CN105367605B - Tetradentate platinum(II) complexes and their analogs cyclometallated with functionalized phenylcarbene ligands - Google Patents
Tetradentate platinum(II) complexes and their analogs cyclometallated with functionalized phenylcarbene ligands Download PDFInfo
- Publication number
- CN105367605B CN105367605B CN201510441320.5A CN201510441320A CN105367605B CN 105367605 B CN105367605 B CN 105367605B CN 201510441320 A CN201510441320 A CN 201510441320A CN 105367605 B CN105367605 B CN 105367605B
- Authority
- CN
- China
- Prior art keywords
- alkyl
- term
- complexes
- complex
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F15/00—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table
- C07F15/0006—Compounds containing elements of Groups 8, 9, 10 or 18 of the Periodic Table compounds of the platinum group
- C07F15/0086—Platinum compounds
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F19/00—Metal compounds according to more than one of main groups C07F1/00 - C07F17/00
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/341—Transition metal complexes, e.g. Ru(II)polypyridine complexes
- H10K85/346—Transition metal complexes, e.g. Ru(II)polypyridine complexes comprising platinum
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/361—Polynuclear complexes, i.e. complexes comprising two or more metal centers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1007—Non-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1011—Condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1014—Carbocyclic compounds bridged by heteroatoms, e.g. N, P, Si or B
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1044—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/18—Metal complexes
- C09K2211/185—Metal complexes of the platinum group, i.e. Os, Ir, Pt, Ru, Rh or Pd
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/18—Metal complexes
- C09K2211/188—Metal complexes of other metals not provided for in one of the previous groups
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/10—Triplet emission
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Inorganic Chemistry (AREA)
- Crystallography & Structural Chemistry (AREA)
- Physics & Mathematics (AREA)
- High Energy & Nuclear Physics (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
本发明提供用官能化苯基卡宾配体环金属化的四齿铂(II)络合物和它们的类似物。式I和II的四齿铂络合物适用于显示和照明应用中的磷光或者延迟荧光和磷光发射体。 The present invention provides tetradentate platinum (II) complexes cyclometalated with functionalized phenylcarbene ligands and their analogs. The tetradentate platinum complexes of formula I and II are suitable for use as phosphorescent or delayed fluorescent and phosphorescent emitters in display and lighting applications.
Description
相关申请的交叉引用CROSS-REFERENCE TO RELATED APPLICATIONS
本申请要求2014年7月24日提交的题为“TETRADENTATE PLATINUM(II)COMPLEXESCYCLOMETALATED WITH FUNCTIONALIZED PHENYL CARBENE LIGANDS AND THEIRANALOGUES”的U.S.序列号62/028,562的优先权,将其全部通过参考全部引入本文中。This application claims priority to U.S. Serial No. 62/028,562, entitled "TETRADENTATE PLATINUM(II) COMPLEXESCYCLOMETALATED WITH FUNCTIONALIZED PHENYL CARBENE LIGANDS AND THEIRANALOGUES," filed July 24, 2014, which is incorporated herein by reference in its entirety.
技术领域technical field
本公开内容涉及用于显示和照明应用中的磷光或者延迟荧光和磷光发射体的四齿铂(II)络合物,并具体涉及具有改进的发射光谱的磷光或者延迟荧光和磷光四齿金属络合物。The present disclosure relates to tetradentate platinum(II) complexes for phosphorescent or delayed fluorescence and phosphorescent emitters in display and lighting applications, and in particular to phosphorescent or delayed fluorescence and phosphorescent tetradentate metal complexes with improved emission spectra compound.
背景技术Background technique
能够吸收和/或发射光的化合物可理想地适合用于各种各样的光学和电致发光器件,包括,例如光吸收器件例如太阳能敏感器件和光敏器件、有机发光二极管(OLED)、光发射器件、以及既能够进行光吸收又能够进行光发射和作为用于生物应用的标志器(marker)的器件。许多研究已致力于发现和优化用于在光学和电致发光器件中使用的有机和有机金属材料。通常,该领域中的研究旨在实现许多目标,包括在吸收和发射效率方面的改善和在器件稳定性方面的改善以及在加工能力方面的改善。Compounds capable of absorbing and/or emitting light may be ideally suited for use in a wide variety of optical and electroluminescent devices including, for example, light absorbing devices such as solar and photosensitive devices, organic light emitting diodes (OLEDs), light emitting devices Devices, and devices capable of both light absorption and light emission and as markers for biological applications. Much research has been devoted to the discovery and optimization of organic and organometallic materials for use in optical and electroluminescent devices. In general, research in this area aims to achieve a number of goals, including improvements in absorption and emission efficiencies and improvements in device stability as well as improvements in processability.
尽管在致力于光学和光电材料的研究中取得了显著的进步(例如,红色和绿色磷光有机金属材料可商购和已经在有机发光二极管(OLED)、照明、和高级显示器中用作磷光体),但是许多目前可利用的材料呈现出许多缺点,包括尤其是差的加工能力、效率低的发射或吸收、以及不太理想的稳定性。Despite significant advances in research devoted to optical and optoelectronic materials (eg, red and green phosphorescent organometallic materials are commercially available and have been used as phosphors in organic light-emitting diodes (OLEDs), lighting, and advanced displays) , but many of the currently available materials exhibit a number of drawbacks including, among other things, poor processability, inefficient emission or absorption, and less than ideal stability.
好的蓝光发射体特别稀少,其中一个挑战是蓝光器件的稳定性。主体材料的选择对于器件的稳定性和效率具有影响。与红色和绿色磷光体的最低三重激发态能量相比,蓝色磷光体的最低三重激发态能量非常高,这意味着,蓝光器件的主体材料的最低三重激发态能量应该甚至更高。因此,问题之一是,用于蓝光器件的主体材料有限。因此,需要新的材料,其在光学发射和吸收应用中呈现出改善的性能。Good blue light emitters are particularly rare, and one of the challenges is the stability of blue light devices. The choice of host material has an impact on the stability and efficiency of the device. The lowest triplet energy of the blue phosphor is very high compared to that of the red and green phosphors, which means that the lowest triplet energy of the host material for blue devices should be even higher. Therefore, one of the problems is that there are limited host materials for blue light devices. Therefore, there is a need for new materials that exhibit improved performance in optical emission and absorption applications.
发明内容SUMMARY OF THE INVENTION
设计和合成了一系列用官能化苯基卡宾配体环金属化的(cyclometalated)四齿铂(II)络合物和它们的类似物(用官能化苯基卡宾配体和它们的类似物环金属化的四齿铂(II)络合物)。这些络合物提供改善的色纯度、增强的操作稳定性、以及减少的或者消除的潜在的强的分子间相互作用,并适用于发光标记物(lable)、用于有机发光二极管(OLED)和照明应用的发射体、和光子降频变换器。designed and synthesized a series of cyclometalated tetradentate platinum(II) complexes with functionalized phenylcarbene ligands and their analogs (cyclized with functionalized phenylcarbene ligands and their analogs) Metallized tetradentate platinum(II) complex). These complexes offer improved color purity, enhanced operational stability, and reduced or eliminated potentially strong intermolecular interactions, and are suitable for use in luminescent labels, for use in organic light-emitting diodes (OLEDs) and Emitters for lighting applications, and photon downconverters.
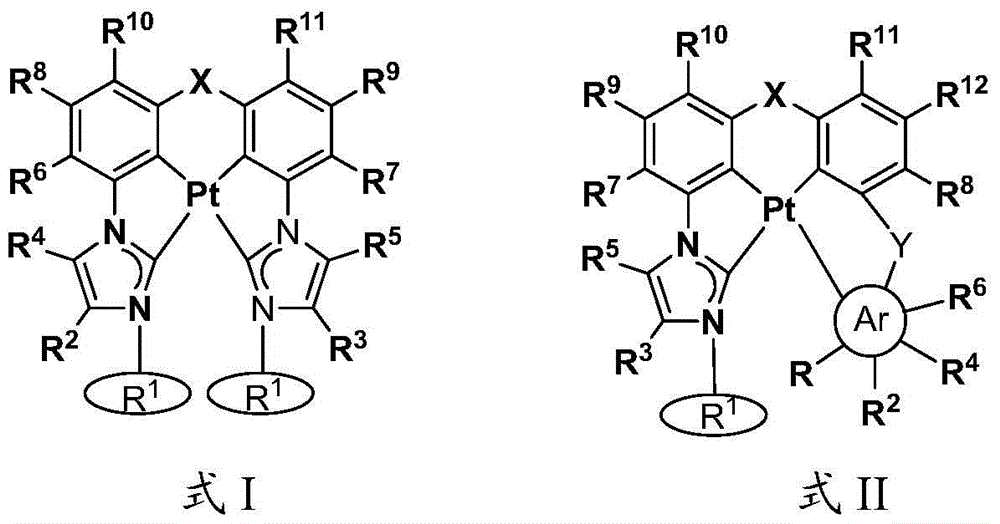
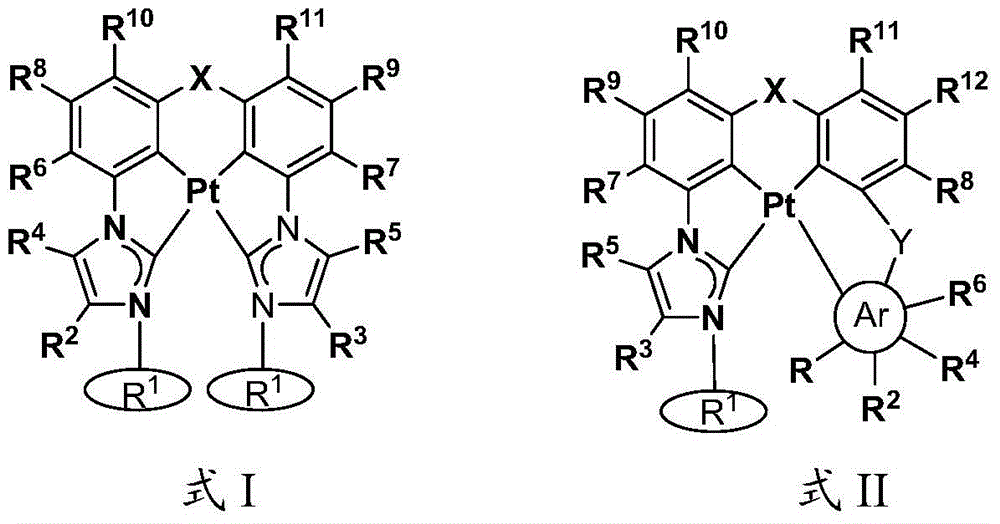
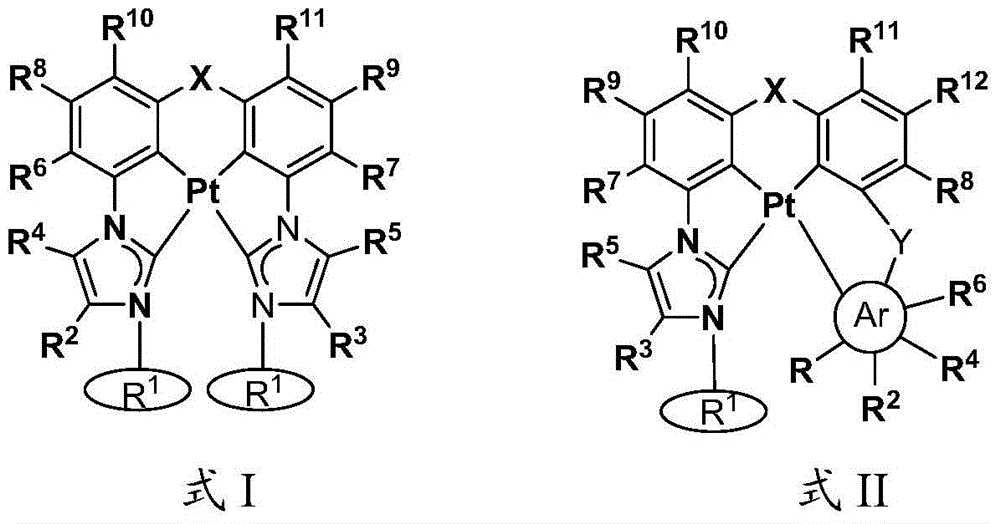
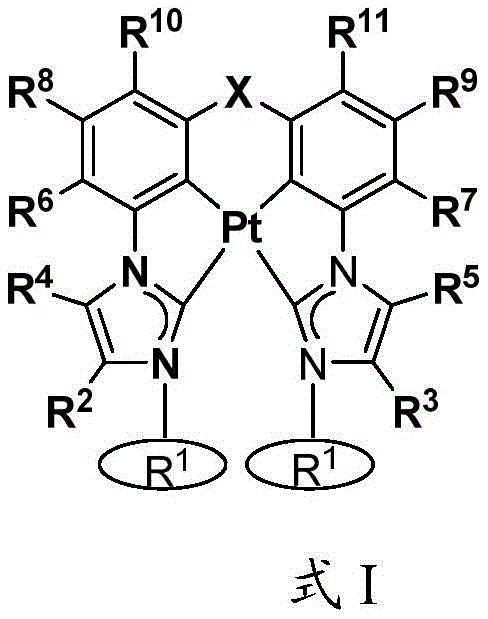
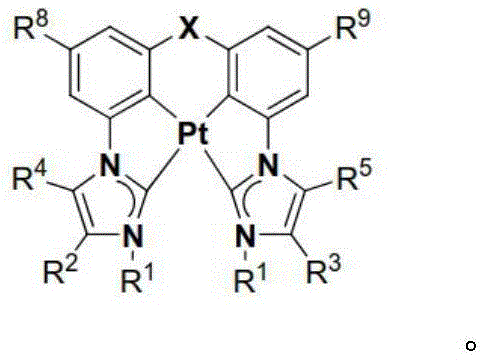
本文中公开了式I和式II的络合物:Disclosed herein are complexes of Formula I and Formula II:
其中:in:
Ar为五元杂芳基、五元卡宾、五元N-杂环卡宾、六元芳基、或者六元杂芳基,Ar is five-membered heteroaryl, five-membered carbene, five-membered N-heterocyclic carbene, six-membered aryl, or six-membered heteroaryl,
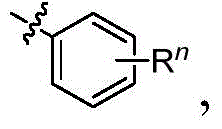
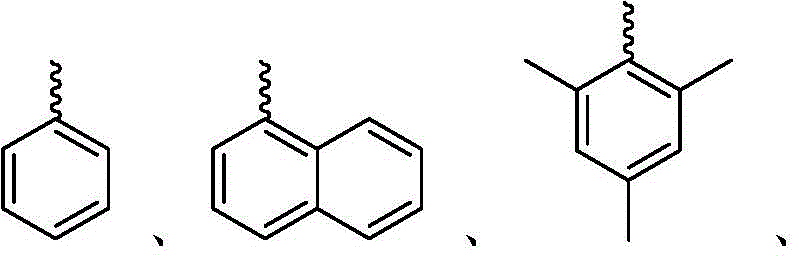
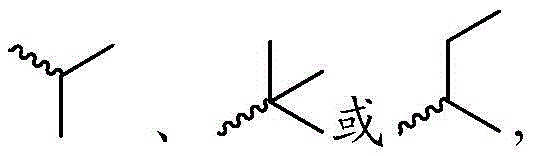
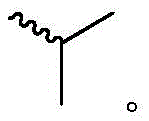
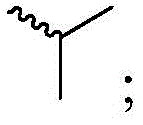
每个R1独立地为 或者 Each R1 is independently or
R、R2、R3、R4、R5、R6、R7、R8、R9、R10、R11、和R12中的每个独立地为氢、卤素、羟基、硝基、硫醇基;或者取代的或者未取代的:C1-C4烷基、烷氧基、芳基、或者氨基,其中当Ar为五元环时R不存在,Each of R, R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , and R 12 is independently hydrogen, halogen, hydroxy, nitro , thiol; or substituted or unsubstituted: C 1 -C 4 alkyl, alkoxy, aryl, or amino, wherein R is absent when Ar is a five-membered ring,
X为O、S、S=O、O=S=O、Se、Se=O、O=Se=O、NR2a、PR2b、AsR2c、CR2dR2e、SiR2fR2g、或者BR2h,X is O, S, S=O, O=S=O, Se, Se=O, O=Se=O, NR 2a , PR 2b , AsR 2c , CR 2d R 2e , SiR 2f R 2g , or BR 2h ,
R2a、R2b、R2c、R2d、R2e、R2f、R2g、和R2h中的每个独立地为取代的或者未取代的C1-C4烷基或者芳基,Each of R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently a substituted or unsubstituted C 1 -C 4 alkyl or aryl,
Y存在或者不存在,以及如果存在,Y为O、S、S=O、O=S=O、Se、Se=O、O=Se=O、NR3a、PR3b、AsR3c、CR3dR3e、SiR3fR3g、或者BR3h、和Y is present or absent, and if present, Y is O, S, S=O, O=S=O, Se, Se=O, O=Se=O, NR 3a , PR 3b , AsR 3c , CR 3d R 3e , SiR 3f R 3g , or BR 3h , and
R3a、R3b、R3c、R3d、R3e、R3f、R3g、和R3h中的每个独立地为取代的或者未取代的C1-C4烷基或者芳基。Each of R 3a , R 3b , R 3c , R 3d , R 3e , R 3f , R 3g , and R 3h is independently substituted or unsubstituted C 1 -C 4 alkyl or aryl.
在一些情况中,Ar为吡唑、咪唑、唑、噻唑、吡啶等。在一些情况中,在相同环或者相邻环上的R、R2、R3、R4、R5、R6、R7、R8、R9、R10、R11、和R12中的任意两个结合(键合)在一起以形成稠环体系。例如,R和R2、R2和R4、或者R4和R6可结合从而与Ar形成稠环体系,例如苯并咪唑、苯并唑、苯并噻唑、吲唑、喹啉、异喹啉、咪唑并[1,5-a]吡啶等。In some cases, Ar is pyrazole, imidazole, azoles, thiazoles, pyridines, etc. In some cases, in R, R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , and R 12 on the same ring or on adjacent rings Any two of are joined (bonded) together to form a fused ring system. For example, R and R 2 , R 2 and R 4 , or R 4 and R 6 can combine to form a fused ring system with Ar, such as benzimidazole, benzo azole, benzothiazole, indazole, quinoline, isoquinoline, imidazo[1,5-a]pyridine, etc.
本文中还公开了包含一种或者多种本文中公开的络合物的组合物、以及包含本文中公开的一种或者多种化合物或者组合物的器件例如OLED。Also disclosed herein are compositions comprising one or more complexes disclosed herein, as well as devices such as OLEDs comprising one or more compounds or compositions disclosed herein.
因此,已经描述了具体实施方案。所述实施方案和其它实施方案的变型、修饰、和改进可基于所描述和说明的内容作出。另外,一个或者多个实施方案的一个或者多个特征可组合。一个或者多个实施以及各种特征和方面的细节在下面的附图、说明书、和权利要求中阐述。Accordingly, specific embodiments have been described. Variations, modifications, and improvements to the described and other embodiments can be made based on what has been described and illustrated. Additionally, one or more features of one or more embodiments may be combined. The details of one or more implementations and various features and aspects are set forth in the accompanying drawings, the description, and the claims below.
附图说明Description of drawings
图1描绘了示例性有机发光器件(OLED)的横截面图。1 depicts a cross-sectional view of an exemplary organic light emitting device (OLED).
图2显示了Pt7O7-dipr在室温和77K的光致发光光谱。Figure 2 shows the photoluminescence spectra of Pt7O7-dipr at room temperature and 77K.
具体实施方式Detailed ways
通过参考下面的详细描述和其中包括的实例,可以更容易地理解本公开内容。The present disclosure may be understood more readily by reference to the following detailed description and the examples included therein.
在公开和描述本络合物、器件、和/或方法之前,将理解,它们不限于具体的合成方法,除非另外说明,或者它们不限于特定的试剂,除非另外说明,因为这些当然可以改变。还将理解,本文中所使用的术语仅为了描述具体方面的目的,且不意图为限制性的。尽管与本文中所描述的那些方法和材料类似或等同的任何方法和材料可用于实践或试验中,但现在描述实例方法和材料。Before the present complexes, devices, and/or methods are disclosed and described, it is to be understood that they are not limited to specific synthetic methods unless otherwise specified, or that they are not limited to specific reagents unless otherwise specified, as these may of course vary. It will also be understood that the terminology used herein is for the purpose of describing particular aspects only and is not intended to be limiting. Although any methods and materials similar or equivalent to those described herein can be used in the practice or testing, example methods and materials are now described.
如本说明书和所附权利要求书中所使用的,单数形式“一个(种)(a,an)”和“该(所述)”包括复数指示物(referent),除非上下文清楚地另外说明。因此,例如,对“一种组分(组分,a component)”的提及包括两种或更多种组分的混合物。As used in this specification and the appended claims, the singular forms "a (a, an)" and "the (the)" include plural referents unless the context clearly dictates otherwise. Thus, for example, reference to "a component" includes a mixture of two or more components.
如本文中所使用的,术语“任选的”或“任选地”表示随后描述的事件或情况可发生或可不发生,以及所述描述包括其中所述事件或情况发生的情形和其中所述事件或情况不发生的情形。As used herein, the terms "optional" or "optionally" mean that the subsequently described event or circumstance may or may not occur, and that the description includes instances in which said event or circumstance occurs and instances in which said event or circumstance occurs Circumstances in which an event or circumstance does not occur.
公开将用于制备本文中所述的组合物的组分以及将在本文中公开的方法中使用的组合物本身。本文中公开这些和其它材料,并且理解,当这些材料的组合、子集、相互作用、集合等被公开时,虽然这些化合物的各种单独的和共同的组合和排列(permutation)各自的具体指示物不能被明确地公开,但每一种在本文中被具体地设计和描述。例如,如果具体化合物被公开和讨论并且对包括所述化合物的许多分子可进行的许多变化被讨论,则可能的变化以及化合物的每种组合和排列被具体地设计,除非具体地相反说明。因此,如果公开了一类分子A、B和C以及一类分子D、E和F,并且公开了组合分子A-D的实例,则即使每个没有被单独地列举,每个也被单独地和共同地被设计,意味着组合A-E、A-F、B-D、B-E、B-F、C-D、C-E和C-F被公开。同样地,这些的任意子集或组合也被公开。因此,例如,A-E、B-F和C-E的子集将被认为被公开。该概念适用于本申请的所有方面,包括,但不限于,制造和使用组合物的方法中的步骤。因此,如果存在可以被执行的多个额外的步骤,则理解,这些额外的步骤各自可用所述方法的任何具体实施方式或实施方式的组合执行。The components to be used in the preparation of the compositions described herein as well as the compositions themselves to be used in the methods disclosed herein are disclosed. These and other materials are disclosed herein, and it is understood that when combinations, subsets, interactions, collections, etc. of these materials are disclosed, notwithstanding the specific indication of each individual and collective combination and permutation of these compounds The items are not explicitly disclosed, but each is specifically designed and described herein. For example, if a particular compound is disclosed and discussed and the many variations that can be made to many molecules comprising the compound are discussed, the possible variations and every combination and permutation of the compounds are specifically designed unless specifically stated to the contrary. Thus, if a class of molecules A, B, and C and a class of molecules D, E, and F are disclosed, and examples of combining molecules A-D are disclosed, each is individually and collectively even if not individually recited is designed, meaning that the combinations A-E, A-F, B-D, B-E, B-F, C-D, C-E and C-F are disclosed. Likewise, any subset or combination of these are also disclosed. Thus, for example, a subset of A-E, B-F, and C-E would be considered disclosed. This concept applies to all aspects of this application, including, but not limited to, steps in methods of making and using compositions. Thus, if there are multiple additional steps that can be performed, it is understood that each of these additional steps can be performed with any specific embodiment or combination of embodiments of the described method.
如本文中提及的,连接原子或者基团可以连接两个原子,例如N原子和C原子。连接原子或者基团在本文中在一个方面中作为X、Y、Y1、Y2和/或Z公开。如果化合价允许的话,连接原子可以任选地具有连接的其它化学部分。例如,在一个方面中,一旦氧键合到两个基团(例如,N和/或C基团),由于化合价被满足,氧将不具有连接的任何其它化学基团。在另一方面中,当碳是连接原子时,两个额外的化学部分可以连接到碳上。合适的化学部分包括胺、酰胺、硫醇、芳基、杂芳基、环烷基、和杂环基部分。As referred to herein, a linking atom or group may link two atoms, eg, a N atom and a C atom. Linking atoms or groups are disclosed herein as X, Y, Y 1 , Y 2 and/or Z in one aspect. The linking atom may optionally have other chemical moieties attached if valence permits. For example, in one aspect, once an oxygen is bonded to two groups (eg, N and/or C groups), the oxygen will not have any other chemical groups attached since the valences are satisfied. In another aspect, when carbon is the linking atom, two additional chemical moieties can be attached to the carbon. Suitable chemical moieties include amine, amide, thiol, aryl, heteroaryl, cycloalkyl, and heterocyclyl moieties.
本文中使用的术语“环状结构”或者类似术语是指任何环状化学结构,其包括,但不限于,芳基、杂芳基、环烷基、环烯基、杂环基、卡宾、和N-杂环卡宾。The term "cyclic structure" or similar terms as used herein refers to any cyclic chemical structure including, but not limited to, aryl, heteroaryl, cycloalkyl, cycloalkenyl, heterocyclyl, carbene, and N-heterocyclic carbene.
如本文中所使用的,术语“取代的”意图包括有机化合物的所有可允许的取代基。在宽的方面中,可允许的取代基包括有机化合物的无环和环状的、支化和非支化的、碳环和杂环的、以及芳族和非芳族的取代基。说明性的取代基包括,例如,下面描述的那些。对于合适的有机化合物,可允许的取代基可以是一个或多个以及相同或不同的。对于本公开内容来说,杂原子,例如氮,可以具有氢取代基和/或满足杂原子的化合价的本文中所描述的有机化合物的任何可允许的取代基。本公开内容不意图被有机化合物的可允许的取代基以任何方式限制。此外,术语“取代”或“被……取代”包括暗示性条件:这样的取代是根据被取代的原子和取代基的允许的化合价,以及该取代产生稳定的化合物,例如不自发地经历转变(例如通过重排、环化、消除等)的化合物。还设计,在一些方面中,除非明确相反说明,单独的取代基可为进一步任选地被取代的(即,进一步取代的或未取代的)。As used herein, the term "substituted" is intended to include all permissible substituents of organic compounds. In broad aspects, permissible substituents include acyclic and cyclic, branched and unbranched, carbocyclic and heterocyclic, and aromatic and non-aromatic substituents of organic compounds. Illustrative substituents include, for example, those described below. The permissible substituents may be one or more and the same or different for suitable organic compounds. For purposes of this disclosure, a heteroatom, such as nitrogen, may have hydrogen substituents and/or any permissible substituents of the organic compounds described herein that satisfy the valences of the heteroatoms. This disclosure is not intended to be limited in any way by the permissible substituents of organic compounds. Furthermore, the terms "substituted" or "substituted by" include the implied condition that such substitution is based on the permissible valences of the substituted atoms and substituents, and that the substitution results in a stable compound, eg, that does not spontaneously undergo a transformation ( Compounds such as by rearrangement, cyclization, elimination, etc.). It is also contemplated that, in some aspects, unless expressly stated to the contrary, individual substituents may be further optionally substituted (ie, further substituted or unsubstituted).
在定义各种术语时,“X”和“Y”在本文中用作一般性符号来表示各种具体的取代基。这些符号可以是任何取代基,不限于本文中公开的那些,并且,当它们在一种情形下被定义为一些取代基时,它们在另一情形下可以被定义为一些其它的取代基。In defining various terms, "X" and "Y" are used herein as general symbols to refer to various specific substituents. These symbols can be any substituents, not limited to those disclosed herein, and when they are defined as some substituents in one context, they can be defined as some other substituents in another context.
如本文中所使用的术语“烷基”是1-24个碳原子的支化或未支化的饱和烃基,例如甲基、乙基、正丙基、异丙基、正丁基、异丁基、仲丁基、叔丁基、正戊基、异戊基、仲戊基、新戊基、己基、庚基、辛基、壬基、癸基、十二烷基、十四烷基、十六烷基、二十烷基、二十四烷基等。烷基可以是环状的或无环的。烷基可以是支化或未支化的。烷基还可以是取代或未取代的。例如,烷基可以被一个或多个基团取代,所述基团包括但不限于,如本文中所描述的烷基、环烷基、烷氧基、氨基、醚基、卤素(卤化物,halide)、羟基、硝基、甲硅烷基、硫-氧代(sulfo-oxo)、或硫醇基。“低级烷基”基团是含有一至六个(例如一至四个)碳原子的烷基。The term "alkyl" as used herein is a branched or unbranched saturated hydrocarbon group of 1-24 carbon atoms, such as methyl, ethyl, n-propyl, isopropyl, n-butyl, isobutyl base, sec-butyl, tert-butyl, n-pentyl, isopentyl, sec-pentyl, neopentyl, hexyl, heptyl, octyl, nonyl, decyl, dodecyl, tetradecyl, hexadecyl, eicosyl, tetracosyl, etc. Alkyl groups can be cyclic or acyclic. Alkyl groups can be branched or unbranched. Alkyl groups can also be substituted or unsubstituted. For example, an alkyl group can be substituted with one or more groups including, but not limited to, alkyl, cycloalkyl, alkoxy, amino, ether, halogen (halide, halide), hydroxyl, nitro, silyl, sulfo-oxo, or thiol. A "lower alkyl" group is an alkyl group containing from one to six (eg, one to four) carbon atoms.
在整个说明书中,“烷基”被概括性地用来指未取代的烷基和取代的烷基两者;然而,取代的烷基通过确定烷基上的具体取代基也在本文中被具体地提到。例如,术语“卤化烷基”或“卤代烷基”具体地指被一种或多种卤素例如氟、氯、溴、或碘取代的烷基。术语“烷氧基烷基”具体地指被如下所述的一个或多个烷氧基取代的烷基。术语“烷基氨基”具体地指被如下所述的一个或多个氨基等取代的烷基。当在一种情形下使用“烷基”并且在另一种情形下使用具体的术语例如“烷基醇”时,其不意图暗示术语“烷基”并非也是指具体术语例如“烷基醇”等。Throughout the specification, "alkyl" is used broadly to refer to both unsubstituted and substituted alkyl groups; however, substituted alkyl groups are also specified herein by identifying the specific substituent on the alkyl group mentioned. For example, the term "haloalkyl" or "haloalkyl" specifically refers to an alkyl group substituted with one or more halogens such as fluorine, chlorine, bromine, or iodine. The term "alkoxyalkyl" specifically refers to an alkyl group substituted with one or more alkoxy groups as described below. The term "alkylamino" specifically refers to an alkyl group substituted with one or more amino groups, etc., as described below. When "alkyl" is used in one context and a specific term such as "alkyl alcohol" is used in another context, it is not intended to imply that the term "alkyl" does not also refer to a specific term such as "alkyl alcohol" Wait.
该实践还可以用于本文中描述的其它基团。即,尽管术语例如“环烷基”是指未取代的和取代的环烷基部分两者,但取代的部分可以另外在本文中具体地确定;例如,具体的取代的环烷基可以被称为例如“烷基环烷基”。类似地,取代的烷氧基可以被具体称为例如“卤化烷氧基”,具体的取代的烯基可以是例如“烯基醇”等。再次,使用概括性术语例如“环烷基”以及具体术语例如“烷基环烷基”的实践不意图暗示所述概括性术语并非也包括所述具体术语。This practice can also be used for other groups described herein. That is, although terms such as "cycloalkyl" refer to both unsubstituted and substituted cycloalkyl moieties, substituted moieties may otherwise be specifically identified herein; for example, a particular substituted cycloalkyl may be referred to as For example "alkylcycloalkyl". Similarly, substituted alkoxy groups can be specifically referred to as, for example, "halogenated alkoxy groups," and specific substituted alkenyl groups can be, for example, "alkenyl alcohols," and the like. Again, the practice of using general terms such as "cycloalkyl" as well as specific terms such as "alkylcycloalkyl" is not intended to imply that the general term does not also include the specific term.
如本文中所用的术语“环烷基”是由至少三个碳原子构成的非芳族的基于碳的环。环烷基的实例包括,但不限于,环丙基、环丁基、环戊基、环己基、降冰片基等。术语“杂环烷基”是一类如上定义的环烷基,并且被包括在术语“环烷基”的含义范围内,其中环的碳原子的至少一个被杂原子替代,所述杂原子例如,但不限于氮、氧、硫、或磷。环烷基和杂环烷基可以是取代或未取代的。环烷基和杂环烷基可以被一个或多个基团取代,所述基团包括,但不限于如本文中描述的烷基、环烷基、烷氧基、氨基、醚基、卤素、羟基、硝基、甲硅烷基、硫-氧代、或硫醇基。The term "cycloalkyl" as used herein is a non-aromatic carbon-based ring composed of at least three carbon atoms. Examples of cycloalkyl groups include, but are not limited to, cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, norbornyl, and the like. The term "heterocycloalkyl" is a class of cycloalkyl groups as defined above, and is included within the meaning of the term "cycloalkyl" wherein at least one of the carbon atoms of the ring is replaced by a heteroatom such as , but not limited to nitrogen, oxygen, sulfur, or phosphorus. Cycloalkyl and heterocycloalkyl can be substituted or unsubstituted. Cycloalkyl and heterocycloalkyl groups may be substituted with one or more groups including, but not limited to, alkyl, cycloalkyl, alkoxy, amino, ether, halogen, Hydroxyl, nitro, silyl, thio-oxo, or thiol.
如本文中所使用的术语“聚烯烃基团”是具有两个或更多个彼此连接的CH2基团的基团。聚烯烃基团可由式-(CH2)a-表示,其中“a”是2-500的整数。The term "polyolefin group" as used herein is a group having two or more CH2 groups attached to each other. The polyolefin group can be represented by the formula -( CH2 ) a- , wherein "a" is an integer from 2-500.
如本文中所使用的术语“烷氧”和“烷氧基”是指通过醚键键合的烷基或环烷基;即,“烷氧”基团可以定义为-OA1,其中A1是如上定义的烷基或环烷基。“烷氧”还包括如刚才所描述的烷氧基团的聚合物;即,烷氧可以是聚醚,例如-OA1-OA2或-OA1-(OA2)a-OA3,其中“a”是1-200的整数并且A1、A2、和A3是烷基和/或环烷基。The terms "alkoxy" and "alkoxy," as used herein, refer to an alkyl or cycloalkyl group bonded through an ether linkage; ie, an "alkoxy" group can be defined as -OA 1 , where A 1 is alkyl or cycloalkyl as defined above. "Alkoxy" also includes polymers of alkoxy groups as just described; that is, the alkoxy may be a polyether, such as -OA 1 -OA 2 or -OA 1 -(OA 2 ) a -OA 3 , wherein "a" is an integer from 1 to 200 and A 1 , A 2 , and A 3 are alkyl and/or cycloalkyl groups.
如本文中所用的“烯基”是结构式含有至少一个碳-碳双键的2-24个碳原子的烃基。不对称结构例如(A1A2)C=C(A3A4)意图包括E和Z异构体两者。这可以从其中存在不对称烯烃的本文中的结构式进行推测,或者其可以通过键符号C=C明确地指示。烯基可以被一个或多个基团取代,所述基团包括,但不限于如本文中所述的烷基、环烷基、烷氧基、烯基、环烯基、炔基、环炔基、芳基、杂芳基、醛基、氨基、羧酸基、酯基、醚基、卤素、羟基、酮基、叠氮基、硝基、甲硅烷基、硫-氧代、或硫醇基。As used herein, "alkenyl" is a hydrocarbyl group of 2-24 carbon atoms having a structural formula containing at least one carbon-carbon double bond. Asymmetric structures such as (A 1 A 2 )C=C(A 3 A 4 ) are intended to include both E and Z isomers. This can be inferred from the structural formulas herein in which the asymmetric olefin is present, or it can be explicitly indicated by the bond symbol C=C. Alkenyl groups may be substituted with one or more groups including, but not limited to, alkyl, cycloalkyl, alkoxy, alkenyl, cycloalkenyl, alkynyl, cycloalkyne as described herein group, aryl, heteroaryl, aldehyde, amino, carboxylate, ester, ether, halogen, hydroxyl, keto, azido, nitro, silyl, thio-oxo, or thiol base.
如本文中所用的术语“环烯基”是由至少三个碳原子构成并且含有至少一个碳-碳双键即C=C的非芳族的基于碳的环。环烯基的实例包括,但不限于,环丙烯基、环丁烯基、环戊烯基、环戊二烯基、环己烯基、环己二烯基、降冰片烯基等。术语“杂环烯基”是一类如上定义的环烯基,并且被包括在术语“环烯基”的含义范围内,其中环的碳原子的至少一个被杂原子替代,所述杂原子例如,但不限于氮、氧、硫或磷。环烯基和杂环烯基可以是取代或未取代的。环烯基和杂环烯基可以被一个或多个基团取代,所述基团包括,但不限于如本文中描述的烷基、环烷基、烷氧基、烯基、环烯基、炔基、环炔基、芳基、杂芳基、醛基、氨基、羧酸基、酯基、醚基、卤素、羟基、酮基、叠氮基、硝基、甲硅烷基、硫-氧代、或硫醇基。The term "cycloalkenyl" as used herein is a non-aromatic carbon-based ring consisting of at least three carbon atoms and containing at least one carbon-carbon double bond, ie, C=C. Examples of cycloalkenyl groups include, but are not limited to, cyclopropenyl, cyclobutenyl, cyclopentenyl, cyclopentadienyl, cyclohexenyl, cyclohexadienyl, norbornenyl, and the like. The term "heterocycloalkenyl" is a class of cycloalkenyl groups as defined above and is included within the meaning of the term "cycloalkenyl" wherein at least one of the carbon atoms of the ring is replaced by a heteroatom such as , but not limited to nitrogen, oxygen, sulfur or phosphorus. Cycloalkenyl and heterocycloalkenyl groups can be substituted or unsubstituted. Cycloalkenyl and heterocycloalkenyl groups may be substituted with one or more groups including, but not limited to, alkyl, cycloalkyl, alkoxy, alkenyl, cycloalkenyl, Alkynyl, Cycloalkynyl, Aryl, Heteroaryl, Aldehyde, Amino, Carboxylic, Ester, Ether, Halogen, Hydroxy, Keto, Azido, Nitro, Silyl, Sulfur-Oxygen Generation, or thiol group.
如本文中所用的术语“炔基”是结构式含有至少一个碳-碳三键的2-24个碳原子的烃基。炔基可以是未取代的或者被一个或多个基团取代的,所述基团包括,但不限于如本文中描述的烷基、环烷基、烷氧基、烯基、环烯基、炔基、环炔基、芳基、杂芳基、醛基、氨基、羧酸基、酯基、醚基、卤素、羟基、酮基、叠氮基、硝基、甲硅烷基、硫-氧代、或硫醇基。The term "alkynyl" as used herein is a hydrocarbyl group of 2-24 carbon atoms having a structural formula containing at least one carbon-carbon triple bond. Alkynyl groups can be unsubstituted or substituted with one or more groups including, but not limited to, alkyl, cycloalkyl, alkoxy, alkenyl, cycloalkenyl, Alkynyl, Cycloalkynyl, Aryl, Heteroaryl, Aldehyde, Amino, Carboxylic, Ester, Ether, Halogen, Hydroxy, Keto, Azido, Nitro, Silyl, Sulfur-Oxygen Generation, or thiol group.
如本文中所用的术语“环炔基”是由至少七个碳原子构成并且含有至少一个碳-碳三键的非芳族的基于碳的环。环炔基的实例包括,但不限于,环庚炔基、环辛炔基、环壬炔基等。术语“杂环炔基”是一类如上定义的环炔基,并且被包括在术语“环炔基”的含义范围内,其中环的碳原子的至少一个被杂原子替代,所述杂原子例如,但不限于氮、氧、硫、或磷。环炔基和杂环炔基可以是取代的或未取代的。环炔基和杂环炔基可以被一个或多个基团取代,所述基团包括,但不限于如本文中描述的烷基、环烷基、烷氧基、烯基、环烯基、炔基、环炔基、芳基、杂芳基、醛基、氨基、羧酸基、酯基、醚基、卤素、羟基、酮基、叠氮基、硝基、甲硅烷基、硫-氧代、或硫醇基。The term "cycloalkynyl" as used herein is a non-aromatic carbon-based ring consisting of at least seven carbon atoms and containing at least one carbon-carbon triple bond. Examples of cycloalkynyl groups include, but are not limited to, cycloheptynyl, cyclooctynyl, cyclononynyl, and the like. The term "heterocycloalkynyl" is a class of cycloalkynyl groups as defined above and is included within the meaning of the term "cycloalkynyl" wherein at least one of the carbon atoms of the ring is replaced by a heteroatom such as , but not limited to nitrogen, oxygen, sulfur, or phosphorus. Cycloalkynyl and heterocycloalkynyl groups can be substituted or unsubstituted. Cycloalkynyl and heterocycloalkynyl groups may be substituted with one or more groups including, but not limited to, alkyl, cycloalkyl, alkoxy, alkenyl, cycloalkenyl, Alkynyl, Cycloalkynyl, Aryl, Heteroaryl, Aldehyde, Amino, Carboxylic, Ester, Ether, Halogen, Hydroxy, Keto, Azido, Nitro, Silyl, Sulfur-Oxygen Generation, or thiol group.
如本文中所用的术语“芳基”是含有任何基于碳的芳族基团的基团,包括,但不限于,苯、萘、苯基、联苯基、苯氧基苯等。术语“芳基”还包括“杂芳基”,其被定义为含有如下芳族基团的基团:所述芳族基团具有至少一个引入到所述芳族基团的环中的杂原子。杂原子的实例包括,但不限于,氮、氧、硫、和磷。同样地,术语“非杂芳基”也包括在术语“芳基”的范围内,其定义包含不含杂原子的芳族基团的基团。芳基可以是取代的或未取代的。芳基可以被一个或多个基团取代,所述基团包括,但不限于如本文中描述的烷基、环烷基、烷氧基、烯基、环烯基、炔基、环炔基、芳基、杂芳基、醛基、氨基、羧酸基、酯基、醚基、卤素、羟基、酮基、叠氮基、硝基、甲硅烷基、硫-氧代、或硫醇基。术语“联芳基(biaryl)”是一种特别类型的芳基并且被包括在术语“芳基”的定义范围内。联芳基是指经由稠环结构结合在一起(如在萘中一样)、或者经由一个或多个碳-碳键连接(如在联苯中一样)的两个芳基。The term "aryl" as used herein is a group containing any carbon-based aromatic group including, but not limited to, benzene, naphthalene, phenyl, biphenyl, phenoxybenzene, and the like. The term "aryl" also includes "heteroaryl", which is defined as a group containing an aromatic group having at least one heteroatom introduced into the ring of the aromatic group . Examples of heteroatoms include, but are not limited to, nitrogen, oxygen, sulfur, and phosphorus. Likewise, the term "non-heteroaryl" is also included within the scope of the term "aryl," which defines a group that includes an aromatic group that does not contain a heteroatom. Aryl groups can be substituted or unsubstituted. Aryl groups may be substituted with one or more groups including, but not limited to, alkyl, cycloalkyl, alkoxy, alkenyl, cycloalkenyl, alkynyl, cycloalkynyl as described herein , aryl, heteroaryl, aldehyde, amino, carboxylate, ester, ether, halogen, hydroxyl, ketone, azide, nitro, silyl, thio-oxo, or thiol . The term "biaryl" is a special type of aryl group and is included within the definition of the term "aryl". Biaryl refers to two aryl groups joined together via a fused ring structure (as in naphthalene), or via one or more carbon-carbon bonds (as in biphenyl).
如本文中所用的术语“醛基”由式-C(O)H表示。在整个本说明书中,“C(O)”是羰基即C=O的简化符号。The term "aldehyde group" as used herein is represented by the formula -C(O)H. Throughout this specification, "C(O)" is a shorthand notation for carbonyl, ie, C=O.
如本文中所用的术语“胺基”或“氨基”由式-NA1A2表示,其中A1和A2可以独立地为氢或如本文中描述的烷基、环烷基、烯基、环烯基、炔基、环炔基、芳基或杂芳基。 The term "amino" or "amino" as used herein is represented by the formula -NA1A2 , wherein A1 and A2 may independently be hydrogen or alkyl, cycloalkyl, alkenyl, Cycloalkenyl, alkynyl, cycloalkynyl, aryl or heteroaryl.
如本文中所用的术语“烷基氨基”由式-NH(-烷基)表示,其中烷基如本文中所述。代表性的实例包括,但不限于,甲基氨基、乙基氨基、丙基氨基、异丙基氨基、丁基氨基、异丁基氨基、(仲丁基)氨基、(叔丁基)氨基、戊基氨基、异戊基氨基、(叔戊基)氨基、己基氨基等。The term "alkylamino" as used herein is represented by the formula -NH(-alkyl), wherein alkyl is as described herein. Representative examples include, but are not limited to, methylamino, ethylamino, propylamino, isopropylamino, butylamino, isobutylamino, (sec-butyl)amino, (tert-butyl)amino, Amylamino, isopentylamino, (tert-amyl)amino, hexylamino and the like.
如本文中所用的术语“二烷基氨基”由式-N(-烷基)2表示,其中烷基如本文中所述。代表性的实例包括,但不限于,二甲基氨基、二乙基氨基、二丙基氨基、二异丙基氨基、二丁基氨基、二异丁基氨基、二(仲丁基)氨基、二(叔丁基)氨基、二戊基氨基、二异戊基氨基、二(叔戊基)氨基、二己基氨基、N-乙基-N-甲基氨基、N-甲基-N-丙基氨基、N-乙基-N-丙基氨基等。The term "dialkylamino" as used herein is represented by the formula -N(-alkyl) 2 , wherein alkyl is as described herein. Representative examples include, but are not limited to, dimethylamino, diethylamino, dipropylamino, diisopropylamino, dibutylamino, diisobutylamino, di(sec-butyl)amino, Di(tert-butyl)amino, dipentylamino, diisoamylamino, di(tert-amyl)amino, dihexylamino, N-ethyl-N-methylamino, N-methyl-N-propane amino, N-ethyl-N-propylamino, etc.
如本文中所用的术语“羧酸基”由式-C(O)OH表示。The term "carboxylate" as used herein is represented by the formula -C(O)OH.
如本文中所用的术语“酯基”由式-OC(O)A1或-C(O)OA1表示,其中A1可为如本文中描述的烷基、环烷基、烯基、环烯基、炔基、环炔基、芳基或杂芳基。如本文中所用的术语“聚酯基”由式-(A1O(O)C-A2-C(O)O)a-或-(A1O(O)C-A2-OC(O))a-表示,其中A1和A2可以独立地为本文中描述的烷基、环烷基、烯基、环烯基、炔基、环炔基、芳基或杂芳基并且“a”是1-500的整数。“聚酯基”作为用来描述通过具有至少两个羧酸基团的化合物与具有至少两个羟基的化合物之间的反应产生的基团的术语。The term "ester group" as used herein is represented by the formula -OC(O)A 1 or -C(O)OA 1 , wherein A 1 can be an alkyl, cycloalkyl, alkenyl, cyclo, as described herein Alkenyl, alkynyl, cycloalkynyl, aryl or heteroaryl. The term "polyester-based" as used herein is represented by the formula -(A 1 O(O)CA 2 -C(O)O) a - or -(A 1 O(O)CA 2 -OC(O)) a - means where A and A can independently be alkyl, cycloalkyl, alkenyl, cycloalkenyl, alkynyl, cycloalkynyl, aryl or heteroaryl as described herein and "a" is 1 -500 integer. "Polyester group" is a term used to describe a group produced by a reaction between a compound having at least two carboxylic acid groups and a compound having at least two hydroxyl groups.
如本文中所用的术语“醚”由式A1OA2表示,其中A1和A2可以独立地为本文中描述的烷基、环烷基、烯基、环烯基、炔基、环炔基、芳基或杂芳基。如本文中所用的术语“聚醚基”由式-(A1O-A2O)a-表示,其中A1和A2可以独立地为本文中描述的烷基、环烷基、烯基、环烯基、炔基、环炔基、芳基或杂芳基并且“a”是1-500的整数。聚醚基团的实例包括聚氧化乙烯、聚氧化丙烯、以及聚氧化丁烯。 The term "ether" as used herein is represented by the formula A1OA2 , wherein A1 and A2 may independently be alkyl, cycloalkyl, alkenyl, cycloalkenyl, alkynyl, cycloalkyne as described herein aryl, aryl or heteroaryl. The term "polyether group" as used herein is represented by the formula -(A 1 OA 2 O) a -, wherein A 1 and A 2 may independently be alkyl, cycloalkyl, alkenyl, cyclic as described herein alkenyl, alkynyl, cycloalkynyl, aryl or heteroaryl and "a" is an integer from 1-500. Examples of polyether groups include polyethylene oxide, polypropylene oxide, and polybutylene oxide.
术语“聚合的”包括聚烯烃、聚醚、聚酯和其它具有重复单元的基团,例如但不限于-(CH2O)n-CH3、-(CH2CH2O)n-CH3、-[CH2CH(CH3)]n-CH3、-[CH2CH(COOCH3)]n-CH3、-[CH2CH(COOCH2CH3)]n-CH3、和-[CH2CH(COOtBu)]n-CH3,其中n为整数(例如,n>1或者n>2)。The term "polymeric" includes polyolefins, polyethers, polyesters, and other groups having repeating units such as, but not limited to, -(CH2O) n - CH3 , - ( CH2CH2O ) n - CH3 , -[CH 2 CH(CH 3 )] n -CH 3 , -[CH 2 CH(COOCH 3 )] n -CH 3 , -[CH 2 CH(COOCH 2 CH 3 )] n -CH 3 , and - [CH2CH( COOtBu )] n- CH3 , where n is an integer (eg, n>1 or n>2).
如本文中所用的术语“卤素”是指卤素氟、氯、溴和碘。The term "halogen" as used herein refers to the halogens fluorine, chlorine, bromine and iodine.
如本文中所用的术语“杂环基”是指单环和多环非芳族环体系且如本文中所用的术语“杂芳基”是指单环和多环芳族环体系:其中环成员的至少一个不同于碳。所述术语包括氮杂环丁烷、二氧六烷、呋喃、咪唑、异噻唑、异唑、吗啉、唑、唑(二唑)(包括1,2,3-二唑、1,2,5-二唑和1,3,4-二唑)、哌嗪、哌啶、吡嗪、吡唑、哒嗪、吡啶、嘧啶、吡咯、吡咯烷、四氢呋喃、四氢吡喃、四嗪(包括1,2,4,5-四嗪)、四唑(包括1,2,3,4-四唑和1,2,4,5-四唑)、噻二唑(包括1,2,3-噻二唑、1,2,5-噻二唑和1,3,4-噻二唑)、噻唑、噻吩、三嗪(包括1,3,5-三嗪和1,2,4-三嗪)、三唑(包括1,2,3-三唑、1,3,4-三唑)等。The term "heterocyclyl" as used herein refers to monocyclic and polycyclic non-aromatic ring systems and the term "heteroaryl" as used herein refers to monocyclic and polycyclic aromatic ring systems: wherein the ring members at least one of which is different from carbon. The term includes azetidine, dioxane, furan, imidazole, isothiazole, iso azole, morpholine, azole, azole ( oxadiazole) (including 1,2,3- oxadiazole, 1,2,5- oxadiazole and 1,3,4- oxadiazole), piperazine, piperidine, pyrazine, pyrazole, pyridazine, pyridine, pyrimidine, pyrrole, pyrrolidine, tetrahydrofuran, tetrahydropyran, tetrazine (including 1,2,4,5-tetrazine) , tetrazole (including 1,2,3,4-tetrazole and 1,2,4,5-tetrazole), thiadiazole (including 1,2,3-thiadiazole, 1,2,5-thiadiazole oxadiazole and 1,3,4-thiadiazole), thiazole, thiophene, triazine (including 1,3,5-triazine and 1,2,4-triazine), triazole (including 1,2,3 - triazole, 1,3,4-triazole) and the like.
如本文中所用的术语“羟基”由式-OH表示。The term "hydroxyl" as used herein is represented by the formula -OH.
如本文中所用的术语“酮”由式A1C(O)A2表示,其中A1和A2可以独立地为如本文中描述的烷基、环烷基、烯基、环烯基、炔基、环炔基、芳基或杂芳基。 The term "ketone" as used herein is represented by the formula A1C ( O)A2, wherein A1 and A2 may independently be alkyl, cycloalkyl, alkenyl, cycloalkenyl, Alkynyl, cycloalkynyl, aryl or heteroaryl.
如本文中所用的术语“叠氮基”由式-N3表示。The term "azido" as used herein is represented by the formula -N 3 .
如本文中所用的术语“硝基”由式-NO2表示。The term "nitro" as used herein is represented by the formula -NO 2 .
如本文中所用的术语“腈基”由式-CN表示。The term "nitrile group" as used herein is represented by the formula -CN.
如本文中所用的术语“甲硅烷基”由式-SiA1A2A3表示,其中A1、A2和A3可以独立地为氢或如本文中描述的烷基、环烷基、烷氧基、烯基、环烯基、炔基、环炔基、芳基或杂芳基。The term "silyl" as used herein is represented by the formula -SiA 1 A 2 A 3 , wherein A 1 , A 2 and A 3 can independently be hydrogen or alkyl, cycloalkyl, alkane as described herein Oxy, alkenyl, cycloalkenyl, alkynyl, cycloalkynyl, aryl or heteroaryl.
如本文中所用的术语“硫-氧代”由式-S(O)A1、-S(O)2A1、-OS(O)2A1、或-OS(O)2OA1表示,其中A1可为氢或如本文中描述的烷基、环烷基、烯基、环烯基、炔基、环炔基、芳基或杂芳基。在整个本说明书中,“S(O)”是S=O的简化符号。如本文中所用的术语“磺酰基”指的是由式-S(O)2A1表示的硫-氧代基团,其中A1可为氢或如本文中描述的烷基、环烷基、烯基、环烯基、炔基、环炔基、芳基或杂芳基。如本文中所用的术语“砜”由式A1S(O)2A2表示,其中A1和A2可以独立地为如本文中描述的烷基、环烷基、烯基、环烯基、炔基、环炔基、芳基或杂芳基。如本文中所用的术语“亚砜”由式A1S(O)A2表示,其中A1和A2可以独立地为如本文中描述的烷基、环烷基、烯基、环烯基、炔基、环炔基、芳基或杂芳基。The term "thio-oxo" as used herein is represented by the formula -S(O)A 1 , -S(O) 2 A 1 , -OS(O) 2 A 1 , or -OS(O) 2 OA 1 , where A 1 can be hydrogen or an alkyl, cycloalkyl, alkenyl, cycloalkenyl, alkynyl, cycloalkynyl, aryl, or heteroaryl group as described herein. Throughout this specification, "S(O)" is a shorthand notation for S=O. The term "sulfonyl" as used herein refers to a thio-oxo group represented by the formula -S(O)2A1, wherein A1 can be hydrogen or an alkyl, cycloalkyl as described herein , alkenyl, cycloalkenyl, alkynyl, cycloalkynyl, aryl or heteroaryl. The term "sulfone" as used herein is represented by the formula A1S (O ) 2A2 , wherein A1 and A2 may independently be alkyl, cycloalkyl, alkenyl, cycloalkenyl as described herein , alkynyl, cycloalkynyl, aryl or heteroaryl. The term "sulfoxide" as used herein is represented by the formula A1S (O ) A2, wherein A1 and A2 may independently be alkyl, cycloalkyl, alkenyl, cycloalkenyl as described herein , alkynyl, cycloalkynyl, aryl or heteroaryl.
如本文中所用的术语“硫醇基”由式-SH表示。The term "thiol" as used herein is represented by the formula -SH.
如本文中所用的“R1”、“R2”、“R3”、“Rn”,其中n是整数,可以独立地具有一个或多个上面所列出的基团。例如,如果R1是直链烷基,则所述烷基的氢原子之一可以任选地被羟基、烷氧基、烷基、卤素等取代。取决于所选择的基团,第一基团可以引入到第二基团内,或者替代地,第一基团可以悬挂(即连接)到第二基团上。例如,对于短语“包含氨基的烷基”,氨基可以引入到烷基的骨架内。替代地,氨基可以连接到烷基的骨架。所选择的基团的性质将决定第一基团是嵌入还是连接到第二基团。As used herein, "R1", "R2", " R3 ", " Rn ", wherein n is an integer, may independently have one or more of the above - listed groups. For example, if R1 is a straight chain alkyl group, one of the hydrogen atoms of the alkyl group may be optionally substituted with hydroxy, alkoxy, alkyl, halogen, and the like. Depending on the group chosen, the first group can be incorporated into the second group, or alternatively, the first group can be pendant (ie, attached) to the second group. For example, for the phrase "an alkyl group containing an amino group," an amino group can be incorporated into the backbone of the alkyl group. Alternatively, the amino group can be attached to the backbone of the alkyl group. The nature of the selected group will determine whether the first group is intercalated or attached to the second group.
本文中所述化合物可以含有“任选地取代的”部分。通常,不管前面是否加上术语“任选地”,术语“取代的”都表示指定部分的一个或多个氢原子被合适的取代基替代。除非另外说明,“任选地取代的”基团可以在所述基团的每个可取代的位置处具有合适的取代基,并且当任意给定结构中的超过一个位置可以被选自规定基团的超过一个取代基取代时,在每个位置处,取代基可以是相同或不同的。由本发明所预期的取代基的组合优选为导致形成稳定的或化学上可行的化合物的那些。还设计,在一些方面中,除非明确地相反说明,否则单独的取代基可以为进一步任选地取代的(即进一步取代的或未取代的)。The compounds described herein may contain "optionally substituted" moieties. In general, the term "substituted", whether or not preceded by the term "optionally", means that one or more hydrogen atoms of the specified moiety have been replaced with a suitable substituent. Unless otherwise specified, an "optionally substituted" group may have a suitable substituent at each substitutable position of the group, and when more than one position in any given structure may be selected from the specified group When more than one substituent of a group is substituted, the substituents may be the same or different at each position. Combinations of substituents contemplated by the present invention are preferably those that result in the formation of stable or chemically feasible compounds. It is also contemplated that, in some aspects, individual substituents may be further optionally substituted (ie, further substituted or unsubstituted) unless explicitly stated to the contrary.
在一些方面中,化合物的结构可以由下式表示:In some aspects, the structure of the compound can be represented by the formula:
其被理解为与下式等同:It is understood to be equivalent to:
其中n典型地为整数。即,Rn理解为表示五个独立的取代基Rn(a)、Rn(b)、Rn(c)、Rn(d)、Rn (e)。“独立的取代基”表示各R取代基可以被独立地定义。例如,如果在一种情形下Rn(a)是卤素,则Rn(b)在该情形下不一定是卤素。where n is typically an integer. That is, R n is understood to represent five independent substituents R n(a) , R n(b) , R n(c) , R n(d) , R n (e) . "Independent substituent" means that each R substituent may be independently defined. For example, if in one instance Rn(a) is halogen, then Rn (b) in that instance is not necessarily halogen.
在本文中公开和描述的化学结构和部分中数次提到了R1、R2、R3、R4、R5、R6等。在本说明书中的R、R1、R2、R3、R4、R5、R6等的任何描述分别可适用于列举R、R1、R2、R3、R4、R5、R6等的任何结构或部分。R1, R2, R3 , R4 , R5 , R6 , etc. are mentioned several times in the chemical structures and moieties disclosed and described herein . Any description of R, R 1 , R 2 , R 3 , R 4 , R 5 , R 6 , etc. in this specification may apply to listing R, R 1 , R 2 , R 3 , R 4 , R 5 , any structure or moiety of R 6 etc.
由于许多原因,利用有机材料的光电子器件正变得越来越合乎需要。用于制造这样的器件的材料中的许多是相对便宜的,因此有机光电子器件由于成本优势而相对于无机器件具有潜力。此外,有机材料的内在性质,例如它们的柔性,可以使得它们非常适合用于特定的应用例如在柔性基板上的制造。有机光电子器件的实例包括有机发光器件(有机发光二极管)(OLED)、有机光电晶体管、有机光伏电池、和有机光电探测器。对于OLED,有机材料相对于常规的材料可具有性能优势。例如,有机发光层发射光的波长可以通常使用合适的掺杂剂容易地调节。Optoelectronic devices utilizing organic materials are becoming increasingly desirable for a number of reasons. Many of the materials used to fabricate such devices are relatively inexpensive, so organic optoelectronic devices have potential relative to inorganic devices due to cost advantages. Furthermore, the intrinsic properties of organic materials, such as their flexibility, can make them well suited for specific applications such as fabrication on flexible substrates. Examples of organic optoelectronic devices include organic light emitting devices (organic light emitting diodes) (OLEDs), organic phototransistors, organic photovoltaic cells, and organic photodetectors. For OLEDs, organic materials can have performance advantages over conventional materials. For example, the wavelength of light emitted by the organic light-emitting layer can be easily adjusted, usually using suitable dopants.
激子从单重激发态衰变至基态以产生迅速的发光,其为荧光。激子从三重激发态衰变至基态以产生发光,其为磷光。因为重金属原子的强的自旋轨道耦合非常有效地增强在单重和三重激发态之间的系间穿越(ISC),磷光金属络合物如铂络合物已经显示了它们俘获单重态和三重态激子以实现100%内量子效率的潜力。因此,磷光金属络合物是在有机发光器件(OLED)的发射层中作为掺杂剂的良好候选者且在学术和工业领域中已得到大量关注。Excitons decay from a singlet excited state to a ground state to produce rapid luminescence, which is fluorescence. The excitons decay from the triplet excited state to the ground state to produce luminescence, which is phosphorescence. Because the strong spin-orbit coupling of heavy metal atoms highly effectively enhances intersystem crossing (ISC) between singlet and triplet excited states, phosphorescent metal complexes such as platinum complexes have been shown to trap singlet and triplet states. Potential for triplet excitons to achieve 100% internal quantum efficiency. Therefore, phosphorescent metal complexes are good candidates as dopants in the emission layer of organic light-emitting devices (OLEDs) and have received a lot of attention in both academic and industrial fields.
然而,迄今为止,蓝光电致发光器件仍然是这种技术的最富挑战性的领域,这至少部分地是由于蓝光器件的不稳定性。通常理解,主体材料的选择是蓝光器件的稳定性中的一个因素。但是,蓝色磷光体的最低三重激发态(T1)能量高,这通常意味着,蓝光器件的主体材料的最低三重激发态(T1)能量应该甚至更高。这导致在用于蓝光器件的主体材料的开发中的困难。However, blue electroluminescent devices remain by far the most challenging area of this technology, due at least in part to the instability of blue light devices. It is generally understood that the choice of host material is a factor in the stability of blue light devices. However, the lowest triplet excited state (T 1 ) energy of blue phosphors is high, which generally means that the lowest triplet excited state (T 1 ) energy of the host material of the blue device should be even higher. This leads to difficulties in the development of host materials for blue light devices.
通过引入桥接到金属络合物的配体的碳类基团(C、Si、Ge),本公开内容提供了材料设计路线。如本文中所述,发现了与具有相同发射基团的氮桥接Pt络合物相比,碳桥接Pt络合物的光致发光光谱具有显著的蓝移。还发现,可改变发射发光团和配体的化学结构,以及可改变金属以调节金属络合物的单重态能量和三重态能量,其均可影响络合物的光学性质。The present disclosure provides a material design route by introducing carbon-based groups (C, Si, Ge) bridging the ligands of the metal complexes. As described herein, it was found that the photoluminescence spectra of carbon-bridged Pt complexes have a significant blue shift compared to nitrogen-bridged Pt complexes with the same emissive groups. It has also been found that the chemical structure of the emitting luminophore and ligand can be altered, as well as the metal can be altered to tune the singlet and triplet energies of the metal complex, which can all affect the optical properties of the complex.
可将本文中所述的金属络合物根据由特定的发射或者吸收特性所促进的具体应用进行修整(tailor)或调整。在本公开内容中的金属络合物的光学性质可通过以下方法调整:改变围绕金属中心的配体的结构或者改变配体上的荧光发光团的结构。例如,具有拥有给电子取代基或者吸电子取代基的配体的金属络合物通常可呈现出不同的光学性质,包括发射和吸收光谱。金属络合物的颜色可通过以下方法调整:改变配体和荧光发光团上的共轭基团。The metal complexes described herein can be tailored or tailored to specific applications facilitated by specific emission or absorption properties. The optical properties of the metal complexes in the present disclosure can be tuned by changing the structure of the ligand surrounding the metal center or by changing the structure of the fluorophore on the ligand. For example, metal complexes with ligands possessing electron-donating or electron-withdrawing substituents can often exhibit different optical properties, including emission and absorption spectra. The color of the metal complex can be adjusted by changing the conjugated groups on the ligand and fluorophore.
通过例如改变配体或者荧光发光团结构,可将这样的络合物的发射例如从紫外调整至近红外。荧光发光团为在有机分子中的可吸收能量以产生一个或多个单重激发态的原子组(group)。一个或多个单重态激子快速产生衰变以产生迅速的发光。在一个方面中,所述络合物可在可见光谱的大部分范围内提供发射。在一个具体实例中,本文中所述络合物可在约400nm至约700nm的范围内发射光。在另一方面中,所述络合物相对于传统发光络合物具有改善的稳定性和效率。在又一方面中,所述络合物作为在例如生物应用、抗癌剂中的标记物、有机发光二极管(OLED)中的发射体、或者其组合可为有用的。在另一方面中,所述络合物在发光器件,例如,紧凑型荧光灯(CFL)、发光二极管(LED)、白炽灯、及其组合中可为有用的。The emission of such complexes can be tuned, for example, from the ultraviolet to the near-infrared by, for example, changing the ligand or fluorophore structure. A fluorophore is a group of atoms in an organic molecule that can absorb energy to produce one or more singlet excited states. One or more singlet excitons rapidly decay to produce rapid luminescence. In one aspect, the complex can provide emission over a substantial portion of the visible spectrum. In a specific example, the complexes described herein can emit light in the range of about 400 nm to about 700 nm. In another aspect, the complexes have improved stability and efficiency relative to traditional light-emitting complexes. In yet another aspect, the complexes may be useful as labels in, for example, biological applications, anticancer agents, emitters in organic light emitting diodes (OLEDs), or combinations thereof. In another aspect, the complexes can be useful in light emitting devices, eg, compact fluorescent lamps (CFLs), light emitting diodes (LEDs), incandescent lamps, and combinations thereof.
本文中公开了铂化合物、化合物络合物、或者络合物。术语化合物、化合物络合物、和络合物在本文中可互换地使用。在一个方面中,本文中公开的化合物具有电中性(neutral charge)。Disclosed herein are platinum compounds, compound complexes, or complexes. The terms compound, compound complex, and complex are used interchangeably herein. In one aspect, the compounds disclosed herein have neutral charge.
本文中公开的化合物可以呈现出期望的性质并且具有可经由合适配体的选择而调整的发射和/或吸收光谱。在另一方面中,可以排除本文中具体叙述的化合物、结构、或其部分的任意一种或多种。The compounds disclosed herein can exhibit desirable properties and possess emission and/or absorption spectra that can be tuned via the selection of suitable ligands. In another aspect, any one or more of the compounds, structures, or portions thereof specifically recited herein may be excluded.
本文中公开的化合物适合用于各种各样的光学和光电器件,包括,但不限于,光吸收器件例如太阳能敏感器件和光敏器件、有机发光二极管(OLED)、光发射器件、或既能够进行光吸收也能够进行光发射和作为用于生物应用的标志器的器件。The compounds disclosed herein are suitable for use in a wide variety of optical and optoelectronic devices, including, but not limited to, light absorbing devices such as solar and photosensitive devices, organic light emitting diodes (OLEDs), light emitting devices, or both capable of Light absorption also enables light emission and devices as markers for biological applications.
如以上简要描述的,所公开的化合物是铂络合物。在一个方面中,本文中公开的化合物可以用作用于OLED应用例如全色显示器的主体材料。As briefly described above, the disclosed compounds are platinum complexes. In one aspect, the compounds disclosed herein can be used as host materials for OLED applications such as full color displays.
本文中公开的化合物在多种应用中可为有用的。作为发光材料,所述化合物在有机发光二极管(OLED)、发光器件和显示器、以及其它发光器件中可为有用的。The compounds disclosed herein can be useful in a variety of applications. As light-emitting materials, the compounds may be useful in organic light-emitting diodes (OLEDs), light-emitting devices and displays, and other light-emitting devices.
在另一方面中,与常规材料相比,所述化合物可以在照明器件例如有机发光器件中提供改善的效率和/或改善的工作寿命。In another aspect, the compounds can provide improved efficiency and/or improved operating lifetime in lighting devices, such as organic light emitting devices, as compared to conventional materials.
本文中所述的化合物可以使用多种方法制造,所述方法包括但不限于实施例中所叙述的那些。The compounds described herein can be made using a variety of methods including, but not limited to, those recited in the Examples.
本文中所公开的化合物包括延迟荧光发射体、磷光发射体、或者其组合。在一个方面中,本文中所公开的化合物为延迟荧光发射体。在另一方面中,本文中所公开的化合物为磷光发射体。在又一方面中,本文中所公开的化合物为延迟荧光发射体和磷光发射体两者。The compounds disclosed herein include delayed fluorescent emitters, phosphorescent emitters, or combinations thereof. In one aspect, the compounds disclosed herein are delayed fluorescence emitters. In another aspect, the compounds disclosed herein are phosphorescent emitters. In yet another aspect, the compounds disclosed herein are both delayed fluorescent emitters and phosphorescent emitters.
本文中公开了式I和式II的络合物:Disclosed herein are complexes of Formula I and Formula II:
其中:in:
Ar为五元杂芳基、五元卡宾、五元N-杂环卡宾、六元芳基、或者六元杂芳基,Ar is five-membered heteroaryl, five-membered carbene, five-membered N-heterocyclic carbene, six-membered aryl, or six-membered heteroaryl,
每个R1独立地为 或者 Each R1 is independently or
R、R2、R3、R4、R5、R6、R7、R8、R9、R10、R11、和R12中的每个独立地为氢、卤素、羟基、硝基、硫醇基;取代的或者未取代的:C1-C4烷基、烷氧基、芳基、或者氨基,其中当Ar为五元环时R不存在,Each of R, R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , and R 12 is independently hydrogen, halogen, hydroxy, nitro , thiol; substituted or unsubstituted: C 1 -C 4 alkyl, alkoxy, aryl, or amino, wherein R is absent when Ar is a five-membered ring,
X为O、S、S=O、O=S=O、Se、Se=O、O=Se=O、NR2a、PR2b、AsR2c、CR2dR2e、SiR2fR2g、或者BR2h,X is O, S, S=O, O=S=O, Se, Se=O, O=Se=O, NR 2a , PR 2b , AsR 2c , CR 2d R 2e , SiR 2f R 2g , or BR 2h ,
R2a、R2b、R2c、R2d、R2e、R2f、R2g、和R2h中的每个独立地为取代的或者未取代的C1-C4烷基或者芳基,Each of R 2a , R 2b , R 2c , R 2d , R 2e , R 2f , R 2g , and R 2h is independently a substituted or unsubstituted C 1 -C 4 alkyl or aryl,
Y存在或者不存在,以及如果存在,Y为O、S、S=O、O=S=O、Se、Se=O、O=Se=O、NR3a、PR3b、AsR3c、CR3dR3e、SiR3fR3g、或者BR3h,和Y is present or absent, and if present, Y is O, S, S=O, O=S=O, Se, Se=O, O=Se=O, NR 3a , PR 3b , AsR 3c , CR 3d R 3e , SiR 3f R 3g , or BR 3h , and
R3a、R3b、R3c、R3d、R3e、R3f、R3g、和R3h中的每个独立地为取代的或者未取代的C1-C4烷基或者芳基。Each of R 3a , R 3b , R 3c , R 3d , R 3e , R 3f , R 3g , and R 3h is independently substituted or unsubstituted C 1 -C 4 alkyl or aryl.
在一些情况中,Ar为吡唑、咪唑、唑、噻唑、吡啶等。在一些情况中,在相同环或者相邻环上的R、R2、R3、R4、R5、R6、R7、R8、R9、R10、R11、和R12中的任意两个结合在一起以形成稠环体系。例如,R和R2、R2和R4、或者R4和R6可结合以与Ar形成稠环体系,例如苯并咪唑、苯并唑、苯并噻唑、吲唑、喹啉、异喹啉、咪唑并[1,5-a]吡啶等。In some cases, Ar is pyrazole, imidazole, azoles, thiazoles, pyridines, etc. In some cases, in R, R 2 , R 3 , R 4 , R 5 , R 6 , R 7 , R 8 , R 9 , R 10 , R 11 , and R 12 on the same ring or on adjacent rings Any two of them are combined together to form a fused ring system. For example, R and R 2 , R 2 and R 4 , or R 4 and R 6 can be combined to form a fused ring system with Ar, such as benzimidazole, benzo azole, benzothiazole, indazole, quinoline, isoquinoline, imidazo[1,5-a]pyridine, etc.
在一些情况中,X直接连接至R10或者R11。在一些情况中,Y直接连接至R6或者R8。In some cases, X is directly linked to R 10 or R 11 . In some cases, Y is directly linked to R 6 or R 8 .
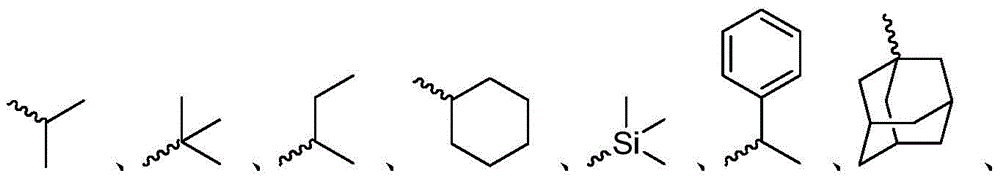
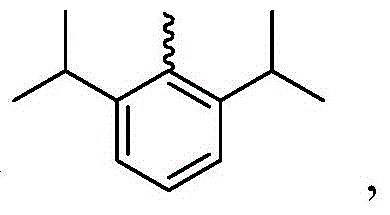
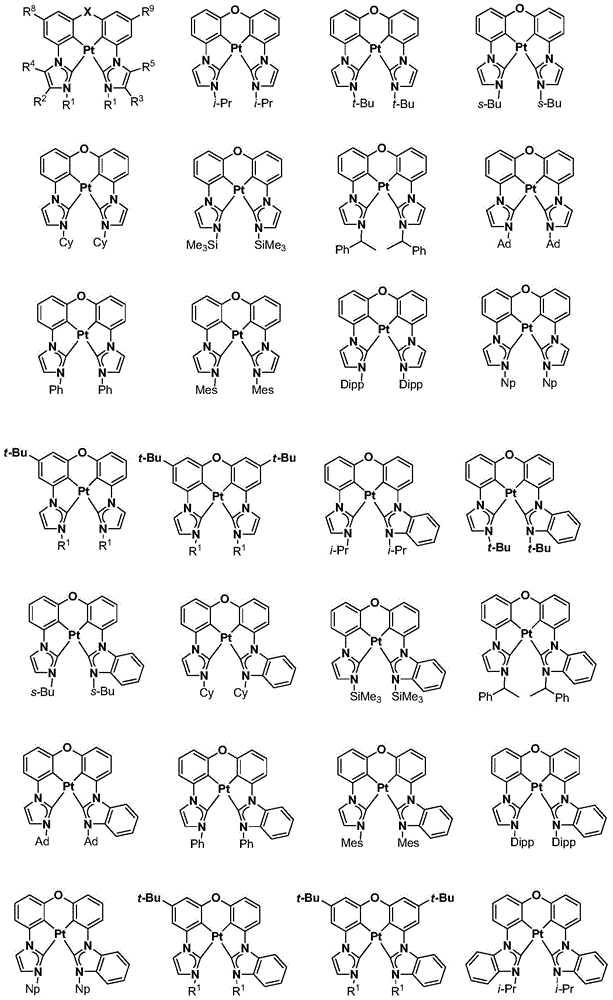
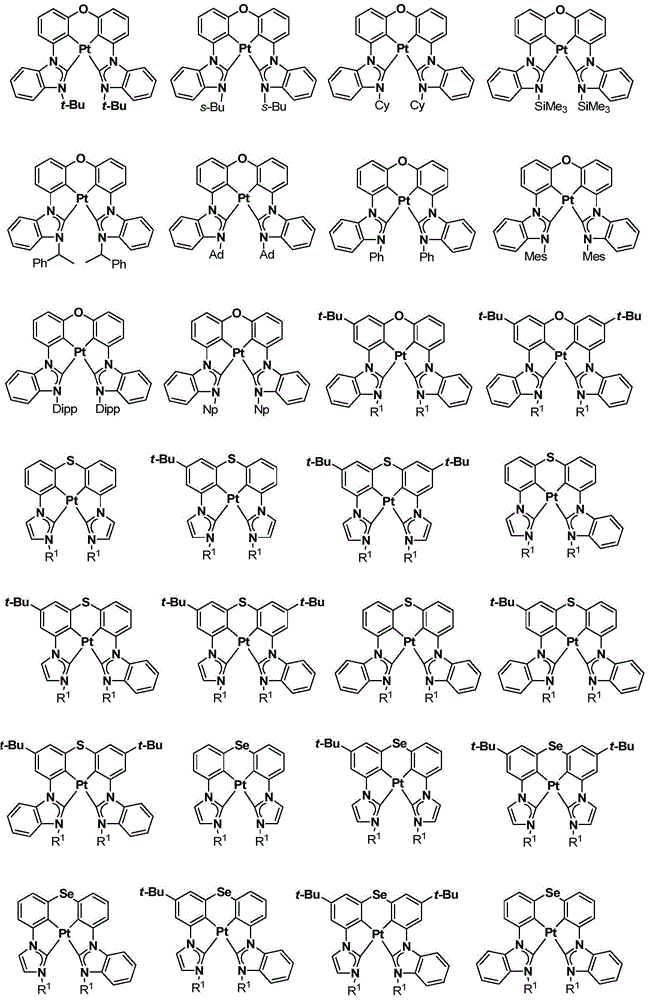
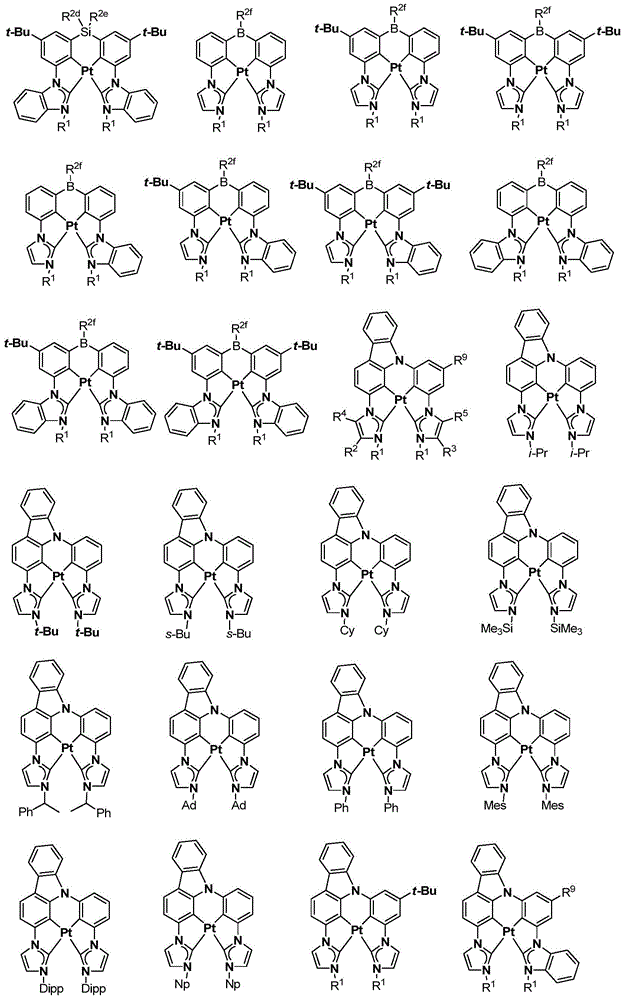
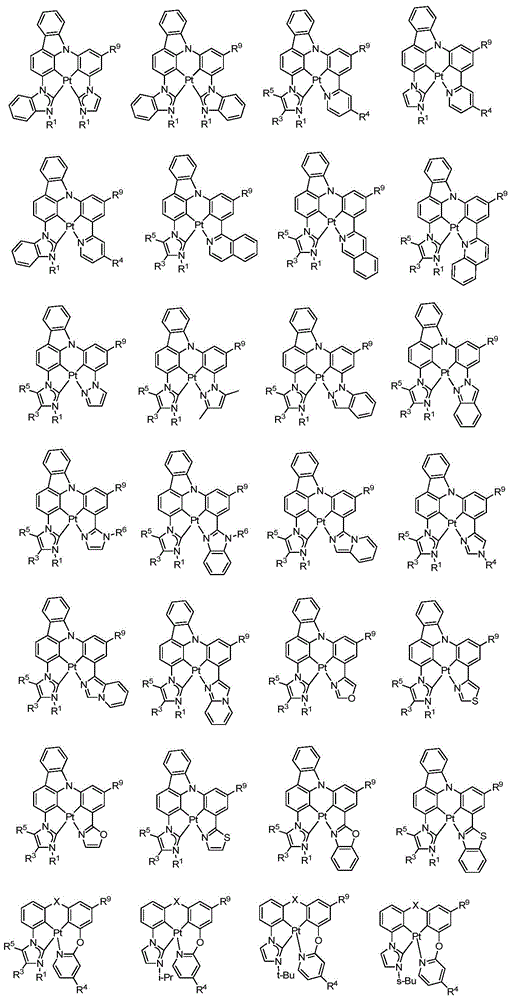
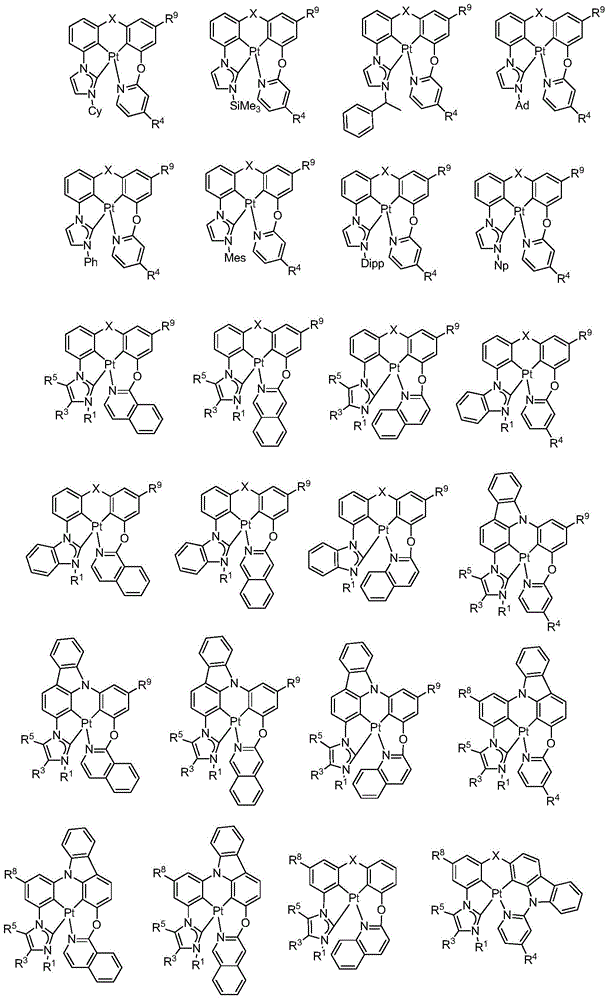
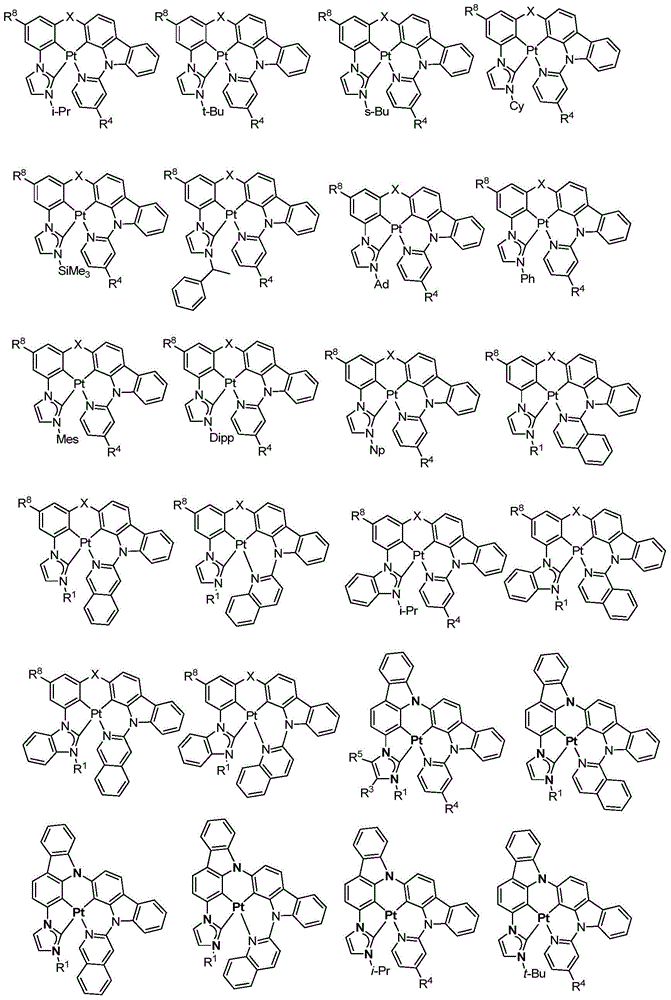
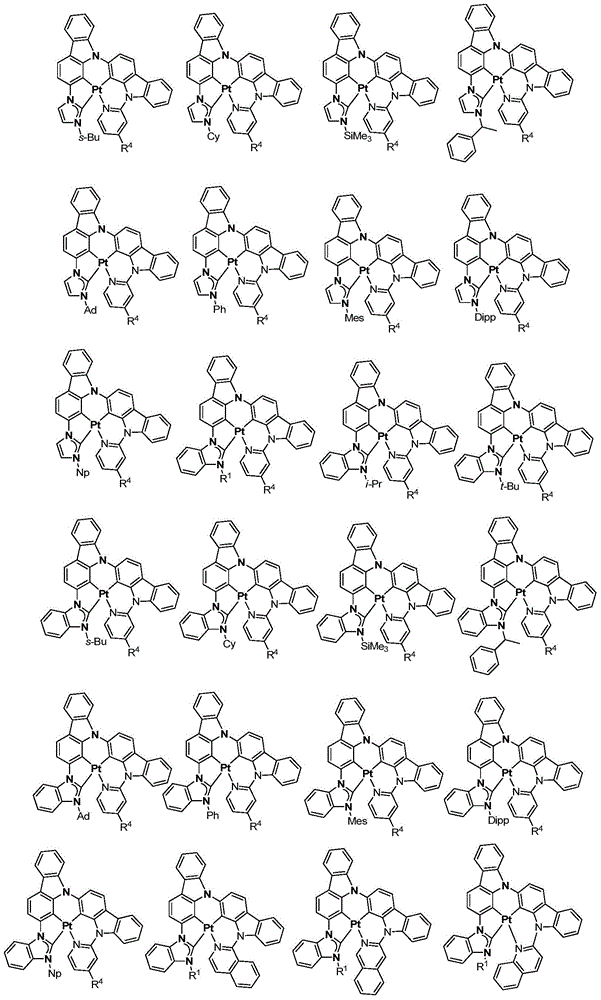
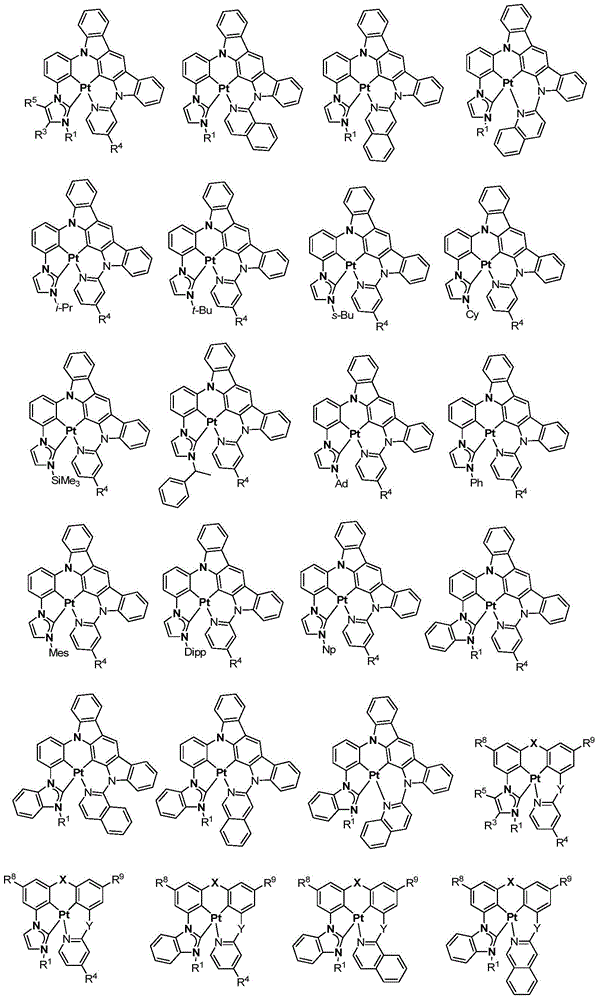
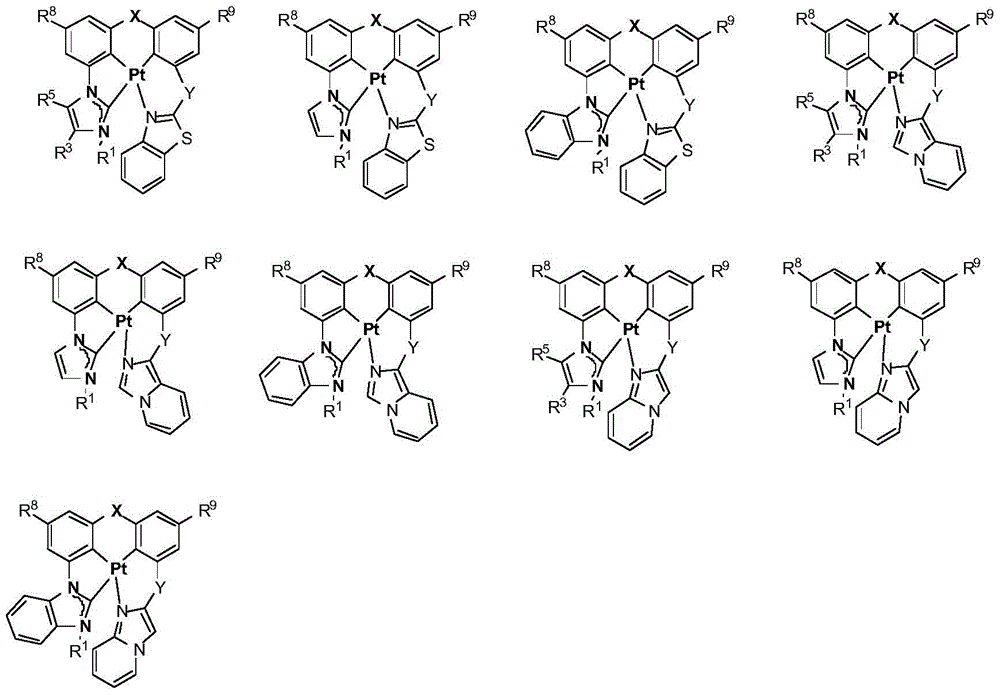
在本公开内容中的金属络合物包括以下结构中的一种或者多种。在本公开内容中的金属络合物也可包括本文中未具体描述的其它结构或者其部分,并且本公开内容不意图限于具体描述的那些结构或者其部分。在下面的结构中,“Ad”是指“金刚烷基”;“Mes”是指“基(mesityl)”;“Dipp”是指“二异丙基苯基”;“Np”是指“新戊基”;和“Cy”是指“环己基”。Metal complexes in the present disclosure include one or more of the following structures. The metal complexes in the present disclosure may also include other structures or portions thereof not specifically described herein, and the present disclosure is not intended to be limited to those structures or portions thereof specifically described. In the structures below, "Ad" refers to "adamantyl";"Mes" refers to " "Dipp" means "diisopropylphenyl";"Np" means "neopentyl"; and "Cy" means "cyclohexyl".
本文中还公开了包含本文中公开的络合物中的一种或者多种的器件。Also disclosed herein are devices comprising one or more of the complexes disclosed herein.
本文中公开的络合物适合用于各种各样的器件,包括例如光学和光电器件,包括,例如光吸收器件例如太阳能敏感器件和光敏器件、有机发光二极管(OLED)、光发射器件、或既能够进行光吸收也能够进行光发射和作为用于生物应用的标志器的器件。The complexes disclosed herein are suitable for use in a wide variety of devices, including, for example, optical and optoelectronic devices, including, for example, light absorbing devices such as solar and photosensitive devices, organic light emitting diodes (OLEDs), light emitting devices, or Devices capable of both light absorption and light emission and as markers for biological applications.
本文中还公开了包含本文中公开的络合物中的一种或者多种的器件。Also disclosed herein are devices comprising one or more of the complexes disclosed herein.
本文中公开的络合物适合用于各种各样的器件,包括例如光学和光电器件,包括,例如光吸收器件例如太阳能敏感器件和光敏器件、有机发光二极管(OLED)、光发射器件、或既能够进行光吸收也能够进行光发射和作为用于生物应用的标志器的器件。The complexes disclosed herein are suitable for use in a wide variety of devices, including, for example, optical and optoelectronic devices, including, for example, light absorbing devices such as solar and photosensitive devices, organic light emitting diodes (OLEDs), light emitting devices, or Devices capable of both light absorption and light emission and as markers for biological applications.
本文中所述的络合物可用于OLED。图1描绘OLED 100的横截面图。OLED 100包含基底102、阳极104、空穴传输材料(HTL)106、光处理材料108、电子传输材料(ETL)110、和金属阴极层112。阳极104典型地为透明材料,例如氧化铟锡。光处理材料108可为包含发射体和主体的发射材料(EML)。The complexes described herein can be used in OLEDs. FIG. 1 depicts a cross-sectional view of
在多个方面中,图1中所描绘的一个或者多个层中的任何层可包含氧化铟锡(ITO)、聚(3,4-亚乙基二氧噻吩)(PEDOT)、聚磺苯乙烯(PSS)、N,N’-二-1-萘基-N,N-二苯基-1,1’-联苯-4,4’-二胺(NPD)、1,1-双((二-4-甲苯基氨基)苯基)环己烷(TAPC)、2,6-双(N-咔唑基)吡啶(mCpy)、2,8-双(二苯基磷酰基)二苯并噻吩(PO15)、LiF、Al、或其组合。In various aspects, any of the one or more layers depicted in FIG. 1 can comprise indium tin oxide (ITO), poly(3,4-ethylenedioxythiophene) (PEDOT), polysulfobenzene Ethylene (PSS), N,N'-di-1-naphthyl-N,N-diphenyl-1,1'-biphenyl-4,4'-diamine (NPD), 1,1-bis( (Bis-4-Tolylamino)phenyl)cyclohexane (TAPC), 2,6-bis(N-carbazolyl)pyridine (mCpy), 2,8-bis(diphenylphosphoryl)diphenyl thiophene (PO15), LiF, Al, or a combination thereof.
光处理材料108可以包括本公开内容的一种或多种络合物以及主体材料或者没有主体材料。主体材料可以是任何合适的已知的主体材料。OLED的发射颜色由光处理材料108的发射能量(光学能隙)决定,其可以通过调整发光络合物和/或主体材料的电子结构而进行调整。HTL层106中的空穴传输材料和ETL层110中的电子传输材料两者都可以包括任何合适的已知的空穴传输物。The
本文中描述的络合物可以呈现出磷光。磷光OLED(即具有磷光发射体的OLED)典型地具有比其它OLED例如荧光OLED高的器件效率。基于电致磷光发射体的发光器件更详细地描述于Baldo等人的WO2000/070655中,由于该文献对于OLED且特别是磷光OLED的教导,将其引入本文中。The complexes described herein can exhibit phosphorescence. Phosphorescent OLEDs (ie, OLEDs with phosphorescent emitters) typically have higher device efficiencies than other OLEDs, such as fluorescent OLEDs. Light emitting devices based on electrophosphorescent emitters are described in more detail in WO2000/070655 to Baldo et al., which is incorporated herein for its teachings on OLEDs and in particular phosphorescent OLEDs.
实施例Example
提出下列实施例以向本领域普通技术人员提供如何制造和评价本文中描述的化合物、组合物、制品、器件和/或方法的完整公开内容和描述,并且所述实施例意图仅是示范性的且不意图限制范围。尽管已经进行努力以确保关于数值(例如,量、温度等)的准确性,但是应当考虑一些误差和偏差。除非另外说明,否则份数为重量份,温度是以℃为单位或者在环境温度下,且压力是在大气压下或附近。The following examples are presented to provide those of ordinary skill in the art with a complete disclosure and description of how to make and evaluate the compounds, compositions, articles, devices and/or methods described herein, and are intended to be exemplary only and is not intended to limit the scope. Although efforts have been made to ensure accuracy with respect to numerical values (eg, amounts, temperature, etc.), some errors and deviations should be accounted for. Unless otherwise stated, parts are parts by weight, temperature is in °C or at ambient temperature, and pressure is at or near atmospheric.
在实施例中叙述用于本文中所描述的化合物的制备方法的多种方法。这些方法被提供用于说明多种制备方法,但是不意图限制本文中所叙述的方法的任一种。因此,本公开内容所属领域的技术人员可容易地修改所叙述的方法或利用不同的方法来制备本文中所述化合物的一种或多种。下列方面仅是示例性的,且不意图限制范围、温度、催化剂、浓度、反应物组成、以及其它工艺条件可改变,并且对于期望的络合物,本公开内容所属领域的技术人员可容易地选择合适的反应物和条件。Various methods for the preparation of the compounds described herein are described in the Examples. These methods are provided to illustrate various methods of preparation, but are not intended to limit any of the methods described herein. Accordingly, one skilled in the art to which this disclosure pertains can readily modify the described methods or utilize different methods to prepare one or more of the compounds described herein. The following aspects are exemplary only, and are not intended to be limiting. The ranges, temperatures, catalysts, concentrations, reactant compositions, and other process conditions may vary, and for desired complexes, those skilled in the art to which this disclosure pertains can readily Choose appropriate reactants and conditions.
在Varian Liquid State NMR仪器上于CDCl3或DMSO-d6溶液中以400MHz记录1H谱,以100MHz记录13C NMR谱,且化学位移参照残留的质子化(protiated)溶剂。如果将CDCl3用作溶剂,则采用四甲基硅烷(δ=0.00ppm)作为内标记录1H NMR谱;采用CDCl3(δ=77.00ppm)作为内标记录13C NMR谱。如果将DMSO-d6用作溶剂,则采用残留的H2O(δ=3.33ppm)作为内标记录1H NMR谱;采用DMSO-d6(δ=39.52ppm)作为内标记录13C NMR谱。使用下列缩写(或其组合)来说明1H NMR的多重性:s=单重,d=双重,t=三重,q=四重,p=五重,m=多重,br=宽。 1 H spectra were recorded at 400 MHz and 13 C NMR spectra were recorded at 100 MHz in CDCl 3 or DMSO-d 6 solution on a Varian Liquid State NMR instrument and chemical shifts were referenced to residual protonated solvent. If CDCl3 was used as solvent, 1H NMR spectra were recorded using tetramethylsilane (δ=0.00 ppm) as internal standard; 13C NMR spectra were recorded using CDCl3 (δ=77.00 ppm) as internal standard. If DMSO-d 6 was used as solvent, 1 H NMR spectra were recorded using residual H 2 O (δ = 3.33 ppm) as internal standard; 13 C NMR spectra were recorded using DMSO-d 6 (δ = 39.52 ppm) as internal standard spectrum. The following abbreviations (or combinations thereof) are used to illustrate the multiplicity of1H NMR: s = single, d=double, t=triple, q=quad, p=quintet, m=multiple, br=broad.
参照下面的对于Pt7O7-dipr的方案1描述用于本文中公开的络合物的示例性合成方法。Exemplary synthetic methods for the complexes disclosed herein are described with reference to Scheme 1 below for Pt7O7-dipr.
方案1plan 1
向经烘箱干燥的烧瓶添加Pt7O7-dipr的配体(610mg,0.9mmol)、K2PtCl4(392mg,0.945mmol)、和n-Bu4NBr(29mg,0.09mmol)。将烧瓶用N2排空和回填,然后在N2的保护下添加HOAc(45mL,0.02M)。然后将混合物在120℃加热3天。将所得混合物冷却至室温并在减压下浓缩。通过快速柱色谱法在硅胶上的纯化(DCM/己烷=1/1至3/1)提供作为淡黄色固体的Pt7O7-dipr(99mg,19%的产率)。1H NMR(DMSO-d6,400MHz):δ8.04(d,J=2.0Hz,2H),7.65(d,J=2.0Hz,2H),7.20(d,J=7.4Hz,2H),7.11(t,J=7.8Hz,2H),6.96-6.85(m,2H),4.78(七重峰(sept),J=6.6Hz,2H),1.47(d,J=6.6Hz,12H)。Pt7O7-dipr在室温和77K的光致发光光谱在图2中示出。To the oven dried flask was added the ligand for Pt7O7 - dipr (610 mg, 0.9 mmol), K2PtCl4 ( 392 mg, 0.945 mmol), and n-Bu4NBr (29 mg, 0.09 mmol). The flask was evacuated and backfilled with N2 , then HOAc (45 mL, 0.02M) was added under the protection of N2 . The mixture was then heated at 120°C for 3 days. The resulting mixture was cooled to room temperature and concentrated under reduced pressure. Purification by flash column chromatography on silica gel (DCM/hexane = 1/1 to 3/1) provided Pt7O7-dipr (99 mg, 19% yield) as a pale yellow solid. 1 H NMR (DMSO-d 6 , 400MHz): δ 8.04 (d, J=2.0 Hz, 2H), 7.65 (d, J=2.0 Hz, 2H), 7.20 (d, J=7.4 Hz, 2H), 7.11 (t, J=7.8 Hz, 2H), 6.96-6.85 (m, 2H), 4.78 (sept, J=6.6 Hz, 2H), 1.47 (d, J=6.6 Hz, 12H). The photoluminescence spectra of Pt7O7-dipr at room temperature and 77 K are shown in Fig. 2.
本文中所述络合物适于作为用于发光器件如OLED(例如,用于全色显示器和照明应用)的发射体。The complexes described herein are suitable as emitters for light-emitting devices such as OLEDs (eg, for full-color displays and lighting applications).
考虑到本说明书,多个方面的进一步修饰和替代实施方案对于本领域技术人员而言将为明晰的。因此,本说明书将被解释为仅说明性的。将理解,本文中示出和描述的形式将被视为实施方案的实例。要素和材料可代替本文中说明和描述的那些要素和材料,部件和工艺可被颠倒,和一些特征可被独立地利用,所有这些在了解本说明书的益处后对于本领域技术人员而言是明晰的。在不脱离如在以下权利要求中所述的精神和范围的情况下,可对本文中所述要素作出改变。Further modifications and alternative embodiments of various aspects will be apparent to those skilled in the art in view of this specification. Accordingly, this specification is to be construed as illustrative only. It is to be understood that the forms shown and described herein are to be considered as examples of embodiments. Elements and materials may be substituted for those illustrated and described herein, components and processes may be reversed, and some features may be utilized independently, all of which will be apparent to those skilled in the art having the benefit of this description of. Changes may be made in elements described herein without departing from the spirit and scope as set forth in the following claims.
Claims (16)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202010883430.8A CN112159432B (en) | 2014-07-24 | 2015-07-24 | Tetradentate platinum(II) complexes cyclometalated with functionalized phenylcarbene ligands and their analogs |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US201462028562P | 2014-07-24 | 2014-07-24 | |
| US62/028,562 | 2014-07-24 |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010883430.8A Division CN112159432B (en) | 2014-07-24 | 2015-07-24 | Tetradentate platinum(II) complexes cyclometalated with functionalized phenylcarbene ligands and their analogs |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN105367605A CN105367605A (en) | 2016-03-02 |
| CN105367605B true CN105367605B (en) | 2020-09-25 |
Family
ID=55167408
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201510441320.5A Active CN105367605B (en) | 2014-07-24 | 2015-07-24 | Tetradentate platinum(II) complexes and their analogs cyclometallated with functionalized phenylcarbene ligands |
| CN202010883430.8A Expired - Fee Related CN112159432B (en) | 2014-07-24 | 2015-07-24 | Tetradentate platinum(II) complexes cyclometalated with functionalized phenylcarbene ligands and their analogs |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202010883430.8A Expired - Fee Related CN112159432B (en) | 2014-07-24 | 2015-07-24 | Tetradentate platinum(II) complexes cyclometalated with functionalized phenylcarbene ligands and their analogs |
Country Status (3)
| Country | Link |
|---|---|
| US (4) | US9923155B2 (en) |
| KR (2) | KR102433486B1 (en) |
| CN (2) | CN105367605B (en) |
Families Citing this family (87)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP5604505B2 (en) | 2009-04-06 | 2014-10-08 | アリゾナ ボード オブ リージェンツ アクティング フォー アンド オン ビハーフ オブ アリゾナ ステイト ユニバーシティ | Synthesis of four-coordinate platinum complexes and their application to light-emitting devices |
| KR20130067276A (en) | 2010-04-30 | 2013-06-21 | 아리조나 보드 오브 리젠츠 퍼 앤 온 비하프 오브 아리조나 스테이트 유니버시티 | Synthesis of four coordinated palladium complexes and their applications in light emitting devices thereof |
| US8816080B2 (en) | 2011-02-18 | 2014-08-26 | Arizona Board Of Regents Acting For And On Behalf Of Arizona State University | Four coordinated platinum and palladium complexes with geometrically distorted charge transfer state and their applications in light emitting devices |
| US9238668B2 (en) | 2011-05-26 | 2016-01-19 | Arizona Board Of Regents, Acting For And On Behalf Of Arizona State University | Synthesis of platinum and palladium complexes as narrow-band phosphorescent emitters for full color displays |
| WO2014031977A1 (en) | 2012-08-24 | 2014-02-27 | Arizona Board Of Regents For And On Behalf Of Arizona State University | Metal compounds and methods and uses thereof |
| WO2014047616A1 (en) | 2012-09-24 | 2014-03-27 | Arizona Board Of Regents For And On Behalf Of Arizona State University | Metal compounds, methods, and uses thereof |
| WO2014109814A2 (en) | 2012-10-26 | 2014-07-17 | Arizona Board Of Regents Acting For And On Behalf Of Arizona State University | Metal complexes, methods, and uses thereof |
| KR102349659B1 (en) | 2013-06-10 | 2022-01-11 | 아리조나 보드 오브 리젠츠 온 비하프 오브 아리조나 스테이트 유니버시티 | Phosphorescent tetradentate metal complexes having modified emission spectra |
| JP6804823B2 (en) | 2013-10-14 | 2020-12-23 | アリゾナ・ボード・オブ・リージェンツ・オン・ビハーフ・オブ・アリゾナ・ステイト・ユニバーシティーArizona Board of Regents on behalf of Arizona State University | Platinum complex and device |
| US10020455B2 (en) | 2014-01-07 | 2018-07-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum and palladium complex emitters containing phenyl-pyrazole and its analogues |
| WO2015131158A1 (en) | 2014-02-28 | 2015-09-03 | Arizona Board Of Regents On Behalf Of Arizona State University | Chiral metal complexes as emitters for organic polarized electroluminescent devices |
| US9941479B2 (en) | 2014-06-02 | 2018-04-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate cyclometalated platinum complexes containing 9,10-dihydroacridine and its analogues |
| US9923155B2 (en) | 2014-07-24 | 2018-03-20 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum (II) complexes cyclometalated with functionalized phenyl carbene ligands and their analogues |
| US9502671B2 (en) | 2014-07-28 | 2016-11-22 | Arizona Board Of Regents On Behalf Of Arizona State University | Tridentate cyclometalated metal complexes with six-membered coordination rings |
| US9818959B2 (en) | 2014-07-29 | 2017-11-14 | Arizona Board of Regents on behlaf of Arizona State University | Metal-assisted delayed fluorescent emitters containing tridentate ligands |
| US10793546B2 (en) | 2014-08-15 | 2020-10-06 | Arizona Board Of Regents On Behalf Of Arizona State University | Non-platinum metal complexes for excimer based single dopant white organic light emitting diodes |
| US9920242B2 (en) | 2014-08-22 | 2018-03-20 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal-assisted delayed fluorescent materials as co-host materials for fluorescent OLEDs |
| US11329244B2 (en) | 2014-08-22 | 2022-05-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Organic light-emitting diodes with fluorescent and phosphorescent emitters |
| US10033003B2 (en) | 2014-11-10 | 2018-07-24 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate metal complexes with carbon group bridging ligands |
| US9865825B2 (en) | 2014-11-10 | 2018-01-09 | Arizona Board Of Regents On Behalf Of Arizona State University | Emitters based on octahedral metal complexes |
| US9711739B2 (en) | 2015-06-02 | 2017-07-18 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate metal complexes containing indoloacridine and its analogues |
| US9879039B2 (en) | 2015-06-03 | 2018-01-30 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate and octahedral metal complexes containing naphthyridinocarbazole and its analogues |
| US11930662B2 (en) | 2015-06-04 | 2024-03-12 | Arizona Board Of Regents On Behalf Of Arizona State University | Transparent electroluminescent devices with controlled one-side emissive displays |
| US10158091B2 (en) | 2015-08-04 | 2018-12-18 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum (II) and palladium (II) complexes, devices, and uses thereof |
| US10211411B2 (en) | 2015-08-25 | 2019-02-19 | Arizona Board Of Regents On Behalf Of Arizona State University | Thermally activated delayed fluorescent material based on 9,10-dihydro-9,9-dimethylacridine analogues for prolonging device longevity |
| DE102016203823B3 (en) * | 2016-03-09 | 2017-08-24 | Ford Global Technologies, Llc | Method for operating a exhaust-gas-charged internal combustion engine with partial deactivation |
| US11335865B2 (en) | 2016-04-15 | 2022-05-17 | Arizona Board Of Regents On Behalf Of Arizona State University | OLED with multi-emissive material layer |
| CN106220645B (en) * | 2016-04-25 | 2018-08-14 | 中节能万润股份有限公司 | Compound based on monosubstituted-9-fluorenone and application thereof |
| US10461266B2 (en) * | 2016-06-14 | 2019-10-29 | Queen's University At Kingston | Luminescent compounds and methods of using same |
| US10177323B2 (en) | 2016-08-22 | 2019-01-08 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum (II) and palladium (II) complexes and octahedral iridium complexes employing azepine functional groups and their analogues |
| US10822363B2 (en) | 2016-10-12 | 2020-11-03 | Arizona Board Of Regents On Behalf Of Arizona State University | Narrow band red phosphorescent tetradentate platinum (II) complexes |
| US11183670B2 (en) | 2016-12-16 | 2021-11-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Organic light emitting diode with split emissive layer |
| US11053268B2 (en) | 2017-01-20 | 2021-07-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| WO2018140765A1 (en) | 2017-01-27 | 2018-08-02 | Jian Li | Metal-assisted delayed fluorescent emitters employing pyrido-pyrrolo-acridine and analogues |
| US10392387B2 (en) | 2017-05-19 | 2019-08-27 | Arizona Board Of Regents On Behalf Of Arizona State University | Substituted benzo[4,5]imidazo[1,2-a]phenanthro[9,10-c][1,8]naphthyridines, benzo[4,5]imidazo[1,2-a]phenanthro[9,10-c][1,5]naphthyridines and dibenzo[f,h]benzo[4,5]imidazo[2,1-a]pyrazino[2,3-c]isoquinolines as thermally assisted delayed fluorescent materials |
| US10615349B2 (en) | 2017-05-19 | 2020-04-07 | Arizona Board Of Regents On Behalf Of Arizona State University | Donor-acceptor type thermally activated delayed fluorescent materials based on imidazo[1,2-F]phenanthridine and analogues |
| US10516117B2 (en) | 2017-05-19 | 2019-12-24 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal-assisted delayed fluorescent emttters employing benzo-imidazo-phenanthridine and analogues |
| US11101435B2 (en) | 2017-05-19 | 2021-08-24 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum and palladium complexes based on biscarbazole and analogues |
| US11832510B2 (en) | 2017-06-23 | 2023-11-28 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11802136B2 (en) | 2017-06-23 | 2023-10-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11814403B2 (en) | 2017-06-23 | 2023-11-14 | Universal Display Corporation | Organic electroluminescent materials and devices |
| KR102606281B1 (en) | 2017-07-14 | 2023-11-27 | 삼성디스플레이 주식회사 | Organometallic compound and organic light-emitting device including the same |
| KR102474204B1 (en) | 2017-07-21 | 2022-12-06 | 삼성디스플레이 주식회사 | Organometallic compound and organic light-emitting device including the same |
| KR102718677B1 (en) | 2017-10-17 | 2024-10-16 | 지안 리 | Phosphorescent excimers with desirable molecular orientation as monochromatic emitters for display and lighting applications |
| US11647643B2 (en) | 2017-10-17 | 2023-05-09 | Arizona Board Of Regents On Behalf Of Arizona State University | Hole-blocking materials for organic light emitting diodes |
| KR102817395B1 (en) | 2017-12-04 | 2025-06-10 | 토미 엘. 로이스터 | Bridged bis(azinyl)amine phosphorescent emitting composition |
| US11081659B2 (en) * | 2018-01-10 | 2021-08-03 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11700765B2 (en) | 2018-01-10 | 2023-07-11 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US12037348B2 (en) | 2018-03-09 | 2024-07-16 | Arizona Board Of Regents On Behalf Of Arizona State University | Blue and narrow band green and red emitting metal complexes |
| KR102661471B1 (en) | 2018-04-27 | 2024-04-29 | 삼성디스플레이 주식회사 | Organometallic compound and organic light emitting device comprising the same |
| WO2020018476A1 (en) | 2018-07-16 | 2020-01-23 | Jian Li | Fluorinated porphyrin derivatives for optoelectronic applications |
| KR102725259B1 (en) | 2018-12-17 | 2024-11-04 | 삼성디스플레이 주식회사 | Organometallic compound and organic light emitting device comprising the same |
| CN109535205B (en) * | 2018-12-30 | 2021-10-08 | 浙江工业大学 | A platinum complex containing a tetradentate ligand and an aryloxy derivative |
| US11878988B2 (en) | 2019-01-24 | 2024-01-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Blue phosphorescent emitters employing functionalized imidazophenthridine and analogues |
| US11594691B2 (en) | 2019-01-25 | 2023-02-28 | Arizona Board Of Regents On Behalf Of Arizona State University | Light outcoupling efficiency of phosphorescent OLEDs by mixing horizontally aligned fluorescent emitters |
| JP2020158491A (en) * | 2019-03-26 | 2020-10-01 | ユニバーサル ディスプレイ コーポレイション | Organic electroluminescent materials and devices |
| KR102804493B1 (en) | 2019-04-30 | 2025-05-09 | 삼성디스플레이 주식회사 | Organic light emitting device |
| KR20200133872A (en) | 2019-05-20 | 2020-12-01 | 삼성디스플레이 주식회사 | Organic electroluminescence device and organometallic complex for organic electroluminescence device |
| EP3750899A1 (en) | 2019-06-13 | 2020-12-16 | Samsung Electronics Co., Ltd. | Organometallic compound, organic light-emitting device including the same, and diagnostic composition including the organometallic compound |
| EP3750901B1 (en) | 2019-06-13 | 2023-12-20 | Samsung Electronics Co., Ltd. | Organometallic compound, organic light-emitting device including the same, and diagnostic composition including the organometallic compound |
| EP3750900A1 (en) * | 2019-06-13 | 2020-12-16 | Samsung Electronics Co., Ltd. | Organometallic compound, organic light-emitting device including the same, and diagnostic composition including the organometallic compound |
| KR102784238B1 (en) | 2019-08-02 | 2025-03-24 | 삼성디스플레이 주식회사 | Organic electroluminescence device and organometallic complex for organic electroluminescence device |
| KR102894388B1 (en) | 2019-08-23 | 2025-12-02 | 삼성디스플레이 주식회사 | Organometallic compound and organic light emitting device comprising the same |
| KR102556834B1 (en) | 2019-10-01 | 2023-07-19 | 삼성디스플레이 주식회사 | Organic electroluminescence device |
| KR102472168B1 (en) * | 2019-10-01 | 2022-11-30 | 삼성디스플레이 주식회사 | Organic electroluminescence device |
| KR102460911B1 (en) | 2019-10-01 | 2022-11-01 | 삼성디스플레이 주식회사 | Organic electroluminescence device |
| US20210119168A1 (en) * | 2019-10-01 | 2021-04-22 | Samsung Display Co., Ltd. | Organic electroluminescence device |
| US11785838B2 (en) | 2019-10-02 | 2023-10-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Green and red organic light-emitting diodes employing excimer emitters |
| KR102508499B1 (en) * | 2019-10-04 | 2023-03-10 | 삼성디스플레이 주식회사 | Organic light emitting device and device with same |
| KR102806420B1 (en) * | 2019-10-21 | 2025-05-13 | 삼성디스플레이 주식회사 | Organic electroluminescence device and organometallic compound for organic electroluminescence device |
| US12382824B2 (en) * | 2019-11-14 | 2025-08-05 | Samsung Display Co., Ltd. | Organic electroluminescence device and organometallic compound for organic electroluminescence device |
| US12247039B2 (en) | 2019-11-27 | 2025-03-11 | Samsung Electronics Co., Ltd. | Organometallic compound, organic light-emitting device including organometallic compound and electronic apparatus including the organic light-emitting device |
| CN112979710B (en) * | 2019-12-16 | 2022-02-15 | 广东阿格蕾雅光电材料有限公司 | A platinum metal complex and its application in organic electroluminescent devices |
| KR102821747B1 (en) * | 2019-12-20 | 2025-06-18 | 삼성디스플레이 주식회사 | Organometallic compound and organic light emitting device comprising the same |
| US12168661B2 (en) | 2020-02-21 | 2024-12-17 | Arizona Board Of Regents On Behalf Of Arizona State University | Functional materials based on stable chemical structure |
| KR102838031B1 (en) | 2020-03-04 | 2025-07-25 | 삼성디스플레이 주식회사 | Organic electroluminescence device and organometallic compound for organic electroluminescence device |
| KR102850626B1 (en) | 2020-03-17 | 2025-08-26 | 삼성디스플레이 주식회사 | Organometallic compound and organic light emitting device comprising the same |
| KR20210124605A (en) | 2020-04-06 | 2021-10-15 | 삼성디스플레이 주식회사 | Organometallic compound and organic light emitting device including the same |
| KR102853327B1 (en) | 2020-05-14 | 2025-09-01 | 삼성디스플레이 주식회사 | Organometallic compound and organic light emitting device comprising the same |
| CN113683625B (en) | 2020-05-19 | 2025-06-10 | 亚利桑那州立大学董事会 | Metal-assisted delayed fluorescence emitters for organic light emitting diodes |
| US11945985B2 (en) | 2020-05-19 | 2024-04-02 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal assisted delayed fluorescent emitters for organic light-emitting diodes |
| KR20220016406A (en) * | 2020-07-31 | 2022-02-09 | 삼성디스플레이 주식회사 | Organometallic compound and light emitting device comprising the same and electronic apparatus comprising the same |
| KR20220037040A (en) | 2020-09-16 | 2022-03-24 | 삼성디스플레이 주식회사 | Organometallic compound and light emitting device comprising the same and electronic apparatus comprising the same |
| CN112125932B (en) * | 2020-09-24 | 2023-12-12 | 中国石油化工股份有限公司 | A divalent platinum complex and its application |
| KR102812310B1 (en) | 2020-10-28 | 2025-05-26 | 삼성전자주식회사 | Organometallic compound, organic light emitting device including the same and an electronic apparatus including the organic light emitting device |
| KR20220085924A (en) | 2020-12-15 | 2022-06-23 | 삼성디스플레이 주식회사 | Light emitting device and electronic apparatus comprising same |
| CN116925148A (en) * | 2022-03-31 | 2023-10-24 | 北京夏禾科技有限公司 | Organic electroluminescent material and device thereof |
Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20130341600A1 (en) * | 2012-06-21 | 2013-12-26 | Universal Display Corporation | Phosphorescent emitters |
| US20140014922A1 (en) * | 2012-07-10 | 2014-01-16 | Universal Display Corporation | Phosphorescent emitters containing dibenzo[1,4]azaborinine structure |
| US20140027733A1 (en) * | 2012-07-19 | 2014-01-30 | Universal Display Corporation | Transition metal complexes containing substituted imidazole carbene as ligands and their application in oleds |
| WO2014047616A1 (en) * | 2012-09-24 | 2014-03-27 | Arizona Board Of Regents For And On Behalf Of Arizona State University | Metal compounds, methods, and uses thereof |
Family Cites Families (301)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS53120687A (en) | 1977-03-30 | 1978-10-21 | Taki Chem Co Ltd | Hardener |
| JPS564505U (en) | 1979-06-21 | 1981-01-16 | ||
| US4769292A (en) | 1987-03-02 | 1988-09-06 | Eastman Kodak Company | Electroluminescent device with modified thin film luminescent zone |
| GB2239705B (en) | 1989-11-08 | 1993-05-12 | Nat Res Dev | Gas sensors and compounds suitable therefor |
| US5641878A (en) | 1991-05-15 | 1997-06-24 | Diatron Corporation | Porphyrin, azaporphyrin, and related fluorescent dyes free of aggregation and serum binding |
| US5707745A (en) | 1994-12-13 | 1998-01-13 | The Trustees Of Princeton University | Multicolor organic light emitting devices |
| US5844363A (en) | 1997-01-23 | 1998-12-01 | The Trustees Of Princeton Univ. | Vacuum deposited, non-polymeric flexible organic light emitting devices |
| US6303238B1 (en) | 1997-12-01 | 2001-10-16 | The Trustees Of Princeton University | OLEDs doped with phosphorescent compounds |
| JP4142782B2 (en) | 1998-06-26 | 2008-09-03 | Tdk株式会社 | Organic EL device |
| WO2000070655A2 (en) | 1999-05-13 | 2000-11-23 | The Trustees Of Princeton University | Very high efficiency organic light emitting devices based on electrophosphorescence |
| US6821645B2 (en) | 1999-12-27 | 2004-11-23 | Fuji Photo Film Co., Ltd. | Light-emitting material comprising orthometalated iridium complex, light-emitting device, high efficiency red light-emitting device, and novel iridium complex |
| JP2002010505A (en) | 2000-06-16 | 2002-01-11 | Fuji Electric Co Ltd | Charge control device |
| JP4154140B2 (en) | 2000-09-26 | 2008-09-24 | キヤノン株式会社 | Metal coordination compounds |
| JP4460743B2 (en) | 2000-09-29 | 2010-05-12 | 富士フイルム株式会社 | Method for producing iridium complex or tautomer thereof |
| WO2003015720A1 (en) | 2001-08-15 | 2003-02-27 | 3M Innovative Properties Company | Hardenable self-supporting structures and methods |
| JP4166455B2 (en) | 2001-10-01 | 2008-10-15 | 株式会社半導体エネルギー研究所 | Polarizing film and light emitting device |
| US20030186077A1 (en) | 2001-12-31 | 2003-10-02 | Chen Jian P. | Bis- and tris- (di) benzocarbazole-based materials as hole transport materials for organic light emitting devices |
| TW536924B (en) | 2002-02-22 | 2003-06-11 | E Ray Optoelectronics Technolo | Efficient organic electroluminescent device with new red fluorescent dopants |
| JP2003342284A (en) | 2002-05-30 | 2003-12-03 | Canon Inc | Metal coordination compound, light emitting element and display device |
| GB0215153D0 (en) | 2002-07-01 | 2002-08-07 | Univ Hull | Luminescent compositions |
| DE10238903A1 (en) | 2002-08-24 | 2004-03-04 | Covion Organic Semiconductors Gmbh | New heteroaromatic rhodium and iridium complexes, useful in electroluminescent and/or phosphorescent devices as the emission layer and for use in solar cells, photovoltaic devices and organic photodetectors |
| US20060094875A1 (en) | 2002-11-01 | 2006-05-04 | Hisanori Itoh | Platinum complexes |
| WO2004070655A2 (en) | 2003-02-04 | 2004-08-19 | Vanderbilt University | Apparatus and methods of determining marker orientation in fiducial registration |
| US7037599B2 (en) | 2003-02-28 | 2006-05-02 | Eastman Kodak Company | Organic light emitting diodes for production of polarized light |
| EP1606296B1 (en) | 2003-03-24 | 2009-08-05 | University Of Southern California | PHENYL-PYRAZOLE COMPLEXES OF Ir |
| US6998492B2 (en) | 2003-05-16 | 2006-02-14 | Semiconductor Energy Laboratory Co., Ltd. | Organometallic complex and light-emitting element containing the same |
| EP3623444B1 (en) | 2003-06-02 | 2021-05-26 | UDC Ireland Limited | Organic electroluminescent devices and metal complex compounds |
| JP4460952B2 (en) | 2003-06-02 | 2010-05-12 | 富士フイルム株式会社 | Organic electroluminescent device and complex compound |
| CN101667626B (en) | 2003-06-02 | 2012-11-28 | 富士胶片株式会社 | Organic electroluminescent devices and metal complex compounds |
| JP4498841B2 (en) | 2003-07-11 | 2010-07-07 | 三星電子株式会社 | GPS correlation peak signal search method and system therefor. |
| US6917159B2 (en) | 2003-08-14 | 2005-07-12 | Eastman Kodak Company | Microcavity OLED device |
| US7268485B2 (en) | 2003-10-07 | 2007-09-11 | Eastman Kodak Company | White-emitting microcavity OLED device |
| DE10350722A1 (en) | 2003-10-30 | 2005-05-25 | Covion Organic Semiconductors Gmbh | metal complexes |
| US7442797B2 (en) | 2003-11-04 | 2008-10-28 | Takasago International Corporation | Platinum complex and light emitting device |
| DE10357044A1 (en) | 2003-12-04 | 2005-07-14 | Novaled Gmbh | Process for doping organic semiconductors with quinonediimine derivatives |
| DE10359341A1 (en) | 2003-12-16 | 2005-07-28 | Basell Polyolefine Gmbh | New monocyclopentadienyl complex useful in catalyst system used in prepolymerized catalyst system and for the polymerization or copolymerization of olefins |
| US7332232B2 (en) | 2004-02-03 | 2008-02-19 | Universal Display Corporation | OLEDs utilizing multidentate ligand systems |
| JP2005267557A (en) | 2004-03-22 | 2005-09-29 | Ntt Docomo Inc | Server device |
| US20050211974A1 (en) | 2004-03-26 | 2005-09-29 | Thompson Mark E | Organic photosensitive devices |
| US7279704B2 (en) | 2004-05-18 | 2007-10-09 | The University Of Southern California | Complexes with tridentate ligands |
| WO2005113704A2 (en) | 2004-05-18 | 2005-12-01 | The University Of Southern California | Luminescent compounds with carbene ligands |
| US7445855B2 (en) | 2004-05-18 | 2008-11-04 | The University Of Southern California | Cationic metal-carbene complexes |
| US7393599B2 (en) | 2004-05-18 | 2008-07-01 | The University Of Southern California | Luminescent compounds with carbene ligands |
| JP4925569B2 (en) | 2004-07-08 | 2012-04-25 | ローム株式会社 | Organic electroluminescent device |
| KR20060011537A (en) | 2004-07-30 | 2006-02-03 | 주식회사 하이닉스반도체 | Device Separation Method of Semiconductor Devices |
| JP4576605B2 (en) | 2004-08-09 | 2010-11-10 | 独立行政法人産業技術総合研究所 | Identification method of oligosaccharide |
| US7300731B2 (en) | 2004-08-10 | 2007-11-27 | E.I. Du Pont De Nemours And Company | Spatially-doped charge transport layers |
| KR20060015371A (en) | 2004-08-14 | 2006-02-17 | 윤희찬 | Hybrid one-chip data recognition device |
| JP4500735B2 (en) | 2004-09-22 | 2010-07-14 | 富士フイルム株式会社 | Organic electroluminescence device |
| US7002013B1 (en) | 2004-09-23 | 2006-02-21 | National Tsing Hua University | Pt complexes as phosphorescent emitters in the fabrication of organic light emitting diodes |
| JP4531509B2 (en) | 2004-09-27 | 2010-08-25 | 富士フイルム株式会社 | Light emitting element |
| US7489074B2 (en) | 2004-09-28 | 2009-02-10 | Osram Opto Semiconductors Gmbh | Reducing or eliminating color change for microcavity OLED devices |
| CA2589711A1 (en) | 2004-12-23 | 2006-06-29 | Ciba Specialty Chemicals Holding Inc. | Electroluminescent metal complexes with nucleophilic carbene ligands |
| CN100348594C (en) | 2005-01-12 | 2007-11-14 | 武汉大学 | Bidentate ligand and its iridium complex and electroluminescent device therewith |
| WO2006081780A1 (en) | 2005-02-04 | 2006-08-10 | Novaled Ag | Dopants for organic semiconductors |
| JP4773109B2 (en) | 2005-02-28 | 2011-09-14 | 高砂香料工業株式会社 | Platinum complex and light emitting device |
| JP2006242080A (en) | 2005-03-02 | 2006-09-14 | Denso Corp | Abnormality diagnostic device for exhaust gas recirculating device |
| JP4425816B2 (en) | 2005-03-02 | 2010-03-03 | 富士重工業株式会社 | Electronically controlled throttle device |
| JP4399382B2 (en) | 2005-03-16 | 2010-01-13 | 富士フイルム株式会社 | Organic electroluminescence device |
| JP4399429B2 (en) | 2005-03-16 | 2010-01-13 | 富士フイルム株式会社 | Organic electroluminescence device |
| US7981524B2 (en) | 2005-03-16 | 2011-07-19 | Fujifilm Corporation | Platinum complex compound and organic electroluminescent device |
| JP4727262B2 (en) | 2005-03-16 | 2011-07-20 | 富士フイルム株式会社 | Organic electroluminescence device |
| US20090126796A1 (en) | 2005-04-07 | 2009-05-21 | The Regents Of The University Of California | Highly Efficient Polymer Solar Cell by Polymer Self-Organization |
| JP4790298B2 (en) | 2005-04-08 | 2011-10-12 | 日本放送協会 | Good solubility iridium complex and organic EL device |
| US9070884B2 (en) | 2005-04-13 | 2015-06-30 | Universal Display Corporation | Hybrid OLED having phosphorescent and fluorescent emitters |
| US7758971B2 (en) | 2005-04-25 | 2010-07-20 | Fujifilm Corporation | Organic electroluminescent device |
| JP4934346B2 (en) | 2005-04-25 | 2012-05-16 | 富士フイルム株式会社 | Organic electroluminescence device |
| TWI418606B (en) | 2005-04-25 | 2013-12-11 | Udc Ireland Ltd | Organic electroluminescent device |
| TWI391027B (en) | 2005-04-25 | 2013-03-21 | Fujifilm Corp | Organic electroluminescent device |
| JP2006303394A (en) | 2005-04-25 | 2006-11-02 | Fuji Photo Film Co Ltd | Organic electroluminescent element |
| JP5046548B2 (en) | 2005-04-25 | 2012-10-10 | 富士フイルム株式会社 | Organic electroluminescence device |
| JP4533796B2 (en) | 2005-05-06 | 2010-09-01 | 富士フイルム株式会社 | Organic electroluminescence device |
| EP1887640A4 (en) | 2005-05-24 | 2012-04-04 | Pioneer Corp | ORGANIC ELECTROLUMINESCENCE DEVICE |
| JP2006351638A (en) | 2005-06-13 | 2006-12-28 | Fujifilm Holdings Corp | Light emitting device |
| JP2007031678A (en) | 2005-07-29 | 2007-02-08 | Showa Denko Kk | Polymeric luminescent material and organic electroluminescence element using the polymeric luminescent material |
| JP2007042875A (en) | 2005-08-03 | 2007-02-15 | Fujifilm Holdings Corp | Organic electroluminescence element |
| JP4796802B2 (en) | 2005-08-15 | 2011-10-19 | 富士フイルム株式会社 | Organic electroluminescence device |
| JP4923478B2 (en) | 2005-08-19 | 2012-04-25 | コニカミノルタホールディングス株式会社 | Organic electroluminescence element, display device and lighting device |
| JP2007066581A (en) | 2005-08-29 | 2007-03-15 | Fujifilm Holdings Corp | Organic electroluminescent element |
| JP5151481B2 (en) | 2005-09-01 | 2013-02-27 | コニカミノルタホールディングス株式会社 | Organic electroluminescence element, display device and lighting device |
| JP2007073620A (en) | 2005-09-05 | 2007-03-22 | Fujifilm Corp | Organic electroluminescence device |
| WO2007029466A1 (en) | 2005-09-06 | 2007-03-15 | Konica Minolta Holdings, Inc. | Organic electroluminescent device, display and illuminating device |
| JP2007073845A (en) | 2005-09-08 | 2007-03-22 | Fujifilm Holdings Corp | Organic laser oscillator |
| JP5208391B2 (en) | 2005-09-09 | 2013-06-12 | 住友化学株式会社 | Metal complex, light emitting material and light emitting device |
| JP2007073900A (en) | 2005-09-09 | 2007-03-22 | Fujifilm Corp | Organic electroluminescence device |
| JP2007080593A (en) | 2005-09-12 | 2007-03-29 | Fujifilm Corp | Electrochemiluminescence device |
| JP2007080677A (en) | 2005-09-14 | 2007-03-29 | Fujifilm Corp | Organic electroluminescent device and manufacturing method thereof |
| JP2007110067A (en) | 2005-09-14 | 2007-04-26 | Fujifilm Corp | Composition for organic electroluminescent device, method for producing organic electroluminescent device, and organic electroluminescent device |
| US7839078B2 (en) | 2005-09-15 | 2010-11-23 | Fujifilm Corporation | Organic electroluminescent element having a luminescent layer and a buffer layer adjacent thereto |
| JP2007110102A (en) | 2005-09-15 | 2007-04-26 | Fujifilm Corp | Organic electroluminescence device |
| JP2007088105A (en) | 2005-09-20 | 2007-04-05 | Fujifilm Corp | Organic electroluminescence device |
| JP4789556B2 (en) | 2005-09-21 | 2011-10-12 | 富士フイルム株式会社 | Organic electroluminescence device |
| TWI268952B (en) | 2005-09-21 | 2006-12-21 | Au Optronics Corp | Spiro silane compound and organic electroluminescent device using the same |
| JP2007186490A (en) | 2005-12-14 | 2007-07-26 | Sumitomo Seika Chem Co Ltd | Compound for electroluminescent device and method for producing the same |
| EP1968131A4 (en) | 2005-12-27 | 2009-08-19 | Idemitsu Kosan Co | ORGANIC ELECTROLUMINESCENT DEVICE AND MATERIAL THEREFOR |
| WO2007080801A1 (en) | 2006-01-11 | 2007-07-19 | Idemitsu Kosan Co., Ltd. | Novel imide derivative, material for organic electroluminescent element, and organic electroluminescent element comprising the same |
| WO2007097149A1 (en) | 2006-02-20 | 2007-08-30 | Konica Minolta Holdings, Inc. | Organic electroluminescence element, white light emitting element, display device and illuminating device |
| US7854513B2 (en) | 2006-03-03 | 2010-12-21 | Quach Cang V | One-way transparent display systems |
| ATE394800T1 (en) | 2006-03-21 | 2008-05-15 | Novaled Ag | HETEROCYCLIC RADICAL OR DIRADICAL, THEIR DIMERS, OLIGOMERS, POLYMERS, DISPIR COMPOUNDS AND POLYCYCLES, THEIR USE, ORGANIC SEMICONDUCTIVE MATERIAL AND ELECTRONIC COMPONENT |
| JP4945156B2 (en) | 2006-03-24 | 2012-06-06 | 富士フイルム株式会社 | Organic electroluminescence device |
| KR100972895B1 (en) | 2006-04-20 | 2010-07-28 | 이데미쓰 고산 가부시키가이샤 | Organic light emitting device |
| JP5144034B2 (en) | 2006-05-31 | 2013-02-13 | 富士フイルム株式会社 | Organic electroluminescence device |
| TW200815446A (en) | 2006-06-05 | 2008-04-01 | Idemitsu Kosan Co | Organic electroluminescent device and material for organic electroluminescent device |
| JP2008010353A (en) | 2006-06-30 | 2008-01-17 | Seiko Epson Corp | Mask manufacturing method, wiring pattern manufacturing method, and plasma display manufacturing method |
| DE102006035018B4 (en) | 2006-07-28 | 2009-07-23 | Novaled Ag | Oxazole triplet emitter for OLED applications |
| JP2008037848A (en) | 2006-08-10 | 2008-02-21 | Takasago Internatl Corp | Platinum complex and light emitting device |
| JP5205584B2 (en) | 2006-09-06 | 2013-06-05 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device and display device |
| US7598381B2 (en) | 2006-09-11 | 2009-10-06 | The Trustees Of Princeton University | Near-infrared emitting organic compounds and organic devices using the same |
| JP5049711B2 (en) | 2006-09-27 | 2012-10-17 | 富士フイルム株式会社 | Organic electroluminescence device |
| JP2008109085A (en) | 2006-09-29 | 2008-05-08 | Fujifilm Corp | Organic electroluminescence device |
| JP2008103535A (en) | 2006-10-19 | 2008-05-01 | Takasago Internatl Corp | Light emitting element |
| JP2008108617A (en) | 2006-10-26 | 2008-05-08 | Fujifilm Corp | Organic electroluminescence device |
| US8945722B2 (en) | 2006-10-27 | 2015-02-03 | The University Of Southern California | Materials and architectures for efficient harvesting of singlet and triplet excitons for white light emitting OLEDs |
| JP2008117545A (en) | 2006-11-01 | 2008-05-22 | Nix Inc | Liquid transmission / reception joint device and fuel cell system including the same |
| JP4924878B2 (en) | 2006-11-06 | 2012-04-25 | 株式会社ニコン | Absolute encoder |
| JP4478166B2 (en) | 2006-11-09 | 2010-06-09 | 三星モバイルディスプレイ株式會社 | Organic light-emitting device provided with organic film containing organometallic complex |
| JP5187493B2 (en) | 2006-11-27 | 2013-04-24 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescent devices and novel indole derivatives |
| JP5282260B2 (en) | 2006-11-27 | 2013-09-04 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| JP2009076834A (en) | 2006-11-27 | 2009-04-09 | Fujifilm Corp | Organic electroluminescent devices and novel indole derivatives |
| JP2010512005A (en) | 2006-12-01 | 2010-04-15 | ザ リージェンツ オブ ザ ユニバーシティ オブ カリフォルニア | Improved performance characteristics of organic semiconductor films by improved solution processing methods |
| US8129494B2 (en) | 2006-12-26 | 2012-03-06 | Asahi Kasei E-Materials Corporation | Resin composition for printing plate |
| JP4833106B2 (en) | 2007-02-13 | 2011-12-07 | 富士フイルム株式会社 | Organic light emitting device |
| US8106199B2 (en) | 2007-02-13 | 2012-01-31 | Arizona Board Of Regents For And On Behalf Of Arizona State University | Organometallic materials for optical emission, optical absorption, and devices including organometallic materials |
| WO2008101842A1 (en) | 2007-02-23 | 2008-08-28 | Basf Se | Electroluminescent metal complexes with benzotriazoles |
| JP5081010B2 (en) | 2007-03-26 | 2012-11-21 | 富士フイルム株式会社 | Organic electroluminescence device |
| US20080241518A1 (en) | 2007-03-26 | 2008-10-02 | Tasuku Satou | Organic electroluminescence element |
| JP5230218B2 (en) | 2007-03-26 | 2013-07-10 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| WO2008117889A1 (en) | 2007-03-28 | 2008-10-02 | Fujifilm Corporation | Organic electroluminescent device |
| JP5430073B2 (en) | 2007-03-30 | 2014-02-26 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| US9006711B2 (en) | 2007-04-17 | 2015-04-14 | Konica Minolta Holdings, Inc. | White organic electroluminescent element and lighting device |
| EP1988079A1 (en) | 2007-04-25 | 2008-11-05 | Lonza Ag | Process for the preparation of optically active ethenylphenyl-alcohols |
| JP5084361B2 (en) | 2007-06-18 | 2012-11-28 | 株式会社リコー | Image forming apparatus |
| JP2009016579A (en) | 2007-07-04 | 2009-01-22 | Fujifilm Corp | Organic electroluminescence device and manufacturing method |
| DE102007031220B4 (en) | 2007-07-04 | 2022-04-28 | Novaled Gmbh | Quinoid compounds and their use in semiconducting matrix materials, electronic and optoelectronic components |
| JP2009016184A (en) | 2007-07-04 | 2009-01-22 | Fujifilm Corp | Organic electroluminescence device |
| US8372527B2 (en) | 2007-07-11 | 2013-02-12 | Idemitsu Kosan Co., Ltd. | Material for organic electroluminescent element, and organic electroluminescent element |
| GB2451106A (en) | 2007-07-18 | 2009-01-21 | Cis Bio Int | Lanthanide (III) ion complexing pyrazoyl-aza(thio)xanthone comprising compounds, their complexes and their use as fluorescent labels |
| EP2045848B1 (en) | 2007-07-18 | 2017-09-27 | Idemitsu Kosan Co., Ltd. | Organic electroluminescent device material and organic electroluminescent device |
| JP2010534394A (en) | 2007-07-25 | 2010-11-04 | ポリマーズ シーアールシー リミテッド | Solar cell and method for manufacturing the same |
| JP2009032989A (en) | 2007-07-27 | 2009-02-12 | Fujifilm Corp | Organic electroluminescence device |
| JP5255794B2 (en) | 2007-07-27 | 2013-08-07 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| JP2009055010A (en) | 2007-07-27 | 2009-03-12 | Fujifilm Corp | Organic electroluminescence device |
| JP5497259B2 (en) | 2007-07-27 | 2014-05-21 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| KR101508024B1 (en) | 2007-08-13 | 2015-04-06 | 유니버시티 오브 써던 캘리포니아 | Organic photosensitive optoelectronic devices with triplet harvesting |
| JP5119812B2 (en) | 2007-09-03 | 2013-01-16 | コニカミノルタホールディングス株式会社 | Organic electroluminescence element, display device and lighting device |
| KR101548382B1 (en) | 2007-09-14 | 2015-08-28 | 유디씨 아일랜드 리미티드 | Organic electroluminescence device |
| JP5014036B2 (en) | 2007-09-18 | 2012-08-29 | 富士フイルム株式会社 | Organic electroluminescence device |
| GB0718577D0 (en) | 2007-09-24 | 2007-10-31 | Acal Energy Ltd | Fuel cells |
| JP5438941B2 (en) | 2007-09-25 | 2014-03-12 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| US7862908B2 (en) | 2007-11-26 | 2011-01-04 | National Tsing Hua University | Conjugated compounds containing hydroindoloacridine structural elements, and their use |
| KR100905951B1 (en) | 2007-12-06 | 2009-07-06 | 주식회사 잉크테크 | Iridium-based compounds having carbazole pyridine and phenyl derivatives as main ligands and organic electroluminescent devices comprising the same |
| JP5438955B2 (en) | 2007-12-14 | 2014-03-12 | ユー・ディー・シー アイルランド リミテッド | Platinum complex compound and organic electroluminescence device using the same |
| WO2009086209A2 (en) | 2007-12-21 | 2009-07-09 | Arizona Board Of Regents For And On Behalf Of Arizona State University | Platinum(ii) di(2-pyrazolyl)benzene chloride analogs and uses |
| US9293720B2 (en) | 2008-02-19 | 2016-03-22 | New Jersey Institute Of Technology | Carbon nanotubes as charge carriers in organic and hybrid solar cells |
| JP5243972B2 (en) | 2008-02-28 | 2013-07-24 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| WO2009111299A2 (en) | 2008-02-29 | 2009-09-11 | Arizona Board Of Regents For And On Behalf Of Arizona State University | Tridentate platinum (ii) complexes |
| JP2009211892A (en) | 2008-03-03 | 2009-09-17 | Fujifilm Corp | Organic electroluminescent element |
| DE102008015526B4 (en) | 2008-03-25 | 2021-11-11 | Merck Patent Gmbh | Metal complexes |
| JP5228578B2 (en) | 2008-03-31 | 2013-07-03 | 株式会社ジェイテクト | Motor control device and electric power steering device |
| JP4531836B2 (en) | 2008-04-22 | 2010-08-25 | 富士フイルム株式会社 | Organic electroluminescent device, novel platinum complex compound and novel compound that can be a ligand |
| JP2009266943A (en) | 2008-04-23 | 2009-11-12 | Fujifilm Corp | Organic field light-emitting element |
| JP4531842B2 (en) | 2008-04-24 | 2010-08-25 | 富士フイルム株式会社 | Organic electroluminescence device |
| JP2009267171A (en) | 2008-04-25 | 2009-11-12 | Fujifilm Corp | Organic electric field light emitting element |
| JP2009267244A (en) | 2008-04-28 | 2009-11-12 | Fujifilm Corp | Organic electroluminescent element |
| JP2009272339A (en) | 2008-04-30 | 2009-11-19 | Fujifilm Corp | Organic electric field light-emitting element |
| JP2011528024A (en) | 2008-07-16 | 2011-11-10 | ソルヴェイ(ソシエテ アノニム) | Luminescent materials containing polynuclear complexes |
| EP2327116A1 (en) | 2008-08-20 | 2011-06-01 | Plextronics, Inc. | Improved solvent system for fabrication of organic solar cells |
| US10105875B2 (en) | 2008-08-21 | 2018-10-23 | Cam Holding Corporation | Enhanced surfaces, coatings, and related methods |
| US7635792B1 (en) | 2008-10-14 | 2009-12-22 | General Electric Company | 2,5-linked polyfluorenes for optoelectronic devices |
| EP2362889B1 (en) | 2008-11-11 | 2018-12-26 | Universal Display Corporation | Phosphorescent emitters |
| JP5507185B2 (en) | 2008-11-13 | 2014-05-28 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| WO2010059240A1 (en) | 2008-11-21 | 2010-05-27 | Plextronics, Inc. | Doped interfacial modification layers for stability enhancement for bulk heterojunction organic solar cells |
| JP5497284B2 (en) | 2008-12-08 | 2014-05-21 | ユー・ディー・シー アイルランド リミテッド | White organic electroluminescence device |
| US8420234B2 (en) | 2009-01-06 | 2013-04-16 | Udc Ireland Limited | Organic electroluminescent device |
| JP5627883B2 (en) | 2009-01-07 | 2014-11-19 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| JP5210187B2 (en) | 2009-01-22 | 2013-06-12 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| KR101066743B1 (en) | 2009-02-13 | 2011-09-21 | 부산대학교 산학협력단 | Iridium complex and organic light emitting device comprising the same |
| US8469401B2 (en) | 2009-02-23 | 2013-06-25 | Amsafe, Inc. | Seat harness pretensioner |
| US20120108806A1 (en) | 2009-03-12 | 2012-05-03 | Jian Li | Azaporphyrins and applications thereof |
| JP5644143B2 (en) | 2009-03-25 | 2014-12-24 | 住友化学株式会社 | Coating method and manufacturing method of organic electroluminescence element |
| JP5604505B2 (en) | 2009-04-06 | 2014-10-08 | アリゾナ ボード オブ リージェンツ アクティング フォー アンド オン ビハーフ オブ アリゾナ ステイト ユニバーシティ | Synthesis of four-coordinate platinum complexes and their application to light-emitting devices |
| US8804682B2 (en) | 2009-04-17 | 2014-08-12 | Panasonic Intellectual Property Corporation Of America | Apparatus for management of local IP access in a segmented mobile communication system |
| US8603642B2 (en) | 2009-05-13 | 2013-12-10 | Global Oled Technology Llc | Internal connector for organic electronic devices |
| KR101763954B1 (en) | 2009-05-21 | 2017-08-01 | 라이너지 테크 인코포레이션 | Conjugated polymers and their use in optoelectronic devices |
| JP5456554B2 (en) | 2009-06-01 | 2014-04-02 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| JP2010185068A (en) | 2009-08-31 | 2010-08-26 | Fujifilm Corp | Organic electroluminescent device |
| JP5770441B2 (en) | 2009-09-30 | 2015-08-26 | ユー・ディー・シー アイルランド リミテッド | Material for organic electroluminescence device and organic electroluminescence device |
| DE102009048791A1 (en) | 2009-10-08 | 2011-04-14 | Merck Patent Gmbh | Materials for organic electroluminescent devices |
| WO2011045337A1 (en) | 2009-10-14 | 2011-04-21 | Basf Se | Dinuclear platinum-carbene complexes and the use thereof in oleds |
| WO2011052570A1 (en) | 2009-10-30 | 2011-05-05 | 住友化学株式会社 | Organic photoelectric conversion element and process for production thereof |
| KR20190057162A (en) | 2009-11-06 | 2019-05-27 | 나노-씨, 인크. | Fullerene-functionalized particles, methods for making the same and their use in blukheterojunction organic photovoltaic devices |
| US20120326103A1 (en) | 2009-11-27 | 2012-12-27 | Shuk Kwan Mak | Functionalized triplet emitters for electro-luminescent devices |
| JP5495746B2 (en) | 2009-12-08 | 2014-05-21 | キヤノン株式会社 | Novel iridium complex and organic light emitting device having the same |
| WO2011076325A1 (en) | 2009-12-23 | 2011-06-30 | Merck Patent Gmbh | Compositions comprising polymeric binders |
| DE102010005463A1 (en) | 2010-01-20 | 2011-07-21 | cynora GmbH, 76344 | Singlet Harvesting Blue Light Emitter for use in OLEDs and other organic electronic devices |
| KR20130067276A (en) | 2010-04-30 | 2013-06-21 | 아리조나 보드 오브 리젠츠 퍼 앤 온 비하프 오브 아리조나 스테이트 유니버시티 | Synthesis of four coordinated palladium complexes and their applications in light emitting devices thereof |
| US9324957B2 (en) | 2010-04-30 | 2016-04-26 | Arizona Board Of Regents On Behalf Of Arizona State University | Synthesis of four coordinated gold complexes and their applications in light emitting devices thereof |
| JP5707818B2 (en) | 2010-09-28 | 2015-04-30 | コニカミノルタ株式会社 | Material for organic electroluminescence element, organic electroluminescence element, display element, lighting device and metal complex compound |
| JP5973692B2 (en) | 2010-09-30 | 2016-08-23 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| JP5627979B2 (en) | 2010-09-30 | 2014-11-19 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescence device |
| US20140147996A1 (en) | 2010-11-29 | 2014-05-29 | Arizon Board of Regents Acting for and on Behalf Arizona State University | Methods for fabricating bulk heterojunctions using solution processing techniques |
| DE102010054893A1 (en) | 2010-12-17 | 2012-06-21 | Osram Opto Semiconductors Gmbh | Radiation-emitting organic-electronic device and method for its production |
| US8816080B2 (en) | 2011-02-18 | 2014-08-26 | Arizona Board Of Regents Acting For And On Behalf Of Arizona State University | Four coordinated platinum and palladium complexes with geometrically distorted charge transfer state and their applications in light emitting devices |
| TWI560191B (en) | 2011-02-23 | 2016-12-01 | Universal Display Corp | Novel tetradentate platinum complexes |
| JP5794813B2 (en) | 2011-04-12 | 2015-10-14 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescent element, organic electroluminescent element material, film, and method for producing organic electroluminescent element |
| JP6014350B2 (en) | 2011-04-12 | 2016-10-25 | ユー・ディー・シー アイルランド リミテッド | Organic electroluminescent device, organic electroluminescent device material, film, light emitting layer, and organic electroluminescent device manufacturing method |
| TWI558713B (en) | 2011-04-14 | 2016-11-21 | 美國亞利桑那州立大學董事會 | Pyridine-oxyphenyl coordinated iridium (iii) complexes and methods of making and using |
| US9238668B2 (en) | 2011-05-26 | 2016-01-19 | Arizona Board Of Regents, Acting For And On Behalf Of Arizona State University | Synthesis of platinum and palladium complexes as narrow-band phosphorescent emitters for full color displays |
| KR101972184B1 (en) | 2011-06-03 | 2019-04-24 | 메르크 파텐트 게엠베하 | Metal complexes |
| US9783564B2 (en) | 2011-07-25 | 2017-10-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| JP6234666B2 (en) | 2011-07-25 | 2017-11-22 | ユニバーサル ディスプレイ コーポレイション | Tetradentate platinum complex |
| US9493698B2 (en) | 2011-08-31 | 2016-11-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| KR101897044B1 (en) | 2011-10-20 | 2018-10-23 | 에스에프씨 주식회사 | Organic metal compounds and organic light emitting diodes comprising the same |
| US9461254B2 (en) | 2012-01-03 | 2016-10-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US8987451B2 (en) | 2012-01-03 | 2015-03-24 | Universal Display Corporation | Synthesis of cyclometallated platinum(II) complexes |
| US9705099B2 (en) | 2012-01-26 | 2017-07-11 | Universal Display Corporation | Phosphorescent organic light emitting devices having a hole transporting cohost material in the emissive region |
| JP5978843B2 (en) | 2012-02-02 | 2016-08-24 | コニカミノルタ株式会社 | Iridium complex compound, organic electroluminescence device material, organic electroluminescence device, lighting device and display device |
| WO2013130483A1 (en) | 2012-02-27 | 2013-09-06 | Jian Li | Microcavity oled device with narrow band phosphorescent emitters |
| DE102012205945A1 (en) | 2012-04-12 | 2013-10-17 | Siemens Aktiengesellschaft | Organic super donors with at least two coupled carbene groups and their use as n-dopants |
| JP6158542B2 (en) | 2012-04-13 | 2017-07-05 | 株式会社半導体エネルギー研究所 | LIGHT EMITTING ELEMENT, LIGHT EMITTING DEVICE, ELECTRONIC DEVICE, AND LIGHTING DEVICE |
| JP6076153B2 (en) | 2012-04-20 | 2017-02-08 | 株式会社半導体エネルギー研究所 | LIGHT EMITTING ELEMENT, LIGHT EMITTING DEVICE, DISPLAY DEVICE, ELECTRONIC DEVICE, AND LIGHTING DEVICE |
| CN202549937U (en) | 2012-05-10 | 2012-11-21 | 京东方科技集团股份有限公司 | Organic light-emitting diode (OLED) display structure and OLED display device |
| DE102012209523A1 (en) | 2012-06-06 | 2013-12-12 | Osram Opto Semiconductors Gmbh | Main group metal complexes as p-dopants for organic electronic matrix materials |
| KR101338250B1 (en) | 2012-06-07 | 2013-12-09 | 삼성디스플레이 주식회사 | Display device |
| EP2684932B8 (en) | 2012-07-09 | 2016-12-21 | Hodogaya Chemical Co., Ltd. | Diarylamino matrix material doped with a mesomeric radialene compound |
| GB201213392D0 (en) | 2012-07-27 | 2012-09-12 | Imp Innovations Ltd | Electroluminescent compositions |
| KR101947815B1 (en) | 2012-08-07 | 2019-02-14 | 한국전자통신연구원 | The dual display device with the vertical structure |
| US9312502B2 (en) | 2012-08-10 | 2016-04-12 | Arizona Board Of Regents Acting For And On Behalf Of Arizona State University | Iridium complexes demonstrating broadband emission through controlled geometric distortion and applications thereof |
| WO2014031977A1 (en) | 2012-08-24 | 2014-02-27 | Arizona Board Of Regents For And On Behalf Of Arizona State University | Metal compounds and methods and uses thereof |
| US9312505B2 (en) | 2012-09-25 | 2016-04-12 | Universal Display Corporation | Organic electroluminescent materials and devices |
| KR102145982B1 (en) | 2012-10-24 | 2020-08-19 | 엘지디스플레이 주식회사 | Method for mnufacturing of blue phosphorescence composition and organic light emittin diode comprising the same |
| WO2014109814A2 (en) | 2012-10-26 | 2014-07-17 | Arizona Board Of Regents Acting For And On Behalf Of Arizona State University | Metal complexes, methods, and uses thereof |
| KR102017743B1 (en) | 2013-01-04 | 2019-09-04 | 삼성디스플레이 주식회사 | Organic light-emitting device having improved efficiency characterisitics and organic light-emitting display apparatus including the same |
| KR102120894B1 (en) | 2013-05-03 | 2020-06-10 | 삼성디스플레이 주식회사 | Organic light emitting device |
| WO2015027060A1 (en) | 2013-08-21 | 2015-02-26 | Arizona Board Of Regents On Behalf Of Arizona State University | Phosphorescent tetradentate metal complexes having modified emission spectra |
| KR102349659B1 (en) | 2013-06-10 | 2022-01-11 | 아리조나 보드 오브 리젠츠 온 비하프 오브 아리조나 스테이트 유니버시티 | Phosphorescent tetradentate metal complexes having modified emission spectra |
| JPWO2014208271A1 (en) | 2013-06-28 | 2017-02-23 | コニカミノルタ株式会社 | ORGANIC ELECTROLUMINESCENT ELEMENT, ITS MANUFACTURING METHOD, AND ORGANIC ELECTROLUMINESCENT DEVICE |
| KR102239408B1 (en) | 2013-07-02 | 2021-04-12 | 메르크 파텐트 게엠베하 | Polycyclic compounds |
| US9735378B2 (en) | 2013-09-09 | 2017-08-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| JP6804823B2 (en) | 2013-10-14 | 2020-12-23 | アリゾナ・ボード・オブ・リージェンツ・オン・ビハーフ・オブ・アリゾナ・ステイト・ユニバーシティーArizona Board of Regents on behalf of Arizona State University | Platinum complex and device |
| CN104576934A (en) | 2013-10-16 | 2015-04-29 | 海洋王照明科技股份有限公司 | White-light OLED (organic light emission diode) device and preparation method thereof |
| US9224963B2 (en) | 2013-12-09 | 2015-12-29 | Arizona Board Of Regents On Behalf Of Arizona State University | Stable emitters |
| US9666822B2 (en) | 2013-12-17 | 2017-05-30 | The Regents Of The University Of Michigan | Extended OLED operational lifetime through phosphorescent dopant profile management |
| US10020455B2 (en) | 2014-01-07 | 2018-07-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum and palladium complex emitters containing phenyl-pyrazole and its analogues |
| WO2015131158A1 (en) | 2014-02-28 | 2015-09-03 | Arizona Board Of Regents On Behalf Of Arizona State University | Chiral metal complexes as emitters for organic polarized electroluminescent devices |
| WO2015171627A1 (en) | 2014-05-08 | 2015-11-12 | Universal Display Corporation | Stabilized imidazophenanthridine materials |
| US9941479B2 (en) | 2014-06-02 | 2018-04-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate cyclometalated platinum complexes containing 9,10-dihydroacridine and its analogues |
| US9911931B2 (en) | 2014-06-26 | 2018-03-06 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9923155B2 (en) | 2014-07-24 | 2018-03-20 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum (II) complexes cyclometalated with functionalized phenyl carbene ligands and their analogues |
| US9502671B2 (en) | 2014-07-28 | 2016-11-22 | Arizona Board Of Regents On Behalf Of Arizona State University | Tridentate cyclometalated metal complexes with six-membered coordination rings |
| US9818959B2 (en) | 2014-07-29 | 2017-11-14 | Arizona Board of Regents on behlaf of Arizona State University | Metal-assisted delayed fluorescent emitters containing tridentate ligands |
| US10793546B2 (en) | 2014-08-15 | 2020-10-06 | Arizona Board Of Regents On Behalf Of Arizona State University | Non-platinum metal complexes for excimer based single dopant white organic light emitting diodes |
| US9920242B2 (en) | 2014-08-22 | 2018-03-20 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal-assisted delayed fluorescent materials as co-host materials for fluorescent OLEDs |
| US11329244B2 (en) | 2014-08-22 | 2022-05-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Organic light-emitting diodes with fluorescent and phosphorescent emitters |
| WO2016044324A1 (en) | 2014-09-15 | 2016-03-24 | Arizona Board Of Regents For And On Behalf Of Arizona State University | Ionic liquid catholytes and electrochemical devices containing same |
| US9865825B2 (en) | 2014-11-10 | 2018-01-09 | Arizona Board Of Regents On Behalf Of Arizona State University | Emitters based on octahedral metal complexes |
| US10033003B2 (en) | 2014-11-10 | 2018-07-24 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate metal complexes with carbon group bridging ligands |
| CN104377231B (en) | 2014-12-03 | 2019-12-31 | 京东方科技集团股份有限公司 | A double-sided OLED display panel and display device |
| JPWO2016088354A1 (en) | 2014-12-05 | 2017-09-07 | 出光興産株式会社 | Metal complex compound, material for organic electroluminescence device, composition, organic electroluminescence device and electronic device |
| KR101604339B1 (en) | 2014-12-09 | 2016-03-18 | 엘지전자 주식회사 | Light conversion film, baclight unit and display devive comprising the same |
| US9450195B2 (en) * | 2014-12-17 | 2016-09-20 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US9406892B2 (en) | 2015-01-07 | 2016-08-02 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10144867B2 (en) | 2015-02-13 | 2018-12-04 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US10870796B2 (en) | 2015-02-18 | 2020-12-22 | Cambridge Display Technology Limited | Organic light emitting polymer comprising light-emitting repeat unit in backbone of polymer and device therewith |
| GB201511300D0 (en) | 2015-06-26 | 2015-08-12 | Cambridge Display Tech Ltd | Metal complex and organic light-emitting device |
| US9711739B2 (en) | 2015-06-02 | 2017-07-18 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate metal complexes containing indoloacridine and its analogues |
| US9879039B2 (en) | 2015-06-03 | 2018-01-30 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate and octahedral metal complexes containing naphthyridinocarbazole and its analogues |
| US11930662B2 (en) | 2015-06-04 | 2024-03-12 | Arizona Board Of Regents On Behalf Of Arizona State University | Transparent electroluminescent devices with controlled one-side emissive displays |
| US20190221757A1 (en) | 2015-06-26 | 2019-07-18 | Cambridge Display Technology Limited | Metal complex and organic light-emitting device |
| US10158091B2 (en) | 2015-08-04 | 2018-12-18 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum (II) and palladium (II) complexes, devices, and uses thereof |
| US10211411B2 (en) | 2015-08-25 | 2019-02-19 | Arizona Board Of Regents On Behalf Of Arizona State University | Thermally activated delayed fluorescent material based on 9,10-dihydro-9,9-dimethylacridine analogues for prolonging device longevity |
| US20180353771A1 (en) | 2015-12-03 | 2018-12-13 | Sabic Global Technologies B.V. | Flexible phototherapy device for wound treatment |
| CN105609656B (en) | 2016-01-06 | 2017-05-17 | 京东方科技集团股份有限公司 | Organic light-emitting diode (OLED) and display device |
| US11335865B2 (en) | 2016-04-15 | 2022-05-17 | Arizona Board Of Regents On Behalf Of Arizona State University | OLED with multi-emissive material layer |
| JP6807178B2 (en) | 2016-07-07 | 2021-01-06 | 株式会社ジャパンディスプレイ | Display device, manufacturing method of display device |
| US10177323B2 (en) | 2016-08-22 | 2019-01-08 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum (II) and palladium (II) complexes and octahedral iridium complexes employing azepine functional groups and their analogues |
| CN107799658B (en) | 2016-08-29 | 2021-05-28 | 株式会社半导体能源研究所 | Light-emitting element, light-emitting device, electronic equipment, lighting device and organometallic complex |
| US10822363B2 (en) | 2016-10-12 | 2020-11-03 | Arizona Board Of Regents On Behalf Of Arizona State University | Narrow band red phosphorescent tetradentate platinum (II) complexes |
| US11183670B2 (en) | 2016-12-16 | 2021-11-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Organic light emitting diode with split emissive layer |
| CN106783922A (en) | 2016-12-26 | 2017-05-31 | 武汉华星光电技术有限公司 | Oled display |
| US10804475B2 (en) | 2017-01-11 | 2020-10-13 | Universal Display Corporation | Organic electroluminescent materials and devices |
| WO2018140765A1 (en) | 2017-01-27 | 2018-08-02 | Jian Li | Metal-assisted delayed fluorescent emitters employing pyrido-pyrrolo-acridine and analogues |
| US11101435B2 (en) | 2017-05-19 | 2021-08-24 | Arizona Board Of Regents On Behalf Of Arizona State University | Tetradentate platinum and palladium complexes based on biscarbazole and analogues |
| US10615349B2 (en) | 2017-05-19 | 2020-04-07 | Arizona Board Of Regents On Behalf Of Arizona State University | Donor-acceptor type thermally activated delayed fluorescent materials based on imidazo[1,2-F]phenanthridine and analogues |
| US10392387B2 (en) | 2017-05-19 | 2019-08-27 | Arizona Board Of Regents On Behalf Of Arizona State University | Substituted benzo[4,5]imidazo[1,2-a]phenanthro[9,10-c][1,8]naphthyridines, benzo[4,5]imidazo[1,2-a]phenanthro[9,10-c][1,5]naphthyridines and dibenzo[f,h]benzo[4,5]imidazo[2,1-a]pyrazino[2,3-c]isoquinolines as thermally assisted delayed fluorescent materials |
| US10516117B2 (en) | 2017-05-19 | 2019-12-24 | Arizona Board Of Regents On Behalf Of Arizona State University | Metal-assisted delayed fluorescent emttters employing benzo-imidazo-phenanthridine and analogues |
| US11802136B2 (en) | 2017-06-23 | 2023-10-31 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US11725022B2 (en) | 2017-06-23 | 2023-08-15 | Universal Display Corporation | Organic electroluminescent materials and devices |
| KR102504132B1 (en) | 2017-08-21 | 2023-02-28 | 삼성디스플레이 주식회사 | Cyclometallic compound, organic light emitting device comprising the same and emitting apparatus comprising the organic light emitting device |
| US11647643B2 (en) | 2017-10-17 | 2023-05-09 | Arizona Board Of Regents On Behalf Of Arizona State University | Hole-blocking materials for organic light emitting diodes |
| US20200287153A1 (en) | 2017-10-17 | 2020-09-10 | Arizona Board Of Regents On Behalf Of Arizona State University | Single-doped white oleds with extraction layer doped with down-conversion red emitters |
| KR102718677B1 (en) | 2017-10-17 | 2024-10-16 | 지안 리 | Phosphorescent excimers with desirable molecular orientation as monochromatic emitters for display and lighting applications |
| US12037348B2 (en) | 2018-03-09 | 2024-07-16 | Arizona Board Of Regents On Behalf Of Arizona State University | Blue and narrow band green and red emitting metal complexes |
| WO2019236541A1 (en) | 2018-06-04 | 2019-12-12 | Jian Li | Color tunable hybrid led-oled illumination devices |
| WO2020018476A1 (en) | 2018-07-16 | 2020-01-23 | Jian Li | Fluorinated porphyrin derivatives for optoelectronic applications |
| US11476430B2 (en) | 2018-10-15 | 2022-10-18 | Universal Display Corporation | Organic electroluminescent materials and devices |
| KR102738039B1 (en) | 2018-11-23 | 2024-12-06 | 삼성디스플레이 주식회사 | Organometallic compound and organic light-emitting device including the same |
| US11878988B2 (en) | 2019-01-24 | 2024-01-23 | Arizona Board Of Regents On Behalf Of Arizona State University | Blue phosphorescent emitters employing functionalized imidazophenthridine and analogues |
| US11594691B2 (en) | 2019-01-25 | 2023-02-28 | Arizona Board Of Regents On Behalf Of Arizona State University | Light outcoupling efficiency of phosphorescent OLEDs by mixing horizontally aligned fluorescent emitters |
| US12084465B2 (en) | 2020-02-24 | 2024-09-10 | Universal Display Corporation | Organic electroluminescent materials and devices |
| US20210376260A1 (en) | 2020-06-02 | 2021-12-02 | Arizona Board Of Regents On Behalf Of Arizona State University | Efficient and stable near-infrared oled employing metal complex aggregates as host materials |
-
2015
- 2015-07-22 US US14/805,691 patent/US9923155B2/en active Active
- 2015-07-23 KR KR1020150104260A patent/KR102433486B1/en active Active
- 2015-07-24 CN CN201510441320.5A patent/CN105367605B/en active Active
- 2015-07-24 CN CN202010883430.8A patent/CN112159432B/en not_active Expired - Fee Related
-
2018
- 2018-03-19 US US15/925,084 patent/US10886478B2/en active Active
-
2020
- 2020-12-03 US US17/110,353 patent/US12082486B2/en active Active
-
2022
- 2022-08-09 KR KR1020220099370A patent/KR102764844B1/en active Active
-
2024
- 2024-08-30 US US18/821,041 patent/US20240431196A1/en active Pending
Patent Citations (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20130341600A1 (en) * | 2012-06-21 | 2013-12-26 | Universal Display Corporation | Phosphorescent emitters |
| US20140014922A1 (en) * | 2012-07-10 | 2014-01-16 | Universal Display Corporation | Phosphorescent emitters containing dibenzo[1,4]azaborinine structure |
| US20140027733A1 (en) * | 2012-07-19 | 2014-01-30 | Universal Display Corporation | Transition metal complexes containing substituted imidazole carbene as ligands and their application in oleds |
| WO2014047616A1 (en) * | 2012-09-24 | 2014-03-27 | Arizona Board Of Regents For And On Behalf Of Arizona State University | Metal compounds, methods, and uses thereof |
Non-Patent Citations (1)
| Title |
|---|
| Effi cient and Stable White Organic Light-Emitting Diodes Employing a Single Emitter;Guijie Li等;《Adv. Mater.》;20140125;第26卷;第2931-2936页 * |
Also Published As
| Publication number | Publication date |
|---|---|
| US20240431196A1 (en) | 2024-12-26 |
| US10886478B2 (en) | 2021-01-05 |
| US12082486B2 (en) | 2024-09-03 |
| US20180219161A1 (en) | 2018-08-02 |
| KR102433486B1 (en) | 2022-08-17 |
| CN105367605A (en) | 2016-03-02 |
| KR102764844B1 (en) | 2025-02-06 |
| KR20160012941A (en) | 2016-02-03 |
| KR20220115904A (en) | 2022-08-19 |
| CN112159432A (en) | 2021-01-01 |
| US20160028028A1 (en) | 2016-01-28 |
| US20210091316A1 (en) | 2021-03-25 |
| CN112159432B (en) | 2024-04-02 |
| US9923155B2 (en) | 2018-03-20 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN105367605B (en) | Tetradentate platinum(II) complexes and their analogs cyclometallated with functionalized phenylcarbene ligands | |
| US10964897B2 (en) | Tridentate cyclometalated metal complexes with six-membered coordination rings | |
| KR102904935B1 (en) | Platinum complexes, devices, and uses thereof | |
| US20230015063A1 (en) | Metal-assisted delayed fluorescent emitters containing tridentate ligands | |
| WO2018140765A1 (en) | Metal-assisted delayed fluorescent emitters employing pyrido-pyrrolo-acridine and analogues | |
| JP6427623B2 (en) | Organic light emitting material | |
| CN110240606A (en) | Blue- and narrow-band green- and red-emitting metal complexes, and organic light-emitting diodes and light-emitting devices incorporating same | |
| JP2019065289A (en) | Donor-Acceptor Compound Comprising Nitrogen-Containing Polycyclic Aromatic as an Acceptor | |
| CN108840886A (en) | Platinum complex blue light material and organic light-emitting device | |
| WO2016025921A1 (en) | Non-platinum metal complexes for excimer based single dopant white organic light emitting diodes | |
| US10246475B2 (en) | Multidentate dinuclear cyclometallated platinum complexes containing N-(pyrimidin-2-yl)-carbazole and its analogues | |
| JP2020186236A (en) | Organic luminescent material containing 3-deuterium-substituted isoquinoline ligand | |
| KR102329178B1 (en) | A silicon-containing electron transporting material and its application | |
| Jung et al. | Deep‐red light‐emitting phosphorescent dendrimer encapsulated tris‐[2‐benzo [b] thiophen‐2‐yl‐pyridyl] iridium (III) core for light‐emitting device applications | |
| KR102840831B1 (en) | Organic electroluminescent material and device thereof | |
| KR102776330B1 (en) | Light-emitting material with a polycyclic ligand |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |