CN112010927A - EGH and hydroxymethyl modified benzimidazole quinazoline, and synthesis, activity and application thereof - Google Patents
EGH and hydroxymethyl modified benzimidazole quinazoline, and synthesis, activity and application thereof Download PDFInfo
- Publication number
- CN112010927A CN112010927A CN201910452670.XA CN201910452670A CN112010927A CN 112010927 A CN112010927 A CN 112010927A CN 201910452670 A CN201910452670 A CN 201910452670A CN 112010927 A CN112010927 A CN 112010927A
- Authority
- CN
- China
- Prior art keywords
- quinazoline
- hydroxymethyl
- boc
- dihydrobenzo
- glu
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Granted
Links
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K5/00—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof
- C07K5/04—Peptides containing up to four amino acids in a fully defined sequence; Derivatives thereof containing only normal peptide links
- C07K5/08—Tripeptides
- C07K5/0821—Tripeptides with the first amino acid being heterocyclic, e.g. His, Pro, Trp
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P37/00—Drugs for immunological or allergic disorders
- A61P37/02—Immunomodulators
- A61P37/06—Immunosuppressants, e.g. drugs for graft rejection
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Immunology (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Engineering & Computer Science (AREA)
- Life Sciences & Earth Sciences (AREA)
- Medicinal Chemistry (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- General Chemical & Material Sciences (AREA)
- Biophysics (AREA)
- Molecular Biology (AREA)
- Genetics & Genomics (AREA)
- Transplantation (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Biochemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Plural Heterocyclic Compounds (AREA)
Abstract
Description
技术领域technical field
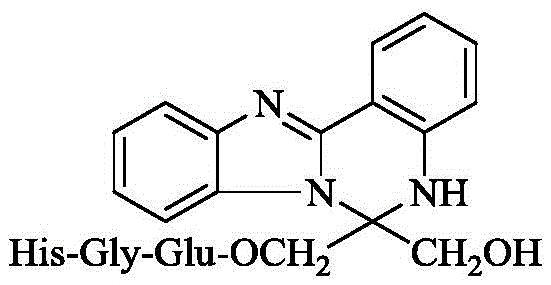
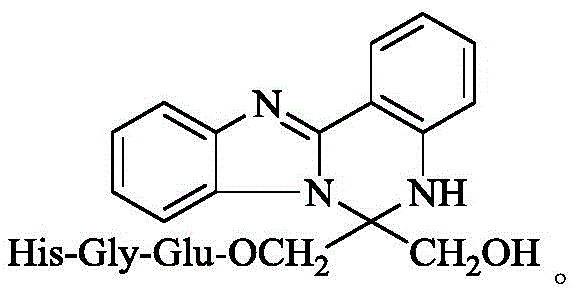
本发明涉及6-羟甲基-6-(His-Gly-Glu-OCH2)-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉,涉及它的制备方法,涉及它的免疫抑制活性。因而本发明涉及该化合物在制备免疫抑制药物中的应用。本发明属于生物医药领域。The present invention relates to 6-hydroxymethyl-6-(His-Gly-Glu-OCH 2 )-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline, and to its The preparation method relates to its immunosuppressive activity. Therefore, the present invention relates to the application of the compound in the preparation of immunosuppressive drugs. The invention belongs to the field of biomedicine.
背景技术Background technique
上世纪60年代,第一例器官移植手术的成功。经过数十年发展,使丧失器官功能的患者得到救治。可是,人体自身免疫系统对异体器官的免疫排斥反应严重影响患者近期和远期健康。最终,免疫排斥反应影响移植术后患者的生存时间。如何避免这种免疫排斥反应至关重要。虽然临床实施了各种措施,但是成效并不理想。以肾移植为例,虽然通过血浆置换和使用强效免疫抑制剂干预急性的细胞排斥和体液排斥可得到有效控制,但这种治疗对慢性排斥反应疗效果尚不理想。这样一来,发明新型免疫抑制剂受到广泛重视。同样, 发明新型免疫抑制剂的效果也不理想。出于对发明新型免疫抑制剂的兴趣,发明人一直致力于用肽修饰含N杂环。经过3年探索,发明人发现,发明人设计该化合物的目的是将两个具有免疫抑制活性的结构进行拼合,得到一种新型免疫抑制剂。发明人发现用羟甲基和 His-Gly-Glu-OCH2修饰5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉生成的下式的6-羟甲基 -6-(His-Gly-Glu-OCH2)-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉在小鼠耳后心肌移植实验模型中显示优秀的活性。根据这个发现,发明人提了出本发明。In the 1960s, the first successful organ transplant was performed. After decades of development, patients with loss of organ function can be treated. However, the immune rejection of the human body's own immune system to the allogeneic organs seriously affects the short-term and long-term health of patients. Ultimately, immune rejection affects the survival time of patients after transplantation. How to avoid this immune rejection is crucial. Although various clinical measures have been implemented, the results are not satisfactory. Taking kidney transplantation as an example, although acute cellular rejection and humoral rejection can be effectively controlled by plasma exchange and the use of potent immunosuppressive agents, the effect of this treatment on chronic rejection is not satisfactory. As a result, the invention of new immunosuppressants has received extensive attention. Similarly, the effect of inventing new immunosuppressants is not ideal. Interested in inventing novel immunosuppressants, the inventors have been working on modifying N-containing heterocycles with peptides. After 3 years of exploration, the inventor found that the purpose of designing the compound was to combine two structures with immunosuppressive activity to obtain a new type of immunosuppressant. The inventors found that 6-hydroxymethyl of the following formula was generated by modifying 5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline with hydroxymethyl and His-Gly-Glu-OCH 2 Radix-6-(His-Gly-Glu-OCH 2 )-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline showed in a mouse retroauricular myocardial transplantation experimental model Excellent activity. Based on this finding, the inventors have come up with the present invention.
