CN102031235B - Enterococcus faecium ANSE228 and application thereof - Google Patents
Enterococcus faecium ANSE228 and application thereof Download PDFInfo
- Publication number
- CN102031235B CN102031235B CN2010105399880A CN201010539988A CN102031235B CN 102031235 B CN102031235 B CN 102031235B CN 2010105399880 A CN2010105399880 A CN 2010105399880A CN 201010539988 A CN201010539988 A CN 201010539988A CN 102031235 B CN102031235 B CN 102031235B
- Authority
- CN
- China
- Prior art keywords
- anse228
- enterococcus faecalis
- enterococcus faecium
- enterococcus
- feed
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Active
Links
- 241000194031 Enterococcus faecium Species 0.000 title claims abstract description 16
- 241001465754 Metazoa Species 0.000 claims abstract description 18
- 239000003651 drinking water Substances 0.000 claims abstract description 13
- 235000020188 drinking water Nutrition 0.000 claims abstract description 13
- 238000000034 method Methods 0.000 claims abstract description 11
- 238000009395 breeding Methods 0.000 claims abstract description 3
- 230000001488 breeding effect Effects 0.000 claims abstract description 3
- 241000194032 Enterococcus faecalis Species 0.000 claims description 63
- 229940032049 enterococcus faecalis Drugs 0.000 claims description 63
- 238000000855 fermentation Methods 0.000 claims description 30
- 230000004151 fermentation Effects 0.000 claims description 30
- 239000007788 liquid Substances 0.000 claims description 20
- 239000006041 probiotic Substances 0.000 claims description 19
- 235000018291 probiotics Nutrition 0.000 claims description 19
- 241000287828 Gallus gallus Species 0.000 claims description 13
- 239000001963 growth medium Substances 0.000 claims description 11
- XLYOFNOQVPJJNP-UHFFFAOYSA-N water Chemical compound O XLYOFNOQVPJJNP-UHFFFAOYSA-N 0.000 claims description 11
- 239000002609 medium Substances 0.000 claims description 10
- WQZGKKKJIJFFOK-GASJEMHNSA-N Glucose Natural products OC[C@H]1OC(O)[C@H](O)[C@@H](O)[C@@H]1O WQZGKKKJIJFFOK-GASJEMHNSA-N 0.000 claims description 6
- 239000012153 distilled water Substances 0.000 claims description 6
- 239000008103 glucose Substances 0.000 claims description 6
- 230000003068 static effect Effects 0.000 claims description 6
- 238000004321 preservation Methods 0.000 claims description 5
- 235000019764 Soybean Meal Nutrition 0.000 claims description 4
- 240000008042 Zea mays Species 0.000 claims description 4
- 235000005824 Zea mays ssp. parviglumis Nutrition 0.000 claims description 4
- 235000002017 Zea mays subsp mays Nutrition 0.000 claims description 4
- 239000000654 additive Substances 0.000 claims description 4
- 235000005822 corn Nutrition 0.000 claims description 4
- ZPWVASYFFYYZEW-UHFFFAOYSA-L dipotassium hydrogen phosphate Chemical compound [K+].[K+].OP([O-])([O-])=O ZPWVASYFFYYZEW-UHFFFAOYSA-L 0.000 claims description 4
- 235000013312 flour Nutrition 0.000 claims description 4
- ISPYRSDWRDQNSW-UHFFFAOYSA-L manganese(II) sulfate monohydrate Chemical compound O.[Mn+2].[O-]S([O-])(=O)=O ISPYRSDWRDQNSW-UHFFFAOYSA-L 0.000 claims description 4
- 229910000402 monopotassium phosphate Inorganic materials 0.000 claims description 4
- 235000019796 monopotassium phosphate Nutrition 0.000 claims description 4
- PJNZPQUBCPKICU-UHFFFAOYSA-N phosphoric acid;potassium Chemical compound [K].OP(O)(O)=O PJNZPQUBCPKICU-UHFFFAOYSA-N 0.000 claims description 4
- 239000004455 soybean meal Substances 0.000 claims description 4
- 241000282887 Suidae Species 0.000 claims description 3
- 238000012364 cultivation method Methods 0.000 claims description 3
- WRUGWIBCXHJTDG-UHFFFAOYSA-L magnesium sulfate heptahydrate Chemical compound O.O.O.O.O.O.O.[Mg+2].[O-]S([O-])(=O)=O WRUGWIBCXHJTDG-UHFFFAOYSA-L 0.000 claims description 3
- 229940061634 magnesium sulfate heptahydrate Drugs 0.000 claims description 3
- 241000283690 Bos taurus Species 0.000 claims description 2
- 241001494479 Pecora Species 0.000 claims description 2
- 230000000996 additive effect Effects 0.000 claims description 2
- 241000272525 Anas platyrhynchos Species 0.000 claims 1
- 230000000529 probiotic effect Effects 0.000 abstract description 10
- 230000005764 inhibitory process Effects 0.000 abstract description 8
- 241000588724 Escherichia coli Species 0.000 abstract description 7
- 230000000694 effects Effects 0.000 abstract description 7
- 241000607683 Salmonella enterica subsp. enterica serovar Pullorum Species 0.000 abstract description 6
- 241000191967 Staphylococcus aureus Species 0.000 abstract description 6
- 230000003115 biocidal effect Effects 0.000 abstract description 5
- 230000000968 intestinal effect Effects 0.000 abstract description 4
- 230000008569 process Effects 0.000 abstract description 4
- 230000009286 beneficial effect Effects 0.000 abstract description 3
- 230000004071 biological effect Effects 0.000 abstract description 3
- 239000003795 chemical substances by application Substances 0.000 abstract description 3
- 239000003814 drug Substances 0.000 abstract description 3
- 238000000746 purification Methods 0.000 abstract description 3
- 230000003716 rejuvenation Effects 0.000 abstract description 3
- 238000000926 separation method Methods 0.000 abstract description 2
- 241000894006 Bacteria Species 0.000 description 25
- 239000000243 solution Substances 0.000 description 15
- 238000012360 testing method Methods 0.000 description 14
- HEMHJVSKTPXQMS-UHFFFAOYSA-M Sodium hydroxide Chemical compound [OH-].[Na+] HEMHJVSKTPXQMS-UHFFFAOYSA-M 0.000 description 12
- 239000003833 bile salt Substances 0.000 description 11
- 238000004519 manufacturing process Methods 0.000 description 10
- 238000011282 treatment Methods 0.000 description 10
- 230000001580 bacterial effect Effects 0.000 description 9
- 235000013305 food Nutrition 0.000 description 8
- 238000002474 experimental method Methods 0.000 description 7
- FAPWRFPIFSIZLT-UHFFFAOYSA-M Sodium chloride Chemical compound [Na+].[Cl-] FAPWRFPIFSIZLT-UHFFFAOYSA-M 0.000 description 6
- 210000004051 gastric juice Anatomy 0.000 description 6
- 230000004083 survival effect Effects 0.000 description 6
- 244000052616 bacterial pathogen Species 0.000 description 5
- 230000008901 benefit Effects 0.000 description 5
- 238000010790 dilution Methods 0.000 description 5
- 239000012895 dilution Substances 0.000 description 5
- VEXZGXHMUGYJMC-UHFFFAOYSA-N Hydrochloric acid Chemical compound Cl VEXZGXHMUGYJMC-UHFFFAOYSA-N 0.000 description 4
- 239000003242 anti bacterial agent Substances 0.000 description 4
- 229940088710 antibiotic agent Drugs 0.000 description 4
- 229940093761 bile salts Drugs 0.000 description 4
- 235000013330 chicken meat Nutrition 0.000 description 4
- 210000001035 gastrointestinal tract Anatomy 0.000 description 4
- 238000002360 preparation method Methods 0.000 description 4
- 239000000725 suspension Substances 0.000 description 4
- 229920001817 Agar Polymers 0.000 description 3
- 239000008272 agar Substances 0.000 description 3
- 239000003674 animal food additive Substances 0.000 description 3
- 230000000844 anti-bacterial effect Effects 0.000 description 3
- 238000011161 development Methods 0.000 description 3
- 210000004211 gastric acid Anatomy 0.000 description 3
- 238000010438 heat treatment Methods 0.000 description 3
- 238000002649 immunization Methods 0.000 description 3
- 230000003053 immunization Effects 0.000 description 3
- 239000000203 mixture Substances 0.000 description 3
- 238000011056 performance test Methods 0.000 description 3
- 238000013207 serial dilution Methods 0.000 description 3
- 238000003756 stirring Methods 0.000 description 3
- 108010019160 Pancreatin Proteins 0.000 description 2
- 102000057297 Pepsin A Human genes 0.000 description 2
- 108090000284 Pepsin A Proteins 0.000 description 2
- 239000001888 Peptone Substances 0.000 description 2
- 108010080698 Peptones Proteins 0.000 description 2
- 241000607142 Salmonella Species 0.000 description 2
- VMHLLURERBWHNL-UHFFFAOYSA-M Sodium acetate Chemical compound [Na+].CC([O-])=O VMHLLURERBWHNL-UHFFFAOYSA-M 0.000 description 2
- 235000015278 beef Nutrition 0.000 description 2
- 230000037396 body weight Effects 0.000 description 2
- 229940041514 candida albicans extract Drugs 0.000 description 2
- KLOIYEQEVSIOOO-UHFFFAOYSA-N carbocromen Chemical compound CC1=C(CCN(CC)CC)C(=O)OC2=CC(OCC(=O)OCC)=CC=C21 KLOIYEQEVSIOOO-UHFFFAOYSA-N 0.000 description 2
- 238000006243 chemical reaction Methods 0.000 description 2
- 229940079593 drug Drugs 0.000 description 2
- 239000000284 extract Substances 0.000 description 2
- 230000036541 health Effects 0.000 description 2
- 239000002054 inoculum Substances 0.000 description 2
- 239000012528 membrane Substances 0.000 description 2
- 238000001471 micro-filtration Methods 0.000 description 2
- 238000012986 modification Methods 0.000 description 2
- 230000004048 modification Effects 0.000 description 2
- 239000006872 mrs medium Substances 0.000 description 2
- 229940055695 pancreatin Drugs 0.000 description 2
- 229940111202 pepsin Drugs 0.000 description 2
- 235000019319 peptone Nutrition 0.000 description 2
- 230000001737 promoting effect Effects 0.000 description 2
- 238000011160 research Methods 0.000 description 2
- 239000001632 sodium acetate Substances 0.000 description 2
- 235000017281 sodium acetate Nutrition 0.000 description 2
- 238000012795 verification Methods 0.000 description 2
- 239000012138 yeast extract Substances 0.000 description 2
- 241000251468 Actinopterygii Species 0.000 description 1
- 241000272517 Anseriformes Species 0.000 description 1
- 108010001478 Bacitracin Proteins 0.000 description 1
- 206010012735 Diarrhoea Diseases 0.000 description 1
- 208000035240 Disease Resistance Diseases 0.000 description 1
- 241000282412 Homo Species 0.000 description 1
- 241000282849 Ruminantia Species 0.000 description 1
- QBRPIXOGXMPIMA-UHFFFAOYSA-N [Mg].S(O)(O)(=O)=O.O.O.O.O.O.O.O Chemical compound [Mg].S(O)(O)(=O)=O.O.O.O.O.O.O.O QBRPIXOGXMPIMA-UHFFFAOYSA-N 0.000 description 1
- 238000010521 absorption reaction Methods 0.000 description 1
- 230000002411 adverse Effects 0.000 description 1
- 229960005364 bacitracin zinc Drugs 0.000 description 1
- 235000014590 basal diet Nutrition 0.000 description 1
- CYDMQBQPVICBEU-UHFFFAOYSA-N chlorotetracycline Natural products C1=CC(Cl)=C2C(O)(C)C3CC4C(N(C)C)C(O)=C(C(N)=O)C(=O)C4(O)C(O)=C3C(=O)C2=C1O CYDMQBQPVICBEU-UHFFFAOYSA-N 0.000 description 1
- 229960004475 chlortetracycline Drugs 0.000 description 1
- CYDMQBQPVICBEU-XRNKAMNCSA-N chlortetracycline Chemical compound C1=CC(Cl)=C2[C@](O)(C)[C@H]3C[C@H]4[C@H](N(C)C)C(O)=C(C(N)=O)C(=O)[C@@]4(O)C(O)=C3C(=O)C2=C1O CYDMQBQPVICBEU-XRNKAMNCSA-N 0.000 description 1
- 235000019365 chlortetracycline Nutrition 0.000 description 1
- 238000013461 design Methods 0.000 description 1
- 239000003085 diluting agent Substances 0.000 description 1
- 201000010099 disease Diseases 0.000 description 1
- 208000037265 diseases, disorders, signs and symptoms Diseases 0.000 description 1
- 230000000816 effect on animals Effects 0.000 description 1
- 230000007613 environmental effect Effects 0.000 description 1
- 230000015784 hyperosmotic salinity response Effects 0.000 description 1
- 238000011534 incubation Methods 0.000 description 1
- 230000002401 inhibitory effect Effects 0.000 description 1
- 238000002955 isolation Methods 0.000 description 1
- 238000009630 liquid culture Methods 0.000 description 1
- 244000144972 livestock Species 0.000 description 1
- 230000007774 longterm Effects 0.000 description 1
- 244000005700 microbiome Species 0.000 description 1
- 239000013642 negative control Substances 0.000 description 1
- 235000015097 nutrients Nutrition 0.000 description 1
- 239000002504 physiological saline solution Substances 0.000 description 1
- 239000013641 positive control Substances 0.000 description 1
- 244000144977 poultry Species 0.000 description 1
- 235000013594 poultry meat Nutrition 0.000 description 1
- 238000012545 processing Methods 0.000 description 1
- 230000002829 reductive effect Effects 0.000 description 1
- 239000011780 sodium chloride Substances 0.000 description 1
- 229910001220 stainless steel Inorganic materials 0.000 description 1
- 239000010935 stainless steel Substances 0.000 description 1
- 238000006467 substitution reaction Methods 0.000 description 1
- 238000010998 test method Methods 0.000 description 1
- UCRLQOPRDMGYOA-DFTDUNEMSA-L zinc;(4r)-4-[[(2s)-2-[[(4r)-2-[(1s,2s)-1-amino-2-methylbutyl]-4,5-dihydro-1,3-thiazole-4-carbonyl]amino]-4-methylpentanoyl]amino]-5-[[(2s,3s)-1-[[(3s,6r,9s,12r,15s,18r,21s)-3-(2-amino-2-oxoethyl)-18-(3-aminopropyl)-12-benzyl-15-[(2s)-butan-2-yl]-6-(carbox Chemical compound [Zn+2].C1SC([C@@H](N)[C@@H](C)CC)=N[C@@H]1C(=O)N[C@@H](CC(C)C)C(=O)N[C@H](CCC([O-])=O)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@@H]1C(=O)N[C@H](CCCN)C(=O)N[C@@H]([C@@H](C)CC)C(=O)N[C@H](CC=2C=CC=CC=2)C(=O)N[C@@H](CC=2NC=NC=2)C(=O)N[C@H](CC([O-])=O)C(=O)N[C@@H](CC(N)=O)C(=O)NCCCC1 UCRLQOPRDMGYOA-DFTDUNEMSA-L 0.000 description 1
Images
Classifications
-
- Y—GENERAL TAGGING OF NEW TECHNOLOGICAL DEVELOPMENTS; GENERAL TAGGING OF CROSS-SECTIONAL TECHNOLOGIES SPANNING OVER SEVERAL SECTIONS OF THE IPC; TECHNICAL SUBJECTS COVERED BY FORMER USPC CROSS-REFERENCE ART COLLECTIONS [XRACs] AND DIGESTS
- Y02—TECHNOLOGIES OR APPLICATIONS FOR MITIGATION OR ADAPTATION AGAINST CLIMATE CHANGE
- Y02A—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE
- Y02A50/00—TECHNOLOGIES FOR ADAPTATION TO CLIMATE CHANGE in human health protection, e.g. against extreme weather
- Y02A50/30—Against vector-borne diseases, e.g. mosquito-borne, fly-borne, tick-borne or waterborne diseases whose impact is exacerbated by climate change
Landscapes
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
Abstract
Description
技术领域 technical field
本发明涉及一种粪肠球菌及其应用。 The invention relates to an enterococcus faecalis and its application. the
背景技术 Background technique
随着社会和科学的发展,食品安全性日益受到人们的关注,因为不安全食品不仅危害人体健康和生命,甚至还可能对子孙后代产生不利影响。社会呼吁绿色、安全的食品,而饲料的安全就是食品的安全。 With the development of society and science, people pay more and more attention to food safety, because unsafe food not only endangers human health and life, but may even have adverse effects on future generations. The society calls for green and safe food, and the safety of feed is the safety of food. the
抗生素作为饲料添加剂,对于促进动物生长发育、提高疾病抵抗力等方面具有重要作用。但其长时间的大量使用导致了一些负面效应的产生,譬如破坏了动物自身的微生态平衡,增加了畜禽消化道疾病发生的几率,并能使病原菌突变为耐药菌株等。更为重要的是,抗生素在动物体内的残留,通过食物链传给人类,给人们健康带来隐患。鉴于此,很多国家都在饲料中禁用和限用抗生素作为饲料添加剂。因此,抗生素替代产品的开发也成了世界各国研究的重点。其中微生态制剂由于其独特的优势,如广谱的非特异性抑菌和杀菌作用、绿色环保、无残留等,在使用抗生素替代领域正得到越来越多的重视。 Antibiotics, as feed additives, play an important role in promoting animal growth and development, and improving disease resistance. However, its long-term and large-scale use has led to some negative effects, such as destroying the micro-ecological balance of animals, increasing the incidence of digestive tract diseases in livestock and poultry, and causing pathogenic bacteria to mutate into drug-resistant strains. More importantly, the residues of antibiotics in animals are transmitted to humans through the food chain, bringing hidden dangers to human health. In view of this, many countries have prohibited and restricted the use of antibiotics as feed additives in feed. Therefore, the development of alternative products to antibiotics has become the focus of research in countries all over the world. Among them, due to their unique advantages, such as broad-spectrum non-specific antibacterial and bactericidal effects, green environmental protection, and no residue, etc., more and more attention is being paid to the field of antibiotic substitution. the
本研究以粪肠球菌(Enterococcus faecium)作为研究对象,整合了现有的对益生粪肠球菌的理论和实践认识,筛选出具有抗菌活性、抗逆性以及安全性的益生粪肠球菌,优化该菌的发酵工艺并,研究其作为微生态制剂对动物生产性能的影响,为益生粪肠球菌的应用和实践奠定基础。 This study takes Enterococcus faecium as the research object, integrates the existing theoretical and practical understanding of probiotic Enterococcus faecium, screens out the probiotic Enterococcus faecium with antibacterial activity, stress resistance and safety, and optimizes the probiotic Enterococcus faecium. The fermentation process of bacteria and its effect on animal production performance as a microecological agent are studied, laying the foundation for the application and practice of probiotic Enterococcus faecalis. the
发明内容 Contents of the invention
本发明的目的在于提供一种粪肠球菌。 The object of the present invention is to provide a kind of enterococcus faecalis. the
本发明的另一目的在于提供上述粪肠球菌的应用。 Another object of the present invention is to provide the application of the above Enterococcus faecalis. the
本发明的目的还在于提供一种微生态制剂。 The object of the present invention is also to provide a probiotics. the
本发明的再一目的在于提供上述微生态制剂的应用。 Another object of the present invention is to provide the application of the above-mentioned probiotics. the
为了实现本发明的目的,本发明提供一种粪肠球菌(Enterococcusfaecium)ANSE228,其保藏编号为CGMCC No.4082,保藏单位:中国微生物菌种保藏管理委员会普通微生物中心(CGMCC),地址:北京市朝阳区北辰西路1号院3号,保藏日期:2010年8月16日。所述粪肠球菌ANSE228是通过反复分离、纯化、复壮等工艺得到,具有生物活性强、益生性显著并且抗逆性能好等优点。 In order to realize the purpose of the present invention, the present invention provides a kind of Enterococcus faecium (Enterococcusfaecium) ANSE228, and its preservation number is CGMCC No.4082, depository unit: China Microorganism Culture Preservation Management Committee Common Microbiology Center (CGMCC), address: Beijing Municipality No. 3, Courtyard No. 1, Beichen West Road, Chaoyang District, date of preservation: August 16, 2010. The Enterococcus faecalis ANSE228 is obtained through repeated isolation, purification, rejuvenation and other processes, and has the advantages of strong biological activity, remarkable probiotics and good stress resistance. the
本发明中所述粪肠球菌ANSE228的培养方法如下:取粪肠球菌ANSE228种子液1-5ml(种子液是挑取的单个菌落在种子液培养基中培养的,其活菌浓度以109CFU/ml计;种子液的培养基和培养条件与发酵的培养基和培养条件相同),接种到50-100ml培养基中进行摇瓶发酵培养。所述培养基由如下组分组成:玉米粉10-25g/L、豆粕10-30g/L、葡萄糖0-15g/L、磷酸氢二钾0.1-0.2g、磷酸二氢钾1.0-2.0g、一水硫酸锰0.5-2.0g、七水硫酸镁0.5-2.0g、蒸馏水800-1200mL,pH值为6.5-7.5。 The cultivation method of Enterococcus faecalis ANSE228 described in the present invention is as follows: get Enterococcus faecalis ANSE228 seed liquid 1-5ml (seed liquid is that the single bacterium colony of picking is cultivated in seed liquid culture medium, and its live bacteria concentration is 10 9 CFU /ml meter; the medium and culture conditions of the seed liquid are the same as the culture medium and culture conditions of fermentation), inoculated into 50-100ml culture medium to carry out shake flask fermentation culture. The culture medium is composed of the following components: corn flour 10-25g/L, soybean meal 10-30g/L, glucose 0-15g/L, dipotassium hydrogen phosphate 0.1-0.2g, potassium dihydrogen phosphate 1.0-2.0g, Manganese sulfate monohydrate 0.5-2.0g, magnesium sulfate heptahydrate 0.5-2.0g, distilled water 800-1200mL, pH value 6.5-7.5.
其中,优选为:玉米粉15g,豆粕20g,葡萄糖5g、磷酸氢二钾0.15g、磷酸二氢钾1.5g、一水硫酸锰1.0g、七水硫酸镁1.5g、蒸馏水1000mL,pH值为7.0。 Among them, the preferred ones are: 15g corn flour, 20g soybean meal, 5g glucose, 0.15g dipotassium hydrogen phosphate, 1.5g potassium dihydrogen phosphate, 1.0g manganese sulfate monohydrate, 1.5g magnesium sulfate heptahydrate, 1000mL distilled water, pH 7.0 . the
本发明中所述摇瓶发酵的条件为:发酵温度25-45℃,发酵时间18-36h,pH值6.5-7.5,静置培养;优选为:发酵温度37℃、发酵时间24h、pH值7.0。 The conditions for shake flask fermentation in the present invention are: fermentation temperature 25-45°C, fermentation time 18-36h, pH value 6.5-7.5, static culture; preferably: fermentation temperature 37°C, fermentation time 24h, pH value 7.0 . the
10L发酵罐中试培养基与摇瓶发酵培养基相同。10L发酵罐中试发酵条件为:装液量为4-7L培养基,接种量为30-100ml摇瓶种子液,发酵温度为25-45℃,发酵时间18-36h,pH值6.5-7.5,搅拌转速200-400r/min。其中,优选为:装液量为5L培养基,接种量为50ml摇瓶种子液发酵温度为37℃,发酵时间24h,pH值7.0,搅拌转速300r/min。 The 10L fermenter pilot test medium is the same as the shake flask fermentation medium. The pilot test fermentation conditions of 10L fermenter are: liquid volume is 4-7L medium, inoculum volume is 30-100ml shake flask seed liquid, fermentation temperature is 25-45℃, fermentation time is 18-36h, pH value is 6.5-7.5, The stirring speed is 200-400r/min. Among them, it is preferred that the liquid volume is 5L of culture medium, the inoculum volume is 50ml, the shake flask seed liquid fermentation temperature is 37°C, the fermentation time is 24h, the pH value is 7.0, and the stirring speed is 300r/min. the
本发明还提供粪肠球菌ANSE228在抑制鸡白痢沙门氏菌、大肠杆菌和/或金黄色葡萄球菌中的应用。所述菌株具有显著的益生性,可显著抑制鸡白痢沙门氏菌、大肠杆菌以及金黄色葡萄球菌等致病菌的生长繁殖。 The present invention also provides the application of Enterococcus faecalis ANSE228 in inhibiting Salmonella pullorum, Escherichia coli and/or Staphylococcus aureus. The bacterial strain has significant probiotic properties and can significantly inhibit the growth and reproduction of pathogenic bacteria such as Salmonella pullorum, Escherichia coli and Staphylococcus aureus. the
本发明还提供所述粪肠球菌ANSE228在模拟胃液和/或模拟胆盐和/或 75-90℃的高温环境中的应用。所述菌株具有较强的抗逆性,可以耐受模拟胃酸、模拟胆盐以及高温环境,并且能保持80%以上较高的活菌存活率。 The present invention also provides the application of the Enterococcus faecalis ANSE228 in simulated gastric juice and/or simulated bile salt and/or high temperature environment of 75-90°C. The bacterial strain has strong stress resistance, can tolerate simulated gastric acid, simulated bile salt and high temperature environment, and can maintain a high survival rate of more than 80% of live bacteria. the
本发明粪肠球菌ANSE228的益生性的检验方法如下: The probiotic testing method of Enterococcus faecalis ANSE228 of the present invention is as follows:
在无菌操作台上,制备厚度为4mm左右的MRS平板,将致病菌(大肠杆菌、金黄色葡萄球菌及沙门氏菌)浓度为106的菌悬液100-200μL涂布于该平板上,将灭菌的不锈钢小管(内径6mm、外径8mm、高10mm的圆形小管,管的两端要光滑)放置在培养基上,轻轻加压,使其与培养基接触无空隙,待数分钟后,分别向各小管中滴加一定数量的粪肠球菌ANSE228活化菌种菌液,勿使其外溢,35℃-40℃培养18-36h,然后测量抑菌圈直径。每个实验三个重复,取平均值。 Prepare an MRS plate with a thickness of about 4 mm on a sterile operating table, and spread 100-200 μL of a bacterial suspension of pathogenic bacteria (E. coli, Staphylococcus aureus, and Salmonella) at a concentration of 10 6 on the plate, and Place a sterilized stainless steel small tube (circular small tube with an inner diameter of 6 mm, an outer diameter of 8 mm, and a height of 10 mm, with both ends smooth) on the culture medium, pressurize gently to make it contact with the culture medium without gaps, and wait for several minutes Finally, drop a certain amount of Enterococcus faecalis ANSE228 activated strain bacteria liquid into each small tube respectively to prevent it from overflowing, incubate at 35°C-40°C for 18-36h, and then measure the diameter of the inhibition zone. Each experiment was repeated three times, and the average value was taken.
其中,所述MRS平板是将121℃高温灭菌后的MRS培养基,倒入灭菌后的平板,冷却后形成表面光滑的4mm厚的MRS平板。所述MRS培养基的配方为:蛋白胨10g、牛肉膏10g、酵母浸膏5g、K2HPO42g、柠檬酸氢二铵2g、葡萄糖20g、乙酸钠5g、Tween-801mL、MgSO4·7H2O0.58g、MnSO4·4H2O 0.25g、琼脂16g、蒸馏水1000ml,pH=7.0。 Wherein, the MRS plate is the MRS culture medium sterilized at 121° C., poured into the sterilized plate, and cooled to form a 4 mm thick MRS plate with a smooth surface. The formula of the MRS medium is: peptone 10g, beef extract 10g, yeast extract 5g, K 2 HPO 4 2g, diammonium hydrogen citrate 2g, glucose 20g, sodium acetate 5g, Tween-801mL, MgSO 4 7H 2 O0.58g, MnSO 4 ·4H 2 O 0.25g, agar 16g, distilled water 1000ml, pH=7.0.
本发明粪肠球菌抗逆性的检验方法如下: The test method of Enterococcus faecalis resistance of the present invention is as follows:
1、高温耐受试验:取5mL粪肠球菌ANSE228活菌菌液注入到离心管1中,用无菌生理盐水十倍逐级稀释,选择合适稀释度的稀释液,在MRS平板1上涂布。另取5mL粪肠球菌ANSE228活菌菌液注入到离心管2中,置于75-90℃水浴锅中加热10-20min,取加热后的粪肠球菌ANSE228活菌菌液用无菌生理盐水进行十倍逐级稀释,选择与前述相同稀释度的稀释液,在MRS平板2上涂布。将平板1和平板2在30-45℃条件下培养18-36h,计算粪肠球菌加热前后的数量。 1. High temperature tolerance test: Take 5mL Enterococcus faecalis ANSE228 live bacteria liquid and inject it into the centrifuge tube 1, dilute it step by step with sterile normal saline ten times, choose the appropriate diluent, and spread it on the MRS plate 1 . Another 5mL live Enterococcus faecalis ANSE228 bacteria solution was injected into centrifuge tube 2, placed in a 75-90°C water bath and heated for 10-20min, and the heated Enterococcus faecalis ANSE228 live bacteria solution was treated with sterile saline Ten-fold serial dilutions were made, and the dilutions with the same dilution as above were selected and coated on MRS plate 2. Plate 1 and plate 2 were cultured at 30-45° C. for 18-36 hours, and the number of Enterococcus faecalis before and after heating was calculated. the
2、模拟胃液的耐受试验:将胃蛋白酶溶解在0.5%-0.85%生理盐水中,使其终浓度为3g/L,并用浓盐酸或者10%的NaOH调整pH值到2.0-4.0。取0.5mL粪肠球菌ANSE228菌悬液加入到4.5mL的模拟胃液中(即十倍逐级稀释),并迅速在振荡器上充分混和,然后置于30-45℃静置培养 2-4h。分别在2h、4h的时候取出培养液并立即计数残存活菌数,与原活菌数进行比较。 2. Tolerance test of simulated gastric juice: Dissolve pepsin in 0.5%-0.85% normal saline to make the final concentration 3g/L, and adjust the pH value to 2.0-4.0 with concentrated hydrochloric acid or 10% NaOH. Take 0.5mL of Enterococcus faecalis ANSE228 suspension and add it to 4.5mL of simulated gastric juice (i.e., ten-fold serial dilution), and quickly mix it fully on a shaker, and then place it at 30-45°C for static culture for 2-4h. The culture solution was taken out at 2h and 4h respectively, and the number of residual viable bacteria was counted immediately, and compared with the original number of viable bacteria. the
3、模拟胆盐的耐受试验: 3. Tolerance test of simulated bile salts:
用胰液素制成1g/L-1.5g/L的溶液,并在溶液中加入0.3%的猪胆盐,用10%的NaOH调整pH为7.0-9.0,然后用0.45μm微滤膜过滤并除菌。将0.5ml粪肠球菌ANSE228活菌菌液接种到4.5ml模拟胆盐中,培养24h后得到培养液,计数残存粪肠球菌ANSE228的活菌数。将菌液在灭菌生理盐水中十倍逐级稀释,并进行MRS平板涂布,然后置于30-45℃静置培养18-36h(粪肠球菌一般要培养18h以上才能长成明显单菌落)。 Use pancreatin to make a solution of 1g/L-1.5g/L, and add 0.3% porcine bile salt to the solution, adjust the pH to 7.0-9.0 with 10% NaOH, then filter with a 0.45μm microfiltration membrane and remove bacteria. Inoculate 0.5ml of Enterococcus faecalis ANSE228 live bacteria solution into 4.5ml of simulated bile salt, cultivate for 24 hours to obtain a culture solution, and count the number of viable bacteria of Enterococcus faecalis ANSE228 remaining. Dilute the bacterial solution tenfold in sterilized normal saline, spread it on an MRS plate, and then place it at 30-45°C for static culture for 18-36 hours (Enterococcus faecalis generally needs to be cultured for more than 18 hours before it can grow into an obvious single colony ). the
模拟胃液和模拟胆盐耐受试验中的计数方法: Counting methods in simulated gastric juice and simulated bile salt tolerance test:
样品用无菌生理盐水十倍梯度稀释,选择合适的稀释度用于计数。采用MRS琼脂培养基,取0.5mL稀释液涂布在平板上,在37℃培养24h。 Samples were serially diluted ten-fold with sterile normal saline, and an appropriate dilution was selected for counting. Using MRS agar medium, 0.5mL of the diluted solution was spread on the plate, and cultured at 37°C for 24h. the
本发明还提供一种微生态制剂,其含有上述粪肠球菌ANSE228。所述微生态制剂是将通过反复分离、纯化、复壮等工艺得到的生物活性强、益生性显著并且抗逆性能良好的粪肠球菌ANSE228,经过液体深层发酵等生产工艺得到发酵液,然后经干燥后制得。 The present invention also provides a microecological preparation, which contains the above-mentioned Enterococcus faecalis ANSE228. The micro-ecological preparation is made of Enterococcus faecalis ANSE228 obtained through repeated separation, purification, rejuvenation and other processes, which has strong biological activity, significant probiotics and good stress resistance, and then undergoes production processes such as liquid submerged fermentation to obtain a fermented liquid, and then dried made later. the
本发明还提供上述微生态制剂在养殖中的应用,其是将微生态制剂作为添加剂,添加到养殖动物的饮水或饲料中。其中,所述粪肠球菌ANSE228在饮水中的添加量为108-1011CFU/L饮水;所述粪肠球菌ANSE228在饲料中的添加量为107-1010CFU/kg饲料。本发明中所述养殖动物包括肉鸡、蛋鸡和猪,还包括鱼、鸭或反刍动物,如牛或羊。 The present invention also provides the application of the above-mentioned probiotics in breeding, which is to add the probiotics as an additive to drinking water or feed of farmed animals. Wherein, the addition amount of Enterococcus faecalis ANSE228 in drinking water is 10 8 -10 11 CFU/L drinking water; the addition amount of Enterococcus faecalis ANSE228 in feed is 10 7 -10 10 CFU/kg feed. The farm animals mentioned in the present invention include broiler hens, laying hens and pigs, and also include fish, ducks or ruminants such as cattle or sheep.
本发明的粪肠球菌ANSE228在其用量较少,例如在饲料中仅为107CFU/kg时即可显著提高动物的生产性能,使肉鸡料肉比降低33.14%、仔猪死淘率降低至0%、蛋鸡料蛋比降低19.8%,显著提高了动物的生产性能,降低了生产成本。 The Enterococcus faecalis ANSE228 of the present invention can significantly improve the production performance of animals when the amount of Enterococcus faecalis ANSE228 is only 10 7 CFU/kg in the feed, reduce the feed-to-meat ratio of broilers by 33.14%, and reduce the death rate of piglets to 0 %, the feed-to-egg ratio of laying hens is reduced by 19.8%, which significantly improves the production performance of animals and reduces production costs.
本发明的有益效果在于: The beneficial effects of the present invention are:
1.本发明的粪肠球菌ANSE228具有显著的益生性,能显著抑制鸡白 痢沙门氏菌、大肠杆菌以及金黄色葡萄球菌等致病菌的生长繁殖。 1. Enterococcus faecalis ANSE228 of the present invention has significant probiotics, can significantly inhibit the growth and reproduction of pathogenic bacteria such as Salmonella pullorum, Escherichia coli and Staphylococcus aureus. the
2.本发明的粪肠球菌ANSE228具有较强的抗逆性,能够耐受模拟胃酸、模拟胆盐以及高温环境,并且能保持较高的活菌存活率,其活菌存活率可达到80%以上。由于饲料加工制粒的过程中温度较高,达到75-90℃,因此菌株需要耐受住这个温度范围才能保证动物吃到的是活菌,而活菌才具有益生性。耐受胃酸和胆盐是为了使粪肠球菌在动物消化道内可以发挥其作用。 2. The Enterococcus faecalis ANSE228 of the present invention has strong stress resistance, can tolerate simulated gastric acid, simulated bile salt and high temperature environment, and can maintain a high survival rate of viable bacteria, and the survival rate of viable bacteria can reach 80% above. Due to the high temperature in the process of feed processing and pelleting, reaching 75-90°C, the strain needs to tolerate this temperature range to ensure that the animals eat live bacteria, and live bacteria have probiotic properties. Tolerance to gastric acid and bile salts is required to allow Enterococcus faecalis to perform its role in the animal's digestive tract. the
3.本发明发掘了已知的粪肠球菌新的用途,开拓了一个新的应用领域。 3. The present invention discovers new uses of the known Enterococcus faecalis and opens up a new application field. the
4.本发明中粪肠球菌ANSE228制成的微生态制剂进入动物肠道后,功能强大的粪肠球菌ANSE228迅速活化、增殖,同时在肠道微生态环境中形成有益优势菌群。 4. After the microecological preparation made of Enterococcus faecalis ANSE228 enters the intestinal tract of animals, the powerful Enterococcus faecalis ANSE228 rapidly activates and proliferates, and at the same time forms beneficial dominant flora in the intestinal microecological environment. the
5.本发明的粪肠球菌ANSE228,具有显著的益生性以及抗逆性,不但可以提高动物的饲料利用效率,节约成本,而且改善了动物体内消化道内环境的稳态,促进营养的吸收利用,促进动物生长,提高经济效益等功效,预示着良好的应用前景。 5. Enterococcus faecalis ANSE228 of the present invention has significant probiotic and stress resistance, not only can improve the feed utilization efficiency of animals, save costs, but also improve the stability of the internal environment of the digestive tract in animals, and promote the absorption and utilization of nutrients. The effects of promoting animal growth and improving economic benefits indicate a good application prospect. the
附图说明 Description of drawings
图1为粪肠球菌ANSE228对鸡白痢沙门氏菌的抑菌圈。 Fig. 1 is the inhibition zone of Enterococcus faecalis ANSE228 to Salmonella pullorum. the
图2为粪肠球菌ANSE228对大肠杆菌的抑菌圈。 Figure 2 is the inhibition zone of Enterococcus faecalis ANSE228 on Escherichia coli. the
图3为粪肠球菌ANSE228对金黄色沙门氏菌的抑菌圈。 Figure 3 is the inhibition zone of Enterococcus faecalis ANSE228 to Salmonella aureus. the
具体实施方式 Detailed ways
以下实施例用于说明本发明,但不用来限制本发明的范围。 The following examples are used to illustrate the present invention, but are not intended to limit the scope of the present invention. the
实施例1粪肠球菌ANSE228发酵液的制备 The preparation of embodiment 1 enterococcus faecalis ANSE228 fermented liquid
取粪肠球菌ANSE228(保藏编号为CGMCC No.4082,活菌浓度为109CFU/ml),接种于50ml培养基中进行摇瓶发酵培养,发酵温度为37℃、pH值7.0、静置培养,发酵时间24h。 Take Enterococcus faecalis ANSE228 (preservation number is CGMCC No.4082, the concentration of viable bacteria is 10 9 CFU/ml), inoculate it in 50ml culture medium for shake flask fermentation culture, the fermentation temperature is 37 ℃, pH value 7.0, static culture , fermentation time 24h.
其中,摇瓶发酵培养基由下述组分组成:玉米粉15g、豆粕20g、葡 萄糖5g、磷酸氢二钾0.15g、磷酸二氢钾1.5g、一水硫酸锰1.0g、七水硫酸镁1.5g、蒸馏水1000mL,pH值为7.0。 Among them, the shake flask fermentation medium is composed of the following components: corn flour 15g, soybean meal 20g, glucose 5g, dipotassium hydrogen phosphate 0.15g, potassium dihydrogen phosphate 1.5g, manganese sulfate monohydrate 1.0g, sulfuric acid heptahydrate Magnesium 1.5g, distilled water 1000mL, pH value 7.0. the
摇瓶发酵结束后进行发酵罐中试试验,取50ml摇瓶发酵种子液接种到10L发酵罐中,装液量为5L、发酵温度为37℃、pH值7.0,搅拌转速300r/min,发酵时间36h。发酵罐中试培养基组成同摇瓶发酵。 After the shake flask fermentation, carry out the fermenter pilot test, take 50ml of shake flask fermentation seed liquid and inoculate it into a 10L fermenter, the liquid volume is 5L, the fermentation temperature is 37°C, the pH value is 7.0, the stirring speed is 300r/min, the fermentation time 36h. The composition of the medium in the pilot test of the fermenter was the same as that of the shake flask fermentation. the
发酵结束后,将发酵液保存在4℃冰箱内备用。 After the fermentation, the fermentation broth was stored in a refrigerator at 4°C for later use. the
实施例2粪肠球菌ANSE228益生性验证 Example 2 Probiotic verification of Enterococcus faecalis ANSE228
制备厚度为4mm左右的MRS平板,将致病菌(大肠杆菌、金黄色葡萄球菌及沙门氏菌)浓度为106的菌悬液100μL涂布于该平板上,以无菌操作将灭菌的不锈钢小管(内径6mm、外径8mm、高10mm的圆形小管,管的两端要光滑),放置在培养基上,轻轻加压,使其与培养基接触无空隙,待数分钟后,分别向各小管中滴加200-300微升的实施例1制备得到的发酵液(活化菌种菌液),勿使其外溢,37℃温度下培养24h,然后测量抑菌圈直径。每个实验三个重复,取平均值。结果如图1-3所示。 Prepare an MRS plate with a thickness of about 4 mm, spread 100 μL of bacterial suspension with a concentration of 10 6 of pathogenic bacteria (E. (a small round tube with an inner diameter of 6 mm, an outer diameter of 8 mm, and a height of 10 mm, the two ends of the tube should be smooth), placed on the medium, and gently pressurized to make it contact with the medium without gaps. Add 200-300 microliters of the fermented liquid (activated strain bacterial liquid) prepared in Example 1 dropwise in each small tube, do not allow it to overflow, cultivate at a temperature of 37° C. for 24 hours, and then measure the diameter of the inhibition zone. Each experiment was repeated three times, and the average value was taken. The results are shown in Figure 1-3.
其中,所述MRS平板是将121℃高温灭菌后的MRS培养基,倒入灭菌后的平板,冷却后形成表面光滑的4mm厚的MRS平板。所述MRS培养基的配方为:蛋白胨10g、牛肉膏10g、酵母浸膏5g、K2HPO42g、柠檬酸氢二铵2g、葡萄糖20g、乙酸钠5g、Tween-801mL、MgSO4·7H2O 0.58g、MnSO4·4H2O 0.25g、琼脂16g、蒸馏水1000ml,pH=7.0。 Wherein, the MRS plate is the MRS culture medium sterilized at 121° C., poured into the sterilized plate, and cooled to form a 4 mm thick MRS plate with a smooth surface. The formula of the MRS medium is: peptone 10g, beef extract 10g, yeast extract 5g, K 2 HPO 4 2g, diammonium hydrogen citrate 2g, glucose 20g, sodium acetate 5g, Tween-801mL, MgSO 4 7H 2 O 0.58g, MnSO 4 ·4H 2 O 0.25g, agar 16g, distilled water 1000ml, pH=7.0.
粪肠球菌ANSE228对鸡白痢沙门氏菌的抑菌圈直径为1.20cm,对大肠杆菌以及金黄色葡萄球菌的抑菌圈直径分别为1.16cm和1.12cm。 The diameter of the inhibition zone of Enterococcus faecalis ANSE228 against Salmonella pullorum was 1.20 cm, and the diameter of the inhibition zone against Escherichia coli and Staphylococcus aureus was 1.16 cm and 1.12 cm, respectively. the
实施例3粪肠球菌ANSE228抗逆性验证 Example 3 Stress resistance verification of Enterococcus faecalis ANSE228
1、取保存好的5mL实施例1制备得到的发酵液注入到离心管1中,采用十倍逐级稀释,在MRS平板1上涂布。再将装有5mL实施例1制备得到的发酵液的离心管2置于80℃水浴锅中加热15min,取加热后的粪肠球菌菌液进行十倍逐级稀释,在MRS平板2上涂布。最后将加热前和加热后的平板均在37℃条件下培养24h,计算粪肠球菌ANSE228加热前后的数量。 1. Take the preserved 5mL fermented liquid prepared in Example 1 and pour it into the centrifuge tube 1, use ten-fold serial dilution, and spread it on the MRS plate 1. Then put the centrifuge tube 2 containing 5mL of the fermented liquid prepared in Example 1 in a water bath at 80°C and heat it for 15 minutes, take the heated Enterococcus faecalis bacterial liquid and carry out ten-fold stepwise dilution, and spread it on the MRS plate 2 . Finally, the plates before and after heating were cultured at 37° C. for 24 hours, and the number of Enterococcus faecalis ANSE228 before and after heating was calculated. the
结果表明,活菌存活率达到了85%。 The results showed that the survival rate of live bacteria reached 85%. the
2、模拟胃液的耐受试验:将胃蛋白酶溶解在0.5%生理盐水中,使其终浓度为3g/L,并用浓盐酸或者10%的NaOH调整pH值到2.0。取0.5mL粪肠球菌ANSE228菌悬液加入到4.5mL的模拟胃液中(即十倍稀释),并迅速在振荡器上充分混和,然后置于37℃静置培养2h。在处理2h后取出培养液并立即计数残存活菌数,与原活菌数进行比较。结果表明,活菌存活率为96%。 2. Tolerance test of simulated gastric juice: Dissolve pepsin in 0.5% normal saline to make the final concentration 3g/L, and adjust the pH value to 2.0 with concentrated hydrochloric acid or 10% NaOH. Take 0.5mL Enterococcus faecalis ANSE228 bacterial suspension and add it into 4.5mL simulated gastric juice (i.e. ten-fold dilution), and quickly mix it fully on a shaker, and then place it at 37°C for static incubation for 2h. After 2 hours of treatment, the culture solution was taken out and the number of residual viable bacteria was counted immediately, and compared with the original number of viable bacteria. The results showed that the survival rate of live bacteria was 96%. the
3、模拟胆盐的耐受试验:用胰液素制成1g/L的溶液,并在溶液中加入0.3%的猪胆盐,用10%的NaOH调整pH为8.0,然后用0.45μm微滤膜过滤并除菌。将0.5ml粪肠球菌ANSE228活菌菌液接种到4.5ml模拟胆盐中,培养24h后得到培养液,计数残存粪肠球菌ANSE228的活菌数。将菌液在灭菌生理盐水中十倍逐级稀释,并进行MRS平板涂布,然后置于37℃静置培养24h。结果表明,活菌存活率为96%。 3. Tolerance test of simulated bile salts: Use pancreatin to make a 1g/L solution, add 0.3% porcine bile salts to the solution, adjust the pH to 8.0 with 10% NaOH, and then use a 0.45 μm microfiltration membrane Filter and sterilize. Inoculate 0.5ml of Enterococcus faecalis ANSE228 live bacteria solution into 4.5ml of simulated bile salt, cultivate for 24 hours to obtain a culture solution, and count the number of viable bacteria of Enterococcus faecalis ANSE228 remaining. The bacterial solution was diluted tenfold in sterilized physiological saline, spread on the MRS plate, and then cultured at 37°C for 24 hours. The results showed that the survival rate of live bacteria was 96%. the
实施例4肉鸡生长性能试验 Embodiment 4 broiler growth performance test
选用120只30日龄健康肉鸡,称重,随机分成4个处理组,每个处理设5个重复,每个重复6只鸡,公母各占1/2。试验饮水分别为:(1)普通饮水(对照组);(2)饮水中粪肠球菌ANSE228活菌浓度为1010CFU/L;(3)饮水中粪肠球菌ANSE228活菌浓度为109CFU/L;(4)饮水中粪肠球菌ANSE228活菌浓度为108CFU/L。试验鸡自由采食和饮水,饲养管理和免疫程序参照肉鸡饲养管理手册。 120 30-day-old healthy broilers were selected, weighed, and randomly divided into 4 treatment groups. Each treatment had 5 replicates, and each replicate had 6 chickens, 1/2 of which were male and female. The test drinking water is: (1) ordinary drinking water (control group); (2) the concentration of Enterococcus faecalis ANSE228 in drinking water is 10 10 CFU/L; (3) the concentration of Enterococcus faecalis ANSE228 in drinking water is 10 9 CFU /L; (4) The concentration of Enterococcus faecalis ANSE228 in drinking water was 10 8 CFU/L. The experimental chickens had free access to food and water, and the feeding management and immunization procedures were in accordance with the broiler feeding management manual.
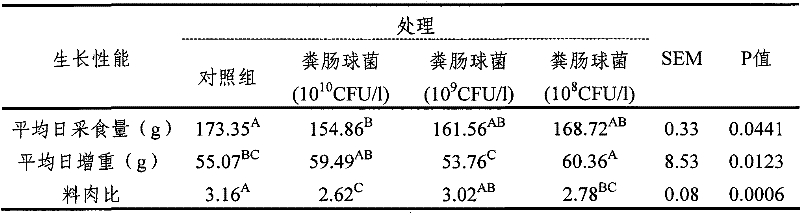
表1粪肠球菌ANSE228对肉鸡生长性能的影响 Table 1 Effect of Enterococcus faecalis ANSE228 on growth performance of broilers
注:同行数据肩标不含相同字母者表示差异显著(P<0.05)(下同) Note: The shoulders of peer data without the same letters indicate significant differences (P<0.05) (the same below)
本试验结果显示,粪肠球菌ANSE228显著提高了肉鸡平均日增重(P=0.0123)和饲料转化效率(P=0.0006)。同时,饮水中粪肠球菌ANSE228活菌浓度为1010CFU/L时,肉鸡饲料转化率最高。 The results of this experiment showed that Enterococcus faecalis ANSE228 significantly increased the average daily gain (P=0.0123) and feed conversion efficiency (P=0.0006) of broilers. At the same time, when the concentration of Enterococcus faecalis ANSE228 in drinking water was 10 10 CFU/L, the feed conversion rate of broilers was the highest.
实施例5蛋鸡生产性能试验: Embodiment 5 laying hen production performance test:
试验为单因子设计,576只罗曼褐商品代蛋鸡随即分为3个处理,每个处理6个重复,每个重复8个笼,每笼4只鸡。处理1为负对照,饲喂基础日粮,不含任何药物饲料添加剂;处理2为正对照,在基础日粮基础上添加杆菌肽锌20mg/kg褐硫酸抗敌素4mg/kg;处理3在基础日粮基础上添加3×109CFU/kg的粪肠球菌ANSE228微生态制剂。试验鸡自由采食和饮水,饲养管理和免疫程序参照蛋鸡饲养管理手册。 The experiment was a single-factor design, and 576 commercial laying hens were randomly divided into 3 treatments, each treatment had 6 replicates, each replicate had 8 cages, and each cage had 4 chickens. Treatment 1 was a negative control, fed a basal ration without any drug feed additives; treatment 2 was a positive control, and bacitracin zinc 20 mg/kg was added to the basal ration; Add 3×10 9 CFU/kg Enterococcus faecalis ANSE228 probiotics to the basal diet. The experimental chickens had free access to food and water, and the feeding management and immunization procedures were referred to the laying hen feeding management manual.
表2粪肠球菌ANSE228对蛋鸡生产性能的影响 Table 2 Effect of Enterococcus faecalis ANSE228 on the performance of laying hens
本试验的结果显示粪肠球菌ANSE228可以显著改善蛋鸡的料蛋比以及提高蛋鸡的产蛋量(p<0.05),并且可以降低生产成本,改善蛋鸡的生产性能,提高经济效益,有效替代抗生素添加剂,适合大规模推广使用。 The results of this test show that Enterococcus faecalis ANSE228 can significantly improve the feed-to-egg ratio of laying hens and increase the egg production of laying hens (p<0.05), and can reduce production costs, improve the production performance of laying hens, increase economic benefits, and effectively It can replace antibiotic additives and is suitable for large-scale promotion and use. the
实施例6仔猪生长性能试验 Embodiment 6 piglet growth performance test
本试验选用35±1日龄、平均体重7.86±0.06kg健康的三元(杜×长×大)杂交断奶仔猪90头随即分配进三个处理,每个处理5个重复,每个 重复6头仔猪(公母各半),各重复组间初始体重无明显差异(p>0.05)。 In this experiment, 90 weaned piglets of 35±1 days old and with an average body weight of 7.86±0.06kg were selected and randomly assigned to three treatments. Each treatment had 5 replicates, and each replicate had 6 piglets. For piglets (half male and half female), there was no significant difference in initial body weight among the repeated groups (p>0.05). the
三个处理分别为:空白对照组、抗生素对照组(75mg/kg金霉素)和试验组(添加4×109CFU/kg粪肠球菌ANSE228微生态制剂)。试验猪自由采食和饮水,饲养管理和免疫程序参照仔猪饲养管理手册。 The three treatments were: blank control group, antibiotic control group (75 mg/kg aureomycin) and test group (adding 4×10 9 CFU/kg Enterococcus faecalis ANSE228 probiotics). The experimental pigs had free access to food and water, and the feeding management and immunization procedures were referred to the piglet feeding management manual.
表3粪肠球菌ANSE228对仔猪生长性能的影响 Table 3 Effect of Enterococcus faecalis ANSE228 on growth performance of piglets
本试验结果显示粪肠球菌ANSE228可显著提高仔猪的平均日增重(p<0.05),改善料肉比,并且显著降低了仔猪的腹泻率以及死淘率(p<0.05)。提高了仔猪的生产性能以及经济效益,能有效替代抗生素添加剂,适合大规模推广使用。 The results of this experiment show that Enterococcus faecalis ANSE228 can significantly increase the average daily gain of piglets (p<0.05), improve the feed-to-meat ratio, and significantly reduce the diarrhea rate and death rate of piglets (p<0.05). The production performance and economic benefits of piglets are improved, the antibiotic additives can be effectively replaced, and the method is suitable for large-scale popularization and use. the
虽然,上文中已经用一般性说明、具体实施方案及实验,对本发明作了详尽的描述,但在本发明基础上,可以对之作一些修改或改进,这对本领域技术人员而言是显而易见的。因此,在不偏离本发明精神的基础上所做的这些修改或改进,均属于本发明要求保护的范围。 Although, the present invention has been described in detail with general description, specific embodiments and experiments above, but on the basis of the present invention, some modifications or improvements can be made to it, which will be obvious to those skilled in the art . Therefore, the modifications or improvements made on the basis of not departing from the spirit of the present invention all belong to the protection scope of the present invention. the
Claims (7)
Priority Applications (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2010105399880A CN102031235B (en) | 2010-11-09 | 2010-11-09 | Enterococcus faecium ANSE228 and application thereof |
Applications Claiming Priority (1)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| CN2010105399880A CN102031235B (en) | 2010-11-09 | 2010-11-09 | Enterococcus faecium ANSE228 and application thereof |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN102031235A CN102031235A (en) | 2011-04-27 |
| CN102031235B true CN102031235B (en) | 2012-07-25 |
Family
ID=43884692
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2010105399880A Active CN102031235B (en) | 2010-11-09 | 2010-11-09 | Enterococcus faecium ANSE228 and application thereof |
Country Status (1)
| Country | Link |
|---|---|
| CN (1) | CN102031235B (en) |
Cited By (24)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN106167785A (en) * | 2016-08-29 | 2016-11-30 | 内蒙古工业大学 | A kind of E. Faecium strains and application thereof |
| US10322151B2 (en) | 2015-06-15 | 2019-06-18 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10391130B2 (en) | 2015-06-15 | 2019-08-27 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10391128B2 (en) | 2015-11-23 | 2019-08-27 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10456444B2 (en) | 2014-12-23 | 2019-10-29 | 4D Pharma Research Limited | Pirin polypeptide and immune modulation |
| US10471108B2 (en) | 2015-11-20 | 2019-11-12 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10485830B2 (en) | 2016-12-12 | 2019-11-26 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US10493112B2 (en) | 2015-06-15 | 2019-12-03 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10500237B2 (en) | 2015-06-15 | 2019-12-10 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10583158B2 (en) | 2016-03-04 | 2020-03-10 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US10610548B2 (en) | 2016-07-13 | 2020-04-07 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US10610550B2 (en) | 2015-11-20 | 2020-04-07 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10736926B2 (en) | 2015-06-15 | 2020-08-11 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10744166B2 (en) | 2015-11-23 | 2020-08-18 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10851137B2 (en) | 2013-04-10 | 2020-12-01 | 4D Pharma Research Limited | Polypeptide and immune modulation |
| US10987387B2 (en) | 2017-05-24 | 2021-04-27 | 4D Pharma Research Limited | Compositions comprising bacterial strain |
| US11007233B2 (en) | 2017-06-14 | 2021-05-18 | 4D Pharma Research Limited | Compositions comprising a bacterial strain of the genus Megasphera and uses thereof |
| US11013773B2 (en) | 2011-07-14 | 2021-05-25 | 4D Pharma Research Limited | Lactic acid bacterial strains |
| US11123379B2 (en) | 2017-06-14 | 2021-09-21 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11123378B2 (en) | 2017-05-22 | 2021-09-21 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11224620B2 (en) | 2016-07-13 | 2022-01-18 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US11266698B2 (en) | 2011-10-07 | 2022-03-08 | 4D Pharma Research Limited | Bacterium for use as a probiotic for nutritional and medical applications |
| US11723933B2 (en) | 2014-12-23 | 2023-08-15 | Cj Bioscience, Inc. | Composition of bacteroides thetaiotaomicron for immune modulation |
| US12048720B2 (en) | 2017-06-14 | 2024-07-30 | Cj Bioscience, Inc. | Compositions comprising bacterial strains |
Families Citing this family (19)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN102373172B (en) * | 2011-11-03 | 2013-03-20 | 北京龙科方舟生物工程技术有限公司 | Enterococcus faecium and application thereof |
| CN102517227B (en) * | 2011-11-24 | 2014-09-17 | 北京大北农科技集团股份有限公司 | Enterococcus faecalis and applications and feed additive and leavening agent thereof |
| CN102657288B (en) * | 2012-03-12 | 2014-09-17 | 北京大北农科技集团股份有限公司 | Formula feed for chickens and preparation method thereof |
| CN103074281A (en) * | 2013-01-21 | 2013-05-01 | 黑龙江八一农垦大学 | Enterococcus faecalis FJL19 and application thereof |
| CN104774782B (en) * | 2014-12-25 | 2017-12-19 | 东北农业大学 | Chicken source enterococcus faecalis separation strains and its application |
| CN104862254B (en) * | 2015-05-26 | 2018-02-16 | 北京好实沃生物技术有限公司 | One Enterococcus faecalis HEW A588 and its application |
| CN105110489A (en) * | 2015-10-10 | 2015-12-02 | 中国水产科学研究院淡水渔业研究中心 | Water-purifying and weed-protecting biological agent for shrimp and crab culture in high-temperature period as well as preparation method and application of biological agent |
| CN105779346B (en) * | 2016-03-25 | 2019-05-17 | 沈阳农业大学 | A kind of enterococcus faecium and its application of bacteriocinogeny |
| CN107338203B (en) * | 2017-07-11 | 2020-02-21 | 福建贝迪药业有限公司 | Enterococcus faecalis for feed, microecological preparation for preventing and treating heat stress of livestock and poultry and preparation method thereof |
| CN107828682A (en) * | 2017-11-06 | 2018-03-23 | 四川恒通动保生物科技有限公司 | A kind of fermentation medium of alctasin and application |
| CN109266576A (en) * | 2018-09-18 | 2019-01-25 | 北京大伟嘉生物技术股份有限公司 | A kind of enterococcus faecalis and its application method |
| CN110004072B (en) * | 2018-11-12 | 2021-08-13 | 西安交通大学 | A probiotic Enterococcus faecalis isolate A3-1 and its application |
| CN109464470B (en) * | 2018-12-28 | 2021-09-07 | 天津坤禾生物科技集团股份有限公司 | Microbial inoculum for preventing and treating litopenaeus vannamei white feces syndrome as well as preparation method and application thereof |
| CN110731422A (en) * | 2019-11-27 | 2020-01-31 | 四川成邦药业有限公司 | fermented product containing thymol and live enterococcus faecalis, and its preparation method and application |
| CN111088184B (en) * | 2019-12-29 | 2021-09-24 | 江南大学 | Enterococcus faecium CCFM1106, its feed additive and its application |
| CN111321104B (en) * | 2020-04-22 | 2023-01-20 | 河南岐伯实业有限公司 | Preparation method of compound organic trace element feed additive for laying hens |
| CN113215019A (en) * | 2021-02-04 | 2021-08-06 | 郑州大学 | Enterococcus faecalis and application thereof |
| CN114480206B (en) * | 2022-02-22 | 2022-12-02 | 江苏三仪生物工程有限公司 | High-temperature-resistant enterococcus faecalis and preparation method and application thereof |
| CN120399991A (en) * | 2025-07-03 | 2025-08-01 | 广西大学 | Enterococcus faecalis FC-1 useful for lowering blood lipids and its application |
Family Cites Families (4)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| CN100506974C (en) * | 2006-11-29 | 2009-07-01 | 康哲医药研究(深圳)有限公司 | Enterococcus faecalis CMS-H001 and its application |
| CN101275158A (en) * | 2007-11-05 | 2008-10-01 | 孙卫 | Screening preparation for superior enterococcus faecalis and application thereof |
| CN101220342B (en) * | 2007-11-14 | 2010-06-23 | 大连三仪动物药品有限公司 | FQ15 enterococcus faecalis and method for producing growth-promoting feed additive by using same |
| CN101701201B (en) * | 2009-11-27 | 2012-06-06 | 南京农业大学 | Enterococcus faecalis and application thereof |
-
2010
- 2010-11-09 CN CN2010105399880A patent/CN102031235B/en active Active
Cited By (45)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US11013773B2 (en) | 2011-07-14 | 2021-05-25 | 4D Pharma Research Limited | Lactic acid bacterial strains |
| US11266698B2 (en) | 2011-10-07 | 2022-03-08 | 4D Pharma Research Limited | Bacterium for use as a probiotic for nutritional and medical applications |
| US11414463B2 (en) | 2013-04-10 | 2022-08-16 | 4D Pharma Research Limited | Polypeptide and immune modulation |
| US10851137B2 (en) | 2013-04-10 | 2020-12-01 | 4D Pharma Research Limited | Polypeptide and immune modulation |
| US11723933B2 (en) | 2014-12-23 | 2023-08-15 | Cj Bioscience, Inc. | Composition of bacteroides thetaiotaomicron for immune modulation |
| US10973872B2 (en) | 2014-12-23 | 2021-04-13 | 4D Pharma Research Limited | Pirin polypeptide and immune modulation |
| US10456444B2 (en) | 2014-12-23 | 2019-10-29 | 4D Pharma Research Limited | Pirin polypeptide and immune modulation |
| US10780134B2 (en) | 2015-06-15 | 2020-09-22 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10744167B2 (en) | 2015-06-15 | 2020-08-18 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10500237B2 (en) | 2015-06-15 | 2019-12-10 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11433106B2 (en) | 2015-06-15 | 2022-09-06 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11389493B2 (en) | 2015-06-15 | 2022-07-19 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11331352B2 (en) | 2015-06-15 | 2022-05-17 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11273185B2 (en) | 2015-06-15 | 2022-03-15 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10322151B2 (en) | 2015-06-15 | 2019-06-18 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10736926B2 (en) | 2015-06-15 | 2020-08-11 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10493112B2 (en) | 2015-06-15 | 2019-12-03 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11040075B2 (en) | 2015-06-15 | 2021-06-22 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10391130B2 (en) | 2015-06-15 | 2019-08-27 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10864236B2 (en) | 2015-06-15 | 2020-12-15 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10471108B2 (en) | 2015-11-20 | 2019-11-12 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11058732B2 (en) | 2015-11-20 | 2021-07-13 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10610550B2 (en) | 2015-11-20 | 2020-04-07 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10391128B2 (en) | 2015-11-23 | 2019-08-27 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10744166B2 (en) | 2015-11-23 | 2020-08-18 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10583158B2 (en) | 2016-03-04 | 2020-03-10 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US11224620B2 (en) | 2016-07-13 | 2022-01-18 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US10610548B2 (en) | 2016-07-13 | 2020-04-07 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US10967010B2 (en) | 2016-07-13 | 2021-04-06 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US10960031B2 (en) | 2016-07-13 | 2021-03-30 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US10610549B2 (en) | 2016-07-13 | 2020-04-07 | 4D Pharma Plc | Composition comprising bacterial strains |
| CN106167785A (en) * | 2016-08-29 | 2016-11-30 | 内蒙古工业大学 | A kind of E. Faecium strains and application thereof |
| CN106167785B (en) * | 2016-08-29 | 2019-05-28 | 内蒙古工业大学 | A kind of E. Faecium strains and its application |
| US10485830B2 (en) | 2016-12-12 | 2019-11-26 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US10898526B2 (en) | 2016-12-12 | 2021-01-26 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US10543238B2 (en) | 2016-12-12 | 2020-01-28 | 4D Pharma Plc | Compositions comprising bacterial strains |
| US11376284B2 (en) | 2017-05-22 | 2022-07-05 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11382936B2 (en) | 2017-05-22 | 2022-07-12 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11123378B2 (en) | 2017-05-22 | 2021-09-21 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US10987387B2 (en) | 2017-05-24 | 2021-04-27 | 4D Pharma Research Limited | Compositions comprising bacterial strain |
| US11007233B2 (en) | 2017-06-14 | 2021-05-18 | 4D Pharma Research Limited | Compositions comprising a bacterial strain of the genus Megasphera and uses thereof |
| US11660319B2 (en) | 2017-06-14 | 2023-05-30 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11123379B2 (en) | 2017-06-14 | 2021-09-21 | 4D Pharma Research Limited | Compositions comprising bacterial strains |
| US11779613B2 (en) | 2017-06-14 | 2023-10-10 | Cj Bioscience, Inc. | Compositions comprising a bacterial strain of the genus Megasphera and uses thereof |
| US12048720B2 (en) | 2017-06-14 | 2024-07-30 | Cj Bioscience, Inc. | Compositions comprising bacterial strains |
Also Published As
| Publication number | Publication date |
|---|---|
| CN102031235A (en) | 2011-04-27 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN102031235B (en) | Enterococcus faecium ANSE228 and application thereof | |
| CN103981118B (en) | A kind of bacillus subtilis feed addictive and its preparation method and application | |
| CN102409015B (en) | Composite micro-ecological preparation as well as premixed material and application of preparation in feed additive | |
| CN102399733B (en) | Lactobacillus johnsonii, microbial inoculum, application and premix thereof | |
| CN101580799B (en) | A kind of compound probiotics and its application | |
| CN101638627B (en) | Bacillus subtilis and application thereof in biological feed additives | |
| CN101463329B (en) | Saccharomyces cerevisiae, yeast preparation including the same, feed, and preparation and use thereof | |
| CN102212495B (en) | Lactobacillus acidophilus and application thereof | |
| CN112011481B (en) | Lactobacillus reuteri for preventing and treating bacterial diarrhea of livestock and poultry and application thereof | |
| CN103005159A (en) | Preparation method of ginkgo leaf biological feed additive | |
| CN101978849B (en) | Composite micro-ecological preparation and application thereof | |
| CN102093967A (en) | Mink source enterococcus faecium and application thereof | |
| CN101919494A (en) | Composite microbial additive and preparation method and application thereof | |
| CN102028110A (en) | Probiotic-astragalus polysaccharide composition for feeding commercial chicken | |
| CN106906154A (en) | One lactobacillus plantarum strain and its feed addictive and feed | |
| CN102657288B (en) | Formula feed for chickens and preparation method thereof | |
| CN102517227B (en) | Enterococcus faecalis and applications and feed additive and leavening agent thereof | |
| CN113717881A (en) | Bacillus coagulans BC66 and application thereof | |
| CN110878266A (en) | A strain of Lactobacillus johnsonii and its use | |
| CN101904411B (en) | Composite microbial ecological agent and application thereof | |
| Suprayudi et al. | Screening of probiotics from the digestive tract of gouramy (Osphronemus goramy) and their potency to enhance the growth of tilapia (Oreochromis niloticus) | |
| CN102919557A (en) | Growing fattening pig probiotics composite premix and accessory material | |
| CN105199978A (en) | Bacillus coagulans preparation for livestock breeding and preparation method thereof | |
| CN103555634B (en) | Lactobacillus acidophilus and applications thereof | |
| CN108102950B (en) | Preparation method and application of poultry composite probiotic feed |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C53 | Correction of patent for invention or patent application | ||
| CB03 | Change of inventor or designer information |
Inventor after: Ma Qiugang Inventor after: Zhao Lihong Inventor after: Gao Xin Inventor after: Ji Cheng Inventor after: Fan Yu Inventor after: Li Xiaoying Inventor before: Ji Cheng Inventor before: Ma Qiugang Inventor before: Gao Xin Inventor before: Fan Yu Inventor before: Li Xiaoying |
|
| COR | Change of bibliographic data |
Free format text: CORRECT: INVENTOR; FROM: JI CHENG MA QIUGANG GAO XIN FAN YU LI XIAOYING TO: MA QIUGANG ZHAO LIHONG GAO XIN JI CHENG FAN YU LI XIAOYING |
|
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| TR01 | Transfer of patent right | ||
| TR01 | Transfer of patent right |
Effective date of registration: 20181008 Address after: 063200 new industrial park of Caofeidian Industrial Zone, Tangshan City, Hebei Patentee after: Ke run Sheng Technology Development Co., Ltd. Address before: 100193 West Road, Old Summer Palace, Haidian District, Haidian District, Beijing Patentee before: China Agricultural University |