
| US20100009902A1 (en) * | 2005-01-06 | 2010-01-14 | Neose Technologies, Inc. | Glycoconjugation Using Saccharyl Fragments |
| US20100048456A1 (en) * | 2003-04-09 | 2010-02-25 | Novo Nordisk A/S | Glycopegylation methods and proteins/peptides produced by the methods |
| US20100075375A1 (en) * | 2006-10-03 | 2010-03-25 | Novo Nordisk A/S | Methods for the purification of polypeptide conjugates |
| US20100120666A1 (en) * | 2007-04-03 | 2010-05-13 | Biogenerix Ag | Methods of treatment using glycopegylated g-csf |
| US20100174059A1 (en) * | 2007-06-12 | 2010-07-08 | Novo Nordisk A/S | Process for the production of nucleotide sugars |
| US20100261872A1 (en) * | 2001-10-10 | 2010-10-14 | Neose Technologies, Inc. | Factor VIII: remodeling and glycoconjugation of factor VIII |
| US20100286067A1 (en) * | 2008-01-08 | 2010-11-11 | Biogenerix Ag | Glycoconjugation of polypeptides using oligosaccharyltransferases |
| US20110003744A1 (en) * | 2005-05-25 | 2011-01-06 | Novo Nordisk A/S | Glycopegylated Erythropoietin Formulations |
| US7956032B2 (en) | 2003-12-03 | 2011-06-07 | Novo Nordisk A/S | Glycopegylated granulocyte colony stimulating factor |
| US20110177029A1 (en) * | 2007-06-04 | 2011-07-21 | Novo Nordisk A/S | O-linked glycosylation using n-acetylglucosaminyl transferases |
| US8008252B2 (en) | 2001-10-10 | 2011-08-30 | Novo Nordisk A/S | Factor VII: remodeling and glycoconjugation of Factor VII |
| US20120121781A1 (en) * | 2009-03-17 | 2012-05-17 | Separation Technologies Investments Limited | Whey or raw milk demineralisation and fractionation |
| US20120121788A1 (en) * | 2009-03-17 | 2012-05-17 | Separation Technologies Investments Limited | Isolation and purification of components of whey |
| WO2012151300A2 (en) | 2011-05-02 | 2012-11-08 | Verasonics, Inc. | Enhanced ultrasound image formation using qualified regions of overlapping transmit beams |
| US8404809B2 (en) | 2005-05-25 | 2013-03-26 | Novo Nordisk A/S | Glycopegylated factor IX |
| US8632770B2 (en) | 2003-12-03 | 2014-01-21 | Novo Nordisk A/S | Glycopegylated factor IX |
| US20140039237A1 (en) * | 2012-08-06 | 2014-02-06 | Ming-Hsin Li | Medication dispensing device |
| US8716240B2 (en) | 2001-10-10 | 2014-05-06 | Novo Nordisk A/S | Erythropoietin: remodeling and glycoconjugation of erythropoietin |
| US8791070B2 (en) | 2003-04-09 | 2014-07-29 | Novo Nordisk A/S | Glycopegylated factor IX |
| US8791066B2 (en) | 2004-07-13 | 2014-07-29 | Novo Nordisk A/S | Branched PEG remodeling and glycosylation of glucagon-like peptide-1 [GLP-1] |
| US8841439B2 (en) | 2005-11-03 | 2014-09-23 | Novo Nordisk A/S | Nucleotide sugar purification using membranes |
| US8911967B2 (en) | 2005-08-19 | 2014-12-16 | Novo Nordisk A/S | One pot desialylation and glycopegylation of therapeutic peptides |
| US9150848B2 (en) | 2008-02-27 | 2015-10-06 | Novo Nordisk A/S | Conjugated factor VIII molecules |
| US9187546B2 (en) | 2005-04-08 | 2015-11-17 | Novo Nordisk A/S | Compositions and methods for the preparation of protease resistant human growth hormone glycosylation mutants |
| KR101938604B1 (en) | 2011-02-18 | 2019-01-15 | 도레이 카부시키가이샤 | Method for producing sugar solution |
| CN110272461A (en) * | 2019-06-29 | 2019-09-24 | 赤峰蒙广生物科技有限公司 | A method of purifying beta-thymidine from fermentation liquid |
Families Citing this family (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN105153313A (en) | 2010-02-16 | 2015-12-16 | 诺沃—诺迪斯克有限公司 | Factor viii fusion protein |
| CN102812039B (en) | 2010-02-16 | 2016-01-20 | 诺沃—诺迪斯克有限公司 | Conjugated protein |
| EP2536753B1 (en) | 2010-02-16 | 2017-12-20 | Novo Nordisk A/S | Factor viii molecules with reduced vwf binding |
| US20130183280A1 (en) | 2010-07-15 | 2013-07-18 | Novo Nordisk A/S | Stabilized factor viii variants |
| BR112013005872A2 (en) | 2010-09-22 | 2019-09-24 | Alios Biopharma Inc | compounds, pharmaceutical composition and their uses |
| US9862893B2 (en) | 2011-05-12 | 2018-01-09 | Virent, Inc. | Process for purifying lignocellulosic feedstocks |
| DE102011101995A1 (en) | 2011-05-19 | 2012-11-22 | Sartorius Stedim Biotech Gmbh | Process for separating a mixture of a protein and its reaction product with a polyalkylene glycol |
| CA2860234A1 (en) | 2011-12-22 | 2013-06-27 | Alios Biopharma, Inc. | Substituted phosphorothioate nucleotide analogs |
| US8916538B2 (en) | 2012-03-21 | 2014-12-23 | Vertex Pharmaceuticals Incorporated | Solid forms of a thiophosphoramidate nucleotide prodrug |
| HK1203356A1 (en) | 2012-03-22 | 2015-10-30 | 艾丽奥斯生物制药有限公司 | Pharmaceutical combinations comprising a thionucleotide analog |
| AR101060A1 (en) * | 2014-02-12 | 2016-11-23 | Novo Nordisk As | FVIII CONJUGATES |
| AR099340A1 (en) * | 2014-02-12 | 2016-07-13 | Novo Nordisk As | CONJUGATES OF THE COAGULATION FACTOR IX |
| ES2991621T3 (en) | 2016-03-07 | 2024-12-04 | Glycom As | Separation of oligosaccharides from fermentation broth |
| EP3490567A4 (en) | 2016-07-28 | 2020-03-18 | Fonterra Co-Operative Group Limited | Dairy product and process |
| WO2018115309A1 (en) * | 2016-12-22 | 2018-06-28 | Novo Nordisk A/S | Preparation of sugar-nucleotides |
| EP3645159A4 (en) | 2017-06-30 | 2021-03-31 | Circulomics Inc. | Size selection purification using a thermoplastic silica nanomaterial |
| AU2019264788B8 (en) * | 2018-05-07 | 2023-07-27 | Chr. Hansen HMO GmbH | A simple method for the purification of lacto-N-Neotetraose (LNnT) From Carbohydrates Obtained By Microbial Fermentation |
| ES2933959T3 (en) | 2018-06-01 | 2023-02-15 | Chr Hansen Hmo Gmbh | A simple method for the purification of a sialyl-lactose |
| EP4329923A1 (en) * | 2021-04-30 | 2024-03-06 | Hydranautics | Concentration and diafiltration of oligonucleotides |
| KR20240125623A (en) * | 2021-12-20 | 2024-08-19 | 89바이오 인코포레이티드 | Chemical synthesis of cytidine-5'-monophospho-N-glycyl-sialic acid |
Citations (89)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4088538A (en) * | 1975-05-30 | 1978-05-09 | Battelle Memorial Institute | Reversibly precipitable immobilized enzyme complex and a method for its use |
| US4385260A (en) * | 1975-09-09 | 1983-05-24 | Beckman Instruments, Inc. | Bargraph display |
| US4438253A (en) * | 1982-11-12 | 1984-03-20 | American Cyanamid Company | Poly(glycolic acid)/poly(alkylene glycol) block copolymers and method of manufacturing the same |
| US4496689A (en) * | 1983-12-27 | 1985-01-29 | Miles Laboratories, Inc. | Covalently attached complex of alpha-1-proteinase inhibitor with a water soluble polymer |
| US4565653A (en) * | 1984-03-30 | 1986-01-21 | Pfizer Inc. | Acyltripeptide immunostimulants |
| US4806595A (en) * | 1985-08-12 | 1989-02-21 | Koken Co., Ltd. | Method of preparing antithrombogenic medical materials |
| US4826945A (en) * | 1987-06-09 | 1989-05-02 | Yissum Research Development Company | Biodegradable polymeric materials based on polyether glycols, processes for the preparation thereof and surgical articles made therefrom |
| US4925796A (en) * | 1986-03-07 | 1990-05-15 | Massachusetts Institute Of Technology | Method for enhancing glycoprotein stability |
| US5104651A (en) * | 1988-12-16 | 1992-04-14 | Amgen Inc. | Stabilized hydrophobic protein formulations of g-csf |
| US5122614A (en) * | 1989-04-19 | 1992-06-16 | Enzon, Inc. | Active carbonates of polyalkylene oxides for modification of polypeptides |
| US5182107A (en) * | 1989-09-07 | 1993-01-26 | Alkermes, Inc. | Transferrin receptor specific antibody-neuropharmaceutical or diagnostic agent conjugates |
| US5194376A (en) * | 1989-02-28 | 1993-03-16 | University Of Ottawa | Baculovirus expression system capable of producing foreign gene proteins at high levels |
| US5202413A (en) * | 1993-02-16 | 1993-04-13 | E. I. Du Pont De Nemours And Company | Alternating (ABA)N polylactide block copolymers |
| US5219564A (en) * | 1990-07-06 | 1993-06-15 | Enzon, Inc. | Poly(alkylene oxide) amino acid copolymers and drug carriers and charged copolymers based thereon |
| US5281698A (en) * | 1991-07-23 | 1994-01-25 | Cetus Oncology Corporation | Preparation of an activated polymer ester for protein conjugation |
| US5324844A (en) * | 1989-04-19 | 1994-06-28 | Enzon, Inc. | Active carbonates of polyalkylene oxides for modification of polypeptides |
| US5324663A (en) * | 1990-02-14 | 1994-06-28 | The Regents Of The University Of Michigan | Methods and products for the synthesis of oligosaccharide structures on glycoproteins, glycolipids, or as free molecules, and for the isolation of cloned genetic sequences that determine these structures |
| US5405753A (en) * | 1990-03-26 | 1995-04-11 | Brossmer; Reinhard | CMP-activated, fluorescing sialic acids, as well as processes for their preparation |
| US5410016A (en) * | 1990-10-15 | 1995-04-25 | Board Of Regents, The University Of Texas System | Photopolymerizable biodegradable hydrogels as tissue contacting materials and controlled-release carriers |
| US5492841A (en) * | 1994-02-18 | 1996-02-20 | E. I. Du Pont De Nemours And Company | Quaternary ammonium immunogenic conjugates and immunoassay reagents |
| US5605793A (en) * | 1994-02-17 | 1997-02-25 | Affymax Technologies N.V. | Methods for in vitro recombination |
| US5614184A (en) * | 1992-07-28 | 1997-03-25 | New England Deaconess Hospital | Recombinant human erythropoietin mutants and therapeutic methods employing them |
| US5621039A (en) * | 1993-06-08 | 1997-04-15 | Hallahan; Terrence W. | Factor IX- polymeric conjugates |
| US5629384A (en) * | 1994-05-17 | 1997-05-13 | Consiglio Nazionale Delle Ricerche | Polymers of N-acryloylmorpholine activated at one end and conjugates with bioactive materials and surfaces |
| US5705367A (en) * | 1994-09-26 | 1998-01-06 | The Rockefeller University | Glycosyltransferases for biosynthesis of oligosaccharides, and genes encoding them |
| US5716812A (en) * | 1995-12-12 | 1998-02-10 | The University Of British Columbia | Methods and compositions for synthesis of oligosaccharides, and the products formed thereby |
| US5728554A (en) * | 1995-04-11 | 1998-03-17 | Cytel Corporation | Enzymatic synthesis of glycosidic linkages |
| US5858752A (en) * | 1995-06-07 | 1999-01-12 | The General Hospital Corporation | Fucosyltransferase genes and uses thereof |
| US5858751A (en) * | 1992-03-09 | 1999-01-12 | The Regents Of The University Of California | Compositions and methods for producing sialyltransferases |
| US5876980A (en) * | 1995-04-11 | 1999-03-02 | Cytel Corporation | Enzymatic synthesis of oligosaccharides |
| US6010999A (en) * | 1990-05-04 | 2000-01-04 | American Cyanamid Company | Stabilization of fibroblast growth factors by modification of cysteine residues |
| US6015555A (en) * | 1995-05-19 | 2000-01-18 | Alkermes, Inc. | Transferrin receptor specific antibody-neuropharmaceutical or diagnostic agent conjugates |
| US6030815A (en) * | 1995-04-11 | 2000-02-29 | Neose Technologies, Inc. | Enzymatic synthesis of oligosaccharides |
| US6037452A (en) * | 1992-04-10 | 2000-03-14 | Alpha Therapeutic Corporation | Poly(alkylene oxide)-Factor VIII or Factor IX conjugate |
| US6057292A (en) * | 1995-09-21 | 2000-05-02 | Genentech, Inc. | Method for inhibiting growth hormone action |
| US6183738B1 (en) * | 1997-05-12 | 2001-02-06 | Phoenix Pharamacologics, Inc. | Modified arginine deiminase |
| US6348558B1 (en) * | 1999-12-10 | 2002-02-19 | Shearwater Corporation | Hydrolytically degradable polymers and hydrogels made therefrom |
| US6362254B2 (en) * | 1998-03-12 | 2002-03-26 | Shearwater Corporation | Poly(ethylene glycol) derivatives with proximal reactive groups |
| US20020037841A1 (en) * | 2000-05-15 | 2002-03-28 | Apollon Papadimitriou | Erythropoietin composition |
| US6376604B2 (en) * | 1999-12-22 | 2002-04-23 | Shearwater Corporation | Method for the preparation of 1-benzotriazolylcarbonate esters of poly(ethylene glycol) |
| US20030027257A1 (en) * | 1997-08-21 | 2003-02-06 | University Technologies International, Inc. | Sequences for improving the efficiency of secretion of non-secreted protein from mammalian and insect cells |
| US20030040037A1 (en) * | 2000-05-12 | 2003-02-27 | Neose Technologies, Inc. | In vitro modification of glycosylation patterns of recombinant glycopeptides |
| US6531121B2 (en) * | 2000-12-29 | 2003-03-11 | The Kenneth S. Warren Institute, Inc. | Protection and enhancement of erythropoietin-responsive cells, tissues and organs |
| US6555660B2 (en) * | 2000-01-10 | 2003-04-29 | Maxygen Holdings Ltd. | G-CSF conjugates |
| US6555346B1 (en) * | 1997-12-18 | 2003-04-29 | Stichting Instituut Voor Dierhouderij En Diergezondheid | Protein expression in baculovirus vector expression systems |
| US6693183B2 (en) * | 1996-03-08 | 2004-02-17 | The Regents Of The University Of Michigan | MURINE α (1,3) FUCOSYLTRANSFERASE FUC-TVII, DNA ENCODING THE SAME, METHOD FOR PREPARING THE SAME, ANTIBODIES RECOGNIZING THE SAME, IMMUNOASSAYS FOR DETECTING THE SAME, PLASMIDS CONTAINING SUCH DNA, AND CELLS CONTAINING SUCH A PLASMID |
| US6692931B1 (en) * | 1998-11-16 | 2004-02-17 | Werner Reutter | Recombinant glycoproteins, method for the production thereof, medicaments containing said glycoproteins and use thereof |
| US20040063911A1 (en) * | 2001-10-10 | 2004-04-01 | Neose Technologies, Inc. | Protein remodeling methods and proteins/peptides produced by the methods |
| US6716626B1 (en) * | 1999-11-18 | 2004-04-06 | Chiron Corporation | Human FGF-21 nucleic acids |
| US20040102607A1 (en) * | 1998-03-25 | 2004-05-27 | Danishefsky Samuel J. | Trimeric antigenic O-linked glycopeptide conjugates, methods of preparation and uses thereof |
| US20050026266A1 (en) * | 2002-11-08 | 2005-02-03 | Glycozym Aps | Methods to identify agents modulating functions of polypeptide galnac-transferases, pharmaceutical compositions comprising such agents and the use of such agents for preparing medicaments |
| US20050031584A1 (en) * | 2001-10-10 | 2005-02-10 | Neose Technologies, Inc. | Interleukin-2:remodeling and glycoconjugation of interleukin-2 |
| US20050064540A1 (en) * | 2002-11-27 | 2005-03-24 | Defrees Shawn Ph.D | Glycoprotein remodeling using endoglycanases |
| US20050100982A1 (en) * | 2001-10-10 | 2005-05-12 | Neose Technologies, Inc. | Factor IX: remodeling and glycoconjugation of factor IX |
| US20050106658A1 (en) * | 2001-10-10 | 2005-05-19 | Defrees Shawn | Remodeling and glycoconjugation of peptides |
| US20060024286A1 (en) * | 2004-08-02 | 2006-02-02 | Paul Glidden | Variants of tRNA synthetase fragments and uses thereof |
| US20060030521A1 (en) * | 2001-11-28 | 2006-02-09 | Neose Technologies, Inc. | Remodeling and glycoconjugation of peptides |
| US20060035224A1 (en) * | 2002-03-21 | 2006-02-16 | Johansen Jack T | Purification methods for oligonucleotides and their analogs |
| US20060111279A1 (en) * | 2003-11-24 | 2006-05-25 | Neose Technologies, Inc. | Glycopegylated erythropoietin |
| US7157277B2 (en) * | 2001-11-28 | 2007-01-02 | Neose Technologies, Inc. | Factor VIII remodeling and glycoconjugation of Factor VIII |
| US20070014759A1 (en) * | 2003-12-03 | 2007-01-18 | Neose Technologies, Inc. | Glycopegylated granulocyte colony stimulating factor |
| US20070027068A1 (en) * | 2001-10-10 | 2007-02-01 | Defrees Shawn | Erythropoietin: remodeling and glycoconjugation of erythropoietin |
| US20070026485A1 (en) * | 2003-04-09 | 2007-02-01 | Neose Technologies, Inc. | Glycopegylation methods and proteins/peptides produced by the methods |
| US7173003B2 (en) * | 2001-10-10 | 2007-02-06 | Neose Technologies, Inc. | Granulocyte colony stimulating factor: remodeling and glycoconjugation of G-CSF |
| US20070032405A1 (en) * | 2003-03-14 | 2007-02-08 | Neose Technologies, Inc. | Branched water-soluble polymers and their conjugates |
| US20070059275A1 (en) * | 2003-07-25 | 2007-03-15 | Defrees Shawn | Antibody toxin conjugates |
| US7214660B2 (en) * | 2001-10-10 | 2007-05-08 | Neose Technologies, Inc. | Erythropoietin: remodeling and glycoconjugation of erythropoietin |
| US20070105755A1 (en) * | 2005-10-26 | 2007-05-10 | Neose Technologies, Inc. | One pot desialylation and glycopegylation of therapeutic peptides |
| US20080015142A1 (en) * | 2003-12-03 | 2008-01-17 | Defrees Shawn | Glycopegylated Follicle Stimulating Hormone |
| US7338933B2 (en) * | 2004-01-08 | 2008-03-04 | Neose Technologies, Inc. | O-linked glycosylation of peptides |
| US20080102083A1 (en) * | 2003-05-09 | 2008-05-01 | Neose Technologies, Inc. | Compositions and Methods for the Preparation of Human Growth Hormone Glycosylation Mutants |
| US7368108B2 (en) * | 2001-11-28 | 2008-05-06 | Neose Technologies, Inc. | Glycopeptide remodeling using amidases |
| US20080108557A1 (en) * | 2004-09-29 | 2008-05-08 | Novo Nordisk Healthcare A/G | Modified Proteins |
| US20090028822A1 (en) * | 2004-09-10 | 2009-01-29 | Neose Technologies, Inc. | Glycopegylated Interferon Alpha |
| US20090055942A1 (en) * | 2005-09-14 | 2009-02-26 | Novo Nordisk Healthcare A/G | Human Coagulation Factor VII Polypeptides |
| US20090054623A1 (en) * | 2004-12-17 | 2009-02-26 | Neose Technologies, Inc. | Lipo-Conjugation of Peptides |
| US20090053167A1 (en) * | 2007-05-14 | 2009-02-26 | Neose Technologies, Inc. | C-, S- and N-glycosylation of peptides |
| US20090093399A1 (en) * | 2001-10-10 | 2009-04-09 | Neose Technologies, Inc. | Glycopegylation methods and proteins/peptides produced by the methods |
| US20090124544A1 (en) * | 2005-04-08 | 2009-05-14 | Neose Technologies ,Inc. A Delaware Corporation | Compositions and methods for the preparation of protease resistant human growth hormone glycosylation mutants |
| US20100009902A1 (en) * | 2005-01-06 | 2010-01-14 | Neose Technologies, Inc. | Glycoconjugation Using Saccharyl Fragments |
| US20100015684A1 (en) * | 2001-10-10 | 2010-01-21 | Neose Technologies, Inc. | Factor vii: remodeling and glycoconjugation of factor vii |
| US20100029555A1 (en) * | 2006-08-11 | 2010-02-04 | Bio-Ker S.r.l | G-csf site-specific mono-conjugates |
| US20100028939A1 (en) * | 2003-08-08 | 2010-02-04 | Novo Nordisk Healthcare A/G | Use of Galactose Oxidase for Selective Chemical Conjugation of Protractor Molecules to Proteins of Therapeutic Interest |
| US20100035299A1 (en) * | 2006-10-03 | 2010-02-11 | Novo Nordisk A/S | Methods for the purification of polypeptide conjugates |
| US20100041872A1 (en) * | 2006-10-04 | 2010-02-18 | Defrees Shawn | Glycerol linked pegylated sugars and glycopeptides |
| US20100056428A1 (en) * | 2006-09-01 | 2010-03-04 | Novo Nordisk Health Care Ag | Modified proteins |
| US20100081791A1 (en) * | 2005-05-25 | 2010-04-01 | Novo Nordisk A/S | Glycopegylated factor ix |
| US7691603B2 (en) * | 2003-04-09 | 2010-04-06 | Novo Nordisk A/S | Intracellular formation of peptide conjugates |
| US20100120666A1 (en) * | 2007-04-03 | 2010-05-13 | Biogenerix Ag | Methods of treatment using glycopegylated g-csf |
Family Cites Families (304)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1479268A (en) | 1973-07-05 | 1977-07-13 | Beecham Group Ltd | Pharmaceutical compositions |
| US4179337A (en) | 1973-07-20 | 1979-12-18 | Davis Frank F | Non-immunogenic polypeptides |
| GB1451798A (en) | 1973-08-02 | 1976-10-06 | Ici Ltd | Prostanoic acid derivatives |
| US4414147A (en) | 1981-04-17 | 1983-11-08 | Massachusetts Institute Of Technology | Methods of decreasing the hydrophobicity of fibroblast and other interferons |
| JPS57206622A (en) | 1981-06-10 | 1982-12-18 | Ajinomoto Co Inc | Blood substitute |
| US4451566A (en) | 1981-12-04 | 1984-05-29 | Spencer Donald B | Methods and apparatus for enzymatically producing ethanol |
| DE3308806A1 (en) | 1983-03-12 | 1984-09-13 | Basf Ag, 6700 Ludwigshafen | FUNGICIDAL AGENTS, SUBSTITUTED GLUCOPYRANOSYLAMINE AND METHOD FOR CONTROLLING FUNGI |
| JPS59172425A (en) | 1983-03-18 | 1984-09-29 | Nippon Chemiphar Co Ltd | Novel blood coagulation factor derivative, its preparation and blood coagulation promoting agent containing the same |
| US4879236A (en) | 1984-05-16 | 1989-11-07 | The Texas A&M University System | Method for producing a recombinant baculovirus expression vector |
| US4675414A (en) | 1985-03-08 | 1987-06-23 | The United States Of America As Represented By The Secretary Of The Navy | Maleimidomethyl-carbonate polyethers |
| GR860984B (en) | 1985-04-17 | 1986-08-18 | Zymogenetics Inc | Expression of factor vii and ix activities in mammalian cells |
| DE3676670D1 (en) | 1985-06-26 | 1991-02-07 | Cetus Corp | SOLUBILIZATION OF PROTEINS FOR PHARMACEUTICAL COMPOSITIONS BY POLYMER CONJUGATION. |
| US5206344A (en) | 1985-06-26 | 1993-04-27 | Cetus Oncology Corporation | Interleukin-2 muteins and polymer conjugation thereof |
| SE451849B (en) | 1985-12-11 | 1987-11-02 | Svenska Sockerfabriks Ab | VIEW TO SYNTHETIZE GYCLOSIDIC BINDINGS AND USE OF THIS RECEIVED PRODUCTS |
| IT1213029B (en) | 1986-01-30 | 1989-12-07 | Bracco Ind Chimica Spa | PARAMAGNETIC METAL ION CHELATES. |
| US4767702A (en) | 1986-02-06 | 1988-08-30 | Cohenford Menashi A | Paper strip assay for neisseria species |
| WO1987005330A1 (en) | 1986-03-07 | 1987-09-11 | Michel Louis Eugene Bergh | Method for enhancing glycoprotein stability |
| US5272066A (en) | 1986-03-07 | 1993-12-21 | Massachusetts Institute Of Technology | Synthetic method for enhancing glycoprotein stability |
| US4902505A (en) | 1986-07-30 | 1990-02-20 | Alkermes | Chimeric peptides for neuropeptide delivery through the blood-brain barrier |
| US5153265A (en) | 1988-01-20 | 1992-10-06 | Cetus Corporation | Conjugation of polymer to colony stimulating factor-1 |
| US4847325A (en) | 1988-01-20 | 1989-07-11 | Cetus Corporation | Conjugation of polymer to colony stimulating factor-1 |
| GB8810808D0 (en) | 1988-05-06 | 1988-06-08 | Wellcome Found | Vectors |
| US5169933A (en) | 1988-08-15 | 1992-12-08 | Neorx Corporation | Covalently-linked complexes and methods for enhanced cytotoxicity and imaging |
| US5874261A (en) | 1988-09-02 | 1999-02-23 | The Trustees Of The University Of Pennsylvania | Method for the purification of glycosyltransferases |
| US5218092A (en) | 1988-09-29 | 1993-06-08 | Kyowa Hakko Kogyo Co., Ltd. | Modified granulocyte-colony stimulating factor polypeptide with added carbohydrate chains |
| US5047335A (en) | 1988-12-21 | 1991-09-10 | The Regents Of The University Of Calif. | Process for controlling intracellular glycosylation of proteins |
| US6166183A (en) | 1992-11-30 | 2000-12-26 | Kirin-Amgen, Inc. | Chemically-modified G-CSF |
| CA2006596C (en) | 1988-12-22 | 2000-09-05 | Rika Ishikawa | Chemically-modified g-csf |
| EP0449968B1 (en) | 1988-12-23 | 1999-02-24 | Genentech, Inc. | Process for the preparation of human dnase |
| AU634517B2 (en) | 1989-01-19 | 1993-02-25 | Pharmacia & Upjohn Company | Somatotropin analogs |
| EP0456642A1 (en) | 1989-01-31 | 1991-11-21 | The Upjohn Company | Somatotropin analogs |
| US5059535A (en) | 1989-04-12 | 1991-10-22 | Chembiomed, Ltd. | Process for the separation and purification of sialyl transferases |
| WO1990013540A1 (en) | 1989-04-19 | 1990-11-15 | Enzon, Inc. | Active carbonates of polyalkylene oxides for modification of polypeptides |
| US5166322A (en) | 1989-04-21 | 1992-11-24 | Genetics Institute | Cysteine added variants of interleukin-3 and chemical modifications thereof |
| US5342940A (en) | 1989-05-27 | 1994-08-30 | Sumitomo Pharmaceuticals Company, Limited | Polyethylene glycol derivatives, process for preparing the same |
| US5154924A (en) | 1989-09-07 | 1992-10-13 | Alkermes, Inc. | Transferrin receptor specific antibody-neuropharmaceutical agent conjugates |
| US5672683A (en) | 1989-09-07 | 1997-09-30 | Alkermes, Inc. | Transferrin neuropharmaceutical agent fusion protein |
| US5977307A (en) | 1989-09-07 | 1999-11-02 | Alkermes, Inc. | Transferrin receptor specific ligand-neuropharmaceutical agent fusion proteins |
| US5527527A (en) | 1989-09-07 | 1996-06-18 | Alkermes, Inc. | Transferrin receptor specific antibody-neuropharmaceutical agent conjugates |
| US5032519A (en) | 1989-10-24 | 1991-07-16 | The Regents Of The Univ. Of California | Method for producing secretable glycosyltransferases and other Golgi processing enzymes |
| US5312808A (en) | 1989-11-22 | 1994-05-17 | Enzon, Inc. | Fractionation of polyalkylene oxide-conjugated hemoglobin solutions |
| IL96477A0 (en) | 1989-12-01 | 1991-08-16 | Amgen Inc | Megakaryocyte production |
| SE465222C5 (en) | 1989-12-15 | 1998-02-10 | Pharmacia & Upjohn Ab | A recombinant human factor VIII derivative and process for its preparation |
| US5595900A (en) | 1990-02-14 | 1997-01-21 | The Regents Of The University Of Michigan | Methods and products for the synthesis of oligosaccharide structures on glycoproteins, glycolipids, or as free molecules, and for the isolation of cloned genetic sequences that determine these structures |
| EP0481038B1 (en) | 1990-04-16 | 2002-10-02 | The Trustees Of The University Of Pennsylvania | Saccharide compositions, methods and apparatus for their synthesis |
| US5583042A (en) | 1990-04-16 | 1996-12-10 | Neose Pharmaceuticals, Inc. | Apparatus for the synthesis of saccharide compositions |
| GB9107846D0 (en) | 1990-04-30 | 1991-05-29 | Ici Plc | Polypeptides |
| US5399345A (en) | 1990-05-08 | 1995-03-21 | Boehringer Mannheim, Gmbh | Muteins of the granulocyte colony stimulating factor |
| CU22302A1 (en) | 1990-09-07 | 1995-01-31 | Cigb | Codifying nucleotidic sequence for a protein of the external membrane of neisseria meningitidis and the use of that protein in preparing vaccines. |
| CA2084514A1 (en) | 1990-07-10 | 1992-01-11 | Guenther Adolf | O-glycosylated ifn-alpha |
| DE4028800A1 (en) | 1990-09-11 | 1992-03-12 | Behringwerke Ag | GENETIC SIALYLATION OF GLYCOPROTEINS |
| US5529914A (en) | 1990-10-15 | 1996-06-25 | The Board Of Regents The Univeristy Of Texas System | Gels for encapsulation of biological materials |
| CA2073511A1 (en) | 1990-11-14 | 1992-05-29 | Matthew R. Callstrom | Conjugates of poly(vinylsaccharide) with proteins for the stabilization of proteins |
| US5164374A (en) | 1990-12-17 | 1992-11-17 | Monsanto Company | Use of oligosaccharides for treatment of arthritis |
| US5861374A (en) | 1991-02-28 | 1999-01-19 | Novo Nordisk A/S | Modified Factor VII |
| US5788965A (en) | 1991-02-28 | 1998-08-04 | Novo Nordisk A/S | Modified factor VII |
| ATE273393T1 (en) | 1991-02-28 | 2004-08-15 | Zymogenetics Inc | MODIFIED FACTOR-VII |
| US5833982A (en) | 1991-02-28 | 1998-11-10 | Zymogenetics, Inc. | Modified factor VII |
| AU1676992A (en) | 1991-03-18 | 1992-10-21 | Enzon, Inc. | Hydrazine containing conjugates of polypeptides and glycopolypeptides with polymers |
| ATE193551T1 (en) | 1991-03-18 | 2000-06-15 | Scripps Research Inst | OLIGOSACCHARIDES AS ENZYME SUBSTRATES AND INHIBITORS: METHODS AND COMPOSITIONS |
| US5278299A (en) | 1991-03-18 | 1994-01-11 | Scripps Clinic And Research Foundation | Method and composition for synthesizing sialylated glycosyl compounds |
| US5212075A (en) | 1991-04-15 | 1993-05-18 | The Regents Of The University Of California | Compositions and methods for introducing effectors to pathogens and cells |
| DE69231467T2 (en) | 1991-05-10 | 2001-01-25 | Genentech, Inc. | SELECTION OF AGONISTS AND ANTAGONISTS OF LIGANDS |
| GB2256197B (en) | 1991-05-31 | 1995-11-22 | Ciba Geigy Ag | Yeast as host for expression of heterologous glycosyl transferase enzymes |
| US5374655A (en) | 1991-06-10 | 1994-12-20 | Alberta Research Council | Methods for the synthesis of monofucosylated oligosaccharides terminating in di-N-acetyllactosaminyl structures |
| US5352670A (en) * | 1991-06-10 | 1994-10-04 | Alberta Research Council | Methods for the enzymatic synthesis of alpha-sialylated oligosaccharide glycosides |
| KR950014915B1 (en) | 1991-06-19 | 1995-12-18 | 주식회사녹십자 | Asialoglycoprotein-conjugated compounds |
| EP0863154A1 (en) | 1991-10-12 | 1998-09-09 | The Regents Of The University Of California | Use of thiol redox proteins for reducing protein intramolecular disulfide bonds, for improving the quality of cereal products, dough and baked goods |
| US6319695B1 (en) | 1991-10-15 | 2001-11-20 | The Scripps Research Insitute | Production of fucosylated carbohydrates by enzymatic fucosylation synthesis of sugar nucleotides; and in situ regeneration of GDP-fucose |
| DE69226197T2 (en) | 1991-11-08 | 1999-02-11 | Somatogen, Inc., Boulder | HEMOGLOBINE AS A MEDICINE DELIVERY SYSTEM |
| US5384249A (en) | 1991-12-17 | 1995-01-24 | Kyowa Hakko Kogyo Co., Ltd. | α2→3 sialyltransferase |
| IT1260468B (en) | 1992-01-29 | 1996-04-09 | METHOD FOR MAINTAINING THE ACTIVITY OF PROTEOLYTIC ENZYMES MODIFIED WITH POLYETHYLENGLYCOL | |
| CZ220494A3 (en) | 1992-03-09 | 1995-03-15 | Univ California | Composition and methods of identification and synthesis of siallyltransferases |
| US5962294A (en) | 1992-03-09 | 1999-10-05 | The Regents Of The University Of California | Compositions and methods for the identification and synthesis of sialyltransferases |
| AU3937593A (en) | 1992-03-25 | 1993-10-21 | New York University | Trans-sialidase and methods of use and making thereof |
| JPH0686684A (en) | 1992-05-26 | 1994-03-29 | Monsanto Co | Synthesis of sialo-conjugate |
| EP0664710A4 (en) | 1992-08-07 | 1998-09-30 | Progenics Pharm Inc | NON-PEPTIDYL MOIETY-CONJUGATED CD4-GAMMA2 AND CD4-IgG2 IMMUNOCONJUGATES, AND USES THEREOF. |
| AU5006993A (en) | 1992-08-21 | 1994-03-15 | Enzon, Inc. | Novel attachment of polyalkylene oxides to bio-effecting substances |
| JP3979678B2 (en) | 1992-08-24 | 2007-09-19 | サントリー株式会社 | Novel glycosyltransferase, gene encoding the same, and method for producing the enzyme |
| AU5098193A (en) | 1992-09-01 | 1994-03-29 | Berlex Laboratories, Inc. | Glycolation of glycosylated macromolecules |
| US5308460A (en) | 1992-10-30 | 1994-05-03 | Glyko, Incorporated | Rapid synthesis and analysis of carbohydrates |
| US6361977B1 (en) | 1992-11-24 | 2002-03-26 | S. Christopher Bauer | Methods of using multivariant IL-3 hematopoiesis fusion protein |
| FI935485L (en) | 1992-12-09 | 1994-06-10 | Ortho Pharma Corp | PEG-hydrazone and PEG-oxime bond-forming reagents and their protein derivatives |
| NZ250375A (en) | 1992-12-09 | 1995-07-26 | Ortho Pharma Corp | Peg hydrazone and peg oxime linkage forming reagents and protein derivatives |
| CA2110543A1 (en) | 1992-12-09 | 1994-06-10 | David E. Wright | Peg hydrazone and peg oxime linkage forming reagents and protein derivatives thereof |
| WO1994015625A1 (en) | 1993-01-15 | 1994-07-21 | Enzon, Inc. | Factor viii - polymeric conjugates |
| US5349001A (en) | 1993-01-19 | 1994-09-20 | Enzon, Inc. | Cyclic imide thione activated polyalkylene oxides |
| US5321095A (en) | 1993-02-02 | 1994-06-14 | Enzon, Inc. | Azlactone activated polyalkylene oxides |
| US6180134B1 (en) | 1993-03-23 | 2001-01-30 | Sequus Pharmaceuticals, Inc. | Enhanced ciruclation effector composition and method |
| WO1994023021A1 (en) | 1993-03-29 | 1994-10-13 | Kyowa Hakko Kogyo Co., Ltd. | α-1,3-FUCOSYLTRANSFERASE |
| US5409817A (en) | 1993-05-04 | 1995-04-25 | Cytel, Inc. | Use of trans-sialidase and sialyltransferase for synthesis of sialylα2→3βgalactosides |
| US5374541A (en) | 1993-05-04 | 1994-12-20 | The Scripps Research Institute | Combined use of β-galactosidase and sialyltransferase coupled with in situ regeneration of CMP-sialic acid for one pot synthesis of oligosaccharides |
| WO1994026906A2 (en) | 1993-05-14 | 1994-11-24 | The Upjohn Company | CLONED DNA ENCODING A UDP-GALNAc:POLYPEPTIDE,N-ACETYLGALACTOS AMINYLTRANSFERASE |
| CA2162478A1 (en) | 1993-05-14 | 1994-11-24 | Shawn A. Defrees | Sialyl lex analogues as inhibitors of cellular adhesion |
| ES2169074T3 (en) | 1993-05-21 | 2002-07-01 | Novo Nordisk Healthcare Ag | FACTOR VII MODIFIED TO INHIBIT THE VASCULAR RESENOSIS OR THE TANK DEPOSIT. |
| WO1994028024A1 (en) | 1993-06-01 | 1994-12-08 | Enzon, Inc. | Carbohydrate-modified polymer conjugates with erythropoietic activity |
| FI960173L (en) | 1993-07-15 | 1996-03-15 | Neose Pharm Inc | Method for synthesizing saccharide compositions |
| DE4325317C2 (en) | 1993-07-29 | 1998-05-20 | Univ Dresden Tech | Process for the radioactive labeling of immunoglobulins |
| ZA946122B (en) | 1993-08-17 | 1995-03-20 | Amgen Inc | Erythropoietin analogs |
| JPH0770195A (en) | 1993-08-23 | 1995-03-14 | Yutaka Mizushima | Sugar-modified interferon |
| US6485930B1 (en) | 1993-09-15 | 2002-11-26 | The Scripps Research Institute | Mannosyl transfer with regeneration of GDP-mannose |
| CN1057775C (en) | 1993-09-22 | 2000-10-25 | 味之素株式会社 | Peptide with antithrombotic activity and its preparation method |
| US5874075A (en) | 1993-10-06 | 1999-02-23 | Amgen Inc. | Stable protein: phospholipid compositions and methods |
| US5643575A (en) | 1993-10-27 | 1997-07-01 | Enzon, Inc. | Non-antigenic branched polymer conjugates |
| US5919455A (en) | 1993-10-27 | 1999-07-06 | Enzon, Inc. | Non-antigenic branched polymer conjugates |
| US5446090A (en) | 1993-11-12 | 1995-08-29 | Shearwater Polymers, Inc. | Isolatable, water soluble, and hydrolytically stable active sulfones of poly(ethylene glycol) and related polymers for modification of surfaces and molecules |
| US5443953A (en) | 1993-12-08 | 1995-08-22 | Immunomedics, Inc. | Preparation and use of immunoconjugates |
| US5369017A (en) | 1994-02-04 | 1994-11-29 | The Scripps Research Institute | Process for solid phase glycopeptide synthesis |
| JP3516272B2 (en) | 1994-02-10 | 2004-04-05 | 株式会社成和化成 | Cosmetic base material |
| US5837458A (en) | 1994-02-17 | 1998-11-17 | Maxygen, Inc. | Methods and compositions for cellular and metabolic engineering |
| MX9504664A (en) | 1994-03-07 | 1997-05-31 | Dow Chemical Co | CONJUGATES OF BIOACTIVE AND / OR DIRECTED DENDRIMEROS. |
| IL113010A (en) | 1994-03-31 | 1999-10-28 | Pharmacia & Upjohn Ab | Pharmaceutical formulation comprising factor viii with an activity of at least 500iu/ml and an enhancer for improved subcutaneous intramuscular or intradermal administration |
| US5432059A (en) | 1994-04-01 | 1995-07-11 | Specialty Laboratories, Inc. | Assay for glycosylation deficiency disorders |
| US5646113A (en) | 1994-04-07 | 1997-07-08 | Genentech, Inc. | Treatment of partial growth hormone insensitivity syndrome |
| WO1996010089A1 (en) | 1994-09-29 | 1996-04-04 | Ajinomoto Co., Inc. | Modification of peptide and protein |
| US5824784A (en) | 1994-10-12 | 1998-10-20 | Amgen Inc. | N-terminally chemically modified protein compositions and methods |
| US5834251A (en) | 1994-12-30 | 1998-11-10 | Alko Group Ltd. | Methods of modifying carbohydrate moieties |
| US5932462A (en) | 1995-01-10 | 1999-08-03 | Shearwater Polymers, Inc. | Multiarmed, monofunctional, polymer for coupling to molecules and surfaces |
| SE9501285L (en) | 1995-04-06 | 1996-10-07 | Johanna Ljung | Process for producing biologically active proteins |
| DK0820520T3 (en) | 1995-04-11 | 2002-12-02 | Neose Technologies Inc | Enhanced enzymatic synthesis of oliogosaccharides |
| US5922577A (en) | 1995-04-11 | 1999-07-13 | Cytel Corporation | Enzymatic synthesis of glycosidic linkages |
| US5695760A (en) | 1995-04-24 | 1997-12-09 | Boehringer Inglehiem Pharmaceuticals, Inc. | Modified anti-ICAM-1 antibodies and their use in the treatment of inflammation |
| EP0833663A4 (en) | 1995-05-15 | 1999-04-07 | Constantin A Bona | Carbohydrate-mediated coupling of peptides to immunoglobulins |
| US5824864A (en) | 1995-05-25 | 1998-10-20 | Pioneer Hi-Bred International, Inc. | Maize gene and protein for insect control |
| US6127153A (en) | 1995-06-07 | 2000-10-03 | Neose Technologies, Inc. | Method of transferring at least two saccharide units with a polyglycosyltransferase, a polyglycosyltransferase and gene encoding a polyglycosyltransferase |
| WO1996040731A1 (en) | 1995-06-07 | 1996-12-19 | Mount Sinai School Of Medicine Of The City University Of New York | Pegylated modified proteins |
| US6251864B1 (en) | 1995-06-07 | 2001-06-26 | Glaxo Group Limited | Peptides and compounds that bind to a receptor |
| US5672662A (en) | 1995-07-07 | 1997-09-30 | Shearwater Polymers, Inc. | Poly(ethylene glycol) and related polymers monosubstituted with propionic or butanoic acids and functional derivatives thereof for biotechnical applications |
| MX9800482A (en) | 1995-07-27 | 1998-04-30 | Cytec Tech Corp | Synthetic cationic polymers as promoters for asa sizing. |
| US5770420A (en) | 1995-09-08 | 1998-06-23 | The Regents Of The University Of Michigan | Methods and products for the synthesis of oligosaccharide structures on glycoproteins, glycolipids, or as free molecules, and for the isolation of cloned genetic sequences that determine these structures |
| SE9503380D0 (en) | 1995-09-29 | 1995-09-29 | Pharmacia Ab | Protein derivatives |
| CA2165041C (en) | 1995-12-12 | 2005-07-05 | The University Of British Columbia | Methods and compositions for synthesis of oligosaccharides, and the products formed thereby |
| WO1997021822A2 (en) | 1995-12-12 | 1997-06-19 | The University Of British Columbia | Methods and compositions for synthesis of oligosaccharides using mutant glycosidase enzymes |
| JP3065925B2 (en) | 1996-01-30 | 2000-07-17 | 日清製油株式会社 | Active oxygen species scavenger and anti-fading agent |
| ATE341344T1 (en) | 1996-08-02 | 2006-10-15 | Ortho Mcneil Pharm Inc | POLYPEPTIDES WITH SINGLE COVALENTLY BONDED N-TERMINAL WATER SOLUBLE POLYMER |
| US6495365B1 (en) | 1996-08-13 | 2002-12-17 | Fujisawa Pharmaceutical Co., Ltd. | Hematopoietic stem cell proliferating agents |
| US20020064546A1 (en) | 1996-09-13 | 2002-05-30 | J. Milton Harris | Degradable poly(ethylene glycol) hydrogels with controlled half-life and precursors therefor |
| NZ335203A (en) * | 1996-10-10 | 2000-10-27 | Neose Technologies Inc | Carbohydrate, purification using ultrafiltration, reverse osmosis and nanofiltration |
| DK0964690T3 (en) | 1996-10-15 | 2003-10-20 | Liposome Co Inc | Peptide-lipid conjugates, liposomes and liposomal drug administration |
| US6117651A (en) | 1996-11-08 | 2000-09-12 | Neose Technologies, Inc. | Expression vectors |
| AU736993B2 (en) | 1997-01-16 | 2001-08-09 | Novo Nordisk A/S | Practical in vitro sialylation of recombinant glycoproteins |
| DE69800640T2 (en) | 1997-01-29 | 2001-07-05 | Polymasc Pharmaceuticals Plc, London | PEGYLATION PROCEDURE |
| DE19709787A1 (en) | 1997-03-11 | 1998-09-17 | Bayer Ag | Oligosaccaride and their derivatives as well as a chemo-enzymatic process for their production |
| US5945314A (en) | 1997-03-31 | 1999-08-31 | Abbott Laboratories | Process for synthesizing oligosaccharides |
| JP2002505574A (en) | 1997-04-30 | 2002-02-19 | エンゾン,インコーポレイテッド | Polyalkylene oxide-modified single-chain polypeptides |
| JPH10307356A (en) | 1997-05-08 | 1998-11-17 | Konica Corp | Silver halide emulsion and silver halide photographic sensitive material using the same |
| US6075134A (en) | 1997-05-15 | 2000-06-13 | The Regents Of The University Of California | Glycoconjugates and methods |
| AU8005098A (en) | 1997-06-06 | 1998-12-21 | Governors Of The University Of Alberta, The | Alpha1,3-fucosyltransferase of helicobacter pylori |
| ES2244066T3 (en) | 1997-06-24 | 2005-12-01 | Genentech, Inc. | PROCEDURE AND COMPOSITIONS OF GALACTOSILATED GLICOPROTEINS. |
| WO1999000150A2 (en) | 1997-06-27 | 1999-01-07 | Regents Of The University Of California | Drug targeting of a peptide radiopharmaceutical through the primate blood-brain barrier in vivo with a monoclonal antibody to the human insulin receptor |
| WO1999013063A1 (en) | 1997-09-09 | 1999-03-18 | Nycomed Imaging As | Factor vii fragments and analogs thereof and their use in the treatment of blood clottng disorders |
| WO1999022764A1 (en) | 1997-10-31 | 1999-05-14 | Genentech, Inc. | Methods and compositions comprising glycoprotein glycoforms |
| MXPA00005376A (en) | 1997-12-01 | 2002-07-02 | Neose Technologies Inc | Enzymatic synthesis of gangliosides. |
| CA2316834C (en) | 1998-01-07 | 2006-01-03 | Shearwater Polymers, Inc. | Degradable heterobifunctional poly(ethylene glycol) acrylates and gels and conjugates derived therefrom |
| AU2559799A (en) | 1998-01-22 | 1999-08-09 | Genentech Inc. | Antibody fragment-polymer conjugates and humanized anti-il-8 monoclonal antibodies and uses of same |
| PT1071700E (en) | 1998-04-20 | 2010-04-23 | Glycart Biotechnology Ag | Glycosylation engineering of antibodies for improving antibody-dependent cellular cytotoxicity |
| WO1999055376A1 (en) | 1998-04-28 | 1999-11-04 | Applied Research Systems Ars Holding N.V. | Peg-lhrh analog conjugates |
| US20030166525A1 (en) | 1998-07-23 | 2003-09-04 | Hoffmann James Arthur | FSH Formulation |
| SG2008070138A (en) | 1998-10-16 | 2017-08-30 | Biogen Ma Inc | Polymer conjugates of interferon beta- 1a and their uses |
| US7304150B1 (en) | 1998-10-23 | 2007-12-04 | Amgen Inc. | Methods and compositions for the prevention and treatment of anemia |
| CN1325443A (en) | 1998-10-30 | 2001-12-05 | 诺沃奇梅兹有限公司 | Glycosylated proteins with reduced allergenicity |
| ATE408006T1 (en) | 1998-11-13 | 2008-09-15 | Henrick Clausen | UDP-GALACTOSE: BETA-I(N)-ACETYL-GLUCOSAMINE BETA1, 3 GALACTOSYL TRANSFERASES, BETA3GAL-T5 |
| JP2002530087A (en) | 1998-11-18 | 2002-09-17 | ネオーズ テクノロジーズ, インコーポレイテッド | Inexpensive production of oligosaccharides |
| US6465220B1 (en) | 1998-12-21 | 2002-10-15 | Glycozym Aps | Glycosylation using GalNac-T4 transferase |
| KR100689212B1 (en) | 1999-01-29 | 2007-03-09 | 암겐 인코포레이티드 | BCS combination |
| US6503744B1 (en) | 1999-02-01 | 2003-01-07 | National Research Council Of Canada | Campylobacter glycosyltransferases for biosynthesis of gangliosides and ganglioside mimics |
| US6949372B2 (en) | 1999-03-02 | 2005-09-27 | The Johns Hopkins University | Engineering intracellular sialylation pathways |
| ATE279531T1 (en) | 1999-04-22 | 2004-10-15 | Astrazeneca Ab | TEST TO DETECT THE ACTIVITY OF PHOSPHO-N-ACETYLMURAMYL PENTAPEPTIDE TRANSLOCASE |
| JO2291B1 (en) | 1999-07-02 | 2005-09-12 | اف . هوفمان لاروش ايه جي | Erythopintin derivatives |
| US6261805B1 (en) | 1999-07-15 | 2001-07-17 | Boyce Thompson Institute For Plant Research, Inc. | Sialyiation of N-linked glycoproteins in the baculovirus expression vector system |
| WO2001005434A2 (en) | 1999-07-20 | 2001-01-25 | Amgen Inc. | Hyaluronic acid-protein conjugates |
| US6534300B1 (en) | 1999-09-14 | 2003-03-18 | Genzyme Glycobiology Research Institute, Inc. | Methods for producing highly phosphorylated lysosomal hydrolases |
| JP5209161B2 (en) | 1999-12-02 | 2013-06-12 | ザイモジェネティクス, インコーポレイテッド | Method for targeting cells expressing glandular fibroblast growth receptor-3 or -2 |
| AU2223401A (en) | 1999-12-24 | 2001-07-09 | Kyowa Hakko Kogyo Co. Ltd. | Branched polyalkylene glycols |
| IL150314A0 (en) | 1999-12-30 | 2002-12-01 | Maxygen Aps | Improved lysosomal enzymes and lysosomal enzyme activators |
| US6646110B2 (en) | 2000-01-10 | 2003-11-11 | Maxygen Holdings Ltd. | G-CSF polypeptides and conjugates |
| CZ20022727A3 (en) | 2000-01-10 | 2002-11-13 | Maxygen Holdings Ltd | Polypeptide conjugate, process of its preparation and pharmaceutical preparation |
| EP1982732A3 (en) | 2000-02-11 | 2011-06-08 | Bayer HealthCare LLC | Factor VII or VIIA-like conjugates |
| AU2001231531A1 (en) | 2000-02-11 | 2001-08-20 | Maxygen Aps | Follicle stimulating hormones |
| AU2001232337A1 (en) | 2000-02-18 | 2001-08-27 | Kanagawa Academy Of Science And Technology | Pharmaceutical composition, reagent and method for intracerebral delivery of pharmaceutically active ingredient or labeling substance |
| US20010041683A1 (en) | 2000-03-09 | 2001-11-15 | Schmitz Harold H. | Cocoa sphingolipids, cocoa extracts containing sphingolipids and methods of making and using same |
| AU2001281463A1 (en) | 2000-03-16 | 2001-09-24 | The Regents Of The University Of California | Chemoselective ligation |
| US6586398B1 (en) | 2000-04-07 | 2003-07-01 | Amgen, Inc. | Chemically modified novel erythropoietin stimulating protein compositions and methods |
| EP1282693B1 (en) | 2000-05-03 | 2010-10-20 | Novo Nordisk Health Care AG | Human coagulation factor vii variants |
| US6905683B2 (en) | 2000-05-03 | 2005-06-14 | Novo Nordisk Healthcare A/G | Human coagulation factor VII variants |
| US7338932B2 (en) | 2000-05-11 | 2008-03-04 | Glycozym Aps | Methods of modulating functions of polypeptide GalNAc-transferases and of screening test substances to find agents herefor, pharmaceutical compositions comprising such agents and the use of such agents for preparing medicaments |
| AU2001274853B2 (en) | 2000-05-16 | 2007-03-22 | Bolder Biotechnology, Inc. | Methods for refolding proteins containing free cysteine residues |
| JP2004501642A (en) | 2000-06-28 | 2004-01-22 | グライコフィ, インコーポレイテッド | Methods for producing modified glycoproteins |
| WO2002002597A2 (en) | 2000-06-30 | 2002-01-10 | Maxygen Aps | Peptide extended glycosylated polypeptides |
| US6423826B1 (en) | 2000-06-30 | 2002-07-23 | Regents Of The University Of Minnesota | High molecular weight derivatives of vitamin K-dependent polypeptides |
| KR100396983B1 (en) | 2000-07-29 | 2003-09-02 | 이강춘 | Highly reactive branched polymer and proteins or peptides conjugated with the polymer |
| WO2002013843A2 (en) | 2000-08-17 | 2002-02-21 | University Of British Columbia | Chemotherapeutic agents conjugated to p97 and their methods of use in treating neurological tumours |
| AU2001285020A1 (en) | 2000-08-17 | 2002-02-25 | Synapse Technologies, Inc. | P97-active agent conjugates and their methods of use |
| EP1325127B1 (en) | 2000-10-02 | 2009-03-11 | Novo Nordisk Health Care AG | Method for the production of vitamin k-dependent proteins |
| US20020142964A1 (en) | 2000-11-02 | 2002-10-03 | Nissen Torben Lauesgaard | Single-chain polypeptides |
| CN1830999A (en) | 2000-11-27 | 2006-09-13 | Rmf迪克塔吉恩有限公司 | Process for folding chemically synthesized polypeptides |
| US20030165849A1 (en) | 2000-11-28 | 2003-09-04 | Biliang Zhang | Methods and reagents for introducing a sulfhydryl group into the 5'-terminus of RNA |
| WO2002049673A2 (en) | 2000-12-20 | 2002-06-27 | F. Hoffmann-La Roche Ag | Conjugates of erythropoietin (pep) with polyethylene glycol (peg) |
| JP4656814B2 (en) | 2000-12-20 | 2011-03-23 | エフ.ホフマン−ラ ロシュ アーゲー | Erythropoietin conjugate |
| US7892730B2 (en) | 2000-12-22 | 2011-02-22 | Sagres Discovery, Inc. | Compositions and methods for cancer |
| PA8536201A1 (en) | 2000-12-29 | 2002-08-29 | Kenneth S Warren Inst Inc | PROTECTION AND IMPROVEMENT OF CELLS, FABRICS AND ORGANS RESPONDING TO Erythropoietin |
| SE0004932D0 (en) | 2000-12-31 | 2000-12-31 | Apbiotech Ab | A method for mixed mode adsorption and mixed mode adsorbents |
| PL367154A1 (en) | 2001-02-27 | 2005-02-21 | Maxygen Aps | New interferon beta-like molecules |
| US7235638B2 (en) | 2001-03-22 | 2007-06-26 | Novo Nordisk Healthcare A/G | Coagulation factor VII derivatives |
| EP1373493B1 (en) | 2001-03-22 | 2013-07-31 | Novo Nordisk Health Care AG | Coagulation factor vii derivative |
| HUP0400284A3 (en) | 2001-05-03 | 2012-09-28 | Merck Patent Gmbh | Recombinant tumor specific antibody and use thereof |
| CA2445494A1 (en) | 2001-05-11 | 2002-11-21 | Aradigm Corporation | Optimization of the molecular properties and formulation of proteins delivered by inhalation |
| US7271150B2 (en) | 2001-05-14 | 2007-09-18 | United States Of America, Represented By The Secretary, Department Of Health And Human Services | Modified growth hormone |
| BR0211071A (en) | 2001-07-11 | 2004-12-21 | Maxygen Holdings Ltd | Polypeptide conjugate showing g-csf activity, method for preparing a g-csf conjugate, composition, method for treating a mammal suffering from an insufficient neutrophilic level, and use of the polypeptide conjugate |
| KR100453877B1 (en) | 2001-07-26 | 2004-10-20 | 메덱스젠 주식회사 | METHOD OF MANUFACTURING Ig-FUSION PROTEINS BY CONCATAMERIZATION, TNFR/Fc FUSION PROTEINS MANUFACTURED BY THE METHOD, DNA CODING THE PROTEINS, VECTORS INCLUDING THE DNA, AND CELLS TRANSFORMED BY THE VECTOR |
| MXPA04000927A (en) | 2001-08-01 | 2004-04-02 | Neose Technologies Inc | Neutral glycosphingolipids and glycosyl-sphingosines and methods for isolating the same. |
| EP1435972B1 (en) | 2001-08-29 | 2016-03-09 | Seneb Biosciences Inc. | Novel synthetic ganglioside derivatives and compositions thereof |
| US6930086B2 (en) | 2001-09-25 | 2005-08-16 | Hoffmann-La Roche Inc. | Diglycosylated erythropoietin |
| US7052868B2 (en) | 2001-09-27 | 2006-05-30 | Novo Nordisk Healthcare A/G | Human coagulation factor VII polypeptides |
| US7226903B2 (en) | 2001-10-10 | 2007-06-05 | Neose Technologies, Inc. | Interferon beta: remodeling and glycoconjugation of interferon beta |
| US7297511B2 (en) | 2001-10-10 | 2007-11-20 | Neose Technologies, Inc. | Interferon alpha: remodeling and glycoconjugation of interferon alpha |
| US7439043B2 (en) | 2001-10-10 | 2008-10-21 | Neose Technologies, Inc. | Galactosyl nucleotide sugars |
| US7125843B2 (en) | 2001-10-19 | 2006-10-24 | Neose Technologies, Inc. | Glycoconjugates including more than one peptide |
| US7265085B2 (en) | 2001-10-10 | 2007-09-04 | Neose Technologies, Inc. | Glycoconjugation methods and proteins/peptides produced by the methods |
| US6784154B2 (en) | 2001-11-01 | 2004-08-31 | University Of Utah Research Foundation | Method of use of erythropoietin to treat ischemic acute renal failure |
| AU2002351199A1 (en) | 2001-11-28 | 2003-06-10 | Neose Technologies, Inc. | Glycoprotein remodeling using endoglycanases |
| EP1504023A4 (en) | 2002-05-03 | 2006-03-22 | Neose Technologies Inc | Recombinant glycosyltransferase fusion proteins |
| DE60336555D1 (en) | 2002-06-21 | 2011-05-12 | Novo Nordisk Healthcare Ag | PEGYLATED GLYCO FORMS OF FACTOR VII |
| BR0311978A (en) | 2002-06-21 | 2005-03-22 | Novo Nordisk Healthcare Ag | Preparation, method for preparing it, pharmaceutical formulation, methods for treating a factor responsive syndrome vii, to prevent unwanted bleeding, to prevent unwanted blood clotting and to prevent tissue factor mediated reactions, and to use a preparation |
| DE10232916B4 (en) | 2002-07-19 | 2008-08-07 | Fraunhofer-Gesellschaft zur Förderung der angewandten Forschung e.V. | Apparatus and method for characterizing an information signal |
| MXPA05000872A (en) | 2002-07-23 | 2005-04-28 | Neose Technologies Inc | Synthesis of glycoproteins using bacterial gycosyltransferases. |
| AU2003254665C1 (en) | 2002-08-01 | 2010-08-19 | National Research Council Of Canada | Campylobacter glycans and glycopeptides |
| CA2493977A1 (en) | 2002-08-02 | 2004-02-19 | Glaxosmithkline Biologicals Sa | Vaccine composition comprising lipooligosaccharide with reduced phase variability |
| JP2006508918A (en) | 2002-09-05 | 2006-03-16 | ザ ジェネラル ホスピタル コーポレーション | Modified asialointerferon and uses thereof |
| CA2497794A1 (en) | 2002-09-06 | 2004-03-18 | Bayer Pharmaceuticals Corporation | Modified glp-1 receptor agonists and their pharmacological methods of use |
| US7470779B2 (en) | 2002-09-20 | 2008-12-30 | Pfizer Inc. | Process for decreasing aggregate levels of pegylated protein |
| JP4597678B2 (en) | 2002-09-25 | 2010-12-15 | ノボ ノルディスク ヘルス ケア アクチェンゲゼルシャフト | Human coagulation factor VII polypeptide |
| ATE505487T1 (en) | 2002-09-30 | 2011-04-15 | Bayer Healthcare Llc | FVII OR FVIIA VARIANTS WITH INCREASED COAGULATION EFFECT |
| US20040062748A1 (en) | 2002-09-30 | 2004-04-01 | Mountain View Pharmaceuticals, Inc. | Polymer conjugates with decreased antigenicity, methods of preparation and uses thereof |
| NZ539415A (en) | 2002-10-09 | 2008-12-24 | Neose Technologies Inc | Remodelling and glycoconjugation of erythropoietin |
| JP4412461B2 (en) | 2002-11-20 | 2010-02-10 | 日油株式会社 | Modified bio-related substance, production method thereof and intermediate |
| US7459436B2 (en) | 2002-11-22 | 2008-12-02 | Hoffmann-La Roche Inc. | Treatment of disturbances of iron distribution |
| EP1424344A1 (en) | 2002-11-29 | 2004-06-02 | Aventis Behring Gesellschaft mit beschränkter Haftung | Modified cDNA factor VIII and its derivates |
| PT1428878E (en) | 2002-12-13 | 2008-11-18 | Bioceuticals Arzneimittel Ag | Process for the production and purification of erythropoietin |
| US9125880B2 (en) | 2002-12-26 | 2015-09-08 | Mountain View Pharmaceuticals, Inc. | Polymer conjugates of interferon-beta with enhanced biological potency |
| US7041635B2 (en) | 2003-01-28 | 2006-05-09 | In2Gen Co., Ltd. | Factor VIII polypeptide |
| DK1596887T3 (en) | 2003-02-26 | 2022-06-20 | Nektar Therapeutics | Polymer-Factor VIII conjugate |
| JP2006520840A (en) | 2003-03-18 | 2006-09-14 | ネオス テクノロジーズ インコーポレイテッド | Activated form of water-soluble polymer |
| PT1605897E (en) | 2003-03-19 | 2012-09-10 | Lilly Co Eli | Polyethelene glycol link glp-1 compounds |
| US7718363B2 (en) | 2003-04-25 | 2010-05-18 | The Kenneth S. Warren Institute, Inc. | Tissue protective cytokine receptor complex and assays for identifying tissue protective compounds |
| TWI353991B (en) | 2003-05-06 | 2011-12-11 | Syntonix Pharmaceuticals Inc | Immunoglobulin chimeric monomer-dimer hybrids |
| EP2077121B1 (en) | 2003-05-06 | 2011-02-09 | Syntonix Pharmaceuticals, Inc. | Clotting factor VII-Fc chimeric proteins for treatment of a hemostatic disorder |
| WO2004101597A2 (en) | 2003-05-13 | 2004-11-25 | Frutarom Ltd. | Methods for the reduction of disulfide bonds |
| US7074755B2 (en) | 2003-05-17 | 2006-07-11 | Centocor, Inc. | Erythropoietin conjugate compounds with extended half-lives |
| EP1481985A1 (en) | 2003-05-28 | 2004-12-01 | Innogenetics N.V. | Modified hepatitis C virus (HCV) NS3 for medical treatment |
| GB0315457D0 (en) | 2003-07-01 | 2003-08-06 | Celltech R&D Ltd | Biological products |
| EP1653996A2 (en) | 2003-08-08 | 2006-05-10 | Novo Nordisk Health Care AG | Use of galactose oxidase for selective chemical conjugation of protractor molecules to proteins of therapeutic interest |
| EP1653991A2 (en) | 2003-08-08 | 2006-05-10 | Fresenius Kabi Deutschland GmbH | Conjugates of a polymer and a protein linked by an oxime linking group |
| CN1882355A (en) | 2003-09-09 | 2006-12-20 | 沃伦药品公司 | Long acting erythropoietins that maintain tissue protective activity of endogenous erythropoietin |
| US7524813B2 (en) | 2003-10-10 | 2009-04-28 | Novo Nordisk Health Care Ag | Selectively conjugated peptides and methods of making the same |
| EP1694347B1 (en) | 2003-11-24 | 2013-11-20 | BioGeneriX AG | Glycopegylated erythropoietin |
| US20080305992A1 (en) | 2003-11-24 | 2008-12-11 | Neose Technologies, Inc. | Glycopegylated erythropoietin |
| WO2005072371A2 (en) | 2004-01-26 | 2005-08-11 | Neose Technologies, Inc. | Branched polymeric sugars and nucleotides thereof |
| US20070254836A1 (en) | 2003-12-03 | 2007-11-01 | Defrees Shawn | Glycopegylated Granulocyte Colony Stimulating Factor |
| US20080318850A1 (en) | 2003-12-03 | 2008-12-25 | Neose Technologies, Inc. | Glycopegylated Factor Ix |
| US20060040856A1 (en) * | 2003-12-03 | 2006-02-23 | Neose Technologies, Inc. | Glycopegylated factor IX |
| WO2005055950A2 (en) | 2003-12-03 | 2005-06-23 | Neose Technologies, Inc. | Glycopegylated factor ix |
| WO2005067601A2 (en) | 2004-01-09 | 2005-07-28 | Neose Technologies, Inc. | Vectors for recombinant protein expression in e.coli |
| US20070105770A1 (en) | 2004-01-21 | 2007-05-10 | Novo Nordisk A/S | Transglutaminase mediated conjugation of peptides |
| EP3385384B1 (en) | 2004-02-12 | 2020-04-08 | Archemix LLC | Aptamer therapeutics useful in the treatment of complement-related disorders |
| WO2005091944A2 (en) | 2004-03-17 | 2005-10-06 | Eli Lilly And Company | Glycol linked fgf-21 compounds |
| ES2339953T5 (en) | 2004-05-04 | 2020-05-06 | Novo Nordisk Healthcare Ag | O-linked factor VII glycoforms and method of manufacture |
| US20070037966A1 (en) | 2004-05-04 | 2007-02-15 | Novo Nordisk A/S | Hydrophobic interaction chromatography purification of factor VII polypeptides |
| JP4251399B2 (en) | 2004-05-21 | 2009-04-08 | 独立行政法人産業技術総合研究所 | Screening method for peptides to which O-linked sugar chains are added |
| JP2008512085A (en) | 2004-06-03 | 2008-04-24 | ネオス テクノロジーズ インコーポレイティッド | Cleaved GalNAcT2 polypeptides and nucleic acids |
| US20080206810A1 (en) | 2004-06-03 | 2008-08-28 | Neose Technologies, Inc. | Truncated St6galnaci Polypeptides and Nucleic Acids |
| KR101146160B1 (en) | 2004-06-30 | 2012-07-16 | 넥타르 테라퓨틱스 | Polymer-factor ix moiety conjugates |
| AU2005260664A1 (en) | 2004-06-30 | 2006-01-12 | Egen Corporation | Pegylated interferon alpha-1b |
| WO2006014349A2 (en) | 2004-07-02 | 2006-02-09 | The Kenneth S. Warren Institute, Inc. | Method of producing fully carbamylated erythropoietin |
| WO2006014466A2 (en) | 2004-07-02 | 2006-02-09 | The Kenneth S. Warren Institute, Inc. | Novel carbamylated epo and method for its production |
| WO2006010143A2 (en) | 2004-07-13 | 2006-01-26 | Neose Technologies, Inc. | Branched peg remodeling and glycosylation of glucagon-like peptide-1 [glp-1] |
| US20090292110A1 (en) | 2004-07-23 | 2009-11-26 | Defrees Shawn | Enzymatic modification of glycopeptides |
| SE0401951D0 (en) | 2004-07-29 | 2004-07-29 | Amersham Biosciences Ab | Chromatography method |
| US20090176967A1 (en) | 2004-08-02 | 2009-07-09 | Novo Nordisk Healthcare A/G | Conjugation of FVII |
| DK1781782T3 (en) | 2004-08-17 | 2010-08-23 | Csl Behring Gmbh | Modified vitamin K-dependent polypeptides |
| ES2566670T3 (en) | 2004-10-29 | 2016-04-14 | Ratiopharm Gmbh | Remodeling and glucopegilation of fibroblast growth factor (FGF) |
| HRP20070268B1 (en) | 2004-11-12 | 2018-04-20 | Bayer Healthcare Llc | Site-directed modification of fviii |
| WO2006073846A2 (en) | 2004-12-22 | 2006-07-13 | Ambrx, Inc. | Methods for expression and purification of recombinant human growth hormone |
| JP4951527B2 (en) | 2005-01-10 | 2012-06-13 | バイオジェネリックス アーゲー | GlycoPEGylated granulocyte colony stimulating factor |
| WO2006078645A2 (en) | 2005-01-19 | 2006-07-27 | Neose Technologies, Inc. | Heterologous polypeptide expression using low multiplicity of infection of viruses |
| PA8660701A1 (en) | 2005-02-04 | 2006-09-22 | Pfizer Prod Inc | SMALL AGONISTS AND THEIR USES |
| US8137928B2 (en) | 2005-03-24 | 2012-03-20 | BioGeneriX | Expression of soluble, active eukaryotic glycosyltransferases in prokaryotic organisms |
| US20060246544A1 (en) | 2005-03-30 | 2006-11-02 | Neose Technologies,Inc. | Manufacturing process for the production of peptides grown in insect cell lines |
| US20080227691A1 (en) | 2005-04-01 | 2008-09-18 | Novo Nordisk Health Care Ag | Blood Coagulation FVIII Analogues |
| EP2311972B1 (en) | 2005-05-11 | 2015-01-21 | ETH Zurich | Recombinant N-glycosylated proteins from procaryotic cells |
| WO2006127910A2 (en) | 2005-05-25 | 2006-11-30 | Neose Technologies, Inc. | Glycopegylated erythropoietin formulations |
| US20110003744A1 (en) | 2005-05-25 | 2011-01-06 | Novo Nordisk A/S | Glycopegylated Erythropoietin Formulations |
| EP1893632B1 (en) | 2005-06-17 | 2015-08-12 | Novo Nordisk Health Care AG | Selective reduction and derivatization of engineered factor vii proteins comprising at least one non-native cysteine |
| CA2619969A1 (en) | 2005-08-19 | 2007-02-22 | Neose Technologies, Inc. | Glycopegylated factor vii and factor viia |
| US20090048440A1 (en) | 2005-11-03 | 2009-02-19 | Neose Technologies, Inc. | Nucleotide Sugar Purification Using Membranes |
| DE102006009437A1 (en) | 2006-03-01 | 2007-09-13 | Bioceuticals Arzneimittel Ag | G-CSF liquid formulation |
| US7645860B2 (en) | 2006-03-31 | 2010-01-12 | Baxter Healthcare S.A. | Factor VIII polymer conjugates |
| RU2008145084A (en) | 2006-05-24 | 2010-06-27 | Ново Нордиск Хелс Кеа Аг (Ch) | ANALOGUES OF FACTOR IX HAVING A LONG-TERM HALF-TIME IN VIVO |
| US20080248959A1 (en) | 2006-07-21 | 2008-10-09 | Neose Technologies, Inc. | Glycosylation of peptides via o-linked glycosylation sequences |
| US20080207487A1 (en) | 2006-11-02 | 2008-08-28 | Neose Technologies, Inc. | Manufacturing process for the production of polypeptides expressed in insect cell-lines |
| WO2008151258A2 (en) | 2007-06-04 | 2008-12-11 | Neose Technologies, Inc. | O-linked glycosylation using n-acetylglucosaminyl transferases |
| EP2170919B8 (en) | 2007-06-12 | 2016-01-20 | ratiopharm GmbH | Improved process for the production of nucleotide sugars |
| US8207112B2 (en) | 2007-08-29 | 2012-06-26 | Biogenerix Ag | Liquid formulation of G-CSF conjugate |
| CN102037004A (en) | 2008-01-08 | 2011-04-27 | 生物种属学股份公司 | Glycoconjugation of polypeptides using oligosaccharyltransferases |
-
2006
- 2006-11-03 US US12/092,563 patent/US20090048440A1/en not_active Abandoned
- 2006-11-03 WO PCT/US2006/043048 patent/WO2007056191A2/en not_active Ceased
-
2011
- 2011-08-23 US US13/215,439 patent/US8841439B2/en not_active Expired - Fee Related
Patent Citations (98)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4088538A (en) * | 1975-05-30 | 1978-05-09 | Battelle Memorial Institute | Reversibly precipitable immobilized enzyme complex and a method for its use |
| US4385260A (en) * | 1975-09-09 | 1983-05-24 | Beckman Instruments, Inc. | Bargraph display |
| US4438253A (en) * | 1982-11-12 | 1984-03-20 | American Cyanamid Company | Poly(glycolic acid)/poly(alkylene glycol) block copolymers and method of manufacturing the same |
| US4496689A (en) * | 1983-12-27 | 1985-01-29 | Miles Laboratories, Inc. | Covalently attached complex of alpha-1-proteinase inhibitor with a water soluble polymer |
| US4565653A (en) * | 1984-03-30 | 1986-01-21 | Pfizer Inc. | Acyltripeptide immunostimulants |
| US4806595A (en) * | 1985-08-12 | 1989-02-21 | Koken Co., Ltd. | Method of preparing antithrombogenic medical materials |
| US4925796A (en) * | 1986-03-07 | 1990-05-15 | Massachusetts Institute Of Technology | Method for enhancing glycoprotein stability |
| US4826945A (en) * | 1987-06-09 | 1989-05-02 | Yissum Research Development Company | Biodegradable polymeric materials based on polyether glycols, processes for the preparation thereof and surgical articles made therefrom |
| US5104651A (en) * | 1988-12-16 | 1992-04-14 | Amgen Inc. | Stabilized hydrophobic protein formulations of g-csf |
| US5194376A (en) * | 1989-02-28 | 1993-03-16 | University Of Ottawa | Baculovirus expression system capable of producing foreign gene proteins at high levels |
| US5122614A (en) * | 1989-04-19 | 1992-06-16 | Enzon, Inc. | Active carbonates of polyalkylene oxides for modification of polypeptides |
| US5324844A (en) * | 1989-04-19 | 1994-06-28 | Enzon, Inc. | Active carbonates of polyalkylene oxides for modification of polypeptides |
| US5182107A (en) * | 1989-09-07 | 1993-01-26 | Alkermes, Inc. | Transferrin receptor specific antibody-neuropharmaceutical or diagnostic agent conjugates |
| US5324663A (en) * | 1990-02-14 | 1994-06-28 | The Regents Of The University Of Michigan | Methods and products for the synthesis of oligosaccharide structures on glycoproteins, glycolipids, or as free molecules, and for the isolation of cloned genetic sequences that determine these structures |
| US5405753A (en) * | 1990-03-26 | 1995-04-11 | Brossmer; Reinhard | CMP-activated, fluorescing sialic acids, as well as processes for their preparation |
| US6010999A (en) * | 1990-05-04 | 2000-01-04 | American Cyanamid Company | Stabilization of fibroblast growth factors by modification of cysteine residues |
| US5219564A (en) * | 1990-07-06 | 1993-06-15 | Enzon, Inc. | Poly(alkylene oxide) amino acid copolymers and drug carriers and charged copolymers based thereon |
| US5410016A (en) * | 1990-10-15 | 1995-04-25 | Board Of Regents, The University Of Texas System | Photopolymerizable biodegradable hydrogels as tissue contacting materials and controlled-release carriers |
| US5281698A (en) * | 1991-07-23 | 1994-01-25 | Cetus Oncology Corporation | Preparation of an activated polymer ester for protein conjugation |
| US5858751A (en) * | 1992-03-09 | 1999-01-12 | The Regents Of The University Of California | Compositions and methods for producing sialyltransferases |
| US6037452A (en) * | 1992-04-10 | 2000-03-14 | Alpha Therapeutic Corporation | Poly(alkylene oxide)-Factor VIII or Factor IX conjugate |
| US5614184A (en) * | 1992-07-28 | 1997-03-25 | New England Deaconess Hospital | Recombinant human erythropoietin mutants and therapeutic methods employing them |
| US5202413A (en) * | 1993-02-16 | 1993-04-13 | E. I. Du Pont De Nemours And Company | Alternating (ABA)N polylactide block copolymers |
| US5621039A (en) * | 1993-06-08 | 1997-04-15 | Hallahan; Terrence W. | Factor IX- polymeric conjugates |
| US5605793A (en) * | 1994-02-17 | 1997-02-25 | Affymax Technologies N.V. | Methods for in vitro recombination |
| US5492841A (en) * | 1994-02-18 | 1996-02-20 | E. I. Du Pont De Nemours And Company | Quaternary ammonium immunogenic conjugates and immunoassay reagents |
| US5629384A (en) * | 1994-05-17 | 1997-05-13 | Consiglio Nazionale Delle Ricerche | Polymers of N-acryloylmorpholine activated at one end and conjugates with bioactive materials and surfaces |
| US6342382B1 (en) * | 1994-09-26 | 2002-01-29 | The Rockefeller University | Glycosyltransferases for biosynthesis of oligosaccharides, and genes encoding them |
| US5705367A (en) * | 1994-09-26 | 1998-01-06 | The Rockefeller University | Glycosyltransferases for biosynthesis of oligosaccharides, and genes encoding them |
| US5876980A (en) * | 1995-04-11 | 1999-03-02 | Cytel Corporation | Enzymatic synthesis of oligosaccharides |
| US6030815A (en) * | 1995-04-11 | 2000-02-29 | Neose Technologies, Inc. | Enzymatic synthesis of oligosaccharides |
| US5728554A (en) * | 1995-04-11 | 1998-03-17 | Cytel Corporation | Enzymatic synthesis of glycosidic linkages |
| US6015555A (en) * | 1995-05-19 | 2000-01-18 | Alkermes, Inc. | Transferrin receptor specific antibody-neuropharmaceutical or diagnostic agent conjugates |
| US5858752A (en) * | 1995-06-07 | 1999-01-12 | The General Hospital Corporation | Fucosyltransferase genes and uses thereof |
| US6057292A (en) * | 1995-09-21 | 2000-05-02 | Genentech, Inc. | Method for inhibiting growth hormone action |
| US5716812A (en) * | 1995-12-12 | 1998-02-10 | The University Of British Columbia | Methods and compositions for synthesis of oligosaccharides, and the products formed thereby |
| US6693183B2 (en) * | 1996-03-08 | 2004-02-17 | The Regents Of The University Of Michigan | MURINE α (1,3) FUCOSYLTRANSFERASE FUC-TVII, DNA ENCODING THE SAME, METHOD FOR PREPARING THE SAME, ANTIBODIES RECOGNIZING THE SAME, IMMUNOASSAYS FOR DETECTING THE SAME, PLASMIDS CONTAINING SUCH DNA, AND CELLS CONTAINING SUCH A PLASMID |
| US6183738B1 (en) * | 1997-05-12 | 2001-02-06 | Phoenix Pharamacologics, Inc. | Modified arginine deiminase |
| US20030027257A1 (en) * | 1997-08-21 | 2003-02-06 | University Technologies International, Inc. | Sequences for improving the efficiency of secretion of non-secreted protein from mammalian and insect cells |
| US6555346B1 (en) * | 1997-12-18 | 2003-04-29 | Stichting Instituut Voor Dierhouderij En Diergezondheid | Protein expression in baculovirus vector expression systems |
| US6362254B2 (en) * | 1998-03-12 | 2002-03-26 | Shearwater Corporation | Poly(ethylene glycol) derivatives with proximal reactive groups |
| US20040102607A1 (en) * | 1998-03-25 | 2004-05-27 | Danishefsky Samuel J. | Trimeric antigenic O-linked glycopeptide conjugates, methods of preparation and uses thereof |
| US6692931B1 (en) * | 1998-11-16 | 2004-02-17 | Werner Reutter | Recombinant glycoproteins, method for the production thereof, medicaments containing said glycoproteins and use thereof |
| US6716626B1 (en) * | 1999-11-18 | 2004-04-06 | Chiron Corporation | Human FGF-21 nucleic acids |
| US6348558B1 (en) * | 1999-12-10 | 2002-02-19 | Shearwater Corporation | Hydrolytically degradable polymers and hydrogels made therefrom |
| US6376604B2 (en) * | 1999-12-22 | 2002-04-23 | Shearwater Corporation | Method for the preparation of 1-benzotriazolylcarbonate esters of poly(ethylene glycol) |
| US6555660B2 (en) * | 2000-01-10 | 2003-04-29 | Maxygen Holdings Ltd. | G-CSF conjugates |
| US20030040037A1 (en) * | 2000-05-12 | 2003-02-27 | Neose Technologies, Inc. | In vitro modification of glycosylation patterns of recombinant glycopeptides |
| US7202208B2 (en) * | 2000-05-15 | 2007-04-10 | Hoffman-La Roche Inc. | Erythropoietin composition |
| US20020037841A1 (en) * | 2000-05-15 | 2002-03-28 | Apollon Papadimitriou | Erythropoietin composition |
| US6531121B2 (en) * | 2000-12-29 | 2003-03-11 | The Kenneth S. Warren Institute, Inc. | Protection and enhancement of erythropoietin-responsive cells, tissues and organs |
| US20090137763A1 (en) * | 2001-10-10 | 2009-05-28 | Neose Technologies, Inc. | Glucosamine nucleotide sugars |
| US7173003B2 (en) * | 2001-10-10 | 2007-02-06 | Neose Technologies, Inc. | Granulocyte colony stimulating factor: remodeling and glycoconjugation of G-CSF |
| US20100015684A1 (en) * | 2001-10-10 | 2010-01-21 | Neose Technologies, Inc. | Factor vii: remodeling and glycoconjugation of factor vii |
| US20050100982A1 (en) * | 2001-10-10 | 2005-05-12 | Neose Technologies, Inc. | Factor IX: remodeling and glycoconjugation of factor IX |
| US20050106658A1 (en) * | 2001-10-10 | 2005-05-19 | Defrees Shawn | Remodeling and glycoconjugation of peptides |
| US7696163B2 (en) * | 2001-10-10 | 2010-04-13 | Novo Nordisk A/S | Erythropoietin: remodeling and glycoconjugation of erythropoietin |
| US7214660B2 (en) * | 2001-10-10 | 2007-05-08 | Neose Technologies, Inc. | Erythropoietin: remodeling and glycoconjugation of erythropoietin |
| US20040063911A1 (en) * | 2001-10-10 | 2004-04-01 | Neose Technologies, Inc. | Protein remodeling methods and proteins/peptides produced by the methods |
| US20090093399A1 (en) * | 2001-10-10 | 2009-04-09 | Neose Technologies, Inc. | Glycopegylation methods and proteins/peptides produced by the methods |
| US7179617B2 (en) * | 2001-10-10 | 2007-02-20 | Neose Technologies, Inc. | Factor IX: remolding and glycoconjugation of Factor IX |
| US20080050772A1 (en) * | 2001-10-10 | 2008-02-28 | Neose Technologies, Inc. | Granulocyte colony stimulating factor: remodeling and glycoconjugation of G-CSF |
| US20070027068A1 (en) * | 2001-10-10 | 2007-02-01 | Defrees Shawn | Erythropoietin: remodeling and glycoconjugation of erythropoietin |
| US20050031584A1 (en) * | 2001-10-10 | 2005-02-10 | Neose Technologies, Inc. | Interleukin-2:remodeling and glycoconjugation of interleukin-2 |
| US7368108B2 (en) * | 2001-11-28 | 2008-05-06 | Neose Technologies, Inc. | Glycopeptide remodeling using amidases |
| US7473680B2 (en) * | 2001-11-28 | 2009-01-06 | Neose Technologies, Inc. | Remodeling and glycoconjugation of peptides |
| US7157277B2 (en) * | 2001-11-28 | 2007-01-02 | Neose Technologies, Inc. | Factor VIII remodeling and glycoconjugation of Factor VIII |
| US20060030521A1 (en) * | 2001-11-28 | 2006-02-09 | Neose Technologies, Inc. | Remodeling and glycoconjugation of peptides |
| US20060035224A1 (en) * | 2002-03-21 | 2006-02-16 | Johansen Jack T | Purification methods for oligonucleotides and their analogs |
| US20050026266A1 (en) * | 2002-11-08 | 2005-02-03 | Glycozym Aps | Methods to identify agents modulating functions of polypeptide galnac-transferases, pharmaceutical compositions comprising such agents and the use of such agents for preparing medicaments |
| US20050064540A1 (en) * | 2002-11-27 | 2005-03-24 | Defrees Shawn Ph.D | Glycoprotein remodeling using endoglycanases |
| US20070032405A1 (en) * | 2003-03-14 | 2007-02-08 | Neose Technologies, Inc. | Branched water-soluble polymers and their conjugates |
| US7691603B2 (en) * | 2003-04-09 | 2010-04-06 | Novo Nordisk A/S | Intracellular formation of peptide conjugates |
| US20070026485A1 (en) * | 2003-04-09 | 2007-02-01 | Neose Technologies, Inc. | Glycopegylation methods and proteins/peptides produced by the methods |
| US20100048456A1 (en) * | 2003-04-09 | 2010-02-25 | Novo Nordisk A/S | Glycopegylation methods and proteins/peptides produced by the methods |
| US20080102083A1 (en) * | 2003-05-09 | 2008-05-01 | Neose Technologies, Inc. | Compositions and Methods for the Preparation of Human Growth Hormone Glycosylation Mutants |
| US20070059275A1 (en) * | 2003-07-25 | 2007-03-15 | Defrees Shawn | Antibody toxin conjugates |
| US20100028939A1 (en) * | 2003-08-08 | 2010-02-04 | Novo Nordisk Healthcare A/G | Use of Galactose Oxidase for Selective Chemical Conjugation of Protractor Molecules to Proteins of Therapeutic Interest |
| US20060111279A1 (en) * | 2003-11-24 | 2006-05-25 | Neose Technologies, Inc. | Glycopegylated erythropoietin |
| US20070014759A1 (en) * | 2003-12-03 | 2007-01-18 | Neose Technologies, Inc. | Glycopegylated granulocyte colony stimulating factor |
| US20080015142A1 (en) * | 2003-12-03 | 2008-01-17 | Defrees Shawn | Glycopegylated Follicle Stimulating Hormone |
| US7338933B2 (en) * | 2004-01-08 | 2008-03-04 | Neose Technologies, Inc. | O-linked glycosylation of peptides |
| US20060024286A1 (en) * | 2004-08-02 | 2006-02-02 | Paul Glidden | Variants of tRNA synthetase fragments and uses thereof |
| US20090028822A1 (en) * | 2004-09-10 | 2009-01-29 | Neose Technologies, Inc. | Glycopegylated Interferon Alpha |
| US20080108557A1 (en) * | 2004-09-29 | 2008-05-08 | Novo Nordisk Healthcare A/G | Modified Proteins |
| US20090054623A1 (en) * | 2004-12-17 | 2009-02-26 | Neose Technologies, Inc. | Lipo-Conjugation of Peptides |
| US20100009902A1 (en) * | 2005-01-06 | 2010-01-14 | Neose Technologies, Inc. | Glycoconjugation Using Saccharyl Fragments |
| US20090124544A1 (en) * | 2005-04-08 | 2009-05-14 | Neose Technologies ,Inc. A Delaware Corporation | Compositions and methods for the preparation of protease resistant human growth hormone glycosylation mutants |
| US20100081791A1 (en) * | 2005-05-25 | 2010-04-01 | Novo Nordisk A/S | Glycopegylated factor ix |
| US20090055942A1 (en) * | 2005-09-14 | 2009-02-26 | Novo Nordisk Healthcare A/G | Human Coagulation Factor VII Polypeptides |
| US20070105755A1 (en) * | 2005-10-26 | 2007-05-10 | Neose Technologies, Inc. | One pot desialylation and glycopegylation of therapeutic peptides |
| US20100029555A1 (en) * | 2006-08-11 | 2010-02-04 | Bio-Ker S.r.l | G-csf site-specific mono-conjugates |
| US20100056428A1 (en) * | 2006-09-01 | 2010-03-04 | Novo Nordisk Health Care Ag | Modified proteins |
| US20100035299A1 (en) * | 2006-10-03 | 2010-02-11 | Novo Nordisk A/S | Methods for the purification of polypeptide conjugates |
| US20100075375A1 (en) * | 2006-10-03 | 2010-03-25 | Novo Nordisk A/S | Methods for the purification of polypeptide conjugates |
| US20100041872A1 (en) * | 2006-10-04 | 2010-02-18 | Defrees Shawn | Glycerol linked pegylated sugars and glycopeptides |
| US20100120666A1 (en) * | 2007-04-03 | 2010-05-13 | Biogenerix Ag | Methods of treatment using glycopegylated g-csf |
| US20090053167A1 (en) * | 2007-05-14 | 2009-02-26 | Neose Technologies, Inc. | C-, S- and N-glycosylation of peptides |
Cited By (75)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US8008252B2 (en) | 2001-10-10 | 2011-08-30 | Novo Nordisk A/S | Factor VII: remodeling and glycoconjugation of Factor VII |
| US8716239B2 (en) | 2001-10-10 | 2014-05-06 | Novo Nordisk A/S | Granulocyte colony stimulating factor: remodeling and glycoconjugation G-CSF |
| US7795210B2 (en) | 2001-10-10 | 2010-09-14 | Novo Nordisk A/S | Protein remodeling methods and proteins/peptides produced by the methods |
| US8716240B2 (en) | 2001-10-10 | 2014-05-06 | Novo Nordisk A/S | Erythropoietin: remodeling and glycoconjugation of erythropoietin |
| US20100261872A1 (en) * | 2001-10-10 | 2010-10-14 | Neose Technologies, Inc. | Factor VIII: remodeling and glycoconjugation of factor VIII |
| US20080050772A1 (en) * | 2001-10-10 | 2008-02-28 | Neose Technologies, Inc. | Granulocyte colony stimulating factor: remodeling and glycoconjugation of G-CSF |
