WO2024105104A1 - Microbiocidal tetrahydroisoquinoline derivatives - Google Patents
Microbiocidal tetrahydroisoquinoline derivatives Download PDFInfo
- Publication number
- WO2024105104A1 WO2024105104A1 PCT/EP2023/081908 EP2023081908W WO2024105104A1 WO 2024105104 A1 WO2024105104 A1 WO 2024105104A1 EP 2023081908 W EP2023081908 W EP 2023081908W WO 2024105104 A1 WO2024105104 A1 WO 2024105104A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- methyl
- hydrogen
- compounds
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing three or more hetero rings
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/80—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,2
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/82—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with three ring hetero atoms
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01P—BIOCIDAL, PEST REPELLANT, PEST ATTRACTANT OR PLANT GROWTH REGULATORY ACTIVITY OF CHEMICAL COMPOUNDS OR PREPARATIONS
- A01P3/00—Fungicides
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D405/00—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom
- C07D405/14—Heterocyclic compounds containing both one or more hetero rings having oxygen atoms as the only ring hetero atoms, and one or more rings having nitrogen as the only ring hetero atom containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D409/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms
- C07D409/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having sulfur atoms as the only ring hetero atoms containing three or more hetero rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D417/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00
- C07D417/14—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and sulfur atoms as the only ring hetero atoms, not provided for by group C07D415/00 containing three or more hetero rings
Definitions
- the present invention relates to microbiocidal tetrahydroisoquinoline derivatives, e.g., as active ingredients, which have microbiocidal activity, in particular fungicidal activity.
- the invention also relates to preparation of these tetrahydroisoquinoline derivatives, to intermediates useful in the preparation of these tetrahydroisoquinoline derivatives, to the preparation of these intermediates, to agrochemical compositions which comprise at least one of the tetrahydroisoquinoline derivatives, to preparation of these compositions and to the use of the tetrahydroisoquinoline derivatives or compositions in agriculture or horticulture for controlling or preventing infestation of plants, harvested food crops, seeds or non-living materials by phytopathogenic microorganisms, in particular fungi.
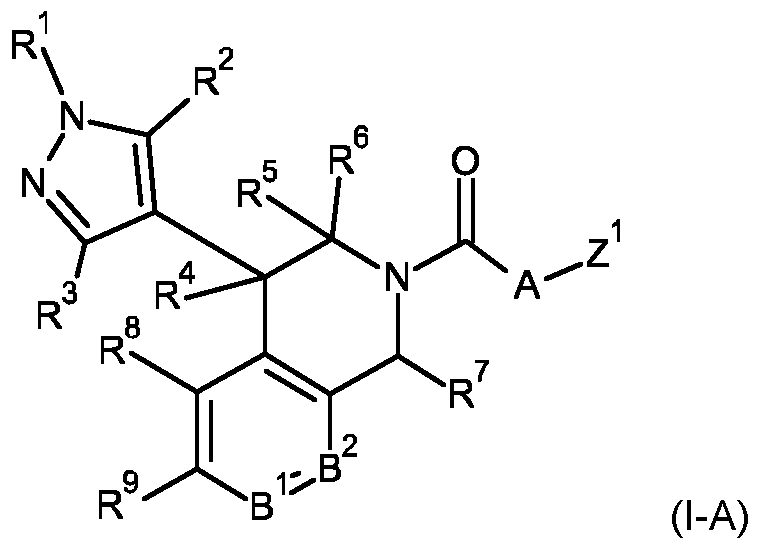
- R 1 is selected from hydrogen, Ci-C4-alkyl, C2-C4-alkenyl, C2-C4-alkynyl, or Cs-Cecycloalkyl;
- R 2 is selected from hydrogen, halogen, Ci-C4-alkyl, C2-C4-alkenyl, C2-C4-alkynyl, Ci-C4-haloalkyl, C3- Cecycloalkyl, Ci-C4-alkylcarbonyl, N-Ci-C4-alkoxy-C-Ci-C4-alkyl-carbonimidoyl, N-hydroxy-C-Ci-C4-alkyl- carbonimidoyl, or Ci-C4-alkoxycarbonyl;
- R 3 is selected from hydrogen, halogen, C1-C4 haloalkyl, or C1-C4 alkyl;
- R 4 is selected from hydrogen, halogen, C1-C4 haloalkyl, C3-C6 cycloalkyl, or C1-C4 alkyl;
- R 5 and R 6 are independently selected from hydrogen, or Ci-C4-alkyl
- R 7 is selected from hydrogen, Ci-C4-lkyl, Ci-C4-alkylcarbonyl, N-Ci-C4-alkoxy-C-Ci-C4-alkyl-carbonimidoyl, N- hydroxy-C-Ci-C4-alkyl-carbonimidoyl, Ci-C4-alkoxycarbonyl, N-methoxy-N-methyl-carbonyl, C1-C4 alkylaminocarbonyl, di (Ci-C4-alkyl)aminocarbonyl, phenyl, a 5- or 6-membered heteroaryl, or Cs-Ce-cycloalkyl; wherein any of said 5- or 6-membered heteroaryl contains 1 , 2, 3 or 4 heteroatoms independently selected from N, O or S, with the proviso that no more than one is O or S; and wherein any of said phenyl and 5- or 6- membered heteroaryl are unsubstituted or substituted with 1
- B 1 is selected from CR 10 , or N;
- B 2 is selected from CR 11 , or N;
- R 8 , R 9 , R 10 and R 11 are independently selected from hydrogen, halogen, C1-C4 alkyl, C1-C4 haloalkyl, C1-C4 alkoxy, Ci-C4-haloalkoxy, C2-C4-alkenyloxy, C2-C4-alkynyloxy, Ci-C4-alkylsulfanyl, Ci-C4-alkylsulfinyl, C1-C4 alkylsulfonyl, C1-C4 alkoxy-Ci-C4 alkyl, N-Ci-C4-alkylamino, N,N-di(Ci-C4 alkyl)amino, C1-C4 alkoxycarbonyl, C1-C4 alkylcarbonyl, Ci-C4-alkylaminocarbonyl, di(Ci-C4-alkyl)aminocarbonyl, cyano-Ci-C4alkyl, N
- a 1 , A 2 and A 3 are independently selected from CR 12 , N, NR 13 , O or S, with the proviso that at least one of A 1 , A 2 and A 3 is selected from N, O or S, and that no more than one of A 1 , A 2 and A 3 is O or S;
- R 12 , R 13 are independently selected from hydrogen, halogen, C1-C4 alkyl, Ci-C4haloalkyl, C2-C4 alkenyl, or C2- C4 alkynyl; and
- Z 1 is selected from 6-membered heteroaryl containing 1 or 2 heteroatoms selected from N; wherein any of said 6-membered heteroaryl is unsubstituted or substituted with 1 , 2 or 3 substituents independently selected from halogen, C1-C4 haloalkyl, cyano, C1-C4 alkyl, C2-C4 alkynyl, C1-C4 haloalkoxy, C1-C4 alkoxy, C1-C4 alkylsulfanyl, C1-C4 alkylsulfinyl, or Ci-C4 alkylsulfonyl; or an agrochemically acceptable salt, stereoisomer, or N-oxide thereof.
- an agrochemical composition comprising a fungicidally effective amount of a compound of formula (I) according to the invention.
- Such an agricultural composition may further comprise at least one additional active ingredient and/or an agrochemically- acceptable diluent or carrier.
- a method of controlling or preventing infestation of useful plants by phytopathogenic microorganisms wherein a fungicidally effective amount of a compound of formula (I) according to the invention, or a composition comprising the compound of formula (I), is applied to the plants, to parts thereof or the locus thereof.
- a compound of formula (I) according to the invention as a fungicide.
- the use may exclude methods for treatment of the human or animal body by surgery or therapy and diagnostic methods practised on the human or animal body.
- Compounds of formula (I) which have at least one basic centre can form, for example, acid addition salts, for example with strong inorganic acids such as mineral acids, for example perchloric acid, sulfuric acid, nitric acid, nitrous acid, a phosphorus acid or a hydrohalic acid, with strong organic carboxylic acids, such as Ci- C4alkanecarboxylic acids which are unsubstituted or substituted, for example by halogen, for example acetic acid, such as saturated or unsaturated dicarboxylic acids, for example oxalic acid, malonic acid, succinic acid, maleic acid, fumaric acid or phthalic acid, such as hydroxycarboxylic acids, for example ascorbic acid, lactic acid, malic acid, tartaric acid or citric acid, or such as benzoic acid, or with organic sulfonic acids, such as Ci- 04 alkane- or arylsulfonic acids which are unsubstituted or substituted, for example
- Compounds of formula (I) which have at least one acidic group can form, for example, salts with bases, for example mineral salts such as alkali metal or alkaline earth metal salts, for example sodium, potassium or magnesium salts, or salts with ammonia or an organic amine, such as morpholine, piperidine, pyrrolidine, a mono-, di- or tri-lower-alkylamine, for example ethyl-, diethyl-, triethyl- or dimethylpropylamine, or a mono-, di- or trihydroxy-lower-alkylamine, for example mono-, di- or triethanolamine.
- bases for example mineral salts such as alkali metal or alkaline earth metal salts, for example sodium, potassium or magnesium salts
- salts with ammonia or an organic amine such as morpholine, piperidine, pyrrolidine, a mono-, di- or tri-lower-alkylamine, for example ethyl-, die
- the compounds of formula (I) according to the invention are in free form, in oxidized form as a N-oxide or in salt form, e.g., an agronomically usable salt form.
- N-oxides are oxidized forms of tertiary amines or oxidized forms of nitrogen containing heteroaromatic compounds. They are described for instance in the book “Heterocyclic N-oxides” by A. Albini and S. Pietra, CRC Press, Boca Raton 1991.
- the compounds of formula (I) according to the invention also include hydrates which may be formed during the salt formation.
- Ci-Cealkyl substituted by 1 , 2 or 3 halogens may include, but not be limited to, -CH2CI, -CHCI2, -CCh, -CH2F, -CHF2, - CF3, -CH2CF3 or -CF2CH3 groups.
- Ci-Cealkoxy substituted by 1 , 2 or 3 halogens may include, but not be limited to, CH2CIO-, CHCI2O-, CCI3O-, CH2FO-, CHF2O-, CF3O-, CF3CH2O- or CH3CF2O- groups.
- optionally substituted as used herein, can be used interchangeably with the term “unsubstituted or substituted”.
- halogen refers to fluorine (fluoro), chlorine (chloro), bromine (bromo), or iodine (iodo), preferably fluorine, chlorine, or bromine. This also applies, correspondingly, to halogen in combination with other meanings, such as haloalkyl, haloalkenyl, haloalkynyl, haloalkoxy, and halocycloalkyl.
- amino means a -NH2 group.
- cyano means a -CN group.
- hydroxyl or “hydroxy” means an -OH group.
- carboxylic acid means a -COOH group.
- Ci-C n -alkyl refers to a saturated straight-chain or branched hydrocarbon radical attached via any of the carbon atoms having 1 to n carbon atoms, for example, any one of the radicals methyl, ethyl, n-propyl, 1 -methylbutyl, 2-methylbutyl, 3-methylbutyl, 2, 2-dimethylpropyl, 1 -ethylpropyl, n-hexyl, n- pentyl, 1 ,1 -dimethylpropyl, 1 , 2-dimethylpropyl, 1 -methylpentyl, 2-methylpentyl, 3-methylpentyl, 4-methylpentyl, 1 ,1 -dimethylbutyl, 1 ,2-dimethylbutyl, 1 ,3-dimethylbutyl, 2,2-dimethylbutyl, 2,3-dimethylbutyl, 3,3-dimethylbutyl,
- C2-C n -alkenyl refers to a straight or branched alkenyl chain moiety having from two to n carbon atoms and one or two double bonds, for example, ethenyl, prop-1 -enyl, but-2-enyl.
- C2-C n -alkynyl refers to a straight or branched alkynyl chain moiety having from two to n carbon atoms and one triple bond, for example, ethynyl, prop-2-ynyl, but-3-ynyl,
- Cs-Cn-cycloalkyl refers to three (3) to n membered cycloalkyl radical such as cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl.
- Ci-C n -alkoxy refers to a straight-chain or branched saturated alkyl radical having one (1) to n carbon atoms (as mentioned above) which is attached via an oxygen atom, i.e., for example, any one of the radicals methoxy, ethoxy, n-propoxy, 1 -methylethoxy, n-butoxy, 1 -methylpropoxy, 2-methylpropoxy and 1 ,1 -dimethylethoxy.
- C2-Cn-alkenyloxy refers to a straight-chain or branched alkenyl chain having two (2) to n carbon atoms (as mentioned above) which is attached via an oxygen atom.
- C2-Cn alkynyloxy refers to a radical of the formula -OR a where R a is a C2-Cn alkynyl radical as generally defined above.
- Ci-C n -alkoxy-Ci-C n -alkyl refers to an alkyl radical (as mentioned above) substituted with a Ci-Cn-alkoxy group. Examples are methoxymethyl, methoxyethyl, ethoxymethyl and propoxymethyl.
- C3-C n -cycloalkyl-Ci-C n -alkyl refers to an alkyl radical (as mentioned above) substituted with a Cs-Cn-cycloalkyl group. Examples are cyclopropylmethyl, cyclopropylethyl.
- C3-C n -halocycloalkyl-Ci-C n -alkyl refers to an alkyl radical substituted with cycloalkyl group, wherein the cycloalkyl group is substituted by one or more of the same or different halogen atoms. Examples are 3,3- difluorobutylmethyl and 1 -chlorocyclopropylmethyl.
- Ci-C n -haloalkyl refers to a straight-chain or branched saturated alkyl radical attached via any of the carbon atoms having 1 to n carbon atoms (as mentioned above), where some or all of the hydrogen atoms in these radicals may be replaced by fluorine, chlorine, bromine and/or iodine, i.e., for example, any one of chloromethyl, dichloromethyl, trichloromethyl, fluoromethyl, difluoromethyl, trifluoromethyl, chlorofluoromethyl, dichlorofluoromethyl, chlorodifluoromethyl, 2-fluoroethyl, 2-chloroethyl, 2- bromoethyl, 2-iodoethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, 2-chloro-2-fluoroethyl, 2-chloro-2,2-difluoroe
- Ci-C2fluoroalkyl would refer to a Ci-C2alkyl radical which carries 1 , 2, 3, 4, or 5 fluorine atoms, for example, any one of difluoromethyl, trifluoromethyl, 1- fluoroethyl, 2-fluoroethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, 1 ,1 ,2,2-tetrafluoroethyl or pentafluoroethyl.
- C2-C n -haloalkenyl or “C2-C n -haloalkynyl” as used herein refers to a C2-C n -alkenyl or C2- Cn-alkynyl radical respectively substituted with one or more halogen atoms which may be the same or different.
- Cs-Cn-halocycloalkyl or “Ci-C n -haloalkoxy” as used herein refers to a Cs-Cn-cycloalkyl radical or Ci-C n -alkoxyl radical respectively substituted with one or more halo atoms which may be the same or different.
- cyano-Ci-C n -alkyl refers to Ci-C n -alkyl radical having 1 to n carbon atoms (as mentioned above), where one of the hydrogen atoms in the radical is be replaced by a cyano group: for example, cyano-methyl, 2-cyano-ethyl, 2-cyano-propyl, 3-cyano-propyl, 1-(cyano-methyl)-2-ethyl, 1-(methyl)- 2-cyano-ethyl, 4-cyanobutyl, and the like.
- cyano-Cs-Cn-cycloalkyl refers to a C3-C n - cycloalkyl radical substituted with one of the hydrogen atoms by a cyano group
- cyano-Cs-Cn- cycloalkyl-Ci-C n -alkyl refers to an Ci-C n -alkyl radical having a cyano-Cs-Cn-cycloalkyl group.
- Ci-Cn-alkylthio“ or “Ci-Cn-alkylsulfanyl“ refers to a Ci-Cn-alkyl group linked through a sulfur atom.
- Ci-C n -haloalkylthio“ or “Ci-C n -haloalkylsulfanyl“ refers to a Ci-C n haloalkyl group linked through a sulfur atom.
- Ci-C n -alkylsulfonyl-Ci-C n -alkyl refers to an a Ci-C n alkyl radical substituted with a Ci-C n alkylsulfonyl group.
- Ci-C n -alkoxycarbonyl-Ci-C n -alkyl refers to a Ci-C n -alkyl radical substituted by a “C1- Cn-alkoxycarbonyl group.
- N-Ci-C n alkylamino refers to a radical of the formula -NH-R a where R a is a Ci-C n alkyl radical as defined above.
- N,N-di(Ci-C n alkyl)amino refers to a radical of the formula -N(R a )R a where each R a is a Ci-C n alkyl radical, which may be the same or different, as defined above.
- the Ci-C n -alkoxy group linked to the nitrogen may be substituted.
- heteroaryl refers to a 5- or 6-membered aromatic monocyclic ring radical which comprises 1 , 2, 3 or 4 heteroatoms independently selected from N, O or S.
- heteroaryl include, but are not limited to, furanyl, pyrrolyl, thienyl, pyrazolyl, imidazolyl, thiazolyl, isothiazolyl, oxazolyl, isoxazolyl, triazolyl, tetrazolyl, pyrazinyl, pyridazinyl, pyrimidyl or pyridyl.
- heteroaryl-Ci-C n -alkyl or “heteroaryl- Cs-Cn-cycloalkyl” refers to an Ci-C n -alkyl or Cs-Cn-cycloalkyl radical respectively substituted by a heteroaryl group.
- the heteroaryl-Ci-C n -alkyl or heteroaryl-Cs-Cn-cycloalkyl radical may be substituted on heteroaryl, alkyl and/or cycloalkyl group as appropriate.
- controlling refers to reducing the number of pests, eliminating pests and/or preventing further pest damage such that damage to a plant or to a plant derived product is reduced.
- pest refers to insects, and molluscs that are found in agriculture, horticulture, forestry, the storage of products of vegetable origin (such as fruit, grain, and timber); and those pests associated with the damage of man-made structures.
- the term pest encompasses all stages in the life cycle of the pest.
- the term "effective amount” refers to the amount of the compound, or a salt thereof, which, upon single or multiple applications provides the desired effect.
- an effective amount is readily determined by the skilled person in the art, using known techniques and by observing results obtained under analogous circumstances. In determining the effective amount, a number of factors are considered including, but not limited to the type of plant or derived product to be applied; the pest to be controlled and its lifecycle; the particular compound applied; the type of application; and other relevant circumstances.
- room temperature or “RT” or “rt” or “ambient temperature” refer to a temperature of about 15° C to about 35° C.
- rt can refer to a temperature of about 20° C to about 30° C.
- R 1 is selected from hydrogen, Ci-C4-alkyl, C2-C4-alkenyl, C2-C4-alkynyl, or Cs-Cecycloalkyl.
- R 1 is Ci-C4alkyl.
- R 1 is methyl, ethyl or isopropyl. More preferably, R 1 is methyl.
- R 2 is selected from hydrogen, halogen, Ci-C4-alkyl, C2-C4-alkenyl, C2-C4- alkynyl, Ci-C4-haloalkyl, Cs-Cecycloalkyl, Ci-C4-alkylcarbonyl, N-C1-C4 alkoxy-C-Ci-C4 alkyl-carbonimidoyl, N- hydroxy-C-Ci-C4 alkyl-carbonimidoyl, or Ci-C4-alkoxycarbonyl.
- R 2 is hydrogen, halogen, Ci-C4-alkyl, Cs-Cecycloalkyl, Ci-C4alkylcarbonyl, N-Ci-C4alkoxy-Ci-C4 alkyl-carbonimidoyl, or N-hydroxy-Ci-C4 alkyl-carbonimidoyl.
- R 2 is hydrogen, halogen, methyl, ethyl, cyclopropyl, C1-C2 alkylcarbonyl, N-C1-C2 alkoxy-C-Ci-C2 alkyl-carbonimidoyl, or N-hydroxy-C-Ci-C2 alkyl-carbonimidoyl.
- R 2 is hydrogen, halogen, or C1-C4 alkyl. Most preferably R 2 is hydrogen, chlorine, or methyl. In one preferred embodiment R 2 is hydrogen. In another preferred embodiment R 2 is methyl. In still another preferred embodiment R 2 is chlorine.
- R 3 is selected from hydrogen, halogen, C1-C4 haloalkyl, or C1-C4 alkyl.
- R 3 is hydrogen or C1-C4 alkyl. More preferably R 3 is hydrogen or methyl. Most preferably R 3 is hydrogen.
- R 4 is selected from hydrogen, halogen, C1-C4 alkyl, C1-C4 haloalkyl, or Cs- Ce-cycloalkyl.
- R 4 is hydrogen, chlorine, bromine, fluorine, methyl, ethyl, trifluoromethyl, difluoromethyl, or cyclopropyl. More preferably R 4 is hydrogen, chlorine, bromine, or methyl. Still more preferably R 4 is hydrogen or methyl. Most preferably R 4 is hydrogen. In one embodiment of the invention R 4 is hydrogen. In another embodiment of the invention R 4 is methyl.
- R 5 and R 6 are independently selected from hydrogen or Ci-C4-alkyl.
- R 5 and R 6 are independently selected from hydrogen, methyl, or ethyl. More preferably, R 5 and R 6 are independently selected from hydrogen or methyl. Even more preferably, R 5 and R 6 are hydrogen.
- R 7 is selected from hydrogen, Ci-C4alkyl, Ci-C4-alkylcarbonyl, N-C1-C4 alkoxy-C-Ci-C4 alkyl-carbonimidoyl, N-hydroxy-C-Ci-C4-alkyl-carbonimidoyl, Ci-C4-alkoxycarbonyl, N-methoxy-N-methyl- carbonyl, Ci-C4alkylaminocarbonyl, di(Ci-C4alkyl)aminocarbonyl, phenyl, a 5- or 6-membered heteroaryl, or Cs-Ce-cycloalkyl; wherein any of said 5- or 6-membered heteroaryl conatins 1 , 2, 3 or 4 heteroatoms independently selected from N, O or S, with the proviso that no more than one is O or S; and wherein any of said phenyl and 5- or 6-membered heteroaryl are unsubstituted or substituted with 1 ,
- R 7 is selected from hydrogen, Ci-C4alkyl, C3- Ci-C4alkylcarbonyl, Ci- 04 alkoxycarbonyl, N-C1-C4 alkoxy-C-Ci-C4alkyl-carbonimidoyl, N-hydroxy-C-Ci-C4 alkyl-carbonimidoyl, N- methoxy-N-methyl-carbonyl, C1-C4 alkylaminocarbonyl, di(Ci-C4alkyl)aminocarbonyl, phenyl, a 5- or 6- membered heteroaryl, or Cs-Ce-cycloalkyl; wherein any of said 5- or 6-membered heteroaryl contains 1 heteroatom selected from N; and wherein any of said phenyl and 5- or 6-membered heteroaryl are unsubstituted or substituted with 1 or 2 substituents independently selected from halogen, Ci-C4haloalkyl, cyano, or Ci-
- R 7 is selected from hydrogen, C1-C4 alkyl, or Cs-Ce-cycloalkyl.
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl. Even more preferably R 7 is hydrogen, methyl, or cyclopropyl.
- R 7 is C1-C4 alkyl.
- R 7 is methyl or ethyl. More preferably R 7 is methyl.
- R 8 , R 9 , R 10 and R 11 are independently selected hydrogen, halogen, C1-C4 alkyl, C1-C4 haloalkyl, C1-C4 alkoxy, C1-C4 haloalkoxy, C2-C4alkenyloxy, C2-C4 alkynyloxy, C1-C4 alkylsulfanyl, C1-C4 alkylsulfinyl, C1-C4 alkylsulfonyl, Ci-C4 alkoxy-Ci-C4 alkyl, N- C1-C4 alkylamino, N,N-di C1-C4 alkylamino, C1-C4 alkoxycarbonyl, C1-C4 alkylcarbonyl, C1-C4 alkylamin
- R 8 and R 9 are independently selected from hydrogen, halogen, cyano, Ci- 04 alkyl, or C1-C4 alkoxy.
- R 8 and R 9 are independently selected from hydrogen, halogen, methyl, methoxy, or cyano. More preferably, R 8 and R 9 are independently selected from hydrogen, methyl, chlorine, fluorine, bromine, or methoxy. Even more preferably, R 8 and R 9 are independently selected from hydrogen or methoxy.
- R 8 is hydrogen, halogen, or cyano.
- R8 is hydrogen, bromine, chlorine, or cyano. More preferably, R 8 is hydrogen, cyano or bromine. Even more preferably, R 8 is hydrogen.
- R 9 is selected from hydrogen, halogen, C1-C3 alkyl, C1-C2 haloalkyl, C1-C3 haloalkoxy, Ci- 04 alkoxy, Ci-Csalkenyloxy, Ci-Csalkynyloxy, Ci-C2alkylsulfanyl, C1-C2 alkylsulfinyl, C1-C2 alkylsulfonyl, C1-C2 alkoxy-Ci-C2 alkyl, Ci-Csalkoxycarbonyl, Ci-C2alkylcarbonyl, Ci-C2alkylaminocarbonyl, di(Ci- C2alkyl)aminocarbonyl, cyano-Ci-C2alkyl, N-C1-C2 alkoxy-C-Ci-C2 alkyl-carbonimidoyl, N-hydroxy-C-Ci- C2alkyl-carbonimidoyl, hydroxy
- R 9 is hydrogen, chloro, bromo, fluoro, cano, methyl, methoxy, propoxy, allyloxy, methoxymethyl, 2- methoxyethoxymethyl, phenyl, 2-cyanophenyl, 3-cyanophenyl, 4-cyanophenyl, [4-(trifluoromethyl)pyrazol-1- yl], [3-(trifluoromethyl)pyrazol-1-yl], 3-cyanopyrazol-1-yl, 4-cyanopyrazol-1-yl, 5-chloropyrazol-1-yl, 4- chloropyrazol-1-yl, 3-chloropyrazol-1-yl, 5-fluoropyrazol-1-yl, 4-fluoropyrazol-1-yl, 3-fluoropyrazol-1-yl, 3,5- dimethylpyrazol-1-yl, 5-methylpyrazol-1-yl, 4-methylpyrazol-1-yl, 3-methylpyrazol-1-yl, pyrazol-1-yl
- B 1 is CR 10 and B 2 is CR 11 or B 1 is N and B 2 is CR 11 or B 1 is CR 10 and B 2 is N.
- B 1 is CR 10 and B 2 is CR 11 .
- R 10 and R 11 are independently selected from hydrogen, halogen, hydroxy, cyano, amino, C1-C4 alkyl, C1-C4 haloalkyl, C1-C4 alkoxy, C1-C4 haloalkoxy, C2-C4alkenyloxy, C2-C4 alkynyloxy, C1-C4 alkylsulfanyl, C1-C4 alkylsulfinyl, C1-C4 alkylsulfonyl, Ci-C4 alkoxy-Ci-C4 alkyl, N- C1-C4 alkylamino, N,N- di C1-C4 alkylamino, C1-C4 alkoxycarbonyl, C1-C4 alkylcarbonyl, Ci-C2alkylaminocarbonyl, di(Ci- C2alkyl)aminocarbonyl, cyano-Ci-C4alkyl, N-C1
- R 10 and R 11 are independently selected from hydrogen, chloro, bromo, fluoro, cyano, methyl, methoxy, propoxy, allyloxy, methoxymethyl, 2-methoxyethoxymethyl, phenyl, 2-cyanophenyl, 3-cyanophenyl, 4-cyanophenyl, [4- (trifluoromethyl)pyrazol-l-yl], [3-(trifluoromethyl)pyrazol-1-yl], 3-cyanopyrazol-1-yl, 4-cyanopyrazol-1-yl, 5- chloropyrazol-1-yl, 4-chloropyrazol-1-yl, 3-chloropyrazol-1-yl, 5-fluoropyrazol-1-yl, 4-fluoropyrazol-1-yl, 3- fluoropyrazol-1-yl, 3,5-dimethylpyrazol-1-yl, 5-methylpyrazol-1-yl, 4-methylpyrazol-1-yl, 3-methylpyrazol-1-yl,
- R 10 and R 11 are independently selected from hydrogen, chlorine, bromine, or cyano.
- R 10 and R 11 are independently selected from hydrogen, halogen, C1-C3 alkyl, or Ci-Cs alkoxy.
- R 10 and R 11 are independently selected from hydrogen or halogen. More preferably R 10 and R 11 are hydrogen, bromine, or chlorine. Even more preferably R 10 and R 11 are hydrogen.
- a 1 , A 2 and A 3 are independently selected from CR 12 , N, NR 13 , O or S, with the proviso that at least one of A 1 , A 2 and A 3 is selected from N, O or S, and that no more than one of A 1 , A 2 and A 3 is O or S.
- a 1 and A 2 are independently selected from CR 12 , N or O
- a 3 is CR 12 , N, O or S, with the proviso that at least one of A 1 , A 2 and A 3 is selected from N, O or S, and that no more than one of A 1 , A 2 and A 3 is O or S.
- a 1 and A 2 are independently selected from N or O; and A 3 is CR 12 , O or S, with the proviso that at least one of A 1 , A 2 and A 3 is N or O, and that no more than one of A 1 , A 2 and A 3 is O.
- R 12 is hydrogen or C1-C4 alkyl.
- R 12 is hydrogen or methyl. More preferably R 12 is hydrogen.
- R 13 is hydrogen or C1-C4 alkyl.
- R 13 is hydrogen or methyl. More preferably R 13 is hydrogen.
- Z 1 is selected from a 6-membered heteroaryl; wherein any of said 6- membered heteroaryl contains 1 or 2 heteroatoms selected from N; and wherein any of said 6-membered heteroaryl is unsubstituted or substituted with 1 , 2 or 3 substituents independently selected from halogen, cyano, C1-C4 alkyl, C2-C4 alkynyl, C1-C4 haloalkyl, C1-C4 alkoxy, C1-C4 haloalkoxy, C1-C4 alkylsulfanyl, C1-C4 alkylsulfinyl, or C1-C4 alkylsulfonyl.
- Z 1 is selected from a 6-membered heteroaryl, wherein any of said 6-membered heteroaryl contains 1 or 2 heteroatoms selected from N; and wherein any of said 6-membered heteroaryl is unsubstituted or substituted with 1 or 2 substituents independently selected from halogen, cyano, C1-C4 alkyl, C1-C4 haloalkyl, C1-C4 haloalkoxy, or C1-C4 alkoxy.
- Z 1 is selected from a 6-membered heteroaryl, wherein any of said 6-membered heteroaryl contains 1 or 2 heteroatoms selected from N; and wherein any of said 6-membered heteroaryl is unsubstituted or substituted with 1 or 2 substituents independently selected from halogen or C1-C4 haloalkyl. More preferably Z 1 is selected from a 6-membered heteroaryl, wherein any of said 6-membered heteroaryl contains 1 or 2 heteroatoms selected from N; and wherein any of said 6-membered heteroaryl is unsubstituted or substituted with 1 or 2 substituents selected from fluorine.
- Z 1 is selected from a 6-membered heteroaryl; wherein any of said 6- membered heteroaryl contains 1 heteroatom selected from N; and wherein any of said 6-membered heteroaryl is unsubstituted or substituted with 1 or 2 substituents independently selected from halogen, cyano, C1-C4 alkyl, C1-C4 haloalkyl, C1-C4 haloalkoxy, or C1-C4 alkoxy.
- Z 1 is selected from a 6-membered heteroaryl, wherein any of said 6-membered heteroaryl contains 1 heteroatom selected from N; and wherein any of said 6-membered heteroaryl is unsubstituted or substituted with 1 or 2 substituents independently selected from halogen or C1-C4 haloalkyl. More preferably Z 1 is selected from a 6-membered heteroaryl; wherein any of said heteroaryl contains 1 heteroatom selected from N; and wherein any of said 6-membered heteroaryl is unsubstituted or substituted with 1 or 2 substituents selected from fluorine.
- Z 1 is selected from 2-pyridyl, 3-pyridyl, 4-pyridyl, pyrazin-2-yl, pyridazin-3-yl, pyridazin- 4-yl, pyrimidin-2-yl, pyrimidin-4-yl, or pyrimidin-5-yl; wherein any of said pyridyl-, pyrazin- pyridazine, or pyrimidin-moities are unsubstituted or substituted with 1 , 2 or 3 substituents independently selected from halogen, C1-C4 haloalkyl, cyano, C1-C4 alkyl, C2-C4 alkynyl, C1-C4 haloalkoxy, C1-C4 alkoxy, C1-C4 alkylsulfanyl, C1-C4 alkylsulfinyl, or Ci-C4 alkylsulfonyl.
- Z 1 is selected from 2-pyridyl, 3-pyridyl, 4-pyridyl, pyrazin- 2-yl, pyridazin-3-yl, pyridazin-4-yl, pyrimidin-2-yl, pyrimidin-4-yl, or pyrimidin-5-yl; wherein any of said pyridyl-, pyrazin- pyridazine or pyrimidin-moities are unsubstituted or substituted with 1 , 2 or 3 substituents independently selected from halogen, C1-C4 haloalkyl, or C1-C4 alkyl.
- Z 1 is selected from 2- pyridyl, 3-pyridyl, 4-pyridyl, pyrazin-2-yl, pyridazin-3-yl, pyridazin-4-yl, pyrimidin-2-yl, pyrimidin-4-yl, or pyrimidin-5-yl; wherein any of said pyridyl-, pyrazin- pyridazine or pyrimidin-moities are unsubstituted or substituted with 1 , 2 or 3 substituents independently selected from fluorine, trifluoromethyl, difluoromethyl, or methyl.
- Z 1 is selected from 2-pyridyl, 3-pyridyl, 4-pyridyl, pyrazin-2-yl, pyridazin-3-yl, pyridazin- 4-yl, pyrimidin-2-yl, pyrimidin-4-yl, or pyrimidin-5-yl; wherein any of said pyridyl-, pyrazin- pyridazine or pyrimidin-moities are unsubstituted or substituted with 1 or 2 substituents selected from fluorine.
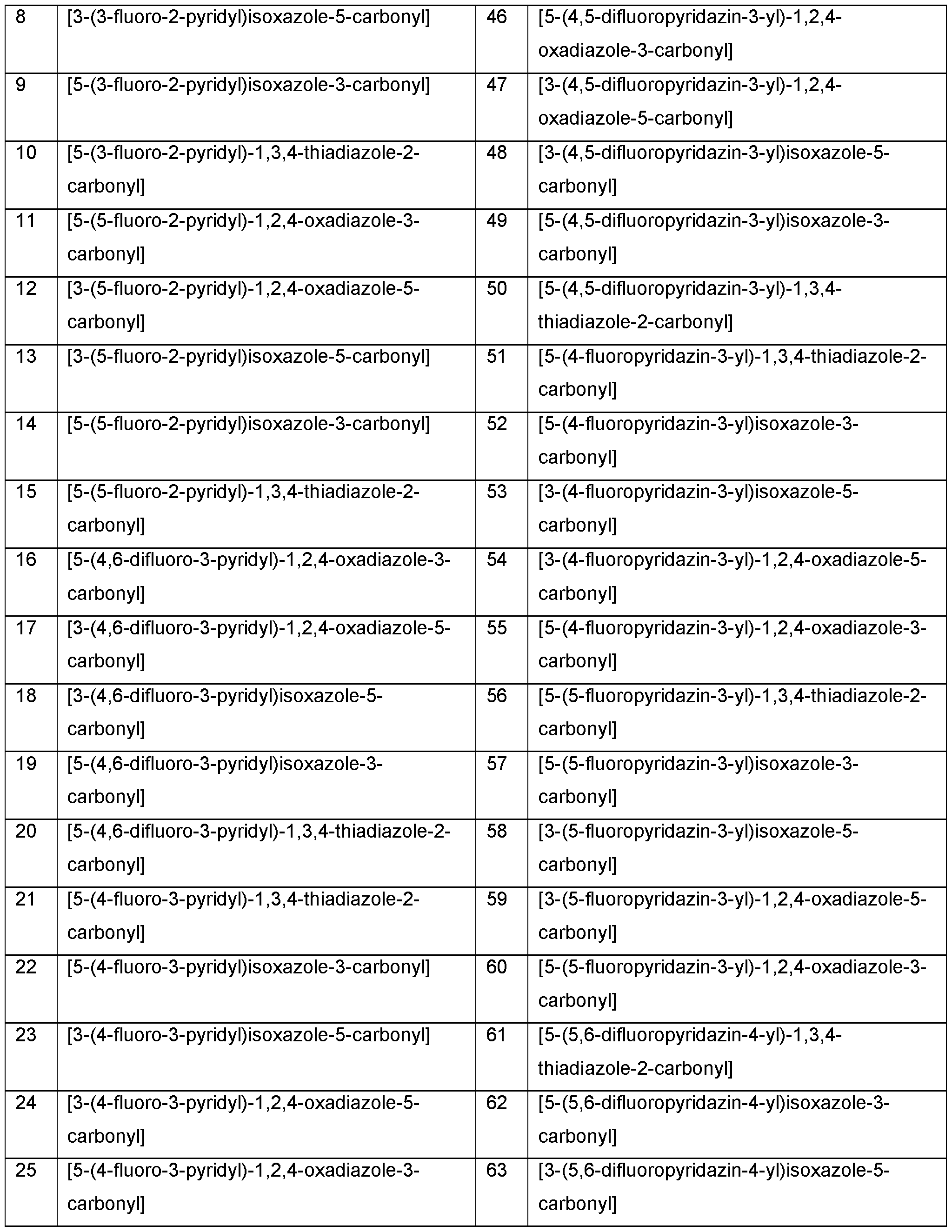
- Z 1 is selected from 5-fluoropyrimidin-4-yl, 3,6-difluoro- 2-pyridyl, 4,6-difluoro-2-pyridyl, 4,5-difluoro-2-pyridyl, 5,6-difluoro-2-pyridyl, 3-fluoro-4-pyridyl, 2-fluoro-4- pyridyl, 2,3-difluoro-4-pyridyl, 2,5-difluoro-4-pyridyl, 2,6-difluoro-4-pyridyl, 3,5-difluoro-4-pyridyl, 2,5-difluoro-4- pyridyl, 2-fluoro-3-pyridyl, 6-fluoro-3-pyridyl, 5-fluoro-3-pyridyl, 4-fluoro-3-pyridyl, 2,6-difluoro-3-pyridyl,
- Z 1 is selected from 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 6-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, 3,5-difluoro-2-pyridyl, 2-fluoro-4-pyridyl, 5-fluoropyrimidin-4-yl, 5-fluoropyrimidin-2-yl, 4-fluoropyridazin-3-yl, 5- fluoropyridazin-3-yl, 4,5-difluoropyridazin-3-yl, or 5-fluoropyridazin-4-yl.
- Z 1 is selected from 3- fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, 3,5-difluoro-2-pyridyl, 4-fluoropyridazin-3-yl, 4,5- difluoropyridazin-3-yl or 5-fluoropyridazin-4-yl. Most preferably Z 1 is selected from 3-fluoro-2-pyridyl, 5-fluoro- 2-pyridyl, 3,4-difluoro-2-pyridyl, or 3,5-difluoro-2-pyridyl.
- the present invention accordingly, makes available a compound of formula (I) having R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , B 1 , B 2 , A 1 , A 2 , A 3 and Z 1 as defined above in all combinations / each permutation.
- Embodiments according to the invention are provided as set out below.
- R 1 is methyl, R 2 is hydrogen, chlorine, or methyl;
- R 3 is hydrogen
- R 4 is hydrogen, or methyl
- R 5 and R 6 are hydrogen
- R 7 is hydrogen, C1-C4 alkyl, or Cs-Ce-cycloalkyl
- R 8 is hydrogen, bromine, chlorine, or cyano
- R 9 is hydrogen, bromine, chlorine, cyano, methyl, or methoxy
- B 1 is N or CR 10 , wherein R 10 is hydrogen, bromine, chlorine, or cyano;
- B 2 is N or CR 11 , wherein R 11 is hydrogen, bromine, chlorine, or cyano;
- a 1 , A 2 , A 3 and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- R 1 is methyl
- R 2 is hydrogen, chlorine, or methyl
- R 3 is hydrogen
- R 4 is hydrogen, or methyl
- R 5 and R 6 are hydrogen
- R 7 is hydrogen, C1-C4 alkyl, or Cs-Ce-cycloalkyl
- R 8 is hydrogen, bromine, chlorine, or cyano
- R 9 is hydrogen, bromine, chlorine, cyano, methyl, or methoxy
- B 1 is N or CR 10 , wherein R 10 is hydrogen, bromine, chlorine, or cyano;
- B 2 is N or CR 11 , wherein R 11 is hydrogen, bromine, chlorine, or cyano;
- Z 1 is 2-pyridyl, 3-pyridyl, 4-pyridyl, pyrazin-2-yl, pyridazin-3-yl, pyridazin-4-yl, pyrimidin-2-yl, pyrimidin-4-yl, or pyrimidin-5-yl; wherein any of said pyridyl-, pyrazin- pyridazine or pyrimidin-moities are unsubstituted or substituted with 1 or 2 substituents selected from fluorine; and
- a 1 , A 2 and A 3 are as defined for the compounds of formula (I) according to the present invention.
- the compound of formula (I) may be a compound of formula (l-A) wherein
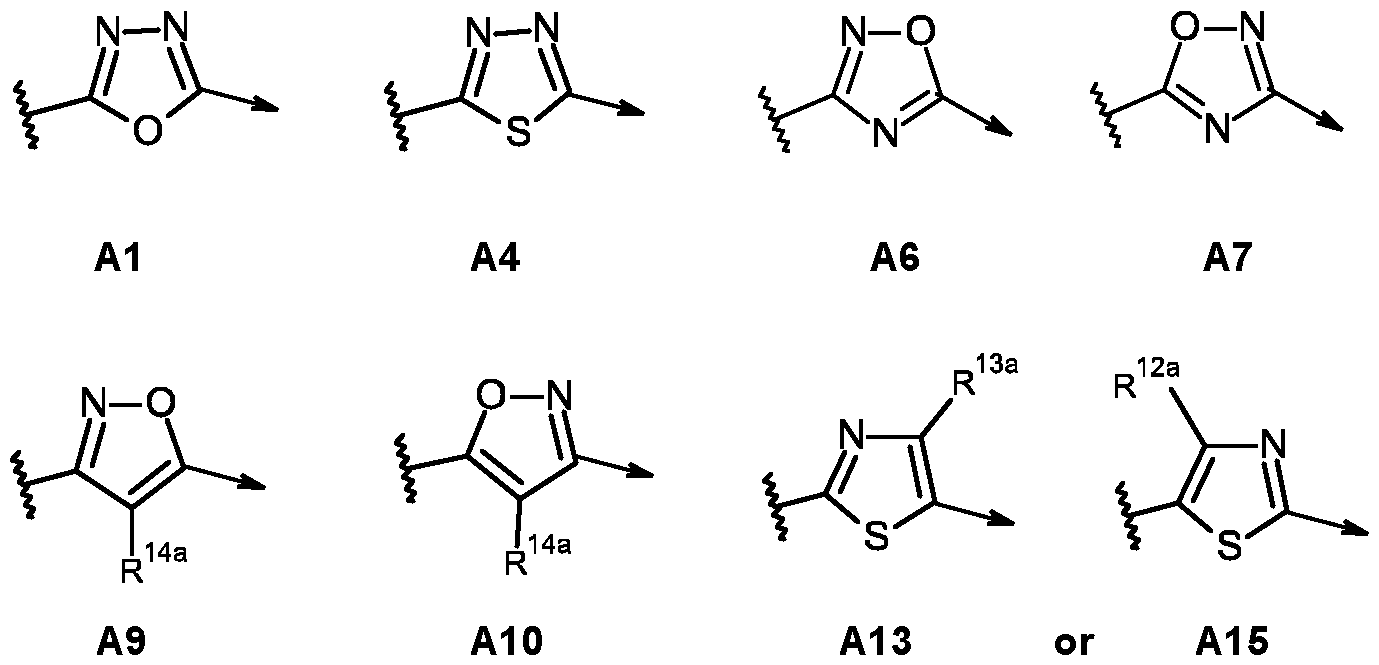
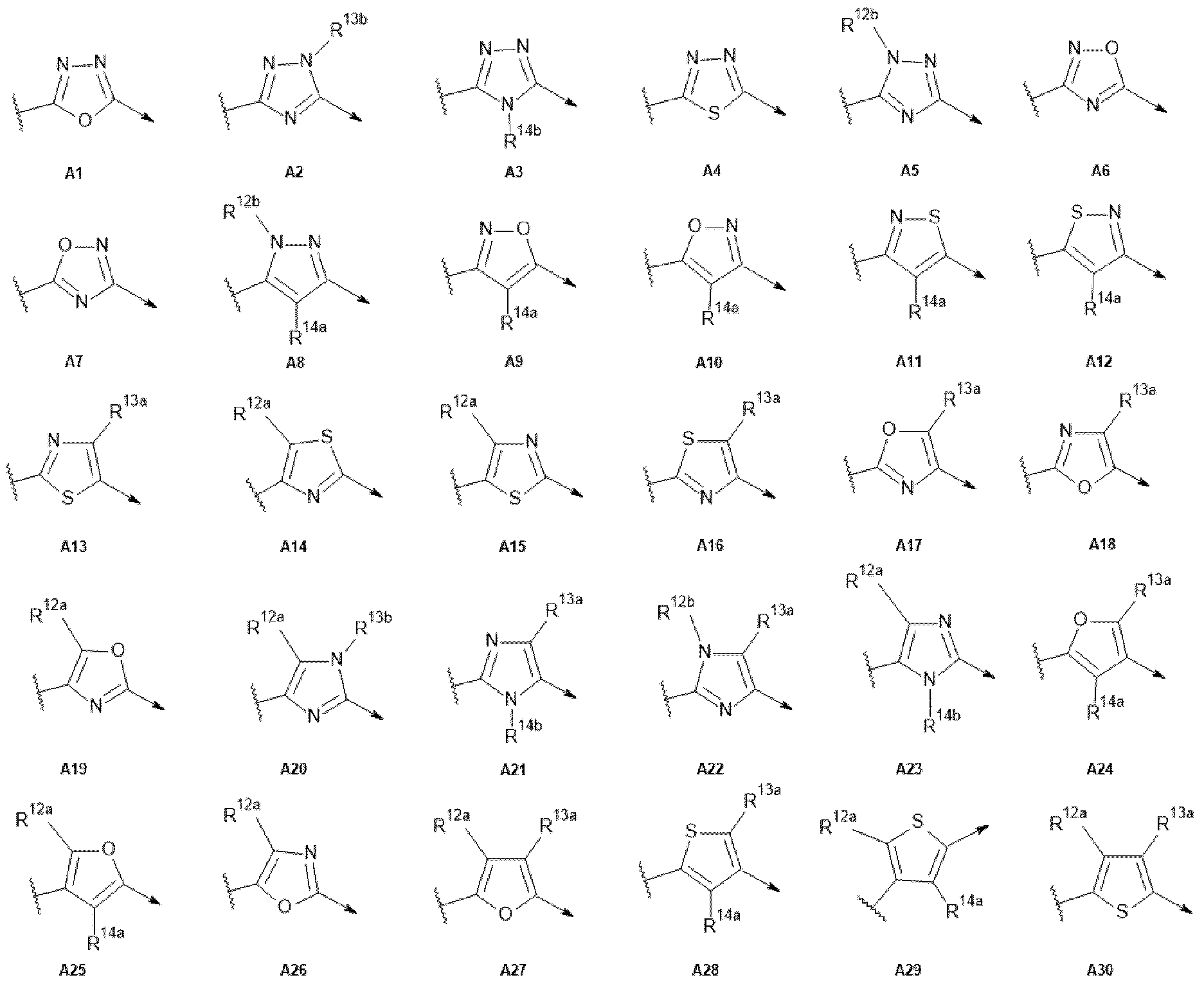
- R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , B 1 and B 2 and Z 1 are as defined for the compounds of formula (I) according to the present invention, and A is selected from A1 to A36:
- R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , B 1 , B 2 and Z 1 are as defined for the compounds of formula (I) according to the present invention, and R 12a , R 13a , R 14a , R 12b , R 13b and R 14b are independently selected from hydrogen, C1-C4 alkyl, C2-C4 alkenyl or C2-C4 alkynyl.
- R 12a , R 13a and R 14a are hydrogen.
- R 12a , R 13a and R 14a are hydrogen.
- R 14a is selected from hydrogen or C1-C4 alkyl.
- R 14a is hydrogen.
- R 14a is hydrogen.
- R 12a , R 13a , R 14a , R 12b , R 13b and R 14b are independently selected from hydrogen or methyl.
- R 12a , R 13a , R 14a , R 12b , R 13b and R 14b are hydrogen.
- R 12a , R 13a , R 14a , R 12b , R 13b and R 14b are methyl.
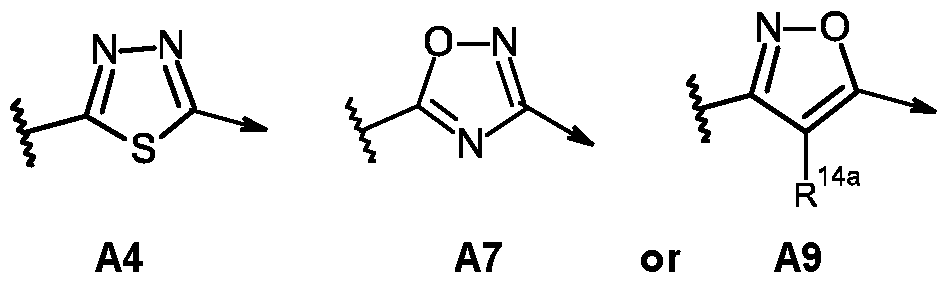
- the compound of formula (l-A) is selected from A4, A7 or A9, and R 14a is hydrogen.
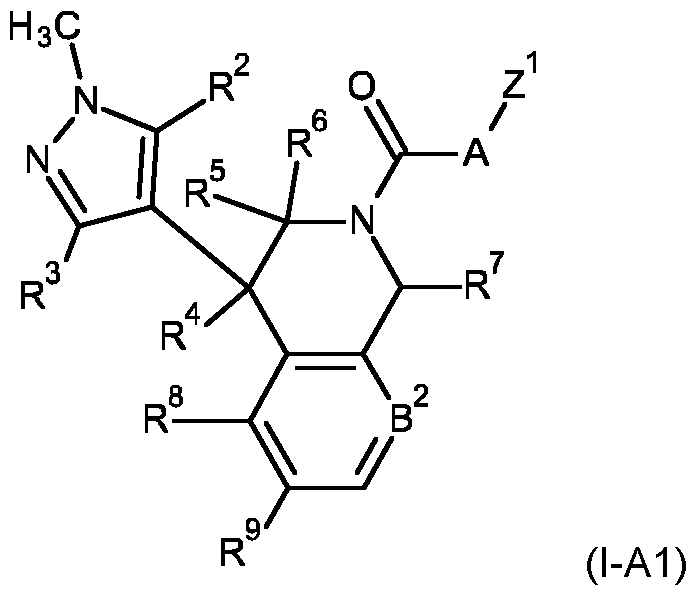
- the compound of formula (l-A) may be a compound of formula (I-A1) wherein R 1 is methyl, B 1 is CH, and A is defined as for compound (l-A), wherein R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , B 2 and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- a compound of formula (I-A1) wherein B 2 is N, and R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- a compound of formula (I-A1) wherein B 2 is CR 11 , and R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 11 and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- the compound of formula (l-A) may be a compound of formula (I-A2) wherein B 1 is CH; R 1 is methyl, R 3 , R 5 and R 6 are hydrogen; and A is defined as for compound (l-A); and wherein R 2 , R 4 , R 7 , R 8 , R 9 , B 2 and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl or chlorine
- R 4 is hydrogen or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen or methoxy
- B 2 is CR 11 , wherein R 11 is hydrogen
- a and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl, or chlorine
- R 4 is hydrogen or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen or methoxy
- B 2 is CR 11 , wherein R 11 is hydrogen
- A is A4, A7 or A9;
- Z 1 is as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl, or chlorine
- R 4 is hydrogen or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen or methoxy
- B 2 is CR 11 , wherein R 11 is hydrogen
- A is A4, A7 or A9;
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, 3,5-difluoro-2-pyridyl, 4-fluoropyridazin-3-yl, 4,5-difluoropyridazin-3-yl, or 5-fluoropyridazin-4-yl.
- the compound of formula (l-A) may be a compound of formula (I-A3) wherein R 1 is methyl; B 1 and B 2 are CH; and A is defined as for compound (l-A), and wherein R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- the compound of formula (l-A) may be a compound of formula (I-A4) wherein B 1 and B 2 are CH; R 1 is methyl; R 3 , R 4 , R 5 and R 6 are hydrogen and A is defined as for compound
- R 2 is hydrogen, methyl or chlorine
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen, or methoxy
- a and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl, or chlorine
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen, or methoxy
- A is A4, A7, or A9;
- Z 1 is as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl, or chlorine
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen, or methoxy
- A is A4, A7, or A9;
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, 3,5-difluoro-2-pyridyl, 4-fluoropyridazin-3-yl, 4,5-difluoropyridazin-3-yl, or 5-fluoropyridazin-4-yl.
- R 2 is hydrogen, methyl, or chlorine
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen, or methoxy
- A is A4, A7, or A9;
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, or 3,5-difluoro-2-pyridyl.
- the compound of formula (l-A) may be a compound of formula (I-A5) wherein R 1 is methyl; R 4 is methyl; R 3 , R 4 , R 5 and R 6 are hydrogen; B 1 and B 2 are CH; and A is defined as for compound (l-A), racemic syn-(l-A5) and wherein R 2 , R 7 , R 8 , R 9 and Z 1 are as defined for the compounds of formula (I) according to the present invention, and wherein when R 7 is not hydrogen the stereochemistry of the pyrazole-derivative-substituent and R 7 at the carbons with the “*” have a syn-relationship to each other.
- R 2 is hydrogen, methyl, or chlorine
- R 7 is methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen, or methoxy
- a and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl, or chlorine
- R 7 is methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen, or methoxy
- A is A4, A7, or A9;
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, 3,5-difluoro-2-pyridyl, 4-fluoropyridazin-3-yl, 4,5-difluoropyridazin-3-yl, or 5-fluoropyridazin-4-yl.
- R 2 is hydrogen, methyl, or chlorine
- R 7 is methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen, or methoxy
- A is A4, A7, or A9;
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, or 3,5-difluoro-2-pyridyl.
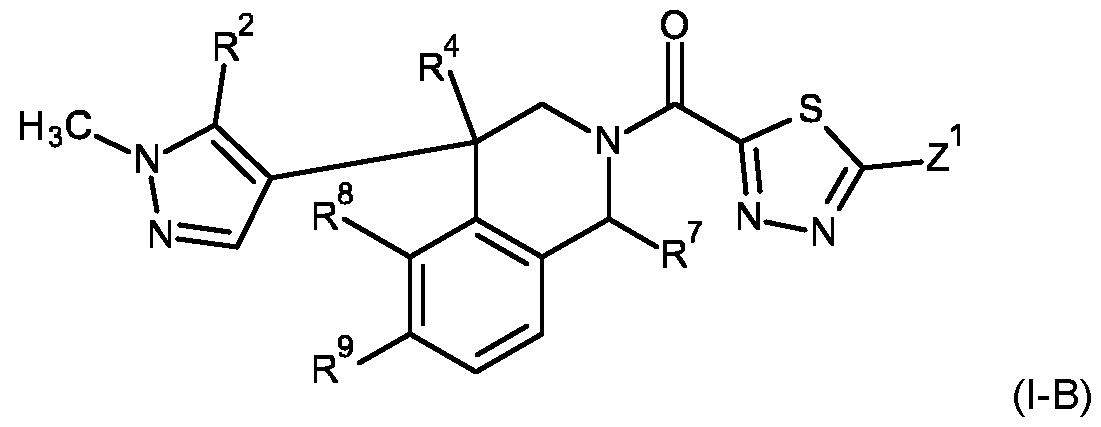
- the compound of formula (l-A) may be a compound of formula (l-B) wherein R 1 is methyl; R 3 , R 5 , R 6 are hydrogen; B 1 and B 2 are CH; and A is A4 and wherein R 2 , R 4 , R 7 , R 8 , R 9 and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl, or chlorine
- R 4 is hydrogen, or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen, or methoxy
- Z 1 is as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl, or chlorine
- R 4 is hydrogen, or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen, or methoxy
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, 3,5-difluoro-2-pyridyl, 4-fluoropyridazin-3-yl, 4,5-difluoropyridazin-3-yl, or 5-fluoropyridazin-4-yl.
- R 2 is hydrogen, methyl, or chlorine
- R 4 is hydrogen, or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano or bromine
- R 9 is hydrogen or methoxy
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl or 3,5-difluoro-2-pyridyl.
- R 2 is hydrogen, methyl or chlorine
- R 4 is hydrogen
- R 7 is methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen or methoxy
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, or 3,5-difluoro-2-pyridyl.
- the compound of formula (l-A) may be a compound of formula (l-C) wherein R 1 is methyl; R 3 , R 5 , R 6 are hydrogen; B 1 and B 2 are CH; and A is A7 and wherein R 2 , R 4 , R 7 , R 8 , R 9 and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl, or chlorine
- R 4 is hydrogen or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen or methoxy
- Z 1 is as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl, or chlorine
- R 4 is hydrogen or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen or methoxy
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, 3,5-difluoro-2-pyridyl, 4-fluoropyridazin-3-yl, 4,5-difluoropyridazin-3-yl, or 5-fluoropyridazin-4-yl.
- R 2 is hydrogen, methyl, or chlorine
- R 4 is hydrogen or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen or methoxy
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, or 3,5-difluoro-2-pyridyl.
- the compound of formula (l-A) may be a compound of formula (l-D) wherein R 1 is methyl; R 3 , R 5 , R 6 are hydrogen; B 1 and B 2 are CH; and A is A9 and wherein R 2 , R 4 , R 7 , R 8 , R 9 and Z 1 are as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl, or chlorine
- R 4 is hydrogen or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen or methoxy
- Z 1 is as defined for the compounds of formula (I) according to the present invention.
- R 2 is hydrogen, methyl, or chlorine
- R 4 is hydrogen or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano, or bromine
- R 9 is hydrogen or methoxy
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, 3,5-difluoro-2-pyridyl, 4-fluoropyridazin-3-yl, 4,5-difluoropyridazin-3-yl, or 5-fluoropyridazin-4-yl.
- R 2 is hydrogen, methyl, or chlorine
- R 4 is hydrogen or methyl
- R 7 is hydrogen, methyl, cyclopropyl, or 1 -cyanocyclopropyl
- R 8 is hydrogen, cyano or bromine
- R 9 is hydrogen or methoxy
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4-difluoro-2-pyridyl, or 3,5-difluoro-2-pyridyl.
- the intermediate compounds of formula (lib) possess the same definitions for A 1 , A 2 , A 3 and Z 1 as for the compounds of formula (I) according to the invention and their corresponding preferences.
- R° is C1-C4 alkyl
- a 1 , A 2 , A 3 and Z 1 are as defined forthe compounds of formula (I) according to the present invention.
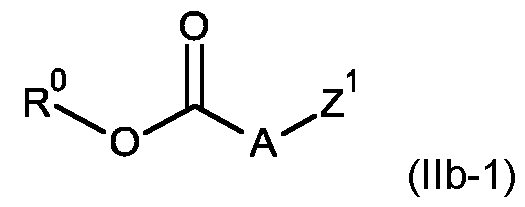
- the compound of formula (lib) may be a compound of formula (llb-1): wherein
- Z 1 is as defined for the compounds of formula (I) according to the present invention; R° is as defined for compounds of formula (lib); and A is selected from A1 to A36 as defined above for compounds of formula (I- A).
- R 14a is selected from hydrogen or C1-C4 alkyl, wherein Z 1 is as defined for the compounds of formula (I) according to the present invention, and R° is as defined for compounds of formula (lib).
- R 14a is hydrogen.
- R 14a is selected from hydrogen or C1-C4 alkyl, wherein Z 1 is as defined for the compounds of formula (I) according to the present invention, and R° is as defined for compounds of formula (lib).
- R 14a is hydrogen.
- A is A4;
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4- difluoro-2-pyridyl, or 3,5-difluoro-2-pyridyl; and R° is as defined for compounds of formula (lib).
- A is A7;
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4- difluoro-2-pyridyl, or 3,5-difluoro-2-pyridyl; and R° is as defined for compounds of formula (lib).
- A is A9;
- Z 1 is 3-fluoro-2-pyridyl, 5-fluoro-2-pyridyl, 3,4- difluoro-2-pyridyl or 3,5-difluoro-2-pyridyl;
- R 14a is hydrogen, and R° is as defined for compounds of formula (Hb).
- the presence of one or more possible asymmetric carbon atoms in any of the compounds selected from compounds of formula (I), (l-A), (I-A1), (I-A2), (I-A3), (I-A4), (I-A5), (l-B), (l-C), or (l-D), or compounds selected from compounds listed in Tables A-1 to A-26, or compounds listed in Table P (below), according to the invention means that the compounds may occur in chiral isomeric forms, i.e., enantiomeric or diastereomeric forms.
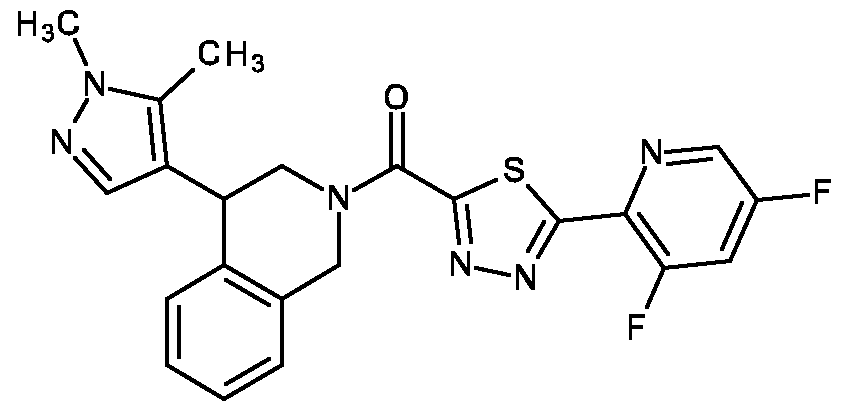
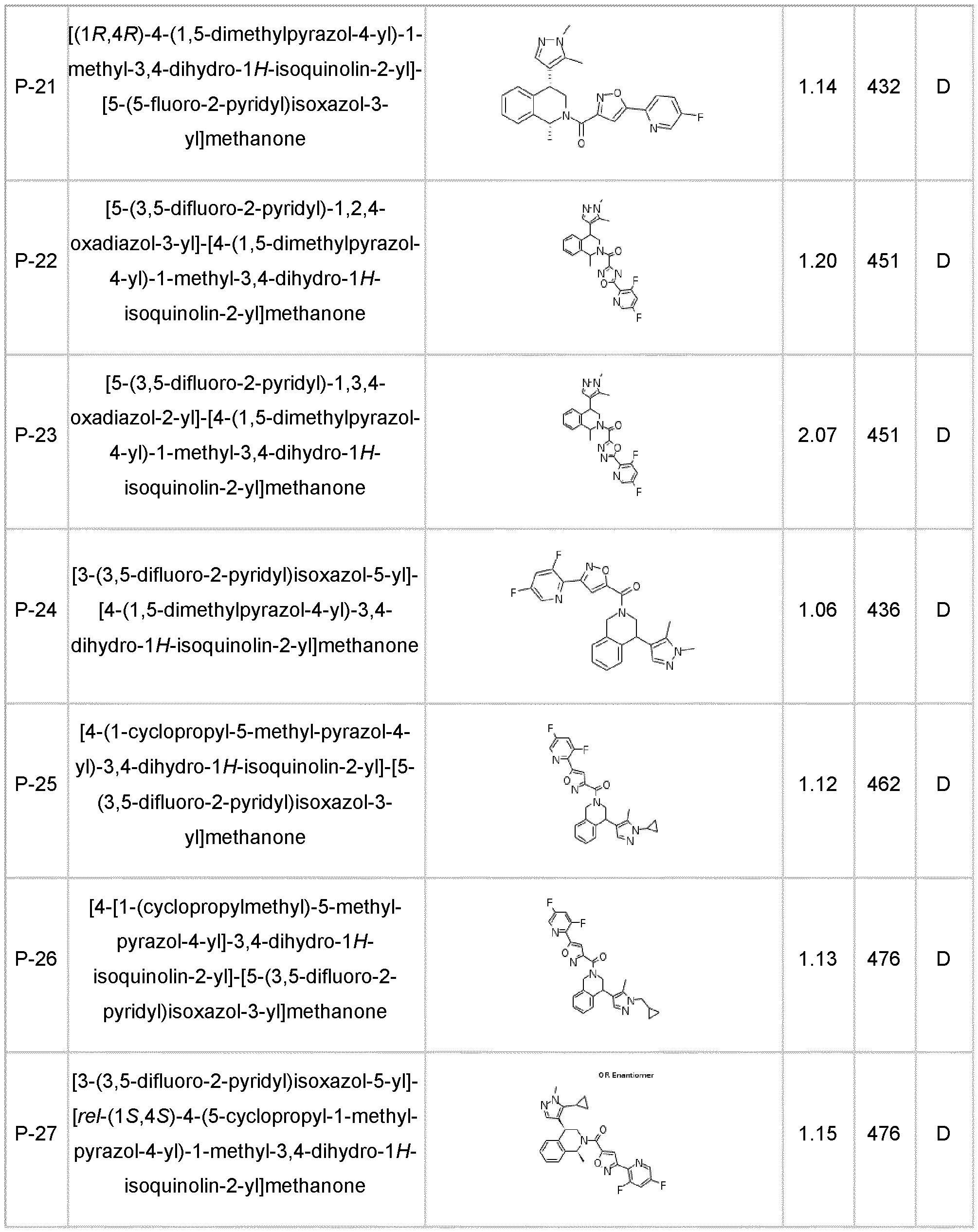
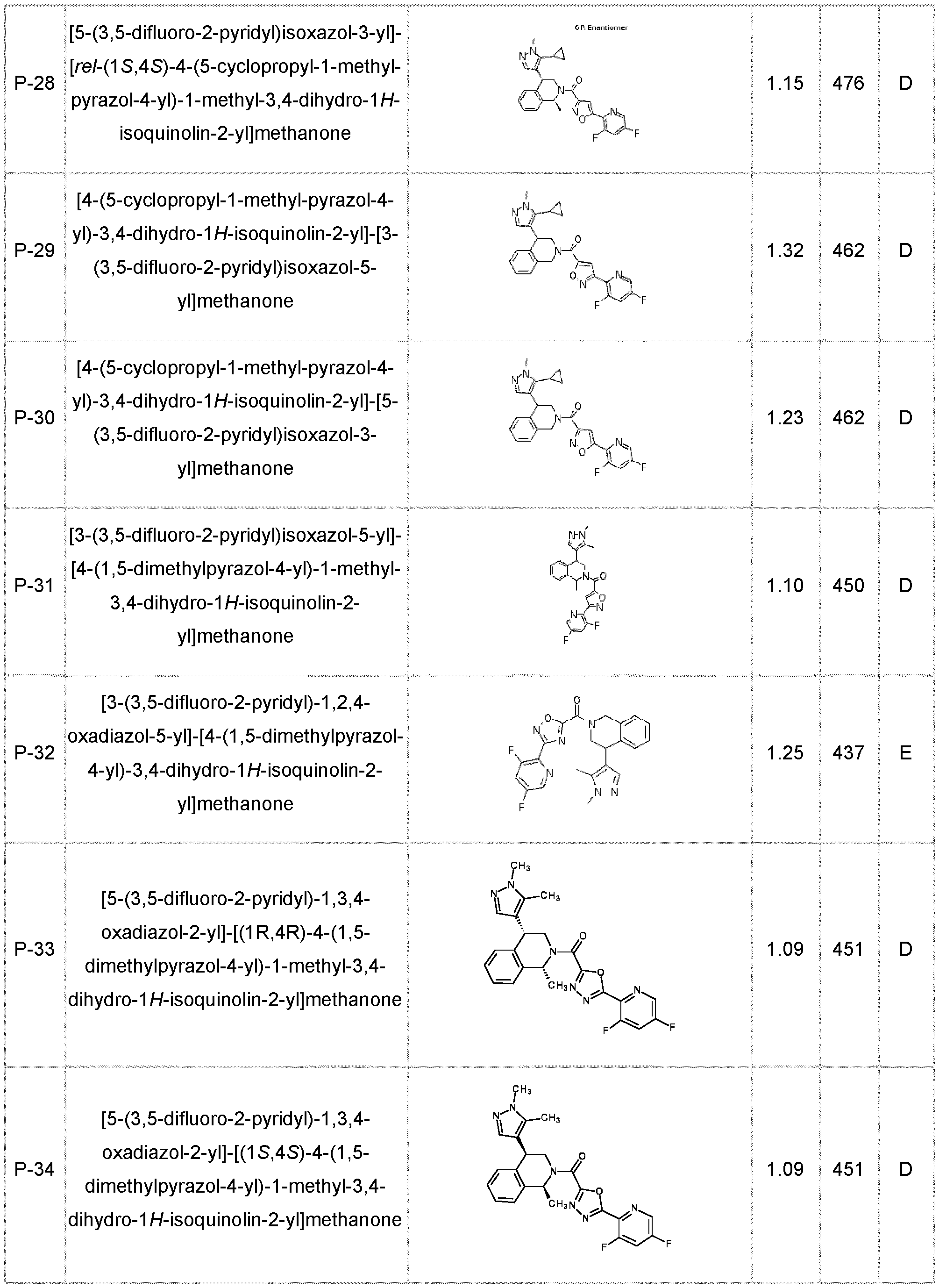
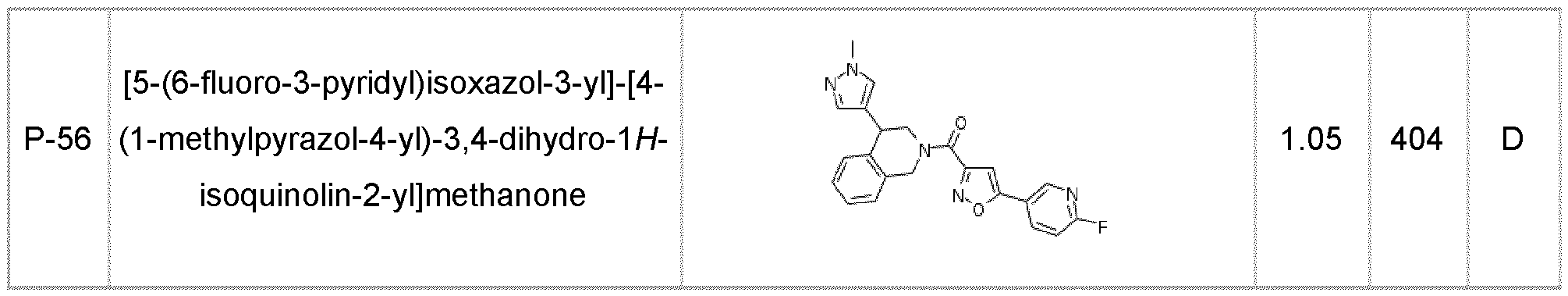
- the compound of formula (I) according to the invention is selected from compounds listed in any one of Tables A-1 to A-26.
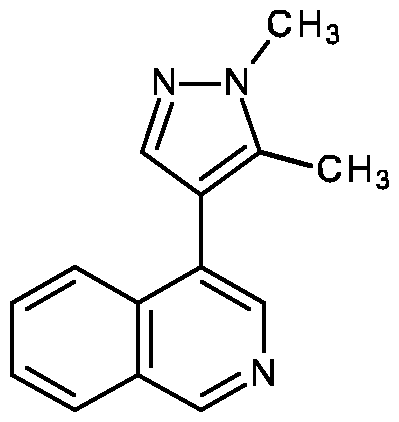
- the compound of formula (I) according to the invention is selected from [4-(1-methylpyrazol-4-yl)-3,4-dihydro-1 H-isoquinolin-2-yl]-[5-(4-pyridyl)isoxazol-3-yl]methanone (P-1), [4-(1-methylpyrazol-4-yl)-3,4-dihydro-1 H-isoquinolin-2-yl]-[5-(3-pyridyl)isoxazol-3-yl]methanone (P-2), [4-(1-methylpyrazol-4-yl)-3,4-dihydro-1 H-isoquinolin-2-yl]-[5-(2-pyridyl)isoxazol-3-yl]methanone (P-3), [5-(6-methoxy-3-pyridyl)isoxazol-3-yl]-[4-(1-methylpyrazol-4-yl]
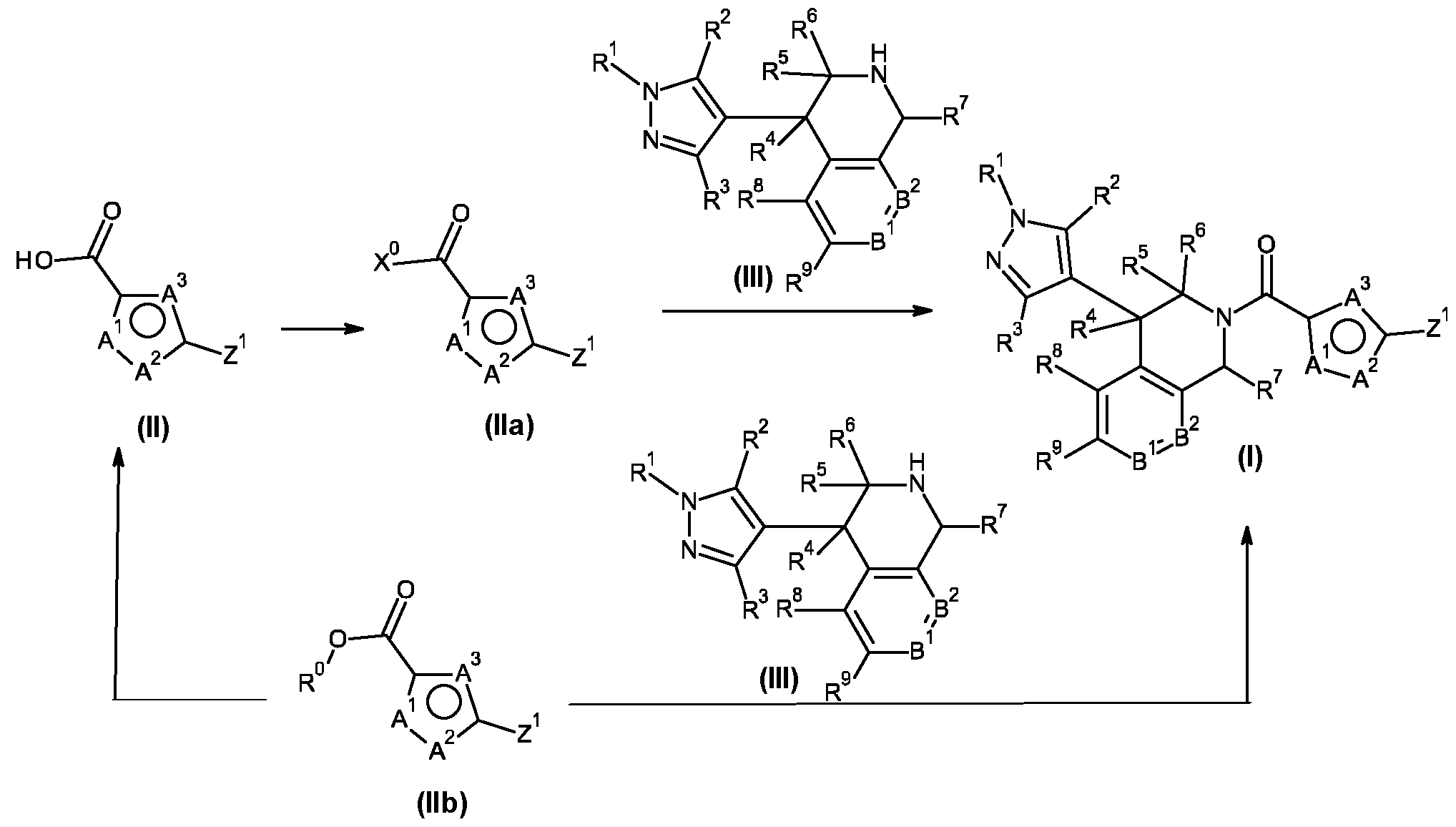
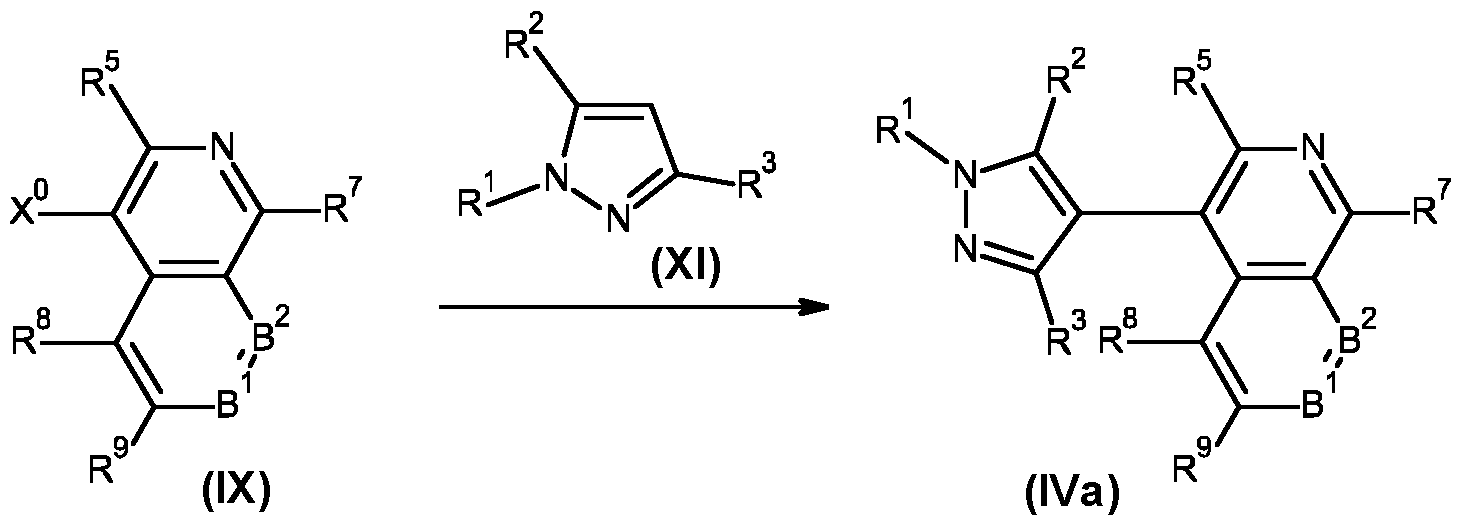
- compounds of formula (I) may be prepared by a person skilled in the art following known methods. More specifically, compounds of formula (I) may be prepared from compounds of formula (III) or a salt thereof, wherein R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , B 1 and B 2 are as defined for the compounds of formula (I) by reaction with a compound of formula (II), wherein A 1 , A 2 , A 3 and Z 1 are as defined for the compounds of formula (I). This reaction is shown in Scheme 1.
- compounds of formula (Ila), where X° is halogen are formed by treatment of compounds of formula (II) with, for example, oxalyl chloride or thionyl chloride in the presence of catalytic quantities of N,N-dimethylformamide (DMF) in inert solvents such as methylene dichloride or tetrahydrofuran (THF) at temperatures between 20°C to 100°C, preferably 25°C.
- DMF N,N-dimethylformamide
- inert solvents such as methylene dichloride or tetrahydrofuran (THF)
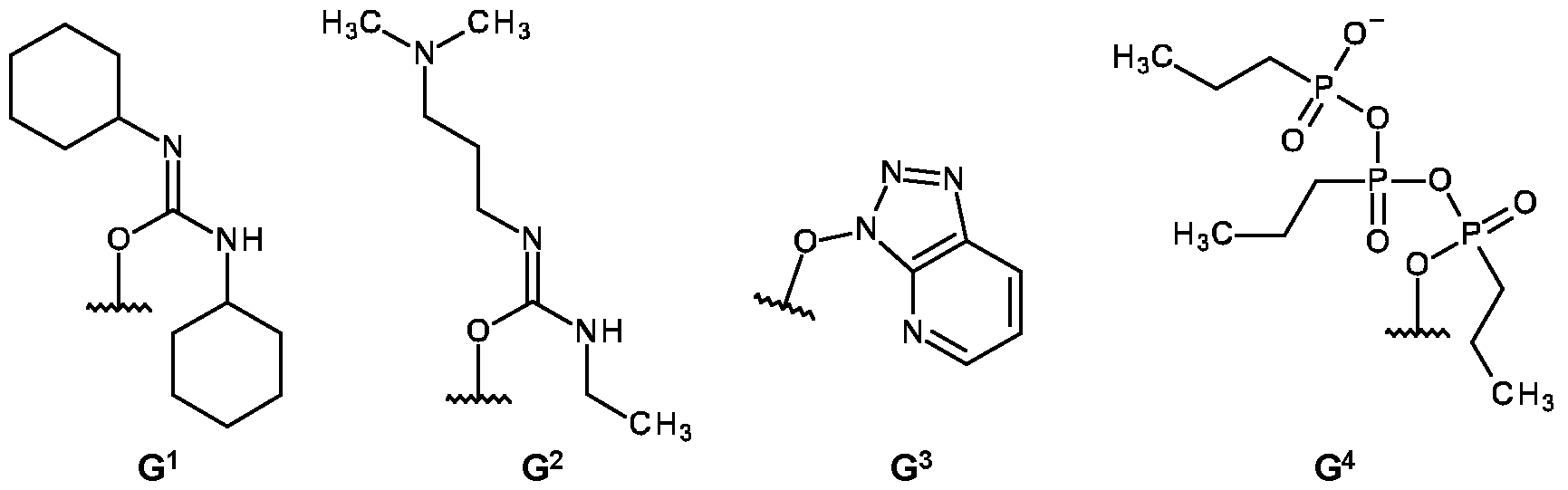
- compounds of formula (I) may be prepared by treatment of compounds of formula (II) with dicyclohexyl carbodiimide (DCC), 1 -ethyl-3-(3- dimethylaminopropyl)carbodiimide (EDC) or 1-[bis(dimethylamino)methylene]-1 H-1 ,2,3-triazolo[4,5- b]pyridinium 3-oxide hexafluorophosphate (HATU) to give the activated compound of formula (Ila), wherein X° is G 1 , G 2 or G 3 as set forth in Scheme 1 a, in an inert solvent, e.g.
- DCC dicyclohexyl carbodiimide
- EDC 1 -ethyl-3-(3- dimethylaminopropyl)carbodiimide
- HATU 1-[bis(dimethylamino)methylene]-1 H-1 ,2,3-triazolo[4,5- b]pyridinium
- Compounds of formula (II) can be prepared from compounds of formula (lib), wherein A 1 , A 2 , A 3 are N and Z 1 are as described in formula (I), and R° is Ci-C4alkyl, by ester hydrolysis.
- a variety of conditions can be used, as for example aqueous sodium hydroxide or lithium hydroxide, and an organic water miscible solvent like THF, dimethoxyethane, methanol, or ethanol.
- ester hydrolyses are well known to those skilled in the art.
- compounds of formula (Illa), wherein R 4 and R 6 are hydrogen, R 5 is hydrogen or methyl and R 1 , R 2 , R 3 , R 7 , R 8 , R 9 , R 10 , R 11 , B 1 and B 2 are as defined for the compounds of formula (I), may be prepared from compounds of formula (IVa), wherein R 4 and R 6 are hydrogen, R 5 is hydrogen or methyl and R 1 , R 2 , R 3 , R 7 , R 8 , R 9 , R 10 , R 11 , B 1 and B 2 are as defined for the compounds of formula (I), by treatment with a reducing agent such as NaBHsCN and an acid, for example hydrochloric acid, or acetic acid in a protic solvent such as methanol or ethanol and the like.
- a reducing agent such as NaBHsCN
- an acid for example hydrochloric acid, or acetic acid in a protic solvent such as methanol or ethanol and the like.
- compounds of formula (Illa) may be prepared from compounds of formula (IVa) by reduction with hydrogen in the presence of a suitable metal catalyst, such as Pd, Ir, Rh with a suitable ligand, e.g. diphosphine [1 ,2-bis(diphenylphosphino)ethane (dppe), 1 ,3-bis(diphenylphosphino)propane (dppp) or 1 ,4- bis(diphenylphosphino)butane (dppb)]. Similar reactions have been reported for example in React. Kinet. Cat. Lett. 2007, 92, 99-104. This reaction is shown in Scheme 2.
- compounds of formula (lllb), wherein R 4 , R 6 and R 7 are hydrogen, R 5 is hydrogen or methyl and R 1 , R 2 , R 3 , R 8 , R 9 , R 10 , R 11 , B 1 and B 2 are as defined for the compounds of formula (I), can be converted to compounds of formula (V), wherein R 4 , R 6 and R 7 are hydrogen, R 5 is hydrogen or methyl and R 1 , R 2 , R 3 , R 8 , R 9 , R 10 , R 11 , B 1 and B 2 are as defined for the compounds of formula (I), by treatment of compounds of formula (lllb) with a compound of formula (VI), wherein X° is a leaving group, such as halogen, and R° is Ci-Cealkyl, by methods known to a person skilled in the art and by those described in Scheme 1 .
- compounds of formula (V) may be prepared by treatment with an anhydride of formula (R°CO)2O, wherein R° is Ci-Cealkyl, in an inert solvent such as methylene chloride, THF or 2-methyl-THF, optionally in the presence of a base, such as triethylamine or dimethylaminopyridine, at temperatures between 0°C and 60°C.
- R°CO anhydride of formula (R°CO)2O, wherein R° is Ci-Cealkyl
- an inert solvent such as methylene chloride, THF or 2-methyl-THF
- a base such as triethylamine or dimethylaminopyridine
- a base for example an alkyl metal base, such as tert-butyl lithium, and an additive such as /V,/V,/V,/V'-tetramethylethylendiamine (TMEDA) at low temperature, for example -78°C to room temperature, in an inert polar solvent such as THF or 2-methyl-THF.
- a base for example an alkyl metal base, such as tert-butyl lithium
- TEDA /V,/V,/V,/V'-tetramethylethylendiamine
- Compounds of formula (Va) may be converted to compounds of formula (Illa), wherein R 4 and R 6 are hydrogen, R 5 is hydrogen or methyl and R 1 , R 2 , R 3 , R 7 , R 8 , R 9 , R 10 , R 11 , B 1 and B 2 are as defined for the compounds of formula (I), by methods known to a person skilled in the art.
- compounds of formula (Va), wherein R° is tert-butyl may be treated with an organic or inorganic acid such as trifluoroacetic acid or HCI to give compounds of formula (Illa). This reaction is shown in Scheme 4.
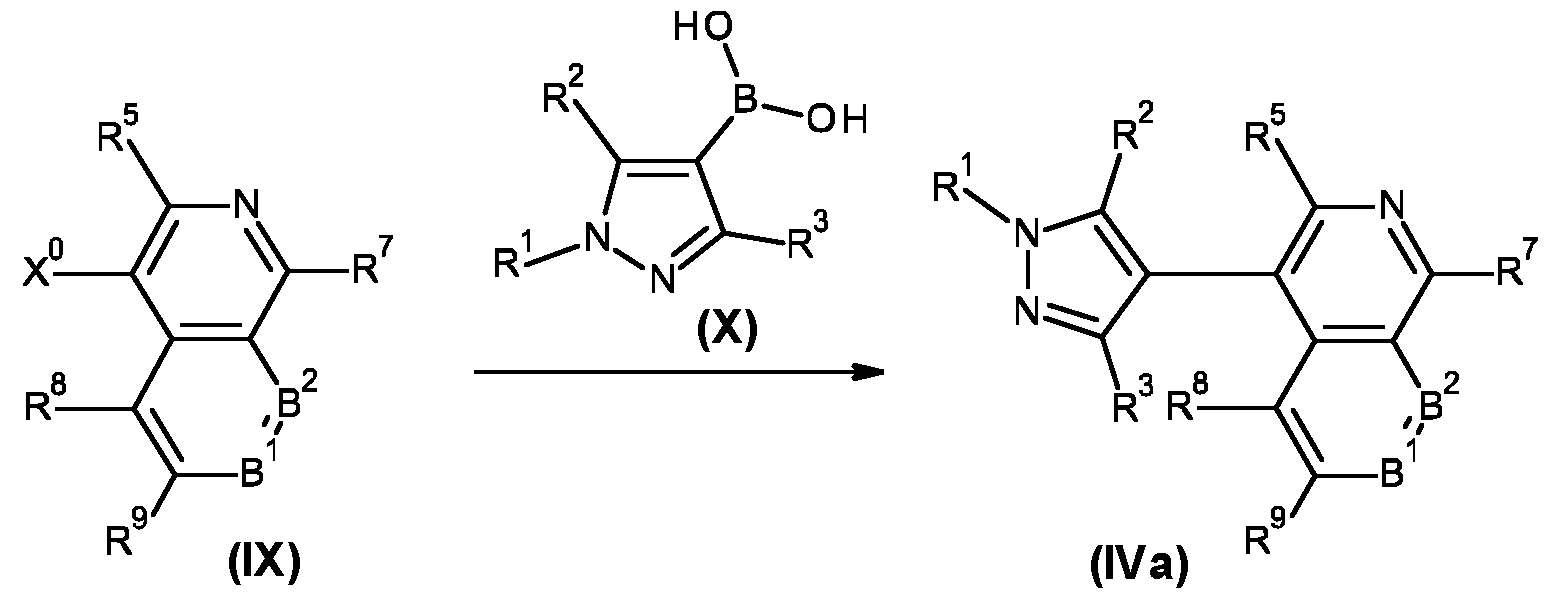
- Suzuki-Miyaura cross-coupling reactions between compounds of formula (VIII) and compound of formula (VII) are well known to a person skilled in the art and are usually carried out in the presence of a palladium catalyst, such as tetrakis(triphenylphosphine)-palladium(0) or [1 ,1 '-bis(diphenylphosphino)ferrocene]palladium(ll) dichloride dichloromethane complex, and a base, such as sodium or potassium carbonate, in a solvent, such as N,N-dimethylformamide, dioxane or dioxane-water mixtures, at temperatures between room temperature and 160°C, optionally under microwave heating conditions, and preferably under inert atmosphere.
- a palladium catalyst such as tetrakis(triphenylphosphine)-palladium(0) or [1 ,1 '-bis(diphenylphosphino)ferrocene]palladium(ll) dich
- a further cross-coupling chemistry namely C-H activation, can also be used to prepare compounds of formula (IVa), wherein R 4 and R 6 are hydrogen, R 5 is hydrogen or methyl and R 1 , R 2 , R 3 , R 7 , R 8 , R 9 , B 1 and B 2 are as defined for the compounds of formula (I) (Scheme 7).
- compounds of formula (III) may be prepared from compounds of formula (XVI) (Scheme 8).
- compounds of formula (III) may be prepared by a person skilled in the art by a carbamate deprotection reaction of compounds of formula (XVI), wherein R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , B 1 and B 2 are as defined for the compounds of formula (I) and R 01 may be a member of a common carbamate protecting group substituent, for example methyl, te/Y-butyl, allyl, 2,2,2-trichloroethyl or benzyl.
- R 01 is methyl
- a suitable solvent such as dichloromethane and a suitable reagent such as iodotrimethylsilane may be employed to afford the product upon heating at temperatures between room temperature and 200°C, preferably between 20°C and the boiling point of the reaction mixture as described, for example, in J. Am. Chem. Soc. 1992, 114, 5959.
- the compounds of formula (III) thus obtained are converted to compounds of formula (I) (Scheme 1).
- Grignard reagents R 5 MgBr or R 6 MgBr, wherein R 5 and R 6 are as defined for the compounds of formula (I) may be added as nucleophiles to compounds of formula (XII), sequentially or simultaneously, to allow more highly substituted amines of formula (XIII) to be prepared.
- Such Grignard additions to nitriles are carried out in an inert solvent such as diethyl ether, tert-butylmethyl ether, and cyclopentyl methyl ether in the presence of a Lewis acid such as Ti(O-'Pr)4 (see Synlett 2007, (4), 652-654). This reaction is shown in Scheme 11.
- compounds of formula (XII), wherein R 1 , R 2 , R 3 , R 8 , R 9 , B 1 and B 2 are as defined for the compounds of formula (I) and R 4 is different from hydrogen may be prepared by a person skilled in the art by deprotonation of compound of formula (Xlla) wherein R 1 , R 2 , R 3 , R 8 , R 9 , B 1 and B 2 are as defined for the compounds of formula (I), using a strong base such as n-butyl lithium or sodium hydride at cryogenic temperatures in an inert solvent such as tetrahydrofuran, followed by addition of a suitable alkylating agent R 4 - X, wherein R 4 is Ci-C4alkyl and X is halogen, for example iodomethane.
- a strong base such as n-butyl lithium or sodium hydride
- an inert solvent such as tetrahydrofuran
- Compounds of formula (Xlla), wherein R 1 , R 2 , R 3 , R 8 , R 9 , B 1 and B 2 are as defined for the compounds of formula (I), may be prepared from alcohols of formula (XVII) by treatment with cyanotrimethylsilane (TMSCN) in the presence of a base such as lithium carbonate in a nonpolar solvent such as dichloromethane at temperatures between 0°C and the boiling point of the reaction mixture.
- TMSCN cyanotrimethylsilane
- a base such as lithium carbonate
- a nonpolar solvent such as dichloromethane
- R 1 is Ci-C4alkyl
- R 2 is hydrogen, halogen, or Ci-C4alkyl
- R 3 is hydrogen
- R 4 is hydrogen or Ci-C4alkyl
- R 5 , R 6 , R 7 are hydrogen or Ci-C4alkyl
- R 8 , R 9 , R 10 , and R 11 are as previously defined, with strong acids, for example sulphuric, hydrochloric, hydrobromic, trifluoroacetic, trifllic, or methane sulphonic acids and the like, or Lewis acids, such as aluminium trichloride, or bismuth (I I l)triflate , in an inert solvent such as chlorobenzene, nitrobenzene at temperature between 0°C to 180°C to yield compounds of formula (lllc).
- strong acids for example sulphuric, hydrochloric, hydrobromic, trifluoroacetic, trifllic, or methane sulphonic acids and the like
- a benzyl amine of formula (XXI) is used to alkylate a compound of formula (XXII), in the presence of a base, such as EtsN in an inert solvent, such as DMF.
- a base such as EtsN
- an inert solvent such as DMF.
- the compound (XXIII) so obtained may be isolated, or treated directly with BOC-anhydride in situ, to give a compound of formula (XXIV).
- Compound of formula (XXIV) can be reduced with a hydride source (e.g., NaBH4 in MeOH/THF) to give the target molecule (XVI 11 b) , which can then be cyclized with, for example with camphor sulphonic acid in EtOAc, to give compounds of formula (XlXb).
- a hydride source e.g., NaBH4 in MeOH/THF
- compounds of formula (XXIV) can be reacted with a Grignard reagent R 4 MgBr in an inert ethereal solvent (e.g., THF) to give compounds of formula (XVI Ila), which can the cyclized with camphorsulphonic acid in, e.g. EtOAc, to give compounds of formula (XIXa).
- R 1a is Ci-C4alkyl
- R 2a is hydrogen, halogen, or Ci-C4alkyl
- R 4a is hydrogen or Ci- C4alkyl
- R 5a , R 6a , and R 7a are hydrogen or C1-C4 alkyl
- R 8 , R 9 , R 10 , and R 11 are as defined for the compounds of formula (I).
- a further aspect of this Friedel-Crafts chemistry should be noted. If the chemistry is carried out starting with a chiral amine (XXIa), e.g., R 7 is Ci-C4alkyl, the stereochemistry is retained in the final compounds of formula (I). This is illustrated below in Scheme 16, for when R 7 is methyl:
- compounds of formula (lie) wherein Z 1 is as defined for the compounds of formula (I), and X 05 is C1-C4 alkyl may be prepared by hydrolysis of compounds of formula (lib) by treatment with, for example, an alkaline earth metal hydroxide in water, or with a water miscible organic solvent, such as THF, methanol, ethanol and the like.
- a water miscible organic solvent such as THF, methanol, ethanol and the like.
- Compounds of formula (lib) can be obtained by the treatment of compounds of formula (XXVII) wherein Z 1 is as defined for the compounds of formula (I) and X 05 is C1-C4 alkyl with hydroxylamine hydrochloride in a polar solvent, for example ethanol and optionally in the presence of a base, e.g., triethyl amine, K2CO3 and the like.
- a polar solvent for example ethanol
- a base e.g., triethyl amine, K2CO3 and the like.
- Compounds of formula (XXVIII) wherein Z 1 is as defined for the compounds of formula (I) may be also prepared by “one-pot” synthesis via Pd-catalyzed cyanation and amidoximation of compound of formula (XXX) wherein Z 1 is as defined for the compounds of formula (I), using potassium ferrocyanide trihydrate and hydroxylamine hydrochloride as described in for example Org. Biomol. Chem. 2015, 13(9), 2541-2545.
- Scheme 19 As shown in Scheme 19, compounds of formula (llg) and (Hi) are obtained by ester hydrolysis of (Ilf) and (llg), respectively. In the latter compounds, X 05 and Z 1 are as previously described.
- Compounds of formula (Ilf) can be obtained from compounds of formula (Ilf) by dehydration of compounds of formula (XXXI).
- Compounds of formula (XXXI) are obtained by acylation of hydrazides of formula (XXXII) with compounds of formula (XXVIa).
- Such sequences of reactions to produce oxadiazoles is well known to those skilled in the art. Similar reactions are described in Bioorg. Med. Chem. Let. 2005, 15, 1423-1428 and W02006/044617.
- Compounds of formula (XXXI) can also be prepared by reaction of activated carboxylic acids of formula (XXXIIIa), wherein Z 1 is as defined for the compounds of formula (I), and X° are as described in Scheme 1 /Scheme 1 a respectively, with compounds of formula (XXXIV).
- Compounds of formula (XXXIIIa) can be prepared from the corresponding acids of formula (XXXIII) as described in scheme 1 .
- Such reactions are described for example in, for example, J. Prakt. Chem. 1985, 327, 109-116.
- compounds of formula (Ilk) are readily obtained by hydrolysis of esters of formula (llj) by methods known to those skilled in the art and described vide supra.
- Compounds of formula (llj) can be obtained by reaction of compounds of formula (XXXV) with compounds of formula (XXXVI) in the presence of an oxidizing agent, for example (diacetoxyiodo)benzene or N-chlorosuccinimide, in an inert solvent such as methanol or DMF respectively.
- an oxidizing agent for example (diacetoxyiodo)benzene or N-chlorosuccinimide
- an inert solvent such as methanol or DMF respectively.
- Such reaction sequences have been described, for example, in J. Het. Chem. 2013, 50(4), 774-780 and J. Chin. Chem. Soc. 2007, 54(3), 643-652.
- Compounds of formula (XXXV) are readily prepared from compounds of formula (XXXVII
- Salts of compounds of formula (I) may be prepared in a manner known perse.
- acid addition salts of compounds of formula (I) are obtained by treatment with a suitable acid or a suitable ion exchanger reagent and salts with bases are obtained by treatment with a suitable base or with a suitable ion exchanger reagent.
- Salts of compounds of formula (I) can be converted in the customary manner into the free compounds (I), acid addition salts, for example, by treatment with a suitable basic compound or with a suitable ion exchanger reagent and salts with bases, for example, by treatment with a suitable acid or with a suitable ion exchanger reagent.
- Salts of compounds of formula (I) can be converted in a manner known per se into other salts of compounds of formula (I), acid addition salts, for example, into other acid addition salts, for example by treatment of a salt of inorganic acid such as hydrochloride with a suitable metal salt such as a sodium, barium or silver salt, of an acid, for example with silver acetate, in a suitable solvent in which an inorganic salt which forms, for example silver chloride, is insoluble and thus precipitates from the reaction mixture.
- a salt of inorganic acid such as hydrochloride
- a suitable metal salt such as a sodium, barium or silver salt
- an acid for example with silver acetate
- an inorganic salt which forms, for example silver chloride is insoluble and thus precipitates from the reaction mixture.
- the compounds of formula (I) which have salt-forming properties, can be obtained in free form or in the form of salts.
- the compounds of formula (I) and, where appropriate, the tautomer’s thereof, in each case in free form or in salt form, can be present in the form of one of the isomers which are possible or as a mixture of these, for example in the form of pure isomers, such as antipodes and/or diastereomers, or as isomer mixtures, such as enantiomer mixtures, for example racemates, or diastereomer mixtures, depending on the number, absolute and relative configuration of asymmetric carbon atoms which occur in the molecule and/or depending on the configuration of non-aromatic double bonds which occur in the molecule, the invention relates to the pure isomers and also to all isomer mixtures which are possible and is to be understood in each case in this sense hereinabove and herein below, even when stereochemical details are not mentioned specifically in each case.
- Diastereomeric mixtures or racemic mixtures of compounds of formula (I), in free form or in salt form, which can be obtained depending on which starting materials and procedures have been chosen can be separated in a known manner into the pure diastereomers or racemates on the basis of the physicochemical differences of the components, for example by fractional crystallization, distillation and/or chromatography.
- Enantiomeric mixtures such as racemates, which can be obtained in a similar manner can be resolved into the optical antipodes by known methods, for example by recrystallization from an optically active solvent, by chromatography on chiral adsorbents, for example high-performance liquid chromatography (HPLC) on acetyl cellulose, with the aid of suitable microorganisms, by cleavage with specific, immobilized enzymes, via the formation of inclusion compounds, for example using chiral crown ethers, where only one enantiomer is complexed, or by conversion into diastereomeric salts, for example by reacting a basic end-product racemate with an optically active acid, such as a carboxylic acid, for example camphor, tartaric or malic acid, or sulfonic acid, for example camphorsulfonic acid, and separating the diastereomer mixture which can be obtained in this manner, for example by fractional crystallization based on their differing solubilities, to give the di
- Pure diastereomers or enantiomers can be obtained according to the invention not only by separating suitable isomer mixtures, but also by generally known methods of diastereoselective or enantioselective synthesis, for example by carrying out the process according to the invention with starting materials of a suitable stereochemistry.
- compounds with more than one asymmetric carbon atoms may exist in diastereomeric forms which can be optionally separated using for example supercritical fluid chromatography (SFC) chromatography with chiral columns.
- SFC supercritical fluid chromatography
- Such diastereomers can show a different fungicidal activity profile, but all isomers and diastereomers form part of this invention.
- the compounds of formula (I) have three chiral carbon atoms, (three stereocenters, wherein the star (*) indicates the chiral carbon atom), such there are eight stereoisomers available. These eight stereoisomers consist of four sets of enantiomers.
- the compounds of formula (I), wherein R 1 is methyl, R 3 , R 4 , R 5 , R 6 are hydrogen and R 2 , R 7 , R 8 , R 9 , B 1 , B 2 , A (A 1 , A 2 , A 3 ) and Z 1 are as defined for the compounds of formula (I), and wherein R 7 is not hydrogen the pyrazole-moiety and R 7 have a syn-relationship to each other.
- the compounds of formula (I) and, where appropriate, the tautomers thereof, in each case in free form or in salt form, can, if appropriate, also be obtained in the form of hydrates and/or include other solvents, for example those which may have been used for the crystallization of compounds which are present in solid form.
- the compounds of formula (I) of the present invention have, for practical purposes, a very advantageous level of biological activity for protecting plants against diseases that are caused by fungi.
- the compounds of formula (I) according to the invention can be used in the agricultural sector and related fields of use, e.g., as active ingredients for controlling plant pests or on non-living materials for the control of spoilage microorganisms or organisms potentially harmful to man.
- the novel compounds are distinguished by excellent activity at low rates of application, by being well tolerated by plants and by being environmentally safe. They have very useful curative, preventive and systemic properties and can be used for protecting numerous cultivated plants.
- the compounds of formula (I) can be used to inhibit or destroy the pests that occur on plants or parts of plants (fruit, blossoms, leaves, stems, tubers, roots) of different crops of useful plants, while at the same time protecting also those parts of the plants that grow later, e.g., from phytopathogenic microorganisms.
- the present invention further relates to a method for controlling or preventing infestation of plants or plant propagation material and/or harvested food crops susceptible to microbial attack by treating plants or plant propagation material and/or harvested food crops wherein an effective amount a compound of formula (I) according to the invention is applied to the plants, to parts thereof or the locus thereof.
- fungicide as used herein means a compound that controls, modifies, or prevents the growth of fungi.
- fungicidally effective amount where used means the quantity of such a compound or combination of such compounds that is capable of producing an effect on the growth of fungi. Controlling or modifying effects include all deviation from natural development, such as killing, retardation and the like, and prevention includes barrier or other defensive formation in or on a plant to prevent fungal infection.
- compounds of formula (I) may also be possible to use compounds of formula (I) according to the invention as dressing agents for the treatment of plant propagation material, e.g., seed, such as fruits, tubers or grains, or plant cuttings, for the protection against fungal infections as well as against phytopathogenic fungi occurring in the soil.
