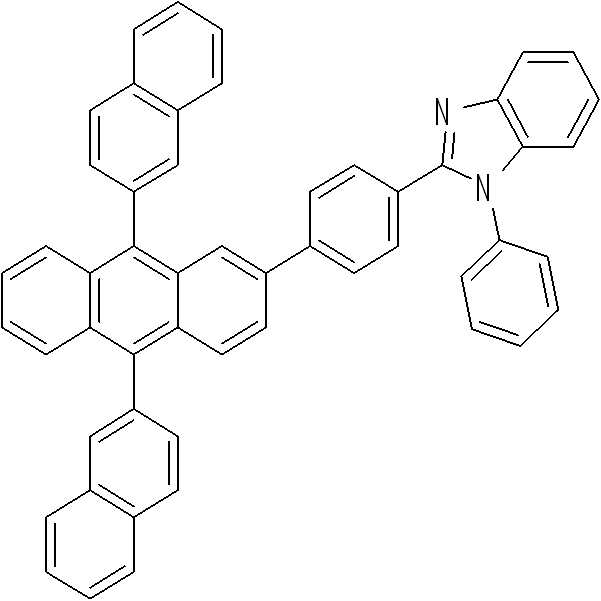
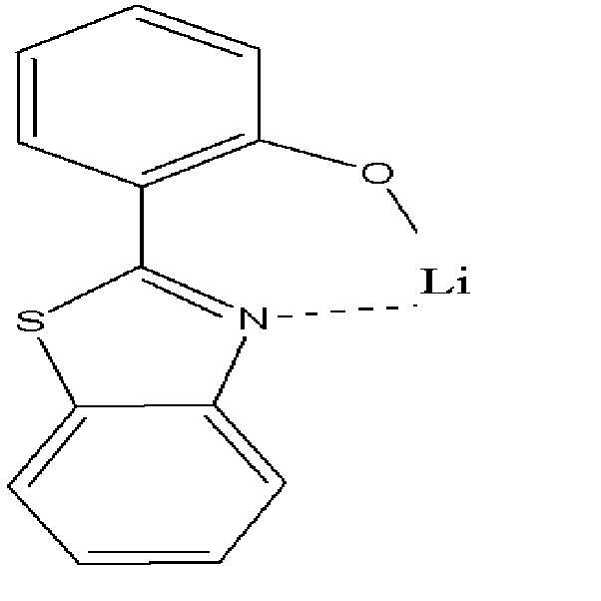
KR101994837B1 - 신규 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 - Google Patents
신규 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 Download PDFInfo
- Publication number
- KR101994837B1 KR101994837B1 KR1020120081965A KR20120081965A KR101994837B1 KR 101994837 B1 KR101994837 B1 KR 101994837B1 KR 1020120081965 A KR1020120081965 A KR 1020120081965A KR 20120081965 A KR20120081965 A KR 20120081965A KR 101994837 B1 KR101994837 B1 KR 101994837B1
- Authority
- KR
- South Korea
- Prior art keywords
- group
- layer
- substituted
- light emitting
- compound
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- -1 heterocycle compound Chemical class 0.000 title claims description 28
- 150000002391 heterocyclic compounds Chemical class 0.000 claims abstract description 14
- 239000000126 substance Substances 0.000 claims abstract description 6
- 239000010410 layer Substances 0.000 claims description 183
- 150000001875 compounds Chemical class 0.000 claims description 105
- 125000004432 carbon atom Chemical group C* 0.000 claims description 68
- 239000000463 material Substances 0.000 claims description 50
- 238000000034 method Methods 0.000 claims description 48
- 238000002347 injection Methods 0.000 claims description 41
- 239000007924 injection Substances 0.000 claims description 41
- 239000002019 doping agent Substances 0.000 claims description 35
- 239000012044 organic layer Substances 0.000 claims description 32
- 125000003118 aryl group Chemical group 0.000 claims description 27
- 125000000217 alkyl group Chemical group 0.000 claims description 26
- 239000002346 layers by function Substances 0.000 claims description 26
- 125000004093 cyano group Chemical group *C#N 0.000 claims description 16
- YZCKVEUIGOORGS-OUBTZVSYSA-N Deuterium Chemical compound [2H] YZCKVEUIGOORGS-OUBTZVSYSA-N 0.000 claims description 14
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 claims description 13
- MWPLVEDNUUSJAV-UHFFFAOYSA-N anthracene Natural products C1=CC=CC2=CC3=CC=CC=C3C=C21 MWPLVEDNUUSJAV-UHFFFAOYSA-N 0.000 claims description 13
- 125000005843 halogen group Chemical group 0.000 claims description 12
- 125000001072 heteroaryl group Chemical group 0.000 claims description 12
- 125000004435 hydrogen atom Chemical group [H]* 0.000 claims description 11
- 229910052805 deuterium Inorganic materials 0.000 claims description 9
- 229910052739 hydrogen Inorganic materials 0.000 claims description 9
- 239000001257 hydrogen Substances 0.000 claims description 9
- 125000002887 hydroxy group Chemical group [H]O* 0.000 claims description 9
- 125000003277 amino group Chemical group 0.000 claims description 8
- 125000003178 carboxy group Chemical group [H]OC(*)=O 0.000 claims description 8
- 125000000449 nitro group Chemical group [O-][N+](*)=O 0.000 claims description 8
- 125000003367 polycyclic group Chemical group 0.000 claims description 8
- 150000002431 hydrogen Chemical class 0.000 claims description 6
- YLQBMQCUIZJEEH-UHFFFAOYSA-N Furan Chemical group C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 5
- 150000004696 coordination complex Chemical class 0.000 claims description 5
- 125000005647 linker group Chemical group 0.000 claims description 4
- IMKMFBIYHXBKRX-UHFFFAOYSA-M lithium;quinoline-2-carboxylate Chemical group [Li+].C1=CC=CC2=NC(C(=O)[O-])=CC=C21 IMKMFBIYHXBKRX-UHFFFAOYSA-M 0.000 claims description 4
- 229910044991 metal oxide Inorganic materials 0.000 claims description 4
- 150000004706 metal oxides Chemical class 0.000 claims description 4
- 150000002894 organic compounds Chemical class 0.000 claims description 4
- 150000003233 pyrroles Chemical group 0.000 claims description 4
- 150000004059 quinone derivatives Chemical group 0.000 claims description 4
- 239000010409 thin film Substances 0.000 claims description 4
- IANQTJSKSUMEQM-UHFFFAOYSA-N 1-benzofuran Chemical group C1=CC=C2OC=CC2=C1 IANQTJSKSUMEQM-UHFFFAOYSA-N 0.000 claims description 3
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 claims description 3
- UFHFLCQGNIYNRP-UHFFFAOYSA-N Hydrogen Chemical compound [H][H] UFHFLCQGNIYNRP-UHFFFAOYSA-N 0.000 claims description 3
- 230000015572 biosynthetic process Effects 0.000 description 32
- 238000006243 chemical reaction Methods 0.000 description 31
- 239000000543 intermediate Substances 0.000 description 27
- 125000001424 substituent group Chemical group 0.000 description 22
- 238000003786 synthesis reaction Methods 0.000 description 22
- 230000005525 hole transport Effects 0.000 description 19
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 16
- 150000003839 salts Chemical class 0.000 description 15
- CSNNHWWHGAXBCP-UHFFFAOYSA-L Magnesium sulfate Chemical compound [Mg+2].[O-][S+2]([O-])([O-])[O-] CSNNHWWHGAXBCP-UHFFFAOYSA-L 0.000 description 14
- 238000004895 liquid chromatography mass spectrometry Methods 0.000 description 13
- 125000001997 phenyl group Chemical group [H]C1=C([H])C([H])=C(*)C([H])=C1[H] 0.000 description 13
- OAKJQQAXSVQMHS-UHFFFAOYSA-N Hydrazine Chemical compound NN OAKJQQAXSVQMHS-UHFFFAOYSA-N 0.000 description 12
- 238000001771 vacuum deposition Methods 0.000 description 12
- 125000001624 naphthyl group Chemical group 0.000 description 11
- 229910052757 nitrogen Inorganic materials 0.000 description 10
- 239000002904 solvent Substances 0.000 description 10
- 239000000758 substrate Substances 0.000 description 10
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 10
- 125000005428 anthryl group Chemical group [H]C1=C([H])C([H])=C2C([H])=C3C(*)=C([H])C([H])=C([H])C3=C([H])C2=C1[H] 0.000 description 9
- 230000000903 blocking effect Effects 0.000 description 9
- 238000004528 spin coating Methods 0.000 description 9
- VFUDMQLBKNMONU-UHFFFAOYSA-N 9-[4-(4-carbazol-9-ylphenyl)phenyl]carbazole Chemical compound C12=CC=CC=C2C2=CC=CC=C2N1C1=CC=C(C=2C=CC(=CC=2)N2C3=CC=CC=C3C3=CC=CC=C32)C=C1 VFUDMQLBKNMONU-UHFFFAOYSA-N 0.000 description 8
- NBIIXXVUZAFLBC-UHFFFAOYSA-N Phosphoric acid Chemical compound OP(O)(O)=O NBIIXXVUZAFLBC-UHFFFAOYSA-N 0.000 description 8
- 239000000872 buffer Substances 0.000 description 8
- 238000000151 deposition Methods 0.000 description 8
- 230000000052 comparative effect Effects 0.000 description 7
- 125000003983 fluorenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3CC12)* 0.000 description 7
- 229910052943 magnesium sulfate Inorganic materials 0.000 description 7
- 235000019341 magnesium sulphate Nutrition 0.000 description 7
- 238000010898 silica gel chromatography Methods 0.000 description 7
- 125000000008 (C1-C10) alkyl group Chemical group 0.000 description 6
- UHXOHPVVEHBKKT-UHFFFAOYSA-N 1-(2,2-diphenylethenyl)-4-[4-(2,2-diphenylethenyl)phenyl]benzene Chemical compound C=1C=C(C=2C=CC(C=C(C=3C=CC=CC=3)C=3C=CC=CC=3)=CC=2)C=CC=1C=C(C=1C=CC=CC=1)C1=CC=CC=C1 UHXOHPVVEHBKKT-UHFFFAOYSA-N 0.000 description 6
- ZMANZCXQSJIPKH-UHFFFAOYSA-N Triethylamine Chemical compound CCN(CC)CC ZMANZCXQSJIPKH-UHFFFAOYSA-N 0.000 description 6
- 125000003739 carbamimidoyl group Chemical group C(N)(=N)* 0.000 description 6
- 238000000576 coating method Methods 0.000 description 6
- 150000007857 hydrazones Chemical class 0.000 description 6
- 239000000243 solution Substances 0.000 description 6
- 230000032258 transport Effects 0.000 description 6
- 238000005266 casting Methods 0.000 description 5
- 239000011248 coating agent Substances 0.000 description 5
- 230000008021 deposition Effects 0.000 description 5
- 229910052751 metal Inorganic materials 0.000 description 5
- 239000002184 metal Substances 0.000 description 5
- 125000001725 pyrenyl group Chemical group 0.000 description 5
- 125000000542 sulfonic acid group Chemical group 0.000 description 5
- 238000005481 NMR spectroscopy Methods 0.000 description 4
- 125000003342 alkenyl group Chemical group 0.000 description 4
- 229910000147 aluminium phosphate Inorganic materials 0.000 description 4
- 125000005110 aryl thio group Chemical group 0.000 description 4
- 125000004104 aryloxy group Chemical group 0.000 description 4
- 239000011575 calcium Substances 0.000 description 4
- 239000011777 magnesium Substances 0.000 description 4
- 239000000203 mixture Substances 0.000 description 4
- QIQXTHQIDYTFRH-UHFFFAOYSA-N octadecanoic acid Chemical compound CCCCCCCCCCCCCCCCCC(O)=O QIQXTHQIDYTFRH-UHFFFAOYSA-N 0.000 description 4
- 125000000027 (C1-C10) alkoxy group Chemical group 0.000 description 3
- FCEHBMOGCRZNNI-UHFFFAOYSA-N 1-benzothiophene Chemical group C1=CC=C2SC=CC2=C1 FCEHBMOGCRZNNI-UHFFFAOYSA-N 0.000 description 3
- MAGFQRLKWCCTQJ-UHFFFAOYSA-M 4-ethenylbenzenesulfonate Chemical compound [O-]S(=O)(=O)C1=CC=C(C=C)C=C1 MAGFQRLKWCCTQJ-UHFFFAOYSA-M 0.000 description 3
- VIZUPBYFLORCRA-UHFFFAOYSA-N 9,10-dinaphthalen-2-ylanthracene Chemical compound C12=CC=CC=C2C(C2=CC3=CC=CC=C3C=C2)=C(C=CC=C2)C2=C1C1=CC=C(C=CC=C2)C2=C1 VIZUPBYFLORCRA-UHFFFAOYSA-N 0.000 description 3
- SIKJAQJRHWYJAI-UHFFFAOYSA-N Indole Chemical group C1=CC=C2NC=CC2=C1 SIKJAQJRHWYJAI-UHFFFAOYSA-N 0.000 description 3
- KFZMGEQAYNKOFK-UHFFFAOYSA-N Isopropanol Chemical compound CC(C)O KFZMGEQAYNKOFK-UHFFFAOYSA-N 0.000 description 3
- YTPLMLYBLZKORZ-UHFFFAOYSA-N Thiophene Chemical group C=1C=CSC=1 YTPLMLYBLZKORZ-UHFFFAOYSA-N 0.000 description 3
- 125000003545 alkoxy group Chemical group 0.000 description 3
- 125000000304 alkynyl group Chemical group 0.000 description 3
- 125000006267 biphenyl group Chemical group 0.000 description 3
- 125000001495 ethyl group Chemical group [H]C([H])([H])C([H])([H])* 0.000 description 3
- 239000011521 glass Substances 0.000 description 3
- 125000002496 methyl group Chemical group [H]C([H])([H])* 0.000 description 3
- 125000001792 phenanthrenyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3C=CC12)* 0.000 description 3
- 229920003227 poly(N-vinyl carbazole) Polymers 0.000 description 3
- 229920000767 polyaniline Polymers 0.000 description 3
- 125000001436 propyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])[H] 0.000 description 3
- 125000004076 pyridyl group Chemical group 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 125000006746 (C1-C60) alkoxy group Chemical group 0.000 description 2
- 125000006745 (C2-C60) alkynyl group Chemical group 0.000 description 2
- 0 *c(c-1c2*c3ccccc-13)c(*)c1c2c(*)c(*)c2c(*)c(*)c(CCI)c(*)c12 Chemical compound *c(c-1c2*c3ccccc-13)c(*)c1c2c(*)c(*)c2c(*)c(*)c(CCI)c(*)c12 0.000 description 2
- WBIQQQGBSDOWNP-UHFFFAOYSA-N 2-dodecylbenzenesulfonic acid Chemical compound CCCCCCCCCCCCC1=CC=CC=C1S(O)(=O)=O WBIQQQGBSDOWNP-UHFFFAOYSA-N 0.000 description 2
- VQGHOUODWALEFC-UHFFFAOYSA-N 2-phenylpyridine Chemical group C1=CC=CC=C1C1=CC=CC=N1 VQGHOUODWALEFC-UHFFFAOYSA-N 0.000 description 2
- 229910001148 Al-Li alloy Inorganic materials 0.000 description 2
- OYPRJOBELJOOCE-UHFFFAOYSA-N Calcium Chemical compound [Ca] OYPRJOBELJOOCE-UHFFFAOYSA-N 0.000 description 2
- UEXCJVNBTNXOEH-UHFFFAOYSA-N Ethynylbenzene Chemical group C#CC1=CC=CC=C1 UEXCJVNBTNXOEH-UHFFFAOYSA-N 0.000 description 2
- FYYHWMGAXLPEAU-UHFFFAOYSA-N Magnesium Chemical compound [Mg] FYYHWMGAXLPEAU-UHFFFAOYSA-N 0.000 description 2
- XQVWYOYUZDUNRW-UHFFFAOYSA-N N-Phenyl-1-naphthylamine Chemical compound C=1C=CC2=CC=CC=C2C=1NC1=CC=CC=C1 XQVWYOYUZDUNRW-UHFFFAOYSA-N 0.000 description 2
- 229920001609 Poly(3,4-ethylenedioxythiophene) Polymers 0.000 description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 2
- HEDRZPFGACZZDS-MICDWDOJSA-N Trichloro(2H)methane Chemical compound [2H]C(Cl)(Cl)Cl HEDRZPFGACZZDS-MICDWDOJSA-N 0.000 description 2
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 2
- JHYLKGDXMUDNEO-UHFFFAOYSA-N [Mg].[In] Chemical compound [Mg].[In] JHYLKGDXMUDNEO-UHFFFAOYSA-N 0.000 description 2
- 125000005037 alkyl phenyl group Chemical group 0.000 description 2
- 229910052782 aluminium Inorganic materials 0.000 description 2
- XAGFODPZIPBFFR-UHFFFAOYSA-N aluminium Chemical compound [Al] XAGFODPZIPBFFR-UHFFFAOYSA-N 0.000 description 2
- 125000000732 arylene group Chemical group 0.000 description 2
- 229910052791 calcium Inorganic materials 0.000 description 2
- 125000000609 carbazolyl group Chemical group C1(=CC=CC=2C3=CC=CC=C3NC12)* 0.000 description 2
- 229910052799 carbon Inorganic materials 0.000 description 2
- 125000000753 cycloalkyl group Chemical group 0.000 description 2
- 239000012153 distilled water Substances 0.000 description 2
- 229940060296 dodecylbenzenesulfonic acid Drugs 0.000 description 2
- 239000007772 electrode material Substances 0.000 description 2
- 230000009477 glass transition Effects 0.000 description 2
- 125000000623 heterocyclic group Chemical group 0.000 description 2
- 125000004051 hexyl group Chemical group [H]C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])* 0.000 description 2
- 230000006872 improvement Effects 0.000 description 2
- 238000004020 luminiscence type Methods 0.000 description 2
- 229910052749 magnesium Inorganic materials 0.000 description 2
- 239000011159 matrix material Substances 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- IBHBKWKFFTZAHE-UHFFFAOYSA-N n-[4-[4-(n-naphthalen-1-ylanilino)phenyl]phenyl]-n-phenylnaphthalen-1-amine Chemical compound C1=CC=CC=C1N(C=1C2=CC=CC=C2C=CC=1)C1=CC=C(C=2C=CC(=CC=2)N(C=2C=CC=CC=2)C=2C3=CC=CC=C3C=CC=2)C=C1 IBHBKWKFFTZAHE-UHFFFAOYSA-N 0.000 description 2
- 125000004957 naphthylene group Chemical group 0.000 description 2
- 239000012299 nitrogen atmosphere Substances 0.000 description 2
- 125000001147 pentyl group Chemical group C(CCCC)* 0.000 description 2
- 125000005560 phenanthrenylene group Chemical group 0.000 description 2
- 125000000843 phenylene group Chemical group C1(=C(C=CC=C1)*)* 0.000 description 2
- 229910052698 phosphorus Inorganic materials 0.000 description 2
- 125000005548 pyrenylene group Chemical group 0.000 description 2
- 238000010992 reflux Methods 0.000 description 2
- 230000004044 response Effects 0.000 description 2
- 239000002356 single layer Substances 0.000 description 2
- 125000000547 substituted alkyl group Chemical group 0.000 description 2
- FPGGTKZVZWFYPV-UHFFFAOYSA-M tetrabutylammonium fluoride Chemical compound [F-].CCCC[N+](CCCC)(CCCC)CCCC FPGGTKZVZWFYPV-UHFFFAOYSA-M 0.000 description 2
- PCCVSPMFGIFTHU-UHFFFAOYSA-N tetracyanoquinodimethane Chemical compound N#CC(C#N)=C1C=CC(=C(C#N)C#N)C=C1 PCCVSPMFGIFTHU-UHFFFAOYSA-N 0.000 description 2
- TVIVIEFSHFOWTE-UHFFFAOYSA-K tri(quinolin-8-yloxy)alumane Chemical compound [Al+3].C1=CN=C2C([O-])=CC=CC2=C1.C1=CN=C2C([O-])=CC=CC2=C1.C1=CN=C2C([O-])=CC=CC2=C1 TVIVIEFSHFOWTE-UHFFFAOYSA-K 0.000 description 2
- ITOFPJRDSCGOSA-KZLRUDJFSA-N (2s)-2-[[(4r)-4-[(3r,5r,8r,9s,10s,13r,14s,17r)-3-hydroxy-10,13-dimethyl-2,3,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydro-1h-cyclopenta[a]phenanthren-17-yl]pentanoyl]amino]-3-(1h-indol-3-yl)propanoic acid Chemical compound C([C@H]1CC2)[C@H](O)CC[C@]1(C)[C@@H](CC[C@]13C)[C@@H]2[C@@H]3CC[C@@H]1[C@H](C)CCC(=O)N[C@H](C(O)=O)CC1=CNC2=CC=CC=C12 ITOFPJRDSCGOSA-KZLRUDJFSA-N 0.000 description 1
- 125000004169 (C1-C6) alkyl group Chemical group 0.000 description 1
- 125000006743 (C1-C60) alkyl group Chemical group 0.000 description 1
- 125000006744 (C2-C60) alkenyl group Chemical group 0.000 description 1
- 125000006818 (C3-C60) cycloalkyl group Chemical group 0.000 description 1
- MIOPJNTWMNEORI-GMSGAONNSA-N (S)-camphorsulfonic acid Chemical compound C1C[C@@]2(CS(O)(=O)=O)C(=O)C[C@@H]1C2(C)C MIOPJNTWMNEORI-GMSGAONNSA-N 0.000 description 1
- 229940005561 1,4-benzoquinone Drugs 0.000 description 1
- MCZUXEWWARACSP-UHFFFAOYSA-N 1-ethynylnaphthalene Chemical group C1=CC=C2C(C#C)=CC=CC2=C1 MCZUXEWWARACSP-UHFFFAOYSA-N 0.000 description 1
- IYZMXHQDXZKNCY-UHFFFAOYSA-N 1-n,1-n-diphenyl-4-n,4-n-bis[4-(n-phenylanilino)phenyl]benzene-1,4-diamine Chemical compound C1=CC=CC=C1N(C=1C=CC(=CC=1)N(C=1C=CC(=CC=1)N(C=1C=CC=CC=1)C=1C=CC=CC=1)C=1C=CC(=CC=1)N(C=1C=CC=CC=1)C=1C=CC=CC=1)C1=CC=CC=C1 IYZMXHQDXZKNCY-UHFFFAOYSA-N 0.000 description 1
- SPDPTFAJSFKAMT-UHFFFAOYSA-N 1-n-[4-[4-(n-[4-(3-methyl-n-(3-methylphenyl)anilino)phenyl]anilino)phenyl]phenyl]-4-n,4-n-bis(3-methylphenyl)-1-n-phenylbenzene-1,4-diamine Chemical compound CC1=CC=CC(N(C=2C=CC(=CC=2)N(C=2C=CC=CC=2)C=2C=CC(=CC=2)C=2C=CC(=CC=2)N(C=2C=CC=CC=2)C=2C=CC(=CC=2)N(C=2C=C(C)C=CC=2)C=2C=C(C)C=CC=2)C=2C=C(C)C=CC=2)=C1 SPDPTFAJSFKAMT-UHFFFAOYSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- GEQBRULPNIVQPP-UHFFFAOYSA-N 2-[3,5-bis(1-phenylbenzimidazol-2-yl)phenyl]-1-phenylbenzimidazole Chemical compound C1=CC=CC=C1N1C2=CC=CC=C2N=C1C1=CC(C=2N(C3=CC=CC=C3N=2)C=2C=CC=CC=2)=CC(C=2N(C3=CC=CC=C3N=2)C=2C=CC=CC=2)=C1 GEQBRULPNIVQPP-UHFFFAOYSA-N 0.000 description 1
- 125000004198 2-fluorophenyl group Chemical group [H]C1=C([H])C(F)=C(*)C([H])=C1[H] 0.000 description 1
- WLPATYNQCGVFFH-UHFFFAOYSA-N 2-phenylbenzonitrile Chemical group N#CC1=CC=CC=C1C1=CC=CC=C1 WLPATYNQCGVFFH-UHFFFAOYSA-N 0.000 description 1
- PPWNCLVNXGCGAF-UHFFFAOYSA-N 3,3-dimethylbut-1-yne Chemical group CC(C)(C)C#C PPWNCLVNXGCGAF-UHFFFAOYSA-N 0.000 description 1
- 125000004180 3-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C(F)=C1[H] 0.000 description 1
- PJUAIXDOXUXBDR-UHFFFAOYSA-N 3-iodo-9-phenylcarbazole Chemical compound C12=CC=CC=C2C2=CC(I)=CC=C2N1C1=CC=CC=C1 PJUAIXDOXUXBDR-UHFFFAOYSA-N 0.000 description 1
- USCSRAJGJYMJFZ-UHFFFAOYSA-N 3-methyl-1-butyne Chemical group CC(C)C#C USCSRAJGJYMJFZ-UHFFFAOYSA-N 0.000 description 1
- FDRNXKXKFNHNCA-UHFFFAOYSA-N 4-(4-anilinophenyl)-n-phenylaniline Chemical compound C=1C=C(C=2C=CC(NC=3C=CC=CC=3)=CC=2)C=CC=1NC1=CC=CC=C1 FDRNXKXKFNHNCA-UHFFFAOYSA-N 0.000 description 1
- AWXGSYPUMWKTBR-UHFFFAOYSA-N 4-carbazol-9-yl-n,n-bis(4-carbazol-9-ylphenyl)aniline Chemical compound C12=CC=CC=C2C2=CC=CC=C2N1C1=CC=C(N(C=2C=CC(=CC=2)N2C3=CC=CC=C3C3=CC=CC=C32)C=2C=CC(=CC=2)N2C3=CC=CC=C3C3=CC=CC=C32)C=C1 AWXGSYPUMWKTBR-UHFFFAOYSA-N 0.000 description 1
- 125000001255 4-fluorophenyl group Chemical group [H]C1=C([H])C(*)=C([H])C([H])=C1F 0.000 description 1
- OSQXTXTYKAEHQV-WXUKJITCSA-N 4-methyl-n-[4-[(e)-2-[4-[4-[(e)-2-[4-(4-methyl-n-(4-methylphenyl)anilino)phenyl]ethenyl]phenyl]phenyl]ethenyl]phenyl]-n-(4-methylphenyl)aniline Chemical compound C1=CC(C)=CC=C1N(C=1C=CC(\C=C\C=2C=CC(=CC=2)C=2C=CC(\C=C\C=3C=CC(=CC=3)N(C=3C=CC(C)=CC=3)C=3C=CC(C)=CC=3)=CC=2)=CC=1)C1=CC=C(C)C=C1 OSQXTXTYKAEHQV-WXUKJITCSA-N 0.000 description 1
- DIVZFUBWFAOMCW-UHFFFAOYSA-N 4-n-(3-methylphenyl)-1-n,1-n-bis[4-(n-(3-methylphenyl)anilino)phenyl]-4-n-phenylbenzene-1,4-diamine Chemical compound CC1=CC=CC(N(C=2C=CC=CC=2)C=2C=CC(=CC=2)N(C=2C=CC(=CC=2)N(C=2C=CC=CC=2)C=2C=C(C)C=CC=2)C=2C=CC(=CC=2)N(C=2C=CC=CC=2)C=2C=C(C)C=CC=2)=C1 DIVZFUBWFAOMCW-UHFFFAOYSA-N 0.000 description 1
- WXAIEIRYBSKHDP-UHFFFAOYSA-N 4-phenyl-n-(4-phenylphenyl)-n-[4-[4-(4-phenyl-n-(4-phenylphenyl)anilino)phenyl]phenyl]aniline Chemical compound C1=CC=CC=C1C1=CC=C(N(C=2C=CC(=CC=2)C=2C=CC=CC=2)C=2C=CC(=CC=2)C=2C=CC(=CC=2)N(C=2C=CC(=CC=2)C=2C=CC=CC=2)C=2C=CC(=CC=2)C=2C=CC=CC=2)C=C1 WXAIEIRYBSKHDP-UHFFFAOYSA-N 0.000 description 1
- VIJYEGDOKCKUOL-UHFFFAOYSA-N 9-phenylcarbazole Chemical compound C1=CC=CC=C1N1C2=CC=CC=C2C2=CC=CC=C21 VIJYEGDOKCKUOL-UHFFFAOYSA-N 0.000 description 1
- 229920001621 AMOLED Polymers 0.000 description 1
- RDCJANRJPSXSHO-UHFFFAOYSA-N C1(=CC=CC=C1)N1C(=NC2=C1C=CC=C2)C=2CC(C=C1C(=C3C(=CC=CC3=C(C21)C2=CC1=CC=CC=C1C=C2)C2=NC1=C(N2C2=CC=CC=C2)C=CC=C1)C1=CC2=CC=CC=C2C=C1)(C(C)(C)C)C1=NC2=C(N1C1=CC=CC=C1)C=CC=C2 Chemical compound C1(=CC=CC=C1)N1C(=NC2=C1C=CC=C2)C=2CC(C=C1C(=C3C(=CC=CC3=C(C21)C2=CC1=CC=CC=C1C=C2)C2=NC1=C(N2C2=CC=CC=C2)C=CC=C1)C1=CC2=CC=CC=C2C=C1)(C(C)(C)C)C1=NC2=C(N1C1=CC=CC=C1)C=CC=C2 RDCJANRJPSXSHO-UHFFFAOYSA-N 0.000 description 1
- 125000003860 C1-C20 alkoxy group Chemical group 0.000 description 1
- LNTJOKGLMLBNPS-UHFFFAOYSA-N C1=C(I)C(Br)=CC2=C1OC=C2 Chemical compound C1=C(I)C(Br)=CC2=C1OC=C2 LNTJOKGLMLBNPS-UHFFFAOYSA-N 0.000 description 1
- KESRRRLHHXXBRW-UHFFFAOYSA-N C1=CC=NC2=C3C(O)=CC=CC3=CC=C21 Chemical compound C1=CC=NC2=C3C(O)=CC=CC3=CC=C21 KESRRRLHHXXBRW-UHFFFAOYSA-N 0.000 description 1
- ZPMBWVLMTYUNEX-UHFFFAOYSA-N CC(C1)C=Cc2c1[o]c(cc1C3(C)C4)c2cc1C(c(cc1)ccc1N(c1ccccc1)c1ccccc1)=CC3=Cc1c4cc[o]1 Chemical compound CC(C1)C=Cc2c1[o]c(cc1C3(C)C4)c2cc1C(c(cc1)ccc1N(c1ccccc1)c1ccccc1)=CC3=Cc1c4cc[o]1 ZPMBWVLMTYUNEX-UHFFFAOYSA-N 0.000 description 1
- PTGURWQRKACDPT-UHFFFAOYSA-N CC(C1)C=Cc2c1[o]c(cc1C3(C)C4)c2cc1C(c(cc1)ccc1OC)=CC3=Cc1c4cc[o]1 Chemical compound CC(C1)C=Cc2c1[o]c(cc1C3(C)C4)c2cc1C(c(cc1)ccc1OC)=CC3=Cc1c4cc[o]1 PTGURWQRKACDPT-UHFFFAOYSA-N 0.000 description 1
- ZULQRIRQCREOHR-UHFFFAOYSA-N CC(C1)C=Cc2c1[o]c1cc3c(cc(c(cc4)c5)[n]4-c4ccccc4)c5cc(-c4cnccc4)c3cc21 Chemical compound CC(C1)C=Cc2c1[o]c1cc3c(cc(c(cc4)c5)[n]4-c4ccccc4)c5cc(-c4cnccc4)c3cc21 ZULQRIRQCREOHR-UHFFFAOYSA-N 0.000 description 1
- RQAYGMLGXZTLKU-UHFFFAOYSA-N CC(C1)C=Cc2c1[s]c1cc3c(cc(cc[n]4-c5ccccc5)c4c4)c4cc(-c(cc4)cc(c5ccccc55)c4[n]5-c4ccccc4)c3cc21 Chemical compound CC(C1)C=Cc2c1[s]c1cc3c(cc(cc[n]4-c5ccccc5)c4c4)c4cc(-c(cc4)cc(c5ccccc55)c4[n]5-c4ccccc4)c3cc21 RQAYGMLGXZTLKU-UHFFFAOYSA-N 0.000 description 1
- VFOVYRSWMVEUOY-UHFFFAOYSA-N CC(CC=C1)C=C1[n]1c2cc(C(c3cnccc3)=CC(C3(C)C4)=Cc5c4cc[n]5-c4ccccc4)c3cc2c2c1cccc2 Chemical compound CC(CC=C1)C=C1[n]1c2cc(C(c3cnccc3)=CC(C3(C)C4)=Cc5c4cc[n]5-c4ccccc4)c3cc2c2c1cccc2 VFOVYRSWMVEUOY-UHFFFAOYSA-N 0.000 description 1
- WZOPTUNUFWEJKG-UHFFFAOYSA-N CC1(C)c(cc(cc2)C(C(C3(C)C4)=Cc5c4cc[o]5)=C(c(cc4)ccc4N(c4ccccc4)c4ccccc4)c4c3c(OC3(C)C=CC=CC33)c3cc4)c2-c2ccccc12 Chemical compound CC1(C)c(cc(cc2)C(C(C3(C)C4)=Cc5c4cc[o]5)=C(c(cc4)ccc4N(c4ccccc4)c4ccccc4)c4c3c(OC3(C)C=CC=CC33)c3cc4)c2-c2ccccc12 WZOPTUNUFWEJKG-UHFFFAOYSA-N 0.000 description 1
- BLCZAHVEZLEWFM-UHFFFAOYSA-N CC1C([n]2c3cc(C(c(cc4)ccc4OC)=CC(C4(C)C5)=Cc6c5cc[n]6C5(C)C=CC=CC5)c4cc3c3c2cccc3)=CC=CC1 Chemical compound CC1C([n]2c3cc(C(c(cc4)ccc4OC)=CC(C4(C)C5)=Cc6c5cc[n]6C5(C)C=CC=CC5)c4cc3c3c2cccc3)=CC=CC1 BLCZAHVEZLEWFM-UHFFFAOYSA-N 0.000 description 1
- RCTVWLWHLWQDTH-UHFFFAOYSA-N COc(cc1)ccc1-c(c1ccc(c2ccccc2[o]2)c2c1c1c2)cc1cc(cc1)c2[n]1-c1ccccc1 Chemical compound COc(cc1)ccc1-c(c1ccc(c2ccccc2[o]2)c2c1c1c2)cc1cc(cc1)c2[n]1-c1ccccc1 RCTVWLWHLWQDTH-UHFFFAOYSA-N 0.000 description 1
- ABUSXXDPWFMTBS-UHFFFAOYSA-N COc(cc1)ccc1-c1c(cc(c(cccc2)c2[o]2)c2c2)c2c(cc(c(cc2)c3)[n]2-c2ccccc2)c3c1 Chemical compound COc(cc1)ccc1-c1c(cc(c(cccc2)c2[o]2)c2c2)c2c(cc(c(cc2)c3)[n]2-c2ccccc2)c3c1 ABUSXXDPWFMTBS-UHFFFAOYSA-N 0.000 description 1
- SQKJYCAKWYCWEH-UHFFFAOYSA-N COc(cc1)ccc1-c1c(cc(c(cccc2)c2[s]2)c2c2)c2c(cc(cc[n]2-c3ccccc3)c2c2)c2c1 Chemical compound COc(cc1)ccc1-c1c(cc(c(cccc2)c2[s]2)c2c2)c2c(cc(cc[n]2-c3ccccc3)c2c2)c2c1 SQKJYCAKWYCWEH-UHFFFAOYSA-N 0.000 description 1
- ASLPBNYRVVANQH-UHFFFAOYSA-N COc(cc1)ccc1-c1cc2cc([n](cc3)-c4ccccc4)c3cc2c2c1ccc1c2[o]c2ccccc12 Chemical compound COc(cc1)ccc1-c1cc2cc([n](cc3)-c4ccccc4)c3cc2c2c1ccc1c2[o]c2ccccc12 ASLPBNYRVVANQH-UHFFFAOYSA-N 0.000 description 1
- DHJWKMOBDCEHEO-UHFFFAOYSA-O C[NH2+]C(c1c(cc(c(cc2)c3)[n]2-c2ccccc2)c3c(cc2[s]c(cccc3)c3c2c2)c2c1-c1c[s]c2c1cccc2)=CC=N Chemical compound C[NH2+]C(c1c(cc(c(cc2)c3)[n]2-c2ccccc2)c3c(cc2[s]c(cccc3)c3c2c2)c2c1-c1c[s]c2c1cccc2)=CC=N DHJWKMOBDCEHEO-UHFFFAOYSA-O 0.000 description 1
- 101000837344 Homo sapiens T-cell leukemia translocation-altered gene protein Proteins 0.000 description 1
- 229910018068 Li 2 O Inorganic materials 0.000 description 1
- WHXSMMKQMYFTQS-UHFFFAOYSA-N Lithium Chemical compound [Li] WHXSMMKQMYFTQS-UHFFFAOYSA-N 0.000 description 1
- CBENFWSGALASAD-UHFFFAOYSA-N Ozone Chemical compound [O-][O+]=O CBENFWSGALASAD-UHFFFAOYSA-N 0.000 description 1
- OAICVXFJPJFONN-UHFFFAOYSA-N Phosphorus Chemical compound [P] OAICVXFJPJFONN-UHFFFAOYSA-N 0.000 description 1
- 229910006404 SnO 2 Inorganic materials 0.000 description 1
- 102100028692 T-cell leukemia translocation-altered gene protein Human genes 0.000 description 1
- XLOMVQKBTHCTTD-UHFFFAOYSA-N Zinc monoxide Chemical compound [Zn]=O XLOMVQKBTHCTTD-UHFFFAOYSA-N 0.000 description 1
- 229910001508 alkali metal halide Inorganic materials 0.000 description 1
- 150000008045 alkali metal halides Chemical class 0.000 description 1
- 229910045601 alloy Inorganic materials 0.000 description 1
- 239000000956 alloy Substances 0.000 description 1
- HSFWRNGVRCDJHI-UHFFFAOYSA-N alpha-acetylene Natural products C#C HSFWRNGVRCDJHI-UHFFFAOYSA-N 0.000 description 1
- 125000002178 anthracenyl group Chemical group C1(=CC=CC2=CC3=CC=CC=C3C=C12)* 0.000 description 1
- 229940027991 antiseptic and disinfectant quinoline derivative Drugs 0.000 description 1
- QVQLCTNNEUAWMS-UHFFFAOYSA-N barium oxide Inorganic materials [Ba]=O QVQLCTNNEUAWMS-UHFFFAOYSA-N 0.000 description 1
- 125000005605 benzo group Chemical group 0.000 description 1
- 125000003236 benzoyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C(*)=O 0.000 description 1
- 229910052790 beryllium Inorganic materials 0.000 description 1
- ATBAMAFKBVZNFJ-UHFFFAOYSA-N beryllium atom Chemical compound [Be] ATBAMAFKBVZNFJ-UHFFFAOYSA-N 0.000 description 1
- 235000010290 biphenyl Nutrition 0.000 description 1
- 239000004305 biphenyl Substances 0.000 description 1
- JRXXLCKWQFKACW-UHFFFAOYSA-N biphenylacetylene Chemical group C1=CC=CC=C1C#CC1=CC=CC=C1 JRXXLCKWQFKACW-UHFFFAOYSA-N 0.000 description 1
- 125000004369 butenyl group Chemical group C(=CCC)* 0.000 description 1
- 125000004106 butoxy group Chemical group [*]OC([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 125000000484 butyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- XMYOPWYUCBPIIZ-UHFFFAOYSA-N c(c(c1c2)cc3c2c(c2c(cc4)c5ccccc5[o]2)c4c(-c(cc2)ccc2N(c2ccccc2)c2ccccc2)c3)c[n]1-c1ccccc1 Chemical compound c(c(c1c2)cc3c2c(c2c(cc4)c5ccccc5[o]2)c4c(-c(cc2)ccc2N(c2ccccc2)c2ccccc2)c3)c[n]1-c1ccccc1 XMYOPWYUCBPIIZ-UHFFFAOYSA-N 0.000 description 1
- HFWGNOFMMOTJIO-UHFFFAOYSA-N c(c(c1c2)cc3c2c(c2c(cc4)c5ccccc5[o]2)c4c(-c2cnccc2)c3)c[n]1-c1ccccc1 Chemical compound c(c(c1c2)cc3c2c(c2c(cc4)c5ccccc5[o]2)c4c(-c2cnccc2)c3)c[n]1-c1ccccc1 HFWGNOFMMOTJIO-UHFFFAOYSA-N 0.000 description 1
- YHCXSWKWQDFNBV-UHFFFAOYSA-N c(c(c1c2)cc3c2cc(-c(cc2)ccc2N(c2ccccc2)c2ccccc2)c(cc2)c3c3c2c2ccccc2[s]3)c[n]1-c1ccccc1 Chemical compound c(c(c1c2)cc3c2cc(-c(cc2)ccc2N(c2ccccc2)c2ccccc2)c(cc2)c3c3c2c2ccccc2[s]3)c[n]1-c1ccccc1 YHCXSWKWQDFNBV-UHFFFAOYSA-N 0.000 description 1
- AUZJPOGOZAFGEW-UHFFFAOYSA-N c(c(c1c2)cc3c2cc(-c2c[s]c4c2cccc4)c(cc2)c3c3c2c2ccccc2[s]3)c[n]1-c1ccccc1 Chemical compound c(c(c1c2)cc3c2cc(-c2c[s]c4c2cccc4)c(cc2)c3c3c2c2ccccc2[s]3)c[n]1-c1ccccc1 AUZJPOGOZAFGEW-UHFFFAOYSA-N 0.000 description 1
- DANAPIYKJYVPKH-UHFFFAOYSA-N c(c(c1cc2c3)cc2c(cc2[s]c(cccc4)c4c2c2)c2c3-c(cc2)cc3c2[o]c2ccccc32)c[n]1-c1ccccc1 Chemical compound c(c(c1cc2c3)cc2c(cc2[s]c(cccc4)c4c2c2)c2c3-c(cc2)cc3c2[o]c2ccccc32)c[n]1-c1ccccc1 DANAPIYKJYVPKH-UHFFFAOYSA-N 0.000 description 1
- XCFBMSYVZBBYNQ-UHFFFAOYSA-N c(c(c1cc2c3)cc2c(cc2[s]c(cccc4)c4c2c2)c2c3-c(cc2)ccc2N(c2ccccc2)c2ccccc2)c[n]1-c1ccccc1 Chemical compound c(c(c1cc2c3)cc2c(cc2[s]c(cccc4)c4c2c2)c2c3-c(cc2)ccc2N(c2ccccc2)c2ccccc2)c[n]1-c1ccccc1 XCFBMSYVZBBYNQ-UHFFFAOYSA-N 0.000 description 1
- WKZIZWKAQFEHBF-UHFFFAOYSA-N c(c(c1cc2c3)cc2c(cc2[s]c(cccc4)c4c2c2)c2c3-c2c3[s]c(cccc4)c4c3ccc2)c[n]1-c1ccccc1 Chemical compound c(c(c1cc2c3)cc2c(cc2[s]c(cccc4)c4c2c2)c2c3-c2c3[s]c(cccc4)c4c3ccc2)c[n]1-c1ccccc1 WKZIZWKAQFEHBF-UHFFFAOYSA-N 0.000 description 1
- ZSKHZYBBRUPUDG-UHFFFAOYSA-N c(c(c1cc2c3)cc2c(cc2[s]c(cccc4)c4c2c2)c2c3-c2c[s]c3c2cccc3)c[n]1-c1ccccc1 Chemical compound c(c(c1cc2c3)cc2c(cc2[s]c(cccc4)c4c2c2)c2c3-c2c[s]c3c2cccc3)c[n]1-c1ccccc1 ZSKHZYBBRUPUDG-UHFFFAOYSA-N 0.000 description 1
- MCVWECAYYNWKMX-UHFFFAOYSA-N c(c(c1cc2c3)cc2c(cc2[s]c(cccc4)c4c2c2)c2c3-c2cnccc2)c[n]1-c1ccccc1 Chemical compound c(c(c1cc2c3)cc2c(cc2[s]c(cccc4)c4c2c2)c2c3-c2cnccc2)c[n]1-c1ccccc1 MCVWECAYYNWKMX-UHFFFAOYSA-N 0.000 description 1
- BEKMXYIBLYNQLJ-UHFFFAOYSA-N c(c1c2cc(cc(-c(cc3)cc(c4ccccc44)c3[n]4-c3ccccc3)c(cc3)c4c5c3c3ccccc3[s]5)c4c1)c[n]2-c1ccccc1 Chemical compound c(c1c2cc(cc(-c(cc3)cc(c4ccccc44)c3[n]4-c3ccccc3)c(cc3)c4c5c3c3ccccc3[s]5)c4c1)c[n]2-c1ccccc1 BEKMXYIBLYNQLJ-UHFFFAOYSA-N 0.000 description 1
- BVISRBZXHDRBNZ-UHFFFAOYSA-N c(c1c2cc(cc(-c(cc3)cc4c3[o]c3ccccc43)c3ccc(c4ccccc4[s]4)c4c33)c3c1)c[n]2-c1ccccc1 Chemical compound c(c1c2cc(cc(-c(cc3)cc4c3[o]c3ccccc43)c3ccc(c4ccccc4[s]4)c4c33)c3c1)c[n]2-c1ccccc1 BVISRBZXHDRBNZ-UHFFFAOYSA-N 0.000 description 1
- MDHLWXRNOQGDHV-UHFFFAOYSA-N c(c1c2cc(cc(-c3c4[s]c5ccccc5c4ccc3)c(cc3)c4c5c3c3ccccc3[s]5)c4c1)c[n]2-c1ccccc1 Chemical compound c(c1c2cc(cc(-c3c4[s]c5ccccc5c4ccc3)c(cc3)c4c5c3c3ccccc3[s]5)c4c1)c[n]2-c1ccccc1 MDHLWXRNOQGDHV-UHFFFAOYSA-N 0.000 description 1
- FZWJWWSVLKLMHU-UHFFFAOYSA-N c(c1c2cc3c(-c(cc4)ccc4N(c4ccccc4)c4ccccc4)c(-c4cnccc4)c(ccc4c5[s]c6ccccc46)c5c3c1)c[n]2-c1ccccc1 Chemical compound c(c1c2cc3c(-c(cc4)ccc4N(c4ccccc4)c4ccccc4)c(-c4cnccc4)c(ccc4c5[s]c6ccccc46)c5c3c1)c[n]2-c1ccccc1 FZWJWWSVLKLMHU-UHFFFAOYSA-N 0.000 description 1
- GFKQLAWLHNGWAE-UHFFFAOYSA-N c(c1c2cc3c(c4c(cc5)c(cccc6)c6[o]4)c5c(-c4c5[s]c(cccc6)c6c5ccc4)cc3c1)c[n]2-c1ccccc1 Chemical compound c(c1c2cc3c(c4c(cc5)c(cccc6)c6[o]4)c5c(-c4c5[s]c(cccc6)c6c5ccc4)cc3c1)c[n]2-c1ccccc1 GFKQLAWLHNGWAE-UHFFFAOYSA-N 0.000 description 1
- YSPHGTCXUWEOFF-UHFFFAOYSA-N c(c1c2cc3c(c4c(cc5)c6ccccc6[o]4)c5c(-c(cc4)cc(c5ccccc55)c4[n]5-c4ccccc4)cc3c1)c[n]2-c1ccccc1 Chemical compound c(c1c2cc3c(c4c(cc5)c6ccccc6[o]4)c5c(-c(cc4)cc(c5ccccc55)c4[n]5-c4ccccc4)cc3c1)c[n]2-c1ccccc1 YSPHGTCXUWEOFF-UHFFFAOYSA-N 0.000 description 1
- UQGMEFHZWAMORQ-UHFFFAOYSA-N c(c1c2cc3c(c4c(cc5)c6ccccc6[o]4)c5c(-c4c[s]c5c4cccc5)c(-c(cc4)ccc4N(c4ccccc4)c4ccccc4)c3c1)c[n]2-c1ccccc1 Chemical compound c(c1c2cc3c(c4c(cc5)c6ccccc6[o]4)c5c(-c4c[s]c5c4cccc5)c(-c(cc4)ccc4N(c4ccccc4)c4ccccc4)c3c1)c[n]2-c1ccccc1 UQGMEFHZWAMORQ-UHFFFAOYSA-N 0.000 description 1
- QCNDSDWCRBJDDA-UHFFFAOYSA-N c(c1c2cc3c(c4c(cc5)c6ccccc6[o]4)c5c(-c4c[s]c5c4cccc5)cc3c1)c[n]2-c1ccccc1 Chemical compound c(c1c2cc3c(c4c(cc5)c6ccccc6[o]4)c5c(-c4c[s]c5c4cccc5)cc3c1)c[n]2-c1ccccc1 QCNDSDWCRBJDDA-UHFFFAOYSA-N 0.000 description 1
- NIKNGWWENLHOIU-UHFFFAOYSA-N c(c1c2cc3c4c5[o]c6ccccc6c5ccc4c(-c(cc4)cc5c4[o]c4ccccc54)cc3c1)c[n]2-c1ccccc1 Chemical compound c(c1c2cc3c4c5[o]c6ccccc6c5ccc4c(-c(cc4)cc5c4[o]c4ccccc54)cc3c1)c[n]2-c1ccccc1 NIKNGWWENLHOIU-UHFFFAOYSA-N 0.000 description 1
- SIDAJOFUMNEJES-UHFFFAOYSA-N c(c1cc2c(-c(cc3)ccc3-c3ccccc3)c(-c3cnccc3)c(cc(c(cccc3)c3[s]3)c3c3)c3c2cc11)c[n]1-c1ccccc1 Chemical compound c(c1cc2c(-c(cc3)ccc3-c3ccccc3)c(-c3cnccc3)c(cc(c(cccc3)c3[s]3)c3c3)c3c2cc11)c[n]1-c1ccccc1 SIDAJOFUMNEJES-UHFFFAOYSA-N 0.000 description 1
- FSLIIIYTYNROIH-UHFFFAOYSA-N c(c1cc2cc(-c(cc3)cc4c3[o]c3ccccc43)c(cc(c(cccc3)c3[s]3)c3c3)c3c2cc11)c[n]1-c1ccccc1 Chemical compound c(c1cc2cc(-c(cc3)cc4c3[o]c3ccccc43)c(cc(c(cccc3)c3[s]3)c3c3)c3c2cc11)c[n]1-c1ccccc1 FSLIIIYTYNROIH-UHFFFAOYSA-N 0.000 description 1
- QISDIAPCEUYPTD-UHFFFAOYSA-N c1c[o]c2cc3cc(-c4c5[s]c(cccc6)c6c5ccc4)c(cc(c(cccc4)c4[o]4)c4c4)c4c3cc12 Chemical compound c1c[o]c2cc3cc(-c4c5[s]c(cccc6)c6c5ccc4)c(cc(c(cccc4)c4[o]4)c4c4)c4c3cc12 QISDIAPCEUYPTD-UHFFFAOYSA-N 0.000 description 1
- XJHCXCQVJFPJIK-UHFFFAOYSA-M caesium fluoride Inorganic materials [F-].[Cs+] XJHCXCQVJFPJIK-UHFFFAOYSA-M 0.000 description 1
- 150000001716 carbazoles Chemical class 0.000 description 1
- 239000000969 carrier Substances 0.000 description 1
- 125000002676 chrysenyl group Chemical group C1(=CC=CC=2C3=CC=C4C=CC=CC4=C3C=CC12)* 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 229940125904 compound 1 Drugs 0.000 description 1
- 229940125773 compound 10 Drugs 0.000 description 1
- 229940125810 compound 20 Drugs 0.000 description 1
- 229940126214 compound 3 Drugs 0.000 description 1
- 229940125898 compound 5 Drugs 0.000 description 1
- 125000003336 coronenyl group Chemical group C1(=CC2=CC=C3C=CC4=CC=C5C=CC6=CC=C1C1=C6C5=C4C3=C21)* 0.000 description 1
- 238000002425 crystallisation Methods 0.000 description 1
- 230000008025 crystallization Effects 0.000 description 1
- 125000004802 cyanophenyl group Chemical group 0.000 description 1
- 125000006165 cyclic alkyl group Chemical group 0.000 description 1
- 150000001975 deuterium Chemical group 0.000 description 1
- 125000004188 dichlorophenyl group Chemical group 0.000 description 1
- 238000005401 electroluminescence Methods 0.000 description 1
- 125000001301 ethoxy group Chemical group [H]C([H])([H])C([H])([H])O* 0.000 description 1
- 125000002534 ethynyl group Chemical group [H]C#C* 0.000 description 1
- 230000005281 excited state Effects 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- JVZRCNQLWOELDU-UHFFFAOYSA-N gamma-Phenylpyridine Natural products C1=CC=CC=C1C1=CC=NC=C1 JVZRCNQLWOELDU-UHFFFAOYSA-N 0.000 description 1
- 230000005283 ground state Effects 0.000 description 1
- 238000010438 heat treatment Methods 0.000 description 1
- RBTKNAXYKSUFRK-UHFFFAOYSA-N heliogen blue Chemical compound [Cu].[N-]1C2=C(C=CC=C3)C3=C1N=C([N-]1)C3=CC=CC=C3C1=NC([N-]1)=C(C=CC=C3)C3=C1N=C([N-]1)C3=CC=CC=C3C1=N2 RBTKNAXYKSUFRK-UHFFFAOYSA-N 0.000 description 1
- 125000003824 heptacenyl group Chemical group C1(=CC=CC2=CC3=CC4=CC5=CC6=CC7=CC=CC=C7C=C6C=C5C=C4C=C3C=C12)* 0.000 description 1
- 125000003187 heptyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 125000005842 heteroatom Chemical group 0.000 description 1
- 125000001633 hexacenyl group Chemical group C1(=CC=CC2=CC3=CC4=CC5=CC6=CC=CC=C6C=C5C=C4C=C3C=C12)* 0.000 description 1
- 125000002883 imidazolyl group Chemical group 0.000 description 1
- AMGQUBHHOARCQH-UHFFFAOYSA-N indium;oxotin Chemical compound [In].[Sn]=O AMGQUBHHOARCQH-UHFFFAOYSA-N 0.000 description 1
- 125000001041 indolyl group Chemical group 0.000 description 1
- 239000011229 interlayer Substances 0.000 description 1
- 125000000959 isobutyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])* 0.000 description 1
- 125000001972 isopentyl group Chemical group [H]C([H])([H])C([H])(C([H])([H])[H])C([H])([H])C([H])([H])* 0.000 description 1
- 125000002183 isoquinolinyl group Chemical group C1(=NC=CC2=CC=CC=C12)* 0.000 description 1
- ZLVXBBHTMQJRSX-VMGNSXQWSA-N jdtic Chemical compound C1([C@]2(C)CCN(C[C@@H]2C)C[C@H](C(C)C)NC(=O)[C@@H]2NCC3=CC(O)=CC=C3C2)=CC=CC(O)=C1 ZLVXBBHTMQJRSX-VMGNSXQWSA-N 0.000 description 1
- 229910052744 lithium Inorganic materials 0.000 description 1
- PQXKHYXIUOZZFA-UHFFFAOYSA-M lithium fluoride Inorganic materials [Li+].[F-] PQXKHYXIUOZZFA-UHFFFAOYSA-M 0.000 description 1
- 125000000040 m-tolyl group Chemical group [H]C1=C([H])C(*)=C([H])C(=C1[H])C([H])([H])[H] 0.000 description 1
- 238000004519 manufacturing process Methods 0.000 description 1
- 230000008018 melting Effects 0.000 description 1
- 238000002844 melting Methods 0.000 description 1
- 125000000956 methoxy group Chemical group [H]C([H])([H])O* 0.000 description 1
- 125000001570 methylene group Chemical group [H]C([H])([*:1])[*:2] 0.000 description 1
- 229910000476 molybdenum oxide Inorganic materials 0.000 description 1
- 125000005029 naphthylthio group Chemical group C1(=CC=CC2=CC=CC=C12)S* 0.000 description 1
- 125000006574 non-aromatic ring group Chemical group 0.000 description 1
- QGLKJKCYBOYXKC-UHFFFAOYSA-N nonaoxidotritungsten Chemical compound O=[W]1(=O)O[W](=O)(=O)O[W](=O)(=O)O1 QGLKJKCYBOYXKC-UHFFFAOYSA-N 0.000 description 1
- CTQNGGLPUBDAKN-UHFFFAOYSA-N o-dimethylbenzene Natural products CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 1
- 125000002347 octyl group Chemical group [H]C([*])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])C([H])([H])[H] 0.000 description 1
- 230000003287 optical effect Effects 0.000 description 1
- 150000004866 oxadiazoles Chemical class 0.000 description 1
- 125000001715 oxadiazolyl group Chemical group 0.000 description 1
- 125000002971 oxazolyl group Chemical group 0.000 description 1
- PQQKPALAQIIWST-UHFFFAOYSA-N oxomolybdenum Chemical compound [Mo]=O PQQKPALAQIIWST-UHFFFAOYSA-N 0.000 description 1
- 229910052760 oxygen Inorganic materials 0.000 description 1
- 125000001037 p-tolyl group Chemical group [H]C1=C([H])C(=C([H])C([H])=C1*)C([H])([H])[H] 0.000 description 1
- 125000003933 pentacenyl group Chemical group C1(=CC=CC2=CC3=CC4=CC5=CC=CC=C5C=C4C=C3C=C12)* 0.000 description 1
- JQQSUOJIMKJQHS-UHFFFAOYSA-N pentaphenyl group Chemical group C1=CC=CC2=CC3=CC=C4C=C5C=CC=CC5=CC4=C3C=C12 JQQSUOJIMKJQHS-UHFFFAOYSA-N 0.000 description 1
- 125000004115 pentoxy group Chemical group [*]OC([H])([H])C([H])([H])C([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 125000002080 perylenyl group Chemical group C1(=CC=C2C=CC=C3C4=CC=CC5=CC=CC(C1=C23)=C45)* 0.000 description 1
- 150000005041 phenanthrolines Chemical class 0.000 description 1
- 125000005561 phenanthryl group Chemical group 0.000 description 1
- 125000000951 phenoxy group Chemical group [H]C1=C([H])C([H])=C(O*)C([H])=C1[H] 0.000 description 1
- ZUOUZKKEUPVFJK-UHFFFAOYSA-N phenylbenzene Natural products C1=CC=CC=C1C1=CC=CC=C1 ZUOUZKKEUPVFJK-UHFFFAOYSA-N 0.000 description 1
- 239000011574 phosphorus Substances 0.000 description 1
- 125000001388 picenyl group Chemical group C1(=CC=CC2=CC=C3C4=CC=C5C=CC=CC5=C4C=CC3=C21)* 0.000 description 1
- 229920003023 plastic Polymers 0.000 description 1
- 125000004368 propenyl group Chemical group C(=CC)* 0.000 description 1
- 125000002572 propoxy group Chemical group [*]OC([H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- QQONPFPTGQHPMA-UHFFFAOYSA-N propylene Natural products CC=C QQONPFPTGQHPMA-UHFFFAOYSA-N 0.000 description 1
- 125000004805 propylene group Chemical group [H]C([H])([H])C([H])([*:1])C([H])([H])[*:2] 0.000 description 1
- 125000003226 pyrazolyl group Chemical group 0.000 description 1
- 125000002098 pyridazinyl group Chemical group 0.000 description 1
- 125000000714 pyrimidinyl group Chemical group 0.000 description 1
- 150000003248 quinolines Chemical class 0.000 description 1
- 125000002943 quinolinyl group Chemical group N1=C(C=CC2=CC=CC=C12)* 0.000 description 1
- 239000000376 reactant Substances 0.000 description 1
- 239000011541 reaction mixture Substances 0.000 description 1
- 125000002914 sec-butyl group Chemical group [H]C([H])([H])C([H])([H])C([H])(*)C([H])([H])[H] 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 238000009987 spinning Methods 0.000 description 1
- 238000004544 sputter deposition Methods 0.000 description 1
- 238000003860 storage Methods 0.000 description 1
- 229910052717 sulfur Inorganic materials 0.000 description 1
- 229940042055 systemic antimycotics triazole derivative Drugs 0.000 description 1
- 125000003831 tetrazolyl group Chemical group 0.000 description 1
- 125000000335 thiazolyl group Chemical group 0.000 description 1
- RMVRSNDYEFQCLF-UHFFFAOYSA-N thiophenol Chemical group SC1=CC=CC=C1 RMVRSNDYEFQCLF-UHFFFAOYSA-N 0.000 description 1
- XOLBLPGZBRYERU-UHFFFAOYSA-N tin dioxide Chemical compound O=[Sn]=O XOLBLPGZBRYERU-UHFFFAOYSA-N 0.000 description 1
- 229910001887 tin oxide Inorganic materials 0.000 description 1
- 125000001425 triazolyl group Chemical group 0.000 description 1
- CWMFRHBXRUITQE-UHFFFAOYSA-N trimethylsilylacetylene Chemical group C[Si](C)(C)C#C CWMFRHBXRUITQE-UHFFFAOYSA-N 0.000 description 1
- ODHXBMXNKOYIBV-UHFFFAOYSA-N triphenylamine Chemical compound C1=CC=CC=C1N(C=1C=CC=CC=1)C1=CC=CC=C1 ODHXBMXNKOYIBV-UHFFFAOYSA-N 0.000 description 1
- 125000005580 triphenylene group Chemical group 0.000 description 1
- 229910001930 tungsten oxide Inorganic materials 0.000 description 1
- 125000000391 vinyl group Chemical group [H]C([*])=C([H])[H] 0.000 description 1
- YVTHLONGBIQYBO-UHFFFAOYSA-N zinc indium(3+) oxygen(2-) Chemical compound [O--].[Zn++].[In+3] YVTHLONGBIQYBO-UHFFFAOYSA-N 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/02—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains two hetero rings
- C07D491/04—Ortho-condensed systems
- C07D491/044—Ortho-condensed systems with only one oxygen atom as ring hetero atom in the oxygen-containing ring
- C07D491/048—Ortho-condensed systems with only one oxygen atom as ring hetero atom in the oxygen-containing ring the oxygen-containing ring being five-membered
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/02—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains two hetero rings
- C07D491/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D487/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00
- C07D487/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, not provided for by groups C07D451/00 - C07D477/00 in which the condensed system contains two hetero rings
- C07D487/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D491/00—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00
- C07D491/12—Heterocyclic compounds containing in the condensed ring system both one or more rings having oxygen atoms as the only ring hetero atoms and one or more rings having nitrogen atoms as the only ring hetero atoms, not provided for by groups C07D451/00 - C07D459/00, C07D463/00, C07D477/00 or C07D489/00 in which the condensed system contains three hetero rings
- C07D491/14—Ortho-condensed systems
- C07D491/153—Ortho-condensed systems the condensed system containing two rings with oxygen as ring hetero atom and one ring with nitrogen as ring hetero atom
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D493/00—Heterocyclic compounds containing oxygen atoms as the only ring hetero atoms in the condensed system
- C07D493/02—Heterocyclic compounds containing oxygen atoms as the only ring hetero atoms in the condensed system in which the condensed system contains two hetero rings
- C07D493/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/02—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains two hetero rings
- C07D495/04—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D495/00—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms
- C07D495/12—Heterocyclic compounds containing in the condensed system at least one hetero ring having sulfur atoms as the only ring hetero atoms in which the condensed system contains three hetero rings
- C07D495/14—Ortho-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H05—ELECTRIC TECHNIQUES NOT OTHERWISE PROVIDED FOR
- H05B—ELECTRIC HEATING; ELECTRIC LIGHT SOURCES NOT OTHERWISE PROVIDED FOR; CIRCUIT ARRANGEMENTS FOR ELECTRIC LIGHT SOURCES, IN GENERAL
- H05B33/00—Electroluminescent light sources
- H05B33/12—Light sources with substantially two-dimensional radiating surfaces
- H05B33/14—Light sources with substantially two-dimensional radiating surfaces characterised by the chemical or physical composition or the arrangement of the electroluminescent material, or by the simultaneous addition of the electroluminescent material in or onto the light source
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/16—Electron transporting layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
- H10K85/636—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine comprising heteroaromatic hydrocarbons as substituents on the nitrogen atom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/654—Aromatic compounds comprising a hetero atom comprising only nitrogen as heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6572—Polycyclic condensed heteroaromatic hydrocarbons comprising only nitrogen in the heteroaromatic polycondensed ring system, e.g. phenanthroline or carbazole
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6574—Polycyclic condensed heteroaromatic hydrocarbons comprising only oxygen in the heteroaromatic polycondensed ring system, e.g. cumarine dyes
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/649—Aromatic compounds comprising a hetero atom
- H10K85/657—Polycyclic condensed heteroaromatic hydrocarbons
- H10K85/6576—Polycyclic condensed heteroaromatic hydrocarbons comprising only sulfur in the heteroaromatic polycondensed ring system, e.g. benzothiophene
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1007—Non-condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1003—Carbocyclic compounds
- C09K2211/1011—Condensed systems
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1022—Heterocyclic compounds bridged by heteroatoms, e.g. N, P, Si or B
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
- C09K2211/1033—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom with oxygen
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1029—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom
- C09K2211/1037—Heterocyclic compounds characterised by ligands containing one nitrogen atom as the heteroatom with sulfur
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1044—Heterocyclic compounds characterised by ligands containing two nitrogen atoms as heteroatoms
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1088—Heterocyclic compounds characterised by ligands containing oxygen as the only heteroatom
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K2211/00—Chemical nature of organic luminescent or tenebrescent compounds
- C09K2211/10—Non-macromolecular compounds
- C09K2211/1018—Heterocyclic compounds
- C09K2211/1025—Heterocyclic compounds characterised by ligands
- C09K2211/1092—Heterocyclic compounds characterised by ligands containing sulfur as the only heteroatom
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K2101/00—Properties of the organic materials covered by group H10K85/00
- H10K2101/10—Triplet emission
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/14—Carrier transporting layers
- H10K50/15—Hole transporting layers
- H10K50/155—Hole transporting layers comprising dopants
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Physics & Mathematics (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Optics & Photonics (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
(상기 화학식 1~4에 대해서는 발명의 상세한 설명을 참조한다).
Description
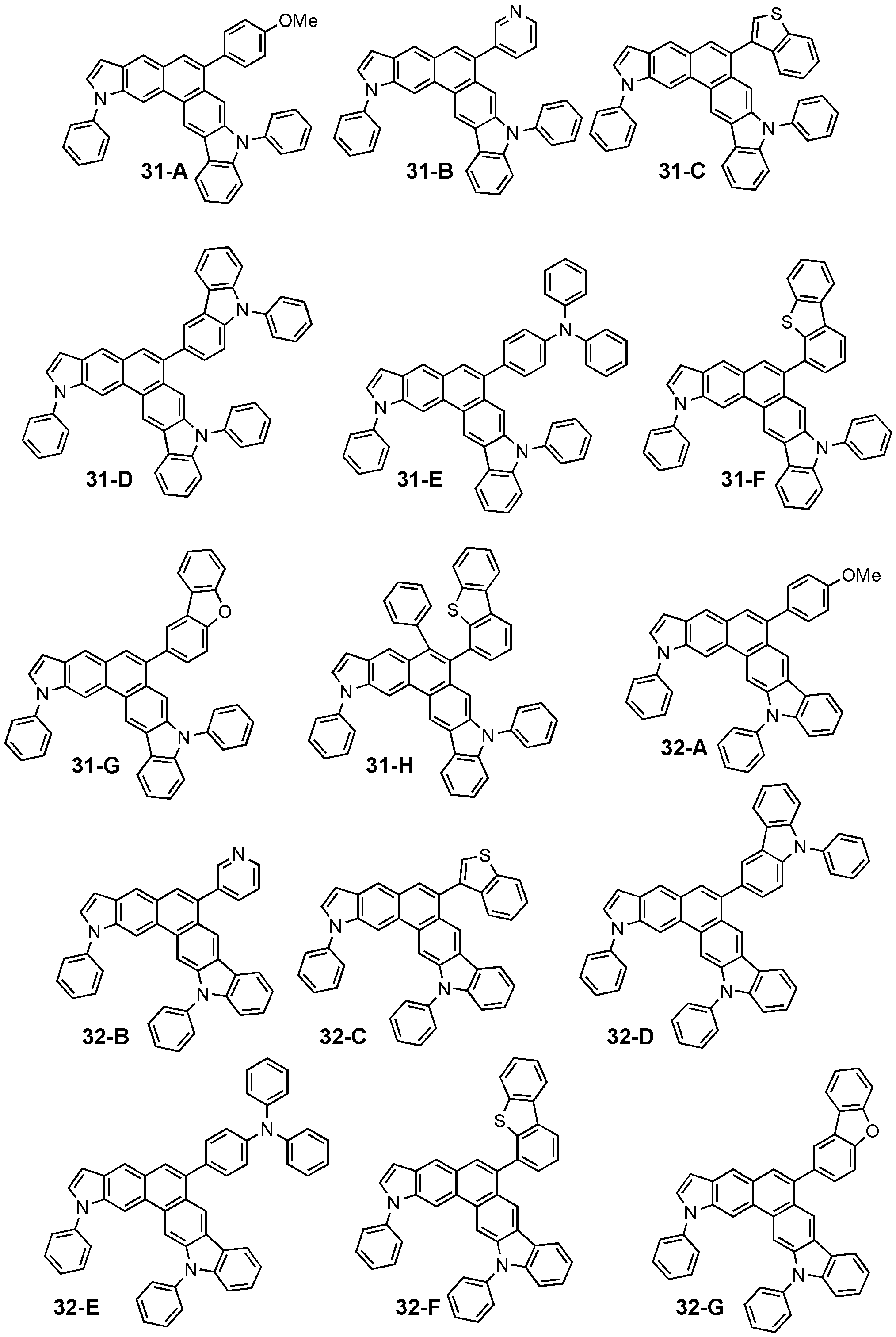
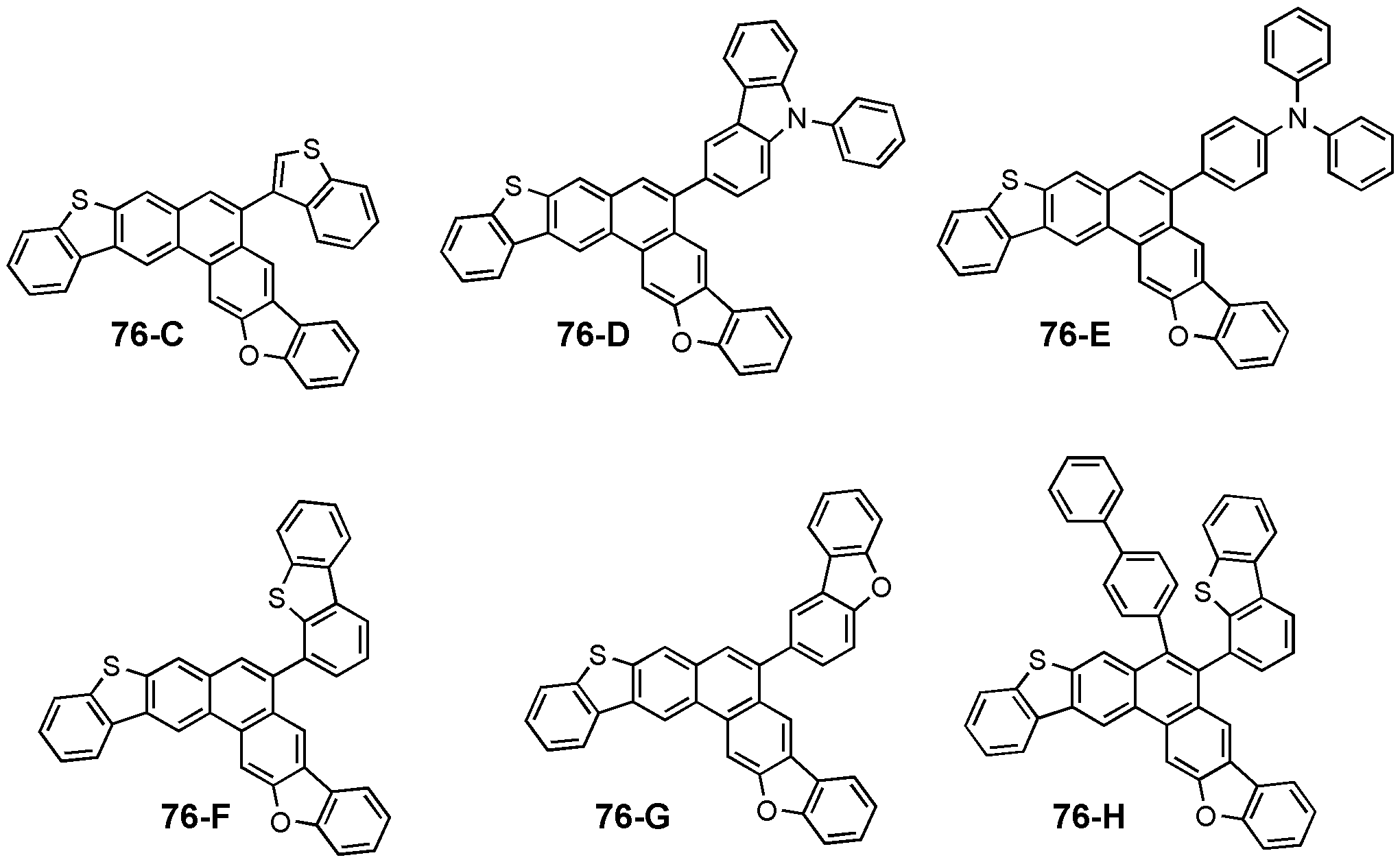
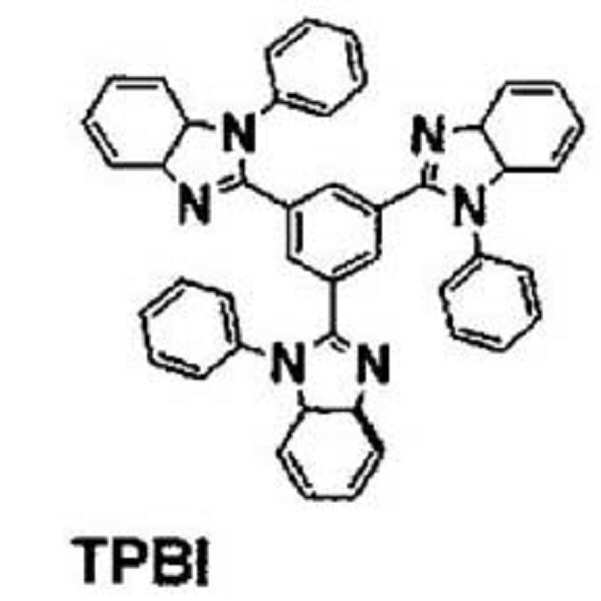
도 2는 일 구현예를 따르는 화학식 1 내지 4의 구조를 나타낸 도면이다.
| 도판트 또는 정공수송 또는 호스트 |
구동전압 (V) |
전류밀도 (㎃/㎠) |
휘도 (cd/㎡) |
효율 (cd/A) |
발광색 | LT97(hr) | |
| 실시예 1 |
청색형광도판트 46-E | 6.21 | 50 | 2,280 | 4.68 | 청색 | 32 |
| 실시예 2 |
정공수송 15-D | 6.18 | 50 | 2,183 | 4.37 | 청색 | 29 |
| 실시예 3 |
정공수송 23-H | 6.03 | 50 | 2,309 | 4.62 | 청색 | 27 |
| 실시예 4 |
정공수송 43-E | 6.32 | 50 | 2,366 | 4.73 | 청색 | 30 |
| 실시예 5 |
녹색인광호스트 15-F | 5.67 | 50 | 16,654 | 33.3 | 녹색 | 84 |
| 실시예 6 |
녹색인광호스트 32-D | 5.33 | 50 | 17,102 | 34.2 | 녹색 | 91 |
| 실시예 7 |
녹색인광호스트 43-D | 5.51 | 50 | 18,248 | 36.5 | 녹색 | 93 |
| 비교예 1 |
DNA/DPBVi | 7.35 | 50 | 1,522 | 3.04 | 청색 | 15 |
| 비교예 2 |
CBP/Irppy | 6.8 | 50 | 10,902 | 21.8 | 녹색 | 60 |
Claims (20)
- 하기 화학식 1, 화학식 2, 화학식 3 또는 화학식 4로 표시되는 헤테로고리 화합물:
<화학식 1>
<화학식 2>
<화학식 3>
<화학식 4>
상기 식 중, R1, R4, R5, R6 및 R8은 각각 독립적으로 수소 또는 중수소이고;
R7은 수소, 중수소, 할로겐기, 시아노기, 치환 또는 비치환된 탄소수 1 내지 60의 알킬기, 치환 또는 비치환된 탄소수 5 내지 60의 아릴기, 치환 또는 비치환된 탄소수 3 내지 60의 헤테로아릴기, 또는 치환 또는 비치환된 탄소수 6 내지 60의 축합다환기를 나타내고,
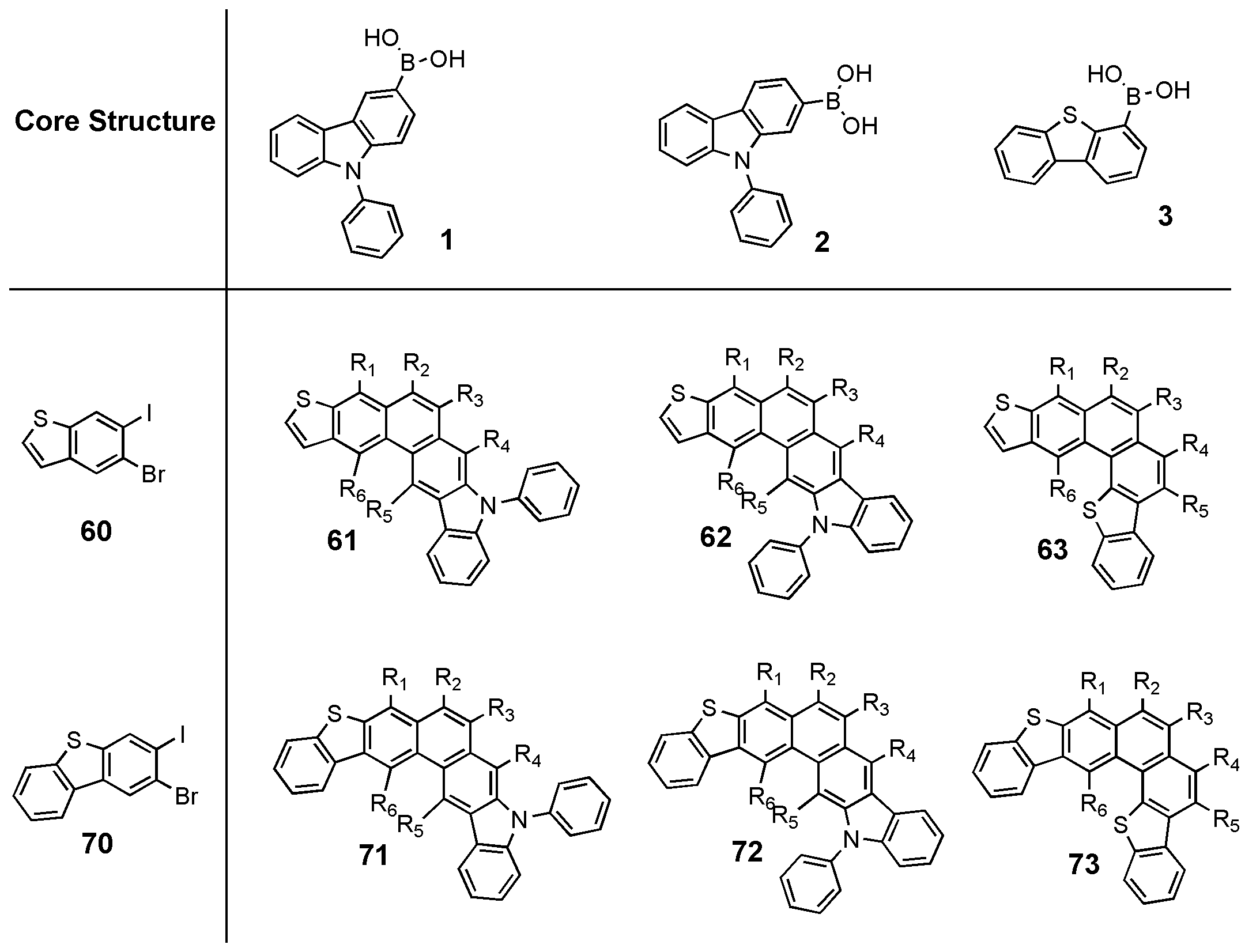
R2 및 R3은 각각 독립적으로 하기 화학식 2a 내지 2f 중 어느 하나이고;
상기 화학식 2a 내지 2f 중,
Q2는 -N(R30)-, -CR31R32-, -S- 또는 -O-로 표시되는 연결기들이고;
Q3는 -S- 또는 -O-로 표시되는 연결기들이고;
Y1 및 Y2는 각각 독립적으로 -N=, -N(R30)- 또는 -C=이고;
Z1, R30, R31 및 R32는 서로 독립적으로, 수소 원자, 중수소, 치환 또는 비치환된 탄소수 1 내지 20의 알킬기, 치환 또는 비치환된 탄소수 5 내지 20의 아릴기, 치환 또는 비치환된 탄소수 3 내지 20의 헤테로아릴기, 치환 또는 비치환된 탄소수 6 내지 20의 축합 다환기, 탄소수 5 내지 20의 아릴기 또는 탄소수 3 내지 20의 헤테로아릴기로 치환된 아미노기, 할로겐기, 시아노기, 니트로기, 하이드록시기 또는 카르복시기이고;
p는 1 내지 9의 정수이고;
*는 결합을 나타내며;
X는 -O-, -S- 또는 -NR20-을 나타내며, R20은 치환 또는 비치환된 탄소수 1 내지 30의 알킬기, 치환 또는 비치환된 탄소수 5 내지 30의 아릴기를 나타내고,
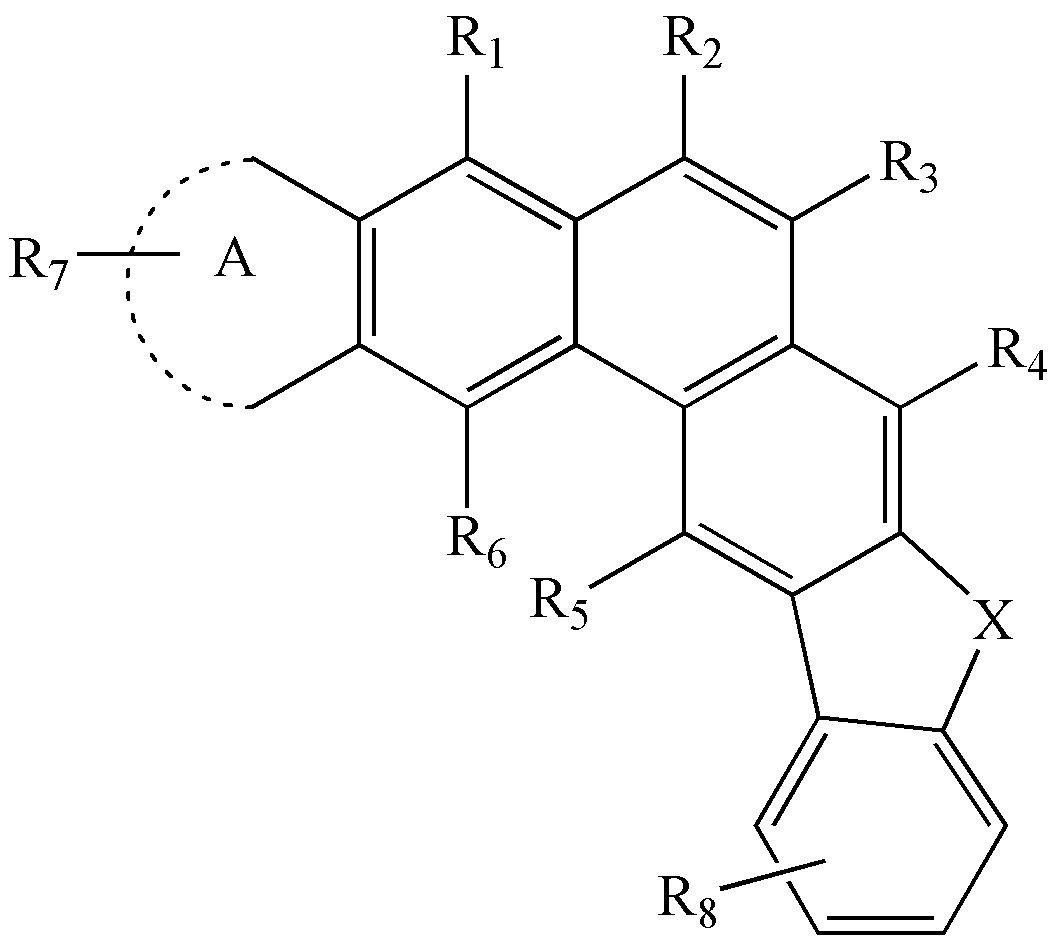
A는 치환 또는 비치환된 퓨란기, 치환 또는 비치환된 벤조퓨란기, 또는 치환 또는 비치환된 피롤기로서, 하기 화학식 11 의 2 위치 및 3 위치 또는 하기 화학식 12의 2 위치 및 3 위치에서 상기 화학식 1 내지 화학식 4의 골격과 융합하는 것을 나타낸다:
<화학식 11>
<화학식 12>
상기 화학식 11 및 12에서 Q1은 -O- 또는 -NR30-를 나타내며,
R30은 수소, 중수소, 치환 또는 비치환된 탄소수 1 내지 20의 알킬기, 치환 또는 비치환된 탄소수 5 내지 20의 아릴기, 치환 또는 비치환된 탄소수 3 내지 20의 헤테로아릴기, 또는 치환 또는 비치환된 탄소수 6 내지 20의 축합다환기를 나타낸다. - 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 제 1 전극;
제 2 전극; 및
상기 제 1 전극 및 제 2 전극 사이에 개재된 유기층을 구비한 유기 발광 소자로서,
상기 유기층이 제 1 항 또는 제 7 항 중 어느 한 항의 화합물을 포함하는 유기 발광 소자. - 제 8 항에 있어서,
상기 유기층이 발광층이며 상기 화합물이 형광 도펀트로 사용되는 유기 발광 소자. - 제 8 항에 있어서,
상기 유기층이 발광층이며 상기 화합물이 인광 호스트로 사용되는 유기 발광 소자. - 제 8 항에 있어서,
상기 유기층이 청색 발광층 또는 녹색 발광층인 유기 발광 소자. - 제 8 항에 있어서,
상기 유기 발광 소자가 발광층, 정공 주입층, 정공 수송층, 또는 정공 주입 및 정공 수송기능을 동시에 갖는 기능층을 포함하고,
상기 발광층, 정공 주입층, 정공 수송층, 또는 정공 주입 및 정공 수송기능을 동시에 갖는 기능층은 제 1 항의 화합물을 포함하며,
상기 발광층은 안트라센계 화합물, 아릴아민계 화합물 또는 스티릴계 화합물을 포함하는 유기 발광 소자. - 제 8 항에 있어서,
상기 유기 발광 소자가 발광층, 정공 주입층, 정공 수송층, 또는 정공 주입 및 정공 수송기능을 동시에 갖는 기능층을 포함하고,
상기 발광층, 정공 주입층, 정공 수송층, 또는 정공 주입 및 정공 수송기능을 동시에 갖는 기능층은 제 1 항의 화합물을 포함하며,
상기 발광층의 적색층, 녹색층, 청색층 또는 흰색층의 어느 한 층은 인광 화합물을 포함하는 유기 발광 소자. - 제 13 항에 있어서,
상기 정공 주입층, 정공 수송층, 또는 정공 주입 기능 및 정공 수송 기능을 동시에 갖는 기능층이 전하 생성 물질을 포함하는 유기 발광 소자. - 제 14 항에 있어서,
상기 전하 생성 물질이 p-도펀트이고, 상기 p-도펀트가 퀴논 유도체, 금속 산화물 또는 시아노기-함유 화합물인 유기 발광 소자. - 제 8 항에 있어서,
상기 유기층이 전자 수송층을 포함하고, 상기 전자 수송층이 전자 수송성 유기 화합물 및 금속 착체를 포함하는 유기 발광 소자. - 제 16 항에 있어서,
상기 금속 착체가 리튬 퀴놀레이트(LiQ)인 유기 발광 소자 - 제 8 항에 있어서,
상기 유기층이 제 1 항 또는 제 7 항 중 어느 한 항의 화합물을 사용하여 습식 공정으로 형성되는 유기 발광 소자. - 제 8 항의 유기 발광 소자를 구비하고, 상기 유기 발광 소자의 제 1 전극이 박막 트랜지스터의 소스 전극 또는 드레인 전극과 전기적으로 연결된 평판 표시 장치.
Priority Applications (5)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020120081965A KR101994837B1 (ko) | 2012-07-26 | 2012-07-26 | 신규 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 |
| US13/690,600 US9118021B2 (en) | 2012-07-26 | 2012-11-30 | Heterocyclic compound and organic light-emitting device including the same |
| CN201310043667.5A CN103570735B (zh) | 2012-07-26 | 2013-02-04 | 新型杂环化合物及包含该杂环化合物的有机发光元件 |
| DE102013202562.9A DE102013202562B4 (de) | 2012-07-26 | 2013-02-18 | Neue heterocyclische verbindung und organische lichtemittierende vorrichtung, die diese aufweist |
| TW102105712A TWI592414B (zh) | 2012-07-26 | 2013-02-19 | 新穎雜環化合物以及包含其之有機發光裝置 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020120081965A KR101994837B1 (ko) | 2012-07-26 | 2012-07-26 | 신규 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20140019082A KR20140019082A (ko) | 2014-02-14 |
| KR101994837B1 true KR101994837B1 (ko) | 2019-07-02 |
Family
ID=49912367
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020120081965A Active KR101994837B1 (ko) | 2012-07-26 | 2012-07-26 | 신규 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 |
Country Status (5)
| Country | Link |
|---|---|
| US (1) | US9118021B2 (ko) |
| KR (1) | KR101994837B1 (ko) |
| CN (1) | CN103570735B (ko) |
| DE (1) | DE102013202562B4 (ko) |
| TW (1) | TWI592414B (ko) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20210014859A (ko) | 2019-07-31 | 2021-02-10 | 한국화학연구원 | N-헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 |
Families Citing this family (10)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR102098737B1 (ko) * | 2012-10-30 | 2020-04-09 | 삼성디스플레이 주식회사 | 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 |
| KR102162791B1 (ko) * | 2013-09-17 | 2020-10-08 | 삼성디스플레이 주식회사 | 유기 발광 소자 |
| WO2015174639A1 (ko) * | 2014-05-12 | 2015-11-19 | 삼성에스디아이 주식회사 | 유기 화합물, 유기 광전자 소자 및 표시 장치 |
| KR101904299B1 (ko) | 2014-05-12 | 2018-10-04 | 제일모직 주식회사 | 유기 화합물, 유기 광전자 소자 및 표시 장치 |
| KR102337550B1 (ko) | 2015-04-16 | 2021-12-13 | 에스에프씨주식회사 | 신규한 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 |
| TWI713625B (zh) * | 2015-11-06 | 2020-12-21 | Lt素材股份有限公司 | 雜環化合物及使用其之有機發光裝置 |
| KR102419178B1 (ko) | 2015-12-29 | 2022-07-11 | 삼성디스플레이 주식회사 | 유기 발광 소자 |
| CN107880055B (zh) * | 2016-09-30 | 2020-08-28 | 南京高光半导体材料有限公司 | 一种蓝色荧光掺杂物质以及包含此物质的有机电致发光器件 |
| CN112552301A (zh) * | 2019-09-26 | 2021-03-26 | 广州华睿光电材料有限公司 | 吲哚醌类有机化合物及其应用 |
| CN113444100A (zh) * | 2020-03-27 | 2021-09-28 | 苏州深通新材料有限公司 | 新型杂环化合物及包含该化合物的有机电子元件 |
Family Cites Families (18)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| DE69511755T2 (de) | 1994-04-26 | 2000-01-13 | Tdk Corp | Phenylanthracenderivat und organisches EL-Element |
| JP3816969B2 (ja) | 1994-04-26 | 2006-08-30 | Tdk株式会社 | 有機el素子 |
| US5645948A (en) | 1996-08-20 | 1997-07-08 | Eastman Kodak Company | Blue organic electroluminescent devices |
| US5972247A (en) | 1998-03-20 | 1999-10-26 | Eastman Kodak Company | Organic electroluminescent elements for stable blue electroluminescent devices |
| JP2000003782A (ja) | 1998-06-12 | 2000-01-07 | Casio Comput Co Ltd | 電界発光素子 |
| EP1009043A3 (en) | 1998-12-09 | 2002-07-03 | Eastman Kodak Company | Electroluminescent device with polyphenyl hydrocarbon hole transport layer |
| US6465115B2 (en) | 1998-12-09 | 2002-10-15 | Eastman Kodak Company | Electroluminescent device with anthracene derivatives hole transport layer |
| KR100787425B1 (ko) * | 2004-11-29 | 2007-12-26 | 삼성에스디아이 주식회사 | 페닐카바졸계 화합물 및 이를 이용한 유기 전계 발광 소자 |
| KR101359631B1 (ko) * | 2006-11-10 | 2014-02-10 | 삼성디스플레이 주식회사 | 안트라센 유도체 화합물 및 이를 이용한 유기 발광 소자 |
| CN101687837A (zh) | 2007-08-06 | 2010-03-31 | 出光兴产株式会社 | 芳族胺衍生物以及使用该衍生物的有机电致发光元件 |
| KR101026171B1 (ko) | 2008-07-01 | 2011-04-05 | 덕산하이메탈(주) | 신규의 축합 카바졸 유도체 및 이를 포함하는 유기전계발광 소자 |
| KR20110043625A (ko) | 2008-07-28 | 2011-04-27 | 이데미쓰 고산 가부시키가이샤 | 유기 발광 매체 및 유기 el 소자 |
| WO2010021524A2 (ko) | 2008-08-22 | 2010-02-25 | 주식회사 엘지화학 | 유기 전자 소자 재료 및 이를 이용한 유기 전자 소자 |
| DE102009005746A1 (de) | 2009-01-23 | 2010-07-29 | Merck Patent Gmbh | Materialien für organische Elektrolumineszenzvorrichtungen |
| KR101108154B1 (ko) * | 2009-08-10 | 2012-02-08 | 삼성모바일디스플레이주식회사 | 축합환 화합물 및 이를 포함한 유기층을 구비한 유기 발광 소자 |
| KR101117722B1 (ko) * | 2009-08-28 | 2012-03-07 | 삼성모바일디스플레이주식회사 | 유기 발광 소자 |
| KR101117724B1 (ko) * | 2009-09-04 | 2012-03-07 | 삼성모바일디스플레이주식회사 | 유기 발광 소자 |
| KR20120081539A (ko) | 2011-01-11 | 2012-07-19 | (주)씨에스엘쏠라 | 유기발광화합물 및 이를 이용한 유기 광소자 |
-
2012
- 2012-07-26 KR KR1020120081965A patent/KR101994837B1/ko active Active
- 2012-11-30 US US13/690,600 patent/US9118021B2/en active Active
-
2013
- 2013-02-04 CN CN201310043667.5A patent/CN103570735B/zh active Active
- 2013-02-18 DE DE102013202562.9A patent/DE102013202562B4/de active Active
- 2013-02-19 TW TW102105712A patent/TWI592414B/zh active
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20210014859A (ko) | 2019-07-31 | 2021-02-10 | 한국화학연구원 | N-헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 |
Also Published As
| Publication number | Publication date |
|---|---|
| TW201404778A (zh) | 2014-02-01 |
| CN103570735B (zh) | 2017-06-09 |
| US20140103300A1 (en) | 2014-04-17 |
| CN103570735A (zh) | 2014-02-12 |
| DE102013202562B4 (de) | 2023-08-10 |
| US9118021B2 (en) | 2015-08-25 |
| DE102013202562A1 (de) | 2014-01-30 |
| KR20140019082A (ko) | 2014-02-14 |
| TWI592414B (zh) | 2017-07-21 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TWI602813B (zh) | 雜環化合物、包含其之有機發光裝置以及包含有機發光裝置的有機發光顯示設備 | |
| KR101994837B1 (ko) | 신규 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 | |
| KR102098737B1 (ko) | 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 | |
| KR102096047B1 (ko) | 신규 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 | |
| KR102098736B1 (ko) | 화합물 및 이를 포함한 유기 발광 소자 | |
| KR20150106501A (ko) | 화합물 및 이를 포함한 유기 발광 소자 | |
| KR20150014778A (ko) | 유기 발광 소자 | |
| KR102203102B1 (ko) | 화합물 및 이를 포함한 유기 발광 소자 | |
| KR101894024B1 (ko) | 헤테로고리 화합물 및 이를 포함하는 유기 발광 소자 | |
| KR20150025259A (ko) | 화합물 및 이를 포함한 유기 발광 소자 | |
| KR20140058755A (ko) | 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 | |
| KR102102350B1 (ko) | 아릴아민 화합물 및 이를 포함한 유기 발광 소자 | |
| KR102086546B1 (ko) | 화합물 및 이를 포함한 유기 발광 소자 | |
| KR20140003259A (ko) | 신규한 유기 발광 화합물 및 이를 포함한 유기 발광 소자 | |
| KR20130000229A (ko) | 헤테로고리 화합물 및 이를 포함한 유기발광 소자 | |
| KR102154705B1 (ko) | 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 | |
| KR102040871B1 (ko) | 신규한 유기 발광 소자용 화합물 및 이를 포함한 유기 발광 소자 | |
| KR102173042B1 (ko) | 헤테로고리 화합물 및 이를 포함한 유기 발광 소자 | |
| KR20150064804A (ko) | 화합물 및 이를 포함한 유기 발광 소자 | |
| KR102278600B1 (ko) | 화합물 및 이를 포함한 유기 발광 소자 | |
| KR102075523B1 (ko) | 신규 축합 화합물 및 이를 포함한 유기 발광 소자 | |
| KR101706742B1 (ko) | 화합물 및 이를 포함한 유기 발광 소자 | |
| KR20140096662A (ko) | 화합물 및 이를 포함한 유기 발광 소자 | |
| KR102006879B1 (ko) | 화합물 및 이를 포함한 유기 발광 소자 | |
| KR20150068144A (ko) | 화합물 및 이를 포함한 유기 발광 소자 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0109 | Patent application |
Patent event code: PA01091R01D Comment text: Patent Application Patent event date: 20120726 |
|
| PG1501 | Laying open of application | ||
| A201 | Request for examination | ||
| PA0201 | Request for examination |
Patent event code: PA02012R01D Patent event date: 20170526 Comment text: Request for Examination of Application Patent event code: PA02011R01I Patent event date: 20120726 Comment text: Patent Application |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20181114 Patent event code: PE09021S01D |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20190401 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20190625 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20190626 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration | ||
| PR1001 | Payment of annual fee |
Payment date: 20220523 Start annual number: 4 End annual number: 4 |
|
| PR1001 | Payment of annual fee |
Payment date: 20230524 Start annual number: 5 End annual number: 5 |
|
| PR1001 | Payment of annual fee |
Payment date: 20240523 Start annual number: 6 End annual number: 6 |