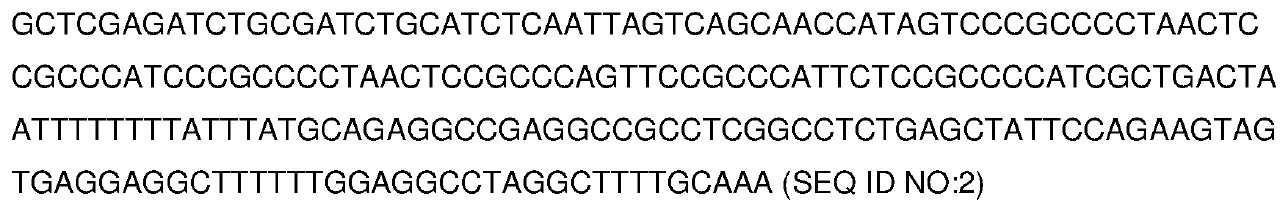KR20200084028A - 영장류 망막 색소 상피 세포-특이적 프로모터 - Google Patents
영장류 망막 색소 상피 세포-특이적 프로모터 Download PDFInfo
- Publication number
- KR20200084028A KR20200084028A KR1020207016484A KR20207016484A KR20200084028A KR 20200084028 A KR20200084028 A KR 20200084028A KR 1020207016484 A KR1020207016484 A KR 1020207016484A KR 20207016484 A KR20207016484 A KR 20207016484A KR 20200084028 A KR20200084028 A KR 20200084028A
- Authority
- KR
- South Korea
- Prior art keywords
- nucleic acid
- acid molecule
- sequence
- acid sequence
- isolated nucleic
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Ceased
Links
- 210000000844 retinal pigment epithelial cell Anatomy 0.000 title claims abstract description 34
- 241000288906 Primates Species 0.000 title claims abstract description 32
- 150000007523 nucleic acids Chemical group 0.000 claims abstract description 132
- 108091028043 Nucleic acid sequence Proteins 0.000 claims abstract description 80
- 108090000623 proteins and genes Proteins 0.000 claims abstract description 75
- 102000039446 nucleic acids Human genes 0.000 claims abstract description 57
- 108020004707 nucleic acids Proteins 0.000 claims abstract description 57
- 230000014509 gene expression Effects 0.000 claims abstract description 51
- 238000000034 method Methods 0.000 claims abstract description 37
- 239000013598 vector Substances 0.000 claims description 19
- 201000010099 disease Diseases 0.000 claims description 10
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 claims description 10
- 239000000790 retinal pigment Substances 0.000 claims description 10
- 238000011282 treatment Methods 0.000 claims description 9
- 239000000049 pigment Substances 0.000 claims description 8
- 210000003583 retinal pigment epithelium Anatomy 0.000 claims description 8
- 239000013603 viral vector Substances 0.000 claims description 7
- 206010012689 Diabetic retinopathy Diseases 0.000 claims description 5
- 206010020880 Hypertrophy Diseases 0.000 claims description 5
- 206010038910 Retinitis Diseases 0.000 claims description 5
- 206010064930 age-related macular degeneration Diseases 0.000 claims description 5
- 208000002780 macular degeneration Diseases 0.000 claims description 5
- FWMNVWWHGCHHJJ-SKKKGAJSSA-N 4-amino-1-[(2r)-6-amino-2-[[(2r)-2-[[(2r)-2-[[(2r)-2-amino-3-phenylpropanoyl]amino]-3-phenylpropanoyl]amino]-4-methylpentanoyl]amino]hexanoyl]piperidine-4-carboxylic acid Chemical compound C([C@H](C(=O)N[C@H](CC(C)C)C(=O)N[C@H](CCCCN)C(=O)N1CCC(N)(CC1)C(O)=O)NC(=O)[C@H](N)CC=1C=CC=CC=1)C1=CC=CC=C1 FWMNVWWHGCHHJJ-SKKKGAJSSA-N 0.000 claims description 3
- 210000004027 cell Anatomy 0.000 description 55
- 108090000765 processed proteins & peptides Proteins 0.000 description 34
- 102000004196 processed proteins & peptides Human genes 0.000 description 32
- 229920001184 polypeptide Polymers 0.000 description 31
- 108020004414 DNA Proteins 0.000 description 28
- 210000001525 retina Anatomy 0.000 description 21
- 241000700605 Viruses Species 0.000 description 18
- 239000000203 mixture Substances 0.000 description 18
- 241001465754 Metazoa Species 0.000 description 16
- 102000040430 polynucleotide Human genes 0.000 description 14
- 108091033319 polynucleotide Proteins 0.000 description 14
- 239000002157 polynucleotide Substances 0.000 description 14
- 125000003275 alpha amino acid group Chemical group 0.000 description 12
- 102000004169 proteins and genes Human genes 0.000 description 12
- 108091032973 (ribonucleotides)n+m Proteins 0.000 description 11
- 150000001875 compounds Chemical class 0.000 description 11
- 230000000694 effects Effects 0.000 description 11
- 230000004048 modification Effects 0.000 description 11
- 238000012986 modification Methods 0.000 description 11
- 210000002569 neuron Anatomy 0.000 description 11
- 235000018102 proteins Nutrition 0.000 description 11
- 235000001014 amino acid Nutrition 0.000 description 10
- 150000001413 amino acids Chemical class 0.000 description 10
- 239000000243 solution Substances 0.000 description 10
- 108700019146 Transgenes Proteins 0.000 description 9
- 239000005090 green fluorescent protein Substances 0.000 description 9
- 230000002207 retinal effect Effects 0.000 description 9
- ZHNUHDYFZUAESO-UHFFFAOYSA-N Formamide Chemical compound NC=O ZHNUHDYFZUAESO-UHFFFAOYSA-N 0.000 description 8
- 230000002068 genetic effect Effects 0.000 description 8
- 238000002864 sequence alignment Methods 0.000 description 8
- 230000000946 synaptic effect Effects 0.000 description 8
- 239000003795 chemical substances by application Substances 0.000 description 7
- 238000012217 deletion Methods 0.000 description 7
- 230000037430 deletion Effects 0.000 description 7
- 238000013518 transcription Methods 0.000 description 7
- 230000035897 transcription Effects 0.000 description 7
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- 230000015572 biosynthetic process Effects 0.000 description 6
- 238000002360 preparation method Methods 0.000 description 6
- 230000003612 virological effect Effects 0.000 description 6
- 241000711798 Rabies lyssavirus Species 0.000 description 5
- 108091023040 Transcription factor Proteins 0.000 description 5
- 102000040945 Transcription factor Human genes 0.000 description 5
- 239000003153 chemical reaction reagent Substances 0.000 description 5
- 108010048367 enhanced green fluorescent protein Proteins 0.000 description 5
- 238000011156 evaluation Methods 0.000 description 5
- 238000009472 formulation Methods 0.000 description 5
- 238000009396 hybridization Methods 0.000 description 5
- 238000000338 in vitro Methods 0.000 description 5
- 239000007924 injection Substances 0.000 description 5
- 238000002347 injection Methods 0.000 description 5
- 238000004519 manufacturing process Methods 0.000 description 5
- 239000000463 material Substances 0.000 description 5
- 125000003729 nucleotide group Chemical group 0.000 description 5
- 239000002953 phosphate buffered saline Substances 0.000 description 5
- 108091008695 photoreceptors Proteins 0.000 description 5
- 230000001105 regulatory effect Effects 0.000 description 5
- -1 silencers Substances 0.000 description 5
- 210000000225 synapse Anatomy 0.000 description 5
- 230000008685 targeting Effects 0.000 description 5
- 230000000903 blocking effect Effects 0.000 description 4
- 230000001413 cellular effect Effects 0.000 description 4
- 210000000349 chromosome Anatomy 0.000 description 4
- 239000002299 complementary DNA Substances 0.000 description 4
- 238000004590 computer program Methods 0.000 description 4
- 238000001514 detection method Methods 0.000 description 4
- 239000003814 drug Substances 0.000 description 4
- 230000005284 excitation Effects 0.000 description 4
- 238000005755 formation reaction Methods 0.000 description 4
- 230000003834 intracellular effect Effects 0.000 description 4
- 239000012528 membrane Substances 0.000 description 4
- 239000002773 nucleotide Substances 0.000 description 4
- 241000894007 species Species 0.000 description 4
- 238000011144 upstream manufacturing Methods 0.000 description 4
- 230000000007 visual effect Effects 0.000 description 4
- 238000005406 washing Methods 0.000 description 4
- 241000972773 Aulopiformes Species 0.000 description 3
- 108091026890 Coding region Proteins 0.000 description 3
- 102000053602 DNA Human genes 0.000 description 3
- 206010028980 Neoplasm Diseases 0.000 description 3
- 108020004511 Recombinant DNA Proteins 0.000 description 3
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 3
- 108700009124 Transcription Initiation Site Proteins 0.000 description 3
- 239000004480 active ingredient Substances 0.000 description 3
- 210000004899 c-terminal region Anatomy 0.000 description 3
- 125000004122 cyclic group Chemical group 0.000 description 3
- 238000009826 distribution Methods 0.000 description 3
- 239000000839 emulsion Substances 0.000 description 3
- 210000000981 epithelium Anatomy 0.000 description 3
- 238000001917 fluorescence detection Methods 0.000 description 3
- 239000012634 fragment Substances 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- 210000004602 germ cell Anatomy 0.000 description 3
- 230000033001 locomotion Effects 0.000 description 3
- 108020004999 messenger RNA Proteins 0.000 description 3
- 239000003068 molecular probe Substances 0.000 description 3
- 230000035772 mutation Effects 0.000 description 3
- 230000001537 neural effect Effects 0.000 description 3
- 230000003287 optical effect Effects 0.000 description 3
- 230000001323 posttranslational effect Effects 0.000 description 3
- 239000000843 powder Substances 0.000 description 3
- 230000008569 process Effects 0.000 description 3
- 235000019515 salmon Nutrition 0.000 description 3
- 150000003839 salts Chemical class 0.000 description 3
- 239000011780 sodium chloride Substances 0.000 description 3
- 235000019333 sodium laurylsulphate Nutrition 0.000 description 3
- 238000001228 spectrum Methods 0.000 description 3
- 239000000725 suspension Substances 0.000 description 3
- 241001529453 unidentified herpesvirus Species 0.000 description 3
- 239000003981 vehicle Substances 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- 108700028369 Alleles Proteins 0.000 description 2
- 241000272517 Anseriformes Species 0.000 description 2
- 241000894006 Bacteria Species 0.000 description 2
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 2
- 241000701022 Cytomegalovirus Species 0.000 description 2
- 241000283074 Equus asinus Species 0.000 description 2
- 108700028146 Genetic Enhancer Elements Proteins 0.000 description 2
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Chemical compound OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 2
- 108010043121 Green Fluorescent Proteins Proteins 0.000 description 2
- 102000004144 Green Fluorescent Proteins Human genes 0.000 description 2
- 241001074968 Halobacteria Species 0.000 description 2
- 108091092195 Intron Proteins 0.000 description 2
- 102000004310 Ion Channels Human genes 0.000 description 2
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 2
- 241000713666 Lentivirus Species 0.000 description 2
- 241000699660 Mus musculus Species 0.000 description 2
- 241000699670 Mus sp. Species 0.000 description 2
- 102000010175 Opsin Human genes 0.000 description 2
- 108050001704 Opsin Proteins 0.000 description 2
- 241000700159 Rattus Species 0.000 description 2
- 108700008625 Reporter Genes Proteins 0.000 description 2
- 241000714474 Rous sarcoma virus Species 0.000 description 2
- VYPSYNLAJGMNEJ-UHFFFAOYSA-N Silicium dioxide Chemical compound O=[Si]=O VYPSYNLAJGMNEJ-UHFFFAOYSA-N 0.000 description 2
- 208000037065 Subacute sclerosing leukoencephalitis Diseases 0.000 description 2
- 206010042297 Subacute sclerosing panencephalitis Diseases 0.000 description 2
- 239000013504 Triton X-100 Substances 0.000 description 2
- 229920004890 Triton X-100 Polymers 0.000 description 2
- 241000251539 Vertebrata <Metazoa> Species 0.000 description 2
- 230000004913 activation Effects 0.000 description 2
- 239000000443 aerosol Substances 0.000 description 2
- 210000000411 amacrine cell Anatomy 0.000 description 2
- 230000004071 biological effect Effects 0.000 description 2
- 210000002459 blastocyst Anatomy 0.000 description 2
- 210000004556 brain Anatomy 0.000 description 2
- 239000000872 buffer Substances 0.000 description 2
- 210000000234 capsid Anatomy 0.000 description 2
- 239000002775 capsule Substances 0.000 description 2
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 description 2
- 238000003776 cleavage reaction Methods 0.000 description 2
- 239000002552 dosage form Substances 0.000 description 2
- 239000003937 drug carrier Substances 0.000 description 2
- 238000010291 electrical method Methods 0.000 description 2
- 239000003623 enhancer Substances 0.000 description 2
- 230000002964 excitative effect Effects 0.000 description 2
- 108091006047 fluorescent proteins Proteins 0.000 description 2
- 102000034287 fluorescent proteins Human genes 0.000 description 2
- 238000001727 in vivo Methods 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 208000015181 infectious disease Diseases 0.000 description 2
- 239000012212 insulator Substances 0.000 description 2
- 239000008101 lactose Substances 0.000 description 2
- 239000010410 layer Substances 0.000 description 2
- 150000002632 lipids Chemical class 0.000 description 2
- 239000007788 liquid Substances 0.000 description 2
- HQKMJHAJHXVSDF-UHFFFAOYSA-L magnesium stearate Chemical compound [Mg+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O HQKMJHAJHXVSDF-UHFFFAOYSA-L 0.000 description 2
- 210000004962 mammalian cell Anatomy 0.000 description 2
- 210000001161 mammalian embryo Anatomy 0.000 description 2
- 239000003550 marker Substances 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 230000001404 mediated effect Effects 0.000 description 2
- 238000010172 mouse model Methods 0.000 description 2
- 230000003647 oxidation Effects 0.000 description 2
- 238000007254 oxidation reaction Methods 0.000 description 2
- 239000002245 particle Substances 0.000 description 2
- 230000008488 polyadenylation Effects 0.000 description 2
- 239000003755 preservative agent Substances 0.000 description 2
- 210000005215 presynaptic neuron Anatomy 0.000 description 2
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 2
- 238000012545 processing Methods 0.000 description 2
- 238000011084 recovery Methods 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 230000007017 scission Effects 0.000 description 2
- 238000012216 screening Methods 0.000 description 2
- 239000008174 sterile solution Substances 0.000 description 2
- 239000000375 suspending agent Substances 0.000 description 2
- 238000003786 synthesis reaction Methods 0.000 description 2
- 229940124597 therapeutic agent Drugs 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 238000010798 ubiquitination Methods 0.000 description 2
- 230000034512 ubiquitination Effects 0.000 description 2
- 108091005957 yellow fluorescent proteins Proteins 0.000 description 2
- 102000040650 (ribonucleotides)n+m Human genes 0.000 description 1
- NWUYHJFMYQTDRP-UHFFFAOYSA-N 1,2-bis(ethenyl)benzene;1-ethenyl-2-ethylbenzene;styrene Chemical compound C=CC1=CC=CC=C1.CCC1=CC=CC=C1C=C.C=CC1=CC=CC=C1C=C NWUYHJFMYQTDRP-UHFFFAOYSA-N 0.000 description 1
- DDMOUSALMHHKOS-UHFFFAOYSA-N 1,2-dichloro-1,1,2,2-tetrafluoroethane Chemical compound FC(F)(Cl)C(F)(F)Cl DDMOUSALMHHKOS-UHFFFAOYSA-N 0.000 description 1
- IIZPXYDJLKNOIY-JXPKJXOSSA-N 1-palmitoyl-2-arachidonoyl-sn-glycero-3-phosphocholine Chemical compound CCCCCCCCCCCCCCCC(=O)OC[C@H](COP([O-])(=O)OCC[N+](C)(C)C)OC(=O)CCC\C=C/C\C=C/C\C=C/C\C=C/CCCCC IIZPXYDJLKNOIY-JXPKJXOSSA-N 0.000 description 1
- FWBHETKCLVMNFS-UHFFFAOYSA-N 4',6-Diamino-2-phenylindol Chemical compound C1=CC(C(=N)N)=CC=C1C1=CC2=CC=C(C(N)=N)C=C2N1 FWBHETKCLVMNFS-UHFFFAOYSA-N 0.000 description 1
- ODHCTXKNWHHXJC-VKHMYHEASA-N 5-oxo-L-proline Chemical compound OC(=O)[C@@H]1CCC(=O)N1 ODHCTXKNWHHXJC-VKHMYHEASA-N 0.000 description 1
- 230000005730 ADP ribosylation Effects 0.000 description 1
- 102000007469 Actins Human genes 0.000 description 1
- 108010085238 Actins Proteins 0.000 description 1
- 241000243290 Aequorea Species 0.000 description 1
- 241000426851 Aequorea aequorea Species 0.000 description 1
- 241000242764 Aequorea victoria Species 0.000 description 1
- 229920001817 Agar Polymers 0.000 description 1
- 206010001497 Agitation Diseases 0.000 description 1
- 239000012103 Alexa Fluor 488 Substances 0.000 description 1
- 239000012112 Alexa Fluor 633 Substances 0.000 description 1
- 235000019489 Almond oil Nutrition 0.000 description 1
- 241000283690 Bos taurus Species 0.000 description 1
- 125000001433 C-terminal amino-acid group Chemical group 0.000 description 1
- 241000282472 Canis lupus familiaris Species 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- VEXZGXHMUGYJMC-UHFFFAOYSA-M Chloride anion Chemical compound [Cl-] VEXZGXHMUGYJMC-UHFFFAOYSA-M 0.000 description 1
- 241000195628 Chlorophyta Species 0.000 description 1
- 108010077544 Chromatin Proteins 0.000 description 1
- 108091062157 Cis-regulatory element Proteins 0.000 description 1
- 229920002261 Corn starch Polymers 0.000 description 1
- 102000018832 Cytochromes Human genes 0.000 description 1
- 108010052832 Cytochromes Proteins 0.000 description 1
- 239000004338 Dichlorodifluoromethane Substances 0.000 description 1
- 206010067671 Disease complication Diseases 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 241000196324 Embryophyta Species 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 241000283086 Equidae Species 0.000 description 1
- 108700039887 Essential Genes Proteins 0.000 description 1
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 1
- 108700024394 Exon Proteins 0.000 description 1
- 241000282326 Felis catus Species 0.000 description 1
- 241000287828 Gallus gallus Species 0.000 description 1
- 208000003098 Ganglion Cysts Diseases 0.000 description 1
- 108010010803 Gelatin Proteins 0.000 description 1
- 101150009006 HIS3 gene Proteins 0.000 description 1
- 241000204946 Halobacterium salinarum Species 0.000 description 1
- 101100246753 Halobacterium salinarum (strain ATCC 700922 / JCM 11081 / NRC-1) pyrF gene Proteins 0.000 description 1
- 102000001554 Hemoglobins Human genes 0.000 description 1
- 108010054147 Hemoglobins Proteins 0.000 description 1
- HTTJABKRGRZYRN-UHFFFAOYSA-N Heparin Chemical compound OC1C(NC(=O)C)C(O)OC(COS(O)(=O)=O)C1OC1C(OS(O)(=O)=O)C(O)C(OC2C(C(OS(O)(=O)=O)C(OC3C(C(O)C(O)C(O3)C(O)=O)OS(O)(=O)=O)C(CO)O2)NS(O)(=O)=O)C(C(O)=O)O1 HTTJABKRGRZYRN-UHFFFAOYSA-N 0.000 description 1
- SQUHHTBVTRBESD-UHFFFAOYSA-N Hexa-Ac-myo-Inositol Natural products CC(=O)OC1C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C(OC(C)=O)C1OC(C)=O SQUHHTBVTRBESD-UHFFFAOYSA-N 0.000 description 1
- 108010033040 Histones Proteins 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 229930010555 Inosine Natural products 0.000 description 1
- UGQMRVRMYYASKQ-KQYNXXCUSA-N Inosine Chemical compound O[C@@H]1[C@H](O)[C@@H](CO)O[C@H]1N1C2=NC=NC(O)=C2N=C1 UGQMRVRMYYASKQ-KQYNXXCUSA-N 0.000 description 1
- 108010083687 Ion Pumps Proteins 0.000 description 1
- 240000000599 Lentinula edodes Species 0.000 description 1
- 235000001715 Lentinula edodes Nutrition 0.000 description 1
- 235000010643 Leucaena leucocephala Nutrition 0.000 description 1
- 240000007472 Leucaena leucocephala Species 0.000 description 1
- 241000282553 Macaca Species 0.000 description 1
- 241000282560 Macaca mulatta Species 0.000 description 1
- 108010052285 Membrane Proteins Proteins 0.000 description 1
- 229920000168 Microcrystalline cellulose Polymers 0.000 description 1
- 241000699666 Mus <mouse, genus> Species 0.000 description 1
- 241000204971 Natronomonas pharaonis Species 0.000 description 1
- 208000012902 Nervous system disease Diseases 0.000 description 1
- 208000025966 Neurological disease Diseases 0.000 description 1
- 108010047956 Nucleosomes Proteins 0.000 description 1
- 206010057249 Phagocytosis Diseases 0.000 description 1
- 239000002202 Polyethylene glycol Substances 0.000 description 1
- 102100021904 Potassium-transporting ATPase alpha chain 1 Human genes 0.000 description 1
- 108010029485 Protein Isoforms Proteins 0.000 description 1
- 102000001708 Protein Isoforms Human genes 0.000 description 1
- 108010083204 Proton Pumps Proteins 0.000 description 1
- 206010037742 Rabies Diseases 0.000 description 1
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 1
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 1
- 241000242743 Renilla reniformis Species 0.000 description 1
- 208000017442 Retinal disease Diseases 0.000 description 1
- 101100394989 Rhodopseudomonas palustris (strain ATCC BAA-98 / CGA009) hisI gene Proteins 0.000 description 1
- 102000005801 Rod Opsins Human genes 0.000 description 1
- 108010005063 Rod Opsins Proteins 0.000 description 1
- 241000242583 Scyphozoa Species 0.000 description 1
- 108010022999 Serine Proteases Proteins 0.000 description 1
- 102000012479 Serine Proteases Human genes 0.000 description 1
- 241000700584 Simplexvirus Species 0.000 description 1
- 108020004682 Single-Stranded DNA Proteins 0.000 description 1
- 229920002472 Starch Polymers 0.000 description 1
- 108090000787 Subtilisin Proteins 0.000 description 1
- 241000282887 Suidae Species 0.000 description 1
- 208000005400 Synovial Cyst Diseases 0.000 description 1
- 101150006914 TRP1 gene Proteins 0.000 description 1
- 108020004566 Transfer RNA Proteins 0.000 description 1
- LVTKHGUGBGNBPL-UHFFFAOYSA-N Trp-P-1 Chemical compound N1C2=CC=CC=C2C2=C1C(C)=C(N)N=C2C LVTKHGUGBGNBPL-UHFFFAOYSA-N 0.000 description 1
- 101150050575 URA3 gene Proteins 0.000 description 1
- 108090000848 Ubiquitin Proteins 0.000 description 1
- 102000044159 Ubiquitin Human genes 0.000 description 1
- 241000195615 Volvox Species 0.000 description 1
- 108091093126 WHP Posttrascriptional Response Element Proteins 0.000 description 1
- 241001492404 Woodchuck hepatitis virus Species 0.000 description 1
- 238000002835 absorbance Methods 0.000 description 1
- 230000021736 acetylation Effects 0.000 description 1
- 238000006640 acetylation reaction Methods 0.000 description 1
- 230000010933 acylation Effects 0.000 description 1
- 238000005917 acylation reaction Methods 0.000 description 1
- 239000000654 additive Substances 0.000 description 1
- 239000008272 agar Substances 0.000 description 1
- 230000002776 aggregation Effects 0.000 description 1
- 238000004220 aggregation Methods 0.000 description 1
- 150000004347 all-trans-retinol derivatives Chemical class 0.000 description 1
- 239000008168 almond oil Substances 0.000 description 1
- 230000004075 alteration Effects 0.000 description 1
- 230000009435 amidation Effects 0.000 description 1
- 238000007112 amidation reaction Methods 0.000 description 1
- 125000000539 amino acid group Chemical group 0.000 description 1
- 238000004458 analytical method Methods 0.000 description 1
- 238000013459 approach Methods 0.000 description 1
- 239000003125 aqueous solvent Substances 0.000 description 1
- 239000008135 aqueous vehicle Substances 0.000 description 1
- 230000010516 arginylation Effects 0.000 description 1
- 230000001580 bacterial effect Effects 0.000 description 1
- 239000011230 binding agent Substances 0.000 description 1
- 238000004166 bioassay Methods 0.000 description 1
- 230000008033 biological extinction Effects 0.000 description 1
- 230000008827 biological function Effects 0.000 description 1
- 230000033228 biological regulation Effects 0.000 description 1
- 230000005540 biological transmission Effects 0.000 description 1
- 238000006664 bond formation reaction Methods 0.000 description 1
- 239000007853 buffer solution Substances 0.000 description 1
- 239000011575 calcium Substances 0.000 description 1
- FUFJGUQYACFECW-UHFFFAOYSA-L calcium hydrogenphosphate Chemical compound [Ca+2].OP([O-])([O-])=O FUFJGUQYACFECW-UHFFFAOYSA-L 0.000 description 1
- 230000009460 calcium influx Effects 0.000 description 1
- 239000001569 carbon dioxide Substances 0.000 description 1
- 229910002092 carbon dioxide Inorganic materials 0.000 description 1
- 229960004424 carbon dioxide Drugs 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 125000002091 cationic group Chemical group 0.000 description 1
- 230000006037 cell lysis Effects 0.000 description 1
- 230000033077 cellular process Effects 0.000 description 1
- 239000001913 cellulose Substances 0.000 description 1
- 235000010980 cellulose Nutrition 0.000 description 1
- 229920002678 cellulose Polymers 0.000 description 1
- 238000007385 chemical modification Methods 0.000 description 1
- 235000013330 chicken meat Nutrition 0.000 description 1
- 210000003161 choroid Anatomy 0.000 description 1
- 210000003483 chromatin Anatomy 0.000 description 1
- 230000002759 chromosomal effect Effects 0.000 description 1
- 210000004081 cilia Anatomy 0.000 description 1
- 230000001886 ciliary effect Effects 0.000 description 1
- 238000010367 cloning Methods 0.000 description 1
- 230000001149 cognitive effect Effects 0.000 description 1
- 238000004891 communication Methods 0.000 description 1
- 238000004624 confocal microscopy Methods 0.000 description 1
- 238000013270 controlled release Methods 0.000 description 1
- 239000008120 corn starch Substances 0.000 description 1
- 210000004087 cornea Anatomy 0.000 description 1
- 239000006071 cream Substances 0.000 description 1
- 238000004132 cross linking Methods 0.000 description 1
- 239000013078 crystal Substances 0.000 description 1
- 235000018417 cysteine Nutrition 0.000 description 1
- 150000001945 cysteines Chemical class 0.000 description 1
- 230000017858 demethylation Effects 0.000 description 1
- 238000010520 demethylation reaction Methods 0.000 description 1
- 238000001212 derivatisation Methods 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 229960000633 dextran sulfate Drugs 0.000 description 1
- 235000019700 dicalcium phosphate Nutrition 0.000 description 1
- PXBRQCKWGAHEHS-UHFFFAOYSA-N dichlorodifluoromethane Chemical compound FC(F)(Cl)Cl PXBRQCKWGAHEHS-UHFFFAOYSA-N 0.000 description 1
- 235000019404 dichlorodifluoromethane Nutrition 0.000 description 1
- 229940042935 dichlorodifluoromethane Drugs 0.000 description 1
- 229940087091 dichlorotetrafluoroethane Drugs 0.000 description 1
- LOKCTEFSRHRXRJ-UHFFFAOYSA-I dipotassium trisodium dihydrogen phosphate hydrogen phosphate dichloride Chemical compound P(=O)(O)(O)[O-].[K+].P(=O)(O)([O-])[O-].[Na+].[Na+].[Cl-].[K+].[Cl-].[Na+] LOKCTEFSRHRXRJ-UHFFFAOYSA-I 0.000 description 1
- 239000007884 disintegrant Substances 0.000 description 1
- 239000002270 dispersing agent Substances 0.000 description 1
- 229940079593 drug Drugs 0.000 description 1
- 238000007876 drug discovery Methods 0.000 description 1
- 239000000975 dye Substances 0.000 description 1
- 238000001962 electrophoresis Methods 0.000 description 1
- 238000004520 electroporation Methods 0.000 description 1
- 210000002257 embryonic structure Anatomy 0.000 description 1
- 239000003995 emulsifying agent Substances 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 230000001973 epigenetic effect Effects 0.000 description 1
- 210000002919 epithelial cell Anatomy 0.000 description 1
- 150000002148 esters Chemical class 0.000 description 1
- BEFDCLMNVWHSGT-UHFFFAOYSA-N ethenylcyclopentane Chemical compound C=CC1CCCC1 BEFDCLMNVWHSGT-UHFFFAOYSA-N 0.000 description 1
- 238000002474 experimental method Methods 0.000 description 1
- 239000003925 fat Substances 0.000 description 1
- 230000004720 fertilization Effects 0.000 description 1
- 230000001605 fetal effect Effects 0.000 description 1
- 210000003754 fetus Anatomy 0.000 description 1
- 239000000835 fiber Substances 0.000 description 1
- 239000000945 filler Substances 0.000 description 1
- 150000002211 flavins Chemical class 0.000 description 1
- GNBHRKFJIUUOQI-UHFFFAOYSA-N fluorescein Chemical compound O1C(=O)C2=CC=CC=C2C21C1=CC=C(O)C=C1OC1=CC(O)=CC=C21 GNBHRKFJIUUOQI-UHFFFAOYSA-N 0.000 description 1
- 239000007850 fluorescent dye Substances 0.000 description 1
- 239000011888 foil Substances 0.000 description 1
- 230000022244 formylation Effects 0.000 description 1
- 238000006170 formylation reaction Methods 0.000 description 1
- 239000007789 gas Substances 0.000 description 1
- 239000008273 gelatin Substances 0.000 description 1
- 229920000159 gelatin Polymers 0.000 description 1
- 235000019322 gelatine Nutrition 0.000 description 1
- 235000011852 gelatine desserts Nutrition 0.000 description 1
- 230000004545 gene duplication Effects 0.000 description 1
- 230000030279 gene silencing Effects 0.000 description 1
- 230000007274 generation of a signal involved in cell-cell signaling Effects 0.000 description 1
- 238000010353 genetic engineering Methods 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 230000013595 glycosylation Effects 0.000 description 1
- 238000006206 glycosylation reaction Methods 0.000 description 1
- PCHJSUWPFVWCPO-UHFFFAOYSA-N gold Chemical compound [Au] PCHJSUWPFVWCPO-UHFFFAOYSA-N 0.000 description 1
- 239000010931 gold Substances 0.000 description 1
- 229910052737 gold Inorganic materials 0.000 description 1
- 239000008187 granular material Substances 0.000 description 1
- 150000003278 haem Chemical group 0.000 description 1
- 229960002897 heparin Drugs 0.000 description 1
- 229920000669 heparin Polymers 0.000 description 1
- 239000012456 homogeneous solution Substances 0.000 description 1
- 238000002744 homologous recombination Methods 0.000 description 1
- 230000006801 homologous recombination Effects 0.000 description 1
- 230000002209 hydrophobic effect Effects 0.000 description 1
- 230000033444 hydroxylation Effects 0.000 description 1
- 238000005805 hydroxylation reaction Methods 0.000 description 1
- 239000001866 hydroxypropyl methyl cellulose Substances 0.000 description 1
- 235000010979 hydroxypropyl methyl cellulose Nutrition 0.000 description 1
- 229920003088 hydroxypropyl methyl cellulose Polymers 0.000 description 1
- UFVKGYZPFZQRLF-UHFFFAOYSA-N hydroxypropyl methyl cellulose Chemical compound OC1C(O)C(OC)OC(CO)C1OC1C(O)C(O)C(OC2C(C(O)C(OC3C(C(O)C(O)C(CO)O3)O)C(CO)O2)O)C(CO)O1 UFVKGYZPFZQRLF-UHFFFAOYSA-N 0.000 description 1
- 238000010191 image analysis Methods 0.000 description 1
- 210000002865 immune cell Anatomy 0.000 description 1
- 230000003832 immune regulation Effects 0.000 description 1
- 238000002513 implantation Methods 0.000 description 1
- 230000006872 improvement Effects 0.000 description 1
- 230000000415 inactivating effect Effects 0.000 description 1
- 239000012678 infectious agent Substances 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 230000005764 inhibitory process Effects 0.000 description 1
- 230000000977 initiatory effect Effects 0.000 description 1
- 239000007972 injectable composition Substances 0.000 description 1
- 229960003786 inosine Drugs 0.000 description 1
- 229960000367 inositol Drugs 0.000 description 1
- CDAISMWEOUEBRE-GPIVLXJGSA-N inositol Chemical compound O[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@H](O)[C@@H]1O CDAISMWEOUEBRE-GPIVLXJGSA-N 0.000 description 1
- 230000003993 interaction Effects 0.000 description 1
- 239000003456 ion exchange resin Substances 0.000 description 1
- 229920003303 ion-exchange polymer Polymers 0.000 description 1
- 150000002500 ions Chemical class 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 239000000787 lecithin Substances 0.000 description 1
- 235000010445 lecithin Nutrition 0.000 description 1
- 229940067606 lecithin Drugs 0.000 description 1
- 239000003446 ligand Substances 0.000 description 1
- 230000031700 light absorption Effects 0.000 description 1
- 239000002502 liposome Substances 0.000 description 1
- 239000006210 lotion Substances 0.000 description 1
- 239000000314 lubricant Substances 0.000 description 1
- 235000019359 magnesium stearate Nutrition 0.000 description 1
- 238000012423 maintenance Methods 0.000 description 1
- 230000007246 mechanism Effects 0.000 description 1
- 229910052751 metal Inorganic materials 0.000 description 1
- 239000002184 metal Substances 0.000 description 1
- MYWUZJCMWCOHBA-VIFPVBQESA-N methamphetamine Chemical compound CN[C@@H](C)CC1=CC=CC=C1 MYWUZJCMWCOHBA-VIFPVBQESA-N 0.000 description 1
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 1
- 239000004292 methyl p-hydroxybenzoate Substances 0.000 description 1
- 235000010270 methyl p-hydroxybenzoate Nutrition 0.000 description 1
- 230000011987 methylation Effects 0.000 description 1
- 238000007069 methylation reaction Methods 0.000 description 1
- 230000000813 microbial effect Effects 0.000 description 1
- 235000019813 microcrystalline cellulose Nutrition 0.000 description 1
- 239000008108 microcrystalline cellulose Substances 0.000 description 1
- 229940016286 microcrystalline cellulose Drugs 0.000 description 1
- 238000000520 microinjection Methods 0.000 description 1
- 238000002406 microsurgery Methods 0.000 description 1
- 210000000663 muscle cell Anatomy 0.000 description 1
- 238000002703 mutagenesis Methods 0.000 description 1
- 231100000350 mutagenesis Toxicity 0.000 description 1
- 230000007498 myristoylation Effects 0.000 description 1
- 239000006199 nebulizer Substances 0.000 description 1
- 230000004770 neurodegeneration Effects 0.000 description 1
- 208000015122 neurodegenerative disease Diseases 0.000 description 1
- 230000000926 neurological effect Effects 0.000 description 1
- 239000002687 nonaqueous vehicle Substances 0.000 description 1
- 239000002736 nonionic surfactant Substances 0.000 description 1
- 231100000252 nontoxic Toxicity 0.000 description 1
- 230000003000 nontoxic effect Effects 0.000 description 1
- 210000001623 nucleosome Anatomy 0.000 description 1
- 210000004940 nucleus Anatomy 0.000 description 1
- 239000003921 oil Substances 0.000 description 1
- 235000019198 oils Nutrition 0.000 description 1
- 230000008520 organization Effects 0.000 description 1
- 238000007911 parenteral administration Methods 0.000 description 1
- 230000001575 pathological effect Effects 0.000 description 1
- 230000037361 pathway Effects 0.000 description 1
- 230000008782 phagocytosis Effects 0.000 description 1
- 239000008194 pharmaceutical composition Substances 0.000 description 1
- 239000000546 pharmaceutical excipient Substances 0.000 description 1
- 239000000825 pharmaceutical preparation Substances 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 210000004694 pigment cell Anatomy 0.000 description 1
- 239000013600 plasmid vector Substances 0.000 description 1
- 239000004033 plastic Substances 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 229920001223 polyethylene glycol Polymers 0.000 description 1
- 229920000136 polysorbate Polymers 0.000 description 1
- 239000001267 polyvinylpyrrolidone Substances 0.000 description 1
- 229920000036 polyvinylpyrrolidone Polymers 0.000 description 1
- 235000013855 polyvinylpyrrolidone Nutrition 0.000 description 1
- 230000001242 postsynaptic effect Effects 0.000 description 1
- 229920001592 potato starch Polymers 0.000 description 1
- 230000003389 potentiating effect Effects 0.000 description 1
- 230000013823 prenylation Effects 0.000 description 1
- 230000002335 preservative effect Effects 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 239000003380 propellant Substances 0.000 description 1
- 239000004405 propyl p-hydroxybenzoate Substances 0.000 description 1
- 235000010232 propyl p-hydroxybenzoate Nutrition 0.000 description 1
- QELSKZZBTMNZEB-UHFFFAOYSA-N propylparaben Chemical compound CCCOC(=O)C1=CC=C(O)C=C1 QELSKZZBTMNZEB-UHFFFAOYSA-N 0.000 description 1
- 125000006239 protecting group Chemical group 0.000 description 1
- 230000001681 protective effect Effects 0.000 description 1
- 210000004777 protein coat Anatomy 0.000 description 1
- 230000002797 proteolythic effect Effects 0.000 description 1
- 229940043131 pyroglutamate Drugs 0.000 description 1
- 238000004445 quantitative analysis Methods 0.000 description 1
- 230000006340 racemization Effects 0.000 description 1
- 230000006798 recombination Effects 0.000 description 1
- 238000005215 recombination Methods 0.000 description 1
- 230000022532 regulation of transcription, DNA-dependent Effects 0.000 description 1
- 230000010076 replication Effects 0.000 description 1
- 108091008146 restriction endonucleases Proteins 0.000 description 1
- 210000001116 retinal neuron Anatomy 0.000 description 1
- 238000012552 review Methods 0.000 description 1
- PYWVYCXTNDRMGF-UHFFFAOYSA-N rhodamine B Chemical compound [Cl-].C=12C=CC(=[N+](CC)CC)C=C2OC2=CC(N(CC)CC)=CC=C2C=1C1=CC=CC=C1C(O)=O PYWVYCXTNDRMGF-UHFFFAOYSA-N 0.000 description 1
- 238000007363 ring formation reaction Methods 0.000 description 1
- 239000000523 sample Substances 0.000 description 1
- 210000003786 sclera Anatomy 0.000 description 1
- CDAISMWEOUEBRE-UHFFFAOYSA-N scyllo-inosotol Natural products OC1C(O)C(O)C(O)C(O)C1O CDAISMWEOUEBRE-UHFFFAOYSA-N 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 230000001953 sensory effect Effects 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 239000000377 silicon dioxide Substances 0.000 description 1
- 239000002356 single layer Substances 0.000 description 1
- 239000001509 sodium citrate Substances 0.000 description 1
- 239000001488 sodium phosphate Substances 0.000 description 1
- 229910000162 sodium phosphate Inorganic materials 0.000 description 1
- 229940079832 sodium starch glycolate Drugs 0.000 description 1
- 239000008109 sodium starch glycolate Substances 0.000 description 1
- 229920003109 sodium starch glycolate Polymers 0.000 description 1
- 239000012453 solvate Substances 0.000 description 1
- 239000004334 sorbic acid Substances 0.000 description 1
- 235000010199 sorbic acid Nutrition 0.000 description 1
- 229940075582 sorbic acid Drugs 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 235000010356 sorbitol Nutrition 0.000 description 1
- 230000003595 spectral effect Effects 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 230000007480 spreading Effects 0.000 description 1
- 238000003892 spreading Methods 0.000 description 1
- 239000003381 stabilizer Substances 0.000 description 1
- 238000010561 standard procedure Methods 0.000 description 1
- 239000008107 starch Substances 0.000 description 1
- 229940032147 starch Drugs 0.000 description 1
- 235000019698 starch Nutrition 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 230000019635 sulfation Effects 0.000 description 1
- 238000005670 sulfation reaction Methods 0.000 description 1
- 238000010189 synthetic method Methods 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000000454 talc Substances 0.000 description 1
- 229910052623 talc Inorganic materials 0.000 description 1
- 238000002560 therapeutic procedure Methods 0.000 description 1
- ANRHNWWPFJCPAZ-UHFFFAOYSA-M thionine Chemical compound [Cl-].C1=CC(N)=CC2=[S+]C3=CC(N)=CC=C3N=C21 ANRHNWWPFJCPAZ-UHFFFAOYSA-M 0.000 description 1
- 230000000699 topical effect Effects 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 238000010361 transduction Methods 0.000 description 1
- 230000026683 transduction Effects 0.000 description 1
- 238000001890 transfection Methods 0.000 description 1
- 230000032258 transport Effects 0.000 description 1
- 238000011269 treatment regimen Methods 0.000 description 1
- CYRMSUTZVYGINF-UHFFFAOYSA-N trichlorofluoromethane Chemical compound FC(Cl)(Cl)Cl CYRMSUTZVYGINF-UHFFFAOYSA-N 0.000 description 1
- 229940029284 trichlorofluoromethane Drugs 0.000 description 1
- HRXKRNGNAMMEHJ-UHFFFAOYSA-K trisodium citrate Chemical compound [Na+].[Na+].[Na+].[O-]C(=O)CC(O)(CC([O-])=O)C([O-])=O HRXKRNGNAMMEHJ-UHFFFAOYSA-K 0.000 description 1
- 229940038773 trisodium citrate Drugs 0.000 description 1
- RYFMWSXOAZQYPI-UHFFFAOYSA-K trisodium phosphate Chemical compound [Na+].[Na+].[Na+].[O-]P([O-])([O-])=O RYFMWSXOAZQYPI-UHFFFAOYSA-K 0.000 description 1
- 230000001810 trypsinlike Effects 0.000 description 1
- 241001430294 unidentified retrovirus Species 0.000 description 1
- 210000001364 upper extremity Anatomy 0.000 description 1
- 235000015112 vegetable and seed oil Nutrition 0.000 description 1
- 239000008158 vegetable oil Substances 0.000 description 1
- 210000002845 virion Anatomy 0.000 description 1
- 238000001429 visible spectrum Methods 0.000 description 1
- 239000000080 wetting agent Substances 0.000 description 1
- 108010066753 yellow cameleon Proteins 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/85—Vectors or expression systems specially adapted for eukaryotic hosts for animal cells
- C12N15/86—Viral vectors
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K67/00—Rearing or breeding animals, not otherwise provided for; New or modified breeds of animals
- A01K67/027—New or modified breeds of vertebrates
- A01K67/0275—Genetically modified vertebrates, e.g. transgenic
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
- A61K38/16—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- A61K38/17—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- A61K38/177—Receptors; Cell surface antigens; Cell surface determinants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
- A61K48/005—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy characterised by an aspect of the 'active' part of the composition delivered, i.e. the nucleic acid delivered
- A61K48/0058—Nucleic acids adapted for tissue specific expression, e.g. having tissue specific promoters as part of a contruct
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/435—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from animals; from humans
- C07K14/705—Receptors; Cell surface antigens; Cell surface determinants
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/85—Vectors or expression systems specially adapted for eukaryotic hosts for animal cells
-
- A—HUMAN NECESSITIES
- A01—AGRICULTURE; FORESTRY; ANIMAL HUSBANDRY; HUNTING; TRAPPING; FISHING
- A01K—ANIMAL HUSBANDRY; AVICULTURE; APICULTURE; PISCICULTURE; FISHING; REARING OR BREEDING ANIMALS, NOT OTHERWISE PROVIDED FOR; NEW BREEDS OF ANIMALS
- A01K2267/00—Animals characterised by purpose
- A01K2267/03—Animal model, e.g. for test or diseases
- A01K2267/0393—Animal model comprising a reporter system for screening tests
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/14011—Parvoviridae
- C12N2750/14111—Dependovirus, e.g. adenoassociated viruses
- C12N2750/14141—Use of virus, viral particle or viral elements as a vector
- C12N2750/14143—Use of virus, viral particle or viral elements as a vector viral genome or elements thereof as genetic vector
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2830/00—Vector systems having a special element relevant for transcription
- C12N2830/008—Vector systems having a special element relevant for transcription cell type or tissue specific enhancer/promoter combination
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2830/00—Vector systems having a special element relevant for transcription
- C12N2830/15—Vector systems having a special element relevant for transcription chimeric enhancer/promoter combination
Landscapes
- Life Sciences & Earth Sciences (AREA)
- Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Genetics & Genomics (AREA)
- Chemical & Material Sciences (AREA)
- Biotechnology (AREA)
- Zoology (AREA)
- General Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- Molecular Biology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biomedical Technology (AREA)
- General Engineering & Computer Science (AREA)
- Veterinary Medicine (AREA)
- Wood Science & Technology (AREA)
- Animal Behavior & Ethology (AREA)
- Biochemistry (AREA)
- Medicinal Chemistry (AREA)
- Environmental Sciences (AREA)
- Biophysics (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Public Health (AREA)
- Plant Pathology (AREA)
- Physics & Mathematics (AREA)
- Microbiology (AREA)
- Animal Husbandry (AREA)
- Biodiversity & Conservation Biology (AREA)
- Gastroenterology & Hepatology (AREA)
- Cell Biology (AREA)
- Immunology (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Virology (AREA)
- Toxicology (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Medicinal Preparation (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Preparation Of Compounds By Using Micro-Organisms (AREA)
Abstract
Description
Claims (12)
- 영장류의 망막 색소 상피 세포에서 특이적으로 외인성 유전자를 발현하는 방법으로서, SEQ ID NO:1의 핵산 서열을 포함하거나 이로 구성되는, 또는 상기 SEQ ID NO:1의 서열과 적어도 80% 전반적 동일성을 갖는 적어도 400 bp의 핵산 서열로 구성되는 단리된 핵산 분자를 상기 영장류의 망막 색소 상피 세포에 전달하는 단계를 포함하며, 상기 단리된 핵산 분자는 상기 외인성 유전자를 코딩하는 핵산 서열이 상기 단리된 핵산 분자에 작동 가능하게 연결되는 경우 영장류의 망막 색소 상피 세포에서 특이적으로 외인성 유전자의 발현을 야기하는 방법.
- 제1항에 있어서, 상기 단리된 핵산 분자가 최소 프로모터, 예컨대 SEQ ID NO: 2의 최소 프로모터를 추가로 포함하는 방법.
- 제1항 또는 제2항에 있어서, 상기 단리된 핵산 분자가 발현 카세트의 일부인 방법.
- 제3항에 있어서, 상기 발현 카세트가 벡터의 일부인 방법.
- 제4항에 있어서, 상기 벡터가 바이러스 벡터인 방법.
- 영장류의 망막 색소 상피 세포에서 특이적으로 외인성 유전자를 발현하기 위한, SEQ ID NO:1의 핵산 서열을 포함하거나 이로 구성되는, 또는 상기 SEQ ID NO:1의 서열과 적어도 80% 전반적 동일성을 갖는 적어도 400 bp의 핵산 서열로 구성되는 단리된 핵산 분자의 용도로서, 상기 단리된 핵산 분자는 상기 외인성 유전자를 코딩하는 핵산 서열이 상기 단리된 핵산 분자에 작동 가능하게 연결되는 경우 영장류의 망막 색소 상피 세포에서 특이적으로 외인성 유전자의 발현을 야기하는 용도.
- 제6항에 있어서, 단리된 핵산 분자가 최소 프로모터, 예컨대 SEQ ID NO: 2의 최소 프로모터를 추가로 포함하는 용도.
- 제6항 또는 제7항에 있어서, 상기 단리된 핵산 분자가 발현 카세트의 일부인 용도.
- 제8항에 있어서, 상기 발현 카세트가 벡터의 일부인 용도.
- 제9항에 있어서, 상기 벡터가 바이러스 벡터인 용도.
- 망막 색소 상피와 연관된 질병의 치료에서 사용하기 위한, SEQ ID NO: 1의 핵산 서열을 포함하거나 이로 구성되는, 또는 상기 SEQ ID NO: 1의 서열과 적어도 80% 전반적 동일성을 갖는 적어도 400 bp의 핵산 서열로 구성되는 단리된 핵산 분자로서, 상기 단리된 핵산 분자는 상기 외인성 유전자를 코딩하는 핵산 서열이 상기 단리된 핵산 분자에 작동 가능하게 연결되는 경우, 영장류의 망막 색소 상피 세포에서 특이적으로 외인성 유전자의 발현을 야기하는 단리된 핵산 분자.
- 제11항에 있어서, 상기 망막 색소 상피와 연관된 질병이 연령-관련 황반 변성, 색소 망막염, 당뇨병성 망막병증 및 망막 색소 상피 비대로 구성되는 군으로부터 선택되는, 사용하기 위한 단리된 핵산 분자.
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| EP17201828 | 2017-11-15 | ||
| EP17201828.5 | 2017-11-15 | ||
| PCT/IB2018/059015 WO2019097454A1 (en) | 2017-11-15 | 2018-11-15 | Primate retinal pigment epithelium cell-specific promoter |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| KR20200084028A true KR20200084028A (ko) | 2020-07-09 |
Family
ID=60331462
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020207016484A Ceased KR20200084028A (ko) | 2017-11-15 | 2018-11-15 | 영장류 망막 색소 상피 세포-특이적 프로모터 |
Country Status (14)
| Country | Link |
|---|---|
| US (1) | US11739349B2 (ko) |
| EP (1) | EP3709798B1 (ko) |
| JP (2) | JP7348176B2 (ko) |
| KR (1) | KR20200084028A (ko) |
| CN (1) | CN111726985B (ko) |
| AU (1) | AU2018369975B2 (ko) |
| BR (1) | BR112020009579A2 (ko) |
| CA (1) | CA3082080A1 (ko) |
| CR (1) | CR20200210A (ko) |
| IL (1) | IL274460A (ko) |
| MX (1) | MX2020005033A (ko) |
| PH (1) | PH12020550757A1 (ko) |
| SG (1) | SG11202004205YA (ko) |
| WO (1) | WO2019097454A1 (ko) |
Families Citing this family (15)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20100136550A (ko) | 2008-04-18 | 2010-12-28 | 노파르티스 포르슝스티프퉁 쯔바이크니덜라쑹 프리드리히 미셔 인스티튜트 포 바이오메디칼 리서치 | 실명을 치료하기 위한 신규의 치료 도구 및 방법 |
| ES2747433T3 (es) | 2015-04-30 | 2020-03-10 | Friedrich Miescher Institute For Biomedical Res | Promotor para la expresión específica de genes en células de Müller |
| CN108350463B (zh) | 2015-09-15 | 2022-06-24 | 弗里德里克·米谢尔生物医学研究所 | 通过靶向光受体治疗失明的新型治疗工具和方法 |
| ES2881782T3 (es) | 2015-10-14 | 2021-11-30 | Friedrich Miescher Institute For Biomedical Res | Promotor para la expresión específica de genes en células endoteliales retinianas. |
| US10995344B2 (en) | 2015-12-03 | 2021-05-04 | Friedrich Miescher Institute For Biomedical Research | SYNP159, a promoter for the specific expression of genes in rod photoreceptors |
| US10994026B2 (en) | 2015-12-03 | 2021-05-04 | Friedrich Miescher Institute For Biomedical Research | SynP160, a promoter for the specific expression of genes in rod photoreceptors |
| CN108472390B (zh) | 2015-12-03 | 2022-04-15 | 弗里德里克·米谢尔生物医学研究所 | SynP162,用于基因在视杆光感受器中特异性表达的启动子 |
| RU2758211C2 (ru) | 2015-12-03 | 2021-10-26 | Фридрих Мишер Инститьют Фор Байомедикал Рисерч | Synp161, промотор для специфической экспрессии генов в палочковых фоторецепторах |
| WO2018083607A1 (en) | 2016-11-02 | 2018-05-11 | Friedrich Miescher Institute For Biomedical Research | Synp198, a promoter for the specific expression of genes in direction selective retinal ganglion cells |
| WO2018146588A1 (en) | 2017-02-08 | 2018-08-16 | Friedrich Miescher Institute For Biomedical Research | Synp88, a promoter for the specific expression of genes in retinal ganglion cells |
| KR20200084028A (ko) | 2017-11-15 | 2020-07-09 | 프리드리히 미셔 인스티튜트 포 바이오메디칼 리서치 | 영장류 망막 색소 상피 세포-특이적 프로모터 |
| WO2019106035A1 (en) | 2017-11-30 | 2019-06-06 | Friedrich Miescher Institute For Biomedical Research | Synpiii, a promoter for the specific expression of genes in retinal pigment epithelium |
| JP7390290B2 (ja) * | 2017-11-30 | 2023-12-01 | フリードリッヒ ミーシェー インスティトゥート フォー バイオメディカル リサーチ | 霊長類網膜色素上皮細胞特異的プロモーターSynP61 |
| CN114045264B (zh) * | 2021-11-16 | 2024-05-14 | 珠海中科先进技术研究院有限公司 | 一种获取驱动元件的方法 |
| EP4568710A1 (en) * | 2022-08-11 | 2025-06-18 | Institute Of Molecular And Clinical Ophthalmology Basel (Iob) | Human cone photoreceptor optogenetic constructs |
Family Cites Families (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6582908B2 (en) | 1990-12-06 | 2003-06-24 | Affymetrix, Inc. | Oligonucleotides |
| US5474796A (en) | 1991-09-04 | 1995-12-12 | Protogene Laboratories, Inc. | Method and apparatus for conducting an array of chemical reactions on a support surface |
| US20020052332A1 (en) * | 2000-04-11 | 2002-05-02 | The Board Of Regents, University Of Texas System | Methods of inhibiting retinal pigment epithelial cell degeneration, stable cell lines, and uses |
| US20040208847A1 (en) * | 2003-03-28 | 2004-10-21 | Fabienne Rolling | Method and vectors for selectively transducing retinal pigment epithelium cells |
| MX375061B (es) | 2004-01-23 | 2025-03-06 | Harvard College | Modalidades mejoradas para el tratamiento de enfermedades degenerativas de la retina. |
| US7309487B2 (en) * | 2004-02-09 | 2007-12-18 | George Inana | Methods and compositions for detecting and treating retinal diseases |
| EP1891976A1 (en) | 2006-08-23 | 2008-02-27 | Novartis Forschungsstiftung, Zweigniederlassung Friedrich Miescher Institute for Biomedical Research | Use of light sensitive genes |
| KR20100136550A (ko) | 2008-04-18 | 2010-12-28 | 노파르티스 포르슝스티프퉁 쯔바이크니덜라쑹 프리드리히 미셔 인스티튜트 포 바이오메디칼 리서치 | 실명을 치료하기 위한 신규의 치료 도구 및 방법 |
| WO2009134681A2 (en) * | 2008-04-30 | 2009-11-05 | The Trustees Of The University Of Pennsylvania | Aav7 viral vectors for targeted delivery of rpe cells |
| CN101747425B (zh) * | 2008-12-16 | 2012-03-28 | 清华大学 | 与视网膜光反应相关的蛋白及其编码基因与应用 |
| WO2011034947A2 (en) * | 2009-09-15 | 2011-03-24 | University Of Washington | Reagents and methods for modulating cone photoreceptor activity |
| AU2011232435B2 (en) | 2010-03-23 | 2016-01-28 | Intrexon Corporation | Vectors conditionally expressing therapeutic proteins, host cells comprising the vectors, and uses thereof |
| US20140099284A1 (en) * | 2010-10-15 | 2014-04-10 | Eos Neuroscience, Inc | Modulation neural pathways |
| WO2014033095A1 (en) | 2012-08-27 | 2014-03-06 | Friedrich Miescher Institute For Biomedical Research | Retinal off circuit-specific promoter |
| EP3104895A1 (en) | 2014-02-10 | 2016-12-21 | Friedrich Miescher Institute for Biomedical Research | Aii retinal amacrine cell-specific promoter |
| EP3105334B1 (en) | 2014-02-11 | 2019-07-03 | Friedrich Miescher Institute for Biomedical Research | Müller cell-specific promoter |
| PL3137497T5 (pl) * | 2014-05-02 | 2025-06-09 | Genzyme Corporation | Wektory AAV do terapii genowej siatkówki i OUN |
| ES2747433T3 (es) | 2015-04-30 | 2020-03-10 | Friedrich Miescher Institute For Biomedical Res | Promotor para la expresión específica de genes en células de Müller |
| CN108350463B (zh) | 2015-09-15 | 2022-06-24 | 弗里德里克·米谢尔生物医学研究所 | 通过靶向光受体治疗失明的新型治疗工具和方法 |
| ES2881782T3 (es) | 2015-10-14 | 2021-11-30 | Friedrich Miescher Institute For Biomedical Res | Promotor para la expresión específica de genes en células endoteliales retinianas. |
| US10995344B2 (en) | 2015-12-03 | 2021-05-04 | Friedrich Miescher Institute For Biomedical Research | SYNP159, a promoter for the specific expression of genes in rod photoreceptors |
| US10994026B2 (en) | 2015-12-03 | 2021-05-04 | Friedrich Miescher Institute For Biomedical Research | SynP160, a promoter for the specific expression of genes in rod photoreceptors |
| CN108472390B (zh) | 2015-12-03 | 2022-04-15 | 弗里德里克·米谢尔生物医学研究所 | SynP162,用于基因在视杆光感受器中特异性表达的启动子 |
| RU2758211C2 (ru) | 2015-12-03 | 2021-10-26 | Фридрих Мишер Инститьют Фор Байомедикал Рисерч | Synp161, промотор для специфической экспрессии генов в палочковых фоторецепторах |
| ES2872798T3 (es) | 2015-12-04 | 2021-11-02 | Univ Sorbonne | Promotores y usos de los mismos |
| CN107287238B (zh) * | 2016-04-11 | 2020-10-16 | 厦门继景生物技术有限责任公司 | 一种基因载体及其用于治疗雷柏氏先天性黑矇2型疾病的基因治疗药物 |
| EP3458476A1 (en) | 2016-05-17 | 2019-03-27 | Friedrich Miescher Institute for Biomedical Research | Novel therapeutic tools and methods for treating blindness |
| WO2018083607A1 (en) | 2016-11-02 | 2018-05-11 | Friedrich Miescher Institute For Biomedical Research | Synp198, a promoter for the specific expression of genes in direction selective retinal ganglion cells |
| JP7071361B2 (ja) | 2016-12-01 | 2022-05-18 | フリードリッヒ ミーシェー インスティトゥート フォー バイオメディカル リサーチ | インターニューロン中の遺伝子の特異的発現のためのプロモーターsynp107 |
| EP3548094A1 (en) | 2016-12-01 | 2019-10-09 | Friedrich Miescher Institute for Biomedical Research | Synpi, a promoter for the specific expression of genes in interneurons |
| WO2018146588A1 (en) | 2017-02-08 | 2018-08-16 | Friedrich Miescher Institute For Biomedical Research | Synp88, a promoter for the specific expression of genes in retinal ganglion cells |
| KR20200084028A (ko) | 2017-11-15 | 2020-07-09 | 프리드리히 미셔 인스티튜트 포 바이오메디칼 리서치 | 영장류 망막 색소 상피 세포-특이적 프로모터 |
| JP7390290B2 (ja) | 2017-11-30 | 2023-12-01 | フリードリッヒ ミーシェー インスティトゥート フォー バイオメディカル リサーチ | 霊長類網膜色素上皮細胞特異的プロモーターSynP61 |
| WO2019106035A1 (en) | 2017-11-30 | 2019-06-06 | Friedrich Miescher Institute For Biomedical Research | Synpiii, a promoter for the specific expression of genes in retinal pigment epithelium |
| JP2022505516A (ja) | 2018-10-25 | 2022-01-14 | フリードリッヒ ミーシェー インスティトゥート フォー バイオメディカル リサーチ | 網膜神経節細胞中の遺伝子の特異的発現のためのプロモーターSynP17(ProB1) |
| CN112930201A (zh) | 2018-10-25 | 2021-06-08 | 弗里德里克·米谢尔生物医学研究所 | 用于视网膜神经节细胞中特异表达基因的启动子SynP78(ProA27) |
| EP3870709A1 (en) | 2018-10-25 | 2021-09-01 | Friedrich Miescher Institute for Biomedical Research | Synp27 (prob12), a promoter for the specific expression of genes in protoplasmic astrocytes |
| WO2020084539A1 (en) | 2018-10-25 | 2020-04-30 | Friedrich Miescher Institute For Biomedical Research | Synp57 (proa14), a promoter for the specific expression of genes in photoreceptors |
| EP3870242A1 (en) | 2018-10-25 | 2021-09-01 | Friedrich Miescher Institute for Biomedical Research | Synp151 (proc29), a promoter for the specific expression of genes in retinal ganglion cells |
| JP2022512780A (ja) | 2018-10-25 | 2022-02-07 | フリードリッヒ ミーシェー インスティトゥート フォー バイオメディカル リサーチ | 網膜神経節細胞中の遺伝子の特異的発現のためのプロモーターSynP194(ProB15) |
| WO2020152623A1 (en) | 2019-01-24 | 2020-07-30 | Friedrich Miescher Institute For Biomedical Research | Synp5 (proa9), a promoter for the specific expression of genes in retinal ganglion cells |
| WO2020152626A1 (en) | 2019-01-24 | 2020-07-30 | Friedrich Miescher Institute For Biomedical Research | Synp166 (proa36), a promoter for the specific expression of genes in photoreceptors |
| US20220119807A1 (en) | 2019-01-24 | 2022-04-21 | Friedrich Miescher Institute For Biomedical Research | Synp35 (proc8), a promoter for the specific expression of genes in retinal ganglion cells |
| EP3914300A1 (en) | 2019-01-24 | 2021-12-01 | Friedrich Miescher Institute for Biomedical Research | Synp66 (proa21), a promoter for the specific expression of genes in retinal ganglion cells |
| JP2022520875A (ja) | 2019-02-25 | 2022-04-01 | ノバルティス アーゲー | Biettiクリスタリン網膜症を治療するための組成物及び方法 |
| BR112021016501A2 (pt) | 2019-02-25 | 2021-10-26 | Novartis Ag | Composições e métodos para tratar distrofia cristalina de bietti |
-
2018
- 2018-11-15 KR KR1020207016484A patent/KR20200084028A/ko not_active Ceased
- 2018-11-15 WO PCT/IB2018/059015 patent/WO2019097454A1/en not_active Ceased
- 2018-11-15 EP EP18808513.8A patent/EP3709798B1/en active Active
- 2018-11-15 SG SG11202004205YA patent/SG11202004205YA/en unknown
- 2018-11-15 MX MX2020005033A patent/MX2020005033A/es unknown
- 2018-11-15 BR BR112020009579-6A patent/BR112020009579A2/pt not_active Application Discontinuation
- 2018-11-15 AU AU2018369975A patent/AU2018369975B2/en not_active Ceased
- 2018-11-15 CA CA3082080A patent/CA3082080A1/en active Pending
- 2018-11-15 CN CN201880074227.7A patent/CN111726985B/zh not_active Expired - Fee Related
- 2018-11-15 US US16/764,306 patent/US11739349B2/en active Active
- 2018-11-15 CR CR20200210A patent/CR20200210A/es unknown
- 2018-11-15 JP JP2020526394A patent/JP7348176B2/ja active Active
-
2020
- 2020-05-05 IL IL274460A patent/IL274460A/en unknown
- 2020-05-13 PH PH12020550757A patent/PH12020550757A1/en unknown
-
2023
- 2023-02-22 JP JP2023025663A patent/JP2023075139A/ja active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| AU2018369975B2 (en) | 2022-05-19 |
| EP3709798A1 (en) | 2020-09-23 |
| EP3709798B1 (en) | 2024-04-17 |
| PH12020550757A1 (en) | 2021-05-10 |
| CR20200210A (es) | 2020-09-23 |
| SG11202004205YA (en) | 2020-06-29 |
| MX2020005033A (es) | 2020-10-28 |
| JP7348176B2 (ja) | 2023-09-20 |
| CN111726985B (zh) | 2022-06-10 |
| WO2019097454A1 (en) | 2019-05-23 |
| CA3082080A1 (en) | 2019-05-23 |
| RU2020119176A3 (ko) | 2022-01-26 |
| JP2023075139A (ja) | 2023-05-30 |
| US11739349B2 (en) | 2023-08-29 |
| AU2018369975A1 (en) | 2020-05-28 |
| IL274460A (en) | 2020-06-30 |
| JP2021502813A (ja) | 2021-02-04 |
| BR112020009579A2 (pt) | 2020-10-13 |
| US20210054408A1 (en) | 2021-02-25 |
| RU2020119176A (ru) | 2021-12-16 |
| CN111726985A (zh) | 2020-09-29 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR20200084028A (ko) | 영장류 망막 색소 상피 세포-특이적 프로모터 | |
| AU2018376544B2 (en) | SynP61, a primate retinal pigment epithelium cell-specific promoter | |
| RU2766353C2 (ru) | Synp198, промотор для специфической экспрессии генов в ганглионарных клетках сетчатки, избирательных в отношении направления | |
| US9999685B2 (en) | AII retinal amacrine cell-specific promoter | |
| EP3105334B1 (en) | Müller cell-specific promoter | |
| EP3383438B1 (en) | Synp159, a promoter for the specific expression of genes in rod photoreceptors | |
| EP3289090B1 (en) | Promoter for the specific expression of genes in müller cells | |
| KR20200091432A (ko) | 망막 색소 상피에서의 유전자의 특이적 발현을 위한 프로모터 SynPIII | |
| EP3579884A1 (en) | Synp88, a promoter for the specific expression of genes in retinal ganglion cells | |
| KR20180084136A (ko) | 간상 광수용체에서의 유전자의 특이적 발현을 위한 프로모터 SynP161 | |
| EP3914721A1 (en) | Synp35 (proc8), a promoter for the specific expression of genes in retinal ganglion cells | |
| WO2017093934A1 (en) | Synp160, a promoter for the specific expression of genes in rod photoreceptors | |
| WO2014033095A1 (en) | Retinal off circuit-specific promoter | |
| WO2017093936A1 (en) | Synp162, a promoter for the specific expression of genes in rod photoreceptors | |
| EP3007730A1 (en) | Retinal on bipolar cells-specific artificial promoter | |
| RU2773940C2 (ru) | Промотор, специфический в отношении клеток пигментного эпителия сетчатки приматов |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
Patent event date: 20200609 Patent event code: PA01051R01D Comment text: International Patent Application |
|
| PG1501 | Laying open of application | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20211027 Comment text: Request for Examination of Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20240422 Patent event code: PE09021S01D |
|
| E601 | Decision to refuse application | ||
| PE0601 | Decision on rejection of patent |
Patent event date: 20240708 Comment text: Decision to Refuse Application Patent event code: PE06012S01D |