CN112300245B - RGDS and theanine co-modified 5-fluorouracil, and synthesis, activity and application thereof - Google Patents
RGDS and theanine co-modified 5-fluorouracil, and synthesis, activity and application thereof Download PDFInfo
- Publication number
- CN112300245B CN112300245B CN201910695564.4A CN201910695564A CN112300245B CN 112300245 B CN112300245 B CN 112300245B CN 201910695564 A CN201910695564 A CN 201910695564A CN 112300245 B CN112300245 B CN 112300245B
- Authority
- CN
- China
- Prior art keywords
- obzl
- fluorouracil
- gly
- arg
- asp
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 230000000694 effects Effects 0.000 title abstract description 25
- NNRFRJQMBSBXGO-CIUDSAMLSA-N (3s)-3-[[2-[[(2s)-2-amino-5-(diaminomethylideneamino)pentanoyl]amino]acetyl]amino]-4-[[(1s)-1-carboxy-2-hydroxyethyl]amino]-4-oxobutanoic acid Chemical compound NC(N)=NCCC[C@H](N)C(=O)NCC(=O)N[C@@H](CC(O)=O)C(=O)N[C@@H](CO)C(O)=O NNRFRJQMBSBXGO-CIUDSAMLSA-N 0.000 title abstract description 12
- 101000829980 Homo sapiens Ral guanine nucleotide dissociation stimulator Proteins 0.000 title abstract description 9
- 102100023320 Ral guanine nucleotide dissociation stimulator Human genes 0.000 title abstract description 9
- DATAGRPVKZEWHA-YFKPBYRVSA-N L-Theanine Natural products CCNC(=O)CC[C@H]([NH3+])C([O-])=O DATAGRPVKZEWHA-YFKPBYRVSA-N 0.000 title abstract 3
- 229940026510 theanine Drugs 0.000 title abstract 2
- -1 theanine co-modified 5-fluorouracil Chemical class 0.000 title description 2
- 230000015572 biosynthetic process Effects 0.000 title 1
- 238000003786 synthesis reaction Methods 0.000 title 1
- 229960002949 fluorouracil Drugs 0.000 claims abstract description 76
- GHASVSINZRGABV-UHFFFAOYSA-N Fluorouracil Chemical compound FC1=CNC(=O)NC1=O GHASVSINZRGABV-UHFFFAOYSA-N 0.000 claims abstract description 24
- 206010028980 Neoplasm Diseases 0.000 claims abstract description 24
- 230000000259 anti-tumor effect Effects 0.000 claims abstract description 23
- 206010027476 Metastases Diseases 0.000 claims abstract description 15
- 230000009401 metastasis Effects 0.000 claims abstract description 15
- 239000003814 drug Substances 0.000 claims abstract description 6
- 239000002246 antineoplastic agent Substances 0.000 claims abstract description 4
- 229940041181 antineoplastic drug Drugs 0.000 claims abstract description 4
- 230000009977 dual effect Effects 0.000 claims abstract 2
- 239000000243 solution Substances 0.000 claims description 19
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 claims description 17
- 238000006243 chemical reaction Methods 0.000 claims description 17
- CPMUSDZOVZYEJJ-UHFFFAOYSA-N 2-(5-fluoro-2,4-dioxopyrimidin-1-yl)acetic acid Chemical compound OC(=O)CN1C=C(F)C(=O)NC1=O CPMUSDZOVZYEJJ-UHFFFAOYSA-N 0.000 claims description 16
- XEKOWRVHYACXOJ-UHFFFAOYSA-N Ethyl acetate Chemical compound CCOC(C)=O XEKOWRVHYACXOJ-UHFFFAOYSA-N 0.000 claims description 15
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 claims description 12
- 238000000034 method Methods 0.000 claims description 9
- BWHMMNNQKKPAPP-UHFFFAOYSA-L potassium carbonate Chemical compound [K+].[K+].[O-]C([O-])=O BWHMMNNQKKPAPP-UHFFFAOYSA-L 0.000 claims description 8
- 125000000999 tert-butyl group Chemical group [H]C([H])([H])C(*)(C([H])([H])[H])C([H])([H])[H] 0.000 claims description 8
- IXCSERBJSXMMFS-UHFFFAOYSA-N hydrogen chloride Substances Cl.Cl IXCSERBJSXMMFS-UHFFFAOYSA-N 0.000 claims description 5
- 229910000041 hydrogen chloride Inorganic materials 0.000 claims description 5
- KWYUFKZDYYNOTN-UHFFFAOYSA-M potassium hydroxide Substances [OH-].[K+] KWYUFKZDYYNOTN-UHFFFAOYSA-M 0.000 claims description 5
- 230000008878 coupling Effects 0.000 claims description 4
- 238000010168 coupling process Methods 0.000 claims description 4
- 238000005859 coupling reaction Methods 0.000 claims description 4
- WBJINCZRORDGAQ-UHFFFAOYSA-N ethyl formate Chemical compound CCOC=O WBJINCZRORDGAQ-UHFFFAOYSA-N 0.000 claims description 4
- 229910000027 potassium carbonate Inorganic materials 0.000 claims description 4
- 125000006239 protecting group Chemical group 0.000 claims description 4
- BNWCETAHAJSBFG-UHFFFAOYSA-N tert-butyl 2-bromoacetate Chemical compound CC(C)(C)OC(=O)CBr BNWCETAHAJSBFG-UHFFFAOYSA-N 0.000 claims description 4
- 239000007864 aqueous solution Substances 0.000 claims description 3
- 239000002253 acid Substances 0.000 claims description 2
- 239000003153 chemical reaction reagent Substances 0.000 claims description 2
- 150000002148 esters Chemical group 0.000 claims description 2
- 125000000896 monocarboxylic acid group Chemical group 0.000 claims 2
- VGALFAWDSNRXJK-VIFPVBQESA-N L-aspartic acid beta-benzyl ester Chemical compound OC(=O)[C@@H](N)CC(=O)OCC1=CC=CC=C1 VGALFAWDSNRXJK-VIFPVBQESA-N 0.000 claims 1
- 125000001797 benzyl group Chemical group [H]C1=C([H])C([H])=C(C([H])=C1[H])C([H])([H])* 0.000 claims 1
- 238000002360 preparation method Methods 0.000 abstract description 21
- 229940079593 drug Drugs 0.000 abstract description 4
- 150000001875 compounds Chemical class 0.000 description 43
- 210000004027 cell Anatomy 0.000 description 32
- 241000699670 Mus sp. Species 0.000 description 23
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 17
- 239000007787 solid Substances 0.000 description 13
- 238000002330 electrospray ionisation mass spectrometry Methods 0.000 description 12
- 241000699666 Mus <mouse, genus> Species 0.000 description 11
- RTZKZFJDLAIYFH-UHFFFAOYSA-N Diethyl ether Chemical compound CCOCC RTZKZFJDLAIYFH-UHFFFAOYSA-N 0.000 description 8
- LFQSCWFLJHTTHZ-UHFFFAOYSA-N Ethanol Chemical compound CCO LFQSCWFLJHTTHZ-UHFFFAOYSA-N 0.000 description 8
- 210000004369 blood Anatomy 0.000 description 8
- 239000008280 blood Substances 0.000 description 8
- 239000002609 medium Substances 0.000 description 8
- 210000004881 tumor cell Anatomy 0.000 description 7
- 206010051779 Bone marrow toxicity Diseases 0.000 description 6
- IAZDPXIOMUYVGZ-UHFFFAOYSA-N Dimethylsulphoxide Chemical compound CS(C)=O IAZDPXIOMUYVGZ-UHFFFAOYSA-N 0.000 description 6
- OKKJLVBELUTLKV-UHFFFAOYSA-N Methanol Chemical compound OC OKKJLVBELUTLKV-UHFFFAOYSA-N 0.000 description 6
- SJRJJKPEHAURKC-UHFFFAOYSA-N N-Methylmorpholine Chemical compound CN1CCOCC1 SJRJJKPEHAURKC-UHFFFAOYSA-N 0.000 description 6
- WYURNTSHIVDZCO-UHFFFAOYSA-N Tetrahydrofuran Chemical compound C1CCOC1 WYURNTSHIVDZCO-UHFFFAOYSA-N 0.000 description 6
- 210000001772 blood platelet Anatomy 0.000 description 6
- 231100000366 bone marrow toxicity Toxicity 0.000 description 6
- 210000000265 leukocyte Anatomy 0.000 description 6
- UCSJYZPVAKXKNQ-HZYVHMACSA-N streptomycin Chemical compound CN[C@H]1[C@H](O)[C@@H](O)[C@H](CO)O[C@H]1O[C@@H]1[C@](C=O)(O)[C@H](C)O[C@H]1O[C@@H]1[C@@H](NC(N)=N)[C@H](O)[C@@H](NC(N)=N)[C@H](O)[C@H]1O UCSJYZPVAKXKNQ-HZYVHMACSA-N 0.000 description 6
- 239000012091 fetal bovine serum Substances 0.000 description 5
- 230000012010 growth Effects 0.000 description 5
- 230000002829 reductive effect Effects 0.000 description 5
- CURLTUGMZLYLDI-UHFFFAOYSA-N Carbon dioxide Chemical compound O=C=O CURLTUGMZLYLDI-UHFFFAOYSA-N 0.000 description 4
- DTQVDTLACAAQTR-UHFFFAOYSA-N Trifluoroacetic acid Chemical compound OC(=O)C(F)(F)F DTQVDTLACAAQTR-UHFFFAOYSA-N 0.000 description 4
- 210000001099 axilla Anatomy 0.000 description 4
- 239000000706 filtrate Substances 0.000 description 4
- 239000000834 fixative Substances 0.000 description 4
- 210000004072 lung Anatomy 0.000 description 4
- 208000002154 non-small cell lung carcinoma Diseases 0.000 description 4
- 239000011541 reaction mixture Substances 0.000 description 4
- 238000003756 stirring Methods 0.000 description 4
- 210000001519 tissue Anatomy 0.000 description 4
- 208000029729 tumor suppressor gene on chromosome 11 Diseases 0.000 description 4
- YMWUJEATGCHHMB-UHFFFAOYSA-N Dichloromethane Chemical compound ClCCl YMWUJEATGCHHMB-UHFFFAOYSA-N 0.000 description 3
- 229930182555 Penicillin Natural products 0.000 description 3
- JGSARLDLIJGVTE-MBNYWOFBSA-N Penicillin G Chemical compound N([C@H]1[C@H]2SC([C@@H](N2C1=O)C(O)=O)(C)C)C(=O)CC1=CC=CC=C1 JGSARLDLIJGVTE-MBNYWOFBSA-N 0.000 description 3
- 230000003698 anagen phase Effects 0.000 description 3
- 108010089975 arginyl-glycyl-aspartyl-serine Proteins 0.000 description 3
- 230000037396 body weight Effects 0.000 description 3
- 239000006285 cell suspension Substances 0.000 description 3
- 230000007547 defect Effects 0.000 description 3
- 230000003203 everyday effect Effects 0.000 description 3
- 239000012530 fluid Substances 0.000 description 3
- 238000000227 grinding Methods 0.000 description 3
- 238000011081 inoculation Methods 0.000 description 3
- 206010061289 metastatic neoplasm Diseases 0.000 description 3
- 229940049954 penicillin Drugs 0.000 description 3
- 229960005322 streptomycin Drugs 0.000 description 3
- YLQBMQCUIZJEEH-UHFFFAOYSA-N tetrahydrofuran Natural products C=1C=COC=1 YLQBMQCUIZJEEH-UHFFFAOYSA-N 0.000 description 3
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 description 3
- ASOKPJOREAFHNY-UHFFFAOYSA-N 1-Hydroxybenzotriazole Chemical compound C1=CC=C2N(O)N=NC2=C1 ASOKPJOREAFHNY-UHFFFAOYSA-N 0.000 description 2
- 206010002091 Anaesthesia Diseases 0.000 description 2
- 206010003445 Ascites Diseases 0.000 description 2
- 108091003079 Bovine Serum Albumin Proteins 0.000 description 2
- 238000011740 C57BL/6 mouse Methods 0.000 description 2
- 229920000742 Cotton Polymers 0.000 description 2
- 206010058467 Lung neoplasm malignant Diseases 0.000 description 2
- 229930040373 Paraformaldehyde Natural products 0.000 description 2
- 239000012980 RPMI-1640 medium Substances 0.000 description 2
- 230000037005 anaesthesia Effects 0.000 description 2
- 210000001185 bone marrow Anatomy 0.000 description 2
- 239000001569 carbon dioxide Substances 0.000 description 2
- 229910002092 carbon dioxide Inorganic materials 0.000 description 2
- 230000004709 cell invasion Effects 0.000 description 2
- 230000012292 cell migration Effects 0.000 description 2
- 230000003833 cell viability Effects 0.000 description 2
- 238000004440 column chromatography Methods 0.000 description 2
- 239000013078 crystal Substances 0.000 description 2
- 238000001914 filtration Methods 0.000 description 2
- 210000003194 forelimb Anatomy 0.000 description 2
- 238000010253 intravenous injection Methods 0.000 description 2
- 230000009545 invasion Effects 0.000 description 2
- 201000005202 lung cancer Diseases 0.000 description 2
- 208000020816 lung neoplasm Diseases 0.000 description 2
- 230000001394 metastastic effect Effects 0.000 description 2
- 230000005012 migration Effects 0.000 description 2
- 238000013508 migration Methods 0.000 description 2
- 229920002866 paraformaldehyde Polymers 0.000 description 2
- 239000002504 physiological saline solution Substances 0.000 description 2
- 239000013641 positive control Substances 0.000 description 2
- 239000012679 serum free medium Substances 0.000 description 2
- 238000010898 silica gel chromatography Methods 0.000 description 2
- 239000011780 sodium chloride Substances 0.000 description 2
- 238000004659 sterilization and disinfection Methods 0.000 description 2
- 239000006228 supernatant Substances 0.000 description 2
- 230000001629 suppression Effects 0.000 description 2
- ITMCEJHCFYSIIV-UHFFFAOYSA-N triflic acid Chemical compound OS(=O)(=O)C(F)(F)F ITMCEJHCFYSIIV-UHFFFAOYSA-N 0.000 description 2
- 230000004614 tumor growth Effects 0.000 description 2
- SOHLZANWVLCPHK-LBPRGKRZSA-N (2s)-2-[(2-methylpropan-2-yl)oxycarbonylamino]-4-oxo-4-phenylmethoxybutanoic acid Chemical compound CC(C)(C)OC(=O)N[C@H](C(O)=O)CC(=O)OCC1=CC=CC=C1 SOHLZANWVLCPHK-LBPRGKRZSA-N 0.000 description 1
- 206010067484 Adverse reaction Diseases 0.000 description 1
- JXYACYYPACQCDM-UHFFFAOYSA-N Benzyl glycinate Chemical compound NCC(=O)OCC1=CC=CC=C1 JXYACYYPACQCDM-UHFFFAOYSA-N 0.000 description 1
- 206010005003 Bladder cancer Diseases 0.000 description 1
- 206010006187 Breast cancer Diseases 0.000 description 1
- 208000026310 Breast neoplasm Diseases 0.000 description 1
- 206010012735 Diarrhoea Diseases 0.000 description 1
- 239000006144 Dulbecco’s modified Eagle's medium Substances 0.000 description 1
- KCXVZYZYPLLWCC-UHFFFAOYSA-N EDTA Chemical compound OC(=O)CN(CC(O)=O)CCN(CC(O)=O)CC(O)=O KCXVZYZYPLLWCC-UHFFFAOYSA-N 0.000 description 1
- 201000008808 Fibrosarcoma Diseases 0.000 description 1
- 241001465754 Metazoa Species 0.000 description 1
- 206010028813 Nausea Diseases 0.000 description 1
- 239000004677 Nylon Substances 0.000 description 1
- 206010033128 Ovarian cancer Diseases 0.000 description 1
- 206010061535 Ovarian neoplasm Diseases 0.000 description 1
- CZPWVGJYEJSRLH-UHFFFAOYSA-N Pyrimidine Chemical compound C1=CN=CN=C1 CZPWVGJYEJSRLH-UHFFFAOYSA-N 0.000 description 1
- 241000700159 Rattus Species 0.000 description 1
- 206010039491 Sarcoma Diseases 0.000 description 1
- UIIMBOGNXHQVGW-UHFFFAOYSA-M Sodium bicarbonate Chemical class [Na+].OC([O-])=O UIIMBOGNXHQVGW-UHFFFAOYSA-M 0.000 description 1
- GLNADSQYFUSGOU-GPTZEZBUSA-J Trypan blue Chemical compound [Na+].[Na+].[Na+].[Na+].C1=C(S([O-])(=O)=O)C=C2C=C(S([O-])(=O)=O)C(/N=N/C3=CC=C(C=C3C)C=3C=C(C(=CC=3)\N=N\C=3C(=CC4=CC(=CC(N)=C4C=3O)S([O-])(=O)=O)S([O-])(=O)=O)C)=C(O)C2=C1N GLNADSQYFUSGOU-GPTZEZBUSA-J 0.000 description 1
- 208000007097 Urinary Bladder Neoplasms Diseases 0.000 description 1
- 206010047700 Vomiting Diseases 0.000 description 1
- 230000006838 adverse reaction Effects 0.000 description 1
- 238000010171 animal model Methods 0.000 description 1
- 230000000340 anti-metabolite Effects 0.000 description 1
- 229940100197 antimetabolite Drugs 0.000 description 1
- 239000002256 antimetabolite Substances 0.000 description 1
- 125000002619 bicyclic group Chemical group 0.000 description 1
- 210000004204 blood vessel Anatomy 0.000 description 1
- 210000005252 bulbus oculi Anatomy 0.000 description 1
- 230000001413 cellular effect Effects 0.000 description 1
- 238000005119 centrifugation Methods 0.000 description 1
- 239000012141 concentrate Substances 0.000 description 1
- 239000012153 distilled water Substances 0.000 description 1
- CCGKOQOJPYTBIH-UHFFFAOYSA-N ethenone Chemical compound C=C=O CCGKOQOJPYTBIH-UHFFFAOYSA-N 0.000 description 1
- 239000012065 filter cake Substances 0.000 description 1
- 238000010579 first pass effect Methods 0.000 description 1
- 238000004108 freeze drying Methods 0.000 description 1
- 230000002496 gastric effect Effects 0.000 description 1
- 210000001035 gastrointestinal tract Anatomy 0.000 description 1
- 239000000499 gel Substances 0.000 description 1
- 239000011521 glass Substances 0.000 description 1
- 230000036541 health Effects 0.000 description 1
- 238000001802 infusion Methods 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 238000001990 intravenous administration Methods 0.000 description 1
- 230000000670 limiting effect Effects 0.000 description 1
- 201000007270 liver cancer Diseases 0.000 description 1
- 208000014018 liver neoplasm Diseases 0.000 description 1
- 238000012986 modification Methods 0.000 description 1
- 230000004048 modification Effects 0.000 description 1
- PBMIETCUUSQZCG-UHFFFAOYSA-N n'-cyclohexylmethanediimine Chemical compound N=C=NC1CCCCC1 PBMIETCUUSQZCG-UHFFFAOYSA-N 0.000 description 1
- JKRHDMPWBFBQDZ-UHFFFAOYSA-N n'-hexylmethanediimine Chemical compound CCCCCCN=C=N JKRHDMPWBFBQDZ-UHFFFAOYSA-N 0.000 description 1
- 230000008693 nausea Effects 0.000 description 1
- 230000007935 neutral effect Effects 0.000 description 1
- 229920001778 nylon Polymers 0.000 description 1
- 210000003200 peritoneal cavity Anatomy 0.000 description 1
- 239000000843 powder Substances 0.000 description 1
- 239000002244 precipitate Substances 0.000 description 1
- 238000000746 purification Methods 0.000 description 1
- 230000009467 reduction Effects 0.000 description 1
- 238000001228 spectrum Methods 0.000 description 1
- 238000010186 staining Methods 0.000 description 1
- 238000007920 subcutaneous administration Methods 0.000 description 1
- 239000000725 suspension Substances 0.000 description 1
- 238000001308 synthesis method Methods 0.000 description 1
- 238000012360 testing method Methods 0.000 description 1
- 238000011287 therapeutic dose Methods 0.000 description 1
- 231100000419 toxicity Toxicity 0.000 description 1
- 230000001988 toxicity Effects 0.000 description 1
- 201000005112 urinary bladder cancer Diseases 0.000 description 1
- 230000008673 vomiting Effects 0.000 description 1
Images
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K7/00—Peptides having 5 to 20 amino acids in a fully defined sequence; Derivatives thereof
- C07K7/04—Linear peptides containing only normal peptide links
- C07K7/06—Linear peptides containing only normal peptide links having 5 to 11 amino acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/513—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim having oxo groups directly attached to the heterocyclic ring, e.g. cytosine
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K47/00—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient
- A61K47/50—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates
- A61K47/51—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent
- A61K47/62—Medicinal preparations characterised by the non-active ingredients used, e.g. carriers or inert additives; Targeting or modifying agents chemically bound to the active ingredient the non-active ingredient being chemically bound to the active ingredient, e.g. polymer-drug conjugates the non-active ingredient being a modifying agent the modifying agent being a protein, peptide or polyamino acid
- A61K47/64—Drug-peptide, drug-protein or drug-polyamino acid conjugates, i.e. the modifying agent being a peptide, protein or polyamino acid which is covalently bonded or complexed to a therapeutically active agent
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
- A61P35/04—Antineoplastic agents specific for metastasis
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02P—CLIMATE CHANGE MITIGATION TECHNOLOGIES IN THE PRODUCTION OR PROCESSING OF GOODS
- Y02P20/00—Technologies relating to chemical industry
- Y02P20/50—Improvements relating to the production of bulk chemicals
- Y02P20/55—Design of synthesis routes, e.g. reducing the use of auxiliary or protecting groups
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Medicinal Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Organic Chemistry (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Pharmacology & Pharmacy (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Epidemiology (AREA)
- Molecular Biology (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Engineering & Computer Science (AREA)
- Oncology (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
本发明公开了下式的RGDS和茶氨酸共同修饰的5‑氟尿嘧啶。公开了它的制备方法,公开了它的抗肿瘤和抗肿瘤转移活性,因而本发明公开了它在制备抗肿瘤药物中的应用,在制备抗肿瘤转移药物中的应用和在制备抗肿瘤及抗肿瘤转移双重活性药物中的应用。
The invention discloses 5-fluorouracil co-modified by RGDS of the following formula and theanine. Its preparation method is disclosed, its anti-tumor and anti-tumor metastasis activities are disclosed, so the present invention discloses its application in the preparation of anti-tumor drugs, its application in the preparation of anti-tumor metastasis drugs and its application in the preparation of anti-tumor and anti-tumor metastasis Application of Dual Active Drugs in Tumor Metastasis.
Description
技术领域technical field
本发明涉及1-(CH2CO-Arg-Gly-Asp-Ser)-3-(CH2CO-The)-5-氟尿嘧啶,涉及它的制备方法,涉及它的抗肿瘤活性和抗肿瘤转移活性,因而本发明涉及它在制备抗肿瘤药物中的应用,涉及它在制备抗肿瘤转移药物中的应用,以及涉及它在制备具有抗肿瘤及抗肿瘤转移双重作用药物中的应用。本发明属于生物医药领域。The present invention relates to 1-(CH 2 CO-Arg-Gly-Asp-Ser)-3-(CH 2 CO-The)-5-fluorouracil, to its preparation method, to its anti-tumor activity and anti-tumor metastasis activity , and thus the present invention relates to its application in the preparation of antitumor drugs, its application in the preparation of anti-tumor metastasis drugs, and its application in the preparation of anti-tumor and anti-tumor metastasis drugs. The invention belongs to the field of biomedicine.
背景技术Background technique
5-氟尿嘧啶(5-FU)是嘧啶类抗代谢的抗肿瘤药物,抗瘤谱较广。多用于治疗消化道肿瘤,乳腺癌,卵巢癌,膀胱癌,肝癌。5-FU在临床应用中存在一些缺陷。例如口服首过效应明显,临床给药途径为静脉注射。静脉注射的半衰期不超过20min。为此,5-FU在临床应用中多采用静脉持续滴注。患者对静脉持续滴注依从性差。又例如5-FU的治疗剂量较大,对肿瘤的选择性差,会出现明显胃肠道反应(恶心,呕吐,腹泻)及骨髓抑制(血小板及白细胞数降低)等不良反应。这些缺陷限制了5-FU的临床应用。为了克服5-FU的缺陷,曾经完成了大量结构修饰。可是没有达到预期效果。发明人经过数年探索发现,在5-氟尿嘧啶的1位和3位分别用CH2CO-Arg-Gly-Asp-Ser及CH2CO-The修饰不仅可以在极低剂量下口服,可以避免骨髓毒性,可以增强抗肿瘤活性,而且可以获得抗肿瘤转移活性。根据这些发现,发明人提出了本发明。5-Fluorouracil (5-FU) is a pyrimidine antimetabolite antineoplastic drug with a broad antitumor spectrum. It is mostly used in the treatment of digestive tract tumors, breast cancer, ovarian cancer, bladder cancer, and liver cancer. 5-FU has some defects in clinical application. For example, the oral first-pass effect is obvious, and the clinical route of administration is intravenous injection. The half-life of intravenous injection does not exceed 20 minutes. For this reason, 5-FU is often infused intravenously in clinical practice. Patients have poor compliance with continuous intravenous infusion. Another example is the large therapeutic dose of 5-FU, which has poor selectivity for tumors, and adverse reactions such as obvious gastrointestinal reactions (nausea, vomiting, diarrhea) and bone marrow suppression (decrease in the number of platelets and white blood cells) may occur. These defects limit the clinical application of 5-FU. In order to overcome the defects of 5-FU, a large number of structural modifications have been done. But it didn't achieve the desired effect. After several years of exploration, the inventors found that modifying the 1-position and 3-position of 5-fluorouracil with CH 2 CO-Arg-Gly-Asp-Ser and CH 2 CO-The can not only be administered orally at a very low dose, but also can avoid bone marrow toxicity, can enhance anti-tumor activity, and can obtain anti-tumor metastasis activity. Based on these findings, the inventors have made the present invention.
发明内容Contents of the invention
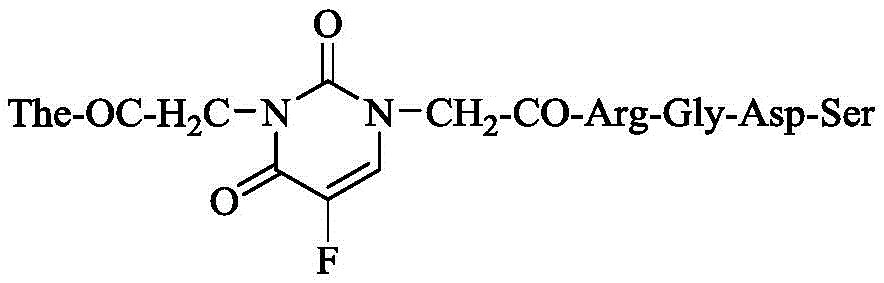
本发明的第一个内容是提供下式的1-(CH2CO-Arg-Gly-Asp-Ser)-3-(CH2CO-The)-5-氟尿嘧啶。The first content of the present invention is to provide 1-(CH 2 CO-Arg-Gly-Asp-Ser)-3-(CH 2 CO-The)-5-fluorouracil of the following formula.
