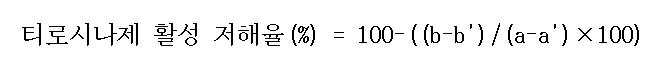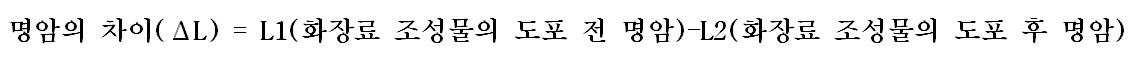KR101452770B1 - 유산균 발효액을 함유하는 항산화, 미백 및 주름개선 효과를 갖는 화장료 조성물 - Google Patents
유산균 발효액을 함유하는 항산화, 미백 및 주름개선 효과를 갖는 화장료 조성물 Download PDFInfo
- Publication number
- KR101452770B1 KR101452770B1 KR1020130019841A KR20130019841A KR101452770B1 KR 101452770 B1 KR101452770 B1 KR 101452770B1 KR 1020130019841 A KR1020130019841 A KR 1020130019841A KR 20130019841 A KR20130019841 A KR 20130019841A KR 101452770 B1 KR101452770 B1 KR 101452770B1
- Authority
- KR
- South Korea
- Prior art keywords
- lactic acid
- root
- cosmetic composition
- fermentation broth
- brix
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 239000000203 mixture Substances 0.000 title claims abstract description 93
- 239000002537 cosmetic Substances 0.000 title claims abstract description 84
- 230000002087 whitening effect Effects 0.000 title abstract description 25
- 241000186660 Lactobacillus Species 0.000 title description 4
- 229940039696 lactobacillus Drugs 0.000 title description 4
- 230000003064 anti-oxidating effect Effects 0.000 title description 2
- 230000001153 anti-wrinkle effect Effects 0.000 title 1
- JVTAAEKCZFNVCJ-UHFFFAOYSA-N lactic acid Chemical compound CC(O)C(O)=O JVTAAEKCZFNVCJ-UHFFFAOYSA-N 0.000 claims abstract description 154
- 238000000855 fermentation Methods 0.000 claims abstract description 78
- 230000004151 fermentation Effects 0.000 claims abstract description 78
- 239000004310 lactic acid Substances 0.000 claims abstract description 77
- 235000014655 lactic acid Nutrition 0.000 claims abstract description 77
- 241000894006 Bacteria Species 0.000 claims abstract description 43
- 239000000284 extract Substances 0.000 claims abstract description 38
- 230000000694 effects Effects 0.000 claims abstract description 37
- 230000003078 antioxidant effect Effects 0.000 claims abstract description 34
- 241000219991 Lythraceae Species 0.000 claims abstract description 32
- 235000014360 Punica granatum Nutrition 0.000 claims abstract description 31
- 230000037303 wrinkles Effects 0.000 claims abstract description 14
- 244000020518 Carthamus tinctorius Species 0.000 claims abstract description 12
- 235000003255 Carthamus tinctorius Nutrition 0.000 claims abstract description 12
- 241001672694 Citrus reticulata Species 0.000 claims abstract description 6
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 claims description 30
- 240000002853 Nelumbo nucifera Species 0.000 claims description 25
- 235000006508 Nelumbo nucifera Nutrition 0.000 claims description 25
- 238000001914 filtration Methods 0.000 claims description 14
- 238000000034 method Methods 0.000 claims description 13
- 241000196324 Embryophyta Species 0.000 claims description 11
- 238000009472 formulation Methods 0.000 claims description 10
- 210000004209 hair Anatomy 0.000 claims description 10
- 238000010564 aerobic fermentation Methods 0.000 claims description 7
- 230000001580 bacterial effect Effects 0.000 claims description 5
- 238000002156 mixing Methods 0.000 claims description 5
- 239000006872 mrs medium Substances 0.000 claims description 5
- 241000186610 Lactobacillus sp. Species 0.000 claims description 4
- 230000008569 process Effects 0.000 claims description 4
- 238000010298 pulverizing process Methods 0.000 claims description 3
- 230000035784 germination Effects 0.000 claims description 2
- 244000194101 Ginkgo biloba Species 0.000 claims 1
- 239000004599 antimicrobial Substances 0.000 claims 1
- 239000000243 solution Substances 0.000 description 40
- 230000000052 comparative effect Effects 0.000 description 32
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 19
- 238000005259 measurement Methods 0.000 description 16
- 238000012360 testing method Methods 0.000 description 16
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 15
- 238000002835 absorbance Methods 0.000 description 14
- FPIPGXGPPPQFEQ-OVSJKPMPSA-N all-trans-retinol Chemical compound OC\C=C(/C)\C=C\C=C(/C)\C=C\C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-OVSJKPMPSA-N 0.000 description 14
- 210000004027 cell Anatomy 0.000 description 13
- -1 goblet Natural products 0.000 description 11
- 238000004519 manufacturing process Methods 0.000 description 11
- 235000013824 polyphenols Nutrition 0.000 description 11
- 230000001603 reducing effect Effects 0.000 description 11
- CIWBSHSKHKDKBQ-JLAZNSOCSA-N Ascorbic acid Chemical compound OC[C@H](O)[C@H]1OC(=O)C(O)=C1O CIWBSHSKHKDKBQ-JLAZNSOCSA-N 0.000 description 10
- 230000005764 inhibitory process Effects 0.000 description 10
- 239000000463 material Substances 0.000 description 10
- 239000012528 membrane Substances 0.000 description 10
- 239000011148 porous material Substances 0.000 description 10
- 235000006510 Nelumbo pentapetala Nutrition 0.000 description 9
- 240000004808 Saccharomyces cerevisiae Species 0.000 description 9
- 102000003425 Tyrosinase Human genes 0.000 description 9
- 108060008724 Tyrosinase Proteins 0.000 description 9
- 238000004458 analytical method Methods 0.000 description 9
- 239000012153 distilled water Substances 0.000 description 9
- 239000012530 fluid Substances 0.000 description 9
- 235000000346 sugar Nutrition 0.000 description 9
- FPIPGXGPPPQFEQ-UHFFFAOYSA-N 13-cis retinol Natural products OCC=C(C)C=CC=C(C)C=CC1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-UHFFFAOYSA-N 0.000 description 8
- 239000003963 antioxidant agent Substances 0.000 description 8
- 235000006708 antioxidants Nutrition 0.000 description 8
- 229930003935 flavonoid Natural products 0.000 description 8
- 235000017173 flavonoids Nutrition 0.000 description 8
- 230000002401 inhibitory effect Effects 0.000 description 8
- 102000029816 Collagenase Human genes 0.000 description 7
- 108060005980 Collagenase Proteins 0.000 description 7
- 229960002424 collagenase Drugs 0.000 description 7
- 239000012091 fetal bovine serum Substances 0.000 description 7
- 150000002215 flavonoids Chemical class 0.000 description 7
- 239000000523 sample Substances 0.000 description 7
- 239000002609 medium Substances 0.000 description 6
- 244000005700 microbiome Species 0.000 description 6
- 229960003471 retinol Drugs 0.000 description 6
- 235000020944 retinol Nutrition 0.000 description 6
- 239000011607 retinol Substances 0.000 description 6
- 239000007858 starting material Substances 0.000 description 6
- 239000000126 substance Substances 0.000 description 6
- 241000186612 Lactobacillus sakei Species 0.000 description 5
- 102100033220 Xanthine oxidase Human genes 0.000 description 5
- 108010093894 Xanthine oxidase Proteins 0.000 description 5
- 235000010323 ascorbic acid Nutrition 0.000 description 5
- 239000011668 ascorbic acid Substances 0.000 description 5
- 229960005070 ascorbic acid Drugs 0.000 description 5
- 239000003153 chemical reaction reagent Substances 0.000 description 5
- 239000006071 cream Substances 0.000 description 5
- 239000007788 liquid Substances 0.000 description 5
- ISWSIDIOOBJBQZ-UHFFFAOYSA-N phenol group Chemical group C1(=CC=CC=C1)O ISWSIDIOOBJBQZ-UHFFFAOYSA-N 0.000 description 5
- 239000001397 quillaja saponaria molina bark Substances 0.000 description 5
- 229940119485 safflower extract Drugs 0.000 description 5
- 229930182490 saponin Natural products 0.000 description 5
- 150000007949 saponins Chemical class 0.000 description 5
- LWFUFLREGJMOIZ-UHFFFAOYSA-N 3,5-dinitrosalicylic acid Chemical compound OC(=O)C1=CC([N+]([O-])=O)=CC([N+]([O-])=O)=C1O LWFUFLREGJMOIZ-UHFFFAOYSA-N 0.000 description 4
- 108010035532 Collagen Proteins 0.000 description 4
- 102000008186 Collagen Human genes 0.000 description 4
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 4
- 239000002253 acid Substances 0.000 description 4
- SQVRNKJHWKZAKO-UHFFFAOYSA-N beta-N-Acetyl-D-neuraminic acid Natural products CC(=O)NC1C(O)CC(O)(C(O)=O)OC1C(O)C(O)CO SQVRNKJHWKZAKO-UHFFFAOYSA-N 0.000 description 4
- 229920001436 collagen Polymers 0.000 description 4
- 230000003013 cytotoxicity Effects 0.000 description 4
- 231100000135 cytotoxicity Toxicity 0.000 description 4
- MTHSVFCYNBDYFN-UHFFFAOYSA-N diethylene glycol Chemical compound OCCOCCO MTHSVFCYNBDYFN-UHFFFAOYSA-N 0.000 description 4
- 239000000706 filtrate Substances 0.000 description 4
- BJRNKVDFDLYUGJ-RMPHRYRLSA-N hydroquinone O-beta-D-glucopyranoside Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@H]1OC1=CC=C(O)C=C1 BJRNKVDFDLYUGJ-RMPHRYRLSA-N 0.000 description 4
- 230000003020 moisturizing effect Effects 0.000 description 4
- SQVRNKJHWKZAKO-OQPLDHBCSA-N sialic acid Chemical compound CC(=O)N[C@@H]1[C@@H](O)C[C@@](O)(C(O)=O)OC1[C@H](O)[C@H](O)CO SQVRNKJHWKZAKO-OQPLDHBCSA-N 0.000 description 4
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 description 3
- CSCPPACGZOOCGX-UHFFFAOYSA-N Acetone Chemical compound CC(C)=O CSCPPACGZOOCGX-UHFFFAOYSA-N 0.000 description 3
- UHOVQNZJYSORNB-UHFFFAOYSA-N Benzene Chemical compound C1=CC=CC=C1 UHOVQNZJYSORNB-UHFFFAOYSA-N 0.000 description 3
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 description 3
- LYCAIKOWRPUZTN-UHFFFAOYSA-N Ethylene glycol Chemical compound OCCO LYCAIKOWRPUZTN-UHFFFAOYSA-N 0.000 description 3
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 description 3
- DNIAPMSPPWPWGF-UHFFFAOYSA-N Propylene glycol Chemical compound CC(O)CO DNIAPMSPPWPWGF-UHFFFAOYSA-N 0.000 description 3
- 230000002292 Radical scavenging effect Effects 0.000 description 3
- 239000004480 active ingredient Substances 0.000 description 3
- 230000003833 cell viability Effects 0.000 description 3
- 230000008859 change Effects 0.000 description 3
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 description 3
- 230000037319 collagen production Effects 0.000 description 3
- 230000001186 cumulative effect Effects 0.000 description 3
- MGJZITXUQXWAKY-UHFFFAOYSA-N diphenyl-(2,4,6-trinitrophenyl)iminoazanium Chemical compound [O-][N+](=O)C1=CC([N+](=O)[O-])=CC([N+]([O-])=O)=C1N=[N+](C=1C=CC=CC=1)C1=CC=CC=C1 MGJZITXUQXWAKY-UHFFFAOYSA-N 0.000 description 3
- 239000000686 essence Substances 0.000 description 3
- 239000008103 glucose Substances 0.000 description 3
- 230000006872 improvement Effects 0.000 description 3
- 230000001965 increasing effect Effects 0.000 description 3
- 239000006210 lotion Substances 0.000 description 3
- VLKZOEOYAKHREP-UHFFFAOYSA-N n-Hexane Chemical compound CCCCCC VLKZOEOYAKHREP-UHFFFAOYSA-N 0.000 description 3
- JPXMTWWFLBLUCD-UHFFFAOYSA-N nitro blue tetrazolium(2+) Chemical compound COC1=CC(C=2C=C(OC)C(=CC=2)[N+]=2N(N=C(N=2)C=2C=CC=CC=2)C=2C=CC(=CC=2)[N+]([O-])=O)=CC=C1[N+]1=NC(C=2C=CC=CC=2)=NN1C1=CC=C([N+]([O-])=O)C=C1 JPXMTWWFLBLUCD-UHFFFAOYSA-N 0.000 description 3
- 235000015097 nutrients Nutrition 0.000 description 3
- 239000005416 organic matter Substances 0.000 description 3
- 239000008363 phosphate buffer Substances 0.000 description 3
- 238000011160 research Methods 0.000 description 3
- 238000011076 safety test Methods 0.000 description 3
- 210000002235 sarcomere Anatomy 0.000 description 3
- 239000000934 spermatocidal agent Substances 0.000 description 3
- 230000000638 stimulation Effects 0.000 description 3
- 230000004083 survival effect Effects 0.000 description 3
- 239000011534 wash buffer Substances 0.000 description 3
- 230000037331 wrinkle reduction Effects 0.000 description 3
- OVBFMEVBMNZIBR-UHFFFAOYSA-N -2-Methylpentanoic acid Natural products CCCC(C)C(O)=O OVBFMEVBMNZIBR-UHFFFAOYSA-N 0.000 description 2
- LRFVTYWOQMYALW-UHFFFAOYSA-N 9H-xanthine Chemical compound O=C1NC(=O)NC2=C1NC=N2 LRFVTYWOQMYALW-UHFFFAOYSA-N 0.000 description 2
- RSWGJHLUYNHPMX-UHFFFAOYSA-N Abietic-Saeure Natural products C12CCC(C(C)C)=CC2=CCC2C1(C)CCCC2(C)C(O)=O RSWGJHLUYNHPMX-UHFFFAOYSA-N 0.000 description 2
- HEDRZPFGACZZDS-UHFFFAOYSA-N Chloroform Chemical compound ClC(Cl)Cl HEDRZPFGACZZDS-UHFFFAOYSA-N 0.000 description 2
- 238000002965 ELISA Methods 0.000 description 2
- 102000004190 Enzymes Human genes 0.000 description 2
- 108090000790 Enzymes Proteins 0.000 description 2
- 206010015150 Erythema Diseases 0.000 description 2
- PEDCQBHIVMGVHV-UHFFFAOYSA-N Glycerine Chemical compound OCC(O)CO PEDCQBHIVMGVHV-UHFFFAOYSA-N 0.000 description 2
- OUYCCCASQSFEME-QMMMGPOBSA-N L-tyrosine Chemical compound OC(=O)[C@@H](N)CC1=CC=C(O)C=C1 OUYCCCASQSFEME-QMMMGPOBSA-N 0.000 description 2
- 102000000380 Matrix Metalloproteinase 1 Human genes 0.000 description 2
- 108010016113 Matrix Metalloproteinase 1 Proteins 0.000 description 2
- 229920000715 Mucilage Polymers 0.000 description 2
- JEIZLWNUBXHADF-UHFFFAOYSA-N Pelletierine Chemical compound CC(=O)CC1CCCCN1 JEIZLWNUBXHADF-UHFFFAOYSA-N 0.000 description 2
- 244000184734 Pyrus japonica Species 0.000 description 2
- KHPCPRHQVVSZAH-HUOMCSJISA-N Rosin Natural products O(C/C=C/c1ccccc1)[C@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 KHPCPRHQVVSZAH-HUOMCSJISA-N 0.000 description 2
- FPIPGXGPPPQFEQ-BOOMUCAASA-N Vitamin A Natural products OC/C=C(/C)\C=C\C=C(\C)/C=C/C1=C(C)CCCC1(C)C FPIPGXGPPPQFEQ-BOOMUCAASA-N 0.000 description 2
- 239000000853 adhesive Substances 0.000 description 2
- 229930013930 alkaloid Natural products 0.000 description 2
- 239000003708 ampul Substances 0.000 description 2
- 230000003712 anti-aging effect Effects 0.000 description 2
- 229960000271 arbutin Drugs 0.000 description 2
- 239000006227 byproduct Substances 0.000 description 2
- 238000004113 cell culture Methods 0.000 description 2
- 239000003795 chemical substances by application Substances 0.000 description 2
- 239000012141 concentrate Substances 0.000 description 2
- 235000008504 concentrate Nutrition 0.000 description 2
- 230000001419 dependent effect Effects 0.000 description 2
- 238000010790 dilution Methods 0.000 description 2
- 239000012895 dilution Substances 0.000 description 2
- XBDQKXXYIPTUBI-UHFFFAOYSA-N dimethylselenoniopropionate Natural products CCC(O)=O XBDQKXXYIPTUBI-UHFFFAOYSA-N 0.000 description 2
- POULHZVOKOAJMA-UHFFFAOYSA-N dodecanoic acid Chemical compound CCCCCCCCCCCC(O)=O POULHZVOKOAJMA-UHFFFAOYSA-N 0.000 description 2
- 235000013399 edible fruits Nutrition 0.000 description 2
- 229940088598 enzyme Drugs 0.000 description 2
- 210000002950 fibroblast Anatomy 0.000 description 2
- 235000013305 food Nutrition 0.000 description 2
- 210000000245 forearm Anatomy 0.000 description 2
- LNTHITQWFMADLM-UHFFFAOYSA-N gallic acid Chemical compound OC(=O)C1=CC(O)=C(O)C(O)=C1 LNTHITQWFMADLM-UHFFFAOYSA-N 0.000 description 2
- 230000035876 healing Effects 0.000 description 2
- 241000411851 herbal medicine Species 0.000 description 2
- 238000011534 incubation Methods 0.000 description 2
- 230000000968 intestinal effect Effects 0.000 description 2
- 239000002075 main ingredient Substances 0.000 description 2
- VNWKTOKETHGBQD-UHFFFAOYSA-N methane Chemical compound C VNWKTOKETHGBQD-UHFFFAOYSA-N 0.000 description 2
- 210000003097 mucus Anatomy 0.000 description 2
- 229930014626 natural product Natural products 0.000 description 2
- BJRNKVDFDLYUGJ-UHFFFAOYSA-N p-hydroxyphenyl beta-D-alloside Natural products OC1C(O)C(O)C(CO)OC1OC1=CC=C(O)C=C1 BJRNKVDFDLYUGJ-UHFFFAOYSA-N 0.000 description 2
- KJFMBFZCATUALV-UHFFFAOYSA-N phenolphthalein Chemical compound C1=CC(O)=CC=C1C1(C=2C=CC(O)=CC=2)C2=CC=CC=C2C(=O)O1 KJFMBFZCATUALV-UHFFFAOYSA-N 0.000 description 2
- 239000013641 positive control Substances 0.000 description 2
- 230000002335 preservative effect Effects 0.000 description 2
- 108090000623 proteins and genes Proteins 0.000 description 2
- 239000012488 sample solution Substances 0.000 description 2
- 230000009759 skin aging Effects 0.000 description 2
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 2
- 239000006228 supernatant Substances 0.000 description 2
- 230000001256 tonic effect Effects 0.000 description 2
- KHPCPRHQVVSZAH-UHFFFAOYSA-N trans-cinnamyl beta-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OCC=CC1=CC=CC=C1 KHPCPRHQVVSZAH-UHFFFAOYSA-N 0.000 description 2
- NQPDZGIKBAWPEJ-UHFFFAOYSA-N valeric acid Chemical compound CCCCC(O)=O NQPDZGIKBAWPEJ-UHFFFAOYSA-N 0.000 description 2
- 235000019155 vitamin A Nutrition 0.000 description 2
- 239000011719 vitamin A Substances 0.000 description 2
- 229940045997 vitamin a Drugs 0.000 description 2
- PUPZLCDOIYMWBV-UHFFFAOYSA-N (+/-)-1,3-Butanediol Chemical compound CC(O)CCO PUPZLCDOIYMWBV-UHFFFAOYSA-N 0.000 description 1
- KIUKXJAPPMFGSW-DNGZLQJQSA-N (2S,3S,4S,5R,6R)-6-[(2S,3R,4R,5S,6R)-3-Acetamido-2-[(2S,3S,4R,5R,6R)-6-[(2R,3R,4R,5S,6R)-3-acetamido-2,5-dihydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-2-carboxy-4,5-dihydroxyoxan-3-yl]oxy-5-hydroxy-6-(hydroxymethyl)oxan-4-yl]oxy-3,4,5-trihydroxyoxane-2-carboxylic acid Chemical compound CC(=O)N[C@H]1[C@H](O)O[C@H](CO)[C@@H](O)[C@@H]1O[C@H]1[C@H](O)[C@@H](O)[C@H](O[C@H]2[C@@H]([C@@H](O[C@H]3[C@@H]([C@@H](O)[C@H](O)[C@H](O3)C(O)=O)O)[C@H](O)[C@@H](CO)O2)NC(C)=O)[C@@H](C(O)=O)O1 KIUKXJAPPMFGSW-DNGZLQJQSA-N 0.000 description 1
- WRIDQFICGBMAFQ-UHFFFAOYSA-N (E)-8-Octadecenoic acid Natural products CCCCCCCCCC=CCCCCCCC(O)=O WRIDQFICGBMAFQ-UHFFFAOYSA-N 0.000 description 1
- LQJBNNIYVWPHFW-UHFFFAOYSA-N 20:1omega9c fatty acid Natural products CCCCCCCCCCC=CCCCCCCCC(O)=O LQJBNNIYVWPHFW-UHFFFAOYSA-N 0.000 description 1
- UAIUNKRWKOVEES-UHFFFAOYSA-N 3,3',5,5'-tetramethylbenzidine Chemical compound CC1=C(N)C(C)=CC(C=2C=C(C)C(N)=C(C)C=2)=C1 UAIUNKRWKOVEES-UHFFFAOYSA-N 0.000 description 1
- FTZIQBGFCYJWKA-UHFFFAOYSA-N 3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyltetrazolium Chemical compound S1C(C)=C(C)N=C1[N+]1=NC(C=2C=CC=CC=2)=NN1C1=CC=CC=C1 FTZIQBGFCYJWKA-UHFFFAOYSA-N 0.000 description 1
- VOUAQYXWVJDEQY-QENPJCQMSA-N 33017-11-7 Chemical compound OC(=O)CC[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](C(C)C)C(=O)NCC(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(=O)NCC(=O)NCC(=O)NCC(=O)N1CCC[C@H]1C(=O)NCC(=O)N[C@@H](C)C(=O)NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(=O)N1[C@H](C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(O)=O)C(=O)NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O)CCC1 VOUAQYXWVJDEQY-QENPJCQMSA-N 0.000 description 1
- MIJYXULNPSFWEK-GTOFXWBISA-N 3beta-hydroxyolean-12-en-28-oic acid Chemical compound C1C[C@H](O)C(C)(C)[C@@H]2CC[C@@]3(C)[C@]4(C)CC[C@@]5(C(O)=O)CCC(C)(C)C[C@H]5C4=CC[C@@H]3[C@]21C MIJYXULNPSFWEK-GTOFXWBISA-N 0.000 description 1
- QSBYPNXLFMSGKH-UHFFFAOYSA-N 9-Heptadecensaeure Natural products CCCCCCCC=CCCCCCCCC(O)=O QSBYPNXLFMSGKH-UHFFFAOYSA-N 0.000 description 1
- 235000001674 Agaricus brunnescens Nutrition 0.000 description 1
- GUBGYTABKSRVRQ-XLOQQCSPSA-N Alpha-Lactose Chemical compound O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@H]1O[C@@H]1[C@@H](CO)O[C@H](O)[C@H](O)[C@H]1O GUBGYTABKSRVRQ-XLOQQCSPSA-N 0.000 description 1
- 239000004382 Amylase Substances 0.000 description 1
- 108010065511 Amylases Proteins 0.000 description 1
- 102000013142 Amylases Human genes 0.000 description 1
- 206010003645 Atopy Diseases 0.000 description 1
- JMGZEFIQIZZSBH-UHFFFAOYSA-N Bioquercetin Natural products CC1OC(OCC(O)C2OC(OC3=C(Oc4cc(O)cc(O)c4C3=O)c5ccc(O)c(O)c5)C(O)C2O)C(O)C(O)C1O JMGZEFIQIZZSBH-UHFFFAOYSA-N 0.000 description 1
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 1
- 241000195940 Bryophyta Species 0.000 description 1
- 108010075254 C-Peptide Proteins 0.000 description 1
- 108010059892 Cellulase Proteins 0.000 description 1
- 108010065152 Coagulase Proteins 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- CKLJMWTZIZZHCS-UHFFFAOYSA-N D-OH-Asp Natural products OC(=O)C(N)CC(O)=O CKLJMWTZIZZHCS-UHFFFAOYSA-N 0.000 description 1
- 206010012735 Diarrhoea Diseases 0.000 description 1
- 238000011891 EIA kit Methods 0.000 description 1
- 208000004232 Enteritis Diseases 0.000 description 1
- 101710128038 Hyaluronan synthase Proteins 0.000 description 1
- 206010020649 Hyperkeratosis Diseases 0.000 description 1
- 206010061218 Inflammation Diseases 0.000 description 1
- CKLJMWTZIZZHCS-REOHCLBHSA-N L-aspartic acid Chemical compound OC(=O)[C@@H](N)CC(O)=O CKLJMWTZIZZHCS-REOHCLBHSA-N 0.000 description 1
- 241001147723 Lactobacillus japonicus Species 0.000 description 1
- GUBGYTABKSRVRQ-QKKXKWKRSA-N Lactose Natural products OC[C@H]1O[C@@H](O[C@H]2[C@H](O)[C@@H](O)C(O)O[C@@H]2CO)[C@H](O)[C@@H](O)[C@H]1O GUBGYTABKSRVRQ-QKKXKWKRSA-N 0.000 description 1
- 239000005639 Lauric acid Substances 0.000 description 1
- 108090001060 Lipase Proteins 0.000 description 1
- 102000004882 Lipase Human genes 0.000 description 1
- 239000004367 Lipase Substances 0.000 description 1
- 241000045552 Longidorus orientalis Species 0.000 description 1
- 241001633628 Lycoris Species 0.000 description 1
- 101150101095 Mmp12 gene Proteins 0.000 description 1
- 102000016943 Muramidase Human genes 0.000 description 1
- 108010014251 Muramidase Proteins 0.000 description 1
- 108010062010 N-Acetylmuramoyl-L-alanine Amidase Proteins 0.000 description 1
- 241000244206 Nematoda Species 0.000 description 1
- MIJYXULNPSFWEK-UHFFFAOYSA-N Oleanolinsaeure Natural products C1CC(O)C(C)(C)C2CCC3(C)C4(C)CCC5(C(O)=O)CCC(C)(C)CC5C4=CCC3C21C MIJYXULNPSFWEK-UHFFFAOYSA-N 0.000 description 1
- 239000005642 Oleic acid Substances 0.000 description 1
- ZQPPMHVWECSIRJ-UHFFFAOYSA-N Oleic acid Natural products CCCCCCCCC=CCCCCCCCC(O)=O ZQPPMHVWECSIRJ-UHFFFAOYSA-N 0.000 description 1
- 208000005141 Otitis Diseases 0.000 description 1
- 241001504519 Papio ursinus Species 0.000 description 1
- 229930182555 Penicillin Natural products 0.000 description 1
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 1
- 108091005804 Peptidases Proteins 0.000 description 1
- 108010050808 Procollagen Proteins 0.000 description 1
- 239000004365 Protease Substances 0.000 description 1
- 244000018795 Prunus mume Species 0.000 description 1
- 235000011158 Prunus mume Nutrition 0.000 description 1
- 208000003251 Pruritus Diseases 0.000 description 1
- 102100037486 Reverse transcriptase/ribonuclease H Human genes 0.000 description 1
- 244000178231 Rosmarinus officinalis Species 0.000 description 1
- 241001125046 Sardina pilchardus Species 0.000 description 1
- 206010040880 Skin irritation Diseases 0.000 description 1
- 206010040914 Skin reaction Diseases 0.000 description 1
- CDBYLPFSWZWCQE-UHFFFAOYSA-L Sodium Carbonate Chemical class [Na+].[Na+].[O-]C([O-])=O CDBYLPFSWZWCQE-UHFFFAOYSA-L 0.000 description 1
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 1
- 240000002871 Tectona grandis Species 0.000 description 1
- 244000269722 Thea sinensis Species 0.000 description 1
- 235000006468 Thea sinensis Nutrition 0.000 description 1
- 230000003187 abdominal effect Effects 0.000 description 1
- 150000007513 acids Chemical class 0.000 description 1
- 230000009471 action Effects 0.000 description 1
- 235000019418 amylase Nutrition 0.000 description 1
- 230000004099 anaerobic respiration Effects 0.000 description 1
- 230000000844 anti-bacterial effect Effects 0.000 description 1
- 230000003110 anti-inflammatory effect Effects 0.000 description 1
- 238000003556 assay Methods 0.000 description 1
- 230000003796 beauty Effects 0.000 description 1
- 229940076810 beta sitosterol Drugs 0.000 description 1
- WQZGKKKJIJFFOK-VFUOTHLCSA-N beta-D-glucose Chemical compound OC[C@H]1O[C@@H](O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-VFUOTHLCSA-N 0.000 description 1
- LGJMUZUPVCAVPU-UHFFFAOYSA-N beta-Sitostanol Natural products C1CC2CC(O)CCC2(C)C2C1C1CCC(C(C)CCC(CC)C(C)C)C1(C)CC2 LGJMUZUPVCAVPU-UHFFFAOYSA-N 0.000 description 1
- NJKOMDUNNDKEAI-UHFFFAOYSA-N beta-sitosterol Natural products CCC(CCC(C)C1CCC2(C)C3CC=C4CC(O)CCC4C3CCC12C)C(C)C NJKOMDUNNDKEAI-UHFFFAOYSA-N 0.000 description 1
- 239000012496 blank sample Substances 0.000 description 1
- 238000009835 boiling Methods 0.000 description 1
- 239000000872 buffer Substances 0.000 description 1
- 125000004432 carbon atom Chemical group C* 0.000 description 1
- 239000006143 cell culture medium Substances 0.000 description 1
- 239000013592 cell lysate Substances 0.000 description 1
- 230000004663 cell proliferation Effects 0.000 description 1
- 229940106157 cellulase Drugs 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 238000004040 coloring Methods 0.000 description 1
- 239000000470 constituent Substances 0.000 description 1
- 238000012258 culturing Methods 0.000 description 1
- 230000003247 decreasing effect Effects 0.000 description 1
- 238000011161 development Methods 0.000 description 1
- 230000018109 developmental process Effects 0.000 description 1
- 102000038379 digestive enzymes Human genes 0.000 description 1
- 108091007734 digestive enzymes Proteins 0.000 description 1
- 239000012470 diluted sample Substances 0.000 description 1
- 238000007865 diluting Methods 0.000 description 1
- 238000007598 dipping method Methods 0.000 description 1
- 239000003814 drug Substances 0.000 description 1
- 208000001848 dysentery Diseases 0.000 description 1
- 208000019258 ear infection Diseases 0.000 description 1
- 239000000839 emulsion Substances 0.000 description 1
- 230000002708 enhancing effect Effects 0.000 description 1
- IVTMALDHFAHOGL-UHFFFAOYSA-N eriodictyol 7-O-rutinoside Natural products OC1C(O)C(O)C(C)OC1OCC1C(O)C(O)C(O)C(OC=2C=C3C(C(C(O)=C(O3)C=3C=C(O)C(O)=CC=3)=O)=C(O)C=2)O1 IVTMALDHFAHOGL-UHFFFAOYSA-N 0.000 description 1
- 231100000321 erythema Toxicity 0.000 description 1
- 210000003743 erythrocyte Anatomy 0.000 description 1
- 238000000605 extraction Methods 0.000 description 1
- 210000004709 eyebrow Anatomy 0.000 description 1
- 235000021107 fermented food Nutrition 0.000 description 1
- 239000012737 fresh medium Substances 0.000 description 1
- 235000011389 fruit/vegetable juice Nutrition 0.000 description 1
- 235000004515 gallic acid Nutrition 0.000 description 1
- 229940074391 gallic acid Drugs 0.000 description 1
- 150000004676 glycans Chemical class 0.000 description 1
- 235000011187 glycerol Nutrition 0.000 description 1
- 229940093915 gynecological organic acid Drugs 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 235000008216 herbs Nutrition 0.000 description 1
- 229920002674 hyaluronan Polymers 0.000 description 1
- 229960003160 hyaluronic acid Drugs 0.000 description 1
- 230000004054 inflammatory process Effects 0.000 description 1
- 210000004347 intestinal mucosa Anatomy 0.000 description 1
- 210000000936 intestine Anatomy 0.000 description 1
- QXJSBBXBKPUZAA-UHFFFAOYSA-N isooleic acid Natural products CCCCCCCC=CCCCCCCCCC(O)=O QXJSBBXBKPUZAA-UHFFFAOYSA-N 0.000 description 1
- 230000007803 itching Effects 0.000 description 1
- 230000002147 killing effect Effects 0.000 description 1
- 235000021109 kimchi Nutrition 0.000 description 1
- 239000008101 lactose Substances 0.000 description 1
- 239000007934 lip balm Substances 0.000 description 1
- 235000019421 lipase Nutrition 0.000 description 1
- 239000004325 lysozyme Substances 0.000 description 1
- 235000010335 lysozyme Nutrition 0.000 description 1
- 229960000274 lysozyme Drugs 0.000 description 1
- 210000004379 membrane Anatomy 0.000 description 1
- 239000011259 mixed solution Substances 0.000 description 1
- 239000012046 mixed solvent Substances 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 235000011929 mousse Nutrition 0.000 description 1
- 239000002362 mulch Substances 0.000 description 1
- 210000003205 muscle Anatomy 0.000 description 1
- VMGAPWLDMVPYIA-HIDZBRGKSA-N n'-amino-n-iminomethanimidamide Chemical compound N\N=C\N=N VMGAPWLDMVPYIA-HIDZBRGKSA-N 0.000 description 1
- 235000016709 nutrition Nutrition 0.000 description 1
- 230000035764 nutrition Effects 0.000 description 1
- MXEMKMNFLXVQBW-UHFFFAOYSA-N oleanoic acid Natural products C1CCC(C)(C)C2CCC3(C)C4(C)CCC5(C(O)=O)CCC(C)(C)CC5C4=CCC3C21C MXEMKMNFLXVQBW-UHFFFAOYSA-N 0.000 description 1
- ZQPPMHVWECSIRJ-KTKRTIGZSA-N oleic acid Chemical compound CCCCCCCC\C=C/CCCCCCCC(O)=O ZQPPMHVWECSIRJ-KTKRTIGZSA-N 0.000 description 1
- 150000007524 organic acids Chemical class 0.000 description 1
- 235000005985 organic acids Nutrition 0.000 description 1
- 229940124595 oriental medicine Drugs 0.000 description 1
- 230000001590 oxidative effect Effects 0.000 description 1
- 230000035515 penetration Effects 0.000 description 1
- 229940049954 penicillin Drugs 0.000 description 1
- 150000002972 pentoses Chemical class 0.000 description 1
- 230000001766 physiological effect Effects 0.000 description 1
- 229920001282 polysaccharide Polymers 0.000 description 1
- 239000005017 polysaccharide Substances 0.000 description 1
- 229920001296 polysiloxane Polymers 0.000 description 1
- LJCNRYVRMXRIQR-OLXYHTOASA-L potassium sodium L-tartrate Chemical compound [Na+].[K+].[O-]C(=O)[C@H](O)[C@@H](O)C([O-])=O LJCNRYVRMXRIQR-OLXYHTOASA-L 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000003755 preservative agent Substances 0.000 description 1
- 230000002265 prevention Effects 0.000 description 1
- 239000000047 product Substances 0.000 description 1
- 230000001737 promoting effect Effects 0.000 description 1
- 235000019260 propionic acid Nutrition 0.000 description 1
- 239000008213 purified water Substances 0.000 description 1
- FDRQPMVGJOQVTL-UHFFFAOYSA-N quercetin rutinoside Natural products OC1C(O)C(O)C(CO)OC1OCC1C(O)C(O)C(O)C(OC=2C(C3=C(O)C=C(O)C=C3OC=2C=2C=C(O)C(O)=CC=2)=O)O1 FDRQPMVGJOQVTL-UHFFFAOYSA-N 0.000 description 1
- IUVKMZGDUIUOCP-BTNSXGMBSA-N quinbolone Chemical compound O([C@H]1CC[C@H]2[C@H]3[C@@H]([C@]4(C=CC(=O)C=C4CC3)C)CC[C@@]21C)C1=CCCC1 IUVKMZGDUIUOCP-BTNSXGMBSA-N 0.000 description 1
- 230000009257 reactivity Effects 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- BOLDJAUMGUJJKM-LSDHHAIUSA-N renifolin D Natural products CC(=C)[C@@H]1Cc2c(O)c(O)ccc2[C@H]1CC(=O)c3ccc(O)cc3O BOLDJAUMGUJJKM-LSDHHAIUSA-N 0.000 description 1
- 230000004044 response Effects 0.000 description 1
- IKGXIBQEEMLURG-BKUODXTLSA-N rutin Chemical compound O[C@H]1[C@H](O)[C@@H](O)[C@H](C)O[C@@H]1OC[C@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](OC=2C(C3=C(O)C=C(O)C=C3OC=2C=2C=C(O)C(O)=CC=2)=O)O1 IKGXIBQEEMLURG-BKUODXTLSA-N 0.000 description 1
- ALABRVAAKCSLSC-UHFFFAOYSA-N rutin Natural products CC1OC(OCC2OC(O)C(O)C(O)C2O)C(O)C(O)C1OC3=C(Oc4cc(O)cc(O)c4C3=O)c5ccc(O)c(O)c5 ALABRVAAKCSLSC-UHFFFAOYSA-N 0.000 description 1
- 235000005493 rutin Nutrition 0.000 description 1
- 229960004555 rutoside Drugs 0.000 description 1
- 229940071089 sarcosinate Drugs 0.000 description 1
- FSYKKLYZXJSNPZ-UHFFFAOYSA-N sarcosine Chemical compound C[NH2+]CC([O-])=O FSYKKLYZXJSNPZ-UHFFFAOYSA-N 0.000 description 1
- 235000019512 sardine Nutrition 0.000 description 1
- 230000036186 satiety Effects 0.000 description 1
- 235000019627 satiety Nutrition 0.000 description 1
- 210000004761 scalp Anatomy 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 230000028327 secretion Effects 0.000 description 1
- 210000002966 serum Anatomy 0.000 description 1
- 239000002453 shampoo Substances 0.000 description 1
- 238000007873 sieving Methods 0.000 description 1
- KZJWDPNRJALLNS-VJSFXXLFSA-N sitosterol Chemical compound C1C=C2C[C@@H](O)CC[C@]2(C)[C@@H]2[C@@H]1[C@@H]1CC[C@H]([C@H](C)CC[C@@H](CC)C(C)C)[C@@]1(C)CC2 KZJWDPNRJALLNS-VJSFXXLFSA-N 0.000 description 1
- 229950005143 sitosterol Drugs 0.000 description 1
- 208000017520 skin disease Diseases 0.000 description 1
- 230000036556 skin irritation Effects 0.000 description 1
- 231100000475 skin irritation Toxicity 0.000 description 1
- 230000035483 skin reaction Effects 0.000 description 1
- 231100000430 skin reaction Toxicity 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- HRZFUMHJMZEROT-UHFFFAOYSA-L sodium disulfite Chemical compound [Na+].[Na+].[O-]S(=O)S([O-])(=O)=O HRZFUMHJMZEROT-UHFFFAOYSA-L 0.000 description 1
- 239000011684 sodium molybdate Substances 0.000 description 1
- 235000015393 sodium molybdate Nutrition 0.000 description 1
- TVXXNOYZHKPKGW-UHFFFAOYSA-N sodium molybdate (anhydrous) Chemical compound [Na+].[Na+].[O-][Mo]([O-])(=O)=O TVXXNOYZHKPKGW-UHFFFAOYSA-N 0.000 description 1
- 239000012064 sodium phosphate buffer Substances 0.000 description 1
- 235000011006 sodium potassium tartrate Nutrition 0.000 description 1
- 239000007787 solid Substances 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 239000007921 spray Substances 0.000 description 1
- 238000005507 spraying Methods 0.000 description 1
- 239000012086 standard solution Substances 0.000 description 1
- 230000001954 sterilising effect Effects 0.000 description 1
- 239000012089 stop solution Substances 0.000 description 1
- 229960005322 streptomycin Drugs 0.000 description 1
- 239000000758 substrate Substances 0.000 description 1
- 150000008163 sugars Chemical class 0.000 description 1
- 230000008961 swelling Effects 0.000 description 1
- 208000024891 symptom Diseases 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 229920002994 synthetic fiber Polymers 0.000 description 1
- 235000020357 syrup Nutrition 0.000 description 1
- 239000006188 syrup Substances 0.000 description 1
- 235000018553 tannin Nutrition 0.000 description 1
- 229920001864 tannin Polymers 0.000 description 1
- 239000001648 tannin Substances 0.000 description 1
- 239000012085 test solution Substances 0.000 description 1
- 238000004448 titration Methods 0.000 description 1
- 229960004441 tyrosine Drugs 0.000 description 1
- 230000036642 wellbeing Effects 0.000 description 1
- 230000029663 wound healing Effects 0.000 description 1
- 229940075420 xanthine Drugs 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/97—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from algae, fungi, lichens or plants; from derivatives thereof
- A61K8/9783—Angiosperms [Magnoliophyta]
- A61K8/9789—Magnoliopsida [dicotyledons]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K35/00—Medicinal preparations containing materials or reaction products thereof with undetermined constitution
- A61K35/66—Microorganisms or materials therefrom
- A61K35/74—Bacteria
- A61K35/741—Probiotics
- A61K35/744—Lactic acid bacteria, e.g. enterococci, pediococci, lactococci, streptococci or leuconostocs
- A61K35/747—Lactobacilli, e.g. L. acidophilus or L. brevis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K36/00—Medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicines
- A61K36/18—Magnoliophyta (angiosperms)
- A61K36/185—Magnoliopsida (dicotyledons)
- A61K36/73—Rosaceae (Rose family), e.g. strawberry, chokeberry, blackberry, pear or firethorn
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/96—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution
- A61K8/99—Cosmetics or similar toiletry preparations characterised by the composition containing materials, or derivatives thereof of undetermined constitution from microorganisms other than algae or fungi, e.g. protozoa or bacteria
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q19/00—Preparations for care of the skin
- A61Q19/08—Anti-ageing preparations
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2236/00—Isolation or extraction methods of medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicine
- A61K2236/10—Preparation or pretreatment of starting material
- A61K2236/19—Preparation or pretreatment of starting material involving fermentation using yeast, bacteria or both; enzymatic treatment
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2236/00—Isolation or extraction methods of medicinal preparations of undetermined constitution containing material from algae, lichens, fungi or plants, or derivatives thereof, e.g. traditional herbal medicine
- A61K2236/30—Extraction of the material
- A61K2236/33—Extraction of the material involving extraction with hydrophilic solvents, e.g. lower alcohols, esters or ketones
- A61K2236/333—Extraction of the material involving extraction with hydrophilic solvents, e.g. lower alcohols, esters or ketones using mixed solvents, e.g. 70% EtOH
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/52—Stabilizers
- A61K2800/522—Antioxidants; Radical scavengers
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/80—Process related aspects concerning the preparation of the cosmetic composition or the storage or application thereof
- A61K2800/85—Products or compounds obtained by fermentation, e.g. yoghurt, beer, wine
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Natural Medicines & Medicinal Plants (AREA)
- Epidemiology (AREA)
- Microbiology (AREA)
- Mycology (AREA)
- Engineering & Computer Science (AREA)
- Biotechnology (AREA)
- Pharmacology & Pharmacy (AREA)
- Medicinal Chemistry (AREA)
- Botany (AREA)
- Chemical & Material Sciences (AREA)
- Birds (AREA)
- Medical Informatics (AREA)
- Alternative & Traditional Medicine (AREA)
- Tropical Medicine & Parasitology (AREA)
- Gerontology & Geriatric Medicine (AREA)
- Dermatology (AREA)
- Molecular Biology (AREA)
- Cosmetics (AREA)
Abstract
본 발명에 따르면, 상기 혼합추출물을 유산균으로 발효시킨 유산균 발효액을 화장료 조성물로 포함하는 화장품을 사용하는 경우 한 종류의 화장품으로 항산화 효과, 피부 미백효과 및 피부주름 개선효과를 모두 나타낼 수 있음을 확인할 수 있다.
Description
| 단 위 | 사화근액 (실시예 1) |
사화근효액 (실시예 2) |
삼화근효액 (비교예 1) |
이화근효액 (비교예 2) |
|
| pH | - | 5.33 | 5.08 | 5.10 | 5.14 |
| 굴절률 | - | 1.322 | 1.323 | 1.322 | 1.321 |
| 폴리페놀화합물 함량 | ㎎/g | 184 | 302 | 201 | 195 |
| 플라보노이드 함량 | ㎎/g | 168 | 275 | 175 | 170 |
| 환원당 함량 | ㎎/g | 292 | 367 | 300 | 295 |
| 유기산 산도 | % | 0.00072 | 0.00076 | 0.00075 | 0.00074 |
| 단 위 | 사화근액 (실시예 1) |
사화근효액 (실시예 2) |
삼화근효액 (비교예 1) |
이화근효액 (비교예 2) |
|
| pH | - | 5.33 | 5.08 | 5.10 | 5.14 |
| 굴절률 | - | 1.322 | 1.323 | 1.322 | 1.321 |
| 폴리페놀화합물 함량 | ㎎/g | 184 | 302 | 201 | 195 |
| 플라보노이드 함량 | ㎎/g | 168 | 275 | 175 | 170 |
| 환원당 함량 | ㎎/g | 292 | 367 | 300 | 295 |
| 유기산 산도 | % | 0.00072 | 0.00076 | 0.00075 | 0.00074 |
| DPPH 라디칼 소거능(%) | ||
| Ascorbic Acid(100ppm) | 96.92 | |
| Brix 0.5 |
사화근액 | 94.62 |
| 사화근효액 | 95.08 | |
| 삼화근효액 | 94.79 | |
| 이화근효액 | 94.70 | |
| Brix 5 |
사화근액 | 95.17 |
| 사화근효액 | 99.80 | |
| 삼화근효액 | 99.17 | |
| 이화근효액 | 96.01 | |
| Ascorbic Acid(100ppm) | 사화근액 (Brix 5) |
사화근효액 (Brix 5) |
삼화근효액 (Brix 5) |
이화근효액 (Brix 5) |
|
| Xanthine Oxidase 저해 활성(%) |
46.81 |
53.06 |
87.89 |
61.07 |
59.84 |
| 알부틴 (500/) |
사화근액 (Brix 5) |
사화근효액 (Brix 5) |
삼화근효액 (Brix 5 |
이화근효액 (Brix 5) |
|
| 티로시나제 활성 저해율(%) |
50.09 |
63.13 |
79.73 |
64.29 |
63.98 |
| 1차 생존율(%) | 2차 생존율(%) | 평균(%) | ||
| 사화근액 (Brix 5) |
0% | 100 | 100 | 100 |
| 0.01% | 97.44 | 107.02 | 102.23 | |
| 0.03% | 101.74 | 101.65 | 101.70 | |
| 0.05% | 100.00 | 99.09 | 99.55 | |
| 0.10% | 99.28 | 102.99 | 101.14 | |
| 0.30% | 98.56 | 111.66 | 105.11 | |
| 0.50% | 92.00 | 117.96 | 104.98 | |
| 1.00% | 113.94 | 93.49 | 103.72 | |
| 3.00% | 99.74 | 102.99 | 101.37 | |
| 5.00% | 98.44 | 99.42 | 98.93 | |
| 10.00% | 97.38 | 96.79 | 97.09 | |
| 사화근효액 (Brix 5) |
0% | 100 | 100 | 100 |
| 0.01% | 98.19 | 97.93 | 98.06 | |
| 0.03% | 114.27 | 98.34 | 106.31 | |
| 0.05% | 105.51 | 92.13 | 98.82 | |
| 0.10% | 94.67 | 116.40 | 105.54 | |
| 0.30% | 99.01 | 103.07 | 101.04 | |
| 0.50% | 108.84 | 103.15 | 105.78 | |
| 1.00% | 97.74 | 108.03 | 102.89 | |
| 3.00% | 105.84 | 95.11 | 100.48 | |
| 5.00% | 100.99 | 98.48 | 99.74 | |
| 10.00% | 97.23 | 98.18 | 97.71 | |
| 삼화근효액 (Brix 5) |
0% | 100 | 100 | 100 |
| 0.01% | 97.59 | 96.73 | 97.16 | |
| 0.03% | 102.98 | 97.24 | 100.11 | |
| 0.05% | 101.98 | 91.09 | 96.54 | |
| 0.10% | 92.26 | 104.87 | 98.56 | |
| 0.30% | 98.79 | 102.09 | 100.44 | |
| 0.50% | 95.13 | 101.57 | 98.35 | |
| 1.00% | 96.54 | 95.87 | 96.19 | |
| 3.00% | 100.65 | 93.65 | 97.15 | |
| 5.00% | 98.54 | 97.13 | 97.84 | |
| 10.00% | 96.54 | 97.16 | 96.85 | |
| 이화근효액 (Brix 5) |
0% | 100 | 100 | 100 |
| 0.01% | 96.01 | 96.24 | 96.13 | |
| 0.03% | 101.54 | 96.15 | 98.85 | |
| 0.05% | 101.11 | 90.78 | 95.95 | |
| 0.10% | 91.69 | 103.56 | 97.63 | |
| 0.30% | 97.36 | 101.86 | 99.61 | |
| 0.50% | 94.65 | 100.92 | 97.79 | |
| 1.00% | 95.35 | 95.06 | 95.20 | |
| 3.00% | 99.88 | 92.43 | 96.16 | |
| 5.00% | 97.38 | 96.09 | 96.74 | |
| 10.00% | 95.54 | 96.52 | 96.03 | |
| 시료 | 1차 생성율(%) | 2차 생성율(%) | 평균(%) | |
| control | 100 | 100 | 100 | |
| Retinol(1㎎/㎖) | 168.75 | 166.96 | 167.86 | |
| 사화근액 (Brix 5) |
0.1% | 102.67 | 117.85 | 110.26 |
| 0.2% | 117.86 | 111.60 | 114.73 | |
| 0.5% | 124.11 | 126.79 | 125.45 | |
| 1.0% | 131.25 | 124.11 | 127.68 | |
| 사화근효액 (Brix 5) |
0.1% | 113.39 | 108.92 | 111.16 |
| 0.2% | 119.64 | 123.89 | 121.77 | |
| 0.5% | 149.11 | 138.39 | 143.75 | |
| 1.0%. | 152.68 | 156.25 | 154.47 | |
| 삼화근효액 (Brix 5) |
0.1% | 103.66 | 107.13 | 105.40 |
| 0.2% | 118.03 | 112.97 | 115.50 | |
| 0.5% | 126.36 | 127.79 | 127.08 | |
| 1.0% | 135.91 | 126.99 | 131.45 | |
| 이화근효액 (Brix 5) |
0.1% | 102.11 | 106.93 | 104.52 |
| 0.2% | 116.29 | 110.47 | 113.38 | |
| 0.5% | 124.73 | 122.87 | 123.80 | |
| 1.0% | 132.96 | 125.11 | 129.04 | |
| 시료 | 1차 생성율(%) | 2차 생성율(%) | 평균(%) | 억제율(%) | |
| control | 100 | 100 | 100 | ||
| Retinol(1㎎/㎖) | 21.57 | 22.76 | 22.17 | 77.83 | |
| 사화근액 (Brix 5) |
0.1% | 90.4 | 91.13 | 90.77 | 9.23 |
| 0.2% | 83.15 | 84.92 | 84.04 | 15.96 | |
| 0.5% | 65.56 | 71.93 | 68.75 | 31.25 | |
| 1.0% | 59.67 | 62.39 | 61.03 | 38.97 | |
| 사화근효액 (Brix 5) |
0.1% | 70.09 | 73.54 | 71.82 | 28.18 |
| 0.2% | 59.83 | 55.80 | 57.82 | 42.18 | |
| 0.5% | 41.29 | 37.25 | 39.27 | 60.73 | |
| 1.0% | 38.79 | 34.83 | 36.81 | 63.19 | |
| 삼화근효액 (Brix 5) |
0.1% | 85.65 | 86.76 | 86.21 | 13.80 |
| 0.2% | 80.79 | 81.29 | 81.04 | 19.00 | |
| 0.5% | 63.47 | 68.47 | 65.97 | 34.03 | |
| 1.0% | 57.39 | 60.17 | 58.78 | 41.22 | |
| 이화근효액 (Brix 5) |
0.1% | 86.97 | 87.26 | 87.12 | 12.89 |
| 0.2% | 82.31 | 82.73 | 82.52 | 17.48 | |
| 0.5% | 64.73 | 69.73 | 67.23 | 32.77 | |
| 1.0% | 59.01 | 61.68 | 60.35 | 39.66 | |
| 시험 물질 |
반응이 나타난 피검자 수 | 평균 반응도 | ||||||||||||||||||||||||||
| 1주 | 2주 | 3주 | ||||||||||||||||||||||||||
| 1차 | 2차 | 3차 | 4차 | 5차 | 6차 | 7차 | 8차 | 9차 | ||||||||||||||||||||
| ± | + | ++ | ± | + | ++ | ± | + | ++ | ± | + | ++ | ± | + | ++ | ± | + | ++ | ± | + | ++ | ± | + | ++ | ± | + | ++ | ||
| 실시예 3 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.00 |
| 비교예 3 | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | - | 0.00 |
| 피검인원 | 20 | 20 | 20 | 20 | 20 | 20 | 20 | 20 | 20 | |||||||||||||||||||
| 각 피검자별 L의 평균값 | |
| 실시예 3 | 3.73 |
| 비교예 3 | 1.25 |
| 평균 주름 감소율(%) | 평균 보습 증가율(%) | |
| 실시예 3 | 35 | 86 |
| 비교예 3 | 13 | 45 |
Claims (14)
- 석류꽃, 석류나무뿌리(석류근), 접시꽃(촉규화), 촉규근, 매실꽃, 매실나무뿌리(매근), 연꽃 및 연근의 혼합추출물을 유산균으로 발효시킨 유산균 발효액을 유효성분으로 포함하는 항산화 및 주름개선 효과를 갖는 화장료 조성물.
- 제 1항에 있어서, 상기 유산균 발효액은 항산화 효과를 갖는 것을 특징으로 하는 항산화용 화장료 조성물.
- 삭제
- 제 1항에 있어서, 상기 유산균 발효액은 주름개선 효과를 갖는 것을 특징으로 하는 주름개선용 화장료 조성물.
- 제 1항에 있어서, 상기 혼합추출물은 50 ~ 90% 에탄올로 추출하는 것을 특징으로 하는 화장료 조성물.
- 제 1항에 있어서, 상기 유산균은 락토바실러스(Lactobacillus sp.)인 것을 특징으로 하는 화장료 조성물.
- 제 1항에 있어서, 상기 유산균 발효액을 호기성 발효하는 것을 특징으로 하는 화장료 조성물.
- 제 1항에 있어서, 상기 유산균 발효액은 기초화장료 제형, 색조화장료 제형, 헤어화장료 제형 및 바디화장료 제형으로 이루어진 군에서 선택되는 어느 하나인 것을 특징으로 하는 화장료 조성물.
- 제 1항, 제 2항 또는 제 4항 중 어느 한 항에 있어서, 상기 유산균 발효액은 화장료 조성물 총 중량 대비 0.01 ~ 10중량% 함유하는 것을 특징으로 하는 화장료 조성물.
- 하기 단계를 통해 유산균 발효액을 제조하는 제조방법:
a) 석류꽃, 석류나무뿌리(석류근), 접시꽃(촉규화), 촉규근, 매실꽃, 매실나무뿌리(매근), 연꽃 및 연근을 실온에서 침적하여 얻는 단계;
b) 상기 a) 단계에서 얻은 식물을 각각 1~2:1~2:1~2:1~2:1~2:1~2:1~2:1~2의 중량비로 혼합하여 분쇄하는 단계;
c) 상기 b)단계의 혼합물을 50 ~ 90% 에탄올로 혼합하여 추출하는 단계;
d) 상기 c)단계에서 추출한 추출물을 농축하여 혼합추출물을 얻는 단계; 및
e) 상기 d)단계의 혼합추출물에 유산균을 처리하여 유산균 발효액을 얻는 단계를 포함하는 유산균 발효액 제조방법.
- 제 10항에 있어서, 상기 e)단계의 유산균은 락토바실러스(Lactobacillus sp.)인 것을 특징으로 하는 유산균 발효액 제조방법.
- 제 10항에 있어서, 상기 e)단계의 유산균은 엠알에스 배지(MRS medium)에 접종 후 진탕 배양기에서 배양 후 계대배양하는 것을 특징으로 하는 유산균 발효액 제조방법.
- 제 10항에 있어서, 상기 e)단계의 유산균은 초기 균수를 107 ~ 109CFU/ml로 조절하는 것을 특징으로 하는 유산균 발효액 제조방법.
- 제 10항에 있어서, 상기 e)단계의 유산균 발효액은 1차 내지 3차 여과 하는 것을 특징으로 하는 유산균 발효액 제조방법.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020130019841A KR101452770B1 (ko) | 2013-02-25 | 2013-02-25 | 유산균 발효액을 함유하는 항산화, 미백 및 주름개선 효과를 갖는 화장료 조성물 |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| KR1020130019841A KR101452770B1 (ko) | 2013-02-25 | 2013-02-25 | 유산균 발효액을 함유하는 항산화, 미백 및 주름개선 효과를 갖는 화장료 조성물 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20140105976A KR20140105976A (ko) | 2014-09-03 |
| KR101452770B1 true KR101452770B1 (ko) | 2014-10-22 |
Family
ID=51754544
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020130019841A Active KR101452770B1 (ko) | 2013-02-25 | 2013-02-25 | 유산균 발효액을 함유하는 항산화, 미백 및 주름개선 효과를 갖는 화장료 조성물 |
Country Status (1)
| Country | Link |
|---|---|
| KR (1) | KR101452770B1 (ko) |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20160075056A (ko) | 2014-12-19 | 2016-06-29 | 코웨이 주식회사 | 혼합 꽃 추출물의 유산균 발효물을 유효성분으로 함유하는 화장료 조성물 |
| KR101963597B1 (ko) | 2018-07-19 | 2019-04-01 | 주식회사 한국화장품제조 | 접시꽃 캘러스 추출물 또는 수정목 캘러스 추출물을 유효성분으로 포함하는 항산화, 미백 및 보습용 화장료 조성물 |
| KR20190125685A (ko) | 2018-04-30 | 2019-11-07 | 주식회사 단정바이오 | 수수 잎, 줄기 발효액 또는 추출액을 유효성분으로 함유하는 피부 미백 및 주름개선용 화장료 조성물, 및 그의 제조방법 |
| KR102374974B1 (ko) | 2021-09-07 | 2022-03-17 | 일동바이오사이언스(주) | 비피도박테리움 비피덤 idcc 4201을 포함하는 피부 미백용 화장료 조성물 |
| KR102451022B1 (ko) * | 2021-10-26 | 2022-10-06 | 주식회사 더가든오브내추럴솔루션 | 버블 추출 공법을 기반으로 한 광노화 방지용 화장료 조성물 |
| WO2024227065A1 (en) | 2023-04-28 | 2024-10-31 | Actera Ingredients, Inc. | Fermented binding serum |
Families Citing this family (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR101964723B1 (ko) * | 2017-08-30 | 2019-04-02 | 김은주 | 유산균발효 한방추출물을 함유하는 피부 항산화, 주름, 미백, 트러블 개선 화장료 조성물 및 그 제조방법 |
| KR102167786B1 (ko) * | 2020-07-28 | 2020-10-19 | 주식회사 토리든 | 락토바실러스를 이용한 병풀, 접시꽃, 푸른연꽃, 쓴풀 화장료용 발효물 및 그 제조방법과 화장료용 발효물을 함유한 상처치유, 피부 재생, 진정, 및 항염 효능을 갖는 화장료 조성물 |
| EP4288032A2 (en) * | 2021-02-08 | 2023-12-13 | Galderma Holding SA | Reduction of gene expression for the use for treating acne |
| CN113444766B (zh) * | 2021-06-04 | 2024-05-24 | 海天醋业集团有限公司 | 用于发酵酒陈酿过程中变质菌的富集培养基及的检测方法 |
Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006327988A (ja) * | 2005-05-26 | 2006-12-07 | Ichimaru Pharcos Co Ltd | 美白剤及び美白用皮膚外用剤 |
| KR20080094459A (ko) * | 2007-04-20 | 2008-10-23 | 주식회사 엘지생활건강 | 항산화 활성을 갖는 화장료 조성물 |
| KR100994987B1 (ko) * | 2008-03-31 | 2010-11-18 | 주식회사 씨앤피차앤박화장품 | 피부 진정 효과를 가지는 화장료 조성물 |
-
2013
- 2013-02-25 KR KR1020130019841A patent/KR101452770B1/ko active Active
Patent Citations (3)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006327988A (ja) * | 2005-05-26 | 2006-12-07 | Ichimaru Pharcos Co Ltd | 美白剤及び美白用皮膚外用剤 |
| KR20080094459A (ko) * | 2007-04-20 | 2008-10-23 | 주식회사 엘지생활건강 | 항산화 활성을 갖는 화장료 조성물 |
| KR100994987B1 (ko) * | 2008-03-31 | 2010-11-18 | 주식회사 씨앤피차앤박화장품 | 피부 진정 효과를 가지는 화장료 조성물 |
Cited By (6)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR20160075056A (ko) | 2014-12-19 | 2016-06-29 | 코웨이 주식회사 | 혼합 꽃 추출물의 유산균 발효물을 유효성분으로 함유하는 화장료 조성물 |
| KR20190125685A (ko) | 2018-04-30 | 2019-11-07 | 주식회사 단정바이오 | 수수 잎, 줄기 발효액 또는 추출액을 유효성분으로 함유하는 피부 미백 및 주름개선용 화장료 조성물, 및 그의 제조방법 |
| KR101963597B1 (ko) | 2018-07-19 | 2019-04-01 | 주식회사 한국화장품제조 | 접시꽃 캘러스 추출물 또는 수정목 캘러스 추출물을 유효성분으로 포함하는 항산화, 미백 및 보습용 화장료 조성물 |
| KR102374974B1 (ko) | 2021-09-07 | 2022-03-17 | 일동바이오사이언스(주) | 비피도박테리움 비피덤 idcc 4201을 포함하는 피부 미백용 화장료 조성물 |
| KR102451022B1 (ko) * | 2021-10-26 | 2022-10-06 | 주식회사 더가든오브내추럴솔루션 | 버블 추출 공법을 기반으로 한 광노화 방지용 화장료 조성물 |
| WO2024227065A1 (en) | 2023-04-28 | 2024-10-31 | Actera Ingredients, Inc. | Fermented binding serum |
Also Published As
| Publication number | Publication date |
|---|---|
| KR20140105976A (ko) | 2014-09-03 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR101452770B1 (ko) | 유산균 발효액을 함유하는 항산화, 미백 및 주름개선 효과를 갖는 화장료 조성물 | |
| KR101769422B1 (ko) | 다중기능성 세럼형 화장품의 혼합 조성물 및 제조방법 | |
| KR101045310B1 (ko) | 유산균 발효 커피추출물을 함유하는 화장료 조성물 제조방법 | |
| KR101086225B1 (ko) | 청미래덩굴 추출물을 유효성분으로 함유하는 화장료 조성물 | |
| KR101566320B1 (ko) | 봉선화, 영실, 마카, 비파나무 잎 및 얌빈 뿌리 복합발효추출물을 포함하는 피부 재생, 진정 및 자극완화용 화장료조성물 | |
| KR20110091137A (ko) | 백합나무 추출물을 유효성분으로 함유하는 화장료 조성물 | |
| KR101209124B1 (ko) | 식물 복합 추출물을 함유하는 기능성 화장료 조성물 | |
| KR102096703B1 (ko) | 무화과잎 주정 추출물을 포함하는 화장료 조성물 | |
| KR101825704B1 (ko) | 커피 실버스킨 추출물을 유효성분으로 함유하는 화장료 조성물 | |
| KR102594733B1 (ko) | 율피, 백지, 마치현, 행인, 및 상백피 발효 추출물을 포함하는 기미, 주근깨, 미백 및 주름 개선용 화장료 조성물 및 이의 제조 방법 | |
| KR102063686B1 (ko) | 콩뿌리 추출물을 함유하는 피부 외용제 조성물 | |
| CN113631227A (zh) | 抗老化剂、抗氧化剂、抗炎剂、及美白剂、以及化妆品 | |
| KR101822092B1 (ko) | 알로에베라 잎 복합 추출물을 함유하는 열노화 방지용 화장료 조성물 | |
| KR20110122448A (ko) | 지리터리풀 추출물을 유효성분으로 함유하는 화장료 조성물 | |
| KR101819060B1 (ko) | 황련해독탕 발효물을 유효성분으로 포함하는 화장료 조성물 | |
| KR102303400B1 (ko) | 제주 용암해수, 제주 보리누룩 및 천연식물을 이용한 발효물 제조방법 및 이를 포함하는 화장품 조성물 및 그 제조방법 | |
| KR101002012B1 (ko) | 산딸나무, 찔레나무, 비자나무, 파초 및 죽절초 혼합물의 추출물을 함유하는 화장료 조성물 | |
| KR20110085371A (ko) | 천연식물 추출물을 함유한 두피 상태 개선용 화장료 조성물 | |
| KR20190049067A (ko) | 커피 실버스킨 및 커피박 추출물을 유효성분으로 함유하는 화장료 조성물 | |
| KR101985381B1 (ko) | 포도, 오렌지, 사과, 레몬 및 라임을 발효하여 만든 과일 발효액 콤플렉스를 함유하는 피부 각질 박리용 화장료 조성물 | |
| KR101790185B1 (ko) | 발효콩 추출 분획물을 유효 성분으로 포함하는 화장료 조성물 | |
| KR20220116780A (ko) | 찔레꽃을 이용한 미백용 조성물의 제조 방법 | |
| KR101921518B1 (ko) | 스피루리나 발효 추출물을 유효성분으로 함유하는 항산화 및 주름개선용 화장료 조성물 | |
| KR102504878B1 (ko) | 능소화 꽃, 매화 꽃, 붉가시나무의 가지 또는 잎의 혼합발효추출물을 유효성분으로 하는 항염, 피부진정 및 가려움증 개선용 화장료 조성물 | |
| KR101073107B1 (ko) | 베리 칵테일 조성물을 함유하는 화장료의 제조방법 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| A201 | Request for examination | ||
| PA0109 | Patent application |
Patent event code: PA01091R01D Comment text: Patent Application Patent event date: 20130225 |
|
| PA0201 | Request for examination | ||
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
Comment text: Notification of reason for refusal Patent event date: 20140331 Patent event code: PE09021S01D |
|
| PG1501 | Laying open of application | ||
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
Patent event code: PE07011S01D Comment text: Decision to Grant Registration Patent event date: 20140923 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
Comment text: Registration of Establishment Patent event date: 20141014 Patent event code: PR07011E01D |
|
| PR1002 | Payment of registration fee |
Payment date: 20141015 End annual number: 3 Start annual number: 1 |
|
| PG1601 | Publication of registration | ||
| FPAY | Annual fee payment |
Payment date: 20180731 Year of fee payment: 5 |
|
| PR1001 | Payment of annual fee |
Payment date: 20180731 Start annual number: 5 End annual number: 5 |
|
| PR1001 | Payment of annual fee |
Payment date: 20211005 Start annual number: 8 End annual number: 8 |
|
| PR1001 | Payment of annual fee |
Payment date: 20231115 Start annual number: 10 End annual number: 10 |
|
| PR1001 | Payment of annual fee |
Payment date: 20241002 Start annual number: 11 End annual number: 11 |