- the propagation material can be treated with a composition comprising a compound of formula (I) before planting: seed, for example, can be dressed before being sown.
- the active compounds of formula (I) can also be applied to grains (coating), either by impregnating the seeds in a liquid formulation or by coating them with a solid formulation.
- the composition can also be applied to the planting site when the propagation material is being planted, for example, to the seed furrow during sowing.
- the invention relates also to such methods of treating plant propagation material and to the plant propagation material so treated.
- the compounds of formula (I) according to the invention can be used for controlling fungi in related areas, for example in the protection of technical materials, including wood and wood related technical products, in food storage, in hygiene management.
- the invention could be used to protect non-living materials from fungal attack, e.g., lumber, wall boards, and paint.
- the compounds of formula (I) according to the invention are for example, effective against fungi and fungal vectors of disease as well as phytopathogenic bacteria and viruses.
- These fungi and fungal vectors of disease as well as phytopathogenic bacteria and viruses are for example: Absidia corymbifera, Alternaria spp., Aphanomyces spp., Ascochyta spp., Aspergillus spp. including A. flavus, A. fumigatus, A. nidulans, A. niger, A. terrus, Aureobasidium spp. including A.
- Botryosphaeria spp. including B. dothidea, B. obtusa, Botrytis spp. including B. cinerea, Candida spp. including C. albicans, C. glabrata, C. krusei, C. lusitaniae, C. parapsilosis, C. tropicalis, Cephaloascus fragrans, Ceratocystis spp., Cercospora spp. including C.
- capsulatum Laetisaria fuciformis, Leptographium lindbergi, Leveillula taurica, Lophodermium seditiosum, Microdochium nivale, Microsporum spp., Monilinia spp., Mucor spp., Mycosphaerella spp. including M. graminicola, M. pomi, Oncobasidium theobromaeon, Ophiostoma piceae, Paracoccidioides spp., Penicillium spp. including P. digitatum, P. italicum, Petriellidium spp., Peronosclerospora spp. Including P. maydis, P.
- leucotricha Polymyxa graminis, Polymyxa betae, Pseudocercosporella herpotrichoides, Pseudomonas spp., Pseudoperonospora spp. including P. cubensis, P. humuli, Pseudopeziza tracheiphila, Puccinia spp. including P. hordei, P. recondita, P. striiformis, P. triticina, Pyrenopeziza spp., Pyrenophora spp., Pyricularia spp. including P. oryzae, Pythium spp. including P.
- the compounds of formula (I) according to the invention may be used for example on turf, ornamentals, such as flowers, shrubs, broad-leaved trees, or evergreens, for example conifers, as well as for tree injection, pest management and the like.
- target crops and/or useful plants to be protected typically comprise perennial and annual crops, such as berry plants for example blackberries, blueberries, cranberries, raspberries and strawberries; cereals for example barley, maize (corn), millet, oats, rice, rye, sorghum triticale and wheat; fibre plants for example cotton, flax, hemp, jute and sisal; field crops for example sugar and fodder beet, coffee, hops, mustard, oilseed rape (canola), poppy, sugar cane, sunflower, tea and tobacco; fruit trees for example apple, apricot, avocado, banana, cherry, citrus, nectarine, peach, pear and plum; grasses for example Bermuda grass, bluegrass, bentgrass, centipede grass, fescue, ryegrass, St.
- perennial and annual crops such as berry plants for example blackberries, blueberries, cranberries, raspberries and strawberries
- cereals for example barley, maize (corn), millet, oats
- Augustine grass and Zoysia grass herbs such as basil, borage, chives, coriander, lavender, lovage, mint, oregano, parsley, rosemary, sage and thyme; legumes for example beans, lentils, peas and soya beans; nuts for example almond, cashew, ground nut, hazelnut, peanut, pecan, pistachio and walnut; palms for example oil palm; ornamentals for example flowers, shrubs and trees; other trees, for example cacao, coconut, olive and rubber; vegetables for example asparagus, aubergine, broccoli, cabbage, carrot, cucumber, garlic, lettuce, marrow, melon, okra, onion, pepper, potato, pumpkin, rhubarb, spinach and tomato; and vines for example grapes.
- herbs such as basil, borage, chives, coriander, lavender, lovage, mint, oregano, parsley, rosemary, sage and thyme
- legumes for example beans, lentils, peas and soya beans
- useful plants is to be understood as also including useful plants that have been rendered tolerant to herbicides like bromoxynil or classes of herbicides (such as, for example, HPPD inhibitors, ALS inhibitors, for example primisulfuron, prosulfuron and trifloxysulfuron, EPSPS (5-enol-pyrovyl-shikimate-3-phosphate- synthase) inhibitors, GS (glutamine synthetase) inhibitors or PPO (protoporphyrinogen-oxidase) inhibitors) as a result of conventional methods of breeding or genetic engineering.
- herbicides like bromoxynil or classes of herbicides
- ALS inhibitors for example primisulfuron, prosulfuron and trifloxysulfuron
- EPSPS (5-enol-pyrovyl-shikimate-3-phosphate- synthase) inhibitors
- GS glutamine synthetase
- PPO protoporphyrinogen-oxida
- An example of a crop that has been rendered tolerant to imidazolinones, e.g., imazamox, by conventional methods of breeding (mutagenesis) is Clearfield® summer rape (Canola).
- crops that have been rendered tolerant to herbicides or classes of herbicides by genetic engineering methods include glyphosate- and glufosinate-resistant maize varieties commercially available under the trade names RoundupReady®, Herculex I® and LibertyLink®.
- useful plants is to be understood as also including useful plants which have been so transformed by the use of recombinant DNA techniques that they are capable of synthesising one or more selectively acting toxins, such as are known, for example, from toxin-producing bacteria, especially those of the genus Bacillus.
- YieldGard® (maize variety that expresses a CrylA(b) toxin); YieldGard Rootworm® (maize variety that expresses a Cry II IB(b1 ) toxin); YieldGard Plus® (maize variety that expresses a CrylA(b) and a CrylllB(bl) toxin); Starlink® (maize variety that expresses a Cry9(c) toxin); Herculex I® (maize variety that expresses a CrylF(a2) toxin and the enzyme phosphinothricine N-acetyltransferase (PAT) to achieve tolerance to the herbicide glufosinate ammonium); NuCOTN 33B® (cotton variety that expresses a CrylA(c) toxin); Bollgard I® (cotton variety that expresses a CrylA(c) toxin); Bollgard II® (cotton variety that
- crops is to be understood as including also crop plants which have been so transformed using recombinant DNA techniques that they are capable of synthesising one or more selectively acting toxins, such as are known, for example, from toxin-producing bacteria, especially those of the genus Bacillus.
- Toxins that can be expressed by such transgenic plants include, for example, insecticidal proteins from Bacillus cereus or Bacillus popilliae; or insecticidal proteins from Bacillus thuringiensis, such as delta-endotoxins, e.g. CrylAb, CrylAc, Cry1 F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), e.g. Vip1 , Vip2, Vip3 or Vip3A; or insecticidal proteins of bacteria colonising nematodes, for example Photorhabdus spp.
- insecticidal proteins from Bacillus cereus or Bacillus popilliae such as delta-endotoxins, e.g. CrylAb, CrylAc, Cry1 F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins
- Xenorhabdus spp. such as Photorhabdus luminescens, Xenorhabdus nematophilus
- toxins produced by animals such as scorpion toxins, arachnid toxins, wasp toxins and other insect-specific neurotoxins
- toxins produced by fungi such as Streptomycetes toxins, plant lectins, such as pea lectins, barley lectins or snowdrop lectins
- agglutinins proteinase inhibitors, such as trypsin inhibitors, serine protease inhibitors, patatin, cystatin, papain inhibitors
- steroid metabolism enzymes such as 3-hydroxysteroidoxidase, ecdysteroid- UDP-glycosyl-transferase, cholesterol oxidases, ecd
- delta-endotoxins for example CrylAb, CrylAc, Cry1 F, Cry1 Fa2, Cry2Ab, Cry3A, Cry3Bb1 or Cry9C, or vegetative insecticidal proteins (Vip), for example Vip1 , Vip2, Vip3 or Vip3A, expressly also hybrid toxins, truncated toxins and modified toxins.
- Hybrid toxins are produced recombinantly by a new combination of different domains of those proteins (see, for example, W002/15701).
- Truncated toxins for example a truncated CrylAb, are known.
- modified toxins one or more amino acids of the naturally occurring toxin are replaced.
- amino acid replacements preferably non-naturally present protease recognition sequences are inserted into the toxin, such as, for example, in the case of Cry3A055, a cathepsin-G-recognition sequence is inserted into a Cry3A toxin (see W02003/018810).
- Examples of such toxins or transgenic plants capable of synthesising such toxins are disclosed, for example, in EP-0374753, WO93/07278, WO95/34656, EP0427529, EP0451878 and W003/052073.
- the toxin contained in the transgenic plants imparts to the plants tolerance to harmful insects.
- insects can occur in any taxonomic group of insects but are especially commonly found in the beetles (Coleoptera), two-winged insects (Diptera) and butterflies (Lepidoptera).
- Transgenic plants containing one or more genes that code for an insecticidal resistance and express one or more toxins are known and some of them are commercially available. Examples of such plants are: YieldGard® (maize variety that expresses a Cry1 Ab toxin); YieldGard Rootworm® (maize variety that expresses a Cry3Bb1 toxin); YieldGard Plus® (maize variety that expresses a CrylAb and a Cry3Bb1 toxin); Starlink® (maize variety that expresses a Cry9C toxin); Herculex I® (maize variety that expresses a Cry1 Fa2 toxin and the enzyme phosphinothricine N-acetyltransferase (PAT) to achieve tolerance to the herbicide glufosinate ammonium); NuCOTN 33B® (cotton variety that expresses a CrylAc toxin); Bollgard I® (cotton variety that expresses
- transgenic crops are:
- Bt1 1 Maize from Syngenta Seeds SAS, Chemin de I'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Genetically modified Zea mays which has been rendered resistant to attack by the European corn borer (Ostrinia nubilalis and Sesamia nonagrioides) by transgenic expression of a truncated CrylAb toxin. Bt11 maize also transgenically expresses the enzyme PAT to achieve tolerance to the herbicide glufosinate ammonium.
- MIR604 Maize from Syngenta Seeds SAS, Chemin de I'Hobit 27, F-31 790 St. Sauveur, France, registration number C/FR/96/05/10. Maize which has been rendered insect-resistant by transgenic expression of a modified Cry3A toxin. This toxin is Cry3A055 modified by insertion of a cathepsin-G-protease recognition sequence. The preparation of such transgenic maize plants is described in W02003/018810. 4.
- MON 863 Maize from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B-1150 Brussels, Belgium, registration number C/DE/02/9. MON 863 expresses a Cry3Bb1 toxin and has resistance to certain Coleoptera insects.
- NK603 x MON 810 Maize from Monsanto Europe S.A. 270-272 Avenue de Tervuren, B 1150 Brussels, Belgium, registration number C/GB/02/M3/03. Consists of conventionally bred hybrid maize varieties by crossing the genetically modified varieties NK603 and MON 810.
- NK603 x MON 810 Maize transgenically expresses the protein CP4 EPSPS, obtained from Agrobacterium sp. strain CP4, which imparts tolerance to the herbicide Roundup® (contains glyphosate), and also a CrylAb toxin obtained from Bacillus thuringiensis subsp. kurstaki which brings about tolerance to certain Lepidoptera, include the European corn borer.
- the compounds of formula (I) according to the invention may be used in controlling or preventing phytopathogenic diseases, especially phytopathogenic fungi such as Alternaria species in fruits, vegetables and potatoes; Botrytis cinerea in strawberries, tomatoes, sunflower, pulse crops, vegetables and grapes; Rhizoctonia solani in potatoes and vegetables; Uncinula necator in grapes; Cladosporium cucumerinum, Didymella bryoniae, Sphaerotheca fuliginea and Glomerella lagenarium in cucurbits; Leveillula taurica in cucurbits and solanacious crops; Fusarium spp. in cereals; Leptosphaeria spp. in cereals; and Zymospetoria spp. in cereals.
- phytopathogenic diseases especially phytopathogenic fungi such as Alternaria species in fruits, vegetables and potatoes; Botrytis cinerea in strawberries, tomatoes, sunflower, pulse crops, vegetables and grapes; Rhizoctonia solani in potatoes and vegetables; Uncinul
- locus means fields in or on which plants are growing, or where seeds of cultivated plants are sown, or where seed will be placed into the soil. It includes soil, seeds, and seedlings, as well as established vegetation.
- plants refers to all physical parts of a plant, including seeds, seedlings, saplings, roots, tubers, stems, stalks, foliage, and fruits.
- plant propagation material is understood to denote generative parts of the plant, such as seeds, which can be used for the multiplication of the latter, and vegetative material, such as cuttings or tubers, for example potatoes.
- vegetative material such as cuttings or tubers, for example potatoes.
- seeds in the strict sense
- roots in the strict sense
- fruits in the tubers
- bulbs rhizomes
- parts of plants may also be mentioned.
- Germinated plants and young plants which are to be transplanted after germination or after emergence from the soil, may also be mentioned. These young plants can be protected before transplantation by a total or partial treatment by immersion.
- plant propagation material is understood to denote seeds.
- the compounds of formula (I) according to the invention may be used in unmodified form or, preferably, together with the adjuvants conventionally employed in the art of formulation. To this end they may be conveniently formulated in known manner to emulsifiable concentrates, coatable pastes, directly sprayable or dilutable solutions or suspensions, dilute emulsions, wettable powders, soluble powders, dusts, granulates, and also encapsulations e.g., in polymeric substances. As with the type of the compositions, the methods of application, such as spraying, atomising, dusting, scattering, coating or pouring, are chosen in accordance with the intended objectives and the prevailing circumstances. The compositions may also contain further adjuvants such as stabilizers, antifoams, viscosity regulators, binders or tackifiers as well as fertilizers, micronutrient donors or other formulations for obtaining special effects.
- Suitable carriers and adjuvants can be solid or liquid and are substances useful in formulation technology, e.g. natural or regenerated mineral substances, solvents, dispersants, wetting agents, tackifiers, thickeners, binders or fertilizers.
- Such carriers are for example described in WO1997/33890.
- Suspension concentrates are aqueous formulations in which finely divided solid particles of the active compound are suspended. Such formulations include anti-settling agents and dispersing agents and may further include a wetting agent to enhance activity as well an anti-foam and a crystal growth inhibitor. In use, these concentrates are diluted in water and normally applied as a spray to the area to be treated. The amount of active ingredient may range from 0.5% to 95% of the concentrate.
- Wettable powders are in the form of finely divided particles which disperse readily in water or other liquid carriers.
- the particles contain the active ingredient retained in a solid matrix.
- Typical solid matrices include fuller’s earth, kaolin clays, silicas and other readily wet organic or inorganic solids. Wettable powders normally contain from 5% to 95% of the active ingredient plus a small amount of wetting, dispersing or emulsifying agent.
- Emulsifiable concentrates are homogeneous liquid compositions dispersible in water or other liquid and may consist entirely of the active compound with a liquid or solid emulsifying agent, or may also contain a liquid carrier, such as xylene, heavy aromatic naphthas, isophorone and other non-volatile organic solvents. In use, these concentrates are dispersed in water or other liquid and normally applied as a spray to the area to be treated. The amount of active ingredient may range from 0.5% to 95% of the concentrate.
- Granular formulations include both extrudates and relatively coarse particles and are usually applied without dilution to the area in which treatment is required.
- Typical carriers for granular formulations include sand, fuller’s earth, attapulgite clay, bentonite clays, montmorillonite clay, vermiculite, perlite, calcium carbonate, brick, pumice, pyrophyllite, kaolin, dolomite, plaster, wood flour, ground corn cobs, ground peanut hulls, sugars, sodium chloride, sodium sulphate, sodium silicate, sodium borate, magnesia, mica, iron oxide, zinc oxide, titanium oxide, antimony oxide, cryolite, gypsum, diatomaceous earth, calcium sulphate and other organic or inorganic materials which absorb or which can be coated with the active compound.
- Granular formulations normally contain 5% to 25% of active ingredients which may include surface-active agents such as heavy aromatic naphthas, kerosene and other petroleum fractions, or vegetable oils
- Dusts are free-flowing admixtures of the active ingredient with finely divided solids such as talc, clays, flours, and other organic and inorganic solids which act as dispersants and carriers.
- Microcapsules are typically droplets or granules of the active ingredient enclosed in an inert porous shell which allows escape of the enclosed material to the surroundings at controlled rates. Encapsulated droplets are typically 1 to 50 microns in diameter.
- the enclosed liquid typically constitutes 50 to 95% of the weight of the capsule and may include solvent in addition to the active compound.
- Encapsulated granules are generally porous granules with porous membranes sealing the granule pore openings, retaining the active species in liquid form inside the granule pores.
- Granules typically range from 1 millimetre to 1 centimetre and preferably 1 to 2 millimetres in diameter. Granules are formed by extrusion, agglomeration or prilling, or are naturally occurring. Examples of such materials are vermiculite, sintered clay, kaolin, attapulgite clay, sawdust, and granular carbon.
- Shell or membrane materials include natural and synthetic rubbers, cellulosic materials, styrene-butadiene copolymers, polyacrylonitriles, polyacrylates, polyesters, polyamides, polyureas, polyurethanes and starch xanthates.
- compositions for agrochemical applications include simple solutions of the active ingredient in a solvent in which it is completely soluble at the desired concentration, such as acetone, alkylated naphthalenes, xylene and other organic solvents.
- Pressurised sprayers wherein the active ingredient is dispersed in finely divided form as a result of vaporisation of a low boiling dispersant solvent carrier, may also be used.
- Suitable agricultural adjuvants and carriers that are useful in formulating the compositions of the invention in the formulation types described above are well known to a person skilled in the art.
- Liquid carriers that can be employed include, for example, water, toluene, xylene, petroleum naphtha, crop oil, acetone, methyl ethyl ketone, cyclohexanone, acetic anhydride, acetonitrile, acetophenone, amyl acetate, 2- butanone, chlorobenzene, cyclohexane, cyclohexanol, alkyl acetates, diacetonalcohol, 1 ,2-dichloropropane, diethanolamine, p diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, N,N-dimethyl formamide, dimethyl sulfoxide, 1 ,4- dioxane, dipropylene glycol, dipropylene glycol methyl ether, dipropylene glycol dibenz
- Water is generally the carrier of choice for the dilution of concentrates.
- suitable solid carriers include, for example, talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, chalk, diatomaxeous earth, lime, calcium carbonate, bentonite clay, fuller’s earth, cotton seed hulls, wheat flour, soybean flour, pumice, wood flour, walnut shell flour and lignin.
- a broad range of surface-active agents are advantageously employed in both said liquid and solid compositions, especially those designed to be diluted with carrier before application.
- These agents when used, normally comprise from 0.1 % to 15% by weight of the formulation. They can be anionic, cationic, non-ionic or polymeric in character and can be employed as emulsifying agents, wetting agents, suspending agents or for other purposes.
- Typical surface-active agents include salts of alkyl sulfates, such as diethanolammonium lauryl sulphate; alkylarylsulfonate salts, such as calcium dodecylbenzenesulfonate; alkylphenol-alkylene oxide addition products, such as nonylphenol-C.sub.
- alcohol-alkylene oxide addition products such as tridecyl alcohol-C.sub. 16 ethoxylate
- soaps such as sodium stearate
- alkylnaphthalenesulfonate salts such as sodium dibutylnaphthalenesulfonate
- dialkyl esters of sulfosuccinate salts such as sodium di(2 ethylhexyl) sulfosuccinate
- sorbitol esters such as sorbitol oleate
- quaternary amines such as lauryl trimethylammonium chloride
- polyethylene glycol esters of fatty acids such as polyethylene glycol stearate
- salts of mono and dialkyl phosphate esters such as mono and dialkyl phosphate esters.
- adjuvants commonly utilized in agricultural compositions include crystallisation inhibitors, viscosity modifiers, suspending agents, spray droplet modifiers, pigments, antioxidants, foaming agents, anti-foaming agents, light-blocking agents, compatibilizing agents, antifoam agents, sequestering agents, neutralising agents and buffers, corrosion inhibitors, dyes, odorants, spreading agents, penetration aids, micronutrients, emollients, lubricants and sticking agents.
- biocidal active ingredients or compositions may be combined with the compositions of the invention and used in the methods of the invention and applied simultaneously or sequentially with the compositions of the invention. When applied simultaneously, these further active ingredients may be formulated together with the compositions of the invention or mixed in, for example, the spray tank. These further biocidal active ingredients may be fungicides, herbicides, insecticides, bactericides, acaricides, nematicides and/or plant growth regulators.
- Pesticidal agents are referred to herein using their common name are known, for example, from “The Pesticide Manual”, 15th Ed., British Crop Protection Council 2009.
- compositions of the invention may also be applied with one or more systemically acquired resistance inducers (“SAR” inducer).
- SAR inducers are known and described in, for example, United States Patent No. US 6,919,298 and include, for example, salicylates and the commercial SAR inducer acibenzolar- S-methyl.
- the compounds of formula (I) according to the invention are normally used in the form of agrochemical compositions and can be applied to the crop area or plant to be treated, simultaneously or in succession with further compounds.
- further compounds can be e.g., fertilizers or micronutrient donors or other preparations, which influence the growth of plants. They can also be selective herbicides or non-selective herbicides as well as insecticides, fungicides, bactericides, nematicides, molluscicides or mixtures of several of these preparations, if desired together with further carriers, surfactants or application promoting adjuvants customarily employed in the art of formulation.
- the compounds of formula (I) according to the invention may be used in the form of (fungicidal) compositions for controlling or protecting against phytopathogenic microorganisms, comprising as active ingredient at least one compound of formula (I) or of at least one preferred individual compound as defined herein, in free form or in agrochemical usable salt form, and at least one of the above-mentioned adjuvants.
- the invention therefore provides a composition, preferably a fungicidal composition, comprising at least one compound of formula (I) according to the invention, an agriculturally acceptable carrier and optionally an adjuvant.
- An agricultural acceptable carrier is for example a carrier that is suitable for agricultural use.
- Agricultural carriers are well known in the art.
- said composition may comprise at least one or more pesticidal-active compounds, for example an additional fungicidal active ingredient in addition to the compound of formula (I).
- the compound of formula (I) according to the invention may be the sole active ingredient of a composition or it may be admixed with one or more additional active ingredients such as a pesticide, fungicide, synergist, herbicide or plant growth regulator where appropriate.
- An additional active ingredient may, in some cases, result in unexpected synergistic activities.
- Suitable additional active ingredients include the following: acycloamino acid fungicides, aliphatic nitrogen fungicides, amide fungicides, anilide fungicides, antibiotic fungicides, aromatic fungicides, arsenical fungicides, aryl phenyl ketone fungicides, benzamide fungicides, benzanilide fungicides, benzimidazole fungicides, benzothiazole fungicides, botanical fungicides, bridged diphenyl fungicides, carbamate fungicides, carbanilate fungicides, conazole fungicides, copper fungicides, dicarboximide fungicides, dinitrophenol fungicides, dithiocarbamate fungicides, dithiolane fungicides, furamide fungicides, furanilide fungicides, hydrazide fungicides, imidazole fungicides, mercury fungicides, morpholine fung
- Suitable additional active ingredients include the following: petroleum oils, 1 ,1 -bis(4-chlorophenyl)- 2-ethoxyethanol, 2,4-dichlorophenyl benzenesulfonate, 2-fluoro-N-methyl-N-1 -naphthylacetamide, 4- chlorophenyl phenyl sulfone, acetoprole, aldoxycarb, amidithion, amidothioate, amiton, amiton hydrogen oxalate, amitraz, aramite, arsenous oxide, azobenzene, azothoate, benomyl, benoxa-fos, benzyl benzoate, bixafen, brofenvalerate, bromocyclen, bromophos, bromopropylate, buprofezin, butocarboxim, butoxycarboxim, butylpyridaben, calcium polysulfide, camphechlor, carban
- Adoxophyes orana GV Agrobacterium radiobacter, Amblyseius spp., Anagrapha falcifera NPV, Anagrus atomus, Aphelinus abdominalis, Aphidius colemani, Aphidoletes aphidimyza, Autographa californica NPV, Bacillus sphaericus Neide, Beauveria brongniartii, Chrysoperla carnea, Cryptolaemus montrouzieri, Cydia pomonella GV, Dacnusa sibirica, Diglyphus isaea, Encarsia formosa, Eretmocerus eremicus, Heterorhabditis bacteriophora and H.
- lecontei NPV, Orius spp. Paecilomyces fumosoroseus, Phytoseiulus persimilis, Steinernema bibionis, Steinernema carpocapsae, Steinernema feltiae, Steinernema glaseri, Steinernema riobrave, Steinernema riobravis, Steinernema scapterisci, Steinernema spp., Trichogramma spp., Typhlodromus occidentalis, Verticillium lecanii, apholate, bisazir, busulfan, dimatif, hemel, hempa, metepa, methiotepa, methyl apholate, morzid, penfluron, tepa, thiohempa, thiotepa, tretamine, uredepa, (E)-dec-5-en-1-yl acetate
- this compound may be prepared from the methods described in WO2016/156290); 3-[2-(1-chlorocyclopropyl)- 3-(3-chloro-2-fluoro-phenyl)-2-hydroxy-propyl]imidazole-4-carbonitrile (this compound may be prepared from the methods described in WO2016/156290); (4-phenoxyphenyl)methyl 2-amino-6-methyl-pyridine-3- carboxylate (this compound may be prepared from the methods described in WO2014/006945); 2,6-Dimethyl- 1 H,5H-[1 ,4]dithiino[2,3-c:5,6-c']dipyrrole-1 ,3,5,7(2H,6H)-tetrone (this compound may be prepared from the methods described in WO2011/138281) N-methyl-4-[5-(trifluoromethyl)-1 ,2,4-oxadiazol-3- yl]benzenecarbothioamide; N-methyl-4-[5-(
- the compounds of the invention may also be used in combination with anthelmintic agents.
- anthelmintic agents include, compounds selected from the macrocyclic lactone class of compounds such as ivermectin, avermectin, abamectin, emamectin, eprinomectin, doramectin, selamectin, moxidectin, nemadectin and milbemycin derivatives as described in EP0357460, EP0444964 and EP0594291 .
- Additional anthelmintic agents include semisynthetic and biosynthetic avermectin/milbemycin derivatives such as those described in US5, 015,630, WO9415944 and WO9522552. Additional anthelmintic agents include the benzimidazoles such as albendazole, cambendazole, fenbendazole, flubendazole, mebendazole, oxfendazole, oxibendazole, parbendazole, and other members of the class. Additional anthelmintic agents include imidazothiazoles and tetrahydropyrimidines such as tetramisole, levamisole, pyrantel pamoate, oxantel or morantel. Additional anthelmintic agents include flukicides, such as triclabendazole and clorsulon and the cestocides, such as praziquantel and epsiprantel.
- the compounds of the invention may be used in combination with derivatives and analogues of the paraherquamide/marcfortine class of anthelmintic agents, as well as the antiparasitic oxazolines such as those disclosed in US5478855, US4639771 and DE-19520936.
- the compounds of the invention may be used in combination with derivatives and analogues of the general class of dioxomorpholine antiparasitic agents as described in WO9615121 and also with anthelmintic active cyclic depsipeptides such as those described in WO961 1945, WO9319053, WO9325543, EP0626375, EP0382173, WO9419334, EP0382173, and EP0503538.
- the compounds of the invention may be used in combination with other ectoparasiticides; for example, fipronil; pyrethroids; organophosphates; insect growth regulators such as lufenuron; ecdysone agonists such as tebufenozide and the like; neonicotinoids such as imidacloprid and the like.
- ectoparasiticides for example, fipronil; pyrethroids; organophosphates; insect growth regulators such as lufenuron; ecdysone agonists such as tebufenozide and the like; neonicotinoids such as imidacloprid and the like.
- the compounds of the invention may be used in combination with terpene alkaloids, for example those described in WO95/19363 or W004/72086, particularly the compounds disclosed therein.
- Organophosphates acephate, azamethiphos, azinphos-ethyl, azinphos- methyl, bromophos, bromophos- ethyl, cadusafos, chlorethoxyphos, chlorpyrifos, chlorfenvinphos, chlormephos, demeton, demeton-S-methyl, demeton-S-methyl sulphone, dialifos, diazinon, dichlorvos, dicrotophos, dimethoate, disulfoton, ethion, ethoprophos, etrimfos, famphur, fenamiphos, fenitrothion, fensulfothion, fenthion, flupyrazofos, fonofos, formothion, fosthiazate, heptenophos, isazophos, isothioate, isoxathion, malathion,
- Carbamates alanycarb, aldicarb, 2-sec-butylphenyl methylcarbamate, benfuracarb, carbaryl, carbofuran, carbosulfan, cloethocarb, ethiofencarb, fenoxycarb, fenthiocarb, furathiocarb, HCN-801 , isoprocarb, indoxacarb, methiocarb, methomyl, 5-methyl-m-cumenylbutyryl(methyl)carbamate, oxamyl, pirimicarb, propoxur, thiodicarb, thiofanox, triazamate, UC-51717.
- Pyrethroids acrinathin, allethrin, alphametrin, 5-benzyl-3-furylmethyl (E)-(1 R)-cis-2,2-dimethyl-3-(2- oxothiolan-3-ylidenemethyl)cyclopropanecarboxylate, bifenthrin, beta-cyfluthrin, cyfluthrin, a-cypermethrin, beta-cypermethrin, bioallethrin, bioallethrin((S)-cyclopentylisomer), bioresmethrin, bifenthrin, NCI-85193, cycloprothrin, cyhalothrin, cythithrin, cyphenothrin, deltamethrin, empenthrin, esfenvalerate, ethofenprox, fenfluthrin, fenpropathrin, fenvaler
- Arthropod growth regulators a) chitin synthesis inhibitors: benzoylureas: chlorfluazuron, diflubenzuron, fluazuron, flucycloxuron, flufenoxuron, hexaflumuron, lufenuron, novaluron, teflubenzuron, triflumuron, buprofezin, diofenolan, hexythiazox, etoxazole, chlorfentazine; b) ecdysone antagonists: halofenozide, methoxyfenozide, tebufenozide; c) juvenoids: pyriproxyfen, methoprene (including S-methoprene), fenoxycarb; d) lipid biosynthesis inhibitors: spirodiclofen.
- antiparasitics acequinocyl, amitraz, AKD-1022, ANS-118, azadirachtin, Bacillus thuringiensis, bensultap, bifenazate, binapacryl, bromopropylate, BTG-504, BTG-505, camphechlor, cartap, chlorobenzilate, chlordimeform, chlorfenapyr, chromafenozide, clothianidine, cyromazine, diacloden, diafenthiuron, DBI-3204, dinactin, dihydroxymethyldihydroxypyrrolidine, dinobuton, dinocap, endosulfan, ethiprole, ethofenprox, fenazaquin, flumite, MTI-800, fenpyroximate, fluacrypyrim, flubenzimine, flubrocythrinate, flufenzine, flufenprox, fluproxyfen, halofenprox, hydr
- Biological agents Bacillus thuringiensis ssp. aizawai, kurstaki, Bacillus thuringiensis delta endotoxin, baculovirus, entomopathogenic bacteria, virus, and fungi.
- Bactericides chlortetracycline, oxytetracycline, streptomycin.
- TX means one compound selected from compounds of formula (I), (l-A), (I-A1), (I-A2), (I-A3), (I-A4), (I-A5), (l-B), (l-C), or (l-D), or compounds selected from compounds listed in Tables A-1 to A-26, or compounds listed in Table P (below), and a compound selected from the group of substances consisting of petroleum oils + TX, 1 ,1-bis(4-chlorophenyl)-2-ethoxyethanol + TX, 2,4-dichlorophenyl benzenesulfonate + TX, 2-fluoro-N-methyl- N-1 -naphthylacetamide + TX, 4-chlorophenyl phenyl sulfone + TX, acetoprole + TX, aldoxycarb + TX, amidithion + TX,
- TX Paecilomyces fumosoroseus + TX, Phytoseiulus persimilis + TX, Steinernema bibionis + TX, Steinernema carpocapsae + TX, Steinernema feltiae + TX, Steinernema glaseri + TX, Steinernema riobrave + TX, Steinernema riobravis + TX, Steinernema scapterisci + TX, Steinernema spp. + TX, Trichogramma spp.
- the compounds in this paragraph may be prepared from the methods described in WO2017/055473, WO2017/055469, WO2017/093348 and WO2017/118689; 2-[6-(4- chlorophenoxy)-2-(trifluoromethyl)-3-pyridyl]-1-(1 ,2,4-triazol-1-yl)propan-2-ol + TX (this compound may be prepared from the methods described in WO2017/029179); 2-[6-(4-bromophenoxy)-2-(trifluoromethyl)-3- pyridyl]-1-(1 ,2,4-triazol-1-yl)propan-2-ol + TX (this compound may be prepared from the methods described in WO2017/029179); 3-[2-(1-chlorocyclopropyl)-3-(2-fluorophenyl)-2-hydroxy-propyl]imidazole-4-carbonitrile + TX (this compound may be prepared from the methods described in WO2016
- the active ingredient mixture of the compound selected from compounds of (I), (l-A), (I-A1), (I-A2), (I-A3), (I- A4), (I-A5), (l-B), (l-C), or (l-D), or compounds selected from compounds listed in Tables A-1 to A-26, or compounds listed in Table P (below), is preferably in a mixing ratio of from 100:1 to 1 :100, especially from 50:1 to 1 :50, more especially in a ratio of from 20:1 to 1 :20, even more especially from 10:1 to 1 :10, and still more especially from 5:1 to 1 :5 Those mixing ratios are by weight.
- the mixtures as described above can be used in a method for controlling pests, which comprises applying a composition comprising a mixture as described above to the pests or their environment, with the exception of a method for treatment of the human or animal body by surgery or therapy and diagnostic methods practised on the human or animal body.
- the mixtures comprising a compound selected from compounds of formula (I), (l-A), (I-A1), (I-A2), (I-A3), (I- A4), (I-A5), (l-B), (l-C), or (l-D), or compounds selected from compounds listed in Tables A-1 to A-26, or compounds listed in Table P (below), and one or more active ingredients as described above can be applied, for example, in a single “ready-mix” form, in a combined spray mixture composed from separate formulations of the single active ingredient components, such as a “tank-mix”, and in a combined use of the single active ingredients when applied in a sequential manner, i.e. one after the other with a reasonably short period, such as a few hours or days.
- compositions according to the invention can also comprise further solid or liquid auxiliaries, such as stabilizers, for example unepoxidized or epoxidized vegetable oils (for example epoxidized coconut oil, rapeseed oil or soya oil), antifoams, for example silicone oil, preservatives, viscosity regulators, binders and/or tackifiers, fertilizers or other active ingredients for achieving specific effects, for example bactericides, fungicides, nematocides, plant activators, molluscicides or herbicides.
- auxiliaries such as stabilizers, for example unepoxidized or epoxidized vegetable oils (for example epoxidized coconut oil, rapeseed oil or soya oil), antifoams, for example silicone oil, preservatives, viscosity regulators, binders and/or tackifiers, fertilizers or other active ingredients for achieving specific effects, for example bactericides, fungicides, nematocides
- compositions according to the invention are prepared in a manner known per se, in the absence of auxiliaries for example by grinding, screening and/or compressing a solid active ingredient and in the presence of at least one auxiliary for example by intimately mixing and/or grinding the active ingredient with the auxiliary (auxiliaries).
- auxiliaries for example by grinding, screening and/or compressing a solid active ingredient and in the presence of at least one auxiliary for example by intimately mixing and/or grinding the active ingredient with the auxiliary (auxiliaries).
- Another aspect of the invention is related to the use of a compound of formula (I) according to the invention or of a preferred individual compound as defined herein, of a composition comprising at least one compound of formula (I) or at least one preferred individual compound as defined herein, or of a fungicidal or insecticidal mixture comprising at least one compound of formula (I) or at least one preferred individual compound as defined herein, in admixture with other fungicides or insecticides as described above, for controlling or preventing infestation of plants, e.g. useful plants such as crop plants, propagation material thereof, e.g. seeds, harvested crops, e.g. harvested food crops, or non-living materials by insects or by phytopathogenic microorganisms, preferably fungal organisms.
- useful plants such as crop plants, propagation material thereof, e.g. seeds, harvested crops, e.g. harvested food crops, or non-living materials by insects or by phytopathogenic microorganisms, preferably fungal organisms.
- a further aspect of invention is related to a method of controlling or preventing an infestation of plants, e.g. useful plants such as crop plants, propagation material thereof, e.g. seeds, harvested crops, e.g. harvested food crops, or of non-living materials by phytopathogenic or spoilage microorganisms or organisms potentially harmful to man, especially fungal organisms, which comprises the application of a compound of formula (I) according to the invention or of a preferred individual compound as defined herein as active ingredient to the plants, to parts of the plants or to the locus thereof, to the propagation material thereof, or to any part of the non-living materials.
- plants e.g. useful plants such as crop plants, propagation material thereof, e.g. seeds, harvested crops, e.g. harvested food crops, or of non-living materials by phytopathogenic or spoilage microorganisms or organisms potentially harmful to man, especially fungal organisms
- a compound of formula (I) according to the invention or of a preferred individual compound as defined herein as active ingredient to the
- Controlling or preventing means reducing infestation by insects or by phytopathogenic or spoilage microorganisms or organisms potentially harmful to man, especially fungal organisms, to such a level that an improvement is demonstrated.
- a preferred method of controlling or preventing an infestation of crop plants by phytopathogenic microorganisms, especially fungal organisms, or insects which comprises the application of a compound of formula (I) according to the invention, or an agrochemical composition which contains at least one compound of formula (I), is foliar application.
- the frequency of application and the rate of application will depend on the risk of infestation by the corresponding pathogen or insect.
- the compounds of formula (I) according to the invention can also penetrate the plant through the roots via the soil (systemic action) by drenching the locus of the plant with a liquid formulation, or by applying the compounds in solid form to the soil, e.g. in granular form (soil application). In crops of water rice such granulates can be applied to the flooded rice field.
- the compounds of formula (I) may also be applied to seeds (coating) by impregnating the seeds or tubers either with a liquid formulation of the fungicide or coating them with a solid formulation.
- a formulation e.g. a composition containing the compound of formula (I) according to the invention and, if desired, a solid or liquid adjuvant or monomers for encapsulating the compound of formula (I), may be prepared in a known manner, typically by intimately mixing and/or grinding the compound with extenders, for example solvents, solid carriers and, optionally, surface active compounds (surfactants).
- Advantageous rates of application are normally from 5g to 2kg of active ingredient (a.i.) per hectare (ha), preferably from 10g to I kg a.i./ha, most preferably from 20g to 600g a.i./ha.
- convenient dosages are from 10mg to 1 g of active substance per kg of seeds.
- g a.i./ha refers to the application rate given in gram [g] of active ingredient [a.i.] per unit of surface [ha].
- the unit hectare symbol ha is the metric unit of area that equals a square with 100 m side (1 hm 2 ) or 10,000 square meters. Hectare is a commonly used unit of area in the metric system.
- rates of 0.001 to 50 g of a compound of formula (I) per kg of seed preferably from 0.01 to 10g per kg of seed are generally sufficient.
- composition comprising a compound of formula (I) according to the present invention is applied either preventative, meaning prior to disease development or curative, meaning after disease development.
- compositions of the invention may be employed in any conventional form, for example in the form of a twin pack, a powder for dry seed treatment (DS), an emulsion for seed treatment (ES), a flowable concentrate for seed treatment (FS), a solution for seed treatment (LS), a water dispersible powder for seed treatment (WS), a capsule suspension for seed treatment (CF), a gel for seed treatment (GF), an emulsion concentrate (EC), a suspension concentrate (SC), a suspo-emulsion (SE), a capsule suspension (CS), a water dispersible granule (WG), an emulsifiable granule (EG), an emulsion, water in oil (EG), an emulsion, oil in water (EW), a micro-emulsion (ME), an oil dispersion (OD), an oil miscible flowable (OF), an oil miscible liquid (OL), a soluble concentrate (SL), an ultra-low volume suspension (SU), an ultra-low volume liquid (UL), a technical concentrate (TK
- compositions may be produced in conventional manner, e.g. by mixing the active ingredients with appropriate formulation inerts (diluents, solvents, fillers and optionally other formulating ingredients such as surfactants, biocides, anti-freeze, stickers, thickeners and compounds that provide adjuvancy effects).
- appropriate formulation inerts diiluents, solvents, fillers and optionally other formulating ingredients such as surfactants, biocides, anti-freeze, stickers, thickeners and compounds that provide adjuvancy effects.
- conventional slow release formulations may be employed where long lasting efficacy is intended.
- Particularly formulations to be applied in spraying forms such as water dispersible concentrates (e.g. EC, SC, DC, OD, SE, EW, EO and the like), wettable powders and granules, may contain surfactants such as wetting and dispersing agents and other compounds that provide adjuvancy effects, e.g.
- a seed dressing formulation is applied in a manner known per se to the seeds employing the combination of the invention and a diluent in suitable seed dressing formulation form, e.g. as an aqueous suspension or in a dry powder form having good adherence to the seeds.
- suitable seed dressing formulation form e.g. as an aqueous suspension or in a dry powder form having good adherence to the seeds.
- seed dressing formulations are known in the art.
- Seed dressing formulations may contain the single active ingredients or the combination of active ingredients in encapsulated form, e.g., as slow release capsules or microcapsules.
- the formulations include from 0.01 to 90% by weight of active agent, from 0 to 20% agriculturally acceptable surfactant and 10 to 99.99% solid or liquid formulation inerts and adjuvant(s), the active agent consisting of at least the compound of formula (I) according to the invention optionally together with other active agents, particularly microbiocides or conservatives or the like.
- Concentrated forms of compositions generally contain in between about 2 and 80%, preferably between about 5 and 70% by weight of active agent.
- Application forms of formulation may for example contain from 0.01 to 20% by weight, preferably from 0.01 to 5% by weight of active agent. Whereas commercial products will preferably be formulated as concentrates, the end user will normally employ diluted formulations.
- the rates of application vary within wide limits and depend on the nature of the soil, the method of application, the crop plant, the pest to be controlled, the prevailing climatic conditions, and other factors governed by the method of application, the time of application and the target crop.
- compounds may be applied at a rate of from 1 to 2000 l/ha, especially from 10 to 1000 l/ha.
- Preferred formulations can have the following compositions (weight %):
- Emulsifiable concentrates active ingredient: 1 to 95 %, preferably 60 to 90 % surface-active agent: 1 to 30 %, preferably 5 to 20 % liquid carrier: 1 to 80 %, preferably 1 to 35 %
- Dusts active ingredient: 0.1 to 10 %, preferably 0.1 to 5 % solid carrier: 99.9 to 90 %, preferably 99.9 to 99 %
- Suspension concentrates active ingredient: 5 to 75 %, preferably 10 to 50 % water: 94 to 24 %, preferably 88 to 30 % surface-active agent: 1 to 40 %, preferably 2 to 30 %
- Wettable powders active ingredient: 0.5 to 90 %, preferably 1 to 80 % surface-active agent: 0.5 to 20 %, preferably 1 to 15 % solid carrier: 5 to 95 %, preferably 15 to 90 %
- Granules active ingredient: 0.1 to 30 %, preferably 0.1 to 15 % solid carrier: 99.5 to 70 %, preferably 97 to 85 %
- active ingredient 0.1 to 30 %, preferably 0.1 to 15 % solid carrier: 99.5 to 70 %, preferably 97 to 85 %
- Table A This table discloses 75 compounds of formula (la) according to the invention wherein: w as defined below:
- the following compounds represent specific compounds of formula (la) as described in Tables A-1 to A-26 wherein G is as defined in Table A.
- the compound A-1.G1 represents a compound of formula (la) wherein R 2 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 and B 2 are as defined in Table A-1 and G is G1 as defined in Table A.
- Table A-1 This table provides 75 compounds A-1.G1 to A-1.G75 of formula (la) wherein R 2 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 are H, B 2 is CH, and G is as defined in Table A.
- compound A-1 ,G3 has the following structure:
- Table A-2 This table provides 75 compounds A-2.G1 to A-2.G75 of formula (la) wherein R 2 is CH3, R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 are H, B 2 is CH, and G is as defined in Table A.
- compound A-2.G2 has the following structure:
- Table A-3 This table provides 75 compounds A-3.G1 to A-3.G75 of formula (la) wherein R 2 is F, R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 are H, B 2 is CH, and G is as defined in Table A.
- Table A-4 This table provides 75 compounds A-4.G1 to A-4.G75 of formula (la) wherein R 2 is Cl, R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 are H, B 2 is CH, and G is as defined in Table A.
- Table A-5 This table provides 75 compounds A-5.G1 to A-5.G75 of formula (la) wherein R 2 is cyclopropyl, R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 are H, B 2 is CH, and G is as defined in Table A.
- Table A-6 This table provides 75 compounds A-6.G1 to A-6.G75 of formula (la) wherein R 2 is COCH3, R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 are H, B 2 is CH, and G is as defined in Table A.
- Table A-8 This table provides 75 compounds A-8.G1 to A-8.G75 of formula (la) wherein R 2 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 are H, R 4 is CH3, B 2 is CH, and G is as defined in Table A.
- compound A-8.G32 has the following structure:
- Table A-10 This table provides 75 compounds A-10.G1 to A-10.G75 of formula (la) wherein R 5 , R 6 , R 7 , R 8 , R 9 , R 10 are H, R 2 and R 4 are CH3, B 2 is N, and G is as defined in Table A.
- Table A-11 This table provides 75 racem/c-compounds A-11 ,G1 to A-11 .G75 of formula (la) wherein R 5 , R 6 , R 8 , R 9 , R 10 are H, R 2 and R 7 are CH3, B 2 is CH, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- compound rac-A- 11 ,G2 has the following structure
- Table A-12 This table provides 75 racem/c-compounds A-12.G1 to A-12.G75 of formula (la) wherein R 5 , R 6 , R 8 , R 9 , R 10 are H, R 2 is Cl, R 7 is CH3, B 2 is CH and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- compound rac-A- 12.G67 has the following structure;
- Table A-13 This table provides 75 compounds A-13.G1 to A-13.G75 of formula (la) wherein R 5 , R 6 , R 8 , R 9 , R 10 are H, R 2 , R 7 , and R 4 are CH3, B 2 is CH, and G is as defined in Table A.
- Table A-14 This table provides 75 racem/c-compounds A-14.G1 to A-14.G75 of formula (la) wherein R 5 , R 6 , R 8 , R 9 , R 10 are H, R 2 and R 7 are CH3, B 2 is C-Br, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- Table A-15 This table provides 75 racem/c-compounds A-15.G1 to A-15.G75 of formula (la) wherein R 5 , R 6 , R 8 , R 9 , R 10 are H, R 2 and R 7 are CH3, B 2 is C-CN, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- Table A-16 This table provides 75 /'acem/c-compounds A-16.G1 to A-16.G75 of formula (la) wherein R 5 , R 6 , R 8 , R 9 , R 10 are H, R 2 is Cl, R 7 is CH3, B 2 is C-CI, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- Table A-17 This table provides 75 /'acem/c-compounds A-17.G1 to A-17.G75 of formula (la) wherein R 5 , R 6 , R 8 , R 9 , R 10 are H, R 2 is Cl, R 7 is CH3, B 2 is C-CN, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- compound rac-A- 17.G53 has the following structure;
- Table A-18 This table provides 15 compounds A-18.G1 to A-18.G15 of formula (la) wherein R 4 , R 5 , R 6 , R 8 , R 9 , and R 10 are H, R 2 and R 7 are CH3, B 2 is N and G is as defined in Table A.
- Table A-19 This table provides 15 racem/c-compounds A-19.G1 to A-19.G15 of formula (la) wherein R 4 , R 5 , R 6 , R 8 , and R 10 are H, R 9 is OCH3, R 2 and R 7 are CH3, B 2 is C-CI, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- compound rac-A-19.G15 has the following structure compound rac-A-19.G15
- Table A-20 This table provides 15 /'acem/c-compounds A-20.G1 to A-20.G15 of formula (la) wherein R 4 , R 5 , R 6 , R 8 , and R 10 are H, R 9 is OCH3, R 2 and R 7 are CH3, B 2 is C-H, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- Table A-21 This table provides 15 /'acem/c-compounds A-21 ,G1 to A-21 ,G15 of formula (la) wherein R 4 , R 5 , R 6 , R 8 , and R 10 are H, R 9 is Br, R 2 and R 7 are CH3, B 2 is C-H, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- Table A-22 This table provides 15 /'acem/c-compounds A-22.G1 to A-22.G15 of formula (la) wherein R 4 , R 5 , R 6 , R 8 , and R 10 are H, R 9 is Cl, R 2 and R 7 are CH3, B 2 is C-H, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- Table A-23 This table provides 15 racem/c-compounds A-23.G1 to A-23.G15 of formula (la) wherein R 4 , R 5 , R 6 , R 8 , and R 10 are H, R 9 is CN, R 2 and R 7 are CH3, B 2 is C-H, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- Table A-24 This table provides 15 /'acem/c-compounds A-24.G1 to A-24.G15 of formula (la) wherein R 4 , R 5 , R 6 , R 8 , and R 9 are H, R 10 is Br, R 2 and R 7 are CH3, B 2 is C-H, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- Table A-25 This table provides 15 /'acem/c-compounds A-25.G1 to A-25.G15 of formula (la) wherein R 4 , R 5 , R 6 , R 8 , and R 9 are H, R 10 is Cl, R 2 and R 7 are CH3, B 2 is C-H, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- Table A-26 This table provides 15 /'acem/c-compounds A-26.G1 to A-26.G15 of formula (la) wherein R 4 , R 5 , R 6 , R 8 , and R 9 are H, R 10 is CN, R 2 and R 7 are CH3, B 2 is C-H, and G is as defined in Table A.
- the pyrazole substituent and the R 7 CH3 substituent have a syn-relationship to each other.
- compound A-26.G6 has the following structure: compound rac-/ ⁇ -2Q.GQ
- the compounds of the invention can be distinguished from known compounds by virtue of greater efficacy at low application rates, which can be verified by a person skilled in the art using the experimental procedures outlined in the Examples, using lower application rates if necessary, for example 60 ppm, 20 ppm or 2 ppm.
- Compounds of formula (I) may possess any number of benefits including, inter alia, advantageous levels of biological activity for protecting plants against diseases that are caused by fungi or superior properties for use as agrochemical active ingredients (for example, greater biological activity, an advantageous spectrum of activity, an increased safety profile (including improved crop tolerance), improved physico-chemical properties, or increased biodegradability).
- advantageous levels of biological activity for protecting plants against diseases that are caused by fungi or superior properties for use as agrochemical active ingredients (for example, greater biological activity, an advantageous spectrum of activity, an increased safety profile (including improved crop tolerance), improved physico-chemical properties, or increased biodegradability).
- temperatures are given in degrees Celsius and “m.p.” means melting point.
- LC/MS means Liquid Chromatography Mass Spectroscopy and the description of the apparatus, and the methods is as follows.
- LC-MS Method A Spectra were recorded on a mass spectrometer from Waters (SQD, SQDII Single quadrupole mass spectrometer) equipped with an electrospray source (Polarity: positive and negative ions, Capillary: 3.00 kV, Cone range: 30 V, Extractor: 2.00 V, Source Temperature: 150°C, Desolvation Temperature: 350°C, Cone Gas Flow: 50 l/h, Desolvation Gas Flow: 650 l/h, Mass range: 100 to 900 Da) and an Acquity UPLC from Waters: binary pump, heated column compartment, diode-array detector and ELSD detector.
- LC-MS Method B Spectra were recorded on a ACQUITY Mass Spectrometer from Waters Corporations (SQD or SQDII Single quadrupole mass spectrometer) equipped with an electrospray source (Polarity: positive or negative ions, Capillary: 3.0 kV, Cone: 30V, Extractor: 3.00 V, Source Temperature: 150°C, Desolvation Temperature: 400°C, Cone Gas Flow: 60 L/hr, Desolvation Gas Flow: 700 L/hr, Mass range: 140 to 800 Da) and an ACQUITY UPLC from Waters Corporations with solvent degasser, binary pump, heated column compartment and diode-array detector.
- an electrospray source Polyity: positive or negative ions, Capillary: 3.0 kV, Cone: 30V, Extractor: 3.00 V, Source Temperature: 150°C, Desolvation Temperature: 400°C, Cone Gas Flow: 60 L/hr, Desolvation Gas Flow:
- LC-MS Method C Spectra were recorded on a Mass Spectrometer from Waters (Acquity QDa Mass Spectrometer) equipped with an electrospray source (Polarity: Positive and Negative Polarity Switch), Capillary: 0.8 kV, Cone range: 25 V, Extractor: V (No extractor voltage for Qda detector) Source Temperature: 120°C, Desolvation Temperature: 600°C, Cone Gas Flow: 50 L/h, Desolvation Gas Flow: 1000 L/h, Mass range: 110 to 850 Da) and an Acquity UPLC from Waters: Quaternary solvent manager, heated column compartment, diode-array detector.
- LC-MS Method D Spectra were recorded on a Mass Spectrometer from Waters (SQD2 or QDA Single quadrupole mass spectrometer) equipped with an electrospray source (Polarity: Positive and Negative Polarity Switch), Capillary: 0.8-3.00 kV, Cone range: 25 Source Temperature: 120-150°C, Desolvation Temperature: 500-600°C, Cone Gas Flow: 50 L/h, Desolvation Gas Flow: 1000 L/h, Mass range: 110 to 850 Da) and an Acquity UPLC from Waters: Quaternary solvent manager, heated column compartment, diode-array detector.
- LC-MS Method E Spectra were recorded on a Mass Spectrometer from Agilent Technologies (6410 Triple Quadrupole mass spectrometer) equipped with an electrospray source (Polarity: positive or negative ions, MS2 Scan, Capillary: 4.00 kV, Fragmentor: 100 V, Desolvation Temperature: 350°C, Gas Flow: 11 L/min, Nebulizer Gas: 45 psi, Mass range: 110 to 1000 Da) and a 1200 Series HPLC from Agilent: quaternary pump, heated column compartment and VWD detector.
- Wettable powders a) b) c) active ingredients 25 % 50 % 75 % sodium lignosulfonate 5 % 5 % - sodium lauryl sulfate 3 % - 5 % sodium diisobutylnaphthalenesulfonate 6 % 10 % phenol polyethylene glycol ether (7-8 mol ethylene oxide) - 2 % - highly dispersed silicic acid 5 % 10 % 10 % Kaolin 62 % 27 % -
- the combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording wettable powders that can be diluted with water to give suspensions of the desired concentration.
- Powders for dry seed treatment a) b) c) active ingredients 25 % 50 % 75 % light mineral oil 5 % 5 % 5 % highly dispersed silicic acid 5 % 5 % Kaolin 65 % 40 % -
- the combination is thoroughly mixed with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording powders that can be used directly for seed treatment.
- Emulsifiable concentrate active ingredients 10 % octylphenol polyethylene glycol ether (4-5 mol of ethylene oxide) 3 % calcium dodecylbenzene sulfonate 3 % castor oil polyglycol ether (35 mol of ethylene oxide) 4 %
- Emulsions of any required dilution which can be used in plant protection, can be obtained from this concentrate by dilution with water.
- Ready-for-use dusts are obtained by mixing the combination with the carrier and grinding the mixture in a suitable mill. Such dusts can also be used for dry dressings for seed.
- the combination is mixed and ground with the adjuvants, and the mixture is moistened with water.
- the mixture is extruded and then dried in a stream of air.
- the finely ground combination is uniformly applied, in a mixer, to the kaolin moistened with polyethylene glycol.
- Non-dusty coated granules are obtained in this manner.
- the finely ground combination is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- a suspension concentrate from which suspensions of any desired dilution can be obtained by dilution with water.
- living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- Flowable concentrate for seed treatment active ingredients 40 % propylene glycol 5 % copolymer butanol PO/EO 2 %
- Silicone oil (in the form of a 75 % emulsion in water) 0.2 %
- the finely ground combination is intimately mixed with the adjuvants, giving a flowable concentrate from which solutions of any desired dilution can be obtained by dilution with water, that can be used directly for seed treatment.
- solutions of any desired dilution can be obtained by dilution with water, that can be used directly for seed treatment.
- dilutions living plants as well as plant propagation material can be treated and protected against infestation by microorganisms, by spraying, pouring or immersion.
- 28 parts of the combination are mixed with 2 parts of an aromatic solvent and 7 parts of toluene diisocyanate/polymethylene-polyphenylisocyanate-mixture (8:1).
- This mixture is emulsified in a mixture of 1.2 parts of polyvinylalcohol, 0.05 parts of a defoamer and 51 .6 parts of water until the desired particle size is achieved.
- a mixture of 2.8 parts 1 ,6-diaminohexane in 5.3 parts of water is added.
- the mixture is agitated until the polymerization reaction is completed.
- the obtained capsule suspension is stabilized by adding 0.25 parts of a thickener and 3 parts of a dispersing agent.
- the capsule suspension formulation contains 28% of the active ingredients.
- the medium capsule diameter is 8-15 microns.
- the resulting formulation is applied to seeds as an aqueous suspension in an apparatus suitable for that purpose.
- Formulation types include an emulsion concentrate (EC), a suspension concentrate (SC), a suspo-emulsion (SE), a capsule suspension (CS), a water dispersible granule (WG), an emulsifiable granule (EG), an emulsion, water in oil (EO), an emulsion, oil in water (EW), a micro-emulsion (ME), an oil dispersion (OD), an oil miscible flowable (OF), an oil miscible liquid (OL), a soluble concentrate (SL), an ultra-low volume suspension (SU), an ultra-low volume liquid (UL), a technical concentrate (TK), a dispersible concentrate (DC), a wettable powder (WP), a soluble granule (SG) or any technically feasible formulation in combination with agriculturally acceptable adjuvants.
- EC emulsion concentrate
- SC suspension concentrate
- SE suspo-emulsion
- CS capsule suspension
- WG water dispersible granule
- EG
- DABCO 1 ,4-diazabicyclo[2.2.2]octane also known as triethylenediamine or TEDA
- T3P propanephosphonic acid anhydride also called 2,4,6-tripropyl-1 , 3, 5, 2,4,6- trioxatriphosphorinane-2,4,6-trioxide
- the compounds of formula (I) according to the invention may be prepared using the synthetic techniques described both above and below.
- Mp means melting point in °C. Free radicals represent methyl groups.
- 1 H NMR and 19 F NMR measurements were recorded on a Bruker 400MHz spectrometer (or 600MHz as indicated), chemical shifts are given in ppm relevant to a TMS ( 1 H) and CFCI3 ( 19 F) standard. Spectra measured in deuterated solvents as indicated. Either one of the LC-MS methods below was used to characterize the compounds. The characteristic LCMS values obtained for each compound were the retention time (“Rt”, recorded in minutes) and the measured molecular ion (M+H) + or (M-H)’.
- Example P1 Synthesis of [5-(3,5-difluoro-2-pyridyl)isoxazol-3-yl1-[rac-(1 S,4S)-4-(1 ,5-dimethylpyrazol-4-yl)-1- methyl-3,4-dihydro-1 H-isoquinolin-2-yl1methanone (Compound P-6, Table P)
- Step 1 Preparation of 3,5-difluoro-N-methoxy-N-methyl-pyridine-2-carboxamide
- Methyl magnesium bromide solution (7.9 mL, 23.59 mmol) was added dropwise to a cooled pale brown solution (0-5°C) of 3,5-difluoro-N-methoxy-N-methyl-pyridine-2-carboxamide (2.38 g, 11 .79 mmol) in THF (35 mL) under argon.
- the resulting suspension was allowed to reach rt and stirred for 30 minutes under an argon atmosphere.
- the reaction mixture was slowly quenched with saturated aqueous ammonium chloride and extracted with EtOAc. The combined organic phases were dried over anhydrous Na2SO4, filtered, and concentrated in vacuo to afford the title compound.
- Step 3 Preparation of ethyl 4-(3,5-difluoro-2-pyridyl)-2,4-dioxo-butanoate
- Step 4 Preparation of ethyl 5-(3,5-difluoro-2-pyridyl)isoxazole-3-carboxylate
- Step 6 Preparation of (1 ,5-dimethylpyrazol-4-yD-phenyl-methanol
- Step 7 Preparation of 2-(1 ,5-dimethylpyrazol-4-yl)-2-phenyl-acetonitrile
- Step 8 Preparation of 2-(1 ,5-dimethylpyrazol-4-yl)-2-phenyl-ethanamine (Option A)
- reaction mixture was cooled to 0°C before adding hydrochloric acid (23 g, 34.71 mL, 208.2 mmol) dropwise (strong gas evolution) and the reaction mixture was stirred at 65°C for 1 hour and allowed to stand overnight at rt.
- the mixture was diluted with water and treated with NaOH 6M (to pH 12).
- the mixture extracted twice with EtOAc and the combined organic layers were washed with brine, dried over Na2SC>4, and concentrated in vacuo to give the title compound as a yellow oil which was used without further purification in the next step.
- Step 9 Preparation of methyl N-[2-(1 ,5-dimethylpyrazol-4-yl)-2-phenyl-ethyl1carbamate
- Step 11 Preparation of rac-(1 S,4S)-4-(1 ,5-dimethylpyrazol-4-yl)-1-methyl-1 ,2,3,4-tetrahydroisoquinoline
- Step 12 Synthesis of [5-(3,5-difluoro-2-pyridyl)isoxazol-3-yl]-[rac-(1 S,4S)-4-(1 ,5-dimethylpyrazol-4-yl)-1- methyl-3,4-dihydro-1 H-isoquinolin-2-yl]methanone (Compound P-6, Table P)
- the reaction mixture was stirred for 1 hr at rt under argon diluted with EtOAc and quenched with aqueous NaHCOs.
- the organic phase was separated, and the aqueous phase back extracted with EtOAc.
- the combined organic phases were dried over anhydrous Na2SO4, filtered, and concentrated in vacuo.
- the crude product was purified by flash chromatography eluting with EtOAc/cyclohexane to afford the title compound.
- Example P2 Preparation of [5-(3,5-difluoro-2-pyridyl)-1 ,3,4-thiadiazol-2-yl]-[4-(1 ,5-dimethylpyrazol-4-yl)-3,4- dihydro-1 H-isoquinolin-2-yl1methanone (Compound P-7, Table P)
- Step 2 Preparation of tert-butyl N-[(3,5-difluoropyridine-2-carbonyl)amino]carbamate
- Step 4 Preparation of methyl 2-[2-(3,5-difluoropyridine-2-carbonyl)hydrazino1-2-oxo-acetate
- Step 5 Preparation of methyl 5-(3,5-difluoro-2-pyridyl)-1 ,3,4-thiadiazole-2-carboxylate
- reaction mixture was heated at 100°C and stirred for 1 hour under microwave irradiation. After cooling down to rt, the reaction mixture was partitioned between water and EtOAc and the organic layer separated, dried over Na2SO4, filtered and concentrated in vacuo. Purification of the crude material by flash chromatography over silica gel (eluting with EtOAc acetate/30% methanol) afforded the title compound.
- hydrochloride salt of the title product (4-(1 ,5-dimethylpyrazol-4-yl)-1 ,2,3,4-tetrahydroisoquinoline; hydrochloride) could be obtained by treating the yellow oil with 2 M HCI in diethyl ether and then concentration in vacuo.
- Step 8 Preparation of [5-(3,5-difluoro-2-pyridyl)-1 ,3,4-thiadiazol-2-yl1-[4-(1 ,5-dimethylpyrazol-4-yl)-3,4- dihydro-1 H-isoquinolin-2-yl]methanone (Compound P-7, Table P)
- Example P3 Preparation of [5-(2,6-difluoro-3-pyridyl)-1 ,3,4-thiadiazol-2-yl1-[4-(1 ,5-dimethylpyrazol-4-yl)-3,4- dihydro-1 H-isoquinolin-2-yl1methanone (Compound P-8, Table P)
- the desired compound was prepared from 2,6-difluoropyridine-3-carboxylic acid and oxalyl chloride, analogously to the example described in example 2, step 1 , to afford 2,6-difluoropyridine-3-carbonyl chloride.
- Step 2 Preparation of tert-butyl N-[(2,6-difluoropyridine-3-carbonyl)amino]carbamate
- Step 4 Preparation of methyl 2-[2-(2,6-difluoropyridine-3-carbonyl)hydrazino]-2-oxo-acetate
- Step 5 Preparation of methyl 5-(2,6-difluoro-3-pyridyl)-1 ,3,4-thiadiazole-2-carboxylate
- Step 6 Preparation of [5-(2,6-difluoro-3-pyridyl)-1 ,3,4-thiadiazol-2-yl1-[4-(1 ,5-dimethylpyrazol-4-yl)-3,4- dihydro-1 H-isoquinolin-2-yl1methanone (Compound P-8, Table P)
- Example P4 Preparation of [5-(2,6-difluoro-3-pyridyl)-1 ,3,4-thiadiazol-2-yl]-[rac-(1 S,4S)-4-(1 ,5- dimethylpyrazol-4-yl)-1-methyl-3,4-dihydro-1 H-isoquinolin-2-yl]methanone (Compound P-9, Table P)
- reaction mixture was heated and stirred at 90°C. After ⁇ 90', the mixture was allowed to cool to rt and was then added to 10ml NaOH 2M aqueous solution. After dilution with water (30ml) the mixture was extracted with EtOAc (2x). The combined organic layers were washed with brine, dried over Na 2 SO 4 , filtered, and concentrated in vacuo to afford crude product. The crude was purified by chromatography over silica gel to afford the title compound.
- Example B1 Altemaria solani / tomato / leaf disc (early blight)
- Tomato leaf disks cv. Baby are placed on agar in multiwell plates (24-well format) and sprayed with the formulated test compound diluted in water.
- the leaf disks are inoculated with a spore suspension of the fungus 2 days after application.
- the inoculated leaf disks are incubated at 23°C / 21 °C (day/night) and 80% rh under a light regime of 12 h light / 12 h darkness in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears on untreated check disk leaf disks (5 to 7 days after application).
- Example B2 Botryotinia fuckeliana (Botrytis cinerea) / liquid culture (Gray mould)
- Conidia of the fungus from cryogenic storage are directly mixed into nutrient broth (Vogels broth). After placing a (DMSO) solution of test compound into a microtiter plate (96-well format), the nutrient broth containing the fungal spores is added. The test plates are incubated at 24°C and the inhibition of growth is determined photometrically 3 to 4 days after application.
- DMSO fetal sulfate
- Example B3 Glomerella laqenarium (Colletotrichum laqenarium) / liquid culture (Anthracnose)
- Conidia of the fungus from cryogenic storage are directly mixed into nutrient broth (PDB potato dextrose broth). After placing a (DMSO) solution of test compound into a microtiter plate (96-well format), the nutrient broth containing the fungal spores is added. The test plates are incubated at 24°C and the inhibition of growth is measured photometrically 3 to 4 days after application.
- nutrient broth PDB potato dextrose broth
- Example B4 Blumeria graminis f. sp. tritici (Erysiphe graminis f. sp. tritici) / wheat / leaf disc preventative (Powdery mildew on wheat)
- Wheat leaf segments cv. Kanzler are placed on agar in a multiwell plate (24-well format) and sprayed with the formulated test compound diluted in water.
- the leaf disks are inoculated by shaking powdery mildew infected plants above the test plates 1 day after application.
- the inoculated leaf disks are incubated at 20°C and 60% rh under a light regime of 24 h darkness followed by 12 h light / 12 h darkness in a climate chamber and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears on untreated check leaf segments (6 to 8 days after application).
- Example B5 Fusarium culmorum / liquid culture (Head blight)
- Conidia of the fungus from cryogenic storage are directly mixed into nutrient broth (PDB potato dextrose broth). After placing a (DMSO) solution of test compound into a microtiter plate (96-well format), the nutrient broth containing the fungal spores is added. The test plates are incubated at 24°C and the inhibition of growth is determined photometrically 3 to 4 days after application.
- nutrient broth PDB potato dextrose broth
- Example B6 Fusarium cu/morum / wheat / spikelet preventative (Head blight)
- Wheat spikelets cv. Monsun are placed on agar in multiwell plates (24-well format) and sprayed with the formulated test compound diluted in water.
- the spikelets are inoculated with a spore suspension of the fungus 1 day after application.
- the inoculated spikelets are incubated at 20°C and 60% rh under a light regime of 72 h semi darkness followed by 12 h light / 12 h darkness in a climate chamber and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears on untreated check spikelets (6 to 8 days after application).
- Example B7 Gibberella zeae (Fusarium graminearum) / wheat / spikelet preventative (Head blight) Wheat spikelets cv. Monsun are placed on agar in multiwell plates (24-well format) and sprayed with the formulated test compound diluted in water. One day after application, the spikelets are inoculated with a spore suspension of the fungus.
- the inoculated test leaf disks are incubated at 20°C and 60% rh under a light regime of 72 h semi darkness followed by 12 h light / 12 h darkness in a climate chamber, the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears on untreated check spikelets (6 to 8 days after application).
- Example B8 Phaeosphaeria nodorum (Septoria nodorum) / wheat / leaf disc preventative (Glume blotch)
- Wheat leaf segments cv. Kanzler are placed on agar in a multiwell plate (24-well format) and sprayed with the formulated test compound diluted in water.
- the leaf disks are inoculated with a spore suspension of the fungus 2 days after application.
- the inoculated test leaf disks are incubated at 20°C and 75% rh under a light regime of 12 h light / 12 h darkness in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf disks (5 to 7 days after application).
- Example B9 Monoqraphella nivalis (Microdochium nivale) / liquid culture (foot rot cereals)
- Conidia of the fungus from cryogenic storage are directly mixed into nutrient broth (PDB potato dextrose broth). After placing a (DMSO) solution of test compound into a microtiter plate (96-well format), the nutrient broth containing the fungal spores is added. The test plates are incubated at 24°C and the inhibition of growth is determined photometrically 4 to 5 days after application.
- nutrient broth PDB potato dextrose broth
- Example B10 Mycosphaerella arachidis (Cercospora arachidicola) / liquid culture (early leaf spot)
- Conidia of the fungus from cryogenic storage are directly mixed into nutrient broth (PDB potato dextrose broth). After placing a (DMSO) solution of test compound into a microtiter plate (96-well format), the nutrient broth containing the fungal spores is added. The test plates are incubated at 24°C and the inhibition of growth is determined photometrically 4 to 5 days after application. The following compounds gave at least 80% control of Mycosphaerella arachidis at 20 ppm when compared to untreated control under the same conditions, which showed extensive disease development:
- Example B11 Pyrenophora teres / barley / leaf disc preventative (Net blotch)
- Barley leaf segments cv. Hasso are placed on agar in a multiwell plate (24-well format) and sprayed with the formulated test compound diluted in water.
- the leaf segments are inoculated with a spore suspension of the fungus 2 days after application.
- the inoculated leaf segments are incubated at 20°C and 65% rh under a light regime of 12 h light / 12 h darkness in a climate cabinet and the activity of a compound is assessed as disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (5 to 7 days after application).
- Example B12 Sclerotinia sclerotiorum / liquid culture (cottony rot)
- Mycelia fragments of a newly grown liquid culture of the fungus are directly mixed into nutrient broth (PDB potato dextrose broth). After placing a (DMSO) solution of test compound into a microtiter plate (96-well format) the nutrient broth containing the fungal material is added. The test plates are incubated at 24°C and the inhibition of growth is determined photometrically 3 to 4 days after application.
- nutrient broth PDB potato dextrose broth
- Example B13 Mycosphaerella qraminicola (Septoria tritici) / liquid culture (Septoria blotch)
- Conidia of the fungus from cryogenic storage are directly mixed into nutrient broth (PDB potato dextrose broth). After placing a (DMSO) solution of test compound into a microtiter plate (96-well format), the nutrient broth containing the fungal spores is added. The test plates are incubated at 24°C and the inhibition of growth is determined photometrically 4 to 5 days after application.
- nutrient broth PDB potato dextrose broth
- Example B14 Puccinia recondita f. sp. tritici / wheat / leaf disc curative (Brown rust) Wheat leaf segments cv. Kanzler are placed on agar in multiwell plates (24-well format). The leaf segments are inoculated with a spore suspension of the fungus. Plates are stored in darkness at 19 °C and 75% rh. The formulated test compound diluted in water is applied 1 day after inoculation.
- the leaf segments are incubated at 19°C and 75% rh under a light regime of 12 h light / 12 h darkness in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (6 to 8 days after application).
- Example B15 Puccinia recondita f. sp. tritici / wheat / leaf disc preventative (Brown rust)
- Wheat leaf segments cv. Kanzler are placed on agar in multiwell plates (24-well format) and sprayed with the formulated test compound diluted in water.
- the leaf disks are inoculated with a spore suspension of the fungus 1 day after application.
- the inoculated leaf segments are incubated at 19°C and 75% rh under a light regime of 12 h light / 12 h darkness in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (7 to 9 days after application).
- Example B16 Maqnaporthe qrisea (Pyricularia oryzae) / liquid culture (Rice Blast)
- Conidia of the fungus from cryogenic storage are directly mixed into nutrient broth (PDB potato dextrose broth). After placing a (DMSO) solution of test compound into a microtiter plate (96-well format), the nutrient broth containing the fungal spores is added. The test plates are incubated at 24°C and the inhibition of growth is determined photometrically 3 to 4 days after application.
- nutrient broth PDB potato dextrose broth
- Example B17 Maqnaporthe qrisea (Pyricularia oryzae) / rice / leaf disc preventative (Rice Blast)
- Rice leaf segments cv. Ballila are placed on agar in a multiwell plate (24-well format) and sprayed with the formulated test compound diluted in water.
- the leaf segments are inoculated with a spore suspension of the fungus 2 days after application.
- the inoculated leaf segments are incubated at 22°C and 80% rh under a light regime of 24 h darkness followed by 12 h light / 12 h darkness in a climate cabinet and the activity of a compound is assessed as percent disease control compared to untreated when an appropriate level of disease damage appears in untreated check leaf segments (5 to 7 days after application).
- the following compounds gave at least 80% control of Magnaporthe grisea at 200 ppm when compared to untreated control under the same conditions, which showed extensive disease development:
- Example B18 Thanatephorus cucumeris (Rhizoctonia solani) / liquid culture (foot rot, damping-off)
- Mycelia fragments of a newly grown liquid culture of the fungus are directly mixed into nutrient broth (PDB potato dextrose broth).
- nutrient broth PDB potato dextrose broth
- DMSO DMSO
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Plant Pathology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Engineering & Computer Science (AREA)
- Pest Control & Pesticides (AREA)
- Wood Science & Technology (AREA)
- Agronomy & Crop Science (AREA)
- Health & Medical Sciences (AREA)
- Dentistry (AREA)
- General Health & Medical Sciences (AREA)
- Mycology (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Microbiology (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
Claims
Priority Applications (9)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CR20250206A CR20250206A (en) | 2022-11-16 | 2023-11-15 | TETRAHYDROISOQUINOLINE MICROBICIDE DERIVATIVES |
| EP23805614.7A EP4619396A1 (en) | 2022-11-16 | 2023-11-15 | Microbiocidal tetrahydroisoquinoline derivatives |
| AU2023381091A AU2023381091A1 (en) | 2022-11-16 | 2023-11-15 | Microbiocidal tetrahydroisoquinoline derivatives |
| JP2025528472A JP2025537818A (en) | 2022-11-16 | 2023-11-15 | Microbicide tetrahydroisoquinoline derivatives |
| CN202380079234.7A CN120225510A (en) | 2022-11-16 | 2023-11-15 | Microbicidal tetrahydroisoquinoline derivatives |
| KR1020257019054A KR20250095743A (en) | 2022-11-16 | 2023-11-15 | Microbial tetrahydroisoquinoline derivatives |
| IL320722A IL320722A (en) | 2022-11-16 | 2025-05-06 | Microbiocidal tetrahydroisoquinoline derivatives |
| CONC2025/0006132A CO2025006132A2 (en) | 2022-11-16 | 2025-05-13 | Microbicidal tetrahydroisoquinoline derivatives |
| MX2025005559A MX2025005559A (en) | 2022-11-16 | 2025-05-13 | Microbiocidal tetrahydroisoquinoline derivatives |
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN202211065741 | 2022-11-16 | ||
| IN202211065741 | 2022-11-16 | ||
| EP23151012.4 | 2023-01-10 | ||
| EP23151012 | 2023-01-10 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024105104A1 true WO2024105104A1 (en) | 2024-05-23 |
Family
ID=88793315
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2023/081908 Ceased WO2024105104A1 (en) | 2022-11-16 | 2023-11-15 | Microbiocidal tetrahydroisoquinoline derivatives |
Country Status (14)
| Country | Link |
|---|---|
| EP (1) | EP4619396A1 (en) |
| JP (1) | JP2025537818A (en) |
| KR (1) | KR20250095743A (en) |
| CN (1) | CN120225510A (en) |
| AR (1) | AR131059A1 (en) |
| AU (1) | AU2023381091A1 (en) |
| CL (1) | CL2025001342A1 (en) |
| CO (1) | CO2025006132A2 (en) |
| CR (1) | CR20250206A (en) |
| IL (1) | IL320722A (en) |
| MX (1) | MX2025005559A (en) |
| TW (1) | TW202434579A (en) |
| UY (1) | UY40520A (en) |
| WO (1) | WO2024105104A1 (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025146624A1 (en) * | 2024-01-03 | 2025-07-10 | Pi Industries Ltd. | Bicyclic heterocyclic amides for combating phytopathogenic fungi |
Citations (68)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4639771A (en) | 1984-10-31 | 1987-01-27 | Kabushiki Kaisha Toshiba | Image processing system |
| EP0357460A2 (en) | 1988-09-02 | 1990-03-07 | Sankyo Company Limited | 13-Substituted milbemycin derivatives, their preparation and use |
| EP0367474A1 (en) | 1988-11-01 | 1990-05-09 | Mycogen Corporation | Novel bacillus thuringiensis isolate denoted b.t. ps81gg, active against lepidopteran pests, and a gene encoding a lepidopteran-active toxin |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0382173A2 (en) | 1989-02-07 | 1990-08-16 | Meiji Seika Kaisha Ltd. | PF 1022 substance, method of producing same and anthelmintic composition containing same |
| WO1990013651A1 (en) | 1989-05-09 | 1990-11-15 | Imperial Chemical Industries Plc | Bacterial genes |
| EP0401979A2 (en) | 1989-05-18 | 1990-12-12 | Mycogen Corporation | Novel bacillus thuringiensis isolates active against lepidopteran pests, and genes encoding novel lepidopteran-active toxins |
| US5015630A (en) | 1989-01-19 | 1991-05-14 | Merck & Co., Inc. | 5-oxime avermectin derivatives |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| EP0444964A1 (en) | 1990-03-01 | 1991-09-04 | Sankyo Company Limited | Milbemycin ether derivatives, their preparation and their anthelmintic uses |
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| EP0503538A1 (en) | 1991-03-08 | 1992-09-16 | Meiji Seika Kaisha Ltd. | Medicinal composition containing an anthelmintic cyclic depsipeptide |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1993019053A1 (en) | 1992-03-17 | 1993-09-30 | Fujisawa Pharmaceutical Co., Ltd. | Depsipeptide derivative, production thereof and use thereof |
| WO1993025543A2 (en) | 1992-06-11 | 1993-12-23 | Bayer Aktiengesellschaft | Enniatines and enniatine derivates used to control endoparasites |
| EP0594291A1 (en) | 1992-09-01 | 1994-04-27 | Sankyo Company Limited | Novel processes for the production of 13-ether derivatives of milbemycins, and novel intermediates therefor |
| WO1994015944A1 (en) | 1993-01-18 | 1994-07-21 | Pfizer Limited | New antiparasitic agents related to the milbemycins and avermectins |
| WO1994019334A1 (en) | 1993-02-19 | 1994-09-01 | Meiji Seika Kaisha, Ltd. | Pf1022 derivative, cyclic depsipeptide |
| EP0626375A1 (en) | 1993-05-26 | 1994-11-30 | Bayer Ag | Octacyclodepsipeptides having endoparasiticidal activity |
| WO1995019363A1 (en) | 1994-01-14 | 1995-07-20 | Pfizer Inc. | Antiparasitic pyrrolobenzoxazine compounds |
| WO1995022552A1 (en) | 1994-02-16 | 1995-08-24 | Pfizer Limited | Antiparasitic agents |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| US5478855A (en) | 1992-04-28 | 1995-12-26 | Yashima Chemical Industry Co., Ltd. | 2-(2,6-difluorophenyl)-4-(2-ethoxy-4-tert-butylphenyl)-2-oxazoline |
| WO1996011945A2 (en) | 1994-10-18 | 1996-04-25 | Bayer Aktiengesellschaft | Cyclic depsipeptide sulfonylation, sulfenylation and phosphorylation process |
| WO1996015121A1 (en) | 1994-11-10 | 1996-05-23 | Bayer Aktiengesellschaft | Use of dioxomorpholines to combat endoparasites, novel dioxomorpholines and process for their production |
| DE19520936A1 (en) | 1995-06-08 | 1996-12-12 | Bayer Ag | Ectoparasiticides means |
| WO1997033890A1 (en) | 1996-03-11 | 1997-09-18 | Novartis Ag | Pyrimidin-4-one derivatives as pesticide |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| WO2004072086A2 (en) | 2003-02-14 | 2004-08-26 | Pfizer Limited | Antiparasitic terpene alkaloids |
| US6919298B2 (en) | 2002-04-04 | 2005-07-19 | Valent Biosciences Corporation | Enhanced herbicide composition |
| WO2006044617A1 (en) | 2004-10-15 | 2006-04-27 | The Scripps Research Institute | Oxadiazole ketone inhibitors of fatty acid amide hydrolase |
| WO2009149858A1 (en) | 2008-06-13 | 2009-12-17 | Bayer Cropscience Ag | Novel heteroaromatic amides and thioamides as pesticides |
| WO2010006713A2 (en) | 2008-07-17 | 2010-01-21 | Bayer Cropscience Ag | Heterocyclic compounds used as pesticides |
| WO2011138281A2 (en) | 2010-05-06 | 2011-11-10 | Bayer Cropscience Ag | Process for the preparation of dithiine tetracarboxydiimides |
| WO2014006945A1 (en) | 2012-07-04 | 2014-01-09 | アグロカネショウ株式会社 | 2-aminonicotinic acid ester derivative and bactericide containing same as active ingredient |
| CN103772278A (en) | 2014-01-02 | 2014-05-07 | 湖南华腾制药有限公司 | Important tetrahydroisoquinoline derivative midbody and synthesis method thereof |
| WO2014095675A1 (en) | 2012-12-19 | 2014-06-26 | Bayer Cropscience Ag | Difluoromethyl-nicotinic-indanyl carboxamides as fungicides |
| WO2015155075A1 (en) | 2014-04-11 | 2015-10-15 | Syngenta Participations Ag | Fungicidal n'-[2-methyl-6-[2-alkoxy-ethoxy]-3-pyridyl]-n-alkyl-formamidine derivatives for use in agriculture |
| WO2016156085A1 (en) | 2015-03-27 | 2016-10-06 | Syngenta Participations Ag | Microbiocidal heterobicyclic derivatives |
| WO2016156290A1 (en) | 2015-04-02 | 2016-10-06 | Bayer Cropscience Aktiengesellschaft | Novel 5-substituted imidazole derivatives |
| WO2016202742A1 (en) | 2015-06-15 | 2016-12-22 | Bayer Cropscience Aktiengesellschaft | Halogen-substituted phenoxyphenylamidines and the use thereof as fungicides |
| WO2017025510A1 (en) | 2015-08-12 | 2017-02-16 | Syngenta Participations Ag | Microbiocidal heterobicyclic derivatives |
| WO2017029179A1 (en) | 2015-08-14 | 2017-02-23 | Bayer Cropscience Aktiengesellschaft | Triazole derivatives, intermediates thereof and their use as fungicides |
| CN106467537A (en) * | 2015-08-14 | 2017-03-01 | 沈阳中化农药化工研发有限公司 | A kind of substituted amides compound and purposes |
| WO2017055473A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017055469A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017093348A1 (en) | 2015-12-02 | 2017-06-08 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017118689A1 (en) | 2016-01-08 | 2017-07-13 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017153380A1 (en) | 2016-03-10 | 2017-09-14 | Syngenta Participations Ag | Microbiocidal quinoline (thio)carboxamide derivatives |
| WO2017220485A1 (en) | 2016-06-21 | 2017-12-28 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018019929A1 (en) | 2016-07-28 | 2018-02-01 | Idorsia Pharmaceuticals Ltd | Piperidine cxcr7 receptor modulators |
| WO2018065414A1 (en) | 2016-10-06 | 2018-04-12 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018153707A1 (en) | 2017-02-22 | 2018-08-30 | Basf Se | Crystalline forms of a strobilurin type compound for combating phytopathogenic fungi |
| WO2018158365A1 (en) | 2017-03-03 | 2018-09-07 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018202428A1 (en) | 2017-05-02 | 2018-11-08 | Basf Se | Fungicidal mixture comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| WO2018228896A1 (en) | 2017-06-14 | 2018-12-20 | Syngenta Participations Ag | Fungicidal compositions |
| WO2019110427A1 (en) | 2017-12-04 | 2019-06-13 | Syngenta Participations Ag | Microbiocidal phenylamidine derivatives |
| WO2019195810A2 (en) | 2018-04-06 | 2019-10-10 | Praxis Biotech LLC | Atf6 inhibitors and uses thereof |
| WO2020056090A1 (en) | 2018-09-14 | 2020-03-19 | Fmc Corporation | Fungicidal halomethyl ketones and hydrates |
| WO2020079111A1 (en) | 2018-10-18 | 2020-04-23 | Syngenta Crop Protection Ag | Microbiocidal compounds |
| CN111072582A (en) | 2018-10-18 | 2020-04-28 | 中国药科大学 | A kind of N-hydroxy aromatic heterocycle-2-carboxamide compound and its preparation method and use |
| WO2020097012A1 (en) | 2018-11-06 | 2020-05-14 | Fmc Corporation | Substituted tolyl as fungicides |
| WO2020109391A1 (en) | 2018-11-28 | 2020-06-04 | Bayer Aktiengesellschaft | Pyridazine (thio)amides as fungicidal compounds |
| WO2020193387A1 (en) | 2019-03-22 | 2020-10-01 | Syngenta Crop Protection Ag | Fungicidal compounds |
| CN114933573A (en) | 2022-05-06 | 2022-08-23 | 贵州大学 | 3-substituted-1, 2, 4-oxadiazole-5-carboxylic acid derivative, and preparation method and application thereof |
| WO2022253645A1 (en) * | 2021-06-01 | 2022-12-08 | Syngenta Crop Protection Ag | Microbiocidal tetrahydroisoquinoline derivatives |
-
2023
- 2023-11-13 TW TW112143691A patent/TW202434579A/en unknown
- 2023-11-14 AR ARP230103062A patent/AR131059A1/en unknown
- 2023-11-14 UY UY0001040520A patent/UY40520A/en unknown
- 2023-11-15 CN CN202380079234.7A patent/CN120225510A/en active Pending
- 2023-11-15 WO PCT/EP2023/081908 patent/WO2024105104A1/en not_active Ceased
- 2023-11-15 KR KR1020257019054A patent/KR20250095743A/en active Pending
- 2023-11-15 EP EP23805614.7A patent/EP4619396A1/en active Pending
- 2023-11-15 JP JP2025528472A patent/JP2025537818A/en active Pending
- 2023-11-15 AU AU2023381091A patent/AU2023381091A1/en active Pending
- 2023-11-15 CR CR20250206A patent/CR20250206A/en unknown
-
2025
- 2025-05-06 IL IL320722A patent/IL320722A/en unknown
- 2025-05-07 CL CL2025001342A patent/CL2025001342A1/en unknown
- 2025-05-13 CO CONC2025/0006132A patent/CO2025006132A2/en unknown
- 2025-05-13 MX MX2025005559A patent/MX2025005559A/en unknown
Patent Citations (68)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4639771A (en) | 1984-10-31 | 1987-01-27 | Kabushiki Kaisha Toshiba | Image processing system |
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| EP0357460A2 (en) | 1988-09-02 | 1990-03-07 | Sankyo Company Limited | 13-Substituted milbemycin derivatives, their preparation and use |
| EP0367474A1 (en) | 1988-11-01 | 1990-05-09 | Mycogen Corporation | Novel bacillus thuringiensis isolate denoted b.t. ps81gg, active against lepidopteran pests, and a gene encoding a lepidopteran-active toxin |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| US5015630A (en) | 1989-01-19 | 1991-05-14 | Merck & Co., Inc. | 5-oxime avermectin derivatives |
| EP0382173A2 (en) | 1989-02-07 | 1990-08-16 | Meiji Seika Kaisha Ltd. | PF 1022 substance, method of producing same and anthelmintic composition containing same |
| WO1990013651A1 (en) | 1989-05-09 | 1990-11-15 | Imperial Chemical Industries Plc | Bacterial genes |
| EP0401979A2 (en) | 1989-05-18 | 1990-12-12 | Mycogen Corporation | Novel bacillus thuringiensis isolates active against lepidopteran pests, and genes encoding novel lepidopteran-active toxins |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| EP0444964A1 (en) | 1990-03-01 | 1991-09-04 | Sankyo Company Limited | Milbemycin ether derivatives, their preparation and their anthelmintic uses |
| EP0503538A1 (en) | 1991-03-08 | 1992-09-16 | Meiji Seika Kaisha Ltd. | Medicinal composition containing an anthelmintic cyclic depsipeptide |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1993019053A1 (en) | 1992-03-17 | 1993-09-30 | Fujisawa Pharmaceutical Co., Ltd. | Depsipeptide derivative, production thereof and use thereof |
| US5478855A (en) | 1992-04-28 | 1995-12-26 | Yashima Chemical Industry Co., Ltd. | 2-(2,6-difluorophenyl)-4-(2-ethoxy-4-tert-butylphenyl)-2-oxazoline |
| WO1993025543A2 (en) | 1992-06-11 | 1993-12-23 | Bayer Aktiengesellschaft | Enniatines and enniatine derivates used to control endoparasites |
| EP0594291A1 (en) | 1992-09-01 | 1994-04-27 | Sankyo Company Limited | Novel processes for the production of 13-ether derivatives of milbemycins, and novel intermediates therefor |
| WO1994015944A1 (en) | 1993-01-18 | 1994-07-21 | Pfizer Limited | New antiparasitic agents related to the milbemycins and avermectins |
| WO1994019334A1 (en) | 1993-02-19 | 1994-09-01 | Meiji Seika Kaisha, Ltd. | Pf1022 derivative, cyclic depsipeptide |
| EP0626375A1 (en) | 1993-05-26 | 1994-11-30 | Bayer Ag | Octacyclodepsipeptides having endoparasiticidal activity |
| WO1995019363A1 (en) | 1994-01-14 | 1995-07-20 | Pfizer Inc. | Antiparasitic pyrrolobenzoxazine compounds |
| WO1995022552A1 (en) | 1994-02-16 | 1995-08-24 | Pfizer Limited | Antiparasitic agents |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| WO1996011945A2 (en) | 1994-10-18 | 1996-04-25 | Bayer Aktiengesellschaft | Cyclic depsipeptide sulfonylation, sulfenylation and phosphorylation process |
| WO1996015121A1 (en) | 1994-11-10 | 1996-05-23 | Bayer Aktiengesellschaft | Use of dioxomorpholines to combat endoparasites, novel dioxomorpholines and process for their production |
| DE19520936A1 (en) | 1995-06-08 | 1996-12-12 | Bayer Ag | Ectoparasiticides means |
| WO1997033890A1 (en) | 1996-03-11 | 1997-09-18 | Novartis Ag | Pyrimidin-4-one derivatives as pesticide |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| US6919298B2 (en) | 2002-04-04 | 2005-07-19 | Valent Biosciences Corporation | Enhanced herbicide composition |
| WO2004072086A2 (en) | 2003-02-14 | 2004-08-26 | Pfizer Limited | Antiparasitic terpene alkaloids |
| WO2006044617A1 (en) | 2004-10-15 | 2006-04-27 | The Scripps Research Institute | Oxadiazole ketone inhibitors of fatty acid amide hydrolase |
| WO2009149858A1 (en) | 2008-06-13 | 2009-12-17 | Bayer Cropscience Ag | Novel heteroaromatic amides and thioamides as pesticides |
| WO2010006713A2 (en) | 2008-07-17 | 2010-01-21 | Bayer Cropscience Ag | Heterocyclic compounds used as pesticides |
| WO2011138281A2 (en) | 2010-05-06 | 2011-11-10 | Bayer Cropscience Ag | Process for the preparation of dithiine tetracarboxydiimides |
| WO2014006945A1 (en) | 2012-07-04 | 2014-01-09 | アグロカネショウ株式会社 | 2-aminonicotinic acid ester derivative and bactericide containing same as active ingredient |
| WO2014095675A1 (en) | 2012-12-19 | 2014-06-26 | Bayer Cropscience Ag | Difluoromethyl-nicotinic-indanyl carboxamides as fungicides |
| CN103772278A (en) | 2014-01-02 | 2014-05-07 | 湖南华腾制药有限公司 | Important tetrahydroisoquinoline derivative midbody and synthesis method thereof |
| WO2015155075A1 (en) | 2014-04-11 | 2015-10-15 | Syngenta Participations Ag | Fungicidal n'-[2-methyl-6-[2-alkoxy-ethoxy]-3-pyridyl]-n-alkyl-formamidine derivatives for use in agriculture |
| WO2016156085A1 (en) | 2015-03-27 | 2016-10-06 | Syngenta Participations Ag | Microbiocidal heterobicyclic derivatives |
| WO2016156290A1 (en) | 2015-04-02 | 2016-10-06 | Bayer Cropscience Aktiengesellschaft | Novel 5-substituted imidazole derivatives |
| WO2016202742A1 (en) | 2015-06-15 | 2016-12-22 | Bayer Cropscience Aktiengesellschaft | Halogen-substituted phenoxyphenylamidines and the use thereof as fungicides |
| WO2017025510A1 (en) | 2015-08-12 | 2017-02-16 | Syngenta Participations Ag | Microbiocidal heterobicyclic derivatives |
| WO2017029179A1 (en) | 2015-08-14 | 2017-02-23 | Bayer Cropscience Aktiengesellschaft | Triazole derivatives, intermediates thereof and their use as fungicides |
| CN106467537A (en) * | 2015-08-14 | 2017-03-01 | 沈阳中化农药化工研发有限公司 | A kind of substituted amides compound and purposes |
| WO2017055473A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017055469A1 (en) | 2015-10-02 | 2017-04-06 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017093348A1 (en) | 2015-12-02 | 2017-06-08 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017118689A1 (en) | 2016-01-08 | 2017-07-13 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2017153380A1 (en) | 2016-03-10 | 2017-09-14 | Syngenta Participations Ag | Microbiocidal quinoline (thio)carboxamide derivatives |
| WO2017220485A1 (en) | 2016-06-21 | 2017-12-28 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018019929A1 (en) | 2016-07-28 | 2018-02-01 | Idorsia Pharmaceuticals Ltd | Piperidine cxcr7 receptor modulators |
| WO2018065414A1 (en) | 2016-10-06 | 2018-04-12 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018153707A1 (en) | 2017-02-22 | 2018-08-30 | Basf Se | Crystalline forms of a strobilurin type compound for combating phytopathogenic fungi |
| WO2018158365A1 (en) | 2017-03-03 | 2018-09-07 | Syngenta Participations Ag | Microbiocidal oxadiazole derivatives |
| WO2018202428A1 (en) | 2017-05-02 | 2018-11-08 | Basf Se | Fungicidal mixture comprising substituted 3-phenyl-5-(trifluoromethyl)-1,2,4-oxadiazoles |
| WO2018228896A1 (en) | 2017-06-14 | 2018-12-20 | Syngenta Participations Ag | Fungicidal compositions |
| WO2019110427A1 (en) | 2017-12-04 | 2019-06-13 | Syngenta Participations Ag | Microbiocidal phenylamidine derivatives |
| WO2019195810A2 (en) | 2018-04-06 | 2019-10-10 | Praxis Biotech LLC | Atf6 inhibitors and uses thereof |
| WO2020056090A1 (en) | 2018-09-14 | 2020-03-19 | Fmc Corporation | Fungicidal halomethyl ketones and hydrates |
| WO2020079111A1 (en) | 2018-10-18 | 2020-04-23 | Syngenta Crop Protection Ag | Microbiocidal compounds |
| CN111072582A (en) | 2018-10-18 | 2020-04-28 | 中国药科大学 | A kind of N-hydroxy aromatic heterocycle-2-carboxamide compound and its preparation method and use |
| WO2020097012A1 (en) | 2018-11-06 | 2020-05-14 | Fmc Corporation | Substituted tolyl as fungicides |
| WO2020109391A1 (en) | 2018-11-28 | 2020-06-04 | Bayer Aktiengesellschaft | Pyridazine (thio)amides as fungicidal compounds |
| WO2020193387A1 (en) | 2019-03-22 | 2020-10-01 | Syngenta Crop Protection Ag | Fungicidal compounds |
| WO2022253645A1 (en) * | 2021-06-01 | 2022-12-08 | Syngenta Crop Protection Ag | Microbiocidal tetrahydroisoquinoline derivatives |
| CN114933573A (en) | 2022-05-06 | 2022-08-23 | 贵州大学 | 3-substituted-1, 2, 4-oxadiazole-5-carboxylic acid derivative, and preparation method and application thereof |
Non-Patent Citations (25)
| Title |
|---|
| "The Pesticide Manual - A World Compendium", THE BRITISH CROP PROTECTION COUNCIL |
| "The Pesticide Manual", 2009, BRITISH CROP PROTECTION COUNCIL |
| BIOORG. MED. CHEM. LETT., vol. 15, 2005, pages 1423 - 1428 |
| BIOORG. MED. CHEM. LETT., vol. 26, no. 23, 2016, pages 5679 - 5684 |
| BIOORG. MED. CHEM. LETT., vol. 30, no. 21, 2020, pages 127508 |
| BIOORG. MED. CHEM., vol. 109, no. 22, 2016, pages 5693 - 5701 |
| CHEM. SCI., vol. 4, 2013, pages 2374 - 2379 |
| J. AM. CHEM. SOC., vol. 114, 1992, pages 5959 |
| J. CHIN. CHEM. SOC., vol. 54, no. 3, 2007, pages 643 - 652 |
| J. HET. CHEM., vol. 50, no. 4, 2013, pages 774 - 780 |
| J. ORG CHEM., vol. 26, 1961, pages 4410 - 12 |
| J. ORG CHEM., vol. 47, 1981, pages 3153 |
| J. ORGANOMET. CHEM., vol. 576, 1999, pages 147 - 168 |
| J. PRAKT. CHEM., vol. 327, 1985, pages 109 - 116 |
| ORG. BIOMOL. CHEM., vol. 13, no. 9, 2015, pages 2541 - 2545 |
| ORG. BIOMOL. CHEM., vol. 14, 2016, pages 6853 |
| ORG. LETT., vol. 10, 2008, pages 4570 |
| REACT. KINET. CAT. LETT., vol. 92, 2007, pages 99 - 104 |
| SYNLETT, vol. 4, 2007, pages 652 - 654 |
| SYNTHESIS, vol. 4, 1979, pages 281 - 283 |
| SYNTHESIS, vol. 45, 2013, pages 1569 |
| TETRAHEDRON LETT., 1977, pages 4171 - 4174 |
| TETRAHEDRON LETT., 2006, pages 5767 - 5769 |
| TETRAHEDRON, vol. 43, 1987, pages 439 |
| TETRAHEDRON, vol. 61, no. 46, 2005, pages 10827 - 10852 |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025146624A1 (en) * | 2024-01-03 | 2025-07-10 | Pi Industries Ltd. | Bicyclic heterocyclic amides for combating phytopathogenic fungi |
Also Published As
| Publication number | Publication date |
|---|---|
| EP4619396A1 (en) | 2025-09-24 |
| CO2025006132A2 (en) | 2025-05-29 |
| JP2025537818A (en) | 2025-11-20 |
| KR20250095743A (en) | 2025-06-26 |
| MX2025005559A (en) | 2025-06-02 |
| CR20250206A (en) | 2025-06-20 |
| UY40520A (en) | 2024-06-14 |
| TW202434579A (en) | 2024-09-01 |
| IL320722A (en) | 2025-07-01 |
| AR131059A1 (en) | 2025-02-12 |
| CL2025001342A1 (en) | 2025-08-22 |
| AU2023381091A1 (en) | 2025-05-15 |
| CN120225510A (en) | 2025-06-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP4380933A1 (en) | Microbiocidal pyrazole derivatives | |
| AU2023376706A1 (en) | Microbiocidal pyrazole derivatives | |
| AU2022415492A1 (en) | Microbiocidal bicycle heterocyclic derivatives | |
| EP4608836A1 (en) | Microbiocidal heterobicyclic dihydrooxadiazine derivatives | |
| AU2023348299A1 (en) | Microbiocidal pyrazole derivatives | |
| AU2024211865A1 (en) | Microbiocidal pyrazole derivatives | |
| WO2024132895A1 (en) | Microbiocidal dihydrooxadiazinyl pyridazinone compounds | |
| EP4447682A1 (en) | Microbiocidal pyrazole derivatives | |
| WO2024132901A1 (en) | Microbiocidal pyridazine dihydrooxadiazine derivatives | |
| WO2024068947A1 (en) | Microbiocidal pyrazole derivatives | |
| US20250368652A1 (en) | Microbiocidal bicyclic heterocyclic carboxamide derivatives | |
| US20250034097A1 (en) | Microbiocidal isonicotinic amide derivatives | |
| EP4313949A1 (en) | Microbiocidal isonicotinic amide derivatives | |
| WO2024105104A1 (en) | Microbiocidal tetrahydroisoquinoline derivatives | |
| WO2024115512A1 (en) | Microbiocidal tetrahydroisoquinoline derivatives | |
| WO2023166067A1 (en) | Microbiocidal pyridazinone amide derivatives | |
| WO2024115509A1 (en) | Microbiocidal tetrahydroisoquinoline derivatives | |
| WO2023094304A1 (en) | Microbiocidal heterobiaryl amide derivatives | |
| WO2023148206A1 (en) | Microbiocidal n-amide derivatives | |
| WO2025172368A1 (en) | (5-isoxazol-3-yl)-[4-(pyrazol-4-yl)-3,4-dihydro-1h-isoquinolin-2-yl]methanone derivatives for use as fungicides | |
| WO2025210095A1 (en) | Microbiocidal tetrahydroisoquinoline compounds | |
| WO2025078263A1 (en) | Microbiocidal pyridyl pyrazole derivatives |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 23805614 Country of ref document: EP Kind code of ref document: A1 |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 820988 Country of ref document: NZ Ref document number: AU2023381091 Country of ref document: AU |
|
| WWP | Wipo information: published in national office |
Ref document number: 820988 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202517043990 Country of ref document: IN Ref document number: 320722 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/A/2025/005559 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2501003136 Country of ref document: TH |
|
| ENP | Entry into the national phase |
Ref document number: 2023381091 Country of ref document: AU Date of ref document: 20231115 Kind code of ref document: A Ref document number: 2025528472 Country of ref document: JP Kind code of ref document: A |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202380079234.7 Country of ref document: CN Ref document number: 2025528472 Country of ref document: JP |
|
| WWP | Wipo information: published in national office |
Ref document number: 202517043990 Country of ref document: IN |
|
| WWP | Wipo information: published in national office |
Ref document number: MX/A/2025/005559 Country of ref document: MX |
|
| ENP | Entry into the national phase |
Ref document number: 20257019054 Country of ref document: KR Kind code of ref document: A |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01A Ref document number: 112025009724 Country of ref document: BR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020257019054 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 202591518 Country of ref document: EA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2023805614 Country of ref document: EP Ref document number: 2025116242 Country of ref document: RU |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| ENP | Entry into the national phase |
Ref document number: 2023805614 Country of ref document: EP Effective date: 20250616 |
|
| WWP | Wipo information: published in national office |
Ref document number: 1020257019054 Country of ref document: KR |
|
| WWP | Wipo information: published in national office |
Ref document number: 202380079234.7 Country of ref document: CN |
|
| WWP | Wipo information: published in national office |
Ref document number: 2025116242 Country of ref document: RU |
|
| WWP | Wipo information: published in national office |
Ref document number: 2023805614 Country of ref document: EP |
|
| REG | Reference to national code |
Ref country code: BR Ref legal event code: B01E Ref document number: 112025009724 Country of ref document: BR Free format text: APRESENTE NOVAS FOLHAS DAS REIVINDICACOES ADAPTADAS AO ART. 18 DA PORTARIA NO 14/2024, UMA VEZ QUE O CONTEUDO ENVIADO ENCONTRA-SE FORA DA NORMA. A EXIGENCIA DEVE SER RESPONDIDA EM ATE 60 (SESSENTA) DIAS DE SUA PUBLICACAO E DEVE SER REALIZADA POR MEIO DA PETICAO GRU CODIGO DE SERVICO 207. |