本发明的第二个内容是提供1-(CH2CO-Arg-Gly-Asp-Ser)-3-(CH2CO-The)-5-氟尿嘧啶的合成方法,该方法包括:The second content of the present invention is to provide a synthesis method of 1-(CH 2 CO-Arg-Gly-Asp-Ser)-3-(CH 2 CO-The)-5-fluorouracil, the method comprising:
(1)在KOH水溶液中5-氟尿嘧啶与溴乙酸60℃反应8h,然后0℃用浓盐酸处理,生成1-羧甲基-5-氟尿嘧啶;(1) React 5-fluorouracil with bromoacetic acid at 60°C for 8 hours in KOH aqueous solution, then treat with concentrated hydrochloric acid at 0°C to generate 1-carboxymethyl-5-fluorouracil;
(2)将1-羧甲基-5-氟尿嘧啶与Arg(NO2)-Gly-OBzl偶联,制备1-[CH2CO-Arg(NO2)-Gly-OBzl]-5-氟尿嘧啶;(2) Coupling 1-carboxymethyl-5-fluorouracil with Arg(NO 2 )-Gly-OBzl to prepare 1-[CH 2 CO-Arg(NO 2 )-Gly-OBzl]-5-fluorouracil;
(3)1-[CH2CO-Arg(NO2)-Gly-OBzl]-5-氟尿嘧啶在碳酸钾条件下,与溴乙酸叔丁酯反应,制备1-[CH2CO-Arg(NO2)-Gly-OBzl]-3-(CH2CO2tBu)-5-氟尿嘧啶;(3) 1-[CH 2 CO-Arg(NO 2 )-Gly-OBzl]-5-fluorouracil reacts with tert-butyl bromoacetate under the condition of potassium carbonate to prepare 1-[CH 2 CO-Arg(NO 2 )-Gly-OBzl]-3-( CH2CO2tBu )-5 - fluorouracil;
(4)1-[CH2CO-Arg(NO2)-Gly-OBzl]-3-(CH2CO2tBu)-5-氟尿嘧啶在2N NaOH溶液中脱除苄酯保护基,制备1-[CH2CO-Arg(NO2)-Gly]-3-(CH2CO2tBu)-5-氟尿嘧啶;(4) 1-[CH 2 CO-Arg(NO 2 )-Gly-OBzl]-3-(CH 2 CO 2 tBu)-5-fluorouracil deprotected benzyl ester in 2N NaOH solution to prepare 1-[ CH 2 CO-Arg(NO 2 )-Gly]-3-(CH 2 CO 2 tBu)-5-fluorouracil;
(5)1-[CH2CO-Arg(NO2)-Gly]-3-(CH2CO2tBu)-5-氟尿嘧啶与Asp(OBzl)-Ser-OBzl偶联,制备1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2CO2tBu)-5-氟尿嘧啶;(5) 1-[CH 2 CO-Arg(NO 2 )-Gly]-3-(CH 2 CO 2 tBu)-5-fluorouracil coupled with Asp(OBzl)-Ser-OBzl to prepare 1-[CH 2 CO-Arg(NO 2 )-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH 2 CO 2 tBu)-5-fluorouracil;
(6)1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2CO2tBu)-5-氟尿嘧啶在浓度为4N的氯化氢/乙酸乙酯试剂中脱除叔丁酯保护基,制备1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2COOH)-5-氟尿嘧啶;(6) 1-[CH 2 CO-Arg(NO 2 )-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH 2 CO 2 tBu)-5-fluorouracil in 4N hydrogen chloride/ethyl acetate Remove the tert-butyl ester protecting group in the ester reagent to prepare 1-[CH 2 CO-Arg(NO 2 )-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH 2 COOH)-5-fluorouracil;
(7)1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2COOH)-5-氟尿嘧啶与The-OBzl偶联,制备1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2CO-The-OBzl)-5-氟尿嘧啶;(7) 1-[CH 2 CO-Arg(NO 2 )-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH 2 COOH)-5-fluorouracil is coupled with The-OBzl to prepare 1-[ CH2CO -Arg(NO2)-Gly - Asp(OBzl)-Ser-OBzl]-3-( CH2CO -The-OBzl)-5-fluorouracil;
(8)将1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2CO-The-OBzl)-5-氟尿嘧啶经酸脱反应脱除保护基,制备1-(CH2CO-Arg-Gly-Asp-Ser)-3-(CH2CO-The)-5-氟尿嘧啶。(8) Remove 1-[CH 2 CO-Arg(NO 2 )-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH 2 CO-The-OBzl)-5-fluorouracil by acid dereaction Protecting group, preparation of 1-(CH 2 CO-Arg-Gly-Asp-Ser)-3-(CH 2 CO-The)-5-fluorouracil.
本发明的第三个内容是评价1-(CH2CO-Arg-Gly-Asp-Ser)-3-(CH2CO-The)-5-氟尿嘧啶的抗肿瘤活性。The third content of the present invention is to evaluate the antitumor activity of 1-(CH 2 CO-Arg-Gly-Asp-Ser)-3-(CH 2 CO-The)-5-fluorouracil.
本发明的第四个内容是评价1-(CH2CO-Arg-Gly-Asp-Ser)-3-(CH2CO-The)-5-氟尿嘧啶的抗肿瘤转移活性。The fourth content of the present invention is to evaluate the anti-tumor metastasis activity of 1-(CH 2 CO-Arg-Gly-Asp-Ser)-3-(CH 2 CO-The)-5-fluorouracil.
本发明的第五个内容是评价1-(CH2CO-Arg-Gly-Asp-Ser)-3-(CH2CO-The)-5-氟尿嘧啶的骨髓毒性。The fifth content of the present invention is to evaluate the bone marrow toxicity of 1-(CH 2 CO-Arg-Gly-Asp-Ser)-3-(CH 2 CO-The)-5-fluorouracil.
附图说明Description of drawings
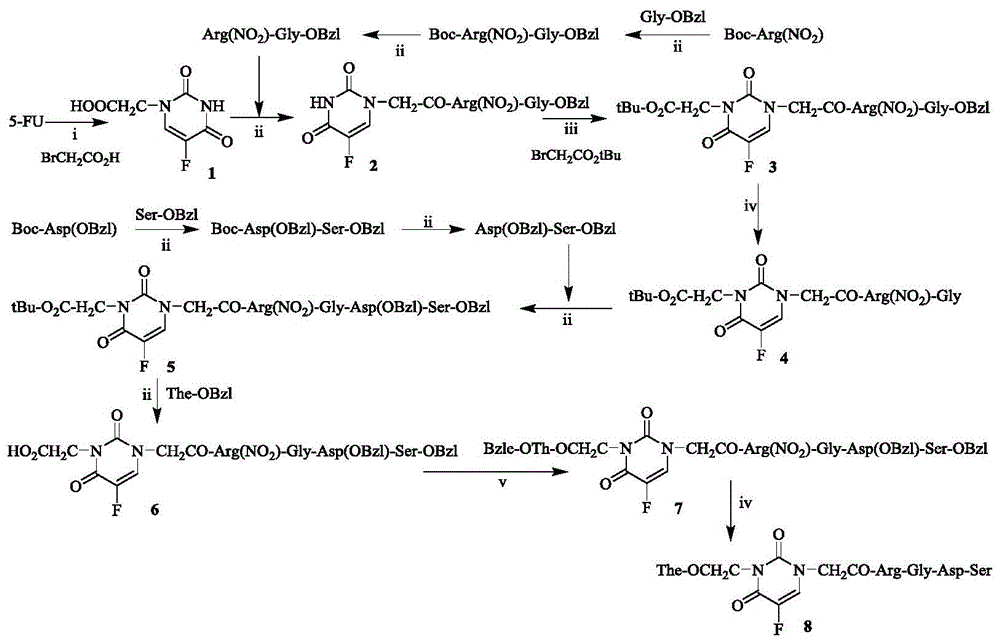
图1 1-(CH2CO-Arg-Gly-Asp-Ser)-3-(CH2CO-The)-5-氟尿嘧啶的合成路线.i)60℃,溴乙酸,浓盐酸;ii)二环己基碳二亚胺,1-羟基苯并三唑,N-甲基吗啉;iii)碳酸钾,溴乙酸叔丁酯;iv)氢氧化钠溶液(2M);v)氯化氢/乙酸乙酯溶液(4M);vi)三氟乙酸,三氟甲磺酸。Figure 1 Synthetic route of 1-(CH 2 CO-Arg-Gly-Asp-Ser)-3-(CH 2 CO-The)-5-fluorouracil. i) 60℃, bromoacetic acid, concentrated hydrochloric acid; ii) bicyclic Hexylcarbodiimide, 1-hydroxybenzotriazole, N-methylmorpholine; iii) potassium carbonate, tert-butyl bromoacetate; iv) sodium hydroxide solution (2M); v) hydrogen chloride/ethyl acetate solution (4M); vi) trifluoroacetic acid, trifluoromethanesulfonic acid.
具体实施方式detailed description
为了进一步阐述本发明,下面给出一系列实施例。这些实施例完全是例证性的,它们仅用来对本发明进行具体描述,不应当理解为对本发明的限制。In order to further illustrate the present invention, a series of examples are given below. These examples are entirely illustrative, and they are only used to specifically describe the present invention, and should not be construed as limiting the present invention.
实施例1制备1-(CH2CO2H)-5-氟尿嘧啶(1)Example 1 Preparation of 1-(CH 2 CO 2 H)-5-fluorouracil (1)
0℃将2.6g(20mmol)5-氟尿嘧啶(5FU)用氢氧化钾水溶液溶解。之后于60℃活化1小时。向内滴加4.14g(30mmol)溴乙酸水溶液,60℃搅拌8h。TLC检测反应完全后反应混合物冷却至室温。反应混合物于0℃用浓盐酸调节pH至5,搅拌30min。过滤,滤液滴加浓盐酸调节pH至2。0℃搅拌2.5h。过滤,滤渣用蒸馏水洗3次,晾干得3.05g(81%)标题化合物,为无色固体。ESI-MS(m/e):187[M-H]-。1H NMR(300MHz,DMSO-d6):δ/ppm=11.889(d,J=2.7Hz,1H),8.069(d,J=3.9Hz,1H),4.366(s,1H)。2.6 g (20 mmol) of 5-fluorouracil (5FU) was dissolved in an aqueous potassium hydroxide solution at 0°C. Then activate at 60°C for 1 hour. Add 4.14g (30mmol) bromoacetic acid aqueous solution dropwise, and stir at 60°C for 8h. After the reaction was detected by TLC, the reaction mixture was cooled to room temperature. The reaction mixture was adjusted to
实施例2制备Boc-Arg(NO2)-Gly-OBzlExample 2 Preparation of Boc-Arg(NO 2 )-Gly-OBzl
0℃将3.19g(10mmol)Boc-Arg(NO2)用无水四氢呋喃溶解(THF),向得到的溶液中依次加入1.35g(10mmol)1-羟基苯并三唑和2.68g(13mmol)二环己基羰二亚胺。搅拌30min后,向反应液中加入3.71g(1.1mmol)Gly-OBzl的四氢呋喃溶液,反应液于0℃滴加N-甲基吗啉,调节pH 8。反应液室温搅拌至TLC显示反应完全。反应混合物过滤,滤液减压浓缩,残留物用乙酸乙酯溶液溶解。经硅胶柱层析纯化得3.53g(75%)标题化合物,为无色固体。ESI-MS(m/e):467[M+H]+。Dissolve 3.19g (10mmol) Boc-Arg (NO 2 ) in anhydrous tetrahydrofuran (THF) at 0°C, and add 1.35g (10mmol) 1-hydroxybenzotriazole and 2.68g (13mmol) bis Cyclohexylcarbodiimide. After stirring for 30 min, 3.71 g (1.1 mmol) of Gly-OBzl in tetrahydrofuran was added to the reaction solution, and N-methylmorpholine was added dropwise to the reaction solution at 0°C to adjust the pH to 8. The reaction solution was stirred at room temperature until TLC showed that the reaction was complete. The reaction mixture was filtered, the filtrate was concentrated under reduced pressure, and the residue was dissolved in ethyl acetate solution. Purification by silica gel column chromatography gave 3.53 g (75%) of the title compound as a colorless solid. ESI-MS (m/e): 467 [M+H] + .
实施例3制备Arg(NO2)-Gly-OBzlExample 3 Preparation of Arg(NO 2 )-Gly-OBzl
0℃将466mg(1mmol)Boc-Arg(NO2)-Gly-OBzl溶于氯化氢的乙酸乙酯溶液(4M),搅拌至TLC显示反应完全。之后,反应液37℃减压浓缩,使彻底除去游离氯化氢。得到的固体悬浮于5mL无水乙醚中,充分洗涤。收集沉淀,得389mg(96%)标题化合物,为无色固体。ESI-MS(m/e):365[M-H]-。466 mg (1 mmol) Boc-Arg(NO 2 )-Gly-OBzl was dissolved in hydrogen chloride in ethyl acetate solution (4M) at 0°C, and stirred until TLC showed that the reaction was complete. Afterwards, the reaction solution was concentrated under reduced pressure at 37°C to completely remove free hydrogen chloride. The obtained solid was suspended in 5mL of anhydrous ether and washed thoroughly. The precipitate was collected to give 389 mg (96%) of the title compound as a colorless solid. ESI-MS (m/e): 365 [MH] - .
实施例4制备1-[CH2CO-Arg(NO2)-Gly-OBzl]-5-氟尿嘧啶(2)Example 4 Preparation of 1-[CH 2 CO-Arg(NO 2 )-Gly-OBzl]-5-fluorouracil (2)
采用实施例2的方法从376mg(2mmol)1-(CH2CO2H)-5-氟尿嘧啶(1)和805mg(2mmol)Arg(NO2)-Gly-OBzl得到491mg(46%)标题化合物,为无色固体。ESI-MS(m/e):535[M-H]-;1HNMR(300MHz,DMSO-d6):δ/ppm=11.833(s,1H),8.481(m,3H),7.989(d,J=6.9Hz,1H),7.792(s,2H),7.365(s,5H),5.129(s,2H),4.346(m,3H),3.917(m,2H),3.128(m,2H),1.689(m,1H),1.512(m,3H)。Using the method of Example 2, 491 mg (46%) of the title compound were obtained from 376 mg (2 mmol) 1-(CH 2 CO 2 H)-5-fluorouracil (1) and 805 mg (2 mmol) Arg(NO 2 )-Gly-OBzl, It is a colorless solid. ESI-MS (m/e): 535[MH] - ; 1 HNMR (300MHz, DMSO-d 6 ): δ/ppm = 11.833 (s, 1H), 8.481 (m, 3H), 7.989 (d, J = 6.9Hz,1H),7.792(s,2H),7.365(s,5H),5.129(s,2H),4.346(m,3H),3.917(m,2H),3.128(m,2H),1.689( m,1H), 1.512(m,3H).
实施例5制备1-[CH2CO-Arg(NO2)-Gly-OBzl]-3-(CH2CO2tBu)-5-氟尿嘧啶(3)Example 5 Preparation of 1-[CH 2 CO-Arg(NO 2 )-Gly-OBzl]-3-(CH 2 CO 2 tBu)-5-fluorouracil (3)
将536mg(1mmol)1-[CH2CO-Arg(NO2)-Gly-OBzl]-5-氟尿嘧啶(2)用DMF溶解,于0℃加入276mg碳酸钾,搅拌1h。之后,缓慢滴加390mg(2mmol)溴乙酸叔丁酯。反应混合物搅拌至TLC检测反应完全,滤除不溶物,滤液减压浓缩。得到的黄色油状物用二氯甲烷复溶,经硅胶柱层析,得到271mg(41%)标题化合物,为无色固体。Dissolve 536mg (1mmol) of 1-[CH 2 CO-Arg(NO 2 )-Gly-OBzl]-5-fluorouracil (2) in DMF, add 276mg of potassium carbonate at 0°C, and stir for 1h. After that, 390 mg (2 mmol) tert-butyl bromoacetate was slowly added dropwise. The reaction mixture was stirred until the reaction was complete as detected by TLC, the insoluble matter was filtered off, and the filtrate was concentrated under reduced pressure. The obtained yellow oil was redissolved in dichloromethane and subjected to silica gel column chromatography to obtain 271 mg (41%) of the title compound as a colorless solid.
ESI-MS(m/e):649[M-H]-;1H NMR(300MHz,DMSO-d6):δ/ppm=8.527(m,3H),8.138(d,J=6.6Hz,1H),7.885(m,3H),7.369(s,5H),5.134(s,2H),4.452(m,4H),4.394(m,1H),3.924(m,2H),3.141(m,2H),1.691(m,1H),1.518(m,3H),1.398(s,9H)。ESI-MS (m/e): 649[MH] - ; 1 H NMR (300MHz, DMSO-d 6 ): δ/ppm=8.527(m, 3H), 8.138(d, J=6.6Hz, 1H), 7.885(m,3H),7.369(s,5H),5.134(s,2H),4.452(m,4H),4.394(m,1H),3.924(m,2H),3.141(m,2H),1.691 (m,1H), 1.518(m,3H), 1.398(s,9H).
实施例6制备1-[CH2CO-Arg(NO2)-Gly]-3-(CH2CO2tBu)-5-氟尿嘧啶(4)Example 6 Preparation of 1-[CH 2 CO-Arg(NO 2 )-Gly]-3-(CH 2 CO 2 tBu)-5-fluorouracil (4)
0℃将792mg(1.2mmol)1-[CH2CO-Arg(NO2)-Gly-OBzl]-3-(CH2CO2tBu)-5-氟尿嘧啶(3)用甲醇溶解,用氢氧化钠溶液(2M)调节反应液pH值至13。持续在0℃反应至TLC显示反应完全。反应液用2N盐酸溶液调节pH值至中性。减压浓缩除去甲醇。加入无水乙醇,超声震荡10min后,过滤,滤饼用无水乙醇洗涤3遍。减压浓缩除,得淡黄色油状标题化合物,直接用于后续反应。ESI-MS(m/e):559[M-H]-。Dissolve 792 mg (1.2 mmol) 1-[CH 2 CO-Arg(NO 2 )-Gly-OBzl]-3-(CH 2 CO 2 tBu)-5-fluorouracil (3) in methanol at 0°C, and dissolve it with sodium hydroxide solution (2M) to adjust the pH value of the reaction solution to 13. The reaction was continued at 0°C until TLC showed that the reaction was complete. The pH of the reaction solution was adjusted to neutral with 2N hydrochloric acid solution. Concentrate under reduced pressure to remove methanol. Add absolute ethanol, ultrasonically shake for 10 min, filter, and wash the
实施例7制备Boc-Asp(OBzl)-Ser-OBzlExample 7 Preparation of Boc-Asp(OBzl)-Ser-OBzl
采用实施例2的方法从3.32g(10mmol)Boc-Asp(OBzl)和2.78g(1.2mmol)Ser-OBzl得3.86g(77%)标题化合物,为淡黄色固体。ESI-MS(m/e):501[M+H]+。Using the method of Example 2, 3.86 g (77%) of the title compound was obtained from 3.32 g (10 mmol) Boc-Asp (OBzl) and 2.78 g (1.2 mmol) Ser-OBzl as a pale yellow solid. ESI-MS (m/e): 501 [M+H] + .
实施例8制备Asp(OBzl)-Ser-OBzlExample 8 Preparation of Asp(OBzl)-Ser-OBzl
采用实施例3的方法从2.50g(5mmol)Boc-Asp(OBzl)-Ser-OBzl得2.05g(94%)标题化合物,为淡黄色固体。ESI-MS(m/e):399[M-H]-。Using the method of Example 3, 2.05 g (94%) of the title compound was obtained from 2.50 g (5 mmol) of Boc-Asp(OBzl)-Ser-OBzl as a pale yellow solid. ESI-MS (m/e): 399 [MH] - .
实施例9制备1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2CO2tBu)-5-氟尿嘧啶(5)Example 9 Preparation of 1-[CH 2 CO-Arg(NO 2 )-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH 2 CO 2 tBu)-5-fluorouracil (5)
采用实施例2的方法从682mg(1.2mmol)1-[CH2CO-Arg(NO2)-Gly]-3-(CH2CO2tBu)-5-氟尿嘧啶(4)和532mg(1.2mmol)Asp(OBzl)-Ser-OBzl得到509mg(44%)标题化合物,为无色固体。ESI-MS(m/e):941[M-H]-。1H NMR(300MHz,DMSO-d6):δ/ppm=8.503(d,J=7.8Hz,1H),8.294(m,2H),8.160(d,J=6.6Hz,1H),7.357(m,10H),5.190(m,5H),4.758(m,1H),4.446(m,3H),4.378(m,2H),4.028(m,1H),3.718(m,4H),3.138(m,2H),2.729(m,1H),2.601(m,1H),1.687(m,2H),1.519(m,2H),1.392(m,9H)。Using the method of Example 2, from 682mg (1.2mmol) 1-[CH 2 CO-Arg(NO 2 )-Gly]-3-(CH 2 CO 2 tBu)-5-fluorouracil (4) and 532mg (1.2mmol) Asp(OBzl)-Ser-OBzl afforded 509 mg (44%) of the title compound as a colorless solid. ESI-MS (m/e): 941 [MH] - . 1 H NMR (300MHz, DMSO-d 6 ): δ/ppm=8.503(d, J=7.8Hz, 1H), 8.294(m, 2H), 8.160(d, J=6.6Hz, 1H), 7.357(m ,10H),5.190(m,5H),4.758(m,1H),4.446(m,3H),4.378(m,2H),4.028(m,1H),3.718(m,4H),3.138(m, 2H), 2.729(m, 1H), 2.601(m, 1H), 1.687(m, 2H), 1.519(m, 2H), 1.392(m, 9H).
实施例10制备1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2COOH)-5-氟尿嘧啶(6)Example 10 Preparation of 1-[CH 2 CO-Arg(NO 2 )-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH 2 COOH)-5-fluorouracil (6)
采用实施例3的方法从496mg(0.5mmol)1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2CO2tBu)-5-氟尿嘧啶(5)得450mg(98%)标题化合物,为淡黄色粉末。ESI-MS(m/e):885[M-H]-。Using the method of Example 3, from 496mg (0.5mmol) 1-[CH 2 CO-Arg(NO 2 )-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH 2 CO 2 tBu)-5-fluorouracil (5) Obtained 450 mg (98%) of the title compound as light yellow powder. ESI-MS (m/e): 885 [MH] - .
实施例11制备1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2CO-The-OBzl)-5-氟尿嘧啶(7)Example 11 Preparation of 1-[CH 2 CO-Arg(NO 2 )-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH 2 CO-The-OBzl)-5-fluorouracil (7)
采用实施例3的方法从485mg(0.5mmol)1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2COOH)-5-氟尿嘧啶(6)和206mg(1.5mmol)The-OBzl得106mg(17%)标题化合物,为无色固体。ESI-MS(m/e):1131[M-H]-;1H NMR(300MHz,DMSO-d6):δ/ppm=8.678(d,J=6.6Hz,1H),8.493(m,2H),8.276(m,3H),8.130(d,J=6.3Hz,1H),7.757(s,1H),7.361(s,15H),5.098(m,7H),4.792(m,1H),4.412(m,7H),3.745(m,4H),3.142(m,1H),3.306(t,J=7.2Hz,2H),2.738(m,1H),2.600(m,1H),2.137(t,J=7.2Hz,2H),1.914(m,1H),1.843(m,1H),1.693(m,1H),1.572(m,3H),0.983(t,J=7.2Hz,3H);13C NMR(75MHz,DMSO-d6):δ/ppm=171.9,1701.1,170.9,170.5,170.2,169.0,166.8,166.6,159.7,157.1,150.1,136.4,136.3,128.8,128.4,128.3,128.2,128.0,66.4,66.1,61.5,56.4,55.4,52.7,52.3,50.6,49.5,42.2,33.7,31.8,30.0,27.4,21.5,19.0,15.1。Using the method of Example 3, from 485 mg (0.5 mmol) 1-[CH 2 CO-Arg(NO 2 )-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH 2 COOH)-5-fluorouracil (6 ) and 206 mg (1.5 mmol) of The-OBzl yielded 106 mg (17%) of the title compound as a colorless solid. ESI-MS (m/e): 1131[MH] - ; 1 H NMR (300MHz, DMSO-d 6 ): δ/ppm=8.678(d, J=6.6Hz, 1H), 8.493(m, 2H), 8.276(m,3H),8.130(d,J=6.3Hz,1H),7.757(s,1H),7.361(s,15H),5.098(m,7H),4.792(m,1H),4.412(m ,7H),3.745(m,4H),3.142(m,1H),3.306(t,J=7.2Hz,2H),2.738(m,1H),2.600(m,1H),2.137(t,J= 7.2Hz, 2H), 1.914(m, 1H), 1.843(m, 1H), 1.693(m, 1H), 1.572(m, 3H), 0.983(t, J= 7.2Hz , 3H); 75MHz, DMSO-d 6 ): δ/ppm=171.9, 1701.1, 170.9, 170.5, 170.2, 169.0, 166.8, 166.6, 159.7, 157.1, 150.1, 136.4, 136.3, 128.8, 128.4, 128.3, 128.2, 1628, 66.1, 61.5, 56.4, 55.4, 52.7, 52.3, 50.6, 49.5, 42.2, 33.7, 31.8, 30.0, 27.4, 21.5, 19.0, 15.1.
实施例12制备1-(CH2CO-Arg-Gly-Asp-Ser)-3-(CH2CO-The)-5-氟尿嘧啶(8)Example 12 Preparation of 1-(CH 2 CO-Arg-Gly-Asp-Ser)-3-(CH 2 CO-The)-5-fluorouracil (8)
0℃将120mg(0.1mmol)1-[CH2CO-Arg(NO2)-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH2CO-The-OBzl)-5-氟尿嘧啶(7)溶于2mL三氟乙酸中,加入0.7mL三氟甲磺酸溶解,反应完全后加入预冷的无水乙醚,搅拌40min至有固体析出。离心,弃去上清液,反复三次,将离心所得固体晾干。加入H2O溶解,0℃下用饱和碳酸氢钠溶液调节pH值至7,过滤除去滤渣。滤液经Sephdax-G10凝胶柱层析及C18柱层析纯化,冷冻干燥后得到47mg(54%)标题化合物,为无色固体。Mp 180~181℃,(c=0.10,H2O);ESI-MS(m/e):816[M-H]-;1H NMR(300MHz,DMSO-d6):δ/ppm=9.384(s,1H),8.569(d,J=5.7Hz,1H),8.476(d,J=8.7Hz,1H),8.155(d,J=6.6Hz,1H),7.877(d,J=7.8Hz,1H),7.763(d,J=5.4Hz,1H),7.284(s,1H),4.441(m,5H),3.873(m,3H),3.634(s,4H),3.029(m,5H),2.052(m,2H),1.901(m,1H),1.725(m,2H),1.513(m,3H),0.984(t,J=7.2Hz,3H)。13C NMR(125MHz,DMSO-d6):δ/ppm=175.5,172.1,170.5,166.9,165.7,157.6,157.3,156.9,150.1,140.7,137.7,130.8,130.3,54.4,52.2,52.1,51.5,44.3,33.7,32.9,30.4,29.1,25.6,15.1。120 mg (0.1 mmol) 1-[CH 2 CO-Arg(NO 2 )-Gly-Asp(OBzl)-Ser-OBzl]-3-(CH 2 CO-The-OBzl)-5-fluorouracil (7 ) was dissolved in 2 mL of trifluoroacetic acid, and 0.7 mL of trifluoromethanesulfonic acid was added to dissolve it. After the reaction was complete, pre-cooled anhydrous diethyl ether was added, and stirred for 40 min until a solid precipitated. Centrifuge, discard the supernatant, repeat three times, and dry the solid obtained by centrifugation. Add H 2 O to dissolve, adjust the pH value to 7 with saturated sodium bicarbonate solution at 0°C, and filter to remove the filter residue. The filtrate was purified by Sephdax-G10 gel column chromatography and C18 column chromatography, and 47 mg (54%) of the title compound was obtained as a colorless solid after lyophilization. Mp 180~181℃, (c=0.10, H 2 O); ESI-MS (m/e): 816[MH] - ; 1 H NMR (300MHz, DMSO-d 6 ): δ/ppm=9.384 (s, 1H), 8.569 ( d,J=5.7Hz,1H),8.476(d,J=8.7Hz,1H),8.155(d,J=6.6Hz,1H),7.877(d,J=7.8Hz,1H),7.763(d, J=5.4Hz,1H),7.284(s,1H),4.441(m,5H),3.873(m,3H),3.634(s,4H),3.029(m,5H),2.052(m,2H), 1.901 (m, 1H), 1.725 (m, 2H), 1.513 (m, 3H), 0.984 (t, J=7.2Hz, 3H). 13 C NMR (125MHz, DMSO-d 6 ): δ/ppm=175.5, 172.1, 170.5, 166.9, 165.7, 157.6, 157.3, 156.9, 150.1, 140.7, 137.7, 130.8, 130.3, 54.4, 52.2, 52.1, 51.5, 44.3, 33.7, 32.9, 30.4, 29.1, 25.6, 15.1.
实施例13测定化合物8的抗肿瘤活性Example 13 Determination of the antitumor activity of
1)化合物8以及阳性对照5-FU用生理盐水溶解,生理盐水作为空白对照。1)
2)化合物8,5-FU和生理盐水均灌胃,化合物8的剂量为1nmol/kg/天,5-FU的剂量为150μmol/kg/天,生理盐水的给药量为0.1mL/10g/天。2)
3)实验动物为清洁级ICR雄性小鼠,体重为20±2g。3) The experimental animals are clean grade ICR male mice with a body weight of 20±2g.
4)移植性小鼠S180腹水型纤维肉瘤建模用的瘤源为S180小鼠腹水瘤细胞,购自北京大学医学部动物实验中心。取传代一周后,生长状态良好的ICR雄性小鼠(瘤源鼠),经麻醉后处死,无菌条件下取其腹腔中S 180瘤液,经1000rpm离心10min,弃去上清,残留物用少量4℃生理盐水洗涤,除去浮血和组织细胞碎片及其他非细胞成分。之后经台盼蓝染色,细胞计数并按公式计算活细胞浓度及细胞活力。4) The tumor source used for the modeling of transplanted mouse S180 ascites fibrosarcoma was S180 mouse ascites tumor cells, which were purchased from the Animal Experiment Center of Peking University Health Science Center. One week after subculture, ICR male mice (tumor-derived mice) in good growth state were taken and killed after anesthesia. Under sterile conditions, the S 180 tumor fluid in the peritoneal cavity was collected, centrifuged at 1000rpm for 10min, the supernatant was discarded, and the residue was used for Wash with a small amount of 4°C normal saline to remove floating blood, tissue cell debris and other non-cellular components. After trypan blue staining, the cells were counted and the concentration of viable cells and cell viability were calculated according to the formula.
将细胞活力大于90%的瘤液用4℃生理盐水稀释,制成2×107个/mL的细胞悬液。以0.2mL/只瘤液接种于ICR小鼠右侧腋下(尽快完成接种)。每天观察小鼠腋下肿瘤生长情况。按照瘤体积进行均匀分组,每组12只。分组后小鼠,按照上面描述的剂量每天口服化合物8或5-FU或生理盐水,连续给药8天。第9天将各组小鼠称重,麻醉,眼球取血于含EDTA的取血管中,进行血常规计数,观察受试化合物8对血常规细胞的影响。将小鼠处死,钝性分离小鼠腋下肿瘤组织,取出肉瘤后,称重。肿瘤重量以均值±SD g表示,用SPSS统计分析软件对实验数据进行统计学分析。结果见表1。可以看出,在1nmol/kg/天口服剂量下的化合物8能够有效抑制肿瘤的生长,其活性与剂量为150μmol/kg/天的5-氟尿嘧啶的活性无显著性差异(P>0.05)。可见,化合物8的抗肿瘤活性相当于5-FU的抗肿瘤活性的150000倍。本发明有显著的技术效果。The tumor fluid with cell viability greater than 90% was diluted with 4°C saline to make a cell suspension of 2×10 7 cells/mL. Inoculate the right axilla of ICR mice with 0.2 mL/mouse tumor fluid (complete the inoculation as soon as possible). The tumor growth in the axilla of the mice was observed every day. According to tumor volume, they were evenly divided into groups, with 12 rats in each group. After grouping, the mice were orally administered
表1化合物8的抗肿瘤活性Antitumor activity of
a)与生理盐水比P<0.01;b)与生理盐水比P<0.01,与5-FU比P>0.05;n=12.a) P<0.01 compared with normal saline; b) P<0.01 compared with normal saline, P>0.05 compared with 5-FU; n=12.
实施例14测定化合物8的抗肿瘤细胞迁移活性Example 14 Determination of anti-tumor cell migration activity of
1)化合物8用含0.1%DMSO的培养基配制成所需浓度。1)
2)肿瘤细胞为A549(人非小细胞肺癌细胞)及95D(人高转移非小细胞肺癌细胞)均选用RPMI-1640培养基培养,培养基中均含有10%经灭活的胎牛血清和1×105U/L青霉素和100mg/L链霉素。2) The tumor cells were A549 (human non-small cell lung cancer cells) and 95D (human highly metastatic non-small cell lung cancer cells) were cultured in RPMI-1640 medium, and the medium contained 10% inactivated fetal bovine serum and 1×10 5 U/L penicillin and 100 mg/L streptomycin.
3)将生长状态良好,处于对数生长期的A549细胞按照5×105个/mL的密度而95D细胞按照1×106个/mL的密度制备细胞悬液。使用无血清的培养基接种在Transwell的上室,每室加入100μL,加入化合物8(终浓度为20μM)。同时在下室加入600μL含有10%FBS的培养基,将Transwell小室放入24孔培养板中,于37℃的5%二氧化碳培养箱中培养48h,用棉签拭去上室的细胞,吸弃下室的培养基,用4%多聚甲醛固定液固定细胞0.5h,弃固定液,用PBS洗2次,用结晶紫染色10min,清水洗去浮色,用400倍显微镜观察。随机选取9个不同的视野观察细胞并计算迁移数。结果见表2。可以确认,在20μM浓度下化合物8有效地抑制肿瘤细胞迁移。此外,它们的活性与20μM浓度下Arg-Gly-Asp-Ser(RGDS)的活性无显著性差异。这是本发明的突出的技术效果。3) A549 cells in good growth state and in the logarithmic growth phase were prepared at a density of 5×10 5 cells/mL and 95D cells were prepared at a density of 1×10 6 cells/mL. Inoculate the upper chamber of the Transwell with serum-free medium, add 100 μL to each chamber, and add compound 8 (final concentration: 20 μM). At the same time, add 600 μL of medium containing 10% FBS to the lower chamber, put the Transwell chamber into a 24-well culture plate, and culture it in a 5% carbon dioxide incubator at 37°C for 48 hours, wipe off the cells in the upper chamber with a cotton swab, and discard the lower chamber The cells were fixed with 4% paraformaldehyde fixative solution for 0.5h, discarded the fixative solution, washed twice with PBS, stained with crystal violet for 10min, washed with water to remove the floating color, and observed under a microscope at 400 times. Randomly select 9 different fields of view to observe cells and calculate the number of migration. The results are shown in Table 2. It can be confirmed that
表2化合物8对A549和95D细胞迁移的影响Table 2 The effect of
a)与空白对照比P<0.01;b)与空白对照比P<0.01,与RGDS比P>0.05;n=3实施例15测定化合物8的抗肿瘤细胞侵袭活性a) P<0.01 compared with blank control; b) P<0.01 compared with blank control, P>0.05 compared with RGDS; n=3 Example 15 Determination of the anti-tumor cell invasion activity of
1)化合物8用含0.1%DMSO的培养基配制成所需浓度。1)
2)肿瘤细胞为A549(人非小细胞肺癌细胞)及95D(人高转移非小细胞肺癌细胞)均选用RPMI-1640培养基培养,培养基中均含有10%经灭活的胎牛血清和1×105U/L青霉素和100mg/L链霉素。2) The tumor cells were A549 (human non-small cell lung cancer cells) and 95D (human highly metastatic non-small cell lung cancer cells) were cultured in RPMI-1640 medium, and the medium contained 10% inactivated fetal bovine serum and 1×10 5 U/L penicillin and 100mg/L streptomycin.
3)将生长状态良好,处于对数生长期的A549细胞按照5×105个/mL的密度而95D细胞按照1×106个/mL的密度使用无血清的培养基接种在Transwell的上室,每室加入100μL,加入化合物8(终浓度为20μM)同时在下室加入600μL含有10%FBS的培养基,将Transwell小室放入24孔培养板中,37℃的5%二氧化碳培养箱中培养48h,用棉签拭去上室的细胞,弃下室的培养基,用4%多聚甲醛固定液固定细胞0.5h,弃固定液,用PBS洗2次,用结晶紫染色10min,清水洗去浮色,用400倍显微镜观察。随机选取9个不同的视野观察细胞并计算侵袭数。结果见表3,可以看到,在20μM浓度下化合物8有效地抑制肿瘤细胞侵袭。此外,它们的活性与20μM浓度下Arg-Gly-Asp-Ser(RGDS)的活性无显著性差异。这是本发明的突出的技术效果。3) A549 cells in good growth state and in the logarithmic growth phase were inoculated in the upper chamber of the Transwell at a density of 5×10 5 cells/mL and 95D cells at a density of 1×10 6 cells/mL with serum-free medium. , add 100 μL to each chamber, add compound 8 (final concentration 20 μM) and add 600 μL medium containing 10% FBS to the lower chamber, put the Transwell chamber into a 24-well culture plate, and incubate in a 5% carbon dioxide incubator at 37 °C for 48 h , wipe off the cells in the upper chamber with a cotton swab, discard the medium in the lower chamber, fix the cells with 4% paraformaldehyde fixative solution for 0.5h, discard the fixative solution, wash twice with PBS, stain with crystal violet for 10min, wash with water to remove the floating cells. Color, observed with a 400X microscope. Randomly select 9 different fields of view to observe cells and calculate the number of invasion. The results are shown in Table 3. It can be seen that
表3化合物8对A549和95D细胞侵袭的影响Table 3 The effect of
a)与空白对照比P<0.01;b)与空白对照比P<0.01,与RGDS比P>0.05;n=3.a) P<0.01 compared with blank control; b) P<0.01 compared with blank control, P>0.05 compared with RGDS; n=3.
实施例16测定化合物8的抗肿瘤转移活性Example 16 Determination of the anti-tumor metastasis activity of
将本化合物8溶于生理盐水。Lewis小鼠肺癌细胞(LLC,购自ATCC)用含有10%FBS和1×105U·L-1青霉素和100mg·L-1链霉素的DMEM培养基培养。每天传代一次,富集细胞。待细胞处于对数生长期且生长状态良好时消化细胞。用生理盐水调整细胞密度至2×107个/mL。This
体重为20±2g的近交系C57BL/6雄性小鼠用左手固定,用75%乙醇涂在小鼠右前肢腋窝皮肤处消毒,右手以1mL无菌注射器于消毒皮下处注射肿瘤细胞悬液,每只注射0.2mL,取接种10天生长状态良好的Lewis肺癌荷瘤小鼠,乙醚麻醉后颈椎脱臼处死。用75%乙醇浸泡消毒10min,在超净台上操作剥离瘤体,选择生长良好的肿瘤组织,在无菌培养皿中剪碎,放置于玻璃组织匀浆器中研磨。研磨时按照瘤块重量(g)/生理盐水体积(mL)为1/3的比例加4℃预冷的生理盐水。研磨制得的细胞悬液用200目的尼龙网过滤,收集的细胞用生理盐水调浓度为2×107个/mL。取体重为20±2g的近交系C57BL/6雄性小鼠,左手固定小鼠,用75%乙醇涂在小鼠右前肢腋窝皮肤处消毒,右手以1mL无菌注射器于消毒皮下处注射肿瘤细胞悬液,每只注射0.2mL,接种后10天可以长成绿豆大小的肿瘤。测量肿瘤体积,肿瘤直径为4-6mm的小鼠随机分组。化合物8组小鼠每天一次口服给药,剂量为0.1nmol/kg/天,连续给10天。Arg-Gly-Asp-Ser(RGDS,腹腔注射剂量为20μmol/kg/天,连续给10天)作阳性对照。空白组小鼠每日口服生理盐水,剂量为0.2mL/只/天,连续给10天。给药第11天称小鼠体重,乙醚麻醉,剖取各组小鼠的肺计算转移的瘤节数,并剖取各组小鼠的肿瘤称重。结果见表4。可以看到,化合物8有效地抑制肿瘤向肺转移。在0.1nmol/kg/天剂量下它抑制肿瘤向肺转移的活性与20μmol/kg/天剂量下RGDS的活性无显著性差异。可见,本发明有突出的技术效果。Inbred C57BL/6 male mice with a body weight of 20±2 g were fixed with the left hand, and 75% ethanol was applied to the skin of the axilla of the right forelimb of the mouse for disinfection, and the right hand was injected with a 1 mL sterile syringe into the subcutaneous place of the tumor cell suspension. Each mouse was injected with 0.2 mL, and Lewis lung cancer tumor-bearing mice with good growth status were taken 10 days after inoculation, and sacrificed by cervical dislocation after ether anesthesia. Soak and sterilize with 75% ethanol for 10min, operate and strip the tumor body on an ultra-clean bench, select a well-growing tumor tissue, cut it into pieces in a sterile petri dish, and place it in a glass tissue homogenizer for grinding. When grinding, add 4°C pre-cooled physiological saline according to the ratio of tumor mass (g)/normal saline volume (mL) to 1/3. The cell suspension prepared by grinding was filtered with a 200-mesh nylon mesh, and the collected cells were adjusted to a concentration of 2×10 7 cells/mL with normal saline. Take an inbred C57BL/6 male mouse with a body weight of 20±2 g, fix the mouse with the left hand, apply 75% ethanol to the skin of the right forelimb armpit of the mouse for disinfection, and inject tumor cells subcutaneously with a 1 mL sterile syringe in the right hand Suspension, each injected 0.2mL, 10 days after inoculation can grow into mung bean-sized tumors. Tumor volume was measured, and mice with tumor diameters of 4-6 mm were randomly divided into groups. Mice in
表4化合物8抑制肿瘤肺转移活性Table 4
a)与空白对照比P<0.01;b)与空白对照比P<0.01,与RGDS比P>0.05;n=10.a) P<0.01 compared with blank control; b) P<0.01 compared with blank control, P>0.05 compared with RGDS; n=10.
实施例17测定化合物8对S180小鼠的骨髓毒性Example 17 Determination of the bone marrow toxicity of
5-FU的骨髓抑制毒性主要表现在降低血液中白细胞和血小板计数。为了考察化合物8治疗的潜在骨髓毒性,本发明采用迈瑞全自动三分类血球分析仪BC3000测定了化合物8治疗的S180小鼠血液中白细胞和血小板计数。数据见表5。数据表明在1nmol/kg/天剂量下化合物8对S180小鼠血液中白细胞和血小板计数的影响与生理盐水没有差异。可见,化合物8治疗对S180小鼠没有骨髓毒性。相反,在150μmol/kg/天剂量下5-FU对S180小鼠血液中白细胞和血小板计数的影响与生理盐水有显著性差异。可见,5-FU对S180小鼠有骨髓毒性。在抗肿瘤活性与5-FU相同的前提下化合物8没有骨髓毒性,体现了本发明的突出技术效果。The bone marrow suppression toxicity of 5-FU is mainly manifested in the reduction of white blood cell and platelet counts in the blood. In order to investigate the potential bone marrow toxicity of
表5化合物8对S180小鼠白细胞和血小板数的影响The influence of table 5
a)与生理盐水比P<0.05;b)与生理盐水比P>0.05,n=8。a) P<0.05 compared with normal saline; b) P>0.05 compared with normal saline, n=8.
Claims (5)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910695564.4A CN112300245B (en) | 2019-07-30 | 2019-07-30 | RGDS and theanine co-modified 5-fluorouracil, and synthesis, activity and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN201910695564.4A CN112300245B (en) | 2019-07-30 | 2019-07-30 | RGDS and theanine co-modified 5-fluorouracil, and synthesis, activity and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN112300245A CN112300245A (en) | 2021-02-02 |
| CN112300245B true CN112300245B (en) | 2023-01-13 |
Family
ID=74485103
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN201910695564.4A Expired - Fee Related CN112300245B (en) | 2019-07-30 | 2019-07-30 | RGDS and theanine co-modified 5-fluorouracil, and synthesis, activity and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN112300245B (en) |
Family Cites Families (5)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| KR100746962B1 (en) * | 2006-04-04 | 2007-08-07 | 한국과학기술연구원 | Thermosensitive polyphosphazene-bioactive molecule conjugates, preparation method thereof and use thereof |
| CN103450199B (en) * | 2012-06-01 | 2015-09-09 | 首都医科大学 | Carbolinyl amino acid benzyl ester modified by theanine, its preparation, antitumor activity and application |
| CN105198961A (en) * | 2014-06-11 | 2015-12-30 | 首都医科大学 | RGD modified 5-fluorouracil and preparation method, nanostructure, activity and application thereof |
| CN109134596B (en) * | 2017-06-16 | 2021-10-26 | 首都医科大学 | Theanyl amino acid modified curcumin, and synthesis, activity and application thereof |
| CN109134598B (en) * | 2017-06-16 | 2021-10-26 | 首都医科大学 | Theanyl amino acid benzyl ester modified curcumin, and synthesis, activity and application thereof |
-
2019
- 2019-07-30 CN CN201910695564.4A patent/CN112300245B/en not_active Expired - Fee Related
Also Published As
| Publication number | Publication date |
|---|---|
| CN112300245A (en) | 2021-02-02 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN112300245B (en) | RGDS and theanine co-modified 5-fluorouracil, and synthesis, activity and application thereof | |
| CN112300244B (en) | 5-fluorouracil modified by theanine alone or together with RGDS, and synthesis, activity and application thereof | |
| CN112300246B (en) | 5-fluorouracil modified by aspartyl theanine RGDS, and synthesis, activity and application thereof | |
| CN112110987B (en) | 5-fluorouracil modified by asparaginyl theanine and phenylalanine, synthesis, activity and application thereof | |
| CN109912590B (en) | Amino acid and amino-n-hexanoic acid modified benzyl carboline carboxylate, its preparation, activity and application | |
| CN112390854B (en) | 5-Fluorouracil co-modified with theanine and RGDS, its synthesis, activity and application | |
| CN112010811B (en) | 5-fluorouracil modified by theanine and phenylalanine together, and synthesis, activity and application thereof | |
| CN112898379B (en) | Dioxane-modified tetrahydrocarboline-3-formyl-The-HGE, preparation thereof, antitumor activity thereof and application thereof | |
| CN109134595B (en) | Curcumin modified by theanyl amino acid benzyl ester, its synthesis, activity and application | |
| CN109912588B (en) | 6-amino amido n-hexanoyl carboline benzyl carboxylate, preparation, activity and application thereof | |
| CN111793114B (en) | Dihydroxymethyltetrahydro-carboline-3-formyl-The-HGE, its synthesis, activity and application | |
| CN111793115A (en) | Dimethyloltetrahydrocarboline-3-formyl-The-EDG, its synthesis, activity and applications | |
| CN112979750B (en) | Dioxane-modified tetrahydrocarboline-3-formyl-The-EDG, its preparation, anti-metastatic activity and application | |
| CN111793113B (en) | Dihydroxymethyltetrahydrocarboline-3-formyl-The-HGK, its synthesis, activity and application | |
| CN112979752B (en) | Dioxane-modified tetrahydrocarboline-3-formyl-The-EDG, preparation thereof, antitumor activity thereof and application thereof | |
| CN112898376B (en) | Dioxane-modified tetrahydrocarboline-3-formyl-The-HGK, preparation thereof, antitumor activity thereof and application thereof | |
| CN110577568B (en) | Methyl indole and amide side chain amino acid modified diketopiperazine, synthesis, activity and application thereof | |
| CN110551121A (en) | Glutamine amido n-hexyl carboline carboxylic acid benzyl ester, preparation, activity and application thereof | |
| CN108976204B (en) | 3S-indolylmethyl-6R-aspartic acid modified piperazine-2, 5-dione, and synthesis, activity and application thereof | |
| CN110577518B (en) | Methyl indole and amide side chain amino acid modified diketopiperazine, synthesis and application thereof | |
| CN108976208B (en) | 3S-indolylmethyl-6R-Met-modified piperazine-2,5-diones, their synthesis, activities and applications | |
| CN108929320A (en) | Piperazine -2,5- diketone of 3R- indole methyl -6R- oxazolidone modification, synthesis, activity and application | |
| CN108976211A (en) | Piperazine -2,5- diketone of 3S- indole methyl -6R- fatty acid/amino acid modification, synthesis, activity and application | |
| CN108929313A (en) | Piperazine -2,5- diketone of 3R- indole methyl -6R- fatty acid/amino acid modification, synthesis, activity and application | |
| CN108929369A (en) | Piperazine -2,5- diketone of 3R- indole methyl -6R- acidic amino acid modification, synthesis, activity and application |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| PB01 | Publication | ||
| PB01 | Publication | ||
| SE01 | Entry into force of request for substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| GR01 | Patent grant | ||
| GR01 | Patent grant | ||
| CF01 | Termination of patent right due to non-payment of annual fee | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20230113 |