| US8076292B2 (en) | 2001-10-10 | 2011-12-13 | Novo Nordisk A/S | Factor VIII: remodeling and glycoconjugation of factor VIII |
| US20040063911A1 (en) * | 2001-10-10 | 2004-04-01 | Neose Technologies, Inc. | Protein remodeling methods and proteins/peptides produced by the methods |
| US20090227504A1 (en) * | 2002-06-21 | 2009-09-10 | Novo Nordisk A/S | Pegylated Factor VII Glycoforms |
| US8053410B2 (en) | 2002-06-21 | 2011-11-08 | Novo Nordisk Health Care A/G | Pegylated factor VII glycoforms |
| US8247381B2 (en) | 2003-03-14 | 2012-08-21 | Biogenerix Ag | Branched water-soluble polymers and their conjugates |
| US20070032405A1 (en) * | 2003-03-14 | 2007-02-08 | Neose Technologies, Inc. | Branched water-soluble polymers and their conjugates |
| US7803777B2 (en) | 2003-03-14 | 2010-09-28 | Biogenerix Ag | Branched water-soluble polymers and their conjugates |
| US20100331489A1 (en) * | 2003-03-14 | 2010-12-30 | Biogenerix Ag | Branched water-soluble polymers and their conjugates |
| US20100048456A1 (en) * | 2003-04-09 | 2010-02-25 | Novo Nordisk A/S | Glycopegylation methods and proteins/peptides produced by the methods |
| US8063015B2 (en) | 2003-04-09 | 2011-11-22 | Novo Nordisk A/S | Glycopegylation methods and proteins/peptides produced by the methods |
| US8853161B2 (en) | 2003-04-09 | 2014-10-07 | Novo Nordisk A/S | Glycopegylation methods and proteins/peptides produced by the methods |
| US8791070B2 (en) | 2003-04-09 | 2014-07-29 | Novo Nordisk A/S | Glycopegylated factor IX |
| US7932364B2 (en) | 2003-05-09 | 2011-04-26 | Novo Nordisk A/S | Compositions and methods for the preparation of human growth hormone glycosylation mutants |
| US20080102083A1 (en) * | 2003-05-09 | 2008-05-01 | Neose Technologies, Inc. | Compositions and Methods for the Preparation of Human Growth Hormone Glycosylation Mutants |
| US9005625B2 (en) | 2003-07-25 | 2015-04-14 | Novo Nordisk A/S | Antibody toxin conjugates |
| US20070059275A1 (en) * | 2003-07-25 | 2007-03-15 | Defrees Shawn | Antibody toxin conjugates |
| US8633157B2 (en) | 2003-11-24 | 2014-01-21 | Novo Nordisk A/S | Glycopegylated erythropoietin |
| US20060111279A1 (en) * | 2003-11-24 | 2006-05-25 | Neose Technologies, Inc. | Glycopegylated erythropoietin |
| US8916360B2 (en) | 2003-11-24 | 2014-12-23 | Novo Nordisk A/S | Glycopegylated erythropoietin |
| US20080305992A1 (en) * | 2003-11-24 | 2008-12-11 | Neose Technologies, Inc. | Glycopegylated erythropoietin |
| US7842661B2 (en) | 2003-11-24 | 2010-11-30 | Novo Nordisk A/S | Glycopegylated erythropoietin formulations |
| US20100210507A9 (en) * | 2003-11-24 | 2010-08-19 | Novo Nordisk A/S | Glycopegylated erythropoietin |
| US20060287224A1 (en) * | 2003-11-24 | 2006-12-21 | Neose Technologies, Inc. | Glycopegylated erythropoietin formulations |
| US20080300175A1 (en) * | 2003-11-24 | 2008-12-04 | Neose Technologies, Inc. | Glycopegylated erythropoietin |
| US7956032B2 (en) | 2003-12-03 | 2011-06-07 | Novo Nordisk A/S | Glycopegylated granulocyte colony stimulating factor |
| US20070254836A1 (en) * | 2003-12-03 | 2007-11-01 | Defrees Shawn | Glycopegylated Granulocyte Colony Stimulating Factor |
| US8632770B2 (en) | 2003-12-03 | 2014-01-21 | Novo Nordisk A/S | Glycopegylated factor IX |
| US8361961B2 (en) | 2004-01-08 | 2013-01-29 | Biogenerix Ag | O-linked glycosylation of peptides |
| US20080242846A1 (en) * | 2004-01-08 | 2008-10-02 | Neose Technologies, Inc. | O-linked glycosylation of peptides |
| US20090169509A1 (en) * | 2004-01-08 | 2009-07-02 | Defrees Shawn | O-linked glycosylation of peptides |
| US8791066B2 (en) | 2004-07-13 | 2014-07-29 | Novo Nordisk A/S | Branched PEG remodeling and glycosylation of glucagon-like peptide-1 [GLP-1] |
| US20090292110A1 (en) * | 2004-07-23 | 2009-11-26 | Defrees Shawn | Enzymatic modification of glycopeptides |
| US20090028822A1 (en) * | 2004-09-10 | 2009-01-29 | Neose Technologies, Inc. | Glycopegylated Interferon Alpha |
| US8268967B2 (en) | 2004-09-10 | 2012-09-18 | Novo Nordisk A/S | Glycopegylated interferon α |
| US10874714B2 (en) | 2004-10-29 | 2020-12-29 | 89Bio Ltd. | Method of treating fibroblast growth factor 21 (FGF-21) deficiency |
| US9200049B2 (en) | 2004-10-29 | 2015-12-01 | Novo Nordisk A/S | Remodeling and glycopegylation of fibroblast growth factor (FGF) |
| US20080176790A1 (en) * | 2004-10-29 | 2008-07-24 | Defrees Shawn | Remodeling and Glycopegylation of Fibroblast Growth Factor (Fgf) |
| US20100009902A1 (en) * | 2005-01-06 | 2010-01-14 | Neose Technologies, Inc. | Glycoconjugation Using Saccharyl Fragments |
| US20090203579A1 (en) * | 2005-01-10 | 2009-08-13 | Defrees Shawn | Glycopegylated Granulocyte Colony Stimulating Factor |
| US9029331B2 (en) | 2005-01-10 | 2015-05-12 | Novo Nordisk A/S | Glycopegylated granulocyte colony stimulating factor |
| US9187546B2 (en) | 2005-04-08 | 2015-11-17 | Novo Nordisk A/S | Compositions and methods for the preparation of protease resistant human growth hormone glycosylation mutants |
| US8404809B2 (en) | 2005-05-25 | 2013-03-26 | Novo Nordisk A/S | Glycopegylated factor IX |
| US20110003744A1 (en) * | 2005-05-25 | 2011-01-06 | Novo Nordisk A/S | Glycopegylated Erythropoietin Formulations |
| US8911967B2 (en) | 2005-08-19 | 2014-12-16 | Novo Nordisk A/S | One pot desialylation and glycopegylation of therapeutic peptides |
| US8841439B2 (en) | 2005-11-03 | 2014-09-23 | Novo Nordisk A/S | Nucleotide sugar purification using membranes |
| US9187532B2 (en) | 2006-07-21 | 2015-11-17 | Novo Nordisk A/S | Glycosylation of peptides via O-linked glycosylation sequences |
| US20080255040A1 (en) * | 2006-07-21 | 2008-10-16 | Neose Technologies, Inc. | Glycosylation of peptides via o-linked glycosylation sequences |
| US20100075375A1 (en) * | 2006-10-03 | 2010-03-25 | Novo Nordisk A/S | Methods for the purification of polypeptide conjugates |
| US8969532B2 (en) | 2006-10-03 | 2015-03-03 | Novo Nordisk A/S | Methods for the purification of polypeptide conjugates comprising polyalkylene oxide using hydrophobic interaction chromatography |
| US20100041872A1 (en) * | 2006-10-04 | 2010-02-18 | Defrees Shawn | Glycerol linked pegylated sugars and glycopeptides |
| US20080146782A1 (en) * | 2006-10-04 | 2008-06-19 | Neose Technologies, Inc. | Glycerol linked pegylated sugars and glycopeptides |
| US9050304B2 (en) | 2007-04-03 | 2015-06-09 | Ratiopharm Gmbh | Methods of treatment using glycopegylated G-CSF |
| US20100120666A1 (en) * | 2007-04-03 | 2010-05-13 | Biogenerix Ag | Methods of treatment using glycopegylated g-csf |
| US20110177029A1 (en) * | 2007-06-04 | 2011-07-21 | Novo Nordisk A/S | O-linked glycosylation using n-acetylglucosaminyl transferases |
| US20100174059A1 (en) * | 2007-06-12 | 2010-07-08 | Novo Nordisk A/S | Process for the production of nucleotide sugars |
| US9493499B2 (en) | 2007-06-12 | 2016-11-15 | Novo Nordisk A/S | Process for the production of purified cytidinemonophosphate-sialic acid-polyalkylene oxide (CMP-SA-PEG) as modified nucleotide sugars via anion exchange chromatography |
| US20090000924A1 (en) * | 2007-06-29 | 2009-01-01 | Harley-Davidson Motor Company Group, Inc. | Integrated ignition and key switch |
| US20090143292A1 (en) * | 2007-08-29 | 2009-06-04 | Neose Technologies, Inc. | Liquid Formulation of G-CSF Conjugate |
| US8207112B2 (en) | 2007-08-29 | 2012-06-26 | Biogenerix Ag | Liquid formulation of G-CSF conjugate |
| US20100286067A1 (en) * | 2008-01-08 | 2010-11-11 | Biogenerix Ag | Glycoconjugation of polypeptides using oligosaccharyltransferases |
| US9150848B2 (en) | 2008-02-27 | 2015-10-06 | Novo Nordisk A/S | Conjugated factor VIII molecules |
| US8932659B2 (en) * | 2009-03-17 | 2015-01-13 | Separation Technologies Investments Limited | Isolation and purification of components of whey |
| US8840947B2 (en) * | 2009-03-17 | 2014-09-23 | Separation Technologies Investments Limited | Whey or raw milk demineralisation and fractionation |
| US20120121788A1 (en) * | 2009-03-17 | 2012-05-17 | Separation Technologies Investments Limited | Isolation and purification of components of whey |
| US20120121781A1 (en) * | 2009-03-17 | 2012-05-17 | Separation Technologies Investments Limited | Whey or raw milk demineralisation and fractionation |
| KR101938604B1 (en) | 2011-02-18 | 2019-01-15 | 도레이 카부시키가이샤 | Method for producing sugar solution |
| WO2012151300A2 (en) | 2011-05-02 | 2012-11-08 | Verasonics, Inc. | Enhanced ultrasound image formation using qualified regions of overlapping transmit beams |
| US20140039237A1 (en) * | 2012-08-06 | 2014-02-06 | Ming-Hsin Li | Medication dispensing device |
| CN110272461A (en) * | 2019-06-29 | 2019-09-24 | 赤峰蒙广生物科技有限公司 | A method of purifying beta-thymidine from fermentation liquid |
Also Published As
| Publication number | Publication date |
|---|---|
| WO2007056191A2 (en) | 2007-05-18 |
| US20120083600A1 (en) | 2012-04-05 |
| WO2007056191A3 (en) | 2007-10-25 |
| US8841439B2 (en) | 2014-09-23 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8841439B2 (en) | Nucleotide sugar purification using membranes | |
| US9493499B2 (en) | Process for the production of purified cytidinemonophosphate-sialic acid-polyalkylene oxide (CMP-SA-PEG) as modified nucleotide sugars via anion exchange chromatography | |
| EP0931097B1 (en) | Carbohydrate purification using reverse osmosis and nanofiltration | |
| EP0820520B1 (en) | Improved enzymatic synthesis of oligosaccharides | |
| ES2991621T3 (en) | Separation of oligosaccharides from fermentation broth | |
| US6030815A (en) | Enzymatic synthesis of oligosaccharides | |
| JP2022550680A (en) | Separation of neutral oligosaccharides from fermentation broth | |
| CN1242776A (en) | Carbohybrate purification using ultrafiltration, reverse osmosis and nanofiltration | |
| KR102872519B1 (en) | Separation of oligosaccharides | |
| WO2018115309A1 (en) | Preparation of sugar-nucleotides | |
| BR112023018066B1 (en) | METHOD FOR PURIFICATION OF SIALO-OLIGOSACCHARIDES | |
| CA2218120C (en) | Improved enzymatic synthesis of oligosaccharides | |
| MXPA99003318A (en) | Carbohydrate purification using ultrafiltration, reverse osmosis and nanofiltration |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: NEOSE TECHNOLOGIES, INC., PENNSYLVANIA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:FELO, MICHAEL;DEFREES, SHAWN;REEL/FRAME:021086/0269;SIGNING DATES FROM 20080512 TO 20080519 |
|
| AS | Assignment |
Owner name: NOVO NORDISK A/S, DENMARK Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:NEOSE TECHNOLOGIES, INC.;REEL/FRAME:022441/0937 Effective date: 20090127 Owner name: NOVO NORDISK A/S,DENMARK Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:NEOSE TECHNOLOGIES, INC.;REEL/FRAME:022441/0937 Effective date: 20090127 |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |