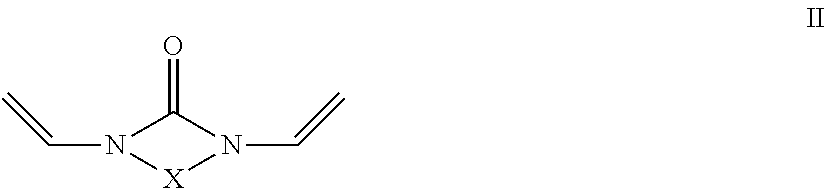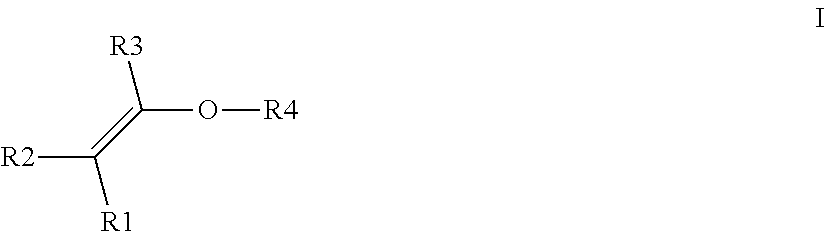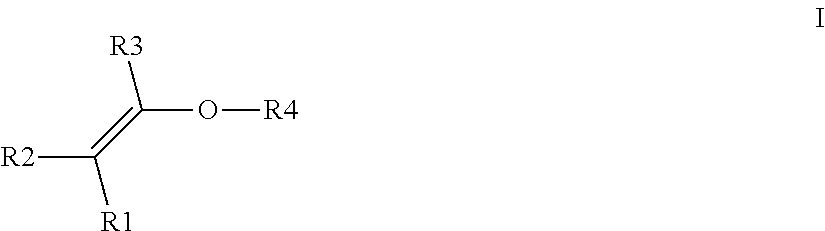US20140155565A1 - Hydrophilic crosslinked polymer - Google Patents
Hydrophilic crosslinked polymer Download PDFInfo
- Publication number
- US20140155565A1 US20140155565A1 US14/173,218 US201414173218A US2014155565A1 US 20140155565 A1 US20140155565 A1 US 20140155565A1 US 201414173218 A US201414173218 A US 201414173218A US 2014155565 A1 US2014155565 A1 US 2014155565A1
- Authority
- US
- United States
- Prior art keywords
- alkyl
- radical
- aryl
- substituted
- independently
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 229920006037 cross link polymer Polymers 0.000 title abstract description 9
- -1 heterocyclic divinyl ethers Chemical class 0.000 claims abstract description 208
- 229920000642 polymer Polymers 0.000 claims abstract description 77
- 239000002245 particle Substances 0.000 claims abstract description 21
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 65
- 229910052760 oxygen Inorganic materials 0.000 claims description 51
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 claims description 46
- 150000003254 radicals Chemical group 0.000 claims description 42
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 claims description 41
- 125000004191 (C1-C6) alkoxy group Chemical group 0.000 claims description 40
- 125000000172 C5-C10 aryl group Chemical group 0.000 claims description 36
- MDFFNEOEWAXZRQ-UHFFFAOYSA-N aminyl Chemical compound [NH2] MDFFNEOEWAXZRQ-UHFFFAOYSA-N 0.000 claims description 36
- 229910052736 halogen Inorganic materials 0.000 claims description 36
- 150000002367 halogens Chemical class 0.000 claims description 36
- 125000004209 (C1-C8) alkyl group Chemical group 0.000 claims description 32
- 125000001424 substituent group Chemical group 0.000 claims description 32
- 125000004432 carbon atom Chemical group C* 0.000 claims description 30
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 30
- 229910052717 sulfur Inorganic materials 0.000 claims description 26
- 150000001875 compounds Chemical class 0.000 claims description 23
- 229910052799 carbon Inorganic materials 0.000 claims description 21
- 150000005840 aryl radicals Chemical class 0.000 claims description 20
- 239000003431 cross linking reagent Substances 0.000 claims description 18
- 238000000034 method Methods 0.000 claims description 17
- 229920001577 copolymer Polymers 0.000 claims description 12
- 125000003118 aryl group Chemical group 0.000 claims description 10
- WULAHPYSGCVQHM-UHFFFAOYSA-N 2-(2-ethenoxyethoxy)ethanol Chemical compound OCCOCCOC=C WULAHPYSGCVQHM-UHFFFAOYSA-N 0.000 claims description 6
- HMBNQNDUEFFFNZ-UHFFFAOYSA-N 4-ethenoxybutan-1-ol Chemical compound OCCCCOC=C HMBNQNDUEFFFNZ-UHFFFAOYSA-N 0.000 claims description 6
- BCTDCDYHRUIHSF-UHFFFAOYSA-N 5-ethenoxypentan-1-ol Chemical compound OCCCCCOC=C BCTDCDYHRUIHSF-UHFFFAOYSA-N 0.000 claims description 6
- MOOIXEMFUKBQLJ-UHFFFAOYSA-N [1-(ethenoxymethyl)cyclohexyl]methanol Chemical compound C=COCC1(CO)CCCCC1 MOOIXEMFUKBQLJ-UHFFFAOYSA-N 0.000 claims description 6
- 229920001222 biopolymer Polymers 0.000 claims description 6
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 claims description 6
- 108090000765 processed proteins & peptides Proteins 0.000 claims description 5
- 238000010532 solid phase synthesis reaction Methods 0.000 claims description 5
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 claims description 4
- JVGOEGSMSPAINF-UHFFFAOYSA-N 1,3-bis(ethenyl)-1,3-diazinan-2-one Chemical compound C=CN1CCCN(C=C)C1=O JVGOEGSMSPAINF-UHFFFAOYSA-N 0.000 claims description 4
- HMYBDZFSXBJDGL-UHFFFAOYSA-N 1,3-bis(ethenyl)imidazolidin-2-one Chemical compound C=CN1CCN(C=C)C1=O HMYBDZFSXBJDGL-UHFFFAOYSA-N 0.000 claims description 4
- VUIWJRYTWUGOOF-UHFFFAOYSA-N 2-ethenoxyethanol Chemical group OCCOC=C VUIWJRYTWUGOOF-UHFFFAOYSA-N 0.000 claims description 4
- OJXVWULQHYTXRF-UHFFFAOYSA-N 3-ethenoxypropan-1-ol Chemical compound OCCCOC=C OJXVWULQHYTXRF-UHFFFAOYSA-N 0.000 claims description 4
- ASPUDHDPXIBNAP-UHFFFAOYSA-N 6-ethenoxyhexan-1-ol Chemical compound OCCCCCCOC=C ASPUDHDPXIBNAP-UHFFFAOYSA-N 0.000 claims description 4
- 125000000753 cycloalkyl group Chemical group 0.000 claims description 4
- 108020004707 nucleic acids Proteins 0.000 claims description 4
- 150000007523 nucleic acids Chemical class 0.000 claims description 4
- 102000039446 nucleic acids Human genes 0.000 claims description 4
- 229910052740 iodine Inorganic materials 0.000 claims description 3
- 238000004519 manufacturing process Methods 0.000 claims description 2
- 238000002360 preparation method Methods 0.000 abstract description 9
- QYKIQEUNHZKYBP-UHFFFAOYSA-N Vinyl ether Chemical class C=COC=C QYKIQEUNHZKYBP-UHFFFAOYSA-N 0.000 abstract description 5
- 238000004132 cross linking Methods 0.000 abstract description 4
- 238000000926 separation method Methods 0.000 description 40
- 239000000463 material Substances 0.000 description 38
- 239000012636 effector Substances 0.000 description 29
- 239000000178 monomer Substances 0.000 description 24
- 239000000725 suspension Substances 0.000 description 24
- 238000002955 isolation Methods 0.000 description 21
- 125000000217 alkyl group Chemical group 0.000 description 20
- 239000002594 sorbent Substances 0.000 description 20
- 239000002585 base Substances 0.000 description 17
- 239000000203 mixture Substances 0.000 description 16
- 102000004169 proteins and genes Human genes 0.000 description 16
- 108090000623 proteins and genes Proteins 0.000 description 16
- RAHZWNYVWXNFOC-UHFFFAOYSA-N Sulphur dioxide Chemical compound O=S=O RAHZWNYVWXNFOC-UHFFFAOYSA-N 0.000 description 14
- 238000004587 chromatography analysis Methods 0.000 description 13
- 239000012071 phase Substances 0.000 description 13
- 239000011148 porous material Substances 0.000 description 13
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 12
- 0 [1*]/C([2*])=C(/[3*])O[4*] Chemical compound [1*]/C([2*])=C(/[3*])O[4*] 0.000 description 12
- 238000006243 chemical reaction Methods 0.000 description 12
- 229910052757 nitrogen Inorganic materials 0.000 description 11
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 11
- 239000000243 solution Substances 0.000 description 11
- 239000011159 matrix material Substances 0.000 description 9
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 9
- 238000009826 distribution Methods 0.000 description 8
- 239000003446 ligand Substances 0.000 description 8
- 239000003381 stabilizer Substances 0.000 description 8
- 239000008346 aqueous phase Substances 0.000 description 7
- 230000027455 binding Effects 0.000 description 7
- 238000001542 size-exclusion chromatography Methods 0.000 description 7
- 239000002904 solvent Substances 0.000 description 7
- IJGRMHOSHXDMSA-UHFFFAOYSA-N Atomic nitrogen Chemical compound N#N IJGRMHOSHXDMSA-UHFFFAOYSA-N 0.000 description 6
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- YXFVVABEGXRONW-UHFFFAOYSA-N Toluene Chemical compound CC1=CC=CC=C1 YXFVVABEGXRONW-UHFFFAOYSA-N 0.000 description 6
- 239000008186 active pharmaceutical agent Substances 0.000 description 6
- 239000003999 initiator Substances 0.000 description 6
- AJPJDKMHJJGVTQ-UHFFFAOYSA-M sodium dihydrogen phosphate Chemical compound [Na+].OP(O)([O-])=O AJPJDKMHJJGVTQ-UHFFFAOYSA-M 0.000 description 6
- 229910000162 sodium phosphate Inorganic materials 0.000 description 6
- FPSURBCYSCOZSE-UHFFFAOYSA-N 1-ethenoxybutan-1-ol Chemical compound CCCC(O)OC=C FPSURBCYSCOZSE-UHFFFAOYSA-N 0.000 description 5
- 230000015572 biosynthetic process Effects 0.000 description 5
- 230000007062 hydrolysis Effects 0.000 description 5
- 238000006460 hydrolysis reaction Methods 0.000 description 5
- 239000003960 organic solvent Substances 0.000 description 5
- 239000003361 porogen Substances 0.000 description 5
- 150000003839 salts Chemical class 0.000 description 5
- 239000000126 substance Substances 0.000 description 5
- 125000000547 substituted alkyl group Chemical group 0.000 description 5
- 125000000472 sulfonyl group Chemical group *S(*)(=O)=O 0.000 description 5
- FJTATUCRGJDPSG-UHFFFAOYSA-N C=CN1CN(C=C)C1=O Chemical compound C=CN1CN(C=C)C1=O FJTATUCRGJDPSG-UHFFFAOYSA-N 0.000 description 4
- 125000004183 alkoxy alkyl group Chemical group 0.000 description 4
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 4
- 125000004181 carboxyalkyl group Chemical group 0.000 description 4
- 125000002091 cationic group Chemical group 0.000 description 4
- 238000011437 continuous method Methods 0.000 description 4
- 125000004093 cyano group Chemical group *C#N 0.000 description 4
- 125000004966 cyanoalkyl group Chemical group 0.000 description 4
- LQZZUXJYWNFBMV-UHFFFAOYSA-N dodecan-1-ol Chemical compound CCCCCCCCCCCCO LQZZUXJYWNFBMV-UHFFFAOYSA-N 0.000 description 4
- SNRUBQQJIBEYMU-UHFFFAOYSA-N dodecane Chemical compound CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 4
- 150000002148 esters Chemical class 0.000 description 4
- 229920001477 hydrophilic polymer Polymers 0.000 description 4
- 230000014759 maintenance of location Effects 0.000 description 4
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 4
- 238000000465 moulding Methods 0.000 description 4
- 230000007935 neutral effect Effects 0.000 description 4
- 102000004196 processed proteins & peptides Human genes 0.000 description 4
- 239000000047 product Substances 0.000 description 4
- 125000004963 sulfonylalkyl group Chemical group 0.000 description 4
- OZAIFHULBGXAKX-UHFFFAOYSA-N 2-(2-cyanopropan-2-yldiazenyl)-2-methylpropanenitrile Chemical compound N#CC(C)(C)N=NC(C)(C)C#N OZAIFHULBGXAKX-UHFFFAOYSA-N 0.000 description 3
- 108020004414 DNA Proteins 0.000 description 3
- 229920002307 Dextran Polymers 0.000 description 3
- RWRDLPDLKQPQOW-UHFFFAOYSA-N Pyrrolidine Chemical compound C1CCNC1 RWRDLPDLKQPQOW-UHFFFAOYSA-N 0.000 description 3
- 239000002253 acid Substances 0.000 description 3
- 230000002378 acidificating effect Effects 0.000 description 3
- 239000013543 active substance Substances 0.000 description 3
- 150000001298 alcohols Chemical class 0.000 description 3
- 125000003545 alkoxy group Chemical group 0.000 description 3
- 239000000470 constituent Substances 0.000 description 3
- 238000001212 derivatisation Methods 0.000 description 3
- 125000004663 dialkyl amino group Chemical group 0.000 description 3
- 150000002118 epoxides Chemical group 0.000 description 3
- 150000002500 ions Chemical class 0.000 description 3
- 125000005647 linker group Chemical group 0.000 description 3
- 238000012434 mixed-mode chromatography Methods 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 239000012074 organic phase Substances 0.000 description 3
- 229920002451 polyvinyl alcohol Polymers 0.000 description 3
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 238000001179 sorption measurement Methods 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 230000008961 swelling Effects 0.000 description 3
- 238000003786 synthesis reaction Methods 0.000 description 3
- 125000005208 trialkylammonium group Chemical group 0.000 description 3
- YBJHBAHKTGYVGT-ZKWXMUAHSA-N (+)-Biotin Chemical compound N1C(=O)N[C@@H]2[C@H](CCCCC(=O)O)SC[C@@H]21 YBJHBAHKTGYVGT-ZKWXMUAHSA-N 0.000 description 2
- BBMCTIGTTCKYKF-UHFFFAOYSA-N 1-heptanol Chemical compound CCCCCCCO BBMCTIGTTCKYKF-UHFFFAOYSA-N 0.000 description 2
- 125000001731 2-cyanoethyl group Chemical group [H]C([H])(*)C([H])([H])C#N 0.000 description 2
- 125000004200 2-methoxyethyl group Chemical group [H]C([H])([H])OC([H])([H])C([H])([H])* 0.000 description 2
- 125000004493 2-methylbut-1-yl group Chemical group CC(C*)CC 0.000 description 2
- 125000005916 2-methylpentyl group Chemical group 0.000 description 2
- 125000005917 3-methylpentyl group Chemical group 0.000 description 2
- KDCGOANMDULRCW-UHFFFAOYSA-N 7H-purine Chemical compound N1=CNC2=NC=NC2=C1 KDCGOANMDULRCW-UHFFFAOYSA-N 0.000 description 2
- NIXOWILDQLNWCW-UHFFFAOYSA-N Acrylic acid Chemical class OC(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-N 0.000 description 2
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 2
- CSCPPACGZOOCGX-UHFFFAOYSA-N CC(C)=O Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 2
- 102000053602 DNA Human genes 0.000 description 2
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 2
- CERQOIWHTDAKMF-UHFFFAOYSA-N Methacrylic acid Chemical compound CC(=C)C(O)=O CERQOIWHTDAKMF-UHFFFAOYSA-N 0.000 description 2
- YNAVUWVOSKDBBP-UHFFFAOYSA-N Morpholine Chemical compound C1COCCN1 YNAVUWVOSKDBBP-UHFFFAOYSA-N 0.000 description 2
- 102000016943 Muramidase Human genes 0.000 description 2
- 108010014251 Muramidase Proteins 0.000 description 2
- 108010062010 N-Acetylmuramoyl-L-alanine Amidase Proteins 0.000 description 2
- GLUUGHFHXGJENI-UHFFFAOYSA-N Piperazine Chemical compound C1CNCCN1 GLUUGHFHXGJENI-UHFFFAOYSA-N 0.000 description 2
- 239000004372 Polyvinyl alcohol Substances 0.000 description 2
- KYQCOXFCLRTKLS-UHFFFAOYSA-N Pyrazine Chemical compound C1=CN=CC=N1 KYQCOXFCLRTKLS-UHFFFAOYSA-N 0.000 description 2
- KAESVJOAVNADME-UHFFFAOYSA-N Pyrrole Chemical compound C=1C=CNC=1 KAESVJOAVNADME-UHFFFAOYSA-N 0.000 description 2
- SMWDFEZZVXVKRB-UHFFFAOYSA-N Quinoline Chemical compound N1=CC=CC2=CC=CC=C21 SMWDFEZZVXVKRB-UHFFFAOYSA-N 0.000 description 2
- MZVQCMJNVPIDEA-UHFFFAOYSA-N [CH2]CN(CC)CC Chemical group [CH2]CN(CC)CC MZVQCMJNVPIDEA-UHFFFAOYSA-N 0.000 description 2
- 150000007513 acids Chemical class 0.000 description 2
- 239000000654 additive Substances 0.000 description 2
- 125000001931 aliphatic group Chemical group 0.000 description 2
- 150000001338 aliphatic hydrocarbons Chemical class 0.000 description 2
- 125000005037 alkyl phenyl group Chemical group 0.000 description 2
- 125000004103 aminoalkyl group Chemical group 0.000 description 2
- 239000007864 aqueous solution Substances 0.000 description 2
- 150000004945 aromatic hydrocarbons Chemical class 0.000 description 2
- 239000011324 bead Substances 0.000 description 2
- IOJUPLGTWVMSFF-UHFFFAOYSA-N benzothiazole Chemical compound C1=CC=C2SC=NC2=C1 IOJUPLGTWVMSFF-UHFFFAOYSA-N 0.000 description 2
- 230000002902 bimodal effect Effects 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 239000003054 catalyst Substances 0.000 description 2
- 238000006555 catalytic reaction Methods 0.000 description 2
- 210000004027 cell Anatomy 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 239000011248 coating agent Substances 0.000 description 2
- 238000000576 coating method Methods 0.000 description 2
- 239000012228 culture supernatant Substances 0.000 description 2
- 125000004122 cyclic group Chemical group 0.000 description 2
- 125000004985 dialkyl amino alkyl group Chemical group 0.000 description 2
- USIUVYZYUHIAEV-UHFFFAOYSA-N diphenyl ether Chemical compound C=1C=CC=CC=1OC1=CC=CC=C1 USIUVYZYUHIAEV-UHFFFAOYSA-N 0.000 description 2
- 238000004090 dissolution Methods 0.000 description 2
- 238000010828 elution Methods 0.000 description 2
- 239000000839 emulsion Substances 0.000 description 2
- 125000005745 ethoxymethyl group Chemical group [H]C([H])([H])C([H])([H])OC([H])([H])* 0.000 description 2
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- 125000005842 heteroatom Chemical group 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- 230000002209 hydrophobic effect Effects 0.000 description 2
- 125000001165 hydrophobic group Chemical group 0.000 description 2
- 150000002484 inorganic compounds Chemical class 0.000 description 2
- 229910010272 inorganic material Inorganic materials 0.000 description 2
- 230000003993 interaction Effects 0.000 description 2
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 2
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 2
- 125000001449 isopropyl group Chemical group [H]C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- 239000002502 liposome Substances 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 125000004184 methoxymethyl group Chemical group [H]C([H])([H])OC([H])([H])* 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- ZIUHHBKFKCYYJD-UHFFFAOYSA-N n,n'-methylenebisacrylamide Chemical compound C=CC(=O)NCNC(=O)C=C ZIUHHBKFKCYYJD-UHFFFAOYSA-N 0.000 description 2
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 2
- 125000003884 phenylalkyl group Chemical group 0.000 description 2
- 229920001223 polyethylene glycol Polymers 0.000 description 2
- 229920001289 polyvinyl ether Polymers 0.000 description 2
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 description 2
- 238000004237 preparative chromatography Methods 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 2
- 230000005855 radiation Effects 0.000 description 2
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 2
- VWDWKYIASSYTQR-UHFFFAOYSA-N sodium nitrate Chemical compound [Na+].[O-][N+]([O-])=O VWDWKYIASSYTQR-UHFFFAOYSA-N 0.000 description 2
- 230000003068 static effect Effects 0.000 description 2
- 238000001256 steam distillation Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- CIHOLLKRGTVIJN-UHFFFAOYSA-N tert‐butyl hydroperoxide Chemical compound CC(C)(C)OO CIHOLLKRGTVIJN-UHFFFAOYSA-N 0.000 description 2
- URAYPUMNDPQOKB-UHFFFAOYSA-N triacetin Chemical compound CC(=O)OCC(OC(C)=O)COC(C)=O URAYPUMNDPQOKB-UHFFFAOYSA-N 0.000 description 2
- ZICNIEOYWVIEQJ-UHFFFAOYSA-N (2-methylbenzoyl) 2-methylbenzenecarboperoxoate Chemical compound CC1=CC=CC=C1C(=O)OOC(=O)C1=CC=CC=C1C ZICNIEOYWVIEQJ-UHFFFAOYSA-N 0.000 description 1
- 125000003837 (C1-C20) alkyl group Chemical group 0.000 description 1
- JYEUMXHLPRZUAT-UHFFFAOYSA-N 1,2,3-triazine Chemical compound C1=CN=NN=C1 JYEUMXHLPRZUAT-UHFFFAOYSA-N 0.000 description 1
- LZDKZFUFMNSQCJ-UHFFFAOYSA-N 1,2-diethoxyethane Chemical compound CCOCCOCC LZDKZFUFMNSQCJ-UHFFFAOYSA-N 0.000 description 1
- UICXTANXZJJIBC-UHFFFAOYSA-N 1-(1-hydroperoxycyclohexyl)peroxycyclohexan-1-ol Chemical compound C1CCCCC1(O)OOC1(OO)CCCCC1 UICXTANXZJJIBC-UHFFFAOYSA-N 0.000 description 1
- DURPTKYDGMDSBL-UHFFFAOYSA-N 1-butoxybutane Chemical compound CCCCOCCCC DURPTKYDGMDSBL-UHFFFAOYSA-N 0.000 description 1
- AOPDRZXCEAKHHW-UHFFFAOYSA-N 1-pentoxypentane Chemical compound CCCCCOCCCCC AOPDRZXCEAKHHW-UHFFFAOYSA-N 0.000 description 1
- UHOPWFKONJYLCF-UHFFFAOYSA-N 2-(2-sulfanylethyl)isoindole-1,3-dione Chemical compound C1=CC=C2C(=O)N(CCS)C(=O)C2=C1 UHOPWFKONJYLCF-UHFFFAOYSA-N 0.000 description 1
- XHZPRMZZQOIPDS-UHFFFAOYSA-N 2-Methyl-2-[(1-oxo-2-propenyl)amino]-1-propanesulfonic acid Chemical compound OS(=O)(=O)CC(C)(C)NC(=O)C=C XHZPRMZZQOIPDS-UHFFFAOYSA-N 0.000 description 1
- HPILSDOMLLYBQF-UHFFFAOYSA-N 2-[1-(oxiran-2-ylmethoxy)butoxymethyl]oxirane Chemical compound C1OC1COC(CCC)OCC1CO1 HPILSDOMLLYBQF-UHFFFAOYSA-N 0.000 description 1
- 125000000022 2-aminoethyl group Chemical group [H]C([*])([H])C([H])([H])N([H])[H] 0.000 description 1
- NQBXSWAWVZHKBZ-UHFFFAOYSA-N 2-butoxyethyl acetate Chemical compound CCCCOCCOC(C)=O NQBXSWAWVZHKBZ-UHFFFAOYSA-N 0.000 description 1
- QCDWFXQBSFUVSP-UHFFFAOYSA-N 2-phenoxyethanol Chemical compound OCCOC1=CC=CC=C1 QCDWFXQBSFUVSP-UHFFFAOYSA-N 0.000 description 1
- 125000003903 2-propenyl group Chemical group [H]C([*])([H])C([H])=C([H])[H] 0.000 description 1
- SLRMQYXOBQWXCR-UHFFFAOYSA-N 2154-56-5 Chemical compound [CH2]C1=CC=CC=C1 SLRMQYXOBQWXCR-UHFFFAOYSA-N 0.000 description 1
- FRIBMENBGGCKPD-UHFFFAOYSA-N 3-(2,3-dimethoxyphenyl)prop-2-enal Chemical compound COC1=CC=CC(C=CC=O)=C1OC FRIBMENBGGCKPD-UHFFFAOYSA-N 0.000 description 1
- LNYTUARMNSFFBE-UHFFFAOYSA-N 4-(diethylazaniumyl)benzoate Chemical compound CCN(CC)C1=CC=C(C(O)=O)C=C1 LNYTUARMNSFFBE-UHFFFAOYSA-N 0.000 description 1
- VHYFNPMBLIVWCW-UHFFFAOYSA-N 4-Dimethylaminopyridine Chemical compound CN(C)C1=CC=NC=C1 VHYFNPMBLIVWCW-UHFFFAOYSA-N 0.000 description 1
- 125000004801 4-cyanophenyl group Chemical group [H]C1=C([H])C(C#N)=C([H])C([H])=C1* 0.000 description 1
- CRQJERFKOCCFPE-UHFFFAOYSA-N 5-methyl-1,3-bis(piperidin-1-ylmethyl)-1,3,5-triazinane-2-thione Chemical compound S=C1N(CN2CCCCC2)CN(C)CN1CN1CCCCC1 CRQJERFKOCCFPE-UHFFFAOYSA-N 0.000 description 1
- HBAQYPYDRFILMT-UHFFFAOYSA-N 8-[3-(1-cyclopropylpyrazol-4-yl)-1H-pyrazolo[4,3-d]pyrimidin-5-yl]-3-methyl-3,8-diazabicyclo[3.2.1]octan-2-one Chemical class C1(CC1)N1N=CC(=C1)C1=NNC2=C1N=C(N=C2)N1C2C(N(CC1CC2)C)=O HBAQYPYDRFILMT-UHFFFAOYSA-N 0.000 description 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical group CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 1
- OMPJBNCRMGITSC-UHFFFAOYSA-N Benzoylperoxide Chemical compound C=1C=CC=CC=1C(=O)OOC(=O)C1=CC=CC=C1 OMPJBNCRMGITSC-UHFFFAOYSA-N 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- WKBOTKDWSSQWDR-UHFFFAOYSA-N Bromine atom Chemical class [Br] WKBOTKDWSSQWDR-UHFFFAOYSA-N 0.000 description 1
- DKPFZGUDAPQIHT-UHFFFAOYSA-N Butyl acetate Natural products CCCCOC(C)=O DKPFZGUDAPQIHT-UHFFFAOYSA-N 0.000 description 1
- TVLUJRPNGFUHOR-UHFFFAOYSA-N C1C2CC3CC1CN(C2)C3.C1CN2CCN1CC2.C1CNC2NCCCN2C1.CN1C2CC(O)CC1C1OC12.CN1C2CC3CC1C(O3)C2O.CN1C2CCCC1CCC2.O=C(O)C1CC2CCCCC2N1.[H]C1(O)C2CN3CC1CN(C2)C3 Chemical compound C1C2CC3CC1CN(C2)C3.C1CN2CCN1CC2.C1CNC2NCCCN2C1.CN1C2CC(O)CC1C1OC12.CN1C2CC3CC1C(O3)C2O.CN1C2CCCC1CCC2.O=C(O)C1CC2CCCCC2N1.[H]C1(O)C2CN3CC1CN(C2)C3 TVLUJRPNGFUHOR-UHFFFAOYSA-N 0.000 description 1
- AMOAJONGPOMNNP-OXLPQMEASA-N C1CC2CCCN2C1.C1CC2CNCCN2C1.C1CCN2CCCCC2C1.C1CCN2CCNCC2C1.NC1CN2CCC1CC2.O=COC1CN2CCC1CC2.OC1CN2CCC1CC2.[H]C1(C=C)CN2CCC1C[C@@H]2CN.[H]C1(C=C)CN2CCC1C[C@@H]2CO.[H]C1(O)CC2CCC(C1)N2C Chemical compound C1CC2CCCN2C1.C1CC2CNCCN2C1.C1CCN2CCCCC2C1.C1CCN2CCNCC2C1.NC1CN2CCC1CC2.O=COC1CN2CCC1CC2.OC1CN2CCC1CC2.[H]C1(C=C)CN2CCC1C[C@@H]2CN.[H]C1(C=C)CN2CCC1C[C@@H]2CO.[H]C1(O)CC2CCC(C1)N2C AMOAJONGPOMNNP-OXLPQMEASA-N 0.000 description 1
- 150000000703 Cerium Chemical class 0.000 description 1
- 229910052684 Cerium Inorganic materials 0.000 description 1
- 108010038061 Chymotrypsinogen Proteins 0.000 description 1
- XDTMQSROBMDMFD-UHFFFAOYSA-N Cyclohexane Chemical compound C1CCCCC1 XDTMQSROBMDMFD-UHFFFAOYSA-N 0.000 description 1
- 102000018832 Cytochromes Human genes 0.000 description 1
- 108010052832 Cytochromes Proteins 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 1
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 1
- 108091034117 Oligonucleotide Proteins 0.000 description 1
- PCNDJXKNXGMECE-UHFFFAOYSA-N Phenazine Natural products C1=CC=CC2=NC3=CC=CC=C3N=C21 PCNDJXKNXGMECE-UHFFFAOYSA-N 0.000 description 1
- 229920001218 Pullulan Polymers 0.000 description 1
- WTKZEGDFNFYCGP-UHFFFAOYSA-N Pyrazole Chemical compound C=1C=NNC=1 WTKZEGDFNFYCGP-UHFFFAOYSA-N 0.000 description 1
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 1
- 108020004682 Single-Stranded DNA Proteins 0.000 description 1
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 1
- FZWLAAWBMGSTSO-UHFFFAOYSA-N Thiazole Chemical compound C1=CSC=N1 FZWLAAWBMGSTSO-UHFFFAOYSA-N 0.000 description 1
- XTXRWKRVRITETP-UHFFFAOYSA-N Vinyl acetate Chemical class CC(=O)OC=C XTXRWKRVRITETP-UHFFFAOYSA-N 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 125000003668 acetyloxy group Chemical group [H]C([H])([H])C(=O)O[*] 0.000 description 1
- 150000001252 acrylic acid derivatives Chemical class 0.000 description 1
- 150000008360 acrylonitriles Chemical class 0.000 description 1
- 238000001042 affinity chromatography Methods 0.000 description 1
- 150000007933 aliphatic carboxylic acids Chemical class 0.000 description 1
- 229910052783 alkali metal Inorganic materials 0.000 description 1
- 150000001340 alkali metals Chemical class 0.000 description 1
- 150000001413 amino acids Chemical class 0.000 description 1
- 150000003863 ammonium salts Chemical class 0.000 description 1
- 238000005349 anion exchange Methods 0.000 description 1
- 125000000129 anionic group Chemical group 0.000 description 1
- 239000003945 anionic surfactant Substances 0.000 description 1
- 239000012062 aqueous buffer Substances 0.000 description 1
- 239000007900 aqueous suspension Substances 0.000 description 1
- 239000012298 atmosphere Substances 0.000 description 1
- QVGXLLKOCUKJST-UHFFFAOYSA-N atomic oxygen Chemical compound [O] QVGXLLKOCUKJST-UHFFFAOYSA-N 0.000 description 1
- XOZUGNYVDXMRKW-AATRIKPKSA-N azodicarbonamide Chemical compound NC(=O)\N=N\C(N)=O XOZUGNYVDXMRKW-AATRIKPKSA-N 0.000 description 1
- 235000019399 azodicarbonamide Nutrition 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- UDEWPOVQBGFNGE-UHFFFAOYSA-N benzoic acid n-propyl ester Natural products CCCOC(=O)C1=CC=CC=C1 UDEWPOVQBGFNGE-UHFFFAOYSA-N 0.000 description 1
- 235000019400 benzoyl peroxide Nutrition 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 230000001588 bifunctional effect Effects 0.000 description 1
- 229960002685 biotin Drugs 0.000 description 1
- 235000020958 biotin Nutrition 0.000 description 1
- 239000011616 biotin Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 229940098773 bovine serum albumin Drugs 0.000 description 1
- 239000001506 calcium phosphate Substances 0.000 description 1
- 229910000389 calcium phosphate Inorganic materials 0.000 description 1
- 235000011010 calcium phosphates Nutrition 0.000 description 1
- 150000001720 carbohydrates Chemical class 0.000 description 1
- IGBSAAYMSMEDQY-UHFFFAOYSA-N carboxy hydrogen carbonate;2-propan-2-ylperoxypropane Chemical compound OC(=O)OC(O)=O.CC(C)OOC(C)C IGBSAAYMSMEDQY-UHFFFAOYSA-N 0.000 description 1
- 150000001735 carboxylic acids Chemical class 0.000 description 1
- ZMIGMASIKSOYAM-UHFFFAOYSA-N cerium Chemical compound [Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce][Ce] ZMIGMASIKSOYAM-UHFFFAOYSA-N 0.000 description 1
- ITZXULOAYIAYNU-UHFFFAOYSA-N cerium(4+) Chemical compound [Ce+4] ITZXULOAYIAYNU-UHFFFAOYSA-N 0.000 description 1
- 238000012512 characterization method Methods 0.000 description 1
- 239000013522 chelant Substances 0.000 description 1
- 239000007795 chemical reaction product Substances 0.000 description 1
- 125000001309 chloro group Chemical class Cl* 0.000 description 1
- 239000002131 composite material Substances 0.000 description 1
- 238000001816 cooling Methods 0.000 description 1
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 1
- UKJLNMAFNRKWGR-UHFFFAOYSA-N cyclohexatrienamine Chemical group NC1=CC=C=C[CH]1 UKJLNMAFNRKWGR-UHFFFAOYSA-N 0.000 description 1
- 125000002704 decyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000008367 deionised water Substances 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- LSXWFXONGKSEMY-UHFFFAOYSA-N di-tert-butyl peroxide Chemical compound CC(C)(C)OOC(C)(C)C LSXWFXONGKSEMY-UHFFFAOYSA-N 0.000 description 1
- XXBDWLFCJWSEKW-UHFFFAOYSA-N dimethylbenzylamine Chemical compound CN(C)CC1=CC=CC=C1 XXBDWLFCJWSEKW-UHFFFAOYSA-N 0.000 description 1
- BNIILDVGGAEEIG-UHFFFAOYSA-L disodium hydrogen phosphate Chemical compound [Na+].[Na+].OP([O-])([O-])=O BNIILDVGGAEEIG-UHFFFAOYSA-L 0.000 description 1
- 229910000397 disodium phosphate Inorganic materials 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 230000000694 effects Effects 0.000 description 1
- 239000003480 eluent Substances 0.000 description 1
- 238000003821 enantio-separation Methods 0.000 description 1
- 229940088598 enzyme Drugs 0.000 description 1
- 125000001033 ether group Chemical group 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 150000005194 ethylbenzenes Chemical class 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 238000003818 flash chromatography Methods 0.000 description 1
- 238000011010 flushing procedure Methods 0.000 description 1
- 235000013373 food additive Nutrition 0.000 description 1
- 239000002778 food additive Substances 0.000 description 1
- 125000000524 functional group Chemical group 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- 235000013773 glyceryl triacetate Nutrition 0.000 description 1
- 125000003055 glycidyl group Chemical group C(C1CO1)* 0.000 description 1
- 102000035122 glycosylated proteins Human genes 0.000 description 1
- 108091005608 glycosylated proteins Proteins 0.000 description 1
- 229920000578 graft copolymer Polymers 0.000 description 1
- ZRALSGWEFCBTJO-UHFFFAOYSA-O guanidinium Chemical compound NC(N)=[NH2+] ZRALSGWEFCBTJO-UHFFFAOYSA-O 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- FUZZWVXGSFPDMH-UHFFFAOYSA-N hexanoic acid Chemical compound CCCCCC(O)=O FUZZWVXGSFPDMH-UHFFFAOYSA-N 0.000 description 1
- 238000004128 high performance liquid chromatography Methods 0.000 description 1
- 210000004408 hybridoma Anatomy 0.000 description 1
- 150000002430 hydrocarbons Chemical group 0.000 description 1
- 238000004191 hydrophobic interaction chromatography Methods 0.000 description 1
- 229920013821 hydroxy alkyl cellulose Polymers 0.000 description 1
- 239000012442 inert solvent Substances 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 239000010954 inorganic particle Substances 0.000 description 1
- 238000011835 investigation Methods 0.000 description 1
- 238000004255 ion exchange chromatography Methods 0.000 description 1
- 125000003010 ionic group Chemical group 0.000 description 1
- 239000002608 ionic liquid Substances 0.000 description 1
- 230000001788 irregular Effects 0.000 description 1
- AWJUIBRHMBBTKR-UHFFFAOYSA-N iso-quinoline Natural products C1=NC=CC2=CC=CC=C21 AWJUIBRHMBBTKR-UHFFFAOYSA-N 0.000 description 1
- 238000000622 liquid--liquid extraction Methods 0.000 description 1
- 210000002751 lymph Anatomy 0.000 description 1
- 239000004325 lysozyme Substances 0.000 description 1
- 229960000274 lysozyme Drugs 0.000 description 1
- 235000010335 lysozyme Nutrition 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- VTHJTEIRLNZDEV-UHFFFAOYSA-L magnesium dihydroxide Chemical compound [OH-].[OH-].[Mg+2] VTHJTEIRLNZDEV-UHFFFAOYSA-L 0.000 description 1
- 239000000347 magnesium hydroxide Substances 0.000 description 1
- 229910001862 magnesium hydroxide Inorganic materials 0.000 description 1
- 239000003550 marker Substances 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 238000010327 methods by industry Methods 0.000 description 1
- 229920000609 methyl cellulose Polymers 0.000 description 1
- OLXYLDUSSBULGU-UHFFFAOYSA-N methyl pyridine-4-carboxylate Chemical compound COC(=O)C1=CC=NC=C1 OLXYLDUSSBULGU-UHFFFAOYSA-N 0.000 description 1
- 239000001923 methylcellulose Substances 0.000 description 1
- 239000008267 milk Substances 0.000 description 1
- 210000004080 milk Anatomy 0.000 description 1
- 235000013336 milk Nutrition 0.000 description 1
- 150000002772 monosaccharides Chemical class 0.000 description 1
- 239000002105 nanoparticle Substances 0.000 description 1
- 150000002825 nitriles Chemical class 0.000 description 1
- 125000004433 nitrogen atom Chemical group N* 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 230000009871 nonspecific binding Effects 0.000 description 1
- 125000001400 nonyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 150000001451 organic peroxides Chemical class 0.000 description 1
- 239000001301 oxygen Substances 0.000 description 1
- 229960005323 phenoxyethanol Drugs 0.000 description 1
- 125000000286 phenylethyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])C([H])([H])* 0.000 description 1
- 239000013612 plasmid Substances 0.000 description 1
- 239000002798 polar solvent Substances 0.000 description 1
- 229920002401 polyacrylamide Polymers 0.000 description 1
- 229920000058 polyacrylate Polymers 0.000 description 1
- 229920005646 polycarboxylate Polymers 0.000 description 1
- 108091033319 polynucleotide Proteins 0.000 description 1
- 102000040430 polynucleotide Human genes 0.000 description 1
- 239000002157 polynucleotide Substances 0.000 description 1
- 229920001451 polypropylene glycol Polymers 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 229910000027 potassium carbonate Inorganic materials 0.000 description 1
- 125000006235 propyl amino ethyl group Chemical group [H]N(C([H])([H])C([H])([H])*)C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000006239 protecting group Chemical group 0.000 description 1
- 235000019423 pullulan Nutrition 0.000 description 1
- JUJWROOIHBZHMG-UHFFFAOYSA-N pyridine Substances C1=CC=NC=C1 JUJWROOIHBZHMG-UHFFFAOYSA-N 0.000 description 1
- UMJSCPRVCHMLSP-UHFFFAOYSA-N pyridine Natural products COC1=CC=CN=C1 UMJSCPRVCHMLSP-UHFFFAOYSA-N 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 238000011084 recovery Methods 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- 238000007127 saponification reaction Methods 0.000 description 1
- 229920006395 saturated elastomer Polymers 0.000 description 1
- 229930195734 saturated hydrocarbon Natural products 0.000 description 1
- 238000007873 sieving Methods 0.000 description 1
- 229910052938 sodium sulfate Inorganic materials 0.000 description 1
- 235000011152 sodium sulphate Nutrition 0.000 description 1
- DAJSVUQLFFJUSX-UHFFFAOYSA-M sodium;dodecane-1-sulfonate Chemical compound [Na+].CCCCCCCCCCCCS([O-])(=O)=O DAJSVUQLFFJUSX-UHFFFAOYSA-M 0.000 description 1
- 239000007790 solid phase Substances 0.000 description 1
- 125000006850 spacer group Chemical group 0.000 description 1
- 239000007858 starting material Substances 0.000 description 1
- 125000001302 tertiary amino group Chemical group 0.000 description 1
- VLLMWSRANPNYQX-UHFFFAOYSA-N thiadiazole Chemical compound C1=CSN=N1.C1=CSN=N1 VLLMWSRANPNYQX-UHFFFAOYSA-N 0.000 description 1
- 238000004809 thin layer chromatography Methods 0.000 description 1
- 230000009261 transgenic effect Effects 0.000 description 1
- 229960002622 triacetin Drugs 0.000 description 1
- 150000003852 triazoles Chemical class 0.000 description 1
- STCOOQWBFONSKY-UHFFFAOYSA-N tributyl phosphate Chemical compound CCCCOP(=O)(OCCCC)OCCCC STCOOQWBFONSKY-UHFFFAOYSA-N 0.000 description 1
- QORWJWZARLRLPR-UHFFFAOYSA-H tricalcium bis(phosphate) Chemical compound [Ca+2].[Ca+2].[Ca+2].[O-]P([O-])([O-])=O.[O-]P([O-])([O-])=O QORWJWZARLRLPR-UHFFFAOYSA-H 0.000 description 1
- 238000005406 washing Methods 0.000 description 1
- 239000008096 xylene Substances 0.000 description 1
- 150000003738 xylenes Chemical class 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F216/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical
- C08F216/12—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical by an ether radical
- C08F216/14—Monomers containing only one unsaturated aliphatic radical
- C08F216/1416—Monomers containing oxygen in addition to the ether oxygen, e.g. allyl glycidyl ether
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
- B01J20/261—Synthetic macromolecular compounds obtained by reactions only involving carbon to carbon unsaturated bonds
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
- B01J20/265—Synthetic macromolecular compounds modified or post-treated polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/22—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof comprising organic material
- B01J20/26—Synthetic macromolecular compounds
- B01J20/265—Synthetic macromolecular compounds modified or post-treated polymers
- B01J20/267—Cross-linked polymers
-
- B—PERFORMING OPERATIONS; TRANSPORTING
- B01—PHYSICAL OR CHEMICAL PROCESSES OR APPARATUS IN GENERAL
- B01J—CHEMICAL OR PHYSICAL PROCESSES, e.g. CATALYSIS OR COLLOID CHEMISTRY; THEIR RELEVANT APPARATUS
- B01J20/00—Solid sorbent compositions or filter aid compositions; Sorbents for chromatography; Processes for preparing, regenerating or reactivating thereof
- B01J20/281—Sorbents specially adapted for preparative, analytical or investigative chromatography
- B01J20/282—Porous sorbents
- B01J20/285—Porous sorbents based on polymers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F216/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical
- C08F216/12—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by an alcohol, ether, aldehydo, ketonic, acetal or ketal radical by an ether radical
- C08F216/14—Monomers containing only one unsaturated aliphatic radical
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F261/00—Macromolecular compounds obtained by polymerising monomers on to polymers of oxygen-containing monomers as defined in group C08F16/00
- C08F261/06—Macromolecular compounds obtained by polymerising monomers on to polymers of oxygen-containing monomers as defined in group C08F16/00 on to polymers of unsaturated ethers
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08F—MACROMOLECULAR COMPOUNDS OBTAINED BY REACTIONS ONLY INVOLVING CARBON-TO-CARBON UNSATURATED BONDS
- C08F222/00—Copolymers of compounds having one or more unsaturated aliphatic radicals, each having only one carbon-to-carbon double bond, and at least one being terminated by a carboxyl radical and containing at least one other carboxyl radical in the molecule; Salts, anhydrides, esters, amides, imides, or nitriles thereof
- C08F222/36—Amides or imides
- C08F222/38—Amides
- C08F222/385—Monomers containing two or more (meth)acrylamide groups, e.g. N,N'-methylenebisacrylamide
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y10—TECHNICAL SUBJECTS COVERED BY FORMER USPC
- Y10T—TECHNICAL SUBJECTS COVERED BY FORMER US CLASSIFICATION
- Y10T428/00—Stock material or miscellaneous articles
- Y10T428/29—Coated or structually defined flake, particle, cell, strand, strand portion, rod, filament, macroscopic fiber or mass thereof
- Y10T428/2982—Particulate matter [e.g., sphere, flake, etc.]
Definitions
- the present invention relates to a hydrophilic crosslinked polymer, preferably in the form of porous particles, and to the preparation and use thereof.
- the polymer according to the invention is produced by polymerisation from chain-forming hydrophilic vinyl ethers and crosslinking N,N′-divinyl compounds, preferably heterocyclic N,N′-divinyl compounds.
- Hydrophilic, crosslinked, polymeric support materials are frequently employed in areas such as chromatography, solid-phase synthesis or catalysis.
- polymers of this type should have the following properties:
- WO 03/104294 discloses polyvinyl ethers, in particular for use in solid-phase synthesis.
- the polymers disclosed usually exhibit very strong swelling behaviour.
- WO 9513861 discloses polyvinyl ethers for separation purposes which are prepared by cationic polymerisation. The cationic polymerisation described must be carried out in combination with complex protecting-group chemistry.
- EP 0 266 503 discloses crosslinked polymers which are prepared by means of suspension polymerisation from monomers containing epoxide groups. The resultant polymers containing epoxide groups can then be used for the immobilisation of biologically active substances.
- EP 0 482 339 discloses a copolymer based on allyl monomers.
- the copolymer based on EP 0 482 339 comprising 1,2,-dihydroxy-3-allyloxypropane and N,N′-methylenebisacrylamide, commercially available under the name FractoPrep® (Merck KGaA, Germany), exhibits good properties for biochromatography with respect to hydrophilicity, stability to hydrolysis and pressure/flow stability.
- FractoPrep® Merck KGaA, Germany
- the object of the present invention was therefore to provide a hydrophilic polymeric support material which meets the above-mentioned requirements with respect to porosity, hydrophilicity, stability to hydrolysis and pressure/flow stability and in addition is simple to prepare.
- copolymerisation of hydrophilically substituted alkyl vinyl ethers using crosslinking, preferably heterocyclic N,N′-divinyl compounds enables the preparation of hydrophilic polymers having good properties, in particular for biochromatography.
- the polymers are preferably prepared by means of suspension polymerisation.
- the present invention therefore relates to a hydrophilic crosslinked polymer based on a copolymer at least comprising
- R1, R2, R3, independently of one another can be H or C1 to C6 alkyl, preferably H or —CH 3
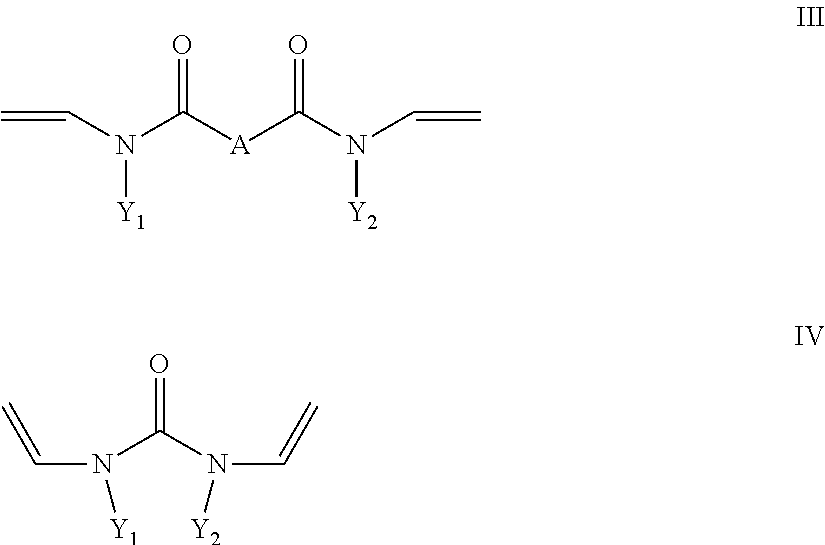
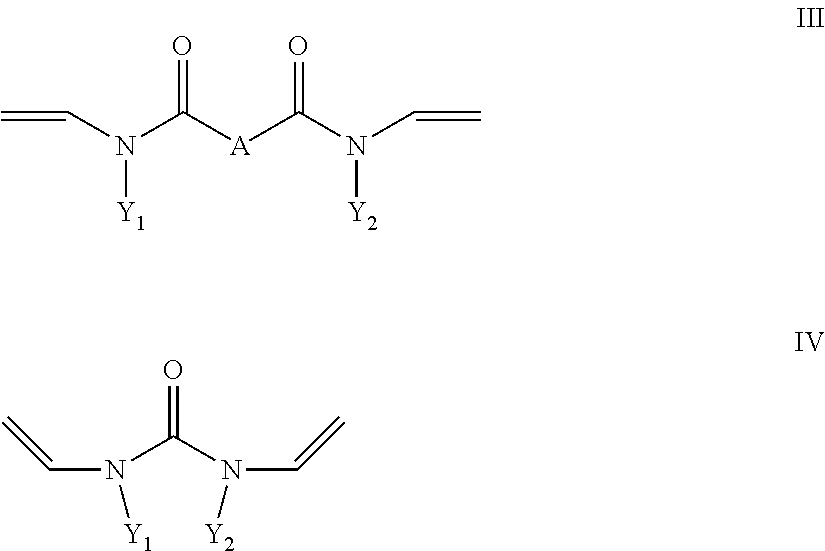
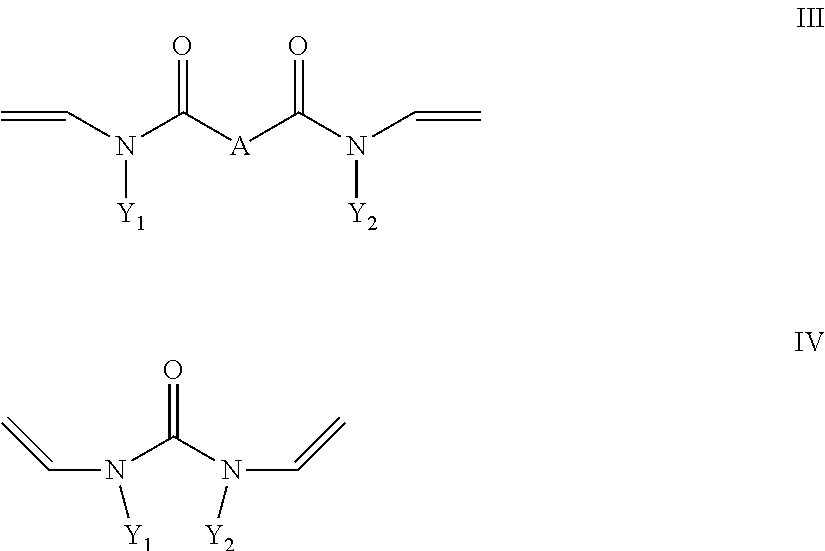
- R4 is a radical which carries at least one hydroxyl group and b) at least one crosslinking agent conforming to formula II and/or III and/or IV with
- X is a divalent alkyl radical having 2 to 5 C atoms, preferably 2 or 3 C atoms, in which one or more methylene groups which are not adjacent and are not located in the direct vicinity of N may be replaced by O, C ⁇ O, S, S ⁇ O, SO 2 , NH, NOH or N and one or more H atoms of the methylene groups may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH 2 , C5-C10-aryl, NH—(C1-C8)-alkyl, N—(C1-C8)-alkyl 2 , C1-C6-alkoxy or C1-C6-alkyl-OH, and
- Y1 and Y2 in formula III and IV are, independently of one another, C1 to C10 alkyl or cycloalkyl, where one or more non-adjacent methylene groups or methylene groups which are not located in the direct vicinity of N may be replaced by O, O ⁇ O, S, S ⁇ O, SO 2 , NH, NOH or N and one or more H of the methylene groups may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH 2 , C5-C10-aryl, NH(C1-C8)alkyl, N(C1-C8)alkyl 2 , C1-C6-alkoxy or 01-C6-alkyl-OH, or C6 to C18 aryl, where one or more H in the aryl system may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH 2 , NH(C1-
- R4 in formula I is
- a straight-chain or branched C1 to C10 alkyl radical in which one or more non-adjacent methylene groups may be replaced by O, C ⁇ O, S, S ⁇ O, SO 2 , NH, NOH, N and/or in which one or more H atoms may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH 2 , NH(C1-C8)alkyl, N(C1-C8)alkyl 2 , C1-C6-alkoxy or C1-C6-alkyl-OH and in which at least one OH group is present either on the C1 to C10 alkyl radical or on a substituent, or a cycloaliphatic radical, typically having 5 to 10 C atoms, in which one or more non-adjacent methylene groups may be replaced by O, C ⁇ O, S, S ⁇ O, SO 2 , NH, NOH, N and/or in
- R4 in formula I is
- a straight-chain or branched C1 to C10 alkyl radical in which one or more non-adjacent methylene groups may be replaced by O, S, SO 2 or NH and/or in which one or more H atoms may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, C1-C6-alkoxy or C1-C6-alkyl-OH and in which at least one OH group is present either on the C1 to C10 alkyl radical or on a substituent, or a cycloaliphatic radical, typically having 5 to 10 C atoms, in which one or more non-adjacent methylene groups may be replaced by O, S, SO 2 or NH and/or in which one or more H atoms of the cycloaliphatic radical may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, C1-C6-alkoxy or C1-C6
- the hydrophilically substituted alkyl vinyl ether employed is a compound of the formula I in which R4 is a radical which carries a hydroxyl group.
- the hydrophilically substituted alkyl vinyl ether employed is 1,2-ethanediol monovinyl ether, 1,3-propanediol monovinyl ether, 1,4-butanediol monovinyl ether, 1,5-pentanediol monovinyl ether, 1,6-hexanediol monovinyl ether or diethylene glycol monovinyl ether and the cycloaliphatic vinyl ether employed is cyclohexanedimethanol monovinyl ether.
- the crosslinking agent employed is divinylethyleneurea (1,3-divinylimidazolin-2-one) or divinylpropyleneurea (1,3-divinyltetrahydropyrimidin-2-one).
- the polymer is porous having pore sizes between 2 and 200 nm.
- the polymer is in the form of particles having a diameter between 3 and 300 ⁇ m.
- the polymer has been derivatised by means of separation effectors.
- the polymer has been derivatised by means of structures which have been attached to the polymer by graft polymerisation.
- the polymer has been derivatised by means of structures which have been attached to the polymer by graft polymerisation with cerium(IV) catalysis.
- the structures are copolymers which have been produced from two or more different monomers.
- the present invention also relates to a process for the preparation of polymers, characterised in that at least one hydrophilically substituted alkyl vinyl ether of the formula I and at least one crosslinking agent conforming to formula II and/or III and/or IV are copolymerised, with
- R1, R2, R3, independently of one another can be H or C1 to C6 alkyl, preferably H or —CH 3 , and R4 is a radical which carries at least one hydroxyl group and
- X is a divalent alkyl radical having 2 to 5 C atoms, preferably 2 or 3 C atoms, in which one or more methylene groups which are not adjacent and are not located in the direct vicinity of N may be replaced by O, C ⁇ O, S, S ⁇ O, SO 2 , NH, NOH or N and one or more H atoms of the methylene groups may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH 2 , C5-C10-aryl, NH—C1-C8-alkyl, N—C1-C8-alkyl 2 , C1-C6-alkoxy or C1-C6-alkyl-OH, and
- Y1 and Y2 in formula III and IV are, independently of one another, C1 to C10 alkyl or cycloalkyl, where one or more non-adjacent methylene groups or methylene groups which are not located in the direct vicinity of N may be replaced by O, C ⁇ O, S, S ⁇ O, SO 2 , NH, NOH or N and one or more H of the methylene groups may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH 2 , C5-C10-aryl, NH(C1-C8)alkyl, N(C1-C8)alkyl 2 , C1-C6-alkoxy or C1-C6-alkyl-OH, or C6 to C18 aryl, where one or more H in the aryl system may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH 2 , NH(C1
- R4 in formula I is
- a straight-chain or branched C1 to C10 alkyl radical in which one or more non-adjacent methylene groups may be replaced by O, C ⁇ O, S, S ⁇ O, SO 2 , NH, NOH, N and/or in which one or more H atoms may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH 2 , NH(C1-C8)alkyl, N(C1-C8)alkyl 2 , C1-C6-alkoxy or C1-C6-alkyl-OH and in which at least one OH group is present either on the C1 to C10 alkyl radical or on a substituent, or a cycloaliphatic radical, typically having 5 to 10 C atoms, in which one or more non-adjacent methylene groups may be replaced by O, C ⁇ O, S, S ⁇ O, SO 2 , NH, NOH, N and/or in
- a free-radical suspension polymerisation is carried out.
- a free-radical suspension polymeri sation is carried out in water as suspension medium in the presence of at least one suspension stabiliser and optional further additives, preferably inorganic salts and interface-active compounds.
- the temperature during the polymerisation is between 40 and 100° C.
- hydrophilically substituted alkyl vinyl ethers and crosslinking agents are employed in a weight ratio between 10:90 and 80:20.
- the polymerisation is carried out with addition of one or more porogens.
- the present invention also relates to a chromatography column, capillary, cartridge, stirred reactor or reactor containing a polymer according to the invention as sorbent or support material.
- the present invention also relates to the use of a hydrophilic crosslinked polymer according to the invention as sorbent in chromatography, as support material for the immobilisation of biologically or catalytically active substances or as support material for a solid-phase synthesis.
- the present invention also relates to polymers obtainable by the process according to the invention.
- FIG. 1 shows the retention behaviour of the polymer according to the invention on application of proteins. Further details are given in Example 2.
- FIG. 2 shows the experimentally determined distribution coefficient Kd of dextrans having different molecular weights. Further details are given in Example 3.
- FIG. 3 shows the storage stability of a polymer according to the invention in dilute sodium hydroxide solution. Further details are given in Example 4.
- FIG. 4 shows the separation of a protein mixture on a polymer according to the invention provided with cationic separation effectors. Further details are given in Example 5.
- FIG. 5 shows the pressure/flow behaviour of the polymer according to the invention in a chromatography column.
- cycloaliphatic radical denotes a saturated hydrocarbon radical in which all or at least some of the hydrocarbon units are a constituent of a cyclic structure.
- N(C1-C8)alkyl 2 denotes a nitrogen which is substituted by at least two alkyl radicals.
- the two alkyl radicals here may have, independently of one another, 1 to 8 C atoms in straight-chain or branched form.
- a heteroaryl radical is an at least partially aromatic radical which differs from an aryl radical in that one or more non adjacent C atoms have been replaced by N, S or O. It is clear to the person skilled in the art that, owing to the valences, the heteroatoms may optionally be substituted by, for example, H, C1-C6-alkyl or C1-C6-alkyl-OH.
- the polymer according to the invention is a hydrophilic, crosslinked polymer based on a copolymer at least comprising
- radicals R1, R2, R3 and R4 have the above-mentioned meanings, and b) at least one crosslinking agent conforming to formula II and/or III and/or IV
- the polymer is formed by copolymerisation of at least one compound from the group of the hydrophilically substituted alkyl vinyl ethers of the formula I and at least one compound from the group of the crosslinking agents of the formula II and/or III and/or IV.
- polymerisable compounds which are copolymerised into the polymer backbone can be added to the polymerisation mixture.
- These are typically compounds having at least one polymerisable double bond.
- only one compound from the group of the hydrophilically sub stituted alkyl vinyl ethers of the formula I and one compound from the group of the crosslinking agents of the formula II, Ill or IV is employed.
- the hydrophilically substituted alkyl vinyl ether employed is a compound of the formula I in which R4 is a radical which carries a hydroxyl group.
- the hydrophilically substituted alkyl vinyl ether employed is 1,2-ethanediol monovinyl ether, 1,3-propanediol monovinyl ether, 1,4-butanediol monovinyl ether, 1,5-pentanediol monovinyl ether, 1,6-hexanediol monovinyl ether or diethylene glycol monovinyl ether and the cycloaliphatic vinyl ether employed is cyclohexanedimethanol monovinyl ether, particularly preferably 1,4-butanediol monovinyl ether, 1,5-pentanediol monovinyl ether, diethylene glycol monovinyl ether or cyclohexanedimethanol monovinyl ether.
- crosslinking agents employed are preferably compounds of the formula II. Preference is given to the use of divinylpropyleneurea (1,3-divinyl-tetrahydropyrimidin-2-one) or particularly preferably divinylethyleneurea (1,3-divinylimidazolin-2-one).
- the proportion of the hydrophilically substituted alkyl vinyl ethers with respect to the weight of the polymer is typically between 1% (by weight) and 90% (by weight) or a maximum proportion by weight of the alkyl vinyl ether which corresponds to a molar ratio of 2:1, based on a bifunctional crosslinking agent, if the alkyl vinyl ether does not homopolymerise.
- the proportion of the hydrophilically substituted alkyl vinyl ethers is preferably between 10 and 80% (% by weight), particularly preferably between 35 and 60%.
- the proportion of the crosslinking agent is between 10 and 99 (% by weight), preferably between 20 and 90%, particularly preferably between 40 and 65%.
- an oil-in-water suspension polymerisation (normal-phase polymerisation) is carried out in a particularly simple procedure in the case of water-insoluble or sparingly water-soluble monomers, which are also known as oil.
- water-soluble monomers by contrast, the latter are dissolved in water and suspended and polymerised in a water-immiscible or only sparingly water-miscible organic solvent (inverse polymerisation).
- a further major advantage of the hydrophilically substituted alkyl vinyl ethers employed in accordance with the invention is that a subsequent hydrolysis step does not have to be carried out in order to produce a hydrophilic polymer. Instead, the use of the hydroxyl-containing vinyl ethers as starting materials results in the direct formation of a hydroxyl-containing polymer.
- the suspension polymerisation is carried out in a known manner.
- the suspension medium used in the normal-phase suspension polymerisation is typically water.
- One or more stabilisers and/or one or more interface-active compounds can, as is known, be added to the suspension medium.
- Stabilisers which can be employed are a multiplicity of water-soluble macromolecular compounds or also finely divided inorganic compounds.
- macromolecular stabilisers are polyvinylpyrrolidone, polyacrylates, polycarboxylates, polyacrylamide, polyvinyl alcohol, hydroxyalkylcellulose, methylcellulose or polyethylene glycols. Preference is given to the use of polyvinyl alcohols.
- Inorganic compounds which can be employed as stabilisers are, for example, calcium phosphate or magnesium hydroxide.
- Salts, ionic liquids or buffer substances such as, for example, Na 2 HPO 4 and NaH 2 PO 4 , can furthermore be added to the aqueous phase.
- Suitable interface-active compounds are, in particular, anionic and nonionic surfactants, such as ethoxylates of long-chain alcohols, ethoxylated mono-, di- and trialkylphenols, alkali metal and ammonium salts of C 12 -C 18 alkylsulfonic acids or arylsulfonic acids.
- anionic and nonionic surfactants such as ethoxylates of long-chain alcohols, ethoxylated mono-, di- and trialkylphenols, alkali metal and ammonium salts of C 12 -C 18 alkylsulfonic acids or arylsulfonic acids.
- the organic phase also known as the oil phase
- the polymer is suspended in the aqueous phase.
- the polymer can typically be filtered off or removed from the reaction mixture in another manner. Residues of solvents or other additives can be removed by washing with suitable solvents or, for example, steam distillation.
- the oil phase comprises inert solvents (porogens) in order to set the desired pore sizes, in addition to the monomers.
- Suitable organic solvents are, for example, aliphatic hydro-carbons (C 6 -C 18 ), such as, for example, hexane, dodecane or benzine mixtures, cycloaliphatic compounds, such as cyclohexane, aromatic hydrocarbons, such as toluene, ethylbenzenes or xylenes, or alcohols, such as aliphatic C 4 -C 12 -alcohols, for example heptanol, dodecanol, cyclohexanol, polyethylene glycols or polypropylene glycols of various molecular weight, or esters of aliphatic carboxylic acids, such as butyl acetate or propyl propionate, or esters of aromatic carboxylic acids, such as propyl benzoate, or esters, such as butyl glycol acetate or glycerol triacetate, or ethers, such as di-n-butylenes (C 6
- solvents can be employed individually or as a mixture of two or more solvents. They can either act as swelling agent or precipitant for the polymers and in this way influence the porosity.
- Examples of swelling agents for the polymers according to the invention are lower alcohols, aliphatic esters or aromatic hydrocarbons, such as toluene.
- Examples of precipitants are aliphatic hydrocarbons, such as dodecane or benzine mixtures.
- the porosity of the polymers can be fixed through the type and amount of the solvents in combination with the degree of crosslinking of the polymer.
- one or more initiators acting as free radicals are typically added to the organic phase.
- free-radical initiators are organic peroxides, such as di-tertbutyl peroxide, dibenzoyl peroxide, bis(o-methylbenzoyl) peroxide, tert-butyl hydroperoxide, cumene hydroperoxide, diisopropyl peroxide dicarbonate, cyclohexanone peroxide, or aliphatic azo compounds, such as ⁇ , ⁇ ′-azobisisobutyronitrile, azobiscyanovaleric acid, 1,1′-azocyclohexane-1,1′-dicarbo-nitrile or azodicarboxamide. Preference is given to ⁇ , ⁇ ′-azobisisobutyronitrile.
- Initiators which can be activated by means of radiation, for example UV radiation, can also be employed in accordance with the invention as free-radical initiators.
- phase ratios in the suspension polymerisation can typically be varied in the volume ratio between 1:20 (oil phase to aqueous phase) and 2:1, ratios between 1:5 and 1:1, particularly preferably between 1:3 and 1:1, are preferred.
- the ratio of organic solvent to monomer is typically between 1:20 and 4:1, preferably between 1:5 and 3:1, particularly preferably between 1:1 and 2.5:1.
- the concentration of the initiator is typically between 0.01% (by weight) and 10% (by weight), preferably between 0.1 and 7% (by weight) and particularly preferably between 0.2 and 7% (by weight).
- the concentration of stabilisers based on the amount of suspension medium, generally water, is between 0.01 and 7% (by weight), typically between 0.02 and 5% (by weight), particularly preferably between 0.05 and 2.5% (by weight).
- the suspension polymerisation is then carried out by mixing the two phases with vigorous stirring.
- the temperature of the mixture is typically kept at 20 to 100° C., preferably 40 to 100° C., for a period of 1 to 20 hours.
- the mixture is typically heated at 40 to 100° C. over a period of 0.5 to 5 hours and subsequently post-polymerised again for several hours at 70 to 100° C.
- the mixture should be stirred throughout the time.
- the mixture is generally cooled to room temperature, the organic solvents are removed, if they were added during the polymerisation, and the product is stored in water, an aqueous buffer or a mixture of water with an organic solvent, such as ethanol.
- the polymer can be brought to the desired particle-size distribution by sieving or another type of classification.
- the polymerisation according to the invention can likewise be carried out continuously.
- the particle production here is particularly preferably carried out using static mixers, in particular micromixers or caterpillar mixers.
- static mixers in particular micromixers or caterpillar mixers.
- the spatial distribution of two components to be mixed is influenced, with utilisation of the flow energy, by the flow management, for example by separation and recombination, twisting, distortion and expansion, so that large interfaces are formed for diffusive exchange between the components and a particularly uniform particle-size distribution can be achieved due to the high shear forces.
- Different mixers and their structure are known. For example, systems of this type are described in EP 1177243.
- the aqueous phase flows, for example, continuously with a defined volume flow through the micromixer.
- the oil phase is subsequently fed in via the second inlet, likewise with a defined, precisely set volume flow.
- the ratio of the two volume flows can be in a broad range and depends on the desired particle size, the nature and distribution of the particle sizes, the viscosity of the aqueous and the oil phase, depending on the reaction temperature, and the reaction kinetics and the requisite ratio of the porogens in the end mixture in order to achieve a suitable pore structure.
- the volume flow ratio between the aqueous phase and the oil phase is preferably 1:10 to 100:1, in particular 2:1 to 20:1.
- the suspension exiting from the static mixer is preferably polymerised in a continuously operating reactor.
- particle size of the resultant polymer can be influenced, inter alia through the nature and amount of the addition of suspension stabilisers and interface-active compounds and the stirring intensity.
- particle diameters between 3 ⁇ m and 500 ⁇ m, preferably between 3 ⁇ m and 300 ⁇ m, can be produced by means of suspension polymerisation, depending on the type of stirrer and the rotational speed.
- the particles can have an irregular shape or can preferably be spherical.
- particle diameters for example, between 20 nm and 3000 nm, preferably between 100 and 500 nm, can be produced.
- the polymers according to the invention can furthermore be prepared or exist in the form of membranes, fibres, hollow fibres, a coating or as a monolithic moulding.
- Monolithic mouldings are three-dimensional bodies, for example in cylindrical form.
- the polymers according to the invention can equally be employed as composite materials, i.e., for example, as a coating, for example of inorganic particles, or a moulding, or mixed with, for example, inorganic constituents.
- An example thereof are particles of the polymer according to the invention which can themselves be magnetised by copolymerisation of magnetisable particles or of a magnetisable core.
- the polymers according to the invention are preferably in the form of nonporous or particularly preferably porous particles.
- the pore sizes can typically be between 2 and 300 nm.
- pore size denotes pore radius. Preference is given to pore sizes or pore radii between 2 and 200 nm.
- the polymers according to the invention are in the form of monolithic mouldings, they can also have pore sizes up to 1000 nm.
- the pore distribution in the materials according to the invention can be monomodal, bimodal or oligomodal, preferably monomodal or bimodal.
- the pore sizes are preferably measured by means of size exclusion chromatography (SEC).
- SEC size exclusion chromatography
- hydrophilic, crosslinked polymers according to the invention are particularly suitable as sorbents in chromatography, as support materials for the immobilisation of biologically and/or catalytically active substances or as support materials for solid-phase syntheses of, for example, biopolymers, such as nucleic acids or peptides, or for combinatorial chemistry.
- the materials according to the invention are distinguished by good stability to hydrolysis, in particular in basic medium, and by good pressure/flow stability. Furthermore, their high hydrophilicity makes them particularly suitable for biochromatographic methods.
- the examples contain additional experimental data on these properties.
- the polymers according to the invention are particularly suitable as sorbents. For this purpose, they are introduced in a known manner into chromatography columns or capillaries.
- the polymer according to the invention can equally be packed in cartridges for chromatographic or other purposes.
- the present invention therefore also relates to chromatography columns, capillaries or cartridges which contain a polymer according to the invention as sorbent or support material.
- polymers according to the invention can be employed in their native form, i.e. without further derivatisation steps, for example, depending on the porosity set, for size exclusion chromatography (SEC) or for reduction of the salt content of solutions if these solutions contain target molecules whose molecular weight differs significantly from the molecular weight of the salt,
- SEC size exclusion chromatography
- separation effectors may be either ionic or neutral or have a preferred affinity or selectivity for one or more target molecules.
- the covalent bonding of the separation effectors generally takes place via the functional groups present on the polymer, such as, for example, hydroxyl groups, for example with formation of an ester or preferably an ether function, directly or via a linker or spacer.
- the linking to the base material takes place via graft polymerisation, such as, for example, a cerium(IV)-catalysed graft polymerisation with formation of a C—C link to the base material.
- Separation effectors are known to the person skilled in the art in the area of chromatography. Separation effectors are substituents which can be intro pokerd into the support material as early as during the synthesis of the base material or subsequently and influence the surface properties of the support material. In particular, targeted derivatisation of support materials by means of separation effectors produces support materials having certain chromatographic properties. In particular, separation effectors can have the following terminal groups:
- the polymer If it is intended firstly to provide the polymer with a universal linker, it can, for example for the introduction of epoxide groups, be reacted with glycidyl compounds, such as butanediol diglycidyl ether.
- glycidyl compounds such as butanediol diglycidyl ether.
- the polymer according to the invention can, as base material, be provided by graft polymerisation with tentacle-like structures, which can in turn carry the corresponding separation effectors or be functionalised by means of the latter.
- the grafting is preferably carried out in accordance with EP 0 337 144.
- the chain produced is linked to the base material via a monomer unit.
- the base material according to the invention is suspended in a solution of monomers, preferably in an aqueous solution.
- the grafting-on of the polymeric material is effected in the course of a conventional redox polymerisation with exclusion of oxygen.
- the polymerisation catalyst employed is cerium(IV) ions, since this catalyst forms free-radical sites on the surface of the base material, from which the graft polymerisation of the monomers is initiated.
- the polymerisation is terminated by termination reactions involving the cerium salts.
- the (average) chain length can be influenced by the concentration ratios of the base material, the initiator and the mono mers.
- uniform monomers or also mixtures of different monomers can be employed; in the latter case, grafted copolymers are formed.
- Suitable monomers for the preparation of the graft polymers are monomers conforming to formula V or VI.
- All these monomers are polymerisable substances containing reversibly bonding groups which can be polymerised by means of free radicals in aqueous solution and which may be neutral, acidic or basic.
- the monomers employed are vinylenecarbonates of the formula VI or vinylcarboxylates CR*R** ⁇ CR 11 —OCOCHR 15 R 16 of the formula V
- the product obtained is preferably subsequently converted into a separating material containing hydroxyl groups. This conversion into a hydroxyl phase is achieved by a mild alkaline or acidic saponification known per se.
- the reaction can be carried out using methanolic K 2 CO 3 solution at room temperature, described, for example, by Y. Tezuka et al., in Macromol. Chem. 186, 685-694 (1985).
- R 11 preferably denotes H, i.e. the acrylic acid derivatives are preferred.
- Y in formula V preferably denotes
- R 15 and R 16 independently of one another, denote H or an alkyl group having up to 5 C atoms. At least one of the radicals R 15 and R 16 is preferably H.
- the following radicals are particularly preferred: acetoxy, propionyloxy, butyryloxy, valeryloxy and hexanoyloxy radical.
- Z in formula V denotes —OR 14 , —OH or —NR 12 R 13 , preferably —NR 12 R 13 .
- the radicals R 12 and/or R 13 preferably denote an alkyl, phenyl, phenylalkyl or alkylphenyl group, where the alkyl and/or phenyl group may be mono- or polysubstituted, preferably mono- or disubstituted, particularly preferably monosubstituted, by an alkoxy, cyano, amino, mono- or dialkylamino, trialkylammonium, carboxyl, sulfonyl, acetoxy or acetamino radical.
- the radicals R 12 and/or R 13 preferably denote alkyl, alkoxyalkyl, cyanoalkyl, aminoalkyl, mono- or dialkylaminoalkyl, trialkylammoniumalkyl, carboxyalkyl or sulfonylalkyl having up to 10 C atoms, preferably up to 6 C atoms, particularly preferably up to 4 C atoms in the alkyl group, which may be linear or branched.
- R 12 and/or R 13 accordingly preferably denote methyl, ethyl, propyl, butyl, pentyl, hexyl, methoxymethyl, ethoxymethyl, 2-methoxyethyl, 2-, 3- or 4-oxapentyl, 2-, 3-, 4- or 5-oxahexyl, 2-, 3-, 4-, 5- or 6-oxaheptyl, isopropyl, 2-butyl, isobutyl, 2-methylbutyl, isopentyl, 2-methylpentyl, 3-methylpentyl, 2-oxa-3-methylbutyl, 3-oxa-4-methylbutyl, 2-methyl-3-oxa pentyl, 2-methyl-3-oxahexyl, furthermore also heptyl, octyl, nonyl or decyl.
- R 12 and/or R 13 preferably denote cyanomethyl, cyanoethyl, cyanopropyl, cyanobutyl, cyanopentyl, cyanohexyl, 2-cyanopropyl, 2-cyanobutyl, carboxylmethyl, carboxylethyl, carboxylpropyl, carboxyl isopropyl, carboxylbutyl, carboxylpentyl, carboxylhexyl, carboxyl-2-methylpropyl, carboxyl-2-methylbutyl, sulfonylmethyl, sulfonylethyl, sulfonylpropyl, sulfonylbutyl, sulfonylpentyl, sulfonylhexyl, sulfonyl-2-methylpropyl
- the alkyl groups are furthermore preferably monosubstituted by an amino, mono- or dialkylamino or trialkylammonium group.
- the alkyl groups here may be identical or different and have up to 10, preferably up to 6 C atoms, particularly preferably up to 4 C atoms, and accordingly preferably denote dimethylaminoethyl, diethylaminoethyl, methylaminoethyl, methylamino propyl, dimethylaminopropyl, ethylaminoethyl, propylaminoethyl, propylaminopropyl, dipropylaminoethyl, dipropylaminobutyl, diethylaminoethyl, trimethylammoniumethyl, trimethylammoniumpropyl, trimethylammoniumbutyl, triethylammoniumethyl, triethylammoniumpropyl, triethylammoniumethyl, aminoeth
- R 12 and/or R 13 preferably also has the meaning of a phenyl group, which is preferably monosubstituted by cyano, cyanoalkyl, amino, aminoalkyl, mono- or dialkylamino, alkyl, alkoxy, alkoxyalkyl, mono- or dialkylaminoalkyl, trialkylammonium or trialkylammoniumalkyl, carboxyl, carboxyalkyl, sulfonyl or sulfonylalkyl.
- the preferred meanings of these substituents correspond to the preferred alkyl groups and substituted alkyl groups indicated above.
- the substituent on the phenyl group is preferably in the p-position.
- p-Acetoxyphenyl, p-aminophenyl or p-acetaminophenyl are likewise preferred meanings for R 12 and/or R 13 .
- R 12 and/or R 13 Preference is furthermore given for R 12 and/or R 13 to an alkylphenyl or phenylalkyl group, where the preferred meanings indicated for the alkyl, substituted alkyl or substituted phenyl groups shall likewise apply.
- substituted phenyl groups for example, are regarded as particularly preferred: 4-cyanophenyl, 4-alkylphenyl, 4-(N,N-dimethylamino)phenyl, 4-(N,N-dialkylaminoethyl) phenyl, 4-ethoxyphenyl, 4-ethoxyethylphenyl, 4-trialkylammoniumphenyl, 4-carboxylphenyl, 4-sulfonylphenyl, phenylethyl, 4-(N-ethylamino) phenylpropyl or 4-cyanophenylethyl.
- preferred monomers of the formula V are those in which R 12 and/or R 13 denote a cyclic or bicyclic radical, which may be aromatic or saturated, having 5-10 C atoms in which one or more CH or CH 2 groups have been replaced by N or NH, N or NH and S, or N or NH and O.
- R 12 and/or R 13 accordingly preferably also denote a pyridine radical, imidazolyl radical, indolyl radical, furthermore preferably a pyrrole, pyrimidine, pyrazine, quinoline or isoquinoline radical.
- R 12 and/or R 13 may also, for example, denote a thiazole, thiadiazole, morpholine, triazine, piperazine, benzothiazole, purine, pyrazole, triazole, pyrrolidine or isoxazole radical.
- radicals R 12 and R 13 must, in order to obtain suitable exchangers, be matched to one another in such a way that either both radicals contain an acidic or basic group or alternatively one of the radicals is neutral.
- the person skilled in the art is presented with no difficulty in assigning the groups correspondingly and thus assembling suitable radicals for R 12 and R 13 , depending on the function and task of the desired ion exchanger.
- One of the two radicals R 12 and R 13 is preferably a neutral radical.
- R 14 preferably denotes alkyl, alkoxyalkyl, cyanoalkyl, carboxyalkyl or sulfonylalkyl having up to 10 C atoms, preferably having up to 6 C atoms, particularly preferably having up to 4 C atoms, in the alkyl group, which may be linear or branched.
- R 14 accordingly preferably denotes methyl, ethyl, propyl, butyl, pentyl, hexyl, methoxymethyl, ethoxymethyl, 2-methoxyethyl, 2-, 3- or 4-oxapentyl, isopropyl, 2-butyl, isobutyl, 2-methylbutyl, isopentyl, 2-methyl pentyl, 3-methylpentyl, 2-oxa-3-methylbutyl, 3-oxa-4-methylbutyl, 2-methyl-3-oxapentyl or 2-methyl-3-oxahexyl.
- R 14 preferably denotes cyanomethyl, cyanoethyl, cyanopropyl, cyanobutyl, cyanopentyl, cyano hexyl, 2-cyanopropyl, 2-cyanobutyl, carboxylmethyl, carboxylethyl, carboxylpropyl, carboxyl isopropyl, carboxylbutyl, carboxylpentyl, carboxylhexyl, carboxyl-2-methylpropyl, carboxyl-2-methylbutyl, sulfonylmethyl, sulfonylethyl, sulfonylpropyl, sulfonylbutyl, sulfonylpentyl, sulfonylhexyl, sulfonyl-2-methylpropyl,
- R 14 preferably also has the meaning of a phenyl group, which is preferably monosubstituted by cyano, cyanoalkyl, alkyl, alkoxy, alkoxyalkyl, carboxyl, carboxyalkyl, sulfonyl or sulfonylalkyl.
- the preferred meanings of these substituents correspond to the preferred alkyl groups and substituted alkyl groups indicated above.
- the substituent on the phenyl group is preferably in the p-position.
- R* and R** in the monomers of the formula V preferably denote H.
- R* and R 11 in formula VI preferably denote H.
- Chains having between 2 and 100, preferably 5 and 60, in particular between 10 and 30, monomer units are typically grafted onto the base material.
- sorbents in which exclusively one type of separation effector is present in the base matrix (“single mode” sorbents), and copolymers or “mixed mode” sorbents in which at least two different types of separation effectors are present in the sorbent.
- Single mode sorbents separate the dissolved substances (solutes) on the basis of one type of interaction between separation effector and solutes.
- mixed mode sorbents the separation of the dissolved substances (solutes) is based on different types of interactions between separation effector and solutes. Chromatographic methods of this type are known as mixed mode chromatography.
- the hydrophobicity of the base matrix is utilised in order to provide a sorbent which is suitable for mixed mode chromatography by introduction of an ionic separation effector.
- Mixed mode sorbents are also accessible by reaction of a base matrix with two different reactants, one of which introduces, for example, a hydrophobic group, the other an ionic group into the base matrix.
- the disadvantage of the two procedures consists in that the steric relationship between the different types of separation effectors are not defined.
- a mixed mode sorbent whose different separation effectors are bonded in a single ligand can be prepared, for example, from a chloropropyl-substituted support material by conversion of the chlorine derivatives into bromine derivatives followed by reaction with benzyldimethylamine: the ligand is bonded to the base matrix via a C3 chain. This is followed by a dimethylsubstituted ammonium group, to which a lipophilic benzyl radical is bonded.
- a reverse arrangement of hydrophobic and ionic separation effectors in a ligand is obtainable by reaction of aminopropyl-substituted support material with p-diethylaminobenzoic acid: in this ligand, the tertiary amino group is oriented distally of the base matrix, while in the former variant, the hydrophobic benzyl group is arranged distally.
- the use of the polymers according to the invention corresponds by way of example to affinity chromatography, ion exchange chromatography, hydrophobic interaction chromatography, size exclusion chromatography, chiral chromatography or mixed mode chromatography or liquid-liquid partition chromatography
- the polymers according to the invention in their native form or provided with separation effectors corresponding to the application, can be used in known chromatographic conceptional methods in which a sorbent is used. These methods can in principle be divided into discontinuous and continuous methods. Examples of discontinuous methods are mentioned in “Preparative Chromatography” (Ed. H. Schmidt-Traub, Wiley-VCH Verlag Weinheim, 2005, ISBN 3-527-30643-9, page 183-189). Further examples are flash chromatography, expanded bed chromatography, inter alia. Furthermore, the polymers according to the invention, in their native form or provided with separation effectors corresponding to the application, can be used in continuous methods, such as, for example, simulated moving bed chromatography. Further examples of continuous methods are described in “Preparative Chromatography” (Ed. H. Schmidt-Traub, Wiley-VCH Verlag Weinheim, 2005, ISBN 3-527-30643-9, page 190-204).
- Both the continuous methods and also the discontinuous methods can be carried out, depending on the objective, either isocratically or also using the gradient technique. It is known to the person skilled in the art how the sorbent according to the invention, native or provided with separation effectors, is to be employed for the desired objective in one of the said methods.
- the polymers according to the invention can also be used in thin-layer chromatography.
- DI water deionised water
- tributyl phosphate 4.2 g of sodium sulfate, 0.01 g of sodium laurylsulfonate and 2.1 g of polyvinyl alcohol as stabiliser are dissolved therein.
- the organic phase consisting of 96.25 g of butanediol monovinyl ether, 78.45 g of divinylethyleneurea, 175 g of dodecanol and 4.2 g of ⁇ , ⁇ ′-azobisisobutyronitrile, is added to the aqueous phase with stirring at room temperature after mixing and dissolution of the constituents.
- the suspension is stirred vigorously and subsequently warmed to 70° C. while flushing with nitrogen. After about 2 hours, the polymerisation is continued at 80° C. for 4 hours. After cooling to room temperature, the polymer is filtered off with suction, the porogen is removed by steam distillation, and the product is stored in 20% ethanolic solution.
- the average particle size is 80 ⁇ m.
- the swollen volume of the reaction product is 4.3 ml/g.
- FIG. 1 shows the binding behaviour of the proteins lysozyme and bovine serum albumin and of NaNO 3 .
- the abbreviation FP stands for FractoPrep®
- B/D stands for the copolymer according to the invention comprising butanediol monovinyl ether and divinylethyleneurea.
- An illustrative pore structure of the polymer according to the invention is shown by way of example in FIG. 2 through the experimentally determined distribution coefficient Kd of dextrans of various molecular weights, expressed by the viscosity radius of the dextrans.
- FIG. 3 shows the results. It is clearly evident that storage in dilute sodium hydroxide solution has absolutely no effect on the distribution coefficient.
- the polymer according to the invention consisting in this example of a copolymer of 65% by weight of butanediol monovinyl ether and 35% by weight of divinylethyleneurea in the form of spherical porous particles having an average particle diameter of 107 ⁇ m, is provided with cationic separation effectors by means of Ce(IV)-initiated graft polymerisation of 2-acrylamido-2-methyl-1-propanesulfonic acid corresponding to the procedure described on page 10 under Preparation Example A2 in WO 03/031062.
- the density of functional SO 3 groups in this polymer is obtained as 1195 ⁇ mol, based on 1 g of dry matter.
- the ion exchanger material prepared (BATCH: BDM SO3 DZ38) is introduced into a SuperformanceTM glass column, Goetec Labortechnik (depth of the sorbent bed 40 mm, diameter 10 mm) and equilibrated with a buffer consisting of 20 mM NaH 2 PO 4 , pH 6.0. 500 ⁇ l of a solution of 5 mg/ml of chymotrypsinogen A, 5 mg/ml of lysozyme and 5 mg/ml of cytochrome C in 20 mM NaH 2 PO 4 are applied to the column.
- FIG. 4 shows the associated chromatogram.
- the polymer according to the invention in its native form consisting of 65% by weight of butanediol monovinyl ether and 35% by weight of divinylethyleneurea (BATCH: BDM PS02/E), is introduced into a SuperformanceTM glass column, Goetec Labortechnik, having a diameter of 50 mm.
- the depth of the sorbent bed is 20 cm.
- the average particle diameter of the spherical porous polymer is 107 ⁇ m.
- the pressure drop over this column is measured as a function of the flow rate of a solution of 100 mM NaCl, 20 mM NaH 2 PO 4 , pH 6.0. Even at a very high flow rate of 1400 cm/h, the pressure drop is less than 3 bar.
- FIG. 5 shows the associated graphical representation of the pressure/flow measurement.
Landscapes
- Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Organic Chemistry (AREA)
- Analytical Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Polymers & Plastics (AREA)
- Addition Polymer Or Copolymer, Post-Treatments, Or Chemical Modifications (AREA)
- Treatment Of Liquids With Adsorbents In General (AREA)
- Solid-Sorbent Or Filter-Aiding Compositions (AREA)
- Graft Or Block Polymers (AREA)
- Cosmetics (AREA)
Abstract
The present invention relates to a hydrophilic crosslinked polymer, preferably in the form of porous particles, and to the preparation and use thereof. The polymer according to the invention is produced by polymerisation from chain-forming hydrophilic vinyl ethers and crosslinking, preferably heterocyclic divinyl ethers.
Description
- The present invention relates to a hydrophilic crosslinked polymer, preferably in the form of porous particles, and to the preparation and use thereof. The polymer according to the invention is produced by polymerisation from chain-forming hydrophilic vinyl ethers and crosslinking N,N′-divinyl compounds, preferably heterocyclic N,N′-divinyl compounds.
- Hydrophilic, crosslinked, polymeric support materials are frequently employed in areas such as chromatography, solid-phase synthesis or catalysis. In particular for use as support material in chromatography, polymers of this type should have the following properties:
-
- the materials should be sufficiently hydrophilic that, in particular, use in biochromatography is possible
- it should be possible for the materials to be modified, if required, by means of separation effectors in order that their separation properties can be matched to the particular separation problem
- the polymer must be sufficiently stable to hydrolysis
- in particular for applications in the area of high-pressure liquid chromatography, the material must have adequate pressure/flow stability
- the materials should have adequate pore sizes
- In addition, it should be possible to prepare polymers of this type in as simple and reproducible a manner as possible.
- WO 03/104294 discloses polyvinyl ethers, in particular for use in solid-phase synthesis. The polymers disclosed usually exhibit very strong swelling behaviour.
- DE 37 14 276 and DE 3344912 disclose crosslinked polymers based on vinyl acetates. In order to produce a hydrophilic polymer, the acetate groups have to be saponified subsequently.
- WO 9513861 discloses polyvinyl ethers for separation purposes which are prepared by cationic polymerisation. The cationic polymerisation described must be carried out in combination with complex protecting-group chemistry.
-
EP 0 266 503 discloses crosslinked polymers which are prepared by means of suspension polymerisation from monomers containing epoxide groups. The resultant polymers containing epoxide groups can then be used for the immobilisation of biologically active substances. -
EP 0 482 339 discloses a copolymer based on allyl monomers. In particular, the copolymer based onEP 0 482 339 comprising 1,2,-dihydroxy-3-allyloxypropane and N,N′-methylenebisacrylamide, commercially available under the name FractoPrep® (Merck KGaA, Germany), exhibits good properties for biochromatography with respect to hydrophilicity, stability to hydrolysis and pressure/flow stability. However, the disadvantage of this material is the complex preparation process by means of inverse suspension polymerisation. - The object of the present invention was therefore to provide a hydrophilic polymeric support material which meets the above-mentioned requirements with respect to porosity, hydrophilicity, stability to hydrolysis and pressure/flow stability and in addition is simple to prepare.
- It has been found that copolymerisation of hydrophilically substituted alkyl vinyl ethers using crosslinking, preferably heterocyclic N,N′-divinyl compounds enables the preparation of hydrophilic polymers having good properties, in particular for biochromatography. The polymers are preferably prepared by means of suspension polymerisation.
- The present invention therefore relates to a hydrophilic crosslinked polymer based on a copolymer at least comprising
- a) at least one hydrophilically substituted alkyl vinyl ether of the formula I
- where R1, R2, R3, independently of one another, can be H or C1 to C6 alkyl, preferably H or —CH3,
and R4 is a radical which carries at least one hydroxyl group and
b)
at least one crosslinking agent conforming to formula II and/or III and/or IV with - where X is a divalent alkyl radical having 2 to 5 C atoms, preferably 2 or 3 C atoms, in which one or more methylene groups which are not adjacent and are not located in the direct vicinity of N may be replaced by O, C═O, S, S═O, SO2, NH, NOH or N and one or more H atoms of the methylene groups may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH—(C1-C8)-alkyl, N—(C1-C8)-alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, and
- where Y1 and Y2 in formula III and IV are, independently of one another,
C1 to C10 alkyl or cycloalkyl, where one or more non-adjacent methylene groups or methylene groups which are not located in the direct vicinity of N may be replaced by O, O═O, S, S═O, SO2, NH, NOH or N and one or more H of the methylene groups may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or 01-C6-alkyl-OH,
or C6 to C18 aryl, where one or more H in the aryl system may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH and
A is a divalent alkyl radical having 2 to 5 C atoms, preferably 2 or 3 C atoms, in which one or more non-adjacent methylene groups or methylene groups which are not located in the direct vicinity of N may be replaced by O, C═O, S, S═O, SO2, NH, NOH or N and one or more H of the methylene groups may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH.
R4 in formula I is typically an alkyl radical, a cycloaliphatic radical or an aryl radical which carries at least one hydroxyl group. - In a preferred embodiment, R4 in formula I is
- a straight-chain or branched C1 to C10 alkyl radical, in which one or more non-adjacent methylene groups may be replaced by O, C═O, S, S═O, SO2, NH, NOH, N and/or in which one or more H atoms may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH and in which at least one OH group is present either on the C1 to C10 alkyl radical or on a substituent,
or a cycloaliphatic radical, typically having 5 to 10 C atoms, in which one or more non-adjacent methylene groups may be replaced by O, C═O, S, S═O, SO2, NH, NOH, N and/or in which one or more H atoms of the cycloaliphatic radical may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the cycloaliphatic ring or on a side chain or substituent, or a
C6 to C18 aryl radical, where one or more H atoms in the aryl radical may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the aryl radical or on a side chain or substituent, or a
C5 to C18 heteroaryl radical, where one or more H atoms in the heteroaryl radical may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the heteroaryl radical or on a side chain or substituent. - In a particularly preferred embodiment, R4 in formula I is
- a straight-chain or branched C1 to C10 alkyl radical, in which one or more non-adjacent methylene groups may be replaced by O, S, SO2 or NH and/or in which one or more H atoms may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, C1-C6-alkoxy or C1-C6-alkyl-OH and in which at least one OH group is present either on the C1 to C10 alkyl radical or on a substituent,
or a cycloaliphatic radical, typically having 5 to 10 C atoms, in which one or more non-adjacent methylene groups may be replaced by O, S, SO2 or NH and/or in which one or more H atoms of the cycloaliphatic radical may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the cycloaliphatic ring or on a side chain or substituent, or a
C6 to C14 aryl radical, where one or more H atoms in the aryl radical may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, C5-C10-aryl, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the aryl radical or on a side chain or substituent, or a
C6 to C14 heteroaryl radical, in which at least one N atom is present as heteroatom and where one or more H atoms in the heteroaryl radical may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, C5-C10-aryl, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the heteroaryl radical or on a side chain or substituent. - In a preferred embodiment, the hydrophilically substituted alkyl vinyl ether employed is a compound of the formula I in which R4 is a radical which carries a hydroxyl group.
- In a particularly preferred embodiment, the hydrophilically substituted alkyl vinyl ether employed is 1,2-ethanediol monovinyl ether, 1,3-propanediol monovinyl ether, 1,4-butanediol monovinyl ether, 1,5-pentanediol monovinyl ether, 1,6-hexanediol monovinyl ether or diethylene glycol monovinyl ether and the cycloaliphatic vinyl ether employed is cyclohexanedimethanol monovinyl ether.
- In a further preferred embodiment, the crosslinking agent employed is divinylethyleneurea (1,3-divinylimidazolin-2-one) or divinylpropyleneurea (1,3-divinyltetrahydropyrimidin-2-one).
- In another preferred embodiment, the polymer is porous having pore sizes between 2 and 200 nm.
- In another embodiment, the polymer is in the form of particles having a diameter between 3 and 300 μm.
- In another embodiment, the polymer has been derivatised by means of separation effectors.
- In a preferred embodiment, the polymer has been derivatised by means of structures which have been attached to the polymer by graft polymerisation.
- In a preferred embodiment, the polymer has been derivatised by means of structures which have been attached to the polymer by graft polymerisation with cerium(IV) catalysis.
- In a particularly preferred embodiment, the structures are copolymers which have been produced from two or more different monomers.
- The present invention also relates to a process for the preparation of polymers, characterised in that at least one hydrophilically substituted alkyl vinyl ether of the formula I and at least one crosslinking agent conforming to formula II and/or III and/or IV are copolymerised, with
- where R1, R2, R3, independently of one another, can be H or C1 to C6 alkyl, preferably H or —CH3,
and R4 is a radical which carries at least one hydroxyl group
and - where X is a divalent alkyl radical having 2 to 5 C atoms, preferably 2 or 3 C atoms, in which one or more methylene groups which are not adjacent and are not located in the direct vicinity of N may be replaced by O, C═O, S, S═O, SO2, NH, NOH or N and one or more H atoms of the methylene groups may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH—C1-C8-alkyl, N—C1-C8-alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, and
- where Y1 and Y2 in formula III and IV are, independently of one another,
C1 to C10 alkyl or cycloalkyl, where one or more non-adjacent methylene groups or methylene groups which are not located in the direct vicinity of N may be replaced by O, C═O, S, S═O, SO2, NH, NOH or N and one or more H of the methylene groups may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH,
or C6 to C18 aryl, where one or more H in the aryl system may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH and
A is a divalent alkyl radical having 2 to 5 C atoms, preferably 2 or 3 C atoms, in which one or more non-adjacent methylene groups or methylene groups which are not located in the direct vicinity of N may be replaced by O, C═O, S, S═O, SO2, NH, NOH or N and one or more H of the methylene groups may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH. - In a preferred embodiment, R4 in formula I is
- a straight-chain or branched C1 to C10 alkyl radical, in which one or more non-adjacent methylene groups may be replaced by O, C═O, S, S═O, SO2, NH, NOH, N and/or in which one or more H atoms may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH and in which at least one OH group is present either on the C1 to C10 alkyl radical or on a substituent,
or a cycloaliphatic radical, typically having 5 to 10 C atoms, in which one or more non-adjacent methylene groups may be replaced by O, C═O, S, S═O, SO2, NH, NOH, N and/or in which one or more H atoms of the cycloaliphatic radical may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the cycloaliphatic ring or on a side chain or substituent, or a
C6 to C18 aryl radical, where one or more H atoms in the aryl radical may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the aryl radical or on a side chain or substituent, or a
C5 to C18 heteroaryl radical, where one or more H atoms in the heteroaryl radical may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the heteroaryl radical or on a side chain or substituent. - In a preferred embodiment, a free-radical suspension polymerisation is carried out.
- In a particularly preferred embodiment, a free-radical suspension polymeri sation is carried out in water as suspension medium in the presence of at least one suspension stabiliser and optional further additives, preferably inorganic salts and interface-active compounds.
- In an embodiment, the temperature during the polymerisation is between 40 and 100° C.
- In a preferred embodiment, hydrophilically substituted alkyl vinyl ethers and crosslinking agents are employed in a weight ratio between 10:90 and 80:20.
- In a preferred embodiment, the polymerisation is carried out with addition of one or more porogens.
- The present invention also relates to a chromatography column, capillary, cartridge, stirred reactor or reactor containing a polymer according to the invention as sorbent or support material.
- The present invention also relates to the use of a hydrophilic crosslinked polymer according to the invention as sorbent in chromatography, as support material for the immobilisation of biologically or catalytically active substances or as support material for a solid-phase synthesis.
- The present invention also relates to polymers obtainable by the process according to the invention.
- Further combinations and preferred embodiments are disclosed in the patent claims.
-
FIG. 1 shows the retention behaviour of the polymer according to the invention on application of proteins. Further details are given in Example 2. -
FIG. 2 shows the experimentally determined distribution coefficient Kd of dextrans having different molecular weights. Further details are given in Example 3. -
FIG. 3 shows the storage stability of a polymer according to the invention in dilute sodium hydroxide solution. Further details are given in Example 4. -
FIG. 4 shows the separation of a protein mixture on a polymer according to the invention provided with cationic separation effectors. Further details are given in Example 5. -
FIG. 5 shows the pressure/flow behaviour of the polymer according to the invention in a chromatography column. - In accordance with the invention, the term “cycloaliphatic radical” denotes a saturated hydrocarbon radical in which all or at least some of the hydrocarbon units are a constituent of a cyclic structure.
- In accordance with the invention, N(C1-C8)alkyl2 denotes a nitrogen which is substituted by at least two alkyl radicals. The two alkyl radicals here may have, independently of one another, 1 to 8 C atoms in straight-chain or branched form.
- In accordance with the invention, a heteroaryl radical is an at least partially aromatic radical which differs from an aryl radical in that one or more non adjacent C atoms have been replaced by N, S or O. It is clear to the person skilled in the art that, owing to the valences, the heteroatoms may optionally be substituted by, for example, H, C1-C6-alkyl or C1-C6-alkyl-OH.
- The polymer according to the invention is a hydrophilic, crosslinked polymer based on a copolymer at least comprising
- a) at least one hydrophilically substituted alkyl vinyl ether of the formula I
- where the radicals R1, R2, R3 and R4 have the above-mentioned meanings, and
b)
at least one crosslinking agent conforming to formula II and/or III and/or IV - where the substituents X, A, Y1 and Y2 have the above-mentioned meanings.
- This means the polymer is formed by copolymerisation of at least one compound from the group of the hydrophilically substituted alkyl vinyl ethers of the formula I and at least one compound from the group of the crosslinking agents of the formula II and/or III and/or IV. Preferably, only one compound from the group of the hydrophilically substituted alkyl vinyl ethers of the formula I and one compound from the group of the crosslinking agents of the formula II, Ill or IV is employed. However, it is also possible to employ one or more compounds from the group of the hydrophilically substituted alkyl vinyl ethers of the formula I and/or one or more compounds from the group of the crosslinking agents of the formula II and/or III and/or IV. Furthermore, further polymerisable compounds which are copolymerised into the polymer backbone can be added to the polymerisation mixture. These are typically compounds having at least one polymerisable double bond. Preferably, only one compound from the group of the hydrophilically sub stituted alkyl vinyl ethers of the formula I and one compound from the group of the crosslinking agents of the formula II, Ill or IV is employed.
- In a preferred embodiment, the hydrophilically substituted alkyl vinyl ether employed is a compound of the formula I in which R4 is a radical which carries a hydroxyl group.
- In a preferred embodiment, the hydrophilically substituted alkyl vinyl ether employed is 1,2-ethanediol monovinyl ether, 1,3-propanediol monovinyl ether, 1,4-butanediol monovinyl ether, 1,5-pentanediol monovinyl ether, 1,6-hexanediol monovinyl ether or diethylene glycol monovinyl ether and the cycloaliphatic vinyl ether employed is cyclohexanedimethanol monovinyl ether, particularly preferably 1,4-butanediol monovinyl ether, 1,5-pentanediol monovinyl ether, diethylene glycol monovinyl ether or cyclohexanedimethanol monovinyl ether.
- The crosslinking agents employed are preferably compounds of the formula II. Preference is given to the use of divinylpropyleneurea (1,3-divinyl-tetrahydropyrimidin-2-one) or particularly preferably divinylethyleneurea (1,3-divinylimidazolin-2-one).
- The proportion of the hydrophilically substituted alkyl vinyl ethers with respect to the weight of the polymer is typically between 1% (by weight) and 90% (by weight) or a maximum proportion by weight of the alkyl vinyl ether which corresponds to a molar ratio of 2:1, based on a bifunctional crosslinking agent, if the alkyl vinyl ether does not homopolymerise. The proportion of the hydrophilically substituted alkyl vinyl ethers is preferably between 10 and 80% (% by weight), particularly preferably between 35 and 60%. Accordingly, the proportion of the crosslinking agent is between 10 and 99 (% by weight), preferably between 20 and 90%, particularly preferably between 40 and 65%.
- The process-engineering design of the copolymerisation of unsaturated compounds is known to the person skilled in the art. To this end, a free-radical polymerisation is carried out. A suspension polymerisation initiated by free radicals, also known as bead polymerisation, is preferably carried out. Descriptions of the way in which a polymerisation of this type is carried out are given, for example, in EP 1179732 A2 (
general description page 4, lines 18 to 26, andpage 5, line 3, to page 6, line 9, and, in particular, Example 1) or in EP 0006199 B1 (general description page 3, line 19, topage 4, line 13, and, in particular, Example 1). Depending on the dissolution behaviour of the monomers, an oil-in-water suspension polymerisation (normal-phase polymerisation) is carried out in a particularly simple procedure in the case of water-insoluble or sparingly water-soluble monomers, which are also known as oil. In the case of water-soluble monomers, by contrast, the latter are dissolved in water and suspended and polymerised in a water-immiscible or only sparingly water-miscible organic solvent (inverse polymerisation). - It has been found that, in particular in the case of the use of divinylethyleneurea or divinylpropyleneurea in combination with the preferred hydrophilically substituted alkyl vinyl ethers, such as 1,4-butanediol monovinyl ether, 1,5-pentanediol monovinyl ether, diethylene glycol monovinyl ether or cyclohexanedimethanol monovinyl ether, a (normal-phase) suspension polymerisation can be carried out. This offers the advantage of a very simple preparation process for the polymers according to the invention since the hydrophilic polymer is obtained in a single reaction step without having to carry out the complex inverse suspension polymerisation. A further major advantage of the hydrophilically substituted alkyl vinyl ethers employed in accordance with the invention is that a subsequent hydrolysis step does not have to be carried out in order to produce a hydrophilic polymer. Instead, the use of the hydroxyl-containing vinyl ethers as starting materials results in the direct formation of a hydroxyl-containing polymer.
- The suspension polymerisation is carried out in a known manner.
- The suspension medium used in the normal-phase suspension polymerisation is typically water.
- One or more stabilisers and/or one or more interface-active compounds can, as is known, be added to the suspension medium.
- Stabilisers which can be employed are a multiplicity of water-soluble macromolecular compounds or also finely divided inorganic compounds.
- Examples of macromolecular stabilisers are polyvinylpyrrolidone, polyacrylates, polycarboxylates, polyacrylamide, polyvinyl alcohol, hydroxyalkylcellulose, methylcellulose or polyethylene glycols. Preference is given to the use of polyvinyl alcohols.
- Inorganic compounds which can be employed as stabilisers are, for example, calcium phosphate or magnesium hydroxide.
- Salts, ionic liquids or buffer substances, such as, for example, Na2HPO4 and NaH2PO4, can furthermore be added to the aqueous phase.
- Suitable interface-active compounds are, in particular, anionic and nonionic surfactants, such as ethoxylates of long-chain alcohols, ethoxylated mono-, di- and trialkylphenols, alkali metal and ammonium salts of C12-C18 alkylsulfonic acids or arylsulfonic acids.
- For carrying out the polymerisation according to the invention, the organic phase, also known as the oil phase, is suspended in the aqueous phase. After initiation and performance of the polymerisation, the polymer can typically be filtered off or removed from the reaction mixture in another manner. Residues of solvents or other additives can be removed by washing with suitable solvents or, for example, steam distillation.
- In an embodiment, the oil phase comprises inert solvents (porogens) in order to set the desired pore sizes, in addition to the monomers.
- Suitable organic solvents (porogens) are, for example, aliphatic hydro-carbons (C6-C18), such as, for example, hexane, dodecane or benzine mixtures, cycloaliphatic compounds, such as cyclohexane, aromatic hydrocarbons, such as toluene, ethylbenzenes or xylenes, or alcohols, such as aliphatic C4-C12-alcohols, for example heptanol, dodecanol, cyclohexanol, polyethylene glycols or polypropylene glycols of various molecular weight, or esters of aliphatic carboxylic acids, such as butyl acetate or propyl propionate, or esters of aromatic carboxylic acids, such as propyl benzoate, or esters, such as butyl glycol acetate or glycerol triacetate, or ethers, such as di-n-butyl ether, di-n-amyl ether, diphenyl ether, ethylene glycol monophenyl ether and ethylene glycol diethyl ether.
- These solvents can be employed individually or as a mixture of two or more solvents. They can either act as swelling agent or precipitant for the polymers and in this way influence the porosity.
- Examples of swelling agents for the polymers according to the invention are lower alcohols, aliphatic esters or aromatic hydrocarbons, such as toluene. Examples of precipitants are aliphatic hydrocarbons, such as dodecane or benzine mixtures. The porosity of the polymers can be fixed through the type and amount of the solvents in combination with the degree of crosslinking of the polymer.
- Furthermore, one or more initiators acting as free radicals are typically added to the organic phase.
- Examples of free-radical initiators are organic peroxides, such as di-tertbutyl peroxide, dibenzoyl peroxide, bis(o-methylbenzoyl) peroxide, tert-butyl hydroperoxide, cumene hydroperoxide, diisopropyl peroxide dicarbonate, cyclohexanone peroxide, or aliphatic azo compounds, such as α,α′-azobisisobutyronitrile, azobiscyanovaleric acid, 1,1′-azocyclohexane-1,1′-dicarbo-nitrile or azodicarboxamide. Preference is given to α,α′-azobisisobutyronitrile.
- Initiators which can be activated by means of radiation, for example UV radiation, can also be employed in accordance with the invention as free-radical initiators.
- The phase ratios in the suspension polymerisation can typically be varied in the volume ratio between 1:20 (oil phase to aqueous phase) and 2:1, ratios between 1:5 and 1:1, particularly preferably between 1:3 and 1:1, are preferred.
- The ratio of organic solvent to monomer is typically between 1:20 and 4:1, preferably between 1:5 and 3:1, particularly preferably between 1:1 and 2.5:1.
- The concentration of the initiator, based on the amount of monomer, is typically between 0.01% (by weight) and 10% (by weight), preferably between 0.1 and 7% (by weight) and particularly preferably between 0.2 and 7% (by weight).
- The concentration of stabilisers, based on the amount of suspension medium, generally water, is between 0.01 and 7% (by weight), typically between 0.02 and 5% (by weight), particularly preferably between 0.05 and 2.5% (by weight).
- The suspension polymerisation is then carried out by mixing the two phases with vigorous stirring. In addition, the temperature of the mixture is typically kept at 20 to 100° C., preferably 40 to 100° C., for a period of 1 to 20 hours. The mixture is typically heated at 40 to 100° C. over a period of 0.5 to 5 hours and subsequently post-polymerised again for several hours at 70 to 100° C. The mixture should be stirred throughout the time. These temperature data relate to a system in which water is used as suspension medium. On use of other polar solvents instead of water, other temperature ranges may be possible or appropriate, depending on the boiling point of the solvent.
- It is generally advantageous to carry out the polymerisation according to the invention in a protective-gas atmosphere.
- Finally, the mixture is generally cooled to room temperature, the organic solvents are removed, if they were added during the polymerisation, and the product is stored in water, an aqueous buffer or a mixture of water with an organic solvent, such as ethanol.
- If necessary, the polymer can be brought to the desired particle-size distribution by sieving or another type of classification.
- If an inverse suspension polymerisation or an emulsion polymerisation is to be carried out instead of the normal-phase suspension polymerisation, it is known to the person skilled in the art how to modify the composition of the reaction solutions.
- The polymerisation according to the invention can likewise be carried out continuously. The particle production here is particularly preferably carried out using static mixers, in particular micromixers or caterpillar mixers. In these mixers, the spatial distribution of two components to be mixed is influenced, with utilisation of the flow energy, by the flow management, for example by separation and recombination, twisting, distortion and expansion, so that large interfaces are formed for diffusive exchange between the components and a particularly uniform particle-size distribution can be achieved due to the high shear forces. Different mixers and their structure are known. For example, systems of this type are described in EP 1177243.
- In order to carry out the reactions, the aqueous phase flows, for example, continuously with a defined volume flow through the micromixer. The oil phase is subsequently fed in via the second inlet, likewise with a defined, precisely set volume flow. The ratio of the two volume flows can be in a broad range and depends on the desired particle size, the nature and distribution of the particle sizes, the viscosity of the aqueous and the oil phase, depending on the reaction temperature, and the reaction kinetics and the requisite ratio of the porogens in the end mixture in order to achieve a suitable pore structure. The volume flow ratio between the aqueous phase and the oil phase is preferably 1:10 to 100:1, in particular 2:1 to 20:1. The suspension exiting from the static mixer is preferably polymerised in a continuously operating reactor.
- It is likewise possible to reverse the procedure described above. For example, for the polymerisation of particularly hydrophilic monomers, the latter are supplied via the aqueous phase. Further details on the synthesis of bead polymers with the aid of micromixers are given in EP 1177243.
- It is furthermore known to the person skilled in the art how the particle size of the resultant polymer can be influenced, inter alia through the nature and amount of the addition of suspension stabilisers and interface-active compounds and the stirring intensity. In general, particle diameters between 3 μm and 500 μm, preferably between 3 μm and 300 μm, can be produced by means of suspension polymerisation, depending on the type of stirrer and the rotational speed. The particles can have an irregular shape or can preferably be spherical.
- If the polymers according to the invention are prepared by means of emulsion polymerisation, particle diameters, for example, between 20 nm and 3000 nm, preferably between 100 and 500 nm, can be produced.
- The polymers according to the invention can furthermore be prepared or exist in the form of membranes, fibres, hollow fibres, a coating or as a monolithic moulding. Monolithic mouldings are three-dimensional bodies, for example in cylindrical form.
- The polymers according to the invention can equally be employed as composite materials, i.e., for example, as a coating, for example of inorganic particles, or a moulding, or mixed with, for example, inorganic constituents. An example thereof are particles of the polymer according to the invention which can themselves be magnetised by copolymerisation of magnetisable particles or of a magnetisable core.
- The polymers according to the invention are preferably in the form of nonporous or particularly preferably porous particles. Depending on the type and amount of the porogenic solvent employed, the pore sizes can typically be between 2 and 300 nm. In accordance with the invention, pore size denotes pore radius. Preference is given to pore sizes or pore radii between 2 and 200 nm. In particular, if the polymers according to the invention are in the form of monolithic mouldings, they can also have pore sizes up to 1000 nm.
- The pore distribution in the materials according to the invention can be monomodal, bimodal or oligomodal, preferably monomodal or bimodal.
- The pore sizes are preferably measured by means of size exclusion chromatography (SEC). A measurement example in this respect is given in Example 3 and
FIG. 2 . - The hydrophilic, crosslinked polymers according to the invention are particularly suitable as sorbents in chromatography, as support materials for the immobilisation of biologically and/or catalytically active substances or as support materials for solid-phase syntheses of, for example, biopolymers, such as nucleic acids or peptides, or for combinatorial chemistry.
- The materials according to the invention are distinguished by good stability to hydrolysis, in particular in basic medium, and by good pressure/flow stability. Furthermore, their high hydrophilicity makes them particularly suitable for biochromatographic methods. The examples contain additional experimental data on these properties.
- The polymers according to the invention are particularly suitable as sorbents. For this purpose, they are introduced in a known manner into chromatography columns or capillaries. The polymer according to the invention can equally be packed in cartridges for chromatographic or other purposes. The present invention therefore also relates to chromatography columns, capillaries or cartridges which contain a polymer according to the invention as sorbent or support material.
- The polymers according to the invention can be employed in their native form, i.e. without further derivatisation steps, for example, depending on the porosity set, for size exclusion chromatography (SEC) or for reduction of the salt content of solutions if these solutions contain target molecules whose molecular weight differs significantly from the molecular weight of the salt,
- or provided with other or additional functionalities by one or more derivatisation steps. In particular, they can be derivatised by means of separation effectors. These separation effectors may be either ionic or neutral or have a preferred affinity or selectivity for one or more target molecules.
- The covalent bonding of the separation effectors generally takes place via the functional groups present on the polymer, such as, for example, hydroxyl groups, for example with formation of an ester or preferably an ether function, directly or via a linker or spacer. In another preferred embodiment, the linking to the base material takes place via graft polymerisation, such as, for example, a cerium(IV)-catalysed graft polymerisation with formation of a C—C link to the base material.
- Separation effectors are known to the person skilled in the art in the area of chromatography. Separation effectors are substituents which can be intro duced into the support material as early as during the synthesis of the base material or subsequently and influence the surface properties of the support material. In particular, targeted derivatisation of support materials by means of separation effectors produces support materials having certain chromatographic properties. In particular, separation effectors can have the following terminal groups:
- a) an ionic or ionisable group, for example
- in which
R7 and R8, independently of one another, -
- H, alkyl having 1-5 C atoms
and
R9 alkyl having 1-5 C atoms
with the proviso that, if X=—N+R7R8R9, R7 and R8 cannot be H, - guanidinium
- SO3 −
- carboxylic acids
- H, alkyl having 1-5 C atoms
- b) a hydrophobic group, for example —OR10 or —NHR10, where R10 denote C1-C20-alkyl, C6-C25-aryl, C7-C25-alkylaryl or C7-C25-arylalkyl, and where these radicals may also have been derivatised by means of nitrile or C1-C5-alkoxy, and where, in addition, one or more non-adjacent CH2 groups may have been replaced by NH or O or, in addition, one or more CH groups may have been replaced by N;
- c) a metal chelate group;
- d) a thiophilic radical;
- e) a chiral radical.
- f) biomolecules, such as proteins (for example antibodies, protein A), peptides, amino acids, nucleic acids, saccharides, biotin, etc.
- g) organic radicals which have increased affinity or selectivity for certain target molecules (for example mono- or bistriazine-based separation effectors in accordance with WO 9710887 and WO 04/052870).
- Thiophilic radicals are disclosed, for example, in
EP 0 165 912. - If it is intended firstly to provide the polymer with a universal linker, it can, for example for the introduction of epoxide groups, be reacted with glycidyl compounds, such as butanediol diglycidyl ether.
- Furthermore, the polymer according to the invention can, as base material, be provided by graft polymerisation with tentacle-like structures, which can in turn carry the corresponding separation effectors or be functionalised by means of the latter. The grafting is preferably carried out in accordance with
EP 0 337 144. The chain produced is linked to the base material via a monomer unit. To this end, the base material according to the invention is suspended in a solution of monomers, preferably in an aqueous solution. The grafting-on of the polymeric material is effected in the course of a conventional redox polymerisation with exclusion of oxygen. The polymerisation catalyst employed is cerium(IV) ions, since this catalyst forms free-radical sites on the surface of the base material, from which the graft polymerisation of the monomers is initiated. - The polymerisation is terminated by termination reactions involving the cerium salts. For this reason, the (average) chain length can be influenced by the concentration ratios of the base material, the initiator and the mono mers. Furthermore, uniform monomers or also mixtures of different monomers can be employed; in the latter case, grafted copolymers are formed.
- Suitable monomers for the preparation of the graft polymers are monomers conforming to formula V or VI.
-
CR*R**═CR11—Y (formula V) - These monomers are (meth) acrylic acid (Y=—COOH), (meth)acrylic acid derivatives where
- allylamines (Y=—CH2NH2, —CH2NR12R13), (meth)acrylonitriles (Y=—CN), acroleins (Y=—CHO), vinylcarboxylates (Y=−OCOCHR15R16) or vinylenecarbonates of the formula VI.
- All these monomers are polymerisable substances containing reversibly bonding groups which can be polymerised by means of free radicals in aqueous solution and which may be neutral, acidic or basic. If the monomers employed are vinylenecarbonates of the formula VI or vinylcarboxylates CR*R**═CR11—OCOCHR15R16 of the formula V, the product obtained is preferably subsequently converted into a separating material containing hydroxyl groups. This conversion into a hydroxyl phase is achieved by a mild alkaline or acidic saponification known per se. For example, the reaction can be carried out using methanolic K2CO3 solution at room temperature, described, for example, by Y. Tezuka et al., in Macromol. Chem. 186, 685-694 (1985).
- In the formulae V and VI, R11 preferably denotes H, i.e. the acrylic acid derivatives are preferred.
- Y in formula V preferably denotes
- —COCHR15R16 or —CH2NH2, secondarily preferably
- R15 and R16, independently of one another, denote H or an alkyl group having up to 5 C atoms. At least one of the radicals R15 and R16 is preferably H. The following radicals are particularly preferred: acetoxy, propionyloxy, butyryloxy, valeryloxy and hexanoyloxy radical.
Z in formula V denotes —OR14, —OH or —NR12R13, preferably —NR12R13. - Preference is given here to compounds in which Z denotes —NR12R13 and one of the radicals R12 and R13 is H.
- The radicals R12 and/or R13 preferably denote an alkyl, phenyl, phenylalkyl or alkylphenyl group, where the alkyl and/or phenyl group may be mono- or polysubstituted, preferably mono- or disubstituted, particularly preferably monosubstituted, by an alkoxy, cyano, amino, mono- or dialkylamino, trialkylammonium, carboxyl, sulfonyl, acetoxy or acetamino radical.
- The radicals R12 and/or R13 preferably denote alkyl, alkoxyalkyl, cyanoalkyl, aminoalkyl, mono- or dialkylaminoalkyl, trialkylammoniumalkyl, carboxyalkyl or sulfonylalkyl having up to 10 C atoms, preferably up to 6 C atoms, particularly preferably up to 4 C atoms in the alkyl group, which may be linear or branched. R12 and/or R13 accordingly preferably denote methyl, ethyl, propyl, butyl, pentyl, hexyl, methoxymethyl, ethoxymethyl, 2-methoxyethyl, 2-, 3- or 4-oxapentyl, 2-, 3-, 4- or 5-oxahexyl, 2-, 3-, 4-, 5- or 6-oxaheptyl, isopropyl, 2-butyl, isobutyl, 2-methylbutyl, isopentyl, 2-methylpentyl, 3-methylpentyl, 2-oxa-3-methylbutyl, 3-oxa-4-methylbutyl, 2-methyl-3-oxa pentyl, 2-methyl-3-oxahexyl, furthermore also heptyl, octyl, nonyl or decyl. Preference is furthermore also given to alkyl groups which are substituted by a cyano, carboxyl or sulfonyl group. Accordingly, R12 and/or R13 preferably denote cyanomethyl, cyanoethyl, cyanopropyl, cyanobutyl, cyanopentyl, cyanohexyl, 2-cyanopropyl, 2-cyanobutyl, carboxylmethyl, carboxylethyl, carboxylpropyl, carboxyl isopropyl, carboxylbutyl, carboxylpentyl, carboxylhexyl, carboxyl-2-methylpropyl, carboxyl-2-methylbutyl, sulfonylmethyl, sulfonylethyl, sulfonylpropyl, sulfonylbutyl, sulfonylpentyl, sulfonylhexyl, sulfonyl-2-methylpropyl, sulfonyl-2-methylbutyl, sulfonyl-3-methylbutyl, sulfonyl-2-methylpentyl, sulfonyl-3-methylhexyl or sulfonyl-2-ethylpentyl. The alkyl groups are furthermore preferably monosubstituted by an amino, mono- or dialkylamino or trialkylammonium group. The alkyl groups here may be identical or different and have up to 10, preferably up to 6 C atoms, particularly preferably up to 4 C atoms, and accordingly preferably denote dimethylaminoethyl, diethylaminoethyl, methylaminoethyl, methylamino propyl, dimethylaminopropyl, ethylaminoethyl, propylaminoethyl, propylaminopropyl, dipropylaminoethyl, dipropylaminobutyl, diethylaminoethyl, trimethylammoniumethyl, trimethylammoniumpropyl, trimethylammoniumbutyl, triethylammoniumethyl, triethylammoniumpropyl, triethylammoniumethyl, aminoethyl, aminopropyl, aminobutyl or aminopentyl. All these alkyl and substituted alkyl groups are likewise preferred as substituents on the phenyl group.
