CN101184789B - High-molecular compound and high-molecular luminescent device using the same - Google Patents
High-molecular compound and high-molecular luminescent device using the same Download PDFInfo
- Publication number
- CN101184789B CN101184789B CN2006800189430A CN200680018943A CN101184789B CN 101184789 B CN101184789 B CN 101184789B CN 2006800189430 A CN2006800189430 A CN 2006800189430A CN 200680018943 A CN200680018943 A CN 200680018943A CN 101184789 B CN101184789 B CN 101184789B
- Authority
- CN
- China
- Prior art keywords
- group
- formula
- independently represent
- ring
- polymer
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Expired - Fee Related
Links
- 0 CC1**=CC1 Chemical compound CC1**=CC1 0.000 description 41
- DZYSMGRTXSUCRH-UHFFFAOYSA-N C(C1c2ccccc2-2)OCc3c1c-2ccc3 Chemical compound C(C1c2ccccc2-2)OCc3c1c-2ccc3 DZYSMGRTXSUCRH-UHFFFAOYSA-N 0.000 description 1
- HKMJHVGVVFVBGN-UHFFFAOYSA-N C(C1c2ccccc2-2)SCc3c1c-2ccc3 Chemical compound C(C1c2ccccc2-2)SCc3c1c-2ccc3 HKMJHVGVVFVBGN-UHFFFAOYSA-N 0.000 description 1
- NYIMZOMNEWZVKM-UHFFFAOYSA-N C(CC1c2cccnc2-2)Cc3c1c-2ncc3 Chemical compound C(CC1c2cccnc2-2)Cc3c1c-2ncc3 NYIMZOMNEWZVKM-UHFFFAOYSA-N 0.000 description 1
- HSZMVSDWJHNPBN-UHFFFAOYSA-N C(Cc1ccc2)CS3c1c2-c1ccccc31 Chemical compound C(Cc1ccc2)CS3c1c2-c1ccccc31 HSZMVSDWJHNPBN-UHFFFAOYSA-N 0.000 description 1
- POCIHYGQOZRCEK-UHFFFAOYSA-N C(Cc1ccc2)C[n]3c1c2c1c3cccc1 Chemical compound C(Cc1ccc2)C[n]3c1c2c1c3cccc1 POCIHYGQOZRCEK-UHFFFAOYSA-N 0.000 description 1
- WDKORMQZLXGGGB-UHFFFAOYSA-N C1C(c2ccccc2-c2ccc3)c2c3OC1 Chemical compound C1C(c2ccccc2-c2ccc3)c2c3OC1 WDKORMQZLXGGGB-UHFFFAOYSA-N 0.000 description 1
- NGRMQWWOAJNWDZ-UHFFFAOYSA-N C1NCc2cccc-3c2C1c1ccccc-31 Chemical compound C1NCc2cccc-3c2C1c1ccccc-31 NGRMQWWOAJNWDZ-UHFFFAOYSA-N 0.000 description 1
- OZKBDTBXALXQRN-UHFFFAOYSA-N C=C(CCCC1c2ccccc22)C1=C2N=N Chemical compound C=C(CCCC1c2ccccc22)C1=C2N=N OZKBDTBXALXQRN-UHFFFAOYSA-N 0.000 description 1
- SBLUBMSVDLDCJH-UHFFFAOYSA-N C=CC(C1c2c3nccc2CCC1)=C3N=N Chemical compound C=CC(C1c2c3nccc2CCC1)=C3N=N SBLUBMSVDLDCJH-UHFFFAOYSA-N 0.000 description 1
- AVWBMBWKBOWSTB-UHFFFAOYSA-N CB1OC(C)(CCB2Oc(cccc3)c3O2)C(C)(C)O1 Chemical compound CB1OC(C)(CCB2Oc(cccc3)c3O2)C(C)(C)O1 AVWBMBWKBOWSTB-UHFFFAOYSA-N 0.000 description 1
- YGYNBBAUIYTWBF-UHFFFAOYSA-N Cc1cc2ccc(C)cc2cc1 Chemical compound Cc1cc2ccc(C)cc2cc1 YGYNBBAUIYTWBF-UHFFFAOYSA-N 0.000 description 1
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/62—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with more than three condensed rings
- C07C13/66—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with more than three condensed rings the condensed ring system contains only four rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C25/00—Compounds containing at least one halogen atom bound to a six-membered aromatic ring
- C07C25/18—Polycyclic aromatic halogenated hydrocarbons
- C07C25/22—Polycyclic aromatic halogenated hydrocarbons with condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/62—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings with more than three condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C13/00—Cyclic hydrocarbons containing rings other than, or in addition to, six-membered aromatic rings
- C07C13/28—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof
- C07C13/32—Polycyclic hydrocarbons or acyclic hydrocarbon derivatives thereof with condensed rings
- C07C13/72—Spiro hydrocarbons
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C211/00—Compounds containing amino groups bound to a carbon skeleton
- C07C211/43—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton
- C07C211/54—Compounds containing amino groups bound to a carbon skeleton having amino groups bound to carbon atoms of six-membered aromatic rings of the carbon skeleton having amino groups bound to two or three six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C255/00—Carboxylic acid nitriles
- C07C255/49—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton
- C07C255/54—Carboxylic acid nitriles having cyano groups bound to carbon atoms of six-membered aromatic rings of a carbon skeleton containing cyano groups and etherified hydroxy groups bound to the carbon skeleton
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C43/00—Ethers; Compounds having groups, groups or groups
- C07C43/02—Ethers
- C07C43/20—Ethers having an ether-oxygen atom bound to a carbon atom of a six-membered aromatic ring
- C07C43/21—Ethers having an ether-oxygen atom bound to a carbon atom of a six-membered aromatic ring containing rings other than six-membered aromatic rings
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G61/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
-
- C—CHEMISTRY; METALLURGY
- C08—ORGANIC MACROMOLECULAR COMPOUNDS; THEIR PREPARATION OR CHEMICAL WORKING-UP; COMPOSITIONS BASED THEREON
- C08G—MACROMOLECULAR COMPOUNDS OBTAINED OTHERWISE THAN BY REACTIONS ONLY INVOLVING UNSATURATED CARBON-TO-CARBON BONDS
- C08G61/00—Macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain of the macromolecule
- C08G61/02—Macromolecular compounds containing only carbon atoms in the main chain of the macromolecule, e.g. polyxylylenes
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09D—COATING COMPOSITIONS, e.g. PAINTS, VARNISHES OR LACQUERS; FILLING PASTES; CHEMICAL PAINT OR INK REMOVERS; INKS; CORRECTING FLUIDS; WOODSTAINS; PASTES OR SOLIDS FOR COLOURING OR PRINTING; USE OF MATERIALS THEREFOR
- C09D165/00—Coating compositions based on macromolecular compounds obtained by reactions forming a carbon-to-carbon link in the main chain; Coating compositions based on derivatives of such polymers
-
- C—CHEMISTRY; METALLURGY
- C09—DYES; PAINTS; POLISHES; NATURAL RESINS; ADHESIVES; COMPOSITIONS NOT OTHERWISE PROVIDED FOR; APPLICATIONS OF MATERIALS NOT OTHERWISE PROVIDED FOR
- C09K—MATERIALS FOR MISCELLANEOUS APPLICATIONS, NOT PROVIDED FOR ELSEWHERE
- C09K11/00—Luminescent, e.g. electroluminescent, chemiluminescent materials
- C09K11/06—Luminescent, e.g. electroluminescent, chemiluminescent materials containing organic luminescent materials
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K50/00—Organic light-emitting devices
- H10K50/10—OLEDs or polymer light-emitting diodes [PLED]
- H10K50/11—OLEDs or polymer light-emitting diodes [PLED] characterised by the electroluminescent [EL] layers
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/10—Organic polymers or oligomers
- H10K85/111—Organic polymers or oligomers comprising aromatic, heteroaromatic, or aryl chains, e.g. polyaniline, polyphenylene or polyphenylene vinylene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/10—Organic polymers or oligomers
- H10K85/111—Organic polymers or oligomers comprising aromatic, heteroaromatic, or aryl chains, e.g. polyaniline, polyphenylene or polyphenylene vinylene
- H10K85/113—Heteroaromatic compounds comprising sulfur or selene, e.g. polythiophene
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/10—Organic polymers or oligomers
- H10K85/111—Organic polymers or oligomers comprising aromatic, heteroaromatic, or aryl chains, e.g. polyaniline, polyphenylene or polyphenylene vinylene
- H10K85/115—Polyfluorene; Derivatives thereof
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/60—Organic compounds having low molecular weight
- H10K85/631—Amine compounds having at least two aryl rest on at least one amine-nitrogen atom, e.g. triphenylamine
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/40—Ortho- or ortho- and peri-condensed systems containing four condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/52—Ortho- or ortho- and peri-condensed systems containing five condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/02—Ortho- or ortho- and peri-condensed systems
- C07C2603/54—Ortho- or ortho- and peri-condensed systems containing more than five condensed rings
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/93—Spiro compounds
- C07C2603/94—Spiro compounds containing "free" spiro atoms
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07C—ACYCLIC OR CARBOCYCLIC COMPOUNDS
- C07C2603/00—Systems containing at least three condensed rings
- C07C2603/93—Spiro compounds
- C07C2603/95—Spiro compounds containing "not free" spiro atoms
- C07C2603/96—Spiro compounds containing "not free" spiro atoms containing at least one ring with less than six members
- C07C2603/97—Spiro compounds containing "not free" spiro atoms containing at least one ring with less than six members containing five-membered rings
-
- H—ELECTRICITY
- H10—SEMICONDUCTOR DEVICES; ELECTRIC SOLID-STATE DEVICES NOT OTHERWISE PROVIDED FOR
- H10K—ORGANIC ELECTRIC SOLID-STATE DEVICES
- H10K85/00—Organic materials used in the body or electrodes of devices covered by this subclass
- H10K85/30—Coordination compounds
- H10K85/341—Transition metal complexes, e.g. Ru(II)polypyridine complexes
- H10K85/342—Transition metal complexes, e.g. Ru(II)polypyridine complexes comprising iridium
Landscapes
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Materials Engineering (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Physics & Mathematics (AREA)
- Medicinal Chemistry (AREA)
- Health & Medical Sciences (AREA)
- Polymers & Plastics (AREA)
- Optics & Photonics (AREA)
- Life Sciences & Earth Sciences (AREA)
- Wood Science & Technology (AREA)
- Spectroscopy & Molecular Physics (AREA)
- Polyoxymethylene Polymers And Polymers With Carbon-To-Carbon Bonds (AREA)
- Electroluminescent Light Sources (AREA)
Abstract
Description
技术领域 technical field
本发明涉及高分子化合物及使用了它的高分子发光元件。 The present invention relates to a polymer compound and a polymer light-emitting device using the same. the
背景技术 Background technique
高分子量的发光材料或电荷输送材料由于可溶于溶剂中,并可以利用涂布法形成发光元件的有机层,因此被进行各种研究。作为能够作为发光材料或电荷输送材料用于高分子发光元件(高分子LED)等电子元件中的高分子化合物,已知有聚芴类(国际公开第99/54385)。 High-molecular-weight light-emitting materials and charge-transporting materials have been studied in various ways because they are soluble in solvents and can form an organic layer of a light-emitting device by a coating method. Polyfluorenes are known as polymer compounds that can be used in electronic devices such as polymer light-emitting devices (polymer LEDs) as light-emitting materials or charge transport materials (International Publication No. 99/54385). the
但是,将上述高分子化合物作为发光材料或电荷输送材料等使用的元件的元件性能不一定是在实用上可以令人满意的水平。 However, the device performance of a device using the above-mentioned polymer compound as a light-emitting material, a charge transport material, etc. is not always at a practically satisfactory level. the
例如,使用了上述高分子化合物的高分子LED在其发光效率、发光颜色的色调等元件性能方面,尚不是在实用上令人满意的水平。 For example, polymer LEDs using the above-mentioned polymer compounds are not yet at a practically satisfactory level in terms of device performance such as luminous efficiency and luminous color tone. the
发明内容 Contents of the invention
本发明的目的在于,提供一种在作为电子元件的材料使用的情况下可以提供元件性能优良的电子元件的高分子化合物。 An object of the present invention is to provide a polymer compound capable of providing an electronic device having excellent device performance when used as a material of an electronic device. the
即,本发明提供含有以下述式(1)表示的化合物的残基的至少一个的高分子化合物。 That is, the present invention provides a polymer compound containing at least one residue of a compound represented by the following formula (1). the
(式1) (Formula 1)
[式中,A环、B环及C环分别独立地表示也可以具有取代基的芳香族环或非芳香族环,Z1、Z2、Z3、Z4及Z5分别独立地表示C-(Q)z或 氮原子,Q表示取代基或氢原子,z表示0或1,A环与B环也可以相互共有构成各自的环的Z5以外的原子,A环、B环及C环的一个以上两个以下为非芳香族环。] [In the formula, Ring A, Ring B, and Ring C independently represent an aromatic ring or a non-aromatic ring that may have a substituent, and Z 1 , Z 2 , Z 3 , Z 4 , and Z 5 independently represent C -(Q)z or a nitrogen atom, Q represents a substituent or a hydrogen atom, z represents 0 or 1, and the A ring and the B ring can also share atoms other than Z 5 constituting their respective rings, the A ring, the B ring and the C ring One or more and two or less rings are non-aromatic rings. ]
具体实施方式 Detailed ways
式(1)中,A环、B环及C环分别独立地表示也可以具有取代基的芳香族环或非芳香族环。 In formula (1), ring A, ring B, and ring C each independently represent an aromatic ring or a non-aromatic ring which may have a substituent. the
作为芳香族环,可以举出在环构造中含有4n+2个π电子的物质。具体来说,可以举出苯环、环癸五烯环等芳香族烃类;呋喃环、噻吩环、吡咯类、吡啶环、嘧啶环、哒嗪环等芳香族杂环。 Examples of the aromatic ring include those containing 4n+2 π electrons in the ring structure. Specifically, aromatic hydrocarbons such as benzene rings and cyclodepentacene rings; aromatic heterocycles such as furan rings, thiophene rings, pyrroles, pyridine rings, pyrimidine rings, and pyridazine rings are mentioned. the
作为非芳香族环,可以举出环戊烷环、环戊烯环、环戊二烯环、环己烷环、环己烯环、环己二烯环、环庚烷环、环庚烯环、环庚二烯环、环庚三烯环、环辛烷环、环辛烯环、环辛二烯环、环辛三烯环、环辛四烯环、环壬烷环、环壬烯环、环壬二烯环、环壬三烯环、环癸烷环、环癸烯环、环癸二烯环、环癸三烯环、环癸四烯环、环十二烷环、环十二烯环、环十二烷二烯环、环十二烷三烯环、环十二烷四烯环、环十二烷五烯环、环十一烷环、环十一烯环、环十一烷二烯环、环十一烷三烯环、环十一烷四烯环、环十一烷五烯环、环十一烷六烯环等脂环式环;吡喃环、噻喃环等非芳香族性的杂环。 Examples of the non-aromatic ring include a cyclopentane ring, a cyclopentene ring, a cyclopentadiene ring, a cyclohexane ring, a cyclohexene ring, a cyclohexadiene ring, a cycloheptane ring, and a cycloheptene ring. , cycloheptadiene ring, cycloheptatriene ring, cyclooctane ring, cyclooctene ring, cyclooctadiene ring, cyclooctatriene ring, cyclooctene tetraene ring, cyclononane ring, cyclononene ring , cyclononadiene ring, cyclononatriene ring, cyclodecane ring, cyclodecene ring, cyclodecadiene ring, cyclodecatriene ring, cyclodecatetraene ring, cyclododecane ring, cyclododeca Alkene ring, cyclododecanadiene ring, cyclododecanetriene ring, cyclododecanetraene ring, cyclododecanpentaene ring, cycloundecane ring, cycloundecene ring, cycloundeca Alicyclic rings such as alkanadiene ring, cycloundecanetriene ring, cycloundecanetetraene ring, cycloundecanepentaene ring, cycloundecane hexaene ring; pyran ring, thiopyran ring, etc. non-aromatic heterocycle. the
在芳香族环或非芳香族环具有取代基的情况下,作为取代基可以举出烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、取代氨基、取代甲硅烷基、氟原子、酰基、酰氧基、酰胺基、酰亚胺基、1价杂环基、羧基、取代羧基、氰基及硝基,更优选烷基、烷氧基、芳基、芳氧基、芳烷基、芳基烷氧基、芳基烷硫基。 When the aromatic ring or non-aromatic ring has a substituent, examples of the substituent include alkyl, alkoxy, alkylthio, aryl, aryloxy, arylthio, aralkyl, aryl Alkoxy group, arylalkylthio group, arylalkenyl group, arylalkynyl group, substituted amino group, substituted silyl group, fluorine atom, acyl group, acyloxy group, amido group, imide group, monovalent heterocyclic ring group, carboxyl group, substituted carboxyl group, cyano group and nitro group, more preferably alkyl group, alkoxy group, aryl group, aryloxy group, aralkyl group, arylalkoxy group, arylalkylthio group. the
这里,烷基无论是直链、支链或环状的哪种都可以,碳数通常为1~20左右,优选碳数3~20,作为其具体例,可以举出甲基、乙基、丙基、异丙基、丁基、异丁基、叔丁基、戊基、异戊基、己基、环己基、庚基、辛基、2-乙基己基、壬基、癸基、3,7-二甲基辛基、月桂基、三氟甲基、五氟乙基、全氟丁基、全氟己基、全氟辛基等,优选戊基、异戊基、 己基、辛基、2-乙基己基、癸基、3,7-二甲基辛基。 Here, the alkyl group may be linear, branched or cyclic, and generally has about 1 to 20 carbon atoms, preferably 3 to 20 carbon atoms. Specific examples thereof include methyl, ethyl, Propyl, isopropyl, butyl, isobutyl, tert-butyl, pentyl, isopentyl, hexyl, cyclohexyl, heptyl, octyl, 2-ethylhexyl, nonyl, decyl, 3, 7-Dimethyloctyl, lauryl, trifluoromethyl, pentafluoroethyl, perfluorobutyl, perfluorohexyl, perfluorooctyl, etc., preferably pentyl, isopentyl, hexyl, octyl, 2 - ethylhexyl, decyl, 3,7-dimethyloctyl. the
烷氧基无论是直链、支链或环状的哪种都可以,碳数通常为1~20左右,优选碳数3~20,作为其具体例,可以举出甲氧基、乙氧基、丙氧基、异丙氧基、丁氧基、异丁氧基、叔丁氧基、戊氧基、己氧基、环己氧基、庚氧基、辛氧基、2-乙基己氧基、壬氧基、癸氧基、3,7-二甲基辛氧基、月桂基氧基、三氟甲氧基、五氟乙氧基、全氟丁氧基、全氟己基、全氟辛基、甲氧基甲基氧基、2-甲氧基乙基氧基等,优选戊氧基、己氧基、辛氧基、2-乙基己氧基、癸氧基、3,7-二甲基辛氧基。 The alkoxy group may be linear, branched or cyclic, and generally has about 1 to 20 carbon atoms, preferably 3 to 20 carbon atoms. Specific examples thereof include methoxy and ethoxy. , propoxy, isopropoxy, butoxy, isobutoxy, tert-butoxy, pentyloxy, hexyloxy, cyclohexyloxy, heptyloxy, octyloxy, 2-ethylhexyl Oxygen, nonyloxy, decyloxy, 3,7-dimethyloctyloxy, lauryloxy, trifluoromethoxy, pentafluoroethoxy, perfluorobutoxy, perfluorohexyl, perfluorobutoxy Fluorooctyl, methoxymethyloxy, 2-methoxyethyloxy, etc., preferably pentyloxy, hexyloxy, octyloxy, 2-ethylhexyloxy, decyloxy, 3, 7-Dimethyloctyloxy. the
烷硫基无论是直链、支链或环状的哪种都可以,碳数通常为1~20左右,优选碳数3~20,作为其具体例,可以举出甲硫基、乙硫基、丙硫基、异丙硫基、丁硫基、异丁硫基、叔丁硫基、戊硫基、己硫基、环己硫基、庚硫基、辛硫基、2-乙基己基硫基、壬硫基、癸硫基、3,7-二甲基辛硫基、月桂基硫基、三氟甲基硫基等,优选戊硫基、己硫基、辛硫基、2-乙基己基硫基、癸硫基、3,7-二甲基辛基硫基。 The alkylthio group may be linear, branched or cyclic, and generally has about 1 to 20 carbon atoms, preferably 3 to 20 carbon atoms. Specific examples thereof include methylthio and ethylthio , propylthio, isopropylthio, butylthio, isobutylthio, tert-butylthio, pentylthio, hexylthio, cyclohexylthio, heptylthio, octylthio, 2-ethylhexyl Sulfuryl, nonylthio, decylthio, 3,7-dimethyloctylthio, laurylthio, trifluoromethylthio, etc., preferably pentylthio, hexylthio, octylthio, 2- Ethylhexylthio, decylthio, 3,7-dimethyloctylthio. the
芳基是从芳香族烃中去除了1个氢原子的原子团,包括具有稠环的物质、2个以上独立的苯环或稠环直接或借助亚乙烯基等基结合的物质。芳基的碳数通常为6~60左右,优选7~48,作为其具体例,可以例示出苯基、C1~C12烷氧基苯基(C1~C12表示碳数为1~12。以下相同。)、C1~C12烷基苯基、1-萘基、2-萘基、1-蒽基、2-蒽基、9-蒽基、五氟苯基等,优选C1~C12烷氧基苯基、C1~C12烷基苯基。作为C1~C12烷氧基,具体来说可以例示出甲氧基、乙氧基、丙氧基、异丙氧基、丁氧基、异丁氧基、叔丁氧基、戊氧基、己氧基、环己氧基、庚氧基、辛氧基、2-乙基己氧基、壬氧基、癸氧基、3,7-二甲基辛氧基、月桂基氧基等。 An aryl group is an atomic group obtained by removing one hydrogen atom from an aromatic hydrocarbon, and includes a substance having a condensed ring, and a substance in which two or more independent benzene rings or fused rings are bonded directly or through a group such as a vinylidene group. The carbon number of the aryl group is usually about 6 to 60, preferably 7 to 48. Specific examples thereof include phenyl, C 1 to C 12 alkoxyphenyl (C 1 to C 12 means that the carbon number is 1 to 12. Same below.), C 1 ~C 12 alkylphenyl, 1-naphthyl, 2-naphthyl, 1-anthracenyl, 2-anthracenyl, 9-anthracenyl, pentafluorophenyl, etc., preferably C 1 -C 12 alkoxyphenyl, C 1 -C 12 alkylphenyl. Specific examples of the C 1 -C 12 alkoxy group include methoxy, ethoxy, propoxy, isopropoxy, butoxy, isobutoxy, tert-butoxy, and pentyloxy. , hexyloxy, cyclohexyloxy, heptyloxy, octyloxy, 2-ethylhexyloxy, nonyloxy, decyloxy, 3,7-dimethyloctyloxy, lauryloxy, etc. .
作为C1~C12烷基苯基,具体来说可以例示出甲基苯基、乙基苯基、二甲基苯基、丙基苯基、基、甲基乙基苯基、异丙基苯基、丁基苯基、异丁基苯基、叔丁基苯基、戊基苯基、异戊基苯基、己基苯基、庚基苯基、辛基苯基、壬基苯基、癸基苯基、十二烷基苯基等。 Specific examples of C 1 -C 12 alkylphenyl groups include methylphenyl, ethylphenyl, dimethylphenyl, propylphenyl, base, methylethylphenyl, isopropylphenyl, butylphenyl, isobutylphenyl, tert-butylphenyl, pentylphenyl, isopentylphenyl, hexylphenyl, heptylphenyl Base, octylphenyl, nonylphenyl, decylphenyl, dodecylphenyl, etc.
芳氧基的碳数通常为6~60左右,优选7~48,作为其具体例,可以例示出苯氧基、C1~C12烷氧基苯氧基、C1~C12烷基苯氧基、1-萘氧基、2-萘氧基、五氟苯基氧基等,优选C1~C12烷氧基苯氧基、C1~C12烷基 苯氧基。 The carbon number of the aryloxy group is usually about 6 to 60, preferably 7 to 48. Specific examples thereof include phenoxy, C 1 to C 12 alkoxyphenoxy, C 1 to C 12 alkylbenzene Oxy, 1-naphthyloxy, 2-naphthyloxy, pentafluorophenyloxy, etc., preferably C 1 -C 12 alkoxyphenoxy, C 1 -C 12 alkylphenoxy.
作为C1~C12烷氧基,具体来说可以例示出甲氧基、乙氧基、丙氧基、异丙氧基、丁氧基、异丁氧基、叔丁氧基、戊氧基、己氧基、环己氧基、庚氧基、辛氧基、2-乙基己氧基、壬氧基、癸氧基、3,7-二甲基辛氧基、月桂基氧基等。 Specific examples of the C 1 -C 12 alkoxy group include methoxy, ethoxy, propoxy, isopropoxy, butoxy, isobutoxy, tert-butoxy, and pentyloxy. , hexyloxy, cyclohexyloxy, heptyloxy, octyloxy, 2-ethylhexyloxy, nonyloxy, decyloxy, 3,7-dimethyloctyloxy, lauryloxy, etc. .
作为C1~C12烷基苯氧基,具体来说可以例示出甲基苯氧基、乙基苯氧基、二甲基苯氧基、丙基苯氧基、1,3,5-三甲基苯氧基、甲基乙基苯氧基、异丙基苯氧基、丁基苯氧基、异丁基苯氧基、叔丁基苯氧基、戊基苯氧基、异戊基苯氧基、己基苯氧基、庚基苯氧基、辛基苯氧基、壬基苯氧基、癸基苯氧基、十二烷基苯氧基等。 Specific examples of C 1 -C 12 alkylphenoxy groups include methylphenoxy, ethylphenoxy, dimethylphenoxy, propylphenoxy, 1,3,5-tris Methylphenoxy, methylethylphenoxy, isopropylphenoxy, butylphenoxy, isobutylphenoxy, tert-butylphenoxy, pentylphenoxy, isopentyl Phenoxy, hexylphenoxy, heptylphenoxy, octylphenoxy, nonylphenoxy, decylphenoxy, dodecylphenoxy and the like.
芳硫基的碳数通常为3~60,作为其具体例,可以例示出苯基硫基、C1~C12烷氧基苯基硫基、C1~C12烷基苯基硫基、1-萘硫基、2-萘硫基、五氟苯基硫基等,优选C1~C12烷氧基苯基硫基、C1~C12烷基苯基硫基。 The carbon number of the arylthio group is usually 3 to 60, and specific examples thereof include phenylthio, C 1 to C 12 alkoxyphenylthio, C 1 to C 12 alkylphenylthio, 1-naphthylthio, 2-naphthylthio, pentafluorophenylthio, etc., preferably C 1 -C 12 alkoxyphenylthio, C 1 -C 12 alkylphenylthio.
芳烷基的碳数通常为7~60左右,优选7~48,作为其具体例,可以例示出苯基-C1~C12烷基、C1~C12烷氧基苯基-C1~C12烷基、C1~C12 烷基苯基-C1~C12烷基、1-萘基-C1~C12烷基、2-萘基-C1~C12烷基等,优选C1~C12烷氧基苯基-C1~C12烷基、C1~C12烷基苯基-C1~C12烷基。 The carbon number of the aralkyl group is usually about 7 to 60, preferably 7 to 48. Specific examples thereof include phenyl-C 1 -C 12 alkyl, C 1 -C 12 alkoxyphenyl-C 1 ~C 12 alkyl, C 1 ~C 12 alkylphenyl-C 1 ~C 12 alkyl, 1-naphthyl-C 1 ~C 12 alkyl, 2-naphthyl-C 1 ~C 12 alkyl, etc. , preferably C 1 -C 12 alkoxyphenyl-C 1 -C 12 alkyl, C 1 -C 12 alkylphenyl-C 1 -C 12 alkyl.
芳基烷氧基的碳数通常为7~60左右,优选碳数7~48,作为其具体例,可以例示出苯基甲氧基、苯基乙氧基、苯基丁氧基、苯基戊氧基、苯基己氧基、苯基庚氧基、苯基辛氧基等苯基-C1~C12烷氧基、C1~C12烷氧基苯基-C1~C12烷氧基、C1~C12烷基苯基-C1~C12烷氧基、1-萘基-C1~C12烷氧基、2-萘基-C1~C12烷氧基等,优选C1~C12烷氧基苯基-C1~C12烷氧基、C1~C12烷基苯基-C1~C12烷氧基。 The carbon number of the arylalkoxy group is usually about 7 to 60, preferably 7 to 48 carbon atoms. Specific examples thereof include phenylmethoxy, phenylethoxy, phenylbutoxy, phenyl Pentyloxy, phenylhexyloxy, phenylheptyloxy, phenyloctyloxy, etc. phenyl-C 1 ~C 12 alkoxy, C 1 ~C 12 alkoxyphenyl-C 1 ~C 12 Alkoxy, C 1 ~C 12 alkylphenyl-C 1 ~C 12 alkoxy, 1-naphthyl-C 1 ~C 12 alkoxy, 2-naphthyl-C 1 ~C 12 alkoxy etc., preferably C 1 -C 12 alkoxyphenyl-C 1 -C 12 alkoxy, C 1 -C 12 alkylphenyl-C 1 -C 12 alkoxy.
芳烷基硫基的碳数通常为7~60左右,优选碳数7~48,作为其具体例,可以例示出苯基-C1~C12烷基硫基、C1~C12烷氧基苯基-C1~C12 烷基硫基、C1~C12烷基苯基-C1~C12烷基硫基、1-萘基-C1~C12烷基硫基、2-萘基-C1~C12烷基硫基等,优选C1~C12烷氧基苯基-C1~C12 烷基硫基、C1~C12烷基苯基-C1~C12烷基硫基。 The carbon number of the aralkylthio group is usually about 7 to 60, preferably 7 to 48, and specific examples thereof include phenyl-C 1 -C 12 alkylthio, C 1 -C 12 alkoxy phenyl-C 1 ~C 12 alkylthio, C 1 ~C 12 alkylphenyl-C 1 ~C 12 alkylthio, 1-naphthyl-C 1 ~C 12 alkylthio, 2 -Naphthyl-C 1 ~C 12 alkylthio, etc., preferably C 1 ~C 12 alkoxyphenyl-C 1 ~C 12 alkylthio, C 1 ~C 12 alkylphenyl-C 1 ~ C 12 alkylthio.
芳基链烯基的碳数通常为8~60左右,作为其具体例,可以例示出苯 基-C2~C12链烯基、C1~C12烷氧基苯基-C2~C12链烯基、C1~C12烷基苯基-C2~C12链烯基、1-萘基-C2~C12链烯基、2-萘基-C2~C12链烯基等,优选C1~C12烷氧基苯基-C2~C12链烯基、C2~C12烷基苯基-C1~C12链烯基。 The carbon number of the arylalkenyl group is usually about 8 to 60, and specific examples thereof include phenyl-C 2 -C 12 alkenyl, C 1 -C 12 alkoxyphenyl-C 2 -C 12 alkenyl, C 1 ~C 12 alkylphenyl-C 2 ~C 12 alkenyl, 1-naphthyl-C 2 ~C 12 alkenyl, 2-naphthyl-C 2 ~C 12 alkenyl group, etc., preferably C 1 -C 12 alkoxyphenyl-C 2 -C 12 alkenyl, C 2 -C 12 alkylphenyl-C 1 -C 12 alkenyl.
芳基炔基的碳数通常为8~60左右,作为其具体例,可以例示出苯基~C2~C12炔基、C1~C12烷氧基苯基-C2~C12炔基、C1~C12烷基苯基-C2~C12炔基、1-萘基-C2~C12炔基、2-萘基-C2~C12炔基等,优选C1~C12烷氧基苯基-C2~C12炔基、C1~C12烷基苯基-C2~C12炔基。 The carbon number of the arylalkynyl group is usually about 8 to 60. Specific examples thereof include phenyl to C 2 to C 12 alkynyl, C 1 to C 12 alkoxyphenyl-C 2 to C 12 alkynyl C 1 -C 12 alkylphenyl-C 2 -C 12 alkynyl, 1-naphthyl-C 2 -C 12 alkynyl, 2-naphthyl-C 2 -C 12 alkynyl, etc., preferably C 1 -C 12 alkoxyphenyl-C 2 -C 12 alkynyl, C 1 -C 12 alkylphenyl-C 2 -C 12 alkynyl.
作为取代氨基,可以举出由选自烷基、芳基、芳烷基或1价杂环基中的1个或2个基团取代了的氨基,该烷基、芳基、芳烷基或1价的杂环基也可以取代基。对于取代氨基的碳数,不包括该取代基的碳数,通常为1~60左右,优选碳数为2~48。 As the substituted amino group, an amino group substituted by one or two groups selected from an alkyl group, an aryl group, an aralkyl group or a monovalent heterocyclic group, the alkyl group, an aryl group, an aralkyl group or A monovalent heterocyclic group may also be a substituent. The carbon number of the substituted amino group, not including the carbon number of the substituent, is usually about 1 to 60, preferably 2 to 48 carbons. the
具体来说,可以例示出甲基氨基、二甲基氨基、乙基氨基、二乙基氨基、丙基氨基、二丙基氨基、异丙基氨基、二异丙基氨基、丁基氨基、异丁基氨基、叔丁基氨基、戊基烷基、己基氨基、环己基氨基、庚基氨基、辛基氨基、2-乙基己基氨基、壬基氨基、癸基氨基、3,7-二甲基辛基氨基、月桂基氨基、环戊基氨基、二环戊基氨基、环己基氨基、二环己基氨基、吡咯烷基、哌啶基、双三氟甲基氨基苯基氨基、联苯基氨基、C1~C12烷氧基苯基氨基、二(C1~C12烷氧基苯基)氨基、二(C1~C12烷基苯基)氨基、1-萘基氨基、2-萘基氨基、五氟苯基氨基、吡啶基氨基、哒嗪基氨基、嘧啶基氨基、吡嗪基氨基、triazyl氨基苯基-C1~C12烷基氨基、C1~C12烷氧基苯基-C1~C12烷基氨基、C1~C12烷基苯基-C1~C12 烷基氨基、二(C1~C12烷氧基苯基-C1~C12烷基)氨基、二(C1~C12 烷基苯基-C1~C12烷基)氨基、1-萘基-C1~C12烷基氨基、2-萘基-C1~C12烷基氨基等。 Specifically, methylamino, dimethylamino, ethylamino, diethylamino, propylamino, dipropylamino, isopropylamino, diisopropylamino, butylamino, isopropylamino, Butylamino, tert-butylamino, pentylalkyl, hexylamino, cyclohexylamino, heptylamino, octylamino, 2-ethylhexylamino, nonylamino, decylamino, 3,7-dimethyl Octylamino, laurylamino, cyclopentylamino, dicyclopentylamino, cyclohexylamino, dicyclohexylamino, pyrrolidinyl, piperidinyl, bistrifluoromethylaminophenylamino, biphenyl Amino, C 1 -C 12 alkoxyphenylamino, di(C 1 -C 12 alkoxyphenyl)amino, di(C 1 -C 12 alkylphenyl)amino, 1-naphthylamino, 2 -Naphthylamino, pentafluorophenylamino, pyridylamino, pyridazinylamino, pyrimidinylamino, pyrazinylamino, triazylaminophenyl-C 1 ~C 12 alkylamino, C 1 ~C 12 alkoxy phenyl-C 1 ~C 12 alkylamino, C 1 ~C 12 alkylphenyl-C 1 ~C 12 alkylamino, di(C 1 ~C 12 alkoxyphenyl-C 1 ~C 12 Alkyl)amino, di(C 1 ~C 12 alkylphenyl-C 1 ~C 12 alkyl)amino, 1-naphthyl-C 1 ~C 12 alkylamino, 2-naphthyl-C 1 ~C 12 Alkylamino, etc.
作为取代甲硅烷基,可以举出由选自烷基、芳基、芳烷基或1价杂环基中的1、2或3个基团取代的甲硅烷基。取代甲硅烷基的碳数通常为1~60左右,优选碳数为3~48。而且,该烷基、芳基、芳烷基或1价的杂环基也可以具有取代基。 Examples of the substituted silyl group include silyl groups substituted with 1, 2 or 3 groups selected from an alkyl group, an aryl group, an aralkyl group or a monovalent heterocyclic group. The carbon number of the substituted silyl group is usually about 1 to 60, preferably 3 to 48 carbon atoms. Furthermore, the alkyl group, aryl group, aralkyl group or monovalent heterocyclic group may have a substituent. the
具体来说,可以例示出三甲基甲硅烷基、三乙基甲硅烷基、三丙基甲 硅烷基、三异丙基甲硅烷基、二甲基异丙基甲硅烷基、二乙基异丙基甲硅烷基、叔丁基甲硅烷基二甲基甲硅烷基、戊基二甲基甲硅烷基、己基二甲基甲硅烷基、庚基二甲基甲硅烷基、辛基二甲基甲硅烷基、2-乙基己基二甲基甲硅烷基、壬基二甲基甲硅烷基、癸基二甲基甲硅烷基、3,7-二甲基辛基-二甲基甲硅烷基、月桂基二甲基甲硅烷基、苯基-C1~C12烷基甲硅烷基、C1~C12烷氧基苯基-C1~C12烷基甲硅烷基、C1~C12烷基苯基-C1~C12烷基甲硅烷基、1-萘基-C1~C12烷基甲硅烷基、2-萘基-C1~C12烷基甲硅烷基、苯基-C1~C12烷基二甲基甲硅烷基、三苯基甲硅烷基、三-对二甲苯基甲硅烷基、三苄基甲硅烷基、联苯基甲基甲硅烷基、叔丁基联苯基甲硅烷基、二甲基苯基甲硅烷基等。 Specifically, trimethylsilyl, triethylsilyl, tripropylsilyl, triisopropylsilyl, dimethylisopropylsilyl, diethylisopropylsilyl, diethylsilyl Propylsilyl, tert-Butylsilyldimethylsilyl, Amyldimethylsilyl, Hexyldimethylsilyl, Heptyldimethylsilyl, Octyldimethylsilyl base, 2-ethylhexyldimethylsilyl, nonyldimethylsilyl, decyldimethylsilyl, 3,7-dimethyloctyl-dimethylsilyl, lauryl Dimethylsilyl group, phenyl-C 1 ~C 12 alkylsilyl group, C 1 ~C 12 alkoxyphenyl-C 1 ~C 12 alkylsilyl group, C 1 ~C 12 alkane phenyl-C 1 ~C 12 alkylsilyl, 1-naphthyl-C 1 ~C 12 alkylsilyl, 2-naphthyl-C 1 ~C 12 alkylsilyl, phenyl- C 1 ~C 12 alkyldimethylsilyl, triphenylsilyl, tri-p-xylylsilyl, tribenzylsilyl, biphenylmethylsilyl, tert-butyl Biphenylsilyl group, dimethylphenylsilyl group and the like.
酰基的碳数通常为2~20左右,优选碳数为2~18,作为其具体例,可以例示出乙酰基、丙酰基、丁酰基、异丁酰基、新戊酰基、苯甲酰基、三氟乙酰基、五氟苯甲酰基等。 The acyl group usually has about 2 to 20 carbon atoms, preferably 2 to 18 carbon atoms. Specific examples thereof include acetyl, propionyl, butyryl, isobutyryl, pivaloyl, benzoyl, trifluoro Acetyl, pentafluorobenzoyl, etc. the
酰氧基的碳数通常为2~20左右,优选碳数为2~18,作为其具体例,可以例示出乙酰氧基、丙酰氧基、丁酰氧基、异丁酰氧基、新戊酰氧基、苯甲酰氧基、三氟乙酰氧基、五氟苯甲酰氧基等。 The carbon number of the acyloxy group is usually about 2 to 20, preferably 2 to 18, and specific examples thereof include acetyloxy, propionyloxy, butyryloxy, isobutyryloxy, and neobutyryloxy. Valeryloxy, benzoyloxy, trifluoroacetoxy, pentafluorobenzoyloxy, etc. the
酰胺基的碳数通常为2~20左右,优选碳数为2~18,作为其具体例,可以例示出甲酰胺基、乙酰胺基、丙酰胺基、丁酰胺基、苯甲酰胺基、三氟乙酰胺基、五氟苯甲酰胺基、二甲酰胺基、二乙酰胺基、二丙酰胺基、二丁酰胺基、二苯甲酰胺基、二三氟乙酰胺基、二五氟苯甲酰胺基等。 The carbon number of the amide group is usually about 2 to 20, preferably 2 to 18, and specific examples thereof include formamide, acetamide, propionamide, butyramide, benzamide, trisamide, Fluoroacetamide, pentafluorobenzamide, diformamide, diacetamide, dipropionamide, dibutyramide, dibenzamide, bistrifluoroacetamide, dipentafluorobenzyl Amide, etc. the
作为酰亚胺基,可以举出从酰亚胺化合物中去除与其氮原子结合的氢原子而得的残基,碳数为4~20左右,具体来说可以例示出以下所示的基团等。 Examples of the imide group include residues obtained by removing a hydrogen atom bonded to the nitrogen atom of an imide compound, and have a carbon number of about 4 to 20. Specifically, the groups shown below are exemplified. . the
所谓1价杂环基是指从杂环化合物中去除了1个氢原子后剩下的原子团,碳数通常为4~60左右,优选为4~20。而且,在杂环基的碳数中,不包含取代基的碳数。这里所谓杂环化合物是指,具有环式构造的有机化合物当中构成环的元素不仅是碳原子,在环内还含有氧、硫、氮、磷、硼等杂原子的化合物。具体来说,可以给出下述构造。 The monovalent heterocyclic group refers to an atomic group remaining after removing one hydrogen atom from a heterocyclic compound, and the carbon number is usually about 4-60, preferably 4-20. In addition, the carbon number of the substituent is not included in the carbon number of the heterocyclic group. The term "heterocyclic compound" herein refers to an organic compound having a cyclic structure in which the elements constituting the ring are not only carbon atoms but also contain heteroatoms such as oxygen, sulfur, nitrogen, phosphorus, and boron in the ring. Specifically, the following configurations can be given. the
上述式中,R分别独立地表示氢原子、烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、氨基、取代氨基、甲硅烷基、取代甲硅烷基、卤原子、酰基、酰氧基、亚胺残基、酰胺基、酰亚胺基、1价杂环基、羧基、取代羧基或氰基。 In the above formula, R independently represent a hydrogen atom, an alkyl group, an alkoxy group, an alkylthio group, an aryl group, an aryloxy group, an arylthio group, an aralkyl group, an arylalkoxy group, an arylalkylthio group, Aryl alkenyl group, aryl alkynyl group, amino group, substituted amino group, silyl group, substituted silyl group, halogen atom, acyl group, acyloxy group, imine residue, amido group, imide group, monovalent hetero Cyclic, carboxyl, substituted carboxyl or cyano. the
其中,优选噻吩基、C1~C12烷基噻吩基、吡咯基、呋喃基、吡啶基、C1~C12烷基吡啶基、哌啶基、喹啉基、异喹啉基等,更优选噻吩基、C1~C12烷基噻吩基、吡啶基、C1~C12烷基吡啶基。 Among them, thienyl, C 1 ~C 12 alkylthienyl, pyrrolyl, furyl, pyridyl, C 1 ~C 12 alkylpyridyl, piperidyl, quinolinyl, isoquinolyl, etc. are preferred, and more Preferred are thienyl, C 1 -C 12 alkylthienyl, pyridyl, and C 1 -C 12 alkylpyridyl.
取代羧基是指由烷基、芳基、芳烷基或1价杂环基取代了的羧基,碳数通常为2~60左右,优选碳数为2~48,作为其具体例,可以举出甲氧 基羰基、乙氧基羰基、丙氧基羰基、异丙氧基羰基、丁氧基羰基、异丁氧基羰基、叔丁氧基羰基、戊氧基羰基、己氧基羰基、环己氧基羰基、庚氧基羰基、辛氧基羰基、2-乙基己氧基羰基、壬氧基羰基、癸氧基羰基、3,-二甲基辛氧基羰基、十二烷氧基羰基、三氟甲氧基羰基、五氟乙氧基羰基、全氟丁氧基羰基、全氟己氧基羰基、全氟辛氧基羰基、苯氧基羰基、萘氧基羰基、吡啶氧基羰基等。而且,该烷基、芳基、芳烷基或1价杂环基也可以具有取代基。在取代羧基的碳数中不包含该取代基的碳数。 The substituted carboxyl group refers to a carboxyl group substituted by an alkyl group, an aryl group, an aralkyl group or a monovalent heterocyclic group, and generally has about 2 to 60 carbon atoms, preferably 2 to 48 carbon atoms. Specific examples thereof include: Methoxycarbonyl, ethoxycarbonyl, propoxycarbonyl, isopropoxycarbonyl, butoxycarbonyl, isobutoxycarbonyl, tert-butoxycarbonyl, pentyloxycarbonyl, hexyloxycarbonyl, cyclohexyl Oxycarbonyl, Heptyloxycarbonyl, Octyloxycarbonyl, 2-Ethylhexyloxycarbonyl, Nonyloxycarbonyl, Decyloxycarbonyl, 3,-Dimethyloctyloxycarbonyl, Dodecyloxycarbonyl , trifluoromethoxycarbonyl, pentafluoroethoxycarbonyl, perfluorobutoxycarbonyl, perfluorohexyloxycarbonyl, perfluorooctyloxycarbonyl, phenoxycarbonyl, naphthyloxycarbonyl, pyridyloxycarbonyl wait. Furthermore, the alkyl group, aryl group, aralkyl group or monovalent heterocyclic group may have a substituent. The carbon number of the substituent is not included in the carbon number of the substituted carboxyl group. the
上述式(1)中,Z1、Z2、Z3、Z4及Z5分别独立,表示C-(Q)z或氮原子,Q表示取代基或氢原子,z表示0或1。 In the above formula (1), Z 1 , Z 2 , Z 3 , Z 4 and Z 5 each independently represent C-(Q)z or a nitrogen atom, Q represents a substituent or a hydrogen atom, and z represents 0 or 1.
作为Q的取代基,可以举出烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、取代氨基、取代甲硅烷基、氟原子、酰基、酰氧基、酰胺基、酰亚胺基、1价杂环基、羧基、取代羧基、氰基及硝基等,其定义、具体例等与上述的内容相同。 Examples of substituents for Q include alkyl, alkoxy, alkylthio, aryl, aryloxy, arylthio, aralkyl, arylalkoxy, arylalkylthio, aryl chain Alkenyl group, arylalkynyl group, substituted amino group, substituted silyl group, fluorine atom, acyl group, acyloxy group, amido group, imide group, monovalent heterocyclic group, carboxyl group, substituted carboxyl group, cyano group and nitro group, etc. , and its definition, specific examples, etc. are the same as those described above. the
A环与B环也可以共有构成各自的环的Z5以外的环的原子,A环、B环及C环的一个以上两个以下为非芳香族环。优选A环与B环除了Z5以外共有一个环的原子的情况。另外,优选非芳香族环为一个的情况。 The A ring and the B ring may share atoms of rings other than Z 5 constituting the respective rings, and one or more of the A ring, B ring, and C ring are non-aromatic rings. The case where the A ring and the B ring share one ring atom except Z5 is preferred. In addition, it is preferable that there is one non-aromatic ring.
另外,本发明的高分子化合物作为重复单元优选含有以下述式(1-1)~(1-3)表示的重复单元。 In addition, the polymer compound of the present invention preferably contains a repeating unit represented by the following formulas (1-1) to (1-3) as a repeating unit. the
式(1-1) 式(1-2) 式(1-3) Formula (1-1) Formula (1-2) Formula (1-3)
[式中,A环、B环及C环分别独立地表示也可以具有取代基的芳香族环或非芳香族环,Z1、Z2、Z3、Z4及Z5分别独立地表示C-(Q)z或氮原子,Q表示取代基或氢原子,z表示0或1,A环与B环也可以共有Z5以外的环的原子,另外,也可以是各环的取代基之间结合,再形成环,A环、B环、C环当中的不具有结合键(connecting bond)的环的一个以 上为非芳香族环。] [In the formula, Ring A, Ring B, and Ring C independently represent an aromatic ring or a non-aromatic ring that may have a substituent, and Z 1 , Z 2 , Z 3 , Z 4 , and Z 5 independently represent C -(Q)z or a nitrogen atom, Q represents a substituent or a hydrogen atom, z represents 0 or 1, ring A and ring B may share atoms other than Z 5 , and may be one of the substituents of each ring The rings are combined to form a ring, and one or more of the rings without a connecting bond among the A ring, B ring, and C ring are non-aromatic rings. ]
作为以式(1-1)表示的重复单元的具体例,可以举出 Specific examples of the repeating unit represented by formula (1-1) include
等及它们具有取代基的例子。 etc. and examples where they have substituents. the
作为以式(1-2)表示的重复单元的具体例,可以举出 Specific examples of the repeating unit represented by formula (1-2) include
等及它们具有取代基的例子。 etc. and examples where they have substituents. the
作为以式(1-3)表示的重复单元的具体例,可以举出 Specific examples of the repeating unit represented by formula (1-3) include
等及它们具有取代基的例子。 etc. and examples where they have substituents. the
由上述式(1-1)~(1-3)表示的重复单元当中,对于构成A环、B环及C环的原子(所谓「构成环的原子」是指形成环的骨架的原子)而言,除了碳原子以外,也可以包括氮、氧、硫、硅、硒等原子,但是从调整电荷输送性的观点考虑,最好构成A环、B环及C环的原子全都是碳原子。 Among the repeating units represented by the above formulas (1-1) to (1-3), for the atoms constituting the A ring, the B ring, and the C ring (the so-called "atoms constituting the ring" means the atoms forming the skeleton of the ring) In other words, atoms such as nitrogen, oxygen, sulfur, silicon, and selenium may be included in addition to carbon atoms, but from the viewpoint of adjusting charge transport properties, it is preferable that all the atoms constituting the A ring, B ring, and C ring are carbon atoms. the
从提高高分子化合物的溶解性、调整发光波长的观点、调整电荷输送性的观点考虑,最好A环、B环、C环的某个具有取代基。 From the viewpoint of improving the solubility of the polymer compound, adjusting the emission wavelength, and adjusting the charge transport property, it is preferable that any one of the A ring, B ring, and C ring has a substituent. the
另外,从电荷输送性的观点考虑,优选以上述式(1-1)、(1-2)表示的构造的重复单元,从合成的容易度的观点考虑,更优选以上述式(1-1)表示的构造。 In addition, from the viewpoint of charge transportability, the repeating unit of the structure represented by the above formulas (1-1) and (1-2) is preferable, and from the viewpoint of easiness of synthesis, it is more preferable to have the repeating units represented by the above formula (1-1) ) represents the structure. the
另外,上述式(1-1)更优选下述式(2-1)。 In addition, the above formula (1-1) is more preferably the following formula (2-1). the
式(2-1) Formula (2-1)
[式中,R1及R2分别独立地表示取代基,D环表示也可以具有取代基的非芳香族环,a表示0~2的整数,b表示0~3的整数,在R1及R2分别存在多个的情况下,它们既可以相同,也可以不同,也可以R1与R2相互结合而形成环。另外,也可以R1及/或R2与D环结合而形成环,Q及z表示与上述相同的意思。] [wherein, R 1 and R 2 independently represent a substituent, D ring represents a non-aromatic ring that may also have a substituent, a represents an integer of 0 to 2, b represents an integer of 0 to 3, and in R 1 and When a plurality of R 2 are present, they may be the same or different, and R 1 and R 2 may be combined to form a ring. In addition, R 1 and/or R 2 may combine with D ring to form a ring, and Q and z have the same meanings as above. ]
包括上述式(2-1)的重复单元优选以下述式(3-1)表示的重复单元。 The repeating unit including the above formula (2-1) is preferably a repeating unit represented by the following formula (3-1). the
式(3-1) Formula (3-1)
[式中,R1、R2、D环、Q、z、a及b表示与上述相同的意思。] [In the formula, R 1 , R 2 , D ring, Q, z, a, and b have the same meanings as above. ]
以式(3-1)表示的重复单元当中,从调整电荷输送性的观点考虑,更优选以下述式(4-1)、(4-2)、(4-3)及(4-4)表示的重复单元。 Among the repeating units represented by the formula (3-1), the following formulas (4-1), (4-2), (4-3) and (4-4) are more preferable from the viewpoint of adjusting the charge transport property. Represents the repeating unit. the
式(4-1) 式(4-2) Formula (4-1) Formula (4-2)
式(4-3) 式(4-4) Formula (4-3) Formula (4-4)
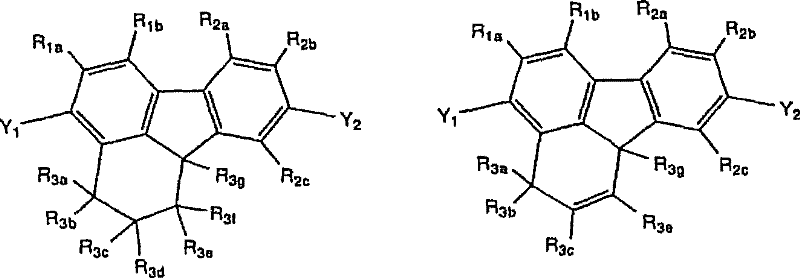
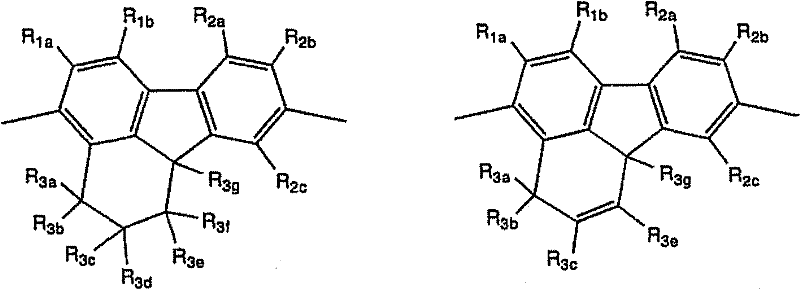
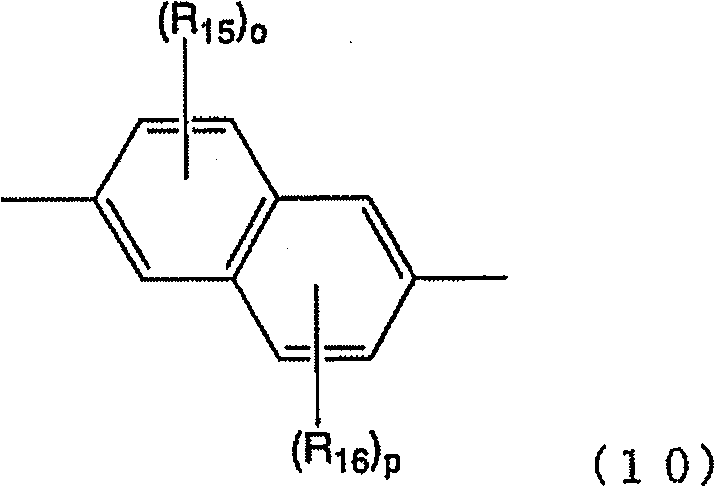
[式中,R1a、R1b、R2a~R2c及R3a~R3g表示取代基。式(4-1)~(4-3)中,也可以R2c与R3g相互结合而形成环。式(4-4)中,也可以R2c 与R3g相互结合而形成环。] [In the formula, R 1a , R 1b , R 2a to R 2c and R 3a to R 3g represent substituents. In formulas (4-1) to (4-3), R 2c and R 3g may combine with each other to form a ring. In formula (4-4), R 2c and R 3g may combine with each other to form a ring. ]
作为相互结合而形成的环,可以举出芳香族环、非芳香族环等,其具体例等与上述中所记载的相同。 Examples of the rings bonded to each other include aromatic rings, non-aromatic rings, and the like, and specific examples thereof are the same as those described above. the
而且,式(4-1)~(4-3)的单元被包含在式(3-1)中z=1的情况中,式(4-4)的单元被包含在式(3-1)中z=0的情况中。 And, the unit of formula (4-1)~(4-3) is included in the case of z=1 in formula (3-1), the unit of formula (4-4) is included in formula (3-1) In the case of z=0. the
作为以上述式(4-1)表示的重复单元的具体例,可以举出 Specific examples of the repeating unit represented by the above formula (4-1) include
作为以上述式(4-2)表示的重复单元的具体例,可以举出 Specific examples of the repeating unit represented by the above formula (4-2) include
作为以上述式(4-3)表示的重复单元的具体例,可以举出 Specific examples of the repeating unit represented by the above formula (4-3) include
作为以上述式(4-4)表示的重复单元的具体例,可以举出 Specific examples of the repeating unit represented by the above formula (4-4) include
式中,Me表示甲基,Et表示乙基。 In the formula, Me represents a methyl group, and Et represents an ethyl group. the
属于以上述式(1-1)、(1-2)、(1-3)、(2-1)、(3-1)、(4-1)、(4-2)、(4-3)及(4-4)表示的构造的重复单元的总数通常为本发明的高分子化合物所具有的全部重复单元的总数的1摩尔%以上100摩尔%以下,优选5摩尔%以上100摩尔%以下。 Belong to the above formula (1-1), (1-2), (1-3), (2-1), (3-1), (4-1), (4-2), (4-3 ) and (4-4) the total number of repeating units of the structure represented by the polymer compound of the present invention is usually 1 mol% to 100 mol% of the total number of all repeating units, preferably 5 mol% to 100 mol% . the
从改变发光波长的观点,提高发光效率的观点,提高耐热性的观点等方面考虑,本发明的高分子化合物优选除了以上述式(1-1)、(1-2)、(1-3)、(2-1)、(3-1)、(4-1)、(4-2)、(4-3)及(4-4)表示的重复单元以外,还含有1种以上除此以外的重复单元的共聚物。 From the viewpoint of changing the emission wavelength, the viewpoint of improving luminous efficiency, the viewpoint of improving heat resistance, etc., the polymer compound of the present invention is preferably in addition to the above-mentioned formula (1-1), (1-2), (1-3 ), (2-1), (3-1), (4-1), (4-2), (4-3) and (4-4) in addition to the repeating units represented by one or more of these Copolymers of other repeating units. the
作为以上述式(1-1)、(1-2)、(1-3)、(2-1)、(3-1)、(4-1)、(4-2)、(4-3)及(4-4)表示的重复单元以外的重复单元,优选以下述式(5)、式(6)、式(7)或式(8)表示的重复单元。 As above-mentioned formula (1-1), (1-2), (1-3), (2-1), (3-1), (4-1), (4-2), (4-3 ) and (4-4), the repeating unit is preferably a repeating unit represented by the following formula (5), formula (6), formula (7) or formula (8). the
-Ar1- (5) -Ar 1 - (5)
-(Ar2-X1)ff-Ar3- (6) -(Ar 2 -X 1 ) ff -Ar 3 - (6)
-Ar4-X2- (7) -Ar 4 -X 2 - (7)
-X3- (8) -X 3 - (8)
式中,Ar1、Ar2、Ar3及Ar4分别独立地表示亚芳基、2价杂环基或具有金属络合物构造的2价基团。X1、X2及X3分别独立地表示-CR9=CR10 -、-C≡C-、-N(R11)-或-(SiR12R13)m-。R9及R10分别独立地表示氢原子、烷基、芳基、1价杂环基、羧基、取代羧基或氰基。R11、R12 及R13分别独立地表示氢原子、烷基、芳基、1价杂环基、芳烷基或取代氨基。ff表示1或2的整数。m表示1~12的整数。在R9、R10、R11、R12 及R12分别存在多个的情况下,它们既可以相同,也可以不同。 In the formula, Ar 1 , Ar 2 , Ar 3 and Ar 4 each independently represent an arylene group, a divalent heterocyclic group or a divalent group having a metal complex structure. X 1 , X 2 and X 3 each independently represent -CR 9 =CR 10 -, -C≡C-, -N(R 11 )- or -(SiR 12 R 13 ) m -. R 9 and R 10 each independently represent a hydrogen atom, an alkyl group, an aryl group, a monovalent heterocyclic group, a carboxyl group, a substituted carboxyl group, or a cyano group. R 11 , R 12 and R 13 each independently represent a hydrogen atom, an alkyl group, an aryl group, a monovalent heterocyclic group, an aralkyl group or a substituted amino group. ff represents an integer of 1 or 2. m represents the integer of 1-12. When there are multiple R 9 , R 10 , R 11 , R 12 and R 12 , they may be the same or different.
这里所说的亚芳基是从芳香族烃中去除了2个氢原子后的原子团,包括具有稠环的原子团、以及2个以上独立的苯环或稠环直接或借助亚乙烯基等基团结合的原子团。亚芳基也可以具有取代基。 The arylene group mentioned here is an atomic group after removing two hydrogen atoms from an aromatic hydrocarbon, including an atomic group with a condensed ring, and two or more independent benzene rings or fused rings directly or through groups such as vinylidene groups bonded atoms. The arylene group may also have a substituent. the
作为取代基,可以举出烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、氨基、取代氨基、甲硅烷基、取代甲硅烷基、卤原子、酰基、酰氧基、亚胺残基、酰胺基、酰亚胺基、1价杂环基、羧基、取代羧基、氰基。 Examples of substituents include alkyl, alkoxy, alkylthio, aryl, aryloxy, arylthio, aralkyl, arylalkoxy, arylalkylthio, arylalkenyl , arylalkynyl group, amino group, substituted amino group, silyl group, substituted silyl group, halogen atom, acyl group, acyloxy group, imine residue, amide group, imide group, monovalent heterocyclic group, carboxyl group, Substitute carboxyl, cyano. the
亚芳基中的除去取代基后的部分的碳数通常为6~60左右,优选为6~20。另外,亚芳基的包括取代基的总碳数通常为6~100左右。 The carbon number of the part except the substituent in an arylene group is about 6-60 normally, Preferably it is 6-20. In addition, the total carbon number of the arylene group including the substituent is usually about 6 to 100. the
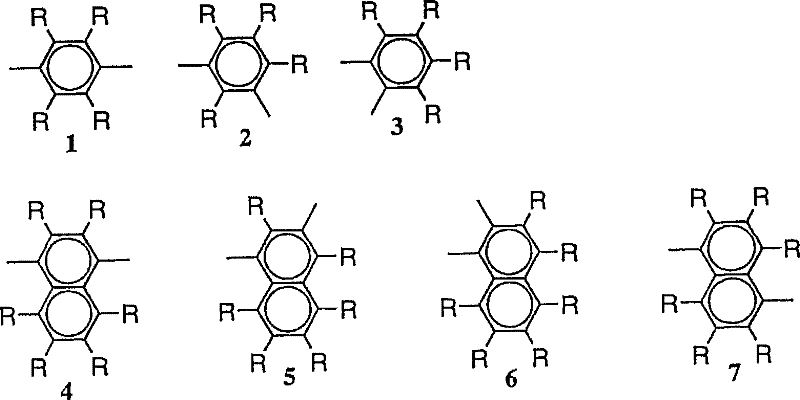
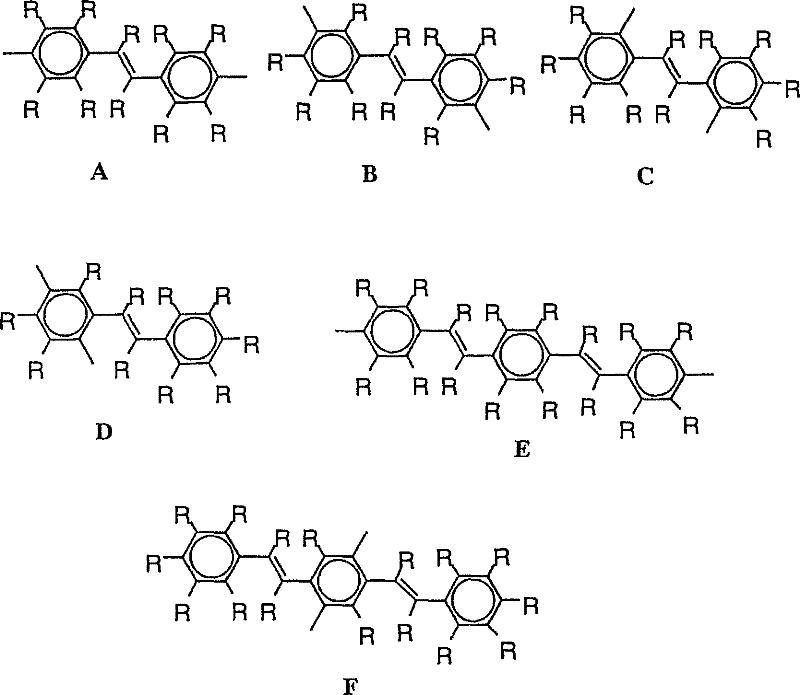
作为亚芳基,可以例示出亚苯基(例如下图的式1~3)、萘二基(下图的式4~13)、蒽二基(下图的式14~19)、联苯二基(下图的式20~25)、芴二基(下图的式36~38)、三联苯二基(下图的式26~28)、稠环化合 物基(下图的式29~35)、苯乙烯二基(下图的式A~D)、二苯乙烯二基(下图的式E、F)等。其中优选亚苯基、亚联苯基、芴二基、茋二基。 Examples of the arylene group include phenylene (for example, formulas 1 to 3 in the figure below), naphthalenediyl (formulas 4 to 13 in the figure below), anthracene diyl groups (formulas 14 to 19 in the figure below), biphenyl Diyl (formula 20-25 in the figure below), fluorene diyl (formula 36-38 in the figure below), terphenyldiyl (formula 26-28 in the figure below), fused ring compound base (formula 29 in the figure below) ~35), styrene diyl groups (formulas A to D in the figure below), distyryl diyl groups (formulas E and F in the figure below), etc. Among them, phenylene, biphenylene, fluorenediyl, and stilylenediyl are preferable. the
另外,Ar1、Ar2、Ar3及Ar4中的所谓2价杂环基是指从杂环化合物中去除了2个氢原子后剩余的原子团,该基团也可以具有取代基。这里所说的杂环化合物是指,具有环式构造的有机化合物当中构成环的元素不仅是碳原子,在环内还包括氧、硫、氮、磷、硼、砷等杂原子的化合物。2价的杂环基当中,优选芳香族杂环基。 In addition, the divalent heterocyclic group in Ar 1 , Ar 2 , Ar 3 and Ar 4 refers to an atomic group remaining after removing two hydrogen atoms from a heterocyclic compound, and this group may have a substituent. The term "heterocyclic compound" as used herein refers to an organic compound having a ring structure in which the elements constituting the ring are not only carbon atoms, but also heteroatoms such as oxygen, sulfur, nitrogen, phosphorus, boron, and arsenic are included in the ring. Among divalent heterocyclic groups, aromatic heterocyclic groups are preferred.