发明内容SUMMARY OF THE INVENTION
本发明的第一个内容是提供下式的6-羟甲基-6-(His-Gly-Glu-OCH2)-5,6-二氢苯并[4,5] 咪唑[1,2-c]喹唑啉。The first content of the present invention is to provide 6-hydroxymethyl-6-(His-Gly-Glu-OCH 2 )-5,6-dihydrobenzo[4,5]imidazo[1,2- c] Quinazoline.
本发明的第二个内容是提供6-羟甲基-6-(His-Gly-Glu-OCH2)-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉的制备方法,该方法包括:The second aspect of the present invention is to provide 6-hydroxymethyl-6-(His-Gly-Glu-OCH 2 )-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinoline The preparation method of oxazoline, the method comprises:
1)合成(6,6-二羟甲基-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉;1) Synthesis of (6,6-dimethylol-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline;
2)合成6-羟甲基-6-[Boc-Glu(OBzl)-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉;2) Synthesis of 6-hydroxymethyl-6-[Boc-Glu(OBzl)-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline;
3)合成6-羟甲基-6-[Glu(OBzl)-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉;3) Synthesis of 6-hydroxymethyl-6-[Glu(OBzl)-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline;
4)采用二环己基碳二亚胺为缩合剂,1-羟基苯并三唑为催化剂的液相法合成Boc-His(Boc)-Gly;4) Synthesize Boc-His(Boc)-Gly by a liquid phase method using dicyclohexylcarbodiimide as a condensing agent and 1-hydroxybenzotriazole as a catalyst;
5)采用二环己基碳二亚胺为缩合剂,1-羟基苯并三唑为催化剂的液相法将6-羟甲基 -6-[Glu(OBzl)-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉与Boc-His(Boc)-Gly缩合成6- 羟甲基-6-[Boc-His(Boc)-Gly-Glu(OBzl)-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉;5) 6-hydroxymethyl-6-[Glu(OBzl)-OCH 2 ]-5,6- Condensation of dihydrobenzo[4,5]imidazo[1,2-c]quinazoline with Boc-His(Boc)-Gly to 6-hydroxymethyl-6-[Boc-His(Boc)-Gly-Glu (OBzl)-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline;
6)合成6-羟甲基-6-[Boc-His(Boc)-Gly-Glu-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉;6) Synthesis of 6-hydroxymethyl-6-[Boc-His(Boc)-Gly-Glu-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazole Lin;
7)合成6-羟甲基-6-(His-Gly-Glu-OCH2)-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉。7) Synthesis of 6-hydroxymethyl-6-(His-Gly-Glu-OCH 2 )-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline.
本发明的第三个内容是评价6-羟甲基-6-(His-Gly-Glu-OCH2)-5,6-二氢苯并[4,5]咪唑[1,2-c] 喹唑啉免疫抑制的作用。The third aspect of the present invention is to evaluate 6-hydroxymethyl-6-(His-Gly-Glu-OCH 2 )-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinoline The immunosuppressive effects of oxazolines.
附图说明Description of drawings
图1.6-羟甲基-6-(His-Gly-Glu-OCH2)-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉的合成路线.(i) 2M盐酸溶液,1,3-二羟基丙酮;(ii)二环己基碳二亚胺,1-羟基苯并三唑,Boc-Glu(OBzl), N-甲基吗啉;(iii)氯化氢的乙酸乙酯溶液(4M);(iv)二环己基碳二亚胺,1-羟基苯并三唑, N-甲基吗啉;(v)Pd/C,H2。Figure 1. Synthetic route of 6-hydroxymethyl-6-(His-Gly-Glu-OCH 2 )-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline.(i ) 2M hydrochloric acid solution, 1,3-dihydroxyacetone; (ii) dicyclohexylcarbodiimide, 1-hydroxybenzotriazole, Boc-Glu(OBzl), N-methylmorpholine; (iii) hydrogen chloride in ethyl acetate (4M); (iv) dicyclohexylcarbodiimide, 1-hydroxybenzotriazole, N-methylmorpholine; (v) Pd/C, H 2 .
具体实施方式Detailed ways
为了进一步阐述本发明,下面给出一系列实施例。这些实施例完全是例证性的,它们仅用来对本发明进行具体描述,不应当理解为对本发明的限制。In order to further illustrate the present invention, a series of examples are given below. These examples are purely illustrative, they are only used to specifically describe the present invention, and should not be construed as limiting the present invention.
实施例1制备(6,6-二羟甲基-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉(1)Example 1 Preparation of (6,6-dimethylol-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline (1)
将6.40g(30.6mmol)2-(2-氨基苯基)-1H-苯并咪唑与100mL甲醇悬浮,向该悬浮液加入2.76g(30.7mmol)1,3二羟基丙酮。反应混合物用2M盐酸调pH为2,此时反应液变为绿色。反应混合物室温搅拌12小时,TLC(二氯甲烷/甲醇=20/1)显示原料点消失,并有大量黄绿色固体析出。终止反应,向反应液中滴加4M氢氧化钠水溶液将反应液的pH调至中性,过滤,得到7.03g标题化合物,为黄绿色固体。滤液减压浓缩并过滤,再得到2.52g 标题化合物,为深绿色固体。本步反应的产率为100%。ESI-MS(m/e):282[M+H]+。 Mp 214-215℃。1H-NMR(300MHz,DMSO-d6):δ/ppm=7.86(d,J=7.2Hz,1H),7.69(m,1H),7.59(m,1H),7.15(m,3H),6.85(d,J=8.1Hz,1H),6.70(m,2H),5.26(t,J=5.4,2H),4.08(dd,J1=6.0Hz,J2=5.7Hz,2H),3.82(dd,J1=5.7Hz,J2=6.0Hz,2H)。6.40 g (30.6 mmol) of 2-(2-aminophenyl)-1H-benzimidazole was suspended in 100 mL of methanol, and 2.76 g (30.7 mmol) of 1,3-dihydroxyacetone was added to the suspension. The pH of the reaction mixture was adjusted to 2 with 2M hydrochloric acid, at which point the reaction solution turned green. The reaction mixture was stirred at room temperature for 12 hours, TLC (dichloromethane/methanol=20/1) showed that the starting point disappeared, and a large amount of yellow-green solid was precipitated. The reaction was terminated, and 4M aqueous sodium hydroxide solution was added dropwise to the reaction solution to adjust the pH of the reaction solution to neutrality, and filtered to obtain 7.03 g of the title compound as a yellow-green solid. The filtrate was concentrated under reduced pressure and filtered to give 2.52 g of the title compound as a dark green solid. The yield of this step is 100%. ESI-MS (m/e): 282 [M+H] + . Mp 214-215°C. 1 H-NMR (300MHz, DMSO-d 6 ): δ/ppm=7.86(d, J=7.2Hz, 1H), 7.69(m, 1H), 7.59(m, 1H), 7.15(m, 3H), 6.85(d,J=8.1Hz,1H),6.70(m,2H),5.26(t,J= 5.4,2H ),4.08(dd,J1 = 6.0Hz,J2=5.7Hz,2H),3.82 (dd, J 1 =5.7 Hz, J 2 =6.0 Hz, 2H).
实施例2制备6-羟甲基-6-[Boc-Glu(OBzl)-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉(2)Example 2 Preparation of 6-hydroxymethyl-6-[Boc-Glu(OBzl)-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline (2 )
将337mg(1.00mmol)Boc-Glu(OBzl),250mg(1.20mmol)二环己基碳二亚胺和135mg(1.00mmol)1-羟基苯并三唑用10mL DMF溶解。得到的溶液于冰浴搅拌30分钟,之后加1.12g(4.00mmol)6,6-二羟甲基-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉(1)。反应混合物用 N-甲基吗啉调pH为7-8。之后,反应混合物室温搅拌12小时,TLC(二氯甲烷/甲醇=20/1)显示Boc-Glu(OBzl)消失。反应混合物减压浓缩,残留物用50mL乙酸乙酯溶解,过滤,滤液用饱和碳酸氢钠水溶液洗(30mL×3),饱和氯化钠水溶液洗(30mL×3),用无水硫酸钠干燥12小时,过滤,滤液减压浓缩,残留物用二氯甲烷稀释,超声,过滤,滤液减压浓缩,残留物经硅胶柱层析纯化(二氯甲烷/甲醇=80/1),得到370mg(62%)标题化合物,为淡黄绿色固体。ESI-MS(m/e):635[M+Cl]-。Mp 80-82℃;1H-NMR(300 MHz,DMSO-d6):δ/ppm=7.86(d,J=7.8Hz,1H),7.72(dd,J1=7.8Hz,J2=19.5Hz,1H), 7.59(m,1H),7.35(s,5H),7.14(m,4H),6.96(d,J=19.5Hz 1H),6.82(t,J=6.3Hz,1H), 5.54(m,1H),5.03(s,2H),4.79(m,1H),4.45(m,1H),4.16(m,1H),3.95(m,1H),3.78(m, 1H),2.13(m,1H),1.45(m,1H),1.32(s,9H)。337 mg (1.00 mmol) of Boc-Glu(OBzl), 250 mg (1.20 mmol) of dicyclohexylcarbodiimide and 135 mg (1.00 mmol) of 1-hydroxybenzotriazole were dissolved in 10 mL of DMF. The resulting solution was stirred in an ice bath for 30 minutes, after which 1.12 g (4.00 mmol) of 6,6-dimethylol-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazole was added Phosphine (1). The reaction mixture was adjusted to pH 7-8 with N-methylmorpholine. After that, the reaction mixture was stirred at room temperature for 12 hours, and TLC (dichloromethane/methanol=20/1) showed that Boc-Glu(OBzl) disappeared. The reaction mixture was concentrated under reduced pressure, the residue was dissolved in 50 mL of ethyl acetate, filtered, and the filtrate was washed with saturated aqueous sodium bicarbonate solution (30 mL × 3), washed with saturated aqueous sodium chloride solution (30 mL × 3), and dried over anhydrous sodium sulfate for 12 hours, filtered, the filtrate was concentrated under reduced pressure, the residue was diluted with dichloromethane, sonicated, filtered, the filtrate was concentrated under reduced pressure, and the residue was purified by silica gel column chromatography (dichloromethane/methanol=80/1) to obtain 370 mg (62 %) title compound as a pale yellow-green solid. ESI-MS (m/e): 635 [M+Cl] − . Mp 80-82°C; 1 H-NMR (300 MHz, DMSO-d 6 ): δ/ppm=7.86 (d, J=7.8 Hz, 1H), 7.72 (dd, J 1 =7.8 Hz, J 2 =19.5 Hz, 1H), 7.59(m, 1H), 7.35(s, 5H), 7.14(m, 4H), 6.96(d, J=19.5Hz 1H), 6.82(t, J=6.3Hz, 1H), 5.54 (m, 1H), 5.03(s, 2H), 4.79(m, 1H), 4.45(m, 1H), 4.16(m, 1H), 3.95(m, 1H), 3.78(m, 1H), 2.13( m, 1H), 1.45 (m, 1H), 1.32 (s, 9H).
实施例3制备6-羟甲基-6-[Glu(OBzl)-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉(3)Example 3 Preparation of 6-hydroxymethyl-6-[Glu(OBzl)-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline (3)
将300mg(0.500mmol)6-羟甲基-6-[Boc-Glu(OBzl)-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c] 喹唑啉(2)用1mL无水乙酸乙酯溶解,冰浴下加5mL氯化氢乙酸乙酯溶液(4M),反应4 小时后TLC(二氯甲烷/甲醇=20/1)显示化合物2消失,反应液减压浓缩,残留物用无水乙酸乙酯溶解,再减压浓缩,残留物再用无水乙酸乙酯溶解。该操作重复3次。残留物分散在10mL无水乙醚中,静置,弃乙醚,得到280mg(100%)标题化合物,为绿色固体。 ESI-MS(m/e):535[M+Cl]-。300 mg (0.500 mmol) of 6-hydroxymethyl-6-[Boc-Glu(OBzl)-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline (2) Dissolve with 1 mL of anhydrous ethyl acetate, add 5 mL of hydrogen chloride ethyl acetate solution (4M) under ice bath, after 4 hours of reaction, TLC (dichloromethane/methanol=20/1) shows that
实施例4制备Boc-His(Boc)-Gly-OBzlExample 4 Preparation of Boc-His(Boc)-Gly-OBzl
将5.23mg(14.7mmol)的Boc-His(Boc),3.70g(16.0mmol)二环己基碳二亚胺及2.00g (15.0mmol)1-羟基苯并三唑用150mL无水四氢呋喃溶解,0℃搅拌30分钟。向反应液中加 3.00g(15.0mmol)Gly-OBzl,反应液用N-甲基吗啉调pH为7-8,室温搅拌12小时,TLC(二氯甲烷/甲醇=20/1)显示反应完成。反应混合物减浓缩,残留物用100mL乙酸乙酯稀释,过滤,滤液用饱和碳酸氢钠水溶液洗(30mL×3),饱和氯化钠水溶液洗(30mL×3),用无水硫酸钠干燥12小时,过滤,滤液减压浓缩,残留物用硅胶柱层析纯化(二氯甲烷/甲醇=80/1),得到2.23g(30%)标题化合物,为黄色油状物。ESI-MS(m/e):503[M+H]+。5.23 mg (14.7 mmol) of Boc-His (Boc), 3.70 g (16.0 mmol) of dicyclohexylcarbodiimide and 2.00 g (15.0 mmol) of 1-hydroxybenzotriazole were dissolved in 150 mL of anhydrous tetrahydrofuran, 0 °C was stirred for 30 minutes. Add 3.00g (15.0mmol) Gly-OBzl to the reaction solution, adjust the pH of the reaction solution to 7-8 with N-methylmorpholine, stir at room temperature for 12 hours, TLC (dichloromethane/methanol=20/1) shows the reaction Finish. The reaction mixture was deconcentrated, the residue was diluted with 100 mL of ethyl acetate, filtered, the filtrate was washed with saturated aqueous sodium bicarbonate solution (30 mL×3), washed with saturated aqueous sodium chloride solution (30 mL×3), and dried over anhydrous sodium sulfate for 12 hours , filtered, the filtrate was concentrated under reduced pressure, and the residue was purified by silica gel column chromatography (dichloromethane/methanol=80/1) to obtain 2.23 g (30%) of the title compound as a yellow oil. ESI-MS (m/e): 503 [M+H] + .
实施例5制备Boc-His(Boc)-GlyExample 5 Preparation of Boc-His(Boc)-Gly
将1.32g(2.64mmoL)Boc-His(Boc)-Gly-OBzl用100mL甲醇溶解,向得到的溶液中加 140mg Pd/C,通氢气,室温搅拌12小时,TLC(二氯甲烷/甲醇=20/1)显示反应完全。反应液过滤,滤液减压浓缩,得到1.16g(100%)标题化合物,为无色固体。ESI-MS(m/e):413 [M+H]+。Dissolve 1.32 g (2.64 mmol) of Boc-His(Boc)-Gly-OBzl in 100 mL of methanol, add 140 mg of Pd/C to the resulting solution, pass hydrogen gas, stir at room temperature for 12 hours, TLC (dichloromethane/methanol=20 /1) shows that the reaction is complete. The reaction solution was filtered, and the filtrate was concentrated under reduced pressure to obtain 1.16 g (100%) of the title compound as a colorless solid. ESI-MS (m/e): 413 [M+H] + .
实施例6制备6-羟甲基-6-[Boc-His(Boc)-Gly-Glu(OBzl)-OCH2]-5,6-二氢苯并[4,5]咪唑 [1,2-c]喹唑啉(4)Example 6 Preparation of 6-hydroxymethyl-6-[Boc-His(Boc)-Gly-Glu(OBzl)-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazo[1,2- c]quinazoline (4)
冰浴下将240mg(0.60mmol)的Boc-His(Boc)-Gly,130mg(0.630mmol)二环己基碳二亚胺及75.0mg(0.550mmol)1-羟基苯并三唑用10mL无水四氢呋喃溶解,搅拌30分钟,向反应液中加270mg(0.60mmol)6-羟甲基-6-[Glu(OBzl)-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c] 喹唑啉(3)。反应混合物用N-甲基吗啉调pH值8,室温搅拌6小时,TLC(二氯甲烷:甲醇=15:1)显示化合物3消失。反应液减压浓缩,残留物用50mL乙酸乙酯稀释,过滤,滤液用饱和碳酸氢钠水溶液洗(30mL×3),饱和氯化钠水溶液洗(30mL×3),用无水硫酸钠干燥12 小时,过滤,滤液减压浓缩,得到的淡黄色固体用硅胶柱层析纯化(二氯甲烷/甲醇=30/1),得到81.0mg(41%)标题化合物,为淡黄色固体。ESI-MS(m/e):896[M+H]+。 Mp 110-111℃。1H-NMR(300MHz,DMSO-d6):δ/ppm=8.18(d,J=7.2Hz,1H),8.06(m,2H),7.87(d,J=6.9Hz,1H),7.73(m,1H),7.59(m,1H),7.34(m,5H),7.16 (m,4H),6.89(m,2H),6.82(d,J=6.9Hz,1H),6.72(t,J=6.9Hz,1H),5.55(q,J=6.6Hz,1 H),5.02(s,2H),4.56(m,2H),4.20(m,2H),4.04(m,2H),3.62(m,2H),2.87(m,1H),2.70 (m,1H),2.15(t,J=8.1Hz,2H),1.53(s,11H),1.32(s,9H)。13C-NMR(300MHz, DMSO-d6):δ/ppm=172.1,171.4,169.4,155.7,148.0,147.9,147.1,144.4,143.5,139.9,137.0, 136.6,133.4,133.3,131.9,128.8,128.4,124.9,122.5,122.3,119.2,118.1,144.8,133.0,122.7, 111.4,78.7,76.4,65.9,63.9,63.6,54,2,51.4,42.3,30.8,30.0,28.5,27.8,26.0。Under ice bath, 240 mg (0.60 mmol) of Boc-His(Boc)-Gly, 130 mg (0.630 mmol) of dicyclohexylcarbodiimide and 75.0 mg (0.550 mmol) of 1-hydroxybenzotriazole were mixed with 10 mL of anhydrous tetrahydrofuran. Dissolve, stir for 30 minutes, add 270 mg (0.60 mmol) of 6-hydroxymethyl-6-[Glu(OBzl)-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazole[1 to the reaction solution ,2-c]quinazoline (3). The reaction mixture was adjusted to pH 8 with N-methylmorpholine, stirred at room temperature for 6 hours, TLC (dichloromethane:methanol=15:1) showed that
实施例7制备6-羟甲基-6-[Boc-His(Boc)-Gly-Glu-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉(5)Example 7 Preparation of 6-hydroxymethyl-6-[Boc-His(Boc)-Gly-Glu-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinoline oxazoline (5)
用实施例5的方法次60.0mg(0.067mmoL)的6-羟甲基 -6-[Boc-His(Boc)-Gly-Glu(OBzl)-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉(4)得到55 mg(100%)标题化合物,为绿色固体。ESI-MS(m/e):806[M+H]+。60.0 mg (0.067 mmol) of 6-hydroxymethyl-6-[Boc-His(Boc)-Gly-Glu(OBzl) -OCH2 ]-5,6-dihydrobenzo[ 4,5]imidazo[1,2-c]quinazoline (4) gave 55 mg (100%) of the title compound as a green solid. ESI-MS (m/e): 806 [M+H] + .
实施例8制备6-羟甲基-6-[His-Gly-Glu-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉(6)Example 8 Preparation of 6-hydroxymethyl-6-[His-Gly-Glu-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazo[1,2-c]quinazoline (6)
冰浴下将55.0mg(0.067mmol)6-羟甲基-6-[Boc-His(Boc)-Gly-Glu-OCH2]-5,6-二氢苯并[4,5]咪唑[1,2-c]喹唑啉(5)用2mL氯化氢的乙酸乙酯溶液(4M)溶解并搅拌4小时。TLC (乙酸乙酯/水/冰醋酸=2/1/1)显示化合物5消失。反应液减压浓缩,残留物加无水乙酸乙酯溶解,得到的溶液再减压浓缩。该操作重复3次。残留物用无水乙醚洗3次。得到绿色固体用C18柱纯化(水/甲醇=80/20),馏分冻干得到9.60mg(26%)标题化合物,为绿色固体。 ESI-MS(m/e):606[M+H]+。Mp 181-183℃。IR(cm-1):3029.60, 2321.11,2166.95,2109.11,1741.91,1682.54,1628.73,1568.08,1531.98,1492.60,1386.95, 1337.38,1261.81,1186.85,1073,10,1019.19,823.60,750.73,622.09。1H-NMR(300MHz, DMSO-d6):δ/ppm=8.89(s,2H),8.53(dd,J1=6.3Hz,J2=15.0Hz,1H),7.89(d,J=7.5Hz, 1H),7.74(dd,J1=6.3Hz,J2=15.0Hz,1H),7.62(d,J=6.0Hz,1H),7.44(s,1H),7.20(m,4 H),6.88(m,1H),6.76(q,J=6.6Hz,1H),5.62(m,1H),4.75(m,qH),,4.47(m,1H),4.18 (m,3H),3.97(m,2H),3.81(m,1H),3.62(m,2H),2.04(m,2H),1.53(m,2H)。13C-NMR (300MHz,DMSO-d6):δ/ppm=174.1,173.8,171.4,169.2,168.1,147.4,144.1,134.6,133.3, 132.4,126.9,125.4,123.6,118.5,118.3,114.9,113.6,77.2,65.8,63.8,63.5,51.6,42.0,30.1, 26.6,26.1。55.0 mg (0.067 mmol) of 6-hydroxymethyl-6-[Boc-His(Boc)-Gly-Glu-OCH 2 ]-5,6-dihydrobenzo[4,5]imidazo[1 ,2-c]quinazoline (5) was dissolved with 2 mL of hydrogen chloride in ethyl acetate (4M) and stirred for 4 hours. TLC (ethyl acetate/water/glacial acetic acid = 2/1/1) showed that
实施例9评价化合物6的免疫抑制作用Example 9 Evaluation of the immunosuppressive effect of
雄性Balb/c小鼠(20±2g)用10%乌拉坦(10mg/10g体重)腹腔注射麻醉。用75%酒精为小鼠耳廓局部消毒。于耳廓背侧中线约1/3处剪开一个与耳廓中线垂直的长3-4毫米口, 注意不要损伤耳廓静脉。通过该开口向耳尖方向钝性分离皮下组织使之成一人工管腔。将 C57bl/6j供心乳鼠于碎冰中冻一分钟,用75%酒精消毒皮肤,剖胸后摘取心脏。将心脏置于铺有生理盐水润湿纱布的表面皿中内搏动,使排净心腔中的余血。将供乳鼠心脏纵向剖成基本等大的肌纤维呈斜面的两瓣心肌。将心肌填入Balb/c受体鼠的人工管腔。操作时心肌组织离体时间不超过2分钟。轻按填有心肌的人工管腔,排出多余的生理盐水及气泡, 使移植心肌贴紧受体鼠耳部周围组织。心肌移植术的当天开始口服化合物6(剂量为0.1μmol/kg/天)或口服环孢菌素A(剂量为2.5μmol/kg/天)或口服生理盐水。连续给药至移植心肌的心电图消失。Male Balb/c mice (20±2 g) were anesthetized with 10% urethane (10 mg/10 g body weight) by intraperitoneal injection. Topically sterilize mouse auricles with 75% alcohol. Cut a 3-4 mm long incision perpendicular to the midline of the auricle at about 1/3 of the dorsal midline of the auricle, taking care not to damage the auricular vein. The subcutaneous tissue is bluntly dissected towards the ear tip through this opening to create an artificial lumen. The C57bl/6j donor rat was frozen in crushed ice for one minute, the skin was disinfected with 75% alcohol, and the heart was harvested after thoracotomy. The heart was placed in a watch dish covered with saline-wet gauze and beat to drain the remaining blood in the heart chamber. The donor rat heart was longitudinally sectioned into two myocardium with substantially equal muscle fibers and inclined planes. The myocardium was filled into the artificial lumen of Balb/c recipient mice. During the operation, the myocardial tissue was isolated for no more than 2 minutes. Gently press the artificial lumen filled with myocardium to drain excess saline and air bubbles, so that the transplanted myocardium is close to the tissue around the ear of the recipient mouse. Compound 6 (dose of 0.1 μmol/kg/day) or cyclosporine A (dose of 2.5 μmol/kg/day) or normal saline was administered orally on the day of myocardial transplantation. Continuous administration until the electrocardiogram of the transplanted myocardium disappeared.
心肌移植术的第七日起,每天用生物信号二道仪测量移植心肌组织的心电信号(若第七日至以后都没有心电信号则判定心肌移植手术失败)。测量异位心电图时,正负电极分别置于移植心肌组织的两侧,接地极连接在小鼠后肢。移植心肌组织心电信号全部消失后结束异位心电图测量,统计移植心肌存活时间(均值±SD),结果见表1。可见在0.1μmol/kg/天剂量下化合物6延长移植心肌存活的活性与2.5μmol/kg/天剂量下的环孢菌素A没有显著性差异。本发明有突出的技术效果。From the seventh day of myocardial transplantation, the ECG signal of the transplanted myocardial tissue was measured with a biosignal device every day (if there was no ECG signal from the seventh day to the later, it was judged that the myocardial transplantation operation failed). When measuring ectopic ECG, the positive and negative electrodes were placed on both sides of the transplanted myocardial tissue, and the ground electrodes were connected to the hind limbs of mice. After all the ECG signals of the transplanted myocardial tissue disappeared, the ectopic ECG measurement was terminated, and the survival time of the transplanted myocardium was counted (mean ± SD). The results are shown in Table 1. It can be seen that the activity of
表1化合物6对小鼠耳后心肌移植存活时间的影响Table 1 The effect of
a)与生理盐水比P<0.01;b)与生理盐水相比P<0.05,与环孢菌素A比P>0.05;n=12。a) Compared with normal saline, P<0.01; b) Compared with normal saline, P<0.05, and compared with cyclosporin A, P>0.05; n=12.
Claims (3)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910452670.XA CN112010927B (en) | 2019-05-28 | 2019-05-28 | EGH and hydroxymethyl modified benzimidazole quinazoline, and synthesis, activity and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910452670.XA CN112010927B (en) | 2019-05-28 | 2019-05-28 | EGH and hydroxymethyl modified benzimidazole quinazoline, and synthesis, activity and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN112010927A true CN112010927A (en) | 2020-12-01 |
| CN112010927B CN112010927B (en) | 2021-10-26 |
Family
ID=73501299
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910452670.XA Expired - Fee Related CN112010927B (en) | 2019-05-28 | 2019-05-28 | EGH and hydroxymethyl modified benzimidazole quinazoline, and synthesis, activity and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN112010927B (en) |
Cited By (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN118562020A (en) * | 2024-06-20 | 2024-08-30 | 首都医科大学 | Glu-His-Gly trimer, its preparation and application in enhancing immunity |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4000275A (en) * | 1969-11-24 | 1976-12-28 | Eli Lilly And Company | Immunosuppressants |
| US20020025968A1 (en) * | 1998-04-15 | 2002-02-28 | Rifat Pamukcu | Method for inhibiting neoplastic cells and related conditions by exposure to 4-aminoquinazoline derivatives |
| CN102796171A (en) * | 2011-05-27 | 2012-11-28 | 首都医科大学 | Double saturated aliphatic chain alcohol His-Gly-Glu-Asp tetrapeptide esters and preparation method and application thereof |
| CN104211761A (en) * | 2013-06-05 | 2014-12-17 | 首都医科大学 | Conjugates of saturated aliphatic chain alcohol, dexamethasone, and His-Gly-Glu, preparation, nano structure, and applications thereof |
| US20160311828A1 (en) * | 2013-12-20 | 2016-10-27 | Laboratorios Del Dr. Esteve S.A. | Fused imidazolyl derivatives, their preparation and use as medicaments |
| CN106317185A (en) * | 2015-06-23 | 2017-01-11 | 首都医科大学 | His-Gly-Tyr-Asp and synthesis, activity and application thereof |
| CN106349244A (en) * | 2015-07-13 | 2017-01-25 | 首都医科大学 | Benzimidazoloquinazoline dimethoxyphenoxyacetyl-AA-OBzl, its synthesis, activity and applications |
| CN107629056A (en) * | 2015-06-23 | 2018-01-26 | 首都医科大学 | Benzimidazole quinazoline Salicyloy lamino acid benzyl ester, its preparation, antitumor activity and application |
-
2019
- 2019-05-28 CN CN201910452670.XA patent/CN112010927B/en not_active Expired - Fee Related
Patent Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US4000275A (en) * | 1969-11-24 | 1976-12-28 | Eli Lilly And Company | Immunosuppressants |
| US20020025968A1 (en) * | 1998-04-15 | 2002-02-28 | Rifat Pamukcu | Method for inhibiting neoplastic cells and related conditions by exposure to 4-aminoquinazoline derivatives |
| CN102796171A (en) * | 2011-05-27 | 2012-11-28 | 首都医科大学 | Double saturated aliphatic chain alcohol His-Gly-Glu-Asp tetrapeptide esters and preparation method and application thereof |
| CN104211761A (en) * | 2013-06-05 | 2014-12-17 | 首都医科大学 | Conjugates of saturated aliphatic chain alcohol, dexamethasone, and His-Gly-Glu, preparation, nano structure, and applications thereof |
| US20160311828A1 (en) * | 2013-12-20 | 2016-10-27 | Laboratorios Del Dr. Esteve S.A. | Fused imidazolyl derivatives, their preparation and use as medicaments |
| CN106317185A (en) * | 2015-06-23 | 2017-01-11 | 首都医科大学 | His-Gly-Tyr-Asp and synthesis, activity and application thereof |
| CN107629056A (en) * | 2015-06-23 | 2018-01-26 | 首都医科大学 | Benzimidazole quinazoline Salicyloy lamino acid benzyl ester, its preparation, antitumor activity and application |
| CN106349244A (en) * | 2015-07-13 | 2017-01-25 | 首都医科大学 | Benzimidazoloquinazoline dimethoxyphenoxyacetyl-AA-OBzl, its synthesis, activity and applications |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN118562020A (en) * | 2024-06-20 | 2024-08-30 | 首都医科大学 | Glu-His-Gly trimer, its preparation and application in enhancing immunity |
| CN118562020B (en) * | 2024-06-20 | 2025-07-29 | 首都医科大学 | Glu-His-Gly trimer, preparation and application thereof in enhancing immunity |
Also Published As
| Publication number | Publication date |
|---|---|
| CN112010927B (en) | 2021-10-26 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US20230312644A1 (en) | Compounds having triple activities of thrombolysis, antithrombotic and radical scavenging | |
| CN102883736A (en) | Peptides for promoting angiogenesis and uses thereof | |
| CN112010927A (en) | EGH and hydroxymethyl modified benzimidazole quinazoline, and synthesis, activity and application thereof | |
| CN112010926B (en) | KGH and hydroxymethyl modified benzimidazole quinazolines, synthesis, activity and applications thereof | |
| CN112010925A (en) | GDE and hydroxymethyl modified benzimidazole quinazoline, synthesis, activity and application thereof | |
| CN109912693A (en) | RGDS-modified heptacyclic aldehyde, its synthesis, antithrombotic activity and application | |
| WO2016062234A1 (en) | Octapeptide modified dexamethasone, and preparation, nanostructure and use thereof | |
| CN110551119B (en) | 6-Aminoamido-n-hexanoylcarbolinecarboxylic acid benzyl ester, its preparation and application | |
| CN109912590B (en) | Amino acid and amino-n-hexanoic acid modified benzyl carboline carboxylate, its preparation, activity and application | |
| CN108948146B (en) | 1R-methyl-β-tetrahydrocarbolinyl-K(ARPAK)-RGDV, its synthesis, activity and application | |
| CN110551121B (en) | Glutaminamido-n-hexanoylcarboline carboxylate benzyl ester, its preparation, activity and application | |
| CN109912588B (en) | 6-amino amido n-hexanoyl carboline benzyl carboxylate, preparation, activity and application thereof | |
| CN110577583B (en) | RGDF-modified heptacyclic aldehyde, its synthesis, antithrombotic activity and application | |
| CN108929320B (en) | 3R-indolylmethyl-6R-oxazolidinone modified piperazine-2, 5-dione, synthesis, activity and application thereof | |
| CN109912692A (en) | YIGSK-modified heptacyclic aldehyde, its preparation, antithrombotic activity and application | |
| CN110551120B (en) | 6-amino amido n-hexanoyl carboline benzyl carboxylate, preparation, activity and application thereof | |
| CN115477655B (en) | Preparation and application of dioxane-tetrahydrocarboline-3-carboxylic acid amino acid docked with tumor necrosis factor-α | |
| CN110577578B (en) | 1S-methyl-β-tetrahydrocarbolinyl-K (RPAK) and its synthesis, activity and application | |
| CN112300245B (en) | RGDS and theanine co-modified 5-fluorouracil, and synthesis, activity and application thereof | |
| CN112390854B (en) | 5-Fluorouracil co-modified with theanine and RGDS, its synthesis, activity and application | |
| CN110577569B (en) | RGDV-modified heptacyclic aldehyde, synthesis, antithrombotic activity and application thereof | |
| CN112110987B (en) | 5-fluorouracil modified by asparaginyl theanine and phenylalanine, synthesis, activity and application thereof | |
| CN112300246B (en) | 5-fluorouracil modified by aspartyl theanine RGDS, and synthesis, activity and application thereof | |
| CN108976280A (en) | The amine-modified LDV of fat, synthesis, activity and application | |
| CN112300244B (en) | 5-fluorouracil modified by theanine alone or together with RGDS, and synthesis, activity and application thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20211026 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |