KR100609800B1 - Mek 저해제를 사용한 관절염 치료 방법 - Google Patents
Mek 저해제를 사용한 관절염 치료 방법 Download PDFInfo
- Publication number
- KR100609800B1 KR100609800B1 KR1020017007502A KR20017007502A KR100609800B1 KR 100609800 B1 KR100609800 B1 KR 100609800B1 KR 1020017007502 A KR1020017007502 A KR 1020017007502A KR 20017007502 A KR20017007502 A KR 20017007502A KR 100609800 B1 KR100609800 B1 KR 100609800B1
- Authority
- KR
- South Korea
- Prior art keywords
- methyl
- phenylamino
- iodo
- benzamide
- difluoro
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 0 CC(*)/C(/C(C)(*)/C=C1\C(*(*)O*)=O)=C\C(C)(*)/C=C1/N(*)C(C=C1)=CC(C)(C)C=C1*#IC=CI Chemical compound CC(*)/C(/C(C)(*)/C=C1\C(*(*)O*)=O)=C\C(C)(*)/C=C1/N(*)C(C=C1)=CC(C)(C)C=C1*#IC=CI 0.000 description 1
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/535—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with at least one nitrogen and one oxygen as the ring hetero atoms, e.g. 1,2-oxazines
- A61K31/5375—1,4-Oxazines, e.g. morpholine
- A61K31/5377—1,4-Oxazines, e.g. morpholine not condensed and containing further heterocyclic rings, e.g. timolol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/135—Amines having aromatic rings, e.g. ketamine, nortriptyline
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/13—Amines
- A61K31/135—Amines having aromatic rings, e.g. ketamine, nortriptyline
- A61K31/136—Amines having aromatic rings, e.g. ketamine, nortriptyline having the amino group directly attached to the aromatic ring, e.g. benzeneamine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/165—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide
- A61K31/166—Amides, e.g. hydroxamic acids having aromatic rings, e.g. colchicine, atenolol, progabide having the carbon of a carboxamide group directly attached to the aromatic ring, e.g. procainamide, procarbazine, metoclopramide, labetalol
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/16—Amides, e.g. hydroxamic acids
- A61K31/18—Sulfonamides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/185—Acids; Anhydrides, halides or salts thereof, e.g. sulfur acids, imidic, hydrazonic or hydroximic acids
- A61K31/19—Carboxylic acids, e.g. valproic acid
- A61K31/192—Carboxylic acids, e.g. valproic acid having aromatic groups, e.g. sulindac, 2-aryl-propionic acids, ethacrynic acid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/34—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having five-membered rings with one oxygen as the only ring hetero atom, e.g. isosorbide
- A61K31/341—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having five-membered rings with one oxygen as the only ring hetero atom, e.g. isosorbide not condensed with another ring, e.g. ranitidine, furosemide, bufetolol, muscarine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/335—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin
- A61K31/35—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom
- A61K31/351—Heterocyclic compounds having oxygen as the only ring hetero atom, e.g. fungichromin having six-membered rings with one oxygen as the only ring hetero atom not condensed with another ring
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/38—Heterocyclic compounds having sulfur as a ring hetero atom
- A61K31/381—Heterocyclic compounds having sulfur as a ring hetero atom having five-membered rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/40—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with one nitrogen as the only ring hetero atom, e.g. sulpiride, succinimide, tolmetin, buflomedil
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/41—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having five-membered rings with two or more ring hetero atoms, at least one of which being nitrogen, e.g. tetrazole
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4406—Non condensed pyridines; Hydrogenated derivatives thereof only substituted in position 3, e.g. zimeldine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/4409—Non condensed pyridines; Hydrogenated derivatives thereof only substituted in position 4, e.g. isoniazid, iproniazid
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/435—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with one nitrogen as the only ring hetero atom
- A61K31/44—Non condensed pyridines; Hydrogenated derivatives thereof
- A61K31/445—Non condensed piperidines, e.g. piperocaine
- A61K31/4453—Non condensed piperidines, e.g. piperocaine only substituted in position 1, e.g. propipocaine, diperodon
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P19/00—Drugs for skeletal disorders
- A61P19/02—Drugs for skeletal disorders for joint disorders, e.g. arthritis, arthrosis
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P43/00—Drugs for specific purposes, not provided for in groups A61P1/00-A61P41/00
Landscapes
- Health & Medical Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Epidemiology (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Rheumatology (AREA)
- Physical Education & Sports Medicine (AREA)
- Orthopedic Medicine & Surgery (AREA)
- Pain & Pain Management (AREA)
- Immunology (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
- Acyclic And Carbocyclic Compounds In Medicinal Compositions (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Pyridine Compounds (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
- Hydrogenated Pyridines (AREA)
Abstract
Description
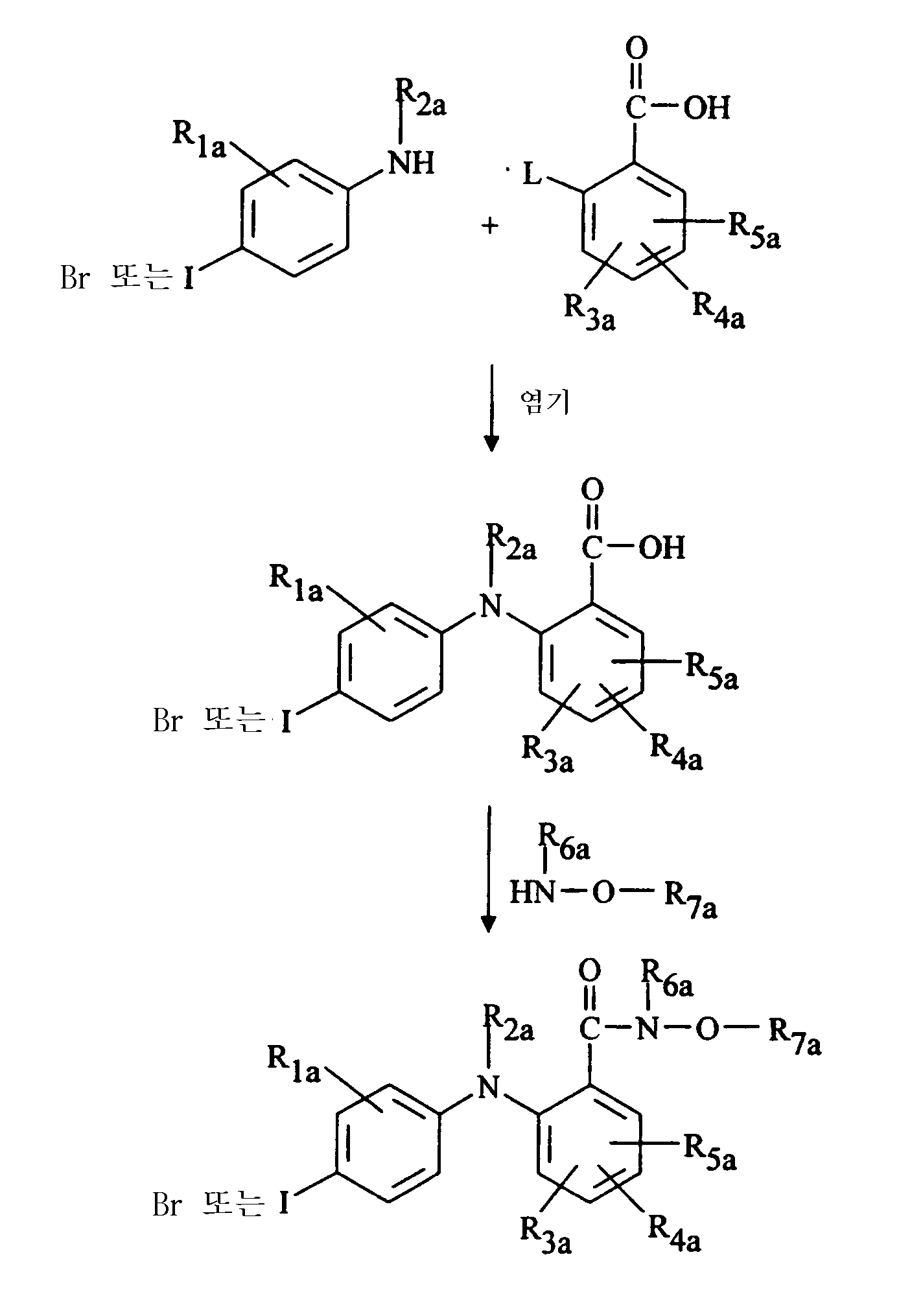
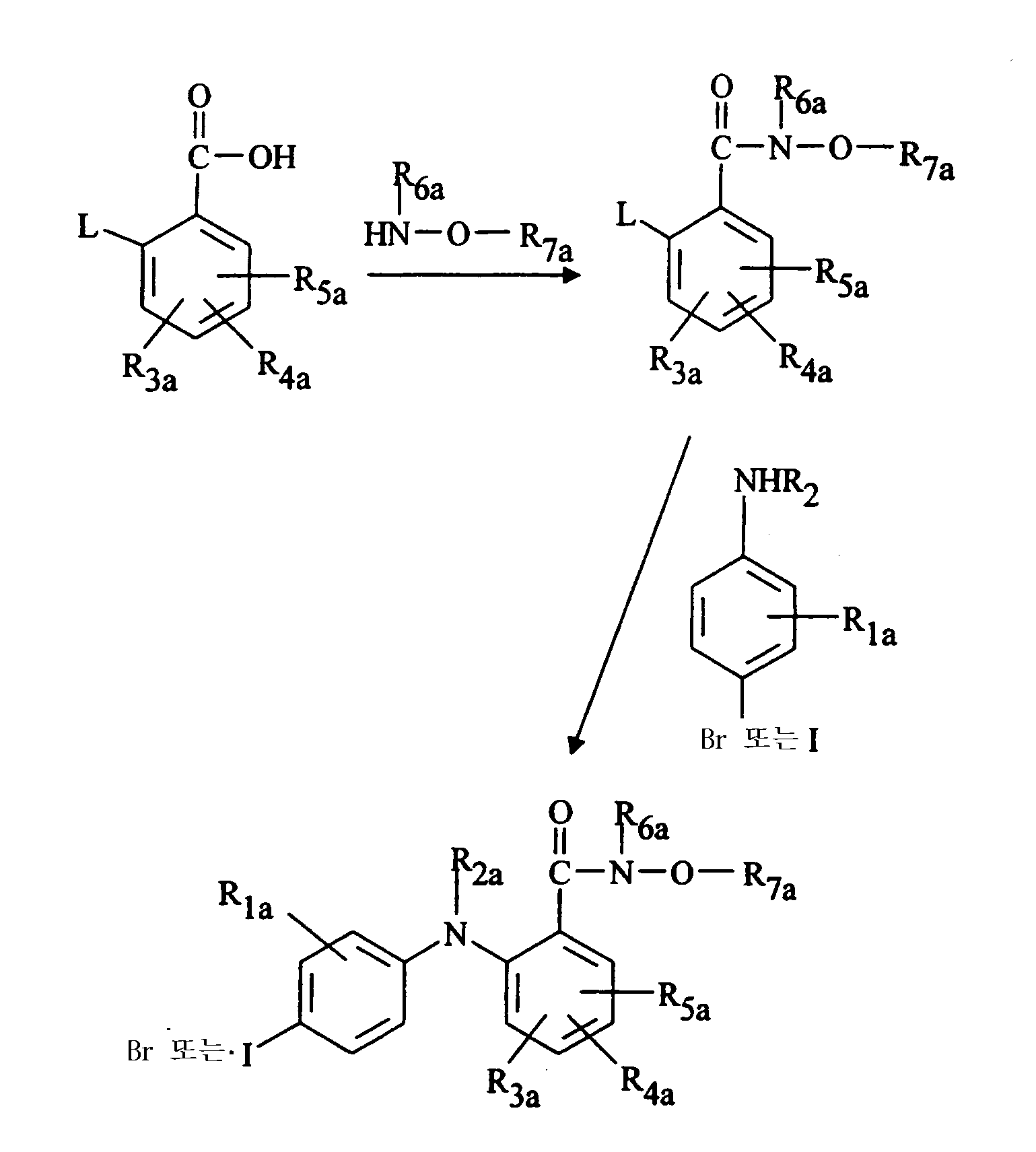
화학식 (Ⅰ)의 본 발명의 몇몇 화합물을 조합 합성 기술을 사용하여 제조했다. 일반적인 방법은 하기와 같다:
시험 화합물 IC50 (nM)
| 모델 | 184352 | 198306 | 203311 |
| 쥐 카라게난 족저 부종 (CFE)(ID40) | 75.8 | 14.7 | 18.9 mg/kg |
| 쥐 SCW 관절염 (SCW)(IC50) | 10.0 | 11.2 | >100 " |
| 쥐 애주반트 관절염 (AIP)(IC50) | 6.9 | 6.6 | >30 " |
| 쥐 IL-1 관절염 (IL-1)(%억제 프로테오글리칸 손실 @ 30mg/kg) | 57.9 | 42.9 | 29.2 |
Claims (19)
- 삭제
- 삭제
- 삭제
- 삭제
- 삭제
- 항관절염 유효량의 하기 화학식 Ⅰ의 화합물 및 그의 제약학적으로 허용되는 염으로부터 선택되는 MEK 저해제를 포함하는, 관절염을 앓고 있어 그 치료를 필요로 하거나 관절염이 진행될 위험이 있는 환자에서의 관절염 치료 또는 예방용 제약 조성물.<화학식 Ⅰ>상기 화학식에서,R1은 수소, 히드록시, C1-C8 알킬, C1-C8 알콕시, 할로, 트리플루오로메틸, 또는 CN이고;R2는 수소이고;R3, R4 및 R5는 독립적으로 수소, 히드록시, 할로, 트리플루오로메틸, C1-C8 알킬, C1-C8 알콕시, 니트로, CN 또는 -(O 또는 NH)m-(CH2)n-R9이고, 여기에서 R9는 수소, 히드록시, COOH 또는 NR10R11이고;n 은 0-4이고;m 은 0 또는 1이고;R10 및 R11은 독립적으로 수소 또는 C1-C8 알킬이거나, 그들이 부착되는 질소와 함께, O, S, NH, 또는 N-C1-C8 알킬로부터 선택되는 1, 2, 또는 3개의 추가의 헤테로 원자를 함유할 수 있는 3-10원 환상 고리를 형성할 수 있고;Z는 COOR7, 테트라졸릴, CONR6R7, CONHNR10R11, 또는 CH2OR7이고;R6 및 R7은 독립적으로 수소, C1-C8 알킬, C2-C8 알케닐, C2-C8 알키닐, (CO)-C1-C8 알킬, C5-C12 아릴, C4-C11 헤테로아릴, O, S, NH, 또는 N 알킬로부터 선택되는 1, 2 또는 3개의 헤테로 원자를 함유할 수 있는 C3-C10 시클로알킬이거나; R6 및 R7은 그들이 부착되는 질소와 함께 O, S, NH, 또는 N 알킬로부터 선택되는 1, 2 또는 3개의 추가의 헤테로 원자를 함유할 수 있는 3-10원 환상 고리를 형성하며; 여기에서, 상기 알킬, 알케닐, C5-C12 아릴, 헤테로시클릭, 및 알키닐 기는 비치환되거나 할로, 히드록시, C1-C6 알콕시, 아미노, 니트로, C1-C4 알킬아미노, 디(C1-C4)알킬아미노, C3-C6 시클로알킬, 페닐, 페녹시, C3-C5 헤테로아릴, 또는 C3-C5 헤테로아릴옥시로 치환될 수 있다.
- 제6항에 있어서, 상기 MEK 저해제가,[4-클로로-2-(1H-테트라졸-5-일)-페닐-(4-요오도-2-메틸-페닐)-아민;(4-요오도-2-메틸-페닐)-[2-(1H-테트라졸-5-일)-페닐]아민;[4-니트로-2-(1H-테트라졸-5-일)-페닐-(4-요오도-2-메틸-페닐)-아민;4-플루오로-2-(4-요오도-2-메틸페닐아미노)벤조산;3,4,5-트리플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤조산;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤조산;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤조산;5-클로로-2-(4-요오도-2-메틸-페닐아미노)-벤조산;나트륨 5-클로로-2-(4-요오도-2-메틸-페닐아미노)-벤조에이트;5-브로모-2-(4-요오도-2-메틸-페닐아미노)-벤조산;2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤조산;4-클로로-2-(4-요오도-2-메틸-페닐아미노)-벤조산;2-(4-요오도-2-메틸-페닐아미노)-벤조산;5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤조산;5-요오도-2-(4-요오도-2-메틸-페닐아미노)-벤조산;2,3,5-트리플루오로-4-(4-요오도-2-메틸-페닐아미노)-벤조산;2-(4-요오도-페닐아미노)-5-메톡시-벤조산;5-메틸-2-(4-요오도-2-메틸-페닐아미노)-벤조산;2-(4-요오도-2-메틸-페닐아미노)-4-니트로-벤조산;2-(4-브로모-2-메틸-페닐아미노)-4-플루오로-벤조산;2-(2-브로모-4-요오도-페닐아미노)-5-니트로-벤조산;2-(4-브로모-2-메틸-페닐아미노)-3,4-디플루오로-벤조산;5-클로로-N-(2-히드록시에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-메틸-벤즈아미드;N-에틸-4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N,N-디메틸-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(1H-테트라졸-5-일)-벤즈아미드;5-브로모-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-2-(4-요오도-2-메틸-페닐아미노)-N,N-디메틸-벤즈아미드;[5-클로로-2-(4-요오도-2-메틸-페닐아미노)-벤조일아미노]-아세트산;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-프로필-벤즈아미드;5-브로모-N-(2-히드록시-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N,N-디에틸-4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;4-플루오로-N-{3-[4-(2-히드록시-에틸)-피페라진-1-일]-프로필}-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N,N-디에틸-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드;N-부틸-4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-N,N-디에틸-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-2-(4-요오도-2-메틸-페닐아미노)-N,N-디메틸-벤즈아미드;5-브로모-3,4-디플루오로-N-(2-히드록시-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-(2,3-디히드록시-프로필)-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피페리딘-1-일-에틸)-벤즈아미드;3,4-디플루오로-N-(2-히드록시-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-(2,3-디히드록시-프로필)-4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;3,4-디플루오로-N-(3-히드록시-프로필)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피롤리딘-1-일-에틸)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피리딘-4-일-에틸)-벤즈아미드;4-플루오로-N-(2-히드록시-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-N-(3-디메틸아미노-프로필)-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-2-페닐아미노)-N-(2-모르폴린-4-일-에틸)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-모르폴린-4-일-에틸)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피롤리딘-1-일-에틸)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피리딘-4-일-에틸)-벤즈아미드;N-(3-디메틸아미노-프로필)-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-벤질-4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;2-(4-브로모-2-메틸-페닐아미노)-3,4-디플루오로-N-(2-히드록시-에틸)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-모르폴린-4-일-에틸)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-피페리딘-1-일-프로필)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-피페리딘-1-일-프로필)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-티오펜-2-일-에틸)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피롤리딘-1-일-에틸)-벤즈아미드;2-(4-브로모-2-메틸-페닐아미노)-3,4-디플루오로-N-(2-모르폴린-4-일-에틸)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-피리딘-4-일메틸-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-피리딘-4-일메틸-벤즈아미드;2-(4-브로모-2-일메틸-페닐아미노)-N-(3-디메틸아미노-프로필)-3,4-디플루오로-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-피리딘-4-일메틸-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피리딘-4-일-에틸)-벤즈아미드;2-(4-브로모-2-메틸-페닐아미노)-3,4-디플루오로-N-(2-피리딘-4-일-에틸)-벤즈아미드;2-(4-브로모-2-메틸-페닐아미노)-3,4-디플루오로-N-(3-히드록시-프로필)-벤즈아미드;2-(4-브로모-2-메틸-페닐아미노)-3,4-디플루오로-N-(2-피롤리딘-1-일-에틸)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-페네틸-벤즈아미드;2-(4-브로모-2-메틸-페닐아미노)-3,4-디플루오로-N-(2-티오펜-2-일-에틸)-벤즈아미드;2-(4-브로모-2-메틸-페닐아미노)-3,4-디플루오로-N-피리딘-4-일메틸-벤즈아미드;2-(4-브로모-2-메틸-페닐아미노)-3,4-디플루오로-N-페네틸-벤즈아미드;2-(4-브로모-2-메틸-페닐아미노)-3,4-디플루오로-N-(2-피페리딘-1-일-에틸)-벤즈아미드;5-클로로-N-{3-[4-(2-히드록시-에틸)-피페라진-1-일]-프로필}-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-플루오로-N-{3-[4-(2-히드록시-에틸)-피페라진-1-일]-프로필}-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;2-(4-요오도-2-메틸-페닐아미노)-5-니트로-N-피리딘-4-일 메틸-벤즈아미드;5-브로모-N-{3-[4-(2-히드록시-에틸)-피페라진-1-일]-프로필}-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-N-(2-디에틸아미노-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피페리딘-1-일-에틸)-벤즈아미드;(3-히드록시-피롤리딘-1-일)-[2-(4-요오도-2-메틸-페닐아미노)-5-니트로-페닐]-메타논;5-클로로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피롤리딘-1-일-에틸)-벤즈아미드;5-브로모-N-(2-디에틸아미노-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-{2-[비스-(2-히드록시-에틸)-아미노]-에틸}-5-클로로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-{2-[비스-(2-히드록시-에틸)-아미노]-에틸}-5-브로모-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-{3-[4-(2-히드록시-에틸)-피페라진-1-일]-프로필}-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-피리딘-4-일메틸-벤즈아미드;5-브로모-2-(4-요오도-2-에틸-페닐아미노)-N-(2-피롤리딘-1-일-에틸)-벤즈아미드;5-브로모-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피페리딘-1-일-에틸)-벤즈아미드;5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피롤리딘-1-일-에틸)-벤즈아미드;5-클로로-N-(3-디메틸아미노-프로필)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-{2-[비스-(2-히드록시-에틸)-아미노]-에틸}-5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-N-(3-히드록시-프로필)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-N-(3-디에틸아미노-2-히드록시-프로필)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피페리딘-1-일-에틸)-벤즈아미드;5-브로모-N-(3-히드록시-프로필)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-2-(4-요오도-2-메틸-페닐아미노)-N-(3-피페리딘-1-일-프로필)-벤즈아미드;N-{2-[비스-(2-히드록시-에틸)-아미노]-에틸}-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드;5-클로로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-모르폴린-4-일-에틸)-벤즈아미드;5-클로로-N-(3-디에틸아미노-프로필)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-N-(2-디이소프로필아미노-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-피페리딘-1-일-프로필)-벤즈아미드;2-(4-요오도-2-메틸-페닐아미노)-5-니트로-N-(2-피페리딘-1-일-에틸)-벤즈아미드;5-브로모-2-(4-요오도-2-메틸-페닐아미노)-N-(2-피페라진-1-일-에틸)-벤즈아미드;N-(2-디에틸아미노-에틸)-5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-N-(3-디메틸아미노-프로필)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-(3-히드록시-프로필)-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드;5-플루오로-N-(3-히드록시-프로필)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-(3-디에틸아미노-프로필)-5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-(3-디에틸아미노-프로필)-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드;5-브로모-2-(4-요오도-2-메틸-페닐아미노)-N-(2-모르폴린-4-일-에틸)-벤즈아미드;2-(4-요오도-2-메틸-페닐아미노)-5-니트로-N-(3-피페리딘-1-일-프로필)-벤즈아미드;[5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-페닐]-(3-히드록시-피롤리딘-1-일)-메타논;5-브로모-N-(2-디이소프로필아미노-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-모르폴린-4-일-에틸)-벤즈아미드;5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-피페리딘-1-일-프로필)-벤즈아미드;[5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-페닐]-[4-(2-히드록시-에틸)-피페라진-1-일]-메타논;N-(3-디에틸아미노-2-히드록시-프로필)-5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-시클로프로필-5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-N-(2-히드록시-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-플루오로-N-(2-히드록시-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-벤질옥시-5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-벤질옥시-5-브로모-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-N-(2-히드록시-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-(2-히드록시-에틸)-5-요오도-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-(2-히드록시-에틸)-2-(4-요오도-2-에틸-페닐아미노)-5-니트로-벤즈아미드;2-(4-요오도-2-메틸-페닐아미노)-N-메틸-5-니트로-N-페닐-벤즈아미드;5-클로로-N-시클로프로필-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-메틸-N-페닐-벤즈아미드;N-알릴-5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-벤질옥시-5-요오도-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-알릴-5-클로로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-시클로프로필-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드;5-브로모-N-시클로프로필-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-2-(4-요오도-2-메틸-페닐아미노)-N-메틸-N-페닐-벤즈아미드;N-알릴-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드;N-알릴-5-브로모-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-메틸-벤질)-벤즈아미드;N-시클로프로필-5-요오도-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-2-(4-요오도-2-메틸-페닐아미노)-N-메틸-N-페닐-벤즈아미드;N-벤질옥시-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드;N-시클로헥실-5-요오도-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-알릴-5-요오도-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-요오도-2-(4-요오도-2-메틸-페닐아미노)-N-(3-메틸-벤질)-벤즈아미드;2-(4-요오도-2-메틸-페닐아미노)-N-(3-메틸-벤질)-5-니트로-벤즈아미드;5-요오도-2-(4-요오도-2-메틸-페닐아미노)-N-메틸-N-페닐-벤즈아미드;N-시클로헥실-5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-N-시클로헥실-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-2-(4-요오도-2-메틸-페닐아미노)-N-(3-메틸-벤질)-벤즈아미드;5-브로모-N-시클로헥실-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-메틸-벤질)-벤즈아미드;N-시클로헥실-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드;N-벤질옥시-5-브로모-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-벤질옥시-5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-N-(2-히드록시-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-N-(2-히드록시-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;2-(4-요오도-2-메틸-페닐아미노)-N-메틸-5-니트로-N-페닐-벤즈아미드;5-클로로-2-(4-요오도-2-메틸-페닐아미노)-N-메틸-N-페닐-벤즈아미드;N-(2-히드록시-에틸)-5-요오도-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-N-시클로프로필-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-알릴-5-클로로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-메틸-N-페닐-벤즈아미드;N-(2-히드록시-에틸)-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드;5-플루오로-N-(2-히드록시-에틸)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-N-시클로프로필-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-시클로프로필-5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-시클로프로필-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드;N-알릴-5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-벤질옥시-5-요오도-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;N-알릴-5-브로모-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-2-(4-요오도-2-메틸-페닐아미노)-N-메틸-N-페닐-벤즈아미드;N-알릴-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤질 알콜;[5-클로로-2-(4-요오도-2-메틸-페닐아미노)-페닐]-메탄올;[2-(4-요오도-2-메틸-페닐아미노)-5-니트로-페닐]-메탄올;[5-브로모-2-(4-요오도-2-메틸-페닐아미노)-페닐]-메탄올; 및N-알릴-2-(4-요오도-2-메틸-페닐아미노)-5-니트로-벤즈아미드로부터 선택되는 화합물인 조성물.
- 제6항에 있어서, 상기 MEK 저해제가,(a) R1은 수소, 메틸, 메톡시, 플루오로, 클로로, 또는 브로모이고; (b) R2는 수소이며; (c) R3, R4 및 R5는 독립적으로 수소, 플루오로, 클로로, 브로모, 요오도, 메틸, 메톡시, 또는 니트로이고; (d) R10 및 R11은 독립적으로 수소 또는 메틸이고; (e) Z는 COOR7, 테트라졸릴, CONR6R7, CONHNR10R11, 또는 CH2OR7이고; R6 및 R7은 독립적으로 수소, C1-C4 알킬, C4-C11 헤테로아릴, 또는 O, S, 또는 NH로부터 선택되는 1 또는 2개의 헤테로 원자를 함유할 수 있는 C3-C5 시클로알킬이거나; R6 및 R7은 그들이 부착되는 질소와 함께 O, NH, 또는 N-알킬로부터 선택되는 1 또는 2개의 추가의 헤테로 원자를 함유할 수 있는 5-6 원 환상 고리를 형성하며; 여기에서 상기 알킬 또는 아릴기는 비치환되거나 또는 할로, 히드록시, 메톡시, 에톡시, 또는 헤테로아릴옥시에 의해 치환될 수 있는 화학식 Ⅰ의 화합물인 조성물.
- 제8항에 있어서, 상기 MEK 저해제가, Z는 COOR7이고; R7은 H, 펜타플루오로페닐, 또는 테트라졸릴이고; R3 및 R5는 독립적으로 H, 플루오로, 또는 클로로이고; R4 는 플루오로인 화학식 Ⅰ의 화합물인 조성물.
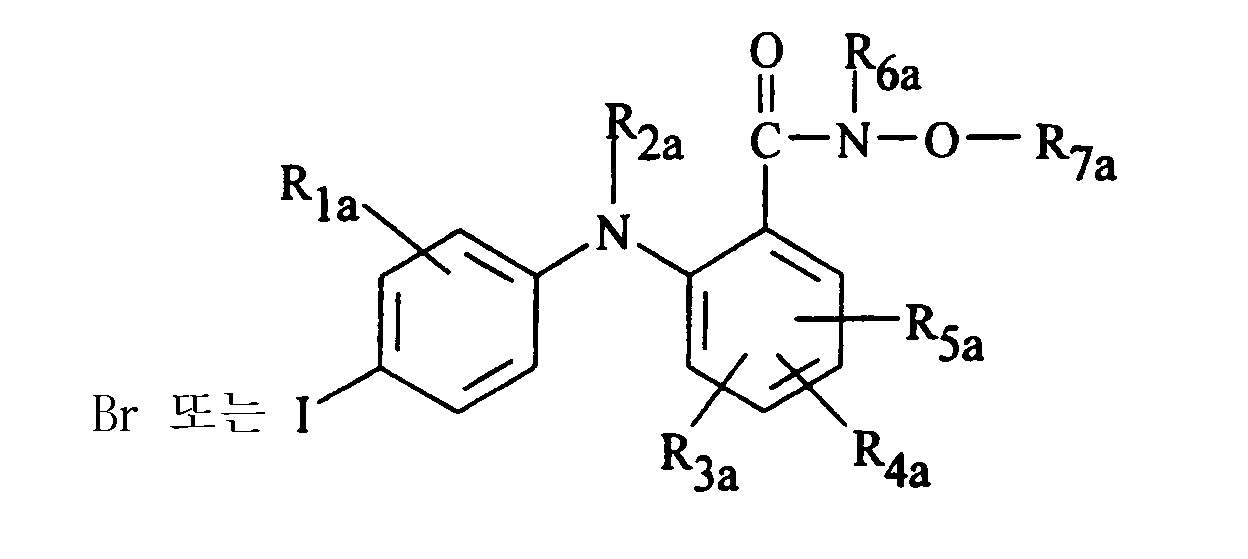
- 항관절염 유효량의 하기 화학식 Ⅱ의 화합물 및 그의 제약학적으로 허용되는 염으로부터 선택되는 MEK 저해제를 포함하는, 관절염을 앓고 있어 그 치료를 필요로 하거나 관절염이 진행될 위험이 있는 환자에서의 관절염 치료 또는 예방용 제약 조성물.<화학식 Ⅱ>상기 화학식에서,R1a는 수소, 히드록시, C1-C8 알킬, C1-C8 알콕시, 할로, 트리플루오로메틸, 또는 CN 이고;R2a는 수소이고;R3a, R4a, 및 R5a는 독립적으로 수소, 히드록시, 할로, 트리플루오로메틸, C1-C8 알킬, C1-C8 알콕시, 니트로, CN, 또는 (O 또는 NH)m-(CH2)n-R9a이며, 여기에서 R9a는 수소, 히드록시, CO2H 또는 NR10aR11a이고;n은 0-4이고;m은 0 또는 1이고;R10a 및 R11a는 독립적으로 수소 또는 C1-C8 알킬이거나, 그들이 부착되는 질소와 함께 O, S, NH, 또는 N-C1-C8 알킬로부터 선택된 1, 2 또는 3개의 추가의 헤테로 원자를 함유할 수 있는 3- 내지 10-원 환상 고리를 형성할 수 있고;R6a는 수소, C1-C8 알킬, (CO)-C1-C8 알킬, C5-C12 아릴, C6-C20 아르알킬, 또는 C3-C10 시클로알킬이고;R7a는 수소, C1-C8 알킬, C2-C8 알케닐, C2-C8 알키닐, C3-C10 (시클로알킬, 또는 O, S, 또는 NR9a로부터 선택된 헤테로 원자를 함유할 수 있는 시클로알킬)이며;여기에서, 상기 알킬, 알케닐, C5-C12 아릴, C4-C11 헤테로아릴, 헤테로시클릭 및 알키닐 기는 비치환되거나 할로, 히드록시, C1-C6 알콕시, 아미노, 니트로, C1-C4 알킬아미노, 디(C1-C4)알킬아미노, C3-C6 시클로알킬, 페닐, 페녹시, C3-C5 헤테로아릴 또는 헤테로시클릭 라디칼, 또는 C3-C5 헤테로아릴옥시 또는 헤테로시클릭 라디칼-옥시로 치환될 수 있거나; 또는 R6a 및 R7a는 그들이 부착되는 N과 함께, O, S, 또는 NR10aR11a로부터 선택된 1, 2 또는 3개의 추가의 헤테로 원자를 함유할 수 있는 5- 내지 10-원 환상 고리를 형성할 수 있다.
- 제10항에 있어서, 상기 MEK 저해제가,(a) R1a는 H, 메틸, 플루오로, 또는 클로로이고; (b) R2a는 H이고; R3a, R4a, 및 R5a는 각각 H, Cl, 니트로, 또는 F이고; (c) R6a는 H이고; (d) R7a는 메틸, 에틸, 2-프로페닐, 프로필, 부틸, 펜틸, 헥실, 시클로프로필메틸, 시클로부틸메틸, 시클로프로필메틸, 또는 시클로프로필에틸이고; (e) 4' 위치는 Br이 아닌 I인 화학식 Ⅱ의 구조를 갖는 화합물인 조성물.
- 제11항에 있어서, 상기 MEK 저해제가, R4a는 CO-N-R6a-OR7a 기에 대하여는 파라 위치이고 가교 질소에 대하여는 메타 위치인 4위치의 F이고; R3a 및 R5a 중 하나 또는 둘다는 F 또는 Cl이고; R1a는 메틸 또는 클로로인 화학식 Ⅱ의 구조를 갖는 화합물인 조성물.
- 제10항에 있어서, 상기 MEK 저해제가,4-플루오로-N-히드록시-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(메톡시)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(프로프-2-이닐옥시)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-페녹시에톡시)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-티에닐메톡시)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(프로프-2-에닐옥시)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(시클로프로필메톡시)-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(시클로펜톡시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-푸릴메톡시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-에톡시-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(부트-2-에닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(시클로프로필메톡시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(1-메틸프로프-2-이닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-페닐프로프-2-이닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-메틸-5-페닐펜트-2-엔-4-이닐옥시)-벤즈아미드3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(프로프-2-이닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(프로폭시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(시클로부틸옥시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-티에닐메톡시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-메틸-프로프-2-에닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(2-페녹시에톡시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(부트-2-에닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(부트-3-이닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(시클로펜틸옥시)-벤즈아미드;3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-(2-플루오로페닐)-프로프-2-이닐옥시)-벤즈아미드;5-브로모-3,4-디플루오로-N-히드록시-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(n-프로폭시)-벤즈아미드;5-브로모-3,4-디플루오로-N-(푸란-3-일메톡시)-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-N-(부트-2-에닐옥시)-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-N-부톡시-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-메틸-부트-2-에닐옥시)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-메틸-펜트-2-엔-4-이닐옥시)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-벤질)-N-[5-(3-메톡시-페닐)-3-메틸-펜트-2-엔-4-이닐옥시]-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(프로프-2-이닐옥시)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-[3-(3-메톡시-페닐)-프로프-2-이닐옥시]-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(티오펜-2-일메톡시)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(피리딘-3-일메톡시)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(3-(2-플루오로페닐)-프로프-2-이닐옥시)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(에톡시)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(시클로프로필메톡시)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(이소프로폭시)-벤즈아미드;5-브로모-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(부트-3-이닐옥시)-벤즈아미드;5-클로로-N-히드록시-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-2-(4-요오도-2-메틸-페닐아미노)-N-(테트라히드로-피란-2-일옥시)-벤즈아미드;5-클로로-2-(4-요오도-2-메틸-페닐아미노)-N-메톡시-벤즈아미드;4-브로모-2-(4-요오도-2-메틸-페닐아미노)-N-페닐메톡시-벤즈아미드;4-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-페닐메톡시-벤즈아미드;5-플루오로-N-히드록시-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-요오도-2-(4-요오도-2-메틸-페닐아미노)-N-페닐메톡시-벤즈아미드;5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(테트라히드로피란-2-일옥시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(3-페닐프로프-2-이닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(3-푸릴메톡시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(2-티에닐메톡시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(부트-3-이닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(2-메틸-프로프-2-에닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(부트-2-에닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(메톡시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(에톡시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(시클로부톡시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(이소프로폭시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(2-페녹시에톡시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(시클로프로필메톡시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(n-프로폭시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(1-메틸-프로프-2-이닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(3-(3-플루오로페닐)-프로프-2-이닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(4,4-디메틸펜트-2-이닐옥시)-벤즈아미드;3,4-디플루오로-2-(4-브로모-2-메틸-페닐아미노)-N-(시클로펜톡시)-벤즈아미드;3,4,5-트리플루오로-N-히드록시-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-3,4-디플루오로-N-히드록시-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-3,4-디플루오로-2-(2-플루오로-4-요오도-페닐아미노)-N-히드록시-벤즈아미드;N-히드록시-2-(4-요오도-2-메틸-페닐아미노)-4-니트로-벤즈아미드;3,4,5-트리플루오로-2-(2-플루오로-4-요오도-페닐아미노)-N-히드록시-벤즈아미드;5-클로로-3,4-디플루오로-2-(2-플루오로-4-요오도-페닐아미노)-N-히드록시-벤즈아미드;5-브로모-2-(2-클로로-4-요오도-페닐아미노)-3,4-디플루오로-N-히드록시-벤즈아미드;2-(2-플루오로-4-요오도-페닐아미노)-N-히드록시-4-니트로-벤즈아미드;2-(2-클로로-4-요오도-페닐아미노)-3,4,5-트리플루오로-N-히드록시-벤즈아미드;5-클로로-2-(2-클로로-4-요오도-페닐아미노)-3,4-디플루오로-N-히드록시-벤즈아미드;5-브로모-2-(2-브로모-4-요오도-페닐아미노)-3,4-디플루오로-N-히드록시-벤즈아미드;2-(2-클로로-4-요오도-페닐아미노)-N-히드록시-4-메틸-벤즈아미드;2-(2-브로모-4-요오도-페닐아미노)-3,4,5-트리플루오로-N-히드록시-벤즈아미드;2-(2-브로모-4-요오도-페닐아미노)-5-클로로-3,4-디플루오로-N-히드록시-벤즈아미드;2-(2-브로모-4-요오도-페닐아미노)-N-히드록시-4-니트로-벤즈아미드;4-플루오로-2-(2-플루오로-4-요오도-페닐아미노)-N-히드록시-벤즈아미드;3,4-디플루오로-2-(2-플루오로-4-요오도-페닐아미노)-N-히드록시-벤즈아미드;2-(2-클로로-4-요오도-페닐아미노)-4-플루오로-N-히드록시-벤즈아미드;2-(2-클로로-4-요오도-페닐아미노)-3,4-디플루오로-N-히드록시-벤즈아미드;2-(2-브로모-4-요오도-페닐아미노)-4-플루오로-N-히드록시-벤즈아미드;2-(2-브로모-4-요오도-페닐아미노)-3,4-디플루오로-N-히드록시-벤즈아미드;N-시클로프로필메톡시-3,4,5-트리플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-클로로-N-시클로프로필메톡시-3,4-디플루오로-2-(4-요오도-2-메틸-페닐아미노)-벤즈아미드;5-브로모-N-시클로프로필메톡시-3,4-디플루오로-2-(2-플루오로-4-요오도-페닐아미노)-벤즈아미드;N-시클로프로필메톡시-2-(4-요오도-2-메틸-페닐아미노)-4-니트로-벤즈아미드;N-시클로프로필메톡시-3,4,5-트리플루오로-2-(2-플루오로-4-요오도-페닐아미노)-벤즈아미드;5-클로로-N-시클로프로필메톡시-3,4-디플루오로-2-(2-플루오로-4-요오도-페닐아미노)-벤즈아미드;5-브로모-2-(2-클로로-4-요오도-페닐아미노)-N-시클로프로필메톡시-3,4-디플루오로-벤즈아미드;N-시클로프로필메톡시-2-(2-플루오로-4-요오도-페닐아미노)-4-니트로-벤즈아미드;2-(2-클로로-4-요오도-페닐아미노)-N-시클로프로필메톡시-3,4,5-트리플루오로-벤즈아미드;5-클로로-2-(2-클로로-4-요오도-페닐아미노)-N-시클로프로필메톡시-3,4-디플루오로-벤즈아미드;5-브로모-2-(2-브로모-4-요오도-페닐아미노)-N-에톡시-3,4-디플루오로-벤즈아미드;2-(2-클로로-4-요오도-페닐아미노)-N-에톡시-4-니트로-벤즈아미드;2-(2-브로모-4-요오도-페닐아미노)-N-시클로프로필메톡시-3,4,5-트리플루오로-벤즈아미드;2-(2-브로모-4-요오도-페닐아미노)-5-클로로-N-시클로프로필메톡시-3,4-디플루오로-벤즈아미드;2-(2-브로모-4-요오도-페닐아미노)-N-시클로프로필메톡시-4-니트로-벤즈아미드;N-시클로프로필메톡시-4-플루오로-2-(2-플루오로-4-요오도-페닐아미노)-벤즈아미드;N-시클로프로필메톡시-3,4-디플루오로-2-(2-플루오로-4-요오도-페닐아미노)-벤즈아미드;2-(2-클로로-4-요오도-페닐아미노)-N-시클로프로필메톡시-4-플루오로-벤즈아미드;2-(2-클로로-4-요오도-페닐아미노)-N-시클로프로필메톡시-3,4-디플루오로-벤즈아미드;2-(2-브로모-4-요오도-페닐아미노)-N-시클로프로필메톡시-4-플루오로-벤즈아미드; 및2-(2-브로모-4-요오도-페닐아미노)-N-시클로프로필메톡시-3,4-디플루오로-벤즈아미드로부터 선택되는 화학식을 갖는 화합물인 조성물.
- 2-(2-클로로-4-요오도페닐아미노)-5-클로로-N-시클로프로필메톡시-3,4-디플루오로벤즈아미드; 2-(4-요오도페닐아미노)-N-시클로프로필메톡시-5-클로로-3,4-디플루오로벤즈아미드; 2-(4-요오도페닐아미노)-5-클로로-3,4-디플루오로벤조산; 2-(2-클로로-4-요오도페닐아미노)-5-클로로-3,4-디플루오로벤조산; 5-클로로-3,4-디플루오로-2-(4-요오도-2-메틸페닐아미노)-벤조산; 및 5-클로로-N-시클로프로필메톡시-3,4-디플루오로-2-(4-요오도-2-메틸페닐아미노)-벤즈아미드로부터 선택되는 화합물의 항관절염 유효량을 포함하는, 관절염을 앓고 있어 그 치료를 필요로 하거나 관절염이 진행될 위험이 있는 환자에서의 관절염의 치료 또는 예방용 제약 조성물.
- 2-(2-클로로-4-요오도페닐아미노)-N-시클로프로필메톡시-3,4-디플루오로벤즈아미드;2-(2-메틸-4-요오도페닐아미노)-N-히드록시-4-플루오로벤즈아미드;2-(2-메틸-4-요오도페닐아미노)-N-히드록시-3,4-디플루오로-5-브로모벤즈아미드;2-(2-메틸-4-요오도페닐아미노)-N-시클로프로필메톡시-3,4-디플루오로-5-브로모벤즈아미드;2-(2-메틸-4-요오도페닐아미노)-N-시클로부틸메톡시-3,4-디플루오로-5-브로모벤즈아미드;2-(2-클로로-4-요오도페닐아미노)-N-시클로프로필메톡시-3,4-디플루오로-5-브로모벤즈아미드;2-(2-클로로-4-요오도페닐아미노)-N-히드록시-3,4-디플루오로-5-브로모벤즈아미드;2-(2-클로로-4-요오도페닐아미노)-N-시클로부틸메톡시-3,4-디플루오로벤즈아미드;2-(2-클로로-4-요오도페닐아미노)-N-히드록시-4-플루오로벤즈아미드;2-(2-메틸-4-요오도페닐아미노)-N-히드록시-3,4-디플루오로벤즈아미드;2-(2-메틸-4-요오도페닐아미노)-N-시클로프로필메톡시-3,4,5-트리플루오로벤즈아미드; 및2-(2-클로로-4-요오도페닐아미노)-N-시클로프로필메톡시-4-플루오로벤즈아미드로부터 선택되는 화합물의 항관절염 유효량을 포함하는, 관절염을 앓고 있어 그 치료를 필요로 하거나 관절염이 진행될 위험이 있는 환자에서의 관절염의 치료 또는 예방용 제약 조성물.
- 제15항에 있어서, 상기 화합물이 2-(2-클로로-4-요오도페닐아미노)-N-시클로프로필메톡시-3,4-디플루오로벤즈아미드, 2-(2-메틸-4-요오도페닐아미노)-N-시클로프로필메톡시-3,4,5-트리플루오로벤즈아미드; 및 2-(2-클로로-4-요오도페닐아미노)-N-시클로프로필메톡시-4-플루오로벤즈아미드로부터 선택되는 것인 조성물.
- 제8항에 있어서, 상기 MEK 저해제가, Z는 COOR7이고; R7은 H, 펜타플루오로페닐, 또는 테트라졸릴이고; R3, R4 및 R5는 독립적으로 H, 플루오로, 또는 클로로인 화학식 Ⅰ의 화합물인 조성물.
- 제8항에 있어서, 상기 MEK 저해제가, Z는 COOR7이고; R7은 H, 펜타플루오로페닐, 또는 테트라졸릴이고; R3, R4 및 R5 중 두개는 독립적으로 플루오로인 화학식 I의 화합물인 조성물.
- 2-(4-요오도-2-메틸-페닐아미노)-5-니트로-N-(4-술파모일-벤질)-벤즈아미드;5-플루오로-2-(4-요오도-2-메틸-페닐아미노)-N-(4-술파모일-벤질)-벤즈아미드;5-요오도-2-(4-요오도-2-메틸-페닐아미노)-N-(4-술파모일-벤질)-벤즈아미드; 및5-브로모-2-(4-요오도-2-메틸-페닐아미노)-N-(4-술파모일-벤질)-벤즈아미드로부터 선택되는 화합물의 항관절염 유효량을 포함하는, 관절염을 앓고 있어 그 치료를 필요로 하거나 관절염이 진행될 위험이 있는 환자에서의 관절염 치료 또는 예방용 제약 조성물.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US11254498P | 1998-12-16 | 1998-12-16 | |
| US60/112,544 | 1998-12-16 | ||
| US16465199P | 1999-11-10 | 1999-11-10 | |
| US60/164,651 | 1999-11-10 |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| KR20010093840A KR20010093840A (ko) | 2001-10-29 |
| KR100609800B1 true KR100609800B1 (ko) | 2006-08-09 |
Family
ID=26810075
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| KR1020017007502A Expired - Fee Related KR100609800B1 (ko) | 1998-12-16 | 1999-12-15 | Mek 저해제를 사용한 관절염 치료 방법 |
Country Status (8)
| Country | Link |
|---|---|
| EP (1) | EP1143957A3 (ko) |
| JP (1) | JP2002532415A (ko) |
| KR (1) | KR100609800B1 (ko) |
| AU (1) | AU776788C (ko) |
| CA (1) | CA2346448A1 (ko) |
| HU (1) | HUP0104693A3 (ko) |
| IL (1) | IL143236A0 (ko) |
| WO (1) | WO2000035436A2 (ko) |
Families Citing this family (94)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| IL147617A0 (en) * | 1999-07-16 | 2002-08-14 | Warner Lambert Co | Method for treating chronic pain using mek inhibitors |
| US7030119B1 (en) | 1999-07-16 | 2006-04-18 | Warner-Lambert Company | Method for treating chronic pain using MEK inhibitors |
| NZ515567A (en) * | 1999-07-16 | 2004-03-26 | Warner Lambert Co | Method for treating chronic pain using MEK inhibitors |
| HU230251B1 (hu) | 2000-07-19 | 2015-11-30 | Warner-Lambert Co. | 4-Jód-fenil-amino-benzhidroxámsav-észter-származékok és ezeket tartalmazó gyógyászati készítmények |
| IL149462A0 (en) | 2001-05-09 | 2002-11-10 | Warner Lambert Co | Method of treating or inhibiting neutrophil chemotaxis by administering a mek inhibitor |
| KR20020096367A (ko) * | 2001-06-19 | 2002-12-31 | 주식회사 티지 바이오텍 | 관절염 예방 또는 치료제 및 그것의 스크리닝 방법 |
| KR20020096368A (ko) * | 2001-06-19 | 2002-12-31 | 주식회사 티지 바이오텍 | 연골세포의 분화촉진, 연골세포의 탈분화 억제 또는탈분화된 연골세포의 재분화 촉진제, 그것의 스크리닝방법 및 그것을 이용한 연골세포의 제조방법 |
| JP4896717B2 (ja) | 2003-07-24 | 2012-03-14 | ワーナー−ランバート カンパニー リミテッド ライアビリティー カンパニー | N−メチル−置換ベンゾアミダゾール |
| EP1674452A4 (en) | 2003-09-19 | 2007-10-10 | Chugai Pharmaceutical Co Ltd | NEW 4-PHENYLAMINOBENZALDOXIMDERIVATIE AND ITS USE AS MEK INHIBITOR |
| NZ546011A (en) | 2003-10-21 | 2009-09-25 | Warner Lambert Co | Polymorphic form of N-[(R)-2,3-dihydroxy-propoxy]-3,4-difluoro-2-(2-fluoro-4-iodophenylamino)-benzamide |
| UA89035C2 (ru) * | 2003-12-03 | 2009-12-25 | Лео Фарма А/С | Эфиры гидроксамовых кислот и их фармацевтическое применение |
| SI1761528T1 (sl) | 2004-06-11 | 2008-06-30 | Japan Tobacco Inc | 5-amino-2,4,7-triokso-3,4,7,8-tetrahidro-2H-pirido(2,3-D)pirimidinski derivati in sorodne spojine za zdravljenje raka |
| US7378423B2 (en) | 2004-06-11 | 2008-05-27 | Japan Tobacco Inc. | Pyrimidine compound and medical use thereof |
| MY144232A (en) | 2004-07-26 | 2011-08-15 | Chugai Pharmaceutical Co Ltd | 5-substituted-2-phenylamino benzamides as mek inhibitors |
| SI1934174T1 (sl) * | 2005-10-07 | 2011-08-31 | Exelixis Inc | Inhibitorji MEK in postopki za njihovo uporabo |
| TR201900306T4 (tr) | 2006-12-14 | 2019-02-21 | Exelixis Inc | Mek inhibitörlerini kullanma yöntemleri. |
| BRPI0821227A2 (pt) | 2007-12-19 | 2015-06-16 | Cancer Rec Tech Ltd | Composto, composição farmacêutica, método para preparar a mesma, uso de um composto, método para tratar uma doença ou distúrbio, para inibir função de raf e para inibir proliferação celular, inibir progressão do ciclo celular, promover apoptose, ou uma combinação de um ou mais dos mesmos |
| JP5746630B2 (ja) | 2008-11-10 | 2015-07-08 | バイエル・インテレクチュアル・プロパティ・ゲゼルシャフト・ミット・ベシュレンクテル・ハフツングBayer Intellectual Property GmbH | 置換スルホンアミドフェノキシベンズアミド |
| EP2380023A2 (en) * | 2008-12-19 | 2011-10-26 | Medirista Biotechnologies AB | Oxidized cardiolipin as a novel pro-inflammatory factor |
| US20120269803A1 (en) | 2009-10-21 | 2012-10-25 | Bayer Intellectual Property Gmbh | Substituted benzosulphonamides |
| US8962606B2 (en) | 2009-10-21 | 2015-02-24 | Bayer Intellectual Property Gmbh | Substituted benzosulphonamides |
| JP2013508320A (ja) | 2009-10-21 | 2013-03-07 | バイエル・ファルマ・アクチェンゲゼルシャフト | 置換されたハロフェノキシベンズアミド誘導体 |
| CN104945401B (zh) | 2010-02-01 | 2017-09-05 | 癌症研究技术有限公司 | Ip化合物及它们在治疗中的应用 |
| CA2816188A1 (en) | 2010-10-29 | 2012-05-03 | Marion Hitchcock | Substituted phenoxypyridines |
| PT3409278T (pt) | 2011-07-21 | 2020-12-18 | Sumitomo Dainippon Pharma Oncology Inc | Inibidores de proteína cinase heterocíclicos |
| CA2860676A1 (en) | 2012-01-09 | 2013-07-18 | Novartis Ag | Organic compositions to treat beta-catenin-related diseases |
| BR112015008113B1 (pt) | 2012-10-12 | 2022-05-24 | Exelixis, Inc. | Novo processo para preparar compostos para uso no tratamento de câncer |
| US9394257B2 (en) | 2012-10-16 | 2016-07-19 | Tolero Pharmaceuticals, Inc. | PKM2 modulators and methods for their use |
| US9498532B2 (en) | 2013-03-13 | 2016-11-22 | Novartis Ag | Antibody drug conjugates |
| ES2738493T3 (es) | 2013-03-14 | 2020-01-23 | Tolero Pharmaceuticals Inc | Inhibidores de JAK2 y ALK2 y métodos para su uso |
| JP6333943B2 (ja) | 2013-03-15 | 2018-05-30 | ノバルティス アーゲー | 抗体薬物コンジュゲート |
| GB201320729D0 (en) | 2013-11-25 | 2014-01-08 | Cancer Rec Tech Ltd | Therapeutic compounds and their use |
| GB201320732D0 (en) | 2013-11-25 | 2014-01-08 | Cancer Rec Tech Ltd | Methods of chemical synthesis |
| US10786578B2 (en) | 2014-08-05 | 2020-09-29 | Novartis Ag | CKIT antibody drug conjugates |
| BR112017001588A2 (pt) | 2014-08-12 | 2017-11-21 | Novartis Ag | conjugados de anticorpo/fármaco anti-cdh6 |
| CA2960824A1 (en) | 2014-09-13 | 2016-03-17 | Novartis Ag | Combination therapies of alk inhibitors |
| ES2774448T3 (es) | 2014-10-03 | 2020-07-21 | Novartis Ag | Terapias de combinación |
| AU2015347015B2 (en) | 2014-11-14 | 2019-02-14 | Novartis Ag | Antibody drug conjugates |
| WO2016100882A1 (en) | 2014-12-19 | 2016-06-23 | Novartis Ag | Combination therapies |
| PL3237418T3 (pl) | 2014-12-23 | 2019-07-31 | Novartis Ag | Związki triazolopirymidyny i ich zastosowania |
| KR20170129757A (ko) | 2015-03-25 | 2017-11-27 | 노파르티스 아게 | Fgfr4 억제제로서의 포르밀화 n-헤테로시클릭 유도체 |
| US20190194315A1 (en) | 2015-06-17 | 2019-06-27 | Novartis Ag | Antibody drug conjugates |
| MA44334A (fr) | 2015-10-29 | 2018-09-05 | Novartis Ag | Conjugués d'anticorps comprenant un agoniste du récepteur de type toll |
| EP3472161B1 (en) | 2016-06-20 | 2020-03-25 | Novartis AG | Triazolopyridine compounds and uses thereof |
| MX388824B (es) | 2016-06-20 | 2025-03-20 | Novartis Ag | Formas cristalinas de compuesto de triazolopirimidina. |
| US10676479B2 (en) | 2016-06-20 | 2020-06-09 | Novartis Ag | Imidazolepyridine compounds and uses thereof |
| GB201702144D0 (en) | 2017-02-09 | 2017-03-29 | Annexin Pharmaceuticals Ab | Therapeutic compositions |
| US11179413B2 (en) | 2017-03-06 | 2021-11-23 | Novartis Ag | Methods of treatment of cancer with reduced UBB expression |
| WO2018185618A1 (en) | 2017-04-03 | 2018-10-11 | Novartis Ag | Anti-cdh6 antibody drug conjugates and anti-gitr antibody combinations and methods of treatment |
| AR111651A1 (es) | 2017-04-28 | 2019-08-07 | Novartis Ag | Conjugados de anticuerpos que comprenden agonistas del receptor de tipo toll y terapias de combinación |
| WO2018215937A1 (en) | 2017-05-24 | 2018-11-29 | Novartis Ag | Interleukin-7 antibody cytokine engrafted proteins and methods of use in the treatment of cancer |
| MX2019014023A (es) | 2017-05-24 | 2020-02-17 | Novartis Ag | Proteinas de anticuerpo injertadas con citocina y metodos de uso en el tratamiento del cancer. |
| CN111107868A (zh) | 2017-05-24 | 2020-05-05 | 诺华股份有限公司 | 抗体细胞因子移植蛋白及使用方法 |
| CA3081602A1 (en) | 2017-11-16 | 2019-05-23 | Novartis Ag | Combination therapies |
| JP2021524835A (ja) | 2018-04-05 | 2021-09-16 | スミトモ ダイニッポン ファーマ オンコロジー, インコーポレイテッド | Axlキナーゼ阻害剤およびその使用 |
| WO2020012334A1 (en) | 2018-07-10 | 2020-01-16 | Novartis Ag | 3-(5-hydroxy-1-oxoisoindolin-2-yl)piperidine-2,6-dione derivatives and their use in the treatment of ikaros family zinc finger 2 (ikzf2)-dependent diseases |
| AR116109A1 (es) | 2018-07-10 | 2021-03-31 | Novartis Ag | Derivados de 3-(5-amino-1-oxoisoindolin-2-il)piperidina-2,6-diona y usos de los mismos |
| HRP20250912T1 (hr) | 2018-07-25 | 2025-09-26 | Advanced Accelerator Applications S.A. | Stabilne, koncentrirane otopine radionuklidnih kompleksa |
| WO2020021465A1 (en) | 2018-07-25 | 2020-01-30 | Advanced Accelerator Applications (Italy) S.R.L. | Method of treatment of neuroendocrine tumors |
| US11040038B2 (en) | 2018-07-26 | 2021-06-22 | Sumitomo Dainippon Pharma Oncology, Inc. | Methods for treating diseases associated with abnormal ACVR1 expression and ACVR1 inhibitors for use in the same |
| CN118666832A (zh) | 2018-08-17 | 2024-09-20 | 诺华股份有限公司 | 作为smarca2/brm atp酶抑制剂的脲化合物和组合物 |
| JP7358484B2 (ja) | 2018-09-25 | 2023-10-10 | アドヴァンスド・アクセラレーター・アプリケーションズ・(イタリー)・エッセエッレエッレ | 併用療法 |
| EP3873532A1 (en) | 2018-10-31 | 2021-09-08 | Novartis AG | Dc-sign antibody drug conjugates |
| EP3897637A1 (en) | 2018-12-20 | 2021-10-27 | Novartis AG | Dosing regimen and pharmaceutical combination comprising 3-(1-oxoisoindolin-2-yl)piperidine-2,6-dione derivatives |
| NZ778055A (en) | 2019-02-12 | 2025-11-28 | Sumitomo Pharma America Inc | Formulations comprising heterocyclic protein kinase inhibitors |
| ES2982474T3 (es) | 2019-02-15 | 2024-10-16 | Novartis Ag | Derivados de 3-(1-oxoisoindolin-2-il)piperidin-1,6-diona sustituidos y usos de estos |
| CN113490528B (zh) | 2019-02-15 | 2024-12-03 | 诺华股份有限公司 | 3-(1-氧代-5-(哌啶-4-基)异吲哚啉-2-基)哌啶-2,6-二酮衍生物及其用途 |
| EP3941463A1 (en) | 2019-03-22 | 2022-01-26 | Sumitomo Dainippon Pharma Oncology, Inc. | Compositions comprising pkm2 modulators and methods of treatment using the same |
| JP2022539208A (ja) | 2019-07-03 | 2022-09-07 | スミトモ ファーマ オンコロジー, インコーポレイテッド | チロシンキナーゼ非受容体1(tnk1)阻害剤およびその使用 |
| KR102889562B1 (ko) | 2019-09-26 | 2025-11-24 | 노파르티스 아게 | 아자-퀴놀린 화합물 및 그의 용도 |
| IL293889A (en) | 2019-12-20 | 2022-08-01 | Novartis Ag | Uses of anti-tgf-beta antibodies and checkpoint inhibitors for the treatment of proliferative diseases |
| AU2021288224A1 (en) | 2020-06-11 | 2023-01-05 | Novartis Ag | ZBTB32 inhibitors and uses thereof |
| WO2021260528A1 (en) | 2020-06-23 | 2021-12-30 | Novartis Ag | Dosing regimen comprising 3-(1-oxoisoindolin-2-yl)piperidine-2,6-dione derivatives |
| WO2022029573A1 (en) | 2020-08-03 | 2022-02-10 | Novartis Ag | Heteroaryl substituted 3-(1-oxoisoindolin-2-yl)piperidine-2,6-dione derivatives and uses thereof |
| AR123185A1 (es) | 2020-08-10 | 2022-11-09 | Novartis Ag | Compuestos y composiciones para inhibir ezh2 |
| EP4204021A1 (en) | 2020-08-31 | 2023-07-05 | Advanced Accelerator Applications International S.A. | Method of treating psma-expressing cancers |
| EP4204020A1 (en) | 2020-08-31 | 2023-07-05 | Advanced Accelerator Applications International S.A. | Method of treating psma-expressing cancers |
| WO2022043556A1 (en) | 2020-08-31 | 2022-03-03 | Novartis Ag | Stable radiopharmaceutical composition |
| TW202237119A (zh) | 2020-12-10 | 2022-10-01 | 美商住友製藥腫瘤公司 | Alk﹘5抑制劑和彼之用途 |
| US20240376094A2 (en) | 2021-02-02 | 2024-11-14 | Les Laboratoires Servier | Selective bcl-xl protac compounds and methods of use |
| WO2022195551A1 (en) | 2021-03-18 | 2022-09-22 | Novartis Ag | Biomarkers for cancer and methods of use thereof |
| TW202304979A (zh) | 2021-04-07 | 2023-02-01 | 瑞士商諾華公司 | 抗TGFβ抗體及其他治療劑用於治療增殖性疾病之用途 |
| WO2022221227A1 (en) | 2021-04-13 | 2022-10-20 | Nuvalent, Inc. | Amino-substituted heterocycles for treating cancers with egfr mutations |
| AR125874A1 (es) | 2021-05-18 | 2023-08-23 | Novartis Ag | Terapias de combinación |
| WO2023008978A1 (ko) * | 2021-07-30 | 2023-02-02 | 아주대학교산학협력단 | Sncg를 표적으로 하는 퇴행성 질환 치료 또는 진단용 조성물 |
| KR102734644B1 (ko) * | 2021-07-30 | 2024-11-27 | 아주대학교산학협력단 | Az-628을 유효성분으로 포함하는 sncg 과발현 질환의 예방 또는 치료용 약학적 조성물 |
| KR20230018813A (ko) * | 2021-07-30 | 2023-02-07 | 아주대학교산학협력단 | Pd-184352를 유효성분으로 포함하는 sncg 과발현 질환의 예방 또는 치료용 약학적 조성물 |
| WO2023214325A1 (en) | 2022-05-05 | 2023-11-09 | Novartis Ag | Pyrazolopyrimidine derivatives and uses thereof as tet2 inhibitors |
| CA3256012A1 (en) | 2022-05-20 | 2023-11-23 | Les Laboratoires Servier | ANTIBODY-DRUG CONJUGATES, MET INHIBITORS BCL-XL, AND THEIR METHODS OF USE |
| KR20250027281A (ko) | 2022-05-20 | 2025-02-25 | 노파르티스 아게 | Epha2 bcl-xl 억제제 항체-약물 접합체 및 그의 사용 방법 |
| CN119301125A (zh) | 2022-07-26 | 2025-01-10 | 诺华股份有限公司 | Akr1c3依赖性kars抑制剂的晶型 |
| KR20250160354A (ko) | 2023-03-10 | 2025-11-12 | 노파르티스 아게 | Panras 억제제 항체-약물 콘쥬게이트 및 이의 사용 방법 |
| TW202540185A (zh) | 2023-11-22 | 2025-10-16 | 法商施維雅藥廠 | 抗cd74抗體藥物結合物及其使用方法 |
| WO2025215536A1 (en) | 2024-04-10 | 2025-10-16 | Novartis Ag | Macrocyclic panras inhibitors for the treatment of cancer |
Family Cites Families (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB9412711D0 (en) * | 1994-06-24 | 1994-08-17 | Cortecs Ltd | Medical use of bromelain |
| US5525625A (en) * | 1995-01-24 | 1996-06-11 | Warner-Lambert Company | 2-(2-Amino-3-methoxyphenyl)-4-oxo-4H-[1]benzopyran for treating proliferative disorders |
| US6251943B1 (en) * | 1997-02-28 | 2001-06-26 | Warner-Lambert Company | Method of treating or preventing septic shock by administering a MEK inhibitor |
| GB9713726D0 (en) * | 1997-06-30 | 1997-09-03 | Ciba Geigy Ag | Organic compounds |
| JP2002515891A (ja) * | 1997-12-19 | 2002-05-28 | スミスクライン・ビーチャム・コーポレイション | 新規なピペリジン含有化合物 |
| YU67600A (sh) * | 1998-05-05 | 2003-07-07 | F.Hofmann - La Roche Ag. | Derivati pirazola kao inhibitori p-38 map kinaze |
| EP1082320A4 (en) * | 1998-05-26 | 2001-11-21 | Smithkline Beecham Corp | NEW SUBSTITUTED IMIDAZOLE COMPOUNDS |
-
1999
- 1999-12-15 KR KR1020017007502A patent/KR100609800B1/ko not_active Expired - Fee Related
- 1999-12-15 HU HU0104693A patent/HUP0104693A3/hu unknown
- 1999-12-15 WO PCT/US1999/029783 patent/WO2000035436A2/en not_active Ceased
- 1999-12-15 AU AU21858/00A patent/AU776788C/en not_active Ceased
- 1999-12-15 IL IL14323699A patent/IL143236A0/xx unknown
- 1999-12-15 EP EP99966278A patent/EP1143957A3/en not_active Withdrawn
- 1999-12-15 CA CA002346448A patent/CA2346448A1/en not_active Abandoned
- 1999-12-15 JP JP2000587757A patent/JP2002532415A/ja active Pending
Also Published As
| Publication number | Publication date |
|---|---|
| HUP0104693A3 (en) | 2003-12-29 |
| AU2185800A (en) | 2000-07-03 |
| KR20010093840A (ko) | 2001-10-29 |
| EP1143957A2 (en) | 2001-10-17 |
| AU776788B2 (en) | 2004-09-23 |
| WO2000035436A3 (en) | 2001-10-18 |
| AU776788C (en) | 2005-10-27 |
| JP2002532415A (ja) | 2002-10-02 |
| EP1143957A3 (en) | 2002-02-27 |
| IL143236A0 (en) | 2002-04-21 |
| CA2346448A1 (en) | 2000-06-22 |
| HUP0104693A2 (hu) | 2002-03-28 |
| WO2000035436A2 (en) | 2000-06-22 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| KR100609800B1 (ko) | Mek 저해제를 사용한 관절염 치료 방법 | |
| US6251943B1 (en) | Method of treating or preventing septic shock by administering a MEK inhibitor | |
| KR20010099877A (ko) | 배합물 화학요법 | |
| US6696440B1 (en) | Treatment of asthma with MEK inhibitors | |
| US6492363B2 (en) | 2-(4-bromo or 4-iodo phenylamino) benzoic acid derivatives | |
| JP2002534381A (ja) | Mek阻害剤を用いた抗ウィルス法 | |
| KR20010101203A (ko) | Mek 저해제의 이식 조직 거부를 예방하기 위한 용도 | |
| KR20010014360A (ko) | 2-(4-브로모 또는 4-요오도 페닐아미노)벤조산 유도체 및mek 억제제로서의 그의 용도 | |
| US20040171632A1 (en) | Combination chemotherapy | |
| MXPA01005476A (en) | Combination chemotherapy |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PA0105 | International application |
St.27 status event code: A-0-1-A10-A15-nap-PA0105 |
|
| PG1501 | Laying open of application |
St.27 status event code: A-1-1-Q10-Q12-nap-PG1501 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| PN2301 | Change of applicant |
St.27 status event code: A-3-3-R10-R13-asn-PN2301 St.27 status event code: A-3-3-R10-R11-asn-PN2301 |
|
| A201 | Request for examination | ||
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| PA0201 | Request for examination |
St.27 status event code: A-1-2-D10-D11-exm-PA0201 |
|
| E902 | Notification of reason for refusal | ||
| PE0902 | Notice of grounds for rejection |
St.27 status event code: A-1-2-D10-D21-exm-PE0902 |
|
| E13-X000 | Pre-grant limitation requested |
St.27 status event code: A-2-3-E10-E13-lim-X000 |
|
| P11-X000 | Amendment of application requested |
St.27 status event code: A-2-2-P10-P11-nap-X000 |
|
| P13-X000 | Application amended |
St.27 status event code: A-2-2-P10-P13-nap-X000 |
|
| E701 | Decision to grant or registration of patent right | ||
| PE0701 | Decision of registration |
St.27 status event code: A-1-2-D10-D22-exm-PE0701 |
|
| GRNT | Written decision to grant | ||
| PR0701 | Registration of establishment |
St.27 status event code: A-2-4-F10-F11-exm-PR0701 |
|
| PR1002 | Payment of registration fee |
St.27 status event code: A-2-2-U10-U12-oth-PR1002 Fee payment year number: 1 |
|
| PG1601 | Publication of registration |
St.27 status event code: A-4-4-Q10-Q13-nap-PG1601 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |
|
| LAPS | Lapse due to unpaid annual fee | ||
| PC1903 | Unpaid annual fee |
St.27 status event code: A-4-4-U10-U13-oth-PC1903 Not in force date: 20090801 Payment event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE |
|
| PC1903 | Unpaid annual fee |
St.27 status event code: N-4-6-H10-H13-oth-PC1903 Ip right cessation event data comment text: Termination Category : DEFAULT_OF_REGISTRATION_FEE Not in force date: 20090801 |
|
| R18-X000 | Changes to party contact information recorded |
St.27 status event code: A-5-5-R10-R18-oth-X000 |