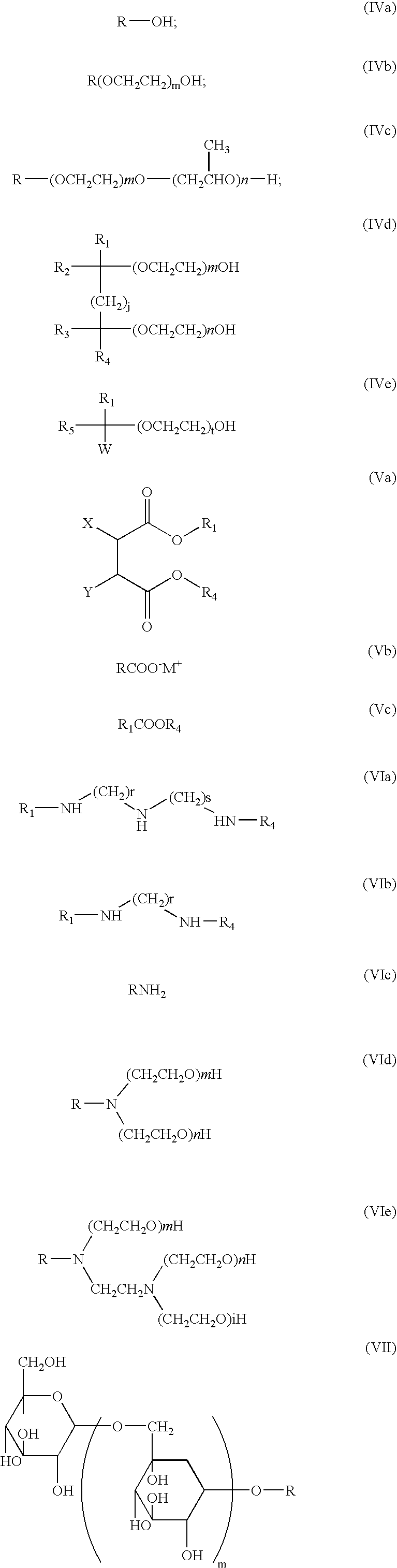US20050161644A1 - Immersion lithography fluids - Google Patents
Immersion lithography fluids Download PDFInfo
- Publication number
- US20050161644A1 US20050161644A1 US10/764,227 US76422704A US2005161644A1 US 20050161644 A1 US20050161644 A1 US 20050161644A1 US 76422704 A US76422704 A US 76422704A US 2005161644 A1 US2005161644 A1 US 2005161644A1
- Authority
- US
- United States
- Prior art keywords
- alkyl
- immersion fluid
- additive
- alcohol
- fluid
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Abandoned
Links
- 239000012530 fluid Substances 0.000 title claims abstract description 203
- 238000000671 immersion lithography Methods 0.000 title abstract description 12
- 238000007654 immersion Methods 0.000 claims abstract description 139
- 239000000654 additive Substances 0.000 claims abstract description 105
- 230000000996 additive effect Effects 0.000 claims abstract description 95
- 239000000203 mixture Substances 0.000 claims abstract description 18
- 125000000217 alkyl group Chemical group 0.000 claims description 46
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 44
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 43
- -1 alkyl diamine Chemical class 0.000 claims description 43
- 239000002609 medium Substances 0.000 claims description 42
- 150000003973 alkyl amines Chemical class 0.000 claims description 26
- GOOHAUXETOMSMM-UHFFFAOYSA-N Propylene oxide Chemical class CC1CO1 GOOHAUXETOMSMM-UHFFFAOYSA-N 0.000 claims description 25
- 150000002009 diols Chemical class 0.000 claims description 24
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 claims description 21
- 125000005233 alkylalcohol group Chemical group 0.000 claims description 20
- 238000000034 method Methods 0.000 claims description 19
- 238000002835 absorbance Methods 0.000 claims description 18
- XSQUKJJJFZCRTK-UHFFFAOYSA-N Urea Chemical compound NC(N)=O XSQUKJJJFZCRTK-UHFFFAOYSA-N 0.000 claims description 16
- 239000004202 carbamide Substances 0.000 claims description 16
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 claims description 15
- 150000003839 salts Chemical class 0.000 claims description 15
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 claims description 14
- 239000003792 electrolyte Substances 0.000 claims description 14
- IAYPIBMASNFSPL-UHFFFAOYSA-N Ethylene oxide Chemical compound C1CO1 IAYPIBMASNFSPL-UHFFFAOYSA-N 0.000 claims description 13
- 229920001296 polysiloxane Polymers 0.000 claims description 13
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 claims description 12
- 150000002148 esters Chemical class 0.000 claims description 11
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 claims description 11
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 10
- 229920000642 polymer Polymers 0.000 claims description 10
- IWYRWIUNAVNFPE-UHFFFAOYSA-N Glycidaldehyde Chemical compound O=CC1CO1 IWYRWIUNAVNFPE-UHFFFAOYSA-N 0.000 claims description 9
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 claims description 9
- QAOWNCQODCNURD-UHFFFAOYSA-L Sulfate Chemical compound [O-]S([O-])(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-L 0.000 claims description 9
- 239000002253 acid Substances 0.000 claims description 9
- 125000005599 alkyl carboxylate group Chemical group 0.000 claims description 9
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 claims description 9
- 229920000728 polyester Polymers 0.000 claims description 9
- BDHFUVZGWQCTTF-UHFFFAOYSA-M sulfonate Chemical compound [O-]S(=O)=O BDHFUVZGWQCTTF-UHFFFAOYSA-M 0.000 claims description 9
- VGGSQFUCUMXWEO-UHFFFAOYSA-N Ethene Chemical compound C=C VGGSQFUCUMXWEO-UHFFFAOYSA-N 0.000 claims description 8
- 239000005977 Ethylene Substances 0.000 claims description 8
- 125000005210 alkyl ammonium group Chemical group 0.000 claims description 8
- 150000008051 alkyl sulfates Chemical class 0.000 claims description 8
- 150000008052 alkyl sulfonates Chemical class 0.000 claims description 8
- 125000003277 amino group Chemical group 0.000 claims description 8
- 229940083124 ganglion-blocking antiadrenergic secondary and tertiary amines Drugs 0.000 claims description 8
- 150000003141 primary amines Chemical class 0.000 claims description 8
- 230000005540 biological transmission Effects 0.000 claims description 7
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 claims description 7
- WYURNTSHIVDZCO-UHFFFAOYSA-N tetrahydrofuran Substances C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims description 7
- 239000002202 Polyethylene glycol Substances 0.000 claims description 4
- 229920001223 polyethylene glycol Polymers 0.000 claims description 4
- NDVWOBYBJYUSMF-NKWVEPMBSA-N (1r,2s)-2-methylcyclohexan-1-ol Chemical compound C[C@H]1CCCC[C@H]1O NDVWOBYBJYUSMF-NKWVEPMBSA-N 0.000 claims description 3
- WVIIMZNLDWSIRH-UHFFFAOYSA-N cyclohexylcyclohexane Chemical group C1CCCCC1C1CCCCC1 WVIIMZNLDWSIRH-UHFFFAOYSA-N 0.000 claims description 3
- 239000012736 aqueous medium Substances 0.000 claims description 2
- 239000000758 substrate Substances 0.000 description 32
- 229920002120 photoresistant polymer Polymers 0.000 description 23
- 229910021642 ultra pure water Inorganic materials 0.000 description 13
- 239000012498 ultrapure water Substances 0.000 description 12
- 238000000576 coating method Methods 0.000 description 10
- 239000000243 solution Substances 0.000 description 10
- 239000004094 surface-active agent Substances 0.000 description 10
- 125000004432 carbon atom Chemical group C* 0.000 description 9
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 8
- 239000011248 coating agent Substances 0.000 description 8
- 230000008569 process Effects 0.000 description 8
- 238000009736 wetting Methods 0.000 description 8
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 7
- 230000005855 radiation Effects 0.000 description 7
- BTRWELPXUDWAGW-UHFFFAOYSA-N 2,4,7,9-tetramethyldecane-4,7-diol Chemical group CC(C)CC(C)(O)CCC(C)(O)CC(C)C BTRWELPXUDWAGW-UHFFFAOYSA-N 0.000 description 6
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 6
- 239000013078 crystal Substances 0.000 description 6
- 125000006165 cyclic alkyl group Chemical group 0.000 description 6
- 239000006260 foam Substances 0.000 description 6
- 239000007788 liquid Substances 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 125000003709 fluoroalkyl group Chemical group 0.000 description 5
- 125000004435 hydrogen atom Chemical group [H]* 0.000 description 5
- 125000005010 perfluoroalkyl group Chemical group 0.000 description 5
- 230000008859 change Effects 0.000 description 4
- 239000008240 homogeneous mixture Substances 0.000 description 4
- 229910052710 silicon Inorganic materials 0.000 description 4
- 239000010703 silicon Substances 0.000 description 4
- 235000012239 silicon dioxide Nutrition 0.000 description 4
- WGTYBPLFGIVFAS-UHFFFAOYSA-M tetramethylammonium hydroxide Chemical compound [OH-].C[N+](C)(C)C WGTYBPLFGIVFAS-UHFFFAOYSA-M 0.000 description 4
- ZWEHNKRNPOVVGH-UHFFFAOYSA-N 2-Butanone Chemical compound CCC(C)=O ZWEHNKRNPOVVGH-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- ZMXDDKWLCZADIW-UHFFFAOYSA-N N,N-Dimethylformamide Chemical compound CN(C)C=O ZMXDDKWLCZADIW-UHFFFAOYSA-N 0.000 description 3
- KWYUFKZDYYNOTN-UHFFFAOYSA-M Potassium hydroxide Chemical compound [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 description 3
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 3
- 230000015572 biosynthetic process Effects 0.000 description 3
- 239000012141 concentrate Substances 0.000 description 3
- 238000001459 lithography Methods 0.000 description 3
- 239000000463 material Substances 0.000 description 3
- 238000005259 measurement Methods 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- 230000003287 optical effect Effects 0.000 description 3
- 239000000377 silicon dioxide Substances 0.000 description 3
- RWNUSVWFHDHRCJ-UHFFFAOYSA-N 1-butoxypropan-2-ol Chemical compound CCCCOCC(C)O RWNUSVWFHDHRCJ-UHFFFAOYSA-N 0.000 description 2
- ARXJGSRGQADJSQ-UHFFFAOYSA-N 1-methoxypropan-2-ol Chemical compound COCC(C)O ARXJGSRGQADJSQ-UHFFFAOYSA-N 0.000 description 2
- FENFUOGYJVOCRY-UHFFFAOYSA-N 1-propoxypropan-2-ol Chemical compound CCCOCC(C)O FENFUOGYJVOCRY-UHFFFAOYSA-N 0.000 description 2
- CETWDUZRCINIHU-UHFFFAOYSA-N 2-heptanol Chemical compound CCCCCC(C)O CETWDUZRCINIHU-UHFFFAOYSA-N 0.000 description 2
- XLLIQLLCWZCATF-UHFFFAOYSA-N 2-methoxyethyl acetate Chemical compound COCCOC(C)=O XLLIQLLCWZCATF-UHFFFAOYSA-N 0.000 description 2
- PFNHSEQQEPMLNI-UHFFFAOYSA-N 2-methyl-1-pentanol Chemical compound CCCC(C)CO PFNHSEQQEPMLNI-UHFFFAOYSA-N 0.000 description 2
- NECRQCBKTGZNMH-UHFFFAOYSA-N 3,5-dimethylhex-1-yn-3-ol Chemical compound CC(C)CC(C)(O)C#C NECRQCBKTGZNMH-UHFFFAOYSA-N 0.000 description 2
- JBRZTFJDHDCESZ-UHFFFAOYSA-N AsGa Chemical compound [As]#[Ga] JBRZTFJDHDCESZ-UHFFFAOYSA-N 0.000 description 2
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 2
- 229910001218 Gallium arsenide Inorganic materials 0.000 description 2
- RZKSECIXORKHQS-UHFFFAOYSA-N Heptan-3-ol Chemical compound CCCCC(O)CC RZKSECIXORKHQS-UHFFFAOYSA-N 0.000 description 2
- AMQJEAYHLZJPGS-UHFFFAOYSA-N N-Pentanol Chemical compound CCCCCO AMQJEAYHLZJPGS-UHFFFAOYSA-N 0.000 description 2
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- 238000010521 absorption reaction Methods 0.000 description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 2
- 150000003863 ammonium salts Chemical class 0.000 description 2
- 238000005452 bending Methods 0.000 description 2
- WERYXYBDKMZEQL-UHFFFAOYSA-N butane-1,4-diol Chemical compound OCCCCO WERYXYBDKMZEQL-UHFFFAOYSA-N 0.000 description 2
- 230000000052 comparative effect Effects 0.000 description 2
- 238000007796 conventional method Methods 0.000 description 2
- 229920001577 copolymer Polymers 0.000 description 2
- JHIVVAPYMSGYDF-UHFFFAOYSA-N cyclohexanone Chemical compound O=C1CCCCC1 JHIVVAPYMSGYDF-UHFFFAOYSA-N 0.000 description 2
- 239000008367 deionised water Substances 0.000 description 2
- 229910021641 deionized water Inorganic materials 0.000 description 2
- 238000011161 development Methods 0.000 description 2
- JXTHNDFMNIQAHM-UHFFFAOYSA-N dichloroacetic acid Chemical compound OC(=O)C(Cl)Cl JXTHNDFMNIQAHM-UHFFFAOYSA-N 0.000 description 2
- SBZXBUIDTXKZTM-UHFFFAOYSA-N diglyme Chemical compound COCCOCCOC SBZXBUIDTXKZTM-UHFFFAOYSA-N 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- KZTYYGOKRVBIMI-UHFFFAOYSA-N diphenyl sulfone Chemical compound C=1C=CC=CC=1S(=O)(=O)C1=CC=CC=C1 KZTYYGOKRVBIMI-UHFFFAOYSA-N 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- LQZZUXJYWNFBMV-UHFFFAOYSA-N dodecan-1-ol Chemical compound CCCCCCCCCCCCO LQZZUXJYWNFBMV-UHFFFAOYSA-N 0.000 description 2
- SNRUBQQJIBEYMU-UHFFFAOYSA-N dodecane Chemical compound CCCCCCCCCCCC SNRUBQQJIBEYMU-UHFFFAOYSA-N 0.000 description 2
- 239000011521 glass Substances 0.000 description 2
- QNVRIHYSUZMSGM-UHFFFAOYSA-N hexan-2-ol Chemical compound CCCCC(C)O QNVRIHYSUZMSGM-UHFFFAOYSA-N 0.000 description 2
- ZOCHHNOQQHDWHG-UHFFFAOYSA-N hexan-3-ol Chemical compound CCCC(O)CC ZOCHHNOQQHDWHG-UHFFFAOYSA-N 0.000 description 2
- 239000012456 homogeneous solution Substances 0.000 description 2
- 230000002209 hydrophobic effect Effects 0.000 description 2
- RLSSMJSEOOYNOY-UHFFFAOYSA-N m-cresol Chemical compound CC1=CC=CC(O)=C1 RLSSMJSEOOYNOY-UHFFFAOYSA-N 0.000 description 2
- BJEPYKJPYRNKOW-UHFFFAOYSA-N malic acid Chemical compound OC(=O)C(O)CC(O)=O BJEPYKJPYRNKOW-UHFFFAOYSA-N 0.000 description 2
- 238000002156 mixing Methods 0.000 description 2
- 239000010702 perfluoropolyether Substances 0.000 description 2
- YPFDHNVEDLHUCE-UHFFFAOYSA-N propane-1,3-diol Chemical compound OCCCO YPFDHNVEDLHUCE-UHFFFAOYSA-N 0.000 description 2
- LLHKCFNBLRBOGN-UHFFFAOYSA-N propylene glycol methyl ether acetate Chemical compound COCC(C)OC(C)=O LLHKCFNBLRBOGN-UHFFFAOYSA-N 0.000 description 2
- 239000010453 quartz Substances 0.000 description 2
- 238000003380 quartz crystal microbalance Methods 0.000 description 2
- 230000003068 static effect Effects 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- HHVIBTZHLRERCL-UHFFFAOYSA-N sulfonyldimethane Chemical compound CS(C)(=O)=O HHVIBTZHLRERCL-UHFFFAOYSA-N 0.000 description 2
- 238000012360 testing method Methods 0.000 description 2
- NDVWOBYBJYUSMF-RNFRBKRXSA-N (1r,2r)-2-methylcyclohexan-1-ol Chemical compound C[C@@H]1CCCC[C@H]1O NDVWOBYBJYUSMF-RNFRBKRXSA-N 0.000 description 1
- AJDIZQLSFPQPEY-UHFFFAOYSA-N 1,1,2-Trichlorotrifluoroethane Chemical compound FC(F)(Cl)C(F)(Cl)Cl AJDIZQLSFPQPEY-UHFFFAOYSA-N 0.000 description 1
- WNXJIVFYUVYPPR-UHFFFAOYSA-N 1,3-dioxolane Chemical compound C1COCO1 WNXJIVFYUVYPPR-UHFFFAOYSA-N 0.000 description 1
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 description 1
- VTBOTOBFGSVRMA-UHFFFAOYSA-N 1-Methylcyclohexanol Chemical compound CC1(O)CCCCC1 VTBOTOBFGSVRMA-UHFFFAOYSA-N 0.000 description 1
- AZUYLZMQTIKGSC-UHFFFAOYSA-N 1-[6-[4-(5-chloro-6-methyl-1H-indazol-4-yl)-5-methyl-3-(1-methylindazol-5-yl)pyrazol-1-yl]-2-azaspiro[3.3]heptan-2-yl]prop-2-en-1-one Chemical compound ClC=1C(=C2C=NNC2=CC=1C)C=1C(=NN(C=1C)C1CC2(CN(C2)C(C=C)=O)C1)C=1C=C2C=NN(C2=CC=1)C AZUYLZMQTIKGSC-UHFFFAOYSA-N 0.000 description 1
- DURPTKYDGMDSBL-UHFFFAOYSA-N 1-butoxybutane Chemical compound CCCCOCCCC DURPTKYDGMDSBL-UHFFFAOYSA-N 0.000 description 1
- RFZHJHSNHYIRNE-UHFFFAOYSA-N 2,3-dimethylpentan-3-ol Chemical compound CCC(C)(O)C(C)C RFZHJHSNHYIRNE-UHFFFAOYSA-N 0.000 description 1
- LXOFYPKXCSULTL-UHFFFAOYSA-N 2,4,7,9-tetramethyldec-5-yne-4,7-diol Chemical compound CC(C)CC(C)(O)C#CC(C)(O)CC(C)C LXOFYPKXCSULTL-UHFFFAOYSA-N 0.000 description 1
- RHRRUYIZUBAQTQ-UHFFFAOYSA-N 2,5,8,11-tetramethyldodec-6-yne-5,8-diol Chemical compound CC(C)CCC(C)(O)C#CC(C)(O)CCC(C)C RHRRUYIZUBAQTQ-UHFFFAOYSA-N 0.000 description 1
- YSUQLAYJZDEMOT-UHFFFAOYSA-N 2-(butoxymethyl)oxirane Chemical compound CCCCOCC1CO1 YSUQLAYJZDEMOT-UHFFFAOYSA-N 0.000 description 1
- QNVRIHYSUZMSGM-LURJTMIESA-N 2-Hexanol Natural products CCCC[C@H](C)O QNVRIHYSUZMSGM-LURJTMIESA-N 0.000 description 1
- POAOYUHQDCAZBD-UHFFFAOYSA-N 2-butoxyethanol Chemical compound CCCCOCCO POAOYUHQDCAZBD-UHFFFAOYSA-N 0.000 description 1
- JKUOURUUCRHADD-UHFFFAOYSA-N 2-ethoxyethyl 3-oxobutanoate Chemical compound CCOCCOC(=O)CC(C)=O JKUOURUUCRHADD-UHFFFAOYSA-N 0.000 description 1
- ISTJMQSHILQAEC-UHFFFAOYSA-N 2-methyl-3-pentanol Chemical compound CCC(O)C(C)C ISTJMQSHILQAEC-UHFFFAOYSA-N 0.000 description 1
- FUGYGGDSWSUORM-UHFFFAOYSA-N 4-hydroxystyrene Chemical compound OC1=CC=C(C=C)C=C1 FUGYGGDSWSUORM-UHFFFAOYSA-N 0.000 description 1
- ZDVJGWXFXGJSIU-UHFFFAOYSA-N 5-methylhexan-2-ol Chemical compound CC(C)CCC(C)O ZDVJGWXFXGJSIU-UHFFFAOYSA-N 0.000 description 1
- KJMBBHZOLRRVMV-UHFFFAOYSA-N 7-methyloctan-4-ol Chemical compound CCCC(O)CCC(C)C KJMBBHZOLRRVMV-UHFFFAOYSA-N 0.000 description 1
- NIXOWILDQLNWCW-UHFFFAOYSA-M Acrylate Chemical compound [O-]C(=O)C=C NIXOWILDQLNWCW-UHFFFAOYSA-M 0.000 description 1
- 229910000838 Al alloy Inorganic materials 0.000 description 1
- QGZKDVFQNNGYKY-UHFFFAOYSA-O Ammonium Chemical compound [NH4+] QGZKDVFQNNGYKY-UHFFFAOYSA-O 0.000 description 1
- RYGMFSIKBFXOCR-UHFFFAOYSA-N Copper Chemical compound [Cu] RYGMFSIKBFXOCR-UHFFFAOYSA-N 0.000 description 1
- 229910000881 Cu alloy Inorganic materials 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- RPNUMPOLZDHAAY-UHFFFAOYSA-N Diethylenetriamine Chemical compound NCCNCCN RPNUMPOLZDHAAY-UHFFFAOYSA-N 0.000 description 1
- HXQPUEQDBSPXTE-UHFFFAOYSA-N Diisobutylcarbinol Chemical compound CC(C)CC(O)CC(C)C HXQPUEQDBSPXTE-UHFFFAOYSA-N 0.000 description 1
- LCGLNKUTAGEVQW-UHFFFAOYSA-N Dimethyl ether Chemical compound COC LCGLNKUTAGEVQW-UHFFFAOYSA-N 0.000 description 1
- KMTRUDSVKNLOMY-UHFFFAOYSA-N Ethylene carbonate Chemical compound O=C1OCCO1 KMTRUDSVKNLOMY-UHFFFAOYSA-N 0.000 description 1
- PIICEJLVQHRZGT-UHFFFAOYSA-N Ethylenediamine Chemical compound NCCN PIICEJLVQHRZGT-UHFFFAOYSA-N 0.000 description 1
- FXHOOIRPVKKKFG-UHFFFAOYSA-N N,N-Dimethylacetamide Chemical compound CN(C)C(C)=O FXHOOIRPVKKKFG-UHFFFAOYSA-N 0.000 description 1
- SECXISVLQFMRJM-UHFFFAOYSA-N N-Methylpyrrolidone Chemical compound CN1CCCC1=O SECXISVLQFMRJM-UHFFFAOYSA-N 0.000 description 1
- 229910019142 PO4 Inorganic materials 0.000 description 1
- ALQSHHUCVQOPAS-UHFFFAOYSA-N Pentane-1,5-diol Chemical compound OCCCCCO ALQSHHUCVQOPAS-UHFFFAOYSA-N 0.000 description 1
- ABLZXFCXXLZCGV-UHFFFAOYSA-N Phosphorous acid Chemical class OP(O)=O ABLZXFCXXLZCGV-UHFFFAOYSA-N 0.000 description 1
- 229920001665 Poly-4-vinylphenol Polymers 0.000 description 1
- 239000004642 Polyimide Substances 0.000 description 1
- 229910052581 Si3N4 Inorganic materials 0.000 description 1
- XUIMIQQOPSSXEZ-UHFFFAOYSA-N Silicon Chemical compound [Si] XUIMIQQOPSSXEZ-UHFFFAOYSA-N 0.000 description 1
- 239000001089 [(2R)-oxolan-2-yl]methanol Substances 0.000 description 1
- 238000000862 absorption spectrum Methods 0.000 description 1
- RYRMWCTWXJAFMK-UHFFFAOYSA-N acetylene;1,1,2,2,3,3-hexafluoropropan-1-ol Chemical group C#C.OC(F)(F)C(F)(F)C(F)F RYRMWCTWXJAFMK-UHFFFAOYSA-N 0.000 description 1
- 230000001154 acute effect Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 150000001298 alcohols Chemical class 0.000 description 1
- 125000002947 alkylene group Chemical group 0.000 description 1
- 125000000304 alkynyl group Chemical group 0.000 description 1
- 229910052782 aluminium Inorganic materials 0.000 description 1
- 229910021417 amorphous silicon Inorganic materials 0.000 description 1
- 125000000129 anionic group Chemical group 0.000 description 1
- VOBHVQRBBHSZAZ-VXGBXAGGSA-N bis(3-methylbutyl) (2r,3r)-2,3-dihydroxybutanedioate Chemical compound CC(C)CCOC(=O)[C@H](O)[C@@H](O)C(=O)OCCC(C)C VOBHVQRBBHSZAZ-VXGBXAGGSA-N 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- WUKWITHWXAAZEY-UHFFFAOYSA-L calcium difluoride Chemical compound [F-].[F-].[Ca+2] WUKWITHWXAAZEY-UHFFFAOYSA-L 0.000 description 1
- 229910001634 calcium fluoride Inorganic materials 0.000 description 1
- ABDBNWQRPYOPDF-UHFFFAOYSA-N carbonofluoridic acid Chemical class OC(F)=O ABDBNWQRPYOPDF-UHFFFAOYSA-N 0.000 description 1
- 150000007942 carboxylates Chemical group 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 239000000919 ceramic Substances 0.000 description 1
- 229910052681 coesite Inorganic materials 0.000 description 1
- 239000003086 colorant Substances 0.000 description 1
- 238000010924 continuous production Methods 0.000 description 1
- 229910052802 copper Inorganic materials 0.000 description 1
- 239000010949 copper Substances 0.000 description 1
- 229910052906 cristobalite Inorganic materials 0.000 description 1
- 229910021419 crystalline silicon Inorganic materials 0.000 description 1
- 125000004122 cyclic group Chemical group 0.000 description 1
- HPXRVTGHNJAIIH-UHFFFAOYSA-N cyclohexanol Chemical compound OC1CCCCC1 HPXRVTGHNJAIIH-UHFFFAOYSA-N 0.000 description 1
- WJTCGQSWYFHTAC-UHFFFAOYSA-N cyclooctane Chemical compound C1CCCCCCC1 WJTCGQSWYFHTAC-UHFFFAOYSA-N 0.000 description 1
- 239000004914 cyclooctane Substances 0.000 description 1
- DPWCXHJKJXRDBZ-UHFFFAOYSA-N dec-5-yne-4,7-diol Chemical compound CCCC(O)C#CC(O)CCC DPWCXHJKJXRDBZ-UHFFFAOYSA-N 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 230000007547 defect Effects 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 239000003599 detergent Substances 0.000 description 1
- 229960005215 dichloroacetic acid Drugs 0.000 description 1
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 1
- 125000001028 difluoromethyl group Chemical group [H]C(F)(F)* 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 238000010894 electron beam technology Methods 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 150000002170 ethers Chemical class 0.000 description 1
- 229940035423 ethyl ether Drugs 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 125000001153 fluoro group Chemical group F* 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- JEGUKCSWCFPDGT-UHFFFAOYSA-N h2o hydrate Chemical compound O.O JEGUKCSWCFPDGT-UHFFFAOYSA-N 0.000 description 1
- 150000004820 halides Chemical group 0.000 description 1
- 150000008282 halocarbons Chemical class 0.000 description 1
- XXMIOPMDWAUFGU-UHFFFAOYSA-N hexane-1,6-diol Chemical compound OCCCCCCO XXMIOPMDWAUFGU-UHFFFAOYSA-N 0.000 description 1
- 229930195733 hydrocarbon Natural products 0.000 description 1
- 150000002430 hydrocarbons Chemical class 0.000 description 1
- 239000004615 ingredient Substances 0.000 description 1
- 238000002347 injection Methods 0.000 description 1
- 239000007924 injection Substances 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 238000010884 ion-beam technique Methods 0.000 description 1
- 150000003893 lactate salts Chemical class 0.000 description 1
- 238000002386 leaching Methods 0.000 description 1
- 235000011090 malic acid Nutrition 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- 229910021645 metal ion Inorganic materials 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- QQZOPKMRPOGIEB-UHFFFAOYSA-N n-butyl methyl ketone Natural products CCCCC(C)=O QQZOPKMRPOGIEB-UHFFFAOYSA-N 0.000 description 1
- 239000002101 nanobubble Substances 0.000 description 1
- 229920003986 novolac Polymers 0.000 description 1
- 239000002245 particle Substances 0.000 description 1
- SNGREZUHAYWORS-UHFFFAOYSA-N perfluorooctanoic acid Chemical compound OC(=O)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)C(F)(F)F SNGREZUHAYWORS-UHFFFAOYSA-N 0.000 description 1
- 238000005191 phase separation Methods 0.000 description 1
- NBIIXXVUZAFLBC-UHFFFAOYSA-K phosphate Chemical compound [O-]P([O-])([O-])=O NBIIXXVUZAFLBC-UHFFFAOYSA-K 0.000 description 1
- 239000010452 phosphate Substances 0.000 description 1
- UEZVMMHDMIWARA-UHFFFAOYSA-M phosphonate Chemical compound [O-]P(=O)=O UEZVMMHDMIWARA-UHFFFAOYSA-M 0.000 description 1
- 238000000206 photolithography Methods 0.000 description 1
- 229910021420 polycrystalline silicon Inorganic materials 0.000 description 1
- 229920001721 polyimide Polymers 0.000 description 1
- 239000002952 polymeric resin Substances 0.000 description 1
- 229920005591 polysilicon Polymers 0.000 description 1
- 229920002451 polyvinyl alcohol Polymers 0.000 description 1
- 235000019422 polyvinyl alcohol Nutrition 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- WYVAMUWZEOHJOQ-UHFFFAOYSA-N propionic anhydride Chemical compound CCC(=O)OC(=O)CC WYVAMUWZEOHJOQ-UHFFFAOYSA-N 0.000 description 1
- RUOJZAUFBMNUDX-UHFFFAOYSA-N propylene carbonate Chemical compound CC1COC(=O)O1 RUOJZAUFBMNUDX-UHFFFAOYSA-N 0.000 description 1
- 239000011241 protective layer Substances 0.000 description 1
- 150000004728 pyruvic acid derivatives Chemical class 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 239000000523 sample Substances 0.000 description 1
- 239000004065 semiconductor Substances 0.000 description 1
- HQVNEWCFYHHQES-UHFFFAOYSA-N silicon nitride Chemical compound N12[Si]34N5[Si]62N3[Si]51N64 HQVNEWCFYHHQES-UHFFFAOYSA-N 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000003892 spreading Methods 0.000 description 1
- 230000007480 spreading Effects 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 229910052682 stishovite Inorganic materials 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 150000005846 sugar alcohols Chemical class 0.000 description 1
- 229920003002 synthetic resin Polymers 0.000 description 1
- 229910052715 tantalum Inorganic materials 0.000 description 1
- GUVRBAGPIYLISA-UHFFFAOYSA-N tantalum atom Chemical compound [Ta] GUVRBAGPIYLISA-UHFFFAOYSA-N 0.000 description 1
- ISXSCDLOGDJUNJ-UHFFFAOYSA-N tert-butyl prop-2-enoate Chemical compound CC(C)(C)OC(=O)C=C ISXSCDLOGDJUNJ-UHFFFAOYSA-N 0.000 description 1
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 1
- BSYVTEYKTMYBMK-UHFFFAOYSA-N tetrahydrofurfuryl alcohol Chemical compound OCC1CCCO1 BSYVTEYKTMYBMK-UHFFFAOYSA-N 0.000 description 1
- 229910052905 tridymite Inorganic materials 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
Images
Classifications
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/20—Exposure; Apparatus therefor
- G03F7/2041—Exposure; Apparatus therefor in the presence of a fluid, e.g. immersion; using fluid cooling means
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/0042—Photosensitive materials with inorganic or organometallic light-sensitive compounds not otherwise provided for, e.g. inorganic resists
- G03F7/0043—Chalcogenides; Silicon, germanium, arsenic or derivatives thereof; Metals, oxides or alloys thereof
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/0045—Photosensitive materials with organic non-macromolecular light-sensitive compounds not otherwise provided for, e.g. dissolution inhibitors
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/004—Photosensitive materials
- G03F7/0046—Photosensitive materials with perfluoro compounds, e.g. for dry lithography
-
- G—PHYSICS
- G03—PHOTOGRAPHY; CINEMATOGRAPHY; ANALOGOUS TECHNIQUES USING WAVES OTHER THAN OPTICAL WAVES; ELECTROGRAPHY; HOLOGRAPHY
- G03F—PHOTOMECHANICAL PRODUCTION OF TEXTURED OR PATTERNED SURFACES, e.g. FOR PRINTING, FOR PROCESSING OF SEMICONDUCTOR DEVICES; MATERIALS THEREFOR; ORIGINALS THEREFOR; APPARATUS SPECIALLY ADAPTED THEREFOR
- G03F7/00—Photomechanical, e.g. photolithographic, production of textured or patterned surfaces, e.g. printing surfaces; Materials therefor, e.g. comprising photoresists; Apparatus specially adapted therefor
- G03F7/70—Microphotolithographic exposure; Apparatus therefor
- G03F7/70216—Mask projection systems
- G03F7/70341—Details of immersion lithography aspects, e.g. exposure media or control of immersion liquid supply
Definitions
- Immersion lithography may offer better resolution enhancement and higher numerical apertures at a given exposure wavelength over conventional projection lithography.
- immersion lithography could extend lithography at the 193 nm wavelength down to the 45 nm node and below thereby providing an alternative to 157 nm exposure wavelengths, extreme ultraviolet (EUV), and other potential technologies.
- EUV extreme ultraviolet
- NA numerical aperture
- immersion lithography the space between the lens and the substrate is filled with a liquid, referred to herein as an immersion fluid, that has a refractive index greater than 1.
- the immersion fluid should preferably exhibit a low optical absorption at the operating wavelength such as, for example 193 nm and 157 nm, be compatible with the photoresist and the lens material, be uniform and be non-contaminating.
- a preferred immersion fluid for 193 nm immersion lithography is ultra pure water. Ultra pure water has an index of refraction of approximately 1.44, exhibits absorption of less than 5% at working distances of up to 6 mm, is compatible with photoresist and lens, and is non-contaminating in its ultra pure form.
- immersion lithography has not been widely implemented in commercial semiconductor processing partly because improvements in resolution by conventional methods have been possible, but also partly because of practical limitations in implementing immersion lithography.
- the wafer stage of a typical 193 nm exposure tool steps from location to location across the wafer scanning the reticle image for each field. In order to achieve high throughput, the stage should accelerate rapidly, move accurately to the next field location, settle, scan the image, and then step to the next location within a short time interval.
- the immersion fluid is typically introduced between the lens and the resist surface of the substrate using a jet stream of the immersion fluid.
- the space between the lens and the resist surface referred to herein as the working distance, is less than 6 mm or typically 1 mm.
- an immersion fluid comprising: from about 10 ppm to the maximum solubility limit of at least one additive selected from an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups; an alkyl ethoxylate or a propylene (PO) derivative thereof; an alkyl carboxylate or an alkyl acid ester; an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate; an acetylenic alcohol, an acetylenic diol or ethylene oxide/propylene oxide derivatives thereof; an alkyl polyglycoside; a block oligomer or a polymer of ethylene and propylene oxide; an alkyl sulfate, an alkyl ethoxylate
- immersion fluid having a transmission of 80% or greater at an operating wavelength ranging from 140 nm to 248 nm comprising: at least one carrier medium selected from the group consisting of an aqueous fluid, a non-aqueous fluid, and mixtures thereof wherein the at least one carrier medium has a refractive index greater than or equal to water at the operating wavelength; and from about 10 ppm to the maximum solubility limit of at least one additive selected from an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups; an alkyl ethoxylate or a propylene (PO) derivative thereof; an alkyl carboxylate or an alkyl acid ester; an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate; an acetylenic alcohol, an acetylenic diol or ethylene oxide/propylene oxide derivatives thereof; an alkyl polygly
- an immersion fluid having a transmission of 80% or greater at an operating wavelength ranging from 140 nm to 248 nm comprising: at least one carrier medium selected from a non-aqueous fluid and a mixture of the non-aqueous fluid and an aqueous fluid wherein the at least one carrier medium has a refractive index greater than or equal to water at the operating wavelength and wherein if the at least one carrier medium is a mixture then the non-aqueous medium is water miscible.
- FIG. 1 provides the dynamic contact angle measurements for various embodiments of the immersion fluid of the present invention on unexposed 193 nm photoresists.
- FIG. 2 provides the absorbance spectra of ultra pure water and an embodiment of the immersion fluid of the present invention that was measured using a UV spectrometer.
- FIG. 3 provides comparison of the change in film thickness (nm) for various embodiments of the immersion fluid of the present invention compared to ultra pure water.
- Immersion fluids comprising at least one carrier medium or comprising at least one carrier medium and at least one additive, useful for performing immersion lithography at operating wavelengths ranging from 140 to 248 nm, particularly 157 nm and/or 193 nm wavelengths are disclosed herein.
- carrier medium as used herein relates to an aqueous fluid, a non-aqueous fluid, or a mixture thereof that may be used by itself or have at least one additive added thereto to provide an immersion fluid.
- the presence of the at least one additive within the at least one carrier medium such as a salt, a surfactant, an electrolyte or mixtures thereof within an immersion fluid, or the immersion fluid comprising at least one carrier medium without the addition of at least one additive may provide at least one of the following benefits: improve the wetting of the immersion fluid onto the resist-coated substrate; reduce defect formation by substantially minimizing the formation of micro-bubbles and nano-bubbles; protect the resist surface by forming aggregations of one or more monolayers at the resist-immersion fluid interface or resist-protective-layer and immersion fluid interface thereby preventing leaching of any chemicals from the resist; minimize the feature size and maximize the resolution when added into an immersion fluid with a refractive index equal to or greater than water at the operating wavelength, such as for example a refractive index equal to or greater than 1.44 at an operating wavelength of 193 nm, thereby allowing smaller feature sizes to be achieved; increase the refractive index of the immersion fluid if the carrier medium such as water has a low
- the addition of at least one additive to a carrier medium or the carrier medium itself may provide an immersion fluid does not significantly increase the absorbance of the immersion fluid at one or more operating wavelengths or maintains the absorbance below 5%, or below 1%, or below 0.5%.
- the immersion fluid containing at least one carrier medium and at least one additive, or the immersion fluid containing at least one carrier medium may exhibit 80% or greater, 90% or greater, or 95% or greater total transmission.
- the carrier medium may be an aqueous fluid, a non-aqueous fluid, or a mixture thereof that may have at least one additive added thereto to provide an immersion fluid, or alternatively, the immersion fluid may be the carrier medium itself.
- the immersion fluid may be an aqueous fluid.
- the refractive index may be equal to or greater than the refractive index of water at the operating wavelength such as, for example, a refractive index of 1.44 at an operating wavelength of 193 nm.
- the aqueous fluid transmits light at the operating wavelengths of the lithography system such as a wavelength ranging from 140 to 248 nm.
- aqueous as used herein, describes a fluid or liquid dispersing medium, which comprises at least 80 weight percent, preferably 90 weight percent, and more preferably at least 95 weight percent water.
- suitable aqueous fluids include deionized water, ultra pure water, distilled water, doubly distilled water, and high performance liquid chemical (HPLC) grade water or deionized water having a low metal content.
- the carrier medium may also be a non-aqueous fluid.
- the non-aqueous fluid is used in addition to or in place of an aqueous fluid.
- the non-aqueous fluid selected will not react with other components in the immersion fluid, the photoresist coating on the substrate, the system optics, or the substrate itself.
- the immersion fluid has at least one additive contained therein
- the non-aqueous fluid will not react with the at least one additive contained therein.
- Suitable fluids include, but are not limited to, hydrocarbons (e.g. dodecane or hexane); halocarbons (e.g. Freon 113); and ethers (e.g.
- ethylether Et 2 O
- THF tetrahydrofuran
- ethylene glycol and derivatives thereof monomethyl ether
- 2-methoxyethyl ether diglyme
- Still further exemplary fluids include lactates, pyruvates, and diols. These fluids include, but are not limited to, acetone, 1,4-dioxane, 1,3-dioxolane, ethyl acetate, cyclohexanone, acetone, and methyl ethyl ketone.
- Other fluids include dimethylformamide, dimethylacetamide, N-methylpyrrolidone, ethylene carbonate, propylene carbonate, glycerol and derivatives, acetic acid anyhydride, dichloroacetic acid, propionic acid and propionic acid anhydride, dimethyl sulfone, diphenyl sulfone, m-cresol, dimethyl sulfoxide, and the like.
- Still further fluids include citric acid, bicyclohexyl, propylene glycol propyl ether (PGPE), methanol, tetrahydrofurfuryl alcohol, 1-methylcyclohexanol, cyclohexanol, trans-2-methylcyclohexanol, 1,5-pentanediol, 1,6-hexanediol, 1,4-butanediol, 1,2-propylene glycol, 1,3-propylene glycol, 1-dodecanol, cyclooctane, ethanol, 3-heptanol, 2-methyl-1-pentanol, 5-methyl-2-hexanol, cis-2-methylcyclohexanol, 3-hexanol, 2-heptanol, 2-hexanol, 2,3-dimethyl-3-pentanol, propylene glycol methyl ether acetate (PGMEA), ethylene glycol and derivatives thereof, polyethylene glyco
- the carrier medium may contain a mixture of at least one aqueous fluid and at least one non-aqueous fluid.
- the immersion fluid may contain at least one non-aqueous fluid that is miscible in the aqueous fluid or is water-miscible.
- the amount of non-aqueous fluid within the immersion fluid may range from about 1 to about 99%, or from about 1 to about 50% by weight with the balance of the carrier medium within the immersion fluid comprising an aqueous fluid.
- water-miscible non-aqueous fluids include methanol, ethanol, isopropyl alcohol, glycerol, ethylene glycol and derivatives thereof, polyethylene glycol and derivatives thereof and THF.
- certain non-aqueous fluids having a refractive index greater than or equal to that of water and a specific absorbance of less than 1 cm ⁇ 1 or less than 0.5 cm ⁇ 1 at one or more operating wavelengths ranging from 140 to 248 nm may be added to the immersion fluid in the amount ranging from 0.1 to 100%, or from 1 to 50% to increase the refractive index of the immersion fluid.
- the non-aqueous fluid may have a refractive index equal to or greater than that of water or greater than 1.44.
- the immersion fluid comprises from 10 parts per million (ppm) to the maximum solubility limit, from 10 ppm to 10,000 ppm of at least one additive.
- maximum solubility limit as used herein relates to the maximum amount of the at least one additive that can be added to the carrier medium to provide a homogenous solution without phase separation and/or precipitation of the at least one additive.
- Examples of at least one additive that may be used alone, or in combination with one or more other at least one additives, within an immersion fluid include: an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups; an alkyl ethoxylate or propylene (PO) derivatives thereof including mono- and multi-hydrophilic units (such as diols); an alkyl carboxylate or an alkyl acid ester with mono- and multi-carboxyl units; an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate; an acetylenic alcohol, an acetylenic diol or ethylene oxide/propylene oxide derivatives thereof; an alkyl polyglycoside; a block oligomer or a polymer of ethylene and propylene oxide; an alkyl sulfate, an alkyl ethoxylate sulfate, an alkyl sulfonate,
- At least one additive is a surfactant.
- Typical surfactants exhibit an amphiphilic nature, meaning that they can be both hydrophilic and hydrophobic at the same time.
- Amphiphilic surfactants possess a hydrophilic head group or groups, which have a strong affinity for water and a long hydrophobic tail, which is organophilic and repels water.
- the surfactant may be ionic (i.e., anionic, cationic, amphoteric) or nonionic.
- the immersion fluid may contain at least one additive that is an acetylenic alcohol, an acetylenic diol, or an ethylene oxide/propylene oxide derivative thereof.
- exemplary acetylenic alcohol, acetylenic diol or ethylene oxide/propylene oxide derivatives that can be used as the at least one additive within an immersion fluid may be represented by the following formulas I through III: wherein R 1 and R 4 are each independently a straight or a branched alkyl chain having from 3 to 10 carbon atoms; R 2 and R 3 are each independently a hydrogen atom or an alkyl chain having from 1 to 5 carbon atoms; and m, n, p, and q are each independently a number that ranges from 0 to 20.
- the at least one additive having the formula I, II or III are commercially available from Air Products and Chemicals, Inc. of Allentown, Pa., the assignee of the present invention, under the trade names SURFYNOL® and DYNOL®.
- the acetylenic diol portion of the molecule of formulas 1 or 11 is 2,4,5,9-tetramethyl-5-decyne-4,7-diol or 2,5,8,11-tetramethyl-6-dodecyne-5,8-diol.
- the at least one additives having the formulas I through III may be prepared in a number of ways including the methods described, for example, in U.S. Pat. No. 6,313,182 and EP 1115035A1, which are assigned to the assignee of the present invention and incorporated herein by reference in their entirety.
- the alkylene oxide moieties represented by (OC 2 H 4 ) are the (n+m) polymerized ethylene oxide (EO) molar units and the moieties represented by (OC 3 H 6 ) are the (p+q) polymerized propylene oxide (PO) molar units.
- the value of (n+m) may range from 0 to 30, preferably from 1.3 to 15, and more preferably from 1.3 to 10.
- the value of (p+q) may range from 0 to 30, preferably from 1 to 10, and more preferably from 1 to 2.
- the immersion fluid may contain from 10 ppm to the maximum solubility limit or from 10 ppm to 10,000 ppm of at least one additive that is represented by the following formulas (IV) through (XI):
- R, R 1 and R 4 are each independently a straight, branched, or cyclic alkyl, fluoroalkyl, or perfluoroalkyl group having from 2 to 25, or from 3 to 10 carbon atoms;
- R 2 and R 3 are each independently a hydrogen atom, a straight, a branched or a cyclic alkyl group, fluoroalkyl group, or perfluoroalkyl group having from 1 to 10 or from 1 to 5 carbon atoms;
- R 5 is a straight, a branched, or a cyclic alkyl, fluoroalkyl, or perfluoroalkyl group having from 1 to 10 carbon atoms;
- R 6 is a straight, a branched, or a cyclic alkyl, fluoroalkyl, or perfluoroalkyl group having from 4 to 16 carbon atoms;
- R 7 , R 8 and R 9 are each independently a straight, a branched, or a
- the at least one additive may be an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups.
- Exemplary alkyl alcohols or polymeric alcohols include sugar alcohols such as sorbitol or polyvinyl alcohols.
- the at least one additive may be an alkyl alcohol, an alkyl ethoxylate, or a propylene oxide derivative thereof.
- Exemplary alkyl alcohols, alkyl ethoxylates, or propylene oxide derivatives that can be used as at least one additive within an immersion fluid may be represented by formulas IVa through IVe.
- a specific example of a Formula IVd additive is 2,4,7,9-tetramethyl-4,7-decane diol.
- Formula IVe additives include, but are not limited to, 3,5-dimethyl-1-hexyn-3-ol and 2,6-dimethyl-4-heptanol.
- the at least one additive may be an alkyl carboxylate or an alkyl acid ester.
- Exemplary alkyl carboxylates or alkyl acid esters that can be used as at least one additive within an immersion fluid may be represented by formulas Va through Vc.
- An example of a Formula Va additive includes, but is not limited to, diisopentyl tartrate.
- the at least one additive may be an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate.
- Exemplary alkyl amines or alkyl amine ethoxylates that can be used as at least one additive within an immersion fluid may be represented by formulas VIa through VIe.
- An example of a Formula VIa additive includes, but is not limited to, N,N′-bis(1,3-dimethylbutyl) ethylene diamine.
- the at least one additive may be an alkyl polyglycoside.
- An exemplary alkyl polyglycoside that can be used as at least one additive within an immersion fluid is represented by formula VII.
- the at least one additive may be a block oligomer or a polymer of ethylene and propylene oxide.
- Exemplary block oligomers or polymers of ethylene and propylene oxide that can be used as at least one additive within an immersion fluid may be represented by formulas VIIIa through VIIIc.
- the at least one additive may be an alkyl sulfate, an alkyl ethoxylate sulfate, an alkyl sulfonate, or an alkyl ethyoxylate sulfonate.
- the at least one additive may be an alkyl ammonium salt.
- the at least one additive may be a glycidal ether or a glucamine derivative with an alkyl amine, an alkyl diamine, an alkyl alcohol, or an acetylenic alcohol.
- Exemplary glycidal ether or glucamine derivatives that can be used as at least one additive within an immersion fluid may be represented by formulas IXa through IXb.
- An example of a Formula IXb additive includes, but is not limited to, an adduct of diethylenetriamine and n-butyl glycidyl ether.
- the at least one additive may be an alkyl urea or a dialkyl urea.
- the at least one additive may be a fluorinated or partially fluorinated acetylenic alcohol or diol and derivates thereof.
- Exemplary fluorinated or partially fluorinated acetylenic alcohol or diol and derivates thereof that can be used as at least one additive within an immersion fluid may be represented by formulas XIa through Xle.
- An example of a Formula XIa additive includes, but is not limited to, hexafluoropropanol acetylene.
- the at least one additive may be a fluorosurfactant provided that the carrier medium comprises at least 1% by weight or greater of an aqueous fluid.
- fluorosurfactants include: straight, branched, or cyclic hydrofluorocarbons having 2 to 10 carbon atoms wherein there are more fluorine atoms than hydrogen atoms; F[CF(CF 2 )CF 2 ] n —O—[CH 2 CH 2 O] m —H; F(CF 2 (CF 3 )CF 2 O] n CFHCF 3 wherein n is a number ranging from 1 to 5; F[CF(CF 3 )CF 2 O] n CF 2 CF 3 wherein n is a number ranging from 1 to 5; and HCF 2 (OCF 2 ) n (OCF 2 CF 2 ) m —O—CF 2 H wherein (n+m) is a number ranging from 1 to 8; a mixture of the ammonium salts of perfluorocarbox
- the at least one additive may be a salt having a refractive index equal to or greater than that of water and a specific absorbance less than 1 cm ⁇ 1 , or less than 0.5 cm ⁇ 1 , at an operating wavelength ranging from 140 to 248 nm. In embodiments wherein the operating wavelength is 193 nm, the refractive index is equal to or greater than 1.44.
- the at least one additive may be an electrolyte having a refractive index equal to or greater than that of water and a specific absorbance less than 1 cm ⁇ 1 , or less than 0.5 cm ⁇ 1 , at an operating wavelength ranging from 140 to 248 nm. In embodiments wherein the operating wavelength is 193 nm, the refractive index is equal to or greater than 1.44.
- Various other components may be optionally added to the immersion fluid. These components may include, but are not limited to, stabilizers, dissolving aids, colorants, wetting agents, antifoamers, buffering agents, and other additional surfactants. Generally, the amount of each of these additives would be about 0.0001 to 1 percent by weight, or about 0.0001 to 0.1 percent by weight, based upon the total weight of the immersion fluid. In embodiments where one or more additional surfactants are added to the immersion fluid, the surfactant may be any of the surfactants disclosed herein or provided in the reference McCutcheon's Emulsifiers and Detergents.
- the immersion fluid may be prepared by mixing the at least one additive with at least one carrier medium which can be an aqueous and/or non-aqueous fluid and any additional components.
- at least one carrier medium which can be an aqueous and/or non-aqueous fluid and any additional components.
- one or more aqueous fluids such as ultra pure or HPLC water is combined with from about 10 ppm to the maximum solubility limit, or from about 10 ppm to about 10,000 ppm, of at least one additive to provide the immersion fluid.
- from about 10 ppm to the maximum solubility limit, or from about 10 ppm to about 10,000 ppm, of at least one additive is combined with one or more aqueous fluids until a homogeneous mixture is formed and is then combined with one or more non-aqueous fluids to provide the immersion fluid.
- one or more non-aqueous fluids such as the fluids disclosed herein are combined with from about 10 ppm to the maximum solubility limit of at least one additive to provide the immersion fluid.
- the combining may be done at a temperature range of about 20 to 60° C. or from 40 to 60° C. to affect dissolution of the ingredients contained therein and provide a homogeneous mixture.
- the resulting immersion fluid may optionally be filtered to remove any undissolved particles that could potentially harm the substrate.
- the immersion fluid may comprise the carrier medium, i.e., at least one aqueous fluid, at least one non-aqueous fluid, and mixtures thereof.
- the combining may be done at a temperature range of about 20 to 60° C. or from 40 to 60° C. to provide a homogeneous mixture.
- the immersion fluid is preferably used to treat at least a portion of the surface of a substrate that is coated with a photoresist or resist coating.
- Suitable substrates include, but are not limited to, materials such as gallium arsenide (“GaAs”), silicon, tantalum, copper, ceramics, aluminum/copper alloys, polyimides, and compositions containing silicon such as crystalline silicon, polysilicon, amorphous silicon, epitaxial silicon, silicon dioxide (“SiO 2 ”), silicon nitride, doped silicon dioxide, and the like.
- Further exemplary substrates include silicon, aluminum, or polymeric resins.
- the immersion fluid contacts at least a portion of a substrate coated with a photoresist, or a resist-coated substrate and is substantially non-reactive to the resist coating.
- exemplary resist coatings include, but are not limited to, novolac resin, polyvinyl phenol copolymer, or copolymers of p-hydroxystyrene and t-butyl acrylate.
- the immersion fluid is applied to at least a portion of a substrate having a photoresist coating applied thereto.
- the photoresist-coated substrate is then exposed to radiation through an optical device to provide a pattern that is imposed upon the photoresist coating.
- radiation sources include ultraviolet (uv) light, electron beam, x-ray, laser, lamp, or ion beams.
- the radiation source emits light at wavelengths at an operating wavelength ranging from 140 nm to 248 nm such as 193 nm and 157 nm.
- a pre-bake or soft-bake step may be conducted prior to the exposure step. This pre-bake or soft bake step may be conducted, for example, at a temperature ranging from 90° C. to 150° C. for a time or from 30 to 120 seconds on a hot plate.
- the radiation either increases or decreased its solubility in a subsequently applied alkaline developer solution such as a process solution containing tetramethylammonium hydroxide (TMAH), potassium hydroxide, sodium hydroxide, or other base.
- alkaline developer solution such as a process solution containing tetramethylammonium hydroxide (TMAH), potassium hydroxide, sodium hydroxide, or other base.
- developer solutions include those provided in U.S. Pat. Nos. 6,455,234; 6,268,115; 6,238,849; 6,127,101; and 6,120,978.
- TMAH tetramethylammonium hydroxide
- the immersion fluid of the present invention may be suitable to treat substrates having either positive or negative photoresist coatings.
- the patterned photoresist image may be developed by a variety of different means, including by not limited to quiescence, immersion, spray, or puddle development.
- the substrate is baked to harden the polymer contained within the photoresist.
- the bake step may be conducted, for example, at a temperature ranging from 70° C. to 150° C. for a time duration of from 30 to 120 seconds.
- a typical immersion lithography process uses an apparatus that has a servo motor driven wafer stage that supports and positions a resist-coated substrate or wafer underneath an optic device such as a lens.
- the optic device may also be a prism, a mirror or combinations thereof.
- the immersion fluid is dispensed onto at least a portion of resist-coated substrate through one or more nozzles to form a puddle.
- a radiation source that emits light at the operating wavelength then passes through lens and the puddle of immersion fluid prior to exposure to at least a portion of the resist-coated substrate.
- the immersion fluid is preferably applied to the photoresist-coated substrate as a prepared solution.
- the immersion fluid can be prepared within the application stream just prior to or during contact with the substrate surface.
- a certain quantity of one or more additives can be injected into a continuous stream of water and/or non-aqueous fluid medium or immersion fluid that optionally includes other components thereby forming the immersion fluid.
- a portion of the at least one additive may be added to the substrate after application of the immersion fluid.
- the at least one additive can be also deposited upon or comprise the material of a high surface area device such as a cartridge or filter (which may or may not include other components).
- a stream of water and/or non-aqueous fluid then passes through the cartridge or filter thereby forming the immersion fluid.
- the immersion fluid is prepared during the contacting step.
- at least one additive is introduced via a dropper or other means to the surface of the substrate.
- Water and/or non-aqueous fluid medium is then introduced to the surface of the substrate and mixes with the at least one additive on the surface of the substrate thereby forming the immersion fluid.
- a concentrated composition comprising at least one additive is provided that may be diluted in water and/or non-aqueous fluids to provide the immersion fluid.
- a concentrated composition of the invention, or “concentrate” allows one to dilute the concentrate to the desired strength and pH.
- a concentrate also permits longer shelf life and easier shipping and storage of the product.
- a variety of means can be employed in contacting the immersion fluid with the substrate surface.
- the actual conditions of the contacting step i.e., temperature, time, and the like
- the contact step can be conducted in either a dynamic method such as, for example, a streamline process for applying the immersion fluid over the surface of the substrate or in a static method such as, for example, a puddle application.
- the immersion fluid may also be sprayed onto the surface of the substrate in a dynamic method such as in a continuous process or sprayed onto the surface and allowed to remain there in a static method.
- the duration of the contacting step, or time of contact of the immersion fluid to the substrate surface can vary from a fraction of a second to hundreds of seconds. Preferably, the duration can range from 1 to 200 seconds, or from 1 to 150 seconds, or from 1 to 40 seconds.
- the temperature range for the contacting step can vary from 10 to 100° C., or from 10 to 40° C.
- the dynamic surface tension (DST) data was collected via the maximum bubble pressure method described in Langmuir 1986, 2, pp. 428-432. The data was collected at bubble rates that range from 0.1 bubbles/second (b/s) to 20 b/s using the Kruss BP2 bubble pressure tensiometer manufactured by Kruss, Inc. of Charlotte, N.C.
- the dynamic surface tension data provides information about the performance of the at least one additive at conditions from near-equilibrium (0.1 b/s) to relatively high surface creation rates (20 b/s).
- High bubble rates may correspond to a dynamic liquid injection process in an immersion lithography process. It is desirable that the dynamic surface tension by reduced below that of ultrapure water at high bubble rates (i.e., 70-72 dyne/cm at 20 b/s) to provide, inter alia, better wetting of the photoresist-coated substrate.
- Table I provides the DST of immersion fluids containing varying concentrations of the alkyl diol additive 2,4,7,9-tetramethyl-4,7-decane diol added to a 100 mL quantity of ultra pure water.
- the alkyl diol additive is added to the ultra pure water at ambient temperature while stirring to provide a homogeneous solution.
- the immersion fluids exhibited dynamic surface tensions at high bubble rates below that of water or 72 dyne/cm. This indicates that the immersion fluid of the present invention may be effective at reducing the surface tension of water in a dynamic process.
- TABLE I Dynamic Surface Tension DST DST DST DST DST Additives (dyne/cm) (dyne/cm) (dyne/cm) (dyne/cm) (dyne/cm) Conc.
- a quantity of 0.01% by weight or 100 ppm of the following additives: hydrophobically modified acetylenic diol or oxirane [(2-Ethyl Hexyl) Oxy]Methyl]-, Rx prod w/polyethylene glycol ether with 2,4,7,9-tetramethyl-5-decyne-4,7-diol (2:1); 2,4,7,9-tetramethyl-4,7-decane diol; ethoxylated 2,4,7,9-tetramethyl-4,7-decane diol; polysiloxane-polyester copolymer or TEGOWETTM manufactured by Goldschmidt Chemical of McDonald, Pa.; and an ethoxylated nonionic fluorosurfactant or ZONYLTM FSO with the formula: F(CF 2 CF 2 )1-7CH 2 CH 2 O(CH 2 CH 2 O) y H where y 0 to ca.
- the wetting properties of a localized region on the surface of a photoresist-coated substrate are estimated by measuring the contact angle between the baseline of a droplet of aqueous developer solution and the tangent at the droplet base.
- a high-speed camera captured the spreading of the droplet at a speed of 2 frames per second for 2 minutes and the contact angle was measured.
- the photoresist is a typical acrylate type 193 nm resist.
- the absorbance of immersion fluids was measured with a UV spectrometer. The wavelength was scanned from 210 nm to 185 nm. As shown in FIG. 1 , at 193 nm wavelength, the addition of 0.02 wt % of the additive 2,4,7,9-tetramethyl-4,7-decane diol only increased the absorbance slightly. The additive is enough to lower the contact angle on resist by 15% while only adding 0.03-0.05 cm ⁇ 1 to the absorbance. This absorbance is low enough to allow 1 mm working distance between optics and resist surface which maintaining the total transmission >95%. The improved wetting may lead to more uniform image formation across the wafer and enhance the image resolution.
- a quartz crystal microbalance was used to study changes in film thickness of unexposed 193 nm photoresist solutions.
- a CH Instruments model CHI 405 was used as a driver and high-resolution frequency counter. The instrument was fitted with a flow cell. The quartz crystals were made by International Crystal Manufacturing. The electrode was gold, 1000 ⁇ thick and 0.201′′ in diameter. The crystal resonance frequency was 7.995 MHz +/ ⁇ 10 Hz. The quartz crystals were spin-coated with a photoresist solution. The spin recipe was 1200 revolutions per minute (RPM) for 30 seconds and 3000 RPM for 10 seconds. The post apply bake (PAB) was 4 min at 125° C. The crystals were then exposed to UV light for the desired time.
- RPM revolutions per minute
- PAB post apply bake
Landscapes
- Physics & Mathematics (AREA)
- General Physics & Mathematics (AREA)
- Engineering & Computer Science (AREA)
- Metallurgy (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Exposure Of Semiconductors, Excluding Electron Or Ion Beam Exposure (AREA)
- Exposure And Positioning Against Photoresist Photosensitive Materials (AREA)
Abstract
Suitable additives that may be added into immersion fluids, immersion fluids comprising at least one carrier medium selected from an aqueous fluid, a non-aqueous fluid, and mixtures thereof, and immersions fluids comprising at least one carrier medium and at least one additive useful for performing immersion lithography at an operating wavelength ranging from 140 nm to 248 nm are disclosed herein.
Description
- Immersion lithography may offer better resolution enhancement and higher numerical apertures at a given exposure wavelength over conventional projection lithography. For example, immersion lithography could extend lithography at the 193 nm wavelength down to the 45 nm node and below thereby providing an alternative to 157 nm exposure wavelengths, extreme ultraviolet (EUV), and other potential technologies.
- The minimum feature width (W) that may be printed with an optical lithography system is determined by the Rayleigh equation: W=(k1λ)/(NA) where k1 is the resolution factor, λ is the wavelength of the exposing radiation and NA is the numerical aperture. Numerical aperture (NA) is determined using the equation: NA=n sin α where n is the index of refraction of the medium surrounding the lens and α is the acceptance angle of the lens. The physical limit to NA for exposure systems using air as a medium between the lens and the wafer is 1. Air is the worst medium because its index of refraction may cause a relatively high amount of bending when light leaves the glass. Since the index of refraction for water and glass is approximately 1.44 and 1.52 respectively, far less bending occurs thereby providing a sharper and deeper focus.
- In immersion lithography, the space between the lens and the substrate is filled with a liquid, referred to herein as an immersion fluid, that has a refractive index greater than 1. The immersion fluid should preferably exhibit a low optical absorption at the operating wavelength such as, for example 193 nm and 157 nm, be compatible with the photoresist and the lens material, be uniform and be non-contaminating. A preferred immersion fluid for 193 nm immersion lithography is ultra pure water. Ultra pure water has an index of refraction of approximately 1.44, exhibits absorption of less than 5% at working distances of up to 6 mm, is compatible with photoresist and lens, and is non-contaminating in its ultra pure form. Applying the Rayleigh equation using n=1.44 and assuming sin a can reach 0.93, the feature width for 193 nm could reach a theoretical minimum resolution of 36 nm. Still other immersion fluids that have been considered for 15 nm immersion lithography are KRYTOX™ and perfluoropolyether (PFPE).
- To date, immersion lithography has not been widely implemented in commercial semiconductor processing partly because improvements in resolution by conventional methods have been possible, but also partly because of practical limitations in implementing immersion lithography. The wafer stage of a typical 193 nm exposure tool steps from location to location across the wafer scanning the reticle image for each field. In order to achieve high throughput, the stage should accelerate rapidly, move accurately to the next field location, settle, scan the image, and then step to the next location within a short time interval. The immersion fluid is typically introduced between the lens and the resist surface of the substrate using a jet stream of the immersion fluid. The space between the lens and the resist surface, referred to herein as the working distance, is less than 6 mm or typically 1 mm. Due to a variety of factors such as short process cycle time, minimal working distance, and dynamics of the immersion stream, maintaining a consistent bubble free liquid between the lens and the resist-coated wafer is very difficult. Further, there is a lack of immersion fluids that have appropriate optical transmission characteristics and chemical compatibility with lithographic systems.
- The desire to develop immersion systems is growing more acute because the ability to achieve resolution improvements via conventional means, such as wavelength reduction, appears to be increasingly difficult, particularly at wavelengths below 248 nm. In addition, with numerical apertures or NAs produced by lithographic methods using air as the immersion medium approaching the theoretical limit, progress using conventional methods is bounded. Accordingly, there is a need for an immersion fluid that is compatible with immersion lithographic systems, particularly those systems having an operative wavelength below 248 nm.
- Immersion fluids comprising at least one carrier medium, immersion fluids comprising at least one carrier medium and at least one additive, and the additives used therein are disclosed herein. In one aspect of the present invention, there is provided an immersion fluid comprising: from about 10 ppm to the maximum solubility limit of at least one additive selected from an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups; an alkyl ethoxylate or a propylene (PO) derivative thereof; an alkyl carboxylate or an alkyl acid ester; an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate; an acetylenic alcohol, an acetylenic diol or ethylene oxide/propylene oxide derivatives thereof; an alkyl polyglycoside; a block oligomer or a polymer of ethylene and propylene oxide; an alkyl sulfate, an alkyl ethoxylate sulfate, an alkyl sulfonate, or an alkyl ethoxylate sulfonate; an alkyl ammonium salt; a glycidal ether or a glucamine derivative with an alkyl amine, an alkyl diamine, an alkyl alcohol, or an acetylenic alcohol; an alkyl urea or a dialkyl urea; a polysiloxane, a poly(dimethyl)siloxane, a polysiloxane polyester copolymer or derivatives thereof; a fluorinated or partially fluorinated acetylenic alcohol, diol or derivates thereof; a fluorosurfactant; a salt; and an electrolyte; wherein the salt and the electrolyte have a specific absorbance <1 cm−1 at an operating wavelength ranging from 140 nm to 248 nm and a refractive index equal to or greater than water at the operating wavelength, provided that if the at least one additive is a fluorosurfactant then the immersion fluid comprises about 1% by weight or greater of an aqueous fluid.
- In another aspect of the invention, there is provided immersion fluid having a transmission of 80% or greater at an operating wavelength ranging from 140 nm to 248 nm comprising: at least one carrier medium selected from the group consisting of an aqueous fluid, a non-aqueous fluid, and mixtures thereof wherein the at least one carrier medium has a refractive index greater than or equal to water at the operating wavelength; and from about 10 ppm to the maximum solubility limit of at least one additive selected from an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups; an alkyl ethoxylate or a propylene (PO) derivative thereof; an alkyl carboxylate or an alkyl acid ester; an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate; an acetylenic alcohol, an acetylenic diol or ethylene oxide/propylene oxide derivatives thereof; an alkyl polyglycoside; a block oligomer or a polymer of ethylene and propylene oxide; an alkyl sulfate, an alkyl ethoxylate sulfate, an alkyl sulfonate, or an alkyl ethoxylate sulfonate; an alkyl ammonium salt; a glycidal ether or a glucamine derivative with an alkyl amine, an alkyl diamine, an alkyl alcohol, or an acetylenic alcohol; an alkyl urea or a dialkyl urea; a polysiloxane, a poly(dimethyl)siloxane, a polysiloxane polyester copolymer, or derivatives thereof; a fluorinated or partially fluorinated acetylenic alcohol, diol or derivates thereof; a fluorosurfactant; a salt; and an electrolyte; wherein the salt and the electrolyte have a specific absorbance <1 cm−1 at an operating wavelength ranging from 140 nm to 248 nm and a refractive index equal to or greater than water at the operating wavelength, provided that if the at least one additive is a fluorosurfactant then the immersion fluid comprises about 1% by weight or greater of an aqueous fluid.
- In a further aspect of the present invention, there is provided an immersion fluid having a transmission of 80% or greater at an operating wavelength ranging from 140 nm to 248 nm comprising: at least one carrier medium selected from a non-aqueous fluid and a mixture of the non-aqueous fluid and an aqueous fluid wherein the at least one carrier medium has a refractive index greater than or equal to water at the operating wavelength and wherein if the at least one carrier medium is a mixture then the non-aqueous medium is water miscible.
-
FIG. 1 provides the dynamic contact angle measurements for various embodiments of the immersion fluid of the present invention on unexposed 193 nm photoresists. -
FIG. 2 provides the absorbance spectra of ultra pure water and an embodiment of the immersion fluid of the present invention that was measured using a UV spectrometer. -
FIG. 3 provides comparison of the change in film thickness (nm) for various embodiments of the immersion fluid of the present invention compared to ultra pure water. - Immersion fluids, comprising at least one carrier medium or comprising at least one carrier medium and at least one additive, useful for performing immersion lithography at operating wavelengths ranging from 140 to 248 nm, particularly 157 nm and/or 193 nm wavelengths are disclosed herein. The term “carrier medium” as used herein relates to an aqueous fluid, a non-aqueous fluid, or a mixture thereof that may be used by itself or have at least one additive added thereto to provide an immersion fluid. It is believed that the presence of the at least one additive within the at least one carrier medium such as a salt, a surfactant, an electrolyte or mixtures thereof within an immersion fluid, or the immersion fluid comprising at least one carrier medium without the addition of at least one additive, may provide at least one of the following benefits: improve the wetting of the immersion fluid onto the resist-coated substrate; reduce defect formation by substantially minimizing the formation of micro-bubbles and nano-bubbles; protect the resist surface by forming aggregations of one or more monolayers at the resist-immersion fluid interface or resist-protective-layer and immersion fluid interface thereby preventing leaching of any chemicals from the resist; minimize the feature size and maximize the resolution when added into an immersion fluid with a refractive index equal to or greater than water at the operating wavelength, such as for example a refractive index equal to or greater than 1.44 at an operating wavelength of 193 nm, thereby allowing smaller feature sizes to be achieved; increase the refractive index of the immersion fluid if the carrier medium such as water has a low absorbance at wavelengths ranging from 140 to 248 nm provided that there is no interaction between the photoresist and the optics; and minimize the change in feature size upon exposure to light or heat by adding at least one additive to the carrier medium having the opposite refractive index/temperature characteristics (dn/dt) that may minimize or eliminate the change in refractive index upon exposure to laser light or heat. Further, the addition of at least one additive to a carrier medium or the carrier medium itself may provide an immersion fluid does not significantly increase the absorbance of the immersion fluid at one or more operating wavelengths or maintains the absorbance below 5%, or below 1%, or below 0.5%. The immersion fluid containing at least one carrier medium and at least one additive, or the immersion fluid containing at least one carrier medium, may exhibit 80% or greater, 90% or greater, or 95% or greater total transmission.
- As mentioned previously, the carrier medium may be an aqueous fluid, a non-aqueous fluid, or a mixture thereof that may have at least one additive added thereto to provide an immersion fluid, or alternatively, the immersion fluid may be the carrier medium itself. In certain embodiments, the immersion fluid may be an aqueous fluid. In these embodiments, the refractive index may be equal to or greater than the refractive index of water at the operating wavelength such as, for example, a refractive index of 1.44 at an operating wavelength of 193 nm. Further, the aqueous fluid transmits light at the operating wavelengths of the lithography system such as a wavelength ranging from 140 to 248 nm. The term “aqueous” as used herein, describes a fluid or liquid dispersing medium, which comprises at least 80 weight percent, preferably 90 weight percent, and more preferably at least 95 weight percent water. Examples of suitable aqueous fluids include deionized water, ultra pure water, distilled water, doubly distilled water, and high performance liquid chemical (HPLC) grade water or deionized water having a low metal content.
- The carrier medium may also be a non-aqueous fluid. In these embodiments, the non-aqueous fluid is used in addition to or in place of an aqueous fluid. The non-aqueous fluid selected will not react with other components in the immersion fluid, the photoresist coating on the substrate, the system optics, or the substrate itself. In embodiments wherein the immersion fluid has at least one additive contained therein, the non-aqueous fluid will not react with the at least one additive contained therein. Suitable fluids include, but are not limited to, hydrocarbons (e.g. dodecane or hexane); halocarbons (e.g. Freon 113); and ethers (e.g. ethylether (Et2O), tetrahydrofuran (“THF”), ethylene glycol and derivatives thereof, monomethyl ether, or 2-methoxyethyl ether (diglyme)). Still further exemplary fluids include lactates, pyruvates, and diols. These fluids include, but are not limited to, acetone, 1,4-dioxane, 1,3-dioxolane, ethyl acetate, cyclohexanone, acetone, and methyl ethyl ketone. Other fluids, include dimethylformamide, dimethylacetamide, N-methylpyrrolidone, ethylene carbonate, propylene carbonate, glycerol and derivatives, acetic acid anyhydride, dichloroacetic acid, propionic acid and propionic acid anhydride, dimethyl sulfone, diphenyl sulfone, m-cresol, dimethyl sulfoxide, and the like. Still further fluids include citric acid, bicyclohexyl, propylene glycol propyl ether (PGPE), methanol, tetrahydrofurfuryl alcohol, 1-methylcyclohexanol, cyclohexanol, trans-2-methylcyclohexanol, 1,5-pentanediol, 1,6-hexanediol, 1,4-butanediol, 1,2-propylene glycol, 1,3-propylene glycol, 1-dodecanol, cyclooctane, ethanol, 3-heptanol, 2-methyl-1-pentanol, 5-methyl-2-hexanol, cis-2-methylcyclohexanol, 3-hexanol, 2-heptanol, 2-hexanol, 2,3-dimethyl-3-pentanol, propylene glycol methyl ether acetate (PGMEA), ethylene glycol and derivatives thereof, polyethylene glycol and derivatives thereof, isopropyl alcohol (IPA), n-butyl ether, propylene glycol n-butyl ether (PGBE), 1-butoxy-2-propanol, 2-methyl-3-pentanol, 2-methoxyethyl acetate, 2-butoxyethanol, 2-ethoxyethyl acetoacetate, 1-pentanol, and propylene glycol methyl ether. The non-aqueous fluids enumerated above may be used alone, in combination with two or more fluids, or in combination with an aqueous fluid.
- The carrier medium may contain a mixture of at least one aqueous fluid and at least one non-aqueous fluid. In these embodiments, the immersion fluid may contain at least one non-aqueous fluid that is miscible in the aqueous fluid or is water-miscible. The amount of non-aqueous fluid within the immersion fluid may range from about 1 to about 99%, or from about 1 to about 50% by weight with the balance of the carrier medium within the immersion fluid comprising an aqueous fluid. Examples of water-miscible non-aqueous fluids include methanol, ethanol, isopropyl alcohol, glycerol, ethylene glycol and derivatives thereof, polyethylene glycol and derivatives thereof and THF.
- In certain embodiments, certain non-aqueous fluids, having a refractive index greater than or equal to that of water and a specific absorbance of less than 1 cm−1 or less than 0.5 cm−1 at one or more operating wavelengths ranging from 140 to 248 nm may be added to the immersion fluid in the amount ranging from 0.1 to 100%, or from 1 to 50% to increase the refractive index of the immersion fluid. At an operating wavelength of 193 nm, the non-aqueous fluid may have a refractive index equal to or greater than that of water or greater than 1.44. Exemplary non-aqueous fluids that may be used at this operating wavelength include, but are not limited to, citric acid (n=1.496), bicyclohexyl (n=1.477), glycerol (n=1.4730), or cis-2-methylcyclohexanol (n 1.4633).
- In certain embodiments, the immersion fluid comprises from 10 parts per million (ppm) to the maximum solubility limit, from 10 ppm to 10,000 ppm of at least one additive. The term “maximum solubility limit” as used herein relates to the maximum amount of the at least one additive that can be added to the carrier medium to provide a homogenous solution without phase separation and/or precipitation of the at least one additive. Examples of at least one additive that may be used alone, or in combination with one or more other at least one additives, within an immersion fluid include: an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups; an alkyl ethoxylate or propylene (PO) derivatives thereof including mono- and multi-hydrophilic units (such as diols); an alkyl carboxylate or an alkyl acid ester with mono- and multi-carboxyl units; an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate; an acetylenic alcohol, an acetylenic diol or ethylene oxide/propylene oxide derivatives thereof; an alkyl polyglycoside; a block oligomer or a polymer of ethylene and propylene oxide; an alkyl sulfate, an alkyl ethoxylate sulfate, an alkyl sulfonate, or an alkyl ethoxylate sulfonate; an alkyl ammonium salt; a glycidal ether or a glucamine derivative with an alkyl amine, an alkyl diamine, an alkyl alcohol, or an acetylenic alcohol; an alkyl urea or a dialkyl urea; a polysiloxane, a poly(dimethyl)siloxane, a polyester copolymer or derivatives thereof; a fluorinated or partially fluorinated acetylenic alcohol, diol or derivates thereof; a fluorosurfactant; a salt having a refractive index greater than or equal to that of water and a specific absorbance of less than 1 cm−1 at an operating wavelength ranging from 140 to 248 nm; and an electrolyte having a refractive index greater than or equal to that of water and a specific absorbance of less than 1 cm−1 at an operating wavelength ranging from 140 to 248 nm.
- In certain embodiments, at least one additive is a surfactant. Typical surfactants exhibit an amphiphilic nature, meaning that they can be both hydrophilic and hydrophobic at the same time. Amphiphilic surfactants possess a hydrophilic head group or groups, which have a strong affinity for water and a long hydrophobic tail, which is organophilic and repels water. In embodiments wherein the at least one additive is a surfactant, the surfactant may be ionic (i.e., anionic, cationic, amphoteric) or nonionic.
- In certain embodiments of the present invention, the immersion fluid may contain at least one additive that is an acetylenic alcohol, an acetylenic diol, or an ethylene oxide/propylene oxide derivative thereof. Exemplary acetylenic alcohol, acetylenic diol or ethylene oxide/propylene oxide derivatives that can be used as the at least one additive within an immersion fluid may be represented by the following formulas I through III:
wherein R1 and R4 are each independently a straight or a branched alkyl chain having from 3 to 10 carbon atoms; R2 and R3 are each independently a hydrogen atom or an alkyl chain having from 1 to 5 carbon atoms; and m, n, p, and q are each independently a number that ranges from 0 to 20. The at least one additive having the formula I, II or III are commercially available from Air Products and Chemicals, Inc. of Allentown, Pa., the assignee of the present invention, under the trade names SURFYNOL® and DYNOL®. In certain embodiments, the acetylenic diol portion of the molecule of formulas 1 or 11 is 2,4,5,9-tetramethyl-5-decyne-4,7-diol or 2,5,8,11-tetramethyl-6-dodecyne-5,8-diol. The at least one additives having the formulas I through III may be prepared in a number of ways including the methods described, for example, in U.S. Pat. No. 6,313,182 and EP 1115035A1, which are assigned to the assignee of the present invention and incorporated herein by reference in their entirety. - In formulas I and II, the alkylene oxide moieties represented by (OC2H4) are the (n+m) polymerized ethylene oxide (EO) molar units and the moieties represented by (OC3H6) are the (p+q) polymerized propylene oxide (PO) molar units. The value of (n+m) may range from 0 to 30, preferably from 1.3 to 15, and more preferably from 1.3 to 10. The value of (p+q) may range from 0 to 30, preferably from 1 to 10, and more preferably from 1 to 2.
-
- In each of the above formulas, R, R1 and R4 are each independently a straight, branched, or cyclic alkyl, fluoroalkyl, or perfluoroalkyl group having from 2 to 25, or from 3 to 10 carbon atoms; R2 and R3 are each independently a hydrogen atom, a straight, a branched or a cyclic alkyl group, fluoroalkyl group, or perfluoroalkyl group having from 1 to 10 or from 1 to 5 carbon atoms; R5 is a straight, a branched, or a cyclic alkyl, fluoroalkyl, or perfluoroalkyl group having from 1 to 10 carbon atoms; R6 is a straight, a branched, or a cyclic alkyl, fluoroalkyl, or perfluoroalkyl group having from 4 to 16 carbon atoms; R7, R8 and R9 are each independently a straight, a branched, or a cyclic alkyl, fluoroalkyl or perfluoroalkyl group having from 1 to 6 carbon atoms; R10 is independently H or a group represented by the formula
R11 is a straight, a branched, or a cyclic alkyl group having from 4 to 22 carbon atoms; W is a hydrogen atom or an alkynyl group; X and Y are either a hydrogen atom or a hydroxyl group; Z is either a halide atom, a hydroxyl group, an acetate group, or a carboxylate group; m, n, p, q are each independently a number ranging from 0 to 20; r and s are each independently 2 or 3; t is a number ranging from 0 to 2; i is a number ranging from 0 to 20; x is a number ranging from 1 to 6, and M is a monovalent metal ion or an ammonium ion. - The at least one additive may be an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups. Exemplary alkyl alcohols or polymeric alcohols include sugar alcohols such as sorbitol or polyvinyl alcohols. The at least one additive may be an alkyl alcohol, an alkyl ethoxylate, or a propylene oxide derivative thereof. Exemplary alkyl alcohols, alkyl ethoxylates, or propylene oxide derivatives that can be used as at least one additive within an immersion fluid may be represented by formulas IVa through IVe. A specific example of a Formula IVd additive is 2,4,7,9-tetramethyl-4,7-decane diol. Examples of Formula IVe additives include, but are not limited to, 3,5-dimethyl-1-hexyn-3-ol and 2,6-dimethyl-4-heptanol. The at least one additive may be an alkyl carboxylate or an alkyl acid ester. Exemplary alkyl carboxylates or alkyl acid esters that can be used as at least one additive within an immersion fluid may be represented by formulas Va through Vc. An example of a Formula Va additive includes, but is not limited to, diisopentyl tartrate. The at least one additive may be an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate. Exemplary alkyl amines or alkyl amine ethoxylates that can be used as at least one additive within an immersion fluid may be represented by formulas VIa through VIe. An example of a Formula VIa additive includes, but is not limited to, N,N′-bis(1,3-dimethylbutyl) ethylene diamine. The at least one additive may be an alkyl polyglycoside. An exemplary alkyl polyglycoside that can be used as at least one additive within an immersion fluid is represented by formula VII. The at least one additive may be a block oligomer or a polymer of ethylene and propylene oxide. Exemplary block oligomers or polymers of ethylene and propylene oxide that can be used as at least one additive within an immersion fluid may be represented by formulas VIIIa through VIIIc. The at least one additive may be an alkyl sulfate, an alkyl ethoxylate sulfate, an alkyl sulfonate, or an alkyl ethyoxylate sulfonate. The at least one additive may be an alkyl ammonium salt. The at least one additive may be a glycidal ether or a glucamine derivative with an alkyl amine, an alkyl diamine, an alkyl alcohol, or an acetylenic alcohol. Exemplary glycidal ether or glucamine derivatives that can be used as at least one additive within an immersion fluid may be represented by formulas IXa through IXb. An example of a Formula IXb additive includes, but is not limited to, an adduct of diethylenetriamine and n-butyl glycidyl ether. The at least one additive may be an alkyl urea or a dialkyl urea. The at least one additive may be a fluorinated or partially fluorinated acetylenic alcohol or diol and derivates thereof. Exemplary fluorinated or partially fluorinated acetylenic alcohol or diol and derivates thereof that can be used as at least one additive within an immersion fluid may be represented by formulas XIa through Xle. An example of a Formula XIa additive includes, but is not limited to, hexafluoropropanol acetylene.
- The at least one additive may be a fluorosurfactant provided that the carrier medium comprises at least 1% by weight or greater of an aqueous fluid. Exemplary fluorosurfactants include: straight, branched, or cyclic hydrofluorocarbons having 2 to 10 carbon atoms wherein there are more fluorine atoms than hydrogen atoms; F[CF(CF2)CF2]n—O—[CH2CH2O]m—H; F(CF2(CF3)CF2O]nCFHCF3 wherein n is a number ranging from 1 to 5; F[CF(CF3)CF2O]nCF2CF3 wherein n is a number ranging from 1 to 5; and HCF2(OCF2)n(OCF2CF2)m—O—CF2H wherein (n+m) is a number ranging from 1 to 8; a mixture of the ammonium salts of perfluorocarboxylic acids; fluoroaliphatic esters, [F(CF2CF2)1-7CH2CH2O]xP(O)(ONH4)y where x=1 or 2; y=2 or 1; and x+y=3 in various weight solution of isopropanol in water ranging from 25-70%; [F(CF2CF2)1-7CH2CH2O]xP(O)(OH)y where x=1 or 2; y=2 or 1; and x+y=3; F(CF2CF2)3-8CH2CH2503H in a 4.5% by weight solution of acetic acid in water; ammonium salt of perfluoro-octanoic acid.
- The at least one additive may be a salt having a refractive index equal to or greater than that of water and a specific absorbance less than 1 cm−1, or less than 0.5 cm−1, at an operating wavelength ranging from 140 to 248 nm. In embodiments wherein the operating wavelength is 193 nm, the refractive index is equal to or greater than 1.44. Exemplary salts include: sodium chloride (n=1.5443), calcium fluoride (n=1.4338), sulfate, sulfonate, phosphate, phosphonate, and phosphite salts and alkyl derivatives thereof.
- The at least one additive may be an electrolyte having a refractive index equal to or greater than that of water and a specific absorbance less than 1 cm−1, or less than 0.5 cm−1, at an operating wavelength ranging from 140 to 248 nm. In embodiments wherein the operating wavelength is 193 nm, the refractive index is equal to or greater than 1.44.
- Various other components may be optionally added to the immersion fluid. These components may include, but are not limited to, stabilizers, dissolving aids, colorants, wetting agents, antifoamers, buffering agents, and other additional surfactants. Generally, the amount of each of these additives would be about 0.0001 to 1 percent by weight, or about 0.0001 to 0.1 percent by weight, based upon the total weight of the immersion fluid. In embodiments where one or more additional surfactants are added to the immersion fluid, the surfactant may be any of the surfactants disclosed herein or provided in the reference McCutcheon's Emulsifiers and Detergents.
- In embodiments where the immersion fluid contains at least one additive, the immersion fluid may be prepared by mixing the at least one additive with at least one carrier medium which can be an aqueous and/or non-aqueous fluid and any additional components. In certain embodiments, one or more aqueous fluids such as ultra pure or HPLC water is combined with from about 10 ppm to the maximum solubility limit, or from about 10 ppm to about 10,000 ppm, of at least one additive to provide the immersion fluid. In an alternative embodiment, from about 10 ppm to the maximum solubility limit, or from about 10 ppm to about 10,000 ppm, of at least one additive is combined with one or more aqueous fluids until a homogeneous mixture is formed and is then combined with one or more non-aqueous fluids to provide the immersion fluid. In a still further embodiment, one or more non-aqueous fluids such as the fluids disclosed herein are combined with from about 10 ppm to the maximum solubility limit of at least one additive to provide the immersion fluid. In foregoing embodiments, the combining may be done at a temperature range of about 20 to 60° C. or from 40 to 60° C. to affect dissolution of the ingredients contained therein and provide a homogeneous mixture. The resulting immersion fluid may optionally be filtered to remove any undissolved particles that could potentially harm the substrate.
- In alternative embodiments, the immersion fluid may comprise the carrier medium, i.e., at least one aqueous fluid, at least one non-aqueous fluid, and mixtures thereof. In embodiments wherein the immersion fluid is a mixture of carrier media, the combining may be done at a temperature range of about 20 to 60° C. or from 40 to 60° C. to provide a homogeneous mixture.
- The immersion fluid is preferably used to treat at least a portion of the surface of a substrate that is coated with a photoresist or resist coating. Suitable substrates include, but are not limited to, materials such as gallium arsenide (“GaAs”), silicon, tantalum, copper, ceramics, aluminum/copper alloys, polyimides, and compositions containing silicon such as crystalline silicon, polysilicon, amorphous silicon, epitaxial silicon, silicon dioxide (“SiO2”), silicon nitride, doped silicon dioxide, and the like. Further exemplary substrates include silicon, aluminum, or polymeric resins. In certain embodiments, the immersion fluid contacts at least a portion of a substrate coated with a photoresist, or a resist-coated substrate and is substantially non-reactive to the resist coating. Exemplary resist coatings include, but are not limited to, novolac resin, polyvinyl phenol copolymer, or copolymers of p-hydroxystyrene and t-butyl acrylate.
- The immersion fluid is applied to at least a portion of a substrate having a photoresist coating applied thereto. The photoresist-coated substrate is then exposed to radiation through an optical device to provide a pattern that is imposed upon the photoresist coating. Examples of radiation sources that may be used include ultraviolet (uv) light, electron beam, x-ray, laser, lamp, or ion beams. In certain embodiments, the radiation source emits light at wavelengths at an operating wavelength ranging from 140 nm to 248 nm such as 193 nm and 157 nm. In some embodiments, a pre-bake or soft-bake step may be conducted prior to the exposure step. This pre-bake or soft bake step may be conducted, for example, at a temperature ranging from 90° C. to 150° C. for a time or from 30 to 120 seconds on a hot plate.
- Depending upon whether the photoresist coating is positive or negative, the radiation either increases or decreased its solubility in a subsequently applied alkaline developer solution such as a process solution containing tetramethylammonium hydroxide (TMAH), potassium hydroxide, sodium hydroxide, or other base. Further examples of developer solutions include those provided in U.S. Pat. Nos. 6,455,234; 6,268,115; 6,238,849; 6,127,101; and 6,120,978. In a positive photoresist coating, the areas masked from radiation remain after development while the exposed areas are dissolved away. In a negative photoresist coating, the opposite occurs. The immersion fluid of the present invention may be suitable to treat substrates having either positive or negative photoresist coatings. The patterned photoresist image may be developed by a variety of different means, including by not limited to quiescence, immersion, spray, or puddle development. After the patterned photoresist image is developed, the substrate is baked to harden the polymer contained within the photoresist. The bake step may be conducted, for example, at a temperature ranging from 70° C. to 150° C. for a time duration of from 30 to 120 seconds.
- A typical immersion lithography process uses an apparatus that has a servo motor driven wafer stage that supports and positions a resist-coated substrate or wafer underneath an optic device such as a lens. The optic device may also be a prism, a mirror or combinations thereof. The immersion fluid is dispensed onto at least a portion of resist-coated substrate through one or more nozzles to form a puddle. A radiation source that emits light at the operating wavelength then passes through lens and the puddle of immersion fluid prior to exposure to at least a portion of the resist-coated substrate.
- The immersion fluid is preferably applied to the photoresist-coated substrate as a prepared solution. In alternative embodiments, however, the immersion fluid can be prepared within the application stream just prior to or during contact with the substrate surface. For example, a certain quantity of one or more additives can be injected into a continuous stream of water and/or non-aqueous fluid medium or immersion fluid that optionally includes other components thereby forming the immersion fluid. In some embodiments of the present invention, a portion of the at least one additive may be added to the substrate after application of the immersion fluid. In still other embodiments of the present invention, the at least one additive can be also deposited upon or comprise the material of a high surface area device such as a cartridge or filter (which may or may not include other components). A stream of water and/or non-aqueous fluid then passes through the cartridge or filter thereby forming the immersion fluid. In still another embodiment of the present invention, the immersion fluid is prepared during the contacting step. In this connection, at least one additive is introduced via a dropper or other means to the surface of the substrate. Water and/or non-aqueous fluid medium is then introduced to the surface of the substrate and mixes with the at least one additive on the surface of the substrate thereby forming the immersion fluid.
- In an alternative embodiment of the invention, a concentrated composition comprising at least one additive is provided that may be diluted in water and/or non-aqueous fluids to provide the immersion fluid. A concentrated composition of the invention, or “concentrate” allows one to dilute the concentrate to the desired strength and pH. A concentrate also permits longer shelf life and easier shipping and storage of the product.
- A variety of means can be employed in contacting the immersion fluid with the substrate surface. The actual conditions of the contacting step (i.e., temperature, time, and the like) may vary over wide ranges and are generally dependent on a variety of factors such as, but not limited to, the nature and amount of residue on the surface of the substrate and the hydrophobicity or hydrophilicity of the substrate surface, etc. The contact step can be conducted in either a dynamic method such as, for example, a streamline process for applying the immersion fluid over the surface of the substrate or in a static method such as, for example, a puddle application. The immersion fluid may also be sprayed onto the surface of the substrate in a dynamic method such as in a continuous process or sprayed onto the surface and allowed to remain there in a static method. The duration of the contacting step, or time of contact of the immersion fluid to the substrate surface, can vary from a fraction of a second to hundreds of seconds. Preferably, the duration can range from 1 to 200 seconds, or from 1 to 150 seconds, or from 1 to 40 seconds. The temperature range for the contacting step can vary from 10 to 100° C., or from 10 to 40° C.
- The dynamic surface tension (DST) data was collected via the maximum bubble pressure method described in
Langmuir 1986, 2, pp. 428-432. The data was collected at bubble rates that range from 0.1 bubbles/second (b/s) to 20 b/s using the Kruss BP2 bubble pressure tensiometer manufactured by Kruss, Inc. of Charlotte, N.C. - The dynamic surface tension data provides information about the performance of the at least one additive at conditions from near-equilibrium (0.1 b/s) to relatively high surface creation rates (20 b/s). High bubble rates may correspond to a dynamic liquid injection process in an immersion lithography process. It is desirable that the dynamic surface tension by reduced below that of ultrapure water at high bubble rates (i.e., 70-72 dyne/cm at 20 b/s) to provide, inter alia, better wetting of the photoresist-coated substrate. Table I provides the DST of immersion fluids containing varying concentrations of the
alkyl diol additive TABLE I Dynamic Surface Tension DST DST DST DST DST Additives (dyne/cm) (dyne/cm) (dyne/cm) (dyne/cm) (dyne/cm) Conc. 0.1 b/s 1 b/s 6 b/s 15 b/s 20 b/s 0.01 wt % 48.7 54.0 61.9 67.8 69.0 0.03 wt % 41.8 43.7 47.5 53.6 56.5 0.05 wt % 38.5 39.6 41.8 45.6 47.6 - A quantity of 0.01% by weight or 100 ppm of the following additives: hydrophobically modified acetylenic diol or oxirane [(2-Ethyl Hexyl) Oxy]Methyl]-, Rx prod w/polyethylene glycol ether with 2,4,7,9-tetramethyl-5-decyne-4,7-diol (2:1); 2,4,7,9-tetramethyl-4,7-decane diol; ethoxylated 2,4,7,9-tetramethyl-4,7-decane diol; polysiloxane-polyester copolymer or TEGOWET™ manufactured by Goldschmidt Chemical of McDonald, Pa.; and an ethoxylated nonionic fluorosurfactant or ZONYL™ FSO with the formula: F(CF2CF2)1-7CH2CH2O(CH2CH2O)yH where y=0 to ca. 15 manufactured by Dupont of Wilmington, Del. were each combined with 100 mL of HPLC water manufactured by Aldrich Chemical to provide immersion fluids 1a through 1e, respectively. The additives were added to the HPLC water at ambient temperature while stirring to provide a homogeneous mixture. Dynamic contact angle measurements for each of the immersion fluids 1a through 1e and HPLC water as a comparative were obtained in accordance with the method disclosed in Example 1. The results of the dynamic contact angle measurements are provided in
FIG. 1 . - The wetting properties of immersion fluids containing an alkyl diol additive, 2,4,7,9-tetramethyl-4,7-decane diol, and ultra pure water as a comparison, was measured on the G10/DSA10 Kruss drop shape analyzer provided by Kruss USA of Charlotte, N.C. using the Sessile drop method. In this method, the wetting properties of a localized region on the surface of a photoresist-coated substrate are estimated by measuring the contact angle between the baseline of a droplet of aqueous developer solution and the tangent at the droplet base. A high-speed camera captured the spreading of the droplet at a speed of 2 frames per second for 2 minutes and the contact angle was measured. The photoresist is a typical acrylate type 193 nm resist.
- In general, lower contact angles indicate better wetting properties on resist surface. As Table II illustrates, the addition of additive improved the wetting on the resist surface.
TABLE II Comparison of Contact Angles of UPW vs. Immersion Fluid Contact Contact Contact Contact Angle Angle Angle Angle Fluid (0 sec) (5 sec) (10 sec) (30 sec) Ultra pure 62.9 62.9 62.5 61.6 water 0.02 wt % 54.1 53.5 52.8 51.8 additive - The absorbance of immersion fluids was measured with a UV spectrometer. The wavelength was scanned from 210 nm to 185 nm. As shown in
FIG. 1 , at 193 nm wavelength, the addition of 0.02 wt % of theadditive - A quartz crystal microbalance (QCM) was used to study changes in film thickness of unexposed 193 nm photoresist solutions. A CH Instruments model CHI 405 was used as a driver and high-resolution frequency counter. The instrument was fitted with a flow cell. The quartz crystals were made by International Crystal Manufacturing. The electrode was gold, 1000 Å thick and 0.201″ in diameter. The crystal resonance frequency was 7.995 MHz +/−10 Hz. The quartz crystals were spin-coated with a photoresist solution. The spin recipe was 1200 revolutions per minute (RPM) for 30 seconds and 3000 RPM for 10 seconds. The post apply bake (PAB) was 4 min at 125° C. The crystals were then exposed to UV light for the desired time. Each crystal was then mounted on a sensor probe and placed in the liquid flow cell. The instrument was controlled by a Dell PC using CHI software program. Frequency data was collected every 0.01 second. The flow of immersion fluid containing 0.01 weight percent of at least one additive was started 15 seconds after data acquisition started, to ensure that there were no glitches in the data acquisition process. The residence time through the flow cell was approximately 2 seconds. The experiment was stopped after 2 minutes.
FIG. 3 provides a comparison of the change in film thickness in nm for immersion fluids containing 0.01% by weight or 100 ppm of dimethyl-4-heptanol, hydrophobically modified acetylenic diol, ethoxylated nonionic fluorosurfactant in 100 ml of ultrapure water and ultrapure water. - Various immersion fluids containing 0.01% by weight of at least one additive in 200 ml ultrapure water were prepared at ambient temperature by mixing to form a homogeneous solution. Each immersion fluid was tested for foam dissipation using the Ross-Miles foam height test or ASTM D 1173-53. The results of this test for each immersion fluid is provided in Table III.
TABLE III Time to 0 Dynamic Initial Foam foam Foam Height Additive Height (cm) (seconds) (1) 2,4,7,9-tetramethyl-decane-4, 0 0 0 7-diol 2-Hydroxy-succinic acid dibutyl 1.5 0 0 ester Ethoxylated 2,4,7,9-tetramethyl- 1.3 30 0 5-decyn-4,7-diol Hydrophobically modified 1.7 >300 1.0 acetylenic diol Polysiloxane-polyester 0.4 3 0 copolymer
(1) Remaining foam after 1 minute measured in cm.
Claims (28)
1. An immersion fluid comprising: about 10 ppm to a maximum solubility limit of at least one additive selected from an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups; an alkyl ethoxylate or a propylene (PO) derivative thereof; an alkyl carboxylate or an alkyl acid ester; an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate; an acetylenic alcohol, an acetylenic diol or ethylene oxide/propylene oxide derivatives thereof; an alkyl polyglycoside; a block oligomer or a polymer of ethylene and propylene oxide; an alkyl sulfate, an alkyl ethoxylate sulfate, an alkyl sulfonate, or an alkyl ethyoxylate sulfonate; an alkyl ammonium salt; a glycidal ether or a glucamine derivative with an alkyl amine, an alkyl diamine, an alkyl alcohol, or an acetylenic alcohol; an alkyl urea or a dialkyl urea; a polysiloxane, a poly(dimethyl)siloxane, a polysiloxane polyester copolymer, or derivatives thereof; a fluorinated or partially fluorinated acetylenic alcohol, diol or derivates thereof; a fluorosurfactant; a salt; and an electrolyte; wherein the salt and the electrolyte have a specific absorbance <1 cm−1 at an operating wavelength ranging from 140 nm to 248 nm and a refractive index equal to or greater than water at the operating wavelength, provided that if the at least one additive is a fluorosurfactant then the immersion fluid comprises about 1% by weight or greater of an aqueous fluid.
2. The immersion fluid of claim 1 wherein the at least one additive is an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups.
3. The immersion fluid of claim 1 wherein the at least one additive is an acetylenic alcohol, an acetylenic diol, or a ethylene oxide/propylene oxide derivative thereof.
4. The immersion fluid of claim 1 wherein the at least one additive is an alkyl alcohol, an alkyl ethoxylate, or a propylene (PO) derivative thereof.
5. The immersion fluid of claim 1 wherein the at least one additive is an alkyl carboxylate or an alkyl acid ester.
6. The immersion fluid of claim 1 wherein the at least one additive is an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate.
7. The immersion fluid of claim 1 wherein the at least one additive is an alkyl polyglycoside.
8. The immersion fluid of claim 1 wherein the at least one additive is a block oligomer or a polymer of ethylene oxide and propylene oxide.
9. The immersion fluid of claim 1 wherein the at least one additive is an alkyl sulfate, an alkyl ethoxylate sulfate, an alkyl sulfonate, an alkyl ethoxylate, or an ethoxylate sulfonate.
10. The immersion fluid of claim 1 wherein the at least additive is an alkyl ammonium salt.
11. The immersion fluid of claim 1 wherein the at least one additive is a glycidal ether or a glucamine derivative with an alkyl amine, an alkyl diamine, an alkyl alcohol, or an acetylenic alcohol.
12. The immersion fluid of claim 1 wherein the at least one additive is an alkyl urea or a dialkyl urea.
13. The immersion fluid of claim 1 wherein the at least one additive is a fluorosurfactant.
14. The immersion fluid of claim 1 wherein the at least one additive is a salt.
15. The immersion fluid of claim 1 wherein the at least one additive is an electrolyte.
16. The immersion fluid of claim 1 wherein the at least one additive is a fluorinated or partially fluorinated acetylenic alcohol, diol or derivate thereof.
17. The immersion fluid of claim 1 wherein the at least one additive is a polysiloxane, a poly(dimethyl)siloxane, a polysiloxane polyester copolymer, or derivative thereof.
18. An immersion fluid having a transmission of 80% or greater at an operating wavelength ranging from 140 nm to 248 nm, the immersion fluid comprising:
at least one carrier medium selected from the group consisting of an aqueous fluid, a non-aqueous fluid, and mixtures thereof wherein the at least one carrier medium has a refractive index greater than or equal to water at the operating wavelength; and
about 10 ppm to a maximum solubility limit of at least one additive selected from an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups; an alkyl ethoxylate or a propylene (PO) derivative thereof; an alkyl carboxylate or an alkyl acid ester; an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate; an acetylenic alcohol, an acetylenic diol or ethylene oxide/propylene oxide derivatives thereof; an alkyl polyglycoside; a block oligomer or a polymer of ethylene and propylene oxide; an alkyl sulfate, an alkyl ethoxylate sulfate, an alkyl sulfonate, or an alkyl ethyoxylate sulfonate; an alkyl ammonium salt; a glycidal ether or a glucamine derivative with an alkyl amine, an alkyl diamine, an alkyl alcohol, or an acetylenic alcohol; an alkyl urea or a dialkyl urea; a polysiloxane, a poly(dimethyl)siloxane, a polysiloxane polyester copolymer, or derivatives thereof; a fluorinated or partially fluorinated acetylenic alcohol or diol or derivates thereof; a fluorosurfactant; a salt; and an electrolyte; wherein the salt and the electrolyte have a specific absorbance <1 cm−1 and a refractive index equal to or greater than water at the operating wavelength, provided that if the at least one additive is a fluorosurfactant then the immersion fluid comprises about 1% by weight or greater of an aqueous fluid.
19. The immersion fluid of claim 18 wherein the immersion fluid exhibits a dynamic surface tension of about 55 dynes/cm2 or less at 23° C. and 1 bubble/second according to the maximum-bubble-pressure method.
20. The immersion fluid of claim 18 wherein the immersion fluid exhibits a contact angle of about 500 or less at 30 seconds.
21. The immersion fluid of claim 18 wherein the immersion fluid exhibits a specific absorbance of 0.5 cm−1 or less at an operating wavelength of 193 nm.
22. The immersion fluid of claim 18 wherein the at least one carrier medium is an aqueous fluid.
23. The immersion fluid of claim 18 wherein the at least one carrier medium is a non-aqueous fluid.
24. The immersion fluid of claim 18 wherein the at least one carrier medium is the mixture of the aqueous and the non-aqueous fluids and wherein the non-aqueous fluid is water miscible.
25. The immersion fluid of claim 24 wherein the non-aqueous fluid is at least one selected from methanol, ethanol, isopropyl alcohol, glycerol, ethylene glycol and derivatives thereof, polyethylene glycol and derivatives thereof, and THF.
26. An immersion fluid having a transmission of 80% or greater at an operating wavelength ranging from 140 nm to 248 nm comprising: at least one carrier medium selected a non-aqueous fluid and a mixture of the non-aqueous fluid and an aqueous fluid wherein the at least one carrier medium has a refractive index greater than or equal to water at the operating wavelength and wherein if the at least one carrier medium is a mixture then the non-aqueous medium is water miscible.
27. The immersion fluid of claim 26 further comprising from 10 ppm to a maximum solubility limit of at least one additive selected from an alkyl alcohol or a polymeric alcohol having one or more hydroxyl groups; an alkyl ethoxylate or a propylene (PO) derivative thereof; an alkyl carboxylate or an alkyl acid ester; an alkyl amine having one or more amine groups including primary, secondary and tertiary amines or an alkyl amine ethoxylate; an acetylenic alcohol, an acetylenic diol or ethylene oxide/propylene oxide derivatives thereof; an alkyl polyglycoside; a block oligomer or a polymer of ethylene and propylene oxide; an alkyl sulfate, an alkyl ethoxylate sulfate, an alkyl sulfonate, or an alkyl ethyoxylate sulfonate; an alkyl ammonium salt; a glycidal ether or a glucamine derivative with an alkyl amine, an alkyl diamine, an alkyl alcohol, or an acetylenic alcohol; an alkyl urea or a dialkyl urea; a polysiloxane, a poly(dimethyl)siloxane, a polysiloxane polyester copolymer, or derivatives thereof; a fluorinated or partially fluorinated acetylenic alcohol, diol, or derivates thereof; a fluorosurfactant; a salt; and an electrolyte; wherein the salt and the electrolyte have a specific absorbance <1 cm−1 and a refractive index equal to or greater than water at the operating wavelength, provided that if the at least one additive is a fluorosurfactant then the immersion fluid comprises about 1% by weight or greater of an aqueous fluid.
28. The immersion fluid of claim 26 wherein the non-aqueous fluid is at least one selected from citric acid, bicyclohexyl, glycerol, and cis-2-methylcyclohexanol.
Priority Applications (11)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/764,227 US20050161644A1 (en) | 2004-01-23 | 2004-01-23 | Immersion lithography fluids |
| US11/030,132 US7879531B2 (en) | 2004-01-23 | 2005-01-07 | Immersion lithography fluids |
| TW094101464A TWI259319B (en) | 2004-01-23 | 2005-01-18 | Immersion lithography fluids |
| EP05000989.3A EP1557721B1 (en) | 2004-01-23 | 2005-01-19 | Use of fluids for immersion lithography and pattern forming method |
| EP14159576.9A EP2762976A1 (en) | 2004-01-23 | 2005-01-19 | Use of immersion liquids |
| CN2008101780274A CN101430509B (en) | 2004-01-23 | 2005-01-21 | Immersion lithography fluids |
| CN200510054221A CN100582935C (en) | 2004-01-23 | 2005-01-21 | Immersion lithographic fluids |
| KR1020050006037A KR20050076772A (en) | 2004-01-23 | 2005-01-22 | Immersion lithography fluids |
| JP2005015928A JP4828833B2 (en) | 2004-01-23 | 2005-01-24 | Immersion lithography fluid |
| US11/754,725 US8007986B2 (en) | 2004-01-23 | 2007-05-29 | Immersion lithography fluids |
| KR1020080070623A KR100967372B1 (en) | 2004-01-23 | 2008-07-21 | Immersion lithography fluid |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US10/764,227 US20050161644A1 (en) | 2004-01-23 | 2004-01-23 | Immersion lithography fluids |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/030,132 Continuation-In-Part US7879531B2 (en) | 2004-01-23 | 2005-01-07 | Immersion lithography fluids |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| US20050161644A1 true US20050161644A1 (en) | 2005-07-28 |
Family
ID=34795247
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US10/764,227 Abandoned US20050161644A1 (en) | 2004-01-23 | 2004-01-23 | Immersion lithography fluids |
| US11/030,132 Expired - Fee Related US7879531B2 (en) | 2004-01-23 | 2005-01-07 | Immersion lithography fluids |
Family Applications After (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| US11/030,132 Expired - Fee Related US7879531B2 (en) | 2004-01-23 | 2005-01-07 | Immersion lithography fluids |
Country Status (4)
| Country | Link |
|---|---|
| US (2) | US20050161644A1 (en) |
| EP (1) | EP2762976A1 (en) |
| KR (1) | KR100967372B1 (en) |
| CN (1) | CN101430509B (en) |
Cited By (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20050205108A1 (en) * | 2004-03-16 | 2005-09-22 | Taiwan Semiconductor Manufacturing Co., Ltd. | Method and system for immersion lithography lens cleaning |
| US20050233254A1 (en) * | 2004-04-16 | 2005-10-20 | Shin-Etsu Chemical Co., Ltd. | Patterning process and resist overcoat material |
| US20050231695A1 (en) * | 2004-04-15 | 2005-10-20 | Taiwan Semiconductor Manufacturing Company, Ltd. | Method and system for immersion lithography using high PH immersion fluid |
| US20050255413A1 (en) * | 2004-05-17 | 2005-11-17 | Matsushita Electric Industrial Co., Ltd. | Semiconductor manufacturing apparatus and pattern formation method |
| US20050270505A1 (en) * | 2004-02-03 | 2005-12-08 | Smith Bruce W | Method of photolithography using a fluid and a system thereof |
| US20050277068A1 (en) * | 2004-06-09 | 2005-12-15 | Matsushita Electric Industrial Co., Ltd. | Semiconductor manufacturing apparatus and pattern formation method |
| US20050286031A1 (en) * | 2004-06-01 | 2005-12-29 | French Roger H | Use of highly purified hydrocarbons in vacuum ultraviolet applications |
| US20060014105A1 (en) * | 2004-07-13 | 2006-01-19 | Matsushita Electric Industrial Co., Ltd. | Immersion exposure liquid and pattern formation method |
| US20060024613A1 (en) * | 2004-07-30 | 2006-02-02 | Asahi Glass Company, Limited | Composition for photoresist protective film, photoresist protective film and photoresist pattern formation process |
| US20060028628A1 (en) * | 2004-08-03 | 2006-02-09 | Taiwan Semiconductor Manufacturing Co., Ltd. | Lens cleaning module |
| US20060066962A1 (en) * | 2003-12-15 | 2006-03-30 | Carl Zeiss Smt Ag | Arrangement of optical elements in a microlithographic projection exposure apparatus |
| US20060072088A1 (en) * | 2004-10-05 | 2006-04-06 | Asml Netherlands B.V. | Lithographic apparatus and device manufacturing method |
| US20060244938A1 (en) * | 2004-05-04 | 2006-11-02 | Karl-Heinz Schuster | Microlitographic projection exposure apparatus and immersion liquid therefore |
| US20060257553A1 (en) * | 2003-10-08 | 2006-11-16 | Zao Nikon Co., Ltd. | Substrate conveyance device and substrate conveyance method, exposure apparatus and exposure method, device manufacturing method |
| KR100682152B1 (en) | 2004-07-02 | 2007-02-12 | 주식회사 하이닉스반도체 | Liquid composition for immersion lithography and lithography method using same |
| US20070105050A1 (en) * | 2003-11-05 | 2007-05-10 | Dsm Ip Assets B.V. | Method and apparatus for producing microchips |
| US20070109517A1 (en) * | 2004-02-03 | 2007-05-17 | Nikon Corporation | Exposure apparatus and device manufacturing method |
| US20070117232A1 (en) * | 2005-11-07 | 2007-05-24 | Roel Gronheid | Method of studying interaction between immersion fluid and substrate |
| US20070117040A1 (en) * | 2005-11-21 | 2007-05-24 | International Business Machines Corporation | Water castable-water strippable top coats for 193 nm immersion lithography |
| US20070182896A1 (en) * | 2005-11-23 | 2007-08-09 | Adelman Douglas J | Apparatus for recycling alkane immersion liquids and methods of employment |
| US20070229957A1 (en) * | 2006-04-03 | 2007-10-04 | Nikon Corporation | Incidence surfaces and optical windows that are solvophobic to immersion liquids |
| US20070285637A1 (en) * | 2004-02-13 | 2007-12-13 | Carl Zeiss Smt Ag | Imaging System for a Microlithographical Projection Light System |
| US20080063984A1 (en) * | 2002-08-12 | 2008-03-13 | Air Products And Chemicals, Inc. | Process Solutions Containing Surfactants |
| US20080084549A1 (en) * | 2006-10-09 | 2008-04-10 | Rottmayer Robert E | High refractive index media for immersion lithography and method of immersion lithography using same |
| US20080176047A1 (en) * | 2004-05-22 | 2008-07-24 | Hynix Semiconductor Inc. | Liquid Composition for Immersion Lithography and Lithography Method Using the Same |
| WO2007140012A3 (en) * | 2006-05-26 | 2008-09-18 | Massachusetts Inst Technology | Immersion fluids for lithography |
| US20080304033A1 (en) * | 2004-02-13 | 2008-12-11 | Carl Zeiss Smt Ag | Projection objective for a microlithographic projection exposure apparatus |
| US20090227111A1 (en) * | 2004-02-25 | 2009-09-10 | Panasonic Corporation | Barrier film material and pattern formation method using the same |
| US20090294693A1 (en) * | 2008-06-02 | 2009-12-03 | Lumatech Gmbh | Apparatus and method for optical examination of documents |
| US20100129526A1 (en) * | 2004-12-06 | 2010-05-27 | Sokudo Co., Ltd. | Substrate processing apparatus |
| US20100245789A1 (en) * | 2006-01-18 | 2010-09-30 | Canon Kabushiki Kaisha | Exposure apparatus |
| US20110229824A1 (en) * | 2004-02-09 | 2011-09-22 | Yoshihiko Okamoto | method of manufacturing a semiconductor integrated circuit device |
| US8859188B2 (en) | 2005-02-10 | 2014-10-14 | Asml Netherlands B.V. | Immersion liquid, exposure apparatus, and exposure process |
| CN104297194A (en) * | 2014-10-17 | 2015-01-21 | 浙江理工大学 | Method and device for detecting average EO number of alkylphenol ethoxylates |
| CN104297411A (en) * | 2014-10-17 | 2015-01-21 | 浙江理工大学 | Method for determining average EO number of alkylphenol polyoxyethylene in mixture |
| US9703199B2 (en) | 2004-12-06 | 2017-07-11 | Screen Semiconductor Solutions Co., Ltd. | Substrate processing apparatus |
Families Citing this family (22)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006523383A (en) | 2003-03-04 | 2006-10-12 | ピクセリジェント・テクノロジーズ・エルエルシー | Application of nano-sized semiconductor particles for photolithography |
| US8993221B2 (en) | 2012-02-10 | 2015-03-31 | Pixelligent Technologies, Llc | Block co-polymer photoresist |
| TWI259319B (en) * | 2004-01-23 | 2006-08-01 | Air Prod & Chem | Immersion lithography fluids |
| US20050260528A1 (en) * | 2004-05-22 | 2005-11-24 | Hynix Semiconductor Inc. | Liquid composition for immersion lithography and lithography method using the same |
| JP4488890B2 (en) * | 2004-12-27 | 2010-06-23 | 株式会社東芝 | Resist pattern forming method and semiconductor device manufacturing method |
| KR20070095399A (en) * | 2005-01-25 | 2007-09-28 | 제이에스알 가부시끼가이샤 | Immersion type exposure system, recycling method and supply method of liquid for immersion type exposure |
| WO2006115268A1 (en) * | 2005-04-26 | 2006-11-02 | Mitsui Chemicals, Inc. | Liquid for immersion exposure, method for purifying liquid for immersion exposure, and immersion exposure method |
| EP1720072B1 (en) * | 2005-05-01 | 2019-06-05 | Rohm and Haas Electronic Materials, L.L.C. | Compositons and processes for immersion lithography |
| US7435528B2 (en) * | 2005-06-09 | 2008-10-14 | E.I. Du Pont De Nemours And Company | Processes and devices using polycyclic fluoroalkanes in vacuum and deep ultraviolet applications |
| US20090130604A1 (en) * | 2005-08-29 | 2009-05-21 | Mitsui Chemicals, Inc. | Solution for immersion exposure and immersion exposure method |
| WO2008008275A2 (en) * | 2006-07-10 | 2008-01-17 | Pixelligent Technologies Llc | Resists for photolithography |
| US7586103B2 (en) | 2006-09-09 | 2009-09-08 | E. I. Du Pont De Nemours And Company | High refractive index fluids for immersion lithography |
| US7771919B2 (en) * | 2006-09-09 | 2010-08-10 | E. I. Du Pont De Nemours And Company | High refractive index fluids for immersion lithography |
| US7671247B2 (en) * | 2006-09-13 | 2010-03-02 | E.I. Du Pont De Nemours And Company | Methods for purifying alkane liquids |
| JP2008089952A (en) * | 2006-10-02 | 2008-04-17 | Matsushita Electric Ind Co Ltd | Chemically amplified resist material and pattern forming method using the same |
| US8045135B2 (en) | 2006-11-22 | 2011-10-25 | Asml Netherlands B.V. | Lithographic apparatus with a fluid combining unit and related device manufacturing method |
| US20090299113A1 (en) * | 2008-05-28 | 2009-12-03 | E. I. Dupont De Nemours And Company | Process for preparing alkane liquids for use in immersion lithography |
| EP2479616A1 (en) * | 2011-01-25 | 2012-07-25 | Basf Se | The use of surfactants having at least three short-chain perfluorinated groups Rf for manufacturing integrated circuits having patterns with line-space dimensions below 50 nm |
| KR102004148B1 (en) * | 2011-01-25 | 2019-07-26 | 바스프 에스이 | Use of surfactants having at least three short-chain perfluorinated groups for manufacturing integrated circuits having patterns with line-space dimensions below 50nm |
| US11292002B2 (en) | 2016-04-08 | 2022-04-05 | The Governing Council Of The University Of Toronto | Surfactant additives for digital microfluidic devices for high protein content droplets transport |
| WO2020236518A1 (en) * | 2019-05-22 | 2020-11-26 | The Board Of Trustees Of The University Of Illinois | Photoresist-free deposition and patterning with vacuum ultraviolet lamps |
| CN112987510A (en) * | 2021-03-09 | 2021-06-18 | 史耿共我 | Interlayer immersion type photoetching method using negative black photoresist |
Citations (14)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4275243A (en) * | 1978-09-29 | 1981-06-23 | Toyo Soda Manufacturing Co., Ltd. | Process for producing hydroxybutyraldehyde |
| US4770802A (en) * | 1986-02-04 | 1988-09-13 | Nippon Oil Co., Ltd. | Lubricating oil compositions |
| US4966977A (en) * | 1988-03-17 | 1990-10-30 | Mitsui Toatsu Chemicals, Incorporated | Process for the preparation of indigo compounds |
| US20020163629A1 (en) * | 2001-05-07 | 2002-11-07 | Michael Switkes | Methods and apparatus employing an index matching medium |
| US20040144399A1 (en) * | 2002-09-24 | 2004-07-29 | Mcdermott Wayne Thomas | Processing of semiconductor components with dense processing fluids and ultrasonic energy |
| US20050029490A1 (en) * | 2003-08-05 | 2005-02-10 | Hoshang Subawalla | Processing of substrates with dense fluids comprising acetylenic diols and/or alcohols |
| US20050036183A1 (en) * | 2003-08-11 | 2005-02-17 | Yee-Chia Yeo | Immersion fluid for immersion Lithography, and method of performing immersion lithography |
| US20050074686A1 (en) * | 2003-10-03 | 2005-04-07 | Solvay Solexis S.P.A. | Perfluoropolyethers |
| US20050094116A1 (en) * | 2003-08-29 | 2005-05-05 | Asml Netherlands B.V. | Gradient immersion lithography |
| US20050164522A1 (en) * | 2003-03-24 | 2005-07-28 | Kunz Roderick R. | Optical fluids, and systems and methods of making and using the same |
| US20050164502A1 (en) * | 2004-01-22 | 2005-07-28 | Hai Deng | Immersion liquids for immersion lithography |
| US20050186513A1 (en) * | 2004-02-24 | 2005-08-25 | Martin Letz | Liquid and method for liquid immersion lithography |
| US20050186514A1 (en) * | 2004-02-20 | 2005-08-25 | French Roger H. | Use of Perfluoro-n-alkanes in vacuum ultraviolet applications |
| US7029832B2 (en) * | 2003-03-11 | 2006-04-18 | Samsung Electronics Co., Ltd. | Immersion lithography methods using carbon dioxide |
Family Cites Families (38)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| SU948994A1 (en) | 1980-01-29 | 1982-08-07 | Институт теплофизики СО АН СССР | Liquid for optical investigations |
| JPS57153433A (en) | 1981-03-18 | 1982-09-22 | Hitachi Ltd | Manufacturing device for semiconductor |
| EP0063684A1 (en) | 1981-04-27 | 1982-11-03 | F. HOFFMANN-LA ROCHE & CO. Aktiengesellschaft | Immersion oil |
| SU1204623A1 (en) | 1983-11-16 | 1986-01-15 | Новосибирский государственный университет | Liquid for optical investigations |
| US4617490A (en) * | 1984-12-07 | 1986-10-14 | North American Philips Corporation | Cathode ray tube device with improved color filtering system |
| JPH04305915A (en) | 1991-04-02 | 1992-10-28 | Nikon Corp | Adhesion type exposure device |
| JPH06167653A (en) | 1992-11-27 | 1994-06-14 | Olympus Optical Co Ltd | Immersion oil |
| RU2051940C1 (en) | 1993-06-22 | 1996-01-10 | Производственное объединение "Новосибирский приборостроительный завод" | Immersion liquid |
| JP3747566B2 (en) | 1997-04-23 | 2006-02-22 | 株式会社ニコン | Immersion exposure equipment |
| JP3817836B2 (en) | 1997-06-10 | 2006-09-06 | 株式会社ニコン | EXPOSURE APPARATUS, ITS MANUFACTURING METHOD, EXPOSURE METHOD, AND DEVICE MANUFACTURING METHOD |
| US5900354A (en) | 1997-07-03 | 1999-05-04 | Batchelder; John Samuel | Method for optical inspection and lithography |
| JPH11269317A (en) * | 1998-01-23 | 1999-10-05 | Nikon Corp | Immersion oil |
| AU2747999A (en) | 1998-03-26 | 1999-10-18 | Nikon Corporation | Projection exposure method and system |
| US6313182B1 (en) | 1999-05-04 | 2001-11-06 | Air Products And Chemicals, Inc. | Acetylenic diol ethylene oxide/propylene oxide adducts and processes for their manufacture |
| US6455234B1 (en) | 1999-05-04 | 2002-09-24 | Air Products And Chemicals, Inc. | Acetylenic diol ethylene oxide/propylene oxide adducts and their use in photoresist developers |
| US6238849B1 (en) | 1999-06-21 | 2001-05-29 | Air Products And Chemicals, Inc. | Cyclic ureas in photoresist developers |
| US6127101A (en) | 1999-10-12 | 2000-10-03 | Air Products And Chemicals, Inc. | Alkylated aminoalkylpiperazine surfactants and their use in photoresist developers |
| JP2001215690A (en) | 2000-01-04 | 2001-08-10 | Air Prod And Chem Inc | Acetylenic diol ethylene oxide / propylene oxide adducts and their use in developers |
| US6120978A (en) | 2000-01-06 | 2000-09-19 | Air Products And Chemicals, Inc. | Use of N,N-dialkyl ureas in photoresist developers |
| US6268115B1 (en) | 2000-01-06 | 2001-07-31 | Air Products And Chemicals, Inc. | Use of alkylated polyamines in photoresist developers |
| JP2002053839A (en) | 2000-08-08 | 2002-02-19 | Nikon Corp | High refractive index liquid |
| JP2002144752A (en) | 2000-11-17 | 2002-05-22 | Chugai Photo Chemical Co Ltd | A fountain solution composition for lithographic printing |
| US6890619B2 (en) * | 2001-11-13 | 2005-05-10 | Agilent Technologies, Inc. | Optical systems and refractive index-matching compositions |
| TW200304044A (en) | 2002-03-06 | 2003-09-16 | Du Pont | Radiation durable organic compounds with high transparency in the vacuum ultraviolet, and method for preparing |
| DE10210899A1 (en) | 2002-03-08 | 2003-09-18 | Zeiss Carl Smt Ag | Refractive projection lens for immersion lithography |
| KR20040058966A (en) | 2002-12-27 | 2004-07-05 | 주식회사 하이닉스반도체 | Immersion lithography method |
| JP2005101498A (en) | 2003-03-04 | 2005-04-14 | Tokyo Ohka Kogyo Co Ltd | Immersion liquid for liquid immersion lithography process, and resist-pattern forming method using immersion liquid |
| JP4376574B2 (en) | 2003-08-25 | 2009-12-02 | パナソニック株式会社 | Pattern formation method |
| JP2007525824A (en) | 2003-11-05 | 2007-09-06 | ディーエスエム アイピー アセッツ ビー.ブイ. | Method and apparatus for manufacturing a microchip |
| US7033523B2 (en) | 2003-11-14 | 2006-04-25 | Photintech Inc. | Composite liquid crystalline mixture |
| US7125652B2 (en) * | 2003-12-03 | 2006-10-24 | Advanced Micro Devices, Inc. | Immersion lithographic process using a conforming immersion medium |
| KR20070039869A (en) | 2004-02-03 | 2007-04-13 | 브루스 더블유. 스미스 | Photolithography Method Using Solution and Related Systems |
| EP1564592A1 (en) | 2004-02-17 | 2005-08-17 | Freescale Semiconductor, Inc. | Protection of resist for immersion lithography technique |
| US7465667B2 (en) | 2004-03-08 | 2008-12-16 | E.I. Du Pont De Nemours And Company | Highly purified liquid perfluoro-n-alkanes and method for preparing |
| EP1748469A4 (en) * | 2004-05-21 | 2012-04-04 | Jsr Corp | Liquid for immersion exposure and immersion exposure method |
| EP1751624A1 (en) | 2004-06-01 | 2007-02-14 | E.I. Dupont De Nemours And Company | Ultraviolet-transparent alkanes and processes using same in vacuum and deep ultraviolet applications |
| US20090213342A1 (en) | 2004-10-22 | 2009-08-27 | Carl Zeiss Smt Ag | Projection exposure apparatus for microlithography |
| US7317507B2 (en) | 2005-05-03 | 2008-01-08 | Asml Netherlands B.V. | Lithographic apparatus and device manufacturing method |
-
2004
- 2004-01-23 US US10/764,227 patent/US20050161644A1/en not_active Abandoned
-
2005
- 2005-01-07 US US11/030,132 patent/US7879531B2/en not_active Expired - Fee Related
- 2005-01-19 EP EP14159576.9A patent/EP2762976A1/en not_active Withdrawn
- 2005-01-21 CN CN2008101780274A patent/CN101430509B/en not_active Expired - Fee Related
-
2008
- 2008-07-21 KR KR1020080070623A patent/KR100967372B1/en not_active Expired - Fee Related
Patent Citations (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4275243A (en) * | 1978-09-29 | 1981-06-23 | Toyo Soda Manufacturing Co., Ltd. | Process for producing hydroxybutyraldehyde |
| US4770802A (en) * | 1986-02-04 | 1988-09-13 | Nippon Oil Co., Ltd. | Lubricating oil compositions |
| US4966977A (en) * | 1988-03-17 | 1990-10-30 | Mitsui Toatsu Chemicals, Incorporated | Process for the preparation of indigo compounds |
| US20020163629A1 (en) * | 2001-05-07 | 2002-11-07 | Michael Switkes | Methods and apparatus employing an index matching medium |
| US20040144399A1 (en) * | 2002-09-24 | 2004-07-29 | Mcdermott Wayne Thomas | Processing of semiconductor components with dense processing fluids and ultrasonic energy |
| US7029832B2 (en) * | 2003-03-11 | 2006-04-18 | Samsung Electronics Co., Ltd. | Immersion lithography methods using carbon dioxide |
| US20050164522A1 (en) * | 2003-03-24 | 2005-07-28 | Kunz Roderick R. | Optical fluids, and systems and methods of making and using the same |
| US20050029492A1 (en) * | 2003-08-05 | 2005-02-10 | Hoshang Subawalla | Processing of semiconductor substrates with dense fluids comprising acetylenic diols and/or alcohols |
| US20050029490A1 (en) * | 2003-08-05 | 2005-02-10 | Hoshang Subawalla | Processing of substrates with dense fluids comprising acetylenic diols and/or alcohols |
| US20050036183A1 (en) * | 2003-08-11 | 2005-02-17 | Yee-Chia Yeo | Immersion fluid for immersion Lithography, and method of performing immersion lithography |
| US20050094116A1 (en) * | 2003-08-29 | 2005-05-05 | Asml Netherlands B.V. | Gradient immersion lithography |
| US20050074686A1 (en) * | 2003-10-03 | 2005-04-07 | Solvay Solexis S.P.A. | Perfluoropolyethers |
| US20050164502A1 (en) * | 2004-01-22 | 2005-07-28 | Hai Deng | Immersion liquids for immersion lithography |
| US20050186514A1 (en) * | 2004-02-20 | 2005-08-25 | French Roger H. | Use of Perfluoro-n-alkanes in vacuum ultraviolet applications |
| US20050186513A1 (en) * | 2004-02-24 | 2005-08-25 | Martin Letz | Liquid and method for liquid immersion lithography |
Cited By (74)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20080063984A1 (en) * | 2002-08-12 | 2008-03-13 | Air Products And Chemicals, Inc. | Process Solutions Containing Surfactants |
| US7995186B2 (en) | 2003-10-08 | 2011-08-09 | Zao Nikon Co., Ltd. | Substrate conveyance device and substrate conveyance method, exposure apparatus and exposure method, device manufacturing method |
| US20070296940A1 (en) * | 2003-10-08 | 2007-12-27 | Zao Nikon Co., Ltd. | Substrate conveyance device and substrate conveyance method, exposure apparatus and exposure method, device manufacturing method |
| US9110381B2 (en) | 2003-10-08 | 2015-08-18 | Nikon Corporation | Substrate conveyance device and substrate conveyance method, exposure apparatus and exposure method, device manufacturing method |
| US9097986B2 (en) | 2003-10-08 | 2015-08-04 | Nikon Corporation | Substrate conveyance device and substrate conveyance method, exposure apparatus and exposure method, device manufacturing method |
| US8345216B2 (en) | 2003-10-08 | 2013-01-01 | Nikon Corporation | Substrate conveyance device and substrate conveyance method, exposure apparatus and exposure method, device manufacturing method |
| US8107055B2 (en) | 2003-10-08 | 2012-01-31 | Zao Nikon Co., Ltd. | Substrate conveyance device and substrate conveyance method, exposure apparatus and exposure method, device manufacturing method |
| US20060257553A1 (en) * | 2003-10-08 | 2006-11-16 | Zao Nikon Co., Ltd. | Substrate conveyance device and substrate conveyance method, exposure apparatus and exposure method, device manufacturing method |
| US20070105050A1 (en) * | 2003-11-05 | 2007-05-10 | Dsm Ip Assets B.V. | Method and apparatus for producing microchips |
| US7385764B2 (en) | 2003-12-15 | 2008-06-10 | Carl Zeiss Smt Ag | Objectives as a microlithography projection objective with at least one liquid lens |
| US20080030869A1 (en) * | 2003-12-15 | 2008-02-07 | Carl Zeiss Smt Ag | Objectives as a microlithography projection objective with at least one liquid lens |
| US7474469B2 (en) | 2003-12-15 | 2009-01-06 | Carl Zeiss Smt Ag | Arrangement of optical elements in a microlithographic projection exposure apparatus |
| US20060066962A1 (en) * | 2003-12-15 | 2006-03-30 | Carl Zeiss Smt Ag | Arrangement of optical elements in a microlithographic projection exposure apparatus |
| US7428105B2 (en) | 2003-12-15 | 2008-09-23 | Carl Zeiss Smt Ag | Objectives as a microlithography projection objective with at least one liquid lens |
| US8767168B2 (en) | 2004-02-03 | 2014-07-01 | Nikon Corporation | Immersion exposure apparatus and method that detects residual liquid on substrate held by substrate table after exposure |
| US20050270505A1 (en) * | 2004-02-03 | 2005-12-08 | Smith Bruce W | Method of photolithography using a fluid and a system thereof |
| US9041906B2 (en) | 2004-02-03 | 2015-05-26 | Nikon Corporation | Immersion exposure apparatus and method that detects liquid adhered to rear surface of substrate |
| US7990516B2 (en) | 2004-02-03 | 2011-08-02 | Nikon Corporation | Immersion exposure apparatus and device manufacturing method with liquid detection apparatus |
| US7990517B2 (en) | 2004-02-03 | 2011-08-02 | Nikon Corporation | Immersion exposure apparatus and device manufacturing method with residual liquid detector |
| US8852850B2 (en) * | 2004-02-03 | 2014-10-07 | Rochester Institute Of Technology | Method of photolithography using a fluid and a system thereof |
| US10151983B2 (en) | 2004-02-03 | 2018-12-11 | Nikon Corporation | Exposure apparatus and device manufacturing method |
| US20070109517A1 (en) * | 2004-02-03 | 2007-05-17 | Nikon Corporation | Exposure apparatus and device manufacturing method |
| US8488101B2 (en) | 2004-02-03 | 2013-07-16 | Nikon Corporation | Immersion exposure apparatus and method that detects residual liquid on substrate held by substrate table on way from exposure position to unload position |
| US20110229824A1 (en) * | 2004-02-09 | 2011-09-22 | Yoshihiko Okamoto | method of manufacturing a semiconductor integrated circuit device |
| US20070285637A1 (en) * | 2004-02-13 | 2007-12-13 | Carl Zeiss Smt Ag | Imaging System for a Microlithographical Projection Light System |
| US7719658B2 (en) | 2004-02-13 | 2010-05-18 | Carl Zeiss Smt Ag | Imaging system for a microlithographical projection light system |
| US20110228246A1 (en) * | 2004-02-13 | 2011-09-22 | Carl Zeiss Smt Ag | Projection objective for a microlithographic projection exposure apparatus |
| US20080304033A1 (en) * | 2004-02-13 | 2008-12-11 | Carl Zeiss Smt Ag | Projection objective for a microlithographic projection exposure apparatus |
| US20090227111A1 (en) * | 2004-02-25 | 2009-09-10 | Panasonic Corporation | Barrier film material and pattern formation method using the same |
| US20050205108A1 (en) * | 2004-03-16 | 2005-09-22 | Taiwan Semiconductor Manufacturing Co., Ltd. | Method and system for immersion lithography lens cleaning |
| US20050231695A1 (en) * | 2004-04-15 | 2005-10-20 | Taiwan Semiconductor Manufacturing Company, Ltd. | Method and system for immersion lithography using high PH immersion fluid |
| US7455952B2 (en) * | 2004-04-16 | 2008-11-25 | Shin-Etsu Chemical Co., Ltd. | Patterning process and resist overcoat material |
| US20050233254A1 (en) * | 2004-04-16 | 2005-10-20 | Shin-Etsu Chemical Co., Ltd. | Patterning process and resist overcoat material |
| US20060244938A1 (en) * | 2004-05-04 | 2006-11-02 | Karl-Heinz Schuster | Microlitographic projection exposure apparatus and immersion liquid therefore |
| US20080304032A1 (en) * | 2004-05-04 | 2008-12-11 | Carl Zeiss Smt Ag | Microlitographic projection exposure apparatus and immersion liquid therefore |
| US20050255413A1 (en) * | 2004-05-17 | 2005-11-17 | Matsushita Electric Industrial Co., Ltd. | Semiconductor manufacturing apparatus and pattern formation method |
| US20080176047A1 (en) * | 2004-05-22 | 2008-07-24 | Hynix Semiconductor Inc. | Liquid Composition for Immersion Lithography and Lithography Method Using the Same |
| US20050286031A1 (en) * | 2004-06-01 | 2005-12-29 | French Roger H | Use of highly purified hydrocarbons in vacuum ultraviolet applications |
| US7589242B2 (en) * | 2004-06-01 | 2009-09-15 | E. I. Du Pont De Nemours And Company | Use of highly purified hydrocarbons in vacuum ultraviolet applications |
| US20050277068A1 (en) * | 2004-06-09 | 2005-12-15 | Matsushita Electric Industrial Co., Ltd. | Semiconductor manufacturing apparatus and pattern formation method |
| US7771918B2 (en) * | 2004-06-09 | 2010-08-10 | Panasonic Corporation | Semiconductor manufacturing apparatus and pattern formation method |
| US20100265477A1 (en) * | 2004-06-09 | 2010-10-21 | Panasonic Coporation | Semiconductor manufacturing apparatus and pattern formation method |
| KR100682152B1 (en) | 2004-07-02 | 2007-02-12 | 주식회사 하이닉스반도체 | Liquid composition for immersion lithography and lithography method using same |
| US20060014105A1 (en) * | 2004-07-13 | 2006-01-19 | Matsushita Electric Industrial Co., Ltd. | Immersion exposure liquid and pattern formation method |
| US20060024613A1 (en) * | 2004-07-30 | 2006-02-02 | Asahi Glass Company, Limited | Composition for photoresist protective film, photoresist protective film and photoresist pattern formation process |
| US8054444B2 (en) * | 2004-08-03 | 2011-11-08 | Taiwan Semiconductor Manufacturing Company, Ltd. | Lens cleaning module for immersion lithography apparatus |
| US20060028628A1 (en) * | 2004-08-03 | 2006-02-09 | Taiwan Semiconductor Manufacturing Co., Ltd. | Lens cleaning module |
| US20110134402A1 (en) * | 2004-10-05 | 2011-06-09 | Asml Netherlands B.V. | Lithographic apparatus and device manufacturing method |
| US20060072088A1 (en) * | 2004-10-05 | 2006-04-06 | Asml Netherlands B.V. | Lithographic apparatus and device manufacturing method |
| US7894040B2 (en) * | 2004-10-05 | 2011-02-22 | Asml Netherlands B.V. | Lithographic apparatus and device manufacturing method |
| US8027026B2 (en) | 2004-10-05 | 2011-09-27 | Asml Netherlands B.V. | Lithographic apparatus and device manufacturing method |
| US8755027B2 (en) | 2004-10-05 | 2014-06-17 | Asml Netherlands B.V. | Lithographic apparatus and device manufacturing method involving fluid mixing and control of the physical property of a fluid |
| US20100129526A1 (en) * | 2004-12-06 | 2010-05-27 | Sokudo Co., Ltd. | Substrate processing apparatus |
| US9703199B2 (en) | 2004-12-06 | 2017-07-11 | Screen Semiconductor Solutions Co., Ltd. | Substrate processing apparatus |
| US10712675B2 (en) | 2005-02-10 | 2020-07-14 | Asml Netherlands B.V. | Immersion liquid, exposure apparatus, and exposure process |
| US9772565B2 (en) | 2005-02-10 | 2017-09-26 | Asml Netherlands B.V. | Immersion liquid, exposure apparatus, and exposure process |
| US8859188B2 (en) | 2005-02-10 | 2014-10-14 | Asml Netherlands B.V. | Immersion liquid, exposure apparatus, and exposure process |
| US9164391B2 (en) | 2005-02-10 | 2015-10-20 | Asml Netherlands B.V. | Immersion liquid, exposure apparatus, and exposure process |
| US9454088B2 (en) | 2005-02-10 | 2016-09-27 | Asml Netherlands B.V. | Immersion liquid, exposure apparatus, and exposure process |
| US20070117232A1 (en) * | 2005-11-07 | 2007-05-24 | Roel Gronheid | Method of studying interaction between immersion fluid and substrate |
| US20070117040A1 (en) * | 2005-11-21 | 2007-05-24 | International Business Machines Corporation | Water castable-water strippable top coats for 193 nm immersion lithography |
| US20070182896A1 (en) * | 2005-11-23 | 2007-08-09 | Adelman Douglas J | Apparatus for recycling alkane immersion liquids and methods of employment |
| US8363205B2 (en) | 2006-01-18 | 2013-01-29 | Canon Kabishiki Kaisha | Exposure apparatus |
| US20100245789A1 (en) * | 2006-01-18 | 2010-09-30 | Canon Kabushiki Kaisha | Exposure apparatus |
| US7709813B2 (en) * | 2006-04-03 | 2010-05-04 | Nikon Corporation | Incidence surfaces and optical windows that are solvophobic to immersion liquids |
| US20070229957A1 (en) * | 2006-04-03 | 2007-10-04 | Nikon Corporation | Incidence surfaces and optical windows that are solvophobic to immersion liquids |
| US20100176304A1 (en) * | 2006-04-03 | 2010-07-15 | Nikon Corporation | Incidence surfaces and optical windows that are solvophobic to immersion liquids |
| WO2007140012A3 (en) * | 2006-05-26 | 2008-09-18 | Massachusetts Inst Technology | Immersion fluids for lithography |
| US7745102B2 (en) | 2006-05-26 | 2010-06-29 | Massachusetts Institute Of Technology | Immersion fluids for lithography |
| US20080084549A1 (en) * | 2006-10-09 | 2008-04-10 | Rottmayer Robert E | High refractive index media for immersion lithography and method of immersion lithography using same |
| US20090294693A1 (en) * | 2008-06-02 | 2009-12-03 | Lumatech Gmbh | Apparatus and method for optical examination of documents |
| US8308330B2 (en) * | 2008-06-02 | 2012-11-13 | Nath Guenther | Backlight apparatus with remote light source |
| CN104297411A (en) * | 2014-10-17 | 2015-01-21 | 浙江理工大学 | Method for determining average EO number of alkylphenol polyoxyethylene in mixture |
| CN104297194A (en) * | 2014-10-17 | 2015-01-21 | 浙江理工大学 | Method and device for detecting average EO number of alkylphenol ethoxylates |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20080074831A (en) | 2008-08-13 |
| EP2762976A1 (en) | 2014-08-06 |
| US7879531B2 (en) | 2011-02-01 |
| CN101430509A (en) | 2009-05-13 |
| US20050173682A1 (en) | 2005-08-11 |
| CN101430509B (en) | 2013-05-01 |
| KR100967372B1 (en) | 2010-07-05 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20050161644A1 (en) | Immersion lithography fluids | |
| US8007986B2 (en) | Immersion lithography fluids | |
| KR101202860B1 (en) | Process solutions containing surfactants | |
| US20040029395A1 (en) | Process solutions containing acetylenic diol surfactants | |
| US20060154171A1 (en) | Photoresist composition and method of forming resist pattern | |
| US20070010412A1 (en) | Process solutions containing surfactants | |
| US20120034558A1 (en) | Photolithography material for immersion lithography processes | |
| EP0736809B1 (en) | Composition for anti-reflective coating on resist | |
| US7364837B2 (en) | Method for pattern formation using photoresist cleaning solution | |
| WO2004108780A1 (en) | Resin for photoresist composition, photoresist composition and method for forming resist pattern | |
| JPH10171128A (en) | Concentrated tetramethylammonium hydroxide aqueous solution | |
| US7537882B2 (en) | Anti-reflective coating composition and production method for pattern using the same | |
| KR102593066B1 (en) | Resist pattern forming method and developer for lithography | |
| WO2007052544A1 (en) | Washing liquid and washing method | |
| JP2003107744A (en) | Rinse liquid | |
| KR20080044478A (en) | Photoresist composition useful for immersion process | |
| JP2006259537A (en) | Alkali developer for photosensitive resin composition and concentrated solution thereof | |
| JP2010164609A (en) | Liquid for modifying resist surface and method for forming resist pattern |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| AS | Assignment |
Owner name: AIR PRODUCTS AND CHEMICALS, INC., PENNSYLVANIA Free format text: ASSIGNMENT OF ASSIGNORS INTEREST;ASSIGNORS:ZHANG, PENG;BUDHLALL, BRIDGETTE MARIA;PARRIS, GENE EVERAD;AND OTHERS;REEL/FRAME:015523/0043;SIGNING DATES FROM 20040512 TO 20040626 |
|
| STCB | Information on status: application discontinuation |
Free format text: ABANDONED -- FAILURE TO RESPOND TO AN OFFICE ACTION |