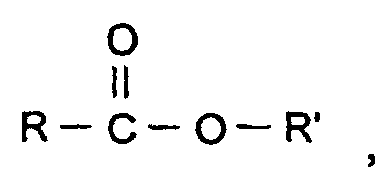EP1252268B1 - Method of reducing the vapour pressure of ethanol-containing motor fuels for spark ignition combustion engines - Google Patents
Method of reducing the vapour pressure of ethanol-containing motor fuels for spark ignition combustion engines Download PDFInfo
- Publication number
- EP1252268B1 EP1252268B1 EP01942659A EP01942659A EP1252268B1 EP 1252268 B1 EP1252268 B1 EP 1252268B1 EP 01942659 A EP01942659 A EP 01942659A EP 01942659 A EP01942659 A EP 01942659A EP 1252268 B1 EP1252268 B1 EP 1252268B1
- Authority
- EP
- European Patent Office
- Prior art keywords
- ethanol
- volume
- dvpe
- gasoline
- kpa
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Lifetime
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/02—Liquid carbonaceous fuels essentially based on components consisting of carbon, hydrogen, and oxygen only
- C10L1/023—Liquid carbonaceous fuels essentially based on components consisting of carbon, hydrogen, and oxygen only for spark ignition
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/18—Organic compounds containing oxygen
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L10/00—Use of additives to fuels or fires for particular purposes
- C10L10/02—Use of additives to fuels or fires for particular purposes for reducing smoke development
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L10/00—Use of additives to fuels or fires for particular purposes
- C10L10/10—Use of additives to fuels or fires for particular purposes for improving the octane number
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/16—Hydrocarbons
- C10L1/1608—Well defined compounds, e.g. hexane, benzene
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/16—Hydrocarbons
- C10L1/1616—Hydrocarbons fractions, e.g. lubricants, solvents, naphta, bitumen, tars, terpentine
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/18—Organic compounds containing oxygen
- C10L1/182—Organic compounds containing oxygen containing hydroxy groups; Salts thereof
- C10L1/1822—Organic compounds containing oxygen containing hydroxy groups; Salts thereof hydroxy group directly attached to (cyclo)aliphatic carbon atoms
- C10L1/1824—Organic compounds containing oxygen containing hydroxy groups; Salts thereof hydroxy group directly attached to (cyclo)aliphatic carbon atoms mono-hydroxy
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/18—Organic compounds containing oxygen
- C10L1/185—Ethers; Acetals; Ketals; Aldehydes; Ketones
- C10L1/1852—Ethers; Acetals; Ketals; Orthoesters
- C10L1/1855—Cyclic ethers, e.g. epoxides, lactides, lactones
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/18—Organic compounds containing oxygen
- C10L1/185—Ethers; Acetals; Ketals; Aldehydes; Ketones
- C10L1/1857—Aldehydes; Ketones
-
- C—CHEMISTRY; METALLURGY
- C10—PETROLEUM, GAS OR COKE INDUSTRIES; TECHNICAL GASES CONTAINING CARBON MONOXIDE; FUELS; LUBRICANTS; PEAT
- C10L—FUELS NOT OTHERWISE PROVIDED FOR; NATURAL GAS; SYNTHETIC NATURAL GAS OBTAINED BY PROCESSES NOT COVERED BY SUBCLASSES C10G OR C10K; LIQUIFIED PETROLEUM GAS; USE OF ADDITIVES TO FUELS OR FIRES; FIRE-LIGHTERS
- C10L1/00—Liquid carbonaceous fuels
- C10L1/10—Liquid carbonaceous fuels containing additives
- C10L1/14—Organic compounds
- C10L1/18—Organic compounds containing oxygen
- C10L1/19—Esters ester radical containing compounds; ester ethers; carbonic acid esters
Definitions
- This invention relates to motor fuel for spark ignition internal combustion engines. More particularly the invention relates to a method for lowering the dry vapour pressure equivalent (DVPE) of a fuel composition including a hydrocarbon liquid and ethanol by using an oxygen-containing additive.
- the ethanol and DVPE adjusting components used to obtain the fuel composition are preferably derived from renewable raw materials.
- motor fuels containing up to 20 % by volume of ethanol meeting standard requirements for spark ignition internal combustion engines operating with gasoline are obtainable.
- Gasoline is the major fuel for spark ignition internal combustion engines.
- the extensive use of gasoline results in the pollution of the environment.
- the combustion of gasoline derived from crude oil or mineral gas disturbs the carbon dioxide balance in the atmosphere, and causes the greenhouse effect. Crude oil reserves are decreasing steadily with some countries already facing crude oil shortages.
- U.S. Patent No. 2,365,009, issued in 1944 describes the combination of C 1-5 , alcohols and C 3-5 hydrocarbons for use as a fuel.
- U.S. Patent No. 4,818,250 issued in 1989 it is proposed to use limonene obtained from citrus and other plants as a motor fuel, or as a component in blends with gasoline.
- U.S. Patent No. 5,607,486 issued in 1997 there are disclosed novel engine fuel additives comprising terpenes, aliphatic hydrocarbons and lower alcohols.
- Zudkevitch D. et al. in "Thermodynamics of reformulated automotive fuels" Hydrocarbon processing, vol 74, No. 6, 1995 disclose in table 5 thereof two fuel compositions comprising base gasoline, ethanol (5% and 3.2%, respectively) and 12% of ETBE. Using the vapour pressure data given in table 5 it can be calculated that the two fuel compositions exhibit a remaining ethanol induced vapour pressure increase of 32.1 and 30.3%, respectively.
- tert-butyl ethers are widely used as components of gasolines.
- Motor fuels comprising tert-butyl ethers are described in U.S. Patent No. 4,468,233 issued in 1984.
- the major portion of these ethers is obtained from petroleum refining, but can equally be produced from renewable resources.
- Ethanol is a most promising product for use as a motor fuel component in mixtures with gasoline.
- Ethanol is obtained from the processing of renewable raw material, known generically as biomass, which, in turn, is derived from carbon dioxide under the influence of solar energy.
- Figure 1 shows the behaviour of the dry vapour pressure equivalent (DVPE) as a function of the ethanol content of mixtures of ethanol and gasoline A92 summer, and gasoline A95 summer and winter at 37.8°C.
- the gasolines known as A92 and A95 are standard gasolines purchased at gas stations in the United States and Sweden.
- Gasoline A92 originated in the United States and gasoline A95, in Sweden.
- the ethanol employed was fuel grade ethanol produced by Williams, USA.
- the DVPE of the mixtures was determined according to the standard ASTM D5191 method at the SGS laboratory in Sweden.
- Fig. 1 For the range of concentrations by volume of ethanol between 5 and 10% which is of particular interest for use as a motor fuel for standard spark ignition engines, the data in Fig. 1 show that the DVPE of mixtures of gasoline and ethanol can exceed the DVPE of source gasoline by more than 10%. Since the commercial petroleum companies normally supply the market with gasoline already at the maximum allowed DVPE, which is strictly limited by current regulations, the addition of ethanol to such presently commercially available gasolines is not possible.
- U.S. Patent No. 5,688,295 granted on November 18, 1997 provides a chemical compound as an additive to gasoline or as a fuel for standard gasoline engines.
- an alcohol-based fuel additive is proposed.
- the fuel additive comprises from 20 - 70% alcohol, from 2.5 - 20% ketone and ether, from 0.03 - 20% aliphatic and silicon compounds, from 5 - 20% toluene and from 4 - 45% mineral spirits.
- the alcohol is methanol or ethanol. It is noted in the patent that the additive improves gasoline quality and specifically decreases DVPE.
- the disadvantages of this method of motor fuel DVPE adjustment are that there is a need for large quantities of the additive, namely, not less than 15 % by volume of the mixture; and the use of silicon compounds, which form silicon oxide upon combustion, results in increased engine wear.
- a spark ignition motor fuel composition including a hydrocarbon component of C 5 - C 8 straight-chained or branched alkanes, essentially free of olefins, aromatics, benzene and sulphur, in which the hydrocarbon component has a minimum anti-knock index of 65, according to ASTM D2699 and D2700 and a maximum DVPE of 15 psi, according to ASTM D5191; a fuel grade alcohol; and a co-solvent for the hydrocarbon component and alcohol in which the components of the fuel composition are present in amounts selected to provide a motor fuel with a minimum anti-knock index of 87 and a maximum DVPE of 15 psi.
- the co-solvent used is biomass-derived 2-methyltetrahydrofuran (MTHF) and other heterocyclical ether
- the present inventors have found that specific types of compounds exhibiting an oxygen-containing group surprisingly lower the vapour pressure of a gasoline-ethanol mixture.
- up to about 20 % by volume of fuel grade ethanol (b) can be used in the whole fuel compositions.
- the oxygen-containing additives (c) used can be obtained from renewable raw materials, and the hydrocarbon component (a) used can for example be any standard gasoline (which does not have to be reformulated) and can optionally contain aromatic fractions and sulphur, and also hydrocarbons obtained from renewable raw materials.
- fuels for standard spark ignition internal combustion engines can be prepared, which fuels allow such engines to have the same maximum performance as when operated on standard gasoline currently on the market.
- a decrease in the level of toxic emissions in the exhaust and a decrease in the fuel consumption can also be obtained by using the method of the invention.
- the anti-knock index in addition to the dry vapour pressure equivalent (DVPE), the anti-knock index (octane number) can also be desirably controlled.
- the mixture of (b), (c), and (d) can also be used per se as a fuel for modified engines, i.e., not standard-type gasoline engines.
- the additive mixture can also be used for adjusting the octane number and/or for lowering the vapour pressure of a high vapour pressure hydrocarbon component.
- the present method enables the use of C 3 - C 12 hydrocarbon fractions as hydrocarbon component (a), including narrower ranges within this broader range, without restriction on the presence of saturated and unsaturated hydrocarbons, aromatics and sulphur.
- the hydrocarbon component can be a standard gasoline currently on the market, as well as other mixtures of hydrocarbons obtained in the refining of petroleum, off-gas of chemical-recovery coal carbonisation, natural gas and synthesis gas. Hydrocarbons obtained from renewable raw materials can also be included.
- the C 3 - C 12 fractions are usually prepared by fractional distillation or by blending various hydrocarbons.
- the component (a) can contain aromatics and sulphur, which are either co-produced or naturally found in the hydrocarbon component.
- the DVPE can be reduced for fuel mixtures containing up to 20% volume of ethanol, calculated as pure ethanol.
- the vapour pressure of the hydrocarbon based ethanol-containing fuel mixture is reduced by 80% of the ethanol-induced vapour pressure increase, and more preferably the vapour pressure of the hydrocarbon based ethanol-containing fuel mixture is reduced to a vapour pressure corresponding to that of the hydrocarbon component alone.
- the DVPE can be reduced if desired to a level even lower than that of the hydrocarbon component used.
- the other properties of the fuel such as for example the octane number, are kept within the required standard limits.
- the oxygen-containing organic compound enables adjustment of (i) the dry vapour pressure equivalent, (ii) the anti-knock index and other performance parameters of the motor fuel composition as well as (iii) the reduction of the fuel consumption and the reduction of toxic substances in the engine exhaust emissions.
- the oxygen-containing compound (c) has oxygen bound in at least any one of the following functional groups:
- Such functional groups are present, for example, in the following classes of organic compounds and which can be used in the present invention: alcohols, ketones, ethers, esters, hydroxy-ketones, ketone esters, and heterocyclics with oxygen-containing rings.
- the fuel additive can be derived from fossil-based sources or preferably from renewable sources such as biomass.
- the oxygen-containing fuel additive (c) can typically be an alcohol, other than ethanol.
- aliphatic or alicyclic alcohols both saturated and unsaturated, preferably alkanols, are employed. More preferably, alkanols of the general formula: R-OH where R is alkyl with 3 to 10 carbon atoms, most preferably 3 to 8 carbon atoms, such as propanol, isopropanol, n-butanol, isobutanol, tert-butanol, n-pentanol, isopentanol, tert-pentanol, 4-methyl-2-pentanol, diethylcarbinol, diisopropylcarbinol, 2-ethylhexanol, 2,4,4-trimethylpentanol, 2,6-dimethyl-4-heptanol, linalool, 3,6-dimethyl-3-octanol, phenol, phenyl
- the component (c) can also be an aliphatic or alicyclic ketone, both saturated and unsaturated, of the general formula where R and R' are the same or different and are each C 1 -C 6 hydrocarbons, which also can be cyclic, and are preferably C 1 -C 4 hydrocarbons.
- ketones have a total (R+R') of 4 to 9 carbon atoms and include methylethyl ketone, methylpropyl ketone, diethylketone, methylisobutyl ketone, 3-heptanone, 2-octanone, diisobutyl ketone, cyclohexanon, acetofenone, trimethylcycohexanone, or similar ketones, and mixtures thereof.
- the component (c) can also be an aliphatic or alicyclic ether, including both saturated and unsaturated ethers, of the general formula R-O-R', wherein R and R' are the same or different and are each a C 1 -C 10 hydrocarbon group.
- R and R' are the same or different and are each a C 1 -C 10 hydrocarbon group.
- lower (C 1 -C 6 ) dialkyl ethers are preferred.
- the total number of carbon atoms in the ether is preferably from 6 to 10.
- Typical ethers include methyltertamyl ether, methylisoamyl ether, ethylisobutyl ether, ethyltertbutyl ether, dibutyl ether, diisobutyl ether, diisoamyl ether, anisole, methylanisole, phenetole or similar ethers and mixtures thereof.
- the component (c) may further be an aliphatic or alicyclic ester, including saturated and unsaturated esters, of the general formula where R and R' are the same or different.
- R and R' are preferably hydrocarbon groups, more preferably alkyl groups and most preferably alkyl and phenyl having 1 to 6 carbon atoms. Especially preferred is an ester where R is C 1 -C 4 and R' is C 4 -C 6 .
- Typical esters are alkyl esters of alkanoic acids, including n-butylacetate, isobutylacetate, tert-butylacetate, isobutylpropionate, isobutylisobutyrate, n-amylacetate, isoamylacetate, isoamylpropionate, methylbenzoate, phenylacetate, cyclohexylacetate, or similar esters and mixtures thereof. In general, it is preferred to employ an ester having from 5 to 8 carbon atoms.
- the additive (c) can simultaneously contain two oxygen-containing groups connected in the same molecule with different carbon atoms.
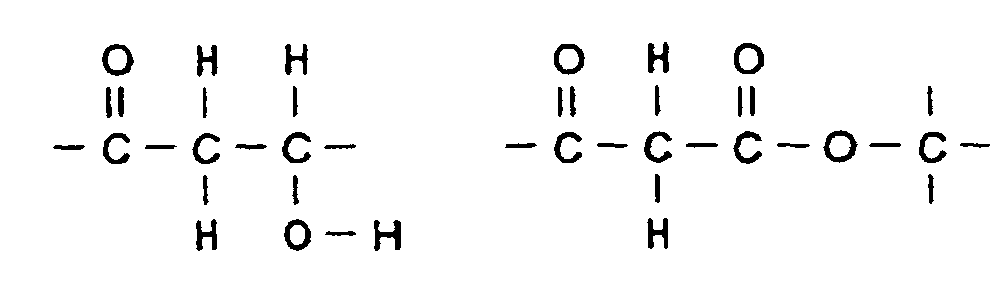
- the additive (c) can be a hydroxyketone.
- a preferred hydroxyketone has the general formula: where R is hydrocarbyl, and R 1 is hydrogen or hydrocarbyl, preferably lower alkyl, i.e. (C 1 -C 4 ). In general, it is preferred to employ a ketol having 4 to 6 carbon atoms.
- Typical hydroxy-ketones include 1-hydroxy-2-butanone, 3-hydroxy-2-butanone, 4-hydroxy-4-methyl-2-pentanone, or similar ketols or mixture thereof.
- the fuel additive (c) is a ketone ester, preferably of the general formula: where R is hydrocarbyl, preferably lower alkyl, i.e. (C 1 -C 4 ).
- Typical ketone esters include methylacetoacetate, ethyl acetoacetate and tert-butyl acetoacetate. Preferably, such ketone esters have 6 to 8 carbon atoms.
- the additive (c) can also be a ring-oxygen-containing heterocyclic compound and, preferably, the oxygen-containing heterocycle has a C 4 - C 5 ring. More preferably, the heterocycle additive has a total of 5 to 8 carbon atoms.
- the additive can preferably have the formula (1) or (2) as follows: where R is hydrogen or hydrocarbyl, preferably -CH 3 , and R 1 is -CH 3 , or -OH, or -CH 2 OH, or CH 3 CO 2 CH 2 -.
- a typical heterocyclic additive (c) is tetrahydrofurfuryl alcohol, tetrahydrofurfurylacetate, dimethyltetrahydrofurane, tetramethyltetrahydrofurane, methyltetrahydropyrane, 4-methyl-4-oxytetrahydropyrane or similar heterocyclic additives, or mixtures thereof.
- Component (c) can also be a mixture of any of the compounds set out above from one or more of the above-mentioned different compound classes.
- Suitable fuel grade ethanol (b) to be used according to the present invention can readily be identified by the person skilled in the art.
- a suitable example of the ethanol component is ethanol containing 99.5% of the main substance.
- Any impurities included in the ethanol in an amount of at least 0.5 % by volume thereof and falling within the above-mentioned definition of component (c) should be taken into account when determining the amount used of component (c). That is, such impurities must be included in an amount of at least 0.5% in the ethanol in order to be taken into account as a part of component (c).
- Any water, if present in the ethanol should preferably amount to no more than about 0.25 % by volume of the total fuel mixture, in order to meet the current standard requirements on fuels for gasoline engines.
- a denatured ethanol mixture as supplied to the market containing about 92% of ethanol, hydrocarbons and by-products, can also be used as the ethanol component in the fuel composition according to the invention.
- the ethanol (b) is employed in amounts from 0.1% to 20%, typically from about 1% to 20 % by volume, preferably 3% to 15 % by volume and more preferably from about 5 to 10 % by volume.
- the oxygen-containing additive (c) is generally employed in amounts from 0.05% to about 15 % by volume, more generally from 0.1 to about 15 % by volume, preferably from about 3 - 10 % by volume and most preferably from about 5 to 10 % by volume.
- the total volume of ethanol (b) and oxygen-containing additive (c) employed is from 0.15 to 25 % by volume, normally from about 0.5 to 25 % by volume, preferably from about 1 to 20 % by volume, more preferably from 3 to 15 % by volume, and most preferably from 5 to 15 % by volume.
- the ratio of ethanol (b) to oxygen-containing additive (c) in the motor fuel composition is thus generally from 1:150 to 400:1, and is more preferably from 1:10 to 10:1.
- the total oxygen content of motor fuel composition based on the ethanol and the oxygen additive is preferably no greater than about 7 wt.%, more preferably no greater than about 5 wt.%.
- a motor fuel suitable for the operation of a standard spark ignition internal combustion engine the aforesaid hydrocarbon component, ethanol, and additional oxygen-containing component are admixed to obtain the following properties of the resulting motor fuel composition:
- the hydrocarbon component and ethanol should be added together, followed by the addition of the additional oxygen-containing compound or compounds to the mix. Afterwards, the resulting motor fuel composition should preferably be maintained at a temperature not lower than -35°C, for at least about one hour. It is a feature of this invention that the components of the motor fuel composition can be merely added to each other to form the desired composition. It is generally not required to agitate or otherwise provide any significant mixing to form the composition.
- oxygen-containing component(s) originating from renewable raw material(s).
- Component (d) is used for further lowering the vapour pressure of the fuel mixture of components (a), (b) and (c).
- An individual hydrocarbon selected from a C 6 - C 12 fraction of aliphatic or alicyclic saturated and unsaturated hydrocarbons can be used as component (d).
- the hydrocarbon component (d) is selected from a C 8 -C 11 fraction.
- Suitable examples of (d) are benzene, toluene, xylene, ethylbenzene, isopropylbenzene, isopropyltoluene, diethylbenzene, isopropylxylene, tert-butylbenzene, tert-butyltoluene, tert-butylxylene, cyclooctadiene, cyclooctotetraene, limonene, isooctane, isononane, isodecane, isooctene, myrcene, allocymene, tert-butylcyclohexane or similar hydrocarbons and mixtures hereof.
- Hydrocarbon component (d) can also be a fraction boiling at 100-200°C, obtained in the distillation of oil, bituminous coal resin, or synthesis gas processing products.
- the invention further relates to an additive mixture consisting of components (b) and (c) and, optionally also component (d), which subsequently can be added to the hydrocarbon component (a) and is also possible to use as such as a fuel for a modified spark ignition combustion engine.
- the additive mixture preferably has a ratio of ethanol (b) to additive (c) of 1: 150 to 200:1 by volume.
- said mixture comprises the oxygen-containing component (c) in an amount from 0.5 up to 99.5 % by volume, and ethanol (b) in an amount from 0.5 up to 99.5 % by volume, and component (d) comprising at least one C 6 - C 12 hydrocarbon, more preferably C 8 -C 11 hydrocarbon, in an amount from 0 up to 99 % by volume, preferably from 0% up to 90%, more preferably from 0 up to 79,5%, and most preferably from 5 up to 77% of the additive mixture.
- the additive mixture has a ratio of ethanol (b) to the sum of the other additive components (c)+(d) from 1:200 to 200:1 by volume, more preferable a ratio of ethanol (b) to the sum of the components (c) + (d) is from 1:10 to 10:1 by volume.
- the octane number of the additive mixture can be established, and the mixture be used to adjust the octane number of the component (a) to a desired level by admixing a corresponding portion of the mixture (b), (c), (d) to component (a).
- the drivability testing was performed on a 1987 VOLVO 240 DL according to the standard test method EU2000 NEDC EC 98/69.
- EU 2000 European 2000
- NEDC New European Driving Cycle
- 91/441 EEC resp. ECE-R 83/01 and 93/116 EEC
- These standardised EU tests include city driving cycles and extra urban driving cycles and require that specific emission regulations be met.
- Exhaust emission analysis is conducted with a constant volume sampling procedure and utilises a flame ionisation detector for hydrocarbon determination.
- Exhaust Emission Directive 91/441 EEC Phase I
- EU Fuel Consumption Directive 93/116 EEC (1996) implements consumption standards.
- the testing was performed on a 1987 Volvo 240 DL with a B230F, 4-cylinder, 2.32 litre engine (No. LG4F20-87) developing 83 kW at 90 revolutions/second and a torque of 185 Nm at 46 revolutions/ second.
- Example 1 demonstrates the possibility of reducing the dry vapour pressure equivalent of the ethanol-containing motor fuel for the cases when gasolines with dry vapour pressure equivalent according to ASTM D-5191 at a level of 90 kPa (about 13 psi) are used as a hydrocarbon base.
- Fig. 1 demonstrates the behaviour of the DVPE of the ethanol-containing motor fuel based on winter A95 gasoline.
- the ethanol-containing motor fuels based on winter A92 and A98 used in this example also demonstrate a similar behaviour.
- the source gasoline comprised aliphatic and alicyclic C 4 -C 12 hydrocarbons, including both saturated and unsaturated ones.
- the following different embodiments of the fuels 1-3 demonstrate the possibility of adjusting the dry vapor pressure equivalent (DVPE) of the ethanol-containing motor fuel based on winter A92 gasoline.
- DVPE dry vapor pressure equivalent
- the inventive fuel 1-3 contained A92 winter gasoline (a), ethanol (b), oxygen-containing additives (c) and hydrocarbons C 6 -C 12 (d), and had the following properties for the various compositions:
- A92 : Ethanol : Isoamyl alcohol : Alkylate 79 : 9 : 2 : 10 % by volume
- Ethanol : Isobutyl acetate : Naphtha 80 : 5 : 5 : 10 % by volume
- Ethanol: Tert-butanol: Naphtha 81 : 5 : 5 : 9 % by volume
- the boiling temperature for the naphtha is
- the motor fuel compositions below demonstrate that it is not always necessary to reduce the excess DVPE of the motor fuel induced by the presence of ethanol to the level of DVPE of the source gasoline. In some cases it is sufficient just to bring it in compliance with the requirements of the regulations in force for the corresponding gasoline.
- the DVPE level for the winter gasoline is 90 kPa.
- compositions 1-6 demonstrate the possibility of adjusting the dry vapor pressure equivalent (DVPE) of the ethanol-containing motor fuel based on winter A98 gasoline.
- the fuel 1-6 contained A98 winter gasoline (a), ethanol (b), oxygen-containing additives (c), and C 6 -C 12 hydrocarbons (d) and had the following properties for the various compositions:
- A98 : Ethanol: Isobutanol : Naphtha 80 : 5 : 5 : 10 % by volume The boiling point of the naphtha is 100-200°C.
- the motor fuel compositions below demonstrate that it is not always necessary to reduce the excess DVPE of the motor fuel caused by presence of ethanol to the level of DVPE of the source gasoline. In some cases it is sufficient just to bring it in compliance with the requirements of the regulations in force for the corresponding gasoline.
- the DVPE level for the winter gasoline is 90 kPa.
- the motor fuel compositions below demonstrate that it might be necessary to reduce the excess DVPE of the motor fuel caused by presence of ethanol below the level of DVPE of the source gasoline. Normally, this is required when DVPE of the source gasoline is higher than the limits of the regulations in force for the corresponding spending gasoline. In this way, for example, it is possible to transform the winter grade gasoline into the summer grade gasoline.
- the DVPE level for the summer gasoline is 70 kPa.
- A98 : Ethanol : Isobutanol : Isooctane : Naphtha 60 : 9.5 : 0.5 : 15 : 15 % by volume
- the boiling point of the naphtha is 100-200°C.
- the following fuels 1-9 and 1-10 demonstrate the possibility of adjusting the dry vapor pressure equivalent (DVPE) of the ethanol-containing motor fuel based on winter A95 gasoline.
- DVPE dry vapor pressure equivalent
- the fuel 1-9 contained A95 winter gasoline (a), ethanol (b), the oxygen-containing additives (c), and C 6 -C 12 hydrocarbons (d) and had the following properties for the various compositions:
- A95 : Ethanol: Isoamyl alcohol : Alkylate 83.7 : 5 : 2 : 9.3 % by volume
- the motor fuel compositions below demonstrate that it is not always necessary to reduce the excess DVPE of the motor fuel caused by presence of ethanol to the level of DVPE of the source gasoline. In some cases it is sufficient just to bring it in compliance with the requirements of the regulations in force for the corresponding gasoline.
- the DVPE level for the winter gasoline is 90 kPa.
- the motor fuel compositions below demonstrate that it might be necessary to reduce the excess DVPE of the motor fuel caused by presence of ethanol below the level of DVPE of the source gasoline. Normally, this is required when DVPE of the source gasoline is higher than the limits of the regulations in force for the corresponding gasoline. In this way, for example, it is possible to transform the winter grade gasoline into the summer grade gasoline.
- the DVPE level for the summer gasoline is 70 kPa.
- A95 : Ethanol : Isobutanol : Isoamyl alcohol : Naphtha : Isooctane 60 : 9.2 : 0.2 : 0.6 : 15 : 15 % by volume
- the boiling temperature of the naphtha is 100-200°C.
- Ethanol: Tert-butyl acetate : Naphtha 60 : 9 : 1 : 30 % by volume
- the boiling temperature of the naphtha is 100-200°C.
- the fuel 1-10 contains 75 % by volume A95 winter gasoline, 9.6 % by volume ethanol, 0.4 % by volume isobutyl alcohol, 4.5 % by volume m-isopropyl toluene and 10.5 % by volume naphtha with boiling temperature of 100-200°C.
- This fuel formulation demonstrates the possibility of decreasing the DVPE, increasing the octane number, decreasing the level of toxic emissions in the exhaust and decreasing the fuel consumption in comparison with the reference mixture of gasoline and ethanol (RFM 1).
- the motor fuel composition has the following properties: density at 15°C, according to ASTM D 4052 749.2 kg /m3; initial boiling point, according to ASTM D 86 29°C; vaporizable portion - 70°C 47.6 % by volume; vaporizable portion - 100°C 55.6 % by volume; vaporizable portion - 150°C 84.2 % by volume; vaporizable portion - 180°C 97.5 % by volume; final boiling point 194.9°C; evaporation residue 1.3 % by volume; loss by evaporation 1.6 % by volume; oxygen content, according to ASTM D4815 acidity, according to ASTM D 1613 3.7%w/w; weight% HAc 0.004; pH, according to ASTM D1287 6.6; sulfur content, according to ASTM D 5453 18mg/kg; gum content, according to ASTM D381 1 mg/100ml; water content, according to ASTM D6304 aromatics, according to SS 155120,
- the motor fuel formulation 1-10 was tested in accordance with the standard test method EU 2000 NEDC EC 98/69 and the following results, as compared to winter A95 gasoline, were obtained: CO -21%; HC -9%; NOx + 12.8%; CO2 +2.38%; NMHC -6.4%; Fuel consumption, Fc 1/100km +3.2%
- the fuel formulations 1-1 to 1-10 showed reduced DVPE over the tested ethanol-containing motor fuels based on summer grade gasoline. Similar results are obtained when other oxygen-containing compounds of this invention are substituted for the additives of the examples 1-1 to 1-10.
- Example 2 demonstrates the possibility of reducing the dry vapour pressure equivalent of the ethanol-containing motor fuel for the cases when gasolines with a dry vapour pressure equivalent according to ASTM D-5191 at a level of 70 kPa (about 10 psi) are used as a hydrocarbon base.
- the source gasoline comprised aliphatic and alicyclic C 4 -C 12 hydrocarbons, including saturated and unsaturated ones.
- Figure 1 shows the behaviour of the DVPE of the ethanol-containing motor fuel based on summer A95 gasoline.
- the fuel 2-3 contained A92 summer gasoline (a), ethanol (b), the oxygen-containing additives (c), and C 6 -C 12 hydrocarbons (d) and had the following properties for the various compositions:
- Ethanol Me
- the motor fuel compositions below demonstrate that it is not always necessary to reduce the excess DVPE of the motor fuel caused by presence of ethanol to the level of DVPE of the source gasoline. In some cases it is sufficient just to bring it in compliance with the requirements of the regulations in force for the corresponding gasoline.
- the DVPE level for the summer gasoline is 70 kPa.
- the following fuels 2-6 demonstrate the possibility of adjusting the dry vapour pressure equivalent (DVPE) of the ethanol-containing motor fuel based on summer A98 gasoline.
- DVPE dry vapour pressure equivalent
- the fuel 2-6 contained A98 summer gasoline (a), ethanol (b), the oxygen-containing additives (c), and C 6 -C 12 hydrocarbons (d) and had the following properties for the various compositions:
- the motor fuel compositions below demonstrate that it is not always necessary to reduce the excess DVPE of the motor fuel caused by the presence of ethanol to the level of DVPE of the source gasoline. In some cases it is sufficient just to bring it in compliance with the requirements of the regulations in force for the corresponding gasoline.
- the DVPE level for the summer gasoline is 70 kPa.
- the following fuels 2-9 to 2-10 demonstrate the possibility of adjusting the dry vapour pressure equivalent (DVPE) of the ethanol-containing motor fuel based on summer A95 gasoline.
- DVPE dry vapour pressure equivalent
- the fuel 2-9 contained A95 summer gasoline (a), ethanol (b), the oxygen-containing additives (c), and C 6 -C 12 hydrocarbons (d) and had the following properties for the various compositions:
- Ethanol: Diethylketone : Xylene 80 : 9.5
- the motor fuel compositions below demonstrate that it is not always necessary to reduce the excess DVPE of the motor fuel caused by the presence of ethanol to the level of DVPE of the source gasoline. In some cases it is sufficient just to bring it in compliance with the requirements of the regulations in force for the corresponding gasoline.
- the DVPE level for the summer gasoline is 70 kPa.
- the fuel formulation 2-10 contained 81.5% by volume of A95 summer gasoline, 8.5% by volume of m-isopropyltoluene, 9.2% by volume of ethanol, and 0.8% by volume of isoamyl alcohol.
- Formulation 2-10 was tested to demonstrate how the inventive composition maintained the dry vapour pressure equivalent at a same level as the source gasoline while increasing the octane number, while decreasing the level of toxic emissions in the exhaust and decreasing the fuel consumption in comparison with the mixture RFM 2 of gasoline and ethanol.
- Formulation 2-10 had the following specific properties: density at 15°C, according to ASTM D4052 754.1kg/m3; initial boiling point, according to ASTM D 86 26.6°C; vaporisable portion - 70°C 45.2 % by volume; vaporisable portion - 100°C 56.4 % by volume; vaporisable portion - 150°C 88.8 % by volume; vaporisable portion - 180°C 97.6 % by volume; final boiling point 186.3°C; evaporation residue 1.6 % by volume; loss by evaporation 0.1 % by volume; oxygen content, according to ASTM D4815 3.56% w/w; acidity, according to ASTM D1613 weight% HAc 0.007; pH, according to ASTM D 1287 8.9; sulfur content, according to ASTM D 5453 16mg/kg; gum content, according to ASTM D381 ⁇ 1mg/100ml; water content, according to ASTM D6304 0.12% w/w; aromatic
- the motor fuel Formulation 2-10 was tested in accordance with test method EU 2000 NEDC EC 98/69 as above and gave the following results in comparison (+) or (-)% with the results for the source A95 summer gasoline: CO -0.18% HC -8.5%; NOx +5.3%; CO 2 +2.8%; NMHC -9%; Fuel consumption, Fc, 1/100km +3.1%
- the fuel formulations 2-1 to 2-10 showed reduced DVPE over the tested ethanol-containing motor fuels based on summer grade gasoline. Similar results are obtained when other oxygen-containing additives of the invention are substituted for the additives of the examples 2-1 to 2-10.
- Figure 2 shows the behaviour of the dry vapour pressure equivalent (DVPE) as a function of the ethanol content when mixing summer A95 gasoline with the additive mixture 3 comprising 35 % by volume of ethanol , 5 % by volume of isoamyl alcohol, and 60 % by volume of naphtha boiling at temperatures between 100-170°C.
- DVPE dry vapour pressure equivalent
- Figure 2 demonstrates that varying the ethanol content in gasoline within the range from 0 to 20% does not induce an increase of the vapour pressure for these compositions higher than the requirements of the standards for DVPE of the summer grade gasolines, which is 70 kPa.
- the ratio between ethanol and the oxygen-containing compound other than ethanol in the additive mixture, which is used for preparation of the ethanol-containing gasolines, is of substantial importance.
- the ratio between the components of the additive established by the present invention enables to adjust the vapour pressure of the ethanol-containing gasolines over a wide range.
- the additive mixture comprising ethanol and the oxygen-containing compound of this invention other than ethanol with the ratio of the present invention can be used as an independent motor fuel for the engines adapted for operation on ethanol.
- Example 3 demonstrates the possibility of reducing the dry vapour pressure equivalent of the ethanol-containing motor fuel for the cases when gasolines with dry vapour pressure equivalent according to ASTM D-5191 at a level of 48 kPa (about 7 pSi) are used as the hydrocarbon base.
- the source gasolines comprised aliphatic and alicyclic C 5 -C 12 hydrocarbons, including both saturated and unsaturated ones.
- Fig. 1 shows the behaviour of the DVPE of the ethanol-containing motor fuel based on US summer grade A92 gasoline.
- the fuel 3-3 contained US A92 summer gasoline (a), ethanol (b), the oxygen-containing additives (c), and C 6 -C 12 hydrocarbons (d) and had the following properties for the various compositions:
- A92 Ethanol : Isoamyl alcohol : Isobut
- the motor fuel compositions below demonstrate that it is not always necessary to reduce the excess DVPE of the motor fuel caused by the presence of ethanol to the level of DVPE of the source gasoline. In some cases it is sufficient just to bring it in compliance with the requirements of the regulations in force for the corresponding gasoline.
- the DVPE level for the US summer grade gasoline is 7 psi, which corresponds to 48.28 kPa.
- the fuel 3-6 contained US A98 summer gasoline (a), ethanol (b), the oxygen-containing additives (c), and C 6 -C 12 hydrocarbons (d) and had the following properties for the various compositions:
- A98 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Naphtha 75 : 9.2 : 0.3 : 0.1 : 15.4 % by volume
- the US summer A95 gasoline was used as a reference fuel for the testing performed according to EU2000 NEDC EC 98/69 test cycle on a 1987 Volvo 240 DL with a B230F, 4-cylinder, 2.32 litre engine (No. LG4F20-87) developing 83 kW at 90 revolutions/second and a torque of 185 Nm at 46 revolutions/second.
- the fuel 3-9 contained US A95 summer gasoline (a), ethanol (b), the oxygen-containing additives (c), and C 6 -C 12 hydrocarbons (d) and had the following properties for the various compositions:
- A95 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Naphtha 75 : 9.2 : 0.3 : 0.1 : 15.4 % by volume
- the motor fuel compositions below demonstrate that it is not always necessary to reduce the excess DVPE of the motor fuel caused by the presence of ethanol to the level of DVPE of the source gasoline. In some cases it is sufficient just to bring it in compliance with the requirements of the regulations in force for the corresponding gasoline.
- the DVPE level for the US summer grade gasoline is 7 pSi, which corresponds to 48.28 kPa.
- the boiling temperature for the naphtha is 100-200°C.
- the fuel formulation 3-10 contained 76 % by volume of US A95 summer gasoline, 9.2 % by volume of ethanol, 0.25 % by volume of isoamyl alcohol, 0.05 % by volume of isobutyl alcohol, 11.5 % by volume of naphtha with boiling temperature of 100-200°C, and 3 % by volume of isopropyltoluene.
- Formulation 3-10 was tested to demonstrate how the invention enables the production of ethanol-containing gasoline entirely meeting the requirements of the standards in force, firstly for the level of the DVPE and also for the other parameters. At the same time this gasoline secures a decrease of toxic emissions in the exhaust and lower fuel consumption in comparison to the mixture RFM 3 of source US A95 summer gasoline with 10% of ethanol.
- Formulation 3-10 had the following specific properties: density at 15°C, according to ASTM D4052 774.9kg /m3; initial boiling point, according to ASTM D 86 36.1°C; vaporisable portion - 70°C 33.6 % by volume; vaporisable portion - 100°C 50.8 % by volume; vaporisable portion - 150°C 86.1 % by volume; vaporisable portion - 190°C 97.0 % by volume; final boiling point 204.8°C; evaporation residue 1.5 % by volume; loss by evaporation 1.5 % by volume; oxygen content, according to ASTM D4815 3.37% w/w; acidity, according to ASTM D1613 weight% HAc 0.007; pH, according to ASTM D1287 7.58; sulfur content, according to ASTM D 5453 47mg/kg; gum content, according to ASTM D381 2.8mg/ 100ml; water content, according to ASTM D6304 0.02% w/w; aromatics, according
- the motor fuel Formulation 3-10 was tested on a 1987 Volvo 240 DL with a B230F, 4-cylinder, 2.32 litre engine (No. LG4F20-87) in accordance with test method EU 2000 NEDC EC 98/69 as above and gave the following results in comparison (+) or (-)% with the results for the source US A95 summer gasoline: CO -15.1% HC -5.6%; NOx +0.5%; CO2 unchanged; NMHC -4.5%; Fuel consumption, Fc, 1/100km unchanged.
- Figure 2 shows the behaviour of the dry vapour pressure equivalent (DVPE) as a function of the ethanol content in the mixtures of US summer A92 gasoline and the additive mixture 4 comprising 35 % by volume of ethanol, 1 % by volume of isoamyl alcohol, 0.2 % by volume of isobutanol, 43.8 % by volume of naphtha boiling at temperatures between 100-170°C, and 20% of isopropyl toluene.
- Figure 2 demonstrates that employment of this additive mixture in formulation of ethanol-containing gasoline enables the reduction of more than 100% of the excess vapour pressure induced by the presence of ethanol.
- the additive mixture comprising ethanol, the oxygen-containing compound other than ethanol, and C 6 -C 12 hydrocarbons in the proportion and composition of the present invention, can be used as an independent motor fuel for the engines adopted for operation on ethanol.
- Example 4 demonstrates the possibility of reducing the dry vapour pressure equivalent of the ethanol-containing motor fuel for the cases when the hydrocarbon base of the fuel is a non-standard gasoline with a dry vapour pressure equivalent according to ASTM D-5191 at a level of 110 kPa (about 16 psi).
- the hydrocarbon component (HCC) for the motor fuel compositions was prepared by mixing about 85 % by volume of winter A92, A95 or A98 gasoline with about 15 % by volume of gas condensate hydrocarbon liquid (GC).
- hydrocarbon component (HCC) for the fuel formulations 4-1 to 4-10 of this motor fuel composition, about 85 % by volume of winter A92, A95 or A98 gasoline was first mixed with the gas condensate hydrocarbon liquid (GC). The obtained hydrocarbon component (HCC) was then allowed to stand for 24 hours. The resulting gasoline contained aliphatic and alicyclic C 3 -C 12 hydrocarbons, including saturated and unsaturated ones.
- Fig. 1 demonstrates the behaviour of the DVPE of the ethanol-containing motor fuel based on winter A98 gasoline and gas condensate.
- the fuel 4-3 contained winter A92 gasoline, gas condensate (GC), ethanol, the oxygen-containing additive and C 6 -C 12 hydrocarbons and had the following properties for the various compositions:
- A92 : GC : Ethanol : Tert-butylethyl ether : Naphtha 68 : 12 : 9.5 : 0.5 : 10 % by volume
- the boiling temperature for the naphtha is 100-200°C.
- the fuel compositions below demonstrate that the invention enables the reduction of the excess DVPE of the non-standard gasoline to the level of the corresponding standrd gasoline.
- the DVPE for the standard A92 winter gasoline is 90 kPa.
- A92 : GC : Ethanol : Isoamyl alcohol : Naphtha : Alkylate 55 : 10 : 9.5 : 0.5 : 12.5 : 12.5 % by volume
- the boiling temperature for the naphtha is 100-200°C.
- the boiling temperature for the alkylate is 100-130°C.
- compositions demonstrate the possibility of adjusting the dry vapour pressure equivalent (DVPE) of the ethanol-containing fuel mixtures based on about 85 % by volume of winter A98 gasoline and about 15 % by volume of gas condensate.
- DVPE dry vapour pressure equivalent
- the motor fuel compositions below demonstrate that the invention enables the reduction of the excess DVPE of non-standard gasoline to the level of DVPE of the corresponding standard gasoline.
- the DVPE for the standard winter A98 gasoline is 90.0 kPa.
- A92 : GC : Ethanol: Isoamyl alcohol: Naphtha : Alkylate 55 : 10 : 9.5 : 0.5 : 12.5 : 12.5 % by volume
- the boiling temperature for the naphtha is 100-200°C.
- the boiling temperature for the alkylate is 100-130°C.
- A92 : GC : Ethanol : Isobutanol : Naphtha : Isopropyltoluene 55 : 10 : 5 : 5 : 20 : 5 % by volume
- the boiling temperature for the naphtha is 100-200°C.
- compositions demonstrate the possibility of adjusting the dry vapour pressure equivalent (DVPE) of the ethanol-containing fuel mixtures based on about 85 % by volume of winter A95 gasoline and about 15 % by volume of gas condensate.
- DVPE dry vapour pressure equivalent
- HCC hydrocarbon component
- GC gas condensate
- the reference fuel mixture (RFM4) comprising 80.75% of winter A95 gasoline, 14.25% of gas condensate (GC) and 5% of ethanol was tested as described above and gave the following results in comparison (+) or (-)% with the results for the gasoline comprising 85 % by volume of winter gasoline A95 and 15 % by volume of gas condensate (GC): CO -6.98% HC -7.3%; NOx +12.1%; CO2 + 1.1%; NMHC -5.3%; Fuel consumption, Fc, 1/100km +2.62%.
- the motor fuel compositions below demonstrate that the invention enables the reduction of the excess vapour pressure equivalent (DVPE) of the non-standard gasoline to the level of the corresponding standard gasoline.
- the DVPE of the standard winter gasoline A95 is 90.0 kPa.
- A95 : GC : Ethanol: Isoamyl alcohol : Isobutanol : Naphtha : Alkylate 55 : 10 : 9.2 : 0.6 : 0.2 : 12.5 : 12.5 % by volume
- the boiling temperature for the naphtha is 100-200°C.
- the boiling temperature for the alkylate is 100-130°C.
- the motor fuel 4-10 contained 55% by volume of A95 winter gasoline, 10% by volume of gas condensate (GC), 5% by volume of ethanol, 5% by volume of tert-butanol, 20% by volume of naphtha with boiling temperature of 100-200°C and 5% by volume of isopropyltoluene.
- Formulation 4-10 was tested to demonstrate how the invention enables the formulation of ethanol-containing gasoline entirely meeting requirements of the standards in force, firstly in respect of the dry vapour pressure equivalent limit, and also for the other parameters of the fuel, even when the source hydrocarbon component (HCC) has a DVPE considerably higher than the requirements of the standards.
- HCC source hydrocarbon component
- the formulation 4-10 had the following specific properties: density at 15°C, according to ASTM D4052 698.6 kg/m3; initial boiling point, according to ASTM D 86 20.5°C; vaporisable portion - 70°C 47.0 % by volume; vaporisable portion - 100°C 65.2 % by volume; vaporisable portion - 150°C 92.4 % by volume; vaporisable portion - 180°C 97.3 % by volume; final boiling point 189.9°C; evaporation residue 0.5 % by volume; loss by evaporation 1.1 % by volume; oxygen content, according to ASTM D4815 3.2% w/w; acidity, according to ASTM D1613 weight% HAc 0.001; pH, according to ASTM D 1287 7.0; sulfur content, according to ASTM D 5453 18 mg/kg; gum content,
- the motor fuel Formulation 4-10 was tested as above and gave the following results in comparison (+) or (-)% with the results for the motor fuel comprising 85 % by volume of winter A95 gasoline and 15 % by volume of gas condensate: CO -14.0% HC -8.6%; NOx unchanged; CO 2 + 1.0%; NMHC -6.7%; Fuel consumption, Fc, 1/100km +2.0%.
- HCC hydrocarbon component
- GC gas condensate
- inventive fuel formulations demonstrated the possibility of adjusting the vapour pressure of the ethanol-ccontaining motor fuels for the standard internal combustion spark ignition engines based on non-standard gasolines having a high vapour pressure.
- Example 5 demonstrates the possibility of reducing the dry vapour pressure equivalent of the ethanol-containing motor fuel for the cases when the hydrocarbon base base of the fuel is a reformulated gasoline with dry vapour pressure equivalent according to ASTM D-5191 at a level of 27.5 kPa (about 4 psi).
- the hydrocarbon component (HCC) for the motor fuel compositions was prepared by mixing about 85 % by volume of winter A92, A95 or A98 gasoline with about 15 % by volume of gas condensate hydrocarbon liquid (GC).
- the source gasolines comprised aliphatic and alicyclic C 6 -C 12 hydrocarbons, including saturated and unsaturated.
- Fig. 1 demonstrates the behaviour of the DVPE of the ethanol-containing motor fuel based on reformulated gasoline A92 and Petroleum benzine. Similar behaviour was observed for the ethanol-containing motor fuel based on reformulated A95 and A98 gasoline, and Petroleum benzine.
- the fuel 5-3 contained reformulated A92 gasoline, Petroleum benzine (PB), ethanol, the oxygen-containing additives and also C 8 -C 12 hydrocarbons and had the following properties for the various compositions:
- the boiling temperature for the naphtha is 140-200°C.
- the fuel 5-6 contained A98 reformulated gasoline, Petroleum benzine (PB), ethanol, the oxygen-containing additives, and C 8 -C 12 hydrocarbons (d) and had the following properties for the various compositions:
- the boiling temperature for the naphtha is 140-200°C.
- HCC hydrocarbon component
- PB Petroleum benzine
- the reference fuel mixture (RFM5) comprising 72 % by volume of reformulated A95 gasoline, 18 % by volume of Petroleum benzine (PB) and 10 % by volume of ethanol was tested on a 1987 Volvo 240 DL with a B230F, 4-cylinder, 2.32 litre engine (No.
- LG4F20-87 in accordance with test method EU 2000 NEDC EC 98/69 as above and gave the following results in comparison (+) or (-)% with the results for the gasoline comprising 80 % by volume of reformulated gasoline A95 and 20 % by volume of Petroleum benzine (GC): CO -4.8% HC -1.3%; NOx +26.3%; CO2 +4.4%; NMHC -0.6%; Fuel consumption, Fc, 1/100km +5.7%.
- GC Petroleum benzine
- the motor fuel 5-10 contained 60% by volume of A95 reformulated gasoline, 15% by volume of Petroleum benzine (PB), 10% by volume of ethanol, 5% by volume of 2,5-Dimethyltetrahydrofuran and 10% by volume of isopropyltoluene.
- Formulation 5-10 was tested to demonstrate how the invention enables the formulation of ethanol-containing gasoline with a low vapour pressure, wherein the presence in the motor fuel composition of ethanol does not induce an increase of dry vapour pressure equivalent in comparison to the source hydrocarbon component (HCC).
- HCC source hydrocarbon component
- this gasoline secures a decrease of toxic emissions in the exhaust and a decrease of the fuel consumption in comparison with the above mixture RFM 5.
- the formulation 5-10 had the following specific properties: density at 15°C, according to ASTM D4052 764.6 kg/m3; initial boiling point, according to ASTM D 86 48.9°C; vaporisable portion - 70°C 25.3 % by volume; vaporisable portion - 100°C 50.8 % by volume; vaporisable portion - 150°C 76.5 % by volume; vaporisable portion - 190°C 95.6 % by volume; final boiling point 204.5°C; evaporation residue 1.4 % by volume; loss by evaporation 0.5 % by volume; oxygen content, according to ASTM D4815 4.6% w/w; acidity, according to ASTM D 1613 weight% HAc 0.08; pH, according to ASTM D1287 7.5; sulfur content, according to ASTM D 5453 39 mg/kg; gum content, according to ASTM D381 1.5 mg/100ml; water content, according to ASTM D6304 0.1% w/w; aromatics, according to SS
- the motor fuel Formulation 5-10 was tested as described previously and gave the following results in comparison (+) or (-)% with the results for the motor fuel comprising 80 % by volume of reformulated A95 gasoline and 20 % by volume of Petroleum benzine: CO -12.3% HC -6.2%; NOx unchanged; CO 2 +2.6%; NMHC -6.4%; Fuel consumption, Fc, 1/ 100km +3.7%
- the invention demonstrated the possibility of adjusting the vapour pressure of the ethanol-containing motor fuels for the standard internal combustion spark ignition engines based on non-standard gasolines having a low vapour pressure.
- Figure 2 shows the behaviour of the dry vapour pressure equivalent (DVPE) when mixing the hydrocarbon component (HCC), comprising 80 % by volume of reformulated A92 gasoline and 20 % by volume of Petroleum benzine, with the oxygen-containing additive mixture 5, comprising 40 % by volume of ethanol, 20 % by volume of 3,3,5-trimethylcyclohexanone, and 20 % by volume of naphtha with boiling temperature 130-170°C and 20 % by volume of tert-butyltoluene.
- HCC hydrocarbon component
- HCC hydrocarbon component
- gasolines have a vapour pressure equivalent (DVPE) not higher than the DVPE of the source hydrocarbon component (HCC).
- DVPE vapour pressure equivalent
- HCC source hydrocarbon component
Landscapes
- Chemical & Material Sciences (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- General Chemical & Material Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Health & Medical Sciences (AREA)
- Emergency Medicine (AREA)
- Combustion & Propulsion (AREA)
- Liquid Carbonaceous Fuels (AREA)
- Ignition Installations For Internal Combustion Engines (AREA)
- Air-Conditioning For Vehicles (AREA)
- Output Control And Ontrol Of Special Type Engine (AREA)
- Organic Low-Molecular-Weight Compounds And Preparation Thereof (AREA)
- Fuel-Injection Apparatus (AREA)
- Combustion Methods Of Internal-Combustion Engines (AREA)
Abstract
Description
- density at 15°C and at normal atmospheric pressure of not less than 690 kg/m3;
- oxygen content, based on the amount of oxygen-containing components, of not more than 7% w/w of the motor fuel composition;
- anti-knock index (octane number) of not lower than the anti-knock index (octane number) of the source hydrocarbon component and preferably for 0.5(RON+MON) of not less than 80;
- dry vapour pressure equivalent (DVPE) essentially the same as the DVPE of the source hydrocarbon component and preferably from 20 kPa to 120 kPa;
- acid content of not more than 0.1% by weight HAc;
- pH from 5 to 9;
- aromatic hydrocarbons content of not more than 40 % by volume, including benzene, and for benzene alone, not more than 1 % by volume;
- limits of evaporation of the liquid at normal atmospheric pressure in % of source volume of the motor fuel composition:
| initial boiling point, | 20°C; |
| volume (at 70°C, min) of the liquid | 25% by |
| evaporated | volume; |
| volume (at 100°C, min) of the liquid | 50% by |
| evaporated | volume; |
| volume (at 150°C, min) of the liquid | 75% by |
| evaporated | volume; |
| volume (at 190°C, min) of the liquid | 95% by |
| evaporated | volume; |
| residue of distillation, max. | 2% by volume; |
| final boiling point, max. | 205°C; |
- sulfur content of not more than 50mg/kg;
- resins content of not more than 2mg/ 100ml.
- fuel grade ethanol purchased in Sweden at Sekab and in the USA from ADM Corp. and Williams;
- oxygen-containing compounds, individual unsubstituted hydrocarbons and mixtures hereof purchased in Germany from Merck and in Russia from Lukoil.
- Naphtha, which is an oil straight run gasoline containing aliphatic and alicyclic saturated and unsaturated hydrocarbons. Alkylate, which is a hydrocarbon fraction consisting almost completely of isoparaffine hydrocarbons obtained in alkylation of isobutene by butanol. Alkylbenzene, which is a mixture of aromatic hydrocarbons obtained in benzene alkylation. Mostly, technical grade alkylbenzene comprises ethylbenzene, propylbenzene, isopropylbenzene, butylbenzene and others.
DVPE = 89,0 kPa
Anti-knock index 0.5(RON + MON)=87.7
A92 : Ethanol = 95 : 5 % by volume
DVPE = 94.4 kPa
0.5(RON + MON) = 89.1
A92 : Ethanol = 90 : 10 % by volume
DVPE = 94.0 kPa
0.5(RON + MON) = 90.2
A92 : Ethanol : Isoamyl alcohol : Alkylate = 79 : 9 : 2 : 10 % by volume
The boiling temperature of the alkylate is 100-130°C
DVPE = 88.5 kPa
0.5(RON + MON) = 90.25
A92 : Ethanol : Isobutyl acetate : Naphtha = 80 : 5 : 5 : 10 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 88.7 kPa
0.5(RON + MON) = 88.6
A92 : Ethanol: Tert-butanol: Naphtha = 81 : 5 : 5 : 9 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 87.5 kPa
0.5(RON + MON) = 89.6
A92 : Ethanol: Isoamyl alcohol: Benzene : Ethylbenzene : Diethyl benzene = 82.5 : 9.5 : 0.5 : 0.5 : 3 : 4 % by volume
DVPE = 90 kPa
0.5(RON + MON) = 91.0
A92 : Ethanol: Isobutyl acetate : Toluene = 82.5 : 9.5 : 0.5 : 7.5 % by volume DVPE = 90 kPa
0.5(RON + MON) = 90.8
A92 : Ethanol : Isobutanol : Isoamyl alcohol : m-Xylene = 82.5 : 9.2 : 0.2 : 0.6 : 7.5 % by volume
DVPE = 90 kPa
0.5(RON + MON) = 90.9
DVPE = 89,5 kPa
Anti-knock index 0.5(RON + MON)=92.35
A98 : Ethanol = 95 : 5 % by volume
DVPE = 95.0 kPa
0.5(RON + MON) = 92.85
A98 : Ethanol = 90 : 10 % by volume
DVPE = 94.5 kPa
0.5(RON + MON) = 93.1
A98 : Ethanol: Isoamyl alcohol : Isooctane = 80 : 5 : 5 : 10 % by volume
DVPE = 82.0 kPa
0.5(RON + MON) = 93.2
A98 : Ethanol : Isoamyl alcohol: m-Isopropyl toluene = 78.2 : 6.1 : 6.1 : 9.6 % by volume
DVPE = 81.0 kPa
0.5(RON + MON) = 93.8
A98 : Ethanol: Isobutanol : Naphtha = 80 : 5 : 5 : 10 % by volume
The boiling point of the naphtha is 100-200°C.
DVPE = 82.5 kPa
0.5(RON + MON) = 92.35
A98 : Ethanol : Isobutanol : Naphtha : m-Isopropyl toluene = 80 : 5 : 5 : 5 : 5 % by volume
The boiling point of the naphtha is 100-200°C.
DVPE = 82.0 kPa
0.5(RON + MON) = 93.25
A98 : Ethanol : Tert-butyl acetate : Naphtha = 83 : 5 : 5 : 7 % by volume
The boiling temperature of the naphtha is 100-200°C
DVPE = 82.1 kPa
0.5(RON + MON) = 92.5
A98 : Ethanol : Isoamyl alcohol : Isooctane = 85 : 5 : 5 : 5 % by volume
DVPE = 90.0 kPa
0.5(RON + MON) = 93.3
A98 : Ethanol: Isobutanol : Naphtha = 85 : 5 : 5 : 5 % by volume
The boiling temperature of the naphtha is 100-200°C
DVPE = 90.0 kPa
0.5(RON + MON) = 93.0
A98 : Ethanol: Isobutanol : Isopropyl xylene = 85 : 9.5 : 0.5 : 5 % by volume
DVPE = 90 kPa
0.5(RON + MON) = 93.1
A98 : Ethanol : Isobutanol : Isooctane : Naphtha = 60 : 9.5 : 0.5 : 15 : 15 % by volume
The boiling point of the naphtha is 100-200°C.
DVPE = 70 kPa
0.5(RON + MON) = 92.85
A98 : Ethanol : Isobutanol : Alkylate : Naphtha = 60 : 9.5 : 0.5 : 15 : 15 % by volume
The boiling point of the naphtha is 100-200°C.
The boiling point of the alkylate is 100-130°C.
DVPE = 70 kPa
0.5(RON + MON) = 92.6
A98 : Ethanol: Tert-butyl acetate : Naphtha = 60 : 9 : 3 : 28 % by volume
The boiling point of the naphtha is 100-200°C.
DVPE = 70 kPa
0.5(RON + MON) = 91.4
DVPE = 89.5 kPa
Anti-knock index 0.5(RON + MON)= 90.1
| CO (carbon monoxide) | 2.13g/km; |
| HC (hydrocarbons) | 0.280g/km; |
| NOx (nitrogen oxides) | 0.265g/km; |
| CO2 (carbon dioxide) | 227.0g/km; |
| NMHC | 0.276g/km; |
| Fuel consumption, Fc 1/100km | 9.84 |
A95 : Ethanol = 95 : 5 % by volume
DVPE = 94.9 kPa
0.5(RON + MON) = 91.6
A95 : Ethanol = 90 : 10 % by volume (referred to as RFM 1 below)
DVPE = 94.5 kPa
0.5(RON + MON) = 92.4
| CO | -15.0%; |
| HC | -7.3%; |
| NOx | + 15.5%; |
| CO2 | +2.4%; |
| NMHC* | -0.5%; |
| Fuel consumption, Fc, 1/100km | +4.7% |
| "-" represents a reduction in emission, while "+" represents an increase in emission. |
A95 : Ethanol: Isoamyl alcohol : Alkylate = 83.7 : 5 : 2 : 9.3 % by volume
The boiling temperature of the alkylate is 100-130°C
DVPE = 88.0 kPa
0.5(RON + MON) = 91.65
A95 : Ethanol: Isoamyl alcohol: Naphtha = 83.7 : 5 : 2 : 9.3% by vol.
The boiling temperature of the naphtha is 100-200°C
DVPE = 88.5 kPa
0.5(RON + MON) = 90.8
A95 : Ethanol : Isobutyl acetate : Alkylate =81:5:5:9% by volume
The boiling temperature of the alkylate is 100-130°C
DVPE = 87.0 kPa
0.5(RON + MON) = 92.0
A95 : Ethanol: Isobutyl acetate : Naphtha = 81 : 5 : 5 : 9 % by volume
The boiling temperature of the naphtha is 100-200°C
DVPE = 87.5 kPa
0.5(RON + MON) = 91.1
A95 : Ethanol: Isoamyl alcohol: Xylene = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 90.0 kPa
0.5(RON + MON) = 92.1
A95 : Ethanol : Isobutanol : Isoamyl alcohol : Naphtha = 80 : 9.2 : 0.2 : 0.6 : 10% by volume
The boiling temperature of the naphtha is 100-200°C
DVPE = 90.0 kPa
0.5(RON + MON) = 91.0
A95 : Ethanol : Isobutanol : Isoamyl alcohol: Naphtha : Alkylate = 80 : 9.2 : 0.2 : 0.6 : 5 : 5 % by volume
The boiling temperature of the naphtha is 100-200°C.
The boiling point of the alkylate is 100-130°C.
DVPE = 90.0 kPa
0.5(RON + MON) = 91.6
A95 : Ethanol : Isobutanol : Isoamyl alcohol : Naphtha : Isooctane = 60 : 9.2 : 0.2 : 0.6 : 15 : 15 % by volume
The boiling temperature of the naphtha is 100-200°C.
DVPE = 70.0 kPa
0.5(RON + MON) = 91.8
A95 : Ethanol: Tert-butyl acetate : Naphtha = 60 : 9 : 1 : 30 % by volume
The boiling temperature of the naphtha is 100-200°C.
DVPE = 70.0 kPa
0.5(RON + MON) = 90.4
| density at 15°C, according to ASTM D 4052 | 749.2 kg /m3; |
| initial boiling point, according to ASTM D 86 | 29°C; |
| vaporizable portion - 70°C | 47.6 % by volume; |
| vaporizable portion - 100°C | 55.6 % by volume; |
| vaporizable portion - 150°C | 84.2 % by volume; |
| vaporizable portion - 180°C | 97.5 % by volume; |
| final boiling point | 194.9°C; |
| evaporation residue | 1.3 % by volume; |
| loss by evaporation | 1.6 % by volume; |
| oxygen content, according to ASTM D4815 acidity, according to ASTM D 1613 | 3.7%w/w; |
| weight% HAc | 0.004; |
| pH, according to ASTM D1287 | 6.6; |
| sulfur content, according to ASTM D 5453 | 18mg/kg; |
| gum content, according to ASTM D381 | 1 mg/100ml; |
| water content, according to ASTM D6304 aromatics, according to SS 155120, | 0.03% w/w; |
| including benzene | 30.2 % by volume; |
| benzene alone, according to EN 238 | 0.7 % by volume; |
| DVPE, according to ASTM D 5191 | 89.0kPa; |
| anti-knock index 0.5(RON+MON), according to ASTM D 2699-86 and ASTM D 2700-86 | 92.6 |
| CO | -21%; |
| HC | -9%; |
| NOx | + 12.8%; |
| CO2 | +2.38%; |
| NMHC | -6.4%; |
| Fuel consumption, Fc 1/100km | +3.2% |
A92 : Ethanol: Isobutanol : Isopropyltoluene = 85 : 6 : 1.5 : 7.5 % by volume
DVPE = 84.9 kPa
0.5(RON + MON) = 93.9
A95 : Ethanol: Isobutanol : Isopropyltoluene = 80 : 8 : 2 : 10 % by volume
DVPE = 84.0 kPa
0.5(RON + MON) = 94.1
A98 : Ethanol: Isobutanol : Isopropyltoluene = 86 : 5.6 : 1.4 : 7 % by volume
DVPE = 85.5 kPa
0.5(RON + MON) = 93.8
DVPE = 70,0 kPa
Anti-knock index 0.5(RON + MON)=87.5
A92 : Ethanol = 95 : 5 % by volume
DVPE = 77.0 kPa
0.5(RON + MON) = 89.3
A92 : Ethanol = 90 : 10 % by volume
DVPE = 76.5 kPa
0.5(RON + MON) = 90.5
A92 : Ethanol : Methylethyl ketone : Isooctane = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 69.0 kPa
0.5(RON + MON) = 91.0
A92 : Ethanol: Isobutanol : Isooctane = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 69.0 kPa
0.5(RON + MON) = 91.1
A92 : Ethanol: Isobutanol : Isononane = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 68.8 kPa
0.5(RON + MON) = 91.0
A92 : Ethanol: Isobutanol : Isodecane = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 68.5 kPa
0.5(RON + MON) = 90.8
A92 : Ethanol: Isobutanol : Isooctene = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 68.9 kPa
0.5(RON + MON) = 91.2
A92 : Ethanol: Isobutanol : Toluene = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 68.5 kPa
0.5(RON + MON) = 91.4
A92 : Ethanol: Isobutanol : Naphtha = 80 : 9.5 : 0.5 : 10 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 67.5 kPa
0.5(RON + MON) = 90.4
A92 : Ethanol : Isobutanol : Naphtha : Toluene = 80 : 9.5 : 0.5 : 5 : 5 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 67.5 kPa
0.5(RON + MON) = 90.9
A92 : Ethanol : Isobutanol : Naphtha : Isopropyltoluene = 80 : 9.5 : 0.5 : 5 : 5 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 67.5 kPa
0.5(RON + MON) = 91.2
A92 : Ethanol: Isobutanol: Isodecane = 82.5 : 9.5 : 0.5 : 7.5 % by volume
DVPE = 70.0 kPa
0.5(RON + MON) = 90.85
A92 : Ethanol : Isobutanol : Tert-butylbenzene = 82.5 : 9.5 : 0.5 : 7.5 % by volume
DVPE = 70.0 kPa
0.5(RON + MON) = 91.5
A92 : Ethanol : Isobutanol : Isoamyl alcohol : Naphtha : Tert-butyltoluene = 82.5 : 9.2 : 0.2 : 0.6 : 5 : 2.5 % by volume
DVPE = 70.0 kPa
0.5(RON + MON) = 91.1
DVPE = 69,5 kPa
Anti-knock index 0.5(RON + MON)=92.5
A98 : Ethanol = 95 : 5 % by volume
DVPE = 76.5 kPa
0.5(RON + MON) = 93.3
A98 : Ethanol = 90 : 10 % by volume
DVPE = 76.0 kPa
0.5(RON + MON) = 93.7
A98: Ethanol : Isobutanol : Isooctane = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 69.0 kPa
0.5(RON + MON) = 93.7
A98 : Ethanol : Isopropanol : Alkylbenzene = 80 : 5 : 5 : 10 % by volume
DVPE = 68.5 kPa
0.5(RON + MON) = 94.0
A98 : Ethanol: Isobutanol: Isooctane = 81.5 : 9.5 : 0.5 : 8.5 % by volume
DVPE = 70.0 kPa
0.5(RON + MON) = 93.5
A98 : Ethanol : Tert-butanol : Limonene = 86 : 7 : 4 : 4 % by volume
DVPE = 70.0 kPa
0.5(RON + MON) = 93.6
DVPE = 68,5 kPa
Anti-knock index 0.5(RON + MON) = 89.8
| CO (carbon monoxide) | 2.198g/km; |
| HC (hydrocarbons) | 0.245g/km; |
| NOx (nitrogen oxides) | 0.252g/km; |
| CO2 (carbon dioxide) | 230.0g/km; |
| NMHC | 0.238g/km; |
| Fuel consumption, Fc 1/100km | 9.95 |
A95 : Ethanol = 95% : 5 % by volume
DVPE = 75.5 kPa
0.5(RON + MON) = 90.9
A95 : Ethanol = 90% : 10 % by volume (also referred to as RFM2 below)
DVPE = 75.0 kPa
0.5(RON + MON) = 92.25
| CO | -9.1%; |
| HC | -4.5%; |
| NOx | +7.3%; |
| CO2 | +4.0%; |
| NMHC* | -4.4%; |
| Fuel consumption, F, 1/100km | +3.6% |
| "-" represents a reduction in emission, while "+" represents an increase in emission |
A95 : Ethanol : Tert-pentanol: Alkylbenzene = 80 : 7 : 4 : 9 % by volume
DVPE = 67.5 kPa
0.5(RON + MON) = 93.6
A95 : Ethanol: Tert-butanol: Alkylbenzene = 80 : 7 : 4 : 9 % by volume
DVPE = 68.0 kPa
0.5(RON + MON) = 93.8
A95 : Ethanol: Propanol: Xylene = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 68.0 kPa
0.5(RON + MON) = 93.1
A95 : Ethanol: Diethylketone : Xylene = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 68.0 kPa
0.5(RON + MON) = 93.2
A95 : Ethanol : Isobutanol : Naphtha : Isopropyltoluene = 80 : 9.5 : 0.5 : 5 : 5 % by volume
The boiling temperature for the naphtha is 100-170°C
DVPE = 68.0 kPa
0.5(RON + MON) = 92.4
A95 : Ethanol : Isobutanol : Naphtha : Alkylate = 80 : 9.5 : 0.5 : 5 : 5 % by volume
The boiling temperature for the naphtha is 100-170°C
The boiling temperature for the alkylate is 100-130°C
DVPE = 68.5 kPa
0.5(RON + MON) = 92.2
A95 : Ethanol : Isobutanol : Isoamyl alcohol : Xylene = 82.5 : 9.2 : 0.2 : 0.6 : 7.5 % by volume
DVPE = 70.0 kPa
0.5(RON + MON) = 93.0
A95 : Ethanol : Isobutanol : Isoamyl alcohol : Cyclooctadiene = 82.5 : 9.2 : 0.2 : 0.6 : 7.5 % by volume
DVPE = 70.0 kPa
0.5(RON + MON) = 92.1
| density at 15°C, according to ASTM D4052 | 754.1kg/m3; |
| initial boiling point, according to ASTM D 86 | 26.6°C; |
| vaporisable portion - 70°C | 45.2 % by volume; |
| vaporisable portion - 100°C | 56.4 % by volume; |
| vaporisable portion - 150°C | 88.8 % by volume; |
| vaporisable portion - 180°C | 97.6 % by volume; |
| final boiling point | 186.3°C; |
| evaporation residue | 1.6 % by volume; |
| loss by evaporation | 0.1 % by volume; |
| oxygen content, according to ASTM D4815 | 3.56% w/w; |
| acidity, according to ASTM D1613 weight% HAc | 0.007; |
| pH, according to ASTM D 1287 | 8.9; |
| sulfur content, according to ASTM D 5453 | 16mg/kg; |
| gum content, according to ASTM D381 | <1mg/100ml; |
| water content, according to ASTM D6304 | 0.12% w/w; |
| aromatics, according to SS 155120, including benzene | 30.3 % by volume; |
| benzene alone, according to EN 238 | 0.8 % by volume; |
| DVPE, according to ASTM D 5191 | 68.5kPa; |
| anti-knock index 0.5(RON+MON), according to ASTM D 2699-86 and ASTM D 2700-86 | 92.7 |
| CO | -0.18% |
| HC | -8.5%; |
| NOx | +5.3%; |
| CO2 | +2.8%; |
| NMHC | -9%; |
| Fuel consumption, Fc, 1/100km | +3.1% |
DVPE = 47,8 kPa
Anti-knock index 0.5(RON + MON)=87.7
A92 : Ethanol = 95 : 5 % by volume
DVPE = 55.9 kPa
0.5(RON + MON) = 89.0
A92 : Ethanol = 90 : 10 % by volume
DVPE = 55.4 kPa
0.5(RON + MON) = 90.1
A92 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Naphtha = 75 : 9.2 : 0.3 : 0.1 : 15.4 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 47.8 kPa
0.5(RON + MON) = 89.5
A92 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : m-Isopropyltoluene = 75 : 9.2 : 0.3 : 0.1 : 15.4 % by volume
DVPE = 47.0 kPa
0.5(RON + MON) = 90.5
A92 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Isooctane = 75 : 9.2 : 0.3 : 0.1 : 15.4 % by volume
DVPE = 47.8 kPa
0.5(RON + MON) = 90.3
A92 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Naphtha = 76 : 9.2 : 0.3 : 0.1 : 14.4 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 48.2 kPa
0.5(RON + MON) = 89.6
A92 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Naphtha : Isooctane = 76 : 9.2 : 0.3 : 0.1 : 10.4 : 4 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 48.2 kPa
0.5(RON + MON) = 89.8
A92 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Naphtha : m-Isopropyl toluene = 77 : 9.2 : 0.3 : 0.1 : 10.4 : 3 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 48.2 kPa
0.5(RON + MON) = 89.9
DVPE = 48.2 kPa
Anti-knock index 0.5(RON + MON)= 92.2
A98 : Ethanol = 95 : 5 % by volume
DVPE = 56.3 kPa
0.5(RON + MON) = 93.0
A98 : Ethanol = 90 : 10 % by volume
DVPE = 55.8 kPa
0.5(RON + MON) = 93.6
A98 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Naphtha = 75 : 9.2 : 0.3 : 0.1 : 15.4 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 48.2 kPa
0.5(RON + MON) = 93.3
A98 : Ethanol: Isoamyl alcohol: Isobutyl alcohol: Isooctane = 75 : 9.2 : 0.3 : 0.1 : 15.4 % by volume
DVPE = 48.2 kPa
0.5(RON + MON) = 93.9
A98 : Ethanol: Isoamyl alcohol: Isobutyl alcohol: m-Isopropyltoluene = 75.5 : 9.2 : 0.3 : 0.1 : 14.9 % by volume
DVPE = 47.5 kPa
0.5(RON + MON) = 94.4
A98 : Ethanol: Isoamyl alcohol: Isobutyl alcohol: Naphtha : Isooctane = 75 : 9.2 : 0.3 : 0.1 : 8.4 : 7 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 48.2 kPa
0.5(RON + MON) = 93.6
A98 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Naphtha : m-Isopropyl toluene = 75 : 9.2 : 0.3 : 0.1 : 10.4 : 5 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 48.0 kPa
0.5(RON + MON) = 93.7
A98 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Naphtha : Alkylate = 75 : 9.2 : 0.3 : 0.1 : 7.9 : 7.5 % by volume
The boiling temperature for the naphtha is 100-200°C.
The boiling temperature for the alkylate is 100-130°C.
DVPE = 48.2 kPa
0.5(RON + MON) = 93.6
DVPE = 47.0 kPa
Anti-knock index 0.5(RON + MON) = 90.9
| CO (carbon monoxide) | 2.406g/km; |
| HC (hydrocarbons) | 0.356g/km; |
| NOx (nitrogen oxides) | 0.278g/km; |
| CO2 (carbon dioxide) | 232.6g/km; |
| NMHC | 0.258g/km; |
| Fuel consumption, Fc 1/100km | 9.93 |
A95 : Ethanol = 95 : 5 % by volume
DVPE = 55.3 kPa
0.5(RON + MON) = 91.5
A95 : Ethanol = 90 : 10 % by volume
DVPE = 54.8 kPa
0.5(RON + MON) = 92.0
| CO | -12.5%; |
| HC | -4.8%; |
| NOx | +2.3%; |
| CO2 | +3.7%; |
| NMHC* | -4.0%; |
| Fuel consumption, F, 1/100km | +3.1% |
| "-" represents a reduction in emission, while "+" represents an increase in emission. |
A95 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Naphtha = 75 : 9.2 : 0.3 : 0.1 : 15.4 % by volume
The boiling temperature for the naphtha is 100-200°C
DVPE = 47.0 kPa
0.5(RON + MON) = 91.6
A95 : Ethanol: Isoamyl alcohol: Isobutyl alcohol: Isooctane = 75 : 9.2 : 0.3 : 0.1 : 15.4 % by volume
DVPE = 47.0 kPa
0.5(RON + MON) = 92.2
A95 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : m-Isopropyltoluene = 75 : 9.2 : 0.3 : 0.1 : 15.4 % by volume
DVPE = 46.8 kPa
0.5(RON + MON) = 93.0
A95 : Ethanol : Tetrahydrofurfuryl alcohol: Cyclooctatetraene = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 46.6 kPa
0.5(RON + MON) = 92.5
A95 : Ethanol : 4-Methyl-4-oxytetrahydropyrane : Allocymene = 80 : 9.5 : 0.5 : 10 % by volume
DVPE = 46.7 kPa
0.5(RON + MON) = 92.1
A95 : Ethanol : Isoamyl alcohol: Isobutyl alcohol : Naphtha = 76.5 : 9.2 : 0.3 : 0.1 : 7 : 6,9 % by volume
The boiling temperature for the naphtha is 100-200°C.
DVPE = 48.2 kPa
0.5(RON + MON) = 91.7
A95 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : Naphtha : Isooctane = 76.5 : 9.2 : 0.3 : 0.1 : 7 : 6.9 % by volume
The boiling temperature for the naphtha is 100-200°C.
DVPE = 48.2 kPa
0.5(RON + MON) = 92.2
A95 : Ethanol : Isoamyl alcohol : Isobutyl alcohol : m-Isopropyltoluene = 77 : 9.2 : 0.3 : 0.1 : 13.4 % by volume
DVPE = 48.2 kPa
0.5(RON + MON) = 92.9
| density at 15°C, according to ASTM D4052 | 774.9kg /m3; |
| initial boiling point, according to ASTM D 86 | 36.1°C; |
| vaporisable portion - 70°C | 33.6 % by volume; |
| vaporisable portion - 100°C | 50.8 % by volume; |
| vaporisable portion - 150°C | 86.1 % by volume; |
| vaporisable portion - 190°C | 97.0 % by volume; |
| final boiling point | 204.8°C; |
| evaporation residue | 1.5 % by volume; |
| loss by evaporation | 1.5 % by volume; |
| oxygen content, according to ASTM D4815 | 3.37% w/w; |
| acidity, according to ASTM D1613 weight% HAc | 0.007; |
| pH, according to ASTM D1287 | 7.58; |
| sulfur content, according to ASTM D 5453 | 47mg/kg; |
| gum content, according to ASTM D381 | 2.8mg/ 100ml; |
| water content, according to ASTM D6304 | 0.02% w/w; |
| aromatics, according to SS 155120, including benzene | 31.2 % by volume; |
| benzene alone, according to EN 238 | 0.7 % by volume; |
| DVPE, according to ASTM D 5191 | 48.0kPa; |
| anti-knock index 0.5(RON+MON), according to ASTM D 2699-86 and ASTM D 2700-86 | 92.2 |
| CO | -15.1% |
| HC | -5.6%; |
| NOx | +0.5%; |
| CO2 | unchanged; |
| NMHC | -4.5%; |
| Fuel consumption, Fc, 1/100km | unchanged. |
Figure 2 demonstrates that employment of this additive mixture in formulation of ethanol-containing gasoline enables the reduction of more than 100% of the excess vapour pressure induced by the presence of ethanol.
DVPE = 110.0 kPa
Anti-knock index 0.5(RON + MON)=87.9
A92 : GC : Ethanol= 80.75 : 14.25 : 5 % by volume
DVPE = 115.5 kPa
0.5(RON + MON) = 89.4
A92 : GC : Ethanol= 76.5 : 13.5 : 10 % by volume
DVPE = 115.0 kPa
0.5(RON + MON) = 90.6
A92 : GC : Ethanol : Isobutanol : Isopropylbenzene = 68 : 12 : 9.5 : 0.5 : 10 % by volume
DVPE = 108.5 kPa
0.5(RON + MON) = 91.7
A92 : GC : Ethanol : Tert-butylethyl ether : Naphtha = 68 : 12 : 9.5 : 0.5 : 10 % by volume
The boiling temperature for the naphtha is 100-200°C.
DVPE = 108.5 kPa
0.5(RON + MON) = 90.6
A92 : GC : Ethanol : Isoamylmethyl ether : Toluene = 68 : 12 : 9.5 : 0.5 : 10 % by volume
DVPE = 107.5 kPa
0.5(RON + MON) = 91.6
A92 : GC : Ethanol : Isoamyl alcohol : Naphtha : Alkylate = 55 : 10 : 9.5 : 0.5 : 12.5 : 12.5 % by volume
The boiling temperature for the naphtha is 100-200°C.
The boiling temperature for the alkylate is 100-130°C.
DVPE = 90.0 kPa
0.5(RON + MON) = 90.6
A92 : GC : Ethanol : Isoamyl alcohol : Naphtha : Ethylbenzene = 55 : 10 : 9.5 : 0.5 : 15 : 10 % by volume
The boiling temperature for the naphtha is 100-200°C.
DVPE = 89.8 kPa
0.5(RON + MON) = 90.9
A92 : GC : Ethanol : Isoamyl alcohol : Naphtha : Isopropyltoluene = 55 : 10 : 9.5 : 0.5 : 20 : 5 % by volume
The boiling temperature for the naphtha is 100-200°C.
DVPE = 90.0 kPa
0.5(RON + MON) = 90.6
DVPE = 109.8 kPa
Anti-knock index 0.5(RON + MON)=92.0
A98 : GC : Ethanol = 80.75 : 14.25 : 5 % by volume
DVPE = 115.3 kPa
0.5(RON + MON) = 93.1
A98 : GC : Ethanol = 76.5 : 13.5 : 10 % by volume
DVPE = 114.8 kPa
0.5(RON + MON) = 94.0
A98 : GC : Ethanol: Isoamyl alcohol : Isobutyl alcohol : Naphtha = 68 : 12 : 9.2 : 0.6 : 0.2 : 10 % by volume
The boiling temperature for the naphtha is 100-200°C.
DVPE = 107.4 kPa
0.5(RON + MON) = 93.8
A98 : GC : Ethanol: Ethylisobutyl ether : Myrzene = 72 : 13 : 9.5 : 0.5 : 5 % by volume
DVPE = 110.0 kPa
0.5(RON + MON) = 93.6
A98 : GC : Ethanol : Isobutanol : Isooctane = 68 : 12 : 5 : 5 : 10 % by volume
DVPE = 102.5 kPa
0.5(RON + MON) = 93.5
A92 : GC : Ethanol: Isoamyl alcohol: Naphtha : Alkylate = 55 : 10 : 9.5 : 0.5 : 12.5 : 12.5 % by volume
The boiling temperature for the naphtha is 100-200°C.
The boiling temperature for the alkylate is 100-130°C.
DVPE = 89.8 kPa
0.5(RON + MON) = 94.0
A92 : GC : Ethanol : Isoamyl alcohol: Naphtha : Isopropylbenzene = 55 : 10 : 9.5 : 0.5 : 15 : 10 % by volume
The boiling temperature for the naphtha is 100-200°C.
DVPE = 89.6 kPa
0.5(RON + MON) = 94.2
A92 : GC : Ethanol : Isobutanol : Naphtha : Isopropyltoluene = 55 : 10 : 5 : 5 : 20 : 5 % by volume
The boiling temperature for the naphtha is 100-200°C.
DVPE = 88.5 kPa
0.5(RON + MON) = 94.1
DVPE = 109.5 kPa
Anti-knock index 0.5(RON + MON)=90.2
| CO | 2.033 g/km; |
| HC | 0.279 g/km; |
| NOx | 0.279 g/km; |
| CO2 | 229.5 g/km; |
| NMHC | 0.255 g/km; |
| Fuel consumption, Fc, 1/ 100km | 9.89 |
A95 : GC : Ethanol = 80.75 : 14.25 : 5 % by volume
DVPE = 115.0 kPa
0.5(RON + MON) = 91.7
A95 : GC : Ethanol = 76.5 : 13.5 : 10 % by volume
DVPE = 114.5 kPa
0.5(RON + MON) = 92.5
| CO | -6.98% |
| HC | -7.3%; |
| NOx | +12.1%; |
| CO2 | + 1.1%; |
| NMHC | -5.3%; |
| Fuel consumption, Fc, 1/100km | +2.62%. |
A95 : GC : Ethanol: Isoamyl alcohol : Isobutyl alcohol : Naphtha = 68 : 12 : 9.2 : 0.6 : 0.2 : 10 % by volume
The boiling temperature for the naphtha is 100-200°C.
DVPE = 107.0 kPa
0.5(RON + MON) = 92.1
A95 : GC : Ethanol: Isobutanol : Cyclooctatetraene = 72 : 13 : 9.5 : 0.5 : 5 % by volume
DVPE = 108.5 kPa
0.5(RON + MON) = 92.6
A95 : GC : Ethanol: Isoamyl alcohol : Isobutanol : Naphtha : Alkylate = 55 : 10 : 9.2 : 0.6 : 0.2 : 12.5 : 12.5 % by volume
The boiling temperature for the naphtha is 100-200°C.
The boiling temperature for the alkylate is 100-130°C.
DVPE = 89.5 kPa
0.5(RON + MON) = 92.4
A95 : GC : Ethanol: Isoamyl alcohol : Naphtha : Tert-butylxylene = 55 : 10 : 9.5 : 0.5 : 20 : 5 % by volume
The boiling temperature for the naphtha is 100-200°C.
DVPE = 89.8 kPa
0.5(RON + MON) = 92.5
A95 : GC : Ethanol: Isobutanol : Naphtha : Isopropylbenzene = 55 : 10 : 5 : 5 : 20 : 5 % by volume
The boiling temperature for the naphtha is 100-200°C.
DVPE = 89.9 kPa
0.5(RON + MON) = 92.2
| density at 15°C, according to ASTM D4052 | 698.6 kg/m3; |
| initial boiling point, according to ASTM D 86 | 20.5°C; |
| vaporisable portion - 70°C | 47.0 % by volume; |
| vaporisable portion - 100°C | 65.2 % by volume; |
| vaporisable portion - 150°C | 92.4 % by volume; |
| vaporisable portion - 180°C | 97.3 % by volume; |
| final boiling point | 189.9°C; |
| evaporation residue | 0.5 % by volume; |
| loss by evaporation | 1.1 % by volume; |
| oxygen content, according to ASTM D4815 | 3.2% w/w; |
| acidity, according to ASTM D1613 weight% HAc | 0.001; |
| pH, according to ASTM D 1287 | 7.0; |
| sulfur content, according to ASTM D 5453 | 18 mg/kg; |
| gum content, according to ASTM D381 | 2 mg/100ml; |
| water content, according to ASTM D6304 | 0.01% w/w; |
| aromatics, according to SS 155120, including benzene | 30.9 % by volume; |
| benzene alone, according to EN 238 | 0.7 % by volume; |
| DVPE, according to ASTM D 5191 | 90.0 kPa; |
| anti-knock index 0.5(RON+MON), according to ASTM D 2699-86 and ASTM D 2700-86 | 92.3 |
| CO | -14.0% |
| HC | -8.6%; |
| NOx | unchanged; |
| CO2 | + 1.0%; |
| NMHC | -6.7%; |
| Fuel consumption, Fc, 1/100km | +2.0%. |
DVPE = 27.5 kPa
Anti-knock index 0.5(RON + MON)=85.5
A92 : PB : Ethanol= 76 : 19 : 5 % by volume
DVPE = 36.5 kPa
0.5(RON + MON) = 89.0
A92 : PB : Ethanol= 72 : 18 : 10 % by volume
DVPE = 36.0 kPa
0.5(RON + MON) = 90.7
A92 : PB : Ethanol : Isoamyl alcohol : Naphtha = 60 : 15 : 9.2 : 0.8 : 15 % by volume
The boiling temperature for the naphtha is 140-200°C.
DVPE = 27.5 kPa
0.5(RON + MON) = 89.3
A92 : PB : Ethanol : n-Butanol : Naphtha : Xylene = 60 : 15 : 9.2 : 0.8 : 7.5 : 7.5 % by volume
The boiling temperature for the naphtha is 140-200°C.
DVPE = 27.5 kPa
0.5(RON + MON) = 91.2
A92 : PB : Ethanol : Tetrahydrofurfuryl alcohol : Isopropylbenzene = 60 : 15 : 9: 1 : 15 % by volume
DVPE = 27.5 kPa
0.5(RON + MON) = 91.3
DVPE = 27.3 kPa
Anti-knock index 0.5(RON + MON) = 88.0
A98 : PB : Ethanol = 76 : 19 : 5 % by volume
DVPE = 36.3 kPa
0.5(RON + MON) = 91.0
A98 : PB : Ethanol = 72 : 18 : 10 % by volume
DVPE = 35.8 kPa
0.5(RON + MON) = 92.5
A98 : PB : Ethanol: Isoamyl alcohol : Naphtha = 60 : 15 : 9.2 : 0.8 : 15 % by volume
The boiling temperature for the naphtha is 140-200°C.
DVPE = 27.0 kPa
0.5(RON + MON) = 91.7
A98 : PB : Ethanol: Linalool : Allocymene = 60 : 15 : 9 : 1 : 15 % by volume
DVPE = 26.0 kPa
0.5(RON + MON) = 93.0
A98 : PB : Ethanol : Methylcyclohexanol : Limonene = 60 : 15 : 9.5 : 1 : 14.5 % by volume
DVPE = 25.4 kPa
0.5(RON + MON) = 93.2
DVPE = 27.6 kPa
Anti-knock index 0.5(RON + MON) = 86.3
| CO | 2.631 g/km; |
| HC | 0.348 g/km; |
| NOx | 0.313 g/km; |
| CO2 | 235.1 g/km; |
| NMHC | 0.308 g/km; |
| Fuel consumption, Fc, 1/100km | 10.68 |
A95 : PB : Ethanol = 76 : 19 : 5 % by volume
DVPE = 36.6 kPa
0.5(RON + MON) = 90.2
A95 : PB : Ethanol = 72 : 18 : 10 % by volume
DVPE = 36.1 kPa
0.5(RON + MON) = 91.7
| CO | -4.8% |
| HC | -1.3%; |
| NOx | +26.3%; |
| CO2 | +4.4%; |
| NMHC | -0.6%; |
| Fuel consumption, Fc, 1/100km | +5.7%. |
A95 : PB : Ethanol: Isoamyl alcohol : Naphtha = 60 : 15 : 9.2 : 0.8 : 15 % by volume
The boiling temperature for the naphtha is 140-200°C.
DVPE = 27.1 kPa
0.5(RON + MON) = 91.4
A95 : PB : Ethanol: Tetrahydrofurfuryl alcohol : Tert-butylcyclohexane = 60 : 15 : 9.2 : 0.8 : 15 % by volume
DVPE = 26.5 kPa
0.5(RON + MON) = 90.7
A95 : PB : Ethanol: 4-Methyl-4-hydroxytetrahydropyran : Isopropyltoluene = 60 : 15 : 9.2 : 0.8 : 15 % by volume
DVPE = 26.1 kPa
0.5(RON + MON) = 92.0
| density at 15°C, according to ASTM D4052 | 764.6 kg/m3; |
| initial boiling point, according to ASTM D 86 | 48.9°C; |
| vaporisable portion - 70°C | 25.3 % by volume; |
| vaporisable portion - 100°C | 50.8 % by volume; |
| vaporisable portion - 150°C | 76.5 % by volume; |
| vaporisable portion - 190°C | 95.6 % by volume; |
| final boiling point | 204.5°C; |
| evaporation residue | 1.4 % by volume; |
| loss by evaporation | 0.5 % by volume; |
| oxygen content, according to ASTM D4815 | 4.6% w/w; |
| acidity, according to ASTM D 1613 weight% HAc | 0.08; |
| pH, according to ASTM D1287 | 7.5; |
| sulfur content, according to ASTM D 5453 | 39 mg/kg; |
| gum content, according to ASTM D381 | 1.5 mg/100ml; |
| water content, according to ASTM D6304 | 0.1% w/w; |
| aromatics, according to SS 155120, including benzene | 38 % by volume; |
| benzene alone, according to EN 238 | 0.4 % by volume; |
| DVPE, according to ASTM D 5191 | 27.2 kPa; |
| anti-knock index 0.5(RON+MON), according to ASTM D 2699-86 and ASTM D 2700-86 | 91.8 |
| CO | -12.3% |
| HC | -6.2%; |
| NOx | unchanged; |
| CO2 | +2.6%; |
| NMHC | -6.4%; |
| Fuel consumption, Fc, 1/ 100km | +3.7% |
Claims (9)
- A method of reducing the vapour pressure of a C3-C12 hydrocarbon-based motor fuel mixture for conventional spark ignition internal combustion engines containing 0.1 to 20% by volume of ethanol, not more than 0.25% by weight of water according to ASTM D 6304, and not more than 7% by weight of oxygen according to ASTM D 4815, by at least 80 % of the ethanol induced vapour pressure increase, and more preferably to the vapour pressure of the C3-C12 hydrocarbon component (a) alone, wherein, in addition to the C3-C12 hydrocarbon component (a) and an ethanol component (b), an oxygen-containing component (c) is present in the fuel mixture in an amount from 0.05 up to 15% by volume of the total volume of the fuel mixture; the component (c) being selected from at least one of the following types of compounds:wherein a component (d), selected from at least one C6-C12 hydrocarbon, is present in the fuel mixture in an amount such that the ratio (b):((c)+(d)) is from 1:200 to 200:1 by volume.alkanol, having from 3 to 10 carbon atoms;dialkyl ether, having from 6 to 10 carbon atoms;ketone, having from 4 to 9 carbon atoms;alkyl ester of alkanoic acid, having from 5 to 8 carbon atoms;hydroxyketone, having from 4 to 6 carbon atoms;ketone ester of alkanoic acid, having from 5 to 8 carbon atoms;oxygen-containing heterocyclic compound selected from the following: tetrahydrofurfuryl alcohol, tetrahydrofurfuryl acetate, dimethyltetrahydro-furan, tetramethyltetrahydrofuran, methyl tetrahydropyrane, 4-methyl-4-oxytetrahydropyrane, and the mixtures hereof; and
- The method of claim 1, characterised in that the oxygen-containing component (c) and component (d) are added to the ethanol component (b), which mixture of (c), (b) and (d) is subsequently added to the hydrocarbon component (a).
- The method of claim 1, characterised in that the ethanol component (b) is added to the hydrocarbon component (a) to which mixture of (b) and (a) the oxygen-containing component (c) is added, thereby obtaining a mixture of (a), (b) and (c), to which mixture component (d) is added.
- The method of any of the preceding claims, characterised in that the C3-C12 hydrocarbon component (a) is selected from the group consisting of a non-reformulated standard type gasoline, a hydrocarbon liquid from petroleum refining, a hydrocarbon liquid from natural gas, a hydrocarbon liquid from an off-gas of chemical-recovery carbonisation, a hydrocarbon liquid from synthesis gas processing, or mixtures thereof, a non-reformulated standard type gasoline being preferred.
- The method of any of the preceding claims, characterised in that the fuel composition obtained exhibits the following characteristics;(i) a density at 15°C, according to ASTM D 4052 of at least 690 kg /m3;(ii) a dry vapour pressure equivalent according to ASTM D 5191 from 20 kPa to 120 kPa;(iii) an acids content according to ASTM D 1613 of no greater than 0.1 weight % HAc;(iv) a pH according to ASTM D 1287 from 5 to 9;(v) an aromatics content according to SS 155120 of no greater than 40% by volume, wherein benzene is present in amounts according to EN 238 no greater than 1% by volume;(vi) a sulphur content according to ASTM D 5453 of no greater than 50 mg/kg;(vii) a gum content according to ASTM D 381 of no greater than 2 mg/100 ml;(viii) distillation properties according to ASTM D86 wherein initial boiling point is at least 20°C; a vaporisable portion at 70°C is at least 25% by volume; a vaporisable portion at 100°C is at least 50% by volume; a vaporisable portion at 150°C is at least 75% by volume; a vaporisable portion at 190°C is at least 95% by volume; a final boiling point no greater than 205°C; and an evaporation residue no greater than 2% by volume; and(ix) an anti-knock index 0.5 (RON+MON) according to ASTM D 2699-86 and ASTM D 2700-86 of at least 80.
- The method of any of the preceding claims, characterised in that hydrocarbon component (d) is selected from benzene, toluene, xylene, ethylbenzene, isopropylbenzene, isopropyltoluene, diethylbenzene, isopropylxylene, tert-butylbenzene, tert-butyltoluene, tert-butylxylene, cyclooctadiene, cyclooctotetraene, limonene, isooctane, isononane, isodecane, isooctene, myrcene, allocymene, tert-butylcyclohexane and mixtures hereof.
- The method of any of the preceding claims, characterised in that hydrocarbon component (d) is selected from a C8-C11 fraction.
- The method of any of the preceding claims, characterised in that hydrocarbon component (d) is selected from a fraction boiling at 100-200°C, obtained in the distillation of oil, bituminous coal resin, or synthesis gas processing products.
- The method of any of the preceding claims, characterised in that the ratio of (b):((c)+(d)) is from 1: 10 to 10:1 by volume.
Priority Applications (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| SI200130387T SI1252268T1 (en) | 2000-01-24 | 2001-01-24 | Method of reducing the vapour pressure of ethanol-containing motor fuels for spark ignition combustion engines |
| EP05104353A EP1589091A1 (en) | 2000-01-24 | 2001-01-24 | Ethanol-Containing motor fuels for spark ignition combustion engines having reduced vapour pressure |
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| WOPCT/SE00/00139 | 2000-01-24 | ||
| PCT/SE2000/000139 WO2001053436A1 (en) | 2000-01-24 | 2000-01-24 | Motor fuel for spark ignition internal combustion engines |
| PCT/SE2001/000040 WO2001053437A1 (en) | 2000-01-24 | 2001-01-24 | Method of reducing the vapour pressure of ethanol-containing motor fuels for spark ignition combustion engines |
Related Child Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP05104353A Division EP1589091A1 (en) | 2000-01-24 | 2001-01-24 | Ethanol-Containing motor fuels for spark ignition combustion engines having reduced vapour pressure |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| EP1252268A1 EP1252268A1 (en) | 2002-10-30 |
| EP1252268B1 true EP1252268B1 (en) | 2005-05-25 |
Family
ID=20278139
Family Applications (2)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP05104353A Ceased EP1589091A1 (en) | 2000-01-24 | 2001-01-24 | Ethanol-Containing motor fuels for spark ignition combustion engines having reduced vapour pressure |
| EP01942659A Expired - Lifetime EP1252268B1 (en) | 2000-01-24 | 2001-01-24 | Method of reducing the vapour pressure of ethanol-containing motor fuels for spark ignition combustion engines |
Family Applications Before (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| EP05104353A Ceased EP1589091A1 (en) | 2000-01-24 | 2001-01-24 | Ethanol-Containing motor fuels for spark ignition combustion engines having reduced vapour pressure |
Country Status (29)
| Country | Link |
|---|---|
| EP (2) | EP1589091A1 (en) |
| JP (1) | JP4871475B2 (en) |
| KR (1) | KR100545054B1 (en) |
| CN (1) | CN1177914C (en) |
| AT (1) | ATE296342T1 (en) |
| AU (2) | AU3684800A (en) |
| BG (1) | BG66039B1 (en) |
| BR (1) | BR0107817B1 (en) |
| CA (1) | CA2397579C (en) |
| CU (1) | CU23146A3 (en) |
| CZ (1) | CZ305710B6 (en) |
| DE (1) | DE60111018T2 (en) |
| DK (1) | DK1252268T3 (en) |
| EA (1) | EA006855B1 (en) |
| EE (1) | EE05647B1 (en) |
| ES (1) | ES2243510T3 (en) |
| HR (1) | HRP20020670B1 (en) |
| HU (1) | HU230551B1 (en) |
| IL (1) | IL150624A (en) |
| MX (1) | MXPA02007148A (en) |
| NO (1) | NO336184B1 (en) |
| PL (1) | PL194561B1 (en) |
| PT (1) | PT1252268E (en) |
| RS (1) | RS52075B (en) |
| SI (1) | SI1252268T1 (en) |
| SK (1) | SK287660B6 (en) |
| UA (1) | UA76945C2 (en) |
| WO (2) | WO2001053436A1 (en) |
| ZA (1) | ZA200205833B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3187570A1 (en) | 2015-12-29 | 2017-07-05 | Neste Oyj | Renewable dvpe adjustment material, fuel blend containing the same, and method for producing a fuel blend |
Families Citing this family (46)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6761745B2 (en) | 2000-01-24 | 2004-07-13 | Angelica Hull | Method of reducing the vapor pressure of ethanol-containing motor fuels for spark ignition combustion engines |
| US7981170B1 (en) * | 2000-04-21 | 2011-07-19 | Shell Oil Company | Gasoline-oxygenate blend and method of producing the same |
| US6565617B2 (en) * | 2000-08-24 | 2003-05-20 | Shell Oil Company | Gasoline composition |
| WO2003062354A1 (en) * | 2002-01-21 | 2003-07-31 | Ramar Ponniah | Hydrocarbon fuel |
| JP2005187706A (en) * | 2003-12-26 | 2005-07-14 | Japan Energy Corp | Ethanol-containing gasoline and method for producing the same |
| BRPI0614026A2 (en) * | 2005-01-25 | 2012-12-25 | Bp Corp North America Inc | petrol composition and rvp reduction methods of a hydrogen peroxide and rvp construction over in gasoline mixture stock in the production of peroxide having a predetermined maximum limit of rvp |
| AU2011226816B2 (en) * | 2005-01-25 | 2012-08-30 | Bp Corporation North America Inc. | Reduced RVP oxygenated gasoline composition and method |
| US20090199464A1 (en) * | 2008-02-12 | 2009-08-13 | Bp Corporation North America Inc. | Reduced RVP Oxygenated Gasoline Composition And Method |
| JP4624143B2 (en) * | 2005-03-11 | 2011-02-02 | コスモ石油株式会社 | Ethanol blended gasoline |
| JP4624142B2 (en) * | 2005-03-11 | 2011-02-02 | コスモ石油株式会社 | Ethanol blended gasoline |
| US7389751B2 (en) * | 2006-03-17 | 2008-06-24 | Ford Global Technology, Llc | Control for knock suppression fluid separator in a motor vehicle |
| US7854774B2 (en) | 2006-05-26 | 2010-12-21 | Amyris Biotechnologies, Inc. | Fuel components, fuel compositions and methods of making and using same |
| DK2024504T4 (en) | 2006-05-26 | 2023-02-27 | Amyris Inc | Production of isoprenoids |
| KR100812423B1 (en) * | 2006-12-13 | 2008-03-10 | 현대자동차주식회사 | Method and apparatus for calculating ethanol concentration in fuel |
| NL1033228C2 (en) * | 2007-01-15 | 2008-07-16 | Univ Eindhoven Tech | Liquid fuel composition useful in compression-ignition engine, comprises a mixture of hydrocarbons containing a cyclic hydrocarbon compound having at least five carbon atoms and at least one oxygen atom |
| WO2008088212A1 (en) | 2007-01-15 | 2008-07-24 | Technische Universiteit Eindhoven | A liquid fuel composition and the use thereof |
| DE102008008818A1 (en) * | 2008-02-12 | 2009-08-20 | Deutsche Bp Ag | Fuels for petrol engines |
| AU2008352537A1 (en) * | 2008-03-12 | 2009-09-17 | Ramar Ponnupillai | Velar bio hydrocarbon fuel |
| US9476004B2 (en) | 2009-09-08 | 2016-10-25 | Technische Universiteit Eindhoven | Liquid fuel composition and the use thereof |
| JP5144729B2 (en) * | 2010-09-10 | 2013-02-13 | コスモ石油株式会社 | Production method of ethanol blended gasoline |
| JP5214688B2 (en) * | 2010-09-10 | 2013-06-19 | コスモ石油株式会社 | Production method of ethanol blended gasoline |
| EP2683798B1 (en) | 2011-03-10 | 2017-03-08 | Shell Internationale Research Maatschappij B.V. | Use of camphene in a gasoline fuel formulations |
| WO2013023372A1 (en) * | 2011-08-17 | 2013-02-21 | 湖南中创化工股份有限公司 | A gasoline composition and its preparation method |
| NL2007304C2 (en) * | 2011-08-26 | 2013-02-27 | Progression Industry B V | Use of perfume composition as fuel for internal combustion engines. |
| US8968429B2 (en) | 2011-09-23 | 2015-03-03 | Butamax Advanced Biofuels Llc | Butanol compositions for fuel blending and methods for the production thereof |
| CN102746909B (en) * | 2012-07-20 | 2014-04-16 | 杨如平 | Methanol fuel modifier and high-proportion methanol fuel for gasoline engine |
| CN102876404B (en) * | 2012-10-11 | 2014-10-22 | 陕西延长石油(集团)有限责任公司研究院 | Additive capable of reducing methanol gasoline saturated vapor pressure |
| ITMI20122006A1 (en) * | 2012-11-26 | 2014-05-27 | Eni Spa | USEFUL COMPOSITIONS AS FUELS INCLUDING HYDROPHOBIC OXYGENATED COMPOUNDS |
| CN104004554B (en) * | 2014-06-05 | 2016-01-20 | 上海化工研究院 | Vehicle alcohol-ether substitute fuel tail gas activator and its preparation method and application |
| TR201902852T4 (en) * | 2014-10-06 | 2019-03-21 | Shell Int Research | Low vapor pressure fuel composition. |
| US11913166B2 (en) | 2015-09-21 | 2024-02-27 | Modern Meadow, Inc. | Fiber reinforced tissue composites |
| RU2605952C1 (en) * | 2015-12-25 | 2017-01-10 | Акционерное общество "Всероссийский научно-исследовательский институт по переработке нефти" (АО "ВНИИ НП") | Alternative motor fuel and production method thereof |
| RU2605954C1 (en) * | 2015-12-25 | 2017-01-10 | Акционерное общество "Всероссийский научно-исследовательский институт по переработке нефти" (АО "ВНИИ НП") | Alternative motor fuel and production method thereof |
| EP3205702A1 (en) | 2016-02-11 | 2017-08-16 | Bp Oil International Limited | Fuel compositions with additives |
| EP3205703A1 (en) | 2016-02-11 | 2017-08-16 | Bp Oil International Limited | Fuel additives |
| EP3205701A1 (en) | 2016-02-11 | 2017-08-16 | Bp Oil International Limited | Fuel compositions |
| KR20170096096A (en) | 2016-02-15 | 2017-08-23 | 브렌던 패트릭 퍼셀 | Method for making a biofabricated material containing collagen fibrils |
| RU2616606C1 (en) * | 2016-04-14 | 2017-04-18 | Акционерное общество "Всероссийский научно-исследовательский институт по переработке нефти" (АО "ВНИИ НП") | High-octane motor gasoline and anti-knock additive for production thereof |
| ES2862192T3 (en) | 2016-09-29 | 2021-10-07 | Neste Oyj | Diesel fuel comprising 5-nonanone |
| EP3399008B1 (en) | 2017-05-02 | 2020-03-18 | ASG Analytik-Service Gesellschaft mbH | Potentially co2-neutral and ecological gasoline based on c1-chemistry |
| AU2018253595A1 (en) | 2017-11-13 | 2019-05-30 | Modern Meadow, Inc. | Biofabricated leather articles having zonal properties |
| WO2020150443A1 (en) | 2019-01-17 | 2020-07-23 | Modern Meadow, Inc. | Layered collagen materials and methods of making the same |
| RU2740554C1 (en) * | 2020-08-13 | 2021-01-15 | федеральное государственное автономное образовательное учреждение высшего образования "Российский государственный университет нефти и газа (национальный исследовательский университет) имени И.М. Губкина" | High-octane gasoline |
| FI20205840A1 (en) * | 2020-08-31 | 2022-03-01 | Neste Oyj | Octane enhanced intermediate hydrocarbon composition |
| FI129568B (en) | 2021-04-15 | 2022-04-29 | Neste Oyj | 2-butanone and ethanol as gasoline components |
| CA3237233A1 (en) * | 2021-11-16 | 2023-05-25 | Richard HEDIGER | Method for producing of a fuel additive |
Family Cites Families (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US2104021A (en) * | 1935-04-24 | 1938-01-04 | Callis Conral Cleo | Fuel |
| US2365009A (en) * | 1940-12-19 | 1944-12-12 | Standard Oil Dev Co | Motor fuels |
| RO75851A2 (en) * | 1978-02-22 | 1981-02-28 | Institutul National De Motoare Termice,Ro | COMBUSTIBLE MIXTURE |
| US4207076A (en) * | 1979-02-23 | 1980-06-10 | Texaco Inc. | Gasoline-ethanol fuel mixture solubilized with ethyl-t-butyl ether |
| DK148747C (en) * | 1980-06-09 | 1986-02-24 | Inst Francais Du Petrole | motor fuel |
| US4328004A (en) * | 1980-08-13 | 1982-05-04 | United International Research, Inc. | Stabilization of ethanol-gasoline mixtures |
| FR2493863A1 (en) * | 1980-11-07 | 1982-05-14 | Inst Francais Du Petrole | NEW FUEL BASED ON FUEL CONTAINING ETHANOL HYDRATE AND AN ADDITIVE |
| DE3150989A1 (en) * | 1980-12-30 | 1982-08-05 | Institut Français du Pétrole, 92502 Rueil-Malmaison, Hauts-de-Seine | COMBUSTIBLE COMPOSITIONS CONTAINING A GAS OIL, AT LEAST ONE FATTY ACID ESTER AND AN ALCOHOLIC COMPONENT BASED ON N-BUTANOL, AND ARE USED AS DIESEL FUELS |
| FR2500844A1 (en) * | 1981-03-02 | 1982-09-03 | Realisations Sarl Et | Preventing phase-sepn. of mixt. of hydrocarbon fuel and an alcohol - using additive mixt. contg. benzyl alcohol and acetate |
| DE3116734C2 (en) * | 1981-04-28 | 1985-07-25 | Veba Oel AG, 4650 Gelsenkirchen | Carburetor fuel |
| US4451266A (en) * | 1982-01-22 | 1984-05-29 | John D. Barclay | Additive for improving performance of liquid hydrocarbon fuels |
| US4541836A (en) * | 1982-12-09 | 1985-09-17 | Union Carbide Corporation | Fuel compositions |
| ATE43625T1 (en) * | 1983-03-03 | 1989-06-15 | Union Rheinische Braunkohlen | ENGINE FUEL. |
| DE3330165C2 (en) * | 1983-08-20 | 1985-10-03 | Union Rheinische Braunkohlen Kraftstoff AG, 5000 Köln | Engine fuel |
| US4891050A (en) * | 1985-11-08 | 1990-01-02 | Fuel Tech, Inc. | Gasoline additives and gasoline containing soluble platinum group metal compounds and use in internal combustion engines |
| US4806129A (en) * | 1987-09-21 | 1989-02-21 | Prepolene Industries, Inc. | Fuel extender |
| US4818250A (en) * | 1987-10-21 | 1989-04-04 | Lemco Energy, Inc. | Process for producing fuel from plant sources and fuel blends containing same |
| ES2012729A6 (en) * | 1989-06-07 | 1990-04-01 | Vicente Rodriguez Heliodoro | Oxygenated organic fuel additive prepn. - by making seven mixts., emulsifying and standing, followed by combining, homogenising and standing |
| CN1017061B (en) * | 1990-02-13 | 1992-06-17 | 唐昌干 | Anti-knocking, fume-reducing, oil-saving agent for vehicles and ships and process for producing same |
| DE4308053C2 (en) * | 1993-03-13 | 1997-05-15 | Veba Oel Ag | Liquid unleaded fuels |
| US5607486A (en) * | 1994-05-04 | 1997-03-04 | Wilkins, Jr.; Joe S. | Engine fuels |
| US5688295A (en) * | 1996-05-08 | 1997-11-18 | H. E. W. D. Enterprises-America, Inc. | Gasoline fuel additive |
| US5697987A (en) * | 1996-05-10 | 1997-12-16 | The Trustees Of Princeton University | Alternative fuel |
| EP1047756A2 (en) * | 1998-01-12 | 2000-11-02 | Deborah Wenzel | An additive composition also used as a fuel composition comprising water soluble alcohols |
-
2000
- 2000-01-24 AU AU36848/00A patent/AU3684800A/en not_active Abandoned
- 2000-01-24 WO PCT/SE2000/000139 patent/WO2001053436A1/en not_active Ceased
-
2001
- 2001-01-11 PL PL01356843A patent/PL194561B1/en unknown
- 2001-01-11 HU HUP0204201A patent/HU230551B1/en unknown
- 2001-01-24 BR BRPI0107817-8A patent/BR0107817B1/en not_active IP Right Cessation
- 2001-01-24 DK DK01942659T patent/DK1252268T3/en active
- 2001-01-24 UA UA2002076155A patent/UA76945C2/en unknown
- 2001-01-24 EE EEP200200407A patent/EE05647B1/en active IP Right Revival
- 2001-01-24 EA EA200200796A patent/EA006855B1/en not_active IP Right Cessation
- 2001-01-24 EP EP05104353A patent/EP1589091A1/en not_active Ceased
- 2001-01-24 DE DE60111018T patent/DE60111018T2/en not_active Expired - Lifetime
- 2001-01-24 AU AU28949/01A patent/AU782062B2/en not_active Ceased
- 2001-01-24 AT AT01942659T patent/ATE296342T1/en active
- 2001-01-24 IL IL15062401A patent/IL150624A/en active IP Right Grant
- 2001-01-24 CZ CZ2002-2869A patent/CZ305710B6/en not_active IP Right Cessation
- 2001-01-24 CN CNB018040500A patent/CN1177914C/en not_active Expired - Fee Related
- 2001-01-24 EP EP01942659A patent/EP1252268B1/en not_active Expired - Lifetime
- 2001-01-24 ES ES01942659T patent/ES2243510T3/en not_active Expired - Lifetime
- 2001-01-24 SK SK1222-2002A patent/SK287660B6/en not_active IP Right Cessation
- 2001-01-24 RS YU55802A patent/RS52075B/en unknown
- 2001-01-24 MX MXPA02007148A patent/MXPA02007148A/en active IP Right Grant
- 2001-01-24 WO PCT/SE2001/000040 patent/WO2001053437A1/en not_active Ceased
- 2001-01-24 HR HR20020670A patent/HRP20020670B1/en not_active IP Right Cessation
- 2001-01-24 CA CA2397579A patent/CA2397579C/en not_active Expired - Lifetime
- 2001-01-24 SI SI200130387T patent/SI1252268T1/en unknown
- 2001-01-24 KR KR1020027009521A patent/KR100545054B1/en not_active Expired - Fee Related
- 2001-01-24 JP JP2001553900A patent/JP4871475B2/en not_active Expired - Fee Related
- 2001-01-24 PT PT01942659T patent/PT1252268E/en unknown
-
2002
- 2002-07-17 CU CU149A patent/CU23146A3/en unknown
- 2002-07-22 ZA ZA200205833A patent/ZA200205833B/en unknown
- 2002-07-23 NO NO20023502A patent/NO336184B1/en not_active IP Right Cessation
- 2002-08-15 BG BG107007A patent/BG66039B1/en unknown
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| EP3187570A1 (en) | 2015-12-29 | 2017-07-05 | Neste Oyj | Renewable dvpe adjustment material, fuel blend containing the same, and method for producing a fuel blend |
Also Published As
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| EP1252268B1 (en) | Method of reducing the vapour pressure of ethanol-containing motor fuels for spark ignition combustion engines | |
| US6761745B2 (en) | Method of reducing the vapor pressure of ethanol-containing motor fuels for spark ignition combustion engines | |
| US5697987A (en) | Alternative fuel | |
| US20010034966A1 (en) | Method of reducing the vapor pressure of ethanol-containing motor fuels for spark ignition combustion engines | |
| JP2006515377A (en) | High alcohol alternative fuel | |
| BRPI0619727A2 (en) | unleaded aviation fuel | |
| JP2000026871A (en) | Low-pollution fuel composition | |
| JP4691269B2 (en) | Fuel oil composition for gasoline engines | |
| JP4920185B2 (en) | Gasoline composition | |
| JP4746869B2 (en) | High performance gasoline | |
| JPH06192667A (en) | Gasoline composition | |
| US20100000483A1 (en) | Gasoline compositions |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PUAI | Public reference made under article 153(3) epc to a published international application that has entered the european phase |
Free format text: ORIGINAL CODE: 0009012 |
|
| 17P | Request for examination filed |
Effective date: 20020815 |
|
| AK | Designated contracting states |
Kind code of ref document: A1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE TR |
|
| AX | Request for extension of the european patent |
Free format text: AL PAYMENT 20020815;LT PAYMENT 20020815;LV PAYMENT 20020815;MK PAYMENT 20020815;RO PAYMENT 20020815;SI PAYMENT 20020815 |
|
| 17Q | First examination report despatched |
Effective date: 20031022 |
|
| GRAP | Despatch of communication of intention to grant a patent |
Free format text: ORIGINAL CODE: EPIDOSNIGR1 |
|
| GRAS | Grant fee paid |
Free format text: ORIGINAL CODE: EPIDOSNIGR3 |
|
| GRAA | (expected) grant |
Free format text: ORIGINAL CODE: 0009210 |
|
| AK | Designated contracting states |
Kind code of ref document: B1 Designated state(s): AT BE CH CY DE DK ES FI FR GB GR IE IT LI LU MC NL PT SE TR |
|
| AX | Request for extension of the european patent |
Extension state: AL LT LV MK RO SI |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: FG4D |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: EP |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: FG4D |
|
| REF | Corresponds to: |
Ref document number: 60111018 Country of ref document: DE Date of ref document: 20050630 Kind code of ref document: P |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: TRGR |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: T3 |
|
| REG | Reference to a national code |
Ref country code: GR Ref legal event code: EP Ref document number: 20050402596 Country of ref document: GR |
|
| REG | Reference to a national code |
Ref country code: PT Ref legal event code: SC4A Effective date: 20050729 Ref country code: CH Ref legal event code: NV Representative=s name: DR. LUSUARDI AG |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FG2A Ref document number: 2243510 Country of ref document: ES Kind code of ref document: T3 |
|
| PLBI | Opposition filed |
Free format text: ORIGINAL CODE: 0009260 |
|
| PLAX | Notice of opposition and request to file observation + time limit sent |
Free format text: ORIGINAL CODE: EPIDOSNOBS2 |
|
| ET | Fr: translation filed | ||
| 26 | Opposition filed |
Opponent name: BP OIL INTERNATIONAL LIMITED Effective date: 20060223 |
|
| NLR1 | Nl: opposition has been filed with the epo |
Opponent name: BP OIL INTERNATIONAL LIMITED |
|
| PLAF | Information modified related to communication of a notice of opposition and request to file observations + time limit |
Free format text: ORIGINAL CODE: EPIDOSCOBS2 |
|
| PLBB | Reply of patent proprietor to notice(s) of opposition received |
Free format text: ORIGINAL CODE: EPIDOSNOBS3 |
|
| PLCK | Communication despatched that opposition was rejected |
Free format text: ORIGINAL CODE: EPIDOSNREJ1 |
|
| APBM | Appeal reference recorded |
Free format text: ORIGINAL CODE: EPIDOSNREFNO |
|
| APBP | Date of receipt of notice of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA2O |
|
| APAH | Appeal reference modified |
Free format text: ORIGINAL CODE: EPIDOSCREFNO |
|
| APBQ | Date of receipt of statement of grounds of appeal recorded |
Free format text: ORIGINAL CODE: EPIDOSNNOA3O |
|
| APBU | Appeal procedure closed |
Free format text: ORIGINAL CODE: EPIDOSNNOA9O |
|
| PLBN | Opposition rejected |
Free format text: ORIGINAL CODE: 0009273 |
|
| STAA | Information on the status of an ep patent application or granted ep patent |
Free format text: STATUS: OPPOSITION REJECTED |
|
| 27O | Opposition rejected |
Effective date: 20110325 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R100 Ref document number: 60111018 Country of ref document: DE Effective date: 20110325 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R082 Ref document number: 60111018 Country of ref document: DE Representative=s name: WHITE & CASE LLP, DE |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R082 Ref document number: 60111018 Country of ref document: DE Representative=s name: WHITE & CASE LLP, DE Effective date: 20150119 Ref country code: DE Ref legal event code: R081 Ref document number: 60111018 Country of ref document: DE Owner name: SWEDISH BIOFUELS (IP) LIMITED, GB Free format text: FORMER OWNER: HULL, ANGELICA, LIDINGOE, SE Effective date: 20150119 |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: 732E Free format text: REGISTERED BETWEEN 20150205 AND 20150211 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R008 Ref document number: 60111018 Country of ref document: DE Ref country code: DE Ref legal event code: R039 Ref document number: 60111018 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 16 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: TR Payment date: 20160112 Year of fee payment: 16 |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 17 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R040 Ref document number: 60111018 Country of ref document: DE |
|
| REG | Reference to a national code |
Ref country code: FR Ref legal event code: PLFP Year of fee payment: 18 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: LU Payment date: 20171228 Year of fee payment: 18 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R082 Ref document number: 60111018 Country of ref document: DE Representative=s name: VOSSIUS & PARTNER PATENTANWAELTE RECHTSANWAELT, DE |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: LU Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20190124 |
|
| REG | Reference to a national code |
Ref country code: SI Ref legal event code: KO00 Effective date: 20190926 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: NL Payment date: 20200120 Year of fee payment: 20 Ref country code: PT Payment date: 20200108 Year of fee payment: 20 Ref country code: DE Payment date: 20190920 Year of fee payment: 20 Ref country code: ES Payment date: 20200203 Year of fee payment: 20 Ref country code: AT Payment date: 20200121 Year of fee payment: 20 Ref country code: IT Payment date: 20200117 Year of fee payment: 20 Ref country code: DK Payment date: 20200122 Year of fee payment: 20 Ref country code: IE Payment date: 20200116 Year of fee payment: 20 Ref country code: SE Payment date: 20200122 Year of fee payment: 20 Ref country code: MC Payment date: 20200116 Year of fee payment: 20 Ref country code: GR Payment date: 20200121 Year of fee payment: 20 Ref country code: GB Payment date: 20200120 Year of fee payment: 20 Ref country code: FI Payment date: 20200120 Year of fee payment: 20 |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: CH Payment date: 20200120 Year of fee payment: 20 Ref country code: BE Payment date: 20200120 Year of fee payment: 20 Ref country code: CY Payment date: 20191231 Year of fee payment: 20 |
|
| REG | Reference to a national code |
Ref country code: DE Ref legal event code: R042 Ref document number: 60111018 Country of ref document: DE |
|
| PGFP | Annual fee paid to national office [announced via postgrant information from national office to epo] |
Ref country code: FR Payment date: 20200116 Year of fee payment: 20 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: DE Free format text: THE PATENT HAS BEEN ANNULLED BY A DECISION OF A NATIONAL AUTHORITY Effective date: 20200609 |
|
| REG | Reference to a national code |
Ref country code: DK Ref legal event code: EUP Expiry date: 20210124 |
|
| REG | Reference to a national code |
Ref country code: NL Ref legal event code: MK Effective date: 20210123 |
|
| REG | Reference to a national code |
Ref country code: CH Ref legal event code: PL |
|
| REG | Reference to a national code |
Ref country code: GB Ref legal event code: PE20 Expiry date: 20210123 |
|
| REG | Reference to a national code |
Ref country code: FI Ref legal event code: MAE |
|
| REG | Reference to a national code |
Ref country code: SE Ref legal event code: EUG |
|
| REG | Reference to a national code |
Ref country code: IE Ref legal event code: MK9A |
|
| REG | Reference to a national code |
Ref country code: AT Ref legal event code: MK07 Ref document number: 296342 Country of ref document: AT Kind code of ref document: T Effective date: 20210124 |
|
| REG | Reference to a national code |
Ref country code: BE Ref legal event code: MK Effective date: 20210124 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: GB Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20210123 Ref country code: PT Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20210204 Ref country code: IE Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20210124 |
|
| REG | Reference to a national code |
Ref country code: ES Ref legal event code: FD2A Effective date: 20211229 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: ES Free format text: LAPSE BECAUSE OF EXPIRATION OF PROTECTION Effective date: 20210125 |
|
| PG25 | Lapsed in a contracting state [announced via postgrant information from national office to epo] |
Ref country code: TR Free format text: LAPSE BECAUSE OF NON-PAYMENT OF DUE FEES Effective date: 20200124 |