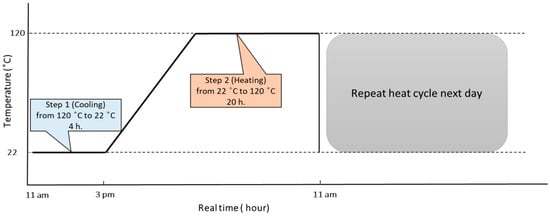
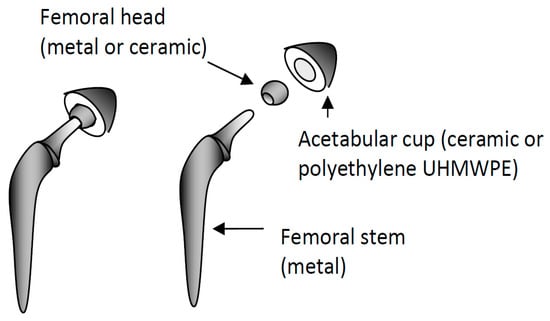
<p>Differential thermogram of corrosion product collected from the dark-grey areas of Mg anode (1), monitoring of temperature (2) and volume of HE (3). The released hydrogen was identified separately by gas chromatography [<a href="#B40-cmd-05-00015" class="html-bibr">40</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 2
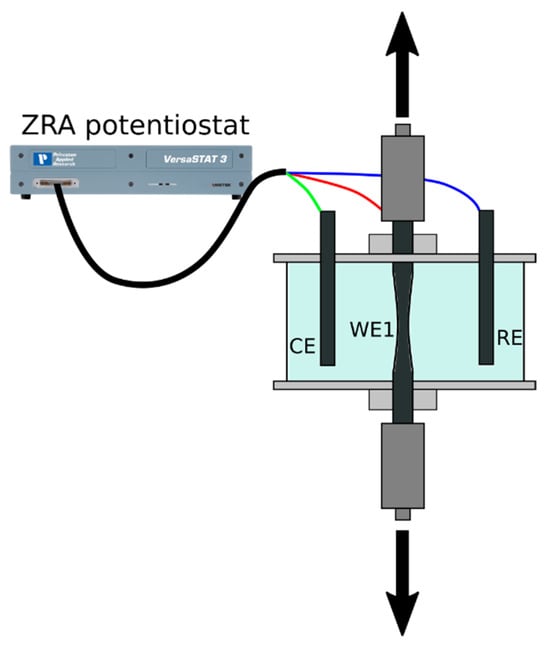
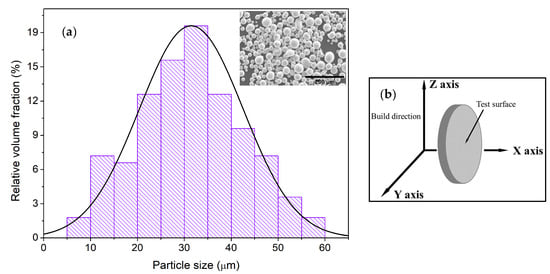
<p>(<b>a</b>) The setup of the electrochemical cell for HE rate measurement, RE-Ag/AgCl reference electrode, CE–Pt counter electrodes, RE-working Mg (99.92%) electrode. (<b>b</b>) monitoring of the volume of released gaseous hydrogen (1), the volume of hydrogen stored in the surface hydride phase (2) at galvanostatic anodic polarization by 8.2 A/m<sup>2</sup> in 0.1 M NaCl aqueous electrolyte. (<b>c</b>) influence of the anodic current density on the volume of hydride hydrogen, after 3.5 h of anodic polarization (8.2 A/m<sup>2</sup>) in 0.1 M NaCl aqueous electrolyte. The volume of hydrogen stored in the surface product was determined by treatment of the Mg electrode in chromic acid–silver nitrate solution [<a href="#B40-cmd-05-00015" class="html-bibr">40</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 3
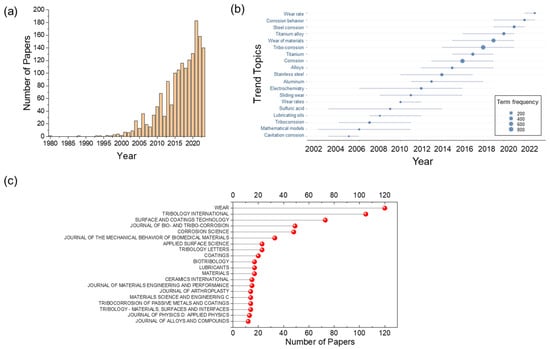
<p>(<b>a</b>) Anodic polarization curves of Mg in 1 M NaOH aqueous electrolyte (potential sweep rate 1 mV/sec). The electrode was preliminary polarized in 0.1 M NaCl at 8 A/m<sup>2</sup> during 5 min (1), 2 h (2) and 4 h (3); (<b>b</b>) Anodic oxidation of Mg with the surface phase at −1.2 V (SHE) in 1 M NaOH electrolyte after preliminary free corrosion 24 h in 0.1 M NaCl aqueous electrolyte (1) and anodic polarization (2) of Mg at 8 A/m<sup>2</sup> in 0.1 M NaCl, pH 10.2. (<b>c</b>) Thermal ion mass spectra of H<sub>2</sub><sup>+</sup> ions of the grey corrosion product [<a href="#B43-cmd-05-00015" class="html-bibr">43</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 4
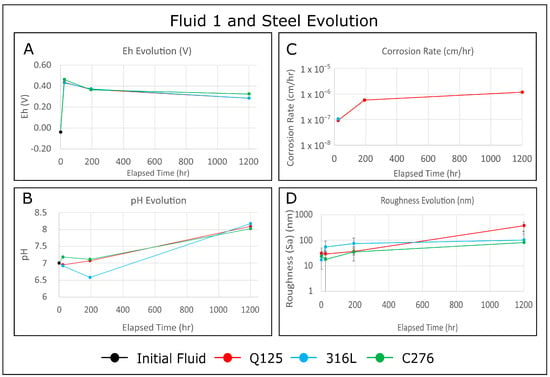
<p>(<b>a</b>) Monitoring of anodic current (1), the current of HE (2), and the surface concentration of hydride ions (3) at the fixed potential −1.25 V (SHE) in 0.1 M NaCl, pH 10.2; (<b>b</b>) Anodic (1), HE (2), and hydride formation (3) current densities vs. the potential of Mg anode. The amount of hydride ions (4) at the anode surface was measured after passing 150 coulombs of anodic electricity [<a href="#B50-cmd-05-00015" class="html-bibr">50</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 5
