WO2024151541A1 - Type-1 diabetes autoimmune mouse - Google Patents
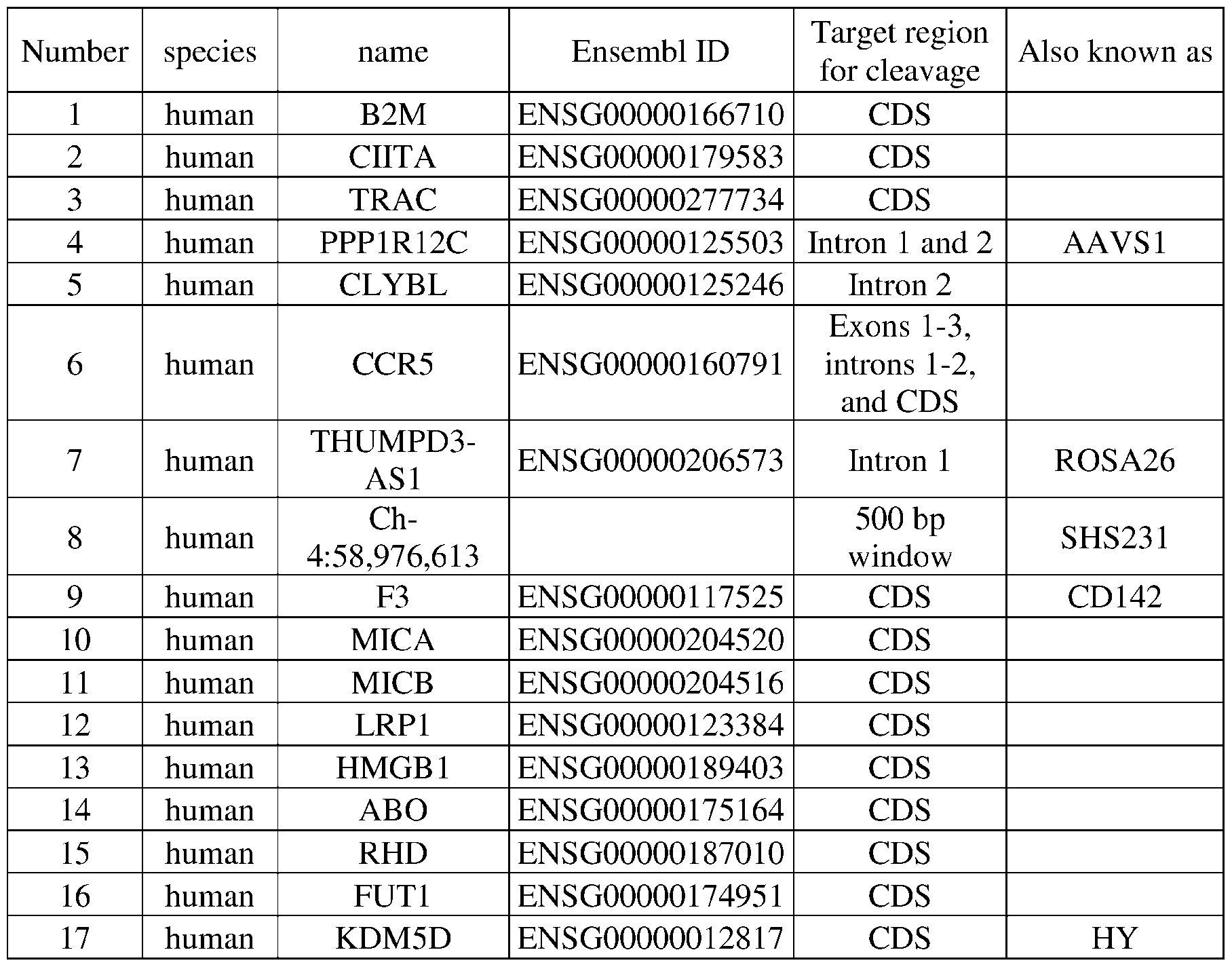
Type-1 diabetes autoimmune mouse Download PDFInfo
- Publication number
- WO2024151541A1 WO2024151541A1 PCT/US2024/010713 US2024010713W WO2024151541A1 WO 2024151541 A1 WO2024151541 A1 WO 2024151541A1 US 2024010713 W US2024010713 W US 2024010713W WO 2024151541 A1 WO2024151541 A1 WO 2024151541A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- cells
- human animal
- islet
- cell
- human
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
Classifications
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K67/00—Rearing or breeding animals, not otherwise provided for; New or modified breeds of animals
- A01K67/027—New or modified breeds of vertebrates
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/12—Materials from mammals; Compositions comprising non-specified tissues or cells; Compositions comprising non-embryonic stem cells; Genetically modified cells
- A61K35/37—Digestive system
- A61K35/39—Pancreas; Islets of Langerhans
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P3/00—Drugs for disorders of the metabolism
- A61P3/08—Drugs for disorders of the metabolism for glucose homeostasis
- A61P3/10—Drugs for disorders of the metabolism for glucose homeostasis for hyperglycaemia, e.g. antidiabetics
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
- C07K14/70503—Immunoglobulin superfamily
- C07K14/70539—MHC-molecules, e.g. HLA-molecules
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N5/00—Undifferentiated human, animal or plant cells, e.g. cell lines; Tissues; Cultivation or maintenance thereof; Culture media therefor
- C12N5/06—Animal cells or tissues; Human cells or tissues
- C12N5/0602—Vertebrate cells
- C12N5/0676—Pancreatic cells
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/5005—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells
- G01N33/5008—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving human or animal cells for testing or evaluating the effect of chemical or biological compounds, e.g. drugs, cosmetics
- G01N33/5082—Supracellular entities, e.g. tissue, organisms
- G01N33/5088—Supracellular entities, e.g. tissue, organisms of vertebrates
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N33/00—Investigating or analysing materials by specific methods not covered by groups G01N1/00 - G01N31/00
- G01N33/48—Biological material, e.g. blood, urine; Haemocytometers
- G01N33/50—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing
- G01N33/66—Chemical analysis of biological material, e.g. blood, urine; Testing involving biospecific ligand binding methods; Immunological testing involving blood sugars, e.g. galactose
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2207/00—Modified animals
- A01K2207/12—Animals modified by administration of exogenous cells
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2207/00—Modified animals
- A01K2207/15—Humanized animals
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2217/00—Genetically modified animals
- A01K2217/07—Animals genetically altered by homologous recombination
- A01K2217/075—Animals genetically altered by homologous recombination inducing loss of function, i.e. knock out
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2217/00—Genetically modified animals
- A01K2217/15—Animals comprising multiple alterations of the genome, by transgenesis or homologous recombination, e.g. obtained by cross-breeding
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2227/00—Animals characterised by species
- A01K2227/10—Mammal
- A01K2227/105—Murine
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2267/00—Animals characterised by purpose
- A01K2267/03—Animal model, e.g. for test or diseases
- A01K2267/035—Animal model for multifactorial diseases
- A01K2267/0362—Animal model for lipid/glucose metabolism, e.g. obesity, type-2 diabetes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/87—Introduction of foreign genetic material using processes not otherwise provided for, e.g. co-transformation
- C12N15/90—Stable introduction of foreign DNA into chromosome
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2800/00—Detection or diagnosis of diseases
- G01N2800/04—Endocrine or metabolic disorders
- G01N2800/042—Disorders of carbohydrate metabolism, e.g. diabetes, glucose metabolism
-
- G—PHYSICS
- G01—MEASURING; TESTING
- G01N—INVESTIGATING OR ANALYSING MATERIALS BY DETERMINING THEIR CHEMICAL OR PHYSICAL PROPERTIES
- G01N2800/00—Detection or diagnosis of diseases
- G01N2800/52—Predicting or monitoring the response to treatment, e.g. for selection of therapy based on assay results in personalised medicine; Prognosis
Definitions
- Type-1 diabetes also referred to as T1D, juvenile diabetes or insulin-dependent diabetes
- T1D juvenile diabetes
- insulin-dependent diabetes is a chronic condition that was reported to affect around 1 .6 million Americans in 2019.
- T1D pancreas makes little or no insulin, which is a hormone the body uses to allow sugar (e.g., glucose) to enter cells to produce energy. Different factors, such as genetics and some viruses, may cause T1D. Although T1D usually appears during childhood or adolescence, it can develop in adults.
- sugar e.g., glucose
- T1D has no cure. Rather, treatment for T1D is largely directed toward managing the amount of sugar in the blood of a patient using insulin, and adjusting diet and lifestyle to prevent complications.
- Insulin has been the predominant treatment for T1D, and has been used to treat patients with T1D for more than 100 years. Insulin has transformed the once-fatal diagnosis into a chronic, medically manageable condition (sec Sims, E.K., Carr, A.L.J., Oram, R.A., DiMeglio, L.A. & Evans-Molina, C. 100 years of insulin: celebrating the past, present and future of diabetes therapy. Nat Med 27, 1154- 1164 (2021), which is incorporated herein by reference in its entirety).
- T1D The risk for severe hypoglycemic events increases with the duration of T1D (see Pedersen-Bjergaard, U., et al. Severe hypoglycaemia in 1076 adult patients with type 1 diabetes: influence of risk markers and selection. Diabetes Metab Res Rev 20, 479-486 (2004), which is incorporated by reference herein).
- Continuous glucose monitoring has shown the ability to catch peaks or drops in glucose levels better than conventional fingerstick blood glucose meter checks.
- Continuous glucose monitoring has improved glycemic control in adults, as well as adolescents and young adults with T1D (see Juvenile Diabetes Research Loundation Continuous Glucose Monitoring Study, G., et al. Continuous glucose monitoring and intensive treatment of type 1 diabetes.
- Such wearable bioartificial devices provide glucose readings, trends, and alerts to the user in real time to inform diabetes treatment decisions (see Bekiari, E., et al. Artificial pancreas treatment for outpatients with type 1 diabetes: systematic review and meta-analysis. BMJ 361, kl310 (2016), which is incorporated herein by reference in its entirety). Nonetheless, compared to the physiological, immediate islet cell (e.g., beta islet cell) response, integrated closed-loop systems still face delays in subcutaneous glucose sensing and insulin delivery. In fact, patients experience an average of at least 30 minutes each day with hypoglycemia, e.g., with glucose less than 3.9 mmol/L (Boughton, C.K., et al.
- hypoglycemia e.g., with glucose less than 3.9 mmol/L
- Hybrid closed- loop glucose control with faster insulin aspart compared with standard insulin aspart in adults with type 1 diabetes A double-blind, multicentre, multinational, randomized, crossover study. Diabetes Obes Metab 23, 1389-1396 (2021), which is incorporated herein by reference in its entirety).
- the systems do not provide curative therapy of T1D.
- the systems also do not completely remove the burden of glucose uncertainty and unremitting daily self-management.
- Negative themes associated with the systems include technical difficulties, intrusiveness of alarms, and equipment size (see Barnard, K.D., et al. Psychosocial aspects of closed- and open-loop insulin delivery: closing the loop in adults with Type 1 diabetes in the home setting. Diabet Med 32, 601-608 (2015), which is incorporated herein by reference in its entirety).
- T1D One possible treatment for T1D is transplantation of allogeneic pancreatic donor islets to a patient who has T1D.
- Transplantation of allogeneic pancreatic donor islets has successfully been performed in selected patients, who have difficult-to-control insulin-dependent diabetes and impaired awareness of hypoglycemia (IAH).
- IAH insulin-dependent diabetes and impaired awareness of hypoglycemia
- Allogeneic islet transplantation replenishes the vanished beta cell population, and clinical trials in patients with IAH and history of severe hypoglycemic events showed that it provides glycemic control, restoration of hypoglycemia awareness, and protection from severe hypoglycemic events (see Brooks, A.M., et al. Attainment of metabolic goals in the integrated UK islet transplant program with locally isolated and transported preparations.
- the present disclosure provides technologies that will aid in the identification and/or characterization of islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) that can be transplanted in a subject suffering from T ID.
- islet cells e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells
- the present disclosure provides technologies that enable transplantation of islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) without the need for concomitant immunosuppression.
- the present disclosure provides non-human animals that can be used to identify and/or assess islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) for use in a subject (e.g., a human) for glycemic control.
- the present disclosure also describes non-human animals that can be used to identify and/or assess islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) survive, engraft, and/or ameliorate T1D.
- the present disclosure recognizes that additional therapeutic agents can be identified and/or assessed in non-human animals provided herein, and that such identification and/or assessment can be made separate from or concurrently with an identification and/or assessment of islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells).
- islet cells e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells.
- the present disclosure further provides the insight that non-human animals described herein can be useful in assessing an autoimmune response of a subject.
- autologous immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- PBMCs peripheral blood mononuclear cells
- islet cells derived from the subject can be transplanted to a non-human animal, where the interaction of such cells with each other and with other agents can be assessed.
- Use of autologous immune cells (e.g., PBMCs) and autologous islet cells from a particular subject allows for a subject specific analysis of autoimmunity, disease pathology, and disease etiology to be evaluated. Such information can be useful in determining how a particular subject may respond to certain therapies and provide an individualized prognostic information.
- the present disclosure provides a non-human animal comprising immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a subject and islet cells derived from cells of the subject.
- PBMCs peripheral blood mononuclear cells
- a subject is a human patient.
- a human patient has T1D.
- immune cells e.g., PBMCs
- islet cells are autologous.
- the present disclosure provides a non-human animal comprising immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with type 1 diabetes (T1D) and islet cells derived from cells of the human patient with T1D, wherein the immune cells (e.g., PBMCs) and the islet cells are autologous.
- a non- human animal is a non-human mammal.
- a non-human animal is a rodent.
- a non-human animal is a mouse.
- a non-human animal comprises one or more genetic modifications that are suitable for maintaining a xenograft.
- one or more genetic modifications comprise reducing or eliminating expression of one or more genes. In some embodiments, one or more genetic modifications comprise increasing expression of one or more genes. In some embodiments, one or more genetic modifications comprise reducing or eliminating expression of one or more genes and increasing expression of one or more genes.
- a mouse is a NOD mouse, optionally an NSG-SGM3 mouse.
- a mouse is a SCID mouse, a NOD/SCID mouse, a NOD/SCID/ IL-2Rg(null) mouse, a NOD.Cg-/We scid Z/2rg tmlwjl /SzJ mouse, a NOD.Cg- /r cld I12rg tmlwjl Tg(CMV-L3,CSF2,KITLG)lEav/MloySzJ (NSG-SGM3) mouse, a NSG-human IL3, NSG-SCF, NSG-GM-GSF mouse, a nude mouse, a ragl knockout mouse, or rag2 knockout mouse.
- a human patient with T1D is eligible for islet transplant therapy.
- a human patient with T1D exhibits one or more of the following: (a) a clinical history compatible with T1D onset of disease at younger than 40 years old; (b) insulin dependence for at least 5 years; (c) a sum of age and years of insulin dependent diabetes duration of at least 28 years; (d) absent stimulated C-peptide (less than 0.3 ng/mL) 60 and 90 minutes post-mixed-meal tolerance test; (e) involvement of intensive diabetes management, wherein intensive diabetes management comprises: (i) self-monitoring of glucose values no less than a mean of three times each day averaged over each week; (ii) administration of three or more insulin injections each day or insulin pump therapy; and/or (iii) under the direction of an endocrinologist, diabetologist, or diabetes specialist with at least three clinical evaluations during the past 12 months; (f) at least one episode of severe hypoglycemia in the past
- a human patient with T1D has had T1D for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 years, at least 8 years, at least 9 years, at least 10 years, at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 years, or at least 90 years.
- a human patient with T1D has impaired awareness of hypoglycemia (IAH).
- IAH comprises: a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more; a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more; marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more; or a composite of: a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile.
- LI liability index
- a human patient with T1D has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years.
- SHE severe hypoglycemic event
- a human patient with T1D has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2).
- antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 IDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
- JDFU juvenile diabetes foundation units
- antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 IDFU
- insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU
- antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU
- antibodies to ZnT8 are present at a level greater than or equal
- one or more of the immune cells comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
- one or more of the immune cells are isolated from peripheral blood mononuclear cells (PBMCs).
- PBMCs peripheral blood mononuclear cells
- one or more of the PBMCs comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
- one or more of the immune cells produce autoantibodies against cells or antigens of the human patient with T1D.
- one or more of the immune cells is a B cell.
- one or more of the immune cells produce antibodies against one or more islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2. In some embodiments, one or more of the immune cells produce islet cell antibodies (ICA) and/or insulin autoantibodies (IAA). In some embodiments, one or more of the immune cells is an islet-reactive T cell. In some embodiments, one or more of the immune cells are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal.
- ICA islet cell antibodies
- IAA insulin autoantibodies
- one or more of the immune cells is an islet-reactive T cell. In some embodiments, one or more of the immune cells are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal.
- a non-human animal comprises at least about 2.5xl0 5 , at least about 5xl0 5 , at least about IxlO 6 , at least about 2xl0 6 , at least about 3xl0 6 , at least about 4xl0 6 , at least about 5xl0 6 , at least about 6xl0 6 , at least about 7xl0 6 , at least about 8xl0 6 , at least about 9xl0 6 , at least about IxlO 7 , at least about 1.5xl0 7 , or at least about 2xl0 7 immune cells.
- a non-human animal comprises at least about 2.5xl0 5 , at least about 5xl0 5 , at least about IxlO 6 , at least about 2xl0 6 , at least about 3xl0 6 , at least about 4xl0 6 , at least about 5xl0 6 , at least about 6xl0 6 , at least about 7xl0 6 , at least about 8xl0 6 , at least about 9xl0 6 , at least about IxlO 7 , at least about 1.5xl0 7 , or at least about 2xl0 7 PBMCs.
- a non-human animal comprises reduced function of non- human islet cells relative to a comparable wild- type non-human animal. In some embodiments, a non-human animal comprises reduced activity of non-human islet cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal comprises a reduced number of non-human islet cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal does not comprise non-human islet cells. In some embodiments, a non-human animal comprises streptozotocin (STZ). In some embodiments, a non-human animal was previously administered STZ. In some embodiments, a non-human animal comprises or was previously administered alloxan.
- STZ streptozotocin
- a non-human animal comprises decreased expression of a PDX1 gene. In some embodiments, a non-human animal has reduced pancreatic function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced pancreatic activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has a reduced number of pancreatic cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal does not have a pancreas. In some embodiments, a non-human animal comprises splenocytes from an autoimmune and diabetic NOD/ShiLt mouse.
- a non-human animal has glucose levels greater than about 200 mg/dL, greater than about 300 mg/dL, greater than about 400 mg/dL, or greater than about
- a non-human animal has glucose levels greater than about
- a non-human animal has glucose levels greater than about
- a non-human animal has glucose levels greater than about
- islet cells are introduced via intramuscular injection, intravenous injection, intraportal injection, ocular injection, injection into omentum, and/or injection into a kidney capsule.
- islet cells are or comprise human alpha, beta, and/or delta islet cells. In some embodiments, islet cells are or comprise about 5%, about 10%, about 15%, about 20%, about 25%, about 35%, or about 40% alpha cells. In some embodiments, islet cells are or comprise about 10%, about 15%, about 20%, about 25%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, or about 80% beta cells. In some embodiments, islet cells are or comprise about 1%, about 2%, about 3%, about 4%, about 5%, about 6%, about 7%, about 8%, about 9%, about 10%, about 15%, about 20%, or about 25% delta cells.
- islet cells are or comprise differentiated cells.
- differentiated cells are derived from a stem cell.
- differentiated cells are derived from a pluripotent stem cell.
- differentiated cells are derived from an induced pluripotent stem cell (iPSC).
- iPSC induced pluripotent stem cell
- an iPSC is derived from a cell obtained from the human patient with T ID.
- a cell obtained from the human patient with T1D is selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell.
- a cell obtained from the human patient with T1D is a PBMC.
- a stem cell is derived from a cell obtained from the human patient with T ID.
- a stem cell is an induced pluripotent stem cell (iPSC) derived from a cell selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell.
- a stem cell is an induced pluripotent stem cell (iPSC) derived from a PBMC.
- differentiated cells are derived from a multipotent stem cell.
- a multipotent stem cell is derived from a cell obtained from the human patient with T1D.
- differentiated cells are derived from a lineage- specific stem cell.
- a lineage- specific stem cell is derived from a cell obtained from the human patient with T1D.
- differentiated cells are derived from a definitive endoderm cell.
- a definitive endoderm cell expresses FOXA2 and/or SOX 17.
- differentiated cells are derived from a primitive gut tube cell.
- a primitive gut tube cell expresses FOXA2 and/or SOX 17.
- differentiated cells are derived from a pancreatic progenitor cell.
- a pancreatic progenitor cell expresses PDX1 and/or NKX6-1.
- differentiated cells are derived from an endocrine cell.
- an endocrine cell expresses NKX6-1, NEUROG3, and/or NKX2-2.
- islet cells are or comprise engineered islet cells.
- engineered islet cells have been modified to be hypoimmune islet cells.
- hypoimmune islet cells comprise one or more modifications that (a) inactivate or disrupt one or more alleles of: (i) one or more major histocompatibility complex (MHC) class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or (ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules, and/or (b) increase expression of one or more tolerogenic factors, wherein the increased expression of (b) is relative to an islet cell that does not comprise the modifications.
- MHC major histocompatibility complex
- hypoimmune islet cells are (i) HLA class I-deficient and/or HLA class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells comprise one or more safety switches. In some embodiments, a non-human animal comprises an agent that activates the one or more safety switches. [00201 In some embodiments, islet cells produce insulin, c-peptide, glucagon, somatostatin, and/or pancreatic polypeptide.
- a non-human animal of the present disclosure comprises a first therapeutic agent.
- a first therapeutic agent is or comprises islet cells.
- a first therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid.
- a non-human animal of the present disclosure further comprised a second therapeutic agent.
- a second therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid.
- the present disclosure provides methods of characterizing human islet cells for delivery to a patient, comprising taking a sample of the islet cells derived from cells of the human patient with T1D and administering the sample to a non-human animal of the present disclosure.
- the present disclosure provides methods of making a non- human animal model of type 1 diabetes (T1D), the method comprising: (a) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to the non-human animal, (b) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous.
- a non-human animal is a non-human mammal.
- a non-human animal is a rodent.
- a non-human animal is a mouse.
- a non-human animal comprises one or more genetic modifications that are suitable for maintaining a xenograft.
- one or more genetic modifications comprise reducing or eliminating expression of one or more genes.
- one or more genetic modifications comprise increasing expression of one or more genes.
- one or more genetic modifications comprise reducing or eliminating expression of one or more genes and increasing expression of one or more genes.
- a mouse is a NOD mouse, optionally an NSG-SGM3 mouse.
- a mouse is a SCID mouse, a NOD/SCID mouse, a NOD/SCID/ IL-2Rg(null) mouse, a NOD.Cg-Prkdc scld Il2rg tml ⁇ /SzJ mouse, a NOD.Cg-Pr£e?c scld H2rg tmlw -’ 1 Tg(CMV- L3,CSF2,KITLG)lEav/MloySzJ (NSG-SGM3) mouse, a NSG-human IL3, NSG-SCF, NSG- GM-GSF mouse, a nude mouse, a ragl knockout mouse, or rag2 knockout mouse.
- a human patient with T1D is eligible for islet transplant therapy.
- a human patient exhibits one or more of the following: (a) a clinical history compatible with T1D onset of disease at younger than 40 years old; (b) insulin dependence for at least 5 years; (c) a sum of age and years of insulin dependent diabetes duration of at least 28 years; (d) absent stimulated C-peptide (less than 0.3 ng/mE) 60 and 90 minutes post-mixed-meal tolerance test; (e) involvement of intensive diabetes management, wherein intensive diabetes management comprises: (i) self-monitoring of glucose values no less than a mean of three times each day averaged over each week; (ii) administration of three or more insulin injections each day or insulin pump therapy; and/or (iii) under the direction of an endocrinologist, diabetologist, or diabetes specialist with at least three clinical evaluations during the past 12 months; (f) at least one episode of severe hypoglycemia in the past 12 months,
- a human patient with T1D has had T ID for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 years, at least 8 years, at least 9 years, at least 10 years at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 years, or at least 90 years.
- a human patient with T1D has impaired awareness of hypoglycemia (IAH).
- IAH comprises: a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more; a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more; marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more; or a composite of: a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile.
- LI liability index
- a human patient with T1D has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years.
- SHE severe hypoglycemic event
- a human patient with T1D has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2).
- antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
- JDFU juvenile diabetes foundation units
- glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU
- insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU
- antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU
- antibodies to ZnT8 are present at a level greater than or equal
- one or more of the immune cells comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
- one or more of the immune cells are isolated from peripheral blood mononuclear cells (PBMCs).
- PBMCs peripheral blood mononuclear cells
- one or more of the PBMCs comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
- one or more of the immune cells produce autoantibodies against cells or antigens of the human patient with T1D.
- one or more of the immune cells is a B cell.
- one or more of the immune cells produce antibodies against one or more islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2. In some embodiments, one or more of the immune cells produce islet cell antibodies (ICA) and/or insulin autoantibodies (IAA). In some embodiments, one or more of the immune cells is an islet-reactive T cell. In some embodiments, one or more of the immune cells are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal.
- ICA islet cell antibodies
- IAA insulin autoantibodies
- one or more of the immune cells is an islet-reactive T cell. In some embodiments, one or more of the immune cells are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal.
- a nonhuman animal of the present disclosure comprises at least 2.5 xlO 5 , at least 5xl0 5 , at least IxlO 6 , at least 2xl0 6 , at least 3xl0 6 , at least 4xl0 6 , at least 5xl0 6 , at least 6xl0 6 , at least 7xl0 6 , at least 8xl0 6 , at least 9xl0 6 , at least IxlO 7 , at least 1.5xl0 7 , or at least 2xl0 7 immune cells.
- a non-human animal comprises at least 2.5 xlO 5 , at least 5xl0 5 , at least I xlO 6 , at least 2xl0 6 , at least 3xl0 6 , at least 4xl0 6 , at least 5xl0 6 , at least 6xl0 6 , at least 7xl0 6 , at least 8xl0 6 , at least 9xl0 6 , at least IxlO 7 , at least 1.5xl0 7 , or at least 2xl0 7 PBMCs.
- islet cells are administered to the non-human animal via intramuscular injection, intravenous injection, intraportal injection, ocular injection, injection into omentum, and/or injection into a kidney capsule.
- islet cells are or comprise human alpha, beta, and/or delta islet cells. In some embodiments, islet cells are or comprise about 5%, about 10%, about 15%, about 20%, about 25%, about 35%, or about 40% alpha cells. In some embodiments, islet cells are or comprise about 10%, about 15%, about 20%, about 25%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, or about 80% beta cells. In some embodiments, islet cells are or comprise about 1%, about 2%, about 3%, about 4%, about 5%, about 6%, about 7%, about 8%, about 9%, about 10%, about 15%, about 20%, or about 25% delta cells.
- islet cells are or comprise differentiated cells.
- differentiated cells are derived from a stem cell.
- differentiated cells are derived from a pluripotent stem cell.
- differentiated cells are derived from an induced pluripotent stem cell (iPSC).
- iPSC induced pluripotent stem cell
- an iPSC is derived from a cell obtained from the human patient with T ID.
- a cell obtained from the human patient with T1D is selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell.
- a cell obtained from the human patient with T1D is a PBMC.
- a stem cell is derived from a cell obtained from a human patient with T1D.
- a stem cell is an induced pluripotent stem cell (iPSC) derived from a cell selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell.
- a stem cell is an induced pluripotent stem cell (iPSC) derived from a PBMC.
- differentiated cells are derived from a multipotent stem cell.
- a multipotent stem cell is derived from a cell obtained from the human patient with T1D.
- differentiated cells are derived from a lineage- specific stem cell.
- a lineage- specific stem cell is derived from a cell obtained from the human patient with T1D.
- differentiated cells are derived from a definitive endoderm cell.
- a definitive endoderm cell expresses FOXA2 and/or SOX17.
- differentiated cells are derived from a primitive gut tube cell.
- a primitive gut tube cell expresses FOXA2 and/or SOX 17.
- differentiated cells are derived from a pancreatic progenitor cell.
- a pancreatic progenitor cell expresses PDX1 and/or NKX6-1.
- differentiated cells are derived from an endocrine cell.
- an endocrine cell expresses NKX6-1, NEUROG3, and/or NKX2-2.
- islet cells are or comprise engineered islet cells.
- engineered islet cells have been modified to be hypoimmune islet cells.
- hypoimmune islet cells comprise one or more modifications, wherein the modifications (a) inactivate or disrupt one or more alleles of: (i) one or more major histocompatibility complex (MHC) class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or (ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules, and/or (b) increase expression of one or more tolerogenic factors, wherein the increased expression of (ii) is relative to an islet cell that does not comprise the modifications.
- MHC major histocompatibility complex
- one or more modifications reduce expression of the one or more MHC class I molecules and/or one or more molecules that regulate expression of the one or more MHC class 1 molecules, and/or reduce expression of the one or more MHC class 11 molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications.
- hypoimmune islet cells are (i) HLA class I-deficient and/or HLA class Il-deficient and (ii) overexpress CD47.
- hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47.
- hypoimmune islet cells comprise one or more safety switches.
- a non-human animal of the present disclosure comprises an agent that activates the one or more safety switches.
- islet cells produce insulin, glucagon, somatostatin, and/or pancreatic polypeptide.
- methods of the present disclosure further comprise a step of: (c) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal.
- reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises administering an islet-cell destroying composition to the non-human animal.
- an islet-cell destroying composition is or comprises STZ.
- an islet-cell destroying composition is or comprises alloxan.
- an islet-cell destroying composition is or comprises splenocytes from an autoimmune and diabetic NOD/ShiLt mouse.
- reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises removing the non-human animal’s pancreas.
- reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises decreasing expression of a PDX1 gene in the non-human animal.
- a non-human animal has reduced endogenous islet cell function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced endogenous islet cell activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced levels of endogenous islet cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal does not comprise endogenous islet cells.
- a non-human animal has glucose levels greater than about 200mg/dL, greater than about 300mg/dL, greater than about 400mg/dL, or greater than about 450mg/dL. In some embodiments, a non-human animal has glucose levels greater than about 300mg/dL. In some embodiments, a non-human animal has glucose levels greater than about 400mg/dL. In some embodiments, a non-human animal has glucose levels greater than about 450mg/dL.
- a method of the present disclosure comprises, in sequential order: (a) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from the human patient with T1D to the non-human animal, (c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, and (b) administering the islet cells derived from cells of the human patient with T1D to the non-human animal.
- immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- PBMCs peripheral blood mononuclear cells
- method of the present disclosure comprises, in sequential order: (c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (b) administering the islet cells derived from cells of the human patient with T1D to the non-human animal, and (a) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from the human patient with T1D to the non-human animal.
- PBMCs peripheral blood mononuclear cells
- method of the present disclosure comprises, in sequential order: (c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (a) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from the human patient with T1D to the non-human animal, and (b) administering islet cells derived from cells of the human patient with T1D to the non-human animal.
- immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- an islet-cell destroying composition is administered to the non-human animal for 5 continuous days, 4 continuous days, 3 continuous days, 2 continuous days, or 1 continuous day. In some embodiments, an islet-cell destroying composition is administered to the non-human animal 5 days, 4 days, 3 days, 2 days, or 1 day before the immune cells are administered to the non-human patient. In some embodiments, no more than 1 day, 2 days, 3 days, 4 days, or 5 days elapse between step (a) and step (b).
- the present disclosure provides methods comprising administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) and islet cells derived from cells of a human patient with type 1 diabetes (T1D) to a non-human animal, wherein the immune cells and the islet cells are autologous.
- the non- human animal comprises immune cells from a human patient with T1D.
- the present disclosure provides methods of using a non-human animal model of type 1 diabetes (T1D), the method comprising: (a) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to the non- human animal, (b) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous and (c) determining the survival and/or endocrine function of the islet cells.
- immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- PBMCs peripheral blood mononuclear cells
- hypoimmune islet cells comprise one or more modifications, wherein the modifications (a) inactivate or disrupt one or more alleles of: (i) one or more MHC class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or (ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules, and/or (b) increase expression of one or more tolerogenic factors, wherein the increased expression of (ii) is relative to an islet cell that does not comprise the modifications.
- one or more modifications reduce expression of the one or more MHC class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or reduce expression of the one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications.
- hypoimmune islet cells are (i) HLA class I-deficient and/or HLA class Il-deficient and (ii) overexpress CD47.
- hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47.
- hypoimmune islet cells comprise one or more safety switches.
- islet cells are a therapeutic agent.
- step (c) comprises determining the survival and/or endocrine function of the islet cells after an autoimmune challenge.
- a method of the present disclosure further comprises: (d) administering an agent to the non-human animal.
- an agent activates the one or more safety switches.
- step (c) further comprises evaluating effects of the one or more modifications on the hypoimmune islet cells.
- an agent is a therapeutic agent.
- a method of the present disclosure further comprises: (e) administering an immunosuppressant to the non- human animal.
- the present disclosure provides methods of characterizing one or more therapeutic agents, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells arc a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non- human animal, and (e) assessing the effect of the islet cells and/or the one or more therapeutic agents on one or more therapeutically relevant T1D measures.
- immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- PBMCs peripheral blood monon
- the present disclosure provides methods of characterizing one or more therapeutic agents, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (c) administering islet cells derived from cells of a human patient with T1D to the non-human animal, wherein the islet cells are a first therapeutic agent, (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, (d) optionally administering a second therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the one or more therapeutic agent on one or more therapeutically relevant T1D measures.
- PBMCs peripheral blood mononuclear cells
- the present disclosure provides methods of characterizing one or more therapeutic agents, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non- human animal, and (e) assessing the effect of the islet cells and/or the one or more therapeutic agent on one or more therapeutically relevant T1D measures.
- immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- PBMCs peripheral blood mononu
- the present disclosure provides methods of determining if one or more therapeutic agents are suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells derived from the human patient with T1D are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non- human animal, and (e) determining the suitability of the one or more therapeutic agents to make a T1D therapeutic by assessing the effect of the one
- immune cells e.
- the present disclosure provides methods of determining if one or more therapeutic agents are suitable for use in treating type 1 diabetes (T1D), the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells derived from the human patient with T1D are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non- human animal, and (e) determining the suitability of the one or more therapeutic agents to treat T1D by assessing the effect of the one or more therapeutic agents on one or
- immune cells e.
- the present disclosure provides methods of making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells derived from the human patient with T1D are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non-human animal, (e) determining the suitability of the first therapeutic agent, and optionally the second therapeutic agent, to make a T1D therapeutic by assessing the effect of the first therapeutic agent, and optionally the second
- immune cells e.
- the present disclosure provides methods of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous, (d) administering a therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
- immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- PBMCs peripheral blood mononuclear cells
- the present disclosure provides methods of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (c) administering islet cells derived from cells of a human patient with T1D to the non-human animal, (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, (d) administering a therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
- T1D candidate type 1 diabetes
- the present disclosure provides methods of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous, (d) administering a therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
- immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- PBMCs peripheral blood mononuclear cells
- the present disclosure provides methods of determining if a therapeutic agent is suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, (d) administering the therapeutic agent to the non-human animal, and (e) determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures.
- immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- the present disclosure provides methods of determining if a therapeutic agent is suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, (d) administering the therapeutic agent to the non-human animal, and (e) determining the suitability of the therapeutic agent to treat T1D by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures.
- immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- PBMCs
- the present disclosure provides methods of making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, (d) administering a therapeutic agent to the non-human animal, (e) determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures, and (f) making the T1D therapeutic.
- immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- one or more therapeutically relevant T1D measures comprise the survival and/or endocrine function of the islet cells. In some embodiments, one or more therapeutically relevant T1D measures comprise levels of insulin, glucagon, somatostatin, pancreatic polypeptide, C-peptide, and/or glucose in the non-human animal.
- the method further comprises a step of making a T1D therapeutic. In some embodiments, if a therapeutic agent is determined to be a suitable T1D therapeutic, the method further comprises a step of making a T1D therapeutic.
- a suitable T1D therapeutic comprises one or more of the following effects of the therapeutic agent on one or more therapeutically relevant T1D measures: (a) C-peptide levels greater than at least 100 pmol/L, at least 200 pmol/L, at least 300 pmol/L, at least 400 pmol/L, at least 500 pmol/L, at least 1000 pmol/L, at least 1500 pmol/L, or at least 1750 pmol/L, optionally wherein C-peptide levels are determined using 60 and 90 minutes post-mixed-meal tolerance test; (b) glucose levels lower than about 200mg/dL, lower than about 300mg/dL, lower than about 400mg/dL, or lower than about 450mg/dL; and/or (c) islet cells survive for at least 5 days, at least 10 days, at least 15 days, at least 20 days, at least 25 days, or at least 30 days.
- reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal comprises administering an islet-cell destroying composition to the non-human animal.
- an islet-cell destroying composition is or comprises STZ.
- an islet-cell destroying composition is or comprises splenocytes from an autoimmune and diabetic NOD/ShiLt mouse.
- reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal comprises removing the non-human animal’s pancreas.
- a non-human animal has reduced endogenous islet cell function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced endogenous islet cell activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced levels of endogenous islet cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal does not comprise endogenous islet cells.
- a second therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid.
- a therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid.
- a step of determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of islet cells and/or one or more therapeutic agents on one or more therapeutically relevant T1D measures comprises the use of two or more assays to characterize the islet cells and/or the one or more therapeutic agents and/or to determine the suitability of islet cells and/or the one or more therapeutic agents for making a T1D therapeutic.
- the present disclosure provides methods of treating a human patient, comprising administering a pharmaceutical composition comprising a T1D therapeutic made in a method of the present disclosure.
- a human patient has been diagnosed as having type 1 diabetes (T1D).
- T1D type 1 diabetes
- a human patient is eligible for islet transplant therapy.
- a human patient exhibits one or more of the following: (a) a clinical history compatible with T1D onset of disease at younger than 40 years old; (b) insulin dependence for at least 5 years; (c) a sum of age and years of insulin dependent diabetes duration of at least 28 years; (d) absent stimulated C-peptide (less than 0.3 ng/mL) 60 and 90 minutes post-mixed-meal tolerance test; (e) involvement of intensive diabetes management, wherein intensive diabetes management comprises: (i) self-monitoring of glucose values no less than a mean of three times each day averaged over each week; (ii) administration of three or more insulin injections each day or insulin pump therapy; and/or (iii) under the direction of an endocrinologist, diabetologist, or diabetes specialist with at least three clinical evaluations during the past 12 months; (f) at least one episode of severe hypoglycemia in the past 12 months, wherein an episode of severe hypoglycemia comprises an event with one or more of the following symptoms: (i) a clinical
- a human patient has had T1D for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 year’s, at least 8 year’s, at least 9 years, at least 10 years, at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 years, or at least 90 years.
- a human patient has impaired awareness of hypoglycemia (IAH).
- IAH comprises: a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more; a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more; marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more; or a composite of: a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile.
- LI liability index
- a human patient has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2).
- ICA beta islet cell cytoplasmic proteins
- GAD65 antibodies to glutamic acid decarboxylase
- IAA insulin autoantibodies
- ZnT8 antibodies against ZnT8
- proteins to protein tyrosine phosphatase e.g., IA-2
- antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
- JDFU juvenile diabetes foundation units
- glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU
- insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU
- antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU
- antibodies to ZnT8 are present at a level greater than or equal
- the present disclosure provides methods of monitoring a human patient treated with a pharmaceutical composition comprising a T1D therapeutic made in any one of the methods of the present disclosure, comprising assessing the effect of the T1D therapeutic on glucose levels.
- a non-human animal of the present disclosure or a method of the present disclosure comprises hypoimmune islet cells derived from induced pluripotent stem cells (iPSCs), wherein one or more of the modifications present in the hypoimmune islet cells are also present in the iPSCs.
- a non-human animal of the present disclosure or a method of the present disclosure comprises hypoimmune islet cells derived from induced pluripotent stem cells (iPSCs), wherein none of the modifications present in the hypoimmune islet cells are also present in the iPSCs.
- one or more of the immune cells comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
- one or more of the immune cells are isolated from peripheral blood mononuclear cells (PBMCs).
- PBMCs peripheral blood mononuclear cells
- one or more of the PBMCs comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
- FIG 1 shows an exemplary illustration of the process of removing human peripheral blood mononuclear cells (PBMCs) from a patient with type 1 diabetes (T1D) and engrafting a mouse with the PBMCs to create type 1 diabetes mice (T1D mice).
- PBMCs peripheral blood mononuclear cells
- T1D mice type 1 diabetes mice
- auto iPSCs autologous induced pluripotent stem cells
- iPSC-derived p-islet cells are generated and transplanted into the T1D mice.
- Some autologous iPSC-derived islet cells were additionally engineered to be hypoimmune (HIP autologous iPSC-derived islet cells).
- Figure 2 shows exemplary in vitro immunofluorescence staining of auto iPSC- derived and HIP iPSC-derived pseudo-islets (p-islets) for somatostatin, insulin, and glucagon (top two images) and for CD47, HLA class I, and DAPI (bottom two images).
- p-islets auto iPSC- derived and HIP iPSC-derived pseudo-islets
- Figure 3 shows exemplary graphs of HLA class I, HLA class II, and CD47 expression as assessed by flow cytometry in auto iPSC-derived p-islets (top row) and in HIP iPSC-derived p-islets (bottom row).
- Figure 4 shows an exemplary graph of the composition of cell types (e.g., alpha cells, beta cells, delta cells, and other cells) in auto iPSC-derived and HIP iPSC-derived p-islets as assessed by flow cytometry.
- cell types e.g., alpha cells, beta cells, delta cells, and other cells
- Figure 5 shows an exemplary graph of insulin release by in vitro human auto iPSC-derived and HIP iPSC-derived p-islets as assessed by ELISA (mean ⁇ SD, three independent replicates per group).
- Figure 6 shows an exemplary illustration of iPSC-derived p-islets transplantations.
- auto iPSC-derived p-islets were transplanted into in autologous T1D mice.
- HIP iPSC-derived p-islets were transplanted into autologous T1D mice, followed by subsequent transplantation of auto p-islets, via a second injection, on Day 15.
- survival and glucose control of the p-islets in the mice were monitored.
- Figure 7 shows exemplary in vivo bioluminescent images of three different batches of auto iPSC-derived p-islets transplanted into autologous T1D mice.
- Figure 8 shows an exemplary graph of bioluminescent signal from auto iPSC- derived p-islets transplanted into autologous T1D mice.
- Figure 9 shows an exemplary graph of fasting blood glucose levels and blood glucose levels after a 30 minute glucose challenge (mean ⁇ SD, 15 animals).
- Figure 10 shows an exemplary graph of results from an impedance killing assay with splenocytes and serum from T1D mice (that received auto p-islets) against auto iPSC- derived p-islet cells from the T1D mice (mean ⁇ SD, 5 animals).
- Figure 11 shows exemplary graphs of results from impedance killing assays using NK cells or macrophages as effector cells from T1D mice that received auto iPSC-derived p-islets against auto iPSC-derived p-islet cells from the T1D mice (mean ⁇ SD, 3 independent experiments).
- Figure 12 shows exemplary in vivo bioluminescent images of three different batches of HIP iPSC-derived p-islets transplanted into the right thigh muscle of autologous T1D mice. On Day 15, auto iPSC-derived p-islets were transplanted into the right thigh muscle.
- the BLI scale on the left side of the figure is from the HIP iPSC-derived p-islet imaging, the scale on the right from the auto p-islet imaging.
- Figure 13 shows an exemplary graph of bioluminescent signal from HIP iPSC- derived p-islets transplanted into autologous T1D mice.
- Figure 14 shows an exemplary graph of fasting blood glucose levels and blood glucose levels after a 30 minute glucose challenge (mean ⁇ SD, 15 animals).
- Figure 15 shows an exemplary graph of results from an impedance killing assay with splenocytes and serum from T1D mice (that received HIP p-islets) against HIP iPSC- derived p-islet cells from the T1D mice (mean ⁇ SD, 5 animals).
- Figure 16 shows exemplary graphs of results from impedance killing assays using NK cells or macrophages as effector cells from T1D mice that received HIP iPSC-derived p-islets against HIP iPSC-derived p-islet cells from the T1D mice (mean ⁇ SD, 3 independent experiments).
- Figure 17 shows an exemplary graph of c-peptide levels in mice 30 days after they received auto iPSC-derived p-islets or HIP iPSC-derived p-islets (mean ⁇ SD, 15 animals each).
- Administration includes the administration of a composition (e.g., an agent, e.g., cells, e.g., PBMCs or islet cells) to a subject or system (e.g., to a cell, organ, tissue, organism, or relevant component or set of components thereof).
- a composition e.g., an agent, e.g., cells, e.g., PBMCs or islet cells
- a subject or system e.g., to a cell, organ, tissue, organism, or relevant component or set of components thereof.
- route of administration may vary depending, for example, on the subject or system to which the composition is being administered, the nature of the composition, the purpose of the administration, etc.
- administration to an animal subject may be bronchial (including by bronchial instillation), buccal, enteral, interdermal, intra-arterial, intradermal, intragastric, intramedullary, intramuscular, intranasal, intraperitoneal, intrathecal, intravenous, intraventricular, mucosal, nasal, oral, rectal, subcutaneous, sublingual, topical, tracheal (including by intratracheal instillation), transdermal, vaginal and/or vitreal.
- administration to an animal subject may be performed via transplantation.
- islet cells may be administered via transplantation.
- islet cells e.g., autologous islet cells
- islet cells may be administered by intramuscular injection, intravenous injection, intraportal injection, ocular injection, injection into omentum, and/or injection into a kidney capsule.
- administration to an animal subject e.g., to a human or a rodent
- administration to an animal subject may be intraarterial or intravenous.
- peripheral blood mononuclear cells e.g., autologous PBMCs
- PBMCs peripheral blood mononuclear cells
- agent may refer to a compound, molecule, or entity of any chemical class (including, for example, a small molecule, polypeptide, nucleic acid, saccharide, lipid, metal, or a combination or complex thereof), system (e.g., cell, tissue, or organism), or a mixture.
- Allogeneic refers to an agent (e.g., compound, complex, cell, tissue, etc.) that is found in, originated from, and/or was obtained or derived (e.g., as progeny of an agent obtained from) from a different subject of the same species.
- an allogeneic agent is sufficiently different genetically such that it is capable of interacting antigenically.
- an allogeneic agent may be immunologically incompatible with a subject to which it is administered.
- Approximately and About as applied to one or more values of interest, includes to a value that is similar to a stated reference value.
- the term “approximately” or “about” refers to a range of values that fall within ⁇ 10% (greater than or less than) of the stated reference value unless otherwise stated or otherwise evident from the context (except where such number would exceed 100% of a possible value). In some embodiments, the term “approximately” or “about” refers to a range of values that would be rounded to the stated value based on the appropriate number of significant digits.
- Autoantibodies as used herein, the term “autoantibodies” is used to refer to antibodies produced by a subject in response to an agent from or derived from the subject.
- autoantibodies associated with T1D include islet cell autoantibodies (ICA), autoantibodies against insulin (IAA), autoantibodies against glutamic acid decarboxylase (GAA or GAD), autoantibodies against protein tyrosine phosphatase, or a combination thereof.
- Autoantigen is used to refer to an antigen produced by a subject, a subject’s cell or tissue, or a cell or tissue derived from a subject that are recognized by the immune system of that subject.
- an autoantigen is one whose recognition by the subject’s immune system is associated with an autoimmune disease, disorder or condition (e.g., T1D).
- an autoantigen may be or include any chemical entity such as, for example, a small molecule, a nucleic acid, a polypeptide, a carbohydrate, a lipid, etc.
- autoantigens associated with T1D include (but are not limited to) cytoplasmic proteins in an islet cell (e.g., a beta islet cell), glutamic acid decarboxylase (GAD-65), insulin, Zinc transporter 8 (ZnT8), and tyrosine phosphatase (IA2 or ICA512).
- cytoplasmic proteins in an islet cell e.g., a beta islet cell
- GAD-65 glutamic acid decarboxylase
- insulin Zinc transporter 8
- ZnT8 Zinc transporter 8
- IA2 or ICA512 tyrosine phosphatase
- Autoimmunity is used to refer to a collection of immune responses of a subject against an antigen produced by the subject, the subject’s cell or tissue, or a cell or tissue derived from the subject.
- Autologous refers to an agent (e.g., molecule, cell, or tissue) that is from or derives from the same individual to which the agent will be administered. Said differently, an autologous agent is obtained from a subject that is both the donor and recipient of the agent.
- agent e.g., molecule, cell, or tissue
- Biologically active refers to a characteristic of any agent that has activity in a biological system, in vitro or in vivo (e.g., in a subject). For instance, an agent that, when present in a subject, has a biological effect within that subject is considered to be biologically active. In particular embodiments, where an agent is biologically active, a portion of that agent that shares at least one biological activity of the agent is typically referred to as a “biologically active” portion.
- biological sample typically refers to a sample obtained or derived from a biological source (e.g., a tissue or organism or cell culture) of interest, as described herein.
- a source of interest comprises an organism, such as an animal or human.
- a biological sample is or comprises biological tissue or fluid.
- a biological sample may be or comprise bone marrow; blood; blood cells; ascites; tissue or fine needle biopsy samples; cellcontaining body fluids; free floating nucleic acids; sputum; saliva; urine; cerebrospinal fluid, peritoneal fluid; pleural fluid; feces; lymph; gynecological fluids; skin swabs; vaginal swabs; oral swabs; nasal swabs; washings or lavages such as a ductal lavages or broncheoalveolar lavages; aspirates; scrapings; bone marrow specimens; tissue biopsy specimens; surgical specimens; feces, other body fluids, secretions, and/or excretions; and/or cells therefrom, etc.
- a biological sample is or comprises cells obtained from an individual.
- obtained cells are or include cells from an individual from whom the sample is obtained.
- a sample is a “primary sample” obtained directly from a source of interest by any appropriate means.
- a primary biological sample is obtained by methods selected from the group consisting of biopsy (e.g., fine needle aspiration or tissue biopsy), surgery, collection of body fluid (e.g., blood, lymph, feces etc.), etc.
- sample refers to a preparation that is obtained by processing (e.g., by removing one or more components of and/or by adding one or more agents to) a primary sample. For example, filtering using a semi- permeable membrane.
- processing e.g., by removing one or more components of and/or by adding one or more agents to
- a primary sample For example, filtering using a semi- permeable membrane.
- Such a “processed sample” may comprise, for example nucleic acids or proteins extracted from a sample or obtained by subjecting a primary sample to techniques such as amplification or reverse transcription of mRNA, isolation and/or purification of certain components, etc.
- Comparable refers to two or more agents, situations, sets of conditions, etc. that may not be identical to one another but that are sufficiently similar to permit comparison there between so that conclusions may reasonably be drawn based on differences or similarities observed. Persons of ordinary skill in the art will understand, in context, what degree of identity is required in any given circumstance for two or more such agents, situations, sets of conditions, etc. to be considered comparable.
- Control refers to the art-understood meaning of a “control” being a standard against which results are compared. Typically, controls are used to augment integrity in experiments by isolating variables in order to make a conclusion about such variables.
- a control is a reaction or assay that is performed simultaneously with a test reaction or assay to provide a comparator.
- a “control” also includes a “control animal.”
- a “control animal” may have a modification as described herein, a modification that is different as described herein, or no modification (i.e., a wild-type animal).
- a "test" parameter e.g., a variable being tested
- a control is a historical control (i.e., of a test or assay performed previously, or an amount or result that is previously known).
- a control is or comprises a printed or otherwise saved record.
- a control may be a positive control or a negative control. In some embodiments, a control may be comparable.
- Detectable means a level that is detectable by standard techniques known to a skilled artisan, and include for example, differential display, RT (reverse transcriptasej-coupled polymerase chain reaction (PCR), Northern Blot, and/or RNase protection analyses as well as immunoaffinity-based methods for protein detection, such as flow cytometry, ELISA, or western blot.
- PCR reverse transcriptasej-coupled polymerase chain reaction
- Determining, measuring, evaluating, assessing, assaying and analyzing are used interchangeably herein to refer to any form of measurement, and include determining if an element is present or not. These terms include both quantitative and/or qualitative determinations. Assaying may be relative or absolute. “Assaying for the presence of” can be determining the amount of something present and/or determining whether or not it is present or absent.
- Engineered refers, in general, to the aspect of having been manipulated by the hand of man. Furthermore, as will be appreciated by persons of skill in the art, a variety of methodologies are available through which “engineering” as described herein may be achieved. For example, in some embodiments, “engineering” may involve selection or design (e.g., of nucleic acid sequences, polypeptide sequences, cells, tissues, and/or organisms) through use of computer systems programmed to perform analysis or comparison, or otherwise to analyze, recommend, and/or select sequences, alterations, etc.).
- “engineering” may involve use of in vitro chemical synthesis methodologies and/or recombinant nucleic acid technologies, mutation, transformation, transfection, etc., and/or any of a variety of controlled mating methodologies.
- in vitro chemical synthesis methodologies and/or recombinant nucleic acid technologies, mutation, transformation, transfection, etc., and/or any of a variety of controlled mating methodologies.
- a variety of established such techniques e.g., for recombinant DNA, oligonucleotide synthesis, tissue culture, transformation (e.g., electroporation, lipofection, etc.), transplantation
- transformation e.g., electroporation, lipofection, etc.
- expression refers to the conversion of the information, contained in a gene, into a gene product.
- a gene product can be the direct transcriptional product of a gene (e.g., mRNA, tRNA, rRNA, antisense RNA, ribozyme, structural RNA or any other type of RNA) or can be a protein produced by translation of an mRNA.
- Gene products also include RNAs which are modified, by processes such as capping, polyadenylation, methylation, and editing, and proteins modified by, for example, methylation, acetylation, phosphorylation, ubiquitination, ADP-ribosylation, myristoylation, and glycosylation.
- reference to expression or gene expression includes protein (or polypeptide) expression or expression of a transcribable product of or a gene such as mRNA.
- the protein expression may include intracellular expression or surface expression of a protein.
- expression of a gene product, such as mRNA or protein is at a level that is detectable in the cell.
- Function refers to a property and/or activity of a form or fragment of an agent (e.g., a biological agent, e.g., a cell, e.g., an islet cell or a PBMC).
- an agent e.g., a biological agent, e.g., a cell, e.g., an islet cell or a PBMC.
- Gene includes a DNA region encoding a gene product, as well as all DNA regions which regulate the production of the gene product, whether or not such regulatory sequences are adjacent to coding and/or transcribed sequences. Accordingly, a gene includes, but is not necessarily limited to, promoter sequences, terminators, translational regulatory sequences such as ribosome binding sites and internal ribosome entry sites, enhancers, silencers, insulators, boundary elements, replication origins, matrix attachment sites and locus control regions. The sequence of a gene is typically present at a fixed chromosomal position or locus on a chromosome in the cell.
- hypoimmunogenic refers to a cell that is less prone to immune rejection by a subject to which such cells are transplanted.
- a hypoimmunogenic cell may be about 2.5%, 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, 97.5%, 99% or more less prone to immune rejection by a subject into which such cells are transplanted.
- the hypoimmunogenic cells are allogenic to the subject and a hypoimmunogenic cell evades immune rejection in an MHC-mismatched allogeneic recipient.
- a hypoimmunogenic cell is protected from T cell-mediated adaptive immune rejection and/or innate immune cell rejection.
- hypoimmunogenicity as used herein, “hypoimmunogenicity” of a cell can be determined by evaluating the immunogenicity of the cell such as the cell’s ability to elicit adaptive and innate immune responses.
- Knock out includes deleting all or a portion of the target polynucleotide sequence in a way that interferes with the function of the target polynucleotide sequence.
- a knock out can be achieved by altering a target polynucleotide sequence by inducing an indel in the target polynucleotide sequence in a functional domain of the target polynucleotide sequence (e.g., a DNA binding domain).
- CRISPR/Cas systems can be used to knock out a target polynucleotide sequence or a portion thereof based upon the details described herein. Knocking out a target polynucleotide sequence or a portion thereof can be useful for a variety of applications. For example, knocking out a target polynucleotide sequence in a cell can be performed in vitro for research purposes.
- knocking out a target polynucleotide sequence in a cell can be useful for treating or preventing a disorder associated with expression of the target polynucleotide sequence (e.g., by knocking out a mutant allele in a cell ex vivo and introducing those cells comprising the knocked out mutant allele into a subject).
- Knock in refers to a process that adds a genetic function to a host cell.
- a knock in can cause increased levels of the knocked in gene product, e.g., an RNA or encoded protein. As will be appreciated by those in the art, this can be accomplished in several ways, including adding one or more additional copies of the gene to the host cell or altering a regulatory component of the endogenous gene increasing expression of the protein is made. This may be accomplished by modifying the promoter, adding a different promoter, adding an enhancer, or modifying other gene expression sequences.
- Modification refers to any change or alteration in a cell that impacts gene expression in the cell.
- the modification is a genetic modification that directly changes the gene or regulatory elements thereof encoding a protein product in a cell, such as by gene editing, mutagenesis or by genetic engineering of an exogenous polynucleotide or transgene.
- an alteration or modification described herein results in reduced expression of a target or selected polynucleotide sequence.
- an alteration or modification described herein results in reduced expression of a target or selected polypeptide sequence.
- an alteration or modification described herein results in increased expression of a target or selected polynucleotide sequence.
- an alteration or modification described herein results in increased expression of a target or selected polypeptide sequence.
- Non-human animal refers to any vertebrate organism that is not a human.
- a non-human animal is a cyclostome, a bony fish, a cartilaginous fish (e.g., a shark or a ray), an amphibian, a reptile, a mammal, and a bird.
- a non-human animal is a mammal.
- a non-human mammal is a primate, a goat, a sheep, a pig, a dog, a cow, or a rodent.
- a non- human animal is a rodent such as a rat or a mouse.
- a patient refers to any organism to which a provided composition is or may be administered, e.g., for experimental, diagnostic, prophylactic, cosmetic, and/or therapeutic purposes. Typical patients include animals (e.g., mammals such as mice, rats, rabbits, non-human primates, and/or humans). In some embodiments, a patient is a human. In some embodiments, a patient is suffering from or susceptible to one or more disorders or conditions (e.g., T1D). In some embodiments, a patient displays one or more symptoms of a disorder or condition (e.g., T1D). In some embodiments, a patient has been diagnosed with one or more disorders or conditions (e.g., T1D).
- animals e.g., mammals such as mice, rats, rabbits, non-human primates, and/or humans.
- a patient is a human.
- a patient is suffering from or susceptible to one or more disorders or conditions (e.g., T1D).
- a patient displays one or more symptoms of
- polypeptide As used herein refers to a polymeric chain of amino acids.
- a polypeptide is a protein.
- a polypeptide has an amino acid sequence that occurs in nature.
- a polypeptide has an amino acid sequence that does not occur in nature.
- a polypeptide has an amino acid sequence that is engineered in that it is designed and/or produced through action of the hand of man.
- a polypeptide may comprise or consist of natural amino acids, non-natural amino acids, or both.
- a polypeptide may comprise or consist of only natural amino acids or only non-natural amino acids.
- a polypeptide may comprise D-amino acids, L-amino acids, or both.
- a polypeptide may comprise only D-amino acids.
- a polypeptide may comprise only L-amino acids.
- a polypeptide may include one or more pendant groups or other modifications, e.g., modifying or attached to one or more amino acid side chains, at the polypeptide’s N-terminus, at the polypeptide’s C-terminus, or any combination thereof.
- such pendant groups or modifications may be selected from the group consisting of acetylation, amidation, lipidation, methylation, pegylation, etc., including combinations thereof.
- a polypeptide may be cyclic, and/or may comprise a cyclic portion. In some embodiments, a polypeptide is not cyclic and/or does not comprise any cyclic portion. In some embodiments, a polypeptide is linear. In some embodiments, a polypeptide may be or comprise a stapled polypeptide.
- polypeptide may be appended to a name of a reference polypeptide, activity, or structure; in such instances it is used herein to refer to polypeptides that share the relevant activity or structure and thus can be considered to be members of the same class or family of polypeptides.
- the present specification provides and/or those skilled in the art will be aware of exemplary polypeptides within the class whose amino acid sequences and/or functions are known; in some embodiments, such exemplary polypeptides are reference polypeptides for the polypeptide class or family.
- a member of a polypeptide class or family shows significant sequence homology or identity with, shares a common sequence motif (e.g., a characteristic sequence element) with, and/or shares a common activity (in some embodiments at a comparable level or within a designated range) with a reference polypeptide of the class; in some embodiments with all polypeptides within the class).
- a common sequence motif e.g., a characteristic sequence element
- shares a common activity in some embodiments at a comparable level or within a designated range
- a member polypeptide shows an overall degree of sequence homology or identity with a reference polypeptide that is at least about 30-40%, and is often greater than about 50%, 60%, 70%, 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or more and/or includes at least one region (e.g., a conserved region that may in some embodiments be or comprise a characteristic sequence element) that shows very high sequence identity, often greater than 90% or even 95%, 96%, 97%, 98%, or 99%.
- a conserved region that may in some embodiments be or comprise a characteristic sequence element
- Such a conserved region usually encompasses at least 3-4 and often up to 20 or more amino acids; in some embodiments, a conserved region encompasses at least one stretch of at least 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15 or more contiguous amino acids.
- a relevant polypeptide may comprise or consist of a fragment of a parent polypeptide.
- a useful polypeptide as may comprise or consist of a plurality of fragments, each of which is found in the same parent polypeptide in a different spatial arrangement relative to one another than is found in the polypeptide of interest (e.g., fragments that are directly linked in the parent may be spatially separated in the polypeptide of interest or vice versa, and/or fragments may be present in a different order in the polypeptide of interest than in the parent), so that the polypeptide of interest is a derivative of its parent polypeptide.
- Prognostic and predictive information are used to refer to any information that may be used to indicate any aspect of the course of a disease or condition (e.g., T1D) either in the absence or presence of treatment. Such information may include, but is not limited to, the average life expectancy of a patient, the likelihood that a patient will survive for a given amount of time (e.g., 6 months, 1 year, 5 years, etc.), the likelihood that a patient will be cured of a disease, the likelihood that a patient’ s disease will respond to a particular therapy (wherein response may be defined in any of a variety of ways).
- Subject refers an organism, typically a mammal (e.g., a human). In some embodiments, a subject is suffering from a relevant disease, disorder or condition (e.g., T1D). In some embodiments, a subject is a patient.
- a relevant disease, disorder or condition e.g., T1D
- a subject is a patient.
- Substantially refers to the qualitative condition of exhibiting total or near-total extent or degree of a characteristic or property of interest.
- One of ordinary skill in the biological arts will understand that biological and chemical phenomena rarely, if ever, go to completion and/or proceed to completeness or achieve or avoid an absolute result.
- the term “substantially” is therefore used herein to capture the potential lack of completeness inherent in many biological and chemical phenomena.
- Therapeutic agent refers to an agent that, when administered to a subject, has a therapeutic effect and/or elicits a desired biological and/or pharmacological effect.
- a therapeutic agent is any substance that can be used to alleviate, ameliorate, relieve, inhibit, prevent, delay onset of, reduce severity of, and/or reduce incidence of one or more symptoms or features of a disease, disorder, and/or condition (e.g., T1D).
- Tolerogenic factor includes immunosuppressive factors or immune-regulatory factors that modulate or affect the ability of a cell to be recognized by the immune system of a host or recipient subject upon administration, transplantation, or engraftment.
- a tolerogenic factor is a factor that induces immunological tolerance to an engineered primary cell so that the engineered primary cell is not targeted, such as rejected, by the host immune system of a recipient.
- a tolerogenic factor may be a hypoimmunity factor.
- Examples of tolerogenic factors include immune cell inhibitory receptors (e.g. CD47), proteins that engage immune cell inhibitory receptors, checkpoint inhibitors and other molecules that reduce innate or adaptive immune recognition.
- treatment refers to administration of a therapy that partially or completely alleviates, ameliorates, relives, inhibits, delays onset of, reduces severity of, and/or reduces incidence of one or more symptoms, features, and/or causes of a particular disease, disorder, and/or condition (e.g., T1D).
- treatment may be prophylactic; in some embodiments, treatment may be therapeutic.
- Wild-type refers to an entity having a structure and/or activity as found in nature in a “normal” (as contrasted with mutant, diseased, altered, engineered, etc.) state or context.
- T1D cure and/or T1D treatments there remains a need in the art for a T1D cure and/or T1D treatments. Part of the reason such a need remains is the lack of technologies available for sufficiently identifying and/or assessing the potential T1D cures and treatment. While T1D models exist, such models are not useful for robust characterization of T1D and the underlying mechanisms that lead to T1D. Thus, a need for technologies useful for identifying and characterizing possible T1D cures and/or T1D treatments also remains in the ail. The present disclosure addresses the need for technologies that can be utilized in understanding an individual patient’s T1D etiology and/or pathogenesis.
- the present disclosure provides technologies that will aid in the identification and/or characterization of islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) that can be transplanted in a subject suffering from T1D.
- islet cells e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells
- the present disclosure provides technologies that enable transplantation of islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) without the need for concomitant immunosuppression.
- the present disclosure provides non-human animals that can be used to identify and/or assess islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) for use in a subject (e.g., a human) for glycemic control.
- the present disclosure also describes non-human animals that can be used to identify and/or assess islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) survive, engraft, and/or ameliorate T1D.
- the present disclosure recognizes that additional therapeutic agents can be identified and/or assessed in non-human animals provided herein, and that such identification and/or assessment can be made separate from or concurrently with an identification and/or assessment of islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells).
- islet cells e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells.
- the present disclosure further provides the insight that non-human animals described herein can be useful in assessing an autoimmune response of a subject.
- PBMCs peripheral blood mononuclear cells
- islet cells derived from the subject can be transplanted to a non- human animal, where the interaction of such cells with each other and with other agents can be assessed.
- Use of autologous PBMCs and autologous islet cells from a particular subject allows for a subject specific analysis of autoimmunity, disease pathology, and disease etiology to be evaluated. Such information can be useful in determining how a particular subject may respond to certain therapies and provide an individualized prognostic information.
- the present disclosure provides a non-human animal comprising peripheral blood mononuclear cells (PBMCs) from a subject and islet cells derived from cells of the subject.
- PBMCs peripheral blood mononuclear cells
- a subject is a human patient.
- a human patient has T1D.
- PBMCs are autologous.
- islet cells are autologous.
- T1D Type-1 Diabetes
- the present disclosure provides technologies that allow for the study of autoimmune diseases.
- the present disclosure provides the recognition that the interaction between immune system cells (e.g., PBMCs or cells derived from PBMCs) from a subject (e.g., a mammal, e.g., a human) and other cells or tissues from the same subject can be assessed in a different non-human animal (e.g., a mammal, a rodent, e.g., a mouse or rat).
- a subject e.g., a mammal, e.g., a human
- a rodent e.g., a mouse or rat
- Non-human animals e.g., a mammal, e.g., a rodent, e.g., a mouse or rat
- a non-human animal comprising peripheral blood mononuclear cells (PBMCs) from a subject (e.g., a mammal, e.g., a human) and islet cells derived from cells of the subject.
- PBMCs peripheral blood mononuclear cells
- T1D also referred to Type-1 diabetes mellitus, T1DM, or insulin-dependent diabetes mellitus
- T1DM Type-1 diabetes mellitus
- T1D is a chronic disease characterized by hyperglycemia secondary to an insulin deficiency.
- T1D affects approximately 20 million people worldwide, and accounts for 5-10% of all cases of all diabetes disorders.
- T1D is most common in childhood and adolescence, T ID can present at any age.
- T1D is caused via an autoimmune response of insulin-producing -cells in the pancreatic islets of Langerhans. Because a T1D subject produces little to no insulin, the subject cannot properly process glucose and glucose build up in the subject’s blood. The increased level of glucose has metabolic consequences, including (but not limited to) weight loss, polyuria, polydipsia, and ketoacidosis.
- Insulitis is the term for the infiltration of the pancreatic islets by immune cells, which are predominantly T and B lymphocytes, macrophages and dendritic cells. Of these immune cells, T cells can play an important role in initiating the autoimmune process and it has been shown that transfer of a single T cell clone can cause T1D in animal models of T1D. A normal immune system eliminates islet-reactive T cells or control their activity; however, the immune systems of subjects with T1D lack this ability.
- immune cells are predominantly T and B lymphocytes, macrophages and dendritic cells.
- T cells can play an important role in initiating the autoimmune process and it has been shown that transfer of a single T cell clone can cause T1D in animal models of T1D.
- a normal immune system eliminates islet-reactive T cells or control their activity; however, the immune systems of subjects with T1D lack this ability.
- autoimmunity to [3-islet cells can be mediated by autoantibodies produced by a subject, which are directed to proteins present on [3-islet cells. Autoantibodies are frequently directed to glutamic acid decarboxylase and/or insulin, but other autoantibody targets are known, as shown in Table 1 below.
- T1D can be diagnosed in a subject by measuring circulating autoantibodies directed to, e.g., insulin, GAD65, ZnT8, ICA512, and/or IA-2.
- circulating autoantibodies directed to e.g., insulin, GAD65, ZnT8, ICA512, and/or IA-2.
- GAD glutamic acid decarboxylase
- children tend to have higher levels of insulin autoantibodies.
- Not all subjects with T1D are antibody-positive, however.
- a positive measure of autoantibodies can be helpful for diagnosing T1D, a negative measure is not sufficient to exclude a diagnosis of T1D.
- Measuring autoantibodies can be helpful because autoantibodies develop prior to the onset of T1D, and therefore can be useful to predict a diagnosis of T1D.
- Assays for T1D autoantibodies are known in the art.
- T1D presents in subjects without a family history, but genetics is understood to play a role in the development of T1D. Only 10-15% of the T1D patients have a first- or second-degree relative with the disease. However, the lifetime risk for developing T1D is increased for relatives of T1D patients, as about 6% of children, 5% of siblings and 50% of monozygotic twins of T1D patients develop T1D compared to 0.4% prevalence of the general population.
- T1D genetic risk loci More than 50 T1D genetic risk loci have been identified by genome-wide association studies and meta-analyses.
- MHC major histocompatibility complex
- HLA human leucocyte antigen
- HLA polymorphic alleles are understood to be responsible for 40-50% of the genetic risk of T1D development.
- Insulin gene Ins-VNTR, IDDM 2
- CTL-4 cytotoxic T lymphocyte-associated antigen-4 gene
- MHC is a group of closely related genetic loci encoding molecules expressed on the surface of immune or other cells. MHC molecules can limit the specificity of T lymphocytes to recognize antigens. HLA genes are distinguished into two classes. MHC class I (MHC-I) molecules are typically associated with antigens recognized by the receptor of cytotoxic T lymphocytes (CD8+), which eventually destroy the antigenic target and are expressed by most cells. MHC class II (MHC -II) molecules are essential for the recognition of antigens by T helper lymphocytes (CD4+), which initiate the immune response and promote cellular co-operation and are expressed mostly by immune cells.
- MHC-I cytotoxic T lymphocytes
- CD4+ T helper lymphocytes
- MHC-II genes which is similar to other autoimmune diseases.
- the ability of MHC-II molecules to present antigens is dependent on their alpha- and beta-chain amino acid composition. Substitutions at one or two sites can impact the binding capacity of the relevant autoantigens.
- more than 90% of T1D patients have either HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotypes as compared to 40% of the healthy individuals.
- about 30% of the patients have both haplotypes (DR3/4 heterozygotes).
- DRB 1 *0403 and DPB 1 *0402 reduces the risk of T1 D development even in the presence of DQB 1*0302 high-risk allele.
- HLA allele DQB 1*0602 also seems to provide protection against T1D development. This allele is present in approximately 20% of the general US population but only in 1% of the children who have developed T1D.
- a subject as described herein can have a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
- a subject has a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) and HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
- a PBMC as described herein can have a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
- a PBMC has a HLA-DR3, DQBl*0201 (also referred as DR3-DQ2) and HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
- VNTRs variable numbers of tandem repeats
- Two VNTR classes are associated with T1D risk: the small class (26-63 repeats, type I) or the big class (140- 243 repeats, type III).
- Class II VNTRs have between 63-140 repeats and is not understood to impact T1D susceptibility.
- Type III is present in approximately 30% of the general population and provides protection from T ID.
- the presence of a big class allele facilitates transcription and epithelial expression of insulin. Accordingly, the autoantigen presentation of insulin in the fetal thymus and is protective by promoting the negative selection and deletion of T lymphocytes reactive to insulin that contribute to the autoimmune destruction of 0 cells. Type 1 is associated with a susceptibility to T1D.
- the present disclosure provides that a subject as described herein can comprise a class I VNTR at an insulin gene promoter.
- a PBMC as described herein can comprise a class I VNTR at an insulin gene promoter.
- the cytotoxic T lymphocyte antigen-4 (CTLA-4) gene is located on the long arm of chromosome 2 (2q33) and has also been associated with T1D. It has a suppressive role in T- cell activation, and specifically, a role of transmitting negative signal and inducing anergy in activated T-cell.
- Identification of an antigen by a naive T-cell is achieved through a triple complex consisting of an antigen, an HLA molecule of the antigen presenting cell (APC) and a T-cell antigen receptor.
- APC antigen presenting cell
- T-cell antigen receptor a triple complex consisting of an antigen, an HLA molecule of the antigen presenting cell (APC) and a T-cell antigen receptor.
- APC antigen presenting cell
- T-cell antigen receptor T-cell antigen receptor
- Such molecules include the B7.1 (CD80) and B7.2 (CD86) glycoproteins on APCs and a CD28 receptor, a member of the immunoglobulin superfamily, on T-cells.
- a T-cell is not stimulated and switches to the anergy state.
- An active T-cell further expresses the CTLA-4 receptor, which resembles the sequence of CD28, but has 20-fold greater affinity for B7 glycoproteins.
- a CTLA-4 receptor when linked to one of the B7 (CD80/CD86) molecules of an APC, transmits negative messages to a T-cell. This limits the production of IL-2 and the proliferation of stimulated cells. As a result, excessive stimulation and proliferation of T-cell are avoided.
- CTLA-4 Polymorphisms of CTLA-4 have been associated with T1D and with other autoimmune diseases. These polymorphisms tend to lead to decreased intracellular expression of CTLA-4. Consequently, inhibition of excessive stimulation and proliferation of T-cell is not inhibited resulting in uncontrolled progression of immune response and autoimmune imbalance.
- a subject as described herein can comprise a polymorphism within a CTLA-4 gene that results in a decreased level of CTLA-4 expression as compared to level of CTLA-4 expression observed with a comparable subject that does not have the polymorphism.
- a cell as described herein can comprise a polymorphism within a CTLA-4 gene that results in a decreased level of CTLA-4 expression as compared to level of CTLA-4 expression observed with a comparable cell that does not have the polymorphism.
- the protein tyrosine phosphatase non-receptor type 22 (PTPN22) gene encodes the lymphoid tyrosyl phosphatase known as LYP.
- LYP is a potent inhibitor of the activation of naive T-cells, preventing spontaneous T-cell activation by dephosphorylating and inactivating a kinase, named CSK.
- CSK a kinase
- a missense mutation (C1858T (Arg620Trp) polymorphism) in the PTPN22 gene was found to be associated with various autoimmune diseases, including T1D. This mutation decreases the binding affinity of LYP to CSK, leading to uncontrolled T-cell activation and autoimmune reaction.
- a subject as described herein can comprise a C1858T (Arg620Trp) polymorphism in a PTPN22 gene.
- a PBMC as described herein can comprise a C1858T (Arg620Trp) polymorphism in a PTPN22 gene.
- AIRE autoimmune regulator
- APS 1 autoimmune polyendocrine syndrome 1
- the present disclosure provides that a subject as described herein can comprise one or more mutations in an AIRE gene.
- the present disclosure also provides that a PBMC as described herein can comprise one or more mutations in an AIRE gene.
- expression of AIRE in a subject or a PBMC with one or more mutations in an AIRE gene may be decreased compared to a subject or PBMC without the one or more mutations.
- the gene of the transcription factor FoxP3 is located on chromosome X and encodes for a protein known as scurfin.
- the congenital dysfunction of FoxP3 is manifested by an autoimmune phenotype.
- congenital dysfunction of FoxP3 appears as an autoimmune polyendocrine disorder associated with chromosome X (IPEX syndrome) and is manifested with a milder phenotype comprising, e.g., T1D, allergies, enteropathies and eczema.
- FoxP3 transcription factor is the hallmark of T regulatory lymphocytes (Tregs).
- Tregs suppress the proliferation and secretory activity of CD4+ and CD8+ T cells, as well as the cytolytic activity of the latter.
- a subject as described herein can comprise one or more mutations in a FoxP3 gene.
- a PBMC as described herein can comprise one or more mutations in a FoxP3 gene.
- expression of scurfin in a subject or a PBMC with one or more mutations in a FoxP3 gene may be decreased compared to a subject or PBMC without the one or more mutations.
- IL-12 produced by activated macrophages and dendritic cells induces Thl differentiation by activation of the transcription factors STAT1, STAT3 and STAT4.
- Mutations in the STAT3 transcription factor have been identified as monogenic causes of autoimmune diseases. De novo activating mutations of STAT3 are associated with an early onset spectrum of autoimmune diseases, such as T1D and autoimmune thyroid dysfunction.
- the present disclosure provides that a subject as described herein can comprise one or more mutations in a STAT3 gene.
- a PBMC as described herein can comprise one or more mutations in a STAT3 gene.
- expression of STAT3 in a subject or a PBMC with one or more mutations in a STAT3 gene may be decreased compared to a subject or PBMC without the one or more mutations.
- the interferon-induced helicase C domain 1 (IFIH1) gene is located on Chr2q24.2. This gene encodes the melanoma differentiation-associated protein 5 (MDA5) protein, a cytoplasmic sensor of viral double- stranded RNA, that activates a cascade of anti-viral immune responses. Rare variants of IFIH1 through a lost or reduced expression of the protein are protective against T1D, whereas common IFIH1 single nuclear polymorphisms (SNPs) are associated with the disease.
- MDA5 melanoma differentiation-associated protein 5
- SNPs single nuclear polymorphisms
- a subject as described herein can comprise one or more mutations in an IFIH1 gene.
- a PBMC as described herein can comprise one or more mutations in an IFIH1 gene.
- expression of MDA5 in a subject or a PBMC with one or more mutations in an IFIH1 gene may be decreased compared to a subject or PBMC without the one or more mutations.
- the Huntingtin-interacting protein 14 gene is located on chromosome 12 and encodes for a palmitoyl transferase, which is involved in the metabolism and trafficking of proteins met with neurons. Knockdown of the genes in mice revealed increased apoptosis for pancreatic 0 cells. On the other hand, increased expression of HIP 14 was protected against apoptosis and this was in association with reduced nuclear-factor kappa B (NFKB) activity.
- NFKB nuclear-factor kappa B
- the present disclosure provides that a subject as described herein can comprise one or more mutations in an HIP14 gene.
- a PBMC as described herein can comprise one or more mutations in an HIP14 gene.
- expression of HIP14 in a subject or a PBMC with one or more mutations in an HIP14 gene may be decreased compared to a subject or PBMC without the one or more mutations.
- ERBB3 Erb-B2 receptor tyrosine kinase 3
- SNPs SNPs (especially the rs2292239) have been associated with the presence of T ID, as well as the glycemic control of patients with the disease.
- the present disclosure provides that a subject as described herein can comprise one or more mutations in an ERBB3 gene.
- a PBMC as described herein can comprise one or more mutations in an ERBB3 gene.
- expression of ERBB3 in a subject or a PBMC with one or more mutations in an ERBB3 gene may be decreased compared to a subject or PBMC without the one or more mutations.
- Environmental factors also play an important role in the pathogenesis of T1D.
- Evidence for the role of environmental factors in T1D derives from the study of monozygotic twins, where occurrence of the disease in both siblings varies around 50% and never reaches 100%.
- Relevant environmental factors involved include viruses (e.g., rubella, coxsackievirus B, or enteroviruses), toxins, and nutrients (e.g., cow’s milk, cereals).
- Viruses can trigger T1D pathogenesis. Children exposed during fetal life to rubella have an increased incidence of T1D (and other autoimmune disorders, such as autoimmune thyroiditis). Furthermore, RNA or proteins from such viruses have been detected in the pancreas and other neighboring tissues from patients with T1D. Enteroviruses can play an important role in the early phase of the development of T1D through the activation of innate immunity. There is also a link between T1D and the interferon-induced helicase 1 (IFIH1) gene, which encodes for the MDA5, as discussed above. Upon viral infection, MDA5 acts as a cytoplasmic sensor especially for coxsackieviruses B and stimulates the production of molecular mediators of an innate immune response.
- IFIH1 interferon-induced helicase 1
- Another mechanism by which the autoimmune response can be induced by viruses is that of molecular mimicry.
- molecular mimicry suggests that the immune response is directed against autoantigens that resemble viral antigens, which results in cellular destruction.
- the best studied paradigm of molecular mimicry is the P2-C protein of the Coxsackie B4 virus.
- GID65 glutamic acid decarboxylase
- T-cell autoactivation after Coxsackie virus infection which leads to inflammation, tissue damage and release of islet autoantigens can further compound issues and increase T1D risk.
- the present disclosure provides that a subject as described herein has been exposed to coxsackieviruses strains B4, Bl, A4, A2, and/or A16.
- the present disclosure provides that a subject as described herein has been exposed to an enterovirus.
- the present disclosure also provides that a PBMC as described herein can be obtained from a subject that has been exposed to coxsackieviruses strains B4, Bl, A4, A2, and/or A 16.
- the present disclosure also provides that a PBMC as described herein can be obtained from a subject that has been exposed an enterovirus.
- gut microbiota the microflora of trillions of microorganisms living in the gastrointestinal tract, may be involved in the pathogenesis of T1D.
- Patients with T1D exhibit differences in their gut microbiota compared to healthy controls, specifically a reduced Firmicutes:Bacteroidetes ratio. Whether the changes in the composition of the gut microbiota derive from environmental factors or T1D itself is still being investigated.
- the birthing process may play a role in the microbiota and its role in T1D, as suggested by the increased risk of T1D in children born by cesarean section. Lack of contact with the vaginal microbiome of the mother and subsequent differences in their gut microbiota due to abnormal colonization may be a pathogenic mechanism and indicate the possible protective role of normal labor and vaginal delivery.
- the present disclosure provides that a subject as described herein may produce antibodies ABBOS.
- the present disclosure also provides that a subject as described herein may have a microbiome with a reduced Firmicutes:Bacteroidetes ratio as compared to a comparable subject that has not been diagnosed with an autoimmune disorder (e.g., T1D).
- the present disclosure also provides that a subject as described herein may have been birthed via cesarean section. iii. Immunologic factors
- the immune systems of mammals are exposed to large numbers of antigens, including autoantigens. Part of the purpose of the immune system is to distinguish foreign antigens from autoantignes. Immune system repertoires are extremely large and diverse. One consequence of such a robust and diverse immune system is that antigenic receptors that are self- reactive can develop, including against, e.g., pancreatic cells. Potentially dangerous immune cells during growth of T-cells in the thymus and B-cells in the bone marrow are typically negatively selected and eliminated (referred to as central tolerance). Self-reactive lymphocytes (e.g., T-cells and B-cells), which escape the mechanisms of central tolerance and end up in the periphery, naturally enter into processes that either neutralize or suppress them (referred to as peripheral tolerance).
- peripheral tolerance Self-reactive lymphocytes
- disorders of impacting central and peripheral tolerance can result in various autoimmune conditions.
- disruption in the function of the thymus can lead to the development of auto-immune diseases, including T1D.
- mutations in the AIRE gene can disturb central tolerance in thymus and lead to the development of T1D.
- Other non-genetic factors can have a similar effect.
- viral infections that affect human thymus can lead to autoimmune disorders, including T1D.
- Thymic epithelial cells and thymocytes have been proven to be targets of infection with coxsackievirus B4 virus and other enteroviruses in experimental studies.
- T regulatory lymphocytes formerly known as suppressive lymphocytes
- a subject as described herein may have a disrupted thymus function.
- a subject as described herein comprises one or more mutations in an AIRE gene and has had prior exposure to a coxsackieviruses strains B4, Bl, A4, A2, and/or A16 or an enterovirus.
- the present disclosure also provides that a subject as described herein may have a disrupted Treg function.
- the destruction of the P cells of the endocrine pancreas in T1D may occur via apoptosis, which includes a cascade of cysteine-asparaginase (caspase) activations.
- caspase cysteine-asparaginase
- animal studies and in vitro experiments support apoptosis as a major cell death mechanisms for beta cells.
- necrosis and necroptosis may also play a role in beta cell death with T1D.
- TNF-a tumor necrosis factor-a
- INF-y interferon-y
- cytokines activate a caspase cascade.
- apoptosis is induced directly by contact of autoreactive T-cells with beta cells via the perforating system or Fas/Fas ligand interaction.
- Fas/Fas ligand interaction a perforating system or Fas/Fas ligand interaction.
- dysfunctional cytokine secretion is involved.
- T1D a chronic atrophic inflammation within the islets of Langerhans is observed histologically, with the participation of T-cells, macrophages, B-ells, and dendritic cells. This condition usually evolves over months or years, when patients are asymptomatic and euglycemic.
- Symptomatic hyperglycemia then occurs after a long latency period, which reflects that a large number of functioning beta cells are destroyed before T1D clinically manifests.
- a subject as described herein may have inflammation (e.g., chronic inflammation, e.g., chronic atrophic inflammation) within the islets of Langerhans.
- the present disclosure also provides that a subject as described herein may have an increased level of proinflammatory cytokines IL-1, TNF-a, and INF-y when compared to a comparable healthy subject.
- Humoral immunity may also play a role in T1D by producing autoantibodies that may precede the onset of clinical manifestation of T1D, e.g., for years.
- autoantibodies detected in T1D patients include against GAD65, tyrosyl phosphatase (IA-2), insulin (IAA) and zinc transporter (ZnT8).
- IA-2 tyrosyl phosphatase
- IAA insulin
- ZnT8 zinc transporter
- Studies in mice with early presentation of these autoantibodies suggest that proinsulin is a potential primary target.
- the importance of insulin as an autoantigen has confirmed by the fact that deletion of its gene in mice affects disease progression, while its administration during the pre-diabetic phase prevents or delays clinical manifestation of T1D.
- T1D Another autoantigen implicated in T1D is the GAD enzyme, which is present in beta cells, the central nervous system, and the testes.
- IA-2 is another autoantigen involved in T1D. Autoantibodies to IA-2 usually appear later in T1D progression than autoantibodies to insulin and GAD. Zinc transporter 8 (ZnT8) has also been identified as autoantigen for T1D. In children monitored from birth to T1D development, it was observed that ZnT8 autoantibodies appear later than autoantibodies to insulin and typically disappear very early after the clinical manifestation of the disease.
- T1D One of the best markers of progression to clinical T1D is the simultaneous expression of two or three autoantibodies from the above.
- the study also investigated important factors that can contribute to the variable rate of progression to T1D among children who were already positive for autoantibodies.
- the data suggested that the risk of progression to T1D was not different among patients with or without a family history of T1D.
- the risk of progression to T1D was higher for females, when a few high-risk HLA-DR and HLA-DQ subtypes were present.
- the risk of progression to T1D was inversely related to the age of first appearance of positive autoantibodies.
- Subjects that have detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2) are provided herein.
- PBMCs that produce antibodies against one or more islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2 are provided.
- subjects as described herein can be female.
- female subjects can have a HLA-DR3, DQBl*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
- a T1D therapeutic agent as described herein may include shortacting insulin. Short-acting insulin generally starts working 30 minutes after administration (e.g., via injection) and reaches peak effect at 90 to 120 minutes. Exemplary short-acting insulin therapeutics include Humulin R, NovolinR, and Afrezza.
- a T1D therapeutic agent as described herein may include rapid-acting insulin. Rapid- acting insulin generally starts working within 15 minutes after administration (e.g., via injection) and reaches peak effect at 60 minutes. Rapid-acting insulin can last for periods of up to 4 hours after administration. Exemplary rapid-acting insulin therapeutics include glulisine, lispro, and aspart.
- a T1D therapeutic agent as described herein may include intermediate- acting insulin.
- Intermediate-acting insulin generally starts working 1 to 3 hours after administration (e.g., via injection) and reaches peak effect at 6-8 hours after administration.
- Intermediate-acting insulin can last for periods of up to 12 to 24 hours after administration.
- An exemplary intermediate-acting insulin therapeutics includes insulin NPH.
- a T1D therapeutic agent as described herein may include long-acting insulin.
- Long-acting insulin generally works for 14 to 40 hours after administration (e.g., via injection).
- Exemplary intermediate- acting insulin therapeutics include insulin glargine, detemir, and degludec.
- T1D therapeutic agent While insulin is one T1D therapeutic agent, it is not the only one. Additional T1D therapeutic agents include metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
- a subject described herein may have received one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
- a PBMC or islet cell as provided herein may have been exposed to one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
- technologies described herein can be used to determine how assess how a subject’s PBMCs and/or islet cells respond to T1D therapeutics, including one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
- Technologies described herein may also be useful for providing prognostic and/or predictive information about how a subject may respond to T1D therapeutics, including one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
- T1D therapeutics including one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
- non-human animals can be engineered such that the animals sufficiently model human disease, disorders, and conditions. Such non-human animals can provide insights into disease pathology and etiology. Non-human animals can also be useful in assessing the safety and efficacy of treatment (e.g., a therapeutic agent).
- non-human animals can be engineered such that the animals sufficiently model autoimmune disorders, such as T1D. These animals can be utilized to determine the pathology and etiology of autoimmune disorders, such as T1D.
- a powerful benefit to non-human animals is that the animals can be engineered to model a particular subject’s (e.g., a particular human’s) autoimmune disorder, such as T1D, and can be used to assess how that particular subject will respond to a treatment, including the safety and efficacy of such a treatment (e.g., a therapeutic agent).
- a particular subject e.g., a particular human
- autoimmune disorder such as T1D
- non-human animals of the present disclosure represent a robust tool for evaluating autoimmunity, T1D, and potential therapeutics at a high level, but also provide a tool for determining a personalized approach to treating a patient.
- Non-human animals are provided by the present disclosure.
- a non-human animal is a mammal.
- a non-human mammal is a primate, a goat, a sheep, a pig, a dog, a cow, or a rodent.
- a non- human animal of the present disclosure is a rodent.
- a rodent is a mouse or a rat.
- a non-human animal is a mouse.
- a mouse is a NOD/ShiLtJ strain (sometimes referred to as NOD strain) mouse.
- a mouse is a NSG mouse (also referred to as a N0D.Cg-Prkdc Sl l ⁇ l IL2rg"" l wjl /SzJ mouse). In some embodiments, a mouse is a NSG-SGM3 mouse.
- a non-human animal is a mouse.
- a mouse comprises reduced function of non-human islet cells relative to a comparable wild-type mouse.
- a mouse comprises reduced activity of non-human islet cells relative to a comparable wild-type mouse.
- a mouse comprises a reduced number of non-human islet cells relative to a comparable wild-type mouse.
- a mouse does not comprise murine islet cells.
- a mouse has reduced pancreatic function relative to a comparable wild-type mouse.
- a mouse has reduced pancreatic activity relative to a comparable wild- type mouse.
- a mouse has a reduced number of pancreatic cells relative to a comparable wildtype mouse. In some embodiments, a mouse does not have a pancreas. In some embodiments, a non-human animal is a mouse that has had its pancreas removed. In some embodiments, a non- human animal is a mouse that has had its islet cells removed or destroyed. In some embodiments, a non-human animal is a mouse that has had its beta cells removed or destroyed. In some embodiments, a non-human animal is a mouse that has been treated with STZ. In some embodiments, a mouse is a NOD/ShiLtJ strain (sometimes referred to as NOD strain) mouse. In some embodiments, a mouse is a NSG mouse.
- NOD/ShiLtJ strain sometimes referred to as NOD strain
- a mouse is a NSG-SGM3 mouse.
- a mouse is a C57BL strain mouse.
- a C57BL mouse is a C57BL/A mouse, C57BL/An mouse, C57BL/GrFa mouse, C57BL/KaLwN mouse, C57BL/6 mouse, C57BL/6J mouse, C57BL/6ByJ mouse, C57BL/6NJ mouse, C57BL/10 mouse, C57BL/10ScSn mouse, C57BL/10Cr mouse, or C57BL/01a mouse.
- a mouse is a BALB strain mouse, e.g., a BALB/c mouse.
- a mouse is a 129 strain mouse.
- a 129 strain mouse is a 129P1 mouse, 129P2 mouse, 129P3 mouse, 129X1 mouse, 129S1 mouse (e.g., 129S1/SV mouse, 129Sl/SvIm mouse), 129S2 mouse, 129S4 mouse, 129S5 mouse, 129S9/SvEvH mouse, 129/SvJac mouse, 129S6 (129/SvEvTac) mouse, 129S7 mouse, 129S8 mouse, 129T1 mouse, or 129T2 mouse (see, e.g., Festing et al., 1999, Mammalian Genome 10:836; Auerbach, W.
- a mouse is a mix of two or more of the aforementioned strains.
- a non-human animal is a rat.
- a rat is a BB Wistar rat, Goto-Kakizaki (GK) rat, Zucker diabetic fatty (ZDF) rat, Komeda diabetes- prone (KDP) rat, BBDP rat, LETL rat, LEW.lARl/Ztm-iddm rat, BBDR rat, PVG rat, PVG.RT1 U rat, PVG.R8 rat, WAG rat, WF.iddm4 d congenic rat, or LEW.1WR1 rat.
- a rat is a BB Wistar rat.
- a rat is a KDP rat.
- a non-human animal is a rat.
- a non-human animal is a rat.
- a rat comprises reduced function of non- human islet cells relative to a comparable wild-type rat.
- a rat comprises reduced activity of non-human islet cells relative to a comparable wild- type rat.
- a rat comprises a reduced number of non-human islet cells relative to a comparable wild-type rat.
- a rat does not comprise murine islet cells.
- a rat has reduced pancreatic function relative to a comparable wild-type rat.
- a rat has reduced pancreatic activity relative to a comparable wild-type rat. In some embodiments, a rat has a reduced number of pancreatic cells relative to a comparable wild-type rat. In some embodiments, a rat does not have a pancreas. In some embodiments, a rat has had its pancreas removed. In some embodiments, a non-human animal is a rat that has had its islet cells removed or destroyed. In some embodiments, a non-human animal is a rat that has had its beta cells removed or destroyed. In some embodiments, a non- human animal is a rat that has been treated with STZ.
- a rat is a BB Wistar rat, Goto-Kakizaki (GK) rat, Zucker diabetic fatty (ZDF) rat, Komeda diabetes-prone (KDP) rat, BBDP rat, LETL rat, LEW.l ARl/Ztm-iddm rat, BBDR rat, PVG rat, PVG.RT1 U rat, PVG.R8 rat, WAG rat, WF.iddm4 d congenic rat, or LEW.1WR1 rat, a Wistar rat, LEA strain rat, Sprague Dawley strain rat, Fischer strain rat, F344 rat, F6 rat, or Dark Agouti rat.
- a rat is a mix of two or more of the aforementioned strains. [01921 Female mice tend to develop diabetes sooner than corresponding male mice.
- a mouse is a female mouse.
- the present disclosure also contemplates using male mice.
- a mouse is a male mouse.
- a plurality of mice are each the same strain.
- a plurality of mice can be a colony.
- a plurality of mice can be a cohort.
- a cohort is defined by a generation of mice.
- a cohort is defined by the sex of the mice.
- a cohort comprises female mice.
- a cohort comprises male mice.
- a cohort is defined by two or more criteria.
- a cohort comprises female mice from the same generation of mice.
- non-human animals as described above, which comprise PBMCs and islet cells derived from cells of a subject.
- a subject of the present disclosure is a donor of PBMCs and/or other cells from which islet cells are differentiated.
- characteristics and features of a subject can also be or affect characteristics and features of PBMCs and islet cells derived from cells of the subject. Further, in some embodiments, a subject may also be a recipient of islet cells which were derived from the subject’s cells. In some embodiments, a subject can receive islet cells as part of a cell therapy, e.g., for T1D.
- a subject is a mammal. In some embodiments, a subject is a primate, a goat, a sheep, a pig, a dog, a cow, or a rodent. In some embodiments, a subject is a human. [01961 In some embodiments, a subject has T1D. In some embodiments, a subject is at risk for developing T1D. In some embodiments, a subject has early indications of T1D, such as inflammation of the islets of Langerhans.
- a subject provided herein may be in need of a T1D therapeutic agent.
- a subject is eligible for islet transplant therapy.
- a subject had T1 D onset at an age of less than 50 years old, less than 45 years old, less than 40 years old, less than 35 years old, less than 30 years old, or less than 25 years old. In some embodiments, a subject had T ID onset at an age of less than 40 years old.
- a subject has had insulin dependence for at least 2 years, at least 3 years, at least 4 years, at least 5 year’s, at least 6 year’s, at least 7 years, at least 8 years, at least 9 years, or at least 10 years. In some embodiments, a subject has had insulin dependence for at least 5 years.
- Diabetes can increase in severity with age.
- a subject is at least 15 years old, at least 20 year’s old, at least 25 years old, at least 30 year’s old, at least 35 years old, at least 40 years old, at least 45 years old, or at least 50 years old.
- a subject with T1D has had T1D for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 years, at least 8 years, at least 9 years, at least 10 years, at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 year’s, or at least 90 years. In some embodiments, a subject with T1D has had T1D for at least 5 years.
- a sum of age and years of insulin dependent diabetes is at least 15 years, at least 20 years, at least 25 years, at least 26 years, at least 27 years, at least 28 years, at least 29 years, or at least 30 years. In some embodiments, a sum of age and years of insulin dependent diabetes is at least 28 years.
- a C-peptide test measures the amount of C-peptide in the blood or urine. The pancreas releases C-peptide when it makes insulin.
- a C-peptide test can help determine the type of diabetes a patient has (i.e., type 1 diabetes or type 2 diabetes) or how well diabetes treatments are working. C-peptide is a byproduct the pancreas releases into the body when it makes insulin. The pancreas produces about the same amount of insulin and C-peptide. Because C-peptide is a byproduct of insulin production, this test can reveal how much insulin the body is producing. When people take insulin, their bodies do not make or release C-peptide. This test can tell the difference between insulin made by the body (endogenous) and insulin from medication (exogenous). Therefore, a C-peptide test indicates if a subject’s body has the ability to make its own insulin from the pancreas.
- a subject has less than 0.5 ng/mL, less than 0.4 ng/mL, less than 0.3 ng/mL, less than 0.2 ng/mL, less than 0.1 ng/mL, or an undetectable level of stimulated C-peptide at 60 minutes post-mixed-meal tolerance test. In some embodiments, a subject has less than 0.5 ng/mL, less than 0.4 ng/mL, less than 0.3 ng/mL, less than 0.2 ng/mL, less than 0.1 ng/mL, or an undetectable level of stimulated C-peptide at 90 minutes post-mixed- meal tolerance test.
- a subject has less than 0.5 ng/mL, less than 0.4 ng/mL, less than 0.3 ng/mL, less than 0.2 ng/mL, less than 0.1 ng/mL, or an undetectable level of stimulated C-peptide at 60 minutes post-mixed-meal tolerance test. In some embodiments, a subject has less than 0.5 ng/mL, less than 0.4 ng/mL, less than 0.3 ng/mL, less than 0.2 ng/mL, less than 0.1 ng/mL, or an undetectable level of stimulated C-peptide at 60 minutes and 90 minutes post-mixed-meal tolerance test.
- a subject has less than 0.3 ng/mL of stimulated C-peptide at 60 minutes post-mixed-meal tolerance test. In some embodiments, a subject has less than 0.3 ng/mL of stimulated C-peptide at 90 minutes post- mixed-meal tolerance test. In some embodiments, a subject has less than 0.3 ng/mL of stimulated C-peptide at 60 minutes and 90 minutes post-mixed-meal tolerance test.
- a subject has intensive diabetes management.
- intensive diabetes management comprises self-monitoring of glucose values no less than a mean of three times each day averaged over each week.
- intensive diabetes management comprises administration of three or more insulin injections each day or insulin pump therapy.
- intensive diabetes management comprises at least three clinical evaluations during the past 12 months with an endocrinologist, diabetologist, or diabetes specialist.
- intensive diabetes management comprises self-monitoring of glucose values no less than a mean of three times each day averaged over each week, administration of three or more insulin injections each day or insulin pump therapy, and/or at least three clinical evaluations during the past 12 months with an endocrinologist, diabetologist, or diabetes specialist.
- a subject has had at least one episode of severe hypoglycemia in the past 12 months.
- an episode of severe hypoglycemia comprises an event with one or more of the following symptoms (i) memory loss, (ii) confusion, (iii) uncontrollable behavior, (iv) irrational behavior, (v) unusual difficulty in awakening, (vi) suspected seizure, (vii) seizure, (viii) loss of consciousness, and (ix) visual symptoms.
- a subject has impaired awareness of hypoglycemia (IAH).
- IAH is defined as “a diminished ability to perceive the onset of acute hypoglycaemia.” IAH can arise when repeated episodes of hypoglycaemia raise the glycaemic threshold for symptoms to occur.
- IAH can be assessed using a Clarke reduced awareness score.
- a subject has a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more.
- a subject has a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more.
- a subject has a marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more.
- LI liability index
- a subject has a composite of: a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile.
- a subject has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years.
- SHE severe hypoglycemic event
- a subject has islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2).
- a subject has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2).
- a subject has antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
- JDFU juvenile diabetes foundation units
- JDFU juvenile diabetes foundation units
- glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU
- insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU
- antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU
- a subject has one or more genetic factors associated with T1D.
- a subject has a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
- a subject has a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) and HLA- DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
- a subject comprises a class I VNTR at an insulin gene promoter.
- a subject comprises a polymorphism within a CTLA-4 gene that results in a decreased level of CTLA-4 expression as compared to level of CTLA-4 expression observed with a comparable subject that does not have the polymorphism.
- a subject comprises a C1858T (Arg620Trp) polymorphism in a PTPN22 gene.
- a subject comprises one or more mutations in an AIRE gene.
- expression of AIRE in a subject with one or more mutations in an AIRE gene may be decreased compared to a subject without the one or more mutations.
- a subject as described herein can comprise one or more mutations in a FoxP3 gene.
- expression of scurfin in a subject with one or more mutations in a FoxP3 gene may be decreased compared to a subject without the one or more mutations.
- a subject comprises one or more mutations in a STAT3 gene.
- expression of STAT3 in a subject with one or more mutations in a STAT3 gene may be decreased compared to a subject without the one or more mutations.
- a subject comprises one or more mutations in an IFIH1 gene.
- expression of MDA5 in a subject with one or more mutations in an 1F1H1 gene may be decreased compared to a subject without the one or more mutations.
- a subject comprises one or more mutations in an HIP14 gene.
- expression of HIP14 in a subject with one or more mutations in an HIP14 gene may be decreased compared to a subject without the one or more mutations.
- a subject comprises one or more mutations in an ERBB3 gene.
- expression of ERBB3 in a subject with one or more mutations in an ERBB3 gene may be decreased compared to a subject without the one or more mutations.
- a subject has been exposed to one or more environmental factors associated with the development of T1D.
- a subject has been exposed to and/or been infected with a coxsackieviruses strain B4, Bl, A4, A2, and/or A16.
- a subject has been exposed to and/or infected with an enterovirus.
- a subject produces antibodies ABBOS.
- a subject has a microbiome with a reduced Firmicutes:Bacteroidetes ratio as compared to a comparable subject that has not been diagnosed with an autoimmune disorder (e.g., T1D).
- an autoimmune disorder e.g., T1D
- a subject was birthed via cesarean section.
- a subject has one or more immunological factors associated with T1D.
- a subject has a disrupted thymus function.
- a subject comprises one or more mutations in an AIRE gene and has had prior exposure to a coxsackieviruses strain B4, Bl, A4, A2, and/or A16 or an enterovirus.
- a subject has a disrupted Treg function.
- a subject has inflammation (e.g., chronic inflammation, e.g., chronic atrophic inflammation) within the islets of Langerhans.
- inflammation e.g., chronic inflammation, e.g., chronic atrophic inflammation
- a subject has an increased level of proinflammatory cytokines IL-1, TNF-a, and INF-y when compared to a comparable healthy subject.
- a subject is female.
- a female subject has a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
- Subjects provided herein may have previously been administered a T1D therapeutic agent. Subjects provided herein may currently be receiving a T1D therapeutic agent. In some embodiments, a subject has received one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
- a PBMC or islet cell as provided herein may have been exposed to one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
- immune cells e.g., peripheral blood mononuclear cells (PBMCs)
- PBMCs peripheral blood mononuclear cells
- T ID type 1 diabetes
- immune cells e.g., PBMCs (and/or other cells from which islet cells are differentiated)
- PBMCs peripheral blood mononuclear cells
- characteristics and features of a subject can also be or affect characteristics and features of immune cells (e.g., PBMCs) from the subject.
- immune cells e.g., PBMCs
- PBMCs mammalian immune cells
- human immune cells e.g., human PBMCs
- immune cells e.g., PBMCs
- PBMCs immune cells
- PBMCs can be used as obtained.
- immune cells e.g., PBMCs
- immune cells e.g., PBMCs
- immune cells can be modified.
- immune cells e.g., PBMCs
- iPSCs can then be differentiated into islet cells.
- immune cells e.g., PBMCs
- immune cells as described herein comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, dendritic cells, or a combination thereof.
- PBMCs as described herein comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, dendritic cells, or a combination thereof.
- one or more of the immune cells are T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
- one or more of the PBMCs are T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
- one or more of immune cells is a B cell.
- one or more of immune cells produce autoantibodies against autoantigens of a subject (e.g., a mammal, e.g., a human).
- one or more immune cells produce antibodies against cells or antigens of a subject (e.g., a mammal, e.g., a human).
- one or more of immune cells produce antibodies against islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2.
- one or more of immune cells produce islet cell antibodies (ICA) and/or insulin autoantibodies (IAA).
- ICA islet cell antibodies
- IAA insulin autoantibodies
- one or more of PBMCs is a B cell.
- one or more of PBMCs produce autoantibodies against autoantigens of a subject (e.g., a mammal, e.g., a human).
- one or more PBMCs produce antibodies against cells or antigens of a subject (e.g., a mammal, e.g., a human).
- one or more of PBMCs produce antibodies against islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2.
- one or more of PBMCs produce islet cell antibodies (ICA) and/or insulin autoantibodies (IAA).
- ICA islet cell antibodies
- IAA insulin autoantibodies
- one or more of immune cells is an islet-reactive T cell.
- one or more of PBMCs is an islet-reactive T cell.
- one or more of immune cells are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal.
- one or more of PBMCs are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal.
- a subject e.g., a mammal, e.g., a human
- a non-human animal e.g.,
- a non-human animal as described herein comprises at least about 2.5xl0 5 , at least about 5xl0 5 , at least about IxlO 6 , at least about 2xl0 6 , at least about 3xl0 6 , at least about 4xl0 6 , at least about 5xl0 6 , at least about 6xl0 6 , at least about 7xl0 6 , at least about 8xl0 6 , at least about 9xl0 6 , at least about IxlO 7 , at least about 1.5xl0 7 , or at least about 2xl0 7 immune cells from a subject (e.g., a mammal, e.g., a human).
- a subject e.g., a mammal, e.g., a human
- a subject e.g., a mammal, e.g., a human
- a non-human animal e.g.
- a non-human animal as described herein comprises at least about 2.5xl0 5 , at least about 5xl0 5 , at least about IxlO 6 , at least about 2xl0 6 , at least about 3xl0 6 , at least about 4xl0 6 , at least about 5xl0 6 , at least about 6xl0 6 , at least about 7xl0 6 , at least about 8xl0 6 , at least about 9xl0 6 , at least about IxlO 7 , at least about 1.5xl0 7 , or at least about 2xl0 7 PBMCs from a subject (e.g., a mammal, e.g., a human).
- a subject e.g., a mammal, e.g., a human
- one or more of immune cells comprise one or more genetic factors associated with T1D.
- one or more of immune cells comprise a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
- one or more of immune cells e.g., PBMCs
- a HLA-DR3, DQB 1*0201 also referred as DR3-DQ2
- HLA-DR4, DQB 1*0302 also referred as DR4-DQ8 haplotype.
- one or more of immune cells comprise a class I VNTR at an insulin gene promoter.
- one or more of immune cells comprise a polymorphism within a CTLA-4 gene that results in a decreased level of CTLA-4 expression as compared to level of CTLA-4 expression observed with a comparable subject that does not have the polymorphism.
- one or more of immune cells e.g., PBMCsjcomprise a C1858T (Arg620Trp) polymorphism in a PTPN22 gene.
- one or more of immune cells e.g., PBMCsjcomprise one or more mutations in an AIRE gene.
- expression of AIRE in a PBMC with one or more mutations in an AIRE gene may be decreased compared to a comparable immune cell (e.g., PBMCjwithout the one or more mutations.
- one or more of immune cells e.g., PBMCsJcomprisc one or more mutations in a FoxP3 gene.
- expression of scurfin in an immune cell (e.g., PBMC) with one or more mutations in a FoxP3 gene may be decreased compared to a comparable immune cell (e.g., PBMC) without the one or more mutations.
- one or more of immune cells comprise one or more mutations in a STAT3 gene.
- expression of STAT3 in an immune cell (e.g., PBMC) with one or more mutations in a STAT3 gene may be decreased compared to a comparable immune cell (e.g., PBMC) without the one or more mutations.
- one or more of immune cells e.g., PBMCsjcomprise one or more mutations in an IFIH1 gene.
- expression of MDA5 in an immune cell e.g., PBMCjwith one or more mutations in an IFIH1 gene may be decreased compared to a comparable immune cell (e.g., PBMC)without the one or more mutations.
- one or more of immune cells comprise one or more mutations in an HIP14 gene.
- expression of HIP14 in an immune cell (e.g., PBMC) with one or more mutations in an HIP14 gene may be decreased compared to a comparable immune cell (e.g., PBMC) without the one or more mutations.
- one or more of immune cells comprise one or more mutations in an ERBB3 gene.
- expression of ERBB3 in an immune cell (e.g., PBMC) with one or more mutations in an ERBB3 gene may be decreased compared to a comparable immune cell (e.g., PBMC) without the one or more mutations.
- an immune cell e.g., PBMC
- PBMC coxsackieviruses strain B4, Bl, A4, A2, and/or A16.
- an immune cell e.g., PBMC
- an enterovirus e.g., PBMC
- an immune cell e.g., PBMC
- PBMC PBMC
- an immune cell has been obtained from a subject that produces antibodies ABBOS.
- an immune cell e.g., PBMC
- PBMC has been obtained from a subject that has a microbiome with a reduced Firmicutes:Bacteroidetes ratio as compared to a comparable subject that has not been diagnosed with an autoimmune disorder (e.g., T1D).
- an immune cell e.g., PBMC
- PBMC has been obtained from a subject that was birthed via cesarean section.
- an immune cell e.g., PBMC
- PBMC has been obtained from a subject that has one or more immunological factors associated with T1D.
- an immune cell e.g., PBMC
- PBMC has been obtained from a subject that has a disrupted thymus function.
- an immune cell e.g., PBMC
- an immune cell e.g., PBMC
- PBMC PBMC
- an immune cell e.g., PBMC
- inflammation e.g., chronic inflammation, e.g., chronic atrophic inflammation
- an immune cell e.g., PBMC
- an immune cell e.g., PBMC
- PBMC has been obtained from a subject that is female.
- an immune cell e.g., PBMC
- Immune cells can be obtained from subjects that may have previously been administered a T1D therapeutic agent. Immune cells (e.g., PBMCs) can be obtained from subjects that may currently be receiving a T1D therapeutic agent.
- an immune cell e.g., PBMC
- An immune cell e.g., PBMC
- PBMC may have been exposed to one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
- the pancreas contains clusters of cells, known as islets, that produce hormones. There are several different types of cells in an islet. For example, alpha cells produce glucagon; delta cells produce somatostatin; and beta cells produce insulin. A sample of pancreatic islet cells may comprise at least alpha cells, delta cells, and beta cells. As used throughout, a “beta cell” should be understood to be a beta islet cell. In some embodiments, an islet cell is an islet progenitor cell, an immature islet cell, or a mature islet cell.
- beta-islet cells used in a cell therapy product may be pluripotent stem cell (iPSC)-derived beta-islet cells.
- iPSC pluripotent stem cell
- Methods for profiling a population of cells for donor capability as described anywhere herein may also be performed on stem cells capable of differentiating to form beta-islet cells.
- Methods for profiling a population of cells for donor capability as described anywhere herein may also be performed on stem cell derived (e.g., genome-edited) beta-islet cells.
- Beta-islet cells as referred to in the context of the present disclosure is known in the art, including certain information regarding desired features of beta-islet cells when used for cell therapy. It will be understood that embodiments concerning beta-islet cells described herein may be readily and appropriately combined with embodiments describing HIP cells (e.g., exhibiting reduced expression of one or more molecules of the MHC class I and/or MHC class II molecules and increased expression of at least one tolerogenic factor), as well as embodiments describing safety switches, and other modified/ gene edited cells as described herein. Beta-islet cells to be used in a cell therapy product may be profiled for donor capability at any stage of the editing process during manufacturing of the cell therapy product.
- a key functional feature of a P cell is its ability to repeatedly perform glucose stimulated insulin secretion (GSIS).
- GSIS glucose stimulated insulin secretion
- assays can be performed to determine the physiological function in vitro of secreting insulin in response to glucose.
- the GSIS assay may be a perifusion GSIS (dynamic GSIS) assay (for example as in Velazco-Cruz, Stem Cell Reports, 2019).
- other assays can be performed to examine the expression of specific genes, pathways, and transcription factors.
- Such assays include those detecting the presence of Yap (Rosado-Olivieri et al., 2019, which is incorporated herein by reference in its entirety), the ROCKII pathway (Ghazizadeh et al., 2017, which is incorporated herein by reference in its entirety), the transforming growth factor b (TGF-b) pathway (Velazco-Cruz, et al., 2019, which is incorporated herein by reference in its entirety), the cytoskeleton (Hogrebe et al., 2020, which is incorporated herein by reference in its entirety), and the expression of SIX2 (Velazco-Cruz et al., Cell Reports, 2020, which is incorporated herein by reference in its entirety).
- transcription factors important for the SC-P cell phenotype include PDX1, NKX6-1, NKX2-2, and NEURODI (Hogrebe et al., 2020, which is incorporated herein by reference in its entirety).
- an assay measuring changes in intracellular Ca2+ may be performed as described in Pagliuca et al. (Cell, 2014, which is incorporated herein by reference in its entirety).
- 0 cells sense changing glucose levels through calcium signaling; increasing glucose levels leads to membrane depolarization causing an influx of calcium ions which triggers insulin exocytosis (Mohammed et al., 2009, which is incorporated herein by reference in its entirety).
- the functional SC-0 cells exhibit calcium flux similarly to primary human islet cells.
- assays can also be performed to assess in vivo functionality of the SC-0 cells.
- An example of such an assay can be found in Pagliuca et al. (Cell, 2014, which is incorporated herein by reference in its entirety). Briefly, to test their capacity to function in vivo, SC-0 cells are transplanted under the kidney capsule of immunocompromised mice and the ability of the cells to produce insulin is analyzed.
- beta-islet cells described herein may be used to treat or prevent a disease in a subject.
- islet cells that are derived (e.g., differentiated) from cells of a subject (e.g., a patient with type 1 diabetes (T1D)).
- a subject e.g., a patient with type 1 diabetes (T1D)
- cells from which islet cells are differentiated (and/or PBMCs) are obtained from the same subject (e.g., the PBMCs and the islet cells are autologous).
- characteristics and features of a subject can also be or affect characteristics and features of islet cells derived from cells of the subject and PBMCs from the subject.
- PBMCs are obtained from a subject as described herein, PBMCs provided can be mammalian PBMCs, e.g., human PBMCs.
- Beta-islet cells as described herein to be used in a cell therapy product may be present with alpha cells and delta cells in the cell therapy product. Islet cells to be used in a cell therapy product may be profiled for donor capability at any stage of the manufacturing process of the cell therapy product.
- islet cells are or comprise alpha, beta, and/or delta islet cells.
- islet cells are or comprise about 5%, about 10%, about 15%, about 20%, about 25%, about 35%, or about 40% alpha cells.
- islet cells are or comprise about 10%, about 15%, about 20%, about 25%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, or about 80% beta cells. In some embodiments, islet cells are or comprise about 1%, about 2%, about 3%, about 4%, about 5%, about 6%, about 7%, about 8%, about 9%, about 10%, about 15%, about 20%, or about 25% delta cells.
- islet cells are or comprise differentiated cells.
- differentiated cells are derived from a stem cell.
- differentiated cells are derived from a pluripotent stem cell.
- differentiated cells are derived from an induced pluripotent stem cell (iPSC).
- iPSC is derived from a cell obtained from the human patient with T1D.
- a cell obtained from a human patient with T1D is selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell.
- a cell obtained from a human patient with T1D is a PBMC.
- a stem cell is derived from a cell obtained from a human patient with T ID.
- a stem cell is an induced pluripotent stem cell (iPSC) derived from a cell selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell.
- a stem cell is an induced pluripotent stem cell (iPSC) derived from a PBMC.
- differentiated cells are derived from a multipotent stem cell.
- a multipotent stem cell is derived from a cell obtained from a human patient with T1D.
- differentiated cells are derived from a lineage- specific stem cell.
- a lineage- specific stem cell is derived from a cell obtained from a human patient with T1D.
- differentiated cells are derived from a definitive endoderm cell.
- a definitive endoderm cell expresses FOXA2 and/or SOX 17.
- differentiated cells are derived from a primitive gut tube cell.
- a primitive gut tube cell expresses FOXA2 and/or SOX 17.
- differentiated cells are derived from a pancreatic progenitor cell.
- a pancreatic progenitor cell expresses PDX1 and/or NKX6-1.
- differentiated cells are derived from an endocrine cell.
- an endocrine cell expresses NKX6-1, NEUROG3, and/or NKX2-2.
- islet cells produce insulin, c-peptide, glucagon, somatostatin, and/or pancreatic polypeptide.
- islet cells as described herein are used to make a first therapeutic agent as described herein.
- islet cells are introduced to a non-human animal of the present disclosure via intramuscular injection, intravenous injection, intraportal injection, ocular injection, injection into omentum, and/or injection into a kidney capsule.
- islet cells are or comprise engineered islet cells.
- engineered islet cells are derived (e.g., differentiated) from cells as described herein that have been engineered.
- the cells as provided herein are beta islet cells derived from engineered iPSCs that contain modifications (e.g., genetic modifications) described herein and that are differentiated into beta islet cells.
- modifications e.g., genetic modifications
- the methods for differentiation depend on the desired cell type using known techniques.
- the cells differentiated into various beta islet cells may be used for subsequent transplantation or engraftment into subjects (e.g., recipients).
- pancreatic islet cells are derived from the engineered pluripotent cells described herein.
- Useful methods for differentiating pluripotent stem cells into beta islet cells are described, for example, in U.S. Patent No. 9,683,215; U.S. Patent No. 9,157,062; U.S. Patent No. 8,927,280; U.S. Patent Pub. No. 2021/0207099; Hogrebe et al., "Targeting the cytoskeleton to direct pancreatic differentiation of human pluripotent stem cells,” Nat. Biotechnol., 2020, 38:460-470; and Hogrebe et al., “Generation of insulin-producing pancreatic beta cells from multiple human stem cell lines,” Nat. Protoc., 2021, the contents of which are herein incorporated by reference in their entirety.
- the engineered pluripotent cells described herein are differentiated into beta-like cells or islet organoids for transplantation to address type I diabetes mellitus (T1DM).
- T1DM type I diabetes mellitus
- Pagliuca et al. reports on the successful differentiation of P-cells from hiPSCs, the contents incorporated herein by reference in its entirety and in particular for the methods and reagents outlined there for the large-scale production of functional human P cells from human pluripotent stem cells).
- Vegas et al. shows the production of human P cells from human pluripotent stem cells followed by encapsulation to avoid immune rejection by the host; Vegas et al., Nat Med, 2016, 22(3):306- 11, incorporated herein by reference in its entirety and in particular for the methods and reagents outlined there for the large-scale production of functional human P cells from human pluripotent stem cells.
- engineered islet cells are derived (e.g., differentiated) from engineered induced pluripotent stem cells (iPSCs) modified to be hypoimmune iPSCs.
- iPSCs have been derived (e.g., reprogrammed) from cells (e.g., PBMCs) of a subject back into a pluripotent state.
- engineered islet cells have been modified to be hypoimmune islet cells.
- all modifications present in a hypoimmune islet cell are present in the iPSC that the hypoimmune islet cell is derived from.
- a hypoimmune islet cell comprises one or more modifications in addition to the one or more modifications present in the iPSC the islet cell is derived from.
- a hypoimmune islet cell comprises one or more modifications, none of which are present in the iPSC the hypoimmune islet cell is derived from.
- an iPSC comprising one or more modifications is differentiated into a hypoimmune islet cell, wherein the hypoimmune islet cell comprises no additional modifications relative to the iPSC from which it is derived. In some embodiments, an iPSC comprising one or more modifications is differentiated into a hypoimmune islet cell, wherein the hypoimmune islet cell comprises one or more additional modifications relative to the iPSC from which it is derived. In some embodiments, an iPSC comprising no hypoimmune modifications is differentiated into an islet cell, wherein the islet cell is modified to comprise one or more modifications relative to the iPSC from which it is derived.
- the present disclosure provides methods of creating hypoimmune islet cells derived from iPSCs.
- the iPSCs are hypoimmune iPSCs.
- a method comprises engineering an iPSC to generate a hypoimmune iPSC comprising one or more modifications, and differentiating the hypoimmune iPSC into a hypoimmune islet cell, wherein the hypoimmune islet cell comprises the same one or more modifications as the hypoimmune iPSC.
- a method comprises engineering an iPSC to generate an iPSC comprising one or more modifications, differentiating the iPSC into a hypoimmune islet cell, and then engineering the hypoimmune islet cells to comprise one or more additional modifications, wherein the hypoimmune islet cells comprises one or more additional modifications relative to the iPSC.
- a method comprises differentiating an iPSC into an islet cell and then engineering the islet cell to comprise one or more modifications, thereby generating a hypoimmune islet cell, wherein the hypoimmune islet cell comprises one or more modifications relative to the iPSC.
- hypoimmune islet cells comprise one or more modifications that (a) inactivate or disrupt one or more alleles of: (i) one or more major histocompatibility complex (MHC) class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or (ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules, and/or (b) increase expression of one or more tolerogenic factors, wherein the increased expression of (b) is relative to an islet cell that does not comprise the modifications.
- MHC major histocompatibility complex
- one or more modifications that disrupt one or more MHC class I molecules and/or one or more MHC class 11 molecules reduce expression of the one or more MHC class 1 molecules and/or the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications.
- hypoimmune islet cells are (i) HLA class I- deficient and/or HLA class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47.
- engineered stem cell-derived islet cells obtained by in vitro differentiation of a modified pluripotent stem cell.
- the modified pluripotent stem cell can be any as described below.
- the provided engineered islet cells are differentiated in vitro from the modified pluripotent stem cell by any method able to generate a functional islet cell.
- the differentiated engineered islet cell is a modified iPSC-derived beta islet cell.
- the provided engineered islet cells retain the one or more modifications of the modified pluripotent stem cells and/or retain or exhibit similar expression of the target immune molecules (e.g. reduced expression of MHC class I and/or II and increased expression of a tolerogenic factor, such as CD47).
- the modified islet cells provided herein also are functional and exhibit one or more functions of primary islet cells or beta islet cells, such as the ability to secrete insulin, for example glucose stimulated insulin secretion (GSIS).
- GSIS glucose stimulated insulin secretion
- populations of cells containing the engineered islet cells are also provided. It is understood that differentiation from a population may not result in 100% having fully differentiated to the same stage in the differentiation pathway. Thus, it should be appreciated that not all cells in a particular population progress through these stages at the same rate, i.e., some cells may have progressed less, or more, down the differentiation pathway than the majority of cells present in the population. Accordingly, a population of islet cells may also include cells that are partially differentiated from the modified pluripotent stem cell or is a precursor of the cell stage such as precursor of the differentiated islet cell. In some cases, a percentage or portion of the cells may be at an earlier stage.
- engineered islet cells are differentiated in vitro (e.g., from pluripotent stem cells) and are cells that display at least one marker indicative of a pancreatic beta cell (e.g., PDX-1 or NKX6-1), express insulin, and display a GSIS response characteristic of an endogenous mature beta cell both in vitro and in vivo.
- a marker indicative of a beta cell is a marker selected from INS, CHGA, NKX2-2, PDX1, NKX6-1, MAFB, GCK and GLUT1.
- the GSIS response of an engineered islet cell can be observed within two weeks of transplantation of the engineered islet cell into a host (e.g., a human or animal).
- a host e.g., a human or animal.
- islet cells need not be derived (e.g., directly) from stem cells, as any method can be used that is capable of deriving engineered islet cells from any endocrine progenitor cell that expresses insulin or precursor thereof using any cell as a starting point in which such starting cell has been modified by the one or more modifications described herein.
- a starting cell may be a modified cell according to the present disclosure that is an induced-pluripotent stem cells, progenitor cells, partially reprogrammed somatic cells (e.g., a somatic cell which has been partially reprogrammed to an intermediate state between an induced pluripotent stem cell and the somatic cell from which it was derived), multipotent cells, totipotent cells, a transdifferentiated version of any of the foregoing cells.
- induced-pluripotent stem cells e.g., a somatic cell which has been partially reprogrammed to an intermediate state between an induced pluripotent stem cell and the somatic cell from which it was derived
- multipotent cells e.g., a somatic cell which has been partially reprogrammed to an intermediate state between an induced pluripotent stem cell and the somatic cell from which it was derived
- multipotent cells e.g., a somatic cell which has been partially reprogrammed to an intermediate state between an
- engineered islet cells have regulated or modulated (e.g. reduced or eliminated) expression of MHC class I molecules, MHC class II molecules, or MHC class I and MHC class II molecules.
- the regulated or modulated expression of MHC class I and/or Class II is due to gene editing in which the DNA of the gene loci involved in regulation of expression of MHC class I and/or class II have been edited to delete genomic DNA of a gene involved in regulation of expression of the immune molecule.
- an engineered islet cell has an edit to delete genomic DNA of beta-2 microglobulin (B2M) and is thus reduced or eliminated for expression of MHC class I.
- B2M beta-2 microglobulin
- the B2M gene is knocked out in an engineered islet cell. In some embodiments, both alleles of B2M are knocked out. In some embodiments, an engineered islet cell has an edit to delete genomic DNA of CIITA and is thus reduced or eliminated for expression of MHC class II. In some embodiments, the CIITA gene is knocked out in an engineered islet cell. In some embodiments, both alleles of CIITA are knocked out.
- engineered islet cells have regulated or modulated (e.g. increase) expression of a tolerogenic factor, such as CD47.
- the tolerogenic factor is one or more of DUX4, B2M-HLA-E, CD16, CD52, CD47, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, CCL21, MFGE8, SERPINB9, CD35, IL-39, CD16 Fc Receptor, IL15-RF, and H2- M3, or any combination thereof.
- the one or more tolerogenic factors are selected from the group consisting of CD 16, CD24, CD35, CD39, CD46, CD47, CD52, CD55, CD59, CD64, CD200, CCL22, CTLA4-Ig, Cl inhibitor, FASL, IDO1, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, IL-10, IL-35, PD-L1, SERPINB9, CCL21, MFGE8, DUX4, B2M-HLA-E, CD27, IL-39, CD16 Fc Receptor, IL15-RF, H2-M3 (HLA-G), A20/TNFAIP3, CR1, HLA-F, and MANF.
- the increased or overexpressed tolerogenic factor is or includes increased expression of CCL21, PD-L1, FasL, Serpinb9, H2-M3 (HLA-G), CD47, CD200, and Mfge8.
- the tolerogenic factor is CD47 and the engineered islet cell has increased expression of CD47.
- the tolerogenic factor is PD-L1 and the engineered islet cell includes increased expression of PD-LL
- the tolerogenic factor is HLA-E and the engineered islet cell includes increased expression of HLA- E.
- the tolerogenic factor is HLA-G and the engineered islet includes increased expression of HLA-G.
- the tolerogenic factor is expressed as an exogenous polynucleotide or transgene in the genome of the engineered islet cell.
- the exogenous polynucleotide or transgene is integrated or inserted into a genome locus of the cells, such as a safe harbor locus.
- the genomic locus is an ABO, CCR5, CLYBL, CXCR4, F3, FUT1, HMGB1, KDM5D, LRP1, MICA, MICB, RHD, ROSA26, or SHS231 locus.
- engineered islet cells e.g. iPSC-derived islet cells having (1) reduced expression of MHC I and/or MHC II; and (2) a transgene comprising CD47 and a safety switch inserted at a safe harbor locus, wherein the safe harbor locus is selected from the group consisting of an AAVS1, ABO, CCR5, CLYBL, CXCR4, F3, FUT1, HMGB1, KDM5D, LRP1, MICA, MICB, RHD, ROSA26, and SHS231 locus.
- a safe harbor locus is selected from the group consisting of an AAVS1, ABO, CCR5, CLYBL, CXCR4, F3, FUT1, HMGB1, KDM5D, LRP1, MICA, MICB, RHD, ROSA26, and SHS231 locus.
- modified pluripotent stem cells having (1) reduced expression of MHC I and/or MHC II; and (2) a transgene comprising CD47 and HSVtk flanked by CLYBL homology arms, wherein the transgene is inserted at the CLYBL locus.
- the modified pluripotent stem cell has B2M and/or CIITA knockout.
- the B2M and/or CIITA knockout occur in both alleles.
- an engineered islet cell provided herein comprises a safety switch.
- the introduction of safety switches improves the safety of cell therapies developed using hypoimmunogenic cells (HIP cells, e.g., engineered islet cells).
- HIP cells hypoimmunogenic cells
- a feature of the HIP cells described herein is the inducible expression of one or more immune regulatory (immunosuppressive) factors.
- an immunosuppressive factor (also referred to herein as “an hypoimmunity factor”) includes, but is not limited to, CD47, CD24, CD200, HLA-G, HLA-E, HLA-C, HLA-E heavy chain, PD-L1, IDO1, CTLA4-Ig, Cl -Inhibitor, IL-10, IL-35, FASL, Serpmb9, CC121, and Mfge8.
- an immunosuppressive factor is CD47.
- the regulatable or inducible expression of an immunosuppressive factor functions to control an immune response by a recipient subject to an engrafted hypoimmunogenic cell.
- Described herein are methods for the expression of an immunosuppressive factor that requires a mechanism to “turn-off’ expression of the immune regulatory protein in a controlled manner. Also described are engineered islet cells possessing controllable expression of one or more immunosuppressive factors. In some cases, the cells overexpress one or more immunosuppressive factors and can be induced to downregulate expression of the one or more immunosuppressive factors. As such, the cells are no longer hypoimmunogenic and are recognized by the recipient's immune cells for cell death.
- the hypoimmunity of engineered islet cells that are introduced to a recipient subject is achieved through the overexpression of an immunosuppressive molecule including hypoimmunity factors and complement inhibitors accompanied with the repression or genetic disruption of the HLA-I and HLA-II loci.
- an immunosuppressive molecule including hypoimmunity factors and complement inhibitors accompanied with the repression or genetic disruption of the HLA-I and HLA-II loci.
- These modifications cloak the cell from the recipient immune system's effector cells that are responsible for the clearance of infected, malignant or non-self cells, such as T cells, B cells, NK cells and macrophages. Cloaking of a cell from the immune system allows for existence and persistence of allogeneic cells within the body. Controlled removal of the engineered cells from the body is crucial for patient safety and can be achieved by uncloaking the cells from the immune system.
- Uncloaking serves as a safety switch and can be achieved through the downregulation of the immunosuppressive molecules or the upregulation of immune signaling molecules.
- the level of expression of any of the immunosuppressive molecules described can be controlled on the protein level, mRNA level, or DNA level in the cells.
- the level of expression of any of the immune signaling molecules described can be controlled on the protein level, mRNA level, or DNA level in the cells.
- any of the safety switch methods described are used to decrease the level of an immunosuppressive factor in the cells such that the lower level of the immunosuppressive factor is below a threshold level.
- the level of the immunosuppressive factor in the cells is decreased by about 10-fold, 9-fold, 8-fold, 7-fold, 6-fold, 5-fold, 4-fold, 3-fold, 2- fold, 1- fold or 0.5-fold below a threshold level of expression.
- the level of the immunosuppressive factor in the cells is decreased by about 10-fold to 5-fold, 10-fold to
- the threshold level of expression of the immunosuppressive factor is established based on the expression of such factor in an induced pluripotent stem cell. In some embodiments, the threshold level of the immunosuppressive factor expression is established based on the expression level of the immunosuppressive factor in a corresponding hypoimmune cell, such as any of the engineered islet cells described herein.
- transcriptional regulation of immunosuppressive factors through employing inducible promoters provides the ability to turn expression of the switch on or off at will through the addition or removal of small molecules, such as, but not limited to, doxycycline. Genetic disruption via targeted nuclease activity can eliminate expression of the immunosuppressive factor to uncloak the cells as well.
- Exemplary safety switches are described in WO2021146627A1, the content of which is herein incorporated by reference in its entirety.
- any of the above engineered islet cells further have regulated or modulated (e.g. reduced or eliminated) expression of CD142.
- the regulated or modulated expression of CD 142 is due to gene editing in which the DNA of the CD 142 gene loci has been edited to delete genomic DNA.
- the engineered islet cell has an edit to delete genomic DNA of CD 142 and is thus reduced or eliminated for expression of CD 142.
- the CD 142 gene is knocked out in the engineered islet cell. In some embodiments, both alleles of B2M are knocked out.
- any of the above engineered islet cells further have regulated or modulated (e.g. increased) expression of one or more complement inhibitor.
- the one or more complement inhibitors is any one of CD46, CD59 and CD55 or is a combination thereof (e.g. CD46 and CD59 or CD46, CD59 and CD55).
- the one or more complement inhibitor is expressed as an exogenous polynucleotide(s) or transgene(s) in the genome of an engineered islet cell.
- the exogenous polynucleotide(s) or transgene(s) is integrated or inserted into a genome locus of the cells, such as a safe harbor locus.
- the genomic locus is an ABO, CCR5, CLYBL, CXCR4, F3, FUT1, HMGB1, KDM5D, LRP1, MICA, MICB, RHD, ROSA26, or SHS231 locus.
- the exogenous polynucleotide or transgene is expressed at the same or a different locus from CD47 and/or from a suicide gene.
- the methods used to differentiate the engineered islet cells are known by one skilled in the ait. Such methods are described, for example, in W02019018818, US8507274, US10030229, US10190096, US10253298, US10443042, W02016100925, WO2019217493, US7510876, US8216836, US8633024, US8647873, US 10421942, US9404086, US20190359943, US 10358628, US8633024, US8647873, US9222069, US10465162, US10370645, US9725699, US10253298, US9499795, US9650610, US9062290, US10494609, US20210060083, US8129182, US86O3811, US9328331, US9012218, US9109245, US9982235, US9988604, US10358628, US10138465,
- stage 1 refers to the first step in the differentiation process, the differentiation of pluripotent stem cells into cells expressing markers characteristic of definitive endoderm cells.
- Stage 2 refers to the second step, the differentiation of cells expressing markers characteristic of definitive endoderm cells into cells expressing markers characteristic of gut tube cells.
- Stage 3 refers to the third step, the differentiation of cells expressing markers characteristic of gut tube cells into cells expressing markers characteristic of early pancreas progenitor cells.
- Stage 4 refers to the fourth step, the differentiation of cells expressing markers characteristic of early pancreas progenitor cells into cells expressing markers characteristic of pancreatic progenitor cell.
- Stage 5 refers to the fifth step, the differentiation of cells expressing markers characteristic of pancreatic progenitor cells into cells expressing markers characteristic of pancreatic endoderm cells and/or pancreatic endocrine progenitor cells. It should be appreciated, however, that not all cells in a particular population progress through these stages at the same rate, i.e., some cells may have progressed less, or more, down the differentiation pathway than the majority of cells present in the population.
- a definitive endoderm cell is a cell that bears the characteristics of cells arising from the epiblast during gastrulation and which form the gastrointestinal tract and its derivatives.
- Definitive endoderm cells express at least one of the following markers: FOXA2 (also known as hepatocyte nuclear factor 3 (“HNF30”)), GATA4, SOX17, CXCR4, Brachyury, Cerberus, OTX2, goosecoid, C-Kit, CD99, and MIXL1. Markers characteristic of the definitive endoderm cells include CXCR4, FOXA2 and SOX17. Thus, definitive endoderm cells may be characterized by their expression of CXCR4, FOXA2 and SOX17. In addition, depending on the length of time cells are allowed to remain in Stage 1, an increase in HNF4a may be observed.
- FOXA2 also known as hepatocyte nuclear factor 3 (“HNF30”)
- GATA4 also known as hepatocyte nuclear factor 3 (“HNF30”)
- SOX17 also known as hepatocyte nuclear factor 3 (“HNF30”)
- CXCR4 also known as hepatocyte nuclear factor 3 (“HNF30”)
- GATA4 also known as hepatocyte nuclear
- gut tube cells are cells derived from definitive endoderm that can give rise to all endodermal organs, such as lungs, liver, pancreas, stomach, and intestine.
- Gut tube cells may be characterized by their substantially increased expression of HNF4a over that expressed by definitive endoderm cells. For example, a ten to forty fold increase in mRNA expression of HNF4a may be observed during Stage 2.
- early pancreas progenitor cells refer to endoderm cells that give rise to the esophagus, lungs, stomach, liver, pancreas, gall bladder, and a portion of the duodenum.
- Early pancreatic progenitor cells express at least one of the following markers: PDX1, FOXA2, CDX2, SOX2, and HNF4a.
- Early pancreatic progenitor cells may be characterized by an increase in expression of PDX1, compared to gut tube cells. For example, greater than fifty percent of the cells in Stage 3 cultures typically express PDX1.
- pancreatic progenitor cells refer to cells that express at least one of the following markers: PDX1, NKX6.1, HNF6, NGN3, SOX9, PAX4, PAX6, ISL1, gastrin, FOXA2, PTFla, PROXI and HNF4a.
- Pancreatic progenitor cells may be characterized as positive for the expression of PDX1, NKX6.1, and SOX9.
- a pancreatic endoderm cell (also sometimes called a pancreatic endocrine progenitor cells) is a cell capable of becoming a pancreatic hormone expressing cell.
- Pancreatic endoderm cells express at least one of the following markers: NGN3; NKX2.2; NeuroDl; ISL1; PAX4; PAX6; or ARX.
- Pancreatic endoderm cells may be characterized by their expression of NKX2.2 and NeuroDl.
- Islet cells as provided herein can be generated, e.g., from induced pluripotent stem cells (iPSCs).
- iPSCs induced pluripotent stem cells
- engineered islet cells as provided herein are derived from induced pluripotent stem cells.
- iPSCs are derived from PBMCs from a subject.
- miPSCs for murine cells or hiPSCs for human cells
- miPSCs for murine cells
- hiPSCs for human cells
- the first induction performed was done from mouse embryonic or adult fibroblasts using the viral introduction of four transcription factors, Oct3/4, Sox2, c-Myc and Klf4; see Takahashi and Yamanaka Cell 126:663-676 (2006), hereby incorporated by reference in its entirety and specifically for the techniques outlined therein. Since then, a number of methods have been developed; see Seki et al, World J.
- iPSCs are generated by the transient expression of one or more reprogramming factors" in the host cell, usually introduced using episomal vectors. Under these conditions, small amounts of the cells are induced to become iPSCs (in general, the efficiency of this step is low, as no selection markers are used). Once the cells are “reprogrammed,” and become pluripotent, they lose the episomal vector(s) and produce the factors using the endogeneous genes.
- the number of reprogramming factors that can be used or are used can vary.
- the efficiency of the transformation of the cells to a pluripotent state goes down, as well as the “pluripotency,” e.g., fewer reprogramming factors may result in cells that are not fully pluripotent but may only be able to differentiate into fewer cell types.
- a single reprogramming factor, OCT4, is used.
- two reprogramming factors, OCT4 and KLF4, are used.
- three reprogramming factors, OCT4, KLF4 and SOX2, are used.
- four reprogramming factors, OCT4, KLF4, SOX2 and c-Myc are used.
- 5, 6 or 7 reprogramming factors can be used selected from SOKMNLT; SOX2, OCT4 (POU5F1), KLF4, MYC, NANOG, LIN28, and SV40L T antigen.
- these reprogramming factor genes are provided on episomal vectors such as are known in the art and commercially available.
- cells used for transfecting the one or more reprogamming factors are non-pluripotent stem cells.
- iPSCs are made from non-pluripotent cells such as, but not limited to, PBMCs, blood cells, fibroblasts, etc., by transiently expressing the reprogramming factors as described herein.
- the non-pluripotent cells, such as PBMCs are obtained or isolated from one or more individual subjects or donors prior to reprogamming the cells.
- iPSCs are made from a pool of isolated non- pluripotent stems cells, e.g., PBMCs, obtained from one or more (e.g., two or more, three or more, four or more, five or more, ten or more, twenty or more, fifty or more, or one hundred or more) different donor subjects.
- PBMCs isolated non- pluripotent stems cells
- the non-pluripotent cells such as PBMCs
- PBMCs are isolated or obtained from a plurality of different donor subjects (e.g., two or more, three or more, four or more, five or more, ten or more, twenty or more, fifty or more, or one hundred or more), pooled together in a batch, reprogrammed as iPSCs and are engineered in accord with the provided methods.
- donor subjects e.g., two or more, three or more, four or more, five or more, ten or more, twenty or more, fifty or more, or one hundred or more
- iPSCs reprogrammed as iPSCs
- iPSCs are derived from, such as by transiently transfecting one or more reprogramming factors into cells from a pool of non-pluripotent cells (e.g., PBMCs) from a subject.
- non-pluripotent cells e.g., PBMCs
- non-pluripotent cells are harvested from one or a plurality of individuals, and in some instances, the non-pluripotent cells (e.g., PBMCs) or a pool of non-pluripotent cells (e.g., PBMCs) are cultured in vitro and transfected with one or more reprogramming factors to induce generation of iPSCs.
- non- pluripotent cells e.g., PBMCs
- a pool of non-pluripotent cells e.g., PBMCs
- engineered iPSCs or a pool of engineered iPSCs are then subjected to a differentiation process for differentiation into any cells of an organism and tissue.
- hypoimmunogenicity is assayed using a number of techniques as exemplified in Figure 13 and Figure 15 of WO2018132783. These techniques include transplantation into allogeneic hosts and monitoring for hypoimmunogenic pluripotent cell growth (e.g., teratomas) that escape the host immune system. In some instances, hypoimmunogenic pluripotent cell derivatives are transduced to express luciferase and can then followed using bioluminescence imaging.
- T cell and/or B cell response of the host animal to such cells are tested to confirm that the cells do not cause an immune reaction in the host animal.
- T cell responses can be assessed by Elispot, ELISA, FACS, PCR, or mass cytometry (CYTOF).
- B cell responses or antibody responses are assessed using FACS or Luminex.
- the cells may be assayed for their ability to avoid innate immune responses, e.g., NK cell killing, as is generally shown in Figures 14 and 15 of WO2018132783.
- the immunogenicity of the cells is evaluated using T cell immunoassays such as T cell proliferation assays, T cell activation assays, and T cell killing assays recognized by those skilled in the art.
- T cell proliferation assay includes pretreating the cells with interferon-gamma and coculturing the cells with labelled T cells and assaying the presence of the T cell population (or the proliferating T cell population) after a preselected amount of time.
- the T cell activation assay includes coculturing T cells with the cells outlined herein and determining the expression levels of T cell activation markers in the T cells.
- In vivo assays can be performed to assess the immunogenicity of the cells outlined herein.
- the survival and immunogenicity of engineered or modified iPSCs is determined using an allogeneic humanized immunodeficient mouse model.
- the engineered or modified iPSCs are transplanted into an allogeneic humanized NSG-SGM3 mouse and assayed for cell rejection, cell survival, and teratoma formation.
- grafted engineered iPSCs or differentiated cells thereof display long-term survival in the mouse model.
- pluripotency is assayed by the expression of certain pluripotency-specific factors as generally described herein and shown in Figure 29 of WO2018132783. Additionally or alternatively, the pluripotent cells are differentiated into one or more cell types as an indication of pluripotency.
- engineered pluripotent stem cells (engineered iPSCs) have been generated, they can be maintained in an undifferentiated state as is known for maintaining iPSCs.
- the cells can be cultured on Matrigel using culture media that prevents differentiation and maintains pluripotency.
- they can be in culture medium under conditions to maintain pluripotency.
- any of the iPSCs described herein can be differentiated into any cells of an organism and tissue.
- engineered cells that are differentiated into different cell types from iPSCs for subsequent transplantation into recipient subjects.
- Differentiation can be assayed as is known in the art, generally by evaluating the presence of cell-specific markers.
- the differentiated engineered (e.g., hypoimmunogenic) pluripotent cell derivatives can be transplanted using techniques known in the art that depends on both the cell type and the ultimate use of these cells. Exemplary types of differentiated cells and methods for producing the same are described below.
- the iPSCs may be differentiated to any type of cell described herein, including any described in Section II.C.3.
- the iPSCs are differentiated into beta islet cells.
- cells such as non-pluripotent cells (e.g., PBMCs) from a subject are isolated or obtained, generated into iPSCs in which the iPSCs are then engineered to contain modifications (e.g., genetic modifications) described herein and then differentiated into a desired cell type.
- modifications e.g., genetic modifications
- islet cells includes islet cells made from iPSCs (sometimes termed iPSC-derived pseudo-islet cells, pseudo-islet cells, or p-islets).
- iPSCs sometimes termed iPSC-derived pseudo-islet cells, pseudo-islet cells, or p-islets.
- a method of generating insulin-producing islet cells comprising: providing a stem cell (e.g. modified stem cell, such as modified induced pluripotent stem cell (iPSC), or a stem cell that does not comprise one or more modifications); providing serum-free media; contacting the stem cell with a TGF /Activin agonist and/or a glycogen synthase kinase 3 (GSK) inhibitor and/or WNT agonist for an amount of time sufficient to form a definitive endoderm cell; contacting the definitive endoderm cell with a FGFR2b agonist for an amount of time sufficient to form a primitive gut tube cell; contacting the primitive gut tube cell with an RAR agonist, a rho kinase inhibitor, a Smoothened antagonist, a FGFR2b agonist, a protein kinase C activator, and/or a BMP type 1 receptor inhibitor for an amount of time sufficient to form an early pancreas progenitor cell; in
- the serum-free media comprises one or more selected from the group consisting of: MCDB131, glucose, NaHCOs, BSA, ITS- X, Glutamax, vitamin C, penicillin-streptomycin, CMRL 10666, FBS, Heparin, NEAA, trace elements A, trace elements B, or ZnS04.
- the TGF /Activin agonist is Activin A; the glycogen synthase kinase 3 (GSK) inhibitor or the WNT agonist is CHIR; the FGFR2b agonist is KGF; the Smoothened antagonist or hedgehog pathway inhibitor is SANT-1 ; the FGF family member/FGFR2b agonist is KGF; the RAR agonist is RA; the protein kinase 3 activator is TPPB; the BMP inhibitor is LDN; the rho kinase inhibitor is Y27632; the Alk5 inhibitor/TGF-b receptor inhibitor is Alk5i; the thyroid hormone is T3; or the gamma secretase inhibitor is XXI.
- the TGF /Activin agonist is Activin A.
- the concentration of Activin A is between 50 ng/ml-150 ng/ml. In certain embodiments, the concentration of Activin A is 50 ng/ml, 55 ng/ml, 60 ng/ml, 65 ng/ml, 70 ng/ml, 75 ng/ml, 80 ng/ml, 85 ng/ml, 90 ng/ml, 95 ng/ml, 100 ng/ml, 105 ng/ml, 110 ng/ml, 115 ng/ml, 120 ng/ml, 125 ng/ml, 130 ng/ml, 135 ng/ml, 140 ng/ml, 145 ng/ml, or 150 ng/ml.
- the concentration of Activin A is between 50 ng/ml-60 ng/ml, 55 ng/ml-65 ng/ml, 60 ng/ml-70 ng/ml, 65 ng/ml-75 ng/ml, 70 ng/ml-80 ng/ml, 75 ng/ml-85 ng/ml, 80 ng/ml- 90 ng/ml, 85 ng/ml-95 ng/ml, 90 ng/ml- 100 ng/ml, 95 ng/ml- 105 ng/ml, 100 ng/ml- 110 ng/ml, 105 ng/ml- 115 ng/ml, 110 ng/ml- 120 ng/ml, 115 ng/ml- 125 ng/ml, 120 ng/ml- 130 ng/ml, 125 ng/ml-135 ng/ml, 130 ng/ml-140 ng/m
- the glycogen synthase kinase 3 (GSK) inhibitor or the WNT agonist is CHIR.
- the concentration of CHIR is between 0.5 pM and 5 pM.
- the concentration of the CHIR is 0.5 pM, 1.0 pM, 1.5 pM, 2.0 pM, 2.5 pM, 3.0 pM, 3.5 pM, 4.0 pM, 4.5 pM, or 5.0 pM.
- the concentration of CHIR is between 0.5 pM-1.5 pM, 1.0 pM-2.0 pM, 1.5 pM-2.5 pM, 2.0 pM- 3.0 pM, 2.5 pM-3.5 pM, 3.0 pM-4.0 pM, 3.5 pM-4.5 pM, or 4.0 pM-5.0 pM. In a specific embodiment, the concentration of CHIR is 3.0 pM.
- the FGFR2b agonist is KGF.
- the concentration of KGF is between 5 ng/ml-100 ng/ml. In certain embodiments, the concentration of KGF is 5 ng/ml, 10 ng/ml, 15 ng/ml, 20 ng/ml, 25 ng/ml, 30 ng/ml, 35 ng/ml, 40 ng/ml, 45 ng/ml, 50 ng/ml, 55 ng/ml, 60 ng/ml, 65 ng/ml, 70 ng/ml, 75 ng/ml, 80 ng/ml, 85 ng/ml, 90 ng/ml, 95 ng/ml, or 100 ng/ml.
- the concentration of KGF is between 5 ng/ml- 15 ng/ml, 10 ng/ml-20 ng/ml, 15 ng/ml-25 ng/ml, 20 ng/ml-30 ng/ml, 25 ng/ml - 35 ng/ml, 30 ng/ml-40 ng/ml, 35 ng/ml-45 ng/ml, 40 ng/ml-50 ng/ml, 45 ng/ml-55 ng/ml, 50 ng/ml-60 ng/ml, 55 ng/ml-65 ng/ml, 60 ng/ml-70 ng/ml, 65 ng/ml-75 ng/ml, 70 ng/ml-80 ng/ml, 75 ng/ml-85 ng/ml, 80 ng/ml-90 ng/ml, 85 ng/ml-95 ng/ml, 90 ng/ml-100 ng
- the Smoothened antagonist is SANT-1.
- the concentration of SANT-1 is between 0.05 pM and 0.50 p M. In certain embodiments, the concentration of the SANT-1 is 0.05 pM, 0.10 pM, 0.15 pM, 0.20 pM, 0.25 pM, 0.30 pM, 0.35 pM, 0.40 pM, 0.45 pM, or 0.5 pM.
- the concentration of SANT-1 is between 0.05 pM-0.15 pM, 0.10 pM-0.20 pM, 0.15 pM-0.25 pM, 0.20 pM-0.30 pM, 0.25 pM-0.35 pM, 0.30 pM-0.40 pM, 0.35 pM-0.45 pM, or 0.40 pM-0.50 pM. In a specific embodiment, the concentration of SANT-1 is 0.25 pM.
- the RAR agonist is retinoic acid (RA).
- the concentration of RA is between 0.05 pM and 2.5 pM. In certain embodiments, the concentration of RA is 0.05 pM, 0.1 pM, 0.15 pM, 0.2 pM, 0.5 pM, 1.0 pM, 1.5 pM, 2.0 pM, or 2.5 pM.
- the concentration of RA is between 0.005 pM-0.15 pM, 0.10 pM-0.2 pM, 0.15 pM-0.5 pM, 0.2 pM-1.0 pM, 0.5 pM-1.5 pM, 1.0 pM-2.0 pM, or 1.5 pM-2.5 pM.
- the concentration of RA is 0.10 pM.
- the concentration of RA is 2.0 pM.
- the protein kinase C activator is TPPB.
- the concentration of TPPB is between 0.05 pM and 0.50 pM. In certain embodiments, the concentration of the TPPB is 0.05 pM, 0.10 pM, 0.15 pM, 0.20 pM, 0.25 pM, 0.30 pM, 0.35 pM, 0.40 pM, 0.45 pM, or 0.5 pM.
- the concentration of TPPB is between 0.05 pM-0.15 pM, 0.10 pM-0.20 pM, 0.15 pM-0.25 pM, 0.20 pM-0.30 pM, 0.25 pM-0.35 pM, 0.30 pM-0.40 pM, 0.35 pM-0.45 pM, or 0.40 pM-0.50 pM. In a specific embodiment, the concentration of TPPB is 0.20 pM.
- the BMP type 1 receptor inhibitor is LDN193189.
- the concentration of LDN193189 is between 0.05 pM and 0.50 pM.
- the concentration of the LDN193189 is 0.05 pM, 0.10 pM, 0.15 pM, 0.20 pM, 0.25 pM, 0.30 pM, 0.35 pM, 0.40 pM, 0.45 pM, or 0.5 pM.
- the concentration of LDN193189 is between 0.05 pM-0.15 pM, 0.10 pM-0.20 pM, 0.15 pM-0.25 pM, 0.20 pM-0.30 pM, 0.25 pM-0.35 pM, 0.30 pM-0.40 pM, 0.35 pM-0.45 pM, or 0.40 pM- 0.50 pM. In a specific embodiment, the concentration of LDN193189 is 0.20 pM.
- the Alk5 inhibitor is Alk5i.
- the concentration of Alk5i is between 5.0 pM and 15 pM. In certain embodiments, the concentration of Alk5i is 5.0 pM, 6.0 pM, 7.0 pM, 8.0 pM, 9.0 pM, 10.0 pM, 11.0 pM, 12.0 pM, 13.0 pM, 14.0 pM, or 15.0 pM.
- the concentration of Alk5i is between 5.0 pM-7.0 pM, 6.0 pM-8.0 pM, 7.0 pM-9.0 pM, 8.0 pM-10.0 pM, 9.0 pM-11.0 pM, 10.0 pM-12.0 pM, 11.0 pM-13.0 pM, 12.0 pM-14.0 pM, or 13.0 pM-15.0 pM.
- the concentration of Alk5i is 10.0 pM.
- latrunculin A is utilized to chemically depolymerize the actin cytoskeleton.
- the concentration of latrunculin A is 0.5 pM and 1.5 pM.
- the concentration of latrunculin A is 0.5 pM, 0.6 pM, 0.7 pM, 0.8 pM, 0.9 pM, 1.0 pM, 1.1 pM, 1.2 pM, 1.3 pM, 1.4 pM, or 1.5 pM.
- the concentration of latrunculin A is between 0.5 pM-0.7 pM, 0.6 pM-0.8 pM, 0.7 pM-0.9 pM, 0.8 pM-1.0 pM, 0.9 pM-1.1 pM, 1.0 pM-1.2 pM, 1.1 pM-1.3 pM, 1.2 pM-1.4 pM, or 1.3 pM-1.5 pM.
- the concentration of latrunculin A is 1.0 pM.
- the thyroid hormone is T3.
- the concentration of T3 is between 0.1 pM and 2 pM. In certain embodiments, the concentration of T3 is 0.1 pM, 0.2 pM, 0.3 pM, 0.4 pM, 0.5 pM, 0.6 pM, 0.7 pM, 0.8 pM, 0.9 pM, 1.0 pM, 1.1 pM, 1.2 pM, 1.3 pM, 1.4 pM, 1.5 pM, 1.6 pM, 1.7 pM, 1.8 pM, 1.9 pM, or 2.0 pM.
- the concentration of T3 is between 0.1 pM-0.3 pM, 0.2 pM-0.4 pM, 0.3 pM-0.5 pM, 0.4 pM-0.6 pM, 0.5 pM-0.7 pM, 0.6 pM-0.8 pM, 0.7 pM-0.9 pM, 0.8 pM-1.0 pM, 0.9 pM-1.1 pM, 1.0 pM-1.2 pM, 1.1 pM-1.3 pM, 1.2 pM-1.4 pM, 1.3 pM-1.5 pM, 1.4 pM-1.6 pM, 1.5 pM-1.7 pM, 1.6 pM-1.8 pM, 1.7 pM-1.9 pM, or 1.8 pM -2.0 pM.
- the concentration of T3 is 1.0 pM.
- the gamma secretase inhibitor is XXI. In certain embodiments, the concentration of XXI is between 0.1 pM and 2 pM.
- the concentration of XXI is 0.1 pM, 0.2 pM, 0.3 pM, 0.4 pM, 0.5 pM, 0.6 pM, 0.7 pM, 0.8 pM, 0.9 pM, 1.0 pM, 1.1 pM, 1.2 pM, 1.3 pM, 1.4 pM, 1.5 pM, 1.6 pM, 1.7 pM 1.8 pM, 1.9 pM, or 2.0 pM.
- the concentration of XXI is between 0.1 pM-0.3 pM, 0.2 pM- 0.4 pM, 0.3 pM-0.5 pM, 0.4 pM-0.6 pM, 0.5 pM-0.7 pM, 0.6 pM-0.8 pM, 0.7 pM-0.9 pM, 0.8 pM-l .O pM, 0.9 pM-l .l pM, 1.0 pM-l .2 pM, 1.1 pM-1.3 pM, 1.2 pM-1.4 pM, 1.3 pM-1.5 pM, 1.4 pM-1.6 pM, 1.5 pM-1.7 pM, 1.6 pM-1.8 pM, 1.7 pM-1.9 pM, or 1.8 pM-2.0 pM.
- the concentration of XXI is 1.0 pM.
- the methods herein detail a differentiation protocol for generating highly functional SC- P cells.
- the methods provided herein comprise six stages that attempt to recreate phases of pancreatic organogenesis by activating and repressing specific developmental pathways with growth factors and small molecules in serum-free medium.
- hPSCs are seeded onto Matrigel-coated TCP plates at a density of 0.8 x 105 cells/cm2 and cultured in medium.
- the methods provided herein comprise six stages of stem cell differentiation.
- Stage 1 comprises incubating a HPSC of Stage 0 in media comprising Activin A and CHIR for about 24 hours followed by about 3 days of incubating the cells in media comprising Activin A in the absence of CHIR.
- Stage 2 comprises incubating the Stage 1 cells for 2 days in media comprising KGF.
- the CHIR is CH1R99021.
- Stage 3 comprises incubating Stage 2 cells for 2 days in media comprising KGF, LDN193189, TPPB, RA (high), and SANT1.
- Stage 4 comprises incubating Stage 3 cells for about 4 days in media comprising KGF, LDN193189, TPPB, RA (low), and SANT1.
- Stage 5 comprises incubating the Stage 4 cells in media comprising XXI, Alk5i, T3, SANT1, and RA for 7 days. Additionally, latrunculin A is added to the media for about the first 24 hours of incubation.
- Stage 6 comprises incubating the cells in an enriched serum-free medium which allows the SC-P cells the time needed to mature before they become glucose responsive.
- the methods provided herein generate islet cells that function better (undergoing glucose- stimulated insulin secretion) than cells in the published literature (Pagliuca et al. Cell 2014).
- the amount of time sufficient to form a definitive endoderm cell, a primitive gut tube cell, an early pancreas progenitor cell, a pancreatic progenitor cell, an endoderm cell, or an islet cell is between about 1 day and about 15 days.
- islet cells can be useful as a cellular therapy for diabetes.
- the presently disclosed method enhances differentiation of human pluripotent stem cells to insulinproducing islet cells. This process is modified from a previously described 6-step differentiation protocol published by Pagliuca et al. Cell 2014. Using the methods disclosed herein, cells that can respond to glucose appropriately to near islet-like levels have been generated, demonstrating both a first phase and second phase response.
- stage 6 shorten stage 3 to 1 day; (2) allow for TGFbeta signaling in stage 6 by removal of Alk5 inhibitor II (3) remove T3 from stage 6; (4) perform stage 6 in a serum-free basal media; and (5) break apart and reaggregate clusters at the beginning of stage 6.
- a cell to be engineered or modified is a stem or progenitor cell that is capable of being differentiated (e.g., the stem cell is totipotent, pluripotent, or multipotent).
- a cell is isolated from neonatal tissue.
- a cell is an induced pluripotent stem cell derived from somatic cells (e.g., skin or blood cells) and reprogrammed into an embryonic-like pluripotent state.
- an induced pluripotent stem cell is derived from a fibroblast.
- cells that are modified as provided herein are pluripotent stems cells or are cells differentiated from pluripotent stem cells.
- a cell may be a vertebrate cell, for example, a mammalian cell, such as a human cell or a mouse cell.
- a cell may also be a vertebrate stem cell, for example, a mammalian stem cell, such as a human stem cell or a mouse stem cell.
- a cell or stem cell is amenable to modification.
- a cell or stem cell, or a cell derived from such a stem cell has or is believed to have therapeutic value, such that a cell or stem cell or a cell derived or differentiated from such stem cell may be used to treat a disease, disorder, defect or injury in a subject in need of treatment for same.
- cells that are modified as provided herein are modified pluripotent stem cells (e.g., modified iPSCs).
- modified pluripotent stem cells e.g., modified iPSCs
- the generation of mammalian (e.g., mouse and human) pluripotent stem cells is generally known in the art.
- iPSCs mammalian pluripotent stem cells
- miPSCs for murine cells
- iPSCs are generated by the transient expression of one or more reprogramming factors" in the host cell, usually introduced using episomal vectors. Under these conditions, small amounts of the cells are induced to become iPSCs (in general, the efficiency of this step is low, as no selection markers are used). Without wishing to be bound by theory, it is believed that once the cells are "reprogrammed", and become pluripotent, they lose the episomal vector(s) and produce the factors using the endogenous genes.
- the number of reprogramming factors that can be used or are used can vary. Commonly, when fewer reprogramming factors are used, the efficiency of the transformation of the cells to a pluripotent state goes down, as well as the "pluripotency", e.g., fewer reprogramming factors may result in cells that are not fully pluripotent but may only be able to differentiate into fewer cell types.
- a single reprogramming factor, OCT4 is used.
- two reprogramming factors, OCT4 and KLF4 are used.
- three reprogramming factors, OCT4, KLF4 and SOX2 are used.
- reprogramming factors OCT4, K.LF4, SOX2 and c-Myc
- 5 reprogramming factors can be used selected from SOKMNLT; SOX2, OCT4 (POU5F1), KLF4, MYC, NANOG, LIN28, and SV40L T antigen.
- SOKMNLT SOX2, OCT4 (POU5F1), KLF4, MYC, NANOG, LIN28, and SV40L T antigen.
- these reprogramming factor genes are provided on episomal vectors such as are known in the art and commercially available.
- hosts cells used for transfecting the one or more reprogramming factors are non-pluripotent stem cells.
- iPSCs are made from non-pluripotent cells such as, but not limited to, blood cells, fibroblasts, etc., by transiently expressing the reprogramming factors as described herein.
- the non-pluripotent cells, such as fibroblasts are obtained or isolated from one or more individual subjects or donors prior to reprogramming the cells.
- iPSCs are made from a pool of isolated non-pluripotent stems cells, e.g., fibroblasts, obtained from one or more (e.g.
- the non- pluripotent cells such as fibroblasts
- the non- pluripotent cells are isolated or obtained from a plurality of different donor subjects (e.g., two or more, three or more, four or more, five or more, ten or more, twenty or more, fifty or more, or one hundred or more), pooled together in a batch, reprogrammed as iPSCs, and are modified in accord with the provided methods.
- the iPSCs are derived from, such as by transiently transfecting one or more reprogramming factors into cells from a pool of non-pluripotent cells (e.g., fibroblasts) from one or more donor subjects that are different than the recipient subject (e.g., the patient administered the cells).
- the non-pluripotent cells (e.g., fibroblasts) to be induced to iPSCs can be obtained from 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 20, 50, 100 or more donor subjects and pooled together.
- the non-pluripotent cells e.g., fibroblasts
- the non-pluripotent cells can be obtained from 1 or more, 2 or more, 3 or more, 4 or more, 5 or more, 6 or more, 7 or more, 8 or more, 9 or more, 10, or more 20 or more, 50 or more, or 100 or more donor subjects and pooled together.
- the non-pluripotent cells arc harvested from one or a plurality of individuals, and in some instances, the non-pluripotent cells (e.g., fibroblasts) or the pool of non- pluripotent cells (e.g., fibroblasts) are cultured in vitro and transfected with one or more reprogramming factors to induce generation of iPSCs.
- the non- pluripotent cells e.g., fibroblasts
- the pool of non-pluripotent cells e.g., fibroblasts
- the modified iPSCs or a pool of modified iPSCs are then subjected to a differentiation process for differentiation into any cells of an organism and tissue.
- modified iPSCs cells may be assayed for their hypoimmunogenicity and/or retention of pluripotency as is described in WO2016183041 and WO2018132783.
- hypoimmunogenicity is assayed using a number of techniques as exemplified in Figure 13 and Figure 15 of WO2018132783. These techniques include transplantation into allogeneic hosts and monitoring for hypoimmunogenic pluripotent cell growth (e.g., teratomas) that escape the host immune system.
- hypoimmunogenic pluripotent cell derivatives are transduced to express luciferase and can then followed using bioluminescence imaging.
- T cell and/or B cell response of the host animal to such cells are tested to confirm that the cells do not cause an immune reaction in the host animal.
- T cell responses can be assessed by Elispot, ELISA, FACS, PCR, or mass cytometry (CYTOF).
- B cell responses or antibody responses are assessed using FACS or Luminex.
- the cells may be assayed for their ability to avoid innate immune responses, e.g., NK cell killing, as is generally shown in Figures 14 and 15 of WO2018132783.
- the immunogenicity of the cells is evaluated using T cell immunoassays such as T cell proliferation assays, T cell activation assays, and T cell killing assays recognized by those skilled in the art.
- the T cell proliferation assay includes pretreating the cells with interferon-gamma and co-culturing the cells with labelled T cells and assaying the presence of the T cell population (or the proliferating T cell population) after a preselected amount of time.
- the T cell activation assay includes co- culturing T cells with the cells outlined herein and determining the expression levels of T cell activation markers in the T cells.
- In vivo assays can be performed to assess the immunogenicity of the cells outlined herein.
- the survival and immunogenicity of modified iPSCs is determined using an allogeneic humanized immunodeficient mouse model.
- the modified iPSCs are transplanted into an allogeneic humanized NSG-SGM3 mouse and assayed for cell rejection, cell survival, and teratoma formation.
- grafted modified iPSCs or differentiated cells thereof display long-term survival in the mouse model.
- pluripotency is assayed by the expression of certain pluripotency- specific factors as generally described herein and shown in Figure 29 of WO2018132783. Additionally or alternatively, the pluripotent cells are differentiated into one or more cell types as an indication of pluripotency.
- modified pluripotent stem cells Once modified pluripotent stem cells (modified iPSCs) have been generated, they can be maintained in an undifferentiated state as is known for maintaining iPSCs.
- the cells can be cultured on Matrigel using culture media that prevents differentiation and maintains pluripotency.
- they can be in culture medium under conditions to maintain pluripotency.
- any of the pluripotent stem cells described herein can be differentiated into any cells of an organism and tissue.
- modified cells that are differentiated into different cell types from iPSCs for subsequent transplantation into recipient subjects. Differentiation can be assayed as is known in the art, generally by evaluating the presence of cell-specific markers.
- the differentiated modified (e.g., hypoimmunogenic) pluripotent cell derivatives can be transplanted using techniques known in the ail that depends on both the cell type and the ultimate use of these cells. Exemplary types of differentiated cells and methods for producing the same are described above and below.
- iPSCs may be differentiated to any type of cell described herein.
- iPSCs are differentiated into islet cells.
- host cells such as non-pluripotent cells (e.g., fibroblasts) from an individual donor or a pool of individual donors are isolated or obtained, generated into iPSCs in which the iPSCs are then modified to contain modifications (e.g., genetic modifications) described herein and then differentiated into a desired cell type.
- cells as provided herein are islet cells derived from modified iPSCs that contain modifications (e.g., genetic modifications) described herein and that are differentiated into islet cells.
- modifications e.g., genetic modifications
- the methods for differentiation depend on the desired cell type using known techniques.
- cells differentiated into various islet cells may be used for subsequent transplantation or engraftment into subjects (e.g., recipients).
- pancreatic islet cells are derived from the modified pluripotent cells described herein. Useful methods for differentiating pluripotent stem cells into islet cells are described, for example, in U.S. Patent No. 9,683,215; U.S. Patent No. 9,157,062; U.S. Patent No. 8,927,280; U.S. Patent Pub. No. 2021/0207099;
- modified pluripotent cells described herein are differentiated into islet-like cells or islet organoids for transplantation to address T1D.
- Cell systems are a promising way to address T1DM, see, e.g., Ellis et al, Nat Rev Gastroenterol Hepatol. 2017 Oct;14(10):612-628, incorporated herein by reference.
- Pagliuca et al. (Cell, 2014, 159(2):428-39) reports on the successful differentiation of islet cells from hiPSCs, the contents incorporated herein by reference in its entirety and in particular for the methods and reagents outlined there for the large-scale production of functional human islet cells from human pluripotent stem cells).
- the method of producing a population of modified pancreatic islet cells from a population of modified pluripotent cells by in vitro differentiation comprises: (a) culturing the population of modified iPSCs in a first culture medium comprising one or more factors selected from the group consisting insulin-like growth factor, transforming growth factor, FGF, EGF, HGF, SHH, VEGF, transforming growth factor-b superfamily, BMP2, BMP7, a GSK inhibitor, an ALK inhibitor, a BMP type 1 receptor inhibitor, and retinoic acid to produce a population of immature pancreatic islet cells; and (b) culturing the population of immature pancreatic islet cells in a second culture medium that is different than the first culture medium to produce a population of modified pancreatic islet cells.
- the GSK inhibitor is CHIR-99021, a derivative thereof, or a variant thereof. In some instances, the GSK inhibitor is at a concentration ranging from about 2 mM to about 10 mM. In some embodiments, the ALK inhibitor is SB-431542, a derivative thereof, or a variant thereof. In some instances, the ALK inhibitor is at a concentration ranging from about 1 pM to about 10 pM. In some embodiments, the first culture medium and/or second culture medium are absent of animal serum.
- Differentiation is assayed as is known in the art, generally by evaluating the presence of islet cell associated or specific markers, including but not limited to, insulin. Differentiation can also be measured functionally, such as measuring glucose metabolism, see generally Muraro et al., Cell Syst. 2016 Oct 26; 3(4): 385-394.e3, hereby incorporated by reference in its entirety, and specifically for the biomarkers outlined there.
- islet cells Once islet cells are generated, they can be transplanted (either as a cell suspension, cell clusters, or within a permeable or semipermeable device or gel matrix as discussed herein) into the portal vein/liver, the omentum, the gastrointestinal mucosa, the bone marrow, a muscle, or subcutaneous pouches.
- pancreatic islet cells including for use in the present technology are found in W02020/018615, the disclosure is herein incorporated by reference in its entirety.
- a population of modified islet cells such as endothelial cells differentiated from iPSCs derived from one or more individual donors (e.g., healthy donors), are maintained in culture, in some cases expanded, prior to administration.
- the population of engineered islet cells are cryopreserved prior to administration.
- pancreatic islet cell types include, but are not limited to, pancreatic islet progenitor cell, immature pancreatic islet cell, mature pancreatic islet cell, and the like.
- pancreatic cells described herein are administered to a subject to treat diabetes.
- pancreatic islet cells modified as disclosed herein, such as islet cells differentiated from iPSCs derived from one or more individual donors (e.g., healthy donors), secrete insulin.
- a pancreatic islet cell exhibits at least two characteristics of an endogenous pancreatic islet cell, for example, but not limited to, secretion of insulin in response to glucose, and expression of beta cell markers.
- beta cell markers or beta cell progenitor markers include, but are not limited to, c-peptide, Pdxl, glucose transporter 2 (Glut2), HNF6, VEGF, glucokinase (GCK), prohormone convertase (PC 1/3), Cdcpl, NeuroD, Ngn3, Nkx2.2, Nkx6.1, Nkx6.2, Pax4, Pax6, Ptfla, Isll, Sox9, Soxl7, and FoxA2.
- pancreatic islet cells such as islet cells differentiated from iPSCs derived from one or more individual donors (e.g., healthy donors), produce insulin in response to an increase in glucose.
- pancreatic islet cells secrete insulin in response to an increase in glucose.
- islet cells have a distinct morphology such as a cobblestone cell morphology and/or a diameter of about 17 pm to about 25 pm.
- provided modified pluripotent stem cells comprise a modification (e.g., genetic modifications) of one or more target polynucleotide or protein sequences (also interchangeably referred to as a target gene) that regulate (e.g., reduce or eliminate) the expression of either MHC class I molecules, MHC class II molecules, or MHC class I and MHC class II molecules.
- a cell to be modified is an unmodified cell that has not previously been introduced with the one or more modifications.
- a genetic editing system is used to modify one or more target polynucleotide sequences that regulate (e.g., reduce or eliminate) the expression of either MHC class I molecules, MHC class II molecules, or MHC class I and MHC class II molecules.
- the genome of the cell has been altered to reduce or delete components required or involved in facilitating HLA expression, such as expression of MHC class I and/or MHC class II molecules on the surface of the cell.
- expression of a beta-2- microgloublin (B2M), a component of MHC class I molecules, is reduced or eliminated in the cell, thereby reducing or elimination the protein expression (e.g., cell surface expression) of MHC class I by the modified pluripotent stem cells.
- B2M beta-2- microgloublin
- any of the described modifications in the modified pluripotent stem cells that regulate (e.g., reduce or eliminate) expression of one or more target polynucleotide or protein in the modified pluripotent stem cells may be combined with one or more modifications to overexpress a polynucleotide (e.g., tolerogenic factor, such as CD47).
- a polynucleotide e.g., tolerogenic factor, such as CD47
- reduction of MHC class I and/or MHC class II expression can be accomplished, for example, by one or more of the following: (1) directly targeting the MHC class I genes such as the polymorphic HLA alleles (HLA- A, HLA-B, HLA -C) and/or the MHC class II genes such as HLA-DP, HLA-DQ, and/or HLA-DR; (2) removal of B2M, which will reduce surface trafficking of all MHC class I molecules; and/or (3) deletion of one or more components of the MHC enhanceosomes, such as LRC5, RFX-5, RFXANK, RFXAP, IRF1, NF- Y (including NFY-A, NFY-B, NFY-C), and CIITA that are critical for HLA expression.
- reduction of MHC class II also may be accomplished by reducing expression, such as by knocking out the gene encoding CD74 in a cell, which is involved in the formation and transport of MHC
- HLA expression is interfered with.
- HLA expression is interfered with by targeting individual HLAs (e.g., knocking out expression of one or more HLA class I molecules such as HLA-A, HLA-B and/or HLA-C and/or knocking out expression of one or more HLA class I molecules such as HLA-DP, HLA- DQ, and/or HLA-DR), targeting transcriptional regulators of HLA expression (e.g., knocking out expression of NLRC5, CIITA, RFX5, RFXAP, RFXANK, NFY-A, NFY-B, NFY-C and/or IRF- 1), blocking surface trafficking of MHC class I molecules (e.g., knocking out expression of B2M and/or TAPI), and/or targeting with HLA-Razor (see, e.g., W02016183041).
- reduction of HLA class II also may be accomplished by reducing expression, such
- the modified pluripotent stem cells disclosed herein do not express one or more human leukocyte antigens corresponding to MHC class I (e.g., HLA-A, HLA-B and/or HLA-C) and/or MHC class II (e.g., HLA-DP, HLA-DQ, and/or HLA-DR) and are thus characterized as being hypoimmunogenic.
- MHC class I e.g., HLA-A, HLA-B and/or HLA-C
- MHC class II e.g., HLA-DP, HLA-DQ, and/or HLA-DR
- the modified pluripotent stem cells disclosed herein have been modified such that the cells, including any stem cell or a differentiated stem cell prepared therefrom, do not express or exhibit reduced expression of one or more of the following MHC class I molecules: HLA-A, HLA-B and HLA-C.
- one or more of HLA-A, HLA-B and HLA-C may be "knocked-out" of a cell.
- a cell that has a knocked-out HLA-A gene, HLA-B gene, and/or HLA-C gene may exhibit reduced or eliminated expression of each knocked-out gene.
- the modified pluripotent stem cells disclosed herein have been modified such that the cells, including any stem cell or a differentiated stem cell prepared therefrom, do not express or exhibit reduced expression of one
- HLA-DP HLA-DQ
- HLA-DR HLA-DR
- one or more of HLA-DP, HLA-DQ, and HLA-DR may be "knocked-out" of a cell.
- a cell that has a knocked-out HLA-DP gene, HLA-DQ gene and/or HLA-DR gene may exhibit reduced or eliminated expression of each knocked-out gene.
- MHC class I molecules and/or MHC class II molecules is modulated by targeting and deleting a contiguous stretch of genomic DNA, thereby reducing or eliminating expression of a target gene selected from the group consisting of B2M, CIITA, and NLRC5.
- MHC class I molecules can alternatively or additionally be modulated by reducing or eliminating expression of TAPI .
- MHC class II molecules can alternatively or additionally be modulated by reducing or eliminating expression of CD74.
- the provided modified pluripotent stem cells comprise a modification of one or more target polynucleotide sequence that regulate MHC class I. Exemplary methods for reducing expression of MHC class I are described in sections below.
- the targeted polynucleotide sequence is one or both of B2M and NLRC5.
- the cell comprises a genetic editing modification (e.g., an indel) to the B2M gene. In some embodiments, the cell comprises a genetic editing modification (e.g., an indel) to the NLRC5 gene. In some embodiments, the cell comprises a genetic editing modification (e.g., an indel) to the TAPI gene. In some embodiments, the cell comprises genetic editing modifications (e.g., indels) to the B2M and CIITA genes.
- a modification that reduces expression of an MHC class I molecule is a modification that reduces expression of B2M.
- the modification that reduces B2M expression reduces B2M mRNA expression.
- the reduced mRNA expression of B2M is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification.
- the mRNA expression of B2M is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more.
- the mRNA expression of B2M is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the mRNA expression of B2M is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of B2M is eliminated (e.g., 0% expression of B2M mRNA). In some embodiments, the modification that reduces B2M mRNA expression eliminates B2M gene activity.
- the modification that reduces B2M expression reduces B2M protein expression.
- the reduced protein expression of B2M is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification.
- the protein expression of B2M is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more.
- the protein expression of B2M is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less.
- the protein expression of B2M is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the protein expression of B2M is eliminated (e.g., no detectable expression of B2M protein). In some embodiments, the modification that reduces B2M protein expression eliminates B2M gene activity.
- the modification that reduces B2M expression comprises inactivation or disruption of the B2M gene. In some embodiments, the modification that reduces B2M expression comprises inactivation or disruption of one allele of the B2M gene. In some embodiments, the modification that reduces B2M expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the B2M gene.
- the modification comprises inactivation or disruption of one or more B2M coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all B2M coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the B2M gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the B2M gene. In some embodiments, the modification is a deletion of genomic DNA of the B2M gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the B2M gene. In some embodiments, the B2M gene is knocked out.
- a modification that reduces expression of an MHC class I molecule is a modification that reduces expression of NLRC5.
- decreased or eliminated expression of NLRC5 reduces or eliminates expression of one or more of the following MHC I molecules HLA-A, HLA-B, and HLA-C.
- the modification that reduces NLRC5 expression reduces NLRC5 mRNA expression.
- the reduced mRNA expression of NLRC5 is relative to an unmodified or wildtype cell of the same cell type that does not comprise the modification.
- the mRNA expression of NLRC5 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the mRNA expression of NLRC5 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the mRNA expression of NLRC5 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of NLRC5 is eliminated (e.g., 0% expression of NLRC5 mRNA). In some embodiments, the modification that reduces NLRC5 mRNA expression eliminates NLRC5 gene activity.
- the modification that reduces NLRC5 expression reduces NLRC5 protein expression.
- the reduced protein expression of NLRC5 is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification.
- the protein expression of NLRC5 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more.
- the protein expression of NLRC5 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less.
- the protein expression of NLRC5 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the protein expression of NLRC5 is eliminated (e.g., no detectable expression of NLRC5 protein). In some embodiments, the modification that reduces NLRC5 protein expression eliminates NLRC5 gene activity.
- the modification that reduces NLRC5 expression comprises inactivation or disruption of the NLRC5 gene. In some embodiments, the modification that reduces NLCR5 expression comprises inactivation or disruption of one allele of the NLRC5 gene. In some embodiments, the modification that reduces NLRC5 expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the NLRC5 gene.
- the modification comprises inactivation or disruption of one or more NLRC5 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all NLRC5 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the NLRC5 gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the NLRC5 gene. In some embodiments, the modification is a deletion of genomic DNA of the NLRC5 gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the NLRC5 gene. In some embodiments, the NLRC5 gene is knocked out.
- a modification that reduces expression of an MHC class I molecule is a modification that reduces expression of TAPI.
- decreased or eliminated expression of TAPI reduces or eliminates expression of one or more of the following MHC I molecules - HLA-A, HLA-B, and HLA-C.
- the modification that reduces TAPI expression reduces TAPI mRNA expression.
- the reduced mRNA expression of TAPI is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification.
- the mRNA expression of TAPI is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the mRNA expression of TAPI is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the mRNA expression of TAPI is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of
- TAPI is eliminated (e.g., 0% expression of TAPI mRNA).
- the modification that reduces TAPI mRNA expression eliminates TAPI gene activity.
- the modification that reduces TAPI expression reduces TAPI protein expression.
- the reduced protein expression of TAPI is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification.
- the protein expression of TAPI is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more.
- the protein expression of TAPI is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less.
- the protein expression of TAPI is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the protein expression of TAPI is eliminated (e.g., no detectable expression of TAPI protein). In some embodiments, the modification that reduces TAPI protein expression eliminates TAPI gene activity.
- the modification that reduces TAPI expression comprises inactivation or disruption of the TAPI gene. In some embodiments, the modification that reduces TAPI expression comprises inactivation or disruption of one allele of the TAPI gene. In some embodiments, the modification that reduces TAPI expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the TAPI gene.
- the modification comprises inactivation or disruption of one or more TAPI coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all TAPI coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the TAPI gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the TAPI gene. In some embodiments, the modification is a deletion of genomic DNA of the TAPI gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the TAPI gene. In some embodiments, the TAPI gene is knocked out.
- the provided modified pluripotent stem cells comprise a modification of one or more target polynucleotide sequence that regulate MHC class II molecule expression. Exemplary methods for reducing expression of MHC class II are described in sections below.
- the cell comprises a genetic editing modification to the CIITA gene. In some embodiments, the cell comprises a genetic editing modification to the CD74 gene.
- a modification that reduces expression of an MHC class II molecule is a modification that reduces expression of CIITA.
- the modification that reduces CIITA expression reduces CIITA mRNA expression.
- the reduced mRNA expression of CIITA is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification.
- the mRNA expression of CIITA is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more.
- the mRNA expression of CIITA is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the mRNA expression of CIITA is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of CIITA is eliminated (e.g., 0% expression of CIITA mRNA). In some embodiments, the modification that reduces CIITA mRNA expression eliminates CIITA gene activity.
- the modification that reduces CIITA expression reduces CIITA protein expression.
- the reduced protein expression of CIITA is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification.
- the protein expression of CIITA is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more.
- the protein expression of CIITA is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less.
- the protein expression of CIITA is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the protein expression of CIITA is eliminated (e.g., 0% expression of CIITA protein). In some embodiments, the modification that reduces CIITA protein expression eliminates CIITA gene activity.
- the modification that reduces CIITA expression comprises inactivation or disruption of the CIITA gene. In some embodiments, the modification that reduces CIITA expression comprises inactivation or disruption of one allele of the CIITA gene. In some embodiments, the modification that reduces CIITA expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the CIITA gene.
- the modification comprises inactivation or disruption of one or more CIITA coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all CIITA coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the CIITA gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the CIITA gene. In some embodiments, the modification is a deletion of genomic DNA of the CIITA gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the CIITA gene. In some embodiments, the CIITA gene is knocked out.
- a modification that reduces expression of an MHC class II molecule is a modification that reduces expression of CD74.
- the modification that reduces CD74 expression reduces CD74 mRNA expression.
- the reduced mRNA expression of CD74 is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification.
- the mRNA expression of CD74 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more.
- the mRNA expression of CD74 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the mRNA expression of CD74 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of CD74 is eliminated (e.g., 0% expression of CD74 mRNA). In some embodiments, the modification that reduces CD74 mRNA expression eliminates CD74 gene activity.
- the modification that reduces CD74 expression reduces CD74 protein expression.
- the reduced protein expression of CD74 is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification.
- the protein expression of CD74 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more.
- the protein expression of CD74 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less.
- the protein expression of CD74 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the protein expression of CD74 is eliminated (e.g., 0% expression of CD74 protein). In some embodiments, the modification that reduces CD74 protein expression eliminates CD74 gene activity.
- the modification that reduces CD74 expression comprises inactivation or disruption of the CD74 gene. In some embodiments, the modification that reduces CD74 expression comprises inactivation or disruption of one allele of the CD74 gene. In some embodiments, the modification that reduces CD74 expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the CD74 gene.
- the modification comprises inactivation or disruption of one or more CD74 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all CD74 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the CD74 gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the CD74 gene. In some embodiments, the modification is a deletion of genomic DNA of the CD74 gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the CD74 gene. In some embodiments, the CD74 gene is knocked out.
- the provided modified cells comprise a modification of one or more target polynucleotide sequence that regulate expression of MHC class I molecules and MHC class II molecules. Exemplary methods for reducing expression of MHC class I molecules and MHC class 11 molecules including any as described in sections below.
- the cell comprises genetic editing modifications to the B2M and NLRC5 genes. In some embodiments, the cell comprises genetic editing modifications to the CIITA and NLRC5 genes. In some embodiments, the cell comprises genetic editing modifications to the B2M and CIITA genes. In particular embodiments, the cell comprises genetic editing modifications to the B2M, CIITA and NLRC5 genes.
- the present disclosure comprises the modulation (e.g., reduction or elimination) the expression of CD142, which is also known as tissue factor, factor III, and F3.
- the modulation occurs using a CRISPR/Cas system.
- the target polynucleotide sequence is CD 142 or a variant of CD142. In some embodiments, the target polynucleotide sequence is a homolog of CD142. In some embodiments, the target polynucleotide sequence is an ortholog of CD 142.
- the cells outlined herein comprise a modification targeting the CD 142 gene.
- the modification targeting the CD 142 gene by the rare- cutting endonuclease comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid (gRNA) sequence for specifically targeting the CD 142 gene.
- gRNA guide ribonucleic acid
- Assays to test whether the CD 142 gene has been inactivated are known and described herein.
- the resulting modification of the CD 142 gene by PCR and the reduction of CD 142 expression can be assays by FACS analysis.
- CD 142 protein expression is detected using a Western blot of cells lysates probed with antibodies to the CD142 protein.
- reverse transcriptase polymerase chain reactions RT-PCR
- Useful genomic, polynucleotide and polypeptide information about the human CD142 are provided in, for example, the GeneCard Identifier GC01M094530, HGNC No.
- the target polynucleotide sequence is PD-1 or a variant of PD-1. In some embodiments, the target polynucleotide sequence is a homolog of PD-1. In some embodiments, the target polynucleotide sequence is an ortholog of PD-1.
- the cells outlined herein comprise a genetic modification targeting the gene encoding the programmed cell death protein 1 (PD- 1 ) protein or the PDCD 1 gene.
- primary T cells comprise a genetic modification targeting the PDCD1 gene.
- the genetic modification can reduce expression of PD-1 polynucleotides and PD- 1 polypeptides in T cells includes primary T cells and CAR-T cells.
- the genetic modification targeting the PDCD1 gene by the rare-cutting endonuclease comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid (gRNA) sequence for specifically targeting the PDCD1 gene.
- gRNA guide ribonucleic acid
- Assays to test whether the PDCD1 gene has been inactivated are known and described herein.
- the resulting genetic modification of the PDCD1 gene by PCR and the reduction of PD-1 expression can be assays by FACS analysis.
- PD-1 protein expression is detected using a Western blot of cells lysates probed with antibodies to the PD-1 protein.
- reverse transcriptase polymerase chain reactions RT-PCR are used to confirm the presence of the inactivating genetic modification.
- the cells provided herein are modified (e.g., genetically modified) to inactivate or disrupt one or more target polynucleotides or proteins as described. In some embodiments, the cells provided herein are modified (e.g., genetically modified) to reduce expression of the one or more target polynucleotides or proteins as described. In some embodiments, the cell that is modified with the one or more modification to reduce (e.g., eliminate) expression of a polynucleotide or protein is any source cell as described herein. In certain embodiments, the modified pluripotent stem cells (e.g., differentiated cells such as islet cells) disclosed herein comprise one or more modifications to reduce expression of one or more target polynucleotides.
- the modified pluripotent stem cells e.g., differentiated cells such as islet cells
- Non-limiting examples of the one or more target polynucleotides include any as described above, such as CIITA, B2M, CD 142, NLRC5, HLA-A, HLA-B, HLA-C, LRC5, RFX-ANK, RFX5, RFX-AP, NFY-A, NFY-B, NFY-C, IRF1, and TAPI.
- the target polynucleotide may be CD74.
- the modifications to reduce expression of the one or more target polynucleotides is combined with one or more modifications to increase expression of a desired transgene.
- the modifications create modified cells that are immune-privileged or hypoimmunogenic cells.
- such cells By modulating (e.g., reducing or deleting) expression of one or a plurality of the target polynucleotides, such cells exhibit decreased immune activation when engrafted into a recipient subject.
- the cell is considered hypoimmunogenic, e.g., in a recipient subject or patient upon administration.
- any method for reducing expression of a target polynucleotide may be used.
- the modifications result in permanent elimination or reduction in expression of the target polynucleotide.
- the target polynucleotide or gene is disrupted by introducing a DNA break in the target polynucleotide, such as by using a targeting endonuclease.
- the modifications result in transient reduction in expression of the target polynucleotide.
- gene repression is achieved using an inhibitory nucleic acid that is complementary to the target polynucleotide to selectively suppress or repress expression of the gene, for instance using antisense techniques, such as by RNA interference (RNAi), short interfering RNA (siRNA), short hairpin (shRNA), and/or ribozymes.
- RNAi RNA interference
- siRNA short interfering RNA
- shRNA short hairpin
- the target polynucleotide sequence is a genomic sequence. In some embodiments, the target polynucleotide sequence is a human genomic sequence. In some embodiments, the target polynucleotide sequence is a mammalian genomic sequence. In some embodiments, the target polynucleotide sequence is a vertebrate genomic sequence.
- gene disruption is carried out by induction of one or more double-stranded breaks and/or one or more single- stranded breaks in the gene, typically in a targeted manner.
- the double-stranded or single-stranded breaks are made by a nuclease, e.g., an endonuclease, such as a gene-targeted nuclease.
- the targeted nuclease is selected from zinc finger nucleases (ZFN), transcription activator-like effector nucleases (TALENs), and RNA-guided nucleases such as a CRISPR-associated nuclease (Cas), specifically designed to be targeted to the sequence of a gene or a portion thereof.
- ZFN zinc finger nucleases
- TALENs transcription activator-like effector nucleases
- Cas RNA-guided nucleases
- the targeted nuclease generates double- stranded or single-stranded breaks that then undergo repair through error prone non-homologous end joining (NHEJ) or, in some cases, precise homology directed repair (HDR) in which a template is used.
- NHEJ error prone non-homologous end joining
- HDR precise homology directed repair
- the targeted nuclease generates DNA double strand breaks (DSBs).
- the process of producing and repairing the breaks is typically error prone and results in insertions and deletions (indels) of DNA bases from NHEJ repair.
- the modification may induce a deletion, insertion, or mutation of the nucleotide sequence of the target gene.
- the modification may result in a frameshift mutation, which can result in a premature stop codon.
- the targeted edits occur on both alleles of the gene resulting in a biallelic disruption or edit of the gene.
- all alleles of the gene are targeted by the gene editing.
- the nuclease such as a rare-cutting endonuclease
- the nuclease is introduced into a cell containing the target polynucleotide sequence.
- the nuclease may be introduced into the cell in the form of a nucleic acid encoding the nuclease.
- the process of introducing the nucleic acids into cells can be achieved by any suitable technique. Suitable techniques include calcium phosphate or lipid-mediated transfection, electroporation, and transduction or infection using a viral vector.
- the nucleic acid that is introduced into the cell is DNA.
- the nuclease is introduced into the cell in the form of a protein. For instance, in the case of a CRISPR/Cas system a ribonucleoprotein (RNP) may be introduced into the cell.
- RNP ribonucleoprotein
- the modification occurs using a CRISPR/Cas system.
- Any CRISPR/Cas system that is capable of altering a target polynucleotide sequence in a cell can be used.
- Such CRISPR-Cas systems can employ a variety of Cas proteins (Haft et al. PLoS Comput Biol. 2005; l(6)e60).
- the molecular machinery of such Cas proteins that allows the CRISPR/Cas system to alter target polynucleotide sequences in cells include RNA binding proteins, endo- and exo-nucleases, helicases, and polymerases.
- the CRISPR/Cas system is a CRISPR type I system.
- the CRISPR/Cas system is a CRISPR type II system.
- the CRISPR/Cas system is a CRISPR type V system.
- the CRISPR/Cas systems include targeted systems that can be used to alter any target polynucleotide sequence in a cell.
- a CRISPR/Cas system provided herein includes a Cas protein and one or more, such as at least one to two, ribonucleic acids (e.g., guide RNA (gRNA)) that are capable of directing the Cas protein to and hybridizing to a target motif of a target polynucleotide sequence.
- gRNA guide RNA
- a Cas protein comprises one or more amino acid substitutions or modifications.
- the one or more amino acid substitutions comprises a conservative amino acid substitution.
- substitutions and/or modifications can prevent or reduce proteolytic degradation and/or extend the half-life of the polypeptide in a cell.
- the Cas protein can comprise a peptide bond replacement (e.g., urea, thiourea, carbamate, sulfonyl urea, etc.).
- the Cas protein can comprise a naturally occurring amino acid.
- the Cas protein can comprise an alternative amino acid (e.g., D-amino acids, beta-amino acids, homocysteine, phosphoserine, etc.).
- a Cas protein can comprise a modification to include a moiety (e.g., PEGylation, glycosylation, lipidation, acetylation, end-capping, etc.).
- a Cas protein comprises a core Cas protein.
- Exemplary Cas core proteins include, but are not limited to Cast, Cas2, Cas3, Cas4, Cas5, Cas6, Cas7, Cas8 and Cas9.
- a Cas protein comprises a Cas protein of an E. coli subtype (also known as CASS2).
- Exemplary Cas proteins of the E. Coli subtype include, but are not limited to Csel, Cse2, Cse3, Cse4, and Cas5e.
- a Cas protein comprises a Cas protein of the Ypest subtype (also known as CASS3).
- Exemplary Cas proteins of the Ypest subtype include, but are not limited to Csyl, Csy2, Csy3, and Csy4.
- a Cas protein comprises a Cas protein of the Nmeni subtype (also known as CASS4).
- Exemplary Cas proteins of the Nmeni subtype include, but are not limited to, Csnl and Csn2.
- a Cas protein comprises a Cas protein of the Dvulg subtype (also known as CASS1).
- Exemplary Cas proteins of the Dvulg subtype include Csdl, Csd2, and Cas5d.
- a Cas protein comprises a Cas protein of the Tneap subtype (also known as CASS7).
- Exemplary Cas proteins of the Tneap subtype include, but are not limited to, Cstl, Cst2, Cas5t.
- a Cas protein comprises a Cas protein of the Hmari subtype.
- Exemplary Cas proteins of the Hmari subtype include, but are not limited to Cshl, Csh2, and Cas5h.
- a Cas protein comprises a Cas protein of the Apem subtype (also known as CASS5).
- Exemplary Cas proteins of the Apem subtype include, but are not limited to Csal, Csa2, Csa3, Csa4, Csa5, and Cas5a.
- a Cas protein comprises a Cas protein of the Mtube subtype (also known as CASS6).
- Exemplary Cas proteins of the Mtube subtype include, but are not limited to Csml, Csm2, Csm3, Csm4, and Csm5.
- a Cas protein comprises a RAMP module Cas protein.
- Exemplary RAMP module Cas proteins include, but are not limited to, CrmT, Cmr2, Cmr3, Cmr4, Cmr5, and Cmr6. See, e.g., Klompe et al., Nature 571, 219-225 (2019); Strecker et al., Science 365, 48-53 (2019).
- CRISPR systems of the present disclosure comprise TnpB polypeptides.
- TnpB polypeptides may comprise a Ruv-C-like domain.
- the RuvC domain may be a split RuvC domain comprising RuvC-I, RuvC-II, and RuvC-III subdomains.
- a TnpB may further comprise one or more of a HTH domain, a bridge helix domain, and a zinc finger domain.
- TnpB polypeptides do not comprise an HNH domain.
- a TnpB protein comprises, starting at the N-terminus: a HTH domain, a RuvC-1 subdomain, a bridge helix domain, a RuvC-11 sub-domain, a zinger finger domain, and a RuvC-III sub-domain.
- a RuvC-III sub-domain forms the C-terminus of a TnpB polypeptide.
- a TnpB polypeptide is from Epsilonproteobacteria bacterium, Actinoplanes lobatus strain DSM 43150, Actinomadura celluolosilytica strain DSM 45823, Actinomadura namibiensis strain DSM 44197, Alicyclobacillus macrosprangiidus strain DSM 17980, Lipingzhangella halophila strain DSM 102030, or Ktedonobacter recemifer.
- a TnpB polypeptide is from Ktedonobacter racemifer, or comprises a conserved RNA region with similarity to the 5’ ITR of K. racemifer TnpB loci.
- a TnpB may comprise a Fanzor protein, a TnpB homolog found in eukaryotic genomes.
- a CRISPR system comprising a TnpB polypeptide binds a target adjacent motif (TAM) sequence 5’ of a target polynucleotide.
- TAM is a transposon-associated motif.
- a TAM sequence comprises TCA.
- a TAM sequence comprises TTCAN.
- a TAM sequence comprises TTGAT.
- a TAM sequence comprises ATAAA.
- the methods for genetically modifying cells to knock out, knock down, or otherwise modify one or more genes comprise using a site-directed nuclease, including, for example, zinc finger nucleases (ZFNs), transcription activator-like effector nucleases (TALENs), meganucleases, transposases, and clustered regularly interspaced short palindromic repeat (CRISPR)/Cas systems
- ZFNs are fusion proteins comprising an array of site-specific DNA binding domains adapted from zinc finger-containing transcription factors attached to the endonuclease domain of the bacterial FokI restriction enzyme.
- a ZFN may have one or more (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, or more) of the DNA binding domains or zinc finger domains. See, e.g., Carroll et al., Genetics Society of America (2011) 188:773-782; Kim et al., Proc. Natl. Acad. Sci. USA (1996) 93:1156-1160.
- Each zinc finger domain is a small protein structural motif stabilized by one or more zinc ions and usually recognizes a 3- to 4-bp DNA sequence. Tandem domains can thus potentially bind to an extended nucleotide sequence that is unique within a cell’s genome.
- Zinc fingers can be engineered to bind a predetermined nucleic acid sequence. Criteria to engineer a zinc finger to bind to a predetermined nucleic acid sequence are known in the art. See, e.g., Sera et al., Biochemistry (2002) 41:7074-7081; Liu et al., Bioinformatics (2008) 24:1850-1857.
- ZFNs containing FokI nuclease domains or other dimeric nuclease domains function as a dimer.
- a pair of ZFNs are required to target non-palindromic DNA sites.
- the two individual ZFNs must bind opposite strands of the DNA with their nucleases properly spaced apart. See Bitinaite et al., Proc. Natl. Acad. Sci. USA (1998) 95:10570-10575.
- a pair of ZFNs arc designed to recognize two sequences flanking the site, one on the forward strand and the other on the reverse strand.
- the nuclease domains dimerize and cleave the DNA at the site, generating a DSB with 5' overhangs.
- HDR can then be utilized to introduce a specific mutation, with the help of a repair template containing the desired mutation flanked by homology arms.
- the repair template is usually an exogenous double-stranded DNA vector introduced to the cell. See Miller et al., Nat. Biotechnol. (2011) 29:143-148; Hockemeyer et al., Nat. Biotechnol. (2011) 29:731-734.
- TALENs are another example of an artificial nuclease which can be used to edit a target gene.
- TALENs are derived from DNA binding domains termed TALE repeats, which usually comprise tandem arrays with 10 to 30 repeats that bind and recognize extended DNA sequences. Each repeat is 33 to 35 amino acids in length, with two adjacent amino acids (termed the repeat-variable di-residue, or RVD) conferring specificity for one of the four DNA base pairs.
- RVD repeat-variable di-residue
- TALENs are produced artificially by fusing one or more TALE DNA binding domains (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more) to a nuclease domain, for example, a FokI endonuclease domain.
- TALE DNA binding domains e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more
- a nuclease domain for example, a FokI endonuclease domain.
- the FokI domain functions as a dimer, requiring two constructs with unique DNA binding domains for sites in the target genome with proper orientation and spacing. Both the number of amino acid residues between the TALE DNA binding domain and the FokI nuclease domain and the number of bases between the two individual TALEN binding sites appear to be important parameters for achieving high levels of activity. Miller et al., Nature Biotech. (2011) 29: 143-148.
- a site-specific nuclease can be produced specific to any desired DNA sequence. Similar to ZFNs, TALENs can be introduced into a cell to generate DSBs at a desired target site in the genome, and so can be used to knock out genes or knock in mutations in similar, HDR-mediated pathways. See Boch, Nature Biotech. (2011) 29:135-136; Boch et al., Science (2009) 326:1509-1512; Moscou et al., Science (2009) 326:3501.
- Meganucleases are enzymes in the endonuclease family which are characterized by their capacity to recognize and cut large DNA sequences (from 14 to 40 base pairs). Meganucleases are grouped into families based on their structural motifs which affect nuclease activity and/or DNA recognition. The most widespread and best known meganucleases are the proteins in the LAGLIDADG family, which owe their name to a conserved amino acid sequence. See Chevalier et al., Nucleic Acids Res. (2001) 29(18): 3757-3774.
- the GIY- YIG family members have a GIY-YIG module, which is 70-100 residues long and includes four or five conserved sequence motifs with four invariant residues, two of which arc required for activity. See Van Roey et al., Nature Struct. Biol. (2002) 9:806-811.
- the His-Cys family meganucleases are characterized by a highly conserved series of histidines and cysteines over a region encompassing several hundred amino acid residues. See Chevalier et al., Nucleic Acids Res. (2001) 29(18):3757-3774.
- NHN family are defined by motifs containing two pairs of conserved histidines surrounded by asparagine residues. See Chevalier et al., Nucleic Acids Res. (2001 ) 29(18):3757-3774.
- Meganucleases can create DSBs in the genomic DNA, which can create a frame-shift mutation if improperly repaired, e.g., via NHEJ, leading to a decrease in the expression of a target gene in a cell.
- foreign DNA can be introduced into the cell along with the meganuclease. Depending on the sequences of the foreign DNA and chromosomal sequence, this process can be used to modify the target gene. See Silva et al., Current Gene Therapy (2011) 11:11-27.
- Transposases are enzymes that bind to the end of a transposon and catalyze its movement to another part of the genome by a cut and paste mechanism or a replicative transposition mechanism.
- transposases By linking transposases to other systems such as the CRISPR/Cas system, new gene editing tools can be developed to enable site specific insertions or manipulations of the genomic DNA.
- transposons which use a catalytically inactive Cas effector protein and Tn7-like transposons.
- the transposase-dependent DNA integration does not provoke DSBs in the genome, which may guarantee safer and more specific DNA integration.
- the CRISPR system was originally discovered in prokaryotic organisms (e.g., bacteria and archaea) as a system involved in defense against invading phages and plasmids that provides a form of acquired immunity. Now it has been adapted and used as a po ular gene editing tool in research and clinical applications.
- prokaryotic organisms e.g., bacteria and archaea
- CRISPR/Cas systems generally comprise at least two components: one or more guide RNAs (gRNAs) and a Cas protein.
- the Cas protein is a nuclease that introduces a DSB into the target site.
- CRISPR-Cas systems fall into two major classes: class 1 systems use a complex of multiple Cas proteins to degrade nucleic acids; class 2 systems use a single large Cas protein for the same purpose. Class 1 is divided into types I, III, and IV; class 2 is divided into types II, V, and VI.
- Cas proteins adapted for gene editing applications include, but are not limited to, Cas3, Cas4, Cas5, Cas8a, Cas8b, Cas8c, Cas9, CaslO, Casl2, Casl2a (Cpfl), Casl2b (C2cl), Casl2c (C2c3), Casl2d (CasY), Casl2e (CasX), Casl2f (C2cl0), Casl2g, Casl2h, Casl2i, Casl2k (C2c5), Casl3, Casl3a (C2c2), Casl3b, Casl3c, Casl3d, C2c4, C2c8, C2c9, Cmr5, Csel, Cse2, Csfl, Csm2, Csn2, CsxlO, Csxl l, Csyl, Csy2, Csy3, and Mad7.
- Cas9 is a type II Cas protein and is described herein as illustrative. These Cas proteins may be originated from different source species. For example, Cas9 can be derived from S. pyogenes or S. aureus.
- the type II CRISPR system incorporates sequences from invading DNA between CRISPR repeat sequences encoded as arrays within the host genome. Transcripts from the CRISPR repeat arrays are processed into CRISPR RNAs (crRNAs) each harboring a variable sequence transcribed from the invading DNA, known as the “protospacer” sequence, as well as part of the CRISPR repeat. Each crRNA hybridizes with a second transactivating CRISPR RNA (tracrRNA), and these two RNAs form a complex with the Cas9 nuclease. The protospacer-encoded portion of the crRNA directs the Cas9 complex to cleave complementary target DNA sequences, provided that they are adjacent to short sequences known as “protospacer adjacent motifs” (PAMs).
- PAMs protospacer adjacent motifs
- the CRISPR system Since its discovery, the CRISPR system has been adapted for inducing sequence specific DSBs and targeted genome editing in a wide range of cells and organisms spanning from bacteria to eukaryotic cells including human cells.
- synthetic gRNAs have replaced the original crRNA:tracrRNA complex.
- the gRNAs can be single guide RNAs (sgRNAs) composed of a crRNA, a tetraloop, and a tracrRNA.
- the crRNA usually comprises a complementary region (also called a spacer, usually about 20 nucleotides in length) that is user-designed to recognize a target DNA of interest.
- the tracrRNA sequence comprises a scaffold region for Cas nuclease binding.
- the crRNA sequence and the tracrRNA sequence are linked by the tetraloop and each have a short repeat sequence for hybridization with each other, thus generating a chimeric sgRNA.
- the complementary region will direct the Cas nuclease to the target DNA site through standard RNA-DNA complementary base pairing rules.
- R A or G
- Y C or T
- W A or T
- V A or C or G
- N any base
- Cas nucleases may comprise one or more mutations to alter their activity, specificity, recognition, and/or other characteristics.
- the Cas nuclease may have one or more mutations that alter its fidelity to mitigate off-target effects (e.g., eSpCas9, SpCas9-HFl, HypaSpCas9, HeFSpCas9, and evoSpCas9 high-fidelity variants of SpCas9).
- the Cas nuclease may have one or more mutations that alter its PAM specificity.
- a Cas protein comprises any one of the Cas proteins described herein or a functional portion thereof.
- “functional portion” refers to a portion of a peptide which retains its ability to complex with at least one ribonucleic acid (e.g., guide RNA (gRNA)) and cleave a target polynucleotide sequence.
- the functional portion comprises a combination of operably linked Cas9 protein functional domains selected from the group consisting of a DNA binding domain, at least one RNA binding domain, a helicase domain, and an endonuclease domain.
- the functional portion comprises a combination of operably linked Cas 12a (also known as Cpfl) protein functional domains selected from the group consisting of a DNA binding domain, at least one RNA binding domain, a helicase domain, and an endonuclease domain.
- the functional domains form a complex.
- a functional portion of the Cas9 protein comprises a functional portion of a RuvC-like domain.
- a functional portion of the Cas9 protein comprises a functional portion of the HNH nuclease domain.
- a functional portion of the Casl2a protein comprises a functional portion of a RuvC-like domain.
- suitable Cas proteins include, but are not limited to, CasO, Casl2a (i.e., Cpfl), Casl2b, Casl2i, CasX, and Mad7.
- exogenous Cas protein can be introduced into the cell in polypeptide form.
- Cas proteins can be conjugated to or fused to a cellpenetrating polypeptide or cell-penetrating peptide.
- cell-penetrating polypeptide and “cell-penetrating peptide” refers to a polypeptide or peptide, respectively, which facilitates the uptake of molecule into a cell.
- the cell-penetrating polypeptides can contain a detectable label.
- Cas proteins can be conjugated to or fused to a charged protein (e.g., that carries a positive, negative or overall neutral electric charge). Such linkage may be covalent.
- the Cas protein can be fused to a superpositively charged GFP to significantly increase the ability of the Cas protein to penetrate a cell (Cronican et al. ACS Chem Biol. 2010; 5(8):747-52).
- the Cas protein can be fused to a protein transduction domain (PTD) to facilitate its entry into a cell.
- PTDs protein transduction domain
- Exemplary PTDs include Tat, oligoarginine, and penetratin.
- the Cas9 protein comprises a Cas9 polypeptide fused to a cell-penetrating peptide. In some embodiments, the Cas9 protein comprises a Cas9 polypeptide fused to a PTD. In some embodiments, the Cas9 protein comprises a Cas9 polypeptide fused to a tat domain. In some embodiments, the Cas9 protein comprises a Cas9 polypeptide fused to an oligoarginine domain. In some embodiments, the Cas9 protein comprises a Cas9 polypeptide fused to a penetrating domain. In some embodiments, the Cas9 protein comprises a Cas9 polypeptide fused to a superpositively charged GFP.
- the Casl2a protein comprises a Casl2a polypeptide fused to a cell-penetrating peptide.
- the Cas 12a protein comprises a Cas 12a polypeptide fused to a PTD.
- the Casl2a protein comprises a Casl2a polypeptide fused to a tat domain.
- the Cas 12a protein comprises a Cas 12a polypeptide fused to an oligoarginine domain.
- the Cas 12a protein comprises a Cas 12a polypeptide fused to a penetrating domain.
- the Casl2a protein comprises a Casl2a polypeptide fused to a superpositively charged GFP.
- the Cas protein can be introduced into a cell containing the target polynucleotide sequence in the form of a nucleic acid encoding the Cas protein.
- the process of introducing the nucleic acids into cells can be achieved by any suitable technique. Suitable techniques include calcium phosphate or lipid-mediated transfection, electroporation, and transduction or infection using a viral vector.
- the nucleic acid comprises DNA.
- the nucleic acid comprises a modified DNA, as described herein.
- the nucleic acid comprises mRNA.
- the nucleic acid comprises a modified mRNA, as described herein (e.g., a synthetic, modified mRNA).
- a CRISPR/Cas system generally includes two components; one or more guide RNA (gRNA) and a Cas protein.
- the Cas protein is complexed with the one or more, such as one to two, ribonucleic acids (e.g., guide RNA (gRNA)).
- the Cas protein is complexed with two ribonucleic acids.
- the Cas protein is complexed with one ribonucleic acid.
- the Cas protein is encoded by a modified nucleic acid, as described herein (e.g., a synthetic, modified mRNA).
- gRNAs are short synthetic RNAs composed of a scaffold sequence for Cas binding and a user-designed spacer or complementary portion designated crRNA.
- the crRNA is composed of a crRNA targeting sequence (herein after also called a gRNA targeting sequence; usually about 20 nucleotides in length) that defines the genomic target to be modified and a region of crRNA repeat (e.g., GUUUUAGAGCUA; SEQ ID NO: 19).
- a crRNA targeting sequence herein after also called a gRNA targeting sequence; usually about 20 nucleotides in length
- crRNA repeat e.g., GUUUUAGAGCUA; SEQ ID NO: 19
- One can change the genomic target of the Cas protein by simply changing the complementary portion sequence (e.g., gRNA targeting sequence) present in the gRNA.
- the scaffold sequence for Cas binding is made up of a tracrRNA sequence (e.g., UAGCAAGUUAAAAUAAGGCUAGUCCGUUAUCAACUUGAAAAAGUGGCACCGAGU CGGUGCUUU; SEQ ID NO: 20) that hybridizes to the crRNA through its anti-repeat sequence.
- the complex between crRNA: tracrRNA recruits the Cas nuclease (e.g., Cas9) and cleaves upstream of a protospacer-adjacent motif (PAM).
- PAM protospacer-adjacent motif
- the specific sequence of PAM varies depending on the species of the Cas gene. For example, the most commonly used Cas9 nuclease, derived from S. pyogenes, recognizes a PAM sequence of NGG. Other Cas9 variants and other nucleases with alternative PAMs have also been characterized and successfully used for genome editing.
- the CRISPR/Cas system can be used to create targeted DSBs at specified genomic loci that are complementary to the gRNA designed for the target loci.
- the crRNA and tracrRNA can be linked together with a loop sequence (e.g., a tetraloop; GAAA, SEQ ID NO:21) for generation of a gRNA that is a chimeric single guide RNA (sgRNA; Hsu et al. 2013).
- sgRNA can be generated for DNA-based expression or by chemical synthesis.
- the complementary portion sequences (e.g., gRNA targeting sequence) of the gRNA will vary depending on the target site of interest.
- the gRNAs comprise complementary portions specific to a sequence of a gene set forth in Table 3 or Table 4.
- the genomic locus targeted by the gRNAs is located within 4000 bp, within 3500 bp, within 3000 bp, within 2500 bp, within 2000 bp, within 1500 bp, within 1000 bp, or within 500 bp of any of the loci as described.
- ribonucleic acid that is capable of directing a Cas protein to and hybridizing to a target motif of a target polynucleotide sequence.
- at least one of the ribonucleic acids comprises
- the Cas protein is complexed with one to two ribonucleic acids (e.g., guide RNA (gRNA)). In some embodiments, the Cas protein is complexed with two ribonucleic acids. In some embodiments, the Cas protein is complexed with one ribonucleic acid. In some embodiments, the Cas protein is encoded by a modified nucleic acid, as described herein (e.g., a synthetic, modified mRNA). [04341 The methods disclosed herein contemplate the use of any ribonucleic acid that is capable of directing a Cas protein to and hybridizing to a target motif of a target polynucleotide sequence.
- gRNA guide RNA
- the Cas protein is complexed with two ribonucleic acids.
- the Cas protein is complexed with one ribonucleic acid.
- the Cas protein is encoded by a modified nucleic acid, as described herein (e.g., a synthetic,
- At least one of the ribonucleic acids comprises tracrRNA. In some embodiments, at least one of the ribonucleic acids comprises CR1SPR RNA (crRNA). In some embodiments, a single ribonucleic acid comprises a guide RNA that directs the Cas protein to and hybridizes to a target motif of the target polynucleotide sequence in a cell. In some embodiments, at least one of the ribonucleic acids comprises a guide RNA that directs the Cas protein to and hybridizes to a target motif of the target polynucleotide sequence in a cell.
- both of the one to two ribonucleic acids comprise a guide RNA that directs the Cas protein to and hybridizes to a target motif of the target polynucleotide sequence in a cell.
- the ribonucleic acids provided herein can be selected to hybridize to a variety of different target motifs, depending on the particular CRISPR/Cas system employed, and the sequence of the target polynucleotide, as will be appreciated by those skilled in the art.
- the one to two ribonucleic acids can also be selected to minimize hybridization with nucleic acid sequences other than the target polynucleotide sequence.
- the one to two ribonucleic acids hybridize to a target motif that contains at least two mismatches when compared with all other genomic nucleotide sequences in the cell. In some embodiments, the one to two ribonucleic acids hybridize to a target motif that contains at least one mismatch when compared with all other genomic nucleotide sequences in the cell. In some embodiments, the one to two ribonucleic acids are designed to hybridize to a target motif immediately adjacent to a deoxyribonucleic acid motif recognized by the Cas protein. In some embodiments, each of the one to two ribonucleic acids are designed to hybridize to target motifs immediately adjacent to deoxyribonucleic acid motifs recognized by the Cas protein which flank a mutant allele located between the target motifs.
- each of the one to two ribonucleic acids comprises guide RNAs that directs the Cas protein to and hybridizes to a target motif of the target polynucleotide sequence in a cell.
- one or two ribonucleic acids e.g., guide RNAs
- one or two ribonucleic acids are complementary to and/or hybridize to sequences on the opposite strands of a target polynucleotide sequence.
- the one or two ribonucleic acids are not complementary to and/or do not hybridize to sequences on the opposite strands of a target polynucleotide sequence.
- the one or two ribonucleic acids are complementary to and/or hybridize to overlapping target motifs of a target polynucleotide sequence.
- the one or two ribonucleic acids are complementary to and/or hybridize to offset target motifs of a target polynucleotide sequence.
- nucleic acids encoding Cas protein and nucleic acids encoding the at least one to two ribonucleic acids are introduced into a cell via viral transduction (e.g., lentiviral transduction).
- the Cas protein is complexed with 1-2 ribonucleic acids.
- the Cas protein is complexed with two ribonucleic acids.
- the Cas protein is complexed with one ribonucleic acid.
- the Cas protein is encoded by a modified nucleic acid, as described herein (e.g., a synthetic, modified mRNA).
- gRNA targeting sequences useful for CRISPR/Cas-based targeting of genes described herein are provided in Table 3 or Table 4.
- gRNA targeting sequences for use in methods of genetic disruption to reduce or eliminate expression of a gene as described.
- an existing gRNA targeting sequence for a particular locus e.g., within a target gene, e.g. set forth in Table 3 or Table 4
- an "inch worming" approach can be used to identify additional loci for targeted insertion of transgenes by scanning the flanking regions on either side of the locus for PAM sequences, which usually occurs about every 100 base pairs (bp) across the genome.
- the PAM sequence will depend on the particular Cas nuclease used because different nucleases usually have different corresponding PAM sequences.
- the flanking regions on either side of the locus can be between about 500 to 4000 bp long, for example, about 500 bp, about 1000 bp, about 1500 bp, about 2000 bp, about 2500 bp, about 3000 bp, about 3500 bp, or about 4000 bp long.
- a new guide can be designed according to the sequence of that locus for use in genetic disruption methods.
- the CRISPR/Cas system is described as illustrative, any gene-editing approaches as described can be used in this method of identifying new loci, including those using ZFNs, TALENS, meganucleases and transposases.
- the cells described herein are made using Transcription Activator-Like Effector Nucleases (TALEN) methodologies.
- TALEN Transcription Activator-Like Effector Nucleases
- TALEN Transcription Activator-Like Effector Nucleases
- TALEN Transcription Activator-Like Effector Nucleases
- TALEN Transcription Activator-Like Effector Nucleases
- the catalytic domain is preferably a nuclease domain and more preferably a domain having endonuclease activity, like for instance I-TevI, ColE7, NucA and Fok-I.
- the TALE domain can be fused to a meganuclease like for instance I-Crel and I-Onul or functional variant thereof.
- said nuclease is a monomeric TALE-Nuclease.
- a monomeric TALE-Nuclease is a TALE-Nuclease that does not require dimerization for specific recognition and cleavage, such as the fusions of engineered TAL repeats with the catalytic domain of I-TevI described in WO2012138927.
- Transcription Activator like Effector are proteins from the bacterial species Xanthomonas comprise a plurality of repeated sequences, each repeat comprising di-residues in position 12 and 13 (RVD) that are specific to each nucleotide base of the nucleic acid targeted sequence.
- Binding domains with similar modular basc-pcr-basc nucleic acid binding properties can also be derived from new modular proteins recently discovered by the applicant in a different bacterial species.
- the new modular proteins have the advantage of displaying more sequence variability than TAL repeats.
- RVDs associated with recognition of the different nucleotides are HD for recognizing C, NG for recognizing T, N1 for recognizing A, NN for recognizing G or A, NS for recognizing A, C, G or T, HG for recognizing T, IG for recognizing T, NK for recognizing G, HA for recognizing C, ND for recognizing C, HI for recognizing C, HN for recognizing G, NA for recognizing G, SN for recognizing G or A and YG for recognizing T, TL for recognizing A, VT for recognizing A or G and SW for recognizing A.
- critical amino acids 12 and 13 can be mutated towards other amino acid residues in order to modulate their specificity towards nucleotides A, T, C and G and in particular to enhance this specificity.
- TALEN kits are sold commercially.
- the cells are manipulated using zinc finger nuclease (ZFN).
- ZFN zinc finger nuclease
- a "zinc finger binding protein” is a protein or polypeptide that binds DNA, RNA and/or protein, preferably in a sequence- specific manner, as a result of stabilization of protein structure through coordination of a zinc ion.
- the term zinc finger binding protein is often abbreviated as zinc finger protein or ZFP.
- the individual DNA binding domains are typically referred to as "fingers.”
- a ZFP has least one finger, typically two fingers, three fingers, or six fingers. Each finger binds from two to four base pairs of DNA, typically three or four base pairs of DNA.
- a ZFP binds to a nucleic acid sequence called a target site or target segment.
- Each finger typically comprises an approximately 30 amino acid, zinc-chelating, DNA-binding subdomain.
- Studies have demonstrated that a single zinc finger of this class consists of an alpha helix containing the two invariant histidine residues coordinated with zinc along with the two cysteine residues of a single beta turn (see, e.g., Berg & Shi, Science 271:1081-1085 (1996)).
- the cells described herein are made using a homing endonuclease.
- a homing endonuclease Such homing endonucleases are well-known to the art (Stoddard 2005). Homing endonucleases recognize a DNA target sequence and generate a single- or double-strand break. Homing endonucleases are highly specific, recognizing DNA target sites ranging from 12 to 45 base pairs (bp) in length, usually ranging from 14 to 40 bp in length.
- the homing endonuclease may for example correspond to a LAGLID ADG endonuclease, to an HNH endonuclease, or to a GIY-YIG endonuclease.
- the homing endonuclease can be an I-Crel variant.
- the cells described herein are made using a meganuclease.
- Meganucleases are by definition sequence- specific endonucleases recognizing large sequences (Chevalier, B. S. and B. L. Stoddard, Nucleic Acids Res., 2001, 29, 3757-3774). They can cleave unique sites in living cells, thereby enhancing gene targeting by 1000-fold or more in the vicinity of the cleavage site (Puchta et al., Nucleic Acids Res., 1993, 21, 5034-5040; Rouet et al., Mol. Cell. Biol., 1994, 14, 8096-8106; Choulika et al., Mol. Cell.
- the cells provided herein are made using RNA silencing or RNA interference (RNAi) to knockdown (e.g., decrease, eliminate, or inhibit) the expression of a polypeptide.
- RNAi RNA silencing or RNA interference
- Useful RNAi methods include those that utilize synthetic RNAi molecules, short interfering RNAs (siRNAs), PlWI-interacting NRAs (piRNAs), short hairpin RNAs (shRNAs), microRNAs (miRNAs), and other transient knockdown methods recognized by those skilled in the art.
- RNAi short interfering RNAs
- piRNAs PlWI-interacting NRAs
- shRNAs short hairpin RNAs
- miRNAs microRNAs
- Reagents for RNAi including sequence specific shRNAs, siRNA, miRNAs and the like are commercially available.
- a target polynucleotide such as any described above, e.g., CIITA, B2M, or NLRC5
- a target polynucleotide can be knocked down in a cell by RNA interference by introducing an inhibitory nucleic acid complementary to a target motif of the target polynucleotide, such as an siRNA, into the cells.
- a target polynucleotide such as any described above, e.g., CIITA, B2M, or NLRC5
- RNA interference is employed to reduce or inhibit the expression of at least one selected from the group consisting of CIITA, B2M, and NLRC5.
- the modification reduces or eliminates, such as knocks out, the expression of MHC class I molecules (e.g., MHC class I genes encoding MHC class I molecules) by targeting the accessory chain B2M.
- the modification occurs using a CRISPR/Cas system.
- the target polynucleotide sequence provided herein is a variant of B2M. In some embodiments, the target polynucleotide sequence is a homolog of B2M. In some embodiments, the target polynucleotide sequence is an ortholog of B2M.
- decreased or eliminated expression of B2M reduces or eliminates expression of one or more of the following MHC class I molecules - HLA-A, HLA- B, and HLA-C.
- the modified pluripotent stem cells cell comprises a modification targeting the B2M gene.
- the modification targeting the B2M gene is by using a targeted nuclease system that comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid sequence for specifically targeting the B2M gene.
- the at least one guide ribonucleic acid sequence e.g., gRNA targeting sequence
- the at least one guide ribonucleic acid sequence for specifically targeting the B2M gene is selected from the group consisting of SEQ ID NOS:81240-85644 of Appendix 2 or Table 15 of W02016/183041, the disclosure of which is herein incorporated by reference in its entirety.
- an exogenous nucleic acid or transgene encoding a polypeptide as disclosed herein is inserted at the B2M gene.
- a polypeptide as disclosed herein e.g., a chimeric antigen receptor, CD47, or another tolerogenic factor disclosed herein
- Exemplary transgenes for targeted insertion at the B2M locus include any as described herein.
- Assays to test whether the B2M gene has been inactivated are known and described herein.
- the resulting modification of the B2M gene by PCR and the reduction of HLA-I expression can be assays by flow cytometry, such as by FACS analysis.
- B2M protein expression is detected using a Western blot of cells lysates probed with antibodies to the B2M protein.
- reverse transcriptase polymerase chain reactions RT-PCR
- the technologies disclosed herein modulate (e.g., reduce or eliminate) the expression of MHC-I genes by targeting and modulating (e.g., reducing or eliminating) expression of the NLR family, CARD domain containing 5/NOD27/CLR16.1 (NLRC5).
- the modulation occurs using a CRISPR/Cas system.
- NLRC5 is a critical regulator of MHC-I- mediated immune responses and, similar to CIITA, NLRC5 is highly inducible by IFN-y and can translocate into the nucleus. NLRC5 activates the promoters of MHC-I genes and induces the transcription of MHC-I as well as related genes involved in MHC-I antigen presentation.
- the target polynucleotide sequence is a variant of NLRC5. In some embodiments, the target polynucleotide sequence is a homolog of NLRC5. In some embodiments, the target polynucleotide sequence is an ortholog of NLRC5.
- the cells outlined herein comprise a genetic modification targeting the NLRC5 gene.
- the genetic modification targeting the NLRC5 gene by the rare-cutting endonuclease comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid sequence for specifically targeting the NLRC5 gene.
- the at least one guide ribonucleic acid sequence for specifically targeting the NLRC5 gene is selected from the group consisting of SEQ ID NOS:36353-81239 of Appendix 3 or Table 14 of WO2016183041, the disclosure is incorporated by reference in its entirety.
- RNA expression is detected using a Western blot of cells lysates probed with antibodies to the NLRC5 protein.
- RT-PCR reverse transcriptase polymerase chain reactions
- the reduction of the MHC class I expression or function (HLA I when the cells are derived from human cells) in the modified cells can be measured using techniques known in the art; for example, FACS techniques using labeled antibodies that bind the HLA complex; for example, using commercially available HLA-A, B, C antibodies that bind to the alpha chain of the human major histocompatibility HLA Class I antigens.
- the cells can be tested to confirm that the HLA I complex is not expressed on the cell surface. This may be assayed by FACS analysis using antibodies to one or more HLA cell surface components as discussed above.
- the modified pluripotent stem cells provided herein have a reduced susceptibility to macrophage phagocytosis and NK cell killing. Methods to assay for hypoimmunogenic phenotypes of the modified cells are described further below.
- the modification reduces or eliminates, such as knocks out, the expression of MHC class II genes by targeting Class II transactivator (CIITA) expression.
- CIITA is a member of the LR or nucleotide binding domain (NBD) leucine-rich repeat (LRR) family of proteins and regulates the transcription of MHC class II by associating with the MHC enhanceosome.
- NBD nucleotide binding domain
- LRR leucine-rich repeat
- the target polynucleotide sequence is a variant of CIITA. In some embodiments, the target polynucleotide sequence is a homolog of CIITA. In some embodiments, the target polynucleotide sequence is an ortholog of CIITA. [04601 In some embodiments, reduced or eliminated expression of CIITA reduces or eliminates expression of one or more of the following MHC class II are HLA-DP, HLA-DM, HLA-DOA, HLA-DOB, HLA-DQ, and HLA-DR.
- the modified cell comprises a modification targeting the CIITA gene.
- the modification targeting the CIITA gene is by a targeted nuclease system that comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid sequence for specifically targeting the CIITA gene.
- the at least one guide ribonucleic acid sequence e.g., gRNA targeting sequence
- the at least one guide ribonucleic acid sequence for specifically targeting the CIITA gene is selected from the group consisting of SEQ ID NOS:5184-36352 of Appendix 1 or Table 12 of W02016183041, the disclosure is incorporated by reference in its entirety.
- an exogenous nucleic acid or transgene encoding a polypeptide as disclosed herein is inserted at the CIITA gene.
- a polypeptide as disclosed herein e.g., a chimeric antigen receptor, CD47, or another tolerogenic factor disclosed herein
- exemplary transgenes for targeted insertion at the B2M locus include any as described herein.
- Assays to test whether the CIITA gene has been inactivated are known and described herein.
- the resulting modification of the CIITA gene by PCR and the reduction of HLA-II expression can be assays by flow cytometry, such as by FACS analysis.
- CIITA protein expression is detected using a Western blot of cells lysates probed with antibodies to the CIITA protein.
- reverse transcriptase polymerase chain reactions RT-PCR
- the reduction of the MHC class II expression or function (HLA II when the cells are derived from human cells) in the modified cells can be measured using techniques known in the art, such as Western blotting using antibodies to the protein, FACS techniques, RT-PCR techniques, etc.
- the modified cells can be tested to confirm that the HLA II complex is not expressed on the cell surface.
- Methods to assess surface expression include methods known in the art (See Figure 21 of WO2018132783, for example) and generally is done using either Western Blots or FACS analysis based on commercial antibodies that bind to human HLA Class II HLA-DR, DP and most DQ antigens.
- the modified pluripotent stem cells provided herein have a reduced susceptibility to macrophage phagocytosis and NK cell killing. Methods to assay for hypoimmunogenic phenotypes of the modified cells are described further below.
- the modification reduces or eliminates, such as knocks out, the expression of CD 142.
- the modification occurs using a CRISPR/Cas system.
- CD 142 also known as tissue factor (F3) is a membrane-bound protein that initiates blood coagulation by forming a complex with circulating factor VII or factor Vila.
- the CD142(TF):VIIa complex activates factors IX or X by specific limited proteolysis.
- CD 142 (TF) plays a role in normal hemostasis by initiating the cell-surface assembly and propagation of the coagulation protease cascade.
- reducing or eliminating, such as knocking out, expression of CD 142 expression of MHC class II molecules is reduced thereby also reducing surface expression.
- such cells exhibit immune tolerance when engrafted into a recipient subject.
- the cell is considered hypoimmunogenic, e.g., in a recipient subject or patient upon administration.
- the target polynucleotide sequence is a variant of CD 142. In some embodiments, the target polynucleotide sequence is a homolog of CD 142. In some embodiments, the target polynucleotide sequence is an ortholog of CD 142.
- the modified pluripotent stem cells comprises a modification targeting the CD 142 gene.
- the modification targeting the CD 142 gene is by a targeted nuclease system that comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid sequence for specifically targeting the CD142 gene.
- the target polynucleotide sequence is CD142 or a variant of CD 142.
- the target polynucleotide sequence is a homolog of CD 142.
- the target polynucleotide sequence is an ortholog of CD 142.
- the cells outlined herein may comprise a modification targeting the CD142 gene.
- the modification targeting the CD142 gene by the rare-cutting endonuclease comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid (gRNA) sequence for specifically targeting the CD 142 gene.
- gRNA guide ribonucleic acid
- Assays to test whether the CD 142 gene has been inactivated are known and described herein.
- the resulting modification of the CD 142 gene by PCR and the reduction of CD 142 expression can be assays by FACS analysis.
- CD 142 protein expression is detected using a Western blot of cells lysates probed with antibodies to the CD142 protein.
- reverse transcriptase polymerase chain reactions RT-PCR
- Useful genomic, polynucleotide and polypeptide information about the human CD 142 are provided in, for example, the GeneCard Identifier GC01M094530, HGNC No.
- an exogenous nucleic acid or transgene encoding a polypeptide as disclosed herein e.g., a chimeric antigen receptor, CD46, CD59, CD55, or CD47 or another tolerogenic factor disclosed herein
- a polypeptide as disclosed herein e.g., a chimeric antigen receptor, CD46, CD59, CD55, or CD47 or another tolerogenic factor disclosed herein
- exemplary transgenes for targeted insertion at the CD142 locus include any as described herein.
- the reduction of the CD 142 expression or function in the modified cells can be measured using techniques known in the ail, such as Western blotting using antibodies to the protein, FACS techniques, RT-PCR techniques, etc.
- the modified cells can be tested to confirm that CD 142 is not expressed on the cell surface.
- Methods to assess surface expression include methods known in the ail (See Figure 21 of WO2018132783, for example) and generally is done using either Western Blots or FACS analysis based on commercial antibodies that bind to human CD142.
- the modified cells provided herein have a reduced susceptibility to IBMIR. Methods to assay for hypoimmunogenic phenotypes of the modified cells are described further below.
- the modification that reduces CD142 expression reduces CD 142 mRNA expression.
- the reduced mRNA expression of CD 142 is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification.
- the mRNA expression of CD 142 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more.
- the mRNA expression of CD 142 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less.
- the mRNA expression of CD142 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of CD 142 is eliminated (e.g., 0% expression of CD 142 mRNA). In some embodiments, the modification that reduces CD142 mRNA expression eliminates CD 142 gene activity.
- the modification that reduces CD142 expression reduces CD 142 protein expression.
- the reduced protein expression of CD 142 is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification.
- the protein expression of CD 142 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more.
- the protein expression of CD142 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less.
- the protein expression of CD142 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the protein expression of CD 142 is eliminated (e.g., 0% expression of CD 142 protein). In some embodiments, the modification that reduces CD 142 protein expression eliminates CD 142 gene activity.
- the modification that reduces CD142 expression comprises inactivation or disruption of the CD 142 gene. In some embodiments, the modification that reduces CD 142 expression comprises inactivation or disruption of one allele of the CD 142 gene. In some embodiments, the modification that reduces CD 142 expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the CD 142 gene.
- the modification comprises inactivation or disruption of one or more CD 142 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all CD142 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the CD 142 gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the CD 142 gene. In some embodiments, the modification is a deletion of genomic DNA of the CD 142 gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the CD 142 gene.
- Exemplary guide target sequences for CD 142 are known, for example, as shown in Table 5.
- modified pluripotent stem cells provided herein arc genetically modified, such as by introduction of one or more modifications into a cell to overexpress a desired polynucleotide in the cell.
- the cell to be modified is an unmodified cell that has not previously been introduced with the one or more modifications.
- the modified pluripotent stem cells provided herein are genetically modified to include one or more exogenous polynucleotides encoding an exogenous protein (also interchangeably used with the term “transgene”).
- the cells are modified to increase expression of certain genes that are tolerogenic (e.g., immune) factors that affect immune recognition and tolerance in a recipient.
- the one or more polynucleotides may be expressed (e.g. overexpressed) in the modified pluripotent stem cells together with one or more genetic modifications to reduce expression of a target polynucleotide described above, such as an MHC class I and/or MHC class II molecule or CD 142.
- a target polynucleotide described above such as an MHC class I and/or MHC class II molecule or CD 142.
- the provided modified pluripotent stem cells do not trigger or activate an immune response upon administration to a recipient subject.
- the modified pluripotent stem cell includes 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more different overexpressed polynucleotides.
- the overexpressed polynucleotide is an exogenous polynucleotide.
- the modified pluripotent stem cell includes 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more different exogenous polynucleotides.
- the overexpressed polynucleotide is an exogenous polynucleotide that is expressed episomally in the cells.
- the overexpressed polynucleotide is an exogenous polynucleotide that is inserted or integrated into one or more genomic loci of the modified cell.
- expression of a polynucleotide is increased, i.e., the polynucleotide is overexpressed, using a fusion protein containing a DNA-targeting domain and a transcriptional activator.
- a fusion protein containing a DNA-targeting domain and a transcriptional activator is known to a skilled artisan.
- the modified pluripotent stem cell contains one or more exogenous polynucleotides in which the one or more exogenous polynucleotides are inserted or integrated into a genomic locus of the cell by non-targeted insertion methods, such as by transduction with a lentiviral vector.
- the one or more exogenous polynucleotides are inserted or integrated into the genome of the cell by targeted insertion methods, such as by using homology directed repair (HDR). Any suitable method can be used to insert the exogenous polynucleotide into the genomic locus of the modified cell by HDR including the gene editing methods described herein (e.g., a CRISPR/Cas system).
- the one or more exogenous polynucleotides are inserted into one or more genomic locus, such as any genomic locus described herein (e.g., Table 6). In some embodiments, the exogenous polynucleotides are inserted into the same genomic loci. In some embodiments, the exogenous polynucleotides are inserted into different genomic loci. In some embodiments, the two or more of the exogenous polynucleotides are inserted into the same genomic loci, such as any genomic locus described herein (e.g., Table 6). In some embodiments, two or more exogenous polynucleotides are inserted into a different genomic loci, such as two or more genomic loci as described herein (e.g., Table 6).
- expression of a tolerogenic factor is overexpressed or increased in the cell.
- the modified pluripotent stem cell includes increased expression, i.e., overexpression, of at least one tolerogenic factor.
- the tolerogenic factor is any factor that promotes or contributes to promoting or inducing tolerance to the modified cell by the immune system (e.g., innate or adaptive immune system).
- the tolerogenic factor is DUX4, B2M-HLA-E, CD16, CD52, CD47, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, CCL21, MFGE8, SERPINB9, CD35, IL-39, CD16 Fc Receptor, IL15-RF, and H2-M3.
- the tolerogenic factor is CD47, PD-L1, HLA-E or HLA-G, CCL21, FasL, Serpinb9, CD200 or Mfge8, or any combination thereof.
- the one or more tolerogenic factors are selected from the group consisting of CD16, CD24, CD35, CD39, CD46, CD47, CD52, CD55, CD59, CD64, CD200, CCL22, CTLA4-lg, Cl inhibitor, FASL, 1DO1, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, IL- 10, IL-35, PD-L1, SERPINB9, CCL21, MFGE8, DUX4, B2M-HLA-E, CD27, IL-39, CD16 Fc Receptor, IL15-RF, H2-M3 (HLA-G), A20/TNFAIP3, CR1, HLA-F, and MANF.
- the cell includes at least one exogenous polynucleotide that includes a polynucleotide that encodes for a tolerogenic factor.
- at least one of the exogenous polynucleotides is a polynucleotide that encodes CD47.
- Provided herein are cells that do not trigger or activate an immune response upon administration to a recipient subject. As described above, in some embodiments, the cells are modified to increase expression of genes and tolerogenic (e.g., immune) factors that affect immune recognition and tolerance in a recipient.
- the present disclosure provides a cell or population thereof that has been modified to express the tolerogenic factor (e.g., immunomodulatory polypeptide), such as CD47.
- the present disclosure provides a method for altering a cell genome to express the tolerogenic factor (e.g., immunomodulatory polypeptide), such as CD47.
- the modified cell expresses an exogenous tolerogenic factor (e.g., immunomodulatory polypeptide), such as an exogenous CD47.
- overexpression or increasing expression of the exogenous polynucleotide is achieved by introducing into the cell (e.g., transducing the cell) within expression vector comprising a nucleotide sequence encoding a human CD47 polypeptide.
- the expression vector may be a viral vector, such as a lentiviral vector) or may be a non- viral vector.
- the cell is modified to contain one or more exogenous polynucleotides in which at least one of the exogenous polynucleotides includes a polynucleotide that encodes for a tolerogenic factor.
- the tolerogenic factor is DUX4, B2M-HLA-E, CD16, CD52, CD47, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, CCL21, MFGE8, SERPINB9, CD35, IL- 39, CD16 Fc Receptor, IL15-RF, and H2-M3.
- the tolerogenic factor is selected from CD47, PD-L1, HLA-E or HLA-G, CCL21, FasL, Serpinb9, CD200 or Mfge8, or any combination thereof (e.g., all thereof).
- the one or more tolerogenic factors are selected from the group consisting of CD16, CD24, CD35, CD39, CD46, CD47, CD52, CD55, CD59, CD64, CD200, CCL22, CTLA4-Ig, Cl inhibitor, FASL, IDO1, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, IL-10, IL-35, PD-L1, SERPINB9, CCL21, MFGE8, DUX4, B2M-HLA-E, CD27, IL-39, CD16 Fc Receptor, IL15-RF, H2-M3 (HLA-G), A20/TNFAIP3, CR1, HLA-F, and MANF.
- the exogenous polynucleotides is a polynucleotide that encodes CD47.
- the tolerogenic factor is CD47.
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD47, such as human CD47.
- CD47 is overexpressed in the cell.
- the expression of CD47 is overexpressed or increased in the modified cell compared to a similar cell of the same cell type that has not been modified with the modification, such as a reference or unmodified cell, e.g. a cell not modified with an exogenous polynucleotide encoding CD47.
- CD47 is a leukocyte surface antigen and has a role in cell adhesion and modulation of integrins. It is normally expressed on the surface of a cell and signals to circulating macrophages not to eat the cell.
- Useful genomic, polynucleotide and polypeptide information about human CD47 are provided in, for example, the NP_001768.1, NP_942088.1, NM_001777.3 and NM_198793.2.
- the modified pluripotent stem cell includes increased expression, i.e., overexpression, of at least one tolerogenic factor.
- the cell includes at least one exogenous polynucleotide that includes a polynucleotide that encodes for a tolerogenic factor.
- tolerogenic factors include DUX4, B2M-HLA-E, CD16, CD52, CD47, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, CCL21, MFGE8, SERPINB9, CD35, IL- 39, CD16 Fc Receptor, IL15-RF, and H2-M3, or any combination thereof.
- the one or more tolerogenic factors are selected from the group consisting of CD16, CD24, CD35, CD39, CD46, CD47, CD52, CD55, CD59, CD64, CD200, CCL22, CTLA4-Ig, Cl inhibitor, FASL, IDOL HLA-C, HLA-E, HLA-E heavy chain, HLA-G, IL- 10, IL-35, PD-L1, SERPINB9, CCL21, MFGE8, DUX4, B2M-HLA-E, CD27, IL-39, CDI6 Fc Receptor, IL15-RF, H2-M3 (HLA-G), A20/TNFAIP3, CR1, HLA-F, and MANF.
- at least one of the overexpressed (e.g., exogenous) polynucleotides is a polynucleotide that encodes CD47.
- the present disclosure provides a cell or population thereof that has been modified to express the tolerogenic factor (e.g., immunomodulatory polypeptide), such as CD47.
- the present disclosure provides a method for altering a cell genome to express the tolerogenic factor (e.g., immunomodulatory polypeptide), such as CD47.
- the modified pluripotent stem cell expresses an exogenous tolerogenic factor (e.g., immunomodulatory polypeptide), such as an exogenous CD47.
- the cell expresses an expression vector comprising a nucleotide sequence encoding a human CD47 polypeptide.
- the modified pluripotent stem cell contains an overexpressed polynucleotide that encodes CD47, such as human CD47.
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD47, such as human CD47.
- CD47 is overexpressed in the cell.
- the expression of CD47 is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CD47.
- the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD47 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1. In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD47 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1.
- the cell comprises an exogenous nucleotide sequence for CD47 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_001777.3 and NM_198793.2.
- the cell comprises an exogenous nucleotide sequence for CD47 as set forth in NCBI Ref. Sequence Nos. NM_001777.3 and NM_198793.2.
- the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD47 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1. In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD47 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1.
- the cell comprises an exogenous nucleotide sequence for CD47 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_001777.3 and NM_198793.2.
- the cell comprises an exogenous nucleotide sequence for CD47 as set forth in NCBI Ref. Sequence Nos. NM_001777.3 and NM_198793.2.
- the cell comprises an exogenous CD47 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1. In some embodiments, the cell outlined herein comprises an exogenous CD47 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1.
- the cell comprises an overexpressed polynucleotide encoding a CD47 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 1.
- the cell comprises an exogenous polynucleotide encoding a CD47 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 1.
- the cell comprises an overexpressed polynucleotide encoding a CD47 polypeptide having the amino acid sequence as set forth in SEQ ID NO: 1. In some embodiments, the cell comprises an exogenous polynucleotide encoding a CD47 polypeptide having the amino acid sequence as set forth in SEQ ID NO: 1.
- the cell comprises an overexpressed CD47 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 2.
- the cell comprises an exogenous CD47 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 2.
- the cell comprises an overexpressed CD47 polypeptide having the amino acid sequence as set forth in SEQ ID NO: 2.
- the cell comprises an exogenous CD47 polypeptide having the amino acid sequence as set forth in SEQ ID NO: 2.
- the exogenous nucleotide sequence encoding the CD59 polypeptide is operably linked to a sequence encoding a heterologous signal peptide.
- an exogenous polynucleotide encoding CD47 is integrated into the genome of the cell by targeted or non-targeted methods of insertion, such as described further below.
- targeted insertion is by homology-dependent insertion into a target locus, such as by insertion into any one of the gene loci depicted in Table 6, e.g. a B2M gene or a CIITA gene.
- targeted insertion is by homology-independent insertion, such as by insertion into a safe harbor locus.
- the polynucleotide encoding CD47 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding CD47 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- all or a functional portion of CD47 can be linked to other components such as a signal peptide, a leader sequence, a secretory signal, a label (e.g., a reporter gene), or any combination thereof.
- the nucleic acid sequence encoding a signal peptide of CD47 is replaced with a nucleic acid sequence encoding a signal peptide from a heterologous protein.
- the heterologous protein can be, for example, CD8a, CD28, tissue plasminogen activator (tPA), growth hormone, granulocyte-macrophage colony stimulating factor (GM-CSF), GM-CSF receptor (GM-CSFRa), or an immunoglobulin (e.g., IgE or IgK).
- the signal peptide is a signal peptide from an immunoglobulin (such as IgG heavy chain or IgG-kappa light chain), a cytokine (such as interleukin-2 (IL-2), or CD33), a serum albumin protein (e.g., HSA or albumin), a human azurocidin preprotein signal sequence, a luciferase, a trypsinogen (e.g. chymotrypsinogen or trypsinogen) or other signal peptide able to efficiently express a protein by or on a cell.
- an immunoglobulin such as IgG heavy chain or IgG-kappa light chain
- a cytokine such as interleukin-2 (IL-2), or CD33
- a serum albumin protein e.g., HSA or albumin
- a human azurocidin preprotein signal sequence e.g., a luciferase
- a trypsinogen e.
- the exogenous polynucleotide encoding CD47 is operably linked to a promoter.
- the exogenous polynucleotide encoding CD47 is inserted into any one of the gene loci depicted in Table 6.
- the exogenous polynucleotide encoding CD47 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the exogenous polynucleotide encoding CD47 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the exogenous polynucleotide encoding CD47 is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CRISPR/Cas system or any of the gene editing systems described herein
- CD47 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CD47 protein.
- reverse transcriptase polymerase chain reactions RT-PCR are used to confirm the presence of the exogenous CD47 mRNA.
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD200, such as human CD200.
- CD200 is overexpressed in the cell.
- the expression of CD200 is increased in the modified cell compared to a si milar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CD200.
- Useful genomic, polynucleotide and polypeptide information about human CD200 are provided in, for example, the GeneCard Identifier GC03P112332, HGNC No. 7203, NCBI Gene ID 4345, Uniprot No. P41217, and NCBI RefSeq Nos.
- the polynucleotide encoding CD200 is operably linked to a promoter.
- the polynucleotide encoding CD200 is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding CD200 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding CD200 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding CD200 is inserted into a B2M gene locus, a CIITA gene locus, or a CD142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CRISPR/Cas system or any of the gene editing systems described herein
- CD200 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CD200 protein.
- reverse transcriptase polymerase chain reactions RT-PCR
- RT-PCR reverse transcriptase polymerase chain reactions
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes HLA-E, such as human HLA-E.
- HLA-E is overexpressed in the cell.
- the expression of HLA-E is increased in the modified pluripotent stem cell compared to a similar’ reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding HLA-E.
- Useful genomic, polynucleotide and polypeptide information about human HLA-E are provided in, for example, the GeneCard Identifier GC06P047281, HGNC No.
- the polynucleotide encoding HLA-E is operably linked to a promoter.
- the polynucleotide encoding HLA-E is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding HLA-E is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding HLA-E is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding HLA- E is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CRISPR/Cas system or any of the gene editing systems described herein
- HLA-E protein expression is detected using a Western blot of cell lysates probed with antibodies against the HLA-E protein.
- RT-PCR reverse transcriptase polymerase chain reactions
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes HLA-G, such as human HLA-G.
- HLA-G is overexpressed in the cell.
- the expression of HLA-G is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding HLA-G.
- Useful genomic, polynucleotide and polypeptide information about human HLA-G are provided in, for example, the GeneCard Identifier GC06P047256, HGNC No.
- the polynucleotide encoding HLA-G is operably linked to a promoter.
- the polynucleotide encoding HLA-G is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding HLA-G is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding HLA-G is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding HLA- G is inserted into a B2M gene locus, a CIITA gene locus, or a CD142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- HLA-G protein expression is detected using a Western blot of cell lysates probed with antibodies against the HLA-G protein.
- RT-PCR reverse transcriptase polymerase chain reactions
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes PD-L1, such as human PD-LL
- PD-L1 is overexpressed in the cell.
- the expression of PD-L1 is increased in the modified cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding PD-LL
- Useful genomic, polynucleotide and polypeptide information about human PD-L1 or CD274 are provided in, for example, the GeneCard Identifier GC09P005450, HGNC No.
- polynucleotide encoding PD-L1 is operably linked to a promoter.
- the polynucleotide encoding PD-L1 is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding PD-L1 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding PD-L1 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding PD-L1 is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CRISPR/Cas system or any of the gene editing systems described herein
- PD-L1 protein expression is detected using a Western blot of cell lysates probed with antibodies against the PD-L1 protein.
- reverse transcriptase polymerase chain reactions RT-PCR
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes FasL, such as human FasL.
- FasL is overexpressed in the cell.
- the expression of FasL is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding FasL.
- Useful genomic, polynucleotide and polypeptide information about human Fas ligand are provided in, for example, the GeneCard Identifier GC01P172628, HGNC No. 11936, NCBI Gene ID 356, Uniprot No. P48023, and NCBI RefSeq Nos. NP_000630.1, NM_000639.2, NP_001289675.1, and NM_001302746.1.
- the polynucleotide encoding Fas-L is operably linked to a promoter.
- the polynucleotide encoding Fas-L is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding Fas-L is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding Fas-L is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding Fas-L is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CRISPR/Cas system or any of the gene editing systems described herein
- Fas-L protein expression is detected using a Western blot of cell lysates probed with antibodies against the Fas-L protein.
- RT-PCR reverse transcriptase polymerase chain reactions
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CCL21, such as human CCL21.
- CCL21 is overexpressed in the cell.
- the expression of CCL21 is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CCL21.
- Useful genomic, polynucleotide and polypeptide information about human CCL21 are provided in, for example, the GeneCard Identifier GC09M034709, HGNC No.
- the polynucleotide encoding CCL21 is operably linked to a promoter.
- the polynucleotide encoding CCL21 is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding CCL21 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding CCL21 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding CCL21 is inserted into a B2M gene locus, a CIITA gene locus, or a CD142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CRISPR/Cas system or any of the gene editing systems described herein
- CCL21 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CCL21 protein.
- RT-PCR reverse transcriptase polymerase chain reactions
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CCL22, such as human CCL22.
- CCL22 is overexpressed in the cell.
- the expression of CCL22 is increased in the modified cell compared to a similar- reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CCL22.
- Useful genomic, polynucleotide and polypeptide information about human CCL22 are provided in, for example, the GeneCard Identifier GC16P057359, HGNC No. 10621, NCBI Gene ID 6367, Uniprot No.
- the polynucleotide encoding CCL22 is operably linked to a promoter.
- the polynucleotide encoding CCL22 is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding CCL22 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding CCL22 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding CCL22 is inserted into a B2M gene locus, a CIITA gene locus, or a CD142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CRISPR/Cas system or any of the gene editing systems described herein
- CCL22 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CCL22 protein.
- RT-PCR reverse transcriptase polymerase chain reactions
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes Mfge8, such as human Mfge8.
- Mfge8 is overexpressed in the cell.
- the expression of Mfge8 is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding Mfge8.
- Useful genomic, polynucleotide and polypeptide information about human Mfge8 are provided in, for example, the GeneCard Identifier GC15MO88898, HGNC No.
- the polynucleotide encoding Mfge8 is operably linked to a promoter.
- the polynucleotide encoding Mfge8 is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding Mfge8 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding Mfge8 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding Mfge8 is inserted into a B2M gene locus, a CIITA gene locus, a CD 142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CRISPR/Cas system or any of the gene editing systems described herein
- Mfge8 protein expression is detected using a Western blot of cell lysates probed with antibodies against the Mfge8 protein.
- RT-PCR reverse transcriptase polymerase chain reactions
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes SerpinB9, such as human SerpinB9.
- SerpinB9 is overexpressed in the cell.
- the expression of SerpinB9 is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding SerpinB9.
- Useful genomic, polynucleotide and polypeptide information about human SerpinB9 are provided in, for example, the GeneCard Identifier GC06M002887, HGNC No.
- the polynucleotide encoding SerpinB9 is operably linked to a promoter. [05221 In some embodiments, the polynucleotide encoding SerpinB9 is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding SerpinB9 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- a safe harbor locus such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding SerpinB9 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding SerpinB9 is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CRISPR/Cas system or any of the gene editing systems described herein
- SerpinB9 protein expression is detected using a Western blot of cell lysates probed with antibodies against the SerpinB9 protein.
- reverse transcriptase polymerase chain reactions RT-PCR are used to confirm the presence of the exogenous SerpinB9 mRNA.
- the tolerogenic factor is CD47 and the cell includes an exogenous polynucleotide encoding a CD47 protein. In some embodiments, the cell expresses an exogenous CD47 polypeptide.
- a method disclosed herein comprises administering to a subject in need thereof a CD47-SIRPa blockade agent, wherein the subject was previously administered a population of cells engineered to express an exogenous CD47 polypeptide.
- the CD47-SIRPa blockade agent comprises a CD47 -binding domain.
- the CD47-binding domain comprises signal regulatory protein alpha (SIRPa) or a fragment thereof.
- the CD47-SIRPa blockade agent comprises an immunoglobulin G (IgG) Fc domain.
- the IgG Fc domain comprises an IgGl Fc domain.
- the IgGl Fc domain comprises a fragment of a human antibody.
- the CD47-SIRPa blockade agent is selected from the group consisting of TTI-621, TTI-622, and ALX148.
- the CD47- SIRPa blockade agent is TTI-621, TTI-622, and ALX148.
- the CD47- SIRPa blockade agent is TTI-622.
- the CD47-SIRPa blockade agent is ALX148.
- the IgG Fc domain comprises an IgG4 Fc domain.
- the CD47-SIRPa blockade agent is an antibody.
- the antibody is selected from the group consisting of M1AP410, B6H12, and Magrolimab. In some embodiments, the antibody is MIAP410. In some embodiments, the antibody is B6H12. In some embodiments, the antibody is Magrolimab. In some embodiments, the antibody is selected from the group consisting of AO- 176, IBI188 (letaplimab), STI-6643, and ZL-1201. In some embodiments, the antibody is AO- 176 (Arch). In some embodiments, the antibody is IBI188 (letaplimab) (Innovent). In some embodiments, the antibody is STI-6643 (Sorrento). In some embodiments, the antibody is ZL-1201 (Zai).
- useful antibodies or fragments thereof that bind CD47 can be selected from a group that includes magrolimab ((Hu5F9-G4)) (Forty Seven, Inc.; Gilead Sciences, Inc.), urabrelimab, CC-90002 (Celgene; Bristol-Myers Squibb), IBL188 (Innovent Biologies), IBL322 (Innovent Biologies), TG-1801 (TG Therapeutics; also known as NI-1701, Novimmune SA), ALX148 (ALX Oncology), TJ011133 (also known as TJC4, 1-Mab Biopharma), FA3M3, ZL-1201 (Zai Lab Co., Ltd), AK117 (Akesbio Australia Pty, Ltd.), AO- 176 (Arch Oncology), SRF231 (Surface Oncology), GenSci-059 (GeneScience), C47B157 (Janssen Research and Development), C47B161 (Janssen Research
- the antibody or fragment thereof does not compete for CD47 binding with an antibody selected from a group that includes magrolimab, urabrelimab, CC-90002, IBI-188, IBI-322, TG-1801 (NI-1701), ALX148, TJO11133, FA3M3, ZL1201, AK117, AO-176, SRF231, GenSci-059, C47B157, C47B161, C47B167, C47B222, C47B227, Vx-1004, HMBD004, SHR-1603, AMMS4-G4, RTX-CD47, and IMC-002.
- the antibody or fragment thereof competes for CD47 binding with an antibody selected from magrolimab, urabrelimab, CC-90002, IBI-188, IBI-322, TG-1801 (NI-1701), ALX148, TJ011133, FA3M3, ZL1201, AK117, AO-176, SRF231, GenSci-059, C47B157, C47B161, C47B167, C47B222, C47B227, Vx-1004, HMBD004, SHR-1603, AMMS4-G4, RTX-CD47, and 1MC-002.
- an antibody selected from magrolimab, urabrelimab, CC-90002, IBI-188, IBI-322, TG-1801 (NI-1701), ALX148, TJ011133, FA3M3, ZL1201, AK117, AO-176, SRF231, GenSci-059, C47B157, C47B161, C47B167, C47B222,
- the antibody or fragment thereof that binds CD47 is selected from a group that includes a single-chain Fv fragment (scFv) against CD47, a Fab against CD47, a VHH nanobody against CD47, a DARPin against CD47, and variants thereof.
- scFv single-chain Fv fragment
- the scFv against CD47, a Fab against CD47, and variants thereof are based on the antigen binding domains of any of the antibodies selected from a group that includes magrolimab, urabrelimab, CC-90002, IBI-188, IBI-322, TG-1801 (NI-1701), ALX148, TJO11133, FA3M3, ZL1201, AK117, AO-176, SRF231, GenSci-059, C47B157, C47B161, C47B167, C47B222, C47B227, Vx-1004, HMBD004, SHR-1603, AMMS4-G4, RTX-CD47, and IMC-002.
- the CD47 antagonist provides CD47 blockade.
- Methods and agents for CD47 blockade are described in PCT/US2021/054326, which is incorporated by reference in its entirety.
- the tolerogenic factor (e.g., CD47) is overexpressed in the modified PSC relative to the control or wild-type PSC.
- the tolerogenic factor (e.g. CD47) is expressed at a first level that is greater than at or about 3-fold, greater than at or about 5-fold, greater than at or about 10-fold, greater than at or about 20-fold, greater than at or about 30-fold, greater than at or about 40-fold, greater than at or about 50-fold, greater than at or about 60-fold, or greater than at or about 70-fold over a second level expressed by the control or wild-type PSC.
- the tolerogenic factor (e.g.
- CD47 is expressed by the modified PSC at greater than at or about 20,000 molecules per cell, at greater than at or about 30,000 molecules per cell, greater than at or about 50,000 molecules per cell, greater than at or about 100,000 molecules per cell, greater than at or about 200,000 molecules per cell, greater than at or about 300,000 molecules per cell, greater than at or about 400,000 molecules per cell, greater than at or about 500,000 molecules per cell, or greater than at or about 600,000 molecules per cell.
- the tolerogenic factor e.g., CD47
- CD47 is overexpressed in the engineered islet cell relative to the control or wild-type islet cell, such as an unmodified isslet cell differentiated from an unmodified PSC that does not contain the modifications.
- the tolerogenic factor (e.g. CD47) is expressed at a first level that is greater than at or about 3-fold, greater than at or about 5-fold, greater than at or about 10-fold, greater than at or about 20-fold, greater than at or about 30-fold, greater than at or about 40-fold, greater than at or about 50-fold, greater than at or about 60-fold, or greater than at or about 70-fold over a second level expressed by the control or wild-type islet cell.
- the tolerogenic factor (e.g. CD47) is expressed at a first level that is greater than at or about 3-fold, greater than at or about 5-fold, greater than at or about 10-fold, greater than at or about 20-fold, greater than at or about 30-fold, greater than at or about 40-fold, greater than at or about 50-fold, greater than at or about 60-fold, or greater than at or about 70-fold over a second level expressed by the control or wild-type islet cell.
- the tolerogenic factor (e.g.
- CD47 is expressed by the engineered islet cell at greater than at or about 20,000 molecules per cell, at greater than at or about 30,000 molecules per cell, greater than at or about 50,000 molecules per cell, greater than at or about 100,000 molecules per cell, greater than at or about 200,000 molecules per cell, greater than at or about 300,000 molecules per cell, greater than at or about 400,000 molecules per cell, greater than at or about 500,000 molecules per cell, or greater than at or about 600,000 molecules per cell.
- expression of one or more complement inhibitor is increased in the cell.
- the one or more complement inhibitor is one or more membrane-bound complement inhibitor.
- at least one of the exogenous polynucleotides includes a polynucleotide that encodes for a complement inhibitor.
- the one or more complement inhibitor is CD46, CD59, CD55, or CD35 or any combination thereof.
- the one or more complement inhibitor is CD46, CD59, CD55, or any combination thereof.
- at least one of the exogenous polynucleotides is a polynucleotide that encodes one or more complement inhibitors, such as CD46.
- the one or more complement inhibitors are CD46 and CD59, or CD46, CD59, and CD55.
- expression of CD46 and CD59 or CD46, CD59, and CD55 protects a cell or population thereof from complement-dependent cytotoxicity, including in the presence of antibodies against cell surface antigens expressed by the cell.
- the present disclosure provides a cell or population thereof that has been modified to express the one or more complement inhibitor, such as CD46, CD59, CD55, or any combination thereof.
- the one or more complement inhibitor is CD46 and CD59.
- the one or more complement inhibitor is CD46, CD59, and CD55.
- the present disclosure provides a method for altering a cell genome to express one or more complement inhibitor.
- the modified cell expresses one or more exogenous complement inhibitor, such as exogenous CD46 and CD59 or CD46, CD59, and CD55.
- the cell expresses an expression vector comprising a nucleotide sequence encoding a human CD46 polypeptide.
- the cell expresses an expression vector comprising a nucleotide sequence encoding a human CD59 polypeptide. In some instances, the cell expresses an expression vector comprising a nucleotide sequence encoding a human CD55 polypeptide. In some embodiments, the expression vector comprises nucleotide sequences encoding two or more complement inhibitors in any combination. In some embodiments, the expression vector comprises nucleotide sequences encoding CD46 and CD59. In some embodiments, the expression vector comprises nucleotide sequences encoding CD46, CD59, and CD55.
- the modified pluripotent stem cells contain an overexpressed polynucleotide that encodes CD46, such as human CD46.
- the modified pluripotent stem cells contain an exogenous polynucleotide that encodes CD46, such as human CD46.
- CD46 is overexpressed in the cell.
- the expression of CD46 is increased in the modified pluripotent stem cells compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CD46.
- CD46 is a membrane-bound complement inhibitor.
- complement factor I a serine protease which protects autologous cells against complement- mediated injury by cleaving C3b and C4b.
- Useful genomic, polynucleotide and polypeptide information about human CD46 are provided in, for example, the GeneCard Identifier GC01P207752, HGNC No. 6953, NCBI Gene ID 4179, Uniprot No. P15529, and NCBI Ref Scq Nos.
- the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD46 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_002380.3, NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1.
- the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD46 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_002380.3, NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1.
- the cell comprises an overexpressed nucleotide sequence for CD46 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_002389.4, NM_153826.3, NM_172350.2, NM_172351.2, NM_172352.2 NP_758860.1, NM_172353.2, NM_172359.2, and NM_172361.2.
- the cell comprises an overexpressed nucleotide sequence for CD46 as set forth in NCBI Ref.
- the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_002380.3, NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1.
- the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_002380.3, NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1.
- the cell comprises an exogenous nucleotide sequence for CD46 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_002389.4, NM_153826.3, NM_172350.2, NM_172351.2, NM_172352.2 NP_758860.1, NM_172353.2, NM_172359.2, and NM_172361.2.
- the cell comprises an exogenous nucleotide sequence for CD46 as set forth in NCBI Ref.
- the cell comprises an overexpressed CD46 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1.
- the cell comprises an exogenous CD46 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref.
- the cell outlined herein comprises an overexpressed CD46 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1.
- the cell outlined herein comprises an exogenous CD46 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1.
- a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD46 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 4.
- a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 4.
- a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD46 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 4.
- a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 4.
- a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 3.
- a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 3.
- the exogenous nucleotide sequence encoding the CD46 polypeptide is operably linked to a sequence encoding a heterologous signal peptide.
- all or a functional portion of CD46 can be linked to other components such as a signal peptide, a leader sequence, a secretory signal, a label (e.g., a reporter gene), or any combination thereof.
- the nucleic acid sequence encoding a signal peptide of CD46 is replaced with a nucleic acid sequence encoding a signal peptide from a heterologous protein.
- the heterologous protein can be, for example, CD8ot, CD28, tissue plasminogen activator (tPA), growth hormone, granulocyte-macrophage colony stimulating factor (GM-CSF), GM-CSF receptor (GM-CSFRa), or an immunoglobulin (e.g., IgE or IgK).
- the signal peptide is a signal peptide from an immunoglobulin (such as IgG heavy chain or IgG-kappa light chain), a cytokine (such as interleukin-2 (IL-2), or CD33), a serum albumin protein (e.g.
- HSA or albumin a human azurocidin preprotein signal sequence, a luciferase, a trypsinogen (e.g., chymotrypsinogen or trypsinogen) or other signal peptide able to efficiently express a protein by or on a cell.
- trypsinogen e.g., chymotrypsinogen or trypsinogen
- the exogenous polynucleotide encoding CD46 is operably linked to a promoter.
- the polynucleotide encoding CD46 is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding CD46 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, SHS231.
- the polynucleotide encoding CD46 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding CD46 is inserted into a B2M gene locus, a C11TA gene locus, or a CD 142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CRISPR/Cas system or any of the gene editing systems described herein
- CD46 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CD46 protein.
- reverse transcriptase polymerase chain reactions RT-PCR are used to confirm the presence of the exogenous CD46 mRNA.
- the modified pluripotent stem cell contains an overexpressed polynucleotide that encodes CD59, such as human CD59.
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD59, such as human CD59.
- CD59 is overexpressed in the cell.
- the expression of CD59 is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CD59.
- CD59 is a membrane-bound complement inhibitor. More specifically, CD59 is an inhibitor of complement membrane attack complex (MAC) activity.
- MAC complement membrane attack complex
- CD59 acts by binding to the C8 and/or C9 complements of the assembling MAC, thereby preventing incorporation of the multiple copies of C9 required for complete formation of the osmolytic pore.
- Useful genomic, polynucleotide and polypeptide information about human CD59 are provided in, for example, the GeneCard Identifier GC11M033704, HGNC No. 1689, NCBI Gene ID 966, Uniprot No.
- NP_000602.1 NM_000611 .5, NP_001120695. 1 , NM_001127223.1, NP_001120697.1, NM_001127225.1, NP_001120698.1, NM_001127226.1, NP_001120699.1, NM_001127227.1, NP_976074.1, NM_203329.2, NP_976075.1, NM_203330.2, NP_976076.1, and NM_203331.2.
- the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. Nos. NP-000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1,
- the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos.
- the cell comprises an overexpressed nucleotide sequence for CD59 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos.
- the cell comprises an overexpressed nucleotide sequence for CD59 as set forth in NCBI Ref. Sequence Nos. NM_000611.5, NM_001127223.1, NM_001127225.1, NM_001127226.1, NM_001127227.1, NM_203329.2, NM_203330.2, and NM_203331.2.
- the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1,
- the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1.
- the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1.
- the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos.
- the cell comprises an overexpressed nucleotide sequence for CD59 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos.
- the cell comprises an exogenous nucleotide sequence for CD59 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos.
- the cell comprises an overexpressed nucleotide sequence for CD59 as set forth in NCBI Ref. Sequence Nos.
- the cell comprises an exogenous nucleotide sequence for CD59 as set forth in NCBI Ref. Sequence Nos.
- NM_000611.5 NM_001127223.1, NM_001127225.1, NM_001127226.1, NM_001127227.1, NM_203329.2, NM_203330.2, and NM_203331.2.
- the cell comprises an overexpressed CD59 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1.
- 95% sequence identity e.g., 95%, 96%, 97%, 98%, 99%, or more
- the cell comprises an exogenous CD59 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1.
- the cell outlined herein comprises an overexpressed CD59 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos.
- the cell outlined herein comprises an exogenous CD59 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1 , NP_976074.1 , NP_976075.1 , and NP_976076.1.
- a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 6.
- a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 6.
- a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 6. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 6.
- a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 5.
- a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 5.
- a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, the exogenous nucleotide sequence encoding the CD59 polypeptide is operably linked to a sequence encoding a heterologous signal peptide.
- all or a functional portion of CD59 can be linked to other components such as a signal peptide, a leader sequence, a secretory signal, a label (e.g., a reporter gene), or any combination thereof.
- the nucleic acid sequence encoding a signal peptide of CD59 is replaced with a nucleic acid sequence encoding a signal peptide from a heterologous protein.
- the heterologous protein can be, for example, CD8a, CD28, tissue plasminogen activator (tPA), growth hormone, granulocyte-macrophage colony stimulating factor (GM-CSF), GM-CSF receptor (GM-CSFRa), or an immunoglobulin (e.g., IgE or IgK).
- the signal peptide is a signal peptide from an immunoglobulin (such as IgG heavy chain or IgG-kappa light chain), a cytokine (such as interleukin-2 (IL-2), or CD33), a serum albumin protein (e.g., HSA or albumin), a human azurocidin preprotein signal sequence, a luciferase, a trypsinogen (e.g. chymotrypsinogen or trypsinogen) or other signal peptide able to efficiently express a protein by or on a cell.
- an immunoglobulin such as IgG heavy chain or IgG-kappa light chain
- a cytokine such as interleukin-2 (IL-2), or CD33
- a serum albumin protein e.g., HSA or albumin
- a human azurocidin preprotein signal sequence e.g., a luciferase
- a trypsinogen e.
- the exogenous polynucleotide encoding CD59 is operably linked to a promoter.
- the polynucleotide encoding CD59 is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding CD59 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding CD59 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding CD59 is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CD59 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CD59 protein.
- reverse transcriptase polymerase chain reactions are used to confirm the presence of the exogenous CD59 mRNA.
- the modified pluripotent stem cell contains an overexpressed polynucleotide that encodes CD55, such as human CD55.
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD55, such as human CD55.
- CD55 is overexpressed in the cell.
- the expression of CD55 is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CD55.
- CD55 is a membrane-bound complement inhibitor.
- interaction of CD55 with cell-associated C4b and C3b polypeptides interferes with their ability to catalyze the conversion of C2 and factor B to enzymatically active C2a and Bb and thereby prevents the formation of C4b2a and C3bBb, the amplification convertases of the complement cascade.
- CD55 inhibits complement activation by destabilizing and preventing the formation of C3 and C5 convertases.
- Useful genomic, polynucleotide and polypeptide information about human CD55 are provided in, for example, the GeneCard Identifier GC01P207321, HGNC No. 2665, NCBI Gene ID 1604, Uniprot No. P08174, and NCBI RefSeq Nos. NM_000574.4,
- NM_001114752.2 NM_001300903.1, NM_001300904.1, NP_000565.I, NP_001108224.1, NP_001287832.1, and NP_001287833.1.
- the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide that has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref.
- the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1.
- the cell comprises an overexpressed nucleotide sequence for CD55 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCB1 Ref. Nos. NM_001777.3 and NM_198793.2.
- the cell comprises an overexpressed nucleotide sequence for CD55 as set forth in NCBI Ref. Sequence Nos. NM_000574.4, NM_001114752.2, NM_001300903.1 , and NM_001300904.1 .
- the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide that has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos.
- the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos.
- the cell comprises an exogenous nucleotide sequence for CD55 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_001777.3 and NM_198793.2.
- the cell comprises an exogenous nucleotide sequence for CD55 as set forth in NCBI Ref. Sequence Nos. NM_000574.4, NM_001114752.2, NM_001300903.1, and NM_001300904.1.
- the cell comprises an overexpressed CD55 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1.
- the cell comprises an exogenous CD55 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos.
- the cell outlined herein comprises an overexpressed CD55 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1. In some embodiments, the cell outlined herein comprises an exogenous CD55 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1.
- a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 9.
- a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 9.
- a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 9. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 9.
- a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 8.
- a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 8.
- a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 8. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 8. In some embodiments, the exogenous nucleotide sequence encoding the CD59 polypeptide is operably linked to a sequence encoding a heterologous signal peptide.
- all or a functional portion of CD55 can be linked to other components such as a signal peptide, a leader sequence, a secretory signal, a label (e.g., a reporter gene), or any combination thereof.
- the nucleic acid sequence encoding a signal peptide of CD55 is replaced with a nucleic acid sequence encoding a signal peptide from a heterologous protein.
- the heterologous protein can be, for example, CD8a, CD28, tissue plasminogen activator (tPA), growth hormone, granulocyte-macrophage colony stimulating factor (GM-CSF), GM-CSF receptor (GM-CSFRa), or an immunoglobulin (e.g., IgE or IgK).
- the signal peptide is a signal peptide from an immunoglobulin (such as IgG heavy chain or IgG-kappa light chain), a cytokine (such as interleukin-2 (IL-2), or CD33), a serum albumin protein (e.g.
- HSA or albumin a human azurocidin preprotein signal sequence, a luciferase, a trypsinogen (e.g., chymotrypsinogen or trypsinogen) or other signal peptide able to efficiently express a protein by or on a cell.
- trypsinogen e.g., chymotrypsinogen or trypsinogen
- the exogenous polynucleotide encoding CD55 is operably linked to a promoter.
- the polynucleotide encoding CD55 is inserted into any one of the gene loci depicted in Table 6.
- the polynucleotide encoding CD55 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231.
- the polynucleotide encoding CD55 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
- the polynucleotide encoding CD55 is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus.
- a suitable gene editing system e.g., CRISPR/Cas system or any of the gene editing systems described herein
- CRISPR/Cas system or any of the gene editing systems described herein
- CD55 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CD55 protein.
- reverse transcriptase polymerase chain reactions RT-PCR
- the cell comprises increased expression of none, one, two, or more complement inhibitors selected from the group consisting of CD46, CD59, and CD55, in any combination.
- the modified pluripotent stem cell contains an overexpressed polynucleotide that encodes CD46, such as any described above, and an overexpressed polynucleotide that encodes CD59, such as any described above.
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD46, such as any described above, and an exogenous polynucleotide that encodes CD59, such as any described above.
- the modified cell (comprising one or more modifications that increase expression of CD46 and CD59) comprises increased expression of CD46 and CD59 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46 and CD59).
- the modified pluripotent stem cell comprises between 1.5-fold and 2-fold, between 2-fold and 3-fold, between 3-fold and 4-fold, between 4-fold and 5-fold, between 5-fold and 10-fold, between 10-fold and 15-fold, between 15-fold and 20-fold, between 20-fold and 40-fold, between 40-fold and 60-fold, between 60-fold and 80-fold, between 80-fold and 100-fold, or between 100-fold and 200-fold increased expression of CD46 and CD59 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46 and CD59).
- the cell without the modification(s) does not have endogenous expression of CD46 and CD59 or does not have detectable expression of CD46 and CD59.
- the fold increase in expression compared to a cell lacking the modifications is greater than 200-fold.
- the modified pluripotent stem cells comprises between 2-fold and 200-fold, between 2-fold and 100-fold, between 2-fold and 50-fold, or between 2-fold and 20- fold increased expression of CD46 and CD59 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46 and CD59).
- the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46 and CD59) comprises between 5-fold and 200-fold, between 5-fold and 100-fold, between 5-fold and 50-fold, or between 5-fold and 20-fold increased expression of CD46 and CD59 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46 and CD59).
- the modified pluripotent stem cells (comprising one or more modifications that increase expression of CD46 and CD59) comprises increased expression of CD46 and CD59 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46 and CD59).
- the modified pluripotent stem cell comprises at least at or about 2-fold, at least at or about 4-fold, at least at or about 6- fold, at least at or about 10-fold, at least at or about and 15-fold, at least at or about 20-fold, at least at or about 30-fold, at least at or about 50-fold, at least at or about 60-fold, at least at or about 70-fold, at least at or about 80-fold, at least at or about 100-fold, or any value between any of the foregoing values, increased expression of CD46 and CD59 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46 and CD59).
- the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46 and CD59) comprises increased expression of CD46 and CD59 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46 and CD59).
- the modified cell comprises at or about 2-fold, at or about 4-fold, at or about 6-fold, at or about 10-fold, at or about and 15- fold, at or about 20-fold, at or about 30-fold, at or about 50-fold, at or about 60-fold, at or about 70-fold, at or about 80-fold, at or about 100-fold, or any value between any of the foregoing values, increased expression of CD46 and CD59 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46 and CD59).
- the cell comprises one or more transgenes encoding the CD46 and CD59.
- the transgenes are monocistronic or multicistronic vectors, as described below.
- the CD46 and CD59 are comprised by the same multicistronic vector, optionally in combination with one or more tolerogenic factors such as CD47.
- the CD46 and CD59 are comprised by different transgenes, optionally in combination with one or more tolerogenic factors such as CD47.
- the modified pluripotent stem cell contains an overexpressed polynucleotide that encodes CD46, such as any described above, an overexpressed polynucleotide that encodes CD59, such as any described above, and an overexpressed polynucleotide that encodes CD55, such as any described above.
- the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD46, such as any described above, an exogenous polynucleotide that encodes CD59, such as any described above, and an exogenous polynucleotide that encodes CD55, such as any described above.
- the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46, CD59, and CD55) comprises increased expression of CD46, CD59, and CD55 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46, CD59, and CD55).
- the modified cell comprises between 1 .5-fold and 2-fold, between 2-fold and 3-fold, between 3-fold and 4-fold, between 4-fold and 5-fold, between 5-fold and 10-fold, between 10- fold and 15-fold, between 15-fold and 20-fold, between 20-fold and 40-fold, between 40-fold and 60-fold, between 60-fold and 80-fold, between 80-fold and 100-fold, or between 100-fold and 200-fold increased expression of CD46, CD59, and CD55 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46, CD59, and CD55).
- the cell without the modification(s) does not have endogenous expression of CD46, CD59, and CD55or does not have detectable expression of CD46, CD59, and CD55.
- the fold increase in expression compared to a cell lacking the modifications is greater than 200-fold.
- the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46, CD59, and CD55) comprises between 2- fold and 200-fold, between 2-fold and 100- fold, between 2-fold and 50-fold, or between 2-fold and 20-fold increased expression of CD46, CD59, and CD55 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46, CD59, and CD55).
- the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46, CD59, and CD55) comprises between 5-fold and 200-fold, between 5-fold and 100-fold, between 5-fold and 50-fold, or between 5-fold and 20-fold increased expression of CD46, CD59, and CD55 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46, CD59, and CD55).
- the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46, CD59, and CD55) comprises increased expression of CD46, CD59, and CD55 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46 and CD59).
- the modified cell comprises at least at or about 2-fold, at least at or about 4-fold, at least at or about 6-fold, at least at or about 10-fold, at least at or about and 15-fold, at least at or about 20-fold, at least at or about 30-fold, at least at or about 50-fold, at least at or about 60-fold, at least at or about 70-fold, at least at or about 80-fold, at least at or about 100-fold, or any value between any of the foregoing values, increased expression of CD46, CD59, and CD55 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46, CD59, and CD55).
- modifications e.g., compared to endogenous expression of CD46, CD59, and CD55.
- the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46, CD59, and CD55) comprises increased expression of CD46, CD59, and CD55 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46, CD59, and CD55).
- the modified cell comprises at or about 2-fold, at or about 4-fold, at or about 6-fold, at or about 10- fold, at or about and 15-fold, at or about 20-fold, at or about 30-fold, at or about 50-fold, at or about 60-fold, at or about 70-fold, at or about 80-fold, at or about 100-fold, or any value between any of the foregoing values, increased expression of CD46, CD59, and CD55compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46, CD59, and CD55).
- the cell comprises one or more transgenes encoding the CD46, CD59, and CD55.
- the transgenes are monocistronic or multicistronic vectors, as described below.
- the CD46, CD59, and CD55 are comprised by the same multicistronic vector, optionally in combination with one or more tolerogenic factors such as CD47.
- the CD46, CD59, and CD55 are comprised by different transgenes, optionally in combination with one or more tolerogenic factors such as CD47. e. Modification Order and Timing
- provided modified pluripotent stem cells comprise a modification (e.g., genetic modifications).
- Modification (e.g., genetic modifications) in an iPSCs can be introduced at various time points and/or stages in the generation of a modified iPSC and/or a cell derived from a modified iPSC (e.g., a hypoimmune islet cell).
- a hypoimmune differentiated cell e.g., hypoimmune islet cell
- a hypoimmune differentiated cell comprises one or more modifications in addition to the one or more modifications present in the iPSC the islet cell is derived from.
- a hypoimmune differentiated cell e.g., hypoimmune islet cell
- a hypoimmune iPSC comprising one or more modifications is differentiated into hypoimmune differentiated cell (e.g., hypoimmune islet cell), wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises no additional modifications relative to the hypoimmune iPSC from which it is derived.
- an iPSC comprising one or more modifications is differentiated into a hypoimmune differentiated cell (e.g., hypoimmune islet cell), wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises one or more additional modifications relative to the iPSC from which it is derived.
- an iPSC comprising no hypoimmune modifications is differentiated into a hypoimmune differentiated cell (e.g., hypoimmune islet cell), wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) is modified to comprise one or more modifications relative to the iPSC from which it is derived.
- a hypoimmune differentiated cell e.g., hypoimmune islet cell
- the present disclosure provides methods of creating hypoimmune differentiated cell (e.g., hypoimmune islet cell) derived from iPSCs.
- the iPSCs are hypoimmune iPSCs.
- a method comprises engineering an iPSC to generate a hypoimmune iPSC comprising one or more modifications, and differentiating the hypoimmune iPSC into a hypoimmune differentiated cell (e.g., hypoimmune islet cell), wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises the same one or more modifications as the hypoimmune iPSC.
- a method comprises engineering an iPSC to generate an iPSC comprising one or more modifications, differentiating the iPSC into a hypoimmune differentiated cell (e.g., hypoimmune islet cell), and then engineering the hypoimmune differentiated cell (e.g., hypoimmune islet cell) to comprise one or more additional modifications, wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises one or more additional modifications relative to the iPSC.
- a hypoimmune differentiated cell e.g., hypoimmune islet cell
- a method comprises differentiating an iPSC into an islet cell and then engineering the islet cell to comprise one or more modifications, thereby generating a hypoimmune differentiated cell (e.g., hypoimmune islet cell), wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises one or more modifications relative to the iPSC.
- increased expression of a polynucleotide may be carried out by any of a variety of techniques. For instance, methods for modulating expression of genes and factors (proteins) include genome editing technologies, and RNA or protein expression technologies and the like. For all of these technologies, well known recombinant techniques are used, to generate recombinant nucleic acids as outlined herein.
- the cell that is modified with the one or more modification for overexpression or increased expression of a polynucleotide is any source cell as described herein.
- expression of a target gene is increased by expression of fusion protein or a protein complex containing (1) a site-specific binding domain specific for the endogenous CD47, or other gene and (2) a transcriptional activator.
- the regulatory factor is comprised of a site-specific DNA- binding nucleic acid molecule, such as a guide RNA (gRNA).
- gRNA guide RNA
- the method is achieved by site specific DNA-binding targeted proteins, such as zinc finger proteins (ZFP) or fusion proteins containing ZFP, which are also known as zinc finger nucleases (ZFNs).
- ZFP zinc finger proteins
- ZFNs zinc finger nucleases
- the regulatory factor comprises a site-specific binding domain, such as using a DNA binding protein or DNA-binding nucleic acid, which specifically binds to or hybridizes to the gene at a targeted region.
- the provided polynucleotides or polypeptides are coupled to or complexed with a site-specific nuclease, such as a modified nuclease.
- the administration is effected using a fusion comprising a DNA-targeting protein of a modified nuclease, such as a meganuclease or an RNA-guided nuclease such as a clustered regularly interspersed short palindromic nucleic acid (CRISPR)-Cas system, such as CRISPR-Cas9 system.
- a modified nuclease such as a meganuclease or an RNA-guided nuclease such as a clustered regularly interspersed short palindromic nucleic acid (CRISPR)-Cas system, such as CRISPR-Cas9 system.
- CRISPR clustered regularly interspersed short palindromic nucleic acid
- the nuclease is modified to lack nuclease activity.
- the modified nuclease is a catalytically dead dCas9.
- the site specific binding domain may be derived from a nuclease.
- the recognition sequences of homing endonucleases and meganucleases such as I-Scel, I-Ceul, PI-PspI, Pl-Sce, I-SceIV, I-CsmI, I-PanI, I-SceII, I-Ppol, I-SceIII, I-Crel, I-TevI, I-TevII and I-TevIII. See also U.S. Patent No. 5,420,032; U.S. Patent No. 6,833,252; Belfort et al., (1997) Nucleic Acids Res.
- Zinc finger, TALE, and CRISPR system binding domains can be “engineered” to bind to a predetermined nucleotide sequence, for example via engineering (altering one or more amino acids) of the recognition helix region of a naturally occurring zinc finger or TALE protein.
- Engineered DNA binding proteins are proteins that are non-naturally occurring. Rational criteria for design include application of substitution rules and computerized algorithms for processing information in a database storing information of existing ZFP and/or TALE designs and binding data. See, for example, U.S. Pat. Nos.
- the site-specific binding domain comprises one or more zinc-finger proteins (ZFPs) or domains thereof that bind to DNA in a sequence- specific manner.
- ZFPs zinc-finger proteins
- a ZFP or domain thereof is a protein or domain within a larger protein that binds DNA in a sequence-specific manner through one or more zinc fingers, regions of amino acid sequence within the binding domain whose structure is stabilized through coordination of a zinc ion.
- ZFPs are artificial ZFP domains targeting specific DNA sequences, typically 9-18 nucleotides long, generated by assembly of individual fingers.
- ZFPs include those in which a single finger domain is approximately 30 amino acids in length and contains an alpha helix containing two invariant histidine residues coordinated through zinc with two cysteines of a single beta turn, and having two, three, four, five, or six fingers.
- sequence-specificity of a ZFP may be altered by making amino acid substitutions at the four helix positions (-1, 2, 3 and 6) on a zinc finger recognition helix.
- the ZFP or ZFP- containing molecule is non-naturally occurring, e.g., is engineered to bind to a target site of choice.
- a target site of choice See, for example, Beerli et al. (2002) Nature Biotechnol. 20:135-141; Pabo et al. (2001) Ann. Rev. Biochem. 70:313-340; Isalan et al. (2001) Nature Biotechnol. 19:656-660; Segal et al. (2001) Curr. Opin. Biotechnol. 12:632-637; Choo et al. (2000) Curr. Opin. Struct. Biol. 10:411- 416; U.S. Pat. Nos. 6,453,242; 6,534,261; 6,599,692; 6,503,717; 6,689,558; 7,030,215;
- the site-specific binding domain comprises a naturally occurring or engineered (non-naturally occurring) transcription activator-like protein (TAL) DNA binding domain, such as in a transcription activator-like protein effector (TALE) protein, See, e.g., U.S. Patent Publication No. 20110301073, incorporated by reference in its entirety herein.
- TAL transcription activator-like protein
- TALE transcription activator-like protein effector
- the site-specific binding domain is derived from the CRISPR/Cas system.
- CRISPR system refers collectively to transcripts and other elements involved in the expression of or directing the activity of CRIS PR-associated (“Cas”) genes, including sequences encoding a Cas gene, a tracr (trans-activating CRISPR) sequence (e.g.
- a guide sequence includes a targeting domain comprising a polynucleotide sequence having sufficient complementarity with a target polynucleotide sequence to hybridize with the target sequence and direct sequence-specific binding of the CR1SPR complex to the target sequence.
- the degree of complementarity between a guide sequence and its corresponding target sequence, when optimally aligned using a suitable alignment algorithm is about or more than about 50%, 60%, 75%, 80%, 85%, 90%, 95%, 97.5%, 99%, or more.
- the targeting domain (e.g., targeting sequence) of the gRNA is complementary, e.g., at least 80, 85, 90, 95, 98 or 99% complementary, e.g., fully complementary, to the target sequence on the target nucleic acid.
- the gRNA may be any as described herein.
- the gRNA has a targeting sequence that is complementary to a target site of CD47, such as set forth in any one of SEQ ID NOS:200784-231885 (Table 29, Appendix 22 of W02016183041); HLA-E, such as set forth in any one of SEQ ID NOS:189859-193183 (Table 19, Appendix 12 of W02016183041); HLA-F, such as set forth in any one of SEQ ID NOS: 688808-699754 (Table 45, Appendix 38 of W02016183041); HLA-G, such as set forth in any one of SEQ ID NOS: 188372-189858 (Table 18, Appendix 11 of WO2016183041); or PD-L1, such as set forth in any one of SEQ ID NOS: 193184-200783 (Table 21, Appendix 14 of W02016183041).
- the target site is upstream of a transcription initiation site of the target gene. In some embodiments, the target site is adjacent to a transcription initiation site of the gene. In some embodiments, the target site is adjacent to an RNA polymerase pause site downstream of a transcription initiation site of the gene.
- the targeting domain is configured to target the promoter region of the target gene to promote transcription initiation, binding of one or more transcription enhancers or activators, and/or RNA polymerase.
- One or more gRNA can be used to target the promoter region of the gene.
- one or more regions of the gene can be targeted.
- the target sites are within 600 base pairs on either side of a transcription start site (TSS) of the gene.
- TSS transcription start site
- gRNA single guide RNA
- the gRNA sequence is or comprises a targeting sequence with minimal off- target binding to a non-target gene.
- the regulatory factor further comprises a functional domain, e.g., a transcriptional activator.
- the transcriptional activator is or contains one or more regulatory elements, such as one or more transcriptional control elements of a target gene, whereby a site-specific domain as provided above is recognized to drive expression of such gene.
- the transcriptional activator drives expression of the target gene.
- the transcriptional activator can be or contain all or a portion of a heterologous transactivation domain.
- the transcriptional activator is selected from Herpes simplex-derived transactivation domain, Dnmt3a methyltransferase domain, p65, VP16, and VP64.
- the regulatory factor is a zinc finger transcription factor (ZF-TF). In some embodiments, the regulatory factor is VP64-p65-Rta (VPR).
- the regulatory factor further comprises a transcriptional regulatory domain.
- Common domains include, e.g., transcription factor domains (activators, repressors, co-activators, co-repressors), silencers, oncogenes (e.g., myc, jun, fos, myb, max, mad, rel, ets, bcl, myb, mos family members etc.); DNA repair enzymes and their associated factors and modifiers; DNA rearrangement enzymes and their associated factors and modifiers; chromatin associated proteins and their modifiers (e.g.
- kinases e.g., kinases, acetylases and deacetylases
- DNA modifying enzymes e.g., methyltransferases such as members of the DNMT family (e.g., DNMT1, DNMT3A, DNMT3B, DNMT3L, etc., topoisomerases, helicases, ligases, kinases, phosphatases, polymerases, endonucleases) and their associated factors and modifiers. See, e.g., U.S. Publication No. 2013/0253040, incorporated by reference in its entirety herein.
- Suitable domains for achieving activation include the HSV VP 16 activation domain (see, e.g., Hagmann et al, J. Virol. 71, 5952-5962 (1 97)) nuclear hormone receptors (see, e.g., Torchia et al., Curr. Opin. Cell. Biol. 10:373-383 (1998)); the p65 subunit of nuclear factor kappa B (Bitko & Bank, J. Virol. 72:5610-5618 (1998) and Doyle & Hunt, Neuroreport 8:2937- 2942 (1997)); Liu et al., Cancer Gene Ther.
- HSV VP 16 activation domain see, e.g., Hagmann et al, J. Virol. 71, 5952-5962 (1 97)
- nuclear hormone receptors see, e.g., Torchia et al., Curr. Opin. Cell. Biol. 10:373-383 (1998)
- chimeric functional domains such as VP64 (Beerli et al., (1998) Proc. Natl. Acad. Sci. USA 95:14623-33), and degron (Molinari et al., (1999) EMBO J. 18, 6439-6447).
- Additional exemplary activation domains include, Oct 1, Oct-2A, Spl, AP-2, and CTF1 (Seipel et al, EMBOJ. 11, 4961-4968 (1992) as well as p300, CBP, PCAF, SRC1 PvALF, AtHD2A and ERF-2. See, for example, Robyr et al, (2000) Mol. Endocrinol.
- Additional exemplary activation domains include, but are not limited to, OsGAI, HALF-1, Cl, API, ARF-5, -6,-1, and -8, CPRF1, CPRF4, MYC-RP/GP, and TRAB1 , See, for example, Ogawa et al, (2000) Gene 245:21-29; Okanami et al, (1996) Genes Cells 1 :87-99; Goff et al, (1991) Genes Dev. 5:298-309; Cho et al, (1999) Plant Mol Biol 40:419-429; Ulmason et al, (1999) Proc. Natl. Acad. Sci.
- Exemplary repression domains that can be used to make genetic repressors include, but are not limited to, KRAB A/B, KOX, TGF-beta- inducible early gene (TIEG), v- erbA, SID, MBD2, MBD3, members of the DNMT family (e.g., DNMT1, DNMT3A, DNMT3B, DNMT3L, etc.), Rb, and MeCP2.
- KRAB A/B KOX
- TGF-beta- inducible early gene TIEG
- v- erbA TGF-beta- inducible early gene
- SID v- erbA
- MBD2 MBD3, members of the DNMT family (e.g., DNMT1, DNMT3A, DNMT3B, DNMT3L, etc.), Rb, and MeCP2.
- Additional exemplary repression domains include, but are not limited to, R0M2 and AtHD2A. See, for example, Chem et al, (1996) Plant Cell 8:305-321; and Wu et al, (2000) Plant J. 22:19-27.
- the domain is involved in epigenetic regulation of a chromosome.
- the domain is a histone acetyltransferase (HAT), e.g., type- A, nuclear localized such as MYST family members MOZ, Ybf2/Sas3, MOF, and Tip60, GNAT family members Gcn5 or pCAF, the p300 family members CBP, p300 or RttlO9 (Bemdsen and Denu (2008) Curr Opin Struct Biol 18(6):682-689).
- HAT histone acetyltransferase
- the domain is a histone deacetylase (HD AC) such as the class I (HDAC-1, 2, 3, and 8), class II (HD AC IIA (HDAC-4, 5, 7 and 9), HD AC IIB (HDAC 6 and 10)), class IV (HDAC-1 1), class III (also known as sirtuins (SIRTs); SIRT1-7) (see Mottamal et al., (2015) Molecules 20(3):3898-3941).
- HD AC histone deacetylase
- Another domain that is used in some embodiments is a histone phosphorylase or kinase, where examples include MSK1, MSK2, ATR, ATM, DNA-PK, Bubl, VprBP, IKK-a, PKCpi, Dik/Zip, JAK2, PKC5, WSTF and CK2.
- a methylation domain is used and may be chosen from groups such as Ezh2, PRMT1/6, PRMT5/7, PRMT 2/6, CARMI, set7/9, MLL, ALL-1, Suv 39h, G9a, SETDB1, Ezh2, Set2, Doti, PRMT 1/6, PRMT 5/7, PR-Set7 and Suv4-20h, Domains involved in sumoylation and biotinylation (Lys9, 13, 4, 18 and 12) may also be used in some embodiments (review see Kousarides (2007) Cell 128:693-705).
- Fusion molecules are constructed by methods of cloning and biochemical conjugation that are well known to those of skill in the art. Fusion molecules comprise a DNA- binding domain and a functional domain (e.g., a transcriptional activation or repression domain). Fusion molecules also optionally comprise nuclear localization signals (such as, for example, that from the SV40 medium T-antigen) and epitope tags (such as, for example, FLAG and hemagglutinin). Fusion proteins (and nucleic acids encoding them) are designed such that the translational reading frame is preserved among the components of the fusion.
- nuclear localization signals such as, for example, that from the SV40 medium T-antigen
- epitope tags such as, for example, FLAG and hemagglutinin
- Fusions between a polypeptide component of a functional domain (or a functional fragment thereof) on the one hand, and a non-protein DNA-binding domain (e.g., antibiotic, intercalator, minor groove binder, nucleic acid) on the other, arc constructed by methods of biochemical conjugation known to those of skill in the art. See, for example, the Pierce Chemical Company (Rockford, IL) Catalogue. Methods and compositions for making fusions between a minor groove binder and a polypeptide have been described. Mapp et al, (2000) Proc. Natl. Acad. Sci. USA 97:3930-3935. Likewise, CRISPR/Cas TFs and nucleases comprising a sgRNA nucleic acid component in association with a polypeptide component function domain arc also known to those of skill in the art and detailed herein.
- a non-protein DNA-binding domain e.g., antibiotic, intercalator, minor groove binder, nucleic acid
- increased expression (i.e., overexpression) of the polynucleotide is mediated by introducing into the cell an exogenous polynucleotide encoding the polynucleotide to be overexpressed.
- the exogenous polynucleotide is a recombinant nucleic acid.
- Well-known recombinant techniques can be used to generate recombinant nucleic acids as outlined herein.
- an exogenous polynucleotide encoding an exogenous polypeptide herein comprises a codon-optimized nucleic acid sequence.
- the recombinant nucleic acids encoding an exogenous polypeptide may be operably linked to one or more regulatory nucleotide sequences in an expression construct.
- Regulatory nucleotide sequences will generally be appropriate for the host cell and recipient subject to be treated. Numerous types of appropriate expression vectors and suitable regulatory sequences are known in the art for a variety of host cells.
- the one or more regulatory nucleotide sequences may include, but are not limited to, promoter sequences, leader or signal sequences, ribosomal binding sites, transcriptional start and termination sequences, translational start and termination sequences, and enhancer or activator sequences.
- the promoters may be either naturally occurring promoters, or hybrid promoters that combine elements of more than one promoter.
- An expression construct may be present in a cell on an episome, such as a plasmid, or the expression construct may be inserted in a chromosome.
- the expression vector includes a selectable marker gene to allow the selection of transformed host cells.
- Certain embodiments include an expression vector comprising a nucleotide sequence encoding a variant polypeptide operably linked to at least one regulatory sequence. Regulatory sequence for use herein include promoters, enhancers, and other expression control elements.
- an expression vector is designed for the choice of the host cell to be transformed, the particular variant polypeptide desired to be expressed, the vector's copy number, the ability to control that copy number, and/or the expression of any other protein encoded by the vector, such as antibiotic markers.
- the exogenous polynucleotide is operably linked to a promoter for expression of the exogenous polynucleotide in the modified cell.
- suitable mammalian promoters include, for example, promoters from the following genes: elongation factor 1 alpha (EFla) promoter, ubiquitin/S27a promoter of the hamster (WO 97/15664), Simian vacuolating virus 40 (SV40) early promoter, adenovirus major late promoter, mouse metallothionein-I promoter, the long terminal repeat region of Rous Sarcoma Virus (RSV), mouse mammary tumor virus promoter (MMTV), Moloney murine leukemia virus Long Terminal repeat region, and the early promoter of human Cytomegalovirus (CMV).
- EFla elongation factor 1 alpha
- SV40 Simian vacuolating virus 40
- SV40 Simian vacuolating virus 40
- SV40 Simian vacuo
- heterologous mammalian promoters examples include the actin, immunoglobulin or heat shock promoter(s).
- promoters for use in mammalian host cells can be obtained from the genomes of viruses such as polyoma virus, fowlpox virus (UK 2,211,504 published 5 Jul. 1989), bovine papilloma virus, avian sarcoma virus, cytomegalovirus, a retrovirus, hepatitis-B virus and Simian Virus 40 (SV40).
- viruses such as polyoma virus, fowlpox virus (UK 2,211,504 published 5 Jul. 1989), bovine papilloma virus, avian sarcoma virus, cytomegalovirus, a retrovirus, hepatitis-B virus and Simian Virus 40 (SV40).
- heterologous mammalian promoters are used. Examples include the actin promoter, an immunoglobulin promoter, and heat-shock promoters.
- the early and late promoters of SV40 are conveniently obtained as an SV40 restriction fragment which also contains the SV40 viral origin of replication (Fiers et al, Nature 273: 113-120 (1978)).
- the immediate early promoter of the human cytomegalovirus is conveniently obtained as a Hindlll restriction enzyme fragment (Greenaway et al, Gene 18: 355-360 (1982)).
- the foregoing references are incorporated by reference in their entirety.
- the expression vector is a bicistronic or multicistronic expression vector.
- Bicistronic or multicistronic expression vectors may include (1) multiple promoters fused to each of the open reading frames; (2) insertion of splicing signals between genes; (3) fusion of genes whose expressions are driven by a single promoter; and/or (4) insertion of proteolytic cleavage sites between genes (self-cleavage peptide) or insertion of internal ribosomal entry sites (IRESs) between genes.
- an expression vector or construct herein is a multicistronic construct.
- multicistronic construct and “multicistronic vector” are used interchangeably herein and refer to a recombinant DNA construct that is to be transcribed into a single mRNA molecule, wherein the single mRNA molecule encodes two or more genes (e.g., two or more transgenes).
- the multi-cistronic construct is referred to as bicistronic construct if it encodes two genes, and tricistronic construct if it encodes three genes, and quadrocistronic construct if it encodes four genes, and so on.
- two or more exogenous polynucleotides comprised by a vector or construct are each separated by a multicistronic separation element.
- the multicistronic separation element is an IRES or a sequence encoding a cleavable peptide or ribosomal skip element.
- the multicistronic separation element is an IRES, such as an encephalomyocarditis (EMCV) virus IRES.
- the multicistronic separation element is a cleavable peptide such as a 2A peptide.
- Exemplary 2A peptides include a P2A peptide, a T2A peptide, an E2A peptide, and an F2Apeptide.
- the cleavable peptide is a T2A.
- the two or more exogenous polynucleotides e.g. the first exogenous polynucleotide and second exogenous polynucleotide
- the first exogenous polynucleotide and the second exogenous polynucleotide are each operably linked to a promoter.
- the promoter is the same promoter.
- the promoter is an EFl promoter.
- an exogenous polynucleotide encoding an exogenous polypeptide encodes a cleavable peptide or ribosomal skip element, such as T2A at the N- terminus or C-terminus of an exogenous polypeptide encoded by a multicistronic vector.
- a cleavable peptide or ribosomal skip element such as T2A at the N- terminus or C-terminus of an exogenous polypeptide encoded by a multicistronic vector.
- inclusion of the cleavable peptide or ribosomal skip element allows for expression of two or more polypeptides from a single translation initiation site.
- the cleavable peptide is a T2A.
- the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 11. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 12. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 17. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 18.
- the vector or construct includes a single promoter that drives the expression of one or more transcription units of an exogenous polynucleotide.
- such vectors or constructs can be multicistronic (bicistronic or tricistronic, see e.g., U.S. Patent No. 6,060,273).
- transcription units can be engineered as a bicistronic unit containing an IRES (internal ribosome entry site), which allows coexpression of gene products (e.g., one or more tolerogenic factors such as CD47 and/or one or more complement inhibitor such as CD46, CD59, and CD55) from an RNA transcribed from a single promoter.
- IRES internal ribosome entry site
- the vectors or constructs provided herein are bicistronic, allowing the vector or construct to express two separate polypeptides.
- the two separate polypeptides encoded by the vector or construct are tolerogenic factors (e.g., two factors selected from CD47, DUX4, CD24, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA- G, PD-L1, IDOL CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, IL-39, FasL, CCL21, CCL22, Mfge8, and Serpinb9).
- the one or more tolerogenic factors are selected from the group consisting of CD16, CD24, CD35, CD39, CD46, CD47, CD52, CD55, CD59, CD64, CD200, CCL22, CTLA4-Ig, Cl inhibitor, FASL, IDOL HLA-C, HLA-E, HLA-E heavy chain, HLA-G, IL-10, IL-35, PD-L1, SERPINB9, CCL21, MFGE8, DUX4, B2M-HLA-E, CD27, IL- 39, CD16 Fc Receptor, IL15-RF, H2-M3 (HLA-G), A20/TNFAIP3, CR1, HLA-F, and MANF.
- the two separate polypeptides encoded by the vector or construct are CD46 and CD59. In some embodiments, the two separate polypeptides encoded by the vector or construct are a tolerogenic factor (e.g., CD47) and a complement inhibitor selected from CD46, CD59, and CD55.
- the vectors or constructs provided herein are tricistronic, allowing the vector or construct to express three separate polypeptides. In some cases, the three nucleic acid sequences of the tricistronic vector or construct are a tolerogenic factor such as CD47, CD46, and CD59. In some cases, the three nucleic acid sequences of the tricistronic vector or construct are CD46, CD59, and CD55.
- the three nucleic acid sequences of the tricistronic vector or construct are three tolerogenic factors selected from CD47, DUX4, CD24, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl- Inhibitor, IL-10, IL-35, IL-39, EasL, CCL21, CCL22, Mfge8, and Serpinb9.
- the vectors or constructs provided herein are quadrocistronic, allowing the vector or construct to express four separate polypeptides.
- the four separate polypeptides of the quadrocistronic vector or construct are CD47, CD46, CD59, and CD55. In some cases, the four separate polypeptides of the quadrocistronic vector or construct are four tolerogenic factors selected from CD47, DUX4, CD24, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA- G, PD-L1, IDOL CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, IL-39, LasL, CCL21, CCL22, Mfge8, and Serpinb9.
- the cell comprises one or more vectors or constructs, wherein each vector or construct is a monocistronic or a multicistronic construct as described above, and the monocistronic or multicistronic constructs encode one or more tolerogenic factors and/or complement inhibitors, in any combination or order.
- a single promoter directs expression of an RNA that contains, in a single open reading frame (ORE), two, three, or four genes (e.g., encoding a tolerogenic factor (e.g., CD47) and/or one or more complement inhibitors selected from CD46, CD59, and CD55) separated from one another by sequences encoding a self-cleavage peptide (e.g., 2A sequences) or a protease recognition site (e.g., furin).
- the ORE thus encodes a single polypeptide, which, either during (in the case of 2A) or after translation, is processed into the individual proteins.
- the peptide such as T2A
- T2A can cause the ribosome to skip (ribosome skipping) synthesis of a peptide bond at the C-terminus of a 2A element, leading to separation between the end of the 2A sequence and the next peptide downstream (see, for example, de Felipe. Genetic Vaccines and Ther. 2:13 (2004) and deFelipe et al. Traffic 5:616- 626 (2004)).
- Many 2A elements are known in the ail.
- Examples of 2A sequences that can be used in the methods and nucleic acids disclosed herein include, without limitation, 2A sequences from the foot-and-mouth disease virus (F2A, e.g., SEQ ID NO: 16), equine rhinitis A virus (E2A, e.g., SEQ ID NO: 15), thosea asigna vims (T2A, e.g., SEQ ID NO: 11, 12, 17, or 18), and porcine teschovirus- 1 (P2A, e.g., SEQ ID NO: 13 or 14) as described in U.S. Patent Publication No. 20070116690.
- F2A foot-and-mouth disease virus
- E2A equine rhinitis A virus
- T2A e.g., SEQ ID NO: 11, 12, 17, or 18
- P2A porcine teschovirus- 1
- the vector or construct (e.g., transgene) contains more than one nucleic acid sequence encoding a protein, e.g., a first exogenous polynucleotide encoding CD46 and a second exogenous polynucleotide encoding CD59, or a first exogenous polynucleotide encoding CD47, a second exogenous polynucleotide encoding CD56, and a third exogenous polynucleotide encoding CD59
- the vector or construct (e.g., transgene) may further include a nucleic acid sequence encoding a peptide between the first and second exogenous polynucleotide sequences.
- the nucleic acid sequence positioned between the first and second exogenous polynucleotides encodes a peptide that separates the translation products of the first and second exogenous polynucleotides during or after translation.
- the peptide contains a self-cleaving peptide or a peptide that causes ribosome skipping (a ribosomal skip element), such as a T2A peptide.
- inclusion of the cleavable peptide or ribosomal skip element allows for expression of two or more polypeptides from a single translation initiation site.
- the peptide is a self-cleaving peptide that is a T2A peptide.
- the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 11. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 12. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 17. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 18.
- the process of introducing the polynucleotides described herein into cells can be achieved by any suitable technique.
- Suitable techniques include calcium phosphate or lipid- mediated transfection, electroporation, fusogens, and transduction or infection using a viral vector.
- the polynucleotides are introduced into a cell via viral transduction (e.g., lentiviral transduction) or otherwise delivered on a viral vector (e.g., fusogen- mediated delivery).
- Suitable techniques include calcium phosphate or lipid-mediated transfection, electroporation, transposase-mediated delivery, and transduction or infection using a viral vector.
- the polynucleotides are introduced into a cell via viral transduction (e.g., lentiviral transduction) or otherwise delivered on a viral vector (e.g., fusogen- mediated delivery).
- viral transduction e.g., lentiviral transduction
- viral vector e.g., fusogen- mediated delivery
- vectors that package a polynucleotide encoding an exogenous polynucleotide may be used to deliver the packaged polynucleotides to a cell or population of cells.
- These vectors may be of any kind, including DNA vectors, RNA vectors, plasmids, viral vectors and particles.
- lipid nanoparticles can be used to deliver an exogenous polynucleotide to a cell.
- viral vectors can be used to deliver an exogenous polynucleotide to a cell.
- Viral vector technology is well known and described in Sambrook et al. (2001, Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory, New York).
- Viruses, which are useful as vectors include, but are not limited to lentiviral vectors, adenoviral vectors, adeno-associated viral (AAV) vectors, herpes simplex viral vectors, retroviral vectors, oncolytic viruses, and the like.
- the introduction of the exogenous polynucleotide into the cell can be specific (targeted) or nonspecific (e.g., non-targeted).
- the introduction of the exogenous polynucleotide into the cell can result in integration or insertion into the genome in the cell.
- the introduced exogenous polynucleotide may be non-integrating or episomal in the cell.
- a skilled artisan is familial' with methods of introducing nucleic acid transgenes into a cell, including any of the exemplary methods described herein, and can choose a suitable method.
- an exogenous polynucleotide is introduced into a cell (e.g., source cell) by any of a variety of non-targeted methods.
- the exogenous polynucleotide is inserted into a random genomic locus of a host cell.
- viral vectors including, for example, retroviral vectors and lentiviral vectors are commonly used to deliver genetic material into host cells and randomly insert the foreign or exogenous gene into the host cell genome to facilitate stable expression and replication of the gene.
- the non-targeted introduction of the exogenous polynucleotide into the cell is under conditions for stable expression of the exogenous polynucleotide in the cell.
- methods for introducing a nucleic acid for stable expression in a cell involves any method that results in stable integration of the nucleic acid into the genome of the cell, such that it may be propagated if the cell it has integrated into divides.
- the viral vector is a lentiviral vector.
- Lentiviral vectors are particularly useful means for successful viral transduction as they permit stable expression of the gene contained within the delivered nucleic acid transcript.
- Lentiviral vectors express reverse transcriptase and integrase, two enzymes required for stable expression of the gene contained within the delivered nucleic acid transcript.
- Reverse transcriptase converts an RNA transcript into DNA, while integrase inserts and integrates the DNA into the genome of the target cell. Once the DNA has been integrated stably into the genome, it divides along with the host.
- the gene of interest contained within the integrated DNA may be expressed constitutively or it may be inducible. As part of the host cell genome, it may be subject to cellular regulation, including activation or repression, depending on a host of factors in the target cell.
- Lentiviruses are subgroup of the Retroviridae family of viruses, named because reverse transcription of viral RNA genomes to DNA is required before integration into the host genome. As such, the most important features of lentiviral vehicles/particles are the integration of their genetic material into the genome of a target/host cell.
- lentivirus examples include the Human Immunodeficiency Viruses: HIV-1 and HIV -2, the Simian Immunodeficiency Vims (SIV), feline immunodeficiency vims (FIV), bovine immunodeficiency vims (BIV), Jembrana Disease Virus (JDV), equine infectious anemia virus (EIAV), equine infectious anemia, virus, visna-maedi and caprine arthritis encephalitis vims (CAEV).
- SIV Simian Immunodeficiency Vims
- FV feline immunodeficiency vims
- BIV bovine immunodeficiency vims
- JDV Jembrana Disease Virus
- EIAV equine infectious anemia virus
- CAEV visna-maedi and caprine arthritis encephalitis vims
- lentiviral particles making up the gene delivery vehicle are replication defective on their own (also referred to as "self-inactivating"). Lentivimses are able to infect both dividing and non-dividing cells by virtue of the entry mechanism through the intact host nuclear envelope (Naldini L et al., Curr. Opin. Biotechnol, 1998, 9: 457-463). Recombinant lentiviral vehicles/particles have been generated by multiply attenuating the HIV virulence genes, for example, the genes Env, Vif, Vpr, Vpu, Nef and Tat are deleted making the vector biologically safe.
- lentiviral vehicles for example, derived from HIV- 1 /HIV-2 can mediate the efficient delivery, integration and long-term expression of transgenes into non- dividing cells.
- Lentiviral particles may be generated by co-expressing the virus packaging elements and the vector genome itself in a producer cell such as human HEK293T cells. These elements are usually provided in three (in second generation lentiviral systems) or four separate plasmids (in third generation lentiviral systems).
- the producer cells arc co-transfected with plasmids that encode lentiviral components including the core (i.e., structural proteins) and enzymatic components of the virus, and the envelope protein(s) (referred to as the packaging systems), and a plasmid that encodes the genome including a foreign transgene, to be transferred to the target cell, the vehicle itself (also referred to as the transfer vector).
- the plasmids or vectors are included in a producer cell line.
- the plasmids/vectors are introduced via transfection, transduction or infection into the producer cell line. Methods for transfection, transduction or infection are well known by those of skill in the art.
- the packaging and transfer constructs can be introduced into producer cell lines by calcium phosphate transfection, lipofection or electroporation, generally together with a dominant selectable marker, such as neomyocin (neo), dihydrofolate reductase (DHFR), glutamine synthetase or adenosine deaminase (ADA), followed by selection in the presence of the appropriate drug and isolation of clones.
- a dominant selectable marker such as neomyocin (neo), dihydrofolate reductase (DHFR), glutamine synthetase or adenosine deaminase (ADA), followed by selection in the presence of the appropriate drug and isolation of
- the producer cell produces recombinant viral particles that contain the foreign gene, for example, the polynucleotides encoding the exogenous polynucleotide.
- the recombinant viral particles are recovered from the culture media and titrated by standard methods used by those of skill in the art.
- the recombinant lentiviral vehicles can be used to infect target cells, such source cells including any described herein.
- Cells that can be used to produce high-titer lentiviral particles may include, but are not limited to, HEK293T cells, 293G cells, STAR cells (Relander et al., Mol Ther.
- Additional elements provided in lentiviral particles may comprise retroviral LTR (long- terminal repeat) at either 5' or 3' terminus, a retroviral export element, optionally a lentiviral reverse response element (RRE), a promoter or active portion thereof, and a locus control region (LCR) or active portion thereof.
- retroviral LTR long- terminal repeat
- RRE lentiviral reverse response element
- Other elements include central polypurine tract (cPPT) sequence to improve transduction efficiency in non-dividing cells, Woodchuck Hepatitis Virus (WHP) Posttranscriptional Regulatory Element (WPRE) which enhances the expression of the transgene, and increases titer.
- WPRE Posttranscriptional Regulatory Element
- Lentivirus vectors used may be selected from, but are not limited to pLVX, pLenti, pLenti6, pLJMl, FUGW, pWPXL, pWPI, pLenti CMV puro DEST, pLJMl-EGFP, pULTRA, p!nducer2Q, pHIV-EGFP, pCW57.1 , pTRPE, pELPS, pRRL, and pLionll, Any known lentiviral vehicles may also be used (See, U.S. Pat. NOs.
- the exogenous polynucleotide is introduced into the cell under conditions for transient expression of the cell, such as by methods that result in episomal delivery of an exogenous polynucleotide.
- polynucleotides encoding the exogenous polynucleotide may be packaged into recombinant adeno-associated viral (rAAV) vectors.
- rAAV adeno-associated viral
- Such vectors or viral particles may be designed to utilize any of the known serotype capsids or combinations of serotype capsids.
- the serotype capsids may include capsids from any identified AAV serotypes and variants thereof, for example, AAV1, AAV2, AAV2G9, AAV3, AAV4, AAV4-4, AAV5, AAV6, AAV7, AAV8, AAV9, AAV 10, AAV 11, AAV 12 and AAVrhlO.
- the AAV serotype may be or have a sequence as described in United States Publication No. US20030138772; Pulichla et al. Molecular Therapy, 2011, 19(6): 1070-1078; U.S. Pat. Nos. : 6,156,303; 7,198,951; U.S. Patent Publication Nos. : US2015/0159173 and US2014/0359799: and International Patent Publication NOs.: WO1998/011244, W02005/033321 and WO2014/14422.
- AAV vectors include not only single stranded vectors but self-complementary AAV vectors (scAAVs).
- scAAV vectors contain DNA which anneals together to form double stranded vector genome. By skipping second strand synthesis, scAAVs allow for rapid expression in the cell.
- the rAAV vectors may be manufactured by standard methods in the art such as by triple transfection, in sf’9 insect cells or in suspension cell cultures of human cells such as HEK293 cells.
- non-viral based methods may be used.
- vectors comprising the polynucleotides may be transferred to cells by non-viral methods by physical methods such as needles, electroporation, sonoporation, hyrdoporation; chemical carriers such as inorganic particles (e.g., calcium phosphate, silica, gold) and/or chemical methods.
- synthetic or natural biodegradable agents may be used for delivery such as cationic lipids, lipid nano emulsions, nanoparticles, peptide -based vectors, or polymer-based vectors.
- the exogenous polynucleotide can be inserted into a specific genomic locus of the host cell.
- a number of gene editing methods can be used to insert an exogenous polynucleotide (e.g., a transgene) into a specific genomic locus of choice.
- the exogenous polynucleotide can be inserted into any suitable target genomic loci of the cell.
- the exogenous polynucleotide is introduced into the cell by targeted integration into a target loci.
- targeted integration can be achieved by gene editing using one or more nucleases and/or nickases and a donor template in a process involving homology-dependent or homology-independent recombination.
- Gene editing is a type of genetic engineering in which a nucleotide sequence may be inserted, deleted, modified, or replaced in the genome of a living organism.
- a number of gene editing methods can be used to insert an exogenous polynucleotide into the specific genomic locus of choice, including for example homology-directed repair (HOR), homology-mediated end-joining (HMEJ), homology-independent targeted integration (HITI), obligate ligation-gated recombination (ObliGaRe), or precise integration into target chromosome (PITCh).
- HOR homology-directed repair
- HMEJ homology-mediated end-joining
- HITI homology-independent targeted integration
- OFTaRe obligate ligation-gated recombination
- PITCh precise integration into target chromosome
- the gene editing technology can include systems involving nucleases, integrases, transposases, and/or recombinases.
- the gene editing technology mediates single-strand breaks (SSB). In some embodiments, the gene editing technology mediates doublestrand breaks (DSB), including in connection with non-homologous end-joining (NHEJ) or homology-directed repair (HDR). In some embodiments, the gene editing technology can include DNA-based editing or prime-editing. In some embodiments, the gene editing technology can include Programmable Addition via Site- specific Targeting Elements (PASTE). In some embodiments, the gene editing technology can include TnpB polypeptides. Many gene editing techniques generally utilize the innate mechanism for cells to repair double-strand breaks (DSBs) in DNA.
- DSBs single-strand breaks
- Eukaryotic cells repair DSBs by two primary repair pathways: non-homologous end-joining (NHEJ) and homology-directed repair (HDR).
- HDR typically occurs during late S phase or G2 phase, when a sister chromatid is available to serve as a repair template.
- NHEJ is more common and can occur during any phase of the cell cycle, but it is more error prone.
- NHEJ is generally used to produce insertion/deletion mutations (indels), which can produce targeted loss of function in a target gene by shifting the open reading frame (ORF) and producing alterations in the coding region or an associated regulatory region.
- HDR is a preferred pathway for producing targeted knock-ins, knockouts, or insertions of specific mutations in the presence of a repair template with homologous sequences.
- chemical modulation e.g., treating cells with inhibitors of key enzymes in the NHEJ pathway
- timed delivery of the gene editing system at S and G2 phases of the cell cycle e.g., cell cycle arrest at S and G2 phases
- introduction of repair templates with homology sequences e.g., chemical modulation (e.g., treating cells with inhibitors of key enzymes in the NHEJ pathway); timed delivery of the gene editing system at S and G2 phases of the cell cycle; cell cycle arrest at S and G2 phases; and introduction of repair templates with homology sequences.
- the methods provided herein may utilize HDR-mediated repair, NHEJ-mediated repair, or a combination thereof.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Engineering & Computer Science (AREA)
- Chemical & Material Sciences (AREA)
- Biomedical Technology (AREA)
- Immunology (AREA)
- Cell Biology (AREA)
- General Health & Medical Sciences (AREA)
- Hematology (AREA)
- Biotechnology (AREA)
- Molecular Biology (AREA)
- Medicinal Chemistry (AREA)
- Zoology (AREA)
- Organic Chemistry (AREA)
- Urology & Nephrology (AREA)
- Diabetes (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biochemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Genetics & Genomics (AREA)
- Microbiology (AREA)
- Pathology (AREA)
- Toxicology (AREA)
- Wood Science & Technology (AREA)
- Environmental Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- General Physics & Mathematics (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Gastroenterology & Hepatology (AREA)
- Analytical Chemistry (AREA)
- Physics & Mathematics (AREA)
- Food Science & Technology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Epidemiology (AREA)
- Endocrinology (AREA)
- Emergency Medicine (AREA)
- Developmental Biology & Embryology (AREA)
- Physiology (AREA)
Abstract
Among other things, provided are non-human animals comprising immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with type 1 diabetes (T1D) and islet cells derived from the human patient with type 1 diabetes (T1D), wherein the immune cells (e.g., PBMCs) and the islet cells are autologous. Methods for use of such non-human animals are also described.
Description
TYPE-1 DIABETES AUTOIMMUNE MOUSE
CROSS REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to and the benefit of United States Provisional Application No. 63/437,976, filed January 9, 2023, the contents of which are incorporated herein by reference in their entirety.
BACKGROUND
[0002] Type-1 diabetes (also referred to as T1D, juvenile diabetes or insulin-dependent diabetes) is a chronic condition that was reported to affect around 1 .6 million Americans in 2019.
With T1D, a patient’s pancreas makes little or no insulin, which is a hormone the body uses to allow sugar (e.g., glucose) to enter cells to produce energy. Different factors, such as genetics and some viruses, may cause T1D. Although T1D usually appears during childhood or adolescence, it can develop in adults.
SUMMARY OF THE INVENTION
[0003] Despite significant research in the field, T1D has no cure. Rather, treatment for T1D is largely directed toward managing the amount of sugar in the blood of a patient using insulin, and adjusting diet and lifestyle to prevent complications. Insulin has been the predominant treatment for T1D, and has been used to treat patients with T1D for more than 100 years. Insulin has transformed the once-fatal diagnosis into a chronic, medically manageable condition (sec Sims, E.K., Carr, A.L.J., Oram, R.A., DiMeglio, L.A. & Evans-Molina, C. 100 years of insulin: celebrating the past, present and future of diabetes therapy. Nat Med 27, 1154- 1164 (2021), which is incorporated herein by reference in its entirety). However, morbidity, mortality, and quality of life remain a problem, specifically for about 25% of patients with impaired awareness of hypoglycemia (IAH), who are at high risk of severe hypoglycemic events
and associated morbidities (see DiMeglio, L.A., Evans-Molina, C. & Oram, R.A. Type 1 diabetes. Lancet 391, 2449-2462 (2018); Livingstone, S J., et al. Estimated life expectancy in a Scottish cohort with type 1 diabetes, 2008-2010. JAMA 313, 37-44 (2015); Lin, Y.K., Eisher, S.J. & Pop-Busui, R. Hypoglycemia unawareness and autonomic dysfunction in diabetes: Lessons learned and roles of diabetes technologies. J Diabetes Investig 11, 1388-1402 (2020); Clarke, W.L., et al. Reduced awareness of hypoglycemia in adults with IDDM. A prospective study of hypoglycemic frequency and associated symptoms. Diabetes Care 18, 517-522 (1995), each of which is incorporated by reference herein in its entirety). Additionally, about half of all patients with long-standing T1D require assistance for severe hypoglycemic events, which increases the risk these patients face to a comparable level of patients with IAH (see Group, U.K.H.S. Risk of hypoglycaemia in types 1 and 2 diabetes: effects of treatment modalities and their duration. Diabetologia 50, 1140-1147 (2007), which is incorporated herein by reference in its entirety). The risk for severe hypoglycemic events increases with the duration of T1D (see Pedersen-Bjergaard, U., et al. Severe hypoglycaemia in 1076 adult patients with type 1 diabetes: influence of risk markers and selection. Diabetes Metab Res Rev 20, 479-486 (2004), which is incorporated by reference herein). Continuous glucose monitoring has shown the ability to catch peaks or drops in glucose levels better than conventional fingerstick blood glucose meter checks. Continuous glucose monitoring has improved glycemic control in adults, as well as adolescents and young adults with T1D (see Juvenile Diabetes Research Loundation Continuous Glucose Monitoring Study, G., et al. Continuous glucose monitoring and intensive treatment of type 1 diabetes. N Engl J Med 359, 1464-1476 (2008); Ruedy, K.J., Parkin, C.G., Riddlesworth, T.D., Graham, C. & Group, D.S. Continuous Glucose Monitoring in Older Adults With Type 1 and Type 2 Diabetes Using Multiple Daily Injections of Insulin: Results From the DIAMOND Trial. J Diabetes Sci Technol 11, 1138-1146 (2017); Laffel, L.M., et al. Effect of Continuous Glucose Monitoring on Glycemic Control in Adolescents and Young Adults With Type 1 Diabetes: A Randomized Clinical Trial. JAMA 323, 2388-2396 (2020), each of which is incorporated herein by reference in its entirety). Such wearable bioartificial devices provide glucose readings, trends, and alerts to the user in real time to inform diabetes treatment decisions (see Bekiari, E.,
et al. Artificial pancreas treatment for outpatients with type 1 diabetes: systematic review and meta-analysis. BMJ 361, kl310 (2018), which is incorporated herein by reference in its entirety). Nonetheless, compared to the physiological, immediate islet cell (e.g., beta islet cell) response, integrated closed-loop systems still face delays in subcutaneous glucose sensing and insulin delivery. In fact, patients experience an average of at least 30 minutes each day with hypoglycemia, e.g., with glucose less than 3.9 mmol/L (Boughton, C.K., et al. Hybrid closed- loop glucose control with faster insulin aspart compared with standard insulin aspart in adults with type 1 diabetes: A double-blind, multicentre, multinational, randomized, crossover study. Diabetes Obes Metab 23, 1389-1396 (2021), which is incorporated herein by reference in its entirety). Thus, while these systems are typically appreciated by patients, the systems do not provide curative therapy of T1D. The systems also do not completely remove the burden of glucose uncertainty and unremitting daily self-management. Negative themes associated with the systems include technical difficulties, intrusiveness of alarms, and equipment size (see Barnard, K.D., et al. Psychosocial aspects of closed- and open-loop insulin delivery: closing the loop in adults with Type 1 diabetes in the home setting. Diabet Med 32, 601-608 (2015), which is incorporated herein by reference in its entirety).
[0004] One possible treatment for T1D is transplantation of allogeneic pancreatic donor islets to a patient who has T1D. Transplantation of allogeneic pancreatic donor islets has successfully been performed in selected patients, who have difficult-to-control insulin-dependent diabetes and impaired awareness of hypoglycemia (IAH). Allogeneic islet transplantation replenishes the vanished beta cell population, and clinical trials in patients with IAH and history of severe hypoglycemic events showed that it provides glycemic control, restoration of hypoglycemia awareness, and protection from severe hypoglycemic events (see Brooks, A.M., et al. Attainment of metabolic goals in the integrated UK islet transplant program with locally isolated and transported preparations. Am J Transplant 13, 3236-3243 (2013); Hering, B.J., et al. Phase 3 Trial of Transplantation of Human Islets in Type 1 Diabetes Complicated by Severe Hypoglycemia. Diabetes Care 39, 1230-1240 (2016); Markmann, J.F., et al. Phase 3 trial of human islct-aftcr-kidncy transplantation in type 1 diabetes. Am J Transplant 21, 1477-1492
(2021), which is incorporated herein by reference in its entirety). Twenty-year follow-up data of allogeneic islet transplantation reported a mean duration of islet graft function of 4.4 to 5.9 years when the patient was taking immunosuppressive agents (see Lemos, J.R.N., et al. Survival After Islet Transplantation in Patients With Type 1 Diabetes: Twenty-Year Follow-Up. Diabetes Care 44, e67-e68 (2021); MarfiLGarza, B.A., et al. Pancreatic islet transplantation in type 1 diabetes: 20-year experience from a single-centre cohort in Canada. Lancet Diabetes Endocrinol 10, 519- 532 (2022), each of which is incorporated herein by reference in its entirety). These results support that islet transplantation may prove to be a successful T1D treatment.
[0005] However, transplantation of allogeneic pancreatic donor islets has required concomitant systemic immunosuppression to minimize the chances that a patient receiving allogeneic pancreatic donor islets will develop an immune response against the donor cells. Generally, the need for immunosuppression is life-long so that the islet grafts remain protected from allo- and autoimmunity. This presents a challenge to the successful implementation of islet cell transplantation, as immunosuppression can present serious additional risks, including beta cell toxicity, kidney toxicity, infections, and cancer (see Dai, C., et al. Tacrolimus- and sirolimus-induced human beta cell dysfunction is reversible and preventable. JCI Insight 5(2020); Kim, J.H., et al. Influence of cyclosporine A on glomerular growth and the effect of mizoribine and losartan on cyclosporine nephrotoxicity in young rats. Sci Rep 6, 22374 (2016); Naesens, M., Kuypers, D.R. & Sarwal, M. Calcineurin inhibitor nephrotoxicity. Clin J Am Soc Nephrol 4, 481-508 (2009); Higdon, L.E., Tan, J.C. & Maltzman, J.S. Infection, Rejection, and the Connection. Transplantation (2022); Rama, I. & Grinyo, J.M. Malignancy after renal transplantation: the role of immunosuppression. Nat Rev Nephrol 6, 511-519 (2010), each of which is incorporated herein by reference in its entirety). Additional side effects include (but are not limited to) increased risk for infections and cancer, digestive side effects (e.g., vomiting, nausea, and diarrhea), headaches, tremors, confusion, high blood pressure, high glucose levels, high cholesterol and triglyceride levels, and kidney damage. Because of the severity and frequency of the risks associated with immunosuppression, transplantation of allogeneic pancreatic donor islets has not experienced wide-spread adoption.
[00061 Additional significant challenges to allogeneic islet transplantation include an unsolved regulatory framework and early loss of transplanted islets from instant blood-mediated inflammatory reaction (see Witkowski, P., et al. The demise of islet allotransplantation in the United States: A call for an urgent regulatory update. Am J Transplant 21, 1365-1375 (2021); Naziruddin, B., et al. Evidence for instant blood-mediated inflammatory reaction in clinical autologous islet transplantation. Am J Transplant 14, 428-437 (2014); Kanak, M.A., et al. Inflammatory response in islet transplantation. Int J Endocrinol 2014, 451035 (2014), each of which is incorporated herein by reference in its entirety). As such, a need for a T1D cure and/or T1D treatments remains in the art. Further, a need for technologies useful for identifying and characterizing possible T1D cures and/or T1D treatments remains in the art.
[0007] The present disclosure provides technologies that will aid in the identification and/or characterization of islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) that can be transplanted in a subject suffering from T ID. In particular, the present disclosure provides technologies that enable transplantation of islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) without the need for concomitant immunosuppression.
[0008] In particular, the present disclosure provides non-human animals that can be used to identify and/or assess islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) for use in a subject (e.g., a human) for glycemic control. The present disclosure also describes non-human animals that can be used to identify and/or assess islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) survive, engraft, and/or ameliorate T1D. The present disclosure recognizes that additional therapeutic agents can be identified and/or assessed in non-human animals provided herein, and that such identification and/or assessment can be made separate from or concurrently with an identification and/or assessment of islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells).
[0009] The present disclosure further provides the insight that non-human animals described herein can be useful in assessing an autoimmune response of a subject. Specifically, the present disclosure provides that autologous immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a subject and islet cells derived from the subject can be transplanted to a
non-human animal, where the interaction of such cells with each other and with other agents can be assessed. Use of autologous immune cells (e.g., PBMCs) and autologous islet cells from a particular subject allows for a subject specific analysis of autoimmunity, disease pathology, and disease etiology to be evaluated. Such information can be useful in determining how a particular subject may respond to certain therapies and provide an individualized prognostic information.
[0010] Thus, among other things, the present disclosure provides a non-human animal comprising immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a subject and islet cells derived from cells of the subject. In some embodiments, a subject is a human patient. In some embodiments, a human patient has T1D. In some embodiments, immune cells (e.g., PBMCs) are autologous. In some embodiments, islet cells are autologous.
[0011] In one aspect, the present disclosure provides a non-human animal comprising immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with type 1 diabetes (T1D) and islet cells derived from cells of the human patient with T1D, wherein the immune cells (e.g., PBMCs) and the islet cells are autologous. In some embodiments, a non- human animal is a non-human mammal. In some embodiments, a non-human animal is a rodent. In some embodiments, a non-human animal is a mouse. In some embodiments, a non-human animal comprises one or more genetic modifications that are suitable for maintaining a xenograft. In some embodiments, one or more genetic modifications comprise reducing or eliminating expression of one or more genes. In some embodiments, one or more genetic modifications comprise increasing expression of one or more genes. In some embodiments, one or more genetic modifications comprise reducing or eliminating expression of one or more genes and increasing expression of one or more genes. In some embodiments, a mouse is a NOD mouse, optionally an NSG-SGM3 mouse. In some embodiments, a mouse is a SCID mouse, a NOD/SCID mouse, a NOD/SCID/ IL-2Rg(null) mouse, a NOD.Cg-/Wescid Z/2rgtmlwjl/SzJ mouse, a NOD.Cg- /rcld I12rgtmlwjl Tg(CMV-L3,CSF2,KITLG)lEav/MloySzJ (NSG-SGM3) mouse, a NSG-human IL3, NSG-SCF, NSG-GM-GSF mouse, a nude mouse, a ragl knockout mouse, or rag2 knockout mouse.
[00121 In some embodiments, a human patient with T1D is eligible for islet transplant therapy. In some embodiments, a human patient with T1D exhibits one or more of the following: (a) a clinical history compatible with T1D onset of disease at younger than 40 years old; (b) insulin dependence for at least 5 years; (c) a sum of age and years of insulin dependent diabetes duration of at least 28 years; (d) absent stimulated C-peptide (less than 0.3 ng/mL) 60 and 90 minutes post-mixed-meal tolerance test; (e) involvement of intensive diabetes management, wherein intensive diabetes management comprises: (i) self-monitoring of glucose values no less than a mean of three times each day averaged over each week; (ii) administration of three or more insulin injections each day or insulin pump therapy; and/or (iii) under the direction of an endocrinologist, diabetologist, or diabetes specialist with at least three clinical evaluations during the past 12 months; (f) at least one episode of severe hypoglycemia in the past 12 months, wherein an episode of severe hypoglycemia comprises an event with one or more of the following symptoms: (i) memory loss; (ii) confusion; (iii) uncontrollable behavior; (iv) irrational behavior; (v) unusual difficulty in awakening; (vi) suspected seizure; (vii) seizure;
(viii) loss of consciousness; (ix) visual symptoms, and hypoglycemia during the past 12 months; and/or (g) impaired awareness of hypoglycemia (IAH). In some embodiments, a human patient with T1D has had T1D for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 years, at least 8 years, at least 9 years, at least 10 years, at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 years, or at least 90 years. In some embodiments, a human patient with T1D has impaired awareness of hypoglycemia (IAH). In some embodiments, IAH comprises: a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more; a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more; marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more; or a composite of: a Clarke score of 4 or
more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile. In some embodiments, a human patient with T1D has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years. In some embodiments, a human patient with T1D has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2). In some embodiments, antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 IDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
[0013] In some embodiments, one or more of the immune cells comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells. In some embodiments, one or more of the immune cells are isolated from peripheral blood mononuclear cells (PBMCs). In some embodiments, one or more of the PBMCs comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells. In some embodiments, one or more of the immune cells produce autoantibodies against cells or antigens of the human patient with T1D. In some embodiments, one or more of the immune cells is a B cell. In some embodiments, one or more of the immune cells produce antibodies against one or more islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2. In some embodiments, one or more of the immune cells produce islet cell antibodies (ICA) and/or insulin autoantibodies (IAA). In some embodiments, one or more of the immune cells is an islet-reactive T cell. In some embodiments, one or more of the immune cells are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal. In some embodiments, a non-human animal comprises at least about 2.5xl05, at least about 5xl05, at least about IxlO6, at least about 2xl06, at least about 3xl06, at
least about 4xl06, at least about 5xl06, at least about 6xl06, at least about 7xl06, at least about 8xl06, at least about 9xl06, at least about IxlO7, at least about 1.5xl07, or at least about 2xl07 immune cells. In some embodiments, a non-human animal comprises at least about 2.5xl05, at least about 5xl05, at least about IxlO6, at least about 2xl06, at least about 3xl06, at least about 4xl06, at least about 5xl06, at least about 6xl06, at least about 7xl06, at least about 8xl06, at least about 9xl06, at least about IxlO7, at least about 1.5xl07, or at least about 2xl07 PBMCs.
[0014] In some embodiments, a non-human animal comprises reduced function of non- human islet cells relative to a comparable wild- type non-human animal. In some embodiments, a non-human animal comprises reduced activity of non-human islet cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal comprises a reduced number of non-human islet cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal does not comprise non-human islet cells. In some embodiments, a non-human animal comprises streptozotocin (STZ). In some embodiments, a non-human animal was previously administered STZ. In some embodiments, a non-human animal comprises or was previously administered alloxan. In some embodiments, a non-human animal comprises decreased expression of a PDX1 gene. In some embodiments, a non-human animal has reduced pancreatic function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced pancreatic activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has a reduced number of pancreatic cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal does not have a pancreas. In some embodiments, a non-human animal comprises splenocytes from an autoimmune and diabetic NOD/ShiLt mouse.
[0015] In some embodiments, a non-human animal has glucose levels greater than about 200 mg/dL, greater than about 300 mg/dL, greater than about 400 mg/dL, or greater than about
450 mg/dL. In some embodiments, a non-human animal has glucose levels greater than about
300 mg/dL. In some embodiments, a non-human animal has glucose levels greater than about
400 mg/dL. In some embodiments, a non-human animal has glucose levels greater than about
450 mg/dL.
[00161 In some embodiments, islet cells are introduced via intramuscular injection, intravenous injection, intraportal injection, ocular injection, injection into omentum, and/or injection into a kidney capsule.
[0017] In some embodiments, islet cells are or comprise human alpha, beta, and/or delta islet cells. In some embodiments, islet cells are or comprise about 5%, about 10%, about 15%, about 20%, about 25%, about 35%, or about 40% alpha cells. In some embodiments, islet cells are or comprise about 10%, about 15%, about 20%, about 25%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, or about 80% beta cells. In some embodiments, islet cells are or comprise about 1%, about 2%, about 3%, about 4%, about 5%, about 6%, about 7%, about 8%, about 9%, about 10%, about 15%, about 20%, or about 25% delta cells.
[0018] In some embodiments, islet cells are or comprise differentiated cells. In some embodiments, differentiated cells are derived from a stem cell. In some embodiments, differentiated cells are derived from a pluripotent stem cell. In some embodiments, differentiated cells are derived from an induced pluripotent stem cell (iPSC). In some embodiments, an iPSC is derived from a cell obtained from the human patient with T ID. In some embodiments, a cell obtained from the human patient with T1D is selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell. In some embodiments, a cell obtained from the human patient with T1D is a PBMC. In some embodiments, a stem cell is derived from a cell obtained from the human patient with T ID. In some embodiments, a stem cell is an induced pluripotent stem cell (iPSC) derived from a cell selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell. In some embodiments, a stem cell is an induced pluripotent stem cell (iPSC) derived from a PBMC. In some embodiments, differentiated cells are derived from a multipotent stem cell. In
some embodiments, a multipotent stem cell is derived from a cell obtained from the human patient with T1D. In some embodiments, differentiated cells are derived from a lineage- specific stem cell. In some embodiments, a lineage- specific stem cell is derived from a cell obtained from the human patient with T1D. In some embodiments, differentiated cells are derived from a definitive endoderm cell. In some embodiments, a definitive endoderm cell expresses FOXA2 and/or SOX 17. In some embodiments, differentiated cells are derived from a primitive gut tube cell. In some embodiments, a primitive gut tube cell expresses FOXA2 and/or SOX 17. In some embodiments, differentiated cells are derived from a pancreatic progenitor cell. In some embodiments, a pancreatic progenitor cell expresses PDX1 and/or NKX6-1. In some embodiments, differentiated cells are derived from an endocrine cell. In some embodiments, an endocrine cell expresses NKX6-1, NEUROG3, and/or NKX2-2.
[0019] In some embodiments, islet cells are or comprise engineered islet cells. In some embodiments, engineered islet cells have been modified to be hypoimmune islet cells. In some embodiments, hypoimmune islet cells comprise one or more modifications that (a) inactivate or disrupt one or more alleles of: (i) one or more major histocompatibility complex (MHC) class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or (ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules, and/or (b) increase expression of one or more tolerogenic factors, wherein the increased expression of (b) is relative to an islet cell that does not comprise the modifications. In some embodiments, one or more modifications that disrupt one or more MHC class I molecules and/or one or more MHC class II molecules reduce expression of the one or more MHC class I molecules and/or the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications. In some embodiments, hypoimmune islet cells are (i) HLA class I-deficient and/or HLA class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells comprise one or more safety switches. In some embodiments, a non-human animal comprises an agent that activates the one or more safety switches.
[00201 In some embodiments, islet cells produce insulin, c-peptide, glucagon, somatostatin, and/or pancreatic polypeptide.
[0021] In some embodiments, a non-human animal of the present disclosure comprises a first therapeutic agent. In some embodiments, a first therapeutic agent is or comprises islet cells. In some embodiments, a first therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid. In some embodiments, a non-human animal of the present disclosure further comprised a second therapeutic agent. In some embodiments, a second therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid.
[0022] In another aspect, the present disclosure provides methods of characterizing human islet cells for delivery to a patient, comprising taking a sample of the islet cells derived from cells of the human patient with T1D and administering the sample to a non-human animal of the present disclosure.
[0023] In another aspect, the present disclosure provides methods of making a non- human animal model of type 1 diabetes (T1D), the method comprising: (a) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to the non-human animal, (b) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous. In some embodiments, a non-human animal is a non-human mammal. In some embodiments, a non-human animal is a rodent. In some embodiments, a non-human animal is a mouse. In some embodiments, a non-human animal comprises one or more genetic modifications that are suitable for maintaining a xenograft. In some embodiments, one or more genetic modifications comprise reducing or eliminating expression of one or more genes. In some embodiments, one or more genetic modifications comprise increasing expression of one or more genes. In some embodiments, one or more genetic modifications comprise reducing or eliminating expression of one or more genes and increasing expression of one or more genes. In some embodiments, a mouse is a NOD mouse, optionally an NSG-SGM3 mouse. In some
embodiments, a mouse is a SCID mouse, a NOD/SCID mouse, a NOD/SCID/ IL-2Rg(null) mouse, a NOD.Cg-Prkdcscld Il2rgtml^ /SzJ mouse, a NOD.Cg-Pr£e?cscld H2rgtmlw-’1 Tg(CMV- L3,CSF2,KITLG)lEav/MloySzJ (NSG-SGM3) mouse, a NSG-human IL3, NSG-SCF, NSG- GM-GSF mouse, a nude mouse, a ragl knockout mouse, or rag2 knockout mouse.
[0024] In some embodiments, a human patient with T1D is eligible for islet transplant therapy. In some embodiments, a human patient exhibits one or more of the following: (a) a clinical history compatible with T1D onset of disease at younger than 40 years old; (b) insulin dependence for at least 5 years; (c) a sum of age and years of insulin dependent diabetes duration of at least 28 years; (d) absent stimulated C-peptide (less than 0.3 ng/mE) 60 and 90 minutes post-mixed-meal tolerance test; (e) involvement of intensive diabetes management, wherein intensive diabetes management comprises: (i) self-monitoring of glucose values no less than a mean of three times each day averaged over each week; (ii) administration of three or more insulin injections each day or insulin pump therapy; and/or (iii) under the direction of an endocrinologist, diabetologist, or diabetes specialist with at least three clinical evaluations during the past 12 months; (f) at least one episode of severe hypoglycemia in the past 12 months, wherein an episode of severe hypoglycemia comprises an event with one or more of the following symptoms: (i) memory loss; (ii) confusion; (iii) uncontrollable behavior; (iv) irrational behavior; (v) unusual difficulty in awakening; (vi) suspected seizure; (vii) seizure; (viii) loss of consciousness; (ix) visual symptoms, and hypoglycemia during the past 12 months; and/or (g) impaired awareness of hypoglycemia (IAH). In some embodiments, a human patient with T1D has had T ID for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 years, at least 8 years, at least 9 years, at least 10 years at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 years, or at least 90 years. In some embodiments, a human patient with T1D has impaired awareness of hypoglycemia (IAH). In some embodiments, IAH comprises: a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or
more; a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more; marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more; or a composite of: a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile. In some embodiments, a human patient with T1D has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years. In some embodiments, a human patient with T1D has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2). In some embodiments, antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
[0025] In some embodiments, wherein one or more of the immune cells comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells. In some embodiments, one or more of the immune cells are isolated from peripheral blood mononuclear cells (PBMCs). In some embodiments, one or more of the PBMCs comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells. In some embodiments, one or more of the immune cells produce autoantibodies against cells or antigens of the human patient with T1D. In some embodiments, one or more of the immune cells is a B cell. In some embodiments, one or more of the immune cells produce antibodies against one or more islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2. In some embodiments, one or more of the immune cells produce islet cell antibodies (ICA) and/or insulin autoantibodies (IAA). In
some embodiments, one or more of the immune cells is an islet-reactive T cell. In some embodiments, one or more of the immune cells are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal. In some embodiments, a nonhuman animal of the present disclosure comprises at least 2.5 xlO5, at least 5xl05, at least IxlO6, at least 2xl06, at least 3xl06, at least 4xl06, at least 5xl06, at least 6xl06, at least 7xl06, at least 8xl06, at least 9xl06, at least IxlO7, at least 1.5xl07, or at least 2xl07 immune cells. In some embodiments, a non-human animal comprises at least 2.5 xlO5, at least 5xl05, at least I xlO6, at least 2xl06, at least 3xl06, at least 4xl06, at least 5xl06, at least 6xl06, at least 7xl06, at least 8xl06, at least 9xl06, at least IxlO7, at least 1.5xl07, or at least 2xl07 PBMCs.
[0026] In some embodiments, islet cells are administered to the non-human animal via intramuscular injection, intravenous injection, intraportal injection, ocular injection, injection into omentum, and/or injection into a kidney capsule.
[0027] In some embodiments, islet cells are or comprise human alpha, beta, and/or delta islet cells. In some embodiments, islet cells are or comprise about 5%, about 10%, about 15%, about 20%, about 25%, about 35%, or about 40% alpha cells. In some embodiments, islet cells are or comprise about 10%, about 15%, about 20%, about 25%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, or about 80% beta cells. In some embodiments, islet cells are or comprise about 1%, about 2%, about 3%, about 4%, about 5%, about 6%, about 7%, about 8%, about 9%, about 10%, about 15%, about 20%, or about 25% delta cells.
[0028] In some embodiments, islet cells are or comprise differentiated cells. In some embodiments, differentiated cells are derived from a stem cell. In some embodiments, differentiated cells are derived from a pluripotent stem cell. In some embodiments, differentiated cells are derived from an induced pluripotent stem cell (iPSC). In some embodiments, an iPSC is derived from a cell obtained from the human patient with T ID. In some embodiments, a cell obtained from the human patient with T1D is selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell,
a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell. In some embodiments, a cell obtained from the human patient with T1D is a PBMC. In some embodiments, a stem cell is derived from a cell obtained from a human patient with T1D. In some embodiments, a stem cell is an induced pluripotent stem cell (iPSC) derived from a cell selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell. In some embodiments, a stem cell is an induced pluripotent stem cell (iPSC) derived from a PBMC. In some embodiments, differentiated cells are derived from a multipotent stem cell. In some embodiments, a multipotent stem cell is derived from a cell obtained from the human patient with T1D. In some embodiments, differentiated cells are derived from a lineage- specific stem cell. In some embodiments, a lineage- specific stem cell is derived from a cell obtained from the human patient with T1D. In some embodiments, differentiated cells are derived from a definitive endoderm cell. In some embodiments, a definitive endoderm cell expresses FOXA2 and/or SOX17. In some embodiments, differentiated cells are derived from a primitive gut tube cell. In some embodiments, a primitive gut tube cell expresses FOXA2 and/or SOX 17. In some embodiments, differentiated cells are derived from a pancreatic progenitor cell. In some embodiments, a pancreatic progenitor cell expresses PDX1 and/or NKX6-1. In some embodiments, differentiated cells are derived from an endocrine cell. In some embodiments, an endocrine cell expresses NKX6-1, NEUROG3, and/or NKX2-2.
[0029] In some embodiments, islet cells are or comprise engineered islet cells. In some embodiments, engineered islet cells have been modified to be hypoimmune islet cells. In some embodiments, hypoimmune islet cells comprise one or more modifications, wherein the modifications (a) inactivate or disrupt one or more alleles of: (i) one or more major histocompatibility complex (MHC) class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or (ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules, and/or (b) increase expression of one or more tolerogenic factors, wherein the
increased expression of (ii) is relative to an islet cell that does not comprise the modifications. In some embodiments, one or more modifications reduce expression of the one or more MHC class I molecules and/or one or more molecules that regulate expression of the one or more MHC class 1 molecules, and/or reduce expression of the one or more MHC class 11 molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications. In some embodiments, hypoimmune islet cells are (i) HLA class I-deficient and/or HLA class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells comprise one or more safety switches. In some embodiments, a non-human animal of the present disclosure comprises an agent that activates the one or more safety switches.
[0030] In some embodiments, islet cells produce insulin, glucagon, somatostatin, and/or pancreatic polypeptide.
[0031] In some embodiments, methods of the present disclosure further comprise a step of: (c) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal. In some embodiments, reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises administering an islet-cell destroying composition to the non-human animal. In some embodiments, an islet-cell destroying composition is or comprises STZ. In some embodiments, an islet-cell destroying composition is or comprises alloxan. In some embodiments, an islet-cell destroying composition is or comprises splenocytes from an autoimmune and diabetic NOD/ShiLt mouse. In some embodiments, reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises removing the non-human animal’s pancreas. In some embodiments, reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises decreasing expression of a PDX1 gene in the non-human animal. In some embodiments, a non-human animal has reduced endogenous islet cell function relative
to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced endogenous islet cell activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced levels of endogenous islet cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal does not comprise endogenous islet cells.
[0032] In some embodiments, a non-human animal has glucose levels greater than about 200mg/dL, greater than about 300mg/dL, greater than about 400mg/dL, or greater than about 450mg/dL. In some embodiments, a non-human animal has glucose levels greater than about 300mg/dL. In some embodiments, a non-human animal has glucose levels greater than about 400mg/dL. In some embodiments, a non-human animal has glucose levels greater than about 450mg/dL.
[0033] In some embodiments, a method of the present disclosure comprises, in sequential order: (a) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from the human patient with T1D to the non-human animal, (c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, and (b) administering the islet cells derived from cells of the human patient with T1D to the non-human animal.
[0034] In some embodiments, method of the present disclosure comprises, in sequential order: (c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (b) administering the islet cells derived from cells of the human patient with T1D to the non-human animal, and (a) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from the human patient with T1D to the non-human animal.
[00351 In some embodiments, method of the present disclosure comprises, in sequential order: (c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (a) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from the human patient with T1D to the non-human animal, and (b) administering islet cells derived from cells of the human patient with T1D to the non-human animal.
[0036] In some embodiments, an islet-cell destroying composition is administered to the non-human animal for 5 continuous days, 4 continuous days, 3 continuous days, 2 continuous days, or 1 continuous day. In some embodiments, an islet-cell destroying composition is administered to the non-human animal 5 days, 4 days, 3 days, 2 days, or 1 day before the immune cells are administered to the non-human patient. In some embodiments, no more than 1 day, 2 days, 3 days, 4 days, or 5 days elapse between step (a) and step (b).
[0037] In another aspect, the present disclosure provides methods comprising administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) and islet cells derived from cells of a human patient with type 1 diabetes (T1D) to a non-human animal, wherein the immune cells and the islet cells are autologous. In some embodiments, the non- human animal comprises immune cells from a human patient with T1D.
[0038] In another aspect, the present disclosure provides methods of using a non-human animal model of type 1 diabetes (T1D), the method comprising: (a) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to the non- human animal, (b) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous and (c) determining the survival and/or endocrine function of the islet cells. In some embodiments, islet cells are or comprise engineered islet cells. In some embodiments, engineered islet cells have been modified to be hypo immune islet cells. In some embodiments, hypoimmune islet cells comprise one or more modifications, wherein the modifications (a) inactivate or disrupt one or more alleles of: (i) one or more MHC class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or (ii) one or more MHC
class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules, and/or (b) increase expression of one or more tolerogenic factors, wherein the increased expression of (ii) is relative to an islet cell that does not comprise the modifications. In some embodiments, one or more modifications reduce expression of the one or more MHC class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or reduce expression of the one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications. In some embodiments, hypoimmune islet cells are (i) HLA class I-deficient and/or HLA class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells comprise one or more safety switches. In some embodiments, islet cells are a therapeutic agent. In some embodiments, step (c) comprises determining the survival and/or endocrine function of the islet cells after an autoimmune challenge. In some embodiments, a method of the present disclosure further comprises: (d) administering an agent to the non-human animal. In some embodiments, an agent activates the one or more safety switches. In some embodiments, step (c) further comprises evaluating effects of the one or more modifications on the hypoimmune islet cells. In some embodiments, an agent is a therapeutic agent. In some embodiments, a method of the present disclosure further comprises: (e) administering an immunosuppressant to the non- human animal.
[0039] In another aspect, the present disclosure provides methods of characterizing one or more therapeutic agents, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells arc a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non-
human animal, and (e) assessing the effect of the islet cells and/or the one or more therapeutic agents on one or more therapeutically relevant T1D measures.
[0040] In another aspect, the present disclosure provides methods of characterizing one or more therapeutic agents, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (c) administering islet cells derived from cells of a human patient with T1D to the non-human animal, wherein the islet cells are a first therapeutic agent, (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, (d) optionally administering a second therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the one or more therapeutic agent on one or more therapeutically relevant T1D measures.
[0041] In another aspect, the present disclosure provides methods of characterizing one or more therapeutic agents, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non- human animal, and (e) assessing the effect of the islet cells and/or the one or more therapeutic agent on one or more therapeutically relevant T1D measures.
[0042] In another aspect, the present disclosure provides methods of determining if one or more therapeutic agents are suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from
cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells derived from the human patient with T1D are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non- human animal, and (e) determining the suitability of the one or more therapeutic agents to make a T1D therapeutic by assessing the effect of the one or more therapeutic agents on one or more therapeutically relevant T1D measures.
[0043] In another aspect, the present disclosure provides methods of determining if one or more therapeutic agents are suitable for use in treating type 1 diabetes (T1D), the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells derived from the human patient with T1D are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non- human animal, and (e) determining the suitability of the one or more therapeutic agents to treat T1D by assessing the effect of the one or more therapeutic agents on one or more therapeutically relevant T1D measures.
[0044] In another aspect, the present disclosure provides methods of making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells derived from the human patient with T1D are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non-human animal, (e) determining the suitability of the first therapeutic agent, and optionally the second therapeutic agent, to make a
T1D therapeutic by assessing the effect of the first therapeutic agent, and optionally the second therapeutic agent, on one or more therapeutically relevant T1D measures, and (f) making the T1D therapeutic.
[0045] In another aspect, the present disclosure provides methods of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous, (d) administering a therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
[0046] In another aspect, the present disclosure provides methods of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (c) administering islet cells derived from cells of a human patient with T1D to the non-human animal, (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, (d) administering a therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
[0047] In another aspect, the present disclosure provides methods of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous, (d) administering a
therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
[0048] In another aspect, the present disclosure provides methods of determining if a therapeutic agent is suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, (d) administering the therapeutic agent to the non-human animal, and (e) determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures.
[0049] In another aspect, the present disclosure provides methods of determining if a therapeutic agent is suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous, (d) administering the therapeutic agent to the non-human animal, and (e) determining the suitability of the therapeutic agent to treat T1D by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures.
[0050] In another aspect, the present disclosure provides methods of making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human
animal, wherein the immune cells and the islet cells are autologous, (d) administering a therapeutic agent to the non-human animal, (e) determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures, and (f) making the T1D therapeutic.
[0051] In some embodiments, one or more therapeutically relevant T1D measures comprise the survival and/or endocrine function of the islet cells. In some embodiments, one or more therapeutically relevant T1D measures comprise levels of insulin, glucagon, somatostatin, pancreatic polypeptide, C-peptide, and/or glucose in the non-human animal.
[0052] In some embodiments, if a therapeutic agent is characterized as a suitable T1D therapeutic, the method further comprises a step of making a T1D therapeutic. In some embodiments, if a therapeutic agent is determined to be a suitable T1D therapeutic, the method further comprises a step of making a T1D therapeutic. In some embodiments, a suitable T1D therapeutic comprises one or more of the following effects of the therapeutic agent on one or more therapeutically relevant T1D measures: (a) C-peptide levels greater than at least 100 pmol/L, at least 200 pmol/L, at least 300 pmol/L, at least 400 pmol/L, at least 500 pmol/L, at least 1000 pmol/L, at least 1500 pmol/L, or at least 1750 pmol/L, optionally wherein C-peptide levels are determined using 60 and 90 minutes post-mixed-meal tolerance test; (b) glucose levels lower than about 200mg/dL, lower than about 300mg/dL, lower than about 400mg/dL, or lower than about 450mg/dL; and/or (c) islet cells survive for at least 5 days, at least 10 days, at least 15 days, at least 20 days, at least 25 days, or at least 30 days.
[0053] In some embodiments, reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal comprises administering an islet-cell destroying composition to the non-human animal. In some embodiments, an islet-cell destroying composition is or comprises STZ. In some embodiments, an islet-cell destroying composition is or comprises splenocytes from an autoimmune and diabetic NOD/ShiLt mouse. In some embodiments, reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal comprises removing the non-human animal’s pancreas. In some
embodiments, a non-human animal has reduced endogenous islet cell function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced endogenous islet cell activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced levels of endogenous islet cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal does not comprise endogenous islet cells.
[0054] In some embodiments, a second therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid. In some embodiments, a therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid.
[0055] In some embodiments, a step of determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of islet cells and/or one or more therapeutic agents on one or more therapeutically relevant T1D measures comprises the use of two or more assays to characterize the islet cells and/or the one or more therapeutic agents and/or to determine the suitability of islet cells and/or the one or more therapeutic agents for making a T1D therapeutic.
[0056] In another aspect, the present disclosure provides methods of treating a human patient, comprising administering a pharmaceutical composition comprising a T1D therapeutic made in a method of the present disclosure. In some embodiments, a human patient has been diagnosed as having type 1 diabetes (T1D). In some embodiments, a human patient is eligible for islet transplant therapy. In some embodiments, a human patient exhibits one or more of the following: (a) a clinical history compatible with T1D onset of disease at younger than 40 years old; (b) insulin dependence for at least 5 years; (c) a sum of age and years of insulin dependent diabetes duration of at least 28 years; (d) absent stimulated C-peptide (less than 0.3 ng/mL) 60 and 90 minutes post-mixed-meal tolerance test; (e) involvement of intensive diabetes
management, wherein intensive diabetes management comprises: (i) self-monitoring of glucose values no less than a mean of three times each day averaged over each week; (ii) administration of three or more insulin injections each day or insulin pump therapy; and/or (iii) under the direction of an endocrinologist, diabetologist, or diabetes specialist with at least three clinical evaluations during the past 12 months; (f) at least one episode of severe hypoglycemia in the past 12 months, wherein an episode of severe hypoglycemia comprises an event with one or more of the following symptoms: (i) memory loss; (ii) confusion; (iii) uncontrollable behavior; (iv) irrational behavior; (v) unusual difficulty in awakening; (vi) suspected seizure; (vii) seizure;
(viii) loss of consciousness; (ix) visual symptoms, and hypoglycemia during the past 12 months; and/or (g) impaired awareness of hypoglycemia (IAH). In some embodiments, a human patient has had T1D for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 year’s, at least 8 year’s, at least 9 years, at least 10 years, at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 years, or at least 90 years. In some embodiments, a human patient has impaired awareness of hypoglycemia (IAH). In some embodiments, IAH comprises: a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more; a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more; marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more; or a composite of: a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile.
[0057] 67.35. The method of any one of claims 67.28-67-34, wherein the human patient has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years.
[0058] In some embodiments, a human patient has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to
glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2). In some embodiments, antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
[0059] In some embodiments, a human patient has glucose levels greater than about 200 mg/dL, greater than about 300 mg/dL, greater than about 400 mg/dL, or greater than about 450 mg/dL. In some embodiments, a human patient has glucose levels greater than about 300 mg/dL. In some embodiments, a human patient has glucose levels greater than about 400 mg/dL. In some embodiments, a human patient has glucose levels greater than about 450 mg/dL.
[0060] In another aspect, the present disclosure provides methods of monitoring a human patient treated with a pharmaceutical composition comprising a T1D therapeutic made in any one of the methods of the present disclosure, comprising assessing the effect of the T1D therapeutic on glucose levels.
[0061] In some embodiments, a non-human animal of the present disclosure or a method of the present disclosure comprises hypoimmune islet cells derived from induced pluripotent stem cells (iPSCs), wherein one or more of the modifications present in the hypoimmune islet cells are also present in the iPSCs. In some embodiments, a non-human animal of the present disclosure or a method of the present disclosure comprises hypoimmune islet cells derived from induced pluripotent stem cells (iPSCs), wherein none of the modifications present in the hypoimmune islet cells are also present in the iPSCs. In some embodiments, one or more of the immune cells comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells. In some embodiments, one or more of the immune cells are isolated from peripheral blood mononuclear cells (PBMCs). In some embodiments, one
or more of the PBMCs comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
BRIEF DESCRIPTION OF THE DRAWING
[0062] The Drawing included herein, which is composed of the following Figures, is for illustration purposes only and not for limitation.
[0063] Figure 1 shows an exemplary illustration of the process of removing human peripheral blood mononuclear cells (PBMCs) from a patient with type 1 diabetes (T1D) and engrafting a mouse with the PBMCs to create type 1 diabetes mice (T1D mice). In parallel PBMCs from the T ID patient are reprogrammed into autologous induced pluripotent stem cells (auto iPSCs), and then autologous iPSC-derived p-islet cells are generated and transplanted into the T1D mice. Some autologous iPSC-derived islet cells were additionally engineered to be hypoimmune (HIP autologous iPSC-derived islet cells).
[0064] Figure 2 shows exemplary in vitro immunofluorescence staining of auto iPSC- derived and HIP iPSC-derived pseudo-islets (p-islets) for somatostatin, insulin, and glucagon (top two images) and for CD47, HLA class I, and DAPI (bottom two images).
[0065] Figure 3 shows exemplary graphs of HLA class I, HLA class II, and CD47 expression as assessed by flow cytometry in auto iPSC-derived p-islets (top row) and in HIP iPSC-derived p-islets (bottom row).
[0066] Figure 4 shows an exemplary graph of the composition of cell types (e.g., alpha cells, beta cells, delta cells, and other cells) in auto iPSC-derived and HIP iPSC-derived p-islets as assessed by flow cytometry.
[0067] Figure 5 shows an exemplary graph of insulin release by in vitro human auto iPSC-derived and HIP iPSC-derived p-islets as assessed by ELISA (mean ± SD, three independent replicates per group).
[00681 Figure 6 shows an exemplary illustration of iPSC-derived p-islets transplantations. In one scenario, auto iPSC-derived p-islets were transplanted into in autologous T1D mice. In a second scenario, HIP iPSC-derived p-islets were transplanted into autologous T1D mice, followed by subsequent transplantation of auto p-islets, via a second injection, on Day 15. In both scenarios, survival and glucose control of the p-islets in the mice were monitored.
[0069] Figure 7 shows exemplary in vivo bioluminescent images of three different batches of auto iPSC-derived p-islets transplanted into autologous T1D mice.
[0070] Figure 8 shows an exemplary graph of bioluminescent signal from auto iPSC- derived p-islets transplanted into autologous T1D mice.
[0071] Figure 9 shows an exemplary graph of fasting blood glucose levels and blood glucose levels after a 30 minute glucose challenge (mean ± SD, 15 animals).
[0072] Figure 10 shows an exemplary graph of results from an impedance killing assay with splenocytes and serum from T1D mice (that received auto p-islets) against auto iPSC- derived p-islet cells from the T1D mice (mean ± SD, 5 animals).
[0073] Figure 11 shows exemplary graphs of results from impedance killing assays using NK cells or macrophages as effector cells from T1D mice that received auto iPSC-derived p-islets against auto iPSC-derived p-islet cells from the T1D mice (mean ± SD, 3 independent experiments).
[0074] Figure 12 shows exemplary in vivo bioluminescent images of three different batches of HIP iPSC-derived p-islets transplanted into the right thigh muscle of autologous T1D mice. On Day 15, auto iPSC-derived p-islets were transplanted into the right thigh muscle. The BLI scale on the left side of the figure is from the HIP iPSC-derived p-islet imaging, the scale on the right from the auto p-islet imaging.
[0075] Figure 13 shows an exemplary graph of bioluminescent signal from HIP iPSC- derived p-islets transplanted into autologous T1D mice.
[0076] Figure 14 shows an exemplary graph of fasting blood glucose levels and blood glucose levels after a 30 minute glucose challenge (mean ± SD, 15 animals).
[0077] Figure 15 shows an exemplary graph of results from an impedance killing assay with splenocytes and serum from T1D mice (that received HIP p-islets) against HIP iPSC- derived p-islet cells from the T1D mice (mean ± SD, 5 animals).
[0078] Figure 16 shows exemplary graphs of results from impedance killing assays using NK cells or macrophages as effector cells from T1D mice that received HIP iPSC-derived p-islets against HIP iPSC-derived p-islet cells from the T1D mice (mean ± SD, 3 independent experiments).
[0079] Figure 17 shows an exemplary graph of c-peptide levels in mice 30 days after they received auto iPSC-derived p-islets or HIP iPSC-derived p-islets (mean ± SD, 15 animals each).
CERTAIN DEFINITIONS
[0080] The scope of the present invention is defined by the claims appended hereto and is not limited by certain embodiments described herein. Those skilled in the art, reading the present specification, will be aware of various modifications that may be equivalent to such described embodiments, or otherwise within the scope of the claims. In general, terms used herein are in accordance with their understood meaning in the art, unless clearly indicated otherwise. Explicit definitions of certain terms are provided below; meanings of these and other terms in particular instances throughout this specification will be clear to those skilled in the art from context. Additional definitions for the following and other terms are set forth throughout the specification. Patent and non-patent literature references cited within this specification, or relevant portions thereof, are incorporated herein by reference in their entireties.
[0081] In order that the present invention may be more readily understood, certain terms are first defined below. Additional definitions for the following terms and other terms are set forth throughout the specification.
[0082] Use of ordinal terms such as “first,” “second,” “third,” etc., in the claims to modify a claim element does not by itself connote any priority, precedence, or order of one claim element over another or the temporal order in which acts of a method are performed, but are used merely as labels to distinguish one claim element having a certain name from another element having a same name (but for use of the ordinal term) to distinguish the claim elements.
[0083] The articles “a” and “an,” as used herein, should be understood to include the plural referents unless clearly indicated to the contrary. Claims or descriptions that include “or” between one or more members of a group are considered satisfied if one, more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process unless indicated to the contrary or otherwise evident from the context. In some embodiments, exactly one member of a group is present in, employed in, or otherwise relevant to a given product or process. In some embodiments, more than one, or all group members are present in, employed in, or otherwise relevant to a given product or process. It is to be understood that the invention encompasses all variations, combinations, and permutations in which one or more limitations, elements, clauses, descriptive terms, etc., from one or more of the listed claims is introduced into another claim dependent on the same base claim (or, as relevant, any other claim) unless otherwise indicated or unless it would be evident to one of ordinary skill in the ail that a contradiction or inconsistency would arise. Where elements are presented as lists (e.g., in Markush group or similar format), it is to be understood that each subgroup of the elements is also disclosed, and any element(s) can be removed from the group. It should be understood that, in general, where embodiments or aspects are referred to as “comprising” particular elements, features, etc., certain embodiments or aspects “consist of,” or “consist essentially of,” such elements, features, etc. For purposes of simplicity, those embodiments have not in every case been specifically set forth in so many words herein. It should also be understood that any embodiment or aspect can be explicitly excluded from the claims, regardless of whether the specific exclusion is recited in the specification.
[0084] Administration: as used herein, includes the administration of a composition (e.g., an agent, e.g., cells, e.g., PBMCs or islet cells) to a subject or system (e.g., to a cell, organ,
tissue, organism, or relevant component or set of components thereof). The skilled artisan will appreciate that route of administration may vary depending, for example, on the subject or system to which the composition is being administered, the nature of the composition, the purpose of the administration, etc. For example, in certain embodiments, administration to an animal subject (e.g., to a human or a rodent) may be bronchial (including by bronchial instillation), buccal, enteral, interdermal, intra-arterial, intradermal, intragastric, intramedullary, intramuscular, intranasal, intraperitoneal, intrathecal, intravenous, intraventricular, mucosal, nasal, oral, rectal, subcutaneous, sublingual, topical, tracheal (including by intratracheal instillation), transdermal, vaginal and/or vitreal. In some embodiments, administration to an animal subject (e.g., to a human or a rodent) may be performed via transplantation. For instance, in some embodiments provided herein, islet cells (e.g., autologous islet cells) may be administered via transplantation. In some embodiments, islet cells (e.g., autologous islet cells) may be administered by intramuscular injection, intravenous injection, intraportal injection, ocular injection, injection into omentum, and/or injection into a kidney capsule. In some embodiments, administration to an animal subject (e.g., to a human or a rodent) may be intraarterial or intravenous. For instance, in some embodiments provided herein, peripheral blood mononuclear cells (PBMCs) (e.g., autologous PBMCs) may be administered intravenously.
[0085] Agent: as used herein, the term “agent,” may refer to a compound, molecule, or entity of any chemical class (including, for example, a small molecule, polypeptide, nucleic acid, saccharide, lipid, metal, or a combination or complex thereof), system (e.g., cell, tissue, or organism), or a mixture.
[0086] Allogeneic: as used herein, refers to an agent (e.g., compound, complex, cell, tissue, etc.) that is found in, originated from, and/or was obtained or derived (e.g., as progeny of an agent obtained from) from a different subject of the same species. In some embodiments, an allogeneic agent is sufficiently different genetically such that it is capable of interacting antigenically. In some embodiments, an allogeneic agent may be immunologically incompatible with a subject to which it is administered.
[0087] Approximately and About: as applied to one or more values of interest, includes to a value that is similar to a stated reference value. In certain embodiments, the term “approximately” or “about” refers to a range of values that fall within ± 10% (greater than or less than) of the stated reference value unless otherwise stated or otherwise evident from the context (except where such number would exceed 100% of a possible value). In some embodiments, the term “approximately” or “about” refers to a range of values that would be rounded to the stated value based on the appropriate number of significant digits.
[0088] Autoantibodies: as used herein, the term “autoantibodies” is used to refer to antibodies produced by a subject in response to an agent from or derived from the subject. In some embodiments, autoantibodies associated with T1D include islet cell autoantibodies (ICA), autoantibodies against insulin (IAA), autoantibodies against glutamic acid decarboxylase (GAA or GAD), autoantibodies against protein tyrosine phosphatase, or a combination thereof.
[0089] Autoantigen: as used herein, the term “autoantigen” is used to refer to an antigen produced by a subject, a subject’s cell or tissue, or a cell or tissue derived from a subject that are recognized by the immune system of that subject. In some embodiments, an autoantigen is one whose recognition by the subject’s immune system is associated with an autoimmune disease, disorder or condition (e.g., T1D). In general, an autoantigen may be or include any chemical entity such as, for example, a small molecule, a nucleic acid, a polypeptide, a carbohydrate, a lipid, etc. In some embodiments, autoantigens associated with T1D include (but are not limited to) cytoplasmic proteins in an islet cell (e.g., a beta islet cell), glutamic acid decarboxylase (GAD-65), insulin, Zinc transporter 8 (ZnT8), and tyrosine phosphatase (IA2 or ICA512).
[0090] Autoimmunity: as used herein, the term “autoimmunity” is used to refer to a collection of immune responses of a subject against an antigen produced by the subject, the subject’s cell or tissue, or a cell or tissue derived from the subject.
[0091] Autologous: as used herein, refers to an agent (e.g., molecule, cell, or tissue) that is from or derives from the same individual to which the agent will be administered. Said
differently, an autologous agent is obtained from a subject that is both the donor and recipient of the agent.
[0092] Biologically active: as used herein, refers to a characteristic of any agent that has activity in a biological system, in vitro or in vivo (e.g., in a subject). For instance, an agent that, when present in a subject, has a biological effect within that subject is considered to be biologically active. In particular embodiments, where an agent is biologically active, a portion of that agent that shares at least one biological activity of the agent is typically referred to as a “biologically active” portion.
[0093] Biological Sample: as used herein, the term “biological sample” typically refers to a sample obtained or derived from a biological source (e.g., a tissue or organism or cell culture) of interest, as described herein. In some embodiments, a source of interest comprises an organism, such as an animal or human. In some embodiments, a biological sample is or comprises biological tissue or fluid. In some embodiments, a biological sample may be or comprise bone marrow; blood; blood cells; ascites; tissue or fine needle biopsy samples; cellcontaining body fluids; free floating nucleic acids; sputum; saliva; urine; cerebrospinal fluid, peritoneal fluid; pleural fluid; feces; lymph; gynecological fluids; skin swabs; vaginal swabs; oral swabs; nasal swabs; washings or lavages such as a ductal lavages or broncheoalveolar lavages; aspirates; scrapings; bone marrow specimens; tissue biopsy specimens; surgical specimens; feces, other body fluids, secretions, and/or excretions; and/or cells therefrom, etc. In some embodiments, a biological sample is or comprises cells obtained from an individual. In some embodiments, obtained cells are or include cells from an individual from whom the sample is obtained. In some embodiments, a sample is a “primary sample” obtained directly from a source of interest by any appropriate means. For example, in some embodiments, a primary biological sample is obtained by methods selected from the group consisting of biopsy (e.g., fine needle aspiration or tissue biopsy), surgery, collection of body fluid (e.g., blood, lymph, feces etc.), etc. In some embodiments, as will be clear from context, the term “sample” refers to a preparation that is obtained by processing (e.g., by removing one or more components of and/or by adding one or more agents to) a primary sample. For example, filtering using a semi-
permeable membrane. Such a “processed sample” may comprise, for example nucleic acids or proteins extracted from a sample or obtained by subjecting a primary sample to techniques such as amplification or reverse transcription of mRNA, isolation and/or purification of certain components, etc.
[0094] Comparable: as used herein, refers to two or more agents, situations, sets of conditions, etc. that may not be identical to one another but that are sufficiently similar to permit comparison there between so that conclusions may reasonably be drawn based on differences or similarities observed. Persons of ordinary skill in the art will understand, in context, what degree of identity is required in any given circumstance for two or more such agents, situations, sets of conditions, etc. to be considered comparable.
[0095] Control: as used herein, refers to the art-understood meaning of a “control” being a standard against which results are compared. Typically, controls are used to augment integrity in experiments by isolating variables in order to make a conclusion about such variables. In some embodiments, a control is a reaction or assay that is performed simultaneously with a test reaction or assay to provide a comparator. A “control” also includes a “control animal.” A “control animal” may have a modification as described herein, a modification that is different as described herein, or no modification (i.e., a wild-type animal). In one experiment, a "test" parameter (e.g., a variable being tested) is applied. In a second experiment, the “control,” the variable being tested is not applied. In some embodiments, a control is a historical control (i.e., of a test or assay performed previously, or an amount or result that is previously known). In some embodiments, a control is or comprises a printed or otherwise saved record. A control may be a positive control or a negative control. In some embodiments, a control may be comparable.
[0096] Detectable: as used herein, a “detectable” expression level, means a level that is detectable by standard techniques known to a skilled artisan, and include for example, differential display, RT (reverse transcriptasej-coupled polymerase chain reaction (PCR), Northern Blot, and/or RNase protection analyses as well as immunoaffinity-based methods for protein detection, such as flow cytometry, ELISA, or western blot. The degree of expression
levels need only be large enough to be visualized or measured via standard characterization techniques.
[0097] Determining, measuring, evaluating, assessing, assaying and analyzing: are used interchangeably herein to refer to any form of measurement, and include determining if an element is present or not. These terms include both quantitative and/or qualitative determinations. Assaying may be relative or absolute. “Assaying for the presence of” can be determining the amount of something present and/or determining whether or not it is present or absent.
[0098] Engineered: as used herein refers, in general, to the aspect of having been manipulated by the hand of man. Furthermore, as will be appreciated by persons of skill in the art, a variety of methodologies are available through which “engineering” as described herein may be achieved. For example, in some embodiments, “engineering” may involve selection or design (e.g., of nucleic acid sequences, polypeptide sequences, cells, tissues, and/or organisms) through use of computer systems programmed to perform analysis or comparison, or otherwise to analyze, recommend, and/or select sequences, alterations, etc.). Alternatively, or additionally, in some embodiments, “engineering” may involve use of in vitro chemical synthesis methodologies and/or recombinant nucleic acid technologies, mutation, transformation, transfection, etc., and/or any of a variety of controlled mating methodologies. As will be appreciated by those skilled in the art, a variety of established such techniques (e.g., for recombinant DNA, oligonucleotide synthesis, tissue culture, transformation (e.g., electroporation, lipofection, etc.), transplantation) are well known in the ail and described in various references that are cited and/or discussed throughout the present specification. See e.g., Sambrook et al., Molecular Cloning: A Laboratory Manual 2nd ed., Cold Spring Harbor Laboratory Press, Cold Spring Harbor, N.Y., 1989 and Principles of Gene Manipulation: An Introduction to Genetic Manipulation, 5th Ed., ed. By Old, R.W. and S.B. Primrose, Blackwell Science, Inc., 1994, each of which is incorporated herein by reference in its entirety.
[0099] Expression: The term “expression” with reference to a gene or “gene expression” refers to the conversion of the information, contained in a gene, into a gene product.
A gene product can be the direct transcriptional product of a gene (e.g., mRNA, tRNA, rRNA, antisense RNA, ribozyme, structural RNA or any other type of RNA) or can be a protein produced by translation of an mRNA. Gene products also include RNAs which are modified, by processes such as capping, polyadenylation, methylation, and editing, and proteins modified by, for example, methylation, acetylation, phosphorylation, ubiquitination, ADP-ribosylation, myristoylation, and glycosylation. Hence, reference to expression or gene expression includes protein (or polypeptide) expression or expression of a transcribable product of or a gene such as mRNA. The protein expression may include intracellular expression or surface expression of a protein. Typically, expression of a gene product, such as mRNA or protein, is at a level that is detectable in the cell.
[0100] Function: as used herein, “function” refers to a property and/or activity of a form or fragment of an agent (e.g., a biological agent, e.g., a cell, e.g., an islet cell or a PBMC).
[0101] Gene: as used herein, a “gene,” includes a DNA region encoding a gene product, as well as all DNA regions which regulate the production of the gene product, whether or not such regulatory sequences are adjacent to coding and/or transcribed sequences. Accordingly, a gene includes, but is not necessarily limited to, promoter sequences, terminators, translational regulatory sequences such as ribosome binding sites and internal ribosome entry sites, enhancers, silencers, insulators, boundary elements, replication origins, matrix attachment sites and locus control regions. The sequence of a gene is typically present at a fixed chromosomal position or locus on a chromosome in the cell.
[0102] Hypoimmunogenic: The term “hypoimmunogenic” refers to a cell that is less prone to immune rejection by a subject to which such cells are transplanted. For example, relative to a comparable cell that does not contain certain modifications (e.g., a wild-type cell or a cell without one or more modifications intended to address immunogenicity (e.g., a modification to an HLA-I, HLA-II and/or tolerogenic factor gene)), such a hypoimmunogenic cell may be about 2.5%, 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, 97.5%, 99% or more less prone to immune rejection by a subject into which such cells are transplanted. Typically, the hypoimmunogenic cells are allogenic to the subject and a hypoimmunogenic cell
evades immune rejection in an MHC-mismatched allogeneic recipient. In some embodiments, a hypoimmunogenic cell is protected from T cell-mediated adaptive immune rejection and/or innate immune cell rejection.
[0103] Hypoimmunogenicity: as used herein, “hypoimmunogenicity” of a cell can be determined by evaluating the immunogenicity of the cell such as the cell’s ability to elicit adaptive and innate immune responses.
[0104] Knock out: as used herein, “knock out” includes deleting all or a portion of the target polynucleotide sequence in a way that interferes with the function of the target polynucleotide sequence. For example, a knock out can be achieved by altering a target polynucleotide sequence by inducing an indel in the target polynucleotide sequence in a functional domain of the target polynucleotide sequence (e.g., a DNA binding domain).
CRISPR/Cas systems can be used to knock out a target polynucleotide sequence or a portion thereof based upon the details described herein. Knocking out a target polynucleotide sequence or a portion thereof can be useful for a variety of applications. For example, knocking out a target polynucleotide sequence in a cell can be performed in vitro for research purposes. For ex vivo purposes, knocking out a target polynucleotide sequence in a cell can be useful for treating or preventing a disorder associated with expression of the target polynucleotide sequence (e.g., by knocking out a mutant allele in a cell ex vivo and introducing those cells comprising the knocked out mutant allele into a subject).
[0105] Knock in: as used herein, “knock in” refers to a process that adds a genetic function to a host cell. A knock in can cause increased levels of the knocked in gene product, e.g., an RNA or encoded protein. As will be appreciated by those in the art, this can be accomplished in several ways, including adding one or more additional copies of the gene to the host cell or altering a regulatory component of the endogenous gene increasing expression of the protein is made. This may be accomplished by modifying the promoter, adding a different promoter, adding an enhancer, or modifying other gene expression sequences.
[01061 Modification: as used herein, the term “modification” refers to any change or alteration in a cell that impacts gene expression in the cell. In some embodiments, the modification is a genetic modification that directly changes the gene or regulatory elements thereof encoding a protein product in a cell, such as by gene editing, mutagenesis or by genetic engineering of an exogenous polynucleotide or transgene. In some embodiments, an alteration or modification described herein results in reduced expression of a target or selected polynucleotide sequence. In some embodiments, an alteration or modification described herein results in reduced expression of a target or selected polypeptide sequence. In some embodiments, an alteration or modification described herein results in increased expression of a target or selected polynucleotide sequence. In some embodiments, an alteration or modification described herein results in increased expression of a target or selected polypeptide sequence.
[0107] Non-human animal: as used herein, refers to any vertebrate organism that is not a human. In some embodiments, a non-human animal is a cyclostome, a bony fish, a cartilaginous fish (e.g., a shark or a ray), an amphibian, a reptile, a mammal, and a bird. In some embodiments, a non-human animal is a mammal. In some embodiments, a non-human mammal is a primate, a goat, a sheep, a pig, a dog, a cow, or a rodent. In some embodiments, a non- human animal is a rodent such as a rat or a mouse.
[0108] Patient: as used herein, the term “patient” refers to any organism to which a provided composition is or may be administered, e.g., for experimental, diagnostic, prophylactic, cosmetic, and/or therapeutic purposes. Typical patients include animals (e.g., mammals such as mice, rats, rabbits, non-human primates, and/or humans). In some embodiments, a patient is a human. In some embodiments, a patient is suffering from or susceptible to one or more disorders or conditions (e.g., T1D). In some embodiments, a patient displays one or more symptoms of a disorder or condition (e.g., T1D). In some embodiments, a patient has been diagnosed with one or more disorders or conditions (e.g., T1D). In some embodiments, the patient is receiving or has received certain therapy to diagnose and/or to treat a disease, disorder, or condition (e.g., T1D).
[01091 Polypeptide: As used herein refers to a polymeric chain of amino acids. In some embodiments, a polypeptide is a protein. In some embodiments, a polypeptide has an amino acid sequence that occurs in nature. In some embodiments, a polypeptide has an amino acid sequence that does not occur in nature. In some embodiments, a polypeptide has an amino acid sequence that is engineered in that it is designed and/or produced through action of the hand of man. In some embodiments, a polypeptide may comprise or consist of natural amino acids, non-natural amino acids, or both. In some embodiments, a polypeptide may comprise or consist of only natural amino acids or only non-natural amino acids. In some embodiments, a polypeptide may comprise D-amino acids, L-amino acids, or both. In some embodiments, a polypeptide may comprise only D-amino acids. In some embodiments, a polypeptide may comprise only L-amino acids. In some embodiments, a polypeptide may include one or more pendant groups or other modifications, e.g., modifying or attached to one or more amino acid side chains, at the polypeptide’s N-terminus, at the polypeptide’s C-terminus, or any combination thereof. In some embodiments, such pendant groups or modifications may be selected from the group consisting of acetylation, amidation, lipidation, methylation, pegylation, etc., including combinations thereof. In some embodiments, a polypeptide may be cyclic, and/or may comprise a cyclic portion. In some embodiments, a polypeptide is not cyclic and/or does not comprise any cyclic portion. In some embodiments, a polypeptide is linear. In some embodiments, a polypeptide may be or comprise a stapled polypeptide. In some embodiments, the term “polypeptide” may be appended to a name of a reference polypeptide, activity, or structure; in such instances it is used herein to refer to polypeptides that share the relevant activity or structure and thus can be considered to be members of the same class or family of polypeptides. For each such class, the present specification provides and/or those skilled in the art will be aware of exemplary polypeptides within the class whose amino acid sequences and/or functions are known; in some embodiments, such exemplary polypeptides are reference polypeptides for the polypeptide class or family. In some embodiments, a member of a polypeptide class or family shows significant sequence homology or identity with, shares a common sequence motif (e.g., a characteristic sequence element) with, and/or shares a common activity (in some embodiments at a comparable
level or within a designated range) with a reference polypeptide of the class; in some embodiments with all polypeptides within the class). For example, in some embodiments, a member polypeptide shows an overall degree of sequence homology or identity with a reference polypeptide that is at least about 30-40%, and is often greater than about 50%, 60%, 70%, 80%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99% or more and/or includes at least one region (e.g., a conserved region that may in some embodiments be or comprise a characteristic sequence element) that shows very high sequence identity, often greater than 90% or even 95%, 96%, 97%, 98%, or 99%. Such a conserved region usually encompasses at least 3-4 and often up to 20 or more amino acids; in some embodiments, a conserved region encompasses at least one stretch of at least 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15 or more contiguous amino acids. In some embodiments, a relevant polypeptide may comprise or consist of a fragment of a parent polypeptide. In some embodiments, a useful polypeptide as may comprise or consist of a plurality of fragments, each of which is found in the same parent polypeptide in a different spatial arrangement relative to one another than is found in the polypeptide of interest (e.g., fragments that are directly linked in the parent may be spatially separated in the polypeptide of interest or vice versa, and/or fragments may be present in a different order in the polypeptide of interest than in the parent), so that the polypeptide of interest is a derivative of its parent polypeptide.
[0110] Prognostic and predictive information: as used herein, the terms “prognostic information” and “predictive information” are used to refer to any information that may be used to indicate any aspect of the course of a disease or condition (e.g., T1D) either in the absence or presence of treatment. Such information may include, but is not limited to, the average life expectancy of a patient, the likelihood that a patient will survive for a given amount of time (e.g., 6 months, 1 year, 5 years, etc.), the likelihood that a patient will be cured of a disease, the likelihood that a patient’ s disease will respond to a particular therapy (wherein response may be defined in any of a variety of ways). Prognostic and predictive information are included within the broad category of diagnostic information.
[0111] Subject: As used herein, the term “subject” refers an organism, typically a mammal (e.g., a human). In some embodiments, a subject is suffering from a relevant disease, disorder or condition (e.g., T1D). In some embodiments, a subject is a patient.
[0112] Substantially: as used herein, refers to the qualitative condition of exhibiting total or near-total extent or degree of a characteristic or property of interest. One of ordinary skill in the biological arts will understand that biological and chemical phenomena rarely, if ever, go to completion and/or proceed to completeness or achieve or avoid an absolute result. The term “substantially” is therefore used herein to capture the potential lack of completeness inherent in many biological and chemical phenomena.
[0113] Therapeutic agent: as used herein, the phrase “therapeutic agent” refers to an agent that, when administered to a subject, has a therapeutic effect and/or elicits a desired biological and/or pharmacological effect. In some embodiments, a therapeutic agent is any substance that can be used to alleviate, ameliorate, relieve, inhibit, prevent, delay onset of, reduce severity of, and/or reduce incidence of one or more symptoms or features of a disease, disorder, and/or condition (e.g., T1D).
[0114] Tolerogenic factor: as used herein, the term “tolerogenic factor” includes immunosuppressive factors or immune-regulatory factors that modulate or affect the ability of a cell to be recognized by the immune system of a host or recipient subject upon administration, transplantation, or engraftment. Typically, a tolerogenic factor is a factor that induces immunological tolerance to an engineered primary cell so that the engineered primary cell is not targeted, such as rejected, by the host immune system of a recipient. Hence, a tolerogenic factor may be a hypoimmunity factor. Examples of tolerogenic factors include immune cell inhibitory receptors (e.g. CD47), proteins that engage immune cell inhibitory receptors, checkpoint inhibitors and other molecules that reduce innate or adaptive immune recognition.
[0115] Treatment: as used herein, the term “treatment” (also “treat” or “treating”) refers to administration of a therapy that partially or completely alleviates, ameliorates, relives, inhibits, delays onset of, reduces severity of, and/or reduces incidence of one or more symptoms, features,
and/or causes of a particular disease, disorder, and/or condition (e.g., T1D). In some embodiments, treatment may be prophylactic; in some embodiments, treatment may be therapeutic.
[0116] Wild-type: as used herein, refers to an entity having a structure and/or activity as found in nature in a “normal” (as contrasted with mutant, diseased, altered, engineered, etc.) state or context.
[0117] All literature and similar material cited in this application, including, but not limited to, patents, patent applications, articles, books, treatises, and web pages, regardless of the format of such literature and similar materials, are expressly incorporated by reference in their entirety. In the event that one or more of the incorporated literature and similar materials differs from or contradicts this application, including but not limited to defined terms, term usage, described techniques, or the like, this application controls. The section headings used herein are for organizational purposes only and are not to be construed as limiting the patient matter described in any way.
DETAILED DESCRIPTION OF CERTAIN EMBODIMENTS
[0118] As discussed above, there remains a need in the art for a T1D cure and/or T1D treatments. Part of the reason such a need remains is the lack of technologies available for sufficiently identifying and/or assessing the potential T1D cures and treatment. While T1D models exist, such models are not useful for robust characterization of T1D and the underlying mechanisms that lead to T1D. Thus, a need for technologies useful for identifying and characterizing possible T1D cures and/or T1D treatments also remains in the ail. The present disclosure addresses the need for technologies that can be utilized in understanding an individual patient’s T1D etiology and/or pathogenesis.
[0119] For example, the present disclosure provides technologies that will aid in the identification and/or characterization of islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) that can be transplanted in a subject suffering from T1D. In particular, the present disclosure provides technologies that enable transplantation of islet cells
(e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) without the need for concomitant immunosuppression.
[0120] Further, the present disclosure provides non-human animals that can be used to identify and/or assess islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) for use in a subject (e.g., a human) for glycemic control. The present disclosure also describes non-human animals that can be used to identify and/or assess islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells) survive, engraft, and/or ameliorate T1D. The present disclosure recognizes that additional therapeutic agents can be identified and/or assessed in non-human animals provided herein, and that such identification and/or assessment can be made separate from or concurrently with an identification and/or assessment of islet cells (e.g., hypoimmune islet cells, e.g., hypoimmune human islet cells).
[0121] Additionally, the present disclosure further provides the insight that non-human animals described herein can be useful in assessing an autoimmune response of a subject. Specifically, the present disclosure provides that autologous peripheral blood mononuclear cells (PBMCs) from a subject and islet cells derived from the subject can be transplanted to a non- human animal, where the interaction of such cells with each other and with other agents can be assessed. Use of autologous PBMCs and autologous islet cells from a particular subject allows for a subject specific analysis of autoimmunity, disease pathology, and disease etiology to be evaluated. Such information can be useful in determining how a particular subject may respond to certain therapies and provide an individualized prognostic information.
[0122] Thus, among other things, the present disclosure provides a non-human animal comprising peripheral blood mononuclear cells (PBMCs) from a subject and islet cells derived from cells of the subject. In some embodiments, a subject is a human patient. In some embodiments, a human patient has T1D. In some embodiments, PBMCs are autologous. In some embodiments, islet cells are autologous.
[0123] Related methods are also provided, including methods for making non-human animals described herein. Methods for using non-human animals described herein are also
provided. Among uses for provided non-human animals are methods of characterizing islet cells for delivery to a patient and methods for assessing and characterizing T1D therapeutic agents.
I. Type-1 Diabetes (T1D)
[0124] Among other things, the present disclosure provides technologies that allow for the study of autoimmune diseases. In particular, the present disclosure provides the recognition that the interaction between immune system cells (e.g., PBMCs or cells derived from PBMCs) from a subject (e.g., a mammal, e.g., a human) and other cells or tissues from the same subject can be assessed in a different non-human animal (e.g., a mammal, a rodent, e.g., a mouse or rat).
[0125] Specifically, the present disclosure provides technologies that allow for the study of Type- 1 diabetes (T1D). Non-human animals (e.g., a mammal, e.g., a rodent, e.g., a mouse or rat) are provided that include a non-human animal comprising peripheral blood mononuclear cells (PBMCs) from a subject (e.g., a mammal, e.g., a human) and islet cells derived from cells of the subject.
A. Overview of Type-1 Diabetes
[0126] In Type-1 diabetes (T1D, also referred to Type-1 diabetes mellitus, T1DM, or insulin-dependent diabetes mellitus) is a chronic disease characterized by hyperglycemia secondary to an insulin deficiency. T1D affects approximately 20 million people worldwide, and accounts for 5-10% of all cases of all diabetes disorders. Although T1D is most common in childhood and adolescence, T ID can present at any age.
[0127] T1D is caused via an autoimmune response of insulin-producing -cells in the pancreatic islets of Langerhans. Because a T1D subject produces little to no insulin, the subject cannot properly process glucose and glucose build up in the subject’s blood. The increased level of glucose has metabolic consequences, including (but not limited to) weight loss, polyuria, polydipsia, and ketoacidosis.
[0128] Insulitis is the term for the infiltration of the pancreatic islets by immune cells, which are predominantly T and B lymphocytes, macrophages and dendritic cells. Of these
immune cells, T cells can play an important role in initiating the autoimmune process and it has been shown that transfer of a single T cell clone can cause T1D in animal models of T1D. A normal immune system eliminates islet-reactive T cells or control their activity; however, the immune systems of subjects with T1D lack this ability.
[0129] In addition to T-cells, autoimmunity to [3-islet cells can be mediated by autoantibodies produced by a subject, which are directed to proteins present on [3-islet cells. Autoantibodies are frequently directed to glutamic acid decarboxylase and/or insulin, but other autoantibody targets are known, as shown in Table 1 below.
(see Chetan, MR, et al., “What is Type 1 diabetes?,” Medicine, Vol. 47, Iss. 1, pp. 5-9 (Jan. 2019), which is incorporated by reference in its entirety).
[0130] T1D can be diagnosed in a subject by measuring circulating autoantibodies directed to, e.g., insulin, GAD65, ZnT8, ICA512, and/or IA-2. Adults tend to have higher levels of glutamic acid decarboxylase (GAD) autoantibodies, and children tend to have higher levels of insulin autoantibodies. Not all subjects with T1D are antibody-positive, however. Thus, while a positive measure of autoantibodies can be helpful for diagnosing T1D, a negative measure is not sufficient to exclude a diagnosis of T1D. Measuring autoantibodies can be helpful because autoantibodies develop prior to the onset of T1D, and therefore can be useful to predict a diagnosis of T1D. Assays for T1D autoantibodies are known in the art.
B. Mechanism of T1D
[0131] Various factors have been implicated in the pathogenesis of T1D, as outlined below.
i. Genetic factors
[0132] Typically, T1D presents in subjects without a family history, but genetics is understood to play a role in the development of T1D. Only 10-15% of the T1D patients have a first- or second-degree relative with the disease. However, the lifetime risk for developing T1D is increased for relatives of T1D patients, as about 6% of children, 5% of siblings and 50% of monozygotic twins of T1D patients develop T1D compared to 0.4% prevalence of the general population.
[0133] More than 50 T1D genetic risk loci have been identified by genome-wide association studies and meta-analyses. The main genes predisposing to T1D include major histocompatibility complex (MHC) genes (also called human leucocyte antigen (HLA) genes), which are located on human chromosome 6. HLA polymorphic alleles are understood to be responsible for 40-50% of the genetic risk of T1D development. Insulin gene (Ins-VNTR, IDDM 2) polymorphisms on chromosome 11 and cytotoxic T lymphocyte-associated antigen-4 gene (CTLA-4) on chromosome 2 are also implicated in a significant number of T1D cases. Other different genetic loci have been found to contribute in a lesser degree to the genetic susceptibility for T1D, either alone or in combination with other autoimmune diseases.
MHC
[0134] MHC is a group of closely related genetic loci encoding molecules expressed on the surface of immune or other cells. MHC molecules can limit the specificity of T lymphocytes to recognize antigens. HLA genes are distinguished into two classes. MHC class I (MHC-I) molecules are typically associated with antigens recognized by the receptor of cytotoxic T lymphocytes (CD8+), which eventually destroy the antigenic target and are expressed by most cells. MHC class II (MHC -II) molecules are essential for the recognition of antigens by T helper lymphocytes (CD4+), which initiate the immune response and promote cellular co-operation and are expressed mostly by immune cells.
[0135] In T1D, most associations with specific MHC alleles or haplotypes are found in MHC-II genes, which is similar to other autoimmune diseases. The ability of MHC-II molecules
to present antigens is dependent on their alpha- and beta-chain amino acid composition. Substitutions at one or two sites can impact the binding capacity of the relevant autoantigens. For example, more than 90% of T1D patients have either HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotypes as compared to 40% of the healthy individuals. In addition, about 30% of the patients have both haplotypes (DR3/4 heterozygotes). On the other hand, the presence of some DR4 alleles, such as DRB 1 *0403 and DPB 1 *0402, reduces the risk of T1 D development even in the presence of DQB 1*0302 high-risk allele. HLA allele DQB 1*0602 also seems to provide protection against T1D development. This allele is present in approximately 20% of the general US population but only in 1% of the children who have developed T1D.
[0136] The present disclosure thus provides that a subject as described herein can have a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype. In some embodiments, a subject has a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) and HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
[0137] The present disclosure also provides that a PBMC as described herein can have a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype. In some embodiments, a PBMC has a HLA-DR3, DQBl*0201 (also referred as DR3-DQ2) and HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
Insulin and Insulin- VNTR
[0138] Polymorphisms in the region of the insulin gene promoter are estimated to account for about 10% of the genetic predisposition for T1D. The insulin gene promoter is located on the short arm of chromosome 11 and is close to the gene encoding for proinsulin. This polymorphic site consists of a variable numbers of tandem repeats (VNTRs). Two VNTR classes are associated with T1D risk: the small class (26-63 repeats, type I) or the big class (140- 243 repeats, type III). Class II VNTRs have between 63-140 repeats and is not understood to impact T1D susceptibility. Type III is present in approximately 30% of the general population and provides protection from T ID. Without wishing to be bound to any particular theory, it is
believed that the presence of a big class allele facilitates transcription and epithelial expression of insulin. Accordingly, the autoantigen presentation of insulin in the fetal thymus and is protective by promoting the negative selection and deletion of T lymphocytes reactive to insulin that contribute to the autoimmune destruction of 0 cells. Type 1 is associated with a susceptibility to T1D.
[0139] The present disclosure provides that a subject as described herein can comprise a class I VNTR at an insulin gene promoter. The present disclosure also provides that a PBMC as described herein can comprise a class I VNTR at an insulin gene promoter.
CTLA-4
[0140] The cytotoxic T lymphocyte antigen-4 (CTLA-4) gene is located on the long arm of chromosome 2 (2q33) and has also been associated with T1D. It has a suppressive role in T- cell activation, and specifically, a role of transmitting negative signal and inducing anergy in activated T-cell. Identification of an antigen by a naive T-cell is achieved through a triple complex consisting of an antigen, an HLA molecule of the antigen presenting cell (APC) and a T-cell antigen receptor. However, this complex recognition is not capable itself of stimulating a naive T-cell for proliferation and differentiation into an active T-cell; rather, costimulatory molecules are necessary. Such molecules include the B7.1 (CD80) and B7.2 (CD86) glycoproteins on APCs and a CD28 receptor, a member of the immunoglobulin superfamily, on T-cells. In the absence of costimulatory molecules, a T-cell is not stimulated and switches to the anergy state. An active T-cell further expresses the CTLA-4 receptor, which resembles the sequence of CD28, but has 20-fold greater affinity for B7 glycoproteins. A CTLA-4 receptor, when linked to one of the B7 (CD80/CD86) molecules of an APC, transmits negative messages to a T-cell. This limits the production of IL-2 and the proliferation of stimulated cells. As a result, excessive stimulation and proliferation of T-cell are avoided.
[0141] Polymorphisms of CTLA-4 have been associated with T1D and with other autoimmune diseases. These polymorphisms tend to lead to decreased intracellular expression of
CTLA-4. Consequently, inhibition of excessive stimulation and proliferation of T-cell is not inhibited resulting in uncontrolled progression of immune response and autoimmune imbalance.
[0142] The present disclosure provides that a subject as described herein can comprise a polymorphism within a CTLA-4 gene that results in a decreased level of CTLA-4 expression as compared to level of CTLA-4 expression observed with a comparable subject that does not have the polymorphism. The present disclosure provides that a cell as described herein can comprise a polymorphism within a CTLA-4 gene that results in a decreased level of CTLA-4 expression as compared to level of CTLA-4 expression observed with a comparable cell that does not have the polymorphism.
PTPN22
[0143] The protein tyrosine phosphatase non-receptor type 22 (PTPN22) gene encodes the lymphoid tyrosyl phosphatase known as LYP. LYP is a potent inhibitor of the activation of naive T-cells, preventing spontaneous T-cell activation by dephosphorylating and inactivating a kinase, named CSK. A missense mutation (C1858T (Arg620Trp) polymorphism) in the PTPN22 gene was found to be associated with various autoimmune diseases, including T1D. This mutation decreases the binding affinity of LYP to CSK, leading to uncontrolled T-cell activation and autoimmune reaction.
[0144] The present disclosure provides that a subject as described herein can comprise a C1858T (Arg620Trp) polymorphism in a PTPN22 gene. The present disclosure also provides that a PBMC as described herein can comprise a C1858T (Arg620Trp) polymorphism in a PTPN22 gene.
AIRE
[0145] Presentation of autoantigens in both the thymus and periphery to negatively select and destroy T-cells that are highly reactive (a process referred to as central and peripheral immune tolerance, respectively) is an important regulatory mechanism of autoimmunity. The autoimmune regulator (AIRE) protein is mainly expressed in the epithelial and dendritic cells of the thymus marrow. It is a transcription factor that is understood to control expression of many
specific autopeptides. Patients with mutations in the AIRE gene, located on the long arm of the chromosome 21 (21q22.3), exhibit autoimmune polyendocrine syndrome 1 (APS 1), also known as APECED syndrome, which presents, among other things, with T1D. The presence of T1D in patients with AIRE mutations suggests that the expression and presentation of relevant autoantigens (e.g., insulin, GAD, and/or IA-2) in the thymus are under the surveillance of this transcription factor.
[0146] The present disclosure provides that a subject as described herein can comprise one or more mutations in an AIRE gene. The present disclosure also provides that a PBMC as described herein can comprise one or more mutations in an AIRE gene. In some embodiments, expression of AIRE in a subject or a PBMC with one or more mutations in an AIRE gene may be decreased compared to a subject or PBMC without the one or more mutations.
FoxP3
[0147] The gene of the transcription factor FoxP3 is located on chromosome X and encodes for a protein known as scurfin. The congenital dysfunction of FoxP3 is manifested by an autoimmune phenotype. In humans, congenital dysfunction of FoxP3 appears as an autoimmune polyendocrine disorder associated with chromosome X (IPEX syndrome) and is manifested with a milder phenotype comprising, e.g., T1D, allergies, enteropathies and eczema. FoxP3 transcription factor is the hallmark of T regulatory lymphocytes (Tregs). The level of expression of Foxp3 in Tregs is positively associated with the a-chain expression of the IL-2 receptor (CD25) and inversely associated with the expression of the a-chain of the receptor for IL-7 (CD 127). Tregs suppress the proliferation and secretory activity of CD4+ and CD8+ T cells, as well as the cytolytic activity of the latter.
[0148] The present disclosure provides that a subject as described herein can comprise one or more mutations in a FoxP3 gene. The present disclosure also provides that a PBMC as described herein can comprise one or more mutations in a FoxP3 gene. In some embodiments, expression of scurfin in a subject or a PBMC with one or more mutations in a FoxP3 gene may be decreased compared to a subject or PBMC without the one or more mutations.
STAT3
[0149] The two major subpopulations of CD4+ T lymphocytes, Thl and Th2, derive from the same precursor T lymphocyte under the influence of specific cytokines within the lymph nodes. IL-12 produced by activated macrophages and dendritic cells induces Thl differentiation by activation of the transcription factors STAT1, STAT3 and STAT4. Mutations in the STAT3 transcription factor have been identified as monogenic causes of autoimmune diseases. De novo activating mutations of STAT3 are associated with an early onset spectrum of autoimmune diseases, such as T1D and autoimmune thyroid dysfunction.
[0150] The present disclosure provides that a subject as described herein can comprise one or more mutations in a STAT3 gene. The present disclosure also provides that a PBMC as described herein can comprise one or more mutations in a STAT3 gene. In some embodiments, expression of STAT3 in a subject or a PBMC with one or more mutations in a STAT3 gene may be decreased compared to a subject or PBMC without the one or more mutations.
IFIH1
[0151] The interferon-induced helicase C domain 1 (IFIH1) gene is located on Chr2q24.2. This gene encodes the melanoma differentiation-associated protein 5 (MDA5) protein, a cytoplasmic sensor of viral double- stranded RNA, that activates a cascade of anti-viral immune responses. Rare variants of IFIH1 through a lost or reduced expression of the protein are protective against T1D, whereas common IFIH1 single nuclear polymorphisms (SNPs) are associated with the disease.
[0152] The present disclosure provides that a subject as described herein can comprise one or more mutations in an IFIH1 gene. The present disclosure also provides that a PBMC as described herein can comprise one or more mutations in an IFIH1 gene. In some embodiments, expression of MDA5 in a subject or a PBMC with one or more mutations in an IFIH1 gene may be decreased compared to a subject or PBMC without the one or more mutations.
HIP14
[0153] The Huntingtin-interacting protein 14 gene is located on chromosome 12 and encodes for a palmitoyl transferase, which is involved in the metabolism and trafficking of proteins met with neurons. Knockdown of the genes in mice revealed increased apoptosis for pancreatic 0 cells. On the other hand, increased expression of HIP 14 was protected against apoptosis and this was in association with reduced nuclear-factor kappa B (NFKB) activity.
[0154] The present disclosure provides that a subject as described herein can comprise one or more mutations in an HIP14 gene. The present disclosure also provides that a PBMC as described herein can comprise one or more mutations in an HIP14 gene. In some embodiments, expression of HIP14 in a subject or a PBMC with one or more mutations in an HIP14 gene may be decreased compared to a subject or PBMC without the one or more mutations.
ERBB3
[0155] The Erb-B2 receptor tyrosine kinase 3 (ERBB3) gene is located on chromosome 12 and encodes for an enzyme interacting with various receptors, but lacks intrinsic activity. ERBB3 seems to play an important role in cytokine-induced apoptosis of 0 cells. Several SNPs (especially the rs2292239) have been associated with the presence of T ID, as well as the glycemic control of patients with the disease.
[0156] The present disclosure provides that a subject as described herein can comprise one or more mutations in an ERBB3 gene. The present disclosure also provides that a PBMC as described herein can comprise one or more mutations in an ERBB3 gene. In some embodiments, expression of ERBB3 in a subject or a PBMC with one or more mutations in an ERBB3 gene may be decreased compared to a subject or PBMC without the one or more mutations. ii. Environmental factors
[0157] Environmental factors also play an important role in the pathogenesis of T1D. Evidence for the role of environmental factors in T1D derives from the study of monozygotic twins, where occurrence of the disease in both siblings varies around 50% and never reaches
100%. Relevant environmental factors involved include viruses (e.g., rubella, coxsackievirus B, or enteroviruses), toxins, and nutrients (e.g., cow’s milk, cereals).
Viruses
[0158] Viruses can trigger T1D pathogenesis. Children exposed during fetal life to rubella have an increased incidence of T1D (and other autoimmune disorders, such as autoimmune thyroiditis). Furthermore, RNA or proteins from such viruses have been detected in the pancreas and other neighboring tissues from patients with T1D. Enteroviruses can play an important role in the early phase of the development of T1D through the activation of innate immunity. There is also a link between T1D and the interferon-induced helicase 1 (IFIH1) gene, which encodes for the MDA5, as discussed above. Upon viral infection, MDA5 acts as a cytoplasmic sensor especially for coxsackieviruses B and stimulates the production of molecular mediators of an innate immune response.
[0159] Another mechanism by which the autoimmune response can be induced by viruses is that of molecular mimicry. One hypothesis of molecular mimicry suggests that the immune response is directed against autoantigens that resemble viral antigens, which results in cellular destruction. In T1D, the best studied paradigm of molecular mimicry is the P2-C protein of the Coxsackie B4 virus. There is significant similarity between the amino acid sequences of Coxsackie B4 P2-C protein and glutamic acid decarboxylase (GAD65) enzyme found in 0 cells of the endocrine pancreas. Experimental studies have shown that clones from T-cell lines that recognized both GAD65 and P2C peptides, reacted with both using different restriction elements and without cross-reaction needed. Of course, T-cell autoactivation after Coxsackie virus infection which leads to inflammation, tissue damage and release of islet autoantigens can further compound issues and increase T1D risk.
[0160] The present disclosure provides that a subject as described herein has been exposed to coxsackieviruses strains B4, Bl, A4, A2, and/or A16. The present disclosure provides that a subject as described herein has been exposed to an enterovirus. The present disclosure also provides that a PBMC as described herein can be obtained from a subject that has
been exposed to coxsackieviruses strains B4, Bl, A4, A2, and/or A 16. The present disclosure also provides that a PBMC as described herein can be obtained from a subject that has been exposed an enterovirus.
Diet and gut microbiota
[0161] The importance of diet in the development of T1D is still being explored. In several studies, associations of early introduction in the infant’s diet of cow’s milk with an increased risk for the disease have been reported, supporting that infant’s exposure to insulin contained in the milk is triggering the autoimmune response. In addition, earlier experimental studies mainly have demonstrated that a particular part of the albumin contained in cow’s milk, known as ABBOS (17 peptide, positions 152-167), can function as a self-reactive epitope since it resembles the p69 protein found in the surface of pancreatic P cells. Early integration of cereals into the diet, nitrate exposure from water intake, inadequate intake of omega-3 fatty acids and vitamin D deficiency have also been implicated. On the other hand, meta-analysis of observational studies and vitamin D supplementation studies do not seem to confirm its protective role.
[0162] Further data indicates that gut microbiota, the microflora of trillions of microorganisms living in the gastrointestinal tract, may be involved in the pathogenesis of T1D. Patients with T1D exhibit differences in their gut microbiota compared to healthy controls, specifically a reduced Firmicutes:Bacteroidetes ratio. Whether the changes in the composition of the gut microbiota derive from environmental factors or T1D itself is still being investigated. The birthing process may play a role in the microbiota and its role in T1D, as suggested by the increased risk of T1D in children born by cesarean section. Lack of contact with the vaginal microbiome of the mother and subsequent differences in their gut microbiota due to abnormal colonization may be a pathogenic mechanism and indicate the possible protective role of normal labor and vaginal delivery.
[0163] The present disclosure provides that a subject as described herein may produce antibodies ABBOS. The present disclosure also provides that a subject as described herein may
have a microbiome with a reduced Firmicutes:Bacteroidetes ratio as compared to a comparable subject that has not been diagnosed with an autoimmune disorder (e.g., T1D). The present disclosure also provides that a subject as described herein may have been birthed via cesarean section. iii. Immunologic factors
Immune tolerance
[0164] The immune systems of mammals are exposed to large numbers of antigens, including autoantigens. Part of the purpose of the immune system is to distinguish foreign antigens from autoantignes. Immune system repertoires are extremely large and diverse. One consequence of such a robust and diverse immune system is that antigenic receptors that are self- reactive can develop, including against, e.g., pancreatic cells. Potentially dangerous immune cells during growth of T-cells in the thymus and B-cells in the bone marrow are typically negatively selected and eliminated (referred to as central tolerance). Self-reactive lymphocytes (e.g., T-cells and B-cells), which escape the mechanisms of central tolerance and end up in the periphery, naturally enter into processes that either neutralize or suppress them (referred to as peripheral tolerance).
[0165] Disorders of impacting central and peripheral tolerance can result in various autoimmune conditions. For example, in some situations, disruption in the function of the thymus can lead to the development of auto-immune diseases, including T1D. As discussed above, for instance, mutations in the AIRE gene can disturb central tolerance in thymus and lead to the development of T1D. Other non-genetic factors can have a similar effect. One hypothesis is that viral infections that affect human thymus can lead to autoimmune disorders, including T1D. Thymic epithelial cells and thymocytes have been proven to be targets of infection with coxsackievirus B4 virus and other enteroviruses in experimental studies. When thymic epithelial cells and thymocytes have been infected with such viruses, several abnormalities have been observed, including the maturation and differentiation processes of T cells.
[01661 Additionally, it has been demonstrated that the subpopulation of T regulatory lymphocytes (Tregs, formerly known as suppressive lymphocytes) plays an important role in the immune response network, especially for peripheral tolerance. It has been found that some patients with T1D present quantitative and qualitative deficits in Tregs, which may explain the increased (without limits) immune response, which eventually leads to the autoimmune response.
[0167] The present disclosure provides that a subject as described herein may have a disrupted thymus function. In some embodiments, a subject as described herein comprises one or more mutations in an AIRE gene and has had prior exposure to a coxsackieviruses strains B4, Bl, A4, A2, and/or A16 or an enterovirus.
[0168] The present disclosure also provides that a subject as described herein may have a disrupted Treg function.
Cellular immunity
[0169] The destruction of the P cells of the endocrine pancreas in T1D may occur via apoptosis, which includes a cascade of cysteine-asparaginase (caspase) activations. Indeed, animal studies and in vitro experiments support apoptosis as a major cell death mechanisms for beta cells. However, necrosis and necroptosis may also play a role in beta cell death with T1D. One hypothesis provides that inflammatory reaction with high levels of the proinflammatory cytokines IL-1, TNF-a (tumor necrosis factor-a) and INF-y (interferon-y) is induced by the autoreactive T-cells within the islet microenvironment. These cytokines activate a caspase cascade. Another hypothesis suggests that apoptosis is induced directly by contact of autoreactive T-cells with beta cells via the perforating system or Fas/Fas ligand interaction. Under this hypothesis, dysfunctional cytokine secretion is involved. Prior to the onset of T1D, a chronic atrophic inflammation within the islets of Langerhans is observed histologically, with the participation of T-cells, macrophages, B-ells, and dendritic cells. This condition usually evolves over months or years, when patients are asymptomatic and euglycemic. Symptomatic hyperglycemia then occurs after a long latency period, which reflects that a large number of functioning beta cells are destroyed before T1D clinically manifests.
[01701 The present disclosure provides that a subject as described herein may have inflammation (e.g., chronic inflammation, e.g., chronic atrophic inflammation) within the islets of Langerhans.
[0171] The present disclosure also provides that a subject as described herein may have an increased level of proinflammatory cytokines IL-1, TNF-a, and INF-y when compared to a comparable healthy subject.
Humoral immunity
[0172] Humoral immunity may also play a role in T1D by producing autoantibodies that may precede the onset of clinical manifestation of T1D, e.g., for years. As discussed above, autoantibodies detected in T1D patients include against GAD65, tyrosyl phosphatase (IA-2), insulin (IAA) and zinc transporter (ZnT8). Studies in mice with early presentation of these autoantibodies suggest that proinsulin is a potential primary target. The importance of insulin as an autoantigen has confirmed by the fact that deletion of its gene in mice affects disease progression, while its administration during the pre-diabetic phase prevents or delays clinical manifestation of T1D. Another autoantigen implicated in T1D is the GAD enzyme, which is present in beta cells, the central nervous system, and the testes. IA-2 is another autoantigen involved in T1D. Autoantibodies to IA-2 usually appear later in T1D progression than autoantibodies to insulin and GAD. Zinc transporter 8 (ZnT8) has also been identified as autoantigen for T1D. In children monitored from birth to T1D development, it was observed that ZnT8 autoantibodies appear later than autoantibodies to insulin and typically disappear very early after the clinical manifestation of the disease.
[0173] One of the best markers of progression to clinical T1D is the simultaneous expression of two or three autoantibodies from the above. The National Institutes of Health funded a study referred to as TEDDY (the environmental determinants of diabetes in the young). This study was a prospective cohort study with an aim to identify environmental causes of T1D and included children that arc at genetic risk. The children were followed from birth with
regular blood sampling. Certain examined patients were shown to have developed two or more autoantibodies.
[0174] The study also investigated important factors that can contribute to the variable rate of progression to T1D among children who were already positive for autoantibodies. The data suggested that the risk of progression to T1D was not different among patients with or without a family history of T1D. The risk of progression to T1D was higher for females, when a few high-risk HLA-DR and HLA-DQ subtypes were present. The risk of progression to T1D was inversely related to the age of first appearance of positive autoantibodies.
[0175] Subjects that have detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2) are provided herein. Additionally, PBMCs that produce antibodies against one or more islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2 are provided.
[0176] It is also recognized that subjects as described herein can be female. In some embodiments, female subjects can have a HLA-DR3, DQBl*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
C. T1D Treatments
[0177] As discussed herein, insulin is the predominant treatment for T1D. Various types of insulin can be used, however. A T1D therapeutic agent as described herein may include shortacting insulin. Short-acting insulin generally starts working 30 minutes after administration (e.g., via injection) and reaches peak effect at 90 to 120 minutes. Exemplary short-acting insulin therapeutics include Humulin R, NovolinR, and Afrezza.
[0178] A T1D therapeutic agent as described herein may include rapid-acting insulin. Rapid- acting insulin generally starts working within 15 minutes after administration (e.g., via injection) and reaches peak effect at 60 minutes. Rapid-acting insulin can last for periods of up
to 4 hours after administration. Exemplary rapid-acting insulin therapeutics include glulisine, lispro, and aspart.
[0179] A T1D therapeutic agent as described herein may include intermediate- acting insulin. Intermediate-acting insulin generally starts working 1 to 3 hours after administration (e.g., via injection) and reaches peak effect at 6-8 hours after administration. Intermediate-acting insulin can last for periods of up to 12 to 24 hours after administration. An exemplary intermediate-acting insulin therapeutics includes insulin NPH.
[0180] A T1D therapeutic agent as described herein may include long-acting insulin. Long-acting insulin generally works for 14 to 40 hours after administration (e.g., via injection). Exemplary intermediate- acting insulin therapeutics include insulin glargine, detemir, and degludec.
[0181] While insulin is one T1D therapeutic agent, it is not the only one. Additional T1D therapeutic agents include metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
[0182] The present disclosure provides that a subject described herein may have received one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin. A PBMC or islet cell as provided herein may have been exposed to one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
[0183] Additionally, technologies described herein can be used to determine how assess how a subject’s PBMCs and/or islet cells respond to T1D therapeutics, including one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin. Technologies described herein may also be useful for providing prognostic and/or predictive information about how a subject may respond to T1D therapeutics,
including one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
II. Non-Human Animals
[0184] The present disclosure recognizes that non-human animals can be engineered such that the animals sufficiently model human disease, disorders, and conditions. Such non-human animals can provide insights into disease pathology and etiology. Non-human animals can also be useful in assessing the safety and efficacy of treatment (e.g., a therapeutic agent).
[0185] The present disclosure provides that non-human animals can be engineered such that the animals sufficiently model autoimmune disorders, such as T1D. These animals can be utilized to determine the pathology and etiology of autoimmune disorders, such as T1D.
However, a powerful benefit to non-human animals provided herein is that the animals can be engineered to model a particular subject’s (e.g., a particular human’s) autoimmune disorder, such as T1D, and can be used to assess how that particular subject will respond to a treatment, including the safety and efficacy of such a treatment (e.g., a therapeutic agent).
[0186] Accordingly, non-human animals of the present disclosure represent a robust tool for evaluating autoimmunity, T1D, and potential therapeutics at a high level, but also provide a tool for determining a personalized approach to treating a patient.
A. Exemplary Non-Human Animals
[0187] Non-human animals are provided by the present disclosure. In some embodiments, a non-human animal is a mammal. In some embodiments, a non-human mammal is a primate, a goat, a sheep, a pig, a dog, a cow, or a rodent. In some embodiments, a non- human animal of the present disclosure is a rodent. In some embodiments, a rodent is a mouse or a rat.
[01881 In some embodiments, a non-human animal is a mouse. In some embodiments, a mouse is a NOD/ShiLtJ strain (sometimes referred to as NOD strain) mouse. In some embodiments, a mouse is a NSG mouse (also referred to as a N0D.Cg-PrkdcSl l<lIL2rg""l wjl/SzJ mouse). In some embodiments, a mouse is a NSG-SGM3 mouse.
[0189] In some embodiments, a non-human animal is a mouse. In some embodiments, a mouse comprises reduced function of non-human islet cells relative to a comparable wild-type mouse. In some embodiments, a mouse comprises reduced activity of non-human islet cells relative to a comparable wild-type mouse. In some embodiments, a mouse comprises a reduced number of non-human islet cells relative to a comparable wild-type mouse. In some embodiments, a mouse does not comprise murine islet cells. In some embodiments, a mouse has reduced pancreatic function relative to a comparable wild-type mouse. In some embodiments, a mouse has reduced pancreatic activity relative to a comparable wild- type mouse. In some embodiments, a mouse has a reduced number of pancreatic cells relative to a comparable wildtype mouse. In some embodiments, a mouse does not have a pancreas. In some embodiments, a non-human animal is a mouse that has had its pancreas removed. In some embodiments, a non- human animal is a mouse that has had its islet cells removed or destroyed. In some embodiments, a non-human animal is a mouse that has had its beta cells removed or destroyed. In some embodiments, a non-human animal is a mouse that has been treated with STZ. In some embodiments, a mouse is a NOD/ShiLtJ strain (sometimes referred to as NOD strain) mouse. In some embodiments, a mouse is a NSG mouse. In some embodiments, a mouse is a NSG-SGM3 mouse. In some embodiments, a mouse is a C57BL strain mouse. In some embodiments, a C57BL mouse is a C57BL/A mouse, C57BL/An mouse, C57BL/GrFa mouse, C57BL/KaLwN mouse, C57BL/6 mouse, C57BL/6J mouse, C57BL/6ByJ mouse, C57BL/6NJ mouse, C57BL/10 mouse, C57BL/10ScSn mouse, C57BL/10Cr mouse, or C57BL/01a mouse. In some embodiments, a mouse is a BALB strain mouse, e.g., a BALB/c mouse. In some embodiments, a mouse is a 129 strain mouse. In some embodiments, a 129 strain mouse is a 129P1 mouse, 129P2 mouse, 129P3 mouse, 129X1 mouse, 129S1 mouse (e.g., 129S1/SV mouse, 129Sl/SvIm mouse), 129S2 mouse, 129S4 mouse, 129S5 mouse, 129S9/SvEvH mouse, 129/SvJac mouse,
129S6 (129/SvEvTac) mouse, 129S7 mouse, 129S8 mouse, 129T1 mouse, or 129T2 mouse (see, e.g., Festing et al., 1999, Mammalian Genome 10:836; Auerbach, W. et al., 2000, Biotechniques 29(5): 1024-1028, 1030, 1032, each of which is incorporated herein by reference in its entirety). In some embodiments, a mouse is a mix of two or more of the aforementioned strains.
[0190] In some embodiments, a non-human animal is a rat. In some embodiments, a rat is a BB Wistar rat, Goto-Kakizaki (GK) rat, Zucker diabetic fatty (ZDF) rat, Komeda diabetes- prone (KDP) rat, BBDP rat, LETL rat, LEW.lARl/Ztm-iddm rat, BBDR rat, PVG rat, PVG.RT1U rat, PVG.R8 rat, WAG rat, WF.iddm4d congenic rat, or LEW.1WR1 rat. In some embodiments, a rat is a BB Wistar rat. In some embodiments, a rat is a KDP rat.
[0191] In some embodiments, a non-human animal is a rat. In some embodiments, a non-human animal is a rat. In some embodiments, a rat comprises reduced function of non- human islet cells relative to a comparable wild-type rat. In some embodiments, a rat comprises reduced activity of non-human islet cells relative to a comparable wild- type rat. In some embodiments, a rat comprises a reduced number of non-human islet cells relative to a comparable wild-type rat. In some embodiments, a rat does not comprise murine islet cells. In some embodiments, a rat has reduced pancreatic function relative to a comparable wild-type rat. In some embodiments, a rat has reduced pancreatic activity relative to a comparable wild-type rat. In some embodiments, a rat has a reduced number of pancreatic cells relative to a comparable wild-type rat. In some embodiments, a rat does not have a pancreas. In some embodiments, a rat has had its pancreas removed. In some embodiments, a non-human animal is a rat that has had its islet cells removed or destroyed. In some embodiments, a non-human animal is a rat that has had its beta cells removed or destroyed. In some embodiments, a non- human animal is a rat that has been treated with STZ. In some certain embodiments, a rat is a BB Wistar rat, Goto-Kakizaki (GK) rat, Zucker diabetic fatty (ZDF) rat, Komeda diabetes-prone (KDP) rat, BBDP rat, LETL rat, LEW.l ARl/Ztm-iddm rat, BBDR rat, PVG rat, PVG.RT1U rat, PVG.R8 rat, WAG rat, WF.iddm4d congenic rat, or LEW.1WR1 rat, a Wistar rat, LEA strain rat, Sprague Dawley strain rat, Fischer strain rat, F344 rat, F6 rat, or Dark Agouti rat. In some embodiments, a rat is a mix of two or more of the aforementioned strains.
[01921 Female mice tend to develop diabetes sooner than corresponding male mice.
Accordingly, in some embodiments, a mouse is a female mouse. However, the present disclosure also contemplates using male mice. Thus, in some embodiments, a mouse is a male mouse.
[0193] The present disclosure recognizes that the use of multiple mice can be particularly helpful for certain uses of mice described herein. Therefore, the present disclosure provides a plurality of mice as described herein. In some embodiments, a plurality of mice are each the same strain. In some embodiments, a plurality of mice can be a colony. In some embodiments, a plurality of mice can be a cohort. In some embodiments, a cohort is defined by a generation of mice. In some embodiments, a cohort is defined by the sex of the mice. For example, in some embodiments, a cohort comprises female mice. In some embodiments, a cohort comprises male mice. In some embodiments, a cohort is defined by two or more criteria. For example, in some embodiments, a cohort comprises female mice from the same generation of mice.
B. Cell Donor Subjects
[0194] Provided herein are non-human animals as described above, which comprise PBMCs and islet cells derived from cells of a subject. Said another way, a subject of the present disclosure is a donor of PBMCs and/or other cells from which islet cells are differentiated.
Accordingly, characteristics and features of a subject can also be or affect characteristics and features of PBMCs and islet cells derived from cells of the subject. Further, in some embodiments, a subject may also be a recipient of islet cells which were derived from the subject’s cells. In some embodiments, a subject can receive islet cells as part of a cell therapy, e.g., for T1D.
[0195] In some embodiments, a subject is a mammal. In some embodiments, a subject is a primate, a goat, a sheep, a pig, a dog, a cow, or a rodent. In some embodiments, a subject is a human.
[01961 In some embodiments, a subject has T1D. In some embodiments, a subject is at risk for developing T1D. In some embodiments, a subject has early indications of T1D, such as inflammation of the islets of Langerhans.
[0197] A subject provided herein may be in need of a T1D therapeutic agent. In some embodiments, a subject is eligible for islet transplant therapy.
[0198] In some embodiments, a subject had T1 D onset at an age of less than 50 years old, less than 45 years old, less than 40 years old, less than 35 years old, less than 30 years old, or less than 25 years old. In some embodiments, a subject had T ID onset at an age of less than 40 years old.
[0199] In some embodiments, a subject has had insulin dependence for at least 2 years, at least 3 years, at least 4 years, at least 5 year’s, at least 6 year’s, at least 7 years, at least 8 years, at least 9 years, or at least 10 years. In some embodiments, a subject has had insulin dependence for at least 5 years.
[0200] Diabetes can increase in severity with age. In some embodiments, a subject is at least 15 years old, at least 20 year’s old, at least 25 years old, at least 30 year’s old, at least 35 years old, at least 40 years old, at least 45 years old, or at least 50 years old.
[0201] In some embodiments, a subject with T1D has had T1D for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 years, at least 8 years, at least 9 years, at least 10 years, at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 year’s, or at least 90 years. In some embodiments, a subject with T1D has had T1D for at least 5 years.
[0202] The age of a subject considered with the number of years the subject has been insulin dependent can be helpful in assessing T1D severity and/or the risk of T1D severity. As the sum of a subject’s age and their years of insulin dependent diabetes increases, the risk of severity and/or severity of T1D for the patient also increases. Thus, in some embodiments, a
sum of age and years of insulin dependent diabetes is at least 15 years, at least 20 years, at least 25 years, at least 26 years, at least 27 years, at least 28 years, at least 29 years, or at least 30 years. In some embodiments, a sum of age and years of insulin dependent diabetes is at least 28 years.
[0203] A C-peptide test measures the amount of C-peptide in the blood or urine. The pancreas releases C-peptide when it makes insulin. A C-peptide test can help determine the type of diabetes a patient has (i.e., type 1 diabetes or type 2 diabetes) or how well diabetes treatments are working. C-peptide is a byproduct the pancreas releases into the body when it makes insulin. The pancreas produces about the same amount of insulin and C-peptide. Because C-peptide is a byproduct of insulin production, this test can reveal how much insulin the body is producing. When people take insulin, their bodies do not make or release C-peptide. This test can tell the difference between insulin made by the body (endogenous) and insulin from medication (exogenous). Therefore, a C-peptide test indicates if a subject’s body has the ability to make its own insulin from the pancreas.
[0204] In some embodiments, a subject has less than 0.5 ng/mL, less than 0.4 ng/mL, less than 0.3 ng/mL, less than 0.2 ng/mL, less than 0.1 ng/mL, or an undetectable level of stimulated C-peptide at 60 minutes post-mixed-meal tolerance test. In some embodiments, a subject has less than 0.5 ng/mL, less than 0.4 ng/mL, less than 0.3 ng/mL, less than 0.2 ng/mL, less than 0.1 ng/mL, or an undetectable level of stimulated C-peptide at 90 minutes post-mixed- meal tolerance test. In some embodiments, a subject has less than 0.5 ng/mL, less than 0.4 ng/mL, less than 0.3 ng/mL, less than 0.2 ng/mL, less than 0.1 ng/mL, or an undetectable level of stimulated C-peptide at 60 minutes post-mixed-meal tolerance test. In some embodiments, a subject has less than 0.5 ng/mL, less than 0.4 ng/mL, less than 0.3 ng/mL, less than 0.2 ng/mL, less than 0.1 ng/mL, or an undetectable level of stimulated C-peptide at 60 minutes and 90 minutes post-mixed-meal tolerance test. In some embodiments, a subject has less than 0.3 ng/mL of stimulated C-peptide at 60 minutes post-mixed-meal tolerance test. In some embodiments, a subject has less than 0.3 ng/mL of stimulated C-peptide at 90 minutes post-
mixed-meal tolerance test. In some embodiments, a subject has less than 0.3 ng/mL of stimulated C-peptide at 60 minutes and 90 minutes post-mixed-meal tolerance test.
[0205] In some embodiments, a subject has intensive diabetes management. In some embodiments, intensive diabetes management comprises self-monitoring of glucose values no less than a mean of three times each day averaged over each week. In some embodiments, intensive diabetes management comprises administration of three or more insulin injections each day or insulin pump therapy. In some embodiments, intensive diabetes management comprises at least three clinical evaluations during the past 12 months with an endocrinologist, diabetologist, or diabetes specialist. In some embodiments, intensive diabetes management comprises self-monitoring of glucose values no less than a mean of three times each day averaged over each week, administration of three or more insulin injections each day or insulin pump therapy, and/or at least three clinical evaluations during the past 12 months with an endocrinologist, diabetologist, or diabetes specialist.
[0206] In some embodiments, a subject has had at least one episode of severe hypoglycemia in the past 12 months. In some embodiments, an episode of severe hypoglycemia comprises an event with one or more of the following symptoms (i) memory loss, (ii) confusion, (iii) uncontrollable behavior, (iv) irrational behavior, (v) unusual difficulty in awakening, (vi) suspected seizure, (vii) seizure, (viii) loss of consciousness, and (ix) visual symptoms.
[0207] In some embodiments, a subject has impaired awareness of hypoglycemia (IAH). IAH is defined as “a diminished ability to perceive the onset of acute hypoglycaemia.” IAH can arise when repeated episodes of hypoglycaemia raise the glycaemic threshold for symptoms to occur.
[0208] In some embodiments, IAH can be assessed using a Clarke reduced awareness score. In some embodiments, a subject has a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more. In some embodiments, a subject has a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more. In some embodiments, a subject
has a marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more. In some embodiments, a subject has a composite of: a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile.
[0209] In some embodiments, a subject has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years.
[0210] In some embodiments, a subject has islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2). In some embodiments, a subject has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2).
[0211] In some embodiments, a subject has antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
[0212] In some embodiments, a subject has one or more genetic factors associated with T1D.
[0213] In some embodiments, a subject has a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype. In some embodiments, a subject has a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) and HLA- DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
[0214] In some embodiments, a subject comprises a class I VNTR at an insulin gene promoter.
[0215] In some embodiments, a subject comprises a polymorphism within a CTLA-4 gene that results in a decreased level of CTLA-4 expression as compared to level of CTLA-4 expression observed with a comparable subject that does not have the polymorphism.
[0216] In some embodiments, a subject comprises a C1858T (Arg620Trp) polymorphism in a PTPN22 gene.
[0217] In some embodiments, a subject comprises one or more mutations in an AIRE gene. In some embodiments, expression of AIRE in a subject with one or more mutations in an AIRE gene may be decreased compared to a subject without the one or more mutations.
[0218] In some embodiments, a subject as described herein can comprise one or more mutations in a FoxP3 gene. In some embodiments, expression of scurfin in a subject with one or more mutations in a FoxP3 gene may be decreased compared to a subject without the one or more mutations.
[0219] In some embodiments, a subject comprises one or more mutations in a STAT3 gene. In some embodiments, expression of STAT3 in a subject with one or more mutations in a STAT3 gene may be decreased compared to a subject without the one or more mutations.
[0220] In some embodiments, a subject comprises one or more mutations in an IFIH1 gene. In some embodiments, expression of MDA5 in a subject with one or more mutations in an 1F1H1 gene may be decreased compared to a subject without the one or more mutations.
[0221] In some embodiments, a subject comprises one or more mutations in an HIP14 gene. In some embodiments, expression of HIP14 in a subject with one or more mutations in an HIP14 gene may be decreased compared to a subject without the one or more mutations.
[0222] In some embodiments, a subject comprises one or more mutations in an ERBB3 gene. In some embodiments, expression of ERBB3 in a subject with one or more mutations in an ERBB3 gene may be decreased compared to a subject without the one or more mutations.
[02231 In some embodiments, a subject has been exposed to one or more environmental factors associated with the development of T1D. In some embodiments, a subject has been exposed to and/or been infected with a coxsackieviruses strain B4, Bl, A4, A2, and/or A16. In some embodiments, a subject has been exposed to and/or infected with an enterovirus.
[0224] In some embodiments, a subject produces antibodies ABBOS. In some embodiments, a subject has a microbiome with a reduced Firmicutes:Bacteroidetes ratio as compared to a comparable subject that has not been diagnosed with an autoimmune disorder (e.g., T1D). In some embodiments, a subject was birthed via cesarean section.
[0225] In some embodiments, a subject has one or more immunological factors associated with T1D. In some embodiments, a subject has a disrupted thymus function. In some embodiments, a subject comprises one or more mutations in an AIRE gene and has had prior exposure to a coxsackieviruses strain B4, Bl, A4, A2, and/or A16 or an enterovirus.
[0226] In some embodiments, a subject has a disrupted Treg function.
[0227] In some embodiments, a subject has inflammation (e.g., chronic inflammation, e.g., chronic atrophic inflammation) within the islets of Langerhans.
[0228] In some embodiments, a subject has an increased level of proinflammatory cytokines IL-1, TNF-a, and INF-y when compared to a comparable healthy subject.
[0229] In some embodiments, a subject is female. In some embodiments, a female subject has a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
[0230] Subjects provided herein may have previously been administered a T1D therapeutic agent. Subjects provided herein may currently be receiving a T1D therapeutic agent. In some embodiments, a subject has received one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin. A PBMC or islet cell as provided herein may have been exposed to one or more of Humulin R, NovolinR, Afrezza,
glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
C. Immune Cells
[0231] As described herein, provided herein are immune cells (e.g., peripheral blood mononuclear cells (PBMCs)) from a subject (e.g., a patient with type 1 diabetes (T ID)), which are delivered to a non-human animal as described herein. In some embodiments, immune cells (e.g., PBMCs (and/or other cells from which islet cells are differentiated)) are obtained from the same subject (e.g., the immune cells and/or the islet cells are autologous). Accordingly, characteristics and features of a subject can also be or affect characteristics and features of immune cells (e.g., PBMCs) from the subject. Also, as immune cells (e.g., PBMCs) are obtained from a subject as described herein, immune cells (e.g., PBMCs) provided can be mammalian immune cells (e.g., PBMCs), e.g., human immune cells (e.g., human PBMCs).
[0232] The present disclosure contemplates that immune cells (e.g., PBMCs) can be used in at least a couple different ways. In some embodiments, immune cells (e.g., PBMCs) can be used as obtained. For instance, in some embodiments, immune cells (e.g., PBMCs) can be administered to a non-human animal.
[0233] In some embodiments, immune cells (e.g., PBMCs) can be modified. For example, immune cells (e.g., PBMCs) can be used to generate iPSCs. In some embodiments, iPSCs can then be differentiated into islet cells. In some embodiments, immune cells (e.g., PBMCs) can also be genetically modified as described herein.
[0234] In some embodiments, immune cells as described herein comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, dendritic cells, or a combination thereof. In some embodiments, PBMCs as described herein comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, dendritic cells, or a combination thereof. In some embodiments, one or more of the immune cells are T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells. In some embodiments, one or more of the PBMCs are T cells, B
cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
[0235] In some embodiments, one or more of immune cells is a B cell. In some embodiments, one or more of immune cells produce autoantibodies against autoantigens of a subject (e.g., a mammal, e.g., a human). In some embodiments, one or more immune cells produce antibodies against cells or antigens of a subject (e.g., a mammal, e.g., a human). In some embodiments, one or more of immune cells produce antibodies against islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2. In some embodiments, one or more of immune cells produce islet cell antibodies (ICA) and/or insulin autoantibodies (IAA).
[0236] In some embodiments, one or more of PBMCs is a B cell. In some embodiments, one or more of PBMCs produce autoantibodies against autoantigens of a subject (e.g., a mammal, e.g., a human). In some embodiments, one or more PBMCs produce antibodies against cells or antigens of a subject (e.g., a mammal, e.g., a human). In some embodiments, one or more of PBMCs produce antibodies against islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2. In some embodiments, one or more of PBMCs produce islet cell antibodies (ICA) and/or insulin autoantibodies (IAA).
[0237] In some embodiments, one or more of immune cells is an islet-reactive T cell. In some embodiments, one or more of PBMCs is an islet-reactive T cell.
[0238] In some embodiments, one or more of immune cells are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal. In some embodiments, one or more of PBMCs are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal.
[0239] In some embodiments, at least about 2.5xl05, at least about 5xl05, at least about IxlO6, at least about 2xl06, at least about 3xl06, at least about 4xl06, at least about 5xl06, at least about 6xl06, at least about 7xl06, at least about 8xl06, at least about 9xl06, at least about IxlO7, at least about 1.5xl07, or at least about 2xl07 immune cells from a subject (e.g., a
mammal, e.g., a human) are administered to a non-human animal (e.g., a mammal, e.g., a rodent, e.g., a rat or a mouse). In some embodiments, a non-human animal (e.g., a mammal, e.g., a rodent, e.g., a rat or a mouse) as described herein comprises at least about 2.5xl05, at least about 5xl05, at least about IxlO6, at least about 2xl06, at least about 3xl06, at least about 4xl06, at least about 5xl06, at least about 6xl06, at least about 7xl06, at least about 8xl06, at least about 9xl06, at least about IxlO7, at least about 1.5xl07, or at least about 2xl07 immune cells from a subject (e.g., a mammal, e.g., a human).
[0240] In some embodiments, at least about 2.5xl05, at least about 5xl05, at least about IxlO6, at least about 2xl06, at least about 3xl06, at least about 4xl06, at least about 5xl06, at least about 6xl06, at least about 7xl06, at least about 8xl06, at least about 9xl06, at least about IxlO7, at least about 1.5xl07, or at least about 2xl07 PBMCs from a subject (e.g., a mammal, e.g., a human) are administered to a non-human animal (e.g., a mammal, e.g., a rodent, e.g., a rat or a mouse). In some embodiments, a non-human animal (e.g., a mammal, e.g., a rodent, e.g., a rat or a mouse) as described herein comprises at least about 2.5xl05, at least about 5xl05, at least about IxlO6, at least about 2xl06, at least about 3xl06, at least about 4xl06, at least about 5xl06, at least about 6xl06, at least about 7xl06, at least about 8xl06, at least about 9xl06, at least about IxlO7, at least about 1.5xl07, or at least about 2xl07 PBMCs from a subject (e.g., a mammal, e.g., a human).
[0241] In some embodiments, one or more of immune cells (e.g., PBMCs) comprise one or more genetic factors associated with T1D.
[0242] In some embodiments, one or more of immune cells (e.g., PBMCs)comprise a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) or HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype. In some embodiments, one or more of immune cells (e.g., PBMCs)comprise a HLA-DR3, DQB 1*0201 (also referred as DR3-DQ2) and HLA-DR4, DQB 1*0302 (also referred as DR4-DQ8) haplotype.
[0243] In some embodiments, one or more of immune cells (e.g., PBMCs)comprise a class I VNTR at an insulin gene promoter.
[02441 In some embodiments, one or more of immune cells (e.g., PBMCs)comprise a polymorphism within a CTLA-4 gene that results in a decreased level of CTLA-4 expression as compared to level of CTLA-4 expression observed with a comparable subject that does not have the polymorphism.
[0245] In some embodiments, one or more of immune cells (e.g., PBMCsjcomprise a C1858T (Arg620Trp) polymorphism in a PTPN22 gene.
[0246] In some embodiments, one or more of immune cells (e.g., PBMCsjcomprise one or more mutations in an AIRE gene. In some embodiments, expression of AIRE in a PBMC with one or more mutations in an AIRE gene may be decreased compared to a comparable immune cell (e.g., PBMCjwithout the one or more mutations.
[0247] In some embodiments, one or more of immune cells (e.g., PBMCsJcomprisc one or more mutations in a FoxP3 gene. In some embodiments, expression of scurfin in an immune cell (e.g., PBMC) with one or more mutations in a FoxP3 gene may be decreased compared to a comparable immune cell (e.g., PBMC) without the one or more mutations.
[0248] In some embodiments, one or more of immune cells (e.g., PBMCs)comprise one or more mutations in a STAT3 gene. In some embodiments, expression of STAT3 in an immune cell (e.g., PBMC) with one or more mutations in a STAT3 gene may be decreased compared to a comparable immune cell (e.g., PBMC) without the one or more mutations.
[0249] In some embodiments, one or more of immune cells (e.g., PBMCsjcomprise one or more mutations in an IFIH1 gene. In some embodiments, expression of MDA5 in an immune cell (e.g., PBMCjwith one or more mutations in an IFIH1 gene may be decreased compared to a comparable immune cell (e.g., PBMC)without the one or more mutations.
[0250] In some embodiments, one or more of immune cells (e.g., PBMCs)comprise one or more mutations in an HIP14 gene. In some embodiments, expression of HIP14 in an immune cell (e.g., PBMC) with one or more mutations in an HIP14 gene may be decreased compared to a comparable immune cell (e.g., PBMC) without the one or more mutations.
[02511 In some embodiments, one or more of immune cells (e.g., PBMCs)comprise one or more mutations in an ERBB3 gene. In some embodiments, expression of ERBB3 in an immune cell (e.g., PBMC) with one or more mutations in an ERBB3 gene may be decreased compared to a comparable immune cell (e.g., PBMC) without the one or more mutations.
[0252] In some embodiments, an immune cell (e.g., PBMC) has been exposed to one or more environmental factors associated with the development of T1D. In some embodiments, an immune cell (e.g., PBMC) has been exposed to a coxsackieviruses strain B4, Bl, A4, A2, and/or A16. In some embodiments, an immune cell (e.g., PBMC) has been exposed to an enterovirus.
[0253] In some embodiments, an immune cell (e.g., PBMC) has been obtained from a subject that produces antibodies ABBOS. In some embodiments, an immune cell (e.g., PBMC) has been obtained from a subject that has a microbiome with a reduced Firmicutes:Bacteroidetes ratio as compared to a comparable subject that has not been diagnosed with an autoimmune disorder (e.g., T1D). In some embodiments, an immune cell (e.g., PBMC) has been obtained from a subject that was birthed via cesarean section.
[0254] In some embodiments, an immune cell (e.g., PBMC) has been obtained from a subject that has one or more immunological factors associated with T1D. In some embodiments, an immune cell (e.g., PBMC) has been obtained from a subject that has a disrupted thymus function. In some embodiments, an immune cell (e.g., PBMC) has been obtained from a subject that comprises one or more mutations in an AIRE gene and that has had prior exposure to a coxsackieviruses strain B4, Bl, A4, A2, and/or A16 or an enterovirus.
[0255] In some embodiments, an immune cell (e.g., PBMC) has been obtained from a subject that has a disrupted Treg function.
[0256] In some embodiments, an immune cell (e.g., PBMC) has been obtained from a subject that has inflammation (e.g., chronic inflammation, e.g., chronic atrophic inflammation) within the islets of Langerhans.
[02571 In some embodiments, an immune cell (e.g., PBMC) has been obtained from a subject that has an increased level of proinflammatory cytokines IL-1, TNF-a, and INF-y when compared to a comparable healthy subject.
[0258] In some embodiments, an immune cell (e.g., PBMC) has been obtained from a subject that is female. In some embodiments, an immune cell (e.g., PBMC) has been obtained from a female subject that has a HLA-DR3, DQBl*0201 (also referred as DR3-DQ2) or HLA- DR4, DQB 1*0302 (also re I erred as DR4-DQ8) haplotype.
[0259] Immune cells (e.g., PBMCs) can be obtained from subjects that may have previously been administered a T1D therapeutic agent. Immune cells (e.g., PBMCs) can be obtained from subjects that may currently be receiving a T1D therapeutic agent. In some embodiments, an immune cell (e.g., PBMC) has been obtained from a subject that has received one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin. An immune cell (e.g., PBMC) may have been exposed to one or more of Humulin R, NovolinR, Afrezza, glulisine, lispro, aspart, insulin NPH, glargine, detemir, degludec, metformin, pramlinide, blood pressure medications, cholesterol regulating medications, and aspirin.
D. Islet Cells
[0260] The pancreas contains clusters of cells, known as islets, that produce hormones. There are several different types of cells in an islet. For example, alpha cells produce glucagon; delta cells produce somatostatin; and beta cells produce insulin. A sample of pancreatic islet cells may comprise at least alpha cells, delta cells, and beta cells. As used throughout, a “beta cell” should be understood to be a beta islet cell. In some embodiments, an islet cell is an islet progenitor cell, an immature islet cell, or a mature islet cell.
[0261] As described elsewhere herein, beta-islet cells used in a cell therapy product may be pluripotent stem cell (iPSC)-derived beta-islet cells. Methods for profiling a population of cells for donor capability as described anywhere herein may also be performed on stem cells
capable of differentiating to form beta-islet cells. Methods for profiling a population of cells for donor capability as described anywhere herein may also be performed on stem cell derived (e.g., genome-edited) beta-islet cells.
[0262] Relevant information concerning beta-islet cells as referred to in the context of the present disclosure is known in the art, including certain information regarding desired features of beta-islet cells when used for cell therapy. It will be understood that embodiments concerning beta-islet cells described herein may be readily and appropriately combined with embodiments describing HIP cells (e.g., exhibiting reduced expression of one or more molecules of the MHC class I and/or MHC class II molecules and increased expression of at least one tolerogenic factor), as well as embodiments describing safety switches, and other modified/ gene edited cells as described herein. Beta-islet cells to be used in a cell therapy product may be profiled for donor capability at any stage of the editing process during manufacturing of the cell therapy product.
[0263] A key functional feature of a P cell is its ability to repeatedly perform glucose stimulated insulin secretion (GSIS). In certain embodiments, assays can be performed to determine the physiological function in vitro of secreting insulin in response to glucose. In certain embodiments, the GSIS assay may be a perifusion GSIS (dynamic GSIS) assay (for example as in Velazco-Cruz, Stem Cell Reports, 2019).
[0264] In certain embodiments, other assays can be performed to examine the expression of specific genes, pathways, and transcription factors. Such assays include those detecting the presence of Yap (Rosado-Olivieri et al., 2019, which is incorporated herein by reference in its entirety), the ROCKII pathway (Ghazizadeh et al., 2017, which is incorporated herein by reference in its entirety), the transforming growth factor b (TGF-b) pathway (Velazco-Cruz, et al., 2019, which is incorporated herein by reference in its entirety), the cytoskeleton (Hogrebe et al., 2020, which is incorporated herein by reference in its entirety), and the expression of SIX2 (Velazco-Cruz et al., Cell Reports, 2020, which is incorporated herein by reference in its entirety). In certain embodiments, other transcription factors important for the SC-P cell
phenotype include PDX1, NKX6-1, NKX2-2, and NEURODI (Hogrebe et al., 2020, which is incorporated herein by reference in its entirety).
[0265] In certain embodiments, an assay measuring changes in intracellular Ca2+ may be performed as described in Pagliuca et al. (Cell, 2014, which is incorporated herein by reference in its entirety). 0 cells sense changing glucose levels through calcium signaling; increasing glucose levels leads to membrane depolarization causing an influx of calcium ions which triggers insulin exocytosis (Mohammed et al., 2009, which is incorporated herein by reference in its entirety). In certain embodiments, the functional SC-0 cells exhibit calcium flux similarly to primary human islet cells.
[0266] In certain embodiments, assays can also be performed to assess in vivo functionality of the SC-0 cells. An example of such an assay can be found in Pagliuca et al. (Cell, 2014, which is incorporated herein by reference in its entirety). Briefly, to test their capacity to function in vivo, SC-0 cells are transplanted under the kidney capsule of immunocompromised mice and the ability of the cells to produce insulin is analyzed.
[0267] The beta-islet cells described herein may be used to treat or prevent a disease in a subject.
[0268] As described herein, provided herein are islet cells that are derived (e.g., differentiated) from cells of a subject (e.g., a patient with type 1 diabetes (T1D)). In some embodiments, cells from which islet cells are differentiated (and/or PBMCs) are obtained from the same subject (e.g., the PBMCs and the islet cells are autologous). Accordingly, characteristics and features of a subject can also be or affect characteristics and features of islet cells derived from cells of the subject and PBMCs from the subject. Also, as PBMCs are obtained from a subject as described herein, PBMCs provided can be mammalian PBMCs, e.g., human PBMCs.
[0269] Beta-islet cells as described herein to be used in a cell therapy product may be present with alpha cells and delta cells in the cell therapy product. Islet cells to be used in a cell therapy product may be profiled for donor capability at any stage of the manufacturing process
of the cell therapy product. In some embodiments, islet cells are or comprise alpha, beta, and/or delta islet cells. In some embodiments, islet cells are or comprise about 5%, about 10%, about 15%, about 20%, about 25%, about 35%, or about 40% alpha cells. In some embodiments, islet cells are or comprise about 10%, about 15%, about 20%, about 25%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, or about 80% beta cells. In some embodiments, islet cells are or comprise about 1%, about 2%, about 3%, about 4%, about 5%, about 6%, about 7%, about 8%, about 9%, about 10%, about 15%, about 20%, or about 25% delta cells.
[0270] In some embodiments, islet cells are or comprise differentiated cells. In some embodiments, differentiated cells are derived from a stem cell. In some embodiments, differentiated cells are derived from a pluripotent stem cell. In some embodiments, differentiated cells are derived from an induced pluripotent stem cell (iPSC). In some embodiments, an iPSC is derived from a cell obtained from the human patient with T1D. In some embodiments, a cell obtained from a human patient with T1D is selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell. In some embodiments, a cell obtained from a human patient with T1D is a PBMC. In some embodiments, a stem cell is derived from a cell obtained from a human patient with T ID. In some embodiments, a stem cell is an induced pluripotent stem cell (iPSC) derived from a cell selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell. In some embodiments, a stem cell is an induced pluripotent stem cell (iPSC) derived from a PBMC. In some embodiments, differentiated cells are derived from a multipotent stem cell. In some embodiments, a multipotent stem cell is derived from a cell obtained from a human patient with T1D. In some embodiments, differentiated cells are derived from a lineage- specific stem cell. In some embodiments, a lineage- specific stem cell is derived from a cell obtained from a
human patient with T1D. In some embodiments, differentiated cells are derived from a definitive endoderm cell. In some embodiments, a definitive endoderm cell expresses FOXA2 and/or SOX 17. In some embodiments, differentiated cells are derived from a primitive gut tube cell. In some embodiments, a primitive gut tube cell expresses FOXA2 and/or SOX 17. In some embodiments, differentiated cells are derived from a pancreatic progenitor cell. In some embodiments, a pancreatic progenitor cell expresses PDX1 and/or NKX6-1. In some embodiments, differentiated cells are derived from an endocrine cell. In some embodiments, an endocrine cell expresses NKX6-1, NEUROG3, and/or NKX2-2.
[0271] In some embodiments, islet cells produce insulin, c-peptide, glucagon, somatostatin, and/or pancreatic polypeptide.
[0272] In some embodiments, islet cells as described herein are used to make a first therapeutic agent as described herein.
[0273] In some embodiments, islet cells are introduced to a non-human animal of the present disclosure via intramuscular injection, intravenous injection, intraportal injection, ocular injection, injection into omentum, and/or injection into a kidney capsule.
1. Hypoimmunogenic Islet Cells
[0274] In some embodiments, islet cells are or comprise engineered islet cells. In some embodiments, engineered islet cells are derived (e.g., differentiated) from cells as described herein that have been engineered. In some embodiments, the cells as provided herein are beta islet cells derived from engineered iPSCs that contain modifications (e.g., genetic modifications) described herein and that are differentiated into beta islet cells. As will be appreciated by those in the art, the methods for differentiation depend on the desired cell type using known techniques. In some embodiments, the cells differentiated into various beta islet cells may be used for subsequent transplantation or engraftment into subjects (e.g., recipients). In some embodiments, pancreatic islet cells are derived from the engineered pluripotent cells described herein. Useful methods for differentiating pluripotent stem cells into beta islet cells are described, for example, in U.S. Patent No. 9,683,215; U.S. Patent No. 9,157,062; U.S. Patent No.
8,927,280; U.S. Patent Pub. No. 2021/0207099; Hogrebe et al., "Targeting the cytoskeleton to direct pancreatic differentiation of human pluripotent stem cells,” Nat. Biotechnol., 2020, 38:460-470; and Hogrebe et al., “Generation of insulin-producing pancreatic beta cells from multiple human stem cell lines,” Nat. Protoc., 2021, the contents of which are herein incorporated by reference in their entirety.
[0275] In some embodiments, the engineered pluripotent cells described herein are differentiated into beta-like cells or islet organoids for transplantation to address type I diabetes mellitus (T1DM). Cell systems are a promising way to address T1DM, see, e.g., Ellis et al, Nat Rev Gastroenterol Hepatol. 2017 Oct;14(10):612-628, incorporated herein by reference.
Additionally, Pagliuca et al. (Cell, 2014, 159(2):428-39) reports on the successful differentiation of P-cells from hiPSCs, the contents incorporated herein by reference in its entirety and in particular for the methods and reagents outlined there for the large-scale production of functional human P cells from human pluripotent stem cells). Furthermore, Vegas et al. shows the production of human P cells from human pluripotent stem cells followed by encapsulation to avoid immune rejection by the host; Vegas et al., Nat Med, 2016, 22(3):306- 11, incorporated herein by reference in its entirety and in particular for the methods and reagents outlined there for the large-scale production of functional human P cells from human pluripotent stem cells.
[0276] In some embodiments, cells as described herein have been modified to be hypoimmune cells. In some embodiments, engineered islet cells are derived (e.g., differentiated) from engineered induced pluripotent stem cells (iPSCs) modified to be hypoimmune iPSCs. In some embodiments, iPSCs have been derived (e.g., reprogrammed) from cells (e.g., PBMCs) of a subject back into a pluripotent state. In some embodiments, engineered islet cells have been modified to be hypoimmune islet cells.
[0277] In some embodiments, all modifications present in a hypoimmune islet cell are present in the iPSC that the hypoimmune islet cell is derived from. In some embodiments, a hypoimmune islet cell comprises one or more modifications in addition to the one or more modifications present in the iPSC the islet cell is derived from. In some embodiments, a
hypoimmune islet cell comprises one or more modifications, none of which are present in the iPSC the hypoimmune islet cell is derived from.
[0278] In some embodiments, an iPSC comprising one or more modifications is differentiated into a hypoimmune islet cell, wherein the hypoimmune islet cell comprises no additional modifications relative to the iPSC from which it is derived. In some embodiments, an iPSC comprising one or more modifications is differentiated into a hypoimmune islet cell, wherein the hypoimmune islet cell comprises one or more additional modifications relative to the iPSC from which it is derived. In some embodiments, an iPSC comprising no hypoimmune modifications is differentiated into an islet cell, wherein the islet cell is modified to comprise one or more modifications relative to the iPSC from which it is derived.
[0279] In some embodiments, the present disclosure provides methods of creating hypoimmune islet cells derived from iPSCs. In some embodiments, the iPSCs are hypoimmune iPSCs. In some embodiments, a method comprises engineering an iPSC to generate a hypoimmune iPSC comprising one or more modifications, and differentiating the hypoimmune iPSC into a hypoimmune islet cell, wherein the hypoimmune islet cell comprises the same one or more modifications as the hypoimmune iPSC. In some embodiments, a method comprises engineering an iPSC to generate an iPSC comprising one or more modifications, differentiating the iPSC into a hypoimmune islet cell, and then engineering the hypoimmune islet cells to comprise one or more additional modifications, wherein the hypoimmune islet cells comprises one or more additional modifications relative to the iPSC. In some embodiments, a method comprises differentiating an iPSC into an islet cell and then engineering the islet cell to comprise one or more modifications, thereby generating a hypoimmune islet cell, wherein the hypoimmune islet cell comprises one or more modifications relative to the iPSC.
[0280] In some embodiments, hypoimmune islet cells comprise one or more modifications that (a) inactivate or disrupt one or more alleles of: (i) one or more major histocompatibility complex (MHC) class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or (ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class
II molecules, and/or (b) increase expression of one or more tolerogenic factors, wherein the increased expression of (b) is relative to an islet cell that does not comprise the modifications. In some embodiments, one or more modifications that disrupt one or more MHC class I molecules and/or one or more MHC class 11 molecules reduce expression of the one or more MHC class 1 molecules and/or the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications. In some embodiments, hypoimmune islet cells are (i) HLA class I- deficient and/or HLA class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47.
[0281] Provided herein are engineered stem cell-derived islet cells obtained by in vitro differentiation of a modified pluripotent stem cell. The modified pluripotent stem cell can be any as described below. The provided engineered islet cells are differentiated in vitro from the modified pluripotent stem cell by any method able to generate a functional islet cell. In some of any embodiments, the differentiated engineered islet cell is a modified iPSC-derived beta islet cell. The provided engineered islet cells retain the one or more modifications of the modified pluripotent stem cells and/or retain or exhibit similar expression of the target immune molecules (e.g. reduced expression of MHC class I and/or II and increased expression of a tolerogenic factor, such as CD47). The modified islet cells provided herein also are functional and exhibit one or more functions of primary islet cells or beta islet cells, such as the ability to secrete insulin, for example glucose stimulated insulin secretion (GSIS).
[0282] Also provided are populations of cells containing the engineered islet cells. It is understood that differentiation from a population may not result in 100% having fully differentiated to the same stage in the differentiation pathway. Thus, it should be appreciated that not all cells in a particular population progress through these stages at the same rate, i.e., some cells may have progressed less, or more, down the differentiation pathway than the majority of cells present in the population. Accordingly, a population of islet cells may also include cells that are partially differentiated from the modified pluripotent stem cell or is a precursor of the cell stage such as precursor of the differentiated islet cell. In some cases, a percentage or portion of the cells may be at an earlier stage.
[02831 In some embodiments, engineered islet cells are differentiated in vitro (e.g., from pluripotent stem cells) and are cells that display at least one marker indicative of a pancreatic beta cell (e.g., PDX-1 or NKX6-1), express insulin, and display a GSIS response characteristic of an endogenous mature beta cell both in vitro and in vivo. In some embodiments, a marker indicative of a beta cell is a marker selected from INS, CHGA, NKX2-2, PDX1, NKX6-1, MAFB, GCK and GLUT1. In some embodiments, the GSIS response of an engineered islet cell can be observed within two weeks of transplantation of the engineered islet cell into a host (e.g., a human or animal). In some embodiments, it is to be understood that islet cells need not be derived (e.g., directly) from stem cells, as any method can be used that is capable of deriving engineered islet cells from any endocrine progenitor cell that expresses insulin or precursor thereof using any cell as a starting point in which such starting cell has been modified by the one or more modifications described herein. In some embodiments, a starting cell may be a modified cell according to the present disclosure that is an induced-pluripotent stem cells, progenitor cells, partially reprogrammed somatic cells (e.g., a somatic cell which has been partially reprogrammed to an intermediate state between an induced pluripotent stem cell and the somatic cell from which it was derived), multipotent cells, totipotent cells, a transdifferentiated version of any of the foregoing cells.
[0284] In some embodiments, engineered islet cells have regulated or modulated (e.g. reduced or eliminated) expression of MHC class I molecules, MHC class II molecules, or MHC class I and MHC class II molecules. In some embodiments, the regulated or modulated expression of MHC class I and/or Class II is due to gene editing in which the DNA of the gene loci involved in regulation of expression of MHC class I and/or class II have been edited to delete genomic DNA of a gene involved in regulation of expression of the immune molecule. In some embodiments, an engineered islet cell has an edit to delete genomic DNA of beta-2 microglobulin (B2M) and is thus reduced or eliminated for expression of MHC class I. In some embodiments, the B2M gene is knocked out in an engineered islet cell. In some embodiments, both alleles of B2M are knocked out. In some embodiments, an engineered islet cell has an edit to delete genomic DNA of CIITA and is thus reduced or eliminated for expression of MHC class
II. In some embodiments, the CIITA gene is knocked out in an engineered islet cell. In some embodiments, both alleles of CIITA are knocked out.
[0285] In some embodiments, engineered islet cells have regulated or modulated (e.g. increase) expression of a tolerogenic factor, such as CD47. In some embodiments, the tolerogenic factor is one or more of DUX4, B2M-HLA-E, CD16, CD52, CD47, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, CCL21, MFGE8, SERPINB9, CD35, IL-39, CD16 Fc Receptor, IL15-RF, and H2- M3, or any combination thereof. In some embodiments, the one or more tolerogenic factors are selected from the group consisting of CD 16, CD24, CD35, CD39, CD46, CD47, CD52, CD55, CD59, CD64, CD200, CCL22, CTLA4-Ig, Cl inhibitor, FASL, IDO1, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, IL-10, IL-35, PD-L1, SERPINB9, CCL21, MFGE8, DUX4, B2M-HLA-E, CD27, IL-39, CD16 Fc Receptor, IL15-RF, H2-M3 (HLA-G), A20/TNFAIP3, CR1, HLA-F, and MANF. In some embodiments, the increased or overexpressed tolerogenic factor is or includes increased expression of CCL21, PD-L1, FasL, Serpinb9, H2-M3 (HLA-G), CD47, CD200, and Mfge8. In some embodiments, the tolerogenic factor is CD47 and the engineered islet cell has increased expression of CD47. In some embodiments, the tolerogenic factor is PD-L1 and the engineered islet cell includes increased expression of PD-LL In some embodiments, the tolerogenic factor is HLA-E and the engineered islet cell includes increased expression of HLA- E. In some embodiments, the tolerogenic factor is HLA-G and the engineered islet includes increased expression of HLA-G. In some embodiments, the tolerogenic factor is expressed as an exogenous polynucleotide or transgene in the genome of the engineered islet cell. In some embodiments, the exogenous polynucleotide or transgene is integrated or inserted into a genome locus of the cells, such as a safe harbor locus. In some embodiments, the genomic locus is an ABO, CCR5, CLYBL, CXCR4, F3, FUT1, HMGB1, KDM5D, LRP1, MICA, MICB, RHD, ROSA26, or SHS231 locus.
[0286] In some aspects, provided are engineered islet cells (e.g. iPSC-derived islet cells) having (1) reduced expression of MHC I and/or MHC II; and (2) a transgene comprising CD47 and a safety switch inserted at a safe harbor locus, wherein the safe harbor locus is selected from
the group consisting of an AAVS1, ABO, CCR5, CLYBL, CXCR4, F3, FUT1, HMGB1, KDM5D, LRP1, MICA, MICB, RHD, ROSA26, and SHS231 locus. In some aspects, provided are modified pluripotent stem cells having (1) reduced expression of MHC I and/or MHC II; and (2) a transgene comprising CD47 and HSVtk flanked by CLYBL homology arms, wherein the transgene is inserted at the CLYBL locus. In some embodiments, the modified pluripotent stem cell has B2M and/or CIITA knockout. In some embodiments, the B2M and/or CIITA knockout occur in both alleles.
[0287] In some embodiments, an engineered islet cell provided herein comprises a safety switch. The introduction of safety switches improves the safety of cell therapies developed using hypoimmunogenic cells (HIP cells, e.g., engineered islet cells). In some embodiments, a feature of the HIP cells described herein is the inducible expression of one or more immune regulatory (immunosuppressive) factors. In some embodiments, an immunosuppressive factor (also referred to herein as “an hypoimmunity factor”) includes, but is not limited to, CD47, CD24, CD200, HLA-G, HLA-E, HLA-C, HLA-E heavy chain, PD-L1, IDO1, CTLA4-Ig, Cl -Inhibitor, IL-10, IL-35, FASL, Serpmb9, CC121, and Mfge8. In certain embodiments, an immunosuppressive factor is CD47. The regulatable or inducible expression of an immunosuppressive factor functions to control an immune response by a recipient subject to an engrafted hypoimmunogenic cell.
[0288] Described herein are methods for the expression of an immunosuppressive factor that requires a mechanism to “turn-off’ expression of the immune regulatory protein in a controlled manner. Also described are engineered islet cells possessing controllable expression of one or more immunosuppressive factors. In some cases, the cells overexpress one or more immunosuppressive factors and can be induced to downregulate expression of the one or more immunosuppressive factors. As such, the cells are no longer hypoimmunogenic and are recognized by the recipient's immune cells for cell death.
[0289] In some embodiments, the hypoimmunity of engineered islet cells that are introduced to a recipient subject is achieved through the overexpression of an immunosuppressive molecule including hypoimmunity factors and complement inhibitors
accompanied with the repression or genetic disruption of the HLA-I and HLA-II loci. These modifications cloak the cell from the recipient immune system's effector cells that are responsible for the clearance of infected, malignant or non-self cells, such as T cells, B cells, NK cells and macrophages. Cloaking of a cell from the immune system allows for existence and persistence of allogeneic cells within the body. Controlled removal of the engineered cells from the body is crucial for patient safety and can be achieved by uncloaking the cells from the immune system. Uncloaking serves as a safety switch and can be achieved through the downregulation of the immunosuppressive molecules or the upregulation of immune signaling molecules. The level of expression of any of the immunosuppressive molecules described can be controlled on the protein level, mRNA level, or DNA level in the cells. Similarly, the level of expression of any of the immune signaling molecules described can be controlled on the protein level, mRNA level, or DNA level in the cells.
[0290] In some embodiments, any of the safety switch methods described (e.g., protein level, RNA level and DNA level safety switches) are used to decrease the level of an immunosuppressive factor in the cells such that the lower level of the immunosuppressive factor is below a threshold level. In some embodiments, the level of the immunosuppressive factor in the cells is decreased by about 10-fold, 9-fold, 8-fold, 7-fold, 6-fold, 5-fold, 4-fold, 3-fold, 2- fold, 1- fold or 0.5-fold below a threshold level of expression. In some embodiments, the level of the immunosuppressive factor in the cells is decreased by about 10-fold to 5-fold, 10-fold to
3-fold, 9- fold to 1-fold, 8-fold to 1-fold, 7-fold to 0.5-fold, 6-fold, to 1-fold, 5-fold to 0.5-fold,
4-fold to 0.5-fold, 3-fold to 0.5-fold, 2-fold to 0.5-fold, or 1-fold to 0.5-fold below a threshold level of expression. In some embodiments, the threshold level of expression of the immunosuppressive factor is established based on the expression of such factor in an induced pluripotent stem cell. In some embodiments, the threshold level of the immunosuppressive factor expression is established based on the expression level of the immunosuppressive factor in a corresponding hypoimmune cell, such as any of the engineered islet cells described herein.
[0291] In some embodiments, transcriptional regulation of immunosuppressive factors through employing inducible promoters provides the ability to turn expression of the switch on
or off at will through the addition or removal of small molecules, such as, but not limited to, doxycycline. Genetic disruption via targeted nuclease activity can eliminate expression of the immunosuppressive factor to uncloak the cells as well. Exemplary safety switches are described in WO2021146627A1, the content of which is herein incorporated by reference in its entirety.
[0292] In some embodiments, any of the above engineered islet cells further have regulated or modulated (e.g. reduced or eliminated) expression of CD142. In some embodiments, the regulated or modulated expression of CD 142 is due to gene editing in which the DNA of the CD 142 gene loci has been edited to delete genomic DNA. In some embodiments, the engineered islet cell has an edit to delete genomic DNA of CD 142 and is thus reduced or eliminated for expression of CD 142. In some embodiments, the CD 142 gene is knocked out in the engineered islet cell. In some embodiments, both alleles of B2M are knocked out.
[0293] In some embodiments, any of the above engineered islet cells further have regulated or modulated (e.g. increased) expression of one or more complement inhibitor. In some embodiments, the one or more complement inhibitors is any one of CD46, CD59 and CD55 or is a combination thereof (e.g. CD46 and CD59 or CD46, CD59 and CD55). In some embodiments, the one or more complement inhibitor is expressed as an exogenous polynucleotide(s) or transgene(s) in the genome of an engineered islet cell. In some embodiments, the exogenous polynucleotide(s) or transgene(s) is integrated or inserted into a genome locus of the cells, such as a safe harbor locus. In some embodiments, the genomic locus is an ABO, CCR5, CLYBL, CXCR4, F3, FUT1, HMGB1, KDM5D, LRP1, MICA, MICB, RHD, ROSA26, or SHS231 locus. In some embodiments, the exogenous polynucleotide or transgene is expressed at the same or a different locus from CD47 and/or from a suicide gene.
[0294] In some embodiments, the methods used to differentiate the engineered islet cells are known by one skilled in the ait. Such methods are described, for example, in W02019018818, US8507274, US10030229, US10190096, US10253298, US10443042, W02016100925, WO2019217493, US7510876, US8216836, US8633024, US8647873, US 10421942, US9404086, US20190359943, US 10358628, US8633024, US8647873,
US9222069, US10465162, US10370645, US9725699, US10253298, US9499795, US9650610, US9062290, US10494609, US20210060083, US8129182, US86O3811, US9328331, US9012218, US9109245, US9982235, US9988604, US10358628, US10138465,
U S20190211309, US10443042, W02020207998A1, US20210230554A1, US20200308548A1, US20190085295A1, US20200002670A1, US20190153394A1, US10487313, US20120135519A1, US20210207099, Pagliuca et al, (Cell 2014), and Hogrebe et al. (2021), all of which are herein incorporated by reference.
[0295] In some embodiments, the process of differentiating pluripotent stem cells into functional pancreatic endocrine cells (i.e., islet cells) in vitro may be viewed in some aspects as progressing through six consecutive stages. In some embodiments, stage 1 refers to the first step in the differentiation process, the differentiation of pluripotent stem cells into cells expressing markers characteristic of definitive endoderm cells. Stage 2 refers to the second step, the differentiation of cells expressing markers characteristic of definitive endoderm cells into cells expressing markers characteristic of gut tube cells. Stage 3 refers to the third step, the differentiation of cells expressing markers characteristic of gut tube cells into cells expressing markers characteristic of early pancreas progenitor cells. Stage 4 refers to the fourth step, the differentiation of cells expressing markers characteristic of early pancreas progenitor cells into cells expressing markers characteristic of pancreatic progenitor cell. Stage 5 refers to the fifth step, the differentiation of cells expressing markers characteristic of pancreatic progenitor cells into cells expressing markers characteristic of pancreatic endoderm cells and/or pancreatic endocrine progenitor cells. It should be appreciated, however, that not all cells in a particular population progress through these stages at the same rate, i.e., some cells may have progressed less, or more, down the differentiation pathway than the majority of cells present in the population. Thus, it is understood that in any step reference to a particular stage may include contacting of the particular cell of a given stage with a compound where cells in the contacted population may include a cell that is partially differentiated from the modified pluripotent stem cell or is a precursor of the cell stage.
[02961 In some embodiments, a definitive endoderm cell is a cell that bears the characteristics of cells arising from the epiblast during gastrulation and which form the gastrointestinal tract and its derivatives. Definitive endoderm cells express at least one of the following markers: FOXA2 (also known as hepatocyte nuclear factor 3 (“HNF30”)), GATA4, SOX17, CXCR4, Brachyury, Cerberus, OTX2, goosecoid, C-Kit, CD99, and MIXL1. Markers characteristic of the definitive endoderm cells include CXCR4, FOXA2 and SOX17. Thus, definitive endoderm cells may be characterized by their expression of CXCR4, FOXA2 and SOX17. In addition, depending on the length of time cells are allowed to remain in Stage 1, an increase in HNF4a may be observed.
[0297] In some embodiments, gut tube cells are cells derived from definitive endoderm that can give rise to all endodermal organs, such as lungs, liver, pancreas, stomach, and intestine. Gut tube cells may be characterized by their substantially increased expression of HNF4a over that expressed by definitive endoderm cells. For example, a ten to forty fold increase in mRNA expression of HNF4a may be observed during Stage 2.
[0298] In some embodiments, early pancreas progenitor cells refer to endoderm cells that give rise to the esophagus, lungs, stomach, liver, pancreas, gall bladder, and a portion of the duodenum. Early pancreatic progenitor cells express at least one of the following markers: PDX1, FOXA2, CDX2, SOX2, and HNF4a. Early pancreatic progenitor cells may be characterized by an increase in expression of PDX1, compared to gut tube cells. For example, greater than fifty percent of the cells in Stage 3 cultures typically express PDX1.
[0299] In some embodiments, pancreatic progenitor cells refer to cells that express at least one of the following markers: PDX1, NKX6.1, HNF6, NGN3, SOX9, PAX4, PAX6, ISL1, gastrin, FOXA2, PTFla, PROXI and HNF4a. Pancreatic progenitor cells may be characterized as positive for the expression of PDX1, NKX6.1, and SOX9.
[0300] In some embodiments, a pancreatic endoderm cell (also sometimes called a pancreatic endocrine progenitor cells) is a cell capable of becoming a pancreatic hormone expressing cell. Pancreatic endoderm cells express at least one of the following markers: NGN3;
NKX2.2; NeuroDl; ISL1; PAX4; PAX6; or ARX. Pancreatic endoderm cells may be characterized by their expression of NKX2.2 and NeuroDl.
2. Methods of Making Induced Pluripotent Stem Cells
[0301] Islet cells as provided herein can be generated, e.g., from induced pluripotent stem cells (iPSCs). In some embodiments, engineered islet cells as provided herein are derived from induced pluripotent stem cells. In some embodiments, iPSCs are derived from PBMCs from a subject.
[0302] Methods for generating mouse and human pluripotent stem cells (generally referred to as miPSCs for murine cells or hiPSCs for human cells) are generally known in the ait. As will be appreciated by those in the art, there are a variety of different methods for the generation of iPCSs. The first induction performed was done from mouse embryonic or adult fibroblasts using the viral introduction of four transcription factors, Oct3/4, Sox2, c-Myc and Klf4; see Takahashi and Yamanaka Cell 126:663-676 (2006), hereby incorporated by reference in its entirety and specifically for the techniques outlined therein. Since then, a number of methods have been developed; see Seki et al, World J. Stem Cells 7(1 ): 1 16-125 (2015) for a review, and Lakshmipathy and Vermuri, editors, Methods in Molecular Biology: Pluripotent Stem Cells, Methods and Protocols, Springer 2013, both of which are hereby expressly incorporated by reference in their entirety, and in particular for the methods for generating hiPSCs (see for example Chapter 3 of the latter reference).
[0303] Generally, iPSCs are generated by the transient expression of one or more reprogramming factors" in the host cell, usually introduced using episomal vectors. Under these conditions, small amounts of the cells are induced to become iPSCs (in general, the efficiency of this step is low, as no selection markers are used). Once the cells are “reprogrammed,” and become pluripotent, they lose the episomal vector(s) and produce the factors using the endogeneous genes.
[0304] The number of reprogramming factors that can be used or are used can vary.
Commonly, when fewer reprogramming factors are used, the efficiency of the transformation of
the cells to a pluripotent state goes down, as well as the “pluripotency,” e.g., fewer reprogramming factors may result in cells that are not fully pluripotent but may only be able to differentiate into fewer cell types.
[0305] In some embodiments, a single reprogramming factor, OCT4, is used. In other embodiments, two reprogramming factors, OCT4 and KLF4, are used. In other embodiments, three reprogramming factors, OCT4, KLF4 and SOX2, are used. In other embodiments, four reprogramming factors, OCT4, KLF4, SOX2 and c-Myc, are used. In other embodiments, 5, 6 or 7 reprogramming factors can be used selected from SOKMNLT; SOX2, OCT4 (POU5F1), KLF4, MYC, NANOG, LIN28, and SV40L T antigen. In general, these reprogramming factor genes are provided on episomal vectors such as are known in the art and commercially available.
[0306] In some embodiments, cells used for transfecting the one or more reprogamming factors are non-pluripotent stem cells. In general, iPSCs are made from non-pluripotent cells such as, but not limited to, PBMCs, blood cells, fibroblasts, etc., by transiently expressing the reprogramming factors as described herein. In some embodiments, the non-pluripotent cells, such as PBMCs, are obtained or isolated from one or more individual subjects or donors prior to reprogamming the cells. In some embodiments, iPSCs are made from a pool of isolated non- pluripotent stems cells, e.g., PBMCs, obtained from one or more (e.g., two or more, three or more, four or more, five or more, ten or more, twenty or more, fifty or more, or one hundred or more) different donor subjects. In some embodiments, the non-pluripotent cells, such as PBMCs, are isolated or obtained from a plurality of different donor subjects (e.g., two or more, three or more, four or more, five or more, ten or more, twenty or more, fifty or more, or one hundred or more), pooled together in a batch, reprogrammed as iPSCs and are engineered in accord with the provided methods. As described herein, however, in preferred embodiments, non-pluripotent cells, such as PBMCs, are obtained or isolated one subject.
[0307] In some embodiments, iPSCs are derived from, such as by transiently transfecting one or more reprogramming factors into cells from a pool of non-pluripotent cells (e.g., PBMCs) from a subject. In some embodiments, non-pluripotent cells (e.g., PBMCs) are harvested from one or a plurality of individuals, and in some instances, the non-pluripotent cells (e.g., PBMCs)
or a pool of non-pluripotent cells (e.g., PBMCs) are cultured in vitro and transfected with one or more reprogramming factors to induce generation of iPSCs. In some embodiments, non- pluripotent cells (e.g., PBMCs) or a pool of non-pluripotent cells (e.g., PBMCs) are engineered or modified in accord with the methods provided herein. In some embodiments, engineered iPSCs or a pool of engineered iPSCs are then subjected to a differentiation process for differentiation into any cells of an organism and tissue.
[0308] Once the engineered iPSCs cells have been generated, they may be assayed for their hypoimmunogenicity and/or retention of pluripotency as is described in W02016183041 and WO2018132783. In some embodiments, hypoimmunogenicity is assayed using a number of techniques as exemplified in Figure 13 and Figure 15 of WO2018132783. These techniques include transplantation into allogeneic hosts and monitoring for hypoimmunogenic pluripotent cell growth (e.g., teratomas) that escape the host immune system. In some instances, hypoimmunogenic pluripotent cell derivatives are transduced to express luciferase and can then followed using bioluminescence imaging. Similarly, the T cell and/or B cell response of the host animal to such cells are tested to confirm that the cells do not cause an immune reaction in the host animal. T cell responses can be assessed by Elispot, ELISA, FACS, PCR, or mass cytometry (CYTOF). B cell responses or antibody responses are assessed using FACS or Luminex. Additionally or alternatively, the cells may be assayed for their ability to avoid innate immune responses, e.g., NK cell killing, as is generally shown in Figures 14 and 15 of WO2018132783.
[0309] In some embodiments, the immunogenicity of the cells is evaluated using T cell immunoassays such as T cell proliferation assays, T cell activation assays, and T cell killing assays recognized by those skilled in the art. In some cases, the T cell proliferation assay includes pretreating the cells with interferon-gamma and coculturing the cells with labelled T cells and assaying the presence of the T cell population (or the proliferating T cell population) after a preselected amount of time. In some cases, the T cell activation assay includes coculturing T cells with the cells outlined herein and determining the expression levels of T cell activation markers in the T cells.
[03101 In vivo assays can be performed to assess the immunogenicity of the cells outlined herein. In some embodiments, the survival and immunogenicity of engineered or modified iPSCs is determined using an allogeneic humanized immunodeficient mouse model. In some instances, the engineered or modified iPSCs are transplanted into an allogeneic humanized NSG-SGM3 mouse and assayed for cell rejection, cell survival, and teratoma formation. In some instances, grafted engineered iPSCs or differentiated cells thereof display long-term survival in the mouse model.
[0311] Additional techniques for determining immunogenicity including hypoimmunogenicity of the cells are described in, for example, Deuse et ah, Nature Biotechnology, 2019, 37, 252-258 and Han et ah, Proc Natl Acad Sci USA, 2019, 116(21), 10441-10446, the disclosures including the figures, figure legends, and description of methods are incorporated herein by reference in their entirety.
[0312] Similarly, the retention of pluripotency is tested in a number of ways. In some embodiments, pluripotency is assayed by the expression of certain pluripotency-specific factors as generally described herein and shown in Figure 29 of WO2018132783. Additionally or alternatively, the pluripotent cells are differentiated into one or more cell types as an indication of pluripotency.
[0313] Once the engineered pluripotent stem cells (engineered iPSCs) have been generated, they can be maintained in an undifferentiated state as is known for maintaining iPSCs. For example, the cells can be cultured on Matrigel using culture media that prevents differentiation and maintains pluripotency. In addition, they can be in culture medium under conditions to maintain pluripotency.
[0314] Any of the iPSCs described herein can be differentiated into any cells of an organism and tissue. In an aspect, provided herein are engineered cells that are differentiated into different cell types from iPSCs for subsequent transplantation into recipient subjects.
Differentiation can be assayed as is known in the art, generally by evaluating the presence of cell-specific markers. As will be appreciated by those in the art, the differentiated engineered
(e.g., hypoimmunogenic) pluripotent cell derivatives can be transplanted using techniques known in the art that depends on both the cell type and the ultimate use of these cells. Exemplary types of differentiated cells and methods for producing the same are described below. In some embodiments, the iPSCs may be differentiated to any type of cell described herein, including any described in Section II.C.3. In some embodiments, the iPSCs are differentiated into beta islet cells. In some embodiments, cells such as non-pluripotent cells (e.g., PBMCs) from a subject are isolated or obtained, generated into iPSCs in which the iPSCs are then engineered to contain modifications (e.g., genetic modifications) described herein and then differentiated into a desired cell type.
3. Methods of making islet cells
[0315] Methods of making islet cells are provided herein. As described herein, the term “islet cells” includes islet cells made from iPSCs (sometimes termed iPSC-derived pseudo-islet cells, pseudo-islet cells, or p-islets).
[0316] Provided herein is a method of generating insulin-producing islet cells comprising: providing a stem cell (e.g. modified stem cell, such as modified iPSC); providing serum-free media; contacting the stem cell with a TGF /Activin agonist or a glycogen synthase kinase 3 (GSK) inhibitor or WNT agonist for an amount of time sufficient to form a definitive endoderm cell; contacting the definitive endoderm cell with a FGFR2b agonist for an amount of time sufficient to form a primitive gut tube cell; contacting the primitive gut tube cell with an RAR agonist, and optionally a rho kinase inhibitor, a Smoothened antagonist, a FGFR2b agonist, a protein kinase C activator, or a BMP type 1 receptor inhibitor for an amount of time sufficient to form an early pancreas progenitor cell; incubating the early pancreas progenitor cell for at least about 3 days and optionally contacting the early pancreas progenitor cell with a rho kinase inhibitor, a TGF- /Activin agonist, a Smoothened antagonist, an FGFR2b agonist, or a RAR agonist for an amount of time sufficient to form a pancreatic progenitor cell; contacting the pancreatic progenitor cell with an Alk5 inhibitor, a gamma secretase inhibitor, a Smoothened antagonist (e.g., SANT1), an Erbbl (EGFR) or Erbb4 agonist, or a RAR agonist for an amount of time sufficient to form an endoderm cell; or resizing cell clusters within about 24 hours and
allowing the endoderm cell to mature for an amount of time in serum-free media sufficient to form an islet cell.
[0317] Provided herein is a method of generating insulin-producing islet cells comprising: providing a stem cell (e.g. modified stem cell, such as modified induced pluripotent stem cell (iPSC), or a stem cell that does not comprise one or more modifications); providing serum-free media; contacting the stem cell with a TGF /Activin agonist and/or a glycogen synthase kinase 3 (GSK) inhibitor and/or WNT agonist for an amount of time sufficient to form a definitive endoderm cell; contacting the definitive endoderm cell with a FGFR2b agonist for an amount of time sufficient to form a primitive gut tube cell; contacting the primitive gut tube cell with an RAR agonist, a rho kinase inhibitor, a Smoothened antagonist, a FGFR2b agonist, a protein kinase C activator, and/or a BMP type 1 receptor inhibitor for an amount of time sufficient to form an early pancreas progenitor cell; incubating the early pancreas progenitor cell for at least about 3 days and optionally contacting the early pancreas progenitor cell with a rho kinase inhibitor, a TGF- /Activin agonist, a Smoothened antagonist, an FGFR2b agonist, and/or a RAR agonist for an amount of time sufficient to form a pancreatic progenitor cell; contacting the pancreatic progenitor cell with an Alk5 inhibitor, a gamma secretase inhibitor, a Smoothened antagonist (e.g., SANT1), an Erbbl (EGFR) and/or Erbb4 agonist, and/or a RAR agonist for an amount of time sufficient to form an endoderm cell; and resizing cell clusters within about 24 hours and allowing the endoderm cell to mature for an amount of time in serum-free media sufficient to form an islet cell.
[0318] In some embodiments, the serum-free media comprises one or more selected from the group consisting of: MCDB131, glucose, NaHCOs, BSA, ITS- X, Glutamax, vitamin C, penicillin-streptomycin, CMRL 10666, FBS, Heparin, NEAA, trace elements A, trace elements B, or ZnS04.
[0319] In some embodiments, the TGF /Activin agonist is Activin A; the glycogen synthase kinase 3 (GSK) inhibitor or the WNT agonist is CHIR; the FGFR2b agonist is KGF; the Smoothened antagonist or hedgehog pathway inhibitor is SANT-1 ; the FGF family member/FGFR2b agonist is KGF; the RAR agonist is RA; the protein kinase 3 activator is
TPPB; the BMP inhibitor is LDN; the rho kinase inhibitor is Y27632; the Alk5 inhibitor/TGF-b receptor inhibitor is Alk5i; the thyroid hormone is T3; or the gamma secretase inhibitor is XXI.
[0320] In some embodiments, the TGF /Activin agonist is Activin A. In certain embodiments, the concentration of Activin A is between 50 ng/ml-150 ng/ml. In certain embodiments, the concentration of Activin A is 50 ng/ml, 55 ng/ml, 60 ng/ml, 65 ng/ml, 70 ng/ml, 75 ng/ml, 80 ng/ml, 85 ng/ml, 90 ng/ml, 95 ng/ml, 100 ng/ml, 105 ng/ml, 110 ng/ml, 115 ng/ml, 120 ng/ml, 125 ng/ml, 130 ng/ml, 135 ng/ml, 140 ng/ml, 145 ng/ml, or 150 ng/ml. In certain embodiments, the concentration of Activin A is between 50 ng/ml-60 ng/ml, 55 ng/ml-65 ng/ml, 60 ng/ml-70 ng/ml, 65 ng/ml-75 ng/ml, 70 ng/ml-80 ng/ml, 75 ng/ml-85 ng/ml, 80 ng/ml- 90 ng/ml, 85 ng/ml-95 ng/ml, 90 ng/ml- 100 ng/ml, 95 ng/ml- 105 ng/ml, 100 ng/ml- 110 ng/ml, 105 ng/ml- 115 ng/ml, 110 ng/ml- 120 ng/ml, 115 ng/ml- 125 ng/ml, 120 ng/ml- 130 ng/ml, 125 ng/ml-135 ng/ml, 130 ng/ml-140 ng/ml, 135 ng/ml-145 ng/ml, or 140ng/ml-150 ng/ml. In a specific embodiment, the concentration of Activin A is 100 ng/ml.
[0321] In some embodiments, the glycogen synthase kinase 3 (GSK) inhibitor or the WNT agonist is CHIR. In certain embodiments, the concentration of CHIR is between 0.5 pM and 5 pM. In certain embodiments, the concentration of the CHIR is 0.5 pM, 1.0 pM, 1.5 pM, 2.0 pM, 2.5 pM, 3.0 pM, 3.5 pM, 4.0 pM, 4.5 pM, or 5.0 pM. In certain embodiments, the concentration of CHIR is between 0.5 pM-1.5 pM, 1.0 pM-2.0 pM, 1.5 pM-2.5 pM, 2.0 pM- 3.0 pM, 2.5 pM-3.5 pM, 3.0 pM-4.0 pM, 3.5 pM-4.5 pM, or 4.0 pM-5.0 pM. In a specific embodiment, the concentration of CHIR is 3.0 pM.
[0322] In certain embodiments, the FGFR2b agonist is KGF. In certain embodiments, the concentration of KGF is between 5 ng/ml-100 ng/ml. In certain embodiments, the concentration of KGF is 5 ng/ml, 10 ng/ml, 15 ng/ml, 20 ng/ml, 25 ng/ml, 30 ng/ml, 35 ng/ml, 40 ng/ml, 45 ng/ml, 50 ng/ml, 55 ng/ml, 60 ng/ml, 65 ng/ml, 70 ng/ml, 75 ng/ml, 80 ng/ml, 85 ng/ml, 90 ng/ml, 95 ng/ml, or 100 ng/ml. In certain embodiments, the concentration of KGF is between 5 ng/ml- 15 ng/ml, 10 ng/ml-20 ng/ml, 15 ng/ml-25 ng/ml, 20 ng/ml-30 ng/ml, 25 ng/ml - 35 ng/ml, 30 ng/ml-40 ng/ml, 35 ng/ml-45 ng/ml, 40 ng/ml-50 ng/ml, 45 ng/ml-55 ng/ml, 50 ng/ml-60 ng/ml, 55 ng/ml-65 ng/ml, 60 ng/ml-70 ng/ml, 65 ng/ml-75 ng/ml, 70 ng/ml-80 ng/ml,
75 ng/ml-85 ng/ml, 80 ng/ml-90 ng/ml, 85 ng/ml-95 ng/ml, 90 ng/ml-100 ng/ml. In a specific embodiment, the concentration of the KGF is 50 ng/ml.
[0323] In certain embodiments, the Smoothened antagonist is SANT-1. In certain embodiments, the concentration of SANT-1 is between 0.05 pM and 0.50 p M. In certain embodiments, the concentration of the SANT-1 is 0.05 pM, 0.10 pM, 0.15 pM, 0.20 pM, 0.25 pM, 0.30 pM, 0.35 pM, 0.40 pM, 0.45 pM, or 0.5 pM. In certain embodiments, the concentration of SANT-1 is between 0.05 pM-0.15 pM, 0.10 pM-0.20 pM, 0.15 pM-0.25 pM, 0.20 pM-0.30 pM, 0.25 pM-0.35 pM, 0.30 pM-0.40 pM, 0.35 pM-0.45 pM, or 0.40 pM-0.50 pM. In a specific embodiment, the concentration of SANT-1 is 0.25 pM.
[0324] In certain embodiments, the RAR agonist is retinoic acid (RA). In certain embodiments, the concentration of RA is between 0.05 pM and 2.5 pM. In certain embodiments, the concentration of RA is 0.05 pM, 0.1 pM, 0.15 pM, 0.2 pM, 0.5 pM, 1.0 pM, 1.5 pM, 2.0 pM, or 2.5 pM. In certain embodiments, the concentration of RA is between 0.005 pM-0.15 pM, 0.10 pM-0.2 pM, 0.15 pM-0.5 pM, 0.2 pM-1.0 pM, 0.5 pM-1.5 pM, 1.0 pM-2.0 pM, or 1.5 pM-2.5 pM. In a specific embodiment, the concentration of RA is 0.10 pM. In a specific embodiment, the concentration of RA is 2.0 pM.
[0325] In some embodiments, the protein kinase C activator is TPPB. In certain embodiments, the concentration of TPPB is between 0.05 pM and 0.50 pM. In certain embodiments, the concentration of the TPPB is 0.05 pM, 0.10 pM, 0.15 pM, 0.20 pM, 0.25 pM, 0.30 pM, 0.35 pM, 0.40 pM, 0.45 pM, or 0.5 pM. In certain embodiments, the concentration of TPPB is between 0.05 pM-0.15 pM, 0.10 pM-0.20 pM, 0.15 pM-0.25 pM, 0.20 pM-0.30 pM, 0.25 pM-0.35 pM, 0.30 pM-0.40 pM, 0.35 pM-0.45 pM, or 0.40 pM-0.50 pM. In a specific embodiment, the concentration of TPPB is 0.20 pM.
[0326] In some embodiments, the BMP type 1 receptor inhibitor is LDN193189. In certain embodiments, the concentration of LDN193189 is between 0.05 pM and 0.50 pM. In certain embodiments, the concentration of the LDN193189 is 0.05 pM, 0.10 pM, 0.15 pM, 0.20 pM, 0.25 pM, 0.30 pM, 0.35 pM, 0.40 pM, 0.45 pM, or 0.5 pM. In certain embodiments, the
concentration of LDN193189 is between 0.05 pM-0.15 pM, 0.10 pM-0.20 pM, 0.15 pM-0.25 pM, 0.20 pM-0.30 pM, 0.25 pM-0.35 pM, 0.30 pM-0.40 pM, 0.35 pM-0.45 pM, or 0.40 pM- 0.50 pM. In a specific embodiment, the concentration of LDN193189 is 0.20 pM.
[0327] In some embodiments, the Alk5 inhibitor is Alk5i. In certain embodiments, the concentration of Alk5i is between 5.0 pM and 15 pM. In certain embodiments, the concentration of Alk5i is 5.0 pM, 6.0 pM, 7.0 pM, 8.0 pM, 9.0 pM, 10.0 pM, 11.0 pM, 12.0 pM, 13.0 pM, 14.0 pM, or 15.0 pM. In certain embodiments, the concentration of Alk5i is between 5.0 pM-7.0 pM, 6.0 pM-8.0 pM, 7.0 pM-9.0 pM, 8.0 pM-10.0 pM, 9.0 pM-11.0 pM, 10.0 pM-12.0 pM, 11.0 pM-13.0 pM, 12.0 pM-14.0 pM, or 13.0 pM-15.0 pM. In a specific embodiment, the concentration of Alk5i is 10.0 pM.
[0328] In certain embodiments, latrunculin A is utilized to chemically depolymerize the actin cytoskeleton. In certain embodiments, the concentration of latrunculin A is 0.5 pM and 1.5 pM. In certain embodiments, the concentration of latrunculin A is 0.5 pM, 0.6 pM, 0.7 pM, 0.8 pM, 0.9 pM, 1.0 pM, 1.1 pM, 1.2 pM, 1.3 pM, 1.4 pM, or 1.5 pM. In certain embodiments, the concentration of latrunculin A is between 0.5 pM-0.7 pM, 0.6 pM-0.8 pM, 0.7 pM-0.9 pM, 0.8 pM-1.0 pM, 0.9 pM-1.1 pM, 1.0 pM-1.2 pM, 1.1 pM-1.3 pM, 1.2 pM-1.4 pM, or 1.3 pM-1.5 pM. In a specific embodiment, the concentration of latrunculin A is 1.0 pM.
[0329] In certain embodiments, the thyroid hormone is T3. In certain embodiments, the concentration of T3 is between 0.1 pM and 2 pM. In certain embodiments, the concentration of T3 is 0.1 pM, 0.2 pM, 0.3 pM, 0.4 pM, 0.5 pM, 0.6 pM, 0.7 pM, 0.8 pM, 0.9 pM, 1.0 pM, 1.1 pM, 1.2 pM, 1.3 pM, 1.4 pM, 1.5 pM, 1.6 pM, 1.7 pM, 1.8 pM, 1.9 pM, or 2.0 pM. In certain embodiments, the concentration of T3 is between 0.1 pM-0.3 pM, 0.2 pM-0.4 pM, 0.3 pM-0.5 pM, 0.4 pM-0.6 pM, 0.5 pM-0.7 pM, 0.6 pM-0.8 pM, 0.7 pM-0.9 pM, 0.8 pM-1.0 pM, 0.9 pM-1.1 pM, 1.0 pM-1.2 pM, 1.1 pM-1.3 pM, 1.2 pM-1.4 pM, 1.3 pM-1.5 pM, 1.4 pM-1.6 pM, 1.5 pM-1.7 pM, 1.6 pM-1.8 pM, 1.7 pM-1.9 pM, or 1.8 pM -2.0 pM. In a specific embodiment, the concentration of T3 is 1.0 pM.
[03301 In certain embodiments, the gamma secretase inhibitor is XXI. In certain embodiments, the concentration of XXI is between 0.1 pM and 2 pM. In certain embodiments, the concentration of XXI is 0.1 pM, 0.2 pM, 0.3 pM, 0.4 pM, 0.5 pM, 0.6 pM, 0.7 pM, 0.8 pM, 0.9 pM, 1.0 pM, 1.1 pM, 1.2 pM, 1.3 pM, 1.4 pM, 1.5 pM, 1.6 pM, 1.7 pM 1.8 pM, 1.9 pM, or 2.0 pM. In certain embodiments, the concentration of XXI is between 0.1 pM-0.3 pM, 0.2 pM- 0.4 pM, 0.3 pM-0.5 pM, 0.4 pM-0.6 pM, 0.5 pM-0.7 pM, 0.6 pM-0.8 pM, 0.7 pM-0.9 pM, 0.8 pM-l .O pM, 0.9 pM-l .l pM, 1.0 pM-l .2 pM, 1.1 pM-1.3 pM, 1.2 pM-1.4 pM, 1.3 pM-1.5 pM, 1.4 pM-1.6 pM, 1.5 pM-1.7 pM, 1.6 pM-1.8 pM, 1.7 pM-1.9 pM, or 1.8 pM-2.0 pM. In a specific embodiment, the concentration of XXI is 1.0 pM.
[0331] In certain embodiments, the methods herein detail a differentiation protocol for generating highly functional SC- P cells. The methods provided herein comprise six stages that attempt to recreate phases of pancreatic organogenesis by activating and repressing specific developmental pathways with growth factors and small molecules in serum-free medium. In certain embodiments, to propagate and expand cells for SC- -cell differentiation, hPSCs are seeded onto Matrigel-coated TCP plates at a density of 0.8 x 105 cells/cm2 and cultured in medium.
[0332] In certain embodiments, the methods provided herein comprise six stages of stem cell differentiation. In certain embodiments, Stage 1 comprises incubating a HPSC of Stage 0 in media comprising Activin A and CHIR for about 24 hours followed by about 3 days of incubating the cells in media comprising Activin A in the absence of CHIR. In certain embodiments, Stage 2 comprises incubating the Stage 1 cells for 2 days in media comprising KGF. In some embodiments, the CHIR is CH1R99021.
[0333] In certain embodiments, Stage 3 comprises incubating Stage 2 cells for 2 days in media comprising KGF, LDN193189, TPPB, RA (high), and SANT1. In certain embodiments, Stage 4 comprises incubating Stage 3 cells for about 4 days in media comprising KGF, LDN193189, TPPB, RA (low), and SANT1. In certain embodiments, Stage 5 comprises incubating the Stage 4 cells in media comprising XXI, Alk5i, T3, SANT1, and RA for 7 days. Additionally, latrunculin A is added to the media for about the first 24 hours of incubation. In
certain embodiments, Stage 6 comprises incubating the cells in an enriched serum-free medium which allows the SC-P cells the time needed to mature before they become glucose responsive. The methods provided herein generate islet cells that function better (undergoing glucose- stimulated insulin secretion) than cells in the published literature (Pagliuca et al. Cell 2014).
[0334] In some embodiments, the amount of time sufficient to form a definitive endoderm cell, a primitive gut tube cell, an early pancreas progenitor cell, a pancreatic progenitor cell, an endoderm cell, or an islet cell is between about 1 day and about 15 days.
[0335] As described herein, islet cells can be useful as a cellular therapy for diabetes. The presently disclosed method enhances differentiation of human pluripotent stem cells to insulinproducing islet cells. This process is modified from a previously described 6-step differentiation protocol published by Pagliuca et al. Cell 2014. Using the methods disclosed herein, cells that can respond to glucose appropriately to near islet-like levels have been generated, demonstrating both a first phase and second phase response. In order to achieve the above modulation, the following was performed: (1) shorten stage 3 to 1 day; (2) allow for TGFbeta signaling in stage 6 by removal of Alk5 inhibitor II (3) remove T3 from stage 6; (4) perform stage 6 in a serum-free basal media; and (5) break apart and reaggregate clusters at the beginning of stage 6.
4. Hypoimmunogenic Islet Cell Precursors
[0336] In some embodiments, a cell to be engineered or modified (prior to differentiation into an islet cell) is a stem or progenitor cell that is capable of being differentiated (e.g., the stem cell is totipotent, pluripotent, or multipotent). In some embodiments, a cell is isolated from neonatal tissue. In some embodiments, a cell is an induced pluripotent stem cell derived from somatic cells (e.g., skin or blood cells) and reprogrammed into an embryonic-like pluripotent state. In some embodiments, an induced pluripotent stem cell is derived from a fibroblast. In some embodiments, cells that are modified as provided herein are pluripotent stems cells or are cells differentiated from pluripotent stem cells. A cell may be a vertebrate cell, for example, a mammalian cell, such as a human cell or a mouse cell. A cell may also be a vertebrate stem cell, for example, a mammalian stem cell, such as a human stem cell or a mouse stem cell.
Preferably, a cell or stem cell is amenable to modification. Preferably, a cell or stem cell, or a cell derived from such a stem cell, has or is believed to have therapeutic value, such that a cell or stem cell or a cell derived or differentiated from such stem cell may be used to treat a disease, disorder, defect or injury in a subject in need of treatment for same.
[0337] In some embodiments, cells that are modified as provided herein are modified pluripotent stem cells (e.g., modified iPSCs). The generation of mammalian (e.g., mouse and human) pluripotent stem cells (generally referred to as iPSCs; miPSCs for murine cells or hiPSCs for human cells) is generally known in the art. As will be appreciated by those in the art, there are a variety of different methods for the generation of iPCSs. The original induction was done from mouse embryonic or adult fibroblasts using the viral introduction of four transcription factors, Oct3/4, Sox2, c-Myc and Klf4; see Takahashi and Yamanaka Cell 126:663-676 (2006), hereby incorporated by reference in its entirety and specifically for the techniques outlined therein. Since then, a number of methods have been developed; see Seki et al, World J. Stem Cells 7(1): 116-125 (2015) for a review, and Lakshmipathy and Vermuri, editors, Methods in Molecular Biology: Pluripotent Stem Cells, Methods and Protocols, Springer 2013, both of which are hereby expressly incorporated by reference in their entirety, and in particular for the methods for generating hiPSCs (see for example Chapter 3 of the latter reference).
[0338] Generally, iPSCs are generated by the transient expression of one or more reprogramming factors" in the host cell, usually introduced using episomal vectors. Under these conditions, small amounts of the cells are induced to become iPSCs (in general, the efficiency of this step is low, as no selection markers are used). Without wishing to be bound by theory, it is believed that once the cells are "reprogrammed", and become pluripotent, they lose the episomal vector(s) and produce the factors using the endogenous genes.
[0339] As is also appreciated by those of skill in the art, the number of reprogramming factors that can be used or are used can vary. Commonly, when fewer reprogramming factors are used, the efficiency of the transformation of the cells to a pluripotent state goes down, as well as the "pluripotency", e.g., fewer reprogramming factors may result in cells that are not fully pluripotent but may only be able to differentiate into fewer cell types.
[03401 In some embodiments, a single reprogramming factor, OCT4, is used. In other embodiments, two reprogramming factors, OCT4 and KLF4, are used. In other embodiments, three reprogramming factors, OCT4, KLF4 and SOX2, are used. In other embodiments, four reprogramming factors, OCT4, K.LF4, SOX2 and c-Myc, are used. In other embodiments, 5, 6 or 7 reprogramming factors can be used selected from SOKMNLT; SOX2, OCT4 (POU5F1), KLF4, MYC, NANOG, LIN28, and SV40L T antigen. In general, these reprogramming factor genes are provided on episomal vectors such as are known in the art and commercially available.
[0341] In some embodiments, hosts cells used for transfecting the one or more reprogramming factors are non-pluripotent stem cells. In general, as is known in the art, iPSCs are made from non-pluripotent cells such as, but not limited to, blood cells, fibroblasts, etc., by transiently expressing the reprogramming factors as described herein. In some embodiments, the non-pluripotent cells, such as fibroblasts, are obtained or isolated from one or more individual subjects or donors prior to reprogramming the cells. In some embodiments, iPSCs are made from a pool of isolated non-pluripotent stems cells, e.g., fibroblasts, obtained from one or more (e.g. two or more, three or more, four or more, five or more, ten or more, twenty or more, fifty or more, or one hundred or more) different donor subjects. In some embodiments, the non- pluripotent cells, such as fibroblasts, are isolated or obtained from a plurality of different donor subjects (e.g., two or more, three or more, four or more, five or more, ten or more, twenty or more, fifty or more, or one hundred or more), pooled together in a batch, reprogrammed as iPSCs, and are modified in accord with the provided methods.
[0342] In some embodiments, the iPSCs are derived from, such as by transiently transfecting one or more reprogramming factors into cells from a pool of non-pluripotent cells (e.g., fibroblasts) from one or more donor subjects that are different than the recipient subject (e.g., the patient administered the cells). The non-pluripotent cells (e.g., fibroblasts) to be induced to iPSCs can be obtained from 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, 20, 50, 100 or more donor subjects and pooled together. The non-pluripotent cells (e.g., fibroblasts) can be obtained from 1 or more, 2 or more, 3 or more, 4 or more, 5 or more, 6 or more, 7 or more, 8 or more, 9 or more, 10, or more 20 or more, 50 or more, or 100 or more donor subjects and pooled together. In some
embodiments, the non-pluripotent cells (e.g., fibroblasts) arc harvested from one or a plurality of individuals, and in some instances, the non-pluripotent cells (e.g., fibroblasts) or the pool of non- pluripotent cells (e.g., fibroblasts) are cultured in vitro and transfected with one or more reprogramming factors to induce generation of iPSCs. In some embodiments, the non- pluripotent cells (e.g., fibroblasts) or the pool of non-pluripotent cells (e.g., fibroblasts) are modified in accord with the methods provided herein. In some embodiments, the modified iPSCs or a pool of modified iPSCs are then subjected to a differentiation process for differentiation into any cells of an organism and tissue.
[0343] Once the modified iPSCs cells have been generated, they may be assayed for their hypoimmunogenicity and/or retention of pluripotency as is described in WO2016183041 and WO2018132783. In some embodiments, hypoimmunogenicity is assayed using a number of techniques as exemplified in Figure 13 and Figure 15 of WO2018132783. These techniques include transplantation into allogeneic hosts and monitoring for hypoimmunogenic pluripotent cell growth (e.g., teratomas) that escape the host immune system. In some instances, hypoimmunogenic pluripotent cell derivatives are transduced to express luciferase and can then followed using bioluminescence imaging. Similarly, the T cell and/or B cell response of the host animal to such cells are tested to confirm that the cells do not cause an immune reaction in the host animal. T cell responses can be assessed by Elispot, ELISA, FACS, PCR, or mass cytometry (CYTOF). B cell responses or antibody responses are assessed using FACS or Luminex. Additionally or alternatively, the cells may be assayed for their ability to avoid innate immune responses, e.g., NK cell killing, as is generally shown in Figures 14 and 15 of WO2018132783.
[0344] In some embodiments, the immunogenicity of the cells is evaluated using T cell immunoassays such as T cell proliferation assays, T cell activation assays, and T cell killing assays recognized by those skilled in the art. In some cases, the T cell proliferation assay includes pretreating the cells with interferon-gamma and co-culturing the cells with labelled T cells and assaying the presence of the T cell population (or the proliferating T cell population) after a preselected amount of time. In some cases, the T cell activation assay includes co-
culturing T cells with the cells outlined herein and determining the expression levels of T cell activation markers in the T cells.
[0345] In vivo assays can be performed to assess the immunogenicity of the cells outlined herein. In some embodiments, the survival and immunogenicity of modified iPSCs is determined using an allogeneic humanized immunodeficient mouse model. In some instances, the modified iPSCs are transplanted into an allogeneic humanized NSG-SGM3 mouse and assayed for cell rejection, cell survival, and teratoma formation. In some instances, grafted modified iPSCs or differentiated cells thereof display long-term survival in the mouse model.
[0346] Additional techniques for determining immunogenicity including hypoimmunogenicity of the cells are described in, for example, Deuse et al., Nature Biotechnology, 2019, 37, 252-258 and Han et al., Proc Natl Acad Sci USA, 2019, 116(21), 10441-10446, the disclosures including the figures, figure legends, and description of methods are incorporated herein by reference in their entirety.
[0347] Similarly, the retention of pluripotency may be tested in a number of ways. In one embodiment, pluripotency is assayed by the expression of certain pluripotency- specific factors as generally described herein and shown in Figure 29 of WO2018132783. Additionally or alternatively, the pluripotent cells are differentiated into one or more cell types as an indication of pluripotency.
[0348] Once modified pluripotent stem cells (modified iPSCs) have been generated, they can be maintained in an undifferentiated state as is known for maintaining iPSCs. For example, the cells can be cultured on Matrigel using culture media that prevents differentiation and maintains pluripotency. In addition, they can be in culture medium under conditions to maintain pluripotency.
[0349] Any of the pluripotent stem cells described herein can be differentiated into any cells of an organism and tissue. In an aspect, provided herein are modified cells that are differentiated into different cell types from iPSCs for subsequent transplantation into recipient subjects. Differentiation can be assayed as is known in the art, generally by evaluating the
presence of cell-specific markers. As will be appreciated by those in the art, the differentiated modified (e.g., hypoimmunogenic) pluripotent cell derivatives can be transplanted using techniques known in the ail that depends on both the cell type and the ultimate use of these cells. Exemplary types of differentiated cells and methods for producing the same are described above and below. In some embodiments, iPSCs may be differentiated to any type of cell described herein. In some embodiments, iPSCs are differentiated into islet cells. In some embodiments, host cells such as non-pluripotent cells (e.g., fibroblasts) from an individual donor or a pool of individual donors are isolated or obtained, generated into iPSCs in which the iPSCs are then modified to contain modifications (e.g., genetic modifications) described herein and then differentiated into a desired cell type.
[0350] In some embodiments, cells as provided herein are islet cells derived from modified iPSCs that contain modifications (e.g., genetic modifications) described herein and that are differentiated into islet cells. As will be appreciated by those in the ail, the methods for differentiation depend on the desired cell type using known techniques. In some embodiments, cells differentiated into various islet cells may be used for subsequent transplantation or engraftment into subjects (e.g., recipients). In some embodiments, pancreatic islet cells are derived from the modified pluripotent cells described herein. Useful methods for differentiating pluripotent stem cells into islet cells are described, for example, in U.S. Patent No. 9,683,215; U.S. Patent No. 9,157,062; U.S. Patent No. 8,927,280; U.S. Patent Pub. No. 2021/0207099;
Hogrebe et al., “Targeting the cytoskeleton to direct pancreatic differentiation of human pluripotent stem cells,” Nat. BiotechnoL, 2020, 38:460-470; and Hogrebe et al., “Generation of insulin-producing pancreatic beta cells from multiple human stem cell lines,” Nat. Protoc., 2021, the contents of which are herein incorporated by reference in their entirety,
[0351] In some embodiments, modified pluripotent cells described herein are differentiated into islet-like cells or islet organoids for transplantation to address T1D. Cell systems are a promising way to address T1DM, see, e.g., Ellis et al, Nat Rev Gastroenterol Hepatol. 2017 Oct;14(10):612-628, incorporated herein by reference. Additionally, Pagliuca et al. (Cell, 2014, 159(2):428-39) reports on the successful differentiation of islet cells from
hiPSCs, the contents incorporated herein by reference in its entirety and in particular for the methods and reagents outlined there for the large-scale production of functional human islet cells from human pluripotent stem cells). Furthermore, Vegas et al. shows the production of human islet cells from human pluripotent stem cells followed by encapsulation to avoid immune rejection by the host; Vegas et al., Nat Med, 2016, 22(3) :306- 11 , incorporated herein by reference in its entirety and in particular for the methods and reagents outlined there for the large-scale production of functional human islet cells from human pluripotent stem cells.
[0352] In some embodiments, the method of producing a population of modified pancreatic islet cells from a population of modified pluripotent cells by in vitro differentiation comprises: (a) culturing the population of modified iPSCs in a first culture medium comprising one or more factors selected from the group consisting insulin-like growth factor, transforming growth factor, FGF, EGF, HGF, SHH, VEGF, transforming growth factor-b superfamily, BMP2, BMP7, a GSK inhibitor, an ALK inhibitor, a BMP type 1 receptor inhibitor, and retinoic acid to produce a population of immature pancreatic islet cells; and (b) culturing the population of immature pancreatic islet cells in a second culture medium that is different than the first culture medium to produce a population of modified pancreatic islet cells. In some embodiments, the GSK inhibitor is CHIR-99021, a derivative thereof, or a variant thereof. In some instances, the GSK inhibitor is at a concentration ranging from about 2 mM to about 10 mM. In some embodiments, the ALK inhibitor is SB-431542, a derivative thereof, or a variant thereof. In some instances, the ALK inhibitor is at a concentration ranging from about 1 pM to about 10 pM. In some embodiments, the first culture medium and/or second culture medium are absent of animal serum.
[0353] Differentiation is assayed as is known in the art, generally by evaluating the presence of islet cell associated or specific markers, including but not limited to, insulin. Differentiation can also be measured functionally, such as measuring glucose metabolism, see generally Muraro et al., Cell Syst. 2016 Oct 26; 3(4): 385-394.e3, hereby incorporated by reference in its entirety, and specifically for the biomarkers outlined there. Once islet cells are generated, they can be transplanted (either as a cell suspension, cell clusters, or within a
permeable or semipermeable device or gel matrix as discussed herein) into the portal vein/liver, the omentum, the gastrointestinal mucosa, the bone marrow, a muscle, or subcutaneous pouches.
[0354] Additional descriptions of pancreatic islet cells including for use in the present technology are found in W02020/018615, the disclosure is herein incorporated by reference in its entirety.
[0355] In some embodiments, a population of modified islet cells, such as endothelial cells differentiated from iPSCs derived from one or more individual donors (e.g., healthy donors), are maintained in culture, in some cases expanded, prior to administration. In certain embodiments, the population of engineered islet cells are cryopreserved prior to administration.
[0356] Exemplary pancreatic islet cell types include, but are not limited to, pancreatic islet progenitor cell, immature pancreatic islet cell, mature pancreatic islet cell, and the like. In some embodiments, pancreatic cells described herein are administered to a subject to treat diabetes.
[0357] In some embodiments, pancreatic islet cells modified as disclosed herein, such as islet cells differentiated from iPSCs derived from one or more individual donors (e.g., healthy donors), secrete insulin. In some embodiments, a pancreatic islet cell exhibits at least two characteristics of an endogenous pancreatic islet cell, for example, but not limited to, secretion of insulin in response to glucose, and expression of beta cell markers.
[0358] Exemplary beta cell markers or beta cell progenitor markers include, but are not limited to, c-peptide, Pdxl, glucose transporter 2 (Glut2), HNF6, VEGF, glucokinase (GCK), prohormone convertase (PC 1/3), Cdcpl, NeuroD, Ngn3, Nkx2.2, Nkx6.1, Nkx6.2, Pax4, Pax6, Ptfla, Isll, Sox9, Soxl7, and FoxA2.
[0359] In some embodiments, pancreatic islet cells, such as islet cells differentiated from iPSCs derived from one or more individual donors (e.g., healthy donors), produce insulin in response to an increase in glucose. In various embodiments, pancreatic islet cells secrete insulin in response to an increase in glucose. In some embodiments, islet cells have a distinct
morphology such as a cobblestone cell morphology and/or a diameter of about 17 pm to about 25 pm. a. Inactivation or Disruption of Target Genes
[0360] In some embodiments, provided modified pluripotent stem cells comprise a modification (e.g., genetic modifications) of one or more target polynucleotide or protein sequences (also interchangeably referred to as a target gene) that regulate (e.g., reduce or eliminate) the expression of either MHC class I molecules, MHC class II molecules, or MHC class I and MHC class II molecules. In some embodiments, a cell to be modified is an unmodified cell that has not previously been introduced with the one or more modifications. In some embodiments, a genetic editing system is used to modify one or more target polynucleotide sequences that regulate (e.g., reduce or eliminate) the expression of either MHC class I molecules, MHC class II molecules, or MHC class I and MHC class II molecules. In certain embodiments, the genome of the cell has been altered to reduce or delete components required or involved in facilitating HLA expression, such as expression of MHC class I and/or MHC class II molecules on the surface of the cell. For instance, in some embodiments, expression of a beta-2- microgloublin (B2M), a component of MHC class I molecules, is reduced or eliminated in the cell, thereby reducing or elimination the protein expression (e.g., cell surface expression) of MHC class I by the modified pluripotent stem cells.
[0361] In some embodiments, any of the described modifications in the modified pluripotent stem cells that regulate (e.g., reduce or eliminate) expression of one or more target polynucleotide or protein in the modified pluripotent stem cells may be combined with one or more modifications to overexpress a polynucleotide (e.g., tolerogenic factor, such as CD47).
[0362] In some embodiments, reduction of MHC class I and/or MHC class II expression can be accomplished, for example, by one or more of the following: (1) directly targeting the MHC class I genes such as the polymorphic HLA alleles (HLA- A, HLA-B, HLA -C) and/or the MHC class II genes such as HLA-DP, HLA-DQ, and/or HLA-DR; (2) removal of B2M, which will reduce surface trafficking of all MHC class I molecules; and/or (3) deletion of one or more
components of the MHC enhanceosomes, such as LRC5, RFX-5, RFXANK, RFXAP, IRF1, NF- Y (including NFY-A, NFY-B, NFY-C), and CIITA that are critical for HLA expression. In some embodiments, reduction of MHC class II also may be accomplished by reducing expression, such as by knocking out the gene encoding CD74 in a cell, which is involved in the formation and transport of MHC class II.
[0363] In certain embodiments, HLA expression is interfered with. In some embodiments, HLA expression is interfered with by targeting individual HLAs (e.g., knocking out expression of one or more HLA class I molecules such as HLA-A, HLA-B and/or HLA-C and/or knocking out expression of one or more HLA class I molecules such as HLA-DP, HLA- DQ, and/or HLA-DR), targeting transcriptional regulators of HLA expression (e.g., knocking out expression of NLRC5, CIITA, RFX5, RFXAP, RFXANK, NFY-A, NFY-B, NFY-C and/or IRF- 1), blocking surface trafficking of MHC class I molecules (e.g., knocking out expression of B2M and/or TAPI), and/or targeting with HLA-Razor (see, e.g., W02016183041). In some embodiments, reduction of HLA class II also may be accomplished by reducing expression, such as by knocking out, the gene encoding CD74 in a human cell, which is involved in the formation and transport of HLA class II molecules.
[0364] In certain aspects, the modified pluripotent stem cells disclosed herein do not express one or more human leukocyte antigens corresponding to MHC class I (e.g., HLA-A, HLA-B and/or HLA-C) and/or MHC class II (e.g., HLA-DP, HLA-DQ, and/or HLA-DR) and are thus characterized as being hypoimmunogenic. For example, in certain aspects, the modified pluripotent stem cells disclosed herein have been modified such that the cells, including any stem cell or a differentiated stem cell prepared therefrom, do not express or exhibit reduced expression of one or more of the following MHC class I molecules: HLA-A, HLA-B and HLA-C. In some embodiments, one or more of HLA-A, HLA-B and HLA-C may be "knocked-out" of a cell. A cell that has a knocked-out HLA-A gene, HLA-B gene, and/or HLA-C gene may exhibit reduced or eliminated expression of each knocked-out gene. In some aspects, the modified pluripotent stem cells disclosed herein have been modified such that the cells, including any stem cell or a differentiated stem cell prepared therefrom, do not express or exhibit reduced expression of one
Ill
or more of the following MHC class II molecules: HLA-DP, HLA-DQ, and HLA-DR. In some embodiments, one or more of HLA-DP, HLA-DQ, and HLA-DR may be "knocked-out" of a cell. A cell that has a knocked-out HLA-DP gene, HLA-DQ gene and/or HLA-DR gene may exhibit reduced or eliminated expression of each knocked-out gene.
[0365] In certain embodiments, the expression of MHC class I molecules and/or MHC class II molecules is modulated by targeting and deleting a contiguous stretch of genomic DNA, thereby reducing or eliminating expression of a target gene selected from the group consisting of B2M, CIITA, and NLRC5. In some embodiments, MHC class I molecules can alternatively or additionally be modulated by reducing or eliminating expression of TAPI . In some embodiments, MHC class II molecules can alternatively or additionally be modulated by reducing or eliminating expression of CD74.
[0366] In some embodiments, the provided modified pluripotent stem cells comprise a modification of one or more target polynucleotide sequence that regulate MHC class I. Exemplary methods for reducing expression of MHC class I are described in sections below. In some embodiments, the targeted polynucleotide sequence is one or both of B2M and NLRC5. In some embodiments, the cell comprises a genetic editing modification (e.g., an indel) to the B2M gene. In some embodiments, the cell comprises a genetic editing modification (e.g., an indel) to the NLRC5 gene. In some embodiments, the cell comprises a genetic editing modification (e.g., an indel) to the TAPI gene. In some embodiments, the cell comprises genetic editing modifications (e.g., indels) to the B2M and CIITA genes.
[0367] In some embodiments, a modification that reduces expression of an MHC class I molecule is a modification that reduces expression of B2M. In some embodiments, the modification that reduces B2M expression reduces B2M mRNA expression. In some embodiments, the reduced mRNA expression of B2M is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification. In some embodiments, the mRNA expression of B2M is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the mRNA expression of B2M is reduced by up to about 100%, such as reduced
by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the mRNA expression of B2M is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of B2M is eliminated (e.g., 0% expression of B2M mRNA). In some embodiments, the modification that reduces B2M mRNA expression eliminates B2M gene activity.
[0368] In some embodiments, the modification that reduces B2M expression reduces B2M protein expression. In some embodiments, the reduced protein expression of B2M is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification. In some embodiments, the protein expression of B2M is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the protein expression of B2M is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the protein expression of B2M is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the protein expression of B2M is eliminated (e.g., no detectable expression of B2M protein). In some embodiments, the modification that reduces B2M protein expression eliminates B2M gene activity.
[0369] In some embodiments, the modification that reduces B2M expression comprises inactivation or disruption of the B2M gene. In some embodiments, the modification that reduces B2M expression comprises inactivation or disruption of one allele of the B2M gene. In some embodiments, the modification that reduces B2M expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the B2M gene.
[0370] In some embodiments, the modification comprises inactivation or disruption of one or more B2M coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all B2M coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the B2M gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the B2M gene. In some embodiments, the modification is a deletion of genomic DNA of the
B2M gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the B2M gene. In some embodiments, the B2M gene is knocked out.
[0371] In some embodiments, a modification that reduces expression of an MHC class I molecule is a modification that reduces expression of NLRC5. In some embodiments, decreased or eliminated expression of NLRC5 reduces or eliminates expression of one or more of the following MHC I molecules HLA-A, HLA-B, and HLA-C. In some embodiments, the modification that reduces NLRC5 expression reduces NLRC5 mRNA expression. In some embodiments, the reduced mRNA expression of NLRC5 is relative to an unmodified or wildtype cell of the same cell type that does not comprise the modification. In some embodiments, the mRNA expression of NLRC5 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the mRNA expression of NLRC5 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the mRNA expression of NLRC5 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of NLRC5 is eliminated (e.g., 0% expression of NLRC5 mRNA). In some embodiments, the modification that reduces NLRC5 mRNA expression eliminates NLRC5 gene activity.
[0372] In some embodiments, the modification that reduces NLRC5 expression reduces NLRC5 protein expression. In some embodiments, the reduced protein expression of NLRC5 is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification. In some embodiments, the protein expression of NLRC5 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the protein expression of NLRC5 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the protein expression of NLRC5 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the protein expression of NLRC5 is eliminated (e.g., no detectable expression of
NLRC5 protein). In some embodiments, the modification that reduces NLRC5 protein expression eliminates NLRC5 gene activity.
[0373] In some embodiments, the modification that reduces NLRC5 expression comprises inactivation or disruption of the NLRC5 gene. In some embodiments, the modification that reduces NLCR5 expression comprises inactivation or disruption of one allele of the NLRC5 gene. In some embodiments, the modification that reduces NLRC5 expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the NLRC5 gene.
[0374] In some embodiments, the modification comprises inactivation or disruption of one or more NLRC5 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all NLRC5 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the NLRC5 gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the NLRC5 gene. In some embodiments, the modification is a deletion of genomic DNA of the NLRC5 gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the NLRC5 gene. In some embodiments, the NLRC5 gene is knocked out.
[0375] In some embodiments, a modification that reduces expression of an MHC class I molecule is a modification that reduces expression of TAPI. In some embodiments, decreased or eliminated expression of TAPI reduces or eliminates expression of one or more of the following MHC I molecules - HLA-A, HLA-B, and HLA-C. In some embodiments, the modification that reduces TAPI expression reduces TAPI mRNA expression. In some embodiments, the reduced mRNA expression of TAPI is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification. In some embodiments, the mRNA expression of TAPI is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the mRNA expression of TAPI is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the mRNA expression of TAPI is reduced by any of about 5%, 10%, 20%, 30%,
40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of
TAPI is eliminated (e.g., 0% expression of TAPI mRNA). In some embodiments, the modification that reduces TAPI mRNA expression eliminates TAPI gene activity.
[0376] In some embodiments, the modification that reduces TAPI expression reduces TAPI protein expression. In some embodiments, the reduced protein expression of TAPI is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification. In some embodiments, the protein expression of TAPI is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the protein expression of TAPI is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the protein expression of TAPI is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the protein expression of TAPI is eliminated (e.g., no detectable expression of TAPI protein). In some embodiments, the modification that reduces TAPI protein expression eliminates TAPI gene activity.
[0377] In some embodiments, the modification that reduces TAPI expression comprises inactivation or disruption of the TAPI gene. In some embodiments, the modification that reduces TAPI expression comprises inactivation or disruption of one allele of the TAPI gene. In some embodiments, the modification that reduces TAPI expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the TAPI gene.
[0378] In some embodiments, the modification comprises inactivation or disruption of one or more TAPI coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all TAPI coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the TAPI gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the TAPI gene. In some embodiments, the modification is a deletion of genomic DNA of the TAPI gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the TAPI gene. In some embodiments, the TAPI gene is knocked out.
[03791 In some embodiments, the provided modified pluripotent stem cells comprise a modification of one or more target polynucleotide sequence that regulate MHC class II molecule expression. Exemplary methods for reducing expression of MHC class II are described in sections below. In some embodiments, the cell comprises a genetic editing modification to the CIITA gene. In some embodiments, the cell comprises a genetic editing modification to the CD74 gene.
[0380] In some embodiments, a modification that reduces expression of an MHC class II molecule is a modification that reduces expression of CIITA. In some embodiments, the modification that reduces CIITA expression reduces CIITA mRNA expression. In some embodiments, the reduced mRNA expression of CIITA is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification. In some embodiments, the mRNA expression of CIITA is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the mRNA expression of CIITA is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the mRNA expression of CIITA is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of CIITA is eliminated (e.g., 0% expression of CIITA mRNA). In some embodiments, the modification that reduces CIITA mRNA expression eliminates CIITA gene activity.
[0381] In some embodiments, the modification that reduces CIITA expression reduces CIITA protein expression. In some embodiments, the reduced protein expression of CIITA is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification. In some embodiments, the protein expression of CIITA is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the protein expression of CIITA is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the protein expression of CIITA is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some
embodiments, the protein expression of CIITA is eliminated (e.g., 0% expression of CIITA protein). In some embodiments, the modification that reduces CIITA protein expression eliminates CIITA gene activity.
[0382] In some embodiments, the modification that reduces CIITA expression comprises inactivation or disruption of the CIITA gene. In some embodiments, the modification that reduces CIITA expression comprises inactivation or disruption of one allele of the CIITA gene. In some embodiments, the modification that reduces CIITA expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the CIITA gene.
[0383] In some embodiments, the modification comprises inactivation or disruption of one or more CIITA coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all CIITA coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the CIITA gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the CIITA gene. In some embodiments, the modification is a deletion of genomic DNA of the CIITA gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the CIITA gene. In some embodiments, the CIITA gene is knocked out.
[0384] In some embodiments, a modification that reduces expression of an MHC class II molecule is a modification that reduces expression of CD74. In some embodiments, the modification that reduces CD74 expression reduces CD74 mRNA expression. In some embodiments, the reduced mRNA expression of CD74 is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification. In some embodiments, the mRNA expression of CD74 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the mRNA expression of CD74 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the mRNA expression of CD74 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of CD74 is
eliminated (e.g., 0% expression of CD74 mRNA). In some embodiments, the modification that reduces CD74 mRNA expression eliminates CD74 gene activity.
[0385] In some embodiments, the modification that reduces CD74 expression reduces CD74 protein expression. In some embodiments, the reduced protein expression of CD74 is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification. In some embodiments, the protein expression of CD74 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the protein expression of CD74 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the protein expression of CD74 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the protein expression of CD74 is eliminated (e.g., 0% expression of CD74 protein). In some embodiments, the modification that reduces CD74 protein expression eliminates CD74 gene activity.
[0386] In some embodiments, the modification that reduces CD74 expression comprises inactivation or disruption of the CD74 gene. In some embodiments, the modification that reduces CD74 expression comprises inactivation or disruption of one allele of the CD74 gene. In some embodiments, the modification that reduces CD74 expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the CD74 gene.
[0387] In some embodiments, the modification comprises inactivation or disruption of one or more CD74 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all CD74 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the CD74 gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the CD74 gene. In some embodiments, the modification is a deletion of genomic DNA of the CD74 gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the CD74 gene. In some embodiments, the CD74 gene is knocked out.
[03881 In some embodiments, the provided modified cells comprise a modification of one or more target polynucleotide sequence that regulate expression of MHC class I molecules and MHC class II molecules. Exemplary methods for reducing expression of MHC class I molecules and MHC class 11 molecules including any as described in sections below. In some embodiments, the cell comprises genetic editing modifications to the B2M and NLRC5 genes. In some embodiments, the cell comprises genetic editing modifications to the CIITA and NLRC5 genes. In some embodiments, the cell comprises genetic editing modifications to the B2M and CIITA genes. In particular embodiments, the cell comprises genetic editing modifications to the B2M, CIITA and NLRC5 genes.
[0389] In certain aspects, the present disclosure comprises the modulation (e.g., reduction or elimination) the expression of CD142, which is also known as tissue factor, factor III, and F3. In some embodiments, the modulation occurs using a CRISPR/Cas system.
[0390] In some embodiments, the target polynucleotide sequence is CD 142 or a variant of CD142. In some embodiments, the target polynucleotide sequence is a homolog of CD142. In some embodiments, the target polynucleotide sequence is an ortholog of CD 142.
[0391] In some embodiments, the cells outlined herein comprise a modification targeting the CD 142 gene. In some embodiments, the modification targeting the CD 142 gene by the rare- cutting endonuclease comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid (gRNA) sequence for specifically targeting the CD 142 gene. Useful methods for identifying gRNA sequences to target CD142 are described below.
[0392] Assays to test whether the CD 142 gene has been inactivated are known and described herein. In one embodiment, the resulting modification of the CD 142 gene by PCR and the reduction of CD 142 expression can be assays by FACS analysis. In another embodiment, CD 142 protein expression is detected using a Western blot of cells lysates probed with antibodies to the CD142 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the inactivating modification.
[03931 Useful genomic, polynucleotide and polypeptide information about the human CD142 are provided in, for example, the GeneCard Identifier GC01M094530, HGNC No. 3541, NCBI Gene ID 2152, NCBI RefSeq Nos. NM_001178096.1, NM_001993.4, NP_001171567.1, and NP_001984.1, UniProt No. P13726, and the like.
[0394] In some embodiments, the target polynucleotide sequence is PD-1 or a variant of PD-1. In some embodiments, the target polynucleotide sequence is a homolog of PD-1. In some embodiments, the target polynucleotide sequence is an ortholog of PD-1.
[0395] In some embodiments, the cells outlined herein comprise a genetic modification targeting the gene encoding the programmed cell death protein 1 (PD- 1 ) protein or the PDCD 1 gene. In certain embodiments, primary T cells comprise a genetic modification targeting the PDCD1 gene. The genetic modification can reduce expression of PD-1 polynucleotides and PD- 1 polypeptides in T cells includes primary T cells and CAR-T cells. In some embodiments, the genetic modification targeting the PDCD1 gene by the rare-cutting endonuclease comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid (gRNA) sequence for specifically targeting the PDCD1 gene. Useful methods for identifying gRNA sequences to target PD-1 are described below.
[0396] Assays to test whether the PDCD1 gene has been inactivated are known and described herein. In some embodiments, the resulting genetic modification of the PDCD1 gene by PCR and the reduction of PD-1 expression can be assays by FACS analysis. In another embodiment, PD-1 protein expression is detected using a Western blot of cells lysates probed with antibodies to the PD-1 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the inactivating genetic modification.
[0397] Useful genomic, polynucleotide and polypeptide information about human PD-1 including the PDCD1 gene are provided in, for example, the GeneCard Identifier GC02M241849, HGNC No. 8760, NCBI Gene ID 5133, Uniprot No. Q15116, and NCBI RefSeq Nos. NM_005018.2 and NP_005009.2.
b. Methods of Inactivating or Disrupting Target Genes (e.g., to Reduce Expression)
[0398] In some embodiments, the cells provided herein are modified (e.g., genetically modified) to inactivate or disrupt one or more target polynucleotides or proteins as described. In some embodiments, the cells provided herein are modified (e.g., genetically modified) to reduce expression of the one or more target polynucleotides or proteins as described. In some embodiments, the cell that is modified with the one or more modification to reduce (e.g., eliminate) expression of a polynucleotide or protein is any source cell as described herein. In certain embodiments, the modified pluripotent stem cells (e.g., differentiated cells such as islet cells) disclosed herein comprise one or more modifications to reduce expression of one or more target polynucleotides. Non-limiting examples of the one or more target polynucleotides include any as described above, such as CIITA, B2M, CD 142, NLRC5, HLA-A, HLA-B, HLA-C, LRC5, RFX-ANK, RFX5, RFX-AP, NFY-A, NFY-B, NFY-C, IRF1, and TAPI. In some embodiment, the target polynucleotide may be CD74. In some embodiments, the modifications to reduce expression of the one or more target polynucleotides is combined with one or more modifications to increase expression of a desired transgene. In some embodiments, the modifications create modified cells that are immune-privileged or hypoimmunogenic cells. By modulating (e.g., reducing or deleting) expression of one or a plurality of the target polynucleotides, such cells exhibit decreased immune activation when engrafted into a recipient subject. In some embodiments, the cell is considered hypoimmunogenic, e.g., in a recipient subject or patient upon administration.
[0399] Any method for reducing expression of a target polynucleotide may be used. In some embodiments, the modifications result in permanent elimination or reduction in expression of the target polynucleotide. For instance, in some embodiments, the target polynucleotide or gene is disrupted by introducing a DNA break in the target polynucleotide, such as by using a targeting endonuclease. In other embodiments, the modifications result in transient reduction in expression of the target polynucleotide. For instance, in some embodiments gene repression is achieved using an inhibitory nucleic acid that is complementary to the target polynucleotide to selectively suppress or repress expression of the gene, for instance using antisense techniques,
such as by RNA interference (RNAi), short interfering RNA (siRNA), short hairpin (shRNA), and/or ribozymes.
[0400] In some embodiments, the target polynucleotide sequence is a genomic sequence. In some embodiments, the target polynucleotide sequence is a human genomic sequence. In some embodiments, the target polynucleotide sequence is a mammalian genomic sequence. In some embodiments, the target polynucleotide sequence is a vertebrate genomic sequence.
[0401] In some embodiments, gene disruption is carried out by induction of one or more double-stranded breaks and/or one or more single- stranded breaks in the gene, typically in a targeted manner. In some embodiments, the double-stranded or single-stranded breaks are made by a nuclease, e.g., an endonuclease, such as a gene-targeted nuclease. In some embodiments, the targeted nuclease is selected from zinc finger nucleases (ZFN), transcription activator-like effector nucleases (TALENs), and RNA-guided nucleases such as a CRISPR-associated nuclease (Cas), specifically designed to be targeted to the sequence of a gene or a portion thereof. In some embodiments, the targeted nuclease generates double- stranded or single-stranded breaks that then undergo repair through error prone non-homologous end joining (NHEJ) or, in some cases, precise homology directed repair (HDR) in which a template is used. In some embodiments, the targeted nuclease generates DNA double strand breaks (DSBs). In some embodiments, the process of producing and repairing the breaks is typically error prone and results in insertions and deletions (indels) of DNA bases from NHEJ repair. In some embodiments, the modification may induce a deletion, insertion, or mutation of the nucleotide sequence of the target gene. In some cases, the modification may result in a frameshift mutation, which can result in a premature stop codon. In examples of nuclease-mediated gene editing the targeted edits occur on both alleles of the gene resulting in a biallelic disruption or edit of the gene. In some embodiments, all alleles of the gene are targeted by the gene editing. In some embodiments, modification with a targeted nuclease, such as using a CRISPR/Cas system, leads to complete knockout of the gene. In some embodiments, the nuclease, such as a rare-cutting endonuclease, is introduced into a cell containing the target polynucleotide sequence. The nuclease may be introduced into the cell in the form of a nucleic acid encoding the nuclease.
The process of introducing the nucleic acids into cells can be achieved by any suitable technique. Suitable techniques include calcium phosphate or lipid-mediated transfection, electroporation, and transduction or infection using a viral vector. In some embodiments, the nucleic acid that is introduced into the cell is DNA. In some embodiments, the nuclease is introduced into the cell in the form of a protein. For instance, in the case of a CRISPR/Cas system a ribonucleoprotein (RNP) may be introduced into the cell.
[0402] In some embodiments, the modification occurs using a CRISPR/Cas system. Any CRISPR/Cas system that is capable of altering a target polynucleotide sequence in a cell can be used. Such CRISPR-Cas systems can employ a variety of Cas proteins (Haft et al. PLoS Comput Biol. 2005; l(6)e60). The molecular machinery of such Cas proteins that allows the CRISPR/Cas system to alter target polynucleotide sequences in cells include RNA binding proteins, endo- and exo-nucleases, helicases, and polymerases. In some embodiments, the CRISPR/Cas system is a CRISPR type I system. In some embodiments, the CRISPR/Cas system is a CRISPR type II system. In some embodiments, the CRISPR/Cas system is a CRISPR type V system.
[0403] The CRISPR/Cas systems include targeted systems that can be used to alter any target polynucleotide sequence in a cell. In some embodiments, a CRISPR/Cas system provided herein includes a Cas protein and one or more, such as at least one to two, ribonucleic acids (e.g., guide RNA (gRNA)) that are capable of directing the Cas protein to and hybridizing to a target motif of a target polynucleotide sequence.
[0404] In some embodiments, a Cas protein comprises one or more amino acid substitutions or modifications. In some embodiments, the one or more amino acid substitutions comprises a conservative amino acid substitution. In some instances, substitutions and/or modifications can prevent or reduce proteolytic degradation and/or extend the half-life of the polypeptide in a cell. In some embodiments, the Cas protein can comprise a peptide bond replacement (e.g., urea, thiourea, carbamate, sulfonyl urea, etc.). In some embodiments, the Cas protein can comprise a naturally occurring amino acid. In some embodiments, the Cas protein can comprise an alternative amino acid (e.g., D-amino acids, beta-amino acids, homocysteine,
phosphoserine, etc.). In some embodiments, a Cas protein can comprise a modification to include a moiety (e.g., PEGylation, glycosylation, lipidation, acetylation, end-capping, etc.).
[0405] In some embodiments, a Cas protein comprises a core Cas protein. Exemplary Cas core proteins include, but are not limited to Cast, Cas2, Cas3, Cas4, Cas5, Cas6, Cas7, Cas8 and Cas9. In some embodiments, a Cas protein comprises a Cas protein of an E. coli subtype (also known as CASS2). Exemplary Cas proteins of the E. Coli subtype include, but are not limited to Csel, Cse2, Cse3, Cse4, and Cas5e. In some embodiments, a Cas protein comprises a Cas protein of the Ypest subtype (also known as CASS3). Exemplary Cas proteins of the Ypest subtype include, but are not limited to Csyl, Csy2, Csy3, and Csy4. In some embodiments, a Cas protein comprises a Cas protein of the Nmeni subtype (also known as CASS4). Exemplary Cas proteins of the Nmeni subtype include, but are not limited to, Csnl and Csn2. In some embodiments, a Cas protein comprises a Cas protein of the Dvulg subtype (also known as CASS1). Exemplary Cas proteins of the Dvulg subtype include Csdl, Csd2, and Cas5d. In some embodiments, a Cas protein comprises a Cas protein of the Tneap subtype (also known as CASS7). Exemplary Cas proteins of the Tneap subtype include, but are not limited to, Cstl, Cst2, Cas5t. In some embodiments, a Cas protein comprises a Cas protein of the Hmari subtype. Exemplary Cas proteins of the Hmari subtype include, but are not limited to Cshl, Csh2, and Cas5h. In some embodiments, a Cas protein comprises a Cas protein of the Apem subtype (also known as CASS5). Exemplary Cas proteins of the Apem subtype include, but are not limited to Csal, Csa2, Csa3, Csa4, Csa5, and Cas5a. In some embodiments, a Cas protein comprises a Cas protein of the Mtube subtype (also known as CASS6). Exemplary Cas proteins of the Mtube subtype include, but are not limited to Csml, Csm2, Csm3, Csm4, and Csm5. In some embodiments, a Cas protein comprises a RAMP module Cas protein. Exemplary RAMP module Cas proteins include, but are not limited to, CrmT, Cmr2, Cmr3, Cmr4, Cmr5, and Cmr6. See, e.g., Klompe et al., Nature 571, 219-225 (2019); Strecker et al., Science 365, 48-53 (2019).
[0406] In some embodiments, CRISPR systems of the present disclosure comprise TnpB polypeptides. In some embodiments, TnpB polypeptides may comprise a Ruv-C-like domain. The RuvC domain may be a split RuvC domain comprising RuvC-I, RuvC-II, and RuvC-III
subdomains. In some embodiments, a TnpB may further comprise one or more of a HTH domain, a bridge helix domain, and a zinc finger domain. TnpB polypeptides do not comprise an HNH domain. In some embodiments, a TnpB protein comprises, starting at the N-terminus: a HTH domain, a RuvC-1 subdomain, a bridge helix domain, a RuvC-11 sub-domain, a zinger finger domain, and a RuvC-III sub-domain. In some embodiments, a RuvC-III sub-domain forms the C-terminus of a TnpB polypeptide. In some embodiments, a TnpB polypeptide is from Epsilonproteobacteria bacterium, Actinoplanes lobatus strain DSM 43150, Actinomadura celluolosilytica strain DSM 45823, Actinomadura namibiensis strain DSM 44197, Alicyclobacillus macrosprangiidus strain DSM 17980, Lipingzhangella halophila strain DSM 102030, or Ktedonobacter recemifer. In some embodiments, a TnpB polypeptide is from Ktedonobacter racemifer, or comprises a conserved RNA region with similarity to the 5’ ITR of K. racemifer TnpB loci. In some embodiments, a TnpB may comprise a Fanzor protein, a TnpB homolog found in eukaryotic genomes. In some embodiments, a CRISPR system comprising a TnpB polypeptide binds a target adjacent motif (TAM) sequence 5’ of a target polynucleotide. In some embodiments, a TAM is a transposon-associated motif. In some embodiments, a TAM sequence comprises TCA. In some embodiments, a TAM sequence comprises TTCAN. In some embodiments, a TAM sequence comprises TTGAT. In some embodiments, a TAM sequence comprises ATAAA.
[0407] In some embodiments, the methods for genetically modifying cells to knock out, knock down, or otherwise modify one or more genes comprise using a site-directed nuclease, including, for example, zinc finger nucleases (ZFNs), transcription activator-like effector nucleases (TALENs), meganucleases, transposases, and clustered regularly interspaced short palindromic repeat (CRISPR)/Cas systems
[0408] ZFNs are fusion proteins comprising an array of site-specific DNA binding domains adapted from zinc finger-containing transcription factors attached to the endonuclease domain of the bacterial FokI restriction enzyme. A ZFN may have one or more (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, or more) of the DNA binding domains or zinc finger domains. See, e.g., Carroll et al., Genetics Society of America (2011) 188:773-782; Kim et al., Proc. Natl. Acad. Sci. USA
(1996) 93:1156-1160. Each zinc finger domain is a small protein structural motif stabilized by one or more zinc ions and usually recognizes a 3- to 4-bp DNA sequence. Tandem domains can thus potentially bind to an extended nucleotide sequence that is unique within a cell’s genome.
[0409] Various zinc fingers of known specificity can be combined to produce multifinger polypeptides which recognize about 6, 9, 12, 15, or 18-bp sequences. Various selection and modular assembly techniques are available to generate zinc fingers (and combinations thereof) recognizing specific sequences, including phage display, yeast one-hybrid systems, bacterial one-hybrid and two-hybrid systems, and mammalian cells. Zinc fingers can be engineered to bind a predetermined nucleic acid sequence. Criteria to engineer a zinc finger to bind to a predetermined nucleic acid sequence are known in the art. See, e.g., Sera et al., Biochemistry (2002) 41:7074-7081; Liu et al., Bioinformatics (2008) 24:1850-1857.
[0410] ZFNs containing FokI nuclease domains or other dimeric nuclease domains function as a dimer. Thus, a pair of ZFNs are required to target non-palindromic DNA sites. The two individual ZFNs must bind opposite strands of the DNA with their nucleases properly spaced apart. See Bitinaite et al., Proc. Natl. Acad. Sci. USA (1998) 95:10570-10575. To cleave a specific site in the genome, a pair of ZFNs arc designed to recognize two sequences flanking the site, one on the forward strand and the other on the reverse strand. Upon binding of the ZFNs on either side of the site, the nuclease domains dimerize and cleave the DNA at the site, generating a DSB with 5' overhangs. HDR can then be utilized to introduce a specific mutation, with the help of a repair template containing the desired mutation flanked by homology arms. The repair template is usually an exogenous double-stranded DNA vector introduced to the cell. See Miller et al., Nat. Biotechnol. (2011) 29:143-148; Hockemeyer et al., Nat. Biotechnol. (2011) 29:731-734.
[0411] TALENs are another example of an artificial nuclease which can be used to edit a target gene. TALENs are derived from DNA binding domains termed TALE repeats, which usually comprise tandem arrays with 10 to 30 repeats that bind and recognize extended DNA sequences. Each repeat is 33 to 35 amino acids in length, with two adjacent amino acids (termed the repeat-variable di-residue, or RVD) conferring specificity for one of the four DNA base
pairs. Thus, there is a one-to-one correspondence between the repeats and the base pairs in the target DNA sequences.
[0412] TALENs are produced artificially by fusing one or more TALE DNA binding domains (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more) to a nuclease domain, for example, a FokI endonuclease domain. See Zhang, Nature Biotech. (2011) 29:149-153. Several mutations to FokI have been made for its use in TALENs; these, for example, improve cleavage specificity or activity. See Cermak et al., Nucl. Acids Res. (2011) 39:e82; Miller et al., Nature Biotech. (2011) 29:143-148; Hockemeyer et al., Nature Biotech. (2011) 29:731-734; Wood et al., Science (2011) 333:307; Doyon et al., Nature Methods (2010) 8:74-79; Szczepek et al., Nature Biotech (2007) 25:786-793; Guo et al., J. Mol. Biol. (2010) 200:96. The FokI domain functions as a dimer, requiring two constructs with unique DNA binding domains for sites in the target genome with proper orientation and spacing. Both the number of amino acid residues between the TALE DNA binding domain and the FokI nuclease domain and the number of bases between the two individual TALEN binding sites appear to be important parameters for achieving high levels of activity. Miller et al., Nature Biotech. (2011) 29: 143-148.
[0413] By combining engineered TALE repeats with a nuclease domain, a site-specific nuclease can be produced specific to any desired DNA sequence. Similar to ZFNs, TALENs can be introduced into a cell to generate DSBs at a desired target site in the genome, and so can be used to knock out genes or knock in mutations in similar, HDR-mediated pathways. See Boch, Nature Biotech. (2011) 29:135-136; Boch et al., Science (2009) 326:1509-1512; Moscou et al., Science (2009) 326:3501.
[0414] Meganucleases are enzymes in the endonuclease family which are characterized by their capacity to recognize and cut large DNA sequences (from 14 to 40 base pairs). Meganucleases are grouped into families based on their structural motifs which affect nuclease activity and/or DNA recognition. The most widespread and best known meganucleases are the proteins in the LAGLIDADG family, which owe their name to a conserved amino acid sequence. See Chevalier et al., Nucleic Acids Res. (2001) 29(18): 3757-3774. On the other hand, the GIY- YIG family members have a GIY-YIG module, which is 70-100 residues long and includes four
or five conserved sequence motifs with four invariant residues, two of which arc required for activity. See Van Roey et al., Nature Struct. Biol. (2002) 9:806-811. The His-Cys family meganucleases are characterized by a highly conserved series of histidines and cysteines over a region encompassing several hundred amino acid residues. See Chevalier et al., Nucleic Acids Res. (2001) 29(18):3757-3774. Members of the NHN family are defined by motifs containing two pairs of conserved histidines surrounded by asparagine residues. See Chevalier et al., Nucleic Acids Res. (2001 ) 29(18):3757-3774.
[0415] Because the chance of identifying a natural meganuclease for a particular target DNA sequence is low due to the high specificity requirement, various methods including mutagenesis and high throughput screening methods have been used to create meganuclease variants that recognize unique sequences. Strategies for engineering a meganuclease with altered DNA-binding specificity, e.g., to bind to a predetermined nucleic acid sequence are known in the art. See, e.g., Chevalier et al., Mol. Cell. (2002) 10:895-905; Epinat et al., Nucleic Acids Res (2003) 31:2952-2962; Silva et al., J Mol. Biol. (2006) 361:744-754; Seligman et al., Nucleic Acids Res (2002) 30:3870-3879; Sussman et al., J Mol Biol (2004) 342:31-41; Doyon et al., J Am Chem Soc (2006) 128:2477-2484; Chen et al., Protein Eng Des Sei (2009) 22:249-256;
Amould et al., J Mol Biol. (2006) 355:443-458; Smith et al., Nucleic Acids Res. (2006) 363(2):283-294.
[0416] Like ZFNs and TALENs, Meganucleases can create DSBs in the genomic DNA, which can create a frame-shift mutation if improperly repaired, e.g., via NHEJ, leading to a decrease in the expression of a target gene in a cell. Alternatively, foreign DNA can be introduced into the cell along with the meganuclease. Depending on the sequences of the foreign DNA and chromosomal sequence, this process can be used to modify the target gene. See Silva et al., Current Gene Therapy (2011) 11:11-27.
[0417] Transposases are enzymes that bind to the end of a transposon and catalyze its movement to another part of the genome by a cut and paste mechanism or a replicative transposition mechanism. By linking transposases to other systems such as the CRISPR/Cas system, new gene editing tools can be developed to enable site specific insertions or
manipulations of the genomic DNA. There are two known DNA integration methods using transposons which use a catalytically inactive Cas effector protein and Tn7-like transposons. The transposase-dependent DNA integration does not provoke DSBs in the genome, which may guarantee safer and more specific DNA integration.
[0418] The CRISPR system was originally discovered in prokaryotic organisms (e.g., bacteria and archaea) as a system involved in defense against invading phages and plasmids that provides a form of acquired immunity. Now it has been adapted and used as a po ular gene editing tool in research and clinical applications.
[0419] CRISPR/Cas systems generally comprise at least two components: one or more guide RNAs (gRNAs) and a Cas protein. The Cas protein is a nuclease that introduces a DSB into the target site. CRISPR-Cas systems fall into two major classes: class 1 systems use a complex of multiple Cas proteins to degrade nucleic acids; class 2 systems use a single large Cas protein for the same purpose. Class 1 is divided into types I, III, and IV; class 2 is divided into types II, V, and VI. Different Cas proteins adapted for gene editing applications include, but are not limited to, Cas3, Cas4, Cas5, Cas8a, Cas8b, Cas8c, Cas9, CaslO, Casl2, Casl2a (Cpfl), Casl2b (C2cl), Casl2c (C2c3), Casl2d (CasY), Casl2e (CasX), Casl2f (C2cl0), Casl2g, Casl2h, Casl2i, Casl2k (C2c5), Casl3, Casl3a (C2c2), Casl3b, Casl3c, Casl3d, C2c4, C2c8, C2c9, Cmr5, Csel, Cse2, Csfl, Csm2, Csn2, CsxlO, Csxl l, Csyl, Csy2, Csy3, and Mad7. The most widely used Cas9 is a type II Cas protein and is described herein as illustrative. These Cas proteins may be originated from different source species. For example, Cas9 can be derived from S. pyogenes or S. aureus.
[0420] In the original microbial genome, the type II CRISPR system incorporates sequences from invading DNA between CRISPR repeat sequences encoded as arrays within the host genome. Transcripts from the CRISPR repeat arrays are processed into CRISPR RNAs (crRNAs) each harboring a variable sequence transcribed from the invading DNA, known as the “protospacer” sequence, as well as part of the CRISPR repeat. Each crRNA hybridizes with a second transactivating CRISPR RNA (tracrRNA), and these two RNAs form a complex with the Cas9 nuclease. The protospacer-encoded portion of the crRNA directs the Cas9 complex to
cleave complementary target DNA sequences, provided that they are adjacent to short sequences known as “protospacer adjacent motifs” (PAMs).
[0421] Since its discovery, the CRISPR system has been adapted for inducing sequence specific DSBs and targeted genome editing in a wide range of cells and organisms spanning from bacteria to eukaryotic cells including human cells. In its use in gene editing applications, artificially designed, synthetic gRNAs have replaced the original crRNA:tracrRNA complex. For example, the gRNAs can be single guide RNAs (sgRNAs) composed of a crRNA, a tetraloop, and a tracrRNA. The crRNA usually comprises a complementary region (also called a spacer, usually about 20 nucleotides in length) that is user-designed to recognize a target DNA of interest. The tracrRNA sequence comprises a scaffold region for Cas nuclease binding. The crRNA sequence and the tracrRNA sequence are linked by the tetraloop and each have a short repeat sequence for hybridization with each other, thus generating a chimeric sgRNA. One can change the genomic target of the Cas nuclease by simply changing the spacer or complementary region sequence present in the gRNA. The complementary region will direct the Cas nuclease to the target DNA site through standard RNA-DNA complementary base pairing rules.
[0422] In order for the Cas nuclease to function, there must be a PAM immediately downstream of the target sequence in the genomic DNA. Recognition of the PAM by the Cas protein is thought to destabilize the adjacent genomic sequence, allowing interrogation of the sequence by the gRNA and resulting in gRNA-DNA pairing when a matching sequence is present. The specific sequence of PAM varies depending on the species of the Cas gene. For example, the most commonly used Cas9 nuclease derived from S. pyogenes recognizes a PAM sequence of 5’-NGG-3’ or, at less efficient rates, 5’-NAG-3’, where “N” can be any nucleotide. Other Cas nuclease variants with alternative PAMs have also been characterized and successfully used for genome editing, which are summarized in Table 2 below.
R = A or G; Y = C or T; W = A or T; V = A or C or G; N = any base
[0423] In some embodiments, Cas nucleases may comprise one or more mutations to alter their activity, specificity, recognition, and/or other characteristics. For example, the Cas nuclease may have one or more mutations that alter its fidelity to mitigate off-target effects (e.g., eSpCas9, SpCas9-HFl, HypaSpCas9, HeFSpCas9, and evoSpCas9 high-fidelity variants of SpCas9). For another example, the Cas nuclease may have one or more mutations that alter its PAM specificity.
[0424] In some embodiments, a Cas protein comprises any one of the Cas proteins described herein or a functional portion thereof. As used herein, "functional portion" refers to a portion of a peptide which retains its ability to complex with at least one ribonucleic acid (e.g., guide RNA (gRNA)) and cleave a target polynucleotide sequence. In some embodiments, the functional portion comprises a combination of operably linked Cas9 protein functional domains selected from the group consisting of a DNA binding domain, at least one RNA binding domain, a helicase domain, and an endonuclease domain. In some embodiments, the functional portion comprises a combination of operably linked Cas 12a (also known as Cpfl) protein functional domains selected from the group consisting of a DNA binding domain, at least one RNA binding domain, a helicase domain, and an endonuclease domain. In some embodiments, the functional domains form a complex. In some embodiments, a functional portion of the Cas9 protein comprises a functional portion of a RuvC-like domain. In some embodiments, a functional portion of the Cas9 protein comprises a functional portion of the HNH nuclease domain. In some
embodiments, a functional portion of the Casl2a protein comprises a functional portion of a RuvC-like domain.
[0425] In some embodiments, suitable Cas proteins include, but are not limited to, CasO, Casl2a (i.e., Cpfl), Casl2b, Casl2i, CasX, and Mad7.
[0426] In some embodiments, exogenous Cas protein can be introduced into the cell in polypeptide form. In certain embodiments, Cas proteins can be conjugated to or fused to a cellpenetrating polypeptide or cell-penetrating peptide. As used herein, "cell-penetrating polypeptide" and "cell-penetrating peptide" refers to a polypeptide or peptide, respectively, which facilitates the uptake of molecule into a cell. The cell-penetrating polypeptides can contain a detectable label.
[0427] In certain embodiments, Cas proteins can be conjugated to or fused to a charged protein (e.g., that carries a positive, negative or overall neutral electric charge). Such linkage may be covalent. In some embodiments, the Cas protein can be fused to a superpositively charged GFP to significantly increase the ability of the Cas protein to penetrate a cell (Cronican et al. ACS Chem Biol. 2010; 5(8):747-52). In certain embodiments, the Cas protein can be fused to a protein transduction domain (PTD) to facilitate its entry into a cell. Exemplary PTDs include Tat, oligoarginine, and penetratin. In some embodiments, the Cas9 protein comprises a Cas9 polypeptide fused to a cell-penetrating peptide. In some embodiments, the Cas9 protein comprises a Cas9 polypeptide fused to a PTD. In some embodiments, the Cas9 protein comprises a Cas9 polypeptide fused to a tat domain. In some embodiments, the Cas9 protein comprises a Cas9 polypeptide fused to an oligoarginine domain. In some embodiments, the Cas9 protein comprises a Cas9 polypeptide fused to a penetrating domain. In some embodiments, the Cas9 protein comprises a Cas9 polypeptide fused to a superpositively charged GFP. In some embodiments, the Casl2a protein comprises a Casl2a polypeptide fused to a cell-penetrating peptide. In some embodiments, the Cas 12a protein comprises a Cas 12a polypeptide fused to a PTD. In some embodiments, the Casl2a protein comprises a Casl2a polypeptide fused to a tat domain. In some embodiments, the Cas 12a protein comprises a Cas 12a polypeptide fused to an oligoarginine domain. In some embodiments, the Cas 12a protein comprises a Cas 12a
polypeptide fused to a penetrating domain. In some embodiments, the Casl2a protein comprises a Casl2a polypeptide fused to a superpositively charged GFP.
[0428] In some embodiments, the Cas protein can be introduced into a cell containing the target polynucleotide sequence in the form of a nucleic acid encoding the Cas protein. The process of introducing the nucleic acids into cells can be achieved by any suitable technique. Suitable techniques include calcium phosphate or lipid-mediated transfection, electroporation, and transduction or infection using a viral vector. In some embodiments, the nucleic acid comprises DNA. In some embodiments, the nucleic acid comprises a modified DNA, as described herein. In some embodiments, the nucleic acid comprises mRNA. In some embodiments, the nucleic acid comprises a modified mRNA, as described herein (e.g., a synthetic, modified mRNA).
[0429] In provided embodiments, a CRISPR/Cas system generally includes two components; one or more guide RNA (gRNA) and a Cas protein. In some embodiments, the Cas protein is complexed with the one or more, such as one to two, ribonucleic acids (e.g., guide RNA (gRNA)). In some embodiments, the Cas protein is complexed with two ribonucleic acids. In some embodiments, the Cas protein is complexed with one ribonucleic acid. In some embodiments, the Cas protein is encoded by a modified nucleic acid, as described herein (e.g., a synthetic, modified mRNA).
[0430] In some embodiments, gRNAs are short synthetic RNAs composed of a scaffold sequence for Cas binding and a user-designed spacer or complementary portion designated crRNA. The crRNA is composed of a crRNA targeting sequence (herein after also called a gRNA targeting sequence; usually about 20 nucleotides in length) that defines the genomic target to be modified and a region of crRNA repeat (e.g., GUUUUAGAGCUA; SEQ ID NO: 19). One can change the genomic target of the Cas protein by simply changing the complementary portion sequence (e.g., gRNA targeting sequence) present in the gRNA. In some embodiments the scaffold sequence for Cas binding is made up of a tracrRNA sequence (e.g., UAGCAAGUUAAAAUAAGGCUAGUCCGUUAUCAACUUGAAAAAGUGGCACCGAGU CGGUGCUUU; SEQ ID NO: 20) that hybridizes to the crRNA through its anti-repeat sequence.
The complex between crRNA: tracrRNA recruits the Cas nuclease (e.g., Cas9) and cleaves upstream of a protospacer-adjacent motif (PAM). For the Cas protein to function, there must be a PAM immediately downstream of the target sequence in the genomic DNA. Recognition of the PAM by the Cas protein is thought to destabilize the adjacent genomic sequence, allowing interrogation of the sequence by the gRNA and resulting in gRNA-DNA pairing when a matching sequence is present. The specific sequence of PAM varies depending on the species of the Cas gene. For example, the most commonly used Cas9 nuclease, derived from S. pyogenes, recognizes a PAM sequence of NGG. Other Cas9 variants and other nucleases with alternative PAMs have also been characterized and successfully used for genome editing. Thus, the CRISPR/Cas system can be used to create targeted DSBs at specified genomic loci that are complementary to the gRNA designed for the target loci. The crRNA and tracrRNA can be linked together with a loop sequence (e.g., a tetraloop; GAAA, SEQ ID NO:21) for generation of a gRNA that is a chimeric single guide RNA (sgRNA; Hsu et al. 2013). sgRNA can be generated for DNA-based expression or by chemical synthesis.
[0431] In some embodiments, the complementary portion sequences (e.g., gRNA targeting sequence) of the gRNA will vary depending on the target site of interest. In some embodiments, the gRNAs comprise complementary portions specific to a sequence of a gene set forth in Table 3 or Table 4. In some embodiments, the genomic locus targeted by the gRNAs is located within 4000 bp, within 3500 bp, within 3000 bp, within 2500 bp, within 2000 bp, within 1500 bp, within 1000 bp, or within 500 bp of any of the loci as described.
[0432] The methods disclosed herein contemplate the use of any ribonucleic acid that is capable of directing a Cas protein to and hybridizing to a target motif of a target polynucleotide sequence. In some embodiments, at least one of the ribonucleic acids comprises
[0433] In some embodiments, the Cas protein is complexed with one to two ribonucleic acids (e.g., guide RNA (gRNA)). In some embodiments, the Cas protein is complexed with two ribonucleic acids. In some embodiments, the Cas protein is complexed with one ribonucleic acid. In some embodiments, the Cas protein is encoded by a modified nucleic acid, as described herein (e.g., a synthetic, modified mRNA).
[04341 The methods disclosed herein contemplate the use of any ribonucleic acid that is capable of directing a Cas protein to and hybridizing to a target motif of a target polynucleotide sequence. In some embodiments, at least one of the ribonucleic acids comprises tracrRNA. In some embodiments, at least one of the ribonucleic acids comprises CR1SPR RNA (crRNA). In some embodiments, a single ribonucleic acid comprises a guide RNA that directs the Cas protein to and hybridizes to a target motif of the target polynucleotide sequence in a cell. In some embodiments, at least one of the ribonucleic acids comprises a guide RNA that directs the Cas protein to and hybridizes to a target motif of the target polynucleotide sequence in a cell. In some embodiments, both of the one to two ribonucleic acids comprise a guide RNA that directs the Cas protein to and hybridizes to a target motif of the target polynucleotide sequence in a cell. The ribonucleic acids provided herein can be selected to hybridize to a variety of different target motifs, depending on the particular CRISPR/Cas system employed, and the sequence of the target polynucleotide, as will be appreciated by those skilled in the art. The one to two ribonucleic acids can also be selected to minimize hybridization with nucleic acid sequences other than the target polynucleotide sequence. In some embodiments, the one to two ribonucleic acids hybridize to a target motif that contains at least two mismatches when compared with all other genomic nucleotide sequences in the cell. In some embodiments, the one to two ribonucleic acids hybridize to a target motif that contains at least one mismatch when compared with all other genomic nucleotide sequences in the cell. In some embodiments, the one to two ribonucleic acids are designed to hybridize to a target motif immediately adjacent to a deoxyribonucleic acid motif recognized by the Cas protein. In some embodiments, each of the one to two ribonucleic acids are designed to hybridize to target motifs immediately adjacent to deoxyribonucleic acid motifs recognized by the Cas protein which flank a mutant allele located between the target motifs.
[0435] In some embodiments, each of the one to two ribonucleic acids comprises guide RNAs that directs the Cas protein to and hybridizes to a target motif of the target polynucleotide sequence in a cell.
[04361 In some embodiments, one or two ribonucleic acids (e.g., guide RNAs) are complementary to and/or hybridize to sequences on the same strand of a target polynucleotide sequence. In some embodiments, one or two ribonucleic acids (e.g., guide RNAs) are complementary to and/or hybridize to sequences on the opposite strands of a target polynucleotide sequence. In some embodiments, the one or two ribonucleic acids (e.g., guide RNAs) are not complementary to and/or do not hybridize to sequences on the opposite strands of a target polynucleotide sequence. In some embodiments, the one or two ribonucleic acids (e.g., guide RNAs) are complementary to and/or hybridize to overlapping target motifs of a target polynucleotide sequence. In some embodiments, the one or two ribonucleic acids (e.g., guide RNAs) are complementary to and/or hybridize to offset target motifs of a target polynucleotide sequence.
[0437] In some embodiments, nucleic acids encoding Cas protein and nucleic acids encoding the at least one to two ribonucleic acids are introduced into a cell via viral transduction (e.g., lentiviral transduction). In some embodiments, the Cas protein is complexed with 1-2 ribonucleic acids. In some embodiments, the Cas protein is complexed with two ribonucleic acids. In some embodiments, the Cas protein is complexed with one ribonucleic acid. In some embodiments, the Cas protein is encoded by a modified nucleic acid, as described herein (e.g., a synthetic, modified mRNA).
[0438] Exemplary gRNA targeting sequences useful for CRISPR/Cas-based targeting of genes described herein are provided in Table 3 or Table 4.
[0439] The sequences can be found in W02016183041 filed May 9, 2016, the disclosure including the Tables, Appendices, and Sequence Listing is incorporated herein by reference in its entirety.
[0440] Additional exemplary Cas9 guide RNA sequences useful for CRISPR/Cas-based targeting of genes described herein are provided in Table 4. It will be understood by one of ordinary skill in the art that uracil and thymine can both be represented by ‘t’, instead of ‘u’ for uracil and ‘t’ for thymine; in the context of a ribonucleic acid, it will be understood that ‘t’ is used to represent uracil unless otherwise indicated.
[0441] In some embodiments, it is within the level of a skilled artisan to identify new loci and/or gRNA targeting sequences for use in methods of genetic disruption to reduce or eliminate expression of a gene as described. For example, for CRISPR/Cas systems, when an existing
gRNA targeting sequence for a particular locus (e.g., within a target gene, e.g. set forth in Table 3 or Table 4) is known, an "inch worming" approach can be used to identify additional loci for targeted insertion of transgenes by scanning the flanking regions on either side of the locus for PAM sequences, which usually occurs about every 100 base pairs (bp) across the genome. The PAM sequence will depend on the particular Cas nuclease used because different nucleases usually have different corresponding PAM sequences. The flanking regions on either side of the locus can be between about 500 to 4000 bp long, for example, about 500 bp, about 1000 bp, about 1500 bp, about 2000 bp, about 2500 bp, about 3000 bp, about 3500 bp, or about 4000 bp long. When a PAM sequence is identified within the search range, a new guide can be designed according to the sequence of that locus for use in genetic disruption methods. Although the CRISPR/Cas system is described as illustrative, any gene-editing approaches as described can be used in this method of identifying new loci, including those using ZFNs, TALENS, meganucleases and transposases.
[0442] [In some embodiments, the cells described herein are made using Transcription Activator-Like Effector Nucleases (TALEN) methodologies. By a "TALE-nuclease" (TALEN) is intended a fusion protein consisting of a nucleic acid-binding domain typically derived from a Transcription Activator Like Effector (TALE) and one nuclease catalytic domain to cleave a nucleic acid target sequence. The catalytic domain is preferably a nuclease domain and more preferably a domain having endonuclease activity, like for instance I-TevI, ColE7, NucA and Fok-I. In a particular embodiment, the TALE domain can be fused to a meganuclease like for instance I-Crel and I-Onul or functional variant thereof. In a more preferred embodiment, said nuclease is a monomeric TALE-Nuclease. A monomeric TALE-Nuclease is a TALE-Nuclease that does not require dimerization for specific recognition and cleavage, such as the fusions of engineered TAL repeats with the catalytic domain of I-TevI described in WO2012138927. Transcription Activator like Effector (TALE) are proteins from the bacterial species Xanthomonas comprise a plurality of repeated sequences, each repeat comprising di-residues in position 12 and 13 (RVD) that are specific to each nucleotide base of the nucleic acid targeted sequence. Binding domains with similar modular basc-pcr-basc nucleic acid binding properties
(MBBBD) can also be derived from new modular proteins recently discovered by the applicant in a different bacterial species. The new modular proteins have the advantage of displaying more sequence variability than TAL repeats. Preferably, RVDs associated with recognition of the different nucleotides are HD for recognizing C, NG for recognizing T, N1 for recognizing A, NN for recognizing G or A, NS for recognizing A, C, G or T, HG for recognizing T, IG for recognizing T, NK for recognizing G, HA for recognizing C, ND for recognizing C, HI for recognizing C, HN for recognizing G, NA for recognizing G, SN for recognizing G or A and YG for recognizing T, TL for recognizing A, VT for recognizing A or G and SW for recognizing A. In another embodiment, critical amino acids 12 and 13 can be mutated towards other amino acid residues in order to modulate their specificity towards nucleotides A, T, C and G and in particular to enhance this specificity. TALEN kits are sold commercially.
[0443] In some embodiments, the cells are manipulated using zinc finger nuclease (ZFN). A "zinc finger binding protein" is a protein or polypeptide that binds DNA, RNA and/or protein, preferably in a sequence- specific manner, as a result of stabilization of protein structure through coordination of a zinc ion. The term zinc finger binding protein is often abbreviated as zinc finger protein or ZFP. The individual DNA binding domains are typically referred to as "fingers." A ZFP has least one finger, typically two fingers, three fingers, or six fingers. Each finger binds from two to four base pairs of DNA, typically three or four base pairs of DNA. A ZFP binds to a nucleic acid sequence called a target site or target segment. Each finger typically comprises an approximately 30 amino acid, zinc-chelating, DNA-binding subdomain. Studies have demonstrated that a single zinc finger of this class consists of an alpha helix containing the two invariant histidine residues coordinated with zinc along with the two cysteine residues of a single beta turn (see, e.g., Berg & Shi, Science 271:1081-1085 (1996)).
[0444] In some embodiments, the cells described herein are made using a homing endonuclease. Such homing endonucleases are well-known to the art (Stoddard 2005). Homing endonucleases recognize a DNA target sequence and generate a single- or double-strand break. Homing endonucleases are highly specific, recognizing DNA target sites ranging from 12 to 45 base pairs (bp) in length, usually ranging from 14 to 40 bp in length. The homing endonuclease
may for example correspond to a LAGLID ADG endonuclease, to an HNH endonuclease, or to a GIY-YIG endonuclease. In some embodiments, the homing endonuclease can be an I-Crel variant.
[0445] In some embodiments, the cells described herein are made using a meganuclease. Meganucleases are by definition sequence- specific endonucleases recognizing large sequences (Chevalier, B. S. and B. L. Stoddard, Nucleic Acids Res., 2001, 29, 3757-3774). They can cleave unique sites in living cells, thereby enhancing gene targeting by 1000-fold or more in the vicinity of the cleavage site (Puchta et al., Nucleic Acids Res., 1993, 21, 5034-5040; Rouet et al., Mol. Cell. Biol., 1994, 14, 8096-8106; Choulika et al., Mol. Cell. Biol., 1995, 15, 1968-1973; Puchta et al., Proc. Natl. Acad. Sci. USA, 1996, 93, 5055-5060; Sargent et al., Mol. Cell. Biol., 1997, 17, 267-77; Donoho et al., Mol. Cell. Biol, 1998, 18, 4070-4078; Elliott et al., Mol. Cell. Biol., 1998, 18, 93-101; Cohen-Tannoudji et al., Mol. Cell. Biol., 1998, 18, 1444-1448).
[0446] In some embodiments, the cells provided herein are made using RNA silencing or RNA interference (RNAi) to knockdown (e.g., decrease, eliminate, or inhibit) the expression of a polypeptide. Useful RNAi methods include those that utilize synthetic RNAi molecules, short interfering RNAs (siRNAs), PlWI-interacting NRAs (piRNAs), short hairpin RNAs (shRNAs), microRNAs (miRNAs), and other transient knockdown methods recognized by those skilled in the art. Reagents for RNAi including sequence specific shRNAs, siRNA, miRNAs and the like are commercially available. For instance, a target polynucleotide, such as any described above, e.g., CIITA, B2M, or NLRC5, can be knocked down in a cell by RNA interference by introducing an inhibitory nucleic acid complementary to a target motif of the target polynucleotide, such as an siRNA, into the cells. In some embodiments, a target polynucleotide, such as any described above, e.g., CIITA, B2M, or NLRC5, can be knocked down in a cell by transducing a shRNA-expressing virus into the cell. In some embodiments, RNA interference is employed to reduce or inhibit the expression of at least one selected from the group consisting of CIITA, B2M, and NLRC5. c. Exemplary Target Polynucleotides and Methods for Reducing Expression
[04471 In certain embodiments, the modification reduces or eliminates, such as knocks out, the expression of MHC class I molecules (e.g., MHC class I genes encoding MHC class I molecules) by targeting the accessory chain B2M. In some embodiments, the modification occurs using a CRISPR/Cas system. By reducing or eliminating, such as knocking out, expression of B2M, surface trafficking of MHC class I molecules is blocked, and such cells exhibit immune tolerance when engrafted into a recipient subject. In some embodiments, the cell is considered hypoimmunogenic, e.g., in a recipient subject or patient upon administration.
[0448] In some embodiments, the target polynucleotide sequence provided herein is a variant of B2M. In some embodiments, the target polynucleotide sequence is a homolog of B2M. In some embodiments, the target polynucleotide sequence is an ortholog of B2M.
[0449] In some embodiments, decreased or eliminated expression of B2M reduces or eliminates expression of one or more of the following MHC class I molecules - HLA-A, HLA- B, and HLA-C.
[0450] In some embodiments, the modified pluripotent stem cells cell comprises a modification targeting the B2M gene. In some embodiments, the modification targeting the B2M gene is by using a targeted nuclease system that comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid sequence for specifically targeting the B2M gene. In some embodiments, the at least one guide ribonucleic acid sequence (e.g., gRNA targeting sequence) for specifically targeting the B2M gene is selected from the group consisting of SEQ ID NOS:81240-85644 of Appendix 2 or Table 15 of W02016/183041, the disclosure of which is herein incorporated by reference in its entirety.
[0451] In some embodiments, an exogenous nucleic acid or transgene encoding a polypeptide as disclosed herein (e.g., a chimeric antigen receptor, CD47, or another tolerogenic factor disclosed herein) is inserted at the B2M gene. Exemplary transgenes for targeted insertion at the B2M locus include any as described herein.
[0452] Assays to test whether the B2M gene has been inactivated are known and described herein. In one embodiment, the resulting modification of the B2M gene by PCR and
the reduction of HLA-I expression can be assays by flow cytometry, such as by FACS analysis. In another embodiment, B2M protein expression is detected using a Western blot of cells lysates probed with antibodies to the B2M protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the inactivating modification.
[0453] In some embodiments, the technologies disclosed herein modulate (e.g., reduce or eliminate) the expression of MHC-I genes by targeting and modulating (e.g., reducing or eliminating) expression of the NLR family, CARD domain containing 5/NOD27/CLR16.1 (NLRC5). In some embodiments, the modulation occurs using a CRISPR/Cas system. NLRC5 is a critical regulator of MHC-I- mediated immune responses and, similar to CIITA, NLRC5 is highly inducible by IFN-y and can translocate into the nucleus. NLRC5 activates the promoters of MHC-I genes and induces the transcription of MHC-I as well as related genes involved in MHC-I antigen presentation.
[0454] In some embodiments, the target polynucleotide sequence is a variant of NLRC5. In some embodiments, the target polynucleotide sequence is a homolog of NLRC5. In some embodiments, the target polynucleotide sequence is an ortholog of NLRC5.
[0455] In some embodiments, the cells outlined herein comprise a genetic modification targeting the NLRC5 gene. In some embodiments, the genetic modification targeting the NLRC5 gene by the rare-cutting endonuclease comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid sequence for specifically targeting the NLRC5 gene. In some embodiments, the at least one guide ribonucleic acid sequence for specifically targeting the NLRC5 gene is selected from the group consisting of SEQ ID NOS:36353-81239 of Appendix 3 or Table 14 of WO2016183041, the disclosure is incorporated by reference in its entirety.
[0456] Assays to test whether the NLRC5 gene has been inactivated are known and described herein. In some embodiments, the resulting genetic modification of the NLRC5 gene by PCR and the reduction of HLA-I expression can be assays by FACS analysis. In another
embodiment, NLRC5 protein expression is detected using a Western blot of cells lysates probed with antibodies to the NLRC5 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the inactivating genetic modification.
[0457] In some embodiments, the reduction of the MHC class I expression or function (HLA I when the cells are derived from human cells) in the modified cells can be measured using techniques known in the art; for example, FACS techniques using labeled antibodies that bind the HLA complex; for example, using commercially available HLA-A, B, C antibodies that bind to the alpha chain of the human major histocompatibility HLA Class I antigens. In addition, the cells can be tested to confirm that the HLA I complex is not expressed on the cell surface. This may be assayed by FACS analysis using antibodies to one or more HLA cell surface components as discussed above. In addition to the reduction of HLA I (or MHC class I), the modified pluripotent stem cells provided herein have a reduced susceptibility to macrophage phagocytosis and NK cell killing. Methods to assay for hypoimmunogenic phenotypes of the modified cells are described further below.
[0458] In certain aspects, the modification reduces or eliminates, such as knocks out, the expression of MHC class II genes by targeting Class II transactivator (CIITA) expression. In some embodiments, the modification occurs using a CRISPR/Cas system. CIITA is a member of the LR or nucleotide binding domain (NBD) leucine-rich repeat (LRR) family of proteins and regulates the transcription of MHC class II by associating with the MHC enhanceosome. By reducing or eliminating, such as knocking out, expression of CIITA, expression of MHC class II molecules is reduced thereby also reducing surface expression. In some cases, such cells exhibit immune tolerance when engrafted into a recipient subject. In some embodiments, the cell is considered hypoimmunogenic, e.g., in a recipient subject or patient upon administration.
[0459] In some embodiments, the target polynucleotide sequence is a variant of CIITA. In some embodiments, the target polynucleotide sequence is a homolog of CIITA. In some embodiments, the target polynucleotide sequence is an ortholog of CIITA.
[04601 In some embodiments, reduced or eliminated expression of CIITA reduces or eliminates expression of one or more of the following MHC class II are HLA-DP, HLA-DM, HLA-DOA, HLA-DOB, HLA-DQ, and HLA-DR.
[0461] In some embodiments, the modified cell comprises a modification targeting the CIITA gene. In some embodiments, the modification targeting the CIITA gene is by a targeted nuclease system that comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid sequence for specifically targeting the CIITA gene. In some embodiments, the at least one guide ribonucleic acid sequence (e.g., gRNA targeting sequence) for specifically targeting the CIITA gene is selected from the group consisting of SEQ ID NOS:5184-36352 of Appendix 1 or Table 12 of W02016183041, the disclosure is incorporated by reference in its entirety.
[0462] In some embodiments, an exogenous nucleic acid or transgene encoding a polypeptide as disclosed herein (e.g., a chimeric antigen receptor, CD47, or another tolerogenic factor disclosed herein) is inserted at the CIITA gene. Exemplary transgenes for targeted insertion at the B2M locus include any as described herein.
[0463] Assays to test whether the CIITA gene has been inactivated are known and described herein. In one embodiment, the resulting modification of the CIITA gene by PCR and the reduction of HLA-II expression can be assays by flow cytometry, such as by FACS analysis. In another embodiment, CIITA protein expression is detected using a Western blot of cells lysates probed with antibodies to the CIITA protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the inactivating modification.
[0464] In some embodiments, the reduction of the MHC class II expression or function (HLA II when the cells are derived from human cells) in the modified cells can be measured using techniques known in the art, such as Western blotting using antibodies to the protein, FACS techniques, RT-PCR techniques, etc. In some embodiments, the modified cells can be tested to confirm that the HLA II complex is not expressed on the cell surface. Methods to
assess surface expression include methods known in the art (See Figure 21 of WO2018132783, for example) and generally is done using either Western Blots or FACS analysis based on commercial antibodies that bind to human HLA Class II HLA-DR, DP and most DQ antigens. In addition to the reduction of HLA 11 (or MHC class 11), the modified pluripotent stem cells provided herein have a reduced susceptibility to macrophage phagocytosis and NK cell killing. Methods to assay for hypoimmunogenic phenotypes of the modified cells are described further below.
[0465] In certain aspects, the modification reduces or eliminates, such as knocks out, the expression of CD 142. In some embodiments, the modification occurs using a CRISPR/Cas system. CD 142, also known as tissue factor (F3) is a membrane-bound protein that initiates blood coagulation by forming a complex with circulating factor VII or factor Vila. The CD142(TF):VIIa complex activates factors IX or X by specific limited proteolysis. CD 142 (TF) plays a role in normal hemostasis by initiating the cell-surface assembly and propagation of the coagulation protease cascade. By reducing or eliminating, such as knocking out, expression of CD 142, expression of MHC class II molecules is reduced thereby also reducing surface expression. In some cases, such cells exhibit immune tolerance when engrafted into a recipient subject. In some embodiments, the cell is considered hypoimmunogenic, e.g., in a recipient subject or patient upon administration.
[0466] In some embodiments, the target polynucleotide sequence is a variant of CD 142. In some embodiments, the target polynucleotide sequence is a homolog of CD 142. In some embodiments, the target polynucleotide sequence is an ortholog of CD 142.
[0467] In some embodiments, the modified pluripotent stem cells comprises a modification targeting the CD 142 gene. In some embodiments, the modification targeting the CD 142 gene is by a targeted nuclease system that comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid sequence for specifically targeting the CD142 gene. In some embodiments, the target polynucleotide sequence is CD142 or a variant of CD 142. In some embodiments, the target polynucleotide sequence is a homolog of CD 142. In some embodiments, the target polynucleotide sequence is an ortholog of CD 142.
[04681 In some embodiments, the cells outlined herein may comprise a modification targeting the CD142 gene. In some embodiments, the modification targeting the CD142 gene by the rare-cutting endonuclease comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid (gRNA) sequence for specifically targeting the CD 142 gene. Useful methods for identifying gRNA sequences to target CD 142 are described below.
[0469] Assays to test whether the CD 142 gene has been inactivated are known and described herein. In one embodiment, the resulting modification of the CD 142 gene by PCR and the reduction of CD 142 expression can be assays by FACS analysis. In another embodiment, CD 142 protein expression is detected using a Western blot of cells lysates probed with antibodies to the CD142 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the inactivating modification. Useful genomic, polynucleotide and polypeptide information about the human CD 142 are provided in, for example, the GeneCard Identifier GC01M094530, HGNC No. 3541, NCBI Gene ID 2152, NCBI RefSeq Nos. NM_001178096.1, NM_001993.4, NP_001171567.1, and NP_001984.1, UniProt No. P13726, and the like.
[0470] In some embodiments, an exogenous nucleic acid or transgene encoding a polypeptide as disclosed herein (e.g., a chimeric antigen receptor, CD46, CD59, CD55, or CD47 or another tolerogenic factor disclosed herein) is inserted at the CD 142 gene. Exemplary transgenes for targeted insertion at the CD142 locus include any as described herein.
[0471] In some embodiments, the reduction of the CD 142 expression or function in the modified cells can be measured using techniques known in the ail, such as Western blotting using antibodies to the protein, FACS techniques, RT-PCR techniques, etc. In some embodiments, the modified cells can be tested to confirm that CD 142 is not expressed on the cell surface. Methods to assess surface expression include methods known in the ail (See Figure 21 of WO2018132783, for example) and generally is done using either Western Blots or FACS analysis based on commercial antibodies that bind to human CD142. In addition to the reduction
of CD 142, the modified cells provided herein have a reduced susceptibility to IBMIR. Methods to assay for hypoimmunogenic phenotypes of the modified cells are described further below.
[0472] In some embodiments, the modification that reduces CD142 expression reduces CD 142 mRNA expression. In some embodiments, the reduced mRNA expression of CD 142 is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification. In some embodiments, the mRNA expression of CD 142 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the mRNA expression of CD 142 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the mRNA expression of CD142 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the mRNA expression of CD 142 is eliminated (e.g., 0% expression of CD 142 mRNA). In some embodiments, the modification that reduces CD142 mRNA expression eliminates CD 142 gene activity.
[0473] In some embodiments, the modification that reduces CD142 expression reduces CD 142 protein expression. In some embodiments, the reduced protein expression of CD 142 is relative to an unmodified or wild-type cell of the same cell type that does not comprise the modification. In some embodiments, the protein expression of CD 142 is reduced by more than about 5%, such as reduced by more than about any of 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or more. In some embodiments, the protein expression of CD142 is reduced by up to about 100%, such as reduced by up to about any of 90%, 80%, 70%, 60%, 50%, 40%, 30%, 20%, 10%, 5%, or less. In some embodiments, the protein expression of CD142 is reduced by any of about 5%, 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, or 100%. In some embodiments, the protein expression of CD 142 is eliminated (e.g., 0% expression of CD 142 protein). In some embodiments, the modification that reduces CD 142 protein expression eliminates CD 142 gene activity.
[0474] In some embodiments, the modification that reduces CD142 expression comprises inactivation or disruption of the CD 142 gene. In some embodiments, the modification that
reduces CD 142 expression comprises inactivation or disruption of one allele of the CD 142 gene. In some embodiments, the modification that reduces CD 142 expression comprises inactivation or disruption comprises inactivation or disruption of both alleles of the CD 142 gene.
[0475] In some embodiments, the modification comprises inactivation or disruption of one or more CD 142 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption of all CD142 coding sequences in the cell. In some embodiments, the modification comprises inactivation or disruption comprises an indel in the CD 142 gene. In some embodiments, the modification is a frameshift mutation of genomic DNA of the CD 142 gene. In some embodiments, the modification is a deletion of genomic DNA of the CD 142 gene. In some embodiments, the modification is a deletion of a contiguous stretch of genomic DNA of the CD 142 gene.
[0476] Exemplary guide target sequences for CD 142 are known, for example, as shown in Table 5.
[0477] In some embodiments, modified pluripotent stem cells provided herein arc genetically modified, such as by introduction of one or more modifications into a cell to overexpress a desired polynucleotide in the cell. In some embodiments, the cell to be modified is an unmodified cell that has not previously been introduced with the one or more modifications. In some embodiments, the modified pluripotent stem cells provided herein are genetically modified to include one or more exogenous polynucleotides encoding an exogenous protein (also interchangeably used with the term “transgene”). As described, in some embodiments, the cells
are modified to increase expression of certain genes that are tolerogenic (e.g., immune) factors that affect immune recognition and tolerance in a recipient. The one or more polynucleotides, e.g., exogenous polynucleotides, may be expressed (e.g. overexpressed) in the modified pluripotent stem cells together with one or more genetic modifications to reduce expression of a target polynucleotide described above, such as an MHC class I and/or MHC class II molecule or CD 142. In some embodiments, the provided modified pluripotent stem cells do not trigger or activate an immune response upon administration to a recipient subject.
[0478] In some embodiments, the modified pluripotent stem cell includes 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more different overexpressed polynucleotides. In some embodiments, the overexpressed polynucleotide is an exogenous polynucleotide. In some embodiments, the modified pluripotent stem cell includes 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more different exogenous polynucleotides. In some embodiments, the overexpressed polynucleotide is an exogenous polynucleotide that is expressed episomally in the cells. In some embodiments, the overexpressed polynucleotide is an exogenous polynucleotide that is inserted or integrated into one or more genomic loci of the modified cell.
[0479] In some embodiments, expression of a polynucleotide is increased, i.e., the polynucleotide is overexpressed, using a fusion protein containing a DNA-targeting domain and a transcriptional activator. Targeted methods of increasing expression using transactivator domains are known to a skilled artisan.
[0480] In some embodiments, the modified pluripotent stem cell contains one or more exogenous polynucleotides in which the one or more exogenous polynucleotides are inserted or integrated into a genomic locus of the cell by non-targeted insertion methods, such as by transduction with a lentiviral vector. In some embodiments, the one or more exogenous polynucleotides are inserted or integrated into the genome of the cell by targeted insertion methods, such as by using homology directed repair (HDR). Any suitable method can be used to insert the exogenous polynucleotide into the genomic locus of the modified cell by HDR including the gene editing methods described herein (e.g., a CRISPR/Cas system). In some embodiments, the one or more exogenous polynucleotides are inserted into one or more genomic
locus, such as any genomic locus described herein (e.g., Table 6). In some embodiments, the exogenous polynucleotides are inserted into the same genomic loci. In some embodiments, the exogenous polynucleotides are inserted into different genomic loci. In some embodiments, the two or more of the exogenous polynucleotides are inserted into the same genomic loci, such as any genomic locus described herein (e.g., Table 6). In some embodiments, two or more exogenous polynucleotides are inserted into a different genomic loci, such as two or more genomic loci as described herein (e.g., Table 6).
[0481] Exemplary polynucleotides for overexpression, and methods for overexpressing the same, are described below.
[0482] In some embodiments, expression of a tolerogenic factor is overexpressed or increased in the cell. In some embodiments, the modified pluripotent stem cell includes increased expression, i.e., overexpression, of at least one tolerogenic factor. In some embodiments, the tolerogenic factor is any factor that promotes or contributes to promoting or inducing tolerance to the modified cell by the immune system (e.g., innate or adaptive immune system). In some embodiments, the tolerogenic factor is DUX4, B2M-HLA-E, CD16, CD52, CD47, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, CCL21, MFGE8, SERPINB9, CD35, IL-39, CD16 Fc Receptor, IL15-RF, and H2-M3. In some embodiments, the tolerogenic factor is CD47, PD-L1, HLA-E or HLA-G, CCL21, FasL, Serpinb9, CD200 or Mfge8, or any combination thereof. In some embodiments, the one or more tolerogenic factors are selected from the group consisting of CD16, CD24, CD35, CD39, CD46, CD47, CD52, CD55, CD59, CD64, CD200, CCL22, CTLA4-lg, Cl inhibitor, FASL, 1DO1, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, IL- 10, IL-35, PD-L1, SERPINB9, CCL21, MFGE8, DUX4, B2M-HLA-E, CD27, IL-39, CD16 Fc Receptor, IL15-RF, H2-M3 (HLA-G), A20/TNFAIP3, CR1, HLA-F, and MANF. In some embodiments, the cell includes at least one exogenous polynucleotide that includes a polynucleotide that encodes for a tolerogenic factor. For instance, in some embodiments, at least one of the exogenous polynucleotides is a polynucleotide that encodes CD47. Provided herein are cells that do not trigger or activate an immune response upon administration to a recipient
subject. As described above, in some embodiments, the cells are modified to increase expression of genes and tolerogenic (e.g., immune) factors that affect immune recognition and tolerance in a recipient.
[0483] In some embodiments, the present disclosure provides a cell or population thereof that has been modified to express the tolerogenic factor (e.g., immunomodulatory polypeptide), such as CD47. In some embodiments, the present disclosure provides a method for altering a cell genome to express the tolerogenic factor (e.g., immunomodulatory polypeptide), such as CD47. In some embodiments, the modified cell expresses an exogenous tolerogenic factor (e.g., immunomodulatory polypeptide), such as an exogenous CD47. In some instances, overexpression or increasing expression of the exogenous polynucleotide is achieved by introducing into the cell (e.g., transducing the cell) within expression vector comprising a nucleotide sequence encoding a human CD47 polypeptide. In some embodiments, the expression vector may be a viral vector, such as a lentiviral vector) or may be a non- viral vector. In some embodiments, the cell is modified to contain one or more exogenous polynucleotides in which at least one of the exogenous polynucleotides includes a polynucleotide that encodes for a tolerogenic factor. In some of any embodiments, the tolerogenic factor is DUX4, B2M-HLA-E, CD16, CD52, CD47, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, CCL21, MFGE8, SERPINB9, CD35, IL- 39, CD16 Fc Receptor, IL15-RF, and H2-M3. In some embodiments, the tolerogenic factor is selected from CD47, PD-L1, HLA-E or HLA-G, CCL21, FasL, Serpinb9, CD200 or Mfge8, or any combination thereof (e.g., all thereof). In some embodiments, the one or more tolerogenic factors are selected from the group consisting of CD16, CD24, CD35, CD39, CD46, CD47, CD52, CD55, CD59, CD64, CD200, CCL22, CTLA4-Ig, Cl inhibitor, FASL, IDO1, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, IL-10, IL-35, PD-L1, SERPINB9, CCL21, MFGE8, DUX4, B2M-HLA-E, CD27, IL-39, CD16 Fc Receptor, IL15-RF, H2-M3 (HLA-G), A20/TNFAIP3, CR1, HLA-F, and MANF. For instance, in some embodiments, at least one of the exogenous polynucleotides is a polynucleotide that encodes CD47.
[04841 In some embodiments, the tolerogenic factor is CD47. In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD47, such as human CD47. In some embodiments, CD47 is overexpressed in the cell. In some embodiments, the expression of CD47 is overexpressed or increased in the modified cell compared to a similar cell of the same cell type that has not been modified with the modification, such as a reference or unmodified cell, e.g. a cell not modified with an exogenous polynucleotide encoding CD47. CD47 is a leukocyte surface antigen and has a role in cell adhesion and modulation of integrins. It is normally expressed on the surface of a cell and signals to circulating macrophages not to eat the cell. Useful genomic, polynucleotide and polypeptide information about human CD47 are provided in, for example, the NP_001768.1, NP_942088.1, NM_001777.3 and NM_198793.2.
[0485] In some embodiments, the modified pluripotent stem cell includes increased expression, i.e., overexpression, of at least one tolerogenic factor. In some embodiments, the cell includes at least one exogenous polynucleotide that includes a polynucleotide that encodes for a tolerogenic factor. In some embodiments, tolerogenic factors include DUX4, B2M-HLA-E, CD16, CD52, CD47, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, CCL21, MFGE8, SERPINB9, CD35, IL- 39, CD16 Fc Receptor, IL15-RF, and H2-M3, or any combination thereof. In some embodiments, the one or more tolerogenic factors are selected from the group consisting of CD16, CD24, CD35, CD39, CD46, CD47, CD52, CD55, CD59, CD64, CD200, CCL22, CTLA4-Ig, Cl inhibitor, FASL, IDOL HLA-C, HLA-E, HLA-E heavy chain, HLA-G, IL- 10, IL-35, PD-L1, SERPINB9, CCL21, MFGE8, DUX4, B2M-HLA-E, CD27, IL-39, CDI6 Fc Receptor, IL15-RF, H2-M3 (HLA-G), A20/TNFAIP3, CR1, HLA-F, and MANF. For instance, in some embodiments, at least one of the overexpressed (e.g., exogenous) polynucleotides is a polynucleotide that encodes CD47.
[0486] In some embodiments, the present disclosure provides a cell or population thereof that has been modified to express the tolerogenic factor (e.g., immunomodulatory polypeptide), such as CD47. In some embodiments, the present disclosure provides a method for altering a cell genome to express the tolerogenic factor (e.g., immunomodulatory polypeptide), such as
CD47. In some embodiments, the modified pluripotent stem cell expresses an exogenous tolerogenic factor (e.g., immunomodulatory polypeptide), such as an exogenous CD47. In some instances, the cell expresses an expression vector comprising a nucleotide sequence encoding a human CD47 polypeptide.
[0487] In some embodiments, the modified pluripotent stem cell contains an overexpressed polynucleotide that encodes CD47, such as human CD47. In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD47, such as human CD47. In some embodiments, CD47 is overexpressed in the cell. In some embodiments, the expression of CD47 is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CD47.
[0488] In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD47 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1. In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD47 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1. In some embodiments, the cell comprises an exogenous nucleotide sequence for CD47 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_001777.3 and NM_198793.2. In some embodiments, the cell comprises an exogenous nucleotide sequence for CD47 as set forth in NCBI Ref. Sequence Nos. NM_001777.3 and NM_198793.2.
[0489] In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD47 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1. In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD47 polypeptide having an amino acid sequence as
set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1. In some embodiments, the cell comprises an exogenous nucleotide sequence for CD47 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_001777.3 and NM_198793.2. In some embodiments, the cell comprises an exogenous nucleotide sequence for CD47 as set forth in NCBI Ref. Sequence Nos. NM_001777.3 and NM_198793.2.
[0490] In some embodiments, the cell comprises an exogenous CD47 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1. In some embodiments, the cell outlined herein comprises an exogenous CD47 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_001768.1 and NP_942088.1.
[0491] In some embodiments, the cell comprises an overexpressed polynucleotide encoding a CD47 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 1. In some embodiments, the cell comprises an exogenous polynucleotide encoding a CD47 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 1. In some embodiments, the cell comprises an overexpressed polynucleotide encoding a CD47 polypeptide having the amino acid sequence as set forth in SEQ ID NO: 1. In some embodiments, the cell comprises an exogenous polynucleotide encoding a CD47 polypeptide having the amino acid sequence as set forth in SEQ ID NO: 1.
[0492] In some embodiments, the cell comprises an overexpressed CD47 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 2. In some embodiments, the cell comprises an exogenous CD47 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 2. In some embodiments, the cell comprises an overexpressed CD47 polypeptide having the amino acid sequence as set forth in SEQ ID NO: 2. In some embodiments, the cell comprises an exogenous CD47 polypeptide having the amino acid sequence as set forth in SEQ ID NO: 2. In some
embodiments, the exogenous nucleotide sequence encoding the CD59 polypeptide is operably linked to a sequence encoding a heterologous signal peptide. In some embodiments, an exogenous polynucleotide encoding CD47 is integrated into the genome of the cell by targeted or non-targeted methods of insertion, such as described further below. In some embodiments, targeted insertion is by homology-dependent insertion into a target locus, such as by insertion into any one of the gene loci depicted in Table 6, e.g. a B2M gene or a CIITA gene. In some embodiments, targeted insertion is by homology-independent insertion, such as by insertion into a safe harbor locus. In some cases, the polynucleotide encoding CD47 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding CD47 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus.
[0493] In some embodiments, all or a functional portion of CD47 can be linked to other components such as a signal peptide, a leader sequence, a secretory signal, a label (e.g., a reporter gene), or any combination thereof. In some embodiments, the nucleic acid sequence encoding a signal peptide of CD47 is replaced with a nucleic acid sequence encoding a signal peptide from a heterologous protein. The heterologous protein can be, for example, CD8a, CD28, tissue plasminogen activator (tPA), growth hormone, granulocyte-macrophage colony stimulating factor (GM-CSF), GM-CSF receptor (GM-CSFRa), or an immunoglobulin (e.g., IgE or IgK). In some embodiments, the signal peptide is a signal peptide from an immunoglobulin (such as IgG heavy chain or IgG-kappa light chain), a cytokine (such as interleukin-2 (IL-2), or CD33), a serum albumin protein (e.g., HSA or albumin), a human azurocidin preprotein signal sequence, a luciferase, a trypsinogen (e.g. chymotrypsinogen or trypsinogen) or other signal peptide able to efficiently express a protein by or on a cell.
[0494] In certain embodiments, the exogenous polynucleotide encoding CD47 is operably linked to a promoter.
[0495] In some embodiments, the exogenous polynucleotide encoding CD47 is inserted into any one of the gene loci depicted in Table 6. In some cases, the exogenous polynucleotide
encoding CD47 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the exogenous polynucleotide encoding CD47 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the exogenous polynucleotide encoding CD47 is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding CD47, into a genomic locus of the cell.
[0496] In some embodiments, CD47 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CD47 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous CD47 mRNA.
[0497] In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD200, such as human CD200. In some embodiments, CD200 is overexpressed in the cell. In some embodiments, the expression of CD200 is increased in the modified cell compared to a si milar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CD200. Useful genomic, polynucleotide and polypeptide information about human CD200 are provided in, for example, the GeneCard Identifier GC03P112332, HGNC No. 7203, NCBI Gene ID 4345, Uniprot No. P41217, and NCBI RefSeq Nos.
NP_001004196.2, NM_001004196.3, NP_001305757.1, NM_001318828.1, NP_005935.4, NM_005944.6, XP_005247539.1, and XM_005247482.2. In certain embodiments, the polynucleotide encoding CD200 is operably linked to a promoter.
[0498] In some embodiments, the polynucleotide encoding CD200 is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding CD200 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding CD200 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1)
gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding CD200 is inserted into a B2M gene locus, a CIITA gene locus, or a CD142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding CD200, into a genomic locus of the cell.
[0499] In some embodiments, CD200 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CD200 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous CD200 mRNA.
[0500] In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes HLA-E, such as human HLA-E. In some embodiments, HLA-E is overexpressed in the cell. In some embodiments, the expression of HLA-E is increased in the modified pluripotent stem cell compared to a similar’ reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding HLA-E. Useful genomic, polynucleotide and polypeptide information about human HLA-E are provided in, for example, the GeneCard Identifier GC06P047281, HGNC No. 4962, NCBI Gene ID 3133, Uniprot No. P13747, and NCBI RefSeq Nos. NP_005507.3 and NM_005516.5. In certain embodiments, the polynucleotide encoding HLA-E is operably linked to a promoter.
[0501] In some embodiments, the polynucleotide encoding HLA-E is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding HLA-E is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding HLA-E is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding HLA- E is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene
editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding HLA-E, into a genomic locus of the cell.
[0502] In some embodiments, HLA-E protein expression is detected using a Western blot of cell lysates probed with antibodies against the HLA-E protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous HLA-E mRNA.
[0503] In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes HLA-G, such as human HLA-G. In some embodiments, HLA-G is overexpressed in the cell. In some embodiments, the expression of HLA-G is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding HLA-G. Useful genomic, polynucleotide and polypeptide information about human HLA-G are provided in, for example, the GeneCard Identifier GC06P047256, HGNC No. 4964, NCBI Gene ID 3135, Uniprot No. P17693, and NCBI RefSeq Nos. NP_002118.1 and NM_002127.5. In certain embodiments, the polynucleotide encoding HLA-G is operably linked to a promoter.
[0504] In some embodiments, the polynucleotide encoding HLA-G is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding HLA-G is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding HLA-G is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding HLA- G is inserted into a B2M gene locus, a CIITA gene locus, or a CD142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding HLA-G, into a genomic locus of the cell.
[05051 In some embodiments, HLA-G protein expression is detected using a Western blot of cell lysates probed with antibodies against the HLA-G protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous HLA-G mRNA.
[0506] In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes PD-L1, such as human PD-LL In some embodiments, PD-L1 is overexpressed in the cell. In some embodiments, the expression of PD-L1 is increased in the modified cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding PD-LL Useful genomic, polynucleotide and polypeptide information about human PD-L1 or CD274 are provided in, for example, the GeneCard Identifier GC09P005450, HGNC No. 17635, NCBI Gene ID 29126, Uniprot No. Q9NZQ7, and NCBI RefSeq Nos. NP_001254635.1, NM_001267706.1, NP_054862.1, and NM_014143.3. In certain embodiments, the polynucleotide encoding PD-L1 is operably linked to a promoter.
[0507] In some embodiments, the polynucleotide encoding PD-L1 is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding PD-L1 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding PD-L1 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding PD-L1 is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding PD-L1, into a genomic locus of the cell.
[0508] In some embodiments, PD-L1 protein expression is detected using a Western blot of cell lysates probed with antibodies against the PD-L1 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous PD-L1 mRNA.
[05091 In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes FasL, such as human FasL. In some embodiments, FasL is overexpressed in the cell. In some embodiments, the expression of FasL is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding FasL. Useful genomic, polynucleotide and polypeptide information about human Fas ligand (which is known as FasL, FASLG, CD 178, TNFSF6, and the like) are provided in, for example, the GeneCard Identifier GC01P172628, HGNC No. 11936, NCBI Gene ID 356, Uniprot No. P48023, and NCBI RefSeq Nos. NP_000630.1, NM_000639.2, NP_001289675.1, and NM_001302746.1. In certain embodiments, the polynucleotide encoding Fas-L is operably linked to a promoter.
[0510] In some embodiments, the polynucleotide encoding Fas-L is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding Fas-L is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding Fas-L is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding Fas-L is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding Fas-L, into a genomic locus of the cell.
[0511] In some embodiments, Fas-L protein expression is detected using a Western blot of cell lysates probed with antibodies against the Fas-L protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous Fas-L mRNA.
[0512] In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CCL21, such as human CCL21. In some embodiments, CCL21 is overexpressed in the cell. In some embodiments, the expression of CCL21 is increased in the
modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CCL21. Useful genomic, polynucleotide and polypeptide information about human CCL21 are provided in, for example, the GeneCard Identifier GC09M034709, HGNC No. 10620, NCBI Gene ID 6366, Uniprot No. 000585, and NCBI RefSeq Nos. NP_002980.1 and NM_002989.3. In certain embodiments, the polynucleotide encoding CCL21 is operably linked to a promoter.
[0513] In some embodiments, the polynucleotide encoding CCL21 is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding CCL21 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding CCL21 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding CCL21 is inserted into a B2M gene locus, a CIITA gene locus, or a CD142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding CCL21, into a genomic locus of the cell.
[0514] In some embodiments, CCL21 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CCL21 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous CCL21 mRNA.
[0515] In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CCL22, such as human CCL22. In some embodiments, CCL22 is overexpressed in the cell. In some embodiments, the expression of CCL22 is increased in the modified cell compared to a similar- reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CCL22. Useful genomic, polynucleotide and polypeptide information about human CCL22 are provided in, for example, the GeneCard Identifier GC16P057359,
HGNC No. 10621, NCBI Gene ID 6367, Uniprot No. 000626, and NCBI RefSeq Nos. NP_002981.2, NM_002990.4, XP_016879020.1, and XM_017023531.1. In certain embodiments, the polynucleotide encoding CCL22 is operably linked to a promoter.
[0516] In some embodiments, the polynucleotide encoding CCL22 is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding CCL22 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding CCL22 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding CCL22 is inserted into a B2M gene locus, a CIITA gene locus, or a CD142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding CCL22, into a genomic locus of the cell.
[0517] In some embodiments, CCL22 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CCL22 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous CCL22 mRNA.
[0518] In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes Mfge8, such as human Mfge8. In some embodiments, Mfge8 is overexpressed in the cell. In some embodiments, the expression of Mfge8 is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding Mfge8. Useful genomic, polynucleotide and polypeptide information about human Mfge8 are provided in, for example, the GeneCard Identifier GC15MO88898, HGNC No. 7036, NCBI Gene ID 4240, Uniprot No. Q08431, and NCBI RefSeq Nos. NP_001108086.1, NM_001114614.2, NP_001297248.1, NM_001310319.1,
NP_001297249.1, NM_001310320.1, NP_001297250.1, NM_001310321.1, NP_005919.2, and
NM_005928.3. In certain embodiments, the polynucleotide encoding Mfge8 is operably linked to a promoter.
[0519] In some embodiments, the polynucleotide encoding Mfge8 is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding Mfge8 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding Mfge8 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding Mfge8 is inserted into a B2M gene locus, a CIITA gene locus, a CD 142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding Mfge8, into a genomic locus of the cell.
[0520] In some embodiments, Mfge8 protein expression is detected using a Western blot of cell lysates probed with antibodies against the Mfge8 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous Mfge8 mRNA.
[0521] In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes SerpinB9, such as human SerpinB9. In some embodiments, SerpinB9 is overexpressed in the cell. In some embodiments, the expression of SerpinB9 is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding SerpinB9. Useful genomic, polynucleotide and polypeptide information about human SerpinB9 are provided in, for example, the GeneCard Identifier GC06M002887, HGNC No. 8955, NCBI Gene ID 5272, Uniprot No. P50453, and NCBI RefSeq Nos. NP_004146.1, NM_004155.5, XP_005249241.1, and XM_005249184.4. In certain embodiments, the polynucleotide encoding SerpinB9 is operably linked to a promoter.
[05221 In some embodiments, the polynucleotide encoding SerpinB9 is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding SerpinB9 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding SerpinB9 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding SerpinB9 is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding SerpinB9, into a genomic locus of the cell.
[0523] In some embodiments, SerpinB9 protein expression is detected using a Western blot of cell lysates probed with antibodies against the SerpinB9 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous SerpinB9 mRNA.
[0524] In some embodiments, the tolerogenic factor is CD47 and the cell includes an exogenous polynucleotide encoding a CD47 protein. In some embodiments, the cell expresses an exogenous CD47 polypeptide.
[0525] In some embodiments, a method disclosed herein comprises administering to a subject in need thereof a CD47-SIRPa blockade agent, wherein the subject was previously administered a population of cells engineered to express an exogenous CD47 polypeptide. In some embodiments, the CD47-SIRPa blockade agent comprises a CD47 -binding domain. In some embodiments, the CD47-binding domain comprises signal regulatory protein alpha (SIRPa) or a fragment thereof. In some embodiments, the CD47-SIRPa blockade agent comprises an immunoglobulin G (IgG) Fc domain. In some embodiments, the IgG Fc domain comprises an IgGl Fc domain. In some embodiments, the IgGl Fc domain comprises a fragment of a human antibody. In some embodiments, the CD47-SIRPa blockade agent is selected from the group consisting of TTI-621, TTI-622, and ALX148. In some embodiments, the CD47- SIRPa blockade agent is TTI-621, TTI-622, and ALX148. In some embodiments, the CD47-
SIRPa blockade agent is TTI-622. In some embodiments, the CD47-SIRPa blockade agent is ALX148. In some embodiments, the IgG Fc domain comprises an IgG4 Fc domain. In some embodiments, the CD47-SIRPa blockade agent is an antibody. In some embodiments, the antibody is selected from the group consisting of M1AP410, B6H12, and Magrolimab. In some embodiments, the antibody is MIAP410. In some embodiments, the antibody is B6H12. In some embodiments, the antibody is Magrolimab. In some embodiments, the antibody is selected from the group consisting of AO- 176, IBI188 (letaplimab), STI-6643, and ZL-1201. In some embodiments, the antibody is AO- 176 (Arch). In some embodiments, the antibody is IBI188 (letaplimab) (Innovent). In some embodiments, the antibody is STI-6643 (Sorrento). In some embodiments, the antibody is ZL-1201 (Zai).
[0526] In some embodiments, useful antibodies or fragments thereof that bind CD47 can be selected from a group that includes magrolimab ((Hu5F9-G4)) (Forty Seven, Inc.; Gilead Sciences, Inc.), urabrelimab, CC-90002 (Celgene; Bristol-Myers Squibb), IBL188 (Innovent Biologies), IBL322 (Innovent Biologies), TG-1801 (TG Therapeutics; also known as NI-1701, Novimmune SA), ALX148 (ALX Oncology), TJ011133 (also known as TJC4, 1-Mab Biopharma), FA3M3, ZL-1201 (Zai Lab Co., Ltd), AK117 (Akesbio Australia Pty, Ltd.), AO- 176 (Arch Oncology), SRF231 (Surface Oncology), GenSci-059 (GeneScience), C47B157 (Janssen Research and Development), C47B161 (Janssen Research and Development), C47B167 (Janssen Research and Development), C47B222 (Janssen Research and Development), C47B227 (Janssen Research and Development), Vx-1004 (Corvus Pharmaceuticals), HMBD004 (Hummingbird Bioscience Pte Ltd), SHR-1603 (Hengrui), AMMS4-G4 (Beijing Institute of Biotechnology), RTX-CD47 (University of Groningen), and IMC-002. (Samsung Biologies; ImmuneOncia Therapeutics). In some embodiments, the antibody or fragment thereof does not compete for CD47 binding with an antibody selected from a group that includes magrolimab, urabrelimab, CC-90002, IBI-188, IBI-322, TG-1801 (NI-1701), ALX148, TJO11133, FA3M3, ZL1201, AK117, AO-176, SRF231, GenSci-059, C47B157, C47B161, C47B167, C47B222, C47B227, Vx-1004, HMBD004, SHR-1603, AMMS4-G4, RTX-CD47, and IMC-002. In some embodiments, the antibody or fragment thereof competes for CD47 binding with an antibody
selected from magrolimab, urabrelimab, CC-90002, IBI-188, IBI-322, TG-1801 (NI-1701), ALX148, TJ011133, FA3M3, ZL1201, AK117, AO-176, SRF231, GenSci-059, C47B157, C47B161, C47B167, C47B222, C47B227, Vx-1004, HMBD004, SHR-1603, AMMS4-G4, RTX-CD47, and 1MC-002. In some embodiments, the antibody or fragment thereof that binds CD47 is selected from a group that includes a single-chain Fv fragment (scFv) against CD47, a Fab against CD47, a VHH nanobody against CD47, a DARPin against CD47, and variants thereof. In some embodiments, the scFv against CD47, a Fab against CD47, and variants thereof are based on the antigen binding domains of any of the antibodies selected from a group that includes magrolimab, urabrelimab, CC-90002, IBI-188, IBI-322, TG-1801 (NI-1701), ALX148, TJO11133, FA3M3, ZL1201, AK117, AO-176, SRF231, GenSci-059, C47B157, C47B161, C47B167, C47B222, C47B227, Vx-1004, HMBD004, SHR-1603, AMMS4-G4, RTX-CD47, and IMC-002.
[0527] In some embodiments, the CD47 antagonist provides CD47 blockade. Methods and agents for CD47 blockade are described in PCT/US2021/054326, which is incorporated by reference in its entirety.
[0528] In some embodiments, the tolerogenic factor (e.g., CD47) is overexpressed in the modified PSC relative to the control or wild-type PSC. In some embodiments, the tolerogenic factor (e.g. CD47) is expressed at a first level that is greater than at or about 3-fold, greater than at or about 5-fold, greater than at or about 10-fold, greater than at or about 20-fold, greater than at or about 30-fold, greater than at or about 40-fold, greater than at or about 50-fold, greater than at or about 60-fold, or greater than at or about 70-fold over a second level expressed by the control or wild-type PSC. In some embodiments, the tolerogenic factor (e.g. CD47) is expressed by the modified PSC at greater than at or about 20,000 molecules per cell, at greater than at or about 30,000 molecules per cell, greater than at or about 50,000 molecules per cell, greater than at or about 100,000 molecules per cell, greater than at or about 200,000 molecules per cell, greater than at or about 300,000 molecules per cell, greater than at or about 400,000 molecules per cell, greater than at or about 500,000 molecules per cell, or greater than at or about 600,000 molecules per cell.
[05291 In some embodiments, the tolerogenic factor (e.g., CD47) is overexpressed in the engineered islet cell relative to the control or wild-type islet cell, such as an unmodified isslet cell differentiated from an unmodified PSC that does not contain the modifications. In some embodiments, the tolerogenic factor (e.g. CD47) is expressed at a first level that is greater than at or about 3-fold, greater than at or about 5-fold, greater than at or about 10-fold, greater than at or about 20-fold, greater than at or about 30-fold, greater than at or about 40-fold, greater than at or about 50-fold, greater than at or about 60-fold, or greater than at or about 70-fold over a second level expressed by the control or wild-type islet cell. In some embodiments, the tolerogenic factor (e.g. CD47) is expressed by the engineered islet cell at greater than at or about 20,000 molecules per cell, at greater than at or about 30,000 molecules per cell, greater than at or about 50,000 molecules per cell, greater than at or about 100,000 molecules per cell, greater than at or about 200,000 molecules per cell, greater than at or about 300,000 molecules per cell, greater than at or about 400,000 molecules per cell, greater than at or about 500,000 molecules per cell, or greater than at or about 600,000 molecules per cell.
[0530] In some embodiments, expression of one or more complement inhibitor is increased in the cell. In some embodiments, the one or more complement inhibitor is one or more membrane-bound complement inhibitor. In some embodiments, at least one of the exogenous polynucleotides includes a polynucleotide that encodes for a complement inhibitor. In some embodiments, the one or more complement inhibitor is CD46, CD59, CD55, or CD35 or any combination thereof.
[0531] In some embodiments, the one or more complement inhibitor is CD46, CD59, CD55, or any combination thereof. For instance, in some embodiments, at least one of the exogenous polynucleotides is a polynucleotide that encodes one or more complement inhibitors, such as CD46. In some embodiments, the one or more complement inhibitors are CD46 and CD59, or CD46, CD59, and CD55. In some embodiments, expression of CD46 and CD59 or CD46, CD59, and CD55 protects a cell or population thereof from complement-dependent cytotoxicity, including in the presence of antibodies against cell surface antigens expressed by the cell.
[05321 In some embodiments, the present disclosure provides a cell or population thereof that has been modified to express the one or more complement inhibitor, such as CD46, CD59, CD55, or any combination thereof. In some embodiments, the one or more complement inhibitor is CD46 and CD59. In some embodiments, the one or more complement inhibitor is CD46, CD59, and CD55. In some embodiments, the present disclosure provides a method for altering a cell genome to express one or more complement inhibitor. In some embodiments, the modified cell expresses one or more exogenous complement inhibitor, such as exogenous CD46 and CD59 or CD46, CD59, and CD55. In some instances, the cell expresses an expression vector comprising a nucleotide sequence encoding a human CD46 polypeptide. In some instances, the cell expresses an expression vector comprising a nucleotide sequence encoding a human CD59 polypeptide. In some instances, the cell expresses an expression vector comprising a nucleotide sequence encoding a human CD55 polypeptide. In some embodiments, the expression vector comprises nucleotide sequences encoding two or more complement inhibitors in any combination. In some embodiments, the expression vector comprises nucleotide sequences encoding CD46 and CD59. In some embodiments, the expression vector comprises nucleotide sequences encoding CD46, CD59, and CD55.
[0533] In some embodiments, the modified pluripotent stem cells contain an overexpressed polynucleotide that encodes CD46, such as human CD46. In some embodiments, the modified pluripotent stem cells contain an exogenous polynucleotide that encodes CD46, such as human CD46. In some embodiments, CD46 is overexpressed in the cell. In some embodiments, the expression of CD46 is increased in the modified pluripotent stem cells compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CD46. CD46 is a membrane-bound complement inhibitor. It acts as a cofactor for complement factor I, a serine protease which protects autologous cells against complement- mediated injury by cleaving C3b and C4b. Useful genomic, polynucleotide and polypeptide information about human CD46 are provided in, for example, the GeneCard Identifier GC01P207752, HGNC No. 6953, NCBI Gene ID 4179, Uniprot No. P15529, and NCBI Ref Scq
Nos. NM_002389.4, NM_153826.3, NM_172350.2, NM_172351.2, NM_172352.2 NP_758860.1, NM_172353.2, NM_172359.2, NM_172361.2, NP_002380.3, NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1.
[0534] In some embodiments, the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD46 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_002380.3, NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1. In some embodiments, the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD46 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_002380.3, NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1. In some embodiments, the cell comprises an overexpressed nucleotide sequence for CD46 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_002389.4, NM_153826.3, NM_172350.2, NM_172351.2, NM_172352.2 NP_758860.1, NM_172353.2, NM_172359.2, and NM_172361.2. In some embodiments, the cell comprises an overexpressed nucleotide sequence for CD46 as set forth in NCBI Ref. Sequence Nos. NM_001777.3 and NM_002389.4, NM_153826.3, NM_172350.2, NM_172351.2, NM_172352.2 NP_758860.1, NM_172353.2, NM_172359.2, and NM_172361.2.
[0535] In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_002380.3, NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1. In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_002380.3, NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1. In some embodiments, the cell comprises an exogenous nucleotide sequence for CD46 having at least
85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_002389.4, NM_153826.3, NM_172350.2, NM_172351.2, NM_172352.2 NP_758860.1, NM_172353.2, NM_172359.2, and NM_172361.2. In some embodiments, the cell comprises an exogenous nucleotide sequence for CD46 as set forth in NCBI Ref. Sequence Nos. NM_001777.3 and NM_002389.4, NM_153826.3, NM_172350.2, NM_172351.2, NM_172352.2 NP_758860.1, NM_172353.2, NM_172359.2, and NM_172361 .2.
[0536] In some embodiments, the cell comprises an overexpressed CD46 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1. In some embodiments, the cell comprises an exogenous CD46 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1. In some embodiments, the cell outlined herein comprises an overexpressed CD46 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1. In some embodiments, the cell outlined herein comprises an exogenous CD46 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_722548.1, NP_758860.1, NP_758861.1, NP_758862.1, NP_758863.1, NP_758869.1, and NP_758871.1.
[0537] In some embodiments, a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD46 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 4. In some embodiments, a cell outlined herein comprises an overexpressed nucleotide sequence
encoding a CD46 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 4.
In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 4.
[0538] In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 3. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD46 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 3. In some embodiments, the exogenous nucleotide sequence encoding the CD46 polypeptide is operably linked to a sequence encoding a heterologous signal peptide.
[0539] In some embodiments, all or a functional portion of CD46 can be linked to other components such as a signal peptide, a leader sequence, a secretory signal, a label (e.g., a reporter gene), or any combination thereof. In some embodiments, the nucleic acid sequence encoding a signal peptide of CD46 is replaced with a nucleic acid sequence encoding a signal peptide from a heterologous protein. The heterologous protein can be, for example, CD8ot, CD28, tissue plasminogen activator (tPA), growth hormone, granulocyte-macrophage colony stimulating factor (GM-CSF), GM-CSF receptor (GM-CSFRa), or an immunoglobulin (e.g., IgE or IgK). In some embodiments, the signal peptide is a signal peptide from an immunoglobulin (such as IgG heavy chain or IgG-kappa light chain), a cytokine (such as interleukin-2 (IL-2), or CD33), a serum albumin protein (e.g. HSA or albumin), a human azurocidin preprotein signal sequence, a luciferase, a trypsinogen (e.g., chymotrypsinogen or trypsinogen) or other signal peptide able to efficiently express a protein by or on a cell.
[0540] In certain embodiments, the exogenous polynucleotide encoding CD46 is operably linked to a promoter.
[0541] In some embodiments, the polynucleotide encoding CD46 is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding CD46 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1,
CCR5, CLYBL, ROSA26, SHS231. In particular embodiments, the polynucleotide encoding CD46 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding CD46 is inserted into a B2M gene locus, a C11TA gene locus, or a CD 142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding CD46, into a genomic locus of the cell.
[0542] In some embodiments, CD46 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CD46 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous CD46 mRNA.
[0543] In some embodiments, the modified pluripotent stem cell contains an overexpressed polynucleotide that encodes CD59, such as human CD59. In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD59, such as human CD59. In some embodiments, CD59 is overexpressed in the cell. In some embodiments, the expression of CD59 is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CD59. CD59 is a membrane-bound complement inhibitor. More specifically, CD59 is an inhibitor of complement membrane attack complex (MAC) activity. CD59 acts by binding to the C8 and/or C9 complements of the assembling MAC, thereby preventing incorporation of the multiple copies of C9 required for complete formation of the osmolytic pore. Useful genomic, polynucleotide and polypeptide information about human CD59 are provided in, for example, the GeneCard Identifier GC11M033704, HGNC No. 1689, NCBI Gene ID 966, Uniprot No.
Pl 3987, and NCBI RefSeq Nos. NP_000602.1 , NM_000611 .5, NP_001120695. 1 , NM_001127223.1, NP_001120697.1, NM_001127225.1, NP_001120698.1, NM_001127226.1, NP_001120699.1, NM_001127227.1, NP_976074.1, NM_203329.2, NP_976075.1, NM_203330.2, NP_976076.1, and NM_203331.2.
[05441 In some embodiments, the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. Nos. NP-000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1,
NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1. In some embodiments, the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos.
NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1. In some embodiments, the cell comprises an overexpressed nucleotide sequence for CD59 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_000611.5, NM_001127223.1, NM_001127225.1, NM_001127226.1, NM_001127227.1, NM_203329.2, NM_203330.2, and NM_203331.2. In some embodiments, the cell comprises an overexpressed nucleotide sequence for CD59 as set forth in NCBI Ref. Sequence Nos. NM_000611.5, NM_001127223.1, NM_001127225.1, NM_001127226.1, NM_001127227.1, NM_203329.2, NM_203330.2, and NM_203331.2.
[0545] In some embodiments, the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1,
NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1. In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1. In some embodiments, the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000602.1, NP_001120695.1, NP_001120697.1,
NP_001120698.1, NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1. In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1. In some embodiments, the cell comprises an overexpressed nucleotide sequence for CD59 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_000611.5, NM_001127223.1, NM_001127225.1, NM_001127226.1, NM_001127227.1, NM_203329.2, NM_203330.2, and NM_203331.2. In some embodiments, the cell comprises an exogenous nucleotide sequence for CD59 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos.
NM_000611.5, NM_001127223.1, NM_001127225.1, NM_001127226.1, NM_001127227.1, NM_203329.2, NM_203330.2, and NM_203331.2. In some embodiments, the cell comprises an overexpressed nucleotide sequence for CD59 as set forth in NCBI Ref. Sequence Nos.
NM_000611.5, NM_001127223.1, NM_001127225.1, NM_001127226.1, NM_001127227.1, NM_203329.2, NM_203330.2, and NM_203331.2. In some embodiments, the cell comprises an exogenous nucleotide sequence for CD59 as set forth in NCBI Ref. Sequence Nos.
NM_000611.5, NM_001127223.1, NM_001127225.1, NM_001127226.1, NM_001127227.1, NM_203329.2, NM_203330.2, and NM_203331.2.
[0546] In some embodiments, the cell comprises an overexpressed CD59 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1. In some embodiments, the cell comprises an exogenous CD59 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1, NP_976074.1, NP_976075.1, and
NP_976076.1. In some embodiments, the cell outlined herein comprises an overexpressed CD59 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos.
NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1, NP_976074.1, NP_976075.1, and NP_976076.1. In some embodiments, the cell outlined herein comprises an exogenous CD59 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000602.1, NP_001120695.1, NP_001120697.1, NP_001120698.1, NP_001120699.1 , NP_976074.1 , NP_976075.1 , and NP_976076.1.
[0547] In some embodiments, a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 6. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 6. In some embodiments, a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 6. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 6.
[0548] In some embodiments, a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD59 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 5. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD59 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 5.
In some embodiments, the exogenous nucleotide sequence encoding the CD59 polypeptide is operably linked to a sequence encoding a heterologous signal peptide.
[0549] In some embodiments, all or a functional portion of CD59 can be linked to other components such as a signal peptide, a leader sequence, a secretory signal, a label (e.g., a reporter gene), or any combination thereof. In some embodiments, the nucleic acid sequence encoding a signal peptide of CD59 is replaced with a nucleic acid sequence encoding a signal peptide from a heterologous protein. The heterologous protein can be, for example, CD8a, CD28, tissue plasminogen activator (tPA), growth hormone, granulocyte-macrophage colony stimulating factor (GM-CSF), GM-CSF receptor (GM-CSFRa), or an immunoglobulin (e.g., IgE or IgK). In some embodiments, the signal peptide is a signal peptide from an immunoglobulin (such as IgG heavy chain or IgG-kappa light chain), a cytokine (such as interleukin-2 (IL-2), or CD33), a serum albumin protein (e.g., HSA or albumin), a human azurocidin preprotein signal sequence, a luciferase, a trypsinogen (e.g. chymotrypsinogen or trypsinogen) or other signal peptide able to efficiently express a protein by or on a cell.
[0550] In certain embodiments, the exogenous polynucleotide encoding CD59 is operably linked to a promoter.
[0551] In some embodiments, the polynucleotide encoding CD59 is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding CD59 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding CD59 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding CD59 is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding CD59, into a genomic locus of the cell.
[05521 In some embodiments, CD59 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CD59 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) are used to confirm the presence of the exogenous CD59 mRNA.
[0553] In some embodiments, the modified pluripotent stem cell contains an overexpressed polynucleotide that encodes CD55, such as human CD55. In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD55, such as human CD55. In some embodiments, CD55 is overexpressed in the cell. In some embodiments, the expression of CD55 is increased in the modified pluripotent stem cell compared to a similar reference or unmodified cell (including with any other modifications) except that the reference or unmodified cell does not include the exogenous polynucleotide encoding CD55. CD55 is a membrane-bound complement inhibitor. In some embodiments, interaction of CD55 with cell-associated C4b and C3b polypeptides interferes with their ability to catalyze the conversion of C2 and factor B to enzymatically active C2a and Bb and thereby prevents the formation of C4b2a and C3bBb, the amplification convertases of the complement cascade. In some embodiments, CD55 inhibits complement activation by destabilizing and preventing the formation of C3 and C5 convertases. Useful genomic, polynucleotide and polypeptide information about human CD55 (also known as complement decay-accelerating factor) are provided in, for example, the GeneCard Identifier GC01P207321, HGNC No. 2665, NCBI Gene ID 1604, Uniprot No. P08174, and NCBI RefSeq Nos. NM_000574.4,
NM_001114752.2, NM_001300903.1, NM_001300904.1, NP_000565.I, NP_001108224.1, NP_001287832.1, and NP_001287833.1.
[0554] In some embodiments, the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide that has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref.
Sequence Nos. NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1. In some embodiments, the cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide having an amino acid sequence as set forth in NCBI Ref.
Sequence Nos. NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1. In some embodiments, the cell comprises an overexpressed nucleotide sequence for CD55 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCB1 Ref. Nos. NM_001777.3 and NM_198793.2. In some embodiments, the cell comprises an overexpressed nucleotide sequence for CD55 as set forth in NCBI Ref. Sequence Nos. NM_000574.4, NM_001114752.2, NM_001300903.1 , and NM_001300904.1 .
[0555] In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide that has at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos.
NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1. In some embodiments, the cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos.
NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1. In some embodiments, the cell comprises an exogenous nucleotide sequence for CD55 having at least 85% sequence identity (e.g., 85%, 86%, 87%, 88%, 89%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, 99%, or more) to the sequence set forth in NCBI Ref. Nos. NM_001777.3 and NM_198793.2. In some embodiments, the cell comprises an exogenous nucleotide sequence for CD55 as set forth in NCBI Ref. Sequence Nos. NM_000574.4, NM_001114752.2, NM_001300903.1, and NM_001300904.1.
[0556] In some embodiments, the cell comprises an overexpressed CD55 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1. In some embodiments, the cell comprises an exogenous CD55 polypeptide having at least 95% sequence identity (e.g., 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in NCBI Ref. Sequence Nos.
NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1. In some embodiments, the cell outlined herein comprises an overexpressed CD55 polypeptide having an
amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1. In some embodiments, the cell outlined herein comprises an exogenous CD55 polypeptide having an amino acid sequence as set forth in NCBI Ref. Sequence Nos. NP_000565.1, NP_001108224.1, NP_001287832.1, and NP_001287833.1.
[0557] In some embodiments, a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 9. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 9. In some embodiments, a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 9. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 9.
[0558] In some embodiments, a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 8. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide that has at least 85% sequence identity (e.g., 85%, 90%, 95%, 96%, 97%, 98%, 99%, or more) to an amino acid sequence as set forth in SEQ ID NO: 8. In some embodiments, a cell outlined herein comprises an overexpressed nucleotide sequence encoding a CD55 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 8. In some embodiments, a cell outlined herein comprises an exogenous nucleotide sequence encoding a CD55 polypeptide comprising an amino acid sequence as set forth in SEQ ID NO: 8. In some embodiments, the exogenous nucleotide sequence encoding the CD59 polypeptide is operably linked to a sequence encoding a heterologous signal peptide.
[0559] In some embodiments, all or a functional portion of CD55 can be linked to other components such as a signal peptide, a leader sequence, a secretory signal, a label (e.g., a
reporter gene), or any combination thereof. In some embodiments, the nucleic acid sequence encoding a signal peptide of CD55 is replaced with a nucleic acid sequence encoding a signal peptide from a heterologous protein. The heterologous protein can be, for example, CD8a, CD28, tissue plasminogen activator (tPA), growth hormone, granulocyte-macrophage colony stimulating factor (GM-CSF), GM-CSF receptor (GM-CSFRa), or an immunoglobulin (e.g., IgE or IgK). In some embodiments, the signal peptide is a signal peptide from an immunoglobulin (such as IgG heavy chain or IgG-kappa light chain), a cytokine (such as interleukin-2 (IL-2), or CD33), a serum albumin protein (e.g. HSA or albumin), a human azurocidin preprotein signal sequence, a luciferase, a trypsinogen (e.g., chymotrypsinogen or trypsinogen) or other signal peptide able to efficiently express a protein by or on a cell.
[0560] In certain embodiments, the exogenous polynucleotide encoding CD55 is operably linked to a promoter.
[0561] In some embodiments, the polynucleotide encoding CD55 is inserted into any one of the gene loci depicted in Table 6. In some cases, the polynucleotide encoding CD55 is inserted into a safe harbor locus, such as but not limited to, a gene locus selected from AAVS1, CCR5, CLYBL, ROSA26, and SHS231. In particular embodiments, the polynucleotide encoding CD55 is inserted into the CCR5 gene locus, the PPP1R12C (also known as AAVS1) gene locus or the CLYBL gene locus. In some embodiments, the polynucleotide encoding CD55 is inserted into a B2M gene locus, a CIITA gene locus, or a CD 142 gene locus. In some embodiments, a suitable gene editing system (e.g., CRISPR/Cas system or any of the gene editing systems described herein) is used to facilitate the insertion of a polynucleotide encoding CD55, into a genomic locus of the cell.
[0562] In some embodiments, CD55 protein expression is detected using a Western blot of cell lysates probed with antibodies against the CD55 protein. In another embodiment, reverse transcriptase polymerase chain reactions (RT-PCR) arc used to confirm the presence of the exogenous CD55 mRNA.
[05631 In some embodiments, the cell comprises increased expression of none, one, two, or more complement inhibitors selected from the group consisting of CD46, CD59, and CD55, in any combination.
[0564] In some embodiments, the modified pluripotent stem cell contains an overexpressed polynucleotide that encodes CD46, such as any described above, and an overexpressed polynucleotide that encodes CD59, such as any described above.
[0565] In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD46, such as any described above, and an exogenous polynucleotide that encodes CD59, such as any described above.
[0566] In some embodiments, the modified cell (comprising one or more modifications that increase expression of CD46 and CD59) comprises increased expression of CD46 and CD59 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46 and CD59). In some embodiments, the modified pluripotent stem cell comprises between 1.5-fold and 2-fold, between 2-fold and 3-fold, between 3-fold and 4-fold, between 4-fold and 5-fold, between 5-fold and 10-fold, between 10-fold and 15-fold, between 15-fold and 20-fold, between 20-fold and 40-fold, between 40-fold and 60-fold, between 60-fold and 80-fold, between 80-fold and 100-fold, or between 100-fold and 200-fold increased expression of CD46 and CD59 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46 and CD59). In some embodiments, the cell without the modification(s) does not have endogenous expression of CD46 and CD59 or does not have detectable expression of CD46 and CD59. In some embodiments, the fold increase in expression compared to a cell lacking the modifications is greater than 200-fold.
[0567] In some embodiments, the modified pluripotent stem cells (comprising one or more modifications that increase expression of CD46 and CD59) comprises between 2-fold and 200-fold, between 2-fold and 100-fold, between 2-fold and 50-fold, or between 2-fold and 20- fold increased expression of CD46 and CD59 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46 and CD59). In some
embodiments, the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46 and CD59) comprises between 5-fold and 200-fold, between 5-fold and 100-fold, between 5-fold and 50-fold, or between 5-fold and 20-fold increased expression of CD46 and CD59 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46 and CD59).
[0568] In some embodiments, the modified pluripotent stem cells (comprising one or more modifications that increase expression of CD46 and CD59) comprises increased expression of CD46 and CD59 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46 and CD59). In some embodiments, the modified pluripotent stem cell comprises at least at or about 2-fold, at least at or about 4-fold, at least at or about 6- fold, at least at or about 10-fold, at least at or about and 15-fold, at least at or about 20-fold, at least at or about 30-fold, at least at or about 50-fold, at least at or about 60-fold, at least at or about 70-fold, at least at or about 80-fold, at least at or about 100-fold, or any value between any of the foregoing values, increased expression of CD46 and CD59 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46 and CD59).
[0569] In some embodiments, the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46 and CD59) comprises increased expression of CD46 and CD59 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46 and CD59). In some embodiments, the modified cell comprises at or about 2-fold, at or about 4-fold, at or about 6-fold, at or about 10-fold, at or about and 15- fold, at or about 20-fold, at or about 30-fold, at or about 50-fold, at or about 60-fold, at or about 70-fold, at or about 80-fold, at or about 100-fold, or any value between any of the foregoing values, increased expression of CD46 and CD59 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46 and CD59).
[0570] In some embodiments, the cell comprises one or more transgenes encoding the CD46 and CD59. In some embodiments, the transgenes are monocistronic or multicistronic vectors, as described below. In some embodiments, the CD46 and CD59 are comprised by the same multicistronic vector, optionally in combination with one or more tolerogenic factors such
as CD47. In some embodiments, the CD46 and CD59 are comprised by different transgenes, optionally in combination with one or more tolerogenic factors such as CD47.
[0571] In some embodiments, the modified pluripotent stem cell contains an overexpressed polynucleotide that encodes CD46, such as any described above, an overexpressed polynucleotide that encodes CD59, such as any described above, and an overexpressed polynucleotide that encodes CD55, such as any described above.
[0572] In some embodiments, the modified pluripotent stem cell contains an exogenous polynucleotide that encodes CD46, such as any described above, an exogenous polynucleotide that encodes CD59, such as any described above, and an exogenous polynucleotide that encodes CD55, such as any described above.
[0573] In some embodiments, the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46, CD59, and CD55) comprises increased expression of CD46, CD59, and CD55 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46, CD59, and CD55). In some embodiments, the modified cell comprises between 1 .5-fold and 2-fold, between 2-fold and 3-fold, between 3-fold and 4-fold, between 4-fold and 5-fold, between 5-fold and 10-fold, between 10- fold and 15-fold, between 15-fold and 20-fold, between 20-fold and 40-fold, between 40-fold and 60-fold, between 60-fold and 80-fold, between 80-fold and 100-fold, or between 100-fold and 200-fold increased expression of CD46, CD59, and CD55 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46, CD59, and CD55). In some embodiments, the cell without the modification(s) does not have endogenous expression of CD46, CD59, and CD55or does not have detectable expression of CD46, CD59, and CD55. In some embodiments, the fold increase in expression compared to a cell lacking the modifications is greater than 200-fold.
[0574] In some embodiments, the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46, CD59, and CD55) comprises between 2- fold and 200-fold, between 2-fold and 100- fold, between 2-fold and 50-fold, or between 2-fold
and 20-fold increased expression of CD46, CD59, and CD55 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46, CD59, and CD55). In some embodiments, the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46, CD59, and CD55) comprises between 5-fold and 200-fold, between 5-fold and 100-fold, between 5-fold and 50-fold, or between 5-fold and 20-fold increased expression of CD46, CD59, and CD55 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46, CD59, and CD55).
[0575] In some embodiments, the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46, CD59, and CD55) comprises increased expression of CD46, CD59, and CD55 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46 and CD59). In some embodiments, the modified cell comprises at least at or about 2-fold, at least at or about 4-fold, at least at or about 6-fold, at least at or about 10-fold, at least at or about and 15-fold, at least at or about 20-fold, at least at or about 30-fold, at least at or about 50-fold, at least at or about 60-fold, at least at or about 70-fold, at least at or about 80-fold, at least at or about 100-fold, or any value between any of the foregoing values, increased expression of CD46, CD59, and CD55 compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46, CD59, and CD55).
[0576] In some embodiments, the modified pluripotent stem cell (comprising one or more modifications that increase expression of CD46, CD59, and CD55) comprises increased expression of CD46, CD59, and CD55 relative to a cell that does not comprise the modifications (e.g., relative to endogenous expression of CD46, CD59, and CD55). In some embodiments, the modified cell comprises at or about 2-fold, at or about 4-fold, at or about 6-fold, at or about 10- fold, at or about and 15-fold, at or about 20-fold, at or about 30-fold, at or about 50-fold, at or about 60-fold, at or about 70-fold, at or about 80-fold, at or about 100-fold, or any value between any of the foregoing values, increased expression of CD46, CD59, and CD55compared to a cell that does not have the modifications (e.g., compared to endogenous expression of CD46, CD59, and CD55).
[05771 In some embodiments, the cell comprises one or more transgenes encoding the CD46, CD59, and CD55. In some embodiments, the transgenes are monocistronic or multicistronic vectors, as described below. In some embodiments, the CD46, CD59, and CD55 are comprised by the same multicistronic vector, optionally in combination with one or more tolerogenic factors such as CD47. In some embodiments, the CD46, CD59, and CD55 are comprised by different transgenes, optionally in combination with one or more tolerogenic factors such as CD47. e. Modification Order and Timing
[0578] In some embodiments, provided modified pluripotent stem cells comprise a modification (e.g., genetic modifications). Modification (e.g., genetic modifications) in an iPSCs can be introduced at various time points and/or stages in the generation of a modified iPSC and/or a cell derived from a modified iPSC (e.g., a hypoimmune islet cell).
[0579] For example, in some embodiments, all modifications present in a hypoimmune differentiated cell (e.g., hypoimmune islet cell) are present in an iPSC that the hypoimmune islet cell is derived from. In some embodiments, a hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises one or more modifications in addition to the one or more modifications present in the iPSC the islet cell is derived from. In some embodiments, a hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises one or more modifications, none of which are present in the iPSC the hypoimmune islet cell is derived from.
[0580] In some embodiments, a hypoimmune iPSC comprising one or more modifications is differentiated into hypoimmune differentiated cell (e.g., hypoimmune islet cell), wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises no additional modifications relative to the hypoimmune iPSC from which it is derived. In some embodiments, an iPSC comprising one or more modifications is differentiated into a hypoimmune differentiated cell (e.g., hypoimmune islet cell), wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises one or more additional modifications relative to the iPSC from which it is derived. In some embodiments, an iPSC comprising no hypoimmune modifications is differentiated into a hypoimmune differentiated cell (e.g.,
hypoimmune islet cell), wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) is modified to comprise one or more modifications relative to the iPSC from which it is derived.
[0581] In some embodiments, the present disclosure provides methods of creating hypoimmune differentiated cell (e.g., hypoimmune islet cell) derived from iPSCs. In some embodiments, the iPSCs are hypoimmune iPSCs. In some embodiments, a method comprises engineering an iPSC to generate a hypoimmune iPSC comprising one or more modifications, and differentiating the hypoimmune iPSC into a hypoimmune differentiated cell (e.g., hypoimmune islet cell), wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises the same one or more modifications as the hypoimmune iPSC. In some embodiments, a method comprises engineering an iPSC to generate an iPSC comprising one or more modifications, differentiating the iPSC into a hypoimmune differentiated cell (e.g., hypoimmune islet cell), and then engineering the hypoimmune differentiated cell (e.g., hypoimmune islet cell) to comprise one or more additional modifications, wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises one or more additional modifications relative to the iPSC. In some embodiments, a method comprises differentiating an iPSC into an islet cell and then engineering the islet cell to comprise one or more modifications, thereby generating a hypoimmune differentiated cell (e.g., hypoimmune islet cell), wherein the hypoimmune differentiated cell (e.g., hypoimmune islet cell) comprises one or more modifications relative to the iPSC. f. Methods of Increasing Expression of (e.g. overexpressing) a Polynucleotide
[0582] In some embodiments, increased expression of a polynucleotide may be carried out by any of a variety of techniques. For instance, methods for modulating expression of genes and factors (proteins) include genome editing technologies, and RNA or protein expression technologies and the like. For all of these technologies, well known recombinant techniques are used, to generate recombinant nucleic acids as outlined herein. In some embodiments, the cell
that is modified with the one or more modification for overexpression or increased expression of a polynucleotide is any source cell as described herein.
DNA-binding Fusion Proteins
[0583] In some embodiments, expression of a target gene (e.g., CD47, or another tolerogenic factor) is increased by expression of fusion protein or a protein complex containing (1) a site-specific binding domain specific for the endogenous CD47, or other gene and (2) a transcriptional activator.
[0584] In some embodiments, the regulatory factor is comprised of a site-specific DNA- binding nucleic acid molecule, such as a guide RNA (gRNA). In some embodiments, the method is achieved by site specific DNA-binding targeted proteins, such as zinc finger proteins (ZFP) or fusion proteins containing ZFP, which are also known as zinc finger nucleases (ZFNs).
[0585] In some embodiments, the regulatory factor comprises a site-specific binding domain, such as using a DNA binding protein or DNA-binding nucleic acid, which specifically binds to or hybridizes to the gene at a targeted region. In some embodiments, the provided polynucleotides or polypeptides are coupled to or complexed with a site-specific nuclease, such as a modified nuclease. For example, in some embodiments, the administration is effected using a fusion comprising a DNA-targeting protein of a modified nuclease, such as a meganuclease or an RNA-guided nuclease such as a clustered regularly interspersed short palindromic nucleic acid (CRISPR)-Cas system, such as CRISPR-Cas9 system. In some embodiments, the nuclease is modified to lack nuclease activity. In some embodiments, the modified nuclease is a catalytically dead dCas9.
[0586] In some embodiments, the site specific binding domain may be derived from a nuclease. For example, the recognition sequences of homing endonucleases and meganucleases such as I-Scel, I-Ceul, PI-PspI, Pl-Sce, I-SceIV, I-CsmI, I-PanI, I-SceII, I-Ppol, I-SceIII, I-Crel, I-TevI, I-TevII and I-TevIII. See also U.S. Patent No. 5,420,032; U.S. Patent No. 6,833,252; Belfort et al., (1997) Nucleic Acids Res. 25:3379-3388; Dujon et al., (1989) Gene 82:115-118; Perler et al, (1994) Nucleic Acids Res. 22, 1125-1127; Jasin (1996) Trends Genet. 12:224-228;
Gimble et al., (1996) J. Mol. Biol. 263:163-180; Argast et al, (1998) J. Mol. Biol. 280:345-353 and the New England Biolabs catalogue. In addition, the DNA-binding specificity of homing endonucleases and meganucleases can be modified to bind non-natural target sites. See, for example, Chevalier et al, (2002) Molec. Cell 10:895-905; Epinat et al, (2003) Nucleic Acids Res. 31 :2952-2962; Ashworth et al, (2006) Nature 441 :656-659; Paques et al, (2007) Current Gene Therapy 7:49-66; U.S. Patent Publication No. 2007/0117128.
[0587] Zinc finger, TALE, and CRISPR system binding domains can be “engineered” to bind to a predetermined nucleotide sequence, for example via engineering (altering one or more amino acids) of the recognition helix region of a naturally occurring zinc finger or TALE protein. Engineered DNA binding proteins (zinc fingers or TALEs) are proteins that are non-naturally occurring. Rational criteria for design include application of substitution rules and computerized algorithms for processing information in a database storing information of existing ZFP and/or TALE designs and binding data. See, for example, U.S. Pat. Nos. 6,140,081; 6,453,242; and 6,534,261; see also WO 98/53058; WO 98/53059; WO 98/53060; WO 02/016536 and WO 03/016496 and U.S. Publication No. 20110301073.
[0588] In some embodiments, the site-specific binding domain comprises one or more zinc-finger proteins (ZFPs) or domains thereof that bind to DNA in a sequence- specific manner. A ZFP or domain thereof is a protein or domain within a larger protein that binds DNA in a sequence-specific manner through one or more zinc fingers, regions of amino acid sequence within the binding domain whose structure is stabilized through coordination of a zinc ion.
[0589] Among the ZFPs are artificial ZFP domains targeting specific DNA sequences, typically 9-18 nucleotides long, generated by assembly of individual fingers. ZFPs include those in which a single finger domain is approximately 30 amino acids in length and contains an alpha helix containing two invariant histidine residues coordinated through zinc with two cysteines of a single beta turn, and having two, three, four, five, or six fingers. Generally, sequence-specificity of a ZFP may be altered by making amino acid substitutions at the four helix positions (-1, 2, 3 and 6) on a zinc finger recognition helix. Thus, in some embodiments, the ZFP or ZFP- containing molecule is non-naturally occurring, e.g., is engineered to bind to a target site of
choice. See, for example, Beerli et al. (2002) Nature Biotechnol. 20:135-141; Pabo et al. (2001) Ann. Rev. Biochem. 70:313-340; Isalan et al. (2001) Nature Biotechnol. 19:656-660; Segal et al. (2001) Curr. Opin. Biotechnol. 12:632-637; Choo et al. (2000) Curr. Opin. Struct. Biol. 10:411- 416; U.S. Pat. Nos. 6,453,242; 6,534,261; 6,599,692; 6,503,717; 6,689,558; 7,030,215;
6,794,136; 7,067,317; 7,262,054; 7,070,934; 7,361,635; 7,253,273; and U.S. Patent Publication Nos. 2005/0064474; 2007/0218528; 2005/0267061, all incorporated herein by reference in their entireties.
[0590] Many gene-specific engineered zinc fingers are available commercially. For example, Sangamo Biosciences (Richmond, CA, USA) has developed a platform (CompoZr) for zinc-finger construction in partnership with Sigma-Aldrich (St. Louis, MO, USA), allowing investigators to bypass zinc-finger construction and validation altogether, and provides specifically targeted zinc fingers for thousands of proteins (Gaj et al., Trends in Biotechnology, 2013, 31(7), 397-405). In some embodiments, commercially available zinc fingers are used or are custom designed.
[0591] In some embodiments, the site-specific binding domain comprises a naturally occurring or engineered (non-naturally occurring) transcription activator-like protein (TAL) DNA binding domain, such as in a transcription activator-like protein effector (TALE) protein, See, e.g., U.S. Patent Publication No. 20110301073, incorporated by reference in its entirety herein.
[0592] In some embodiments, the site-specific binding domain is derived from the CRISPR/Cas system. In general, “CRISPR system” refers collectively to transcripts and other elements involved in the expression of or directing the activity of CRIS PR-associated (“Cas”) genes, including sequences encoding a Cas gene, a tracr (trans-activating CRISPR) sequence (e.g. tracrRNA or an active partial tracrRNA), a tracr-mate sequence (encompassing a “direct repeat” and a tracrRNA-processed partial direct repeat in the context of an endogenous CRISPR system), a guide sequence (also referred to as a “spacer” in the context of an endogenous CRISPR system, or a “targeting sequence”), and/or other sequences and transcripts from a CRISPR locus.
[05931 In general, a guide sequence includes a targeting domain comprising a polynucleotide sequence having sufficient complementarity with a target polynucleotide sequence to hybridize with the target sequence and direct sequence-specific binding of the CR1SPR complex to the target sequence. In some embodiments, the degree of complementarity between a guide sequence and its corresponding target sequence, when optimally aligned using a suitable alignment algorithm, is about or more than about 50%, 60%, 75%, 80%, 85%, 90%, 95%, 97.5%, 99%, or more. Tn some examples, the targeting domain (e.g., targeting sequence) of the gRNA is complementary, e.g., at least 80, 85, 90, 95, 98 or 99% complementary, e.g., fully complementary, to the target sequence on the target nucleic acid.
[0594] In some embodiments, the gRNA may be any as described herein. In particular embodiments, the gRNA has a targeting sequence that is complementary to a target site of CD47, such as set forth in any one of SEQ ID NOS:200784-231885 (Table 29, Appendix 22 of W02016183041); HLA-E, such as set forth in any one of SEQ ID NOS:189859-193183 (Table 19, Appendix 12 of W02016183041); HLA-F, such as set forth in any one of SEQ ID NOS: 688808-699754 (Table 45, Appendix 38 of W02016183041); HLA-G, such as set forth in any one of SEQ ID NOS: 188372-189858 (Table 18, Appendix 11 of WO2016183041); or PD-L1, such as set forth in any one of SEQ ID NOS: 193184-200783 (Table 21, Appendix 14 of W02016183041).
[0595] In some embodiments, the target site is upstream of a transcription initiation site of the target gene. In some embodiments, the target site is adjacent to a transcription initiation site of the gene. In some embodiments, the target site is adjacent to an RNA polymerase pause site downstream of a transcription initiation site of the gene.
[0596] In some embodiments, the targeting domain is configured to target the promoter region of the target gene to promote transcription initiation, binding of one or more transcription enhancers or activators, and/or RNA polymerase. One or more gRNA can be used to target the promoter region of the gene. In some embodiments, one or more regions of the gene can be targeted. In certain aspects, the target sites are within 600 base pairs on either side of a transcription start site (TSS) of the gene.
[05971 It is within the level of a skilled artisan to design or identify a gRNA sequence that is or comprises a sequence targeting a gene (i.e., gRNA targeting sequence), including the exon sequence and sequences of regulatory regions, including promoters and activators. A genome-wide gRNA database for CR1SPR genome editing is publicly available, which contains exemplary single guide RNA (sgRNA) target sequences in constitutive exons of genes in the human genome or mouse genome (see e.g., genescript.com/gRNA-database.html; see also, Sanjana et al. (2014) Nat. Methods, 11 :783-4; www.e-crisp.org/E-CRISP/; crispr.mit.edu/). In some embodiments, the gRNA sequence is or comprises a targeting sequence with minimal off- target binding to a non-target gene.
[0598] In some embodiments, the regulatory factor further comprises a functional domain, e.g., a transcriptional activator.
[0599] In some embodiments, the transcriptional activator is or contains one or more regulatory elements, such as one or more transcriptional control elements of a target gene, whereby a site-specific domain as provided above is recognized to drive expression of such gene. In some embodiments, the transcriptional activator drives expression of the target gene. In some cases, the transcriptional activator, can be or contain all or a portion of a heterologous transactivation domain. For example, in some embodiments, the transcriptional activator is selected from Herpes simplex-derived transactivation domain, Dnmt3a methyltransferase domain, p65, VP16, and VP64.
[0600] In some embodiments, the regulatory factor is a zinc finger transcription factor (ZF-TF). In some embodiments, the regulatory factor is VP64-p65-Rta (VPR).
[0601] In certain embodiments, the regulatory factor further comprises a transcriptional regulatory domain. Common domains include, e.g., transcription factor domains (activators, repressors, co-activators, co-repressors), silencers, oncogenes (e.g., myc, jun, fos, myb, max, mad, rel, ets, bcl, myb, mos family members etc.); DNA repair enzymes and their associated factors and modifiers; DNA rearrangement enzymes and their associated factors and modifiers; chromatin associated proteins and their modifiers (e.g. kinases, acetylases and deacetylases); and
DNA modifying enzymes (e.g., methyltransferases such as members of the DNMT family (e.g., DNMT1, DNMT3A, DNMT3B, DNMT3L, etc., topoisomerases, helicases, ligases, kinases, phosphatases, polymerases, endonucleases) and their associated factors and modifiers. See, e.g., U.S. Publication No. 2013/0253040, incorporated by reference in its entirety herein.
[0602] Suitable domains for achieving activation include the HSV VP 16 activation domain (see, e.g., Hagmann et al, J. Virol. 71, 5952-5962 (1 97)) nuclear hormone receptors (see, e.g., Torchia et al., Curr. Opin. Cell. Biol. 10:373-383 (1998)); the p65 subunit of nuclear factor kappa B (Bitko & Bank, J. Virol. 72:5610-5618 (1998) and Doyle & Hunt, Neuroreport 8:2937- 2942 (1997)); Liu et al., Cancer Gene Ther. 5:3-28 (1998)), or artificial chimeric functional domains such as VP64 (Beerli et al., (1998) Proc. Natl. Acad. Sci. USA 95:14623-33), and degron (Molinari et al., (1999) EMBO J. 18, 6439-6447). Additional exemplary activation domains include, Oct 1, Oct-2A, Spl, AP-2, and CTF1 (Seipel et al, EMBOJ. 11, 4961-4968 (1992) as well as p300, CBP, PCAF, SRC1 PvALF, AtHD2A and ERF-2. See, for example, Robyr et al, (2000) Mol. Endocrinol. 14:329-347; Collingwood et al, (1999) J. Mol. Endocrinol 23:255-275; Leo et al, (2000) Gene 245:1-11; Manteuffel-Cymborowska (1999) Acta Biochim. Pol. 46:77-89; McKenna et al, (1999) J. Steroid Biochem. Mol. Biol. 69:3-12; Malik et al, (2000) Trends Biochem. Sci. 25:277-283; and Lemon et al, (1999) Curr. Opin. Genet. Dev. 9:499-504. Additional exemplary activation domains include, but are not limited to, OsGAI, HALF-1, Cl, API, ARF-5, -6,-1, and -8, CPRF1, CPRF4, MYC-RP/GP, and TRAB1 , See, for example, Ogawa et al, (2000) Gene 245:21-29; Okanami et al, (1996) Genes Cells 1 :87-99; Goff et al, (1991) Genes Dev. 5:298-309; Cho et al, (1999) Plant Mol Biol 40:419-429; Ulmason et al, (1999) Proc. Natl. Acad. Sci. USA 96:5844-5849; Sprenger-Haussels et al, (2000) Plant J. 22:1- 8; Gong et al, (1999) Plant Mol. Biol. 41:33-44; and Hobo et al. , (1999) Proc. Natl. Acad. Sci. USA 96:15,348-15,353.
[0603] Exemplary repression domains that can be used to make genetic repressors include, but are not limited to, KRAB A/B, KOX, TGF-beta- inducible early gene (TIEG), v- erbA, SID, MBD2, MBD3, members of the DNMT family (e.g., DNMT1, DNMT3A, DNMT3B, DNMT3L, etc.), Rb, and MeCP2. See, for example, Bird et al, (1999) Cell 99:451-454; Tyler et
al, (1999) Cell 99:443-446; Knoepfler et al, (1999) Cell 99:447-450; and Robertson et al, (2000) Nature Genet. 25:338-342. Additional exemplary repression domains include, but are not limited to, R0M2 and AtHD2A. See, for example, Chem et al, (1996) Plant Cell 8:305-321; and Wu et al, (2000) Plant J. 22:19-27.
[0604] In some instances, the domain is involved in epigenetic regulation of a chromosome. In some embodiments, the domain is a histone acetyltransferase (HAT), e.g., type- A, nuclear localized such as MYST family members MOZ, Ybf2/Sas3, MOF, and Tip60, GNAT family members Gcn5 or pCAF, the p300 family members CBP, p300 or RttlO9 (Bemdsen and Denu (2008) Curr Opin Struct Biol 18(6):682-689). In other instances, the domain is a histone deacetylase (HD AC) such as the class I (HDAC-1, 2, 3, and 8), class II (HD AC IIA (HDAC-4, 5, 7 and 9), HD AC IIB (HDAC 6 and 10)), class IV (HDAC-1 1), class III (also known as sirtuins (SIRTs); SIRT1-7) (see Mottamal et al., (2015) Molecules 20(3):3898-3941). Another domain that is used in some embodiments is a histone phosphorylase or kinase, where examples include MSK1, MSK2, ATR, ATM, DNA-PK, Bubl, VprBP, IKK-a, PKCpi, Dik/Zip, JAK2, PKC5, WSTF and CK2. In some embodiments, a methylation domain is used and may be chosen from groups such as Ezh2, PRMT1/6, PRMT5/7, PRMT 2/6, CARMI, set7/9, MLL, ALL-1, Suv 39h, G9a, SETDB1, Ezh2, Set2, Doti, PRMT 1/6, PRMT 5/7, PR-Set7 and Suv4-20h, Domains involved in sumoylation and biotinylation (Lys9, 13, 4, 18 and 12) may also be used in some embodiments (review see Kousarides (2007) Cell 128:693-705).
[0605] Fusion molecules are constructed by methods of cloning and biochemical conjugation that are well known to those of skill in the art. Fusion molecules comprise a DNA- binding domain and a functional domain (e.g., a transcriptional activation or repression domain). Fusion molecules also optionally comprise nuclear localization signals (such as, for example, that from the SV40 medium T-antigen) and epitope tags (such as, for example, FLAG and hemagglutinin). Fusion proteins (and nucleic acids encoding them) are designed such that the translational reading frame is preserved among the components of the fusion.
[0606] Fusions between a polypeptide component of a functional domain (or a functional fragment thereof) on the one hand, and a non-protein DNA-binding domain (e.g., antibiotic,
intercalator, minor groove binder, nucleic acid) on the other, arc constructed by methods of biochemical conjugation known to those of skill in the art. See, for example, the Pierce Chemical Company (Rockford, IL) Catalogue. Methods and compositions for making fusions between a minor groove binder and a polypeptide have been described. Mapp et al, (2000) Proc. Natl. Acad. Sci. USA 97:3930-3935. Likewise, CRISPR/Cas TFs and nucleases comprising a sgRNA nucleic acid component in association with a polypeptide component function domain arc also known to those of skill in the art and detailed herein.
Exogenous Polypeptide
[0607] In some embodiments, increased expression (i.e., overexpression) of the polynucleotide is mediated by introducing into the cell an exogenous polynucleotide encoding the polynucleotide to be overexpressed. In some embodiments, the exogenous polynucleotide is a recombinant nucleic acid. Well-known recombinant techniques can be used to generate recombinant nucleic acids as outlined herein. In some embodiments, an exogenous polynucleotide encoding an exogenous polypeptide herein comprises a codon-optimized nucleic acid sequence.
[0608] In certain embodiments, the recombinant nucleic acids encoding an exogenous polypeptide, such as a tolerogenic factor or a chimeric antigen receptor, may be operably linked to one or more regulatory nucleotide sequences in an expression construct. Regulatory nucleotide sequences will generally be appropriate for the host cell and recipient subject to be treated. Numerous types of appropriate expression vectors and suitable regulatory sequences are known in the art for a variety of host cells. Typically, the one or more regulatory nucleotide sequences may include, but are not limited to, promoter sequences, leader or signal sequences, ribosomal binding sites, transcriptional start and termination sequences, translational start and termination sequences, and enhancer or activator sequences. Constitutive or inducible promoters as known in the art are also contemplated. The promoters may be either naturally occurring promoters, or hybrid promoters that combine elements of more than one promoter. An expression construct may be present in a cell on an episome, such as a plasmid, or the expression construct may be inserted in a chromosome. In a specific embodiment, the expression vector
includes a selectable marker gene to allow the selection of transformed host cells. Certain embodiments include an expression vector comprising a nucleotide sequence encoding a variant polypeptide operably linked to at least one regulatory sequence. Regulatory sequence for use herein include promoters, enhancers, and other expression control elements. In certain embodiments, an expression vector is designed for the choice of the host cell to be transformed, the particular variant polypeptide desired to be expressed, the vector's copy number, the ability to control that copy number, and/or the expression of any other protein encoded by the vector, such as antibiotic markers.
[0609] In some embodiments, the exogenous polynucleotide is operably linked to a promoter for expression of the exogenous polynucleotide in the modified cell. Examples of suitable mammalian promoters include, for example, promoters from the following genes: elongation factor 1 alpha (EFla) promoter, ubiquitin/S27a promoter of the hamster (WO 97/15664), Simian vacuolating virus 40 (SV40) early promoter, adenovirus major late promoter, mouse metallothionein-I promoter, the long terminal repeat region of Rous Sarcoma Virus (RSV), mouse mammary tumor virus promoter (MMTV), Moloney murine leukemia virus Long Terminal repeat region, and the early promoter of human Cytomegalovirus (CMV). Examples of other heterologous mammalian promoters are the actin, immunoglobulin or heat shock promoter(s). In additional embodiments, promoters for use in mammalian host cells can be obtained from the genomes of viruses such as polyoma virus, fowlpox virus (UK 2,211,504 published 5 Jul. 1989), bovine papilloma virus, avian sarcoma virus, cytomegalovirus, a retrovirus, hepatitis-B virus and Simian Virus 40 (SV40). In further embodiments, heterologous mammalian promoters are used. Examples include the actin promoter, an immunoglobulin promoter, and heat-shock promoters. The early and late promoters of SV40 are conveniently obtained as an SV40 restriction fragment which also contains the SV40 viral origin of replication (Fiers et al, Nature 273: 113-120 (1978)). The immediate early promoter of the human cytomegalovirus is conveniently obtained as a Hindlll restriction enzyme fragment (Greenaway et al, Gene 18: 355-360 (1982)). The foregoing references are incorporated by reference in their entirety.
[06101 In some embodiments, the expression vector is a bicistronic or multicistronic expression vector. Bicistronic or multicistronic expression vectors may include (1) multiple promoters fused to each of the open reading frames; (2) insertion of splicing signals between genes; (3) fusion of genes whose expressions are driven by a single promoter; and/or (4) insertion of proteolytic cleavage sites between genes (self-cleavage peptide) or insertion of internal ribosomal entry sites (IRESs) between genes.
[0611] In some embodiments, an expression vector or construct herein is a multicistronic construct. The terms “multicistronic construct” and “multicistronic vector” are used interchangeably herein and refer to a recombinant DNA construct that is to be transcribed into a single mRNA molecule, wherein the single mRNA molecule encodes two or more genes (e.g., two or more transgenes). The multi-cistronic construct is referred to as bicistronic construct if it encodes two genes, and tricistronic construct if it encodes three genes, and quadrocistronic construct if it encodes four genes, and so on.
[0612] In some embodiments, two or more exogenous polynucleotides comprised by a vector or construct (e.g., a transgene) are each separated by a multicistronic separation element. In some embodiments, the multicistronic separation element is an IRES or a sequence encoding a cleavable peptide or ribosomal skip element. In some embodiments, the multicistronic separation element is an IRES, such as an encephalomyocarditis (EMCV) virus IRES. In some embodiments, the multicistronic separation element is a cleavable peptide such as a 2A peptide. Exemplary 2A peptides include a P2A peptide, a T2A peptide, an E2A peptide, and an F2Apeptide. In some embodiments, the cleavable peptide is a T2A. In some embodiments, the two or more exogenous polynucleotides (e.g. the first exogenous polynucleotide and second exogenous polynucleotide) are operably linked to a promoter. In some embodiments, the first exogenous polynucleotide and the second exogenous polynucleotide are each operably linked to a promoter. In some embodiments, the promoter is the same promoter. In some embodiments, the promoter is an EFl promoter.
[0613] In some cases, an exogenous polynucleotide encoding an exogenous polypeptide (e.g., an exogenous polynucleotide encoding a tolerogenic factor or complement inhibitor
described herein) encodes a cleavable peptide or ribosomal skip element, such as T2A at the N- terminus or C-terminus of an exogenous polypeptide encoded by a multicistronic vector. In some embodiments, inclusion of the cleavable peptide or ribosomal skip element allows for expression of two or more polypeptides from a single translation initiation site. In some embodiments, the cleavable peptide is a T2A. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 11. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 12. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 17. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 18.
[0614] In some embodiments, the vector or construct includes a single promoter that drives the expression of one or more transcription units of an exogenous polynucleotide. In some embodiments, such vectors or constructs can be multicistronic (bicistronic or tricistronic, see e.g., U.S. Patent No. 6,060,273). For example, in some embodiments, transcription units can be engineered as a bicistronic unit containing an IRES (internal ribosome entry site), which allows coexpression of gene products (e.g., one or more tolerogenic factors such as CD47 and/or one or more complement inhibitor such as CD46, CD59, and CD55) from an RNA transcribed from a single promoter. In some embodiments, the vectors or constructs provided herein are bicistronic, allowing the vector or construct to express two separate polypeptides. In some cases, the two separate polypeptides encoded by the vector or construct are tolerogenic factors (e.g., two factors selected from CD47, DUX4, CD24, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA- G, PD-L1, IDOL CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, IL-39, FasL, CCL21, CCL22, Mfge8, and Serpinb9). In some embodiments, the one or more tolerogenic factors are selected from the group consisting of CD16, CD24, CD35, CD39, CD46, CD47, CD52, CD55, CD59, CD64, CD200, CCL22, CTLA4-Ig, Cl inhibitor, FASL, IDOL HLA-C, HLA-E, HLA-E heavy chain, HLA-G, IL-10, IL-35, PD-L1, SERPINB9, CCL21, MFGE8, DUX4, B2M-HLA-E, CD27, IL- 39, CD16 Fc Receptor, IL15-RF, H2-M3 (HLA-G), A20/TNFAIP3, CR1, HLA-F, and MANF. In some cases, the two separate polypeptides encoded by the vector or construct are CD46 and CD59. In some embodiments, the two separate polypeptides encoded by the vector or construct
are a tolerogenic factor (e.g., CD47) and a complement inhibitor selected from CD46, CD59, and CD55. In some embodiments, the vectors or constructs provided herein are tricistronic, allowing the vector or construct to express three separate polypeptides. In some cases, the three nucleic acid sequences of the tricistronic vector or construct are a tolerogenic factor such as CD47, CD46, and CD59. In some cases, the three nucleic acid sequences of the tricistronic vector or construct are CD46, CD59, and CD55. In some cases, the three nucleic acid sequences of the tricistronic vector or construct are three tolerogenic factors selected from CD47, DUX4, CD24, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl- Inhibitor, IL-10, IL-35, IL-39, EasL, CCL21, CCL22, Mfge8, and Serpinb9. In some embodiments, the vectors or constructs provided herein are quadrocistronic, allowing the vector or construct to express four separate polypeptides. In some cases, the four separate polypeptides of the quadrocistronic vector or construct are CD47, CD46, CD59, and CD55. In some cases, the four separate polypeptides of the quadrocistronic vector or construct are four tolerogenic factors selected from CD47, DUX4, CD24, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA- G, PD-L1, IDOL CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, IL-39, LasL, CCL21, CCL22, Mfge8, and Serpinb9.
[0615] In some embodiments, the cell comprises one or more vectors or constructs, wherein each vector or construct is a monocistronic or a multicistronic construct as described above, and the monocistronic or multicistronic constructs encode one or more tolerogenic factors and/or complement inhibitors, in any combination or order.
[0616] In some embodiments, a single promoter directs expression of an RNA that contains, in a single open reading frame (ORE), two, three, or four genes (e.g., encoding a tolerogenic factor (e.g., CD47) and/or one or more complement inhibitors selected from CD46, CD59, and CD55) separated from one another by sequences encoding a self-cleavage peptide (e.g., 2A sequences) or a protease recognition site (e.g., furin). The ORE thus encodes a single polypeptide, which, either during (in the case of 2A) or after translation, is processed into the individual proteins. In some cases, the peptide, such as T2A, can cause the ribosome to skip (ribosome skipping) synthesis of a peptide bond at the C-terminus of a 2A element, leading to
separation between the end of the 2A sequence and the next peptide downstream (see, for example, de Felipe. Genetic Vaccines and Ther. 2:13 (2004) and deFelipe et al. Traffic 5:616- 626 (2004)). Many 2A elements are known in the ail. Examples of 2A sequences that can be used in the methods and nucleic acids disclosed herein include, without limitation, 2A sequences from the foot-and-mouth disease virus (F2A, e.g., SEQ ID NO: 16), equine rhinitis A virus (E2A, e.g., SEQ ID NO: 15), thosea asigna vims (T2A, e.g., SEQ ID NO: 11, 12, 17, or 18), and porcine teschovirus- 1 (P2A, e.g., SEQ ID NO: 13 or 14) as described in U.S. Patent Publication No. 20070116690.
[0617] In cases where the vector or construct (e.g., transgene) contains more than one nucleic acid sequence encoding a protein, e.g., a first exogenous polynucleotide encoding CD46 and a second exogenous polynucleotide encoding CD59, or a first exogenous polynucleotide encoding CD47, a second exogenous polynucleotide encoding CD56, and a third exogenous polynucleotide encoding CD59, the vector or construct (e.g., transgene) may further include a nucleic acid sequence encoding a peptide between the first and second exogenous polynucleotide sequences. In some cases, the nucleic acid sequence positioned between the first and second exogenous polynucleotides encodes a peptide that separates the translation products of the first and second exogenous polynucleotides during or after translation. In some embodiments, the peptide contains a self-cleaving peptide or a peptide that causes ribosome skipping (a ribosomal skip element), such as a T2A peptide. In some embodiments, inclusion of the cleavable peptide or ribosomal skip element allows for expression of two or more polypeptides from a single translation initiation site. In some embodiments, the peptide is a self-cleaving peptide that is a T2A peptide. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 11. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 12. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 17. In some embodiments, the T2A is or comprises the amino acid sequence set forth by SEQ ID NO: 18.
[0618] The process of introducing the polynucleotides described herein into cells can be achieved by any suitable technique. Suitable techniques include calcium phosphate or lipid-
mediated transfection, electroporation, fusogens, and transduction or infection using a viral vector. In some embodiments, the polynucleotides are introduced into a cell via viral transduction (e.g., lentiviral transduction) or otherwise delivered on a viral vector (e.g., fusogen- mediated delivery). Suitable techniques include calcium phosphate or lipid-mediated transfection, electroporation, transposase-mediated delivery, and transduction or infection using a viral vector. In some embodiments, the polynucleotides are introduced into a cell via viral transduction (e.g., lentiviral transduction) or otherwise delivered on a viral vector (e.g., fusogen- mediated delivery). In some embodiments, vectors that package a polynucleotide encoding an exogenous polynucleotide may be used to deliver the packaged polynucleotides to a cell or population of cells. These vectors may be of any kind, including DNA vectors, RNA vectors, plasmids, viral vectors and particles. In some embodiments, lipid nanoparticles can be used to deliver an exogenous polynucleotide to a cell. In some embodiments, viral vectors can be used to deliver an exogenous polynucleotide to a cell. Viral vector technology is well known and described in Sambrook et al. (2001, Molecular Cloning: A Laboratory Manual, Cold Spring Harbor Laboratory, New York). Viruses, which are useful as vectors include, but are not limited to lentiviral vectors, adenoviral vectors, adeno-associated viral (AAV) vectors, herpes simplex viral vectors, retroviral vectors, oncolytic viruses, and the like. In some embodiments, the introduction of the exogenous polynucleotide into the cell can be specific (targeted) or nonspecific (e.g., non-targeted). In some embodiments, the introduction of the exogenous polynucleotide into the cell can result in integration or insertion into the genome in the cell. In other embodiments, the introduced exogenous polynucleotide may be non-integrating or episomal in the cell. A skilled artisan is familial' with methods of introducing nucleic acid transgenes into a cell, including any of the exemplary methods described herein, and can choose a suitable method.
Non-Targeted Delivery
[0619] In some embodiments, an exogenous polynucleotide is introduced into a cell (e.g., source cell) by any of a variety of non-targeted methods. In some embodiments, the exogenous polynucleotide is inserted into a random genomic locus of a host cell. As known to a person
skilled in the ail, viral vectors, including, for example, retroviral vectors and lentiviral vectors are commonly used to deliver genetic material into host cells and randomly insert the foreign or exogenous gene into the host cell genome to facilitate stable expression and replication of the gene. In some embodiments, the non-targeted introduction of the exogenous polynucleotide into the cell is under conditions for stable expression of the exogenous polynucleotide in the cell. In some embodiments, methods for introducing a nucleic acid for stable expression in a cell involves any method that results in stable integration of the nucleic acid into the genome of the cell, such that it may be propagated if the cell it has integrated into divides.
[0620] In some embodiments, the viral vector is a lentiviral vector. Lentiviral vectors are particularly useful means for successful viral transduction as they permit stable expression of the gene contained within the delivered nucleic acid transcript. Lentiviral vectors express reverse transcriptase and integrase, two enzymes required for stable expression of the gene contained within the delivered nucleic acid transcript. Reverse transcriptase converts an RNA transcript into DNA, while integrase inserts and integrates the DNA into the genome of the target cell. Once the DNA has been integrated stably into the genome, it divides along with the host. The gene of interest contained within the integrated DNA may be expressed constitutively or it may be inducible. As part of the host cell genome, it may be subject to cellular regulation, including activation or repression, depending on a host of factors in the target cell.
[0621] Lentiviruses are subgroup of the Retroviridae family of viruses, named because reverse transcription of viral RNA genomes to DNA is required before integration into the host genome. As such, the most important features of lentiviral vehicles/particles are the integration of their genetic material into the genome of a target/host cell. Some examples of lentivirus include the Human Immunodeficiency Viruses: HIV-1 and HIV -2, the Simian Immunodeficiency Vims (SIV), feline immunodeficiency vims (FIV), bovine immunodeficiency vims (BIV), Jembrana Disease Virus (JDV), equine infectious anemia virus (EIAV), equine infectious anemia, virus, visna-maedi and caprine arthritis encephalitis vims (CAEV).
[0622] Typically, lentiviral particles making up the gene delivery vehicle are replication defective on their own (also referred to as "self-inactivating"). Lentivimses are able to infect both
dividing and non-dividing cells by virtue of the entry mechanism through the intact host nuclear envelope (Naldini L et al., Curr. Opin. Biotechnol, 1998, 9: 457-463). Recombinant lentiviral vehicles/particles have been generated by multiply attenuating the HIV virulence genes, for example, the genes Env, Vif, Vpr, Vpu, Nef and Tat are deleted making the vector biologically safe. Correspondingly, lentiviral vehicles, for example, derived from HIV- 1 /HIV-2 can mediate the efficient delivery, integration and long-term expression of transgenes into non- dividing cells.
[0623] Lentiviral particles may be generated by co-expressing the virus packaging elements and the vector genome itself in a producer cell such as human HEK293T cells. These elements are usually provided in three (in second generation lentiviral systems) or four separate plasmids (in third generation lentiviral systems). The producer cells arc co-transfected with plasmids that encode lentiviral components including the core (i.e., structural proteins) and enzymatic components of the virus, and the envelope protein(s) (referred to as the packaging systems), and a plasmid that encodes the genome including a foreign transgene, to be transferred to the target cell, the vehicle itself (also referred to as the transfer vector). In general, the plasmids or vectors are included in a producer cell line. The plasmids/vectors are introduced via transfection, transduction or infection into the producer cell line. Methods for transfection, transduction or infection are well known by those of skill in the art. As non-limiting example, the packaging and transfer constructs can be introduced into producer cell lines by calcium phosphate transfection, lipofection or electroporation, generally together with a dominant selectable marker, such as neomyocin (neo), dihydrofolate reductase (DHFR), glutamine synthetase or adenosine deaminase (ADA), followed by selection in the presence of the appropriate drug and isolation of clones.
[0624] The producer cell produces recombinant viral particles that contain the foreign gene, for example, the polynucleotides encoding the exogenous polynucleotide. The recombinant viral particles are recovered from the culture media and titrated by standard methods used by those of skill in the art. The recombinant lentiviral vehicles can be used to infect target cells, such source cells including any described herein.
[06251 Cells that can be used to produce high-titer lentiviral particles may include, but are not limited to, HEK293T cells, 293G cells, STAR cells (Relander et al., Mol Ther. 2005, 11: 452- 459), FreeStyle™ 293 Expression System (ThermoFisher, Waltham, MA), and other HEK293T- based producer cell lines (e.g., Stewart et al., Hum Gene Ther. _2011, 2,2.(3):357~369; Lee et al, Biotechnol Bioeng, 2012, 10996): 1551-1560; Throm et al.. Blood. 2009, 113(21): 5104-5110).
[0626] Additional elements provided in lentiviral particles may comprise retroviral LTR (long- terminal repeat) at either 5' or 3' terminus, a retroviral export element, optionally a lentiviral reverse response element (RRE), a promoter or active portion thereof, and a locus control region (LCR) or active portion thereof. Other elements include central polypurine tract (cPPT) sequence to improve transduction efficiency in non-dividing cells, Woodchuck Hepatitis Virus (WHP) Posttranscriptional Regulatory Element (WPRE) which enhances the expression of the transgene, and increases titer.
[0627] Methods for generating recombinant lentiviral particles are known to a skilled artisan, for example, U.S. Pat. NOs.: 8,846,385; 7,745,179; 7,629,153; 7,575,924; 7,179,903; and 6,808,905. Lentivirus vectors used may be selected from, but are not limited to pLVX, pLenti, pLenti6, pLJMl, FUGW, pWPXL, pWPI, pLenti CMV puro DEST, pLJMl-EGFP, pULTRA, p!nducer2Q, pHIV-EGFP, pCW57.1 , pTRPE, pELPS, pRRL, and pLionll, Any known lentiviral vehicles may also be used (See, U.S. Pat. NOs. 9,260,725: 9,068,199: 9,023,646: 8,900,858: 8,748,169; 8,709,799; 8,420,104; 8,329,462; 8,076,106; 6,013,516: and 5,994, 136; International Patent Publication NO.: WO2012079000).
[0628] In some embodiments, the exogenous polynucleotide is introduced into the cell under conditions for transient expression of the cell, such as by methods that result in episomal delivery of an exogenous polynucleotide.
[0629] In some embodiments, polynucleotides encoding the exogenous polynucleotide may be packaged into recombinant adeno-associated viral (rAAV) vectors. Such vectors or viral particles may be designed to utilize any of the known serotype capsids or combinations of
serotype capsids. The serotype capsids may include capsids from any identified AAV serotypes and variants thereof, for example, AAV1, AAV2, AAV2G9, AAV3, AAV4, AAV4-4, AAV5, AAV6, AAV7, AAV8, AAV9, AAV 10, AAV 11, AAV 12 and AAVrhlO. In some embodiments, the AAV serotype may be or have a sequence as described in United States Publication No. US20030138772; Pulicherla et al. Molecular Therapy, 2011, 19(6): 1070-1078; U.S. Pat. Nos. : 6,156,303; 7,198,951; U.S. Patent Publication Nos. : US2015/0159173 and US2014/0359799: and International Patent Publication NOs.: WO1998/011244, W02005/033321 and WO2014/14422.
[0630] AAV vectors include not only single stranded vectors but self-complementary AAV vectors (scAAVs). scAAV vectors contain DNA which anneals together to form double stranded vector genome. By skipping second strand synthesis, scAAVs allow for rapid expression in the cell. The rAAV vectors may be manufactured by standard methods in the art such as by triple transfection, in sf’9 insect cells or in suspension cell cultures of human cells such as HEK293 cells.
[0631] In some embodiments, non-viral based methods may be used. For instance, in some aspects, vectors comprising the polynucleotides may be transferred to cells by non-viral methods by physical methods such as needles, electroporation, sonoporation, hyrdoporation; chemical carriers such as inorganic particles (e.g., calcium phosphate, silica, gold) and/or chemical methods. In other aspects, synthetic or natural biodegradable agents may be used for delivery such as cationic lipids, lipid nano emulsions, nanoparticles, peptide -based vectors, or polymer-based vectors.
Targeted Delivery
[0632] In some embodiments, the exogenous polynucleotide can be inserted into a specific genomic locus of the host cell. A number of gene editing methods can be used to insert an exogenous polynucleotide (e.g., a transgene) into a specific genomic locus of choice. The exogenous polynucleotide can be inserted into any suitable target genomic loci of the cell. In some embodiments, the exogenous polynucleotide is introduced into the cell by targeted
integration into a target loci. In some embodiments, targeted integration can be achieved by gene editing using one or more nucleases and/or nickases and a donor template in a process involving homology-dependent or homology-independent recombination.
[0633] Gene editing is a type of genetic engineering in which a nucleotide sequence may be inserted, deleted, modified, or replaced in the genome of a living organism. A number of gene editing methods can be used to insert an exogenous polynucleotide into the specific genomic locus of choice, including for example homology-directed repair (HOR), homology-mediated end-joining (HMEJ), homology-independent targeted integration (HITI), obligate ligation-gated recombination (ObliGaRe), or precise integration into target chromosome (PITCh). In some embodiments, the gene editing technology can include systems involving nucleases, integrases, transposases, and/or recombinases. In some embodiments, the gene editing technology mediates single-strand breaks (SSB). In some embodiments, the gene editing technology mediates doublestrand breaks (DSB), including in connection with non-homologous end-joining (NHEJ) or homology-directed repair (HDR). In some embodiments, the gene editing technology can include DNA-based editing or prime-editing. In some embodiments, the gene editing technology can include Programmable Addition via Site- specific Targeting Elements (PASTE). In some embodiments, the gene editing technology can include TnpB polypeptides. Many gene editing techniques generally utilize the innate mechanism for cells to repair double-strand breaks (DSBs) in DNA.
[0634] Eukaryotic cells repair DSBs by two primary repair pathways: non-homologous end-joining (NHEJ) and homology-directed repair (HDR). HDR typically occurs during late S phase or G2 phase, when a sister chromatid is available to serve as a repair template. NHEJ is more common and can occur during any phase of the cell cycle, but it is more error prone. In gene editing, NHEJ is generally used to produce insertion/deletion mutations (indels), which can produce targeted loss of function in a target gene by shifting the open reading frame (ORF) and producing alterations in the coding region or an associated regulatory region. HDR, on the other hand, is a preferred pathway for producing targeted knock-ins, knockouts, or insertions of specific mutations in the presence of a repair template with homologous sequences. Several
methods are known to a skilled artisan to improve HDR efficiency, including, for example, chemical modulation (e.g., treating cells with inhibitors of key enzymes in the NHEJ pathway); timed delivery of the gene editing system at S and G2 phases of the cell cycle; cell cycle arrest at S and G2 phases; and introduction of repair templates with homology sequences. The methods provided herein may utilize HDR-mediated repair, NHEJ-mediated repair, or a combination thereof.
[0635] In some embodiments, the methods provided herein for HDR-mediated insertion utilize a site-directed nuclease, including, for example, zinc finger nucleases (ZFNs), transcription activator-like effector nucleases (TALENs), meganucleases, transposases, and clustered regularly interspaced short palindromic repeat (CRISPR)/Cas systems. In some embodiments, the nucleases create specific double-strand breaks (DSBs) at desired locations (e.g., target sites) in the genome, and harness the cell's endogenous mechanisms to repair the induced break. The nickases create specific single- strand breaks at desired locations in the genome. In one non-limiting example, two nickases can be used to create two single-strand breaks on opposite strands of a target DNA, thereby generating a blunt or a sticky end. Any suitable nuclease can be introduced into a cell to induce genome editing of a target DNA sequence including, but not limited to, CRIS PR-associated protein (Cas) nucleases, zinc finger nucleases (ZFNs), transcription activator-like effector nucleases (TALENs), meganucleases, other endo- or exo-nucleases, variants thereof, fragments thereof, and combinations thereof. In some embodiments, when a nuclease or a nickase is introduced with a donor template containing an exogenous polynucleotide sequence (also called a transgene) flanked by homology sequences (e.g., homology arms) that are homologous to sequences at or near the endogenous genomic target locus, e.g., a safe harbor locus, DNA damage repair pathways can result in integration of the transgene sequence at the target site in the cell. This can occur by a homology -dependent process. In some embodiments, the donor template is a circular double- stranded plasmid DNA, single- stranded donor oligonucleotide (ssODN), linear double- stranded polymerase chain reaction (PCR) fragments, or the homologous sequences of the intact sister chromatid.
Depending on the form of the donor template, the homology -mediated gene insertion and
replacement can be carried out via specific DNA repair pathways such as homology-directed repair (HDR), synthesis-dependent strand annealing (SDSA), microhomology-mediated end joining (MMEJ), and homology-mediated end joining (HMEJ) pathways.
[0636] For instance, DNA repair mechanisms can be induced by a nuclease after (i) two SSBs, where there is a SSB on each strand, thereby inducing single strand overhangs; or (ii) a DSB occurring at the same cleavage site on both strands, thereby inducing a blunt end break. Upon cleavage by one of these agents, the target locus with the SSBs or the DSB undergoes one of two major pathways for DNA damage repair: (1) the error-prone non-homologous end joining (NHEJ), or (2) the high-fidelity homology-directed repair (HDR) pathway. In some embodiments, a donor template (e.g., circular plasmid DNA or a linear DNA fragment, such as a ssODN) introduced into cells in which there are SSBs or a DSB can result in HDR and integration of the donor template into the target locus. In general, in the absence of a donor template, the NHEJ process re-ligates the ends of the cleaved DNA strands, which frequently results in nucleotide deletions and insertions at the cleavage site.
[0637] In some embodiments, site-directed insertion of the exogenous polynucleotide into a cell may be achieved through HDR-based approaches. HDR is a mechanism for cells to repair double-strand breaks (DSBs) in DNA and can be utilized to modify genomes in many organisms using various gene editing systems, including clustered regularly interspaced short palindromic repeat (CRISPR)/Cas systems, zinc finger nucleases (ZFNs), transcription activatorlike effector nucleases (TALENs), meganucleases, and transposases.
[0638] In some embodiments, the targeted integration is carried by introducing one or more sequence- specific or targeted nucleases, including DNA-binding targeted nucleases and gene editing nucleases such as zinc finger nucleases (ZFN) and transcription activator-like effector nucleases (TALENs), and RNA-guided nucleases such as a CRISPR-associated nuclease (Cas) system, specifically designed to be targeted to at least one target site(s) sequence of a target gene. Exemplary ZFNs, TALEs, and TALENs are described in, e.g., Lloyd et al., Frontiers in Immunology, 4(221): 1-7 (2013). In particular embodiments, targeted genetic disruption at or near the target site is carried out using clustered regularly interspaced short
palindromic repeats (CRISPR) and CRISPR-associated (Cas) proteins. See Sander and Joung, (2014) Nature Biotechnology, 32(4): 347-355.
Zinc Finger Nucleases (ZFNs)
[0639] ZFNs are fusion proteins comprising an array of site-specific DNA binding domains adapted from zinc finger-containing transcription factors attached to the endonuclease domain of the bacterial FokI restriction enzyme. A ZFN may have one or more (e.g., 1 , 2, 3, 4, 5, 6, 7, 8, 9, 10, or more) of the DNA binding domains or zinc finger domains. See, e.g., Carroll et al., Genetics Society of America (2011) 188:773-782; Kim et al., Proc. Natl. Acad. Sci. USA (1996) 93:1156-1160. Each zinc finger domain is a small protein structural motif stabilized by one or more zinc ions and usually recognizes a 3- to 4-bp DNA sequence. Tandem domains can thus potentially bind to an extended nucleotide sequence that is unique within a cell’s genome.
[0640] Various zinc fingers of known specificity can be combined to produce multifinger polypeptides which recognize about 6, 9, 12, 15, or 18-bp sequences. Various selection and modular assembly techniques are available to generate zinc fingers (and combinations thereof) recognizing specific sequences, including phage display, yeast one-hybrid systems, bacterial one-hybrid and two-hybrid systems, and mammalian cells. Zinc fingers can be engineered to bind a predetermined nucleic acid sequence. Criteria to engineer a zinc finger to bind to a predetermined nucleic acid sequence are known in the art. See, e.g., Sera et al., Biochemistry (2002) 41:7074-7081; Liu et al., Bioinformatics (2008) 24:1850-1857.
[0641] ZFNs containing FokI nuclease domains or other dimeric nuclease domains function as a dimer. Thus, a pair of ZFNs are required to target non-palindromic DNA sites. The two individual ZFNs must bind opposite strands of the DNA with their nucleases properly spaced apart. See Bitinaite et al., Proc. Natl. Acad. Sci. USA (1998) 95:10570-10575. To cleave a specific site in the genome, a pair of ZFNs are designed to recognize two sequences flanking the site, one on the forward strand and the other on the reverse strand. Upon binding of the ZFNs on either side of the site, the nuclease domains dimerize and cleave the DNA at the site, generating a DSB with 5' overhangs. HDR can then be utilized to introduce a specific mutation,
with the help of a repair template containing the desired mutation flanked by homology arms. The repair template is usually an exogenous double-stranded DNA vector introduced to the cell. See Miller et al., Nat. Biotechnol. (2011) 29:143-148; Hockemeyer et al., Nat. Biotechnol. (2011) 29:731-734.
TALENs
[0642] TALENs are another example of an artificial nuclease which can be used to edit a target gene. TALENs are derived from DNA binding domains termed TALE repeats, which usually comprise tandem arrays with 10 to 30 repeats that bind and recognize extended DNA sequences. Each repeat is 33 to 35 amino acids in length, with two adjacent amino acids (termed the repeat-variable di-residue, or RVD) conferring specificity for one of the four DNA base pairs. Thus, there is a one-to-one correspondence between the repeats and the base pairs in the target DNA sequences.
[0643] TALENs are produced artificially by fusing one or more TALE DNA binding domains (e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, 10 or more) to a nuclease domain, for example, a FokI endonuclease domain. See Zhang, Nature Biotech. (2011 ) 29:149-153. Several mutations to FokI have been made for its use in TALENs; these, for example, improve cleavage specificity or activity. See Cermak et al., Nucl. Acids Res. (2011) 39:e82; Miller et al., Nature Biotech. (2011) 29:143-148; Hockemeyer et al., Nature Biotech. (2011) 29:731-734; Wood et al., Science (2011) 333:307; Doyon et al., Nature Methods (2010) 8:74-79; Szczepek et al., Nature Biotech (2007) 25:786-793; Guo et al., J. Mol. Biol. (2010) 200:96. The FokI domain functions as a dimer, requiring two constructs with unique DNA binding domains for sites in the target genome with proper orientation and spacing. Both the number of amino acid residues between the TALE DNA binding domain and the FokI nuclease domain and the number of bases between the two individual TALEN binding sites appear to be important parameters for achieving high levels of activity. Miller et al., Nature Biotech. (2011) 29:143-148.
[0644] By combining engineered TALE repeats with a nuclease domain, a site-specific nuclease can be produced specific to any desired DNA sequence. Similar to ZFNs, TALENs can
be introduced into a cell to generate DSBs at a desired target site in the genome, and so can be used to knock out genes or knock in mutations in similar, HDR-mediated pathways. See Boch, Nature Biotech. (2011) 29:135-136; Boch et al., Science (2009) 326:1509-1512; Moscou et al., Science (2009) 326:3501.
Meganucleases
[0645] Meganucleases are enzymes in the endonuclease family which are characterized by their capacity to recognize and cut large DNA sequences (from 14 to 40 base pairs). Meganucleases are grouped into families based on their structural motifs which affect nuclease activity and/or DNA recognition. The most widespread and best known meganucleases are the proteins in the LAGLID ADG family, which owe their name to a conserved amino acid sequence. See Chevalier et al., Nucleic Acids Res. (2001) 29(18): 3757-3774. On the other hand, the GIY- YIG family members have a GIY-YIG module, which is 70-100 residues long and includes four or five conserved sequence motifs with four invariant residues, two of which are required for activity. See Van Roey et al., Nature Struct. Biol. (2002) 9:806-811. The His-Cys family meganucleases are characterized by a highly conserved series of histidines and cysteines over a region encompassing several hundred amino acid residues. See Chevalier et al., Nucleic Acids Res. (2001) 29(18):3757-3774. Members of the NHN family are defined by motifs containing two pairs of conserved histidines surrounded by asparagine residues. See Chevalier et al., Nucleic Acids Res. (2001) 29(18):3757-3774.
[0646] Because the chance of identifying a natural meganuclease for a particular target DNA sequence is low due to the high specificity requirement, various methods including mutagenesis and high throughput screening methods have been used to create meganuclease variants that recognize unique sequences. Strategies for engineering a meganuclease with altered DNA-binding specificity, e.g., to bind to a predetermined nucleic acid sequence are known in the art. See, e.g., Chevalier et al., Mol. Cell. (2002) 10:895-905; Epinat et al., Nucleic Acids Res (2003) 31:2952-2962; Silva et al., J Mol. Biol. (2006) 361:744-754; Seligman et al., Nucleic Acids Res (2002) 30:3870-3879; Sussman et al., J Mol Biol (2004) 342:31-41; Doyon et al., J Am Chem Soc (2006) 128:2477-2484; Chen et al., Protein Eng Des Sei (2009) 22:249-256;
Amould et al., J Mol Biol. (2006) 355:443-458; Smith et al., Nucleic Acids Res. (2006) 363(2):283-294.
[0647] Like ZFNs and TALENs, Meganucleases can create DSBs in the genomic DNA, which can create a frame-shift mutation if improperly repaired, e.g., via NHEJ, leading to a decrease in the expression of a target gene in a cell. Alternatively, foreign DNA can be introduced into the cell along with the meganuclease. Depending on the sequences of the foreign DNA and chromosomal sequence, this process can be used to modify the target gene. See Silva et al., Current Gene Therapy (2011) 11:11-27.
Transposases
[0648] Transposases are enzymes that bind to the end of a transposon and catalyze its movement to another pail of the genome by a cut and paste mechanism or a replicative transposition mechanism. By linking transposases to other systems such as the CRISPR/Cas system, new gene editing tools can be developed to enable site specific insertions or manipulations of the genomic DNA. There are two known DNA integration methods using transposons which use a catalytically inactive Cas effector protein and Tn7-like transposons. The transposase-dependent DNA integration does not provoke DSBs in the genome, which may guarantee safer and more specific DNA integration.
CRISPR/Cas
[0649] The CRISPR system was originally discovered in prokaryotic organisms (e.g., bacteria and archaea) as a system involved in defense against invading phages and plasmids that provides a form of acquired immunity. Now it has been adapted and used as a popular gene editing tool in research and clinical applications.
[0650] CRISPR/Cas systems generally comprise at least two components: one or more guide RNAs (gRNAs) and a Cas protein. The Cas protein is a nuclease that introduces a DSB into the target site. CRISPR-Cas systems fall into two major classes: class 1 systems use a complex of multiple Cas proteins to degrade nucleic acids; class 2 systems use a single large Cas protein for the same purpose. Class 1 is divided into types I, III, and IV; class 2 is divided into
types II, V, and VI. Different Cas proteins adapted for gene editing applications include, but are not limited to, Cas3, Cas4, Cas5, Cas8a, Cas8b, Cas8c, Cas9, CaslO, Casl2, Casl2a (Cpfl), Casl2b (C2cl), Casl2c (C2c3), Casl2d (CasY), Casl2e (CasX), Casl2f (C2cl0), Casl2g, Casl2h, Casl2i, Casl2k (C2c5), Casl3, Casl3a (C2c2), Casl3b, Casl3c, Casl3d, C2c4, C2c8, C2c9, Cmr5, Csel, Cse2, Csfl, Csm2, Csn2, CsxlO, Csxl l, Csyl, Csy2, Csy3, and Mad7. See, e.g., Jinek et al., Science (2012) 337 (6096):816-821; Dang et al., Genome Biology (2015) 16:280; Ran et al., Nature (2015) 520:186-191 ; Zetsche et al., Cell (2015) 163:759-771 ; Strecker et al., Nature Comm. (2019) 10:212; Yan et al., Science (2019) 363:88-91. The most widely used Cas9 is a type II Cas protein and is described herein as illustrative. These Cas proteins may be originated from different source species. For example, Cas9 can be derived from S. pyogenes or S. aureus.
[0651] In the original microbial genome, the type II CRISPR system incorporates sequences from invading DNA between CRISPR repeat sequences encoded as arrays within the host genome. Transcripts from the CRISPR repeat arrays are processed into CRISPR RNAs (crRNAs) each harboring a variable sequence transcribed from the invading DNA, known as the “protospacer” sequence, as well as part of the CRISPR repeat. Each crRNA hybridizes with a second transactivating CRISPR RNA (tracrRNA), and these two RNAs form a complex with the Cas9 nuclease. The protospacer-encoded portion of the crRNA directs the Cas9 complex to cleave complementary target DNA sequences, provided that they are adjacent to short sequences known as “protospacer adjacent motifs” (PAMs).
[0652] While the foregoing description has focused on Cas9 nuclease, it should be appreciated that other RNA-guided nucleases exist which utilize gRNAs that differ in some ways from those described to this point. For instance, Cpfl (CRISPR from Prevotella and Franciscella 1; also known as Cas 12a) is an RNA-guided nuclease that only requires a crRNA and does not need a tracrRNA to function.
[0653] Since its discovery, the CRISPR system has been adapted for inducing sequence specific DSBs and targeted genome editing in a wide range of cells and organisms spanning from bacteria to eukaryotic cells including human cells. In its use in gene editing applications,
artificially designed, synthetic gRNAs have replaced the original crRNA:tracrRNA complexes, including in certain embodiments via a single gRNA. For example, the gRNAs can be single guide RNAs (sgRNAs) composed of a crRNA, a tetraloop, and a tracrRNA. The crRNA usually comprises a complementary region (also called a spacer, usually about 20 nucleotides in length) that is user-designed to recognize a target DNA of interest. The tracrRNA sequence comprises a scaffold region for Cas nuclease binding. The crRNA sequence and the tracrRNA sequence are linked by the tetraloop and each have a short repeat sequence for hybridization with each other, thus generating a chimeric sgRNA. One can change the genomic target of the Cas nuclease by simply changing the spacer or complementary region sequence present in the gRNA. The complementary region will direct the Cas nuclease to the target DNA site through standard RNA-DNA complementary base pairing rules.
[0654] In order for the Cas nuclease to function, there must be a PAM immediately downstream of the target sequence in the genomic DNA. Recognition of the PAM by the Cas protein is thought to destabilize the adjacent genomic sequence, allowing interrogation of the sequence by the gRNA and resulting in gRNA-DNA pairing when a matching sequence is present. The specific sequence of PAM varies depending on the species of the Cas gene. For example, the most commonly used Cas9 nuclease derived from S. pyogenes recognizes a PAM sequence of 5’-NGG-3’ or, at less efficient rates, 5’-NAG-3’, where “N” can be any nucleotide. Other Cas nuclease variants with alternative PAMs have also been characterized and successfully used for genome editing.
[0655] In some embodiments, Cas nucleases may comprise one or more mutations to alter their activity, specificity, recognition, and/or other characteristics. For example, the Cas nuclease may have one or more mutations that alter its fidelity to mitigate off-target effects (e.g., eSpCas9, SpCas9-HFl, HypaSpCas9, HeFSpCas9, and evoSpCas9 high-fidelity variants of SpCas9). For another example, the Cas nuclease may have one or more mutations that alter its PAM specificity.
[0656] In some embodiments, CRISPR systems of the present disclosure comprise TnpB polypeptides. In some embodiments, TnpB polypeptides may comprise a Ruv-C-like domain.
The RuvC domain may be a split RuvC domain comprising RuvC-I, RuvC-II, and RuvC-III subdomains. In some embodiments, a TnpB may further comprise one or more of a HTH domain, a bridge helix domain and a zinc finger domain. TnpB polypeptides do not comprise an HNH domain. In some embodiments, a TnpB protein comprises, starting at the N-terminus: a HTH domain, a RuvC-I subdomain, a bridge helix domain, a RuvC-II sub-domain, a zinger finger domain, and a RuvC-III sub-domain. In some embodiments, a RuvC-III sub-domain forms the C-terminus of a TnpB polypeptide. In some embodiments, a TnpB polypeptide is from Epsilonproteobacteria bacterium, Actinoplanes lobatus strain DSM 43150, Actinomadura celluolosilytica strain DSM 45823, Actinomadura namibiensis strain DSM 44197, Alicyclobacillus macrosprangiidus strain DSM 17980, Lipingzhangella halophila strain DSM 102030, or Ktedonobacter recemifer. In some embodiments, a TnpB polypeptide is from Ktedonobacter racemifer, or comprises a conserved RNA region with similarity to the 5’ ITR of K. racemifer TnpB loci. In some embodiments, a TnpB may comprise a Fanzor protein, a TnpB homolog found in eukaryotic genomes. In some embodiments, a CRISPR system comprising a TnpB polypeptide binds a target adjacent motif (TAM) sequence 5’ of a target polynucleotide. In some embodiments, a TAM is a transposon-associated motif. In some embodiments, a TAM sequence comprises TCA. In some embodiments, a TAM sequence comprises TTCAN. In some embodiments, a TAM sequence comprises TTGAT. In some embodiments, a TAM sequence comprises ATAAA.
[0657] In certain embodiments, the exogenous polynucleotide may function as a DNA repair template to be integrated into the target site through HDR in associated with a gene editing system (e.g., the CRISPR/Cas system) as described. Generally, the exogenous polynucleotide to be inserted would comprise at least the expression cassette encoding the protein of interest (e.g., the tolerogenic factor) and would optionally also include one or more regulatory elements (e.g., promoters, insulators, enhancers). In certain of these embodiments, the exogenous polynucleotide to be inserted would be flanked by homologous sequence immediately upstream and downstream of the target, i.e., left homology arm (LHA) and right homology arm (RHA), specifically designed for the target genomic locus to serve as template for HDR. The length of
each homology arm is generally dependent on the size of the insert being introduced, with larger insertions requiring longer homology arms.
[0658] In some embodiments, target-primed reverse transcription (TPRT) or prime editing may be used to engineer exogenous genes, such as exogenous transgenes encoding a tolerogenic factor (e.g., CD47) into specific loci. In some embodiments, prime editing mediates targeted insertions, deletions, all 12 possible base-to-base conversions, and combinations thereof in human cells without requiring DSBs or donor DNA templates.
[0659] Prime editing is a genome editing method that directly writes new genetic information into a specified DNA site using a nucleic acid programmable DNA binding protein (“napDNAbp”) working in association with a polymerase (i.e., in the form of a fusion protein or otherwise provided in trans with the napDNAbp), wherein the prime editing system is programmed with a prime editing (PE) guide RNA (“PEgRNA”) that both specifies the target site and templates the synthesis of the desired edit in the form of a replacement DNA strand by way of an extension (either DNA or RNA) engineered onto a guide RNA (e.g., at the 5' or 3' end, or at an internal portion of a guide RNA). The replacement strand containing the desired edit (e.g., a single nucleobase substitution) shares the same sequence as the endogenous strand of the target site to be edited (with the exception that it includes the desired edit). Through DNA repair and/or replication machinery, the endogenous strand of the target site is replaced by the newly synthesized replacement strand containing the desired edit. In some cases, prime editing may be thought of as a “search-and- replace” genome editing technology since the prime editors search and locate the desired target site to be edited, and encode a replacement strand containing a desired edit which is installed in place of the corresponding target site endogenous DNA strand at the same time. For example, prime editing can be adapted for conducting precision CRISPR/Cas-based genome editing in order to bypass double stranded breaks. In some embodiments, a homologous protein is or encodes for a Cas protein-reverse transcriptase fusions or related systems to target a specific DNA sequence with a guide RNA, generate a single strand nick at the target site, and use the nicked DNA as a primer for reverse transcription of an engineered reverse transcriptase template that is integrated with the guide RNA. In some
embodiments, a prime editor protein is paired with two prime editing guide RNAs (pegRNAs) that template the synthesis of complementary DNA flaps on opposing strands of genomic DNA, resulting in the replacement of endogenous DNA sequence between the PE-induced nick sites with pegRNA-encoded sequences.
[0660] In some embodiments, a gene editing technology is associated with a prime editor that is a reverse transcriptase, or any DNA polymerase known in the art. Thus, in one aspect, a prime editor may comprise Cas9 (or an equivalent napDNAbp) which is programmed to target a DNA sequence by associating it with a specialized guide RNA (i.e., PEgRNA) containing a spacer sequence that anneals to a complementary protospacer in the target DNA. Such methods include any disclosed in Anzalone et al., (doi.org/10.1038/s41586-019-1711-4), or in PCT publication Nos. WO2020191248, WO2021226558, or W02022067130, which are hereby incorporated in their entirety.
[0661] In some embodiments, the base editing technology may be used to introduce single-nucleotide variants (SNVs) into DNA or RNA in living cells. Base editing is a CRISPR- Cas9-based genome editing technology that allows the introduction of point mutations in RNAs or DNAs without generating DSBs. Base editors (BEs) are typically fusions of a Cas (“CRISPR- associated”) domain and a nucleobase modification domain (e.g., a natural or evolved deaminase, such as a cytidine deaminase that include APOBEC1 (“apolipoprotein B mRNA editing enzyme, catalytic polypeptide 1”), CDA (“cytidine deaminase”), and AID (“activation- induced cytidine deaminase”)) domains. In some embodiments, base editors may also include proteins or domains that alter cellular DNA repair processes to increase the efficiency and/or stability of the resulting single-nucleotide change. Two major classes of base editors have been developed: cytidine base editors (CBEs) (e.g., BE4) that allow C:G to T:A conversions and adenine base editors (ABEs) (e.g., ABE7.10) that allow A:T to G:C conversions. Base editors are composed by a catalytically dead Cas9 (dCas9) or a nickase Cas9 (nCas9) fused to a deaminase and guided by a sgRNA to the locus of interest. The d/nCas9 recognizes a specific PAM sequence and the DNA unwinds thanks to the complementarity between the sgRNA and the DNA sequence usually located upstream of the PAM (also called protospacer). Then, the
opposite DNA strand is accessible to the deaminase that converts the bases located in a specific DNA stretch of the proto spacer. Compared to HDR-based strategies, base editing is a promising tool to precisely correct genetic mutations as it avoids gene disruption by NHEJ associated with failed HDR- mediated gene correction. Rat deaminase AP0BEC1 (rAPOBECl) fused to deactivated Cas9 (dCas9) has been used to successfully convert cytidines to thymidines upstream of the PAM of the sgRNA. In some embodiments, this first BE system was optimized by changing the dCas9 to a “nickase” Cas9 D 1 OA, which nicks the strand opposite the deaminated cytidine. Without being bound by theory, this is expected to initiate long-patch base excision repair (BER), where the deaminated strand is preferentially used to template the repair to produce a U:A base pair, which is then converted to T:A during DNA replication.
[0662] In some embodiments, a base editor is a nucleobase editor containing a first DNA binding protein domain that is catalytically inactive, a domain having base editing activity, and a second DNA binding protein domain having nickase activity, where the DNA binding protein domains are expressed on a single fusion protein or are expressed separately (e.g., on separate expression vectors). In some embodiments, a base editor is a fusion protein comprising a domain having base editing activity (e.g., cytidine deaminase or adenosine deaminase), and two nucleic acid programmable DNA binding protein domains (napDNAbp), a first comprising nickase activity and a second napDNAbp that is catalytically inactive, wherein at least the two napDNAbp are joined by a linker. In some embodiments, a base editor is a fusion protein that comprises a DNA domain of a CRISPR-Cas (e.g., Cas9) having nickase activity (nCas; nCas9), a catalytically inactive domain of a CRISPR-Cas protein (e.g., Cas9) having nucleic acid programmable DNA binding activity (dCas; e.g., dCas9), and a deaminase domain, wherein the dCas is joined to the nCas by a linker, and the dCas is immediately adjacent to the deaminase domain. In some embodiments, a base editor is an adenine-to-thymine or “ATBE” (or thymine- to-adenine or “TABE”) transversion base editor. Exemplary base editor and base editor systems include any as described in patent publication Nos. US20220127622, US20210079366, US20200248169, US20210093667, US20210071163, W02020181202, WO2021158921,
WO2019126709, W02020181178, W02020181195, WO2020214842, W02020181193, which are hereby incorporated in their entirety.
[0663] In some embodiments, a gene editing technology is Programmable Addition via Site-specific Targeting Elements (PASTE). In some aspects, PASTE is platform in which genomic insertion is directed via a CRISPR-Cas9 nickase fused to both a reverse transcriptase and serine integrase. As described in loannidi et al. (doi.org/10.1101/2021.11.01.466786), PASTE does not generate double stranded breaks, but allows for integration of sequences as large as ~36 kb. In some embodiments, a serine integrase can be any known in the art. In some embodiments, a serine integrase has sufficient orthogonality such that PASTE can be used for multiplexed gene integration, simultaneously integrating at least two different genes at at least two genomic loci. In some embodiments, PASTE has editing efficiencies comparable to or better than those of homology directed repair or non-homologous end joining based integration, with activity in non-dividing cells and fewer detectable off-target events.
[0664] Any of the systems for gene disruption described herein can be used and, when also introduced with an appropriate donor template having with an exogenous polynucleotide, e.g., transgene sequences, can result in targeted integration of the exogenous polynucleotide at or near the target site of the genetic disruption. In particular embodiments, the genetic disruption is mediated using a CRISPR/Cas system containing one or more guide RNAs (gRNA) and a Cas protein. Exemplary Cas proteins and gRNA are described above, any of which can be used in HDR mediated integration of an exogenous polynucleotide into a target locus to which the Crispr/Cas system is specific for. It is within the level of a skilled artisan to choose an appropriate Cas nuclease and gRNA, such as depending on the particular target locus and target site for cleavage and integration of the exogenous polynucleotide by HDR. Further, depending on the target locus a skilled artisan can readily prepare an appropriate donor template, such as described further below.
[0665] In some embodiments, the DNA editing system is an RNA-guided CRISPR/Cas system (such as RNA-based CRISPR/Cas system), wherein the CRISPR/Cas system is capable of creating a double-strand break in the target locus (e.g., safe harbor locus) to induce insertion
of the transgene into the target locus. In some embodiments, the nuclease system is a CRISPR/Cas9 system. In some embodiments, the CRISPR/Cas9 system comprises a plasmidbased Cas9. In some embodiments, the CRISPR/Cas9 system comprises a RNA-based Cas9. In some embodiments, the CR1SPR/Cas9 system comprises a Cas9 mRNA and gRNA. In some embodiments, the CRISPR/Cas9 system comprises a protein/RNA complex, or a plasmid/RNA complex, or a protein/plasmid complex. In some embodiments, there are provided methods for generating modified cells, which comprises introducing into a source cell (e.g., a pluripotent stem cell, e.g., iPSC) a donor template containing a transgene or exogenous polynucleotide sequence and a DNA nuclease system including a DNA nuclease system (e.g., Cas9) and a locusspecific gRNA. In some embodiments, the Cas9 is introduced as an mRNA. In some embodiments, the Cas9 is introduced as a ribonucleoprotein complex with the gRNA.
[0666] Generally, the donor template to be inserted would comprise at least the transgene cassette containing the exogenous polynucleotide of interest (e.g., the tolerogenic factor or CAR) and would optionally also include the promoter. In certain of these embodiments, the transgene cassette containing the exogenous polynucleotide and/or promoter to be inserted would be flanked in the donor template by homology arms with sequences homologous to sequences immediately upstream and downstream of the target cleavage site, i.e., left homology arm (LHA) and right homology arm (RHA). Typically, the homology arms of the donor template arc specifically designed for the target genomic locus to serve as template for HDR. The length of each homology arm is generally dependent on the size of the insert being introduced, with larger insertions requiring longer homology arms.
[0667] In some embodiments, a donor template (e.g., a recombinant donor repair template) comprises: (i) a transgene cassette comprising an exogenous polynucleotide sequence (for example, a transgene operably linked to a promoter, for example, a heterologous promoter); and (ii) two homology arms that flank the transgene cassette and are homologous to portions of a target locus (e.g. safe harbor locus) at either side of a DNA nuclease (e.g., Cas nuclease, such as Cas9 or Cas 12) cleavage site. The donor template can further comprise a selectable marker, a detectable marker, and/or a purification marker.
[06681 In some embodiments, the homology arms are the same length. In other embodiments, the homology arms are different lengths. The homology arms can be at least about 10 base pairs (bp), e.g., at least about 10 bp, 15 bp, 20 bp, 25 bp, 30 bp, 35 bp, 45 bp, 55 bp, 65 bp, 75 bp, 85 bp, 95 bp, 100 bp, 150 bp, 200 bp, 250 bp, 300 bp, 350 bp, 400 bp, 450 bp, 500 bp, 550 bp, 600 bp, 650 bp, 700 bp, 750 bp, 800 bp, 850 bp, 900 bp, 950 bp, 1000 bp, 1.1 kilobases (kb), 1.2 kb, 1.3 kb, 1.4 kb, 1.5 kb, 1.6 kb, 1.7 kb, 1.8 kb, 1.9 kb, 2.0 kb, 2, 1 kb, 2,2 kb, 2,3 kb, 2,4 kb, 2,5 kb, 2,6 kb, 2.7 kb, 2.8 kb, 2.9 kb, 3.0 kb, 3.1 kb, 3.2 kb, 3.3 kb, 3.4 kb, 3.5 kb, 3.6 kb, 3.7 kb, 3.8 kb, 3.9 kb, 4.0 kb, or longer. The homology arms can be about 10 bp to about 4 kb, e.g., about 10 bp to about 20 bp, about 10 bp to about 50 bp, about 10 bp to about 100 bp, about 10 bp to about 200 bp, about 10 bp to about 500 bp, about 10 bp to about I kb, about 10 bp to about 2 kb, about 10 bp to about 4 kb, about 100 bp to about 200 bp, about 100 bp to about 500 bp, about 100 bp to about 1 kb, about 100 bp to about 2 kb, about 100 bp to about 4 kb, about 500 bp to about I kb, about 500 bp to about 2 kb, about 500 bp to about 4 kb, about 1 kb to about 2 kb, about 1 kb to about 2 kb, about 1 kb to about 4 kb, or about 2 kb to about 4 kb.
[0669] In some embodiments, the donor template can be cloned into an expression vector. Conventional viral and non-viral based expression vectors known to those of ordinary skill in the art can be used.
[0670] In some embodiments, the target locus targeted for integration may be any locus in which it would be acceptable or desired to target integration of an exogenous polynucleotide or transgene. Non-limiting examples of a target locus include, but are not limited to, a CXCR4 gene, an albumin gene, a SHS231 locus, an F3 gene (also known as CD142), a MICA gene, a M1CB gene, a LRP1 gene (also known as CD91), a HMGB1 gene, an ABO gene, a RHD gene, a FUT1 gene, a KDM5D gene (also known as HY), a B2M gene, a CIITA gene, a CCR5 gene, a F3 (i.e., CD142) gene, a MICA gene, a MICB gene, a LRP1 gene, a HMGB1 gene, an ABO gene, a RHD gene, a FUT1 gene, a KDM5D (i.e., HY) gene, a PDGFRa gene, a OLIG2 gene, and/or a GFAP gene. In some embodiments, the exogenous polynucleotide can be inserted in a suitable region of the target locus (e.g., safe harbor locus), including, for example, an intron, an exon, and/or gene coding region (also known as a Coding Sequence, or "CDS"). In some
embodiments, the insertion occurs in one allele of the target genomic locus. In some embodiments, the insertion occurs in both alleles of the target genomic locus. In either of these embodiments, the orientation of the transgene inserted into the target genomic locus can be either the same or the reverse of the direction of the gene in that locus.
[0671] In some embodiments, the exogenous polynucleotide is interested into an intron, exon, or coding sequence region of the safe harbor gene locus. In some embodiments, the exogenous polynucleotide is inserted into an endogenous gene wherein the insertion causes silencing or reduced expression of the endogenous gene. Exemplary genomic loci for insertion of an exogenous polynucleotide are depicted in Table 6.
Table 6. Exemplary genomic loci for insertion of exogenous polynucleotides.
[0672] [In some embodiments, the target locus is a safe harbor locus. In some embodiments, a safe harbor locus is a genomic location that allows for stable expression of integrated DNA with minimal impact on nearby or adjacent endogenous genes, regulatory element and the like. In some cases, a safe harbor gene enables sustainable gene expression and can be targeted by engineered nuclease for gene modification in various cell types including pluripotent stem cells, including derivatives thereof, and differentiated cells thereof. Nonlimiting examples of a safe harbor locus include, but are not limited to, a CCR5 gene locus, a PPP1R12C (also known as AAVS1) gene locus, a CLYBL gene locus, and/or a Rosa gene locus (e.g., ROSA26 gene locus), n some embodiments, the safe harbor locus is selected from the group consisting of the AAVS1 locus, the CCR5 locus, and the CLYBL locus. In some cases SHS231 can be targeted as a safe harbor locus in many cell types. In some cases, certain loci can function as a safe harbor locus in certain cell types. For instance, PDGFRa is a safe harbor for glial progenitor cells (GPCs), OLIG2 is a safe harbor locus for oligodendrocytes, and GFAP is a safe harbor locus for astrocytes. It is within the level of a skilled artisan to choose an appropriate safe harbor locus depending on the particular modified cell type. In some cases, more than one safe harbor gene can be targeted, thereby introducing more than one transgene into the genetically modified cell.
[0673] In some embodiments, there are provided methods for generating modified cells, which comprises introducing into a source cell (e.g. a pluripotent stem cell, e.g. iPSC) a donor template containing a transgene or exogenous polynucleotide sequence and a DNA nuclease system including a DNA nuclease system (e.g. Cas9) and a locus-specific gRNA that comprise complementary portions (e.g. gRNA targeting sequence) specific to a CCR5 gene locus, a PPP1R12C (also known as AAVS1) gene locus, a CLYBL gene locus, and/or a Rosa gene locus (e.g., ROSA26 gene locus). In some embodiments, the genomic locus targeted by the gRNAs is located within 4000 bp, within 3500 bp, within 3000 bp, within 2500 bp, within 2000 bp, within 1500 bp, within 1000 bp, or within 500 bp of any of the loci as described.
[0674] In some embodiments, the gRNAs used herein for HDR-mediated insertion of a transgene comprise a complementary portion (e.g. gRNA targeting sequence) that recognizes a
target sequence in AAVS1. In certain of these embodiments, the target sequence is located in intron 1 of AAVS 1. AAVS1 is located at Chromosome 19: 55,090,918-55,117,637 reverse strand, and AAVS1 intron 1 (based on transcript ENSG00000125503) is located at Chromosome 19: 55,117,222-55,112,796 reverse strand. In certain embodiments, the gRNAs target a genomic locus within 4000 bp, within 3500 bp, within 3000 bp, within 2500 bp, within 2000 bp, within 1500 bp, within 1000 bp, or within 500 bp of Chromosome 19: 55, 117,222-55, 112,796. In certain embodiments, the gRNAs target a genomic locus within 4000 bp, within 3500 bp, within 3000 bp, within 2500 bp, within 2000 bp, within 1500 bp, within 1000 bp, or within 500 bp of Chromosome 19: 55,115,674. In certain embodiments, the gRNA is configured to produce a cut site at Chromosome 19: 55, 115,674, or at a position within 5, 10, 15, 20, 30, 40 or 50 nucleotides of Chromosome 19: 55, 115,674. In certain embodiments, the gRNA s GET000046, also known as "sgAAVSl-1," described in Li et al., Nat. Methods 16:866-869 (2019). This gRNA comprises a complementary portion (e.g., gRNA targeting sequence) having the nucleic acid sequence set forth in SEQ ID NO: 36 (e.g., Table 7) and targets intron 1 of AAVS1 (also known as PPP1R12C).
[0675] In some embodiments, the gRNAs used herein for HDR-mediated insertion of a transgene comprise a complementary portion (e.g., gRNA targeting sequence) that recognizes a target sequence in CLYBL. In certain of these embodiments, the target sequence is located in intron 2 of CL YBL. CLYBL is located at Chromosome 13: 99,606,669-99,897, 134 forward strand, and CLYBL intron 2 (based on transcript ENST00000376355.7) is located at Chromosome 13: 99,773,011-99,858,860 forward strand. In certain embodiments, the gRNAs target a genomic locus within 4000 bp, within 3500 bp, within 3000 bp, within 2500 bp, within 2000 bp, within 1500 bp, within 1000 bp, or within 500 bp of Chromosome 13: 99,773,011- 99,858,860. In certain embodiments, the gRNAs target a genomic locus within 4000 bp, within 3500 bp, within 3000 bp, within 2500 bp, within 2000 bp, within 1500 bp, within 1000 bp, or within 500 bp of Chromosome 13: 99,822,980. In certain embodiments, the gRNA is configured to produce a cut site at Chromosome 13: 99,822,980, or at a position within 5, 0, 15, 20, 30, 40 or 50 nucleotides of Chromosome 13: 99,822,980. In certain embodiments, the gRNA is
GET000047, which comprises a complementary portion (e.g., gRNA targeting sequence) having the nucleic acid sequence set forth in SEQ ID NO: 36 (e.g., Table 7) and targets intron 2 of CLYBL. The target site is similar to the target site of the TALENs as described in Cerbini et al., PLoS One, 10(1): eOl 16032 (2015).
[0676] In some embodiments, the gRNAs used herein for HDR-mediated insertion of a transgene comprise a complementary portion (e.g., gRNA targeting sequence) that recognizes a target sequence in CCR5. In certain of these embodiments, the target sequence is located in exon 3 of CCR5. CCR5 is located at Chromosome 3: 46,370,854-46,376,206 forward strand, and CCR5 exon 3 (based on transcript ENST00000292303.4) is located at Chromosome 3: 46,372,892-46,376,206 forward strand. In certain embodiments, the gRNAs target a genomic locus within 4000 bp, within 3500 bp, within 3000 bp, within 2500 bp, within 2000 bp, within 1500 bp, within 1000 bp, or within 500 bp of Chromosome 3: 46,372,892-46,376,206. In certain embodiments, the gRNAs target a genomic locus within 4000 bp, within 3500 bp, within 3000 bp, within 2500 bp, within 2000 bp, within 1500 bp, within 1000 bp, or within 500 bp of Chromosome 3: 46,373,180. In certain embodiments, the gRNA is configured to produce a cut site at Chromosome 3: 46,373,180, or at a position within 5, 10, 15, 20, 30, 40, or 50 nucleotides of Chromosome 3: 46,373,180. In certain embodiments the gRNA is GET000048, also known as "crCCR5_D," described in Mandal et al., Cell Stem Cell 15:643-652 (2014). This gRNA comprises a complementary portion having the nucleic acid sequence set forth in SEQ ID NO: 37 (e.g., Table 7) and targets exon 3 of CCR5 (alternatively annotated as exon 2 in the Ensembl genome database). See Gomez-Ospina et al., Nat. Comm. 10( 1 ):4045 (2019).
[0677] Table 7 sets forth exemplary gRNA targeting sequences. In some embodiments, the gRNA targeting sequence may contain one or more thymines in the complementary portion sequences set forth in Table 7 are substituted with uracil.
[0678] In some embodiments, the target locus is a locus that is desired to be knocked out in the cells. In such embodiments, such a target locus is any target locus whose disruption or elimination is desired in the cell, such as to modulate a phenotype or function of the cell. For instance, any of the gene modifications described herein to reduce expression of a target gene may be a desired target locus for targeted integration of an exogenous polynucleotide, in which the genetic disruption or knockout of a target gene and overexpression by targeted insertion of an exogenous polynucleotide may be achieved at the same target site or locus in the cell. For instance, the HDR process may be used to result in a genetic disruption to eliminate or reduce expression of (e.g. knock out) any target gene set forth herein while also integrating (e.g. knocking in) an exogenous polynucleotide into the target gene by using a donor template with flanking homology arms that are homologous to nucleic acid sequences at or near the target site of the genetic disruption.
[0679] In some embodiments, there are provided methods for generating modified cells, which comprises introducing into a source cell (e.g., a pluripotent stem cell, e.g. iPSC) a donor template containing a transgene or exogenous polynucleotide sequence and a DNA nuclease system including a DNA nuclease system (e.g. Cas9) and a locus- specific gRNA that comprise complementary portions specific to the B2M locus or the CIITA locus. In some embodiments, the genomic locus targeted by the gRNAs is located within 4000 bp, within 3500 bp, within 3000 bp, within 2500 bp, within 2000 bp, within 1500 bp, within 1000 bp, or within 500 bp of any of the loci as described.
[0680] In particular embodiments, the target locus is B2M. In some embodiments, the modified cell comprises a genetic modification targeting the B2M gene. In some embodiments, the genetic modification targeting the B2M gene is by using a targeted nuclease system that comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid sequence for specifically targeting the B2M gene. In some embodiments, the at least one guide ribonucleic acid (gRNA) sequence for specifically targeting the B2M gene is selected from the group consisting of SEQ ID NOS:81240-85644 of Appendix 2 or Table 15 of
WO20 16/183041, the disclosure is incorporated by reference in its entirety. In some embodiments, an exogenous polynucleotide is integrated into the disrupted B2M locus by HDR by introducing a donor template containing the exogenous polynucleotide sequence with flanking homology arms homologous to sequences adjacent to the target site targeted by the gRNA.
[0681] In particular embodiments, the target locus is CIITA. In some embodiments, the modified cell comprises a genetic modification targeting the CIITA gene. In some embodiments, the genetic modification targeting the CIITA gene is by a targeted nuclease system that comprises a Cas protein or a polynucleotide encoding a Cas protein, and at least one guide ribonucleic acid sequence for specifically targeting the CIITA gene. In some embodiments, the at least one guide ribonucleic acid sequence for specifically targeting the CIITA gene is selected from the group consisting of SEQ ID NOS:5184-36352 of Appendix 1 or Table 12 of W02016183041, the disclosure is incorporated by reference in its entirety. In some embodiments, an exogenous polynucleotide is integrated into the disrupted CIITA locus by HDR by introducing a donor template containing the exogenous polynucleotide sequence with flanking homology arms homologous to sequences adjacent to the target site targeted by the gRNA.
[0682] In some embodiments, it is within the level of a skilled artisan to identify new loci and/or gRNA sequences for use in HDR-mediated integration approaches as described. For example, for CRISPR/Cas systems, when an existing gRNA for a particular locus (e.g., within a target gene) is known, an "inch worming" approach can be used to identify additional loci for targeted insertion of transgenes by scanning the flanking regions on either side of the locus for PAM sequences, which usually occurs about every 100 base pairs (bp) across the genome. The PAM sequence will depend on the particular Cas nuclease used because different nucleases usually have different corresponding PAM sequences. The flanking regions on either side of the locus can be between about 500 to 4000 bp long, for example, about 500 bp, about 1000 bp, about 1500 bp, about 2000 bp, about 2500 bp, about 3000 bp, about 3500 bp, or about 4000 bp long. When a PAM sequence is identified within the search range, a new guide can be designed
according to the sequence of that locus for use in genetic disruption methods. Although the CRISPR/Cas system is described as illustrative, any HDR-mediated approaches as described can be used in this method of identifying new loci, including those using ZFNs, TALENS, meganucleases and transposases.
[0683] In some embodiments, the exogenous polynucleotide encodes an exogenous CD47 polypeptide (e.g., a human CD47 polypeptide) and the exogenous polypeptide is inserted into a safe harbor gene loci or a safe harbor site as disclosed herein or a genomic locus that causes silencing or reduced expression of the endogenous gene. In some embodiments, the exogenous polynucleotide encoding CD47 is inserted in a CCR5 gene locus, a PPP1R12C (also known as AAVS1) gene locus, a CLYBL gene locus, and/or a Rosa gene locus (e.g., ROSA26 gene locus). In some embodiments, the polynucleotide is inserted in a B2M, CIITA, PD1 or CTLA4 gene locus.
[0684] In some embodiments, the modified cell that includes the exogenous polynucleotide is an islet cell and includes a first exogenous polynucleotide that encodes a CD47 polypeptide. In some embodiments, the modified pluripotent stem cells (e.g. modified iPSC) further comprises one or more additional exogenous polynucleotides that encode one or more complement inhibitors or other tolerogenic polypeptides described herein. In some embodiments, the modified pluripotent stem cells (e.g. modified iPSC) comprises reduced expression of CD 142 and reduced expression of MHC class I and/or reduced expression of MHC class II. In some embodiments, the first exogenous polynucleotide and the one or more additional exogenous polynucleotide are inserted into the same genomic locus. In some embodiments, the first exogenous polynucleotide and the one or more additional exogenous polynucleotide are inserted into different genomic loci. In exemplary embodiments, the modified (e.g., hypoimmunogenic) cell is an islet cell derived from an modified (e.g., hypoimmunogenic) pluripotent cell (e.g., an iPSC).
[0685] In some embodiments, the cell is an islet cell. In some embodiments, the cell is an iPSC-derived cell that has been differentiated from a modified iPSC. In some embodiments,
the cell comprises reduced or eliminated expression of CD 142. In some embodiments, the cell comprises overexpression or increased expression of one or more complement inhibitor.
[0686] In some embodiments, the cell is an iPSC-derived islet cell that is modified to contain modifications (e.g. genetic modifications) described herein. In some embodiments, the cell comprises reduced or eliminated expression of CD 142. In some embodiments, the cell comprises overexpression or increased expression of one or more complement inhibitor. In some embodiments, the modified (e.g. hypoimmunogenic) islet cell can be used to treat a variety of indications with allogenic cell therapy, including any as described herein. In some embodiments, the modified (e.g. hypoimmunogenic) islet cell can be used to treat diabetes, such as type I diabetes.
[0687] In some embodiments, the cells that are modified as provided herein are cells from a healthy subject, such as a subject that is not known or suspected of having a particular disease or condition to be treated. g. Safety Switches
[0688] In some embodiments, a safety switch can be incorporated, such as introduced, into the engineered cells provided herein to provide the ability to induce death or apoptosis of engineered cells containing the safety switch, for example if the cells grow and divide in an undesired manner or cause excessive toxicity to the host. Thus, the use of safety switches enables one to conditionally eliminate aberrant cells in vivo and can be a critical step for the application of cell therapies in the clinic. Relevant information concerning safety switches as referred to in the context of the present disclosure may be found from WO2021/146627, the contents of which are herein incorporated by reference. It will be understood that embodiments concerning safety switches may be readily applied to any cell type as described herein, as well as combined with embodiments relating to HIP cells and other modification/gene edit as described herein. The following definitions apply to the present disclosure:
[0689] The term “safety switch” used herein refers to a system for controlling the expression of a gene or protein of interest that, when downregulated or upregulated, leads to
clearance or death of the cell, e.g., through recognition by the host’s immune system. A safety switch can be designed to be triggered by an exogenous molecule in case of an adverse clinical event. A safety switch can be engineered by regulating the expression on DNA, RNA and/or protein levels. A safety switch includes a protein or molecule that allows for the control of cellular activity in response to an adverse event. In some embodiments, a safety switch is a ‘kill switch’ that is expressed in an inactive state and is fatal to a cell expressing the safety switch upon activation of the switch by a selective, externally provided agent. In some embodiments, a safety switch gene is cis-acting in relation to the gene of interest in a construct. In some embodiments, a safety switch is an “uncloaking” system, wherein upon activation, cells downregulate expression of immunosuppressive factors and/or upregulate expression of immune signaling molecules, thereby marking the cell for elimination by the host immune system. Activation of a safety switch causes a cell to kill solely itself, causes a cell to kill itself and neighboring cells through apoptosis or necrosis, or causes a cell to be killed by the host immune system.
[0690] ‘HLA” or “human leukocyte antigen” complex is a gene complex encoding the major histocompatibility complex (MHC) proteins in humans. These cell-surface proteins that make up the HLA complex are responsible for the regulation of the immune response to antigens. In humans, there are two MHCs, class I and class II, “HLA-I” and “HLA-II”. HLA-I includes three proteins, HLA-A, HLA-B and HLA-C, which present peptides from the inside of the cell, and antigens presented by the HLA-I complex attract killer T cells (also known as CD8+ T cells or cytotoxic T cells). The HLA-I proteins are associated with P-2 microglobulin (B2M). HLA-II includes five proteins, HLA-DP, HLA-DM, HLA-DOB, HLA-DQ and HLA-DR, which present antigens from outside the cell to T lymphocytes. This stimulates CD4+ T cells (also known as helper T cells). It should be understood that the use of either “MHC” or “HLA” is not meant to be limiting, and while the terms were originally species specific (e.g., humans (HLA) or murine (MHC)), these terms are used interchangeably herein.
Immune signaling gene locus
[06911 Provided herein is an isolated cell or a population thereof comprising a safety switch construct described herein. In some embodiments, a construct has been introduced into a target gene locus. In some embodiments, a gene locus is either a safe harbor locus selected from the group consisting of an AAVS1 locus, a CLBYL locus, a CXCR4 locus, a Rosa26 locus, and a CCR5 locus, or an immune signaling gene locus selected from the group consisting of B2M, HLA-A, HLA-B, HLA-C, HLA-D, HLA-E, RFXANK, CIITA, CTLA-4, PD-1, RAET1E/ULBP4, RAET1G/ULBP5, RAET1H/ULBP2, RAET1/ULBP1 , RAET1L/ULBP6, RAET1N/ULBP3, and other ligands of NKG2D. In some embodiments, an isolated cell is an isolated engineered human cell further comprising deletion or reduced expression of MHC class I human leukocyte antigens and/or deletion or reduced expression of MHC class II human leukocyte antigens compared to an unmodified human cell. In some embodiments, an isolated cell further comprises deletion or reduced expression of CIITA, B2M, and/or NLRC5. In some embodiments, an isolated cell is hypoimmunogenic and is a stem cell.
[0692] In some embodiments, an immune signaling gene locus is selected from the group consisting of an B2M, HLA-A, HLA-B, HLA-C, HLA-D, HLA-E, RFXANK, CIITA, CTLA-4, PD-1, RAET1E/ULBP4, RAET1G/ULBP5, RAET1H/ULBP2, RAET1/ULBP1, RAE11L/ULBP6, RAET1N/ULBP3, and other ligands of NKG2D.
[0693] In some embodiments, an immune signaling gene locus is selected from the group consisting of B2M, HLA-A, HLA-B, HLA-C, HLA-D, and HLA-E.
[0694] In some aspects, reduced or eliminated expression of CIITA reduces or eliminates expression of one or more of the following MHC class II are HLA-DP, HLA-DM, HLA-DOA, HLA-DOB, HLA-DQ, and HLA-DR.
Conditional HIP Cells and Methods for Conditional Downregulation of Immunosuppressive Factors
[0695] The introduction of safety switches improves the safety of cell therapies developed using hypoimmunogenic cells (HIP cells). A feature of the HIP cells described herein is the inducible expression of one or more immune regulatory (immunosuppressive) factors. In
some embodiments, an immunosuppressive factor (also referred to herein as “an hypoimmunity factor” or “a tolerogenic factor”) includes, but is not limited to, CD47, CD24, CD200, HLA-G, HLA-E, HLA-C, HLA-E heavy chain, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, Serpinb9, CC121, and Mfge8. In certain embodiments, an immunosuppressive factor is CD47. The regulatable or inducible expression of an immunosuppressive factor functions to control an immune response by a recipient subject to an engrafted hypoimmunogenic cell.
[0696] Described herein are methods for the expression of an immunosuppressive factor that requires a mechanism to ‘turn-off’ expression of the immune regulatory protein in a controlled manner. Also described herein are HIP cells possessing controllable expression of one or more immunosuppressive factors. In some embodiments, cells of the present disclosure overexpress one or more immunosuppressive factors and can be induced to downregulate expression of the one or more immunosuppressive factors. As such, the cells are no longer hypoimmunogenic and are recognized by the recipient’s immune cells for cell death. In some embodiments, the hypoimmunity of cells that are introduced to a recipient subject is achieved through the overexpression of an immunosuppressive molecule, including hypoimmunity factors and complement inhibitors accompanied with the repression or genetic disruption of the HLA-I and HLA-II loci. These modifications cloak the cell from the recipient immune system’s effector cells that are responsible for the clearance of infected, malignant or non-self cells, such as T cells, B cells, NK cells and macrophages. Cloaking a cell from the immune system allows for the existence and persistence of allogeneic cells within the body. Controlled removal of engineered cells from the body is crucial for patient safety and can be achieved by uncloaking the cells from the immune system. Uncloaking serves as a safety switch and can be achieved through downregulation of the immunosuppressive molecules or upregulation of immune signaling molecules. The level of expression of any of the immunosuppressive molecules described herein can be controlled on the protein level, mRNA level, or DNA level in the cells. Similarly, the level of expression of any of the immune signaling molecules described herein can be controlled on the protein level, mRNA level, or DNA level in the cells.
[06971 In some embodiments, any of the safety switch methods described herein (e.g., protein level, RNA level and DNA level safety switches) are used to decrease the level of an immunosuppressive factor in the cells such that the lower level of the immunosuppressive factor is below a threshold level. In some embodiments, the level of the immunosuppressive factor in the cells is decreased by about 10-fold, 9-fold, 8-fold, 7-fold, 6-fold, 5-fold, 4-fold, 3-fold, 2- fold, 1-fold or 0.5-fold below a threshold level of expression. In some embodiments, the level of the immunosuppressive factor in the cells is decreased by about 10-fold to 5-fold, 10-fold to 3- fold, 9-fold to 1-fold, 8-fold to 1-fold, 7-fold to 0.5-fold, 6-fold, to 1-fold, 5-fold to 0.5-fold, 4- fold to 0.5-fold, 3-fold to 0.5-fold, 2-fold to 0.5-fold, or 1-fold to 0.5-fold below a threshold level of expression. In some embodiments, a threshold level of expression of the immunosuppressive factor is established based on the expression of such factor in an induced pluripotent stem cell. In some embodiments, a threshold level of the immunosuppressive factor expression is established based on the expression level of the immunosuppressive factor in a corresponding hypoimmune cell, such as an MHC I and MHC II knockout cell or an MHC I/MHC II/TCR knockout cell.
Protein Level Control
[0698] In some embodiments, regulated degradation of an immunosuppressive protein is established by incorporating a degron into the amino acid sequence of the immunosuppressive factor that allows recruitment to the endogenous protein turnover machinery. Mechanisms for targeted protein degradation include, but are not limited to, recruitment to an E3 ligase for ubiquitination and subsequent proteasomal degradation, direct recruitment to a proteasome, and recruitment to a lysosome.
[0699] Fusion of inducible degron motifs to immunosuppressive molecules enables exogenous control over the stability of the molecule through the addition or removal of small molecules that stabilize or destabilize the degron, and thus the immunosuppressive molecule.
[0700] In some embodiments, methods for inducible protein degradation by a degron include, but are not limited to, ligand-induced degradation (LID) using a SMASH tag, ligand-
induced degradation using Shield- 1, ligand- induced degradation using auxin, ligand- induced degradation using rapamycin, peptidic degrons (e.g., IKZF3 based degrons), and proteolysistargeting chimeras (PROTACs). In some embodiments of a ligand- induced degradation method, a degron tag is held in an inactive conformation but is induced to adopt a conformation capable of recognition by a proteasome upon binding of a specific molecule, such as but not limited to, a Shield-1 molecule. See, e.g., Roth et al., Cellular Molecular Life Sciences, 2019, 76(14), 2761- 2777 , which is herein incorporated by reference in its entirety. Detailed descriptions of SMASH degron technology can be found in Hannah and Zhou, Nat Chem Biol, 2015, 11:637-638 and Chung et al., Nat Chem Biol, 2015, 11:713-720, which are herein incorporated by reference in their entireties. Detailed descriptions of LID degron technologies can be found in Bonger et al., Nat Chem Biol, 2011, 7(8):531-7, which is herein incorporated by reference in its entirety.
[0701] In some aspects, provided herein are methods for controlling the immunogenicity of a mammalian cell (e.g., a human cell) by obtaining an isolated cell and introducing a construct containing a constitutive promoter operably linked to an inducible degron element that is operably linked to a gene encoding an immunosuppressive factor. In some embodiments, a construct includes a constitutive promoter operably linked to an inducible degron element that is operably linked to a nucleic acid sequence encoding flexible linker that is operable linked to a gene encoding an immunosuppressive factor. In some embodiments, a construct comprising a constitutive promoter operably linked to a gene encoding an immunosuppressive factor that is operably linked to an inducible degron element. In some embodiments, a construct includes a constitutive promoter operably linked to a gene encoding an immunosuppressive factor that is linked to a sequence encoding a flexible linker that is operably linked to an inducible degron element. As such, the degron targets the immunosuppressive factor for degradation upon contacting the cell with a degron ligand or molecule.
[0702] In some embodiments, an inducible degron element is selected from the group consisting of a ligand-inducible degron element such as a small molecule-assisted shutoff (SMASH) degron element, Shield- 1 responsive degron element, auxin responsive degron element, and rapamycin responsive degron element; a peptidic degron element; and a peptidic
proteolysis targeting chimera (PROTAC) element. In some embodiments, a ligand-inducible degron element is a small molecule-assisted shutoff (SMASH) degron element and the exogenous factor for controlling immunogenicity is asunaprevir. In some embodiments, the immunosuppressive factor gene is selected from the group consisting of CD47, CD24, CD200, HLA-G, HLA-E, HLA-C, HLA-E heavy chain, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, Serpinb9, CC121, and Mfge8. In some embodiments, the immunosuppressive factor gene is CD47. In some instances, the constitutive promoter of the construct is selected from the group consisting of an EF1A promoter, an EFS promoter, a CMV promoter, a CAGGS promoter, a SV40 promoter, a COPIA promoter, an ACT5C promoter, a TRE promoter, a CBh promoter, a PGK promoter, and a UBC promoter. In some instances, the optional flexible linker is selected from the group consisting of (GSG)n (SEQ ID NO: 25), (GGGS)n (SEQ ID NO: 26), and (GGGSGGGS)n (SEQ ID NO: 27), wherein n is 1-10.
[0703] In some embodiments, a construct is introduced into a cell to be integrated into a safe harbor locus, such as but not limited to, an AAVS1 locus, a CLBYL locus, a CXCR4 locus, a Rosa26 locus, and a CCR5 locus. In some embodiments, a construct is introduced into an AAVS locus in the cell by way of homology directed recombination. As such, the construct includes 5’ and 3’ homology arms specific to the targeted safe harbor locus. In some embodiments, a construct comprises from 5’ end to 3’ end: a 5’ homology arm to the AAVS1 locus, an exogenous constitutive promoter, an inducible degron element, a gene encoding an immunosuppressive factor, and a 3’ homology arm to the AAVS1 locus. In other embodiments, a construct comprises from 5’ end to 3’ end: a 5’ homology arm to the AAVS1 locus, an exogenous constitutive promoter, an inducible degron element, a sequence encoding a flexible linker, a gene encoding an immunosuppressive factor, and a 3’ homology arm to the AAVS1 locus. In some embodiments, an engineered cell includes an exogenous nucleic acid sequence comprising a constitutive promoter operably linked to an inducible degron element that is operably linked to an optional sequence encoding a flexible linker that is operably linked to a gene encoding an immunosuppressive factor. The engineered cell expresses the inducible degron clement fused or linked to an immunosuppressive factor. In some embodiments, the cell
is contacted by a factor or agent such as, but not limited to, a ligand, molecule, peptide or small molecule, which activates the degron element to degrade the immunosuppressive factor.
[0704] In some embodiments of a peptidic degron, a peptide tag is used that confers small molecule-mediated recruitment to an E3 ligase. In some embodiments, the peptide tag comprises the lymphoid-restricted transcription factor IKZF3 that is recruited to the E3 ligase receptor (CRBN) in an immunomodulatory drug (IMiD) dependent manner, as described in Koduri et al., Proc Natl Acad Sci, 2019, 116(7), 2539-2544, which is herein incorporated by reference in its entirety. In certain embodiments, the degron is capable of targeting immunosuppressive factors for degradation (e.g., through a ubiquitination pathway), inducing protein degradation, or degrading proteins.
[0705] In some aspects, provided herein are methods for controlling the immunogenicity of a mammalian cell (e.g., a human cell) by obtaining an isolated cell and introducing a construct including a constitutive promoter, an inducible peptidic degron element, and a gene encoding an immunosuppressive factor. In some embodiments, the construct includes a constitutive promoter, an inducible peptidic degron element, a nucleic acid sequence encoding flexible linker, and a gene encoding an immunosuppressive factor. Any of the constitutive promoters, immunosuppressive factors, flexible linkers, and cells described herein are applicable to the method.
[0706] In some embodiments of a PROTAC, a bi-functional molecule is used to recruit an immunosuppressive factor to the protein degradation machinery of a cell. In some embodiments, a bi-functional molecule binds to the native or wild-type sequence of the immunosuppressive protein or an engineered version of the immunosuppressive protein expressing a domain that binds to the bi-functional molecule with high affinity. In some embodiments, the bi-functional molecule comprises a small molecule or a biologic agent (e.g., an antibody or fragment thereof). See, e.g., Burslem et al., Cell Chemical Biology, 2018, 25, 67-77 and Roth et al., Cellular Molecular Life Sciences, 2019, 76(14), 2761-2777, which are herein incorporated by reference in their entirety.
[07071 In some embodiments of a bi-functional antibody, the antibody targets an immunosuppressive factor and a second endogenous receptor, which leads to internalization and degradation. Controllable expression of one or more immunosuppressive factors can be provided by way of a bi-functional antibody (e.g., a chemically reprogrammed bi-functional antibody), inducible protein degradation by a degron, inducible RNA regulation, inducible DNA regulation, and an inducible expression method. See, e.g., Natsume and Kanemaki, Annu Rev Genet, 2017, 51 , 82-102; Burslem and Crews, Chem Rev, 2017, 1 17, 11269- 11301 ; Banik et al., ChemRxiv, 2019; which are herein incorporated by reference in their entirety. In some embodiments, a cell expressing an immunosuppressive factor is contacted by an antibody that binds the cell for degradation.
[0708] In some instances, hypoimmune cells are cleared by the immune system through the addition of an antibody that binds an epitope on the extracellular surface of the cell. The epitope can be native to the overexpressed immunosuppressive factor, or can be another epitope located within the immunosuppressive factor or distinctly located at the extracellular surface. Binding of an antibody to the surface uncloaks the cell and leads to antibody-dependent cellular cytotoxicity (ADCC) or complement-dependent cytotoxicity (CDC).
[0709] In some embodiments, an ADCC/CDC safety switch epitope is selected from the group consisting of EGFR, CD20, CD19, CCR4, HER2, MUC1, GD2, PSMA, CD30, CD16, and fragments, derivatives, and variants thereof. In some instances, any of the cells described herein express an epitope selected from an EGFR epitope, CD20 epitope, CD 19 epitope, CCR4 epitope, HER2 epitope, MUC1 epitope, GD2 epitope, PSMA epitope, CD30 epitope, or CD16 epitope. In some embodiments, the cells bind to an antibody specific to EGFR, CD20, CD 19, CCR4, HER2, MUC1, GD2, PSMA, CD30, or CD 16, which leads to ADCC/CDC.
[0710] The methods directed to a protein level safety switch as described herein provides a way for decreasing the level of an immunosuppressive factor (e.g., CD47) in an regulatable manner in engineered cells described herein (e.g., hypoimmune cells). By lowering the level of the immunosuppressive factor such as CD47 below a threshold level in the cells using any of the safety switch methods described herein, the recipient subject’s immune system can initiate an
immune response to such cells. In some embodiments, the level of CD47 in the engineered cells is decreased by the safety switch by about 10-fold, 9-fold, 8-fold, 7-fold, 6-fold, 5-fold, 4-fold, 3- fold, 2-fold, 1-fold or 0.5-fold below a threshold level of expression. In some embodiments, the level of CD47 in the engineered cells is decreased by about 10-fold to 5-fold, 10-fold to 3-fold, 9-fold to 1-fold, 8-fold to 1-fold, 7-fold to 0.5-fold, 6-fold, to 1-fold, 5-fold to 0.5-fold, 4-fold to 0.5-fold, 3-fold to 0.5-fold, 2-fold to 0.5-fold, or 1-fold to 0.5-fold below a threshold level of expression. In some instances, the threshold level of CD47 expression is established based on the exogenous expression of CD47 in an induced pluripotent stem cell. In other instances, the threshold level of CD47 expression is established based on the expression level of CD47 in a corresponding hypoimmune cell, such as an MHC I and MHC II knockout cell or an MHC I/MHC II/TCR knockout cell. In some instances, the level of CD47 is reduced using a degron- based safety switch such as, but not limited to, a SMASH degron or a LID degron. In some embodiments, the cells expressing a SMASH degron linked to an exogenous CD47 transgene are exposed to the small molecule asunaprevir (the degron inducer), which thereby induces a reduction of expression of the exogenous CD47 by the cells.
RNA Level Control
[0711] Immunosuppressive factors can be targeted by siRNAs or miRNAs, thereby leading to the degradation of the transcript encoding the factors. An siRNA can be exogenously provided or genetically encoded to provide control over transcription of the inhibitory RNA. The siRNA or miRNA can anneal to the immunosuppressive factor’s transcript, resulting in degradation by the RISC complex.
[0712] In some embodiments, methods for inducible RNA regulation to downregulate expression of an immunosuppressive factor include, but are not limited to, shRNAs induced by a small molecule or a biologic agent, inducible siRNAs, inducible miRNAs, inducible CRISPR interference (CRISPRi), and inducible RNA targeting nucleases.
[07131 In some embodiments, the method comprises an shRNA or siRNA targeting the RNA of the immunosuppressive factor. In some instances, expression of the shRNA or siRNA is induced by a small molecule or biologic agent.
[0714] In some aspects, provided are methods for controlling the immunogenicity of a mammalian cell (e.g., a human cell) by obtaining an isolated cell and introducing a construct containing an inducible RNA polymerase promoter operably linked an shRNA sequence targeting an immunosuppressive factor that is operably linked to a constitutive promoter that is operably linked to a transactivator element that can control the inducible RNA polymerase promoter. In some embodiments, the construct includes a U6Tet promoter, an shRNA targeting an immunosuppressive factor, a constitutive promoter, and a Tet Repressor element that is responsive to tetracycline or a derivative thereof (e.g., doxycycline). In other instances, the shRNA eliminates expression of the immunosuppressive factor. In other instances, the shRNA decreases expression of the immunosuppressive factor by about 99% or less, e.g., 99%, 98%, 97%, 96%, 95%, 94%, 93%, 92%, 90%, 85% or less. In some embodiments, the inducible promoter is a tetracycline responsive promoter. Any of the constitutive promoters, immunosuppressive factors, and cells described herein are applicable to the method.
[0715] In many embodiments, the engineered cell expresses an inducible shRNA that targets an immunosuppressive factor. In some embodiments, the cell also expresses an exogenous immunosuppressive factor that mediates the hypoimmunogenicity of the cell. In some embodiments, the cell is contacted by a factor such as, but not limited to, a ligand, molecule, peptide or small molecule, that activates the expression of the shRNA to degrade the immunosuppressive factor.
[0716] In some embodiments, the method comprises a CRISPR interference system (CRISPRi) for targeting the promoter of an immunosuppressive factor to downregulate its transcription. In some instances, expression of a CRISPRi and/or a gRNA targeting the immunosuppressive factor is induced by a small molecule or biologic agent. Detailed description of CRISPRi methods are found in, e.g., Engreitz et al., Cold Spring Harb Perspect Biol, 2019, l l:a035386, which is herein incorporated by reference in its entirety. In some embodiments, the
CRISPRi system utilizes a dCas9-repressor fusion protein that is controlled by a constitutive promoter and a gRNA specific to the immunosuppressive factor under the control of an inducible promoter.
[0717] In some aspects, provided are methods for controlling the immunogenicity of a mammalian cell (e.g., a human cell) by obtaining an isolated cell and introducing into the cell (i) a first construct containing a constitutive promoter operably linked to a gene encoding an immunosuppressive factor; (ii) a second construct containing a constitutive promoter operably linked to a gene encoding a Cas9 nuclease or variant thereof such as dCas9-repressor fusion protein; and (iii) a third construct comprising an inducible RNA polymerase promoter operably linked to a gRNA sequence targeting the sequence encoding the immunosuppressive factor such that the gRNA sequence is operably linked to a transactivator element that corresponds to the inducible RNA polymerase promoter. In some instances, the first construct, second construct, and third construct are found in a single vector. In some instances, the first construct, second construct, and third construct are found in two vectors.
[0718] In some embodiments, the CRISPR based method includes a nuclease for targeting the mRNA sequence corresponding to the immunosuppressive factor such as, but not limited to, Casl3, Cas7, or Csxl. In some instances, expression of a nuclease and/or a gRNA targeting the immunosuppressive factor is induced by a small molecule or biologic agent.
[0719] In some aspects, provided are methods for controlling the immunogenicity of a mammalian cell (e.g., a human cell) by obtaining an isolated cell and introducing into the cell (i) a first construct comprising a constitutive promoter operably linked to a gene encoding an immunosuppressive factor; (ii) a second construct comprising a constitutive promoter operably linked to a gene encoding a Casl3a nuclease, a variant thereof, or a fusion protein thereof; and (iii) a third construct comprising an inducible RNA polymerase promoter operably linked to a gRNA sequence targeting the sequence encoding the immunosuppressive factor such that the gRNA sequence is operably linked to a transactivator element that corresponds to the inducible RNA polymerase promoter.
[07201 In some embodiments, inducible expression systems that are useful for RNA level control of the immunosuppressive factor include, but are not limited to, ligand inducible transcription factor systems, receptor mediated expression control systems, and ligand regulated riboswitches. In some embodiments, the inducible expression system comprises a tetracycline- controlled operator system, a synthetic Notch-based (SynNotch) system (see, e.g., Morsut et ah, Cell, 2016, 164:780-791 and Yang et al., Commun Biol, 2020, 3:116), and riboswitch that regulates expression of the immunosuppressive factor gene by ligand (e.g., aptamer, peptide or small molecule) mediated alternative splicing of the resulting pre-mRNA. Useful riboswitches comprise a sensor region and an effector region that sense the presence of a ligand and alter the splice of the target immunosuppressive factor gene. Detailed descriptions and examples of riboswitch gRNAs are found in e.g., US 9,228,207; US 9,993,491; and US 10,421,989; and Seeliger et ah, PLoS One, 2012, 7(l):e29266; the contents are herein incorporated by reference in their entirety.
[0721] In some embodiments, the level of an immunosuppressive factor such as CD47 in the engineered cells is decreased by an RNA level safety switch by about 10-fold, 9-fold, 8-fold, 7-fold, 6-fold, 5-fold, 4-fold, 3-fold, 2-fold, 1-fold or 0.5-fold below a threshold level of expression. In some embodiments, the level of CD47 in the engineered cells is decreased by about 10-fold to 5-fold, 10-fold to 3-fold, 9-fold to 1-fold, 8-fold to 1-fold, 7-fold to 0.5-fold, 6- fold, to 1-fold, 5-fold to 0.5-fold, 4-fold to 0.5-fold, 3-fold to 0.5-fold, 2-fold to 0.5-fold, or 1- fold to 0.5-fold below a threshold level of expression. In some instances, the threshold level of CD47 expression is established based on the exogenous expression of CD47 in an induced pluripotent stem cell. In other instances, the threshold level of CD47 expression is established based on the expression level of CD47 in a corresponding hypoimmune cell, such as an MHC I and MHC II knockout cell or an MHC I/MHC II/TCR knockout cell.
DNA Level Control
[0722] Transcriptional regulation of immunosuppressive factors through employing inducible promoters provides the ability to turn expression of the switch on or off at will through the addition or removal of small molecules, such as, but not limited to, doxycycline. Genetic
disruption via targeted nuclease activity can eliminate expression of the immunosuppressive factor to uncloak the cells as well.
[0723] In some embodiments, methods for inducible DNA regulation include, but are not limited to, using tissue-specific promoters, inducible promoters, controllable riboswitches, and knockout using an inducible nuclease (e.g., inducible CRISPRs, inducible TALENs, inducible zinc finger nucleases, inducible homing endonucleases, inducible meganucleases, and the like) to target the DNA sequence of one or more immunosuppressive factors. In some embodiments, the inducible nuclease comprises a nuclease such that its expression is controlled by the presence of a small molecule. In some embodiments, the inducible nuclease comprises a nuclease such that delivery of the nuclease RNA or protein to a cells is controlled by the presence of a small molecule. In some embodiments, expression of the nuclease is induced by a small molecule or biologic agent. In some embodiments, expression of a Cas nuclease and/or a guide RNA (gRNA) is induced by a small molecule or biologic agent.
[0724] In some embodiments, methods for inducible expression include, but are not limited to, ligand inducible transcription factors systems (e.g., a tetracycline-controlled operator system), receptor mediated control of expression system (e.g., a SynNotch system), and a ligand regulated riboswitch system for control of mRNA or gRNA activity. Detailed description of inducible expression methods are found in, e.g., Kallunki et al., Cells, 2019, 796 (doi:10.3390/cells8080796), which is herein incorporated by reference in its entirety.
[0725] In some embodiments, the immunosuppressive factors are expressed in a cell using an inducible expression vector. The expression vector can be a viral vector, such as but not limited to, a lentiviral vector. In some embodiments, the inducible immunosuppressive factors described herein are introduced into a cell by lentiviral transduction.
[0726] In some embodiments, the silencing of a construct encoding the immunosuppressive factor results in elimination of the engineered cell by a recipient subject’s immune system. Furthermore, the construct containing the immunosuppressive factor and an inducible expression system can be integrated into an endogenous gene locus to safeguard
expression of the cassette, as silencing of the gene will eliminate the engineered cells. In some embodiments, the endogenous gene locus useful for integration is a core essential gene locus or an immune signaling factor gene locus. Non-limiting examples of a core essential gene locus for such integration include RpS2, RpS9, RpSl l, RpS13, RpS18, RpL8, RpLl l, RpL32, RpL36, Rpnl 1, Psmdl4, and PSMA3. Non-limiting examples of an immune signaling factor gene locus for such integration include B2M, MIC-A/B, HLA-A, HLA-B, HLA-C, RFXANK, CTLA4, PD1 , and ligands of NKG2D (e.g., MICA, MICB, RAET1E/ULBP4, RAET1G/ULBP5, RAET1H/ULBP2, RAET1/ULBP1, RAET1L/ULBP6, and RAET1N/ULBP3).
[0727] In some embodiments, the conditional expression of an immunosuppressive factor is based on regulating expression of the immune regulatory factor CD47. CD47 is a component of the innate immune system that functions as a “do not eat me” signal as part of the innate immune system to block phagocytosis by macrophages. Useful immunosuppressive factors that can be engineered for controlled expression include, but are not limited to, CD47, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, Serpinb9, CCL21, and Mfge8.
[0728] In some embodiments, the present disclosure provides a method of producing a stem cell (e.g., hypoimmunogenic pluripotent stem cell or hypoimmunogenic induced pluripotent stem cell) or a differentiated cell thereof that has been modified to conditionally express any one of the immunosuppressive factors selected from the group consisting of CD47, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, IDO1, CTLA4-Ig, Cl-Inhibitor, IL-10, IL-35, FASL, Scrpinb9, CCL21, and Mfge8. In other embodiments, the immunosuppressive factor is selected from the group consisting of HLA-A, HLA-B, HLA-C, RFX-ANK, C11TA, NFY-A, NLRC5, B2M, RFX5, RFX-AP, HLA-G, HLA-E, NFY-B, PD-L1, NFY-C, IRF1, TAPI, GITR, 4-1BB, CD28, B7-1, CD47, B7-2, 0X40, CD27, HVEM, SLAM, CD226, ICOS, LAG3, TIGIT, TIM3, CD 160, BTLA, CD244, LFA-1 , ST2, HLA-F, CD30, B7-H3, VISTA, TLT, PD-L2, CD58, CD2, and HELIOS.
[0729] In some embodiments, the cells conditionally express one or more of the immunosuppressive factors such that in the absence of the exogenous controlling signal, the cells
are hypoimmunogenic or have reduced hypoimmunogenicity. In the presence of the exogenous controlling signal, the cells are recognized by immune cells and are targeted by cell death or clearance. In some instances, the HIP cells express an immunosuppressive factor that functions allow the HIP cell to evade the recipient subject’s immune response. Upon exposing the HIP cells to an exogenous controlling signal, the expression (e.g., the DNA level expression, the RNA level expression, or the protein level expression) of immunosuppressive factor is downregulated, and thus the HIP cells are recognized by the innate immune system in the recipient subject. As such, the HIP cells undergo cell death and/or cell clearance in the recipient.
[0730] In some embodiments, the level of an immunosuppressive factor such as CD47 in the engineered cells is decreased by a DNA level safety switch by about 10-fold, 9-fold, 8-fold, 7-fold, 6-fold, 5-fold, 4-fold, 3-fold, 2-fold, 1-fold or 0.5-fold below a threshold level of expression. In some embodiments, the level of CD47 in the engineered cells is decreased by about 10-fold to 5-fold, 10-fold to 3-fold, 9-fold to 1-fold, 8-fold to 1-fold, 7-fold to 0.5-fold, 6- fold, to 1-fold, 5-fold to 0.5-fold, 4-fold to 0.5-fold, 3-fold to 0.5-fold, 2-fold to 0.5-fold, or 1- fold to 0.5-fold below a threshold level of expression. In some instances, the threshold level of CD47 expression is established based on the exogenous expression of CD47 in an induced pluripotent stem cell. In other instances, the threshold level of CD47 expression is established based on the expression level of CD47 in a corresponding hypoimmune cell, such as an MHC I and MHC II knockout cell or an MHC I/MHC II/TCR knockout cell.
Conditional HIP Cells and Methods Conditional Upregulation of Immune Signaling Factors
[0731] Described herein are methods for the expression of an immune signaling factor in a controllable manner as to increase the expression of the factor to alter the hypoimmunogenicity of the cell. Also described are HIP cells that possess controllable expression of one or more immune signaling factors. In some aspects, the immune signaling factor is selected from the group consisting of B2M, MIC-A/B, HLA-A, HLA-B, HLA-C, RFXANK, CTLA-4, PD-1, and ligands of NKG2D (e.g., MICA, MICB, RAET1E/ULBP4, RAET1G/ULBP5, RAET1H/ULBP2, RAET1/ULBP1, RAET1L/ULBP6, and RAET1N/ULBP3).
[07321 Controllable expression of one or more immune signaling factors can be provided by way of a inducible ligand stabilization system using a degron, an inducible RNA upregulation system (e.g., an inducible CRISPR activation), and an inducible DNA upregulation system. In some embodiments, the inducible DNA upregulation system comprises inducible CRISPR activation (CRISPRa), tissue-specific promoters, inducible promoters, and riboswitches.
[0733] Detailed description of CRISPRa methods are found in, e.g., Engreitz et al., Cold Spring Harb Perspect Biol, 2019, ll:a035386, which is herein incorporated by reference in its entirety. Detailed descriptions and examples of inducible riboswitches are found in e.g., US 9,228,207; US 9,993,491; and US 10,421,989; and Seeliger et al., PLoS One, 2012, 7(l):e29266; the contents are herein incorporated by reference in their entirety.
[0734] In an example of uncloaking Hypo-Immune cells Through Genetic, Post- Transcriptional, and Post-Translational Regulation, hypoimmunity is achieved through the overexpression of hypoimmune molecules such as CD47, complement inhibitors accompanied with the repression or genetic disruption of the HLA-I and HLA-II loci. These modifications cloak the cell from the immune system’s effector cells that are responsible for the clearance of infected, malignant or non-self cells, such as T-cells, B-cells, NK cells and macrophages. Cloaking of a cell from the immune system allows for existence and persistence of allogeneic cells within the body. Removal of the engineered cells from the body is crucial for patient safety and can be achieved by uncloaking the cells from the immune system. Uncloaking serves as a safety switch and can be achieved through the downregulation of the hypoimmune molecules (for example CD47, CD27, CD200, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, PD-L1, 1DO1, CTLA4-lg, Cl-lnhibitor, IL-10, IL-35, FASL, Serpinb9, CCL21, or Mfge8) or the upregulation of immune signaling molecules (for example B2M, MIC-A/B, HLA-A, HLA-B, HLA-C, RFXANK, CTLA-4, PD-1, and ligands of NKG2D (e.g., MICA, MICB, RAET1E/ULBP4, RAET1G/ULBP5, RAET1H/ULBP2, RAET1/ULBP1 , RAET1L/ULBP6, or RAET1N/ULBP3). Either of these activities will avail the cell to native effector cells, resulting in clearance of the allogeneic cell.
III. Methods
A. Methods of Making a Non-Human Animal
[0735] In another aspect, the present disclosure provides methods of making a nonhuman animal model of type 1 diabetes (T1D), the method comprising: (a) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to the non-human animal, (b) administering islet cells derived from cells of the human patient with T1 D to the non- human animal, wherein the PBMCs and the islet cells are autologous. In some embodiments, a non-human animal is a non-human mammal. In some embodiments, a non-human animal is a rodent. In some embodiments, a non-human animal is a mouse. In some embodiments, a mouse is a NOD mouse, optionally an NSG-SGM3 mouse.
[0736] In some embodiments, a human patient with T1D is eligible for islet transplant therapy. In some embodiments, a human patient exhibits one or more of the following: (a) a clinical history compatible with T1D onset of disease at younger than 40 years old; (b) insulin dependence for at least 5 years; (c) a sum of age and years of insulin dependent diabetes duration of at least 28 years; (d) absent stimulated C-peptide (less than 0.3 ng/mL) 60 and 90 minutes post-mixed-meal tolerance test; (e) involvement of intensive diabetes management, wherein intensive diabetes management comprises: (i) self-monitoring of glucose values no less than a mean of three times each day averaged over each week; (ii) administration of three or more insulin injections each day or insulin pump therapy; and/or (iii) under the direction of an endocrinologist, diabetologist, or diabetes specialist with at least three clinical evaluations during the past 12 months; (f) at least one episode of severe hypoglycemia in the past 12 months, wherein an episode of severe hypoglycemia comprises an event with one or more of the following symptoms: (i) memory loss; (ii) confusion; (iii) uncontrollable behavior; (iv) irrational behavior; (v) unusual difficulty in awakening; (vi) suspected seizure; (vii) seizure; (viii) loss of consciousness; (ix) visual symptoms, and hypoglycemia during the past 12 months; and/or (g) impaired awareness of hypoglycemia (IAH). In some embodiments, a human patient with T1D has had T1D for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10
months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 years, at least 8 years, at least 9 years, at least 10 years at least 15 year’s, at least 20 years, at least 30 year’s, at least 40 years, at least 50 year’s, at least 60 years, at least 70 years, at least 80 years, or at least 90 years. In some embodiments, a human patient with T1D has impaired awareness of hypoglycemia (IAH). In some embodiments, IAH comprises: a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more; a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more; marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more; or a composite of: a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile. In some embodiments, a human patient with T1D has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years. In some embodiments, a human patient with T1D has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2). In some embodiments, antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
[0737] In some embodiments, one or more of the PBMCs are selected from the group consisting of T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, and dendritic cells. In some embodiments, one or more of the PBMCs produce autoantibodies against cells or antigens of the human patient with T1D. In some embodiments, one or more of the PBMCs is a B cell. In some embodiments, one or more of the PBMCs
produce antibodies against one or more islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2. In some embodiments, one or more of the PBMCs produce islet cell antibodies (ICA) and/or insulin autoantibodies (IAA). In some embodiments, one or more of the PBMCs is an islet-reactive T cell. In some embodiments, one or more of the PBMCs are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the nonhuman animal. In some embodiments, a non-human animal of the present disclosure comprises at least 2.5 x 105, at least 5x 105, at least 1 x 106, at least 2x 106, at least 3x 106, at least 4x 106, at least 5xl06, at least 6xl06, at least 7xl06, at least 8xl06, at least 9xl06, at least IxlO7, at least 1.5xl07, or at least 2xl07 PBMCs.
[0738] In some embodiments, islet cells are administered to the non-human animal via intramuscular injection, intravenous injection, intraportal injection, ocular injection, injection into omentum, and/or injection into a kidney capsule. In some embodiments, islet cells produce insulin, glucagon, somatostatin, and/or pancreatic polypeptide.
[0739] In some embodiments, islet cells are or comprise human alpha, beta, and/or delta islet cells. In some embodiments, islet cells are or comprise about 5%, about 10%, about 15%, about 20%, about 25%, about 35%, or about 40% alpha cells. In some embodiments, islet cells are or comprise about 10%, about 15%, about 20%, about 25%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, or about 80% beta cells. In some embodiments, islet cells are or comprise about 1%, about 2%, about 3%, about 4%, about 5%, about 6%, about 7%, about 8%, about 9%, about 10%, about 15%, about 20%, or about 25% delta cells.
[0740] In some embodiments, islet cells are or comprise differentiated cells. In some embodiments, differentiated cells are derived from a stem cell. In some embodiments, differentiated cells are derived from a pluripotent stem cell. In some embodiments, differentiated cells are derived from an induced pluripotent stem cell (iPSC). hi some embodiments, an iPSC is derived from a cell obtained from the human patient with T1D. In some embodiments, a cell obtained from the human patient with T1D is selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord
blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell. In some embodiments, a cell obtained from the human patient with T1D is a PBMC. In some embodiments, a stem cell is derived from a cell obtained from a human patient with T ID. In some embodiments, a stem cell is an induced pluripotent stem cell (iPSC) derived from a cell selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell. In some embodiments, a stem cell is an induced pluripotent stem cell (iPSC) derived from a PBMC. In some embodiments, differentiated cells are derived from a multipotent stem cell. In some embodiments, a multipotent stem cell is derived from a cell obtained from the human patient with T1D. In some embodiments, differentiated cells are derived from a lineage- specific stem cell. In some embodiments, a lineage- specific stem cell is derived from a cell obtained from the human patient with T1D. In some embodiments, differentiated cells are derived from a definitive endoderm cell. In some embodiments, a definitive endoderm cell expresses FOXA2 and/or SOX 17. In some embodiments, differentiated cells are derived from a primitive gut tube cell. In some embodiments, a primitive gut tube cell expresses FOXA2 and/or SOX 17. In some embodiments, differentiated cells are derived from a pancreatic progenitor cell. In some embodiments, a pancreatic progenitor cell expresses PDX1 and/or NKX6-1. In some embodiments, differentiated cells are derived from an endocrine cell. In some embodiments, an endocrine cell expresses NKX6-1, NEUROG3, and/or NKX2-2.
[0741] In some embodiments, islet cells are or comprise engineered islet cells. In some embodiments, engineered islet cells have been modified to be hypoimmune islet cells. In some embodiments, hypoimmune islet cells comprise one or more modifications, wherein the modifications (a) inactivate or disrupt one or more alleles of: (i) one or more major histocompatibility complex (MHC) class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or (ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class
II molecules, and/or (b) increase expression of one or more tolerogenic factors, wherein the increased expression of (ii) is relative to an islet cell that does not comprise the modifications. In some embodiments, one or more modifications reduce expression of the one or more MHC class 1 molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or reduce expression of the one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications. In some embodiments, hypoimmune islet cells are (i) HLA class I-deficient and/or HLA class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells comprise one or more safety switches. In some embodiments, a non-human animal of the present disclosure comprises an agent that activates the one or more safety switches.
[0742] In some embodiments, methods of the present disclosure further comprise a step of: (c) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal. In some embodiments, reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises administering an islet-cell destroying composition to the non-human animal. In some embodiments, an islet-cell destroying composition is or comprises STZ. In some embodiments, an islet-cell destroying composition is or comprises splenocytes from an autoimmune and diabetic NOD/ShiLt mouse. In some embodiments, reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises removing the non-human animal’s pancreas. In some embodiments, a non-human animal has reduced endogenous islet cell function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced endogenous islet cell activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell activity relative to
a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced levels of endogenous islet cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal does not comprise endogenous islet cells.
[0743] In some embodiments, a non-human animal has glucose levels greater than about 200mg/dL, greater than about 300mg/dL, greater than about 400mg/dL, or greater than about 450mg/dL. In some embodiments, a non-human animal has glucose levels greater than about 300mg/dL. In some embodiments, a non-human animal has glucose levels greater than about 400mg/dL. In some embodiments, a non-human animal has glucose levels greater than about 450mg/dL.
[0744] In some embodiments, a method of the present disclosure comprises, in sequential order: (a) administering peripheral blood mononuclear cells (PBMCs) from the human patient with T1D to the non-human animal, (c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, and (b) administering the islet cells derived from cells of the human patient with T1D to the non-human animal.
[0745] In some embodiments, method of the present disclosure comprises, in sequential order: (c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (b) administering the islet cells derived from cells of the human patient with T1D to the non-human animal, and (a) administering peripheral blood mononuclear cells (PBMCs) from the human patient with T1D to the non-human animal.
[0746] In some embodiments, method of the present disclosure comprises, in sequential order: (c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (a) administering peripheral blood mononuclear cells (PBMCs) from the human patient with T1D to the non- human animal, and (b) administering islet cells derived from cells of the human patient with T1D to the non-human animal.
[07471 In some embodiments, an islet-cell destroying composition is administered to the non-human animal for 5 continuous days, 4 continuous days, 3 continuous days, 2 continuous days, or 1 continuous day. In some embodiments, an islet-cell destroying composition is administered to the non-human animal 5 days, 4 days, 3 days, 2 days, or 1 day before the PBMCs are administered to the non-human patient. In some embodiments, no more than 1 day, 2 days, 3 days, 4 days, or 5 days elapse between step (a) and step (b).
[0748] In another aspect, the present disclosure provides methods comprising administering peripheral blood mononuclear cells (PBMCs) and islet cells derived from cells of a human patient with type 1 diabetes (T1D) to a non-human animal, wherein the PBMCs and the islet cells are autologous. In some embodiments, the non-human animal comprises PBMCs from a human patient with T1D.
B. Methods of Treatment
[0749] Among other things, provided herein are compositions and methods relating to assessing an autoimmune response of a subject. The present disclosure provides that autologous peripheral blood mononuclear cells (PBMCs) from a subject and islet cells derived from cells of the subject can be transplanted to a non-human animal, where the interaction of such cells with each other and with other agents can be assessed. The ability to evaluate, e.g., islet cells derived from cells of a subject prior to administering such islet cells back to the subject is a powerful tool in the field of cell therapy. The present disclosure, thus, also provides islet cells that can be administered to a subject.
[0750] Provided herein is a method of treating a subject by administering an islet cell or a population islet cells (e.g., hypoimmune islet cells) described herein. In some embodiments, an islet cell or a population islet cells (e.g., hypoimmune islet cells) are formulated for administration in a pharmaceutical composition, such as any described here. Such methods and uses include therapeutic methods and uses, for example, involving administration of an islet cell or a population islet cells (e.g., hypoimmune islet cells), or compositions containing the same, to a subject having a disease, condition, or disorder (e.g., T1D). In some embodiments, an islet
cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same is administered in an effective amount to effect treatment of the disease or disorder (e.g., T1D). Uses include uses of an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same in such methods and treatments, and in the preparation of a medicament in order to carry out such therapeutic methods. In some embodiments, the methods thereby treat the disease or condition or disorder (e.g., T1D) in the subject.
[0751] An islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same provided herein can be administered to any suitable subjects including, for example, a candidate for a cellular therapy for the treatment of a disease or disorder (e.g., T1D). A candidate who benefits from the therapeutic effects of the subject engineered cells provided herein exhibit an elimination, reduction or amelioration of a disease or condition (e.g., T1D).
[0752] In some embodiments, an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same as provided herein, including those produced by any of the methods provided herein, can be used in cell therapy.
[0753] In some embodiments, provided herein is a method of administering an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same to a subject in need thereof. In some embodiments, an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same are administered intravenously or via intramuscular injection. In some embodiments, an islet cell or a population islet cells (e.g., hypoimmune islet cells) overexpress a tolerogenic factor (e.g., CD47), have reduced expression or lack expression of one or more MHC class I molecules. In some embodiments, the engineered cells further comprise overexpression of one or more complement inhibitors.
[0754] In some embodiments, a cellular deficiency is associated with T1D or a cellular therapy is for the treatment of T1D. In some embodiments, a population of engineered cells is a
population of islet cells, including beta islet cells. In some embodiments, the islet cells are selected from the group consisting of an islet progenitor cell, an immature islet cell, and a mature islet cell. In some embodiments, the method comprises administering to the subject a composition comprising a population of engineered beta islet cells, wherein the engineered cells comprise: (i) a multicistronic vector comprising an exogenous polynucleotide encoding CD47, and (ii) inactivation or disruption of both alleles of a B2M gene. In some embodiments, the method comprises administering to the subject a composition comprising a population of engineered beta islet cells, wherein the engineered beta islet cells comprise: (i) a transgene comprising an exogenous polynucleotide encoding CD47. In some embodiments, the engineered beta cells comprise inactivation or disruption of both alleles of a CIITA gene. In some embodiments, the transgene comprising the polynucleotide encoding CD47 is the transgene is a multicistronic vector.
[0755] In some embodiments, a subject undergoing a treatment using an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same received a previous treatment. In some embodiments, an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same are used to treat the same condition (e.g., T1D) as the previous treatment. In some embodiments, an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same administered to the subject exhibit an enhanced therapeutic effect for the treatment of the same condition or disease (e.g., T1D) treated by the previous treatment. In certain embodiments, an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same exhibit a longer therapeutic effect for the treatment of the condition or disease (e.g., T1D) in the subject as compared to the previous treatment.
[0756] The methods provided herein can be used as a second-line treatment for a particular condition or disease (e.g., T1D) after a failed first line treatment. In some embodiments, a previous treatment is a therapeutically ineffective treatment. As used herein, a “therapeutically ineffective” treatment refers to a treatment that produces a less than desired
clinical outcome in a subject. For example, with respect to T ID, a therapeutically ineffective treatment can refer to a treatment that adequately stabilize insulin levels, adequately stabilize glucose levels, prevent or minimize severe hypoglycemic events, and/or prevent or minimize impaired awareness of hypoglycemia.
[0757] In embodiments, the subject does not exhibit an immune response to an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same.
[0758] Methods of treating a subject arc generally through administrations of engineered cells, or a composition containing the same, as provided herein. As will be appreciated, for all the multiple embodiments described herein related to the cells and/or the timing of therapies, the administering of the cells is accomplished by a method or route that results in at least partial localization of the introduced cells at a desired site. The cells can be implanted directly to the desired site, or alternatively be administered by any appropriate route which results in delivery to a desired location in the subject where at least a portion of the implanted cells or components of the cells remain viable.
[0759] In some embodiments, the recovery period begins following the first administration of the population of engineered cells or a composition containing the same, and ends when such cells are no longer present or detectable in the subject. In some embodiments, the duration of the recovery period is at least 1 week (e.g., 1 week, 2 weeks, 3 weeks, 4 weeks, 5 weeks, 6 weeks, 7 weeks, 8 weeks, 9 weeks, 10 weeks, 11 weeks, 12 weeks, 13 weeks, 14 weeks, 15 weeks, 16 weeks, 17 weeks, 18 weeks, 19 weeks, 20 weeks, or more) or more after the initial administration of the cells. In some embodiments, the duration of the recovery period is at least 1 month (e.g., 1 month, 2 months, 3 months, 4 months, 5 months, 6 months, 7 months, 8 months, 9 months, 10 months, 11 months, 12 months, 13 months, 14 months, 15 months, 16 months, 17 months, 18 months, 19 months, 20 months, or more) or more after the initial administration of the cells.
[07601 In some embodiments, the administered islet cell, population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same is hypoimmunogenic when administered to the subject. In some embodiments, islet cells are hypoimmune. In some embodiments, an immune response against an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same is reduced or lower by at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91 %, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% lower compared to the level of the immune response produced by the administration of immunogenic cells (e.g., a population of comparable cells but that do not contain the modifications, e.g., genetic modifications, of the engineered cells). In some embodiments, an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same fails to elicit an immune response against the engineered cells in the subject.
[0761] In some embodiments, the administered islet cell, population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same elicits a decreased or lower level of systemic TH1 activation in a subject. In some instances, the level of systemic TH1 activation elicited by the islet cells is at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% lower compared to the level of systemic TH1 activation produced by the administration of immunogenic cells (e.g., a population of comparable cells but that do not contain the modifications, e.g., genetic modifications, of the engineered cells). In some embodiments, the administered islet cell, population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same fails to elicit systemic TH1 activation in the subject.
[0762] In some embodiments, the administered islet cell, population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same elicits a decreased or lower level of immune activation of peripheral blood mononuclear cells (PBMCs) in the subject. In some instances, a level of immune activation of PBMCs elicited by the cells is at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%,
90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% lower compared to the level of immune activation of PBMCs produced by the administration of immunogenic cells (e.g., a population of cells of the same or similar cell type or phenotype but that do not contain the modifications, e.g., genetic modifications, of the engineered cells). In some embodiments, the administered islet cell, population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same fails to elicit immune activation of PBMCs in the subject.
[0763] In some embodiments, the administered islet cell, population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same elicits a decreased or lower level of donor- specific IgG antibodies in the subject. In some instances, a level of donor- specific IgG antibodies elicited by the cells is at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% lower compared to the level of donor-specific IgG antibodies produced by the administration of immunogenic cells (e.g., a population of cells of the same or similar cell type or phenotype but that do not contain the modifications, e.g., genetic modifications, of the engineered cells). In some embodiments, the administered islet cell, population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same fails to elicit donor- specific IgG antibodies in the subject.
[0764] In some embodiments, the administered population of engineered cells, or a composition containing the same, elicits a decreased or lower level of IgM and IgG antibody production in the subject. In some instances, the level of IgM and IgG antibody production elicited by the cells is at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% lower compared to the level of IgM and IgG antibody production produced by the administration of immunogenic cells (e.g., a population of cells of the same or similar’ cell type or phenotype but that do not contain the modifications, e.g., genetic modifications, of the engineered cells). In some embodiments, the administered population of engineered cells, or a composition containing the same, fails to elicit IgM and IgG antibody production in the subject.
[07651 In some embodiments, an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same elicits a decreased or lower level of cytotoxic T cell killing in the subject. In some instances, the level of cytotoxic T cell killing elicited by the cells is at least 5%, 10%, 15%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 91%, 92%, 93%, 94%, 95%, 96%, 97%, 98%, or 99% lower compared to the level of cytotoxic T cell killing produced by the administration of immunogenic cells (e.g., a population of cells of the same or similar cell type or phenotype but that do not contain the modifications, e.g., genetic modifications, of the engineered cells). In some embodiments, an islet cell, a population islet cells (e.g., hypoimmune islet cells), or pharmaceutical composition comprising the same fails to elicit cytotoxic T cell killing in the subject.
[0766] Dose and Dosage Regimen
[0767] Any therapeutically effective amount of cells described herein can be included in a pharmaceutical composition. Non-limiting examples of the cells include cells differentiated from engineered induced pluripotent stem cells as described (e.g., beta islet cells differentiated from iPSCs). In preferred embodiments, cells are islet cells (e.g., beta islet cells).
[0768] In some embodiments, a pharmaceutical composition includes at least about 1 x 102, 5 x 102, 1 x 103, 5 x 103, 1 x 104, 5 x 104, 1 x 105, 5 x 105, 1 x 106, 5 x 106, 1 x 107, 5 x 107, 1 x 108, 5 x 108, 1 x 109, 5 x 109, 1 x 1010, or 5 x 1010 cells. In some embodiments, a pharmaceutical composition includes up to about 1 x 102, 5 x 102, 1 x 103, 5 x 103, 1 x 104, 5 x
104, 1 x 105, 5 x 105, 1 x 106, 5 x 106, 1 x 107, 5 x 107, 1 x 108, 5 x 108, 1 x 109, 5 x 109, 1 x IO10, or 5 x IO10 cells. In some embodiments, a pharmaceutical composition includes up to about 6.0 x 108 cells. In some embodiments, a pharmaceutical composition includes up to about 8.0 x 108 cells. In some embodiments, a pharmaceutical composition includes at least about 1 x 102-5 x 102, 5 x 102- 1 x 103, 1 x 103-5 x 103, 5 x 103- 1 x 104, 1 x 104-5 x 104, 5 x 104- 1 x 105, 1 x 105-5 x
105, 5 x 105-l x 106, 1 x 106-5 x 106, 5 x 106-l x 107, 1 x 107-5 x 107, 5 x 107- 1 x 108, 1 x 108-5 x 108, 5 x 108-l x 109, 1 x 109-5 x 109, 5 x 109- 1 x IO10, or 1 x 1010-5 x IO10 cells. In exemplary
embodiments, a pharmaceutical composition includes from about 1.0 x 106 to about 2.5 x 108 cells.
[0769] In some embodiments, a pharmaceutical composition has a volume of at least 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 110, 120, 130, 140, 150, 160, 170, 180, 190, 200, 250, 300, 350, 400, or 500 ml. In exemplary embodiments, a pharmaceutical composition has a volume of up to about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 110, 120, 130, 140, 150, 160, 170, 180, 190, 200, 250, 300, 350, 400, or 500 ml. In exemplary embodiments, a pharmaceutical composition has a volume of about 5, 10, 15, 20, 25, 30, 35, 40, 45, 50, 55, 60, 65, 70, 75, 80, 85, 90, 95, 100, 110, 120, 130, 140, 150, 160, 170, 180, 190, 200, 250, 300, 350, 400, or 500 ml. In some embodiments, a pharmaceutical composition has a volume of from about 1-50 ml, 50-100 ml, 100-150 ml, 150- 200 ml, 200-250 ml, 250-300 ml, 300-350 ml, 350-400 ml, 400-450 ml, or 450-500 ml. In some embodiments, a pharmaceutical composition has a volume of from about 1-50 ml, 50-100 ml, 100-150 ml, 150-200 ml, 200-250 ml, 250-300 ml, 300-350 ml, 350-400 ml, 400-450 ml, or 450- 500 ml. In some embodiments, a pharmaceutical composition has a volume of from about 1-10 ml, 10-20 ml, 20-30 ml, 30-40 ml, 40-50 ml, 50-60 ml, 60-70 ml, 70-80 ml, 70-80 ml, 80-90 ml, or 90-100 ml. In some embodiments, a pharmaceutical composition has a volume that ranges from about 5 ml to about 80 ml. In exemplary embodiments, a pharmaceutical composition has a volume that ranges from about 10 ml to about 70 ml. In many embodiments, a pharmaceutical composition has a volume that ranges from about 10 ml to about 50 ml.
[0770] The specific amount/dosage regimen can vary depending on multiple factors including weight, gender, age and health of the individual; the formulation, the biochemical nature, bioactivity, bioavailability and the side effects of the cells and the number and identity of the cells in the complete therapeutic regimen.
[0771] In some embodiments, a dose of the pharmaceutical composition includes about 1.0 x 105 to about 2.5 x 108 cells at a volume of about 10 mL to 50 mL and the pharmaceutical composition is administered as a single dose.
[07721 In some embodiments, a pharmaceutical composition is administered as a single dose of from about 1.0 x 105 to about 1.0 x 107 engineered cells per kg body weight for subjects 50 kg or less. In some embodiments, a pharmaceutical composition is administered as a single dose of from about 0.5 x 105 to about 1.0 x 107, about 1.0 x 105 to about 1.0 x 107, about 1.0 x 105 to about 1.0 x 107, about 5.0 x 105 to about 1 x 107, about 1.0 x 106 to about 1 x 107, about 5.0 x 106 to about 1.0 x 107, about 1.0 x 105 to about 5.0 x 106, about 1.0 x 105 to about 1.0 x 106, about 1 .0 x 105 to about 5.0 x 105, about 1 .0 x 105 to about 5.0 x 106, about 2.0 x 105 to about 5.0 x 106, about 3.0 x 105 to about 5.0 x 106, about 4.0 x 105 to about 5.0 x 106, about 5.0 x 105 to about 5.0 x 106, about 6.0 x 105 to about 5.0 x 106, about 7.0 x 105 to about 5.0 x 106, about 8.0 x
105 to about 5.0 x 106, or about 9.0 x 105 to about 5.0 x 106 cells per kg body weight for subjects 50 kg or less. In some embodiments, a dose is from about 0.2 x 106 to about 5.0 x 106 cells per kg body weight for subjects 50 kg or less. In many embodiments, a dose is at a range that is lower than from about 0.2 x 106 to about 5.0 x 106 cells per kg body weight for subjects 50 kg or less. In many embodiments, a dose is at a range that is higher than from about 0.2 x 106 to about 5.0 x 106 cells per kg body weight for subjects 50 kg or less. In exemplary embodiments, a single dose is at a volume of about 10 ml to 50 ml. In some embodiments, a dose is administered intravenously.
[0773] In exemplary embodiments, cells are administered in a single dose of from about 1.0 x 106 to about 5.0 x 108 cells for subjects above 50 kg. In some embodiments, a pharmaceutical composition is administered as a single dose of from about 0.5 x 106 to about 1.0 x 109, about 1.0 x 106 to about 1.0 x 109, about 1.0 x 106 to about 1.0 x 109, about 5.0 x 106 to about 1.0 x 109, about 1.0 x 107 to about 1.0 x 109, about 5.0 x 107 to about 1.0 x 109, about 1.0 x
106 to about 5.0 x 107, about 1.0 x 106 to about 1.0 x 107, about 1.0 x 106 to about 5.0 x 107, about 1.0 x 107 to about 5.0 x 108, about 2.0 x 107 to about 5.0 x 108, about 3.0 x 107 to about 5.0 x 108, about 4.0 x 107 to about 5.0 x 108, about 5.0 x 107 to about 5.0 x 108, about 6.0 x 107 to about 5.0 x 108, about 7.0 x 107 to about 5.0 x 108, about 8.0 x 107 to about 5.0 x 108, or about 9.0 x 107 to about 5.0 x 108 cells per kg body weight for subjects 50 kg or less. In some embodiments, cells arc administered in a single dose of about 1.0 x 107 to about 2.5 x 108 cells
for subjects above 50 kg. In some embodiments, cells are administered in a single dose of a range that is less than about 1.0 x 107 to about 2.5 x 108 cells for subjects above 50 kg. In some embodiments, cells are administered in a single dose of a range that is higher than about 1.0 x 107 to about 2.5 x 108 cells for subjects above 50 kg. In some embodiments, a dose is administered intravenously. In exemplary embodiments, a single dose is at a volume of about 10 ml to 50 ml. In some embodiments, a dose is administered intravenously.
[0774] In exemplary embodiments, a dose is administered intravenously at a rate of about 1 to 50 ml per minute, 1 to 40 ml per minute, 1 to 30 ml per minute, 1 to 20 ml per minute, 10 to 20 ml per minute, 10 to 30 ml per minute, 10 to 40 ml per minute, 10 to 50 ml per minute, 20 to 50 ml per minute, 30 to 50 ml per minute, 40 to 50 ml per minute. In some embodiments, a pharmaceutical composition is stored in one or more infusion bags for intravenous administration. In some embodiments, a dose is administered completely at no more than 10 minutes, 15 minutes, 20 minutes, 25 minutes, 30 minutes, 35 minutes, 40 minutes, 45 minutes, 50 minutes, 55 minutes, 60 minutes, 70 minutes, 80 minutes, 90 minutes, 120 minutes, 150 minutes, 180 minutes, 240 minutes, or 300 minutes.
[0775] In some embodiments, a single dose of a pharmaceutical composition is present in a single infusion bag. In other embodiments, a single dose of a pharmaceutical composition is divided into 2, 3, 4 or 5 separate infusion bags.
[0776] In some embodiments, cells described herein are administered in a plurality of doses such as 2, 3, 4, 5, 6 or more doses. In some embodiments, each dose of the plurality of doses is administered to the subject ranging from 1 to 24 hours apart. In some instances, a subsequent dose is administered from about 1 hour to about 24 hours (e.g., about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23 or about 24 hours) after an initial or preceding dose. In some embodiments, each dose of the plurality of doses is administered to the subject ranging from about 1 day to 28 days apart. In some instances, a subsequent dose is administered from about 1 day to about 28 days (e.g., about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, or about 28 days) after an initial or preceding dose. In many embodiments, each dose of the plurality of doses is administered to the
subject ranging from 1 week to about 6 weeks apart. In certain instances, a subsequent dose is administered from about 1 week to about 6 weeks (e.g., about 1, 2, 3, 4, 5, or 6 weeks) after an initial or preceding dose. In several embodiments, each dose of the plurality of doses is administered to the subject ranging from about 1 month to about 12 months apart. In several instances, a subsequent dose is administered from about 1 month to about 12 months (e.g., about 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 months) after an initial or preceding dose.
[0777] In some embodiments, a subject is administered a first dosage regimen at a first timepoint, and then subsequently administered a second dosage regimen at a second timepoint. In some embodiments, the first dosage regimen is the same as the second dosage regimen. In other embodiments, the first dosage regimen is different than the second dosage regimen. In some instances, the number of cells in the first dosage regimen and the second dosage regimen are the same. In some instances, the number of cells in the first dosage regimen and the second dosage regimen are different. In some cases, the number of doses of the first dosage regimen and the second dosage regimen are the same. In some cases, the number of doses of the first dosage regimen and the second dosage regimen are different.
[0778] In another aspect, the present disclosure provides methods of treating a human patient, comprising administering a pharmaceutical composition comprising a T1D therapeutic made in a method of the present disclosure. In some embodiments, a human patient has been diagnosed as having type 1 diabetes (T1D). In some embodiments, a human patient is eligible for islet transplant therapy. In some embodiments, a human patient exhibits one or more of the following: (a) a clinical history compatible with T1D onset of disease at younger than 40 years old; (b) insulin dependence for at least 5 years; (c) a sum of age and years of insulin dependent diabetes duration of at least 28 years; (d) absent stimulated C-peptide (less than 0.3 ng/mL) 60 and 90 minutes post-mixed-meal tolerance test; (e) involvement of intensive diabetes management, wherein intensive diabetes management comprises: (i) self-monitoring of glucose values no less than a mean of three times each day averaged over each week; (ii) administration of three or more insulin injections each day or insulin pump therapy; and/or (iii) under the direction of an endocrinologist, diabetologist, or diabetes specialist with at least three clinical
evaluations during the past 12 months; (f) at least one episode of severe hypoglycemia in the past 12 months, wherein an episode of severe hypoglycemia comprises an event with one or more of the following symptoms: (i) memory loss; (ii) confusion; (iii) uncontrollable behavior; (iv) irrational behavior; (v) unusual difficulty in awakening; (vi) suspected seizure; (vii) seizure;
(viii) loss of consciousness; (ix) visual symptoms, and hypoglycemia during the past 12 months; and/or (g) impaired awareness of hypoglycemia (IAH). In some embodiments, a human patient has had T1D for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 years, at least 8 years, at least 9 years, at least 10 years, at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 years, or at least 90 years. In some embodiments, a human patient has impaired awareness of hypoglycemia (IAH). In some embodiments, IAH comprises: a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more; a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more; marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more; or a composite of a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile.
[0779] In some embodiments, a human patient has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years.
[0780] In some embodiments, a human patient has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2). In some embodiments, antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU, insulin autoantibodies are present at a level
greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
[0781] In some embodiments, a human patient has glucose levels greater than about 200 mg/dL, greater than about 300 mg/dL, greater than about 400 mg/dL, or greater than about 450 mg/dL. In some embodiments, a human patient has glucose levels greater than about 300 mg/dL. In some embodiments, a human patient has glucose levels greater than about 400 mg/dL. In some embodiments, a human patient has glucose levels greater than about 450 mg/dL.
[0782] In another aspect, the present disclosure provides methods of monitoring a human patient treated with a pharmaceutical composition comprising a T1D therapeutic made in any one of the methods of the present disclosure, comprising assessing the effect of the T1D therapeutic on glucose levels.
[0783] In some embodiments, an immunosuppressive and/or immunomodulatory agent is not administered to a subject before administration of an islet cell, a population islet cells (e.g., hypoimmune islet cell ), or pharmaceutical composition comprising the same.
C. Methods of Assessing and/or Characterizing
1. Methods of Assessing and/or Characterizing Autoimmune Response of a Subject
[0784] Among other things, provided herein are compositions and methods relating to assessing an autoimmune response of a subject. The present disclosure provides that autologous peripheral blood mononuclear cells (PBMCs) from a subject and islet cells derived from cells of the subject can be transplanted to a non-human animal, where the interaction of such cells with each other and with other agents can be assessed. The ability to evaluate, e.g., islet cells derived from cells of a subject prior to administering such islet cells back to the subject is a powerful tool in the field of cell therapy. The present disclosure, thus, also provides islet cells that can be administered to a subject.
2. Methods of Characterizing Human Islet Cells for Delivery to a Patient
[07851 In another aspect, the present disclosure provides methods of characterizing human islet cells for delivery to a patient, comprising taking a sample of the islet cells derived from cells of the human patient with T1D and administering the sample to a non-human animal of the present disclosure.
[0786] In another aspect, the present disclosure provides methods of using a non-human animal model of type 1 diabetes (T1D), the method comprising: (a) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to the non-human animal, (b) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous and (c) determining the survival and/or endocrine function of the islet cells. In some embodiments, islet cells are or comprise engineered islet cells. In some embodiments, engineered islet cells have been modified to be hypoimmune islet cells. In some embodiments, hypoimmune islet cells comprise one or more modifications, wherein the modifications (a) inactivate or disrupt one or more alleles of: (i) one or more MHC class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or (ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules, and/or (b) increase expression of one or more tolerogenic factors, wherein the increased expression of (ii) is relative to an islet cell that does not comprise the modifications. In some embodiments, one or more modifications reduce expression of the one or more MHC class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or reduce expression of the one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications. In some embodiments, hypoimmune islet cells are (i) HLA class I-deficient and/or HLA class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47. In some embodiments, hypoimmune islet cells comprise one or more safety switches. In some embodiments, islet cells are a therapeutic agent. In some embodiments, step (c) comprises determining the survival and/or endocrine function of the islet cells after an autoimmune
challenge. In some embodiments, a method of the present disclosure further comprises: (d) administering an agent to the non-human animal. In some embodiments, an agent activates the one or more safety switches. In some embodiments, step (c) further comprises evaluating effects of the one or more modifications on the hypoimmune islet cells. In some embodiments, an agent is a therapeutic agent. In some embodiments, a method of the present disclosure further comprises: (e) administering an immunosuppressant to the non-human animal.
3. Methods of Characterizing Therapeutic Agents
[0787] In another aspect, the present disclosure provides methods of characterizing one or more therapeutic agents, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (b) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, and wherein the islet cells are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the one or more therapeutic agents on one or more therapeutically relevant T1D measures.
[0788] In another aspect, the present disclosure provides methods of characterizing one or more therapeutic agents, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (c) administering islet cells derived from cells of a human patient with T1D to the non-human animal, wherein the islet cells are a first therapeutic agent, (b) administering peripheral blood mononuclear cells (PBMCs) from the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, (d) optionally administering a second therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the one or more therapeutic agent on one or more therapeutically relevant T1D measures.
[07891 In another aspect, the present disclosure provides methods of characterizing one or more therapeutic agents, the method comprising, in sequential order: (b) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, and wherein the islet cells are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the one or more therapeutic agent on one or more therapeutically relevant T1D measures.
[0790] In another aspect, the present disclosure provides methods of determining if one or more therapeutic agents are suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, and wherein the islet cells derived from the human patient with T1D are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non-human animal, and (e) determining the suitability of the one or more therapeutic agents to make a T1D therapeutic by assessing the effect of the one or more therapeutic agents on one or more therapeutically relevant T1D measures.
[0791] In another aspect, the present disclosure provides methods of determining if one or more therapeutic agents are suitable for use in treating type 1 diabetes (T1D), the method comprising, in sequential order: (b) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with
T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, and wherein the islet cells derived from the human patient with T1D are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non-human animal, and (e) determining the suitability of the one or more therapeutic agents to treat T1D by assessing the effect of the one or more therapeutic agents on one or more therapeutically relevant T1D measures.
[0792] In another aspect, the present disclosure provides methods of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (b) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, (d) administering a therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
[0793] In another aspect, the present disclosure provides methods of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal, (c) administering islet cells derived from cells of a human patient with T1D to the non-human animal, (b) administering peripheral blood mononuclear cells (PBMCs) from the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, (d) administering a therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
[0794] In another aspect, the present disclosure provides methods of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous
islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, (d) administering a therapeutic agent to the non-human animal, and (e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
[0795] In another aspect, the present disclosure provides methods of determining if a therapeutic agent is suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, (d) administering the therapeutic agent to the non-human animal, and (e) determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures.
[0796] In another aspect, the present disclosure provides methods of determining if a therapeutic agent is suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, (d) administering the therapeutic agent to the non-human animal, and (e) determining the suitability of the therapeutic agent to treat T1D by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures.
D. Methods of Making a Therapeutic
[07971 In another aspect, the present disclosure provides methods of making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, and wherein the islet cells derived from the human patient with T1D are a first therapeutic agent, (d) optionally administering a second therapeutic agent to the non-human animal, (e) determining the suitability of the first therapeutic agent, and optionally the second therapeutic agent, to make a T1D therapeutic by assessing the effect of the first therapeutic agent, and optionally the second therapeutic agent, on one or more therapeutically relevant T1D measures, and (f) making the T ID therapeutic.
[0798] In another aspect, the present disclosure provides methods of making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order: (b) administering peripheral blood mononuclear cells (PBMCs) from a human patient with T1D to a non-human animal, (a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, (c) administering islet cells derived from cells of the human patient with T1D to the non-human animal, wherein the PBMCs and the islet cells are autologous, (d) administering a therapeutic agent to the non-human animal, (e) determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures, and (f) making the T1D therapeutic.
[0799] In some embodiments, one or more therapeutically relevant T1D measures comprise the survival and/or endocrine function of the islet cells. In some embodiments, one or more therapeutically relevant T1D measures comprise levels of insulin, glucagon, somatostatin, pancreatic polypeptide, C-peptide, and/or glucose in the non-human animal.
[0800] In some embodiments, if a therapeutic agent is characterized as a suitable T1D therapeutic, the method further comprises a step of making a T1D therapeutic. In some
embodiments, if a therapeutic agent is determined to be a suitable T1D therapeutic, the method further comprises a step of making a T1D therapeutic. In some embodiments, a suitable T1D therapeutic comprises one or more of the following effects of the therapeutic agent on one or more therapeutically relevant T1D measures: (a) C-peptide levels greater than at least 100 pmol/L, at least 200 pmol/L, at least 300 pmol/L, at least 400 pmol/L, at least 500 pmol/L, at least 1000 pmol/L, at least 1500 pmol/L, or at least 1750 pmol/L, optionally wherein C-peptide levels are determined using 60 and 90 minutes post-mixed-meal tolerance test; (b) glucose levels lower than about 200mg/dL, lower than about 300mg/dL, lower than about 400mg/dL, or lower than about 450mg/dL; and/or (c) islet cells survive for at least 5 days, at least 10 days, at least 15 days, at least 20 days, at least 25 days, or at least 30 days.
[0801] In some embodiments, reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal comprises administering an islet-cell destroying composition to the non-human animal. In some embodiments, an islet-cell destroying composition is or comprises STZ. In some embodiments, an islet-cell destroying composition is or comprises splenocytes from an autoimmune and diabetic NOD/ShiLt mouse. In some embodiments, reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal comprises removing the non-human animal’s pancreas. In some embodiments, a non-human animal has reduced endogenous islet cell function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell function relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced endogenous islet cell activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has eliminated endogenous islet cell activity relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal has reduced levels of endogenous islet cells relative to a comparable wild-type non-human animal. In some embodiments, a non-human animal does not comprise endogenous islet cells.
[08021 In some embodiments, a second therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid. In some embodiments, a therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid.
[0803] In some embodiments, a step of determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of islet cells and/or one or more therapeutic agents on one or more therapeutically relevant T1D measures comprises the use of two or more assays to characterize the islet cells and/or the one or more therapeutic agents and/or to determine the suitability of islet cells and/or the one or more therapeutic agents for making a T1D therapeutic.
IV. Populations of Engineered Cells and Pharmaceutical Compositions
[0804] As described elsewhere herein, in the process of manufacturing a cell therapy, certain modifications may be introduced to the cell. Provided herein are populations of cells containing a plurality of the provided engineered cells. Provided herein are also populations of cells containing a plurality of engineered cells.
[0805] In the ‘end’ population of cells, i.e., the population of cells being used in the cell therapy product (pharmaceutical composition), unedited or partially edited cells may be viewed as contaminants likely to adversely affect the function of the cell therapy product. In some circumstances, the percentage of edited cells may be viewed as a measure of purity with respect to the proportion of edited versus unedited or partially edited cells in a population. This is particularly in relation to hypoimmune gene modifications that enable immune evasion where the presence of cells not having the hypoimmune gene modifications might be expected to adversely affect the in vivo efficacy of the cell therapy product. Surprisingly, engineered cells within a population of cells may be functional even if the population of cells contains unedited or partially edited cells. Accordingly, a novel cell therapy product comprising a population of cells
is hereby provided, wherein the population of cells contains some unedited or partially edited cells.
[0806] In some embodiments, up to 20%, up to 30%, up to 40%, up to 50%, up to 60%, or up to 65% of cells in the population are not HIP modified cells (e.g., do not exhibit reduced expression of one or more molecules of the MHC class I and/or MHC class II molecules and optionally also do not exhibit increased expression of at least one tolerogenic factor).
[0807] In some embodiments, up to 20%, up to 30%, up to 40%, up to 50%, up to 60%, or up to 65% of cells in a cell therapy product do not exhibit reduced expression of one or more molecules of the MHC class I and/or MHC class II molecules. In some embodiments, up to 20%, up to 30%, up to 40%, up to 50%, up to 60%, or up to 65% of cells in a cell therapy product (i) do not exhibit reduced expression of one or more molecules of the MHC class I and/or MHC class II molecules and (ii) do not exhibit increased expression of at least one tolerogenic factor. In some embodiments, at least 10%, 20%, 30%, 40%, 50%, 60%, or 70% of cells in the cell therapy product do not exhibit reduced expression of one or more molecules of the MHC class I and/or MHC class II molecules. In some embodiments, at least 10%, 20%, 30%, 40%, 50%, 60%, or 70% of cells in the cell therapy product (i) do not exhibit reduced expression of one or more molecules of the MHC class I and/or MHC class II molecules and (ii) do not exhibit increased expression of at least one tolerogenic factor.
[0808] In some embodiments, at least 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99%, 99.9%, or 99.99% of cells in a population comprise a set of modifications described herein. In some embodiments, the set of modifications reduce expression of one or more molecules of the MHC class I and/or MHC class II molecules and increase expression of at least one tolerogenic factor, such as tolerogenic factors described herein.
[0809] In some embodiments at least 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99%, 99.9%, or 99.99% of cells in the population comprise a set of modifications that reduce expression of one or more MHC class I molecules and/or one or more MHC class II molecules, and that increase expression of one or more tolerogenic factor. In some
embodiments, the one or more tolerogenic factor is one or more of CD 16, CD24, CD35, CD39, CD46, CD47, CD52, CD55, CD59, CD200, CCL22, CTLA4-Ig, Cl inhibitor, FASL, IDO1, HLA-C, HLA-E, HLA-E heavy chain, HLA-G, IL-10, IL-35, PD-L1, SERPINB9, CCL21, MFGE8, DUX4, B2M-HLA-E, CD27, IL-39, CD16 Fc Receptor, 1L15-RF, H2-M3 (HLA-G), A20/TNFAIP3, CR1, HLA-F, MANF, or any combination thereof. In some embodiments, the one or more tolerogenic factor is CD47. In some embodiments, at least 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99%, 99.9%, or 99.99% of cells in the population comprise an exogenous polynucleotide encoding CD47.
[0810] In some embodiments, at least 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99%, 99.9%, or 99.99% of cells in the population comprise one or more alterations that inactivate both alleles of a B2M gene. In some embodiments, at least 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99%, 99.9%, or 99.99% of cells in the population comprise one or more alterations that inactivate both alleles of a CIITA gene.
[0811] In some embodiments, the proportion of cells in the population that comprise the set of modifications as described herein is 30-90%, 30-80%, 30-70%, 30-60%, 30-50% or 40- 50%. In some embodiments, the proportion of cells in the population that comprise the set of modifications as described herein is 40-90%, 40-80%, 40-70%, 40-60% or 40-50%. In some embodiments, the proportion of cells in the population that comprise the set of modifications as described herein is 50-90%, 50-80%, 50-70% or 50-60%.
[0812] In some embodiments, 30-90%, 30-80%, 30-70%, 30-60%, 30-50% or 40-50% of cells in the cell therapy product are HIP modified cells (e.g., exhibiting reduced expression of one or more molecules of the MHC class I and/or MHC class II molecules and optionally increased expression of at least one tolerogenic factor). In some embodiments, 40-90%, 40-80%, 40-70%, 40-60% or 40-50% of cells in the cell therapy product are HIP modified cells (e.g., exhibiting reduced expression of one or more molecules of the MHC class I and/or MHC class II molecules and optionally increased expression of at least one tolerogenic factor). In some embodiments, 50-90%, 50-80%, 50-70% or 50-60% of cells in the cell therapy product are HIP modified cells (e.g., exhibiting reduced expression of one or more molecules of the MHC class I
and/or MHC class II molecules and optionally increased expression of at least one tolerogenic factor).
[0813] Also provided herein are compositions comprising the engineered cells or populations of engineered cells. In some embodiments, the compositions are pharmaceutical compositions.
[0814] In some embodiments, the pharmaceutical composition provided herein further include a pharmaceutically acceptable excipient or carrier. Acceptable carriers, excipients, or stabilizers are nontoxic to recipients at the dosages and concentrations employed, and include buffers such as phosphate, citrate, and other organic acids; antioxidants including ascorbic acid and methionine; preservatives (such as octadecyldimethylbenzyl ammonium chloride; hexamethonium chloride; benzalkonium chloride, benzethonium chloride; phenol, butyl or benzyl alcohol; alkyl parabens such as methyl or propyl paraben; catechol; resorcinol; cyclohexanol; 3-pentanol; and m-cresol); low molecular’ weight (less than about 10 residues) polypeptides; proteins, such as serum albumin, gelatin, or immunoglobulins; hydrophilic polymers such as polyvinylpyrrolidone; amino acids such as glycine, glutamine, asparagine, histidine, arginine, or lysine; monosaccharides, disaccharides, and other carbohydrates including glucose, mannose, or dextrins; chelating agents such as EDTA; sugars such as sucrose, mannitol, trehalose or sorbitol; salt-forming counter-ions such as sodium; metal complexes (e.g., Zn- protein complexes); and/or non-ionic surfactants such as polysorbates (TWEEN™), poloxamers (PLURONICS™) or polyethylene glycol (PEG). In some embodiments, the pharmaceutical composition includes a pharmaceutically acceptable buffer (e.g., neutral buffer saline or phosphate buffered saline). In some embodiments, the pharmaceutical composition can contain one or more excipients for modifying, maintaining or preserving, for example, the pH, osmolarity, viscosity, clarity, color, isotonicity, odor, sterility, stability, rate of dissolution or release, adsorption, or penetration of the composition. In some aspects, a skilled artisan understands that a pharmaceutical composition containing cells may differ from a pharmaceutical composition containing a protein.
[08151 The term “pharmaceutical formulation” refers to a preparation which is in such form as to permit the biological activity of an active ingredient contained therein to be effective, and which contains no additional components which are unacceptably toxic to a subject to which the formulation would be administered.
[0816] A “pharmaceutically acceptable carrier” refers to an ingredient in a pharmaceutical formulation, other than an active ingredient, which is nontoxic to a subject. A pharmaceutically acceptable carrier includes, but is not limited to, a buffer, excipient, stabilizer, or preservative.
[0817] The pharmaceutical composition in some embodiments contains engineered cells as described herein in amounts effective to treat or prevent the disease or condition, such as a therapeutically effective or prophylactic ally effective amount. In some embodiments, the pharmaceutical composition contains engineered cells as described herein in amounts effective to treat or prevent the disease or condition, such as a therapeutically effective or prophylactically effective amount. Therapeutic or prophylactic efficacy in some embodiments is monitored by periodic assessment of treated subjects. For repeated administrations over several days or longer, depending on the condition, the treatment is repeated until a desired suppression of disease symptoms occurs. However, other dosage regimens may be useful and can be determined. The desired dosage can be delivered by a single bolus administration of the composition, by multiple bolus administrations of the composition, or by continuous infusion administration of the composition.
[0818] In some embodiments, engineered cells as described herein are administered using standard administration techniques, formulations, and/or devices. In some embodiments, engineered cells as described herein are administered using standard administration techniques, formulations, and/or devices. Provided are formulations and devices, such as syringes and vials, for storage and administration of the compositions. Engineered cells can be administered via localized injection, including catheter administration, systemic injection, localized injection, intravenous injection, or parenteral administration. When administering a therapeutic
composition (e.g., a pharmaceutical composition containing an engineered cell), it will generally be formulated in a unit dosage injectable form (solution, suspension, emulsion).
[0819] Formulations include those for intravenous, intraperitoneal, or subcutaneous, administration. In some embodiments, the cell populations are administered parenterally. The term “parenteral,” as used herein, includes intravenous, intramuscular, subcutaneous, rectal, vaginal, and intraperitoneal administration. In some embodiments, the cell populations are administered to a subject using peripheral systemic delivery by intravenous, intraperitoneal, or subcutaneous injection.
[0820] Compositions in some embodiments are provided as sterile liquid preparations, e.g., isotonic aqueous solutions, suspensions, emulsions, or dispersions, which may in some aspects be buffered to a selected pH. Liquid compositions are somewhat more convenient to administer, especially by injection. Liquid compositions can comprise carriers, which can be a solvent or dispersing medium containing, for example, water, saline, phosphate buffered saline, polyol (for example, glycerol, propylene glycol, liquid polyethylene glycol) and suitable mixtures thereof. Sterile injectable solutions can be prepared by incorporating the cells in a solvent, such as in admixture with a suitable carrier, diluent, or excipient such as sterile water, physiological saline, glucose, dextrose, or the like.
[0821] In some embodiments, a pharmaceutically acceptable carrier can include all solvents, dispersion media, coatings, antibacterial and antifungal agents, isotonic and absorption delaying agents, and the like, compatible with pharmaceutical administration (Gennaro, 2000, Remington: The science and practice of pharmacy, Lippincott, Williams & Wilkins, Philadelphia, PA). Examples of such carriers or diluents include, but are not limited to, water, saline, Ringer's solutions, dextrose solution, and 5% human serum albumin. Liposomes and nonaqueous vehicles such as fixed oils may also be used. Supplementary active compounds can also be incorporated into the compositions. The pharmaceutical carrier should be one that is suitable for the engineered cells, such as a saline solution, a dextrose solution or a solution comprising human serum albumin. In some embodiments, the pharmaceutically acceptable carrier or vehicle for such compositions is any non-toxic aqueous solution in which the engineered cells can be
maintained, or remain viable, for a time sufficient to allow administration of live cells. For example, the pharmaceutically acceptable carrier or vehicle can be a saline solution or buffered saline solution.
[0822] In some embodiments, the composition, including pharmaceutical composition, is sterile. In some embodiments, isolation, enrichment, or culturing of the cells is carried out in a closed or sterile environment, for example and for instance in a sterile culture bag, to minimize error, user handling and/or contamination. In some embodiments, sterility may be readily accomplished, e.g., by filtration through sterile filtration membranes. In some embodiments, culturing is carried out using a gas permeable culture vessel. In some embodiments, culturing is carried out using a bioreactor.
[0823] In some embodiments, the cells and compositions provided herein may be stored. In some embodiments, the cells and compositions provided herein may be stored for 1-72 hours. In some embodiments, the cells and compositions provided herein may be stored for 1-7 days. In some embodiments, the cells and compositions provided herein may be stored for 1-5 weeks. In some embodiments, the cells and compositions provided herein may be stored for 1-12 months. In some embodiments, the cells and compositions provided herein may be stored for 1-30 years.
[0824] In some embodiments, the cells and compositions provided herein may be stored after they have been collected from a donor or pool of donors. In some embodiments, the cells and compositions provided herein may be stored before manufacturing. In some embodiments, the cells and compositions provided herein may be stored after starting manufacturing. In some embodiments, the cells and compositions provided herein may be stored after completing 1, 2, 3, 4, 5, 6, 7, 8, 9, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 steps of the manufacturing process. In some embodiments, the cells and compositions provided herein may be stored after completing 1 or more steps of the manufacturing process. In some embodiments, the cells and compositions provided herein may be stored after completing the manufacturing process. In some embodiments, the cells and compositions provided herein may be stored before modification. In some embodiments, the cells and compositions provided herein may be stored after starting modification. In some embodiments, the cells and compositions provided herein may be stored
after completing 1, 2, 3, 4, 5, 6, 7, 8, 9, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 modifications. In some embodiments, the cells and compositions provided herein may be stored after completing 1 or more modifications. In some embodiments, the cells and compositions provided herein may be stored after completing modification. In some embodiments, the cells and compositions provided herein may be stored before gene-editing. In some embodiments, the cells and compositions provided herein may be stored after stalling gene-editing. In some embodiments, the cells and compositions provided herein may be stored after completing 1 , 2, 3, 4, 5, 6, 7, 8, 9, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 gene-edits. In some embodiments, the cells and compositions provided herein may be stored after completing 1 or more gene-edits. In some embodiments, the cells and compositions provided herein may be stored after completing geneediting. In some embodiments, the cells and compositions provided herein may be stored before viral transduction. In some embodiments, the cells and compositions provided herein may be stored after starting viral transduction. In some embodiments, the cells and compositions provided herein may be stored after completing 1, 2, 3, 4, 5, 6, 7, 8, 9, 11, 12, 13, 14, 15, 16, 17, 18, 19, or 20 viral transductions. In some embodiments, the cells and compositions provided herein may be stored after completing 1 or more viral transductions. In some embodiments, the cells and compositions provided herein may be stored after completing viral transduction.
[0825] In some embodiments, the cells and compositions may be stored in liquid nitrogen. In some embodiments, the cells and compositions may be stored in a freezer at -80 °C. In some embodiments, the cells and compositions may be stored in a freezer at -20 °C. In some embodiments, the cells and compositions may be stored on ice. In some embodiments, the cells and compositions may be stored on dry ice. In some embodiments, the cells and compositions may be stored in refrigerator at 4 °C.
[0826] Also provided herein are compositions that are suitable for cryopreserving the provided engineered cells. In some embodiments, the provided engineered cells are cryopreserved in a cry opreservation medium. In some embodiments, the cry opreservation medium is a serum free cryopreservation medium. In some embodiments, the composition comprises a cryoprotectant. In some embodiments, the cryoprotectant is or comprises DMSO
and/or s glycerol. In some embodiments, the cryopreservation medium is between at or about 5% and at or about 10% DMSO (v/v). In some embodiments, the cryopreservation medium is at or about 5% DMSO (v/v). In some embodiments, the cryopreservation medium is at or about 6% DMSO (v/v). In some embodiments, the cryopreservation medium is at or about 7% DMSO (v/v). In some embodiments, the cry opreservation medium is at or about 7.5% DMSO (v/v). In some embodiments, the cryopreservation medium is at or about 8% DMSO (v/v). In some embodiments, the cryopreservation medium is at or about 9% DMSO (v/v). In some embodiments, the cryopreservation medium is at or about 10% DMSO (v/v). In some embodiments, the cryopreservation medium contains a commercially available cryopreservation solution (CryoStor™ CS10). CryoStor™ CS10 is a cryopreservation medium containing 10% dimethyl sulfoxide (DMSO). In some embodiments, compositions formulated for cryopreservation can be stored at low temperatures, such as ultra-low temperatures, for example, storage with temperature ranges from -40 °C to -150 °C, such as or about 80 °C ± 6.0 ° C.
[0827] It will be understood that following cryo preservation some cells in the population may be dead, such as up to 10%, up to 5%, up to 1%, up to 0.5% or up to 0.1% of cells. Alternatively, cell recovers after cryo preservation may be up to 20% or up to 10%. In some embodiments, the pharmaceutical composition comprises engineered cells described herein and a pharmaceutically acceptable carrier comprising 31.25 % (v/v) Plasma- Lyte A, 31.25 % (v/v) of 5% dextrose/0.45% sodium chloride, 10% dextran 40 (LMD)/5% dextrose, 20% (v/v) of 25% human serum albumin (HSA), and 7.5% (v/v) dimethylsulfoxide (DMSO).
[0828] In some embodiments, the cryoprcscrvcd engineered cells are prepared for administration by thawing. In some cases, the engineered cells can be administered to a subject immediately after thawing. In some such embodiments, the composition is ready-to-use without any further processing. In other cases, the engineered cells are further processed after thawing, such as by resuspension with a pharmaceutically acceptable carrier, incubation with an activating or stimulating agent, or are activated washed and resuspended in a pharmaceutically acceptable buffer prior to administration to a subject.
V. Kits, Components, and Articles of Manufacture
[0829] In some aspects, provided herein are kits, components, and compositions (such as consumables) of the methods, devices, and systems described herein. In some embodiments, a kit comprises instructions for use according to the disclosure herein.
[0830] In some embodiments, provided herein is a kit comprising a population of engineered cells (e.g., islet cells, e.g., hypoimmune islet cells, e.g., human hypoimmune islet cells) described herein. In some embodiments, provided herein is a kit comprising: (a) a population of cells comprising a plurality of engineered cells (e.g., hypoimmune islet cells, e.g., human hypoimmune islet cells), wherein the engineered cells comprise (i) increased expression of a tolerogenic factor (e.g., CD47), and (ii) reduced expression of one or more MHC class I molecules and/or one or more MHC class II molecules (e.g., one or more MHC class I human leukocyte antigens and/or one or more MHC class II human leukocyte antigens), wherein the increased expression of (i) and the reduced expression of (ii) is relative to a cell of the same cell type that does not comprise the modifications.
[0831] In some embodiments, there is provided an article of manufacture containing materials useful for clinical transplantation therapies, including cell therapies. In some embodiments, the articles of manufacture contain material useful for the treatment of cellular deficiencies, such as but not limited to diabetes (e.g., T1D). The article of manufacture can comprise a container and a label or package insert on or associated with the container. Suitable containers include, for example, bottles, vials, syringes, etc. (e.g., glass or plastic containers). Generally, the container holds a composition which is effective for allogenic cell therapy and may have a sterile access port (for example the container may be an intravenous solution bag or a vial having a stopper pierceable by a hypodermic injection needle).
[0832] In some aspects, a kit or article of manufacture provided herein comprises a population of engineered cells (e.g., islet cells, e.g., hypoimmune islet cells, e.g., human hypoimmune islet cells), such as any of the engineered cells provided herein. In some embodiments, a kit or article of manufacture comprises a composition comprising a population
of engineered cells (e.g., islet cells, e.g., hypoimmune islet cells, e.g., human hypoimmune islet cells), wherein the engineered cells comprise: (i) a transgene comprising an exogenous polynucleotide encoding CD47, and (ii) inactivation or disruption of both alleles of a B2M gene. In some embodiments, the engineered cells (e.g., islet cells, e.g., hypoimmune islet cells, e.g., human hypoimmune islet cells) further comprise inactivation or disruption of both alleles of a C11TA gene.
[0833] The label or package insert indicates that the composition is used for treating the particular condition (e.g., T1D). The label or package insert will further comprise instructions for administering the pharmaceutical composition to the subject. In some embodiments, the article of manufacture comprises a combination treatment.
[0834] The article of manufacture and/or kit may further comprise a package insert. The insert refers to instructions customarily included in commercial packages of therapeutic products that contain information about the indications, usage, dosage, administration, contraindications and/or warnings concerning the use of such therapeutic products.
[0835] The disclosure is further illustrated by the following examples. The examples are provided for illustrative purposes only. They are not to be construed as limiting the scope or content of the disclosure in any way.
EXAMPLES
[08361 The following examples are provided so as to describe to the skilled artisan how to make and use methods and compositions described herein, and are not intended to limit the scope of the present disclosure.
Example 1: Methods
[0837] This Example provides methods useful for generating and testing of non-human animals provided herein. Specifically, this example describes methods used for the generation
and testing of T1D autoimmune mice that include autologous human PBMCs and human iPSC- derived islet cells.
Generation of humanized diabetic T1D mice and transplantation of TID iPSC-derived islet cells
[0838] Female NSG mice (5-8 weeks old) received daily intraperitoneal (i.p.) injection of streptozotocin (STZ) for 5 consecutive days at 60 mg/kg body weight in 0.1 M citrate buffer to induce diabetes. Glucose levels were determined by glucometer in 10 pL blood samples collected by tail vein venepuncture after 4 hours of fasting. Animals with a glucose level of greater than 450 mg/dL served as recipients for immune cell transfer and cell implantation. For adoptive transfer of T1D immune cells, animals were injected intravenously with 10 million T1D PBMCs 3 days before islet transplantation.
[0839] One thousand islet clusters were resuspended in 60 pL of sterile saline and injected intramuscularly (i.m.) into the hindlimb muscle with a 23 G needle. Glucose tolerance testing was performed at study endpoint (30 days after cell transplantation). Mice were fasted for 4 hours, and a baseline blood glucose level was obtained. A glucose bolus of 2 g/kg glucose was injected intraperitoneally and blood glucose levels were measured at 30 minutes postinjection. Blood was collected from study animals and plasma was stored at -80°C for ELISA c- peptide (Mercodia, Uppsala, Sweden) according to the manufacturer’s protocol. In vivo BLI imaging of transplanted iPSC-derived wild-type and HIP islet cells was performed as described below.
Cell generation for autologous T1D model
[0840] PBMCs and serum from a T1D volunteer were obtained and iPSCs were generated. iPSCs were genetically engineered using CRISPR guide RNAs and Cas9 to inactivate the B2M and CIITA genes. Flow cytometry was then used to sort cells negative for HLA class I and HLA class II expression. The iPSCs were then transduced with lentiviral particles carrying transgenes for CD47 and firefly luciferase. Next, flow cytometry was used to sort iPSCs overexpressing CD47, thereby generating hypoimmune (HIP) cells that overexpress CD47 and
are deficient for HLA class I and HLA class II molecules. Then, the HIP iPSCs were differentiated into islet cells. The islet differentiation protocol was adapted from the previously reported protocol (Millman, J.R., et al. Generation of stem cell-derived beta-cells from patients with type 1 diabetes. Nat Commun 7, 11463 (2016)).
Islet immunofluorescence
[0841] Approximately 20 mL of aggregates were collected into a 1 .5 mL Eppendorf tube and spun down to generate a pellet. The pellet was resuspended in fixation/permeabilization working solution (eBioscience - 00-5523-00) and incubated overnight at 4°C. Cells were washed with permeabilization working buffer (eBioscience - 00-5523-00) and stained with 5 pL each of antibodies that bind Glucagon (Novus Biologicals - NBP2-21803AF647), Somatostatin (Novus Biologicals - NBP2-99309AF350), and Insulin (Fife Technologies - 53-9769-82) for 24 hours at 4°C. Cells were washed with permeabilization working buffer, mounted to slides with Prolong Gold (Fisher Scientific - P36930), and allowed to dry overnight. Slides were then imaged on a Leica Thunder Imaging System.
Imaging of wild-type and HIP iPSC-derived islet cells
[0842] Approximately 20 pL of aggregates were collected into a 1.5 mL Eppendorf tube and washed with stain buffer (DPBS with 0.1% BSA and 5mM EDTA). Aggregates were stained with 5 pL of PE anti-human CD47 (Biolegend - 323108), APC anti-human HLA- ABC (BD Bioscience - 562006) for 45 minutes on ice, and washed with stain buffer. Cells were fixed with BD Cytofix on ice for 30 minutes (Fisher Scientific - 554655), washed with stain buffer, mounted on slides with Prolong Gold with DAPI (Fisher Scientific - P36931), and allowed to dry overnight. Slides were then imaged on a Leica Thunder Imaging System.
Flow cytometry
[0843] To assess HLA class I expression, single islet cells were dissociated and labeled with APC labeled anti-HLA-A,B,C antibody (clone G46_2.6,BD Biosciences) or APC- conjugated IgGl isotype-matched control antibody (clone MOPC-21, BD Biosciences). To assess HLA class II expression, cells were incubated with Alexa- flour647-labeled anti-HLA-
DR,DP,DQ antibody (clone Tu3a, BD Biosciences) or Alexa-flour647-labeled IgG2a isotype- matched control antibody (clone G155-178, BD Biosciences). To assess CD47 expression, the PerCP-Cy5-conjugated anti-CD47 (clone B6H12, BD Biosciences) or PerCP-Cy5-conjugated IgGl isotype-matched control antibody (clone MOPC-21, BD Biosciences) was used. Results have been normalized, and representative histograms are shown in Figure 3.
Islet composition
[0844] Human unmodified (auto iPSC-derived islets) or HIP iPSC-derived islets were transduced with EFla-GFP-LVV with a MOI of 50 and were sorted for GFP+ cells. 300 islet clusters were transplanted into the hindlimb muscle of NGS mice with a 27G needle. Injection site was harvested on Day 0, Day 7, or Day 28 and were subsequently analyzed in flow. To create single cell suspensions, muscle samples were cut into small pieces and filtered through a 70 pm cell strainer. Cells were pre-gated GFP+ cells. For separating the different islet cell types, the following antibodies were used: anti-insulin antibody (clone 2D11-H5, Santa Cruz Biotechnology), anti-glucagon antibody (clone C-l 1, Santa Cruz Biotechnology) and antisomatostatin antibody (clone G-10, Santa Cruz Biotechnology). Results are shown as percentage of auto iPSC-derived islets or percentage of HIP iPSC-derived islets (see Figure 4).
Insulin secretion in vitro
[0845] For the insulin secretion assay, 100 islet clusters were plated in one well of a low attachment 6-well plate (Thermo Fisher) in 2 mL islet media including 5mM glucose (Prodo, PIM(S) media). Supernatant was collected after 24 hours and centrifuged at 10,000 rpm to remove any cells and debris. Human Insulin ELISA assay was performed according to manufacturer’s instructions (Thermo Fisher). Briefly, islet cell supernatant was incubated with an anti-insulin antibody, followed by incubation with a horseradish peroxidase (HRP)- conjugated secondary antibody and a peroxidase substrate. A microplate reader with an absorbance of optical density (OD) 450 nm (Molecular Devices) was used to measure the insulin level of the standards and study samples. Insulin levels were calculated to pLU per million cells.
Glucose monitoring
[08461 Glucose measurements were performed using a glucometer (AccuCheck, Roche, Basel, Switzerland) after 4 hours of fasting on Day -5, Day -3, Day -2, Day -1, Day 0, Day 3, Day 7, Day 10, Day 14, Day 21, and Day 28 after islet transplantation. A glucose level above 200 mg/dL was considered to be diabetic. For a glucose challenge, mice received 2 g/kg glucose solution (Thermo Fisher) intraperitoneally and glucose level was determined 30 minutes after the glucose injection.
Innate killing in vitro by XCelligence
[0847] NK-cell and macrophage killing assays were performed on the XCelligence MP platform (ACEA BioSciences, San Diego, CA). 96-well E-plates (ACEA BioSciences) were coated and 4e4 target islet cells were plated in 100 pL Prodo PIM(S) (Prodo) media. After the Cell Index reached 0.7, effector cells were added at an effector cell to target cell (E:T) ratio of 1:1. NK cells were stimulated with 1 pg/mL human IL2 (Peprotech). In some wells, an anti- CD47 antibody (Magrolimab 5F9, Creative Biolabs) was added at a concentration of 100 ug/mL. As a killing control, cells were treated with 2% TritonXIOO or media only were added (data not shown). Data were standardized and analyzed with the RTCA software (ACEA).
In vivo BLI imaging
[0848] Mice were monitored on Day 0, Day 3, and every second day until Day 13 and subsequently every 4 days until Day 29. D-luciferin firefly potassium salt (375 mg/kg, Biosynth) was dissolved in sterile PBS (pH 7.4, Gibco, Invitrogen) and was injected intraperitoneally (250 pL per mouse) into anesthetized mice. Animals were imaged using the Ami HT (Spectral Instruments Imaging) and ROI bioluminescence was quantified in units of maximum photons per second per centimeter square per steradian (p s-1 cm-2 sr-1). The maximum signal from an ROI was measured using Aura software (Spectral Instruments Imaging).
Splenocyte killing assay
[0849] Splenocyte killing assays were performed on the XCelligence MP platform. 96- well E-plates (ACEA BioSciences) were coated and 4xl04 target islet cells were plated in 100 pL Prodo PIM (Prodo) media. When the cell index reached 0.7, effector cells were added at an
effector cell to target cell (E:T) ratio of 1:1. As a killing control, cells were treated with 2% TritonXIOO or media only were added (data not shown). Data were standardized and analyzed with the RTCA software (ACEA).
C-peptide ELISA
[0850] A c-peptide ELISA kit (Mercodia, Winston Salem, NC) was used to measure human c-peptide in serum. Samples were diluted and pipetted according to manufacturer’s instructions. Briefly, standards and samples were added to pre-coated 96-well ELISA plates and incubated for 1 hour. After the removal of unbound proteins by washing, anti-c-peptide antibodies conjugated with horseradish peroxidase were added. These enzyme-labeled antibodies form complexes with the previously bound c-peptide. The enzyme bound to the immunosorbent was assayed by the addition of a chromogenic substrate, tetramethylbenzidine. Samples were analyzed in a microplate reader (ThermoFisher).
Example 2: Human iPSC-derived HIP p-islets escape autoimmunity in autologous, diabetic humanized mice
[0851] PBMCs from a patient with long-standing type 1 diabetes (T1D) and measurable GAD65, ZT8, and IA-2 antibodies were used to engraft NSG-SGM3 mice (T1D mice, Figure 1). Because beta cells are destroyed in T1D patients and thus are not available for transplantation, beta cells were differentiated from iPSCs derived from the patient’s PBMCs. Beta cells were generated from the same PBMC pool for introduction into a mouse; one fraction of PBMCs underwent HIP engineering (B2M/CIITA inactivation and CD47/luc overexpression), while the other fraction was transduced to overexpress luc only without HIP engineering. Both autologous (auto) and HIP iPSCs were then differentiated into autologous islet cells for subsequent transplantation into T1D mice (Figure 1). As shown in Figure 2, in vitro immunofluorescence staining of auto-derived and HIP iPSC-derived p-islets for somatostatin, insulin, and glucagon (top two images) and for CD47, HLA class I, and DAPI (bottom two images) was performed.
Figure 3 illustrates exemplary results of HLA class I, HLA class II, and CD47 expression as
assessed by flow cytometry in auto-derived p-islets (top row) and in HIP iPSC-derived p-islets (bottom row). Figure 4 illustrates exemplary results of the composition of cell types (alpha cells, beta cells, delta cells, and other cells) in auto-derived and HIP iPSC-derived p-islets as assessed by flow cytometry. Auto-derived and HIP iPSC-derived p-islets showed similar size, morphology, endocrine hormone content and cell composition, but only HIP-derived p-islets showed abundant CD47 and lack of HLA class I expression (Figures 2-4). Both auto-derived and HIP iPSC-derived p-islets released similar amounts of insulin in vitro, as assessed by ELISA (Figure 5).
[0852] Auto-derived and HIP-derived p-islets were then transplanted into autologous humanized T1D mice generated with autologous PBMCs from the same T1D patient used to generate the auto-derived and HIP-derived p-islets. The first injection was made into the right thigh muscle (Figure 6). A second injection, of auto-derived p-islets, was made on day 15 for recipients who received a first injection of the HIP-derived p-islets (Figure 6). The experiment was performed three times. As illustrated by exemplary in vivo bioluminescent images and an exemplary graph of bioluminescent signal, the auto-derived p-islets vanished quickly over just 5 to 10 days, confirming the establishment of transferred human autoimmune disease (via the human PBMCs) in these T1D mice (Figures 7 and 8). Transplantation of auto-derived p-islets had no effect on either fasting blood glucose levels or blood glucose levels after a 30 minute glucose challenge (Figure 9). Impedance-based killing assays with splenocytes and serum from T1D mice that rejected auto-derived p-islets also killed auto-derived p-islet cells in vitro (Figure 10). Auto-derived p-islet cells were not killed by NK cells or macrophages (Figure 11). HIP- derived p-islets survived in T1D mice, and their survival was not impacted by the fact that subsequently transplanted auto-derived p-islets were rejected in the same mice (Figures 12 and 13). HIP-derived p-islets very reliably achieved glycemic control in the animals within 2 weeks, as illustrated by fasting blood glucose levels and blood glucose levels after a 30 minute glucose challenge (Figure 14). In vitro killing assays performed with splenocytes and serum from T1D mice that received HIP-derived p-islets, along with NK cells or macrophages from the same mice, confirmed that there was no immune response against HIP-derived p-islet cells (Figures
15 and 16). On Day 30, only T1D mice that received HIP-derived p-islets had relevant c-peptide levels (Figure 17). Together, these results show that HIP-derived p-islets efficiently avoided autoimmune killing in a humanized mouse model.
EXEMPLARY SEQUENCES
[0853] The following tables provide exemplary amino acid and nucleic acid sequences as described herein.
EQUIVALENTS
[0854] It is to be understood that while the disclosure has been described in conjunction with the detailed description thereof, the foregoing description is intended to illustrate and not limit the scope of the invention, which is defined by the scope of the appended claims. Other aspects, advantages, and modifications arc within the scope of the following claims.
Claims
1. A non-human animal comprising immune cells from a human patient with type 1 diabetes (T1D) and islet cells derived from cells of the human patient with T1D, wherein the immune cells and the islet cells are autologous.
2. The non-human animal of claim 1, wherein the non-human animal is a non-human mammal.
3. The non-human animal of any of the previous claims, wherein the non-human animal is a rodent.
4. The non-human animal of any of the previous claims, wherein the non-human animal is a mouse.
5. The non-human aminal of any of the previous claims, wherein the non-human animal comprises one or more genetic modifications that arc suitable for maintaining a xenograft.
6. The non-human animal of claim 5, wherein the one or more genetic modifications comprise reducing or eliminating expression of one or more genes.
7. The non-human animal of claim 5 or 6, wherein the one or more genetic modifications comprise increasing expression of one or more genes.
8. The non-human animal of any one of claims 5-7, wherein the one or more genetic modifications comprise reducing or eliminating expression of one or more genes and increasing expression of one or more genes.
9. The non-human animal of claim 8, wherein the mouse is a NOD mouse, optionally an NSG-SGM3 mouse.
10. The The non-human animal of claim 8, wherein the mouse is a SC1D mouse, a NOD/SCID mouse, a NOD/SCID/ IL-2Rg(null) mouse,
mouse, a NOD.Cg-Prhfcscid I12rgtmlwjl Tg(CMV-L3,CSF2,KITLG)lEav/MloySzJ (NSG-SGM3) mouse, a NSG-human IL3, NSG-SCF, NSG-GM-GSF mouse, a nude mouse, a ragl knockout mouse, or rag2 knockout mouse.
11. The non-human animal of any of the previous claims, wherein the human patient with T1D is eligible for islet transplant therapy.
12. The non-human animal of any of the previous claims, wherein the human patient with T1D has or exhibits one or more of the following:
(a) a clinical history compatible with T1D onset of disease at younger than 40 years old;
(b) insulin dependence for at least 5 years;
(c) a sum of age and years of insulin dependent diabetes duration of at least 28 years;
(d) absent stimulated C-peptide (less than 0.3 ng/mL) 60 and 90 minutes post-mixed- meal tolerance test;
(e) involvement of intensive diabetes management, wherein intensive diabetes management comprises:
(i) self-monitoring of glucose values no less than a mean of three times each day averaged over each week;
(ii) administration of three or more insulin injections each day or insulin pump therapy; and/or
(iii) under the direction of an endocrinologist, diabetologist, or diabetes specialist with at least three clinical evaluations during the past 12 months;
(f) at least one episode of severe hypoglycemia in the past 12 months, wherein an episode of severe hypoglycemia comprises an event with one or more of the following symptoms:
(i) memory loss;
(ii) confusion;
(iii) uncontrollable behavior;
(iv) irrational behavior;
(v) unusual difficulty in awakening;
(vi) suspected seizure;
(vii) seizure;
(viii) loss of consciousness;
(ix) visual symptoms; and/or
(g) impaired awareness of hypoglycemia (IAH).
13. The non-human animal of any of the previous claims, wherein the human patient with T1D has had T1D for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 years, at least 8 years, at least 9 years, at least 10 years, at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 years, or at least 90 years.
14. The non-human animal of any of the previous claims, wherein the human patient with T1D has impaired awareness of hypoglycemia (IAH).
15. The non-human animal of claim 12 or 14, wherein IAH comprises: a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more; a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more;
marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more; or a composite of: a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile.
16. The non-human animal of any of the previous claims, wherein the human patient with T1D has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years.
17. The non-human animal of any of the previous claims, wherein the human patient with T1D has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2).
18. The non-human animal of claim 17, wherein antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
19. The non-human animal of any of the previous claims, wherein one or more of the immune cells comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
20. The non-human animal of any of the previous claims, wherein one or more of the immune cells are isolated from peripheral blood mononuclear cells (PBMCs).
21. The non-human animal of claim 20, wherein one or more of the PBMCs comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
22. The non-human animal of any of the previous claims, wherein one or more of the immune cells produce autoantibodies against cells or antigens of the human patient with T1D.
23. The non-human animal of any of the previous claims, wherein one or more of the immune cells is a B cell.
24. The non-human animal of any of the previous claims, wherein one or more of the immune cells produce antibodies against one or more islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2.
25. The non-human animal of any of the previous claims, wherein one or more of the immune cells produce islet cell antibodies (ICA) and/or insulin autoantibodies (IAA).
26. The non-human animal of any of the previous claims, wherein one or more of the immune cells is an islet-reactive T cell.
27. The non-human animal of any of the previous claims, wherein one or more of the immune cells are present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal.
28. The non-human animal of any of the previous claims, wherein the non-human animal comprises at least about 2.5xl05, at least about 5xl05, at least about IxlO6, at least about 2xl06, at least about 3xl06, at least about 4xl06, at least about 5xl06, at least about 6xl06, at least about 7xl06, at least about 8xl06, at least about 9xl06, at least about IxlO7, at least about 1.5xl07, or at least about 2xl07 immune cells.
29. The non-human animal of claim 20 or 21 , wherein the non-human animal comprises at least about 2.5xl05, at least about 5xl05, at least about IxlO6, at least about 2xl06, at least about 3xl06, at least about 4xl06, at least about 5xl06, at least about 6xl06, at least about 7xl06, at least about 8xl06, at least about 9xl06, at least about IxlO7, at least about 1.5xl07, or at least about 2xl07 PBMCs.
30. The non-human animal of any of the previous claims, wherein the non-human animal comprises reduced function of non-human islet cells relative to a comparable wild-type non- human animal.
31. The non-human animal of any of the previous claims, wherein the non-human animal comprises reduced activity of non-human islet cells relative to a comparable wild-type non- human animal.
32. The non-human animal of any of the previous claims, wherein the non-human animal comprises a reduced number of non-human islet cells relative to a comparable wild-type non- human animal.
33. The non-human animal of any of the previous claims, wherein the non-human animal does not comprise non-human islet cells.
34. The non-human animal of any of the previous claims, wherein the non-human animal comprises streptozotocin (STZ).
35. The non-human animal of any of the previous claims, wherein the non-human animal was previously administered STZ.
36. The non-human animal of any of the previous claims, wherein the non-human animal.
37. The non-human animal of any of the previous claims, wherein the non-human animal comprises decreased expression of a PDX1 gene.
38. The non-human animal of any of the previous claims, wherein the non-human animal has reduced pancreatic function relative to a comparable wild-type non-human animal.
39. The non-human animal of any of the previous claims, wherein the non-human animal has reduced pancreatic activity relative to a comparable wild-type non-human animal.
40. The non-human animal of any of the previous claims, wherein the non-human animal has a reduced number of pancreatic cells relative to a comparable wild-type non-human animal.
41. The non-human animal of any of the previous claims, wherein the non-human animal does not have a pancreas.
42. The non-human animal of any of the previous claims, wherein the non-human animal comprises splenocytes from an autoimmune and diabetic NOD/ShiLt mouse.
43. The non-human animal of any of the previous claims, wherein the non-human animal has glucose levels greater than about 200 mg/dL, greater than about 300 mg/dL, greater than about
400 mg/dL, or greater than about 450 mg/dL.
44. The non-human animal of any of the previous claims, wherein the non-human animal has glucose levels greater than about 300 mg/dL.
45. The non-human animal of any of the previous claims, wherein the non-human animal has glucose levels greater than about 400 mg/dL.
46. The non-human animal of any of the previous claims, wherein the non-human animal has glucose levels greater than about 450 mg/dL.
47. The non-human animal of any of the previous claims, wherein the islet cells are introduced via intramuscular injection, intravenous injection, intraportal injection, ocular injection, injection into omentum, and/or injection into a kidney capsule.
48. The non-human animal of any of the previous claims, wherein the islet cells are or comprise human alpha, beta, and/or delta islet cells.
49. The non-human animal of claim 48, wherein the islet cells are or comprise about 5%, about 10%, about 15%, about 20%, about 25%, about 35%, or about 40% alpha cells.
50. The non-human animal of claim 48 or 49, wherein the islet cells are or comprise about 10%, about 15%, about 20%, about 25%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, or about 80% beta cells.
51. The non-human animal of any one of claims 48-50, wherein the islet cells are or comprise about 1%, about 2%, about 3%, about 4%, about 5%, about 6%, about 7%, about 8%, about 9%, about 10%, about 15%, about 20%, or about 25% delta cells.
52. The non-human animal of any of the previous claims, wherein the islet cells are or comprise differentiated cells.
53. The non-human animal of claim 52, wherein the differentiated cells are derived from a stem cell.
54. The non-human animal of claim 52 or 53, wherein the differentiated cells are derived from a pluripotent stem cell.
55. The non-human animal of any one of claims 52-54, wherein the differentiated cells are derived from an induced pluripotent stem cell (iPSC).
56. The non-human animal of 55, wherein the iPSC is derived from a cell obtained from the human patient with T1D.
57. The non-human animal of claim 56, wherein the cell obtained from the human patient with T1D is selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell.
58. The non-human animal of claim 56 or claim 57, wherein the cell obtained from the human patient with T1D is a PBMC.
59. The non-human animal of claim 53, wherein the stem cell is derived from a cell obtained from the human patient with T1D.
60. The non-human animal of claim 59, wherein the stem cell is an induced pluripotent stem cell (iPSC) derived from a cell selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell.
61. The non-human animal of claim 59, wherein the stem cell is an induced pluripotent stem cell (iPSC) derived from a PBMC.
62. The non-human animal of any one of claims 52-53, wherein the differentiated cells are derived from a multipotent stem cell.
63. The non-human animal of 62, wherein the multipotent stem cell is derived from a cell obtained from the human patient with T1D.
64. The non-human animal of claim 52 or 53, wherein the differentiated cells are derived from a lineage- specific stem cell.
65. The non-human animal of 64, wherein the lineage- specific stem cell is derived from a cell obtained from the human patient with T1D.
66. The non-human animal of claim 52, wherein the differentiated cells are derived from a definitive endoderm cell.
67. The non-human animal of claims 66, wherein the definitive endoderm cell expresses FOXA2 and/or SOX17.
68. The non-human animal of claim 52, wherein the differentiated cells are derived from a primitive gut tube cell.
69. The non-human animal of claim 68, wherein the primitive gut tube cell expresses FOXA2 and/or SOX 17.
70. The non-human animal of claim 52, wherein the differentiated cells are derived from a pancreatic progenitor cell.
71. The non-human animal of claim 70, wherein the pancreatic progenitor cell expresses PDX1 and/or NKX6-1.
72. The non-human animal of claim 52, wherein the differentiated cells are derived from an endocrine cell.
73. The non-human animal of claim 72, wherein the endocrine cell expresses NKX6-1, NEUROG3, and/or NKX2-2.
74. The non-human animal of any one of claims 1-73, wherein the islet cells are or comprise engineered islet cells.
75. The non-human animal of claim 74, wherein the engineered islet cells have been modified to be hypoimmune islet cells.
76. The non-human animal of claim 75, wherein the hypoimmune islet cells comprise one or more modifications that
(a) inactivate or disrupt one or more alleles of:
(i) one or more major histocompatibility complex (MHC) class 1 molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or
(ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules, and/or
(b) increase expression of one or more tolerogenic factors, wherein the increased expression of (b) is relative to an islet cell that does not comprise the modifications.
77. The non-human animal of claim 75 or 76, wherein the one or more modifications that disrupt one or more MHC class I molecules and/or one or more MHC class II molecules reduce expression of the one or more MHC class I molecules and/or the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications.
78. The non-human animal of any one of claims 75-77, wherein the hypoimmune islet cells are (i) HLA class I-deficient and/or HLA class Il-deficient and (ii) overexpress CD47.
79. The non-human animal of any one of claims 75-78, wherein the hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47.
80. The non-human animal of any one of claims 75-79, wherein the hypoimmune islet cells comprise one or more safety switches.
81. The non-human animal of any one of claims 75-80, wherein the non-human animal comprises an agent that activates the one or more safety switches.
82. The non-human animal of any of the previous claims, wherein the islet cells produce insulin, c-peptide, glucagon, somatostatin, and/or pancreatic polypeptide.
83. The non-human animal of any of the previous claims, comprising a first therapeutic agent.
84. The non-human animal of claim 83, wherein the first therapeutic agent is or comprises the islet cells.
85. The non-human animal of claim 83, wherein the first therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid.
86. The non-human animal of any of the previous claims, further comprising a second therapeutic agent.
87. The non-human animal of claim 85, wherein the second therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid.
88. A method of characterizing human islet cells for delivery to a patient, comprising taking a sample of the islet cells derived from cells of the human patient with T1D and administering the sample to a non-human animal of claims 1-87.
89. A method of making a non-human animal model of type 1 diabetes (T1D), the method comprising:
(a) administering immune cells from a human patient with T1D to the non-human animal,
(b) administering islet cells derived from cells of the human patient with T1D to the nonhuman animal, wherein the immune cells and the islet cells are autologous.
90. The method of claim 89, wherein the non-human animal is a non-human mammal.
91 . The method of claim 89 or 90, wherein the non-human animal is a rodent.
92. The method of any one of claims 89-91, wherein the non-human animal is a mouse.
93. The method of any one of claims 89-92, wherein the non-human animal comprises one or more genetic modifications that are suitable for maintaining a xenograft.
94. The method of claim 93, wherein the one or more genetic modifications comprise reducing or eliminating expression of one or more genes.
95. The method of claim 93 or 94, wherein the one or more genetic modifications comprise increasing expression of one or more genes.
96. The method of any one of claims 93-95, wherein the one or more genetic modifications comprise reducing or eliminating expression of one or more genes and increasing expression of one or more genes.
97. The method of claim 96, wherein the mouse is a NOD mouse, optionally an NSG-SGM3 mouse.
98. The method of claim 96, wherein the mouse is a SCID mouse, a NOD/SCID mouse, a NOD/SCID/ IL-2Rg(null) mouse, a NOD.Cg-PrfcZcscld ZZ2rgtmlw-il/SzJ mouse, a NOD.Cg-
Prkdc^ I12rgtmlWjl Tg(CMV-L3,CSF2,KITLG)lEav/MloySzJ (NSG-SGM3) mouse, a NSG- human IL3, NSG-SCF, NSG-GM-GSF mouse, a nude mouse, a ragl knockout mouse, or rag2 knockout mouse.
99. The method of any one of claims 89-98, wherein the human patient with T ID is eligible for islet transplant therapy.
100. The method of any one of claims 89-99, wherein the human patient has or exhibits one or more of the following:
(a) a clinical history compatible with T1D onset of disease at younger than 40 years old;
(b) insulin dependence for at least 5 years;
(c) a sum of age and years of insulin dependent diabetes duration of at least 28 years;
(d) absent stimulated C-peptide (less than 0.3 ng/mL) 60 and 90 minutes post-mixed- meal tolerance test;
(e) involvement of intensive diabetes management, wherein intensive diabetes management comprises:
(i) self-monitoring of glucose values no less than a mean of three times each day averaged over each week;
(ii) administration of three or more insulin injections each day or insulin pump therapy; and/or
(iii) under the direction of an endocrinologist, diabetologist, or diabetes specialist with at least three clinical evaluations during the past 12 months;
(f) at least one episode of severe hypoglycemia in the past 12 months, wherein an episode of severe hypoglycemia comprises an event with one or more of the following symptoms:
(i) memory loss;
(ii) confusion;
(iii) uncontrollable behavior;
(iv) irrational behavior;
(v) unusual difficulty in awakening;
(vi) suspected seizure;
(vii) seizure;
(viii) loss of consciousness;
(ix) visual symptoms; and/or
(g) impaired awareness of hypoglycemia (IAH).
101. The method of any one of claims 89-99, wherein the human patient with T1D has had T1D for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 years, at least 8 years, at least 9 years, at least 10 years at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 years, or at least 90 years.
102. The method of any one of claims 89-101, wherein the human patient with T1D has impaired awareness of hypoglycemia (IAH).
103. The method of claim 100 or 102, wherein IAH comprises: a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more; a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more; marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more; or a composite of: a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile.
104. The method of any one of claims 89-103, wherein the human patient with T1D has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years.
105. The method of any one of claims 89-104, wherein the human patient with T1D has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2).
106. The method of any one of claims 89-105, wherein antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050 JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
107. The method of any one of claims 89-106, wherein one or more of the immune cells comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
108. The method of any one of claims 89-107, wherein one or more of the immune cells are isolated from peripheral blood mononuclear cells (PBMCs).
109. The method of claim 108, wherein one or more of the PBMCs comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
110. The method of any one of claims 89-109, wherein one or more of the immune cells produce autoantibodies against cells or antigens of the human patient with T1D.
111. The method of any one of claims 89- 110, wherein one or more of the immune cells is a B cell.
112. The method of any one of claims 89- 1 11 , wherein one or more of the immune cells produce antibodies against one or more islet cell antigens, beta islet cell cytoplasmic proteins, insulin, GAD65, ZnT8, and/or IA-2.
113. The method of any one of claims 89- 112, wherein one or more of the immune cells produce islet cell antibodies (ICA) and/or insulin autoantibodies (IAA).
114. The method of any one of claims 89- 113, wherein one or more of the immune cells is an islet-reactive T cell.
115. The method of any of one of claims 89-114, wherein one or more of the immune cells arc present in peripheral blood, bone marrow, spleen, lymph nodes, and/or thymus of the non-human animal.
116. The method of any of any one of claims 89-115, wherein the non-human animal comprises at least 2.5 xlO5, at least 5xl05, at least IxlO6, at least 2xl06, at least 3xl06, at least 4xl06, at least 5xl06, at least 6xl06, at least 7xl06, at least 8xl06, at least 9xl06, at least IxlO7, at least 1.5xl07, or at least 2xl07 immune cells.
117. The method of claim 108 or 109, wherein the non-human animal comprises at least 2.5 xlO5, at least 5xl05, at least IxlO6, at least 2xl06, at least 3xl06, at least 4xl06, at least 5xl06, at
least 6xl06, at least 7xl06, at least 8xl06, at least 9xl06, at least IxlO7, at least 1.5xlO7, or at least 2xl07 PBMCs.
118. The method of any one of claims 89-117, wherein the islet cells are administered to the non-human animal via intramuscular injection, intravenous injection, intraportal injection, ocular injection, injection into omentum, and/or injection into a kidney capsule.
119. The method of any one of claims 89-118, wherein the islet cells are or comprise human alpha, beta, and/or delta islet cells.
120. The method of claim 119, wherein the islet cells are or comprise about 5%, about 10%, about 15%, about 20%, about 25%, about 35%, or about 40% alpha cells.
121. The method of claim 119 or 120, wherein the islet cells are or comprise about 10%, about 15%, about 20%, about 25%, about 35%, about 40%, about 45%, about 50%, about 55%, about 60%, about 65%, about 70%, about 75%, or about 80% beta cells.
122. The method of any one of claims 119-121, wherein the islet cells are or comprise about 1%, about 2%, about 3%, about 4%, about 5%, about 6%, about 7%, about 8%, about 9%, about 10%, about 15%, about 20%, or about 25% delta cells.
123. The method of any one of claims 89-122, wherein the islet cells are or comprise differentiated cells.
124. The method of claim 123, wherein the differentiated cells are derived from a stem cell.
125. The method of claim 123 or 124, wherein the differentiated cells are derived from a pluripotent stem cell.
126. The method of any one of claims 123-125, wherein the differentiated cells are derived from an induced pluripotent stem cell (iPSC).
127. The method of claim 126, wherein the iPSC is derived from a cell obtained from the human patient with T1D.
128. The method of claim 127, wherein the cell obtained from the human patient with T1D is selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell.
129. The method of claim 127 or claim 128, wherein the cell obtained from the human patient with T1D is a PBMC.
130. The method of claim 124, wherein the stem cell is derived from a cell obtained from the human patient with T1D.
131. The method of claim 130, wherein the stem cell is an induced pluripotent stem cell (iPSC) derived from a cell selected from the group consisting of a fibroblast, an adipose stem cell, a dental stem cell, a PBMC, an umbilical cord blood cell (UCBC), a keratinocyte, a melanocyte, a nasal epithelium cell, a urine exfoliated cell, a neural stem cell, a hepatocyte, an astrocyte, and a kidney mesangial cell.
132. The method of claim 130, wherein the stem cell is an induced pluripotent stem cell (iPSC) derived from a PBMC.
133. The method of any one of claims 123-124, wherein the differentiated cells are derived from a multipotent stem cell.
134. The method of 133, wherein the multipotent stem cell is derived from a cell obtained from the human patient with T1D.
135. The method of claim 123 or 124, wherein the differentiated cells are derived from a lineage- specific stem cell.
136. The method of 135, wherein the lineage- specific stem cell is derived from a cell obtained from the human patient with T1D.
137. The method of claim 123, wherein the differentiated cells are derived from a definitive endoderm cell.
138. The method of claims 137, wherein the definitive endoderm cell expresses F0XA2 and/or SOX 17.
139. The method of claim 123, wherein the differentiated cells are derived from a primitive gut tube cell.
140. The method of claim 139, wherein the primitive gut tube cell expresses FOXA2 and/or SOX17.
141. The method of claim 123, wherein the differentiated cells are derived from a pancreatic progenitor cell.
142. The method of claim 141, wherein the pancreatic progenitor cell expresses PDX1 and/or NKX6-1.
143. The method of claim 123, wherein the differentiated cells are derived from an endocrine cell.
144. The method of claim 143, wherein the endocrine cell expresses NKX6-1 , NEUR0G3, and/or NKX2-2.
145. The method of any one of claims 89-144, wherein the islet cells are or comprise engineered islet cells.
146. The method of claim 145, wherein the engineered islet cells have been modified to be hypoimmune islet cells.
147. The method of claim 146, wherein the hypoimmune islet cells comprise one or more modifications, wherein the modifications
(a) inactivate or disrupt one or more alleles of:
(i) one or more major histocompatibility complex (MHC) class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or
(ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class 11 molecules, and/or
(b) increase expression of one or more tolerogenic factors, wherein the increased expression of (ii) is relative to an islet cell that does not comprise the modifications.
148. The method of claim 146 or 147, wherein the one or more modifications reduce expression of the one or more MHC class I molecules and/or one or more molecules that regulate
expression of the one or more MHC class I molecules, and/or reduce expression of the one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications.
149. The method of any one of claims 146-148, wherein the hypoimmune islet cells are (i) HLA class I-deficient and/or HLA class Il-deficient and (ii) overexpress CD47.
150. The method of any one of claims 147-149, wherein the hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47.
151. The method of any one of claims 146-150, wherein the hypoimmune islet cells comprise one or more safety switches.
152. The method of any one of claims 146-151, wherein the non-human animal comprises an agent that activates the one or more safety switches.
153. The method of any one of claims 89-145, wherein the islet cells produce insulin, glucagon, somatostatin, and/or pancreatic polypeptide.
154. The method of any one of claims 89-153, further comprising a step of:
(c) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal.
155. The method of claim 154, wherein reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises administering an islet-cell destroying composition to the non-human animal.
156. The method of claim 155, wherein the islet-cell destroying composition is or comprises
STZ.
157. The method of claim 155, wherein the islet-cell destroying composition is or comprises alloxan.
158. The method of claim 155, wherein the islet-cell destroying composition is or comprises splenocytes from an autoimmune and diabetic NOD/ShiLt mouse.
159. The method of claim 154, wherein reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises removing the non-human animal’s pancreas.
160. The method of claim 154, wherein reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises decreasing expression of a PDX1 gene in the non-human animal.
161. The method of any one of claims 154-160, wherein the non-human animal has reduced endogenous islet cell function relative to a comparable wild-type non-human animal.
162. The method of claim any one of claims 154-161, wherein the non-human animal has eliminated endogenous islet cell function relative to a comparable wild-type non-human animal.
163. The method of any one of claims 154-162, wherein the non-human animal has reduced endogenous islet cell activity relative to a comparable wild-type non-human animal.
164. The method of claim any one of claims 154-163, wherein the non-human animal has eliminated endogenous islet cell activity relative to a comparable wild-type non-human animal.
165. The method of any one of claims 154-164, wherein the non-human animal has reduced levels of endogenous islet cells relative to a comparable wild-type non-human animal.
166. The method of any one of claims 154-165, wherein the non-human animal does not comprise endogenous islet cells.
167. The method of any one of claims 154-163, wherein the non-human animal has glucose levels greater than about 200mg/dL, greater than about 300mg/dL, greater than about 400mg/dL, or greater than about 450mg/dL.
168. The method of any one of claims 154-167, wherein the non-human animal has glucose levels greater than about 300mg/dL.
169. The method of any one of claims 154-168, wherein the non-human animal has glucose levels greater than about 400mg/dL.
170. The method of any one of claims 154-169, wherein the non-human animal has glucose levels greater than about 450mg/dL.
171. The method of any one of claims 154-170, wherein the method comprises, in sequential order:
(a) administering immune cells from the human patient with T1D to the non-human animal,
(c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal, and
(b) administering the islet cells derived from cells of the human patient with T1D to the non-human animal.
172. The method of any one of claims 154-168, wherein the method comprises, in sequential order:
(c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal,
(b) administering the islet cells derived from cells of the human patient with T1D to the non-human animal, and
(a) administering immune cells from the human patient with T1D to the non-human animal.
173. The method of any one of claims 154-168, wherein the method comprises, in sequential order:
(c) reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal,
(a) administering immune cells from the human patient with T1D to the non-human animal, and
(b) administering islet cells derived from cells of the human patient with T1D to the non- human animal.
174. The method of any one of claims 154-173, wherein the islet-cell destroying composition is administered to the non-human animal for 5 continuous days, 4 continuous days, 3 continuous days, 2 continuous days, or 1 continuous day.
175. The method of any one of claims 154-174, wherein the islet-cell destroying composition is administered to the non-human animal 5 days, 4 days, 3 days, 2 days, or 1 day before the immune cells are administered to the non-human patient.
176. The method of any one of claims 89-174, wherein no more than 1 day, 2 days, 3 days, 4 days, or 5 days elapse between step (a) and step (b).
177. A method comprising administering immune cells and islet cells derived from cells of a human patient with type 1 diabetes (T1D) to a non-human animal, wherein the immune cells and the islet cells are autologous.
178. The method of claim 177, wherein the non-human animal comprises immune cells from a human patient with T1D.
179. A method of using a non-human animal model of type 1 diabetes (T1D), the method comprising:
(a) administering immune cells from a human patient with T1D to the non-human animal,
(b) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous and
(c) determining the survival and/or endocrine function of the islet cells.
180. The method of claim 179, wherein the islet cells are or comprise engineered islet cells.
181. The method of claim 180, wherein the engineered islet cells have been modified to be hypoimmune islet cells.
182. The method of claim 181, wherein the hypoimmune islet cells comprise one or more modifications, wherein the modifications
(a) inactivate or disrupt one or more alleles of:
(i) one or more MHC class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or
(ii) one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules, and/or
(b) increase expression of one or more tolerogenic factors, wherein the increased expression of (ii) is relative to an islet cell that does not comprise the modifications.
183. The method of claim 181 or 182, wherein the one or more modifications reduce expression of the one or more MHC class I molecules and/or one or more molecules that regulate expression of the one or more MHC class I molecules, and/or reduce expression of the one or more MHC class II molecules and/or one or more molecules that regulate expression of the one or more MHC class II molecules relative to an islet cell that does not comprise the modifications.
184. The method of any one of claims 181-183, wherein the hypoimmune islet cells are (i) HLA class I-deficient and/or HLA class Il-deficient and (ii) overexpress CD47.
185. The method of any one of claims 181-184, wherein the hypoimmune islet cells are (i) HLA class I and class Il-deficient and (ii) overexpress CD47.
186. The method of any one of claims 181-185, wherein the hypoimmune islet cells comprise one or more safety switches.
187. The method of any one of claims 179-184, wherein the islet cells are a therapeutic agent.
188. The method of claim 187, wherein step (c) comprises determining the survival and/or endocrine function of the islet cells after an autoimmune challenge.
189. The method of any one of claims 179-188, further comprising: (d) administering an agent to the non-human animal.
190. The method of claim 189, wherein the agent activates the one or more safety switches.
191. The method of claim 190, wherein step (c) further comprises evaluating effects of the one or more modifications on the hypoimmune islet cells.
192. The method of claim 189, wherein the agent is a therapeutic agent.
193. The method of claim 192, further comprising:
(e) administering an immunosuppressant to the non-human animal.
194. A method of characterizing one or more therapeutic agents, the method comprising, in sequential order:
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal,
(b) administering immune cells from a human patient with T1D to the non-human animal,
(c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells are a first therapeutic agent,
(d) optionally administering a second therapeutic agent to the non-human animal, and
(e) assessing the effect of the islet cells and/or the one or more therapeutic agents on one or more therapeutically relevant T1D measures.
195. A method of characterizing one or more therapeutic agents, the method comprising, in sequential order:
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal,
(c) administering islet cells derived from cells of a human patient with T1D to the non- human animal, wherein the islet cells are a first therapeutic agent,
(b) administering immune cells from the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous,
(d) optionally administering a second therapeutic agent to the non-human animal, and
(e) assessing the effect of the islet cells and/or the one or more therapeutic agent on one or more therapeutically relevant T1D measures.
196. A method of characterizing one or more therapeutic agents, the method comprising, in sequential order:
(b) administering immune cells from a human patient with T1D to a non-human animal,
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal,
(c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells are a first therapeutic agent,
(d) optionally administering a second therapeutic agent to the non-human animal, and
(e) assessing the effect of the islet cells and/or the one or more therapeutic agent on one or more therapeutically relevant T1D measures.
197. A method of determining if one or more therapeutic agents are suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order:
(b) administering immune cells from a human patient with T1D to a non-human animal,
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal,
(c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells derived from the human patient with T1D are a first therapeutic agent,
(d) optionally administering a second therapeutic agent to the non-human animal, and
(e) determining the suitability of the one or more therapeutic agents to make a T1D therapeutic by assessing the effect of the one or more therapeutic agents on one or more therapeutically relevant T1D measures.
198. A method of determining if one or more therapeutic agents are suitable for use in treating type 1 diabetes (T1D), the method comprising, in sequential order:
(b) administering immune cells from a human patient with T1D to a non-human animal,
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal,
(c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells derived from the human patient with T ID are a first therapeutic agent,
(d) optionally administering a second therapeutic agent to the non-human animal, and
(e) determining the suitability of the one or more therapeutic agents to treat T1D by assessing the effect of the one or more therapeutic agents on one or more therapeutically relevant T1D measures.
199. A method of making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order:
(b) administering immune cells from a human patient with T1D to a non-human animal,
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal,
(c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous, and wherein the islet cells derived from the human patient with T ID are a first therapeutic agent,
(d) optionally administering a second therapeutic agent to the non-human animal,
(e) determining the suitability of the first therapeutic agent, and optionally the second therapeutic agent, to make a T1D therapeutic by assessing the effect of the first therapeutic
agent, and optionally the second therapeutic agent, on one or more therapeutically relevant T ID measures, and
(f) making the T1D therapeutic.
200. A method of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order:
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal,
(b) administering immune cells from a human patient with T1D to the non-human animal,
(c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous,
(d) administering a therapeutic agent to the non-human animal, and
(e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
201. A method of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order:
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in a non-human animal,
(c) administering islet cells derived from cells of a human patient with T1D to the non- human animal,
(b) administering immune cells from the human patient with T1D to the non-human animal, wherein the immune cells and the islet cells are autologous,
(d) administering a therapeutic agent to the non-human animal, and
(e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
202. A method of characterizing a candidate type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order:
(b) administering immune cells from a human patient with T1D to a non-human animal,
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal,
(c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous,
(d) administering a therapeutic agent to the non-human animal, and
(e) assessing the effect of the islet cells and/or the therapeutic agent on one or more therapeutically relevant T1D measures.
203. A method of determining if a therapeutic agent is suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order:
(b) administering immune cells from a human patient with T1D to a non-human animal,
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal,
(c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous,
(d) administering the therapeutic agent to the non-human animal, and
(e) determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures.
204. A method of determining if a therapeutic agent is suitable for use in making a type 1 diabetes (T1D) therapeutic, the method comprising, in sequential order:
(b) administering immune cells from a human patient with T1D to a non-human animal,
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal,
(c) administering islet cells derived from cells of the human patient with T1D to the nonhuman animal, wherein the immune cells and the islet cells are autologous,
(d) administering the therapeutic agent to the non-human animal, and
(e) determining the suitability of the therapeutic agent to treat T1D by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures.
205. A method of making a type 1 diabetes (T1 D) therapeutic, the method comprising, in sequential order:
(b) administering immune cells from a human patient with T1D to a non-human animal,
(a) reducing, removing, or destroying endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal,
(c) administering islet cells derived from cells of the human patient with T1D to the non- human animal, wherein the immune cells and the islet cells are autologous,
(d) administering a therapeutic agent to the non-human animal,
(e) determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of the therapeutic agent on one or more therapeutically relevant T1D measures, and
(f) making the T1D therapeutic.
206. The method of any one of claims claim 194-205, wherein one or more therapeutically relevant T1D measures comprise the survival and/or endocrine function of the islet cells.
207. The method of any one of claims 194-206, wherein one or more therapeutically relevant T1D measures comprise levels of insulin, glucagon, somatostatin, pancreatic polypeptide, C- peptide, and/or glucose in the non-human animal.
208. The method of any one of claims 194, 195, 196, 200, 201, or 202, wherein if a therapeutic agent is characterized as a suitable T1D therapeutic, the method further comprises a step of making a T1D therapeutic.
209. The method of any one of claims 197, 198, 203, 204, wherein if a therapeutic agent is determined to be a suitable T1D therapeutic, the method further comprises a step of making a T1D therapeutic.
210. The method of claim 208 or 209, wherein a suitable T1D therapeutic comprises one or more of the following effects of the therapeutic agent on one or more therapeutically relevant T1D measures:
(a) C-peptide levels greater than at least 100 pmol/L, at least 200 pmol/L, at least 300 pmol/L, at least 400 pmol/L, at least 500 pmol/L, at least 1000 pmol/L, at least 1500 pmol/L, or at least 1750 pmol/L, optionally wherein C-peptide levels are determined using 60 and 90 minutes post-mixed-meal tolerance test;
(b) glucose levels lower than about 200mg/dL, lower than about 300mg/dL, lower than about 400mg/dL, or lower than about 450mg/dL; and/or
(c) islet cells survive for at least 5 days, at least 10 days, at least 15 days, at least 20 days, at least 25 days, or at least 30 days.
211. The method of any one of claims 194-210, wherein reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises administering an islet-cell destroying composition to the nonhuman animal.
212. The method of claim 211, wherein the islet-cell destroying composition is or comprises STZ.
213. The method of claim 211, wherein the islet-cell destroying composition is or comprises splenocytes from an autoimmune and diabetic NOD/ShiLt mouse.
214. The method of any one of claims 194-210, wherein reducing, removing, or destroying the endogenous islet cells, endogenous islet cell function, and/or endogenous islet cell activity in the non-human animal comprises removing the non-human animal’s pancreas.
215. The method of any one of claims 194-214, wherein the non-human animal has reduced endogenous islet cell function relative to a comparable wild-type non-human animal.
216. The method of claim any one of claims 194-215, wherein the non-human animal has eliminated endogenous islet cell function relative to a comparable wild-type non-human animal.
217. The method of any one of claims 194-216, wherein the non-human animal has reduced endogenous islet cell activity relative to a comparable wild-type non-human animal.
218. The method of claim any one of claims 194-217, wherein the non-human animal has eliminated endogenous islet cell activity relative to a comparable wild-type non-human animal.
219. The method of any one of claims 194-218, wherein the non-human animal has reduced levels of endogenous islet cells relative to a comparable wild-type non-human animal.
220. The method of any one of claims 194-219, wherein the non-human animal does not comprise endogenous islet cells.
221. The method of any one of claims 194-199, wherein the second therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid.
222. The method of any one of claims 200-205, wherein a therapeutic agent is or comprises a small molecule, an antibody, an antibody fragment, a blocking peptide, a receptor agonist, or a nucleic acid.
223. The method of any one of claims 194-205, wherein the step of determining the suitability of the therapeutic agent to make a T1D therapeutic by assessing the effect of the islet cells and/or the one or more therapeutic agents on one or more therapeutically relevant T1D measures comprises the use of two or more assays to characterize the islet cells and/or the one or more therapeutic agents and/or to determine the suitability of islet cells and/or the one or more therapeutic agents for making a T1D therapeutic.
224. A method of treating a human patient, comprising administering a pharmaceutical composition comprising a T1D therapeutic made in any one of claims 199, 205, 208, or 209.
225. The method of claim 224, wherein the human patient has been diagnosed as having type 1 diabetes (T1D).
226. The method of claim 225, wherein the human patient is eligible for islet transplant therapy.
227. The method of claim 226, wherein the human patient exhibits one or more of the following:
(a) a clinical history compatible with T1D onset of disease at younger than 40 years old;
(b) insulin dependence for at least 5 years;
(c) a sum of age and years of insulin dependent diabetes duration of at least 28 years;
(d) absent stimulated C-peptide (less than 0.3 ng/mL) 60 and 90 minutes post-mixed- meal tolerance test;
(e) involvement of intensive diabetes management, wherein intensive diabetes management comprises:
(i) self-monitoring of glucose values no less than a mean of three times each day averaged over each week;
(ii) administration of three or more insulin injections each day or insulin pump therapy; and/or
(iii) under the direction of an endocrinologist, diabetologist, or diabetes specialist with at least three clinical evaluations during the past 12 months;
(f) at least one episode of severe hypoglycemia in the past 12 months, wherein an episode of severe hypoglycemia comprises an event with one or more of the following symptoms:
(i) memory loss;
(ii) confusion;
(iii) uncontrollable behavior;
(iv) irrational behavior;
(v) unusual difficulty in awakening;
(vi) suspected seizure;
(vii) seizure;
(viii) loss of consciousness;
(ix) visual symptoms; and/or
(g) impaired awareness of hypoglycemia (IAH).
228. The method of claim 224, wherein the human patient has had T1D for at least 1 month, at least 2 months, at least 3 months, at least 4 months, at least 5 months, at least 6 months, at least 7 months, at least 8 months, at least 9 months, at least 10 months, at least 11 months, at least 1 year, at least 2 years, at least 3 years, at least 4 years, at least 5 years, at least 6 years, at least 7 years, at least 8 years, at least 9 years, at least 10 years, at least 15 years, at least 20 years, at least 30 years, at least 40 years, at least 50 years, at least 60 years, at least 70 years, at least 80 years, or at least 90 years.
229. The method of claim 224, wherein the human patient has impaired awareness of hypoglycemia (IAH).
230. The method of claim 227 or 229, wherein IAH comprises: a Clarke reduced awareness score of about 3 or more, about 4 or more, or about 5 or more; a HYPO score greater than or equal to the 90th percentile and/or about 1047 or more; marked glycemic lability characterized by wide swings in blood glucose despite optimal diabetes therapy and a liability index (LI) score greater than or equal to the 90th percentile and/or about 433 mmol/L2/h wk-1 or more; or a composite of: a Clarke score of 4 or more, a HYPO score greater than or equal to the 75th percentile, and an LI greater than or equal to the 75th percentile.
231. The method of any one of claims 224-230, wherein the human patient has had at least one severe hypoglycemic event (SHE) in the last year, last 2 years, last 3 years, last 4 years, or last 5 years.
232. The method of any one of claims 224-231, wherein the human patient has detectable levels of islet cell antibodies, antibodies against beta islet cell cytoplasmic proteins (e.g., ICA), antibodies to glutamic acid decarboxylase (GAD65), insulin autoantibodies (IAA), antibodies against ZnT8, and/or antibodies to protein tyrosine phosphatase (e.g., IA-2).
233. The method of claim 232, wherein antibodies against beta islet cell cytoplasmic proteins are present at a level greater than or equal to 10 juvenile diabetes foundation units (JDFU), antibodies to glutamic acid decarboxylase are present at a level greater than or equal to 0.033 JDFU, insulin autoantibodies are present at a level greater than or equal to 0.010 JDFU, antibodies to protein tyrosine phosphatase are present at a level greater than or equal to 0.050
JDFU, antibodies to ZnT8 are present at a level greater than or equal to 0.021 JDFU, or combinations thereof.
234. The method of any one of claims 224-233, wherein the human patient has glucose levels greater than about 200 mg/dL, greater than about 300 mg/dL, greater than about 400 mg/dL, or greater than about 450 mg/dL.
235. The method of any one of claims 224-234, wherein the human patient has glucose levels greater than about 300 mg/dL.
236. The non-human animal of any one of claims 224-235, wherein the human patient has glucose levels greater than about 400 mg/dL.
237. The non-human animal of any one of claims 224-236, wherein the human patient has glucose levels greater than about 450 mg/dL.
238. A method of monitoring a human patient treated with a pharmaceutical composition comprising a T1D therapeutic made in any one of claims 199, 205, 208, or 209, comprising assessing the effect of the T1D therapeutic on glucose levels.
239. The non-human animal of any one of claims 75-81 or the method of any one of claims 146-152, 181-186, or 191, wherein the hypoimmune islet cells are derived from induced pluripotent stem cells (iPSCs), and wherein one or more of the modifications present in the hypoimmune islet cells are also present in the iPSCs.
240. The non-human animal of any one of claims 75-81 or the method of any one of claims 146-152, 181-186, or 191, wherein the hypoimmune islet cells are derived from induced
pluripotent stem cells (iPSCs), and wherein none of the modifications present in the hypoimmune islet cells are also present in the iPSCs.
241. The method of any one of claims 177-238, wherein one or more of the immune cells comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
242. The method of any one of claims 177-238, wherein one or more of the immune cells are isolated from peripheral blood mononuclear cells (PBMCs).
243. The method of claims 242, wherein one or more of the PBMCs comprise T cells, B cells, natural killer (NK) cells, natural killer T (NKT) cells, monocytes, macrophages, or dendritic cells.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363437976P | 2023-01-09 | 2023-01-09 | |
| US63/437,976 | 2023-01-09 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2024151541A1 true WO2024151541A1 (en) | 2024-07-18 |
Family
ID=89941138
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/010713 Ceased WO2024151541A1 (en) | 2023-01-09 | 2024-01-08 | Type-1 diabetes autoimmune mouse |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2024151541A1 (en) |
Citations (106)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2211504A (en) | 1987-10-23 | 1989-07-05 | Nat Res Dev | Fowlpox virus promoters |
| US5420032A (en) | 1991-12-23 | 1995-05-30 | Universitge Laval | Homing endonuclease which originates from chlamydomonas eugametos and recognizes and cleaves a 15, 17 or 19 degenerate double stranded nucleotide sequence |
| WO1997015664A1 (en) | 1995-10-24 | 1997-05-01 | Dr. Karl Thomae Gmbh | Intensive homologous promoter obtained from hamsters |
| WO1998011244A2 (en) | 1996-09-11 | 1998-03-19 | The Government Of The United States Of America, Represented By The Secretary, Department Of Health And Human Services | Aav4 vector and uses thereof |
| WO1998053060A1 (en) | 1997-05-23 | 1998-11-26 | Gendaq Limited | Nucleic acid binding proteins |
| WO1998053059A1 (en) | 1997-05-23 | 1998-11-26 | Medical Research Council | Nucleic acid binding proteins |
| US5994136A (en) | 1997-12-12 | 1999-11-30 | Cell Genesys, Inc. | Method and means for producing high titer, safe, recombinant lentivirus vectors |
| US6013516A (en) | 1995-10-06 | 2000-01-11 | The Salk Institute For Biological Studies | Vector and method of use for nucleic acid delivery to non-dividing cells |
| US6060273A (en) | 1992-08-27 | 2000-05-09 | Beiersdorf Ag | Multicistronic expression units and their use |
| US6140081A (en) | 1998-10-16 | 2000-10-31 | The Scripps Research Institute | Zinc finger binding domains for GNN |
| US6156303A (en) | 1997-06-11 | 2000-12-05 | University Of Washington | Adeno-associated virus (AAV) isolates and AAV vectors derived therefrom |
| WO2002016536A1 (en) | 2000-08-23 | 2002-02-28 | Kao Corporation | Bactericidal antifouling detergent for hard surface |
| US6453242B1 (en) | 1999-01-12 | 2002-09-17 | Sangamo Biosciences, Inc. | Selection of sites for targeting by zinc finger proteins and methods of designing zinc finger proteins to bind to preselected sites |
| US6503717B2 (en) | 1999-12-06 | 2003-01-07 | Sangamo Biosciences, Inc. | Methods of using randomized libraries of zinc finger proteins for the identification of gene function |
| WO2003016496A2 (en) | 2001-08-20 | 2003-02-27 | The Scripps Research Institute | Zinc finger binding domains for cnn |
| US6534261B1 (en) | 1999-01-12 | 2003-03-18 | Sangamo Biosciences, Inc. | Regulation of endogenous gene expression in cells using zinc finger proteins |
| US20030138772A1 (en) | 2001-11-13 | 2003-07-24 | Guangping Gao | Method of detecting and/or identifying adeno-associated virus (AAV) sequences and isolating novel sequences identified thereby |
| US6599692B1 (en) | 1999-09-14 | 2003-07-29 | Sangamo Bioscience, Inc. | Functional genomics using zinc finger proteins |
| US6689558B2 (en) | 2000-02-08 | 2004-02-10 | Sangamo Biosciences, Inc. | Cells for drug discovery |
| US6794136B1 (en) | 2000-11-20 | 2004-09-21 | Sangamo Biosciences, Inc. | Iterative optimization in the design of binding proteins |
| US6808905B2 (en) | 2001-05-14 | 2004-10-26 | Cell Genesys, Inc. | Lentiviral vectors encoding clotting factors for gene therapy |
| US6833252B1 (en) | 1992-05-05 | 2004-12-21 | Institut Pasteur | Nucleotide sequence encoding the enzyme I-SecI and the uses thereof |
| US20050064474A1 (en) | 2003-08-08 | 2005-03-24 | Sangamo Biosciences, Inc. | Methods and compositions for targeted cleavage and recombination |
| WO2005033321A2 (en) | 2003-09-30 | 2005-04-14 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (aav) clades, sequences, vectors containing same, and uses therefor |
| US20050267061A1 (en) | 2004-04-08 | 2005-12-01 | Sangamo Biosciences, Inc. | Methods and compositions for treating neuropathic and neurodegenerative conditions |
| US7030215B2 (en) | 1999-03-24 | 2006-04-18 | Sangamo Biosciences, Inc. | Position dependent recognition of GNN nucleotide triplets by zinc fingers |
| US7067317B2 (en) | 2000-12-07 | 2006-06-27 | Sangamo Biosciences, Inc. | Regulation of angiogenesis with zinc finger proteins |
| US7070934B2 (en) | 1999-01-12 | 2006-07-04 | Sangamo Biosciences, Inc. | Ligand-controlled regulation of endogenous gene expression |
| US7198951B2 (en) | 2001-12-17 | 2007-04-03 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (AAV) serotype 9 sequences, vectors containing same, and uses therefor |
| US20070116690A1 (en) | 2001-12-10 | 2007-05-24 | Lili Yang | Method for the generation of antigen-specific lymphocytes |
| US20070117128A1 (en) | 2005-10-18 | 2007-05-24 | Smith James J | Rationally-designed meganucleases with altered sequence specificity and DNA-binding affinity |
| US7253273B2 (en) | 2004-04-08 | 2007-08-07 | Sangamo Biosciences, Inc. | Treatment of neuropathic pain with zinc finger proteins |
| US7262054B2 (en) | 2002-01-22 | 2007-08-28 | Sangamo Biosciences, Inc. | Zinc finger proteins for DNA binding and gene regulation in plants |
| US20070218528A1 (en) | 2004-02-05 | 2007-09-20 | Sangamo Biosciences, Inc. | Methods and compositions for targeted cleavage and recombination |
| US7361635B2 (en) | 2002-08-29 | 2008-04-22 | Sangamo Biosciences, Inc. | Simultaneous modulation of multiple genes |
| US7510876B2 (en) | 2003-12-23 | 2009-03-31 | Cythera, Inc. | Definitive endoderm |
| US7575924B2 (en) | 2000-11-13 | 2009-08-18 | Research Development Foundation | Methods and compositions relating to improved lentiviral vectors and their applications |
| US7629153B2 (en) | 2001-08-02 | 2009-12-08 | Research Development Foundation | Methods and compositions relating to improved lentiviral vector production systems |
| US20110301073A1 (en) | 2010-05-17 | 2011-12-08 | Sangamo Biosciences, Inc. | Novel DNA-binding proteins and uses thereof |
| US8076106B2 (en) | 2007-03-02 | 2011-12-13 | Richmond Chemical Corporation | Method to increase the yield and improve purification of products from transaminase reactions |
| US8129182B2 (en) | 2006-03-02 | 2012-03-06 | Viacyte, Inc. | Endocrine precursor cells, pancreatic hormone-expressing cells and methods of production |
| US20120135519A1 (en) | 2009-05-29 | 2012-05-31 | Cellartis Ab | INDUCED DERIVATION OF SPECIFIC ENDODERM FROM hPS CELL-DERIVED DEFINITIVE ENDODERM |
| WO2012079000A1 (en) | 2010-12-09 | 2012-06-14 | The Trustees Of The University Of Pennsylvania | Use of chimeric antigen receptor-modified t cells to treat cancer |
| US8216836B2 (en) | 2003-12-23 | 2012-07-10 | Viacyte, Inc. | Methods for identifying factors for differentiating definitive endoderm |
| WO2012138927A2 (en) | 2011-04-05 | 2012-10-11 | Philippe Duchateau | Method for the generation of compact tale-nucleases and uses thereof |
| US8420104B2 (en) | 2007-08-03 | 2013-04-16 | Institut Pasteur | Lentiviral gene transfer vectors and their medicinal applications |
| US8507274B2 (en) | 2009-02-06 | 2013-08-13 | President And Fellows Of Harvard College | Compositions and methods for promoting the generation of definitive endoderm |
| US20130253040A1 (en) | 2012-02-29 | 2013-09-26 | c/o Sangamo BioSciences, Inc. | Methods and compositions for treating huntington's disease |
| US8633024B2 (en) | 2004-04-27 | 2014-01-21 | Viacyte, Inc. | PDX1 expressing endoderm |
| WO2014014422A1 (en) | 2012-07-20 | 2014-01-23 | National University Of Singapore | Combinatoric encoding methods for microarrays |
| US8647873B2 (en) | 2004-04-27 | 2014-02-11 | Viacyte, Inc. | PDX1 expressing endoderm |
| US8748169B2 (en) | 2001-10-02 | 2014-06-10 | Research Development Foundation | Methods and compositions relating to restricted expression lentiviral vectors and their applications |
| US20140359799A1 (en) | 2011-12-23 | 2014-12-04 | Case Western Reserve University | Targeted gene modification using hybrid recombinant adeno-associated virus |
| WO2014201167A1 (en) * | 2013-06-11 | 2014-12-18 | President And Fellows Of Harvard College | SC-β CELLS AND COMPOSITIONS AND METHODS FOR GENERATING THE SAME |
| US9012218B2 (en) | 2008-10-31 | 2015-04-21 | Janssen Biotech, Inc. | Differentiation of human embryonic stem cells |
| US20150159173A1 (en) | 2005-04-07 | 2015-06-11 | The Trustees Of The University Of Pennsylvania | Method of increasing the function of an aav vector |
| US9062290B2 (en) | 2007-11-27 | 2015-06-23 | Lifescan, Inc. | Differentiation of human embryonic stem cells |
| US9068199B2 (en) | 2002-12-13 | 2015-06-30 | Bluebird Bio, Inc. | Therapeutic retroviral vectors for gene therapy |
| US9109245B2 (en) | 2009-04-22 | 2015-08-18 | Viacyte, Inc. | Cell compositions derived from dedifferentiated reprogrammed cells |
| US9157062B2 (en) | 2008-04-22 | 2015-10-13 | President And Fellows Of Harvard College | Compositions and methods for promoting the generation of PDX1+ pancreatic cells |
| US9228207B2 (en) | 2013-09-06 | 2016-01-05 | President And Fellows Of Harvard College | Switchable gRNAs comprising aptamers |
| US9328331B2 (en) | 2009-05-21 | 2016-05-03 | Joslin Diabetes Center, Inc. | Compositions and methods for promoting beta cell maturity |
| WO2016100925A1 (en) | 2014-12-18 | 2016-06-23 | President And Fellows Of Harvard College | METHODS FOR GENERATING AUGMENTED STEM CELL-DERIVED β CELLS AND USES THEREOF |
| US9404086B2 (en) | 2014-03-26 | 2016-08-02 | Ge Healthcare Uk Limited | Method for cell differentiation |
| WO2016183041A2 (en) | 2015-05-08 | 2016-11-17 | President And Fellows Of Harvard College | Universal donor stem cells and related methods |
| US9499795B2 (en) | 2005-10-27 | 2016-11-22 | Viacyte, Inc. | PDX1-expressing dorsal and ventral foregut endoderm |
| US9650610B2 (en) | 2013-03-14 | 2017-05-16 | Viacyte, Inc. | In vitro differentiation of pluripotent stem cells to pancreatic endoderm cells (PEC) and endocrine cells |
| US9683215B2 (en) | 2008-08-22 | 2017-06-20 | President And Fellows Of Harvard College | Methods of reprogramming cells |
| US9725699B2 (en) | 2006-04-28 | 2017-08-08 | Lifescan, Inc. | Differentiation of human embryonic stem cells |
| US9993491B2 (en) | 2009-06-12 | 2018-06-12 | Socpra—Sciences Et Génie S.E.C. | Guanine riboswitch binding compounds and their use as antibiotics |
| WO2018132783A1 (en) | 2017-01-13 | 2018-07-19 | The Regents Of The University Of California | Immunoengineered pluripotent cells |
| US10138465B2 (en) | 2012-12-31 | 2018-11-27 | Janssen Biotech, Inc. | Differentiation of human embryonic stem cells into pancreatic endocrine cells using HB9 regulators |
| WO2019018818A1 (en) | 2017-07-21 | 2019-01-24 | Semma Therapeutics, Inc. | Re-aggregation of stem cell-derived pancreatic beta cells |
| US10190096B2 (en) | 2014-12-18 | 2019-01-29 | President And Fellows Of Harvard College | Methods for generating stem cell-derived β cells and uses thereof |
| US20190085295A1 (en) | 2016-02-24 | 2019-03-21 | Novo Nordisk A/S | Generation of Functional Beta Cells From Human Pluripotent Stem Cell-Derived Endocrine Progenitors |
| US10253298B2 (en) | 2014-12-18 | 2019-04-09 | President And Fellows Of Harvard College | Methods for generating stem cell-derived beta cells and methods of use thereof |
| US20190153394A1 (en) | 2012-09-03 | 2019-05-23 | Novo Nordisk A/S | Generation of pancreatic endoderm from pluripotent stem cells using small molecules |
| WO2019126709A1 (en) | 2017-12-22 | 2019-06-27 | The Broad Institute, Inc. | Cas12b systems, methods, and compositions for targeted dna base editing |
| US10358628B2 (en) | 2011-12-22 | 2019-07-23 | Janssen Biotech, Inc. | Differentiation of human embryonic stem cells into single hormonal insulin positive cells |
| US10421989B2 (en) | 2014-02-26 | 2019-09-24 | Cornell University | Methods and reagents for riboswitch analysis |
| US10443042B2 (en) | 2014-12-18 | 2019-10-15 | President And Fellows Of Harvard College | Serum-free in vitro directed differentiation protocol for generating stem cell-derived beta cells and uses thereof |
| WO2019217493A2 (en) | 2018-05-07 | 2019-11-14 | President And Fellows Of Harvard College | Cell populations and gene expression associated with in vitro beta cell differentiation |
| US10487313B2 (en) | 2011-06-21 | 2019-11-26 | Novo Nordisk A/S | Efficient induction of definitive endoderm from pluripotent stem cells |
| US20190359943A1 (en) | 2006-03-02 | 2019-11-28 | Viacyte, Inc. | Endocrine precursor cells, pancreatic hormone-expressing cells and methods of production |
| US20200002670A1 (en) | 2013-08-30 | 2020-01-02 | Novo Nordisk A/S | Generation of Endocrine Progenitor Cells from Human Pluripotent Stem Cells Using Small Molecules |
| WO2020018615A2 (en) | 2018-07-17 | 2020-01-23 | The Regents Of The University Of California | Cells differentiated from immunoengineered pluripotent cells |
| US20200248169A1 (en) | 2017-06-26 | 2020-08-06 | The Broad Institute, Inc. | Crispr/cas-cytidine deaminase based compositions, systems, and methods for targeted nucleic acid editing |
| WO2020181202A1 (en) | 2019-03-06 | 2020-09-10 | The Broad Institute, Inc. | A:t to t:a base editing through adenine deamination and oxidation |
| WO2020181193A1 (en) | 2019-03-06 | 2020-09-10 | The Broad Institute, Inc. | T:a to a:t base editing through adenosine methylation |
| WO2020181178A1 (en) | 2019-03-06 | 2020-09-10 | The Broad Institute, Inc. | T:a to a:t base editing through thymine alkylation |
| WO2020181195A1 (en) | 2019-03-06 | 2020-09-10 | The Broad Institute, Inc. | T:a to a:t base editing through adenine excision |
| WO2020191248A1 (en) | 2019-03-19 | 2020-09-24 | The Broad Institute, Inc. | Method and compositions for editing nucleotide sequences |
| US20200308548A1 (en) | 2016-07-01 | 2020-10-01 | Centre National De La Recherche Scientifique (Cnrs) | Amplifying Beta Cell Differentiation with Small Molecules BET (Bromodomain And Extraterminal Family Of Bromodomain-Containing Proteins) Inhibitors |
| WO2020207998A1 (en) | 2019-04-08 | 2020-10-15 | Novo Nordisk A/S | Generation of pancreatic endoderm from stem cell derived definitive endoderm |
| WO2020214842A1 (en) | 2019-04-17 | 2020-10-22 | The Broad Institute, Inc. | Adenine base editors with reduced off-target effects |
| US20210060083A1 (en) | 2014-12-18 | 2021-03-04 | President And Fellows Of Harvard College | Methods for generating stem cell-derived beta cells and uses thereof |
| US20210071163A1 (en) | 2017-12-22 | 2021-03-11 | The Broad Institute, Inc. | Cas12b systems, methods, and compositions for targeted rna base editing |
| US20210079366A1 (en) | 2017-12-22 | 2021-03-18 | The Broad Institute, Inc. | Cas12a systems, methods, and compositions for targeted rna base editing |
| US20210093667A1 (en) | 2017-06-26 | 2021-04-01 | The Broad Institute, Inc. | Crispr/cas-adenine deaminase based compositions, systems, and methods for targeted nucleic acid editing |
| US20210207099A1 (en) | 2018-05-16 | 2021-07-08 | Washington University | Methods and compositions for generating cells of endodermal lineage and beta cells and uses thereof |
| WO2021146627A1 (en) | 2020-01-17 | 2021-07-22 | Sana Biotechnology, Inc. | Safety switches for regulation of gene expression |
| US20210230554A1 (en) | 2018-08-30 | 2021-07-29 | Novo Nordisk A/S | Generation of functional beta cells from human pluripotent stem cell-derived endocrine progenitors |
| WO2021158921A2 (en) | 2020-02-05 | 2021-08-12 | The Broad Institute, Inc. | Adenine base editors and uses thereof |
| WO2021226558A1 (en) | 2020-05-08 | 2021-11-11 | The Broad Institute, Inc. | Methods and compositions for simultaneous editing of both strands of a target double-stranded nucleotide sequence |
| WO2022067130A2 (en) | 2020-09-24 | 2022-03-31 | The Broad Institute, Inc. | Prime editing guide rnas, compositions thereof, and methods of using the same |
| US20220127622A1 (en) | 2018-09-07 | 2022-04-28 | Beam Therapeutics Inc. | Compositions and Methods for Improving Base Editing |
-
2024
- 2024-01-08 WO PCT/US2024/010713 patent/WO2024151541A1/en not_active Ceased
Patent Citations (126)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB2211504A (en) | 1987-10-23 | 1989-07-05 | Nat Res Dev | Fowlpox virus promoters |
| US5420032A (en) | 1991-12-23 | 1995-05-30 | Universitge Laval | Homing endonuclease which originates from chlamydomonas eugametos and recognizes and cleaves a 15, 17 or 19 degenerate double stranded nucleotide sequence |
| US6833252B1 (en) | 1992-05-05 | 2004-12-21 | Institut Pasteur | Nucleotide sequence encoding the enzyme I-SecI and the uses thereof |
| US6060273A (en) | 1992-08-27 | 2000-05-09 | Beiersdorf Ag | Multicistronic expression units and their use |
| US6013516A (en) | 1995-10-06 | 2000-01-11 | The Salk Institute For Biological Studies | Vector and method of use for nucleic acid delivery to non-dividing cells |
| WO1997015664A1 (en) | 1995-10-24 | 1997-05-01 | Dr. Karl Thomae Gmbh | Intensive homologous promoter obtained from hamsters |
| WO1998011244A2 (en) | 1996-09-11 | 1998-03-19 | The Government Of The United States Of America, Represented By The Secretary, Department Of Health And Human Services | Aav4 vector and uses thereof |
| WO1998053058A1 (en) | 1997-05-23 | 1998-11-26 | Gendaq Limited | Nucleic acid binding proteins |
| WO1998053059A1 (en) | 1997-05-23 | 1998-11-26 | Medical Research Council | Nucleic acid binding proteins |
| WO1998053060A1 (en) | 1997-05-23 | 1998-11-26 | Gendaq Limited | Nucleic acid binding proteins |
| US6156303A (en) | 1997-06-11 | 2000-12-05 | University Of Washington | Adeno-associated virus (AAV) isolates and AAV vectors derived therefrom |
| US8846385B2 (en) | 1997-12-12 | 2014-09-30 | Gbp Ip, Llc | Method and means for producing high titer, safe recombinant lentivirus vectors |
| US5994136A (en) | 1997-12-12 | 1999-11-30 | Cell Genesys, Inc. | Method and means for producing high titer, safe, recombinant lentivirus vectors |
| US6140081A (en) | 1998-10-16 | 2000-10-31 | The Scripps Research Institute | Zinc finger binding domains for GNN |
| US6453242B1 (en) | 1999-01-12 | 2002-09-17 | Sangamo Biosciences, Inc. | Selection of sites for targeting by zinc finger proteins and methods of designing zinc finger proteins to bind to preselected sites |
| US6534261B1 (en) | 1999-01-12 | 2003-03-18 | Sangamo Biosciences, Inc. | Regulation of endogenous gene expression in cells using zinc finger proteins |
| US7070934B2 (en) | 1999-01-12 | 2006-07-04 | Sangamo Biosciences, Inc. | Ligand-controlled regulation of endogenous gene expression |
| US7030215B2 (en) | 1999-03-24 | 2006-04-18 | Sangamo Biosciences, Inc. | Position dependent recognition of GNN nucleotide triplets by zinc fingers |
| US6599692B1 (en) | 1999-09-14 | 2003-07-29 | Sangamo Bioscience, Inc. | Functional genomics using zinc finger proteins |
| US6503717B2 (en) | 1999-12-06 | 2003-01-07 | Sangamo Biosciences, Inc. | Methods of using randomized libraries of zinc finger proteins for the identification of gene function |
| US6689558B2 (en) | 2000-02-08 | 2004-02-10 | Sangamo Biosciences, Inc. | Cells for drug discovery |
| WO2002016536A1 (en) | 2000-08-23 | 2002-02-28 | Kao Corporation | Bactericidal antifouling detergent for hard surface |
| US7575924B2 (en) | 2000-11-13 | 2009-08-18 | Research Development Foundation | Methods and compositions relating to improved lentiviral vectors and their applications |
| US9023646B2 (en) | 2000-11-13 | 2015-05-05 | Research Development Foundation | Methods and compositions relating to improved lentiviral vectors and their applications |
| US8329462B2 (en) | 2000-11-13 | 2012-12-11 | Research Development Foundation | Methods and compositions relating to improved lentiviral vectors and their applications |
| US6794136B1 (en) | 2000-11-20 | 2004-09-21 | Sangamo Biosciences, Inc. | Iterative optimization in the design of binding proteins |
| US7067317B2 (en) | 2000-12-07 | 2006-06-27 | Sangamo Biosciences, Inc. | Regulation of angiogenesis with zinc finger proteins |
| US6808905B2 (en) | 2001-05-14 | 2004-10-26 | Cell Genesys, Inc. | Lentiviral vectors encoding clotting factors for gene therapy |
| US7745179B2 (en) | 2001-05-14 | 2010-06-29 | Gbp Ip, Llc | Lentiviral vectors featuring liver specific transcriptional enhancer and methods of using same |
| US7179903B2 (en) | 2001-05-14 | 2007-02-20 | Cell Genesys, Inc | Liver specific transcriptional enhancer |
| US9260725B2 (en) | 2001-08-02 | 2016-02-16 | Research Development Foundation | Methods and compositions relating to improved lentiviral vector production systems |
| US7629153B2 (en) | 2001-08-02 | 2009-12-08 | Research Development Foundation | Methods and compositions relating to improved lentiviral vector production systems |
| US8900858B2 (en) | 2001-08-02 | 2014-12-02 | Research Development Foundation | Methods and compositions relating to improved lentiviral vector production systems |
| WO2003016496A2 (en) | 2001-08-20 | 2003-02-27 | The Scripps Research Institute | Zinc finger binding domains for cnn |
| US8748169B2 (en) | 2001-10-02 | 2014-06-10 | Research Development Foundation | Methods and compositions relating to restricted expression lentiviral vectors and their applications |
| US20030138772A1 (en) | 2001-11-13 | 2003-07-24 | Guangping Gao | Method of detecting and/or identifying adeno-associated virus (AAV) sequences and isolating novel sequences identified thereby |
| US20070116690A1 (en) | 2001-12-10 | 2007-05-24 | Lili Yang | Method for the generation of antigen-specific lymphocytes |
| US7198951B2 (en) | 2001-12-17 | 2007-04-03 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (AAV) serotype 9 sequences, vectors containing same, and uses therefor |
| US7262054B2 (en) | 2002-01-22 | 2007-08-28 | Sangamo Biosciences, Inc. | Zinc finger proteins for DNA binding and gene regulation in plants |
| US7361635B2 (en) | 2002-08-29 | 2008-04-22 | Sangamo Biosciences, Inc. | Simultaneous modulation of multiple genes |
| US9068199B2 (en) | 2002-12-13 | 2015-06-30 | Bluebird Bio, Inc. | Therapeutic retroviral vectors for gene therapy |
| US20050064474A1 (en) | 2003-08-08 | 2005-03-24 | Sangamo Biosciences, Inc. | Methods and compositions for targeted cleavage and recombination |
| WO2005033321A2 (en) | 2003-09-30 | 2005-04-14 | The Trustees Of The University Of Pennsylvania | Adeno-associated virus (aav) clades, sequences, vectors containing same, and uses therefor |
| US7510876B2 (en) | 2003-12-23 | 2009-03-31 | Cythera, Inc. | Definitive endoderm |
| US8216836B2 (en) | 2003-12-23 | 2012-07-10 | Viacyte, Inc. | Methods for identifying factors for differentiating definitive endoderm |
| US10421942B2 (en) | 2003-12-23 | 2019-09-24 | Viacyte, Inc. | Definitive endoderm |
| US20070218528A1 (en) | 2004-02-05 | 2007-09-20 | Sangamo Biosciences, Inc. | Methods and compositions for targeted cleavage and recombination |
| US20050267061A1 (en) | 2004-04-08 | 2005-12-01 | Sangamo Biosciences, Inc. | Methods and compositions for treating neuropathic and neurodegenerative conditions |
| US7253273B2 (en) | 2004-04-08 | 2007-08-07 | Sangamo Biosciences, Inc. | Treatment of neuropathic pain with zinc finger proteins |
| US9222069B2 (en) | 2004-04-27 | 2015-12-29 | Viacyte, Inc. | Methods for making anterior foregut endoderm |
| US10465162B2 (en) | 2004-04-27 | 2019-11-05 | Viacyte, Inc. | Anterior endoderm cells and methods of production |
| US8633024B2 (en) | 2004-04-27 | 2014-01-21 | Viacyte, Inc. | PDX1 expressing endoderm |
| US8647873B2 (en) | 2004-04-27 | 2014-02-11 | Viacyte, Inc. | PDX1 expressing endoderm |
| US20150159173A1 (en) | 2005-04-07 | 2015-06-11 | The Trustees Of The University Of Pennsylvania | Method of increasing the function of an aav vector |
| US20070117128A1 (en) | 2005-10-18 | 2007-05-24 | Smith James J | Rationally-designed meganucleases with altered sequence specificity and DNA-binding affinity |
| US9499795B2 (en) | 2005-10-27 | 2016-11-22 | Viacyte, Inc. | PDX1-expressing dorsal and ventral foregut endoderm |
| US8603811B2 (en) | 2006-03-02 | 2013-12-10 | Viacyte, Inc. | Endocrine precursor cells, pancreatic hormone-expressing cells and methods of production |
| US8129182B2 (en) | 2006-03-02 | 2012-03-06 | Viacyte, Inc. | Endocrine precursor cells, pancreatic hormone-expressing cells and methods of production |
| US10370645B2 (en) | 2006-03-02 | 2019-08-06 | Emory University | Endocrine precursor cells, pancreatic hormone-expressing cells and methods of production |
| US20190359943A1 (en) | 2006-03-02 | 2019-11-28 | Viacyte, Inc. | Endocrine precursor cells, pancreatic hormone-expressing cells and methods of production |
| US9725699B2 (en) | 2006-04-28 | 2017-08-08 | Lifescan, Inc. | Differentiation of human embryonic stem cells |
| US8076106B2 (en) | 2007-03-02 | 2011-12-13 | Richmond Chemical Corporation | Method to increase the yield and improve purification of products from transaminase reactions |
| US8709799B2 (en) | 2007-08-03 | 2014-04-29 | Institut Pasteur | Lentiviral gene transfer vectors and their medicinal applications |
| US8420104B2 (en) | 2007-08-03 | 2013-04-16 | Institut Pasteur | Lentiviral gene transfer vectors and their medicinal applications |
| US10494609B2 (en) | 2007-11-27 | 2019-12-03 | Janssen Biotech, Inc. | Use of a TGF-βR-1 kinase to produce human pancreatic endocrine cells |
| US9062290B2 (en) | 2007-11-27 | 2015-06-23 | Lifescan, Inc. | Differentiation of human embryonic stem cells |
| US9157062B2 (en) | 2008-04-22 | 2015-10-13 | President And Fellows Of Harvard College | Compositions and methods for promoting the generation of PDX1+ pancreatic cells |
| US9683215B2 (en) | 2008-08-22 | 2017-06-20 | President And Fellows Of Harvard College | Methods of reprogramming cells |
| US9012218B2 (en) | 2008-10-31 | 2015-04-21 | Janssen Biotech, Inc. | Differentiation of human embryonic stem cells |
| US8927280B2 (en) | 2009-02-06 | 2015-01-06 | President And Fellows Of Harvard College | Compositions and methods for promoting the generation of definitive endoderm |
| US8507274B2 (en) | 2009-02-06 | 2013-08-13 | President And Fellows Of Harvard College | Compositions and methods for promoting the generation of definitive endoderm |
| US9982235B2 (en) | 2009-04-22 | 2018-05-29 | Viacyte, Inc. | Cell compositions derived from dedifferentiated reprogrammed cells |
| US9109245B2 (en) | 2009-04-22 | 2015-08-18 | Viacyte, Inc. | Cell compositions derived from dedifferentiated reprogrammed cells |
| US9988604B2 (en) | 2009-04-22 | 2018-06-05 | Viacyte, Inc. | Cell compositions derived from dedifferentiated reprogrammed cells |
| US9328331B2 (en) | 2009-05-21 | 2016-05-03 | Joslin Diabetes Center, Inc. | Compositions and methods for promoting beta cell maturity |
| US20120135519A1 (en) | 2009-05-29 | 2012-05-31 | Cellartis Ab | INDUCED DERIVATION OF SPECIFIC ENDODERM FROM hPS CELL-DERIVED DEFINITIVE ENDODERM |
| US9993491B2 (en) | 2009-06-12 | 2018-06-12 | Socpra—Sciences Et Génie S.E.C. | Guanine riboswitch binding compounds and their use as antibiotics |
| US20110301073A1 (en) | 2010-05-17 | 2011-12-08 | Sangamo Biosciences, Inc. | Novel DNA-binding proteins and uses thereof |
| WO2012079000A1 (en) | 2010-12-09 | 2012-06-14 | The Trustees Of The University Of Pennsylvania | Use of chimeric antigen receptor-modified t cells to treat cancer |
| WO2012138927A2 (en) | 2011-04-05 | 2012-10-11 | Philippe Duchateau | Method for the generation of compact tale-nucleases and uses thereof |
| US10487313B2 (en) | 2011-06-21 | 2019-11-26 | Novo Nordisk A/S | Efficient induction of definitive endoderm from pluripotent stem cells |
| US10358628B2 (en) | 2011-12-22 | 2019-07-23 | Janssen Biotech, Inc. | Differentiation of human embryonic stem cells into single hormonal insulin positive cells |
| US20140359799A1 (en) | 2011-12-23 | 2014-12-04 | Case Western Reserve University | Targeted gene modification using hybrid recombinant adeno-associated virus |
| US20130253040A1 (en) | 2012-02-29 | 2013-09-26 | c/o Sangamo BioSciences, Inc. | Methods and compositions for treating huntington's disease |
| WO2014014422A1 (en) | 2012-07-20 | 2014-01-23 | National University Of Singapore | Combinatoric encoding methods for microarrays |
| US20190153394A1 (en) | 2012-09-03 | 2019-05-23 | Novo Nordisk A/S | Generation of pancreatic endoderm from pluripotent stem cells using small molecules |
| US10138465B2 (en) | 2012-12-31 | 2018-11-27 | Janssen Biotech, Inc. | Differentiation of human embryonic stem cells into pancreatic endocrine cells using HB9 regulators |
| US20190211309A1 (en) | 2012-12-31 | 2019-07-11 | Janssen Biotech, Inc. | Differentiation of human embryonic stem cells into pancreatic endocrine cells using hb9 regulators |
| US9650610B2 (en) | 2013-03-14 | 2017-05-16 | Viacyte, Inc. | In vitro differentiation of pluripotent stem cells to pancreatic endoderm cells (PEC) and endocrine cells |
| US10030229B2 (en) | 2013-06-11 | 2018-07-24 | President And Fellows Of Harvard College | SC-β cells and compositions and methods for generating the same |
| WO2014201167A1 (en) * | 2013-06-11 | 2014-12-18 | President And Fellows Of Harvard College | SC-β CELLS AND COMPOSITIONS AND METHODS FOR GENERATING THE SAME |
| US20200002670A1 (en) | 2013-08-30 | 2020-01-02 | Novo Nordisk A/S | Generation of Endocrine Progenitor Cells from Human Pluripotent Stem Cells Using Small Molecules |
| US9228207B2 (en) | 2013-09-06 | 2016-01-05 | President And Fellows Of Harvard College | Switchable gRNAs comprising aptamers |
| US10421989B2 (en) | 2014-02-26 | 2019-09-24 | Cornell University | Methods and reagents for riboswitch analysis |
| US9404086B2 (en) | 2014-03-26 | 2016-08-02 | Ge Healthcare Uk Limited | Method for cell differentiation |
| US10443042B2 (en) | 2014-12-18 | 2019-10-15 | President And Fellows Of Harvard College | Serum-free in vitro directed differentiation protocol for generating stem cell-derived beta cells and uses thereof |
| US10253298B2 (en) | 2014-12-18 | 2019-04-09 | President And Fellows Of Harvard College | Methods for generating stem cell-derived beta cells and methods of use thereof |
| WO2016100925A1 (en) | 2014-12-18 | 2016-06-23 | President And Fellows Of Harvard College | METHODS FOR GENERATING AUGMENTED STEM CELL-DERIVED β CELLS AND USES THEREOF |
| US10190096B2 (en) | 2014-12-18 | 2019-01-29 | President And Fellows Of Harvard College | Methods for generating stem cell-derived β cells and uses thereof |
| US20210060083A1 (en) | 2014-12-18 | 2021-03-04 | President And Fellows Of Harvard College | Methods for generating stem cell-derived beta cells and uses thereof |
| WO2016183041A2 (en) | 2015-05-08 | 2016-11-17 | President And Fellows Of Harvard College | Universal donor stem cells and related methods |
| US20190085295A1 (en) | 2016-02-24 | 2019-03-21 | Novo Nordisk A/S | Generation of Functional Beta Cells From Human Pluripotent Stem Cell-Derived Endocrine Progenitors |
| US20200308548A1 (en) | 2016-07-01 | 2020-10-01 | Centre National De La Recherche Scientifique (Cnrs) | Amplifying Beta Cell Differentiation with Small Molecules BET (Bromodomain And Extraterminal Family Of Bromodomain-Containing Proteins) Inhibitors |
| WO2018132783A1 (en) | 2017-01-13 | 2018-07-19 | The Regents Of The University Of California | Immunoengineered pluripotent cells |
| US20210093667A1 (en) | 2017-06-26 | 2021-04-01 | The Broad Institute, Inc. | Crispr/cas-adenine deaminase based compositions, systems, and methods for targeted nucleic acid editing |
| US20200248169A1 (en) | 2017-06-26 | 2020-08-06 | The Broad Institute, Inc. | Crispr/cas-cytidine deaminase based compositions, systems, and methods for targeted nucleic acid editing |
| WO2019018818A1 (en) | 2017-07-21 | 2019-01-24 | Semma Therapeutics, Inc. | Re-aggregation of stem cell-derived pancreatic beta cells |
| US20210079366A1 (en) | 2017-12-22 | 2021-03-18 | The Broad Institute, Inc. | Cas12a systems, methods, and compositions for targeted rna base editing |
| WO2019126709A1 (en) | 2017-12-22 | 2019-06-27 | The Broad Institute, Inc. | Cas12b systems, methods, and compositions for targeted dna base editing |
| US20210071163A1 (en) | 2017-12-22 | 2021-03-11 | The Broad Institute, Inc. | Cas12b systems, methods, and compositions for targeted rna base editing |
| WO2019217493A2 (en) | 2018-05-07 | 2019-11-14 | President And Fellows Of Harvard College | Cell populations and gene expression associated with in vitro beta cell differentiation |
| US20210207099A1 (en) | 2018-05-16 | 2021-07-08 | Washington University | Methods and compositions for generating cells of endodermal lineage and beta cells and uses thereof |
| WO2020018615A2 (en) | 2018-07-17 | 2020-01-23 | The Regents Of The University Of California | Cells differentiated from immunoengineered pluripotent cells |
| US20210230554A1 (en) | 2018-08-30 | 2021-07-29 | Novo Nordisk A/S | Generation of functional beta cells from human pluripotent stem cell-derived endocrine progenitors |
| US20220127622A1 (en) | 2018-09-07 | 2022-04-28 | Beam Therapeutics Inc. | Compositions and Methods for Improving Base Editing |
| WO2020181202A1 (en) | 2019-03-06 | 2020-09-10 | The Broad Institute, Inc. | A:t to t:a base editing through adenine deamination and oxidation |
| WO2020181193A1 (en) | 2019-03-06 | 2020-09-10 | The Broad Institute, Inc. | T:a to a:t base editing through adenosine methylation |
| WO2020181195A1 (en) | 2019-03-06 | 2020-09-10 | The Broad Institute, Inc. | T:a to a:t base editing through adenine excision |
| WO2020181178A1 (en) | 2019-03-06 | 2020-09-10 | The Broad Institute, Inc. | T:a to a:t base editing through thymine alkylation |
| WO2020191248A1 (en) | 2019-03-19 | 2020-09-24 | The Broad Institute, Inc. | Method and compositions for editing nucleotide sequences |
| WO2020207998A1 (en) | 2019-04-08 | 2020-10-15 | Novo Nordisk A/S | Generation of pancreatic endoderm from stem cell derived definitive endoderm |
| WO2020214842A1 (en) | 2019-04-17 | 2020-10-22 | The Broad Institute, Inc. | Adenine base editors with reduced off-target effects |
| WO2021146627A1 (en) | 2020-01-17 | 2021-07-22 | Sana Biotechnology, Inc. | Safety switches for regulation of gene expression |
| WO2021158921A2 (en) | 2020-02-05 | 2021-08-12 | The Broad Institute, Inc. | Adenine base editors and uses thereof |
| WO2021226558A1 (en) | 2020-05-08 | 2021-11-11 | The Broad Institute, Inc. | Methods and compositions for simultaneous editing of both strands of a target double-stranded nucleotide sequence |
| WO2022067130A2 (en) | 2020-09-24 | 2022-03-31 | The Broad Institute, Inc. | Prime editing guide rnas, compositions thereof, and methods of using the same |
Non-Patent Citations (175)
| Title |
|---|
| "Methods and Protocols", 2013, SPRINGER, article "Methods in Molecular Biology: Pluripotent Stem Cells" |
| "NCBI", Database accession no. NM 001 127223. 1 |
| "UniProt", Database accession no. P13726 |
| "Uniprot", Database accession no. Pl 3987 |
| ABDELALIM ESSAM M: "Modeling different types of diabetes using human pluripotent stem cells", CMLS CELLULAR AND MOLECULAR LIFE SCIENCES, vol. 78, no. 6, 26 November 2020 (2020-11-26), pages 2459 - 2483, XP037390779, ISSN: 1420-682X, DOI: 10.1007/S00018-020-03710-9 * |
| AMOULD ET AL., J MOL BIOL., vol. 355, 2006, pages 443 - 458 |
| ANNE-MARIE AUBIN: "The NOD Mouse Beyond Autoimmune Diabetes", FRONTIERS IN IMMUNOLOGY, vol. 13, 29 April 2022 (2022-04-29), Lausanne, CH, XP093154794, ISSN: 1664-3224, Retrieved from the Internet <URL:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9102607/pdf/fimmu-13-874769.pdf> DOI: 10.3389/fimmu.2022.874769 * |
| ARGAST ET AL., J. MOL. BIOL., vol. 280, 1998, pages 345 - 353 |
| ASHWORTH ET AL., NATURE, vol. 441, 2006, pages 656 - 659 |
| AUERBACH, W. ET AL., BIOTECHNIQUES, vol. 29, no. 5, 2000, pages 1024 - 1028 |
| BALBOA DIEGO ET AL: "Concise Review: Human Pluripotent Stem Cells for the Modeling of Pancreatic [beta]-Cell Pathology", STEM CELLS, vol. 37, no. 1, 2 December 2018 (2018-12-02), pages 33 - 41, XP093082464, ISSN: 1066-5099, Retrieved from the Internet <URL:https://onlinelibrary.wiley.com/doi/full-xml/10.1002/stem.2913> DOI: 10.1002/stem.2913 * |
| BANIK ET AL., CHEMRXIV, 2019 |
| BARNARD, K.D. ET AL.: "Psychosocial aspects of closed- and open-loop insulin delivery: closing the loop in adults with Type 1 diabetes in the home setting", DIABET MED, vol. 32, 2015, pages 601 - 608, XP071687736, DOI: 10.1111/dme.12706 |
| BEERLI ET AL., NATURE BIOTECHNOL., vol. 20, 2002, pages 135 - 141 |
| BEKIARI, E. ET AL.: "Artificial pancreas treatment for outpatients with type 1 diabetes: systematic review and meta-analysis", BMJ, vol. 361, no. k1310, 2018 |
| BELFORT ET AL., NUCLEIC ACIDS RES., vol. 25, 1997, pages 3379 - 3388 |
| BEMDSENDENU, CURR OPIN STRUCT BIOL, vol. 18, no. 6, 2008, pages 682 - 689 |
| BERGSHI, SCIENCE, vol. 271, 1996, pages 1081 - 1085 |
| BITINAITE ET AL., PROC. NATL. ACAD. SCI. USA, vol. 95, 1998, pages 14623 - 10575 |
| BONGER ET AL., NAT CHEM BIOL, vol. 7, no. 8, 2011, pages 531 - 7 |
| BOUGHTON, C.K. ET AL.: "Hybrid closed-loop glucose control with faster insulin aspart compared with standard insulin aspart in adults with type 1 diabetes: A double-blind, multicentre, multinational, randomized, crossover study", DIABETES OBES METAB, vol. 23, 2021, pages 1389 - 1396 |
| BROOKS, A.M. ET AL.: "Attainment of metabolic goals in the integrated UK islet transplant program with locally isolated and transported preparations", AM J TRANSPLANT, vol. 13, 2013, pages 3236 - 3243, XP072345807, DOI: 10.1111/ajt.12469 |
| BURSLEM ET AL., CELL CHEMICAL BIOLOGY, vol. 25, 2018, pages 67 - 77 |
| BURSLEMCREWS, CHEM REV, vol. 117, 2017, pages 11269 - 11301 |
| CARROLL ET AL., GENETICS SOCIETY OF AMERICA, vol. 188, 2011, pages 773 - 782 |
| CERBINI ET AL., PLOS ONE, vol. 10, no. 1, 2015, pages e0116032 |
| CERMAK ET AL., NUCL. ACIDS RES., vol. 39, 2011, pages e82 |
| CHEM ET AL., PLANT CELL, vol. 8, 1996, pages 305 - 321 |
| CHEN ET AL., PROTEIN ENG DES SEL, vol. 22, 2009, pages 249 - 256 |
| CHETAN, MR ET AL.: "What is Type 1 diabetes?", MEDICINE, vol. 47, no. 1, January 2019 (2019-01-01), pages 5 - 9 |
| CHEVALIER ET AL., MOL. CELL, vol. 10, 2002, pages 895 - 905 |
| CHEVALIER ET AL., MOLEC. CELL, vol. 10, 2002, pages 895 - 905 |
| CHEVALIER, B. S.B. L. STODDARD, NUCLEIC ACIDS RES., vol. 29, no. 18, 2001, pages 3757 - 3774 |
| CHO ET AL., PLANT MOL BIOL, vol. 40, 1999, pages 419 - 429 |
| CHOO ET AL., CURR. OPIN. STRUCT. BIOL., vol. 10, 2000, pages 411 - 416 |
| CHOULIKA ET AL., MOL. CELL. BIOL., vol. 15, 1995, pages 1968 - 1973 |
| CHUNG ET AL., NAT CHEM BIOL, vol. 11, 2015, pages 713 - 720 |
| CLARKE, W.L. ET AL.: "Reduced awareness of hypoglycemia in adults with IDDM. A prospective study of hypoglycemic frequency and associated symptoms", DIABETES CARE, vol. 18, 1995, pages 517 - 522, XP008176692, DOI: 10.2337/diacare.18.4.517 |
| COHEN-TANNOUDJI ET AL., MOL. CELL. BIOL., vol. 18, 1998, pages 1444 - 1448 |
| COLLINGWOOD ET AL., J. MOL. ENDOCRINOL, vol. 23, 1999, pages 255 - 275 |
| CRONICAN ET AL., ACS CHEM BIOL., vol. 5, no. 8, 2010, pages 747 - 52 |
| DAI, C. ET AL.: "Tacrolimus- and sirolimus-induced human beta cell dysfunction is reversible and preventable", JCI INSIGHT, vol. 5, 2020 |
| DALE L GREINER ET AL: "Humanized mice for the study of type 1 and type 2 diabetes", ANNALS OF THE NEW YORK ACADEMY OF SCIENCES, NEW YORK ACADEMY OF SCIENCES, US, vol. 1245, no. 1, 29 December 2011 (2011-12-29), pages 55 - 58, XP071408075, ISSN: 0077-8923, DOI: 10.1111/J.1749-6632.2011.06318.X * |
| DANG ET AL., GENOME BIOLOGY, vol. 16, 2015, pages 280 |
| DE FELIPE, GENETIC VACCINES AND THER., vol. 2, 2004, pages 13 |
| DEFELIPE ET AL., TRAFFIC, vol. 5, 2004, pages 616 - 626 |
| DEUSE ET AL., NATURE BIOTECHNOLOGY, vol. 37, 2019, pages 252 - 258 |
| DIMEGLIO, L.A.EVANS-MOLINA, C.ORAM, R.A.: "Type 1 diabetes", LANCET, vol. 391, 2018, pages 2449 - 2462 |
| DONOHO ET AL., MOL. CELL. BIOL, vol. 18, 1998, pages 4070 - 4078 |
| DOYLEHUNT, NEUROREPORT, vol. 8, 1997, pages 2937 - 2942 |
| DOYON ET AL., J AM CHEM SOC, vol. 128, 2006, pages 2477 - 2484 |
| DOYON ET AL., NATURE METHODS, vol. 8, 2010, pages 74 - 79 |
| DUJON ET AL., GENE, vol. 82, 1989, pages 115 - 118 |
| ELLIS ET AL., NAT REV GASTROENTEROL HEPATOL., vol. 14, no. 10, October 2017 (2017-10-01), pages 612 - 628 |
| ENGREITZ ET AL., COLD SPRING HARB PERSPECT BIOL, vol. 11, no. 035386, 2019 |
| EPINAT ET AL., NUCLEIC ACIDS RES, vol. 31, 2003, pages 2952 - 2962 |
| EPINAT ET AL., NUCLEIC ACIDS RES., vol. 31, 2003, pages 2952 - 2962 |
| FATIMA WHITFIELD-LARRY: "HLA-A2-Matched Peripheral Blood Mononuclear Cells From Type 1 Diabetic Patients, but Not Nondiabetic Donors, Transfer Insulitis to NOD- scid /[gamma]cnull/HLA-A2 Transgenic Mice Concurrent With the Expansion of Islet-Specific CD8+ T cells", DIABETES, vol. 60, no. 6, 21 May 2011 (2011-05-21), pages 1726 - 1733, XP093154246, ISSN: 0012-1797, Retrieved from the Internet <URL:https://journals.org/diabetes/diabetes/article-pdf/60/6/1726/664151/1726.pdf> DOI: 10.2337/db10-1287 * |
| FESTING ET AL., MAMMALIAN GENOME, vol. 10, 1999, pages 836 |
| FUJIMOTO K ET AL: "Pdx1 and other factors that regulate pancreatic [beta]-cell survival", DIABETES, OBESITY AND METABOLISM, BLACKWELL SCIENCE, GB, vol. 11, 7 October 2009 (2009-10-07), pages 30 - 37, XP072208109, ISSN: 1462-8902, DOI: 10.1111/J.1463-1326.2009.01121.X * |
| G. ET AL.: "Juvenile Diabetes Research Foundation Continuous Glucose Monitoring Study", N ENGL J MED, vol. 359, 2008, pages 1464 - 1476 |
| GAJ ET AL., TRENDS IN BIOTECHNOLOGY, vol. 31, no. 7, 2013, pages 397 - 405 |
| GENNARO: "Remington: The science and practice of pharmacy", 2000, LIPPINCOTT, WILLIAMS & WILKINS |
| GOFF, GENES DEV., vol. 5, 1991, pages 298 - 309 |
| GOMEZ-OSPINA ET AL., NAT. COMM., vol. 10, no. 1, 2019, pages 4045 |
| GONG ET AL., PLANT MOL. BIOL., vol. 41, 1999, pages 33 - 44 |
| GREENAWAY ET AL., GENE, vol. 18, 1982, pages 355 - 360 |
| GROUP, U.K.H.S.: "Risk of hypoglycaemia in types 1 and 2 diabetes: effects of treatment modalities and their duration", DIABETOLOGIA, vol. 50, 2007, pages 1140 - 1147, XP019510552, DOI: 10.1007/s00125-007-0599-y |
| GUO ET AL., J. MOL. BIOL., vol. 200, 2010, pages 96 |
| HAFT ET AL., PLOS COMPUT BIOL., vol. 1, no. 6, 2005, pages e60 |
| HAGMANN ET AL., J. VIROL., vol. 72, no. 1 97, 1998, pages 5610 - 5618 |
| HAN ET AL., PROC NATL ACAD SCI USA, vol. 116, no. 21, 2019, pages 10441 - 10446 |
| HERING, B.J. ET AL.: "Phase 3 Trial of Transplantation of Human Islets in Type 1 Diabetes Complicated by Severe Hypoglycemia", DIABETES CARE, vol. 39, 2016, pages 1230 - 1240 |
| HIGDON, L.E.TAN, J.C.MALTZMAN, J.S.: "Infection, Rejection, and the Connection", TRANSPLANTATION, 2022 |
| HOCKEMEYER ET AL., NAT. BIOTECHNOL., vol. 29, 2011, pages 731 - 734 |
| HOCKEMEYER ET AL., NATURE BIOTECH., vol. 29, 2011, pages 135 - 136 |
| HOGREBE ET AL.: "Generation of insulin-producing pancreatic beta cells from multiple human stem cell lines", NAT. PROTOC., 2021 |
| HOGREBE ET AL.: "Targeting the cytoskeleton to direct pancreatic differentiation of human pluripotent stem cells", NAT. BIOTECHNOL., vol. 38, 2020, pages 460 - 470, XP037523977, DOI: 10.1038/s41587-020-0430-6 |
| HU XIAOMENG ET AL: "302-OR: Hypoimmune Human Primary Pancreatic Islets Survive and Function in Humanized Diabetic Mice", DIABETES, vol. 71, no. Supplement_1, June 2022 (2022-06-01), XP055979539, ISSN: 0012-1797, Retrieved from the Internet <URL:https://diabetesjournals.org/diabetes/article/71/Supplement_1/302-OR/145089/302-OR-Hypoimmune-Human-Primary-Pancreatic-Islets> DOI: 10.2337/db22-302-OR * |
| ISALAN ET AL., NATURE BIOTECHNOL., vol. 19, 2001, pages 656 - 660 |
| JACQUELINE V. SCHIESSER ET AL: "Derivation of Insulin-Producing Beta-Cells from Human Pluripotent Stem Cells", THE REVIEW OF DIABETIC STUDIES, vol. 11, no. 1, 2014, DE, pages 6 - 18, XP055506943, ISSN: 1613-6071, DOI: 10.1900/RDS.2014.11.6 * |
| JASIN, TRENDS GENET., vol. 12, 1996, pages 224 - 228 |
| JEFFREY R. MILLMAN ET AL: "Generation of stem cell-derived [beta]-cells from patients with type 1 diabetes", NATURE COMMUNICATIONS, VOL. 7, N. 1, 10 May 2016 (2016-05-10), XP055648467, Retrieved from the Internet <URL:https://www.nature.com/articles/ncomms11463.pdf> [retrieved on 20191203], DOI: 10.1038/ncomms11463 * |
| JINEK ET AL., SCIENCE, vol. 337, no. 6096, 2012, pages 816 - 821 |
| KALLUNKI ET AL., CELLS, vol. 796, 2019 |
| KANAK, M.A. ET AL.: "Inflammatory response in islet transplantation", INT J ENDOCRINOL, 2014, pages 451035 |
| KIM, J.H. ET AL.: "Influence of cyclosporine A on glomerular growth and the effect of mizoribine and losartan on cyclosporine nephrotoxicity in young rats", SCI REP, vol. 6, 2016, pages 22374 |
| KLOMPE ET AL., NATURE, vol. 571, 2019, pages 219 - 225 |
| KNOEPFLER ET AL., CELL, vol. 99, 1999, pages 447 - 450 |
| KODURI ET AL., PROC NATL ACAD SCI, vol. 116, no. 7, 2019, pages 2539 - 2544 |
| KOUSARIDES, CELL, vol. 128, 2007, pages 693 - 705 |
| LAFFEL, L.M. ET AL.: "Effect of Continuous Glucose Monitoring on Glycemic Control in Adolescents and Young Adults With Type 1 Diabetes: A Randomized Clinical Trial", JAMA, vol. 323, 2020, pages 2388 - 2396 |
| LAGUNAS-RANGEL FRANCISCO ALEJANDRO ET AL: "Triple drug therapy with GABA, sitagliptin, and omeprazole prevents type 1 diabetes onset and promotes its reversal in non-obese diabetic mice", FRONTIERS IN ENDOCRINOLOGY, vol. 13, 21 October 2022 (2022-10-21), CH, XP093045001, ISSN: 1664-2392, DOI: 10.3389/fendo.2022.1028114 * |
| LEE ET AL., BIOTECHNOL BIOENG, vol. 10996, 2012, pages 1551 - 1560 |
| LEMON ET AL., CURR. OPIN. GENET. DEV., vol. 9, 1999, pages 499 - 504 |
| LEMOS, J.R.N. ET AL.: "Survival After Islet Transplantation in Patients With Type 1 Diabetes: Twenty-Year Follow-Up", DIABETES CARE, vol. 44, 2021, pages e67 - e68 |
| LI ET AL., NAT. METHODS, vol. 16, 2019, pages 866 - 869 |
| LIN, Y.K.FISHER, S.J.POP-BUSUI, R.: "Hypoglycemia unawareness and autonomic dysfunction in diabetes: Lessons learned and roles of diabetes technologies", J DIABETES INVESTIG, vol. 11, 2020, pages 1388 - 1402 |
| LIU ET AL., BIOINFORMATICS, vol. 24, 2008, pages 1850 - 1857 |
| LIU ET AL., CANCER GENE THER., vol. 5, 1998, pages 3 - 28 |
| LIVINGSTONE, S.J. ET AL.: "Estimated life expectancy in a Scottish cohort with type 1 diabetes", JAMA, vol. 313, 2008, pages 37 - 44 |
| LLOYD ET AL., FRONTIERS IN IMMUNOLOGY, vol. 4, no. 221, 2013, pages 1 - 7 |
| MAHMOUD IZADI: "Mesenchymal stem cell transplantation in newly diagnosed type-1 diabetes patients: a phase I/II randomized placebo-controlled clinical trial", STEM CELL RESEARCH & THERAPY, vol. 13, no. 1, December 2022 (2022-12-01), London, UK, XP093154607, ISSN: 1757-6512, Retrieved from the Internet <URL:https://www.ncbi.nlm.nih.gov/pmc/articles/PMC9208234/pdf/13287_2022_Article_2941.pdf> DOI: 10.1186/s13287-022-02941-w * |
| MALIK ET AL., TRENDS BIOCHEM. SCI., vol. 25, 2000, pages 277 - 283 |
| MANDAL ET AL., CELL STEM CELL, vol. 15, 2014, pages 643 - 652 |
| MANTEUFFEL-CYMBOROWSKA, ACTA BIOCHIM. POL., vol. 46, 1999, pages 77 - 89 |
| MAPP ET AL., PROC. NATL. ACAD. SCI. USA, vol. 97, 2000, pages 3930 - 3935 |
| MARFIL-GARZA, B.A. ET AL.: "Pancreatic islet transplantation in type 1 diabetes: 20-year experience from a single-centre cohort in Canada", LANCET DIABETES ENDOCRINOL, vol. 10, 2022, pages 519 - 532 |
| MARKMANN, J.F. ET AL.: "Phase 3 trial of human islet-after-kidney transplantation in type 1 diabetes", AM J TRANSPLANT, vol. 21, 2021, pages 1477 - 1492 |
| MCKENNA ET AL., J. STEROID BIOCHEM. MOL. BIOL., vol. 69, 1999, pages 3 - 12 |
| MILLMAN, J.R. ET AL.: "Generation of stem cell-derived beta-cells from patients with type 1 diabetes", NAT COMMUN, vol. 7, 2016, pages 11463 |
| MOLINARI ET AL., EMBO J., vol. 18, 1999, pages 6439 - 6447 |
| MORSUT ET AL., CELL, vol. 164, 2016, pages 780 - 791 |
| MOSCOU ET AL., SCIENCE, vol. 326, 2009, pages 3501 - 1512 |
| MOTTAMAL ET AL., MOLECULES, vol. 20, no. 3, 2015, pages 3898 - 3941 |
| MURARO ET AL., CELL SYST., vol. 3, no. 4, 26 October 2016 (2016-10-26), pages 385 - 394 |
| NAESENS, M.KUYPERS, D.R.SARWAL, M.: "Calcineurin inhibitor nephrotoxicity", CLIN J AM SOC NEPHROL, vol. 4, 2009, pages 481 - 508 |
| NALDINI L ET AL., CURR. OPIN. BIOTECHNOL, vol. 9, 1998, pages 457 - 463 |
| NATSUMEKANEMAKI, ANNU REV GENET, vol. 51, 2017, pages 82 - 102 |
| NAZIRUDDIN, B. ET AL.: "Evidence for instant blood-mediated inflammatory reaction in clinical autologous islet transplantation", AM J TRANSPLANT, vol. 14, 2014, pages 428 - 437 |
| NICOLE C. WALSH ET AL: "Humanized Mouse Models of Clinical Disease", ANNUAL REVIEW OF PATHOLOGY: MECHANISMS OF DISEASE, vol. 12, no. 1, 24 January 2017 (2017-01-24), US, pages 187 - 215, XP055519301, ISSN: 1553-4006, DOI: 10.1146/annurev-pathol-052016-100332 * |
| OGAWA ET AL., GENE, vol. 245, 2000, pages 21 - 29 |
| OKANAMI ET AL., GENES CELLS, vol. 1, 1996, pages 87 - 99 |
| PABO ET AL., ANN. REV. BIOCHEM., vol. 70, 2001, pages 313 - 340 |
| PAGLIUCA ET AL., CELL, vol. 159, no. 2, 2014, pages 428 - 39 |
| PAQUES ET AL., CURRENT GENE THERAPY, vol. 7, 2007, pages 49 - 66 |
| PEDERSEN-BJERGAARD, U. ET AL.: "Severe hypoglycaemia in 1076 adult patients with type 1 diabetes: influence of risk markers and selection", DIABETES METAB RES REV, vol. 20, 2004, pages 479 - 486, XP072260755, DOI: 10.1002/dmrr.482 |
| PERLER ET AL., NUCLEIC ACIDS RES., vol. 22, 1994, pages 1125 - 1127 |
| PIERS ET AL., NATURE, vol. 273, 1978, pages 113 - 120 |
| PUCHTA ET AL., NUCLEIC ACIDS RES., vol. 21, 1993, pages 5034 - 5040 |
| PUCHTA ET AL., PROC. NATL. ACAD. SCI. USA, vol. 93, 1996, pages 5055 - 5060 |
| PULICHERLA ET AL., MOLECULAR THERAPY, vol. 19, no. 6, 2011, pages 1070 - 1078 |
| RAMA, I.GRINYO, J.M.: "Malignancy after renal transplantation: the role of immunosuppression", NAT REV NEPHROL, vol. 6, 2010, pages 511 - 519 |
| RAN ET AL., NATURE, vol. 520, 2015, pages 186 - 191 |
| RELANDER ET AL., MOL THER., vol. 11, 2005, pages 452 - 459 |
| ROBERTSON ET AL., NATURE GENET., vol. 25, 2000, pages 338 - 342 |
| ROBYR ET AL., MOL. ENDOCRINOL., vol. 14, 2000, pages 329 - 347 |
| ROTH ET AL., CELLULAR MOLECULAR LIFE SCIENCES, vol. 76, no. 14, 2019, pages 2761 - 2777 |
| ROUET ET AL., MOL. CELL. BIOL., vol. 14, 1994, pages 8096 - 8106 |
| RUEDY, K.J.PARKIN, C.G.RIDDLESWORTH, T.D.GRAHAM, C.GROUP, D.S.: "Continuous Glucose Monitoring in Older Adults With Type 1 and Type 2 Diabetes Using Multiple Daily Injections of Insulin: Results From the DIAMOND Trial", J DIABETES SCI TECHNOL, vol. 11, 2017, pages 1138 - 1146 |
| SAMBROOK ET AL.: "Molecular Cloning: A Laboratory Manual", 2001, COLD SPRING HARBOR LABORATORY |
| SANDERJOUNG, NATURE BIOTECHNOLOGY, vol. 32, no. 4, 2014, pages 347 - 355 |
| SANJANA ET AL., NAT. METHODS, vol. 11, 2014, pages 783 - 4, Retrieved from the Internet <URL:www.e-crisp.org/E-CRISP/;crispr.mit.edu> |
| SARGENT ET AL., MOL. CELL. BIOL., vol. 17, 1997, pages 267 - 77 |
| SCOTT J BANUELOS: "Rejection of human islets and human HLA-A2.1 transgenic mouse islets by alloreactive human lymphocytes in immunodeficient NOD-scid and NOD-Rag1nullPrf1null mice", CLINICAL IMMUNOLOGY, vol. 112, no. 3, September 2004 (2004-09-01), AMSTERDAM, NL, pages 273 - 283, XP093154278, ISSN: 1521-6616, DOI: 10.1016/j.clim.2004.04.006 * |
| SEELIGER ET AL., PLOS ONE, vol. 7, no. 1, 2012, pages e29266 |
| SEGAL, CURR. OPIN. BIOTECHNOL., vol. 12, 2001, pages 632 - 637 |
| SEIPEL ET AL., EMBOJ., vol. 11, 1992, pages 4961 - 4968 |
| SEKI ET AL., WORLD J. STEM CELLS, vol. 7, no. 1, 2015, pages 116 - 125 |
| SELIGMAN ET AL., NUCLEIC ACIDS RES, vol. 30, 2002, pages 3870 - 3879 |
| SERA ET AL., BIOCHEMISTRY, vol. 41, 2002, pages 7074 - 7081 |
| SILVA ET AL., CURRENT GENE THERAPY, vol. 11, 2011, pages 11 - 27 |
| SILVA ET AL., J MOL. BIOL., vol. 361, 2006, pages 744 - 754 |
| SIMS, E.K.CARR, A.L.J.ORAM, R.A.DIMEGLIO, L.A.EVANS-MOLINA, C.: "100 years of insulin: celebrating the past, present and future of diabetes therapy", NAT MED, vol. 27, 2021, pages 1154 - 1164, XP037508957, DOI: 10.1038/s41591-021-01418-2 |
| SMITH ET AL., NUCLEIC ACIDS RES., vol. 363, no. 2, 2006, pages 283 - 294 |
| SPRENGER-HAUSSELS ET AL., PLANT J., vol. 22, 2000, pages 19 - 27 |
| STEWART ET AL., HUM GENE THER., vol. 2,2, no. 3, 2011, pages 357 - 369 |
| STRECKER ET AL., NATURE COMM., vol. 10, 2019, pages 212 |
| STRECKER ET AL., SCIENCE, vol. 363, 2019, pages 88 - 91 |
| SUSSMAN ET AL., J MOL BIOL, vol. 342, 2004, pages 31 - 41 |
| SZCZEPEK ET AL., NATURE BIOTECH, vol. 25, 2007, pages 786 - 793 |
| TAKAHASHIYAMANAKA, CELL, vol. 126, 2006, pages 663 - 676 |
| THROM ET AL., BLOOD, vol. 113, no. 21, 2009, pages 5104 - 5110 |
| TORCHIA ET AL., CURR. OPIN. CELL. BIOL., vol. 10, 1998, pages 373 - 383 |
| ULMASON ET AL., PROC. NATL. ACAD. SCI. USA, vol. 96, 1999, pages 15,348 - 15,353 |
| VAN ROEY ET AL., NATURE STRUCT. BIOL., vol. 9, 2002, pages 806 - 811 |
| VEGAS ET AL., NAT MED, vol. 22, no. 3, 2016, pages 306 - 11 |
| VELAZCO-CRUZ ET AL., CELL REPORTS, 2020 |
| WENZLAU JANET M. ET AL: "Novel Diabetes Autoantibodies and Prediction of Type 1 Diabetes", CURRENT DIABETES REPORTS, vol. 13, no. 5, 31 July 2013 (2013-07-31), US, pages 608 - 615, XP055779083, ISSN: 1534-4827, Retrieved from the Internet <URL:http://link.springer.com/article/10.1007/s11892-013-0405-9/fulltext.html> DOI: 10.1007/s11892-013-0405-9 * |
| WITKOWSKI, P. ET AL.: "The demise of islet allotransplantation in the United States: A call for an urgent regulatory update", AM J TRANSPLANT, vol. 21, 2021, pages 1365 - 1375 |
| WOOD ET AL., SCIENCE, vol. 333, 2011, pages 307 |
| XIN-XING WAN: "Stem Cell Transplantation in the Treatment of Type 1 Diabetes Mellitus: From Insulin Replacement to Beta-Cell Replacement", FRONTIERS IN ENDOCRINOLOGY, vol. 13, 18 March 2022 (2022-03-18), CH, XP093154628, ISSN: 1664-2392, DOI: 10.3389/fendo.2022.859638 * |
| YANG ET AL., COMMUN BIOL, vol. 3, 2020, pages 116 |
| ZETSCHE ET AL., CELL, vol. 163, 2015, pages 759 - 771 |
| ZHAO Y ET AL: "Selective destruction of mouse islet beta cells by human T lymphocytes in a newly-established humanized type 1 diabetic model", BIOCHEMICAL AND BIOPHYSICAL RESEARCH COMMUNICATIONS, ELSEVIER, AMSTERDAM NL, vol. 399, no. 4, 3 September 2010 (2010-09-03), pages 629 - 636, XP027259131, ISSN: 0006-291X, [retrieved on 20100804], DOI: 10.1016/J.BBRC.2010.07.128 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7348160B2 (en) | Delivery methods and compositions for nuclease-mediated genome genetic engineering | |
| CN105208866B (en) | Targeted disruption of the T cell receptor gene using engineered zinc finger protein nucleases | |
| US20230272415A1 (en) | Genetic modification of cytokine inducible sh2-containing protein (cish) gene | |
| JP2023510916A (en) | Safety switch for control of gene expression | |
| US20250223564A1 (en) | Hypoimmune beta cells differentiated from pluripotent stem cells and related uses and methods | |
| WO2016019144A2 (en) | Gene correction of scid-related genes in hematopoietic stem and progenitor cells | |
| WO2014081855A1 (en) | Methods and compositions for muscular dystrophies | |
| US20240425820A1 (en) | Genetically modified cells for allogeneic cell therapy to reduce complement-mediated inflammatory reactions | |
| JP2024535677A (en) | Genetically modified cells for allogeneic cell therapy to reduce immediate blood-borne inflammatory responses | |
| KR20240073006A (en) | Genetically modified primary cells for allogeneic cell therapy | |
| US11020492B2 (en) | Gene correction of SCID-related genes in hematopoietic stem and progenitor cells | |
| WO2024151541A1 (en) | Type-1 diabetes autoimmune mouse | |
| CN118076362A (en) | Inducible system for altering gene expression in low-immunogenic cells | |
| CN118871590A (en) | Low-immunity beta cells differentiated from pluripotent stem cells and related uses and methods | |
| US20250101382A1 (en) | Genetically modified cells and compositions and uses thereof | |
| WO2024243236A2 (en) | Methods of delivery of islet cells and related methods | |
| CN118434844A (en) | Genetically modified cells for allogeneic cell therapy to reduce complement-mediated inflammatory responses | |
| CN118382693A (en) | Genetically modified cells for allogeneic cell therapy to reduce immediate blood-mediated inflammatory responses | |
| WO2024229302A1 (en) | Methods of dosing and administration of engineered islet cells | |
| CN118401653A (en) | Genetically modified cells for allogeneic cell therapy |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24705301 Country of ref document: EP Kind code of ref document: A1 |
|
| NENP | Non-entry into the national phase |
Ref country code: DE |
|
| 122 | Ep: pct application non-entry in european phase |
Ref document number: 24705301 Country of ref document: EP Kind code of ref document: A1 |