CN110551119B - 6-Aminoamido-n-hexanoylcarbolinecarboxylic acid benzyl ester, its preparation and application - Google Patents
6-Aminoamido-n-hexanoylcarbolinecarboxylic acid benzyl ester, its preparation and application Download PDFInfo
- Publication number
- CN110551119B CN110551119B CN201810561635.7A CN201810561635A CN110551119B CN 110551119 B CN110551119 B CN 110551119B CN 201810561635 A CN201810561635 A CN 201810561635A CN 110551119 B CN110551119 B CN 110551119B
- Authority
- CN
- China
- Prior art keywords
- residues
- tetrahydro
- carboline
- amino
- hexanoyl
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 238000002360 preparation method Methods 0.000 title claims abstract description 27
- -1 benzyl ester Chemical class 0.000 title claims description 8
- 239000002253 acid Substances 0.000 title description 2
- 230000000259 anti-tumor effect Effects 0.000 claims abstract description 12
- WHUUTDBJXJRKMK-VKHMYHEASA-N L-glutamic acid Chemical compound OC(=O)[C@@H](N)CCC(O)=O WHUUTDBJXJRKMK-VKHMYHEASA-N 0.000 claims abstract description 10
- 230000003110 anti-inflammatory effect Effects 0.000 claims abstract description 8
- 239000003814 drug Substances 0.000 claims abstract description 7
- MTCFGRXMJLQNBG-REOHCLBHSA-N (2S)-2-Amino-3-hydroxypropansäure Chemical group OC[C@H](N)C(O)=O MTCFGRXMJLQNBG-REOHCLBHSA-N 0.000 claims abstract description 5
- ONIBWKKTOPOVIA-BYPYZUCNSA-N L-Proline Chemical compound OC(=O)[C@@H]1CCCN1 ONIBWKKTOPOVIA-BYPYZUCNSA-N 0.000 claims abstract description 5
- AGPKZVBTJJNPAG-WHFBIAKZSA-N L-isoleucine Chemical compound CC[C@H](C)[C@H](N)C(O)=O AGPKZVBTJJNPAG-WHFBIAKZSA-N 0.000 claims abstract description 5
- ROHFNLRQFUQHCH-YFKPBYRVSA-N L-leucine Chemical compound CC(C)C[C@H](N)C(O)=O ROHFNLRQFUQHCH-YFKPBYRVSA-N 0.000 claims abstract description 5
- KDXKERNSBIXSRK-YFKPBYRVSA-N L-lysine Chemical compound NCCCC[C@H](N)C(O)=O KDXKERNSBIXSRK-YFKPBYRVSA-N 0.000 claims abstract description 5
- FFEARJCKVFRZRR-BYPYZUCNSA-N L-methionine Chemical compound CSCC[C@H](N)C(O)=O FFEARJCKVFRZRR-BYPYZUCNSA-N 0.000 claims abstract description 5
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 claims description 48
- 239000000243 solution Substances 0.000 claims description 39
- 150000001875 compounds Chemical class 0.000 claims description 31
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Substances O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 20
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 claims description 18
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 15
- QTBSBXVTEAMEQO-UHFFFAOYSA-N acetic acid Substances CC(O)=O QTBSBXVTEAMEQO-UHFFFAOYSA-N 0.000 claims description 15
- 238000000034 method Methods 0.000 claims description 14
- LWXXGYOXIZBQRH-QHCPKHFHSA-N benzyl (3S)-2-(6-aminohexanoyl)-1,3,4,9-tetrahydropyrido[3,4-b]indole-3-carboxylate Chemical compound C1[C@H](N(CC2=C1C3=CC=CC=C3N2)C(=O)CCCCCN)C(=O)OCC4=CC=CC=C4 LWXXGYOXIZBQRH-QHCPKHFHSA-N 0.000 claims description 12
- PSTDRLUXXSVTJJ-KRWDZBQOSA-N benzyl (3s)-2,3,4,9-tetrahydro-1h-pyrido[3,4-b]indole-3-carboxylate Chemical compound O=C([C@H]1NCC2=C(C3=CC=CC=C3N2)C1)OCC1=CC=CC=C1 PSTDRLUXXSVTJJ-KRWDZBQOSA-N 0.000 claims description 10
- NPZTUJOABDZTLV-UHFFFAOYSA-N hydroxybenzotriazole Substances O=C1C=CC=C2NNN=C12 NPZTUJOABDZTLV-UHFFFAOYSA-N 0.000 claims description 10
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 claims description 9
- WSFSSNUMVMOOMR-UHFFFAOYSA-N Formaldehyde Chemical compound O=C WSFSSNUMVMOOMR-UHFFFAOYSA-N 0.000 claims description 8
- ASOKPJOREAFHNY-UHFFFAOYSA-N 1-Hydroxybenzotriazole Chemical compound C1=CC=C2N(O)N=NC2=C1 ASOKPJOREAFHNY-UHFFFAOYSA-N 0.000 claims description 7
- QOSSAOTZNIDXMA-UHFFFAOYSA-N Dicylcohexylcarbodiimide Chemical compound C1CCCCC1N=C=NC1CCCCC1 QOSSAOTZNIDXMA-UHFFFAOYSA-N 0.000 claims description 7
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 6
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 claims description 6
- 229910000041 hydrogen chloride Inorganic materials 0.000 claims description 6
- DYHSDKLCOJIUFX-UHFFFAOYSA-N tert-butoxycarbonyl anhydride Chemical compound CC(C)(C)OC(=O)OC(=O)OC(C)(C)C DYHSDKLCOJIUFX-UHFFFAOYSA-N 0.000 claims description 6
- SLXKOJJOQWFEFD-UHFFFAOYSA-N 6-aminohexanoic acid Chemical compound NCCCCCC(O)=O SLXKOJJOQWFEFD-UHFFFAOYSA-N 0.000 claims description 5
- RYHBNJHYFVUHQT-UHFFFAOYSA-N 1,4-Dioxane Chemical compound C1COCCO1 RYHBNJHYFVUHQT-UHFFFAOYSA-N 0.000 claims description 4
- YEXNNJWRYPGWGU-UHFFFAOYSA-N 2-amino-7-[(2-methylpropan-2-yl)oxy]-7-oxoheptanoic acid Chemical compound CC(C)(C)OC(=O)CCCCC(N)C(O)=O YEXNNJWRYPGWGU-UHFFFAOYSA-N 0.000 claims description 4
- QAOWNCQODCNURD-UHFFFAOYSA-N Sulfuric acid Chemical compound OS(O)(=O)=O QAOWNCQODCNURD-UHFFFAOYSA-N 0.000 claims description 4
- 229960002684 aminocaproic acid Drugs 0.000 claims description 4
- 238000006929 Pictet-Spengler synthesis reaction Methods 0.000 claims description 2
- TYQYRKDGHAPZRF-INIZCTEOSA-N benzyl (2s)-2-amino-3-(1h-indol-3-yl)propanoate Chemical compound O=C([C@H](CC=1C2=CC=CC=C2NC=1)N)OCC1=CC=CC=C1 TYQYRKDGHAPZRF-INIZCTEOSA-N 0.000 claims description 2
- 238000006555 catalytic reaction Methods 0.000 claims description 2
- 239000011259 mixed solution Substances 0.000 claims description 2
- 206010027476 Metastases Diseases 0.000 abstract description 15
- 230000009401 metastasis Effects 0.000 abstract description 13
- 230000000694 effects Effects 0.000 abstract description 10
- 229940079593 drug Drugs 0.000 abstract description 4
- 229940124599 anti-inflammatory drug Drugs 0.000 abstract description 2
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 32
- 241000699670 Mus sp. Species 0.000 description 22
- 206010028980 Neoplasm Diseases 0.000 description 20
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 18
- 239000007787 solid Substances 0.000 description 17
- IAZDPXIOMUYVGZ-WFGJKAKNSA-N Dimethyl sulfoxide Chemical compound [2H]C([2H])([2H])S(=O)C([2H])([2H])[2H] IAZDPXIOMUYVGZ-WFGJKAKNSA-N 0.000 description 16
- 229920006395 saturated elastomer Polymers 0.000 description 16
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 12
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 12
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 9
- 210000004027 cell Anatomy 0.000 description 8
- 238000006243 chemical reaction Methods 0.000 description 8
- 239000011780 sodium chloride Substances 0.000 description 8
- 241000699666 Mus <mouse, genus> Species 0.000 description 7
- 208000020816 lung neoplasm Diseases 0.000 description 7
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 6
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 6
- 239000000706 filtrate Substances 0.000 description 6
- 201000005202 lung cancer Diseases 0.000 description 6
- NNRFRJQMBSBXGO-CIUDSAMLSA-N (3s)-3-[[2-[[(2s)-2-amino-5-(diaminomethylideneamino)pentanoyl]amino]acetyl]amino]-4-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-oxobutanoic acid Chemical compound NC(N)=NCCC[C@H](N)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(O)=O NNRFRJQMBSBXGO-CIUDSAMLSA-N 0.000 description 5
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 5
- 101000829980 Homo sapiens Ral guanine nucleotide dissociation stimulator Proteins 0.000 description 5
- 102100023320 Ral guanine nucleotide dissociation stimulator Human genes 0.000 description 5
- 239000006285 cell suspension Substances 0.000 description 5
- 238000005481 NMR spectroscopy Methods 0.000 description 4
- 230000008034 disappearance Effects 0.000 description 4
- 150000002148 esters Chemical class 0.000 description 4
- 230000008961 swelling Effects 0.000 description 4
- IGVKWAAPMVVTFX-BUHFOSPRSA-N (e)-octadec-5-en-7,9-diynoic acid Chemical compound CCCCCCCCC#CC#C\C=C\CCCC(O)=O IGVKWAAPMVVTFX-BUHFOSPRSA-N 0.000 description 3
- BSYNRYMUTXBXSQ-UHFFFAOYSA-N Aspirin Chemical compound CC(=O)OC1=CC=CC=C1C(O)=O BSYNRYMUTXBXSQ-UHFFFAOYSA-N 0.000 description 3
- 238000011740 C57BL/6 mouse Methods 0.000 description 3
- 206010061218 Inflammation Diseases 0.000 description 3
- CTQNGGLPUBDAKN-UHFFFAOYSA-N O-Xylene Chemical compound CC1=CC=CC=C1C CTQNGGLPUBDAKN-UHFFFAOYSA-N 0.000 description 3
- 229960001138 acetylsalicylic acid Drugs 0.000 description 3
- 239000002246 antineoplastic agent Substances 0.000 description 3
- 229940041181 antineoplastic drug Drugs 0.000 description 3
- OKOWGETVYFKHMJ-NASUQTAISA-N benzyl (3S)-2-[6-amino-7-[(2-methylpropan-2-yl)oxy]-7-oxoheptanoyl]-1,3,4,9-tetrahydropyrido[3,4-b]indole-3-carboxylate Chemical compound CC(C)(C)OC(=O)C(CCCCC(=O)N1CC2=C(C[C@H]1C(=O)OCC3=CC=CC=C3)C4=CC=CC=C4N2)N OKOWGETVYFKHMJ-NASUQTAISA-N 0.000 description 3
- 210000005069 ears Anatomy 0.000 description 3
- 230000004054 inflammatory process Effects 0.000 description 3
- 238000011081 inoculation Methods 0.000 description 3
- 239000013641 positive control Substances 0.000 description 3
- 238000010898 silica gel chromatography Methods 0.000 description 3
- 239000008096 xylene Substances 0.000 description 3
- RUFDYIJGNPVTAY-UHFFFAOYSA-N 6-[(2-methylpropan-2-yl)oxycarbonylamino]hexanoic acid Chemical compound CC(C)(C)OC(=O)NCCCCCC(O)=O RUFDYIJGNPVTAY-UHFFFAOYSA-N 0.000 description 2
- 206010014025 Ear swelling Diseases 0.000 description 2
- PMZURENOXWZQFD-UHFFFAOYSA-L Sodium Sulfate Chemical compound [Na+].[Na+].[O-]S([O-])(=O)=O PMZURENOXWZQFD-UHFFFAOYSA-L 0.000 description 2
- 229940024606 amino acid Drugs 0.000 description 2
- 235000001014 amino acid Nutrition 0.000 description 2
- 238000000540 analysis of variance Methods 0.000 description 2
- 238000010171 animal model Methods 0.000 description 2
- 210000001099 axilla Anatomy 0.000 description 2
- DOKDMGOWZOTZRA-NTISSMGPSA-N benzyl (2s)-2-amino-3-(1h-indol-3-yl)propanoate;hydrochloride Chemical compound Cl.O=C([C@H](CC=1C2=CC=CC=C2NC=1)N)OCC1=CC=CC=C1 DOKDMGOWZOTZRA-NTISSMGPSA-N 0.000 description 2
- 201000011510 cancer Diseases 0.000 description 2
- 238000011156 evaluation Methods 0.000 description 2
- 230000003203 everyday effect Effects 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 238000003304 gavage Methods 0.000 description 2
- 230000005764 inhibitory process Effects 0.000 description 2
- 210000004072 lung Anatomy 0.000 description 2
- 208000037841 lung tumor Diseases 0.000 description 2
- 239000013642 negative control Substances 0.000 description 2
- 239000011541 reaction mixture Substances 0.000 description 2
- 239000011734 sodium Substances 0.000 description 2
- 238000003756 stirring Methods 0.000 description 2
- 238000012353 t test Methods 0.000 description 2
- 210000004881 tumor cell Anatomy 0.000 description 2
- FHOAKXBXYSJBGX-YFKPBYRVSA-N (2s)-3-hydroxy-2-[(2-methylpropan-2-yl)oxycarbonylamino]propanoic acid Chemical compound CC(C)(C)OC(=O)N[C@@H](CO)C(O)=O FHOAKXBXYSJBGX-YFKPBYRVSA-N 0.000 description 1
- QJCNLJWUIOIMMF-YUMQZZPRSA-N (2s,3s)-3-methyl-2-[(2-methylpropan-2-yl)oxycarbonylamino]pentanoic acid Chemical compound CC[C@H](C)[C@@H](C(O)=O)NC(=O)OC(C)(C)C QJCNLJWUIOIMMF-YUMQZZPRSA-N 0.000 description 1
- 238000005160 1H NMR spectroscopy Methods 0.000 description 1
- VNTPPTFQTSGONS-UHFFFAOYSA-N 6-amino-2-[(2-methylpropan-2-yl)oxycarbonyl]hexanoic acid Chemical compound CC(C)(C)OC(=O)C(C(O)=O)CCCCN VNTPPTFQTSGONS-UHFFFAOYSA-N 0.000 description 1
- VHUUQVKOLVNVRT-UHFFFAOYSA-N Ammonium hydroxide Chemical compound [NH4+].[OH-] VHUUQVKOLVNVRT-UHFFFAOYSA-N 0.000 description 1
- MYMOFIZGZYHOMD-UHFFFAOYSA-N Dioxygen Chemical compound O=O MYMOFIZGZYHOMD-UHFFFAOYSA-N 0.000 description 1
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 1
- 125000003338 L-glutaminyl group Chemical group O=C([*])[C@](N([H])[H])([H])C([H])([H])C([H])([H])C(=O)N([H])[H] 0.000 description 1
- 125000002061 L-isoleucyl group Chemical group [H]N([H])[C@]([H])(C(=O)[*])[C@](C([H])([H])[H])([H])C(C([H])([H])[H])([H])[H] 0.000 description 1
- 125000001176 L-lysyl group Chemical group [H]N([H])[C@]([H])(C(=O)[*])C([H])([H])C([H])([H])C([H])([H])C(N([H])[H])([H])[H] 0.000 description 1
- 125000000393 L-methionino group Chemical group [H]OC(=O)[C@@]([H])(N([H])[*])C([H])([H])C(SC([H])([H])[H])([H])[H] 0.000 description 1
- 125000000174 L-prolyl group Chemical group [H]N1C([H])([H])C([H])([H])C([H])([H])[C@@]1([H])C(*)=O 0.000 description 1
- 125000002842 L-seryl group Chemical group O=C([*])[C@](N([H])[H])([H])C([H])([H])O[H] 0.000 description 1
- 206010027458 Metastases to lung Diseases 0.000 description 1
- 240000001307 Myosotis scorpioides Species 0.000 description 1
- MDXGYYOJGPFFJL-QMMMGPOBSA-N N(alpha)-t-butoxycarbonyl-L-leucine Chemical compound CC(C)C[C@@H](C(O)=O)NC(=O)OC(C)(C)C MDXGYYOJGPFFJL-QMMMGPOBSA-N 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- GLNADSQYFUSGOU-GPTZEZBUSA-J Trypan blue Chemical compound [Na+].[Na+].[Na+].[Na+].C1=C(S([O-])(=O)=O)C=C2C=C(S([O-])(=O)=O)C(/N=N/C3=CC=C(C=C3C)C=3C=C(C(=CC=3)\N=N\C=3C(=CC4=CC(=CC(N)=C4C=3O)S([O-])(=O)=O)S([O-])(=O)=O)C)=C(O)C2=C1N GLNADSQYFUSGOU-GPTZEZBUSA-J 0.000 description 1
- 229960000583 acetic acid Drugs 0.000 description 1
- 230000001464 adherent effect Effects 0.000 description 1
- 235000011114 ammonium hydroxide Nutrition 0.000 description 1
- 230000002001 anti-metastasis Effects 0.000 description 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 description 1
- 239000008280 blood Substances 0.000 description 1
- 210000004369 blood Anatomy 0.000 description 1
- 230000037396 body weight Effects 0.000 description 1
- 150000007942 carboxylates Chemical class 0.000 description 1
- 239000012230 colorless oil Substances 0.000 description 1
- 238000004440 column chromatography Methods 0.000 description 1
- 230000002354 daily effect Effects 0.000 description 1
- 229910001882 dioxygen Inorganic materials 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- 230000007717 exclusion Effects 0.000 description 1
- 239000012065 filter cake Substances 0.000 description 1
- 210000003194 forelimb Anatomy 0.000 description 1
- 239000008098 formaldehyde solution Substances 0.000 description 1
- 239000012362 glacial acetic acid Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 229910052739 hydrogen Inorganic materials 0.000 description 1
- 230000005917 in vivo anti-tumor Effects 0.000 description 1
- 239000007788 liquid Substances 0.000 description 1
- 239000002609 medium Substances 0.000 description 1
- 230000001394 metastastic effect Effects 0.000 description 1
- 206010061289 metastatic neoplasm Diseases 0.000 description 1
- 239000000203 mixture Substances 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- WLJVXDMOQOGPHL-UHFFFAOYSA-N phenylacetic acid Chemical compound OC(=O)CC1=CC=CC=C1 WLJVXDMOQOGPHL-UHFFFAOYSA-N 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 238000004393 prognosis Methods 0.000 description 1
- 238000011160 research Methods 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 231100000331 toxic Toxicity 0.000 description 1
- 230000002588 toxic effect Effects 0.000 description 1
- 230000004614 tumor growth Effects 0.000 description 1
- 238000005303 weighing Methods 0.000 description 1
Images
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P29/00—Non-central analgesic, antipyretic or antiinflammatory agents, e.g. antirheumatic agents; Non-steroidal antiinflammatory drugs [NSAID]
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/55—Design of synthesis routes, e.g. reducing the use of auxiliary or protecting groups
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Rheumatology (AREA)
- Pain & Pain Management (AREA)
- Oncology (AREA)
- Nitrogen Condensed Heterocyclic Rings (AREA)
Abstract
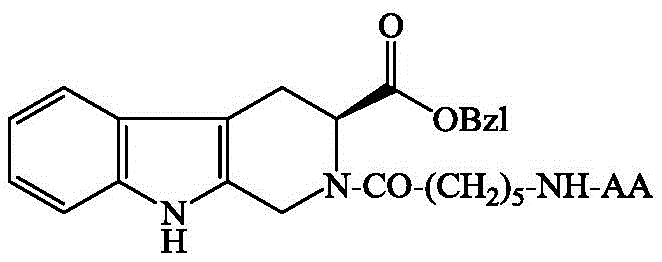
本发明公开了下面结构的(3S)‑N‑(6‑AA‑氨基正己酰)‑2,3,4,9‑四氢‑β‑咔啉‑3‑羧酸苄酯(式中AA选自L‑Glu(OBzl)的残基,L‑Ile的残基,L‑Lys的残基,L‑Leu的残基,L‑Met的残基,L‑Pro的残基,L‑Ser的残基)。公开了它们的制备方法和其抗肿瘤转移作用,以及公开了它们的抗炎作用,阐明了它们在制备抗肿瘤转移作用药物和抗炎药物中的应用。
The invention discloses (3S)-N-(6-AA-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylate benzyl ester with the following structure (where AA is selected as Residues from L-Glu(OBzl), Residues of L-Ile, Residues of L-Lys, Residues of L-Leu, Residues of L-Met, Residues of L-Pro, Residues of L-Ser Residues). Their preparation methods and their anti-tumor metastasis effects are disclosed, as well as their anti-inflammatory effects, and their applications in the preparation of anti-tumor metastasis drugs and anti-inflammatory drugs are clarified.
Description
技术领域technical field
本发明涉及(3S)-N-(6-AA-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯。涉及它的制备方法,涉及它的体内抗肿瘤转移作用,以及它们的抗炎作用。因而本发明涉及它在制备抗肿瘤转移药物和抗炎药物中的应用。本发明属于生物医药领域。The present invention relates to (3S)-N-(6-AA-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester. The invention relates to its preparation method, its in vivo anti-tumor metastasis effect, and their anti-inflammatory effect. Therefore, the present invention relates to its application in the preparation of anti-tumor metastases and anti-inflammatory drugs. The invention belongs to the field of biomedicine.
技术背景technical background
恶性肿瘤严重威胁人类健康。其中肺癌是最具侵略性的人类癌症之一。对于肺癌患者晚期的患者,通常10%-15%的人只能存活5年。在过去的30年里这种困难的局面尚未显著有所改善。在很多临床病例中,肺癌在被诊断之前已经转移到周围组织。肿瘤转移,尤其是肿瘤肺转移是肿瘤患者死亡的最大风险。至今,仍然没有可预防肿瘤转移的抗肿瘤药物用于临床。炎症则会进一步恶化肿瘤及肿瘤转移患者的预后。至今,更没有可预防炎症及肿瘤转移的抗肿瘤药物用于临床。发明具有抗肿瘤,抗肿瘤转移和抗炎三重作用的药物是抗肿瘤药物研究的前沿。发明人的前期发明(专利申请公布号CN 106349148A,申请号CN201510409682.6)曾经公开,在0.2μmol/kg剂量下氨基酸苄酯取代的双吲哚乙酸醇具有抗肿瘤,抗肿瘤转移和抗炎三重作用(左式)。发明人对这类双吲哚乙醇氨基酸酸苄酯发挥抗肿瘤转移作用和抗炎作用的最低有效剂量是0.2μmol/kg不满意。剂量偏高。在过去的两年里,发明人一直在寻找最低有效剂量比0.2μmol/kg低的抗肿瘤转移作用化合物。最终发明人发现,氨基酰-6-氨基己酸酸修饰的四氢咔啉羧酸苄酯在0.02μmol/kg剂量下具有抗肿瘤转移作用和抗炎作用。因为药物的毒副作用都可以随剂量降低而消失,所以有效剂量降低10倍表明了这种结构修饰有突出的技术效果。于是,发明人提出了本发明。Malignant tumors seriously threaten human health. Lung cancer is one of the most aggressive human cancers. For patients with advanced lung cancer, usually 10%-15% only survive 5 years. This difficult situation has not improved significantly over the past 30 years. In many clinical cases, lung cancer has metastasized to surrounding tissues before being diagnosed. Tumor metastases, especially lung metastases, are the greatest risk of death in cancer patients. So far, there are still no antitumor drugs that can prevent tumor metastasis in clinical use. Inflammation further worsens the prognosis of tumors and patients with tumor metastasis. So far, there are no anti-tumor drugs that can prevent inflammation and tumor metastasis in clinical use. The invention of drugs with triple effects of anti-tumor, anti-tumor metastasis and anti-inflammatory is the frontier of anti-tumor drug research. The inventor's previous invention (Patent Application Publication No. CN 106349148A, Application No. CN201510409682.6) once disclosed that the amino acid benzyl ester-substituted bisindole acetic alcohol has triple effects of anti-tumor, anti-tumor metastasis and anti-inflammatory at a dose of 0.2 μmol/kg. function (left). The inventors are not satisfied that the minimum effective dose of this type of bisindoleethanol amino acid benzyl ester to exert anti-tumor metastasis and anti-inflammatory effects is 0.2 μmol/kg. The dose is too high. For the past two years, the inventors have been looking for compounds with anti-metastatic effects at a minimum effective dose lower than 0.2 μmol/kg. Finally, the inventors found that aminoacyl-6-aminocaproic acid-modified benzyl tetrahydrocarboline carboxylate has anti-tumor metastasis and anti-inflammatory effects at a dose of 0.02 μmol/kg. Because the toxic and side effects of the drug can disappear with the dose reduction, the 10-fold reduction in the effective dose shows that this structural modification has outstanding technical effects. Thus, the inventors have come up with the present invention.
发明内容SUMMARY OF THE INVENTION
本发明的第一个内容是提供下面结构的(3S)-N-(6-AA-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(式中AA选自L-Glu(OBzl)的残基,L-Ile的残基,L-Lys的残基,L-Leu的残基,L-Met的残基,L-Pro的残基,L-Ser的残基)。The first aspect of the present invention is to provide (3S)-N-(6-AA-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester of the following structure (where AA is selected from the residues of L-Glu(OBzl), the residues of L-Ile, the residues of L-Lys, the residues of L-Leu, the residues of L-Met, the residues of L-Pro , residues of L-Ser).
本发明的第二个内容是提供(3S)-N-(6-AA-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯的制备方法,该方法包括:The second content of the present invention is to provide a preparation method of (3S)-N-(6-AA-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester , the method includes:
(1)在稀硫酸的催化下,L-色氨酸苄酯和甲醛进行Pictet-Spengler反应生成(3S)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯;(1) Under the catalysis of dilute sulfuric acid, L-tryptophan benzyl ester and formaldehyde undergo Pictet-Spengler reaction to generate benzyl (3S)-2,3,4,9-tetrahydro-β-carboline-3-carboxylate ester;
(2)在氢氧化钠水溶液(2M),水与二氧六环混合溶液中6-氨基己酸与二碳酸二叔丁酯(Boc)2O反应生成6-Boc-氨基己酸;(2) in aqueous sodium hydroxide solution (2M), in water and dioxane mixed solution, 6-aminocaproic acid and di-tert-butyl dicarbonate (Boc) 2 O react to generate 6-Boc-aminocaproic acid;
(3)在N,N-二环己基碳二亚胺和N-羟基苯并三氮唑存在下,在无水四氢呋喃中,6-Boc-氨基己酸与(3S)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯反应,得到(3S)-N-(6-Boc-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯;(3) In the presence of N,N-dicyclohexylcarbodiimide and N-hydroxybenzotriazole in anhydrous tetrahydrofuran, 6-Boc-aminocaproic acid was combined with (3S)-2,3,4 , 9-Tetrahydro-β-carboline-3-carboxylate benzyl ester was reacted to give (3S)-N-(6-Boc-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboyl Benzyl-3-carboxylate;
(4)冰浴下在氯化氢的乙酸乙酯溶液中(4M),(3S)-N-(6-Boc-氨基正己酰)-2,3,4,9-四氢咔啉-3-羧酸苄酯脱除Boc得(3S)-N-(6-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯;(4) In an ethyl acetate solution of hydrogen chloride (4M) under ice bath, (3S)-N-(6-Boc-amino-n-hexanoyl)-2,3,4,9-tetrahydrocarboline-3-carboxyl Acid benzyl ester removes Boc to obtain (3S)-N-(6-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester;
(5)在N,N-二环己基碳二亚胺和N-羟基苯并三氮唑存在下,在无水四氢呋喃中,(3S)-N-(6-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯与Boc-AA(式中AA选自L-Glu(OBzl)的残基,L-Ile的残基,L-Lys的残基,L-Leu的残基,L-Met的残基,L-Pro的残基,L-Ser的残基)反应,得到(3S)-N-(6-Boc-AA-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯;(5) (3S)-N-(6-amino-n-hexanoyl)-2,3 in anhydrous tetrahydrofuran in the presence of N,N-dicyclohexylcarbodiimide and N-hydroxybenzotriazole , 4,9-tetrahydro-β-carboline-3-carboxylate benzyl ester and Boc-AA (where AA is selected from the residues of L-Glu(OBzl), the residues of L-Ile, the residues of L-Lys residues, residues of L-Leu, residues of L-Met, residues of L-Pro, residues of L-Ser) react to give (3S)-N-(6-Boc-AA-amino-n-hexanoyl) )-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester;
(6)冰浴下在氯化氢的乙酸乙酯溶液中(4M),(3S)-N-(6-Boc-AA-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯脱除Boc得(3S)-N-(6-AA-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯。(6) In ethyl acetate solution of hydrogen chloride (4M) under ice bath, (3S)-N-(6-Boc-AA-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboyl Removal of Boc from benzyl phospholine-3-carboxylate to give (3S)-N-(6-AA-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylate benzyl ester .
本发明的第三个内容是评价(3S)-N-(6-AA-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯抑制C57BL/6小鼠肺癌转移活性。The third aspect of the present invention is to evaluate the inhibition of C57BL/ 6 Mice lung cancer metastasis activity.
本发明的第四个内容是评价(3S)-N-(6-AA-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯对ICR小鼠炎症的抑制作用。The fourth aspect of the present invention is to evaluate (3S)-N-(6-AA-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylate benzyl ester with low ICR Inhibition of inflammation in mice.
附图说明Description of drawings
图1.(3S)-N-(6-AA-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯的合成路线.ⅰ)HCHO,H2O,H2SO4;ⅱ)N,N-二环己基碳二亚胺(DCC),N-羟基苯并三氮唑(HOBt),N-甲基吗啉,四氢呋喃;ⅲ)二氧六环,氢氧化钠水溶液,二碳酸二叔丁酯(Boc)2O;ⅳ)氯化氢的乙酸乙酯溶液(4M)。5a,6a中AA=L-谷氨酸残基;5b,6b中AA=L-异亮氨酸残基;5c,6c中AA=L-赖氨酸残基;5d,6d中AA=L-亮氨酸残基;5e,6e中AA=L-甲硫氨酸残基;5f,6f中AA=L-脯氨酸残基;5g,6g中AA=L-丝氨酸残基。Figure 1. Synthetic route of (3S)-N-(6-AA-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester. i)HCHO,H 2 O,H 2 SO 4 ; ii) N,N-dicyclohexylcarbodiimide (DCC), N-hydroxybenzotriazole (HOBt), N-methylmorpholine, tetrahydrofuran; iii) dioxygen Hexacyclic, aqueous sodium hydroxide, di-tert-butyl dicarbonate (Boc) 2O ; iv) hydrogen chloride in ethyl acetate (4M). AA=L-glutamic acid residue in 5a,6a; AA=L-isoleucine residue in 5b,6b; AA=L-lysine residue in 5c,6c; AA=L in 5d,6d - Leucine residue; AA = L-methionine residue in 5e, 6e; AA = L-proline residue in 5f, 6f; AA = L-serine residue in 5g, 6g.
具体实施方式Detailed ways
为了进一步阐述本发明,下面给出一系列实施例。这些实施例完全是例证性的,它们仅用来对本发明进行具体描述,不应当理解为对本发明的限制。In order to further illustrate the present invention, a series of examples are given below. These examples are purely illustrative, they are only used to specifically describe the present invention, and should not be construed as limiting the present invention.
实施例1制备(3S)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(1)Example 1 Preparation of (3S)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (1)
冰浴下向300mL水缓慢加入1.5mL浓H2SO4,搅拌3min。之后加入5.00g(15.1mmol)L-色氨酸苄酯盐酸盐和6mL甲醛溶液(40%)。室温搅拌72h,TLC显示L-色氨酸苄酯盐酸盐消失。反应液在冰浴下用浓氨水调溶液pH为7,过滤。滤饼用100mL乙酸乙酯溶解,得到的溶液用饱和氯化钠水溶液洗(40mL×3),之后用无水硫酸钠干燥。过滤,滤液减压浓缩。残留物经硅胶柱层析纯化(二氯甲烷:甲醇=90:1)得1.9g(41%)目标化合物,为黄色油状固体。ESI-MS(m/e):307[M+H]+。1.5 mL of concentrated H 2 SO 4 was slowly added to 300 mL of water under ice bath, and stirred for 3 min. Then 5.00 g (15.1 mmol) L-tryptophan benzyl ester hydrochloride and 6 mL formaldehyde solution (40%) were added. After stirring at room temperature for 72h, TLC showed the disappearance of L-tryptophan benzyl ester hydrochloride. The reaction solution was adjusted to pH 7 with concentrated ammonia water under ice bath, and filtered. The filter cake was dissolved with 100 mL of ethyl acetate, and the obtained solution was washed with saturated aqueous sodium chloride solution (40 mL×3), and then dried over anhydrous sodium sulfate. After filtration, the filtrate was concentrated under reduced pressure. The residue was purified by silica gel column chromatography (dichloromethane:methanol=90:1) to obtain 1.9 g (41%) of the title compound as a yellow oily solid. ESI-MS (m/e): 307 [M+H] + .
实施例2制备6-叔丁氧羰氨基己酸(2)Example 2 Preparation of 6-tert-butoxycarbonylaminocaproic acid (2)
将5.00g(38.2mmol)6-氨基己酸(EACA)用50mL蒸馏水悬浮。得到的悬浮液于冰浴下加入10mL氢氧化钠水溶液(2M),搅拌至溶解。往得到的溶液中加9.2g(42.2mmol)(Boc)2O与20mL二氧六环的溶液并用氢氧化钠水溶液(2M)调pH至9,搅拌30min。撤去冰浴,室温搅拌至TLC显示6-氨基己酸消失。冰浴下反应液用饱和KHSO4水溶液调节pH至7,减压浓缩除去二氧六环。冰浴下反应液用饱和KHSO4水溶液调节pH至2,用乙酸乙酯萃取(50mL×3)。乙酸乙酯层用饱和NaCl水溶液洗(40mL×3),用无水硫酸钠干燥。过滤,滤液减压浓缩得8.0g(91%)目标化合物,为无色油状物。ESI-MS(m/e):232[M+H]+。5.00 g (38.2 mmol) of 6-aminocaproic acid (EACA) were suspended in 50 mL of distilled water. The resulting suspension was added with 10 mL of aqueous sodium hydroxide solution (2M) under an ice bath and stirred until dissolved. To the resulting solution, a solution of 9.2 g (42.2 mmol) (Boc) 2 O and 20 mL of dioxane was added and the pH was adjusted to 9 with aqueous sodium hydroxide solution (2 M) and stirred for 30 min. The ice bath was removed and stirred at room temperature until TLC showed the disappearance of 6-aminocaproic acid. Under ice bath, the pH of the reaction solution was adjusted to 7 with saturated aqueous KHSO 4 solution, and the dioxane was removed by concentration under reduced pressure. The reaction solution was adjusted to
实施例3制备(3S)-N-(6-Boc-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(3)Example 3 Preparation of (3S)-N-(6-Boc-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (3)
将4.15g(18mmol)Boc-6-氨基己酸用80mL无水四氢呋喃溶解,冰浴下加入2.46g(18.2mmol)N-羟基苯骈三氮唑(HOBt)和4.04g(19.6mmol)N,N-二环己基碳二亚胺(DCC),搅拌60分钟。之后,往得到反应液加5.06g(16.5mmol)(3S)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯与80mL无水四氢呋喃的溶液。反应混合物于冰浴下用N-甲基吗啉(NMM)调pH至9,室温搅拌24小时。TLC显示(3S)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯消失。反应液过滤,滤液减压浓缩,残留物加乙酸乙酯溶解。溶液依次用饱和NaHCO3水溶液洗(40mL×3),饱和NaCl水溶液洗(40mL×3),饱和KHSO4水溶液洗(40mL×3),饱和NaCl水溶液洗(40mL×3),饱和NaHCO3水溶液洗(40mL×3),饱和NaCl水溶液洗(40mL×3)。乙酸乙酯层用无水Na2SO4干燥,过滤,滤液减压浓缩,残留物经硅胶柱层析纯化(二氯甲烷:甲醇=160:1)得5.43g(64%)目标化合物,为黄色油状固体。ESI-MS(m/e):520[M+H]+。4.15g (18mmol) Boc-6-aminocaproic acid was dissolved in 80mL anhydrous tetrahydrofuran, 2.46g (18.2mmol) N-hydroxybenzotriazole (HOBt) and 4.04g (19.6mmol) N were added under ice bath, N-dicyclohexylcarbodiimide (DCC), stirred for 60 minutes. Then, a solution of 5.06 g (16.5 mmol) of (3S)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester and 80 mL of anhydrous tetrahydrofuran was added to the obtained reaction solution. The reaction mixture was adjusted to pH 9 with N-methylmorpholine (NMM) in an ice bath and stirred at room temperature for 24 hours. TLC showed disappearance of (3S)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester. The reaction solution was filtered, the filtrate was concentrated under reduced pressure, and the residue was dissolved in ethyl acetate. The solution was washed successively with saturated aqueous NaHCO 3 solution (40 mL×3), saturated aqueous NaCl solution (40 mL×3), saturated aqueous KHSO 4 solution (40 mL×3), saturated aqueous NaCl solution (40 mL×3), and saturated aqueous NaHCO 3 solution (40 mL×3), washed with saturated aqueous NaCl solution (40 mL×3). The ethyl acetate layer was dried over anhydrous Na 2 SO 4 , filtered, the filtrate was concentrated under reduced pressure, and the residue was purified by silica gel column chromatography (dichloromethane:methanol=160:1) to obtain 5.43 g (64%) of the title compound, which was Yellow oily solid. ESI-MS (m/e): 520 [M+H] + .
实施例4制备(3S)-N-(6-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(4)Example 4 Preparation of (3S)-N-(6-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (4)
冰浴下将3.4g(6.6mmol)(3S)-N-(6-Boc-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯用40mL氯化氢的乙酸乙酯溶液(4M)溶解,搅拌3小时,TLC显示(3S)-N-(6-Boc-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯消失。搅拌下将反应混合物减压浓缩至干。残留物加无水乙酸乙酯溶解,溶液再减压浓缩至干。该操作重复三次。残留物加无水乙醚充分洗,得2.8g(94%)目标化合物,为棕褐色固体。ESI-MS(m/e):420[M+H]+;Mp 223-226℃。1HNMR(300MHz,DMSO-d6:δ/ppm=10.963(d,J=11.4Hz,1H),7.894(s,3H),7.458(d,J=7.5Hz,1H),7.327(t,J=7.5Hz,1H),7.204(dd,J1=1.5Hz,J2=7.2Hz,1H),7.158-6.972(m,6H),5.607(d,J=4.7Hz,1H),5.059(s,2H),4767(d,J=15.6Hz,1H),4.610(d,J=17.4Hz,1H),3.426(m,1H),3.021(m,1H),2.754(m,2H),2.613(m,1H),2.389(m,1H),1.568-1.546(m,4H),1.399-1.323(m,2H)。Under ice bath, 3.4g (6.6mmol) of (3S)-N-(6-Boc-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester was added to 40mL Hydrogen chloride in ethyl acetate (4M) was dissolved, stirred for 3 hours, TLC showed (3S)-N-(6-Boc-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3 - Disappearance of benzyl carboxylate. The reaction mixture was concentrated to dryness under reduced pressure with stirring. The residue was dissolved in anhydrous ethyl acetate, and the solution was concentrated to dryness under reduced pressure. This operation is repeated three times. The residue was thoroughly washed with anhydrous ether to obtain 2.8 g (94%) of the title compound as a tan solid. ESI-MS (m/e): 420 [M+H] + ; Mp 223-226°C. 1 HNMR (300MHz, DMSO-d6:δ/ppm=10.963(d,J=11.4Hz,1H), 7.894(s,3H), 7.458(d,J=7.5Hz,1H), 7.327(t,J= 7.5Hz, 1H), 7.204(dd, J 1 =1.5Hz, J 2 =7.2Hz, 1H), 7.158-6.972(m, 6H), 5.607(d, J=4.7Hz, 1H), 5.059(s, 2H), 4767(d, J=15.6Hz, 1H), 4.610(d, J=17.4Hz, 1H), 3.426(m, 1H), 3.021(m, 1H), 2.754(m, 2H), 2.613( m, 1H), 2.389 (m, 1H), 1.568-1.546 (m, 4H), 1.399-1.323 (m, 2H).
实施例5制备(3S)-N-(6-Boc-Glu(OBzl)-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(5a)Example 5 Preparation of (3S)-N-(6-Boc-Glu(OBzl)-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (5a)
将2.44g(7.2mmol)Boc-Glu(OBzl)和0.97g(7.2mmol)N-羟基苯骈三氮唑(HOBt)用30mL无水四氢呋喃溶解,冰浴下加入1.48g(7.2mmol)N,N-二环己基碳二亚胺(DCC),搅拌40分钟。之后,往得到反应液中加3.00g(6.6mmol)(3S)-N-(6-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯与30mL无水四氢呋喃的溶液,冰浴下用N-甲基吗啉调pH至8,室温搅拌12小时。TLC显示(3S)-N-(6-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯消失,反应液过滤,滤液减压浓缩,残留物加100mL乙酸乙酯溶解。溶液依次用饱和NaHCO3水溶液洗(40mL×3),饱和NaCl水溶液洗(40mL×3),饱和KHSO4水溶液洗(40mL×3),饱和NaCl水溶液洗(40mL×3),饱和NaHCO3水溶液洗(40mL×3),饱和NaCl水溶液洗(40mL×3)。乙酸乙酯层用无水Na2SO4干燥,过滤,滤液减压浓缩,残留物经硅胶柱层析纯化(二氯甲烷:甲醇=60:1)得1.52g(31%)目标化合物,为淡黄色固体。ESI-MS(m/z):739[M+H]+。Dissolve 2.44g (7.2mmol) Boc-Glu (OBzl) and 0.97g (7.2mmol) N-hydroxybenzotriazole (HOBt) with 30mL anhydrous tetrahydrofuran, add 1.48g (7.2mmol) N under ice bath, N-dicyclohexylcarbodiimide (DCC), stirred for 40 minutes. Then, 3.00 g (6.6 mmol) of (3S)-N-(6-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester was added to the obtained reaction solution It was mixed with 30 mL of anhydrous tetrahydrofuran solution, adjusted to pH 8 with N-methylmorpholine under ice bath, and stirred at room temperature for 12 hours. TLC showed that (3S)-N-(6-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester disappeared, the reaction solution was filtered, the filtrate was concentrated under reduced pressure, and the residue The mixture was dissolved in 100 mL of ethyl acetate. The solution was washed successively with saturated aqueous NaHCO 3 solution (40 mL×3), saturated aqueous NaCl solution (40 mL×3), saturated aqueous KHSO 4 solution (40 mL×3), saturated aqueous NaCl solution (40 mL×3), and saturated aqueous NaHCO 3 solution (40 mL×3), washed with saturated aqueous NaCl solution (40 mL×3). The ethyl acetate layer was dried over anhydrous Na 2 SO 4 , filtered, the filtrate was concentrated under reduced pressure, and the residue was purified by silica gel column chromatography (dichloromethane:methanol=60:1) to obtain 1.52g (31%) of the title compound, which was Pale yellow solid. ESI-MS (m/z): 739 [M+H] + .
实施例6制备(3S)-N-(6-Boc-Ile-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(5b)Example 6 Preparation of (3S)-N-(6-Boc-Ile-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (5b)
按照实施例5的方法,从2.60g(5.7mmol)(3S)-N-(6-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯和1.45g(6.3mmol)Boc-Ile得到1.19g(33%)标题化合物,为淡黄色固体。ESI-MS(m/z):633[M+H]+。According to the method of Example 5, from 2.60 g (5.7 mmol) (3S)-N-(6-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester and 1.45 g (6.3 mmol) of Boc-Ile to give 1.19 g (33%) of the title compound as a pale yellow solid. ESI-MS (m/z): 633 [M+H] + .
实施例7制备(3S)-N-(6-Boc-Lys(Boc)-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(5c)Example 7 Preparation of (3S)-N-(6-Boc-Lys(Boc)-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (5c)
按照实施例5的方法从0.86g(1.87mmol)Boc-Lys(Boc)和0.42g(1.37mmol)(3S)-N-(6-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯得到0.54g(39%)标题化合物,为淡黄色固体。ESI-MS(m/z):748[M+H]+。From 0.86 g (1.87 mmol) Boc-Lys(Boc) and 0.42 g (1.37 mmol) (3S)-N-(6-amino-n-hexanoyl)-2,3,4,9-tetrahydro according to the method of Example 5 -β-Carboline-3-carboxylate benzyl ester gave 0.54 g (39%) of the title compound as a pale yellow solid. ESI-MS (m/z): 748 [M+H] + .
实施例8制备(3S)-N-(6-Boc-Leu-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(5d)Example 8 Preparation of (3S)-N-(6-Boc-Leu-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (5d)
按照实施例5的方法,从2.10g(4.6mmol)(3S)-N-(6-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯和1.28g(5.5mmol)Boc-Leu得到1.62g(56%)标题化合物,为淡黄色固体。ESI-MS(m/z):633[M+H]+。According to the method of Example 5, from 2.10 g (4.6 mmol) of (3S)-N-(6-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester and 1.28 g (5.5 mmol) of Boc-Leu to give 1.62 g (56%) of the title compound as a pale yellow solid. ESI-MS (m/z): 633 [M+H] + .
实施例9制备(3S)-N-(6-Boc-Met-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(5e)Example 9 Preparation of (3S)-N-(6-Boc-Met-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (5e)
按照实施例5的方法,从1.80g(4.0mmol)(3S)-N-(6-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯和1.28g(5.1mmol)Boc-Met得到0.35g(14%)标题化合物,为淡黄色固体。ESI-MS(m/z):651[M+H]+。According to the method of Example 5, from 1.80 g (4.0 mmol) of (3S)-N-(6-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester and 1.28 g (5.1 mmol) of Boc-Met to give 0.35 g (14%) of the title compound as a pale yellow solid. ESI-MS (m/z): 651 [M+H] + .
实施例10制备(3S)-N-(6-Boc-Pro-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(5f)Example 10 Preparation of (3S)-N-(6-Boc-Pro-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (5f)
按照实施例5的方法,从2.00g(4.4mmol)(3S)-N-(6-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯和1.04g(4.8mmol)Boc-Pro得到1.20g(44%)标题化合物,为淡黄色固体。ESI-MS(m/z):617[M+H]+。According to the method of Example 5, from 2.00 g (4.4 mmol) (3S)-N-(6-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester and 1.04 g (4.8 mmol) of Boc-Pro to give 1.20 g (44%) of the title compound as a pale yellow solid. ESI-MS (m/z): 617 [M+H] + .
实施例11制备(3S)-N-(6-Boc-Ser-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(5g)Example 11 Preparation of (3S)-N-(6-Boc-Ser-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (5g)
按照实施例5的方法,从1.5g(3.3mmol)(3S)-N-(6-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯和0.74g(3.6mmol)Boc-Ser得到0.30g(15%)标题化合物,为淡黄色固体。ESI-MS(m/z):607[M+H]+。According to the method of Example 5, from 1.5 g (3.3 mmol) of (3S)-N-(6-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester and 0.74 g (3.6 mmol) of Boc-Ser to give 0.30 g (15%) of the title compound as a pale yellow solid. ESI-MS (m/z): 607 [M+H] + .
实施例12制备(3S)-N-(6-Glu(OBzl)-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(6a)Example 12 Preparation of (3S)-N-(6-Glu(OBzl)-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (6a)
将1000mg(3S)-N-(6-Boc-Glu(OBzl)-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯于冰浴下加入15mL氯化氢的乙酸乙酯溶液(4M)并搅拌2.5h,TLC(乙酸乙酯:水:冰醋酸=6:1:1.2)显示(3S)-N-(6-Boc-Glu(OBzl)-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯消失。反应液减压浓缩至干。残留物加无水乙酸乙酯溶解,再减压浓缩至干。该操作重复三次。残留物加无水乙醚充分洗,之后经C18柱层析纯化(甲醇:水=40:60)并冻干得到490mg(54%)标题化合物,为淡黄色固体。ESI-MS(m/e):639[M+H]+.Mp:103-105℃;1H NMR(300MHz,DMSO-d6:δ/ppm=10.963(d,J=10.5Hz,1H),8.608(m,1H),8.328(s,3H),7.455(d,J=7.5Hz,1H),7.395-7.305(m,6H),7.236-7.101(m,7H),5.685(d,J=4.5Hz,1H),5.096(s,2H),5.053(s,2H),4761(d,J=15.6Hz,1H),4.629(d,J=17.1Hz,1H),3.803(dd,J1=5.7Hz,J2=12.0Hz 1H),3.419(m,1H),3.145-2.965(m,3H),2.574(m,1H),2.457-2.389(m,3H),2.039-1.985(m,2H),1.582-1.508(m,2H),1.482-1.421(m,2H),1.374-1.300(m,2H)。1000 mg of (3S)-N-(6-Boc-Glu(OBzl)-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester was added under ice bath 15mL of hydrogen chloride in ethyl acetate (4M) and stirred for 2.5h, TLC (ethyl acetate:water:glacial acetic acid=6:1:1.2) showed (3S)-N-(6-Boc-Glu(OBzl)-amino n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester disappeared. The reaction solution was concentrated to dryness under reduced pressure. The residue was dissolved in anhydrous ethyl acetate, and then concentrated to dryness under reduced pressure. This operation is repeated three times. The residue was thoroughly washed with anhydrous ether, then purified by C18 column chromatography (methanol:water=40:60) and lyophilized to obtain 490 mg (54%) of the title compound as a pale yellow solid. ESI-MS(m/e):639[M+H]+.Mp:103-105℃; 1H NMR(300MHz,DMSO-d6:δ/ppm=10.963(d,J=10.5Hz,1H), 8.608(m, 1H), 8.328(s, 3H), 7.455(d, J=7.5Hz, 1H), 7.395-7.305(m, 6H), 7.236-7.101(m, 7H), 5.685(d, J= 4.5Hz, 1H), 5.096(s, 2H), 5.053(s, 2H), 4761(d, J=15.6Hz, 1H), 4.629(d, J=17.1Hz, 1H), 3.803(dd, J 1 =5.7Hz, J 2 =12.0Hz 1H), 3.419(m, 1H), 3.145-2.965(m, 3H), 2.574(m, 1H), 2.457-2.389(m, 3H), 2.039-1.985(m, 2H), 1.582-1.508 (m, 2H), 1.482-1.421 (m, 2H), 1.374-1.300 (m, 2H).
实施例13制备(3S)-N-(6-Ile-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(6b)Example 13 Preparation of (3S)-N-(6-Ile-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (6b)
按照实施例12的方法,从840mg(3S)-N-(6-Boc-Ile-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯得到560mg(79%)标题化合物,为淡黄色固体。ESI-MS(m/e):533[M+H]+.Mp121-122℃;1H NMR(300MHz,DMSO-d6:δ/ppm=10.997(d,J=13.2Hz,1H),8.541(m,1H),8.240(s,3H),7.454(d,J=7.5Hz,1H),7.329(t,J=7.8Hz,1H),7.205(dd,J1=2.1Hz,J2=5.7Hz,1H),7.162-7.104(m,6H),5.602(d,J=4.8Hz,1H),5.058(s,2H),4769(d,J=15.6Hz,1H),4.637(d,J=17.4Hz,1H),3.586(m,1H),3.422(m,1H),3.196(m,1H),3.028(m,1H),2.566(m,1H),2.417(m,1H),1.815(m,1H),1.592-1.310(m,6H),1.113(m,2H),0.911-0.840(m,6H)。Following the method of Example 12, obtained from 840 mg of (3S)-N-(6-Boc-Ile-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester 560 mg (79%) of the title compound as a pale yellow solid. ESI-MS (m/e): 533[M+H] + .Mp121-122°C; 1 H NMR (300MHz, DMSO-d6: δ/ppm=10.997 (d, J=13.2Hz, 1H), 8.541 ( m, 1H), 8.240 (s, 3H), 7.454 (d, J=7.5Hz, 1H), 7.329 (t, J=7.8Hz, 1H), 7.205 (dd, J 1 =2.1Hz, J 2 =5.7 Hz,1H),7.162-7.104(m,6H),5.602(d,J=4.8Hz,1H),5.058(s,2H),4769(d,J=15.6Hz,1H),4.637(d,J =17.4Hz, 1H), 3.586(m, 1H), 3.422(m, 1H), 3.196(m, 1H), 3.028(m, 1H), 2.566(m, 1H), 2.417(m, 1H), 1.815 (m, 1H), 1.592-1.310 (m, 6H), 1.113 (m, 2H), 0.911-0.840 (m, 6H).
实施例14制备(3S)-N-(6-Lys-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(6c)Example 14 Preparation of (3S)-N-(6-Lys-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (6c)
按照实施例12的方法,从800mg(3S)-N-(6-Boc-Lys(Boc)-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯得到430mg(75%)标题化合物,为淡黄色固体。ESI-MS(m/e):548.3[M+H]+.Mp:118.3-120.1℃;1HNMR(300MHz,DMSO-d6:δ/ppm=10.990(d,J=15.3Hz,1H),8.642(m,1H),8.292(s,3H),8.055(s,3H),7.454(d,J=7.8Hz,1H),7.331(dd,J1=7.8Hz,J2=15.6Hz,1H),7.205(dd,J1=1.8Hz,J2=7.5Hz,1H),7.162-7.086(m,6H),5.596(d,J=4.8Hz,1H),5.061(s,2H),4775(d,J=15.6Hz,1H),4.634(d,J=17.4Hz,1H),3.744(m,1H),3.451(m,1H),3.109-2.937(m,3H),2.746(m,2H),2.584(m,1H),2.430(m,1H),1.742(m,2H),1.621-1.313(m,10H)。According to the method of Example 12, from 800 mg of (3S)-N-(6-Boc-Lys(Boc)-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid The benzyl ester gave 430 mg (75%) of the title compound as a pale yellow solid. ESI-MS (m/e): 548.3[M+H] + .Mp: 118.3-120.1°C; 1 HNMR (300MHz, DMSO-d6: δ/ppm=10.990 (d, J=15.3Hz, 1H), 8.642 (m,1H),8.292(s,3H),8.055(s,3H),7.454(d,J=7.8Hz,1H),7.331(dd,J1 = 7.8Hz,J2= 15.6Hz ,1H) ,7.205(dd,J1 = 1.8Hz,J2=7.5Hz,1H), 7.162-7.086 (m,6H),5.596(d,J=4.8Hz,1H),5.061(s,2H),4775( d, J=15.6Hz, 1H), 4.634(d, J=17.4Hz, 1H), 3.744(m, 1H), 3.451(m, 1H), 3.109-2.937(m, 3H), 2.746(m, 2H) ), 2.584(m, 1H), 2.430(m, 1H), 1.742(m, 2H), 1.621-1.313(m, 10H).
实施例15制备(3S)-N-(6-Leu-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(6d)Example 15 Preparation of (3S)-N-(6-Leu-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (6d)
按照实施例12的方法,从1000mg(3S)-N-(6-Boc-Leu-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯得到605mg(72%)标题化合物,为淡黄色固体。ESI-MS(m/e):533[M+H]+.Mp111-113℃;1H NMR(300MHz,DMSO-d6:δ/ppm=10.998(d,J=13.2Hz,1H),8.635(dd,J1=5.1Hz,J2=10.5Hz 1H),8.281(s,3H),7.455(d,J=7.5Hz,1H),7.328(t,J=7.8Hz,1H),7.200(dd,J1=2.1Hz,J2=7.5Hz,1H),7.158-7.072(m,6H),5.613(d,J=4.5Hz,1H),5.057(s,2H),4769(d,J=15.6Hz,1H),4.630(d,J=17.4Hz,1H),3.707(m,1H),3.447(m,1H),3.174-3.030(m,3H),2.569(m,1H),2.419(m,1H),1.645-1.306(m,9H),0.908(d,J=6.0Hz,3H),0.888(d,J=6.0Hz,3H)。According to the method of Example 12, from 1000 mg of (3S)-N-(6-Boc-Leu-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester 605 mg (72%) of the title compound as a pale yellow solid. ESI-MS (m/e): 533[M+H] + .Mp111-113°C; 1 H NMR (300MHz, DMSO-d6: δ/ppm=10.998 (d, J=13.2Hz, 1H), 8.635 ( dd, J 1 =5.1Hz, J 2 =10.5Hz 1H), 8.281(s, 3H), 7.455(d, J=7.5Hz, 1H), 7.328(t, J=7.8Hz, 1H), 7.200(dd , J 1 =2.1Hz, J 2 =7.5Hz,1H),7.158-7.072(m,6H),5.613(d,J=4.5Hz,1H),5.057(s,2H),4769(d,J= 15.6Hz, 1H), 4.630(d, J=17.4Hz, 1H), 3.707(m, 1H), 3.447(m, 1H), 3.174-3.030(m, 3H), 2.569(m, 1H), 2.419( m, 1H), 1.645-1.306 (m, 9H), 0.908 (d, J=6.0 Hz, 3H), 0.888 (d, J=6.0 Hz, 3H).
实施例16制备(3S)-N-(6-Met-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(6e)Example 16 Preparation of (3S)-N-(6-Met-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (6e)
按照实施例12的方法,从1100mg(3S)-N-(6-Boc-Met-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯得到530mg(57%)标题化合物,为淡黄色固体。ESI-MS(m/e):551.2[M+H]+.Mp:108.8-110.4℃;1HNMR(300MHz,DMSO-d6:δ/ppm=10.962(d,J=10.8Hz,1H),8.599(q,J1=5.4Hz,J2=10.5Hz,1H),8.319(s,3H),7.456(d,J=7.5Hz,1H),7.329(t,J=7.5Hz,1H),7.206(dd,J1=2.1Hz,J2=6.6Hz,1H),7.163-7.074(m,6H),5.604(d,J=4.8Hz,1H),5.059(s,2H),4769(d,J=15.6Hz,1H),4.631(d,J=17.4Hz,1H),3.818(dd,J1=5.4Hz,J2=11.7Hz,1H),3.427(m,1H),3.186-3.050(m,3H),2.568(m,1H),2.466-2.401(m,3H),2.059-1.962(m,5H),1.596-1.520(m,2H),1.495-1.433(m,2H),1.384-1.308(m,2H)。Following the procedure of Example 12, from 1100 mg of (3S)-N-(6-Boc-Met-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester 530 mg (57%) of the title compound as a pale yellow solid. ESI-MS (m/e): 551.2[M+H] + .Mp: 108.8-110.4°C; 1 HNMR (300MHz, DMSO-d6: δ/ppm=10.962 (d, J=10.8Hz, 1H), 8.599 (q, J1 = 5.4Hz, J2=10.5Hz, 1H), 8.319 (s, 3H), 7.456(d, J=7.5Hz, 1H), 7.329(t, J=7.5Hz, 1H), 7.206 (dd, J 1 =2.1Hz, J 2 =6.6Hz,1H),7.163-7.074(m,6H),5.604(d,J=4.8Hz,1H),5.059(s,2H),4769(d, J=15.6Hz, 1H), 4.631 (d, J=17.4Hz, 1H), 3.818 (dd, J1 = 5.4Hz, J2=11.7Hz, 1H), 3.427 (m, 1H), 3.186-3.050( m,3H),2.568(m,1H),2.466-2.401(m,3H),2.059-1.962(m,5H),1.596-1.520(m,2H),1.495-1.433(m,2H),1.384- 1.308 (m, 2H).
实施例17制备(3S)-N-(6-Pro-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(6f)Example 17 Preparation of (3S)-N-(6-Pro-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (6f)
按照实施例12的方法,从950mg(3S)-N-(6-Boc-Pro-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯得到598mg(75%)标题化合物,为淡黄色固体。ESI-MS(m/e):517.2[M+H]+.Mp 116-117℃;1H NMR(300MHz,DMSO-d6:δ/ppm=10.975(d,J=11.1Hz,1H),9.188(m,2H),8.610(dd,J1=5.4Hz,J2=11.1Hz,1H),7.456(d,J=7.8Hz,1H),7.328(t,J=7.5Hz,1H),7.202(dd,J1=2.1Hz,J2=7.5Hz,1H),7.159-7.085(m,6H),5.605(d,J=4.5Hz,1H),5.058(s,2H),4767(d,J=15.6Hz,1H),4.636(d,J=17.7Hz,1H),4.21(m,1H),3.421(m,1H),3.224-3.099(m,5H),2.569(m,1H),2.438(m,1H),2.273(m,1H),1.889-1.786(m,3H),1.552(m,2H),1.457(m,2H),1.322(m,2H)。According to the method of Example 12, from 950 mg of (3S)-N-(6-Boc-Pro-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester 598 mg (75%) of the title compound as a pale yellow solid. ESI-MS (m/e): 517.2 [M+H] + .Mp 116-117°C; 1 H NMR (300 MHz, DMSO-d6: δ/ppm=10.975 (d, J=11.1 Hz, 1H), 9.188 (m, 2H), 8.610(dd, J 1 =5.4Hz, J 2 =11.1Hz, 1H), 7.456(d, J=7.8Hz, 1H), 7.328(t, J=7.5Hz, 1H), 7.202 (dd, J 1 =2.1Hz, J 2 =7.5Hz,1H),7.159-7.085(m,6H),5.605(d,J=4.5Hz,1H),5.058(s,2H),4767(d, J=15.6Hz, 1H), 4.636(d, J=17.7Hz, 1H), 4.21(m, 1H), 3.421(m, 1H), 3.224-3.099(m, 5H), 2.569(m, 1H), 2.438 (m, 1H), 2.273 (m, 1H), 1.889-1.786 (m, 3H), 1.552 (m, 2H), 1.457 (m, 2H), 1.322 (m, 2H).
实施例18制备(3S)-N-(6-Ser-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯(6g)Example 18 Preparation of (3S)-N-(6-Ser-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester (6g)
按照实施例12的方法,从825mg(3S)-N-(6-Boc-Ser-氨基正己酰)-2,3,4,9-四氢-β-咔啉-3-羧酸苄酯得到448mg(65%)标题化合物,为淡黄色固体。ESI-MS(m/e):507.2[M+H]+.Mp147-149℃;1H NMR(300MHz,DMSO-d6:δ/ppm=10.990(d,J=14.1Hz,1H),8.501(dd,J1=5.1Hz,J2=10.2Hz,1H),8.185(s,3H),7.454(d,J=7.8Hz,1H),7.331(t,J=7.8Hz,1H),7.243-7.191(m,1H),7.162-7.085(m,6H),5.602(d,J=4.5Hz,1H),5.506(t,J=4.5Hz,1H),5.060(s,2H),4772(d,J=15.6Hz,1H),4.634(d,J=17.4Hz,1H),3.778-3.700(m,3H),3.426(m,1H),3.126-2.971(m,3H),2.574(m,1H),2.421(m,1H),1.553(m,2H),1.489-1.306(m,4H)。Following the procedure of Example 12, from 825 mg of (3S)-N-(6-Boc-Ser-amino-n-hexanoyl)-2,3,4,9-tetrahydro-β-carboline-3-carboxylic acid benzyl ester 448 mg (65%) of the title compound as a pale yellow solid. ESI-MS (m/e): 507.2[M+H] + .Mp 147-149°C; 1 H NMR (300MHz, DMSO-d6: δ/ppm=10.990 (d, J=14.1Hz, 1H), 8.501 ( dd, J 1 =5.1Hz, J 2 =10.2Hz, 1H), 8.185(s, 3H), 7.454(d, J=7.8Hz, 1H), 7.331(t, J=7.8Hz, 1H), 7.243- 7.191(m, 1H), 7.162-7.085(m, 6H), 5.602(d, J=4.5Hz, 1H), 5.506(t, J=4.5Hz, 1H), 5.060(s, 2H), 4772(d , J=15.6Hz, 1H), 4.634(d, J=17.4Hz, 1H), 3.778-3.700(m, 3H), 3.426(m, 1H), 3.126-2.971(m, 3H), 2.574(m, 1H), 2.421 (m, 1H), 1.553 (m, 2H), 1.489-1.306 (m, 4H).
实验例1评价化合物6a,b,c,d,e,f,g的抗肿瘤转移活性Experimental Example 1 Evaluation of the anti-tumor metastatic activity of
1)化合物6a,b,c,d,e,f,g用生理盐水溶解,RGDS四肽用生理盐水溶解作为阳性对照,生理盐水作为阴性对照;1)
2)化合物6a,b,c,d,e,f,g口服剂量为0.02μmol/kg/day,化合物4的口服剂量为0.2μmol/kg/day,生理盐水的口服剂量为0.2mL/20g/day,RGDS四肽口服剂量为20μmol/kg/day。肿瘤接种两周后小鼠腋下可观察到直径约为4~5mm的肿瘤,开始连续给药10天,共给药10次。2) The oral dose of
3)实验动物为C57BL/6雄性小鼠(清洁级),体重20±2g,每组12只小鼠。3) The experimental animals were C57BL/6 male mice (clean grade) with a body weight of 20±2g, 12 mice in each group.
4)Lewis小鼠肺癌细胞(LLC):LLC细胞购自ATCC,贴壁细胞;DMEM培养基。4) Lewis mouse lung cancer cells (LLC): LLC cells were purchased from ATCC, adherent cells; DMEM medium.
5)将LLC细胞进行消化,用生理盐水稀释至2×107个/mL,胎盘蓝(Tryanblue)拒染实验示活细胞数>95%。取C57BL/6雄性小鼠,雄性,左手固定小鼠,用75%乙醇消毒小鼠右前肢腋窝皮肤,右手持1mL无菌注射器于小鼠腋部皮下注射瘤细胞悬液0.2mL/只(含肿瘤细胞数约为2×106/mL)。小鼠接种后12~15天可以长出直径约1.5~2cm的肿瘤,作为瘤源备用。5) The LLC cells were digested and diluted with physiological saline to 2×10 7 cells/mL. The Tryanblue exclusion test showed that the number of viable cells was >95%. Take C57BL/6 male mice, male, fix the mouse with the left hand, sterilize the axillary skin of the right forelimb of the mouse with 75% ethanol, hold a 1 mL sterile syringe in the right hand and inject 0.2 mL/mouse of the tumor cell suspension into the axilla of the mice subcutaneously. The number of tumor cells was about 2×10 6 /mL). Tumors with a diameter of about 1.5 to 2 cm can grow in mice 12 to 15 days after inoculation, which can be used as tumor sources for future use.
6)取接种12~15天生长良好的Lewis肺癌荷瘤小鼠,颈椎脱臼,用75%乙醇浸泡消毒10min,在超净台内钝性剥离实体瘤,选择生长良好的瘤组织洗去浮血,在表面皿内剪碎,将剪碎的瘤组织放入玻璃组织匀浆器内,加入4℃预冷的生理盐水研磨成细胞悬液,用200目尼龙网过滤两次制成单细胞悬液。稀释细胞悬液至2×107个/mL,利用台盼蓝进行染色,计算活细胞数>95%。取C57BL/6雄性小鼠,将合格的细胞悬液按每只小鼠0.2mL接种于右侧腋皮下,观察。接种10~12天后小鼠腋下可观察到直径约为4~5mm的肿瘤,测量瘤体积,按照瘤体积平均分组,开始给药。治疗组小鼠每日灌胃给药化合物6a,b,c,d,e,f,g剂量为0.02μmol/kg/day,化合物4的口服剂量为0.2μmol/kg/day,。空白组小鼠每日灌胃给药生理盐水剂量为0.2mL/20g/day。阳性对照组小鼠每日灌胃给药RGDS四肽20μmol/kg/day,连续给药十天。6) Take Lewis lung cancer tumor-bearing mice that have been inoculated well for 12 to 15 days, dislocate their cervical vertebrae, soak and disinfect with 75% ethanol for 10 minutes, bluntly dissect solid tumors in an ultra-clean bench, and select well-growing tumor tissues to wash away the floating blood , cut into pieces in a watch dish, put the cut tumor tissue into a glass tissue homogenizer, add 4°C pre-cooled normal saline to grind into a cell suspension, filter twice with a 200-mesh nylon mesh to make a single-cell suspension liquid. Dilute the cell suspension to 2×10 7 cells/mL, stain with trypan blue, and calculate the number of viable cells >95%. Take C57BL/6 male mice, inoculate 0.2 mL of qualified cell suspension into the right armpit subcutaneously for each mouse, and observe. 10 to 12 days after inoculation, tumors with a diameter of about 4 to 5 mm could be observed in the armpits of the mice, and the tumor volume was measured, and the groups were averagely grouped according to the tumor volume, and the administration was started. The mice in the treatment group were given daily oral administration of
7)第十一天称小鼠体重,乙醚麻醉,脱颈椎处死小鼠,然后用镊子固定小鼠右腋肿瘤生长部位,剪开皮肤,暴露肿瘤,钝性剥离,称重;取小鼠的肺并记录肿瘤肺部转移的瘤节数。实验数据采用t检验和方差分析,转移瘤结数以(均值±SD g)表示,结果见表1。由表1可以看出,在0.02μmol/kg/day口服剂量下,化合物6a,b,c,d,e,f,g可有效地抑制小鼠肺肿瘤转移情况。本发明有显著的技术效果。7) On the eleventh day, the mice were weighed, anesthetized with ether, and the mice were sacrificed by removing the cervical vertebrae. Then, the tumor growth site of the right axilla of the mice was fixed with forceps, the skin was cut, the tumor was exposed, bluntly dissected, and weighed; Lungs and the number of tumor nodules with lung metastasis were recorded. The experimental data were analyzed by t test and analysis of variance, and the number of metastatic tumor nodes was expressed as (mean ± SD g). The results are shown in Table 1. It can be seen from Table 1 that at an oral dose of 0.02 μmol/kg/day,
表1各组化合物转移小鼠肺瘤结数Table 1 The number of lung tumor nodules in mice transferred by each group of compounds
a)与生理盐水比,P<0.01,与RGDS比,P>0.05;b)与生理盐水比,P<0.05,与RGDS比,P>0.05;c)与生理盐水比,P<0.05;n=8.a) Compared with normal saline, P<0.01, compared with RGDS, P>0.05; b) Compared with normal saline, P<0.05, compared with RGDS, P>0.05; c) Compared with normal saline, P<0.05; n =8.
实验例2评价化合物6a,b,c,d,e,f,g的抗炎活性Experimental Example 2 Evaluation of the anti-inflammatory activity of
1)化合物6a,b,c,d,e,f,g用生理盐水溶解,阿司匹林用生理盐水溶解作为阳性对照,生理盐水作为阴性对照;1)
2)化合物6a,b,c,d,e,f,g口服剂量为0.02μmol/kg/day,化合物4的口服剂量为0.2μmol/kg/day,生理盐水的口服剂量为0.2mL/20g/day,阿司匹林口服剂量为1.11mmol/kg/day。2) The oral dose of
3)实验动物为ICR雄性小鼠(清洁级),体重20±2g,随机分组,每组12只小鼠。3) The experimental animals were ICR male mice (clean grade), weighing 20±2g, randomly divided into groups, 12 mice in each group.
4)灌胃给药30min后,在小鼠左耳廓均匀涂抹30μL二甲苯,2h后小鼠接受乙醚麻醉,断颈处死,剪下左右两耳,用7mm的打孔器在两耳的相同位置取圆形耳片,称重,求出两耳肿胀差值作为肿胀度。即肿胀度=左耳圆片重量–右耳圆片重量。实验数据采用t检验和方差分析,耳肿胀度以(均值±SD mg)表示,结果见表2。由表2可以看出,在0.02μmol/kg/day口服剂量下,化合物6a,b,c,d,e,f,g可有效地抑制二甲苯引起的小鼠耳肿胀情况。本发明有显著的技术效果。4) After 30 minutes of intragastric administration, 30 μL of xylene was evenly applied to the left auricle of the mouse. After 2 hours, the mouse was anesthetized with ether, killed by cervical dislocation, and the left and right ears were cut off. A round ear piece was taken from the position, weighed, and the difference of the swelling of the two ears was calculated as the swelling degree. That is, the degree of swelling = the weight of the left ear disc - the weight of the right ear disc. The experimental data were analyzed by t test and analysis of variance, and the ear swelling was expressed as (mean ± SD mg). The results are shown in Table 2. It can be seen from Table 2 that at an oral dose of 0.02 μmol/kg/day,
表2各组化合物对二甲苯引起的小鼠耳肿胀的影响Table 2 Effects of each group of compounds on mouse ear swelling caused by xylene
a)与生理盐水比,P<0.01,与Aspirin比,P>0.05;b)与生理盐水比,P<0.01;a) Compared with normal saline, P<0.01, compared with Aspirin, P>0.05; b) Compared with normal saline, P<0.01;
c)与生理盐水比,P<0.05;n=12。c) Compared with normal saline, P<0.05; n=12.
Claims (3)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201810561635.7A CN110551119B (en) | 2018-06-04 | 2018-06-04 | 6-Aminoamido-n-hexanoylcarbolinecarboxylic acid benzyl ester, its preparation and application |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201810561635.7A CN110551119B (en) | 2018-06-04 | 2018-06-04 | 6-Aminoamido-n-hexanoylcarbolinecarboxylic acid benzyl ester, its preparation and application |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN110551119A CN110551119A (en) | 2019-12-10 |
| CN110551119B true CN110551119B (en) | 2020-12-01 |
Family
ID=68734014
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201810561635.7A Expired - Fee Related CN110551119B (en) | 2018-06-04 | 2018-06-04 | 6-Aminoamido-n-hexanoylcarbolinecarboxylic acid benzyl ester, its preparation and application |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN110551119B (en) |
Families Citing this family (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN116804040B (en) * | 2023-05-26 | 2024-08-06 | 首都医科大学 | Carboline oral small molecule anti-inflammatory medicament and preparation method and application thereof |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101597291A (en) * | 2008-06-04 | 2009-12-09 | 首都医科大学 | Benzyl 2-(aminoacyltryptophanyl)-β-tetrahydrocarboline-3-carboxylate and its preparation method and application |
| CN109912587A (en) * | 2017-12-12 | 2019-06-21 | 首都医科大学 | Side chain protected aminoamido-n-hexanoylcarboline carboxylate benzyl ester, its preparation, activity and application |
-
2018
- 2018-06-04 CN CN201810561635.7A patent/CN110551119B/en not_active Expired - Fee Related
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN101597291A (en) * | 2008-06-04 | 2009-12-09 | 首都医科大学 | Benzyl 2-(aminoacyltryptophanyl)-β-tetrahydrocarboline-3-carboxylate and its preparation method and application |
| CN109912587A (en) * | 2017-12-12 | 2019-06-21 | 首都医科大学 | Side chain protected aminoamido-n-hexanoylcarboline carboxylate benzyl ester, its preparation, activity and application |
Also Published As
| Publication number | Publication date |
|---|---|
| CN110551119A (en) | 2019-12-10 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN109912587B (en) | Side chain protection amido n-hexanoyl carboline benzyl carboxylate, preparation, activity and application thereof | |
| CN109111501B (en) | Fatty amino acid-modified indoleethanol derivatives, their synthesis, activities and applications | |
| CN110551119B (en) | 6-Aminoamido-n-hexanoylcarbolinecarboxylic acid benzyl ester, its preparation and application | |
| CN109912590B (en) | Amino acid and amino-n-hexanoic acid modified benzyl carboline carboxylate, its preparation, activity and application | |
| CN110551121B (en) | Glutaminamido-n-hexanoylcarboline carboxylate benzyl ester, its preparation, activity and application | |
| CN109912588B (en) | 6-amino amido n-hexanoyl carboline benzyl carboxylate, preparation, activity and application thereof | |
| CN109912589B (en) | Glutamine amido n-hexyl carboline carboxylic acid benzyl ester, preparation, activity and application thereof | |
| CN110551120B (en) | 6-amino amido n-hexanoyl carboline benzyl carboxylate, preparation, activity and application thereof | |
| CN108976226A (en) | The spinacin of nonpolar amino acid benzyl ester modification, its synthesis, activity rating and application | |
| CN108929320B (en) | 3R-indolylmethyl-6R-oxazolidinone modified piperazine-2, 5-dione, synthesis, activity and application thereof | |
| CN112010927B (en) | EGH and hydroxymethyl modified benzimidazole quinazoline, and synthesis, activity and application thereof | |
| CN108948137B (en) | 3S-Indoleethyl-6S-polar amino acid-modified piperazine-2,5-diones, their synthesis, activities and applications | |
| CN108947976B (en) | 3S-indolylmethyl-6S-aromatic amino acid-modified piperazine-2,5-diones, their synthesis, activities and applications | |
| CN108947978B (en) | 3R-Indolylmethyl-6R-Tyr-modified piperazine-2,5-diones, their synthesis, activities and applications | |
| CN110577568B (en) | Methyl indole and amide side chain amino acid modified diketopiperazine, synthesis, activity and application thereof | |
| CN112010926B (en) | KGH and hydroxymethyl modified benzimidazole quinazolines, synthesis, activity and applications thereof | |
| CN110577518B (en) | Methyl indole and amide side chain amino acid modified diketopiperazine, synthesis and application thereof | |
| CN112010811B (en) | 5-fluorouracil modified by theanine and phenylalanine together, and synthesis, activity and application thereof | |
| CN108976211B (en) | 3S-indolylmethyl-6R-aliphatic amino acid modified piperazine-2, 5-dione, and synthesis, activity and application thereof | |
| CN109081802B (en) | Basic amino acid-modified indoleethanol derivatives, their synthesis, activities and applications | |
| CN108947979B (en) | 3R-indolylmethyl-6R-polar amino acid modified piperazine-2, 5-dione, and synthesis, activity and application thereof | |
| CN109134328B (en) | Amino n-caproyl methyl cyclo amido n-caproyl Met, its synthesis, activity and application | |
| CN108976209B (en) | 3S-indolylmethyl-6R-piperazine-2, 5-dione modified by N-containing heterocyclic amino acid, and synthesis, activity and application thereof | |
| CN108976204B (en) | 3S-indolylmethyl-6R-aspartic acid modified piperazine-2, 5-dione, and synthesis, activity and application thereof | |
| CN108947981B (en) | 3R-indolylmethyl-6S-fatty amino acid modified piperazine-2,5-dione, its synthesis, activity and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20201201 |
|
| CF01 | Termination of patent right due to non-payment of annual fee |