- Preference is also given for R12 and/or R13 to a sulfone sulfide of the structure —(CH2)n— SO2—(CH2)—S—(CH2)nOH where n=2, 3, 4, 5 or 6, preferably 2, 3 or 4.
- R12 and/or R13 preferably also has the meaning of a phenyl group, which is preferably monosubstituted by cyano, cyanoalkyl, amino, aminoalkyl, mono- or dialkylamino, alkyl, alkoxy, alkoxyalkyl, mono- or dialkylaminoalkyl, trialkylammonium or trialkylammoniumalkyl, carboxyl, carboxyalkyl, sulfonyl or sulfonylalkyl. The preferred meanings of these substituents correspond to the preferred alkyl groups and substituted alkyl groups indicated above. The substituent on the phenyl group is preferably in the p-position. p-Acetoxyphenyl, p-aminophenyl or p-acetaminophenyl are likewise preferred meanings for R12 and/or R13.
- Preference is furthermore given for R12 and/or R13 to an alkylphenyl or phenylalkyl group, where the preferred meanings indicated for the alkyl, substituted alkyl or substituted phenyl groups shall likewise apply. Accordingly, the following substituted phenyl groups, for example, are regarded as particularly preferred: 4-cyanophenyl, 4-alkylphenyl, 4-(N,N-dimethylamino)phenyl, 4-(N,N-dialkylaminoethyl) phenyl, 4-ethoxyphenyl, 4-ethoxyethylphenyl, 4-trialkylammoniumphenyl, 4-carboxylphenyl, 4-sulfonylphenyl, phenylethyl, 4-(N-ethylamino) phenylpropyl or 4-cyanophenylethyl.
- Furthermore, preferred monomers of the formula V are those in which R12 and/or R13 denote a cyclic or bicyclic radical, which may be aromatic or saturated, having 5-10 C atoms in which one or more CH or CH2 groups have been replaced by N or NH, N or NH and S, or N or NH and O.
- R12 and/or R13 accordingly preferably also denote a pyridine radical, imidazolyl radical, indolyl radical, furthermore preferably a pyrrole, pyrimidine, pyrazine, quinoline or isoquinoline radical.
- R12 and/or R13 may also, for example, denote a thiazole, thiadiazole, morpholine, triazine, piperazine, benzothiazole, purine, pyrazole, triazole, pyrrolidine or isoxazole radical.
- Particular preference is given here to the aromatic, heterocyclic radicals. The radicals R12 and R13 must, in order to obtain suitable exchangers, be matched to one another in such a way that either both radicals contain an acidic or basic group or alternatively one of the radicals is neutral. The person skilled in the art is presented with no difficulty in assigning the groups correspondingly and thus assembling suitable radicals for R12 and R13, depending on the function and task of the desired ion exchanger. One of the two radicals R12 and R13 is preferably a neutral radical.
- R14 preferably denotes alkyl, alkoxyalkyl, cyanoalkyl, carboxyalkyl or sulfonylalkyl having up to 10 C atoms, preferably having up to 6 C atoms, particularly preferably having up to 4 C atoms, in the alkyl group, which may be linear or branched. R14 accordingly preferably denotes methyl, ethyl, propyl, butyl, pentyl, hexyl, methoxymethyl, ethoxymethyl, 2-methoxyethyl, 2-, 3- or 4-oxapentyl, isopropyl, 2-butyl, isobutyl, 2-methylbutyl, isopentyl, 2-methyl pentyl, 3-methylpentyl, 2-oxa-3-methylbutyl, 3-oxa-4-methylbutyl, 2-methyl-3-oxapentyl or 2-methyl-3-oxahexyl.
- Preference is furthermore also given to alkyl groups which are substituted by a cyano, carboxyl or sulfonyl group. Accordingly, R14 preferably denotes cyanomethyl, cyanoethyl, cyanopropyl, cyanobutyl, cyanopentyl, cyano hexyl, 2-cyanopropyl, 2-cyanobutyl, carboxylmethyl, carboxylethyl, carboxylpropyl, carboxyl isopropyl, carboxylbutyl, carboxylpentyl, carboxylhexyl, carboxyl-2-methylpropyl, carboxyl-2-methylbutyl, sulfonylmethyl, sulfonylethyl, sulfonylpropyl, sulfonylbutyl, sulfonylpentyl, sulfonylhexyl, sulfonyl-2-methylpropyl, sulfonyl-2-methylbutyl, sulfonyl-3-methylbutyl, sulfonyl-2-methylpentyl, sulfonyl-3-methylhexyl or sulfonyl-2-ethylpentyl. All these alkyl and substituted alkyl groups are likewise preferred as substituents on the phenyl group.
- R14 preferably also has the meaning of a phenyl group, which is preferably monosubstituted by cyano, cyanoalkyl, alkyl, alkoxy, alkoxyalkyl, carboxyl, carboxyalkyl, sulfonyl or sulfonylalkyl. The preferred meanings of these substituents correspond to the preferred alkyl groups and substituted alkyl groups indicated above. The substituent on the phenyl group is preferably in the p-position.
- R* and R** in the monomers of the formula V preferably denote H.
- R* and R11 in formula VI preferably denote H.
- Chains having between 2 and 100, preferably 5 and 60, in particular between 10 and 30, monomer units are typically grafted onto the base material.
- As already mentioned above, there are sorbents in which exclusively one type of separation effector is present in the base matrix (“single mode” sorbents), and copolymers or “mixed mode” sorbents in which at least two different types of separation effectors are present in the sorbent. “Single mode” sorbents separate the dissolved substances (solutes) on the basis of one type of interaction between separation effector and solutes. On use of mixed mode sorbents, the separation of the dissolved substances (solutes) is based on different types of interactions between separation effector and solutes. Chromatographic methods of this type are known as mixed mode chromatography.
- In the simplest case, for example, the hydrophobicity of the base matrix is utilised in order to provide a sorbent which is suitable for mixed mode chromatography by introduction of an ionic separation effector. Mixed mode sorbents are also accessible by reaction of a base matrix with two different reactants, one of which introduces, for example, a hydrophobic group, the other an ionic group into the base matrix. The disadvantage of the two procedures consists in that the steric relationship between the different types of separation effectors are not defined.
- This disadvantage is avoided in the case of mixed mode sorbents in which the different separation effectors are combined in a ligand. This ligand is bonded to the base matrix.
- Both types of mixed mode sorbents mentioned above are described in a review article by L. W. McLaughlin (1989) in Chem. Rev. 89, 309-319. A mixed mode sorbent whose different separation effectors are bonded in a single ligand can be prepared, for example, from a chloropropyl-substituted support material by conversion of the chlorine derivatives into bromine derivatives followed by reaction with benzyldimethylamine: the ligand is bonded to the base matrix via a C3 chain. This is followed by a dimethylsubstituted ammonium group, to which a lipophilic benzyl radical is bonded. A reverse arrangement of hydrophobic and ionic separation effectors in a ligand is obtainable by reaction of aminopropyl-substituted support material with p-diethylaminobenzoic acid: in this ligand, the tertiary amino group is oriented distally of the base matrix, while in the former variant, the hydrophobic benzyl group is arranged distally.
- Further embodiments of such ligands having two separation effectors are disclosed in the following documents: WO 96/09116, WO 97/29825, WO 00/69872 and WO01/38228. The patent application EP 04028798.9 discloses more complex ligands having more than one separation effector each, and anion exchange groups whose nitrogen is in the form of an aza derivative of polycyclic structures, for example the quininuclidine system. Examples thereof are:
- The possible applications of native polymers or polymers according to the invention provided with separation effectors may include, for example,
-
- selective, partially selective or nonselective binding or adsorption of one or more target components with the aim of removal from a matrix
- selective, partially selective or nonselective binding or adsorption of one or more secondary components with the aim of removal of the secondary component from a matrix
- separation of a substance mixture without binding or adsorption of one or more components merely on the basis of the molecular size by size exclusion chromatography
- isolation, enrichment and/or depletion of biopolymers from natural sources
- isolation, enrichment and/or depletion of biopolymers from recombinant sources
- isolation, enrichment and/or depletion of biopolymers from immortalised cell lines and culture supernatants thereof or from plants, in particular transgenic plants
- isolation, enrichment and/or depletion of biopolymers from B-cell lines and derivatives thereof, lymph cells and hybridoma cell lines and culture supernatants thereof
- isolation, enrichment and/or depletion of proteins and peptides
- isolation, enrichment and/or depletion of enzymes
- isolation, enrichment and/or depletion of monoclonal and polyclonal anti bodies and naturally occurring or recombinant antibody fragments
- isolation, enrichment and/or depletion of phosphorylated peptides/proteins and nucleic acids
- isolation, enrichment and/or depletion of food additives
- isolation, enrichment and/or depletion of mono- and polysaccharides
- isolation, enrichment and/or depletion of glycosylated proteins
- isolation, enrichment and/or depletion of single-stranded or doublestranded DNA
- isolation, enrichment and/or depletion of plasmid DNA
- isolation, enrichment and/or depletion of RNA
- isolation, enrichment and/or depletion of viruses
- isolation, enrichment and/or depletion of host cell proteins
- isolation, enrichment and/or depletion of oligo- and polynucleotides
- isolation, enrichment and/or depletion of liposomes
- isolation, enrichment and/or depletion of products from blood and milk
- isolation, enrichment and/or depletion of low-molecular-weight active pharmaceutical ingredients (APIs)
- removal of an API from an API drug carrier (for example an API/liposome adduct or an API/nanoparticle adduct)
- isolation, enrichment and/or depletion of enantiomers
- Depending on the type of separation effectors, the use of the polymers according to the invention corresponds by way of example to affinity chromatography, ion exchange chromatography, hydrophobic interaction chromatography, size exclusion chromatography, chiral chromatography or mixed mode chromatography or liquid-liquid partition chromatography
- The polymers according to the invention, in their native form or provided with separation effectors corresponding to the application, can be used in known chromatographic conceptional methods in which a sorbent is used. These methods can in principle be divided into discontinuous and continuous methods. Examples of discontinuous methods are mentioned in “Preparative Chromatography” (Ed. H. Schmidt-Traub, Wiley-VCH Verlag Weinheim, 2005, ISBN 3-527-30643-9, page 183-189). Further examples are flash chromatography, expanded bed chromatography, inter alia. Furthermore, the polymers according to the invention, in their native form or provided with separation effectors corresponding to the application, can be used in continuous methods, such as, for example, simulated moving bed chromatography. Further examples of continuous methods are described in “Preparative Chromatography” (Ed. H. Schmidt-Traub, Wiley-VCH Verlag Weinheim, 2005, ISBN 3-527-30643-9, page 190-204).
- Both the continuous methods and also the discontinuous methods can be carried out, depending on the objective, either isocratically or also using the gradient technique. It is known to the person skilled in the art how the sorbent according to the invention, native or provided with separation effectors, is to be employed for the desired objective in one of the said methods.
- The polymers according to the invention can also be used in thin-layer chromatography.
- Even without further comments, it is assumed that a person skilled in the art will be able to utilise the above description in the broadest scope. The preferred embodiments and examples should therefore merely be regarded as descriptive disclosure which is absolutely not limiting in any way.
- The complete disclosure content of all applications, patents and publications mentioned above and below, in particular the corresponding applicat ion EP 05 016 846.7, filed on Mar. 8, 2005, is incorporated into this application by way of reference.
- 1050 ml of deionised water (DI water) are charged into a double-walled reactor, and 0.32 g of tributyl phosphate, 4.2 g of sodium sulfate, 0.01 g of sodium laurylsulfonate and 2.1 g of polyvinyl alcohol as stabiliser are dissolved therein. The organic phase, consisting of 96.25 g of butanediol monovinyl ether, 78.45 g of divinylethyleneurea, 175 g of dodecanol and 4.2 g of α,α′-azobisisobutyronitrile, is added to the aqueous phase with stirring at room temperature after mixing and dissolution of the constituents. The suspension is stirred vigorously and subsequently warmed to 70° C. while flushing with nitrogen. After about 2 hours, the polymerisation is continued at 80° C. for 4 hours. After cooling to room temperature, the polymer is filtered off with suction, the porogen is removed by steam distillation, and the product is stored in 20% ethanolic solution.
- 130 g of polymer, based on the dry weight, are obtained. The average particle size is 80 μm.
- Specific surface area (BET): 322 m2/g
- The swollen volume of the reaction product is 4.3 ml/g.
- The determination of the retention behaviour of proteins as a function of the salt concentration is employed to investigate the protein-binding behaviour of the unmodified support material. Since proteins only bind nonspecifically to the unmodified support material, it is advantageous if as little background binding as possible occurs.
- The binding behaviour of the material according to the invention (prepared corresponding to Example 1) is depicted in comparison with the unmodified copolymer comprising 1,2,-dihydroxy-3-allylaxypropane and methylenebisacrylamide, commercially available under the name FractoPrep® (Merck KGaA, Germany).
FIG. 1 shows the binding behaviour of the proteins lysozyme and bovine serum albumin and of NaNO3. The abbreviation FP stands for FractoPrep®, B/D stands for the copolymer according to the invention comprising butanediol monovinyl ether and divinylethyleneurea. The recovery of the measured proteins, quoted as the quotient of the photometrically determined amount of protein eluted from the column under the run conditions to the protein applied to the column, is greater than 90% under all experimental conditions. - An illustrative pore structure of the polymer according to the invention is shown by way of example in
FIG. 2 through the experimentally determined distribution coefficient Kd of dextrans of various molecular weights, expressed by the viscosity radius of the dextrans. - The material according to the invention prepared corresponding to Example 1 is stored at room temperature in 0.5 M NaOH (volume ratio NaOH:polymer gel=4:1).
- After certain times, the separation behaviour of the polymer is tested with reference to the retention behaviour in the separation of various pullulanes (PSS, Mainz, Germany) as marker compounds.
- Column: 300×10 mm, flow rate: 0.8 ml/min, refractive index detector.
-
FIG. 3 shows the results. It is clearly evident that storage in dilute sodium hydroxide solution has absolutely no effect on the distribution coefficient. - The polymer according to the invention, consisting in this example of a copolymer of 65% by weight of butanediol monovinyl ether and 35% by weight of divinylethyleneurea in the form of spherical porous particles having an average particle diameter of 107 μm, is provided with cationic separation effectors by means of Ce(IV)-initiated graft polymerisation of 2-acrylamido-2-methyl-1-propanesulfonic acid corresponding to the procedure described on
page 10 under Preparation Example A2 in WO 03/031062. The density of functional SO3 groups in this polymer is obtained as 1195 μmol, based on 1 g of dry matter. The ion exchanger material prepared (BATCH: BDM SO3 DZ38) is introduced into a Superformance™ glass column, Goetec Labortechnik (depth of thesorbent bed 40 mm,diameter 10 mm) and equilibrated with a buffer consisting of 20 mM NaH2PO4, pH 6.0. 500 μl of a solution of 5 mg/ml of chymotrypsinogen A, 5 mg/ml of lysozyme and 5 mg/ml of cytochrome C in 20 mM NaH2PO4 are applied to the column. After the application, elution is carried out via a linear gradient over the course of 12 column volumes to 100% of a solution of 20 mM NaH2PO4, 1 M NaCl, pH 6.0. The elution behaviour of the polymer according to the invention results in separation of the individual proteins of the mixture.FIG. 4 shows the associated chromatogram. - The polymer according to the invention in its native form, consisting of 65% by weight of butanediol monovinyl ether and 35% by weight of divinylethyleneurea (BATCH: BDM PS02/E), is introduced into a Superformance™ glass column, Goetec Labortechnik, having a diameter of 50 mm. The depth of the sorbent bed is 20 cm. The average particle diameter of the spherical porous polymer is 107 μm. The pressure drop over this column is measured as a function of the flow rate of a solution of 100 mM NaCl, 20 mM NaH2PO4, pH 6.0. Even at a very high flow rate of 1400 cm/h, the pressure drop is less than 3 bar.
FIG. 5 shows the associated graphical representation of the pressure/flow measurement.
Claims (14)
1-20. (canceled)
21. A polymer based on a copolymer, which is in the form of polymeric particles, comprising
a) at least one hydrophilically substituted alkyl vinyl ether of formula I
where
R1, R2, R3, independently of one another, is H or C1 to C6 alkyl, and
R4 is a radical which carries at least one hydroxyl group,
and
b) at least one crosslinking agent of formula II and/or III and/or IV
where
X is a divalent alkyl radical having 2 to 5 C atoms in which one or more methylene groups which are not adjacent and are not located in the direct vicinity of N may be replaced by O, C═O, S, S═O, SO2, NH, NOH or N and one or more H atoms of the methylene groups may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH—(C1-C8)-alkyl, N—(C1-C8)-alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH,
where
Y1 and Y2 in formula III and IV
are, independently of one another, C1 to C10 alkyl or cycloalkyl, where one or more non-adjacent methylene groups or methylene groups which are not located in the direct vicinity of N may be replaced by O, C═O, S, S═O, SO2, NH, NOH or N and one or more H atoms of the methylene groups may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, or
C6 to C18 aryl, where one or more H atoms in the aryl system may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, and
A is a divalent alkyl radical having 2 to 5 C atoms in which one or more non-adjacent methylene groups or methylene groups which are not located in the direct vicinity of N may be replaced by O, C═O, S, S═O, SO2, NH, NOH or N and one or more H of the methylene groups may be substituted, independently of one another, by hydroxyl groups, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH,
wherein a) and b) have been copolymerized and the resultant polymer is in the form of polymeric particles.
22. A polymer according to claim 21 , wherein R4 in formula I is a straight-chain or branched C1 to C10 alkyl radical, in which one or more non-adjacent methylene groups may be replaced by O, C═O, S, S═O, SO2, NH, NOH, or N and/or in which one or more H atoms may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH and in which at least one OH group is present either on the C1 to C10 alkyl radical or on a substituent, or
a cycloaliphatic radical, in which one or more non-adjacent methylene groups may be replaced by O, C═O, S, S═O, SO2, NH, NOH, or N and/or in which one or more H atoms of the cycloaliphatic radical may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the cycloaliphatic ring or on a side chain or substituent, or
a C6 to C18 aryl radical, where one or more H atoms in the aryl radical may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the aryl radical or on a side chain or substituent, or
a C5 to C18 heteroaryl radical, where one or more H atoms in the heteroaryl radical may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the heteroaryl radical or on a side chain or substituent.
23. A polymer according to claim 21 , wherein the hydrophilically substituted alkyl vinyl ether is a compound of formula I in which R4 is a radical which carries a hydroxyl group.
24. A polymer according to claim 21 , wherein the hydrophilically substituted alkyl vinyl ether is 1,2-ethanediol monovinyl ether, 1,3-propanediol monovinyl ether, 1,4-butanediol monovinyl ether, 1,5-pentanediol monovinyl ether, 1,6-hexanediol monovinyl ether, diethylene glycol monovinyl ether or cyclohexanedimethanol monovinyl ether.
25. A polymer according to claim 21 , wherein the crosslinking agent is divinylethyleneurea (1,3-divinylimidazolin-2-one) or divinylpropyleneurea (1,3-divinyltetrahydropyrimidin-2-one).
26. A polymer according to claim 21 , wherein the hydrophilically substituted alkyl vinyl ether is 1,2-ethanediol monovinyl ether, 1,3-propanediol monovinyl ether, 1,4-butanediol monovinyl ether, 1,5-pentanediol monovinyl ether, 1,6-hexanediol monovinyl ether, diethylene glycol monovinyl ether or cyclohexanedimethanol monovinyl ether, and the crosslinking agent is divinylethyleneurea (1,3-divinylimidazolin-2-one) or divinylpropyleneurea (1,3-divinyltetrahydropyrimidin-2-one).
27. A polymer according to claim 21 , wherein R4 in formula I is a straight-chain or branched C1 to C10 alkyl radical, in which one or more non-adjacent methylene groups may be replaced by O, C═O, S, S═O, SO2, NH, NOH, or N and/or in which one or more H atoms may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH and in which at least one OH group is present either on the C1 to C10 alkyl radical or on a substituent, or
a cycloaliphatic radical having 5 to 10 C atoms, in which one or more non-adjacent methylene groups may be replaced by O, C═O, S, S═O, SO2, NH, NOH, or N and/or in which one or more H atoms of the cycloaliphatic radical may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the cycloaliphatic ring or on a side chain or substituent, or
a C6 to C18 aryl radical, where one or more H atoms in the aryl radical may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the aryl radical or on a side chain or substituent, or
a C5 to C18 heteroaryl radical, where one or more H atoms in the heteroaryl radical may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the heteroaryl radical or on a side chain or substituent.
28. A polymer according to claim 21 , wherein b) is a crosslinking agent of formula III.
29. A process for preparing a polymer, which is in the form of polymeric particles, comprising copolymerizing at least one hydrophilically substituted alkyl vinyl ether of the formula I and at least one crosslinking agent of formula II and/or III and/or IV to form said polymeric particles
where
R1, R2, R3, independently of one another, are H or C1 to C6 alkyl, and
R4 is a radical which carries at least one hydroxyl group,
and
where
X is a divalent alkyl radical having 2 to 5 C atoms in which one or more methylene groups which are not adjacent and are not located in the direct vicinity of N may be replaced by O, C═O, S, S═O, SO2, NH, NOH or N and one or more H atoms of the methylene groups may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH—(C1-C8)-alkyl, N—(C1-C8)-alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH,
where
Y1 and Y2 in formula III and IV
are, independently of one another, C1 to C10 alkyl or cycloalkyl, where one or more non-adjacent methylene groups or methylene groups which are not located in the direct vicinity of N may be replaced by O, C═O, S, S═O, SO2, NH, NOH or N and one or more H of the methylene groups may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, or
C6 to C18 aryl, where one or more H in the aryl system may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, and
A is a divalent alkyl radical having 2 to 5 C atoms in which one or more non-adjacent methylene groups or methylene groups which are not located in the direct vicinity of N may be replaced by O, C═O, S, S═O, SO2, NH, NOH or N and one or more H of the methylene groups may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, halogen, NH2, C5-C10-aryl, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH.
30. A process according to claim 29 , wherein in the compound of formula I, R4 is a straight-chain or branched C1 to C10 alkyl radical, in which one or more non-adjacent methylene groups may be replaced by O, C═O, S, S═O, SO2, NH, NOH, or N and/or in which one or more H atoms may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH and in which at least one OH group is present either on the C1 to C10 alkyl radical or on a substituent, or a cycloaliphatic radical, in which one or more non-adjacent methylene groups may be replaced by O, C═O, S, S═O, SO2, NH, NOH, or N and/or in which one or more H atoms of the cycloaliphatic radical may be substituted, independently of one another, by C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the cycloaliphatic ring or on a side chain or substituent, or
a C6 to C18 aryl radical, where one or more H atoms in the aryl radical may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the aryl radical or on a side chain or substituent, or
a C5 to C18 heteroaryl radical, where one or more H atoms in the heteroaryl radical may be substituted, independently of one another, by hydroxyl, C1-C6-alkyl, C5-C10-aryl, halogen, NH2, NH(C1-C8)alkyl, N(C1-C8)alkyl2, C1-C6-alkoxy or C1-C6-alkyl-OH, where at least one OH group is present either on the heteroaryl radical or on a side chain or substituent.
31. A process according to claim 29 , wherein the temperature during the polymerisation is between 40 and 100° C.
32. A process according to claim 29 , wherein hydrophilically substituted alkyl vinyl ethers and crosslinking agents are in a weight ratio between 10:90 and 80:20.
33. In a method for solid-phase synthesis of a biopolymer, such as a nucleic acid or peptide, wherein the improvement is to perform said solid-phase synthesis on a polymer according to claim 1.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US14/173,218 US20140155565A1 (en) | 2005-08-03 | 2014-02-05 | Hydrophilic crosslinked polymer |
Applications Claiming Priority (6)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP05016846.7 | 2005-08-03 | ||
| EP05016846A EP1754534A1 (en) | 2005-08-03 | 2005-08-03 | Crosslinked hydrophile polymer |
| EPPCT/EP2006/005296 | 2006-06-02 | ||
| US99761308A | 2008-02-01 | 2008-02-01 | |
| US12/947,900 US8765897B2 (en) | 2005-08-03 | 2010-11-17 | Hydrophilic crosslinked polymer |
| US14/173,218 US20140155565A1 (en) | 2005-08-03 | 2014-02-05 | Hydrophilic crosslinked polymer |
Related Parent Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US12/947,900 Continuation US8765897B2 (en) | 2005-08-03 | 2010-11-17 | Hydrophilic crosslinked polymer |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20140155565A1 true US20140155565A1 (en) | 2014-06-05 |
Family
ID=37110325
Family Applications (3)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/997,613 Active 2028-02-22 US7951885B2 (en) | 2005-08-03 | 2006-06-08 | Hydrophilic crosslinked polymer |
| US12/947,900 Active 2028-01-20 US8765897B2 (en) | 2005-08-03 | 2010-11-17 | Hydrophilic crosslinked polymer |
| US14/173,218 Abandoned US20140155565A1 (en) | 2005-08-03 | 2014-02-05 | Hydrophilic crosslinked polymer |
Family Applications Before (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/997,613 Active 2028-02-22 US7951885B2 (en) | 2005-08-03 | 2006-06-08 | Hydrophilic crosslinked polymer |
| US12/947,900 Active 2028-01-20 US8765897B2 (en) | 2005-08-03 | 2010-11-17 | Hydrophilic crosslinked polymer |
Country Status (8)
| Country | Link |
|---|---|
| US (3) | US7951885B2 (en) |
| EP (1) | EP1910433B1 (en) |
| JP (1) | JP5475284B2 (en) |
| KR (1) | KR101289911B1 (en) |
| CN (2) | CN101258175B (en) |
| CA (1) | CA2617645C (en) |
| ES (1) | ES2528426T3 (en) |
| WO (1) | WO2007014591A1 (en) |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9334353B2 (en) | 2012-05-14 | 2016-05-10 | Jsr Corporation | Method for producing polymer particles, polymer particles, filler for chromatography column, and chromatography column |
| US9777085B2 (en) | 2012-02-14 | 2017-10-03 | Maruzen Petrochemical Co., Ltd. | Method for producing homopolymer or random copolymer of hydroxyl group-containing vinyl ether |
| US9975920B2 (en) | 2014-08-15 | 2018-05-22 | Merck Patent Gmbh | Purification of immunoglobulins from plasma |
| US11623941B2 (en) | 2016-09-30 | 2023-04-11 | Cytiva Bioprocess R&D Ab | Separation method |
| US11667671B2 (en) | 2016-05-11 | 2023-06-06 | Cytiva Bioprocess R&D Ab | Separation method |
| US11685764B2 (en) | 2016-05-11 | 2023-06-27 | Cytiva Bioprocess R&D Ab | Separation matrix |
| US11708390B2 (en) * | 2016-05-11 | 2023-07-25 | Cytiva Bioprocess R&D Ab | Method of storing a separation matrix |
| US12037359B2 (en) | 2016-05-11 | 2024-07-16 | Cytiva Bioprocess R&D Ab | Mutated immunoglobulin-binding polypeptides |
| US12134633B2 (en) | 2016-05-11 | 2024-11-05 | Cytiva Bioprocess R&D Ab | Separation matrix |
| US12221492B2 (en) | 2014-11-17 | 2025-02-11 | Cytiva Bioprocess R&D Ab | Mutated immunoglobulin-binding polypeptides |
| US12448411B2 (en) | 2016-09-30 | 2025-10-21 | Cytiva Bioprocess R&D Ab | Separation method |
Families Citing this family (25)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7951885B2 (en) * | 2005-08-03 | 2011-05-31 | Merck Patent Gmbh | Hydrophilic crosslinked polymer |
| AU2007272494B2 (en) | 2006-07-14 | 2011-07-28 | Wisconsin Alumni Research Foundation | Adsorptive membranes for trapping viruses |
| US20090294362A1 (en) * | 2008-05-30 | 2009-12-03 | Persson Jonas Par | Stationary phase for hydrophilic interaction chromatography |
| JP5631869B2 (en) * | 2008-05-30 | 2014-11-26 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツングMerck Patent Gesellschaft mit beschraenkter Haftung | Graft polymerization initiated by Ce (IV) on a polymer containing no hydroxyl group |
| US8592555B2 (en) | 2008-08-11 | 2013-11-26 | Emd Millipore Corporation | Immunoglobulin-binding proteins with improved specificity |
| SG162687A1 (en) * | 2008-12-24 | 2010-07-29 | Millipore Corp | Caustic stable chromatography ligands |
| SG10201604554WA (en) | 2011-06-08 | 2016-07-28 | Emd Millipore Corp | Chromatography matrices including novel staphylococcus aureus protein a based ligands |
| US10189891B2 (en) * | 2012-03-28 | 2019-01-29 | Ge Healthcare Bioprocess R&D Ab | Affinity chromatography matrix |
| EP2656892A1 (en) | 2012-04-23 | 2013-10-30 | Merck Patent GmbH | Chromatography method |
| JP6220496B2 (en) * | 2012-05-14 | 2017-10-25 | Jsr株式会社 | Method for producing polymer particle, polymer particle, packing material for chromatography column, and chromatography column |
| MY195756A (en) | 2012-09-17 | 2023-02-09 | Grace W R & Co | Functionalized Particulate Support Material and Methods of Making and Using the Same |
| WO2014058570A1 (en) | 2012-09-17 | 2014-04-17 | Alltech Associates, Inc. | Chromatography media and devices |
| KR101880565B1 (en) | 2013-09-04 | 2018-07-23 | 이엠디 밀리포어 코포레이션 | Methods of cleaning a protein a based affinity chromatography coloumn |
| WO2015088677A1 (en) | 2013-12-12 | 2015-06-18 | Emd Millipore Corporation | Protein separations using an acrylamide containing filter |
| JP6853670B2 (en) | 2014-01-16 | 2021-04-07 | ダブリュー・アール・グレース・アンド・カンパニー−コーンW R Grace & Co−Conn | Affinity Chromatography Medium and Chromatography Devices |
| US11389783B2 (en) | 2014-05-02 | 2022-07-19 | W.R. Grace & Co.-Conn. | Functionalized support material and methods of making and using functionalized support material |
| JP2018517559A (en) | 2015-06-05 | 2018-07-05 | ダブリュー・アール・グレース・アンド・カンパニー−コーンW R Grace & Co−Conn | Adsorbing bioprocess clarifier and method for producing and using the same |
| AU2018217433B2 (en) * | 2017-02-09 | 2023-03-09 | Ixom Operations Pty Ltd | Polymer beads and application thereof |
| TWI799441B (en) | 2017-08-23 | 2023-04-21 | 日商Jsr股份有限公司 | Chromatography carrier, ligand-immobilized carrier, chromatography column, purification method of target substance, and production method of chromatography carrier |
| CN117920163A (en) | 2018-11-06 | 2024-04-26 | Jsr株式会社 | Method for producing organic sulfur compound |
| JP7590400B2 (en) * | 2019-07-03 | 2024-11-26 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | Purification of antibody drug conjugates |
| JP7700092B2 (en) | 2019-07-03 | 2025-06-30 | メルク パテント ゲゼルシャフト ミット ベシュレンクテル ハフツング | Glycoform purification |
| WO2025027058A1 (en) | 2023-08-01 | 2025-02-06 | Merck Patent Gmbh | Polishing for aav particles using anion exchange chromatography with improved elution system |
| WO2025027059A1 (en) | 2023-08-02 | 2025-02-06 | Merck Patent Gmbh | Method for purification of plasmid dna |
| WO2025257280A1 (en) | 2024-06-14 | 2025-12-18 | Merck Patent Gmbh | Method for purification of rna |
Citations (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7951885B2 (en) * | 2005-08-03 | 2011-05-31 | Merck Patent Gmbh | Hydrophilic crosslinked polymer |
Family Cites Families (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JPS54160300A (en) | 1978-06-08 | 1979-12-18 | Toyo Soda Mfg Co Ltd | Hydrophilic separating carrier and making method thereof |
| DE3344912A1 (en) | 1983-12-13 | 1985-06-20 | Hoechst Ag, 6230 Frankfurt | CROSSLINKED POLYMERISATES, METHOD FOR THE PRODUCTION THEREOF AND THEIR USE |
| JPH0648267B2 (en) * | 1984-07-26 | 1994-06-22 | 日本エクスラン工業株式会社 | Method for producing gel for liquid chromatography |
| DE3543348A1 (en) * | 1985-12-07 | 1987-06-11 | Bayer Ag | PEARL-SHAPED CROSS-NETWORKED MIXED POLYMERS WITH EPOXY AND BASIC AMINO GROUPS, METHOD FOR THE PRODUCTION THEREOF AND THEIR USE |
| DE3629176A1 (en) * | 1986-08-28 | 1988-03-17 | Hoechst Ag | CROSSLINKED POLYMERISATES WITH CARBONATE TER GROUPS AND METHOD FOR THE PRODUCTION THEREOF |
| DE3629177A1 (en) * | 1986-08-28 | 1988-03-17 | Hoechst Ag | CROSSLINKED POLYMERISATES AND METHOD FOR THE PRODUCTION THEREOF |
| DE3714276C2 (en) | 1987-04-29 | 2002-09-19 | Celanese Ventures Gmbh | Hydrophilic, crosslinked polymers, processes for their preparation and their use |
| DE3811042A1 (en) | 1988-03-31 | 1989-10-19 | Merck Patent Gmbh | ION EXCHANGER |
| DE9013137U1 (en) | 1990-09-15 | 1992-01-23 | Röhm GmbH, 6100 Darmstadt | Highly cross-linked, pearl-shaped, activatable, hydrophilic carrier material |
| TW198064B (en) * | 1990-12-24 | 1993-01-11 | Hoechst Ag | |
| DE4130428A1 (en) * | 1991-09-13 | 1993-03-18 | Basf Ag | COPOLYMERISATES OF HYDROXYALKYLVINYL ETHERS AND THE USE OF HOMO- AND COPOLYMERISES OF HYDROXYALKYLVINYL ETHERS IN WASHING AND CLEANING AGENTS |
| JPH0769686A (en) * | 1993-08-31 | 1995-03-14 | Dainippon Ink & Chem Inc | UV curable resin composition for coating optical fiber |
| SE9303790D0 (en) | 1993-11-17 | 1993-11-17 | Pharmacia Lkb Biotech | A method for separation and synthetic polymers that can be used as separation media in the method |
| US5652348A (en) | 1994-09-23 | 1997-07-29 | Massey University | Chromatographic resins and methods for using same |
| SE9600590D0 (en) | 1996-02-19 | 1996-02-19 | Pharmacia Biotech Ab | Methods for chromatographic separation of peptides and nucleic acid and new high-affinity ion exchange matrix |
| DE19920794A1 (en) | 1999-05-06 | 2000-11-09 | Merck Patent Gmbh | Process for the preparation of bead polymers |
| US6310199B1 (en) | 1999-05-14 | 2001-10-30 | Promega Corporation | pH dependent ion exchange matrix and method of use in the isolation of nucleic acids |
| SE9904197D0 (en) | 1999-11-22 | 1999-11-22 | Amersham Pharm Biotech Ab | An method for anion exchange adsorption on matrices carrying mixed mode ligands |
| EP1179732B2 (en) | 2000-08-11 | 2012-02-29 | Rohm And Haas Company | Polymeric adsorbent and method of preparation |
| ATE376007T1 (en) | 2002-06-05 | 2007-11-15 | Imp College Innovations Ltd | POLYVINYL ETHER |
| EP1693108A1 (en) | 2004-12-04 | 2006-08-23 | MERCK PATENT GmbH | Mixed-modal anion-exchange type separation material |
| DE102005051963A1 (en) | 2005-10-29 | 2007-05-03 | Werner & Pfleiderer Industrielle Backtechnik Gmbh | Pressing device for a plant for the production of biscuit sticks |
-
2006
- 2006-06-08 US US11/997,613 patent/US7951885B2/en active Active
- 2006-06-08 CA CA2617645A patent/CA2617645C/en active Active
- 2006-06-08 EP EP06754224.1A patent/EP1910433B1/en active Active
- 2006-06-08 CN CN2006800285993A patent/CN101258175B/en active Active
- 2006-06-08 CN CN201110266314.2A patent/CN102417557B/en active Active
- 2006-06-08 ES ES06754224.1T patent/ES2528426T3/en active Active
- 2006-06-08 WO PCT/EP2006/005484 patent/WO2007014591A1/en not_active Ceased
- 2006-06-08 KR KR1020087005296A patent/KR101289911B1/en active Active
- 2006-06-08 JP JP2008524371A patent/JP5475284B2/en active Active
-
2010
- 2010-11-17 US US12/947,900 patent/US8765897B2/en active Active
-
2014
- 2014-02-05 US US14/173,218 patent/US20140155565A1/en not_active Abandoned
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US7951885B2 (en) * | 2005-08-03 | 2011-05-31 | Merck Patent Gmbh | Hydrophilic crosslinked polymer |
| US8765897B2 (en) * | 2005-08-03 | 2014-07-01 | Merck Patent Gmbh | Hydrophilic crosslinked polymer |
Non-Patent Citations (1)
| Title |
|---|
| Webster's New Universal Unabridged Dictionary, page 568 (1996) * |
Cited By (11)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US9777085B2 (en) | 2012-02-14 | 2017-10-03 | Maruzen Petrochemical Co., Ltd. | Method for producing homopolymer or random copolymer of hydroxyl group-containing vinyl ether |
| US9334353B2 (en) | 2012-05-14 | 2016-05-10 | Jsr Corporation | Method for producing polymer particles, polymer particles, filler for chromatography column, and chromatography column |
| US9975920B2 (en) | 2014-08-15 | 2018-05-22 | Merck Patent Gmbh | Purification of immunoglobulins from plasma |
| US12221492B2 (en) | 2014-11-17 | 2025-02-11 | Cytiva Bioprocess R&D Ab | Mutated immunoglobulin-binding polypeptides |
| US11667671B2 (en) | 2016-05-11 | 2023-06-06 | Cytiva Bioprocess R&D Ab | Separation method |
| US11685764B2 (en) | 2016-05-11 | 2023-06-27 | Cytiva Bioprocess R&D Ab | Separation matrix |
| US11708390B2 (en) * | 2016-05-11 | 2023-07-25 | Cytiva Bioprocess R&D Ab | Method of storing a separation matrix |
| US12037359B2 (en) | 2016-05-11 | 2024-07-16 | Cytiva Bioprocess R&D Ab | Mutated immunoglobulin-binding polypeptides |
| US12134633B2 (en) | 2016-05-11 | 2024-11-05 | Cytiva Bioprocess R&D Ab | Separation matrix |
| US11623941B2 (en) | 2016-09-30 | 2023-04-11 | Cytiva Bioprocess R&D Ab | Separation method |
| US12448411B2 (en) | 2016-09-30 | 2025-10-21 | Cytiva Bioprocess R&D Ab | Separation method |
Also Published As
| Publication number | Publication date |
|---|---|
| ES2528426T3 (en) | 2015-02-09 |
| CN102417557B (en) | 2015-03-11 |
| EP1910433A1 (en) | 2008-04-16 |
| CA2617645C (en) | 2014-09-09 |
| CN102417557A (en) | 2012-04-18 |
| CA2617645A1 (en) | 2007-02-08 |
| JP2009503203A (en) | 2009-01-29 |
| CN101258175B (en) | 2011-11-16 |
| KR101289911B1 (en) | 2013-07-26 |
| US8765897B2 (en) | 2014-07-01 |
| US7951885B2 (en) | 2011-05-31 |
| US20110091727A1 (en) | 2011-04-21 |
| KR20080039472A (en) | 2008-05-07 |
| JP5475284B2 (en) | 2014-04-16 |
| CN101258175A (en) | 2008-09-03 |
| US20080210615A1 (en) | 2008-09-04 |
| EP1910433B1 (en) | 2014-11-05 |
| WO2007014591A1 (en) | 2007-02-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US8765897B2 (en) | Hydrophilic crosslinked polymer | |
| CA2737404C (en) | Temperature-responsive polymer particles in protein separation | |
| US4208309A (en) | Pearl polymer containing hollow pearls | |
| JPH08502534A (en) | Pore size selective modification method for porous materials | |
| EP2414321B1 (en) | Hydrophobic monomers, hydrophobically-derivatized supports, and methods of making and using the same | |
| US4190713A (en) | Pearl polymer containing hollow pearls | |
| CN1946479A (en) | A method of preparing a separation matrix | |
| US20100047904A1 (en) | Materials, methods and systems for purification and/or separation | |
| WO1993006161A1 (en) | Macroporous polymeric particles as biocompatible supports | |
| US9303098B2 (en) | Ce(IV)-initiated graft polymerisation on polymers containing no hydroxyl groups | |
| WO2019110318A1 (en) | Porous materials, method for producing same and uses thereof | |
| EP1754534A1 (en) | Crosslinked hydrophile polymer | |
| CN101791543B (en) | Preparation method of chromatographic particle medium | |
| CN101791542B (en) | Preparation method of chromatographic particle medium | |
| CZ291396B6 (en) | Process for preparing spherical polymeric sorbents |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: MERCK PATENT GMBH, GERMANY Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:JOEHNCK, MATTHIAS;SABROWSKI, ECKHARD;SIGNING DATES FROM 20140217 TO 20140303;REEL/FRAME:032516/0074 |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |