WO2025146624A1 - Bicyclic heterocyclic amides for combating phytopathogenic fungi - Google Patents
Bicyclic heterocyclic amides for combating phytopathogenic fungi Download PDFInfo
- Publication number
- WO2025146624A1 WO2025146624A1 PCT/IB2025/050007 IB2025050007W WO2025146624A1 WO 2025146624 A1 WO2025146624 A1 WO 2025146624A1 IB 2025050007 W IB2025050007 W IB 2025050007W WO 2025146624 A1 WO2025146624 A1 WO 2025146624A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- formula
- pyrazol
- methyl
- methanone
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D495/04—Ortho-condensed systems
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/48—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with two nitrogen atoms as the only ring hetero atoms
- A01N43/56—1,2-Diazoles; Hydrogenated 1,2-diazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/64—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with three nitrogen atoms as the only ring hetero atoms
- A01N43/647—Triazoles; Hydrogenated triazoles
- A01N43/653—1,2,4-Triazoles; Hydrogenated 1,2,4-triazoles
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/713—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with four or more nitrogen atoms as the only ring hetero atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D401/00—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom
- C07D401/14—Heterocyclic compounds containing two or more hetero rings, having nitrogen atoms as the only ring hetero atoms, at least one ring being a six-membered ring with only one nitrogen atom containing three or more hetero rings
Definitions
- the present invention relates to novel bicyclic heterocyclic amide compounds of formula (I) which are useful in combating phytopathogenic fungi, and to a process for preparing these novel bicyclic heterocyclic amide compounds of formula (I).
- the present invention also relates to compositions and to combinations comprising these novel bicyclic heterocyclic amide compounds of formula (I) and to a method for combating phytopathogenic fungi using the same.
- Bicyclic heterocyclic amide compounds are described as fungicidal agents in WO2022253645A1 and WO2023110869A1.
- the bicyclic heterocyclic amide compounds reported in the above cited literature have disadvantages in certain aspects, such as that they exhibit a narrow spectrum of efficacy or that they do not have a satisfactory fungicidal activity, particularly at low application rates.
- the present invention relates to a compound of formula (I) or an agriculturally acceptable salt, N- oxide, isomer, tautomer or polymorph thereof,
- D is selected from the group consisting of phenyl or 5- or 6-membered heteroaryl ring, wherein said 5- or 6-membered heteroaryl ring comprises 1, 2, 3 or 4 heteroatoms each independently selected from N, O, and S(O)0-2, and wherein said phenyl or 5- or 6-membered heteroaryl ring may be further substituted with one or more different or identical substituents selected from R 1b and/or R 1c ;
- R 1a is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, nitro, C1-C6- alkyl, C1-C6-haloalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C3- C8-cycloalkyl, C3-C8-halocycloalkyl, C1-C6-alkoxy, C1-C6-halo
- the present invention also relates to a process for preparing the compound of formula (I).
- the compounds of formula (I) have been found to be advantageous over the compounds reported in the literature in either of improved fungicidal activity, broader spectrum of biological efficacy, lower application rates, more favourable biological and/or environmental properties, or enhanced plant compatibility.
- the present invention further relates to agrochemical compositions comprising a compound of formula (I) or a compound of formula (I) in combination with one or more further pesticidally active substance(s) for controlling and/or preventing plant diseases, particularly caused by phytopathogenic fungi.
- the present invention still further relates to a method for controlling or preventing an infestation of useful plants by phytopathogenic microorganisms, wherein a fungicidally effective amount of a compound of formula (I), a composition or a combination thereof, is applied to the plants, to parts thereof or the locus thereof.
- a fungicidally effective amount of a compound of formula (I), a composition or a combination thereof is applied to the plants, to parts thereof or the locus thereof.
- the terms “comprises”, “comprising”, “includes”, “including”, “has”, “having”, “contains”, “containing”, “characterized by” or any other variation thereof, are intended to cover a non-exclusive inclusion, subject to any limitation explicitly indicated.
- a composition, mixture, process or method that comprises a list of elements is not necessarily limited to only those elements but may include other elements not expressly listed or inherent to such composition, mixture, process or method.
- the transitional phrase “consisting of” excludes any element, step or ingredient not specified. If in the claim, such would close the claim to the inclusion of materials other than those recited except for impurities ordinarily associated therewith.
- a condition A “or” B is satisfied by any one of the following: A is true (or present) and B is false (or not present), A is false (or not present) and B is true (or present), and both A and B are true (or present).
- the indefinite articles “a” and “an” preceding an element or component of the present invention are intended to be nonrestrictive regarding the number of instances (i.e. occurrences) of the element or component. Therefore “a” or “an” should be read to include one or at least one, and the singular word form of the element or component also includes the plural unless the number is obviously meant to be singular.
- invertebrate pests includes but is not limited to fungi, stramenopiles (oomycetes), bacteria, nematodes, mites, ticks, insects and nematodes of economic importance.
- pest control means inhibition of invertebrate pest development (including necrosis, retarded growth and/or death), and related expressions are defined analogously.
- agronomic refers to the production of field crops such as for food, feed and fiber and includes the growth of corn, soybeans and other legumes, rice, cereal (e.g., wheat, oats, barley, rye, rice, maize), leafy vegetables (e.g., lettuce, cabbage, and other cole crops), fruiting vegetables (e.g., tomatoes, pepper, eggplant, crucifers and cucurbits), potatoes, sweet potatoes, grapes, cotton, tree fruits (e.g., pome, stone and citrus), small fruit (berries, cherries) and other specialty crops (e.g., canola, sunflower, olives).
- wheat e.g., wheat, oats, barley, rye, rice, maize
- leafy vegetables e.g., lettuce, cabbage, and other cole crops
- fruiting vegetables e.g., tomatoes, pepper, eggplant, crucifers and cucurbits
- potatoes e.g., sweet potatoes, grapes, cotton, tree fruits (e.
- nonagronomic refers to other than field crops, such as horticultural crops (e.g., greenhouse, nursery or ornamental plants not grown in a field), residential, agricultural, commercial and industrial structures, turf (e.g., sod farm, pasture, golf course, lawn, sports field, etc.), wood products, stored product, agro-forestry and vegetation management, public health (i.e. human) and animal health (e.g., domesticated animals such as pets, livestock and poultry, undomesticated animals such as wildlife) applications.
- Nonagronomic applications include protecting an animal from an invertebrate parasitic pest by administering a parasiticidally effective (i.e.
- a compound of the present invention typically in the form of a composition formulated for veterinary use, to the animal to be protected.
- parasitic and “parasiticidally” refers to observable effects on an invertebrate parasite pest to provide protection of an animal from the pest. Parasiticidal effects typically relate to diminishing the occurrence or activity of the target invertebrate parasitic pest. Such effects on the pest include necrosis, death, retarded growth, diminished mobility or lessened ability to remain on or in the host animal, reduced feeding and inhibition of reproduction.
- C1-C6 alkyl used either alone or in compound words such as “alkylthio” or “haloalkyl” refers to a straight-chain or branched C1 to C6 alkyl.
- C1-C6 alkyl include methyl, ethyl, propyl, 1-methylethyl, butyl, 1-methylpropyl, 2-methylpropyl, 1,1-dimethylethyl, pentyl, 1-methylbutyl, 2-methylbutyl, 3-methylbutyl, 2,2-dimethylpropyl, 1-ethylpropyl, hexyl, 1,1- dimethylpropyl, 1,2-dimethylpropyl, 1-methylpentyl, 2-methylpentyl, 3-methylpentyl, 4- methylpentyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 1,3-dimethylbutyl, 2,2-dimethylbutyl, 2,3- dimethylbutyl, 3,3-dimethylbutyl, 1-ethylbutyl, 2-ethylbutyl, 1,1,2-trimethylpropyl, 1,2,2- trimethylpropyl, 1-ethylbutyl, 2-e
- C2-C6-alkenyl refers to straight-chain or branched C2 to C6 alkenes.
- Non-limiting examples of C2-C6-alkenyl include ethenyl, 1-propenyl, 2- propenyl, 1-methylethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1-methyl-1-propenyl, 2-methyl-l- propenyl, l-methyl-2 -propenyl, 2-methyl-2-propenyl, 1-pentenyl, 2-pentenyl, 3-pentenyl, 4- pentenyl, 1-methyl-1-butenyl, 2-methyl-1-butenyl, 3-methyl-1-butenyl, l-methyl-2-butenyl, 2- methyl-2-butenyl, 3-methyl-2-butenyl, l-methyl-3-butenyl, 2-methyl-3-butenyl, 3-methyl-3-butenyl, 1,1-di
- Alkenyl also includes polyenes such as 1,2-propadienyl and 2,4-hexadienyl. This definition also applies to alkenyl as a part of a composite substituent, for example haloalkenyl, alkenyloxy or haloalkenyloxy and the like, unless defined specifically elsewhere.
- C 2 -C 6 -alkynyl used either alone or in compound words refers to straight-chain or branched C2 to C6 alkynes.
- C2-C6-alkynes include ethynyl, 1-propynyl, 2- propynyl, 1-butynyl, 2-butynyl, 3-butynyl, 1-methyl-2-propynyl, 1-pentynyl, 2-pentynyl, 3- pentynyl, 4-pentynyl, l-methyl-2-butynyl, l-methyl-3-butynyl, 2-methyl-3-butynyl, 3-methyl-l- butynyl, 1,1-dimethyl-2-propynyl, 1-ethyl -2-propynyl, 1-hexynyl, 2-hexynyl, 3-hexynyl, 4- hexynyl, 5-hexynyl, 1-methyl-2-pentynyl, l-methyl-3-pentynyl, 1-methyl-4-pentynyl, 2-methyl-3- penty
- alkynyl as a part of a composite substituent, for example haloalkynyl etc. alkynyloxy, haloalkynyloxy, unless specifically defined elsewhere.
- alkynyl can also include moieties comprised of multiple triple bonds such as 2,5-hexadiynyl.
- C3-C8-cycloalkyl means a saturated carbocyclic ring containing 3 to 8 carbon atoms. Non-limiting examples of C3-C8-cycloalkyl include cyclopropyl, cyclopentyl and cyclohexyl.
- C3-C6-cycloalkoxy and “C3-C6-cycloalkylthio” and the like are defined analogously.
- Non limiting examples of C3-C6-cycloalkoxy or C3-C6-cycloalkylthio include cyclopropyloxy, cyclopentyloxy, cyclohexyloxy, cyclopropylthio, cyclopentylthio and cyclohexylthio.
- halogen either alone or in compound words such as “haloalkyl”, includes fluorine, chlorine, bromine or iodine. Further, when used in compound words such as “C1-C6-haloalkyl”, said C1-C6-alkyl may be partially or fully substituted with halogen atoms which may be the same or different.
- C 1 -C 6 -haloalkyl include chloromethyl, bromomethyl, dichloromethyl, trichloromethyl, fluoromethyl, difluoromethyl, trifluoromethyl, chlorofluoromethyl, dichlorofluoromethyl, chlorodifluoromethyl, 1-chloroethyl, 1-bromoethyl, 1- fluoroethyl, 2-fluoroethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, 2-chloro-2-fluoroethyl, 2-chloro- 2,2-difluoroethyl, 2,2-dichloro-2-fluoroethyl, 2,2,2-trichloroethyl, pentafluoroethyl, 1,1-dichloro- 2,2,2-trifluoroethyl, and 1,1,1-trifluoroprop-2-yl.
- C 2 -C 6 -haloalkenyl “C 2 -C 6 -haloalkynyl” are defined analogously except that, instead of C 1 -C 6 -alkyl groups, C 2 -C 6 -alkenyl and C 2 -C 6 -alkynyl groups are present as a part of the substituent.
- C 1 -C 6 -alkoxy used either alone or in compound words includes a straight or branched chain C 1 -C 6 -alkoxy.
- C 1 -C 6 -alkoxy examples include methoxy, ethoxy, propoxy, 1-methylethoxy, butoxy, 1-methylpropoxy, 2-methylpropoxy, 1,1-dimethylethoxy, pentoxy, 1-methylbutoxy, 2- methylbutoxy, 3-methylbutoxy, 2,2-dimethylpropoxy, 1-ethylpropoxy, hexoxy, 1,1- dimethylpropoxy, 1,2-dimethylpropoxy, 1-methylpentoxy, 2-methylpentoxy, 3-methylpentoxy, 4- methylpentoxy, 1,1-dimethylbutoxy, 1,2-dimethylbutoxy, 1,3-dimethylbutoxy, 2,2-dimethylbutoxy, 2,3-dimethylbutoxy, 3,3-dimethylbutoxy, 1-ethylbutoxy, 2-ethylbutoxy, 1,1,2-trimethylpropoxy, 1,2,2-trimethylpropoxy, 1-ethyl-1-methylpropoxy and l-ethyl-2-methylpropoxy and the different isomers.
- C1-C6-haloalkoxy means straight-chain or branched C1-C6-alkoxy groups where some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as specified above.
- Non-limiting examples of C1-C6-haloalkoxy include chloromethoxy, bromomethoxy, dichloromethoxy, trichloromethoxy, fluoromethoxy, difluoromethoxy, trifluoromethoxy, chlorofluoromethoxy, dichlorofluoromethoxy, chlorodifluoromethoxy, 1-chloroethoxy, 1- bromoethoxy, 1-fluoroethoxy, 2-fluoroethoxy, 2,2-difluoroethoxy, 2,2,2-trifluoroethoxy, 2-chloro- 2-fluoroethoxy, 2-chloro-2,2-difluoroethoxy, 2,2-dichloro-2-fluoroethoxy, 2,2,2-trichloroethoxy, pentafluoroethoxy and l,l,l-trifluoroprop-2-oxy.
- C1-C6-alkylthio includes branched or straight-chain alkylthio moieties with 1 to 6 carbon atoms.
- C1-C6-alkylthio includes methylthio, ethylthio, propylthio, 1- methylethylthio, butylthio, 1-methylpropylthio, 2-methylpropylthio, 1,1-dimethylethylthio, pentylthio, 1-methylbutylthio, 2-methylbutylthio, 3-methylbutylthio, 2,2-dimethylpropylthio, 1- ethylpropylthio, hexylthio, 1,1-dimethylpropylthio, 1,2-dimethylpropylthio, 1-methylpentylthio, 2- methylpentylthio, 3-methylpentylthio, 4-methylpentylthio, 1,1-dimethylbutylthio, 1,2- dimethylbutylthio, 1,3-dimethylbutylthio, 2,2-dimethylbutylthio, 2,3-dimethylbutylthio
- C 1 -C 6 -haloalkylthio means straight-chain or branched C 1 -C 6 -alkylthio group where some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as specified above.
- Non-limiting examples of haloalkylthio include chloromethylthio, bromomethylthio, dichloromethylthio, trichloromethylthio, fluoromethylthio, difluoromethylthio, trifluoromethylthio, chlorofluoromethylthio, dichlorofluoromethylthio, chlorodifluoromethylthio, 1-chloroethylthio, 1- bromoethylthio, 1- fluoroethylthio, 2-fluoroethylthio, 2,2-difluoroethylthio, 2,2,2-trifluoroethylthio, 2-chloro-2- fluoroethylthio, 2-chloro-2,2-difluoroethylthio, 2,2-dichloro-2-fluoroethylthio, 2,2,2- trichloroethylthio, pentafluoroethylthio and l,l,
- C1-C6-alkylsulphinyl includes branched or straight-chain alkylsulphinyl moiety with 1 to 6 carbon atoms.
- C1-C6-alkylsulfinyl include methylsulphinyl, ethylsulphinyl, propylsulphinyl, 1-methylethylsulphinyl, butylsulphinyl, 1-methylpropylsulphinyl, 2-methylpropylsulphinyl, 1,1-dimethylethylsulphinyl, pentylsulphinyl, 1-methylbutylsulphinyl, 2- methylbutylsulphinyl, 3-methylbutylsulphinyl, 2,2-dimethylpropylsulphinyl, 1- ethylpropylsulphinyl, hexylsulphinyl, 1,1-dimethylpropylsulphinyl, 1,2-dimethylpropylsulphinyl, 1-methylpentylsulphinyl, 2-methylpentylsulphinyl, 3-methylpentylsulphiny
- C1-C6-alkylsulfonyl includes branched or straight-chain alkylsulfonyl moiety with 1 to 6 carbon atoms.
- Non-limiting examples of C1-C6-alkylsulfonyl include methylsulfonyl, ethylsulfonyl, propylsulfonyl, 1-methylethylsulfonyl, butylsulfonyl, 1-methylpropylsulfonyl, 2- methylpropylsulfonyl, 1,1-dimethylethylsulfonyl, pentylsulfonyl, 1-methylbutylsulfonyl, 2- methylbutylsulfonyl, 3-methylbutylsulfonyl, 2,2-dimethylpropylsulfonyl, 1-ethylpropylsulfonyl, hexylsulfonyl, 1,
- C 1 -C 6 -alkylcarbonyl includes branched or straight-chain alkylcarbonyl moiety with 1 to 6 carbon atoms.
- Non-limiting examples of C1-C6-alkylcarbonyl include methylcarbonyl, ethylcarbonyl, propylcarbonyl, 1-methylethylcarbonyl, butylcarbonyl, 1-methylpropylcarbonyl, 2- methylpropylcarbonyl, 1,1-dimethylethylcarbonyl, pentylcarbonyl, 1-methylbutylcarbonyl, 2- methylbutylcarbonyl, 3-methylbutylcarbonyl, 2,2-dimethylpropylcarbonyl, 1-ethylpropylcarbonyl, hexylcarbonyl, 1,1-dimethylpropylcarbonyl, 1,2-dimethylpropylcarbonyl, 1-methylpentylcarbonyl, 2-methylpentylcarbonyl, 3-methylpentylcarbonyl
- C6-C10 aryl refers to mono or bicyclic aromatic carbocyclic ring containing 6 to 10 carbon atoms.
- Non-limiting examples of “C6-C10 aryl” includes phenyl, indenyl, indanyl, naphthyl.
- the term “5- or 6-membered aromatic heterocyclic ring or 5- or 6-membered heteroaromatic ring” refers to a mono cyclic aromatic heterocyclic ring comprising at least one heteroatom selected from nitrogen, oxygen, or suphur or a group selected from S(O) or S(O)2.
- Non-limiting examples for said term includes furanyl, pyrrolyl, thiophenyl (thienyl), oxazolyl, thiazolyl, imidazolyl, pyrazolyl, isoxazolyl, isothiazolyl, oxadiazolyl, thiadiazolyl, triazolyl, tetrazolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, triazinyl etc.
- heterocyclic ring includes "aromatic or non-aromatic heterocyclic ring comprising at least one heteroatom selected from N, O, and S(O)0-2.
- substituents When a compound is substituted with a substituent bearing a subscript that indicates the number of said substituents can exceed 1, said substituents (when they exceed 1) are independently selected from the group of defined substituents. Further, when the subscript m in (R)m indicates an integer ranging from for example 0 to 4 then the number of substituents may be selected from the integers between 0 and 4 inclusive.
- substituents When a group contains a substituent which can be hydrogen, then, when this substituent is taken as hydrogen, it is recognized that said group is being un-substituted.
- Optionally substituted groups may be mono- or polysubstituted, where the substituents in the case of polysubstitutions may be the same or different.
- the inventive compound of the present invention may, if appropriate, be present as mixtures of different possible isomeric forms, especially of stereoisomers, for example E and Z, threo and erythro, and also optical isomers, but if appropriate also of tautomers. Both the E and the Z isomers, but also the threo and erythro isomers, and the optical isomers, and any desired mixtures of these isomers and the possible tautomeric forms are disclosed and claimed.
- polymorph refers to a particular crystalline form of a chemical compound that can crystallize in different crystalline forms, these forms having different arrangements and/or conformations of the molecules in the crystal lattice.
- polymorphs can have the same chemical composition, they can also differ in composition due to the presence or absence of co- crystallized water or other molecules, which can be weakly or strongly bound in the lattice. Polymorphs can differ in such chemical, physical and biological properties as crystal shape, density, hardness, color, chemical stability, melting point, hygroscopicity, suspensibility, dissolution rate and biological availability.
- a polymorph of a compound represented by formula (I), formula (I-A) to formula (I-F) or formula (I-D-a) can exhibit beneficial effects (e.g., suitability for preparation of useful formulations, improved biological performance) relative to another polymorph or a mixture of polymorphs of the same compound represented by formula (I), formula (I-A) to formula (I-F) or formula (I-D-a).
- the preparation and isolation of a particular polymorph of a compound represented by formula (I), formula (I-A) to formula (I-F) or Formula (I-D-a) can be achieved by methods known to those skilled in the art including, for example, crystallization using selected solvents and temperatures.
- plants for the purpose of the present disclosure includes but is not limited to fungi, stramenopiles (oomycetes) and bacteria.
- plant is understood here to mean all plants and plant populations, such as desired and undesired wild plants or crop plants (including naturally occurring crop plants). Crop plants may be plants which can be obtained by conventional breeding and optimization methods or by biotechnological and genetic engineering methods or combinations of these methods, including transgenic plants and including the plant cultivars which are protectable and non-protectable by plant breeders’ rights.
- plant includes a living organism of the kind exemplified by trees, shrubs, herbs, grasses, ferns, and mosses, typically growing in a site, absorbing water and required substances through its roots, and synthesizing nutrients in its leaves by photosynthesis.
- plant for the purpose of the present invention include but are not limited to agricultural crops such as wheat, rye, barley, triticale, oats or rice; beet, e.g. sugar beet or fodder beet; fruits and fruit trees, such as pomes, stone fruits or soft fruits, e.g.
- leguminous plants such as lentils, peas, alfalfa or soybeans; oil plants, such as rape, mustard, olives, sunflowers, coconut, cocoa beans, castor oil plants, oil palms, ground nuts or soybeans; cucurbits, such as squashes, cucumber or melons; fiber plants, such as cotton, flax, hemp or jute; citrus fruit and citrus trees, such as oranges, lemons, grapefruits or mandarins; any horticultural plants, vegetables, such as spinach, lettuce, asparagus, cabbages, carrots, onions, tomatoes, potatoes, cucurbits or paprika; lauraceous plants, such as avocados, cinnamon or camphor; cucurbitaceae; oleaginous plants; energy and raw material plants, such as cereals, corn, soybean, other leguminous plants, rape, sugar cane or oil palm; tobacco; nuts; coffee; tea;
- the plant for the purpose of the present invention includes but is not limited to cereals, corn, rice, soybean and other leguminous plants, fruits and fruit trees, grapes, nuts and nut trees, citrus and citrus trees, any horticultural plants, cucurbitaceae, oleaginous plants, tobacco, coffee, tea, cacao, sugar beet, sugar cane, cotton, potato, tomato, onions, peppers and vegetables, ornamentals, any floricultural plants and other plants for use of humans and animals.
- plant parts is understood to mean all parts and organs of plants above and below the ground.
- plant parts include but is not limited to cuttings, leaves, twigs, tubers, flowers, seeds, branches, roots including taproots, lateral roots, root hairs, root apex, root cap, rhizomes, slips, shoots, fruits, fruit bodies, bark, stem, buds, auxillary buds, meristems, nodes and internodes.
- locus thereof includes soil, surroundings of plant or plant parts and equipment or tools used before, during or after sowing/planting a plant or a plant part.
- compositions optionally comprising other compatible compounds to a plant or a plant material or locus thereof includes application by a technique known to a person skilled in the art which includes but is not limited to spraying, coating, dipping, fumigating, impregnating, injecting and dusting.
- applied means adhered to a plant or plant part either physically or chemically including impregnation.
- the present invention provides a compound of formula (I) or an agriculturally acceptable salt, N- oxide, isomer, tautomer or polymorph thereof,
- D is selected from the group consisting of phenyl or 5- or 6-membered heteroaryl ring, wherein said 5- or 6-membered heteroaryl ring comprises 1, 2, 3 or 4 heteroatoms each independently selected from N, O, and S(O)0-2, and wherein said phenyl or 5- or 6-membered heteroaryl ring may be further substituted with one or more different or identical substituents selected from R 1b and/or R 1c ;
- R 1a is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, nitro, C1-C6- alkyl, C1-C6-haloalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C3- C8-cycloalkyl, C3-C8-halocycloalkyl, C1-C6-alkoxy, C1-C6-halo
- D is selected from the group consisting of phenyl or 5- or 6-membered heteroaryl ring, wherein said 5- or 6-membered heteroaryl ring comprises 1, 2, 3 or 4 heteroatoms each independently selected from N, O, and S(O)0-2, and wherein said phenyl or 5- or 6-membered heteroaryl ring may be further substituted with one or more different or identical substituents selected from R 1b and/or R 1c ;
- R 1a is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, nitro, C1-C6- alkyl, C1-C6-haloalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C
- C3 and C4 carbons of the piperidine ring are part of the 5-membered aromatic heterocyclic ring), wherein the 5-membered aromatic heterocyclic ring comprises 1 to 3 heteroatoms selected from oxygen, nitrogen or S(O)0-2, wherein the ring B is unsubstituted or substituted with one or more different or identical substituents selected from R 6 ;
- R 6 can be selected from the group consisting of hydrogen, halogen, cyano, hydroxyl, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C3-C8-cycloalkyl, C3-C8-halocycloalkyl, C6- C10-aryl which may be substituted with 1 or 2 same or different substitutent selected from the group consisting of halogen, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6-alk
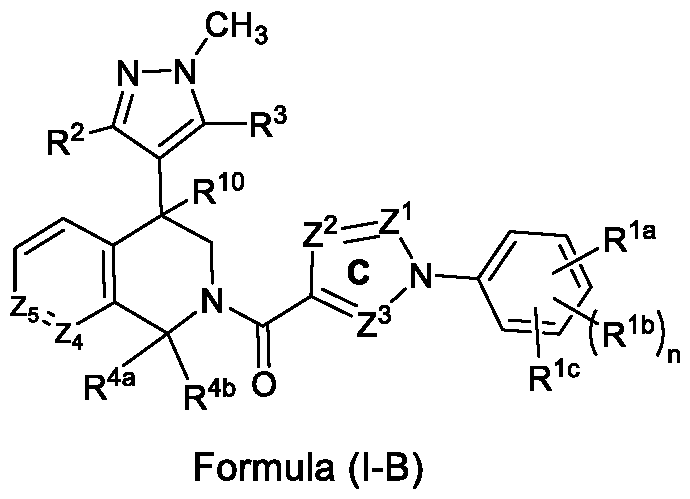
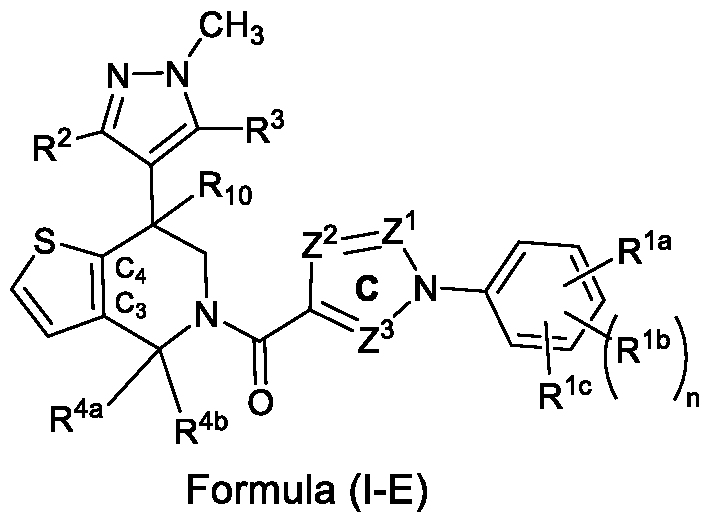
- B is a 5-membered aromatic heterocyclic ring (C3, C4 carbons of the piperidine ring are part of the 5-membered aromatic heterocyclic ring) which may be selected as thiophenyl (thienyl), that is fused at C 3 , C 4 carbons of the piperidine ring, wherein said ring is unsubstituted or substituted with one or more different or identical substituents selected from R 6 ; and n, R 1a -R 1c , R 2 , R 3 , R 4a , R 4b , R 10 and Z 1 -Z 3 are as defined above for the compound of formula (I).
- R 1a is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, nitro, C 1 -C 6 - alkyl, C 1 -C 6 -haloalkyl, C 2 -C 6 -alkenyl, C 2 -C 6 -haloalkenyl, C 2 -C 6 -alkynyl, C 2 -C 6 -haloalkynyl, C 3 - C 8 -cycloalkyl, C 3 -C 8 -halocycloalkyl, C 1 -C 6 -alkoxy, C 1 -C 6 -haloalkoxy, C 2 -C 6 -alkenyloxy, C 2 -C 6 - haloalkenyloxy, C 2 -C 6 -alkynyloxy, C 2 -C 6 -haloalkyl
- R 1a is selected from the group consisting of halogen, cyano, C 1 -C 6 -alkyl, C 1 -C 6 -haloalkyl, C 3 -C 6 - cycloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C3-C6-cycloalkyloxy, C1-C6-alkylthio, C1-C6- haloalkylthio, C1-C6-alkylsulfonyl, more preferably fluoro, chloro, bromo, iodo, cyano, methyl, trifluoromethyl, O-isopropyl or cyclopropyl; R 1b is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6-alkyl, C1-C6-F
- the compound of formula (I) may be selected from a compound of formula (I-D-a), , wherein, R 2 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R 3 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C1-C6-cyanoalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6- haloalkynyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R 4a
- R 1a is selected from fluoro, chloro, bromo, iodo, cyano, methyl, trifluoromethyl, -O-isopropyl, or cyclopropyl
- R 1b is selected from hydrogen, fluoro, chloro, bromo, cyano, methyl, trifluoromethyl and cyclopropyl
- R 1c is selected from hydrogen, fluoro, chloro or bromo or cyano
- R 2 is selected from hydrogen, or chloro
- R 3 is selected from hydrogen, chloro, methyl, CH 2 Cl, or CH 2 CN
- R 4a is selected from hydrogen or methyl
- R 4b is selected from hydrogen
- ring C selected from one of the following rings C-1 to C-7: trifluoromethyl or cyclopropyl and in ring C-7,
- R 5 can be same or different selected from hydrogen, chloro, methyl, ethyl
- Formula (I) or Formula (I-A) to Formula (I-F) is selected from: D-1 D-2 D-3 D-4 D-6 D-5 D-11 D-7 D-8 D-9 D-10 D-12 D-13 D-14 D-15 D-16 D-17 D-18 D-19 D-20 D-21 D-22 D-23 D-24
- the present invention provided the compound of formula (I-A) to formula (I-F), is particularly described in the below table: Table A describes the compounds of formula (I-A) to formula (I- F).
- salts obtainable in this way likewise have fungicidal properties.
- PI External Anions of useful acid addition salts are primarily chloride, bromide, fluoride, hydrogensulfate, sulfate, dihydrogenphosphate, hydrogenphosphate, phosphate, nitrate, bicarbonate, carbonate, hexafluorosilicate, hexafluorophosphate, benzoate, and the anions of C 1 -C 4 -alkanoic acids, preferably formate, acetate, propionate and butyrate.
- the compounds of the present invention can be present either in pure form or as mixtures of different possible isomeric forms such as stereoisomers e.g. a racemate, individual stereoisomers, or constitutional isomers or as an optically active form.
- the various stereoisomers include enantiomers, diastereomers, chiral isomers, atropisomers, conformers, rotamers, tautomers, optical isomers, polymorphs, and geometric isomers. Any desired mixtures of these isomers fall within the scope of the claims of the present disclosure.
- one stereoisomer may be more active and/or may exhibit beneficial effects when enriched relative to the other isomer(s) or when separated from the other isomer(s).
- processes or methods or technologies to separate, enrich, and/or to selectively prepare said isomers are examples of the compounds.
- the compounds of formula (I) or formula (I-A) to formula (I-F) or formula (I-D-a) can be present in different crystal modifications whose biological activity may differ. They also form part of the subject matter of the present invention.
- the compounds of formula (I) or formula (I-A) to formula (I-F) or formula (I-D-a) can be present in atropisomers arising from restricted rotation about a single bond of asymmetric groups. They also form part of the subject matter of the present invention.
- the compounds selected from formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) may typically exist in more than one form.
- the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) thus include all crystalline and non-crystalline forms of the compound that formula (I), or formula (I-A) to formula (I- F), or formula (I-D-a).
- Non-crystalline forms include embodiments which are solids such as waxes and gums as well as embodiments which are liquids such as solutions and melts.
- Crystalline forms include embodiments which represent essentially a single crystal type and embodiments which represent a mixture of polymorphs (i.e. different crystalline types).
- the following schemes illustrate approaches for preparing compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a).
- the present invention provides a process for the synthesis of compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a).
- PI External The following schemes illustrate approaches for preparing the compounds of formula (I), or formula (I- A) to formula (I-F) or formula (I-D-a).
- the following descriptions and examples are provided for illustrative purposes and should not be construed as limiting in terms of substituents or substitution patterns.
- the compounds of the present invention as defined by formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and/or in the table 1 may be prepared, in a known manner, in a variety of ways as described in the schemes 1-6.
- the definitions of B, R 1a -R 1c , R 2 , R 3 , R 4 and Z 1 -Z 3 in the compounds of formulae (I), (I-A to I-F, I-D-a), are as defined above in the detailed description of the invention unless otherwise stated specifically.
- the compounds of general formula (I-A) can be prepared by acid- amine coupling reactions between an amine compound of formula 1 (or a salt thereof) and a compound of general formula 2 wherein X represents -OH or -halo group in the presence or absence of coupling reagents such as 1-propanephosphonic acid cyclic anhydride, hexafluorophosphate azabenzotriazole tetramethyl uronium, N-(3-dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride, and the like or alkyl chloroformates such as ethyl chloroforamate, isobutylchloroformate, and the like and a solvent such as dichloromethane, tetrahydrofuran, N,N-dimethyl formamide etc.
- coupling reagents such as 1-propanephosphonic acid cyclic anhydride, hexafluorophosphate azabenzotri
- Scheme-2 As shown in scheme 2, the compounds of general formula (I-A) can also be prepared by ester-amine coupling reactions between an amine compound of formula 1 (or a salt thereof, particularly HCl salt) and a compound of general formula 3, wherein R is alkyl or aryl group in the presence or absence of a base such as LiHMDS, KOt-Bu, and the like, and in the presence or absence of activating reagents such PI External as Lewis acids. These reactions are carried out in various solvents such as toluene, tetrahydrofuran, methanol and the like.
- Scheme-3 According to scheme 3, the compounds represented by general formula (I-A) can also be prepared by transition metal catalyzed C-N coupling of an amine of formula 4 and aryl halide or pseudo-halide compound of formula 5.
- the compounds of formula 5 with required substitution pattern can either be purchased from commercial sources or can be prepared using literature protocols.
- Another requisite coupling partner of general formula 4 can be constructed via acid-amine coupling reaction between an acid or acyl halide of formula 10 and an amine of general formula 1 (or salt thereof preferably its hydrochloric acid salt).
- the C-N coupling reactions are usually carried out in the presence of solvents, while the solvents which can be used for this reaction are not particularly limited as long as they do not adversely affect the reaction.
- solvents such as 1,4-dioxane, tetrahydrofuran, ethylene glycol, dimethyl ether and diethylene glycol dimethyl ether; aromatic hydrocarbons such as benzene, toluene and xylene; N- amides such as N,N-dimethylformamide, N-dimethylacetamide and 1-methyl-2-pyrrolidone; alcohols such as methanol, ethanol, propanol, butanol, 2-propanol and 2-methyl-2-propanol; nitriles such as acetonitrile; or water or a mixture thereof may serve this purpose.
- Preferable solvents include aromatic hydrocarbons such as toluene and xylene; and ethers such as 1,4-dioxane, tetrahydrofuran; out of which toluene and 1,4-dioxane are the most preferable ones.
- the C-N coupling reaction is carried out in the presence of a base selected from, but not limited to, metal alkoxides such as sodium methoxide, sodium ethoxide, potassium tert-butoxide, and sodium tert- butoxide; inorganic bases such as sodium hydroxide, potassium hydroxide, sodium carbonate, potassium carbonate, cesium carbonate, sodium phosphate, tribasic potassium phosphate, potassium hydrogenphosphate, and potassium hydride; organic bases such as triethylamine, N,N- diisopropylethylamine and pyridine.
- metal alkoxides such as sodium methoxide, sodium ethoxide, potassium tert-butoxide, and sodium tert- butoxide
- inorganic bases such as sodium hydroxide, potassium hydroxide, sodium carbonate, potassium carbonate, cesium carbonate, sodium phosphate, tribasic potassium phosphate, potassium hydrogenphosphate, and potassium hydride
- organic bases such as triethyl
- Preferred bases include metal alkoxides such as potassium tert- butoxide, sodium tert-butoxide and inorganic bases such as cesium carbonate and tribasic potassium phosphate.
- the palladium catalysts that can be used for this C-N coupling reaction include, but are not limited to, inorganic palladium salts such as palladium chloride; organic palladium complexes such as palladium PI External acetate; tetrakis(triphenylphosphine)palladium(0), bis (triphenylphosphine)palladium(II) chloride, 1,1'- bis (diphenylphosphino) phenylpalladium (II) chloride, and tris(dibenzylidene acetone)dipalladium(0).
- Palladium catalysts such as palladium chloride, palladium acetate, tetrakis (triphenylphosphine) palladium (0), bis (triphenylphosphine) palladium (II) chloride, 1'- bis (diphenylphosphino) phenylpalladium (II) chloride and tris (dibenzylidene acetone) dipalladium (0) are more preferable.
- the C-N coupling reaction can also be carried out using further ligands in combination with the above- mentioned catalysts for a fruitful transformation.
- a list of possible ligands for this transformation include, but are not limited to, tris(o-tolyl)phosphine, tricyclohexylphosphine, tri-tert- butylphosphonium tetrafluoroborate, (oxydi-2,1-phenylene)bis[dicyclohexyl] phosphine, 4,5- bis(diphenylphosphino)-9,9-dimethylxanthene, 2-dicyclohexylphosphino-2′-(N,N- dimethylamino)biphenyl, 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl, 2- dicyclohexylphosphino-2′,6′-dimethoxybiphenyl, 2-dicyclohexylphosphino-2′,6′- diisopropoxybiphenyl, 2-di-tert-butylphosphin
- the preferred ligands include 2-(di-tert-butylphosphino)biphenyl, 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl, 2-di-tert- butylphosphino-3,4,5,6-tetramethyl-2′,4′,6′-triisopropyl-1,1′-biphenyl (Oxydi-2,1- phenylene)bis[dicyclohexyl] phosphine, 1,1′-ferrocenediyl-bis(diphenylphosphine and 4,5- bis(diphenylphosphino)-9,9-dimethylxanthene.
- Pre-catalysts can also be utilized for such transformations.
- a list of possible pre-catalysts include, but are not limited to, (SP-4-3)-[dicyclohexyl[3,6-dimethoxy- 2′,4′,6′-tris(1-methylethyl)[1,1′-biphenyl]-2-yl]phosphine- ⁇ P](methanesulfonato- ⁇ O)[2′- (methylamino- ⁇ N)[1,1′-biphenyl]-2-yl- ⁇ C], [(2-di-cyclohexylphosphino-3,6-dimethoxy-2′,4′,6′- triisopropyl-1,1′-biphenyl)-2-(2′-amino-1,1′ -biphenyl)]palladium(II) methanesulfonate methanesulfonate, [(2-di-
- PI External Halogens such as, chloro-, bromo-, and iodo-
- LG trivial leaving groups
- tosylates, mesylates, and triflates in somewhat similar manner, as leaving groups allowing fruitful transformations.
- the reactions can be performed at a temperature range of 0-200 °C, preferably at a temperature range of 50-150 °C for a duration of time between 30 minutes to 24 hours.
- Scheme-4 As shown in scheme 4, the compound denoted by general formula (I-A) can also be synthesized through oxidation of the active methylene group (for instance benzylic -CH 2 -) as present in the compound of formula 7 in the presence or absence of metal oxides such as Iron oxide, Manganese oxide, Selenium dioxide etc or in the presence of oxygen atmosphere. These types of transformations are typically carried out in solvents such as water, ethyl acetate, acetonitrile, dimethyl sulfoxide at a temperature in the range of 25 °C to 100 °C.
- solvents such as water, ethyl acetate, acetonitrile, dimethyl sulfoxide at a temperature in the range of 25 °C to 100 °C.
- the compound represented by formula (I-C) can be prepared via acid-amine coupling reaction of an amine (or it’s Hydrochloric acid salt) of formula 8 and an acid or acyl halide denoted by general formula 2 in the presence of commonly used coupling reagents and conditions.
- Scheme-6 In another preferred embodiment, the compound represented by formula (I-D) can also be prepared via amide coupling reaction of an amine (or salt thereof particularly Hydrochloric acid salt) of formula 9 PI External and an acid or acyl halide denoted by general formula 2 under commonly used coupling reaction conditions.
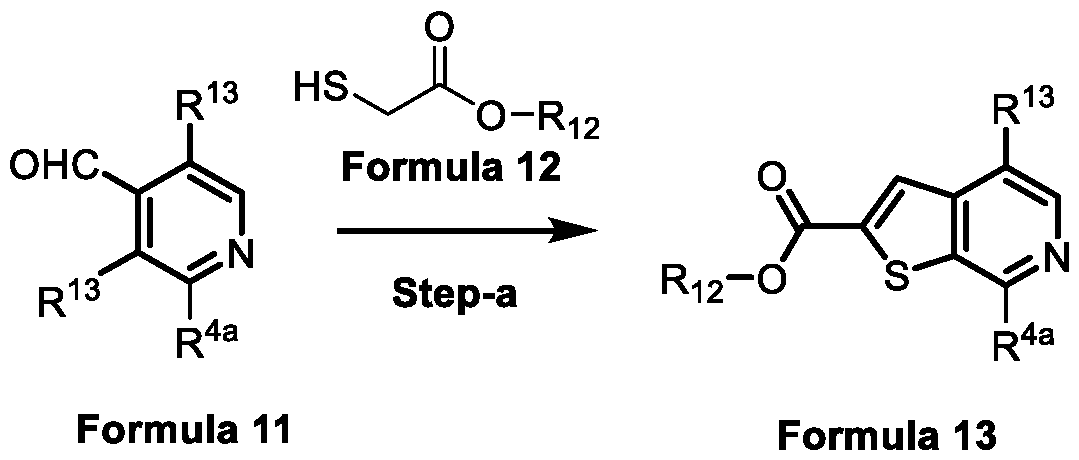
- a compound of formula 9A of the present invention can be prepared by treating a compound of formula 11 with a compound of formula 12 in a solvent and a base to obtain a compound of formula 13 as shown in scheme-7.
- the compound of formula 13 is then subjected to Suzuki coupling reaction with an intermediate compound of formula 14 using a transition metal catalyst and a base, followed by ester hydrolysis and decarboxylation to obtain a compound of formula 17.
- the compound of formula 17 is converted to the compound of formula 9A using mild and economically cheaper reagents such as iodine and NaBH4 in a suitable solvent preferably selected from tetrahydrofuran or 1,4-dioxane.
- Scheme-8 :
- the compound of formula 9A of the present invention can also be prepared according to Scheme-8, wherein a compound of formula 13 is subjected to ester hydrolysis and subsequently to decarboxylation to obtain a compound of formula 19 which is then converted to a compound of formula 17 using a transitional metal catalyst and a base in a solvent under Suzuki C-C coupling reaction conditions.
- the compound of formula 17 is finally reduced using economically cheaper reagents such as iodine and NaBH 4 in a suitable solvent preferably tetrahydrofuran or 1,4- dioxane to obtain the compound of formula 9A.
- Scheme-9 In yet another preferred embodiment, a compound of formula 9A obtained by the above scheme-7 or scheme-8, can be reacted with a compound of formula 2A in the presence of a suitable reagent and a solvent as shown in Scheme-9 to obtain a compound of formula (I-D-a).
- PI External In one embodiment the present invention, it provides a method for preparing a compound of formula (1), wherein said method comprising the step of: A.
- R 2 is selected from the group consisting of hydrogen, halogen, cyano, C 1 -C 6 -alkyl, C 1 -C 6 - haloalkyl, C 3 -C 8 -cycloalkyl, C 1 -C 6 -alkoxy, and C 1 -C 6 -haloalkoxy;
- R 3 is selected from the group consisting of hydrogen, halogen, cyano, C 1 -C 6 -alkyl, C 1 -C 6 - haloalkyl, C1-C6-cyanoalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6- halo
- the present invention provides a method for preparing a compound of formula (1), wherein said method comprising the step of: A. reducing a compound of formula (1A) with NaBH4 and iodine (I2) in a solvent to obtain a compound of formula (1), wherein B represents a phenyl ring or a 5-membered aromatic heterocyclic ring, preferably phenyl or thiophenyl (thienyl) ring; and R 2 , R 3 , R 4a and R 4b are as defined above, preferably R 4b is hydrogen.
- PI External The compounds of the present disclosure may be applied by a variety of known techniques, either as the compounds themselves or as formulations comprising these compounds.
- the compounds may be applied to the roots or foliage of plants for the control of various fungi, without damaging the commercial value of the plants.
- the materials may be applied in the form of any of the generally used formulation types, for example, as solutions, dusts, wettable powders, flowable concentrates, or emulsifiable concentrates.
- the compounds of the present disclosure are applied in the form of a formulation, comprising one or more of the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) together with a phytologically acceptable carrier.
- Concentrated formulations may be dispersed in water, or other liquids, for application, or formulations may be dust-like or granular, which may then be applied without further treatment.
- the formulations can be prepared according to procedures that are conventional in the agricultural chemical art.
- the present disclosure contemplates all vehicles by which one or more of the compounds may be formulated for delivery and use as a fungicide.
- formulations are applied as aqueous suspensions or emulsions.
- Such suspensions or emulsions may be produced from water-soluble, water- suspendible, or emulsifiable formulations which are solids, usually known as wettable powders; or liquids, usually known as emulsifiable concentrates, aqueous suspensions, or suspension concentrates.
- any material to which these compounds may be added may be used, provided it yields the desired utility without significant interference with the activity of these compounds as antifungal agents.
- the present invention provides an agrochemical composition
- agrochemical composition comprising a compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), agriculturally acceptable salts, constitutional isomers, stereo-isomers, diastereoisomers, enantiomers, chiral isomers, atropisomers, conformers, rotamers, tautomers, optical isomers, polymorphs, geometric isomers, or N- oxides thereof, optionally with one or more additional active ingredient(s), and optionally together with an auxiliary such as an inert carrier or any other essential ingredient(s) such as surfactants, additives, solid diluents and liquid diluents.
- an auxiliary such as an inert carrier or any other essential ingredient(s) such as surfactants, additives, solid diluents and liquid diluents.
- An agrochemical composition comprises a fungicidally effective amount of a compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a).
- effective amount denotes an amount of the composition or of the compound of formula (I), or formula (I-A) to formula (I-F) or formula (I- D-a), which is sufficient for controlling harmful fungi on cultivated plants or in the protection of materials and which does not result in a substantial damage to the treated plants.
- the present invention provides a composition comprising at least one compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and seeds.
- the amount of the compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) in the composition ranges from 0.1 g ai (gram per active ingredient) to 1 kg ai (kilogram per active ingredient) per 100 kg of seeds.
- the compounds of formula (I), formula (I-A) or formula (I-B formula (I), or formula (I-A) to formula (I-F), or formula (I-D-a), their N-oxides, isomers, polymophs or the agriculturally acceptable salts thereof can be converted into customary types of agrochemical compositions, e. g.
- composition types are suspensions (e. g. SC, OD, FS), emulsifiable concentrates (e. g. EC), emulsions (e. g. EW, EO, ES, ME), capsules (e. g. CS, ZC), pastes, pastilles, wettable powders or dusts (e. g. WP, SP, WS, DP, DS), pressings (e. g. BR, TB, DT), granules (e. g.
- compositions are defined in the "Catalogue of pesticide Formulation types and international coding system", Technical Monograph No. 2, 6 th Ed. May 2008, CropLife International. The mentioned compositions are prepared in a known manner, such as described by Mollet and Grubemann, “Formulation Technology”, Wiley VCH, Weinheim, 2001; or Knowles, “New Developments in Crop Protection Product Formulation”, Agrow Reports DS243, T&F Informa, London, 2005.
- auxiliaries for formulations and/or agrochemical compositions according to the invention are solvents, liquid carriers, solid carriers or fillers, surfactants, dispersants, emulsifiers, wetters, adjuvants, solubilizers, penetration enhancers, protective colloids, adhesion agents, thickeners, humectants, repellents, attractants, feeding stimulants, compatibilizers, bactericides, anti-freezing agents, anti-foaming agents, colorants, tackifiers and binders.
- the agrochemical compositions generally comprise between 0.01 and 95%, preferably between 0.1 and 90%, and in particular between 0.5 and 75%, by weight of active ingredient (ai).
- the active ingredients (ai) are employed in a purity from 90% to 100%, preferably from 95% to 100% (according to NMR spectrum).
- solutions for seed treatment (LS), suspoemulsions (SE), flowable concentrates (FS), powders for dry treatment (DS), water-dispersible powders for slurry treatment (WS), water-soluble powders (SS), emulsions (ES), PI External emulsifiable concentrates (EC), and gels (GF) are usually employed.
- the compositions in question give, after two-to-tenfold dilution, active substance concentrations from 0.01 to 60% by weight, preferably from 0.1 to 40%, in the ready-to-use preparations.
- the amounts of active substances applied are, depending on the kind of effect desired, from 0.001 to 2 kg per ha, preferably from 0.005 to 2 kg per ha, more preferably from 0.05 to 1.0 kg per ha, and in particular from 0.1 to 0.5 kg per ha.
- the generally required amounts of active substance are ranging from 0.1 to 1000 g, preferably from 1 to 1000 g, more preferably from 1 to 100 g and most preferably from 5 to 100 g, per 100 kg of plant propagation material (preferably seeds).
- the amount of active substance applied depends on the kind of application area and on the desired effect.
- Amounts customarily applied in the protection of materials are ranging from 0.001 g to 2 kg, preferably from 0.005 g to 1 kg, of active substance per cubic meter of treated material.
- Various types of oils, wetters, adjuvants, fertilizer, or micronutrients, and further pesticides e. g. herbicides, insecticides, fungicides, growth regulators, safeners, biopesticides
- pesticides e. g. herbicides, insecticides, fungicides, growth regulators, safeners, biopesticides
- These agents can be mixed with the composition according to the invention in a weight ratio of 1:100 to 100:1, preferably 1:20 to 20:1.
- a pesticide is generally a chemical or biological agent (such as pesticidally active ingredient, compound, composition, virus, bacterium, antimicrobial or disinfectant) that through its effect deters, incapacitates, kills or otherwise discourages pests.
- Target pests can include insects, plant pathogens, weeds, mollusks, birds, mammals, fish, nematodes (roundworms), and microbes that destroy property, cause nuisance, spread disease or are vectors for disease.
- pesticide includes also plant growth regulators that alter the expected growth, flowering, or reproduction rate of plants; defoliants that cause leaves or other foliage to drop from a plant, usually to facilitate harvest; desiccants that promote drying of living tissues, such as unwanted plant tops; plant activators that activate plant physiology for defense against certain pests; safeners that reduce unwanted herbicidal action of pesticides on crop plants; and plant growth promoters that affect plant physiology e.g. to increase plant growth, biomass, yield or any other quality parameter of the harvestable goods of a crop plant.
- the user applies the composition according to the invention usually from a predosage device, a knapsack sprayer, a spray tank, a spray plane, or an irrigation system.
- the agrochemical PI External composition is made up with water, buffer, and/or further auxiliaries to the desired application concentration and the ready-to-use spray liquor or the agrochemical composition according to the invention is thus obtained.
- 20 to 2000 liters, preferably 50 to 400 liters, of the ready-to-use spray liquor are applied per hectare of agricultural useful area.
- the present invention provides a combination comprising the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and at least one further pesticidally active substance selected from the group consisting of fungicides, insecticides, nematicides, acaricides, biopesticides, herbicides, safeners, plant growth regulators, antibiotics, fertilizers and nutrients.
- the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), the combinations and the compositions thereof comprising them in the use as fungicides with other fungicides may result in an expansion of the fungicidal spectrum of activity being obtained or in a prevention of fungicide resistance development.
- the present invention also relates to agrochemical combinations comprising at least one compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), and at least one further pesticidally active substance selected from the group of fungicides, insecticides, nematicides, acaricides, biopesticides, herbicides, safeners, plant growth regulators, antibiotics, fertiliers and nutrients.
- the pesticidally active substances reported in WO2015185485 pages 36-43 and WO2017093019 pages 42- 56 can be used in conjunction with the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a).
- the active substances referred to as component 2 their preparation and their activity e. g. against harmful fungi is known (cf.: http://www.alanwood.net/pesticides/); these substances are commercially available.
- the compounds described by IUPAC nomenclature, their preparation and their pesticidal activity are also known in prior art.
- the present invention furthermore relates to agrochemical mixtures comprising at least one compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) (component 1) and at least one further active substance useful for plant protection.
- the time between both applications may vary e. g. between 2 hours to 7 days. Also, a broader range is possible ranging from 0.25 hour to 30 days, preferably from 0.5 hour to 14 days, particularly from 1 hour to 7 days or from 1.5 hours to 5 days, even more preferred from 2 hours to 1 day.
- the weight ratio of the component 1) and the component 2) generally depends on the properties of the active components used, usually it is in the range of 1:1000 to 1000:1, often in the range of 1:100 to 100:1, regularly in the range of 1:50 to 50:1, preferably in the range of 1:20 to 20:1, more preferably in the range of 1:10 to 10:1, even more preferably in the range of 1:4 to 4:1 and in particular in the range of 1:2 to 2:1.
- the weight ratio of the component 1) and the component 2) usually is in the range of 1000:1 to 1:1000, often in the range of 100:1 to 1:100, regularly in the range of 50:1 to 1:50, preferably in the range of 20:1 to 1:20, more preferably in the range of 10:1 to 1:10, even more preferably in the range of 4:1 to 1:4 and in particular in the range of 2:1 to 1:2.
- the weight ratio of component 1) and component 2) depend of the properties of the active substances being used.
- the weight ratio of component 1) and component 3) usually is in the range of 1:100 to 100:1, regularly in the range of 1:50 to 50:1, preferably in the range of 1:20 to 20:1, more preferably in the range of 1:10 to 10:1 and in particular in the range of 1:4 to 4:1 and 1:2 to 2:1, and the weight ratio of component 1) and component 3) usually is in the range of 1:100 to 100:1, regularly in the range of 1:50 to 50:1, preferably in the range of 1:20 to 20:1, more preferably in the range of 1:10 to 10:1 and in particular in the range of 1:4 to 4:1 as well as 1:2 to 2:1. Any further active components are, if desired, added in a ratio of 20:1 to 1:20 to the component 1. These ratios are also suitable for inventive mixtures applied by seed treatment.
- kits for preparing a usable pesticidal composition comprising a) a composition comprising component 1) as defined herein and at least one auxiliary; and b) a composition comprising component 2) as defined herein and at least one auxiliary; and optionally c) a composition comprising at least one auxiliary and optionally a further active component 3) as defined herein.
- the combinations and the compositions thereof can be carried out before or during sowing.
- Methods for applying the compounds of formula (I), the combinations and the compositions thereof, respectively, are application onto plant propagation material, especially seeds, including dressing, coating, pelleting, dusting, and soaking as well as in- furrow application methods.
- the compounds of formula (I), the combinations and the compositions thereof, respectively are applied on to the plant propagation material by a method such that germination is not induced, e. g. by seed dressing, pelleting, coating and dusting.
- the present invention provides a method for controlling or preventing infestation of plants by phytopathogenic microorganisms in agricultural crops and/or horticultural crops wherein an effective amount of at least one compound of formula (I) or the combinations or the composition, is applied to the plants, to parts thereof or to a locus thereof.
- the present invention provides a method for controlling or preventing infestation of plants by phytopathogenic microorganisms in agricultural crops and or horticultural crops wherein an effective amount of at least one compound of formula (I) or the combination or the composition, is applied to the seeds of plants.
- the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and the compositions according to the invention, respectively, are suitable as fungicides. They are distinguished by an outstanding effectiveness against a broad spectrum of phytopathogenic fungi, including soil-borne fungi, which derive especially from the classes of the Plasmodiophoromycetes, Peronosporomycetes (syn. Oomycetes), Chytridiomycetes, Zygomycetes, Ascomycetes, Basidiomycetes and Deuteromycetes (syn. Fungi imperfecti).
- Some are systemically active and they can be used in crop protection as foliar fungicides, fungicides for seed dressing and soil fungicides. Moreover, they are suitable for controlling harmful fungi, which inter alia occur in wood or roots of plants.
- the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and the compositions according to the invention are particularly important in the control of a multitude of phytopathogenic fungi on various cultivated plants, such as cereals, e. g. wheat, rye, barley, triticale, oats or rice; beet, e. g. sugar beet or fodder beet; fruits, such as pomes, stone fruits or soft fruits, e.
- the compounds of formula (I), or formula (I-A) to formula (I-F) and the composition according to the invention are important in the control of phytopathogenic fungi on cereals (e. g. wheat, rye, barley, triticale, oats or rice) and soybeans and on the plant propagation material, such as seeds, and the crop material of cereals and soybeans.
- the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and composition thereof, respectively are used for controlling a multitude of fungi on field crops, such as potatoes sugar beets, tobacco, wheat, rye, barley, oats, rice, corn, cotton, soybeans, rape, legumes, sunflowers, coffee or sugar cane; fruits; vines; ornamentals; or vegetables, such as cucumbers, tomatoes, beans or squashes.
- plant propagation material is to be understood to denote all the generative or reproductive parts of the plant such as seeds and vegetative plant material such as cuttings and tubers (e. g. potatoes), which can be used for the multiplication of the plant.
- treatment of plant propagation materials with the compounds of formula (I), or formula (I- A) to formula (I-F) or formula (I-D-a), the combinations and or the compositions thereof, respectively, is used for controlling a multitude of fungi on cereals, such as wheat, rye, barley and oats; on rice, corn, cotton and soybeans.
- cultiva plants is to be understood as including plants which have been modified by breeding, mutagenesis or genetic engineering including but not limiting to agricultural biotech products on the market or in development (cf. http://cera-gmc.org/, see GM crop database therein).
- Genetically modified plants are plants, which genetic material has been so modified by recombinant DNA techniques that otherwise cannot readily be obtained by cross breeding under natural circumstances, mutations or natural recombination.
- one or more genes have been integrated into the genetic material of a genetically modified plant to improve certain properties of the plant.
- Such genetic modifications also include but are not limited to targeted post-translational modification of protein(s), oligo-or polypeptides e. g.
- auxin herbicides such as dicamba or 2,4-D
- bleacher herbicides such as hydroxylphenylpyruvate PI External dioxy
- herbicides e. bromoxynil or ioxynil herbicides as a result of conventional methods of breeding or genetic engineering. Furthermore, plants have been made resistant to multiple classes of herbicides through multiple genetic modifications, such as resistance to both glyphosate and glufosinate or to both glyphosate and a herbicide from another class such as ALS inhibitors, HPPD inhibitors, auxin herbicides, or ACCase inhibitors. These herbicide resistance technologies are e. g. described in Pest Managem.
- Bacillus are particularly from Bacillus thuringiensis, such as ⁇ -endotoxins, e. g. CrylA(b), CrylA(c), CrylF, CrylF(a2), CryllA(b), CrylllA, CrylllB(bl) or Cry9c; vegetative insecticidal proteins (VIP), e. g. VIP1, VIP2, VIP3 or VIP3A; insecticidal proteins of bacteria colonizing nematodes, e. g. Photorhabdus spp.
- ⁇ -endotoxins e. g. CrylA(b), CrylA(c), CrylF, CrylF(a2), CryllA(b), CrylllA, CrylllB(bl) or Cry9c
- VIP vegetative insecticidal proteins
- VIP1, VIP2, VIP3 or VIP3A insecticidal proteins of bacteria colonizing nematodes, e. g. Photorhabdus spp.
- toxins produced by animals such as scorpion toxins, arachnid toxins, wasp toxins, or other insect-specific neurotoxins
- toxins produced by fungi such Streptomycetes toxins, plant lectins, such as pea or barley lectins; agglutinins
- proteinase inhibitors such as trypsin inhibitors, serine protease inhibitors, patatin, cystatin or papain inhibitors
- ribosome-inactivating proteins (RIP) such as ricin, maize-RIP, abrin, luffin, saporin or bryodin
- steroid metabolism enzymes such as 3-hydroxysteroid oxidase, ecdysteroid-IDP-glycosyl-transferase, cholesterol oxidases, ecdysone inhibitors or HMG-CoA-reductase
- ion channel blockers such as blockers of sodium or calcium channels
- these insecticidal proteins or toxins are to be understood expressly also as pre- toxins, hybrid proteins, truncated or otherwise modified proteins.
- Hybrid proteins are characterized by PI External a new combination of protein domains, (see, e. g. WO02/015701).
- Further examples of such toxins or genetically modified plants capable of synthesizing such toxins are disclosed, e. g., in EP374753, WO93/007278, WO95/34656, EP427529, EP451878, WO03/18810 und WO03/52073.
- the methods for producing such genetically modified plants are generally known to the person skilled in the art and are described, e. g. in the publications mentioned above.
- insecticidal proteins contained in the genetically modified plants impart to the plants, producing these proteins, tolerance to harmful pests from all taxonomic groups of arthropods, especially to beetles (Coeloptera), two-winged insects (Diptera), and moths (Lepidoptera) and to nematodes (Nematoda).
- Genetically modified plants capable to synthesize one or more insecticidal proteins are, e.
- WO 03/018810 MON 863 from Monsanto Europe S.A., Belgium (corn cultivars producing the Cry3Bb1 toxin), IPC 531 from Monsanto Europe S.A., Belgium (cotton cultivars producing a modified version of the CrylAc toxin) and 1507 from Pioneer Overseas Corporation, Belgium (corn cultivars producing the Cry1 F toxin and PAT enzyme).
- plants capable to synthesize one or more proteins to increase the resistance or tolerance of those plants to bacterial, viral or fungal pathogens by the use of recombinant DNA techniques are also within the scope of the present invention. Examples of such proteins are the so-called "pathogenesis-related proteins" (PR proteins, see, e. g.
- plant disease resistance genes e. g. potato cultivars, which express resistance genes acting against Phytophthora infestans derived from the Mexican wild potato Solanum bulbocastanum
- T4-lysozym e. g. potato cultivars capable of synthesizing these proteins with increased resistance against bacteria such as Erwinia amylvora
- the methods for producing such genetically modified plants are generally known to the person skilled in the art and are described, e. g. in the publications mentioned above.
- plants capable to synthesize one or more proteins by use of recombinant DNA techniques, to increase the productivity (e. g.
- plants that contain a modified quantity of substances of content or new substances of content, by the use of recombinant DNA techniques, to improve human or animal nutrition e. g. oil crops that produce health-promoting long-chain omega-3 fatty acids or unsaturated omega-9 fatty acids (e. g. Nexera ® rape, DOW Agro Sciences, Canada) are also within the scope of the present invention.
- plants that contain a modified quantity of substances of content or new substances of content by the use of recombinant DNA techniques, to improve raw material production, e. g. potatoes that produce increased amounts of amylopectin (e. g. Amflora ® potato, BASF SE, Germany) are also within the scope of the present invention.
- the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) may be, for example, effective against fungi and fungal vectors of disease as well as phytopathogenic bacteria and viruses.
- fungi and fungal vectors of disease as well as phytopathogenic bacteria and viruses are for example: Absidia corymbifera, Alternaria spp, Aphanomyces spp, Ascochyta spp, Aspergillus spp. including A. flavus, A. fumigatus, A. nidulans, A. niger, A. terms, Aureobasidium spp. including A. pullulans, Blastomyces dermatitidis, Blumeria graminis, Bremia lactucae, Botryosphaeria spp. including B. dothidea, B. obtusa, Botrytis spp. comprising B. cinerea, Candida spp. including C.
- albicans C. glabrata, C. krusei, C. lusitaniae, C. parapsilosis, C. tropicalis, Cephaloascus fragrans, Ceratocystis spp, Cercospora spp. including C. arachidicola, Cercosporidium personatum, Cladosporium spp, Claviceps purpurea, Coccidioides immitis, Cochliobolus spp, Colletotrichum spp. including C.
- M. graminicola including M. graminicola, M. pomi, Oncobasidium theobromaeon, Ophiostoma piceae, Paracoccidioides spp, Penicillium spp. including P. digitatum, P. italicum, Petriellidium spp, Peronosclerospora spp. Including P. maydis, P. philippinensis and P.
- Ustilaginales such as Ustilaginoidea virens, Ustilago nuda, Ustilago tritici, Ustilago zeae, Cerotelium fici, Chrysomyxa arctostaphyli, Coleosporium ipomoeae, Hemileia vastatrix, Puccinia arachidis, Puccinia cacabata, Puccinia graminis, Puccinia sorghi, Puccinia striiformis f.sp. Hordei, Puccinia striiformis f.sp.
- Non-limiting examples of pathogens of fungal diseases which can be treated in accordance with the invention include the diseases caused by rust disease pathogens, for example Gymnosporangium species, for example Gymnosporangium sabinae; Hemileia species, for example Hemileia vastatrix; Phakopsora species, for example Phakopsora pachyrhizi or Phakopsora meibomiae; Puccinia species, for example Puccinia recondita, Puccinia graminis oder Puccinia striiformis; Uromyces species, for example Uromyces appendiculatus.
- rust disease pathogens for example Gymnosporangium species, for example Gymnosporangium sabinae
- Hemileia species for example Hemileia vastatrix
- Phakopsora species for example Phakopsora pachyrhizi or Phakopsora meibomiae
- Cronartium ribicola White pine blister rust
- Gymnosporangium juniperi-virginianae Cedar-apple rust
- Hemileia vastatrix Coffee rust
- Phakopsora meibomiae and P. pachyrhizi Soybean rust
- Puccinia coronata Crown Rust of Oats and Ryegrass
- Puccinia graminis Stetem rust of wheat and Kentucky bluegrass, or black rust of cereals
- Puccinia hemerocallidis Daylily rust
- Puccinia persistens subsp.
- Puccinia sorghi rust in corn
- Puccinia striiformis 'Yellow rust' in cereals
- Uromyces appendiculatus rust of beans
- Uromyces phaseoli Bean rust
- Puccinia melanocephala 'Brown rust' in sugarcane
- Puccinia kuehnii 'Orange rust' in sugarcane
- the present invention further relates to the use of the compounds of formula (I), the combinations or the compositions thereof for controlling or preventing against phytopathogenic fungi such as Septoria spp., Blumeria spp., Podosphaera spp., Sphaerotheca spp., Uncinula spp., Erysiphe spp., Erysiphe spp., Microsphaera diffusa spp., Botrytis spp., Colletotrichum spp, Alternaria spp., Venturia inaequalis spp., and Monilinia spp. of agricultural crops and or horticultural crops.
- phytopathogenic fungi such as Septoria spp., Blumeria spp., Podosphaera spp., Sphaerotheca spp., Uncinula spp., Erysiphe spp., Erysiphe
- the present invention further relates to the use of the compounds of formula (I), the combinations or the compositions thereof for controlling or preventing against phytopathogenic fungi such as Septoria spp., Blumeria spp., Podosphaera spp., Sphaerotheca spp., Uncinula spp., Erysiphe spp., Erysiphe spp., Microsphaera diffusa spp., Botrytis spp., Colletotrichum spp, Alternaria spp., Venturia inaequalis spp., and Monilinia spp. in cereals, grapevines, fruits, nuts and vegetables.
- phytopathogenic fungi such as Septoria spp., Blumeria spp., Podosphaera spp., Sphaerotheca spp., Uncinula spp., Erysiphe spp., Erys
- Plants which can be treated in accordance with the invention include the following: cotton, flax, grapevine, fruits, vegetables, such as Rosaceae sp (for example pome fruits such as apples, pears, apricots, cherries, almonds and peaches), Ribesioidae sp., Juglandaceae sp., Betulaceae sp., Anacardiaceae sp., Fagaceae sp., Moraceae sp., Oleaceae sp., Actinidaceae sp., Lauraceae sp., Musaceae sp. (for example banana trees and plantations), Rubiaceae sp.
- Rosaceae sp for example pome fruits such as apples, pears, apricots, cherries, almonds and peaches
- Ribesioidae sp. Juglandaceae sp.
- Betulaceae sp. Ana
- Theaceae sp. for example coffee
- Theaceae sp. Sterculiceae sp.
- Rutaceae sp. for example lemons, oranges and grapefruit
- Vitaceae sp. for example grapes
- Solanaceae sp. for example tomatoes, peppers
- Liliaceae sp. for example lettuce
- Umbelliferae sp. for example Cruciferae sp., Chenopodiaceae sp.
- Cucurbitaceae sp. for example cucumber
- Alliaceae sp. for example leek, onion
- peas for example peas
- major crop plants such as Poaceae/Gramineae sp.
- Poaceae/Gramineae sp. for example maize, turf, cereals such as wheat, rye, rice, barley, oats, millet and triticale
- Asteraceae sp. for example sunflower
- Brassicaceae sp. for example white cabbage, red cabbage, broccoli, cauliflower, Brussels sprouts, pak choi, kohlrabi, radishes, and oilseed rape, mustard, horseradish and cress
- Fabacae sp. for example bean, peanuts
- Papilionaceae sp. for example soya bean
- phytophthora rot (Phytophthora megasperma), brown stem rot (Phialophora gregata), pythium rot (Pythium aphanidennatum, Pythium irregulare, Pythium debaryanum, Pythium myriotylum, Pythium ultimum), rhizoctonia root rot, stem decay, and damping-off (Rhizoctonia solani), sclerotinia stem decay (Sclerotinia sclerotiorum), sclerotinia southern blight (Sclerotinia rolfsii), thielaviopsis root rot (Thielaviopsis basicola).
- the present invention also relates to the use of the compounds of formula (I), the combinations or the compositions thereof for controlling or preventing the following plant diseases: Puccinia spp. (rusts) on various plants, for example, but not limited to P. triticina (brown or leaf rust), P. striiformis (stripe or yellow rust), P. hordei (dwarf rust), P. graminis (stem or black rust) or P. recondita (brown or leaf rust) on cereals, such as e. g. wheat, barley or rye and Phakopsoraceae spp. on various plants, in particular Phakopsora pachyrhizi and P.
- Puccinia spp. rusts
- rusts rusts
- rusts rusts
- P. triticina brown or leaf rust
- P. striiformis stripe or yellow rust
- P. hordei d
- the compounds, the combinations and the compositions of the present invention can be used for controlling or preventing plant diseases.
- the compounds of formula (I), or formula (I-A) to formula (I- F) or formula (I-D-a), the combinations and/or the compositions thereof, respectively, are particularly suitable for controlling the following plant diseases: Albugo spp. (white rust) on ornamentals, vegetables (e. g. A. Candida) and sunflowers (e. g. A. tragopogonis); Altemaria spp.
- B. zeicola Northern leaf blight
- B. zeicola spot blotch
- C. sorokiniana C. sorokiniana
- cereals e. g. B. oryzae on rice and turfs
- Blumeria originally Erysiphe
- graminis prowdery mildew
- wheat or barley e. g. wheat or barley
- Botrytis cinerea teleomorph: Botryotinia fuckeliana: grey mold
- fruits and berries e. g. strawberries
- vegetables e. g.
- Colletotrichum teleomorph: Glomerella
- spp. anthracnose
- cotton e. g. C. gossypii
- corn e. g. C. graminicola: Anthracnose stalk rot
- soft fruits e. g. C. coccodes: black dot
- beans e. g. C. lindemuthianum
- soybeans e. g. C. truncatum or C. gloeosporioides
- Corticium spp. e. g. C. C.
- sasakii sheath blight
- Corynespora cassiicola leaf spots
- Cycloconium spp. e. g. C. oleaginum on olive trees
- Cylindrocarpon spp. e. g. fruit tree canker or young vine decline, teleomorph: Nectria or Neonectria spp.
- liriodendri Neonectria liriodendri: Black Foot Disease) and ornamentals; Dematophora (teleomorph: Rosellinia) necatrix (root and stem rot) on soybeans; Diaporthe spp., e. g. D. phaseolorum (damping off) on soybeans; Drechslera (syn. Helminthosporium, teleomorph: Pyrenophora) spp. on corn, cereals, such as barley (e. g. D. teres, net blotch) and wheat (e. g. D. D.
- tritici-repentis tan spot), rice and turf; Esca (dieback, apoplexy) on vines, caused by Formitiporia (syn. Phellinus) punctata, F. mediterranea, Phaeomoniella chlamydospora (earlier Phaeoacremonium chlamydosporum), Phaeoacremonium aleophilum and/or Botryosphaeria obtusa; Elsinoe spp. on pome fruits (E. pyri), soft fruits (E. veneta: anthracnose) and vines (E.
- ampelina anthracnose
- Entyloma oryzae leaf smut
- Epicoccum spp. black mold
- Erysiphe spp. potowdery mildew
- sugar beets E. betae
- vegetables e. g. E. pisi
- cucurbits e. g. E. cichoracearum
- cabbages e. g. E. cruciferarum
- Eutypa lata Eutypa canker or dieback, anamorph: Cytosporina lata, syn.
- G. sabinae rust on pears
- Helminthosporium spp. syn. Drechslera, teleomorph: Cochliobolus
- Hemileia spp. e. g. H. vastatrix (coffee leaf rust) on coffee
- Isariopsis clavispora syn. Cladosporium vitis
- Macrophomina phaseolina syn. phaseoli
- root and stem rot on soybeans and cotton
- Microdochium syn. Fusarium
- nivale pink snow mold
- brassicae brassicae
- rape e. g. P. parasitica
- onions e. g. P. destructor
- tobacco P. tabacina
- soybeans e. g. P. manshurica
- Phakopsora pachyrhizi and P. meibomiae staybean rust
- Phialophora spp. e. g. on vines (e. g. P. tracheiphila and P. tetraspora) and soybeans (e. g. P. gregata: stem rot); Phoma lingam (root and stem rot) on rape and cabbage and P.
- betae root rot, leaf spot and damping-off
- Phomopsis spp. on sunflowers, vines (e. g. P. viticola: can and leaf spot) and soybeans (e. g. stem rot: P. phaseoli, teleomorph: Diaporthe phaseolorum); Physoderma maydis (brown spots) on corn; Phytophthora spp. (wilt, root, leaf, fruit and stem root) on various plants, such as paprika and cucurbits (e. g. P. capsici), soybeans (e. g. P. megasperma, syn. P. sojae), soybeans, potatoes and tomatoes (e. g. P.
- Plasmodiophora brassicae club root
- Plasmopara spp. e. g. P. viticola (grapevine downy mildew) on vines and P. halstedii on sunflowers
- Plasmopara spp. e. g. P. viticola (grapevine downy mildew) on vines and P. halstedii on sunflowers
- Podosphaera spp. powdery mildew) on rosaceous plants, hop, pome and soft fruits, e. g. P. leucotricha on apples
- Polymyxa spp. e. g. on cereals, such as barley and wheat (P.
- Pseudocercosporella herpotrichoides eyespot, teleomorph: Tapesia yallundae
- Pseudoperonospora downy mildew
- Pseudopezicula tracheiphila red fire disease or .rotbrenner', anamorph: Phialophora) on vines
- Puccinia spp. rusts
- oryzae (teleomorph: Magnaporthe grisea, rice blast) on rice and P. grisea on turf and cereals; Pythium spp. (damping-off) on turf, rice, corn, wheat, cotton, rape, sunflowers, soybeans, sugar beets, vegetables and various other plants (e. g. P. ultimum or P. aphanidermatum); Ramularia spp., e. g. R. collo-cygni (Ramularia leaf spots, Physiological leaf spots) on barley and R. beticola on sugar beets; Rhizoctonia spp.
- R. solani root and stem rot
- S. solani silk and stem rot
- S. solani silk and stem rot
- S. solani silk blight
- R. cerealis Rhizoctonia spring blight
- Rhizopus stolonifer black mold, soft rot
- Rhynchosporium secalis scald
- Sarocladium oryzae and S. attenuatum sheath rot) on rice
- Sclerotinia spp e. g.
- R. solani root and stem rot
- S. solani silk blight
- R. cerealis Rhizoctonia spring blight
- Rhizopus stolonifer black mold, soft rot
- Rhynchosporium secalis scald
- Sarocladium oryzae and S. attenuatum sheath rot
- Sclerotinia spp Sclerotinia spp.
- seed rot or white mold on vegetables and field crops, such as rape, sunflowers (e. g. S. sclerotiorum) and soybeans (e. g. S. rolfsii or S. sclerotiorum); Septoria spp. on various plants, e. g. S. glycines (brown spot) on PI External soybeans, S. tritici (Septoria blotch) on wheat and S. (syn. Stagonospora) nodorum (Stagonospora blotch) on cereals; Uncinula (syn.
- Erysiphe) necator prowdery mildew, anamorph: Oidium tuckeri
- Setospaeria spp. (leaf blight) on corn (e. g. S. turcicum, syn. Helminthosporium turcicum) and turf; Sphacelotheca spp. (smut) on corn, (e. g. S. reiliana: head smut), sorghum und sugar cane; Sphaerotheca fuliginea (powdery mildew) on cucurbits; Spongospora subterranea (powdery scab) on potatoes and thereby transmitted viral diseases; Stagonospora spp.
- S. nodorum Stagonospora blotch, teleomorph: Leptosphaeria [syn. Phaeosphaeria] nodorum
- wheat Synchytrium endobioticum on potatoes (potato wart disease)
- Taphrina spp. e. g. T. deformans (leaf curl disease) on peaches and T. pruni (plum pocket) on plums
- Thielaviopsis spp. black root rot
- tobacco, pome fruits, vegetables, soybeans and cotton e. g. T. basicola (syn. Chalara elegans); Tilletia spp.
- the compounds of formula (I) have broad ranges of activity against fungal pathogens.
- Exemplary pathogens may include, but are not limited to, causing agent of wheat leaf blotch (Zymoseptoria tritici), wheat brown rust (Puccinia triticina), wheat stripe rust (Puccinia striiformis), scab of apple (Venturia inaequalis), powdery mildew of grapevine (Uncinula necator), barley scald (Rhynchosporium secalis), blast of rice (Pyricularia oryzae), rust of soybean (Phakopsora pachyrhizi), glume blotch of wheat (Leptosphaeria nodorum), powdery mildew of wheat (Blumeria graminis f. sp.
- the exact amount of the active material to be applied is dependent not only on the specific active material being applied, but also on the particular action desired, the fungal species to be controlled, and the stage of growth thereof, as well as the parts of the plant or other products to be contacted with the compound. Thus, all the compounds, and formulations containing the same, may not be equally effective at similar concentrations or against the same fungal species.
- the compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), the combinations and the compositions thereof, respectively, are also suitable for controlling harmful fungi in the protection of stored products or harvest and in the protection of materials.
- protection of PI External materials is to be understood to denote the protection of technical and non-living materials, such as adhesives, glues, wood, paper and paperboard, textiles, leather, paint dispersions, plastics, cooling lubricants, fibre or fabrics, against the infestation and destruction by harmful microorganisms, such as fungi and bacteria.
- Ascomycetes such as Ophiostoma spp., Ceratocystis spp., Aureobasidium pullulans, Sclerophoma spp., Chaetomium spp., Humicola spp., Petriella spp., Trichurus spp.; Basidiomycetes such as Coniophora spp., Coriolus spp., Gloeophyllum spp., Lentinus spp., Pleurotus spp., Pora spp., Serpula spp.
- Ascomycetes such as Ophiostoma spp., Ceratocystis spp., Aureobasidium pullulans, Sclerophoma spp., Chaetomium spp., Humicola spp., Petriella spp., Trichurus spp.
- Basidiomycetes such as Coniophora spp
- the present invention provides a method for controlling or preventing phytopathogenic fungi.
- the method comprises treating the fungi or the materials, plants, plant parts, locus thereof, soil or seeds to be protected against fungal attack, with an effective amount of at least one compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), or a combination or a composition comprising at least one compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a).
- the method of treatment according to the invention can also be used in the field of protecting stored products or harvest against attack of fungi and microorganisms.
- the term "stored products” is understood to denote natural substances of plant or animal origin and their processed forms, which have been taken from the natural life cycle and for which long-term protection is desired.
- Stored products of crop plant origin such as plants or parts thereof, for example stalks, leafs, tubers, seeds, fruits or grains, can be protected in the freshly harvested state or in processed form, such as pre-dried, moistened, comminuted, ground, pressed or roasted, which process is also known as post- harvest treatment.
- timber whether in the form of crude timber, such as construction timber, electricity pylons and barriers, or in the form of finished articles, such as furniture or objects made from wood.
- Stored products of animal origin are hides, leather, furs, hairs and the like.
- the combinations according to the present invention can prevent disadvantageous effects such as decay, discoloration or mold.
- stored products is understood to denote natural substances of plant origin and their processed forms, more preferably fruits and their processed forms, such as pomes, stone fruits, soft fruits and citrus fruits and their processed forms.
- PI External It is also possible to use the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I- D-a) as a fungicide.
- fungicide as used herein means a compound that controls, modifies, or prevents the growth of fungi.
- the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), the combinations and the compositions thereof, respectively, may be used for improving the health of a plant.
- the invention also relates to a method for improving plant health by treating a plant, its propagation material and/or the locus where the plant is growing or is to grow with an effective amount of compound of formula (I), or formula (I-A) to formula (I-F) or formual (I-D-a) and the composition thereof, respectively.
- plant health is to be understood to denote a condition of the plant and/or its products which is determined by several indicators alone or in combination with each other such as yield (e. g.
- the above identified indicators for the health condition of a plant may be interdependent or may result from each other.
- the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) are employed as such or in the form of a composition for treating the fungi or the plants, plant propagation materials, such as seeds, soil, surfaces, materials or rooms to be protected from fungal attack with a fungicidally effective amount of the active substances.
- the application can be carried out both before and after the infection of the plants, plant propagation materials, such as seeds, soil, surfaces, materials, or rooms by the fungi.
- Plant propagation material may be treated with the compounds of formula (I), the combinations and the compositions thereof protectively either at or before planting or transplanting.
- the compounds of the invention can be used in combination with models e.g. embedded in computer programs for site specific crop management, satellite farming, precision farming or precision agriculture.
- models support the site specific management of agricultural sites with data from various sources such as soils, weather, crops (e.g. type, growth stage, plant health), weeds (e.g. type, growth stage), diseases, pests, nutrients, water, moisture, biomass, satellite data, yield etc. with the purpose to optimize profitability, sustainability and protection of the environment.
- the compounds of the invention can be applied to a crop plant according to an appropriate dose regime if a model models the development of a pest and calculates that a threshold has been reached for which it is recommendable to apply the compound of the invention to the crop plant.
- PI External Commercially available systems which include agronomic models are e.g. FieldScriptsTM from The climate Corporation, XarvioTM from BASF, AGLogicTM from John Deere, etc.
- the compounds of the invention can also be used in combination with smart spraying equipment such as e.g.
- Such an equipment usually includes input sensors (such as e.g. a camera) and a processing unit configured to analyze the input data and configured to provide a decision based on the analysis of the input data to apply the compound of the invention to the crop plants (respectively the weeds) in a specific and precise manner.
- input sensors such as e.g. a camera
- processing unit configured to analyze the input data and configured to provide a decision based on the analysis of the input data to apply the compound of the invention to the crop plants (respectively the weeds) in a specific and precise manner.
- the use of such smart spraying equipment usually also requires positions systems (e.g. GPS receivers) to localize recorded data and to guide or to control farm vehicles; geographic information systems (GIS) to represent the information on intelligible maps, and appropriate farm vehicles to perform the required farm action such as the spraying.
- GPS geographic information systems
- pests can be detected from imagery acquired by a camera.
- the pests can be identified and/or classified based on that imagery.
- image processing algorithms can utilize machine learning algorithms, such as trained neutral networks, decision trees and utilize artificial intelligence algorithms.
- machine learning algorithms such as trained neutral networks, decision trees and utilize artificial intelligence algorithms.
- the bicyclic amine compounds such as 4- (1-methyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine, 4-(1,5-dimethyl-1H-pyrazol-4- yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine, and 7-methyl-4-(1-methyl-1H-pyrazol-4-yl)-4,5,6,7- tetrahydrothieno[2,3-c]pyridine are prepared according to Scheme-7 and Scheme-8, by adopting the procedure described in Example 6 and Example 7. All other starting materials, reagents and solvents are purchased from commercial sources.
- Example 1 Synthesis of 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylic acid
- Step 1 Synthesis of ethyl 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylate PI
- An solution of 1-azido-2,4-difluorobenzene (2 g, 12.9 mmol) and ethyl propiolate (1.9 g, 19.3 mmol) in tetrahydrofuran (20 mL) was treated with copper (I) iodide (0.25 g, 1.3 mmol) and stirred at 25 °C for 12 h.
- Step 2 Synthesis of 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylic acid
- a solution of ethyl 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylate (3 g, 11.8 mmol) in tetrahydrofuran (21 mL) and water (9 mL) was treated with lithium hydroxide monohydrate (1.5 g, 35.5 mmol), and stirred at 25 °C for 3h. After completion of the reaction, tetrahydrofuran was evaporated under reduced pressure, and pH of the residual material was adjusted to 5 - 6 with 10% aqueous hydrochloric acid.
- Example 2 Synthesis of 1-(4-fluorophenyl)-1H-1,2,4-triazole-3-carboxylic acid
- Step 1 Synthesis of ethyl 1-(4-fluorophenyl)-1H-1,2,4-triazole-3-carboxylate
- a solution of 4-fluoroaniline (8 g, 72 mmol) in water (80 mL) was treated sequentially with 10% hydrochloric acid (27 mL) and sodium nitrite (7.45 g, 108 mmol) at 0 °C, and stirred for 30 minutes to obtain the corresponding diazonium salt.
- a solution of sodium acetate (5.9 g, 72 mmol) in water (30 mL) and methanol (80 mL) was treated dropwise with ethyl isocyanoacetate (8.14 g, 72 mmol) at 0 °C, stirred for 5 minutes, and then treated slowly with the solution of the diazonium salt (
- Step 2 Synthesis of 1-(2,4-difluorophenyl)-1H-1,2,4-triazole-3-carboxylic acid
- ethyl 1-(4-fluorophenyl)-1H-1,2,4-triazole-3-carboxylate (1 g, 4.5 mmol) in a mixture of tetrahydrofuran (20 mL), water (10 mL) and methanol (2 mL) was treated with lithium hydroxide (0.54 g, 22.6 mmol) at 25 °C, and stirred for 16 h. After completion of the reaction, the solvents were evaporated under reduced pressure.
- Example 3 Synthesis of 1-(2,4-difluorophenyl)-1H-pyrazole-4-carboxylic acid
- Step 1 Synthesis of ethyl 1-(2,4-difluorophenyl)-1H-pyrazole-4-carboxylate
- ethanol 20 mL
- ethyl 2-formyl-3-oxopropanoate 1 g, 7.2 mmol
- sodium acetate (0.45 g, 5.54 mmol
- Step 2 Synthesis of 1-(2,4-difluorophenyl)-1H-pyrazole-4-carboxylic acid
- ethyl 1-(2,4-difluorophenyl)-1H-pyrazole-4-carboxylate (1 g, 3.96 mmol) in tetrahydrofuran (10 mL) and water (5 mL) was treated with lithium hydroxide monohydrate (0.25 g, 5.95 mmol) at 25 °C, and stirred for 18 h. After completion of the reaction, volatiles were evaporated under reduced pressure, and the residue was acidified with 10% aqueous hydrochloric acid.
- reaction mixture was evaporated under reduced pressure to remove ethanol, and the resulting residual material was dissolved in pyridine (30 mL), cooled to 0 °C.
- the diazonium salt solution (prepared above) was added slowly to this cold reaction mixture, warmed up to 25 °C, and stirred for 3 h.
- the reaction mixture was diluted with water (60 mL) and extracted with ethyl acetate (2 x 50 mL). The combined organic PI External extracts were washed with saturated brine solution (30 mL), dried over sodium sulphate and evaporated under reduced pressure.
- Step 2 Synthesis of 2-(2,4-difluorophenyl)-2H-tetrazole-5-carboxylic acid
- a solution of ethyl 2-(2,4-difluorophenyl)-2H-tetrazole-5-carboxylate (0.6 g, 2.36 mmol) in 1:1:1 mixture of ethanol, tetrahydrofuran and water (6 mL) was treated with LiOH (226 mg, 9.44 mmol) at 25 °C, and stirred for 16 h. After completion of the reaction, the volatiles were removed under reduced pressure and the residual material was acidified with 10% aqueous hydrochloric acid solution to pH 4.
- Step 2 Synthesis of ethyl (2E)-2-(2-(2,4-difluorophenyl)hydrazineylidene)-3-(hydroxyimino) propanoate
- ethyl (E)-2-(2-(2,4-difluorophenyl)hydrazineylidene)-3-oxopropanoate (18 g, 70 mmol) in ethanol (150 mL) was treated with hydroxylamine hydrochloride (5.86 g, 84 mmol) and potassium acetate (17.24 g, 176 mmol) at 25 °C, heated to 80 °C, and stirred for 30 min.
- reaction mixture was diluted with ethyl acetate (500 mL), washed with water (150 mL) and saturated brine solution (100 mL). The separated organic layer was dried over anhydrous sodium sulphate, filtered and evaporated to isolate crude ethyl (2E)-2-(2-(2,4- difluorophenyl)hydrazineylidene)-3-(hydroxyimino)propanoate (16 g, 59 mmol, 84% yield).
- Step 3 Synthesis of ethyl 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4-carboxylate PI External A solution of ethyl (2E)-2-(2-(2,4-difluorophenyl)hydrazineylidene)-3-(hydroxyimino) propanoate (1 g, 3.69 mmol) in acetic anhydride (10 mL) was heated to 140 °C, and stirred for 30 minutes. After completion of the reaction, the reaction mixture was cooled to 25 °C and diluted with water (50 mL). The resulting dark solution was extracted with ethyl acetate (3 x 30 mL).
- Step 4 Synthesis of 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4-carboxylic acid
- a solution of ethyl 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4-carboxylate (9 g, 35.5 mmol) in tetrahydrofuran (100 mL) and water (10 mL) was treated with lithium hydroxide (1.49 g, 35.5 mmol) at 25 °C, and stirred for 2 h. After completion of the reaction, tetrahydrofuran was evaporated under reduced pressure and the residue was acidified with 1M aqueous hydrochloric acid to pH 2.
- Example 6 Synthesis of 4-(1-methyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine
- Step a Synthesis of ethyl 4-bromothieno[2,3-c]pyridine-2-carboxylate
- K2CO3 5 g, 36.2 mmol
- ethyl thioglycolate 3.64 mL, 33.2 mmol
- reaction mixture was poured into water (200 mL) and extracted with ethyl acetate (2 x 100 mL). The combined organic layers were washed with brine solution (100 mL), dried over anhydrous sodium sulphate and evaporated under reduced PI External pressure. The residue was purified by flash column chromatography to isolate ethyl 4-bromothieno[2,3- c]pyridine-2-carboxylate (7.6 g, 26.6 mmol, 88% yield).
- reaction mixture was cooled to 25 °C, treated with methanol (200 mL) and stirred for 30 min. The volatiles were evaporated under reduced pressure. The residue obtained was dissolved in 1: 1 water: dioxane (50 mL) and then treated with NaOH (4.64 g, 119 mmol) at 25° C followed by stirring at 90 °C for 16 h. After completion of the reaction, the reaction mixture was cooled to 25° C and extracted with ethyl acetate (2 x 100 mL).
- Example 7 Synthesis of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3- c]pyridine (Formula 9A-b)
- Step f Synthesis of 4-bromothieno[2,3-c]pyridine-2-carboxylic acid To a suspension of ethyl 4-bromothieno[2,3-c]pyridine-2-carboxylate (11 g, 38.4 mmol, prepared according to step-1 of Example 6) in tetrahydrofuran (200 mL) and water (50 mL), LiOH (1.381 g, 57.7 mmol) was added and stirred at 25 °C for 5 h.
- Step g Synthesis of 4-bromothieno[2,3-c]pyridine
- 4-bromothieno[2,3-c]pyridine-2-carboxylic acid 5 g, 19.37 mmol
- silver carbonate 2.67 g, 9.69 mmol
- acetic acid 0.06 mL, 0.97 mmol
- the reaction mixture was then stirred at 150 °C for 16 h.
- the reaction mixture was filtered through celite bed. The filtrate was diluted with water (100 mL) and extracted with ethyl acetate (3 ⁇ 100 mL).
- reaction mixture was cooled to 25 °C and treated with methanol (20 mL) and stirred for 30-45 min.
- the reaction mixture was concentrated under reduced pressure to obtain a residue which was dissolved in 1:1 mixture of dixoxane:water (50 mL) and treated with NaOH (1.83 g, 45.8 mmol) at 25 °C and stirred at 90 °C for 16 h.
- the reaction mixture was cooled to 25 °C and extracted with dichloromethane (2 x 50 mL).
- Example 8 Synthesis of 1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3-triazole-4-carboxylic acid
- Step 1 Synthesis of ethyl 1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3-triazole-4-carboxylate
- TMS-N 3 8.25 mL, 62.1 mmol
- tert-butyl nitrite 7.4 mL, 62.1 mmol
- reaction mixture was warmed up to 25 °C, diluted with dimethylsulfoxide (30 mL) and water (10 mL), treated with ethyl acetoacetate (10.3 mL, 79 mmol), piperidine (2.24 mL, 22.6 mmol) and potassium carbonate (781 mg, 5.65 mmol) and stirred for 5 minutes.
- the resulting reaction mixture was heated to 80 °C, stirred for 16 h.
- the reaction mixture was cooled to 0 °C followed by addition of ice cooled water (20 mL) under stirring.
- Step 2 Synthesis of 1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3-triazole-4-carboxylic acid
- a solution of ethyl 1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3-triazole-4-carboxylate (500 mg, 1.79 mmol) in a mixture of tetrahydrofuran (8 mL), methanol (4 mL) and water (2 mL) was treated with LiOH (86 mg, 3.57 mmol) at 25 °C, and stirred for 2 h.
- Example 9 Synthesis of (1-(2,4-difluorophenyl)-1H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol- 4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 1)
- a solution of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-1,2,3,4-tetrahydroisoquinoline hydrochloride (633 mg, 2.4 mmol) and 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylic acid (450 mg, 2 mmol) in dichloromethane (20 mL) was treated sequentially with triethyl amine (1.1 mL, 8 mmol) and 1- propanephosphonic acid cyclic anhydride (T3P, 2.4 mL, 4 mmol) at 25°C, and stirred for 18 h.
- reaction mixture was diluted with water (50 mL) and extracted with ethyl acetate (3 x 40 mL). The combined organic layers were washed with brine solution (40 mL), dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure.
- the crude compound thus obtained was purified by a reverse phase preparative-high performance liquid chromatography to isolate (1-(2,4-difluorophenyl)-1H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)methanone 1 (660 mg, 1.52 mmol, 76% yield) as a beige solid.
- reaction mixture was diluted with water (60 mL) and extracted with dichloromethane (3 x 80 mL). The combined organic extracts were washed with brine solution (40 mL), PI External dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure.
- the crude material thus obtained was purified by a reverse phase preparative-high performance liquid chromatography to isolate (4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)(1-(4- fluorophenyl)-1H-1,2,4-triazol-3-yl)methanone 2 (250 mg, 0.6 mmol, 36% yield) as a white solid.
- reaction mixture was diluted with water (60 mL) and extracted with dichloromethane (3 x 40 mL). The combined organic extracts were washed with brine solution (30 mL), dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure to obtain a crude material.
- Example 12 Synthesis of (2-(2,4-difluorophenyl)-2H-tetrazol-5-yl)(4-(1,5-dimethyl-1H-pyrazol-4- yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 4) PI External A solution of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-1,2,3,4-tetrahydroisoquinoline hydrochloride (583 mg, 2.21 mmol) and 2-(2,4-difluorophenyl)-2H-tetrazole-5-carboxylic acid (500 mg, 2.21 mmol) in dichloromethane (20 mL) was treated sequentially with triethyl amine (0.9 mL, 6.63 mmol) and 1- propanephosphonic acid cyclic anhydride (2.6 mL, 4.42 mmol) at 0 °C, warmed up to 25 °C, and
- reaction mixture was diluted with water (60 mL) and extracted with dichloromethane (3 x 80 mL). The combined organic extracts were washed with saturated brine solution (30 mL), dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure to obtain a crude material.
- reaction mixture was diluted with water (50 mL) and extracted with dichloromethane (3 x 50 mL). The combined organic extracts were washed with saturated brine solution (50 mL), dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure to obtain a crude material.
- reaction mixture was diluted with water (30 mL) and extracted with dichloromethane (2 x 30 mL). The combined organic layer was washed with brine solution (30 mL); dried over sodium sulphate and evaporated under reduced pressure. The residue was purified by reverse phase preparative HPLC method to afford (2-(3-fluorophenyl)-2H- tetrazol-5-yl)(4-(1-methyl-1H-pyrazol-4-yl)-4,7-dihydrothieno[2,3-c]pyridin-6(5H)-yl)methanone (0.12 g, 0.27 mmol, 24% yield) as off white solid.
- Example 20 Synthesis of (4-(1,5-dimethyl-1H-pyrazol-4-yl)-4,7-dihydrothieno[2,3-c]pyridin-6(5H)- yl)(2-(3-fluorophenyl)-2H-tetrazol-5-yl)methanone (Compound 79)
- a suspension of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine (0.25 g, 1.07 mmol) 2-(3-fluorophenyl)-2H-tetrazole-5-carboxylic acid (0.22 g, 1.07 mmol) in dichloromethane (8 mL) was treated with trimethylamine (0.3 mL, 2.14 mmol) and propanephosphonic acid anhydride (0.71 mL, 1.18 mmol) at 0 °C and then stirred at 25 °C for 6 h.
- the compounds of the present invention were assessed for their activity as described in the following tests: PI External Example 1: Pyricularia oryzae (Rice blast): The compounds were dissolved in 0.3% dimethyl sulfoxide and then added to potato dextrose agar medium just prior to dispensing it into petri dishes. 5 mL medium, with the compound in the desired test concentration, was dispensed into a 60 mm sterile petri-plate. After solidification, each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate of Pyricularia oryzae.
- the plates were incubated in growth chambers at 25 o C temperature and 60% relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control.
- Example 2 Alternaria solani (early blight of tomato/potato): The compounds were dissolved in 0.3% dimethyl sulfoxide and then added to potato dextrose agar medium just prior to dispensing it into petri dishes. 5 mL medium, with the compound in the desired concentration, was dispensed into a 60 mm sterile petri-plate. After solidification, each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate of Alternaria solani. The plates were incubated in growth chambers at 25 o C temperature and 60% relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control.
- Example 3 Colletotrichum capsici (anthracnose): The compounds were dissolved in 0.3% dimethyl sulfoxide and then added to potato dextrose agar medium just prior to dispensing it into petri dishes. 5 mL medium, with the compound in the desired concentration, was dispensed into a 60 mm sterile petri-plate. After solidification, each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate of Colletotrichum capsici. The plates were incubated in growth chambers at 25 o C temperature and 60% PI External relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control.
- Example 4 Corynespora cassiicola (Leaf spot of tomato): The compounds were dissolved in 0.3% dimethyl sulfoxide and then added to potato dextrose agar medium just prior to dispensing it into petri dishes. 5 mL medium, with the compound in the desired concentration, was dispensed into a 60 mm sterile petri-plate. After solidification, each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate of Corynespora cassiicola. The plates were incubated in growth chambers at 25 o C temperature and 70% relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control.
- Botrytis cinerea Liquid testing Yeast, bacterial peptone and sodium acetate (YBA) liquid medium containing a BOTRCI (10 4 spores/mL) spore suspension was prepared.
- YBA sodium acetate
- each test compound was solved in dimethyl sulfoxide and 100 ⁇ l of the respective test solution was given into a well of a 96-well microtiter PI External plate, consequently, the same volume (100 ⁇ l) of the media and spore suspension was added to each well to obtain the final test concentration.
- the plates were incubated at 22 °C for 15-18 days. The growth inhibition was evaluated by measuring the OD600.
- 5ml medium with compound in the desired concentration was dispensed into 60mm sterile petri-plates. After solidification each plate was seeded with a 5mm size mycelial disc taken from the periphery of actively growing virulent culture plates. Plates were incubated in growth chambers at 25°C temperature and 60% relative humidity for seven days and radial growth was measured.
- Rhizoctonia solani (Rice sheath blight/Potato black scurf): Compounds were dissolved in 0.3% dimethyl sulfoxide and then added to potato dextrose agar medium just prior to dispensing it into petri dishes. 5 mL medium with a compound in the desired test concentration was dispensed into 60 mm sterile petri-plates. After solidification each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate. Plates were incubated in growth chambers at 25 °C temperature and 60% relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control.
- Example 10 Septoria tritici Potato dextrose (PDB) liquid medium (Difco) containing a Septoria tritici (105 spores/mL) spore suspension was prepared.
- PDB Potato dextrose
- each test compound was dissolved in dimethyl sulfoxide.100 ⁇ L of the test media-solution was added to a 96-well microtiter plate, consequently, the same volume (100 ⁇ L) of spore suspension was added to the well making the final test concentrations and the plate was incubated at 18 °C for 15 to 18 days.
- pycnidiospores were measured under a microscope using software for image acquisition and analysis. Then, for each condition of the test, a mean length of pycnidiospores is calculated by averaging the size of pycnidiospores.
- C is the mean length of pycnidiospores obtained for the control condition (untreated)
- T is the mean length of pycnidiospores for conditions treated with the specific concentration (c) of the fungicide.
- Example B Pseudoperonospora cubensis on cucumber plants The compounds were dissolved in 2% dimethyl sulfoxide/acetone and then mixed with water containing emulsifier to the desired test concentration in a calibrated spray volume of 30ml.
- test solutions were poured into the spray bottles for further applications.
- healthy young cucumber plants raised in the greenhouse were sprayed with the active compound preparation at the stated application rates inside the spray cabinets using hallow cone nozzles.
- the plants were inoculated with a conidial spore suspension containing 2x104Pseudoperonospora cubensis inoculum.
- the inoculated plants were then kept in a greenhouse chamber at 230C temperature & 80-90 % relative humidity for disease expression.
- a visual assessment of the performance of the compounds was carried out by rating the disease severity (0-100% scale) on treated plants 3, 7, 10 and 15 days after application.
- Efficacy (% control) of the compounds was calculated by comparing the disease rating in the treatment with the one of the untreated control. The compounds were also assessed for their plant compatibility by recording symptoms like necrosis, chlorosis and stunting. Compound 21 at 500 ppm gave more than or equal to 70% control in these tests when compared to the untreated check which showed extensive disease development.
- Example C Septoria nodorum test in wheat The single compounds were dissolved in 2% DMSO/ Acetone and then mixed with water containing an emulsifier to a calibrated spray volume of 30 mL. The spray solutions were poured into spray bottles for further applications.
- Efficacy (% control) of the compounds and composition was calculated by comparing the disease rating in the treatment with the one of the untreated, inoculated control plants. The compounds and the respective compositions were also assessed for their plant compatibility by recording symptoms like necrosis, chlorosis & stunting. Compounds 1, 31, 33, 39, 86, 87, 90, 92, 103, 105, 109, 110, 111, 112, 114, 115, 116, 118 at 500 ppm gave more than or equal to 70% control in these tests when compared to the untreated check which showed extensive disease development.
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical & Material Sciences (AREA)
- Dentistry (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Plant Pathology (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Pest Control & Pesticides (AREA)
- Agronomy & Crop Science (AREA)
- Plural Heterocyclic Compounds (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
Abstract
The present invention relates to a compound of formula (I), wherein B, D, R1a, R2, R3, R4a, R4b, R10 and Z1-Z3 are as described in the detailed description and to a process for preparing the compound of formula (I). The present invention also relates to a method for combating phytopathogenic fungi.
Description
Title: BICYCLIC HETEROCYCLIC AMIDES FOR COMBATING PHYTOPATHOGENIC FUNGI FIELD OF THE INVENTION: The present invention relates to novel bicyclic heterocyclic amide compounds of formula (I) which are useful in combating phytopathogenic fungi, and to a process for preparing these novel bicyclic heterocyclic amide compounds of formula (I). The present invention also relates to compositions and to combinations comprising these novel bicyclic heterocyclic amide compounds of formula (I) and to a method for combating phytopathogenic fungi using the same. BACKGROUND OF THE INVENTION: Bicyclic heterocyclic amide compounds are described as fungicidal agents in WO2022253645A1 and WO2023110869A1. The bicyclic heterocyclic amide compounds reported in the above cited literature have disadvantages in certain aspects, such as that they exhibit a narrow spectrum of efficacy or that they do not have a satisfactory fungicidal activity, particularly at low application rates. Therefore, there remains a need for the development of new fungicidal compounds, including such that are belonging to the class of the above cited bicyclic heterocyclic amide compounds, to provide compounds being effective against a broader spectrum of fungi, having a lower toxicity, a higher selectivity, and being used at lower dosage rates to reduce or avoid unfavorable environmental or toxicological effects whilst still allowing an effective and long-lasting control of said fungi. Therefore, it is an objective of the present invention to provide compounds having an improved/enhanced activity and/or a broader efficacy spectrum against phytopathogenic fungi. This objective is achieved by using a compound of formula (I) of the present invention for combating phytopathogenic fungi. SUMMARY OF THE INVENTION: The present invention relates to a compound of formula (I) or an agriculturally acceptable salt, N- oxide, isomer, tautomer or polymorph thereof,
wherein, D is selected from the group consisting of phenyl or 5- or 6-membered heteroaryl ring, wherein said 5- or 6-membered heteroaryl ring comprises 1, 2, 3 or 4 heteroatoms each independently selected from N, O, and S(O)0-2, and wherein said phenyl or 5- or 6-membered heteroaryl ring may be further substituted with one or more different or identical substituents selected from R1b and/or R1c; R1a is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, nitro, C1-C6- alkyl, C1-C6-haloalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C3- C8-cycloalkyl, C3-C8-halocycloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C2-C6-alkenyloxy, C2-C6- haloalkenyloxy, C2-C6-alkynyloxy, C2-C6-haloalkynyloxy, C3-C6-cycloalkyloxy, C1-C6- alkylthio, C1-C6-haloalkylthio, C3-C6-cycloalkylthio,
C6-alkylsulphinyl, C1-C6- haloalkylsulphinyl, C1-C6-alkylsulfonyl, C1-C6-haloalkylsulfonyl, -N(R7)2, -CO-N(R9)2, - S(O)(R8)=NR9, -N=S(O)(R8)2, P(O)(R8)2, Si(R8)3 or phenyl which may be optionally substituted with one or more different or identical substituents selected from halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy or C1-C6-haloalkoxy; R1b is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, C1-C6-alkyl, C1- C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy and C1-C6-haloalkoxy; R1c is selected from the group consisting of hydrogen, halogen, C1-C6-alkyl, C1-C6-haloalkyl, C3- C8-cycloalkyl, hydroxyl and cyano; R2 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R3 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C1-C6-cyanoalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6- haloalkynyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy;
R4a and R4b are each independently selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; or R4a and R4b may together form an oxo group (=O), or a 3- to 6-membered carbocyclic ring; Z1, Z2 and Z3 are each independently selected from CR5 or N; R5 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl and C1-C6-alkoxy; B represents a phenyl ring or a 5- or 6-membered aromatic heterocyclic ring fused at the C3 and C4 carbons of the piperidine ring, preferably B represents a 5-membered aromatic heterocyclic ring fused at the C3 and C4 carbons of the piperidine ring, wherein the 5- or 6-membered aromatic heterocyclic ring comprises 1 to 3 heteroatoms selected from oxygen, nitrogen or S(O)0-2, wherein the ring B is unsubstituted or substituted with one or more different or identical substituents selected from R6; R6 can be selected from the group consisting of hydrogen, halogen, cyano, hydroxyl, C1-C6- alkyl, C1-C6-haloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C3-C8-cycloalkyl, C3-C8- halocycloalkyl, C6-C10-aryl which may be substituted with 1 or 2 same or different substitutent selected from the group consisting of halogen, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6-alkoxy and C1-C6-haloalkoxy, or -N(R7)2; R7 represents hydrogen, C1-C6-alkyl, C3-C8-cycloalkyl, C1-C6-alkylcarbonyl, C3-C8- cycloalkylcarbonyl, C1-C6-alkoxycarbonyl or C1-C6-alkylsulfonyl; R8 is selected from the group consisting of hydrogen, halogen, C1-C6-alkyl, C1-C6-haloalkyl, C2- C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C3-C8-cycloalkyl, C1-C6- alkoxy and C1-C6-haloalkoxy; R9 is selected from the group consisting of hydrogen, C1-C6-alkyl, C1-C6-haloalkyl and C3-C8- cycloalkyl; R10 is selected from the group consisting of hydrogen and C1-C6-alkyl; R1a and R1b, optionally together along with the phenyl or 5- or 6-membered heteroaryl ring, may form a 8- to 10-membered bicyclic carbocyclic or heterocyclic ring system that can be optionally substituted with one or more different or identical substituents selected from R1b and/or R1c, wherein the bicyclic heterocyclic ring system comprises one or more heteroatoms selected from nitrogen, oxygen or S(O)0-2, and wherein one or more carbon atom of the bicyclic carbocyclic or heterocyclic ring system may be replaced with C(O).
The present invention also relates to a process for preparing the compound of formula (I). The compounds of formula (I) have been found to be advantageous over the compounds reported in the literature in either of improved fungicidal activity, broader spectrum of biological efficacy, lower application rates, more favourable biological and/or environmental properties, or enhanced plant compatibility. The present invention further relates to agrochemical compositions comprising a compound of formula (I) or a compound of formula (I) in combination with one or more further pesticidally active substance(s) for controlling and/or preventing plant diseases, particularly caused by phytopathogenic fungi. The present invention still further relates to a method for controlling or preventing an infestation of useful plants by phytopathogenic microorganisms, wherein a fungicidally effective amount of a compound of formula (I), a composition or a combination thereof, is applied to the plants, to parts thereof or the locus thereof. DETAILED DESCRIPTION OF THE INVENTION: DEFINITIONS: The definitions provided herein for the terminologies used in the present disclosure are for illustrative purpose only and in no manner limit the scope of the present invention disclosed in the present disclosure. As used herein, the terms “comprises”, “comprising”, “includes”, “including”, “has”, “having”, “contains”, “containing”, “characterized by” or any other variation thereof, are intended to cover a non-exclusive inclusion, subject to any limitation explicitly indicated. For example, a composition, mixture, process or method that comprises a list of elements is not necessarily limited to only those elements but may include other elements not expressly listed or inherent to such composition, mixture, process or method. The transitional phrase “consisting of” excludes any element, step or ingredient not specified. If in the claim, such would close the claim to the inclusion of materials other than those recited except for impurities ordinarily associated therewith. When the phrase “consisting of” appears in a clause of the body of a claim, rather than immediately following the preamble, it limits only the element set forth in that clause; other elements are not excluded from the claim as a whole.
The transitional phrase “consisting essentially of” is used to define a composition or method that includes materials, steps, features, components or elements, in addition to those literally disclosed, provided that these additional materials, steps, features, components or elements do not materially affect the basic and novel characteristic(s) of the claimed invention. The term “consisting essentially of” occupies a middle ground between “comprising” and “consisting of”. Further, unless expressly stated to the contrary, “or” refers to an inclusive “or” and not to an exclusive “or”. For example, a condition A “or” B is satisfied by any one of the following: A is true (or present) and B is false (or not present), A is false (or not present) and B is true (or present), and both A and B are true (or present). Also, the indefinite articles “a” and “an” preceding an element or component of the present invention are intended to be nonrestrictive regarding the number of instances (i.e. occurrences) of the element or component. Therefore “a” or “an” should be read to include one or at least one, and the singular word form of the element or component also includes the plural unless the number is obviously meant to be singular. As referred to in this disclosure, the term “invertebrate pests” includes but is not limited to fungi, stramenopiles (oomycetes), bacteria, nematodes, mites, ticks, insects and nematodes of economic importance. In the context of this disclosure “pest control” means inhibition of invertebrate pest development (including necrosis, retarded growth and/or death), and related expressions are defined analogously. The term “agronomic” refers to the production of field crops such as for food, feed and fiber and includes the growth of corn, soybeans and other legumes, rice, cereal (e.g., wheat, oats, barley, rye, rice, maize), leafy vegetables (e.g., lettuce, cabbage, and other cole crops), fruiting vegetables (e.g., tomatoes, pepper, eggplant, crucifers and cucurbits), potatoes, sweet potatoes, grapes, cotton, tree fruits (e.g., pome, stone and citrus), small fruit (berries, cherries) and other specialty crops (e.g., canola, sunflower, olives). The term “nonagronomic” refers to other than field crops, such as horticultural crops (e.g., greenhouse, nursery or ornamental plants not grown in a field), residential, agricultural, commercial and industrial structures, turf (e.g., sod farm, pasture, golf course, lawn, sports field, etc.), wood products, stored product, agro-forestry and vegetation management, public health (i.e. human) and animal health (e.g., domesticated animals such as pets, livestock and poultry, undomesticated animals such as wildlife) applications.
Nonagronomic applications include protecting an animal from an invertebrate parasitic pest by administering a parasiticidally effective (i.e. biologically effective) amount of a compound of the present invention, typically in the form of a composition formulated for veterinary use, to the animal to be protected. As referred to in the present disclosure and claims, the terms “parasiticidal” and “parasiticidally” refers to observable effects on an invertebrate parasite pest to provide protection of an animal from the pest. Parasiticidal effects typically relate to diminishing the occurrence or activity of the target invertebrate parasitic pest. Such effects on the pest include necrosis, death, retarded growth, diminished mobility or lessened ability to remain on or in the host animal, reduced feeding and inhibition of reproduction. These effects on invertebrate parasite pests provide control (including prevention, reduction or elimination) of parasitic infestation or infection of the animal. The meaning of various terms used in the description shall now be illustrated. The term “C1-C6 alkyl”, used either alone or in compound words such as “alkylthio” or “haloalkyl” refers to a straight-chain or branched C1 to C6 alkyl. Non-limiting examples of “C1-C6 alkyl” include methyl, ethyl, propyl, 1-methylethyl, butyl, 1-methylpropyl, 2-methylpropyl, 1,1-dimethylethyl, pentyl, 1-methylbutyl, 2-methylbutyl, 3-methylbutyl, 2,2-dimethylpropyl, 1-ethylpropyl, hexyl, 1,1- dimethylpropyl, 1,2-dimethylpropyl, 1-methylpentyl, 2-methylpentyl, 3-methylpentyl, 4- methylpentyl, 1,1-dimethylbutyl, 1,2-dimethylbutyl, 1,3-dimethylbutyl, 2,2-dimethylbutyl, 2,3- dimethylbutyl, 3,3-dimethylbutyl, 1-ethylbutyl, 2-ethylbutyl, 1,1,2-trimethylpropyl, 1,2,2- trimethylpropyl, 1-ethyl-1-methylpropyl and l-ethyl-2-methylpropyl or the different isomers. The term “C2-C6-alkenyl”, used either alone or in compound words refers to straight-chain or branched C2 to C6 alkenes. Non-limiting examples of C2-C6-alkenyl include ethenyl, 1-propenyl, 2- propenyl, 1-methylethenyl, 1-butenyl, 2-butenyl, 3-butenyl, 1-methyl-1-propenyl, 2-methyl-l- propenyl, l-methyl-2 -propenyl, 2-methyl-2-propenyl, 1-pentenyl, 2-pentenyl, 3-pentenyl, 4- pentenyl, 1-methyl-1-butenyl, 2-methyl-1-butenyl, 3-methyl-1-butenyl, l-methyl-2-butenyl, 2- methyl-2-butenyl, 3-methyl-2-butenyl, l-methyl-3-butenyl, 2-methyl-3-butenyl, 3-methyl-3- butenyl, 1,1-dimethyl-2-propenyl, 1,2-dimethyl-1-propenyl, 1,2-dimethyl-2 -propenyl, 1-ethyl-1- propenyl, l-ethyl-2-propenyl, 1-hexenyl, 2-hexenyl, 3-hexenyl, 4-hexenyl, 5-hexenyl, 1-methyl-1- pentenyl, 2-methyl-1-pentenyl, 3-methyl-1-pentenyl, 4-methyl-1-pentenyl, 1-methyl-2-pentenyl, 2- methyl-2-pentenyl, 3-methyl-2-pentenyl, 4-methyl-2-pentenyl, l-methyl-3-pentenyl, 2-methyl-3- pentenyl, 3-methyl-3-pentenyl, 4-methyl-3-pentenyl, 1-methyl-4-pentenyl, 2-methyl-4-pentenyl, 3- methyl-4-pentenyl, 4-methyl-4-pentenyl, 1,1-dimethyl-2-butenyl, l,l-dimethyl-3-butenyl, 1,2- dimethyl-l-butenyl, 1,2-dimethyl-2-butenyl, 1,2-dimethyl-3-butenyl, 1,3-dimethyl-1-butenyl, l,3- dimethyl-2-butenyl, l,3-dimethyl-3-butenyl, 2,2-dimethyl-3-butenyl, 2,3-dimethyl-1-butenyl, 2,3-
dimethyl-2-butenyl, 2,3-dimethyl-3-butenyl, 3,3-dimethyl-l-butenyl, 3,3-dimethyl-2-butenyl, 1- ethyl-1-butenyl, 1-ethyl-2-butenyl, l-ethyl-3-butenyl, 2-ethyl- 1-butenyl, 2-ethyl-2-butenyl, 2-ethyl- 3-butenyl, l,l,2-trimethyl-2-propenyl, 1-ethyl-l-methyl-2-propenyl, l-ethyl-2-methyl-l-propenyl and l-ethyl-2-methyl-2-propenyl and the different isomers. “Alkenyl” also includes polyenes such as 1,2-propadienyl and 2,4-hexadienyl. This definition also applies to alkenyl as a part of a composite substituent, for example haloalkenyl, alkenyloxy or haloalkenyloxy and the like, unless defined specifically elsewhere. The term “C2-C6-alkynyl”, used either alone or in compound words refers to straight-chain or branched C2 to C6 alkynes. Non-limiting examples of C2-C6-alkynes include ethynyl, 1-propynyl, 2- propynyl, 1-butynyl, 2-butynyl, 3-butynyl, 1-methyl-2-propynyl, 1-pentynyl, 2-pentynyl, 3- pentynyl, 4-pentynyl, l-methyl-2-butynyl, l-methyl-3-butynyl, 2-methyl-3-butynyl, 3-methyl-l- butynyl, 1,1-dimethyl-2-propynyl, 1-ethyl -2-propynyl, 1-hexynyl, 2-hexynyl, 3-hexynyl, 4- hexynyl, 5-hexynyl, 1-methyl-2-pentynyl, l-methyl-3-pentynyl, 1-methyl-4-pentynyl, 2-methyl-3- pentynyl, 2-methyl-4-pentynyl, 3-methyl-l-pentynyl, 3-methyl-4-pentynyl, 4-methyl-l-pentynyl, 4- methyl-2-pentynyl, 1,1-dimethyl-2-butynyl, l,l-dimethyl-3-butynyl, l,2-dimethyl-3-butynyl, 2,2- dimethyl-3-butynyl, 3,3-dimethyl-l-butynyl, l-ethyl-2-butynyl, l-ethyl-3-butynyl, 2-ethyl-3-butynyl and 1-ethyl-l-methyl-2-propynyl and the different isomers. This definition also applies to alkynyl as a part of a composite substituent, for example haloalkynyl etc. alkynyloxy, haloalkynyloxy, unless specifically defined elsewhere. The term “alkynyl” can also include moieties comprised of multiple triple bonds such as 2,5-hexadiynyl. The term “C3-C8-cycloalkyl” means a saturated carbocyclic ring containing 3 to 8 carbon atoms. Non-limiting examples of C3-C8-cycloalkyl include cyclopropyl, cyclopentyl and cyclohexyl. This definition also applies to cycloalkyl as a part of a composite substituent, for example C3-C8- halocycloalkyl, cycloalkylalkyl etc., unless specifically defined elsewhere. The term “C3-C6-cycloalkoxy”, and “C3-C6-cycloalkylthio” and the like are defined analogously. Non limiting examples of C3-C6-cycloalkoxy or C3-C6-cycloalkylthio include cyclopropyloxy, cyclopentyloxy, cyclohexyloxy, cyclopropylthio, cyclopentylthio and cyclohexylthio. The term “halogen”, either alone or in compound words such as “haloalkyl”, includes fluorine, chlorine, bromine or iodine. Further, when used in compound words such as “C1-C6-haloalkyl”, said C1-C6-alkyl may be partially or fully substituted with halogen atoms which may be the same or different. Non-limiting examples of “C1-C6-haloalkyl” include chloromethyl, bromomethyl, dichloromethyl, trichloromethyl, fluoromethyl, difluoromethyl, trifluoromethyl, chlorofluoromethyl, dichlorofluoromethyl, chlorodifluoromethyl, 1-chloroethyl, 1-bromoethyl, 1-
fluoroethyl, 2-fluoroethyl, 2,2-difluoroethyl, 2,2,2-trifluoroethyl, 2-chloro-2-fluoroethyl, 2-chloro- 2,2-difluoroethyl, 2,2-dichloro-2-fluoroethyl, 2,2,2-trichloroethyl, pentafluoroethyl, 1,1-dichloro- 2,2,2-trifluoroethyl, and 1,1,1-trifluoroprop-2-yl. The terms “C2-C6-haloalkenyl”, “C2-C6-haloalkynyl” are defined analogously except that, instead of C1-C6-alkyl groups, C2-C6-alkenyl and C2-C6-alkynyl groups are present as a part of the substituent. The term “C1-C6-alkoxy” used either alone or in compound words includes a straight or branched chain C1-C6-alkoxy. Examples of C1-C6-alkoxy include methoxy, ethoxy, propoxy, 1-methylethoxy, butoxy, 1-methylpropoxy, 2-methylpropoxy, 1,1-dimethylethoxy, pentoxy, 1-methylbutoxy, 2- methylbutoxy, 3-methylbutoxy, 2,2-dimethylpropoxy, 1-ethylpropoxy, hexoxy, 1,1- dimethylpropoxy, 1,2-dimethylpropoxy, 1-methylpentoxy, 2-methylpentoxy, 3-methylpentoxy, 4- methylpentoxy, 1,1-dimethylbutoxy, 1,2-dimethylbutoxy, 1,3-dimethylbutoxy, 2,2-dimethylbutoxy, 2,3-dimethylbutoxy, 3,3-dimethylbutoxy, 1-ethylbutoxy, 2-ethylbutoxy, 1,1,2-trimethylpropoxy, 1,2,2-trimethylpropoxy, 1-ethyl-1-methylpropoxy and l-ethyl-2-methylpropoxy and the different isomers. This definition also applies to alkoxy as a part of a composite substituent, for example haloalkoxy, etc., unless specifically defined elsewhere. The term “C1-C6-haloalkoxy” means straight-chain or branched C1-C6-alkoxy groups where some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as specified above. Non-limiting examples of C1-C6-haloalkoxy include chloromethoxy, bromomethoxy, dichloromethoxy, trichloromethoxy, fluoromethoxy, difluoromethoxy, trifluoromethoxy, chlorofluoromethoxy, dichlorofluoromethoxy, chlorodifluoromethoxy, 1-chloroethoxy, 1- bromoethoxy, 1-fluoroethoxy, 2-fluoroethoxy, 2,2-difluoroethoxy, 2,2,2-trifluoroethoxy, 2-chloro- 2-fluoroethoxy, 2-chloro-2,2-difluoroethoxy, 2,2-dichloro-2-fluoroethoxy, 2,2,2-trichloroethoxy, pentafluoroethoxy and l,l,l-trifluoroprop-2-oxy. This definition also applies to haloalkoxy as a part of a composite substituent, for example haloalkoxyalkyl etc., unless specifically defined elsewhere. The term “C1-C6-alkylthio” includes branched or straight-chain alkylthio moieties with 1 to 6 carbon atoms. Non-limiting examples of C1-C6-alkylthio includes methylthio, ethylthio, propylthio, 1- methylethylthio, butylthio, 1-methylpropylthio, 2-methylpropylthio, 1,1-dimethylethylthio, pentylthio, 1-methylbutylthio, 2-methylbutylthio, 3-methylbutylthio, 2,2-dimethylpropylthio, 1- ethylpropylthio, hexylthio, 1,1-dimethylpropylthio, 1,2-dimethylpropylthio, 1-methylpentylthio, 2- methylpentylthio, 3-methylpentylthio, 4-methylpentylthio, 1,1-dimethylbutylthio, 1,2- dimethylbutylthio, 1,3-dimethylbutylthio, 2,2-dimethylbutylthio, 2,3-dimethylbutylthio, 3,3- dimethylbutylthio, 1-ethylbutylthio, 2-ethylbutylthio, 1,1,2-trimethylpropylthio, 1,2,2-
trimethylpropylthio, 1-ethyl-1-methylpropylthio and l-ethyl-2-methylpropylthio and the different isomers. The term “C1-C6-haloalkylthio” means straight-chain or branched C1-C6-alkylthio group where some or all of the hydrogen atoms in these groups may be replaced by halogen atoms as specified above. Non-limiting examples of haloalkylthio include chloromethylthio, bromomethylthio, dichloromethylthio, trichloromethylthio, fluoromethylthio, difluoromethylthio, trifluoromethylthio, chlorofluoromethylthio, dichlorofluoromethylthio, chlorodifluoromethylthio, 1-chloroethylthio, 1- bromoethylthio, 1- fluoroethylthio, 2-fluoroethylthio, 2,2-difluoroethylthio, 2,2,2-trifluoroethylthio, 2-chloro-2- fluoroethylthio, 2-chloro-2,2-difluoroethylthio, 2,2-dichloro-2-fluoroethylthio, 2,2,2- trichloroethylthio, pentafluoroethylthio and l,l,l-trifluoroprop-2-ylthio. This definition also applies to haloalkylthio as a part of a composite substituent, for example haloalkylthioalkyl etc., unless specifically defined elsewhere. The term “C1-C6-alkylsulphinyl” includes branched or straight-chain alkylsulphinyl moiety with 1 to 6 carbon atoms. Non-limiting examples of C1-C6-alkylsulfinyl include methylsulphinyl, ethylsulphinyl, propylsulphinyl, 1-methylethylsulphinyl, butylsulphinyl, 1-methylpropylsulphinyl, 2-methylpropylsulphinyl, 1,1-dimethylethylsulphinyl, pentylsulphinyl, 1-methylbutylsulphinyl, 2- methylbutylsulphinyl, 3-methylbutylsulphinyl, 2,2-dimethylpropylsulphinyl, 1- ethylpropylsulphinyl, hexylsulphinyl, 1,1-dimethylpropylsulphinyl, 1,2-dimethylpropylsulphinyl, 1-methylpentylsulphinyl, 2-methylpentylsulphinyl, 3-methylpentylsulphinyl, 4- methylpentylsulphinyl, 1,1-dimethylbutylsulphinyl, 1,2-dimethylbutylsulphinyl, 1,3- dimethylbutylsulphinyl, 2,2-dimethylbutylsulphinyl, 2,3-dimethylbutylsulphinyl, 3,3- dimethylbutylsulphinyl, 1-ethylbutylsulphinyl, 2-ethylbutylsulphinyl, 1,1,2- trimethylpropylsulphinyl, 1,2,2-trimethylpropylsulphinyl, 1-ethyl-1-methylpropylsulphinyl and 1- ethyl-2-methylpropylsulphinyl and the different isomers. The term “C1-C6-alkylsulfonyl” includes branched or straight-chain alkylsulfonyl moiety with 1 to 6 carbon atoms. Non-limiting examples of C1-C6-alkylsulfonyl include methylsulfonyl, ethylsulfonyl, propylsulfonyl, 1-methylethylsulfonyl, butylsulfonyl, 1-methylpropylsulfonyl, 2- methylpropylsulfonyl, 1,1-dimethylethylsulfonyl, pentylsulfonyl, 1-methylbutylsulfonyl, 2- methylbutylsulfonyl, 3-methylbutylsulfonyl, 2,2-dimethylpropylsulfonyl, 1-ethylpropylsulfonyl, hexylsulfonyl, 1,1-dimethylpropylsulfonyl, 1,2-dimethylpropylsulfonyl, 1-methylpentylsulfonyl, 2- methylpentylsulfonyl, 3-methylpentylsulfonyl, 4-methylpentylsulfonyl, 1,1-dimethylbutylsulfonyl, 1,2-dimethylbutylsulfonyl, 1,3-dimethylbutylsulfonyl, 2,2-dimethylbutylsulfonyl, 2,3- dimethylbutylsulfonyl, 3,3-dimethylbutylsulfonyl, 1-ethylbutylsulfonyl, 2-ethylbutylsulfonyl,
1,1,2-trimethylpropylsulfonyl, 1,2,2-trimethylpropylsulfonyl, 1-ethyl-1-methylpropylsulfonyl and l-ethyl-2-methylpropylsulfonyl and the different isomers. The term “C1-C6-alkylcarbonyl” includes branched or straight-chain alkylcarbonyl moiety with 1 to 6 carbon atoms. Non-limiting examples of C1-C6-alkylcarbonyl include methylcarbonyl, ethylcarbonyl, propylcarbonyl, 1-methylethylcarbonyl, butylcarbonyl, 1-methylpropylcarbonyl, 2- methylpropylcarbonyl, 1,1-dimethylethylcarbonyl, pentylcarbonyl, 1-methylbutylcarbonyl, 2- methylbutylcarbonyl, 3-methylbutylcarbonyl, 2,2-dimethylpropylcarbonyl, 1-ethylpropylcarbonyl, hexylcarbonyl, 1,1-dimethylpropylcarbonyl, 1,2-dimethylpropylcarbonyl, 1-methylpentylcarbonyl, 2-methylpentylcarbonyl, 3-methylpentylcarbonyl, 4-methylpentylcarbonyl, 1,1- dimethylbutylcarbonyl, 1,2-dimethylbutylcarbonyl, 1,3-dimethylbutylcarbonyl, 2,2- dimethylbutylcarbonyl, 2,3-dimethylbutylcarbonyl, 3,3-dimethylbutylcarbonyl, 1- ethylbutylcarbonyl, 2-ethylbutylcarbonyl, 1,1,2-trimethylpropylcarbonyl, 1,2,2- trimethylpropylcarbonyl, 1-ethyl-1-methylpropylcarbonyl and l-ethyl-2-methylpropylcarbonyl and the different isomers. The term “C6-C10 aryl” refers to mono or bicyclic aromatic carbocyclic ring containing 6 to 10 carbon atoms. Non-limiting examples of “C6-C10 aryl” includes phenyl, indenyl, indanyl, naphthyl. The term “5- or 6-membered aromatic heterocyclic ring or 5- or 6-membered heteroaromatic ring” refers to a mono cyclic aromatic heterocyclic ring comprising at least one heteroatom selected from nitrogen, oxygen, or suphur or a group selected from S(O) or S(O)2. Non-limiting examples for said term includes furanyl, pyrrolyl, thiophenyl (thienyl), oxazolyl, thiazolyl, imidazolyl, pyrazolyl, isoxazolyl, isothiazolyl, oxadiazolyl, thiadiazolyl, triazolyl, tetrazolyl, pyridyl, pyridazinyl, pyrimidinyl, pyrazinyl, triazinyl etc. The term "heterocyclic ring” includes "aromatic or non-aromatic heterocyclic ring comprising at least one heteroatom selected from N, O, and S(O)0-2. When a compound is substituted with a substituent bearing a subscript that indicates the number of said substituents can exceed 1, said substituents (when they exceed 1) are independently selected from the group of defined substituents. Further, when the subscript m in (R)m indicates an integer ranging from for example 0 to 4 then the number of substituents may be selected from the integers between 0 and 4 inclusive. When a group contains a substituent which can be hydrogen, then, when this substituent is taken as hydrogen, it is recognized that said group is being un-substituted.
Optionally substituted groups may be mono- or polysubstituted, where the substituents in the case of polysubstitutions may be the same or different. The embodiments herein and the various features and advantageous details thereof are explained with reference to the non-limiting embodiments in the description. Descriptions of well-known components and processing techniques are omitted so as to not unnecessarily obscure the embodiments herein. The examples used herein are intended merely to facilitate an understanding of ways in which the embodiments herein may be practiced and to further enable those of skilled in the art to practice the embodiments herein. Accordingly, the examples should not be construed as limiting the scope of the embodiments herein. The description of the specific embodiments will so fully reveal the general nature of the embodiments herein that others can, by applying current knowledge, readily modify and/or adapt for various applications such specific embodiments without departing from the generic concept, and, therefore, such adaptations and modifications should and are intended to be comprehended within the meaning and range of equivalents of the disclosed embodiments. It is to be understood that the phraseology or terminology employed herein is for the purpose of description and not of limitation. Therefore, while the embodiments herein have been described in terms of preferred embodiments, those skilled in the art will recognize that the embodiments herein can be practiced with modification within the spirit and scope of the embodiments as described herein. Any discussion of documents, acts, materials, devices, articles and the like that has been included in this specification is solely for the purpose of providing a context for the disclosure. It is not to be taken as an admission that any or all these matters form a part of the prior art base or were common general knowledge in the field relevant to the disclosure as it existed anywhere before the priority date of this application. The numerical values mentioned in the description and the description/claims though might form a critical part of the present invention, any deviation from such numerical values shall still fall within the scope of the present invention if that deviation follows the same scientific principle as that of the present invention disclosed in the present invention. The inventive compound of the present invention may, if appropriate, be present as mixtures of different possible isomeric forms, especially of stereoisomers, for example E and Z, threo and erythro, and also optical isomers, but if appropriate also of tautomers. Both the E and the Z isomers, but also the threo and erythro isomers, and the optical isomers, and any desired mixtures of these isomers and the possible tautomeric forms are disclosed and claimed.
The term "polymorph" refers to a particular crystalline form of a chemical compound that can crystallize in different crystalline forms, these forms having different arrangements and/or conformations of the molecules in the crystal lattice. Although polymorphs can have the same chemical composition, they can also differ in composition due to the presence or absence of co- crystallized water or other molecules, which can be weakly or strongly bound in the lattice. Polymorphs can differ in such chemical, physical and biological properties as crystal shape, density, hardness, color, chemical stability, melting point, hygroscopicity, suspensibility, dissolution rate and biological availability. One skilled in the art will appreciate that a polymorph of a compound represented by formula (I), formula (I-A) to formula (I-F) or formula (I-D-a) can exhibit beneficial effects (e.g., suitability for preparation of useful formulations, improved biological performance) relative to another polymorph or a mixture of polymorphs of the same compound represented by formula (I), formula (I-A) to formula (I-F) or formula (I-D-a). The preparation and isolation of a particular polymorph of a compound represented by formula (I), formula (I-A) to formula (I-F) or Formula (I-D-a) can be achieved by methods known to those skilled in the art including, for example, crystallization using selected solvents and temperatures. The term “pest” for the purpose of the present disclosure includes but is not limited to fungi, stramenopiles (oomycetes) and bacteria. The term “plant” is understood here to mean all plants and plant populations, such as desired and undesired wild plants or crop plants (including naturally occurring crop plants). Crop plants may be plants which can be obtained by conventional breeding and optimization methods or by biotechnological and genetic engineering methods or combinations of these methods, including transgenic plants and including the plant cultivars which are protectable and non-protectable by plant breeders’ rights. For the purpose of the present disclosure the term “plant” includes a living organism of the kind exemplified by trees, shrubs, herbs, grasses, ferns, and mosses, typically growing in a site, absorbing water and required substances through its roots, and synthesizing nutrients in its leaves by photosynthesis. Examples of “plant” for the purpose of the present invention include but are not limited to agricultural crops such as wheat, rye, barley, triticale, oats or rice; beet, e.g. sugar beet or fodder beet; fruits and fruit trees, such as pomes, stone fruits or soft fruits, e.g. apples, pears, plums, peaches, almonds, cherries, strawberries, raspberries, blackberries or gooseberries; leguminous plants, such as lentils, peas, alfalfa or soybeans; oil plants, such as rape, mustard, olives, sunflowers, coconut, cocoa beans, castor oil plants, oil palms, ground nuts or soybeans; cucurbits, such as
squashes, cucumber or melons; fiber plants, such as cotton, flax, hemp or jute; citrus fruit and citrus trees, such as oranges, lemons, grapefruits or mandarins; any horticultural plants, vegetables, such as spinach, lettuce, asparagus, cabbages, carrots, onions, tomatoes, potatoes, cucurbits or paprika; lauraceous plants, such as avocados, cinnamon or camphor; cucurbitaceae; oleaginous plants; energy and raw material plants, such as cereals, corn, soybean, other leguminous plants, rape, sugar cane or oil palm; tobacco; nuts; coffee; tea; cacao; bananas; peppers; vines (table grapes and grape juice grape vines); hop; turf; sweet leaf (also called Stevia); natural rubber plants or ornamental and forestry plants, such as flowers, shrubs, broad-leaved trees or evergreens, e.g. conifers; and on the plant propagation material, such as seeds, and the crop material of these plants. Preferably, the plant for the purpose of the present invention includes but is not limited to cereals, corn, rice, soybean and other leguminous plants, fruits and fruit trees, grapes, nuts and nut trees, citrus and citrus trees, any horticultural plants, cucurbitaceae, oleaginous plants, tobacco, coffee, tea, cacao, sugar beet, sugar cane, cotton, potato, tomato, onions, peppers and vegetables, ornamentals, any floricultural plants and other plants for use of humans and animals. The term “plant parts” is understood to mean all parts and organs of plants above and below the ground. For the purpose of the present disclosure the term plant parts include but is not limited to cuttings, leaves, twigs, tubers, flowers, seeds, branches, roots including taproots, lateral roots, root hairs, root apex, root cap, rhizomes, slips, shoots, fruits, fruit bodies, bark, stem, buds, auxillary buds, meristems, nodes and internodes. The term “locus thereof” includes soil, surroundings of plant or plant parts and equipment or tools used before, during or after sowing/planting a plant or a plant part. Application of the compounds of the present disclosure or the compound of the present disclosure in a composition optionally comprising other compatible compounds to a plant or a plant material or locus thereof includes application by a technique known to a person skilled in the art which includes but is not limited to spraying, coating, dipping, fumigating, impregnating, injecting and dusting. The term “applied” means adhered to a plant or plant part either physically or chemically including impregnation. The present invention provides a compound of formula (I) or an agriculturally acceptable salt, N- oxide, isomer, tautomer or polymorph thereof,
wherein, D is selected from the group consisting of phenyl or 5- or 6-membered heteroaryl ring, wherein said 5- or 6-membered heteroaryl ring comprises 1, 2, 3 or 4 heteroatoms each independently selected from N, O, and S(O)0-2, and wherein said phenyl or 5- or 6-membered heteroaryl ring may be further substituted with one or more different or identical substituents selected from R1b and/or R1c; R1a is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, nitro, C1-C6- alkyl, C1-C6-haloalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C3- C8-cycloalkyl, C3-C8-halocycloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C2-C6-alkenyloxy, C2-C6- haloalkenyloxy, C2-C6-alkynyloxy, C2-C6-haloalkynyloxy, C3-C6-cycloalkyloxy, C1-C6- alkylthio, C1-C6-haloalkylthio, C3-C6-cycloalkylthio, C1-C6-alkylsulphinyl, C1-C6- haloalkylsulphinyl, C1-C6-alkylsulfonyl, C1-C6-haloalkylsulfonyl, -N(R7)2, -CO-N(R9)2, - S(O)(R8)=NR9, -N=S(O)(R8)2, P(O)(R8)2, Si(R8)3 or phenyl which may be optionally substituted with one or more different or identical substituents selected from halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy or C1-C6-haloalkoxy; R1b is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, C1-C6-alkyl, C1- C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy and C1-C6-haloalkoxy; R1c is selected from the group consisting of hydrogen, halogen, C1-C6-alkyl, C1-C6-haloalkyl, C3- C8-cycloalkyl, hydroxyl and cyano; R2 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy;
R3 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C1-C6-cyanoalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6- haloalkynyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R4a and R4b are each independently selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; or R4a and R4b may together form an oxo group (=O), or a 3- to 6-membered carbocyclic ring; Z1, Z2 and Z3 are each independently selected from CR5 or N; R5 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl and C1-C6-alkoxy; B represents a phenyl ring or a 5- or 6-membered aromatic heterocyclic ring fused at C3, C4 carbons of the piperidine ring, wherein the 5- or 6-membered aromatic heterocyclic ring comprises 1 to 3 heteroatoms selected from oxygen, nitrogen or S(O)0-2, wherein the ring B is unsubstituted or substituted with one or more different or identical substituents selected from R6; R6 can be selected from the group consisting of hydrogen, halogen, cyano, hydroxyl, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C3-C8-cycloalkyl, C3-C8-halocycloalkyl, C6- C10-aryl which may be substituted with 1 or 2 same or different substitutent selected from the group consisting of halogen, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6-alkoxy and C1-C6-haloalkoxy, or -N(R7)2; R7 represents hydrogen, C1-C6-alkyl, C3-C8-cycloalkyl, C1-C6-alkylcarbonyl, C3-C8- cycloalkylcarbonyl, C1-C6-alkoxycarbonyl or C1-C6-alkylsulfonyl; R8 is selected from the group consisting of hydrogen, halogen, C1-C6-alkyl, C1-C6-haloalkyl, C2- C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C3-C8-cycloalkyl, C1-C6- alkoxy and C1-C6-haloalkoxy; R9 is selected from the group consisting of hydrogen, C1-C6-alkyl, C1-C6-haloalkyl and C3-C8- cycloalkyl; R10 is selected from the group consisting of hydrogen and C1-C6-alkyl; R1a and R1b optionally together along with the phenyl or 5- or 6-membered heteroaryl ring may form an 8- to 10-membered bicyclic carbocyclic or heterocyclic ring system that can be optionally substituted with one or more different or identical substituents selected from R1b and/or R1c, wherein the bicyclic heterocyclic ring system comprises one or more heteroatoms
selected from nitrogen, oxygen or S(O)0-2, and wherein one or more carbon atom of the bicyclic carbocyclic or heterocyclic ring system can be replaced with C(O). In one embodiment of the present invention, it provides a compound of formula (I) or an agriculturally acceptable salt, N-oxide, isomer, tautomer or polymorph thereof, wherein,
D is selected from the group consisting of phenyl or 5- or 6-membered heteroaryl ring, wherein said 5- or 6-membered heteroaryl ring comprises 1, 2, 3 or 4 heteroatoms each independently selected from N, O, and S(O)0-2, and wherein said phenyl or 5- or 6-membered heteroaryl ring may be further substituted with one or more different or identical substituents selected from R1b and/or R1c; R1a is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, nitro, C1-C6- alkyl, C1-C6-haloalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C3- C8-cycloalkyl, C3-C8-halocycloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C2-C6-alkenyloxy, C2-C6- haloalkenyloxy, C2-C6-alkynyloxy, C2-C6-haloalkynyloxy, C3-C6-cycloalkyloxy, C1-C6- alkylthio, C1-C6-haloalkylthio, C3-C6-cycloalkylthio,
C6-alkylsulphinyl, C1-C6- haloalkylsulphinyl, C1-C6-alkylsulfonyl, C1-C6-haloalkylsulfonyl, -N(R7)2, -CO-N(R9)2, - S(O)(R8)=NR9, -N=S(O)(R8)2, P(O)(R8)2, Si(R8)3 or phenyl which may be optionally substituted with one or more different or identical substituents selected from halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy or C1-C6-haloalkoxy; R1b is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, C1-C6-alkyl, C1- C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy and C1-C6-haloalkoxy; R1c is selected from the group consisting of hydrogen, halogen, C1-C6-alkyl, C1-C6-haloalkyl, C3- C8-cycloalkyl, hydroxyl and cyano;
R2 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R3 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C1-C6-cyanoalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6- haloalkynyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R4a and R4b are each independently selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; or R4a and R4b may together form an oxo group (=O), or a 3- to 6-membered carbocyclic ring; Z1, Z2 and Z3 are each independently selected from CR5 or N; R5 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl and C1-C6-alkoxy; B represents a 5-membered aromatic heterocyclic ring fused at C3, C4 carbons of the piperidine ring (i.e. C3 and C4 carbons of the piperidine ring are part of the 5-membered aromatic heterocyclic ring), wherein the 5-membered aromatic heterocyclic ring comprises 1 to 3 heteroatoms selected from oxygen, nitrogen or S(O)0-2, wherein the ring B is unsubstituted or substituted with one or more different or identical substituents selected from R6; R6 can be selected from the group consisting of hydrogen, halogen, cyano, hydroxyl, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C3-C8-cycloalkyl, C3-C8-halocycloalkyl, C6- C10-aryl which may be substituted with 1 or 2 same or different substitutent selected from the group consisting of halogen, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6-alkoxy and C1-C6-haloalkoxy, or -N(R7)2; R7 represents hydrogen, C1-C6-alkyl, C3-C8-cycloalkyl, C1-C6-alkylcarbonyl, C3-C8- cycloalkylcarbonyl, C1-C6-alkoxycarbonyl or C1-C6-alkylsulfonyl; R8 is selected from the group consisting of hydrogen, halogen, C1-C6-alkyl, C1-C6-haloalkyl, C2- C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C3-C8-cycloalkyl, C1-C6- alkoxy and C1-C6-haloalkoxy; R9 is selected from the group consisting of hydrogen, C1-C6-alkyl, C1-C6-haloalkyl and C3-C8- cycloalkyl; R10 is selected from the group consisting of hydrogen and C1-C6-alkyl;
R1a and R1b optionally together along with the phenyl or 5- or 6-membered heteroaryl ring may form an 8- to 10-membered bicyclic carbocyclic or heterocyclic ring system that can be optionally substituted with one or more different or identical substituents selected from R1b and/or R1c, wherein the bicyclic heterocyclic ring system comprises one or more heteroatoms selected from nitrogen, oxygen or S(O)0-2, and wherein one or more carbon atom of the bicyclic carbocyclic or heterocyclic ring system can be replaced with C(O). In an embodiment of the present invention, the compound of formula (I) is represented by a compound of formula (I-A): wherein n = 1-3 and B, R1a-
defined above for the compound of formula (I). In an embodiment of the present invention, the compound of formula (I) is represented by a compound of formula (I-B): wherein n = 1-3, preferably
and Z1-Z3 are as defined above for the compound of formula (I), Z4 and Z5 each independently selected from C-R6 or N. In another embodiment of the present invention, the compound of formula (I) is represented by a compound of formula (I-C):
wherein n = 1-3, preferably n = 1 and R1a-R1c, R2, R3, R4a, R4b, R10 and Z1-Z3 are as defined above for the compound of formula (I). In an embodiment of the present invention, the compound of formula (I-A),
wherein B is selected from a 5-membered aromatic heterocyclic ring fused at C3, C4 carbons of the piperidine ring, wherein the 5-membered aromatic heterocyclic ring comprises 1 to 3 heteroatoms selected from oxygen, nitrogen or S(O)0-2, wherein the ring B is unsubstituted or substituted with one or more different or identical substituents selected from R6; and n, R1a-R1c, R2, R3, R4a, R4b, R10 and Z1-Z3 are as defined above for the compound of formula (I). In a preferred embodiment of the present invention, for the compound of formula (I-A),
B is a 5-membered aromatic heterocyclic ring (C3, C4 carbons of the piperidine ring are part of the 5-membered aromatic heterocyclic ring) which may be selected as thiophenyl (thienyl), that is fused
at C3, C4 carbons of the piperidine ring, wherein said ring is unsubstituted or substituted with one or more different or identical substituents selected from R6; and n, R1a-R1c, R2, R3, R4a, R4b, R10 and Z1-Z3 are as defined above for the compound of formula (I). In another embodiment of the present invention, the compound of formula (I) is a compound of formula (I-D): wherein n = 1-3, preferably n and Z1-Z3 are as defined above
for the compound of formula (I). In another embodiment of the present invention, the compound of formula (I) is a compound of formula (I-E): wherein n = 1-3, preferably
and Z1-Z3 are as defined above for the compound of formula (I). In another embodiment of the present invention, the compound of formula (I) is a compound of formula (I-F):
wherein n = 1-3, preferably n= 1 and R1a-R1c, R2, R3, R4a, R4b, R10 and Z1-Z3 are as defined above for the compound of formula (I). In another embodiment, the compound of formula (I), or formula (I-A) to formula (I-F), wherein the ring C = , is selected from one of the following rings C-1 to C-7:
wherein R5 is
halogen, cyano, C1- C6-alkyl, C1-C6-haloalkyl and C3-C8-cycloalkyl and R5 can be same or different in the C-7 imidazolyl ring. In another preferred embodiment, for the compound of formula (I), or formula (I-A) to formula (I- F) wherein, R1a is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, nitro, C1-C6- alkyl, C1-C6-haloalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C3- C8-cycloalkyl, C3-C8-halocycloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C2-C6-alkenyloxy, C2-C6- haloalkenyloxy, C2-C6-alkynyloxy, C2-C6-haloalkynyloxy, C3-C6-cycloalkyloxy, C1-C6- alkylthio, C1-C6-haloalkylthio, C3-C6-cycloalkylthio, C1-C6-alkylsulphinyl, C1-C6- haloalkylsulphinyl, C1-C6-alkylsulfonyl, C1-C6-haloalkylsulfonyl, -N(R7)2, -CO-N(R9)2, -
S(O)(R8)=NR9, -N=S(O)(R8)2, P(O)(R8)2, Si(R8)3 or phenyl which may be optionally substituted with one or more different or identical substituents selected from halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl , C1-C6-alkoxy or C1-C6-haloalkoxy; R1b is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy and C1-C6-haloalkoxy; R1c is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl and C3-C8-cycloalkyl; R2 is selected from the group consisting of hydrogen, C1-C6-alkyl, and C1-C6-haloalkyl; R3 is selected from the group consisting of hydrogen, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6- cyanoalkyl, C2-C6-alkenyl, C2-C6-alkynyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R4a and R4b are each independently selected from the group consisting of hydrogen, C1-C6-alkyl and C1-C6-haloalkyl or together may form an oxo group (=O) or a 3- to 6-membered carbocyclic ring; ring C = is preferably selected from one of the following rings C-1 to C-7:
hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl and C3-C8-cycloalkyl, and R5 can be same or different in the C-7 imidazolyl ring; R6 is selected from the group consisting of hydrogen, halogen, cyano, hydroxyl, C1-C6-alkyl, C1- C6-haloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C3-C8-cycloalkyl, or -N(R7)2; R7 represents hydrogen, C1-C6-alkyl, C3-C8-cycloalkyl, C1-C6-alkylcarbonyl, or C3-C8- cycloalkylcarbonyl; and R8 is selected from the group consisting of hydrogen, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6-alkoxy and C1-C6-haloalkoxy; R9 is selected from the group consisting of hydrogen, C1-C6-alkyl and C3-C8-cycloalkyl; R10 is selected from the group consisting of hydrogen and C1-C6-alkyl;
R1a and R1b optionally together along with the phenyl ring may form an 8- to 10-membered bicyclic carbocyclic or heterocyclic ring system that can be optionally substituted with one or more different or identical substituents selected from R1b and/or R1c, wherein the bicyclic heterocyclic ring system comprises one or more heteroatoms selected from nitrogen, oxygen or S(O)0-2 and n is 1-3, preferably n = 1-2 and more preferably n = 1. In yet another preferred embodiment, for the compound of formula (I), or formula (I-A) to formula (I-F) wherein, R1a is selected from the group consisting of halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C6- cycloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C3-C6-cycloalkyloxy, C1-C6-alkylthio, C1-C6- haloalkylthio, C1-C6-alkylsulfonyl, more preferably fluoro, chloro, bromo, iodo, cyano, methyl, trifluoromethyl, O-isopropyl or cyclopropyl; R1b is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl and C3-C6-cycloalkyl, more preferably hydrogen, fluoro, chloro, bromo, cyano, methyl, trifluoromethyl and cyclopropyl; R1c is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl and C1-C6- haloalkyl, more preferably hydrogen, halogen such as fluoro, chloro or bromo or cyano; R2 is selected from hydrogen; R3 is selected from the group consisting of hydrogen, C1-C6-alkyl, C1-C6-haloalkyl, or C1-C6- cyanoalkyl, more preferably hydrogen, methyl, CH2Cl, or CH2CN; R4a is selected from the group consisting of hydrogen and C1-C6-alkyl, more preferably hydrogen or methyl; R4b is selected from the group consisting of hydrogen and C1-C6-alkyl, more preferably hydrogen; or R4a and R4b may together form an oxo group (=O) or cyclopropyl ring; ring C = is selected from one of the following rings C-1 to C-7:
wher hydrogen, halogen, C1-C6-alkyl, C1-C6-haloalkyl and C3-C8-cycloalkyl and R5 can be same or different in the C-7 imidazolyl ring. More preferably R5 can be selected from hydrogen, chloro, methyl, ethyl, trifluoromethyl or cyclopropyl; R6 is selected from hydrogen; R10 is selected from hydrogen or C1-C6-alkyl, more preferably hydrogen or methyl and n is 1-3, preferably n = 1-2 and more preferably n = 1. In one preferred embodiment of the present invention, the compound of formula (I) may be selected from a compound of formula (I-D-a),
, wherein, R2 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R3 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C1-C6-cyanoalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6- haloalkynyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R4a is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy;
R1a is selected from halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C6-cycloalkyl, C1-C6- alkoxy, C1-C6-haloalkoxy, C3-C6-cycloalkyloxy, C1-C6-alkylthio, C1-C6-haloalkylthio, C1-C6- alkylsulfonyl; R1b is selected from the
consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl and C3-C6-cycloalkyl; R1c is selected from hydrogen, halogen, cyano, C1-C6-alkyl and C1-C6-haloalkyl; and ring C = is selected from one of the following rings C-1 to C-7:
wherein R5 is
or C3-C8-cycloalkyl and in ring C-7, R5 are independently selected from hydrogen, halogen, C1-C6-alkyl, C1-C6- haloalkyl and C3-C8-cycloalkyl. In another preferred embodiment of the present invention, wherein for the compound of formula (I- D-a),
, wherein: R1a is selected from fluoro, chloro, bromo, iodo, cyano, methyl, trifluoromethyl, -O-isopropyl, or cyclopropyl; R1b is selected from hydrogen, fluoro, chloro, bromo, cyano, methyl, trifluoromethyl and cyclopropyl;
R1c is selected from hydrogen, fluoro, chloro or bromo or cyano; R2 is selected from hydrogen, or chloro; R3 is selected from hydrogen, chloro, methyl, CH2Cl, or CH2CN; R4a is selected from hydrogen or methyl; R4b is selected from hydrogen; ring C = selected from one of the following rings C-1 to C-7:
trifluoromethyl or cyclopropyl and in ring C-7, R5 can be same or different selected from hydrogen, chloro, methyl, ethyl, trifluoromethyl or cyclopropyl. In one preferred embodiment of the present in Formula (I) or Formula (I-A) to Formula (I-F) is selected from:
D-1 D-2 D-3 D-4 D-6 D-5 D-11 D-7 D-8 D-9 D-10 D-12
D-13 D-14 D-15 D-16 D-17 D-18 D-19 D-20 D-21 D-22 D-23 D-24 In another preferred embodiment, the present invention provided the compound of formula (I-A) to formula (I-F), is particularly described in the below table: Table A describes the
the compounds of formula (I-A) to formula (I- F). Table A: Comp R1a R1b R1c Comp . No. R1a R1b R1c . No. A.1 2-F H H A.13 4-CH3 H H A.2 3-F H H A.14 2-OCH3 H H A.3 4-F H H A.15 3-OCH3 H H A.4 2-Cl H H A.16 4-OCH3 H H A.5 3-Cl H H A.17 2-CHF2 H H A.6 4-Cl H H A.18 3- CHF2 H H A.7 2-Br H H A.19 4- CHF2 H H A.8 3-Br H H A.20 2-CF3 H H A.9 4-Br H H A.21 3- CF3 H H A.10 4-I H H A.22 4- CF3 H H A.11 2-CH3 H H A.23 2-OCHF2 H H A.12 3-CH3 H H A.24 3-OCHF2 H H
Comp 1a 1b 1c Comp R R R R1a R1b R1c . No. . No. A.25 4-OCHF2 H H A.55 4-(C≡C(Me)) H H A.26 2-OCF3 H H A.56 3-C≡C(CyPr) H H A.27 3-OCF3 H H A.57 3-NH(COMe) H H A.28 4-OCF3 H H A.58 4-NH(COCyPr) H H A.29 2-OCH2CF3 H H A.59 3-NHMe H H A.30 3-OCH2CF3 H H A.60 4-NH(CyPr) H H A.31 4-OCH2CF3 H H A.61 3-CONHMe H H A.32 2-SCF3 H H A.62 4-CON(Me)2 H H A.33 3-SCF3 H H A.63 3-CONH(CyPr) H H A.34 4-SCF3 H H A.64 4-CONMe(CyPr) H H A.35 2-CN H H A.65 3-S(O)Me(=NH) H H A.36 3-CN H H A.66 3- H H A.37 4-CN H H S(O)Me(=NCyPr) A.67 3-S(O)Me(=NMe) H H A.38 3-SO2CH3 H H A.68 4-S(O)Et(=NH) H H A.39 4-SO2CH3 H H A.69 4-S(O)Et(=NMe) H H A.40 2-Cyclopropyl H H Cyclopropyl H H A. 4- H H A.41 3- 70 S(O)Et(=NCyPr) A.42 4-Cyclopropyl H H A.71 3-N=S(O)Me2 H H A.43 2-N(Me)2 H H A.72 3-N=S(O)MeCyPr H H A.44 3-N(Me)2 H H A.73 4-N=S(O)Me2 H H A.45 4-N(Me)2 H H A.74 4-N=S(O)MeCyPr H H A.46 2-NO2 H H A.75 2-F 3-F H A.47 3-NO2 H H A.76 2-F 4-F H A.48 4-NO2 H H A.77 2-F 5-F H A.49 2-SMe H H A.78 2-F 6-F H A.50 3-SMe H H A.79 3-F 4-F H A.51 4-SMe H H A.80 3-F 5-F H A.52 3-OCH2(C≡CH) H H A.81 2-Cl 3-Cl H A.53 3- H H A.82 2-Cl 4-Cl H OCH2(CH=CH2) A.83 2-Cl 5-Cl H A.54 4-O-cyclopropyl H H A.84 2-Cl 6-Cl H
Comp 1a 1b 1 Comp R R R c R1a R1b R1c . No. . No. A.85 3-Cl 4-Cl H A.116 2-F 6-CH3 H A.86 3-Cl 5-Cl H A.117 2-Br 5-CH3 H A.87 2-F 3-Cl H A.118 2-CH3 3-Br H A.88 2-F 4-Cl H A.119 3-CH3 5-Br H A.89 2-F 5-Cl H A.120 2-CH3 4-Br H A.90 3-F 4-Cl H A.121 3-CN 4-F H A.91 3-F 5-Cl H A.122 3-CN 4-Cl H A.92 2-Cl 3-F H A.123 2-F 5-CN H A.93 2-Cl 4-F H A.124 2-Cl 5-CN H A.94 2-Cl 5-F H A.125 2-F 4-CN H A.95 3-Cl 4-F H A.126 2-Cl 4-CN H A.96 3-Cl 5-Cl H A.127 3-F 4-CN H A.97 3-Cl 5-CF3 H A.128 3-Cl 4-CN H A.98 2-F 4-Br H A.129 3-Cl 5-CN H A.99 2-F 5-Br H A.130 2-Br 5-CN H A.100 3-F 4-Br H A.131 2-CN 3-Br H A.101 2-Cl 4-Br H A.132 3-CN 5-Br H A.102 2-Cl 5-Br H A.133 2-CN 4-Br H A.103 3-Cl 4-Br H A.134 3-Br 4-CN H A.104 2-Br 4-F H A.135 2-Br 4-CN H A.105 3-Br 4-F H A.136 2-F 3-CF3 H A.106 2-Br 5-Cl H A.137 2-F 4-CF3 H A.107 3-Br 4-Cl H A.138 S-CF3 2-F H A.108 2-F 3-CH3 H A.139 3-F 4-CF3 H A.109 2-F 4-CH3 H A.140 S-CF3 3-F H A.110 2-F 5-CH3 H A.141 4-F 3-CF3 H A.111 2-F 6-CH3 H A.142 2-Cl 3-CF3 H A.112 2-Cl 3-CH3 H A.143 2-Cl 4-CF3 H A.113 2-Cl 4-Me H A.144 3-CF3 5-Br H A.114 2-CH3 4-Cl H A.145 3-CF3 5-CF3 H A.115 2-F 5-CH3 H A.146 3-CF3 4-Br H
PI External A.147 3-CF3 5-Me H A.172 3-OCH3 4-Br H A.148 2-Me 4-CF3 H A.173 5-OCH3 3-Br H A.149 2-Me 5-CF3 H A.174 2-Cl 4-Cl H A.150 3-CF3 4-Me H A.176 2-F 3-F 4-F A.151 3-CF3 5-CN H A.177 2-F 3-F 5-F A.152 3-CN 4-CF3 H A.178 2-F 3-F 6-F A.153 2-OCHF2 4-Cl H A.179 3-F 4-F 5-F A.154 2-OCHF2 4-F H A.180 2-F 4-F 6-F A.155 4-OCF3 2-Cl H A.181 2-Cl 4-F 5-F A.156 S-CF3 2-Cl H A.182 3-Cl 4-F 5-Cl A.157 S-CF3 3-Cl H A.183 S-CF3 3-F 4-F A.158 3-CF3 4-Cl H A.184 4-Me 3-Cl 5-Cl A.159 4-SCH3 2-F H A.185 4-Me 3-F 5-F A.160 4-SCH3 2-Cl H A.186 3-Me 5-Me 4-Cl A.161 4- SO2CH3 2-F H A.187 3-Me 5-Me 4-F A.162 4-SO2CH3 2-Cl H A.188 4-OMe 3-Cl 5-Cl A.163 4-SO2CH3 3-F H A.189 4-OMe 3-F 5-F A.164 4-SO2CH3 3-Cl H A.190 2-Cl 4-Cl 6-Cl A.165 3-SO2CH3, 4-F H A.191 2-Cl 4-Cl 6-Me A.166 3-SO2CH3 4-Cl H A.192 2-O-CF2-O-3 H A.167 5-SO2CH3 2-F H A.193 3-O-CF2-O-4 H A.168 5-SO2CH3 2-Cl H A.194 4-O-CF2-O-5 2-Cl A.169 2-OCH3 4-Cl H A.195 2-CH2-CH2-CH2-3 H A.170 3-OCH3 2-F H A.195 3-CH2-CH2-CH2-4 H A.171 4-OCH3 2-Cl H A.196 2-CH2-CH2-CH2-3, 4-F * CyPr =cyclopropyl The agriculturally acceptable salts of the compounds of formula (I), or formula (I-A) to formula (I-F), or formula (I-D-a) encompass especially the salts of those cations or the acid addition salts of those acids whose cations and anions, respectively, have no adverse effect on the fungicidal action of the compounds of formula (I). or formula (I-A) to formula (I-F) or formula (I-D-a). The salts obtainable in this way likewise have fungicidal properties.
PI External Anions of useful acid addition salts are primarily chloride, bromide, fluoride, hydrogensulfate, sulfate, dihydrogenphosphate, hydrogenphosphate, phosphate, nitrate, bicarbonate, carbonate, hexafluorosilicate, hexafluorophosphate, benzoate, and the anions of C1-C4-alkanoic acids, preferably formate, acetate, propionate and butyrate. They can be formed by reacting a compound of formula (I) or any one of the formula (I-A) to formula (I-F) or formula (I-D-a), with an acid of the corresponding anion, preferably of hydrochloric acid, hydrobromic acid, sulfuric acid, phosphoric acid, or nitric acid. The compounds of the present invention may be present either in pure form or as mixtures of different possible isomeric forms such as stereoisomers e.g. a racemate, individual stereoisomers, or constitutional isomers or as an optically active form. The various stereoisomers include enantiomers, diastereomers, chiral isomers, atropisomers, conformers, rotamers, tautomers, optical isomers, polymorphs, and geometric isomers. Any desired mixtures of these isomers fall within the scope of the claims of the present disclosure. One skilled in the art will appreciate that one stereoisomer may be more active and/or may exhibit beneficial effects when enriched relative to the other isomer(s) or when separated from the other isomer(s). Additionally, the person skilled in the art knows processes or methods or technologies to separate, enrich, and/or to selectively prepare said isomers. The compounds of formula (I) or formula (I-A) to formula (I-F) or formula (I-D-a) can be present in different crystal modifications whose biological activity may differ. They also form part of the subject matter of the present invention. The compounds of formula (I) or formula (I-A) to formula (I-F) or formula (I-D-a) can be present in atropisomers arising from restricted rotation about a single bond of asymmetric groups. They also form part of the subject matter of the present invention. The compounds selected from formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) (including all stereoisomers, N-oxides, and salts thereof), may typically exist in more than one form. The compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) thus include all crystalline and non-crystalline forms of the compound that formula (I), or formula (I-A) to formula (I- F), or formula (I-D-a). Non-crystalline forms include embodiments which are solids such as waxes and gums as well as embodiments which are liquids such as solutions and melts. Crystalline forms include embodiments which represent essentially a single crystal type and embodiments which represent a mixture of polymorphs (i.e. different crystalline types). The following schemes illustrate approaches for preparing compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a). The following descriptions and examples are provided for illustrative purposes and should not be construed as limiting in terms of substituents or substitution patterns. Further, the mentioned reagents, solvents and reaction conditions are intended for the purpose of exemplification only and should not be construed as limiting. In one embodiment, the present invention provides a process for the synthesis of compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a).
PI External The following schemes illustrate approaches for preparing the compounds of formula (I), or formula (I- A) to formula (I-F) or formula (I-D-a). The following descriptions and examples are provided for illustrative purposes and should not be construed as limiting in terms of substituents or substitution patterns. Further, the mentioned reagents, solvents and reaction conditions are intended for the purpose of exemplification only and should not be construed as limiting. The compounds of the present invention as defined by formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and/or in the table 1 may be prepared, in a known manner, in a variety of ways as described in the schemes 1-6. The definitions of B, R1a-R1c, R2, R3, R4 and Z1-Z3 in the compounds of formulae (I), (I-A to I-F, I-D-a), are as defined above in the detailed description of the invention unless otherwise stated specifically. Scheme-1:
In one of the preferred embodiments, the compounds of general formula (I-A) can be prepared by acid- amine coupling reactions between an amine compound of formula 1 (or a salt thereof) and a compound of general formula 2 wherein X represents -OH or -halo group in the presence or absence of coupling reagents such as 1-propanephosphonic acid cyclic anhydride, hexafluorophosphate azabenzotriazole tetramethyl uronium, N-(3-dimethylaminopropyl)-N′-ethylcarbodiimide hydrochloride, and the like or alkyl chloroformates such as ethyl chloroforamate, isobutylchloroformate, and the like and a solvent such as dichloromethane, tetrahydrofuran, N,N-dimethyl formamide etc. These transformations are typically run at a temperature ranging from 0 °C to 25 °C. Scheme-2:
As shown in scheme 2, the compounds of general formula (I-A) can also be prepared by ester-amine coupling reactions between an amine compound of formula 1 (or a salt thereof, particularly HCl salt) and a compound of general formula 3, wherein R is alkyl or aryl group in the presence or absence of a base such as LiHMDS, KOt-Bu, and the like, and in the presence or absence of activating reagents such
PI External as Lewis acids. These reactions are carried out in various solvents such as toluene, tetrahydrofuran, methanol and the like. These reactions are run typically at a temperature ranging from 0 °C to 120 °C. Scheme-3:
According to scheme 3, the compounds represented by general formula (I-A) can also be prepared by transition metal catalyzed C-N coupling of an amine of formula 4 and aryl halide or pseudo-halide compound of formula 5. The compounds of formula 5 with required substitution pattern can either be purchased from commercial sources or can be prepared using literature protocols. Another requisite coupling partner of general formula 4 can be constructed via acid-amine coupling reaction between an acid or acyl halide of formula 10 and an amine of general formula 1 (or salt thereof preferably its hydrochloric acid salt). The C-N coupling reactions are usually carried out in the presence of solvents, while the solvents which can be used for this reaction are not particularly limited as long as they do not adversely affect the reaction. For example, ethers such as 1,4-dioxane, tetrahydrofuran, ethylene glycol, dimethyl ether and diethylene glycol dimethyl ether; aromatic hydrocarbons such as benzene, toluene and xylene; N- amides such as N,N-dimethylformamide, N-dimethylacetamide and 1-methyl-2-pyrrolidone; alcohols such as methanol, ethanol, propanol, butanol, 2-propanol and 2-methyl-2-propanol; nitriles such as acetonitrile; or water or a mixture thereof may serve this purpose. Preferable solvents include aromatic hydrocarbons such as toluene and xylene; and ethers such as 1,4-dioxane, tetrahydrofuran; out of which toluene and 1,4-dioxane are the most preferable ones. The C-N coupling reaction is carried out in the presence of a base selected from, but not limited to, metal alkoxides such as sodium methoxide, sodium ethoxide, potassium tert-butoxide, and sodium tert- butoxide; inorganic bases such as sodium hydroxide, potassium hydroxide, sodium carbonate, potassium carbonate, cesium carbonate, sodium phosphate, tribasic potassium phosphate, potassium hydrogenphosphate, and potassium hydride; organic bases such as triethylamine, N,N- diisopropylethylamine and pyridine. Preferred bases include metal alkoxides such as potassium tert- butoxide, sodium tert-butoxide and inorganic bases such as cesium carbonate and tribasic potassium phosphate. The palladium catalysts that can be used for this C-N coupling reaction include, but are not limited to, inorganic palladium salts such as palladium chloride; organic palladium complexes such as palladium
PI External acetate; tetrakis(triphenylphosphine)palladium(0), bis (triphenylphosphine)palladium(II) chloride, 1,1'- bis (diphenylphosphino) phenylpalladium (II) chloride, and tris(dibenzylidene acetone)dipalladium(0). Palladium catalysts such as palladium chloride, palladium acetate, tetrakis (triphenylphosphine) palladium (0), bis (triphenylphosphine) palladium (II) chloride, 1'- bis (diphenylphosphino) phenylpalladium (II) chloride and tris (dibenzylidene acetone) dipalladium (0) are more preferable. The C-N coupling reaction can also be carried out using further ligands in combination with the above- mentioned catalysts for a fruitful transformation. A list of possible ligands for this transformation include, but are not limited to, tris(o-tolyl)phosphine, tricyclohexylphosphine, tri-tert- butylphosphonium tetrafluoroborate, (oxydi-2,1-phenylene)bis[dicyclohexyl] phosphine, 4,5- bis(diphenylphosphino)-9,9-dimethylxanthene, 2-dicyclohexylphosphino-2′-(N,N- dimethylamino)biphenyl, 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl, 2- dicyclohexylphosphino-2′,6′-dimethoxybiphenyl, 2-dicyclohexylphosphino-2′,6′- diisopropoxybiphenyl, 2-di-tert-butylphosphino-2′,4′,6′-triisopropylbiphenyl, 2-di-tert- butylphosphino-3,4,5,6-tetramethyl-2′,4′,6′-triisopropyl-1,1′-biphenyl, (2-biphenylyl) di-tert- butylphosphine; 2-(di-tert-butylphosphino)biphenyl, 2-di-tert-butylphosphino-3,4,5,6-tetramethyl- 2′,4′,6′-triisopropyl-1,1′-biphenyl, 1,1′-ferrocenediyl-bis(diphenylphosphine), 2-di-tert- butylphosphino-2′-methylbiphenyl, 2-methyl-2′-dicyclohexylphosphinobiphenyl and [1,1′-biphenyl]- 3-sulfonic acid, 2′-(dicyclohexylphosphino)-2,6-dimethoxy- sodium salt. The preferred ligands include 2-(di-tert-butylphosphino)biphenyl, 2-dicyclohexylphosphino-2′,4′,6′-triisopropylbiphenyl, 2-di-tert- butylphosphino-3,4,5,6-tetramethyl-2′,4′,6′-triisopropyl-1,1′-biphenyl (Oxydi-2,1- phenylene)bis[dicyclohexyl] phosphine, 1,1′-ferrocenediyl-bis(diphenylphosphine and 4,5- bis(diphenylphosphino)-9,9-dimethylxanthene. Pre-catalysts can also be utilized for such transformations. A list of possible pre-catalysts include, but are not limited to, (SP-4-3)-[dicyclohexyl[3,6-dimethoxy- 2′,4′,6′-tris(1-methylethyl)[1,1′-biphenyl]-2-yl]phosphine-κP](methanesulfonato-κO)[2′- (methylamino-κN)[1,1′-biphenyl]-2-yl-κC], [(2-di-cyclohexylphosphino-3,6-dimethoxy-2′,4′,6′- triisopropyl-1,1′-biphenyl)-2-(2′-amino-1,1′ -biphenyl)]palladium(II) methanesulfonate methanesulfonate, [(2-di-tert-butylphosphino-3,6-dimethoxy-2′,4′,6′-triisopropyl-1,1′-biphenyl)-2-(2′- amino-1,1′-biphenyl)]palladium(II) methanesulfonate, [2-(di-1-adamantylphosphino)-2′,4′,6′- triisopropyl-3,6-dimethoxybiphenyl][2-(2′-amino-1,1′-biphenyl)]palladium(II) methanesulfonate, [2′- (amino-κN)[1,1′-biphenyl]-2-yl-κC][2′-[(1,1-dimethylethyl)phenylphosphino-κP]-N2, N2, N6, N6- tetramethyl[1,1′-biphenyl]-2,6-diamine](methanesulfonato-κO)palladium, [dicyclohexyl[3-(1- methylethoxy)-2′,4′,6′-tris(1-methylethyl)[1,1′-biphenyl]-2-yl]phosphine-κP](methanesulfonato- κO)[2′-(methylamino-κN)[1,1′-biphenyl]-2-yl-κC] palladium.
PI External Halogens, such as, chloro-, bromo-, and iodo-, are the trivial leaving groups (LG) that are used in this transformation. However, it is also possible to use tosylates, mesylates, and triflates, in somewhat similar manner, as leaving groups allowing fruitful transformations. The reactions can be performed at a temperature range of 0-200 °C, preferably at a temperature range of 50-150 °C for a duration of time between 30 minutes to 24 hours. Scheme-4:
As shown in scheme 4, the compound denoted by general formula (I-A) can also be synthesized through oxidation of the active methylene group (for instance benzylic -CH2-) as present in the compound of formula 7 in the presence or absence of metal oxides such as Iron oxide, Manganese oxide, Selenium dioxide etc or in the presence of oxygen atmosphere. These types of transformations are typically carried out in solvents such as water, ethyl acetate, acetonitrile, dimethyl sulfoxide at a temperature in the range of 25 °C to 100 °C. Scheme-5:
In one of the preferred embodiments, the compound represented by formula (I-C) can be prepared via acid-amine coupling reaction of an amine (or it’s Hydrochloric acid salt) of formula 8 and an acid or acyl halide denoted by general formula 2 in the presence of commonly used coupling reagents and conditions. Scheme-6:
In another preferred embodiment, the compound represented by formula (I-D) can also be prepared via amide coupling reaction of an amine (or salt thereof particularly Hydrochloric acid salt) of formula 9
PI External and an acid or acyl halide denoted by general formula 2 under commonly used coupling reaction conditions. Scheme-7:
In another preferred embodiment, a compound of formula 9A of the present invention can be prepared by treating a compound of formula 11 with a compound of formula 12 in a solvent and a base to obtain a compound of formula 13 as shown in scheme-7. The compound of formula 13 is then subjected to Suzuki coupling reaction with an intermediate compound of formula 14 using a transition metal catalyst and a base, followed by ester hydrolysis and decarboxylation to obtain a compound of formula 17. The compound of formula 17 is converted to the compound of formula 9A using mild and economically cheaper reagents such as iodine and NaBH4 in a suitable solvent preferably selected from tetrahydrofuran or 1,4-dioxane. Scheme-8:
PI External
. In another preferred embodiment, the compound of formula 9A of the present invention can also be prepared according to Scheme-8, wherein a compound of formula 13 is subjected to ester hydrolysis and subsequently to decarboxylation to obtain a compound of formula 19 which is then converted to a compound of formula 17 using a transitional metal catalyst and a base in a solvent under Suzuki C-C coupling reaction conditions. The compound of formula 17 is finally reduced using economically cheaper reagents such as iodine and NaBH4 in a suitable solvent preferably tetrahydrofuran or 1,4- dioxane to obtain the compound of formula 9A. Scheme-9:
In yet another preferred embodiment, a compound of formula 9A obtained by the above scheme-7 or scheme-8, can be reacted with a compound of formula 2A in the presence of a suitable reagent and a solvent as shown in Scheme-9 to obtain a compound of formula (I-D-a).
PI External In one embodiment the present invention, it provides a method for preparing a compound of formula (1), wherein said method comprising the step of: A. reducing a compound of formula (1A) with NaBH4 and iodine (I2) in a solvent to obtain a compound of formula (1),
wherein, B represents a phenyl ring or a 5- or 6-membered aromatic heterocyclic ring; R2 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R3 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C1-C6-cyanoalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6- haloalkynyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R4a and R4b are each independently selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; or R4a and R4b may together form an oxo group (=O), or a 3- to 6-membered carbocyclic ring. In one embodiment the present invention, it provides a method for preparing a compound of formula (1), wherein said method comprising the step of: A. reducing a compound of formula (1A) with NaBH4 and iodine (I2) in a solvent to obtain a compound of formula (1),
wherein B represents a phenyl ring or a 5-membered aromatic heterocyclic ring, preferably phenyl or thiophenyl (thienyl) ring; and R2, R3, R4a and R4b are as defined above, preferably R4b is hydrogen.
PI External The compounds of the present disclosure may be applied by a variety of known techniques, either as the compounds themselves or as formulations comprising these compounds. For example, the compounds may be applied to the roots or foliage of plants for the control of various fungi, without damaging the commercial value of the plants. The materials may be applied in the form of any of the generally used formulation types, for example, as solutions, dusts, wettable powders, flowable concentrates, or emulsifiable concentrates. Preferably, the compounds of the present disclosure are applied in the form of a formulation, comprising one or more of the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) together with a phytologically acceptable carrier. Concentrated formulations may be dispersed in water, or other liquids, for application, or formulations may be dust-like or granular, which may then be applied without further treatment. The formulations can be prepared according to procedures that are conventional in the agricultural chemical art. The present disclosure contemplates all vehicles by which one or more of the compounds may be formulated for delivery and use as a fungicide. Typically, formulations are applied as aqueous suspensions or emulsions. Such suspensions or emulsions may be produced from water-soluble, water- suspendible, or emulsifiable formulations which are solids, usually known as wettable powders; or liquids, usually known as emulsifiable concentrates, aqueous suspensions, or suspension concentrates. As will be readily appreciated, any material to which these compounds may be added may be used, provided it yields the desired utility without significant interference with the activity of these compounds as antifungal agents. In one embodiment, the present invention provides an agrochemical composition comprising a compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), agriculturally acceptable salts, constitutional isomers, stereo-isomers, diastereoisomers, enantiomers, chiral isomers, atropisomers, conformers, rotamers, tautomers, optical isomers, polymorphs, geometric isomers, or N- oxides thereof, optionally with one or more additional active ingredient(s), and optionally together with an auxiliary such as an inert carrier or any other essential ingredient(s) such as surfactants, additives, solid diluents and liquid diluents. An agrochemical composition comprises a fungicidally effective amount of a compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a). The term "effective amount" denotes an amount of the composition or of the compound of formula (I), or formula (I-A) to formula (I-F) or formula (I- D-a), which is sufficient for controlling harmful fungi on cultivated plants or in the protection of materials and which does not result in a substantial damage to the treated plants. Such an amount can vary in a broad range and is dependent on various factors, such as the fungal species to be controlled,
PI External the treated cultivated plant or material, the climatic conditions, and the specific compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) being used. In another embodiment, the present invention provides a composition comprising at least one compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and seeds. The amount of the compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) in the composition ranges from 0.1 g ai (gram per active ingredient) to 1 kg ai (kilogram per active ingredient) per 100 kg of seeds. The compounds of formula (I), formula (I-A) or formula (I-B formula (I), or formula (I-A) to formula (I-F), or formula (I-D-a), their N-oxides, isomers, polymophs or the agriculturally acceptable salts thereof can be converted into customary types of agrochemical compositions, e. g. into solutions, emulsions, suspensions, dusts, powders, pastes, granules, pressings, capsules, and mixtures thereof. Examples for such composition types are suspensions (e. g. SC, OD, FS), emulsifiable concentrates (e. g. EC), emulsions (e. g. EW, EO, ES, ME), capsules (e. g. CS, ZC), pastes, pastilles, wettable powders or dusts (e. g. WP, SP, WS, DP, DS), pressings (e. g. BR, TB, DT), granules (e. g. WG, SG, GR, FG, GG, MG), insecticidal articles (e. g. LN), as well as gel formulations for the treatment of plant propagation materials such as seeds (e. g. GF). These and further composition types are defined in the "Catalogue of pesticide Formulation types and international coding system", Technical Monograph No. 2, 6th Ed. May 2008, CropLife International. The mentioned compositions are prepared in a known manner, such as described by Mollet and Grubemann, “Formulation Technology”, Wiley VCH, Weinheim, 2001; or Knowles, “New Developments in Crop Protection Product Formulation”, Agrow Reports DS243, T&F Informa, London, 2005. Examples for suitable auxiliaries for formulations and/or agrochemical compositions according to the invention are solvents, liquid carriers, solid carriers or fillers, surfactants, dispersants, emulsifiers, wetters, adjuvants, solubilizers, penetration enhancers, protective colloids, adhesion agents, thickeners, humectants, repellents, attractants, feeding stimulants, compatibilizers, bactericides, anti-freezing agents, anti-foaming agents, colorants, tackifiers and binders. The agrochemical compositions generally comprise between 0.01 and 95%, preferably between 0.1 and 90%, and in particular between 0.5 and 75%, by weight of active ingredient (ai). The active ingredients (ai) are employed in a purity from 90% to 100%, preferably from 95% to 100% (according to NMR spectrum). For the purposes of treatment of plant propagation materials, particularly seeds, solutions for seed treatment (LS), suspoemulsions (SE), flowable concentrates (FS), powders for dry treatment (DS), water-dispersible powders for slurry treatment (WS), water-soluble powders (SS), emulsions (ES),
PI External emulsifiable concentrates (EC), and gels (GF) are usually employed. The compositions in question give, after two-to-tenfold dilution, active substance concentrations from 0.01 to 60% by weight, preferably from 0.1 to 40%, in the ready-to-use preparations. When employed in plant protection, the amounts of active substances applied are, depending on the kind of effect desired, from 0.001 to 2 kg per ha, preferably from 0.005 to 2 kg per ha, more preferably from 0.05 to 1.0 kg per ha, and in particular from 0.1 to 0.5 kg per ha. In the treatment of plant propagation materials such as seeds, e. g. by dusting, coating or drenching seed, the generally required amounts of active substance are ranging from 0.1 to 1000 g, preferably from 1 to 1000 g, more preferably from 1 to 100 g and most preferably from 5 to 100 g, per 100 kg of plant propagation material (preferably seeds). When used in the protection of materials or stored products, the amount of active substance applied depends on the kind of application area and on the desired effect. Amounts customarily applied in the protection of materials are ranging from 0.001 g to 2 kg, preferably from 0.005 g to 1 kg, of active substance per cubic meter of treated material. Various types of oils, wetters, adjuvants, fertilizer, or micronutrients, and further pesticides (e. g. herbicides, insecticides, fungicides, growth regulators, safeners, biopesticides) may be added to the active substances or the compositions comprising them as premix or, if appropriate not until immediately prior to use (tank mix). These agents can be mixed with the composition according to the invention in a weight ratio of 1:100 to 100:1, preferably 1:20 to 20:1. A pesticide is generally a chemical or biological agent (such as pesticidally active ingredient, compound, composition, virus, bacterium, antimicrobial or disinfectant) that through its effect deters, incapacitates, kills or otherwise discourages pests. Target pests can include insects, plant pathogens, weeds, mollusks, birds, mammals, fish, nematodes (roundworms), and microbes that destroy property, cause nuisance, spread disease or are vectors for disease. The term "pesticide" includes also plant growth regulators that alter the expected growth, flowering, or reproduction rate of plants; defoliants that cause leaves or other foliage to drop from a plant, usually to facilitate harvest; desiccants that promote drying of living tissues, such as unwanted plant tops; plant activators that activate plant physiology for defense against certain pests; safeners that reduce unwanted herbicidal action of pesticides on crop plants; and plant growth promoters that affect plant physiology e.g. to increase plant growth, biomass, yield or any other quality parameter of the harvestable goods of a crop plant. The user applies the composition according to the invention usually from a predosage device, a knapsack sprayer, a spray tank, a spray plane, or an irrigation system. Usually, the agrochemical
PI External composition is made up with water, buffer, and/or further auxiliaries to the desired application concentration and the ready-to-use spray liquor or the agrochemical composition according to the invention is thus obtained. Usually, 20 to 2000 liters, preferably 50 to 400 liters, of the ready-to-use spray liquor are applied per hectare of agricultural useful area. In one embodiment, the present invention provides a combination comprising the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and at least one further pesticidally active substance selected from the group consisting of fungicides, insecticides, nematicides, acaricides, biopesticides, herbicides, safeners, plant growth regulators, antibiotics, fertilizers and nutrients. The compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), the combinations and the compositions thereof comprising them in the use as fungicides with other fungicides may result in an expansion of the fungicidal spectrum of activity being obtained or in a prevention of fungicide resistance development. Furthermore, in many cases, extraordinary and unexpected effects are obtained. The present invention also relates to agrochemical combinations comprising at least one compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), and at least one further pesticidally active substance selected from the group of fungicides, insecticides, nematicides, acaricides, biopesticides, herbicides, safeners, plant growth regulators, antibiotics, fertiliers and nutrients. The pesticidally active substances reported in WO2015185485 pages 36-43 and WO2017093019 pages 42- 56 can be used in conjunction with the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a). The active substances referred to as component 2, their preparation and their activity e. g. against harmful fungi is known (cf.: http://www.alanwood.net/pesticides/); these substances are commercially available. The compounds described by IUPAC nomenclature, their preparation and their pesticidal activity are also known in prior art. The present invention furthermore relates to agrochemical mixtures comprising at least one compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) (component 1) and at least one further active substance useful for plant protection. By applying the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) together with at least one pesticidally active compound an additional effect can be obtained. This can be obtained by applying the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and at least one further pesticidally active substance simultaneously, either jointly (e. g. as tank-mix) or separately, or in succession, wherein the time interval between the individual applications is selected to ensure that the active substance applied first still occurs at the site of action in a sufficient amount at the time of application of the further pesticidally active substance(s). The order of application is not essential for working of the present invention.
PI External When applying the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and a pesticidally active substance sequentially the time between both applications may vary e. g. between 2 hours to 7 days. Also, a broader range is possible ranging from 0.25 hour to 30 days, preferably from 0.5 hour to 14 days, particularly from 1 hour to 7 days or from 1.5 hours to 5 days, even more preferred from 2 hours to 1 day. In the binary mixtures and the composition according to the invention the weight ratio of the component 1) and the component 2) generally depends on the properties of the active components used, usually it is in the range of 1:1000 to 1000:1, often in the range of 1:100 to 100:1, regularly in the range of 1:50 to 50:1, preferably in the range of 1:20 to 20:1, more preferably in the range of 1:10 to 10:1, even more preferably in the range of 1:4 to 4:1 and in particular in the range of 1:2 to 2:1. According to a further embodiment of the binary mixtures and the composition thereof, the weight ratio of the component 1) and the component 2) usually is in the range of 1000:1 to 1:1000, often in the range of 100:1 to 1:100, regularly in the range of 50:1 to 1:50, preferably in the range of 20:1 to 1:20, more preferably in the range of 10:1 to 1:10, even more preferably in the range of 4:1 to 1:4 and in particular in the range of 2:1 to 1:2. In the ternary mixtures, i.e. the composition according to the invention comprising the (component 1 and component 2) and a compound III (component 3), the weight ratio of component 1) and component 2) depend of the properties of the active substances being used. Usually it is in the range of 1:100 to 100:1, regularly in the range of 1:50 to 50:1, preferably in the range of 1:20 to 20:1, more preferably in the range of 1:10 to 10:1 and in particular in the range of 1:4 to 4:1 and 1:2 to 2:1, and the weight ratio of component 1) and component 3) usually is in the range of 1:100 to 100:1, regularly in the range of 1:50 to 50:1, preferably in the range of 1:20 to 20:1, more preferably in the range of 1:10 to 10:1 and in particular in the range of 1:4 to 4:1 as well as 1:2 to 2:1. Any further active components are, if desired, added in a ratio of 20:1 to 1:20 to the component 1. These ratios are also suitable for inventive mixtures applied by seed treatment. According to one embodiment, individual components of the composition according to the invention such as parts of a kit or parts of a binary or ternary mixture may be mixed by the user himself in a spray tank or any other kind of vessel used for applications (e. g. seed treater drums, seed pelleting machinery, knapsack sprayer). Further auxiliaries may be added, if appropriate. Consequently, one embodiment of the invention is a kit for preparing a usable pesticidal composition, comprising a) a composition comprising component 1) as defined herein and at least one auxiliary; and b) a composition comprising component 2) as defined herein and at least one auxiliary; and optionally c) a composition comprising at least one auxiliary and optionally a further active component 3) as defined herein.
PI External Application of the compounds of formula (I), the combinations and the compositions thereof can be carried out before or during sowing. Methods for applying the compounds of formula (I), the combinations and the compositions thereof, respectively, are application onto plant propagation material, especially seeds, including dressing, coating, pelleting, dusting, and soaking as well as in- furrow application methods. Preferably, the compounds of formula (I), the combinations and the compositions thereof, respectively, are applied on to the plant propagation material by a method such that germination is not induced, e. g. by seed dressing, pelleting, coating and dusting. In one embodiment, the present invention provides a method for controlling or preventing infestation of plants by phytopathogenic microorganisms in agricultural crops and/or horticultural crops wherein an effective amount of at least one compound of formula (I) or the combinations or the composition, is applied to the plants, to parts thereof or to a locus thereof. In another embodiment, the present invention provides a method for controlling or preventing infestation of plants by phytopathogenic microorganisms in agricultural crops and or horticultural crops wherein an effective amount of at least one compound of formula (I) or the combination or the composition, is applied to the seeds of plants. The compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and the compositions according to the invention, respectively, are suitable as fungicides. They are distinguished by an outstanding effectiveness against a broad spectrum of phytopathogenic fungi, including soil-borne fungi, which derive especially from the classes of the Plasmodiophoromycetes, Peronosporomycetes (syn. Oomycetes), Chytridiomycetes, Zygomycetes, Ascomycetes, Basidiomycetes and Deuteromycetes (syn. Fungi imperfecti). Some are systemically active and they can be used in crop protection as foliar fungicides, fungicides for seed dressing and soil fungicides. Moreover, they are suitable for controlling harmful fungi, which inter alia occur in wood or roots of plants. The compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and the compositions according to the invention are particularly important in the control of a multitude of phytopathogenic fungi on various cultivated plants, such as cereals, e. g. wheat, rye, barley, triticale, oats or rice; beet, e. g. sugar beet or fodder beet; fruits, such as pomes, stone fruits or soft fruits, e. g. apples, pears, plums, peaches, almonds, cherries, strawberries, raspberries, blackberries or gooseberries; leguminous plants, such as lentils, peas, alfalfa or soybeans; oil plants, such as rape, mustard, olives, sunflowers, coconut, cocoa beans, castor oil plants, oil palms, ground nuts or soybeans; cucurbits, such as squashes, cucumber or melons; fiber plants, such as cotton, flax, hemp or jute; citrus fruit, such as oranges, lemons, grapefruits or mandarins; vegetables, such as spinach, lettuce, asparagus, cabbages, carrots, onions, tomatoes, potatoes, cucurbits or paprika; lauraceous plants, such as avocados, cinnamon or camphor; energy and raw material plants, such as corn, soybean, rape, sugar cane or oil palm; corn;
PI External tobacco; nuts; coffee; tea; bananas; vines (table grapes, grape juice and grape vines); hop; turf; sweet leaf (also called Stevia); natural rubber plants or ornamental and forestry plants, such as flowers, shrubs, broad-leaved trees or evergreens, e. g. conifers; and on the plant propagation material, such as seeds, and the crop material of these plants. Particularly, the compounds of formula (I), or formula (I-A) to formula (I-F) and the composition according to the invention are important in the control of phytopathogenic fungi on cereals (e. g. wheat, rye, barley, triticale, oats or rice) and soybeans and on the plant propagation material, such as seeds, and the crop material of cereals and soybeans. Preferably, the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) and composition thereof, respectively are used for controlling a multitude of fungi on field crops, such as potatoes sugar beets, tobacco, wheat, rye, barley, oats, rice, corn, cotton, soybeans, rape, legumes, sunflowers, coffee or sugar cane; fruits; vines; ornamentals; or vegetables, such as cucumbers, tomatoes, beans or squashes. The term "plant propagation material" is to be understood to denote all the generative or reproductive parts of the plant such as seeds and vegetative plant material such as cuttings and tubers (e. g. potatoes), which can be used for the multiplication of the plant. This includes seeds, roots, fruits, tubers, bulbs, rhizomes, shoots, sprouts, twigs, flowers, and other parts of plants, including seedlings and young plants, which are to be transplanted after germination or after emergence from soil. These young plants may also be protected before transplantation by a total or partial treatment by immersion or pouring. Preferably, treatment of plant propagation materials with the compounds of formula (I), or formula (I- A) to formula (I-F) or formula (I-D-a), the combinations and or the compositions thereof, respectively, is used for controlling a multitude of fungi on cereals, such as wheat, rye, barley and oats; on rice, corn, cotton and soybeans. The term "cultivated plants" is to be understood as including plants which have been modified by breeding, mutagenesis or genetic engineering including but not limiting to agricultural biotech products on the market or in development (cf. http://cera-gmc.org/, see GM crop database therein). Genetically modified plants are plants, which genetic material has been so modified by recombinant DNA techniques that otherwise cannot readily be obtained by cross breeding under natural circumstances, mutations or natural recombination. Typically, one or more genes have been integrated into the genetic material of a genetically modified plant to improve certain properties of the plant. Such genetic modifications also include but are not limited to targeted post-translational modification of protein(s), oligo-or polypeptides e. g. by glycosylation or polymer additions such as prenylated, acetylated or farnesylated moieties or PEG moieties. Plants that have been modified by breeding, mutagenesis or genetic engineering, e. g. have been rendered tolerant to applications of specific classes of herbicides, such as auxin herbicides such as dicamba or 2,4-D; bleacher herbicides such as hydroxylphenylpyruvate
PI External dioxygenase (HPPD) inhibitors or phytoene desaturase (PDS) inhibitors; acetolactate synthase (ALS) inhibitors such as sulfonyl ureas or imidazolinones; enolpyruvylshikimate-3-phosphate synthase (EPSPS) inhibitors, such as glyphosate; glutamine synthetase (GS) inhibitors such as glufosinate; protoporphyrinogen-IX oxidase inhibitors; lipid biosynthesis inhibitors such as acetyl CoA carboxylase (ACCase) inhibitors; or oxynil (i. e. bromoxynil or ioxynil) herbicides as a result of conventional methods of breeding or genetic engineering. Furthermore, plants have been made resistant to multiple classes of herbicides through multiple genetic modifications, such as resistance to both glyphosate and glufosinate or to both glyphosate and a herbicide from another class such as ALS inhibitors, HPPD inhibitors, auxin herbicides, or ACCase inhibitors. These herbicide resistance technologies are e. g. described in Pest Managem. Sci.61, 2005, 246; 61, 2005, 258; 61, 2005, 277; 61, 2005, 269; 61, 2005, 286; 64, 2008, 326; 64, 2008, 332; Weed Sci.57, 2009, 108; Austral. J. Agricult. Res.58, 2007, 708; Science 316, 2007, 1185; and references quoted therein. Several cultivated plants have been rendered tolerant to herbicides by conventional methods of breeding (mutagenesis), e. g. Clearfield® summer rape (Canola, BASF SE, Germany) being tolerant to imidazolinones, e. g. imazamox, or ExpressSun® sunflowers (DuPont, USA) being tolerant to sulfonyl ureas, e. g. tribenuron. Genetic engineering methods have been used to render cultivated plants such as soybean, cotton, corn, beets and rape, tolerant to herbicides such as glyphosate and glufosinate, some of which are commercially available under the trade names RoundupReady® (glyphosate-tolerant, Bayer CropScience), Cultivance® (imidazolinone tolerant, BASF SE, Germany) and LibertyLink® (glufosinate-tolerant, BASF, Germany). Furthermore, plants capable to synthesize one or more insecticidal proteins, especially those known from the bacterial genus (Bacillus), by use of recombinant DNA techniques are within the scope of the present invention. The Bacillus are particularly from Bacillus thuringiensis, such as δ-endotoxins, e. g. CrylA(b), CrylA(c), CrylF, CrylF(a2), CryllA(b), CrylllA, CrylllB(bl) or Cry9c; vegetative insecticidal proteins (VIP), e. g. VIP1, VIP2, VIP3 or VIP3A; insecticidal proteins of bacteria colonizing nematodes, e. g. Photorhabdus spp. or Xenorhabdus spp.; toxins produced by animals, such as scorpion toxins, arachnid toxins, wasp toxins, or other insect-specific neurotoxins; toxins produced by fungi, such Streptomycetes toxins, plant lectins, such as pea or barley lectins; agglutinins; proteinase inhibitors, such as trypsin inhibitors, serine protease inhibitors, patatin, cystatin or papain inhibitors; ribosome-inactivating proteins (RIP), such as ricin, maize-RIP, abrin, luffin, saporin or bryodin; steroid metabolism enzymes, such as 3-hydroxysteroid oxidase, ecdysteroid-IDP-glycosyl-transferase, cholesterol oxidases, ecdysone inhibitors or HMG-CoA-reductase; ion channel blockers, such as blockers of sodium or calcium channels; juvenile hormone esterase; diuretic hormone receptors (helicokinin receptors); stilbene synthase, bibenzyl synthase, chitinases or glucanases. In the context of the present invention these insecticidal proteins or toxins are to be understood expressly also as pre- toxins, hybrid proteins, truncated or otherwise modified proteins. Hybrid proteins are characterized by
PI External a new combination of protein domains, (see, e. g. WO02/015701). Further examples of such toxins or genetically modified plants capable of synthesizing such toxins are disclosed, e. g., in EP374753, WO93/007278, WO95/34656, EP427529, EP451878, WO03/18810 und WO03/52073. The methods for producing such genetically modified plants are generally known to the person skilled in the art and are described, e. g. in the publications mentioned above. These insecticidal proteins contained in the genetically modified plants impart to the plants, producing these proteins, tolerance to harmful pests from all taxonomic groups of arthropods, especially to beetles (Coeloptera), two-winged insects (Diptera), and moths (Lepidoptera) and to nematodes (Nematoda). Genetically modified plants capable to synthesize one or more insecticidal proteins are, e. g., described in the publications mentioned above, and some of which are commercially available such as YieldGard® (corn cultivars producing the CrylAb toxin), YieldGard® Plus (corn cultivars producing CrylAb and Cry3Bb1 toxins), Starlink® (corn cultivars producing the Cry9c toxin), Herculex® RW (corn cultivars producing Cry34Ab1, Cry35Ab1 and the enzyme phosphinothricin-N-acetyltransferase [PAT]); NuCOTN® 33B (cotton cultivars producing the CrylAc toxin), Bollgard® I (cotton cultivars producing the Cry1 Ac toxin), Bollgard® II (cotton cultivars producing CrylAc and Cry2Ab2 toxins); VIPCOT® (cotton cultivars producing a VIP- toxin); NewLeaf®(potato cultivars producing the Cry3A toxin); Bt-Xtra®, NatureGard®, KnockOut®, BiteGard®, Protecta®, Bt11 (e. g. Agrisure® CB) and Bt176 from Syngenta Seeds SAS, France, (corn cultivars producing the CrylAb toxin and PAT enyzme), MIR604 from Syngenta Seeds SAS, France (corn cultivars producing a modified version of the Cry3A toxin, c.f. WO 03/018810), MON 863 from Monsanto Europe S.A., Belgium (corn cultivars producing the Cry3Bb1 toxin), IPC 531 from Monsanto Europe S.A., Belgium (cotton cultivars producing a modified version of the CrylAc toxin) and 1507 from Pioneer Overseas Corporation, Belgium (corn cultivars producing the Cry1 F toxin and PAT enzyme). Furthermore, plants capable to synthesize one or more proteins to increase the resistance or tolerance of those plants to bacterial, viral or fungal pathogens by the use of recombinant DNA techniques are also within the scope of the present invention. Examples of such proteins are the so-called "pathogenesis-related proteins" (PR proteins, see, e. g. EP392225), plant disease resistance genes (e. g. potato cultivars, which express resistance genes acting against Phytophthora infestans derived from the Mexican wild potato Solanum bulbocastanum) or T4-lysozym (e. g. potato cultivars capable of synthesizing these proteins with increased resistance against bacteria such as Erwinia amylvora). The methods for producing such genetically modified plants are generally known to the person skilled in the art and are described, e. g. in the publications mentioned above. Furthermore, plants capable to synthesize one or more proteins, by use of recombinant DNA techniques, to increase the productivity (e. g. biomass production, grain yield, starch content, oil content or protein content), tolerance to drought, salinity or other growth-limiting environmental factors or tolerance to
PI External pests and fungal, bacterial or viral pathogens of those plants are within the scope of the present invention. Furthermore, plants that contain a modified quantity of substances of content or new substances of content, by the use of recombinant DNA techniques, to improve human or animal nutrition, e. g. oil crops that produce health-promoting long-chain omega-3 fatty acids or unsaturated omega-9 fatty acids (e. g. Nexera® rape, DOW Agro Sciences, Canada) are also within the scope of the present invention. Furthermore, plants that contain a modified quantity of substances of content or new substances of content, by the use of recombinant DNA techniques, to improve raw material production, e. g. potatoes that produce increased amounts of amylopectin (e. g. Amflora® potato, BASF SE, Germany) are also within the scope of the present invention. The compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) may be, for example, effective against fungi and fungal vectors of disease as well as phytopathogenic bacteria and viruses. These fungi and fungal vectors of disease as well as phytopathogenic bacteria and viruses are for example: Absidia corymbifera, Alternaria spp, Aphanomyces spp, Ascochyta spp, Aspergillus spp. including A. flavus, A. fumigatus, A. nidulans, A. niger, A. terms, Aureobasidium spp. including A. pullulans, Blastomyces dermatitidis, Blumeria graminis, Bremia lactucae, Botryosphaeria spp. including B. dothidea, B. obtusa, Botrytis spp. inclusing B. cinerea, Candida spp. including C. albicans, C. glabrata, C. krusei, C. lusitaniae, C. parapsilosis, C. tropicalis, Cephaloascus fragrans, Ceratocystis spp, Cercospora spp. including C. arachidicola, Cercosporidium personatum, Cladosporium spp, Claviceps purpurea, Coccidioides immitis, Cochliobolus spp, Colletotrichum spp. including C. musae, Cryptococcus neoformans, Colletotrichum capsici, Diaporthe spp, Didymella spp, Drechslera spp, Elsinoe spp, Epidermophyton spp, Erwinia amylovora, Erysiphe spp. including E. cichoracearum, Eutypa lata, Fusarium spp. including F. culmorum, F. graminearum, F. langsethiae, F. moniliforme, F. oxysporum, F. proliferatum, F. subglutinans, F. solani, Gaeumannomyces graminis, Gibberella fujikuroi, Gloeodes pomigena, Gloeosporium musarum, Glomerella cingulate, Guignardia bidwellii, Gym nosporangium juniperi-virginianae, Helminthosporium spp, Hemileia spp, Histoplasma spp. including H. capsulatum, Laetisaria fuciformis, Leptographium lindbergi, Leveillula taurica, Lophodermium seditiosum, Microdochium nivale, Microsporum spp, Monilinia spp, Mucor spp, Mycosphaerella spp. including M. graminicola, M. pomi, Oncobasidium theobromaeon, Ophiostoma piceae, Paracoccidioides spp, Penicillium spp. including P. digitatum, P. italicum, Petriellidium spp, Peronosclerospora spp. Including P. maydis, P. philippinensis and P. sorghi, Peronospora spp, Parastagonospora nodorum, Phaeosphaeria nodorum, Phakopsora pachyrhizi, Phellinus igniarus, Phialophora spp, Phoma spp, Phomopsis viticola, Phytophthora spp. including P. infestans, Plasmopara spp. including P. halstedii, P. viticola, Pleospora spp., Podosphaera spp. including P.
PI External leucotricha, Polymyxa graminis, Polymyxa betae, Pseudocercosporella herpotrichoides, Pseudomonas spp, Pseudoperonospora spp. including P. cubensis, P. humuli, Pseudopeziza tracheiphila, Puccinia spp. including P. hordei, P. recondita, P. striiformis, P. triticina, Pyrenopeziza spp, Pyrenophora spp, Pyricularia spp. including P. oryzae, Pythium spp. including P. ultimum, Ramularia spp, Rhizoctonia spp, Rhizomucor pusillus, Rhizopus arrhizus, Rhynchosporium spp, Scedosporium spp. including S. apiospermum and S. prolificans, Schizothyrium pomi, Sclerotinia spp, Sclerotium spp, Septoria spp, including S. nodorum, S. tritici, Septoria lycopersici, Sphaerotheca macularis, Sphaerotheca fusca (Sphaerotheca fuliginea), Sporothorix spp, Stagonospora nodorum, Stemphylium spp., Stereum hirsutum, Thanatephorus cucumeris, Thielaviopsis basicola, Tilletia spp, Trichoderma spp., including T. harzianum, T. pseudokoningii, T. viride, Trichophyton spp, Typhula spp, Uncinula necator, Urocystis spp, Ustilago spp, Venturia spp. including V. inaequalis, Verticillium spp., and Xanthomonas spp, Ustilaginales such as Ustilaginoidea virens, Ustilago nuda, Ustilago tritici, Ustilago zeae, Cerotelium fici, Chrysomyxa arctostaphyli, Coleosporium ipomoeae, Hemileia vastatrix, Puccinia arachidis, Puccinia cacabata, Puccinia graminis, Puccinia sorghi, Puccinia striiformis f.sp. Hordei, Puccinia striiformis f.sp. Secalis, Pucciniastrum coryli, or Uredinales such as Cronartium ribicola, Gymnosporangium juniperi-viginianae, Melampsora medusae, Phragmidium mucronatum, Physopella ampelosidis, Tranzschelia discolor and Uromyces viciae-fabae, Cryptococcus spp., Exobasidium vexans, Marasmiellus inoderma, Mycena spp., Sphacelotheca reiliana, Typhula ishikariensis, Urocystis agropyri, ltersonilia perplexans, Corticium invisum, Waitea circinata, Rhizoctonia solani, Thanetephorus cucurmeris, Entyloma dahliae, Entylomella microspora, Neovossia moliniae and Tilletia caries, Blastocladiomycetes, such as Physoderma maydis, Mucoromycetes, such as Choanephora cucurbitarum. Non-limiting examples of pathogens of fungal diseases which can be treated in accordance with the invention include the diseases caused by rust disease pathogens, for example Gymnosporangium species, for example Gymnosporangium sabinae; Hemileia species, for example Hemileia vastatrix; Phakopsora species, for example Phakopsora pachyrhizi or Phakopsora meibomiae; Puccinia species, for example Puccinia recondita, Puccinia graminis oder Puccinia striiformis; Uromyces species, for example Uromyces appendiculatus. In particular, Cronartium ribicola (White pine blister rust); Gymnosporangium juniperi-virginianae (Cedar-apple rust); Hemileia vastatrix (Coffee rust); Phakopsora meibomiae and P. pachyrhizi (Soybean rust); Puccinia coronata (Crown Rust of Oats and Ryegrass); Puccinia graminis (Stem rust of wheat and Kentucky bluegrass, or black rust of cereals); Puccinia hemerocallidis (Daylily rust); Puccinia persistens subsp. triticina (wheat rust or 'brown or red rust'); Puccinia sorghi (rust in corn); Puccinia striiformis ('Yellow rust' in cereals); Uromyces appendiculatus (rust of beans); Uromyces phaseoli (Bean rust); Puccinia melanocephala ('Brown rust' in sugarcane); Puccinia kuehnii ('Orange rust' in sugarcane).
PI External The present invention further relates to the use of the compounds of formula (I), the combinations or the compositions thereof for controlling or preventing against phytopathogenic fungi such as Septoria spp., Blumeria spp., Podosphaera spp., Sphaerotheca spp., Uncinula spp., Erysiphe spp., Erysiphe spp., Microsphaera diffusa spp., Botrytis spp., Colletotrichum spp, Alternaria spp., Venturia inaequalis spp., and Monilinia spp. of agricultural crops and or horticultural crops. The present invention further relates to the use of the compounds of formula (I), the combinations or the compositions thereof for controlling or preventing against phytopathogenic fungi such as Septoria spp., Blumeria spp., Podosphaera spp., Sphaerotheca spp., Uncinula spp., Erysiphe spp., Erysiphe spp., Microsphaera diffusa spp., Botrytis spp., Colletotrichum spp, Alternaria spp., Venturia inaequalis spp., and Monilinia spp. in cereals, grapevines, fruits, nuts and vegetables. Plants which can be treated in accordance with the invention include the following: cotton, flax, grapevine, fruits, vegetables, such as Rosaceae sp (for example pome fruits such as apples, pears, apricots, cherries, almonds and peaches), Ribesioidae sp., Juglandaceae sp., Betulaceae sp., Anacardiaceae sp., Fagaceae sp., Moraceae sp., Oleaceae sp., Actinidaceae sp., Lauraceae sp., Musaceae sp. (for example banana trees and plantations), Rubiaceae sp. (for example coffee), Theaceae sp., Sterculiceae sp., Rutaceae sp. (for example lemons, oranges and grapefruit); Vitaceae sp. (for example grapes); Solanaceae sp. (for example tomatoes, peppers), Liliaceae sp., Asteraceae sp. (for example lettuce), Umbelliferae sp., Cruciferae sp., Chenopodiaceae sp., Cucurbitaceae sp. (for example cucumber), Alliaceae sp. (for example leek, onion), Papilionaceae sp. (for example peas); major crop plants, such as Poaceae/Gramineae sp. (for example maize, turf, cereals such as wheat, rye, rice, barley, oats, millet and triticale), Asteraceae sp. (for example sunflower), Brassicaceae sp. (for example white cabbage, red cabbage, broccoli, cauliflower, Brussels sprouts, pak choi, kohlrabi, radishes, and oilseed rape, mustard, horseradish and cress), Fabacae sp. (for example bean, peanuts), Papilionaceae sp. (for example soya bean), Solanaceae sp. (for example potatoes), Chenopodiaceae sp. (for example sugar beet, fodder beet, swiss chard, beetroot); Malvaceae (for example cotton); useful plants and ornamental plants for gardens and wooded areas; and genetically modified varieties of each of these plants. More preference is given to controlling the following diseases of soya beans: Fungal diseases on leaves, stems, pods and seeds caused, for example, by Altemaria leaf spot (Altemaria spec. atrans tenuissima), Anthracnose (Colletotrichum gloeosporoides dematium var. truncatum), brown spot (Septoria glycines ), cercospora leaf spot and blight ( Cercospora kikuchii), choanephora leaf blight (Choanephora infundibulifera trispora (Syn.)), dactuliophora leaf spot (Dactuliophora glycines), downy mildew (Peronospora manshurica), drechslera blight (Drechslera glycini), frogeye leaf spot (Cercospora sojina), leptosphaerulina leaf spot (Leptosphaerulina trifolii), phyllostica leaf spot (Phyllosticta sojaecola), pod and stem blight (Phomopsis sojae), powdery mildew (Microsphaera diffusa),
PI External pyrenochaeta leaf spot (Pyrenochaeta glycines), rhizoctonia aerial, foliage, and web blight (Rhizoctonia solani), rust (Phakopsora pachyrhizi, Phakopsora meibomiae), scab (Sphaceloma glycines), stemphylium leaf blight (Stemphylium botryosum), target spot (Corynespora cassiicola). Fungal diseases on roots and the stem base caused, for example, by black root rot (Calonectiia crotalariae), charcoal rot (Macrophomina phaseolina), fusarium blight or wilt, root rot, and pod and collar rot (Fusarium oxysporum, Fusarium orthoceras, Fusarium semitectum, Fusarium equiseti), mycoleptodiscus root rot (Mycoleptodiscus terrestris), neocosmospora (Neocosmospora vasinfecta), pod and stem blight (Diaporthe phaseolorum), stem canker (Diaporthe phaseolorum var. caulivora), phytophthora rot (Phytophthora megasperma), brown stem rot (Phialophora gregata), pythium rot (Pythium aphanidennatum, Pythium irregulare, Pythium debaryanum, Pythium myriotylum, Pythium ultimum), rhizoctonia root rot, stem decay, and damping-off (Rhizoctonia solani), sclerotinia stem decay (Sclerotinia sclerotiorum), sclerotinia southern blight (Sclerotinia rolfsii), thielaviopsis root rot (Thielaviopsis basicola). The present invention also relates to the use of the compounds of formula (I), the combinations or the compositions thereof for controlling or preventing the following plant diseases: Puccinia spp. (rusts) on various plants, for example, but not limited to P. triticina (brown or leaf rust), P. striiformis (stripe or yellow rust), P. hordei (dwarf rust), P. graminis (stem or black rust) or P. recondita (brown or leaf rust) on cereals, such as e. g. wheat, barley or rye and Phakopsoraceae spp. on various plants, in particular Phakopsora pachyrhizi and P. meibomiae (soybean rust) on soybeans, Hemileia vastatrix (Coffee rust), Uromyces appendiculatus, Uromyces fabae and Uromyces phaseoli (rust of beans). The compounds, the combinations and the compositions of the present invention can be used for controlling or preventing plant diseases. The compounds of formula (I), or formula (I-A) to formula (I- F) or formula (I-D-a), the combinations and/or the compositions thereof, respectively, are particularly suitable for controlling the following plant diseases: Albugo spp. (white rust) on ornamentals, vegetables (e. g. A. Candida) and sunflowers (e. g. A. tragopogonis); Altemaria spp. (Alternaria leaf spot) on vegetables, rape (A. brassicola or brassicae), sugar beets (A. tenuis), fruits, rice, soybeans, potatoes (e. g. A. solani or A. alternata), tomatoes (e. g. A. solani or A. alternata) and wheat; Aphanomyces spp. on sugar beets and vegetables; Ascochyta spp. on cereals and vegetables, e. g. A. tritici (anthracnose) on wheat and A. hordei on barley; Bipolaris and Drechslera spp. (teleomorph: Cochliobolus spp.), e. g. Southern leaf blight (D. maydis) or Northern leaf blight (B. zeicola) on corn, e. g. spot blotch (C. sorokiniana) on cereals and e. g. B. oryzae on rice and turfs; Blumeria (formerly Erysiphe) graminis (powdery mildew) on cereals (e. g. on wheat or barley); Botrytis cinerea (teleomorph: Botryotinia fuckeliana: grey mold) on fruits and berries (e. g. strawberries), vegetables (e. g. lettuce, carrots, celery and cabbages), rape, flowers, vines, forestry plants
PI External and wheat; Bremia lactucae (downy mildew) on lettuce; Ceratocystis (syn. Ophiostoma) spp. (rot or wilt) on broad-leaved trees and evergreens, e. g. C. ulmi (Dutch elm disease) on elms; Cercospora spp. (Cercospora leaf spots) on corn (e. g. Gray leaf spot: C. zeae-maydis), rice, sugar beets (e. g. C. beticola), sugar cane, vegetables, coffee, soybeans (e. g. C. sojina or C. kikuchii) and rice; Cladosporium spp. on tomatoes (e. g. C. fulvum: leaf mold) and cereals, e. g. C. herbarum (black ear) on wheat; Claviceps purpurea (ergot) on cereals; Cochliobolus (anamorph: Helminthosporium of Bipolaris) spp. (leaf spots) on corn (C. carbonum), cereals (e. g. C. sativus, anamorph: B. sorokiniana) and rice (e. g. C. miyabeanus, anamorph: H. oryzae); Colletotrichum (teleomorph: Glomerella) spp. (anthracnose) on cotton (e. g. C. gossypii), corn (e. g. C. graminicola: Anthracnose stalk rot), soft fruits, potatoes (e. g. C. coccodes: black dot), beans (e. g. C. lindemuthianum) and soybeans (e. g. C. truncatum or C. gloeosporioides); Corticium spp., e. g. C. sasakii (sheath blight) on rice; Corynespora cassiicola (leaf spots) on soybeans and ornamentals; Cycloconium spp., e. g. C. oleaginum on olive trees; Cylindrocarpon spp. (e. g. fruit tree canker or young vine decline, teleomorph: Nectria or Neonectria spp.) on fruit trees, vines (e. g. C. liriodendri, teleomorph: Neonectria liriodendri: Black Foot Disease) and ornamentals; Dematophora (teleomorph: Rosellinia) necatrix (root and stem rot) on soybeans; Diaporthe spp., e. g. D. phaseolorum (damping off) on soybeans; Drechslera (syn. Helminthosporium, teleomorph: Pyrenophora) spp. on corn, cereals, such as barley (e. g. D. teres, net blotch) and wheat (e. g. D. tritici-repentis: tan spot), rice and turf; Esca (dieback, apoplexy) on vines, caused by Formitiporia (syn. Phellinus) punctata, F. mediterranea, Phaeomoniella chlamydospora (earlier Phaeoacremonium chlamydosporum), Phaeoacremonium aleophilum and/or Botryosphaeria obtusa; Elsinoe spp. on pome fruits (E. pyri), soft fruits (E. veneta: anthracnose) and vines (E. ampelina: anthracnose); Entyloma oryzae (leaf smut) on rice; Epicoccum spp. (black mold) on wheat; Erysiphe spp. (powdery mildew) on sugar beets (E. betae), vegetables (e. g. E. pisi), such as cucurbits (e. g. E. cichoracearum), cabbages, rape (e. g. E. cruciferarum); Eutypa lata (Eutypa canker or dieback, anamorph: Cytosporina lata, syn. Libertella blepharis) on fruit trees, vines and ornamental woods; Exserohilum (syn. Helminthosporium) spp. on corn (e. g. E. turcicum); Fusarium (teleomorph: Gibberella) spp. (wilt, root or stem rot) on various plants, such as F. graminearum or F. culmorum (root rot, scab or head blight) on cereals (e. g. wheat or barley), F. oxysporum on tomatoes, F. solani (f. sp. glycines now syn. F. virguliforme) and F. tucumaniae and F. brasiliense each causing sudden death syndrome on soybeans, and F. verticillioides on corn; Gaeumannomyces graminis (take-all) on cereals (e. g. wheat or barley) and corn; Gibberella spp. on cereals (e. g. G. zeae) and rice (e. g. G. fujikuroi: Bakanae disease); Glomerella cingulata on vines, pome fruits and other plants and G. gossypii on cotton; Grainstaining complex on rice; Guignardia bidwellii (black rot) on vines; Gymnosporangium spp. on rosaceous plants and junipers, e. g. G. sabinae (rust) on pears; Helminthosporium spp. (syn. Drechslera, teleomorph: Cochliobolus) on corn, cereals and rice; Hemileia spp., e. g. H. vastatrix (coffee leaf rust) on coffee; Isariopsis clavispora (syn. Cladosporium vitis) on vines; Macrophomina phaseolina (syn. phaseoli) (root and stem rot) on soybeans and cotton; Microdochium (syn. Fusarium) nivale (pink snow mold) on cereals (e. g. wheat
PI External or barley); Microsphaera diffusa (powdery mildew) on soybeans; Monilinia spp., e. g. M. laxa, M. fructicola and M. fructigena (bloom and twig blight, brown rot) on stone fruits and other rosaceous plants; Mycosphaerella spp. on cereals, bananas, soft fruits and ground nuts, such as e. g. M. graminicola (anamorph: Septoria tritici, Septoria blotch) on wheat or M. fijiensis (black Sigatoka disease) on bananas; Peronospora spp. (downy mildew) on cabbage (e. g. P. brassicae), rape (e. g. P. parasitica), onions (e. g. P. destructor), tobacco (P. tabacina) and soybeans (e. g. P. manshurica); Phakopsora pachyrhizi and P. meibomiae (soybean rust) on soybeans; Phialophora spp. e. g. on vines (e. g. P. tracheiphila and P. tetraspora) and soybeans (e. g. P. gregata: stem rot); Phoma lingam (root and stem rot) on rape and cabbage and P. betae (root rot, leaf spot and damping-off) on sugar beets; Phomopsis spp. on sunflowers, vines (e. g. P. viticola: can and leaf spot) and soybeans (e. g. stem rot: P. phaseoli, teleomorph: Diaporthe phaseolorum); Physoderma maydis (brown spots) on corn; Phytophthora spp. (wilt, root, leaf, fruit and stem root) on various plants, such as paprika and cucurbits (e. g. P. capsici), soybeans (e. g. P. megasperma, syn. P. sojae), soybeans, potatoes and tomatoes (e. g. P. infestans: late blight) and broad-leaved trees (e. g. P. ramorum: sudden oak death); Plasmodiophora brassicae (club root) on cabbage, rape, radish and other plants; Plasmopara spp., e. g. P. viticola (grapevine downy mildew) on vines and P. halstedii on sunflowers; Podosphaera spp. (powdery mildew) on rosaceous plants, hop, pome and soft fruits, e. g. P. leucotricha on apples; Polymyxa spp., e. g. on cereals, such as barley and wheat (P. graminis) and sugar beets (P. betae) and thereby transmitted viral diseases; Pseudocercosporella herpotrichoides (eyespot, teleomorph: Tapesia yallundae) on cereals, e. g. wheat or barley; Pseudoperonospora (downy mildew) on various plants, e. g. P. cubensis on cucurbits or P. humili on hop; Pseudopezicula tracheiphila (red fire disease or .rotbrenner', anamorph: Phialophora) on vines; Puccinia spp. (rusts) on various plants, e. g. P. triticina (brown or leaf rust), P. striiformis (stripe or yellow rust), P. hordei (dwarf rust), P. graminis (stem or black rust) or P. recondita (brown or leaf rust) on cereals, such as e. g. wheat, barley or rye, P. kuehnii (orange rust) on sugar cane and P. asparagi on asparagus; Pyrenophora (anamorph: Drechslera) tritici- repentis (tan spot) on wheat or P. teres (net blotch) on barley; Pyricularia spp., e. g. P. oryzae (teleomorph: Magnaporthe grisea, rice blast) on rice and P. grisea on turf and cereals; Pythium spp. (damping-off) on turf, rice, corn, wheat, cotton, rape, sunflowers, soybeans, sugar beets, vegetables and various other plants (e. g. P. ultimum or P. aphanidermatum); Ramularia spp., e. g. R. collo-cygni (Ramularia leaf spots, Physiological leaf spots) on barley and R. beticola on sugar beets; Rhizoctonia spp. on cotton, rice, potatoes, turf, corn, rape, potatoes, sugar beets, vegetables and various other plants, e. g. R. solani (root and stem rot) on soybeans, R. solani (sheath blight) on rice or R. cerealis (Rhizoctonia spring blight) on wheat or barley; Rhizopus stolonifer (black mold, soft rot) on strawberries, carrots, cabbage, vines and tomatoes; Rhynchosporium secalis (scald) on barley, rye and triticale; Sarocladium oryzae and S. attenuatum (sheath rot) on rice; Sclerotinia spp. (stem rot or white mold) on vegetables and field crops, such as rape, sunflowers (e. g. S. sclerotiorum) and soybeans (e. g. S. rolfsii or S. sclerotiorum); Septoria spp. on various plants, e. g. S. glycines (brown spot) on
PI External soybeans, S. tritici (Septoria blotch) on wheat and S. (syn. Stagonospora) nodorum (Stagonospora blotch) on cereals; Uncinula (syn. Erysiphe) necator (powdery mildew, anamorph: Oidium tuckeri) on vines; Setospaeria spp. (leaf blight) on corn (e. g. S. turcicum, syn. Helminthosporium turcicum) and turf; Sphacelotheca spp. (smut) on corn, (e. g. S. reiliana: head smut), sorghum und sugar cane; Sphaerotheca fuliginea (powdery mildew) on cucurbits; Spongospora subterranea (powdery scab) on potatoes and thereby transmitted viral diseases; Stagonospora spp. on cereals, e. g. S. nodorum (Stagonospora blotch, teleomorph: Leptosphaeria [syn. Phaeosphaeria] nodorum) on wheat; Synchytrium endobioticum on potatoes (potato wart disease); Taphrina spp., e. g. T. deformans (leaf curl disease) on peaches and T. pruni (plum pocket) on plums; Thielaviopsis spp. (black root rot) on tobacco, pome fruits, vegetables, soybeans and cotton, e. g. T. basicola (syn. Chalara elegans); Tilletia spp. (common bunt or stinking smut) on cereals, such as e. g. T. tritici (syn. T. caries, wheat bunt) and T. controversa (dwarf bunt) on wheat; Typhula incarnata (grey snow mold) on barley or wheat; Urocystis spp., e. g. U. occulta (stem smut) on rye; Uromyces spp. (rust) on vegetables, such as beans (e. g. U. appendiculatus, syn. U. phaseoli) and sugar beets (e. g. U. betae); Ustilago spp. (loose smut) on cereals (e. g. U. nuda and U. avaenae), corn (e. g. U. maydis: corn smut) and sugar cane; Venturia spp. (scab) on apples (e. g. V. inaequalis) and pears; and Verticillium spp. (wilt) on various plants, such as fruits and ornamentals, vines, soft fruits, vegetables and field crops, e. g. V. dahliae on strawberries, rape, potatoes and tomatoes. In one embodiment, the compounds of formula (I) have broad ranges of activity against fungal pathogens. Exemplary pathogens may include, but are not limited to, causing agent of wheat leaf blotch (Zymoseptoria tritici), wheat brown rust (Puccinia triticina), wheat stripe rust (Puccinia striiformis), scab of apple (Venturia inaequalis), powdery mildew of grapevine (Uncinula necator), barley scald (Rhynchosporium secalis), blast of rice (Pyricularia oryzae), rust of soybean (Phakopsora pachyrhizi), glume blotch of wheat (Leptosphaeria nodorum), powdery mildew of wheat (Blumeria graminis f. sp. tritici), powdery mildew of barley (Blumeria graminis f. sp. hordei), powdery mildew of cucurbits (Erysiphe cichoracearum), anthracnose of cucurbits (Colletotrichum lagenarium), leaf spot of beet (Cercospora beticola), early blight of tomato (Alternaria solani), and spot blotch of barley (Cochliobolus sativus). The exact amount of the active material to be applied is dependent not only on the specific active material being applied, but also on the particular action desired, the fungal species to be controlled, and the stage of growth thereof, as well as the parts of the plant or other products to be contacted with the compound. Thus, all the compounds, and formulations containing the same, may not be equally effective at similar concentrations or against the same fungal species. The compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), the combinations and the compositions thereof, respectively, are also suitable for controlling harmful fungi in the protection of stored products or harvest and in the protection of materials. The term "protection of
PI External materials" is to be understood to denote the protection of technical and non-living materials, such as adhesives, glues, wood, paper and paperboard, textiles, leather, paint dispersions, plastics, cooling lubricants, fibre or fabrics, against the infestation and destruction by harmful microorganisms, such as fungi and bacteria. As to the protection of wood and other materials, the particular attention is paid to the following harmful fungi: Ascomycetes such as Ophiostoma spp., Ceratocystis spp., Aureobasidium pullulans, Sclerophoma spp., Chaetomium spp., Humicola spp., Petriella spp., Trichurus spp.; Basidiomycetes such as Coniophora spp., Coriolus spp., Gloeophyllum spp., Lentinus spp., Pleurotus spp., Pora spp., Serpula spp. and Tyromyces spp., Deuteromycetes such as Aspergillus spp., Cladosporium spp., Penicillium spp., Trichoderma spp., Altemaria spp., Paecilomyces spp. and Zygomycetes such as Mucor spp., and in addition in the protection of stored products and harvest the following yeast fungi are worthy of note: Candida spp. and Saccharomyces cerevisae. In one embodiment, the present invention provides a method for controlling or preventing phytopathogenic fungi. The method comprises treating the fungi or the materials, plants, plant parts, locus thereof, soil or seeds to be protected against fungal attack, with an effective amount of at least one compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), or a combination or a composition comprising at least one compound of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a). The method of treatment according to the invention can also be used in the field of protecting stored products or harvest against attack of fungi and microorganisms. According to the present invention, the term "stored products" is understood to denote natural substances of plant or animal origin and their processed forms, which have been taken from the natural life cycle and for which long-term protection is desired. Stored products of crop plant origin, such as plants or parts thereof, for example stalks, leafs, tubers, seeds, fruits or grains, can be protected in the freshly harvested state or in processed form, such as pre-dried, moistened, comminuted, ground, pressed or roasted, which process is also known as post- harvest treatment. Also falling under the definition of stored products is timber, whether in the form of crude timber, such as construction timber, electricity pylons and barriers, or in the form of finished articles, such as furniture or objects made from wood. Stored products of animal origin are hides, leather, furs, hairs and the like. The combinations according to the present invention can prevent disadvantageous effects such as decay, discoloration or mold. Preferably "stored products" is understood to denote natural substances of plant origin and their processed forms, more preferably fruits and their processed forms, such as pomes, stone fruits, soft fruits and citrus fruits and their processed forms.
PI External It is also possible to use the compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I- D-a) as a fungicide. The term “fungicide” as used herein means a compound that controls, modifies, or prevents the growth of fungi. The compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a), the combinations and the compositions thereof, respectively, may be used for improving the health of a plant. The invention also relates to a method for improving plant health by treating a plant, its propagation material and/or the locus where the plant is growing or is to grow with an effective amount of compound of formula (I), or formula (I-A) to formula (I-F) or formual (I-D-a) and the composition thereof, respectively. The term "plant health" is to be understood to denote a condition of the plant and/or its products which is determined by several indicators alone or in combination with each other such as yield (e. g. increased biomass and/or increased content of valuable ingredients), plant vigor (e. g. improved plant growth and/or greener leaves ("greening effect")), quality (e. g. improved content or composition of certain ingredients) and tolerance to abiotic and/or biotic stress. The above identified indicators for the health condition of a plant may be interdependent or may result from each other. The compounds of formula (I), or formula (I-A) to formula (I-F) or formula (I-D-a) are employed as such or in the form of a composition for treating the fungi or the plants, plant propagation materials, such as seeds, soil, surfaces, materials or rooms to be protected from fungal attack with a fungicidally effective amount of the active substances. The application can be carried out both before and after the infection of the plants, plant propagation materials, such as seeds, soil, surfaces, materials, or rooms by the fungi. Plant propagation material may be treated with the compounds of formula (I), the combinations and the compositions thereof protectively either at or before planting or transplanting. The compounds of the invention can be used in combination with models e.g. embedded in computer programs for site specific crop management, satellite farming, precision farming or precision agriculture. Such models support the site specific management of agricultural sites with data from various sources such as soils, weather, crops (e.g. type, growth stage, plant health), weeds (e.g. type, growth stage), diseases, pests, nutrients, water, moisture, biomass, satellite data, yield etc. with the purpose to optimize profitability, sustainability and protection of the environment. In particular, such models can help to optimize agronomical decisions, control the precision of pesticide applications and record the work performed. As an example, the compounds of the invention can be applied to a crop plant according to an appropriate dose regime if a model models the development of a pest and calculates that a threshold has been reached for which it is recommendable to apply the compound of the invention to the crop plant.
PI External Commercially available systems which include agronomic models are e.g. FieldScriptsTM from The Climate Corporation, XarvioTM from BASF, AGLogicTM from John Deere, etc. The compounds of the invention can also be used in combination with smart spraying equipment such as e.g. spot spraying or precision spraying equipment attached to or housed within a farm vehicle such as a tractor, robot, helicopter, airplane, unmanned aerial vehicle (UAV) such as a drone, etc. Such an equipment usually includes input sensors (such as e.g. a camera) and a processing unit configured to analyze the input data and configured to provide a decision based on the analysis of the input data to apply the compound of the invention to the crop plants (respectively the weeds) in a specific and precise manner. The use of such smart spraying equipment usually also requires positions systems (e.g. GPS receivers) to localize recorded data and to guide or to control farm vehicles; geographic information systems (GIS) to represent the information on intelligible maps, and appropriate farm vehicles to perform the required farm action such as the spraying. In an example, pests can be detected from imagery acquired by a camera. In an example the pests can be identified and/or classified based on that imagery. Such identification and / classification can make use of image processing algorithms. Such image processing algorithms can utilize machine learning algorithms, such as trained neutral networks, decision trees and utilize artificial intelligence algorithms. In this manner, the compounds described herein can be applied only where needed. Any range or desired value given herein may be extended or altered without losing the effects sought, as is apparent to the skilled person for an understanding of the teachings herein. The invention disclosed in the present disclosure shall now be elaborated with the help of non-limiting examples. CHEMISTRY EXAMPLES: Experimental Procedure: The acid intermediates such as 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylic acid, 1-(4- fluorophenyl)-1H-1,2,4-triazole-3-carboxylic acid and 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4- carboxylic acid are prepared according to the procedures reported in WO2018019929Al, WO2014085490A1 and WO2021094434A1 respectively. The bicyclic amine compounds such as 4- (1-methyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine, 4-(1,5-dimethyl-1H-pyrazol-4- yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine, and 7-methyl-4-(1-methyl-1H-pyrazol-4-yl)-4,5,6,7- tetrahydrothieno[2,3-c]pyridine are prepared according to Scheme-7 and Scheme-8, by adopting the procedure described in Example 6 and Example 7. All other starting materials, reagents and solvents are purchased from commercial sources. Example 1: Synthesis of 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylic acid Step 1: Synthesis of ethyl 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylate
PI External A solution of 1-azido-2,4-difluorobenzene (2 g, 12.9 mmol) and ethyl propiolate (1.9 g, 19.3 mmol) in tetrahydrofuran (20 mL) was treated with copper (I) iodide (0.25 g, 1.3 mmol) and stirred at 25 °C for 12 h. After completion of the reaction, the reaction mixture was poured into ice water and the precipitated solid was separated by filteration to obtain ethyl 1-(2,4-difluorophenyl)-1H-1,2,3-triazole- 4-carboxylate (2.1 g, 8.3 mmol, 64% yield) as off-white solid. Step 2: Synthesis of 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylic acid A solution of ethyl 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylate (3 g, 11.8 mmol) in tetrahydrofuran (21 mL) and water (9 mL) was treated with lithium hydroxide monohydrate (1.5 g, 35.5 mmol), and stirred at 25 °C for 3h. After completion of the reaction, tetrahydrofuran was evaporated under reduced pressure, and pH of the residual material was adjusted to 5 - 6 with 10% aqueous hydrochloric acid. The precipitated solid was separated by filtration and dried under reduced pressure to obtain 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylic acid (2.5 g, 11.1 mmol, 94% yield) as off-white solid. Example 2: Synthesis of 1-(4-fluorophenyl)-1H-1,2,4-triazole-3-carboxylic acid Step 1: Synthesis of ethyl 1-(4-fluorophenyl)-1H-1,2,4-triazole-3-carboxylate A solution of 4-fluoroaniline (8 g, 72 mmol) in water (80 mL) was treated sequentially with 10% hydrochloric acid (27 mL) and sodium nitrite (7.45 g, 108 mmol) at 0 °C, and stirred for 30 minutes to obtain the corresponding diazonium salt.In another reaction flask, a solution of sodium acetate (5.9 g, 72 mmol) in water (30 mL) and methanol (80 mL) was treated dropwise with ethyl isocyanoacetate (8.14 g, 72 mmol) at 0 °C, stirred for 5 minutes, and then treated slowly with the solution of the diazonium salt (prepared above) at 0 °C. The resulting reaction mixture was warmed up to 25°C and stirred for 5 h. After completion of the reaction, methanol was removed under reduced pressure and the residual material was extracted with dichloromethane (3 x 100 mL). The combined organic extracts were dried over anhydrous sodium sulfate and evaporated under reduced pressure to give ethyl 1-(4- fluorophenyl)-1H-1,2,4-triazole-3-carboxylate (5.2 g, 23.5 mmol, 33% yield). Step 2: Synthesis of 1-(2,4-difluorophenyl)-1H-1,2,4-triazole-3-carboxylic acid A solution of ethyl 1-(4-fluorophenyl)-1H-1,2,4-triazole-3-carboxylate (1 g, 4.5 mmol) in a mixture of tetrahydrofuran (20 mL), water (10 mL) and methanol (2 mL) was treated with lithium hydroxide (0.54 g, 22.6 mmol) at 25 °C, and stirred for 16 h. After completion of the reaction, the solvents were evaporated under reduced pressure. The resulting residue was dissolved in water (40 mL) and extracted with ethyl acetate (30 mL). The aqueous layer was acidified with 2M aqueous hydrochloric acid to adjust pH 5. The precipitated solid was separated by filteration and dried under reduced pressure to
PI External obtain 1-(4-fluorophenyl)-1H-1,2,4-triazole-3-carboxylic acid (0.5 g, 2.4 mmol, 53% yield) as a white solid. Example 3: Synthesis of 1-(2,4-difluorophenyl)-1H-pyrazole-4-carboxylic acid Step 1: Synthesis of ethyl 1-(2,4-difluorophenyl)-1H-pyrazole-4-carboxylate A solution of (2,4-difluorophenyl) hydrazine hydrochloride (1 g, 5.54 mmol) in ethanol (20 mL) was treated with ethyl 2-formyl-3-oxopropanoate (1.04 g, 7.2 mmol) and sodium acetate (0.45 g, 5.54 mmol) at 25 °C, stirred for 30 minutes, heated to 80 °C, and stirred for 3 h. After completion of the reaction, ethanol was removed under reduced pressure. The residue obtained was diluted with water (50 mL) and extracted with ethyl acetate (3 x 50 mL). The combined organic extracts were dried over anhydrous sodium sulphate and evaporated under reduced pressure to give a crude material. The crude material was then purified by CombiFlash® chromatography on silica gel (0-10% ethyl acetate in hexane) to obtain ethyl 1-(2,4-difluorophenyl)-1H-pyrazole-4-carboxylate (1 g, 3.96 mmol, 72% yield) as a yellow solid. Step 2: Synthesis of 1-(2,4-difluorophenyl)-1H-pyrazole-4-carboxylic acid A solution of ethyl 1-(2,4-difluorophenyl)-1H-pyrazole-4-carboxylate (1 g, 3.96 mmol) in tetrahydrofuran (10 mL) and water (5 mL) was treated with lithium hydroxide monohydrate (0.25 g, 5.95 mmol) at 25 °C, and stirred for 18 h. After completion of the reaction, volatiles were evaporated under reduced pressure, and the residue was acidified with 10% aqueous hydrochloric acid. The precipitated solid product was filtered and dried under reduced pressure to obtain 1-(2,4- difluorophenyl)-1H-pyrazole-4-carboxylic acid (600 mg, 2.68 mmol, 67% yield) as off white solid. Example 4: Synthesis of 2-(2,4-difluorophenyl)-2H-tetrazole-5-carboxylic acid Step 1: Synthesis of ethyl 2-(2,4-difluorophenyl)-2H-tetrazole-5-carboxylate A solution of 2,4-difluoroaniline (1 g, 7.75 mmol) in a 1:1 mixture of ethanol and water (10 mL) was treated slowly with 12M hydrochloric acid (4.6 mL, 56 mmol) and sodium nitrite (0.59 g, 8.52 mmol) at 0 °C, and stirred for 30 minutes to obtain the corresponding diazonium salt solution.In another reaction flask, a solution of ethyl 2-oxoacetate (2.45 g, 24 mmol) in ethanol (5 mL) was treated with 4- methylbenzenesulfonohydrazide (1.44 g, 7.75 mmol) at 0 °C, the resulting reaction mixture was warmed up to 25 °C, and stirred for 30 minutes. The reaction mixture was evaporated under reduced pressure to remove ethanol, and the resulting residual material was dissolved in pyridine (30 mL), cooled to 0 °C. The diazonium salt solution (prepared above) was added slowly to this cold reaction mixture, warmed up to 25 °C, and stirred for 3 h. After completion of the reaction, the reaction mixture was diluted with water (60 mL) and extracted with ethyl acetate (2 x 50 mL). The combined organic
PI External extracts were washed with saturated brine solution (30 mL), dried over sodium sulphate and evaporated under reduced pressure. The residual material thus obtained was purified by CombiFlash® chromatography on silica gel (0-10% ethyl acetate in hexane) to isolate ethyl 2-(2,4-difluorophenyl)- 2H-tetrazole-5-carboxylate (0.6 g, 2.36 mmol, 31% yield) as a brown coloured solid. Step 2: Synthesis of 2-(2,4-difluorophenyl)-2H-tetrazole-5-carboxylic acid A solution of ethyl 2-(2,4-difluorophenyl)-2H-tetrazole-5-carboxylate (0.6 g, 2.36 mmol) in 1:1:1 mixture of ethanol, tetrahydrofuran and water (6 mL) was treated with LiOH (226 mg, 9.44 mmol) at 25 °C, and stirred for 16 h. After completion of the reaction, the volatiles were removed under reduced pressure and the residual material was acidified with 10% aqueous hydrochloric acid solution to pH 4. The precipitated solid was filtered and dried under reduced pressure to obtain 2-(2,4-difluorophenyl)- 2H-tetrazole-5-carboxylic acid (0.3 g, 1.33 mmol, 56% yield) as an off white solid. Example 5: Synthesis of 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4-carboxylic acid Step 1: Synthesis of ethyl (E)-2-(2-(2,4-difluorophenyl)hydrazineylidene)-3-oxopropanoate A solution of 2,4-difluoroaniline (7.9 mL, 77 mmol) in water (100 mL) and 12M hydrochloric acid (20 mL) was treated slowly with a solution of sodium nitrite (5.34 g, 77 mmol) in water (50 mL) at 0 °C and stirred for 5 minutes. This solution was treated with dropwise addition of a solution of ethyl (E)-3- (dimethylamino)acrylate (12.2 g, 85 mmol) and potassium acetate (11.4 g, 116 mmol) in ethanol (150 mL) at 0 °C, and then allowed to warm up to 25 °C, and stirred for 10 minutes. After completion of the reaction, the reaction mixture was diluted with ethyl acetate (400 mL) and washed with water (150 mL) and saturated brine solution (100 mL). The separated organic layer was dried over anhydrous sodium sulfate, filtered and evaporated under reduced pressure to obtain crude ethyl (E)-2-(2-(2,4- difluorophenyl)hydrazineylidene)-3-oxopropanoate (18.92 g, 73.8 mmol, 95% yield). Step 2: Synthesis of ethyl (2E)-2-(2-(2,4-difluorophenyl)hydrazineylidene)-3-(hydroxyimino) propanoate A solution of ethyl (E)-2-(2-(2,4-difluorophenyl)hydrazineylidene)-3-oxopropanoate (18 g, 70 mmol) in ethanol (150 mL) was treated with hydroxylamine hydrochloride (5.86 g, 84 mmol) and potassium acetate (17.24 g, 176 mmol) at 25 °C, heated to 80 °C, and stirred for 30 min. After completion of the reaction, the reaction mixture was diluted with ethyl acetate (500 mL), washed with water (150 mL) and saturated brine solution (100 mL). The separated organic layer was dried over anhydrous sodium sulphate, filtered and evaporated to isolate crude ethyl (2E)-2-(2-(2,4- difluorophenyl)hydrazineylidene)-3-(hydroxyimino)propanoate (16 g, 59 mmol, 84% yield). Step 3: Synthesis of ethyl 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4-carboxylate
PI External A solution of ethyl (2E)-2-(2-(2,4-difluorophenyl)hydrazineylidene)-3-(hydroxyimino) propanoate (1 g, 3.69 mmol) in acetic anhydride (10 mL) was heated to 140 °C, and stirred for 30 minutes. After completion of the reaction, the reaction mixture was cooled to 25 °C and diluted with water (50 mL). The resulting dark solution was extracted with ethyl acetate (3 x 30 mL). The combined organic extracts were dried over anhydrous sodium sulphate and evaporated under reduced pressure to obtain a crude material, which was purified by CombiFlash® chromatography on silica gel (0-20% ethyl acetate in hexane) to isolate ethyl 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4-carboxylate (0.7 g, 2.76 mmol, 75% yield). Step 4: Synthesis of 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4-carboxylic acid A solution of ethyl 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4-carboxylate (9 g, 35.5 mmol) in tetrahydrofuran (100 mL) and water (10 mL) was treated with lithium hydroxide (1.49 g, 35.5 mmol) at 25 °C, and stirred for 2 h. After completion of the reaction, tetrahydrofuran was evaporated under reduced pressure and the residue was acidified with 1M aqueous hydrochloric acid to pH 2. The precipitated solid was filtered, washed with hexane (50 mL) and dried under reduced pressure to afford 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4-carboxylic acid (6 g, 26.6 mmol, 75% yield) as off white solid. Example 6: Synthesis of 4-(1-methyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine
Step a: Synthesis of ethyl 4-bromothieno[2,3-c]pyridine-2-carboxylate A solution of 3,5-dibromoisonicotinaldehyde (8 g, 30.2 mmol) in dimethyl acetamide (70 mL) was treated with K2CO3 (5 g, 36.2 mmol) followed by ethyl thioglycolate (3.64 mL, 33.2 mmol) at 0 °C and stirred at 25 °C for 3 h. After completion of the reaction, the reaction mixture was poured into water (200 mL) and extracted with ethyl acetate (2 x 100 mL). The combined organic layers were washed with brine solution (100 mL), dried over anhydrous sodium sulphate and evaporated under reduced
PI External pressure. The residue was purified by flash column chromatography to isolate ethyl 4-bromothieno[2,3- c]pyridine-2-carboxylate (7.6 g, 26.6 mmol, 88% yield). LCMS (m/z): 287.65 Step b: Synthesis of ethyl 4-(1-methyl-1H-pyrazol-4-yl)thieno[2,3-c]pyridine-2-carboxylate A solution of 1-methyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole (5.51 g, 26.5 mmol), ethyl 4-bromothieno[2,3-c]pyridine-2-carboxylate (6 g, 22.05 mmol) in 1,4-dioxane (70 mL) and water (18 mL), was treated with K2CO3 (9.14 g, 66.1 mmol) and 1,1'- Bis(diphenylphosphino)ferrocene-palladium(II)dichloride dichloromethane complex (1.080 g, 1.323 mmol) at 25 °C and stirred at 70 °C for 1 h. After completion of the reaction, the reaction mixture was filtered through celite bed, and the filtrate was extracted with ethyl acetate (2 x 120 mL). The combined organic phase was washed with brine (80 mL), dried over anhydrous sodium sulphate and evaporated under reduced pressure. The residue was purified by flash column chromatography to obtain ethyl 4- (1-methyl-1H-pyrazol-4-yl)thieno[2,3-c]pyridine-2-carboxylate (6 g, 20.9 mmol, 95% yield). LCMS (m/z): 288.10 Step c: Synthesis of 4-(1-methyl-1H-pyrazol-4-yl)thieno[2,3-c]pyridine-2-carboxylic acid A suspension of ethyl 4-(1-methyl-1H-pyrazol-4-yl)thieno[2,3-c]pyridine-2-carboxylate (6 g, 20.88 mmol) in THF (40 mL) and water (10 mL), was treated with LiOH (0.75 g, 31.3 mmol) at 25 °C and stirred for 3 h. After completion of the reaction, the reaction mixture was concentrated under reduced pressure to evaporate THF, and the residue was acidified with 1 N aq. HCl. The precipitated solid was filtered and dried under reduced pressure to obtain 4-(1-methyl-1H-pyrazol-4-yl)thieno[2,3-c]pyridine- 2-carboxylic acid (5 g, 19.3 mmol, 92% yield). LCMS (m/z): 259.75 Step d: Synthesis of 4-(1-methyl-1H-pyrazol-4-yl)thieno[2,3-c]pyridine A solution of 4-(1-methyl-1H-pyrazol-4-yl)thieno[2,3-c]pyridine-2-carboxylic acid (5 g, 19.3 mmol) in dimethyl sulfoxide (70 mL), was treated with silver carbonate (1.6 g, 5.8 mmol) and acetic acid (0.06 mL, 0.96 mmol) at 25 °C and stirred at 150 °C for 48 h. After completion of the reaction, the reaction mixture was cooled and filtered. The filtrate was diluted with water (200 mL) and extracted with ethyl acetate. The combined organic layer was washed with brine solution (100 mL), dried over anhydrous sodium sulphate and evaporated under reduced pressure to obtain 4-(1-methyl-1H-pyrazol-4- yl)thieno[2,3-c]pyridine (2.82 g, 13.1 mmol, 68% yield). LCMS (m/z): 215.95 Step e: Synthesis of 4-(1-methyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine To a solution of 4-(1-methyl-1H-pyrazol-4-yl)thieno[2,3-c]pyridine (5 g, 23.23 mmol) and iodine (3.54 g, 13.94 mmol) in tetrahydrofuran (25 mL), NaBH4 (3.51 g, 93 mmol) was added slowly at 25 °C and then stirred at 65 °C for 22 h. After completion of the reaction, the reaction mixture was cooled to 25 °C, treated with methanol (200 mL) and stirred for 30 min. The volatiles were evaporated under reduced pressure. The residue obtained was dissolved in 1: 1 water: dioxane (50 mL) and then treated with NaOH (4.64 g, 119 mmol) at 25° C followed by stirring at 90 °C for 16 h. After completion of the reaction, the reaction mixture was cooled to 25° C and extracted with ethyl acetate (2 x 100 mL). The
PI External combined organic layer was dried over anhydrous sodium sulphate and evaporated under reduced pressure to obtain 4-(1-methyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine (4 g, 18.24 mmol, 79% yield). Example 7: Synthesis of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3- c]pyridine (Formula 9A-b)
Step f: Synthesis of 4-bromothieno[2,3-c]pyridine-2-carboxylic acid To a suspension of ethyl 4-bromothieno[2,3-c]pyridine-2-carboxylate (11 g, 38.4 mmol, prepared according to step-1 of Example 6) in tetrahydrofuran (200 mL) and water (50 mL), LiOH (1.381 g, 57.7 mmol) was added and stirred at 25 °C for 5 h. After completion of the reaction, the reaction mixture was concentrated under reduced pressure to remove tetrahydrofuran. The residue obtained was acidified with 1 N aq. HCl solution. The precipitated solid was filtered and dried under reduced pressure to obtain 4-bromothieno[2,3-c]pyridine-2-carboxylic acid (7.5 g, 29.1 mmol, 76% yield). The crude material was used directly for the next step. Step g: Synthesis of 4-bromothieno[2,3-c]pyridine To a solution of 4-bromothieno[2,3-c]pyridine-2-carboxylic acid (5 g, 19.37 mmol) in dimethyl sulfoxide (10 mL), silver carbonate (2.67 g, 9.69 mmol) and acetic acid (0.06 mL, 0.97 mmol) were added at 25 °C. The reaction mixture was then stirred at 150 °C for 16 h. After completion of the reaction, the reaction mixture was filtered through celite bed. The filtrate was diluted with water (100 mL) and extracted with ethyl acetate (3 × 100 mL). The combined organic layer was washed with brine solution (70 mL), dried over anhydrous sodium sulphate and evaporated under reduced pressure to obtain 4-bromothieno[2,3-c]pyridine (3 g, 14.01 mmol, 72% yield). LCMS (m/z):215.65 Step h: Synthesis of 4-(1,5-dimethyl-1H-pyrazol-4-yl)thieno[2,3-c]pyridine To a solution of 1,5-dimethyl-4-(4,4,5,5-tetramethyl-1,3,2-dioxaborolan-2-yl)-1H-pyrazole (14.94 g, 67.3 mmol), 4-bromothieno[2,3-c]pyridine (12 g, 56.1 mmol) and K₂CO₃ (23.24 g, 168 mmol) in 2:1 1,4-dioxane: water (150 mL), 1,1'-Bis(diphenylphosphino)ferrocene-palladium(II) dichloride dichloromethane complex (2.75 g, 3.36 mmol) was added at 25 °C and then stirred at 90 °C for 1 h. After completion of the reaction, the reaction mixture was filtered through celite bed. The filtrate was extracted with ethyl acetate (2 x 150 mL). The combined organic layer was dried over anhydrous sodium sulphate and evaporated under reduced pressure to obtain a residue which was purified by flash column
PI External chromatography to obtain 4-(1,5-dimethyl-1H-pyrazol-4-yl)thieno[2,3-c]pyridine (12 g, 52.3 mmol, 93% yield). LCMS (m/z): 229.85 Step i: Synthesis of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine To a solution of 4-(1,5-dimethyl-1H-pyrazol-4-yl)thieno[2,3-c]pyridine (3.5 g, 15.26 mmol) and iodine (2.32 g, 9.16 mmol) in tetrahydrofuran (20 ml), NaBH4 (2.31 g, 61.1 mmol) was added slowly at 0 °C and then stirred at 60 °C for 20 h. The reaction mixture was cooled to 25 °C and treated with methanol (20 mL) and stirred for 30-45 min. The reaction mixture was concentrated under reduced pressure to obtain a residue which was dissolved in 1:1 mixture of dixoxane:water (50 mL) and treated with NaOH (1.83 g, 45.8 mmol) at 25 °C and stirred at 90 °C for 16 h. After completion of the reaction, the reaction mixture was cooled to 25 °C and extracted with dichloromethane (2 x 50 mL). The combined organic layer was dried over anhydrous sodium sulphate and evaporated under reduced pressure to obtain 4- (1,5-dimethyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine (3.1 g, 13.3 mmol, 87% yield). LCMS (m/z): 234.05. Example 8: Synthesis of 1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3-triazole-4-carboxylic acid Step 1: Synthesis of ethyl 1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3-triazole-4-carboxylate A solution of 2-chloro-4-methylaniline (8 g, 56.5 mmol) in acetonitrile (30 mL) was treated sequentially with TMS-N3 (8.25 mL, 62.1 mmol) and tert-butyl nitrite (7.4 mL, 62.1 mmol) at 0 °C, and stirred for 30 minutes. After completion of the reaction, the reaction mixture was warmed up to 25 °C, diluted with dimethylsulfoxide (30 mL) and water (10 mL), treated with ethyl acetoacetate (10.3 mL, 79 mmol), piperidine (2.24 mL, 22.6 mmol) and potassium carbonate (781 mg, 5.65 mmol) and stirred for 5 minutes. The resulting reaction mixture was heated to 80 °C, stirred for 16 h. After completion of the reaction, the reaction mixture was cooled to 0 °C followed by addition of ice cooled water (20 mL) under stirring. The precipitated solid material was separated by filtration, washed with water and dried to obtain ethyl 1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3-triazole-4-carboxylate (10 g, 35.7 mmol, 63% yield) as off-white solid. Step 2: Synthesis of 1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3-triazole-4-carboxylic acid A solution of ethyl 1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3-triazole-4-carboxylate (500 mg, 1.79 mmol) in a mixture of tetrahydrofuran (8 mL), methanol (4 mL) and water (2 mL) was treated with LiOH (86 mg, 3.57 mmol) at 25 °C, and stirred for 2 h. After completion of the reaction, volatiles were removed under reduced pressure, the residual material was acidified with 1M aqueous hydrochloric acid solution to adjust pH 4 and extracted with ethyl acetate (3 x 30 mL). The combined organic extracts were washed with saturated brine solution (20 mL), dried over anhydrous sodium sulphate, filtered and
PI External concentrated under reduced pressure to obtain 1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3- triazole-4-carboxylic acid (400 mg, 1.59 mmol, 89% yield). Example 9: Synthesis of (1-(2,4-difluorophenyl)-1H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol- 4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 1)
A solution of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-1,2,3,4-tetrahydroisoquinoline hydrochloride (633 mg, 2.4 mmol) and 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylic acid (450 mg, 2 mmol) in dichloromethane (20 mL) was treated sequentially with triethyl amine (1.1 mL, 8 mmol) and 1- propanephosphonic acid cyclic anhydride (T3P, 2.4 mL, 4 mmol) at 25°C, and stirred for 18 h. After completion of the reaction, the reaction mixture was diluted with water (50 mL) and extracted with ethyl acetate (3 x 40 mL). The combined organic layers were washed with brine solution (40 mL), dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure. The crude compound thus obtained was purified by a reverse phase preparative-high performance liquid chromatography to isolate (1-(2,4-difluorophenyl)-1H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)methanone 1 (660 mg, 1.52 mmol, 76% yield) as a beige solid. 1H-NMR Data (400 MHz, DMSO-D6): δ 8.90 (d, J = 104.9 Hz, 1H), 7.99-7.88 (m, 1H), 7.73-7.68 (m, 1H), 7.37 (t, J = 8.1 Hz, 1H), 7.32-7.02 (m, 3H), 6.93 (dd, J = 13.3, 7.5 Hz, 1H), 6.78 (s, 1H), 5.33 (dd, J = 36.3, 17.0 Hz, 1H), 4.93 (dd, J = 20.1, 17.6 Hz, 1H), 4.39-4.23 (m, 1H), 4.26-4.11 (m, 2H), 3.60 (s, 3H), 2.09 (s, 3H). LCMS (m/z): 435.3 (M+H)+ Example 10: Synthesis of (4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)(1-(4- fluorophenyl)-1H-1,2,4-triazol-3-yl)methanone (compound 2)
A solution of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-1,2,3,4-tetrahydroisoquinoline hydrochloride (535 mg, 2.03 mmol) and 1-(4-fluorophenyl)-1H-1,2,4-triazole-3-carboxylic acid (350 mg, 1.69 mmol) in dichloromethane (20 mL) was treated sequentially with triethyl amine (0.94 mL, 6.76 mmol) and 1- propanephosphonic acid cyclic anhydride (2 mL, 3.38 mmol) at 25 °C, and stirred for 18 h. After completion of the reaction, the reaction mixture was diluted with water (60 mL) and extracted with dichloromethane (3 x 80 mL). The combined organic extracts were washed with brine solution (40 mL),
PI External dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure. The crude material thus obtained was purified by a reverse phase preparative-high performance liquid chromatography to isolate (4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)(1-(4- fluorophenyl)-1H-1,2,4-triazol-3-yl)methanone 2 (250 mg, 0.6 mmol, 36% yield) as a white solid. 1H-NMR (400 MHz, DMSO-D6): δ 9.36 (d, J = 26.4 Hz, 1H), 7.95-7.87 (m, 2H), 7.46 (t, J = 8.8 Hz, 2H), 7.32-7.12 (m, 3H), 7.04-6.82 (m, 2H), 5.03-4.95 (m, 1H), 4.85 (d, J = 17.1 Hz, 1H), 4.23-4.08 (m, 1H), 4.01 (dd, J = 13.3, 4.8 Hz, 1H), 3.85-3.77 (m, 1H), 3.60 (s, 3H), 2.01 (s, 3H). LCMS (m/z): 417.25 (M+H)+ Example 11: Synthesis of (1-(2,4-difluorophenyl)-1H-pyrazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol-4- yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 3)
A solution of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-1,2,3,4-tetrahydroisoquinoline hydrochloride (471 mg, 1.78 mmol) and 1-(2,4-difluorophenyl)-1H-pyrazole-4-carboxylic acid (400 mg, 1.78 mmol) in dichloromethane (20 mL) was treated sequentially with triethyl amine (0.75 mL, 5.35 mmol) and 1- propanephosphonic acid cyclic anhydride (2.1 mL, 3.57 mmol) at 25 °C, and stirred for 18 h. After completion of the reaction, the reaction mixture was diluted with water (60 mL) and extracted with dichloromethane (3 x 40 mL). The combined organic extracts were washed with brine solution (30 mL), dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure to obtain a crude material. The crude material thus obtained was purified by a reverse phase preparative-high performance liquid chromatography to isolate (1-(2,4-difluorophenyl)-1H-pyrazol-4-yl)(4-(1,5- dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone 3 (380 mg, 0.88 mmol, 49% yield) as a brown solid. LCMS (m/z): 434.05 (M+H)+ 1H-NMR (400 MHz, DMSO-D6): δ 8.63 (s, 1H), 8.11-7.71 (m, 2H), 7.63-7.58 (m, 1H), 7.31-6.84 (m, 6H), 5.00 (d, J = 15.9 Hz, 2H), 4.73-3.87 (m, 3H), 3.65 (s, 3H), 2.32-2.06 (m, 3H). Example 12: Synthesis of (2-(2,4-difluorophenyl)-2H-tetrazol-5-yl)(4-(1,5-dimethyl-1H-pyrazol-4- yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 4)
PI External
A solution of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-1,2,3,4-tetrahydroisoquinoline hydrochloride (583 mg, 2.21 mmol) and 2-(2,4-difluorophenyl)-2H-tetrazole-5-carboxylic acid (500 mg, 2.21 mmol) in dichloromethane (20 mL) was treated sequentially with triethyl amine (0.9 mL, 6.63 mmol) and 1- propanephosphonic acid cyclic anhydride (2.6 mL, 4.42 mmol) at 0 °C, warmed up to 25 °C, and stirred for 18 h. After completion of the reaction, the reaction mixture was diluted with water (60 mL) and extracted with dichloromethane (3 x 80 mL). The combined organic extracts were washed with saturated brine solution (30 mL), dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure to obtain a crude material. This crude material was purified by a reverse phase preparative- high performance liquid chromatography to isolate (2-(2,4-difluorophenyl)-2H-tetrazol-5-yl)(4-(1,5- dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone 4 (205 mg, 0.471 mmol, 21.29 % yield) as a colorless gum. 1H-NMR (400 MHz, DMSO-D6) δ 8.09 (dtd, J = 28.5, 8.8, 5.8 Hz, 1H), 7.83-7.77 (m, 1H), 7.49-7.44 (m, 1H), 7.35-7.11 (m, 3H), 7.01-6.77 (m, 2H), 5.05-4.90 (m, 2H), 4.25-4.21 (m, 1H), 4.14-4.00 (m, 1H), 3.89-3.83 (m, 1H), 3.65 (d, J = 55.5 Hz, 3H), 2.06 (s, 1H), 1.98 (s, 2H). LCMS (m/z): 436.2 (M+H)+ Example 13: Synthesis of (2-(2,4-difluorophenyl)-2H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol- 4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 5)
A solution of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-1,2,3,4-tetrahydroisoquinoline hydrochloride (422 mg, 1.56 mmol) and 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4-carboxylic acid (300 mg, 1.33 mmol) in dichloromethane (20 mL) was treated sequentially with triethyl amine (0.6 mL, 4 mmol) and 1- propanephosphonic acid cyclic anhydride (1.6 mL, 2.66 mmol) at 25 °C, and stirred for 12 h. After completion of the reaction, water (30 mL) was added to the reaction mixture which then was extracted with dichloromethane (3 x 60 mL). The combined organic extracts were washed with saturated brine solution (50 mL), dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure to obtain a crude material. This crude material was purified by reverse phase preparative-high performance liquid chromatography to isolate (2-(2,4-difluorophenyl)-2H-1,2,3-triazol-4-yl)(4-(1,5-
PI External dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone 5 (540 mg, 1.24 mmol, 93% yield) as a brown gum. 1H-NMR (400 MHz, DMSO-D6) δ 8.29 (d, J = 140.4 Hz, 1H), 7.96 (dtd, J = 45.8, 8.8, 5.9 Hz, 1H), 7.71-7.65 (m, 1H), 7.39-6.69 (m, 6H), 5.20-4.83 (m, 2H), 4.21-3.59 (m, 6H), 2.20-1.94 (m, 3H). LCMS (m/z): 435.15 (M+H)+ Example 14: Synthesis of (1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3-triazol-4-yl)(4-(1-methyl- 1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 6)
A solution of 4-(1-methyl-1H-pyrazol-4-yl)-1,2,3,4-tetrahydroisoquinoline hydrochloride (198 mg, 0.79 mmol) and 1-(2-chloro-4-methylphenyl)-5-methyl-1H-1,2,3-triazole-4-carboxylic acid (200 mg, 0.79 mmol) in dichloromethane (15 mL) was treated with triethyl amine (0.4 mL, 3.18 mmol) and propanephosphonic acid anhydride (0.95 mL, 1.59 mmol) at 25 °C, and stirred for 12 h. After completion of the reaction, the reaction mixture was diluted with water (50 mL) and extracted with dichloromethane (3 x 50 mL). The combined organic extracts were washed with saturated brine solution (50 mL), dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure to obtain a crude material. The crude material thus obtained was then purified by a reverse phase preparative-high performance liquid chromatography to isolate (1-(2-chloro-4-methylphenyl)-5- methyl-1H-1,2,3-triazol-4-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)- yl)methanone 6 (118 mg, 0.26 mmol, 33% yield). 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.42 (s, 1H), 7.29 (t, J = 8.1 Hz, 3H), 7.25-7.11 (m, 5H), 6.99 (s, 1H), 5.44 (dd, J = 39.9, 17.0 Hz, 1H), 5.01 (dd, J = 133.4, 17.3 Hz, 1H), 4.40-4.24 (m, 2H), 3.99-3.93 (m, 1H), 3.77 (s, 3H), 2.45 (s, 3H), 2.22 (s, 3H). LCMS (m/z): 447.1 (M+H)+ Example 15 : Synthesis of (1-(2,4-difluorophenyl)-1H-1,2,3-triazol-4-yl)(7-methyl-4-(1-methyl-1H- pyrazol-4-yl)-4,7-dihydrothieno[2,3-c]pyridin-6(5H)-yl)methanone (compound 7)
A suspension of 7-methyl-4-(1-methyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine (350 mg, 1.5 mmol) and 1-(2,4-difluorophenyl)-1H-1,2,3-triazole-4-carboxylic acid (338 mg, 1.5 mmol) in
PI External dichloromethane (10 mL) was treated sequentially with triethyl amine (0.42 mL, 3 mmol) and propanephosphonic acid anhydride (1 mL, 1.65 mmol) at 25 °C, and stirred for 18 h. After completion of the reaction, water (40 mL) was added to the reaction mixture, which then was extracted with dichloromethane (3 x 30 mL). The combined organic extracts were washed with saturated brine solution (20 mL), dried over anhydrous sodium sulphate and evaporated to isolate a crude material. This crude material was then purified by a reverse phase preparative-high performance liquid chromatography to obtain (1-(2,4-difluorophenyl)-1H-1,2,3-triazol-4-yl)(7-methyl-4-(1-methyl-1H-pyrazol-4-yl)-4,7- dihydrothieno[2,3-c]pyridin-6(5H)-yl)methanone 7 (210 mg, 0.48 mmol, 32% yield). 1H-NMR (400 MHz, DMSO-D6) δ 9.07-8.36 (m, 1H), 7.98-6.57 (m, 7H), 6.01-5.75 (m, 1H), 4.98-3.48 (m, 6H), 1.77-1.52 (m, 3H). LCMS (m/z): 441.2 (M+H)+ Example 16 : Synthesis of (1-(4-fluorophenyl)-1H-1,2,4-triazol-3-yl)(7-methyl-4-(1-methyl-1H- pyrazol-4-yl)-4,7-dihydrothieno[2,3-c]pyridin-6(5H)-yl)methanone (Compound 8)
A suspension of 7-methyl-4-(1-methyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine (0.4 g, 1.71 mmol) and 1-(4-fluorophenyl)-1H-1,2,4-triazole-3-carboxylic acid (355 mg, 1.71 mmol) in dichloromethane (10 mL) was treated with triethyl amine (0.45 mL, 3.43 mmol) and propanephosphonic acid anhydride (1.1 mL, 1.89 mmol) at 25 °C, and stirred for 18 h. After completion of the reaction, water (50 mL) was added to the reaction mixture and extracted with dichloromethane (3 x 30 mL). The combined organic extracts were washed with saturated brine solution (30 mL), dried over anhydrous sodium sulphate and evaporated under reduced pressure. The crude material thus obtained was purified by a reverse phase preparative-high performance liquid chromatography to isolate (1-(4-fluorophenyl)- 1H-1,2,4-triazol-3-yl)(7-methyl-4-(1-methyl-1H-pyrazol-4-yl)-4,7-dihydrothieno[2,3-c]pyridin- 6(5H)-yl)methanone 8 (213 mg, 0.50 mmol, 29% yield). 1H-NMR (400 MHz, DMSO-D6) δ 9.39-9.24 (m, 1H), 7.95-7.81 (m, 2H), 7.68-6.51 (m, 6H), 5.88-5.37 (m, 1H), 4.73-3.42 (m, 6H), 1.69-1.52 (m, 3H). LCMS (m/z): 423.2 (M+H)+ Example 17 : Synthesis of (2-(2,4-difluorophenyl)-2H-1,2,3-triazol-4-yl)(7-methyl-4-(1-methyl-1H- pyrazol-4-yl)-4,7-dihydrothieno[2,3-c]pyridin-6(5H)-yl)methanone (compound 9)
PI External A suspension of 7-methyl-4-(1-methyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine (0.4 g, 1.71 mmol) and 2-(2,4-difluorophenyl)-2H-1,2,3-triazole-4-carboxylic acid (386 mg, 1.71 mmol) in dichloromethane (7 mL) was treated with triethyl amine (0.5 mL, 3.43 mmol) and propanephosphonic acid anhydride (1.1 mL, 1.89 mmol) at 25 °C, and stirred for 18 h. After completion of the reaction, water (50 mL) was added to the reaction mixture, which then was extracted with dichloromethane (3 x 40 mL). The combined organic layers were washed with saturated brine solution (30 mL), dried over anhydrous sodium sulphate and evaporated under reduced pressure. The crude material thus obtained was purified by a reverse phase preparative-high performance liquid chromatography to obtain (2-(2,4- difluorophenyl)-2H-1,2,3-triazol-4-yl)(7-methyl-4-(1-methyl-1H-pyrazol-4-yl)-4,7-dihydrothieno[2,3- c]pyridin-6(5H)-yl)methanone 9 (160 mg, 0.36 mmol, 21% yield). 1H-NMR (400 MHz, DMSO-D6) δ 8.51 (d, J = 13.7 Hz, 1H), 8.04-7.94 (m, 1H), 7.71-7.57 (m, 2H), 7.43-7.32 (m, 3H), 6.56 (dd, J = 12.6, 5.0 Hz, 1H), 5.80-5.70 (m, 1H), 4.63-4.50 (m, 1H), 4.20-3.99 (m, 1H), 3.79 (d, J = 16.1 Hz, 3H), 3.45 (dd, J = 13.7, 11.5 Hz, 1H), 1.64 (dd, J = 48.8, 6.5 Hz, 3H). LCMS (m/z): 440.95 (M+H)+ Example 18 : Synthesis of (2-(2,4-difluorophenyl)-2H-tetrazol-5-yl)(7-methyl-4-(1-methyl-1H- pyrazol-4-yl)-4,7-dihydrothieno[2,3-c]pyridin-6(5H)-yl)methanone (compound 10)
A solution of 7-methyl-4-(1-methyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine (0.35 g, 1.500 mmol) and 2-(2,4-difluorophenyl)-2H-tetrazole-5-carboxylic acid (0.339 g, 1.500 mmol) in dichloromethane (7 mL) was treated sequentially with triethyl amine (0.4 mL, 3 mmol) and propanephosphonic acid anhydride (1 mL, 1.65 mmol) at 25 °C, and stirred for 18 h. After completion of the reaction, water (60 mL) was added and extracted with dichloromethane (3 x 30 mL). The combined organic extracts were washed with saturated brine solution (40 mL), dried over anhydrous sodium sulphate, filtered and evaporated under reduced pressure. The crude material thus obtained was purified by a reverse phase preparative-high performance liquid chromatography to isolate (2-(2,4- difluorophenyl)-2H-tetrazol-5-yl)(7-methl-4-(1-methyl-1H-pyrazol-4-yl)-4,7-dihydrothieno[2,3- c]pyridin-6(5H)-yl)methanone 10 (104 mg, 0.24 mmol, 16% yield). 1H-NMR (400 MHz, DMSO-D6) δ 8.15-6.78 (m, 6H), 6.56 (dd, J = 8.3, 5.1 Hz, 1H), 5.94-5.80 (m, 1H), 4.20-3.88 (m, 2H), 3.79 (d, J = 21.5 Hz, 3H), 3.33 (s, 1H), 1.71-1.57 (m, 3H). LCMS (m/z): 442 (M+H)+ Example 19 : Synthesis of (2-(3-fluorophenyl)-2H-tetrazol-5-yl)(4-(1-methyl-1H-pyrazol-4-yl)-4,7- dihydrothieno[2,3-c]pyridin-6(5H)-yl)methanone (Compound 81)
PI External
A suspension of 4-(1-methyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine (0.25 g, 1.14 mmol) and 2-(3-fluorophenyl)-2H-tetrazole-5-carboxylic acid (0.24 g, 1.14 mmol) in dichloromethane (8 mL) was treated with triethyl amine (0.32 mL, 2.28 mmol) followed by propanephosphonic acid anhydride (0.75 mL, 1.25 mmol) at 0 °C and stirred at 25 °C 16 h. Then, reaction mixture was diluted with water (30 mL) and extracted with dichloromethane (2 x 30 mL). The combined organic layer was washed with brine solution (30 mL); dried over sodium sulphate and evaporated under reduced pressure. The residue was purified by reverse phase preparative HPLC method to afford (2-(3-fluorophenyl)-2H- tetrazol-5-yl)(4-(1-methyl-1H-pyrazol-4-yl)-4,7-dihydrothieno[2,3-c]pyridin-6(5H)-yl)methanone (0.12 g, 0.27 mmol, 24% yield) as off white solid. Example 20: Synthesis of (4-(1,5-dimethyl-1H-pyrazol-4-yl)-4,7-dihydrothieno[2,3-c]pyridin-6(5H)- yl)(2-(3-fluorophenyl)-2H-tetrazol-5-yl)methanone (Compound 79)
A suspension of 4-(1,5-dimethyl-1H-pyrazol-4-yl)-4,5,6,7-tetrahydrothieno[2,3-c]pyridine (0.25 g, 1.07 mmol) 2-(3-fluorophenyl)-2H-tetrazole-5-carboxylic acid (0.22 g, 1.07 mmol) in dichloromethane (8 mL) was treated with trimethylamine (0.3 mL, 2.14 mmol) and propanephosphonic acid anhydride (0.71 mL, 1.18 mmol) at 0 °C and then stirred at 25 °C for 6 h. Upon completion of the reaction, the reaction mixture was taken into water (30 mL) and extracted with dichloromethane (2 x 30 mL). The combined organic layer was washed with brine solution (30 mL), dried over sodium sulphate and evaporated under reduced pressure. The residue was purified by preparative HPLC to isolate (4-(1,5- dimethyl-1H-pyrazol-4-yl)-4,7-dihydrothieno[2,3-c]pyridin-6(5H)-yl)(2-(3-fluorophenyl)-2H- tetrazol-5-yl)methanone (0.14 g, 0.33 mmol, 31% yield). Table 1: Representative compounds of the present disclosure were prepared according to the methods as described above by employing the suitable starting materials: Comp. No IUPAC Name Analytical Data (5-methyl-1-(2,4,6- 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.57 (s, trichlorophenyl)-1H-1,2,3- 2H), 7.26 (s, 2H), 7.22-7.17 (m, 2H), 7.15-7.11 (m, 1H), 11 triazol-4-yl)(4-(1-methyl-1H- 7.01 (s, 1H), 5.57-5.15 (m, 1H), 4.87 (d, J = 17.4 Hz, pyrazol-4-yl)-3,4- 1H), 4.59-4.41 (m, 1H), 4.34-4.27 (m, 1H), 3.96-3.92 dihydroisoquinolin-2(1H)- (m, 1H), 3.78 (s, 3H), 2.25 (s, 3H); LCMS (m/z): yl)methanone 500.95
PI External 1 (1-(2-chloro-4-fluorophenyl)-5- H-NMR (400 MHz, CHLOROFORM-D) δ 7.48-7.41 ethyl-1H-1,2,3-triazol-4-yl)(4- (m, 1H), 7.39-7.35 (m, 2H), 7.23-7.12 (m, 5H), 6.96 (s, 12 (1-methyl-1H-pyrazol-4-yl)-3,4- 1H), 5.46-5.38 (m, 1H), 5.02 (dd, J = 134.5, 17.4 Hz, dihydroisoquinolin-2(1H)- 1H), 4.37-4.28 (m, 2H), 4.22-4.09 (m, 1H), 3.77 (s, 3H), yl)methanone 2.67 (d, J = 45.0 Hz, 2H), 1.04-0.96 (m, 3H); LCMS (m/z): 465.05 (1-(3,5- 1H-NMR (400 MHz, DMSO-D6) δ 9.40 (s, 1H), 8.71 (d, bis(trifluoromethyl)phenyl)-1H- J = 8.3 Hz, 2H), 8.31 (s, 1H), 7.33-7.17 (m, 4H), 7.06 (t, 13 1,2,3-triazol-4-yl)(4-(1-methyl- J = 7.6 Hz, 2H), 5.26 (dd, J = 31.1, 16.9 Hz, 1H), 4.91 1H-pyrazol-4-yl)-3,4- (dd, J = 50.5, 17.2 Hz, 1H), 4.44-4.31 (m, 1H), 4.25 (d, J dihydroisoquinolin-2(1H)- = 4.9 Hz, 1H), 4.07-4.01 (m, 1H), 3.69 (s, 3H); LCMS yl)methanone (m/z): 521.45 1 (1-(2,4-dichlorophenyl)-5- H-NMR (400 MHz, DMSO-D6) δ 8.12 (t, J = 2.4 Hz, (trifluoromethyl)-1H-1,2,3- 1H), 8.06-8.02 (m, 1H), 7.82 (dd, J = 8.6, 2.4 Hz, 1H), triaz 7.33 (d, J = 7.3 Hz, 1H), 7.29-7.25 (m, 1H), 7.22-7.17 14 ol-4-yl)(4-(1-methyl-1H- pyrazol-4-yl)-3,4- (m, 2H), 7.13-7.11 (m, 1H), 7.06 (d, J = 7.8 Hz, 1H), dihydroisoquinolin-2(1H)- 5.20-4.73 (m, 2H), 4.25-4.20 (m, 1H), 4.07-4.01 (m, yl)methanone 1H), 3.91 (dd, J = 13.3, 4.0 Hz, 1H), 3.75 (s, 1H), 3.68 (s, 2H); LCMS (m/z):521 1H-NMR (400 MHz, DMSO-D6) δ 7.91 (dd, J = 10.0, (1-(4-chloro-2-fluorophenyl)-5- 2.2 Hz, 1H), 7.83-7.75 (m, 1H), 7.61 (d, J = 8.3 Hz, 1H), methyl-1H-1,2,3-triazol-4-yl)(4- 7.48-7.30 (m, 1H), 7.25 (t, J = 7.7 Hz, 1H), 7.17 (d, J = 15 (1-methyl-1H-pyrazol-4-yl)-3,4- 11.5 Hz, 2H), 7.05 (t, J = 7.6 Hz, 1H), 6.98 (s, 1H), dihydroisoquinolin-2(1H)- 5.22-5.09 (m, 1H), 4.73 (d, J = 17.1 Hz, 1H), 4.41-4.15 yl)methanone (m, 2H), 4.06-3.95 (m, 1H), 3.66 (s, 3H), 2.10 (s, 3H) LCMS (m/z): 451 (1-(2-chloro-4-methylphenyl)-5- 1H-NMR (400 MHz, DMSO-D6) δ 7.82 (dd, J = 14.3, (trifluoromethyl)-1H-1,2,3- 8.2 Hz, 1H), 7.68 (d, J = 1.0 Hz, 1H), 7.40 (dd, J = 55.4, 16 triazol-4-yl)(4-(1-methyl-1H- 7.7 Hz, 2H), 7.29-7.25 (m, 1H), 7.22-7.02 (m, 4H), 5.13- pyrazol-4-yl)-3,4- 4.72 (m, 2H), 4.26-4.20 (m, 1H), 4.06-4.02 (m, 1H), dihydroisoquinolin-2(1H)- 3.90 (dd, J = 13.2, 4.2 Hz, 1H), 3.75 (s, 1H), 3.66 (s, yl)methanone 2H), 2.45 (s, 3H); LCMS (m/z): 501.15 1 3-fluoro-4-(4-(4-(1-methyl-1H- H-NMR (400 MHz, DMSO-D6) δ 8.40 (d, J = 9.8 Hz, pyrazol-4-yl)-1,2,3,4- 1H), 8.20-8.15 (m, 1H), 8.10 (d, J = 8.3 Hz, 1H), 7.38 tetrahydroisoq (dd, J = 40.8, 7.6 Hz, 1H), 7.27 (t, J = 5.7 Hz, 1H), 7.22- 17 uinoline-2- carbonyl)-5-(trifluoromethyl)- 7.17 (m, 2H), 7.13-7.04 (m, 2H), 5.03 (d, J = 17.6 Hz, 1H-1,2,3-triazol-1- 1H), 4.86-4.78 (m, 1H), 4.26-4.21 (m, 1H), 4.18-4.03 yl)benzonitrile (m, 1H), 3.94 (dt, J = 12.9, 4.1 Hz, 1H), 3.66 (s, 3H) LCMS (m/z): 496.1 (1-(2,4-dichloro-6- 1H-NMR (400 MHz, DMSO-D6) δ 7.88-7.86 (m, 1H), methylphenyl)-5-methyl-1H- 7.70 (d, J = 1.8 Hz, 1H), 7.51-7.15 (m, 4H), 7.07-7.00 18 1,2,3-triazol-4-yl)(4-(1-methyl- (m, 2H), 5.28-5.17 (m, 1H), 4.68 (dd, J = 24.9, 17.6 Hz, 1H-pyrazol-4-yl)-3,4- 1H), 4.42 (ddd, J = 51.7, 12.8, 4.3 Hz, 1H), 4.22-4.01 dihydroisoquinolin-2(1H)- (m, 2H), 3.64 (s, 3H), 2.20 (s, 1H), 2.04-1.96 (m, 5H) yl)methanone LCMS (m/z): 481.05 (1-(3-chloro-5- 1 (trifluoromethyl)phenyl)-1H- H-NMR (400 MHz, DMSO-D6) δ 9.40 (d, J = 84.1 Hz, 1,2,3-triazol- 1H), 8.48-8.36 (m, 2H), 8.07 (s, 1H), 7.31-7.05 (m, 6H), 19 4-yl)(4-(1-methyl- 1H-pyrazol-4-yl)-3,4- 5.25 (d, J = 13.9 Hz, 1H), 4.98-4.82 (m, 1H), 4.37-4.01 dihydroisoquinolin-2(1H)- (m, 3H), 3.73 (d, J = 27.9 Hz, 3H) yl)methanone LCMS (m/z): 487 (1-(2-chloro-4-fluorophenyl)-5- 1H-NMR (400 MHz, DMSO-D6) δ 7.95-7.87 (m, 2H), 20 isopropyl-1H-1,2,3-triazol-4- 7.57-7.52 (m, 1H), 7.43-7.01 (m, 6H), 5.12-4.72 (m, yl)(4-(1-methyl-1H-pyrazol-4- 2H), 4.19 (dd, J = 19.6, 5.6 Hz, 2H), 4.05-3.88 (m, 1H),
PI External yl)-3,4-dihydroisoquinolin- 3.73 (s, 3H), 2.81-2.63 (m, 1H), 1.12-0.91 (m, 6H); 2(1H)-yl)methanone LCMS (m/z): 479.1 (4-(1,5-dimethyl-1H-pyrazol-4- 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.57 (td, J = yl)-3,4-dihydroisoquinolin- 4.0, 2.1 Hz, 2H), 7.25-7.19 (m, 2H), 7.18-7.02 (m, 3H), 21 2(1H)-yl)(5-methyl-1-(2,4,6- 5.76-4.84 (m, 2H), 4.76-4.51 (m, 1H), 4.46-4.21 (m, trichlorophenyl)-1H-1,2,3- 1H), 3.92 (dd, J = 13.1, 8.9 Hz, 1H), 3.76 (s, 3H), 2.34 triazol-4-yl)methanone (s, 3H), 2.24 (s, 3H); LCMS (m/z): 516.75 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.47-8.33 (1-(4-chloro-2-methylphenyl)- (m, 1H), 7.52-7.44 (m, 1H), 7.41 (s, 1H), 7.25-7.07 (m, 1H-1,2,3-triazol-4-yl)(4-(1,5- 5H), 7.04-7.00 (m, 1H), 5.62 (dd, J = 213.7, 17.1 Hz, 22 dimethyl-1H-pyrazol-4-yl)-3,4- 1H), 5.18 (d, J = 17.1 Hz, 1H), 4.68 (ddd, J = 129.2, dihydroisoquinolin-2(1H)- 12.9, 4.8 Hz, 1H), 4.40-4.20 (m, 1H), 4.10 (dd, J = 13.3, yl)methanone 8.7 Hz, 1H), 3.74 (s, 3H), 2.44 (s, 3H), 2.27 (s, 3H) LCMS (m/z): 447.2 1 (1-(2-chloro-4-fluorophenyl)-5- H-NMR (400 MHz, CHLOROFORM-D) δ 7.45-7.36 ethyl-1H-1,2,3-triazol-4-yl)(4- (m, 2H), 7.25-7.07 (m, 5H), 7.03 (d, J = 6.4 Hz, 1H), 23 (1,5-dimethyl-1H-pyrazol-4-yl)- 5.64-5.21 (m, 1H), 4.85 (d, J = 17.1 Hz, 1H), 4.66 (dd, J 3,4-dihydroisoquinolin-2(1H)- = 12.8, 3.7 Hz, 1H), 4.47-4.20 (m, 1H), 3.96-3.90 (m, yl)methanone 1H), 3.75 (s, 3H), 2.75 (s, 2H), 2.25 (d, J= 6.1 Hz, 3H), 1.07-0.97 (m, 3H); LCMS (m/z): 479.05 (1-(2-chloro-4-methylphenyl)-5- 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.42 (s, methyl-1H-1,2,3-triazol-4-yl)(4- 1H), 7.29 (d, 1H), 7.25-7.07 (m, 5H), 7.02 (d, 1H), 24 (1,5-dimethyl-1H-pyrazol-4-yl)- 5.71-4.86 (m, 2H), 4.60 (m, 1H), 4.33 (m, 1H), 3.95 (m, 3,4-dihydroisoquinolin-2(1H)- 1H), 3.78 (s, 3H), 2.44 (m, 3H), 2.33 (m, 3H), 2.25 (m, yl)methanone 3H); LCMS (m/z):461.2 (1-(3,5- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.59 (d, J = bis(trifluoromethyl)phenyl)-1H- 46.5 Hz, 1H), 8.27 (d, J = 5.2 Hz, 2H), 8.00 (s, 1H), 25 1,2,3-triazol-4-yl)(4-(1,5- 7.25-7.16 (m, 3H), 7.09-7.00 (m, 2H), 5.56 (dd, J = dimethyl-1H-pyrazol-4-yl)-3,4- 153.3, 17.0 Hz, 1H), 5.05 (dd, J = 53.5, 17.1 Hz, 1H), dihydroisoquinolin-2(1H)- 4.74-4.41 (m, 1H), 4.37-4.18 (m, 2H), 3.71 (s, 3H), 2.26 yl)methanone (s, 3H); LCMS (m/z): 535.2 (1-(2,4-dichlorophenyl)-5- 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.66 (dd, J (trifluoromethyl)-1H-1,2,3- = 5.8, 2.1 Hz, 1H), 7.52-7.47 (m, 1H), 7.42 (t, J = 8.6 26 triazol-4-yl)(4-(1,5-dimethyl- Hz, 1H), 7.28 (d, J = 7.9 Hz, 1H), 7.25-7.16 (m, 2H), 1H-pyrazol-4-yl)-3,4- 7.11-7.04 (m, 2H), 5.24 (d, J = 17.1 Hz, 1H), 4.92 (d, J = dihydroisoquinolin-2(1H)- 17.4 Hz, 1H), 4.50-4.19 (m, 2H), 4.00 (dd, J = 13.1, 4.6 yl)methanone Hz, 1H), 3.75 (s, 3H), 2.08 (s, 3H); LCMS (m/z): 535.1 (1-(2,4-dichloro-6- 1 methylphenyl)-5-methyl-1H- H-NMR (400 MHz, CHLOROFORM-D) δ 7.44 (t, J = 1,2,3-triazol- 2.6 Hz, 1H), 7.32 (d, J = 1.8 Hz, 1H), 7.22 (dd, J = 11.6, 27 4-yl)(4-(1,5- dimethyl-1H-pyrazol-4-yl)-3,4- 4.0 Hz, 2H), 7.16-7.01 (m, 3H), 5.81-4.83 (m, 2H), 4.81- dihydroisoquinolin-2(1H)- 4.20 (m, 2H), 4.00-3.48 (m, 4H), 2.32-2.23 (m, 6H), yl)methanone 2.02-1.96 (m, 3H); LCMS (m/z): 497.05 1 (1-(4-chloro-2-fluorophenyl)-5- H-NMR (400 MHz, CHLOROFORM-D) δ 7.47-7.35 methyl-1H-1,2,3-triazol-4-yl)(4- (m, 3H), 7.24-6.98 (m, 5H), 5.62-5.24 (m, 1H), 5.04 (dd, 28 (1,5-dimethyl-1H-pyrazol-4-yl)- J = 65.1, 17.1 Hz, 1H), 4.51 (ddd, J = 43.0, 13.1, 4.7 Hz, 3,4-dihydroisoquinolin-2(1H)- 1H), 4.39-4.20 (m, 1H), 4.04 (dd, J = 13.3, 8.1 Hz, 1H), yl)methanone 3.73 (s, 3H), 2.37-2.36 (d, J=1.6Hz, 3H), 2.22 (s, 3H) LCMS (m/z): 465.2 (1-(2-chloro-4-methylphenyl)-5- 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.45-7.43 29 (trifluoromethyl)-1H-1,2,3- (m, 1H), 7.35 (dd, J = 11.0, 8.3 Hz, 1H), 7.28 (t, J = 0.9 triazol-4-yl)(4-(1,5-dimethyl- Hz, 1H), 7.25-7.12 (m, 3H), 7.09-7.00 (m, 2H), 5.26 (d, 1H-pyrazol-4-yl)-3,4- J = 17.1 Hz, 1H), 4.90 (d, J = 18.3 Hz, 1H), 4.51-4.19
PI External dihydroisoquinolin-2(1H)- (m, 1H), 4.00 (dd, J = 13.1, 4.6 Hz, 1H), 3.82-3.61 (m, yl)methanone 4H), 2.47 (s, 3H), 2.09 (s, 3H); LCMS (m/z): 515.25 4-(4-(4-(1,5-dimethyl-1H- 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.74-7.65 pyrazol-4-yl)-1,2,3,4- (m, 3H), 7.29 (d, J = 8.3 Hz, 1H), 7.24-7.16 (m, 2H), 30 tetrahydroisoquinoline-2- 7.11-7.04 (m, 2H), 5.21 (d, J = 17.4 Hz, 1H), 4.94 (d, J = carbonyl)-5-(trifluoromethyl)- 20.2 Hz, 1H), 4.45-4.19 (m, 1H), 4.02 (dd, J = 13.1, 4.6 1H-1,2,3-triazol-1-yl)-3- Hz, 1H), 3.81-3.69 (m, 4H), 2.08 (s, 3H); LCMS (m/z): fluorobenzonitrile 510.25 1 (1-(3-chloro-5- H-NMR (400 MHz, CHLOROFORM-D) δ 8.53 (d, J = (trifluoromethyl)phenyl)-1H- 48.3 Hz, 1H), 8.02 (d, J = 5.8 Hz, 1H), 7.96 (s, 1H), 7.73 1,2, (s, 1H), 7.25-7.15 (m, 3H), 7.10-6.98 (m, 2H), 5.56 (dd, 31 3-triazol-4-yl)(4-(1,5- dimethyl-1H-pyrazol-4-yl)-3,4- J = 161.1, 16.8 Hz, 1H), 5.04 (dd, J = 48.6, 17.1 Hz, dihydroisoquinolin-2(1H)- 1H), 4.58 (ddd, J = 106.0, 13.1, 4.7 Hz, 1H), 4.36-4.17 yl)methanone (m, 2H), 3.70 (s, 3H), 2.25 (s, 3H); LCMS (m/z): 501.15 1 (1-(3,5-dichlorophenyl)-1H- H-NMR (400 MHz, CHLOROFORM-D) δ 8.44 (d, J = 1,2,3-triazol-4-yl)(4-(1,5- 54.7 Hz, 1H), 7.72-7.70 (m, 2H), 7.47 (t, J = 1.7 Hz, 32 dimethyl-1H-pyrazol-4-yl)-3,4- 1H), 7.24-7.15 (m, 3H), 7.11-6.97 (m, 2H), 5.56 (dd, J = dihydroisoquinolin-2(1H)- 169.5, 17.0 Hz, 1H), 5.04 (dd, J = 39.3, 17.3 Hz, 1H), yl)methanone 4.57 (ddd, J = 96.1, 13.1, 4.5 Hz, 1H), 4.34-4.18 (m, 2H), 3.69 (s, 3H), 2.24 (s, 3H); LCMS (m/z): 468.85 1 (1-(2,4-difluorophenyl)-1H- H-NMR (400 MHz, DMSO-D6) δ 8.90 (d, J = 107.1 1,2,3-triazol-4-yl)(4-(1-methyl- Hz, 1H), 7.98-7.89 (m, 1H), 7.74-7.68 (m, 1H), 7.48- 33 1H-pyrazol-4-yl)-3,4- 7.10 (m, 6H), 7.07-7.03 (m, 1H), 5.29 (dd, J = 30.3, 17.1 dihydroisoquinolin-2(1H)- Hz, 1H), 4.90 (dd, J = 53.7, 17.5 Hz, 1H), 4.42-4.30 (m, yl)methanone 1H), 4.27-4.21 (m, 1H), 4.04-4.00 (m, 1H), 3.70 (s, 3H); LCMS (m/z): 420.95 (1-(2,4-difluorophenyl)-5- 1H-NMR (400 MHz, DMSO-D6) δ 7.87-7.70 (m, 2H), methyl-1H-1,2,3-triazol-4-yl)(4- 7.43-7.14 (m, 4H), 7.00-6.91 (m, 1H), 6.69 (s, 1H), 5.26- 34 (1,5-dimethyl-1H-pyrazol-4-yl)- 5.03 (m, 1H), 4.85 (d, J = 17.1 Hz, 1H), 4.25-4.08 (m, 3,4-dihydroisoquinolin-2(1H)- 3H), 3.60 (s, 3H), 2.28-2.05 (m, 6H) yl)methanone LCMS (m/z): 449.3 (4-(1-methyl-1H-pyrazol-4-yl)- 1H-NMR (400 MHz, DMSO-D6) δ 9.16 (m, 1H), 7.96 35 3,4-dihydroisoquinolin-2(1H)- (dd, 2H), 7.61 (t, 2H), 7.54-7.49 (m, 1H), 7.33-7.03 (m, yl)(1-phenyl-1H-1,2,3-triazol-4- 6H), 5.31-5.23 (m, 1H), 4.90 (m, 1H), 4.44-4.00 (m, yl)methanone 3H), 3.76-3.66 (m, 3H); LCMS (m/z): 385.15 1H-NMR (400 MHz, DMSO-D6) δ 9.04 (s, 1H), 7.96 (4-(1,5-dimethyl-1H-pyrazol-4- (dd, J = 13.1, 7.9 Hz, 2H), 7.61 (t, J = 7.8 Hz, 2H), 7.52 yl)-3,4-dihydroisoq (t, J = 7.3 Hz, 1H), 7.32 (d, J = 7.6 Hz, 1H), 7.25-7.14 36 uinolin- 2(1H)-yl)(1-phenyl-1H-1,2,3- (m, 2H), 6.93 (q, J = 7.3 Hz, 1H), 6.76 (s, 1H), 5.33 (d, J triazol-4-yl)methanone = 20.8 Hz, 1H), 4.94 (dd, J = 27.4, 17.1 Hz, 1H), 4.38 (t, J = 9.4 Hz, 1H), 4.25-4.11 (m, 2H), 3.56 (s, 3H), 2.07 (s, 3H); LCMS (m/z): 399.15 1H-NMR (400 MHz, DMSO-D6) δ 8.90 (d, J = 112.2 (4-(1,5-dimethyl-1H-pyrazol-4- Hz, 1H), 7.90-7.82 (m, 1H), 7.67-7.44 (m, 3H), 7.33- 37 yl)-3,4-dihydroisoquinolin- 7.14 (m, 3H), 7.02-6.78 (m, 2H), 5.33 (dd, J = 37.3, 16.8 2(1H)-yl)(1-(2-fluorophenyl)- Hz, 1H), 4.94-4.89 (m, 1H), 4.37 (dd, J = 12.7, 3.8 Hz, 1H-1,2,3-triazol-4-yl)methanone 1H), 4.26-4.13 (m, 2H), 3.59 (s, 3H), 2.09 (s, 3H); LCMS (m/z): 417.05 (1-(4-fluorophenyl)-1H-1,2,4- 1 triazol-3-yl)(4-(1-methyl-1H- H-NMR (400 MHz, DMSO-D6) δ 9.38-9.31 (m, 1H), 38 pyrazol-4-yl)-3,4- 7.95-7.86 (m, 2H), 7.51-7.02 (m, 8H), 4.98-4.84 (m, dihydroisoquinolin-2(1H)- 2H), 4.22 (q, J = 6.0 Hz, 1H), 4.08-3.98 (m, 2H), 3.69 (s, yl)methanone 3H); LCMS (m/z): 403.05
PI External 1 (4-(1,5-dimethyl-1H-pyrazol-4- H-NMR (400 MHz, DMSO-D6) δ 8.68 (d, J = 162.6 yl)-3 Hz, 1H), 7.77 (d, J = 82.2 Hz, 3H), 7.41-7.35 (m, 2H), 39 ,4-dihydroisoquinolin- 2(1H)-yl)(1-(4-fluorophenyl)- 7.28-7.19 (m, 2H), 7.16-6.92 (m, 3H), 5.01-4.74 (m, 1H-pyrazol-4-yl)methanone 2H), 4.19-3.65 (m, 6H), 2.06 (s, 3H) LCMS (m/z): 416 (1-(2,4-difluorophenyl)-5- 1H-NMR (400 MHz, DMSO-D6) δ 7.78-7.59 (m, 2H), methyl-1H-pyrazol-3-yl)(4-(1- 7.48-7.14 (m, 5H), 7.09-7.02 (m, 2H), 6.48 (d, J = 87.8 40 methyl-1H-pyrazol-4-yl)-3,4- Hz, 1H), 5.13 (s, 1H), 4.83 (dd, J = 43.6, 17.5 Hz, 1H), dihydroisoquinolin-2(1H)- 4.29-4.11 (m, 2H), 3.98-3.88 (m, 1H), 3.69 (s, 3H), 2.15 yl)methanone (s, 3H); LCMS (m/z): 434 (1-(4-fluorophenyl)-1H-pyrazol- 1H-NMR (400 MHz, DMSO-D6) δ 8.57-8.51 (m, 1H), 41 3-yl)(4-(1-methyl-1H-pyrazol-4- 7.95-7.85 (m, 2H), 7.50-6.62 (m, 9H), 5.21 (s, 1H), 4.87 yl)-3,4-dihydroisoquinolin- (dd, J = 32.1, 17.4 Hz, 1H), 4.26-4.17 (m, 2H), 4.05-3.90 2(1H)-yl)methanone (m, 1H), 3.70 (s, 3H); LCMS (m/z): 402.35 (1-(2,4-difluorophenyl)-5- 1H-NMR (400 MHz, DMSO-D6) δ 7.78-7.59 (m, 2H), methyl-1H-pyrazol-3-yl)(4-(1,5- 7.34-6.67 (m, 6H), 6.45 (d, J = 107.3 Hz, 1H), 5.16 (d, J 42 dimethyl-1H-pyrazol-4-yl)-3,4- = 46.9 Hz, 1H), 4.87 (dd, J = 24.9, 17.1 Hz, 1H), 4.23- dihydroisoquinolin-2(1H)- 4.11 (m, 2H), 3.91 (dd, J = 13.0, 7.3 Hz, 1H), 3.61 (s, yl)methanone 3H), 2.15 (s, 3H), 1.91 (s, 3H); LCMS (m/z): 448 1 (4-(1,5-dimethyl-1H-pyrazol-4- H-NMR (400 MHz, DMSO-D6) δ 8.57-8.50 (m, 1H), yl)-3,4-dihydroisoq 7.95-7.83 (m, 2H), 7.41-6.77 (m, 7H), 6.56 (d, J = 2.4 43 uinolin- 2(1H)-yl)(1-(4-fluorophenyl)- Hz, 1H), 5.75-5.30 (m, 1H), 5.20-4.91 (m, 1H), 4.24- 1H-pyrazol-3-yl)methanone 4.14 (m, 2H), 4.03 (dd, J = 14.2, 8.7 Hz, 1H), 3.62 (s, 3H), 1.98 (s, 3H); LCMS (m/z): 416.3 (2-(2-fluorophenyl)-2H-1,2,3- 1H-NMR (400 MHz, DMSO-D6) δ 8.32 (d, J = 124.2 triazol-4-yl)(4-(1-methyl-1H- Hz, 1H), 7.96-7.84 (m, 1H), 7.61-7.54 (m, 2H), 7.49- 44 pyrazol-4-yl)-3,4- 7.31 (m, 2H), 7.27-7.22 (m, 1H), 7.20-7.03 (m, 4H), dihydroisoquinolin-2(1H)- 5.19-4.75 (m, 2H), 4.31-4.00 (m, 3H), 3.72 (d, J = 33.3 yl)methanone Hz, 3H); LCMS (m/z): 403.2 (1-(2,4-difluorophenyl)-1H- 1 pyrazol-4-yl)(4-(1-methyl-1H- H-NMR (400 MHz, DMSO-D6) δ 8.13-7.80 (m, 3H), 45 pyrazol-4-yl)-3,4- 7.63-7.57 (m, 1H), 7.47-7.15 (m, 6H), 7.04 (d, J = 7.6 dihydroisoquinolin-2(1H)- Hz, 1H), 4.97-4.72 (m, 2H), 4.23 (s, 1H), 3.97-3.88 (m, yl)methanone 2H), 3.74 (s, 3H); LCMS (m/z): 420.25 (1-(2,4-difluorophenyl)-1H- 1 pyrazol-4-yl)(7-methyl-4-(1- H-NMR (400 MHz, DMSO-D6) δ 8.56-7.04 (m, 8H), 46 methyl-1H-pyrazol-4-yl)-4,7- 6.66 (dd, J = 80.8, 5.0 Hz, 1H), 5.75 (s, 1H), 4.24-3.44 dihydrothieno[2,3-c]pyridin- (m, 6H), 1.70-1.49 (m, 3H) 6(5H)-yl)methanone LCMS (m/z): 440.25 (1-(2-fluorophenyl)-1H-1,2,3- triazol-4-yl)(7-methyl-4-(1- 1H-NMR (400 MHz, DMSO-D6) δ 9.08-8.35 (m, 1H), 47 methyl-1H-pyrazol-4-yl)-4,7- 7.90-6.58 (m, 8H), 6.02-5.74 (m, 1H), 4.98-3.48 (m, dihydrothieno[2,3-c]pyridin- 6H), 1.77-1.52 (m, 3H); LCMS (m/z): 423.15 6(5H)-yl)methanone 1 (4-(1,5-dimethyl-1H-pyrazol-4- H-NMR (400 MHz, DMSO-D6) δ 8.29 (d, J = 144.3 yl)-3,4-dihydroisoquinoli Hz, 1H), 7.97-7.81 (m, 1H), 7.64-7.54 (m, 2H), 7.47- 48 n- 2(1H)-yl)(2-(2-fluorophenyl)- 7.42 (m, 1H), 7.33-7.12 (m, 3H), 7.01-6.67 (m, 2H), 2H-1,2,3-triazol-4-yl)methanone 5.22-4.83 (m, 2H), 4.22-3.58 (m, 6H), 2.20-1.94 (m, 3H) LCMS (m/z): 417.4 1 (4-(1,5-dimethyl-1H-pyrazol-4- H-NMR (400 MHz, DMSO-D6) δ 8.59-7.78 (m, 3H), yl)-3,4-dihydroiso 7.53-7.45 (m, 2H), 7.41-7.36 (m, 1H), 7.28-7.13 (m, 49 quinolin- 2(1H)-yl)(1-(2-fluorophenyl)- 3H), 6.94-6.83 (m, 2H), 5.03-4.74 (m, 2H), 4.19-3.65 1H-pyrazol-4-yl)methanone (m, 6H), 2.07 (d, J = 7.6 Hz, 3H) LCMS (m/z): 416.05
PI External (2-(2,4-difluorophenyl)-2H- 1H-NMR (400 MHz, DMSO-D6) δ 8.32 (d, J = 124.4 1,2,3-triazol-4-yl)(4-(1-methyl- Hz, 1H), 7.97 (dtd, J = 35.7, 8.8, 6.1 Hz, 1H), 7.70-7.64 50 1H-pyrazol-4-yl)-3,4- (m, 1H), 7.48-7.02 (m, 7H), 5.17-4.75 (m, 2H), 4.30- dihydroisoquinolin-2(1H)- 3.99 (m, 3H), 3.76-3.66 (m, 3H) yl)methanone LCMS (m/z): 421.3 (2-(2,4-difluorophenyl)-2H- 1H-NMR (400 MHz, DMSO-D6) δ 8.15-8.05 (m, 1H), tetrazol-5-yl)(4-(1-methyl-1H- 7.83-7.77 (m, 1H), 7.49-7.33 (m, 2H), 7.28-7.25 (m, 51 pyrazol-4-yl)-3,4- 1H), 7.21-7.02 (m, 4H), 5.10-4.82 (m, 2H), 4.30-4.21 dihydroisoquinolin-2(1H)- (m, 1H), 4.13-3.98 (m, 2H), 3.77-3.67 (m, 3H) yl)methanone LCMS (m/z): 422 (2-(4-fluorophenyl)-2H-1,2,3- 1 triazol-4-yl)(4-(1-methyl-1H- H-NMR (400 MHz, DMSO-D6) δ 8.43-8.01 (m, 3H), 52 pyrazol-4-yl)-3,4- 7.50-7.31 (m, 3H), 7.27-7.04 (m, 5H), 5.21-4.74 (m, dihydroisoquinolin-2(1H)- 2H), 4.33-4.00 (m, 3H), 3.77-3.69 (m, 3H) yl)methanone LCMS (m/z): 403.5 (4-(1,5-dimethyl-1H-pyrazol-4- 1H-NMR (400 MHz, DMSO-D6) δ 8.42-7.99 (m, 3H), 53 yl)-3,4-dihydroisoquinolin- 7.48-7.42 (m, 2H), 7.33-6.73 (m, 5H), 5.20-4.83 (m, 2(1H)-yl)(2-(4-fluorophenyl)- 2H), 4.24-3.59 (m, 6H), 2.09 (d, J = 91.1 Hz, 3H) 2H-1,2,3-triazol-4-yl)methanone LCMS (m/z): 417 1 (1-(2,4-difluorophenyl)-1H- H-NMR (400 MHz, DMSO-D6) δ 8.55 (dd, J = 6.6, 2.0 pyrazol-4-yl)(1-methyl-4-(1- Hz, 1H), 8.35 (dt, J = 22.9, 5.7 Hz, 1H), 8.09-8.07 (m, 54 methyl-1H-pyrazol-4-yl)-3,4- 1H), 7.85-7.77 (m, 1H), 7.69-7.51 (m, 2H), 7.46-6.90 dihydroisoquinolin-2(1H)- (m, 5H), 4.73-4.36 (m, 1H), 4.20 (t, J = 7.8 Hz, 1H), yl)methanone 3.83-3.78 (m, 2H), 3.76-3.63 (m, 3H), 1.50 (d, J = 6.7 Hz, 3H); LCMS (m/z): 434.3 (2-(4-fluorophenyl)-2H-1,2,3- 1H-NMR (400 MHz, DMSO-D6) δ 8.47-8.39 (m, 1H), triazol-4-yl)(7-methyl-4-(1- 8.12-7.97 (m, 2H), 7.80-7.62 (m, 1H), 7.49-6.53 (m, 55 methyl-1H-pyrazol-4-yl)-4,7- 5H), 5.91-5.70 (m, 1H), 4.64-4.46 (m, 1H), 4.38-3.99 dihydrothieno[2,3-c]pyridin- (m, 1H), 3.82-3.61 (m, 3H), 3.49-3.43 (m, 1H), 1.77- 6(5H)-yl)methanone 1.53 (m, 3H); LCMS (m/z): 422.95 1 (1-(2,4-difluorophenyl)-1H- H-NMR (400 MHz, DMSO-D6) δ 9.00-8.40 (m, 1H), 1,2,3-triazol-4-yl)((1S,4R)-1- 7.97-7.86 (m, 1H), 7.73-7.66 (m, 1H), 7.53-7.18 (m, 56 methyl-4-(1-methyl-1H-pyrazol- 5H), 7.16-7.05 (m, 1H), 6.88 (d, J = 36.7 Hz, 1H), 6.17- 4-yl)-3,4-dihydroisoquinolin- 5.74 (m, 1H), 4.91-4.68 (m, 1H), 4.18 (d, J = 28.1 Hz, 2(1H)-yl)methanone 1H), 3.93-3.80 (m, 1H), 3.63 (d, J = 32.1 Hz, 3H), 1.56 (dd, J = 17.1, 6.7 Hz, 3H); LCMS (m/z): 434.95 1 (1-(2,4-difluorophenyl)-1H- H-NMR (400 MHz, DMSO-D6) δ 9.06-8.65 (m, 1H), 1,2,3-triazol-4-yl)((1S,4S)-1- 7.98-7.87 (m, 1H), 7.74-7.57 (m, 1H), 7.41-7.27 (m, 57 methyl-4-(1-methyl-1H-pyrazol- 5H), 7.25-7.09 (m, 1H), 6.92 (t, J = 8.3 Hz, 1H), 5.93- 4-yl)-3,4-dihydroisoquinolin- 5.66 (m, 1H), 4.87-4.56 (m, 1H), 4.34-4.09 (m, 2H), 2(1H)-yl)methanone 3.78-3.72 (m, 3H), 1.69 (dd, J = 67.6, 6.7 Hz, 3H); LCMS (m/z): 435 1 (4-(1,5-dimethyl-1H-pyrazol-4- H-NMR (400 MHz, DMSO-D6) δ 8.00 (dtd, J = 31.0, yl)-3,4-d 7.7, 1.6 Hz, 1H), 7.79-7.73 (m, 1H), 7.70-7.65 (m, 1H), 58 ihydroisoquinolin- 2(1H)-yl)(2-(2-fluorophenyl)- 7.56-7.51 (m, 1H), 7.35-7.13 (m, 3H), 7.02-6.77 (m, 2H-tetrazol-5-yl)methanone 2H), 5.06-4.91 (m, 2H), 4.25-3.83 (m, 3H), 3.74-3.57 (m, 3H), 2.22-1.97 (m, 3H); LCMS (m/z): 418.05 1 (2-(2-fluorophenyl)-2H-tetrazol- H-NMR (400 MHz, DMSO-D6) δ 8.01 (dtd, J = 19.6, 5-yl)(4-(1-methyl-1H-pyra 7.7, 1.6 Hz, 1H), 7.78-7.73 (m, 1H), 7.70-7.64 (m, 1H), 59 zol-4- yl)-3,4-dihydroisoquinolin- 7.56-7.02 (m, 7H), 5.11-4.82 (m, 2H), 4.30-4.22 (m, 2(1H)-yl)methanone 1H), 4.14-3.95 (m, 2H), 3.77-3.60 (m, 3H) LCMS (m/z): 403.95
PI External 1 (2-(2-fluorophenyl)-2H-tetrazol- H-NMR1H-NMR (400 MHz, DMSO-D6) δ 8.07-8.01 5-yl)(7-methyl-4-(1-methyl-1H- (m, 1H), 7.78-7.73 (m, 1H), 7.70-7.62 (m, 1H), 7.56- 60 pyrazol-4-yl)-4,7- 7.51 (m, 1H), 7.47-7.32 (m, 3H), 6.92-6.55 (m, 1H), dihydrothieno[2,3-c]pyridin- 5.95-5.36 (m, 1H), 4.66-4.06 (m, 2H), 3.92-3.49 (m, 6(5H)-yl)methanone 4H), 1.67 (dd, J = 31.5, 6.6 Hz, 3H); LCMS (m/z): 423.95 1 (1-(2,4-difluorophenyl)-1H- H-NMR (400 MHz, DMSO-D6) δ 9.09-8.30 (m, 1H), 1,2,3-triazol-4-yl)(1,4-dimethyl- 7.97-7.67 (m, 2H), 7.43-7.34 (m, 2H), 7.32-7.03 (m, 61 4-(1-methyl-1H-pyrazol-4-yl)- 3H), 6.94-6.66 (m, 1H), 5.82-5.72 (m, 1H), 4.91 (dd, J = 3,4-dihydroisoquinolin-2(1H)- 48.4, 13.7 Hz, 1H), 4.56 (dd, J = 74.7, 13.3 Hz, 1H), yl)methanone 3.85-3.44 (m, 3H), 1.74-1.46 (m, 6H); LCMS (m/z): 449.05 (1-(2,4-difluorophenyl)-1H- 1H-NMR (400 MHz, DMSO-D6) δ 7.86-7.32 (m, 6H), pyrazol-3-yl)(7-methyl-4-(1- 7.27-6.55 (m, 3H), 5.57 (dd, J = 24.3, 6.6 Hz, 1H), 4.13- 62 methyl-1H-pyrazol-4-yl)-4,7- 3.98 (m, 1H), 3.80 (d, J = 19.0 Hz, 4H), 3.67-3.41 (m, dihydrothieno[2,3-c]pyridin- 1H), 1.38 (dd, J = 53.8, 6.4 Hz, 3H); 6(5H)-yl)methanone LCMS (m/z): 440.05 (2-(2-chlorophenyl)-2H-tetrazol- 1H-NMR (400 MHz, DMSO-D6) δ 8.00-7.86 (m, 2H), 5-yl)(7-methyl-4-(1-methyl-1H- 7.79-7.75 (m, 1H), 7.72-7.61 (m, 2H), 7.46-7.33 (m, 63 pyrazol-4-yl)-4,7- 2H), 6.79-6.55 (m, 1H), 5.96-5.34 (m, 1H), 4.80-4.06 dihydrothieno[2,3-c]pyridin- (m, 2H), 3.94-3.52 (m, 4H), 1.71-1.57 (m, 3H) 6(5H)-yl)methanone LCMS (m/z): 440.05 (7-methyl-4-(1-methyl-1H- 1H-NMR (400 MHz, DMSO-D6) δ 8.01-7.90 (m, 2H), pyrazol-4-yl)-4,7- 7.70-7.33 (m, 5H), 6.80-6.54 (m, 1H), 5.94-5.34 (m, 64 dihydrothieno[2,3-c]pyridin- 1H), 4.66-4.04 (m, 2H), 3.90-3.47 (m, 4H), 2.42 (s, 3H), 6(5H)-yl)(2-(p-tolyl)-2H- 1.72-1.57 (m, 3H) tetrazol-5-yl)methanone LCMS (m/z): 419.90 (7-methyl-4-(1-methyl-1H- 1H-NMR (400 MHz, DMSO-D6) δ 8.14-8.03 (m, 2H), pyrazol-4-yl)-4,7- 7.72-7.60 (m, 4H), 7.46-7.33 (m, 2H), 6.57 (dd, J = 12.0, 65 dihydrothieno[2,3-c]pyridin- 5.1 Hz, 1H), 5.96-5.35 (m, 1H), 4.79-3.48 (m, 6H), 1.72- 6(5H)-yl)(2-phenyl-2H-tetrazol- 1.57 (m, 3H) 5-yl)methanone LCMS (m/z): 405.95 (2-(3,5-dimethylphenyl)-2H- 1H-NMR (400 MHz, DMSO-D6) δ 7.73 (d, J = 17.6 Hz, tetrazol-5-yl)(7-methyl-4-(1- 2H), 7.66-7.60 (m, 1H), 7.46-7.34 (m, 2H), 7.28 (s, 1H), 66 methyl-1H-pyrazol-4-yl)-4,7- 6.56 (q, J = 5.1 Hz, 1H), 5.94-5.32 (m, 1H), 4.66-3.48 dihydrothieno[2,3-c]pyridin- (m, 6H), 2.40 (s, 6H), 1.71-1.57 (m, 3H) 6(5H)-yl)methanone LCMS (m/z): 434.15 (2-(4-chlorophenyl)-2H-tetrazol- 1H-NMR (400 MHz, DMSO-D6) δ 8.17-8.05 (m, 2H), 5-yl)(7-methyl-4-(1-methyl-1H- 7.77 (dt, J = 6.8, 2.1 Hz, 2H), 7.70-7.60 (m, 1H), 7.47- 67 pyrazol-4-yl)-4,7- 7.33 (m, 2H), 6.56 (dd, J = 11.2, 5.1 Hz, 1H), 5.94-5.34 dihydrothieno[2,3-c]pyridin- (m, 1H), 4.78-3.47 (m, 6H), 1.72-1.57 (m, 3H) 6(5H)-yl)methanone LCMS (m/z): 439.85 1 (2-(4-isopropoxyphenyl)-2H- H-NMR (400 MHz, DMSO-D6) δ 8.02-7.91 (m, 2H), tetrazol-5-yl)(7-methyl-4-(1- 7.70-7.59 (m, 1H), 7.46-7.33 (m, 2H), 7.20-7.15 (m, 68 methyl-1H-pyrazol-4-yl)-4,7- 2H), 6.56 (dd, J = 11.5, 5.1 Hz, 1H), 5.94-5.35 (m, 1H), dihydrothieno[2,3-c]pyridin- 4.75 (td, J = 11.9, 6.0 Hz, 1H), 4.65-3.47 (m, 6H), 1.71- 6(5H)-yl)methanone 1.56 (m, 3H), 1.30 (d, J = 5.9 Hz, 6H) LCMS (m/z): 464 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.17 (ddd, J (4-(1,5-dimethyl-1H-pyrazol-4- = 16.0, 8.4, 1.3 Hz, 2H), 7.61-7.52 (m, 3H), 7.19-7.12 69 yl)-4,7-dihydrothieno[2,3- (m, 2H), 6.68 (dd, J = 8.2, 5.3 Hz, 1H), 5.42-5.33 (m, c]pyridin-6(5H)-yl)(2-phenyl- 1H), 4.98 (dd, J = 80.0, 16.9 Hz, 1H), 4.68-4.18 (m, 2H), 2H-tetrazol-5-yl)methanone 3.87-3.46 (m, 4H), 2.23 (d, J = 56.7 Hz, 3H) LCMS (m/z): 406.25
PI External 1 (4-(1,5-dimethyl-1H-pyrazol-4- H-NMR (500 MHz, CHLOROFORM-D) δ 8.42 (d, J = yl)-4,7-dihydrothieno[2,3- 53.4 Hz, 1H), 7.75-7.71 (m, 2H), 7.28 (d, J = 8.8 Hz, 70 c]pyridin-6(5H)-yl)(1-(4- 1H), 7.23 (d, J = 4.9 Hz, 1H), 7.18-7.05 (m, 2H), 6.68 fluorophenyl)-1H-1,2,3-triazol- (dd, J = 27.6, 5.1 Hz, 1H), 5.98-4.89 (m, 2H), 4.87-4.53 4-yl)methanone (m, 1H), 4.35-3.45 (m, 5H), 2.30 (d, J = 12.5 Hz, 3H) LCMS (m/z): 422.85 1 (2-(2,4-difluorophenyl)-2H- H-NMR (500 MHz, CHLOROFORM-D) δ 8.20 (d, J = 1,2,3-triazol-4-yl)(4-(1,5- 84.1 Hz, 1H), 7.86-7.74 (m, 1H), 7.16-7.04 (m, 4H), 71 dimethyl-1H-pyrazol-4-yl)-4,7- 6.66 (dd, J = 5.0, 3.1 Hz, 1H), 5.67-5.27 (m, 1H), 5.10- dihydrothieno[2,3-c]pyridin- 4.85 (m, 1H), 4.63-4.56 (m, 1H), 4.22-4.13 (m, 1H), 6(5H)-yl)methanone 3.86-3.37 (m, 4H), 2.28-2.00 (m, 3H) LCMS (m/z): 440.85 (4-(1,5-dimethyl-1H-pyrazol-4- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.21-8.14 yl)-4,7-dihydrothieno[2,3- (m, 2H), 7.29 (d, J = 8.1 Hz, 2H), 7.21-7.15 (m, 2H), 72 c]pyridin-6(5H)-yl)(2-(4- 6.72-6.66 (m, 1H), 5.39-5.11 (m, 1H), 4.93-4.19 (m, fluorophenyl)-2H-tetrazol-5- 3H), 3.95-3.62 (m, 4H), 2.26 (d, J = 61.1 Hz, 3H) yl)methanone LCMS (m/z): 424.05 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.56 (dd, J (4-(1,5-dimethyl-1H-pyrazol-4- = 38.9, 2.4 Hz, 1H), 7.98-7.89 (m, 1H), 7.50 (q, J = 6.2 yl)-4,7-dihydrothieno[2,3- Hz, 1H), 7.38-7.31 (m, 2H), 7.19-7.12 (m, 2H), 6.68 (dd, 73 c]pyridin-6(5H)-yl)(1-(2- J = 21.0, 5.1 Hz, 1H), 5.34-5.24 (m, 1H), 4.90-4.52 (m, fluorophenyl)-1H-1,2,3-triazol- 2H), 4.26 (dt, J = 76.6, 6.4 Hz, 1H), 4.02 (dd, J = 13.0, 4-yl)methanone 8.1 Hz, 1H), 3.90-3.82 (m, 3H), 2.33 (d, J = 20.8 Hz, 3H); LCMS (m/z): 423.9 1 (4-(1,5-dimethyl-1H-pyrazol-4- H-NMR (400 MHz, DMSO-D6) δ 8.05-7.95 (m, 1H), yl)-4,7-dihydrothieno[2,3- 7.79-7.73 (m, 1H), 7.70-7.65 (m, 1H), 7.54 (t, J = 7.7 74 c]pyridin-6(5H)-yl)(2-(2- Hz, 1H), 7.38 (dd, J = 21.8, 5.1 Hz, 1H), 6.91 (d, J = fluorophenyl)-2H-tetrazol-5- 86.8 Hz, 1H), 6.62 (dd, J = 14.4, 5.1 Hz, 1H), 5.14-4.95 yl)methanone (m, 2H), 4.28-4.05 (m, 2H), 3.78-3.56 (m, 4H), 2.21- 1.96 (m, 3H); LCMS (m/z): 424.2 1 (1-(2,4-difluorophenyl)-1H- H-NMR (400 MHz, DMSO-D6) δ 8.92 (d, J = 99.0 Hz, 1,2,3-triazol-4-yl)(4-(1,5- 1H), 7.96-7.89 (m, 1H), 7.74-7.68 (m, 1H), 7.40-7.34 75 dimethyl-1H-pyrazol-4-yl)-4,7- (m, 2H), 6.92 (d, J = 85.8 Hz, 1H), 6.61 (dd, J = 21.4, dihydrothieno[2,3-c]pyridin- 5.3 Hz, 1H), 5.53-4.85 (m, 2H), 4.49-4.25 (m, 1H), 4.12 6(5H)-yl)methanone (d, J = 45.7 Hz, 1H), 4.01-3.54 (m, 4H), 2.14 (d, J = 39.9 Hz, 3H); LCMS (m/z): 441.2 1H-NMR (400 MHz, DMSO-D6) δ 9.16 (d, J = 94.6 Hz, (4-(1,5-dimethyl-1H-pyrazol-4- 1H), 7.95 (d, J = 7.8 Hz, 2H), 7.61 (t, J = 7.8 Hz, 2H), 76 yl)-4,7-dihydrothieno[2,3- 7.52 (t, J = 7.3 Hz, 1H), 7.36 (dd, J = 15.2, 5.1 Hz, 1H), c]pyridin-6(5H)-yl)(1-phenyl- 6.91 (d, J = 99.8 Hz, 1H), 6.62 (dd, J = 23.6, 5.0 Hz, 1H-1,2,3-triazol-4-yl)methanone 1H), 5.55-4.88 (m, 2H), 4.51-4.25 (m, 1H), 4.17-3.54 (m, 5H), 2.19-2.06 (m, 3H); LCMS (m/z): 405.1 1 (1-(2,4-difluorophenyl)-1H- H-NMR (400 MHz, DMSO-D6) δ 7.83 (d, J = 26.7 Hz, pyrazol-3-yl)(4-(1,5-dimethyl- 1H), 7.52-7.37 (m, 3H), 7.22 (d, J = 8.6 Hz, 1H), 6.93 77 1H-pyrazol-4-yl)-4,7- (d, J = 19.1 Hz, 1H), 6.83-6.60 (m, 1H), 6.47 (d, J = 66.8 dihydrothieno[2,3-c]pyridin- Hz, 1H), 4.91-4.64 (m, 2H), 4.14 (s, 1H), 3.90 (d, J = 6(5H)-yl)methanone 12.7 Hz, 1H), 3.70 (s, 3H), 3.58 (s, 1H), 2.11 (d, J = 16.9 Hz, 3H); LCMS (m/z): 440.1 (2-(2,4-difluorophenyl)-2H- 1 tetrazol-5-yl)(4-(1,5-dimethyl- H-NMR (400 MHz, DMSO-D6) δ 8.13-8.03 (m, 1H), 78 1H-pyrazol-4-yl)-4,7- 7.83-7.78 (m, 1H), 7.49-7.45 (m, 1H), 7.38 (dd, J = 21.0, dihydrothieno[2,3-c]pyridin- 5.1 Hz, 1H), 6.92 (d, J = 81.2 Hz, 1H), 6.62 (dd, J = 6(5H)-yl)methanone 13.1, 5.0 Hz, 1H), 5.15-4.94 (m, 2H), 4.28-4.05 (m, 2H),
PI External 3.77-3.69 (m, 2H), 3.57 (s, 2H), 2.21 (s, 1H), 1.96 (s, 2H); LCMS (m/z): 442.2 1 (4-(1,5-dimethyl-1H-pyrazol-4- H-NMR (500 MHz, DMSO-D6) δ 8.01-7.94 (m, 2H), yl)-4,7-dihydrothieno[2,3- 7.78-7.74 (m, 1H), 7.55-7.51 (m, 1H), 7.37 (dd, J = 29.5, 79 c]pyridin-6(5H)-yl)(2-(3- 5.1 Hz, 1H), 6.93 (d, J = 101.5 Hz, 1H), 6.61 (dd, J = fluorophenyl)-2H-tetrazol-5- 19.8, 5.1 Hz, 1H), 5.13-4.94 (m, 2H), 4.29-4.05 (m, 2H), yl)methanone 3.78-3.56 (m, 4H), 2.10 (d, J = 111.6 Hz, 3H) LCMS (m/z): 424.1 1 (2-(3,5-difluorophenyl)-2H- H-NMR (500 MHz, DMSO-D6) δ 7.95-7.86 (m, 2H), tetrazol-5-yl)(4-(1,5-dimethyl- 7.64 (tt, J = 9.2, 2.3 Hz, 1H), 7.37 (dd, J = 28.4, 5.1 Hz, 80 1H-pyrazol-4-yl)-4,7- 1H), 6.94 (d, J = 93.2 Hz, 1H), 6.61 (dd, J = 18.6, 5.1 dihydrothieno[2,3-c]pyridin- Hz, 1H), 5.14-4.93 (m, 2H), 4.29-4.03 (m, 2H), 3.76- 6(5H)-yl)methanone 3.58 (m, 4H), 2.10 (d, J = 107.2 Hz, 3H) LCMS (m/z): 442.2 1 (2-(3-fluorophenyl)-2H-tetrazol- H-NMR (500 MHz, DMSO-D6) δ 8.02-7.93 (m, 2H), 5-yl)(4-(1-me 7.78-7.74 (m, 1H), 7.55-7.05 (m, 4H), 6.74 (dd, J = 8.3, 81 thyl-1H-pyrazol-4- yl)-4,7-dihydrothieno[2,3- 5.1 Hz, 1H), 5.10-4.96 (m, 2H), 4.21-4.06 (m, 2H), 3.95- c]pyridin-6(5H)-yl)methanone 3.91 (m, 1H), 3.73 (d, J = 53.4 Hz, 3H) LCMS (m/z): 410 1H-NMR (400 MHz, DMSO-D6) δ 8.19-8.16 (m, 1H), (2-(4-fluorophenyl)-2H-tetrazol- 8.15-8.11 (m, 1H), 7.58-7.53 (m, 2H), 7.41-7.24 (m, 82 5-yl)(4-(1-methyl-1H-pyrazol-4- 2H), 7.05 (s, 1H), 6.73 (dd, J = 7.3, 5.1 Hz, 1H), 5.07 (d, yl)-4,7-dihydrothieno[2,3- J = 16.9 Hz, 1H), 5.00-4.96 (m, 1H), 4.19 (t, J = 9.2 Hz, c]pyridin-6(5H)-yl)methanone 2H), 4.12-3.89 (m, 1H), 3.78 (s, 1H), 3.67 (s, 2H) LCMS (m/z): 409.80 1 (4-(1-methyl-1H-pyrazol-4-yl)- H-NMR (400 MHz, DMSO-D6) δ 8.11 (dd, J = 19.4, 4,7-dihydrothieno[2,3-c]pyri 7.5 Hz, 2H), 7.72-7.63 (m, 3H), 7.54-7.05 (m, 3H), 6.73 83 din- 6(5H)-yl)(2-phenyl-2H-tetrazol- (dd, J = 7.1, 5.1 Hz, 1H), 5.10-4.96 (m, 2H), 4.23-4.08 5-yl)methanone (m, 2H), 3.97-3.89 (m, 1H), 3.78-3.67 (m, 3H) LCMS (m/z): 391.8 (2-(3,5-difluorophenyl)-2H- 1H-NMR (400 MHz, DMSO-D6) δ 7.95-7.87 (m, 2H), tetrazol-5-yl)(4-(1-methyl-1H- 7.66-7.61 (m, 1H), 7.53-7.06 (m, 3H), 6.75-6.72 (m, 84 pyrazol-4-yl)-4,7- 1H), 5.11-4.95 (m, 2H), 4.21-4.03 (m, 2H), 3.96-3.88 dihydrothieno[2,3-c]pyridin- (m, 1H), 3.73 (d, J = 39.9 Hz, 3H) 6(5H)-yl)methanone LCMS (m/z): 428 (1-(2-fluorophenyl)-1H-1,2,3- 1H-NMR (500 MHz, DMSO-D6) δ 8.91 (d, J = 140.5 triazol-4-yl)(4-(1-methyl-1H- Hz, 1H), 7.89-7.85 (m, 1H), 7.67-7.45 (m, 3H), 7.37- 85 pyrazol-4-yl)-4,7- 7.12 (m, 2H), 6.72 (s, 1H), 5.43-4.90 (m, 2H), 4.50-4.15 dihydrothieno[2,3-c]pyridin- (m, 3H), 3.84-3.70 (m, 3H) 6(5H)-yl)methanone LCMS (m/z): 409 (1-(2,4-difluorophenyl)-1H- 1H-NMR (400 MHz, DMSO-D6) δ 8.91 (d, J = 110.0 1,2,3-triazol-4-yl)(4-(1-methyl- Hz, 1H), 7.92 (dd, J = 14.5, 8.7 Hz, 1H), 7.74-7.68 (m, 86 1H-pyrazol-4-yl)-4,7- 1H), 7.52-7.11 (m, 4H), 6.71 (d, J = 5.1 Hz, 1H), 5.43- dihydrothieno[2,3-c]pyridin- 4.90 (m, 2H), 4.50-3.82 (m, 3H), 3.74 (d, J = 25.9 Hz, 6(5H)-yl)methanone 3H) LCMS (m/z): 427.05 (2-(2,4-difluorophenyl)-2H- 1H-NMR (400 MHz, DMSO-D6) δ 8.33 (d, J = 120.6 1,2,3-triazol-4-yl)(4-(1-methyl- Hz, 1H), 7.93 (d, J = 5.9 Hz, 1H), 7.71-7.65 (m, 1H), 87 1H-pyrazol-4-yl)-4,7- 7.52-7.04 (m, 4H), 6.72 (d, J = 5.1 Hz, 1H), 4.97 (d, J = dihydrothieno[2,3-c]pyridin- 22.3 Hz, 2H), 4.21-4.10 (m, 3H), 3.73 (d, J = 33.5 Hz, 6(5H)-yl)methanone 3H) LCMS (m/z): 427.00 1 (1-(2,4-difluorophenyl)-1H- H-NMR (500 MHz, DMSO-D6) δ 7.83 (d, J = 35.9 Hz, 88 pyrazol-3-yl)(4-(1-methyl-1H- 1H), 7.58-6.38 (m, 8H), 4.88-4.68 (m, 2H), 4.15-3.89 pyrazol-4-yl)-4,7- (m, 2H), 3.79-3.67 (m, 4H) LCMS (m/z): 426.1
PI External dihydrothieno[2,3-c]pyridin- 6(5H)-yl)methanone 1 (4-(1-methyl-1H-pyrazol-4-yl)- H-NMR (500 MHz, DMSO-D6) δ 7.98 (dd, J = 24.4, 4,7-dihydrothieno[2,3-c]pyri 8.5 Hz, 2H), 7.53-7.40 (m, 3H), 7.37-7.04 (m, 2H), 6.73 89 din- 6(5H)-yl)(2-(p-tolyl)-2H- (dd, J = 9.5, 5.1 Hz, 1H), 5.09-4.96 (m, 2H), 4.21-4.08 tetrazol-5-yl)methanone (m, 2H), 3.96-3.91 (m, 1H), 3.72 (d, J = 56.3 Hz, 3H), 2.42 (s, 3H); LCMS (m/z): 406.05 (1-(3,5-difluorophenyl)-1H- 1H-NMR (500 MHz, DMSO-D6) δ 9.29 (d, J = 87.1 Hz, 1,2,3-triazol-4-yl)(4-(1,5- 1H), 7.85 (d, J = 5.6 Hz, 2H), 7.49-7.45 (m, 1H), 7.40- 90 dimethyl-1H-pyrazol-4-yl)-4,7- 7.33 (m, 1H), 6.91 (d, J = 119.0 Hz, 1H), 6.64-6.58 (m, dihydrothieno[2,3-c]pyridin- 1H), 5.49-4.87 (m, 2H), 4.50-4.25 (m, 1H), 4.17-3.56 6(5H)-yl)methanone (m, 5H), 2.19-2.06 (m, 3H); LCMS (m/z): 441.05 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.46 (d, J = (4-(1-methyl-1H-pyrazol-4-yl)- 62.1 Hz, 1H), 7.75 (d, J = 7.3 Hz, 2H), 7.56 (t, J = 7.7 91 4,7-dihydrothieno[2,3-c]pyridin- Hz, 2H), 7.52-7.36 (m, 2H), 7.22-7.18 (m, 2H), 6.75 (d, 6(5H)-yl)(1-phenyl-1H-1,2,3- J = 4.9 Hz, 1H), 5.63 (dd, J = 79.6, 17.0 Hz, 1H), 5.06 triazol-4-yl)methanone (s, 1H), 4.75-4.44 (m, 1H), 4.36-3.88 (m, 5H) LCMS (m/z): 391 (7-methyl-4-(1-methyl-1H- 1H-NMR (400 MHz, DMSO-D6) δ 9.31 (d, J = 16.9 Hz, pyrazol-4-yl)-4,7- 1H), 7.94 (dd, J = 31.2, 7.9 Hz, 2H), 7.69-7.50 (m, 3H), 92 dihydrothieno[2,3-c]pyridin- 7.43-7.19 (m, 2H), 6.93-5.75 (m, 2H), 5.03-4.60 (m, 6(5H)-yl)(1-phenyl-1H-1,2,3- 1H), 4.22-3.98 (m, 2H), 3.82-3.47 (m, 4H), 1.76-1.52 triazol-4-yl)methanone (m, 3H); LCMS (m/z): 404.9 1H-NMR (500 MHz, CHLOROFORM-D) δ 8.02 (dd, J (2-(3-fluorophenyl)-2H-tetrazol- = 8.2, 2.0 Hz, 1H), 7.94 (dt, J = 9.0, 2.1 Hz, 1H), 7.60- 5-yl)(7-methyl-4-(1-methyl-1H- 7.54 (m, 1H), 7.41-7.37 (m, 1H), 7.33 (d, J = 6.4 Hz, 93 pyrazol-4-yl)-4,7- 1H), 7.30-7.28 (m, 1H), 7.141(dd, J = 17.5, 5.1 Hz, 1H), dihydrothieno[2,3-c]pyridin- 6.67 (dd, J = 5.2, 2.2 Hz, 1H), 5.99 (q, J = 6.7 Hz, 1H), 6(5H)-yl)methanone 5.54-5.50 (m, 1H), 4.94 (dd, J = 13.0, 5.3 Hz, 1H), 4.40- 4.34 (m, 1H), 3.92-3.70 (m, 3H), 1.85-1.68 (m, 3H) LCMS (m/z): 423.8 (1-(4-fluorophenyl)-1H-1,2,3- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.39 (d, J = triazol-4-yl)(4-(1-methyl-1H- 80.7 Hz, 1H), 7.72 (q, J = 4.5 Hz, 2H), 7.40-7.27 (m, 94 pyrazol-4-yl)-4,7- 2H), 7.23-7.13 (m, 3H), 6.75 (d, J = 5.1 Hz, 1H), 5.60 dihydrothieno[2,3-c]pyridin- (dd, J = 149.7, 16.6 Hz, 1H), 5.12-4.71 (m, 2H), 4.46- 6(5H)-yl)methanone 4.21 (m, 2H), 3.93-3.82 (m, 3H); LCMS (m/z): 409 (2-(4-fluorophenyl)-2H-tetrazol- 1 5-yl)(7-methyl-4-(1-methyl-1H- H-NMR (400 MHz, DMSO-D6) δ 8.20-8.07 (m, 2H), 95 pyrazol-4-yl)-4,7- 7.70-6.54 (m, 6H), 5.94-5.34 (m, 1H), 4.78-4.04 (m, dihydrothieno[2,3-c]pyridin- 2H), 3.90-3.47 (m, 4H), 1.72-1.57 (m, 3H) 6(5H)-yl)methanone LCMS (m/z): 424 (4-(1,5-dimethyl-1H-pyrazol-4- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.48 (d, J = yl)-4,7-dihydrothieno[2,3- 50.1 Hz, 1H), 7.59-7.50 (m, 3H), 7.22-7.00 (m, 3H), 96 c]pyridin-6(5H)-yl)(1-(3- 6.68 (dd, J = 24.6, 5.0 Hz, 1H), 6.05-4.62 (m, 3H), 4.32- fluorophenyl)-1H-1,2,3-triazol- 4.13 (m, 1H), 3.97-3.32 (m, 4H), 2.25 (d, J = 10.3 Hz, 4-yl)methanone 3H); LCMS (m/z): 422.8 (4-(5-chloro-1-methyl-1H- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.21-8.14 pyrazol-4-yl)-4,7- (m, 2H), 7.61-7.52 (m, 3H), 7.24-7.12 (m, 2H), 6.69 (dd, 97 dihydrothieno[2,3-c]pyridin- J = 7.0, 5.3 Hz, 1H), 5.30-5.24 (m, 1H), 5.03 (d, J = 16.9 6(5H)-yl)(2-phenyl-2H-tetrazol- Hz, 1H), 4.28 (dd, J = 14.7, 4.4 Hz, 2H), 3.95-3.70 (m, 5-yl)methanone 4H); LCMS (m/z): 425.95 (4-(5-chloro-1-methyl-1H- 1 98 pyrazol-4-yl)-4,7- H-NMR (500 MHz, CHLOROFORM-D) δ 8.21-8.14 dihydrothieno[2,3-c]pyridin- (m, 2H), 7.31-7.27 (m, 3H), 7.24-7.12 (m, 2H), 6.69 (dd,
PI External 6(5H)-yl)(2-(4-fluorophenyl)- J = 7.2, 5.1 Hz, 1H), 5.32-5.01 (m, 2H), 4.62-4.24 (m, 2H-tetrazol-5-yl)methanone 2H), 3.95-3.69 (m, 3H) LCMS (m/z): 443.95 (4-(5-chloro-1-methyl-1H- pyrazol-4-yl)-4,7- 1H-NMR (500 MHz, CHLOROFORM-D) δ 7.82-7.74 99 dihydrothieno[2,3-c]pyridin- (m, 2H), 7.23-7.12 (m, 2H), 7.03-6.99 (m, 1H), 6.70 (dd, 6(5H)-yl)(2-(3,5- J = 6.4, 5.3 Hz, 1H), 5.30-5.03 (m, 2H), 4.31-4.21 (m, difluorophenyl)-2H-tetrazol-5- 2H), 3.93-3.70 (m, 4H); LCMS (m/z): 461.95 yl)methanone 1 (4-(5-chloro-1-methyl-1H- H-NMR (400 MHz, CHLOROFORM-D) δ 8.53-8.37 pyrazol-4-yl)-4,7- (m, 1H), 7.77-7.73 (m, 2H), 7.56 (t, J = 7.6 Hz, 2H), 100 dihydrothieno[2,3-c]pyridin- 7.52-7.46 (m, 1H), 7.22-7.09 (m, 2H), 6.69 (q, J = 4.9 6(5H)-yl)(1-phenyl-1H-1,2,3- Hz, 1H), 5.65 (dd, J = 214.0, 16.9 Hz, 1H), 5.09 (dd, J = triazol-4-yl)methanone 29.7, 16.8 Hz, 1H), 4.82-4.51 (m, 1H), 4.39-3.63 (m, 5H) ; LCMS (m/z): 425 (4-(5-chloro-1-methyl-1H- 1 pyrazol-4-yl)-4,7- H-NMR (400 MHz, CHLOROFORM-D) δ 8.49 (dd, J dihydrothieno[2,3 = 53.8, 2.4 Hz, 1H), 7.94-7.87 (m, 1H), 7.22-7.08 (m, 101 -c]pyridin- 6(5H)-yl)(1-(2,4- 4H), 6.69 (dd, J = 8.3, 5.1 Hz, 1H), 5.90-4.96 (m, 2H), difluorophenyl)-1H-1,2,3- 4.84-4.51 (m, 1H), 4.33-4.23 (m, 2H), 3.86-3.76 (m, 3H) triazol-4-yl)methanone LCMS (m/z): 461 (4-(5-chloro-1-methyl-1H- 1 pyrazol-4-yl)-4,7- H-NMR (400 MHz, CHLOROFORM-D) δ 8.30-8.05 dihydrothie (m, 1H), 7.87-7.74 (m, 1H), 7.21-7.03 (m, 4H), 6.68 (d, 102 no[2,3-c]pyridin- 6(5H)-yl)(2-(2,4- J = 5.1 Hz, 1H), 5.61-5.09 (m, 2H), 4.57-4.40 (m, 1H), difluorophenyl)-2H-1,2,3- 4.22-4.20 (m, 1H), 4.16-3.61 (m, 4H) triazol-4-yl)methanone LCMS (m/z): 461.05 1 (4-(1,5-dimethyl-1H-pyrazol-4- H-NMR (400 MHz, CHLOROFORM-D) δ 7.70-7.63 yl)-4,7-dihydrothieno[2,3- (m, 1H), 7.44-7.41 (m, 1H), 7.24 (d, J = 7.8 Hz, 1H), 103 c]pyridin-6(5H)-yl)(2-(2-fluoro- 7.18-7.08 (m, 2H), 6.67 (q, J = 5.1 Hz, 1H), 5.40 (dd, J = 3-methylphenyl)-2H-tetrazol-5- 23.6, 16.5 Hz, 1H), 5.05-4.83 (m, 1H), 4.77-4.16 (m, yl)methanone 2H), 3.81-3.33 (m, 4H), 2.43 (t, J = 2.4 Hz, 3H), 2.18 (d, J = 60.4 Hz, 3H); LCMS (m/z): 438.2 (4-(5-chloro-1-methyl-1H- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.01 (dd, J pyrazol-4-yl)-4,7- = 13.4, 8.1 Hz, 1H), 7.93 (qt, J = 9.2, 2.2 Hz, 1H), 7.60- 104 dihydrothieno[2,3-c]pyridin- 7.52 (m, 1H), 7.28 (t, J = 1.2 Hz, 1H), 7.24-7.12 (m, 6(5H)-yl)(2-(3-fluorophenyl)- 2H), 6.71-6.68 (m, 1H), 5.30-5.01 (m, 2H), 4.63-4.23 2H-tetrazol-5-yl)methanone (m, 2H), 3.98-3.68 (m, 4H); LCMS (m/z): 443.75 (2-(3-bromophenyl)-2H-tetrazol- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.38 (dt, J = 5-yl)(4-(5-chloro-1-methyl-1H- 16.5, 1.9 Hz, 1H), 8.13 (dq, J = 8.3, 1.0 Hz, 1H), 7.68 105 pyrazol-4-yl)-4,7- (dq, J = 8.1, 1.0 Hz, 1H), 7.49-7.45 (m, 1H), 7.24-7.12 dihydrothieno[2,3-c]pyridin- (m, 2H), 6.71-6.68 (m, 1H), 5.14 (dd, J = 97.3, 16.9 Hz, 6(5H)-yl)methanone 2H), 4.31-3.72 (m, 6H); LCMS (m/z): 505.55 (2-(2,4-difluorophenyl)-2H- 1H-NMR (500 MHz, CHLOROFORM-D) δ 7.92-7.87 tetrazol-5-yl)(4-(1-methyl-1H- (m, 1H), 7.37-7.29 (m, 1H), 7.19-6.95 (m, 4H), 6.75 (dd, 106 pyrazol-4-yl)-4,7- J = 16.9, 5.1 Hz, 1H), 5.25 (dd, J = 16.7, 7.1 Hz, 1H), dihydrothieno[2,3-c]pyridin- 5.09-5.02 (m, 1H), 4.59-4.10 (m, 3H), 3.88-3.71 (m, 3H) 6(5H)-yl)methanone LCMS (m/z): 427.95 1 (4-(5-chloro-1-methyl-1H- H-NMR (400 MHz, CHLOROFORM-D) δ 8.21-8.18 pyrazol-4-yl)-7-methyl-4,7- (m, 2H), 7.30 (d, J = 9.3 Hz, 2H), 7.15 (dd, J = 19.0, 5.0 107 dihydrothieno[2,3-c]pyridin- Hz, 1H), 6.61 (dd, J = 13.0, 5.1 Hz, 1H), 6.01 (d, J = 6.8 6(5H)-yl)(2-(4-fluorophenyl)- Hz, 1H), 4.37-4.34 (m, 2H), 3.89-3.57 (m, 3H), 3.45 (dd, 2H-tetrazol-5-yl)methanone J = 14.8, 12.1 Hz, 2H), 1.86-1.69 (m, 3H) LCMS (m/z): 457.75
PI External 1 (4-(5-chloro-1-methyl-1H- H-NMR (400 MHz, CHLOROFORM-D) δ 8.03 (d, J = pyrazol-4-yl)-7-methyl-4,7- 8.1 Hz, 1H), 7.97-7.94 (m, 1H), 7.60-7.55 (m, 1H), 7.37- 108 dihydrothieno[2,3-c]pyridin- 7.28 (m, 2H), 7.15 (dd, J = 18.8, 5.1 Hz, 1H), 6.61 (dd, J 6(5H)-yl)(2-(3-fluorophenyl)- = 12.8, 5.0 Hz, 1H), 5.78 (dd, J = 184.3, 6.2 Hz, 1H), 2H-tetrazol-5-yl)methanone 4.96-3.21 (m, 6H), 1.79 (dd, J = 49.6, 6.8 Hz, 3H) LCMS (m/z): 457.8 (4-(5-chloro-1-methyl-1H- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.21-8.08 pyrazol-4-yl)-7-methyl-4,7- (m, 2H), 7.62-7.52 (m, 3H), 7.34 (d, J = 23.5 Hz, 1H), 109 dihydrothieno[2,3-c]pyridin- 7.15 (dd, J = 19.4, 5.0 Hz, 1H), 6.61 (dd, J = 13.7, 5.1 6(5H)-yl)(2-phenyl-2H-tetrazol- Hz, 1H), 6.02-5.58 (m, 1H), 4.38-4.34 (m, 2H), 3.89- 5-yl)methanone 3.21 (m, 4H), 1.87-1.69 (m, 3H); LCMS (m/z): 440 (4-(5-chloro-1-methyl-1H- 1H-NMR (500 MHz, CHLOROFORM-D) δ 8.66-8.56 pyrazol-4-yl)-7-methyl-4,7- (m, 1H), 7.99-7.86 (m, 1H), 7.52-7.45 (m, 1H), 7.40- 110 dihydrothieno[2,3-c]pyridin- 7.29 (m, 3H), 7.21-7.11 (m, 1H), 6.76-6.59 (m, 1H), 6(5H)-yl)(1-(2-fluorophenyl)- 6.48-5.93 (m, 1H), 5.47-4.86 (m, 1H), 4.38-3.17 (m, 1H-1,2,3-triazol-4-yl)methanone 5H), 1.90-1.65 (m, 3H); LCMS (m/z): 458.65 (4-(5-chloro-1-methyl-1H- 1 pyrazol-4-yl)-7-methyl-4,7- H-NMR (500 MHz, CHLOROFORM-D) δ 8.57-8.13 111 dihydrothieno[2,3-c]pyridin- (m, 1H), 7.59-7.47 (m, 3H), 7.23-6.78 (m, 2H), 6.73- 6(5H)-yl)(1-(3-fluorophenyl)- 6.58 (m, 1H), 6.48-5.93 (m, 1H), 5.48-3.17 (m, 7H), 1H-1,2,3-triazol-4-yl)methanone 1.89-1.64 (m, 3H); LCMS (m/z): 457.05 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.56 (d, J = (4-(5-chloro-1-methyl-1H- 5.6 Hz, 1H), 7.76 (d, J = 7.8 Hz, 2H), 7.57 (t, J = 7.2 Hz, pyrazol-4-yl)-7-methyl-4,7- 2H), 7.49 (t, J = 7.2 Hz, 1H), 7.36 (d, J = 4.6 Hz, 1H), 112 dihydrothieno[2,3-c]pyridin- 7.15-7.12 (m, 1H), 6.60 (dd, J = 13.0, 5.1 Hz, 1H), 6.51- 6(5H)-yl)(1-phenyl-1H-1,2,3- 5.93 (m, 1H), 5.45-4.85 (m, 1H), 4.38-4.13 (m, 1H), triazol-4-yl)methanone 3.88 (d, J = 5.6 Hz, 3H), 3.50-3.17 (m, 1H), 1.79 (dd, J = 80.2, 6.8 Hz, 3H) LCMS (m/z): 439.05 (4-(5-chloro-1-methyl-1H- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.31 (d, J = pyrazol-4-yl)-7-methyl-4,7- 4.2 Hz, 1H), 7.90-7.80 (m, 1H), 7.33 (d, J = 16.9 Hz, 113 dihydrothieno[2,3-c]pyridin- 1H), 7.15-7.00 (m, 2H), 6.59 (d, J = 4.9 Hz, 1H), 5.99 6(5H)-yl)(2-(2,4- (dd, J = 26.7, 6.4 Hz, 1H), 4.90-4.82 (m, 1H), 4.31-4.12 difluorophenyl)-2H-1,2,3- (m, 2H), 3.86 (d, J = 18.8 Hz, 3H), 3.29 (dt, J = 97.7, triazol-4-yl)methanone 12.2 Hz, 1H), 2.01-1.68 (m, 3H) LCMS (m/z): 474.85 (4-(5-chloro-1-methyl-1H- 1H-NMR (500 MHz, CHLOROFORM-D) δ 7.72-7.54 pyrazol-4-yl)-7-methyl-4,7- (m, 1H), 7.44-7.40 (m, 1H), 7.37-7.27 (m, 2H), 7.25- 114 dihydrothieno[2,3-c]pyridin- 7.12 (m, 1H), 6.75-6.58 (m, 1H), 6.15-5.56 (m, 1H), 6(5H)-yl)(2-(2-fluoro-3- 5.08-4.02 (m, 2H), 3.91-3.80 (m, 3H), 3.63-3.22 (m, methylphenyl)-2H-tetrazol-5- 1H), 2.48-2.40 (m, 3H), 1.87-1.68 (m, 3H) LCMS yl)methanone (m/z): 472.15 1 (4-(5-chloro-1-methyl-1H- H-NMR (400 MHz, CHLOROFORM-D) δ 8.07-7.95 pyrazol-4-yl)-7-methyl-4,7- (m, 2H), 7.38-7.31 (m, 2H), 7.15 (dd, J = 19.3, 5.1 Hz, 115 dihydrothieno[2,3-c]pyridin- 1H), 6.60 (dd, J = 14.3, 5.3 Hz, 1H), 6.14-5.98 (m, 1H), 6(5H)-yl)(2-(p-tolyl)-2H- 5.60-4.92 (m, 1H), 4.62-4.01 (m, 2H), 3.89-3.81 (m, tetrazol-5-yl)methanone 3H), 3.48-3.21 (m, 1H), 2.46 (s, 3H), 1.86-1.68 (m, 3H) LCMS (m/z): 453.85 (4-(5-chloro-1-methyl-1H- 1 pyrazol-4-yl)-7-methyl-4,7- H-NMR (500 MHz, CHLOROFORM-D) δ 8.56-8.15 dihydrothien (m, 1H), 7.42-7.30 (m, 2H), 7.22-6.58 (m, 3H), 6.44- 116 o[2,3-c]pyridin- 6(5H)-yl)(1-(3,5- 5.92 (m, 1H), 5.45-4.84 (m, 1H), 4.36-4.06 (m, 2H), difluorophenyl)-1H-1,2,3- 3.90-3.18 (m, 4H), 1.89-1.64 (m, 3H) triazol-4-yl)methanone LCMS (m/z): 475.05 117 (2-(3-bromophenyl)-2H-tetrazol- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.41-8.16 5-yl)(4-(5-chloro-1-methyl-1H- (m, 2H), 7.69 (d, J = 8.1 Hz, 1H), 7.52-7.45 (m, 1H),
PI External pyrazol-4-yl)-7-methyl-4,7- 7.34 (d, J = 24.5 Hz, 1H), 7.15 (dd, J = 18.7, 5.0 Hz, dihydrothieno[2,3-c]pyridin- 1H), 6.61 (dd, J = 13.4, 5.1 Hz, 1H), 6.00 (d, J = 6.4 Hz, 6(5H)-yl)methanone 1H), 4.93-4.31 (m, 2H), 3.89-3.81 (m, 3H), 3.62-3.42 (m, 1H), 1.86-1.69 (m, 3H); LCMS (m/z): 519.8 (4-(5-chloro-1-methyl-1H- 1 pyrazol-4-yl)-7-methyl-4,7- H-NMR (500 MHz, CHLOROFORM-D) δ 7.81-7.66 dihydrothien (m, 2H), 7.36-7.28 (m, 1H), 7.23-7.11 (m, 1H), 7.01- 118 o[2,3-c]pyridin- 6(5H)-yl)(2-(3,5- 6.86 (m, 1H), 6.71-6.58 (m, 1H), 6.10-5.49 (m, 1H), difluorophenyl)-2H-tetrazol-5- 4.95-4.01 (m, 2H), 3.87-3.21 (m, 4H), 1.83-1.68 (m, 3H) yl)methanone LCMS (m/z): 475.8 1 (4-(5-chloro-1-methyl-1H- H-NMR (500 MHz, CHLOROFORM-D) δ 8.58 (dd, J pyrazol-4-yl)-7-methyl-4,7- = 12.6, 2.5 Hz, 1H), 7.95-7.91 (m, 1H), 7.38-7.35 (m, dihydroth 1H), 7.15-7.07 (m, 1H), 6.80-6.71 (m, 1H), 6.61 (dd, J = 119 ieno[2,3-c]pyridin- 6(5H)-yl)(1-(2,4- 15.1, 5.2 Hz, 1H), 6.45-5.94 (m, 1H), 5.47-5.30 (m, 1H), difluorophenyl)-1H-1,2,3- 4.38-4.07 (m, 2H), 3.88 (d, J = 8.4 Hz, 3H), 3.49-3.19 triazol-4-yl)methanone (m, 1H), 1.67 (dd, J = 18.7, 6.8 Hz, 3H) LCMS (m/z): 475.1 1 (4-(5-chloro-1-methyl-1H- H-NMR (500 MHz, CHLOROFORM-D) δ 8.53-8.09 pyrazol-4-yl)-7-methyl-4,7- (m, 1H), 7.79-7.67 (m, 2H), 7.39-7.27 (m, 1H), 7.24- 120 dihydrothieno[2,3-c]pyridin- 6.76 (m, 2H), 6.74-6.58 (m, 1H), 6.49-5.93 (m, 1H), 6(5H)-yl)(1-(4-fluorophenyl)- 5.52-4.85 (m, 1H), 4.37-4.02 (m, 2H), 3.92-3.74 (m, 1H-1,2,3-triazol-4-yl)methanone 3H), 3.55-3.17 (m, 1H), 1.93-1.64 (m, 3H) LCMS (m/z): 457.05 (4-(5-chloro-1-methyl-1H- pyrazol-4-yl)-7-methyl-4,7- 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.75-7.43 121 dihydrothieno[2,3-c]pyridin- (m, 2H), 7.21 (d, J = 5.1 Hz, 1H), 7.17-6.64 (m, 5H), 6(5H)-yl)(1-(2,4- 5.36 (s, 1H), 4.08-3.86 (m, 5H), 3.21-3.72 (1H), 1.66- difluorophenyl)-1H-pyrazol-3- 1.25 (m, 3H) LCMS (m/z): 474.1 yl)methanone (4-(5-chloro-1-methyl-1H- 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.95-7.68 pyrazol-4-yl)-7-methyl-4,7- (m, 2H), 7.31 (d, J = 31.3 Hz, 1H), 7.20-6.69 (m, 3H), 122 dihydrothieno[2,3-c]pyridin- 6.59-6.38 (m, 1H), 6.14-5.94 (m, 1H), 5.07-4.81 (m, 6(5H)-yl)(1-(2,4- 1H), 4.33-3.95 (m, 1H), 3.88-3.63 (m, 4H), 3.39-3.15 difluorophenyl)-1H-pyrazol-4- (m, 1H), 1.82-1.63 (m, 3H) yl)methanone LCMS (m/z): 474.05 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.36 (dt, J = (2-(3-bromophenyl)-2H-tetrazol- 24.0, 1.9 Hz, 1H), 8.13 (ddq, J = 21.7, 8.2, 1.0 Hz, 1H), 123 5-yl)(4-(1-methyl-1H-pyrazol-4- 7.69-7.66 (m, 1H), 7.46 (td, J = 8.1, 4.0 Hz, 1H), 7.33 yl)-4,7-dihydrothieno[2,3- (d, J = 33.5 Hz, 1H), 7.19-6.98 (m, 2H), 6.75 (dd, J = c]pyridin-6(5H)-yl)methanone 13.9, 5.1 Hz, 1H), 5.27-5.04 (m, 2H), 4.60-4.20 (m, 2H), 4.11-3.69 (m, 4H) LCMS (m/z): 471.9 (1-(3-fluorophenyl)-1H-1,2,3- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.43 (d, J = triazol-4-yl)(4-(1-methyl-1H- 86.1 Hz, 1H), 7.58-7.52 (m, 3H), 7.25-7.11 (m, 4H), 124 pyrazol-4-yl)-4,7- 6.75 (q, J = 2.5 Hz, 1H), 5.60 (dd, J = 206.1, 16.6 Hz, dihydrothieno[2,3-c]pyridin- 1H), 5.15-4.76 (m, 2H), 4.55-3.61 (m, 5H) 6(5H)-yl)methanone LCMS (m/z): 408.95 (2-(2-fluoro-3-methylphenyl)- 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.69-7.63 2H-tetrazol-5-yl)(4-(1-methyl- (m, 1H), 7.43-7.40 (m, 1H), 7.29 (s, 1H), 7.24-6.99 (m, 125 1H-pyrazol-4-yl)-4,7- 3H), 6.75 (dd, J = 12.2, 5.1 Hz, 1H), 5.29-5.06 (m, 2H), dihydrothieno[2,3-c]pyridin- 4.59-3.69 (m, 6H), 2.42 (d, J = 2.2 Hz, 3H) LCMS 6(5H)-yl)methanone (m/z): 424.1 (4-(5-chlo 1 126 ro-1-methyl-1H- H-NMR (400 MHz, CHLOROFORM-D) δ 8.21-8.13 pyrazol-4-yl)-7-methyl-4,7- (m, 2H), 7.31-7.27 (m, 2H), 7.25-7.15 (m, 2H), 6.72 (dd, dihydrothieno[2,3-c]pyridin- J = 16.9, 5.1 Hz, 1H), 5.40 (dd, J = 22.9, 16.8 Hz, 1H),
PI External 6(5H)-yl)(2-(2,4- 5.13-4.84 (m, 1H), 4.64-4.25 (m, 2H), 3.98-3.89 (m, difluorophenyl)-2H-tetrazol-5- 3H), 3.77-3.53 (m, 3H); LCMS: 449 yl)methanone 1 (4-(5-(chloromethyl)-1-methyl- H-NMR (400 MHz, CHLOROFORM-D) δ 8.03-7.96 1H-pyrazol-4-yl)-4,7- (m, 1H), 7.95-7.88 (m, 1H), 7.60-7.52 (m, 1H), 7.28- 127 dihydrothieno[2,3-c]pyridin- 7.28 (m, 1H), 7.25 (d, J = 5.4 Hz, 1H), 7.21-7.17 (m, 6(5H)-yl)(1-(2,4- 1H), 6.72 (dd, J = 17.4, 5.1 Hz, 1H), 5.38 (dd, J = 24.2, difluorophenyl)-1H-1,2,3- 16.6 Hz, 1H), 4.99 (ddd, J = 88.0, 16.6, 1.5 Hz, 1H), triazol-4-yl)methanone 4.64-4.25 (m, 2H), 3.95-3.89 (m, 3H), 3.76-3.53 (m, 3H); LCMS: 448.8 (4-(5-(chloromethyl)-1-methyl- 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.93-7.85 128 1H-pyrazol-4-yl)-4,7- (m, 1H), 7.24 (t, J = 5.4 Hz, 1H), 7.21-7.09 (m, 3H), dihydrothieno[2,3-c]pyridin- 6.71 (dd, J = 16.1, 5.1 Hz, 1H), 5.40-5.33 (m, 1H), 5.12- 6(5H)-yl)(1-(4-fluorophenyl)- 4.88 (m, 1H), 4.63-4.24 (m, 2H), 3.97-3.84 (m, 3H), 1H-1,2,3-triazol-4-yl)methanone 3.76-3.54 (m, 3H); LCMS: 467.05 (4-(5-(chloromethyl)-1-methyl- 1H-NMR (500 MHz, CHLOROFORM-D) δ 8.50 (d, J = 129 1H-pyrazol-4-yl)-4,7- 2.6 Hz, 1H), 8.13 (t, J = 6.2 Hz, 1H), 7.93-7.88 (m, 1H), dihydrothieno[2,3-c]pyridin- 7.46 (d, J = 0.9 Hz, 1H), 7.34 (d, J = 0.5 Hz, 1H), 7.12- 6(5H)-yl)(2-(4-fluorophenyl)- 7.07 (m, 2H), 4.06 (d, J = 6.4 Hz, 2H), 3.89 (s, 3H), 2.42 2H-tetrazol-5-yl)methanone (s, 3H); LCMS: 477.9 (4-(5-(chloromethyl)-1-methyl- 1H-pyrazol-4-yl)-4,7- 1H-NMR (500 MHz, CHLOROFORM-D) δ 8.58-8.56 130 dihydrothieno[2,3-c]pyridin- (m, 1H), 7.92-7.87 (m, 1H), 7.43 (d, J = 0.8 Hz, 1H), 6(5H)-yl)(2-(2,4- 7.35 (s, 1H), 7.16-7.09 (m, 2H), 4.09-4.08 (m, 2H), 3.88 difluorophenyl)-2H-tetrazol-5- (s, 3H), 2.42 (s, 3H), 1.78 (s, 3H); LCMS: 479.05 yl)methanone (4-(5-chloro-1-methyl-1H- 1H-NMR (500 MHz, CHLOROFORM-D) δ 8.54-8.51 131 pyrazol-4-yl)-4,7- (m, 1H), 8.21-8.17 (m, 2H), 7.43 (d, J = 0.7 Hz, 1H), dihydrothieno[2,3-c]pyridin- 7.35 (d, J = 0.6 Hz, 1H), 7.29-7.27 (m, 2H), 4.09-4.08 6(5H)-yl)(1-(3-fluorophenyl)- (m, 2H), 3.88 (s, 3H), 2.43 (d, J = 0.3 Hz, 3H), 1.77 (s, 1H-1,2,3-triazol-4-yl)methanone 3H); LCMS: 461 (4-(5-chloro-1-methyl-1H- 1 pyrazol-4-yl)-4,7- H-NMR (500 MHz, CHLOROFORM-D) δ 8.58-8.56 dihydrothieno[2,3-c]pyridin- (m, 1H), 8.03 (dd, J = 8.2, 1.6 Hz, 1H), 7.96 (dt, J = 9.0, 132 6(5H)-yl)(2-(2-fluoro-3- 2.2 Hz, 1H), 7.57 (td, J = 8.3, 5.8 Hz, 1H), 7.44 (s, 1H), methylphenyl)-2H-tetrazol-5- 7.35 (s, 1H), 7.25-7.24 (m, 1H), 4.10-4.08 (m, 2H), 3.88 yl)methanone (s, 3H), 2.44 (s, 3H), 1.78 (s, 3H); LCMS: 461.05 (4-(5-chloro-1-methyl-1H- 1 pyrazol-4-yl)-4,7- H-NMR (500 MHz, CHLOROFORM-D) δ 8.48-8.34 133 dihydrothieno[2,3-c]pyridin- (m, 1H), 7.75-7.71 (m, 2H), 7.29-7.27 (m, 1H), 7.25- 6(5H)-yl)(1-(2,4- 7.23 (m, 1H), 7.21-7.09 (m, 2H), 6.69 (dd, J = 11.2, 5.0 difluorophenyl)-1H-pyrazol-3- Hz, 1H), 5.91-5.02 (m, 2H), 4.82-4.49 (m, 1H), 4.37- yl)methanone 3.66 (m, 5H); LCMS: 443.1 1 (4-(5-(chloromethyl)-1-methyl- H-NMR (400 MHz, CHLOROFORM-D) δ 8.21-8.01 1H-pyrazol-4-yl)-4,7- (m, 2H), 7.61-7.50 (m, 2H), 7.24-7.11 (m, 2H), 6.83- 134 dihydrothieno[2,3-c]pyridin- 6.60 (m, 1H), 6.18-5.55 (m, 1H), 4.98-4.26 (m, 1H), 6(5H)-yl)(1-(2-fluorophenyl)- 4.15-3.10 (m, 4H), 2.97-2.80 (m, 3H), 2.27-2.20 (m, 1H-1,2,3-triazol-4-yl)methanone 1H), 2.07-1.98 (m, 1H), 1.86-1.69 (m, 3H); LCMS: 420.05 2-(1-methyl-4-(6-(2-phenyl-2H- 1H-NMR (400 MHz, CHLOROFORM-D) δ 7.83-7.64 135 tetrazole-5-carbonyl)-4,5,6,7- (m, 2H), 7.24-6.81 (m, 3H), 6.74-6.60 (m, 1H), 6.16- tetrahydrothieno[2,3-c]pyridin- 5.46 (m, 1H), 4.97-4.09 (m, 1H), 4.05-3.12 (m, 5H), 4-yl)-1H-pyrazol-5- 2.28 (d, J = 14.7 Hz, 1H), 1.86-1.82 (m, 2H), 1.74-1.69 yl)acetonitrile (m, 3H); LCMS: 456.05
PI External 2-(4-(6-(2-(4-fluorophenyl)-2H- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.04-7.79 136 tetrazole-5-carbonyl)-4,5,6,7- (m, 2H), 7.61-7.52 (m, 1H), 7.24-6.81 (m, 2H), 6.73- tetrahydrothieno[2,3-c]pyridin- 6.60 (m, 1H), 6.17-5.51 (m, 1H), 4.97-4.09 (m, 2H), 4-yl)-1-methyl-1H-pyrazol-5- 4.03-3.11 (m, 5H), 2.29-2.22 (m, 1H), 1.88-1.82 (m, yl)acetonitrile 2H), 1.71 (dd, J = 10.9, 6.7 Hz, 3H); LCMS: 438.1 1 2-(4-(6-(2-(3-fluorophenyl)-2H- H-NMR (400 MHz, CHLOROFORM-D) δ 8.25-8.05 tetrazole-5-carbonyl)-4,5,6, (m, 2H), 7.31-7.27 (m, 1H), 7.24-7.11 (m, 2H), 6.83 (d, 137 7- tetrahydrothieno[2,3-c]pyridin- J = 3.9 Hz, 1H), 6.67 (ddd, J = 37.3, 11.1, 5.1 Hz, 1H), 4-yl)-1-methyl-1H-pyrazol-5- 6.17-5.54 (m, 1H), 4.98-3.90 (m, 2H), 3.84-3.48 (m, yl)acetonitrile 4H), 2.30-2.25 (m, 1H), 1.86-1.81 (m, 2H), 1.74-1.69 (m, 3H); LCMS: 438.05 2-(4-(6-(2-(2,4-difluorophenyl)- 1 2H H-NMR (400 MHz, CHLOROFORM-D) δ 7.94-7.79 138 -tetrazole-5-carbonyl)- 4,5,6,7-tetrahydrothieno[2,3- (m, 1H), 7.22-6.79 (m, 4H), 6.73-6.59 (m, 1H), 6.17- c]pyridin-4-yl)-1-methyl-1H- 5.52 (m, 1H), 4.97-3.11 (m, 6H), 2.68-1.28 (m, 6H); pyrazol-5-yl)acetonitrile LCMS: 455.95 (4-(5-chloro-1-methyl-1H- 1 pyrazol-4-yl)-4,7- H-NMR (400 MHz, CHLOROFORM-D) δ 8.62 (t, J = 139 dihydrothieno[2,3-c]pyridin- 6.0 Hz, 1H), 7.85-7.78 (m, 2H), 7.43 (d, J = 0.7 Hz, 1H), 6(5H)-yl)(1-(4-fluorophenyl)- 7.35 (s, 1H), 7.03-6.98 (m, 1H), 4.08 (d, J = 6.4 Hz, 2H), 1H-1,2,3-triazol-4-yl)methanone 3.89 (s, 3H), 2.44 (s, 3H), 1.78 (s, 3H); LCMS: 479.25 (4-(1,5-dimethyl-1H-pyrazol-4- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.42 (s, 140 yl)-7-methyl-4,7- 1H), 8.11 (t, J = 6.2 Hz, 1H), 7.73-7.70 (m, 2H), 7.45 (d, dihydrothieno[2,3-c]pyridin- J = 0.7 Hz, 1H), 7.34 (s, 1H), 7.25-7.23 (m, 2H), 4.06 (d, 6(5H)-yl)(2-phenyl-2H-tetrazol- J = 6.4 Hz, 2H), 3.88 (s, 3H), 2.43 (s, 3H), 1.76 (s, 3H); 5-yl)methanone LCMS: 460.15 (2-(3,5-difluorophenyl)-2H- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.27 (s, 141 tetrazol-5-yl)(4-(1,5-dimethyl- 1H), 8.12 (t, J = 6.1 Hz, 1H), 7.87-7.82 (m, 1H), 7.44 (d, 1H-pyrazol-4-yl)-7-methyl-4,7- J = 0.7 Hz, 1H), 7.33 (s, 1H), 7.10-7.02 (m, 2H), 4.05- dihydrothieno[2,3-c]pyridin- 3.95 (m, 2H), 3.88 (s, 3H), 2.39 (s, 3H), 1.75 (s, 3H); 6(5H)-yl)methanone LCMS: 478.3 (4-(1,5-dimethyl-1H-pyrazol-4- 1H-NMR (500 MHz, CHLOROFORM-D) δ 8.64 (t, J = 142 yl)-7-methyl-4,7- 6.0 Hz, 1H), 8.21-8.19 (m, 2H), 7.60-7.52 (m, 3H), 7.48 dihydrothieno[2,3-c]pyridin- (s, 1H), 4.25 (dd, J = 13.5, 5.2 Hz, 1H), 4.12 (dd, J = 6(5H)-yl)(2-(3-fluorophenyl)- 13.5, 7.3 Hz, 1H), 3.83 (s, 3H), 2.46 (s, 3H), 1.82 (s, 2H-tetrazol-5-yl)methanone 3H); LCMS: 477 (4-(1,5-dimethyl-1H-pyrazol-4- 1H-NMR (500 MHz, CHLOROFORM-D) δ 8.42 (s, yl)-7-methyl-4,7- 1H), 8.23 (t, J = 6.1 Hz, 1H), 7.74-7.70 (m, 2H), 7.48 (s, 143 dihydrothieno[2,3-c]pyridin- 1H), 7.27 (s, 1H), 7.25-7.23 (m, 1H), 4.21 (dd, J = 13.5, 6(5H)-yl)(2-(4-fluorophenyl)- 5.4 Hz, 1H), 4.11 (dd, J = 13.5, 7.4 Hz, 1H), 3.82 (s, 2H-tetrazol-5-yl)methanone 3H), 2.44 (s, 3H), 1.80 (s, 3H); LCMS: 493.95 (2-(2,4-difluorophenyl)-2H- 1H-NMR (400 MHz, CHLOROFORM-D) δ 8.27 (s, tetrazol-5-yl)(4-(1,5-dimethyl- 1H), 8.25-8.22 (m, 1H), 7.88-7.81 (m, 1H), 7.47 (s, 1H), 144 1H-pyrazol-4-yl)-7-methyl-4,7- 7.10-7.00 (m, 2H), 4.15 (dd, J = 13.4, 5.1 Hz, 1H), 4.04 dihydrothieno[2,3-c]pyridin- (dd, J = 13.4, 7.6 Hz, 1H), 3.82 (s, 3H), 2.40 (s, 3H), 6(5H)-yl)methanone 1.79 (s, 3H); LCMS: 512.2 BIOLOGY EXAMPLES: As described herein the compounds of the general formula (I) show fungicidal activity which is exerted with respect to numerous phytopathogenic fungi which attack on important agricultural crops. The compounds of the present invention were assessed for their activity as described in the following tests:
PI External Example 1: Pyricularia oryzae (Rice blast): The compounds were dissolved in 0.3% dimethyl sulfoxide and then added to potato dextrose agar medium just prior to dispensing it into petri dishes. 5 mL medium, with the compound in the desired test concentration, was dispensed into a 60 mm sterile petri-plate. After solidification, each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate of Pyricularia oryzae. The plates were incubated in growth chambers at 25 oC temperature and 60% relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control. Compounds 1, 3, 4, 7, 9, 10, 12, 32, 33, 36, 37, 39, 46, 47, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 64, 65, 67, 68, 72, 73, 74, 75, 78, 79, 80, 81, 82, 83, 84, 92, 93, 94, 95, 96, 98, 103, 104, 105, 107, 108, 109, 110, 111, 112, 114, 115, 117, 118, 119, 120, 123, 125, 140, 141, 142, 143, 144 at 300 ppm gave more than or equal to 70 % control in these tests when compared to the untreated check which showed extensive pathogen growth. Example 2: Alternaria solani (early blight of tomato/potato): The compounds were dissolved in 0.3% dimethyl sulfoxide and then added to potato dextrose agar medium just prior to dispensing it into petri dishes. 5 mL medium, with the compound in the desired concentration, was dispensed into a 60 mm sterile petri-plate. After solidification, each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate of Alternaria solani. The plates were incubated in growth chambers at 25 oC temperature and 60% relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control. Compounds 1, 3, 4, 10, 14, 33, 36, 37, 39, 49, 51, 53, 54, 55, 56, 57, 58, 59, 60, 61, 64, 65, 67, 69, 70, 71, 72, 73, 74, 75, 76, 78, 79, 80, 81, 82, 83, 84, 85, 86, 92, 93, 95, 96, 97, 98, 99, 100, 101, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 114, 115, 116, 117, 118, 119, 120, 123, 125, 126, 127, 128, 130, 132, 135, 136, 137, 138, 140, 141, 142, 143, 144 at 300 ppm gave more than or equal to 70 % control in these tests when compared to the untreated check which showed extensive pathogen growth. Example 3: Colletotrichum capsici (anthracnose): The compounds were dissolved in 0.3% dimethyl sulfoxide and then added to potato dextrose agar medium just prior to dispensing it into petri dishes. 5 mL medium, with the compound in the desired concentration, was dispensed into a 60 mm sterile petri-plate. After solidification, each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate of Colletotrichum capsici. The plates were incubated in growth chambers at 25 oC temperature and 60%
PI External relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control. Compounds 1, 3, 4, 10, 13, 19, 22, 25, 31, 32, 33, 35, 36, 37, 39, 49, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 64, 65, 66, 67, 69, 70, 72, 73, 74, 75, 76, 78, 79, 80, 81, 82, 83, 84, 85, 86, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 114, 115, 116, 117, 118, 119, 120, 123, 125, 126, 129, 131, 132, 135, 136, 137, 139, 140, 141, 142, 143, 144 at 300 ppm gave more than or equal to 70 % control in these tests when compared to the untreated check which showed extensive pathogen growth. Example 4: Corynespora cassiicola (Leaf spot of tomato): The compounds were dissolved in 0.3% dimethyl sulfoxide and then added to potato dextrose agar medium just prior to dispensing it into petri dishes. 5 mL medium, with the compound in the desired concentration, was dispensed into a 60 mm sterile petri-plate. After solidification, each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate of Corynespora cassiicola. The plates were incubated in growth chambers at 25 oC temperature and 70% relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control. Compounds 1, 3, 10, 25, 28, 32, 33, 36, 37, 39, 51, 53, 54, 57, 58, 59, 60, 61, 72, 73, 74, 81, 84, 86, 91, 92, 104, 106, 110, 118, 119, 120, 123, 125, 129, 130, 132, 137 at 300 ppm gave more than or equal to 70 % control in these tests when compared to the untreated check which showed extensive pathogen growth. Example 5: Phytophthora infestans (Late blight of potato & tomato): The compounds were dissolved in 0.3% dimethyl sulfoxide and then added to Rye Agar medium just prior to dispensing it into petri dishes. 5 mL medium, with the compound in the desired test concentration, was dispensed into a 60 mm sterile petri-plate. After solidification, each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate of Phytophthora infestans. The plates were incubated in growth chambers at 18 ºC temperature and 95% relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control. Compound 10, 50, 60, 77, 121 at 300 ppm gave more than or equal to 70 % control in these tests when compared to the untreated check which showed extensive pathogen growth. Example 6: Botrytis cinerea (Gray mold): Liquid testing Yeast, bacterial peptone and sodium acetate (YBA) liquid medium containing a BOTRCI (104 spores/mL) spore suspension was prepared. For the inhibition assay, each test compound was solved in dimethyl sulfoxide and 100 µl of the respective test solution was given into a well of a 96-well microtiter
PI External plate, consequently, the same volume (100 µl) of the media and spore suspension was added to each well to obtain the final test concentration. The plates were incubated at 22 °C for 15-18 days. The growth inhibition was evaluated by measuring the OD600. Percent inhibition was calculated with the below formula: I= (C-B)-(T-B)/(C-B)*100 where T=treatment, C=control, and B=blank Compounds 3, 4, 7, 10, 11, 19, 20, 25, 28, 30, 31, 33, 34, 36, 37, 42, 43, 45, 48, 49, 51, 52, 53, 54, 55, 57, 58, 60, 61, 64, 65, 66, 68, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 88, 89, 90, 91, 92, 97, 98, 99, 100, 101, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 113, 114, 115, 116, 117, 118, 119, 120, 123, 125, 126, 128, 129, 131, 135, 136, 140, 141, 142, 143, 144 at 300 ppm gave more than or equal to 70 % control in these tests when compared to the untreated check which showed extensive pathogen growth Example 7: Fusarium culmorum (Foot rot/Head blight of cereals): Compounds were dissolved in 0.3% dimethyl sulfoxide and then added to Potato Dextrose Agar medium just prior to dispensing it into petri dishes. 5ml medium with compound in the desired concentration was dispensed into 60mm sterile petri-plates. After solidification each plate was seeded with a 5mm size mycelial disc taken from the periphery of actively growing virulent culture plates. Plates were incubated in growth chambers at 25°C temperature and 60% relative humidity for seven days and radial growth was measured. Compounds 1, 69, 72, 73, 74, 75, 79, 80, 81, 82, 83, 84, 86, 93, 97, 98, 101, 104, 107, 108, 109, 118, 140, 141, 142, 143 at 300 ppm gave more than or equal to 70 % control in these tests when compared to the untreated check which showed extensive pathogen growth. Example 8: Sclerotinia sclerotiorum (White mold): Compounds were dissolved in 0.3% dimethyl sulfoxide and then added to potato dextrose agar medium just prior to dispensing it into petri dishes. 5 mL medium with the compound in the desired test concentration was dispensed into 60 mm sterile petri-plates. After solidification, each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate. Plates were incubated in growth chambers at 20 oC temperature and 70% relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control. Compounds 1, 3, 4, 5, 10, 15, 22, 23, 24, 28, 31, 32, 33, 36, 37, 40, 41, 42, 43, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 65, 69, 70, 71, 72, 73, 74, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 89, 91, 92, 93, 94, 95, 96, 97, 98, 99, 103, 104, 105, 106, 107, 108, 109, 110, 111, 112, 114, 115, 116, 117, 118, 119, 120, 123, 125, 129, 131, 132, 139, 140, 141, 142, 143, 144 at 300 ppm gave more than or equal to 70 % control in these tests when compared to the untreated check which showed extensive pathogen growth.
PI External Example 9: Rhizoctonia solani (Rice sheath blight/Potato black scurf): Compounds were dissolved in 0.3% dimethyl sulfoxide and then added to potato dextrose agar medium just prior to dispensing it into petri dishes. 5 mL medium with a compound in the desired test concentration was dispensed into 60 mm sterile petri-plates. After solidification each plate was seeded with a 5 mm size mycelial disc taken from the periphery of an actively growing virulent culture plate. Plates were incubated in growth chambers at 25 ℃ temperature and 60% relative humidity for seven days and the radial growth was measured and compared to the one of the untreated control. Compound 72 at 300 ppm gave more than or equal to 70% control in these tests when compared to the untreated check which showed extensive pathogen growth. Example 10: Septoria tritici Potato dextrose (PDB) liquid medium (Difco) containing a Septoria tritici (105 spores/mL) spore suspension was prepared. For the inhibition assay, each test compound was dissolved in dimethyl sulfoxide.100 µL of the test media-solution was added to a 96-well microtiter plate, consequently, the same volume (100 µL) of spore suspension was added to the well making the final test concentrations and the plate was incubated at 18 °C for 15 to 18 days. After the period of incubation, the length of pycnidiospores were measured under a microscope using software for image acquisition and analysis. Then, for each condition of the test, a mean length of pycnidiospores is calculated by averaging the size of pycnidiospores. From this mean length of pycnidiospores, the efficacy (E) of the product for each condition can be calculated by comparison to the mean length of pycnidiospores obtained in the control condition (untreated) according to the formula: E = 100 x [(C-T)/C] C is the mean length of pycnidiospores obtained for the control condition (untreated) and T is the mean length of pycnidiospores for conditions treated with the specific concentration (c) of the fungicide. Compounds 1, 4, 7, 19, 22, 25, 31, 32, 33, 37, 45, 46, 49, 50, 51, 52, 53, 57, 59, 64, 65, 66, 67, 68 at 300 ppm gave more than or equal to 70 % control in these tests when compared to the untreated check which showed extensive disease development. Green House Biological Activity: Example A: Phakopsora pachyrhizi test on soybean plants The compounds were dissolved in 2% dimethyl sulfoxide/acetone and then mixed with water containing emulsifier to the calibrated spray volume of 30 mL. Each spray solution was poured into a spray bottle for further application. To test the preventive activity of the compound, healthy young soybean plants, raised in the greenhouse, were sprayed with the active compound preparation at the stated application rate inside the spray cabinets using hollow cone nozzles. One day after treatment, the plants were inoculated with a conidial suspension containing 2 x105 Phakopsora pachyrhizi inoculum. The inoculated plants were then kept
PI External in a greenhouse chamber at 22-24 °C temperature and 80-90% relative humidity for disease expression. A visual assessment of the compound’s performance was carried out by rating the disease severity (0- 100% scale) on treated plants 3, 7, 10 and 15 days after application. Efficacy (% control) of the compound was calculated by comparing the disease rating in the treatment with the one of the untreated control. The treated plants were also assessed for plant damage by recording symptoms like necrosis, chlorosis and stunting. Compounds 1, 2, 3, 4, 5, 6, 14, 23, 27, 33, 39, 41, 42, 47, 49, 54, 61, 62, 65, 68, 70, 71, 87, 110, 111, 112, 116, 118 at 500 ppm gave more than or equal to 70 % control in these tests when compared to the untreated check which showed extensive disease development. Example B: Pseudoperonospora cubensis on cucumber plants The compounds were dissolved in 2% dimethyl sulfoxide/acetone and then mixed with water containing emulsifier to the desired test concentration in a calibrated spray volume of 30ml. The test solutions were poured into the spray bottles for further applications. To test the preventive activity of compounds, healthy young cucumber plants, raised in the greenhouse were sprayed with the active compound preparation at the stated application rates inside the spray cabinets using hallow cone nozzles. One day after treatment, the plants were inoculated with a conidial spore suspension containing 2x104Pseudoperonospora cubensis inoculum. The inoculated plants were then kept in a greenhouse chamber at 230C temperature & 80-90 % relative humidity for disease expression. A visual assessment of the performance of the compounds was carried out by rating the disease severity (0-100% scale) on treated plants 3, 7, 10 and 15 days after application. Efficacy (% control) of the compounds was calculated by comparing the disease rating in the treatment with the one of the untreated control. The compounds were also assessed for their plant compatibility by recording symptoms like necrosis, chlorosis and stunting. Compound 21 at 500 ppm gave more than or equal to 70% control in these tests when compared to the untreated check which showed extensive disease development. Example C: Septoria nodorum test in wheat The single compounds were dissolved in 2% DMSO/ Acetone and then mixed with water containing an emulsifier to a calibrated spray volume of 30 mL. The spray solutions were poured into spray bottles for further applications. To test the preventive activity of the compounds and respective composition, healthy young wheat plants, raised in the greenhouse, were sprayed with the single compound or respective composition at
PI External the stated application rates inside the spray cabinets using hollow cone nozzles. One day after treatment, the plants were inoculated with a suspension containing 2.8x106 Septoria nodorum inoculum. The inoculated plants were then kept in a greenhouse chamber at 22-25 °C temperature and 90-100 % relative humidity for disease expression. A visual assessment of the performance of the compounds and the respective composition was carried out by rating the disease severity (0-100% scale) on treated plants 3, 7 and 10 days after application. Efficacy (% control) of the compounds and composition was calculated by comparing the disease rating in the treatment with the one of the untreated, inoculated control plants. The compounds and the respective compositions were also assessed for their plant compatibility by recording symptoms like necrosis, chlorosis & stunting. Compounds 1, 31, 33, 39, 86, 87, 90, 92, 103, 105, 109, 110, 111, 112, 114, 115, 116, 118 at 500 ppm gave more than or equal to 70% control in these tests when compared to the untreated check which showed extensive disease development. Having described the invention with reference to certain preferred embodiments, other embodiments will become apparent to one skilled in the art from the consideration of the specification. It will be apparent to those skilled in the art that many modifications, both to materials and methods, may be practiced without departing from the scope of the invention.
Claims
PI External CLAIMS: 1. A compound of formula (I) or an agriculturally acceptable salt, N-oxide, isomer, tautomer or polymorph thereof,
wherein, D is selected from the group consisting of phenyl or 5- or 6-membered heteroaryl ring, wherein said 5- or 6-membered heteroaryl ring comprises 1, 2, 3 or 4 heteroatoms each independently selected from N, O, and S(O)0-2, and wherein said phenyl or 5- or 6-membered heteroaryl ring may be further substituted with one or more different or identical substituents selected from R1b and/or R1c; R1a is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, nitro, C1-C6- alkyl, C1-C6-haloalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C3-C8-cycloalkyl, C3-C8-halocycloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C2-C6-alkenyloxy, C2-C6-haloalkenyloxy, C2-C6-alkynyloxy, C2-C6-haloalkynyloxy, C3-C6-cycloalkyloxy, C1-C6- alkylthio, C1-C6-haloalkylthio, C3-C6-cycloalkylthio, C1-C6-alkylsulphinyl, C1-C6- haloalkylsulphinyl, C1-C6-alkylsulfonyl, C1-C6-haloalkylsulfonyl, -N(R7)2, -CO-N(R9)2, - S(O)(R8)=NR9, -N=S(O)(R8)2, P(O)(R8)2, Si(R8)3, or phenyl which may be optionally substituted with one or more different or identical substituents selected from halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy or C1-C6-haloalkoxy; R1b is selected from the group consisting of hydrogen, halogen, hydroxyl, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy and C1-C6-haloalkoxy; R1c is selected from the group consisting of hydrogen, halogen, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, hydroxyl and cyano; R2 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy;
PI External R3 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C1-C6-cyanoalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6- haloalkynyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R4a and R4b are each independently selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; or R4a and R4b may together form an oxo group (=O), or a 3- to 6-membered carbocyclic ring; Z1, Z2 and Z3 are each independently selected from CR5 or N; R5 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl and C1-C6-alkoxy; B represents a 5-membered aromatic heterocyclic ring fused at C3, C4 carbons of the piperidine ring, wherein the 5-membered aromatic heterocyclic ring comprises 1 to 3 heteroatoms selected from oxygen, nitrogen or S(O)0-2, wherein the ring B is unsubstituted or substituted with one or more different or identical substituents selected from R6; R6 can be selected from the group consisting of hydrogen, halogen, cyano, hydroxyl, C1-C6- alkyl, C1-C6-haloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C3-C8-cycloalkyl, C3-C8- halocycloalkyl, C6-C10-aryl which may be substituted with 1 or 2 same or different substitutent selected from the group consisting of halogen, C1-C6-alkyl, C1-C6-haloalkyl, C1-C6-alkoxy and C1-C6-haloalkoxy, and -N(R7)2; R7 represents hydrogen, C1-C6-alkyl, C3-C8-cycloalkyl, C1-C6-alkylcarbonyl, C3-C8- cycloalkylcarbonyl, C1-C6-alkoxycarbonyl or C1-C6-alkylsulfonyl; R8 is selected from the group consisting of hydrogen, halogen, C1-C6-alkyl, C1-C6-haloalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6-haloalkynyl, C3-C8-cycloalkyl, C1-C6- alkoxy and C1-C6-haloalkoxy; R9 is selected from the group consisting of hydrogen, C1-C6-alkyl, C1-C6-haloalkyl and C3-C8- cycloalkyl; R10 is selected from the group consisting of hydrogen and C1-C6-alkyl; R1a and R1b optionally together along with the phenyl or 5- or 6-membered heteroaryl ring may form an 8- to 10-membered bicyclic carbocyclic or heterocyclic ring system that can be optionally substituted with one or more different or identical substituents selected from R1b and/or R1c, wherein the bicyclic heterocyclic ring system comprises one or more heteroatoms/group selected from nitrogen, oxygen, S, S(O) or S(O)2, and wherein one or more carbon atom of the bicyclic carbocyclic or heterocyclic ring system can be replaced with C(O).
PI External 2. The compound of formula (I) as claimed in claim 1, wherein the compound is selected from formula (I-A):
, wherein B represents thiophenyl (thienyl) ring; R1a-R1c, R2, R3, R4a, R4b, R10 and Z1-Z3 are as defined in claim 1; and n = 1-3. 3. The compound of formula (I) as claimed in claim 2, wherein for the compound of formula (I- A), R1a is selected from the group consisting of halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3- C6-cycloalkyl, C1-C6-alkoxy, C1-C6-haloalkoxy, C3-C6-cycloalkyloxy, C1-C6-alkylthio, C1-C6- haloalkylthio, C1-C6-alkylsulfonyl; R1b is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl and C3-C6-cycloalkyl; R1c is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl and C1-C6- haloalkyl; R2 is selected from hydrogen, or halogen; R3 is selected from the group consisting of hydrogen, halogen, C1-C6-alkyl, C1-C6-haloalkyl and C1-C6-cyanoalkyl; R4a is selected from the group consisting of hydrogen and C1-C6-alkyl; R4b is selected from the group consisting of hydrogen and C1-C6-alkyl; or R4a and R4b may together form an oxo group (=O) or cyclopropyl ring; ring C =
selected from one of the following rings C-1 to C-7:
PI External
wherein R5 is independently selected from the group consisting of hydrogen, halogen, C1-C6- alkyl, C1-C6-haloalkyl and C3-C8-cycloalkyl and R5 can be same or different in the C-7 imidazolyl ring; R10 is selected from hydrogen or C1-C6-alkyl and n is 1-3. 4. The compound of formula (I) as claimed in claim 2, wherein for the compound of formula (I- A): R1a is selected from fluoro, chloro, bromo, iodo, cyano, methyl, trifluoromethyl, -O-isopropyl, or cyclopropyl; R1b is selected from hydrogen, fluoro, chloro, bromo, cyano, methyl, trifluoromethyl and cyclopropyl; R1c is selected from the group consisting of hydrogen, fluoro, chloro, bromo or cyano; R2 is selected from hydrogen, or chloro; R3 is selected from hydrogen, chloro, or methyl, CH2Cl or CH2CN; R4a is selected from hydrogen or methyl; R4b is selected from hydrogen;
wherein R5 is independently selected from hydrogen, chloro, methyl, ethyl, trifluoromethyl or cyclopropyl and R5 can be same or different in the C-7 imidazolyl ring;
PI External R10 is selected from hydrogen or methyl and n is 1-3. 5. The compound of formula (I) as claimed in claim 2, wherein the compound is selected from formula (I-D), formula (I-E) or formula (I-F):
wherein B, R1a-R1c, R2, R3, R4a, R4b, R10 and Z1-Z3 are as defined in claim 1; and n = 1-3. 6. A process for preparing the compound of formula (I-A), wherein said process comprising step- i or steps-ii-iii or steps iv-v to obtain the compound of formula (I-A): i. reacting an amine compound of formula 1 or salt thereof with a compound of formula 2 or formula 3 in a solvent, and in the presence of an optional coupling reagent/activating agent and a base, to obtain the compound of formula (I-A),
, or ii. reacting a compound of formula 1 or salt thereof with a compound of formula 10 to obtain a compound of formula 4,
PI External
; iii. treating the compound of formula 4 with a compound of 5 and a base in the presence of a transition metal catalyst and a solvent, to obtain the compound of formula (I-A),
, or iv. reacting a compound of formula 1 or salt thereof with a compound of formula 6 in a solvent to obtain a compound of formula 7,
, and v. oxidizing the compound of formula 7 in the presence of a metal oxide or oxygen in a solvent to obtain the compound of formula (I-A),
, wherein B, R1a-R1c, R2, R3, R4a, R4b, R10 and Z1-Z3 are as defined in claim 1. 7. A method for preparing an intermediate compound of formula 9A, wherein said method comprising the steps of:
PI External a. cyclizing a compound of formula 11 with a compound of formula 12 in a solvent and using a base to obtain a compound of formula 13,
; b. reacting the compound of formula 13 with a compound of formula 14 in the presence of a transition metal catalyst and a base to obtain a compound of formula 15,
; c. hydrolyzing the compound of formula 15 to a compound of formula 16,
d. decarboxylation of the compound of formula 16 to a compound of formula 17,
; e. reducing the compound of formula 17 using NaBH4 and iodine (I2) in a solvent to obtain the compound of formula 9A,
PI External
, wherein R2, R3 and R4a are as defined in claim 5; R12 = C1-C6 alkyl; R13 = Cl, Br or I; R14= H, C1-C6-alkyl or together form a 5-membered ring which may be substituted with one or more C1-C6-alkyl. 8. A method for preparing a compound of formula 9A, wherein said method comprising the steps f. hydrolyzing a compound of formula 13 to a corresponding acid compound of formula 18,
; g. decarboxylation of the compound of formula 18 to a compound of formula 19,
h. reacting the compound of formula 19 with a compound of formula 14 using a transition- metal catalyst and a base in a solvent to obtain a compound of formula 17,
i. reducing the compound of formula 17 with NaBH4 and iodine (I2) in a solvent to obtain the compound of formula 9A,
PI External
, wherein R2, R3 and R4a are as defined in claim 5; R12 = C1-C6 alkyl; R13 = Cl, Br or I; R14= H, C1-C6-alkyl or together form a 5-membered ring which may be substituted with one or more C1-C6-alkyl. 9. A method for preparing a compound of formula (I-D-a), wherein said method comprising the step of: j. reacting the compound of formula 9A obtained according to claim 9 or claim 10, with a compound of formula 2A in the presence of a coupling reagent and a solvent, to obtain the compound of formula (I-D-a),
, wherein ring C, R2, R3, R4a, and R1a-R1c, are as defined in claim 5. 10. A method for preparing a compound of formula (1), wherein said method comprising the step of: A. reducing a compound of formula (1A) with NaBH4 and iodine (I2) in a solvent to obtain a compound of formula (1),
wherein, B represents a phenyl ring or a 5- or 6-membered aromatic heterocyclic ring; R2 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy;
PI External R3 is selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6- haloalkyl, C1-C6-cyanoalkyl, C2-C6-alkenyl, C2-C6-haloalkenyl, C2-C6-alkynyl, C2-C6- haloalkynyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; R4a and R4b are each independently selected from the group consisting of hydrogen, halogen, cyano, C1-C6-alkyl, C1-C6-haloalkyl, C3-C8-cycloalkyl, C1-C6-alkoxy, and C1-C6-haloalkoxy; or R4a and R4b may together form an oxo group (=O), or a 3- to 6-membered carbocyclic ring. 11. A composition for controlling or preventing plant fungal diseases, wherein said composition comprising the compound of formula (I) as claimed in claim 1 and an agrochemically acceptable auxiliary. 12. The composition as claimed in claim 11, wherein said composition may further comprise an additional active ingredient selected from fungicide, insecticide, nematicide, acaricide, biopesticide, herbicide, safener, plant growth regulator, antibiotics, fertilizer or nutrients. 13. The composition as claimed in claim 11, wherein said composition is applied to a seed and the amount of the compound of formula (I) as claimed in claim 1 in said composition is ranges from 0.1 gai to 10 kgai per 100 kg of seeds. 14. The composition as claimed in claim 11, wherein the plant fungal disease is caused by rust pathogens selected from the group comprising of Hemileia vastatrix, Uromyces appendiculatus/fabae/phaseoli, Puccinia spp. on various plants, and Phakopsora spp. on various plants. 15. The composition as claimed in claim 14, wherein the Puccinia spp. on various plants is selected from P. triticina, P. striiformis, P. Hordei, P. graminis or P. recondita on cereals selected from wheat, barley or rye and Phakopsora spp. on various plants is selected from Phakopsora pachyrhizi and Phakopsora meibomiae on soybeans. 16. A combination comprising the compound of formula (I) as claimed in claim 1 and a pesticidally active substance selected from the group consisting of fungicide, insecticide, nematicide, acaricide, biopesticide, herbicide, safener, plant growth regulator, antibiotic, fertilizer and nutrient. 17. A method for controlling or preventing phytopathogenic fungi, wherein the method comprises treating the fungi or the materials, plants, plant parts, locus thereof, soil or seeds to be protected against fungal attack, with an effective amount of the compound of formula (I) as claimed in claim 1 or the composition as claimed in claim 11 or the combination as claimed in claim 16. 18. A method for controlling or preventing infestation of plants by phytopathogenic micro- organisms in agricultural crops and or horticultural crops wherein an effective amount of the
PI External compound of formula (I) as claimed in claim 1 or the composition as claimed in claim 11 or the combination as claimed in claim 16, is applied to the seeds of plants. 19. A compound of formula
, wherein the compound is selected from: (4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)(1-(4-fluorophenyl)-1H- 1,2,4-triazol-3-yl)methanone (compound 2); (1-(2,4-difluorophenyl)-1H-pyrazol-4-yl)(4-(1,5- dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 3); (2- (2,4-difluorophenyl)-2H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)methanone (compound 5); (1-(2-chloro-4-methylphenyl)-5- methyl-1H-1,2,3-triazol-4-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)- yl)methanone (compound 6); (5-methyl-1-(2,4,6-trichlorophenyl)-1H-1,2,3-triazol-4-yl)(4-(1- methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 11); (1-(2- chloro-4-fluorophenyl)-5-ethyl-1H-1,2,3-triazol-4-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)methanone (compound 12); (1-(3,5-bis(trifluoromethyl)phenyl)- 1H-1,2,3-triazol-4-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)- yl)methanone (compound 13); (1-(2,4-dichlorophenyl)-5-(trifluoromethyl)-1H-1,2,3-triazol-4- yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 14); (1-(4-chloro-2-fluorophenyl)-5-methyl-1H-1,2,3-triazol-4-yl)(4-(1-methyl-1H-pyrazol-4- yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 15); (1-(2-chloro-4- methylphenyl)-5-(trifluoromethyl)-1H-1,2,3-triazol-4-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)methanone (compound 16); 3-fluoro-4-(4-(4-(1-methyl-1H- pyrazol-4-yl)-1,2,3,4-tetrahydroisoquinoline-2-carbonyl)-5-(trifluoromethyl)-1H-1,2,3- triazol-1-yl)benzonitrile (compound 17); (1-(2,4-dichloro-6-methylphenyl)-5-methyl-1H- 1,2,3-triazol-4-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 18); (1-(3-chloro-5-(trifluoromethyl)phenyl)-1H-1,2,3-triazol-4-yl)(4-(1-methyl- 1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 19); (1-(2-chloro-4- fluorophenyl)-5-isopropyl-1H-1,2,3-triazol-4-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)methanone (compound 20); (4-(1,5-dimethyl-1H-pyrazol-4-yl)- 3,4-dihydroisoquinolin-2(1H)-yl)(5-methyl-1-(2,4,6-trichlorophenyl)-1H-1,2,3-triazol-4- yl)methanone (compound 21); (1-(4-chloro-2-methylphenyl)-1H-1,2,3-triazol-4-yl)(4-(1,5- dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 22); (1- (2-chloro-4-fluorophenyl)-5-ethyl-1H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)- 3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 23); (1-(2-chloro-4-methylphenyl)-5- methyl-1H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin- 2(1H)-yl)methanone (compound 24); (1-(3,5-bis(trifluoromethyl)phenyl)-1H-1,2,3-triazol-4- yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 25); (1-(2,4-dichlorophenyl)-5-(trifluoromethyl)-1H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-
PI External pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 26); (1-(2,4-dichloro-6- methylphenyl)-5-methyl-1H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)methanone (compound 27); (1-(4-chloro-2-fluorophenyl)-5- methyl-1H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin- 2(1H)-yl)methanone (compound 28); (1-(2-chloro-4-methylphenyl)-5-(trifluoromethyl)-1H- 1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)- yl)methanone (compound 29); 4-(4-(4-(1,5-dimethyl-1H-pyrazol-4-yl)-1,2,3,4- tetrahydroisoquinoline-2-carbonyl)-5-(trifluoromethyl)-1H-1,2,3-triazol-1-yl)-3- fluorobenzonitrile (compound 30); (1-(3-chloro-5-(trifluoromethyl)phenyl)-1H-1,2,3-triazol- 4-yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 31); (1-(3,5-dichlorophenyl)-1H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol- 4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 32); (1-(2,4-difluorophenyl)-5- methyl-1H-1,2,3-triazol-4-yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin- 2(1H)-yl)methanone (compound 34); (4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)(1-phenyl-1H-1,2,3-triazol-4-yl)methanone (compound 36); (4- (1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)(1-(2-fluorophenyl)-1H- 1,2,3-triazol-4-yl)methanone (compound 37); (1-(4-fluorophenyl)-1H-1,2,4-triazol-3-yl)(4-(1- methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 38); (4- (1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)(1-(4-fluorophenyl)-1H- pyrazol-4-yl)methanone (compound 39); (1-(2,4-difluorophenyl)-5-methyl-1H-pyrazol-3- yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 40); (1-(2,4-difluorophenyl)-5-methyl-1H-pyrazol-3-yl)(4-(1,5-dimethyl-1H-pyrazol-4-yl)- 3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 42); (4-(1,5-dimethyl-1H-pyrazol-4- yl)-3,4-dihydroisoquinolin-2(1H)-yl)(1-(4-fluorophenyl)-1H-pyrazol-3-yl)methanone (compound 43); (2-(2-fluorophenyl)-2H-1,2,3-triazol-4-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)methanone (compound 44); (1-(2,4-difluorophenyl)-1H-pyrazol- 4-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 45); (4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)(2-(2-fluorophenyl)- 2H-1,2,3-triazol-4-yl)methanone (compound 48); (4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)(1-(2-fluorophenyl)-1H-pyrazol-4-yl)methanone (compound 49); (2-(2,4-difluorophenyl)-2H-1,2,3-triazol-4-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)methanone (compound 50); (2-(2,4-difluorophenyl)-2H-tetrazol- 5-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 51); (4-(1,5-dimethyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)(2-(4-fluorophenyl)- 2H-1,2,3-triazol-4-yl)methanone (compound 53); (1-(2,4-difluorophenyl)-1H-pyrazol-4-yl)(1- methyl-4-(1-methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 54); (1-(2,4-difluorophenyl)-1H-1,2,3-triazol-4-yl)((1S,4R)-1-methyl-4-(1- methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 56); (1- (2,4-difluorophenyl)-1H-1,2,3-triazol-4-yl)((1S,4S)-1-methyl-4-(1-methyl-1H-pyrazol-4-yl)- 3,4-dihydroisoquinolin-2(1H)-yl)methanone (compound 57); (4-(1,5-dimethyl-1H-pyrazol-4- yl)-3,4-dihydroisoquinolin-2(1H)-yl)(2-(2-fluorophenyl)-2H-tetrazol-5-yl)methanone (compound 58); (2-(2-fluorophenyl)-2H-tetrazol-5-yl)(4-(1-methyl-1H-pyrazol-4-yl)-3,4- dihydroisoquinolin-2(1H)-yl)methanone (compound 59); or (1-(2,4-difluorophenyl)-1H-1,2,3- triazol-4-yl)(1,4-dimethyl-4-(1-methyl-1H-pyrazol-4-yl)-3,4-dihydroisoquinolin-2(1H)- yl)methanone (Compound 61).
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN202411000328 | 2024-01-03 | ||
| IN202411000328 | 2024-01-03 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025146624A1 true WO2025146624A1 (en) | 2025-07-10 |
Family
ID=94481106
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IB2025/050007 Pending WO2025146624A1 (en) | 2024-01-03 | 2025-01-02 | Bicyclic heterocyclic amides for combating phytopathogenic fungi |
Country Status (2)
| Country | Link |
|---|---|
| TW (1) | TW202542133A (en) |
| WO (1) | WO2025146624A1 (en) |
Citations (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| WO2014085490A1 (en) | 2012-11-29 | 2014-06-05 | Chemocentryx, Inc. | Cxcr7 antagonists |
| WO2015185485A1 (en) | 2014-06-06 | 2015-12-10 | Basf Se | Use of substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017093019A1 (en) | 2015-12-03 | 2017-06-08 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018019929A1 (en) | 2016-07-28 | 2018-02-01 | Idorsia Pharmaceuticals Ltd | Piperidine cxcr7 receptor modulators |
| WO2021094434A1 (en) | 2019-11-15 | 2021-05-20 | Bayer Aktiengesellschaft | Substituted hydantoinamides as adamts7 antagonists |
| WO2022253645A1 (en) | 2021-06-01 | 2022-12-08 | Syngenta Crop Protection Ag | Microbiocidal tetrahydroisoquinoline derivatives |
| WO2023110869A1 (en) | 2021-12-15 | 2023-06-22 | Syngenta Crop Protection Ag | Microbiocidal bicycle heterocyclic derivatives |
| WO2024068950A1 (en) * | 2022-09-30 | 2024-04-04 | Syngenta Crop Protection Ag | Microbiocidal pyrazole derivatives |
| WO2024105104A1 (en) * | 2022-11-16 | 2024-05-23 | Syngenta Crop Protection Ag | Microbiocidal tetrahydroisoquinoline derivatives |
| WO2024115546A1 (en) * | 2022-11-30 | 2024-06-06 | Syngenta Crop Protection Ag | Fungicidal compositions |
| WO2024256433A1 (en) * | 2023-06-14 | 2024-12-19 | Syngenta Crop Protection Ag | Fungicidal compositions |
-
2025
- 2025-01-02 WO PCT/IB2025/050007 patent/WO2025146624A1/en active Pending
- 2025-01-03 TW TW114100388A patent/TW202542133A/en unknown
Patent Citations (20)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0451878A1 (en) | 1985-01-18 | 1991-10-16 | Plant Genetic Systems, N.V. | Modifying plants by genetic engineering to combat or control insects |
| EP0374753A2 (en) | 1988-12-19 | 1990-06-27 | American Cyanamid Company | Insecticidal toxines, genes coding therefor, antibodies binding them, transgenic plant cells and plants expressing these toxines |
| EP0392225A2 (en) | 1989-03-24 | 1990-10-17 | Ciba-Geigy Ag | Disease-resistant transgenic plants |
| EP0427529A1 (en) | 1989-11-07 | 1991-05-15 | Pioneer Hi-Bred International, Inc. | Larvicidal lectins and plant insect resistance based thereon |
| WO1993007278A1 (en) | 1991-10-04 | 1993-04-15 | Ciba-Geigy Ag | Synthetic dna sequence having enhanced insecticidal activity in maize |
| WO1995034656A1 (en) | 1994-06-10 | 1995-12-21 | Ciba-Geigy Ag | Novel bacillus thuringiensis genes coding toxins active against lepidopteran pests |
| WO2002015701A2 (en) | 2000-08-25 | 2002-02-28 | Syngenta Participations Ag | Bacillus thuringiensis crystal protein hybrids |
| WO2003018810A2 (en) | 2001-08-31 | 2003-03-06 | Syngenta Participations Ag | Modified cry3a toxins and nucleic acid sequences coding therefor |
| WO2003052073A2 (en) | 2001-12-17 | 2003-06-26 | Syngenta Participations Ag | Novel corn event |
| WO2014085490A1 (en) | 2012-11-29 | 2014-06-05 | Chemocentryx, Inc. | Cxcr7 antagonists |
| WO2015185485A1 (en) | 2014-06-06 | 2015-12-10 | Basf Se | Use of substituted oxadiazoles for combating phytopathogenic fungi |
| WO2017093019A1 (en) | 2015-12-03 | 2017-06-08 | Basf Se | Substituted oxadiazoles for combating phytopathogenic fungi |
| WO2018019929A1 (en) | 2016-07-28 | 2018-02-01 | Idorsia Pharmaceuticals Ltd | Piperidine cxcr7 receptor modulators |
| WO2021094434A1 (en) | 2019-11-15 | 2021-05-20 | Bayer Aktiengesellschaft | Substituted hydantoinamides as adamts7 antagonists |
| WO2022253645A1 (en) | 2021-06-01 | 2022-12-08 | Syngenta Crop Protection Ag | Microbiocidal tetrahydroisoquinoline derivatives |
| WO2023110869A1 (en) | 2021-12-15 | 2023-06-22 | Syngenta Crop Protection Ag | Microbiocidal bicycle heterocyclic derivatives |
| WO2024068950A1 (en) * | 2022-09-30 | 2024-04-04 | Syngenta Crop Protection Ag | Microbiocidal pyrazole derivatives |
| WO2024105104A1 (en) * | 2022-11-16 | 2024-05-23 | Syngenta Crop Protection Ag | Microbiocidal tetrahydroisoquinoline derivatives |
| WO2024115546A1 (en) * | 2022-11-30 | 2024-06-06 | Syngenta Crop Protection Ag | Fungicidal compositions |
| WO2024256433A1 (en) * | 2023-06-14 | 2024-12-19 | Syngenta Crop Protection Ag | Fungicidal compositions |
Non-Patent Citations (6)
| Title |
|---|
| "Catalogue of pesticide Formulation types and international coding system", May 2008, CROPLIFE INTERNATIONAL |
| AUSTRAL., J. AGRICULT. RES., vol. 58, 2007, pages 708 |
| MOLLETGRUBEMANN: "Formulation Technology", 2001, WILEY VCH |
| PEST MANAGEM. SCI., vol. 61, 2005, pages 246 |
| SCIENCE, vol. 316, 2007, pages 1 185 |
| WEED SCI., vol. 57, 2009, pages 108 |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202542133A (en) | 2025-11-01 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| AU2020272217B2 (en) | Novel oxadiazole compounds for controlling or preventing phytopathogenic fungi | |
| EP3953340B1 (en) | Novel oxadiazole compounds for controlling or preventing phytopathogenic fungi | |
| JP2021512088A (en) | New oxadiazoles | |
| WO2022058877A1 (en) | Novel picolinamide compounds for combating phytopathogenic fungi | |
| US20210387954A1 (en) | Oxadiazoles as fungicides | |
| WO2022038500A1 (en) | Novel heterocyclic compounds for combating phytopathogenic fungi | |
| WO2025146625A1 (en) | Pyrazole compounds for combating phytopathogenic fungi | |
| WO2022058878A1 (en) | Novel picolinamide compounds for combating phytopathogenic fungi | |
| WO2021090282A1 (en) | Novel oxadiazole compounds containing fused heterocyclyl rings for controlling or preventing phytopathogenic fungi | |
| WO2025146624A1 (en) | Bicyclic heterocyclic amides for combating phytopathogenic fungi | |
| WO2022234470A1 (en) | Novel fused heterocyclic compounds for combating phytopathogenic fungi | |
| US20220151236A1 (en) | Novel oxadiazole compounds for controlling or preventing phytopathogenic fungi | |
| RU2818085C9 (en) | Novel oxadiazole-based compounds for controlling phytopathogenic fungi or preventing damage by them | |
| AU2022414326A1 (en) | Novel substituted fused bicyclic pyridine carboxamide compounds for combating phytopathogenic fungi | |
| RU2818085C2 (en) | Novel oxadiazole-based compounds for controlling phytopathogenic fungi or preventing damage by them | |
| RU2805208C2 (en) | New oxadiazoles | |
| US20220348567A1 (en) | Novel oxadiazole compounds containing 5- membered heteroaromatic ring for controlling or preventing phytopathogenic fungi |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 25703651 Country of ref document: EP Kind code of ref document: A1 |