<p>The current densities of HE (1–3) and hydride formation (1′–3′) vs. density of galvanostatic anodic current at the different concentrations of NaCl 0.01 M (1,1′), 0.1 M (2,2′), and 5 M (3,3′) (pH 10.2) [<a href="#B50-cmd-05-00015" class="html-bibr">50</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 6
<p>(<b>a</b>) The dependence of the self-corrosion (<b>a</b>) and hydride formation (<b>b</b>) current densities vs. density of galvanostatic anodic current in 0.1 M NaCl (1), NaBr (2), KI (3), Na<sub>2</sub>SO<sub>4</sub> (4), NaCH<sub>3</sub>COO (5) and NaF (6), pH 10.2 [<a href="#B50-cmd-05-00015" class="html-bibr">50</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 7
<p>Influence of additions of KI (M/L) to aqueous electrolyte 0.1 M NaCl, pH 10.2 on the current densities of self-dissolution (1), hydride formation (2), and anodic dissolution (3) at the fixed potential −1.28 V (SHE) [<a href="#B50-cmd-05-00015" class="html-bibr">50</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 8
<p>Effect of addition of acetate sodium to 0.1 M NaCl, pH 10.2, aqueous electrolyte solution on the volume of released gaseous hydrogen at the potentiostatic conditions E = −1.28 V (SHE) vs. the amount of passing anodic electricity. without addition (1), 0.01 M (2), 0.05 M (3), 0.1 M (4), 0.3 M (5) [<a href="#B51-cmd-05-00015" class="html-bibr">51</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 9
<p>Anodic polarization curves of Mg in 0.1 M NaCl aqueous electrolyte with the addition of oxalate Na<sub>2</sub>C<sub>2</sub>O<sub>4</sub>, pH 10.2. 1—0 M/L, 2—0.01 M/L, 3—0.02 M/L, 4—0.1 M/L and 5—0.15 M/L. The arrows mark the potential pitting formation [<a href="#B49-cmd-05-00015" class="html-bibr">49</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 10
<p>(<b>a</b>)—Galvanostatic anodic dissolution at 8.2 A/m<sup>2</sup> in 0.1 M NaCl (pH 10.2), electrolyte vs. concentration of potassium oxalate. (<b>a</b>) The experiment duration is 3.5 h, 1,4—self-corrosion current density determined by mass loss and volumetry correspondingly, 2—current density of hydride formation, 3—coefficient R. (<b>b</b>), the experiment duration 20 h, 1—the current density of HE, 2—the current density of the hydride formation, 3—the potential of Mg electrode vs. SHE. Hydride ions concentration was measured by post-anodic polarization of Mg anode in alkali [<a href="#B49-cmd-05-00015" class="html-bibr">49</a>,<a href="#B51-cmd-05-00015" class="html-bibr">51</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 11
<p>Influence of the addition of oxalate sodium on the current densities of HE (1), hydride formation (2), and anodic dissolution (3) at the potentiostatic anodic polarization in 0.1 M NaCl, pH 10.2, E = −1.26 V (<b>a</b>) E = −1.2 V (<b>b</b>) vs. SHE [<a href="#B51-cmd-05-00015" class="html-bibr">51</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 12
<p>(<b>a</b>)—Influence of additions of sodium glycinate (<b>a</b>) and sodium tartrate (<b>b</b>) on the current densities of HE (1), hydride formation (2), and anodic dissolution (3) at the potentiostatic anodic polarization in 0.1 M NaCl, pH 10.2, E = −1.26 V (SHE) [<a href="#B51-cmd-05-00015" class="html-bibr">51</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 13
<p>Calculation of near electrode pH<sub>s</sub> (1), i—the rate of corrosion (2), E (3)—the potential of Mg/Mg(OH)<sub>2</sub> electrode as a function of volume pH<sub>o</sub> [<a href="#B59-cmd-05-00015" class="html-bibr">59</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 14
<p>Influence of the replacement of Na<sup>+</sup> to NH<sub>4</sub><sup>+</sup>, [Cl<sup>−</sup>] = 0.1 M, E = −1.26 V vs. SHE. 1—the amount of hydride ion. 2—the anodic current density, 3—HE current density. In each experiment, 150 coulombs of anodic electricity were passed. The amount of the surface hydride in corrosion products on the Mg surface after electrolyzing was detected by anodic oxidation in 1 M NaOH [<a href="#B60-cmd-05-00015" class="html-bibr">60</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">Figure 15
<p>(<b>a</b>)—Anodic potentiodinamic (1 mV/s) curves in 0.1 M NH<sub>4</sub>Cl at pH 5.8 (1), 8.4 (2); 9 (3), 9.5 (4), 10.0 (5), 10.5 (6). (<b>b</b>)—HE current density vs. potential of Mg anode (galvanostatic anodic polarization using step-wise 5–10 mA/cm<sup>2</sup>) in 0.1 M NH<sub>4</sub>Cl at pH 5.5 (1) 7.4 (2); 8.7 (3); 9 (4) 9.5 (5). The pH of the electrolytes was adjusted by the addition of NaOH [<a href="#B60-cmd-05-00015" class="html-bibr">60</a>]. The copyright permission of Pleiades Publishing.</p> Full article ">