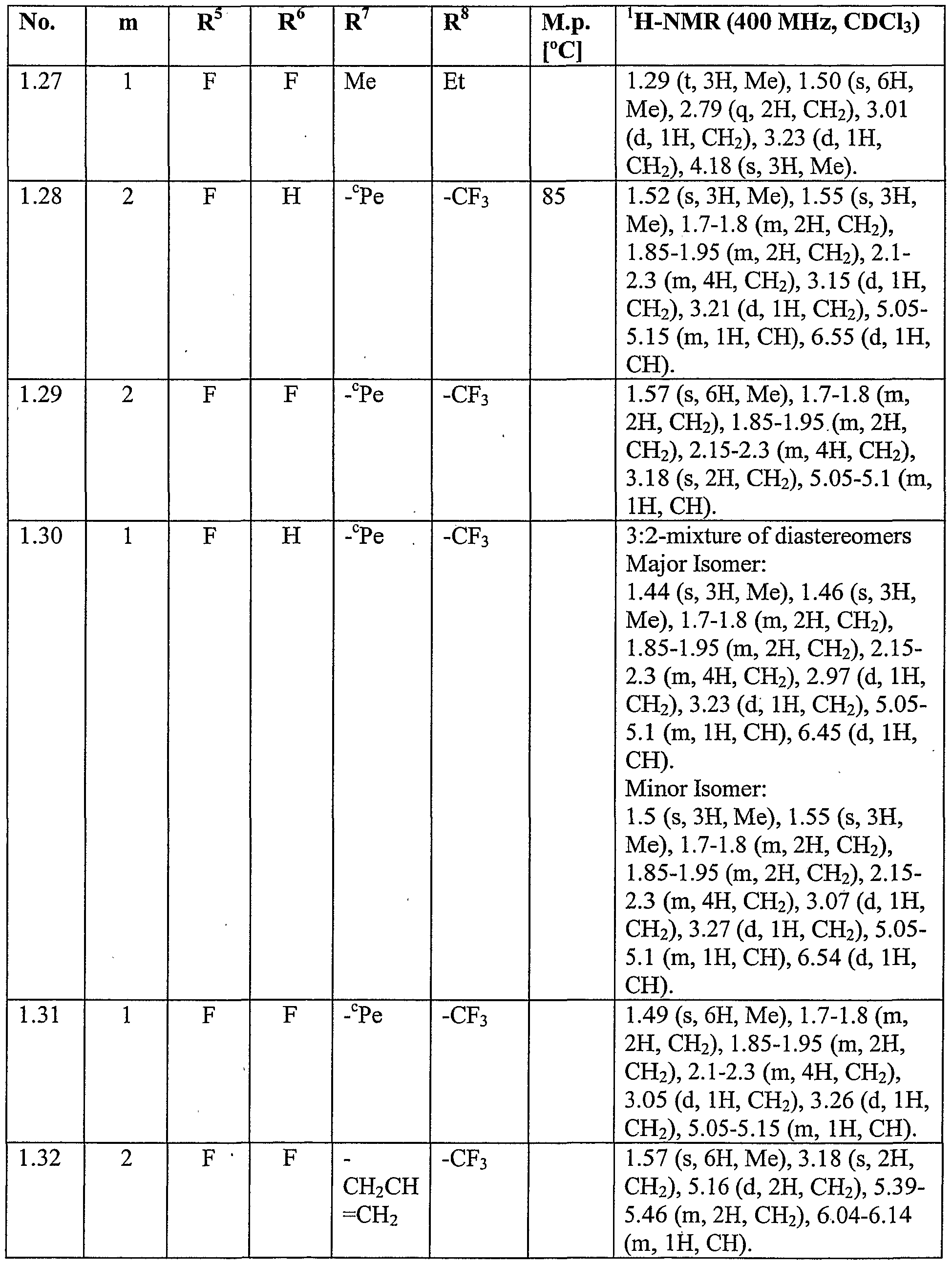
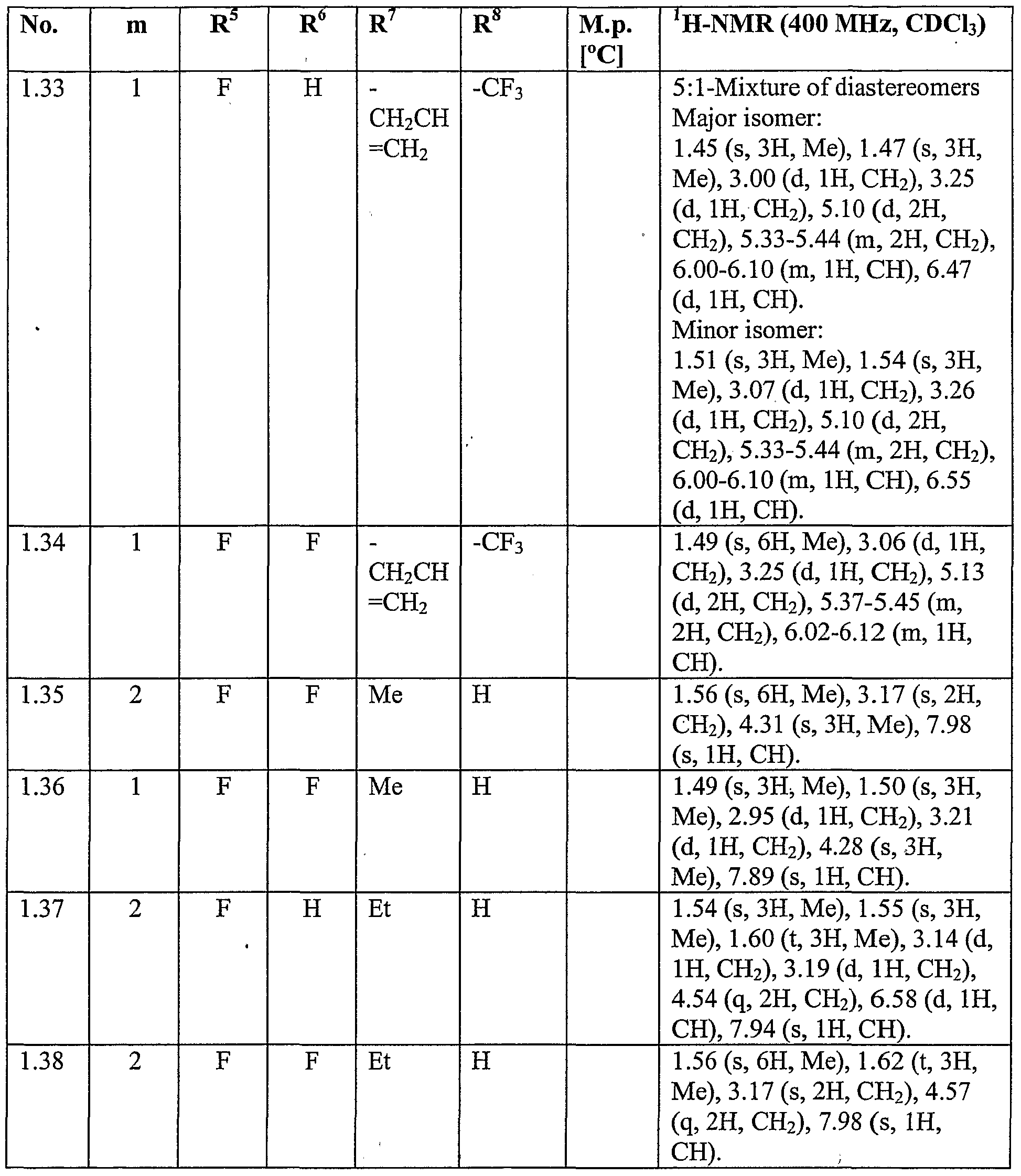
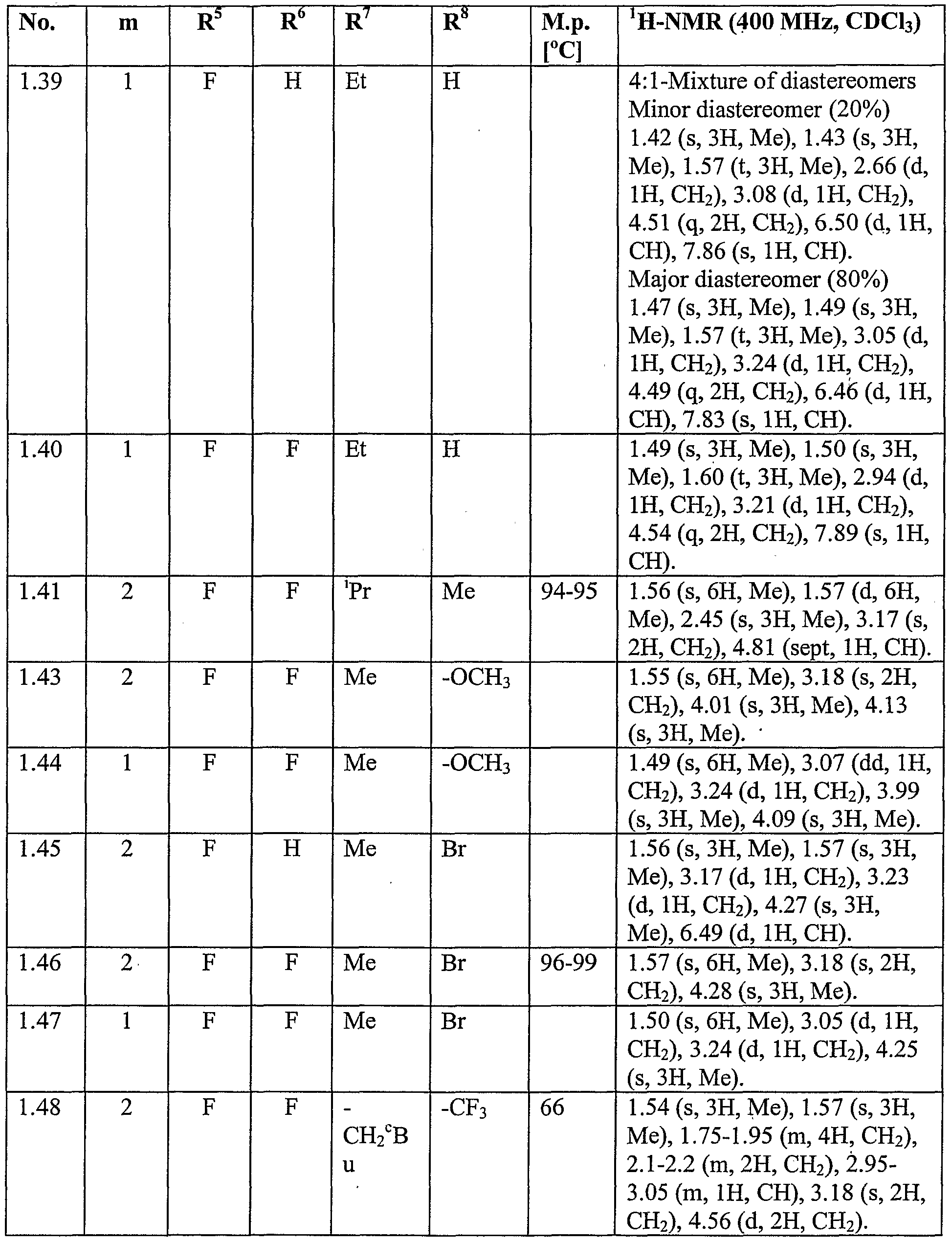
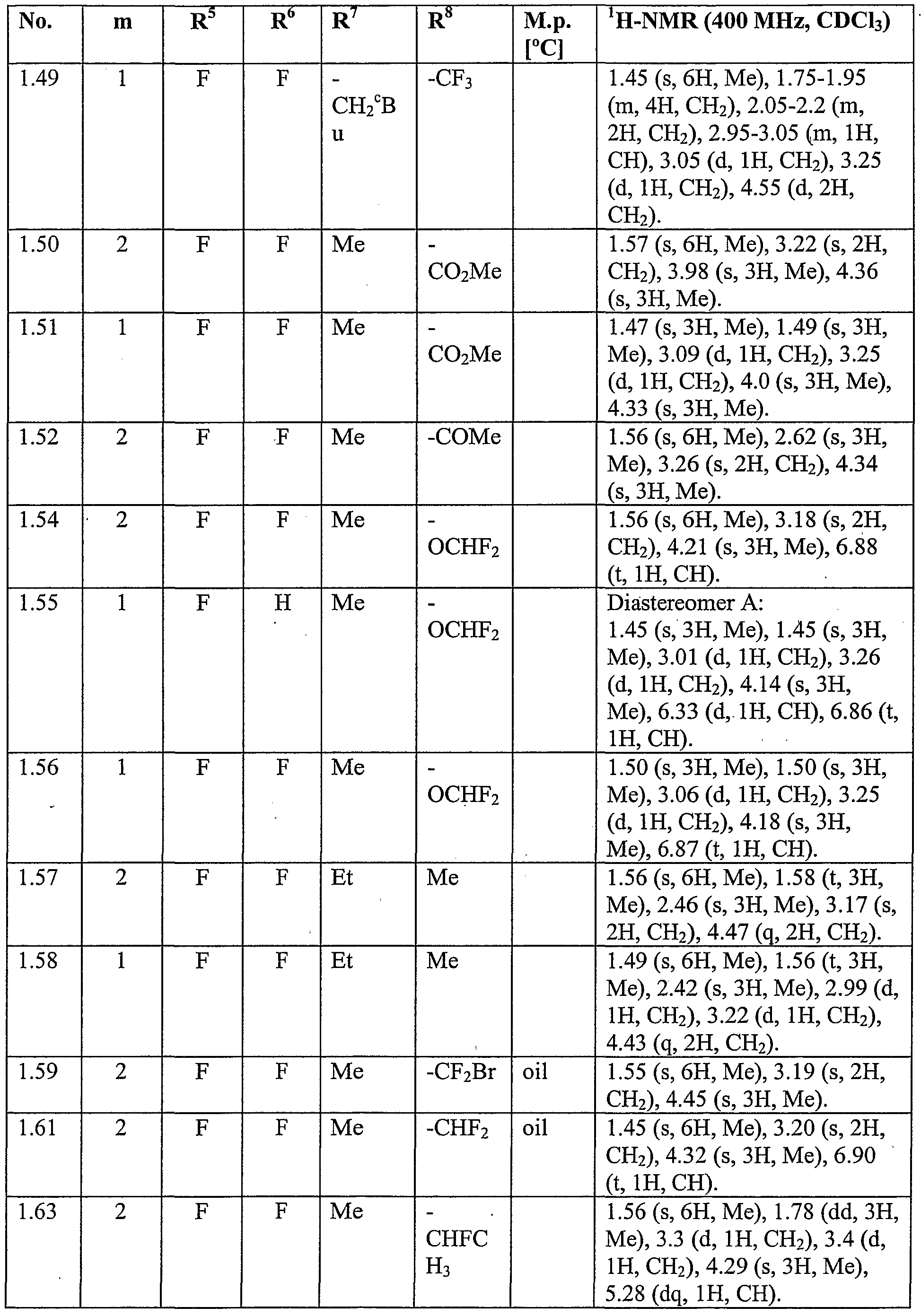
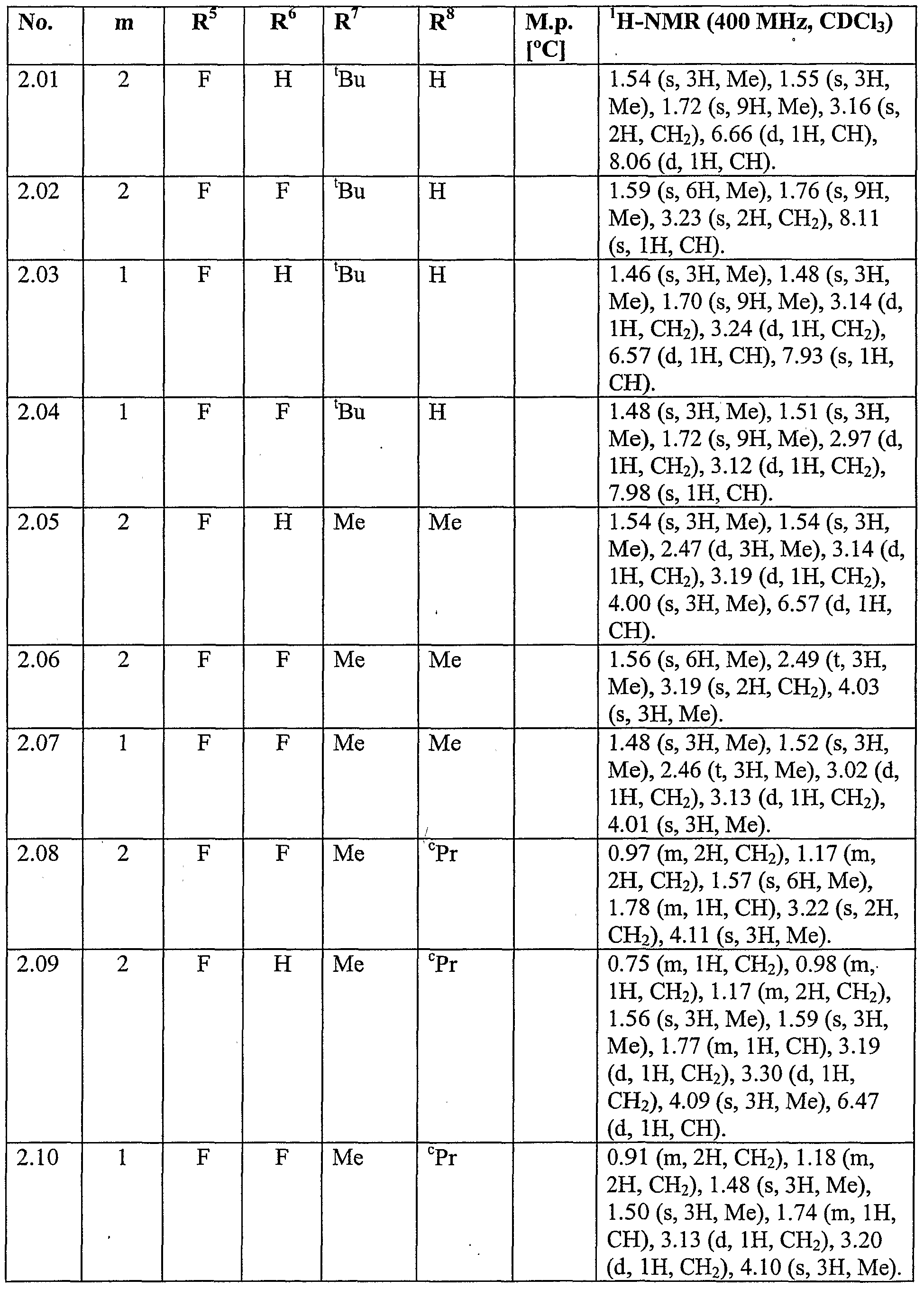
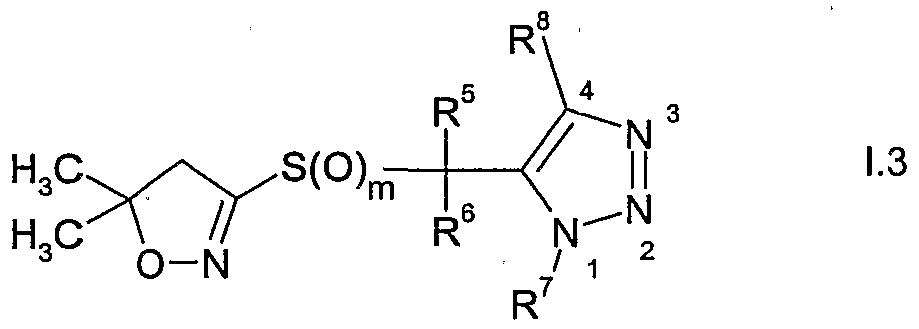
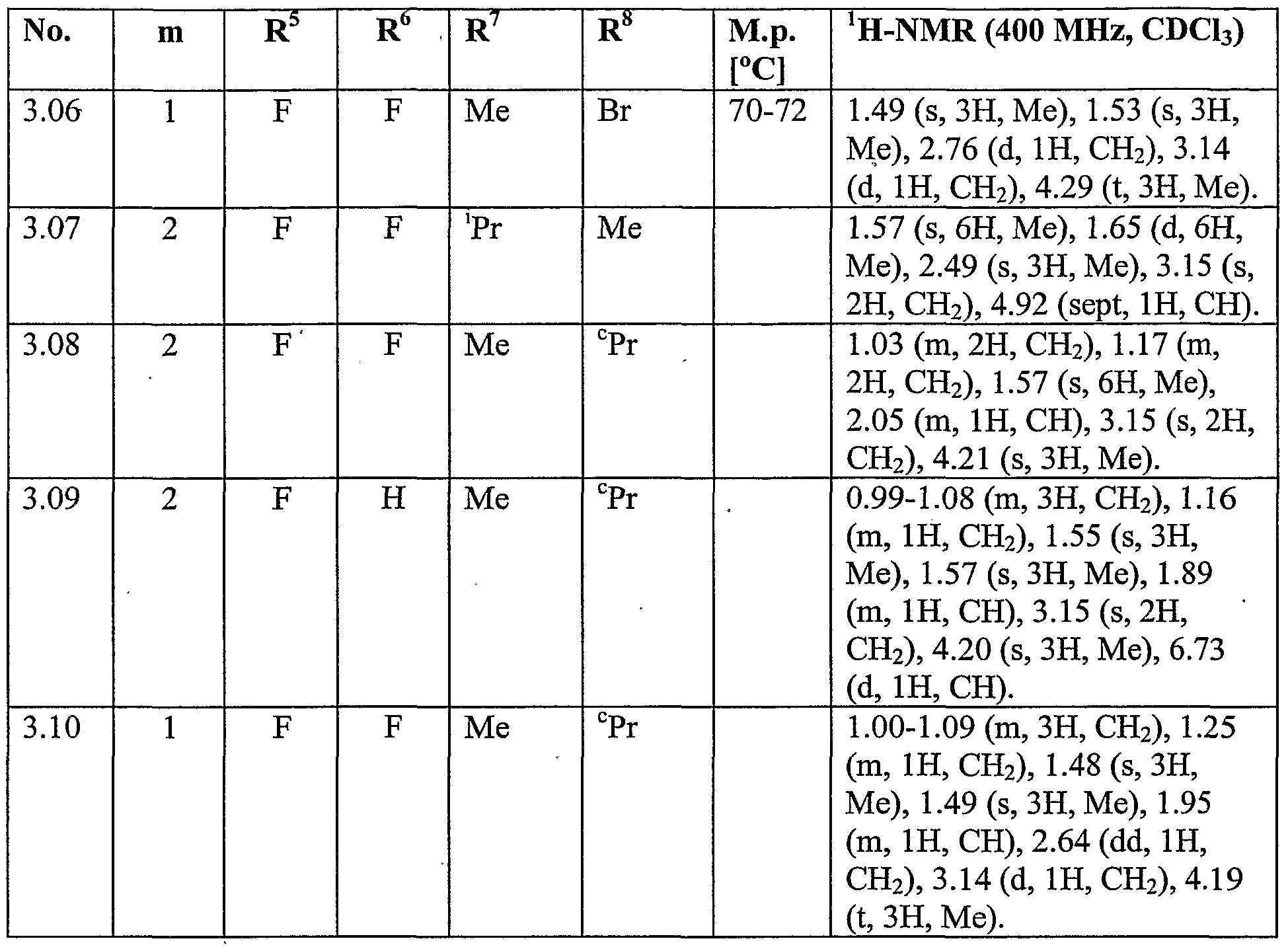
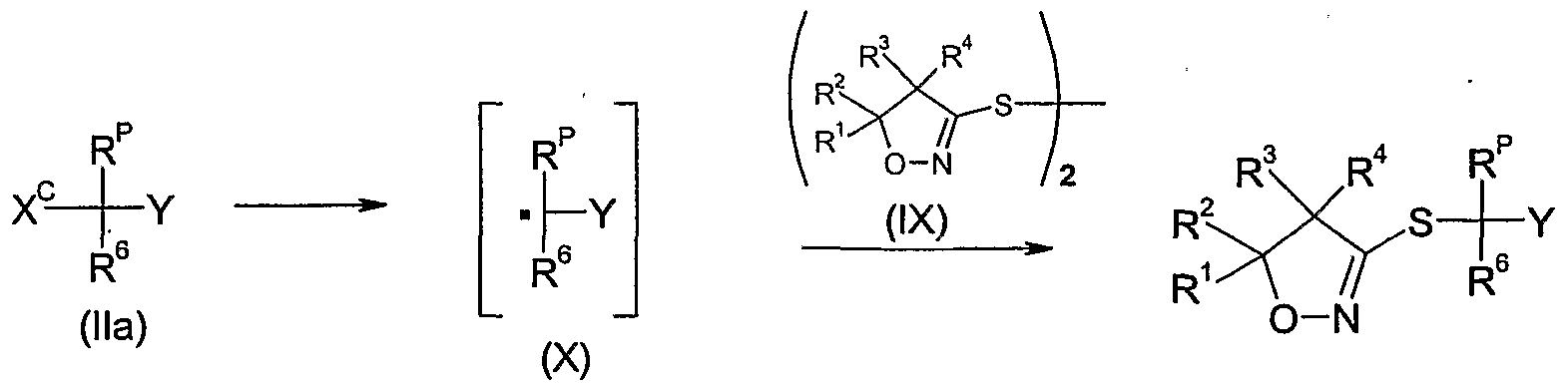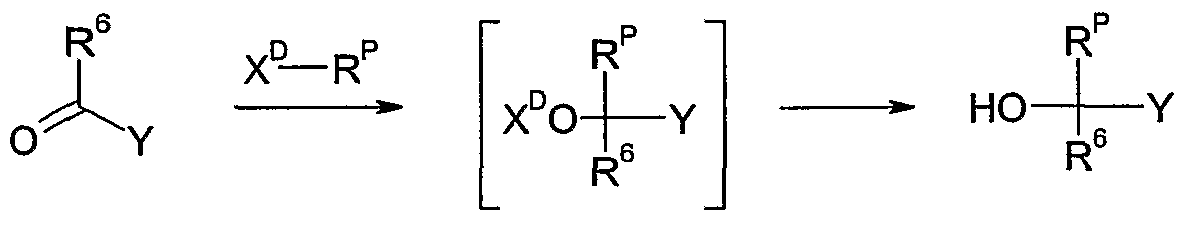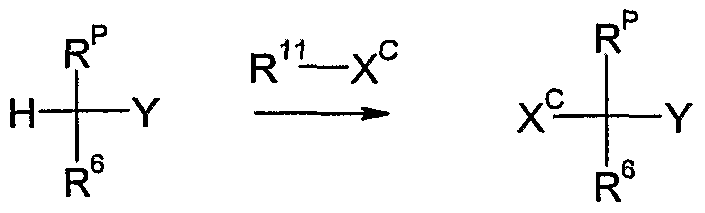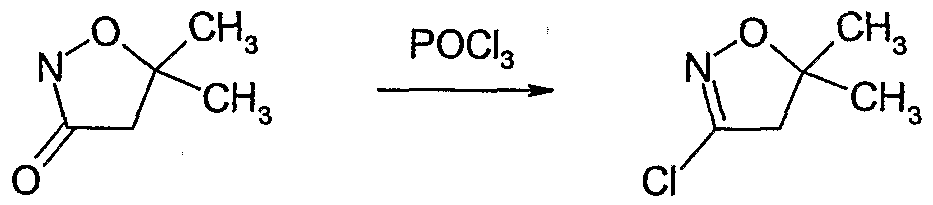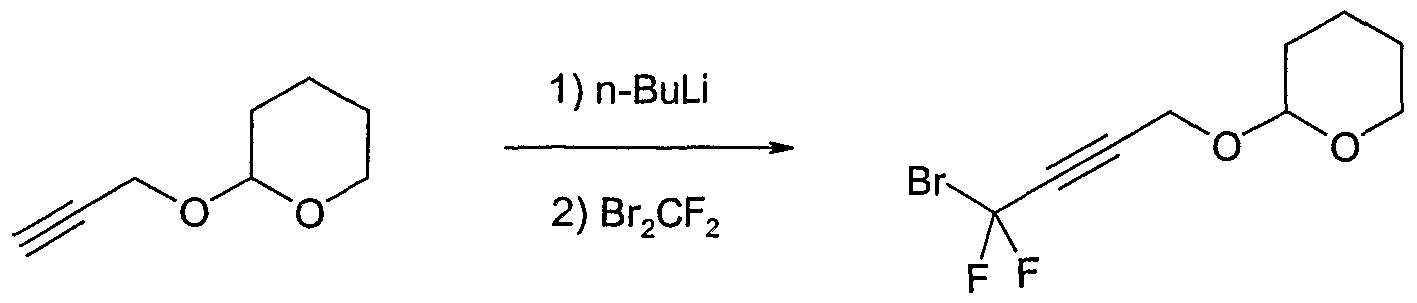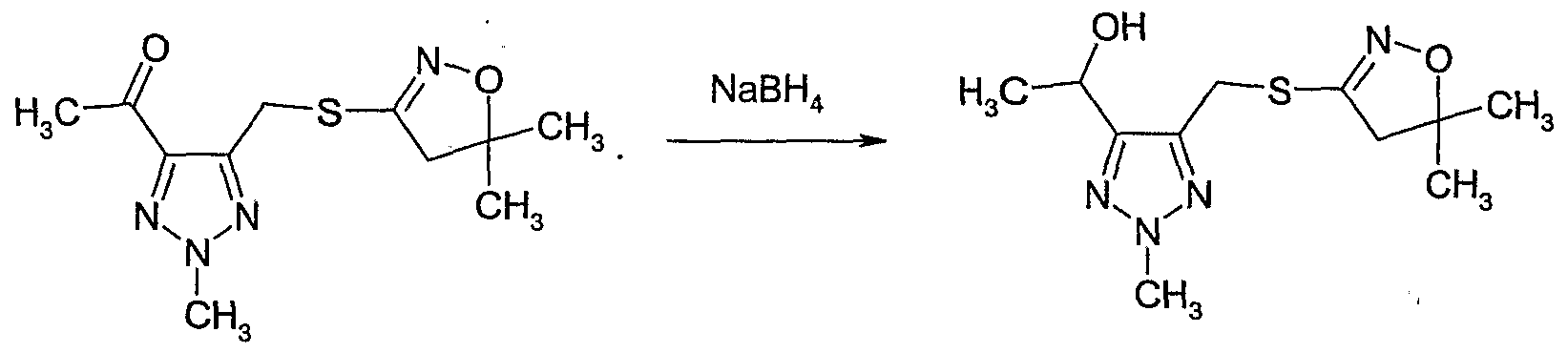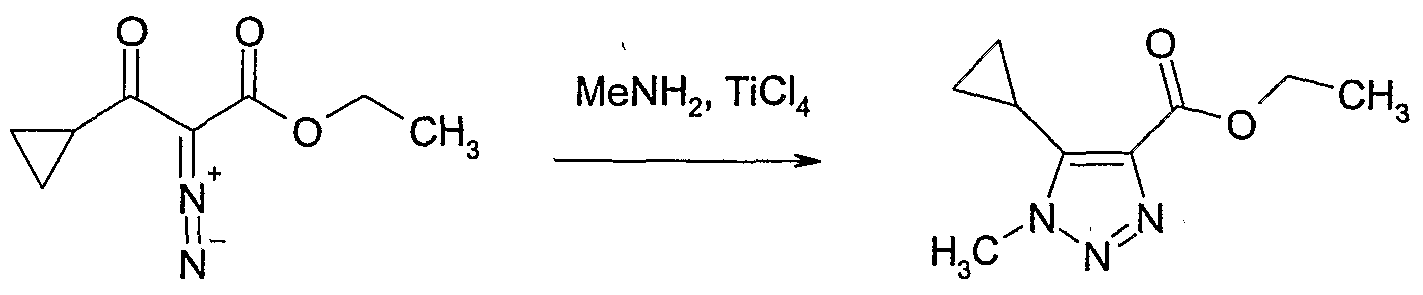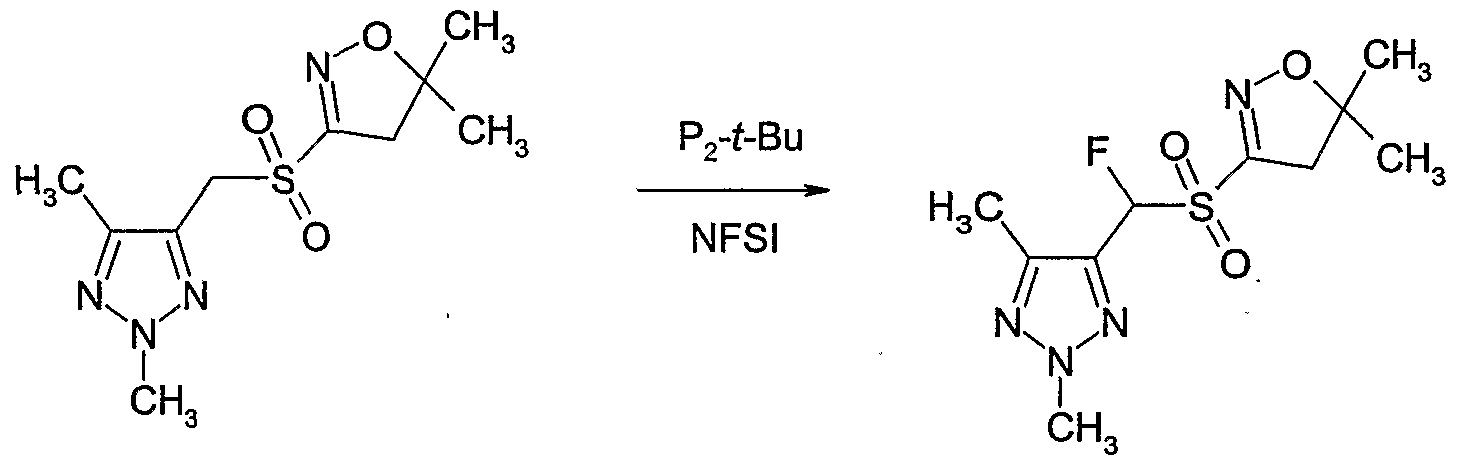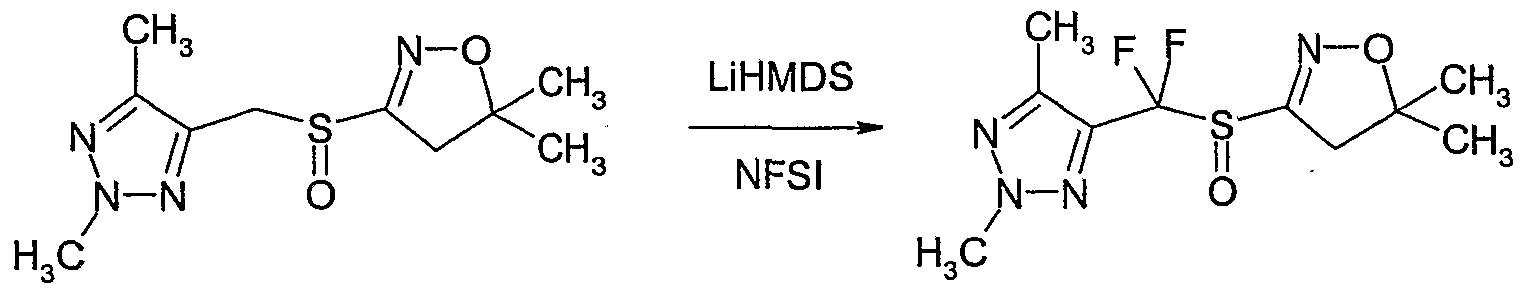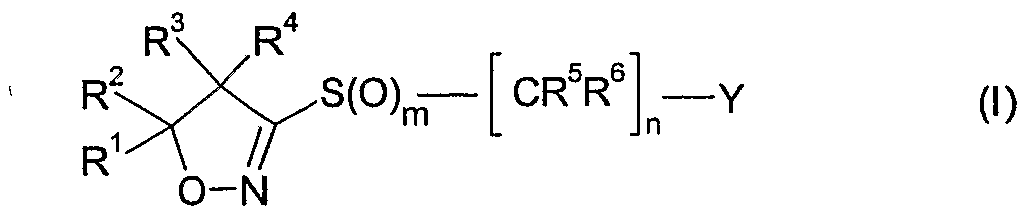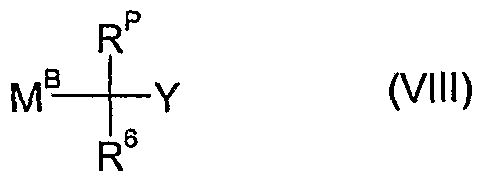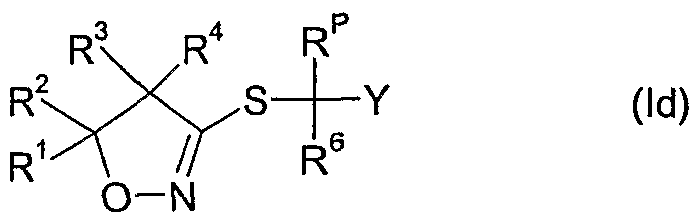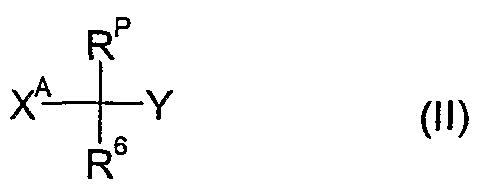WO2007096576A1 - Herbicidal isoxazoline compounds - Google Patents
Herbicidal isoxazoline compounds Download PDFInfo
- Publication number
- WO2007096576A1 WO2007096576A1 PCT/GB2007/000184 GB2007000184W WO2007096576A1 WO 2007096576 A1 WO2007096576 A1 WO 2007096576A1 GB 2007000184 W GB2007000184 W GB 2007000184W WO 2007096576 A1 WO2007096576 A1 WO 2007096576A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- formula
- compound
- methyl
- triazole
- hydrogen
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 0 *C1(*)C(S)=NOC1(*)* Chemical compound *C1(*)C(S)=NOC1(*)* 0.000 description 7
- OXJIOINGGWWRAQ-DKBJKEKUSA-N C/C(/C(/C)=N/O)=N/NC Chemical compound C/C(/C(/C)=N/O)=N/NC OXJIOINGGWWRAQ-DKBJKEKUSA-N 0.000 description 1
- YMCWJQIZJIKFHO-UHFFFAOYSA-N CC(C)(C1)ON=C1Cl Chemical compound CC(C)(C1)ON=C1Cl YMCWJQIZJIKFHO-UHFFFAOYSA-N 0.000 description 1
- RIIASHWHZUKELW-UHFFFAOYSA-N CCOC(C(C(C)=O)=[N+]=N)=O Chemical compound CCOC(C(C(C)=O)=[N+]=N)=O RIIASHWHZUKELW-UHFFFAOYSA-N 0.000 description 1
- HJQKQTDVFSMJAR-UHFFFAOYSA-N CCOC(c1c(C)[n](C)nn1)=O Chemical compound CCOC(c1c(C)[n](C)nn1)=O HJQKQTDVFSMJAR-UHFFFAOYSA-N 0.000 description 1
- CQZZRIFOMOLXGQ-UHFFFAOYSA-N CCOC(c1c(C2CC2)[n](C)nn1)=O Chemical compound CCOC(c1c(C2CC2)[n](C)nn1)=O CQZZRIFOMOLXGQ-UHFFFAOYSA-N 0.000 description 1
- HIXXPHZKNWSVCL-UHFFFAOYSA-N CCOC(c1n[n](C)nc1C1CC1)=O Chemical compound CCOC(c1n[n](C)nc1C1CC1)=O HIXXPHZKNWSVCL-UHFFFAOYSA-N 0.000 description 1
- VEDCNANCOKIATD-VIFPVBQESA-N CCO[C@@H](c1c(C2CC2)nn[n]1C)O Chemical compound CCO[C@@H](c1c(C2CC2)nn[n]1C)O VEDCNANCOKIATD-VIFPVBQESA-N 0.000 description 1
- WVIQAKMDOCNIQZ-UHFFFAOYSA-N COc1ccc(C[n]2nnc(C(F)(F)Br)c2COC2OCCCC2)cc1 Chemical compound COc1ccc(C[n]2nnc(C(F)(F)Br)c2COC2OCCCC2)cc1 WVIQAKMDOCNIQZ-UHFFFAOYSA-N 0.000 description 1
- JSCHJOSIQZYDJO-UHFFFAOYSA-N COc1ccc(C[n]2nnc(CO)c2C(F)(F)Br)cc1 Chemical compound COc1ccc(C[n]2nnc(CO)c2C(F)(F)Br)cc1 JSCHJOSIQZYDJO-UHFFFAOYSA-N 0.000 description 1
- WLQBIEJOTISZQY-UHFFFAOYSA-O COc1ccc(C[n]2nnc(COC3[OH+]CCCC3)c2C(F)(F)Br)cc1 Chemical compound COc1ccc(C[n]2nnc(COC3[OH+]CCCC3)c2C(F)(F)Br)cc1 WLQBIEJOTISZQY-UHFFFAOYSA-O 0.000 description 1
- VRTOTYWHOHTXII-UHFFFAOYSA-N Cc1n[n](C)[n+](O)c1C Chemical compound Cc1n[n](C)[n+](O)c1C VRTOTYWHOHTXII-UHFFFAOYSA-N 0.000 description 1
- DOZNHLSFAUKQMY-UHFFFAOYSA-N Cc1n[n](C)[n+](O)c1CBr Chemical compound Cc1n[n](C)[n+](O)c1CBr DOZNHLSFAUKQMY-UHFFFAOYSA-N 0.000 description 1
- LXXHPXXJHRBKCG-UHFFFAOYSA-N Cc1n[n](C)nc1CBr Chemical compound Cc1n[n](C)nc1CBr LXXHPXXJHRBKCG-UHFFFAOYSA-N 0.000 description 1
- PBZUAIHRZUBBAJ-HNQUOIGGSA-N OC(/C=N/O)=O Chemical compound OC(/C=N/O)=O PBZUAIHRZUBBAJ-HNQUOIGGSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D261/00—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings
- C07D261/02—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings
- C07D261/04—Heterocyclic compounds containing 1,2-oxazole or hydrogenated 1,2-oxazole rings not condensed with other rings having one double bond between ring members or between a ring member and a non-ring member
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01N—PRESERVATION OF BODIES OF HUMANS OR ANIMALS OR PLANTS OR PARTS THEREOF; BIOCIDES, e.g. AS DISINFECTANTS, AS PESTICIDES OR AS HERBICIDES; PEST REPELLANTS OR ATTRACTANTS; PLANT GROWTH REGULATORS
- A01N43/00—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds
- A01N43/72—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms
- A01N43/80—Biocides, pest repellants or attractants, or plant growth regulators containing heterocyclic compounds having rings with nitrogen atoms and oxygen or sulfur atoms as ring hetero atoms five-membered rings with one nitrogen atom and either one oxygen atom or one sulfur atom in positions 1,2
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D413/00—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms
- C07D413/02—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings
- C07D413/12—Heterocyclic compounds containing two or more hetero rings, at least one ring having nitrogen and oxygen atoms as the only ring hetero atoms containing two hetero rings linked by a chain containing hetero atoms as chain links
Definitions
- the present invention relates to novel, herbicidal isoxazoline compounds, to processes for their preparation, to compositions comprising those compounds, and to their use in controlling plants or in inhibiting plant growth. . •
- Isoxazoline compounds which display a herbicidal action are described, for example, in WO 01/012613, WO 02/062770, WO 03/000686, WO 04/010165, JP 2005/035924, JP 2005/213168 and WO 06/024820. The preparation of these compounds is also described in WO 04/013106 and WO 05/095352. Novel isoxazoline compounds which display herbicidal and growth-inhibiting properties have now been found.
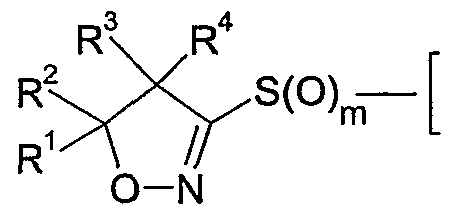
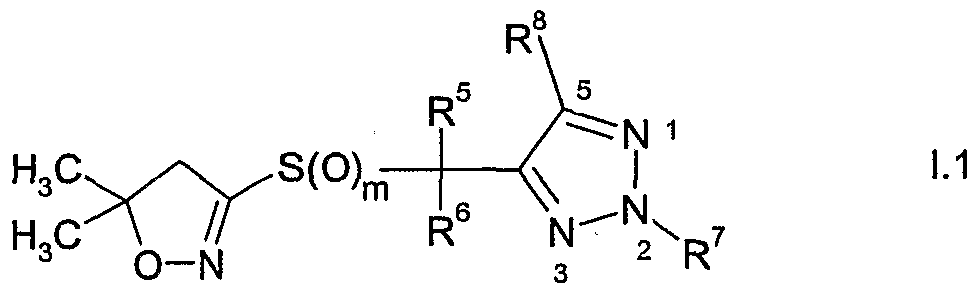
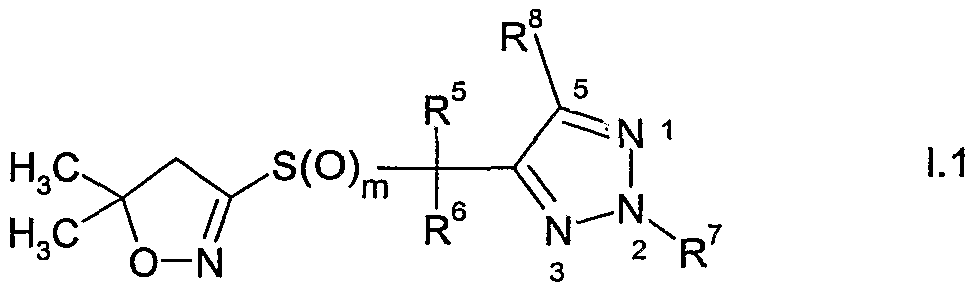
- the present invention accordingly relates to compounds of formula I
- R and R are each independently of the other hydrogen, Q-Qoalkyl, Ci-Qohaloalkyl,
- R 1 and R 2 together with the carbon atom to which they are bonded form a C 3 -C 7 ring
- R 3 and R 4 are each independently of the other hydrogen, Q-Qoalkyl, Ci-Ciohaloalkyl,
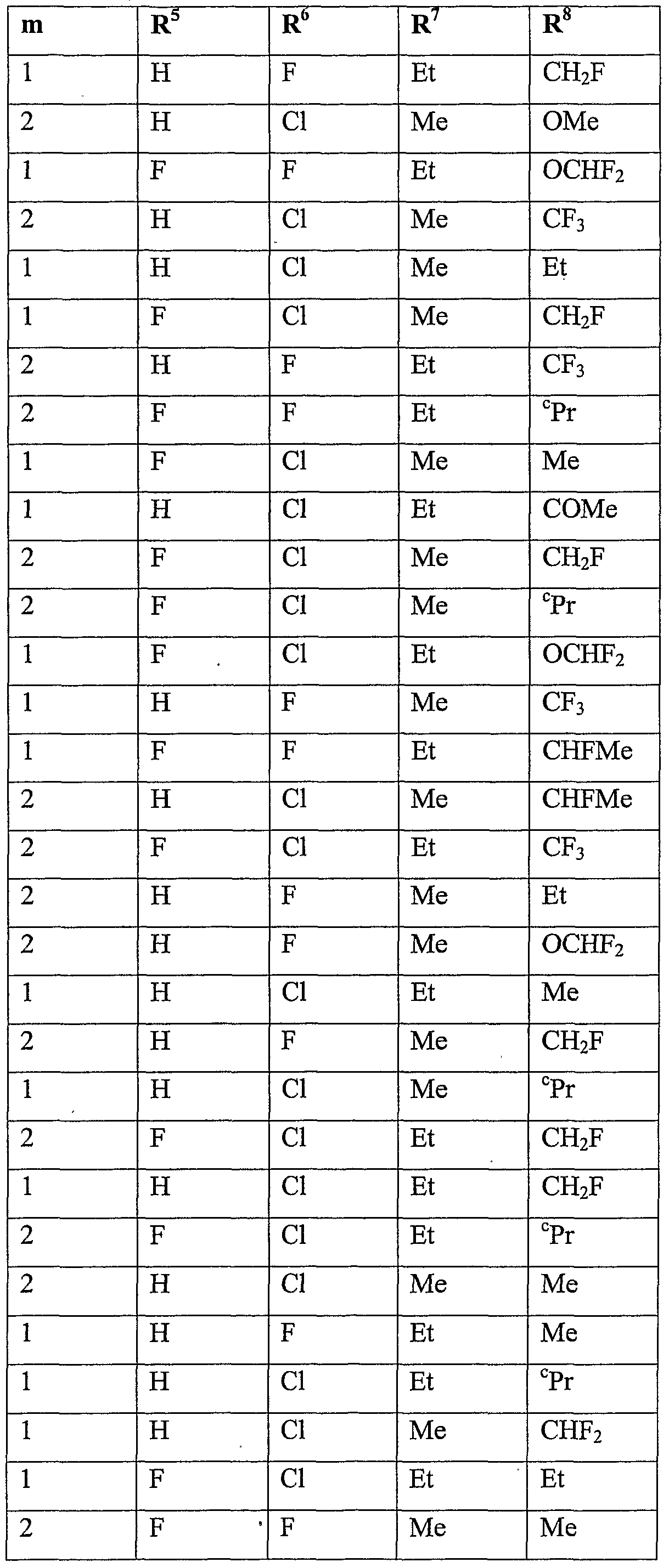
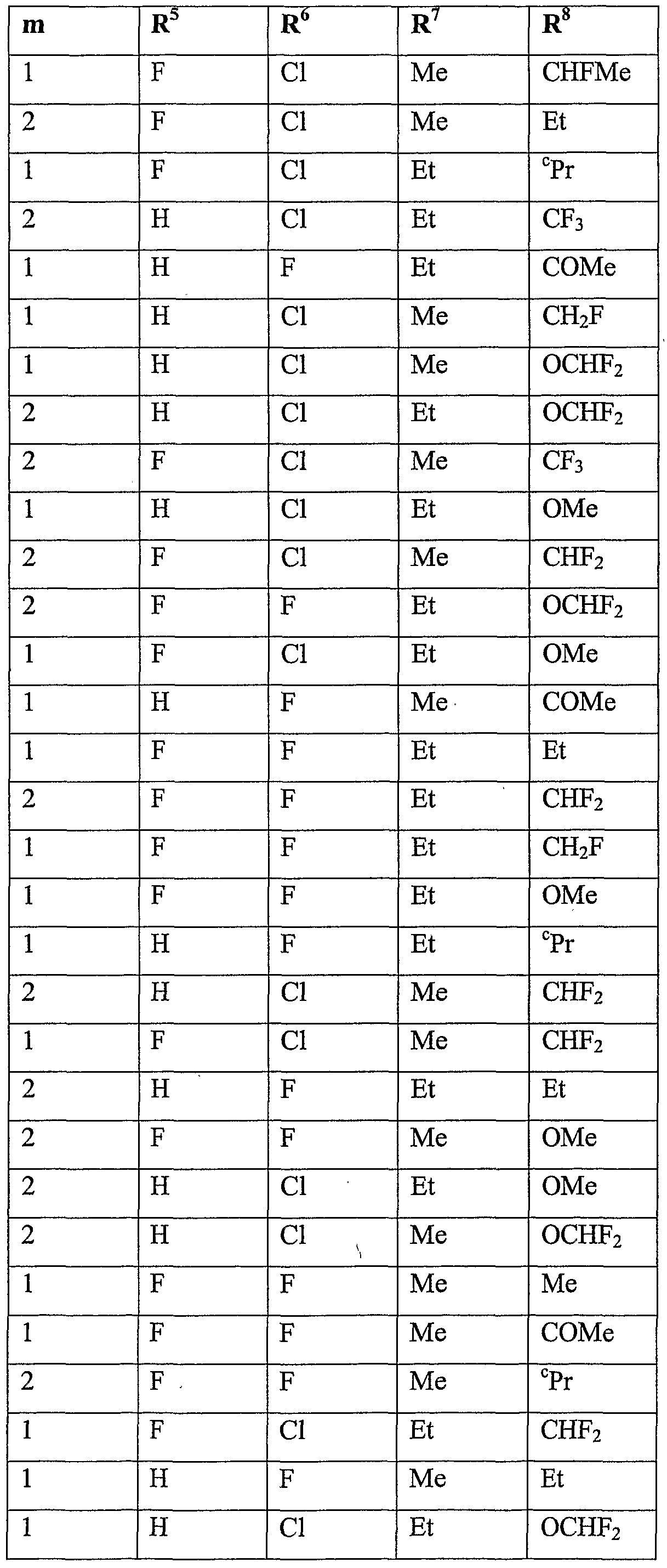
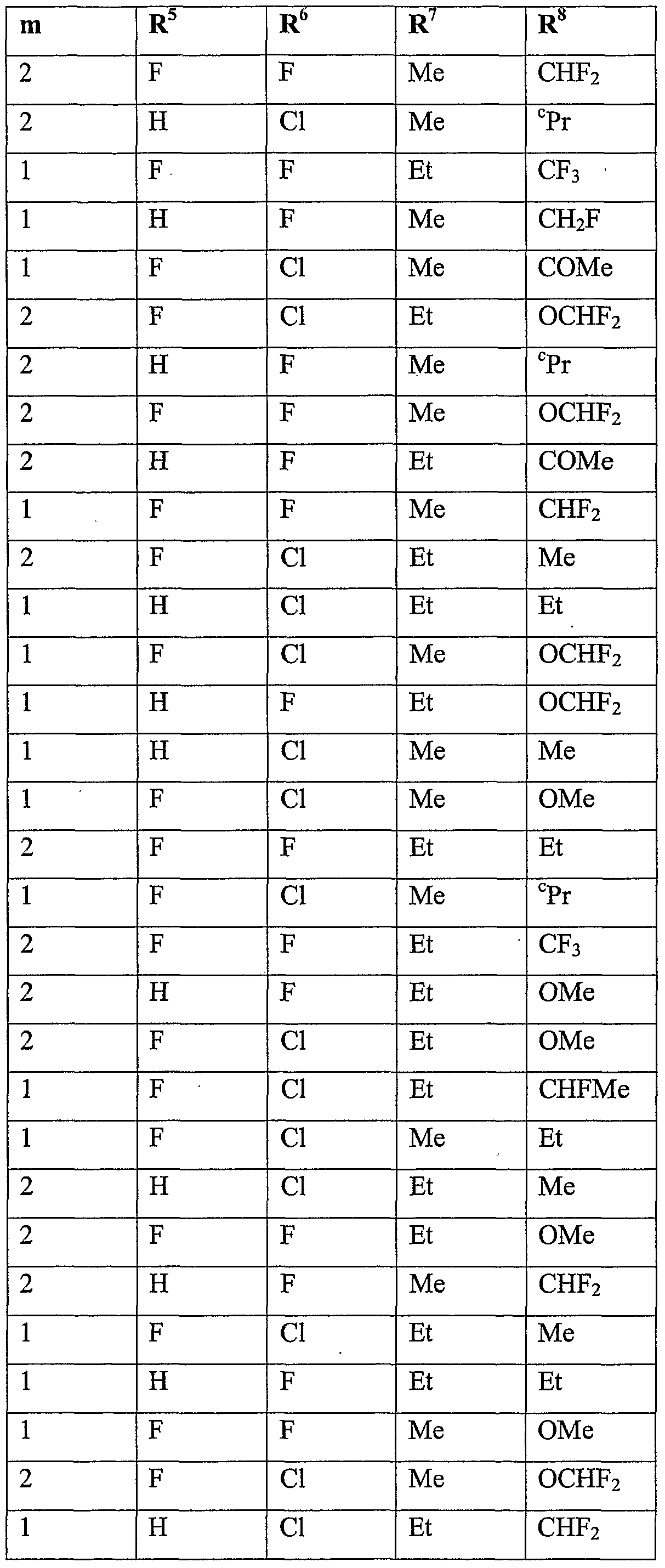
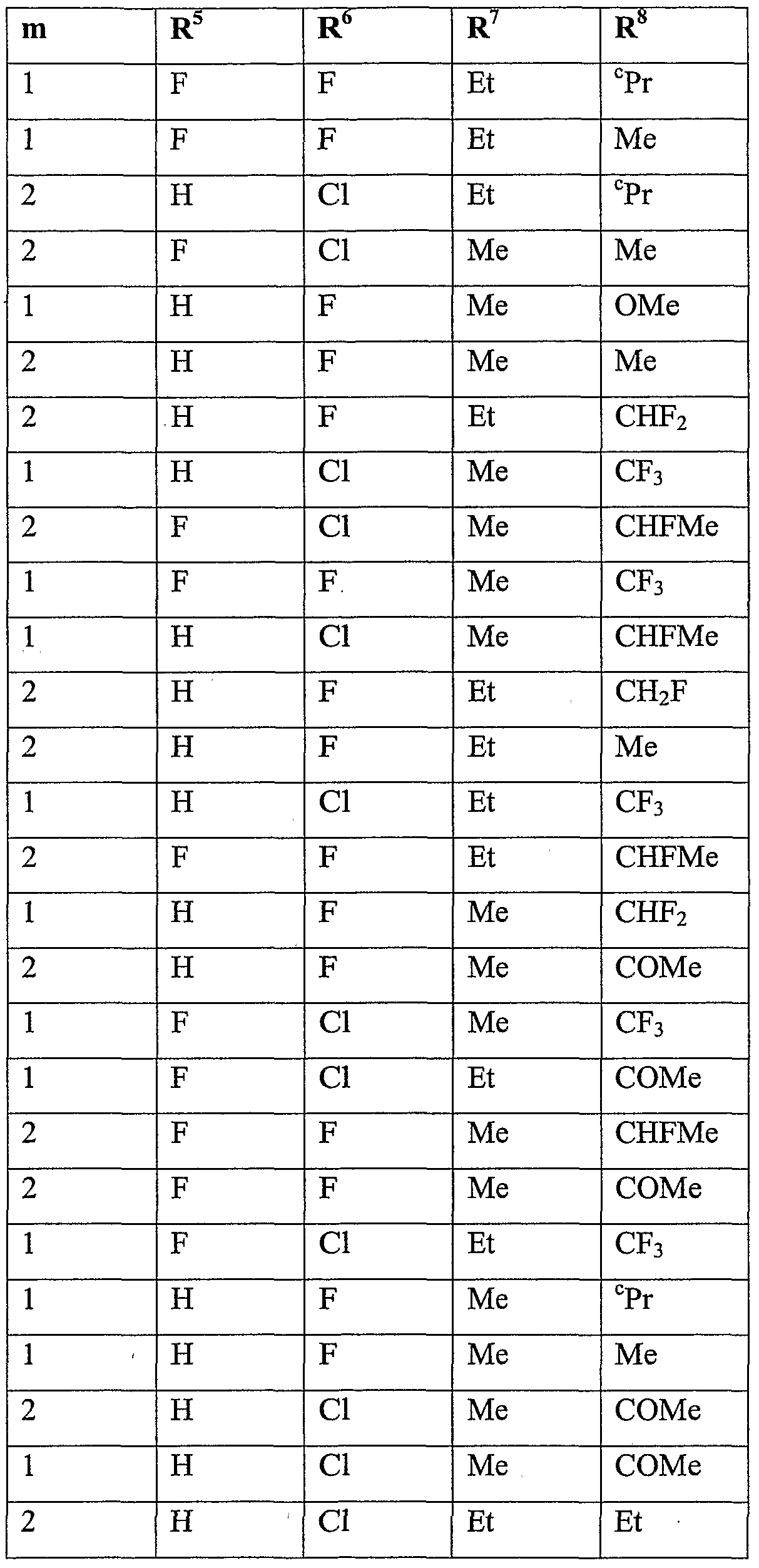
- R 5 is halogen or Ci-C 6 haloalkyl
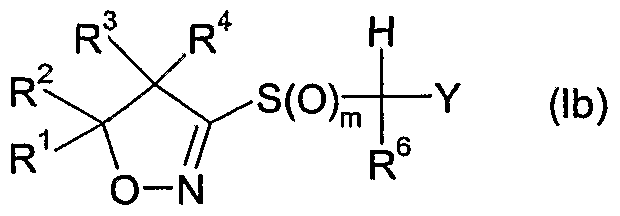
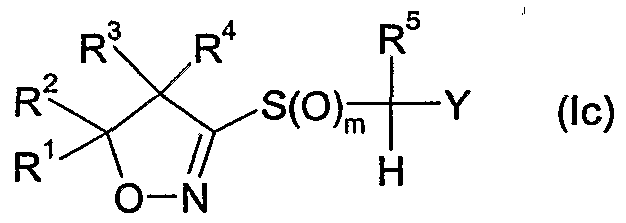
- R 6 is hydrogen, cyano, C 1 -QaIkVl, Ci-C 6 alkoxycarbonyl, halogen or Ci-C 6 haloalkyl; m is 0, 1 or 2; n is 1, 2 or 3; and
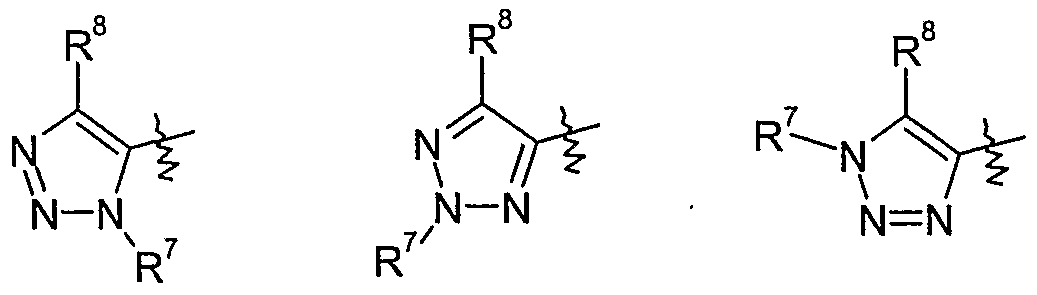
- Y is one of the following groups wherein
- R 7 is hydrogen, Ci-Cioalkyl, Ci-Cioalkylcarbonyl, formyl, d-Ciohaloalkylcarbonyl, C 1 - C 10 alkoxycarbonyl, d-Cjohaloalkyl, cyano, C 2 -C 10 alkenyl, C 2 -C 10 alkynyl, C 3 - C 10 cycloalkyl, C 3 -C 10 cycloalkyl-Ci-C 10 alkyl, d-doalkylcarbonyl-d-doalkyl, C 1 -
- R 13 and R 14 are independently of each other hydrogen, C 1 -QaIkVl, d-C 6 haloalkyl, d-C 6 cycloalkyl, d-C 6 alkylcarbonyl, d-C 6 halo- alkylcarbonyl, d-C 6 alkylsulfonyl, d-C 6 haloalkylsulfonyl, or R 13 and R 14 together form a C 3 -C 8 alkylene group which optionally contains one oxygen, sulfur, amino or
- R 1 and R 2 are independently d-Cioalkyl or d-Ciohaloalkyl, more preferably C 1 -C 6 alkyl or Ci -C 6 haloalkyl, most preferably methyl.
- R 3 and R 4 are independently hydrogen, Ci-Cioalkyl or Ci-Ciohaloalkyl, more preferably hydrogen, Ci-C 6 alkyl or Ci-C 6 haloalkyl, most preferably hydrogen.
- R 5 is halogen or trifluoromethyl, more preferably fluoro, chloro or trifluoromethyl, most preferably fluoro or chloro.
- R 6 is hydrogen, methoxycarbonyl, Ci-C 6 alkyl or halogen, more preferably hydrogen, methyl, fluoro or chloro, most preferably hydrogen or fluoro.
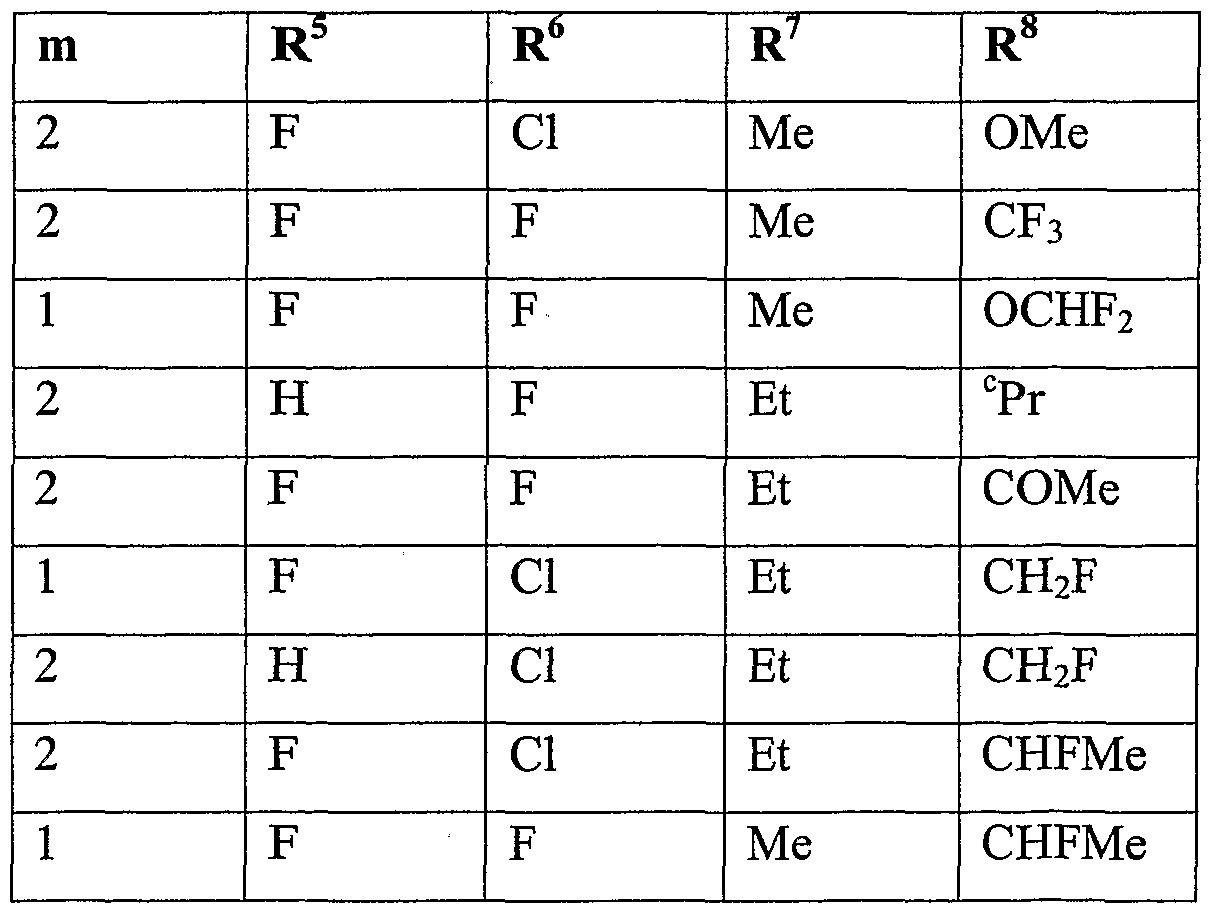
- m is 1 or 2, more preferably 2.
- n is 1.
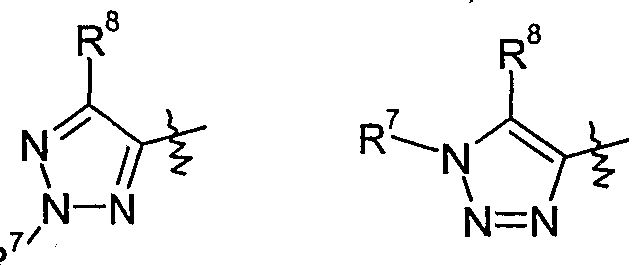
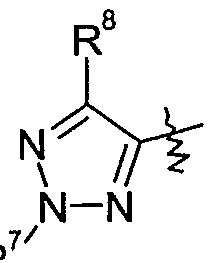
- Y is one of the following groups
- R 7 is hydrogen, C 1 -C 1O aIlCyI, Ci-Qohaloalkyl, C 2 -C 1 oalkenyl, C 2 - Cioalkynyl, C 3 -C 10 cycloalkyl, Q-docycloalkyl-Q-doalkyl, d-Qoalkoxy-Q-Cioalkyl or phenyl which is optionally substituted by one to five substituents selected from cyano, d-doalkyl, d-dohaloalkyl or halogen, more preferably hydrogen, methyl, ethyl, iso- propyl, tert-butyl, monofluoromethyl, difluoromethyl, trifluoromethyl, allyl, propargyl, cyclopropyl, cyclopentyl, cyclopropylmethyl, cyclobutyhnethyl, methoxymethyl, 2- methoxy-ethyl
- R 8 is hydrogen, C 1 -C 1O aUCyI, Ci-C 10 alkylcarbonyl, formyl, C 1 - doalkoxycarbonyl, Ci-C 10 haloalkyl, C 3 -C 10 cycloalkyl, halogen, cyano, C 1 -C 1O aIkOXy or d-dohaloalkoxy, more preferably hydrogen, methyl, ethyl, acetyl, formyl, methoxy- carbonyl, monofluoromethyl, difluoromethyl, trifluoromethyl, bromodifluoromethyl, 1- fluoro-ethyl, cyclopropyl, fluoro, chloro, bromo, cyano, methoxy, difluoromethoxy, trifluoromethoxy or 2,2,2-trifluoroethoxy, even more preferably hydrogen, methyl, ethyl, acetyl, methoxycarbonyl, monoflu
- a group of preferred compounds of formula I comprises those wherein Y is 1,2,3- triazolyl which is optionally substituted by one to two substituents independently selected from Ci-C 6 alkyl, Ci-C 6 haloalkyl, Ci-C 6 alkylcarbonyl, formyl, Q-C ⁇ alkoxycarbonyl, C 2 - C 6 alkenyl, C 3 -C 6 cycloalkyl, C 3 -C 6 cycloalkyl-Ci-C 6 -alkyl, Ci-C 6 alkoxy-C]-C 6 alkyl, halogen, cyano, Ci-C 6 alkoxy, C 1 -C 6 IIaIOaIkOXy or phenyl which is optionally substituted by one to five substituents selected from cyano, Ci-Cioalkyl, Q-Qohaloalkyl or halogen, more preferably wherein Y is 1,2,3-triazolyl which is optionally substituted by one to two
- a group of especially preferred compounds of formula I comprises those wherein Y is l,2,3-triazol-4-yl which is optionally substituted by one to two substituents independently selected from Ci-C ⁇ alkyl, Ci-C 6 haloalkyl, d-C 6 alkylcarbonyl, formyl, Q- C 6 alkoxycarbonyl, C 2 -C 6 alkenyl, C 3 -C 6 cycloalkyl, Cs-C ⁇ cycloalkyl-Ci-C ⁇ -alkyl, C 1 -
- a group of especially preferred compounds of formula I comprises those wherein Y is
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 5-monofluoromethyl-2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2,5-dimethyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 5-cycloproyl-2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein Y is 2,5-diethyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2-difluoromethyl-5-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2-allyl-5-trifluoromethyl-l ,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2-cyclopentyl-5-trifluoromethyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2-cyclobutylmethyl-5-trifluoromethyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein Y is 2-(2-methoxy-ethyl)-5-trifluoromethyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2-wo-propyl-5-trifluoromethyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2-ethyl-5-trifluoromethyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 5-ethyl-2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2-ethyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2-z ' so-propyl-5-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein Y is 2-methyl-5-trifluoromethyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 5-methoxy-2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 5-bromo-2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2-ethyl-5-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 5-difluoromethoxy-2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein Y is 5-methoxycarbonyl-2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 5-acetyl-2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 5-bromodifluoromethyl-2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 5-difluoromethyl-2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 5-(l-fluoro-ethyl)-2-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein Y is 5-methyl-2-phenyl-l ,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein Y is
- a group of especially preferred compounds of formula I comprises those wherein Y is 1 -tert-butyl-1 ,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is l,5-dimethyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 5-cyclopropyl-l-methyl-l,2,3-triazol-4-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is l,2,3-triazol-5-yl which is optionally substituted by one to two substituents independently selected from Ci-C ⁇ alkyl, Q-C ⁇ haloalkyl, d-Cealkylcarbonyl, formyl, C 1 - C 6 alkoxycarbonyl, C 2 -C 6 alkenyl, C 3 -C 6 cycloalkyl, C 3 -C 6 cycloalkyl-Ci-C 6 -alkyl, C 1 - C ⁇ alkoxy-Q-C ⁇ alkyl, halogen, cyano, C 1 -QaIkOXy, C 1 -C 6 IIaIOaIkOXy or phenyl which is optionally substituted by one to five substituents selected from cyano, Ci-Qoalkyl, C 1 - C 10 haloalkyl or halogen, more preferably wherein Y is l,2,3-triazol-5-yl which is
- a group of especially preferred compounds of formula I comprises those wherein Y is
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 4-bromo-l-methyl-l,2,3-triazol-5-yl.
- a group of especially preferred compounds of formula I comprises those wherein Y is 4-methyl-l -zs ⁇ -propyl-l ,2,3-triazol-5-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is l-methyl-4-trifluoromethyl-l,2,3-triazol-5-yl.
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 4-cyclopropyl-l-methyl-l,2,3-triazol-5-yl.
- a group of especially preferred compounds of formula I comprises those wherein
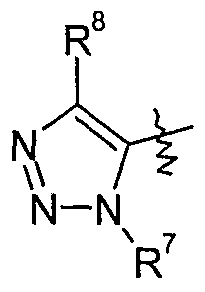
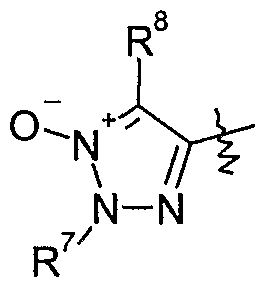
- Y is l,2,3-triazol-4-yl-l-oxide which is optionally substituted by one to two substituents independently selected from Q-C ⁇ alkyl, d-C 6 haloalkyl, Ci-Qalkylcarbonyl, formyl, Q- C 6 alkoxycarbonyl, C 2 -C 6 alkenyl, C 3 -C ⁇ cycloalkyl, CrC ⁇ cycloalkyl-Q-C ⁇ -alkyl, C 1 - Cealkoxy-C t -C ⁇ alkyl, halogen, cyano, Ci-C 6 alkoxy, d-C 6 haloalkoxy or phenyl which is optionally substituted by one to five substituents selected from cyano, Ci-C 10 alkyl, C 1 - C 10 haloalkyl or halogen, more preferably wherein Y is 1 ,2,3-triazol-4-yl-l-oxide which is optionally
- a group of especially preferred compounds of formula I comprises those wherein Y is
- a group of especially preferred compounds of formula I comprises those wherein
- Y is 2,5-dimethyl-l,2,3-triazol-4-yl-l -oxide.
- the invention relates to a compound of formula I wherein R 1 and R 2 are each independently of the other hydrogen, Ci-doalkyl, Ci-C 10 haloalkyl,
- R 1 and R 2 together with the carbon atom to which they are bonded form a C 3 -C 7 ring
- R 3 and R 4 are each independently of the other hydrogen, Ci-C 10 alkyl, Ci-C 10 haloalkyl,
- R 5 is halogen or C ⁇ -C 6 haloalkyl
- R 6 is hydrogen, cyano, d-C 6 alkyl, d-C 6 alkoxycarbonyl, halogen or Ci-C 6 haloalkyl; m is 0, 1 or 2; n is 1, 2 or 3; and
- Y is one of the following groups
- R 7 is hydrogen, d-doalkyl, d-Ci 0 alkylcarbonyl, d-dohaloalkylcarbonyl, C 1 - Cioalkoxycarbonyl, Ci-dohaloalkyl, C 2 -Ci 0 alkenyl, C 2 -Ci 0 alkynyl, C 3 -C 10 cycloalkyl, C 3 - Ciocycloalkyl-d-Cioalkyl, Ci-Cioalkylcarbonyl-d-doalkyl, Q-Cioalkylsulfonyl, C 1 - Ciohaloalkylsulfonyl or CrCioalkoxy-Ci-doalkyl;
- R is hydrogen, Q-Cioalkyl, Crdoalkylcarbonyl, d-dohaloalkylcarbonyl, C 1 - Cioalkoxycarbonyl, Ci-Ciohaloalkyl, C 2 -Ci 0 alkenyl, C 2 -Ci 0 alkynyl, C 3 -C ! ocycloalkyl, C 3 - Ciocycloalkyl-Ci-Cioalkyl, halogen, d-Ci 0 alkoxy or Ci-Ciohaloalkoxy; and to N-oxides, salts and optical isomers of compounds of formula I.
- R 7 is hydrogen, Q-Cioalkyl, Ci-Ciohaloalkyl, C2-C ⁇ oalkenyl, C 2 - Cioalkynyl, C 3 -Ci 0 cycloalkyl, C 3 -C 10 cycloalkyl-Ci-C 10 alkyl or Ci-C t oalkoxy-d- Cioalkyl, more preferably hydrogen, methyl, ethyl, allyl, propargyl, cyclopropyl, cyclopropylmethyl, methoxymethyl, methoxymethyl, difluoromethyl or trifluoromethyl, most preferably methyl.
- R 8 is hydrogen, C 1 -C 1O aUCyI, Ci-Qohaloalkyl, C 3 -C 10 cycloalkyl, halogen, Ci-Ci 0 alkoxy or d-Ciohaloalkoxy, more preferably hydrogen, methyl, ethyl, trifluoromethyl, difluoromethyl, monofluoromethyl, cyclopropyl, fluoro, chloro, trifluoromethoxy or 2,2,2-trifluoroethoxy, most preferably methyl, trifluoromethyl or monofluoromethyl.
- the compounds of the invention may contain one or more asymmetric carbon atoms, for example, in the -CR 5 R 6 - group or in the -CR 3 R 4 -group and may exist as enantiomers (or as pairs of diastereomers) or as mixtures of such.
- m 1, the compounds of the invention are sulfoxides, which can exists in two enantiomeric forms, the adjacent carbon can also exists in two enantiomeric forms and the -CR 3 R 4 - group can also exist in two enantiomeric forms.
- Compounds of general formula I can therefore exist as racemates, diastereoisomers, or single enantiomers, and the invention includes all possible isomers or isomer mixtures in all proportions. It is to be expected that for any given compound, one isomer may be more herbicidal than another.
- Alkyl groups, haloalkyl groups, hydroxyalkyl groups, alkoxy groups, haloalkoxy groups and alkylene groups can be straight or branched chain.
- Preferred alkyl groups, haloalkyl groups, hydroxyalkyl groups, alkoxy groups, haloalkoxy groups and alkylene groups each independently contain 1 to 4 carbons.
- alkyl groups are methyl, ethyl, «-and z ' so-propyl and n ⁇ , sec-, iso- and tert-butyl, hexyl, nonyl and decyl.
- Examples of haloalkyl groups are difluoromethyl and 2,2,2-trifluoroethyl.
- Examples of hydroxyalkyl groups are 1,2-dihydroxyethyl and 3-hydroxypropyl.
- Examples of alkoxy groups are methoxy, ethoxy, propoxy, butoxy, hexyloxy, nonyloxy and decyloxy.
- Examples of haloalkoxy groups are difluoromethoxy and 2,2,2-trifluoroethoxy.
- Examples of alkylene groups are methylene, ethylene, «-and zs ⁇ -propylene and n-, sec-, iso- and fert-butylene.
- Cycloalkyl groups can be in mono-, bi- or tri-cyclic form. Preferred cycloalkyl groups independently contain 3 to 8 carbons. Examples of monocyclic cycloalkyl groups are cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl and cyclooctyl. Alkenyl and alkynyl groups and haloalkenyl groups and haloalkynyl groups can be straight or branched chain. Examples of alkenyl and alkynyl groups are allyl, but-2- enyl, 3-methylbut-2-enyl, ethynyl, propargyl and but-2-ynyl.
- haloalkenyl and haloalkynyl groups are trifluoroallyl and l-chloroprop-l-yn-3-yl.
- Halogen means fluoro, chloro, bromo and iodo, preferably fluoro, chloro or bromo, more preferably fluoro or chloro.
- the invention relates likewise to the salts which the compounds of formula I are able to form with amines, alkali metal and alkaline earth metal bases and quarternary ammonium bases.
- alkali metal and alkaline earth metal hydroxides as salt formers, special mention should be made of the hydroxides of lithium, sodium, potassium, magnesium and calcium, but especially the hydroxides of sodium and potassium.
- the compounds of formula I according to the invention also include hydrates which may be formed during the salt formation.
- amines suitable for ammonium salt formation include ammonia as well as primary, secondary and tertiary Ci-Qsalkylamines, CrC 4 hydroxyalkylamines and C 2 -C 4 alkoxyalkylamines, for example methylamine, ethylamine, n-propylamine, isopropylamine, the four butylamine isomers, n-amylamine, isoamylamine, hexylamine, heptylamine, octylamine, nonylamine, decylamine, pentadecylamine, hexadecylamine, heptadecylamine, octadecylamine, methylethylamine, methylisopropylamine, methylhexylamine, methylnonylamine, methylpentadecylamine, methyloctadecylamine, ethylbutylamine
- Preferred quarternary ammonium bases suitable for salt formation correspond, for example, to the formula [N(Ra R b RcRd )]OH wherein R a , Rb, R 0 and Rd are each independently of the others CrQalkyl.
- Other suitable tetraalkylammonium bases with other anions can be obtained, for example, by anion exchange reactions.
- herbicide as used herein means a compound that controls or modifies the growth of plants.
- herbicidally effective amount means the quantity of such a compound or combination of such compounds that is capable of producing a controlling or modifying effect on the growth of plants. Controlling or modifying effects include all deviation from natural development, for example: killing, retardation, leaf burn, albinism, dwarfing and the like.
- plants refers to all physical parts of a plant, including seeds, seedlings, saplings, roots, tubers, stems, stalks, foliage, and fruits.
- locus is intended to include soil, seeds, and seedlings, as well as established vegetation.
- the compounds of the invention may be made by a variety of methods.
- R 1 , R 2 , R 3 , R 4 and Y are as defined above and m is 1 or 2, in a single step or stepwise in succession with compounds of the formula R 5 -X and/or R 6 -X, wherein R 5 is halogen and R 6 is cyano, Ci-C ⁇ alkyl, CrC ⁇ alkoxycarbonyl or halogen and X is a suitable leaving group e.g.
- halide such as bromide or iodide
- a carboxylate such as acetate
- an alkylsulfonate such as methylsulfonate
- an arylsulfonate such as p-toluenesulfonate
- a haloalkylsulfonate such as trifluoromethylsulfonate
- an imide such as succinimide, a sulfonimide, such as bis(phenylsulfonyl)imide, or an arylsulfinate, such as p-toluene- sulfinate, in the presence of abase, e.g.
- an alkyl-lithium compound such as methyl- lithium, H-butyl-lithium or tert-butyl-lithium, a lithium dialkylamide, such as lithium diisopropylamide
- a metal hydride preferably an alkali metal hydride, such as sodium hydride, or an alkali metal amide, such as sodium amide, a metal bis(tri(C 1 -C 6 alkyl)- silyl)amide, such as lithium bis(trimethylsilyl)amide, a metal alkoxide, such as potassium tert-butoxide, or a phosphazene base, such as N'-tert-butyl-N,N,N',N',N",N"-hexamethyl- phosphorimidic triamide (P ⁇ -t-Bu), l-tert-butyl-2,2,4,4,4-pentakis(dimethylamino)-2- Iambda 5 ,41ambda 5
- a hydrocarbon such as a hydrocarbon, an ether, such as tetrahydrofuran, an amide, such as N,N-dimethylformamide, or a halogenated hydrocarbon, such as dichloromethane, or mixtures thereof and optionally in the presence of a complexing agent, such as hexamethylphosphoramide or tetramethylethylenediamine in a temperature range of from -12CPC to lOCPC, preferably from -8CPC to 5CPC.
- a complexing agent such as hexamethylphosphoramide or tetramethylethylenediamine in a temperature range of from -12CPC to lOCPC, preferably from -8CPC to 5CPC.
- R 1 ; R 2 , R 3 , R 4 , R 6 and Y are as defined above and m is 1 or 2, with compounds of the formula R 5 -X, wherein R 5 is halogen and X is a suitable leaving group as defined in 1), in the presence of a base as defined in 1), optionally in the presence of a diluent as defined in 1), preferably an inert solvent, and optionally in the presence of a complexing agent as defined in 1) in a temperature range of from -12CPC to 10(PC, preferably from -8(TC to 5CPC.
- R 1 , R 2 , R 3 , R 4 , R 5 and Y are as defined above and m is 1 or 2, with compounds of the formula R 6 -X, wherein R 6 is cyano, Q-C ⁇ alkyl, Ci-C 6 alkoxycarbonyl or halogen and X is a suitable leaving group as defined in 1), in the presence of a base as defined in 1), optionally in the presence of a diluent as defined in 1) and optionally in the presence of a complexing agent as defined in 1) in a temperature range of from -120C to lOCPC, preferably from -8CPC to 5CPC.
- R 1 , R 2 , R 3 , R 4 , R 6 and Y are as defined above and R p is hydrogen, halogen or Ci-C 6 haloalkyl, and reacting those compounds with a suitable organic or inorganic oxidising agent, e.g. a peroxy acid, such as 3-chloroperoxybenzoic acid, peracetic acid or hydrogen peroxide, an alkoxyperoxide or a periodate, such as sodium periodate, optionally in the presence of a catalyst, such as ruthenium(III) chloride, optionally in the presence of a diluent, such as a halogenated hydrocarbon, e.g.
- a suitable organic or inorganic oxidising agent e.g. a peroxy acid, such as 3-chloroperoxybenzoic acid, peracetic acid or hydrogen peroxide, an alkoxyperoxide or a periodate, such as sodium periodate, optionally in the presence of a catalyst, such as ruthenium
- halogenating agent e.g. bromine or an N-halo- succinimide, such as N-chlorosuccinimide or N-bromosuccinimide, to form compounds of formula If wherein R 1 , R 2 , R 3 , R 4 and Y are as defined above, and X c is halogen, optionally in the presence of a diluent, e.g. acetic acid or a halogenated hydrocarbon, such as cabon tetrachloride or dichloromethane, in a temperature range of from -8OXU to 12CPC, preferably from -2CPC to 6CPC.
- a halogenating agent e.g. bromine or an N-halo- succinimide, such as N-chlorosuccinimide or N-bromosuccinimide
- R 5 is fluoro and M-R 5 is a suitable salt or an organometal compound in which M is e.g. Li, MgBr, Na, K, Ag or tetraalkylammonium, optionally in the presence of a Lewis acid, e.g. SnCl 4 , optionally in the presence of a complexing agent, e.g. hexa- methylphosphoramide (HMPA) or l,3-dimethyl-3,4,5,6-tetrahydro-2(lH)-pyrimidinone (DMPU), and optionally in the presence of a diluent, e.g.
- M e.g. Li, MgBr, Na, K, Ag or tetraalkylammonium
- a Lewis acid e.g. SnCl 4
- a complexing agent e.g. hexa- methylphosphoramide (HMPA) or l,3-dimethyl-3,4,5,6-tetra
- halogenating agent e.g. bromine or an N-halo- succinimide, such as N-chlorosuccinimide or N-bromosuccinimide
- a halogenating agent e.g. bromine or an N-halo- succinimide, such as N-chlorosuccinimide or N-bromosuccinimide
- X c is halogen, optionally in the presence of a diluent, e.g. acetic acid or a halogenated hydrocarbon, such as carbon tetrachloride or dichloromethane, in a temperature range of from -8CPC to 12CPC, preferably from -2CPC to 6CFC.
- R 5 and / or R 6 are fluoro and M-R 5 and / or M-R 6 are a suitable salt or an organometal compound in which M is e.g. Li, MgBr, Na, K, Ag or tetraalkylammonium, optionally in the presence of a Lewis acid, e.g. SnCl 4 , optionally in the presence of a complexing agent, e.g. hexamethylphosphoramide (HMPA) or l,3-dimethyl-3 ,4,5,6- tetrahydro-2(lH)-pyrimidinone (DMPU), and optionally in the presence of a diluent, e.g.
- HMPA hexamethylphosphoramide
- DMPU l,3-dimethyl-3 ,4,5,6- tetrahydro-2(lH)-pyrimidinone
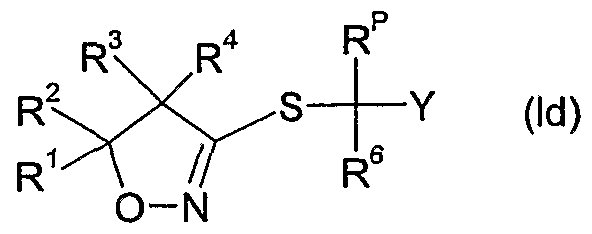
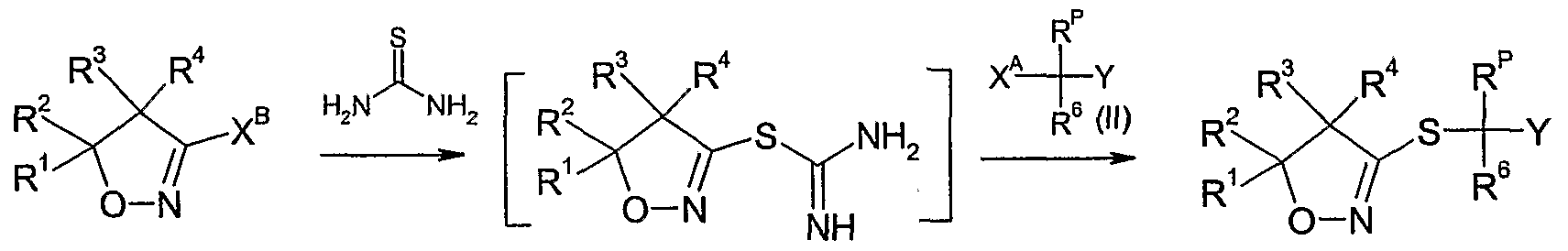
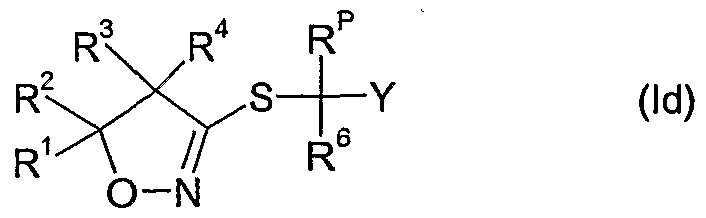
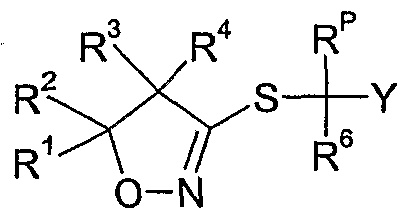
- the compounds of formula Id as defined in 4) can be prepared by reacting a compound of formula II wherein R 6 and Y are as defined above, R p is as defined in 4) and X A is a leaving group such as halide e.g. bromide or chloride, an alkylsulfonate, e.g. methylsulfonate, an arylsulfonate, e.g. p-toluenesulfonate, or a haloalkylsulfonate, e.g. trifluoromethylsulfonate, with thiourea, optionally in the presence of a diluent e.g.
- a diluent e.g.
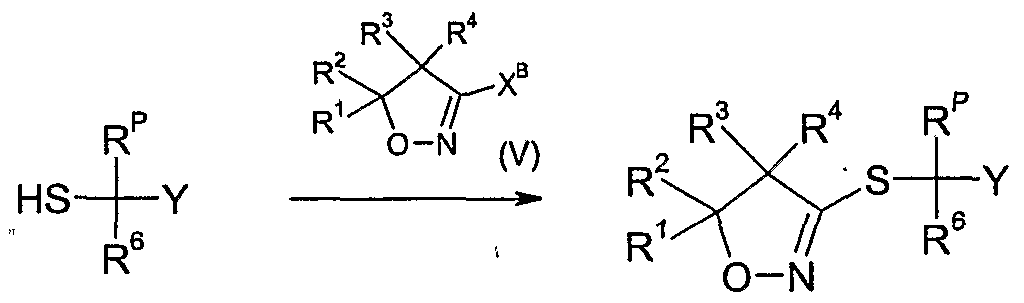
- R ! , R 2 , R 3 and R 4 are as defined above, and X B is a suitable leaving group such as halogen, e.g. chloro, an alkylsulfinyl group, an arylsulfmyl group, a haloalkylsulfinyl group, an alkylsulfonyl group, e.g. methylsulfonyl, an arylsulfonyl group, e.g. p-toluene- sulfonyl, a haloalkylsulfonyl group, e.g.
- a base such as a carbonate, e.g. potassium carbonate, sodium carbonate or potassium hydrogencarbonate, or a hydroxide, e.g. potassium hydroxide, or an alkoxide, e.g. sodium alkoxide, optionally in the presence of an alkali iodide, e.g. sodium iodide or sodium bromide, optionally in the presence of a diluent, such as an alcohol, e.g. ethanol, an ether, e.g. 1,4-dioxane or tetrahydrofuran, a polar solvent, e.g.
- a base such as a carbonate, e.g. potassium carbonate, sodium carbonate or potassium hydrogencarbonate, or a hydroxide, e.g. potassium hydroxide, or an alkoxide, e.g. sodium alkoxide, optionally in the presence of an alkali iodide, e.g. sodium iodide
- a further method of preparing intermediates of formula IV, wherein R 6 and Y are as defined above and R p is as defined in 4), is to react a compound of the formula III, wherein R 6 and Y are as defined above and R p is as defined in 4), with thiourea in the presence of an acid, for example a mineral acid, such as hydrochloric acid or hydrobromic acid, or sulfuric acid, or an organic acid, such as trifluoroacetic acid, and optionally in the presence of a diluent, such as an ether, e.g. 1,4-dioxane or tetrahydrofuran, a polar solvent, e.g.
- an acid for example a mineral acid, such as hydrochloric acid or hydrobromic acid, or sulfuric acid, or an organic acid, such as trifluoroacetic acid
- a diluent such as an ether, e.g. 1,4-dioxane or tetrahydrofur
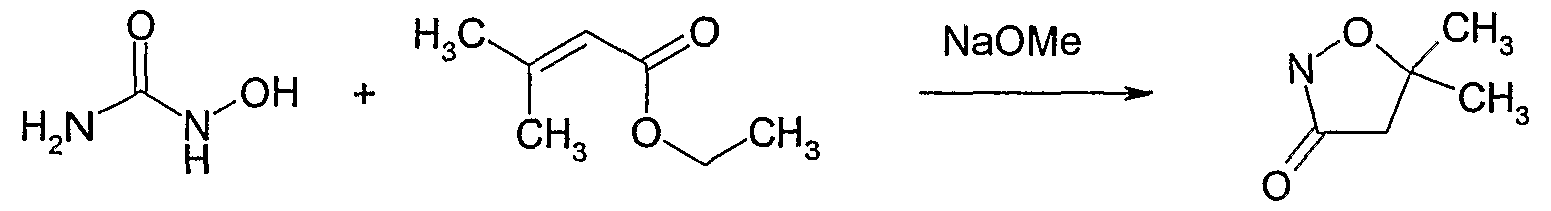
- the compounds of formula Id as defined in 4) can be prepared by reacting a compound of formula V as defined in 7) with thiourea, optionally in the . presence of a diluent e.g. an alcohol, e.g. ethanol, in a temperature range of from -3CPC to 15 CPC, preferably from OC to 8CFC, to give an isothiourea intermediate of formula VII,
- a diluent e.g. an alcohol, e.g. ethanol
- a temperature range of from -3CPC to 15 CPC preferably from OC to 8CFC
- a base such as a carbonate, e.g. potassium carbonate, sodium carbonate or potassium hydrogencarbonate, or a hydroxide, e.g. potassium hydroxide, or an alkoxide, e.g. sodium alkoxide, optionally in the presence of a diluent, such as an alcohol, e.g. ethanol, a polar solvent, e.g. water or N,N-dimethylformamide, or a mixture of solvents, in a temperature range of from CPC to 200C, preferably from CPC to lOCPC.
- a base such as a carbonate, e.g. potassium carbonate, sodium carbonate or potassium hydrogencarbonate, or a hydroxide, e.g. potassium hydroxide, or an alkoxide, e.g. sodium alkoxide, optionally in the presence of a diluent, such as an alcohol, e.g. ethanol, a polar solvent, e.g. water or N,N-
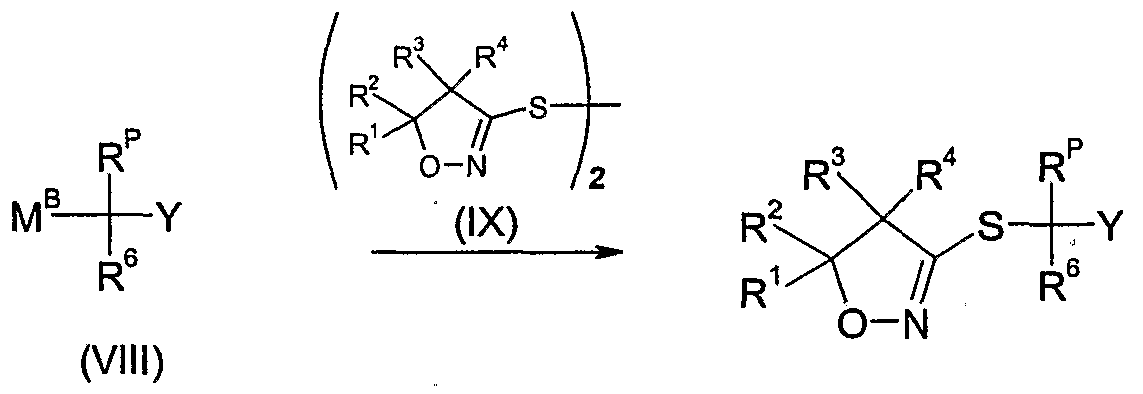
- a further method of preparing the compounds of formula Id as defined in 4) is to react an organometal reagent of the formula VIII wherein R 6 and Y are as defined above, R p is as defined in 4) and M B is a group such as MgCl, MgBr, ZnBr or Li,
- a further method of preparing the compounds of formula Id as defined in 4) is to react a compound of the formula Ha wherein R 6 and Y are as defined above, R p is as defined in 4) and X c is functional group that may be cleaved as a radical, e.g. a halogen, such as bromo or chloro,
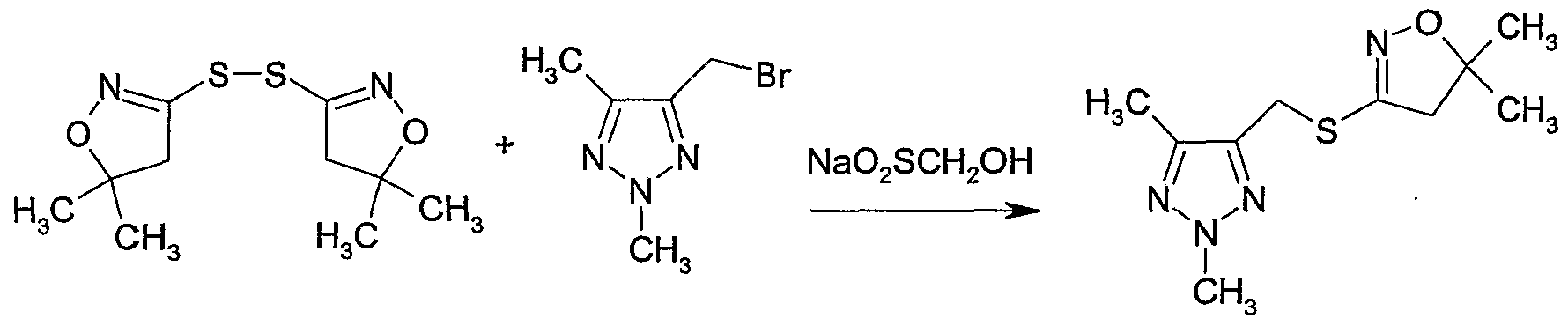
- radical initiator or a precursor thereof and with a compound of formula IX as defined in 11 optionally in the presence of a base, e.g. a phosphate or hydrogen phosphate such as disodium hydrogen phosphate, a carbonate, e.g. potassium carbonate, sodium carbonate or potassium hydrogencarbonate, optionally in the presence of a diluent, e.g. a polar solvent, such as water or N,N-dimethylformamide, or mixtures thereof, in a temperature range of from -5CPC to 18CPC, preferably from -2CPC to 5CfC, and optionally under an inert atmosphere, e.g. nitrogen.
- radical initiator or precursors can be used e.g. sodium dithionite or sodium hydroxymethylsulfinate.
- a further method of preparing the compounds of formula Id as defined in 4) is to react a compound of the formula II as defined in 7),
- a reducing agent e.g. a hydride, such as sodium borohydride, a metal, such as zinc, or a hydrosulfite, such as sodium hydrosulfite, optionally in the presence of a base, e.g. a hydroxide, such as sodium hydroxide, a phosphate or hydrogen phosphate, such as disodium hydrogen phosphate, or an amine, such as triethylamine, optionally in the presence of a diluent, e.g.
- R p is Q-C ⁇ haloalkyl, in particular perfluoroalkyl, for example trifluoromethyl
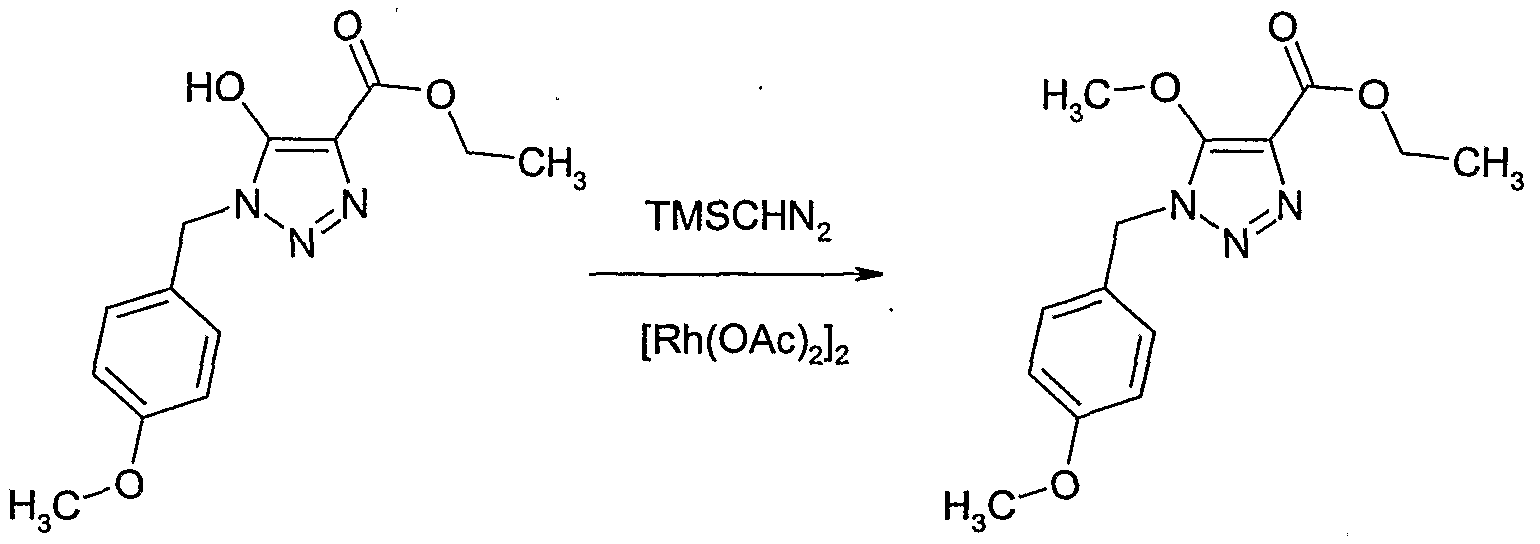
- the compounds of the formula III wherein Y is as defined above and R 6 is hydrogen, CrQalkyl or d-C ⁇ haloalkyl can be conveniently prepared by reacting carbonyl compounds of the formula XI wherein Y is as defined above and R 6 is hydrogen, Q-C ⁇ alkyl or CrC ⁇ haloalkyl, with a reagent R P -X D , wherein X D is a trialkylsilyl group, e.g. trimethylsilyl, in the presence of an initiator, such as a fluoride salt, e.g.
- caesium fluoride tetrabutylammonium fluoride or potassium fluoride, or an alkoxide salt
- an optional diluent such as an ether, e.g. tetrahydrofuran, 1,4-dioxane, in a temperature range of from CPC to lOCPC, preferably from 2CPC to 3CPC, to form the silylated intermediate of the formula XII.
- the silylated intermediate of formula XII is desilylated without isolation or purification in the presence of an acid, e.g. hydrochloric acid or hydrobromic acid, or acetic acid, and optionally in the presence of an additional diluent, such as an ether, e.g. tetrahydrofuran or 1,4-dioxane, a polar solvent, e.g. water or N,N-dimethylformarnide, or a mixture of solvents, in a temperature range of from CPC to 1 OCPC, preferably from 2CPC to 3CPC, to form the compounds of the formula III.
- an acid e.g. hydrochloric acid or hydrobromic acid, or acetic acid
- an additional diluent such as an ether, e.g. tetrahydrofuran or 1,4-dioxane, a polar solvent, e.g. water or N,N-dimethylformarnide, or a
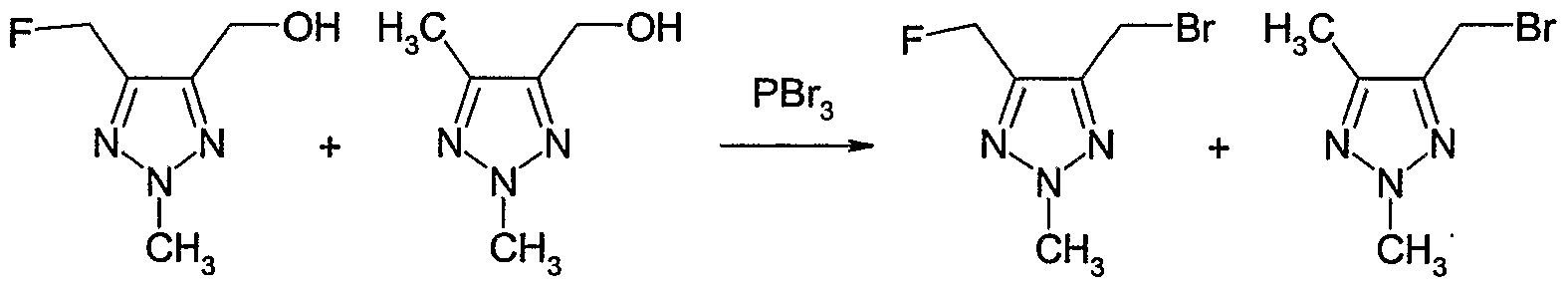
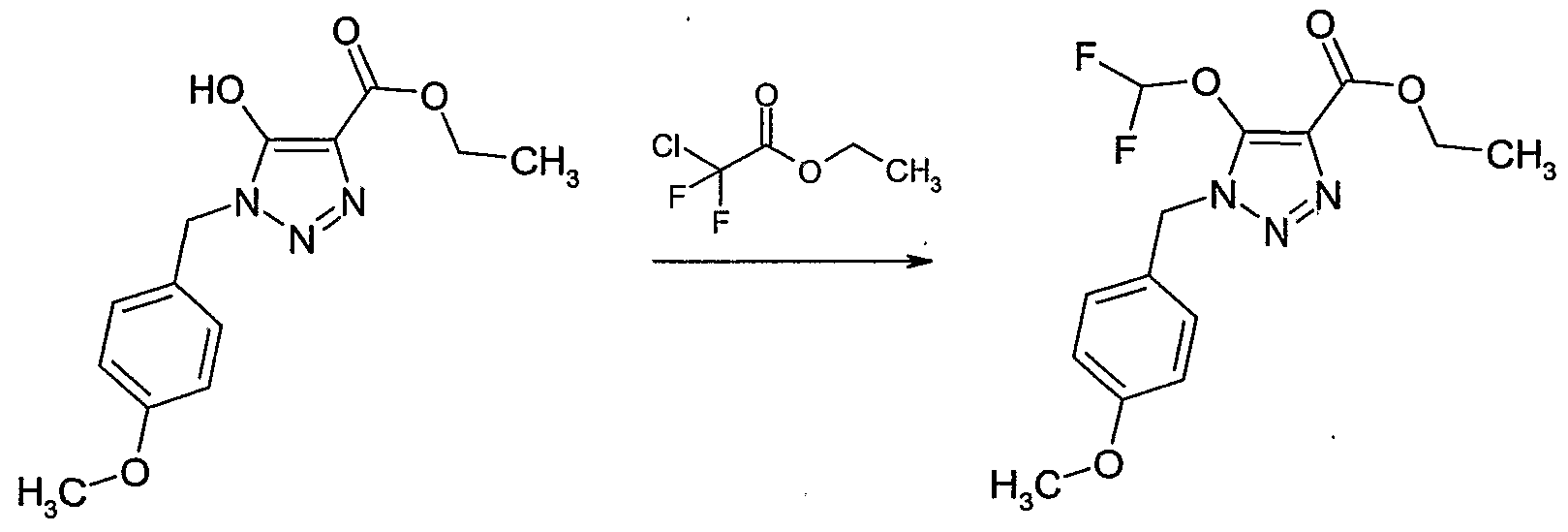
- halogenating agent such as hydrogen chloride, hydrogen bromide, phosphorous tribromide, phosphorous trichloride or thionyl chloride, or with an alkyl-, aryl- or halo- alkylsulfonyl chloride, such as methanesulfonyl chloride, p-toluenesulfonyl chloride or trifluoromethylsulfonyl chloride, or with a combination of carbon tetrabromide and triphenyl phosphine, optionally in the presence of an inert solvent, e.g.
- an inert solvent e.g.
- halogenated hydrocarbon such as dichloromethane, 1,2-dichloroethane or carbon tetrachloride
- an , ether such as diethyl ether or tetrahydrofuran
- an acid such as acetic acid
- a base e.g. an amine, such as triethyl amine, in a temperature range from -5CPC to 1 OCPC, preferably from CPC to 5CPC.
- R -X Suitable compounds of formula R -X include compounds in which R n is a succinimido group, e.g.
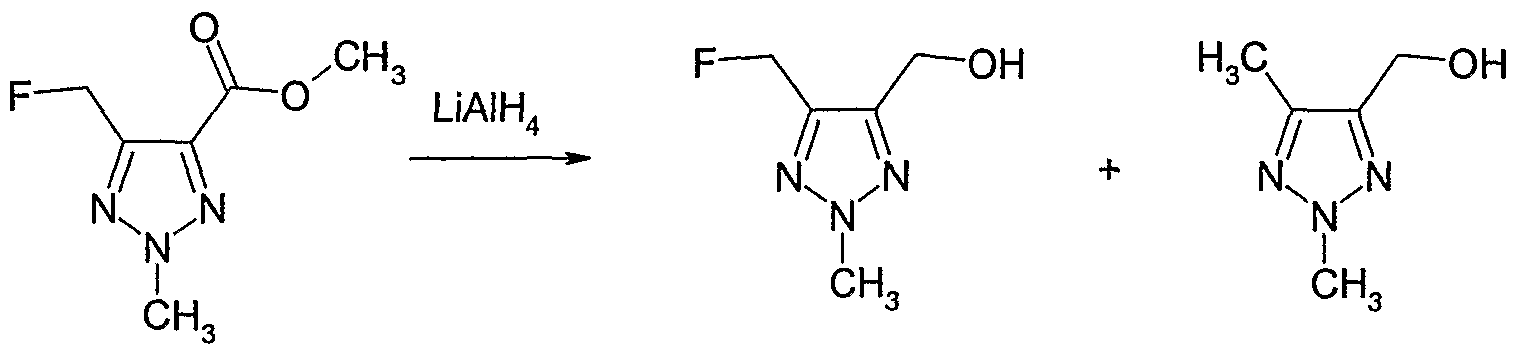
- a reducing agent e.g. a metal hydride, such as diisobutyl aluminium hydride, lithium aluminium hydride, sodium borohydride, lithium borohydride, or diborane, optionally in the presence of an inert solvent, e.g. an ether, such as diethyl ether, 1,4-dioxane or tetrahydrofuran, an alcohol, such as methanol or ethanol, or an aromatic hydrocarbon, such as toluene.
- a metal hydride such as diisobutyl aluminium hydride, lithium aluminium hydride, sodium borohydride, lithium borohydride, or diborane
- an inert solvent e.g. an ether, such as diethyl ether, 1,4-dioxane or tetrahydrofuran, an alcohol, such as methanol or ethanol, or an aromatic hydrocarbon, such as toluene.
- Y is as defined above and R 12 is hydrogen or C 1 -C 1O aIlCyI, with a reducing agent, e.g. a metal hydride, such as diisobutyl aluminium hydride, lithium aluminium hydride, sodium borohydride, lithium borohydride, or diborane, optionally in the presence of an inert solvent, e.g. an ether, such as diethyl ether, 1,4-dioxane or tetrahydrofuran, an alcohol, such as methanol or ethanol, or an aromatic hydrocarbon, such as toluene.
- a metal hydride such as diisobutyl aluminium hydride, lithium aluminium hydride, sodium borohydride, lithium borohydride, or diborane
- an inert solvent e.g. an ether, such as diethyl ether, 1,4-dioxane or tetrahydrofuran, an alcohol, such
- Such reactions are usually carried out in a temperature range from -5CPC to 10(FC, preferably from CPC to 8CPC.
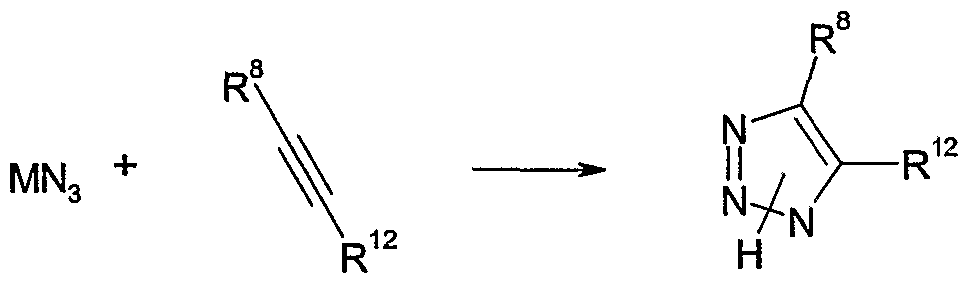
- Such processes are known in the literature and are described, for example, in Tetrahedron Asymmetry 2004 (15) 3719-3722, J. Med. Chem., 2004 (47) 2176-2179, Heterocyclic Communications 2002 (8) 385-390, J. Antibiotics, 1995 (48) 1320-1329.
- R 12 is hydrogen, Ci-Cioalkyl, Ci-Ciohaloalkyl, tetrahydro-pyran-2-yloxymethyl, C 1 - doalkoxycarbonyl, formyl, or the group Q
- a catalyst e.g. a transition metal catalyst, such as CuCl, CuI, CuBr 2 , copper powder
- a diluent such as a halogenated hydrocarbon, e.g. 1,2-dichloroethane or carbon tetrachloride, an ether, e.g. tetrahydrofuran or 1,4-dioxane, an aromatic compound, e.g.
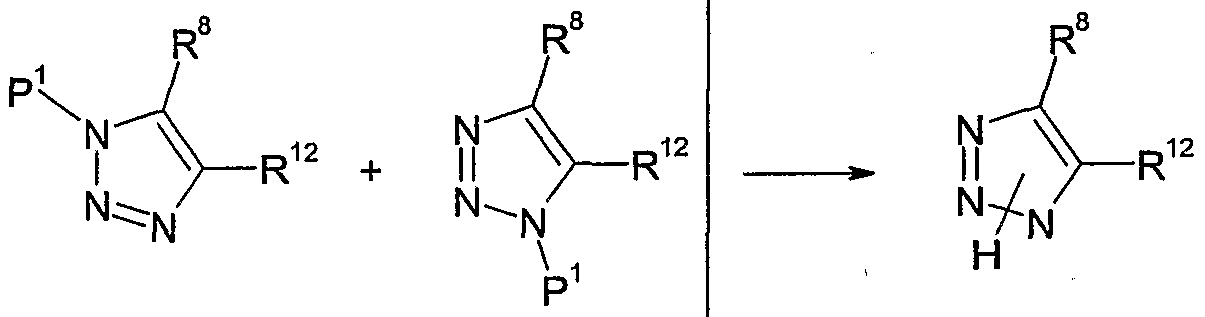
- Suitable groups P 1 include trialkylsilyl groups, such as trimethylsilyl, or optionally substituted benzyl groups, such as benzyl or 4-methoxy- benzyl.
- the protecting group is cleaved either in situ under the reaction conditions or in a separate step. Similar processes are known in the literature and are described in, e.g. Molecular Diversity, 2003 (7) 171-174; J. Heterocyclic Chemistry, 1976 (13) 589-592; WO 04/106324; J. Med. Chem., 2004 (47) 2176-2179.
- R 7 is as defined above and X E is a suitable leaving group, such as halogen, e.g. bromide, chloride or iodide, a carboxylate, such as acetate, or an alkylsulfonate, e.g. methylsulfonate, or an arylsulfonate, e.g. p-toluenesulfonate, in the presence of a base, e.g. a carbonate, such as potassium carbonate, a hydroxide, such as potassium hydroxide, a metal hydride, such as sodium hydride, an amine, such as triethylamine, optionally in the presence of a diluent, e.g.
- a base e.g. a carbonate, such as potassium carbonate, a hydroxide, such as potassium hydroxide, a metal hydride, such as sodium hydride, an amine, such as triethylamine, optionally in
- an ether such as tetrahydrofuran
- an amide such as N,N- dimethylformamide
- an alcohol such as methanol, acetonitrile or acetone or mixtures thereof
- a phase transfer catalyst such as trimethyl- ammonium bromide
- compounds of formula XXa, XXb and XXc are obtained exclusively or as mixtures in varying ratios.
- Such processes are known in the literature and are described, for example, in Bioorg. Med. Chem.
- the compounds of formula I according to the invention can be used as herbicides in unmodified form, as obtained in the synthesis, but they are generally formulated into herbicidal compositions in various ways using formulation adjuvants, such as carriers, solvents and surface-active substances.
- formulation adjuvants such as carriers, solvents and surface-active substances.
- the formulations can be in various physical forms, e.g.
- the formulations can be prepared e.g. by mixing the active ingredient with the formulation adjuvants in order to obtain compositions in the form of finely divided solids, granules, solutions, dispersions or emulsions.
- the active ingredients can also be formulated with other adjuvants, such as finely divided solids, mineral oils, oils of vegetable or animal origin, modified oils of vegetable or animal origin, organic solvents, water, surface-active substances or combinations thereof.
- the active ingredients can also be contained in very fine microcapsules consisting of a polymer. Microcapsules contain the active ingredients in a porous carrier. This enables the active ingredients to be released into the environment in controlled amounts (e.g. slow-release). Microcapsules usually have a diameter of from 0.1 to 500 microns.
- the active ingredients contain active ingredients in an amount of about from 25 to 95 % by weight of the capsule weight.
- the active ingredients can be in the form of a monolithic solid, in the form of fine particles in solid or liquid dispersion or in the form of a suitable solution.
- the encapsulating membranes comprise, for example, natural or synthetic rubbers, cellulose, styrene/butadiene copolymers, polyacrylonitrile, polyacrylate, polyesters, polyamides, polyureas, polyurethane or chemically modified polymers and starch xanthates or other polymers that are known to the person skilled in the art in this connection.
- very fine microcapsules can be formed in which the active ingredient is contained in the form of finely divided particles in a solid matrix of base substance, but the microcapsules are not themselves encapsulated.
- liquid carriers there may be used: water, toluene, xylene, petroleum ether, vegetable oils, acetone, methyl ethyl ketone, cyclohexanone, acid anhydrides, acetonitrile, acetophenone, amyl acetate, 2- butanone, butylene carbonate, chlorobenzene, cyclohexane, cyclohexanol, alkyl esters of acetic acid, diacetone alcohol, 1,2-dichloropropane, diethanolamine, p-diethylbenzene, diethylene glycol, diethylene glycol abietate, diethylene glycol butyl ether, diethylene glycol ethyl ether, diethylene glycol methyl ether, N,N-dimethylformamide, dimethyl sulfoxide, 1,4-dioxane, dipropylene glycol
- Water is generally the carrier of choice for diluting the concentrates.
- suitable solid carriers are, for example, talc, titanium dioxide, pyrophyllite clay, silica, attapulgite clay, kieselguhr, limestone, calcium carbonate, bentonite, calcium montmorillonite, cottonseed husks, wheat flour, soybean flour, pumice, wood flour, ground walnut shells, lignin and similar substances, as described, for example, in CFR 180.1001. (c) & (d).
- a large number of surface-active substances can advantageously be used in both solid and liquid formulations, especially in those formulations which can be diluted with a carrier prior to use.
- Surface-active substances may be anionic, cationic, non-ionic or polymeric and they can be used as emulsifiers, wetting agents or suspending agents or for other purposes.
- Typical surface-active substances include, for example, salts of alkyl sulfates, such as diethanolammonium lauryl sulfate; salts of alkylarylsulfonates, such as calcium dodecylbenzenesulfonate; alkylphenol/alkylene oxide addition products, such as nonylphenol ethoxylate; alcohol/alkylene oxide addition products, such as tridecylalcohol ethoxylate; soaps, such as sodium stearate; salts of alkylnaphthalenesulfonates, such as sodium dibutylnaphthalenesulfonate; dialkyl esters of sulfosuccinate salts, such as sodium di(2-ethylhexyl)sulfosuccinate; sorbitol esters, such as sorbitol oleate; quaternary amines, such as lauryltrimethylammonium chloride, polyethylene glycol esters of fatty
- compositions according to the invention can additionally include an additive comprising an oil of vegetable or animal origin, a mineral oil, alkyl esters of such oils or mixtures of such oils and oil derivatives.
- the amount of oil additive in the composition according to the invention is generally from 0.01 to 10 %, based on the spray mixture.
- the oil additive can be added to the spray tank in the desired concentration after the spray mixture has been prepared.
- Preferred oil additives comprise mineral oils or an oil of vegetable origin, for example rapeseed oil, olive oil or sunflower oil, emulsified vegetable oil, such as AMIGGE)(Rl ⁇ ie-Poulenc Canada Inc.), alkyl esters of oils of vegetable origin, for example the methyl derivatives, or an oil of animal origin, such as fish oil or beef tallow.
- a preferred additive contains, for example, as active components essentially 80 % by weight alkyl esters of fish oils and 15 % by weight methylated rapeseed oil, and also 5 % by weight of customary emulsifiers and pH modifiers.
- Especially preferred oil additives comprise alkyl esters of C 8 -C 22 fatty acids, especially the methyl derivatives OfC 12 -Cj 8 fatty acids, for example the methyl esters of lauric acid, palmitic acid and oleic acid, being of importance. Those esters are known as methyl laurate (CAS-111-82-0), methyl palmitate (CAS-112-39-0) and methyl oleate (CAS-112-62-9).
- a preferred fatty acid methyl ester derivative is Emer ⁇ D2230 and 2231 (Cognis GmbH). Those and other oil derivatives are also known from the Compendium of Herbicide Adjuvants, 5th Edition, Southern Illinois University, 2000. The application and action of the oil additives can be further improved by combination with surface-active substances, such as non-ionic, anionic or cationic surfactants. Examples of suitable anionic, non-ionic and cationic surfactants are listed on pages 7 and 8 of WO 97/34485.
- Preferred surface-active substances are anionic surfactants of the dodecylbenzylsulfonate type, especially the calcium salts thereof, and also non-ionic surfactants of the fatty alcohol ethoxylate type.
- ethoxylated C 12 -C 22 fatty alcohols having a degree of ethoxylation of from 5 to 40.
- examples of commercially available surfactants are the Genapol types (Clariant AG).
- silicone surfactants especially polyalkyl-oxide-modified heptamethyltriloxanes which are commercially available e.g. as Silwet L-77®, and also perfluorinated surfactants.
- the concentration of the surface-active substances in relation to the total additive is generally from 1 to 30 % by weight.
- oil additives consisting of mixtures of oil or mineral oils or derivatives thereof with surfactants are Edenor ME SU®, Turbocharge® (Syngenta AG, CH) or ActipronC (BP Oil UK Limited, GB).
- an organic solvent may contribute to an additional enhancement of action.
- Suitable solvents are, for example, Solvesso® (ESSO) or Aromatic Solvent® (Exxon Corporation). The concentration of such solvents can be from 10 to 80 % by weight of the total weight.
- Oil additives that are present in admixture with solvents are described, for example, in US-A- 4,834,908.
- a commercially available oil additive disclosed therein is known by the name MERGE® (BASF Corporation).
- a further oil additive that is preferred according to the invention is SCORE® (Syngenta Crop Protection Canada).
- alkylpyrrolidones e.g. Agrimax®
- formulations of alkylpyrrolidones e.g. Agrimax®
- synthetic lattices e.g. polyacrylamide, polyvinyl compounds or poly-1-p-menthene (e.g. Bond®, Courier® or Emerald®)
- propionic acid for example Eurogkem Pen-e-trate®
- the herbicidal compositions generally comprise from 0.1 to 99 % by weight, especially from 0.1 to 95 % by weight, compounds of formula I and from 1 to 99.9 % by weight of a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance.
- a formulation adjuvant which preferably includes from 0 to 25 % by weight of a surface-active substance.
- commercial products will preferably be formulated as concentrates, the end user will normally employ dilute formulations.
- the rates of application of compounds of formula I may vary within wide limits and depend on the nature of the soil, the method of application (pre- or post-emergence; seed dressing; application to the seed furrow; no tillage application etc.), the crop plant, the grass or weed to be controlled, the prevailing climatic conditions, and other factors governed by the method of application, the time of application and the target crop.
- the compounds of formula I according to the invention are generally applied at a rate of from 10 to 2000 g/ha, especially from 50 to 1000 g/ha.
- Emulsifiable concentrates active ingredient: 1 to 95 %, preferably 60 to 90 % surface-active agent: 1 to 30 %, preferably 5 to 20 % liquid carrier: 1 to 80 %, preferably 1 to 35 %
- Dusts active ingredient: 0.1 to 10 %, preferably 0.1 to 5 % solid carrier: 99.9 to 90 %, preferably 99.9 to 99 %
- Suspension concentrates active ingredient: 5 to 75 %, preferably 10 to 50 % water: 94 to 24 %, preferably 88 to 30 % surface-active, agent: 1 to 40 %, preferably 2 to 30 %
- Wettable powders active ingredient: 0.5 to 90 %, preferably 1 to 80 % surface-active agent: 0.5 to 20 %, preferably 1 to 15 % solid carrier: 5 to 95 %, preferably 15 to 90 %
- Granules active ingredient: 0.1 to 30 %, preferably 0.1 to 15 % solid carrier: 99.5 to 70 %, preferably
- Emulsifiable concentrates a) b) c) d) active ingredient 5% 10% 25% 50% calcium dodecylbenzenesulfonate 6% 8% 6% 8% castor oil polyglycol ether 4% 4% 4% 4%
- Emulsions of any desired concentration can be obtained from such concentrates by dilution with water.
- Wettable powders a) b) c) ⁇ active ingredient 5% 25% 50% 80% sodium lignosulfonate 4% - 3% - sodium lauryl sulfate 2% 3% - 4% sodium diisobutylnaphthalene- sulfonate - 6% 5% 6% octylphenol polyglycol ether — 1% 2% _ (7-8 mol of ethylene oxide) highly dispersed silicic acid 1% 3% 5% 10% kaolin 88% 62% 35% -
- the active ingredient is mixed thoroughly with the adjuvants and the mixture is thoroughly ground in a suitable mill, affording wettable powders which can be diluted with water to give suspensions of any desired concentration.
- the active ingredient is dissolved in methylene chloride and applied to the carrier by spraying, and the solvent is then evaporated off in vacuo.
- the finely ground active ingredient is uniformly applied, in a mixer, to the carrier moistened with polyethylene glycol.
- Non-dusty coated granules are obtained in this manner.
- Ready-to-use dusts are obtained by mixing the active ingredient with the carriers and grinding the mixture in a suitable mill.
- Suspension concentrates a) b) c) d) active ingredient 3 % 10 % 25 % 50 % ethylene glycol 5 % 5 % 5 % 5 % nonylphenol polyglycol ether - 1 % 2 % - (15 mol of ethylene oxide) sodium lignosulfonate 3 % 3 % 4 % 5 % carboxymethylcellulose 1 % 1 % 1 % 1 %
- the finely ground active ingredient is intimately mixed with the adjuvants, giving a suspension concentrate from which suspensions of any desired concentration can be obtained by dilution with water.
- the invention also relates to a method of controlling plants which comprises applying to the plants or to the locus thereof a herbicidally effective amount of a compound of formula I, or of a composition comprising such a compound.
- the invention also relates to a method of inhibiting plant growth which comprises applying to the plants or to the locus thereof a herbicidally effective amount of a compound of formula I, or of a composition comprising such a compound.
- the invention also relates to a method of selectively controlling grasses and weeds in crops of useful plants which comprises applying to the useful plants or locus thereof or to the area of cultivation a herbicidally effective amount of a compound of formula I, or of a composition comprising such a compound.
- Crops of useful plants in which the composition according to the invention can be used include cereals, for example barley and wheat, cotton,, oilseed rape, maize, rice, soy beans, sugar beet and sugar cane, especially cereals and maize.
- Crops can also include trees, such as palm trees, coconut trees or other nuts, and vines such as grapes.
- the grasses and weeds to be controlled may be both monocotyledonous species, for example Agrostis, Alopecurus, Avena, Bromus, Cyperus, Digitaria, Echinochloa, Lolium, Monochoria, Rottboellia, Sagittaria, Scirpus, Setaria, Sida and Sorghum, and dicotyledonous species, for example Abutilon, Amaranthus, Chenopodium, Chrysanthemum, Galium, Ipomoea, Nasturtium, Sinapis, Solanum, Stellaria, Veronica, Viola and Xanthium.
- monocotyledonous species for example Agrostis, Alopecurus, Avena, Bromus, Cyperus, Digitaria, Echinochloa, Lolium, Monochoria, Rottboellia, Sagittaria, Scirpus, Setaria, Sida and Sorghum
- dicotyledonous species for example Abutilon,
- Crops are to be understood as also including those crops which have been rendered tolerant to herbicides or classes of herbicides (e.g. ALS-, GS-, EPSPS-, PPO- and HPPD-inhibitors) by conventional methods of breeding or by genetic engineering.
- herbicides or classes of herbicides e.g. ALS-, GS-, EPSPS-, PPO- and HPPD-inhibitors
- An example of a crop that has been rendered tolerant to imidazolinones, e.g. imazamox, by conventional methods of breeding is Clearf ⁇ eld&ummer rape (canola).
- crops that have been rendered tolerant to herbicides by genetic engineering methods include e.g. glyphosate- and glufosinate-resistant maize varieties commercially available under the trade names RoundupRead ⁇ and LibertyLinl®
- Crops are also to be understood as being those which have been rendered resistant to harmful insects by genetic engineering methods, for example Bt maize (resistant to European corn borer), Bt cotton (resistant to cotton boll weevil) and also Bt potatoes (resistant to Colorado beetle).
- Bt maize are the Bt 176 maize hybrids of NKg) (Syngenta Seeds).
- the Bt toxin is a protein that is formed naturally by Bacillus thuringiensis soil bacteria. Examples of toxins, or transgenic plants able to synthesise such toxins, are described in EP-A-451 878, EP-A-374 753, WO 93/07278, WO 95/34656, WO 03/052073 and EP-A-427 529.
- transgenic plants comprising one or more genes that code for an insecticidal resistance and express one or more toxins are KnockOut® (maize), Yield Gard® (maize), NuCOTIN33B® (cotton), Bollgard® (cotton), NewLeaf® (potatoes), NatureGard® and Protexcta®.
- Plant crops or seed material thereof can be both resistant to herbicides and, at the same time, resistant to insect feeding ("stacked" transgenic events).
- seed can have the ability to express an insecticidal Cry3 protein while at the same time being tolerant to glyphosate.
- Crops are also to be understood as being those which are obtained by conventional methods of breeding or genetic engineering and contain so-called output traits (e.g. improved storage stability, higher nutritional value and improved flavour).
- output traits e.g. improved storage stability, higher nutritional value and improved flavour.
- Areas under cultivation include land on which the crop plants are already growing and land intended for cultivation with those crop plants.
- the compounds of formula I according to the invention can also be used in combination with other herbicides.
- the following mixtures of the compound of formula I are important:
- a compound of formula I with a PPO inhibitor (e.g. compound of formula I + fomesafen (401), compound of formula I + flumioxazin (376), compound of formula I + sulfentrazone (749), compound of formula I + [3-[2-chloro-4-fluoro-5-(l- methyl-6-trifluoromethyl-2,4-dioxo-l,2,3,4-tetrahydropyrimidin-3-yl)phenoxy]-2- pyridyloxy] acetic acid ethyl ester) (CAS RN 353292-31-6).
- a PPO inhibitor e.g. compound of formula I + fomesafen (401), compound of formula I + flumioxazin (376), compound of formula I + sulfentrazone (749), compound of formula I + [3-[2-chloro-4-fluoro-5-(l- methyl-6-trifluoromethyl-2,4-dioxo-l,2,
- the mixing partners of the compound of formula I may also be in the form of esters or salts, as mentioned e.g. in The Pesticide Manual, 13 th Edition (BCPC), 2003.
- the reference to glufosinate-ammonium also applies to glufosinate
- the reference to cloransulam-methyl also applies to cloransulam
- the reference to pyrithiobac-sodium also applies to pyrithiobac, etc.
- the mixing ratio of the compound of formula I to the mixing partner is preferably from l: 100 to 1000:1.
- the mixtures can advantageously be used in the above-mentioned formulations (in which case "active ingredient” relates to the respective mixture of compound of formula I with the mixing partner).
- the compounds of formula I according to the invention can also be used in combination with other herbicides: compound of formula I + acetochlor (5), compound of formula I + acifluorfen-sodium (7), compound of formula I + aclonifen (8), compound of formula I + acrolein (10), compound of formula I + alachlor (14), compound of formula I + alloxydim (18), compound of formula I + allyl alcohol (CAS RN 107-18-6), compound of formula I + amidosulfixron (22), compound of formula I + aminopyralid (CAS RN 150114-71-9), compound of formula I + amitrole (aminotriazole) (25), compound of formula I + ammonium sulfamate (26), compound of formula I + anilofos (31), compound of formula I + asulam (36), compound of formula I + atraton (CAS RN 1610-17-9), compound of formula I + aviglycine (39), compound of formula I + azafenidin
- the mixing partners of the compound of formula I may also be in the form of esters or salts, as mentioned e.g. in The Pesticide Manual, 13 th Edition (BCPC), 2003.
- the reference to acifluorfen-sodium also applies to acifluorfen, and the reference to bensulfuron-methyl also applies to bensulfuron, etc.
- the mixing ratio of the compound of formula I to the mixing partner is preferably
- mixtures can advantageously be used in the above-mentioned formulations (in which case "active ingredient” relates to the respective mixture of compound of formula I with the mixing partner).
- active ingredient relates to the respective mixture of compound of formula I with the mixing partner.
- the compounds of formula I according to the invention can also be used in combination with one or more safeners.
- mixtures of a compound of formula I according to the invention with one or more further herbicides can also be used in combination with one or more safeners.
- the safeners can be AD 67 (MON 4660) (11), benoxacor (63), cloquintocet-mexyl (163), cyometrinil and the corresponding (Z) isomer, cyprosulfamide (CAS RN 221667-31-8), dichlormid (231), fenchlorazole-ethyl (331), fenclorim (332), flurazole (386), fluxofenim (399), furilazole (413) and the corresponding R isomer, isoxadifen-ethyl (478), mefenpyr-diethyl (506), oxabetrinil (598), naphthalic anhydride (CAS RN 81-84-5) and N-isopropyl-4-(2-methoxy- benzoylsulfamoyl)-benzamide (CAS RN 221668-34-4). Particularly preferred are mixtures of a compound of formula I with benoxacor (i.e.
- the safeners of the compound of formula I may also be in the form of esters or salts, as mentioned e.g. in The Pesticide Manual, 13 th Edition (BCPC), 2003.
- the reference to cloquintocet-mexyl also applies to a lithium, sodium, potassium, calcium, magnesium, aluminium, iron, ammonium, quaternary ammonium, sulfonium or phos- phonium salt thereof as disclosed in WO 02/34048, and the reference to fenchlorazole- ethyl also applies to fenchlorazole, etc.
- the mixing ratio of compound of formula I to safener is from 100: 1 to 1 :10, especially from 20: 1 to 1 : 1.
- mixtures can advantageously be used in the above-mentioned formulations (in which case "active ingredient” relates to the respective mixture of compound of formula I with the safener).
- active ingredient relates to the respective mixture of compound of formula I with the safener.
- Preferred mixtures of a compound of formula I with further herbicides and safeners include:
- Mixtures of a compound of formula I with a triazine and a safener Mixtures of a compound of formula I with glyphosate and a safener. Mixtures of a compound of formula I with glufosinate and a safener. Mixtures of a compound of formula I with isoxaflutole and a safener.
- Mixtures of a compound of formula I with isoxaflutole and glyphosate and a safener Mixtures of a compound of formula I with isoxaflutole and glyphosate and a safener. Mixtures of a compound of formula I with isoxaflutole and glufosinate and a safener.
- Mixtures of a compound of formula I with mesotrione and a safener Mixtures of a compound of formula I with mesotrione and a triazine and a safener. Mixtures of a compound of formula I with mesotrione and glyphosate and a safener.
- Mixtures of a compound of formula I with sulcotrione and a safener Mixtures of a compound of formula I with sulcotrione and a triazine and a safener.
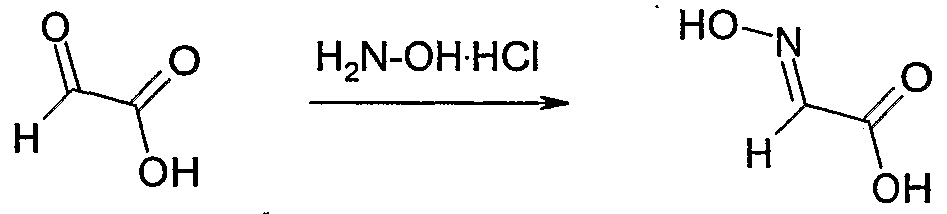
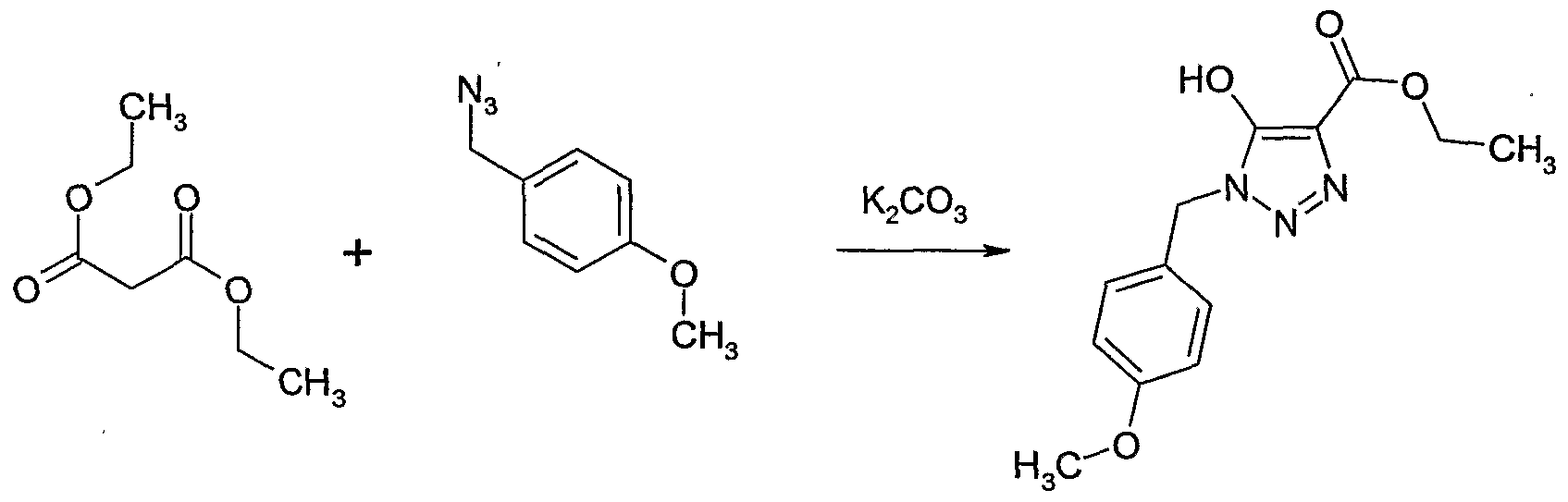
- Aqueous glyoxylic acid (50% by weight) (1 1, 9 mol) and hydroxyl amine hydrochloride (627 g, 9 mol) were mixed and concentrated.
- Acetonitrile (1 1) was added, the solution cooled to 5O and filtered.
- the mother liquor was concentrated to 50% of the volume, stored at 5C overnight and filtered again. This process was repeated twice.
- the solids were combined and dried to give hydroxyimino-acetic acid as white crystals (546 g, 68% yield).
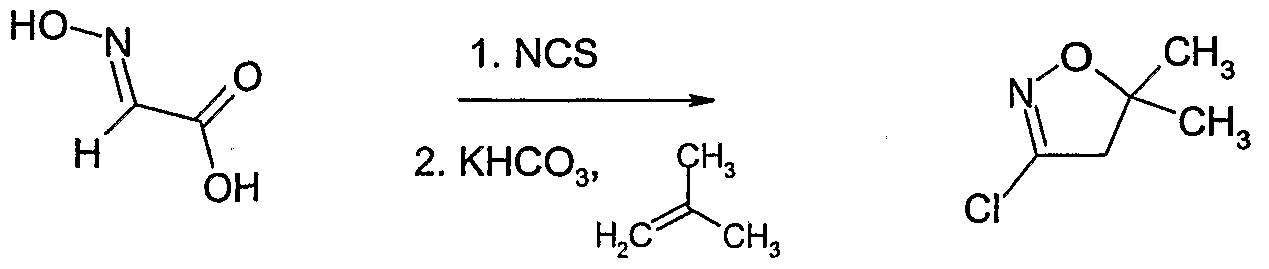
- Example II Hydroxyimino-acetic acid (Example II) (107 g, 1.2 mol) was dissolved in 1,2- dimethoxy-ethane (1.4 1) and heated to 7CPC. N-chlorosuccinimide (NCS) (320.4 g, 2.4 mol) was added in portions within 1 hour at 7CPC. The reaction mixture was stirred at 7CPC for 1 hour. The reaction mixture was cooled to SC and potassium hydrogencarbonate (535 g, 4.45 mol) and water (54 g) were added. 2-Methyl-propene (134.6 g, 2.4 mol) was introduced into the suspension for 20 minutes at 5C. The reaction mixture was allowed to warm to room temperature and was stirred at room temperature for 18 hours.
- NCS N-chlorosuccinimide
- 2-Methyl-propene 134.6 g, 2.4 mol
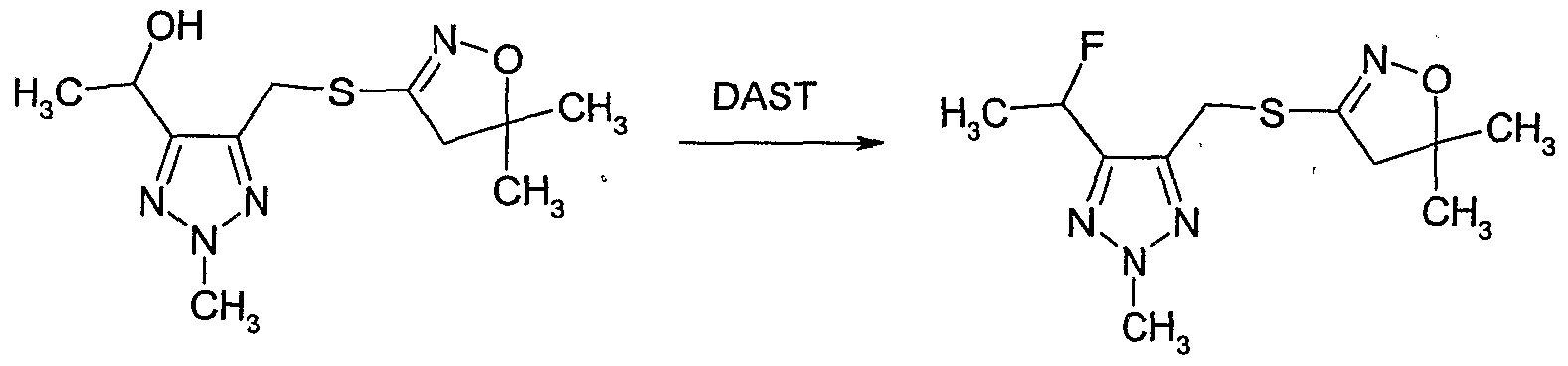
- Example 16 Preparation of (5-fluoromethyl-2-methyl-2H-
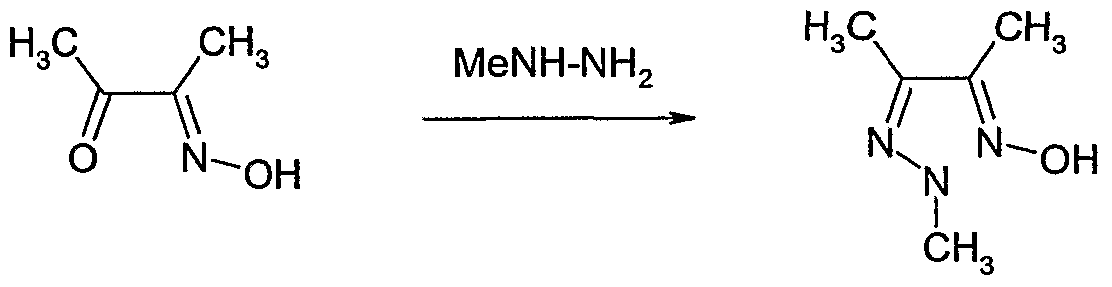
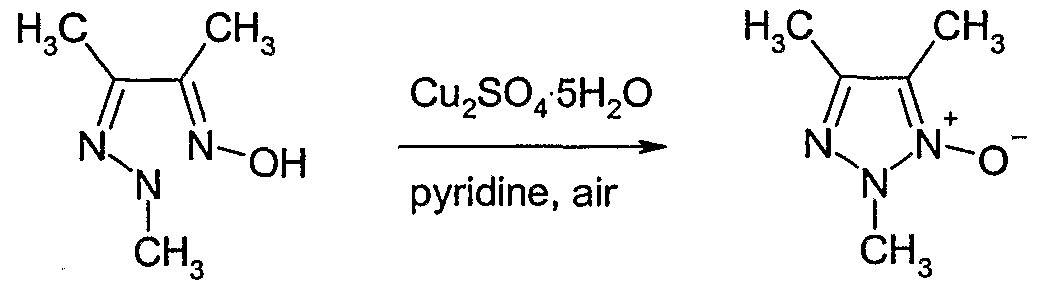
- Butane-2,3-dione monooxime (2.6 g, 25.8 mmol) was dissolved in ethanol (100 ml) then methylhydrazine (1.13 g, 24.5 mmol) was added at room temperature. The reaction mixture was stirred at 8CPC for 2 hours. Methylhydrazine (0.6 g, 12 mmol) was added and the reaction mixture stirred at 8CPC for 1.5 hours. Methylhydrazine (0.6 g, 12 mmol) was added and the reaction mixture stirred at 8CPC for another 1.5 hours, then at room temperature for 48 hours.
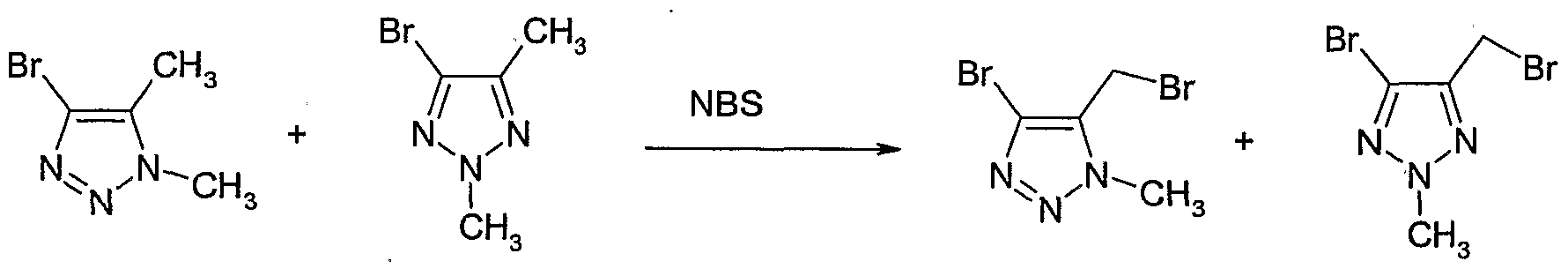
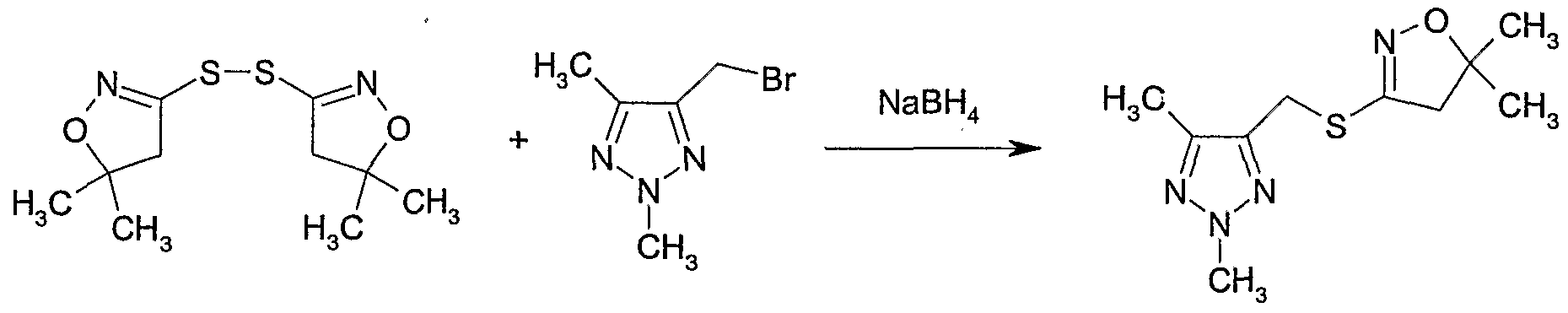
- N-Bromosuccinimide (NBS) (186 mg, 1.0 mmol) and 2,2'-azobisisobutyronitrile (AIBN) (14 mg, 0.087 mmol) were added to a solution of 2,4,5-trimethyl-2H-[ 1,2,3]- triazole 1-oxide (110 mg, 0.87 mmol) (Example 19) in carbon tetrachloride (7 ml).
- the reaction mixture was heated to 70 0 C for 1 hour then cooled to room temperature and filtered. The solvent was removed to give ' 5-bromomethyl-2,4-dimethyl-2uZ-[l, 2,3]- triazole 1-oxide as a brown gum.
- 1 H-NMR 400 MHz, CDCl 3 ): 2.31 (s, 3H, Me), 3.95 (s, 3H, Me), 4.42 (s, 2H, CH 2 ) ppm.
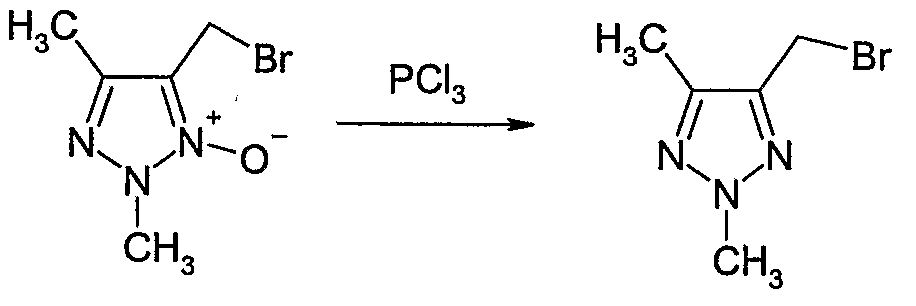
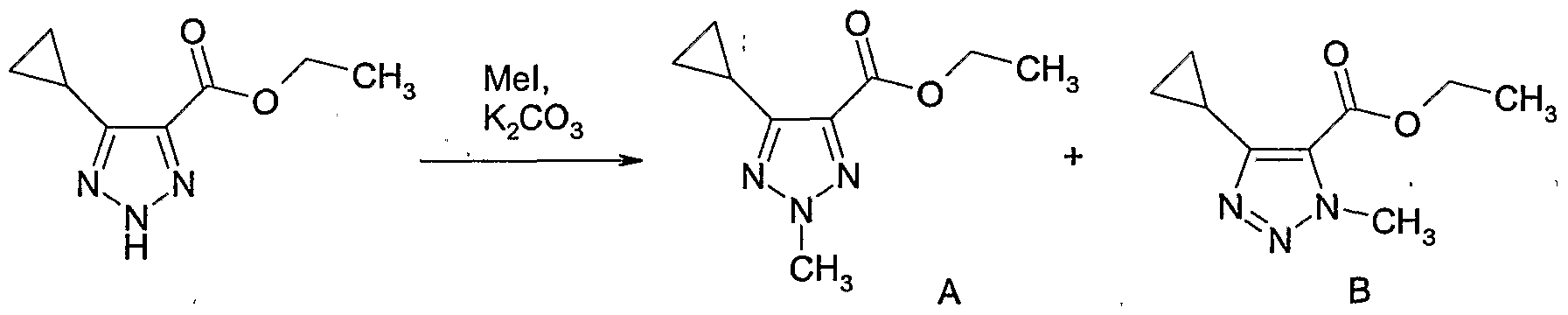
- Example Il 1 Alternative preparation of 4-bromomethyl-2,5-dimethyl-2H-
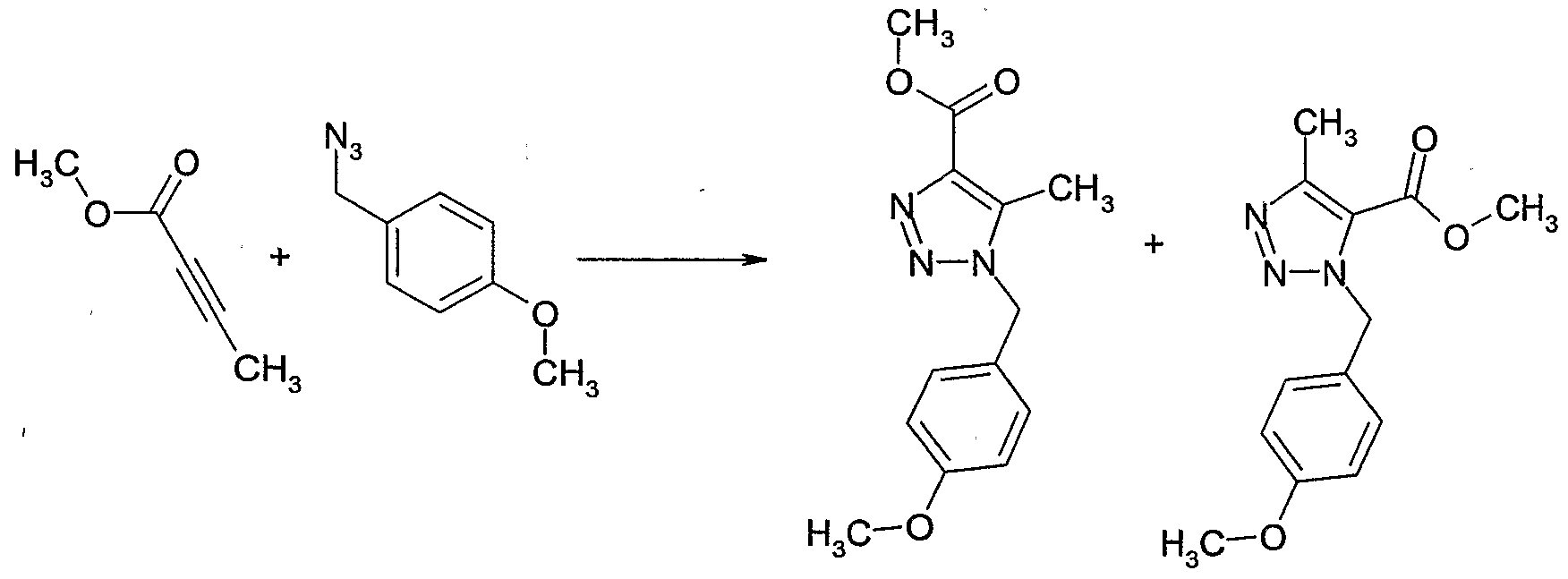
- Example 112 Preparation of l-(4-methoxy-benzylV5-methyl-lH-
- Example 114 Preparation of 2-ethyl-5-methyl-2H-[l,2,3]triazole-4-carboxylic acid methyl ester, 3-ethyl-5-methyl-3H-[l,2,31triazole-4-carboxylic acid methyl ester and 1- ethyl-5-methyl-lH-ri,2,31triazole-4-carboxylic acid methyl ester
- Example 115 Preparation of 2-difluoromethyl-5-methyl-2H-[l,2,3]triazole-4-carboxylic acid ethyl ester and 3-difluoromethyl-5-methyl-3H-[ ' l,2,3 '
- Example 116 Preparation of l-(4-methoxy-benzylV5-trifluoromemyl-lH-[l,2J]triazole- 4-carboxylic acid ethyl ester and 3-(4-methoxy-benzyl)-5-trifluoromethyl-3H " -[l,2,3]- triazole-4-carboxylic acid ethyl ester
- Example 118 Preparation of 2-methyl-5-trifluoromethyl-2H-[l,2,31triazole-4-carboxylic acid ethyl ester, 3-methyl-5-trifluoromethyl-3H-[l,2,3]triazole-4-carboxylic acid ethyl ester and l-methyl-5-trifluoromethyl-lH-ri,231triazole-4-carboxylic acid ethyl ester
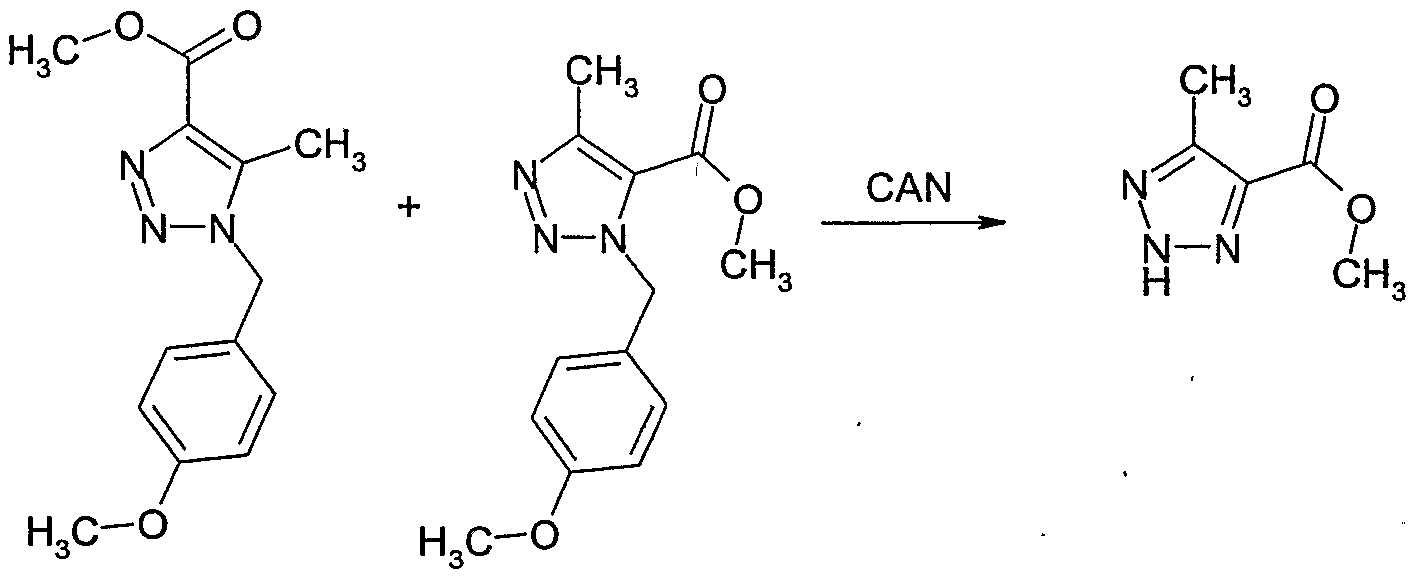
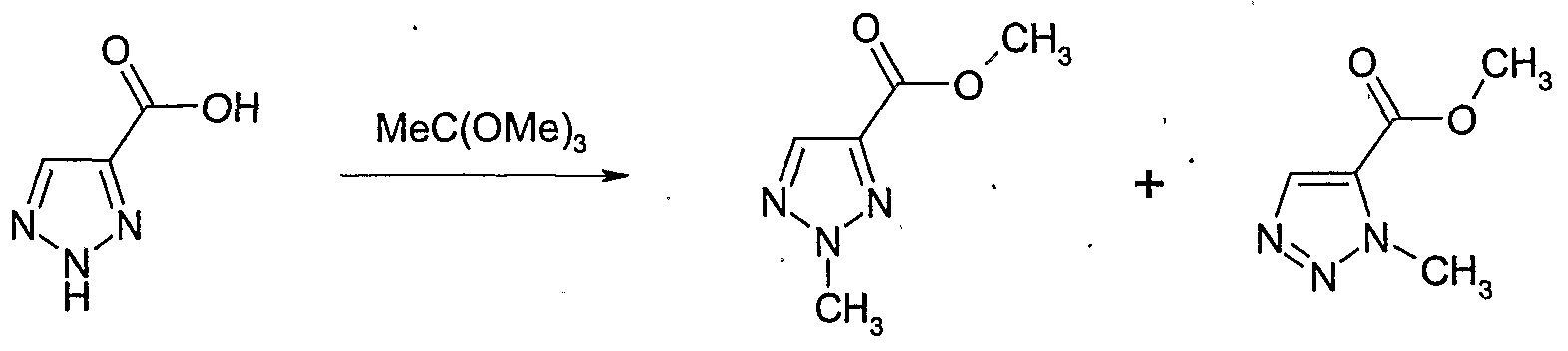
- Example 139 Example 119: Preparation of 2-methyl-2H-[l,2,31triazole-4-carboxylic acid methyl ester and 3-methyl-3H-
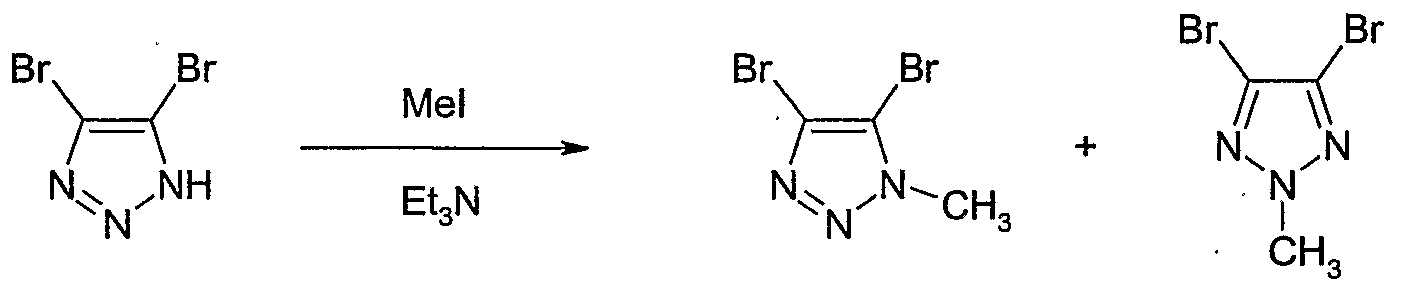
- Example 123 Preparation of 4-bromo-5-bromomethyl-l-methyl-lH-[ ' l,2,3 '
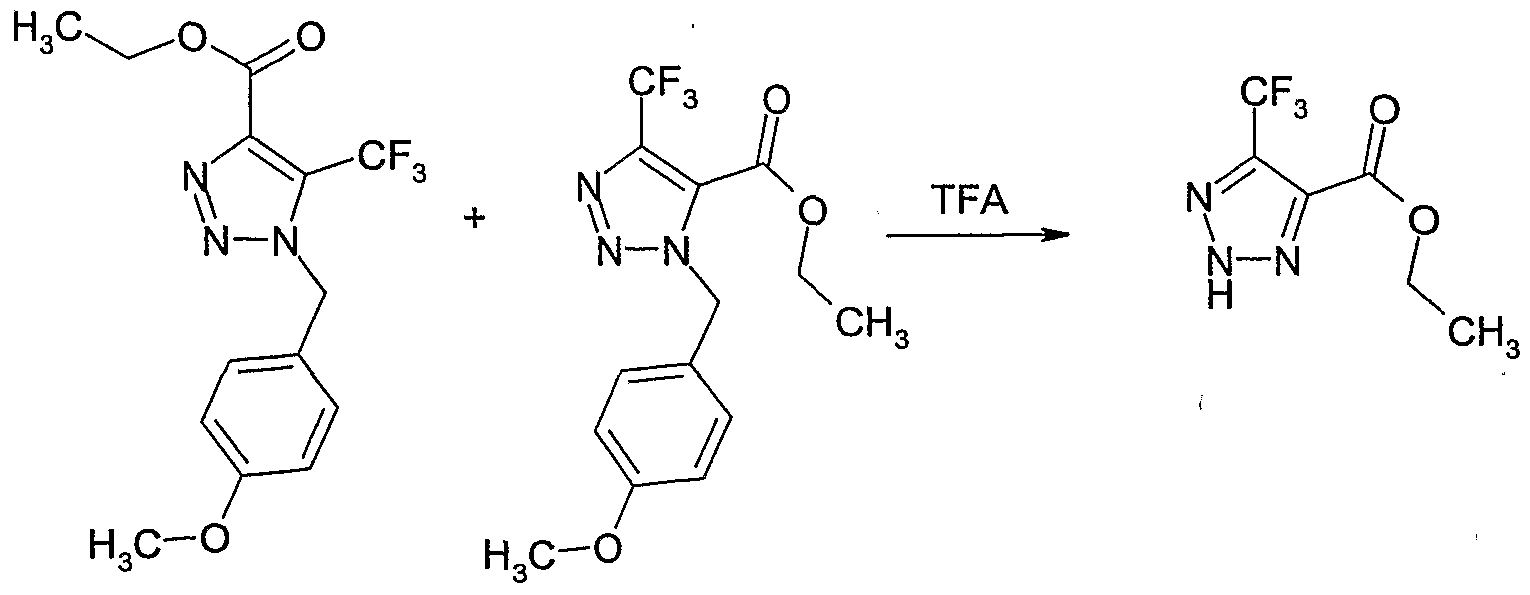
- the reaction mixture was cooled to CPC and quenched by sequential addition of methanol (75 ml), glacial acetic acid (5 ml) and water (1 ml). The mixture was extracted three times with ethyl acetate. The combined organic extracts were washed three times with brine, dried over magnesium sulfate and concentrated. The residue was purified by column chromatography on silica gel (eluent: 10-55% ethyl acetate in hexane) to give 5-methoxy-l-(4-methoxy-benzyl)-lH-[l,2,3]triazole-4-carboxylic acid ethyl ester as a straw coloured oil (15.2 g, 55% yield).
- reaction mixture was allowed to cool to room temperature and was quenched by addition of water (5 ml), ethyl acetate (10 ml) and aqueous hydrochloric acid (2M) (9 ml). The phases were separated and the aqueous phase extracted twice with more ethyl acetate. The combined organic extracts were washed with brine, dried over magnesium sulfate and concentrated.
- 5-Difluoromethoxy-lH-[l,2,3]triazole-4-carboxylic acid ethyl ester was methylated with methyl iodide according to the method described in Example 114 to give 5-difluoromethoxy-l-methyl-lH-[l,2,3]triazole-4-carboxylic acid ethyl ester (isomer A) and 5-difluoromethoxy-2-methyl-2H-[l,2,3]triazole-4-carboxylic acid ethyl ester (isomer
- reaction mixture was quenched with aqueous potassium dihydrogenphosphate (10%) and extracted three times with diethyl ether. The combined organic extracts were washed with water and brine, dried over magnesium sulfate and concentrated. The residue was purified by column chromatography on silica gel (eluent: diethyl ether / hexane) to give 2-(4-bromo-4,4- difluoro-but-2-ynyloxy)-tetrahydro-pyran as a colourless oil (17.6 g, 91.7% yield).
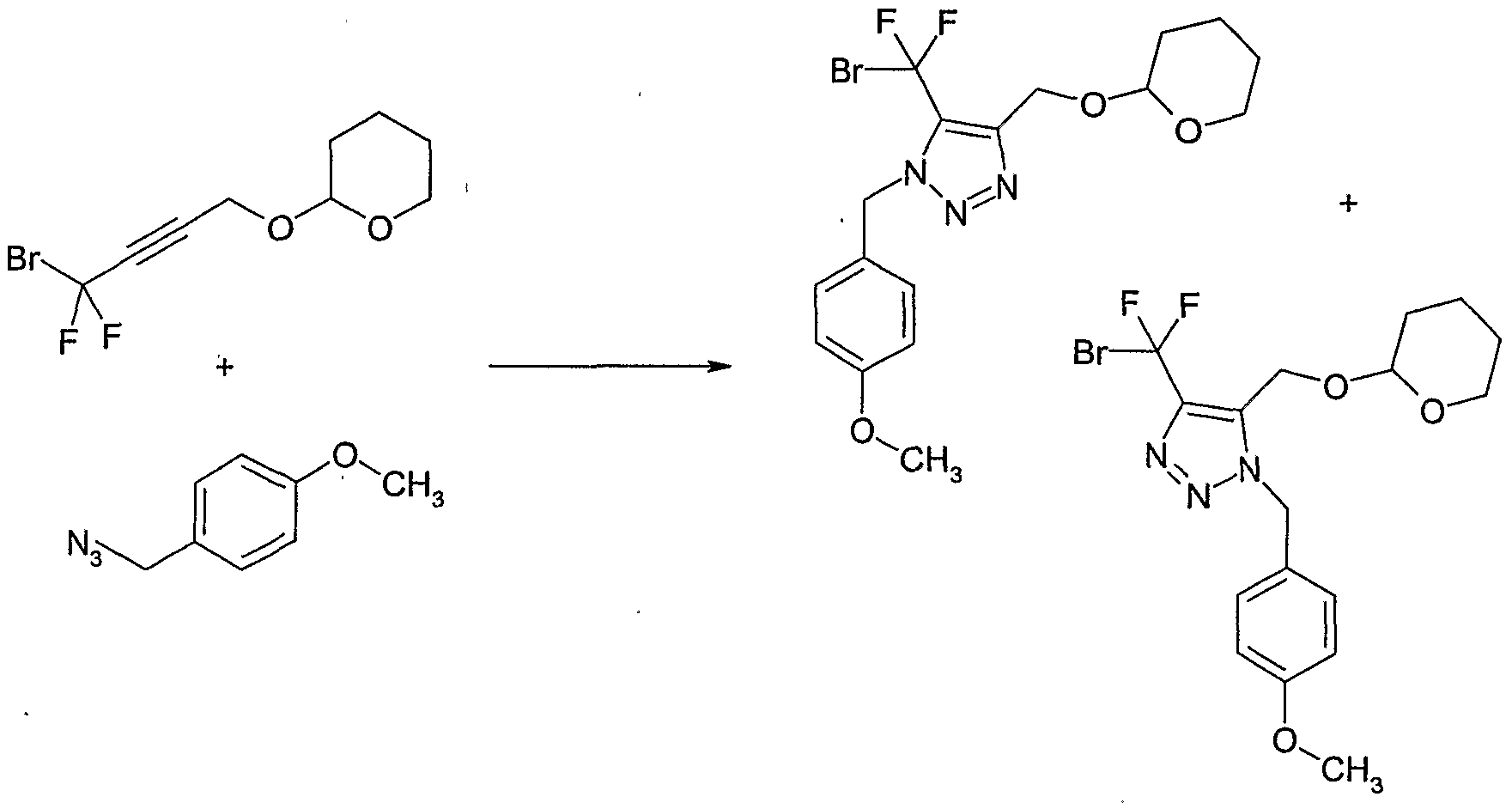
- Example 128 Preparation of 4-(bromo-difluoro-methyl)-l-(4-methoxy-benzyl)-5- (tetrahvdro-pyran-2-yloxymethylVlH-rL2,3]triazole and 5-(bromo-difluoro-methyl)-l- (4-methoxy-benzyl)-4-(tetrahvdro-pyran-2-yloxymethyl)-lH-
- reaction mixture was stirred at room temperature for lhour.
- the reaction mixture was concentrated and the residue purified by column chromatography on silica gel (eluent: ethyl acetate / hexane) to give a 2:1- mixture of [5-(bromo-difluoro-methyl)-3-(4-methoxy-benzyl)-3H-[ 1 ,2,3]triazol-4-yl]- methanol and [5-(bromo-difluoro-methyl)-l-(4-methoxy-benzyl)-lH-[l,2,3]triazol-4-yl]- methanol as a colourless oil (7.04 g, 82% yield).
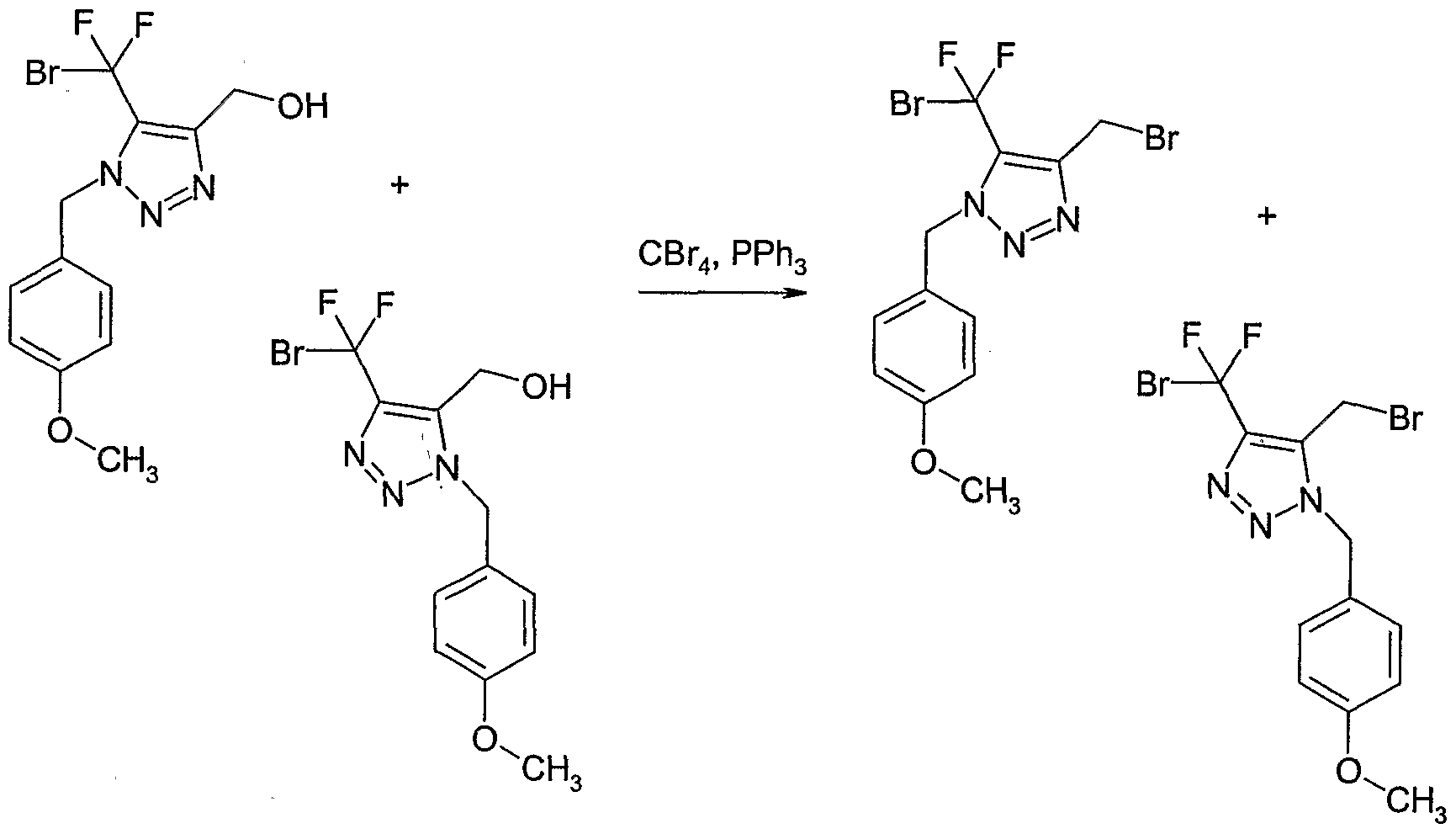
- Example 130 Preparation of 4-(bromo-difluoro-methyl ' )-5-bromomethyl-l-(4-methoxy- benzyl)-lH- ⁇ ,2,31triazole and 5-(bromo-difluoro-methyl)-4-bromomethyl-l-( ' 4- methoxy-benzyl)-lH-[l,2,31triazole
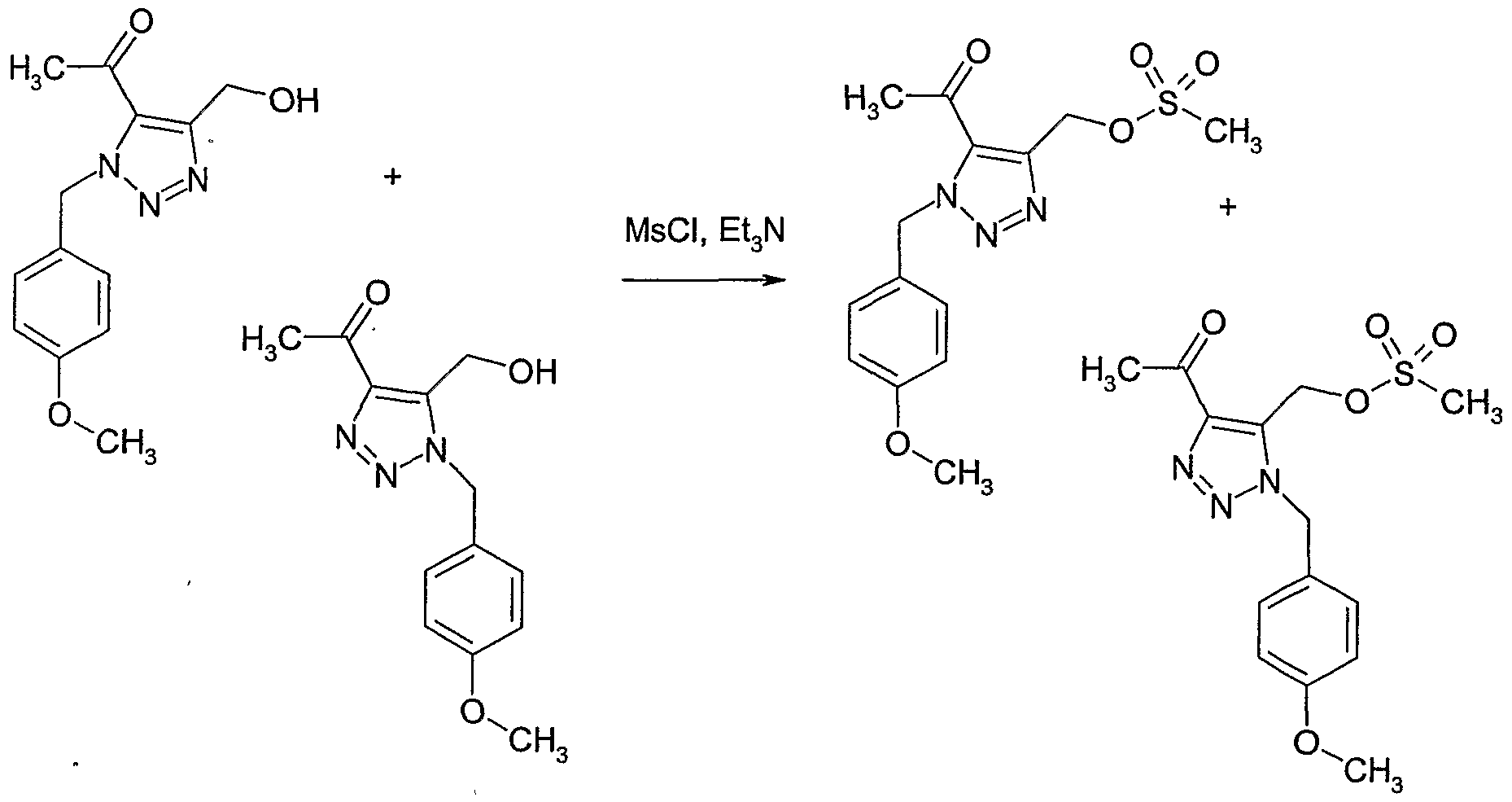
- Example 131 Preparation of methanesulfonic acid 5-acetyl-l-(4-methoxy-benzylH-H- [l,2,31triazol-4-ylmethyl ester and methanesulfonic acid 5-acetyl-3-(4-methoxy-benzyl)-
- reaction mixtures were concentrated and re-dissolved in acetonitrile.
- the solids were removed by filtration and washed with acetonitrile.
- the combined filtrates were concentrated to give a mixture of methanesulfonic acid 5-acetyl- l-(4-methoxy-benzyl)-lH-[l,2,3]triazol-4-ylmethyl ester and methanesulfonic acid 5- acetyl-3-(4-methoxy-benzyl)-3//-[l,2,3]triazol-4-ylmethyl ester.
- the mixture was used without further purification.
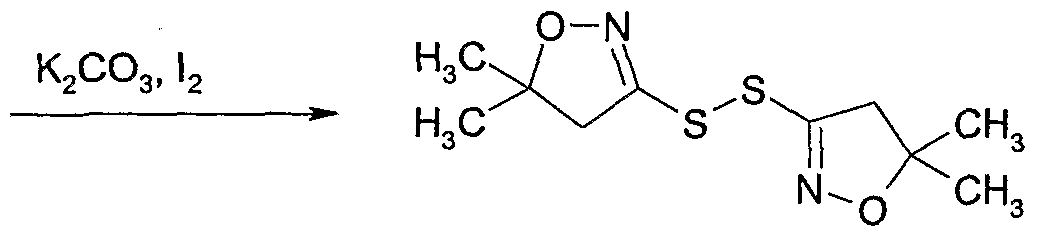
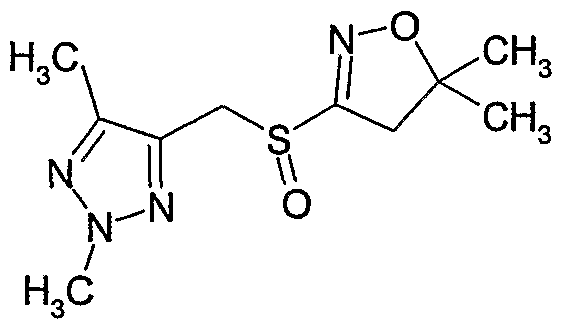
- Example 132 Preparation of 4-(bromo-difluoro-methyl)-5-(5,5-dimethyl-4,5-dihydro- isoxazol-3-ylsulfanylmethyl)-2-methyl-2.H-[ 1 ,2,31triazole
- Example 133 Preparation of l-r5-(5,5-dimethyl-4,5-dihydro-isoxazol-3-ylsulfanyl- methvl)-2-methvl-2#-ri ,2,31triazol-4-vl]-ethanol
- 2-Diazo-3-oxo-butyric acid ethyl ester (5 g, 32 mmol) was dissolved in tetra- hydrofuran (50 ml) and acetic acid (6 ml) and methylhydrazine was passed through the solution until a white precipitate crystallised out. More acetic acid (10 ml) was added and the solution was refluxed for 2 days.
- Example 136 Preparation of 5-cvclopropyl-l-metliyl-l//-[l,2,3]triazole-4-carboxylic acid ethyl ester
- Example 138 Preparation of 5-cyclopropyl-2-methyl-2H-[l,2,31triazole-4-carboxylic acid ethyl ester and 5-cvclopropyl-3-memyl-3H-ri,2.3 "
- Example 140 Preparation of 4-(bromo-difluoro-methyl)-5-(5,5-dimethyl-4,5-dihvdro- isoxazol-3-ylsulfanylmethvD-l-(4-methoxy-benzyl)-l/f-[l,2,3]triazole and 5-(bromo- difluoro-methyl)-4-(5,5-dimethyl-4,5-dihvdro-isoxazol-3-ylsulfanylmethyl)-l-(4- methoxy-benzyl)-l//-ri,2,31triazole
- Example 142 Alternative preparation of 4-(5,5-dimethyl-4,5-dihvdro-isoxazol-3-yl- sulfanylmethvn-2.5-dimethyl-2H-r 1.2.31triazole
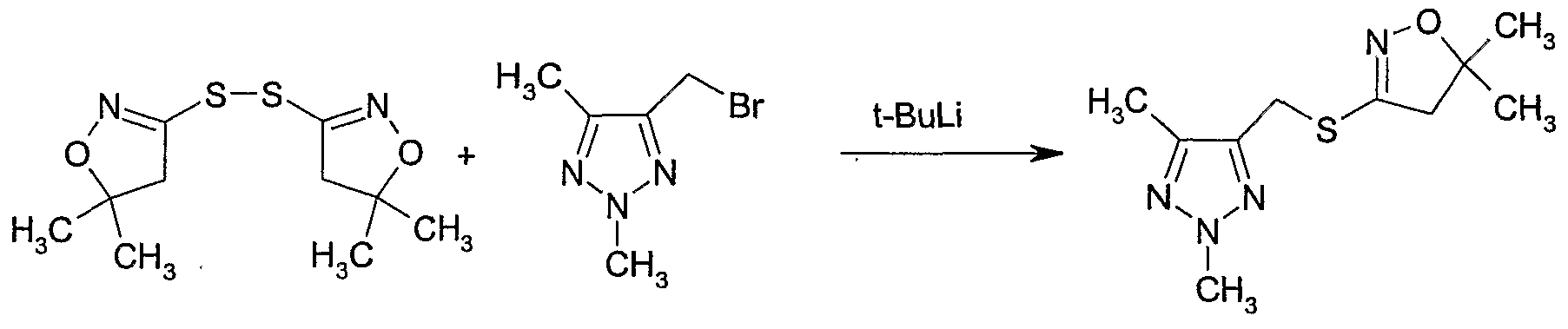
- tert-Buty ⁇ lithium (1.7 M in hexane) (1.12 ml, 1.89 mmol) was added to diethyl ether (5 ml) at -78 3 C.
- a solution of 4-bromomethyl-2,5-dimethyl-2i/-[l,2,3]triazole (Example 17, Example Il 1) (300 mg, 1.58 mmol) in diethyl ether (5 ml) was added at -7S 3 C and the reaction mixture stirred at -78 3 C for 5 minutes.
- Example 143 Alternative preparation of 4-(5,5-dimethyl-4,5-dihvdro-isoxazol-3-yl- sulfanylmethyl)-2,5-dimethyl-2H-r 1 ,2.31triazole
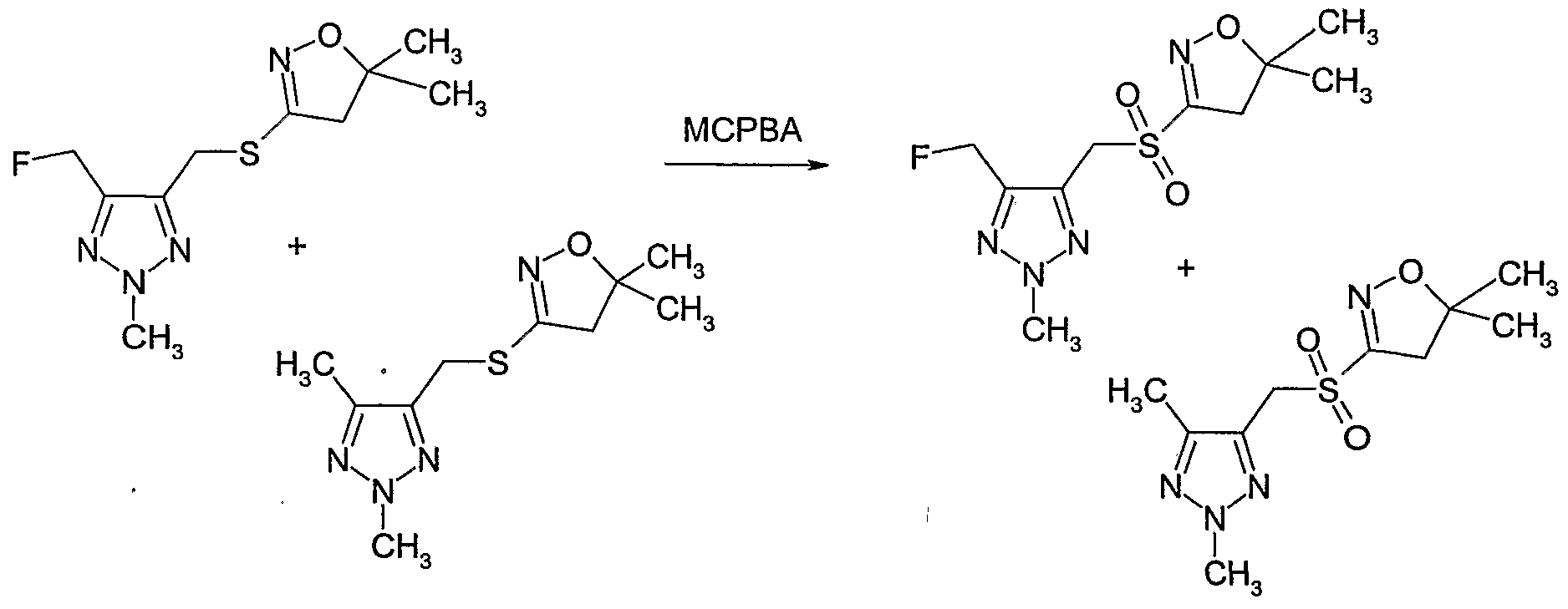
- [l,2,3]triazole (Example 139) (75% pure, 3.53g) was dissolved in dichloromethane and 3- chloroperoxybenzoic acid (MCPBA) (60% by weight) (4.2 g, 14.6 mmol) was added at CPC. The mixture was stirred for 30 minutes at CPC. The reaction mixture was quenched by addition of aqueous sodium metabisulfite (20%) (80 ml) and the mixture stirred for 10 minutes. The phases were separated and the aqueous phase was washed with aqueous sodium bicarbonate (saturated), water and brine, dried over magnesium sulfate and concentrated.
- MCPBA 3- chloroperoxybenzoic acid
- Example 145 Preparation of 4-(5,5-dimetliyl-4,5-dihvdro-isoxazol-3-ylsulfonylmethv ⁇ - 5-fluoromethyl-2-methyl-2H-[T,2,31triazole and 4-(5,5-dimethyl-4,5-dihydro-isoxazol-3- ylsulfonvlmethvl)-2. 5-dimethvl-2H-ri ,2,3
- reaction mixture was quenched by addition of aqueous sodium metabisulfite (10%).
- the mixture was diluted with water and aqueous sodium hydroxide (2M) was added.
- the phases were separated and the aqueous phase extracted with dichloromethane.
- the combined organic extracts were washed twice with aqueous sodium hydroxide (2M), dried over magnesium sulfate and concentrated.
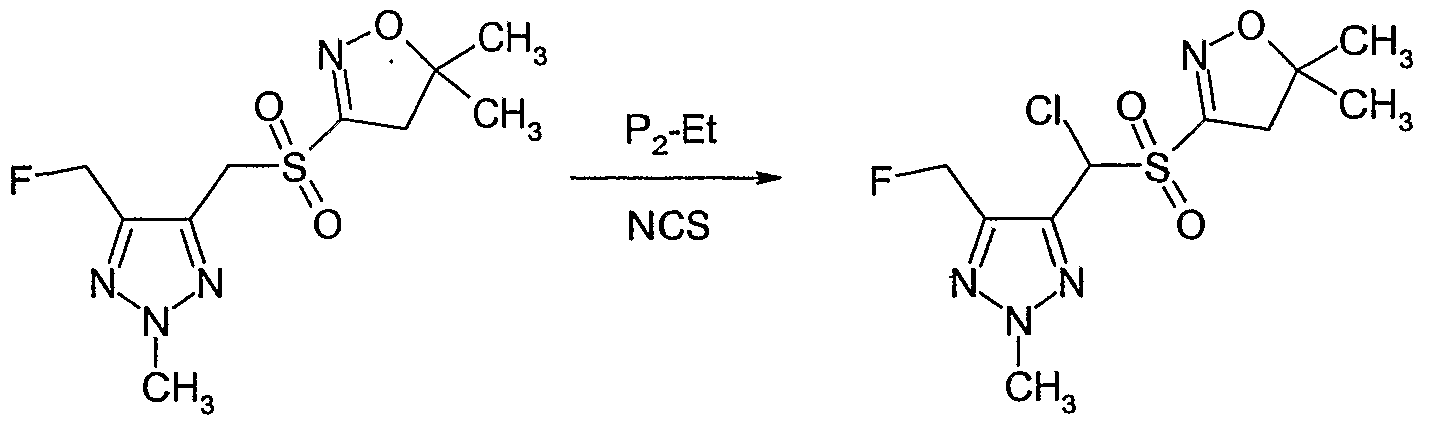
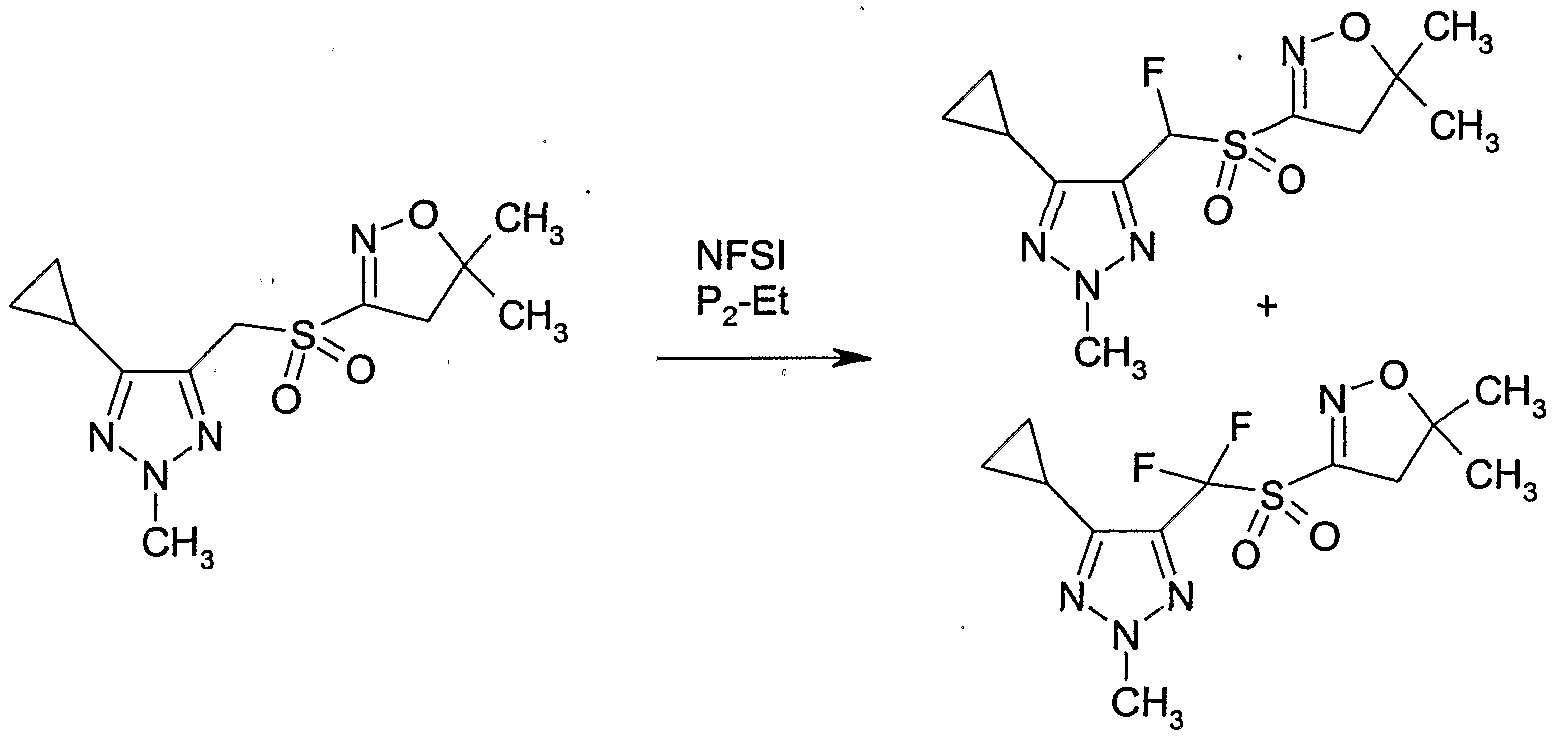
- Example P2 Preparation of 4-[chloro-(5,5-dimethyl-4,5-dihvdro-isoxazole-3-sulfonyP)- methyll-5-fluoromethyl-2-ethyl-2H-rL231triazole
- Example P2 4-[Chloro-(5,5-dimethyl-4,5-dihydro-isoxazole-3-sulfonyl)-methyl]-5-fluoro- methyl-2-ethyl-2H-[l,2,3]triazole (Example P2) (116 mg, 0.36 mmol) was dissolved in tetrahydrofuran (2 ml) under nitrogen and l-ethyl-2,2,4,4,4-pentakis(dimethylamino)-2- Iambda 5 ,41ambda 5 -catenadi(phosphazene) (P 2 -Et) (0.13 ml, 0.38 mmol) was added at room temperature.
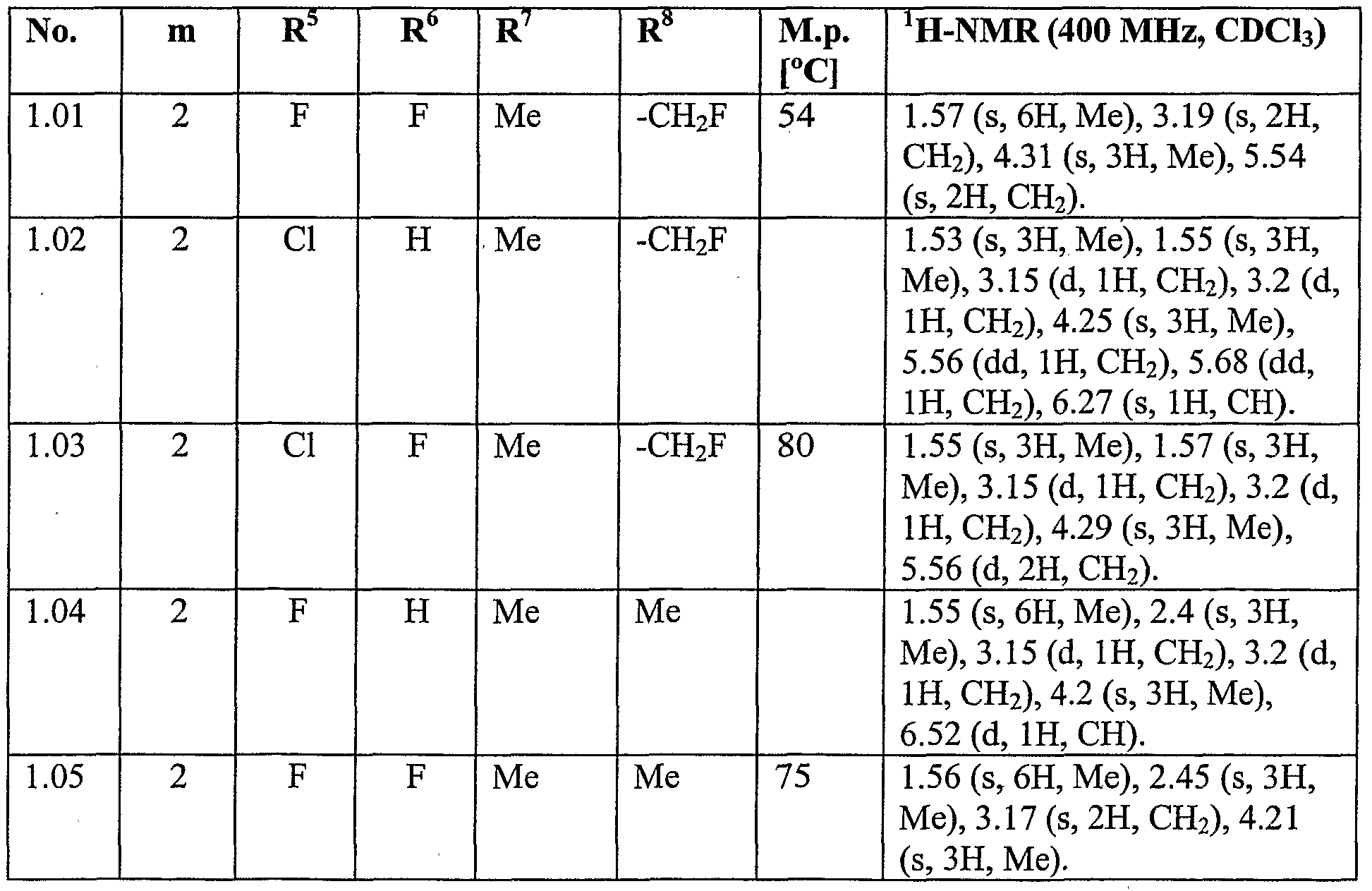
- N-Fluorobenzene sulfonimide (NFSI) (118 mg, 0.38 mmol) was added in one portion at room temperature and the reaction mixture was stirred for 1 hour at room temperature. The reaction mixture was concentrated and the residue was purified by column chromatography on silica gel (eluent: ethyl acetate / hexane) to give Compound No. 1.03 of Table 1 as a white solid (85 mg, 69% yield).
- Example P4 Preparation of 4-r(5,5-dimethyl-4,5-dihvdro-isoxazole-3-sulfonyl * )-fluoro- methvll-2. 5-dimethvl-2H- ⁇ ,2,3]triazole
- Example P5 Preparation of 4-r(5,5-dimethyl-4,5-dihvdro-isoxazole-3-sulfinvD-difluoro- methvll-2.5-dimethvl-2H-r 12 ,3]triazole
- Lithium hexamethyldisilazide (IM solution in THF) (1.64 ml, 1.64 mmol) was added dropwise at -78 0 C and the solution was stirred and allowed to warm slowly to -2O 0 C over 3 hours.
- the reaction mixture was quenched at -2CPCby addition of several drops of aqueous ammonium chloride (saturated).
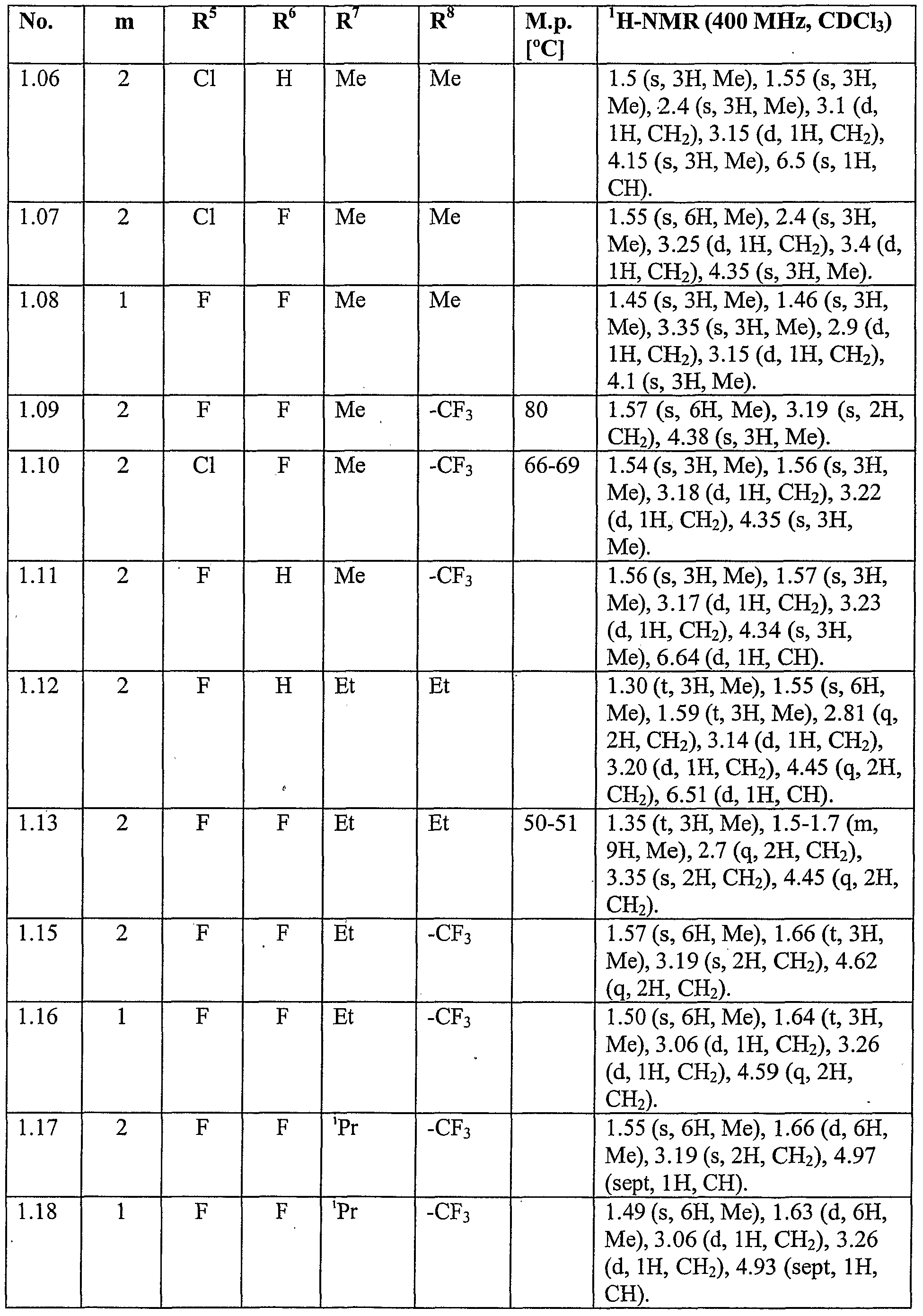
- the mixture was allowed to warm to room temperature, concentrated onto silica gel and purified by column chromatography on silica gel (eluent: hexane / ethyl acetate) to give Compound No. 1.08 of Table 1 (160 mg, 70% yield).
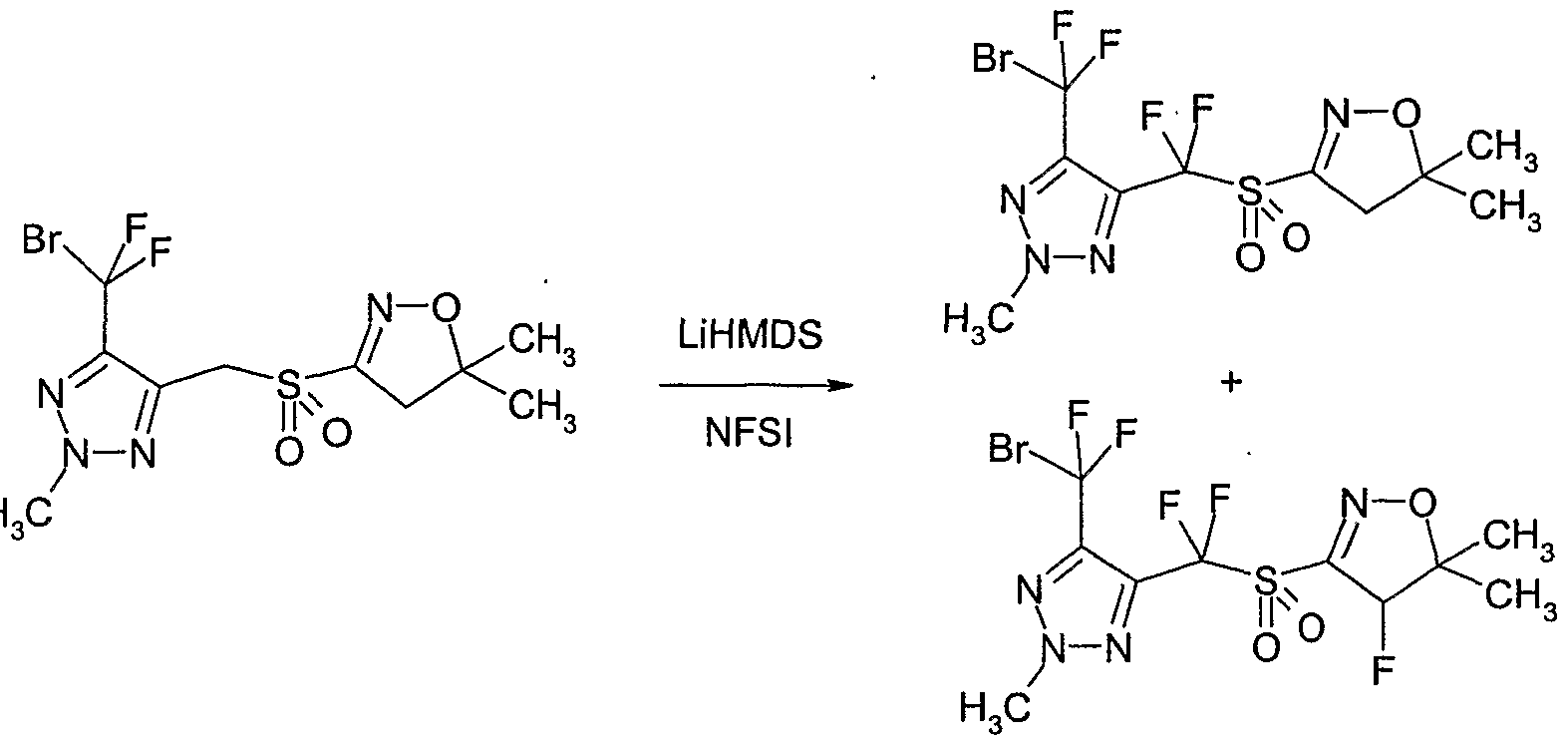
- Example P6 Preparation of 4-( ' bromo-difluoro-methyl)-5-[ " ( ' 5,5-dimethyl-4,5-dihvdro- isoxazole-S-sulfonvD-difluoro-methvn ⁇ -methyl- ⁇ H-fl ⁇ Jjtriazole
- Lithium hexamethyldisilazide (IM in THF) (2.8 ml, 2.8 mmol) was added dropwise at -78 3 C and the solution was stirred and allowed to warm slowly to room temperature over 4 hours.
- the reaction mixture was quenched at by addition of aqueous ammonium chloride (saturated) and was extracted several times with ethyl acetate. The combined organic fractions were washed with water and brine, dried over magnesium sulfate and concentrated. The residue was purified by column chromatography on silica gel (eluent: ethyl acetate / hexane) to give Compound No.
- Example P7 Preparation of 5-cyclopropyl-4-[(5,5-dimethyl-4,5-dihvdro-isoxazole-3- sulfinyl)-fluoro-methyll-l-methyl-lH-[l,2,31triazole and 5-cyclopropyl-4-( ' (5,5- dimethyl-4,5-dihvdro-isoxazole-3-sulfinyl)-difluoro-methvn-l-methyl-lH-ri,2,31triazole
- N- fluorobenzene sulfonimide (NFSI) (160 mg, 0.51 mmol) was added and the reaction mixture stirred for 2 hours at -78?C.
- the reaction mixture was quenched by addition of aqueous ammonium chloride (saturated) (3ml) and extracted with ethyl acetate. The organic phase was washed with water and brine, dried over magnesium sulfate and concentrated. The residue was purified by column chromatography on silica gel (eluent: ethyl acetate / iso-hexane) to give Comppund No. 2.10 of Table 2 (42 mg, 39% yield) and Compound No. 2.11 of Table 2 (45 mg, 44% yield).
- Example P8 Preparation of 5-cvclopropyl-4-[(5,5-dimethyl-4,5-dihvdro-isoxazole-3- sulfonylVfluoro-methvn-l-methyl-l.H-ri,2,3]triazole and 5-cyclopropyl-4-r(5,5- dimethyl-4,5-dihvdro-isoxazole-3-sulfonyl)-difluoro-methyl1-l-methyl-l//-[l,2,3]- ' triazole
- DIGSA Digitaria sanguinalis
- AGRTE Agrostis tenius
- SETIT Setaria italica
- SETFA Setaria faberi
- POATR Poa trivialis
- AMARE Amaranthus retroflexus.
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Life Sciences & Earth Sciences (AREA)
- Dentistry (AREA)
- Pest Control & Pesticides (AREA)
- Plant Pathology (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Agronomy & Crop Science (AREA)
- General Health & Medical Sciences (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Environmental Sciences (AREA)
- Agricultural Chemicals And Associated Chemicals (AREA)
- Plural Heterocyclic Compounds (AREA)
- Heterocyclic Carbon Compounds Containing A Hetero Ring Having Nitrogen And Oxygen As The Only Ring Hetero Atoms (AREA)
Abstract
Description
Claims
Priority Applications (12)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| AU2007217252A AU2007217252A1 (en) | 2006-02-27 | 2007-01-22 | Herbicidal isoxazoline compounds |
| JP2008555855A JP2009536149A (en) | 2006-02-27 | 2007-01-22 | Isoxazoline compounds for herbicidal use |
| DE602007013185T DE602007013185D1 (en) | 2006-02-27 | 2007-01-22 | HERBICIDE ISOXAZOLIN COMPOUNDS |
| PL07704963T PL1991542T3 (en) | 2006-02-27 | 2007-01-22 | Herbicidal isoxazoline compounds |
| AP2008004580A AP2008004580A0 (en) | 2006-02-27 | 2007-01-22 | Herbicidal isoyazoline compounds |
| BRPI0707973-7A BRPI0707973A2 (en) | 2006-02-27 | 2007-01-22 | compound, process in which a compound is formed, herbicidal composition, and method of controlling plants |
| EA200870299A EA200870299A1 (en) | 2006-02-27 | 2007-01-22 | Herbicide Isoxazole Compounds |
| CA002640962A CA2640962A1 (en) | 2006-02-27 | 2007-01-22 | Herbicidal isoxazoline compounds |
| US12/280,877 US20100004128A1 (en) | 2006-02-27 | 2007-01-22 | Herbicidal isoxazoline compounds |
| AT07704963T ATE502033T1 (en) | 2006-02-27 | 2007-01-22 | HERBICIDAL ISOXAZOLINE COMPOUNDS |
| EP07704963A EP1991542B1 (en) | 2006-02-27 | 2007-01-22 | Herbicidal isoxazoline compounds |
| IL193468A IL193468A0 (en) | 2006-02-27 | 2008-08-14 | Herbicidal isoxazoline compounds |
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| GB0603891.3 | 2006-02-27 | ||
| GBGB0603891.3A GB0603891D0 (en) | 2006-02-27 | 2006-02-27 | Novel herbicides |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2007096576A1 true WO2007096576A1 (en) | 2007-08-30 |
Family
ID=36178842
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/GB2007/000184 Ceased WO2007096576A1 (en) | 2006-02-27 | 2007-01-22 | Herbicidal isoxazoline compounds |
Country Status (21)
| Country | Link |
|---|---|
| US (1) | US20100004128A1 (en) |
| EP (1) | EP1991542B1 (en) |
| JP (1) | JP2009536149A (en) |
| KR (1) | KR20080097238A (en) |
| CN (1) | CN101389625A (en) |
| AP (1) | AP2008004580A0 (en) |
| AR (1) | AR059516A1 (en) |
| AT (1) | ATE502033T1 (en) |
| AU (1) | AU2007217252A1 (en) |
| BR (1) | BRPI0707973A2 (en) |
| CA (1) | CA2640962A1 (en) |
| DE (1) | DE602007013185D1 (en) |
| EA (1) | EA200870299A1 (en) |
| ES (1) | ES2363254T3 (en) |
| GB (1) | GB0603891D0 (en) |
| IL (1) | IL193468A0 (en) |
| PL (1) | PL1991542T3 (en) |
| UA (1) | UA92210C2 (en) |
| UY (1) | UY30177A1 (en) |
| WO (1) | WO2007096576A1 (en) |
| ZA (1) | ZA200806687B (en) |
Cited By (190)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008074991A1 (en) * | 2006-12-21 | 2008-06-26 | Syngenta Limited | Novel herbicides |
| WO2010116122A2 (en) | 2009-04-06 | 2010-10-14 | Syngenta Limited | Herbicidal compounds |
| WO2010145992A1 (en) | 2009-06-19 | 2010-12-23 | Basf Se | Herbicidal benzoxazinones |
| WO2011018486A2 (en) | 2009-08-14 | 2011-02-17 | Basf Se | Herbicidally active composition comprising benzoxazinones |
| WO2011033251A1 (en) | 2009-09-16 | 2011-03-24 | Syngenta Limited | Herbicidal isoxazoline derivatives |
| WO2011039172A2 (en) | 2009-09-30 | 2011-04-07 | Basf Se | Low volatile amine salts of anionic pesticides |
| WO2011051393A1 (en) | 2009-11-02 | 2011-05-05 | Basf Se | Herbicidal tetrahydrophthalimides |
| WO2011057935A1 (en) | 2009-11-13 | 2011-05-19 | Basf Se | 3-(3,4-dihydro-2h-benzo [1,4]oxazin-6-yl)-1h-pyrimidin-2,4-dione compounds as herbicides |
| WO2011063843A1 (en) | 2009-11-26 | 2011-06-03 | Basf Se | Method for producing 5,5-disubstituted 2-isoxazolines |
| WO2011063842A1 (en) * | 2009-11-26 | 2011-06-03 | Basf Se | Method for producing 5,5-disubstituted 4,5-dihydroisoxazol-3-thiocarboxamidine salts |
| WO2011134539A1 (en) | 2010-04-30 | 2011-11-03 | Basf Se | Use of oxylipins as safeners and safening herbicidal compositions comprising oxylipins |
| WO2012041789A1 (en) | 2010-10-01 | 2012-04-05 | Basf Se | Herbicidal benzoxazinones |
| WO2012045721A2 (en) | 2010-10-05 | 2012-04-12 | Syngenta Limited | Herbicidal compounds |
| WO2012080239A1 (en) | 2010-12-15 | 2012-06-21 | Basf Se | Herbicidal compositions |
| WO2012084755A1 (en) | 2010-12-23 | 2012-06-28 | Basf Se | Substituted pyridines having herbicidal activity |
| EP2474226A1 (en) | 2011-01-07 | 2012-07-11 | Basf Se | Herbicidally active composition comprising cyanobutyrates |
| US8263630B2 (en) | 2008-02-12 | 2012-09-11 | Bristol-Myers Squibb Company | 1,2,3-triazoles as 11-beta hydroxysteroid dehydrogenase type I inhibitors |
| WO2012136703A1 (en) | 2011-04-08 | 2012-10-11 | Syngenta Limited | Herbicidal compounds |
| WO2012168397A1 (en) | 2011-06-09 | 2012-12-13 | Basf Se | Substituted pyridines having herbicidal activity |
| WO2012168241A1 (en) | 2011-06-09 | 2012-12-13 | Basf Se | Substituted pyrazines having herbicidal activity |
| WO2013039990A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013040021A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013040117A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013040049A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013050421A1 (en) | 2011-10-04 | 2013-04-11 | Syngenta Limited | Herbicidal pyridazinone derivatives |
| WO2013083774A1 (en) | 2011-12-09 | 2013-06-13 | Syngenta Limited | Herbicidal compounds |
| WO2013092834A1 (en) | 2011-12-21 | 2013-06-27 | Syngenta Limited | Herbicidal compounds |
| WO2013139760A1 (en) | 2012-03-20 | 2013-09-26 | Syngenta Limited | Herbicidal compounds |
| WO2013144234A1 (en) | 2012-03-29 | 2013-10-03 | Syngenta Limited | 1,2,4 -triazole derivatives as herbicidals |
| WO2013144231A1 (en) | 2012-03-29 | 2013-10-03 | Syngenta Limited | Herbicidal compounds |
| DE112011104396T5 (en) | 2010-12-16 | 2013-10-10 | Basf Se | Plants with increased tolerance to herbicides |
| WO2013178585A1 (en) | 2012-06-01 | 2013-12-05 | Basf Se | Substituted pyridine compounds having herbicidal activity |
| WO2013189984A2 (en) | 2012-06-19 | 2013-12-27 | Basf Se | Plants having increased tolerance to herbicides |
| EP2700635A1 (en) | 2012-08-20 | 2014-02-26 | Basf Se | 5-Trifluoromethylpyrazole amides having herbicidal activity |
| EP2700634A1 (en) | 2012-08-20 | 2014-02-26 | Basf Se | 5-difluoromethylpyrazole amides having herbicidal activity |
| US8703829B2 (en) | 2007-02-06 | 2014-04-22 | University Of Louisville Research Foundation, Inc. | Therapeutic compounds |
| WO2014066164A1 (en) | 2012-10-26 | 2014-05-01 | E. I. Du Pont De Nemours And Company | Substituted triazoles as herbicides |
| WO2014102065A1 (en) | 2012-12-31 | 2014-07-03 | Basf Se | Herbicidal composition comprising a cornexistin |
| WO2014151255A1 (en) | 2013-03-15 | 2014-09-25 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2014187705A1 (en) | 2013-05-24 | 2014-11-27 | Basf Se | Substituted pyridine compounds having herbicidal activity |
| WO2014206835A1 (en) | 2013-06-26 | 2014-12-31 | Basf Se | Methods for improving the efficacy of anionic herbicides under hard water conditions and suitable compositions |
| EP2868197A1 (en) | 2013-11-05 | 2015-05-06 | Basf Se | Herbicidal compositions |
| EP2868196A1 (en) | 2013-11-05 | 2015-05-06 | Basf Se | Herbicidal compositions |
| WO2015108982A2 (en) | 2014-01-15 | 2015-07-23 | Monsanto Technology Llc | Methods and compositions for weed control using epsps polynucleotides |
| US9121022B2 (en) | 2010-03-08 | 2015-09-01 | Monsanto Technology Llc | Method for controlling herbicide-resistant plants |
| WO2015150541A1 (en) | 2014-04-03 | 2015-10-08 | Basf Se | Diaminotriazine compound useful as herbicide |
| WO2015150465A2 (en) | 2014-04-03 | 2015-10-08 | Basf Se | Plants having increased tolerance to herbicides |
| EP2930174A1 (en) | 2014-04-07 | 2015-10-14 | Basf Se | Diaminotriazine derivatives as herbicides |
| WO2015162169A1 (en) | 2014-04-23 | 2015-10-29 | Basf Se | Diaminotriazine compounds as herbicides |
| US9383364B2 (en) | 2011-03-07 | 2016-07-05 | University Of Louisville Research Foundation, Inc. | Predictive marker of DNMT1 inhibitor therapeutic efficacy and methods of using the marker |
| WO2016116870A1 (en) | 2015-01-21 | 2016-07-28 | Basf Se | Plants having increased tolerance to herbicides |
| WO2016120116A1 (en) | 2015-01-29 | 2016-08-04 | Basf Se | Herbicidal phenylpyridines |
| US9416363B2 (en) | 2011-09-13 | 2016-08-16 | Monsanto Technology Llc | Methods and compositions for weed control |
| US9422557B2 (en) | 2011-09-13 | 2016-08-23 | Monsanto Technology Llc | Methods and compositions for weed control |
| EP3112016A1 (en) | 2015-07-02 | 2017-01-04 | Basf Se | Microcapsules containing benzoxazinones |
| US9540642B2 (en) | 2013-11-04 | 2017-01-10 | The United States Of America, As Represented By The Secretary Of Agriculture | Compositions and methods for controlling arthropod parasite and pest infestations |
| EP3135113A1 (en) | 2015-08-31 | 2017-03-01 | Basf Se | Use of herbicidal compositions for controlling unwanted vegetation |
| WO2017080905A1 (en) | 2015-11-12 | 2017-05-18 | Basf Se | Herbicidal compositions comprising isoxazolo[5,4-b]pyridines |
| WO2017125395A1 (en) | 2016-01-22 | 2017-07-27 | Basf Se | Biodegradable polyester capsules comprising an aqueous core and a pesticide |
| US9737493B2 (en) | 2012-09-07 | 2017-08-22 | University Of Louisville Research Foundation, Inc. | Compositions and methods for modulating DNMT1 inhibitor activity |
| US9777288B2 (en) | 2013-07-19 | 2017-10-03 | Monsanto Technology Llc | Compositions and methods for controlling leptinotarsa |
| WO2017202774A1 (en) | 2016-05-24 | 2017-11-30 | Basf Se | Method for controlling ppo resistant weeds |
| WO2017202768A1 (en) | 2016-05-24 | 2017-11-30 | Basf Se | Herbicidal uracilpyrid |
| US9850496B2 (en) | 2013-07-19 | 2017-12-26 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| WO2018015180A1 (en) | 2016-07-20 | 2018-01-25 | Basf Se | Herbicidal compositions comprising phenylpyrimidines |
| EP3275877A1 (en) | 2016-07-28 | 2018-01-31 | Basf Se | Herbicidal pyridine compounds |
| WO2018019767A1 (en) | 2016-07-27 | 2018-02-01 | Basf Se | Herbicidal pyridine compounds |
| WO2018019758A1 (en) | 2016-07-26 | 2018-02-01 | Basf Se | Herbicidal pyridine compounds |
| WO2018019842A1 (en) | 2016-07-29 | 2018-02-01 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018019770A1 (en) | 2016-07-28 | 2018-02-01 | Basf Se | Herbicidal pyridine compounds |
| WO2018019721A1 (en) | 2016-07-26 | 2018-02-01 | Basf Se | Herbicidal pyridine compounds |
| WO2018019554A1 (en) | 2016-07-25 | 2018-02-01 | Basf Se | Herbicidal pyrimidine compounds |
| WO2018019629A1 (en) | 2016-07-27 | 2018-02-01 | Basf Se | Agroformulation of microcapsules with an anionic c6-c10 codispersant |
| WO2018019765A1 (en) | 2016-07-27 | 2018-02-01 | Basf Se | Herbicidal pyrimidine compounds |
| WO2018019860A1 (en) | 2016-07-27 | 2018-02-01 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| WO2018019552A1 (en) | 2016-07-25 | 2018-02-01 | Basf Se | Herbicidal pyrimidine compounds |
| WO2018019574A1 (en) | 2016-07-28 | 2018-02-01 | Basf Se | Herbicidal pyrimidine compounds |
| WO2018019755A1 (en) | 2016-07-26 | 2018-02-01 | Basf Se | Herbicidal pyridine compounds |
| WO2018019555A1 (en) | 2016-07-26 | 2018-02-01 | Basf Se | Herbicidal pyrimidine compounds |
| WO2018019845A1 (en) | 2016-07-29 | 2018-02-01 | Basf Se | Method for controlling ppo resistant weeds |
| EP3278667A1 (en) | 2016-08-05 | 2018-02-07 | Basf Se | Method for controlling ppo-inhibitor resistant weeds |
| WO2018024695A1 (en) | 2016-08-05 | 2018-02-08 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018024696A1 (en) | 2016-08-05 | 2018-02-08 | Basf Se | Method for controlling ppo resistant weeds |
| EP3281524A1 (en) | 2016-08-09 | 2018-02-14 | Basf Se | Method for controlling ppo resistant weeds |
| EP3281525A1 (en) | 2016-08-09 | 2018-02-14 | Basf Se | Method for controlling ppo resistant weeds |
| EP3281523A1 (en) | 2016-08-09 | 2018-02-14 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018029030A1 (en) | 2016-08-09 | 2018-02-15 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018029031A1 (en) | 2016-08-09 | 2018-02-15 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018029029A1 (en) | 2016-08-09 | 2018-02-15 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018077875A1 (en) | 2016-10-28 | 2018-05-03 | Syngenta Participations Ag | Herbicidal pyridazinone compounds |
| WO2018095811A1 (en) | 2016-11-28 | 2018-05-31 | Basf Se | Diaminotriazine compounds |
| WO2018108695A1 (en) | 2016-12-16 | 2018-06-21 | Basf Se | Herbicidal phenyltriazolinones |
| WO2018114759A1 (en) | 2016-12-20 | 2018-06-28 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| US10041087B2 (en) | 2012-06-19 | 2018-08-07 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| US10041068B2 (en) | 2013-01-01 | 2018-08-07 | A. B. Seeds Ltd. | Isolated dsRNA molecules and methods of using same for silencing target molecules of interest |
| WO2018141594A1 (en) | 2017-02-02 | 2018-08-09 | Basf Se | Enhancement of soil herbicide activity with anionic alkoxylated phenols |
| WO2018166822A1 (en) | 2017-03-14 | 2018-09-20 | Basf Se | Herbicidal azines |
| US10087460B2 (en) | 2013-08-12 | 2018-10-02 | BASF Agro B.V. | Transgenic or non-transgenic plants with mutated protoporphyrinogen oxidase having increased tolerance to herbicides |
| WO2018178039A1 (en) | 2017-03-31 | 2018-10-04 | Basf Se | Crystalline forms of ethyl[3-[2-chloro-4-fluoro-5-(1-methyl-6-trifluoromethyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-3-yl)phenoxy]-2-pyridyloxy]acetate |
| WO2018229041A1 (en) | 2017-06-14 | 2018-12-20 | Basf Se | Herbicidal pyrimidine compounds |
| WO2019030104A1 (en) | 2017-08-09 | 2019-02-14 | Basf Se | Herbicidal mixtures comprising l-glufosinate or its salt and at least one vlcfa inhibitor |
| US10240161B2 (en) | 2012-05-24 | 2019-03-26 | A.B. Seeds Ltd. | Compositions and methods for silencing gene expression |
| WO2019101560A1 (en) | 2017-11-22 | 2019-05-31 | Basf Se | Benzoxaborole compounds |
| WO2019101513A1 (en) | 2017-11-23 | 2019-05-31 | Basf Se | Herbicidal pyridylethers |
| WO2019101533A1 (en) | 2017-11-27 | 2019-05-31 | Basf Se | Crystalline forms of ethyl 2-[[3-[[3-chloro-5-fluoro-6-[3-methyl-2,6-dioxo-4-(trifluoromethyl)pyrimidin-1-yl]-2-pyridyl]oxy]-2-pyridyl]oxy]acetate |
| WO2019101551A1 (en) | 2017-11-23 | 2019-05-31 | Basf Se | Herbicidal phenylethers |
| WO2019106568A1 (en) | 2017-11-29 | 2019-06-06 | Basf Se | Plants having increased tolerance to herbicides |
| WO2019121352A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Herbicidal pyrimidine compounds |
| WO2019121373A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Herbicidal pyrimidine compounds |
| WO2019121408A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Herbicidal pyrimidine compounds |
| WO2019121374A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Herbicidal pyrimidine compounds |
| US10378012B2 (en) | 2014-07-29 | 2019-08-13 | Monsanto Technology Llc | Compositions and methods for controlling insect pests |
| WO2019158378A1 (en) | 2018-02-16 | 2019-08-22 | Basf Se | Herbicidal mixtures |
| US10392630B2 (en) | 2013-08-12 | 2019-08-27 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019238427A1 (en) | 2018-06-13 | 2019-12-19 | Basf Se | Crystalline form of ethyl 2-[2-[2-chloro-4-fluoro-5-[3-methyl-2,6-dioxo-4-(trifluoromethyl)pyrimidin-1-yl]phenoxy]phenoxy]acetate |
| WO2019243358A1 (en) | 2018-06-19 | 2019-12-26 | Syngenta Crop Protection Ag | Herbicidal compounds |
| US10557138B2 (en) | 2013-12-10 | 2020-02-11 | Beeologics, Inc. | Compositions and methods for virus control in Varroa mite and bees |
| WO2020058009A1 (en) | 2018-09-18 | 2020-03-26 | Basf Se | Diaminotriazine compounds |
| US10612019B2 (en) | 2013-03-13 | 2020-04-07 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10609930B2 (en) | 2013-03-13 | 2020-04-07 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10655136B2 (en) | 2015-06-03 | 2020-05-19 | Monsanto Technology Llc | Methods and compositions for introducing nucleic acids into plants |
| US10683505B2 (en) | 2013-01-01 | 2020-06-16 | Monsanto Technology Llc | Methods of introducing dsRNA to plant seeds for modulating gene expression |
| US10760086B2 (en) | 2011-09-13 | 2020-09-01 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10801028B2 (en) | 2009-10-14 | 2020-10-13 | Beeologics Inc. | Compositions for controlling Varroa mites in bees |
| US10808249B2 (en) | 2011-09-13 | 2020-10-20 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10806146B2 (en) | 2011-09-13 | 2020-10-20 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10829828B2 (en) | 2011-09-13 | 2020-11-10 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10883103B2 (en) | 2015-06-02 | 2021-01-05 | Monsanto Technology Llc | Compositions and methods for delivery of a polynucleotide into a plant |
| US10888579B2 (en) | 2007-11-07 | 2021-01-12 | Beeologics Inc. | Compositions for conferring tolerance to viral disease in social insects, and the use thereof |
| WO2021013969A1 (en) | 2019-07-25 | 2021-01-28 | Syngenta Crop Protection Ag | 3-[(hydrazono)methyl]-n-(tetrazol-5-yl)-benzamide and 3-[(hydrazono)methyl]-n-(1,3,4-oxadiazol-2-yl)-benzamide derivatives as herbicides |
| WO2021002484A3 (en) * | 2019-10-31 | 2021-03-11 | クミアイ化学工業株式会社 | Herbicide and production method for intermediate thereof |
| US10968449B2 (en) | 2015-01-22 | 2021-04-06 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US10988764B2 (en) | 2014-06-23 | 2021-04-27 | Monsanto Technology Llc | Compositions and methods for regulating gene expression via RNA interference |
| WO2021148302A1 (en) | 2020-01-23 | 2021-07-29 | Basf Se | Novel additives for agrochemical formulations |
| US11091770B2 (en) | 2014-04-01 | 2021-08-17 | Monsanto Technology Llc | Compositions and methods for controlling insect pests |
| EP3868749A1 (en) | 2016-03-23 | 2021-08-25 | Syngenta Participations Ag | Herbicidal compounds |
| WO2021170464A1 (en) | 2020-02-28 | 2021-09-02 | Basf Se | Herbicidal malonamides |
| US11109591B2 (en) | 2017-04-24 | 2021-09-07 | Taminco Bvba | Single phase liquids of alkanolamine salts of dicamba |
| WO2021191035A1 (en) | 2020-03-25 | 2021-09-30 | Basf Se | Dioxazolines and their use as herbicides |
| WO2021209268A1 (en) | 2020-04-14 | 2021-10-21 | Basf Se | Beta-lactams and their use as herbicides |
| WO2021224040A1 (en) | 2020-05-05 | 2021-11-11 | Basf Se | Condensed isoxazoline derivatives and their use as herbicides |
| CN113754602A (en) * | 2020-06-02 | 2021-12-07 | 山东润博生物科技有限公司 | Synthesis method of 5, 5-dimethyl-4, 5-dihydroisoxazole-3-ketone |
| WO2022008285A1 (en) | 2020-07-07 | 2022-01-13 | BASF Agro B.V. | Method for controlling undesirable vegetation in an aquatic environment |
| US11311014B2 (en) | 2017-03-31 | 2022-04-26 | Basf Se | Crystalline forms of ethyl[3-[2-chloro-4-fluoro-5-(1-methyl-6-trifluoromethyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-3-yl)phenoxy]-2-pyridyloxy]acetate |
| WO2022112351A1 (en) | 2020-11-25 | 2022-06-02 | Basf Se | Herbicidal malonamides |
| WO2022112347A1 (en) | 2020-11-25 | 2022-06-02 | Basf Se | Herbicidal malonamides |
| EP4008185A1 (en) | 2020-12-03 | 2022-06-08 | BASF Agro B.V. | Herbicidal composition comprising a saflufenacil microparticles |
| EP4011205A1 (en) | 2020-12-08 | 2022-06-15 | Basf Se | Microparticle compositions comprising trifludimoxazin |
| WO2022161801A1 (en) | 2021-01-27 | 2022-08-04 | Basf Se | Diaminotriazine compounds |
| WO2022161802A1 (en) | 2021-01-27 | 2022-08-04 | Basf Se | Diaminotriazine compounds |
| EP4049749A1 (en) | 2021-02-25 | 2022-08-31 | Basf Se | New microcapsules containing active substances |
| EP4056038A1 (en) | 2021-03-10 | 2022-09-14 | Basf Se | Microparticles containing active substances |
| WO2022189190A1 (en) | 2021-03-09 | 2022-09-15 | Basf Se | Malonamides and their use as herbicides |
| US11499162B2 (en) | 2016-07-15 | 2022-11-15 | Basf Se | Plants having increased tolerance to herbicides |
| WO2022268564A1 (en) | 2021-06-23 | 2022-12-29 | Basf Se | Herbicidal aryldihydrofurane carboxylates |
| WO2023025854A1 (en) | 2021-08-25 | 2023-03-02 | Basf Se | Herbicidal malonamides |
| WO2023025855A1 (en) | 2021-08-25 | 2023-03-02 | Basf Se | Herbicidal malonamides |
| WO2023030934A1 (en) | 2021-08-31 | 2023-03-09 | Basf Se | Herbicidal composition comprising phenyluracils |
| WO2023031161A1 (en) | 2021-09-03 | 2023-03-09 | BASF Agricultural Solutions Seed US LLC | Plants having increased tolerance to herbicides |
| WO2023031200A1 (en) | 2021-08-31 | 2023-03-09 | Basf Se | Herbicidal malonamides containing a condensed ring system |
| WO2023031199A1 (en) | 2021-08-31 | 2023-03-09 | Basf Se | Herbicidal malonamides containing monocyclic heteroaromatic rings |
| WO2023036636A1 (en) | 2021-09-09 | 2023-03-16 | Basf Se | New microparticles containing active substances |
| WO2023110491A1 (en) | 2021-12-16 | 2023-06-22 | BASF Agro B.V. | Method for controlling undesirable vegetation in an aquatic environment |
| EP4177252A3 (en) * | 2020-07-10 | 2023-08-09 | Adama Agan Ltd. | Process and intermediates for the preparation of pyroxasulfone, fenoxasulfone and various sulfone analogs of 5,5-dimethyl-4h-1,2-oxazole |
| WO2023208447A1 (en) | 2022-04-25 | 2023-11-02 | Basf Se | An emulsifiable concentrate having a (substituted) benzaldehyde-based solvent system |
| US11807857B2 (en) | 2014-06-25 | 2023-11-07 | Monsanto Technology Llc | Methods and compositions for delivering nucleic acids to plant cells and regulating gene expression |
| WO2023227676A1 (en) | 2022-05-25 | 2023-11-30 | Basf Se | Herbicidal malonic acid monoamides and malonamide esters |
| WO2023247489A1 (en) | 2022-06-21 | 2023-12-28 | Astrazeneca Ab | N-(2-(4-cyanothiazolidin-3-yl)-2-oxoethyl)- quinoline-4-carboxamides |
| WO2024012905A1 (en) | 2022-07-13 | 2024-01-18 | Basf Se | Herbicidal composition comprising azine compounds |
| EP4338592A1 (en) | 2022-09-15 | 2024-03-20 | Basf Se | Use of compound for improving the efficacy of herbicides |
| WO2024056517A1 (en) | 2022-09-14 | 2024-03-21 | Basf Se | Use of an alkylether sulfate for improving the efficacy of herbicides |
| US11939587B2 (en) | 2018-01-17 | 2024-03-26 | Basf Se | Plants having increased tolerance to herbicides |
| US11959086B2 (en) | 2016-05-20 | 2024-04-16 | BASF Agro B.V. | Dual transit peptides for targeting polypeptides |
| WO2024126113A1 (en) | 2022-12-12 | 2024-06-20 | BASF Agricultural Solutions Seed US LLC | Plants having increased tolerance to herbicides |
| US12018267B2 (en) | 2015-10-22 | 2024-06-25 | Basf Se | Plants having increased tolerance to herbicides |
| US12043626B2 (en) | 2021-10-01 | 2024-07-23 | Nuvalent, Inc. | Solid forms, pharmaceutical compositions and preparation of heteroaromatic macrocyclic ether compounds |
| US12054498B2 (en) | 2020-05-05 | 2024-08-06 | Nuvalent, Inc. | Heteroaromatic macrocyclic ether chemotherapeutic agents |
| US12203089B2 (en) | 2015-10-22 | 2025-01-21 | Basf Se | Plants having increased tolerance to herbicides |
| US12275742B2 (en) | 2020-05-05 | 2025-04-15 | Nuvalent, Inc. | Heteroaromatic macrocyclic ether chemotherapeutic agents |
| WO2025114052A1 (en) | 2023-11-29 | 2025-06-05 | Basf Se | Ternary herbicidal composition comprising azine compounds |
| US12338446B2 (en) | 2015-06-17 | 2025-06-24 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| WO2025153657A2 (en) | 2024-01-17 | 2025-07-24 | Basf Se | Plants having increased tolerance to herbicides |
| WO2025153595A1 (en) | 2024-01-17 | 2025-07-24 | Basf Se | Plants having increased tolerance to herbicides |
| EP4617275A1 (en) | 2022-11-09 | 2025-09-17 | Qingdao KingAgroot Chemical Compound Co., Ltd. | Oxabicycloalkane compounds, preparation method therefor, and herbicidal composition and use thereof |
| US12459904B2 (en) | 2019-04-16 | 2025-11-04 | Bristol-Myers Squibb Company | Process for preparing carbamoyloxymethyl triazole cyclohexyl acid compounds |
| WO2025242699A1 (en) | 2024-05-22 | 2025-11-27 | Basf Se | Method for improving rainfastness of an agrochemical active ingredient |
| WO2026003150A1 (en) | 2024-06-28 | 2026-01-02 | Basf Se | Plants with mutated tubulin polypeptide having increased tolerance to herbicides |
| WO2026021913A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | Herbicidal 2-phenoxybutanoylamides |
Families Citing this family (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1938689A1 (en) * | 2006-12-18 | 2008-07-02 | Bayer CropScience AG | Penetration promoter for herbicides |
| CN102408385B (en) * | 2011-06-20 | 2014-06-18 | 雅本化学股份有限公司 | Preparation method of 2-substituent-2H-1,2,3-triazole derivative |
| CN102408386B (en) * | 2011-07-27 | 2014-05-07 | 雅本化学股份有限公司 | Preparation method of 2,4-disubstituted-2H-1, 2, 3-triazole derivatives |
| CN102276540A (en) * | 2011-08-08 | 2011-12-14 | 中国科学院长春应用化学研究所 | Method for synthesizing 1,2,3-triazole compounds |
| CN105628837B (en) * | 2015-12-18 | 2017-03-15 | 山东省农业科学院农业质量标准与检测技术研究所 | The detection method of mesotrione and its metabolite in a kind of soil |
| KR20200112860A (en) * | 2017-12-29 | 2020-10-05 | 바이오마린 파머수티컬 인크. | Glycolate oxidase inhibitors for treatment of diseases |
| CN110483424A (en) * | 2018-06-11 | 2019-11-22 | 上海睿升化工科技有限公司 | A kind of preparation method of 1-H-1,2,3- triazole -4- carboxylic acid tert-butyl ester |
| CN111393427B (en) * | 2020-04-30 | 2021-11-30 | 安徽久易农业股份有限公司 | Synthetic method of sulfuryl pyraflufen |
| CN114478507B (en) * | 2020-11-12 | 2025-11-18 | 东莞市东阳光农药研发有限公司 | Pyrimidine oxazole compounds and their applications |
| US20240391909A1 (en) * | 2021-10-29 | 2024-11-28 | Kumiai Chemical Industry Co., Ltd. | Disulfide compound, polysulfide compound, and use thereof |
| WO2023074828A1 (en) * | 2021-10-29 | 2023-05-04 | クミアイ化学工業株式会社 | Disulfide compound, polysulfide compound, and use thereof |
| CN116102454B (en) * | 2021-11-10 | 2025-03-25 | 江西仰立新材料有限公司 | A preparation method of 2-(hydroxyimino)acetic acid |
| CN113929637B (en) * | 2021-11-17 | 2024-02-06 | 山东大学 | Sulfur-containing dihydro isoxazole compound and synthetic method thereof |
| CN114105810A (en) * | 2021-12-07 | 2022-03-01 | 山东科加工业技术研究院有限公司 | Preparation method of chloramine |
Citations (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001012613A1 (en) * | 1999-08-10 | 2001-02-22 | Kumiai Chemical Industry Co., Ltd. | Isoxazoline derivatives and herbicides containing the same as the active ingredient |
| WO2002062770A1 (en) * | 2001-02-08 | 2002-08-15 | Kumiai Chemical Industry Co., Ltd. | Isoxazoline derivative and herbicide comprising the same as active ingredient |
| WO2003000686A1 (en) * | 2001-06-21 | 2003-01-03 | Kumiai Chemical Industry Co., Ltd. | Isoxazoline derivatives and herbicides |
| JP2005035924A (en) * | 2003-07-14 | 2005-02-10 | Kumiai Chem Ind Co Ltd | 2-Isoxazoline derivatives and herbicides containing them as active ingredients |
| WO2005104848A1 (en) * | 2004-04-30 | 2005-11-10 | Syngenta Participations Ag | Herbicidal composition |
| US20050256004A1 (en) * | 2002-08-07 | 2005-11-17 | Satoru Takahashi | Herbicide compositions |
| WO2006024820A1 (en) * | 2004-09-03 | 2006-03-09 | Syngenta Limited | Isoxazoline derivatives and their use as herbicides |
-
2006
- 2006-02-27 GB GBGB0603891.3A patent/GB0603891D0/en not_active Ceased
-
2007
- 2007-01-22 CA CA002640962A patent/CA2640962A1/en not_active Abandoned
- 2007-01-22 BR BRPI0707973-7A patent/BRPI0707973A2/en not_active IP Right Cessation
- 2007-01-22 EP EP07704963A patent/EP1991542B1/en active Active
- 2007-01-22 US US12/280,877 patent/US20100004128A1/en not_active Abandoned
- 2007-01-22 EA EA200870299A patent/EA200870299A1/en unknown
- 2007-01-22 AP AP2008004580A patent/AP2008004580A0/en unknown
- 2007-01-22 KR KR1020087023443A patent/KR20080097238A/en not_active Withdrawn
- 2007-01-22 WO PCT/GB2007/000184 patent/WO2007096576A1/en not_active Ceased
- 2007-01-22 AT AT07704963T patent/ATE502033T1/en not_active IP Right Cessation
- 2007-01-22 AU AU2007217252A patent/AU2007217252A1/en not_active Abandoned
- 2007-01-22 ES ES07704963T patent/ES2363254T3/en active Active
- 2007-01-22 CN CNA2007800067404A patent/CN101389625A/en active Pending
- 2007-01-22 JP JP2008555855A patent/JP2009536149A/en active Pending
- 2007-01-22 PL PL07704963T patent/PL1991542T3/en unknown
- 2007-01-22 UA UAA200811550A patent/UA92210C2/en unknown
- 2007-01-22 DE DE602007013185T patent/DE602007013185D1/en active Active
- 2007-02-15 AR ARP070100654A patent/AR059516A1/en unknown
- 2007-02-27 UY UY30177A patent/UY30177A1/en unknown
-
2008
- 2008-07-31 ZA ZA200806687A patent/ZA200806687B/en unknown
- 2008-08-14 IL IL193468A patent/IL193468A0/en unknown
Patent Citations (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2001012613A1 (en) * | 1999-08-10 | 2001-02-22 | Kumiai Chemical Industry Co., Ltd. | Isoxazoline derivatives and herbicides containing the same as the active ingredient |
| EP1203768A1 (en) * | 1999-08-10 | 2002-05-08 | Kumiai Chemical Industry Co., Ltd. | Isoxazoline derivatives and herbicides containing the same as the active ingredient |
| WO2002062770A1 (en) * | 2001-02-08 | 2002-08-15 | Kumiai Chemical Industry Co., Ltd. | Isoxazoline derivative and herbicide comprising the same as active ingredient |
| EP1364946A1 (en) * | 2001-02-08 | 2003-11-26 | Kumiai Chemical Industry Co., Ltd. | Isoxazoline derivative and herbicide comprising the same as active ingredient |
| WO2003000686A1 (en) * | 2001-06-21 | 2003-01-03 | Kumiai Chemical Industry Co., Ltd. | Isoxazoline derivatives and herbicides |
| EP1405853A1 (en) * | 2001-06-21 | 2004-04-07 | Kumiai Chemical Industry Co., Ltd. | Isoxazoline derivatives and herbicides |
| US20050256004A1 (en) * | 2002-08-07 | 2005-11-17 | Satoru Takahashi | Herbicide compositions |
| JP2005035924A (en) * | 2003-07-14 | 2005-02-10 | Kumiai Chem Ind Co Ltd | 2-Isoxazoline derivatives and herbicides containing them as active ingredients |
| WO2005104848A1 (en) * | 2004-04-30 | 2005-11-10 | Syngenta Participations Ag | Herbicidal composition |
| WO2006024820A1 (en) * | 2004-09-03 | 2006-03-09 | Syngenta Limited | Isoxazoline derivatives and their use as herbicides |
Cited By (239)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2008074991A1 (en) * | 2006-12-21 | 2008-06-26 | Syngenta Limited | Novel herbicides |
| US8703829B2 (en) | 2007-02-06 | 2014-04-22 | University Of Louisville Research Foundation, Inc. | Therapeutic compounds |
| US10888579B2 (en) | 2007-11-07 | 2021-01-12 | Beeologics Inc. | Compositions for conferring tolerance to viral disease in social insects, and the use thereof |
| US8263630B2 (en) | 2008-02-12 | 2012-09-11 | Bristol-Myers Squibb Company | 1,2,3-triazoles as 11-beta hydroxysteroid dehydrogenase type I inhibitors |
| WO2010116122A2 (en) | 2009-04-06 | 2010-10-14 | Syngenta Limited | Herbicidal compounds |
| WO2010145992A1 (en) | 2009-06-19 | 2010-12-23 | Basf Se | Herbicidal benzoxazinones |
| WO2011018486A2 (en) | 2009-08-14 | 2011-02-17 | Basf Se | Herbicidally active composition comprising benzoxazinones |
| WO2011033251A1 (en) | 2009-09-16 | 2011-03-24 | Syngenta Limited | Herbicidal isoxazoline derivatives |
| EP2638803A1 (en) | 2009-09-30 | 2013-09-18 | Basf Se | Low volatile amine salts of anionic pesticides |
| EP2638804A1 (en) | 2009-09-30 | 2013-09-18 | Basf Se | Low volatile amine salts of anionic pesticides |
| WO2011039172A2 (en) | 2009-09-30 | 2011-04-07 | Basf Se | Low volatile amine salts of anionic pesticides |
| US10801028B2 (en) | 2009-10-14 | 2020-10-13 | Beeologics Inc. | Compositions for controlling Varroa mites in bees |
| WO2011051393A1 (en) | 2009-11-02 | 2011-05-05 | Basf Se | Herbicidal tetrahydrophthalimides |
| WO2011057935A1 (en) | 2009-11-13 | 2011-05-19 | Basf Se | 3-(3,4-dihydro-2h-benzo [1,4]oxazin-6-yl)-1h-pyrimidin-2,4-dione compounds as herbicides |
| WO2011063842A1 (en) * | 2009-11-26 | 2011-06-03 | Basf Se | Method for producing 5,5-disubstituted 4,5-dihydroisoxazol-3-thiocarboxamidine salts |
| WO2011063843A1 (en) | 2009-11-26 | 2011-06-03 | Basf Se | Method for producing 5,5-disubstituted 2-isoxazolines |
| US8921572B2 (en) | 2009-11-26 | 2014-12-30 | Basf Se | Method for producing 5,5-disubstituted 4,5-dihydroisoxazol-3-thiocarboxamidine salts |
| US11812738B2 (en) | 2010-03-08 | 2023-11-14 | Monsanto Technology Llc | Polynucleotide molecules for gene regulation in plants |
| US9988634B2 (en) | 2010-03-08 | 2018-06-05 | Monsanto Technology Llc | Polynucleotide molecules for gene regulation in plants |
| US9121022B2 (en) | 2010-03-08 | 2015-09-01 | Monsanto Technology Llc | Method for controlling herbicide-resistant plants |
| WO2011134539A1 (en) | 2010-04-30 | 2011-11-03 | Basf Se | Use of oxylipins as safeners and safening herbicidal compositions comprising oxylipins |
| WO2012041789A1 (en) | 2010-10-01 | 2012-04-05 | Basf Se | Herbicidal benzoxazinones |
| WO2012045721A2 (en) | 2010-10-05 | 2012-04-12 | Syngenta Limited | Herbicidal compounds |
| WO2012080239A1 (en) | 2010-12-15 | 2012-06-21 | Basf Se | Herbicidal compositions |
| US11274313B2 (en) | 2010-12-16 | 2022-03-15 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| DE112011104396T5 (en) | 2010-12-16 | 2013-10-10 | Basf Se | Plants with increased tolerance to herbicides |
| WO2012084755A1 (en) | 2010-12-23 | 2012-06-28 | Basf Se | Substituted pyridines having herbicidal activity |
| EP2474226A1 (en) | 2011-01-07 | 2012-07-11 | Basf Se | Herbicidally active composition comprising cyanobutyrates |
| US9383364B2 (en) | 2011-03-07 | 2016-07-05 | University Of Louisville Research Foundation, Inc. | Predictive marker of DNMT1 inhibitor therapeutic efficacy and methods of using the marker |
| WO2012136703A1 (en) | 2011-04-08 | 2012-10-11 | Syngenta Limited | Herbicidal compounds |
| WO2012168241A1 (en) | 2011-06-09 | 2012-12-13 | Basf Se | Substituted pyrazines having herbicidal activity |
| WO2012168397A1 (en) | 2011-06-09 | 2012-12-13 | Basf Se | Substituted pyridines having herbicidal activity |
| US9422558B2 (en) | 2011-09-13 | 2016-08-23 | Monsanto Technology Llc | Methods and compositions for weed control |
| US9422557B2 (en) | 2011-09-13 | 2016-08-23 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013040021A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10760086B2 (en) | 2011-09-13 | 2020-09-01 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10808249B2 (en) | 2011-09-13 | 2020-10-20 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013040049A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| US9416363B2 (en) | 2011-09-13 | 2016-08-16 | Monsanto Technology Llc | Methods and compositions for weed control |
| EP3434779A1 (en) | 2011-09-13 | 2019-01-30 | Monsanto Technology LLC | Methods and compositions for weed control |
| EP3434780A1 (en) | 2011-09-13 | 2019-01-30 | Monsanto Technology LLC | Methods and compositions for weed control |
| EP3382027A2 (en) | 2011-09-13 | 2018-10-03 | Monsanto Technology LLC | Methods and compositions for weed control |
| US10806146B2 (en) | 2011-09-13 | 2020-10-20 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10829828B2 (en) | 2011-09-13 | 2020-11-10 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013040117A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2013039990A1 (en) | 2011-09-13 | 2013-03-21 | Monsanto Technology Llc | Methods and compositions for weed control |
| EP3296402A2 (en) | 2011-09-13 | 2018-03-21 | Monsanto Technology LLC | Methods and compositions for weed control |
| WO2013050421A1 (en) | 2011-10-04 | 2013-04-11 | Syngenta Limited | Herbicidal pyridazinone derivatives |
| WO2013083774A1 (en) | 2011-12-09 | 2013-06-13 | Syngenta Limited | Herbicidal compounds |
| WO2013092834A1 (en) | 2011-12-21 | 2013-06-27 | Syngenta Limited | Herbicidal compounds |
| WO2013139760A1 (en) | 2012-03-20 | 2013-09-26 | Syngenta Limited | Herbicidal compounds |
| WO2013144234A1 (en) | 2012-03-29 | 2013-10-03 | Syngenta Limited | 1,2,4 -triazole derivatives as herbicidals |
| WO2013144231A1 (en) | 2012-03-29 | 2013-10-03 | Syngenta Limited | Herbicidal compounds |
| US10240161B2 (en) | 2012-05-24 | 2019-03-26 | A.B. Seeds Ltd. | Compositions and methods for silencing gene expression |
| US10240162B2 (en) | 2012-05-24 | 2019-03-26 | A.B. Seeds Ltd. | Compositions and methods for silencing gene expression |
| US10934555B2 (en) | 2012-05-24 | 2021-03-02 | Monsanto Technology Llc | Compositions and methods for silencing gene expression |
| WO2013178585A1 (en) | 2012-06-01 | 2013-12-05 | Basf Se | Substituted pyridine compounds having herbicidal activity |
| US12529066B2 (en) | 2012-06-19 | 2026-01-20 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| US11572571B2 (en) | 2012-06-19 | 2023-02-07 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| US10100329B2 (en) | 2012-06-19 | 2018-10-16 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| US11441154B2 (en) | 2012-06-19 | 2022-09-13 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| US10041087B2 (en) | 2012-06-19 | 2018-08-07 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| WO2013189984A2 (en) | 2012-06-19 | 2013-12-27 | Basf Se | Plants having increased tolerance to herbicides |
| EP2700634A1 (en) | 2012-08-20 | 2014-02-26 | Basf Se | 5-difluoromethylpyrazole amides having herbicidal activity |
| EP2700635A1 (en) | 2012-08-20 | 2014-02-26 | Basf Se | 5-Trifluoromethylpyrazole amides having herbicidal activity |
| US9737493B2 (en) | 2012-09-07 | 2017-08-22 | University Of Louisville Research Foundation, Inc. | Compositions and methods for modulating DNMT1 inhibitor activity |
| WO2014066164A1 (en) | 2012-10-26 | 2014-05-01 | E. I. Du Pont De Nemours And Company | Substituted triazoles as herbicides |
| WO2014102065A1 (en) | 2012-12-31 | 2014-07-03 | Basf Se | Herbicidal composition comprising a cornexistin |
| US10683505B2 (en) | 2013-01-01 | 2020-06-16 | Monsanto Technology Llc | Methods of introducing dsRNA to plant seeds for modulating gene expression |
| US10041068B2 (en) | 2013-01-01 | 2018-08-07 | A. B. Seeds Ltd. | Isolated dsRNA molecules and methods of using same for silencing target molecules of interest |
| US10609930B2 (en) | 2013-03-13 | 2020-04-07 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10612019B2 (en) | 2013-03-13 | 2020-04-07 | Monsanto Technology Llc | Methods and compositions for weed control |
| US10568328B2 (en) | 2013-03-15 | 2020-02-25 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2014151255A1 (en) | 2013-03-15 | 2014-09-25 | Monsanto Technology Llc | Methods and compositions for weed control |
| WO2014187705A1 (en) | 2013-05-24 | 2014-11-27 | Basf Se | Substituted pyridine compounds having herbicidal activity |
| WO2014206835A1 (en) | 2013-06-26 | 2014-12-31 | Basf Se | Methods for improving the efficacy of anionic herbicides under hard water conditions and suitable compositions |
| US9777288B2 (en) | 2013-07-19 | 2017-10-03 | Monsanto Technology Llc | Compositions and methods for controlling leptinotarsa |
| US9856495B2 (en) | 2013-07-19 | 2018-01-02 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US9850496B2 (en) | 2013-07-19 | 2017-12-26 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US11377667B2 (en) | 2013-07-19 | 2022-07-05 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US10597676B2 (en) | 2013-07-19 | 2020-03-24 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| US10392630B2 (en) | 2013-08-12 | 2019-08-27 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| US10087460B2 (en) | 2013-08-12 | 2018-10-02 | BASF Agro B.V. | Transgenic or non-transgenic plants with mutated protoporphyrinogen oxidase having increased tolerance to herbicides |
| US10968462B2 (en) | 2013-08-12 | 2021-04-06 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| US10982227B2 (en) | 2013-08-12 | 2021-04-20 | BASF Agro B.V. | Transgenic or non-transgenic plants with mutated protoporphyrinogen oxidase having increased tolerance to herbicides |
| US11866720B2 (en) | 2013-08-12 | 2024-01-09 | BASF Agro B.V. | Transgenic or non-transgenic plants with mutated protoporphyrinogen oxidase having increased tolerance to herbicides |
| US11827896B2 (en) | 2013-08-12 | 2023-11-28 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| US9540642B2 (en) | 2013-11-04 | 2017-01-10 | The United States Of America, As Represented By The Secretary Of Agriculture | Compositions and methods for controlling arthropod parasite and pest infestations |
| US10927374B2 (en) | 2013-11-04 | 2021-02-23 | Monsanto Technology Llc | Compositions and methods for controlling arthropod parasite and pest infestations |
| US10100306B2 (en) | 2013-11-04 | 2018-10-16 | Monsanto Technology Llc | Compositions and methods for controlling arthropod parasite and pest infestations |
| EP2868196A1 (en) | 2013-11-05 | 2015-05-06 | Basf Se | Herbicidal compositions |
| EP2868197A1 (en) | 2013-11-05 | 2015-05-06 | Basf Se | Herbicidal compositions |
| US10557138B2 (en) | 2013-12-10 | 2020-02-11 | Beeologics, Inc. | Compositions and methods for virus control in Varroa mite and bees |
| WO2015108982A2 (en) | 2014-01-15 | 2015-07-23 | Monsanto Technology Llc | Methods and compositions for weed control using epsps polynucleotides |
| US10334848B2 (en) | 2014-01-15 | 2019-07-02 | Monsanto Technology Llc | Methods and compositions for weed control using EPSPS polynucleotides |
| US11091770B2 (en) | 2014-04-01 | 2021-08-17 | Monsanto Technology Llc | Compositions and methods for controlling insect pests |
| WO2015150541A1 (en) | 2014-04-03 | 2015-10-08 | Basf Se | Diaminotriazine compound useful as herbicide |
| WO2015150465A2 (en) | 2014-04-03 | 2015-10-08 | Basf Se | Plants having increased tolerance to herbicides |
| EP2930174A1 (en) | 2014-04-07 | 2015-10-14 | Basf Se | Diaminotriazine derivatives as herbicides |
| WO2015162169A1 (en) | 2014-04-23 | 2015-10-29 | Basf Se | Diaminotriazine compounds as herbicides |
| US10988764B2 (en) | 2014-06-23 | 2021-04-27 | Monsanto Technology Llc | Compositions and methods for regulating gene expression via RNA interference |
| US11807857B2 (en) | 2014-06-25 | 2023-11-07 | Monsanto Technology Llc | Methods and compositions for delivering nucleic acids to plant cells and regulating gene expression |
| US10378012B2 (en) | 2014-07-29 | 2019-08-13 | Monsanto Technology Llc | Compositions and methods for controlling insect pests |
| US11124792B2 (en) | 2014-07-29 | 2021-09-21 | Monsanto Technology Llc | Compositions and methods for controlling insect pests |
| WO2016116870A1 (en) | 2015-01-21 | 2016-07-28 | Basf Se | Plants having increased tolerance to herbicides |
| US10968449B2 (en) | 2015-01-22 | 2021-04-06 | Monsanto Technology Llc | Compositions and methods for controlling Leptinotarsa |
| WO2016120116A1 (en) | 2015-01-29 | 2016-08-04 | Basf Se | Herbicidal phenylpyridines |
| US10883103B2 (en) | 2015-06-02 | 2021-01-05 | Monsanto Technology Llc | Compositions and methods for delivery of a polynucleotide into a plant |
| US10655136B2 (en) | 2015-06-03 | 2020-05-19 | Monsanto Technology Llc | Methods and compositions for introducing nucleic acids into plants |
| US12338446B2 (en) | 2015-06-17 | 2025-06-24 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| EP3112016A1 (en) | 2015-07-02 | 2017-01-04 | Basf Se | Microcapsules containing benzoxazinones |
| EP3135113A1 (en) | 2015-08-31 | 2017-03-01 | Basf Se | Use of herbicidal compositions for controlling unwanted vegetation |
| US12203089B2 (en) | 2015-10-22 | 2025-01-21 | Basf Se | Plants having increased tolerance to herbicides |
| US12018267B2 (en) | 2015-10-22 | 2024-06-25 | Basf Se | Plants having increased tolerance to herbicides |
| WO2017080905A1 (en) | 2015-11-12 | 2017-05-18 | Basf Se | Herbicidal compositions comprising isoxazolo[5,4-b]pyridines |
| WO2017125395A1 (en) | 2016-01-22 | 2017-07-27 | Basf Se | Biodegradable polyester capsules comprising an aqueous core and a pesticide |
| EP3868749A1 (en) | 2016-03-23 | 2021-08-25 | Syngenta Participations Ag | Herbicidal compounds |
| US11959086B2 (en) | 2016-05-20 | 2024-04-16 | BASF Agro B.V. | Dual transit peptides for targeting polypeptides |
| WO2017202774A1 (en) | 2016-05-24 | 2017-11-30 | Basf Se | Method for controlling ppo resistant weeds |
| WO2017202768A1 (en) | 2016-05-24 | 2017-11-30 | Basf Se | Herbicidal uracilpyrid |
| US11499162B2 (en) | 2016-07-15 | 2022-11-15 | Basf Se | Plants having increased tolerance to herbicides |
| WO2018015180A1 (en) | 2016-07-20 | 2018-01-25 | Basf Se | Herbicidal compositions comprising phenylpyrimidines |
| WO2018019554A1 (en) | 2016-07-25 | 2018-02-01 | Basf Se | Herbicidal pyrimidine compounds |
| WO2018019552A1 (en) | 2016-07-25 | 2018-02-01 | Basf Se | Herbicidal pyrimidine compounds |
| WO2018019758A1 (en) | 2016-07-26 | 2018-02-01 | Basf Se | Herbicidal pyridine compounds |
| WO2018019555A1 (en) | 2016-07-26 | 2018-02-01 | Basf Se | Herbicidal pyrimidine compounds |
| WO2018019755A1 (en) | 2016-07-26 | 2018-02-01 | Basf Se | Herbicidal pyridine compounds |
| WO2018019721A1 (en) | 2016-07-26 | 2018-02-01 | Basf Se | Herbicidal pyridine compounds |
| WO2018019860A1 (en) | 2016-07-27 | 2018-02-01 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| WO2018019767A1 (en) | 2016-07-27 | 2018-02-01 | Basf Se | Herbicidal pyridine compounds |
| US12279612B2 (en) | 2016-07-27 | 2025-04-22 | Basf Se | Agroformulation of microcapsules with an anionic C6—C10 codispersant |
| US11149030B2 (en) | 2016-07-27 | 2021-10-19 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| WO2018019629A1 (en) | 2016-07-27 | 2018-02-01 | Basf Se | Agroformulation of microcapsules with an anionic c6-c10 codispersant |
| WO2018019765A1 (en) | 2016-07-27 | 2018-02-01 | Basf Se | Herbicidal pyrimidine compounds |
| WO2018019770A1 (en) | 2016-07-28 | 2018-02-01 | Basf Se | Herbicidal pyridine compounds |
| WO2018019574A1 (en) | 2016-07-28 | 2018-02-01 | Basf Se | Herbicidal pyrimidine compounds |
| EP3275877A1 (en) | 2016-07-28 | 2018-01-31 | Basf Se | Herbicidal pyridine compounds |
| WO2018019845A1 (en) | 2016-07-29 | 2018-02-01 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018019842A1 (en) | 2016-07-29 | 2018-02-01 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018024695A1 (en) | 2016-08-05 | 2018-02-08 | Basf Se | Method for controlling ppo resistant weeds |
| EP3278667A1 (en) | 2016-08-05 | 2018-02-07 | Basf Se | Method for controlling ppo-inhibitor resistant weeds |
| WO2018024696A1 (en) | 2016-08-05 | 2018-02-08 | Basf Se | Method for controlling ppo resistant weeds |
| EP3281524A1 (en) | 2016-08-09 | 2018-02-14 | Basf Se | Method for controlling ppo resistant weeds |
| EP3281525A1 (en) | 2016-08-09 | 2018-02-14 | Basf Se | Method for controlling ppo resistant weeds |
| EP3281523A1 (en) | 2016-08-09 | 2018-02-14 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018029030A1 (en) | 2016-08-09 | 2018-02-15 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018029031A1 (en) | 2016-08-09 | 2018-02-15 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018029029A1 (en) | 2016-08-09 | 2018-02-15 | Basf Se | Method for controlling ppo resistant weeds |
| WO2018077875A1 (en) | 2016-10-28 | 2018-05-03 | Syngenta Participations Ag | Herbicidal pyridazinone compounds |
| WO2018095811A1 (en) | 2016-11-28 | 2018-05-31 | Basf Se | Diaminotriazine compounds |
| WO2018108695A1 (en) | 2016-12-16 | 2018-06-21 | Basf Se | Herbicidal phenyltriazolinones |
| US11879132B2 (en) | 2016-12-20 | 2024-01-23 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| WO2018114759A1 (en) | 2016-12-20 | 2018-06-28 | BASF Agro B.V. | Plants having increased tolerance to herbicides |
| WO2018141594A1 (en) | 2017-02-02 | 2018-08-09 | Basf Se | Enhancement of soil herbicide activity with anionic alkoxylated phenols |
| WO2018166822A1 (en) | 2017-03-14 | 2018-09-20 | Basf Se | Herbicidal azines |
| WO2018178039A1 (en) | 2017-03-31 | 2018-10-04 | Basf Se | Crystalline forms of ethyl[3-[2-chloro-4-fluoro-5-(1-methyl-6-trifluoromethyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-3-yl)phenoxy]-2-pyridyloxy]acetate |
| US11311014B2 (en) | 2017-03-31 | 2022-04-26 | Basf Se | Crystalline forms of ethyl[3-[2-chloro-4-fluoro-5-(1-methyl-6-trifluoromethyl-2,4-dioxo-1,2,3,4-tetrahydropyrimidin-3-yl)phenoxy]-2-pyridyloxy]acetate |
| US11109591B2 (en) | 2017-04-24 | 2021-09-07 | Taminco Bvba | Single phase liquids of alkanolamine salts of dicamba |
| WO2018229041A1 (en) | 2017-06-14 | 2018-12-20 | Basf Se | Herbicidal pyrimidine compounds |
| US11350631B2 (en) | 2017-08-09 | 2022-06-07 | Basf Se | Herbicidal mixtures comprising l-glufosinate or its salt and at least one VLCFA inhibitor |
| US12133530B2 (en) | 2017-08-09 | 2024-11-05 | Basf Se | Herbicidal mixtures comprising L-glufosinate or its salt and at least one VLCFA inhibitor |
| WO2019030104A1 (en) | 2017-08-09 | 2019-02-14 | Basf Se | Herbicidal mixtures comprising l-glufosinate or its salt and at least one vlcfa inhibitor |
| US12075782B2 (en) | 2017-08-09 | 2024-09-03 | Basf Se | Herbicidal mixtures comprising L-glufosinate or its salt and at least one VLCFA inhibitor |
| WO2019101560A1 (en) | 2017-11-22 | 2019-05-31 | Basf Se | Benzoxaborole compounds |
| WO2019101513A1 (en) | 2017-11-23 | 2019-05-31 | Basf Se | Herbicidal pyridylethers |
| WO2019101551A1 (en) | 2017-11-23 | 2019-05-31 | Basf Se | Herbicidal phenylethers |
| WO2019101533A1 (en) | 2017-11-27 | 2019-05-31 | Basf Se | Crystalline forms of ethyl 2-[[3-[[3-chloro-5-fluoro-6-[3-methyl-2,6-dioxo-4-(trifluoromethyl)pyrimidin-1-yl]-2-pyridyl]oxy]-2-pyridyl]oxy]acetate |
| WO2019106568A1 (en) | 2017-11-29 | 2019-06-06 | Basf Se | Plants having increased tolerance to herbicides |
| US11479786B2 (en) | 2017-11-29 | 2022-10-25 | Basf Se | Plants having increased tolerance to herbicides |
| US12123010B2 (en) | 2017-11-29 | 2024-10-22 | Basf Se | Plants having increased tolerance to herbicides |
| WO2019121352A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Herbicidal pyrimidine compounds |
| WO2019121373A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Herbicidal pyrimidine compounds |
| WO2019121408A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Herbicidal pyrimidine compounds |
| WO2019121374A1 (en) | 2017-12-20 | 2019-06-27 | Basf Se | Herbicidal pyrimidine compounds |
| US11939587B2 (en) | 2018-01-17 | 2024-03-26 | Basf Se | Plants having increased tolerance to herbicides |
| WO2019158378A1 (en) | 2018-02-16 | 2019-08-22 | Basf Se | Herbicidal mixtures |
| WO2019175713A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New catechol molecules and their use as inhibitors to p450 related metabolic pathways |
| WO2019175712A1 (en) | 2018-03-14 | 2019-09-19 | Basf Corporation | New uses for catechol molecules as inhibitors to glutathione s-transferase metabolic pathways |
| WO2019238427A1 (en) | 2018-06-13 | 2019-12-19 | Basf Se | Crystalline form of ethyl 2-[2-[2-chloro-4-fluoro-5-[3-methyl-2,6-dioxo-4-(trifluoromethyl)pyrimidin-1-yl]phenoxy]phenoxy]acetate |
| WO2019243358A1 (en) | 2018-06-19 | 2019-12-26 | Syngenta Crop Protection Ag | Herbicidal compounds |
| WO2020058009A1 (en) | 2018-09-18 | 2020-03-26 | Basf Se | Diaminotriazine compounds |
| US12459904B2 (en) | 2019-04-16 | 2025-11-04 | Bristol-Myers Squibb Company | Process for preparing carbamoyloxymethyl triazole cyclohexyl acid compounds |
| WO2021013969A1 (en) | 2019-07-25 | 2021-01-28 | Syngenta Crop Protection Ag | 3-[(hydrazono)methyl]-n-(tetrazol-5-yl)-benzamide and 3-[(hydrazono)methyl]-n-(1,3,4-oxadiazol-2-yl)-benzamide derivatives as herbicides |
| CN112969697A (en) * | 2019-10-31 | 2021-06-15 | 组合化学工业株式会社 | Process for producing herbicide and intermediate thereof |
| EP4053125A4 (en) * | 2019-10-31 | 2023-07-05 | Kumiai Chemical Industry Co., Ltd. | HERBICIDE AND METHOD OF MANUFACTURE OF AN INTERMEDIATE THEREOF |
| EP4053125B1 (en) | 2019-10-31 | 2024-12-04 | Kumiai Chemical Industry Co., Ltd. | Process for the production of a pyroxasulfone herbicide |
| KR102523028B1 (en) | 2019-10-31 | 2023-04-17 | 구미아이 가가쿠 고교 가부시키가이샤 | Method for manufacturing herbicides and intermediates thereof |
| WO2021002484A3 (en) * | 2019-10-31 | 2021-03-11 | クミアイ化学工業株式会社 | Herbicide and production method for intermediate thereof |
| JPWO2021002484A1 (en) * | 2019-10-31 | 2021-09-13 | クミアイ化学工業株式会社 | Method for manufacturing herbicides and their intermediates |
| KR20220097436A (en) * | 2019-10-31 | 2022-07-07 | 구미아이 가가쿠 고교 가부시키가이샤 | Method for producing herbicides and intermediates thereof |
| IL292532B1 (en) * | 2019-10-31 | 2024-04-01 | Kumiai Chemical Industry Co | Process for the production of herbicide and its intermediates |
| WO2021148302A1 (en) | 2020-01-23 | 2021-07-29 | Basf Se | Novel additives for agrochemical formulations |
| WO2021148303A1 (en) | 2020-01-23 | 2021-07-29 | Basf Se | Glufosinate formulations containing amines or ammonium salts |
| WO2021170464A1 (en) | 2020-02-28 | 2021-09-02 | Basf Se | Herbicidal malonamides |
| WO2021191035A1 (en) | 2020-03-25 | 2021-09-30 | Basf Se | Dioxazolines and their use as herbicides |
| WO2021209268A1 (en) | 2020-04-14 | 2021-10-21 | Basf Se | Beta-lactams and their use as herbicides |
| US12275742B2 (en) | 2020-05-05 | 2025-04-15 | Nuvalent, Inc. | Heteroaromatic macrocyclic ether chemotherapeutic agents |
| US12054498B2 (en) | 2020-05-05 | 2024-08-06 | Nuvalent, Inc. | Heteroaromatic macrocyclic ether chemotherapeutic agents |
| WO2021224040A1 (en) | 2020-05-05 | 2021-11-11 | Basf Se | Condensed isoxazoline derivatives and their use as herbicides |
| CN113754602A (en) * | 2020-06-02 | 2021-12-07 | 山东润博生物科技有限公司 | Synthesis method of 5, 5-dimethyl-4, 5-dihydroisoxazole-3-ketone |
| CN113754602B (en) * | 2020-06-02 | 2023-04-25 | 山东润博生物科技有限公司 | Synthesis method of 5, 5-dimethyl-4, 5-dihydro-isoxazole-3-one |
| WO2022008285A1 (en) | 2020-07-07 | 2022-01-13 | BASF Agro B.V. | Method for controlling undesirable vegetation in an aquatic environment |
| EP4177252A3 (en) * | 2020-07-10 | 2023-08-09 | Adama Agan Ltd. | Process and intermediates for the preparation of pyroxasulfone, fenoxasulfone and various sulfone analogs of 5,5-dimethyl-4h-1,2-oxazole |
| WO2022112351A1 (en) | 2020-11-25 | 2022-06-02 | Basf Se | Herbicidal malonamides |
| WO2022112347A1 (en) | 2020-11-25 | 2022-06-02 | Basf Se | Herbicidal malonamides |
| EP4008185A1 (en) | 2020-12-03 | 2022-06-08 | BASF Agro B.V. | Herbicidal composition comprising a saflufenacil microparticles |
| EP4011205A1 (en) | 2020-12-08 | 2022-06-15 | Basf Se | Microparticle compositions comprising trifludimoxazin |
| WO2022161801A1 (en) | 2021-01-27 | 2022-08-04 | Basf Se | Diaminotriazine compounds |
| WO2022161802A1 (en) | 2021-01-27 | 2022-08-04 | Basf Se | Diaminotriazine compounds |
| EP4049749A1 (en) | 2021-02-25 | 2022-08-31 | Basf Se | New microcapsules containing active substances |
| WO2022179911A1 (en) | 2021-02-25 | 2022-09-01 | Basf Se | New microcapsules containing active substances |
| WO2022189190A1 (en) | 2021-03-09 | 2022-09-15 | Basf Se | Malonamides and their use as herbicides |
| WO2022189208A1 (en) | 2021-03-10 | 2022-09-15 | Basf Se | New microparticles containing active substances |
| EP4056038A1 (en) | 2021-03-10 | 2022-09-14 | Basf Se | Microparticles containing active substances |
| WO2022268563A1 (en) | 2021-06-23 | 2022-12-29 | Basf Se | Herbicidal arylcyclopentene carboxamides |
| WO2022268564A1 (en) | 2021-06-23 | 2022-12-29 | Basf Se | Herbicidal aryldihydrofurane carboxylates |
| WO2023025855A1 (en) | 2021-08-25 | 2023-03-02 | Basf Se | Herbicidal malonamides |
| WO2023025854A1 (en) | 2021-08-25 | 2023-03-02 | Basf Se | Herbicidal malonamides |
| WO2023030934A1 (en) | 2021-08-31 | 2023-03-09 | Basf Se | Herbicidal composition comprising phenyluracils |
| WO2023031199A1 (en) | 2021-08-31 | 2023-03-09 | Basf Se | Herbicidal malonamides containing monocyclic heteroaromatic rings |
| WO2023031200A1 (en) | 2021-08-31 | 2023-03-09 | Basf Se | Herbicidal malonamides containing a condensed ring system |
| WO2023031161A1 (en) | 2021-09-03 | 2023-03-09 | BASF Agricultural Solutions Seed US LLC | Plants having increased tolerance to herbicides |
| WO2023036636A1 (en) | 2021-09-09 | 2023-03-16 | Basf Se | New microparticles containing active substances |
| US12043626B2 (en) | 2021-10-01 | 2024-07-23 | Nuvalent, Inc. | Solid forms, pharmaceutical compositions and preparation of heteroaromatic macrocyclic ether compounds |
| WO2023110491A1 (en) | 2021-12-16 | 2023-06-22 | BASF Agro B.V. | Method for controlling undesirable vegetation in an aquatic environment |
| WO2023208447A1 (en) | 2022-04-25 | 2023-11-02 | Basf Se | An emulsifiable concentrate having a (substituted) benzaldehyde-based solvent system |
| WO2023227676A1 (en) | 2022-05-25 | 2023-11-30 | Basf Se | Herbicidal malonic acid monoamides and malonamide esters |
| WO2023247489A1 (en) | 2022-06-21 | 2023-12-28 | Astrazeneca Ab | N-(2-(4-cyanothiazolidin-3-yl)-2-oxoethyl)- quinoline-4-carboxamides |
| WO2024012905A1 (en) | 2022-07-13 | 2024-01-18 | Basf Se | Herbicidal composition comprising azine compounds |
| WO2024056517A1 (en) | 2022-09-14 | 2024-03-21 | Basf Se | Use of an alkylether sulfate for improving the efficacy of herbicides |
| EP4338592A1 (en) | 2022-09-15 | 2024-03-20 | Basf Se | Use of compound for improving the efficacy of herbicides |
| EP4617275A1 (en) | 2022-11-09 | 2025-09-17 | Qingdao KingAgroot Chemical Compound Co., Ltd. | Oxabicycloalkane compounds, preparation method therefor, and herbicidal composition and use thereof |
| WO2024126113A1 (en) | 2022-12-12 | 2024-06-20 | BASF Agricultural Solutions Seed US LLC | Plants having increased tolerance to herbicides |
| WO2025114052A1 (en) | 2023-11-29 | 2025-06-05 | Basf Se | Ternary herbicidal composition comprising azine compounds |
| WO2025153657A2 (en) | 2024-01-17 | 2025-07-24 | Basf Se | Plants having increased tolerance to herbicides |
| WO2025153595A1 (en) | 2024-01-17 | 2025-07-24 | Basf Se | Plants having increased tolerance to herbicides |
| WO2025242699A1 (en) | 2024-05-22 | 2025-11-27 | Basf Se | Method for improving rainfastness of an agrochemical active ingredient |
| WO2026003150A1 (en) | 2024-06-28 | 2026-01-02 | Basf Se | Plants with mutated tubulin polypeptide having increased tolerance to herbicides |
| WO2026021913A1 (en) | 2024-07-23 | 2026-01-29 | Basf Se | Herbicidal 2-phenoxybutanoylamides |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20080097238A (en) | 2008-11-04 |
| ES2363254T3 (en) | 2011-07-28 |
| IL193468A0 (en) | 2009-05-04 |
| EP1991542A1 (en) | 2008-11-19 |
| AR059516A1 (en) | 2008-04-09 |
| UA92210C2 (en) | 2010-10-11 |
| UY30177A1 (en) | 2007-09-28 |
| CN101389625A (en) | 2009-03-18 |
| EA200870299A1 (en) | 2009-02-27 |
| PL1991542T3 (en) | 2011-08-31 |
| BRPI0707973A2 (en) | 2011-05-17 |
| EP1991542B1 (en) | 2011-03-16 |
| ZA200806687B (en) | 2009-07-29 |
| JP2009536149A (en) | 2009-10-08 |
| CA2640962A1 (en) | 2007-08-30 |
| US20100004128A1 (en) | 2010-01-07 |
| AP2008004580A0 (en) | 2008-08-31 |
| DE602007013185D1 (en) | 2011-04-28 |
| GB0603891D0 (en) | 2006-04-05 |
| ATE502033T1 (en) | 2011-04-15 |
| AU2007217252A1 (en) | 2007-08-30 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1991542B1 (en) | Herbicidal isoxazoline compounds | |
| US7465805B2 (en) | Isoxazoline derivatives and their use as herbicides | |
| WO2008074991A1 (en) | Novel herbicides | |
| US20090042726A1 (en) | Novel Herbicides | |
| CA2577495C (en) | Isoxazoline derivatives and their use as herbicides | |
| US20090048112A1 (en) | Novel Herbicides | |
| US8133849B2 (en) | Herbicidal compounds | |
| BR112017005883B1 (en) | HERBICIDAL COMPOUND, HERBICIDAL COMPOSITION, METHOD FOR CONTROLLING THE GROWTH OF UNWANTED PLANTS AND METHOD OF SELECTIVE WEED CONTROL | |
| US20160168126A1 (en) | Chemical compounds | |
| US20170318810A1 (en) | Herbicidal Compounds |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application | ||
| WWE | Wipo information: entry into national phase |
Ref document number: 2007217252 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2640962 Country of ref document: CA Ref document number: 570174 Country of ref document: NZ |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2007704963 Country of ref document: EP |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 6758/DELNP/2008 Country of ref document: IN |
|
| WWE | Wipo information: entry into national phase |
Ref document number: MX/a/2008/010384 Country of ref document: MX |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 193468 Country of ref document: IL |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 08086983 Country of ref document: CO |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 2008555855 Country of ref document: JP Ref document number: 2008081428 Country of ref document: EG |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200780006740.4 Country of ref document: CN |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| WWP | Wipo information: published in national office |
Ref document number: 2007217252 Country of ref document: AU |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 1020087023443 Country of ref document: KR |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 200870299 Country of ref document: EA |
|
| WWE | Wipo information: entry into national phase |
Ref document number: 12280877 Country of ref document: US |
|
| ENP | Entry into the national phase |
Ref document number: PI0707973 Country of ref document: BR Kind code of ref document: A2 Effective date: 20080819 |