US20150247123A1 - Generation of pancreatic endoderm from Pluripotent Stem cells using small molecules - Google Patents
Generation of pancreatic endoderm from Pluripotent Stem cells using small molecules Download PDFInfo
- Publication number
- US20150247123A1 US20150247123A1 US14/425,136 US201314425136A US2015247123A1 US 20150247123 A1 US20150247123 A1 US 20150247123A1 US 201314425136 A US201314425136 A US 201314425136A US 2015247123 A1 US2015247123 A1 US 2015247123A1
- Authority
- US
- United States
- Prior art keywords
- cells
- pancreatic
- inhibitor
- pancreatic cell
- cell precursors
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 150000003384 small molecules Chemical class 0.000 title abstract description 10
- 210000001900 endoderm Anatomy 0.000 title description 24
- 210000001778 pluripotent stem cell Anatomy 0.000 title description 12
- 210000004027 cell Anatomy 0.000 claims abstract description 286
- 239000002243 precursor Substances 0.000 claims abstract description 74
- 238000000034 method Methods 0.000 claims abstract description 58
- 101000578254 Homo sapiens Homeobox protein Nkx-6.1 Proteins 0.000 claims abstract description 45
- 102100041030 Pancreas/duodenum homeobox protein 1 Human genes 0.000 claims abstract description 45
- 101000578258 Homo sapiens Homeobox protein Nkx-6.2 Proteins 0.000 claims abstract description 44
- 101710183548 Pyridoxal 5'-phosphate synthase subunit PdxS Proteins 0.000 claims abstract description 44
- 210000004039 endoderm cell Anatomy 0.000 claims abstract description 24
- 102100028096 Homeobox protein Nkx-6.2 Human genes 0.000 claims abstract 3
- 150000001875 compounds Chemical class 0.000 claims description 61
- CDOVNWNANFFLFJ-UHFFFAOYSA-N 4-[6-[4-(1-piperazinyl)phenyl]-3-pyrazolo[1,5-a]pyrimidinyl]quinoline Chemical group C1CNCCN1C1=CC=C(C2=CN3N=CC(=C3N=C2)C=2C3=CC=CC=C3N=CC=2)C=C1 CDOVNWNANFFLFJ-UHFFFAOYSA-N 0.000 claims description 45
- 108090000379 Fibroblast growth factor 2 Proteins 0.000 claims description 43
- 229940043355 kinase inhibitor Drugs 0.000 claims description 37
- 239000003757 phosphotransferase inhibitor Substances 0.000 claims description 37
- 102100024785 Fibroblast growth factor 2 Human genes 0.000 claims description 35
- 229940118135 JNK inhibitor Drugs 0.000 claims description 35
- 239000012825 JNK inhibitor Substances 0.000 claims description 35
- 239000003112 inhibitor Substances 0.000 claims description 34
- 229940096885 Retinoic acid receptor agonist Drugs 0.000 claims description 14
- ACPOUJIDANTYHO-UHFFFAOYSA-N anthra[1,9-cd]pyrazol-6(2H)-one Chemical compound C1=CC(C(=O)C=2C3=CC=CC=2)=C2C3=NNC2=C1 ACPOUJIDANTYHO-UHFFFAOYSA-N 0.000 claims description 12
- 102100022086 GRB2-related adapter protein 2 Human genes 0.000 claims description 8
- 101710181935 Phosphate-binding protein PstS 1 Proteins 0.000 claims description 8
- 102000013814 Wnt Human genes 0.000 claims description 8
- 108050003627 Wnt Proteins 0.000 claims description 8
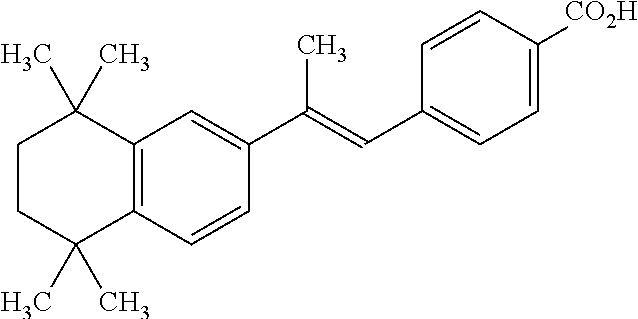
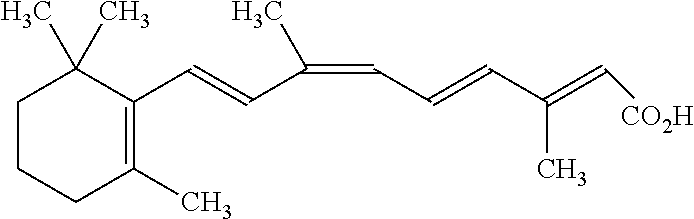
- SHGAZHPCJJPHSC-YCNIQYBTSA-N retinoic acid group Chemical group C\C(=C/C(=O)O)\C=C\C=C(\C=C\C1=C(CCCC1(C)C)C)/C SHGAZHPCJJPHSC-YCNIQYBTSA-N 0.000 claims description 7
- 241000289669 Erinaceus europaeus Species 0.000 claims description 6
- 102100028071 Fibroblast growth factor 7 Human genes 0.000 claims description 6
- 101001060261 Homo sapiens Fibroblast growth factor 7 Proteins 0.000 claims description 6
- 101000950695 Homo sapiens Mitogen-activated protein kinase 8 Proteins 0.000 claims description 6
- 102000043136 MAP kinase family Human genes 0.000 claims description 6
- 108091054455 MAP kinase family Proteins 0.000 claims description 6
- 102100037808 Mitogen-activated protein kinase 8 Human genes 0.000 claims description 6
- 108010053099 Vascular Endothelial Growth Factor Receptor-2 Proteins 0.000 claims description 6
- 101000628949 Homo sapiens Mitogen-activated protein kinase 10 Proteins 0.000 claims description 5
- 101000950669 Homo sapiens Mitogen-activated protein kinase 9 Proteins 0.000 claims description 5
- 102100026931 Mitogen-activated protein kinase 10 Human genes 0.000 claims description 5
- 102100037809 Mitogen-activated protein kinase 9 Human genes 0.000 claims description 5
- 238000007126 N-alkylation reaction Methods 0.000 claims description 5
- 102100028412 Fibroblast growth factor 10 Human genes 0.000 claims description 4
- 101000917237 Homo sapiens Fibroblast growth factor 10 Proteins 0.000 claims description 4
- 102100033177 Vascular endothelial growth factor receptor 2 Human genes 0.000 claims description 4
- 101100220616 Caenorhabditis elegans chk-2 gene Proteins 0.000 claims description 3
- 102000002254 Glycogen Synthase Kinase 3 Human genes 0.000 claims description 3
- 108010014905 Glycogen Synthase Kinase 3 Proteins 0.000 claims description 3
- 102000018967 Platelet-Derived Growth Factor beta Receptor Human genes 0.000 claims description 3
- 108010051742 Platelet-Derived Growth Factor beta Receptor Proteins 0.000 claims description 3
- 102000016549 Vascular Endothelial Growth Factor Receptor-2 Human genes 0.000 claims description 3
- 102100033178 Vascular endothelial growth factor receptor 1 Human genes 0.000 claims description 3
- 102000000568 rho-Associated Kinases Human genes 0.000 claims description 3
- 108010041788 rho-Associated Kinases Proteins 0.000 claims description 3
- 101000851018 Homo sapiens Vascular endothelial growth factor receptor 1 Proteins 0.000 claims description 2
- 102000004232 Mitogen-Activated Protein Kinase Kinases Human genes 0.000 claims description 2
- 108090000744 Mitogen-Activated Protein Kinase Kinases Proteins 0.000 claims description 2
- -1 Syc Proteins 0.000 claims description 2
- 102100033179 Vascular endothelial growth factor receptor 3 Human genes 0.000 claims description 2
- 101000851007 Homo sapiens Vascular endothelial growth factor receptor 2 Proteins 0.000 claims 1
- 101000851030 Homo sapiens Vascular endothelial growth factor receptor 3 Proteins 0.000 claims 1
- 101150001535 SRC gene Proteins 0.000 claims 1
- 230000002401 inhibitory effect Effects 0.000 claims 1
- 102100028098 Homeobox protein Nkx-6.1 Human genes 0.000 description 42
- 210000000130 stem cell Anatomy 0.000 description 29
- 230000004069 differentiation Effects 0.000 description 21
- 102000003702 retinoic acid receptors Human genes 0.000 description 15
- 108090000064 retinoic acid receptors Proteins 0.000 description 15
- QASFUMOKHFSJGL-LAFRSMQTSA-N Cyclopamine Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H](CC2=C3C)[C@@H]1[C@@H]2CC[C@@]13O[C@@H]2C[C@H](C)CN[C@H]2[C@H]1C QASFUMOKHFSJGL-LAFRSMQTSA-N 0.000 description 14
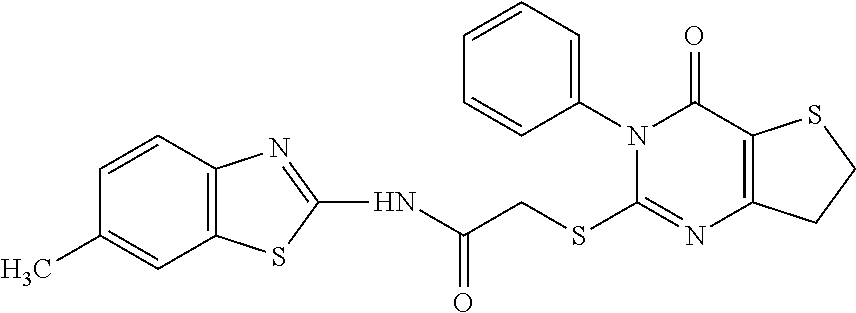
- WRKPZSMRWPJJDH-UHFFFAOYSA-N N-(6-methyl-1,3-benzothiazol-2-yl)-2-[(4-oxo-3-phenyl-6,7-dihydrothieno[3,2-d]pyrimidin-2-yl)thio]acetamide Chemical compound S1C2=CC(C)=CC=C2N=C1NC(=O)CSC1=NC=2CCSC=2C(=O)N1C1=CC=CC=C1 WRKPZSMRWPJJDH-UHFFFAOYSA-N 0.000 description 14
- 238000013459 approach Methods 0.000 description 14
- NOESYZHRGYRDHS-UHFFFAOYSA-N insulin Chemical compound N1C(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(NC(=O)CN)C(C)CC)CSSCC(C(NC(CO)C(=O)NC(CC(C)C)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CCC(N)=O)C(=O)NC(CC(C)C)C(=O)NC(CCC(O)=O)C(=O)NC(CC(N)=O)C(=O)NC(CC=2C=CC(O)=CC=2)C(=O)NC(CSSCC(NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2C=CC(O)=CC=2)NC(=O)C(CC(C)C)NC(=O)C(C)NC(=O)C(CCC(O)=O)NC(=O)C(C(C)C)NC(=O)C(CC(C)C)NC(=O)C(CC=2NC=NC=2)NC(=O)C(CO)NC(=O)CNC2=O)C(=O)NCC(=O)NC(CCC(O)=O)C(=O)NC(CCCNC(N)=N)C(=O)NCC(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC=CC=3)C(=O)NC(CC=3C=CC(O)=CC=3)C(=O)NC(C(C)O)C(=O)N3C(CCC3)C(=O)NC(CCCCN)C(=O)NC(C)C(O)=O)C(=O)NC(CC(N)=O)C(O)=O)=O)NC(=O)C(C(C)CC)NC(=O)C(CO)NC(=O)C(C(C)O)NC(=O)C1CSSCC2NC(=O)C(CC(C)C)NC(=O)C(NC(=O)C(CCC(N)=O)NC(=O)C(CC(N)=O)NC(=O)C(NC(=O)C(N)CC=1C=CC=CC=1)C(C)C)CC1=CN=CN1 NOESYZHRGYRDHS-UHFFFAOYSA-N 0.000 description 14
- 238000012216 screening Methods 0.000 description 14
- QASFUMOKHFSJGL-UHFFFAOYSA-N cyclopamine Natural products C1C=C2CC(O)CCC2(C)C(CC2=C3C)C1C2CCC13OC2CC(C)CNC2C1C QASFUMOKHFSJGL-UHFFFAOYSA-N 0.000 description 13
- 239000012980 RPMI-1640 medium Substances 0.000 description 10
- 210000000227 basophil cell of anterior lobe of hypophysis Anatomy 0.000 description 10
- 230000000975 bioactive effect Effects 0.000 description 10
- 210000004263 induced pluripotent stem cell Anatomy 0.000 description 9
- 102000003974 Fibroblast growth factor 2 Human genes 0.000 description 8
- 238000000338 in vitro Methods 0.000 description 8
- 150000002632 lipids Chemical class 0.000 description 8
- 102000004877 Insulin Human genes 0.000 description 7
- 108090001061 Insulin Proteins 0.000 description 7
- 239000007640 basal medium Substances 0.000 description 7
- 230000000694 effects Effects 0.000 description 7
- 229940125396 insulin Drugs 0.000 description 7
- 229940044601 receptor agonist Drugs 0.000 description 7
- 239000000018 receptor agonist Substances 0.000 description 7
- 241000607479 Yersinia pestis Species 0.000 description 6
- 210000002459 blastocyst Anatomy 0.000 description 6
- 210000001671 embryonic stem cell Anatomy 0.000 description 6
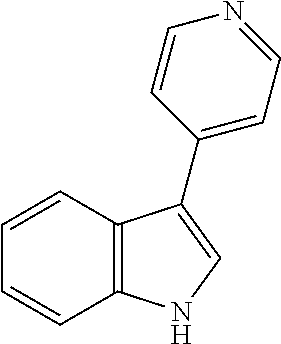
- LLJRXVHJOJRCSM-UHFFFAOYSA-N 3-pyridin-4-yl-1H-indole Chemical compound C=1NC2=CC=CC=C2C=1C1=CC=NC=C1 LLJRXVHJOJRCSM-UHFFFAOYSA-N 0.000 description 5
- 230000008859 change Effects 0.000 description 5
- 230000004186 co-expression Effects 0.000 description 5
- 239000002609 medium Substances 0.000 description 5
- 210000001519 tissue Anatomy 0.000 description 5
- YOELZIQOLWZLQC-UHFFFAOYSA-N 6-(4-fluorophenyl)-5-pyridin-4-yl-2,3-dihydroimidazo[2,1-b]thiazole Chemical compound C1=CC(F)=CC=C1C1=C(C=2C=CN=CC=2)N2CCSC2=N1 YOELZIQOLWZLQC-UHFFFAOYSA-N 0.000 description 4
- 108010023082 activin A Proteins 0.000 description 4
- 210000003890 endocrine cell Anatomy 0.000 description 4
- 210000001654 germ layer Anatomy 0.000 description 4
- ODZGYELAMAOARP-UHFFFAOYSA-N n1-methyl-1,9-pyrazoloanthrone Chemical compound C12=CC=CC=C2C(=O)C2=CC=CC3=C2C1=NN3C ODZGYELAMAOARP-UHFFFAOYSA-N 0.000 description 4
- 210000001082 somatic cell Anatomy 0.000 description 4
- FWBHETKCLVMNFS-UHFFFAOYSA-N 4',6-Diamino-2-phenylindol Chemical compound C1=CC(C(=N)N)=CC=C1C1=CC2=CC=C(C(N)=N)C=C2N1 FWBHETKCLVMNFS-UHFFFAOYSA-N 0.000 description 3
- 102000040945 Transcription factor Human genes 0.000 description 3
- 108091023040 Transcription factor Proteins 0.000 description 3
- 229920004890 Triton X-100 Polymers 0.000 description 3
- 239000013504 Triton X-100 Substances 0.000 description 3
- 210000003981 ectoderm Anatomy 0.000 description 3
- 210000002308 embryonic cell Anatomy 0.000 description 3
- 230000002124 endocrine Effects 0.000 description 3
- 238000007429 general method Methods 0.000 description 3
- 210000003958 hematopoietic stem cell Anatomy 0.000 description 3
- 210000003716 mesoderm Anatomy 0.000 description 3
- 230000003287 optical effect Effects 0.000 description 3
- 230000009996 pancreatic endocrine effect Effects 0.000 description 3
- 229930002330 retinoic acid Natural products 0.000 description 3
- 229960001727 tretinoin Drugs 0.000 description 3
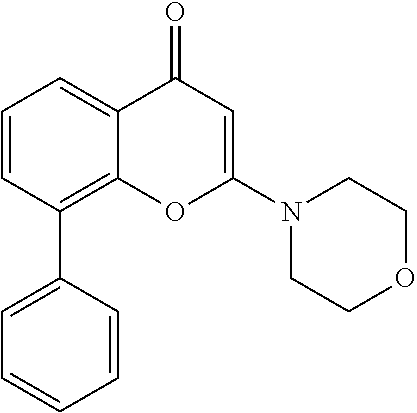
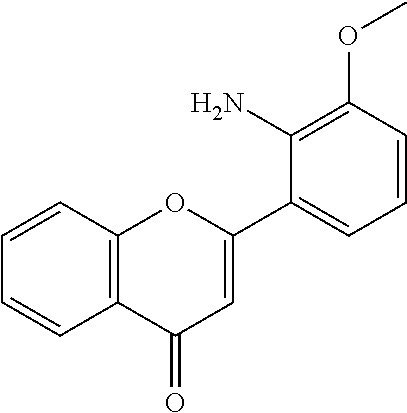
- QFWCYNPOPKQOKV-UHFFFAOYSA-N 2-(2-amino-3-methoxyphenyl)chromen-4-one Chemical compound COC1=CC=CC(C=2OC3=CC=CC=C3C(=O)C=2)=C1N QFWCYNPOPKQOKV-UHFFFAOYSA-N 0.000 description 2
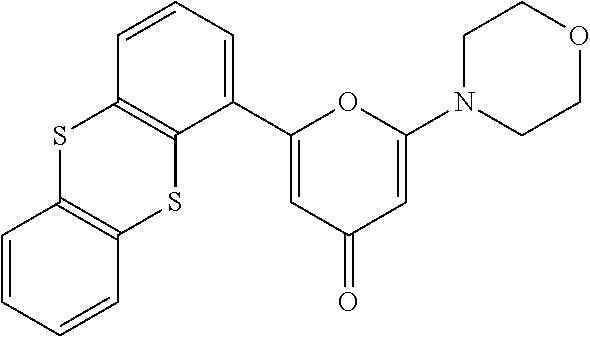
- XRKYMMUGXMWDAO-UHFFFAOYSA-N 2-(4-morpholinyl)-6-(1-thianthrenyl)-4-pyranone Chemical compound O1C(C=2C=3SC4=CC=CC=C4SC=3C=CC=2)=CC(=O)C=C1N1CCOCC1 XRKYMMUGXMWDAO-UHFFFAOYSA-N 0.000 description 2
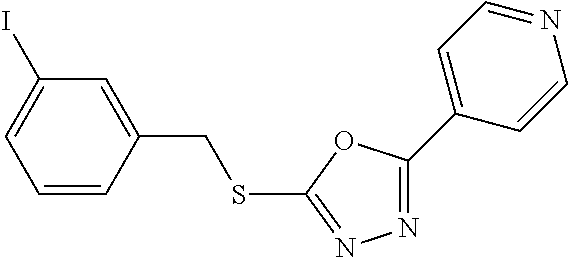
- ZRHRPGSSSVYBRG-UHFFFAOYSA-N 2-[(3-iodophenyl)methylthio]-5-pyridin-4-yl-1,3,4-oxadiazole Chemical compound IC1=CC=CC(CSC=2OC(=NN=2)C=2C=CN=CC=2)=C1 ZRHRPGSSSVYBRG-UHFFFAOYSA-N 0.000 description 2
- UXGJAOIJSROTTN-UHFFFAOYSA-N 2-[4-(4-chlorophenoxy)phenyl]-3h-benzimidazole-5-carboxamide Chemical compound N1C2=CC(C(=O)N)=CC=C2N=C1C(C=C1)=CC=C1OC1=CC=C(Cl)C=C1 UXGJAOIJSROTTN-UHFFFAOYSA-N 0.000 description 2
- BHWCZLOXTLWZAQ-UHFFFAOYSA-N 2-methyl-4-phenyl-5-pyridin-4-yloxazole Chemical compound O1C(C)=NC(C=2C=CC=CC=2)=C1C1=CC=NC=C1 BHWCZLOXTLWZAQ-UHFFFAOYSA-N 0.000 description 2
- SHGAZHPCJJPHSC-ZVCIMWCZSA-N 9-cis-retinoic acid Chemical compound OC(=O)/C=C(\C)/C=C/C=C(/C)\C=C\C1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-ZVCIMWCZSA-N 0.000 description 2
- VLMIVRCMWFXYTM-UHFFFAOYSA-N CC1(C)CCC(C)(C)C2=C1C=CC(C(=O)NC1=CC=C(OC=O)C=C1)=C2 Chemical compound CC1(C)CCC(C)(C)C2=C1C=CC(C(=O)NC1=CC=C(OC=O)C=C1)=C2 VLMIVRCMWFXYTM-UHFFFAOYSA-N 0.000 description 2
- WKYYLDKREKPNKZ-XQSIOCNLSA-N CC1=C(/C=C/C(C)=C/C=C/C(C)=C\OC=O)C(C)(C)CCC1 Chemical compound CC1=C(/C=C/C(C)=C/C=C/C(C)=C\OC=O)C(C)(C)CCC1 WKYYLDKREKPNKZ-XQSIOCNLSA-N 0.000 description 2
- WKYYLDKREKPNKZ-RQIJYFHRSA-N CC1=C(/C=C/C(C)=C\C=C\C(C)=C\OC=O)C(C)(C)CCC1 Chemical compound CC1=C(/C=C/C(C)=C\C=C\C(C)=C\OC=O)C(C)(C)CCC1 WKYYLDKREKPNKZ-RQIJYFHRSA-N 0.000 description 2
- 241000283074 Equus asinus Species 0.000 description 2
- 241000027355 Ferocactus setispinus Species 0.000 description 2
- SHGAZHPCJJPHSC-NUEINMDLSA-N Isotretinoin Chemical compound OC(=O)C=C(C)/C=C/C=C(C)C=CC1=C(C)CCCC1(C)C SHGAZHPCJJPHSC-NUEINMDLSA-N 0.000 description 2
- CZQHHVNHHHRRDU-UHFFFAOYSA-N LY294002 Chemical compound C1=CC=C2C(=O)C=C(N3CCOCC3)OC2=C1C1=CC=CC=C1 CZQHHVNHHHRRDU-UHFFFAOYSA-N 0.000 description 2
- 229960001445 alitretinoin Drugs 0.000 description 2
- 230000015572 biosynthetic process Effects 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- 230000018109 developmental process Effects 0.000 description 2
- 210000002257 embryonic structure Anatomy 0.000 description 2
- 238000002474 experimental method Methods 0.000 description 2
- 238000009472 formulation Methods 0.000 description 2
- 239000001963 growth medium Substances 0.000 description 2
- 238000003384 imaging method Methods 0.000 description 2
- 229960005280 isotretinoin Drugs 0.000 description 2
- 238000004519 manufacturing process Methods 0.000 description 2
- 239000000203 mixture Substances 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 239000013642 negative control Substances 0.000 description 2
- 238000010899 nucleation Methods 0.000 description 2
- 230000015031 pancreas development Effects 0.000 description 2
- 239000013641 positive control Substances 0.000 description 2
- 230000019491 signal transduction Effects 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- 239000000126 substance Substances 0.000 description 2
- 230000001360 synchronised effect Effects 0.000 description 2
- 238000002054 transplantation Methods 0.000 description 2
- IDDDVXIUIXWAGJ-DDSAHXNVSA-N 4-[(1r)-1-aminoethyl]-n-pyridin-4-ylcyclohexane-1-carboxamide;dihydrochloride Chemical compound Cl.Cl.C1CC([C@H](N)C)CCC1C(=O)NC1=CC=NC=C1 IDDDVXIUIXWAGJ-DDSAHXNVSA-N 0.000 description 1
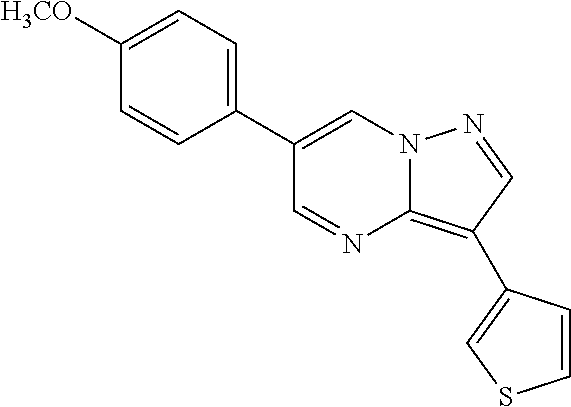
- HBWLNACPIFKNIP-UHFFFAOYSA-N 6-(4-methoxyphenyl)-3-(3-thiophenyl)pyrazolo[1,5-a]pyrimidine Chemical compound C1=CC(OC)=CC=C1C1=CN2N=CC(C3=CSC=C3)=C2N=C1 HBWLNACPIFKNIP-UHFFFAOYSA-N 0.000 description 1
- HFDKKNHCYWNNNQ-YOGANYHLSA-N 75976-10-2 Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(N)=O)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H]1N(CCC1)C(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(O)=O)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@@H](NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](C)N)C(C)C)[C@@H](C)O)C1=CC=C(O)C=C1 HFDKKNHCYWNNNQ-YOGANYHLSA-N 0.000 description 1
- 229940122485 ATM kinase inhibitor Drugs 0.000 description 1
- 102100034111 Activin receptor type-1 Human genes 0.000 description 1
- 239000012103 Alexa Fluor 488 Substances 0.000 description 1
- 239000012110 Alexa Fluor 594 Substances 0.000 description 1
- 210000002237 B-cell of pancreatic islet Anatomy 0.000 description 1
- 102100025423 Bone morphogenetic protein receptor type-1A Human genes 0.000 description 1
- 102100027052 Bone morphogenetic protein receptor type-1B Human genes 0.000 description 1
- JMZLVEYCFJVVCD-SAPNQHFASA-N C/C(=C\C1=CC=C(OC=O)C=C1)C1=CC2=C(C=C1)C(C)(C)CCC2(C)C Chemical compound C/C(=C\C1=CC=C(OC=O)C=C1)C1=CC2=C(C=C1)C(C)(C)CCC2(C)C JMZLVEYCFJVVCD-SAPNQHFASA-N 0.000 description 1
- IDMGVRDNZFQORW-SIKBANFGSA-N CC1(C)C(/C=C/C(/C)=C\C=C\C(\C)=C\C)=C(C)CCC1 Chemical compound CC1(C)C(/C=C/C(/C)=C\C=C\C(\C)=C\C)=C(C)CCC1 IDMGVRDNZFQORW-SIKBANFGSA-N 0.000 description 1
- KHMVFFRIDRPJDO-YCNIQYBTSA-N CC1=C(/C=C/C(C)=C/C=C/C(C)=C/OC(=O)I)C(C)(C)CCC1 Chemical compound CC1=C(/C=C/C(C)=C/C=C/C(C)=C/OC(=O)I)C(C)(C)CCC1 KHMVFFRIDRPJDO-YCNIQYBTSA-N 0.000 description 1
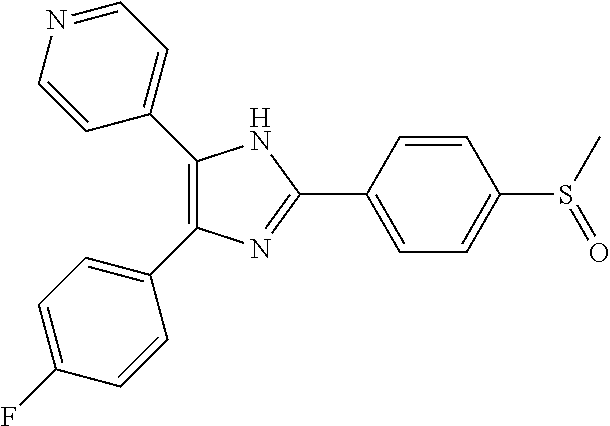
- BVEYUHATIMQURK-UHFFFAOYSA-N CS(=O)C1=CC=C(C2=NC(C3=CC=C(F)C=C3)=C(C3=CC=NC=C3)C2)C=C1 Chemical compound CS(=O)C1=CC=C(C2=NC(C3=CC=C(F)C=C3)=C(C3=CC=NC=C3)C2)C=C1 BVEYUHATIMQURK-UHFFFAOYSA-N 0.000 description 1
- AQGNHMOJWBZFQQ-UHFFFAOYSA-N CT 99021 Chemical compound CC1=CNC(C=2C(=NC(NCCNC=3N=CC(=CC=3)C#N)=NC=2)C=2C(=CC(Cl)=CC=2)Cl)=N1 AQGNHMOJWBZFQQ-UHFFFAOYSA-N 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 101800003838 Epidermal growth factor Proteins 0.000 description 1
- 102400001368 Epidermal growth factor Human genes 0.000 description 1
- 241001481760 Erethizon dorsatum Species 0.000 description 1
- 108010011459 Exenatide Proteins 0.000 description 1
- 102000018233 Fibroblast Growth Factor Human genes 0.000 description 1
- 108050007372 Fibroblast Growth Factor Proteins 0.000 description 1
- 102000003972 Fibroblast growth factor 7 Human genes 0.000 description 1
- 108090000385 Fibroblast growth factor 7 Proteins 0.000 description 1
- 101800001586 Ghrelin Proteins 0.000 description 1
- 102400000442 Ghrelin-28 Human genes 0.000 description 1
- 102400000321 Glucagon Human genes 0.000 description 1
- 108060003199 Glucagon Proteins 0.000 description 1
- 102400000322 Glucagon-like peptide 1 Human genes 0.000 description 1
- 101800000224 Glucagon-like peptide 1 Proteins 0.000 description 1
- DTHNMHAUYICORS-KTKZVXAJSA-N Glucagon-like peptide 1 Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](C)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCCN)C(=O)NCC(=O)N[C@@H](CCCNC(N)=N)C(N)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](C)NC(=O)[C@H](C)NC(=O)[C@H](CCC(N)=O)NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CC=1N=CNC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 DTHNMHAUYICORS-KTKZVXAJSA-N 0.000 description 1
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 1
- 229940121827 Hedgehog pathway inhibitor Drugs 0.000 description 1
- 108090000100 Hepatocyte Growth Factor Proteins 0.000 description 1
- 102100021866 Hepatocyte growth factor Human genes 0.000 description 1
- 102100029284 Hepatocyte nuclear factor 3-beta Human genes 0.000 description 1
- 101000799140 Homo sapiens Activin receptor type-1 Proteins 0.000 description 1
- 101000934638 Homo sapiens Bone morphogenetic protein receptor type-1A Proteins 0.000 description 1
- 101000984546 Homo sapiens Bone morphogenetic protein receptor type-1B Proteins 0.000 description 1
- 101001062347 Homo sapiens Hepatocyte nuclear factor 3-beta Proteins 0.000 description 1
- 101000599951 Homo sapiens Insulin-like growth factor I Proteins 0.000 description 1
- 101000612089 Homo sapiens Pancreas/duodenum homeobox protein 1 Proteins 0.000 description 1
- 101000984042 Homo sapiens Protein lin-28 homolog A Proteins 0.000 description 1
- 101000652324 Homo sapiens Transcription factor SOX-17 Proteins 0.000 description 1
- 101000687905 Homo sapiens Transcription factor SOX-2 Proteins 0.000 description 1
- 102100037852 Insulin-like growth factor I Human genes 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 108010025020 Nerve Growth Factor Proteins 0.000 description 1
- 102000015336 Nerve Growth Factor Human genes 0.000 description 1
- 108020005497 Nuclear hormone receptor Proteins 0.000 description 1
- KFLWBZPSJQPRDD-ONEGZZNKSA-N O=[N+]([O-])/C=C/C1=CC2=C(C=C1)OCO2 Chemical compound O=[N+]([O-])/C=C/C1=CC2=C(C=C1)OCO2 KFLWBZPSJQPRDD-ONEGZZNKSA-N 0.000 description 1
- VRLLXSFPAAIUKN-UHFFFAOYSA-N OCC1=CC=C(C2=CN3N=CC(C4=CSC=C4)=C3N=C2)C=C1 Chemical compound OCC1=CC=C(C2=CN3N=CC(C4=CSC=C4)=C3N=C2)C=C1 VRLLXSFPAAIUKN-UHFFFAOYSA-N 0.000 description 1
- 102000018886 Pancreatic Polypeptide Human genes 0.000 description 1
- 229930040373 Paraformaldehyde Natural products 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- 229940122907 Phosphatase inhibitor Drugs 0.000 description 1
- 102000003993 Phosphatidylinositol 3-kinases Human genes 0.000 description 1
- 108090000430 Phosphatidylinositol 3-kinases Proteins 0.000 description 1
- 108091000080 Phosphotransferase Proteins 0.000 description 1
- 108010038512 Platelet-Derived Growth Factor Proteins 0.000 description 1
- 102000010780 Platelet-Derived Growth Factor Human genes 0.000 description 1
- 102100025460 Protein lin-28 homolog A Human genes 0.000 description 1
- 238000011579 SCID mouse model Methods 0.000 description 1
- 102000005157 Somatostatin Human genes 0.000 description 1
- 108010056088 Somatostatin Proteins 0.000 description 1
- 101000983124 Sus scrofa Pancreatic prohormone precursor Proteins 0.000 description 1
- 206010043276 Teratoma Diseases 0.000 description 1
- 102100030243 Transcription factor SOX-17 Human genes 0.000 description 1
- 102100024270 Transcription factor SOX-2 Human genes 0.000 description 1
- 206010067584 Type 1 diabetes mellitus Diseases 0.000 description 1
- 108010053096 Vascular Endothelial Growth Factor Receptor-1 Proteins 0.000 description 1
- 108010053100 Vascular Endothelial Growth Factor Receptor-3 Proteins 0.000 description 1
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 1
- 239000000556 agonist Substances 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 239000005557 antagonist Substances 0.000 description 1
- FOIVPCKZDPCJJY-JQIJEIRASA-N arotinoid acid Chemical compound C=1C=C(C(CCC2(C)C)(C)C)C2=CC=1C(/C)=C/C1=CC=C(C(O)=O)C=C1 FOIVPCKZDPCJJY-JQIJEIRASA-N 0.000 description 1
- 210000003050 axon Anatomy 0.000 description 1
- 210000001109 blastomere Anatomy 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000000601 blood cell Anatomy 0.000 description 1
- 230000011748 cell maturation Effects 0.000 description 1
- 230000008614 cellular interaction Effects 0.000 description 1
- JUFFVKRROAPVBI-PVOYSMBESA-N chembl1210015 Chemical compound C([C@@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(=O)N[C@H]1[C@@H]([C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O)[C@@H](O)[C@@H](CO[C@]3(O[C@@H](C[C@H](O)[C@H](O)CO)[C@H](NC(C)=O)[C@@H](O)C3)C(O)=O)O2)O)[C@@H](CO)O1)NC(C)=O)C(=O)NCC(=O)NCC(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](C)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CO)C(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@@H](NC(=O)[C@H](C)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@H](CCSC)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(O)=O)NC(=O)CNC(=O)[C@@H](N)CC=1NC=NC=1)[C@@H](C)O)[C@@H](C)O)C(C)C)C1=CC=CC=C1 JUFFVKRROAPVBI-PVOYSMBESA-N 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 230000002759 chromosomal effect Effects 0.000 description 1
- 238000011281 clinical therapy Methods 0.000 description 1
- 238000012790 confirmation Methods 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 230000006378 damage Effects 0.000 description 1
- 206010012601 diabetes mellitus Diseases 0.000 description 1
- 102000038379 digestive enzymes Human genes 0.000 description 1
- 108091007734 digestive enzymes Proteins 0.000 description 1
- 230000013020 embryo development Effects 0.000 description 1
- 210000002242 embryoid body Anatomy 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- 229940116977 epidermal growth factor Drugs 0.000 description 1
- 229960001519 exenatide Drugs 0.000 description 1
- 210000000646 extraembryonic cell Anatomy 0.000 description 1
- 229940126864 fibroblast growth factor Drugs 0.000 description 1
- 210000001647 gastrula Anatomy 0.000 description 1
- 230000014509 gene expression Effects 0.000 description 1
- GNKDKYIHGQKHHM-RJKLHVOGSA-N ghrelin Chemical compound C([C@H](NC(=O)[C@@H](NC(=O)[C@H](CO)NC(=O)CN)COC(=O)CCCCCCC)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC=1N=CNC=1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CCCCN)C(=O)N1[C@@H](CCC1)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N1[C@@H](CCC1)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C1=CC=CC=C1 GNKDKYIHGQKHHM-RJKLHVOGSA-N 0.000 description 1
- MASNOZXLGMXCHN-ZLPAWPGGSA-N glucagon Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCSC)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O)C(C)C)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](C)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CCCNC(N)=N)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CCCCN)NC(=O)[C@H](CO)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](NC(=O)[C@H](CC=1C=CC=CC=1)NC(=O)[C@@H](NC(=O)CNC(=O)[C@H](CCC(N)=O)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC=1NC=NC=1)[C@@H](C)O)[C@@H](C)O)C1=CC=CC=C1 MASNOZXLGMXCHN-ZLPAWPGGSA-N 0.000 description 1
- 229960004666 glucagon Drugs 0.000 description 1
- 239000008103 glucose Substances 0.000 description 1
- 229940088597 hormone Drugs 0.000 description 1
- 239000005556 hormone Substances 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 230000006698 induction Effects 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 239000010410 layer Substances 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- PSGAAPLEWMOORI-PEINSRQWSA-N medroxyprogesterone acetate Chemical compound C([C@@]12C)CC(=O)C=C1[C@@H](C)C[C@@H]1[C@@H]2CC[C@]2(C)[C@@](OC(C)=O)(C(C)=O)CC[C@H]21 PSGAAPLEWMOORI-PEINSRQWSA-N 0.000 description 1
- 230000003278 mimic effect Effects 0.000 description 1
- 238000004264 monolayer culture Methods 0.000 description 1
- 210000002894 multi-fate stem cell Anatomy 0.000 description 1
- 229940053128 nerve growth factor Drugs 0.000 description 1
- 102000006255 nuclear receptors Human genes 0.000 description 1
- 108020004017 nuclear receptors Proteins 0.000 description 1
- 210000000056 organ Anatomy 0.000 description 1
- 229920002866 paraformaldehyde Polymers 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- 102000020233 phosphotransferase Human genes 0.000 description 1
- 230000008569 process Effects 0.000 description 1
- GPTFURBXHJWNHR-UHFFFAOYSA-N protopine Chemical compound C1=C2C(=O)CC3=CC=C4OCOC4=C3CN(C)CCC2=CC2=C1OCO2 GPTFURBXHJWNHR-UHFFFAOYSA-N 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- NHXLMOGPVYXJNR-ATOGVRKGSA-N somatostatin Chemical compound C([C@H]1C(=O)N[C@H](C(N[C@@H](CO)C(=O)N[C@@H](CSSC[C@@H](C(=O)N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@@H](CC=2C=CC=CC=2)C(=O)N[C@@H](CC=2C3=CC=CC=C3NC=2)C(=O)N[C@@H](CCCCN)C(=O)N[C@H](C(=O)N1)[C@@H](C)O)NC(=O)CNC(=O)[C@H](C)N)C(O)=O)=O)[C@H](O)C)C1=CC=CC=C1 NHXLMOGPVYXJNR-ATOGVRKGSA-N 0.000 description 1
- 229960000553 somatostatin Drugs 0.000 description 1
- 230000000920 spermatogeneic effect Effects 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 210000002438 upper gastrointestinal tract Anatomy 0.000 description 1
- VBEQCZHXXJYVRD-GACYYNSASA-N uroanthelone Chemical compound C([C@@H](C(=O)N[C@H](C(=O)N[C@@H](CS)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CS)C(=O)N[C@H](C(=O)N[C@@H]([C@@H](C)CC)C(=O)NCC(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(=O)N[C@@H](CO)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CC=1C2=CC=CC=C2NC=1)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O)C(C)C)[C@@H](C)O)NC(=O)[C@H](CO)NC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CO)NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](NC(=O)[C@H](CC=1NC=NC=1)NC(=O)[C@H](CCSC)NC(=O)[C@H](CS)NC(=O)[C@@H](NC(=O)CNC(=O)CNC(=O)[C@H](CC(N)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@H](CS)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)CNC(=O)[C@H](CC(O)=O)NC(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@H](CO)NC(=O)[C@H]1N(CCC1)C(=O)[C@H](CS)NC(=O)CNC(=O)[C@H]1N(CCC1)C(=O)[C@H](CC=1C=CC(O)=CC=1)NC(=O)[C@H](CO)NC(=O)[C@@H](N)CC(N)=O)C(C)C)[C@@H](C)CC)C1=CC=C(O)C=C1 VBEQCZHXXJYVRD-GACYYNSASA-N 0.000 description 1
- 210000004291 uterus Anatomy 0.000 description 1
- 229940121396 wnt pathway inhibitor Drugs 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0676—Pancreatic cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0676—Pancreatic cells
- C12N5/0678—Stem cells; Progenitor cells; Precursor cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/115—Basic fibroblast growth factor (bFGF, FGF-2)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/117—Keratinocyte growth factors (KGF-1, i.e. FGF-7; KGF-2, i.e. FGF-12)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/119—Other fibroblast growth factors, e.g. FGF-4, FGF-8, FGF-10
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/155—Bone morphogenic proteins [BMP]; Osteogenins; Osteogenic factor; Bone inducing factor
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/10—Growth factors
- C12N2501/16—Activin; Inhibin; Mullerian inhibiting substance
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/30—Hormones
- C12N2501/38—Hormones with nuclear receptors
- C12N2501/385—Hormones with nuclear receptors of the family of the retinoic acid recptor, e.g. RAR, RXR; Peroxisome proliferator-activated receptor [PPAR]
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/40—Regulators of development
- C12N2501/41—Hedgehog proteins; Cyclopamine (inhibitor)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/40—Regulators of development
- C12N2501/415—Wnt; Frizzeled
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/60—Transcription factors
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/70—Enzymes
- C12N2501/72—Transferases [EC 2.]
- C12N2501/727—Kinases (EC 2.7.)
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2501/00—Active agents used in cell culture processes, e.g. differentation
- C12N2501/999—Small molecules not provided for elsewhere
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2503/00—Use of cells in diagnostics
- C12N2503/02—Drug screening
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/02—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from embryonic cells
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2506/00—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells
- C12N2506/45—Differentiation of animal cells from one lineage to another; Differentiation of pluripotent cells from artificially induced pluripotent stem cells
Definitions
- the present invention relates to methods of generating pancreatic endoderm from pluripotent stem (PS) cells, such as human definitive endoderm.
- PS pluripotent stem
- Beta cell transplantation potentially provides the ultimate cure for type I diabetes.
- the limited availability of donor beta cells constrains the use of this treatment as a clinical therapy.
- Pluripotent stem cells can proliferate infinitely and differentiate into many cell types; thus, PS cells are a promising source for beta cells.
- PS cells before PS cells can be used to treat diabetes, they need to be efficiently and reproducibly differentiated to pancreatic cells.
- a pluripotent cell gives rise to the three germ layers; ectoderm, mesoderm and endoderm.
- Induction of definitive endoderm (DE) is the first step towards formation of endoderm derived tissues.
- Generation of pancreatic endoderm (PE) from DE cells is necessary for the generation of insulin-producing beta cells.
- PE cells with the potential to become endocrine progenitors (EP) are characterized by co-expression of two important transcription factors, PDX1 and NKX6.1.
- Stepwise in vitro differentiation protocols have been established for generating pancreatic cells from PS cells. These protocols generally mimic the major events of pancreatic development, which includes several stages such as formation of the DE which co-expresses SOX17 and FOXA2, primitive gut, posterior foregut, PE, EP and ultimately the mature beta cells. To date, efficient DE differentiation of hES cells has been achieved by activin A treatment. The next major step in generating pancreatic beta cells is to generate PE that co-expresses PDX1 and NKX6.1.
- the present invention relates to a method of producing pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing definitive endoderm (DE) cells to an effective amount of at least one compound of the group consisting of:
- the present invention further relates to a method for producing pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing DE cells to an effective amount of at least one compound of the group consisting of:
- the present invention further relates to a method for producing pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing definitive endoderm cells to an effective amount of at least one compound of the group consisting of:
- the present invention further relates to a method for generating pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing definitive endoderm cells to an effective amount of the BMP inhibitor LDN-193189, to differentiate human DE cells into pancreatic or pancreatic cell precursors.
- the present invention further relates to a method for generating pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing DE cells to an effective amount of the BMP inhibitor LDN-193189, and subsequent exposure to one of the following molecules:
- the present invention further relates to a method for generating pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing DE cells to an effective amount of the BMP inhibitor LDN-193189, and subsequent exposure to a combination of JNK inhibitor II, retinoic acid or a retinoic acid derivative, bFGF and one of the following molecules:
- the present invention further relates to a method for generating pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing DE cells to an effective amount of the BMP inhibitor LDN-193189, and subsequent exposure to a combination of JNK inhibitor II in combination with retinoic acid or a retinoic acid derivative, bFGF and LDN-193189 to differentiate DE stem cells into pancreatic or pancreatic cell precursors.
- any one of the retinoic acid receptor agonists or kinase inhibitors may be in combination with bFGF.
- the present invention further relates to pancreatic cells or pancreatic cell precursors obtainable by the methods of the present invention.
- the present invention relates to a pancreatic cell or pancreatic cell precursor produced by exposing a human pluripotent stem cell to at least one compound listed in tables 1 and 2.
- the present invention relates to use of any one of the compounds of tables 1 and 2, to induce pancreatic cells or pancreatic cell precursors from stem cells.
- the present invention relates to use of LDN-193189 to induce pancreatic cells or pancreatic cell precursors from stem cells.
- the present invention relates to use of LDN-193189 followed by Cyclopamine or IWP2, to induce pancreatic cells or pancreatic cell precursors from stem cells.
- the present invention takes an alternative approach to improve the efficiency of differentiating human PS cells toward mature beta cells, by providing a method to increase the fraction of NKX6.1/PDX1 double positive cells, a hallmark for PE cells committed to an endocrine cell fate.
- the invention provides an improved pancreatic cell population, i.e. PE with increased fraction of NKX6.1/PDX1 double positive cells.
- the present invention provides a more homogenous pancreatic cell population, which is important for the further development of these cells towards the endocrine lineage.
- the present invention also provides a more synchronised pancreatic population to get to the next stage.
- the present invention may also solve further problems that will be apparent from the disclosure of the exemplary embodiments.
- FIG. 1 shows the PE screening approach—also referred to as the library screening approach—using small molecule libraries.
- Pluripotent stem (PS) cells were differentiated into definitive endoderm (DE) according to the DE protocol (see general methods) and seeded in 96 well plates for screening.
- the pancreatic endoderm (PE) screen was divided into an early and a late phase. In the early phase compounds were added on top of a published bFGF based protocol (Amen et al., 2010, cf. also WO/2010/136583) for the first seven days of PE differentiation and then continued for another six days without the compounds. In the late phase compounds were only added on top of the bFGF based protocol for the last six days.
- FIG. 2 shows early phase hits for the library screening approach.
- Definitive endoderm cells from human induced pluripotent stem cells (hiPSC) (black) or hESC (white) were seeded in 96 well optical plates and differentiated into pancreatic endoderm using a 14 day protocol based on bFGF.
- Compounds were added on top of the bFGF based protocol for the first seven out of 14 days and analysed for NKX6.1/PDX1 double positive cells using the InCell analyzer 2000 (GE Healthcare).
- the graph shows the % effect of the fraction of NKX6.1/PDX1 double positive cells compared to the Benchmark protocol.
- FIG. 3 shows late phase hits for the library screening approach.
- Definitive endoderm cells from hiPSC (black) or hESC (white) were seeded in 96 well optical plates and differentiated into pancreatic endoderm using a 14 day protocol based on bFGF.
- Compounds were added on top of the bFGF based protocol for the last six days and analysed for NKX6.1/PDX1 double positive cells using the InCell analyzer 2000 (GE Healthcare).
- the graph shows the % effect of the fraction of NKX6.1/PDX1 double positive cells compared to the Benchmark protocol.
- FIG. 4 shows a second, candidate based PE screening approach.
- Pluripotent stem (PS) cells were differentiated into definitive endoderm according to DE protocol (See general methods) and seeded in 96 well plates for screening.
- the pancreatic endoderm screen was divided into two parts. In screen 1, compounds were added to a basal medium (RPMI1640+0.1% PEST+12% KOSR) the first eight days of PE differentiation. Compounds were tested in 4 different time windows having 2 day increments and then cells were left to continue differentiation for another six days in the bFGF based published protocol (Amen et al., 2010). In screen 2, cells were first differentiated for 4 days with the hit compounds from screen 1, then screening compounds were added the last 10 days to basal medium.
- FIG. 5 shows hits from the candidate screen 1 and 2 compared to cells differentiated according to Amen et al, 2010 which was used as a benchmark protocol running in parallel with every screen.
- one hit compound was identified (LDN-193189) and was found to be most effective when added for the first 4 days followed by 4 days basal medium.
- two hit compounds were identified (Cyclopamine and IWP-2) when cells were first exposed to the hit compound from screen 1 for 4 days and hit compounds from screen 2 were added for the last 10 days of differentiation.
- the graph shows the % effect of the fraction of NKX6.1/PDX1 double positive cells compared to the Benchmark protocol (Bars for hiPSC in black and hESC in white).
- FIG. 6 shows the advantageous effect on the amount of PDX1/NKX6.1 double positive cells by the combination of hit compounds found in the two individual screens (small molecule libraries and candidate approach) compared to the benchmark protocol (Ameri et al. (2010)). Bars for hiPSC in black and hESC in white.
- FGF basic Fibroblast Growth Factor
- hBS human Blastocyst derived Stem hBSC; human Blastocyst-derived Stem Cells hES: human Embryonic Stem hESC: human Embryonic Stem Cells hiPSC: human induced Pluripotent Stem Cells hPSC: human Pluripotent Stem Cells
- NKX6.1 NK6 homeobox 1
- PDX1 Pancreatic and duodenal homeobox 1
- PS Pluripotent Stem
- the present invention related to methods of generating pancreatic endoderm from stem cells, such as human definitive endoderm cells and induced pluripotent stem cells.
- the present invention takes an alternative approach to improve the efficiency of differentiating human PS cells toward mature beta cells, by providing a method to improve the percentage of NKX6.1/PDX1 double positive cells, which are markers for a PE cell population, one of the cell stages necessary to reach endocrine cell populations. Furthermore, the present invention provides a more homogenous and synchronised pancreatic cell population, which is important for the further development of these cells towards the endocrine lineage.
- the present invention may also solve further problems that will be apparent from the disclosure of the exemplary embodiments.
- the pancreatic endocrine cells obtainable by the method according to the invention are insulin producing cells, optionally together with cells differentiated towards glucagon, somatostatin, pancreatic polypeptide, and/or ghrelin producing cells.
- insulin producing cells refers to cells that produce and store or secrete detectable amounts of insulin.
- Insulin producing cells can be individual cells or collections of cells.
- the cell population comprising pancreatic cells is obtained from a somatic cell population.
- the somatic cell population has been induced to de-differentiate into an embryonic-like stem (ES, e.g., a pluripotent) cell.
- ES embryonic-like stem
- iPSC induced pluripotent stem cells
- the cell population comprising pancreatic cells is obtained from embryonic stem (ES, e.g., pluripotent) cells.
- ES embryonic stem
- the cell population comprising pancreatic cells is pluripotent cells such as ES like-cells.
- the cell population comprising pancreatic cells is embryonic differentiated stem (ES or pluripotent) cells. Differentiation takes place in embryoid bodies and/or in monolayer cell cultures or a combination thereof.
- ES embryonic differentiated stem
- the cell population is a population of stem cells.
- the cell population is a population of stem cells differentiated to the pancreatic endocrine lineage.
- Stem cells are undifferentiated cells defined by their ability at the single cell level to both self-renew and differentiate to produce progeny cells, including self-renewing progenitors, non-renewing progenitors, and terminally differentiated cells. Stem cells are also characterized by their ability to differentiate in vitro into functional cells of various cell lineages from multiple germ layers (endoderm, mesoderm and ectoderm), as well as to give rise to tissues of multiple germ layers following transplantation and to contribute substantially to most, if not all, tissues following injection into blastocysts.
- Stem cells are classified by their developmental potential as: (1) totipotent, meaning able to give rise to all embryonic and extraembryonic cell types; (2) pluripotent, meaning able to give rise to all embryonic cell types; (3) multi-potent, meaning able to give rise to a subset of cell lineages, but all within a particular tissue, organ, or physiological system (for example, hematopoietic stem cells (HSC) can produce progeny that include HSC (self-renewal), blood cell restricted oligopotent progenitors and all cell types and elements (e.g., platelets) that are normal components of the blood); (4) oligopotent, meaning able to give rise to a more restricted subset of cell lineages than multi-potent stem cells; and (5) unipotent, meaning able to give rise to a single cell lineage (e.g., spermatogenic stem cells).
- HSC hematopoietic stem cells
- a protocol for obtaining pancreatic cells from stem cells is exemplified by, but not limited to, the protocols described in D'Amour, K. A. et al. (2006); Jiang, J. et al. (2007); Kroon, E. et al. (2008).
- a protocol for obtaining pancreatic cells from somatic cells or somatic cells induced to de-differentiate into pluripotent cells such as ES like-cells is exemplified by, but not limited to, the protocols described in Aoi, T. et al. (2008); D'Amour, K. A. et al. (2006); Jiang, J. et al. (2007); Kroon, E. et al. (2008); Takahashi, K. et al. (2007); Takahashi, K., and Yamanaka, S. (2006) and Wernig, M. et al. (2007).
- differentiate refers to a process where cells progress from an undifferentiated state to a differentiated state, from an immature state to a less immature state or from an immature state to a mature state.
- characteristics markers like PDX1, NKX6.1, and PTF1a.
- Mature or differentiated pancreatic cells do not proliferate and do secrete high levels of pancreatic endocrine hormones or digestive enzymes.
- fully differentiated beta cells secrete insulin at high levels in response to glucose. Changes in cell interaction and maturation occur as cells lose markers of undifferentiated cells or gain markers of differentiated cells.
- differentiation factor refers to a compound added to pancreatic cells to enhance their differentiation to mature endocrine cells also containing insulin producing beta cells.
- exemplary differentiation factors include hepatocyte growth factor, keratinocyte growth factor, exendin-4, basic fibroblast growth factor, insulin-like growth factor-1, nerve growth factor, epidermal growth factor platelet-derived growth factor, and glucagon-like peptide 1.
- differentiation of the cells comprises culturing the cells in a medium comprising one or more differentiation factors.
- human pluripotent stem cells refers to cells that may be derived from any source and that are capable, under appropriate conditions, of producing human progeny of different cell types that are derivatives of all of the 3 germinal layers (endoderm, mesoderm, and ectoderm). hPSC may have the ability to form a teratoma in 8-12 week old SCID mice and/or the ability to form identifiable cells of all three germ layers in tissue culture. Included in the definition of human pluripotent stem cells are embryonic cells of various types including human blastocyst derived stem (hBS) cells in 30 literature often denoted as human embryonic stem (hES) cells, (see, e.g., Thomson et al.
- hBS human blastocyst derived stem
- hES human embryonic stem
- hPSC human induced pluripotent stem
- hiPSC human induced pluripotent stem cells
- ES cell lines can also be derived from single blastomeres without the destruction of ex utero embryos and without affecting the clinical outcome (Chung et al. (2006) and Klimanskaya et al. (2006)).
- the term “blastocyst-derived stem cell” is denoted BS cell, and the human form is termed “hBS cells”.
- the pluripotent stem cells used in the present invention can thus be embryonic stem cells prepared from blastocysts, as described in e.g. WO 03/055992 and WO 2007/042225, or be commercially available hBS cells or cell lines.
- any human pluripotent stem cell can be used in the present invention, including differentiated adult cells which are reprogrammed to pluripotent cells by e.g. the treating adult cells with certain transcription factors, such as OCT4, SOX2, NANOG, and LIN28 as disclosed in Yu, et al. (2007); Takahashi et al. (2007) and Yu et al. (2009).
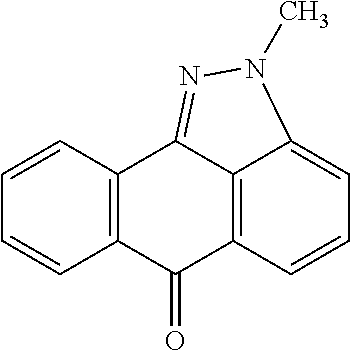
- JNK inhibitor II includes isomers or tautomers of 1,9-pyrazoloanthrone with or without N-alkylation.
- DEF medium or DEF-CS medium/system is a defined balanced culture medium for the establishment and propagation of human pluripotent stem cells, DEF-CS medium/system.
- hES cells line SA121 and human induced pluripotent stem cells (hiPSC) chIPS4 (Cellectis) were grown in DEF-CS culture media (Cellectis) in T75 culture flasks.
- Cells were single cell passaged with 5 ⁇ M Rock inhibitor Y-27632 (Sigma #Y0503) and seeded at a density of 40000 cells/cm2 for experiments.
- Cells were cultured at 37° C. and 5% CO 2 in a humidified incubator (ThermoScientific Model 371).
- hES cells line SA121 and hiPSC (chIPS4) were washed once in RPMI1640 (Gibco #61870) and treated with 3 ⁇ M CHIR99021 (Axon#1386) in RPMI1640. After 24 hours the cells were washed with RPMI1640 and treated with 100 ng/ml Activin A (Peprotech #120-14E) in RPMI1640. 24 hours later, 2% B27 (Invitrogen #17504-044) was added to the Activin A media for 2 days with daily media change. Cells were maintained at 37° C. and 5% CO 2 in a humidified incubator during the differentiation.
- DAPI 4,6-diamidino-2-phenylindole, Applichem, A4099.0010
- secondary antibodies Alexa Fluor 488 donkey anti-goat and Alexa Fluor 594 donkey anti-mouse (both Invitrogen) were diluted 1:1000 in 0.1% Triton X-100 in PBS and added to each well for 45 min. Cells were washed five times and left in 200 ⁇ L PBS for imaging.
- Imaging was performed using the InCell Analyzer 2000 (GE Healthcare). 4 fields per well with 10 ⁇ objective were captured. The total cell number based in the DAPI counterstaining and the number of NKX6.1/PDX1 double positive cells was determined using InCell Developer Toolbox 1.8 (GE Healthcare). The fraction of NKX6.1/PDX1 double positive cells was normalized to the benchmark on each plate and the % effect was calculated. Values above 200% effect were categorized as hits.
- Pancreatic endoderm is characterized by co-expression of two transcription factors, NKX6.1 and PDX1. Many of the published protocols for making PE are ineffective with low outcome of NKX6.1/PDX1 double positive cells. Enhancing the efficacy of the PE protocols is a desirable outcome. We therefore screened libraries of small molecules to identify novel compounds that would improve the existing PE protocols. This was done on the assumption that inhibitors, agonists or antagonists may regulate signaling pathways, or chromosomal accessibility, which would improve the fraction of NKX6.1/PDX1 double positive cells.
- kinase inhibitor library (Calbiochem #539743), a bioactive lipid library (Enzo Life Sciences #BML-2800), a nuclear receptor ligand library (Enzo Life Sciences # BML-2802) and a phosphatase inhibitor library (Enzo Life Sciences #BML-2834).
- the compounds within the bioactive library were tested at 1 uM and 0.1 uM.
- Compounds from the other libraries were tested at 10 uM and 1 uM.
- small molecules that target the main signalling pathways involved in pancreas development were included.
- NKX6.1/PDX1 Screen The library compounds were screened on top of a bFGF based media formulation for making PE (Ameri et al. 2010) (RPMI1640, Gibco#61870; 12% KOSR, Gibco#10828; 0.1% PEST, Gibco#15140; 64 ng/mL bFGF, Peprotech #100-18B).
- the library PE screening approach was divided into an early and a late phase ( FIG. 1 ).
- DE cells were differentiated in the PE media for the first seven days. In the following six days compounds were tested on top of the PE media. 12 positive control wells (PE media) and 12 negative control wells (PE media without bFGF) were included in each 96 well plate. Media change was performed daily. Hits identified in the early phase screen are illustrated in FIG. 2 and listed in table 1. Hits identified in the late phase screen are illustrated in FIG. 3 and listed in table 2.
- the compounds from the candidate approach were screened in basal medium (RPMI1640, Gibco#61870; 12% KOSR, Gibco#10828; 0.1% PEST, Gibco#15140) without the addition of bFGF.
- This candidate approach screen was divided into two parts ( FIG. 4 ). In the first part, compounds were tested in time intervals with 2 day increments for the first eight days of PE differentiation (2 days exposure to compounds followed by 6 days basal medium or 4 days exposure to compounds followed by 4 days basal medium or 6 days exposure to compounds followed by 2 days basal medium or 8 days exposure to compounds).
- DE cells were differentiated according to the hit compounds from the first part, the following 6-10 days compounds were tested in basal media.
- Hits identified in the candidate screening approach are illustrated in FIG. 5 and also contained in Tables 1 and 2.
- DE cells were exposed to 4 days 50 nM LDN-193189, followed by 8 days AM580 (AH Diagnostics, BML GF104 0025), JNK Inhibitor II (Calbiochem, 420119), 50 nM LDN-193189 and 64 ng/ml FGF2, or AM580, JNK Inhibitor II, 50 nM LDN-193189, 64 ng/ml FGF2 and IWP2, or AM580, JNK Inhibitor II, 50 nM LDN-193189, 64 ng/ml FGF2 and Cyclopamine ( FIG. 6 ). Media change was performed daily.
- Hit compounds were screened on top of a bFGF based media formulation for making PE (Amen et al. 2010) (RPMI1640, Gibco#61870; 12% KOSR, Gibco#10828; 0.1% PEST, Gibco#15140; 64 ng/mL bFGF, Peprotech #100-18B).
- the screen was divided into an early and a late phase ( FIG. 1 ).
- the early phase compounds were tested on top of the PE media for the first seven days of PE differentiation, and then continued for additional six days using PE media without compounds.
- the late phase DE cells were differentiated in the PE media for the first seven days. In the following six days compounds were tested on top of the PE media. Twelve positive control wells (PE media) and 12 negative control wells (PE media without bFGF) were included in each 96 well plate. Media change was performed daily.
Landscapes
- Engineering & Computer Science (AREA)
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Biomedical Technology (AREA)
- Chemical & Material Sciences (AREA)
- Biotechnology (AREA)
- Organic Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Genetics & Genomics (AREA)
- Wood Science & Technology (AREA)
- Zoology (AREA)
- Microbiology (AREA)
- Biochemistry (AREA)
- General Engineering & Computer Science (AREA)
- General Health & Medical Sciences (AREA)
- Cell Biology (AREA)
- Developmental Biology & Embryology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
Abstract
Description
- The present invention relates to methods of generating pancreatic endoderm from pluripotent stem (PS) cells, such as human definitive endoderm.
- Beta cell transplantation potentially provides the ultimate cure for type I diabetes. However, the limited availability of donor beta cells constrains the use of this treatment as a clinical therapy. Pluripotent stem cells can proliferate infinitely and differentiate into many cell types; thus, PS cells are a promising source for beta cells. However, before PS cells can be used to treat diabetes, they need to be efficiently and reproducibly differentiated to pancreatic cells.
- During vertebrate embryonic development, a pluripotent cell gives rise to the three germ layers; ectoderm, mesoderm and endoderm. Induction of definitive endoderm (DE) is the first step towards formation of endoderm derived tissues. Generation of pancreatic endoderm (PE) from DE cells is necessary for the generation of insulin-producing beta cells. PE cells with the potential to become endocrine progenitors (EP) are characterized by co-expression of two important transcription factors, PDX1 and NKX6.1.
- Stepwise in vitro differentiation protocols have been established for generating pancreatic cells from PS cells. These protocols generally mimic the major events of pancreatic development, which includes several stages such as formation of the DE which co-expresses SOX17 and FOXA2, primitive gut, posterior foregut, PE, EP and ultimately the mature beta cells. To date, efficient DE differentiation of hES cells has been achieved by activin A treatment. The next major step in generating pancreatic beta cells is to generate PE that co-expresses PDX1 and NKX6.1. Several groups have developed in vitro protocols that can differentiate PS cells into DE and PE, however they are only able to generate a modest fraction of NKX6.1/PDX1 double positive (db+ve) cells, and importantly none of them are able to generate fully mature beta cells in vitro (Cai et al. (2010); D'Amour et al. (2006); Kunisada et al. (2012); Schulz et al. (2012); Zhang et al. (2009); Amen et al. (2010)).
- The present invention relates to a method of producing pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing definitive endoderm (DE) cells to an effective amount of at least one compound of the group consisting of:
-
- a. BMP inhibitor LDN-193189 (listed in table 1)
- b. Kinase inhibitors (listed in tables 1 and 2)
- c. Retinoic acid receptor agonists (listed in table 2)
to differentiate the human DE cells into the pancreatic cells or pancreatic cell precursors.
- The present invention further relates to a method for producing pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing DE cells to an effective amount of at least one compound of the group consisting of:
-
- a. BMP inhibitor LDN-193189 (listed in table 1)
- b. isomers of 1,9-pyrazoloanthrone with or without N-alkylation (listed in table 1 and 2)
- c. Retinoic acid receptor agonists (listed in table 2)
to differentiate the human DE cells into the pancreatic or pancreatic cell precursors.
- The present invention further relates to a method for producing pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing definitive endoderm cells to an effective amount of at least one compound of the group consisting of:
-
- a. BMP inhibitor LDN-193189
- b. JNK inhibitor II
- c. AM580
to differentiate the human DE cells into the pancreatic or pancreatic cell precursors.
- The present invention further relates to a method for generating pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing definitive endoderm cells to an effective amount of the BMP inhibitor LDN-193189, to differentiate human DE cells into pancreatic or pancreatic cell precursors. The present invention further relates to a method for generating pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing DE cells to an effective amount of the BMP inhibitor LDN-193189, and subsequent exposure to one of the following molecules:
-
- a. Wnt inhibitor IWP2
- b. Hedgehog inhibitor Cyclopamine (Cyc)
to differentiate human DE cells into pancreatic or pancreatic cell precursors.
- The present invention further relates to a method for generating pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing DE cells to an effective amount of the BMP inhibitor LDN-193189, and subsequent exposure to a combination of JNK inhibitor II, retinoic acid or a retinoic acid derivative, bFGF and one of the following molecules:
-
- a. Wnt inhibitor IWP2
- b. Hedgehog inhibitor Cyclopamine
to differentiate human DE cells into pancreatic or pancreatic cell precursors.
- The present invention further relates to a method for generating pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing DE cells to an effective amount of the BMP inhibitor LDN-193189, and subsequent exposure to a combination of JNK inhibitor II in combination with retinoic acid or a retinoic acid derivative, bFGF and LDN-193189 to differentiate DE stem cells into pancreatic or pancreatic cell precursors.
- In one embodiment of the present invention, any one of the retinoic acid receptor agonists or kinase inhibitors may be in combination with bFGF.
- The present invention further relates to pancreatic cells or pancreatic cell precursors obtainable by the methods of the present invention.
- The present invention relates to a pancreatic cell or pancreatic cell precursor produced by exposing a human pluripotent stem cell to at least one compound listed in tables 1 and 2.
- The present invention relates to use of any one of the compounds of tables 1 and 2, to induce pancreatic cells or pancreatic cell precursors from stem cells.
- The present invention relates to use of LDN-193189 to induce pancreatic cells or pancreatic cell precursors from stem cells.
- The present invention relates to use of LDN-193189 followed by Cyclopamine or IWP2, to induce pancreatic cells or pancreatic cell precursors from stem cells.
- The present invention takes an alternative approach to improve the efficiency of differentiating human PS cells toward mature beta cells, by providing a method to increase the fraction of NKX6.1/PDX1 double positive cells, a hallmark for PE cells committed to an endocrine cell fate.
- In one aspect, the invention provides an improved pancreatic cell population, i.e. PE with increased fraction of NKX6.1/PDX1 double positive cells.
- Furthermore, the present invention provides a more homogenous pancreatic cell population, which is important for the further development of these cells towards the endocrine lineage.
- The present invention also provides a more synchronised pancreatic population to get to the next stage.
- The present invention may also solve further problems that will be apparent from the disclosure of the exemplary embodiments.
-
FIG. 1 shows the PE screening approach—also referred to as the library screening approach—using small molecule libraries. Pluripotent stem (PS) cells were differentiated into definitive endoderm (DE) according to the DE protocol (see general methods) and seeded in 96 well plates for screening. The pancreatic endoderm (PE) screen was divided into an early and a late phase. In the early phase compounds were added on top of a published bFGF based protocol (Amen et al., 2010, cf. also WO/2010/136583) for the first seven days of PE differentiation and then continued for another six days without the compounds. In the late phase compounds were only added on top of the bFGF based protocol for the last six days. -
FIG. 2 shows early phase hits for the library screening approach. Definitive endoderm cells from human induced pluripotent stem cells (hiPSC) (black) or hESC (white) were seeded in 96 well optical plates and differentiated into pancreatic endoderm using a 14 day protocol based on bFGF. Compounds were added on top of the bFGF based protocol for the first seven out of 14 days and analysed for NKX6.1/PDX1 double positive cells using the InCell analyzer 2000 (GE Healthcare). The graph shows the % effect of the fraction of NKX6.1/PDX1 double positive cells compared to the Benchmark protocol. -
FIG. 3 shows late phase hits for the library screening approach. Definitive endoderm cells from hiPSC (black) or hESC (white) were seeded in 96 well optical plates and differentiated into pancreatic endoderm using a 14 day protocol based on bFGF. Compounds were added on top of the bFGF based protocol for the last six days and analysed for NKX6.1/PDX1 double positive cells using the InCell analyzer 2000 (GE Healthcare). The graph shows the % effect of the fraction of NKX6.1/PDX1 double positive cells compared to the Benchmark protocol. -
FIG. 4 shows a second, candidate based PE screening approach. Pluripotent stem (PS) cells were differentiated into definitive endoderm according to DE protocol (See general methods) and seeded in 96 well plates for screening. The pancreatic endoderm screen was divided into two parts. Inscreen 1, compounds were added to a basal medium (RPMI1640+0.1% PEST+12% KOSR) the first eight days of PE differentiation. Compounds were tested in 4 different time windows having 2 day increments and then cells were left to continue differentiation for another six days in the bFGF based published protocol (Amen et al., 2010). Inscreen 2, cells were first differentiated for 4 days with the hit compounds fromscreen 1, then screening compounds were added the last 10 days to basal medium. -
FIG. 5 shows hits from thecandidate screen screen 1, one hit compound was identified (LDN-193189) and was found to be most effective when added for the first 4 days followed by 4 days basal medium. Forscreen 2, two hit compounds were identified (Cyclopamine and IWP-2) when cells were first exposed to the hit compound fromscreen 1 for 4 days and hit compounds fromscreen 2 were added for the last 10 days of differentiation. The graph shows the % effect of the fraction of NKX6.1/PDX1 double positive cells compared to the Benchmark protocol (Bars for hiPSC in black and hESC in white). -
FIG. 6 shows the advantageous effect on the amount of PDX1/NKX6.1 double positive cells by the combination of hit compounds found in the two individual screens (small molecule libraries and candidate approach) compared to the benchmark protocol (Ameri et al. (2010)). Bars for hiPSC in black and hESC in white. - +ve: positive
bFGF: basic Fibroblast Growth Factor (FGF) (also known as FGF2) - db: double positive
- hBS: human Blastocyst derived Stem
hBSC; human Blastocyst-derived Stem Cells
hES: human Embryonic Stem
hESC: human Embryonic Stem Cells
hiPSC: human induced Pluripotent Stem Cells
hPSC: human Pluripotent Stem Cells - NKX6.1:
NK6 homeobox 1
PDX1: Pancreatic andduodenal homeobox 1 - The present invention related to methods of generating pancreatic endoderm from stem cells, such as human definitive endoderm cells and induced pluripotent stem cells.
- The present invention takes an alternative approach to improve the efficiency of differentiating human PS cells toward mature beta cells, by providing a method to improve the percentage of NKX6.1/PDX1 double positive cells, which are markers for a PE cell population, one of the cell stages necessary to reach endocrine cell populations. Furthermore, the present invention provides a more homogenous and synchronised pancreatic cell population, which is important for the further development of these cells towards the endocrine lineage.
- The present invention may also solve further problems that will be apparent from the disclosure of the exemplary embodiments.
- In one embodiment, the pancreatic endocrine cells obtainable by the method according to the invention are insulin producing cells, optionally together with cells differentiated towards glucagon, somatostatin, pancreatic polypeptide, and/or ghrelin producing cells. As used herein, “insulin producing cells” refers to cells that produce and store or secrete detectable amounts of insulin. “Insulin producing cells” can be individual cells or collections of cells.
- In another embodiment, the cell population comprising pancreatic cells is obtained from a somatic cell population. In some aspects the somatic cell population has been induced to de-differentiate into an embryonic-like stem (ES, e.g., a pluripotent) cell. Such de-differentiated cells are also termed induced pluripotent stem cells (iPSC).
- In another embodiment, the cell population comprising pancreatic cells is obtained from embryonic stem (ES, e.g., pluripotent) cells. In some aspects the cell population comprising pancreatic cells is pluripotent cells such as ES like-cells.
- In another embodiment, the cell population comprising pancreatic cells is embryonic differentiated stem (ES or pluripotent) cells. Differentiation takes place in embryoid bodies and/or in monolayer cell cultures or a combination thereof.
- In another embodiment, the cell population is a population of stem cells. In some aspects the cell population is a population of stem cells differentiated to the pancreatic endocrine lineage.
- Stem cells are undifferentiated cells defined by their ability at the single cell level to both self-renew and differentiate to produce progeny cells, including self-renewing progenitors, non-renewing progenitors, and terminally differentiated cells. Stem cells are also characterized by their ability to differentiate in vitro into functional cells of various cell lineages from multiple germ layers (endoderm, mesoderm and ectoderm), as well as to give rise to tissues of multiple germ layers following transplantation and to contribute substantially to most, if not all, tissues following injection into blastocysts.
- Stem cells are classified by their developmental potential as: (1) totipotent, meaning able to give rise to all embryonic and extraembryonic cell types; (2) pluripotent, meaning able to give rise to all embryonic cell types; (3) multi-potent, meaning able to give rise to a subset of cell lineages, but all within a particular tissue, organ, or physiological system (for example, hematopoietic stem cells (HSC) can produce progeny that include HSC (self-renewal), blood cell restricted oligopotent progenitors and all cell types and elements (e.g., platelets) that are normal components of the blood); (4) oligopotent, meaning able to give rise to a more restricted subset of cell lineages than multi-potent stem cells; and (5) unipotent, meaning able to give rise to a single cell lineage (e.g., spermatogenic stem cells).
- A protocol for obtaining pancreatic cells from stem cells is exemplified by, but not limited to, the protocols described in D'Amour, K. A. et al. (2006); Jiang, J. et al. (2007); Kroon, E. et al. (2008).
- A protocol for obtaining pancreatic cells from somatic cells or somatic cells induced to de-differentiate into pluripotent cells such as ES like-cells is exemplified by, but not limited to, the protocols described in Aoi, T. et al. (2008); D'Amour, K. A. et al. (2006); Jiang, J. et al. (2007); Kroon, E. et al. (2008); Takahashi, K. et al. (2007); Takahashi, K., and Yamanaka, S. (2006) and Wernig, M. et al. (2007).
- As used herein “differentiate” or “differentiation” refers to a process where cells progress from an undifferentiated state to a differentiated state, from an immature state to a less immature state or from an immature state to a mature state. For example, early undifferentiated embryonic pancreatic cells are able to proliferate and express characteristics markers, like PDX1, NKX6.1, and PTF1a. Mature or differentiated pancreatic cells do not proliferate and do secrete high levels of pancreatic endocrine hormones or digestive enzymes. E.g., fully differentiated beta cells secrete insulin at high levels in response to glucose. Changes in cell interaction and maturation occur as cells lose markers of undifferentiated cells or gain markers of differentiated cells. Loss or gain of a single marker can indicate that a cell has “matured or fully differentiated.” The term “differentiation factor” refers to a compound added to pancreatic cells to enhance their differentiation to mature endocrine cells also containing insulin producing beta cells. Exemplary differentiation factors include hepatocyte growth factor, keratinocyte growth factor, exendin-4, basic fibroblast growth factor, insulin-like growth factor-1, nerve growth factor, epidermal growth factor platelet-derived growth factor, and glucagon-
like peptide 1. In some aspects differentiation of the cells comprises culturing the cells in a medium comprising one or more differentiation factors. - As used herein, “human pluripotent stem cells” (hPSC) refers to cells that may be derived from any source and that are capable, under appropriate conditions, of producing human progeny of different cell types that are derivatives of all of the 3 germinal layers (endoderm, mesoderm, and ectoderm). hPSC may have the ability to form a teratoma in 8-12 week old SCID mice and/or the ability to form identifiable cells of all three germ layers in tissue culture. Included in the definition of human pluripotent stem cells are embryonic cells of various types including human blastocyst derived stem (hBS) cells in 30 literature often denoted as human embryonic stem (hES) cells, (see, e.g., Thomson et al. (1998), Heins et al. (2004), as well as induced pluripotent stem cells (see, e.g. Yu et al. (2007); Takahashi et al. (2007)). The various methods and other embodiments described herein may require or utilise hPSC from a variety of sources. For example, hPSC suitable for use may be obtained from developing embryos. Additionally or alternatively, suitable hPSC may be obtained from established cell lines and/or human induced pluripotent stem (hiPS) cells.
- As used herein “hiPSC” refers to human induced pluripotent stem cells.
- ES cell lines can also be derived from single blastomeres without the destruction of ex utero embryos and without affecting the clinical outcome (Chung et al. (2006) and Klimanskaya et al. (2006)).
- As used herein, the term “blastocyst-derived stem cell” is denoted BS cell, and the human form is termed “hBS cells”. In literature the cells are often referred to as embryonic stem cells, and more specifically human embryonic stem cells (hESC). The pluripotent stem cells used in the present invention can thus be embryonic stem cells prepared from blastocysts, as described in e.g. WO 03/055992 and WO 2007/042225, or be commercially available hBS cells or cell lines. However, it is further envisaged that any human pluripotent stem cell can be used in the present invention, including differentiated adult cells which are reprogrammed to pluripotent cells by e.g. the treating adult cells with certain transcription factors, such as OCT4, SOX2, NANOG, and LIN28 as disclosed in Yu, et al. (2007); Takahashi et al. (2007) and Yu et al. (2009).
- As used herein JNK inhibitor II includes isomers or tautomers of 1,9-pyrazoloanthrone with or without N-alkylation. Where 1,9-pyrazoloanthrone can be defined as “SMILES: c1ccc2c(c1)-c3c4c(cccc4[nH]n3)C2=O” or “1,6-dihydrodibenzo[cd,g]indazol-6-one”.
- DEF medium or DEF-CS medium/system is a defined balanced culture medium for the establishment and propagation of human pluripotent stem cells, DEF-CS medium/system.
-
- 1. A method of producing pancreatic cells or pancreatic cell precursors where at least 5% of the cells co-express PDX1 and NKX6.1, comprising exposing embryonic stem cells to an effective amount of at least one compound of the group consisting of:
- a. BMP inhibitors
- b. kinase inhibitors
- c. retinoic acid receptor agonists
- to differentiate the human embryonic stem cells into the pancreatic cells or pancreatic cell precursors.
- 2. The method of
embodiment 1, wherein said compounds are listed in tables 1 or 2. - 3. The method of
embodiments - 4. The method of embodiments 1-3, wherein said kinase inhibitor is JNK inhibitor II.
- 5. The method of embodiments 1-2, wherein said retinoic acid receptor agonist is AM580.
- 6. The method of embodiments 1-5, wherein said JNK inhibitor II is in combination with AM580.
- 7. The method of embodiments 3-6, wherein bFGF is present.
- 8. The method of
embodiments 7, wherein said bFGF is FGF2, FGF7 or FGF10. - 9. The method of embodiment 8, wherein said bFGF is FGF7.
- 10. The method of
embodiment 1, wherein said BMP inhibitor is LDN-193189. - 11. The method of embodiment 10, wherein said LDN-193189 is followed by exposure to a Wnt inhibitor or a hedgehog inhibitor.
- 12. The method of embodiment 11, wherein said Wnt inhibitor is IWP2.
- 13. The method of embodiment 12, wherein said hedgehog inhibitor is cyclopamine.
- 14. The method of embodiments 1-13, wherein said pancreatic cells or pancreatic cell precursors are 10-20%, 10-30%, 10-40%, 5-20%, 5-30%, 5-40%, 5-50%, 5-60% or 5-70%, 5-80%, 40-80% or 5-90% PDX1/NKX6.1 double positive.
- 15. The method of any one of embodiments 14, wherein said pancreatic cells or pancreatic cell precursors are 5-50% PDX1/NKX6.1 double positive.
- 16. The method of any one of embodiments 1-9, wherein said pancreatic cells or pancreatic cell precursors are 40-80% PDX1/NKX6.1 double positive.
- 17. A pancreatic cell or pancreatic cell precursors obtainable by the methods of embodiments 1-16.
- 18. A pancreatic cell or pancreatic cell precursor produced by exposing a human pluripotent stem cell to at least one compound listed in Tables 1 or 2.
- 19. The pancreatic cell or pancreatic cell precursor of embodiment 18, in which said compound is LDN-193189.
- 20. The pancreatic cell or pancreatic cell precursor of embodiment 18, in which said compound is JNK inhibitor II.
- 21. The pancreatic cell or pancreatic cell precursor of embodiment 18, in which said compound is AM580.
- 22. The pancreatic cell or pancreatic cell precursor of embodiment 18, in which the pancreatic cell or pancreatic cell precursor is produced by exposing the stem cell to at least one compound in combination with at least one additional agent.
- 23. The pancreatic cell or pancreatic cell precursor of embodiment 22, in which said LDN-193189 is in combination with JNK inhibitor II and AM580.
- 24. The pancreatic cell or pancreatic cell precursor of embodiment 22, in which said LDN-193189 is followed by exposure to cyclopamine.
- 25. The pancreatic cell or pancreatic cell precursor of embodiment 22, in which said LDN-193189 is followed by exposure to IWP2.
- 26. The pancreatic cell or pancreatic cell precursor of embodiment 22, in which said compound is JNK inhibitor II in combination with a retinoic acid receptor agonist listed in Table 2.
- 27. The pancreatic cell or pancreatic cell precursor of embodiment 22, wherein JNK inhibitor II is in combination with AM580.
- 28. The pancreatic cell or pancreatic cell precursor of embodiment 22, wherein JNK inhibitor II is in combination with AM580 and bFGF.
- 29. The pancreatic cell or pancreatic cell precursor of embodiment 28, wherein bFGF is FGF2, FGF7 or FGF10.
- 30. The pancreatic cell or pancreatic cell precursor of embodiment 29, wherein bFGF is FGF7.
- 31. Use of a compound of Tables 1 or 2 to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 32. Use of JNK inhibitor II to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 33. Use of JNK inhibitor II in combination with a retinoic acid receptor agonist to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 34. Use of JNK inhibitor II in combination with AM580 to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 35. Use of LDN-193189 to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 36. Use of LDN-193189 followed by Cyclopamine or IWP2 to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 37. Use of LDN-193189 followed by Cyclopamine to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 38. Use of LDN-193189 followed by IWP2 to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 39. Use of LDN-193189 followed by combination with JNK inhibitor II, AM580 and bFGF to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 40. Use of LDN-193189 followed by a combination of JNK inhibitor II, AM580, LDN-193189 and bFGF to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 41. Use of LDN-193189 followed by a combination of JNK inhibitor II, AM580, LDN-193189, bFGF and Cyclopamine or IWP2 to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 42. Use of LDN-193189 followed by a combination of JNK inhibitor II, AM580, LDN-193189, bFGF and Cyclopamine to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 43. Use of LDN-193189 followed by a combination of JNK inhibitor II, AM580, LDN-193189, bFGF and IWP2 to induce pancreatic cells or pancreatic cell precursors from stem cells.
- 44. A method of producing pancreatic cells or pancreatic cell precursors expressing at least 5% PDX1/NKX6.1 double positive, comprising exposing definitive endoderm cells to an effective amount of at least one of the compounds from each of the following groups:
- a. BMP inhibitor, and
- b. Kinase inhibitors, and
- c. Retinoic acid receptor agonists
- to differentiate the definitive endoderm cells into the pancreatic cell or pancreatic cell precursors.
- 45. The method of embodiment 44 wherein the BMP inhibitor is LDN-193189.
- 46. The method of embodiment 44 wherein the retinoic acid receptor agonist is AM580.
- 47. The method of embodiment 44 wherein the retinoic acid receptor agonist is a retinoic acid derivative.
- 48. The method of embodiment 44 wherein the kinase inhibitor is an isomer of 1,9-pyrazoloanthrone with or without N-alkylation.
- 49. The method of embodiment 44 wherein the kinase inhibitor is JNK inhibitor II and is in combination with AM580.
- 50. The method of embodiment 44 wherein said kinase inhibitors and retinoic acid receptor agonists are in combination with bFGF.
- 51. The method of embodiment 44 wherein said kinase inhibitors and retinoic acid receptor agonists are in combination with FGF7 or FGF10.
- 52. The method of embodiments 44-46, comprising a first step of exposing definitive endoderm cells to an effective amount of LDN-193189, without bFGF, and a second step of exposure to JNK inhibitor II in combination with AM580, in the presence of bFGF.
- 53. The method of embodiments 44-46, comprising exposing definitive endoderm cells to an effective amount of LDN-193189, following exposure to at least one compound of the group consisting of:
- a wnt inhibitor, such as IWP2, and/or
- a hedgehog inhibitor, such as cyclopamine
- 54. The method of any one of embodiments 44-53, wherein said pancreatic cells or pancreatic cell precursors are at least 5%, at least 10%, 10-30%, 10-40%, 5-70%, 10-80% or 5-100% PDX1/NKX6.1 double positive.
- 55. A pancreatic cell or pancreatic cell precursor obtainable by in vitro use of the methods of embodiments 44-54.
- 56. A pancreatic cell or pancreatic cell precursor produced by in vitro exposing definitive endoderm cells to a kinase inhibitor targeting JNK1, 2 or 3, or Syc or Src or GSK-3 or P38 MAPK or P38 kinase or Rho kinase or MEK or Chk2 or VEGFR1, 2 or 3 or PDGFRb or KDR/Flk-1.
- 57. A pancreatic cell or pancreatic cell precursor produced by the method of embodiment 56 wherein the kinase inhibitor is JNK inhibitor II and wherein the definitive endoderm cells are also exposed to at least one of the following compounds:
- LDN-193189,
- a Wnt inhibitor,
- a hedgehog inhibitor,
- a retinoic acid receptor agonist.
- 58. The pancreatic cell or pancreatic cell precursor of embodiment 56 wherein the definitive endoderm cells are exposed to JNK inhibitor II in combination with a retinoic acid receptor agonist listed in Table 2.
- 59. Use of a kinase inhibitor compound of Table 1 or 2 to induce pancreatic cells or pancreatic cell precursors from a pancreatic endoderm precursor.
- 60. Use of JNK inhibitor II and LDN-193189 in combination to induce pancreatic cells or pancreatic cell precursors from a pancreatic endoderm precursor.
- 61. Use of JNK inhibitor II in combination with a retinoic acid receptor agonist to induce pancreatic cells or pancreatic cell precursors from a pancreatic endoderm precursor.
- Human embryonic stem (hES) cells line SA121 and human induced pluripotent stem cells (hiPSC) chIPS4 (Cellectis) were grown in DEF-CS culture media (Cellectis) in T75 culture flasks. Cells were single cell passaged with 5 μM Rock inhibitor Y-27632 (Sigma #Y0503) and seeded at a density of 40000 cells/cm2 for experiments. Cells were cultured at 37° C. and 5% CO2 in a humidified incubator (ThermoScientific Model 371).
- In Vitro Differentiation of Pluripotent Stem Cells into Definitive Endoderm
- Confluent cultures of hES cells (line SA121) and hiPSC (chIPS4) were washed once in RPMI1640 (Gibco #61870) and treated with 3 μM CHIR99021 (Axon#1386) in RPMI1640. After 24 hours the cells were washed with RPMI1640 and treated with 100 ng/ml Activin A (Peprotech #120-14E) in RPMI1640. 24 hours later, 2% B27 (Invitrogen #17504-044) was added to the Activin A media for 2 days with daily media change. Cells were maintained at 37° C. and 5% CO2 in a humidified incubator during the differentiation.
- Seeding of hES and hiPS Cell Derived Definitive Endoderm.
- Human ES cell derived DE and Human iPS cell derived DE cells were washed in PBS−/− and trypsinized for 5 min. using Tryple Select (Invitrogen, 12563-029). DE cells were carefully suspended in RPMI1640 and washed once before they were resuspended in DE seeding medium (Activin A 100 ng/ml, 2% B27, RPMI1640, 0.1% PEST (Gibco #15140)). The DE cells were seeded at 200 000/cm2 in 96 well optical plates (BD Bioscience), and PE differentiation using screening compounds was initiated the following day.
- At day 8 or 14 of PE differentiation, media were aspirated followed by fixation of the cells at room temperature for 30 min with 4% paraformaldehyde (VWR, 97.131.000). Cells were washed with PBS and permeabilized with 0.5% Triton X-100 (Sigma, 9002-93-1) for 10 min, washed and blocked in 0.5% TNB-buffer (Perkin Elmer) for 30 min at room temperature. Primary antibodies mouse anti-NKX6.1 (Abcore#A55F12) and goat anti-PDX1 (Abcam#47383) were diluted 1:500 and 1:8000, respectively, in 0.1% Triton X-100 in PBS and added to each well for overnight incubation at 4° C. Cells were washed three times with PBS. DAPI (4′,6-diamidino-2-phenylindole, Applichem, A4099.0010) and secondary antibodies, Alexa Fluor 488 donkey anti-goat and Alexa Fluor 594 donkey anti-mouse (both Invitrogen) were diluted 1:1000 in 0.1% Triton X-100 in PBS and added to each well for 45 min. Cells were washed five times and left in 200 μL PBS for imaging.
- Imaging was performed using the InCell Analyzer 2000 (GE Healthcare). 4 fields per well with 10× objective were captured. The total cell number based in the DAPI counterstaining and the number of NKX6.1/PDX1 double positive cells was determined using InCell Developer Toolbox 1.8 (GE Healthcare). The fraction of NKX6.1/PDX1 double positive cells was normalized to the benchmark on each plate and the % effect was calculated. Values above 200% effect were categorized as hits.
- Pancreatic endoderm is characterized by co-expression of two transcription factors, NKX6.1 and PDX1. Many of the published protocols for making PE are ineffective with low outcome of NKX6.1/PDX1 double positive cells. Enhancing the efficacy of the PE protocols is a desirable outcome. We therefore screened libraries of small molecules to identify novel compounds that would improve the existing PE protocols. This was done on the assumption that inhibitors, agonists or antagonists may regulate signaling pathways, or chromosomal accessibility, which would improve the fraction of NKX6.1/PDX1 double positive cells.
- Four different libraries were included in the screen; a kinase inhibitor library (Calbiochem #539743), a bioactive lipid library (Enzo Life Sciences #BML-2800), a nuclear receptor ligand library (Enzo Life Sciences # BML-2802) and a phosphatase inhibitor library (Enzo Life Sciences #BML-2834). The compounds within the bioactive library were tested at 1 uM and 0.1 uM. Compounds from the other libraries were tested at 10 uM and 1 uM. In a second candidate based screening approach, small molecules that target the main signalling pathways involved in pancreas development were included.
- NKX6.1/PDX1 Screen The library compounds were screened on top of a bFGF based media formulation for making PE (Ameri et al. 2010) (RPMI1640, Gibco#61870; 12% KOSR, Gibco#10828; 0.1% PEST, Gibco#15140; 64 ng/mL bFGF, Peprotech #100-18B).
- The library PE screening approach was divided into an early and a late phase (
FIG. 1 ). - In the early phase, compounds were tested on top of the PE media for the first seven days of PE differentiation, and then the differentiation was continued for additional six days using PE media without compounds.
- In the late phase, DE cells were differentiated in the PE media for the first seven days. In the following six days compounds were tested on top of the PE media. 12 positive control wells (PE media) and 12 negative control wells (PE media without bFGF) were included in each 96 well plate. Media change was performed daily. Hits identified in the early phase screen are illustrated in
FIG. 2 and listed in table 1. Hits identified in the late phase screen are illustrated inFIG. 3 and listed in table 2. - The compounds from the candidate approach were screened in basal medium (RPMI1640, Gibco#61870; 12% KOSR, Gibco#10828; 0.1% PEST, Gibco#15140) without the addition of bFGF. This candidate approach screen was divided into two parts (
FIG. 4 ). In the first part, compounds were tested in time intervals with 2 day increments for the first eight days of PE differentiation (2 days exposure to compounds followed by 6 days basal medium or 4 days exposure to compounds followed by 4 days basal medium or 6 days exposure to compounds followed by 2 days basal medium or 8 days exposure to compounds). - One plate was fixed after these 8 days and analysed for PDX1 and NKX6.1 expression. A second plate was further differentiated for additional six days using the published PE protocol (Amen et al. (2010)).
- In the second part, DE cells were differentiated according to the hit compounds from the first part, the following 6-10 days compounds were tested in basal media.
- The benchmark protocol (Amen et al. (2010)) served as a control.
- Media change was performed daily in both first and second part experiments.
- Hits identified in the candidate screening approach are illustrated in
FIG. 5 and also contained in Tables 1 and 2. - Combining Hits from the Candidate Screening Approach with Hits from the Library Approach
- DE cells were exposed to 4 days 50 nM LDN-193189, followed by 8 days AM580 (AH Diagnostics, BML GF104 0025), JNK Inhibitor II (Calbiochem, 420119), 50 nM LDN-193189 and 64 ng/ml FGF2, or AM580, JNK Inhibitor II, 50 nM LDN-193189, 64 ng/ml FGF2 and IWP2, or AM580, JNK Inhibitor II, 50 nM LDN-193189, 64 ng/ml FGF2 and Cyclopamine (
FIG. 6 ). Media change was performed daily. - Hit compounds (Tables 1 and 2) were screened on top of a bFGF based media formulation for making PE (Amen et al. 2010) (RPMI1640, Gibco#61870; 12% KOSR, Gibco#10828; 0.1% PEST, Gibco#15140; 64 ng/mL bFGF, Peprotech #100-18B).
- The screen was divided into an early and a late phase (
FIG. 1 ). In the early phase, compounds were tested on top of the PE media for the first seven days of PE differentiation, and then continued for additional six days using PE media without compounds. In the late phase, DE cells were differentiated in the PE media for the first seven days. In the following six days compounds were tested on top of the PE media. Twelve positive control wells (PE media) and 12 negative control wells (PE media without bFGF) were included in each 96 well plate. Media change was performed daily. - Values above 200% effect were categorized as a hit (
FIG. 2 andFIG. 3 ). - Compounds that enhance the fraction of NKX6.1/PDX1 double positive cells more than 200% compared the PE media. Library, location of compound within the library, target, chemical structure, hit concentration and percentage of PDX1/NKX6.1 double positive cells are listed.
-
TABLE 1 Early phase hits. Plate Library location Target Inhibitor Structure Concentration Kinase inhibitor B3 ATM kinase ATM Kinase Inhibitor 10 uM Kinase inhibitor D18 Chk2 Chk2 Inhibitor II 10 uM Kinase inhibitor N15 P38 MAPK SKF-86002 10 uM Kinase inhibitor P20 KDR/Flk-1, VEGGFR-2, PDGFRb, VEGFR-1, VEGFR-3 VEGF Receptor 2 Kinase Inhibitor IV10 uM Kinase inhibitor H22 JNK1, JNK2, JNK3, JNK Inhibitor II 10 uM Kinase inhibitor H22 JNK1, JNK2, JNK3 JNK Inhibitor II 1 uM Candidate screen BMP inhibitor Candidate approach ALK2, ALK3, ALK6 LDN-193189 50 nM/100 nM - Compounds that enhance the fraction of NKX6.1/PDX1 double positive cells more than 200% compared the PE media. Library, location of compound within the library, target, chemical structure, hit concentration and percentage of PDX1/NKX6.1 double positive cells are listed.
-
TABLE 2 Late phase hits. Plate Library location Target Inhibitor Structure Concentration Kinase inhibitor H22 JNK1, JNK2, JNK3 JNK Inhibitor II 10 uM Kinase inhibitor J4 JNK Inhibitor, Negative Control 10 uM Kinase inhibitor P5 syc, src SYK Inhibitor III 10 uM Kinase inhibitor F20 GSK-3 GSK-3b Inhibitor II 10 uM Kinase inhibitor N15 P38 MAPK SKF-86002 10 uM Kinase inhibitor J14 PI3-kinase LY 294002 10 uM Kinase inhibitor N14 P38 MAPK SC-68376 10 uM Kinase inhibitor N6 Rho kinase Rho Kinase Inhibitor III, Rockout 10 uM Kinase inhibitor L5 MEK PD 98059 10 uM Kinase inhibitor N11 P38 MAPK SB203580 10 uM Bioactive lipids 2-E09 RAR AM-580 1 uM Bioactive lipids 2-E05 RAR RETINOIC ACID, ALL TRANS 1 uM Bioactive lipids 2-E07 RAR/RXR 13-CIS RETINOIC ACID 1 uM Bioactive lipids 2-E06 RAR/RXR 9-CIS RETINOIC ACID 1 uM Bioactive lipids 2-E10 RAR TTNPB 1 uM Kinase inhibitor H22 JNK1, JNK2, JNK3 JNK Inhibitor II 1 uM Kinase inhibitor J4 JNK Inhibitor, Negative Control 1 uM Bioactive lipids 2-E09 RAR AM-580 0.1 uM Bioactive liids 2-E07 RAR/RXR 13-CIS RETINOIC ACID 0.1 uM Bioactive lipids 2-E06 RAR/RXR 9-CIS RETINOIC ACID 0.1 uM Candidate screen Hedgehog pathway inhibitor Smoothened (SMO) Cydopamine 1 μM/5 μM Candidate screen Wnt pathway inhibitor O-acyltransferase Porcupine IWP2 1 μM/5 μM - While certain features of the invention have been illustrated and described herein, many modifications, substitutions, changes, and equivalents will now occur to those of ordinary skill in the art. It is, therefore, to be understood that the appended claims are intended to cover all such modifications and changes as fall within the true spirit of the invention.
-
- Amen et al. (2010) Stem Cells, 28:45-56
- Cai J. et al. (2010). J Mol Cell Biol., February; 2(1):50-60
- Chung Y. et al. (2006). Nature, January 12; 439(7073):216-9
- D'Amour K. A. et al. (2006). Nat Biotechnol, 24: 1392-401
- Heins N. et al. (2004). Stem Cells, 22(3):367-76
- Jiang J. et al. (2007). Stem Cells, 25:1940-53
- Kroon E. et al. (2008). Nat Biotechnol, 26:443-452
- Klimanskaya I. et al. (2006). Nature, November 23; 444(7118):481-5
- Kunisada Y. et al. (2012). Stem Cell Res, 8(2):274-84
- Schulz T C. et al. (2012). PLoS One, 7(5):e37004
- Takahashi K. et al. (2007). Cell, 131:861-72
- Takahashi K. and Yamanaka S. (2006). Cell, 126(4):663-76
- Thomson J A. et al. (1998). Science, November 6; 282(5391):1145-7
- Wernig, M. et al. (2007). Nature, 448:318-24
- Zhang D. et al. (2009). Cell Research, 19:429-438
Claims (16)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/425,136 US20150247123A1 (en) | 2012-09-03 | 2013-09-03 | Generation of pancreatic endoderm from Pluripotent Stem cells using small molecules |
Applications Claiming Priority (7)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP12182747 | 2012-09-03 | ||
| EP12182747.1 | 2012-09-03 | ||
| US201261697970P | 2012-09-07 | 2012-09-07 | |
| EP12198820 | 2012-12-21 | ||
| EP12198820.8 | 2012-12-21 | ||
| PCT/EP2013/068188 WO2014033322A1 (en) | 2012-09-03 | 2013-09-03 | Generation of pancreatic endoderm from pluripotent stem cells using small molecules |
| US14/425,136 US20150247123A1 (en) | 2012-09-03 | 2013-09-03 | Generation of pancreatic endoderm from Pluripotent Stem cells using small molecules |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/EP2013/068188 A-371-Of-International WO2014033322A1 (en) | 2012-09-03 | 2013-09-03 | Generation of pancreatic endoderm from pluripotent stem cells using small molecules |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/215,807 Division US10221392B2 (en) | 2012-09-03 | 2016-07-21 | Generation of pancreatic endoderm from pluripotent stem cells using small molecules |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20150247123A1 true US20150247123A1 (en) | 2015-09-03 |
Family
ID=50182554
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US14/425,136 Abandoned US20150247123A1 (en) | 2012-09-03 | 2013-09-03 | Generation of pancreatic endoderm from Pluripotent Stem cells using small molecules |
| US15/215,807 Active US10221392B2 (en) | 2012-09-03 | 2016-07-21 | Generation of pancreatic endoderm from pluripotent stem cells using small molecules |
| US16/255,347 Abandoned US20190153394A1 (en) | 2012-09-03 | 2019-01-23 | Generation of pancreatic endoderm from pluripotent stem cells using small molecules |
Family Applications After (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US15/215,807 Active US10221392B2 (en) | 2012-09-03 | 2016-07-21 | Generation of pancreatic endoderm from pluripotent stem cells using small molecules |
| US16/255,347 Abandoned US20190153394A1 (en) | 2012-09-03 | 2019-01-23 | Generation of pancreatic endoderm from pluripotent stem cells using small molecules |
Country Status (5)
| Country | Link |
|---|---|
| US (3) | US20150247123A1 (en) |
| EP (1) | EP2893000B1 (en) |
| JP (2) | JP6470687B2 (en) |
| CN (1) | CN104903440B (en) |
| WO (1) | WO2014033322A1 (en) |
Cited By (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20160186143A1 (en) * | 2014-12-18 | 2016-06-30 | President And Fellows Of Harvard College | Methods for generating stem cell-derived beta cells and uses thereof |
| US10030229B2 (en) | 2013-06-11 | 2018-07-24 | President And Fellows Of Harvard College | SC-β cells and compositions and methods for generating the same |
| US10253298B2 (en) | 2014-12-18 | 2019-04-09 | President And Fellows Of Harvard College | Methods for generating stem cell-derived beta cells and methods of use thereof |
| US10443042B2 (en) | 2014-12-18 | 2019-10-15 | President And Fellows Of Harvard College | Serum-free in vitro directed differentiation protocol for generating stem cell-derived beta cells and uses thereof |
| WO2020041608A1 (en) * | 2018-08-22 | 2020-02-27 | The Penn State Research Foundation | Generation of pancreatic progenitor cells and beta cells from human pluripotent stem cells |
| WO2021099532A1 (en) | 2019-11-22 | 2021-05-27 | Novo Nordisk A/S | Spin-aggregated neural microspheres and the application thereof |
| US11466256B2 (en) | 2018-08-10 | 2022-10-11 | Vertex Pharmaceuticals Incorporated | Stem cell derived islet differentiation |
| US11945795B2 (en) | 2017-11-15 | 2024-04-02 | Vertex Pharmaceuticals Incorporated | Islet cell manufacturing compositions and methods of use |
Families Citing this family (35)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9080145B2 (en) | 2007-07-01 | 2015-07-14 | Lifescan Corporation | Single pluripotent stem cell culture |
| US9096832B2 (en) | 2007-07-31 | 2015-08-04 | Lifescan, Inc. | Differentiation of human embryonic stem cells |
| CA2959401C (en) | 2008-02-21 | 2021-12-07 | Centocor Ortho Biotech Inc. | Methods, surface modified plates and compositions for cell attachment, cultivation and detachment |
| KR20180018839A (en) | 2008-06-30 | 2018-02-21 | 얀센 바이오테크 인코포레이티드 | Differentiation of pluripotent stem cells |
| MX356756B (en) | 2008-11-20 | 2018-06-11 | Centocor Ortho Biotech Inc | Pluripotent stem cell culture on micro-carriers. |
| ES2584053T3 (en) | 2008-11-20 | 2016-09-23 | Janssen Biotech, Inc. | Methods and compositions for cell binding and culture in flat substrates |
| WO2011011300A2 (en) | 2009-07-20 | 2011-01-27 | Centocor Ortho Biotech Inc. | Differentiation of human embryonic stem cells |
| KR101773538B1 (en) | 2009-12-23 | 2017-08-31 | 얀센 바이오테크 인코포레이티드 | Differentiation of human embryonic stem cells |
| PH12012501686A1 (en) | 2010-03-01 | 2020-10-19 | Janssen Biotech Inc | Methods for purifying cells derived from pluripotent stem cells |
| RU2620938C2 (en) | 2010-08-31 | 2017-05-30 | Янссен Байотек, Инк. | Differentiation of human embryonic stem cells |
| US9388386B2 (en) | 2011-12-22 | 2016-07-12 | Janssen Biotech, Inc. | Differentiation of human embryonic stem cells into single hormonal insulin positive cells |
| CN108103005A (en) | 2012-06-08 | 2018-06-01 | 詹森生物科技公司 | Differentiation of the human embryo stem cell to pancreatic endocrine cell |
| US20150247123A1 (en) * | 2012-09-03 | 2015-09-03 | Novo Nordisk A/S | Generation of pancreatic endoderm from Pluripotent Stem cells using small molecules |
| JP6529440B2 (en) | 2012-12-31 | 2019-06-12 | ヤンセン バイオテツク,インコーポレーテツド | Suspension and clustering of human pluripotent cells for differentiation to pancreatic endocrine cells |
| ES2942484T3 (en) | 2012-12-31 | 2023-06-01 | Janssen Biotech Inc | Differentiation of human embryonic stem cells into pancreatic endocrine cells using HB9 regulators |
| SG10201709338RA (en) | 2012-12-31 | 2017-12-28 | Janssen Biotech Inc | Culturing of human embryonic stem cells at the air-liquid interface for differentiation into pancreatic endocrine cells |
| US10370644B2 (en) | 2012-12-31 | 2019-08-06 | Janssen Biotech, Inc. | Method for making human pluripotent suspension cultures and cells derived therefrom |
| CN117821369A (en) * | 2014-05-16 | 2024-04-05 | 詹森生物科技公司 | Use of small molecules to enhance MAFA expression in pancreatic endocrine cells |
| CN108366987A (en) | 2015-04-30 | 2018-08-03 | 河谷控股Ip有限责任公司 | Patient is treated by teratogenic drug compound |
| CN108699515A (en) * | 2016-02-24 | 2018-10-23 | 诺和诺德股份有限公司 | The endocrine progenitor cells generating functionality β cells derived from human pluripotent stem cells |
| MA45479A (en) | 2016-04-14 | 2019-02-20 | Janssen Biotech Inc | DIFFERENTIATION OF PLURIPOTENT STEM CELLS IN ENDODERMAL CELLS OF MIDDLE INTESTINE |
| MA45502A (en) | 2016-06-21 | 2019-04-24 | Janssen Biotech Inc | GENERATION OF FUNCTIONAL BETA CELLS DERIVED FROM HUMAN PLURIPOTENT STEM CELLS WITH GLUCOSE-DEPENDENT MITOCHONDRIAL RESPIRATION AND TWO-PHASE INSULIN SECRETION RESPONSE |
| WO2018035454A1 (en) * | 2016-08-19 | 2018-02-22 | Memorial Sloan-Kettering Cancer Center | Methods of differentiating stem cells into endoderm |
| EP3395942A1 (en) * | 2017-04-25 | 2018-10-31 | IMBA-Institut für Molekulare Biotechnologie GmbH | Bi- or multi-differentiated organoid |
| CA3074910A1 (en) | 2017-09-11 | 2019-03-14 | Novo Nordisk A/S | Enrichment of nkx6.1 and c-peptide co-expressing cells derived in vitro from stem cells |
| WO2020043292A1 (en) | 2018-08-30 | 2020-03-05 | Novo Nordisk A/S | Generation of functional beta cells from human pluripotent stem cell-derived endocrine progenitors |
| CN113728090B (en) * | 2019-04-08 | 2024-04-05 | 诺和诺德股份有限公司 | Pancreatic endoderm generation from stem cell-derived definitive endoderm |
| EP4386081A3 (en) * | 2019-05-21 | 2024-10-16 | President and Fellows of Harvard College | Endocrine differentiation-inducing molecule |
| US20230313145A1 (en) | 2020-08-28 | 2023-10-05 | Novo Nordisk A/S | Method for screening in vitro population of stem cell derived beta like cells and novel markers thereof |
| WO2024008810A1 (en) | 2022-07-06 | 2024-01-11 | Novo Nordisk A/S | Differentiation of stem cells to pancreatic endocrine cells |
| WO2024151541A1 (en) | 2023-01-09 | 2024-07-18 | Sana Biotechnology, Inc. | Type-1 diabetes autoimmune mouse |
| WO2024243236A2 (en) | 2023-05-22 | 2024-11-28 | Sana Biotechnology, Inc. | Methods of delivery of islet cells and related methods |
| WO2025093467A1 (en) | 2023-10-31 | 2025-05-08 | Novo Nordisk A/S | Methods for preventing unwanted cells arising during differentiation of human pluripotent stem cells |
| EP4491712A1 (en) | 2024-03-20 | 2025-01-15 | Novo Nordisk A/S | Method for detecting contaminant cells in a stem cell derived cell population |
| WO2025235403A1 (en) * | 2024-05-06 | 2025-11-13 | Vertex Pharmaceuticals Incorporated | Differentiation of pancreatic islet cells |
Family Cites Families (45)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP1298201A1 (en) | 2001-09-27 | 2003-04-02 | Cardion AG | Process for the production of cells exhibiting an islet-beta-cell-like state |
| US20040005301A1 (en) | 2002-02-12 | 2004-01-08 | Goldman Steven A. | Identification and high-yield isolation of human pancreatic islet progenitor and stem cells |
| WO2005005608A2 (en) | 2003-06-30 | 2005-01-20 | Lifescan, Inc. | Compositions and methods for differentiating adipose stromal cells into pancratic beta cells |
| AU2005221095A1 (en) | 2004-03-09 | 2005-09-22 | John J. O'neil | Methods for generating insulin-producing cells |
| US20090053181A1 (en) | 2005-06-13 | 2009-02-26 | Novo Nordisk A/S | Modulation of Cells |
| EP1999253B1 (en) | 2006-03-02 | 2019-05-22 | Viacyte, Inc. | Endocrine precursor cells, pancreatic hormone-expressing cells and methods of production |
| ES2725601T3 (en) | 2006-04-28 | 2019-09-25 | Lifescan Inc | Differentiation of human embryonic stem cells |
| SE534150C2 (en) | 2006-05-02 | 2011-05-10 | Wisconsin Alumni Res Found | Procedure for differentiating stem cells into cells of the endodermal and pancreatic developmental line |
| CA2893679C (en) | 2006-06-26 | 2018-10-30 | Lifescan, Inc. | Conditioned medium for pluripotent stem cell culture |
| CA2666789C (en) | 2006-10-18 | 2016-11-22 | Yong Zhao | Embryonic-like stem cells derived from adult human peripheral blood and methods of use |
| DK3192865T3 (en) | 2007-07-01 | 2021-06-28 | Janssen Biotech Inc | SINGLE PLURIPOTENT STEM CELL CULTURE |
| US9080145B2 (en) | 2007-07-01 | 2015-07-14 | Lifescan Corporation | Single pluripotent stem cell culture |
| EP2185695B1 (en) | 2007-07-18 | 2015-02-25 | Lifescan, Inc. | Differentiation of human embryonic stem cells |
| JP6087043B2 (en) | 2007-07-31 | 2017-03-01 | ライフスキャン・インコーポレイテッドLifescan,Inc. | Differentiation of pluripotent stem cells using human feeder cells |
| US9096832B2 (en) | 2007-07-31 | 2015-08-04 | Lifescan, Inc. | Differentiation of human embryonic stem cells |
| SG154367A1 (en) | 2008-01-31 | 2009-08-28 | Es Cell Int Pte Ltd | Method of differentiating stem cells |
| US8338170B2 (en) | 2008-04-21 | 2012-12-25 | Viacyte, Inc. | Methods for purifying endoderm and pancreatic endoderm cells derived from human embryonic stem cells |
| DK2727998T3 (en) | 2008-04-21 | 2019-08-26 | Viacyte Inc | Methods for purifying pancreatic endoderm cells derived from human embryonic stem cells |
| US8728812B2 (en) | 2008-04-22 | 2014-05-20 | President And Fellows Of Harvard College | Compositions and methods for promoting the generation of PDX1+ pancreatic cells |
| US7939322B2 (en) | 2008-04-24 | 2011-05-10 | Centocor Ortho Biotech Inc. | Cells expressing pluripotency markers and expressing markers characteristic of the definitive endoderm |
| KR20180018839A (en) | 2008-06-30 | 2018-02-21 | 얀센 바이오테크 인코포레이티드 | Differentiation of pluripotent stem cells |
| US20100028307A1 (en) | 2008-07-31 | 2010-02-04 | O'neil John J | Pluripotent stem cell differentiation |
| US9234178B2 (en) * | 2008-10-31 | 2016-01-12 | Janssen Biotech, Inc. | Differentiation of human pluripotent stem cells |
| CN102333862B (en) | 2008-10-31 | 2018-04-27 | 詹森生物科技公司 | Differentiation of human embryonic stem cells to the pancreatic endocrine lineage |
| EP3550012A1 (en) | 2008-11-04 | 2019-10-09 | ViaCyte, Inc. | Stem cell aggregate suspension compositions and methods for differentiation thereof |
| US8008075B2 (en) | 2008-11-04 | 2011-08-30 | Viacyte, Inc. | Stem cell aggregate suspension compositions and methods of differentiation thereof |
| EP2356227B1 (en) | 2008-11-14 | 2018-03-28 | Viacyte, Inc. | Encapsulation of pancreatic cells derived from human pluripotent stem cells |
| MX356756B (en) | 2008-11-20 | 2018-06-11 | Centocor Ortho Biotech Inc | Pluripotent stem cell culture on micro-carriers. |
| US8507274B2 (en) | 2009-02-06 | 2013-08-13 | President And Fellows Of Harvard College | Compositions and methods for promoting the generation of definitive endoderm |
| WO2010096496A2 (en) * | 2009-02-17 | 2010-08-26 | Memorial Sloan-Kettering Cancer Center | Methods of neural conversion of human embryonic stem cells |
| EP2233566A1 (en) | 2009-03-17 | 2010-09-29 | Vrije Universiteit Brussel | Generation of pancreatic progenitor cells |
| EP3904505A1 (en) * | 2009-04-22 | 2021-11-03 | Viacyte, Inc. | Cell compositions derived from dedifferentiated reprogrammed cells |
| JP2012527880A (en) * | 2009-05-29 | 2012-11-12 | ノヴォ ノルディスク アー/エス | Derivation of specific endoderm from hPS cell-derived definitive endoderm |
| JP5819826B2 (en) | 2009-07-20 | 2015-11-24 | ヤンセン バイオテツク,インコーポレーテツド | Differentiation of human embryonic stem cells |
| KR101785626B1 (en) | 2009-07-20 | 2017-10-16 | 얀센 바이오테크 인코포레이티드 | Differentiation of human embryonic stem cells |
| KR101773538B1 (en) * | 2009-12-23 | 2017-08-31 | 얀센 바이오테크 인코포레이티드 | Differentiation of human embryonic stem cells |
| AU2010333840B2 (en) | 2009-12-23 | 2016-01-07 | Janssen Biotech, Inc. | Differentiation of human embryonic stem cells |
| EP2505639B1 (en) * | 2009-12-29 | 2018-09-19 | Takeda Pharmaceutical Company Limited | Method for manufacturing pancreatic-hormone-producing cells |
| WO2011100291A1 (en) | 2010-02-09 | 2011-08-18 | The Regents Of The University Of California | Method for identifying pancreatic progenitor cells |
| CN102884176B (en) * | 2010-05-12 | 2017-09-05 | 詹森生物科技公司 | Differentiation of human embryonic stem cells |
| EP2606122A1 (en) | 2010-08-22 | 2013-06-26 | Ramot at Tel Aviv University, Ltd. | Induced pluripotent stem cells derived from human pancreatic beta cells |
| AU2011296383B2 (en) * | 2010-08-31 | 2016-03-10 | Janssen Biotech, Inc. | Differentiation of pluripotent stem cells |
| ES2902650T3 (en) | 2011-06-21 | 2022-03-29 | Novo Nordisk As | Efficient induction of definitive endoderm from pluripotent stem cells |
| JP6143268B2 (en) * | 2011-12-19 | 2017-06-07 | 国立大学法人京都大学 | Method for inducing differentiation from human pluripotent stem cells to intermediate mesoderm cells |
| US20150247123A1 (en) * | 2012-09-03 | 2015-09-03 | Novo Nordisk A/S | Generation of pancreatic endoderm from Pluripotent Stem cells using small molecules |
-
2013
- 2013-09-03 US US14/425,136 patent/US20150247123A1/en not_active Abandoned
- 2013-09-03 JP JP2015529065A patent/JP6470687B2/en active Active
- 2013-09-03 CN CN201380045948.2A patent/CN104903440B/en active Active
- 2013-09-03 WO PCT/EP2013/068188 patent/WO2014033322A1/en not_active Ceased
- 2013-09-03 EP EP13756893.7A patent/EP2893000B1/en active Active
-
2016
- 2016-07-21 US US15/215,807 patent/US10221392B2/en active Active
-
2018
- 2018-10-11 JP JP2018192572A patent/JP6954877B2/en active Active
-
2019
- 2019-01-23 US US16/255,347 patent/US20190153394A1/en not_active Abandoned
Non-Patent Citations (1)
| Title |
|---|
| Kim et al., Current Opinion in Genetics & Development 2002, 12:540â547 * |
Cited By (28)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11078463B2 (en) | 2013-06-11 | 2021-08-03 | President And Fellows Of Harvard College | SC-beta cells and compositions and methods for generating the same |
| US10030229B2 (en) | 2013-06-11 | 2018-07-24 | President And Fellows Of Harvard College | SC-β cells and compositions and methods for generating the same |
| US11827905B2 (en) | 2013-06-11 | 2023-11-28 | President And Fellows Of Harvard College | SC-beta cells and compositions and methods for generating the same |
| US12049646B2 (en) | 2013-06-11 | 2024-07-30 | President And Fellows Of Harvard College | SC-beta cells and compositions and methods for generating the same |
| US11162078B2 (en) | 2013-06-11 | 2021-11-02 | President And Fellows Of Harvard College | SC-beta cells and compositions and methods for generating the same |
| US12054745B2 (en) | 2013-06-11 | 2024-08-06 | President And Fellows Of Harvard College | SC-beta cells and compositions and methods for generating the same |
| US10655106B2 (en) | 2013-06-11 | 2020-05-19 | President And Fellows Of Harvard College | SC-beta cells and compositions and methods for generating the same |
| US11104883B2 (en) | 2013-06-11 | 2021-08-31 | President And Fellows Of Harvard College | SC-beta cells and compositions and methods for generating the same |
| US11155787B2 (en) | 2014-12-18 | 2021-10-26 | President And Fellows Of Harvard College | Methods for generating stem cell-derived beta cells and methods of use thereof |
| US12123023B2 (en) | 2014-12-18 | 2024-10-22 | President And Fellows Of Harvard College | Methods for generating stem cell-derived β cells and methods of use thereof |
| US11085025B2 (en) | 2014-12-18 | 2021-08-10 | President And Fellows Of Harvard College | Serum-free in vitro directed differentiation protocol for generating stem cell-derived beta cells and uses thereof |
| US11085027B2 (en) | 2014-12-18 | 2021-08-10 | President And Fellows Of Harvard College | Serum-free in vitro directed differentiation protocol for generating stem cell-derived beta cells and uses thereof |
| US11085026B2 (en) | 2014-12-18 | 2021-08-10 | President And Fellows Of Harvard College | Serum-free in vitro directed differentiation protocol for generating stem cell-derived beta cells and uses thereof |
| US10927350B2 (en) | 2014-12-18 | 2021-02-23 | President And Fellows Of Harvard College | Methods for generating stem cell-derived beta cells and uses thereof |
| US10443042B2 (en) | 2014-12-18 | 2019-10-15 | President And Fellows Of Harvard College | Serum-free in vitro directed differentiation protocol for generating stem cell-derived beta cells and uses thereof |
| US10253298B2 (en) | 2014-12-18 | 2019-04-09 | President And Fellows Of Harvard College | Methods for generating stem cell-derived beta cells and methods of use thereof |
| US20160186143A1 (en) * | 2014-12-18 | 2016-06-30 | President And Fellows Of Harvard College | Methods for generating stem cell-derived beta cells and uses thereof |
| US10190096B2 (en) * | 2014-12-18 | 2019-01-29 | President And Fellows Of Harvard College | Methods for generating stem cell-derived β cells and uses thereof |
| US12304900B2 (en) | 2017-11-15 | 2025-05-20 | Vertex Pharmaceuticals Incorporated | Islet cell manufacturing compositions and methods of use |
| US11945795B2 (en) | 2017-11-15 | 2024-04-02 | Vertex Pharmaceuticals Incorporated | Islet cell manufacturing compositions and methods of use |
| US11999971B2 (en) | 2018-08-10 | 2024-06-04 | Vertex Pharmaceuticals Incorporated | Stem cell derived islet differentiation |
| US11525120B2 (en) | 2018-08-10 | 2022-12-13 | Vertex Pharmaceuticals Incorporated | Stem cell derived islet differentiation |
| US11466256B2 (en) | 2018-08-10 | 2022-10-11 | Vertex Pharmaceuticals Incorporated | Stem cell derived islet differentiation |
| US12215355B2 (en) | 2018-08-10 | 2025-02-04 | Vertex Pharmaceuticals Incorporated | Stem cell derived islet differentiation |
| US20210324337A1 (en) * | 2018-08-22 | 2021-10-21 | The Penn State Research Foundation | Generation of pancreatic progenitor cells and beta cells from human pluripotent stem cells |
| WO2020041608A1 (en) * | 2018-08-22 | 2020-02-27 | The Penn State Research Foundation | Generation of pancreatic progenitor cells and beta cells from human pluripotent stem cells |
| US12516295B2 (en) * | 2018-08-22 | 2026-01-06 | The Penn State Research Foundation | Generation of pancreatic progenitor cells and beta cells from human pluripotent stem cells |
| WO2021099532A1 (en) | 2019-11-22 | 2021-05-27 | Novo Nordisk A/S | Spin-aggregated neural microspheres and the application thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| JP2015528289A (en) | 2015-09-28 |
| US20190153394A1 (en) | 2019-05-23 |
| JP2019000126A (en) | 2019-01-10 |
| EP2893000B1 (en) | 2019-04-10 |
| EP2893000A1 (en) | 2015-07-15 |
| US20160326495A1 (en) | 2016-11-10 |
| JP6470687B2 (en) | 2019-02-13 |
| US10221392B2 (en) | 2019-03-05 |
| JP6954877B2 (en) | 2021-10-27 |
| CN104903440A (en) | 2015-09-09 |
| CN104903440B (en) | 2018-04-06 |
| WO2014033322A1 (en) | 2014-03-06 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US10221392B2 (en) | Generation of pancreatic endoderm from pluripotent stem cells using small molecules | |
| JP7420860B2 (en) | Methods and systems for converting progenitor cells into gastric tissue by directed differentiation | |
| US20200002670A1 (en) | Generation of Endocrine Progenitor Cells from Human Pluripotent Stem Cells Using Small Molecules | |
| US11274280B2 (en) | Generation of functional beta cells from human pluripotent stem cell-derived endocrine progenitors | |
| US10457916B2 (en) | Method for inducing differentiation of insulin-producing cells | |
| US20210230554A1 (en) | Generation of functional beta cells from human pluripotent stem cell-derived endocrine progenitors | |
| CN113728090B (en) | Pancreatic endoderm generation from stem cell-derived definitive endoderm | |
| ES2729776T3 (en) | Generation of pancreatic endoderm from pluripotent stem cells through the use of small molecules | |
| CN112119154A (en) | Production of brown fat cells | |
| Horst | Activin-A and Bmp4 Levels Modulate Cell Type Specification during CHIR-Induced Cardiomyogenesis | |
| ES2746232T3 (en) | Generation of endocrine progenitor cells from human pluripotent stem cells using small molecules | |
| Class et al. | Patent application title: Generation of Endocrine Progenitor Cells from Human Pluripotent Stem Cells Using Small Molecules | |
| Rotti | 3D differentiation enhances the efficiency of differentiation of human induced pluripotent stem cells to insulin producing cells |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: NOVO NORDISK A/S, DENMARK Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:EKBERG, JENNY;HANSSON, MATTIAS;DOEHN, ULRIK;AND OTHERS;SIGNING DATES FROM 20150325 TO 20150508;REEL/FRAME:035765/0626 |
|
| AS | Assignment |
Owner name: NOVO NORDISK A/S, DENMARK Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:EKBERG, JENNY;HANSSON, MATTIAS;DOEHN, ULRIK;AND OTHERS;SIGNING DATES FROM 20150818 TO 20150823;REEL/FRAME:036749/0785 Owner name: CELLECTIS SA, FRANCE Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:EKBERG, JENNY;HANSSON, MATTIAS;DOEHN, ULRIK;AND OTHERS;SIGNING DATES FROM 20150818 TO 20150823;REEL/FRAME:036750/0059 |
|
| AS | Assignment |
Owner name: TAKARA BIO EUROPE AB, SWEDEN Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNOR:CELLECTIS SA;REEL/FRAME:037071/0840 Effective date: 20150416 |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |