CN113940997B - Stable preparation of bispecific antibody - Google Patents
Stable preparation of bispecific antibody Download PDFInfo
- Publication number
- CN113940997B CN113940997B CN202111566377.XA CN202111566377A CN113940997B CN 113940997 B CN113940997 B CN 113940997B CN 202111566377 A CN202111566377 A CN 202111566377A CN 113940997 B CN113940997 B CN 113940997B
- Authority
- CN
- China
- Prior art keywords
- antibody
- bispecific antibody
- aqueous composition
- nkg
- seq
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 238000002360 preparation method Methods 0.000 title abstract description 16
- KRKNYBCHXYNGOX-UHFFFAOYSA-N citric acid Chemical compound OC(=O)CC(O)(C(O)=O)CC(O)=O KRKNYBCHXYNGOX-UHFFFAOYSA-N 0.000 claims abstract description 39
- 239000007853 buffer solution Substances 0.000 claims abstract description 15
- 235000010482 polyoxyethylene sorbitan monooleate Nutrition 0.000 claims abstract description 15
- 229920000053 polysorbate 80 Polymers 0.000 claims abstract description 15
- 229930006000 Sucrose Natural products 0.000 claims abstract description 11
- CZMRCDWAGMRECN-UGDNZRGBSA-N Sucrose Chemical compound O[C@H]1[C@H](O)[C@@H](CO)O[C@@]1(CO)O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 CZMRCDWAGMRECN-UGDNZRGBSA-N 0.000 claims abstract description 11
- 239000005720 sucrose Substances 0.000 claims abstract description 11
- 238000002347 injection Methods 0.000 claims abstract description 8
- 239000007924 injection Substances 0.000 claims abstract description 8
- 102000008096 B7-H1 Antigen Human genes 0.000 claims abstract 5
- 108010074708 B7-H1 Antigen Proteins 0.000 claims abstract 5
- 230000027455 binding Effects 0.000 claims description 48
- 239000000203 mixture Substances 0.000 claims description 31
- 239000000427 antigen Substances 0.000 claims description 30
- 102000036639 antigens Human genes 0.000 claims description 30
- 108091007433 antigens Proteins 0.000 claims description 30
- 102100022682 NKG2-A/NKG2-B type II integral membrane protein Human genes 0.000 claims description 28
- 101150069255 KLRC1 gene Proteins 0.000 claims description 25
- 101100404845 Macaca mulatta NKG2A gene Proteins 0.000 claims description 25
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 claims description 22
- 239000000872 buffer Substances 0.000 claims description 17
- 239000011780 sodium chloride Substances 0.000 claims description 11
- 239000003223 protective agent Substances 0.000 claims description 8
- 239000004094 surface-active agent Substances 0.000 claims description 6
- 239000007979 citrate buffer Substances 0.000 claims description 5
- 238000009472 formulation Methods 0.000 claims description 4
- 239000004615 ingredient Substances 0.000 claims 1
- QTBSBXVTEAMEQO-UHFFFAOYSA-N Acetic acid Chemical compound CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 abstract description 30
- 238000012360 testing method Methods 0.000 abstract description 29
- 238000001514 detection method Methods 0.000 abstract description 11
- 230000004071 biological effect Effects 0.000 abstract description 7
- 238000005286 illumination Methods 0.000 abstract description 4
- 239000003381 stabilizer Substances 0.000 abstract description 2
- 210000004027 cell Anatomy 0.000 description 29
- 239000012634 fragment Substances 0.000 description 26
- 238000001542 size-exclusion chromatography Methods 0.000 description 20
- 108090000623 proteins and genes Proteins 0.000 description 19
- 235000018102 proteins Nutrition 0.000 description 18
- 102000004169 proteins and genes Human genes 0.000 description 18
- 239000000523 sample Substances 0.000 description 17
- 238000004255 ion exchange chromatography Methods 0.000 description 14
- 150000001413 amino acids Chemical class 0.000 description 13
- 239000002245 particle Substances 0.000 description 12
- 238000013368 capillary electrophoresis sodium dodecyl sulfate analysis Methods 0.000 description 11
- 230000000694 effects Effects 0.000 description 11
- 230000003993 interaction Effects 0.000 description 10
- 238000011835 investigation Methods 0.000 description 10
- 239000003446 ligand Substances 0.000 description 10
- 239000000244 polyoxyethylene sorbitan monooleate Substances 0.000 description 10
- 229940068968 polysorbate 80 Drugs 0.000 description 10
- 235000001014 amino acid Nutrition 0.000 description 9
- 238000010494 dissociation reaction Methods 0.000 description 9
- 230000005593 dissociations Effects 0.000 description 9
- 238000000034 method Methods 0.000 description 8
- 102100028970 HLA class I histocompatibility antigen, alpha chain E Human genes 0.000 description 7
- 101000986085 Homo sapiens HLA class I histocompatibility antigen, alpha chain E Proteins 0.000 description 7
- 101001109508 Homo sapiens NKG2-A/NKG2-B type II integral membrane protein Proteins 0.000 description 7
- 206010028980 Neoplasm Diseases 0.000 description 7
- 210000001744 T-lymphocyte Anatomy 0.000 description 7
- 230000008859 change Effects 0.000 description 7
- 230000002829 reductive effect Effects 0.000 description 7
- 210000004881 tumor cell Anatomy 0.000 description 7
- 108010047041 Complementarity Determining Regions Proteins 0.000 description 6
- 108010054477 Immunoglobulin Fab Fragments Proteins 0.000 description 5
- 102000001706 Immunoglobulin Fab Fragments Human genes 0.000 description 5
- 230000000903 blocking effect Effects 0.000 description 5
- 238000002474 experimental method Methods 0.000 description 5
- HNDVDQJCIGZPNO-UHFFFAOYSA-N histidine Natural products OC(=O)C(N)CC1=CN=CN1 HNDVDQJCIGZPNO-UHFFFAOYSA-N 0.000 description 5
- 210000000822 natural killer cell Anatomy 0.000 description 5
- 238000005457 optimization Methods 0.000 description 5
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 5
- 101001117317 Homo sapiens Programmed cell death 1 ligand 1 Proteins 0.000 description 4
- 238000003556 assay Methods 0.000 description 4
- 230000008901 benefit Effects 0.000 description 4
- 102000048776 human CD274 Human genes 0.000 description 4
- 102000057310 human KLRC1 Human genes 0.000 description 4
- 238000009169 immunotherapy Methods 0.000 description 4
- 230000006641 stabilisation Effects 0.000 description 4
- 238000011105 stabilization Methods 0.000 description 4
- 239000000126 substance Substances 0.000 description 4
- 238000002198 surface plasmon resonance spectroscopy Methods 0.000 description 4
- 101000971513 Homo sapiens Natural killer cells antigen CD94 Proteins 0.000 description 3
- 102000018071 Immunoglobulin Fc Fragments Human genes 0.000 description 3
- 108010091135 Immunoglobulin Fc Fragments Proteins 0.000 description 3
- 102100021462 Natural killer cells antigen CD94 Human genes 0.000 description 3
- 230000002776 aggregation Effects 0.000 description 3
- 238000004220 aggregation Methods 0.000 description 3
- 108010069205 aspartyl-phenylalanine Proteins 0.000 description 3
- 230000003247 decreasing effect Effects 0.000 description 3
- UQLDLKMNUJERMK-UHFFFAOYSA-L di(octadecanoyloxy)lead Chemical compound [Pb+2].CCCCCCCCCCCCCCCCCC([O-])=O.CCCCCCCCCCCCCCCCCC([O-])=O UQLDLKMNUJERMK-UHFFFAOYSA-L 0.000 description 3
- 239000012636 effector Substances 0.000 description 3
- 238000000684 flow cytometry Methods 0.000 description 3
- 230000006870 function Effects 0.000 description 3
- 210000002865 immune cell Anatomy 0.000 description 3
- 230000002401 inhibitory effect Effects 0.000 description 3
- 238000004519 manufacturing process Methods 0.000 description 3
- 210000000581 natural killer T-cell Anatomy 0.000 description 3
- 229920000136 polysorbate Polymers 0.000 description 3
- 108020003175 receptors Proteins 0.000 description 3
- 102000005962 receptors Human genes 0.000 description 3
- 230000004044 response Effects 0.000 description 3
- 238000005406 washing Methods 0.000 description 3
- MNQMTYSEKZHIDF-GCJQMDKQSA-N Asp-Thr-Ala Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(O)=O MNQMTYSEKZHIDF-GCJQMDKQSA-N 0.000 description 2
- 210000001266 CD8-positive T-lymphocyte Anatomy 0.000 description 2
- YQPFCZVKMUVZIN-AUTRQRHGSA-N Glu-Val-Gln Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O YQPFCZVKMUVZIN-AUTRQRHGSA-N 0.000 description 2
- BYYNJRSNDARRBX-YFKPBYRVSA-N Gly-Gln-Gly Chemical compound NCC(=O)N[C@@H](CCC(N)=O)C(=O)NCC(O)=O BYYNJRSNDARRBX-YFKPBYRVSA-N 0.000 description 2
- 102000008394 Immunoglobulin Fragments Human genes 0.000 description 2
- 108010021625 Immunoglobulin Fragments Proteins 0.000 description 2
- 108010067060 Immunoglobulin Variable Region Proteins 0.000 description 2
- 102000017727 Immunoglobulin Variable Region Human genes 0.000 description 2
- HNDVDQJCIGZPNO-YFKPBYRVSA-N L-histidine Chemical compound OC(=O)[C@@H](N)CC1=CN=CN1 HNDVDQJCIGZPNO-YFKPBYRVSA-N 0.000 description 2
- PDIDTSZKKFEDMB-UWVGGRQHSA-N Lys-Pro-Gly Chemical compound [H]N[C@@H](CCCCN)C(=O)N1CCC[C@H]1C(=O)NCC(O)=O PDIDTSZKKFEDMB-UWVGGRQHSA-N 0.000 description 2
- 108010079364 N-glycylalanine Proteins 0.000 description 2
- 101710094000 Programmed cell death 1 ligand 1 Proteins 0.000 description 2
- 102100024216 Programmed cell death 1 ligand 1 Human genes 0.000 description 2
- 108091007744 Programmed cell death receptors Proteins 0.000 description 2
- 102000001708 Protein Isoforms Human genes 0.000 description 2
- 108010029485 Protein Isoforms Proteins 0.000 description 2
- 102000007056 Recombinant Fusion Proteins Human genes 0.000 description 2
- 108010008281 Recombinant Fusion Proteins Proteins 0.000 description 2
- DBMJMQXJHONAFJ-UHFFFAOYSA-M Sodium laurylsulphate Chemical compound [Na+].CCCCCCCCCCCCOS([O-])(=O)=O DBMJMQXJHONAFJ-UHFFFAOYSA-M 0.000 description 2
- OWFGFHQMSBTKLX-UFYCRDLUSA-N Val-Tyr-Tyr Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H](CC2=CC=C(C=C2)O)C(=O)O)N OWFGFHQMSBTKLX-UFYCRDLUSA-N 0.000 description 2
- 230000021736 acetylation Effects 0.000 description 2
- 238000006640 acetylation reaction Methods 0.000 description 2
- 108010069020 alanyl-prolyl-glycine Proteins 0.000 description 2
- 238000004458 analytical method Methods 0.000 description 2
- 230000009830 antibody antigen interaction Effects 0.000 description 2
- 230000008827 biological function Effects 0.000 description 2
- 210000004899 c-terminal region Anatomy 0.000 description 2
- 238000005251 capillar electrophoresis Methods 0.000 description 2
- 238000003776 cleavage reaction Methods 0.000 description 2
- 235000018417 cysteine Nutrition 0.000 description 2
- 230000001086 cytosolic effect Effects 0.000 description 2
- 239000003814 drug Substances 0.000 description 2
- 239000013604 expression vector Substances 0.000 description 2
- 102000037865 fusion proteins Human genes 0.000 description 2
- 108020001507 fusion proteins Proteins 0.000 description 2
- 230000014509 gene expression Effects 0.000 description 2
- 230000013595 glycosylation Effects 0.000 description 2
- 238000006206 glycosylation reaction Methods 0.000 description 2
- VPZXBVLAVMBEQI-UHFFFAOYSA-N glycyl-DL-alpha-alanine Natural products OC(=O)C(C)NC(=O)CN VPZXBVLAVMBEQI-UHFFFAOYSA-N 0.000 description 2
- XKUKSGPZAADMRA-UHFFFAOYSA-N glycyl-glycyl-glycine Natural products NCC(=O)NCC(=O)NCC(O)=O XKUKSGPZAADMRA-UHFFFAOYSA-N 0.000 description 2
- 201000010536 head and neck cancer Diseases 0.000 description 2
- 208000014829 head and neck neoplasm Diseases 0.000 description 2
- 210000000987 immune system Anatomy 0.000 description 2
- 108091008042 inhibitory receptors Proteins 0.000 description 2
- 210000001165 lymph node Anatomy 0.000 description 2
- 238000005259 measurement Methods 0.000 description 2
- 230000002503 metabolic effect Effects 0.000 description 2
- 239000011859 microparticle Substances 0.000 description 2
- 238000004806 packaging method and process Methods 0.000 description 2
- 230000008569 process Effects 0.000 description 2
- 239000000047 product Substances 0.000 description 2
- 230000035755 proliferation Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 230000007017 scission Effects 0.000 description 2
- 238000013207 serial dilution Methods 0.000 description 2
- 230000009870 specific binding Effects 0.000 description 2
- 238000012289 standard assay Methods 0.000 description 2
- 238000003860 storage Methods 0.000 description 2
- 230000001225 therapeutic effect Effects 0.000 description 2
- 210000001519 tissue Anatomy 0.000 description 2
- 108010044292 tryptophyltyrosine Proteins 0.000 description 2
- 239000013598 vector Substances 0.000 description 2
- HDTRYLNUVZCQOY-UHFFFAOYSA-N α-D-glucopyranosyl-α-D-glucopyranoside Natural products OC1C(O)C(O)C(CO)OC1OC1C(O)C(O)C(O)C(CO)O1 HDTRYLNUVZCQOY-UHFFFAOYSA-N 0.000 description 1
- YYSWCHMLFJLLBJ-ZLUOBGJFSA-N Ala-Ala-Ser Chemical compound C[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](CO)C(O)=O YYSWCHMLFJLLBJ-ZLUOBGJFSA-N 0.000 description 1
- KQFRUSHJPKXBMB-BHDSKKPTSA-N Ala-Ala-Trp Chemical compound C1=CC=C2C(C[C@H](NC(=O)[C@H](C)NC(=O)[C@@H](N)C)C(O)=O)=CNC2=C1 KQFRUSHJPKXBMB-BHDSKKPTSA-N 0.000 description 1
- JBGSZRYCXBPWGX-BQBZGAKWSA-N Ala-Arg-Gly Chemical compound OC(=O)CNC(=O)[C@@H](NC(=O)[C@@H](N)C)CCCN=C(N)N JBGSZRYCXBPWGX-BQBZGAKWSA-N 0.000 description 1
- AWAXZRDKUHOPBO-GUBZILKMSA-N Ala-Gln-Lys Chemical compound C[C@H](N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCCCN)C(O)=O AWAXZRDKUHOPBO-GUBZILKMSA-N 0.000 description 1
- PAIHPOGPJVUFJY-WDSKDSINSA-N Ala-Glu-Gly Chemical compound C[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)NCC(O)=O PAIHPOGPJVUFJY-WDSKDSINSA-N 0.000 description 1
- BEMGNWZECGIJOI-WDSKDSINSA-N Ala-Gly-Glu Chemical compound [H]N[C@@H](C)C(=O)NCC(=O)N[C@@H](CCC(O)=O)C(O)=O BEMGNWZECGIJOI-WDSKDSINSA-N 0.000 description 1
- ARHJJAAWNWOACN-FXQIFTODSA-N Ala-Ser-Val Chemical compound [H]N[C@@H](C)C(=O)N[C@@H](CO)C(=O)N[C@@H](C(C)C)C(O)=O ARHJJAAWNWOACN-FXQIFTODSA-N 0.000 description 1
- CREYEAPXISDKSB-FQPOAREZSA-N Ala-Thr-Tyr Chemical compound [H]N[C@@H](C)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O CREYEAPXISDKSB-FQPOAREZSA-N 0.000 description 1
- VKKYFICVTYKFIO-CIUDSAMLSA-N Arg-Ala-Glu Chemical compound OC(=O)CC[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CCCN=C(N)N VKKYFICVTYKFIO-CIUDSAMLSA-N 0.000 description 1
- HKRXJBBCQBAGIM-FXQIFTODSA-N Arg-Asp-Ser Chemical compound C(C[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)N[C@@H](CO)C(=O)O)N)CN=C(N)N HKRXJBBCQBAGIM-FXQIFTODSA-N 0.000 description 1
- FEZJJKXNPSEYEV-CIUDSAMLSA-N Arg-Gln-Ala Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](C)C(O)=O FEZJJKXNPSEYEV-CIUDSAMLSA-N 0.000 description 1
- AOHKLEBWKMKITA-IHRRRGAJSA-N Arg-Phe-Ser Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)N[C@@H](CO)C(=O)O)NC(=O)[C@H](CCCN=C(N)N)N AOHKLEBWKMKITA-IHRRRGAJSA-N 0.000 description 1
- PRLPSDIHSRITSF-UNQGMJICSA-N Arg-Phe-Thr Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H]([C@@H](C)O)C(O)=O PRLPSDIHSRITSF-UNQGMJICSA-N 0.000 description 1
- ADPACBMPYWJJCE-FXQIFTODSA-N Arg-Ser-Asp Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(O)=O)C(O)=O ADPACBMPYWJJCE-FXQIFTODSA-N 0.000 description 1
- KMFPQTITXUKJOV-DCAQKATOSA-N Arg-Ser-Leu Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(O)=O KMFPQTITXUKJOV-DCAQKATOSA-N 0.000 description 1
- WCZXPVPHUMYLMS-VEVYYDQMSA-N Arg-Thr-Asp Chemical compound [H]N[C@@H](CCCNC(N)=N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(O)=O)C(O)=O WCZXPVPHUMYLMS-VEVYYDQMSA-N 0.000 description 1
- XYBJLTKSGFBLCS-QXEWZRGKSA-N Asp-Arg-Val Chemical compound NC(N)=NCCC[C@@H](C(=O)N[C@@H](C(C)C)C(O)=O)NC(=O)[C@@H](N)CC(O)=O XYBJLTKSGFBLCS-QXEWZRGKSA-N 0.000 description 1
- YNQIDCRRTWGHJD-ZLUOBGJFSA-N Asp-Asn-Ala Chemical compound OC(=O)[C@H](C)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CC(O)=O YNQIDCRRTWGHJD-ZLUOBGJFSA-N 0.000 description 1
- VHQOCWWKXIOAQI-WDSKDSINSA-N Asp-Gln-Gly Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CCC(N)=O)C(=O)NCC(O)=O VHQOCWWKXIOAQI-WDSKDSINSA-N 0.000 description 1
- VFUXXFVCYZPOQG-WDSKDSINSA-N Asp-Glu-Gly Chemical compound [H]N[C@@H](CC(O)=O)C(=O)N[C@@H](CCC(O)=O)C(=O)NCC(O)=O VFUXXFVCYZPOQG-WDSKDSINSA-N 0.000 description 1
- QNFRBNZGVVKBNJ-PEFMBERDSA-N Asp-Ile-Gln Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](CC(=O)O)N QNFRBNZGVVKBNJ-PEFMBERDSA-N 0.000 description 1
- GGRSYTUJHAZTFN-IHRRRGAJSA-N Asp-Pro-Tyr Chemical compound C1C[C@H](N(C1)C(=O)[C@H](CC(=O)O)N)C(=O)N[C@@H](CC2=CC=C(C=C2)O)C(=O)O GGRSYTUJHAZTFN-IHRRRGAJSA-N 0.000 description 1
- BRRPVTUFESPTCP-ACZMJKKPSA-N Asp-Ser-Glu Chemical compound OC(=O)C[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@H](C(O)=O)CCC(O)=O BRRPVTUFESPTCP-ACZMJKKPSA-N 0.000 description 1
- GYNUXDMCDILYIQ-QRTARXTBSA-N Asp-Val-Trp Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)O)NC(=O)[C@H](CC(=O)O)N GYNUXDMCDILYIQ-QRTARXTBSA-N 0.000 description 1
- 101150091609 CD274 gene Proteins 0.000 description 1
- 241000283707 Capra Species 0.000 description 1
- 206010009944 Colon cancer Diseases 0.000 description 1
- 208000001333 Colorectal Neoplasms Diseases 0.000 description 1
- YFXFOZPXVFPBDH-VZFHVOOUSA-N Cys-Ala-Thr Chemical compound C[C@@H](O)[C@H](NC(=O)[C@H](C)NC(=O)[C@@H](N)CS)C(O)=O YFXFOZPXVFPBDH-VZFHVOOUSA-N 0.000 description 1
- SZQCDCKIGWQAQN-FXQIFTODSA-N Cys-Arg-Ala Chemical compound [H]N[C@@H](CS)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](C)C(O)=O SZQCDCKIGWQAQN-FXQIFTODSA-N 0.000 description 1
- FBPFZTCFMRRESA-FSIIMWSLSA-N D-Glucitol Natural products OC[C@H](O)[C@H](O)[C@@H](O)[C@H](O)CO FBPFZTCFMRRESA-FSIIMWSLSA-N 0.000 description 1
- FBPFZTCFMRRESA-KVTDHHQDSA-N D-Mannitol Chemical compound OC[C@@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-KVTDHHQDSA-N 0.000 description 1
- FBPFZTCFMRRESA-JGWLITMVSA-N D-glucitol Chemical compound OC[C@H](O)[C@@H](O)[C@H](O)[C@H](O)CO FBPFZTCFMRRESA-JGWLITMVSA-N 0.000 description 1
- 238000002965 ELISA Methods 0.000 description 1
- 102000004190 Enzymes Human genes 0.000 description 1
- 108090000790 Enzymes Proteins 0.000 description 1
- 108700024394 Exon Proteins 0.000 description 1
- 238000012413 Fluorescence activated cell sorting analysis Methods 0.000 description 1
- OYTPNWYZORARHL-XHNCKOQMSA-N Gln-Ala-Pro Chemical compound C[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CCC(=O)N)N OYTPNWYZORARHL-XHNCKOQMSA-N 0.000 description 1
- IKFZXRLDMYWNBU-YUMQZZPRSA-N Gln-Gly-Arg Chemical compound NC(=O)CC[C@H](N)C(=O)NCC(=O)N[C@H](C(O)=O)CCCN=C(N)N IKFZXRLDMYWNBU-YUMQZZPRSA-N 0.000 description 1
- FQCILXROGNOZON-YUMQZZPRSA-N Gln-Pro-Gly Chemical compound NC(=O)CC[C@H](N)C(=O)N1CCC[C@H]1C(=O)NCC(O)=O FQCILXROGNOZON-YUMQZZPRSA-N 0.000 description 1
- CGYDXNKRIMJMLV-GUBZILKMSA-N Glu-Arg-Glu Chemical compound [H]N[C@@H](CCC(O)=O)C(=O)N[C@@H](CCCNC(N)=N)C(=O)N[C@@H](CCC(O)=O)C(O)=O CGYDXNKRIMJMLV-GUBZILKMSA-N 0.000 description 1
- PAQUJCSYVIBPLC-AVGNSLFASA-N Glu-Asp-Phe Chemical compound OC(=O)CC[C@H](N)C(=O)N[C@@H](CC(O)=O)C(=O)N[C@H](C(O)=O)CC1=CC=CC=C1 PAQUJCSYVIBPLC-AVGNSLFASA-N 0.000 description 1
- ZYRXTRTUCAVNBQ-GVXVVHGQSA-N Glu-Val-Lys Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CCC(=O)O)N ZYRXTRTUCAVNBQ-GVXVVHGQSA-N 0.000 description 1
- YMUFWNJHVPQNQD-ZKWXMUAHSA-N Gly-Ala-Ile Chemical compound CC[C@H](C)[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)CN YMUFWNJHVPQNQD-ZKWXMUAHSA-N 0.000 description 1
- UQJNXZSSGQIPIQ-FBCQKBJTSA-N Gly-Gly-Thr Chemical compound C[C@@H](O)[C@@H](C(O)=O)NC(=O)CNC(=O)CN UQJNXZSSGQIPIQ-FBCQKBJTSA-N 0.000 description 1
- MIIVFRCYJABHTQ-ONGXEEELSA-N Gly-Leu-Val Chemical compound [H]NCC(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(O)=O MIIVFRCYJABHTQ-ONGXEEELSA-N 0.000 description 1
- IMRNSEPSPFQNHF-STQMWFEESA-N Gly-Ser-Trp Chemical compound NCC(=O)N[C@@H](CO)C(=O)N[C@@H](CC1=CNC2=CC=CC=C12)C(=O)O IMRNSEPSPFQNHF-STQMWFEESA-N 0.000 description 1
- NVTPVQLIZCOJFK-FOHZUACHSA-N Gly-Thr-Asp Chemical compound [H]NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(O)=O)C(O)=O NVTPVQLIZCOJFK-FOHZUACHSA-N 0.000 description 1
- ZZWUYQXMIFTIIY-WEDXCCLWSA-N Gly-Thr-Leu Chemical compound [H]NCC(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(O)=O ZZWUYQXMIFTIIY-WEDXCCLWSA-N 0.000 description 1
- MYXNLWDWWOTERK-BHNWBGBOSA-N Gly-Thr-Pro Chemical compound C[C@H]([C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)CN)O MYXNLWDWWOTERK-BHNWBGBOSA-N 0.000 description 1
- UMRIXLHPZZIOML-OALUTQOASA-N Gly-Trp-Phe Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)O)NC(=O)[C@H](CC2=CNC3=CC=CC=C32)NC(=O)CN UMRIXLHPZZIOML-OALUTQOASA-N 0.000 description 1
- GNNJKUYDWFIBTK-QWRGUYRKSA-N Gly-Tyr-Asp Chemical compound [H]NCC(=O)N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CC(O)=O)C(O)=O GNNJKUYDWFIBTK-QWRGUYRKSA-N 0.000 description 1
- 102000003886 Glycoproteins Human genes 0.000 description 1
- 108090000288 Glycoproteins Proteins 0.000 description 1
- 208000002250 Hematologic Neoplasms Diseases 0.000 description 1
- MWWOPNQSBXEUHO-ULQDDVLXSA-N His-Arg-Tyr Chemical compound C([C@H](N)C(=O)N[C@@H](CCCN=C(N)N)C(=O)N[C@@H](CC=1C=CC(O)=CC=1)C(O)=O)C1=CN=CN1 MWWOPNQSBXEUHO-ULQDDVLXSA-N 0.000 description 1
- 101100407305 Homo sapiens CD274 gene Proteins 0.000 description 1
- OTSVBELRDMSPKY-PCBIJLKTSA-N Ile-Phe-Asn Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CC(=O)N)C(=O)O)N OTSVBELRDMSPKY-PCBIJLKTSA-N 0.000 description 1
- JHNJNTMTZHEDLJ-NAKRPEOUSA-N Ile-Ser-Arg Chemical compound CC[C@H](C)[C@H](N)C(=O)N[C@@H](CO)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O JHNJNTMTZHEDLJ-NAKRPEOUSA-N 0.000 description 1
- ZGKVPOSSTGHJAF-HJPIBITLSA-N Ile-Tyr-Ser Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC1=CC=C(C=C1)O)C(=O)N[C@@H](CO)C(=O)O)N ZGKVPOSSTGHJAF-HJPIBITLSA-N 0.000 description 1
- 102000037982 Immune checkpoint proteins Human genes 0.000 description 1
- 108091008036 Immune checkpoint proteins Proteins 0.000 description 1
- 108060003951 Immunoglobulin Proteins 0.000 description 1
- 102100037850 Interferon gamma Human genes 0.000 description 1
- 108010074328 Interferon-gamma Proteins 0.000 description 1
- 102000002698 KIR Receptors Human genes 0.000 description 1
- 108010043610 KIR Receptors Proteins 0.000 description 1
- PMGDADKJMCOXHX-UHFFFAOYSA-N L-Arginyl-L-glutamin-acetat Natural products NC(=N)NCCCC(N)C(=O)NC(CCC(N)=O)C(O)=O PMGDADKJMCOXHX-UHFFFAOYSA-N 0.000 description 1
- TYYLDKGBCJGJGW-UHFFFAOYSA-N L-tryptophan-L-tyrosine Natural products C=1NC2=CC=CC=C2C=1CC(N)C(=O)NC(C(O)=O)CC1=CC=C(O)C=C1 TYYLDKGBCJGJGW-UHFFFAOYSA-N 0.000 description 1
- CQGSYZCULZMEDE-UHFFFAOYSA-N Leu-Gln-Pro Natural products CC(C)CC(N)C(=O)NC(CCC(N)=O)C(=O)N1CCCC1C(O)=O CQGSYZCULZMEDE-UHFFFAOYSA-N 0.000 description 1
- FEHQLKKBVJHSEC-SZMVWBNQSA-N Leu-Glu-Trp Chemical compound C1=CC=C2C(C[C@H](NC(=O)[C@H](CCC(O)=O)NC(=O)[C@@H](N)CC(C)C)C(O)=O)=CNC2=C1 FEHQLKKBVJHSEC-SZMVWBNQSA-N 0.000 description 1
- KIZIOFNVSOSKJI-CIUDSAMLSA-N Leu-Ser-Cys Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)O)N KIZIOFNVSOSKJI-CIUDSAMLSA-N 0.000 description 1
- MVJRBCJCRYGCKV-GVXVVHGQSA-N Leu-Val-Gln Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(N)=O)C(O)=O MVJRBCJCRYGCKV-GVXVVHGQSA-N 0.000 description 1
- AIMGJYMCTAABEN-GVXVVHGQSA-N Leu-Val-Glu Chemical compound [H]N[C@@H](CC(C)C)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(O)=O AIMGJYMCTAABEN-GVXVVHGQSA-N 0.000 description 1
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 1
- IXHKPDJKKCUKHS-GARJFASQSA-N Lys-Ala-Pro Chemical compound C[C@@H](C(=O)N1CCC[C@@H]1C(=O)O)NC(=O)[C@H](CCCCN)N IXHKPDJKKCUKHS-GARJFASQSA-N 0.000 description 1
- UWKNTTJNVSYXPC-CIUDSAMLSA-N Lys-Ala-Ser Chemical compound OC[C@@H](C(O)=O)NC(=O)[C@H](C)NC(=O)[C@@H](N)CCCCN UWKNTTJNVSYXPC-CIUDSAMLSA-N 0.000 description 1
- QUCDKEKDPYISNX-HJGDQZAQSA-N Lys-Asn-Thr Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H]([C@@H](C)O)C(O)=O QUCDKEKDPYISNX-HJGDQZAQSA-N 0.000 description 1
- AIRZWUMAHCDDHR-KKUMJFAQSA-N Lys-Leu-Leu Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC(C)C)C(O)=O AIRZWUMAHCDDHR-KKUMJFAQSA-N 0.000 description 1
- RPWTZTBIFGENIA-VOAKCMCISA-N Lys-Thr-Leu Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(O)=O RPWTZTBIFGENIA-VOAKCMCISA-N 0.000 description 1
- UGCIQUYEJIEHKX-GVXVVHGQSA-N Lys-Val-Glu Chemical compound [H]N[C@@H](CCCCN)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCC(O)=O)C(O)=O UGCIQUYEJIEHKX-GVXVVHGQSA-N 0.000 description 1
- 229930195725 Mannitol Natural products 0.000 description 1
- CAODKDAPYGUMLK-FXQIFTODSA-N Met-Asn-Ser Chemical compound CSCC[C@H](N)C(=O)N[C@@H](CC(N)=O)C(=O)N[C@@H](CO)C(O)=O CAODKDAPYGUMLK-FXQIFTODSA-N 0.000 description 1
- SJDQOYTYNGZZJX-SRVKXCTJSA-N Met-Glu-Leu Chemical compound CSCC[C@H](N)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(C)C)C(O)=O SJDQOYTYNGZZJX-SRVKXCTJSA-N 0.000 description 1
- SPSSJSICDYYTQN-HJGDQZAQSA-N Met-Thr-Gln Chemical compound CSCC[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@H](C(O)=O)CCC(N)=O SPSSJSICDYYTQN-HJGDQZAQSA-N 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 102000018656 Mitogen Receptors Human genes 0.000 description 1
- 108010052006 Mitogen Receptors Proteins 0.000 description 1
- 101150110881 NKG2A gene Proteins 0.000 description 1
- 108091005461 Nucleic proteins Chemical group 0.000 description 1
- 102000057297 Pepsin A Human genes 0.000 description 1
- 108090000284 Pepsin A Proteins 0.000 description 1
- MRWOVVNKSXXLRP-IHPCNDPISA-N Phe-Ser-Trp Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H](CO)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(O)=O MRWOVVNKSXXLRP-IHPCNDPISA-N 0.000 description 1
- KLYYKKGCPOGDPE-OEAJRASXSA-N Phe-Thr-Leu Chemical compound [H]N[C@@H](CC1=CC=CC=C1)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(O)=O KLYYKKGCPOGDPE-OEAJRASXSA-N 0.000 description 1
- UIMCLYYSUCIUJM-UWVGGRQHSA-N Pro-Gly-Lys Chemical compound NCCCC[C@@H](C(O)=O)NC(=O)CNC(=O)[C@@H]1CCCN1 UIMCLYYSUCIUJM-UWVGGRQHSA-N 0.000 description 1
- 206010060862 Prostate cancer Diseases 0.000 description 1
- 208000000236 Prostatic Neoplasms Diseases 0.000 description 1
- UCOYFSCEIWQYNL-FXQIFTODSA-N Ser-Cys-Met Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCSC)C(O)=O UCOYFSCEIWQYNL-FXQIFTODSA-N 0.000 description 1
- SQBLRDDJTUJDMV-ACZMJKKPSA-N Ser-Glu-Asn Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CCC(O)=O)C(=O)N[C@@H](CC(N)=O)C(O)=O SQBLRDDJTUJDMV-ACZMJKKPSA-N 0.000 description 1
- UQFYNFTYDHUIMI-WHFBIAKZSA-N Ser-Gly-Ala Chemical compound OC(=O)[C@H](C)NC(=O)CNC(=O)[C@@H](N)CO UQFYNFTYDHUIMI-WHFBIAKZSA-N 0.000 description 1
- YMTLKLXDFCSCNX-BYPYZUCNSA-N Ser-Gly-Gly Chemical compound OC[C@H](N)C(=O)NCC(=O)NCC(O)=O YMTLKLXDFCSCNX-BYPYZUCNSA-N 0.000 description 1
- UIGMAMGZOJVTDN-WHFBIAKZSA-N Ser-Gly-Ser Chemical compound OC[C@H](N)C(=O)NCC(=O)N[C@@H](CO)C(O)=O UIGMAMGZOJVTDN-WHFBIAKZSA-N 0.000 description 1
- QYSFWUIXDFJUDW-DCAQKATOSA-N Ser-Leu-Arg Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CCCNC(N)=N)C(O)=O QYSFWUIXDFJUDW-DCAQKATOSA-N 0.000 description 1
- GJFYFGOEWLDQGW-GUBZILKMSA-N Ser-Leu-Gln Chemical compound CC(C)C[C@@H](C(=O)N[C@@H](CCC(=O)N)C(=O)O)NC(=O)[C@H](CO)N GJFYFGOEWLDQGW-GUBZILKMSA-N 0.000 description 1
- YUJLIIRMIAGMCQ-CIUDSAMLSA-N Ser-Leu-Ser Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CO)C(O)=O YUJLIIRMIAGMCQ-CIUDSAMLSA-N 0.000 description 1
- AZWNCEBQZXELEZ-FXQIFTODSA-N Ser-Pro-Ser Chemical compound OC[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(O)=O AZWNCEBQZXELEZ-FXQIFTODSA-N 0.000 description 1
- XJDMUQCLVSCRSJ-VZFHVOOUSA-N Ser-Thr-Ala Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C)C(O)=O XJDMUQCLVSCRSJ-VZFHVOOUSA-N 0.000 description 1
- BCAVNDNYOGTQMQ-AAEUAGOBSA-N Ser-Trp-Gly Chemical compound [H]N[C@@H](CO)C(=O)N[C@@H](CC1=CNC2=C1C=CC=C2)C(=O)NCC(O)=O BCAVNDNYOGTQMQ-AAEUAGOBSA-N 0.000 description 1
- GSCVDSBEYVGMJQ-SRVKXCTJSA-N Ser-Tyr-Asp Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)N[C@@H](CC(=O)O)C(=O)O)NC(=O)[C@H](CO)N)O GSCVDSBEYVGMJQ-SRVKXCTJSA-N 0.000 description 1
- LGIMRDKGABDMBN-DCAQKATOSA-N Ser-Val-Lys Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CCCCN)C(=O)O)NC(=O)[C@H](CO)N LGIMRDKGABDMBN-DCAQKATOSA-N 0.000 description 1
- 108010003723 Single-Domain Antibodies Proteins 0.000 description 1
- 210000000662 T-lymphocyte subset Anatomy 0.000 description 1
- YDWLCDQXLCILCZ-BWAGICSOSA-N Thr-His-Tyr Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC1=CNC=N1)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O YDWLCDQXLCILCZ-BWAGICSOSA-N 0.000 description 1
- FQPDRTDDEZXCEC-SVSWQMSJSA-N Thr-Ile-Ser Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H](CO)C(O)=O FQPDRTDDEZXCEC-SVSWQMSJSA-N 0.000 description 1
- GXUWHVZYDAHFSV-FLBSBUHZSA-N Thr-Ile-Thr Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]([C@@H](C)O)C(O)=O GXUWHVZYDAHFSV-FLBSBUHZSA-N 0.000 description 1
- IJVNLNRVDUTWDD-MEYUZBJRSA-N Thr-Leu-Tyr Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CC(C)C)C(=O)N[C@@H](CC1=CC=C(O)C=C1)C(O)=O IJVNLNRVDUTWDD-MEYUZBJRSA-N 0.000 description 1
- BIBYEFRASCNLAA-CDMKHQONSA-N Thr-Phe-Gly Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@H](C(=O)NCC(O)=O)CC1=CC=CC=C1 BIBYEFRASCNLAA-CDMKHQONSA-N 0.000 description 1
- RVMNUBQWPVOUKH-HEIBUPTGSA-N Thr-Ser-Thr Chemical compound [H]N[C@@H]([C@@H](C)O)C(=O)N[C@@H](CO)C(=O)N[C@@H]([C@@H](C)O)C(O)=O RVMNUBQWPVOUKH-HEIBUPTGSA-N 0.000 description 1
- YRJOLUDFVAUXLI-GSSVUCPTSA-N Thr-Thr-Asp Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@H](C(O)=O)CC(O)=O YRJOLUDFVAUXLI-GSSVUCPTSA-N 0.000 description 1
- LECUEEHKUFYOOV-ZJDVBMNYSA-N Thr-Thr-Val Chemical compound CC(C)[C@@H](C(O)=O)NC(=O)[C@H]([C@@H](C)O)NC(=O)[C@@H](N)[C@@H](C)O LECUEEHKUFYOOV-ZJDVBMNYSA-N 0.000 description 1
- MNYNCKZAEIAONY-XGEHTFHBSA-N Thr-Val-Ser Chemical compound C[C@@H](O)[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CO)C(O)=O MNYNCKZAEIAONY-XGEHTFHBSA-N 0.000 description 1
- HDTRYLNUVZCQOY-WSWWMNSNSA-N Trehalose Natural products O[C@@H]1[C@@H](O)[C@@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-WSWWMNSNSA-N 0.000 description 1
- AZBIIKDSDLVJAK-VHWLVUOQSA-N Trp-Ile-Asn Chemical compound CC[C@H](C)[C@@H](C(=O)N[C@@H](CC(=O)N)C(=O)O)NC(=O)[C@H](CC1=CNC2=CC=CC=C21)N AZBIIKDSDLVJAK-VHWLVUOQSA-N 0.000 description 1
- BOMYCJXTWRMKJA-RNXOBYDBSA-N Trp-Phe-Phe Chemical compound C1=CC=C(C=C1)C[C@@H](C(=O)N[C@@H](CC2=CC=CC=C2)C(=O)O)NC(=O)[C@H](CC3=CNC4=CC=CC=C43)N BOMYCJXTWRMKJA-RNXOBYDBSA-N 0.000 description 1
- MBLJBGZWLHTJBH-SZMVWBNQSA-N Trp-Val-Arg Chemical compound C1=CC=C2C(C[C@H](N)C(=O)N[C@@H](C(C)C)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O)=CNC2=C1 MBLJBGZWLHTJBH-SZMVWBNQSA-N 0.000 description 1
- YJQCOFNZVFGCAF-UHFFFAOYSA-N Tunicamycin II Natural products O1C(CC(O)C2C(C(O)C(O2)N2C(NC(=O)C=C2)=O)O)C(O)C(O)C(NC(=O)C=CCCCCCCCCC(C)C)C1OC1OC(CO)C(O)C(O)C1NC(C)=O YJQCOFNZVFGCAF-UHFFFAOYSA-N 0.000 description 1
- 108091005956 Type II transmembrane proteins Proteins 0.000 description 1
- CKKFTIQYURNSEI-IHRRRGAJSA-N Tyr-Asn-Arg Chemical compound NC(N)=NCCC[C@@H](C(O)=O)NC(=O)[C@H](CC(N)=O)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 CKKFTIQYURNSEI-IHRRRGAJSA-N 0.000 description 1
- QOIKZODVIPOPDD-AVGNSLFASA-N Tyr-Cys-Gln Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CS)C(=O)N[C@@H](CCC(N)=O)C(O)=O QOIKZODVIPOPDD-AVGNSLFASA-N 0.000 description 1
- QUILOGWWLXMSAT-IHRRRGAJSA-N Tyr-Gln-Gln Chemical compound [H]N[C@@H](CC1=CC=C(O)C=C1)C(=O)N[C@@H](CCC(N)=O)C(=O)N[C@@H](CCC(N)=O)C(O)=O QUILOGWWLXMSAT-IHRRRGAJSA-N 0.000 description 1
- AKLNEFNQWLHIGY-QWRGUYRKSA-N Tyr-Gly-Asp Chemical compound C1=CC(=CC=C1C[C@@H](C(=O)NCC(=O)N[C@@H](CC(=O)O)C(=O)O)N)O AKLNEFNQWLHIGY-QWRGUYRKSA-N 0.000 description 1
- NKUGCYDFQKFVOJ-JYJNAYRXSA-N Tyr-Leu-Gln Chemical compound NC(=O)CC[C@@H](C(O)=O)NC(=O)[C@H](CC(C)C)NC(=O)[C@@H](N)CC1=CC=C(O)C=C1 NKUGCYDFQKFVOJ-JYJNAYRXSA-N 0.000 description 1
- DDRBQONWVBDQOY-GUBZILKMSA-N Val-Ala-Arg Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](C)C(=O)N[C@@H](CCCN=C(N)N)C(O)=O DDRBQONWVBDQOY-GUBZILKMSA-N 0.000 description 1
- LTFLDDDGWOVIHY-NAKRPEOUSA-N Val-Ala-Ile Chemical compound CC[C@H](C)[C@@H](C(=O)O)NC(=O)[C@H](C)NC(=O)[C@H](C(C)C)N LTFLDDDGWOVIHY-NAKRPEOUSA-N 0.000 description 1
- DIOSYUIWOQCXNR-ONGXEEELSA-N Val-Lys-Gly Chemical compound CC(C)[C@H](N)C(=O)N[C@@H](CCCCN)C(=O)NCC(O)=O DIOSYUIWOQCXNR-ONGXEEELSA-N 0.000 description 1
- SSYBNWFXCFNRFN-GUBZILKMSA-N Val-Pro-Ser Chemical compound CC(C)[C@H](N)C(=O)N1CCC[C@H]1C(=O)N[C@@H](CO)C(O)=O SSYBNWFXCFNRFN-GUBZILKMSA-N 0.000 description 1
- JQTYTBPCSOAZHI-FXQIFTODSA-N Val-Ser-Cys Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CO)C(=O)N[C@@H](CS)C(=O)O)N JQTYTBPCSOAZHI-FXQIFTODSA-N 0.000 description 1
- USXYVSTVPHELAF-RCWTZXSCSA-N Val-Thr-Met Chemical compound C[C@H]([C@@H](C(=O)N[C@@H](CCSC)C(=O)O)NC(=O)[C@H](C(C)C)N)O USXYVSTVPHELAF-RCWTZXSCSA-N 0.000 description 1
- HTONZBWRYUKUKC-RCWTZXSCSA-N Val-Thr-Val Chemical compound CC(C)[C@H](N)C(=O)N[C@@H]([C@@H](C)O)C(=O)N[C@@H](C(C)C)C(O)=O HTONZBWRYUKUKC-RCWTZXSCSA-N 0.000 description 1
- RSEIVHMDTNNEOW-JYJNAYRXSA-N Val-Trp-Cys Chemical compound CC(C)[C@@H](C(=O)N[C@@H](CC1=CNC2=CC=CC=C21)C(=O)N[C@@H](CS)C(=O)O)N RSEIVHMDTNNEOW-JYJNAYRXSA-N 0.000 description 1
- 241000700605 Viruses Species 0.000 description 1
- 239000008351 acetate buffer Substances 0.000 description 1
- 238000001467 acupuncture Methods 0.000 description 1
- 108010008685 alanyl-glutamyl-aspartic acid Proteins 0.000 description 1
- 230000000735 allogeneic effect Effects 0.000 description 1
- HDTRYLNUVZCQOY-LIZSDCNHSA-N alpha,alpha-trehalose Chemical compound O[C@@H]1[C@@H](O)[C@H](O)[C@@H](CO)O[C@@H]1O[C@@H]1[C@H](O)[C@@H](O)[C@H](O)[C@@H](CO)O1 HDTRYLNUVZCQOY-LIZSDCNHSA-N 0.000 description 1
- 230000009435 amidation Effects 0.000 description 1
- 238000007112 amidation reaction Methods 0.000 description 1
- 230000000259 anti-tumor effect Effects 0.000 description 1
- 108010008355 arginyl-glutamine Proteins 0.000 description 1
- 108010062796 arginyllysine Proteins 0.000 description 1
- 108010040443 aspartyl-aspartic acid Proteins 0.000 description 1
- 210000003719 b-lymphocyte Anatomy 0.000 description 1
- 230000015572 biosynthetic process Effects 0.000 description 1
- 239000006172 buffering agent Substances 0.000 description 1
- 230000015556 catabolic process Effects 0.000 description 1
- 239000006285 cell suspension Substances 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 230000003196 chaotropic effect Effects 0.000 description 1
- 238000012412 chemical coupling Methods 0.000 description 1
- 238000007385 chemical modification Methods 0.000 description 1
- 239000003795 chemical substances by application Substances 0.000 description 1
- 238000012875 competitive assay Methods 0.000 description 1
- 230000004154 complement system Effects 0.000 description 1
- 239000002299 complementary DNA Substances 0.000 description 1
- 230000009918 complex formation Effects 0.000 description 1
- 238000007796 conventional method Methods 0.000 description 1
- 238000005520 cutting process Methods 0.000 description 1
- XUJNEKJLAYXESH-UHFFFAOYSA-N cysteine Natural products SCC(N)C(O)=O XUJNEKJLAYXESH-UHFFFAOYSA-N 0.000 description 1
- 150000001945 cysteines Chemical class 0.000 description 1
- 108010016616 cysteinylglycine Proteins 0.000 description 1
- 210000001151 cytotoxic T lymphocyte Anatomy 0.000 description 1
- 230000006240 deamidation Effects 0.000 description 1
- 239000007857 degradation product Substances 0.000 description 1
- 238000006731 degradation reaction Methods 0.000 description 1
- 230000036425 denaturation Effects 0.000 description 1
- 238000004925 denaturation Methods 0.000 description 1
- 230000001419 dependent effect Effects 0.000 description 1
- 238000001212 derivatisation Methods 0.000 description 1
- 230000001627 detrimental effect Effects 0.000 description 1
- 230000004069 differentiation Effects 0.000 description 1
- 230000029087 digestion Effects 0.000 description 1
- 238000004090 dissolution Methods 0.000 description 1
- 239000003937 drug carrier Substances 0.000 description 1
- 230000009977 dual effect Effects 0.000 description 1
- 238000005516 engineering process Methods 0.000 description 1
- 229940088598 enzyme Drugs 0.000 description 1
- 230000017188 evasion or tolerance of host immune response Effects 0.000 description 1
- 238000001943 fluorescence-activated cell sorting Methods 0.000 description 1
- 230000022244 formylation Effects 0.000 description 1
- 238000006170 formylation reaction Methods 0.000 description 1
- 238000013467 fragmentation Methods 0.000 description 1
- 238000006062 fragmentation reaction Methods 0.000 description 1
- 108010090037 glycyl-alanyl-isoleucine Proteins 0.000 description 1
- 108010067216 glycyl-glycyl-glycine Proteins 0.000 description 1
- 108010010096 glycyl-glycyl-tyrosine Proteins 0.000 description 1
- 108010089804 glycyl-threonine Proteins 0.000 description 1
- 108010015792 glycyllysine Proteins 0.000 description 1
- 108010037850 glycylvaline Proteins 0.000 description 1
- 230000005931 immune cell recruitment Effects 0.000 description 1
- 230000001900 immune effect Effects 0.000 description 1
- 230000036737 immune function Effects 0.000 description 1
- 108091008915 immune receptors Proteins 0.000 description 1
- 102000027596 immune receptors Human genes 0.000 description 1
- 230000006058 immune tolerance Effects 0.000 description 1
- 102000018358 immunoglobulin Human genes 0.000 description 1
- 238000007689 inspection Methods 0.000 description 1
- 238000006317 isomerization reaction Methods 0.000 description 1
- 230000002147 killing effect Effects 0.000 description 1
- 238000002372 labelling Methods 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 230000005923 long-lasting effect Effects 0.000 description 1
- 201000005202 lung cancer Diseases 0.000 description 1
- 208000020816 lung neoplasm Diseases 0.000 description 1
- 108010009298 lysylglutamic acid Proteins 0.000 description 1
- 229920002521 macromolecule Polymers 0.000 description 1
- 210000004962 mammalian cell Anatomy 0.000 description 1
- 239000000594 mannitol Substances 0.000 description 1
- 235000010355 mannitol Nutrition 0.000 description 1
- 201000001441 melanoma Diseases 0.000 description 1
- 239000012528 membrane Substances 0.000 description 1
- 108010005942 methionylglycine Proteins 0.000 description 1
- 230000004001 molecular interaction Effects 0.000 description 1
- 239000013642 negative control Substances 0.000 description 1
- 108020004707 nucleic acids Proteins 0.000 description 1
- 102000039446 nucleic acids Human genes 0.000 description 1
- 150000007523 nucleic acids Chemical group 0.000 description 1
- 239000002773 nucleotide Substances 0.000 description 1
- 125000003729 nucleotide group Chemical group 0.000 description 1
- 230000010355 oscillation Effects 0.000 description 1
- 230000003204 osmotic effect Effects 0.000 description 1
- 230000003647 oxidation Effects 0.000 description 1
- 238000007254 oxidation reaction Methods 0.000 description 1
- 230000006320 pegylation Effects 0.000 description 1
- 229940111202 pepsin Drugs 0.000 description 1
- 108010070409 phenylalanyl-glycyl-glycine Proteins 0.000 description 1
- 230000026731 phosphorylation Effects 0.000 description 1
- 238000006366 phosphorylation reaction Methods 0.000 description 1
- 229920001184 polypeptide Polymers 0.000 description 1
- 229950008882 polysorbate Drugs 0.000 description 1
- 230000008092 positive effect Effects 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000001556 precipitation Methods 0.000 description 1
- 125000002924 primary amino group Chemical group [H]N([H])* 0.000 description 1
- 102000004196 processed proteins & peptides Human genes 0.000 description 1
- 108090000765 processed proteins & peptides Proteins 0.000 description 1
- 108010020755 prolyl-glycyl-glycine Proteins 0.000 description 1
- 108010004914 prolylarginine Proteins 0.000 description 1
- 230000006337 proteolytic cleavage Effects 0.000 description 1
- 238000003127 radioimmunoassay Methods 0.000 description 1
- 238000010188 recombinant method Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 230000008261 resistance mechanism Effects 0.000 description 1
- 230000000717 retained effect Effects 0.000 description 1
- 238000012216 screening Methods 0.000 description 1
- 238000007423 screening assay Methods 0.000 description 1
- 230000019491 signal transduction Effects 0.000 description 1
- 239000000600 sorbitol Substances 0.000 description 1
- 210000000952 spleen Anatomy 0.000 description 1
- 230000000087 stabilizing effect Effects 0.000 description 1
- 238000011476 stem cell transplantation Methods 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 230000002195 synergetic effect Effects 0.000 description 1
- 238000003786 synthesis reaction Methods 0.000 description 1
- 230000008685 targeting Effects 0.000 description 1
- 238000010257 thawing Methods 0.000 description 1
- 108010033670 threonyl-aspartyl-tyrosine Proteins 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 108091005703 transmembrane proteins Proteins 0.000 description 1
- 102000035160 transmembrane proteins Human genes 0.000 description 1
- ZHSGGJXRNHWHRS-VIDYELAYSA-N tunicamycin Chemical compound O([C@H]1[C@@H]([C@H]([C@@H](O)[C@@H](CC(O)[C@@H]2[C@H]([C@@H](O)[C@@H](O2)N2C(NC(=O)C=C2)=O)O)O1)O)NC(=O)/C=C/CC(C)C)[C@H]1O[C@H](CO)[C@@H](O)[C@H](O)[C@H]1NC(C)=O ZHSGGJXRNHWHRS-VIDYELAYSA-N 0.000 description 1
- MEYZYGMYMLNUHJ-UHFFFAOYSA-N tunicamycin Natural products CC(C)CCCCCCCCCC=CC(=O)NC1C(O)C(O)C(CC(O)C2OC(C(O)C2O)N3C=CC(=O)NC3=O)OC1OC4OC(CO)C(O)C(O)C4NC(=O)C MEYZYGMYMLNUHJ-UHFFFAOYSA-N 0.000 description 1
- 108010003137 tyrosyltyrosine Proteins 0.000 description 1
- 238000011179 visual inspection Methods 0.000 description 1
- 238000001262 western blot Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/08—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite containing oxygen, e.g. ethers, acetals, ketones, quinones, aldehydes, peroxides
- A61K47/12—Carboxylic acids; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/06—Organic compounds, e.g. natural or synthetic hydrocarbons, polyolefins, mineral oil, petrolatum or ozokerite
- A61K47/26—Carbohydrates, e.g. sugar alcohols, amino sugars, nucleic acids, mono-, di- or oligo-saccharides; Derivatives thereof, e.g. polysorbates, sorbitan fatty acid esters or glycyrrhizin
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2827—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against B7 molecules, e.g. CD80, CD86
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/30—Immunoglobulins specific features characterized by aspects of specificity or valency
- C07K2317/31—Immunoglobulins specific features characterized by aspects of specificity or valency multispecific
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Organic Chemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Molecular Biology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Engineering & Computer Science (AREA)
- Oil, Petroleum & Natural Gas (AREA)
- Epidemiology (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Peptides Or Proteins (AREA)
Abstract
The invention optimizes the components of the bispecific antibody stability preparation aiming at the difference of the molecular structure of the bispecific antibody and the natural antibody and based on the indexes of physical state, microstructure and biological activity. Taking humanized recombinant anti-NKG 2A and PD-L1 bispecific antibody as an example, the optimized stability preparation takes acetic acid buffer solution or citric acid buffer solution with pH of 5.5-6.5 as a buffer system, takes 4% sucrose as a stabilizer, and is added with 0.005-0.08% Tween-80. The detection results of high temperature test, shaking test, repeated freeze-thaw test and illumination test show that the stable preparation of the invention can maintain the appearance and physical state of the injection and maintain the structural stability and biological activity of the bispecific antibody molecule.
Description
Technical Field
The invention relates to the field of antibody medicaments, in particular to a stable preparation suitable for a bispecific antibody, and particularly relates to a hydro-acupuncture preparation of a humanized recombinant anti-NKG 2A and PD-L1 bispecific antibody and a preparation method thereof.
Background
In recent years, tumor immunotherapy has been unsuccessful, and only a few patients have shown long-lasting efficacy. Improving clinical response and overcoming resistance mechanisms are ongoing challenges in the field of tumor immunotherapy, and blocking other inhibitory immune receptors may be a viable strategy.
NKG2A (killer cell receptor C1), also known as KLRC1 or CD159A, is a type II transmembrane protein belonging to the NKG2/CD94 natural killer cell lectin receptor family. NKG2A has a Carbohydrate-recognizing domain CRD (Carbohydrate-recognition domain, usually consisting of 115-130 amino acids, containing 2-3 disulfide bonds, having 2-3N-linked glycosylation sites, the process of ligand recognition is often Ca2+ -dependent) in the extracellular domain. NKG2A is mainly expressed in NK cells, NKT cells and T cells; the relative molecular mass is 43000, which is composed of 233 amino acids, and the extracellular region has 135 amino acids. NKG2A inhibits immune cell activation through interaction with its ligand HLA-E. HLA-E is widely expressed on the surface of various tumor cells such as head and neck cancer, lung cancer, prostate cancer, colorectal cancer and the like, and IFN-gamma released by immune cells can further up-regulate the expression of HLA-E (J Clin invest, 2019; 129(5): 2094-2106). Like other receptor-ligands (such as PD-1/PD-L1), NKG2A/HLA-E is also an important signal path for tumor immune escape for immune checkpoints, and blocking the interaction between NKG2A/HLA-E becomes a very potential target in the field of tumor immunotherapy. Monoclonal antibodies against NKG2A are currently developed by several companies (e.g., Innate Pharma/Novo Nordisk/AstraZeneca and ChemPartner) to kill tumor cells by blocking the NKG2A/HLA-E interaction to increase the immunocompetence of NK cells as well as T cells.
Recent research shows that NKG2A and PD-1 are co-expressed on CD8+ T cells infiltrating head and neck cancer and melanoma, and have strong synergistic antitumor effect on blocking two signal pathways of NKG2A/HLA-E and PD-1/PD-L1 (cell, 2018; 175: 1-13, cell, 2018; 175: 1744-1755). Meanwhile, the treatment aiming at various targets has positive effects of improving the response rate of tumor immunotherapy and reducing immune tolerance. At present, the affinity of a therapeutic antibody aiming at NKG2A to antigen is insufficient, the therapeutic effect of the single target point binding to NKG2A to tumors is poor, and no medicine aiming at NKG2A is on the market in the world. FrancePaoli-CalmettesThe research institute is advancing a phase I clinical safety experiment (NCT 02921685) of combining humanized anti-NKG 2A monoclonal antibody IPH2201 with allogeneic stem cell transplantation for treating hematologic malignancies. The applicant has already constructed a mutant antibody with improved affinity and/or improved dissociation constant by constructing a light-heavy chain mutant antibody library, constructing CDRs (complementary deoxyribonucleic acid) regions of the mutant antibody into a human Fab heavy chain gene expression vector and a mammalian cell expression vector containing human kappa subclass light chain constant region genes, cross-pairing a heavy chain vector and a light chain vector of an affinity mature antibody, screening to obtain a mutant Fab antibody of anti-NKG 2A, and connecting with an Fc segment of a human antibody. The anti-PD-L1 nano antibody is connected to the C end of the Fc segment through a linker, and the antibody with dual specificity to NKG2A and PD-L1 is obtained.
Compared with the common antibody, the bispecific antibody has the advantages of good specificity, strong targeting property, low effect taking dosage, small toxic and side effects and the like, and has great significance in clinical treatment of tumors. The humanized recombinant bispecific antibody is a biological macromolecule, has a complex structure, is easy to generate physical changes such as aggregation, denaturation and precipitation and chemical changes such as isomerization, deamidation and oxidation during the production and storage processes, and the changes can influence the safety and the effectiveness of the product, so a stable preparation is needed to ensure that the antibody still has the biological activity required by treatment before being used in a patient body. No stable preparation specially aiming at the bispecific antibody exists in the market at present, and most of the stable preparations directly use natural antibody molecules. Although bispecific antibodies have more or less components of a generic antibody molecule, bispecific antibodies also differ from a generic antibody molecule by three points: (1) bispecific antibodies are artificially designed molecules, not naturally occurring molecules, whose factors cause bispecific antibody molecules to face more uncertain degradation, aggregation, metabolic effects. (2) The structure of the bispecific antibody is different from that of a natural antibody, and the difference of the molecular structure causes that the bispecific molecule is different from the natural antibody molecule in the physical and chemical parameters of molecular weight, isoelectric point, enzyme cutting site and the like. (3) Bispecific antibody molecules differ from native antibody molecules in activity and function, in that to maintain both specific binding activities, a bispecific antibody molecule must maintain both antigen-binding sites in a functional structure, particularly when both antigen-binding sites are located at two ends, such as Fab-Fc-sdA (Fab fragment linked to the N-terminus of the Fc fragment of an antibody via the C-terminus of the heavy chain, nanobody linked to the C-terminus of the Fc-terminus of an antibody via the N-terminus), and must maintain the entire molecular structure for activity.
Disclosure of Invention
In order to solve the problems, the invention optimizes the components of the bispecific antibody stability preparation according to the indexes of three aspects of physical state, microstructure and biological activity aiming at the difference of the molecular structure of the bispecific antibody and the natural antibody. Taking humanized recombinant anti-NKG 2A and PD-L1 bispecific antibody as an example, the optimized stability preparation takes acetic acid buffer solution or citric acid buffer solution with pH of 5.5-6.5 as a buffer system, takes 4% sucrose as a stabilizer, and is added with 0.005-0.08% Tween-80. The detection results of high temperature test, shaking test, repeated freeze-thaw test and illumination test show that the stable preparation of the invention can maintain the appearance and physical state of the injection and maintain the structural stability and biological activity of the bispecific antibody molecule.
Specifically, the method comprises the following steps:
in one aspect, the present invention provides an aqueous composition comprising:
buffer solution: 10-20mM acetic acid buffer solution or citric acid buffer solution;
a protective agent: 4% (w/v) sucrose;
surfactant (b): 0.005-0.08% (w/v) Tween-80;
the aqueous composition has a pH of 5.5 to 6.5.
Further, the aqueous composition of the present invention is characterized in that: the buffer is 10mM acetic acid buffer, and the pH of the aqueous composition is 5.5.
Further, the aqueous composition of the present invention is characterized in that: the buffer is a 20mM citrate buffer, and the pH of the aqueous composition is 6.0-6.5, preferably 6.2; and the aqueous composition further comprises 50mM NaCl.
Further, the aqueous composition of the present invention is characterized in that: the aqueous composition further comprises at least one bispecific antibody, the concentration of said bispecific antibody being 10-50mg/mL, preferably 20 mg/mL.
Further, the aqueous composition of the present invention is characterized in that the bispecific antibody comprises a Fab providing a first specificity, a nanobody domain providing a second specificity; wherein the Fab segment is linked to the N-terminus of the Fc segment of the antibody via the C-terminus of its heavy chain, and the nanobody domain is linked to the C-terminus of the Fc segment of the antibody via its N-terminus.
Further, the aqueous composition of the present invention is characterized in that the bispecific antibody is a humanized recombinant anti-NKG 2A and PD-L1 bispecific antibody, wherein the light chain variable region of SEQ ID No.1 and the heavy chain variable region of SEQ ID No.2 form an antigen binding site against NKG 2A; the nanobody domain shown in SEQ ID NO 3 forms anti-PD-L1 specificity.
In a second aspect, the present invention also provides the use of an aqueous composition according to any one of the preceding claims for stabilizing a bispecific antibody and/or for preparing a bispecific antibody formulation.
Further, the use according to the invention, characterized in that said bispecific antibody comprises a Fab providing a first specificity, a nanobody domain providing a second specificity; wherein the Fab segment is linked to the N-terminus of the Fc segment of the antibody via the C-terminus of its heavy chain, and the nanobody domain is linked to the C-terminus of the Fc segment of the antibody via its N-terminus.
In a third aspect, the present invention also provides the use of any one of the aqueous compositions described above for improving the high temperature stability, shaking stability, freeze-thaw stability, and/or light stability of a bispecific antibody.
Further, the use according to the invention, characterized in that said bispecific antibody comprises a Fab providing a first specificity, a nanobody domain providing a second specificity; wherein the Fab segment is linked to the N-terminus of the Fc segment of the antibody via the C-terminus of its heavy chain, and the nanobody domain is linked to the C-terminus of the Fc segment of the antibody via its N-terminus.
In a fourth aspect, the invention provides an aqueous needle formulation of a humanized recombinant anti-NKG 2A and PD-L1 bispecific antibody comprising:
bispecific antibody 10-50mg/ml
Citric acid buffer 20mM
50mM sodium chloride
Sucrose 4% (w/v)
Polysorbate 800.005% -0.08% (w/v)
pH 5.5-6.5
For a better understanding of the present invention, certain terms are first defined. Other definitions are listed throughout the detailed description section.
The term "NKG 2A" is an inhibitory receptor present in NK, NKT and T cell subsets, NKG2A (OMIM 161555, the entire disclosure of which is incorporated herein by reference) being a member of the NKG2 group of transcripts (Houchins, et al (1991) J. exp. Med. 173: 1017-. NKG2A is encoded by 7 exons spanning 25kb showing some differential splicing. NKG2A forms together with CD94 the heterodimeric inhibitory receptor CD94/NKG2A found on the surface of NK cells, α/β T cells, γ/δ T cells and subsets of NKT cells. Like the inhibitory KIR receptor, it has an ITIM in its cytoplasmic domain. As used herein, "NKG 2A" refers to any variant, derivative or isoform (isoform) of the NKG2A gene or encoded protein. Also included are any nucleic acid or protein sequences that share one or more biological properties or functions with wild-type full-length NKG2A and share at least 70%, 80%, 90%, 95%, 96%, 97%, 98%, 99% or more nucleotide or amino acid identity. Human NKG2A comprises 233 amino acids in 3 domains, wherein the cytoplasmic domain comprises residues 1-70, the transmembrane region comprises residues 71-93, and the extracellular region comprises residues 94-233.
The term "PD-L1", i.e.PD-L1 (programmed cell depletion 1) is known as programmed death receptor ligand 1, also known as cluster of differentiation 274 (CD 274) of the surface antigen or B7 homolog (B7 homolog 1, B7-H1), encoded by the CD274 gene, is a ligand for PD-1 (programmed cell depletion 1, programmed death receptor 1). PD-L1 is a first type transmembrane protein with the size of 40kDa, is expressed on immune cells such as T cells, B cells and the like and tumor cells, and normally the immune system reacts to foreign antigens gathered in lymph nodes or spleen to promote cytotoxic T cells with antigen specificity (CD 8+ Tcell proliferation). When PD-L1 on the tumor cell membrane is combined with PD-1 on immune cells such as T cells and the like, the tumor cells send inhibitory signals to reduce the proliferation of lymph node CD8+ T cells, so that the T cells cannot recognize the tumor cells and have killing effect on the tumor cells, and the immune function of an organism is inhibited.
The term "specific" refers to the determination of the presence or absence of a protein in a heterogeneous population of proteins and/or other organisms. Thus, under the conditions specified, a particular ligand/antigen binds to a particular receptor/antibody and does not bind in significant amounts to other proteins present in the sample.
The term "antibody" herein is intended to include full-length antibodies and any antigen-binding fragment (i.e., antigen-binding portion) or single chain thereof. Full-length antibodies are glycoproteins comprising at least two heavy (H) chains and two light (L) chains, the heavy and light chains being linked by disulfide bonds. Each heavy chain is composed of a heavy chain variable region (abbreviated VH) and a heavy chain constant region. The heavy chain constant region is composed of three domains, CH1, CH2, and CH 3. Each light chain is composed of a light chain variable region (abbreviated as VL) and a light chain constant region. The light chain constant region is composed of one domain CL. The VH and VL regions can also be divided into hypervariable regions, called Complementarity Determining Regions (CDRs), which are separated by more conserved Framework Regions (FRs). Each VH and VL is composed of three CDRs and four FRs, arranged in the order FR1, CDR1, FR2, CDR2, FR3, CDR3, FR4 from the amino terminus to the carboxy terminus. The variable regions of the heavy and light chains comprise binding domains that interact with antigens. The constant region of the antibody may mediate the binding of the immunoglobulin to host tissues or factors, including various immune system cells (e.g., effector cells) and the first component of the classical complement system (C1 q).
The term "bispecific antibodies" (bispecific antibodies), an antibody structure that binds to different epitopes on the same or different antigens. Thus, bispecific antibodies are capable of bridging two different molecules, serving to recruit effector molecules, effector cells, viruses, and drug carrier systems to a target structure. The bispecific antibody can simultaneously recognize two different molecules (receptor and/or ligand), thereby improving the selectivity and functional affinity of the antibody.
The term "monoclonal antibody" or "monoclonal antibody composition" refers to a preparation of antibody molecules of a single molecular composition. Monoclonal antibody compositions exhibit a single binding specificity and affinity for a particular epitope.
The term "antigen-binding fragment" of an antibody (or simply antibody portion), as used herein, refers to one or more fragments of an antibody that retain the ability to specifically bind antigen. It has been demonstrated that the antigen binding function of an antibody can be performed by fragments of a full-length antibody. Examples of binding fragments comprised in the "antigen-binding portion" of an antibody include (i) Fab fragments, monovalent fragments consisting of VL, VH, CL and CH 1; (ii) a F (ab') 2 fragment, a bivalent fragment comprising two Fab fragments linked by a hinge region disulfide bridge; (iii) an Fd fragment consisting of VH and CH 1; (iv) an Fv fragment consisting of VL and VH antibody single arms; (v) dAb fragments consisting of VH (Ward et al, (1989) Nature 341: 544-546); (vi) an isolated Complementarity Determining Region (CDR); and (vii) a nanobody, a heavy chain variable region comprising a single variable domain and two constant domains. Furthermore, although the two domains of the Fv fragment, VL and VH, are encoded by different genes, they can be joined by recombinant methods via a synthetic linker that makes the two single protein chains, in which the VL and VH regions pair to form monovalent molecules (known as single chain Fc (scFv); see, e.g., Bird et al., (1988) Science 242: 423-. These single chain antibodies are also intended to be included within the term meaning. These antibody fragments can be obtained by conventional techniques known to those skilled in the art, and the fragments can be functionally screened in the same manner as intact antibodies.
Antigen-binding fragments of the invention include those capable of specifically binding to an antigen. Examples of antibody binding fragments include, for example, but are not limited to, Fab ', F (ab')2Fv fragments, single chain Fv (scFv) fragments and single domain fragments.
The Fab fragment contains the constant domain of the light chain and the first constant domain of the heavy chain (CH 1). Fab' fragments differ from Fab fragments by the addition of a few residues at the carboxy terminus of the heavy chain CH1 domain, including one or more cysteines from the antibody hinge region. Fab 'fragments are generated by cleavage of the disulfide bond at the hinge cysteine of the F (ab') 2 pepsin digestion product. Additional chemical couplings of antibody fragments are known to those of ordinary skill in the art. Fab and F (ab') 2 fragments lack the fragment crystallizable (Fc) region of intact antibodies, clear more rapidly from the circulation of animals, and may have less non-specific tissue binding than intact antibodies (see, e.g., Wahl et al, 1983, J. Nucl. Med. 24: 316).
As is generally understood in the art, an "Fc" region is a fragment crystallizable constant region of an antibody that does not comprise an antigen-specific binding region. In IgG, IgA and IgD antibody isotypes, the Fc region consists of two identical protein fragments derived from the second and third constant domains of the two heavy chains of an antibody (CH 2 and CH3 domains, respectively). The IgM and IgE Fc regions contain three heavy chain constant domains (CH 2, CH3, and CH4 domains) in each polypeptide chain.
"Single domain fragments", sda (single domain antibody) consists of a single VH or VL domain showing sufficient affinity for the antigen. In a particular embodiment, the single domain fragments are camelized (see, e.g., Riechmann, 1999, Journal of immunological Methods 231: 25-38).
The anti-NKG 2A antibodies of the invention include derivatized antibodies. For example, derivatized antibodies are typically modified by glycosylation, acetylation, pegylation, phosphorylation, amidation, derivatization by known protecting/blocking groups, proteolytic cleavage, attachment to cellular ligands or other proteins. Any of a number of chemical modifications can be made by known techniques including, but not limited to, specific chemical cleavage, acetylation, formylation, metabolic synthesis of tunicamycin, and the like. In addition, the derivative may contain one or more unnatural amino acid, e.g., using ambrx technology (see, e.g., Wolfson, 2006, chem. biol. 13 (10): 1011-2).
The term "binding affinity" is used herein as a measure of the strength of a non-covalent interaction between two molecules (e.g., an antibody or fragment thereof, and an antigen). The term "binding affinity" is used to describe monovalent interactions (intrinsic activity). The binding affinity between two molecules (e.g., an antibody or fragment thereof, and an antigen) via a monovalent interaction can be quantitatively determined by determining the dissociation constant (KD). KD can then be determined by measurement of complex formation and dissociation kinetics, for example by SPR methods. The rate constants corresponding to the association and dissociation of monovalent complexes are referred to as the association rate constant ka (or kon) and the dissociation rate constant kd (or koff), respectively. KD is linked to ka and KD by the equation KD = KD/ka. According to the above definitions, the binding affinities associated with different molecular interactions, e.g. the binding affinities of different antibodies for a given antigen, can be compared by comparing the KD values of the individual antibody/antigen complexes. Similarly, the specificity of an interaction can be evaluated by determining and comparing the KD value for the interaction of interest (e.g., a specific interaction between an antibody and an antigen) to the KD value for an interaction not of interest. The value of the dissociation constant can be determined directly by well-known methods, e.g., by standard assays that assess the binding ability of a ligand (e.g., an antibody) to a target are known in the art and include, e.g., ELISA, western blot, RIA, and flow cytometry analysis. The binding kinetics and binding affinity of the antibody can also be assessed by standard assays known in the art, such as SPR. A competitive binding assay may be performed in which the binding of an antibody to a target is compared to the binding of another ligand (e.g., another antibody) to the target.
The term "high affinity" for an IgG antibody means a KD for the antigen of 1.0x 10-6M is less, preferably 5.0x 10-8M or less, more preferably 1.0x 10-8M below, 5.0x 10-9M or less, more preferably 1.0x 10-9M is less than or equal to M. For other antibody subtypes, "high affinity" binding may vary. For example, "high affinity" binding of an IgM subtype means a KD of 10-6M is less, preferably 10-7M is less, more preferably 10-8M is less than or equal to M.
The term "Kassoc" or "Ka" refers to the association rate of a particular antibody-antigen interaction, while the term "Kdis" or "Kd" refers to the dissociation rate of a particular antibody-antigen interaction. The term "KD" refers to the dissociation constant, derived from the KD to Ka ratio (KD/Ka), and expressed in molar concentration (M). The KD value of an antibody can be determined by methods known in the art. A preferred way of determining the KD of an antibody is by measurement using a Surface Plasmon Resonance (SPR), preferably a biosensing system such as the Biacore (TM) system.
The term "EC 50," also called half maximal effect concentration, refers to the concentration of antibody that causes 50% of the maximal effect.
Compared with the prior art, the technical scheme of the invention has the following advantages:
firstly, the detection of physical state, chemical state and biological function is carried out aiming at the structural characteristics of the Fab-Fc-sdA type tetravalent bispecific antibody. Based on the change of physical form, the change of molecular structure and the change of biological activity of the water injection, the types and contents of a buffering agent, an osmotic pressure regulator, a protective agent and a surfactant which influence the stability of the water injection are optimized so as to improve the stability of the water injection in production, storage and transportation.
Secondly, the stability of the bispecific antibody is improved under various physicochemical conditions by adopting comprehensive stability indexes. According to physicochemical factors causing the instability of the Fab-Fc-sdA type tetravalent bispecific antibody, the high-temperature stability, the shaking stability, the freeze-thaw stability and the illumination stability of the bispecific antibody are comprehensively improved through the optimization of each component and content of the water injection.
And thirdly, a multi-angle detection method is adopted. According to the change of the aggregate or degradation products of the bispecific antibody in volume, charge and the like, the change of the microscopic molecules of the bispecific antibody is detected by using Size Exclusion Chromatography (SEC), Ion Exchange Chromatography (IEC), Non-reduced Sodium Dodecyl Sulfate Capillary Electrophoresis (NR CE-SDS, Non-reduced Capillary Electrophoresis Sodium Dodecyl Sulfate) multi-angle.
Drawings
Various other advantages and benefits will become apparent to those of ordinary skill in the art upon reading the following detailed description of the preferred embodiments. The drawings are only for purposes of illustrating the preferred embodiments and are not to be construed as limiting the invention. Also, like reference numerals are used to refer to like parts throughout the drawings. In the drawings:
FIG. 1: FACS analysis of 6MW3411 binding activity to NK92 cell surface NKG 2A.
FIG. 2: FACS assay 6MW3411 was assayed for binding activity to PD-L1 on the cell surface of MDA-MB-231.
Detailed Description
It should be understood that the present disclosure may be embodied in various forms and should not be limited by the embodiments set forth herein. Rather, these embodiments are provided so that this disclosure will be thorough and complete, and will fully convey the scope of the disclosure to those skilled in the art.
Example 1 antibody preparation screening assay
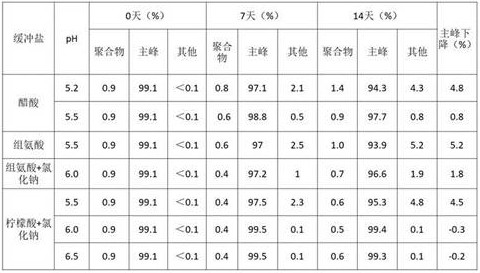
1.1 optimization of pH and buffer systems
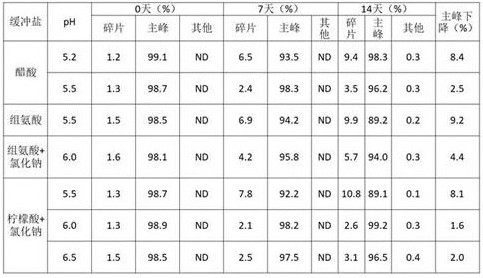
According to the molecular characteristics, a humanized recombinant anti-NKG 2A and PD-L1 bispecific antibody (wherein the amino acid sequence of the light chain variable region of the double anti-NKG 2A is SEQ ID NO.1, the amino acid sequence of the heavy chain variable region of the anti-NKG 2A is SEQ ID NO.2, the amino acid sequence of the variable region of the anti-PD-L1 single variable domain antibody VHH-F2 is SEQ ID NO.3, and the antibody is subsequently abbreviated as 6MW 3411) is replaced into the following target buffer solution, subpackaged and subjected to stability inspection. The buffer system is shown in table 1.
TABLE 1 optimization of pH and buffer systems
After the 6MW3411 sample was replaced with the above buffer system, the samples in the citric acid system were found to delaminate after standing by visual inspection, and histidine precipitated at pH > 6.0. Therefore, other parameters were optimized based on the acetate buffer, while the citrate buffer and the histidine buffer were modified by addition of a chaotropic agent and the like.
1.2 bispecific antibody concentration optimization
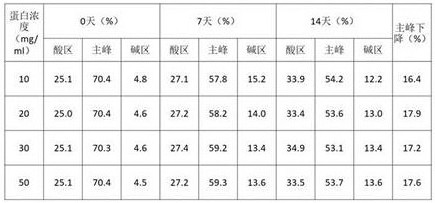
The above experiment shows that the protein does not precipitate under the acetic acid condition, so the stability of different protein concentrations is examined under the condition of 10mM acetic acid pH5.5. The 6MW3411 double antibody is replaced into different protein concentrations, placed at 40 ℃ for high-temperature forced condition test, and sampled at 0 day, 7 days and 14 days for SEC, IEC and NR CE-SDS detection. The experimental results are as follows:
TABLE 2 high temperature test SEC investigation results for different protein concentrations
TABLE 3 high temperature test IEC investigation results for different protein concentrations
TABLE 4 high temperature test of different protein concentrations NR CE-SDS investigation results
The results in tables 2-4 show that after 14 days at 40 ℃, the SEC purity, NR-CE purity and CEX main peak of the 6MW3411 sample all decreased, and the magnitude of the decrease in each key mass attribute was more pronounced the higher the concentration; indicating that higher protein concentrations are detrimental to 6MW3411 stabilization and that samples are more prone to aggregation, fragmentation and charge heterogeneity. In view of the above results, the clinical regimen was considered in combination and subsequently further investigation was carried out around a concentration of 20 mg/ml.
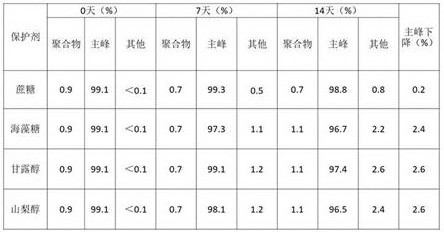
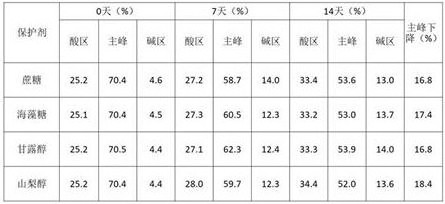
1.3 optimization of protective Agents
The stability of 6MW3411 in different protectant classes was examined at 10mM acetic acid pH 5.5. The 6MW3411 double antibody is replaced into different protective agents, placed at 40 ℃ for high-temperature forced condition test, and sampled at 0 day, 7 days and 14 days for SEC, IEC and NR CE-SDS detection. The experimental results are as follows:
TABLE 5 SEC investigation results of different protective agents
TABLE 6 IEC investigation results of high temperature tests with different protective agents
TABLE 7 high temperature test NR CE-SDS investigation results of different protectants
The results in tables 5-7 show that the major SEC, NR-CE and CEX peaks for the 6MW3411 samples all decreased after 14 days at 40 ℃. The SEC purity, NR-CE purity and CEX main peak reduction of samples in trehalose, mannitol and sorbitol were significantly higher than those in sucrose, indicating that sucrose is advantageous for maintaining the stability of the 6MW3411 sample. Subsequent studies therefore used sucrose as a protective agent at 6MW 3411.
1.4 Effect of sodium chloride on buffer stabilization function
The previous stability examination shows that protein is easy to precipitate in histidine and citric acid, so that sodium chloride is added into two buffer systems to promote dissolution, 6MW3411 double antibodies are replaced into different buffer systems by taking acetic acid as a control, the buffer systems are placed at 40 ℃ for high-temperature forced condition test, and samples are taken at 0 day, 7 days and 14 days for SEC, IEC and NR CE-SDS detection. The experimental results are as follows:
TABLE 8 SEC investigation results of different buffer pH high temperature tests
TABLE 9 IEC finding results of high temperature tests on different buffer pH
TABLE 10 high temperature test of different buffer pH NR CE-SDS investigation results
The results in tables 8-10 show that the major SEC, NR-CE and CEX peaks for the 6MW3411 samples all decreased after 14 days at 40 ℃. Wherein the SEC purity and the NR-CE purity are significantly reduced in acetic acid pH5.2, histidine pH5.5 and citric acid + sodium chloride pH 5.5; the SEC purity and NR-CE purity varied less than acetic acid and histidine at citric acid pH greater than 6.0. In pH6.5, the main CEX peak was reduced significantly, and the variation in charge heterogeneity of CEX was not significantly different in the other conditions. And combining the results, and subsequently selecting about 6.0 of citric acid and sodium chloride for next investigation.
The present inventors have surprisingly found, by comparing the above experiments 1.4 and 1.1, that the stabilization effect of citrate buffer is optimal with a minimum main peak drop under the condition of adding 50mM sodium chloride at pH >6.0, despite the poor stabilization ability of citrate buffer against bispecific antibody in the one-way experiment, resulting in the demixing of the antibody water injection on standing.
1.5 surfactant selection
The 6MW3411 sample was replaced with 20mM citric acid, 4% sucrose, 50mM sodium chloride, ph6.2 buffer solution, at a protein concentration of 20mg/ml, and different proportions of polysorbate 80 were added to the replaced sample. The prepared sample is repeatedly frozen and thawed for 1 time, 3 times and 5 times, shaken for 1 day and 3 days, and placed at 40 ℃ for 14 days, and then the insoluble particles in the sample are measured through MFI. The results are shown in tables 11 to 12.
TABLE 11 polysorbate 80 freeze-thaw samples of different ratios insoluble microparticle assay results
TABLE 12 results of different proportions of polysorbate 80 shaking and high temperature sample insoluble particulates
The results in tables 11-12 show that the samples without polysorbate 80 were not treated at all, i.e., large numbers of particles appeared, and particles were visible to the naked eye, indicating that surfactant stability at 6MW3411 is important. After the samples in different proportions of polysorbate 80 are frozen and thawed 5 times, the sample particles in 0.005% of polysorbate 80 are obviously increased; after shaking for 3 days and standing at 40 ℃ for 14 days, the sample particles in 0.005% and 0.01% of polysorbate 80 were both significantly increased; after the mixture is placed at 40 ℃ for 14 hours, when the concentration of polysorbate 80 is more than or equal to 0.04%, the number of particles more than or equal to 5 mu m is less than 500/ml, and the number of particles more than or equal to 10 mu m is less than 100/ml. Thus, a suitable polysorbate 80 concentration that effectively inhibited the production of insoluble microparticles in the 6MW3411 sample was 0.05% ± 0.01%.
As described above, 20mg/ml protein concentration, 20mM citric acid, 50mM sodium chloride, 4% sucrose, pH 6.0. + -. 0.2 were selected. At the same time, polysorbate 80 at 0.05% was added as a 6MW3411 surfactant.
Example two: prescription influence factor test
2.1 high temperature test
The samples were tested for SEC, IEC, NR CE-SDS, relative binding activity and insoluble particles after 7 days, 14 days and 28 days of standing at 40 ℃ and the results are shown in Table 13. The results show a slight decrease in SEC purity and NR-CE purity at high temperature, a significant decrease in IEC peak, and a small increase in insoluble particles, indicating that 6MW3411 is sensitive to high temperature and that the samples should be stored at low temperature.
TABLE 13.6 MW3411 preferred recipe high temperature test results
*Means not detecting
2.2 shaking test
The samples were examined for SEC, IEC, NR CE-SDS, relative binding activity and insoluble particles after shaking the displaced samples at 10 ℃ and 200 rpm for 0 day, 1 day and 3 days, respectively, and the results are shown in Table 14. The results show that no significant change occurs in each detection index, and the 6MW3411 double-antibody in the preferred prescription is insensitive to oscillation and has good stability.
TABLE 14.6 MW3411 preferred recipe shaking test results
*Means not detecting
2.3 repeated Freeze/thaw test
After the replaced sample is frozen and thawed 0 time, 1 time, 3 times and 5 times respectively at the temperature of-40 ℃ to 4 ℃, SEC, IEC, NR CE-SDS, relative binding activity and insoluble particles of the sample are detected, and the results are shown in Table 15. The results show that the detection indexes are not changed significantly, and the results show that the 6MW3411 double antibody in the optimal formula is insensitive to repeated freeze thawing and has good stability.
TABLE 15.6 MW3411 preferred recipe repeated freeze-thaw test results
*Means not detecting
2.4 light test
The replaced samples were illuminated under 4500 lx ± 500 lx conditions for 0, 5 and 10 days, respectively (while the samples were placed in a common paper packaging box as a light-shielding control group), and the samples were tested for SEC, IEC, NR CE-SDS, relative binding activity and insoluble particle assay, with the results shown in table 16. The result shows that in the illumination test, the SEC and IEC main peak contents of the 6MW3411 double antibody are obviously reduced, the NR-CE main peak is obviously reduced, and the relative binding activity is obviously reduced; in the light-shielding control group, all detection indexes have no significant change. The light irradiation has an effect on 6MW3411 double antibody in the optimal prescription, and the common paper packaging box can play a role in keeping out of the sun.
TABLE 16.6 MW3411 preferred prescription light test results
*Means not detecting
Example 3.6 MW3411 antibody molecule affinity analysis
The affinity of 6MW341 to NKG2A and PD-L1 were determined by using an Octet QKe system instrument from Fortebio, using a 6MW3411 antibody Fc fragment capture Antibody (AHC) bioprobe to capture the antibody Fc fragment. 6MW3411 was diluted to 4. mu.g/ml in PBS buffer and passed over the AHC probe surface for 240 s. Human NKG2A extracellular region fusion protein or human PD-L1 extracellular region fusion protein was used as a mobile phase, the binding time was 300s, and the dissociation time was 300 s. After the experiment, blank control response values were deducted, and the software was run for 1:1 Langmuir binding pattern was fitted and kinetic constants for antigen-antibody binding were calculated.
The affinity (KD) parameters of 6MW3411 and human NKG2A and PD-L1 recombinant proteins are shown in Table 17, and 6MW3411 can be effectively combined with NKG2A and PD-L1.
TABLE 17.6 result of affinity assay of MW3411 and human NKG2A/PD-L1 recombinant protein
Example 4.6MW3411 analysis of cell surface NKG2A and PD-L1 binding Activity
The binding of 6MW3411 to cell surface NKG2A and PD-L1 was evaluated using NK92 cells and MBA-MD-231 cells, respectively.
Specifically, a suspension of natural cells NK92 was incubated with 6MW3411 labeled AF488 and a control Antibody NC-IgG4 (Mix-n-Stain CF488A Antibody Labeling Kit: Cat. MX488AS100-1 KT, sigma) at 4 ℃ for 40 min at cell concentrations: 1 × 105 cells/sample, final antibody concentration: 55 nM starts 3-fold serial dilution of 8 gradients. Cells were washed 3 times with ice-cold PBS (containing 0.05% Tween) and tested on the machine. The Mean Fluorescence Intensity (MFI) of the cells was measured by flow cytometry (model B49007AD, SNAW31211, BECKMAN COULTER) after PBST washing the cells 3 times to examine the binding ability of the humanized antibody to natural cell NK 92. The results are shown in figure 1, 6MW3411 can specifically bind to NKG2A antibody on NK92 cell surface.
On the other hand, MDA-MB-231 cell suspensions were incubated with antibody-gradient diluted detection antibody 6MW3411 and negative control antibody NC-IgG4 at 4 ℃ for 60 min at cell concentrations: 1 x 105 cells/sample, antibody final concentration 10 nM initial, 3 times serial dilution 10 gradient. After washing the cells 3 times with ice cold PBST (0.05% Tween), goat anti-human IgG-FITC (Cat.: F9512, Sigma) diluted 1:100 was added and incubated at 4 ℃ for 30 min. The Mean Fluorescence Intensity (MFI) of the cells was measured by flow cytometry after PBST washing the cells 3 times to examine the binding ability of the humanized antibody to human PD-L1 on the cell surface of MDA-MB-231. The results are shown in FIG. 2, where the bispecific antibodies all retained the binding activity to cell surface PD-L1.
The amino acid sequence involved in the invention is as follows:
SEQ ID NO. 1: anti-NKG 2A light chain variable region amino acid sequence
DIQMTQSPSSLSASVGDRVTITCRASENIYSYAAWYQQKPGKAPKLLIYNRKTLAEGVPSRFSGSGSGTDFTLTISSLQPEDFATYYCQHRYGTPRTFGGGTKVEIK
SEQ ID NO. 2: anti-NKG 2A heavy chain variable region amino acid sequence
EVQLVQSGAEVKKPGASVKVSCKASSYDFSWYWINWVRQAPGQGLEWMGAIDPYDSETHYAQKLQGRVTMTTDTSTSTAYMELRSLRSDDTAVYYCARGGYDFDQGTLYWFFDVWGQGTTVTVSS
SEQ ID NO. 3: anti-PD-L1 single variable domain antibody VHH-F2 variable region amino acid sequence
EVQLVESGGGLVQPGGSLRLSCAASRDSDEGASCMGWFRQAPGKEREGVAIIFNAGERTDYGDSVKGRFTISRDNAKNTLYLQMNSLRAEDTAVYYCATVWCGSWVARSWGQGTLVTVSS
The above description is only for the preferred embodiment of the present invention, but the scope of the present invention is not limited thereto, and any changes or substitutions that can be easily conceived by those skilled in the art within the technical scope of the present invention are included in the scope of the present invention. Therefore, the protection scope of the present invention shall be subject to the protection scope of the appended claims.
Sequence listing
<110> Miwei (Shanghai) Biotech Co., Ltd
<120> a stable formulation of bispecific antibody
<160> 3
<170> SIPOSequenceListing 1.0
<210> 1
<211> 107
<212> PRT
<213> Artificial Sequence (Artificial Sequence)
<400> 1
Asp Ile Gln Met Thr Gln Ser Pro Ser Ser Leu Ser Ala Ser Val Gly
1 5 10 15
Asp Arg Val Thr Ile Thr Cys Arg Ala Ser Glu Asn Ile Tyr Ser Tyr
20 25 30
Ala Ala Trp Tyr Gln Gln Lys Pro Gly Lys Ala Pro Lys Leu Leu Ile
35 40 45
Tyr Asn Arg Lys Thr Leu Ala Glu Gly Val Pro Ser Arg Phe Ser Gly
50 55 60
Ser Gly Ser Gly Thr Asp Phe Thr Leu Thr Ile Ser Ser Leu Gln Pro
65 70 75 80
Glu Asp Phe Ala Thr Tyr Tyr Cys Gln His Arg Tyr Gly Thr Pro Arg
85 90 95
Thr Phe Gly Gly Gly Thr Lys Val Glu Ile Lys
100 105
<210> 2
<211> 125
<212> PRT
<213> Artificial Sequence (Artificial Sequence)
<400> 2
Glu Val Gln Leu Val Gln Ser Gly Ala Glu Val Lys Lys Pro Gly Ala
1 5 10 15
Ser Val Lys Val Ser Cys Lys Ala Ser Ser Tyr Asp Phe Ser Trp Tyr
20 25 30
Trp Ile Asn Trp Val Arg Gln Ala Pro Gly Gln Gly Leu Glu Trp Met
35 40 45
Gly Ala Ile Asp Pro Tyr Asp Ser Glu Thr His Tyr Ala Gln Lys Leu
50 55 60
Gln Gly Arg Val Thr Met Thr Thr Asp Thr Ser Thr Ser Thr Ala Tyr
65 70 75 80
Met Glu Leu Arg Ser Leu Arg Ser Asp Asp Thr Ala Val Tyr Tyr Cys
85 90 95
Ala Arg Gly Gly Tyr Asp Phe Asp Gln Gly Thr Leu Tyr Trp Phe Phe
100 105 110
Asp Val Trp Gly Gln Gly Thr Thr Val Thr Val Ser Ser
115 120 125
<210> 3
<211> 120
<212> PRT
<213> Artificial Sequence (Artificial Sequence)
<400> 3
Glu Val Gln Leu Val Glu Ser Gly Gly Gly Leu Val Gln Pro Gly Gly
1 5 10 15
Ser Leu Arg Leu Ser Cys Ala Ala Ser Arg Asp Ser Asp Glu Gly Ala
20 25 30
Ser Cys Met Gly Trp Phe Arg Gln Ala Pro Gly Lys Glu Arg Glu Gly
35 40 45
Val Ala Ile Ile Phe Asn Ala Gly Glu Arg Thr Asp Tyr Gly Asp Ser
50 55 60
Val Lys Gly Arg Phe Thr Ile Ser Arg Asp Asn Ala Lys Asn Thr Leu
65 70 75 80
Tyr Leu Gln Met Asn Ser Leu Arg Ala Glu Asp Thr Ala Val Tyr Tyr
85 90 95
Cys Ala Thr Val Trp Cys Gly Ser Trp Val Ala Arg Ser Trp Gly Gln
100 105 110
Gly Thr Leu Val Thr Val Ser Ser
115 120
Claims (7)
1. An aqueous composition, the composition ingredients consisting of:
buffer solution: 10-20mM citric acid buffer solution;
a protective agent: 4% (w/v) sucrose;
surfactant (b): 0.005-0.08% (w/v) Tween-80; and 50mM NaCl, and
a bispecific antibody;
the bispecific antibody is a humanized recombinant anti-NKG 2A and PD-L1 bispecific antibody which comprises an anti-NKG 2A specific antigen binding site formed by a light chain variable region shown in SEQ ID NO.1 and a heavy chain variable region shown in SEQ ID NO.2, an anti-PD-L1 specific antigen binding site formed by a nano antibody structure domain shown in SEQ ID NO. 3;
the aqueous composition has a pH of 5.5 to 6.5.
2. The aqueous composition of claim 1, wherein: the buffer is a 20mM citrate buffer, and the pH of the aqueous composition is 6.0-6.5.
3. The aqueous composition of claim 2, wherein: the pH of the aqueous composition was 6.2.
4. The aqueous composition of claim 1, wherein: the concentration of the bispecific antibody is 10-50 mg/mL.
5. The aqueous composition of claim 4, wherein: the concentration of the bispecific antibody was 20 mg/mL.
6. The aqueous composition of claim 4, wherein said bispecific antibody comprises a Fab providing specificity for NKG2A, a nanobody domain providing specificity for PD-L1; wherein the Fab segment is linked to the N-terminus of the Fc segment of the antibody via the C-terminus of its heavy chain, and the nanobody domain is linked to the C-terminus of the Fc segment of the antibody via its N-terminus.
7. An aqueous injection formulation of a humanized recombinant anti-NKG 2A and PD-L1 bispecific antibody comprising:
bispecific antibody 10-50mg/ml
Citric acid buffer 20mM
50mM sodium chloride
Sucrose 4% (w/v)
Tween-80 0.005-0.08%(w/v)
pH 5.5-6.5
The bispecific antibody is a humanized recombinant anti-NKG 2A and PD-L1 bispecific antibody, which comprises an anti-NKG 2A specific antigen binding site formed by a light chain variable region shown in SEQ ID NO.1 and a heavy chain variable region shown in SEQ ID NO.2, and an anti-PD-L1 specific antigen binding site formed by a nano antibody structural domain shown in SEQ ID NO. 3.
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111566377.XA CN113940997B (en) | 2021-12-21 | 2021-12-21 | Stable preparation of bispecific antibody |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN202111566377.XA CN113940997B (en) | 2021-12-21 | 2021-12-21 | Stable preparation of bispecific antibody |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN113940997A CN113940997A (en) | 2022-01-18 |
| CN113940997B true CN113940997B (en) | 2022-04-08 |
Family
ID=79339440
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN202111566377.XA Active CN113940997B (en) | 2021-12-21 | 2021-12-21 | Stable preparation of bispecific antibody |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN113940997B (en) |
Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106999581A (en) * | 2014-11-07 | 2017-08-01 | 诺华股份有限公司 | Stable protein solution dosage containing high concentration anti-VEGF antibody |
| WO2017180769A1 (en) * | 2016-04-13 | 2017-10-19 | Capten Therapeutics Inc. | Small molecules for immunogenic treatment of cancer |
| CN110974958A (en) * | 2019-12-25 | 2020-04-10 | 北京东方百泰生物科技有限公司 | Injection preparation of anti-PD-L1 monoclonal antibody |
| CN113527488A (en) * | 2020-04-22 | 2021-10-22 | 迈威(上海)生物科技股份有限公司 | Single variable domain antibody targeting human programmed death ligand 1(PD-L1) and derivative thereof |
| CN113583127A (en) * | 2020-04-30 | 2021-11-02 | 迈威(上海)生物科技股份有限公司 | Bispecific antibody targeting NKG2A and PD-L1 and application thereof |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| PE20190448A1 (en) * | 2016-06-30 | 2019-03-29 | Celltrion Inc | STABLE LIQUID PHARMACEUTICAL FORMULATION |
-
2021
- 2021-12-21 CN CN202111566377.XA patent/CN113940997B/en active Active
Patent Citations (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106999581A (en) * | 2014-11-07 | 2017-08-01 | 诺华股份有限公司 | Stable protein solution dosage containing high concentration anti-VEGF antibody |
| WO2017180769A1 (en) * | 2016-04-13 | 2017-10-19 | Capten Therapeutics Inc. | Small molecules for immunogenic treatment of cancer |
| CN110974958A (en) * | 2019-12-25 | 2020-04-10 | 北京东方百泰生物科技有限公司 | Injection preparation of anti-PD-L1 monoclonal antibody |
| CN113527488A (en) * | 2020-04-22 | 2021-10-22 | 迈威(上海)生物科技股份有限公司 | Single variable domain antibody targeting human programmed death ligand 1(PD-L1) and derivative thereof |
| CN113583127A (en) * | 2020-04-30 | 2021-11-02 | 迈威(上海)生物科技股份有限公司 | Bispecific antibody targeting NKG2A and PD-L1 and application thereof |
Non-Patent Citations (1)
| Title |
|---|
| Potential prognostic value of PD‑L1 and NKG2A expression in Indonesian patients with skin nodular melanoma;Ridwan Dwi Saputro et al.;《BMC Research Notes》;20210528;第14卷;第1-6页 * |
Also Published As
| Publication number | Publication date |
|---|---|
| CN113940997A (en) | 2022-01-18 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN113286817B (en) | DLL3 binding proteins and methods of use | |
| EP3818086B1 (en) | Treatment and prevention of cancer using her3 antigen-binding molecules | |
| KR102593409B1 (en) | HER3 antigen binding molecule | |
| EP3645570B1 (en) | Vista antigen-binding molecules | |
| JP7436711B2 (en) | Anti-SIRP-alpha antibody | |
| CN111808190B (en) | Antibodies that bind PD-1 | |
| US11117964B2 (en) | Anti-KIR3DL1 antibodies | |
| WO2021219048A1 (en) | Bispecific antibody targeting nkg2a and pd-l1, and application | |
| WO2022082014A2 (en) | Skin targeted immunotolerance | |
| JP2022505925A (en) | Anti-TIM-3 antibody | |
| CN110167959A (en) | For monitoring immunoassays and the engineering protein of the Antybody therapy to immunologic test point inhibitor PD1 and PD-L1 | |
| TW202302651A (en) | Bispecific antibody combination and use thereof | |
| CN112442123B (en) | anti-CD47 monoclonal antibody and application thereof | |
| CN113940997B (en) | Stable preparation of bispecific antibody | |
| WO2023031435A1 (en) | Treatment and prevention of cancer using her3 antigen-binding molecules | |
| JP7753401B2 (en) | Humanized anti-CLEC-1A antibody, its antigen-binding fragment, and its mimetic | |
| CN114085291B (en) | Method for reducing or eliminating CEX acidic peak of recombinant protein | |
| TW202330026A (en) | Combination of an anti-btn3a activating antibody and an il-2 agonist for use in therapy | |
| CN114072175A (en) | Materials and methods for treating tumors | |
| CN121108346A (en) | Anti-LY 6G6D single domain antibody, and preparation method and application thereof | |
| TW202241957A (en) | Anti-pd-1 antibody and use thereof | |
| JP2026505058A (en) | Antibodies with selectivity for the 4Ig isoform of B7-H3 and methods of use thereof | |
| TW202241958A (en) | Anti-pd-l1 antibody and application thereof | |
| HK40063855A (en) | Substances and methods for treating tumors | |
| HK40033623A (en) | Antibody binding to pd-1 |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant |