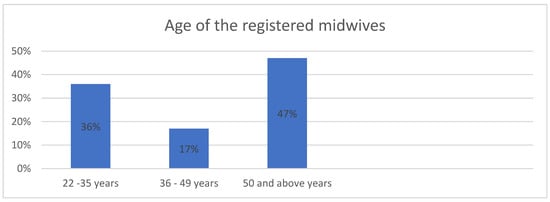
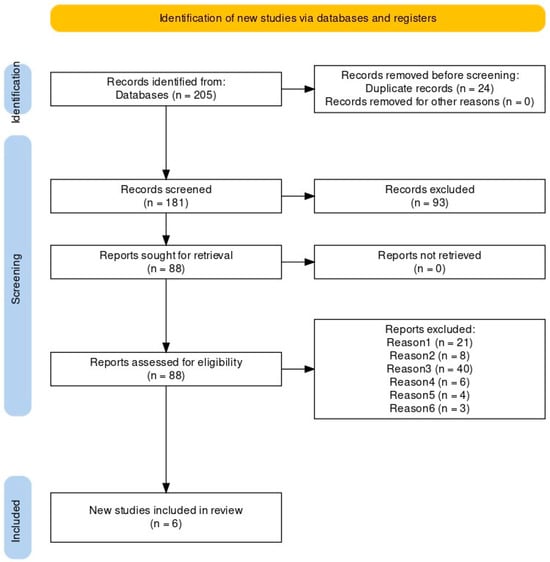
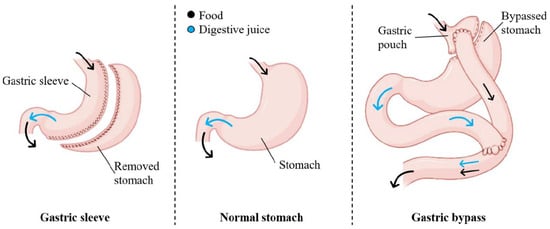
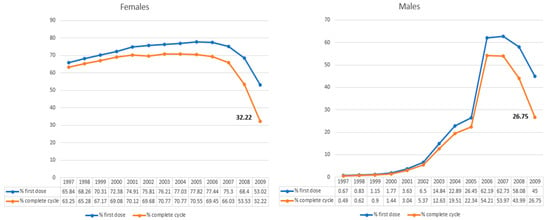
<p>Flowchart of the study population, including HPV mRNA testing, Re-evaluation of the index of cervical cytology, and detection of CIN2+ cases. The HPV mRNA test is a screening tool designed to detect messenger RNA (mRNA) from high-risk types of the human papillomavirus (HPV). For this study, we utilized the PreTect SEE, a 3-type HPV mRNA test specifically targeting the E6/E7 mRNA from HPV types 16, 18, and 45, which are among the most oncogenic and commonly associated with cervical cancer. ASC-US+: atypical squamous cells of undetermined significance or higher (including all categories of abnormalities above ASC-US). CIN2+: cervical intraepithelial neoplasia grade 2 or higher (including CIN2, CIN3, and cancers).</p> Full article ">Figure 2
<p>Distribution of HPV-positive women and detection of CIN2+ cases by age group. CIN2+ represents histological findings of cervical intraepithelial neoplasia grade 2 (CIN2) and more severe lesions, such as cervical intraepithelial neoplasia grade 3 (CIN3), adenocarcinoma in situ (ACIS), and invasive cervical cancer.</p> Full article ">Figure 3
<p>Risk of being HPV-positive and developing CIN2+ by age group. HPV+: indicates a positive result from a 3-type HPV mRNA test (PreTect SEE) that detected E6/E7 mRNA from high-risk human papillomavirus types 16, 18, and 45, suggesting an increased risk of cervical cancer development. CIN2+ represents histological findings of cervical intraepithelial neoplasia grade 2 (CIN2) and more severe lesions, including cervical intraepithelial neoplasia grade 3 (CIN3), adenocarcinoma in situ (ACIS), and invasive cervical cancer, indicating progressively higher risks of cancer progression.</p> Full article ">