作为取代基,可以举出烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、氨基、取代氨基、甲硅烷基、取代甲硅烷基、卤原子、酰基、酰氧基、亚胺残基、酰胺基、酰亚胺基、1价杂环基、羧基、取代羧基、氰基。 Examples of substituents include alkyl, alkoxy, alkylthio, aryl, aryloxy, arylthio, aralkyl, arylalkoxy, arylalkylthio, arylalkenyl , arylalkynyl group, amino group, substituted amino group, silyl group, substituted silyl group, halogen atom, acyl group, acyloxy group, imine residue, amide group, imide group, monovalent heterocyclic group, carboxyl group, Substitute carboxyl, cyano. the
2价杂环基中除去取代基后的部分的碳数通常为3~60左右。另外,2价杂环基的包括取代基的总碳数通常为3~100左右。 The number of carbon atoms of the part excluding the substituent in the divalent heterocyclic group is usually about 3 to 60. In addition, the total carbon number including the substituent of the divalent heterocyclic group is usually about 3-100. the
作为2价杂环基,例如可以举出如下的基团。 As a divalent heterocyclic group, the following groups are mentioned, for example. the
作为杂原子,包括氮的2价杂环基:吡啶二基(下图的式39~44)、二氮杂亚苯基(下图的式45~48)、喹啉二基(下图的式49~63)、喹喔 啉二基(下图的式64~68)、吖啶二基(下图的式69~72)、联吡啶二基(下图的式73~75)、菲绕啉二基(下图的式76~78)等。 As heteroatoms, divalent heterocyclic groups including nitrogen: pyridinediyl (formula 39-44 in the figure below), diazaphenylene (formula 45-48 in the figure below), quinoline diyl (formula 45-48 in the figure below), quinoline diyl ( Formula 49~63), quinoxaline dibase (formula 64~68 in the figure below), acridine dibase (formula 69~72 in the figure below), bipyridine dibase (formula 73~75 in the figure below), phenanthrene Arborindiyl (formula 76-78 in the figure below) and the like. the
作为杂原子含有硅、氮、硒等的具有芴构造的基团(下图的式79~93)。 A group having a fluorene structure containing silicon, nitrogen, selenium, etc. as a heteroatom (formulas 79 to 93 in the figure below). the
作为杂原子含有硅、氮、硫、硒等的五元环杂环基:可以举出(下图的式94~98)。 Five-membered ring heterocyclic groups containing silicon, nitrogen, sulfur, selenium, etc. as heteroatoms: Examples include (formulas 94 to 98 in the diagram below). the
作为杂原子含有硅、氮、硒等的五元环稠合杂环基:可以举出(下图的式99~100、102~110)。 Five-membered ring condensed heterocyclic groups containing silicon, nitrogen, selenium, etc. as heteroatoms: Examples include (formulas 99 to 100, 102 to 110 in the diagram below). the
作为杂原子含有硅、氮、硫、硒等的五元环杂环基且在其杂原子的α位结合而成为二聚体或低聚物的基团:可以举出(下图的式111~112)。 A five-membered ring heterocyclic group containing silicon, nitrogen, sulfur, selenium, etc. as a heteroatom, which is combined at the α position of the heteroatom to form a dimer or oligomer: can be listed (formula 111 in the figure below) ~112). the
作为杂原子含有硅、氮、硫、硒等的五元环杂环基且在其杂原子的α位与苯基结合的基团:可以举出(下图的式113~119)。 A five-membered ring heterocyclic group containing silicon, nitrogen, sulfur, selenium, etc. as a heteroatom, and a group bonded to a phenyl group at the α-position of the heteroatom: (Formulas 113 to 119 in the diagram below). the
在作为杂原子含有氧、氮、硫等的五元环杂环基上苯基、呋喃基、噻吩基取代了的基团:可以举出(下图的式120~125)。 Groups substituted with phenyl, furyl, and thienyl groups on five-membered ring heterocyclic groups containing oxygen, nitrogen, sulfur, etc. as heteroatoms: Examples include (formulas 120 to 125 in the diagram below). the
另外,所谓Ar1、Ar2、Ar3及Ar4中的具有金属络合物构造的2价基团是指从具有有机配位体的金属络合物的有机配位体中去除2个氢原子后剩余的2价基团。 In addition, the divalent group having a metal complex structure among Ar 1 , Ar 2 , Ar 3 and Ar 4 means that two hydrogens are removed from the organic ligand of the metal complex having an organic ligand. The divalent group remaining after the atom.
该有机配位体的碳数通常为4~60左右,例如可以举出8-羟基喹啉及其衍生物、苯并喹啉及其衍生物、2-苯基吡啶及其衍生物、2-苯基苯并噻唑及其衍生物、2-苯基苯并噁唑及其衍生物、卟啉及其衍生物等。 The carbon number of the organic ligand is usually about 4 to 60, for example, 8-hydroxyquinoline and its derivatives, benzoquinoline and its derivatives, 2-phenylpyridine and its derivatives, 2- Phenylbenzothiazole and its derivatives, 2-phenylbenzoxazole and its derivatives, porphyrin and its derivatives, etc. the
另外,作为该络合物的中心金属,例如可以举出铝、锌、铍、铱、铂、金、铕、铽等。 In addition, examples of the central metal of the complex include aluminum, zinc, beryllium, iridium, platinum, gold, europium, terbium, and the like. the
作为具有有机配位体的金属络合物,可以举出低分子的荧光材料、作为磷光材料公知的金属络合物、三重态发光络合物等。 Examples of the metal complex having an organic ligand include low-molecular fluorescent materials, metal complexes known as phosphorescent materials, triplet light-emitting complexes, and the like. the
作为具有金属络合物构造的2价基团,具体来说,可以例示出以下的(126~132)。 Specifically, the following (126-132) can be illustrated as a divalent group which has a metal complex structure. the
上述的式1~132中,R分别独立地表示氢原子、烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、氨基、取代氨基、甲硅烷基、取代甲硅烷基、卤原子、酰基、酰氧基、亚胺残基、酰胺基、酰亚胺基、1价的杂环基、羧基、取代羧基或氰基。另外,式1~132的基团所具有的碳原子也可以取代为氮原子、氧原子或硫原子,氢原子也可以取代为氟原子。 In the above formulas 1 to 132, R independently represent a hydrogen atom, an alkyl group, an alkoxy group, an alkylthio group, an aryl group, an aryloxy group, an arylthio group, an aralkyl group, an arylalkoxy group, an aryl group Alkylthio group, arylalkenyl group, arylalkynyl group, amino group, substituted amino group, silyl group, substituted silyl group, halogen atom, acyl group, acyloxy group, imide residue, amido group, imide group , a monovalent heterocyclic group, a carboxyl group, a substituted carboxyl group or a cyano group. In addition, the carbon atoms contained in the groups of formulas 1 to 132 may be substituted by nitrogen atoms, oxygen atoms or sulfur atoms, and the hydrogen atoms may be substituted by fluorine atoms. the
另外,以上述式(5)、(6)、(7)、(8)表示的重复单元当中,优选以下述式(9)、式(10)、式(11)、式(12)、式(13)或式(14)表示的重复单元。 In addition, among the repeating units represented by the above-mentioned formulas (5), (6), (7), and (8), the following formulas (9), (10), (11), (12), and (13) or a repeating unit represented by formula (14). the
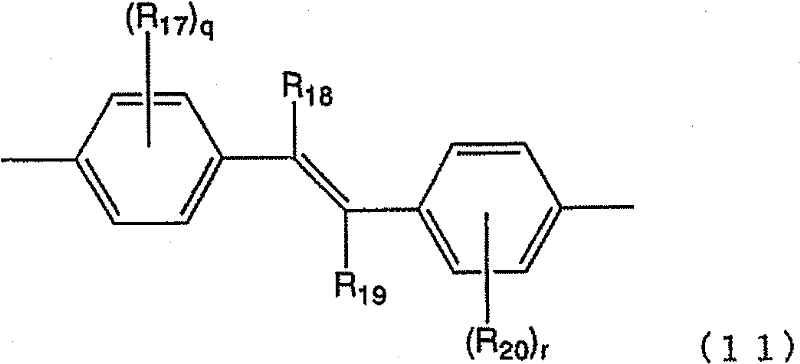
[式中,R14表示烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、氨基、取代氨基、甲硅烷基、取代甲硅烷基、卤原子、酰基、酰氧基、亚胺残基、酰胺基、酰亚胺基、1价的杂环基、羧基、取代羧基或氰基。n表示0~4的整数。在存在多个R14的情况下,它们既可以相同也可以不同。] [ wherein , R represents alkyl, alkoxy, alkylthio, aryl, aryloxy, arylthio, aralkyl, arylalkoxy, arylalkylthio, arylalkenyl , arylalkynyl group, amino group, substituted amino group, silyl group, substituted silyl group, halogen atom, acyl group, acyloxy group, imine residue, amide group, imide group, monovalent heterocyclic group, carboxyl group , substituted carboxyl or cyano. n represents an integer of 0-4. When a plurality of R 14 are present, they may be the same or different. ]
[式中,R15及R16分别独立地表示烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、氨基、取代氨基、甲硅烷基、取代甲硅烷基、卤原子、酰基、酰氧基、亚胺残基、酰胺基、酰亚胺基、1价的杂环基、羧基、取代羧基或氰基。o及p分别独立地表示0~3的整数。在存在多个R15及R16的情况下,它们既可以相同也可以不同。] [wherein, R 15 and R 16 independently represent alkyl, alkoxy, alkylthio, aryl, aryloxy, arylthio, aralkyl, arylalkoxy, arylalkylthio , aryl alkenyl, aryl alkynyl, amino, substituted amino, silyl, substituted silyl, halogen atom, acyl, acyloxy, imine residue, amido, imide, monovalent Heterocyclyl, carboxyl, substituted carboxyl or cyano. o and p each independently represent the integer of 0-3. When there are a plurality of R 15 and R 16 , they may be the same or different. ]
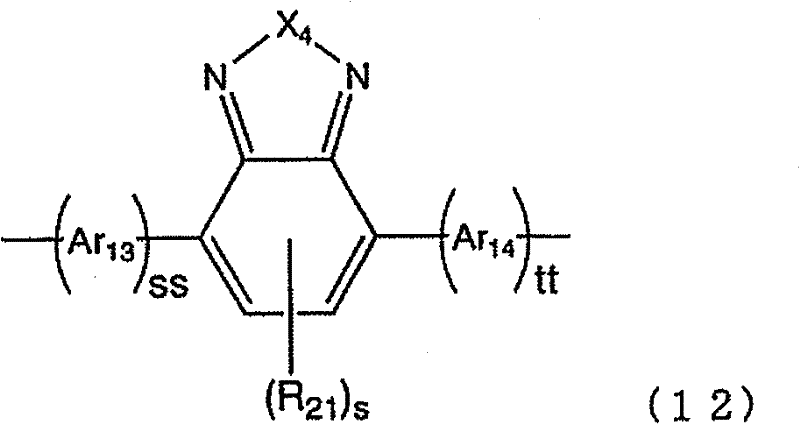
[式中,R17及R20分别独立地表示烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、氨基、取代氨基、甲硅烷基、取代甲硅烷基、卤原子、酰基、酰氧基、亚胺残基、酰胺基、酰亚胺基、1价的杂环基、羧基、取代羧基或氰基。q及r分别独立地表示0~4的整数。R18及R19分别独立地表示氢原子、烷基、芳基、1价的杂环基、羧基、取代羧基或氰基。在存在多个R17及R20 的情况下,它们既可以相同也可以不同。] [wherein, R 17 and R 20 independently represent alkyl, alkoxy, alkylthio, aryl, aryloxy, arylthio, aralkyl, arylalkoxy, arylalkylthio , aryl alkenyl, aryl alkynyl, amino, substituted amino, silyl, substituted silyl, halogen atom, acyl, acyloxy, imine residue, amido, imide, monovalent Heterocyclyl, carboxyl, substituted carboxyl or cyano. q and r each independently represent the integer of 0-4. R 18 and R 19 each independently represent a hydrogen atom, an alkyl group, an aryl group, a monovalent heterocyclic group, a carboxyl group, a substituted carboxyl group, or a cyano group. When there are a plurality of R 17 and R 20 , they may be the same or different. ]
[式中,R21表示烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、氨基、取代氨基、甲硅烷基、取代甲硅烷基、卤原子、酰基、酰氧基、亚胺残基、酰胺基、酰亚胺基、1价的杂环基、羧基、取代羧基或氰基。s表示0~2的整数。Ar13及Ar14分别独立地表示亚芳基、2价的杂环基或具有金属络合物构造的2价基团。ss及tt分别独立地表示0或1。X4表示O、S、SO、SO2、Se或Te。在存在多个R21的情况下,它们既可以相同也可以不同。] [ wherein , R represents alkyl, alkoxy, alkylthio, aryl, aryloxy, arylthio, aralkyl, arylalkoxy, arylalkylthio, arylalkenyl , arylalkynyl group, amino group, substituted amino group, silyl group, substituted silyl group, halogen atom, acyl group, acyloxy group, imine residue, amide group, imide group, monovalent heterocyclic group, carboxyl group , substituted carboxyl or cyano. s represents an integer of 0-2. Ar 13 and Ar 14 each independently represent an arylene group, a divalent heterocyclic group, or a divalent group having a metal complex structure. ss and tt each independently represent 0 or 1. X 4 represents O, S, SO, SO 2 , Se or Te. When a plurality of R 21 are present, they may be the same or different. ]
[式中,R22及R25分别独立地表示烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、氨基、取代氨基、甲硅烷基、取代甲硅烷基、卤原子、酰基、酰氧基、亚胺残基、酰胺基、酰亚胺基、1价的杂环基、羧基、取代羧基或氰基。t及u分别独立地表示0~4的整数。X5表示O、S、SO、SO2、Se、Te、N-R24或SiR25R26。X6及X7分别独立地表示N或C-R27。R24、R25、R26 及R27分别独立地表示氢原子、烷基、芳基、芳烷基或1价杂环基。在存在多个R22、R23及R27的情况下,它们既可以相同也可以不同。] [wherein, R 22 and R 25 independently represent alkyl, alkoxy, alkylthio, aryl, aryloxy, arylthio, aralkyl, arylalkoxy, arylalkylthio , aryl alkenyl, aryl alkynyl, amino, substituted amino, silyl, substituted silyl, halogen atom, acyl, acyloxy, imine residue, amido, imide, monovalent Heterocyclyl, carboxyl, substituted carboxyl or cyano. t and u each independently represent the integer of 0-4. X 5 represents O, S, SO, SO 2 , Se, Te, NR 24 or SiR 25 R 26 . X 6 and X 7 each independently represent N or CR 27 . R 24 , R 25 , R 26 and R 27 each independently represent a hydrogen atom, an alkyl group, an aryl group, an aralkyl group or a monovalent heterocyclic group. When there are a plurality of R 22 , R 23 and R 27 , they may be the same or different. ]
作为以式(11)表示的重复单元的中央的五元环的例子,可以举出噻二唑、噁二唑、三唑、噻吩、呋喃、silole等。 Examples of the central five-membered ring of the repeating unit represented by formula (11) include thiadiazole, oxadiazole, triazole, thiophene, furan, silole and the like. the
[式中,R28及R33分别独立地表示烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、氨基、取代氨基、甲硅烷基、取代甲硅烷基、卤原子、酰基、酰氧基、亚胺残基、酰胺基、酰亚胺基、1价的杂环基、羧基、取代羧基或氰基。v及w分别独立地表示0~4的整数。R29、R30、R31及R36分别独立地表示氢原子、烷基、芳基、1价杂环基、羧基、取代羧基或氰基。Ar5表示亚芳基、2价的杂环基或具有金属络合物构造的2价基团。在存在多个R28及R33的情况下,它们既可以相同也可以不同。] [wherein, R 28 and R 33 independently represent alkyl, alkoxy, alkylthio, aryl, aryloxy, arylthio, aralkyl, arylalkoxy, arylalkylthio , aryl alkenyl, aryl alkynyl, amino, substituted amino, silyl, substituted silyl, halogen atom, acyl, acyloxy, imine residue, amido, imide, monovalent Heterocyclyl, carboxyl, substituted carboxyl or cyano. v and w each independently represent the integer of 0-4. R 29 , R 30 , R 31 and R 36 each independently represent a hydrogen atom, an alkyl group, an aryl group, a monovalent heterocyclic group, a carboxyl group, a substituted carboxyl group or a cyano group. Ar 5 represents an arylene group, a divalent heterocyclic group, or a divalent group having a metal complex structure. When a plurality of R 28 and R 33 exist, they may be the same or different. ]
另外,以上述式(6)表示的重复单元当中,从改变发光波长的观点、提高发光效率的观点、提高耐热性的观点考虑,优选以下述式(15)表示的重复单元。 In addition, among the repeating units represented by the above formula (6), the repeating unit represented by the following formula (15) is preferable from the viewpoint of changing the emission wavelength, improving the luminous efficiency, and improving heat resistance. the
[式中,Ar6、Ar7、Ar8及Ar9分别独立地表示亚芳基或2价的杂环基。Ar10、Ar11及Ar12分别独立地表示芳基或1价的杂环基。Ar6、Ar7、Ar8、Ar9及Ar10也可以具有取代基。x及y分别独立地表示0或1。0≤x+y≤1。] [In the formula, Ar 6 , Ar 7 , Ar 8 and Ar 9 each independently represent an arylene group or a divalent heterocyclic group. Ar 10 , Ar 11 and Ar 12 each independently represent an aryl group or a monovalent heterocyclic group. Ar 6 , Ar 7 , Ar 8 , Ar 9 and Ar 10 may also have a substituent. x and y each independently represent 0 or 1. 0≦x+y≦1. ]
作为以上述式(15)表示的重复单元的具体例,可以举出以下面的(式133~140)表示的例子。 Specific examples of the repeating unit represented by the above formula (15) include those represented by the following (Formulas 133 to 140). the
上述式中,R与上述式1~132的内容相同。 In the above formula, R is the same as in the above formulas 1 to 132. the
为了提高向溶剂中的溶解性,优选具有一个以上氢原子以外的取代基,另外,含有取代基的重复单元的形状的对称性越少越好。 In order to improve the solubility in solvents, it is preferable to have one or more substituents other than hydrogen atoms, and the less symmetrical the shape of the repeating unit containing the substituents, the better. the
上述式中,在R含有烷基的取代基中,为了提高高分子化合物向溶剂中的溶解性,优选含有一个以上环状或具有支链的烷基。 In the above formula, in the substituent R containing an alkyl group, it is preferable to contain one or more cyclic or branched alkyl groups in order to improve the solubility of the polymer compound in a solvent. the
另外,在上述式中R在其一部分中含有芳基或杂环基的情况下,它们也可以还具有一个以上的取代基。 In addition, when R in the above formula contains an aryl group or a heterocyclic group in a part thereof, they may further have one or more substituents. the
以上述式(15)表示的重复单元中,从调节发光波长的观点、元件特性等观点考虑,最好Ar6、Ar7、Ar8及Ar9分别独立地表示亚芳基,Ar10、Ar11及Ar12分别独立地表示芳基。 Among the repeating units represented by the above formula (15), it is preferable that Ar 6 , Ar 7 , Ar 8 and Ar 9 each independently represent an arylene group, and Ar 10 , Ar 11 and Ar 12 each independently represent an aryl group.
作为Ar6、Ar7、Ar8,优选分别独立地为未取代的亚苯基、未取代的联苯基、未取代的亚萘基、未取代的蒽二基的情况。 Ar 6 , Ar 7 , and Ar 8 are each independently preferably unsubstituted phenylene, unsubstituted biphenyl, unsubstituted naphthylene, or unsubstituted anthracendiyl.
作为Ar10、Ar11及Ar12,从溶解性、发光效率、稳定性的观点考虑,优选分别独立地为具有3个以上取代基的芳基,更优选Ar10、Ar11及Ar12 是具有3个以上取代基的苯基、具有3个以上取代基的萘基或具有3个以上取代基的蒽基,进一步优选Ar10、Ar11及Ar12为具有3个以上取代基的苯基。 Ar 10 , Ar 11 , and Ar 12 are each independently preferably an aryl group having three or more substituents from the viewpoint of solubility, luminous efficiency, and stability, and more preferably Ar 10 , Ar 11 , and Ar 12 have A phenyl group having three or more substituents, a naphthyl group having three or more substituents, or an anthracenyl group having three or more substituents, and Ar 10 , Ar 11 and Ar 12 are more preferably phenyl groups having three or more substituents.
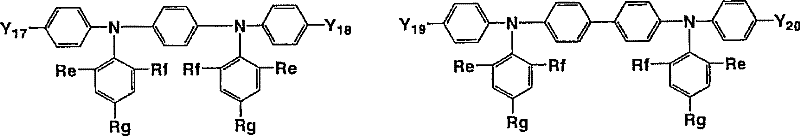
其中,Ar10、Ar11及Ar12优选分别独立地为下述式(15-1)。 Among them, Ar 10 , Ar 11 and Ar 12 are each independently preferably represented by the following formula (15-1).
[式中,Re、Rf及Rg分别独立地表示烷基、烷氧基、烷硫基、芳基、芳氧基、芳硫基、芳烷基、芳基烷氧基、芳基烷硫基、芳基链烯基、芳基炔基、氨基、取代氨基、甲硅烷基、取代甲硅烷基、甲硅烷氧基、取代甲硅烷氧基、1价杂环基或卤原子。] [wherein, Re, Rf and Rg independently represent alkyl, alkoxy, alkylthio, aryl, aryloxy, arylthio, aralkyl, arylalkoxy, arylalkylthio , arylalkenyl, arylalkynyl, amino, substituted amino, silyl, substituted silyl, siloxy, substituted siloxy, monovalent heterocyclic group or halogen atom. ]
以上述式(15)表示的重复单元中,Ar7优选为下述式(15-2)或(15-3)。 Among the repeating units represented by the above formula (15), Ar 7 is preferably the following formula (15-2) or (15-3).
[这里,以(15-2)、(15-3)表示的构造中所含的苯环也可以分别独立地具有1个以上4个以下的取代基。这些取代基既可以相同,也可以不同。另外,也可以多个取代基连结而形成环。另外,也可以与该苯环相邻地结合其他的芳香族烃环或杂环。] [Here, the benzene rings contained in the structures represented by (15-2) and (15-3) may each independently have one to four substituents. These substituents may be the same or different. In addition, a plurality of substituents may be linked to form a ring. In addition, another aromatic hydrocarbon ring or heterocyclic ring may be bonded adjacent to the benzene ring. ]
作为以上述式(15)表示的重复单元,作为特别优选的具体例,可以举出以下面的(式141~142)表示的例子。 As the repeating unit represented by the above-mentioned formula (15), examples represented by the following (formulas 141 to 142) can be given as particularly preferable specific examples. the
[式中,Re~Rg如上述所示。] [wherein, Re to Rg are as described above. ]
上述式中,Re~Rg与上述式1~132的内容相同。为了提高向溶剂中的溶解性,优选具有一个以上氢原子以外的取代基,另外,含有取代基的重复单元的形状的对称性越少越好。 In the above formulas, Re to Rg are the same as those in the above formulas 1 to 132. In order to improve the solubility in solvents, it is preferable to have one or more substituents other than hydrogen atoms, and the less symmetrical the shape of the repeating unit containing the substituents, the better. the
上述式中R含有烷基链的取代基中,为了提高高分子化合物向溶剂中的溶解性,优选含有1个以上环状或具有支链的烷基链。 Among the substituents in which R contains an alkyl chain in the above formula, it is preferable to contain one or more cyclic or branched alkyl chains in order to improve the solubility of the polymer compound in a solvent. the
另外,在上述式中R在其一部分含有芳基或杂环基的情况下,它们也可以还具有1个以上的取代基。 In addition, in the above formula, when R partially contains an aryl group or a heterocyclic group, these may further have one or more substituents. the
而且,在不损害发光特性或电荷输送特性的范围中,本发明的高分子 化合物也可以含有以上述式(1-1)、(1-2)、(1-3)及式(5)~式(15)表示的重复单元以外的重复单元。另外,这些重复单元或其他的重复单元也可以由非共轭的单元连结,还可以在重复单元中含有这些非共轭部分。作为结合构造,可以例示出以下所示的构造及将以下所示的构造当中的2个以上组合了的构造等。这里,R是从与上述的取代基相同的取代基中选择的基团,Ar表示碳数为6~60个的烃基。 In addition, the polymer compound of the present invention may also contain the above-mentioned formula (1-1), (1-2), (1-3) and formula (5) ~ A repeating unit other than the repeating unit represented by formula (15). In addition, these repeating units or other repeating units may be connected by non-conjugated units, and these non-conjugated moieties may be contained in the repeating units. As a combination structure, the structure shown below, the structure which combined two or more of the structures shown below, etc. can be illustrated. Here, R is a group selected from the same substituents as those described above, and Ar represents a hydrocarbon group having 6 to 60 carbon atoms. the
本发明的高分子化合物当中,优选仅由上述式(1-1)表示的重复单元构成的化合物;及/或仅由(1-2)表示的重复单元构成的化合物;及/或仅由(1-3)表示的重复单元构成的化合物,实质上优选由上述式(1-1)及/或(1-2)及/或(1-3),与以上述式(5)~(15)表示的重复单元的1个以上构成的化合物。 Among the polymer compounds of the present invention, preferred are compounds consisting only of repeating units represented by the above formula (1-1); and/or compounds consisting only of repeating units represented by (1-2); and/or compounds consisting only of ( 1-3) The compound composed of the repeating unit represented by the above-mentioned formula (1-1) and/or (1-2) and/or (1-3) is preferably substantially combined with the above-mentioned formula (5) to (15 ) compound consisting of one or more repeating units represented by ). the
另外,本发明的高分子化合物既可以是无规、嵌段或接枝共聚物,也可以是具有它们的中间的构造的高分子,例如是带有嵌段性的无规共聚物。从获得荧光或磷光的量子收率高的高分子发光体的观点考虑,与完全的无规共聚物相比,更优选带有嵌段性的无规共聚物或嵌段或者接枝共聚物。也包括在主链中具有分枝而具有3个以上末端部的情况或枝状物。 In addition, the polymer compound of the present invention may be a random, block or graft copolymer, or may be a polymer having an intermediate structure thereof, for example, a random copolymer having a block property. From the viewpoint of obtaining a high-molecular light-emitting body with a high quantum yield of fluorescence or phosphorescence, a block random copolymer or a block or graft copolymer is more preferable than a complete random copolymer. It also includes the case where the main chain has a branch and has three or more terminal parts, or a dendritic substance. the
另外,本发明的高分子化合物的末端基当原封不动地残留聚合活性基时,由于有可能降低制成元件后的发光特性或寿命,因此可以用稳定的基团将其保护。优选具有与主链的共轭构造连续的共轭键的构造,例如可以 例示出借助碳-碳键与芳基或杂环基结合的构造。具体来说,可以例示出特开平9-45478号公报的化10中记载的取代基等。 In addition, when the terminal group of the polymer compound of the present invention remains as it is, the luminescent characteristics or lifetime of the device may be reduced, so it can be protected with a stable group. A structure having a conjugated bond continuous to the conjugated structure of the main chain is preferred, for example, a structure in which an aryl group or a heterocyclic group is bonded via a carbon-carbon bond can be exemplified. Specifically, substituents described in Formula 10 of JP-A-9-45478 and the like can be exemplified. the
本发明的高分子化合物中,最好其分子链末端的至少一方是选自1价杂环基、1价芳香族胺基、由杂环配位金属络合物衍生的1价基团及芳基中的芳香族末端基。该芳香族末端基既可以是1种,也可以是2种以上。从荧光特性或元件特性的观点考虑,最好实质上不存在芳香族末端基以外的末端基。这里,所谓分子链末端是指,利用本发明的制造方法在存在于高分子化合物的末端的芳香族末端基;属于聚合中所用的单体的离去基,且在聚合时不离去而存在于高分子化合物的末端的离去基;存在于高分子化合物的末端的来源于单体的离去基虽然脱离,但是不结合芳香族末端基而结合的氢原子。这些分子链末端当中,对于属于聚合中所用的单体的离去基,且在聚合时不离去而存在于高分子化合物的末端的离去基,例如在作为原料使用具有卤原子的单体制造本发明的高分子化合物等情况下,当卤素残留于高分子化合物末端时,则会有荧光特性等降低的倾向,因此最好在末端实质上不残留单体的离去基。 In the polymer compound of the present invention, preferably at least one of its molecular chain ends is selected from monovalent heterocyclic groups, monovalent aromatic amine groups, monovalent groups derived from heterocyclic coordination metal complexes, and aromatic Aromatic terminal groups in the group. The aromatic terminal group may be one type, or two or more types. From the viewpoint of fluorescence characteristics or device characteristics, it is preferable that there are substantially no terminal groups other than aromatic terminal groups. Here, the molecular chain terminal refers to the aromatic terminal group present at the terminal of the polymer compound by the production method of the present invention; it belongs to the leaving group of the monomer used in polymerization, and exists at the time of polymerization without leaving. A leaving group at the end of a polymer compound; a hydrogen atom that is bonded to an aromatic terminal group without being bonded to a leaving group derived from a monomer present at the end of a polymer compound. Among these molecular chain terminals, the leaving group that is a leaving group of a monomer used in polymerization and does not leave during polymerization but exists at the end of a polymer compound is produced, for example, using a monomer having a halogen atom as a raw material. In the case of the polymer compound of the present invention, if the halogen remains at the terminal of the polymer compound, the fluorescence characteristics and the like tend to decrease, so it is preferable that substantially no leaving group of the monomer remains at the terminal. the
高分子化合物中,通过将其分子链末端的至少一方用选自1价杂环基、1价芳香族胺基、由杂环配位金属络合物衍生的1价的基团或化学式量在90以上的芳基中的芳香族末端基封堵(blocking),就可以期待对高分子化合物赋予各种各样的特性。具体来说,可以举出延长元件的亮度降低所需要的时间的效果;提高电荷注入性、电荷输送性、发光特性的效果;提高共聚物间的相溶性或相互作用的效果;锚定(anchor-like)的效果等。 In a polymer compound, at least one of its molecular chain ends is selected from a monovalent heterocyclic group, a monovalent aromatic amine group, a monovalent group or a chemical formula derived from a heterocyclic coordination metal complex By blocking the aromatic terminal group in the aryl group of 90 or more, various properties can be expected to be imparted to the polymer compound. Specifically, the effect of prolonging the time required for the brightness of the device to decrease; the effect of improving charge injection, charge transport, and luminescent characteristics; the effect of improving compatibility or interaction between copolymers; anchoring (anchor) -like) effects, etc. the
作为1价的芳香族胺基,可以例示出在上述式(15)的构造中将具有的2个结合键当中的1个用R封堵的构造。 Examples of the monovalent aromatic amine group include a structure in which one of the two bonds in the structure of the formula (15) is blocked with R. the
作为由杂环配位金属络合物衍生的1价的基,可以例示出在上述的具有金属络合物构造的2价的基中将2个所具有的结合键当中的1个用R封堵的构造。 As a monovalent group derived from a heterocyclic coordination metal complex, one of the two bonds in the above-mentioned divalent group having a metal complex structure is sealed with R blocked structure. the
本发明的高分子化合物所具有的芳香族末端基当中,作为化学式量在90以上的芳基,碳数通常为6~60左右。这里所说的芳基的化学式量是指,在将芳基用化学式表达时,对于该化学式中的各元素,在各个元素的原子数上乘以原子量后的值的和。 Among the aromatic terminal groups possessed by the polymer compound of the present invention, the aryl group having a chemical formula weight of 90 or more usually has about 6 to 60 carbon atoms. The chemical formula weight of the aryl group referred to here refers to the sum of values obtained by multiplying the atomic number of each element by the atomic weight for each element in the chemical formula when the aryl group is expressed in a chemical formula. the
作为芳基,可以举出苯基、萘基、蒽基、具有芴构造的基、稠环化合物基等。 Examples of the aryl group include a phenyl group, a naphthyl group, an anthracenyl group, a group having a fluorene structure, a condensed ring compound group, and the like. the
作为将末端封堵的苯基,例如可以举出 As the phenyl group to block the end, for example,
作为将末端封堵的萘基,例如可以举出 As the naphthyl end-blocked, for example, can be cited
作为蒽基,例如可以举出 As the anthracenyl group, for example,
作为含有芴构造的基,例如可以举出 As a group containing a fluorene structure, for example,
作为稠环化合物基,例如可以举出 As the condensed ring compound group, for example, can be cited
作为提高电荷注入性、电荷输送性的末端基,优选1价的杂环基、1价的芳香族胺基、稠环化合物基,更优选1价的杂环基、稠环化合物基。 As the terminal group for improving the charge injection and charge transport properties, a monovalent heterocyclic group, a monovalent aromatic amine group, and a condensed ring compound group are preferable, and a monovalent heterocyclic group and a condensed ring compound group are more preferable. the
作为提高发光特性的末端基,优选萘基、蒽基、稠环化合物基、由杂环配位金属络合物衍生的1价的基团。 As the terminal group for improving luminescent properties, naphthyl, anthracenyl, condensed ring compound groups, and monovalent groups derived from heterocyclic coordination metal complexes are preferable. the
作为具有延长元件的亮度降低所需要的时间的效果的末端基,优选具有取代基的芳基,优选具有1~3个烷基的苯基。 As the terminal group having the effect of prolonging the time required for the brightness of the device to decrease, an aryl group having a substituent is preferable, and a phenyl group having 1 to 3 alkyl groups is preferable. the
作为具有提高高分子化合物间的相溶性或相互作用的效果的末端基,优选具有取代基的芳基。另外,通过使用碳数在6以上的烷基取代了的苯基,可以起到锚定的效果。所谓锚定效果是指,末端基对聚合物的凝聚物起到锚定的作用,提高相互作用的效果。 An aryl group having a substituent is preferable as the terminal group having the effect of improving compatibility or interaction between polymer compounds. In addition, by using a phenyl group substituted with an alkyl group having 6 or more carbon atoms, an anchoring effect can be exerted. The so-called anchoring effect means that the terminal group plays an anchoring role on the aggregate of the polymer and improves the interaction effect. the
作为提高元件特性的基团,优选下述构造。 As a group for improving device characteristics, the following structures are preferable. the
式中的R可以例示出上述的R。 R in the formula can exemplify the above-mentioned R. the
本发明的高分子化合物的聚苯乙烯换算的数均分子量通常为103~108 左右,优选为104~106左右。另外,聚苯乙烯换算的重均分子量为103~108,优选104~5×106。 The polystyrene-equivalent number average molecular weight of the polymer compound of the present invention is usually about 10 3 to 10 8 , preferably about 10 4 to 10 6 . In addition, the polystyrene-equivalent weight average molecular weight is 10 3 to 10 8 , preferably 10 4 to 5×10 6 .
作为针对本发明的高分子化合物的良溶剂,可以例示出氯仿、二氯甲烷、二氯乙烷、四氢呋喃、甲苯、二甲苯、、萘满、萘烷、正丁基苯等。虽然根据高分子化合物的构造或分子量而不同,但是通常可以在这些溶剂中溶解0.1重量%以上。 Examples of good solvents for the polymer compound of the present invention include chloroform, dichloromethane, dichloroethane, tetrahydrofuran, toluene, xylene, , tetralin, decalin, n-butylbenzene, etc. Although it varies depending on the structure and molecular weight of the polymer compound, it can usually be dissolved in these solvents at 0.1% by weight or more.
本发明的高分子化合物与所对应的聚芴衍生物相比,可以在更短波长下发光。 Compared with the corresponding polyfluorene derivatives, the polymer compound of the present invention can emit light at a shorter wavelength. the
下面,对本发明的高分子化合物的制造方法进行说明。 Next, the production method of the polymer compound of the present invention will be described. the
本发明的高分子化合物当中,例如具有以式(1-1)、(1-2)、(1-3)表示的重复单元的化合物作为各自的原料,可以通过使用以下述式(16-1)、(16-2)及(16-3) Among the polymer compounds of the present invention, for example, compounds having repeating units represented by formulas (1-1), (1-2), and (1-3) can be obtained by using the following formula (16-1 ), (16-2) and (16-3)
式(16-1) 式(16-2) 式(16-3) Formula (16-1) Formula (16-2) Formula (16-3)
[式中,A环、B环、C环、Z1~Z5如前所述。Y1、Y2、Y3、Y4、Y5 及Y6分别独立地表示参与聚合的取代基。] [In the formula, ring A, ring B, ring C, and Z 1 to Z 5 are as described above. Y 1 , Y 2 , Y 3 , Y 4 , Y 5 and Y 6 each independently represent a substituent involved in polymerization. ]
表示的化合物来聚合而制造。 The indicated compound is produced by polymerization. the
以式(16-1)表示的化合物当中,优选以式(17-1)表示的化合物。 Among the compounds represented by the formula (16-1), the compound represented by the formula (17-1) is preferable. the
式(17-1) Formula (17-1)
[式中,R1、R2、a、b、D环、Q、z、Y1及Y2表示与上述相同的意思。] [In the formula, R 1 , R 2 , a, b, ring D, Q, z, Y 1 and Y 2 represent the same meanings as above. ]
以式(17-1)表示的化合物当中,更优选以式(18-1)、(18-2)、(18-3)及(18-4)表示的构造。 Among the compounds represented by formula (17-1), structures represented by formulas (18-1), (18-2), (18-3) and (18-4) are more preferable. the
式(18-1) 式(18-2) Formula (18-1) Formula (18-2)
式(18-3) 式(18-4) Formula (18-3) Formula (18-4)
另外,以式(16-2)表示的化合物当中,优选以式(17-2)表示的化合物。 In addition, among the compounds represented by the formula (16-2), the compound represented by the formula (17-2) is preferable. the
式(17-2) Formula (17-2)
[式中,B环、C环、Z2、Z3、Z4、Y3及Y4表示与上述相同的意思。Z6、Z7及Z8分别独立地表示C-(Q)z或氮原子。Z1a、Z5a及Z9分别独立地表示碳原子。Q及z表示与上述相同的意思。R4表示取代基。e表示0~2的整数。在有多个R4的情况下,它们既可以相同也可以不同,也可以R4之间结合而形成环。] [In the formula, ring B, ring C, Z 2 , Z 3 , Z 4 , Y 3 and Y 4 have the same meanings as above. Z 6 , Z 7 and Z 8 each independently represent C-(Q) z or a nitrogen atom. Z 1a , Z 5a and Z 9 each independently represent a carbon atom. Q and z represent the same meaning as above. R 4 represents a substituent. e represents an integer of 0-2. When there are a plurality of R 4 , they may be the same or different, and R 4 may be combined to form a ring. ]
另外,以式(16-3)表示的化合物当中,优选以式(17-3)表示的化合物。 In addition, among the compounds represented by the formula (16-3), the compound represented by the formula (17-3) is preferable. the
式(17-3) Formula (17-3)
[式中,A环、B环、Z1、Z4、Z5、Y5及Y6表示与上述相同的意思。Z10、Z11、Z12及Z13分别独立地表示C-(Q)z或氮原子。Z2a及Z3a分别独立地表示碳原子。Q及z表示与上述相同的意思。R5表示取代基。f表示0~2的整数。在有多个R5的情况下,它们既可以相同也可以不同,也可以R5之间结合而形成环。] [In the formula, ring A, ring B, Z 1 , Z 4 , Z 5 , Y 5 and Y 6 have the same meanings as above. Z 10 , Z 11 , Z 12 and Z 13 each independently represent C-(Q) z or a nitrogen atom. Z 2a and Z 3a each independently represent a carbon atom. Q and z represent the same meaning as above. R 5 represents a substituent. f represents an integer of 0-2. When there are a plurality of R 5 , they may be the same or different, and R 5 may be combined to form a ring. ]
本发明的制造方法中,作为参与聚合的取代基,可以举出卤原子、烷基磺酸酯基、芳基磺酸酯基、芳烷基磺酸酯基、硼酸酯基、锍甲基、鏻甲基、磷酸酯甲基、单卤代甲基、-B(OH)2、甲酰基、氰基、乙烯基等。 In the production method of the present invention, examples of substituents involved in polymerization include halogen atoms, alkylsulfonate groups, arylsulfonate groups, aralkylsulfonate groups, borate groups, sulfonium methyl , phosphonium methyl, phosphate methyl, monohalomethyl, -B(OH) 2 , formyl, cyano, vinyl, etc.
这里,作为卤原子,可以举出氟原子、氯原子、溴原子、碘原子。从提高聚合度的观点考虑,优选溴原子、碘原子。 Here, examples of the halogen atom include a fluorine atom, a chlorine atom, a bromine atom, and an iodine atom. From the viewpoint of increasing the degree of polymerization, bromine atoms and iodine atoms are preferable. the
作为烷基磺酸酯基,可以例示出甲磺酸酯基、乙磺酸酯基、三氟甲烷磺酸酯基等,作为芳基磺酸酯基,可以例示出苯磺酸酯基、对甲苯磺酸酯基等,作为芳基磺酸酯基,可以例示出苄基磺酸酯基等。 As the alkylsulfonate group, mesylate group, ethanesulfonate group, trifluoromethanesulfonate group, etc. can be exemplified, and as the arylsulfonate group, benzenesulfonate group, p- As the tosylate group and the like, examples of the arylsulfonate group include benzylsulfonate group and the like. the
作为硼酸酯基,可以例示出以下式表示的基团。 As boric acid ester group, the group represented by the following formula can be illustrated. the
式中,Me表示甲基,Et表示乙基。 In the formula, Me represents a methyl group, and Et represents an ethyl group. the
作为锍甲基,可以例示出以下式表示的基团。 As a sulfonium methyl group, the group represented by the following formula can be illustrated. the
-CH2S+Me2X-、-CH2S+Ph2X-、 -CH 2 S + Me 2 X - , -CH 2 S + Ph 2 X - ,
(X表示卤原子,Ph表示苯基。) (X represents a halogen atom, Ph represents a phenyl group.)
作为鏻甲基,可以例示出以下式表示的基团。 As a phosphonium methyl group, the group represented by the following formula can be illustrated. the
-CH2P+Ph3X-(X表示卤原子。) -CH 2 P + Ph 3 X - (X represents a halogen atom.)
作为磷酸酯甲基,可以例示出以下式表示的基团。 As the phosphate methyl group, groups represented by the following formulas can be exemplified. the
-CH2PO(OR’)2(X表示卤原子,R’表示烷基、芳基、芳烷基。) -CH 2 PO(OR') 2 (X represents a halogen atom, R' represents an alkyl group, an aryl group, or an aralkyl group.)
作为单卤代甲基,可以例示出氟代甲基、氯代甲基、溴代甲基、碘代甲基。 Examples of the monohalomethyl group include fluoromethyl, chloromethyl, bromomethyl and iodomethyl. the
作为参与聚合的取代基来说优选的取代基虽然根据聚合反应的种类而不同,但是例如在Yamamoto偶联反应等使用0价镍络合物的情况下,可以举出卤原子、烷基磺酸酯基、芳基磺酸酯基或芳烷基磺酸酯基。另外,在Suzuki偶联反应等使用镍催化剂或钯催化剂的情况下,可以举出烷基磺酸酯基、卤原子、硼酸酯基、-B(OH)2等。 Preferred substituents as substituents involved in polymerization vary depending on the type of polymerization reaction, but for example, in the case of using a zero-valent nickel complex such as Yamamoto coupling reaction, halogen atoms, alkylsulfonic acid Ester group, arylsulfonate group or aralkylsulfonate group. In addition, when a nickel catalyst or a palladium catalyst is used such as Suzuki coupling reaction, alkylsulfonate group, halogen atom, borate group, -B(OH) 2 and the like can be mentioned.
特别是,具有式(4-1)的重复单元的高分子化合物可以将具有式(4-2)~(4-4)的重复单元的高分子化合物用在活性碳上担载了钯、铂、铑、钌或混合了它们的贵金属的催化剂氢化而得。 In particular, the polymer compound having the repeating unit of formula (4-1) can use the polymer compound having the repeating unit of formula (4-2) to (4-4) on activated carbon loaded with palladium, platinum , rhodium, ruthenium or a mixture of their noble metal catalyst hydrogenation. the
相反,具有式(4-2)~(4-4)的重复单元的高分子化合物可以将具有式(4-1)的重复单元的高分子化合物在2,3-二氯-5,6-二氰基-1,4-苯醌(DDQ)、碱性条件下用溴化四丁基铵氧化而得。 On the contrary, the macromolecular compound having the repeating unit of formula (4-2)~(4-4) can have the polymer compound of repeating unit of formula (4-1) in 2,3-dichloro-5,6- Dicyano-1,4-benzoquinone (DDQ), obtained by oxidation with tetrabutylammonium bromide under alkaline conditions. the
另外,本发明的高分子化合物在具有式(1-1)或式(1-2)或式(1-3)以外的重复单元的情况下,只要使作为式(1-1)或式(1-2)或式(1-3)以外的重复单元的具有2个参与聚合的取代基的化合物共存而聚合即可。 In addition, when the polymer compound of the present invention has a repeating unit other than formula (1-1) or formula (1-2) or formula (1-3), as long as the formula (1-1) or formula ( 1-2) or compounds having repeating units other than formula (1-3) having two substituents participating in polymerization may coexist and polymerize. the
除了以上述式(16-1)、(16-2)或(16-3)表示的化合物以外,还可以将以下述式(19)~(22)的某个表示的化合物作为原料使用。 In addition to the compound represented by the above formula (16-1), (16-2) or (16-3), a compound represented by any of the following formulas (19) to (22) can also be used as a raw material. the
Y7-Ar1-Y8 (19) Y 7 -Ar 1 -Y 8 (19)
Y9-(Ar2-X1)ff-Ar3-Y10 (20) Y 9 -(Ar 2 -X 1 ) ff -Ar 3 -Y 10 (20)
Y11-Ar4-X2-Y12 (21) Y 11 -Ar 4 -X 2 -Y 12 (21)
Y13-X3-Y14 (22) Y 13 -X 3 -Y 14 (22)
[式中,Ar1、Ar2、Ar3、Ar4、ff、X1、X2及X3与上述相同。Y7、Y8、Y9、Y10、Y11、Y12、Y13、Y14分别独立地表示可以聚合的取代基。] [wherein, Ar 1 , Ar 2 , Ar 3 , Ar 4 , ff, X 1 , X 2 and X 3 are the same as above. Y 7 , Y 8 , Y 9 , Y 10 , Y 11 , Y 12 , Y 13 , and Y 14 each independently represent a polymerizable substituent. ]
还可以制造除了以上述式(1-1)、(1-2)或(1-3)表示的单元以外,依次具有1个以上(5)、(6)、(7)或(8)的单元的高分子化合物。 In addition to the unit represented by the above formula (1-1), (1-2) or (1-3), it is also possible to produce one or more units of (5), (6), (7) or (8) in this order. Unit polymer compound. the
另外,作为以上述式(1-1)、(1-2)或(1-3)表示的重复单元以外的重复单元的具有与上述式(15)对应的2个参与聚合的取代基的化合物,可以举出以下述式(15-7)表示的化合物。 In addition, as a repeating unit other than the repeating unit represented by the above formula (1-1), (1-2) or (1-3), a compound having two substituents corresponding to the above formula (15) participating in polymerization , Compounds represented by the following formula (15-7) can be mentioned. the
[式中,Ar6、Ar7、Ar8、Ar9、Ar10、Ar11、Ar12、x及y与上述相同。Y15及Y16分别独立地表示参与聚合的取代基。] [wherein, Ar 6 , Ar 7 , Ar 8 , Ar 9 , Ar 10 , Ar 11 , Ar 12 , x and y are the same as above. Y15 and Y16 each independently represent a substituent involved in polymerization. ]
更优选以式(15-8)或式(15-9)表示的化合物。 Compounds represented by formula (15-8) or formula (15-9) are more preferred. the
式(15-8) 式(15-9) Formula (15-8) Formula (15-9)
[式中,Re~Rg如前所述。Y17、Y18、Y19、Y20分别独立地表示参与聚合的取代基。] [wherein, Re to Rg are as described above. Y 17 , Y 18 , Y 19 , and Y 20 each independently represent a substituent involved in polymerization. ]
本发明的制造方法具体来说可以将作为单体的具有多个参与缩聚的取代基的化合物根据需要溶解于有机溶剂中,例如使用碱或适当的催化剂,在有机溶剂的熔点以上沸点以下进行。例如可以使用“有机反应(Organic Reactions)”,第14卷、270-490页,John Wiley & Sons,Inc.,1965年;“有机合成(Organic Syntheses)”,合集第6卷(Collective VolumeVI),407-411页,John Wiley & Sons,Inc.,1988年;化学综述(Chem.Rev.),第95卷,2475页(1995年);有机金属化学杂志(J.Organomet.Chem.),第576卷,147页(1999年);大分子化学大分子论丛(Makromol.Chem.,Macromol.Symp.),第12卷,229页(1987年)等中记载的公知的方法。 Specifically, the production method of the present invention can be carried out by dissolving a compound having a plurality of substituents involved in polycondensation as a monomer in an organic solvent, for example, using a base or a suitable catalyst at a temperature above the melting point and below the boiling point of the organic solvent. For example, "Organic Reactions", Vol. 14, pp. 270-490, John Wiley & Sons, Inc., 1965; "Organic Syntheses", Collective Volume VI, pp. 407-411, John Wiley & Sons, Inc., 1988; Chem.Rev., Vol. 95, pp. 2475 (1995); J.Organomet.Chem., pp. 576, p. 147 (1999); known methods described in Makromol. Chem., Macromol. Symp., vol. 12, p. 229 (1987). the
本发明的高分子化合物的制造方法中,作为将其聚合的方法,可以与参与以上述式(16-1)~(16-3)及(22)~(25)表示的化合物的聚合的取代基对应地使用已知的聚合反应来制造。 In the production method of the polymer compound of the present invention, as a method of polymerizing it, substitutions involving the polymerization of the compounds represented by the above formulas (16-1) to (16-3) and (22) to (25) may be carried out. The bases are correspondingly produced using known polymerization reactions. the
在本发明的高分子化合物在聚合中生成双键的情况下,例如可以举出 特开平5-202355号公报中记载的方法。即,利用具有甲酰基的化合物与具有鏻甲基的化合物的、或具有甲酰基和鏻甲基的化合物的Wittig反应的聚合;利用具有乙烯基的化合物与具有卤原子的化合物的Heck反应的聚合;利用具有2个或2个以上单卤代甲基的化合物的脱卤化氢法的聚合;利用具有2个或2个以上锍甲基的化合物的锍盐分解法的聚合;利用具有甲酰基的化合物与具有氰基的化合物的Knoevenagel反应的聚合等方法;利用具有2个或2个以上甲酰基的化合物的McMurry反应的聚合等方法。 When the polymer compound of the present invention forms a double bond during polymerization, for example, the method described in JP-A-5-202355 can be used. That is, polymerization using the Wittig reaction of a compound having a formyl group and a compound having a phosphonium methyl group, or a compound having a formyl group and a phosphonium methyl group; polymerization using a Heck reaction of a compound having a vinyl group and a compound having a halogen atom ; Polymerization using the dehydrohalogenation method of compounds having 2 or more monohalogenated methyl groups; Polymerization using the sulfonium salt decomposition method of compounds having 2 or more sulfonium methyl groups; Utilizing compounds with formyl groups Methods such as polymerization using Knoevenagel reaction of compounds having cyano groups; methods such as polymerization using McMurry reactions of compounds having 2 or more formyl groups. the
在本发明的高分子化合物因聚合而在主链中生成三键的情况下,例如可以利用Heck反应、Sonogashira反应。 When the polymer compound of the present invention forms a triple bond in the main chain by polymerization, for example, Heck reaction and Sonogashira reaction can be utilized. the
另外,在不生成双键或三键的情况下,例如可以例示出由对应的单体利用Suzuki偶联反应聚合的方法;利用Grignard反应聚合的方法;利用Ni(0)络合物聚合的方法;利用FeCl3等氧化剂聚合的方法;电化学地氧化聚合的方法;或者利用具有适当的离去基的中间体高分子的分解的方法等。 In addition, in the case where no double bond or triple bond is formed, for example, a method of polymerizing from a corresponding monomer by Suzuki coupling reaction; a method of polymerizing by Grignard reaction; a method of polymerizing by Ni(0) complex can be exemplified. ; The method of polymerization using an oxidizing agent such as FeCl 3 ; the method of electrochemically oxidative polymerization; or the method of decomposing an intermediate polymer with an appropriate leaving group, etc.
它们当中,由于容易进行构造控制,因此优选利用Wittig反应的聚合、利用Heck反应的聚合、利用Knoevenagel反应的聚合及利用Suzuki偶联反应聚合的方法、利用Grignard反应聚合的方法、利用镍0价络合物聚合的方法。 Among them, the method of polymerization by Wittig reaction, the polymerization by Heck reaction, the polymerization by Knoevenagel reaction, the method of polymerization by Suzuki coupling reaction, the method of polymerization by Grignard reaction, and the method of polymerization by nickel 0 valence complex are preferable because of the ease of structural control. method of compound polymerization. the
本发明的制造方法当中,在将以式(16-1)、(16-2)或(16-3)表示的化合物单独地或者与选自以式(19)~(22)表示的化合物中的至少-种聚合之时,优选如下的制造方法,即,Y1、Y2、Y3、Y4、Y5、Y6、Y7、Y8、Y9、Y10、Y11、Y12、Y13、Y14、Y15、Y16、Y17、Y18、Y19、Y20 分别独立地为卤原子、烷基磺酸酯基、芳基磺酸酯基或芳烷基磺酸酯基,在镍0价络合物存在下进行缩聚。 In the production method of the present invention, in the compound represented by the formula (16-1), (16-2) or (16-3) alone or in the compound selected from the compounds represented by the formula (19) to (22) When polymerizing at least one of the following production methods, that is, Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 , Y 7 , Y 8 , Y 9 , Y 10 , Y 11 , Y 12 , Y 13 , Y 14 , Y 15 , Y 16 , Y 17 , Y 18 , Y 19 , and Y 20 are each independently a halogen atom, an alkylsulfonate group, an arylsulfonate group or an aralkylsulfonate group Ester group, polycondensation in the presence of nickel zero-valent complex.
作为原料化合物,可以举出二卤化化合物、双(烷基磺酸酯)化合物、双(芳基磺酸酯)化合物、双(芳烷基磺酸酯)化合物或卤素-烷基磺酸酯化合物、卤素-芳基磺酸酯化合物、卤素-芳烷基磺酸酯化合物、烷基磺酸酯-芳基磺酸酯化合物、烷基磺酸酯-芳烷基磺酸酯化合物、芳基磺酸酯-芳烷基磺酸酯化合物。 Examples of raw material compounds include dihalogenated compounds, bis(alkylsulfonate) compounds, bis(arylsulfonate) compounds, bis(arylalkylsulfonate) compounds, and halogen-alkylsulfonate compounds. , Halogen-arylsulfonate compound, halogen-aralkylsulfonate compound, alkylsulfonate-arylsulfonate compound, alkylsulfonate-aralkylsulfonate compound, arylsulfonate Ester-aralkylsulfonate compounds. the
另外,本发明的制造方法当中,在将以式(16-1)、(16-2)或(16 -3)表示的化合物单独地或者与选自以式(19)~(22)表示的化合物中的至少一种聚合之时,优选如下的制造方法,即,Y1、Y2、Y3、Y4、Y5、Y6、Y7、Y8、Y9、Y10、Y11、Y12、Y13、Y14、Y15、Y16、Y17、Y18、Y19、Y20分别独立地为卤原子、烷基磺酸酯基、芳基磺酸酯基、芳烷基磺酸酯基、-B(OH)2或硼酸酯基,卤原子、烷基磺酸酯基、芳基磺酸酯基及芳烷基磺酸酯基的摩尔数的总数与-B(OH)2及硼酸酯基的摩尔数的总数的比实质上为1(通常K/J为0.7~1.2的范围),使用镍或钯催化剂进行缩聚。 In addition, in the production method of the present invention, when the compound represented by the formula (16-1), (16-2) or (16-3) is used alone or in combination with a compound selected from the compounds represented by the formulas (19) to (22), When at least one of the compounds is polymerized, the production method of Y 1 , Y 2 , Y 3 , Y 4 , Y 5 , Y 6 , Y 7 , Y 8 , Y 9 , Y 10 , Y 11 is preferable. , Y 12 , Y 13 , Y 14 , Y 15 , Y 16 , Y 17 , Y 18 , Y 19 , and Y 20 are each independently a halogen atom, an alkylsulfonate group, an arylsulfonate group, an aralkyl group Sulfonate group, -B(OH) 2 or borate group, the total number of moles of halogen atom, alkylsulfonate group, arylsulfonate group and aralkylsulfonate group and -B The ratio of the total number of moles of (OH) 2 and borate groups is substantially 1 (usually K/J is in the range of 0.7 to 1.2), and polycondensation is performed using a nickel or palladium catalyst.
作为具体的原料化合物的组合,可以举出二卤化化合物、双(烷基磺酸酯)化合物、双(芳基磺酸酯)化合物或双(芳烷基磺酸酯)化合物与二硼酸化合物或二硼酸酯化合物的组合。 Specific combinations of raw material compounds include dihalogenated compounds, bis(alkylsulfonate) compounds, bis(arylsulfonate) compounds, or bis(arylalkylsulfonate) compounds and diboronic acid compounds or A combination of diboronate compounds. the
另外,可以举出卤素-硼酸化合物、卤素-硼酸酯化合物、芳基磺酸酯-硼酸化合物、烷基磺酸酯-硼酸酯化合物、芳基磺酸酯-硼酸化合物、芳基磺酸酯-硼酸酯化合物、芳烷基磺酸酯-硼酸化合物、芳烷基磺酸酯-硼酸化合物、芳烷基磺酸酯-硼酸酯化合物。 In addition, halogen-boric acid compounds, halogen-boric acid ester compounds, arylsulfonate-boric acid compounds, alkylsulfonate-boric acid ester compounds, arylsulfonate-boric acid compounds, arylsulfonic acid Ester-boronate compounds, aralkylsulfonate-boronic acid compounds, aralkylsulfonate-boronic acid compounds, aralkylsulfonate-boronic acid ester compounds. the
作为有机溶剂,虽然根据所用的化合物或反应而不同,但是一般来说为了抑制副反应,所用的溶剂最好充分地实施脱氧处理,在惰性气氛中进行反应。另外,最好同样地进行脱水处理。但是,在Suzuki偶联反应那样的与水的2相体系中的反应的情况下并不受此限制。 The organic solvent varies depending on the compound used or the reaction, but in general, in order to suppress side reactions, the solvent used is preferably deoxidized sufficiently and the reaction is carried out in an inert atmosphere. In addition, it is preferable to perform dehydration treatment in the same manner. However, in the case of a reaction in a two-phase system with water such as a Suzuki coupling reaction, it is not limited thereto. the
作为溶剂,可以例示出戊烷、己烷、庚烷、辛烷、环己烷等饱和烃;苯、甲苯、乙基苯、二甲苯等不饱和烃;四氯化碳、氯仿、二氯甲烷、氯丁烷、溴丁烷、氯戊烷、溴戊烷、氯己烷、溴己烷、氯环己烷、溴环己烷等卤化饱和烃;氯苯、二氯苯、三氯苯等卤化不饱和烃;甲醇、乙醇、丙醇、异丙醇、丁醇、叔丁醇等醇类;甲酸、乙酸、丙酸等羧酸类;二甲醚、二乙醚、甲基叔丁基醚、四氢呋喃、四氢吡喃、二氧杂环己烷等醚类;三甲胺、三乙胺、N,N,N’,N’-四甲基乙二胺、吡啶等胺类;N,N-二甲基甲酰胺、N,N-二甲基乙酰胺、N,N-二乙基乙酰胺、N-甲基吗啉氧化物等酰胺类,可以使用单一溶剂或它们的混合溶剂。它们当中,优选醚类,更优选四氢呋喃、二乙醚。 Examples of solvents include saturated hydrocarbons such as pentane, hexane, heptane, octane, and cyclohexane; unsaturated hydrocarbons such as benzene, toluene, ethylbenzene, and xylene; carbon tetrachloride, chloroform, and methylene chloride , chlorobutane, bromobutane, chloropentane, bromopentane, chlorohexane, bromohexane, chlorocyclohexane, bromocyclohexane and other halogenated saturated hydrocarbons; chlorobenzene, dichlorobenzene, trichlorobenzene, etc. Halogenated unsaturated hydrocarbons; alcohols such as methanol, ethanol, propanol, isopropanol, butanol, tert-butanol; carboxylic acids such as formic acid, acetic acid, propionic acid; dimethyl ether, diethyl ether, methyl tert-butyl ether , tetrahydrofuran, tetrahydropyran, dioxane and other ethers; trimethylamine, triethylamine, N, N, N', N'-tetramethylethylenediamine, pyridine and other amines; N, N For amides such as dimethylformamide, N,N-dimethylacetamide, N,N-diethylacetamide, and N-methylmorpholine oxide, a single solvent or a mixed solvent thereof can be used. Among them, ethers are preferable, and tetrahydrofuran and diethyl ether are more preferable. the
为了使之反应,适当地添加碱或合适的催化剂。它们可以根据所用的 反应选择。该碱或催化剂优选充分地溶解于反应所用的溶剂中的。作为将碱或催化剂混合的方法,可以例示出将反应液在氩气或氮气等惰性气氛下搅拌的同时慢慢地添加碱或催化剂的溶液;或者相反地向碱或催化剂的溶液中慢慢地添加反应液的方法。 For the reaction, a base or a suitable catalyst is appropriately added. They can be selected according to the reaction used. The base or catalyst is preferably sufficiently dissolved in the solvent used for the reaction. As a method of mixing the base or the catalyst, it can be exemplified that the reaction liquid is stirred under an inert atmosphere such as argon or nitrogen, and slowly adding a solution of the base or the catalyst; The method of adding the reaction solution. the
在将本发明的高分子化合物用于高分子LED等中的情况下,由于其纯度对发光特性等元件的性能产生影响,因此最好将聚合前的单体用蒸馏、升华精制、重结晶等方法精制后聚合。另外,在聚合后,最好进行再沉淀精制、利用色谱的分离等纯化处理。 When the polymer compound of the present invention is used in polymer LEDs, etc., since its purity affects the performance of elements such as light emitting characteristics, it is preferable to distill, sublimate, recrystallize, etc. the monomer before polymerization. Polymerization after refining. In addition, after polymerization, it is preferable to carry out purification treatment such as reprecipitation purification and separation by chromatography. the
作为本发明的高分子化合物的原料来说有用的(16-1)~(16-3)、(17-1)~(17-3)及(18-1)~(18-4)可以通过对将上述式的Y1~Y6以氢原子取代的构造的化合物进行溴化而获得。 (16-1) to (16-3), (17-1) to (17-3), and (18-1) to (18-4) useful as raw materials for the polymer compound of the present invention can be obtained by It is obtained by brominating a compound having a structure in which Y 1 to Y 6 of the above formula is substituted with hydrogen atoms.
特别是,式(18-1)的构造的化合物也可以通过将具有式(18-2)~(18-4)的构造的化合物利用在活性碳上担载了钯、铂、铑、钌或混合了它们的贵金属的催化剂氢化而获得。 In particular, the compound of the structure of formula (18-1) can also be carried by palladium, platinum, rhodium, ruthenium or Mixed catalyst hydrogenation of their precious metals obtained. the
相反,具有式(18-2)~(18-4)的构造的化合物可以通过将具有式(18-1)的构造的化合物在2,3-二氯-5,6-二氰基-1,4-苯醌(DDQ)、碱性条件下使用溴化四丁基铵氧化来获得。 On the contrary, the compound having the structure of formula (18-2)~(18-4) can be obtained by the compound having the structure of formula (18-1) in 2,3-dichloro-5,6-dicyano-1 , 4-Benzoquinone (DDQ), obtained by oxidation with tetrabutylammonium bromide under basic conditions. the
下面,对本发明的高分子LED进行说明。 Next, the polymer LED of the present invention will be described. the
本发明的高分子LED的特征是,在由阳极及阴极构成的电极间,具有有机层,该有机层含有本发明的高分子化合物。 The polymer LED of the present invention is characterized in that it has an organic layer between electrodes composed of an anode and a cathode, and the organic layer contains the polymer compound of the present invention. the
有机层虽然无论是发光层、空穴输送层、电子输送层等的哪种都可以,但是有机层优选为发光层。 The organic layer may be any of a light emitting layer, a hole transport layer, an electron transport layer, etc., but the organic layer is preferably a light emitting layer. the
这里,所谓发光层是指具有发光的功能的层,所谓空穴输送层是指具有输送空穴的功能的层,所谓电子输送层是指具有输送电子的功能的层。而且,将电子输送层和空穴输送层总称为电荷输送层。发光层、空穴输送层、电子输送层可以分别独立地使用2层以上。 Here, the light-emitting layer refers to a layer having a function of emitting light, the so-called hole-transporting layer refers to a layer having a function of transporting holes, and the term “electron-transporting layer” refers to a layer having a function of transporting electrons. Furthermore, the electron transport layer and the hole transport layer are collectively referred to as a charge transport layer. A light-emitting layer, a hole transport layer, and an electron transport layer can be independently used in two or more layers. the
在有机层为发光层的情况下,作为有机层的发光层也可以还含有空穴输送材料、电子输送材料或发光材料。这里,所谓发光性材料是指显示出荧光及/或磷光的材料。 When the organic layer is a light-emitting layer, the light-emitting layer as the organic layer may further contain a hole transport material, an electron transport material, or a light-emitting material. Here, the term "luminescent material" refers to a material that exhibits fluorescence and/or phosphorescence. the
在将本发明的高分子化合物与空穴输送材料混合的情况下,相对于该 混合物整体,空穴输送材料的混合比例为1wt%~80wt%,优选为5wt%~60wt%。在将本发明的高分子化合物与电子输送材料混合的情况下,相对于该混合物整体,电子输送材料的混合比例为1wt%~80wt%,优选为5wt%~60wt%。另外,在将本发明的高分子化合物与发光材料混合的情况下,相对于该混合物整体,发光材料的混合比例为1wt%~80wt%,优选为5wt%~60wt%。在将本发明的高分子化合物与发光材料、空穴输送材料及/或电子输送材料混合的情况下,相对于该混合物整体,发光材料的混合比例为1wt%~50wt%,优选为5wt%~40wt%,空穴输送材料与电子输送材料以它们的合计表示为1wt%~50wt%,优选为5wt%~40wt%,本发明的高分子化合物的含量为99wt%~20wt%。 When the polymer compound of the present invention is mixed with a hole transport material, the mixing ratio of the hole transport material is 1 wt % to 80 wt %, preferably 5 wt % to 60 wt %, based on the entire mixture. When mixing the polymer compound of the present invention with the electron transport material, the mixing ratio of the electron transport material is 1 wt % to 80 wt %, preferably 5 wt % to 60 wt %, based on the entire mixture. In addition, when the polymer compound of the present invention is mixed with a luminescent material, the mixing ratio of the luminescent material is 1 wt % to 80 wt %, preferably 5 wt % to 60 wt %, based on the entire mixture. When mixing the polymer compound of the present invention with a luminescent material, a hole transport material, and/or an electron transport material, the mixing ratio of the luminescent material is 1 wt % to 50 wt %, preferably 5 wt % to 5 wt %, based on the entire mixture. 40wt%, the total of hole transport material and electron transport material is 1wt%-50wt%, preferably 5wt%-40wt%, the content of the polymer compound of the present invention is 99wt%-20wt%. the
所混合的空穴输送材料、电子输送材料、发光材料可以使用公知的低分子化合物、三重态发光络合物或高分子化合物,但是优选使用高分子化合物。作为高分子化合物的空穴输送材料、电子输送材料及发光材料,可以例示出WO99/139692、WO99/48160、GB2340304A、WO00/53656、WO01/19834、WO00/55927、GB2348316、WO00/46321、WO00/06665、WO99/54943、WO99/54385、US5777070、WO98/06773、WO97/05184、WO00/35987、WO00/53655、WO01/34722、WO99/24526、WO00/22027、WO00/22026、WO98/27136、US573636、WO98/21262、US5741921、WO97/09394、WO96/29356、WO96/10617、EP0707020、WO95/07955、特开平2001-181618、特开平2001-123156、特开平2001-3045、特开平2000-351967、特开平2000-303066、特开平2000-299189、特开平2000-252065、特开平2000-136379、特开平2000-104057、特开平2000-80167、特开平10-324870、特开平10-114891、特开平9-111233、特开平9-45478等中所公布的聚芴、其衍生物及共聚物、聚芳撑、其衍生物及共聚物、聚芳撑亚乙烯基、其衍生物及共聚物、芳香族胺及其衍生物的(共)聚合物。 As the hole transport material, electron transport material, and light-emitting material to be mixed, known low-molecular compounds, triplet light-emitting complexes, or high-molecular compounds can be used, but high-molecular compounds are preferably used. Examples of hole-transporting materials, electron-transporting materials, and light-emitting materials of polymer compounds include WO99/139692, WO99/48160, GB2340304A, WO00/53656, WO01/19834, WO00/55927, GB2348316, WO00/46321, WO00/ 06665, WO99/54943, WO99/54385, US5777070, WO98/06773, WO97/05184, WO00/35987, WO00/53655, WO01/34722, WO99/24526, WO00/22027, WO00/22026, WO98/27136, US WO98/21262, US5741921, WO97/09394, WO96/29356, WO96/10617, EP0707020, WO95/07955, JP2001-181618, JP2001-123156, JP2001-3045, JP2000-351967, 2000-303066, TKP 2000-299189, TKP 2000-252065, TKP 2000-136379, TKP 2000-104057, TKP 2000-80167, TKP 10-324870, TKP 10-114891, TKP 9- Polyfluorene, its derivatives and copolymers, polyarylene, its derivatives and copolymers, polyarylene vinylene, its derivatives and copolymers, and aromatic amines disclosed in 111233, JP-A-9-45478, etc. (co)polymers and their derivatives. the
作为低分子化合物的荧光性材料,例如可以使用萘衍生物、蒽或其衍生物、苝或其衍生物、聚甲炔系、呫吨系、香豆素系、花青系等色素类、8-羟基喹啉或其衍生物的金属络合物、芳香族胺、四苯基环戊二烯或其衍生物或者四苯基丁二烯或其衍生物等。 As fluorescent materials of low molecular weight compounds, for example, pigments such as naphthalene derivatives, anthracene or derivatives thereof, perylene or derivatives thereof, polymethines, xanthenes, coumarins, and cyanines, 8 - Metal complexes of hydroxyquinoline or derivatives thereof, aromatic amines, tetraphenylcyclopentadiene or derivatives thereof, tetraphenylbutadiene or derivatives thereof, and the like. the
具体来说,例如可以使用特开昭57-51781号、相同的59-194393号公报中记载的物质等公知的材料。 Specifically, for example, known materials such as those described in JP-A No. 57-51781 and the same publication No. 59-194393 can be used. the
作为三重态发光络合物,例如可以举出以铱作为中心金属的Ir(ppy)3、Btp2Ir(acac)、以铂作为中心金属的PtOEP、以铕作为中心金属的Eu(TTA)3phen等。 Examples of triplet light-emitting complexes include Ir(ppy)3 and Btp2Ir (acac) with iridium as the central metal, PtOEP with platinum as the central metal, Eu(TTA)3phen with europium as the central metal, etc. wait.
作为三重态发光络合物,具体来说,例如记载于Nature,(1998),395,151、Appl.Phys.Lett.(1999),75(1),4、Proc.SPIE-Int.Soc.Opt.Eng.(2001),4105(Organic Light-Emitting Materials and Devices IV),119、J.Am.Chem.Soc.,(2001),123,4304、Appl.Phys.Lett.(1997),71(18), 2596、Syn.Met.,(1998),94(1),103、Syn.Met.,(1999),99(2),1361、Adv.Mater.,(1999),11(10),852、Jpn.J.Appl.Phys.,34,1883(1995)等中。 As a triplet light-emitting complex, specifically, for example, it is described in Nature, (1998), 395, 151, Appl. Phys. Lett. (1999), 75 (1), 4, Proc. SPIE-Int. Soc. Opt.Eng.(2001), 4105(Organic Light-Emitting Materials and Devices IV), 119, J.Am.Chem.Soc., (2001), 123, 4304, Appl.Phys.Lett.(1997), 71 (18), 2596, Syn.Met., (1998), 94(1), 103, Syn.Met., (1999), 99(2), 1361, Adv.Mater., (1999), 11(10 ), 852, Jpn.J.Appl.Phys., 34, 1883 (1995) et al. the
本发明的组合物含有选自空穴输送材料、电子输送材料、发光材料中的至少一种材料和本发明的高分子化合物,可以作为发光材料或电荷输送材料使用。 The composition of the present invention contains at least one material selected from the group consisting of a hole transport material, an electron transport material, and a luminescent material, and the polymer compound of the present invention, and can be used as a luminescent material or a charge transport material. the
该选自空穴输送材料、电子输送材料、发光材料中的至少一种材料与本发明的高分子化合物的含有比率可以根据用途决定。 The content ratio of at least one material selected from the group consisting of a hole transport material, an electron transport material, and a light emitting material to the polymer compound of the present invention can be determined according to the application. the
作为本发明的其他的实施方式,可以例示出含有2种以上本发明的高分子化合物的高分子组合物。 As another embodiment of the present invention, a polymer composition containing two or more polymer compounds of the present invention can be exemplified. the
作为本发明的高分子LED所具有的发光层的膜厚,最佳值根据所用的材料而不同,只要以使驱动电压和发光效率达到适度的值的方式选择即可。例如为1nm到1μm,优选2nm~500nm,更优选5nm~200nm。 The optimum value of the film thickness of the light emitting layer included in the polymer LED of the present invention differs depending on the material used, and it may be selected so that the driving voltage and luminous efficiency become appropriate values. For example, it is 1 nm to 1 μm, preferably 2 nm to 500 nm, more preferably 5 nm to 200 nm. the
作为发光层的形成方法,例如可以例示出由溶液成膜的方法。在高分子LED的制作之时,通过使用本发明的高分子化合物,不仅在由溶液成膜的情况下,可以在涂布该溶液后将溶剂干燥除去,而且在混合了电荷输送材料或发光材料的情况下,也可以应用相同的方法,在制造上非常有利。 As a method of forming the light emitting layer, for example, a method of forming a film from a solution can be exemplified. In the production of polymer LEDs, by using the polymer compound of the present invention, not only when a film is formed from a solution, the solvent can be dried and removed after coating the solution, but also when a charge transport material or a light emitting material is mixed The same method can also be applied in the case of , which is very advantageous in terms of manufacturing. the
作为由溶液成膜的方法,可以使用旋转涂覆法、浇注法(casting)、显微凹版涂覆法、凹版涂覆法、棒涂覆法、辊涂法、绕线棒(wire bar)涂覆法、浸渍涂覆法、喷雾涂覆法、丝网印刷法、苯胺印刷法、胶版印刷法、喷墨印刷法等涂布法。从容易进行图案形成或分别涂布多种颜色的观点考虑,优选丝网印刷法、苯胺印刷法、胶版印刷法、喷墨印刷法等印刷法。 As a method of forming a film from a solution, spin coating, casting, microgravure coating, gravure coating, bar coating, roll coating, wire bar coating, etc. can be used. Coating method, dip coating method, spray coating method, screen printing method, flexographic printing method, offset printing method, inkjet printing method and other coating methods. Printing methods such as screen printing, flexographic printing, offset printing, and inkjet printing are preferable from the viewpoint of easy pattern formation and separate application of multiple colors. the
作为印刷法等中所用的溶液,可以使用本发明的油墨组合物。 The ink composition of the present invention can be used as a solution used in printing methods and the like. the
本发明的油墨组合物只要含有至少一种本发明的高分子化合物和溶剂即可,另外除了本发明的高分子化合物以外,还可以含有空穴输送材料、电子输送材料、发光材料、稳定剂等添加剂。 The ink composition of the present invention only needs to contain at least one polymer compound of the present invention and a solvent. In addition to the polymer compound of the present invention, it may also contain a hole transport material, an electron transport material, a luminescent material, a stabilizer, etc. additive. the
本发明的油墨组合物在元件制作时为液状,典型的情况是指,在常压(即1个大气压)、25℃为液状的材料。 The ink composition of the present invention is liquid at the time of device fabrication, and typically refers to a material that is liquid at normal pressure (ie, 1 atmosphere) and 25°C. the
另外,本发明的油墨组合物不一定需要为有色。 In addition, the ink composition of the present invention does not necessarily need to be colored. the
该油墨组合物中的本发明的高分子化合物的比例为,相对于除去溶剂 后的该油墨组合物的总重量,通常为20wt%~100wt%,优选为40wt%~100wt%。 The ratio of the polymer compound of the present invention in the ink composition is usually 20 wt% to 100 wt%, preferably 40 wt% to 100 wt%, based on the total weight of the ink composition after removing the solvent. the
另外,油墨组合物中的溶剂的比例相对于该油墨组合物的总重量来说为1wt%~99.9wt%,优选为60wt%~99.9wt%,更优选为90wt%~99.8wt%。 In addition, the proportion of the solvent in the ink composition is 1 wt % to 99.9 wt %, preferably 60 wt % to 99.9 wt %, more preferably 90 wt % to 99.8 wt %, based on the total weight of the ink composition. the
该油墨组合物(溶液)的粘度虽然根据印刷法而不同,然而在喷墨印刷法等溶液经由喷出装置中的情况下,为了防止喷出时的堵塞或飞行弯曲,粘度优选为在25℃处于1~20mPa·s的范围。 Although the viscosity of the ink composition (solution) varies depending on the printing method, when the solution is passed through the discharge device such as the inkjet printing method, in order to prevent clogging or flight deflection at the time of discharge, the viscosity is preferably at 25°C. It is in the range of 1 to 20 mPa·s. the
作为本发明的油墨组合物中所用的溶剂,可以例示出氯仿、二氯甲烷、1,2-二氯乙烷、1,1,2-三氯乙烷、氯苯、邻二氯苯等氯系溶剂;四氢呋喃、二氧杂环己烷等醚系溶剂;甲苯、二甲苯等芳香族烃系溶剂;环己烷、甲基环己烷、正戊烷、正己烷、正庚烷、正辛烷、正壬烷、正癸烷等脂肪族烃系溶剂;丙酮、甲乙酮、环己酮等酮系溶剂;乙酸乙酯、乙酸丁酯、乙基溶纤剂乙酸酯等酯系溶剂;乙二醇、乙二醇单丁醚、乙二醇单乙醚、乙二醇单甲醚、二甲氧基乙烷、丙二醇、二乙氧基甲烷、三甘醇单乙醚、甘油、1,2-己二醇等多元醇及其衍生物;甲醇、乙醇、丙醇、异丙醇、环己醇等醇系溶剂;二甲亚砜等砜系溶剂;N-甲基-2-吡咯烷酮、N,N-二甲基甲酰胺等酰胺系溶剂。另外,这些溶剂当中,最好含有一种以上如下的有机溶剂,即,具有包括至少1个以上苯环的构造,并且熔点在0℃以下,沸点在100℃以上。 Examples of solvents used in the ink composition of the present invention include chloroform, dichloromethane, 1,2-dichloroethane, 1,1,2-trichloroethane, chlorobenzene, o-dichlorobenzene, and other chlorine solvents; tetrahydrofuran, dioxane and other ether solvents; toluene, xylene and other aromatic hydrocarbon solvents; cyclohexane, methylcyclohexane, n-pentane, n-hexane, n-heptane, n-octane Aliphatic hydrocarbon-based solvents such as alkane, n-nonane, and n-decane; ketone-based solvents such as acetone, methyl ethyl ketone, and cyclohexanone; ester-based solvents such as ethyl acetate, butyl acetate, and ethyl cellosolve acetate; Glycol, Ethylene Glycol Monobutyl Ether, Ethylene Glycol Monoethyl Ether, Ethylene Glycol Monomethyl Ether, Dimethoxyethane, Propylene Glycol, Diethoxymethane, Triethylene Glycol Monoethyl Ether, Glycerin, 1,2- Polyols such as hexanediol and their derivatives; alcohol solvents such as methanol, ethanol, propanol, isopropanol, and cyclohexanol; sulfone solvents such as dimethyl sulfoxide; N-methyl-2-pyrrolidone, N, N-dimethylformamide and other amide solvents. In addition, among these solvents, it is preferable to contain one or more organic solvents having a structure including at least one benzene ring and having a melting point of 0°C or lower and a boiling point of 100°C or higher. the
作为溶剂的种类,从本发明的高分子化合物向有机溶剂中的溶解性、成膜时的均一性、粘度特性等观点考虑,优选芳香族烃系溶剂、脂肪族烃系溶剂、酯系溶剂、酮系溶剂,优选甲苯、二甲苯、乙基苯、二乙基苯、三甲基苯、、正丙基苯、异丙基苯、正丁基苯、异丁基苯、仲丁基苯、苯甲醚、乙氧基苯、1-甲基萘、环己烷、环己酮、环己基苯、联二环己烷、环己烯基环己酮、正庚基环己烷、正己基环己烷、甲基苯甲酸酯、2-丙基环己酮、2-庚酮、3-庚酮、4-庚酮、2-辛酮、2-壬酮、2-癸酮、二环己基酮,更优选含有二甲苯、苯甲醚、、环己基苯、联二环己烷甲基苯甲酸酯当中的至少一种。 As the type of solvent, from the viewpoints of solubility of the polymer compound of the present invention in an organic solvent, uniformity during film formation, viscosity characteristics, etc., aromatic hydrocarbon solvents, aliphatic hydrocarbon solvents, ester solvents, Ketone solvents, preferably toluene, xylene, ethylbenzene, diethylbenzene, trimethylbenzene, , n-propylbenzene, cumene, n-butylbenzene, isobutylbenzene, sec-butylbenzene, anisole, ethoxybenzene, 1-methylnaphthalene, cyclohexane, cyclohexanone, cyclohexanone Hexylbenzene, bicyclohexyl, cyclohexenylcyclohexanone, n-heptylcyclohexane, n-hexylcyclohexane, methyl benzoate, 2-propylcyclohexanone, 2-heptanone, 3-heptanone, 4-heptanone, 2-octanone, 2-nonanone, 2-decanone, dicyclohexyl ketone, more preferably containing xylene, anisole, , cyclohexylbenzene, and dicyclohexyl methyl benzoate at least one.
从成膜性的观点或元件特性等观点考虑,油墨组合物中的溶剂的种类 优选为2种以上,更优选为2~3种,进一步优选为2种。 The types of solvents in the ink composition are preferably 2 or more types, more preferably 2 to 3 types, and even more preferably 2 types from the viewpoint of film-forming properties and device characteristics. the
在油墨组合物中含有2种溶剂的情况下,其中的1种溶剂在25℃下也可以是固体状态。从成膜性的观点考虑,优选1种溶剂是沸点在180℃以上的溶剂,另一种溶剂是沸点在180℃以下的溶剂,更优选1种溶剂是沸点在200℃以上的溶剂,另一种溶剂是沸点在180℃以下的溶剂。另外,从粘度的观点考虑,优选在2种溶剂中,在60℃下,都溶解0.2wt%以上的本发明的高分子化合物,优选在2种溶剂当中的1种溶剂中,在25℃溶解0.2wt%以上的本发明的高分子化合物。 When two types of solvents are contained in the ink composition, one of the solvents may be in a solid state at 25°C. From the viewpoint of film-forming properties, it is preferable that one solvent has a boiling point of 180° C. or higher, the other solvent has a boiling point of 180° C. or lower, more preferably one solvent has a boiling point of 200° C. or higher, and the other solvent has a boiling point of 200° C. or higher. The first solvent is a solvent with a boiling point below 180°C. In addition, from the viewpoint of viscosity, it is preferable to dissolve 0.2 wt% or more of the polymer compound of the present invention in both of the two solvents at 60°C, and it is preferable to dissolve it in one of the two solvents at 25°C. 0.2 wt% or more of the polymer compound of the present invention. the
在油墨组合物中含有3种溶剂的情况下,其中的1~2种溶剂在25℃也可以是固体状态。从成膜性的观点考虑,优选3种溶剂当中的至少一种溶剂是沸点在180℃以上的溶剂,至少一种溶剂是沸点在180℃以下的溶剂,更优选3种溶剂当中的至少一种溶剂是沸点在200℃以上300℃以下的溶剂,至少一种溶剂是沸点在180℃以下的溶剂。另外,从粘度的观点考虑,优选在3种溶剂当中的2种溶剂中,在60℃下,溶解0.2wt%以上的本发明的高分子化合物,优选在3种溶剂当中的1种溶剂中,在25℃溶解0.2wt%以上的本发明的高分子化合物。 When the ink composition contains three solvents, one or two of them may be in a solid state at 25°C. From the viewpoint of film-forming properties, at least one of the three solvents is preferably a solvent having a boiling point of 180° C. or higher, at least one of the solvents is a solvent having a boiling point of 180° C. or lower, more preferably at least one of the three solvents The solvents have a boiling point of not less than 200°C and not more than 300°C, and at least one solvent has a boiling point of not more than 180°C. In addition, from the viewpoint of viscosity, it is preferable to dissolve 0.2 wt % or more of the polymer compound of the present invention in two of the three solvents at 60° C., preferably in one of the three solvents, 0.2 wt% or more of the polymer compound of the present invention is dissolved at 25°C. the
在油墨组合物中含有2种以上溶剂的情况下,从粘度及成膜性的观点考虑,优选沸点最高的溶剂为油墨组合物的全部溶剂的重量的40~90wt%,更优选为50~90wt%,进一步优选为65~85wt%。 When the ink composition contains two or more solvents, from the viewpoint of viscosity and film-forming properties, the solvent with the highest boiling point is preferably 40 to 90 wt%, more preferably 50 to 90 wt%, based on the weight of all solvents in the ink composition %, more preferably 65 to 85 wt%. the
作为本发明的油墨组合物,从粘度及成膜性的观点考虑,优选由苯甲醚及联二环己烷构成的组合物、由苯甲醚及环己基苯构成的组合物、由二甲苯及联二环己烷构成的组合物、由二甲苯及环己基苯构成的组合物、由 及甲基苯甲酸酯构成的组合物。 As the ink composition of the present invention, a composition composed of anisole and bicyclohexane, a composition composed of anisole and cyclohexylbenzene, a composition composed of xylene Compositions composed of dicyclohexyl and bicyclohexane, compositions composed of xylene and cyclohexylbenzene, compositions composed of and methyl benzoate compositions.
本发明的油墨组合物所可以含有的添加剂当中,作为空穴输送材料,可以举出聚乙烯基咔唑或其衍生物、聚硅烷或其衍生物、在侧链或主链中具有芳香族胺的聚硅氧烷衍生物、吡唑啉衍生物、芳基胺衍生物、茋衍生物、三苯基二胺衍生物、聚苯胺或其衍生物、聚噻吩或其衍生物、聚吡咯或其衍生物、聚(对亚苯基亚乙烯基)或其衍生物或聚(2,5-亚噻吩基亚乙烯基)或其衍生物。 Among the additives that may be contained in the ink composition of the present invention, examples of hole transport materials include polyvinylcarbazole or its derivatives, polysilane or its derivatives, and aromatic amines in the side chain or main chain. Polysiloxane derivatives, pyrazoline derivatives, arylamine derivatives, stilbene derivatives, triphenyldiamine derivatives, polyaniline or its derivatives, polythiophene or its derivatives, polypyrrole or its derivatives, poly(p-phenylenevinylene) or derivatives thereof, or poly(2,5-thienylenevinylene) or derivatives thereof. the
作为电子输送材料,可以举出噁二唑衍生物、蒽醌二甲烷或其衍生物、 苯醌或其衍生物、萘醌或其衍生物、蒽醌或其衍生物、四氰基蒽醌二甲烷或其衍生物、芴衍生物、二苯基二氰基乙烯或其衍生物、苯酚合苯醌衍生物或8-羟基喹啉或其衍生物的金属络合物、聚喹啉或其衍生物、聚喹喔啉或其衍生物、聚芴或其衍生物。 Examples of electron transport materials include oxadiazole derivatives, anthraquinone dimethane or derivatives thereof, benzoquinone or derivatives thereof, naphthoquinone or derivatives thereof, anthraquinone or derivatives thereof, tetracyanoanthraquinone di Methane or its derivatives, fluorene derivatives, diphenyldicyanoethylene or its derivatives, phenoquinone derivatives or metal complexes of 8-hydroxyquinoline or its derivatives, polyquinoline or its derivatives substances, polyquinoxaline or its derivatives, polyfluorene or its derivatives. the
作为发光材料,可以举出萘衍生物、蒽或其衍生物、苝或其衍生物、聚甲炔系、呫吨系、香豆素系、花青系等色素类、8-羟基喹啉或其衍生物的金属络合物、芳香族胺、四苯基环戊二烯或其衍生物或者四苯基丁二烯或其衍生物等。 Examples of light-emitting materials include naphthalene derivatives, anthracene or derivatives thereof, perylene or derivatives thereof, polymethine-based, xanthene-based, coumarin-based, cyanine-based pigments, 8-hydroxyquinoline or Metal complexes of its derivatives, aromatic amines, tetraphenylcyclopentadiene or its derivatives, or tetraphenylbutadiene or its derivatives, etc. the
本发明的油墨组合物(溶液)除了本发明的高分子化合物以外,也可以含有用于调节粘度及/或表面张力的添加剂。作为该添加剂,只要适当地组合使用用于提高粘度的高分子量的高分子化合物(增稠剂)或弱溶剂、用于降低粘度的低分子量的化合物、用于降低表面张力的表面活性剂等即可。 The ink composition (solution) of the present invention may contain additives for adjusting viscosity and/or surface tension in addition to the polymer compound of the present invention. As the additive, a high-molecular-weight high-molecular-weight compound (thickener) or weak solvent for increasing the viscosity, a low-molecular-weight compound for reducing the viscosity, a surfactant for reducing the surface tension, etc. may be used in combination appropriately. Can. the
作为上述的高分子量的高分子化合物,只要是与本发明的高分子化合物同样地在溶剂中是可溶性的,且不会妨碍发光或电荷输送的即可。例如可以使用高分子量的聚苯乙烯、聚甲基丙烯酸甲酯或本发明的高分子化合物当中的分子量大的物质等。重均分子量优选在50万以上,更优选在100万以上。 As the above-mentioned high-molecular-weight polymer compound, it is sufficient that it is soluble in a solvent similarly to the polymer compound of the present invention, and does not hinder light emission or charge transport. For example, high-molecular-weight polystyrene, polymethyl methacrylate, or a substance having a large molecular weight among the high-molecular compounds of the present invention can be used. The weight average molecular weight is preferably at least 500,000, more preferably at least 1 million. the
也可以将弱溶剂作为增稠剂使用。即,通过少量地添加相对于溶液中的固体成分的弱溶剂,就可以提高粘度。在出于该目的添加弱溶剂的情况下,只要在溶液中的固体成分不会析出的范围中,选择溶剂的种类和添加量即可。 Weak solvents can also be used as thickeners. That is, the viscosity can be increased by adding a small amount of weak solvent to the solid content in the solution. When adding a weak solvent for this purpose, the type and amount of the solvent may be selected within a range in which the solid content in the solution does not precipitate. the
另外,为了改善保存稳定性,本发明的油墨组合物(溶液)除了本发明的高分子化合物以外,还可以含有抗氧化剂。作为抗氧化剂,只要是与本发明的高分子化合物同样地在溶剂中是可溶性的,且不会妨碍发光或电荷输送的物质即可,可以例示出酚系抗氧化剂、磷系抗氧化剂等。 In addition, in order to improve storage stability, the ink composition (solution) of the present invention may contain an antioxidant in addition to the polymer compound of the present invention. As the antioxidant, as long as it is soluble in a solvent like the polymer compound of the present invention and does not interfere with light emission or charge transport, examples include phenolic antioxidants, phosphorus antioxidants, and the like. the
从本发明的高分子化合物向溶剂中的溶解性的观点考虑,溶剂的溶解度参数与高分子化合物的溶解度参数的差优选在10以下,更优选在7以下。 From the viewpoint of the solubility of the polymer compound of the present invention in a solvent, the difference between the solubility parameter of the solvent and the solubility parameter of the polymer compound is preferably 10 or less, more preferably 7 or less. the
溶剂的溶解度参数与本发明的高分子化合物的溶解度参数可以利用 「溶剂手册(讲谈社刊,1976年)」中记载的方法求得。 The solubility parameter of the solvent and the solubility parameter of the polymer compound of the present invention can be obtained by the method described in "Solvent Handbook (Kodansha, 1976)". the
作为本发明的高分子LED,可以举出在阴极与发光层之间设置了电子输送层的高分子LED、在阳极与发光层之间设置了空穴输送层的高分子LED、在阴极与发光层之间设置了电子输送层并且在阳极与发光层之间设置了空穴输送层的高分子LED等。 Examples of the polymer LED of the present invention include polymer LEDs in which an electron transport layer is provided between the cathode and the luminescent layer, polymer LEDs in which a hole transport layer is provided between the anode and the luminescent layer, and polymer LEDs in which the cathode and the luminescent layer are provided. Polymer LEDs and the like in which an electron transport layer is provided between layers and a hole transport layer is provided between the anode and the light emitting layer. the
例如,具体来说可以例示出以下的a)~d)的构造。 For example, the following structures a) to d) can be specifically illustrated. the
a)阳极/发光层/阴极 a) anode/luminescent layer/cathode
b)阳极/空穴输送层/发光层/阴极 b) anode/hole transport layer/emissive layer/cathode
c)阳极/发光层/电子输送层/阴极 c) anode/luminescent layer/electron transport layer/cathode
d)阳极/空穴输送层/发光层/电子输送层/阴极 d) Anode/hole transport layer/luminescent layer/electron transport layer/cathode
(这里,/表示各层被相邻地叠层的意思。以下相同。) (Here, / means that each layer is stacked adjacently. The same applies below.)
作为本发明的高分子LED,还包括在空穴输送层及/或电子输送层中含有本发明的高分子化合物的LED。 The polymer LED of the present invention also includes an LED containing the polymer compound of the present invention in a hole transport layer and/or an electron transport layer. the
在将本发明的高分子化合物用于空穴输送层中的情况下,本发明的高分子化合物优选为含有空穴输送性基的高分子化合物,作为其具体例,可以例示出与芳香族胺的共聚物、与茋的共聚物等。 When the polymer compound of the present invention is used in the hole transport layer, the polymer compound of the present invention is preferably a polymer compound containing a hole transport group. Copolymers, copolymers with stilbene, etc. the
另外,在将本发明的高分子化合物用于电子输送层中的情况下,本发明的高分子化合物优选为含有电子输送性基团的高分子化合物,作为其具体例,可以例示出与噁二唑的共聚物、与三唑的共聚物、与喹啉的共聚物、与喹喔啉的共聚物、与苯并噻二唑的共聚物等。 In addition, when the polymer compound of the present invention is used in the electron transport layer, the polymer compound of the present invention is preferably a polymer compound containing an electron transport group, and specific examples thereof include dioxane Copolymers of azoles, copolymers with triazoles, copolymers with quinoline, copolymers with quinoxaline, copolymers with benzothiadiazole, etc. the
在本发明的高分子LED具有空穴输送层的情况下,作为所用的空穴输送材料,可以例示出聚乙烯基咔唑或其衍生物、聚硅烷或其衍生物、在侧链或主链中具有芳香族胺的聚硅氧烷衍生物、吡唑啉衍生物、芳基胺衍生物、茋衍生物、三苯基二胺衍生物、聚苯胺或其衍生物、聚噻吩或其衍生物、聚吡咯或其衍生物、聚(对亚苯基亚乙烯基)或其衍生物或聚(2,5-亚噻吩基亚乙烯基)或其衍生物。 When the polymer LED of the present invention has a hole-transporting layer, examples of the hole-transporting material used include polyvinylcarbazole or its derivatives, polysilane or its derivatives, side chain or main chain Polysiloxane derivatives with aromatic amines, pyrazoline derivatives, arylamine derivatives, stilbene derivatives, triphenyldiamine derivatives, polyaniline or its derivatives, polythiophene or its derivatives , polypyrrole or derivatives thereof, poly(p-phenylenevinylene) or derivatives thereof, or poly(2,5-thienylenevinylene) or derivatives thereof. the
具体来说,作为该空穴输送材料,可以例示出特开昭63-70257号公报、相同的63-175860号公报、特开平2-135359号公报、相同的2-135361号公报、相同的2-209988号公报、相同的3-37992号公报、相同的3-152184号公报中所记载的材料等。 Specifically, examples of the hole transport material include JP-A-63-70257, JP-A-63-175860, JP-2-135359, JP-2-135361, and 2 -Materials described in Gazette No. 209988, the same Gazette No. 3-37992, the same Gazette No. 3-152184, etc. the
它们当中,作为在空穴输送层中所用的空穴输送材料,优选聚乙烯基咔唑或其衍生物、聚硅烷或其衍生物、在侧链或主链中具有芳香族胺化合物基团的聚硅氧烷衍生物、聚苯胺或其衍生物、聚噻吩或其衍生物、聚(对亚苯基亚乙烯基)或其衍生物或聚(2,5-亚噻吩基亚乙烯基)或其衍生物等高分子空穴输送材料,更优选聚乙烯基咔唑或其衍生物、聚硅烷或其衍生物、在侧链或主链中具有芳香族胺的聚硅氧烷衍生物。 Among them, as the hole-transporting material used in the hole-transporting layer, polyvinylcarbazole or its derivatives, polysilane or its derivatives, those having an aromatic amine compound group in the side chain or the main chain are preferable. Polysiloxane derivatives, polyaniline or derivatives thereof, polythiophene or derivatives thereof, poly(p-phenylenevinylene) or derivatives thereof or poly(2,5-thienylenevinylene) or Polymer hole transport materials such as derivatives thereof are more preferably polyvinylcarbazole or derivatives thereof, polysilane or derivatives thereof, and polysiloxane derivatives having aromatic amines in side chains or main chains. the
另外,作为低分子化合物的空穴输送材料,可以例示出吡唑啉衍生物、芳基胺衍生物、茋衍生物、三苯基二胺衍生物。在低分子空穴输送材料的情况下,优选将其分散于高分子粘合剂中使用。 In addition, examples of the hole-transporting material of the low-molecular compound include pyrazoline derivatives, arylamine derivatives, stilbene derivatives, and triphenyldiamine derivatives. In the case of a low-molecular hole transport material, it is preferably used dispersed in a polymer binder. the
作为所混合的高分子粘合剂,优选不会明显阻碍电荷输送的粘合剂,另外可以优选使用对可见光的吸收不强的粘合剂。作为该高分子粘合剂,可以例示出聚(N-乙烯基咔唑)、聚苯胺或其衍生物、聚噻吩或其衍生物、聚(对亚苯基亚乙烯基)或其衍生物、聚(2,5-亚噻吩基亚乙烯基)或其衍生物、聚碳酸酯、聚丙烯酸酯、聚丙烯酸甲酯、聚甲基丙烯酸甲酯、聚苯乙烯、聚氯乙烯、聚硅氧烷等。 The polymer binder to be mixed is preferably one that does not significantly hinder charge transport, and one that does not strongly absorb visible light can be preferably used. Examples of the polymer binder include poly(N-vinylcarbazole), polyaniline or derivatives thereof, polythiophene or derivatives thereof, poly(p-phenylene vinylene) or derivatives thereof, Poly(2,5-thienylenevinylene) or its derivatives, polycarbonate, polyacrylate, polymethyl acrylate, polymethyl methacrylate, polystyrene, polyvinyl chloride, polysiloxane wait. the
聚乙烯基咔唑或其衍生物例如可以由乙烯基单体利用阳离子聚合或自由基聚合来得到。 Polyvinylcarbazole or its derivatives can be obtained, for example, from vinyl monomers by cationic polymerization or radical polymerization. the
作为聚硅烷或其衍生物,可以例示出化学综述(Chem.Rev.)第89卷、1359页(1989年)、英国专利GB2300196号公报说明书中记载的化合物等。合成方法也可以使用它们中所记载的方法,然而特别优选使用Kipping法。 Examples of polysilanes or derivatives thereof include compounds described in Chem. Rev. Vol. 89, p. 1359 (1989), British Patent No. GB2300196, and the like. As the synthesis method, the methods described therein can also be used, but the Kipping method is particularly preferably used. the
聚硅氧烷或其衍生物由于在硅氧烷骨架构造中基本上没有空穴输送性,因此优选使用在侧链或主链中具有上述低分子空穴输送材料的构造的物质。特别可以例示出在侧链或主链中具有空穴输送性的芳香族胺的物质。 Since polysiloxane or its derivative has substantially no hole transport property in the siloxane skeleton structure, it is preferable to use a substance having the structure of the above-mentioned low-molecular hole transport material in the side chain or the main chain. In particular, a substance having a hole-transporting aromatic amine in a side chain or a main chain can be exemplified. the
对于空穴输送层的成膜的方法没有特别限制,但是如果是低分子空穴输送材料,则可以例示出由其与高分子粘合剂的混合溶液成膜的方法。另外如果是高分子空穴输送材料,则可以例示出由溶液成膜的方法。 The method of forming a film of the hole transport layer is not particularly limited, but if it is a low-molecular hole transport material, a method of forming a film from a mixed solution thereof and a polymer binder can be exemplified. In addition, if it is a polymeric hole transport material, a method of forming a film from a solution can be exemplified. the
作为由溶液成膜中所用的溶剂,只要是溶解空穴输送材料的溶剂,就没有特别限制。作为该溶剂,可以例示出氯仿、二氯甲烷、二氯乙烷等氯 系溶剂;四氢呋喃等醚系溶剂;甲苯、二甲苯等芳香族烃系溶剂;丙酮、甲乙酮等酮系溶剂;乙酸乙酯、乙酸丁酯、乙基溶纤剂乙酸酯等酯系溶剂。 The solvent used for film formation from a solution is not particularly limited as long as it dissolves the hole transport material. Examples of the solvent include chlorine-based solvents such as chloroform, dichloromethane, and dichloroethane; ether-based solvents such as tetrahydrofuran; aromatic hydrocarbon-based solvents such as toluene and xylene; ketone-based solvents such as acetone and methyl ethyl ketone; , butyl acetate, ethyl cellosolve acetate and other ester solvents. the
作为由溶液成膜的方法,可以使用溶液的旋转涂覆法、浇注法、显微凹版涂覆法、凹版涂覆法、棒涂覆法、辊涂法、绕线棒涂覆法、浸渍涂覆法、喷雾涂覆法、丝网印刷法、苯胺印刷法、胶版印刷法、喷墨印刷法等涂布法。 As a method of forming a film from a solution, spin coating method, casting method, micro gravure coating method, gravure coating method, rod coating method, roll coating method, wire rod coating method, dip coating method, etc. Coating method, spray coating method, screen printing method, flexographic printing method, offset printing method, inkjet printing method and other coating methods. the
作为空穴输送层的膜厚,最佳值根据所用的材料而不同,只要以使驱动电压和发光效率达到适度的值的方式选择即可,然而至少需要不会产生针孔的厚度,当太厚时,则元件的驱动电压变高,因而不够理想。所以,作为该空穴输送层的膜厚,例如为1nm到1μm,优选为2nm~500nm,更优选为5nm~200nm。 As the film thickness of the hole transport layer, the optimum value varies depending on the material used, and it is sufficient to select such that the driving voltage and luminous efficiency become moderate values. However, at least a thickness that does not cause pinholes is required. When thick, the driving voltage of the element becomes high, which is not preferable. Therefore, the film thickness of the hole transport layer is, for example, 1 nm to 1 μm, preferably 2 nm to 500 nm, and more preferably 5 nm to 200 nm. the
在本发明的高分子LED具有电子输送层的情况下,作为所用的电子输送材料,可以使用公知的材料,可以例示出噁二唑衍生物、蒽醌二甲烷或其衍生物、苯醌或其衍生物、萘醌或其衍生物、蒽醌或其衍生物、四氰基蒽醌二甲烷或其衍生物、芴衍生物、二苯基二氰基乙烯或其衍生物、二苯酚合苯醌衍生物或8-羟基喹啉或其衍生物的金属络合物、聚喹啉或其衍生物、聚喹喔啉或其衍生物、聚芴或其衍生物等。 When the polymer LED of the present invention has an electron transport layer, known materials can be used as the electron transport material used, and oxadiazole derivatives, anthraquinone dimethane or derivatives thereof, benzoquinone or its derivatives can be exemplified. Derivatives, naphthoquinone or its derivatives, anthraquinone or its derivatives, tetracyanoanthraquinone dimethane or its derivatives, fluorene derivatives, diphenyldicyanoethylene or its derivatives, diphenoquinone Derivatives or metal complexes of 8-hydroxyquinoline or its derivatives, polyquinoline or its derivatives, polyquinoxaline or its derivatives, polyfluorene or its derivatives, etc. the
具体来说,可以例示出特开昭63-70257号公报、相同的63-175860号公报、特开平2-135359号公报、相同的2-135361号公报、相同的2-209988号公报、相同的3-37992号公报、相同的3-152184号公报中所记载的材料等。 Specifically, JP-A-63-70257, JP-A-63-175860, JP-2-135359, JP-2-135361, JP-2-209988, and Materials described in Publication No. 3-37992, the same Publication No. 3-152184, and the like. the
它们当中,优选噁二唑衍生物、苯醌或其衍生物、萘醌或其衍生物、蒽醌或其衍生物或8-羟基喹啉或其衍生物的金属络合物、聚喹啉或其衍生物、聚喹喔啉或其衍生物、聚芴或其衍生物等,更优选2-(4-联苯基)-5-(4-叔丁基苯基)-1,3,4-噁二唑、苯醌、蒽醌、三(8-羟基喹啉)铝、聚喹啉。 Among them, metal complexes of oxadiazole derivatives, benzoquinone or derivatives thereof, naphthoquinone or derivatives thereof, anthraquinone or derivatives thereof, or 8-hydroxyquinoline or derivatives thereof, polyquinoline or Its derivatives, polyquinoxaline or its derivatives, polyfluorene or its derivatives, etc., more preferably 2-(4-biphenyl)-5-(4-tert-butylphenyl)-1,3,4 -Oxadiazoles, benzoquinones, anthraquinones, tris(8-quinolinolate)aluminum, polyquinolines. the
作为电子输送层的成膜法没有特别限制,然而如果是低分子电子输送材料,则可以例示出由粉末的真空蒸镀法或由溶液或熔融状态开始的成膜的方法,如果是高分子电子输送材料,则可以例示出由溶液或熔融状态成膜的方法。在由溶液或熔融状态成膜时,也可以并用上述的高分子粘合剂。 The film-forming method as the electron-transporting layer is not particularly limited, but if it is a low-molecular-weight electron-transporting material, a vacuum evaporation method from a powder or a film-forming method from a solution or a molten state can be exemplified. For conveying materials, a method of forming a film from a solution or a molten state can be exemplified. When forming a film from a solution or a molten state, the above-mentioned polymer binder may be used in combination. the
作为由溶液的成膜中所用的溶剂,只要是溶解电子输送材料及/或高分子粘合剂的溶剂,就没有特别限制。作为该溶剂,可以例示出氯仿、二氯甲烷、二氯乙烷等氯系溶剂;四氢呋喃等醚系溶剂;甲苯、二甲苯等芳香族烃系溶剂;丙酮、甲乙酮等酮系溶剂;乙酸乙酯、乙酸丁酯、乙基溶纤剂乙酸酯等酯系溶剂。 The solvent used for film formation from a solution is not particularly limited as long as it dissolves the electron transport material and/or the polymer binder. Examples of the solvent include chlorine-based solvents such as chloroform, dichloromethane, and dichloroethane; ether-based solvents such as tetrahydrofuran; aromatic hydrocarbon-based solvents such as toluene and xylene; ketone-based solvents such as acetone and methyl ethyl ketone; , butyl acetate, ethyl cellosolve acetate and other ester solvents. the
作为由溶液或熔融状态的成膜方法,可以使用旋转涂覆法、浇注法、显微凹版涂覆法、凹版涂覆法、棒涂覆法、辊涂法、绕线棒涂覆法、浸渍涂覆法、喷雾涂覆法、丝网印刷法、苯胺印刷法、胶版印刷法、喷墨印刷法等涂布法。 As a film-forming method from a solution or a molten state, spin coating, casting, microgravure coating, gravure coating, rod coating, roll coating, wire-wound bar coating, dipping, etc., can be used. Coating method, spray coating method, screen printing method, flexographic printing method, offset printing method, inkjet printing method and other coating methods. the
作为电子输送层的膜厚,最佳值根据所用的材料而不同,只要以使驱动电压和发光效率达到适度的值的方式选择即可,然而至少需要不会产生针孔的厚度,当太厚时,则元件的驱动电压变高,因而不够理想。所以,作为该电子输送层的膜厚,例如为1nm到1μm,优选为2nm~500nm,更优选为5nm~200nm。 As the film thickness of the electron transport layer, the optimum value differs depending on the material used, as long as the driving voltage and luminous efficiency can be selected in such a way as to achieve an appropriate value, but at least a thickness that does not cause pinholes is required, and when it is too thick , the driving voltage of the element becomes high, which is not ideal. Therefore, the film thickness of the electron transport layer is, for example, 1 nm to 1 μm, preferably 2 nm to 500 nm, and more preferably 5 nm to 200 nm. the
另外,与电极相邻地设置的电荷输送层当中,具有改善从电极注入电荷的效率的功能,并具有降低元件的驱动电压效果的层有时-般性地特称为电荷注入层(空穴注入层、电子注入层)。 In addition, among the charge transport layers provided adjacent to the electrodes, the layer that has the function of improving the efficiency of injecting charges from the electrodes and has the effect of lowering the driving voltage of the device is sometimes generally referred to as a charge injection layer (hole injection layer). layer, electron injection layer). the
另外,为了提高与电极的密接性或改善从电极中的电荷注入,也可以与电极相邻地设置上述电荷注入层或膜厚在2nm以下的绝缘层,另外,为了提高界面的密接性或防止混合等,也可以在电荷输送层或发光层的界面插入薄的缓冲层。 In addition, in order to improve the adhesion with the electrode or improve the charge injection from the electrode, the above-mentioned charge injection layer or an insulating layer with a film thickness of 2 nm or less may be provided adjacent to the electrode. In addition, in order to improve the adhesion of the interface or prevent A thin buffer layer may also be inserted at the interface of the charge transport layer or the light emitting layer by mixing or the like. the
对于所叠层的层的顺序或数目及各层的厚度,可以考虑发光效率或元件寿命而适当地使用。 The order and number of layers to be laminated and the thickness of each layer can be appropriately used in consideration of luminous efficiency and device life. the
本发明的高分子化合物也可以制成有机半导体薄膜而作为高分子电场效应晶体管使用。作为高分子电场效应晶体管的构造,通常是与由高分子构成的活性层相接地设置源极及漏极,继而夹隔与活性层相接的绝缘层地设置栅极即可。 The polymer compound of the present invention can also be made into an organic semiconductor thin film and used as a polymer field effect transistor. As a structure of a polymer field effect transistor, generally, a source and a drain are provided in contact with an active layer made of a polymer, and then a gate is provided with an insulating layer in contact with the active layer. the
高分子电场效应晶体管通常形成于支撑基板上。作为支撑基板的材质,只要是不妨碍作为电场效应晶体管的特性,就没有特别限制,可以使用玻璃基板、柔性的薄膜基板或塑料基板。 Polymer field effect transistors are usually formed on a support substrate. The material of the supporting substrate is not particularly limited as long as it does not interfere with the characteristics of the field effect transistor, and a glass substrate, a flexible film substrate, or a plastic substrate can be used. the
电场效应晶体管可以利用公知的方法,例如特开平5-110069号公报记载的方法来制造。 The field effect transistor can be produced by a known method, for example, the method described in JP-A-5-110069. the
在形成活性层之时,使用有机溶剂可溶性的高分子的做法在制造上是非常有利的,因而优选。作为由在有机溶剂中溶解了高分子的溶液的成膜方法,可以使用旋转涂覆法、浇注法、显微凹版涂覆法、凹版涂覆法、棒涂覆法、辊涂法、绕线棒涂覆法、浸渍涂覆法、喷雾涂覆法、丝网印刷法、苯胺印刷法、胶版印刷法、喷墨印刷法等涂布法。 When forming the active layer, it is very advantageous in terms of production to use a polymer soluble in an organic solvent, and thus it is preferable. As a film-forming method from a solution in which a polymer is dissolved in an organic solvent, spin coating, casting, microgravure coating, gravure coating, rod coating, roll coating, wire winding, etc., can be used. Coating methods such as bar coating, dip coating, spray coating, screen printing, flexographic printing, offset printing, and inkjet printing. the
优选在制成高分子电场效应晶体管后,密封而成的密封高分子电场效应晶体管。这样,就可以将高分子电场效应晶体管与大气隔断,抑制高分子电场效应晶体管的特性的降低。 It is preferably a sealed polymer field effect transistor formed by sealing the polymer field effect transistor after it is manufactured. In this way, the polymer field effect transistor can be blocked from the atmosphere, and the deterioration of the characteristics of the polymer field effect transistor can be suppressed. the
作为密封的方法,可以举出用UV固化树脂、热固化树脂或无机的SiONx膜等覆盖的方法、将玻璃板或薄膜用UV固化树脂、热固化树脂等贴合的方法等。为了有效地进行与大气的隔断,制成高分子电场效应晶体管后最好不向大气暴露地(例如在干燥的氮气气氛中、真空中等)进行直到密封的工序。 Examples of sealing methods include a method of covering with UV curable resin, thermosetting resin, or inorganic SiONx film, and a method of bonding glass plates or films with UV curable resin, thermosetting resin, and the like. In order to effectively block the atmosphere from the atmosphere, it is preferable to carry out the steps up to sealing without exposing the polymer field effect transistor to the atmosphere (for example, in a dry nitrogen atmosphere, in a vacuum, etc.). the
本发明中,作为设置了电荷注入层(电子注入层、空穴注入层)的高分子LED,可以举出与阴极相邻地设置了电荷注入层的高分子LED、与阳极相邻地设置了电荷注入层的高分子LED。 In the present invention, examples of polymer LEDs provided with a charge injection layer (electron injection layer, hole injection layer) include polymer LEDs provided with a charge injection layer adjacent to a cathode, and polymer LEDs provided with a charge injection layer adjacent to an anode. Polymer LED with charge injection layer. the
例如,具体来说可以举出以下的e)~p)的构造。 For example, the following structures e)-p) are specifically mentioned. the
e)阳极/电荷注入层/发光层/阴极 e) Anode/Charge Injection Layer/Emitting Layer/Cathode
f)阳极/发光层/电荷注入层/阴极 f) anode/luminescent layer/charge injection layer/cathode
g)阳极/电荷注入层/发光层/电荷注入层/阴极 g) anode/charge injection layer/light-emitting layer/charge injection layer/cathode
h)阳极/电荷注入层/空穴输送层/发光层/阴极 h) anode/charge injection layer/hole transport layer/light-emitting layer/cathode
i)阳极/空穴输送层/发光层/电荷注入层/阴极 i) Anode/hole transport layer/emissive layer/charge injection layer/cathode
j)阳极/电荷注入层/空穴输送层/发光层/电荷注入层/阴极 j) anode/charge injection layer/hole transport layer/emissive layer/charge injection layer/cathode
k)阳极/电荷注入层/发光层/电子输送层/阴极 k) anode/charge injection layer/emissive layer/electron transport layer/cathode
l)阳极/发光层/电子输送层/电荷注入层/阴极 l) anode/luminescent layer/electron transport layer/charge injection layer/cathode
m)阳极/电荷注入层/发光层/电子输送层/电荷注入层/阴极 m) anode/charge injection layer/light-emitting layer/electron transport layer/charge injection layer/cathode
n)阳极/电荷注入层/空穴输送层/发光层/电子输送层/阴极 n) anode/charge injection layer/hole transport layer/light emitting layer/electron transport layer/cathode
o)阳极/空穴输送层/发光层/电子输送层/电荷注入层/阴极 o) anode/hole transport layer/light emitting layer/electron transport layer/charge injection layer/cathode
p)阳极/电荷注入层/空穴输送层/发光层/电子输送层/电荷注入层/阴极 p) anode/charge injection layer/hole transport layer/light emitting layer/electron transport layer/charge injection layer/cathode
作为电荷注入层的具体的例子,可以例示出含有导电性高分子的层;设于阳极与空穴输送层之间,含有具有阳极材料和空穴输送层中所含的空穴输送材料的中间的值的离子化电位的材料的层;设于阴极与电子输送层之间,含有具有阴极材料和电子输送层中所含的电子输送材料的中间的值的电子亲和力的材料的层等。 As a specific example of the charge injection layer, a layer containing a conductive polymer can be exemplified; it is provided between the anode and the hole transport layer, and contains an intermediate layer having an anode material and a hole transport material contained in the hole transport layer. A layer of a material having an ionization potential of a value of ; a layer of a material having an electron affinity of an intermediate value between the cathode material and the electron transport material contained in the electron transport layer, etc., provided between the cathode and the electron transport layer. the
在上述电荷注入层是含有导电性高分子的层的情况下,该导电性高分子的电导率优选为10-5S/cm以上103以下,为了减小发光像素之间的漏电流,更优选10-5S/cm以上102以下,进一步优选10-5S/cm以上101以下。 When the above-mentioned charge injection layer is a layer containing a conductive polymer, the conductivity of the conductive polymer is preferably not less than 10 −5 S/cm and not more than 10 3 . In order to reduce the leakage current between light-emitting pixels, more It is preferably 10 -5 S/cm or more and 10 2 or less, more preferably 10 -5 S/cm or more and 10 1 or less.
在上述电荷注入层是含有导电性高分子的层的情况下,该导电性高分子的电导率优选为10-5S/cm以上103S/cm以下,为了减小发光像素之间的漏电流,更优选10-5S/cm以上102S/cm以下,进一步优选10-5S/cm以上101S/cm以下。 When the above-mentioned charge injection layer is a layer containing a conductive polymer, the conductivity of the conductive polymer is preferably not less than 10 −5 S/cm and not more than 10 3 S/cm. In order to reduce leakage between light-emitting pixels, The current is more preferably from 10 -5 S/cm to 10 2 S/cm, and still more preferably from 10 -5 S/cm to 10 1 S/cm.
通常来说,为了将该导电性高分子的电导率设为10-5S/cm以上103 以下,而在该导电性高分子中掺杂适量的离子。 Generally, the conductive polymer is doped with an appropriate amount of ions so that the electrical conductivity of the conductive polymer is not less than 10 −5 S/cm and not more than 10 3 .
对于所掺杂的离子的种类,如果是空穴注入层,则为阴离子,如果是电子注入层,则为阳离子。作为阴离子的例子,可以例示出聚苯乙烯磺酸根离子、烷基苯磺酸根离子、樟脑磺酸根离子等,作为阳离子的例子,可以例示出锂离子、钠离子、钾离子、四丁基铵离子等。 The type of ions to be doped is an anion in the case of a hole injection layer, and a cation in the case of an electron injection layer. Examples of anions include polystyrenesulfonate ions, alkylbenzenesulfonate ions, and camphorsulfonate ions, and examples of cations include lithium ions, sodium ions, potassium ions, and tetrabutylammonium ions. wait. the
作为电荷注入层的膜厚,例如为1nm~100nm,优选2nm~50nm。 The film thickness of the charge injection layer is, for example, 1 nm to 100 nm, preferably 2 nm to 50 nm. the
电荷注入层中所用的材料只要根据其与电极或相邻的层的材料的关系适当地选择即可,可以例示出聚苯胺及其衍生物、聚噻吩及其衍生物、聚吡咯及其衍生物、聚苯撑亚乙烯基及其衍生物、聚亚噻吩亚乙烯基及其衍生物、聚喹啉及其衍生物、聚喹喔啉及其衍生物、在主链或侧链中含有芳香族胺构造的聚合物等导电性高分子、金属酞菁(铜酞菁等)、碳等。 The material used for the charge injection layer may be appropriately selected according to its relationship with the electrode or the material of the adjacent layer, and polyaniline and its derivatives, polythiophene and its derivatives, polypyrrole and its derivatives can be exemplified. , polyphenylene vinylene and its derivatives, polythiophene vinylene and its derivatives, polyquinoline and its derivatives, polyquinoxaline and its derivatives, containing aromatics in the main chain or side chain Conductive polymers such as polymers with an amine structure, metal phthalocyanines (copper phthalocyanines, etc.), carbon, etc. the
膜厚在2nm以下的绝缘层是具有使得电荷注入更为容易的功能的层。作为上述绝缘层的材料,可以举出金属氟化物、金属氧化物、有机绝缘材料等。作为设置了膜厚在2nm以下的绝缘层的高分子LED,可以举出与阴极相邻地设置了膜厚在2nm以下的绝缘层的高分子LED、与阳极相邻地设置了膜厚在2nm以下的绝缘层的高分子LED。 The insulating layer having a film thickness of 2 nm or less has a function of facilitating charge injection. Examples of the material of the insulating layer include metal fluorides, metal oxides, organic insulating materials and the like. Examples of polymer LEDs provided with an insulating layer with a film thickness of 2 nm or less include polymer LEDs provided with an insulating layer with a film thickness of 2 nm or less adjacent to the cathode, and polymer LEDs provided with an insulating layer with a film thickness of 2 nm or less adjacent to the anode. Polymer LED below the insulating layer. the
具体来说,可以举出以下的q)~ab)的构造。 Specifically, the following structures q) to ab) can be mentioned. the
q)阳极/膜厚在2nm以下的绝缘层/发光层/阴极 q) anode/insulating layer/luminescent layer/cathode with film thickness below 2nm
r)阳极/发光层/膜厚在2nm以下的绝缘层/阴极 r) anode/luminescent layer/insulating layer/cathode with film thickness below 2nm
s)阳极/膜厚在2nm以下的绝缘层/发光层/膜厚在2nm以下的绝缘层/阴极 s) Anode/insulating layer with film thickness below 2nm/luminescent layer/insulating layer with film thickness below 2nm/cathode
t)阳极/膜厚在2nm以下的绝缘层/空穴输送层/发光层/阴极 t) anode/insulating layer with film thickness below 2nm/hole transport layer/luminescent layer/cathode
u)阳极/空穴输送层/发光层/膜厚在2nm以下的绝缘层/阴极 u) Anode/hole transport layer/luminescent layer/insulating layer with film thickness below 2nm/cathode
v)阳极/膜厚在2nm以下的绝缘层/空穴输送层/发光层/膜厚在2nm以下的绝缘层/阴极 v) Anode/insulating layer with film thickness below 2nm/hole transport layer/light-emitting layer/insulating layer with film thickness below 2nm/cathode
w)阳极/膜厚在2nm以下的绝缘层/发光层/电子输送层/阴极 w) anode/insulating layer with film thickness below 2nm/luminescent layer/electron transport layer/cathode
x)阳极/发光层/电子输送层/膜厚在2nm以下的绝缘层/阴极 x) Anode/luminescent layer/electron transport layer/insulating layer with film thickness below 2nm/cathode
y)阳极/膜厚在2nm以下的绝缘层/发光层/电子输送层/膜厚在2nm以下的绝缘层/阴极 y) Anode/insulating layer with film thickness below 2nm/luminescent layer/electron transport layer/insulating layer with film thickness below 2nm/cathode
z)阳极/膜厚在2nm以下的绝缘层/空穴输送层/发光层/电子输送层/阴极 z) Anode/insulating layer with film thickness below 2nm/hole transport layer/luminescent layer/electron transport layer/cathode
aa)阳极/空穴输送层/发光层/电子输送层/膜厚在2nm以下的绝缘层/阴极 aa) anode/hole transport layer/luminescent layer/electron transport layer/insulating layer with film thickness below 2nm/cathode
ab)阳极/膜厚在2nm以下的绝缘层/空穴输送层/发光层/电子输送层/膜厚在2nm以下的绝缘层/阴极 ab) Anode/insulating layer with a film thickness of less than 2nm/hole transport layer/luminescent layer/electron transport layer/insulating layer with a film thickness of less than 2nm/cathode
形成本发明的高分子LED的基板只要是在形成电极、形成有机物的层之时不发生变化的基板即可,例如可以例示出玻璃、塑料、高分子薄膜、硅基板等。在不透明的基板的情况下,反面的电极优选为透明或半透明的。 The substrate on which the polymer LED of the present invention is formed can be any substrate that does not change when electrodes and organic layers are formed, and examples thereof include glass, plastic, polymer films, silicon substrates, and the like. In the case of an opaque substrate, the opposite electrode is preferably transparent or translucent. the
通常来说,本发明的高分子LED所具有的阳极及阴极的至少一方是透明或半透明的。阳极侧优选为透明或半透明的。 Generally, at least one of the anode and the cathode of the polymer LED of the present invention is transparent or translucent. The anode side is preferably transparent or translucent. the
作为该阳极的材料,可以使用导电性的金属氧化物膜、半透明的金属薄膜等。具体来说,可以使用氧化铟、氧化锌、氧化锡及作为它们的复合体的铟锡氧化物(ITO)、使用由铟锌氧化物等构成的导电性玻璃制成的膜(NESA等)、金、铂、银、铜等,优选ITO、铟锌氧化物、氧化锡。作为制作方法,可以举出真空蒸镀法、溅射法、离子镀法、镀膜法等。另外,作为该阳极,也可以使用聚苯胺或其衍生物、聚噻吩或其衍生物等的有机 的透明导电膜。 As the material of the anode, a conductive metal oxide film, a semitransparent metal thin film, or the like can be used. Specifically, indium oxide, zinc oxide, tin oxide, and indium tin oxide (ITO) as a composite of these, films made of conductive glass made of indium zinc oxide (NESA, etc.), Gold, platinum, silver, copper, etc., preferably ITO, indium zinc oxide, tin oxide. Examples of the production method include a vacuum vapor deposition method, a sputtering method, an ion plating method, a coating method, and the like. In addition, as the anode, an organic transparent conductive film such as polyaniline or its derivatives, polythiophene or its derivatives, or the like can also be used. the
阳极的膜厚可以考虑光的透过性和电导率而适当地选择,例如为10nm到10μm,优选为20nm~1μm,进一步优选为50nm~500nm。 The film thickness of the anode can be appropriately selected in consideration of light permeability and electrical conductivity, and is, for example, 10 nm to 10 μm, preferably 20 nm to 1 μm, more preferably 50 nm to 500 nm. the
另外,在阳极上,为了使电荷注入更为容易,也可以设置由酞菁衍生物、导电性高分子、碳等构成的层;或由金属氧化物或金属氟化物、有机绝缘材料等构成的平均膜厚在2nm以下的层。 In addition, on the anode, in order to make charge injection easier, a layer composed of phthalocyanine derivatives, conductive polymers, carbon, etc. can also be provided; or a layer composed of metal oxides or metal fluorides, organic insulating materials, etc. A layer with an average film thickness of 2 nm or less. the
作为本发明的高分子LED中所用的阴极材料,优选功函数小的材料。例如可以举出锂、钠、钾、铷、铯、铍、镁、钙、锶、钡、铝、钪、钒、锌、钇、铟、铈、钐、铕、铽、镱等金属;及它们当中的2种以上的合金;或者它们当中的1种以上与金、银、铂、铜、锰、钛、钴、镍、钨、锡当中的1种以上的合金;石墨或石墨层间化合物等。作为合金的例子,可以举出镁-银合金、镁-铟合金、镁-铝合金、铟-银合金、锂-铝合金、锂-镁合金、锂-铟合金、钙-铝合金等。也可以将阴极设为2层以上的叠层构造。 As the cathode material used in the polymer LED of the present invention, a material having a small work function is preferable. Examples include lithium, sodium, potassium, rubidium, cesium, beryllium, magnesium, calcium, strontium, barium, aluminum, scandium, vanadium, zinc, yttrium, indium, cerium, samarium, europium, terbium, ytterbium and other metals; and their Alloys of two or more of them; or alloys of more than one of them with gold, silver, platinum, copper, manganese, titanium, cobalt, nickel, tungsten, and tin; graphite or graphite interlayer compounds, etc. . Examples of alloys include magnesium-silver alloys, magnesium-indium alloys, magnesium-aluminum alloys, indium-silver alloys, lithium-aluminum alloys, lithium-magnesium alloys, lithium-indium alloys, and calcium-aluminum alloys. The cathode may also have a laminated structure of two or more layers. the
阴极的膜厚可以考虑电导率或耐久性而适当地选择,例如为10nm到10μm,优选为20nm~1μm,进一步优选为50nm~500nm。 The film thickness of the cathode can be appropriately selected in consideration of electrical conductivity and durability, and is, for example, 10 nm to 10 μm, preferably 20 nm to 1 μm, more preferably 50 nm to 500 nm. the
作为阴极的制作方法,可以使用真空蒸镀法、溅射法或热压接金属薄膜的层压法等。另外,在阴极与有机物层之间,也可以设置由导电性高分子构成的层,或由金属氧化物或金属氟化物、有机绝缘材料等构成的平均膜厚在2nm以下的层,在阴极制成后,也可以安装保护该高分子LED的保护层。为了长期稳定地使用该高分子LED,最好安装保护层及/或保护外罩,以保护元件免受外部影响。 As a method for producing the cathode, a vacuum vapor deposition method, a sputtering method, a lamination method of thermocompression bonding a metal thin film, or the like can be used. In addition, between the cathode and the organic layer, a layer made of conductive polymers, or a layer made of metal oxides or metal fluorides, organic insulating materials, etc., with an average film thickness of 2 nm or less may be provided. After completion, a protective layer to protect the polymer LED can also be installed. In order to use the polymer LED stably for a long time, it is preferable to install a protective layer and/or a protective cover to protect the element from external influences. the
作为该保护层,可以使用高分子化合物、金属氧化物、金属氟化物、金属硼化物等。另外,作为保护外罩,可以使用玻璃板、对表面实施了低透水率处理的塑料板等,优选使用将该外罩以热固化树脂或光固化树脂与元件基板贴合而密闭的方法。如果使用隔块来维持空间,则很容易防止元件受到损伤。如果向该空间注入氮气或氩气之类的惰性气体,则可以防止阴极的氧化,另外,通过在该空间内设置氧化钡等干燥剂,则可以容易地抑制在制造工序中吸附的水分损害元件。它们当中,优选采用任何一种以上的方法。 As the protective layer, polymer compounds, metal oxides, metal fluorides, metal borides, and the like can be used. In addition, a glass plate, a plastic plate whose surface has been subjected to a low water permeability treatment, etc. can be used as a protective cover, and a method of sealing the cover by bonding a thermosetting resin or a photosetting resin to the element substrate is preferably used. If spacers are used to maintain space, it is easy to prevent damage to components. If an inert gas such as nitrogen or argon is injected into this space, the oxidation of the cathode can be prevented. In addition, by installing a desiccant such as barium oxide in this space, it is possible to easily prevent the moisture absorbed in the manufacturing process from damaging the element. . Among them, any one or more methods are preferably employed. the
本发明的高分子LED可以用于面状光源、分段显示装置、点矩阵显示装置、液晶显示装置(例如液晶显示装置的背光灯)中。 The polymer LED of the present invention can be used in planar light sources, segmented display devices, dot matrix display devices, and liquid crystal display devices (such as backlights of liquid crystal display devices). the
为了使用本发明的高分子LED获得面状的发光,只要将面状的阳极与阴极重合地配置即可。另外,为了获得图案状的发光,有在上述面状的发光元件的表面设置设有图案状的窗口的掩模的方法、将非发光部的有机物层极厚地形成而实质上使之不发光的方法、将阳极或阴极的任意一方或双方电极制成图案状的方法。通过用它们中的任意的方法来形成图案,将某些电极可以独立地On/OFF地配置,就可以获得能够显示数字或文字、简单的记号等的分段型的显示元件。另外,为了制成点矩阵元件,只要将阳极和阴极都制成条纹状而正交地配置即可。利用分别涂布多种发光颜色不同的高分子荧光体的方法、使用滤色片或荧光变换过滤片的方法,就可以实现部分彩色显示、多彩色显示。点矩阵元件既可以是无源驱动,也可以与TFT等组合而进行有源驱动。这些显示元件可以作为计算机、电视、携带终端、携带电话、导航器、摄像机的取景器等显示装置使用。 In order to obtain planar light emission using the polymer LED of the present invention, it is only necessary to arrange planar anodes and cathodes so as to overlap each other. In addition, in order to obtain pattern-like light emission, there is a method of providing a mask with a pattern-like window on the surface of the above-mentioned planar light-emitting element, and a method of forming an organic layer of a non-light-emitting part extremely thick so that it does not emit light substantially. method, a method of patterning either one or both electrodes of the anode or the cathode. By forming a pattern by any of these methods and disposing certain electrodes independently on/off, a segmented display element capable of displaying numerals, letters, simple signs, etc. can be obtained. In addition, in order to form a dot matrix device, both the anode and the cathode may be arranged in stripes and orthogonally arranged. Partial color display and multi-color display can be realized by coating a plurality of polymer phosphors with different luminescent colors, or by using color filters or fluorescence conversion filters. The dot matrix element can be driven passively, or combined with TFT etc. for active driving. These display elements can be used as display devices such as computers, televisions, portable terminals, mobile phones, navigators, and video camera viewfinders. the
另外,上述面状的发光元件为自发光薄型,可以作为液晶显示装置的背光灯用的面状光源或面状的照明用光源合适地使用。另外,如果使用柔性的基板,则也可以作为曲面状的光源或显示装置使用。 In addition, the above-mentioned planar light-emitting element is self-luminous and thin, and can be suitably used as a planar light source for a backlight of a liquid crystal display device or a planar light source for illumination. In addition, if a flexible substrate is used, it can also be used as a curved light source or a display device. the
下面,为了进一步详细地说明本发明而给出实施例,然而本发明并不限定于它们。 Hereinafter, examples are given to explain the present invention in more detail, but the present invention is not limited to them. the
(数均分子量及重均分子量) (number average molecular weight and weight average molecular weight)
这里,对于数均分子量及重均分子量,利用GPC(岛津制作所制:LC-10Avp)求得了聚苯乙烯换算的数均分子量及重均分子量。将所测定的聚合物溶解于四氢呋喃中,使之达到约0.5wt%的浓度,向GPC中注入50μL。GPC的移动相使用四氢呋喃,以0.6mL/min的流速流动。色谱柱是将2根TSKgel SuperHM-H(Toso制)和1根TSKgel SuperH2000(Toso制)串联地连接。在检测器中使用了示差折射率检测器(岛津制作所制:RID-10A)。 Here, the number average molecular weight and weight average molecular weight were calculated|required for the number average molecular weight and weight average molecular weight of polystyrene conversion by GPC (manufactured by Shimadzu Corporation: LC-10Avp). The polymer to be measured was dissolved in tetrahydrofuran to a concentration of about 0.5% by weight, and 50 μL was injected into the GPC. Tetrahydrofuran was used as the mobile phase of GPC at a flow rate of 0.6 mL/min. As a column, two pieces of TSKgel SuperHM-H (manufactured by Toso) and one piece of TSKgel SuperH2000 (manufactured by Toso) were connected in series. As a detector, a differential refractive index detector (manufactured by Shimadzu Corporation: RID-10A) was used. the
(荧光光谱) (Fluorescence Spectroscopy)
荧光光谱的测定是以下面的方法进行的。在石英上旋转涂覆聚合物的0.8wt%溶液,制作了聚合物的薄膜。以350nm的波长激发该薄膜,使用 荧光分光光度计(堀场制作所制Fluorolog)测定了荧光光谱。为了获得薄膜中的相对的荧光强度,以水的拉曼线的强度作为标准,在光谱测定范围中将以波数绘制的荧光光谱积分,使用分光光度计(Varian公司制Cary5E)测定。求得用激发波长下的吸光度除后的值。 The measurement of the fluorescence spectrum was carried out in the following manner. Thin films of the polymer were produced by spin coating a 0.8 wt% solution of the polymer on quartz. The thin film was excited at a wavelength of 350 nm, and the fluorescence spectrum was measured using a fluorescence spectrophotometer (Fluorolog manufactured by Horiba, Ltd.). In order to obtain the relative fluorescence intensity in the thin film, the fluorescence spectrum plotted by the wavenumber was integrated in the spectral measurement range using the Raman line intensity of water as a standard, and measured using a spectrophotometer (Cary5E manufactured by Varian). The value obtained by dividing the absorbance at the excitation wavelength was obtained. the
(HPLC测定) (HPLC determination)
测定仪器:Agilent 1100LC Measuring instrument: Agilent 1100LC
测定条件:L-Column ODS、5μm、2.1mm×150mm; Measuring conditions: L-Column ODS, 5μm, 2.1mm×150mm;
A液:乙腈、B液:THF Liquid A: Acetonitrile, Liquid B: THF
梯度 Gradient
B液: Liquid B:
0%(60min.)→10%up/min→100%(10min)、 0% (60min.) → 10% up/min → 100% (10min),
样品浓度:5.0mg/mL(THF溶液)、 Sample concentration: 5.0mg/mL (THF solution),
注入量:1μL Injection volume: 1μL
检测波长:350nm Detection wavelength: 350nm
合成例1 Synthesis example 1
(1-溴-4-叔丁基-2,6-二甲基苯的合成) (Synthesis of 1-bromo-4-tert-butyl-2,6-dimethylbenzene)
在惰性气氛下,向500ml的三口烧瓶中加入乙酸225g,添加5-叔丁基-间二甲苯。接下来添加了溴31.2g后,在15~20℃下反应3小时。 Under an inert atmosphere, 225 g of acetic acid was added to a 500 ml three-necked flask, and 5-tert-butyl-m-xylene was added. Next, after adding 31.2 g of bromine, it was made to react at 15-20 degreeC for 3 hours. the
将反应液加入500ml水中,过滤析出的沉淀。用250ml水清洗2次,得到了白色的固体34.2g。 The reaction solution was added to 500 ml of water, and the precipitate was filtered. It was washed twice with 250 ml of water to obtain 34.2 g of a white solid. the
<N,N’-二苯基-N,N’-双(4-叔丁基-2,6-二甲基苯基)-1,4-苯二胺的合成> <Synthesis of N,N'-diphenyl-N,N'-bis(4-tert-butyl-2,6-dimethylphenyl)-1,4-phenylenediamine>
在惰性气氛下,向100ml的三口烧瓶中加入脱气了的脱水甲苯36ml,添加三(叔丁基)膦0.63g。接下来加入三(二亚苄基丙酮)二钯0.41g、1-溴-4-叔丁基-2,6-二甲基苯9.6g、叔丁氧基钠5.2g、N,N’-二苯基-1,4-苯二胺4.7g后,在100℃反应3小时。 Under an inert atmosphere, 36 ml of degassed dehydrated toluene was added to a 100 ml three-necked flask, and 0.63 g of tri(tert-butyl)phosphine was added. Next, add 0.41 g of tris(dibenzylideneacetone) dipalladium, 9.6 g of 1-bromo-4-tert-butyl-2,6-dimethylbenzene, 5.2 g of sodium tert-butoxide, N,N'- After 4.7 g of diphenyl-1,4-phenylenediamine was reacted at 100° C. for 3 hours. the
将反应液加到300ml饱和食盐水中,用加温为约50℃的氯仿300ml萃取。蒸馏除去溶剂后,添加甲苯100ml,加热至固体溶解,自然冷却后,过滤沉淀,得到了白色的固体9.9g。 The reaction solution was added to 300 ml of saturated brine, and extracted with 300 ml of chloroform heated to about 50°C. After the solvent was distilled off, 100 ml of toluene was added, the mixture was heated until the solid was dissolved, and after standing to cool, the precipitate was filtered to obtain 9.9 g of a white solid. the
<N,N’-双(4-溴苯基)-N,N’-双(4-叔丁基-2,6-二甲基苯基)-1,4-苯二胺的合成> <Synthesis of N,N'-bis(4-bromophenyl)-N,N'-bis(4-tert-butyl-2,6-dimethylphenyl)-1,4-phenylenediamine>
在惰性气氛下,向100ml的三口烧瓶中加入脱水N,N-二甲基甲酰胺350ml,在溶解了N’-二苯基-N,N’-双(4-叔丁基-2,6-二甲基苯基)-1,4-苯二胺5.2g后,在冰浴中滴加N-溴琥珀酰亚胺3.5g/N,N-二甲基甲酰胺溶液,使之反应一昼夜。 Under an inert atmosphere, add dehydrated N,N-dimethylformamide 350ml to a 100ml three-necked flask, after dissolving N'-diphenyl-N,N'-bis(4-tert-butyl-2,6 After 5.2 g of -dimethylphenyl)-1,4-phenylenediamine, add N-bromosuccinimide 3.5 g/N, N-dimethylformamide solution dropwise in an ice bath, and make it react for a day and night . the
向反应液中加入水150ml,过滤析出的沉淀,用甲醇50ml清洗2次,得到了白色的固体4.4g。 150 ml of water was added to the reaction liquid, and the deposited precipitate was filtered and washed twice with 50 ml of methanol to obtain 4.4 g of a white solid. the
1H-NMR(300MHz/THF-d8) 1 H-NMR (300MHz/THF-d8)
δ(ppm)=1.3[s,18H]、2.0[s,12H]、6.6~6.7[d,4H]、6.8~6.9[br,4H]、7.1[s,4H]、7.2~7.3[d,4H] δ(ppm)=1.3[s, 18H], 2.0[s, 12H], 6.6~6.7[d, 4H], 6.8~6.9[br, 4H], 7.1[s, 4H], 7.2~7.3[d, 4H]
MS(FD+)M+738 MS(FD + )M + 738
合成例2 Synthesis example 2
<N,N’-二苯基-N,N’-双(4-叔丁基-2,6-二甲基苯基)-联苯胺的合成> <Synthesis of N, N'-diphenyl-N, N'-bis(4-tert-butyl-2,6-dimethylphenyl)-benzidine>
在惰性气氛下,向300ml的三口烧瓶中加入脱水甲苯1660ml,添加N,N’-二苯基联苯胺275.0g、4-叔丁基-2,6-二甲基溴苯449.0g。接下来添加了三(二亚苄基丙酮)二钯7.48g、叔丁氧基钠196.4g后,添加三(叔丁基)膦5.0g。其后,在105℃反应7小时。 Under an inert atmosphere, 1660 ml of dehydrated toluene was added to a 300 ml three-necked flask, and 275.0 g of N,N'-diphenylbenzidine and 449.0 g of 4-tert-butyl-2,6-dimethylbromobenzene were added. Next, 7.48 g of tris(dibenzylideneacetone)dipalladium and 196.4 g of sodium tert-butoxide were added, and then 5.0 g of tris(tert-butyl)phosphine was added. Then, it was made to react at 105 degreeC for 7 hours. the
向反应液中加入甲苯2000ml,铈硅石(cerite)过滤,将滤液用1000ml水清洗3次后,浓缩至700ml。向其中添加甲苯/甲醇(1∶1)溶液1600ml,过滤析出的晶体,用甲醇清洗。得到了白色的固体479.4g。 2,000 ml of toluene was added to the reaction liquid, filtered through cerite, and the filtrate was washed three times with 1,000 ml of water, and concentrated to 700 ml. To this was added 1600 ml of a toluene/methanol (1:1) solution, and the precipitated crystals were filtered and washed with methanol. 479.4 g of a white solid were obtained. the
<N,N’-双(4-溴苯基)-N,N’-双(4-叔丁基-2,6-二甲基苯基)-联苯胺的合成> <Synthesis of N,N'-bis(4-bromophenyl)-N,N'-bis(4-tert-butyl-2,6-dimethylphenyl)-benzidine>
在惰性气氛下,在4730g氯仿中溶解了上述N,N’-二苯基-N,N’-双(4-叔丁基-2,6-二甲基苯基)-联苯胺472.8g后,在遮光及冰浴下将N-溴琥珀酰亚胺281.8g分成12份用1小时加入,反应3小时。 Under an inert atmosphere, after dissolving 472.8 g of the above-mentioned N, N'-diphenyl-N, N'-bis(4-tert-butyl-2,6-dimethylphenyl)-benzidine in 4730 g of chloroform 281.8 g of N-bromosuccinimide was divided into 12 parts and added in 1 hour under shading and ice bathing, and reacted for 3 hours. the
向反应液中加入氯仿1439ml,过滤,将滤液的氯仿溶液用5%硫代硫酸钠2159ml清洗,蒸馏除去甲苯,得到了白色晶体。将所得的白色晶体用甲苯/甲醇重结晶,得到了白色晶体678.7g。 1439 ml of chloroform was added to the reaction liquid, followed by filtration, and the chloroform solution of the filtrate was washed with 2159 ml of 5% sodium thiosulfate, and the toluene was distilled off to obtain white crystals. The obtained white crystals were recrystallized from toluene/methanol to obtain 678.7 g of white crystals. the
MS(APCI(+)):(M+H)+815.2 MS (APCI (+)): (M+H) + 815.2
实施例1<化合物B的合成> Embodiment 1<The synthesis of compound B>
<化合物A的合成> <Synthesis of Compound A>
(化合物A) (Compound A)
向反应容器中加入离子交换水31ml,在搅拌的同时一点点地加入氢氧化钠29g(727mmol),完全地溶解。将体系内用氩气置换,加入甲苯30ml和1,2,3,10b-tetrahydrofluoranthene 5.0g(24mmol),搅拌而溶解。接下来加入溴化四丁基铵2.3g(7.3mmol)和辛基溴9.4g(48mmol),在40℃反应3小时。用甲苯和水分液,将有机层萃取后,用硫酸钠干燥。蒸馏除去溶剂后,通过用以己烷作为展开溶剂的硅胶色谱精制,得到了淡黄色的晶体6.45g。 31 ml of ion-exchanged water was added to the reaction vessel, and 29 g (727 mmol) of sodium hydroxide was added little by little while stirring to completely dissolve it. The system was replaced with argon, and 30 ml of toluene and 5.0 g (24 mmol) of 1,2,3,10b-tetrahydrofluoranthene were added, stirred and dissolved. Next, 2.3 g (7.3 mmol) of tetrabutylammonium bromide and 9.4 g (48 mmol) of octyl bromide were added, and it was made to react at 40 degreeC for 3 hours. The organic layer was extracted with toluene and water, and dried over sodium sulfate. After distilling off the solvent, it was purified by silica gel chromatography using hexane as a developing solvent to obtain 6.45 g of pale yellow crystals. the
MS(APCI(+)):318([M+H]+) MS (APCI (+)): 318 ([M+H] + )
1H-NMR(300MHz/CDCl3)δ7.69(1H,d)、7.50(1H,d)、7.40(1H,d)、7.34~7.22(3H,m)、7.03(1H,d)、3.07~2.96(1H,m)、2.77~2.70(1H,m)、2.37~2.18(2H,m)、1.95~1.74(3H,m)、1.27~1.04(11H,m)、0.84~0.69(5H,m) 1 H-NMR (300MHz/CDCl 3 ) δ7.69(1H,d), 7.50(1H,d), 7.40(1H,d), 7.34~7.22(3H,m), 7.03(1H,d), 3.07 ~2.96(1H, m), 2.77~2.70(1H, m), 2.37~2.18(2H, m), 1.95~1.74(3H, m), 1.27~1.04(11H, m), 0.84~0.69(5H, m)
(化合物B的合成) (Synthesis of compound B)
(化合物B) (compound B)
向氩气置换的反应容器中加入化合物A5.8g(18mmol)和乙酸∶二氯甲烷=1∶1的混合溶剂115ml,在室温下搅拌溶解。接下来加入三溴化苄基三甲基铵14g(36mmol),在搅拌的同时添加氯化锌,直至三溴化苄基三甲基铵完全溶解。在用HPCL追踪反应的同时,适当地添加三溴化苄基三甲基铵和氯化锌。反应结束后,用氯仿和水分液,萃取有机层,水洗2次后,用碳酸钾水溶液中和。用硫酸钠干燥后,蒸馏除去溶剂,用以己烷作为展开溶剂的硅胶色谱精制后,从乙醇∶己烷=10∶1的混合溶剂中重结晶,作为白色的粉末得到了化合物B5.08g。 5.8 g (18 mmol) of compound A and 115 ml of a mixed solvent of acetic acid:dichloromethane = 1:1 were added to a reaction vessel replaced with argon, and stirred and dissolved at room temperature. Next, 14 g (36 mmol) of benzyltrimethylammonium tribromide was added, and zinc chloride was added with stirring until benzyltrimethylammonium tribromide was completely dissolved. While following the reaction with HPCL, benzyltrimethylammonium tribromide and zinc chloride were added as appropriate. After the reaction, the organic layer was extracted with chloroform and water, washed twice with water, and then neutralized with potassium carbonate aqueous solution. After drying over sodium sulfate, the solvent was distilled off, purified by silica gel chromatography using hexane as a developing solvent, and then recrystallized from a mixed solvent of ethanol:hexane = 10:1 to obtain 5.08 g of compound B as a white powder. the
MS(APPI(+)):476(M+) MS (APPI (+)): 476 (M + )
1H-NMR(300MHz/CDCl3)δ7.53~7.43(4H,m)、7.33(1H,d)、2.90~2.84(2H,m)、2.33~2.22(2H,m)、2.05~1.96(1H,m)、1.83~1.64(2H,m)、1.32~1.05(13H,m)、0.85~0.81(3H,m) 1 H-NMR (300MHz/CDCl 3 ) δ7.53~7.43(4H, m), 7.33(1H, d), 2.90~2.84(2H, m), 2.33~2.22(2H, m), 2.05~1.96( 1H, m), 1.83~1.64(2H, m), 1.32~1.05(13H, m), 0.85~0.81(3H, m)
实施例2 Example 2
(高分子化合物1的合成) (Synthesis of polymer compound 1)
将化合物B(0.1g)、2,2’-联吡啶(0.089g)溶解于脱水的四氢呋喃19mL中后,在氮气气氛下,向该溶液中加入双(1,5-环辛二烯)镍(0){Ni(COD)2}(0.156g),升温至60℃,反应3小时。将该反应液冷却至室温,向25%氨水1mL/甲醇19mL/离子交换水19mL混合溶液中滴加,搅拌1小时后,过滤析出的沉淀,减压干燥。所得的聚合物(以后称作高分子化合物1)的产量为0.08g。另外,聚苯乙烯换算的数均分子量为2.9×104,重均分子量为6.1×104。 Compound B (0.1 g) and 2,2'-bipyridine (0.089 g) were dissolved in 19 mL of dehydrated tetrahydrofuran, and bis(1,5-cyclooctadiene)nickel was added to the solution under a nitrogen atmosphere (0) {Ni(COD) 2 } (0.156g), heated up to 60°C, and reacted for 3 hours. The reaction solution was cooled to room temperature, added dropwise to a mixed solution of 25% ammonia water 1 mL/methanol 19 mL/ion-exchanged water 19 mL, stirred for 1 hour, and the precipitate was filtered and dried under reduced pressure. The yield of the obtained polymer (hereinafter referred to as polymer compound 1) was 0.08 g. In addition, the polystyrene-equivalent number average molecular weight was 2.9×10 4 , and the weight average molecular weight was 6.1×10 4 .
实施例3 Example 3
<高分子化合物2的合成> <Synthesis of Polymer Compound 2>
将化合物B(0.557g)、N,N’-双(4-溴苯基)-N,N’-双(4-叔丁基-2,6-二甲基苯基)-1,4-苯二胺(0.096g)、2,2’-联吡啶(0.548g)溶解于脱水的四氢呋喃140mL中后,用氩气鼓泡而将体系内进行氮气置换。升温至60℃后,在氮气气氛下,向该溶液中加入双(1,5-二氯辛二烯)镍(0){Ni(COD)2}(0.965g),搅拌,反应3小时。将该反应液冷却至室温,向25%氨水5mL/甲醇140mL/离子交换水140mL混合溶液中滴加,搅拌1小时后,过滤析出的沉淀,减压干燥,溶解于甲苯40ml中。溶解后,添加硅藻土(radiolite)1.6g,搅拌30分钟,过滤不溶物。将所得的滤液穿过氧化铝柱而进行精制。然后添加5.2%盐酸水80mL,搅拌3小时后,除去水层。然后添加4%氨水80mL,搅拌2小时后除去水层。继而向有机层中添加离子交换水约80mL,搅拌1小时后,除去水层。其后,将有机层注入甲醇160ml中,搅拌1小时,过滤析出的沉淀,减压干燥。所得的聚合物(以后称作高分子化合物2)的产量为0.33g。另外,聚苯乙烯换算的数均分子量及重均分子量分别Mn=1.6×104,Mw=8.7×104。 Compound B (0.557g), N, N'-bis(4-bromophenyl)-N,N'-bis(4-tert-butyl-2,6-dimethylphenyl)-1,4- After dissolving phenylenediamine (0.096 g) and 2,2'-bipyridine (0.548 g) in 140 mL of dehydrated tetrahydrofuran, the inside of the system was replaced with nitrogen by bubbling argon. After heating up to 60°C, bis(1,5-dichlorooctadiene)nickel(0){Ni(COD) 2 }(0.965g) was added to the solution under nitrogen atmosphere, stirred and reacted for 3 hours. The reaction solution was cooled to room temperature, added dropwise to a mixed solution of 25% ammonia 5mL/methanol 140mL/ion-exchanged water 140mL, stirred for 1 hour, and the precipitate was filtered, dried under reduced pressure, and dissolved in toluene 40ml. After dissolution, 1.6 g of diatomaceous earth (radiolite) was added, stirred for 30 minutes, and insoluble matter was filtered. The resulting filtrate was purified by passing through an alumina column. Then, 80 mL of 5.2% aqueous hydrochloric acid was added, and after stirring for 3 hours, the aqueous layer was removed. Then, 80 mL of 4% ammonia water was added, and the aqueous layer was removed after stirring for 2 hours. Next, about 80 mL of ion-exchanged water was added to the organic layer, and after stirring for 1 hour, the aqueous layer was removed. Thereafter, the organic layer was poured into 160 ml of methanol, stirred for 1 hour, and the deposited precipitate was filtered and dried under reduced pressure. The yield of the obtained polymer (hereinafter referred to as polymer compound 2) was 0.33 g. In addition, the polystyrene-equivalent number average molecular weight and weight average molecular weight were Mn=1.6×10 4 and Mw=8.7×10 4 , respectively.
实施例4 Example 4
<高分子化合物3的合成> <Synthesis of Polymer Compound 3>
将化合物B(0.433g)、N,N’-双(4-溴苯基)-N,N’-双(4-叔丁基-2,6-二甲基苯基)-联苯胺(0.318g)、2,2’-联吡啶(0.548g)溶解于脱水的四氢呋喃140mL中后,用氩气鼓泡而将体系内进行氮气置换。升温至60℃后,在氮气气氛下,向该溶液中加入双(1,5-二氯辛二烯)镍(0){Ni(COD)2}(0.965g),搅拌,反应3小时。将该反应液冷却至室温,向25%氨水5mL/甲醇140mL/离子交换水140mL混合溶液中滴加,搅拌1小时后,过滤析出的沉淀,减压干燥,溶解于甲苯40ml中。溶解后,添加硅藻土1.6g,搅拌30分钟,过滤不溶物。将所得的滤液穿过氧化铝柱而进行精制。然后添加5.2%盐酸水80mL,搅拌3小时后,除去水层。然后添加4%氨水80mL,搅拌2小时后除去水层。继而向有机层中添加离子交换水约80mL,搅拌1小时后,除去水层。其后,将有机层注入到甲醇160ml中,搅拌1小时,过滤析出的沉淀,减压干燥。所得的聚合物(以后称作高分子化合物3)的产量为0.46g。另外,聚苯乙烯换 算的数均分子量及重均分子量分别Mn=1.0×104,Mw=6.1×104。 Compound B (0.433g), N, N'-bis(4-bromophenyl)-N,N'-bis(4-tert-butyl-2,6-dimethylphenyl)-benzidine (0.318 g) After dissolving 2,2'-bipyridine (0.548 g) in 140 mL of dehydrated tetrahydrofuran, the inside of the system was replaced with nitrogen by bubbling argon. After heating up to 60°C, bis(1,5-dichlorooctadiene)nickel(0){Ni(COD) 2 }(0.965g) was added to the solution under nitrogen atmosphere, stirred and reacted for 3 hours. The reaction solution was cooled to room temperature, added dropwise to a mixed solution of 25% ammonia 5mL/methanol 140mL/ion-exchanged water 140mL, stirred for 1 hour, and the precipitate was filtered, dried under reduced pressure, and dissolved in toluene 40ml. After dissolution, 1.6 g of diatomaceous earth was added, stirred for 30 minutes, and insoluble matter was filtered. The resulting filtrate was purified by passing through an alumina column. Then, 80 mL of 5.2% aqueous hydrochloric acid was added, and after stirring for 3 hours, the aqueous layer was removed. Then, 80 mL of 4% ammonia water was added, and the aqueous layer was removed after stirring for 2 hours. Next, about 80 mL of ion-exchanged water was added to the organic layer, and after stirring for 1 hour, the aqueous layer was removed. Thereafter, the organic layer was poured into 160 ml of methanol, stirred for 1 hour, and the deposited precipitate was filtered and dried under reduced pressure. The yield of the obtained polymer (hereinafter referred to as polymer compound 3) was 0.46 g. In addition, the polystyrene-equivalent number average molecular weight and weight average molecular weight were Mn=1.0×10 4 and Mw=6.1×10 4 , respectively.
实施例5 Example 5
<高分子化合物4的合成> <Synthesis of Polymer Compound 4>
将化合物B(0.588g)、N,N’-双(4-溴苯基)-N,N’-双(4-叔丁基-2,6-二甲基苯基)-联苯胺(0.053g)、2,2’-联吡啶(0.548g)溶解于脱水的四氢呋喃140mL中后,用氩气鼓泡而将体系内进行氮气置换。升温至60℃后,在氮气气氛下,向该溶液中加入双(1,5-环辛二烯)镍(0){Ni(COD)2}(0.965g),搅拌,反应3小时。将该反应液冷却至室温,向25%氨水5mL/甲醇140mL/离子交换水140mL混合溶液中滴加,搅拌1小时后,过滤析出的沉淀,减压干燥,溶解于甲苯40ml中。溶解后,添加硅藻土1.6g,搅拌30分钟,过滤不溶物。将所得的滤液穿过氧化铝柱而进行精制。然后添加5.2%盐酸水80mL,搅拌3小时后,除去水层。然后添加4%氨水80mL,搅拌2小时后除去水层。继而向有机层中添加离子交换水约80mL,搅拌1小时后,除去水层。其后,将有机层注入到甲醇160ml中,搅拌1小时,过滤析出的沉淀,减压干燥。所得的聚合物(以后称作高分子化合物4)的产量为0.31g。另外,聚苯乙烯换算的数均分子量及重均分子量分别Mn=2.5×104,Mw=1.2×105。 Compound B (0.588g), N, N'-bis(4-bromophenyl)-N,N'-bis(4-tert-butyl-2,6-dimethylphenyl)-benzidine (0.053 g) After dissolving 2,2'-bipyridine (0.548 g) in 140 mL of dehydrated tetrahydrofuran, the inside of the system was replaced with nitrogen by bubbling argon. After heating up to 60°C, bis(1,5-cyclooctadiene)nickel(0){Ni(COD) 2 }(0.965g) was added to the solution under a nitrogen atmosphere, stirred, and reacted for 3 hours. The reaction solution was cooled to room temperature, added dropwise to a mixed solution of 25% ammonia 5mL/methanol 140mL/ion-exchanged water 140mL, stirred for 1 hour, and the precipitate was filtered, dried under reduced pressure, and dissolved in toluene 40ml. After dissolution, 1.6 g of diatomaceous earth was added, stirred for 30 minutes, and insoluble matter was filtered. The resulting filtrate was purified by passing through an alumina column. Then, 80 mL of 5.2% aqueous hydrochloric acid was added, and after stirring for 3 hours, the aqueous layer was removed. Then, 80 mL of 4% ammonia water was added, and the aqueous layer was removed after stirring for 2 hours. Next, about 80 mL of ion-exchanged water was added to the organic layer, and after stirring for 1 hour, the aqueous layer was removed. Thereafter, the organic layer was poured into 160 ml of methanol, stirred for 1 hour, and the deposited precipitate was filtered and dried under reduced pressure. The yield of the obtained polymer (hereinafter referred to as polymer compound 4) was 0.31 g. In addition, the polystyrene-equivalent number average molecular weight and weight average molecular weight were Mn=2.5×10 4 and Mw=1.2×10 5 , respectively.
实施例6<EL元件的制作与性能> Embodiment 6<The production and performance of EL element>
(溶液的配制) (preparation of solution)
将上述说明中得到的高分子化合物2溶解于甲苯中,制作了聚合物浓度为1.8重量%的甲苯溶液。 The polymer compound 2 obtained in the above description was dissolved in toluene to prepare a toluene solution having a polymer concentration of 1.8% by weight. the
(EL元件的制作) (Production of EL elements)
在利用溅射法以150nm的厚度附着了ITO膜的玻璃基板上,使用将聚(3,4)亚乙基二氧噻吩/聚苯乙烯磺酸(Bayer制,BaytronP AI4083)的悬浊液用0.2μm薄膜过滤器过滤了的液体,利用旋转涂覆机以70nm的厚度形成薄膜,在热板上以200℃干燥10分钟。然后,使用上述操作中得到的甲苯溶液,利用旋转涂覆机以3400rpm的转速成膜。成膜后的膜厚约为95nm。继而,将其在减压下80℃干燥1小时后,蒸镀约4nm的氟化锂,作为阴极蒸镀约5nm的钙,然后蒸镀约80nm的铝,制作了EL元件。而且,在真空度到达1×10-4Pa以下后,开始金属的蒸镀。 A suspension of poly(3,4)ethylenedioxythiophene/polystyrenesulfonic acid (manufactured by Bayer, BaytronP AI4083) was used on a glass substrate on which an ITO film was attached to a thickness of 150 nm by sputtering. The liquid filtered with a 0.2 μm membrane filter was formed into a thin film with a thickness of 70 nm by a spin coater, and dried on a hot plate at 200° C. for 10 minutes. Then, using the toluene solution obtained in the above operation, a film was formed with a spin coater at a rotation speed of 3400 rpm. The film thickness after film formation was about 95 nm. Next, after drying this under reduced pressure at 80° C. for 1 hour, about 4 nm of lithium fluoride was deposited, about 5 nm of calcium was deposited as a cathode, and about 80 nm of aluminum was deposited to produce an EL element. Then, after the degree of vacuum reached 1×10 -4 Pa or less, vapor deposition of the metal was started.
(EL元件的性能) (Performance of EL element)
通过对所得的元件施加电压,从该元件中得到了在455nm具有峰的EL发光。当将EL发光颜色用C.I.E.颜色坐标值表示时,则x=0.150,y=0.128,显示出非常良好的蓝色。EL发光的强度基本上与电流密度成比例。另外,该元件是从5.2V起可以看到发光开始。发光效率在所测定的施加电压的范围(0V~12V)中单调地增加,而12V处的值显示出1.02cd/m2 这样的比较高的效率。 By applying a voltage to the obtained device, EL emission having a peak at 455 nm was obtained from the device. When the EL emission color is represented by the CIE color coordinate value, x=0.150, y=0.128, showing a very good blue color. The intensity of EL light emission is basically proportional to the current density. In addition, this element can be seen to start emitting light from 5.2V. The luminous efficiency increased monotonously in the range of the measured applied voltage (0 V to 12 V), and the value at 12 V showed a relatively high efficiency of 1.02 cd/m 2 .
实施例7<EL元件的制作与性能> Embodiment 7<The production and performance of EL element>
(溶液的配制) (preparation of solution)
将上述说明中得到的高分子化合物4以90重量%的比率、将高分子化合物3以10重量%的比率溶解于甲苯中,制作了聚合物浓度为1.8重量%的甲苯溶液。 Polymer Compound 4 obtained in the above description was dissolved in toluene at a ratio of 90% by weight and Polymer Compound 3 at a ratio of 10% by weight to prepare a toluene solution having a polymer concentration of 1.8% by weight. the
(EL元件的制作) (Production of EL elements)
除了使用上述操作中得到的甲苯溶液以外,与实施例6中记载的方法完全相同地制成了EL元件。而且,聚合物溶液的在旋转涂覆机中的转速为3300rpm,聚合物膜的成膜后的膜厚为95nm。 An EL element was produced in exactly the same manner as described in Example 6, except that the toluene solution obtained in the above operation was used. Furthermore, the rotational speed of the polymer solution in the spin coater was 3300 rpm, and the film thickness of the polymer film after film formation was 95 nm. the
(EL元件的性能) (Performance of EL element)
通过对所得的元件施加电压,从该元件中得到了在425nm具有峰的EL发光。当将EL发光颜色用C.I.E.颜色坐标值表示时,则x=0.155,y=0.072,显示出非常良好的蓝色。EL发光的强度基本上与电流密度成比例。另外,该元件是从5.5V起可以看到发光开始。发光效率在所测定的施加电压的范围(0V~12V)中单调地增加,而12V处的值显示出0.22cd/m2 这样的比较高的效率。 By applying a voltage to the obtained device, EL emission having a peak at 425 nm was obtained from the device. When the EL emission color is represented by the CIE color coordinate value, x=0.155, y=0.072, showing a very good blue color. The intensity of EL light emission is basically proportional to the current density. In addition, this element can be seen to start emitting light from 5.5V. The luminous efficiency increased monotonously in the range of the measured applied voltage (0 V to 12 V), and the value at 12 V showed a relatively high efficiency of 0.22 cd/m 2 .
实施例8<EL元件的制作与性能> Embodiment 8<The production and performance of EL element>
(溶液的配制) (preparation of solution)
将上述说明中得到的高分子化合物4溶解于甲苯中,制作了聚合物浓度为1.8重量%的甲苯溶液。 The polymer compound 4 obtained in the above description was dissolved in toluene to prepare a toluene solution having a polymer concentration of 1.8% by weight. the
(EL元件的制作) (Production of EL elements)
除了使用上述操作中得到的甲苯溶液以外,与实施例6中记载的方法完全相同地制成了EL元件。而且,聚合物溶液的在旋转涂覆机中的转速 为2500rpm,聚合物膜的成膜后的膜厚为90nm。 An EL element was produced in exactly the same manner as described in Example 6, except that the toluene solution obtained in the above operation was used. Furthermore, the rotational speed of the polymer solution in the spin coater was 2500 rpm, and the film thickness of the polymer film after film formation was 90 nm. the
(EL元件的性能) (Performance of EL element)
通过对所得的元件施加电压,从该元件中得到了在425nm具有峰的EL发光。当将EL发光颜色用C.I.E.颜色坐标值表示时,则x=0.155,y=0.074,显示出非常良好的蓝色。EL发光的强度基本上与电流密度成比例。另外,该元件是从5.8V起可以看到发光开始。发光效率在所测定的施加电压的范围(0V~12V)中单调地增加,而12V处的值显示出0.57cd/m2 这样的比较高的效率。 By applying a voltage to the obtained device, EL emission having a peak at 425 nm was obtained from the device. When the EL emission color is represented by the CIE color coordinate value, x=0.155, y=0.074, showing a very good blue color. The intensity of EL light emission is basically proportional to the current density. In addition, this device can be seen to start emitting light from 5.8V. The luminous efficiency increased monotonously in the measured applied voltage range (0V to 12V), and the value at 12V showed a relatively high efficiency of 0.57cd/m 2 .
实施例9<化合物D的合成> Embodiment 9<The synthesis of compound D>
(化合物C的合成) (Synthesis of Compound C)
(化合物C) (compound C)
向反应容器中加入离子交换水31ml,在搅拌的同时-点点地加入氢氧化钠29g(727mmol),完全地溶解。将体系内用氩气置换,加入甲苯30ml和1,2,3,10b-tetrahydrofluoranthene 5.0g(24mmol),搅拌而溶解。接下来加入溴化四丁基铵2.3g(7.3mmol)和2-乙基己基溴9.4g(48mmol),在40℃反应3小时。用甲苯和水分液,将有机层萃取后,用硫酸钠干燥。蒸馏除去溶剂后,通过用以己烷作为展开溶剂的硅胶色谱精制,得到了黄色的油6.88g。 31 ml of ion-exchanged water was added to the reaction container, and 29 g (727 mmol) of sodium hydroxide was added little by little while stirring to completely dissolve it. The system was replaced with argon, and 30 ml of toluene and 5.0 g (24 mmol) of 1,2,3,10b-tetrahydrofluoranthene were added, stirred and dissolved. Next, 2.3 g (7.3 mmol) of tetrabutylammonium bromide and 9.4 g (48 mmol) of 2-ethylhexyl bromide were added, and it was made to react at 40 degreeC for 3 hours. The organic layer was extracted with toluene and water, and dried over sodium sulfate. After distilling off the solvent, it was purified by silica gel chromatography using hexane as a developing solvent to obtain 6.88 g of a yellow oil. the
MS(APPI(+)):318([M+H]+) MS (APPI (+)): 318 ([M+H] + )
1H-NMR(300MHz/CDCl3)δ7.69(1H,d)、7.50(1H,d)、7.40(1H,d)、7.33~7.22(3H,m)、7.02(1H,d)、3.07~2.96(1H,m)、2.79~2.71(1H,m)、2.36~2.22(2H,m)、2.10~1.99(1H,m)、1.93~1.76(2H,m)、1.24~1.15(1H,m)、0.88~0.37(9H,m) 1 H-NMR (300MHz/CDCl 3 ) δ7.69(1H,d), 7.50(1H,d), 7.40(1H,d), 7.33~7.22(3H,m), 7.02(1H,d), 3.07 ~2.96(1H, m), 2.79~2.71(1H, m), 2.36~2.22(2H, m), 2.10~1.99(1H, m), 1.93~1.76(2H, m), 1.24~1.15(1H, m), 0.88~0.37(9H, m)
(化合物D的合成) (Synthesis of compound D)
(化合物D) (compound D)
向氩气置换的反应容器中加入化合物C6.8g(21mmol)、氯化锌6.7g(49mmol)及乙酸∶二氯甲烷=1∶1的混合溶剂134ml,在室温下搅拌。接下来将三溴化苄基三甲基铵18g(47mmol)溶解于二氯甲烷150ml中滴加。滴加结束后,在室温下反应2小时,接下来在40℃及50℃各反应30分钟后,添加氯仿和5%亚硫酸氢钠水溶液而停止反应。用氯仿和水分液,萃取有机层,水洗2次后,用碳酸钾水溶液中和。用硫酸钠干燥后,蒸馏除去溶剂,通过用以己烷作为展开溶剂的硅胶色谱精制3次,作为黄色的油(室温下放置时慢慢地白色结晶化),得到了化合物D1.73g。 6.8 g (21 mmol) of compound C, 6.7 g (49 mmol) of zinc chloride, and 134 ml of a mixed solvent of acetic acid:dichloromethane=1:1 were added to a reaction vessel replaced with argon, and stirred at room temperature. Next, 18 g (47 mmol) of benzyltrimethylammonium tribromide was dissolved in 150 ml of methylene chloride and added dropwise. After completion of the dropwise addition, the reaction was carried out at room temperature for 2 hours, followed by the reaction at 40° C. and 50° C. for 30 minutes each, and the reaction was stopped by adding chloroform and 5% aqueous sodium bisulfite solution. The organic layer was extracted with chloroform and water, washed twice with water, and then neutralized with an aqueous potassium carbonate solution. After drying over sodium sulfate, the solvent was distilled off and purified three times by silica gel chromatography using hexane as a developing solvent to obtain 1.73 g of Compound D as a yellow oil (slowly crystallized into white when left standing at room temperature). the
MS(APPI(+))476(M+) MS(APPI(+))476(M + )
1H-NMR(300MHz/CDCl3)δ7.52~7.42(4H,m)、7.33(1H,d)、3.00~2.80(2H,m)、2.40~2.20(2H,m)、2.02~1.89(2H,m)、1.75~1.70(1H,m)、1.31~1.15(2H,m)、0.96~0.39(8H,m) 1 H-NMR (300MHz/CDCl 3 ) δ7.52~7.42(4H, m), 7.33(1H, d), 3.00~2.80(2H, m), 2.40~2.20(2H, m), 2.02~1.89( 2H, m), 1.75~1.70(1H, m), 1.31~1.15(2H, m), 0.96~0.39(8H, m)
实施例10 Example 10
<高分子化合物5的合成> <Synthesis of Polymer Compound 5>
在氮气气氛下,将化合物D(0.476g)、2,2’-联吡啶(0.422g)溶解于脱水的四氢呋喃72mL中后,搅拌而使之溶解。向该溶液中加入双(1,5-二氯辛二烯)镍(0){Ni(COD)2}(0.743g),搅拌,在60℃下反应3小时。将该反应液冷却至室温,向25%氨水4mL/甲醇72mL/离子交换水72mL混合溶液中滴加,搅拌1小时后,过滤析出的沉淀,减压干燥,溶解于甲苯20ml中。溶解后,添加硅藻土1.6g,搅拌30分钟,过滤不溶物。将所得的滤液穿过氧化铝柱而进行精制。然后添加5.2%盐酸水40mL,搅拌3小时后,除去水层。然后添加4%氨水40mL,搅拌2小时后除去水层。继而向有机层中添加离子交换水约40mL,搅拌1小时后,除去水层。 其后,将有机层中注入到甲醇80ml中,搅拌1小时,过滤析出的沉淀,减压干燥。所得的聚合物(以后称作高分子化合物5)的产量为0.17g。另外,聚苯乙烯换算的数均分子量及重均分子量分别Mn=1.1×105,Mw=3.2×105。 Under a nitrogen atmosphere, Compound D (0.476 g) and 2,2'-bipyridine (0.422 g) were dissolved in 72 mL of dehydrated tetrahydrofuran, followed by stirring to dissolve. Bis(1,5-dichlorooctadiene) nickel (0) {Ni(COD) 2 } (0.743 g) was added to this solution, stirred, and reacted at 60 degreeC for 3 hours. The reaction solution was cooled to room temperature, added dropwise to a mixed solution of 25% ammonia 4mL/methanol 72mL/ion-exchanged water 72mL, stirred for 1 hour, and the precipitate was filtered, dried under reduced pressure, and dissolved in toluene 20ml. After dissolution, 1.6 g of diatomaceous earth was added, stirred for 30 minutes, and insoluble matter was filtered. The resulting filtrate was purified by passing through an alumina column. Then, 40 mL of 5.2% aqueous hydrochloric acid was added, and after stirring for 3 hours, the aqueous layer was removed. Then, 40 mL of 4% ammonia water was added, and the aqueous layer was removed after stirring for 2 hours. Next, about 40 mL of ion-exchanged water was added to the organic layer, and after stirring for 1 hour, the aqueous layer was removed. Thereafter, the organic layer was poured into 80 ml of methanol, stirred for 1 hour, and the deposited precipitate was filtered and dried under reduced pressure. The yield of the obtained polymer (hereinafter referred to as polymer compound 5) was 0.17 g. In addition, the polystyrene-equivalent number average molecular weight and weight average molecular weight were Mn=1.1×10 5 and Mw=3.2×10 5 , respectively.
比较例1 Comparative example 1
<高分子化合物6的合成> <Synthesis of polymer compound 6>
将2,7-二溴-9,9-二辛基芴0.22g(0.40mmol)、N,N’-双(4-溴苯基)-N,N’-双(4-叔丁基-2,6-二甲基苯基)-1,4-苯二胺0.20g(0.27mmol)和2,2’-联吡啶0.24g(1.5mmol)加入反应容器后,将反应体系内用氮气置换。向其中加入预先用氩气鼓泡而脱气的四氢呋喃(脱水溶剂)。然后,向该混合溶液中,添加双(1,5-环辛二烯)镍(0)0.42g(1.5mmol),在60℃反应3小时。而且,反应是在氮气气氛中进行的。反应后,将该溶液冷却后,注入到25%氨水10mL/甲醇120mL/离子交换水50mL混合溶液中,搅拌约1小时。然后过滤回收所生成的沉淀。将该沉淀用乙醇清洗后,减压干燥2小时。然后,将该沉淀溶解于甲苯50ml中,添加1N盐酸50mL,搅拌1小时,除去水层,向有机层中加入4%氨水50mL,搅拌1小时后除去水层。将有机层向甲醇120mL中滴加,搅拌1小时,过滤析出的沉淀,减压干燥2小时,溶解于40mL甲苯中。其后,穿过氧化铝柱(氧化铝量20g)而进行精制,将所回收的甲苯溶液向甲醇120mL中滴加,搅拌1小时,过滤析出的沉淀,减压干燥2小时。所得的高分子化合物6的产量为0.094g。 0.22g (0.40mmol) of 2,7-dibromo-9,9-dioctylfluorene, N,N'-bis(4-bromophenyl)-N,N'-bis(4-tert-butyl- After adding 0.20g (0.27mmol) of 2,6-dimethylphenyl)-1,4-phenylenediamine and 0.24g (1.5mmol) of 2,2'-bipyridine into the reaction vessel, replace the reaction system with nitrogen . Tetrahydrofuran (dehydration solvent) previously degassed by bubbling argon gas was added thereto. Then, 0.42 g (1.5 mmol) of bis(1,5-cyclooctadiene) nickel (0) was added to this mixed solution, and it was made to react at 60 degreeC for 3 hours. Also, the reaction was carried out in a nitrogen atmosphere. After the reaction, the solution was cooled, poured into a mixed solution of 25% ammonia water 10 mL/methanol 120 mL/ion-exchanged water 50 mL, and stirred for about 1 hour. The resulting precipitate was then recovered by filtration. After the precipitate was washed with ethanol, it was dried under reduced pressure for 2 hours. Then, this precipitate was dissolved in 50 ml of toluene, 50 mL of 1N hydrochloric acid was added, stirred for 1 hour, the water layer was removed, 4% ammonia water 50 mL was added to the organic layer, and the water layer was removed after stirring for 1 hour. The organic layer was added dropwise to 120 mL of methanol, stirred for 1 hour, and the deposited precipitate was filtered, dried under reduced pressure for 2 hours, and dissolved in 40 mL of toluene. Thereafter, it was purified by passing through an alumina column (amount of alumina: 20 g), and the recovered toluene solution was added dropwise to 120 mL of methanol, stirred for 1 hour, and the deposited precipitate was filtered and dried under reduced pressure for 2 hours. The yield of the obtained polymer compound 6 was 0.094 g. the
高分子化合物6的聚苯乙烯换算的数均分子量为2.0×104,聚苯乙烯换算重均分子量为1.1×105。 Polystyrene-equivalent number average molecular weight of polymer compound 6 was 2.0×10 4 , and polystyrene-equivalent weight average molecular weight was 1.1×10 5 .
比较例2<EL元件的制作与性能> Comparative example 2 <Production and performance of EL element>
(溶液的配制) (preparation of solution)
将上述说明中得到的高分子化合物6溶解于氯仿中,制作了聚合物浓度为1.8重量%的氯仿溶液。 The polymer compound 6 obtained in the above description was dissolved in chloroform to prepare a chloroform solution having a polymer concentration of 1.8% by weight. the
(EL元件的制作) (Production of EL elements)
在利用溅射法以150nm的厚度附着了ITO膜的玻璃基板上,使用将聚(亚乙基二氧噻吩)/聚苯乙烯磺酸的溶液(Bayer制,BaytronP),利用 旋转涂覆机以50nm的厚度成膜,在热板上以200℃干燥10分钟。然后,使用上述所配制的氯仿溶液,利用旋转涂覆机以2500rpm的转速成膜。膜厚约为100nm。继而,将其在减压下80℃干燥1小时后,作为阴极缓冲层,蒸镀约4nm的LiF,作为阴极蒸镀约5nm的钙,然后蒸镀约80nm的铝,制作了EL元件。而且,在真空度到达1×10-4Pa以下后,开始金属的蒸镀。 On a glass substrate on which an ITO film was adhered to a thickness of 150 nm by sputtering, a solution of poly(ethylenedioxythiophene)/polystyrenesulfonic acid (manufactured by Bayer, BaytronP) was used to coat the glass substrate with a spin coater. A film was formed with a thickness of 50 nm, and dried on a hot plate at 200° C. for 10 minutes. Then, using the chloroform solution prepared above, a spin coater was used to form a film at a rotational speed of 2500 rpm. The film thickness is about 100 nm. Next, after drying this under reduced pressure at 80° C. for 1 hour, about 4 nm of LiF was deposited as a cathode buffer layer, about 5 nm of calcium was deposited as a cathode, and about 80 nm of aluminum was deposited to fabricate an EL element. Then, after the degree of vacuum reached 1×10 -4 Pa or less, vapor deposition of the metal was started.
(EL元件的性能) (Performance of EL element)
通过对所得的元件施加电压,得到了在448nm具有峰的EL发光。当将EL发光颜色用C.I.E.颜色坐标值表示时,则x=0.155,y=0.133。该元件在大约10V显示出最大发光效率,其值为0.14cd/A。 By applying a voltage to the obtained device, EL emission having a peak at 448 nm was obtained. When the EL emission color is represented by the C.I.E. color coordinate value, then x=0.155, y=0.133. The element exhibited the maximum luminous efficiency at about 10V, which was 0.14 cd/A. the
本发明的高分子化合物在作为电子元件的材料使用时,可以提供元件性能优良的电子元件。 When the polymer compound of the present invention is used as a material of an electronic device, it can provide an electronic device having excellent device performance. the
本发明的高分子化合物通常来说在固体状态下发出荧光或磷光,可以作为高分子发光体(高分子量的发光材料)使用。 The polymer compound of the present invention generally emits fluorescence or phosphorescence in a solid state, and can be used as a polymer luminescent body (a high-molecular-weight light-emitting material). the
另外,该高分子化合物具有优良的电荷输送性能,可以作为高分子LED用材料或电荷输送材料合适地使用。使用了该高分子发光体的高分子LED是能够以低电压、高效率驱动的高性能的高分子LED。所以,该高分子LED可以理想地用于液晶显示器的背光灯、作为照明用的曲面状或平面状的光源、分段型的显示元件、点矩阵的平板显示器等装置中。 In addition, the polymer compound has excellent charge transport performance, and can be suitably used as a polymer LED material or a charge transport material. A polymer LED using this polymer light-emitting body is a high-performance polymer LED that can be driven at low voltage and with high efficiency. Therefore, the polymer LED can be ideally used in devices such as backlights of liquid crystal displays, curved or planar light sources for illumination, segmented display elements, and dot-matrix flat panel displays. the
另外,本发明的高分子化合物也可以作为激光器用色素、有机太阳能电池用材料、有机晶体管用的有机半导体、导电性薄膜、有机半导体薄膜等的传导性薄膜用材料使用。 In addition, the polymer compound of the present invention can also be used as a material for conductive thin films such as dyes for lasers, materials for organic solar cells, organic semiconductors for organic transistors, conductive thin films, and organic semiconductor thin films. the
另外,还可以作为发出荧光或磷光的发光性薄膜材料使用。 In addition, it can also be used as a light-emitting thin film material that emits fluorescence or phosphorescence. the
Claims (25)
Applications Claiming Priority (3)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| JP2005131383 | 2005-04-28 | ||
| JP131383/2005 | 2005-04-28 | ||
| PCT/JP2006/309353 WO2006118345A1 (en) | 2005-04-28 | 2006-04-28 | High-molecular compounds and high-molecular luminescent devices made by using the same |
Publications (2)
| Publication Number | Publication Date |
|---|---|
| CN101184789A CN101184789A (en) | 2008-05-21 |
| CN101184789B true CN101184789B (en) | 2012-05-30 |
Family
ID=37308121
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| CN2006800189430A Expired - Fee Related CN101184789B (en) | 2005-04-28 | 2006-04-28 | High-molecular compound and high-molecular luminescent device using the same |
Country Status (5)
| Country | Link |
|---|---|
| KR (1) | KR20080013926A (en) |
| CN (1) | CN101184789B (en) |
| DE (1) | DE112006001016T5 (en) |
| GB (1) | GB2440482A (en) |
| WO (1) | WO2006118345A1 (en) |
Families Citing this family (63)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| JP2006328387A (en) * | 2005-04-28 | 2006-12-07 | Sumitomo Chemical Co Ltd | Polymer compound and polymer light emitting device using the same |
| CN101316879B (en) * | 2005-11-30 | 2011-10-05 | 住友化学株式会社 | Polymer material and polymer light-emitting device using the polymer material |
| JP5095189B2 (en) * | 2005-11-30 | 2012-12-12 | 住友化学株式会社 | Polymer material and polymer light emitting device using the same |
| CN101490862B (en) | 2006-07-25 | 2013-03-13 | 默克专利有限公司 | Polymer blends and their use in organic light emitting devices |
| EP2462203B1 (en) | 2009-08-04 | 2016-03-02 | Merck Patent GmbH | Electronic devices comprising multi cyclic hydrocarbons |
| EP2517278B1 (en) | 2009-12-22 | 2019-07-17 | Merck Patent GmbH | Electroluminescent formulations |
| JP5897472B2 (en) | 2009-12-22 | 2016-03-30 | メルク パテント ゲーエムベーハー | Electroluminescent functional surfactant |
| WO2011076323A1 (en) | 2009-12-22 | 2011-06-30 | Merck Patent Gmbh | Formulations comprising phase-separated functional materials |
| DE102010009193B4 (en) | 2010-02-24 | 2022-05-19 | MERCK Patent Gesellschaft mit beschränkter Haftung | Composition containing fluorine-fluorine associates, processes for their production, their use and organic electronic devices containing them |
| WO2011110277A1 (en) | 2010-03-11 | 2011-09-15 | Merck Patent Gmbh | Fibers in therapy and cosmetics |
| US9373807B2 (en) | 2010-03-11 | 2016-06-21 | Merck Patent Gmbh | Radiative fibers |
| EP2576723B1 (en) | 2010-05-27 | 2017-09-20 | Merck Patent GmbH | Compositions comprising quantum dots |
| WO2011147521A1 (en) | 2010-05-27 | 2011-12-01 | Merck Patent Gmbh | Down conversion |
| CN103563113A (en) | 2011-06-01 | 2014-02-05 | 默克专利股份有限公司 | Hybrid ambipolar TFTs |
| WO2013013754A1 (en) | 2011-07-25 | 2013-01-31 | Merck Patent Gmbh | Copolymers with functionalized side chains |
| US9246103B2 (en) | 2011-07-25 | 2016-01-26 | Merck Patent Gmbh | Polymers and oligomers with functionalized side groups |
| DE102012016192A1 (en) | 2011-08-19 | 2013-02-21 | Merck Patent Gmbh | New compounds capable of forming hydrogen bonds are useful in electronic device, e.g. organic electroluminescent device, organic light-emitting transistor and organic light-emitting electrochemical cell |
| DE102011121022A1 (en) | 2011-12-13 | 2013-06-13 | Merck Patent Gmbh | Organic sensitizers for up-conversion |
| JP6567519B2 (en) | 2013-07-29 | 2019-08-28 | メルク、パテント、ゲゼルシャフト、ミット、ベシュレンクテル、ハフツングMerck Patent GmbH | Electroluminescence element |
| EP3028318A1 (en) | 2013-07-29 | 2016-06-08 | Merck Patent GmbH | Electro-optical device and the use thereof |
| EP3189551B1 (en) | 2014-09-05 | 2021-01-27 | Merck Patent GmbH | Formulations and method of producing an organic electroluminescent device |
| WO2016086885A1 (en) | 2014-12-04 | 2016-06-09 | 广州华睿光电材料有限公司 | Deuterated organic compound, mixture and composition containing said compound, and organic electronic device |
| CN107001336A (en) | 2014-12-11 | 2017-08-01 | 广州华睿光电材料有限公司 | A kind of organometallic complex, polymer comprising it, mixture, composition, organic electronic device and application |
| WO2016107663A1 (en) | 2014-12-30 | 2016-07-07 | Merck Patent Gmbh | Formulations and electronic devices |
| CN107108862B (en) | 2015-01-13 | 2019-08-02 | 广州华睿光电材料有限公司 | Conjugated polymers containing acetylene crosslinking groups, mixtures containing them, compositions, organic electronic devices and applications thereof |
| WO2016155866A1 (en) | 2015-03-30 | 2016-10-06 | Merck Patent Gmbh | Formulation of an organic functional material comprising a siloxane solvent |
| KR102654992B1 (en) | 2015-06-12 | 2024-04-04 | 메르크 파텐트 게엠베하 | Esters containing non-aromatic cycles as solvents for oled formulations |
| EP3341981B1 (en) | 2015-08-28 | 2020-08-19 | Merck Patent GmbH | Formulation of an organic functional material comprising an epoxy group containing solvent |
| CN108291103B (en) | 2015-11-12 | 2021-12-07 | 广州华睿光电材料有限公司 | Printing composition, electronic device comprising same and preparation method of functional material film |
| CN108368361A (en) | 2015-12-10 | 2018-08-03 | 默克专利有限公司 | Preparation containing the ketone comprising non-aromatic ring |
| JP7051684B2 (en) | 2015-12-15 | 2022-04-11 | メルク パテント ゲーエムベーハー | Esters containing aromatic groups as solvents for organic electronic formulations |
| KR20180095028A (en) | 2015-12-16 | 2018-08-24 | 메르크 파텐트 게엠베하 | A formulation containing a mixture of two or more different solvents |
| EP3390549B1 (en) | 2015-12-16 | 2022-06-29 | Merck Patent GmbH | Formulations containing a solid solvent |
| US10840448B2 (en) | 2016-02-17 | 2020-11-17 | Merck Patent Gmbh | Formulation of an organic functional material |
| DE102016003104A1 (en) | 2016-03-15 | 2017-09-21 | Merck Patent Gmbh | Container comprising a formulation containing at least one organic semiconductor |
| CN109153871A (en) | 2016-06-16 | 2019-01-04 | 默克专利有限公司 | The preparation of organic functional material |
| JP2019523998A (en) | 2016-06-17 | 2019-08-29 | メルク パテント ゲーエムベーハー | Formulation of organic functional materials |
| TW201815998A (en) | 2016-06-28 | 2018-05-01 | 德商麥克專利有限公司 | Organic functional material formulation |
| WO2018024719A1 (en) | 2016-08-04 | 2018-02-08 | Merck Patent Gmbh | Formulation of an organic functional material |
| JP7013459B2 (en) | 2016-10-31 | 2022-01-31 | メルク パテント ゲーエムベーハー | Formulation of organic functional materials |
| CN109863223B (en) | 2016-10-31 | 2023-06-20 | 默克专利有限公司 | Formulation of organic functional materials |
| CN109790407B (en) | 2016-11-23 | 2021-12-07 | 广州华睿光电材料有限公司 | Printing ink composition, preparation method and application thereof |
| KR102472751B1 (en) | 2016-12-06 | 2022-11-30 | 메르크 파텐트 게엠베하 | Manufacturing method of electronic device |
| CN110168047B (en) | 2016-12-13 | 2023-08-08 | 默克专利有限公司 | Preparation of organic functional material |
| TWI791481B (en) | 2017-01-30 | 2023-02-11 | 德商麥克專利有限公司 | Method for forming an organic electroluminescence (el) element |
| TWI763772B (en) | 2017-01-30 | 2022-05-11 | 德商麥克專利有限公司 | Method for forming an organic element of an electronic device |
| JP7123967B2 (en) | 2017-03-31 | 2022-08-23 | メルク パテント ゲーエムベーハー | Printing method for organic light emitting diodes (OLEDs) |
| CN110494514A (en) | 2017-04-10 | 2019-11-22 | 默克专利有限公司 | Formulation of organic functional materials |
| JP7330898B2 (en) | 2017-05-03 | 2023-08-22 | メルク パテント ゲーエムベーハー | Formulation of organic functional material |
| WO2019016184A1 (en) | 2017-07-18 | 2019-01-24 | Merck Patent Gmbh | Formulation of an organic functional material |
| CN111418081B (en) | 2017-12-15 | 2024-09-13 | 默克专利有限公司 | Preparation of organic functional materials |
| JP7247231B2 (en) | 2018-02-26 | 2023-03-28 | メルク パテント ゲーエムベーハー | Formulation of organic functional material |
| EP3807367B1 (en) | 2018-06-15 | 2023-07-19 | Merck Patent GmbH | Formulation of an organic functional material |
| WO2020094538A1 (en) | 2018-11-06 | 2020-05-14 | Merck Patent Gmbh | Method for forming an organic element of an electronic device |
| WO2021213918A1 (en) | 2020-04-21 | 2021-10-28 | Merck Patent Gmbh | Formulation of an organic functional material |
| EP4139971A1 (en) | 2020-04-21 | 2023-03-01 | Merck Patent GmbH | Emulsions comprising organic functional materials |
| WO2022122607A1 (en) | 2020-12-08 | 2022-06-16 | Merck Patent Gmbh | An ink system and a method for inkjet printing |
| WO2022223675A1 (en) | 2021-04-23 | 2022-10-27 | Merck Patent Gmbh | Formulation of an organic functional material |
| CN117730638A (en) | 2021-08-02 | 2024-03-19 | 默克专利有限公司 | Printing method by combining inks |
| TW202349760A (en) | 2021-10-05 | 2023-12-16 | 德商麥克專利有限公司 | Method for forming an organic element of an electronic device |
| TW202411366A (en) | 2022-06-07 | 2024-03-16 | 德商麥克專利有限公司 | Method of printing a functional layer of an electronic device by combining inks |
| TW202440819A (en) | 2022-12-16 | 2024-10-16 | 德商麥克專利有限公司 | Formulation of an organic functional material |
| WO2025032039A1 (en) | 2023-08-07 | 2025-02-13 | Merck Patent Gmbh | Process for the preparation of an electronic device |
Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5858562A (en) * | 1994-10-13 | 1999-01-12 | Nec Corporation | Organic thin film electroluminescent device |
| US6630566B1 (en) * | 1997-12-19 | 2003-10-07 | Avecia Limited | Polymeric material comprising N, P, S, As, or Se and composition for charge transport material |
Family Cites Families (1)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US20060094859A1 (en) * | 2004-11-03 | 2006-05-04 | Marrocco Matthew L Iii | Class of bridged biphenylene polymers |
-
2006
- 2006-04-28 CN CN2006800189430A patent/CN101184789B/en not_active Expired - Fee Related
- 2006-04-28 WO PCT/JP2006/309353 patent/WO2006118345A1/en not_active Ceased
- 2006-04-28 KR KR1020077027267A patent/KR20080013926A/en not_active Ceased
- 2006-04-28 DE DE112006001016T patent/DE112006001016T5/en not_active Withdrawn
-
2007
- 2007-11-15 GB GB0722483A patent/GB2440482A/en not_active Withdrawn
Patent Citations (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5858562A (en) * | 1994-10-13 | 1999-01-12 | Nec Corporation | Organic thin film electroluminescent device |
| US6630566B1 (en) * | 1997-12-19 | 2003-10-07 | Avecia Limited | Polymeric material comprising N, P, S, As, or Se and composition for charge transport material |
Non-Patent Citations (2)
| Title |
|---|
| Holbro Th. et al.Bromofluoranthenes.II. Synthesis of 4,11-dibromofluoranthene.Helvetica Chimica Acta33.1950,332178-2185. * |
| HolbroTh.etal.Bromofluoranthenes.II.Synthesisof4 11-dibromofluoranthene.Helvetica Chimica Acta33.1950 |
Also Published As
| Publication number | Publication date |
|---|---|
| CN101184789A (en) | 2008-05-21 |
| KR20080013926A (en) | 2008-02-13 |
| GB0722483D0 (en) | 2007-12-27 |
| DE112006001016T5 (en) | 2008-04-03 |
| GB2440482A (en) | 2008-01-30 |
| WO2006118345A1 (en) | 2006-11-09 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| CN101184789B (en) | High-molecular compound and high-molecular luminescent device using the same | |
| JP5076433B2 (en) | Copolymer and polymer light emitting device using the same | |
| CN1898292B (en) | Polymer compound and polymer light-emitting device using the polymer compound | |
| CN101516963B (en) | Polymer compound and polymer light-emitting device | |
| JP4736471B2 (en) | Polymer compound and polymer light emitting device using the same | |
| JP5256568B2 (en) | Polymer compound and polymer light emitting device using the same | |
| CN101203539B (en) | Polymer material and polymeric light-emitting element | |
| JP5162888B2 (en) | Polymer compound and polymer light emitting device using the same | |
| JP5274754B2 (en) | Polymer material and polymer light emitting device | |
| JP5162868B2 (en) | Polymer light-emitting device, organic transistor and composition useful for them | |
| CN101356211A (en) | Conjugated polymer compound and polymer light emitting device using the conjugated polymer compound | |
| CN101198633A (en) | Polymer materials and polymer light-emitting devices | |
| CN101316879B (en) | Polymer material and polymer light-emitting device using the polymer material | |
| JP4904805B2 (en) | Polymer compound and polymer light emitting device using the same | |
| JP5617150B2 (en) | Conjugated polymer compound and polymer light emitting device using the same | |
| JP4956918B2 (en) | Polymer compound and polymer light emitting device using the same | |
| JP4896411B2 (en) | Polymer compound and polymer light emitting device using the same | |
| JP4724440B2 (en) | Polymer compound and polymer light emitting device using the same | |
| JP5095189B2 (en) | Polymer material and polymer light emitting device using the same | |
| US8153276B2 (en) | Polymer compound and polymer light emitting device using the same | |
| JP2007016226A (en) | Aromatic polymer | |
| JP2006328387A (en) | Polymer compound and polymer light emitting device using the same | |
| JP2011038103A (en) | Polymer compound and polymer light-emitting element using the same |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| C06 | Publication | ||
| PB01 | Publication | ||
| C10 | Entry into substantive examination | ||
| SE01 | Entry into force of request for substantive examination | ||
| C14 | Grant of patent or utility model | ||
| GR01 | Patent grant | ||
| C17 | Cessation of patent right | ||
| CF01 | Termination of patent right due to non-payment of annual fee |
Granted publication date: 20120530 Termination date: 20140428 |