WO2013021105A2 - Novel derivatives of furanones and pharmaceutical composition containing same - Google Patents
Novel derivatives of furanones and pharmaceutical composition containing same Download PDFInfo
- Publication number
- WO2013021105A2 WO2013021105A2 PCT/FR2012/000335 FR2012000335W WO2013021105A2 WO 2013021105 A2 WO2013021105 A2 WO 2013021105A2 FR 2012000335 W FR2012000335 W FR 2012000335W WO 2013021105 A2 WO2013021105 A2 WO 2013021105A2
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- radicals
- radical
- alkyl
- chosen
- group
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- GBUBYTIPHGERBQ-MPQUPPDSSA-N CC(C)(C)[Si](C)(C)OC[C@@H]([C@H](C(OCc1ccccc1)=C1OCc2ccccc2)OC1=O)OP(OCc1ccccc1)(OCc1ccccc1)=O Chemical compound CC(C)(C)[Si](C)(C)OC[C@@H]([C@H](C(OCc1ccccc1)=C1OCc2ccccc2)OC1=O)OP(OCc1ccccc1)(OCc1ccccc1)=O GBUBYTIPHGERBQ-MPQUPPDSSA-N 0.000 description 1
- IOAUCMVDZYEFFS-JLAZNSOCSA-N OC[C@@H]([C@H](C(O)=C1O)OC1=O)OP(O)(O)=O Chemical compound OC[C@@H]([C@H](C(O)=C1O)OC1=O)OP(O)(O)=O IOAUCMVDZYEFFS-JLAZNSOCSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/655—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having oxygen atoms, with or without sulfur, selenium, or tellurium atoms, as the only ring hetero atoms
- C07F9/65515—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom having oxygen atoms, with or without sulfur, selenium, or tellurium atoms, as the only ring hetero atoms the oxygen atom being part of a five-membered ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07F—ACYCLIC, CARBOCYCLIC OR HETEROCYCLIC COMPOUNDS CONTAINING ELEMENTS OTHER THAN CARBON, HYDROGEN, HALOGEN, OXYGEN, NITROGEN, SULFUR, SELENIUM OR TELLURIUM
- C07F9/00—Compounds containing elements of Groups 5 or 15 of the Periodic Table
- C07F9/02—Phosphorus compounds
- C07F9/547—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom
- C07F9/6558—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom containing at least two different or differently substituted hetero rings neither condensed among themselves nor condensed with a common carbocyclic ring or ring system
- C07F9/65586—Heterocyclic compounds, e.g. containing phosphorus as a ring hetero atom containing at least two different or differently substituted hetero rings neither condensed among themselves nor condensed with a common carbocyclic ring or ring system at least one of the hetero rings does not contain nitrogen as ring hetero atom
Definitions
- the invention relates to pharmaceutical chemistry, and more particularly to novel heterocyclic furan-2 (5H) -one derivatives, to a process for their preparation, to compositions containing them and to their use in the manufacture of medicaments.
- the invention relates to novel heterocyclic furan (5H) -one derivatives having anticancer activity.
- 5H heterocyclic furan
- Vitamin C (ascorbic acid L) has long been thought to have other therapeutic effects than scurvy. According to a controversial hypothesis (see the publication of Rath and Pauling "Hypothesis: Lipoprotein (a) is a surrogate for ascorbate", published in 1990 in Proc Natl Acad Sci USA, vol 87, p 6204-6207), a high plasma concentration of ascorbate could decrease the concentration of lipoprotein Lp (a) and thus act against cardiovascular diseases, cancer and diabetes.
- US Patent 5,278,189 (Rath and Pauling) describes the effect of a drug comprising ascorbate, a lipoprotein inhibitor Lp (a) and an antioxidant against occlusive cardiovascular diseases.
- CMT1A Charcot-Marie-Tooth disease type 1A
- WO 2004/006911 a hereditary peripheral neuropathy
- ⁇ is involved in the regulation of PMP22 gene expression, the expression of which also depends on cyclic ((c-AMP). This property is probably responsible for the phenotypic correction of the CMT1A phenotype.
- AA is a competitive inhibitor of adenylate cyclase, probably by the presence of a common structural element in ⁇ and ascorbic acid, namely the furanic ribose nucleus.
- EP 0 202 589 and EP 0 339 486 disclose a prophylactic and curative effect of blood circulation disorders for certain ascorbic acid derivatives L; US 5,541,221 (Allergan) describes type 3 (4H) substituted furanoses (5H) and their use for the prevention of osteoporosis; EP 0 768 314 (Senju Pharmaceutical Co. Ltd.) discloses diesters of phosphoric acid with ascorbic acid (position 5) and a tocopherol, some of which have an antioxidant effect (ie of free radical scavenger), likely to reduce ischemic-type disorders.
- an antioxidant effect ie of free radical scavenger
- EP 0 875 246 (Showa Denko Kabushiki Kaisha) describes the anticancer effect of certain L ascorbic acid derivatives, and in particular of its 2-phosphate-6-carboxylic derivatives. Other derivatives having a phosphoryl group in position 2 and for the same application are described in EP 0 875 514 of the same applicant.
- a recent publication by Gazivoda et al. (“Synthesis, structural studies, and cytostatic evaluation of 5,6-di-O-modified L-ascorbic acid derivatives", published in 2006 in Carbohydrate Research, Vol 341, pp. 433-442) suggests a cytostatic effect. for certain chlorinated derivatives at position 6 of 2,3-di-O-benzylated ascorbic acid.
- R 1 and R 2 are selected from alkyl, alkene, aryl, arylalkyl, heteroarylalkyl and where R 4 is H,
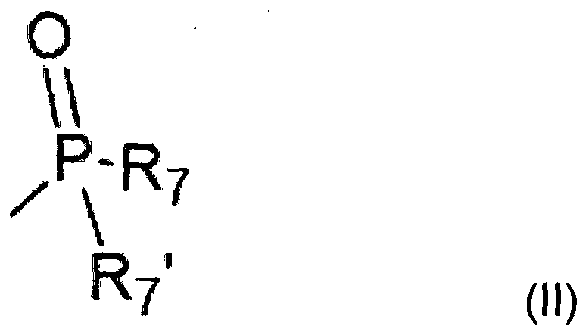
- R 3 is a phosphoryl group of formula (II):
- R 7 and R 71 are OH or substituted arylalkyl as described below; said alkyl, aryl, alkene, heteroalkylaryl and arylalkyl radicals being substituted by one or more identical or different radicals selected from the group consisting of: halogen, CF 3 , OCF 3 , nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide (CONH 2 ), acyl, free carboxy or esterified with an alkyl, aryl, alkene or alkyne radical;
- the halogen term denotes F, Cl, Br and I, and preferably F, Cl and Br;
- the term alkyl denotes a linear or branched radical containing at most 12 carbon atoms, said alkyl radical being chosen from the group formed by the radicals: methyl, ethyl, propyl, isopropyl, butyl, iso-butyl, sec-butyl, tert- butyl, pentyl, iso-pentyl, sec-pentyl, tert-pentyl, neo-pentyl, hexyl, isohexyl, sec-hexyl, tert-hexyl, heptyl, octyl, nonyl, decyl, undecyl, dodecyl, and their isomers linear position and branched, said radical preferably containing at most 6 carbon atoms, and said radical being even more preferably selected from the group formed by
- alkoxy denotes a linear or branched radical containing at most 12 carbon atoms, said alkoxy radical being preferably chosen from the group formed by methoxy, ethoxy, propoxy, iso-propoxy, linear butoxy, secondary or tertiary, pentoxy, hexoxy, heptoxy, as well as their linear and branched positional isomers, and said alkoxy radical being even more preferably selected from radicals of this group containing at most 6 carbon atoms;
- alkylthio denotes a linear or branched radical containing at most 12 carbon atoms, said alkylthio radical being preferentially chosen from the group formed by methylthio, ethylthio, isopropylthio and heptylthio, knowing that in each of these radicals, the sulfur can be oxidized in radical SO or S0 2 , and said alkylthio radical being even more preferably selected from radicals of this group containing at most 6 carbon atoms;
- carboxyamide denotes CONH 2 ;
- sulphonamide denotes SO 2 NH 2 ;
- acyl or R A -C (O) - denotes a linear or branched radical containing at most 12 carbon atoms, and wherein R A represents a radical chosen from the group formed by: H, alkyl, cycloalkyl, cycloalkenyl, heterocycloalkyl heterocycloalkylalkyl aryl, said radicals being optionally substituted; and wherein said acyl radical is preferably selected from the group consisting of: formyl, acetyl, propionyl, butyryl, benzoyl, valeryl, hexanoyl, acroyl, crotonyl and carbamoyl;
- cycloalkyl preferably denotes a monocyclic or bicyclic carbocyclic radical containing from 3 to 10 carbon atoms in the ring, said radical preferably being chosen from the group formed by the cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl radicals;
- cycloalkylalkyl denotes a radical in which cycloalkyl and alkyl are chosen from those indicated above, and preferably from the group formed by: cyclopropylmethyl, cyclopentylmethyl, cyclohexylmethyl and cycloheptylmethyl;
- acyloxy denotes an acyl-O radical in which acyl has the meaning indicated above, said acyloxy radical preferably being an acetoxy or propionyloxy radical;
- acylamino denotes an acyl-N radical in which acyl has the meaning indicated above;
- aryl denotes an unsaturated carbocyclic radical, monocyclic or consisting of condensed rings, and preferably phenyl or naphthyl radicals;
- arylalkyl denotes a radical resulting from the substitution of one or more hydrogen atoms of an aryl radical with one or more alkyl radicals, said arylalkyl radical preferably being chosen from the group formed by the benzyl and phenylethyl radicals, phenylethyl, triphenylmethyl and naphthylenemethyl;
- alkene denotes a linear or branched radical containing at most 12 carbon atoms and having from 1 to 4 double bonds, conjugated or otherwise, said alkene radical being preferably chosen from the group formed by vinyl, allyl or 2-butene radicals; ;
- alkyne denotes a linear or branched radical containing at most 12 carbon atoms and having from 1 to 4 triple bonds, conjugated or otherwise, said alkyne radical being preferably chosen from the group formed by the propargyl, 2-butyne or 2-membered radicals; -pentyne;
- heterocyclic radical denotes a saturated carbocyclic radical (here called heterocycloalkyl) or a partially or totally unsaturated (here called heteroaryl) radical containing from 4 to 10 carbon atoms in the ring, and one or more heteroatoms, which may be identical or different, chosen from oxygen, nitrogen or sulfur atoms;
- said heterocycloalkyl radical being preferably selected from the group consisting of dioxolane, dioxane, dithiolane, thiooxolane, thiooxane, oxiranyl, oxolanyl, dioxolanyl, piperazinyl, N-methylpiperazinyl, piperidyl, pyrrolidinyl, imidazolidinyl, imidazolidin-2, 4-dione, pyrazolidinyl, morpholinyl, tetrahydrofuryl, hexahydropyran, tetrahydro-thienyl, chromanyl, dihydrobenzofuranyl, indolinyl, perhydropyranyl, pyrindolinyl, tetrahydroquinolinyl, tetrahydroisoquinolinyl, thioazolidinyl, these radicals may or may not be substituted; said heteroaryl radical being
- fused heteroaryl radicals are preferably selected from the group consisting of benzothienyl, benzofuryl, benzopyrrolyl, benzothiazolyl, benzimidazolyl, imidazopyridyl, imidazopyrimidinyl, imidazopyrazinyl, purinyl, pyrrolo-pyrimidinyl, pyrrolopyridinyl, benzoxazolyl, benzisoxazolyl, benzisothiazolyl radicals.
- alkylamino denotes a radical in which the alkyl radical is chosen from the alkyl radicals mentioned above, given that the alkyl radicals having at most
- dialkylamino denotes a radical in which the alkyl radicals, which are identical or different, are chosen from the alkyl radicals mentioned above, it being preferred that alkyl radicals having at most 4 carbon atoms, such as the dimethylamino and diethylamino radicals, are chosen, methylethylamino, or dibutylamino, linear or branched;
- Another subject of the invention is a compound addition salt according to the invention with an inorganic or organic acid or with a mineral or organic base.
- Another subject of the invention is a pharmaceutical composition
- a pharmaceutical composition comprising as active ingredient at least one compound according to the invention and / or at least one addition salt according to the invention.
- a final subject of the invention is the use of a pharmaceutical composition according to the invention for the production of medicaments intended for the treatment or prevention of cancers, and preferably of cancers selected from the group formed by: lung, breast cancer, ovarian cancer, prostate cancer, pancreatic cancer, colon cancer, thyroid cancer, carcinoma rectal, glioblastoma, chronic myeloid leukaemias, acute lymphoblastic leukemia, metastatic melanoma, renal carcinoma.
- R some radicals are marked by the letter R followed by one or more digits which can be, indifferently, an index (for example R 3 ) or an exponent (for example R 3 ) or be written on the same line as "R", for example R3.
- stereosiomerism is defined in its broad sense and includes in particular the isomerism of compounds having the same developed formula but whose different groups are arranged differently in space, such as for example in mono-substituted cyclohexanes. whose substituent may be in axial or equatorial position, and in the ethane derivatives which may have different rotational conformations. Stereoisomerism also encompasses the different spatial arrangements of attached substituents, either on double bonds or on rings, also known as geometric isomerism or cis-trans isomerism.
- the structural formulas of the products of formula (I) also cover the addition salts with inorganic and organic acids or with mineral or organic bases.
- patient refers to a human or other mammal.
- the compound of general formula (I) is characterized in that:
- R 1 and R 2 are benzyl radicals substituted by one or more identical or different radicals chosen from: H, fluorine, chlorine, bromine, iodine, CF 3 ,
- OCF 3 nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyl, carboxy free or esterified with an alkyl, aryl, alkene or alkyne radical;
- R 7 and / or R 71 is an OH radical
- R 4 is a H radical.
- the compound of general formula (I) is characterized in that:
- R 1 and R 2 are benzyl radicals substituted by one or more identical or different radicals chosen from: H, fluorine, chlorine, bromine, iodine, CF 3 ,
- OCF 3 nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyl, carboxy free or esterified with an alkyl, aryl, alkene or alkyne radical;
- R 7 and / or R 71 is an O-benzyl radical substituted with one or more identical or different radicals chosen from: H, fluorine, chlorine, bromine, iodine, CF 3 ,
- OCF 3 nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyl, carboxy free or esterified with an alkyl, aryl, alkene or alkyne radical;
- R 4 is a H radical.
- R 3 may advantageously be a substituted or unsubstituted dibenzylphosphonate group, or phosphate.
- R 8 and R 81 which are identical or different, are radicals chosen from the group formed by: H, alkyl, aryl and are preferably identical radicals of alkyl type containing between 1 and 4 carbon atoms, and are more preferably radicals methyl;
- R 1 and R 2 may be identical or different, as defined above, and are preferably identical benzyl radicals, substituted or unsubstituted by radicals selected from the group consisting of F, Cl, Br, I, CF 3 , OCF 3 , nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyl, carboxy free or esterified with an alkyl, aryl, alkene or alkyne radical;
- R 9 is a protecting group of the alcohol functions, preferably a derivative of the silane, and in particular trialkylsilane;
- R 3 is a radical as defined above, and in particular dibenzylphosphonate (possibly substituted as indicated above in the definition of R 7 and R 71 radicals) or phosphate;
- R 4 is a H radical.
- R 3 is a dibenzylphosphonate radical (possibly substituted as indicated above in the definition of R 7 and R 71 radicals) or phosphate.
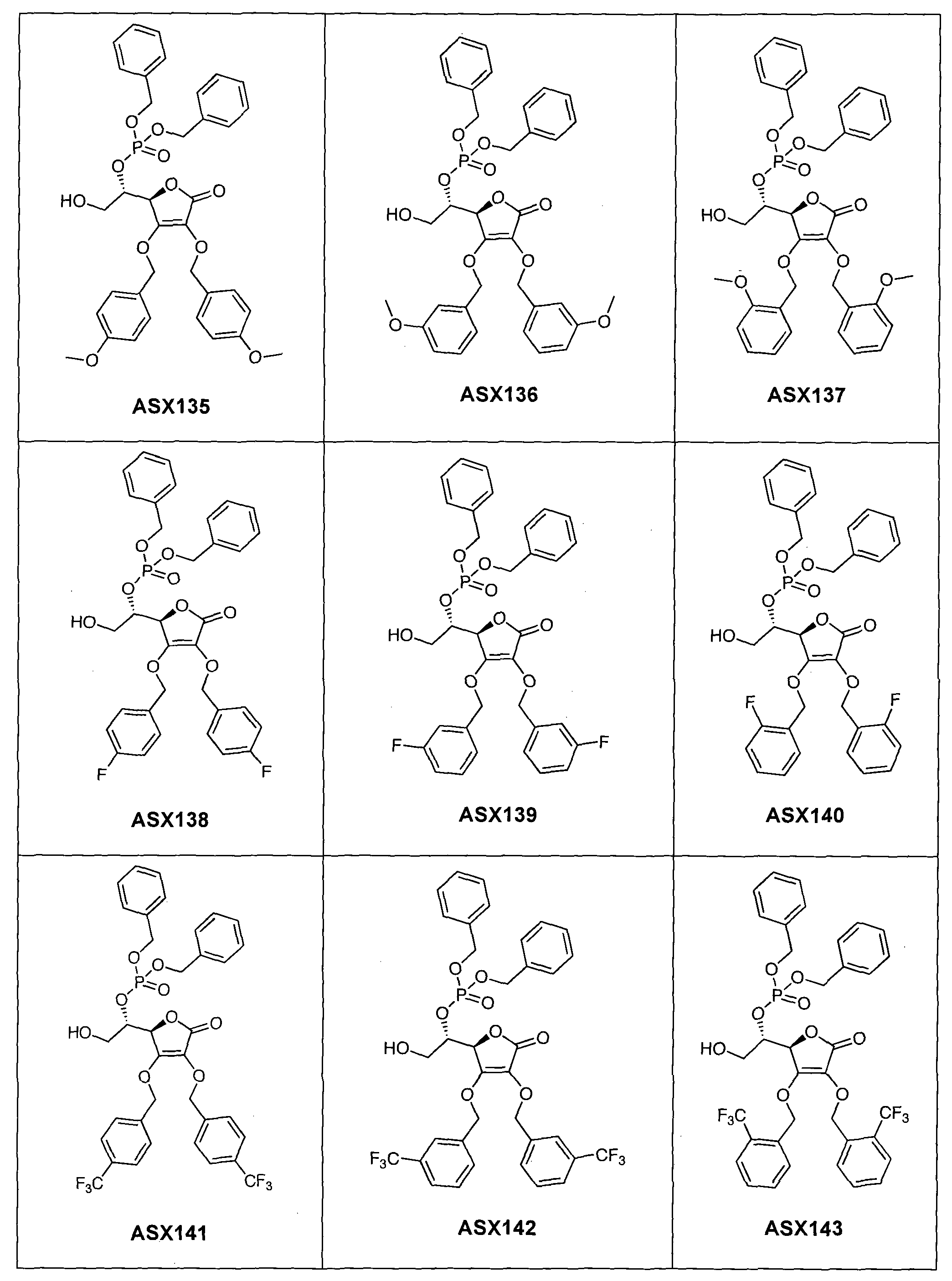
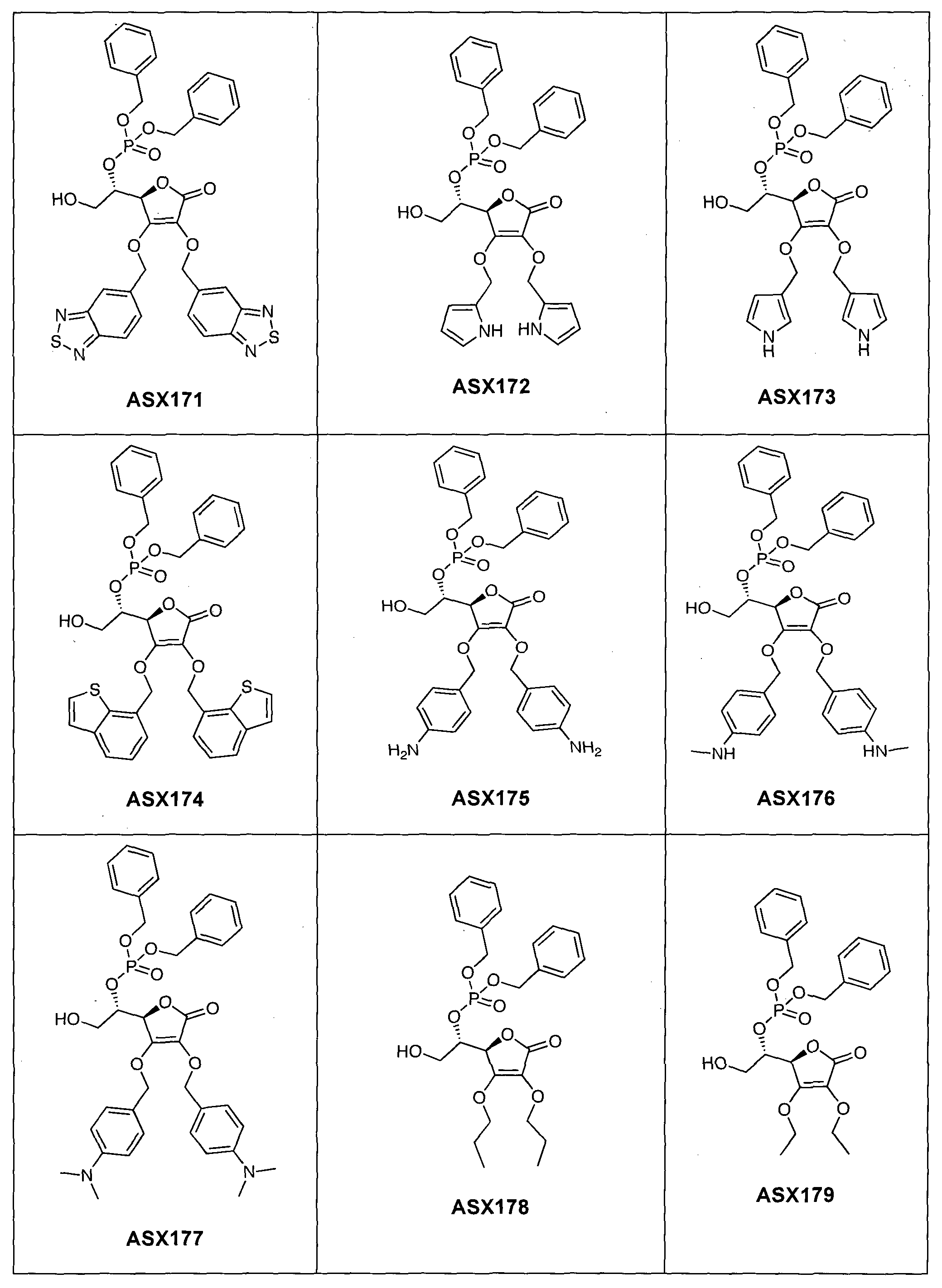
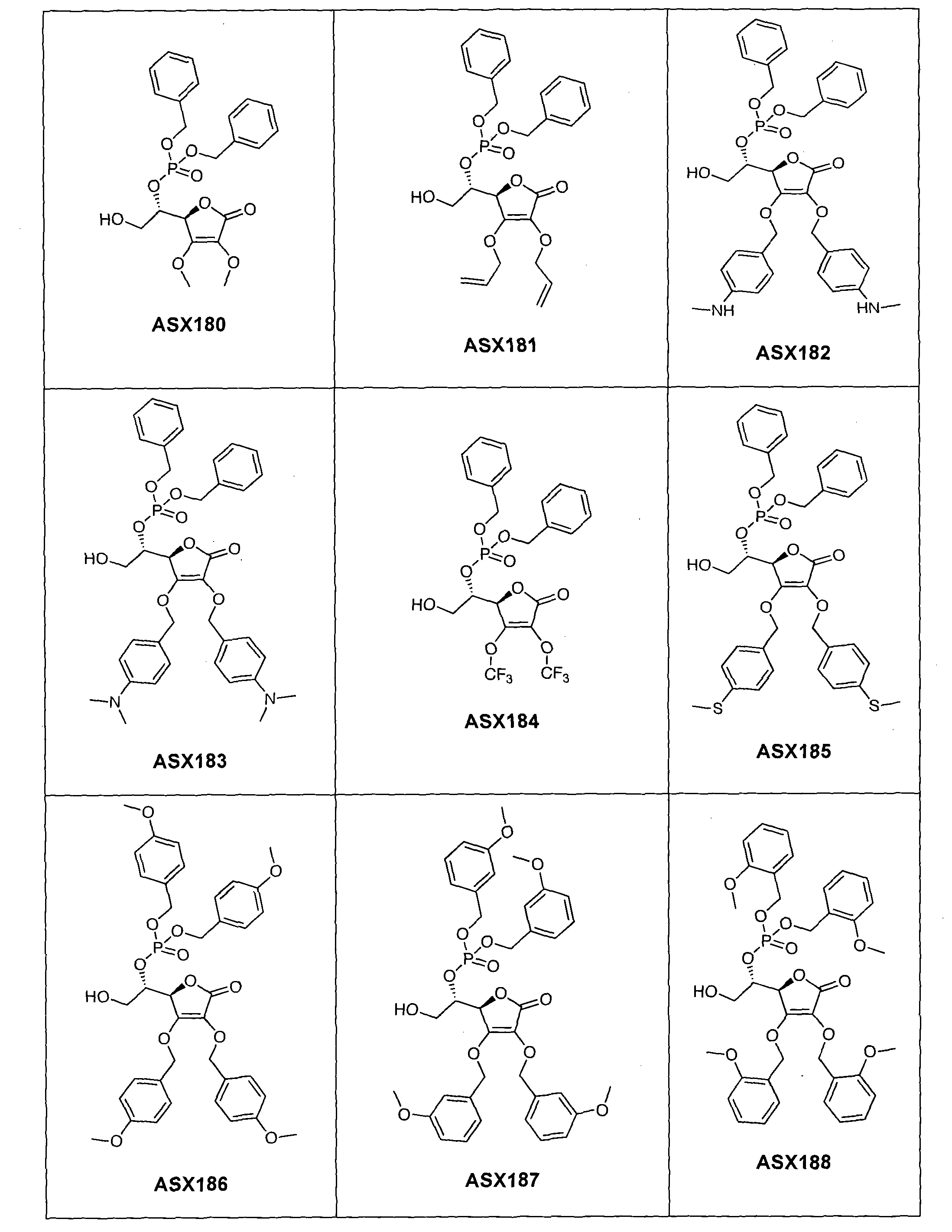
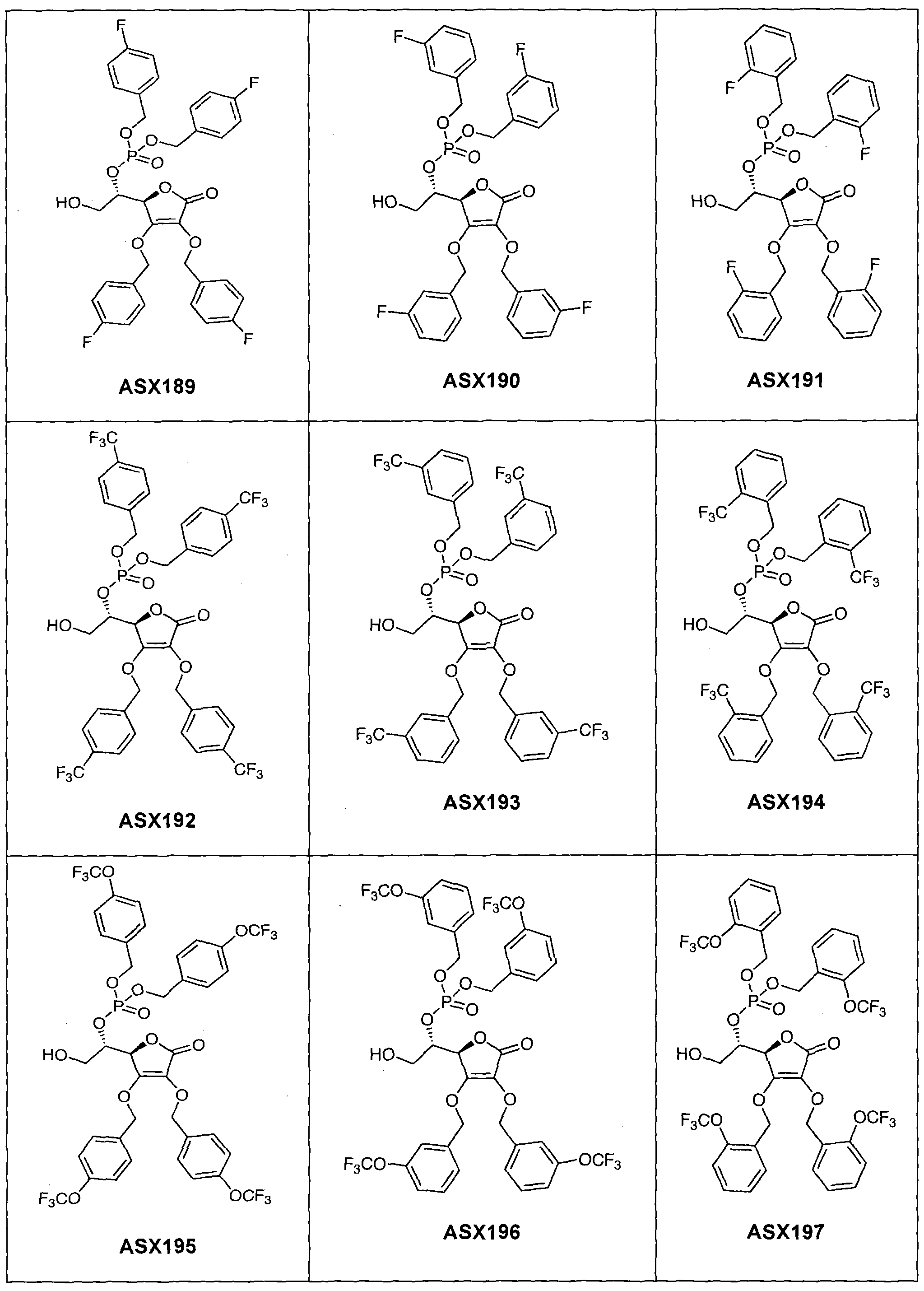
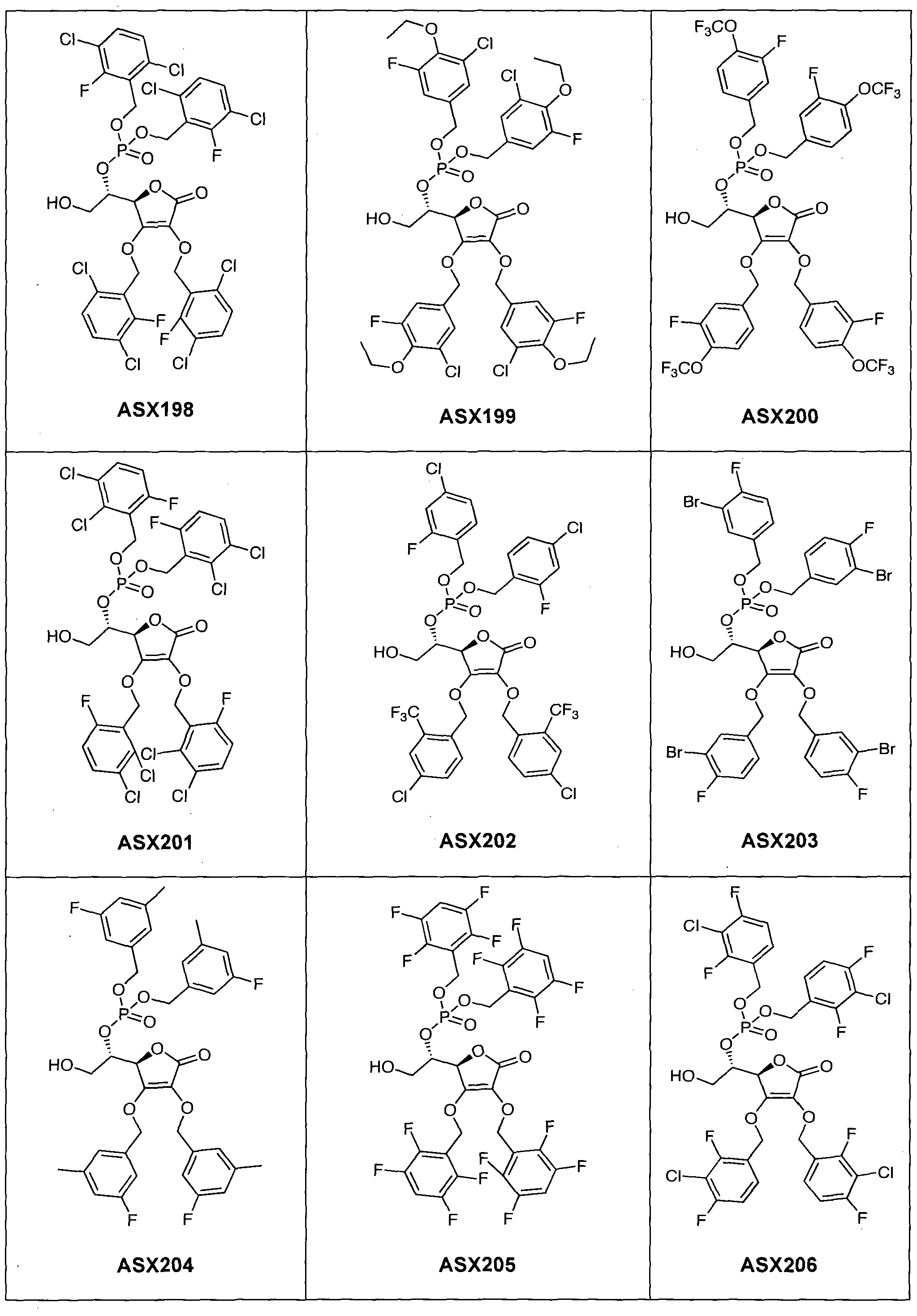
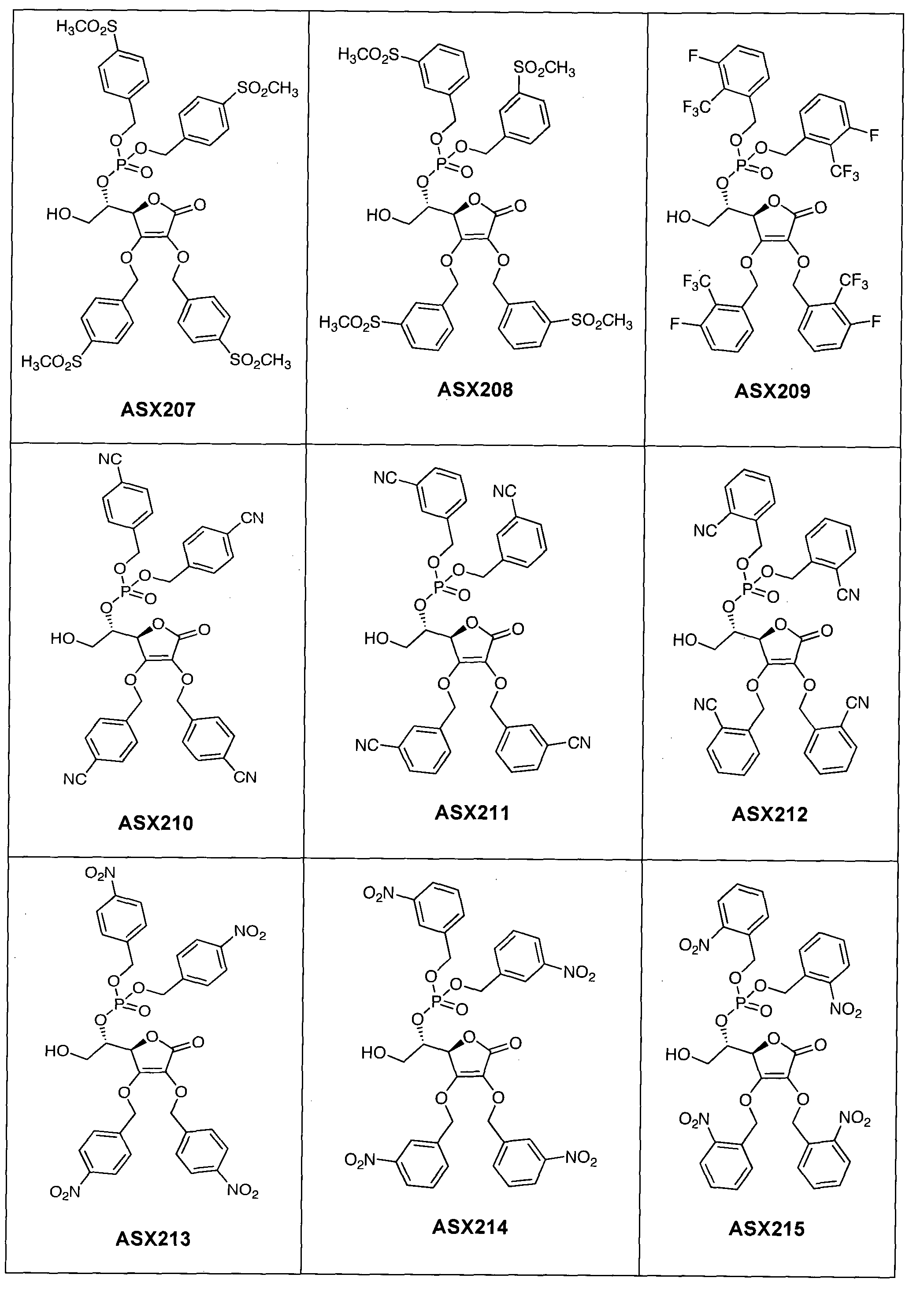
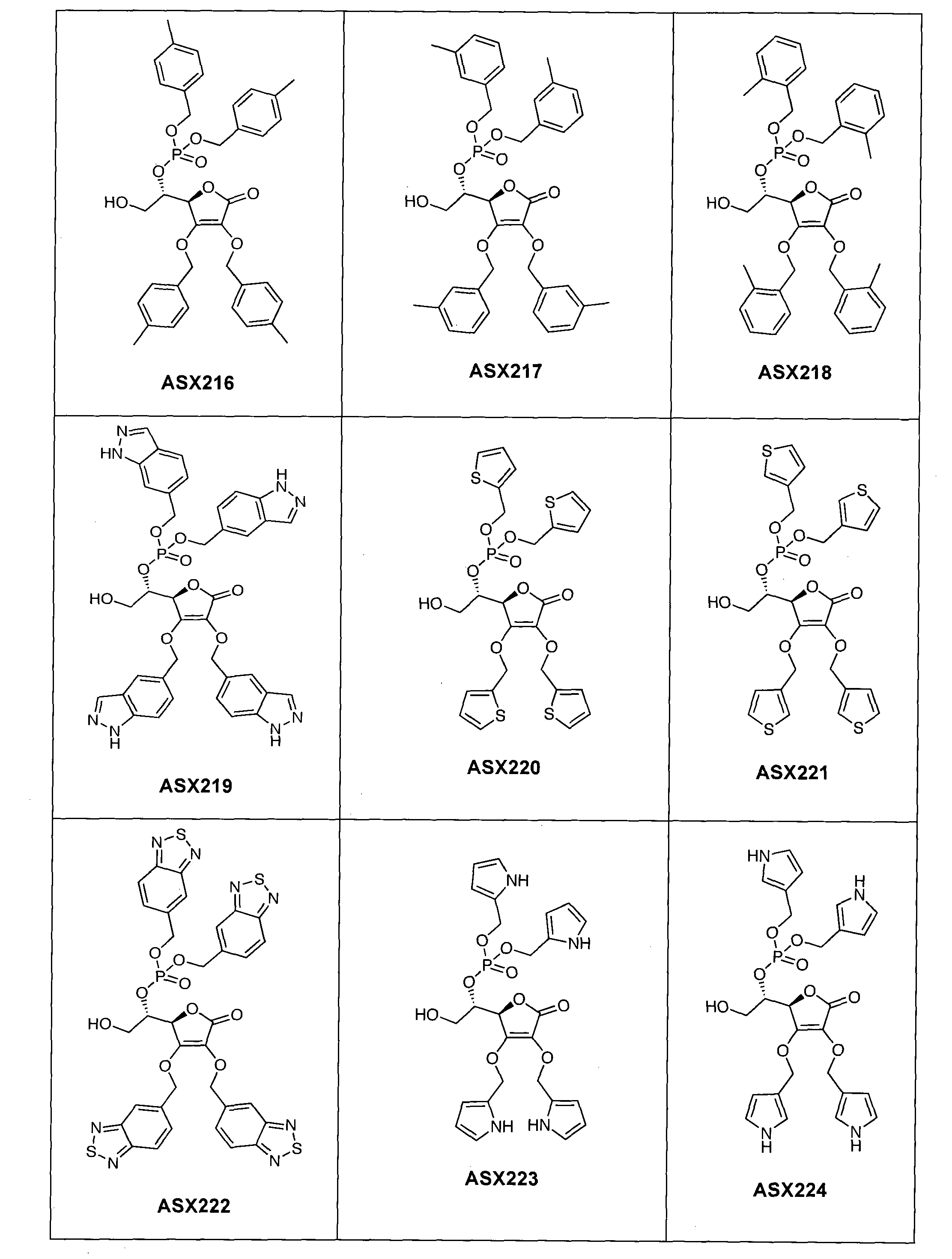
- the compound of formula (I) can in particular be one of the following compounds (the ASX codes followed by three digits are internal codes used in the context of this document): ASX135 to ASX237 and ASX 401 to ASX453.
- the structural formulas of these compounds of formula (I) according to the invention are indicated below; they include all isomers and isomeric mixtures:
- the compound (VI) is supplied and is converted successively with appropriate reagents into compounds (VII), (VIII), (IX) and (I).
- the compounds according to the invention obtained by synthesis according to the process described above, were tested in two ways: firstly on carcinogenic human cell cultures. to determine their IC 50 value, then, for compounds that appear particularly promising, on an animal model of colon cancer that involves monitoring the evolution of the tumor in volume every three days.
- the inventors have found that the toxicity of the compounds of formula (I) is generally low. This promotes their use as a medicine.
- the inventors have furthermore found that the compounds of formula (I) show a biological effect which makes it possible to envisage their therapeutic use in pharmaceutical compositions.
- Dibenzyl (S) -1 - [(R) - 3,4-bis (benzyloxy) -5-oxo-2,5-dihydrofuran-2-yl is used.
- 2-hydroxyethylphosphate (ASX234) derived from ascorbic acid (AA), which is found to be a cytotoxic molecule for cancer cells, at concentrations between 10 and 20 times lower than that of the original molecule (AA) .
- ASX234 is non-toxic for normal cells, which is rare in the treatment of cancer.
- the inventors have found that the compounds according to the invention represent a new class of anti-cancer molecules.
- the observations made by the inventors in the course of their work enabled them to identify at least one such mechanism of action, which is based on on the role of competitors of ATP compounds according to the invention; this new mechanism induces cell death by necrosis and has no visible side effects on non-cancerous cells.
- the compounds according to the invention can be used as active ingredients in pharmaceutical compositions for the treatment or prevention of cancer.
- This use can implement the compounds as such, and / or a pharmaceutically acceptable salt of said compounds, said salt being a salt addition of said compounds with an inorganic or organic acid or with a mineral or organic base.
- a pharmaceutical composition according to the invention comprises as active ingredient at least one compound according to the invention and / or at least one addition salt according to the invention.
- Said pharmaceutical compositions may be administered especially orally, for example in solid form or, especially when it is a question of salts, in a form soluble in water and / or in a alcohol compatible with water (such ethanol, propylene glycol, polypropylene glycol and glycerol). They can also be administered parenterally, for example intravenously, intramuscularly, subcutaneously or rectally. They may include other components, including pharmaceutically acceptable carriers and / or excipients, and / or other active ingredients.
- Cancers whose treatment and / or prevention may be considered are preferably lung cancers, breast cancers, ovarian cancers, prostate cancers, pancreatic cancers, colon cancers, thyroid cancers, rectal carninomas, glioblastomas, chronic myeloid leukemias, acute lymphoblastic leukemias, metastatic melanomas, renal carninomas.
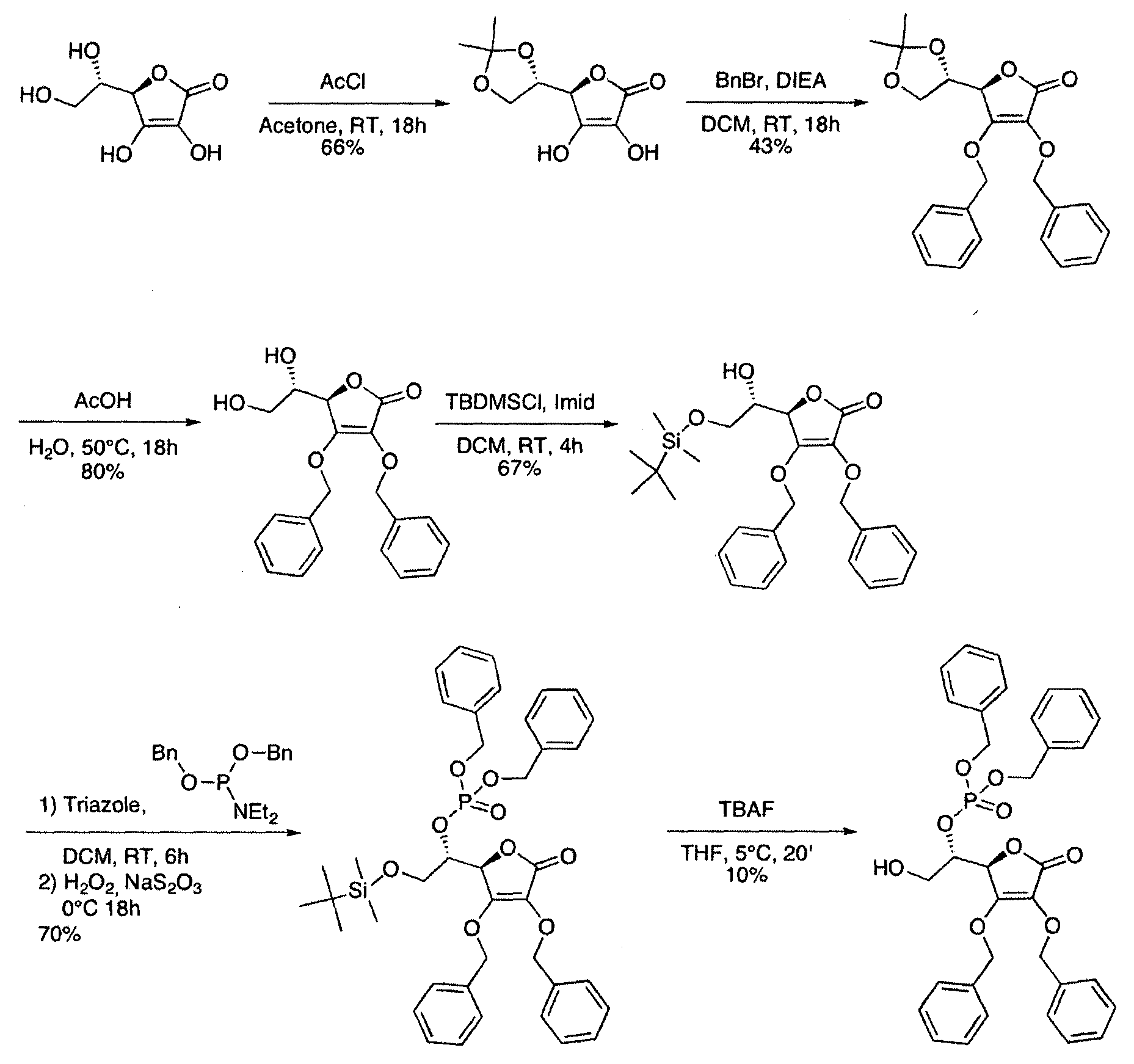
- Example 1 dibenzyl (S) -1 - [(R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydrofuran-2-yn-2-hydroxyethyl phosphate (herein referred to as "ASX234" ):
- Step 1 (R) -5-f (S) -2,2-dimethyl-1,3-dioxolan-4-yn-3,4-dihydroxyfuran-2 (5H) -one
- Step 3 (R) -3,4-bis (benzyloxy) -5-r (S) -1-l 2 dihvdroxyethyllfuran-2 (5H) -one
- step 2 A mixture of the compound obtained in step 2 (9.5 g, 24.2 mmol) in acetic acid (50 mL) and water (15 mL) was heated at 50 ° C overnight. The mixture was evaporated, neutralized with NaHCO 3 and extracted with DCM. The organic phase is dried over Na 2 S0 4 , filtered and evaporated. The residue was purified on silica gel to give the expected product (6.8 g, 80% yield).
- Step 4 (R) -3,4-bis (benzyloxy) -5-i (S) -2- (tert-butyldimethylsilyloxy) -1-hydroxyethynfuran-2 (5H) -one
- step 3 To a solution of the compound obtained in step 3 (4.26 g, 11.97 mmol) and imidazole (1.6 g, 23.93 mmol) in DCM (60 mL) at RT under N 2 TBDMSCI (3.6 g, 23.93 mmol) was added. After 4h, the reaction mixture was filtered and rinsed with DCM. The filtrate was washed with water and then with brine, dried over Na 2 SO 4 , filtered and evaporated. The residue obtained was purified on silica gel to give the expected product (3.7 g, 66% yield).
- Step 6 dibenzyl (S) -1 - [(R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydrofuran-2-yn-2- (tert-butyldimethylsilyloxy) ethyl phosphate
- step 4 To a mixture of the compound obtained in step 4 (2.0 g, 4.25 mmol) was added NaHCO 3 (2.2 g, 20.1 mmol) and triazole (0.6 g, 8.5 mmol) in DCM (40 mL); then a solution of the phosphoramidite obtained in step 5 (3.2 g, 10.2 mmol) in DCM (60 mL) was added at RT. After 6h, the reaction medium was cooled to 0-5 ° C and H 2 O 2 (9 mL) was slowly added. After stirring overnight, the medium was quenched with a solution of NaS 2 O 3 (30 mL). Water was added and the product was extracted with DCM. The organic phase was dried over Na 2 SO 4 , filtered and evaporated. The residue obtained was purified on silica gel to give the expected product (2.2 g, yield 70%).
- Step 7 dibenzyl (S) -1- (R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydrofuran-2-yl-2-hydroxyethyl phosphate
- step 6 To a solution of the compound obtained in step 6 (100 mg, 0.136 mmol) in THF (2 mL), TBAF (1 M in THF, 0.14 mL, 0.136 mmol) was added at 5-10. ° C. After 20 min, the reaction was quenched with NH 4 CI solution and the product was extracted with DCM. The organic phase was dried over Na 2 SO 4 , filtered and evaporated. The residue obtained was purified on silica gel to give the expected product (8 mg, yield 10%)
- step 6 of Example 1 The compound obtained in step 6 of Example 1 (150 mg) was dissolved in methanol (5 ml). Palladium on charcoal (15mg, 10% w / w) was added and the medium was placed in an autoclave under 4 bar of hydrogen at room temperature for 4h. The medium was then filtered through Celite, the methanol evaporated and the residue purified on RP18 phase. The fractions were lyophilized and 25 mg of pure product was recovered as an oil.
- Step 3 (S) -1-r (R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydro-furan-2-yn-2- (tert-butyldimethylsilyloxy) ethyl dihydrogen phosphate
- step 2 To a solution of the product of step 2 (135mg, 214pmol) in THF (5mL) was added formic acid (125 ⁇ , 3.3mmol), triphenylphosphine (35mg, 128mol), butylamine (62mg, 857 ⁇ ), then palladium tetrakis (25mg, 21 ⁇ ). The medium was stirred under nitrogen for 18h. The medium was purified on silica gel (DCM ⁇ DCM / MeOH 9/1) and the expected product was obtained (40 mg, yield 34%).
- DCM ⁇ DCM / MeOH 9/1 silica gel
- Step 4 (S) -1 - [(R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydrofuran-2-yn-2-hydroxyethyl dihydrogen phosphate
- step 3 To a solution of the compound obtained in step 3 (75 mg, 0.136 mmol) in THF (2 mL), TBAF (1 M in THF, 0.14 mL, 0.136 mmol) was added at 5-10. ° C. After 20 min, the reaction was quenched with a solution of NH 4 Cl and the product was extracted with DCM. The organic phase was dried over Na 2 SO 4 , filtered and evaporated. The residue obtained was purified on silica gel (DCM ⁇ DCM / MeOH 9/1) to give the expected product (10 mg, yield 17%)
- ASX410 ASX411, ASX412, ASX413, ASX414, ASX415, ASX416, ASX417, ASX418, ASX419, ASX420, ASX421, ASX422, ASX423, ASX424, ASX425, ASX426, ASX427,
- cells derived from lymphoma, multiple myeloma and colon cancer were used. These lines, obtained from the American Type Culture Collection (ATCC), are recognized as representative of these cancers.
- ATCC American Type Culture Collection
- the first line studied is derived from a plasmocytoma (RPMI 8226), responsible for multiple myeloma, which is an abnormal multiplication of plasma cells (a variety of white blood cells that synthesize circulating antibodies) in the bone marrow.
- RPMI 8226 plasmocytoma responsible for multiple myeloma, which is an abnormal multiplication of plasma cells (a variety of white blood cells that synthesize circulating antibodies) in the bone marrow.
- the second cancer line (Raji) is derived from Burkitt's lymphoma cells. This lymphoma constituting a particularly aggressive malignant tumor has become a model of oncogenesis of lymphomas.
- the third line is derived from an adenocarcinoma of the colon (HT29c2), which is the most common cancer in France (more than 15% of all cancers), which makes it a major public health problem.
- HT29c2 adenocarcinoma of the colon
- Cytotoxicity 10,000 cells were incubated in wells of microtiter plates with 100 ⁇ l of O / N culture medium at 37 ° C., 5% CO 2 . The plating medium was replaced by 100 ⁇ l / well of the test molecules. The cells were incubated 24h, at 37 ° C., 5% CO 2. The cell viability was evaluated by the MTT test described by Mosmann in 1983, well known to those skilled in the art, using a microplate reader (Victor4x). the company Perkin Elmer).
- Proliferation The same incubation protocol was used with the cells. Proliferation was evaluated using the Perkin Elmer Deifia Proliferation Kit, using the protocol recommended by the supplier.
- mice 20 female Athymic Nude (Harlan) mice, 8 weeks old, were used. 3 x 10 6 cells (HT29, human colonic adenocarcinoma cell line) resuspended in PBS, were injected subcutaneously in a volume of 0.2 ml per mouse. Mice were maintained in a sterile environment throughout the test. After 20 days, when the tumor sizes were between 300 and 500 mm 3 , the animals were randomized into 2 groups (homogeneous average sizes), a treated group (T) and a placebo group (P).
- HT29 human colonic adenocarcinoma cell line
- the T group then received a daily IP injection of the ASX234 molecule at 6 mg / kg / day (of which 25% DMSO), while the P group received a daily IP injection of NaCl (0.9%) (25% DMSO).
- Tumor volume was evaluated every three days, using an electronic caliper. After 2 weeks of treatment the animals were sacrificed, the tumors removed and weighed.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Molecular Biology (AREA)
- Biochemistry (AREA)
- Medicinal Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Description
Nouveaux dérivés de furanones et composition pharmaceutique les contenant Domaine technique Novel furanone derivatives and pharmaceutical composition containing them Technical field
L'invention concerne la chimie pharmaceutique, et plus particulièrement des nouveaux dérivés hétérocycliques de furan-2(5H)-one, leur procédé de préparation, des compositions les contenant le leur utilisation pour la fabrication de médicaments. The invention relates to pharmaceutical chemistry, and more particularly to novel heterocyclic furan-2 (5H) -one derivatives, to a process for their preparation, to compositions containing them and to their use in the manufacture of medicaments.
Plus particulièrement, l'invention concerne de nouveaux dérivés hétérocycliques du furan- 2(5H)-one présentant une activité anticancéreuse. Etat de la technique More particularly, the invention relates to novel heterocyclic furan (5H) -one derivatives having anticancer activity. State of the art
On pense depuis longtemps que la vitamine C (acide ascorbique L) pourrait avoir d'autres effets thérapeutiques que celui contre le scorbut. Selon une hypothèse controversée (voir la publication de Rath et Pauling « Hypothesis : Lipoprotein(a) is a surrogate for ascorbate », parue en 1990 dans Proc. Natl. Acad. Sci. USA, vol 87, p. 6204-6207), une forte concentration plasmatique d'ascorbate pourrait diminuer la concentration en lipoprotéine Lp(a) et ainsi agir contre des maladies cardiovasculaires, le cancer et le diabète. Le brevet US 5,278,189 (Rath et Pauling) décrit l'effet d'un médicament comprenant de l'ascorbate, un inihibiteur du lipoprotéine Lp(a) et un antioxydant contre les maladies cardiovasculaires occlusives. Vitamin C (ascorbic acid L) has long been thought to have other therapeutic effects than scurvy. According to a controversial hypothesis (see the publication of Rath and Pauling "Hypothesis: Lipoprotein (a) is a surrogate for ascorbate", published in 1990 in Proc Natl Acad Sci USA, vol 87, p 6204-6207), a high plasma concentration of ascorbate could decrease the concentration of lipoprotein Lp (a) and thus act against cardiovascular diseases, cancer and diabetes. US Patent 5,278,189 (Rath and Pauling) describes the effect of a drug comprising ascorbate, a lipoprotein inhibitor Lp (a) and an antioxidant against occlusive cardiovascular diseases.
L'effet antiprolifératif de l'acide ascorbique L (vitamine C) à haute dose sur certains types de cancers a été démontré in vitro et in vivo sur un modèle murin du cancer de côlon par l'équipe du professeur Fontés à Marseille (voir par exemple la publication électronique « Antiproliferative effect of Ascorbic Acid Is Associated with the Inhibition of Gènes Necessary to Cell Cyle Progression » par S. Belin et al., parue en février 2009 dans PlosOne 4(2), e4409). Cet effet semble être lié au rôle de cette molécule dans la régulation de l'expression de gènes codant la synthèse de deux classes de protéines nécessaires à la progression du cycle cellulaire: les tRNA synthétases et les initiateurs de la traduction. Ceci a été observé aussi bien in vitro qu'in vivo, entraînant la mort des cellules cancéreuses par nécrose (voir la publication précédemment citées et la publication « Ascorbic Acid and Gene Expression : Another Example of Régulation of Gene Expression by Small Molécules » par S. Belin, F. Kaya et al., parue en 2010 dans la revue Current Genomics vol. 11 , p. 52-57). The antiproliferative effect of high-dose ascorbic acid L (vitamin C) on certain types of cancer has been demonstrated in vitro and in vivo in a mouse model of colon cancer by Professor Fontes' team in Marseille (see for example the electronic publication "Antiproliferative effect of Ascorbic Acid Is Associated with the Inhibition of Genes Necessary to Cell Cylation Progression" by S. Belin et al., published in February 2009 in PlosOne 4 (2), e4409). This effect seems to be related to the role of this molecule in the regulation of the expression of genes encoding the synthesis of two classes of proteins necessary for the progression of the cell cycle: the tRNA synthetases and the translational initiators. This has been observed both in vitro and in vivo, resulting in the death of cancer cells by necrosis (see the previously cited publication and the publication "Ascorbic Acid and Gene Expression: Another Example of Regulation of Gene Expression by Small Molecules" by S Belin, F. Kaya et al., Published in 2010 in Current Genomics, Vol 11, pp. 52-57).
De plus, il a été observé que l'acide ascorbique corrige le phéntype d'un modèle animal de la maladie de Charcot-Marie-Tooth type 1A (CMT1A), une neuropathie périphérique héréditaire. Ces travaux ont été décrits dans la publication de E. Passage et al, Nature Medicine 2004 (doi :10.1083/nm1023) et dans la demande de brevet WO 2004/006911 sur la base d'un modèle murin. Toutefois, les effets observés chez l'homme sont moins concluants (voir la publication de J.Micaleff et al., « Effect of ascorbic acid in patients with Cjharcot-Marie-Tooth disease type 1A : a multicentre, randomised, double-blind, placebo- cotnrolled trial » paru en 2009 dans la revue Lancet Neurol., (12) :1 03-10). In addition, it has been observed that ascorbic acid corrects the phenotype of an animal model of Charcot-Marie-Tooth disease type 1A (CMT1A), a hereditary peripheral neuropathy. This work has been described in the publication by E. Passage et al, Nature Medicine 2004 (doi: 10.1083 / nm1023) and in the patent application WO 2004/006911 on the basis of a mouse model. However, the effects observed in humans are less conclusive (see the publication of J.Micaleff et al., "Effect of ascorbic acid in patients with Cjharcot-Marie-Tooth disease type 1A: multicenter, randomized, double-blind, placebo-cotnrolled trial "published in 2009 in Lancet Neurol., (12): 1 03-10).
Kaya et al. (FEBS Letters 528 (2008), p. 3614-1618) ont montré que ΓΑΑ est impliqué dans la régulation de l'expression du gène PMP22, dont l'expression dépend également de ΓΑΜΡ cyclique (c-AMP). Cette propriété est probablement responsable de la correction phénotypique du phénotype CMT1A. Kaya et al, 2008, ont montré que l'acide ascorbique est un régulateur global du pool de cAMP. Ceci est du à une propriété inconnue de ΓΑΑ, découverte par ces auteurs. L'AA est un inhibiteur compétitif des adénylates cyclase, probablement par la présence d'un élément structurel commun dans ΑΤΡ et l'acide ascorbique, à savoir le noyau furanique ribose. Les effets biologiques de certains dérivés de l'acide ascorbique comportant un noyau furanique ribose ont été explorés depuis longtemps. Ainsi, les documents WO 91/16055 et EP 0 579 769 (Allergan), EP 0 799 218 (Merck Frosst Canada & Co) et EP 1 106 175 (Nippon Hypox Laboratoires Inc.) décrivent certaines molécules de ce type présentant un effet anti-inflammatoire in vitro et in vivo dans un modèle murin. Les documents EP 0 202 589 et EP 0 339 486 (Takeda Chemical Industries, Inc.) décrivent un effet prophylactique et curatif de désordres de la circulation sanguine pour certains dérivés de l'acide ascorbique L ; le document US 5,541 ,221 (Allergan) décrit des furanoses de type 2(5H) substitués en position 3 et 4 et leur utilisation pour la prévention de l'ostéoporose ; le document EP 0 768 314 (Senju Pharmaceutical Co. Ltd.) décrit des diesters de l'acide phosphorique avec l'acide ascorbique (position 5) et un tocophérol, dont certains ont un effet antioxidant (i.e. de capteur de radicaux libres), susceptibles de réduire les troubles de type ischémiques. Kaya et al. (FEBS Letters 528 (2008), pp. 3614-1618) have shown that ΓΑΑ is involved in the regulation of PMP22 gene expression, the expression of which also depends on cyclic ((c-AMP). This property is probably responsible for the phenotypic correction of the CMT1A phenotype. Kaya et al, 2008, have shown that ascorbic acid is a global regulator of the cAMP pool. This is due to an unknown property of ΓΑΑ, discovered by these authors. AA is a competitive inhibitor of adenylate cyclase, probably by the presence of a common structural element in ΑΤΡ and ascorbic acid, namely the furanic ribose nucleus. The biological effects of certain ascorbic acid derivatives with a furanic ribose ring have been explored for a long time. Thus, WO 91/16055 and EP 0 579 769 (Allergan), EP 0 799 218 (Merck Frosst Canada & Co) and EP 1 106 175 (Nippon Hypox Laboratories Inc.) describe certain molecules of this type having an anti-aging effect. Inflammatory in vitro and in vivo in a murine model. EP 0 202 589 and EP 0 339 486 (Takeda Chemical Industries, Inc.) disclose a prophylactic and curative effect of blood circulation disorders for certain ascorbic acid derivatives L; US 5,541,221 (Allergan) describes type 3 (4H) substituted furanoses (5H) and their use for the prevention of osteoporosis; EP 0 768 314 (Senju Pharmaceutical Co. Ltd.) discloses diesters of phosphoric acid with ascorbic acid (position 5) and a tocopherol, some of which have an antioxidant effect (ie of free radical scavenger), likely to reduce ischemic-type disorders.
Le document EP 0 875 246 (Showa Denko Kabushiki Kaisha) décrit l'effet anticancéreux de certains dérivés de acide ascorbique L, et notamment de ses dérivés 2-phosphate-6- carboxyliques. D'autres dérivés comportant un groupe phosphoryle en position 2 et visant la même application sont décrits dans EP 0 875 514 du même déposant. Une publication récente de Gazivoda et al. (« Synthesis, structural studies, and cytostatic évaluation of 5,6- di-O-modified L-ascorbic acide derivatives », parue en 2006 dans la revue Carbohydrate Research, vol. 341 , p. 433-442) suggère un effet cytostatique pour certains dérivés chlorés en position 6 de l'acide ascorbique 2,3-di-O-benzylé. Et finalement, des dérivés de l'acide ascorbique ont été testés pour leurs effets dermatologiques et cosmétologiques, voir EP 1 145 710 (L'Oréal), EP 1 409 494 (Tagra Biotechnologies Ltd.) et EP 1 496 889 (Showa Denko K.K.). EP 0 875 246 (Showa Denko Kabushiki Kaisha) describes the anticancer effect of certain L ascorbic acid derivatives, and in particular of its 2-phosphate-6-carboxylic derivatives. Other derivatives having a phosphoryl group in position 2 and for the same application are described in EP 0 875 514 of the same applicant. A recent publication by Gazivoda et al. ("Synthesis, structural studies, and cytostatic evaluation of 5,6-di-O-modified L-ascorbic acid derivatives", published in 2006 in Carbohydrate Research, Vol 341, pp. 433-442) suggests a cytostatic effect. for certain chlorinated derivatives at position 6 of 2,3-di-O-benzylated ascorbic acid. Finally, ascorbic acid derivatives have been tested for their dermatological and cosmetological effects, see EP 1 145 710 (L'Oreal), EP 1 409 494 (Tagra Biotechnologies Ltd.) and EP 1 496 889 (Showa Denko KK ).
Devant cette multitude d'approches structurelles et biologiques pour explorer l'utilité médicale des dérivés de l'acide ascorbique, les présents inventeurs ont porté leur attention sur de nouveaux dérivés de l'acide ascorbique présentant une analogie structurelle avec ΓΑΤΡ, et en particulier sur leurs possibles effets biologiques, en vue de développer de nouvelles molécules d'intérêt thérapeutique. In view of this multitude of structural and biological approaches to explore the medical utility of ascorbic acid derivatives, the present inventors have focused their attention on novel ascorbic acid derivatives having a structural analogy with ΓΑΤΡ, and in particular on their possible biological effects, with a view to developing new molecules of therapeutic interest.
Objets de l'invention Objects of the invention
Le premier objet de l'invention The first object of the invention
composé de formule générale compound of general formula
où R1 et R2 sont choisis parmi alkyle, alcène, aryle, arylalkyle, hétéro-arylalkyle et où R4 est H, where R 1 and R 2 are selected from alkyl, alkene, aryl, arylalkyl, heteroarylalkyl and where R 4 is H,
et où R3 est un groupement phosphoryle de formule (II) : and where R 3 is a phosphoryl group of formula (II):
dans lequel R7 et R71 sont OH ou arylalkyle substitués comme décrits ci-dessous ; lesdits radicaux alkyle, aryle, alcène, hétéro-alkylaryle et arylakyle pouvant être substitués par un ou plusieurs radicaux identiques ou différents choisi dans le groupe formé par : halogène, CF3, OCF3, nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide (CONH2), acyle, carboxy libre ou estérifié par un radical alkyle, aryle, alcène ou alcyne ; wherein R 7 and R 71 are OH or substituted arylalkyl as described below; said alkyl, aryl, alkene, heteroalkylaryl and arylalkyl radicals being substituted by one or more identical or different radicals selected from the group consisting of: halogen, CF 3 , OCF 3 , nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide (CONH 2 ), acyl, free carboxy or esterified with an alkyl, aryl, alkene or alkyne radical;
sachant que knowing that
- le terme halogène désigne F, Cl, Br et I, et de préférence F, Cl et Br ; le terme alkyle désigne un radical linéaire ou ramifié renfermant au plus 12 atomes de carbone, ledit radical alkyle étant choisi dans le groupe formé par les radicaux : méthyle, éthyle, propyle, isopropyle, butyle, iso-butyle, sec-butyle, tert-butyle, pentyle, iso-pentyle, sec-pentyle, tert-pentyle, néo-pentyle, hexyle, iso-hexyle, sec- hexyle, tert-hexyle, heptyle, octyle, nonyle, décyle, undécyle, dodécyle, ainsi que leurs isomères de position linéaires et ramifiés, ledit radical renfermant de préférence au plus 6 atomes de carbone, et ledit radical étant encore plus préférentiellement choisi dans le groupe formé par les radicaux : méthyle, éthyle, propyle, iso-propyle, n-butyle, iso-butyle, tert-butyle, pentyle linéare ou ramifié, hexyle linéaire ou ramifié ; the halogen term denotes F, Cl, Br and I, and preferably F, Cl and Br; the term alkyl denotes a linear or branched radical containing at most 12 carbon atoms, said alkyl radical being chosen from the group formed by the radicals: methyl, ethyl, propyl, isopropyl, butyl, iso-butyl, sec-butyl, tert- butyl, pentyl, iso-pentyl, sec-pentyl, tert-pentyl, neo-pentyl, hexyl, isohexyl, sec-hexyl, tert-hexyl, heptyl, octyl, nonyl, decyl, undecyl, dodecyl, and their isomers linear position and branched, said radical preferably containing at most 6 carbon atoms, and said radical being even more preferably selected from the group formed by the radicals: methyl, ethyl, propyl, iso-propyl, n-butyl, iso-propyl butyl, tert-butyl, linear or branched pentyl, linear or branched hexyl;
- le terme alkoxy désigne un radical linéaire ou ramifié renfermant au plus 12 atomes de carbone, ledit radical alkoxy étant préférentiellement choisi dans le groupe forme par méthoxy, éthoxy, propoxy, iso-propoxy, butoxy linéaire, secondaire ou tertiaire, pentoxy, hexoxy, heptoxy, ainsi que leurs isomères de position linéaires et ramifiés, et ledit radical alkoxy étant encore plus préférentiellement choisi parmi les radicaux de ce groupe renfermant au plus 6 atomes de carbone ; the term alkoxy denotes a linear or branched radical containing at most 12 carbon atoms, said alkoxy radical being preferably chosen from the group formed by methoxy, ethoxy, propoxy, iso-propoxy, linear butoxy, secondary or tertiary, pentoxy, hexoxy, heptoxy, as well as their linear and branched positional isomers, and said alkoxy radical being even more preferably selected from radicals of this group containing at most 6 carbon atoms;
- le terme alkylthio désigne un radical linéaire ou ramifié renfermant au plus 12 atomes de carbone, ledit radical alkylthio étant préférentiellement choisi dans le groupe formé par méthylthio, éthylthio, isopropylthio et heptylthio, sachant que dans chacun de ces radicaux, le soufre peut être oxydé en radical SO ou S02, et ledit radical alkylthio étant encore plus préférentiellement choisi parmi les radicaux de ce groupe renfermant au plus 6 atomes de carbone ; the term alkylthio denotes a linear or branched radical containing at most 12 carbon atoms, said alkylthio radical being preferentially chosen from the group formed by methylthio, ethylthio, isopropylthio and heptylthio, knowing that in each of these radicals, the sulfur can be oxidized in radical SO or S0 2 , and said alkylthio radical being even more preferably selected from radicals of this group containing at most 6 carbon atoms;
- le terme carboxyamide désigne CONH2 ; the term carboxyamide denotes CONH 2 ;
- le terme sulfonamide désigne S02NH2 ; the term sulphonamide denotes SO 2 NH 2 ;
- le terme acyle ou RA-C(0)- désigne un radical linéaire ou ramifié renfermant au plus 12 atomes de carbone, et où RA représente un radical choisi dans le groupe formé par : H, alkyle, cycloalkyle, cycloalkenyle, hétérocycloalkyle, hétérocycloalkylalkyle aryle, lesdits radicaux étant éventuellement substitués ; et ledit radical acyle étant de préférence choisi dans le groupe formé par : formyle, acétyle, propionyle, butyryle, benzoyle, valéryle, hexanoyle, acroyle, crotonyle et carbamoyle ; the term acyl or R A -C (O) - denotes a linear or branched radical containing at most 12 carbon atoms, and wherein R A represents a radical chosen from the group formed by: H, alkyl, cycloalkyl, cycloalkenyl, heterocycloalkyl heterocycloalkylalkyl aryl, said radicals being optionally substituted; and wherein said acyl radical is preferably selected from the group consisting of: formyl, acetyl, propionyl, butyryl, benzoyl, valeryl, hexanoyl, acroyl, crotonyl and carbamoyl;
le terme cycloalkyle désigne de préférence un radical carbocyclique monocyclique ou bicyclique renfermant de 3 à 10 atomes de carbone dans le cycle, ledit radical étant de préférence choisi dans le groupe formé par les radicaux cyclopropyle, cyclobutyle, cyclopentyle et cyclohexyle ; - le terme cycloalylalkyle désigne un radical dans lequel cycloalkyle et alkyle sont choisis parmi ceux indiqués ci-dessus, et de préférence dans le groupe formé par : cyclopropylméthyle, cyclopentylméthyle, cyclohexylméthyle et cycloheptylméthyle ; le terme acyloxy désigne un radical acyl-0 dans lequel acyl a la signification indiqué ci-dessus, ledit radical acyloxy étant préférentiellement un radical acétoxy ou propionyloxy ; the term cycloalkyl preferably denotes a monocyclic or bicyclic carbocyclic radical containing from 3 to 10 carbon atoms in the ring, said radical preferably being chosen from the group formed by the cyclopropyl, cyclobutyl, cyclopentyl and cyclohexyl radicals; the term cycloalkylalkyl denotes a radical in which cycloalkyl and alkyl are chosen from those indicated above, and preferably from the group formed by: cyclopropylmethyl, cyclopentylmethyl, cyclohexylmethyl and cycloheptylmethyl; the term acyloxy denotes an acyl-O radical in which acyl has the meaning indicated above, said acyloxy radical preferably being an acetoxy or propionyloxy radical;
- le terme acylamino désigne un radical acyl-N dans lequel acyl a la signification indiqué ci-dessus ; the term acylamino denotes an acyl-N radical in which acyl has the meaning indicated above;
le terme aryle désigne un radical carbocyclique insaturé, monocyclique ou constitué de cycles condensés, et de préférence des radicaux phényle ou naphtyle ; the term aryl denotes an unsaturated carbocyclic radical, monocyclic or consisting of condensed rings, and preferably phenyl or naphthyl radicals;
le terme arylalkyle désigne un radical résultant de la substitution d'un ou plusieurs atomes d'hydrogène d'un radical aryle par un ou plusieurs radicaux alkyle, ledit radical arylalkyle étant de préférence choisi dans le groupe formé par les radicaux benzyle, phénylethyle, 2-phényléthyle, triphénylméthyle et naphthylèneméthyle ; le terme alcène désigne un radical linéaire ou ramifié renfermant au plus 12 atomes de carbone et présentant de 1 à 4 liaisons doubles, conjuguées ou non, ledit radical alcène étant de préférence choisi dans le groupe formé par les radicaux vinyl, allyl ou 2-butène ; the term arylalkyl denotes a radical resulting from the substitution of one or more hydrogen atoms of an aryl radical with one or more alkyl radicals, said arylalkyl radical preferably being chosen from the group formed by the benzyl and phenylethyl radicals, phenylethyl, triphenylmethyl and naphthylenemethyl; the term alkene denotes a linear or branched radical containing at most 12 carbon atoms and having from 1 to 4 double bonds, conjugated or otherwise, said alkene radical being preferably chosen from the group formed by vinyl, allyl or 2-butene radicals; ;
le terme alcyne désigne un radical linéaire ou ramifié renfermant au plus 12 atomes de carbone et présentant de 1 à 4 liaisons triples, conjuguées ou non, ledit radical alcyne étant de préférence choisi dans le groupe formé par les radicaux propargyl, 2-butyne ou 2-pentyne ; the term "alkyne" denotes a linear or branched radical containing at most 12 carbon atoms and having from 1 to 4 triple bonds, conjugated or otherwise, said alkyne radical being preferably chosen from the group formed by the propargyl, 2-butyne or 2-membered radicals; -pentyne;
le terme radical hétérocyclique désigne un radical carbocyclique saturé (appelé ici hétérocycloalkyle) ou partiellement ou totalement insaturé (appelé ici hétéroaryle) renfermant de 4 à 10 atomes de carbone dans le cycle, ainsi qu'un ou plusieurs hétéroatomes, identiques ou non, choisis parmi les atomes d'oxygène, d'azote ou de soufre ; the term "heterocyclic radical" denotes a saturated carbocyclic radical (here called heterocycloalkyl) or a partially or totally unsaturated (here called heteroaryl) radical containing from 4 to 10 carbon atoms in the ring, and one or more heteroatoms, which may be identical or different, chosen from oxygen, nitrogen or sulfur atoms;
ledit radical hétérocycloalkyle étant choisi de préférence dans le groupe formé par les radicaux dioxolane, dioxane, dithiolane, thiooxolane, thio-oxane, oxirannyle, oxolannyle, dioxolannyle, pipérazinyle, N-méthyl-pipérazinyle, piperidyle, pyrrolidinyle, imidazolidinyle, imidazolidine-2,4-dione, pyrazolidinyle, morpholinyle, tétrahydrofuryle, hexahydropyranne, tétrahydro-thiényle, chromanyle, dihydrobenzofuranyle, indolinyle, perhydropyranyle, pyrindolinyle, tétrahydroquinoléinyle, tétrahydroiso-quinoléinyle, thioazolidinyle, ces radicaux pouvant être substitués ou non ; ledit radical hétéroaryle étant choisi de préférence dans le groupe formé par les radicaux furyle, pyrrolyle, tétrazolyle, thiazolyle, iso-thiazolyle, diazolyle, thiadiazolyle, thiatriazolyle, oxazolyle, oxadiazolyle, isoxazolyle, imidazolyle, pyrazolyle, thiényle, triazolyle, pyridyle, 2-pyridyle, 4-pyridyle, pyrimidyle, 5- pyridazinyle, pyrazinyle, les radicaux hétéroaryles condensés contenant de préférence un hétéroatome choisi parmi S, O et N ; said heterocycloalkyl radical being preferably selected from the group consisting of dioxolane, dioxane, dithiolane, thiooxolane, thiooxane, oxiranyl, oxolanyl, dioxolanyl, piperazinyl, N-methylpiperazinyl, piperidyl, pyrrolidinyl, imidazolidinyl, imidazolidin-2, 4-dione, pyrazolidinyl, morpholinyl, tetrahydrofuryl, hexahydropyran, tetrahydro-thienyl, chromanyl, dihydrobenzofuranyl, indolinyl, perhydropyranyl, pyrindolinyl, tetrahydroquinolinyl, tetrahydroisoquinolinyl, thioazolidinyl, these radicals may or may not be substituted; said heteroaryl radical being preferably selected from the group consisting of furyl, pyrrolyl, tetrazolyl, thiazolyl, isothiazolyl, diazolyl, thiadiazolyl, thiatriazolyl, oxazolyl, oxadiazolyl, isoxazolyl, imidazolyl, pyrazolyl, thienyl, triazolyl, pyridyl, 2- radicals. pyridyl, 4-pyridyl, pyrimidyl, 5-pyridazinyl, pyrazinyl, condensed heteroaryl radicals preferably containing a heteroatom selected from S, O and N;
sachant que les radicaux hétéroaryles condensés sont de préférence choisis dans le groupe formé par les radicaux benzothiényle, benzofuryle, benzopyrrolyle, benzothiazolyle, benzimidazolyle, imidazo-pyridyle, imidazopyrimidinyle, imidazopyrazinyle, purinyle, pyrrolo-pyrimidinyle, pyrrolopyridinyle, benzoxazolyle, benzisoxazolyle, benz-isothiazolyle, thionaphtyle, chroményle, indolizinyle, quinazolinyle, quinoxalinyle, indolyle, indazolyle, purinyle, quinolyle, isoquinolyle, naphtyridinyle ; wherein the fused heteroaryl radicals are preferably selected from the group consisting of benzothienyl, benzofuryl, benzopyrrolyl, benzothiazolyl, benzimidazolyl, imidazopyridyl, imidazopyrimidinyl, imidazopyrazinyl, purinyl, pyrrolo-pyrimidinyl, pyrrolopyridinyl, benzoxazolyl, benzisoxazolyl, benzisothiazolyl radicals. , thionaphthyl, chromenyl, indolizinyl, quinazolinyl, quinoxalinyl, indolyl, indazolyl, purinyl, quinolyl, isoquinolyl, naphthyridinyl;
- le terme alkylamino désigne un radical dans lequel le radical alkyl est choisi parmi les radicaux alkyles cités ci-dessus, sachant que les radicaux alkyle ayant au plus the term alkylamino denotes a radical in which the alkyl radical is chosen from the alkyl radicals mentioned above, given that the alkyl radicals having at most
4 atomes de carbone sont préférés, tels que les radicaux méthylamino, éthylamino, propylamino ou butylamino, linéaire ou ramifié ; 4 carbon atoms are preferred, such as linear or branched methylamino, ethylamino, propylamino or butylamino radicals;
le terme dialkylamino désigne un radical dans lequel les radicaux alkyle, identiques ou différents, sont choisis parmi les radicaux alkyle cités ci-dessus, sachant que les radicaux alkyle ayant au plus 4 atomes de carbone sont préférés, tels que les radicaux diméthylamino, diéthylamino, méthyléthylamino, ou dibutylamino, linéaire ou ramifié ; the term dialkylamino denotes a radical in which the alkyl radicals, which are identical or different, are chosen from the alkyl radicals mentioned above, it being preferred that alkyl radicals having at most 4 carbon atoms, such as the dimethylamino and diethylamino radicals, are chosen, methylethylamino, or dibutylamino, linear or branched;
ou un des ses sels pharmaceutiquement acceptables. or a pharmaceutically acceptable salt thereof.
Un autre objet de l'invention est un sel d'addition composé selon l'invention avec un acide minéral ou organique ou avec une base minérale ou organique. Another subject of the invention is a compound addition salt according to the invention with an inorganic or organic acid or with a mineral or organic base.
Un autre objet de l'invention est une composition pharmaceutique comprenant à titre de principe actif au moins un composé selon l'invention et/ou au moins un sel d'addition selon l'invention. Another subject of the invention is a pharmaceutical composition comprising as active ingredient at least one compound according to the invention and / or at least one addition salt according to the invention.
Un dernier objet de l'invention est l'utilisation d'une composition pharmaceutique selon l'invention pour la réalisation de médicaments destinés au traitement ou à la prévention de cancers, et de préférence de cancers sélectionnés dans le groupe formé par : les cancers du poumon, les cancers du sein, les cancers de l'ovaire, les cancers de la prostate, les cancers du pancréas, les cancers du colon, les cancers de la thyroïde, les carcinomes rectaux, les glioblastomes, les leucémies myéloïdes chroniques, les leucémies lympho- blastiques aiguës, les mélanomes métastatiques, les carcinomes rénaux. A final subject of the invention is the use of a pharmaceutical composition according to the invention for the production of medicaments intended for the treatment or prevention of cancers, and preferably of cancers selected from the group formed by: lung, breast cancer, ovarian cancer, prostate cancer, pancreatic cancer, colon cancer, thyroid cancer, carcinoma rectal, glioblastoma, chronic myeloid leukaemias, acute lymphoblastic leukemia, metastatic melanoma, renal carcinoma.
Description de l'invention Description of the invention
Dans les formules de structure et le présent texte, certains radicaux sont repérés par la lettre R suivie d'un ou plusieurs chiffres qui peut être, indifféremment, un indice (par exemple R3) ou un exposant (par exemple R3) ou encore être écrit sur la même ligne que « R », par exemple R3. In the formulas of the structure and the present text, some radicals are marked by the letter R followed by one or more digits which can be, indifferently, an index (for example R 3 ) or an exponent (for example R 3 ) or be written on the same line as "R", for example R3.
Sauf mention contraire, pour chaque produit de formule (I) et pour chaque radical défini comme indiqué ci-dessus, ces formules de structure désigne tous les isomères, et notamment toutes les formes tautomères, diastéréoisomères et énantiomères qui peuvent être représentés par cette formule, ainsi que des mélanges de ces formes, y compris des mélanges racémiques. Unless otherwise stated, for each product of formula (I) and for each radical defined as indicated above, these structural formulas denote all the isomers, and in particular all the tautomeric, diastereoisomeric and enantiomeric forms which may be represented by this formula, as well as mixtures of these forms, including racemic mixtures.
Dans le cadre de la présente invention, la stéréosiomérie est définie dans son sens large et englobe notamment l'isomérie de composés ayant la même formule développée mais dont les différents groupes sont disposés différemment dans l'espace, tels que par exemple dans les cyclohexanes monosubstitués dont le substituant peut être en position axiale ou équatoriale, et dans les dérivés de l'éthane qui peuvent présenter différentes conformations rotationnelles. La stéréoisomérie englobe aussi les arrangements spatiaux différents de substituants fixés, soit sur des doubles liaisons, soit sur des cycles, que l'on appelle aussi isomérie géométrique ou isomérie cis-trans. In the context of the present invention, the stereosiomerism is defined in its broad sense and includes in particular the isomerism of compounds having the same developed formula but whose different groups are arranged differently in space, such as for example in mono-substituted cyclohexanes. whose substituent may be in axial or equatorial position, and in the ethane derivatives which may have different rotational conformations. Stereoisomerism also encompasses the different spatial arrangements of attached substituents, either on double bonds or on rings, also known as geometric isomerism or cis-trans isomerism.
Les formules de structure des produits de formule (I) couvrent également les sels d'addition avec des acides minéraux et organiques ou avec des bases minérales ou organiques. The structural formulas of the products of formula (I) also cover the addition salts with inorganic and organic acids or with mineral or organic bases.
Le terme « patient » désigne un être humain ou un autre mammifère. The term "patient" refers to a human or other mammal.
Dans un premier mode de réalisation avantageux de l'invention, le composé de formule générale (I) est caractérisé en ce que : In a first advantageous embodiment of the invention, the compound of general formula (I) is characterized in that:
(a) R1 et R2 sont des radicaux benzyl substitués par un ou plusieurs radicaux identiques ou différents choisis parmi : H, fluor, chlore, brome, iode, CF3,(a) R 1 and R 2 are benzyl radicals substituted by one or more identical or different radicals chosen from: H, fluorine, chlorine, bromine, iodine, CF 3 ,
OCF3, nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyle, carboxy libre ou estérifié par un radical alkyle, aryle, alcène ou alcyne ;OCF 3 , nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyl, carboxy free or esterified with an alkyl, aryl, alkene or alkyne radical;
(b) R7 et/ou R71 est un radical OH ; (b) R 7 and / or R 71 is an OH radical;
(c) R4 est un radical H. Dans un deuxième mode de réalisation avantageux de l'invention, le composé de formule générake (I) est caractérisé en ce que : (c) R 4 is a H radical. In a second advantageous embodiment of the invention, the compound of general formula (I) is characterized in that:
(a) R1 et R2 sont des radicaux benzyl substitués par un ou plusieurs radicaux identiques ou différents choisis parmi : H, fluor, chlore, brome, iode, CF3,(a) R 1 and R 2 are benzyl radicals substituted by one or more identical or different radicals chosen from: H, fluorine, chlorine, bromine, iodine, CF 3 ,
OCF3, nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyle, carboxy libre ou estérifié par un radical alkyle, aryle, alcène ou alcyne ; OCF 3 , nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyl, carboxy free or esterified with an alkyl, aryl, alkene or alkyne radical;
(b) R7 et/ou R71 est un radical O-benzyl substitué par un ou plusieurs radicaux identiques ou différents choisis parmi : H, fluor, chlore, brome, iode, CF3,(b) R 7 and / or R 71 is an O-benzyl radical substituted with one or more identical or different radicals chosen from: H, fluorine, chlorine, bromine, iodine, CF 3 ,
OCF3, nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyle, carboxy libre ou estérifié par un radical alkyle, aryle, alcène ou alcyne ; OCF 3 , nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyl, carboxy free or esterified with an alkyl, aryl, alkene or alkyne radical;
(c) R4 est un radical H. (c) R 4 is a H radical.
Dans ces deux modes de réalisation, R3 peut être avantageusement un groupement dibenzylphosphonate substitué ou non, ou phosphate. In these two embodiments, R 3 may advantageously be a substituted or unsubstituted dibenzylphosphonate group, or phosphate.
Les composés de formule (I) selon l'invention peuvent être préparés conformément au schéma réactionnel suivant : The compounds of formula (I) according to the invention may be prepared according to the following reaction scheme:
où or
R8 et R81, identiques ou différents, sont des radicaux choisis dans le groupe formé par : H, alkyl, aryl, et sont préférentiellement des radicaux identiques de type alkyl comportant entre 1 et 4 atomes de carbone, et sont plus préférentiellement des radicaux méthyl ; R 8 and R 81 , which are identical or different, are radicals chosen from the group formed by: H, alkyl, aryl and are preferably identical radicals of alkyl type containing between 1 and 4 carbon atoms, and are more preferably radicals methyl;
R1 et R2 peuvent être identiques ou différents, tel que définis ci-dessus, et sont de préférence des radicaux identiques de type benzyle, substitués ou non par des radicaux sélectionnés dans le groupe formé par F, Cl, Br, I, CF3, OCF3, nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyle, carboxy libre ou estérifié par un radical alkyle, aryle, alcène ou alcyne ; R 1 and R 2 may be identical or different, as defined above, and are preferably identical benzyl radicals, substituted or unsubstituted by radicals selected from the group consisting of F, Cl, Br, I, CF 3 , OCF 3 , nitro, cyano, hydroxy, alkoxy, aryloxy, mercapto, alkylthio, arylthio, amino, alkylamino, dialkylamino, arylamino, diarylamino, carboxamide, acyl, carboxy free or esterified with an alkyl, aryl, alkene or alkyne radical;
R9 est un groupement protecteur des fonctions alcool, préférentiellement un dérivé du silane, et en particulier trialkylsilane ; R 9 is a protecting group of the alcohol functions, preferably a derivative of the silane, and in particular trialkylsilane;
R3 est un radical tel que défini ci-dessus, et en particulier dibenzylphosphonate (possiblement substitué comme indiqué ci-dessus dans la définition des radicaux R7 et R71) ou phosphate ; R 3 is a radical as defined above, and in particular dibenzylphosphonate (possibly substituted as indicated above in the definition of R 7 and R 71 radicals) or phosphate;
R4 est un radical H. R 4 is a H radical.
Dans un mode de réalisation avantageux, R3 est un radical dibenzylphosphonate (possiblement substitué comme indiqué ci-dessus dans la définition des radicaux R7 et R71) ou phosphate. A ce titre, le composé de formule (I) peut notamment être un des composés suivants (les codes ASX suivis de trois chiffres sont des codes internes utilisés dans le cadre du présent document) : ASX135 à ASX237 et ASX 401 à ASX453. Les formules structurelles de ces composés de formuila (I) selon l'invention sont indiqués ci-dessous ; elles incluent tous les isomères et les mélanges d'isomères : In an advantageous embodiment, R 3 is a dibenzylphosphonate radical (possibly substituted as indicated above in the definition of R 7 and R 71 radicals) or phosphate. As such, the compound of formula (I) can in particular be one of the following compounds (the ASX codes followed by three digits are internal codes used in the context of this document): ASX135 to ASX237 and ASX 401 to ASX453. The structural formulas of these compounds of formula (I) according to the invention are indicated below; they include all isomers and isomeric mixtures:
Dans un mode de réalisation du procédé représenté par ce schéma réactionnel, on approvisionne le composé de formule (III) et le transforme à l'aide de réactifs appropriés successivement en composés (IV), (V), (VI), (VII), (VIII), (IX) et (I). Ce mode de réalisation est expliqué de manière détaillée dans l'exemple 1 ci-dessous ; cet exemple contient à titre d'illustration une liste de composés de formule (I) qui peuvent être obtenus de cette manière (composés ASX135 à ASX237). In one embodiment of the process represented by this reaction scheme, supplying the compound of formula (III) and transforming it with appropriate reagents successively into compounds (IV), (V), (VI), (VII) , (VIII), (IX) and (I). This embodiment is explained in detail in Example 1 below; this example contains by way of illustration a list of compounds of formula (I) which can be obtained in this way (compounds ASX135 to ASX237).
Dans un autre mode de réalisation du procédé représenté par ce schéma réactionnel, on approvisionne le composé (VI) et le transforme successivement à l'aide de réactifs appropriés en composés (VII), (VIII), (IX) et (I). In another embodiment of the process represented by this reaction scheme, the compound (VI) is supplied and is converted successively with appropriate reagents into compounds (VII), (VIII), (IX) and (I).
Dans un autre mode de réalisation, qui conduit à un composé (I) dans lequel R3 est un groupement phosphate, on approvisionne le composé (VII) et le fait réagir avec du diallyl diéthylphosphoramidite pour obtenir le composé (VIII) sous la forme de son diallyl- phosphate, c'est-à-dire que dans ce composé (VIII), l'atome hydrogène de chacune des deux fonctions OH du phosphate R3 est substitué par un reste allyl. Ce composé (VIII) est ensuite transformé successivement en composés (IX) et (I). Ce mode de réalisation est expliqué de manière détaillée dans l'exemple 3 ci-dessous ; cet exemple contient à titre d'illustrration une liste de composés de formule (I) qui peuvent être obtenus de cette manière (composés ASX401 à ASX 453). Pour évaluer leurs effets biologiques, et notamment leur toxicité et leur effet thérapeutique, les composés selon l'invention, obtenus par synthèse selon le procédé décrit ci-dessus, ont été testés de deux manières : d'abord sur des cultures de cellules humaines cancérigènes pour déterminer leur valeur IC50, puis, pour les composés qui paraissent particulièrement prometteuses, sur un modèle animal du cancer du côlon qui implique le suivi de l'évolution de la tumeur en volume tous les trois jours. In another embodiment, which leads to a compound (I) in which R 3 is a phosphate group, the compound (VII) is supplied and reacted with diallyl diethylphosphoramidite to obtain the compound (VIII) in the form of its diallylphosphate, that is to say in this compound (VIII), the hydrogen atom of each of the two OH functions of the phosphate R 3 is substituted by an allyl residue. This compound (VIII) is then successively converted into compounds (IX) and (I). This embodiment is explained in detail in Example 3 below; this example contains by way of illustration a list of compounds of formula (I) which can be obtained in this way (compounds ASX401 to ASX 453). To evaluate their biological effects, and in particular their toxicity and their therapeutic effect, the compounds according to the invention, obtained by synthesis according to the process described above, were tested in two ways: firstly on carcinogenic human cell cultures. to determine their IC 50 value, then, for compounds that appear particularly promising, on an animal model of colon cancer that involves monitoring the evolution of the tumor in volume every three days.
Les inventeurs ont constatés que la toxicité des composés de formule (I) est généralement faible. Cela favorise leur utilisation en tant que médicament. The inventors have found that the toxicity of the compounds of formula (I) is generally low. This promotes their use as a medicine.
Les inventeurs ont par ailleurs constatés que les composés de formule (I) montrent un effet biologique qui permet d'envisager leur utilisation thérapeutique dans des compositions pharmaceutiques. The inventors have furthermore found that the compounds of formula (I) show a biological effect which makes it possible to envisage their therapeutic use in pharmaceutical compositions.
A titre d'exemple, dans un mode de réalisation typique, on utilise le Dibenzyl (S)-1-[(R)- 3,4-bis(benzyloxy)-5-oxo-2,5-dihydrofuran-2-yl]-2-hydroxyéthylphosphate (ASX234), dérivé de l'acide ascorbique (AA), qui s'avère être une molécule cytotoxique pour les cellules cancéreuses, à des concentrations entre 10 et 20 fois inférieures à celle de la molécule originelle (AA). Par contre, le ASX234 s'avère non toxique pour des cellules normales, ce qui est rare dans le traitement du cancer. By way of example, in a typical embodiment, Dibenzyl (S) -1 - [(R) - 3,4-bis (benzyloxy) -5-oxo-2,5-dihydrofuran-2-yl is used. 2-hydroxyethylphosphate (ASX234), derived from ascorbic acid (AA), which is found to be a cytotoxic molecule for cancer cells, at concentrations between 10 and 20 times lower than that of the original molecule (AA) . On the other hand, ASX234 is non-toxic for normal cells, which is rare in the treatment of cancer.
Le traitement d'un modèle animal de cancer du côlon par cette molécule, utilisant des concentrations 250 inférieures à celle de l'acide ascorbique, montre qu'elle ne présente pas de toxicité in vivo sur des cellules non pathologiques et qu'elle ralentie fortement la progression des tumeurs. The treatment of an animal model of colon cancer with this molecule, using concentrations 250 lower than that of ascorbic acid, shows that it exhibits no toxicity in vivo on non-pathological cells and that it decelerates strongly. the progression of tumors.
En conséquence, les inventeurs ont constaté que les composés selon l'invention représentent une nouvelle classe de molécules anti-cancéreuses. Sans vouloir être lié par une quelconque théorie concernant le méanisme d'action des composés selon l'invention, les observations réalisées par les inventeurs dans le cadre de leurs travaux leur ont permis d'identifier au moins un tel mécanisme d'action, qui repose sur le rôle de compétiteurs de l'ATP des composés selon l'invention ; ce nouveau mécanisme, induit une mort cellulaire par nécrose et ne présente pas d'effets secondaires visibles sur des cellules non-cancéreuses. As a result, the inventors have found that the compounds according to the invention represent a new class of anti-cancer molecules. Without wishing to be bound by any theory concerning the action mechanism of the compounds according to the invention, the observations made by the inventors in the course of their work enabled them to identify at least one such mechanism of action, which is based on on the role of competitors of ATP compounds according to the invention; this new mechanism induces cell death by necrosis and has no visible side effects on non-cancerous cells.
D'une manière générale, les composés selon l'invention peuvent être utilisés comme principes actifs dans des composition pharmaceutiques pour le traitement ou la prévention du cancer. Cette utilisation peut mettre en œuvre les composés en tant que tels, et/ou un sel pharmaceutiquement acceptable desdits composés, ledit sel pouvant être un sel d'addition desits composés avec un acide minéral ou organique ou avec une base minérale ou organique. Ainsi, une composition pharmaceutique selon l'invention comprend à titre de principe actif au moins un composé selon l'invention et/ou au moins un sel d'addition selon l'invention. In general, the compounds according to the invention can be used as active ingredients in pharmaceutical compositions for the treatment or prevention of cancer. This use can implement the compounds as such, and / or a pharmaceutically acceptable salt of said compounds, said salt being a salt addition of said compounds with an inorganic or organic acid or with a mineral or organic base. Thus, a pharmaceutical composition according to the invention comprises as active ingredient at least one compound according to the invention and / or at least one addition salt according to the invention.
Lesdites compositions pharmaceutiques peuvent être administrées notamment par voie orale, par exemple sous une forme solide ou, notamment lorsqu'il s'agit de sels, sous une forme soluble dans l'eau et/ou dans un alcool compatible avec l'eau (tel que l'éthanole, le propylène glycol, le polypropylène glycol et le glycérol). Elles peuvent aussi être admini- strées par voie parenterale, par exemple intraveineuse, intra-musculaire, sous-cutanée ou rectale. Elles peuvent comprendre d'autres constituants, notamment des véhicules et/ou excipients pharmaceutiquement acceptables, et/ou d'autres principes actifs. Said pharmaceutical compositions may be administered especially orally, for example in solid form or, especially when it is a question of salts, in a form soluble in water and / or in a alcohol compatible with water (such ethanol, propylene glycol, polypropylene glycol and glycerol). They can also be administered parenterally, for example intravenously, intramuscularly, subcutaneously or rectally. They may include other components, including pharmaceutically acceptable carriers and / or excipients, and / or other active ingredients.
Les cancers dont le traitement et/ou la prévention pourra être envisagée sont de préférence les cancers du poumon, les cancers du sein, les cancers de l'ovaire, les cancers de la prostate, les cancers du pancréas, les cancers du colon, les cancers de la thyroïde, les carninomes rectaux, les glioblastomes, les leucémies myéloïdes chroniques, les leucémies lympho-blastiques aiguës, les mélanomes métastatiques, les carninomes rénaux. Cancers whose treatment and / or prevention may be considered are preferably lung cancers, breast cancers, ovarian cancers, prostate cancers, pancreatic cancers, colon cancers, thyroid cancers, rectal carninomas, glioblastomas, chronic myeloid leukemias, acute lymphoblastic leukemias, metastatic melanomas, renal carninomas.
Exemples Examples
Les exemples qui suivent illustrent des modes de réalisation de l'invention, mais ne limitent pas sa portée. 1. Synthèse The following examples illustrate embodiments of the invention, but do not limit its scope. 1. Summary
Dans ces exemples, « TA » ou « RT » signifient indifféremment « température ambiante » et « Rdt » signifie « rendement ». In these examples, "TA" or "RT" mean indifferently "ambient temperature" and "Yield" means "yield".
Exemple 1 : dibenzyl (S)-1-[(R)-3,4-bis(benzyloxy)-5-oxo-2,5-dihydrofuran-2-yn-2-hydroxy- ethyl phosphate (appelé ici « ASX234 ») : Example 1: dibenzyl (S) -1 - [(R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydrofuran-2-yn-2-hydroxyethyl phosphate (herein referred to as "ASX234" ):
Etape 1 : (R)-5-f(S)-2,2-dimethyl-1 ,3-dioxolan-4-yn-3,4-dihydroxyfuran-2(5H)-one Step 1: (R) -5-f (S) -2,2-dimethyl-1,3-dioxolan-4-yn-3,4-dihydroxyfuran-2 (5H) -one
A une suspension de L(+)- acide ascorbique (10 g, 56,8 mmol) dans l'acétone (50 mL), on a ajouté du chlorure d'éthanoyle CH3COCI (4,4 mL, 62,5 mmol) à TA. Après agitation pendant la nuit, le précipité blanc a été filtré, rincé avec de l'acétone puis séché sous vide pour donner le produit attendu (8,1 g, Rdt 66%). To a suspension of L (+) - ascorbic acid (10 g, 56.8 mmol) in acetone (50 mL) was added CH 3 COCI ethanoyl chloride (4.4 mL, 62.5 mmol). ) at TA. After stirring overnight, the white precipitate was filtered, rinsed with acetone and dried under vacuum to give the expected product (8.1 g, 66% yield).
Caractérisation par RMN 1H (200MHz, DMSO-d6) : 1 ,25 (s, 6H), 3,87 (dd, 1 H, J1 =8,4 Hz, J2=6,4 Hz), 4,09 (t, 1 H, J=8,4 Hz), 4,25 (td, 1 H, J1=6,6 Hz, J2=3,0 Hz), 4,71 (d, 1 H, J=3,0 Hz), 8,49 (s, 1 H), 11 ,30 (s, 1 H). Etape 2: (R)-3,4-bis(benzvloxv)-5-((S)-2,2-dimethyl-1 ,3-dioxolan-4-yl)furan-2(5H)-one Characterization by 1 H NMR (200 MHz, DMSO-d6): 1.25 (s, 6H), 3.87 (dd, 1H, J1 = 8.4 Hz, J2 = 6.4 Hz), 4.09 ( t, 1H, J = 8.4 Hz), 4.25 (td, 1H, J1 = 6.6 Hz, J2 = 3.0 Hz), 4.71 (d, 1H, J = 3, 0 Hz), 8.49 (s, 1H), 11.30 (s, 1H). Step 2: (R) -3,4-bis (benzyloxy) -5 - ((S) -2,2-dimethyl-1,3-dioxolan-4-yl) furan-2 (5H) -one
A une suspension du composé obtenu lors de l'étape 1 (3 g, 13,88 mmol, 1 ,0 éq.) dans du DCM (60 mL), on a ajouté du DIEA (5,5 mL, 33,33 mmol, 2,4 éq.), puis du bromure de benzyle (3,3 mL, 27,76 mmol, 2,0 éq.) à TA. Après agitation pendant la nuit, le milieu réactionnel a été lavé avec de l'eau, séché sur Na2S04, filtré et évaporé. Le résidu obtenu a été purifié sur gel de silice pour donner le produit attendu (2,4g, Rdt 43%). To a suspension of the compound obtained in Step 1 (3 g, 13.88 mmol, 1.0 eq.) In DCM (60 mL) was added DIEA (5.5 mL, 33.33 mmol). , 2,4 eq.), Then bromide of benzyl (3.3 mL, 27.76 mmol, 2.0 eq) at RT. After stirring overnight, the reaction medium was washed with water, dried over Na 2 SO 4 , filtered and evaporated. The residue obtained was purified on silica gel to give the expected product (2.4 g, yield 43%).
Caractérisation par RMN 1H (200MHz, CDCI3) : 1 ,37 (s, 3H), 1 ,41 (s, 3H), 3,9-4,3 (m, 3H), 4,54 (d, 1 H, J=3,2 Hz), 5,0-5,3 (m, 4H), 7,2-7,5 (m, 10H). Characterization by 1 H NMR (200 MHz, CDCl 3 ): 1.37 (s, 3H), 1.41 (s, 3H), 3.9-4.3 (m, 3H), 4.54 (d, 1) H, J = 3.2 Hz), 5.0-5.3 (m, 4H), 7.2-7.5 (m, 10H).
Etape 3: (R)-3,4-bis(benzvloxy)-5-r(S)-1 l2-dihvdroxyethyllfuran-2(5H)-one Step 3: (R) -3,4-bis (benzyloxy) -5-r (S) -1-l 2 dihvdroxyethyllfuran-2 (5H) -one
Un mélange du composé obtenu à l'étape 2 (9,5 g, 24,2 mmol) dans l'acide acétique (50 mL) et de l'eau (15 mL) a été chauffé à 50°C pendant la nuit. Le mélange a été évaporé, neutralisé avec NaHC03 puis extrait avec du DCM. La phase est organique est a été séchée sur Na2S04, filtré et évaporé. Le résidu a été purifié sur gel de silice pour donner le produit attendu (6,8 g, Rdt 80%). A mixture of the compound obtained in step 2 (9.5 g, 24.2 mmol) in acetic acid (50 mL) and water (15 mL) was heated at 50 ° C overnight. The mixture was evaporated, neutralized with NaHCO 3 and extracted with DCM. The organic phase is dried over Na 2 S0 4 , filtered and evaporated. The residue was purified on silica gel to give the expected product (6.8 g, 80% yield).
Caractérisation par RMN H (200MHz, CDCI3) : 2,80 (si, 1 H), 3,07 (si, 1 H), 3,6-4,0 (m, 3H), 4,68 (d, 1 H, J=2,2 Hz), 5,07 (s, 2H), 5,17 (d, 2H, J=3,2 Hz), 7,1-7,4 (m, 10H). Characterization by H NMR (200 MHz, CDCl 3 ): 2.80 (Si, 1H), 3.07 (Si, 1H), 3.6-4.0 (m, 3H), 4.68 (d, 1 H, J = 2.2 Hz), 5.07 (s, 2H), 5.17 (d, 2H, J = 3.2 Hz), 7.1-7.4 (m, 10H).
Etape 4: (R)-3,4-bis(benzyloxy)-5-i(S)-2-(tert-butyldimethylsilyloxy)-1 -hydroxyethynfuran- 2(5H)-one Step 4: (R) -3,4-bis (benzyloxy) -5-i (S) -2- (tert-butyldimethylsilyloxy) -1-hydroxyethynfuran-2 (5H) -one
A une solution du composé obtenu lors de l'étape 3 (4,26 g, 11 ,97 mmol) et d'imidazole (1 ,6 g, 23,93 mmol) dans du DCM (60 mL) à TA sous N2, on a ajouté du TBDMSCI (3,6 g, 23,93 mmol). Après 4h, le mélange réactionnel a été filtré et rincé avec du DCM. Le filtrat a été lavé avec de l'eau puis avec de la saumure, séché sur Na2S04, filtré et évaporé. Le résidu obtenu a été purifié sur gel de silice pour donner le produit attendu (3,7g, Rdt 66%).To a solution of the compound obtained in step 3 (4.26 g, 11.97 mmol) and imidazole (1.6 g, 23.93 mmol) in DCM (60 mL) at RT under N 2 TBDMSCI (3.6 g, 23.93 mmol) was added. After 4h, the reaction mixture was filtered and rinsed with DCM. The filtrate was washed with water and then with brine, dried over Na 2 SO 4 , filtered and evaporated. The residue obtained was purified on silica gel to give the expected product (3.7 g, 66% yield).
Caractérisation par RMN 1H (300MHz, CDCI3) : 0,05 (s 6H), 0,87 (s, 9H), 1 ,94 (d, 1 H, J=7,0 Hz), 3,74 (m, 2H), 3,95 (m, 1 H), 4,73 (s, 1 H), 5,08 (s, 2H), 5,19 (d, 2H, J=4,8 Hz), 7,2-7,5 (m, 10H). Characterization by 1 H NMR (300 MHz, CDCl 3 ): 0.05 (s 6H), 0.87 (s, 9H), 1.94 (d, 1H, J = 7.0 Hz), 3.74 ( m, 2H), 3.95 (m, 1H), 4.73 (s, 1H), 5.08 (s, 2H), 5.19 (d, 2H, J = 4.8 Hz), 7.2-7.5 (m, 10H).
Etape 5: (dibenzyl)-N,N-diethylphosphoramidite Step 5: (dibenzyl) -N, N-diethylphosphoramidite
A une solution de PCI3 (10 g, 72,82 mmol) dans Et20 (300 mL) refroidie à 0°C sous N2, on a ajouté de la triéthylamine (10,1 mL, 72,82 mmol) et de la diéthylamine (7,5 mL, 72,82 mmol). Après avoir agité le milieu pendant une nuit à TA, le précipité obtenu a été filtré et lavé avec Et20. Le filtrat résultant a été placé dans un tricol de 500 mL sous N2 à 0°C. A cette solution, ont été ajoutés de la triéthylamine (22,3 mL, 160,2 mmol) et de l'alcool benzylique (15,1 mL, 145,6 mmol) en gardant T<5°C. Après agitation pendant la nuit, le mélange a été filtré et lavé avec Et20. Le filtrat a été évaporé à sec pour donner le phosphoramidite attendu (23g, Rdt 96%). To a solution of PCI 3 (10 g, 72.82 mmol) in Et 2 O (300 mL) cooled to 0 ° C under N 2 was added triethylamine (10.1 mL, 72.82 mmol) and diethylamine (7.5 mL, 72.82 mmol). After stirring the medium overnight at RT, the resulting precipitate was filtered and washed with Et 2 O. The resulting filtrate was placed in a 500 mL three-neck N 2 at 0 ° C. To this solution was added triethylamine (22.3 mL, 160.2 mmol) and benzyl alcohol (15.1 mL, 145.6 mmol) keeping T <5 ° C. After stirring overnight, the mixture was filtered and washed with Et 2 O. The filtrate was evaporated to dryness to give the expected phosphoramidite (23 g, 96% yield).
Caractérisation par RMN 1H (200MHz, CDCI3) : 1 ,10 (t,6H,J=7,0 Hz), 3,12 (q,2H,J=7,0 Hz), 3,13 (q,2H,J=7,0 Hz), 4,77 (m,3H), 4,91 (d,1 H,J=8,0 Hz), 7,34 (m,10H). Characterization by 1 H NMR (200 MHz, CDCl 3 ): 1.10 (t, 6H, J = 7.0 Hz), 3.12 (q, 2H, J = 7.0 Hz), 3.13 (q, 2H, J = 7.0Hz), 4.77 (m, 3H), 4.91 (d, 1H, J = 8.0Hz), 7.34 (m, 10H).
Etape 6: dibenzyl (S)-1-[(R)-3,4-bis(benzyloxy)-5-oxo-2,5-dihydrofuran-2-vn-2-(tert- butyldimethylsilyloxy)éthyl phosphate Step 6: dibenzyl (S) -1 - [(R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydrofuran-2-yn-2- (tert-butyldimethylsilyloxy) ethyl phosphate
A un mélange du composé obtenu lors de l'étape 4 (2,0 g, 4,25 mmol), on a ajouté du NaHC03 (2,2 g, 20,1 mmol) et du triazole (0,6 g, 8,5 mmol) dans du DCM (40 mL) ; puis on a ajouté une solution du phosphoramidite obtenu lors de l'étape 5 (3,2 g, 10,2 mmol) dans du DCM (60 mL) à TA. Après 6h, le milieu réactionnel a été refroidi à 0-5°C et du H202 (9 mL) a été ajouté lentement. Après agitation pendant la nuit, le milieu a été quenché avec une solution de NaS203 (30 mL). De l'eau a été ajoutée et le produit a été extrait avec du DCM. La phase organique a été séchée sur Na2S04, filtrée et évaporée. Le résidu obtenu a été purifiée sur gel de silice pour donner le produit attendu (2,2g, Rdt 70%). To a mixture of the compound obtained in step 4 (2.0 g, 4.25 mmol) was added NaHCO 3 (2.2 g, 20.1 mmol) and triazole (0.6 g, 8.5 mmol) in DCM (40 mL); then a solution of the phosphoramidite obtained in step 5 (3.2 g, 10.2 mmol) in DCM (60 mL) was added at RT. After 6h, the reaction medium was cooled to 0-5 ° C and H 2 O 2 (9 mL) was slowly added. After stirring overnight, the medium was quenched with a solution of NaS 2 O 3 (30 mL). Water was added and the product was extracted with DCM. The organic phase was dried over Na 2 SO 4 , filtered and evaporated. The residue obtained was purified on silica gel to give the expected product (2.2 g, yield 70%).
Caractérisation par RMN H (200MHz, CDCI3) : 0,02 (s,3H), 0,03 (s,3H), 0,85 (s,9H), 3,80 (m,2H), 4,65 (m,1 H), 4,92 (m,5H), 5,02 (s,2H), 5,16 (d,2H,J=6,0 Hz), 7,2-7,4 (m,10H). Characterization by H NMR (200 MHz, CDCl 3 ): 0.02 (s, 3H), 0.03 (s, 3H), 0.85 (s, 9H), 3.80 (m, 2H), 4.65 (m, 1H), 4.92 (m, 5H), 5.02 (s, 2H), 5.16 (d, 2H, J = 6.0 Hz), 7.2-7.4 (m , 10H).
Etape 7: dibenzyl (S)-1-f(R)-3,4-bis(benzyloxy)-5-oxo-2,5-dihydrofuran-2-vll-2- hydroxyéthyl phosphate Step 7: dibenzyl (S) -1- (R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydrofuran-2-yl-2-hydroxyethyl phosphate
A une solution du composé obtenu lors de l'étape 6 (100 mg, 0,136 mmol) dans du THF (2 mL), on a ajouté du TBAF (1 M in THF, 0,14 mL, 0,136 mmol) à 5-10°C. Après 20 min, la réaction a été quenchée avec une solution de NH4CI et le produit a été extrait avec du DCM. La phase organique a été séchée sur Na2S04, filtrée et évaporée. Le résidu obtenu a été purifiée sur gel de silice donner le produit attendu (8mg, Rdt 10%) To a solution of the compound obtained in step 6 (100 mg, 0.136 mmol) in THF (2 mL), TBAF (1 M in THF, 0.14 mL, 0.136 mmol) was added at 5-10. ° C. After 20 min, the reaction was quenched with NH 4 CI solution and the product was extracted with DCM. The organic phase was dried over Na 2 SO 4 , filtered and evaporated. The residue obtained was purified on silica gel to give the expected product (8 mg, yield 10%)
Caractérisation par RMN 1H (300MHz, CDCI3) : 2,76 (d, 1 H, J=6,9 Hz), 4,00-4,15 (m, 3H), 4,56 (d, 1 H, J=2,1 Hz), 4,95-5,25 (m, 8H), 7,20-7,50 (m, 10H). Les composés suivants sont préparés de façon similaire: Characterization by 1 H NMR (300 MHz, CDCl 3 ): 2.76 (d, 1H, J = 6.9 Hz), 4.00-4.15 (m, 3H), 4.56 (d, 1H) , J = 2.1 Hz), 4.95-5.25 (m, 8H), 7.20-7.50 (m, 10H). The following compounds are prepared in a similar manner:
ASX135, ASX136, ASX137, ASX138, ASX139, ASX140, ASX141 , ASX142, ASX143, ASX144, ASX145, ASX 146, ASX147, ASX148, ASX149, ASX150, ASX151 , ASX152, ASX153, ASX154, ASX155, ASX156, ASX157, ASX158, ASX159, ASX160, ASX161 , ASX162, ASX163, ASX164.ASX165, ASX166, ASX167, ASX168, ASX169, ASX170, ASX171 , ASX172, ASX173, ASX174, ASX175, AXS176, ASX177, ASX178, ASX179, ASX180 ; ASX181 , ASX182, ASX183, ASX184, ASX185, ASX186, ASX187, ASX188, ASX189, ASX190, ASX191 , ASX192, ASX193, ASX194, ASX195, ASX196, ASX197, ASX198, ASX199, ASX200, ASX201 , ASX202, ASX203, ASX204, ASX205, ASX206, ASX207, ASX208, ASX209, ASX210, ASX211 , ASX212, ASX213, ASX214, ASX215, ASX2 6, ASX2 7, ASX218, ASX219, ASX220, ASX221 , ASX222, ASX223, ASX224, ASX225, ASX226, ASX227, ASX228, ASX229, ASX230, ASX231 , ASX232, ASX233, ASX234, ASX235, ASX236, ASX237. Exemple 2: (S)-1-f(R)-3,4-dihvdroxy-5-oxo-2,5-dihydrofuran-2-vn-2-hydroxyethyl dihydroqen phosphate (appelé ici « ASX300 ») ASX135, ASX136, ASX137, ASX138, ASX139, ASX140, ASX141, ASX142, ASX143, ASX144, ASX145, ASX146, ASX147, ASX148, ASX149, ASX150, ASX151, ASX152, ASX153, ASX154, ASX155, ASX156, ASX157, ASX158, ASX159 , ASX160, ASX161, ASX162, ASX163, ASX164.ASX165, ASX166, ASX167, ASX168, ASX169, ASX170, ASX171, ASX172, ASX173, ASX174, ASX175, AXS176, ASX177, ASX178, ASX179, ASX180; ASX181, ASX182, ASX183, ASX184, ASX185, ASX186, ASX187, ASX188, ASX189, ASX190, ASX191, ASX192, ASX193, ASX194, ASX195, ASX196, ASX197, ASX198, ASX199, ASX200, ASX201, ASX202, ASX203, ASX204, ASX205, ASX206, ASX207, ASX208, ASX209, ASX210, ASX211, ASX212, ASX213, ASX214, ASX215, ASX2 6, ASX2 7, ASX218, ASX219, ASX220, ASX221, ASX222, ASX223, ASX224, ASX225, ASX226, ASX227, ASX228, ASX229, ASX230, ASX231, ASX232, ASX233, ASX234, ASX235, ASX236, ASX237. Example 2: (S) -1- (R) -3,4-dihydroxy-5-oxo-2,5-dihydrofuran-2-yn-2-hydroxyethyl dihydroxy phosphate (herein referred to as "ASX300")
Le composé obtenu lors de l'étape 6 de l'exemple 1 (150mg) a été dissout dans du méthanol (5mL). On a ajouté du palladium sur charbon (15mg, 10% w/w) et le milieu a été placé dans un autoclave sous 4 bars d'hydrogène à température ambiante pendant 4h. Le milieu a ensuite filtré sur célite, le méthanol évaporé et le résidu purifié sur phase RP18. Les fractions ont été lyophilisées et on a récupéré 25mg de produit pur sous forme d'huile.The compound obtained in step 6 of Example 1 (150 mg) was dissolved in methanol (5 ml). Palladium on charcoal (15mg, 10% w / w) was added and the medium was placed in an autoclave under 4 bar of hydrogen at room temperature for 4h. The medium was then filtered through Celite, the methanol evaporated and the residue purified on RP18 phase. The fractions were lyophilized and 25 mg of pure product was recovered as an oil.
Caractérisation par RMN 1H (200MHz, CD3OD) : 3,36 (m, 1 H), 3,77 (m, 1 H), 3,91 (m, 1 H) 4,51 (m, 1 H), 4,84 (brs, 4H), 4,98 (m, 1 H). Exemple 3: (S)-1 -[(R)-3,4-bis(benzyloxy)-5-oxo-2,5-dihvdrofuran-2-yl]-2-hvdroxyethyl dihydrogen phosphate (appelé ici « ASX400 ») Characterization by 1 H NMR (200 MHz, CD 3 OD): 3.36 (m, 1H), 3.77 (m, 1H), 3.91 (m, 1H) 4.51 (m, 1H) ), 4.84 (brs, 4H), 4.98 (m, 1H). Example 3: (S) -1 - [(R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydro-furan-2-yl] -2-hydroxyethyl dihydrogen phosphate (herein referred to as "ASX400")
Etape 1 : diallyl diéthylphosphoramidite Step 1: diallyl diethylphosphoramidite
A une solution de PCI3 (10 g, 72,82 mmol) dans Et2O (300 mL) refroidie à 0°C sous N2, on a ajouté de la triethylamine (10,1 mL, 72,82 mmol) et de la diethylamine (7,5 mL, 72,82 mmol). Après avoir agité le milieu pendant une nuit à TA, le précipité obtenu a été filtré et lavé avec Et2O. Le filtrat résultant a été placé dans un tricol de 500 mL sous N2 à 0°C. A cette solution, ont été ajoutés de la triethylamine (22,3 mL, 160,2 mmol) et du allyl alcohol (9,95 mL, 145,6 mmol) en gardant T<5°C. Après agitation pendant la nuit, le mélange a été filtré et lavé avec Et20. Le filtrat a été évaporé à sec pour donner le phosphoramidite attendu (15g, Rdt 96%). To a solution of PCI 3 (10 g, 72.82 mmol) in Et 2 O (300 mL) cooled to 0 ° C under N 2 was added triethylamine (10.1 mL, 72.82 mmol) and diethylamine (7.5 mL, 72.82 mmol). After stirring the medium overnight at RT, the resulting precipitate was filtered and washed with Et 2 O. The resulting filtrate was placed in a 500 mL three-neck N 2 at 0 ° C. To this solution was added triethylamine (22.3 mL, 160.2 mmol) and allyl alcohol (9.95 mL, 145.6 mmol) keeping T <5 ° C. After stirring overnight, the mixture It was filtered and washed with Et 2 O. The filtrate was evaporated to dryness to give the expected phosphoramidite (15 g, yield 96%).
Caractérisation par RMN 1H (300MHz, CDCI3) : 1 ,10 (t, 6H, J=7,0 Hz), 3,12 (m, 4H), 4,18 (m, 4H), 5,18 (m, 2H), 5,35 (m, 2H), 5,95 (m, 2H). Etape 2: diallyl (S)-1-f(R)-3,4-bis(benzyloxy)-5-oxo-2,5-dihydrofuran-2-vn-2-(tert- butyldiméthylsilyloxy)éthyl phosphate Characterization by 1 H NMR (300 MHz, CDCl 3 ): 1.10 (t, 6H, J = 7.0 Hz), 3.12 (m, 4H), 4.18 (m, 4H), 5.18 ( m, 2H), 5.35 (m, 2H), 5.95 (m, 2H). Step 2: Diallyl (S) -1- (R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydrofuran-2-yn-2- (tert-butyldimethylsilyloxy) ethyl phosphate
A une solution du produit de l'étape 4 de l'exemple 1 (500mg, 1.06mmol) dans du DCM (10mL) on a ajouté du triazole (150mg, 2.12mmol), du NaHC03 (535mg, 6.36mmol), puis une solution du composé de l'étape 1 (550mg, 2.55mmol) dans du DCM (15mL) goutte à goutte à TA. Le milieu a été agité sous azote pendant 6h. Le milieu a ensuite été porté à 0°C puis une solution de H202 (2.5mL) dans le THF (25mL) a été ajoutée et le milieu a été laissé sous agitation à TA pendant 18h. Une solution de Na2S203 saturée a été ajoutée goutte à goutte à 0°C. Le mélange a été extrait avec du DCM, les phases organiques ont été séchées au Na2S04 et le solvant a été évaporé. Le résidu a été purifié sur gel de silice (EP/EA 9/1) et le produit pur a été obtenu (260mg, Rdt 46%). To a solution of the product of Step 4 of Example 1 (500mg, 1.06mmol) in DCM (10mL) was added triazole (150mg, 2.12mmol), NaHCO 3 (535mg, 6.36mmol), then a solution of the compound of step 1 (550mg, 2.55mmol) in DCM (15mL) dropwise at RT. The medium was stirred under nitrogen for 6 h. The medium was then brought to 0 ° C. and then a solution of H 2 0 2 (2.5 mL) in THF (25 mL) was added and the medium was stirred at RT for 18 h. Saturated Na 2 S 2 O 3 solution was added dropwise at 0 ° C. The mixture was extracted with DCM, the organic phases were dried with Na 2 S0 4 and the solvent was evaporated. The residue was purified on silica gel (EP / EA 9/1) and the pure product was obtained (260 mg, yield 46%).
Caractérisation par RMN 1H (300MHz,CDCI3): 0,088 (s,6H), 0,89 (s,9H), 3,85 (m,1 H), 3,95 (m,1 H), 4,50 (m,5H), 4,97 (s,1 H), 5,12 (m,2H), 5,29 (m,6H), 5,95 (m,2H), 7,39 (m, 10H). Characterization by 1 H NMR (300 MHz, CDCl 3 ): 0.088 (s, 6H), 0.89 (s, 9H), 3.85 (m, 1H), 3.95 (m, 1H), 4, 50 (m, 5H), 4.97 (s, 1H), 5.12 (m, 2H), 5.29 (m, 6H), 5.95 (m, 2H), 7.39 (m, 10H).
Etape 3: (S)-1-r(R)-3,4-bis(benzyloxy)-5-oxo-2,5-dihvdrofuran-2-vn-2-(tert- butyldimethylsilyloxy)éthyl dihydrogen phosphate Step 3: (S) -1-r (R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydro-furan-2-yn-2- (tert-butyldimethylsilyloxy) ethyl dihydrogen phosphate
A une solution du produit de l'étape 2 (135mg, 214pmol) dans le THF (5mL) on a ajouté de l'acide formique (125μί, 3,3mmol), de la triphenylphosphine (35mg, 128 mol), du n- butylamine (62mg, 857μιηοΙ), puis du palladium tétrakis (25mg, 21 ιηοΙ). Le milieu a été agité sous azote pendant 18h. Le milieu a été purifié sur gel de silice (DCM->DCM/MeOH 9/1) et on a obtenu le produit attendu (40mg, Rdt 34%). To a solution of the product of step 2 (135mg, 214pmol) in THF (5mL) was added formic acid (125μ, 3.3mmol), triphenylphosphine (35mg, 128mol), butylamine (62mg, 857μιηοΙ), then palladium tetrakis (25mg, 21 ιηοΙ). The medium was stirred under nitrogen for 18h. The medium was purified on silica gel (DCM → DCM / MeOH 9/1) and the expected product was obtained (40 mg, yield 34%).
Caractérisation par RMN 1H (300MHz, CDCI3) : 0,088 (s, 6H), 0,89 (s, 9H), 3,95 (m, 2H), 4,25 (m, 1 H), 4,97 (m, 3H), 5,22 (m, 2H), 7,39 (m, 10H), 11 ,50 (brs, 2H). Characterization by 1 H NMR (300 MHz, CDCl 3 ): 0.088 (s, 6H), 0.89 (s, 9H), 3.95 (m, 2H), 4.25 (m, 1H), 4.97 (m, 3H), 5.22 (m, 2H), 7.39 (m, 10H), 11.50 (brs, 2H).
Etape 4: (S)-1-[(R)-3,4-bis(benzyloxy)-5-oxo-2,5-dihydrofuran-2-vn-2-hydroxyéthyl dihydrogen phosphate Step 4: (S) -1 - [(R) -3,4-bis (benzyloxy) -5-oxo-2,5-dihydrofuran-2-yn-2-hydroxyethyl dihydrogen phosphate
A une solution du composé obtenu lors de l'étape 3 (75 mg, 0,136 mmol) dans du THF (2 mL), on a ajouté du TBAF (1 M in THF, 0,14 mL, 0,136 mmol) à 5-10°C. Après 20 min, la réaction a été quenchée avec une solution de NH4CI et le produit a été extrait avec du DCM. La phase organique a été est séchée sur Na2S04, filtrée et évaporée. Le résidu obtenu a été purifié sur gel de silice (DCM->DCM/MeOH 9/1) pour donner le produit attendu (10mg, Rdt 17%) To a solution of the compound obtained in step 3 (75 mg, 0.136 mmol) in THF (2 mL), TBAF (1 M in THF, 0.14 mL, 0.136 mmol) was added at 5-10. ° C. After 20 min, the reaction was quenched with a solution of NH 4 Cl and the product was extracted with DCM. The organic phase was dried over Na 2 SO 4 , filtered and evaporated. The residue obtained was purified on silica gel (DCM → DCM / MeOH 9/1) to give the expected product (10 mg, yield 17%)
Caractérisation par RMN H (300MHz, CDCI3) : 3,85 (m, 1 H), 3,95 (m, 2H), 4,78 (s, 1 H), 4,97 (m, 3H), 5,22 (m, 2H), 7,39 (m, 10H), 11 ,48 (brs, 2H). Characterization by H NMR (300 MHz, CDCl 3 ): 3.85 (m, 1H), 3.95 (m, 2H), 4.78 (s, 1H), 4.97 (m, 3H), , 22 (m, 2H), 7.39 (m, 10H), 11.48 (brs, 2H).
Les composés suivants sont préparés de façon similaire: The following compounds are prepared in a similar manner:
ASX401 , ASX402, ASX403, ASX404, ASX405, ASX406, ASX407, ASX408, ASX409, ASX401, ASX402, ASX403, ASX404, ASX405, ASX406, ASX407, ASX408, ASX409,
ASX410, ASX411 , ASX412, ASX413, ASX414, ASX415, ASX416, ASX417, ASX418, ASX419, ASX420, ASX421 , ASX422, ASX423, ASX424, ASX425, ASX426, ASX427,ASX410, ASX411, ASX412, ASX413, ASX414, ASX415, ASX416, ASX417, ASX418, ASX419, ASX420, ASX421, ASX422, ASX423, ASX424, ASX425, ASX426, ASX427,
ASX428, ASX42, ASX430, ASX431 , ASX432, ASX433, ASX434, ASX435, ASX436,ASX428, ASX42, ASX430, ASX431, ASX432, ASX433, ASX434, ASX435, ASX436,
ASX437, ASX438, ASX439, ASX440, ASX441 , ASX442, ASX443, ASX444, ASX445, ASX446, ASX447, ASX448, ASX449, ASX450, ASX451 , ASX452, ASX453. 2. Activité biologique ASX437, ASX438, ASX439, ASX440, ASX441, ASX442, ASX443, ASX444, ASX445, ASX446, ASX447, ASX448, ASX449, ASX450, ASX451, ASX452, ASX453. 2. Biological activity
Dans le but de tester l'effet anti-prolifératif des analogues de l'acide ascorbique, on a traité in vitro par ces molécules différentes lignées cancéreuses, cellules échappant au contrôle du cycle cellulaire. Le test MTT, permettant d'étudier la viabilité cellulaire, a été utilisé. In order to test the anti-proliferative effect of the ascorbic acid analogues, various cancer lines, cells outside the control of the cell cycle, have been treated in vitro with these molecules. The MTT test, to study cell viability, was used.
Dans ce cadre, on a utilisé des cellules dérivées de lymphomes, de myélomes multiples et de cancer du côlon. Ces lignées, obtenues auprès de l'ATCC (American Type Culture Collection), sont reconnues comme représentatives de ces cancers. In this context, cells derived from lymphoma, multiple myeloma and colon cancer were used. These lines, obtained from the American Type Culture Collection (ATCC), are recognized as representative of these cancers.
La première lignée étudiée est dérivée d'un plasmocytome (RPMI 8226), responsable de myélome multiple, qui est une multiplication anormale des plasmocytes 'variété de globules blancs qui synthétisent les anticorps circulant) dans la moelle osseuse. The first line studied is derived from a plasmocytoma (RPMI 8226), responsible for multiple myeloma, which is an abnormal multiplication of plasma cells (a variety of white blood cells that synthesize circulating antibodies) in the bone marrow.
La seconde lignée cancéreuse (Raji) est dérivée de cellules de lymphome de Burkitt. Ce lymphome constituant une tumeur maligne particulièrement agressive est devenu un modèle d'oncogénèse des lymphomes. The second cancer line (Raji) is derived from Burkitt's lymphoma cells. This lymphoma constituting a particularly aggressive malignant tumor has become a model of oncogenesis of lymphomas.
La troisième lignée est issue d'un adénocarcinome du côlon (HT29c2) qui représente le cancer le plus fréquent en France (plus de 15% de l'ensemble des cancers) ce qui en fait un problème majeur de santé publique. The third line is derived from an adenocarcinoma of the colon (HT29c2), which is the most common cancer in France (more than 15% of all cancers), which makes it a major public health problem.
Deux molécules ont été testées dans ces conditions : (ASX234 et AA). Two molecules were tested under these conditions: (ASX234 and AA).
Nous décrivons ici la méthodologie des essais. I - Test in vitro Here we describe the test methodology. I - In vitro test
- Cytotoxicité :10 000 cellules ont été incubées dans des puits de plaques de microtitration aveclOO μΙ de milieu de culture O/N à 37°C, 5% C02. Le milieu de plating a été remplacé par 100 μΙ / puits des molécules à tester. Les cellules ont été incubées 24h, à 37°C, 5% C02 La viabilité cellulaire a été évaluée par le test MTT décrit par Mosmann en 1983, bien connu de l'homme du métier, en utilisant un lecteur de microplaque (Victor4x de la société Perkin Elmer). Cytotoxicity: 10,000 cells were incubated in wells of microtiter plates with 100 μl of O / N culture medium at 37 ° C., 5% CO 2 . The plating medium was replaced by 100 μl / well of the test molecules. The cells were incubated 24h, at 37 ° C., 5% CO 2. The cell viability was evaluated by the MTT test described by Mosmann in 1983, well known to those skilled in the art, using a microplate reader (Victor4x). the company Perkin Elmer).
- Prolifération : On a utilisé le même protocole d'incubation avec les cellules. La prolifération a été évaluée à l'aide du kit de prolifération Déifia de la société Perkin Elmer, en utilisant le protocole recommandé par le fournisseur. Proliferation: The same incubation protocol was used with the cells. Proliferation was evaluated using the Perkin Elmer Deifia Proliferation Kit, using the protocol recommended by the supplier.
II - Tests in vivo, sur modèles animaux. II - In vivo tests, on animal models.
- On a utilisé 20 souris Femelles Athymics Nude (Harlan) âgées de 8 semaines. 3 x 106 cellules (HT29, lignée de cellules d'adénocarcinome colique humain) resuspendues dans du PBS, ont été injectées, en sous cutané, dans un volume de 0,2 ml par souris. Les souris ont été maintenues dans un environnement stérile durant tout l'essai. Après 20 jours, quand les tailles des tumeurs étaient comprises entre 300 et 500mm3, les animaux ont été randomisés en 2 groupes (tailles moyennes homogènes), un groupe traité (T) et un groupe placebo (P). Le groupe T a alors reçu une injection IP quotidienne de la molécule ASX234 à 6mg/kg/j (dont 25% DMSO), tandis que le groupe P a reçu une injection IP quotidienne de NaCI (0,9%) (dont 25% DMSO). Le volume des tumeurs a été évalué tous les trois jours, en utilisant un pied à coulisse électronique. Après 2 semaines de traitement les animaux ont été sacrifiés, les tumeurs prélevées et pesées. 20 female Athymic Nude (Harlan) mice, 8 weeks old, were used. 3 x 10 6 cells (HT29, human colonic adenocarcinoma cell line) resuspended in PBS, were injected subcutaneously in a volume of 0.2 ml per mouse. Mice were maintained in a sterile environment throughout the test. After 20 days, when the tumor sizes were between 300 and 500 mm 3 , the animals were randomized into 2 groups (homogeneous average sizes), a treated group (T) and a placebo group (P). The T group then received a daily IP injection of the ASX234 molecule at 6 mg / kg / day (of which 25% DMSO), while the P group received a daily IP injection of NaCl (0.9%) (25% DMSO). Tumor volume was evaluated every three days, using an electronic caliper. After 2 weeks of treatment the animals were sacrificed, the tumors removed and weighed.
- Traitement statistique : Les données ont été évaluées quand à leur significativité statistique par le test de Mann-Whitney. Conformément aux conventions internationales, les données ont été considérées comme significatives lorsque la valeur de p était inférieure à 0,05. - Statistical treatment: The data were evaluated when their statistical significance by the Mann-Whitney test. In accordance with international conventions, the data were considered significant when the value of p was less than 0.05.
Nous décrivons ici les résultats des essais. Here we describe the results of the tests.
I - Impact du traitement sur des cellules tumorales humaines I - Impact of treatment on human tumor cells
Des lignées cellulaires, issues de cellules cancéreuses de patients, atteints de tumeurs solides (foie, colon) ou de tumeurs liquides (myélome, lymphomes), ont été traitées par des concentrations croissantes soit de la molécule ASX234 soit d'acide ascorbique (AA). La mortalité cellulaire ainsi que la prolifération cellulaire ont été évaluées. Il ressort des résultats montrée sur la figure 1 que la molécule ASX234 présenté à la fois un effet cytotoxique pour les cellules cancéreuses et un effet antiprolifératif ; cette molécule est entre 10 et 20 fois plus active que ΓΑΑ lui-même. Cell lines, derived from cancer cells of patients, with solid tumors (liver, colon) or liquid tumors (myeloma, lymphomas), were treated with increasing concentrations of either the ASX234 or ascorbic acid (AA) molecule. . Cell death and cell proliferation were assessed. It appears from the results shown in FIG. 1 that the ASX234 molecule presented at one and the same time an effect cytotoxic for cancer cells and an antiproliferative effect; this molecule is between 10 and 20 times more active than itself.
On sait (Belin et al, 2009) que ΓΑΑ n'agit que sur les cellules cancéreuses. On a donc évalué la toxicité du ASX234 sur des cultures de cellules primaires humaines (fibroblastes et hépatocytes primaires). Il ressort des résultats rassemblées sur la figure 2, qu'à des concentrations 2 à 3 fois plus élevées que l'IC50 de cytotoxicité observé sur les cellules cancéreuses, cette molécule n'est pas toxique pour des cellules normales, non pathologiques. Ces données, ainsi que leur traitement statistique, sont présentées sur le tableau 1. It is known (Belin et al, 2009) that ΓΑΑ acts only on cancer cells. The toxicity of ASX234 was therefore assessed on human primary cell cultures (fibroblasts and primary hepatocytes). From the results gathered in FIG. 2, it appears that at concentrations 2 to 3 times higher than the IC50 of cytotoxicity observed on the cancer cells, this molecule is not toxic for normal, non-pathological cells. These data, as well as their statistical treatment, are presented in Table 1.
Tableau 1 Table 1
Il - Impact du traitement par l'AA sur des modèles animaux de cancer It - Impact of AA treatment on animal models of cancer
Les preuves in vitro n'étant qu'une indication, on a testé l'efficacité potentielle des molécules sur un modèle in vivo du cancer le plus fréquent chez l'homme, le cancer du colon. Pour cela on a xénogréffé des souris de type nude immunodéficiente avec des cellules d'adénocarcinome colique (HT29). Les souris, porteuses de tumeurs visibles, ont été traitées soit avec un placebo soit avec du ASX234, à une concentration 250 fois inférieure à la concentration efficace de ΓΑΑ (Belin et al, 2009). La progression de la tumeur, ainsi que le poids final, ont été évalués. Les résultats présentés sur la figure 3 et le tableau 2 montrent que la progression des tumeurs a été significative ment ralentie par le traitement au ASX234. The in vitro evidence being only an indication, the potential efficacy of the molecules was tested on an in vivo model of the most common cancer in humans, colon cancer. For this purpose, node-immunodeficient nude mice were xenographed with colonic adenocarcinoma cells (HT29). Mice with visible tumors were treated either with placebo or with ASX234 at a concentration 250-fold lower than the effective concentration of ΓΑΑ (Belin et al, 2009). The progression of the tumor, as well as the final weight, were evaluated. The results shown in Figure 3 and Table 2 show that tumor progression was significantly slowed by ASX234 treatment.
Claims
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| FR1102504 | 2011-08-11 | ||
| FR1102504A FR2978963A1 (en) | 2011-08-11 | 2011-08-11 | NOVEL DERIVATIVES OF FURANONES AND PHARMACEUTICAL COMPOSITION CONTAINING SAME |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| WO2013021105A2 true WO2013021105A2 (en) | 2013-02-14 |
| WO2013021105A3 WO2013021105A3 (en) | 2013-07-18 |
Family
ID=47022940
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/FR2012/000335 Ceased WO2013021105A2 (en) | 2011-08-11 | 2012-08-06 | Novel derivatives of furanones and pharmaceutical composition containing same |
Country Status (2)
| Country | Link |
|---|---|
| FR (1) | FR2978963A1 (en) |
| WO (1) | WO2013021105A2 (en) |
Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0202589A2 (en) | 1985-05-17 | 1986-11-26 | Takeda Chemical Industries, Ltd. | Pharmaceutical compositions containing ascorbic acid derivatives |
| EP0339486A2 (en) | 1988-04-25 | 1989-11-02 | Takeda Chemical Industries, Ltd. | Ester of ascorbic acid 2-phosphate |
| WO1991016055A1 (en) | 1990-04-17 | 1991-10-31 | Allergan, Inc. | 2(5h)-furanones substituted in the 5 and or in the 4 position, as anti-inflammatory agents |
| US5278189A (en) | 1990-06-04 | 1994-01-11 | Rath Matthias W | Prevention and treatment of occlusive cardiovascular disease with ascorbate and substances that inhibit the binding of lipoprotein (A) |
| EP0579769A1 (en) | 1991-04-30 | 1994-01-26 | Allergan, Inc. | 2- and 5-alkyl and phenyl substituted 4-(1-hydroxy, 1-acyloxy or 1-carbamoyloxy)-5-hydroxy-2(5h)-furanones as anti-inflammatory agents |
| US5541221A (en) | 1992-04-24 | 1996-07-30 | Allergan | Pharmaceutical compositions and method for administering 3 and 4-substituted 2(5H)-furanones to a mammal for inhibiting bone loss |
| EP0768314A1 (en) | 1995-10-13 | 1997-04-16 | Senju Pharmaceutical Co., Ltd. | Ascorbic acid tocopheryl phosphate diesters |
| EP0799218A1 (en) | 1994-12-21 | 1997-10-08 | Merck Frosst Canada Inc. | Diaryl-2-(5h)-furanones as cox-2 inhibitors |
| EP0875514A1 (en) | 1997-04-30 | 1998-11-04 | Showa Denko Kabushiki Kaisha | Ascorbic acid derivative and vitamin C preparation containing the same |
| EP0875246A1 (en) | 1997-04-04 | 1998-11-04 | Showa Denko Kabushiki Kaisha | Pharmaceutical preparation of ascorbic acid derivatives for medical treatment of cancer |
| EP1106175A1 (en) | 1998-08-10 | 2001-06-13 | Nippon Hypox Laboratories Incorporated | Anti-inflammatory analgesics |
| EP1145710A1 (en) | 2000-04-10 | 2001-10-17 | L'oreal | Use of ascorbic acid derivatives for increasing epidermal ceramides synthesis |
| WO2004006911A2 (en) | 2002-07-16 | 2004-01-22 | Universite De La Mediterranee | Compositions intended for the treatment of peripheral neuropathies, preparation thereof and uses of same |
| EP1409494A1 (en) | 2001-07-26 | 2004-04-21 | Tagra Biotechnologies Ltd | Stabilized derivatives of ascorbic acid -3- phosphate |
| EP1496889A1 (en) | 2002-04-12 | 2005-01-19 | Showa Denko K.K. | Stabilized ascorbic acid derivatives |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| BR112012005247A2 (en) * | 2009-09-14 | 2019-09-24 | Univ Hokkaido Nat Univ Corp | antifitoviral agent |
-
2011
- 2011-08-11 FR FR1102504A patent/FR2978963A1/en not_active Withdrawn
-
2012
- 2012-08-06 WO PCT/FR2012/000335 patent/WO2013021105A2/en not_active Ceased
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP0202589A2 (en) | 1985-05-17 | 1986-11-26 | Takeda Chemical Industries, Ltd. | Pharmaceutical compositions containing ascorbic acid derivatives |
| EP0339486A2 (en) | 1988-04-25 | 1989-11-02 | Takeda Chemical Industries, Ltd. | Ester of ascorbic acid 2-phosphate |
| WO1991016055A1 (en) | 1990-04-17 | 1991-10-31 | Allergan, Inc. | 2(5h)-furanones substituted in the 5 and or in the 4 position, as anti-inflammatory agents |
| US5278189A (en) | 1990-06-04 | 1994-01-11 | Rath Matthias W | Prevention and treatment of occlusive cardiovascular disease with ascorbate and substances that inhibit the binding of lipoprotein (A) |
| EP0579769A1 (en) | 1991-04-30 | 1994-01-26 | Allergan, Inc. | 2- and 5-alkyl and phenyl substituted 4-(1-hydroxy, 1-acyloxy or 1-carbamoyloxy)-5-hydroxy-2(5h)-furanones as anti-inflammatory agents |
| US5541221A (en) | 1992-04-24 | 1996-07-30 | Allergan | Pharmaceutical compositions and method for administering 3 and 4-substituted 2(5H)-furanones to a mammal for inhibiting bone loss |
| EP0799218A1 (en) | 1994-12-21 | 1997-10-08 | Merck Frosst Canada Inc. | Diaryl-2-(5h)-furanones as cox-2 inhibitors |
| EP0768314A1 (en) | 1995-10-13 | 1997-04-16 | Senju Pharmaceutical Co., Ltd. | Ascorbic acid tocopheryl phosphate diesters |
| EP0875246A1 (en) | 1997-04-04 | 1998-11-04 | Showa Denko Kabushiki Kaisha | Pharmaceutical preparation of ascorbic acid derivatives for medical treatment of cancer |
| EP0875514A1 (en) | 1997-04-30 | 1998-11-04 | Showa Denko Kabushiki Kaisha | Ascorbic acid derivative and vitamin C preparation containing the same |
| EP1106175A1 (en) | 1998-08-10 | 2001-06-13 | Nippon Hypox Laboratories Incorporated | Anti-inflammatory analgesics |
| EP1145710A1 (en) | 2000-04-10 | 2001-10-17 | L'oreal | Use of ascorbic acid derivatives for increasing epidermal ceramides synthesis |
| EP1409494A1 (en) | 2001-07-26 | 2004-04-21 | Tagra Biotechnologies Ltd | Stabilized derivatives of ascorbic acid -3- phosphate |
| EP1496889A1 (en) | 2002-04-12 | 2005-01-19 | Showa Denko K.K. | Stabilized ascorbic acid derivatives |
| WO2004006911A2 (en) | 2002-07-16 | 2004-01-22 | Universite De La Mediterranee | Compositions intended for the treatment of peripheral neuropathies, preparation thereof and uses of same |
Non-Patent Citations (7)
| Title |
|---|
| E.PASSAGE ET AL., NATURE MEDICINE, 2004 |
| GAZIVODA ET AL.: "Synthesis, structural studies, and cytostatic evaluation of 5,6-di-O-modified L-ascorbic acide derivatives", CARBOHYDRATE RESEARCH, vol. 341, 2006, pages 433 - 442 |
| J.MICALEFF ET AL.: "Effect of ascorbic acid in patients with Cjharcot-Marie-Tooth disease type 1A : a multicentre, randomised, double-blind, placebo- cotnrolled trial", LANCET NEUROL., 2009, pages 1103 - 10 |
| KAYA ET AL., FEBS LETTERS, vol. 528, 2008, pages 3614 - 1618 |
| RATH; PAULING: "Hypothesis : Lipoprotein(a) is a surrogate for ascorbate", PROC. NATL. ACAD. SCI. USA, vol. 87, 1990, pages 6204 - 6207, XP000961237, DOI: doi:10.1073/pnas.87.16.6204 |
| S. BELIN ET AL.: "Antiproliferative effect of Ascorbic Acid Is Associated with the Inhibition of Genes Necessary to Cell Cyle Progression", PLOSONE, vol. 4, no. 2, February 2009 (2009-02-01), pages E4409 |
| SMALL MOLECULES O; S. BELIN, F. KAYA ET AL.: "Ascorbic Acid and Gene Expression : Another Example of Regulation of Gene Expression", CURRENT GENOMICS, vol. 11, 2010, pages 52 - 57 |
Also Published As
| Publication number | Publication date |
|---|---|
| FR2978963A1 (en) | 2013-02-15 |
| WO2013021105A3 (en) | 2013-07-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1727544A1 (en) | Use of indazolecarboxamide derivatives for the preparation of a medicament that is intended for the treatment and prevention of paludism | |
| EP3052500B1 (en) | Inhibitors of 5'-nucleotidases and therapeutic uses thereof | |
| EP2240501B1 (en) | Hydroxy-bisphosphonic acid derivatives as vector for targeting bone tissue | |
| EP3418273A1 (en) | Flavagline derivatives | |
| FR3064268A1 (en) | ACYCLONUCLEOSIDES PHOSPHONATES, THEIR PRODUCTS, AND THEIR USE AS MEDICAMENTS. | |
| EP3129014B1 (en) | Novel organoselenium compounds, method for producing same, and pharmaceutical uses thereof as antitumour agents | |
| EP2321323B9 (en) | Dimeric derivatives of artemisinin and application in anti-cancer therapy | |
| CA2342990A1 (en) | Artemisinin derivatives, method for the preparation thereof and pharmaceutical compositions containing the same | |
| WO2019213335A1 (en) | Orally bioavailable prodrugs of edaravone with altered pharmacokinetic properties and methods of use thereof | |
| KR101975299B1 (en) | Compounds containing core structure of indole acetic acid and uses thereof | |
| CN1901901B (en) | Taxoid-fatty acid conjugates and pharmaceutical compositions thereof | |
| CN109369554B (en) | A kind of substituted heterocyclic compound containing hydroxamic acid and its preparation method and use | |
| EP0364350B1 (en) | 4-Methyl-5[2-(4-phenyl-1-piperazinyl)-ethyl] thiazole derivatives, their preparation and pharmaceutical compositions containing them | |
| WO2013021105A2 (en) | Novel derivatives of furanones and pharmaceutical composition containing same | |
| FR2815033A1 (en) | 7-CARBOXY-FLAVONES DERIVATIVES, PORCEDIA FOR THEIR PREPARATION AND THERAPEUTIC APPLICATION | |
| CN115703727A (en) | A kind of hydrogen persulfide prodrug and its pharmaceutical application | |
| WO2013117870A1 (en) | Methods and compositions for treating cancer | |
| JP6608605B2 (en) | Sirtuin expression enhancer | |
| JP2009062314A (en) | Autoimmune disease-treating agent | |
| JP7076090B2 (en) | New organogermanium compounds and analgesics containing them | |
| EP3237015B1 (en) | Hydrosoluble hydroxybisphosphonic doxorubicine derivatives | |
| GB2494595A (en) | Phenylbutyryl curcumin derivatives and uses for preparing anti-tumor drugs thereof | |
| WO2011070152A2 (en) | Novel antiviral compounds, which are aryl 3-deazauridine analogues at c3 position | |
| KR20070071027A (en) | Water-soluble prodrug compounds comprising paclitaxel or derivatives thereof and preparation method thereof | |
| WO2010049451A2 (en) | C-glycosidic analogues of alpha-galactosylceramids having immunostimulating and immunoregulating properties |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 12773005 Country of ref document: EP Kind code of ref document: A2 |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 12773005 Country of ref document: EP Kind code of ref document: A2 |