Journal Description
Journal of Nanotheranostics
Journal of Nanotheranostics
is an international, peer-reviewed, open access journal on nanotheranostics published quarterly online by MDPI.
- Open Access—free for readers, with article processing charges (APC) paid by authors or their institutions.
- Rapid Publication: manuscripts are peer-reviewed and a first decision is provided to authors approximately 14.6 days after submission; acceptance to publication is undertaken in 2 days (median values for papers published in this journal in the first half of 2024).
- Recognition of Reviewers: APC discount vouchers, optional signed peer-review and reviewer names published annually in the journal.
Latest Articles
Anti-Poly(ethylene glycol) (PEG) Antibodies: From Where Are We Coming and Where Are We Going
J. Nanotheranostics 2024, 5(3), 99-103; https://doi.org/10.3390/jnt5030007 - 29 Jul 2024
Abstract
PEGylation technology confers stability and modulates the biological performance of a broad range of preclinical and clinical nanopharmaceuticals. However, the emerging PEG immunogenicity in the general population is thought to impact the efficacy and safety of PEGylated medicines. Despite this, the clinical significance
[...] Read more.
PEGylation technology confers stability and modulates the biological performance of a broad range of preclinical and clinical nanopharmaceuticals. However, the emerging PEG immunogenicity in the general population is thought to impact the efficacy and safety of PEGylated medicines. Despite this, the clinical significance of PEG immunogenicity is still not clear and remains debatable. By considering the strategic importance of the PEGylation technology in nanopharmaceutical engineering, we raise a number of critical questions and briefly discuss gaps in the knowledge of PEG immunogenicity and its clinical significance.
Full article
Open AccessReview
Paradoxical Roles of Carbon Nanotubes in Cancer Therapy and Carcinogenesis
by
Bohan Xu, Shunjie Wu, Yiyang Wang, Yuhe Ji, Shufeng Liang, Chunyan Wang and Xin Tian
J. Nanotheranostics 2024, 5(3), 84-98; https://doi.org/10.3390/jnt5030006 - 8 Jul 2024
Abstract
►▼
Show Figures
Carbon nanotubes (CNTs), members of the nanomaterial family, are increasingly being used in consumer products and extensively studied for various biomedical applications. Due to their benign elemental composition, large surface area, and chemical and biological activities, CNTs demonstrate great potential in cancer therapy,
[...] Read more.
Carbon nanotubes (CNTs), members of the nanomaterial family, are increasingly being used in consumer products and extensively studied for various biomedical applications. Due to their benign elemental composition, large surface area, and chemical and biological activities, CNTs demonstrate great potential in cancer therapy, including drug delivery, imaging analysis, photothermal therapy, photodynamic therapy, and radiotherapy. However, there is still a major knowledge gap when it comes to transitioning from research to clinical applications. One of the important issues is that the biological toxicity of CNTs, especially in terms of carcinogenesis, and the underlying mechanisms are not fully understood. Therefore, a thorough evaluation of toxicity and the underlying mechanisms of carcinogenesis is essential to enable the wide application of CNTs. In this review, we summarize the recent progress of CNTs as multifunctional therapeutics in cancer therapy. Furthermore, a detailed discussion is provided on the carcinogenesis and potential mechanisms of CNTs. Finally, the review ends with further challenges and prospects for CNTs with the expectation of facilitating their broader utilization.
Full article

Figure 1
Figure 1
<p>(<b>A</b>) Functionalized SWCNTs loaded with drugs or gene products enter the cell, releasing the drugs or gene products into the cytoplasm [<a href="#B25-jnt-05-00006" class="html-bibr">25</a>]. Reproduced with permission from ref. [<a href="#B25-jnt-05-00006" class="html-bibr">25</a>]. Copyright © 2020 Elsevier. (<b>B</b>) TEM images of SWCNTs loaded with plasmid DNA [<a href="#B26-jnt-05-00006" class="html-bibr">26</a>]. (<b>a</b>) TEM image of SWCNTs. (<b>b</b>,<b>c</b>) TEM images of SWCNTs loaded with plasmid DNA. Globular and supercoiled structures of plasmid DNA (black arrows in (<b>b</b>,<b>c</b>)). Tighter packing of the SWCNTs (white arrows in (<b>c</b>)). Reproduced with permission from ref. [<a href="#B26-jnt-05-00006" class="html-bibr">26</a>]. Copyright © 2004 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim, respectively.</p> Full article ">Figure 2
<p>CNTs for cancer therapy. (<b>A</b>) Au combined with SWCNTs enhances the sensitivity of cancer cells to X-rays [<a href="#B33-jnt-05-00006" class="html-bibr">33</a>]. Reproduced with permission from ref. [<a href="#B33-jnt-05-00006" class="html-bibr">33</a>]. Copyright © 2020 Elsevier. (<b>B</b>) Ru@SWCNTs are developed for bimodal PTT and PDT [<a href="#B36-jnt-05-00006" class="html-bibr">36</a>]. Reproduced with permission from ref. [<a href="#B36-jnt-05-00006" class="html-bibr">36</a>]. Copyright © 2015 American Chemical Society.</p> Full article ">Figure 3
<p>Schematic diagram of CNTs for the regulation of immunity [<a href="#B38-jnt-05-00006" class="html-bibr">38</a>]. Reproduced with permission from ref. [<a href="#B38-jnt-05-00006" class="html-bibr">38</a>]. Copyright © 2024 Formstack, LLC.</p> Full article ">Figure 4
<p>Molecular imaging methods based on CNTs. (<b>A</b>) Encapsulation of SPIO in the center of polymer-modified MWCNTs by a microwave reaction [<a href="#B48-jnt-05-00006" class="html-bibr">48</a>]. Reproduced with permission from ref. [<a href="#B48-jnt-05-00006" class="html-bibr">48</a>]. Copyright © 2015, American Chemical Society. (<b>B</b>) An inner view of a CNT dynamic micro-CT scanner [<a href="#B50-jnt-05-00006" class="html-bibr">50</a>]. (<b>a</b>,<b>c</b>) CT slice images at the diastolic phase. (<b>b</b>,<b>d</b>) CT slice images at the systolic phase. Reproduced with permission from ref. [<a href="#B50-jnt-05-00006" class="html-bibr">50</a>]. Copyright © 2018, AIP Publishing, respectively.</p> Full article ">Figure 5
<p>Schematic illustration of how CNTs promote ROS production and induce cell damage. Reprinted from Accounts of Chemical Research [<a href="#B68-jnt-05-00006" class="html-bibr">68</a>]. Reproduced with permission from ref. [<a href="#B68-jnt-05-00006" class="html-bibr">68</a>]. Copyright © 2012 American Chemical Society.</p> Full article ">Figure 6
<p>Chronic exposure to SWCNTs induces the malignant transformation of epithelial cells [<a href="#B80-jnt-05-00006" class="html-bibr">80</a>]. (<b>A</b>) Dysregulation of DNA methylation leads to carcinogenesis. (<b>B</b>) H&E staining and IHC staining (Ki67, TTF-1, and P63) of malignant transformed tissues. Reproduced with permission from ref. [<a href="#B80-jnt-05-00006" class="html-bibr">80</a>]. Copyright © 2021 American Chemical Society.</p> Full article ">Figure 7
<p>Schematic illustration of nanoparticles penetrating the endothelium of a blood vessel [<a href="#B86-jnt-05-00006" class="html-bibr">86</a>]. Reproduced with permission from ref. [<a href="#B86-jnt-05-00006" class="html-bibr">86</a>]. Copyright © 2023 Lasak and Ciepluch.</p> Full article ">
<p>(<b>A</b>) Functionalized SWCNTs loaded with drugs or gene products enter the cell, releasing the drugs or gene products into the cytoplasm [<a href="#B25-jnt-05-00006" class="html-bibr">25</a>]. Reproduced with permission from ref. [<a href="#B25-jnt-05-00006" class="html-bibr">25</a>]. Copyright © 2020 Elsevier. (<b>B</b>) TEM images of SWCNTs loaded with plasmid DNA [<a href="#B26-jnt-05-00006" class="html-bibr">26</a>]. (<b>a</b>) TEM image of SWCNTs. (<b>b</b>,<b>c</b>) TEM images of SWCNTs loaded with plasmid DNA. Globular and supercoiled structures of plasmid DNA (black arrows in (<b>b</b>,<b>c</b>)). Tighter packing of the SWCNTs (white arrows in (<b>c</b>)). Reproduced with permission from ref. [<a href="#B26-jnt-05-00006" class="html-bibr">26</a>]. Copyright © 2004 WILEY-VCH Verlag GmbH & Co. KGaA, Weinheim, respectively.</p> Full article ">Figure 2
<p>CNTs for cancer therapy. (<b>A</b>) Au combined with SWCNTs enhances the sensitivity of cancer cells to X-rays [<a href="#B33-jnt-05-00006" class="html-bibr">33</a>]. Reproduced with permission from ref. [<a href="#B33-jnt-05-00006" class="html-bibr">33</a>]. Copyright © 2020 Elsevier. (<b>B</b>) Ru@SWCNTs are developed for bimodal PTT and PDT [<a href="#B36-jnt-05-00006" class="html-bibr">36</a>]. Reproduced with permission from ref. [<a href="#B36-jnt-05-00006" class="html-bibr">36</a>]. Copyright © 2015 American Chemical Society.</p> Full article ">Figure 3
<p>Schematic diagram of CNTs for the regulation of immunity [<a href="#B38-jnt-05-00006" class="html-bibr">38</a>]. Reproduced with permission from ref. [<a href="#B38-jnt-05-00006" class="html-bibr">38</a>]. Copyright © 2024 Formstack, LLC.</p> Full article ">Figure 4
<p>Molecular imaging methods based on CNTs. (<b>A</b>) Encapsulation of SPIO in the center of polymer-modified MWCNTs by a microwave reaction [<a href="#B48-jnt-05-00006" class="html-bibr">48</a>]. Reproduced with permission from ref. [<a href="#B48-jnt-05-00006" class="html-bibr">48</a>]. Copyright © 2015, American Chemical Society. (<b>B</b>) An inner view of a CNT dynamic micro-CT scanner [<a href="#B50-jnt-05-00006" class="html-bibr">50</a>]. (<b>a</b>,<b>c</b>) CT slice images at the diastolic phase. (<b>b</b>,<b>d</b>) CT slice images at the systolic phase. Reproduced with permission from ref. [<a href="#B50-jnt-05-00006" class="html-bibr">50</a>]. Copyright © 2018, AIP Publishing, respectively.</p> Full article ">Figure 5
<p>Schematic illustration of how CNTs promote ROS production and induce cell damage. Reprinted from Accounts of Chemical Research [<a href="#B68-jnt-05-00006" class="html-bibr">68</a>]. Reproduced with permission from ref. [<a href="#B68-jnt-05-00006" class="html-bibr">68</a>]. Copyright © 2012 American Chemical Society.</p> Full article ">Figure 6
<p>Chronic exposure to SWCNTs induces the malignant transformation of epithelial cells [<a href="#B80-jnt-05-00006" class="html-bibr">80</a>]. (<b>A</b>) Dysregulation of DNA methylation leads to carcinogenesis. (<b>B</b>) H&E staining and IHC staining (Ki67, TTF-1, and P63) of malignant transformed tissues. Reproduced with permission from ref. [<a href="#B80-jnt-05-00006" class="html-bibr">80</a>]. Copyright © 2021 American Chemical Society.</p> Full article ">Figure 7
<p>Schematic illustration of nanoparticles penetrating the endothelium of a blood vessel [<a href="#B86-jnt-05-00006" class="html-bibr">86</a>]. Reproduced with permission from ref. [<a href="#B86-jnt-05-00006" class="html-bibr">86</a>]. Copyright © 2023 Lasak and Ciepluch.</p> Full article ">
Open AccessArticle
Nano-Enabled Colorimetric Assay for the Detection of Paracoccidioides lutzii: Advancing Diagnostics with Nanotechnology
by
Olavo O. Comparato Filho, Marcela A. Cândido, Aveline Ventura, Flavia V. Morais and Leandro Raniero
J. Nanotheranostics 2024, 5(3), 75-83; https://doi.org/10.3390/jnt5030005 - 26 Jun 2024
Abstract
►▼
Show Figures
Deforestation is a common occurrence driven by agricultural expansion, urbanization, and infrastructure development. These activities often lead to increased human interaction with ecosystems, potentially exposing individuals to Paracoccidioides spores (P. brasiliensis and P. lutzii) found in the soil, resulting in Paracoccidioidomycosis
[...] Read more.
Deforestation is a common occurrence driven by agricultural expansion, urbanization, and infrastructure development. These activities often lead to increased human interaction with ecosystems, potentially exposing individuals to Paracoccidioides spores (P. brasiliensis and P. lutzii) found in the soil, resulting in Paracoccidioidomycosis (PCM). This fungal infection is endemic to specific regions in Latin America, such as Brazil, Colombia, Venezuela, and Argentina. Diagnosis typically involves a combination of clinical assessment, imaging techniques, and laboratory examinations. P. lutzii lacks the glycoprotein Gp43, a key antigenic protein utilized in serological tests for PCM diagnosis. In this study, a colorimetric test employing gold nanoparticles (AuNPs) and label-free methodology was employed for P. lutzii detection. The effectiveness of the label-free colorimetric test was assessed using a total of 100 samples. This detection was achieved through the amplification of the gp43 gene and the use of a specific probe (5′CAGGGGTGCG3′) in conjunction with AuNPs. The receiver operating characteristic curve was employed to assess the test, revealing that the method can accurately detect P. lutzii with a sensitivity of 100% and a specificity of 100%. The findings indicate a substantial impact on remote endemic regions attributable to the implementation of cost-effective diagnostic methodologies.
Full article

Figure 1
Figure 1
<p>Gold nanoparticle characterization: (<b>a</b>) morphology by TEM; (<b>b</b>) hydrodynamic size distribution and zeta potential value; (<b>c</b>) UV–visible spectrum.</p> Full article ">Figure 2
<p>UV–visible spectra of AuNPs from colorimetric tests. The negative result is the red curve, the positive result is the blue curve, and control AuNPs are shown in black.</p> Full article ">Figure 3
<p>UV–visible spectra of colorimetric test fitted by the Gaussian equation: (<b>a</b>) Gaussian area; (<b>b</b>) FWHM; (<b>c</b>) Gaussian center.</p> Full article ">Figure 4
<p>Automatic discrimination of the colorimetric test as determined by Equation (1). The integrated areas of UV–visible spectra were defined by the regions 488–530 nm and 533–575 nm.</p> Full article ">Figure 5
<p>Receiver operating characteristic curve of the colorimetric tests.</p> Full article ">
<p>Gold nanoparticle characterization: (<b>a</b>) morphology by TEM; (<b>b</b>) hydrodynamic size distribution and zeta potential value; (<b>c</b>) UV–visible spectrum.</p> Full article ">Figure 2
<p>UV–visible spectra of AuNPs from colorimetric tests. The negative result is the red curve, the positive result is the blue curve, and control AuNPs are shown in black.</p> Full article ">Figure 3
<p>UV–visible spectra of colorimetric test fitted by the Gaussian equation: (<b>a</b>) Gaussian area; (<b>b</b>) FWHM; (<b>c</b>) Gaussian center.</p> Full article ">Figure 4
<p>Automatic discrimination of the colorimetric test as determined by Equation (1). The integrated areas of UV–visible spectra were defined by the regions 488–530 nm and 533–575 nm.</p> Full article ">Figure 5
<p>Receiver operating characteristic curve of the colorimetric tests.</p> Full article ">
Open AccessReview
Titanium Dioxide-Based Nanoparticles to Enhance Radiation Therapy for Cancer: A Literature Review
by
Masao Nakayama, Hiroaki Akasaka, Ryohei Sasaki and Moshi Geso
J. Nanotheranostics 2024, 5(2), 60-74; https://doi.org/10.3390/jnt5020004 - 31 May 2024
Abstract
►▼
Show Figures
Titanium dioxide nanoparticles (TiO2 NPs) have been investigated as one of the potential dose enhancement agents for radiation therapy. The role of TiO2 NPs as a photodynamic sensitiser has been well documented, but its sensitisation with X-rays is not highlighted. Unlike
[...] Read more.
Titanium dioxide nanoparticles (TiO2 NPs) have been investigated as one of the potential dose enhancement agents for radiation therapy. The role of TiO2 NPs as a photodynamic sensitiser has been well documented, but its sensitisation with X-rays is not highlighted. Unlike other metal NPs, such as gold NPs, the main challenge for TiO2 NPs as radiosensitisers is their low atomic number, resulting in a small cross-section for X-rays. This review summarises the results of current research in this area to explore the dose enhancement inflicted by TiO2 NPs, which could potentially be of great value in improving radiation therapy efficiency.
Full article

Figure 1
Open AccessReview
Unlocking the Potential of Gold as Nanomedicine in Cancer Immunotherapy
by
Panangattukara Prabhakaran Praveen Kumar, Maggie Lee and Taeho Kim
J. Nanotheranostics 2024, 5(2), 29-59; https://doi.org/10.3390/jnt5020003 - 30 Apr 2024
Cited by 1
Abstract
Nanotechnology advancements have resulted in many sensors and devices for biomedical applications. Among the various nanomaterials, gold nanoparticles (AuNPs), due to their size, shape, biocompatibility, and unique plasmonic property, are an excellent candidate for many biomedical applications. AuNPs, known for their easy surface
[...] Read more.
Nanotechnology advancements have resulted in many sensors and devices for biomedical applications. Among the various nanomaterials, gold nanoparticles (AuNPs), due to their size, shape, biocompatibility, and unique plasmonic property, are an excellent candidate for many biomedical applications. AuNPs, known for their easy surface modifications, robust nature, and photothermal activities, find application in drug delivery and cancer treatment studies. In this review, we are highlighting the recent trends in using AuNPs as nanomedicine for cancer immunotherapy. Cancer immunotherapy not only eliminates the primary tumors but also allows for the treatment of metastasis along with the recurrence of the tumor. AuNPs possess tissue-specific delivery functions that depend on the tunability in size and surface functionalization of AuNPs. AuNPs can be used to activate the tumor’s immune defense ability, or they can be used to enhance the anti-tumor immune response. Understanding the interaction of the tumor environment and nanobiomedicine is very important. In the present review, we give an idea of the mode of action of AuNPs and various combinations of therapies for cancer immunotherapy.
Full article
(This article belongs to the Special Issue Interventional Nanotheranostics for Translational Nano-Immunotherapy)
►▼
Show Figures

Figure 1
Figure 1
<p>Represents the synthetic versatility and functionalization properties of AuNPs. The size and shape of AuNPs can be fine-tuned using different synthetic procedures, and reducing or stabilizing agents. Reproduced with permission from [<a href="#B36-jnt-05-00003" class="html-bibr">36</a>]. Copyright 2017, Elsevier.</p> Full article ">Figure 2
<p>A schematic representation of the cancer immunity cycle. Reproduced with permission from [<a href="#B49-jnt-05-00003" class="html-bibr">49</a>]. Copyright 2013, Elsevier.</p> Full article ">Figure 3
<p>Illustrates various aspects of the experimental procedure and outcomes. (<b>a</b>) outlines the treatment protocol involving intravenous injection in vivo. (<b>b</b>) depicts the progression of tumor growth alongside changes in body weight across different experimental groups of mice over time. (<b>c</b>) showcases the results of ELISA tests conducted on mouse serum to measure levels of IFN-γ and IL-2. (<b>d</b>) presents histological images of tumor sections stained with H&E and TUNEL, illustrating differences between mouse groups. (<b>e</b>,<b>f</b>) display representative immunofluorescence staining of tumor and spleen sections, respectively, highlighting the expression of CD3, CD4, and CD8 markers, with DAPI staining indicating tumor cell nuclei. Scale bar = 100 μm. *** <span class="html-italic">p</span> < 0.001. Reproduced with permission from [<a href="#B59-jnt-05-00003" class="html-bibr">59</a>]. Copyright 2020, Royal Society of Chemistry.</p> Full article ">Figure 4
<p>(<b>a</b>) Illustration of Au nanoprism-based nanocarrier for siRNA-PDL-1 complex in cancer immunotherapy. (<b>b</b>) Impact of hPD-L1 gene silencing on HCC827 cell growth after treatment with PBS, GNPs-siRNA, or GNPs-nsRNA. (<b>c</b>) Sequential photoacoustic imaging pre- and post-injection of GNPs-hPD-L1 siRNA at 1, 8, 24, and 48 h using a 650 nm laser. (<b>d</b>) Fluorescence images of Calcein AM/PI-stained HCC827 cells incubated with PBS, GNPs, and GNPs-siRNA post-laser irradiation (3 min, 0.8 W cm<sup>−2</sup>) for 24 h. Reproduced with permission from [<a href="#B92-jnt-05-00003" class="html-bibr">92</a>]. Copyright, 2019, Elsevier.</p> Full article ">Figure 5
<p>Representing the role of AuNPs for normalizing the tumor vasculature. (<b>a</b>) CD31 staining studies. Results showed that, by the administration of AuNPs, the tumor vessel area decreased. (<b>b</b>) The improved perfusion rate in the tumor by AuNPs as studied by the FITC-conjugated lectin (green) and CD31 (red) staining. (<b>c</b>) The reduced vascular leakage in the tumor vessels by AuNPs as studied by the FITC-dextran (green) and CD31 staining. (<b>d</b>) Improved hypoxia condition in tumor cells as studied by the pimonidazole staining after treatment with AuNPs. * <span class="html-italic">p</span> < 0.05, ** <span class="html-italic">p</span> < 0.01. Reproduced with permission from [<a href="#B95-jnt-05-00003" class="html-bibr">95</a>]. Copyright 2017, Dove Medical Press.</p> Full article ">Figure 6
<p>Insights into the mechanistic induction of the host immune system through the application of AuNP-based strategies for cancer treatment, including PDT and PTT.</p> Full article ">Figure 7
<p>(<b>a</b>) Schematic depiction illustrating the dynamic self-assembly process of AuNPs within liposomes. (<b>b</b>) Evolution of absorption peak shifts in AuNPs as influenced by varying compositions of AuNPs and liposomes. (<b>c</b>) Confocal microscopy images showcasing Calreticulin (CALR) expression in 4T1 tumor cell post-photothermal therapy (PTT) treatment. (<b>d</b>) Immunofluorescence staining analysis revealing the expression levels of HMGB1 in cancer cells following PTT intervention. Reproduced with permission from [<a href="#B108-jnt-05-00003" class="html-bibr">108</a>]. Copyright 2019, American Chemical Society.</p> Full article ">Figure 8
<p>Role of AuNPs in PTT based immunotherapy using immunoadjuvants. (<b>a</b>) A schematic for mPEG-GNRs@BSA/R837 nanocomplexes synthesizes and elucidates the mechanism underlying their stimulation of anti-tumor immune responses. (<b>b</b>) TNF-α, IL-6, and IL-12 cytokines levels measured in the serum of mice after three days of laser treatment. (<b>c</b>) A schematic representation of gold Nanoshell drug delivery system. (<b>d</b>) Induction of dendritic cell maturation through siRNA@CPG@Gold Nanoshell-mediated photothermal therapy in mice with MFC tumors. Cells from the lymph nodes draining the tumors were harvested 72 h post-treatment and analyzed via flow cytometry after staining for CD80 and CD86. Where * <span class="html-italic">p</span> < 0.05, ** <span class="html-italic">p</span> < 0.01 and *** <span class="html-italic">p</span> < 0.001. Reproduced with permission from [<a href="#B109-jnt-05-00003" class="html-bibr">109</a>,<a href="#B110-jnt-05-00003" class="html-bibr">110</a>]. Copyright 2018, Royal Society of Chemistry. Copyright 2019, Springer.</p> Full article ">Figure 9
<p>Working principle of the AuNCs@MnO<sub>2</sub> (AM) nanomaterial for ICD, (<b>a</b>) A synthetic route for the preparation and generation of O<sub>2</sub> from AM. (<b>b</b>) Therapeutic application of AM for the generation of cytokines and imaging studies. (<b>c</b>–<b>e</b>) Detection of various ICD signal molecules after AM + Laser treatment and PB is used as a control for the studies. (<b>c</b>) Fluorescence microscopic images for CRT expression in 4T1 cells. (<b>d</b>,<b>e</b>) Released ATP and HMGB1 in the supernatant after the AM + Laser treatment. (<b>f</b>) Schematic representation for the DC activation by ICD. (<b>g</b>) The expression for CD83 and CD86 after DC maturation. (<b>h</b>) The generated IL-12 in the culture supernatant. The asterisks indicate differences between PBS and other treatments are statistically significant. ** <span class="html-italic">p</span> < 0.01, *** <span class="html-italic">p</span> < 0.001. # Differences between the two groups are statistically significant; # <span class="html-italic">p</span> < 0.05. (n = 5). Reproduced with permission from [<a href="#B126-jnt-05-00003" class="html-bibr">126</a>], Copyright 2018, Elsevier.</p> Full article ">Figure 10
<p>(<b>a</b>) A schematic representation for the glutathione-based AuNC formation and complexation of 177Lu or 99Tc. (<b>b</b>) Cartoon diagram showing the cancer immunotherapy pathway and the immune checkpoint blockade pathways. (<b>c</b>) The internalization and SPECT/CT images for 99Tc and 177Lu@GSH-AuNCs in 4T1 tumor-bearing mice after regular intervals of time. (<b>d</b>) represents the DC maturation in CT26 cells using the various radionuclides and the 99Tc and 177Lu@GSH-AuNCs, where <span class="html-italic">p</span> values were calculated by multiple <span class="html-italic">t</span>-tests (*** <span class="html-italic">p</span> < 0.001). Reproduced with permission from [<a href="#B144-jnt-05-00003" class="html-bibr">144</a>], Copyright 2021, Elsevier.</p> Full article ">
<p>Represents the synthetic versatility and functionalization properties of AuNPs. The size and shape of AuNPs can be fine-tuned using different synthetic procedures, and reducing or stabilizing agents. Reproduced with permission from [<a href="#B36-jnt-05-00003" class="html-bibr">36</a>]. Copyright 2017, Elsevier.</p> Full article ">Figure 2
<p>A schematic representation of the cancer immunity cycle. Reproduced with permission from [<a href="#B49-jnt-05-00003" class="html-bibr">49</a>]. Copyright 2013, Elsevier.</p> Full article ">Figure 3
<p>Illustrates various aspects of the experimental procedure and outcomes. (<b>a</b>) outlines the treatment protocol involving intravenous injection in vivo. (<b>b</b>) depicts the progression of tumor growth alongside changes in body weight across different experimental groups of mice over time. (<b>c</b>) showcases the results of ELISA tests conducted on mouse serum to measure levels of IFN-γ and IL-2. (<b>d</b>) presents histological images of tumor sections stained with H&E and TUNEL, illustrating differences between mouse groups. (<b>e</b>,<b>f</b>) display representative immunofluorescence staining of tumor and spleen sections, respectively, highlighting the expression of CD3, CD4, and CD8 markers, with DAPI staining indicating tumor cell nuclei. Scale bar = 100 μm. *** <span class="html-italic">p</span> < 0.001. Reproduced with permission from [<a href="#B59-jnt-05-00003" class="html-bibr">59</a>]. Copyright 2020, Royal Society of Chemistry.</p> Full article ">Figure 4
<p>(<b>a</b>) Illustration of Au nanoprism-based nanocarrier for siRNA-PDL-1 complex in cancer immunotherapy. (<b>b</b>) Impact of hPD-L1 gene silencing on HCC827 cell growth after treatment with PBS, GNPs-siRNA, or GNPs-nsRNA. (<b>c</b>) Sequential photoacoustic imaging pre- and post-injection of GNPs-hPD-L1 siRNA at 1, 8, 24, and 48 h using a 650 nm laser. (<b>d</b>) Fluorescence images of Calcein AM/PI-stained HCC827 cells incubated with PBS, GNPs, and GNPs-siRNA post-laser irradiation (3 min, 0.8 W cm<sup>−2</sup>) for 24 h. Reproduced with permission from [<a href="#B92-jnt-05-00003" class="html-bibr">92</a>]. Copyright, 2019, Elsevier.</p> Full article ">Figure 5
<p>Representing the role of AuNPs for normalizing the tumor vasculature. (<b>a</b>) CD31 staining studies. Results showed that, by the administration of AuNPs, the tumor vessel area decreased. (<b>b</b>) The improved perfusion rate in the tumor by AuNPs as studied by the FITC-conjugated lectin (green) and CD31 (red) staining. (<b>c</b>) The reduced vascular leakage in the tumor vessels by AuNPs as studied by the FITC-dextran (green) and CD31 staining. (<b>d</b>) Improved hypoxia condition in tumor cells as studied by the pimonidazole staining after treatment with AuNPs. * <span class="html-italic">p</span> < 0.05, ** <span class="html-italic">p</span> < 0.01. Reproduced with permission from [<a href="#B95-jnt-05-00003" class="html-bibr">95</a>]. Copyright 2017, Dove Medical Press.</p> Full article ">Figure 6
<p>Insights into the mechanistic induction of the host immune system through the application of AuNP-based strategies for cancer treatment, including PDT and PTT.</p> Full article ">Figure 7
<p>(<b>a</b>) Schematic depiction illustrating the dynamic self-assembly process of AuNPs within liposomes. (<b>b</b>) Evolution of absorption peak shifts in AuNPs as influenced by varying compositions of AuNPs and liposomes. (<b>c</b>) Confocal microscopy images showcasing Calreticulin (CALR) expression in 4T1 tumor cell post-photothermal therapy (PTT) treatment. (<b>d</b>) Immunofluorescence staining analysis revealing the expression levels of HMGB1 in cancer cells following PTT intervention. Reproduced with permission from [<a href="#B108-jnt-05-00003" class="html-bibr">108</a>]. Copyright 2019, American Chemical Society.</p> Full article ">Figure 8
<p>Role of AuNPs in PTT based immunotherapy using immunoadjuvants. (<b>a</b>) A schematic for mPEG-GNRs@BSA/R837 nanocomplexes synthesizes and elucidates the mechanism underlying their stimulation of anti-tumor immune responses. (<b>b</b>) TNF-α, IL-6, and IL-12 cytokines levels measured in the serum of mice after three days of laser treatment. (<b>c</b>) A schematic representation of gold Nanoshell drug delivery system. (<b>d</b>) Induction of dendritic cell maturation through siRNA@CPG@Gold Nanoshell-mediated photothermal therapy in mice with MFC tumors. Cells from the lymph nodes draining the tumors were harvested 72 h post-treatment and analyzed via flow cytometry after staining for CD80 and CD86. Where * <span class="html-italic">p</span> < 0.05, ** <span class="html-italic">p</span> < 0.01 and *** <span class="html-italic">p</span> < 0.001. Reproduced with permission from [<a href="#B109-jnt-05-00003" class="html-bibr">109</a>,<a href="#B110-jnt-05-00003" class="html-bibr">110</a>]. Copyright 2018, Royal Society of Chemistry. Copyright 2019, Springer.</p> Full article ">Figure 9
<p>Working principle of the AuNCs@MnO<sub>2</sub> (AM) nanomaterial for ICD, (<b>a</b>) A synthetic route for the preparation and generation of O<sub>2</sub> from AM. (<b>b</b>) Therapeutic application of AM for the generation of cytokines and imaging studies. (<b>c</b>–<b>e</b>) Detection of various ICD signal molecules after AM + Laser treatment and PB is used as a control for the studies. (<b>c</b>) Fluorescence microscopic images for CRT expression in 4T1 cells. (<b>d</b>,<b>e</b>) Released ATP and HMGB1 in the supernatant after the AM + Laser treatment. (<b>f</b>) Schematic representation for the DC activation by ICD. (<b>g</b>) The expression for CD83 and CD86 after DC maturation. (<b>h</b>) The generated IL-12 in the culture supernatant. The asterisks indicate differences between PBS and other treatments are statistically significant. ** <span class="html-italic">p</span> < 0.01, *** <span class="html-italic">p</span> < 0.001. # Differences between the two groups are statistically significant; # <span class="html-italic">p</span> < 0.05. (n = 5). Reproduced with permission from [<a href="#B126-jnt-05-00003" class="html-bibr">126</a>], Copyright 2018, Elsevier.</p> Full article ">Figure 10
<p>(<b>a</b>) A schematic representation for the glutathione-based AuNC formation and complexation of 177Lu or 99Tc. (<b>b</b>) Cartoon diagram showing the cancer immunotherapy pathway and the immune checkpoint blockade pathways. (<b>c</b>) The internalization and SPECT/CT images for 99Tc and 177Lu@GSH-AuNCs in 4T1 tumor-bearing mice after regular intervals of time. (<b>d</b>) represents the DC maturation in CT26 cells using the various radionuclides and the 99Tc and 177Lu@GSH-AuNCs, where <span class="html-italic">p</span> values were calculated by multiple <span class="html-italic">t</span>-tests (*** <span class="html-italic">p</span> < 0.001). Reproduced with permission from [<a href="#B144-jnt-05-00003" class="html-bibr">144</a>], Copyright 2021, Elsevier.</p> Full article ">
Open AccessArticle
Graphene Oxide Chemical Refining Screening to Improve Blood Compatibility of Graphene-Based Nanomaterials
by
Fabio Pieretti, Alessandro Moretto, Emanuele Papini and Regina Tavano
J. Nanotheranostics 2024, 5(1), 13-28; https://doi.org/10.3390/jnt5010002 - 20 Feb 2024
Abstract
Graphene oxide (GO) nanoparticles, due to their favorable water solubility, compared to graphene (GA), are a hot research topic in biomedical and pharmaceutical research. However, GO clinical translation may be complicated by its high surface/volume ratio enhancing the interaction with human blood components.
[...] Read more.
Graphene oxide (GO) nanoparticles, due to their favorable water solubility, compared to graphene (GA), are a hot research topic in biomedical and pharmaceutical research. However, GO clinical translation may be complicated by its high surface/volume ratio enhancing the interaction with human blood components. In fact, GO’s bi-dimensional nature and strong negative charge may lead to severe biological effects, such as thrombogenicity and immune cell activation. This study explores the impact of further GO surface chemical modulation on major adverse effects: blood plasma coagulation and hemolysis. To this aim, we refined GO nanoparticles by fine-tuned reduction chemistry, esterification and introduction of negative or positive charges. With this approach, we were able to mitigate plasma coagulation and hemolysis at variable degrees and to identify GO derivatives with improved biocompatibility. This opens the door to the progress of graphene-based nanotheranostic applications.
Full article
(This article belongs to the Special Issue Carbon Nanomaterials as Nano-Theranostic Tools in Disease Treatment)
►▼
Show Figures
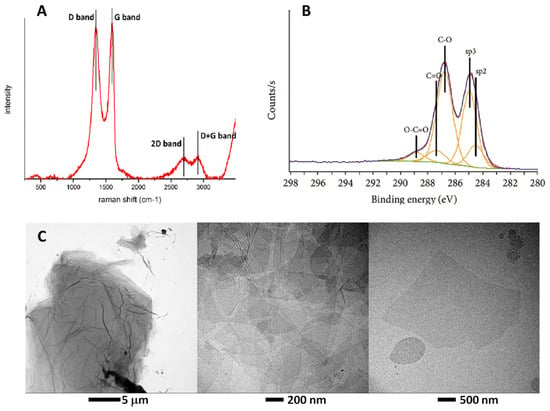
Figure 1
Figure 1
<p>(<b>A</b>) Raman spectra of GO. (<b>B</b>) XPS spectra of GO in the C1s range (purple line) and a multipeak analysis for the carbon 1s (orange line). Green line was used to fit baseline. (<b>C</b>) TEM image of the GO showing aggregates (left), stacked flakes (centre) and single flakes (right).</p> Full article ">Figure 2
<p>(<b>A</b>) UV spectra of reduced GO and rGO samples, obtained at a concentration of 0.05 mg/mL and pH 10. (<b>B</b>) Solid state FT-IR spectra of GO and rGO samples.</p> Full article ">Figure 3
<p>(<b>A</b>) Comparison of UV-Vis spectra of GO and eGO taken, respectively, at the concentrations 0.025 mg/m. (<b>B</b>) comparison of solid-state FT-IR spectra of GO and eGO.</p> Full article ">Figure 4
<p>TEM images of cysGO, met-cysGO and met-cys-e-GO.</p> Full article ">Figure 5
<p>Hemolytic activity for GO and its selected reduced and functionalized derivatives at the concentrations of 2.5 μg/mL and 25 μg/mL, after 18 (left panels) and 24 h (right panels)-incubation with RBCs. Each data point is presented as mean ± absolute error (<span class="html-italic">n</span> = 2). Data are expressed as % of total hemolysis induced by pure distilled water (100% release).</p> Full article ">Figure 6
<p>Hemolytic activity in HS and saline solution for GO and its selected reduced and functionalized derivatives at the concentration of 2.5 μg/mL, after 18 (left panels) and 24 h of incubation with RBCs (right panels). Each data point is presented as mean ± absolute error (<span class="html-italic">n</span> = 2).</p> Full article ">Figure 7
<p>Comparison between <span class="html-italic">ζ</span>-potential measures (black squares) and EC50 (blue squares) for GO and its functionalized derivatives. <span class="html-italic">ζ</span>-potential data are presented as mean ± standard deviation (<span class="html-italic">n</span> = 3).</p> Full article ">Figure 8
<p>Pro-coagulant activity data for GO. Histograms representing values of t<sub>1/2</sub> for the corresponding concentration of nanomaterial.</p> Full article ">Scheme 1
<p>Schematic chemical structure of GO, its derivatives showing different degrees of reduction (top panel), and functionalized derivatives of GO (bottom panel) investigated in this study.</p> Full article ">
<p>(<b>A</b>) Raman spectra of GO. (<b>B</b>) XPS spectra of GO in the C1s range (purple line) and a multipeak analysis for the carbon 1s (orange line). Green line was used to fit baseline. (<b>C</b>) TEM image of the GO showing aggregates (left), stacked flakes (centre) and single flakes (right).</p> Full article ">Figure 2
<p>(<b>A</b>) UV spectra of reduced GO and rGO samples, obtained at a concentration of 0.05 mg/mL and pH 10. (<b>B</b>) Solid state FT-IR spectra of GO and rGO samples.</p> Full article ">Figure 3
<p>(<b>A</b>) Comparison of UV-Vis spectra of GO and eGO taken, respectively, at the concentrations 0.025 mg/m. (<b>B</b>) comparison of solid-state FT-IR spectra of GO and eGO.</p> Full article ">Figure 4
<p>TEM images of cysGO, met-cysGO and met-cys-e-GO.</p> Full article ">Figure 5
<p>Hemolytic activity for GO and its selected reduced and functionalized derivatives at the concentrations of 2.5 μg/mL and 25 μg/mL, after 18 (left panels) and 24 h (right panels)-incubation with RBCs. Each data point is presented as mean ± absolute error (<span class="html-italic">n</span> = 2). Data are expressed as % of total hemolysis induced by pure distilled water (100% release).</p> Full article ">Figure 6
<p>Hemolytic activity in HS and saline solution for GO and its selected reduced and functionalized derivatives at the concentration of 2.5 μg/mL, after 18 (left panels) and 24 h of incubation with RBCs (right panels). Each data point is presented as mean ± absolute error (<span class="html-italic">n</span> = 2).</p> Full article ">Figure 7
<p>Comparison between <span class="html-italic">ζ</span>-potential measures (black squares) and EC50 (blue squares) for GO and its functionalized derivatives. <span class="html-italic">ζ</span>-potential data are presented as mean ± standard deviation (<span class="html-italic">n</span> = 3).</p> Full article ">Figure 8
<p>Pro-coagulant activity data for GO. Histograms representing values of t<sub>1/2</sub> for the corresponding concentration of nanomaterial.</p> Full article ">Scheme 1
<p>Schematic chemical structure of GO, its derivatives showing different degrees of reduction (top panel), and functionalized derivatives of GO (bottom panel) investigated in this study.</p> Full article ">
Open AccessReview
The Role of Fullerenes in Neurodegenerative Disorders
by
Daisy L. Wilson, Jyoti Ahlawat and Mahesh Narayan
J. Nanotheranostics 2024, 5(1), 1-12; https://doi.org/10.3390/jnt5010001 - 16 Jan 2024
Abstract
The use of carbon nanomaterials including fullerenes, carbon nanotubes, carbon nano-onions, carbon dots and carbon quantum dots for environmental applications has increased substantially. These nanoparticles are now used in the development of sensors and switches, in agriculture as smart fertilizers and in the
[...] Read more.
The use of carbon nanomaterials including fullerenes, carbon nanotubes, carbon nano-onions, carbon dots and carbon quantum dots for environmental applications has increased substantially. These nanoparticles are now used in the development of sensors and switches, in agriculture as smart fertilizers and in the biomedical realm for cancer therapy intervention, as antioxidants, in gene delivery and as theranostics. Here, we review the role of fullerenes as neuroprotectants. Their sp2 hybridized architectures and ability to intervene in the soluble-to-toxic transformation of amyloidogenic trajectories is highlighted here, along with other physico–chemical properties that impact interventional efficacy. Also highlighted are drawbacks that need to be overcome and future prospects.
Full article
(This article belongs to the Special Issue Advances and Innovations in Theranostic Nanobiomaterials)
►▼
Show Figures
Figure 1
Figure 1
<p>Schematic representation of C<sub>60</sub> and its properties and applications.</p> Full article ">Figure 2
<p>Trajectory of amyloid aggregation induced by oxidative stress and the subsequent neuronal and cognitive decline.</p> Full article ">Figure 3
<p>TEM image of lysozyme fibrils produced in the lab of Dr. Narayan. The inset shows an image of various stage of amyloid during fibril formation.</p> Full article ">Figure 4
<p>NACore aggregation morphology probed by TEM imaging. TEM images of (<b>a</b>) hydrophobic C<sub>60</sub>, (<b>b</b>) amphiphilic C<sub>60</sub>(OH)<sub>24</sub>, and (<b>c</b>) hydrophilic C<sub>60</sub>(OH)<sub>40</sub>. NAcore aggregates in the absence (<b>d</b>,<b>h</b>) and presence of (<b>e</b>,<b>i</b>) C<sub>60</sub>, (<b>f</b>,<b>g</b>) C<sub>60</sub>(OH)<sub>24</sub>, and (<b>j</b>,<b>k</b>) C<sub>60</sub>(OH)<sub>40</sub>. Reproduced with permission from Ref. [<a href="#B35-jnt-05-00001" class="html-bibr">35</a>].</p> Full article ">Figure 5
<p>Schematic representation of the antioxidative effect of fullerenes.</p> Full article ">
<p>Schematic representation of C<sub>60</sub> and its properties and applications.</p> Full article ">Figure 2
<p>Trajectory of amyloid aggregation induced by oxidative stress and the subsequent neuronal and cognitive decline.</p> Full article ">Figure 3
<p>TEM image of lysozyme fibrils produced in the lab of Dr. Narayan. The inset shows an image of various stage of amyloid during fibril formation.</p> Full article ">Figure 4
<p>NACore aggregation morphology probed by TEM imaging. TEM images of (<b>a</b>) hydrophobic C<sub>60</sub>, (<b>b</b>) amphiphilic C<sub>60</sub>(OH)<sub>24</sub>, and (<b>c</b>) hydrophilic C<sub>60</sub>(OH)<sub>40</sub>. NAcore aggregates in the absence (<b>d</b>,<b>h</b>) and presence of (<b>e</b>,<b>i</b>) C<sub>60</sub>, (<b>f</b>,<b>g</b>) C<sub>60</sub>(OH)<sub>24</sub>, and (<b>j</b>,<b>k</b>) C<sub>60</sub>(OH)<sub>40</sub>. Reproduced with permission from Ref. [<a href="#B35-jnt-05-00001" class="html-bibr">35</a>].</p> Full article ">Figure 5
<p>Schematic representation of the antioxidative effect of fullerenes.</p> Full article ">
Open AccessArticle
Efficacy of 15 nm Gold Nanoparticles for Image-Guided Gliosarcoma Radiotherapy
by
Elette Engels, Michael Lerch, Stéphanie Corde and Moeava Tehei
J. Nanotheranostics 2023, 4(4), 480-495; https://doi.org/10.3390/jnt4040021 - 26 Oct 2023
Cited by 1
Abstract
Targeted brain cancer treatments are sorely needed to improve long-term prognosis, particularly for gliosarcoma and glioblastoma patients. Gold nanoparticles (GNPs) have unique properties including high atomic number, biocompatibility, and small size for cancer cell internalization. GNPs are consequently an ideal candidate for improved
[...] Read more.
Targeted brain cancer treatments are sorely needed to improve long-term prognosis, particularly for gliosarcoma and glioblastoma patients. Gold nanoparticles (GNPs) have unique properties including high atomic number, biocompatibility, and small size for cancer cell internalization. GNPs are consequently an ideal candidate for improved cancer targeting using image-guided radiotherapy. This work investigated 15 nm AuroVistTM GNPs for image-guided gliosarcoma radiotherapy and identified optimum GNP concentrations. The GNPs were found to be 15–20 nm using optical surface plasmon resonance absorption, with a (41.3 ± 0.3) nm hydrodynamic diameter. Confocal imaging showed that 50–500 µg/mL of the GNPs was well-internalized into the 9L cells within 24–48 h. γ-H2AX assays showed that 50–500 µg/mL of the GNPs radiosensitized the 9L cells irradiated with 125 and 150 kVp X-rays. However, only 500 µg/mL of the GNPs produced significant long-term dose enhancement with 150 kVp X-rays (with a sensitization enhancement ratio at 10% survival of 1.43, and 1.13 with 50 µg/mL) using clonogenic assay. CT imaging of the GNPs in the 9L tumors in Fischer rats further showed that GNP concentrations above 500 µg/mL were required to distinguish the tumor from the brain, and the GNPs were detected 48 h after injection. These promising results indicate that the GNPs can be used for selective gliosarcoma treatment with image-guided X-ray radiotherapy at concentrations above 500 µg/mL.
Full article
(This article belongs to the Special Issue Emerging Strategies in Nanomedicine)
►▼
Show Figures
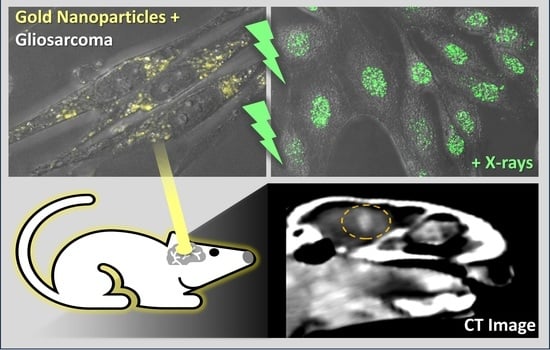
Graphical abstract
Graphical abstract
Full article ">Figure 1
<p>Dynamic light scatter output of the Malvern Zetasizer system analysis of 1 mg/mL GNPs, including raw correlation data (<b>a</b>) and size distribution in solution (<b>b</b>).</p> Full article ">Figure 2
<p>Optimal absorbance of AuroVist<sup>TM</sup> GNPs at concentrations of 0.125–0.625 mg/mL in DPBS, measured with a SpectraMax plate reader.</p> Full article ">Figure 3
<p>Confocal microscopy images of 9LGS cells acquired with 63× oil objective lens without GNPS (<b>a</b>) incubated with 50 µg/mL (<b>b</b>) and with 500 µg/mL (<b>c</b>) GNPs. Images are projected over 16 µm (42 slices in 0.38 µm steps) and the brightfield and 488 nm laser illuminated channels are overlayed. A summary of the estimated quantity of GNPs from the confocal images is shown for each GNP concentration (n = 15) (<b>d</b>). GNP uptake was also investigated with FACS (10,000 events, n = 3) using 50 µg/mL of GNPs incubated with 9LGS cells for 24 h and 48 h (<b>e</b>).</p> Full article ">Figure 4
<p>Clonogenic assay survival data following irradiation of 9LGS cells with and without GNPs in concentrations of 50 µg/mL and 500 µg/mL (also shown in [<a href="#B13-jnt-04-00021" class="html-bibr">13</a>]). Survival curves are fitted with the linear quadratic model equation [<a href="#B34-jnt-04-00021" class="html-bibr">34</a>].</p> Full article ">Figure 5
<p>DNA damage quantification using γ-H2AX confocal imaging following 2 Gy irradiation of 9LGS with GNPs (0–500 µg/mL) using 125 kVp and 150 kVp X-rays. A projection of γ-H2AX foci produced by the 150 kVp X-rays is shown overlayed with brightfield (<b>a</b>), including an insert of the 488 nm channel in one slice for three cells in each group. Confocal images were obtained 61–74 individual cells over four sets to obtain the average number (<b>b</b>) and area (<b>c</b>) of the foci of each treatment. The mass energy absorption coefficient ratio of gold to water [<a href="#B36-jnt-04-00021" class="html-bibr">36</a>] is compared with the 125 kVp and 150 kVp spectra generated with SpekCalc [<a href="#B37-jnt-04-00021" class="html-bibr">37</a>], (<b>d</b>).</p> Full article ">Figure 5 Cont.
<p>DNA damage quantification using γ-H2AX confocal imaging following 2 Gy irradiation of 9LGS with GNPs (0–500 µg/mL) using 125 kVp and 150 kVp X-rays. A projection of γ-H2AX foci produced by the 150 kVp X-rays is shown overlayed with brightfield (<b>a</b>), including an insert of the 488 nm channel in one slice for three cells in each group. Confocal images were obtained 61–74 individual cells over four sets to obtain the average number (<b>b</b>) and area (<b>c</b>) of the foci of each treatment. The mass energy absorption coefficient ratio of gold to water [<a href="#B36-jnt-04-00021" class="html-bibr">36</a>] is compared with the 125 kVp and 150 kVp spectra generated with SpekCalc [<a href="#B37-jnt-04-00021" class="html-bibr">37</a>], (<b>d</b>).</p> Full article ">Figure 6
<p>GNP CT contrast, uptake, and diffusion with time in 9LGS tumors. Overall, 100 kVp images were acquired with a Toshiba Asteion following GNP injection up to 48 h (<b>a</b>). The contrast enhancement ratio was determined between the tumor with GNPs to the brain (<b>b</b>). The GNP concentration and corresponding image contrast were compared using 100 kVp and 120 kVp CTs (<b>c</b>), which determined the measured concentration of GNPs in the tumor at 4 h, 24 h, and 48 h (<b>d</b>). This was compared with the calculated GNP concentration in the tumors using the 40 µg in tumor volumes from day 12 to day 14 (48 h) (<b>e</b>).</p> Full article ">
Full article ">Figure 1
<p>Dynamic light scatter output of the Malvern Zetasizer system analysis of 1 mg/mL GNPs, including raw correlation data (<b>a</b>) and size distribution in solution (<b>b</b>).</p> Full article ">Figure 2
<p>Optimal absorbance of AuroVist<sup>TM</sup> GNPs at concentrations of 0.125–0.625 mg/mL in DPBS, measured with a SpectraMax plate reader.</p> Full article ">Figure 3
<p>Confocal microscopy images of 9LGS cells acquired with 63× oil objective lens without GNPS (<b>a</b>) incubated with 50 µg/mL (<b>b</b>) and with 500 µg/mL (<b>c</b>) GNPs. Images are projected over 16 µm (42 slices in 0.38 µm steps) and the brightfield and 488 nm laser illuminated channels are overlayed. A summary of the estimated quantity of GNPs from the confocal images is shown for each GNP concentration (n = 15) (<b>d</b>). GNP uptake was also investigated with FACS (10,000 events, n = 3) using 50 µg/mL of GNPs incubated with 9LGS cells for 24 h and 48 h (<b>e</b>).</p> Full article ">Figure 4
<p>Clonogenic assay survival data following irradiation of 9LGS cells with and without GNPs in concentrations of 50 µg/mL and 500 µg/mL (also shown in [<a href="#B13-jnt-04-00021" class="html-bibr">13</a>]). Survival curves are fitted with the linear quadratic model equation [<a href="#B34-jnt-04-00021" class="html-bibr">34</a>].</p> Full article ">Figure 5
<p>DNA damage quantification using γ-H2AX confocal imaging following 2 Gy irradiation of 9LGS with GNPs (0–500 µg/mL) using 125 kVp and 150 kVp X-rays. A projection of γ-H2AX foci produced by the 150 kVp X-rays is shown overlayed with brightfield (<b>a</b>), including an insert of the 488 nm channel in one slice for three cells in each group. Confocal images were obtained 61–74 individual cells over four sets to obtain the average number (<b>b</b>) and area (<b>c</b>) of the foci of each treatment. The mass energy absorption coefficient ratio of gold to water [<a href="#B36-jnt-04-00021" class="html-bibr">36</a>] is compared with the 125 kVp and 150 kVp spectra generated with SpekCalc [<a href="#B37-jnt-04-00021" class="html-bibr">37</a>], (<b>d</b>).</p> Full article ">Figure 5 Cont.
<p>DNA damage quantification using γ-H2AX confocal imaging following 2 Gy irradiation of 9LGS with GNPs (0–500 µg/mL) using 125 kVp and 150 kVp X-rays. A projection of γ-H2AX foci produced by the 150 kVp X-rays is shown overlayed with brightfield (<b>a</b>), including an insert of the 488 nm channel in one slice for three cells in each group. Confocal images were obtained 61–74 individual cells over four sets to obtain the average number (<b>b</b>) and area (<b>c</b>) of the foci of each treatment. The mass energy absorption coefficient ratio of gold to water [<a href="#B36-jnt-04-00021" class="html-bibr">36</a>] is compared with the 125 kVp and 150 kVp spectra generated with SpekCalc [<a href="#B37-jnt-04-00021" class="html-bibr">37</a>], (<b>d</b>).</p> Full article ">Figure 6
<p>GNP CT contrast, uptake, and diffusion with time in 9LGS tumors. Overall, 100 kVp images were acquired with a Toshiba Asteion following GNP injection up to 48 h (<b>a</b>). The contrast enhancement ratio was determined between the tumor with GNPs to the brain (<b>b</b>). The GNP concentration and corresponding image contrast were compared using 100 kVp and 120 kVp CTs (<b>c</b>), which determined the measured concentration of GNPs in the tumor at 4 h, 24 h, and 48 h (<b>d</b>). This was compared with the calculated GNP concentration in the tumors using the 40 µg in tumor volumes from day 12 to day 14 (48 h) (<b>e</b>).</p> Full article ">
Open AccessReview
Enhancing Antibody Exposure in the Central Nervous System: Mechanisms of Uptake, Clearance, and Strategies for Improved Brain Delivery
by
Kelly Schwinghamer and Teruna J. Siahaan
J. Nanotheranostics 2023, 4(4), 463-479; https://doi.org/10.3390/jnt4040020 - 2 Oct 2023
Cited by 1
Abstract
Antibodies (mAbs) are attractive molecules for their application as a diagnostic and therapeutic agent for diseases of the central nervous system (CNS). mAbs can be generated to have high affinity and specificity to target molecules in the CNS. Unfortunately, only a very small
[...] Read more.
Antibodies (mAbs) are attractive molecules for their application as a diagnostic and therapeutic agent for diseases of the central nervous system (CNS). mAbs can be generated to have high affinity and specificity to target molecules in the CNS. Unfortunately, only a very small number of mAbs have been specifically developed and approved for neurological indications. This is primarily attributed to their low exposure within the CNS, hindering their ability to reach and effectively engage their potential targets in the brain. This review discusses aspects of various barriers such as the blood–brain barrier (BBB) and blood–cerebrospinal fluid (CSF) barrier (BCSFB) that regulate the entry and clearance of mAbs into and from the brain. The roles of the glymphatic system on brain exposure and clearance are being described. We also discuss the proposed mechanisms of the uptake of mAbs into the brain and for clearance. Finally, several methods of enhancing the exposure of mAbs in the CNS were discussed, including receptor-mediated transcytosis, osmotic BBB opening, focused ultrasound (FUS), BBB-modulating peptides, and enhancement of mAb brain retention.
Full article
(This article belongs to the Special Issue Exclusive Papers of the Editorial Board Members)
►▼
Show Figures
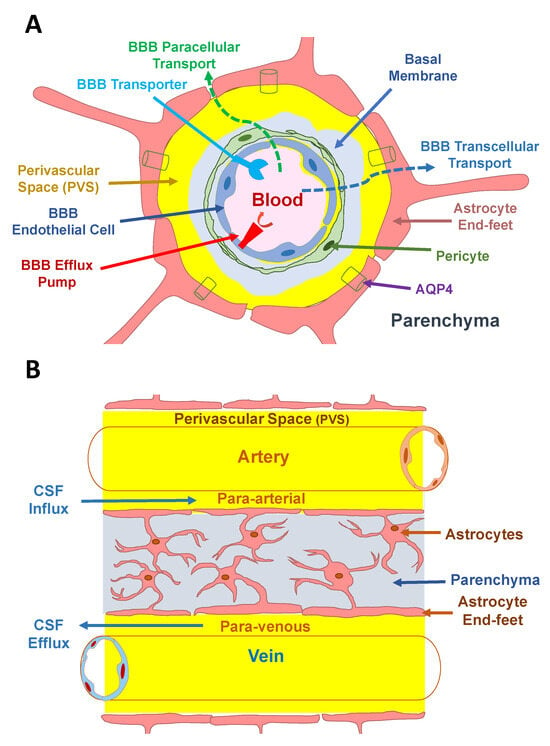
Figure 1
Figure 1
<p>(<b>A</b>) A schematic of the BBB that is composed of endothelial cells (blue) surrounded by supportive pericytes (green) and astrocytic endfeet (pink). The basement membrane (light blue) is shared between pericytes and endothelial, while perivascular spaces (yellow) are located between the basement membrane and astrocytic endfeet and are filled with CSF. AQP4 channels on astrocytic endfeet mediate water flux into PVS. (<b>B</b>) In the glymphatic system, fluid movement is facilitated by AQP4 channels located on astrocytic endfeet, driving convective flow from the CSF-filled periarterial spaces to the perivenous spaces. This convective flow within the brain parenchyma is believed to contribute to the clearance of waste products from the brain. Subsequently, the parenchymal waste present in the CSF is drained into the peripheral lymphatics through the perivenous spaces.</p> Full article ">
<p>(<b>A</b>) A schematic of the BBB that is composed of endothelial cells (blue) surrounded by supportive pericytes (green) and astrocytic endfeet (pink). The basement membrane (light blue) is shared between pericytes and endothelial, while perivascular spaces (yellow) are located between the basement membrane and astrocytic endfeet and are filled with CSF. AQP4 channels on astrocytic endfeet mediate water flux into PVS. (<b>B</b>) In the glymphatic system, fluid movement is facilitated by AQP4 channels located on astrocytic endfeet, driving convective flow from the CSF-filled periarterial spaces to the perivenous spaces. This convective flow within the brain parenchyma is believed to contribute to the clearance of waste products from the brain. Subsequently, the parenchymal waste present in the CSF is drained into the peripheral lymphatics through the perivenous spaces.</p> Full article ">
Open AccessReview
Recent Advances in Combating Bacterial Infections by Using Hybrid Nano-Systems
by
Unnati Patel and Emily C. Hunt
J. Nanotheranostics 2023, 4(3), 429-462; https://doi.org/10.3390/jnt4030019 - 8 Sep 2023
Cited by 4
Abstract
In recent years, antimicrobial resistance in many human pathogens has become a serious health concern. Since infections with resistant pathogens cannot be treated with traditional antimicrobial drugs, new strategies are necessary to fight bacterial infections. Hybrid nano-systems may provide a solution to this
[...] Read more.
In recent years, antimicrobial resistance in many human pathogens has become a serious health concern. Since infections with resistant pathogens cannot be treated with traditional antimicrobial drugs, new strategies are necessary to fight bacterial infections. Hybrid nano-systems may provide a solution to this problem, by combining multiple mechanisms for killing bacteria to synergistically increase the effectiveness of the antimicrobial treatment. In this review, we highlight recent advances in the development of hybrid nano-systems for the treatment of bacterial infections. We discuss the use of hybrid nano-systems for combinational therapy, focusing on various triggering mechanisms for drug release and the development of biomimetic nanomaterials. We also examine inherently antimicrobial nano-systems and their uses in preventing infections due to wounds and medical implants. This review summarizes recent advances and provides insight into the future development of antimicrobial treatments using hybrid nanomaterials.
Full article
(This article belongs to the Special Issue Advances and Innovations in Theranostic Nanobiomaterials)
►▼
Show Figures
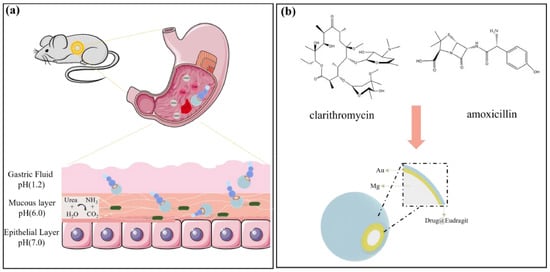
Figure 1
Figure 1
<p>(<b>a</b>) Schematic illustration of in vivo propulsion and drug delivery of Mg/Au/Eur micromotors in a mouse stomach. (<b>b</b>) Drug-loaded Mg/Au/Eur micromotor. Adapted with permission from Ref. [<a href="#B16-jnt-04-00019" class="html-bibr">16</a>]. Copyright 2020, Applied Materials Today.</p> Full article ">Figure 2
<p>(<b>a</b>) Synthetic route of Col@MSN@LL-(LL-37), final nanoassembly. (<b>b</b>) Hypothetical mechanism of the action of Col@MSN@LL-(LL-37) upon interaction with PA14. Adapted with permission from Ref. [<a href="#B31-jnt-04-00019" class="html-bibr">31</a>]. Copyright 2020, American Chemical Society.</p> Full article ">Figure 3
<p>(<b>a</b>) Schematic illustration of stepwise synthesis of the final nano-assembly: (<b>b</b>) Application of final nano-assembly: GNR@MSNP@BDQ@TSL@NZX for targeting and killing Msmeg using 808 nm NIR laser light. Adapted with permission from Ref. [<a href="#B93-jnt-04-00019" class="html-bibr">93</a>]. Copyright 2020 Nano select Wiley.</p> Full article ">Figure 4
<p>Scheme illustration of MLP18 nanoliposomes for diagnosis and eradiation of MDR bacterial infection. Taking advantage of the bacteria-specific maltodextrin transporter pathway, MLP18 after intravenous injection selectively navigates to the bacterial infection site (<b>a</b>). Afterward, the oversecreted PLA2 enzyme in bacterial infection microenvironment effectively degrades the lipid skeleton of MLP18, activating an efficient release and internalization of sonoensitizer into bacteria. Due to the excellent optical properties of P18, the bacteria show an obvious NIR FL/PA signal, changing from yellow to red (<b>b</b>). Upon ultrasound irradiation, cytotoxic ROS are generated and then mediate bacterial death (<b>c</b>). Adapted with permission from Ref. [<a href="#B70-jnt-04-00019" class="html-bibr">70</a>]. Copyright 2020, American Chemical Society.</p> Full article ">Figure 5
<p>Preparation of NIR light-controlled drug-release D-TiO<sub>2</sub>/Au@UCN nanocomposites and its mechanism of action. Adapted with permission from Ref. [<a href="#B74-jnt-04-00019" class="html-bibr">74</a>]. Copyright 2020, American Chemical Society.</p> Full article ">Figure 6
<p>Schematic illustration of the preparation and the POD-like activity of UsAuNPs/MOFs hybrid for antibacterial therapy. Adapted with permission from Ref. [<a href="#B166-jnt-04-00019" class="html-bibr">166</a>]. Copyright 2020 Small, Wiley.</p> Full article ">Scheme 1
<p>Schematic of hybrid nano-systems used to combat antimicrobial resistance.</p> Full article ">
<p>(<b>a</b>) Schematic illustration of in vivo propulsion and drug delivery of Mg/Au/Eur micromotors in a mouse stomach. (<b>b</b>) Drug-loaded Mg/Au/Eur micromotor. Adapted with permission from Ref. [<a href="#B16-jnt-04-00019" class="html-bibr">16</a>]. Copyright 2020, Applied Materials Today.</p> Full article ">Figure 2
<p>(<b>a</b>) Synthetic route of Col@MSN@LL-(LL-37), final nanoassembly. (<b>b</b>) Hypothetical mechanism of the action of Col@MSN@LL-(LL-37) upon interaction with PA14. Adapted with permission from Ref. [<a href="#B31-jnt-04-00019" class="html-bibr">31</a>]. Copyright 2020, American Chemical Society.</p> Full article ">Figure 3
<p>(<b>a</b>) Schematic illustration of stepwise synthesis of the final nano-assembly: (<b>b</b>) Application of final nano-assembly: GNR@MSNP@BDQ@TSL@NZX for targeting and killing Msmeg using 808 nm NIR laser light. Adapted with permission from Ref. [<a href="#B93-jnt-04-00019" class="html-bibr">93</a>]. Copyright 2020 Nano select Wiley.</p> Full article ">Figure 4
<p>Scheme illustration of MLP18 nanoliposomes for diagnosis and eradiation of MDR bacterial infection. Taking advantage of the bacteria-specific maltodextrin transporter pathway, MLP18 after intravenous injection selectively navigates to the bacterial infection site (<b>a</b>). Afterward, the oversecreted PLA2 enzyme in bacterial infection microenvironment effectively degrades the lipid skeleton of MLP18, activating an efficient release and internalization of sonoensitizer into bacteria. Due to the excellent optical properties of P18, the bacteria show an obvious NIR FL/PA signal, changing from yellow to red (<b>b</b>). Upon ultrasound irradiation, cytotoxic ROS are generated and then mediate bacterial death (<b>c</b>). Adapted with permission from Ref. [<a href="#B70-jnt-04-00019" class="html-bibr">70</a>]. Copyright 2020, American Chemical Society.</p> Full article ">Figure 5
<p>Preparation of NIR light-controlled drug-release D-TiO<sub>2</sub>/Au@UCN nanocomposites and its mechanism of action. Adapted with permission from Ref. [<a href="#B74-jnt-04-00019" class="html-bibr">74</a>]. Copyright 2020, American Chemical Society.</p> Full article ">Figure 6
<p>Schematic illustration of the preparation and the POD-like activity of UsAuNPs/MOFs hybrid for antibacterial therapy. Adapted with permission from Ref. [<a href="#B166-jnt-04-00019" class="html-bibr">166</a>]. Copyright 2020 Small, Wiley.</p> Full article ">Scheme 1
<p>Schematic of hybrid nano-systems used to combat antimicrobial resistance.</p> Full article ">
Open AccessReview
Aptamers as Theranostics in Cardiovascular Diseases
by
Manish Ramchandani, Priyanka Kumari and Amit K. Goyal
J. Nanotheranostics 2023, 4(3), 408-428; https://doi.org/10.3390/jnt4030018 - 6 Sep 2023
Cited by 1
Abstract
Cardiovascular disease (particularly atherosclerosis) is a leading cause of death around the world, and there still exists a need for improved diagnostic techniques and treatments to improve patient outcomes as well as minimize the disease’s global burden. Aptamers are short, single-stranded DNA or
[...] Read more.
Cardiovascular disease (particularly atherosclerosis) is a leading cause of death around the world, and there still exists a need for improved diagnostic techniques and treatments to improve patient outcomes as well as minimize the disease’s global burden. Aptamers are short, single-stranded DNA or RNA molecules that are accompanied by unique characteristics such as specificity, high binding affinity, ease of cellular internalization, and rapid tissue accumulation capabilities, offering great potential as theranostic agents in cardiovascular diseases with significantly improved sensitivity and accuracy. These theranostic agents provide a combination of therapy and diagnostics in which aptamers may diagnose and treat disease simultaneously. Therefore, this review article summarizes the role of aptamer-based probes for imaging and theranostics in cardiovascular disease. It also provides insight into current research and future treatment techniques that are very relevant for future clinical practice with the aim of improving the quality of life of cardiovascular disease patients.
Full article
(This article belongs to the Special Issue Interventional Nanotheranostics for Translational Nano-Immunotherapy)
►▼
Show Figures
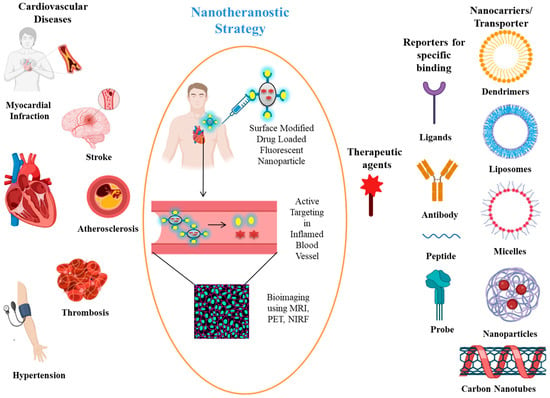
Figure 1
Open AccessReview
Smart Platinum Nanostructures: A Journey from Synthesis to Advanced Theranostic Applications
by
Akash Kumar, Nabojit Das and Raja Gopal Rayavarapu
J. Nanotheranostics 2023, 4(3), 384-407; https://doi.org/10.3390/jnt4030017 - 10 Aug 2023
Cited by 1
Abstract
A significant paradigm shift has been observed in the past decade in the area of theranostics owing to the development of various isotropic and anisotropic metal nanostructures, simultaneous with improved imaging modalities. Platinum-based nanostructures are advancing in a plethora of clinical applications as
[...] Read more.
A significant paradigm shift has been observed in the past decade in the area of theranostics owing to the development of various isotropic and anisotropic metal nanostructures, simultaneous with improved imaging modalities. Platinum-based nanostructures are advancing in a plethora of clinical applications as theranostics tools owing to their unique behavior concerning their size, shape, and surface chemistry at the nanoscale regime. Platinum nanostructures are optically active and provide significant potential to the field of theranostics by simplifying diagnosis and therapeutics, thus providing key solutions through nano-enabled technologies. The review emphasizes the potential of platinum nanostructures that have immense potential in vitro and in vivo scenarios as nanocarriers. Still, their potential in terms of photothermal active agents has not been well explored or reported. Nanotheranostics has emerged as a platform where various noble metal nanoparticles are effectively efficient as photothermal agents in bringing precision to therapy and diagnostics. Platinum, as an antioxidant and a stable nanocarrier, will enable them to act as photosensitizers when conjugated to affinity molecules and plays a key role in efficient treatment and diagnosis. The review envisions bringing together the possibilities of the safe-by-design synthesis of platinum nanostructures and their potential role in both in vitro and in vivo applications. A roadmap describing the challenges, pitfalls, and possibilities of influencing platinum nanostructures to overcome the existing biological/targeting barriers is elaborated. This review provides a literature survey on platinum nanostructures in theranostics, providing novel strategies in bio-imaging, diagnostics, and nanomedicine.
Full article
(This article belongs to the Special Issue Role of Nano-Bio Interaction for Nanotheranostic Applications)
►▼
Show Figures
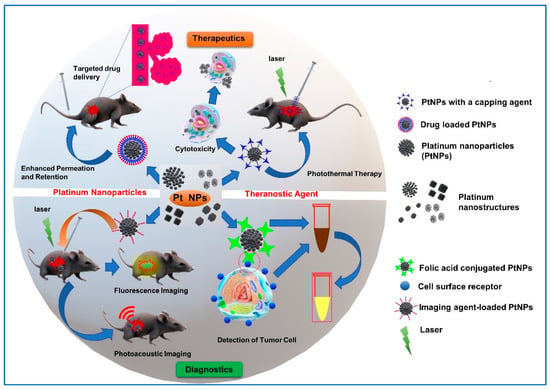
Figure 1
Figure 1
<p>Schematic representation of different platinum nanostructures providing a single platform for the simultaneous diagnosis and treatment of a particular disease.</p> Full article ">Figure 2
<p>Synthesis of platinum nanoparticles through various methods.</p> Full article ">Figure 3
<p>Safety assessment of citrate-stabilized platinum nanoparticles (70 nm) using a liver cell line (HepG2). (<b>a</b>,<b>b</b>) Cell viability assessment using an MTS assay and ROS evaluation using a DCFH-DA dye of PtNPs with different concentrations. (<b>c</b>–<b>f</b>) Assessment of different pro-inflammatory cytokines (IL-1β, IL-6, IL-8, and TNF-α) upon exposure to various concentrations of PtNPs. * denotes statistical significance, where <span class="html-italic">p</span> < 0.05. Adapted from ref. [<a href="#B17-jnt-04-00017" class="html-bibr">17</a>] with permission. Copyright © 2018 Hindawi.</p> Full article ">Figure 4
<p>Safety assessment of synthesized platinum nanoparticles in an in vivo model system: (<b>a</b>) synthesis procedure for obtaining less toxic PtNPs; (<b>b</b>) assessment of a common toxicity marker from serum after the termination of a tolerated dose (3 weeks); (<b>c</b>) histopathological analysis of different organs using H & E stain after the termination of the study; (<b>d</b>) change in mice weight after the injection of PtNP:DSPE-PEG micelles at different doses while phosphate-buffered saline acts as a control; (<b>e</b>) biodistribution analysis of PtNP:DSPE-PEG in BALB/c mice, 4T1 tumors-bearing BALB/c mice, and the control (BALB/c mice receiving saline). The PtNP:DSPE-PEG was injected at 10 mg Pt/kg b.wt, and tissues were collected after 24 h of injection. (<b>f</b>) Bioaccumulation of PtNP:DSPE-PEG in mice at concentrations ranging from 5 to 20 mg Pt/kg was studied using inductively coupled plasma mass spectroscopy (ICP-MS), and the platinum content was quantified in different organs. Adapted from ref. [<a href="#B114-jnt-04-00017" class="html-bibr">114</a>] with permission, MDPI 2018, Basel, Switzerland.</p> Full article ">Figure 5
<p>Diagnostic potential of designed platinum nanoparticles: (<b>a</b>) preparation steps for the synthesis of PEG-stabilized platinum nanoparticles over mesoporous silica nanoparticles, conjugated with imaging dye Dy800 (PEG-MSNs-Pt-Dy800); (<b>b</b>) optical image of a tumor-bearing mouse before (control) and after 24 h of exposure (sample, injected with PEG-MSNs-Pt-Dy800); (<b>c</b>) CT images of a tumor-bearing mouse (encircled in red) after 24 h of exposure (sample, injected with PEG-MSNs-Pt-Dy800). Adapted from ref. [<a href="#B117-jnt-04-00017" class="html-bibr">117</a>] with permission, MDPI 2019, Basel, Switzerland.</p> Full article ">Figure 6
<p>Therapeutic potential of platinum nanoparticles: (<b>a</b>) synthesis steps of doxorubicin-loaded Peglayted mesoporous platinum nanoparticles (PEG@PtNP/Dox); (<b>b</b>) confocal images of PEG@PtNP/Dox in MCF-7/ADR cells at 1 h, 12 h, and 24 h, respectively. z stack images with different cross-sections in XY, XZ, and YZ plane PEG@Pt/Dox-treated MCF-7/ADR cells for (<b>c</b>) 1 h, (<b>d</b>) 12 h, and (<b>e</b>) 24 h, respectively. Blue: DAPI (nucleus), Red: Doxorubicin. (<b>f</b>) TEM images of PEG@Pt/Dox-treated cells treated for 12 h (red arrows confirmed PEG@Pt/Dox’s presence in cells). (<b>g</b>) Flow cytometric analysis of PEG@Pt/Dox-treated MCF-7/ADR cells for 1 h, 12 h, and 24 h, respectively. (<b>h</b>) The corresponding cellular fluorescence intensity. The concentration of Doxorubicin is 5 μg/mL (cells without treatment were considered as the control). (<b>i</b>) Thermal images and (<b>j</b>) change in temperature of PEG@Pt/Dox solutions with different nanoparticle concentrations and irradiation intensities. (<b>k</b>) Change in temperature of PEG@Pt/Dox solution with fixed 20 μg/mL of PEG@Pt over five ON/OFF cycles of irradiation at 1.0 W/cm<sup>2</sup>. (<b>l</b>) Cell viability of the MCF-7/ADR cells line after exposure to free Doxorubicin, PEG@Pt/Dox + laser, and PEG@Pt/Dox after 24 h of incubation. Adapted from ref. [<a href="#B130-jnt-04-00017" class="html-bibr">130</a>] with permission. Copyright 2020, Elsevier.</p> Full article ">Figure 7
<p>Various challenges associated with platinum nanoparticles for their clinical use in cancer theranostics. The challenges include the level of synthesis, toxicity/safety, application, and regulation.</p> Full article ">
<p>Schematic representation of different platinum nanostructures providing a single platform for the simultaneous diagnosis and treatment of a particular disease.</p> Full article ">Figure 2
<p>Synthesis of platinum nanoparticles through various methods.</p> Full article ">Figure 3
<p>Safety assessment of citrate-stabilized platinum nanoparticles (70 nm) using a liver cell line (HepG2). (<b>a</b>,<b>b</b>) Cell viability assessment using an MTS assay and ROS evaluation using a DCFH-DA dye of PtNPs with different concentrations. (<b>c</b>–<b>f</b>) Assessment of different pro-inflammatory cytokines (IL-1β, IL-6, IL-8, and TNF-α) upon exposure to various concentrations of PtNPs. * denotes statistical significance, where <span class="html-italic">p</span> < 0.05. Adapted from ref. [<a href="#B17-jnt-04-00017" class="html-bibr">17</a>] with permission. Copyright © 2018 Hindawi.</p> Full article ">Figure 4
<p>Safety assessment of synthesized platinum nanoparticles in an in vivo model system: (<b>a</b>) synthesis procedure for obtaining less toxic PtNPs; (<b>b</b>) assessment of a common toxicity marker from serum after the termination of a tolerated dose (3 weeks); (<b>c</b>) histopathological analysis of different organs using H & E stain after the termination of the study; (<b>d</b>) change in mice weight after the injection of PtNP:DSPE-PEG micelles at different doses while phosphate-buffered saline acts as a control; (<b>e</b>) biodistribution analysis of PtNP:DSPE-PEG in BALB/c mice, 4T1 tumors-bearing BALB/c mice, and the control (BALB/c mice receiving saline). The PtNP:DSPE-PEG was injected at 10 mg Pt/kg b.wt, and tissues were collected after 24 h of injection. (<b>f</b>) Bioaccumulation of PtNP:DSPE-PEG in mice at concentrations ranging from 5 to 20 mg Pt/kg was studied using inductively coupled plasma mass spectroscopy (ICP-MS), and the platinum content was quantified in different organs. Adapted from ref. [<a href="#B114-jnt-04-00017" class="html-bibr">114</a>] with permission, MDPI 2018, Basel, Switzerland.</p> Full article ">Figure 5
<p>Diagnostic potential of designed platinum nanoparticles: (<b>a</b>) preparation steps for the synthesis of PEG-stabilized platinum nanoparticles over mesoporous silica nanoparticles, conjugated with imaging dye Dy800 (PEG-MSNs-Pt-Dy800); (<b>b</b>) optical image of a tumor-bearing mouse before (control) and after 24 h of exposure (sample, injected with PEG-MSNs-Pt-Dy800); (<b>c</b>) CT images of a tumor-bearing mouse (encircled in red) after 24 h of exposure (sample, injected with PEG-MSNs-Pt-Dy800). Adapted from ref. [<a href="#B117-jnt-04-00017" class="html-bibr">117</a>] with permission, MDPI 2019, Basel, Switzerland.</p> Full article ">Figure 6
<p>Therapeutic potential of platinum nanoparticles: (<b>a</b>) synthesis steps of doxorubicin-loaded Peglayted mesoporous platinum nanoparticles (PEG@PtNP/Dox); (<b>b</b>) confocal images of PEG@PtNP/Dox in MCF-7/ADR cells at 1 h, 12 h, and 24 h, respectively. z stack images with different cross-sections in XY, XZ, and YZ plane PEG@Pt/Dox-treated MCF-7/ADR cells for (<b>c</b>) 1 h, (<b>d</b>) 12 h, and (<b>e</b>) 24 h, respectively. Blue: DAPI (nucleus), Red: Doxorubicin. (<b>f</b>) TEM images of PEG@Pt/Dox-treated cells treated for 12 h (red arrows confirmed PEG@Pt/Dox’s presence in cells). (<b>g</b>) Flow cytometric analysis of PEG@Pt/Dox-treated MCF-7/ADR cells for 1 h, 12 h, and 24 h, respectively. (<b>h</b>) The corresponding cellular fluorescence intensity. The concentration of Doxorubicin is 5 μg/mL (cells without treatment were considered as the control). (<b>i</b>) Thermal images and (<b>j</b>) change in temperature of PEG@Pt/Dox solutions with different nanoparticle concentrations and irradiation intensities. (<b>k</b>) Change in temperature of PEG@Pt/Dox solution with fixed 20 μg/mL of PEG@Pt over five ON/OFF cycles of irradiation at 1.0 W/cm<sup>2</sup>. (<b>l</b>) Cell viability of the MCF-7/ADR cells line after exposure to free Doxorubicin, PEG@Pt/Dox + laser, and PEG@Pt/Dox after 24 h of incubation. Adapted from ref. [<a href="#B130-jnt-04-00017" class="html-bibr">130</a>] with permission. Copyright 2020, Elsevier.</p> Full article ">Figure 7
<p>Various challenges associated with platinum nanoparticles for their clinical use in cancer theranostics. The challenges include the level of synthesis, toxicity/safety, application, and regulation.</p> Full article ">
Open AccessReview
Nanotheranostics: Platforms, Current Applications, and Mechanisms of Targeting in Breast and Prostate Cancers
by
Paul A. Akpa, Ikechukwu E. Peter, Akachukwu M. Onwuka, Bonaventure C. Obi, Maureen O. Akunne, Chukwuemeka S. Nworu, Paul M. Ejikeme, Theophine C. Akunne, Anthony A. Attama and Peter A. Akah
J. Nanotheranostics 2023, 4(3), 346-383; https://doi.org/10.3390/jnt4030016 - 10 Aug 2023
Cited by 1
Abstract
►▼
Show Figures
Globally, cancer is one of the deadliest diseases, needing a meticulous diagnosis and targeted treatment plan to achieve an initial prognosis, followed by precision and optimization in treatment. Nonselective targeting, difficulty in accurately monitoring treatment end-results, serious drug side-effects, and severity of disease
[...] Read more.
Globally, cancer is one of the deadliest diseases, needing a meticulous diagnosis and targeted treatment plan to achieve an initial prognosis, followed by precision and optimization in treatment. Nonselective targeting, difficulty in accurately monitoring treatment end-results, serious drug side-effects, and severity of disease resulting in metastasis are the key flaws of traditional techniques. Nanotechnology and nanoparticles possess special features to completely transform the field of diagnosis and treatment of cancer. A holistic strategy that employs a dual function of diagnosis and therapy while utilizing a nanocarrier is referred to as a nanotheranostic. The nanotheranostic framework was created to surmount a variety of biological and physiological obstacles, effectively delivering the cargo to the intended target location, while simultaneously facilitating therapeutic intervention, surveillance, and validation to demonstrate improved treatment effectiveness. As a result, a nanotheranostic platform can be useful for targeted drug delivery, release, and distribution assessment, in addition to patient classification and survival. Nanotheranostic techniques also lead to reduced drug side-effects compared with conventional therapies. In this review, we outline current studies on nanotheranostics and their advantages over conventional treatment strategies, the applications and challenges/limitations of nanotheranostics, and the mechanisms of targeting in breast and prostate cancers.
Full article
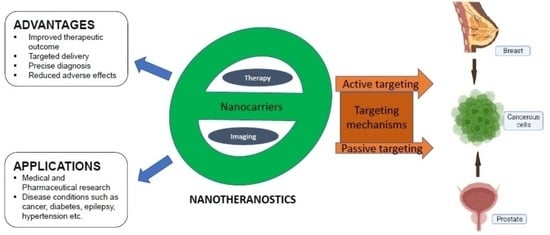
Graphical abstract
Graphical abstract
Full article ">Figure 1
<p>Schematic representation of the composition of a nanotheranostic system.</p> Full article ">Figure 2
<p>Schematic representation of some nanocarriers employed for theranostic purposes in breast and prostate cancer. Adapted with permission from Ref. [<a href="#B43-jnt-04-00016" class="html-bibr">43</a>]. Copyright 2021, Springer.</p> Full article ">Figure 3
<p>Schematic representation of passive targeting. Passive targeting uses the tumor vascular system to enhance the permeability and retention effect (EPR), which passively accumulates at the pathological site and allows the drug to be released near the tumor. Adapted from the Ref. [<a href="#B126-jnt-04-00016" class="html-bibr">126</a>]. Copyright 2023, BioMed Central Ltd.</p> Full article ">Figure 4
<p>Schematic representation of active targeting. Active targeting is achieved by functionalizing the surface of drug-containing nanocarriers, whose targeting component provides selective recognition of different receptors or antigens overexpressed in breast and prostate cancer cells, improving their therapeutic efficacy and overcoming multidrug resistance. Adapted with permission from Ref. [<a href="#B135-jnt-04-00016" class="html-bibr">135</a>]. Copyright 2022, Chinese Anti-Cancer Association (CACA).</p> Full article ">
Full article ">Figure 1
<p>Schematic representation of the composition of a nanotheranostic system.</p> Full article ">Figure 2
<p>Schematic representation of some nanocarriers employed for theranostic purposes in breast and prostate cancer. Adapted with permission from Ref. [<a href="#B43-jnt-04-00016" class="html-bibr">43</a>]. Copyright 2021, Springer.</p> Full article ">Figure 3
<p>Schematic representation of passive targeting. Passive targeting uses the tumor vascular system to enhance the permeability and retention effect (EPR), which passively accumulates at the pathological site and allows the drug to be released near the tumor. Adapted from the Ref. [<a href="#B126-jnt-04-00016" class="html-bibr">126</a>]. Copyright 2023, BioMed Central Ltd.</p> Full article ">Figure 4
<p>Schematic representation of active targeting. Active targeting is achieved by functionalizing the surface of drug-containing nanocarriers, whose targeting component provides selective recognition of different receptors or antigens overexpressed in breast and prostate cancer cells, improving their therapeutic efficacy and overcoming multidrug resistance. Adapted with permission from Ref. [<a href="#B135-jnt-04-00016" class="html-bibr">135</a>]. Copyright 2022, Chinese Anti-Cancer Association (CACA).</p> Full article ">
Open AccessReview
An Overview of Nanotherapeutic Drug Delivery Options for the Management of Glioblastoma
by
William H. Pentz, Vincenzo J. Pizzuti, Matthew E. Halbert, Tritan J. Plute, Paul R. Lockman and Samuel A. Sprowls
J. Nanotheranostics 2023, 4(3), 323-345; https://doi.org/10.3390/jnt4030015 - 1 Aug 2023
Cited by 1
Abstract
Glioblastoma is the most common primary, malignant brain tumor that remains uniformly lethal in nearly all cases as a result of extreme cellular heterogeneity, treatment resistance, and recurrence. A major hurdle in therapeutic delivery to brain tumors is the blood–brain barrier (BBB), which
[...] Read more.
Glioblastoma is the most common primary, malignant brain tumor that remains uniformly lethal in nearly all cases as a result of extreme cellular heterogeneity, treatment resistance, and recurrence. A major hurdle in therapeutic delivery to brain tumors is the blood–brain barrier (BBB), which is the tightly regulated vascular barrier between the brain parenchyma and systemic circulation that prevents distribution of otherwise beneficial chemotherapeutics to central nervous system tumors. To overcome the obstacle of drug delivery beyond the BBB, nanoparticle formulations have come to the forefront, having demonstrated success in preclinical observations, but have not translated well into the clinical setting. In summary, this review article discusses brain tumors and challenges for drug delivery caused by the BBB, explores the benefits of nanoparticle formulations for brain tumor delivery, describes the characteristics these formulations possess that make them attractive therapeutic strategies, and provides preclinical examples that implement nanoparticles within glioma treatment regimens. Additionally, we explore the pitfalls associated with clinical translation and conclude with remarks geared toward overcoming these issues.
Full article
(This article belongs to the Special Issue Emerging Strategies in Nanomedicine)
►▼
Show Figures
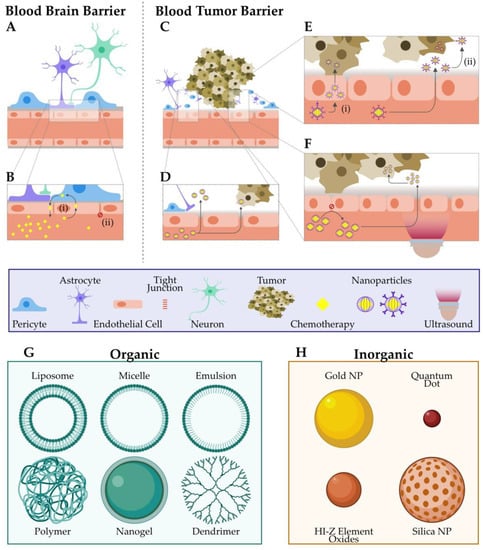
Figure 1
Figure 1
<p><b>Nanoparticle formulations improve drug delivery beyond the blood–tumor barrier.</b> The BBB (<b>A</b>) is a multi-cellular, physio-chemical barrier preventing the distribution of most conventional chemotherapeutics (<b>B</b>) to the brain through both active efflux (<b>Bi</b>) and passive restriction imposed the contiguous tight junction proteins between endothelial cells (<b>Bii</b>). During tumor development, tumor cells disrupt the BBB creating fenestrated, leaky tumor-associated vasculature termed the BTB (<b>C</b>). The disrupted vasculature allows for the passive accumulation of nanoparticles driven through the enhanced permeability and retention effect (<b>D</b>). Nanoparticle surface modifications can allow for the targeting of cell surface receptors on either the endothelium (<b>Ei</b>) or tumor cells themselves (<b>Eii</b>) allowing for receptor-mediated transcytosis across the vascular endothelium and/or tumor-specific targeting. There is a multitude of physically disruptive methods to further increase nanoparticle drug delivery, the most popular of late being transcranial low intensity focused ultrasound (<b>F</b>). (<b>G</b>,<b>H</b>) Illustrations of commonly formulated organic and inorganic nanoparticles seen in translational treatments.</p> Full article ">
<p><b>Nanoparticle formulations improve drug delivery beyond the blood–tumor barrier.</b> The BBB (<b>A</b>) is a multi-cellular, physio-chemical barrier preventing the distribution of most conventional chemotherapeutics (<b>B</b>) to the brain through both active efflux (<b>Bi</b>) and passive restriction imposed the contiguous tight junction proteins between endothelial cells (<b>Bii</b>). During tumor development, tumor cells disrupt the BBB creating fenestrated, leaky tumor-associated vasculature termed the BTB (<b>C</b>). The disrupted vasculature allows for the passive accumulation of nanoparticles driven through the enhanced permeability and retention effect (<b>D</b>). Nanoparticle surface modifications can allow for the targeting of cell surface receptors on either the endothelium (<b>Ei</b>) or tumor cells themselves (<b>Eii</b>) allowing for receptor-mediated transcytosis across the vascular endothelium and/or tumor-specific targeting. There is a multitude of physically disruptive methods to further increase nanoparticle drug delivery, the most popular of late being transcranial low intensity focused ultrasound (<b>F</b>). (<b>G</b>,<b>H</b>) Illustrations of commonly formulated organic and inorganic nanoparticles seen in translational treatments.</p> Full article ">
Open AccessReview
Recent Advancement of Bio-Inspired Nanoparticles in Cancer Theragnostic
by
Divya Tripathi, Kasturee Hajra and Dipak Maity
J. Nanotheranostics 2023, 4(3), 299-322; https://doi.org/10.3390/jnt4030014 - 24 Jul 2023
Cited by 4
Abstract
The introduction of cancer therapeutics and nanotechnology has resulted in a paradigm shift from conventional therapy to precision medicine. Nanotechnology, an interdisciplinary field with a focus on biomedical applications, holds immense promise in bringing about novel approaches for cancer detection, diagnosis, and therapy.
[...] Read more.
The introduction of cancer therapeutics and nanotechnology has resulted in a paradigm shift from conventional therapy to precision medicine. Nanotechnology, an interdisciplinary field with a focus on biomedical applications, holds immense promise in bringing about novel approaches for cancer detection, diagnosis, and therapy. The past decade has witnessed significant research and material applications related to nanoparticles (NPs). NPs differ from small-molecule drugs as they possess unique physicochemical characteristics, such as a large surface-to-volume ratio, enabling them to penetrate live cells efficiently. Traditional cancer therapies, such as chemotherapy, radiation therapy, targeted therapy, and immunotherapy, have limitations, such as cytotoxicity, lack of specificity, and multiple drug resistance, which pose significant challenges for effective cancer treatment. However, nanomaterials have unique properties that enable new therapeutic modalities beyond conventional drug delivery in the fight against cancer. Moreover, nanoparticles (1–100 nm) have numerous benefits, such as biocompatibility, reduced toxicity, excellent stability, enhanced permeability and retention effect, and precise targeting, making them ideal for cancer treatment. The purpose of this article is to provide consolidated information on various bio-inspired nanoparticles that aid in cancer theranostics.
Full article
(This article belongs to the Special Issue Advanced Functional and Tunable Nano-Systems for High-Performance Theranostic and Tissue Engineering Applications)
►▼
Show Figures
Figure 1
Figure 1
<p>The NPs’ routes of administration in the body to treat cancer along with their advantages and disadvantages. Adapted from reference [<a href="#B36-jnt-04-00014" class="html-bibr">36</a>], source is open access.</p> Full article ">Figure 2
<p>These are some of the nanoparticles which are being used in cancer theranostics.</p> Full article ">Figure 3
<p>Radioisotope biodistribution on healthy C57BL/6 mice.</p> Full article ">Figure 4
<p>The figure shows the formation of polymeric micelles used in cancer therapy.</p> Full article ">Figure 5
<p>Schematic diagram of a magnetic Nano capsule which encapsulates the therapeutic agent.</p> Full article ">
<p>The NPs’ routes of administration in the body to treat cancer along with their advantages and disadvantages. Adapted from reference [<a href="#B36-jnt-04-00014" class="html-bibr">36</a>], source is open access.</p> Full article ">Figure 2
<p>These are some of the nanoparticles which are being used in cancer theranostics.</p> Full article ">Figure 3
<p>Radioisotope biodistribution on healthy C57BL/6 mice.</p> Full article ">Figure 4
<p>The figure shows the formation of polymeric micelles used in cancer therapy.</p> Full article ">Figure 5
<p>Schematic diagram of a magnetic Nano capsule which encapsulates the therapeutic agent.</p> Full article ">
Open AccessReview
Polymeric Theragnostic Nanoplatforms for Bone Tissue Engineering
by
Kaushita Banerjee and Harishkumar Madhyastha
J. Nanotheranostics 2023, 4(3), 280-298; https://doi.org/10.3390/jnt4030013 - 20 Jul 2023
Abstract
Nanomaterial-based tissue engineering strategies are precisely designed and tweaked to contest specific patient needs and their end applications. Though theragnostic is a radical term very eminent in cancer prognosis, of late, theragnostic approaches have been explored in the fields of tissue remodulation and
[...] Read more.
Nanomaterial-based tissue engineering strategies are precisely designed and tweaked to contest specific patient needs and their end applications. Though theragnostic is a radical term very eminent in cancer prognosis, of late, theragnostic approaches have been explored in the fields of tissue remodulation and reparation. The engineering of theragnostic nanomaterials has opened up avenues for disease diagnosis, imaging, and therapeutic treatments. The instantaneous monitoring of therapeutic strategy is expected to co-deliver imaging and pharmaceutical agents at the same time, and nanoscale carrier moieties are convenient and efficient platforms in theragnostic applications, especially in soft and hard tissue regeneration. Furthermore, imaging modalities have extensively contributed to the signal-to-noise ratio. Simultaneously, there is an accumulation of high concentrations of therapeutic mediators at the defect site. Given the confines of contemporary bone diagnostic systems, the clinical rationale demands nano/biomaterials that can localize to bone-diseased sites to enhance the precision and prognostic value for osteoporosis, non-healing fractures, and/or infections, etc. Furthermore, bone theragnostics may have an even greater clinical impact and multimodal imaging procedures can overcome the restrictions of individual modalities. The present review introduces representative theragnostic polymeric nanomaterials and their advantages and disadvantages in practical use as well as their unique properties.
Full article
(This article belongs to the Topic Applications of Polymers and Polymer Nanomaterials in Drug Delivery and Nanomedicine)
►▼
Show Figures
Figure 1
Figure 1
<p>Ordered classification of bone tissue over their magnitude and occurrence ranging from calcified outer compact layer to resident cells coated with cell membrane receptors and precise nanoarchitecture of the surrounding extracellular matrix. Reproduced with permission from Reference [<a href="#B22-jnt-04-00013" class="html-bibr">22</a>] © 2005 American Society for the Advancement of Science.</p> Full article ">Figure 2
<p>Nanoplatforms loaded with targeting and therapeutic moieties that serve as contrast agents for several imaging techniques to accomplish theragnostic objectives. AuNP: gold nanoparticle; CT: micro-computed tomography; MRI: magnetic resonance imaging [<a href="#B26-jnt-04-00013" class="html-bibr">26</a>]. Reproduced from open access source.</p> Full article ">Figure 3
<p>Comprehensive design of the contemporary research advances and the future expectations in biomaterials with numerous functions of bone therapeutics and regeneration [<a href="#B93-jnt-04-00013" class="html-bibr">93</a>]. Reproduced from an open access source.</p> Full article ">
<p>Ordered classification of bone tissue over their magnitude and occurrence ranging from calcified outer compact layer to resident cells coated with cell membrane receptors and precise nanoarchitecture of the surrounding extracellular matrix. Reproduced with permission from Reference [<a href="#B22-jnt-04-00013" class="html-bibr">22</a>] © 2005 American Society for the Advancement of Science.</p> Full article ">Figure 2
<p>Nanoplatforms loaded with targeting and therapeutic moieties that serve as contrast agents for several imaging techniques to accomplish theragnostic objectives. AuNP: gold nanoparticle; CT: micro-computed tomography; MRI: magnetic resonance imaging [<a href="#B26-jnt-04-00013" class="html-bibr">26</a>]. Reproduced from open access source.</p> Full article ">Figure 3
<p>Comprehensive design of the contemporary research advances and the future expectations in biomaterials with numerous functions of bone therapeutics and regeneration [<a href="#B93-jnt-04-00013" class="html-bibr">93</a>]. Reproduced from an open access source.</p> Full article ">
Open AccessReview
Emerging Trends in the Application of Green Synthesized Biocompatible ZnO Nanoparticles for Translational Paradigm in Cancer Therapy
by
Shaikh Sheeran Naser, Basab Ghosh, Faizan Zarreen Simnani, Dibyangshee Singh, Anmol Choudhury, Aditya Nandi, Adrija Sinha, Ealisha Jha, Pritam Kumar Panda, Mrutyunjay Suar and Suresh K. Verma
J. Nanotheranostics 2023, 4(3), 248-279; https://doi.org/10.3390/jnt4030012 - 1 Jul 2023
Cited by 19
Abstract
Zinc oxide nanomaterials have been the cynosure of this decade because of their immense potential in different biomedical applications. It includes their usage in the prognosis and treatment of different infectious and cellular diseases, owing to their peculiar physiochemical properties such as variable
[...] Read more.
Zinc oxide nanomaterials have been the cynosure of this decade because of their immense potential in different biomedical applications. It includes their usage in the prognosis and treatment of different infectious and cellular diseases, owing to their peculiar physiochemical properties such as variable shape, size, and surface charge etc. Increasing demand and usage of the ZnO nanomaterials raise concerns about their cellular and molecular toxicity and their biocompatibility with human cells. This review comprehensively details their physiochemical properties for usage in biomedical applications. Furthermore, the toxicological concerns of ZnO nanomaterials with different types of cellular systems have been reviewed. Moreover, the biomedical and biocompatible efficacy of ZnO nanomaterials for cancer specific pathways has been discussed. This review offers insights into the current scenario of ZnO nanomaterials usage and signifies their potential future extension usage on different types of biomedical and environmental applications.
Full article
(This article belongs to the Special Issue Role of Nano-Bio Interaction for Nanotheranostic Applications)
►▼
Show Figures
Graphical abstract
Graphical abstract
Full article ">Figure 1
<p>Schematic diagram explaining the various synthesis methods of ZnO NPs.</p> Full article ">Figure 2
<p>Schematic diagram showing the toxicological concern of Zinc oxide NPs. (<b>a</b>) Toxicology of Zinc oxide nanoparticles is influenced by a number of critical factors, including particle size, solubility, exposure routes, structures, and others. Some of the damaging processes that depict the toxic impact include oxidative stress, non-homeostasis, genotoxicity, and coordination problems [<a href="#B126-jnt-04-00012" class="html-bibr">126</a>]. (<b>b</b>) zinc oxide nanoparticles that cause lung damage [<a href="#B102-jnt-04-00012" class="html-bibr">102</a>] (<b>c</b>) Influence of ZnO nanoparticles size on toxicities to the developing fetus and placenta (reprinted form reference [<a href="#B127-jnt-04-00012" class="html-bibr">127</a>], copyright Elsevier 2019) (<b>d</b>) ZnO nanoparticles that have pain-relieving properties. Glutamate release is decreased by ZnO nanoparticles. They are not competing NMDA glutamate receptor inhibitors, so they reduce the capacity of glutamate to have an impact on these receptors [<a href="#B128-jnt-04-00012" class="html-bibr">128</a>] (<b>e</b>) Skin toxicity is brought on by ZnONPs and UVB, which also inhibits PT [<a href="#B129-jnt-04-00012" class="html-bibr">129</a>].</p> Full article ">Figure 3
<p>Pathogenesis of cancer (<b>a</b>) EGFR pathway (<b>b</b>) PI3K/AKT/mTOR Pathway.</p> Full article ">Figure 4
<p>Pathogenesis of cancer (<b>a</b>) TGFβ (<b>b</b>) WNT β Catenin Pathway.</p> Full article ">Figure 5
<p>ZnO nanorod for doxorubicin delivery that is focused. Reprinted from reference [<a href="#B157-jnt-04-00012" class="html-bibr">157</a>], copyright Royal Society of Chemistry 2012.</p> Full article ">Figure 6
<p><b>(a</b>) Aqueous solution of ZnO@polymer, reprinted from reference [<a href="#B158-jnt-04-00012" class="html-bibr">158</a>], copyright Wiley-VCH 2013; (<b>b</b>) Synthesis of ZnO@MSNsDOX and Effective Methodology for pH-Triggered Release of the Anticancer Drug (DOX) from ZnO@MSNs DOX to the Cytosol via Selective Dissolution of ZnO QDs in the Acidic Intracellular Compartments of Cancer Cell, reprinted from reference [<a href="#B156-jnt-04-00012" class="html-bibr">156</a>] copyright American Chemical Society 2011.</p> Full article ">
Full article ">Figure 1
<p>Schematic diagram explaining the various synthesis methods of ZnO NPs.</p> Full article ">Figure 2
<p>Schematic diagram showing the toxicological concern of Zinc oxide NPs. (<b>a</b>) Toxicology of Zinc oxide nanoparticles is influenced by a number of critical factors, including particle size, solubility, exposure routes, structures, and others. Some of the damaging processes that depict the toxic impact include oxidative stress, non-homeostasis, genotoxicity, and coordination problems [<a href="#B126-jnt-04-00012" class="html-bibr">126</a>]. (<b>b</b>) zinc oxide nanoparticles that cause lung damage [<a href="#B102-jnt-04-00012" class="html-bibr">102</a>] (<b>c</b>) Influence of ZnO nanoparticles size on toxicities to the developing fetus and placenta (reprinted form reference [<a href="#B127-jnt-04-00012" class="html-bibr">127</a>], copyright Elsevier 2019) (<b>d</b>) ZnO nanoparticles that have pain-relieving properties. Glutamate release is decreased by ZnO nanoparticles. They are not competing NMDA glutamate receptor inhibitors, so they reduce the capacity of glutamate to have an impact on these receptors [<a href="#B128-jnt-04-00012" class="html-bibr">128</a>] (<b>e</b>) Skin toxicity is brought on by ZnONPs and UVB, which also inhibits PT [<a href="#B129-jnt-04-00012" class="html-bibr">129</a>].</p> Full article ">Figure 3
<p>Pathogenesis of cancer (<b>a</b>) EGFR pathway (<b>b</b>) PI3K/AKT/mTOR Pathway.</p> Full article ">Figure 4
<p>Pathogenesis of cancer (<b>a</b>) TGFβ (<b>b</b>) WNT β Catenin Pathway.</p> Full article ">Figure 5
<p>ZnO nanorod for doxorubicin delivery that is focused. Reprinted from reference [<a href="#B157-jnt-04-00012" class="html-bibr">157</a>], copyright Royal Society of Chemistry 2012.</p> Full article ">Figure 6
<p><b>(a</b>) Aqueous solution of ZnO@polymer, reprinted from reference [<a href="#B158-jnt-04-00012" class="html-bibr">158</a>], copyright Wiley-VCH 2013; (<b>b</b>) Synthesis of ZnO@MSNsDOX and Effective Methodology for pH-Triggered Release of the Anticancer Drug (DOX) from ZnO@MSNs DOX to the Cytosol via Selective Dissolution of ZnO QDs in the Acidic Intracellular Compartments of Cancer Cell, reprinted from reference [<a href="#B156-jnt-04-00012" class="html-bibr">156</a>] copyright American Chemical Society 2011.</p> Full article ">
Open AccessFeature PaperArticle
Convolutional Neural Network Classification of Exhaled Aerosol Images for Diagnosis of Obstructive Respiratory Diseases
by
Mohamed Talaat, Jensen Xi, Kaiyuan Tan, Xiuhua April Si and Jinxiang Xi
J. Nanotheranostics 2023, 4(3), 228-247; https://doi.org/10.3390/jnt4030011 - 26 Jun 2023
Cited by 6
Abstract
Aerosols exhaled from the lungs have distinctive patterns that can be linked to the abnormalities of the lungs. Yet, due to their intricate nature, it is highly challenging to analyze and distinguish these aerosol patterns. Small airway diseases pose an even greater challenge,
[...] Read more.
Aerosols exhaled from the lungs have distinctive patterns that can be linked to the abnormalities of the lungs. Yet, due to their intricate nature, it is highly challenging to analyze and distinguish these aerosol patterns. Small airway diseases pose an even greater challenge, as the disturbance signals tend to be weak. The objective of this study was to evaluate the performance of four convolutional neural network (CNN) models (AlexNet, ResNet-50, MobileNet, and EfficientNet) in detecting and staging airway abnormalities in small airways using exhaled aerosol images. Specifically, the model’s capacity to classify images inside and outside the original design space was assessed. In doing so, multi-level testing on images with decreasing similarities was conducted for each model. A total of 2745 images were generated using physiology-based simulations from normal and obstructed lungs of varying stages. Multiple-round training on datasets with increasing images (and new features) was also conducted to evaluate the benefits of continuous learning. Results show reasonably high classification accuracy on inbox images for models but significantly lower accuracy on outbox images (i.e., outside design space). ResNet-50 was the most robust among the four models for both diagnostic (2-class: normal vs. disease) and staging (3-class) purposes, as well as on both inbox and outbox test datasets. Variation in flow rate was observed to play a more important role in classification decisions than particle size and throat variation. Continuous learning/training with appropriate images could substantially enhance classification accuracy, even with a small number (~100) of new images. This study shows that CNN transfer-learning models could detect small airway remodeling (<1 mm) amidst a variety of variants and that ResNet-50 can be a promising model for the future development of obstructive lung diagnostic systems.
Full article
(This article belongs to the Special Issue Emerging Strategies in Nanomedicine)
►▼
Show Figures
Graphical abstract
Graphical abstract
Full article ">Figure 1
<p>Computational mouth-lung models: (<b>a</b>) normal and diseased airway geometries with varying levels of bronchiolar constrictions in the left lower lung (i.e., D0, D1, and D2), as well as varying throat openings (i.e., Th0, Th1, Th2, and Th3); (<b>b</b>) a computational mesh with a fine, body-fitted prismatic mesh.</p> Full article ">Figure 2
<p>Dataset architectures: (<b>a</b>) Diagram for data source from a systemic variation in the particle size (dp), respiration flow rate (Q), and throat opening (Th); (<b>b</b>) Baseline dataset (Base, 1080 images) with dp ranging 0.5–10 µm, Q ranging 10–19 L/min, and a normal throat opening (Th0), i.e., breath test design space; (<b>c</b>) Inbox dataset with both Q and dp falling within the design space, but either Q or dp differing from the baseline (i.e., Inbox_Q and Inbox_dp); (<b>d</b>) Outbox dataset with at least one of the three factors (i.e., dp, Q, Th) falling out of the design space, including outbox_Q, Outbox_Q_dp, Outbox_Th, and Outbox_Q_dp_Th. Explanations of the shape and colors of symbols were provided in text (see <a href="#sec2dot3-jnt-04-00011" class="html-sec">Section 2.3</a>, 1st, 2nd, and 3rd paragraphs).</p> Full article ">Figure 3
<p>Comparison of exhaled aerosol distributions between normal (<b>a</b>) and diseased lungs with mild (D1) and severe (D2) constrictions (<b>b</b>,<b>c</b>). Effects from particle size, flow rate, and throat constriction were considered.</p> Full article ">Figure 4
<p>Comparison of exhaled aerosols released only from the disease-afflicted bronchioles during exhalation between normal lung (<b>a</b>) and (<b>b</b>) diseased lung D1, as well as (<b>c</b>) exhalation flows in disease-afflicted bronchioles. The black, green, and red color represents 0.5, 1.0, and 5 µm, respectively.</p> Full article ">Figure 5
<p>Performance comparison on three test datasets with decreasing levels of similarity (Level 1, Inbox, and Outbox) in terms of accuracy, sensitivity, and specificity in Round 1 testing: (<b>a</b>) 2-class classification (normal vs. disease); (<b>b</b>) 3-class classification (D0 vs. D1 vs. D2).</p> Full article ">Figure 6
<p>Performance comparison in 3-class classification among four network models in Round 1 testing: (<b>a</b>) Model accuracy, sensitivity, and specificity; (<b>b</b>) ROC (receiver operating characteristic) profiles.</p> Full article ">Figure 7
<p>Classification accuracy on three test datasets (Level 1, Inbox, and Outbox) in terms of 2-class (normal vs. disease) and 3-class (D0 vs. D1 vs. D2) classifications in different rounds: (<b>a</b>) Round 2; (<b>b</b>) Round 3, 25% Outbox; (<b>c</b>) Round 3, 50% Outbox.</p> Full article ">Figure 8
<p>Receiver operating characteristics (ROC) based on the Outbox test dataset: (<b>a</b>) Round 2 (R2); (<b>b</b>) Round 3, 25% Outbox (R3-25%).</p> Full article ">Figure 9
<p>Comparison of ResNet-50 classification performance on the Outbox test dataset in different rounds: (<b>a</b>) 2-class; (<b>b</b>) 3-class.</p> Full article ">Figure 10
<p>Performance of trained models on the new test dataset (Inbox_dp and Outbox_Q_dp, <a href="#jnt-04-00011-f002" class="html-fig">Figure 2</a>a): (<b>a</b>) Inbox_dp vs. Outbox_Q_dp in 2-class and 3-class classifications; (<b>b</b>) Model effects in 2-class and 3-class classifications; (<b>c</b>) ROC in Rounds 1, 2, and 3.</p> Full article ">Figure 11
<p>Image analysis: (<b>a</b>) Heat map; (<b>b</b>) Features from the 2nd ReLU layer. The true class of the image was D2, with EfficientNet misclassifying it as D1 and the other three models classifying it correctly.</p> Full article ">
Full article ">Figure 1
<p>Computational mouth-lung models: (<b>a</b>) normal and diseased airway geometries with varying levels of bronchiolar constrictions in the left lower lung (i.e., D0, D1, and D2), as well as varying throat openings (i.e., Th0, Th1, Th2, and Th3); (<b>b</b>) a computational mesh with a fine, body-fitted prismatic mesh.</p> Full article ">Figure 2
<p>Dataset architectures: (<b>a</b>) Diagram for data source from a systemic variation in the particle size (dp), respiration flow rate (Q), and throat opening (Th); (<b>b</b>) Baseline dataset (Base, 1080 images) with dp ranging 0.5–10 µm, Q ranging 10–19 L/min, and a normal throat opening (Th0), i.e., breath test design space; (<b>c</b>) Inbox dataset with both Q and dp falling within the design space, but either Q or dp differing from the baseline (i.e., Inbox_Q and Inbox_dp); (<b>d</b>) Outbox dataset with at least one of the three factors (i.e., dp, Q, Th) falling out of the design space, including outbox_Q, Outbox_Q_dp, Outbox_Th, and Outbox_Q_dp_Th. Explanations of the shape and colors of symbols were provided in text (see <a href="#sec2dot3-jnt-04-00011" class="html-sec">Section 2.3</a>, 1st, 2nd, and 3rd paragraphs).</p> Full article ">Figure 3
<p>Comparison of exhaled aerosol distributions between normal (<b>a</b>) and diseased lungs with mild (D1) and severe (D2) constrictions (<b>b</b>,<b>c</b>). Effects from particle size, flow rate, and throat constriction were considered.</p> Full article ">Figure 4
<p>Comparison of exhaled aerosols released only from the disease-afflicted bronchioles during exhalation between normal lung (<b>a</b>) and (<b>b</b>) diseased lung D1, as well as (<b>c</b>) exhalation flows in disease-afflicted bronchioles. The black, green, and red color represents 0.5, 1.0, and 5 µm, respectively.</p> Full article ">Figure 5
<p>Performance comparison on three test datasets with decreasing levels of similarity (Level 1, Inbox, and Outbox) in terms of accuracy, sensitivity, and specificity in Round 1 testing: (<b>a</b>) 2-class classification (normal vs. disease); (<b>b</b>) 3-class classification (D0 vs. D1 vs. D2).</p> Full article ">Figure 6
<p>Performance comparison in 3-class classification among four network models in Round 1 testing: (<b>a</b>) Model accuracy, sensitivity, and specificity; (<b>b</b>) ROC (receiver operating characteristic) profiles.</p> Full article ">Figure 7
<p>Classification accuracy on three test datasets (Level 1, Inbox, and Outbox) in terms of 2-class (normal vs. disease) and 3-class (D0 vs. D1 vs. D2) classifications in different rounds: (<b>a</b>) Round 2; (<b>b</b>) Round 3, 25% Outbox; (<b>c</b>) Round 3, 50% Outbox.</p> Full article ">Figure 8
<p>Receiver operating characteristics (ROC) based on the Outbox test dataset: (<b>a</b>) Round 2 (R2); (<b>b</b>) Round 3, 25% Outbox (R3-25%).</p> Full article ">Figure 9
<p>Comparison of ResNet-50 classification performance on the Outbox test dataset in different rounds: (<b>a</b>) 2-class; (<b>b</b>) 3-class.</p> Full article ">Figure 10
<p>Performance of trained models on the new test dataset (Inbox_dp and Outbox_Q_dp, <a href="#jnt-04-00011-f002" class="html-fig">Figure 2</a>a): (<b>a</b>) Inbox_dp vs. Outbox_Q_dp in 2-class and 3-class classifications; (<b>b</b>) Model effects in 2-class and 3-class classifications; (<b>c</b>) ROC in Rounds 1, 2, and 3.</p> Full article ">Figure 11
<p>Image analysis: (<b>a</b>) Heat map; (<b>b</b>) Features from the 2nd ReLU layer. The true class of the image was D2, with EfficientNet misclassifying it as D1 and the other three models classifying it correctly.</p> Full article ">
Open AccessReview
The Golden Liposomes: Preparation and Biomedical Applications of Gold-Liposome Nanocomposites
by
Sourour Idoudi, Roua Ismail, Ousama Rachid, Abdelbary Elhissi and Alaaldin M. Alkilany
J. Nanotheranostics 2023, 4(3), 201-227; https://doi.org/10.3390/jnt4030010 - 25 Jun 2023
Cited by 2
Abstract
►▼
Show Figures
Gold nanoparticles (AuNP) have received a growing attention due to their fascinating physiochemical properties and promising range of biomedical applications including sensing, diagnosis and cancer photothermal ablation. AuNP enjoy brilliant optical properties and ability to convert light into local heat and function as
[...] Read more.
Gold nanoparticles (AuNP) have received a growing attention due to their fascinating physiochemical properties and promising range of biomedical applications including sensing, diagnosis and cancer photothermal ablation. AuNP enjoy brilliant optical properties and ability to convert light into local heat and function as a “nanoheaters” to fight cancer. However, AuNP are poor drug delivery systems as they do not have reservoirs or matrices to achieve an acceptable drug loading efficiency. On the other end, liposome-based nanocarriers do not exhibit such optical properties but are excellent platform for drug loading and they have been proven clinically with a true presence in the market since the FDA approved Doxil® in 1995. Combining the brilliant optical and photothermal properties of AuNP with the excellent drug loading capability of liposome should yield nanocomposites that enjoy the features of both modalities and enable the development of novel and smart drug delivery systems. Therefore, this review discusses the up-to date research on the AuNP-liposome nanocomposites and the current available approaches and protocols for their preparation and characterization. Finally, the biomedical applications of AuNP-liposome nanocomposites and proposed future directions in this field are discussed.
Full article
Graphical abstract
Graphical abstract
Full article ">Figure 1
<p>Various biomedical applications of gold nanoparticles as labeled. Figure created in BioRender.com.</p> Full article ">Figure 2
<p>Illustration of different architectures of AuNP-liposome nanocomposites. Pre-prepared hydrophilic AuNP on the outermost shell (<b>A</b>) or in the aqueous reservoir (<b>B</b>) of a liposome. In (<b>C</b>), pre-prepared ultrasmall hydrophobic AuNP are embedded in the bilayer of a liposome. Figure created in BioRender.com.</p> Full article ">Figure 3
<p>Schematic illustration of the preparation of AuNP-liposome nanocomposite using hydrophilic AuNP. Note that the preparations of two different architectures are sketched: AuNP in the aqueous core of the liposomes (<b>A</b>) and AuNP on the outermost shell of the liposome (<b>B</b>). For the later, attractive forces (electrostatic or covalent interaction) should exist for stable assembly. Figure created in BioRender.com.</p> Full article ">Figure 4
<p>Schematic illustration of the preparation of AuNP-liposome nanocomposite using ultra-small hydrophobic AuNP. Note that the resulting architectures implies the incorporation of AuNP into the lipid membrane bilayer. Figure created in BioRender.com.</p> Full article ">Figure 5
<p>Schematic illustration of the AuNP-liposome nanocomposites formation using the “in-liposome” (<b>A</b>) and “on-liposome” (<b>B</b>) reduction approaches. Figure created in BioRender.com.</p> Full article ">Figure 6
<p>(<b>A</b>) TEM images of (<b>i</b>) AuNP-surface coated liposomes, (<b>ii</b>) AuNP-Loaded liposomes, (<b>iii</b>) AuNPs within the liposomal bilayer, and (<b>iv</b>) Cryo-EM images of AuNP-surface coated liposomes. (<b>B</b>) Successful incorporation of AuNP is evident from the development of violet color in the liposome suspension. (<b>C</b>) Absorption spectra of free drug, empty liposomes and AuNP-liposome in aqueous environment illustrating the differences in the Vis-NIR absorbance upon AuNP incorporation. (<b>D</b>) Elemental analysis of AuNP-liposome nanocomposites. (<b>E</b>) Spike fractograms from ICP-MS reporting the detected AuNP mass and indicating (<b>Ei</b>) AuNP particle size of 30 nm, and (<b>Eii</b>) AuNP-liposome size around 200 nm. (<b>A</b>) Reproduced with permission from (<b>i</b>) [<a href="#B122-jnt-04-00010" class="html-bibr">122</a>], copyright Frontiers 2015, (<b>ii</b>) [<a href="#B164-jnt-04-00010" class="html-bibr">164</a>], copyright IOP Publishing Ltd. 2018, (<b>iii</b>) [<a href="#B171-jnt-04-00010" class="html-bibr">171</a>], copyright The Royal Society of Chemistry 2012, and (<b>iv</b>) [<a href="#B172-jnt-04-00010" class="html-bibr">172</a>], copyright Elsevier 2009. (<b>B</b>) Reproduced with permission from [<a href="#B169-jnt-04-00010" class="html-bibr">169</a>], copyright PLOS 2020. (<b>C</b>) Reproduced with permission from [<a href="#B130-jnt-04-00010" class="html-bibr">130</a>], copyright Elsevier 2018. (<b>D</b>) Reproduced with permission from [<a href="#B133-jnt-04-00010" class="html-bibr">133</a>], copyright IOP Publishing 2018. (<b>E</b>) Reproduced from [<a href="#B170-jnt-04-00010" class="html-bibr">170</a>].</p> Full article ">Figure 7
<p>Graphical illustration of the protective properties of the AuNP encapsulated into the liposomal core against chemical etching with cyanide ions. Figure created in BioRender.com.</p> Full article ">Figure 8
<p>CT of laboratory mice with Lewis lung carcinoma after intravenous administration of AuNP-Phospholipid nanocomposite in the presence (<b>A</b>) and absence (<b>B</b>) of Poly (ethylene glycol)-distearoylphosphatidylethanolamine (PEG-DSPE) after 90 minutes illustrating a significant gold accumulation in the tumor site from the PEGylated AuNP-Phospholipid nanocomposite compared to the non-PEGylated nanocomposite (shown in the red circles). (<b>C</b>) X-ray CT imaging of AuNP-liposome (shown in the arrow) illustrating a superior CT contrast signal compared to the generated signal from the commercially available gold seeds. (<b>A</b>,<b>B</b>) Reproduced with permission from [<a href="#B33-jnt-04-00010" class="html-bibr">33</a>], copyright Elsevier 2021. (<b>C</b>) Reproduced with permission from [<a href="#B173-jnt-04-00010" class="html-bibr">173</a>], copyright RSC Publishing 2013.</p> Full article ">Figure 9
<p>Graphical illustration of the solution-based (<b>A</b>) and paper-based (<b>B</b>) Assays for detecting the Pore-Forming Toxin Listeriolysin O (LLO). Adapted with permission from [<a href="#B182-jnt-04-00010" class="html-bibr">182</a>], copyright American Chemical Society 2020.</p> Full article ">Figure 10
<p>(<b>A</b>) Fluorescent micrograph images of the photothermal-mediated therapy using AuNP-liposome nanocomposites and laser against MCF-7-fluc2-turboFP tumor cells (absence of red color represents cell death as shown by the arrow). (<b>B</b>) Pre- and post- injecting liposome-AuNP nanocomposite treatment bioluminescence images of mice bearing HT1080-fluc2-turboFP tumor xenograft showing the significant tumor suppression from the combined photothermal and chemothermal treatment. (<b>A</b>,<b>B</b>) Reproduced with permission from [<a href="#B122-jnt-04-00010" class="html-bibr">122</a>], copyright American Chemical Society 2015.</p> Full article ">Figure 11
<p>(<b>A</b>) Thermal images of tumor-bearing mice after exposure to NIR irradiation and injection with wedelolactone (anticancer agent) loaded AuNP-liposome nanocomposites. (<b>B</b>) Changes in the tumor volume in the tumor-bearing mice in different treatment groups. (<b>C</b>) Weights of the tumor in different treatment groups (* means <span class="html-italic">p</span> < 0.05 and ** means <span class="html-italic">p</span> < 0.01). (<b>D</b>) Images showing the removed tumors from the mice bearing S180 tumor after 2 weeks of treatment. All graphs were reproduced with permission from [<a href="#B192-jnt-04-00010" class="html-bibr">192</a>], copyright Elsevier 2019.</p> Full article ">Figure 12
<p>Hepatic uptake and urinary excretion clearances of a variety of macromolecules and nanoparticles in mice after intravenous injection. Nanoparticles include small extracellular vesicles, liposomes, and AuNP [<a href="#B205-jnt-04-00010" class="html-bibr">205</a>,<a href="#B206-jnt-04-00010" class="html-bibr">206</a>,<a href="#B207-jnt-04-00010" class="html-bibr">207</a>,<a href="#B208-jnt-04-00010" class="html-bibr">208</a>,<a href="#B209-jnt-04-00010" class="html-bibr">209</a>]. AuNP (20 nm) and liposomes (150 nm) are shown in red rectangles. Note: Clearances of nanoparticles are approximate values estimated from the figures shown in the references, since raw data were not available. A direct comparison of data from the same reference is possible. However, attention should be paid when absolute clearance values from different references are compared because experimental designs, such as labeling probes and sampling time schedules, were different. The graph was reproduced with permission from [<a href="#B193-jnt-04-00010" class="html-bibr">193</a>], copyright Elsevier 2022.</p> Full article ">
Full article ">Figure 1
<p>Various biomedical applications of gold nanoparticles as labeled. Figure created in BioRender.com.</p> Full article ">Figure 2
<p>Illustration of different architectures of AuNP-liposome nanocomposites. Pre-prepared hydrophilic AuNP on the outermost shell (<b>A</b>) or in the aqueous reservoir (<b>B</b>) of a liposome. In (<b>C</b>), pre-prepared ultrasmall hydrophobic AuNP are embedded in the bilayer of a liposome. Figure created in BioRender.com.</p> Full article ">Figure 3
<p>Schematic illustration of the preparation of AuNP-liposome nanocomposite using hydrophilic AuNP. Note that the preparations of two different architectures are sketched: AuNP in the aqueous core of the liposomes (<b>A</b>) and AuNP on the outermost shell of the liposome (<b>B</b>). For the later, attractive forces (electrostatic or covalent interaction) should exist for stable assembly. Figure created in BioRender.com.</p> Full article ">Figure 4
<p>Schematic illustration of the preparation of AuNP-liposome nanocomposite using ultra-small hydrophobic AuNP. Note that the resulting architectures implies the incorporation of AuNP into the lipid membrane bilayer. Figure created in BioRender.com.</p> Full article ">Figure 5
<p>Schematic illustration of the AuNP-liposome nanocomposites formation using the “in-liposome” (<b>A</b>) and “on-liposome” (<b>B</b>) reduction approaches. Figure created in BioRender.com.</p> Full article ">Figure 6
<p>(<b>A</b>) TEM images of (<b>i</b>) AuNP-surface coated liposomes, (<b>ii</b>) AuNP-Loaded liposomes, (<b>iii</b>) AuNPs within the liposomal bilayer, and (<b>iv</b>) Cryo-EM images of AuNP-surface coated liposomes. (<b>B</b>) Successful incorporation of AuNP is evident from the development of violet color in the liposome suspension. (<b>C</b>) Absorption spectra of free drug, empty liposomes and AuNP-liposome in aqueous environment illustrating the differences in the Vis-NIR absorbance upon AuNP incorporation. (<b>D</b>) Elemental analysis of AuNP-liposome nanocomposites. (<b>E</b>) Spike fractograms from ICP-MS reporting the detected AuNP mass and indicating (<b>Ei</b>) AuNP particle size of 30 nm, and (<b>Eii</b>) AuNP-liposome size around 200 nm. (<b>A</b>) Reproduced with permission from (<b>i</b>) [<a href="#B122-jnt-04-00010" class="html-bibr">122</a>], copyright Frontiers 2015, (<b>ii</b>) [<a href="#B164-jnt-04-00010" class="html-bibr">164</a>], copyright IOP Publishing Ltd. 2018, (<b>iii</b>) [<a href="#B171-jnt-04-00010" class="html-bibr">171</a>], copyright The Royal Society of Chemistry 2012, and (<b>iv</b>) [<a href="#B172-jnt-04-00010" class="html-bibr">172</a>], copyright Elsevier 2009. (<b>B</b>) Reproduced with permission from [<a href="#B169-jnt-04-00010" class="html-bibr">169</a>], copyright PLOS 2020. (<b>C</b>) Reproduced with permission from [<a href="#B130-jnt-04-00010" class="html-bibr">130</a>], copyright Elsevier 2018. (<b>D</b>) Reproduced with permission from [<a href="#B133-jnt-04-00010" class="html-bibr">133</a>], copyright IOP Publishing 2018. (<b>E</b>) Reproduced from [<a href="#B170-jnt-04-00010" class="html-bibr">170</a>].</p> Full article ">Figure 7
<p>Graphical illustration of the protective properties of the AuNP encapsulated into the liposomal core against chemical etching with cyanide ions. Figure created in BioRender.com.</p> Full article ">Figure 8
<p>CT of laboratory mice with Lewis lung carcinoma after intravenous administration of AuNP-Phospholipid nanocomposite in the presence (<b>A</b>) and absence (<b>B</b>) of Poly (ethylene glycol)-distearoylphosphatidylethanolamine (PEG-DSPE) after 90 minutes illustrating a significant gold accumulation in the tumor site from the PEGylated AuNP-Phospholipid nanocomposite compared to the non-PEGylated nanocomposite (shown in the red circles). (<b>C</b>) X-ray CT imaging of AuNP-liposome (shown in the arrow) illustrating a superior CT contrast signal compared to the generated signal from the commercially available gold seeds. (<b>A</b>,<b>B</b>) Reproduced with permission from [<a href="#B33-jnt-04-00010" class="html-bibr">33</a>], copyright Elsevier 2021. (<b>C</b>) Reproduced with permission from [<a href="#B173-jnt-04-00010" class="html-bibr">173</a>], copyright RSC Publishing 2013.</p> Full article ">Figure 9
<p>Graphical illustration of the solution-based (<b>A</b>) and paper-based (<b>B</b>) Assays for detecting the Pore-Forming Toxin Listeriolysin O (LLO). Adapted with permission from [<a href="#B182-jnt-04-00010" class="html-bibr">182</a>], copyright American Chemical Society 2020.</p> Full article ">Figure 10
<p>(<b>A</b>) Fluorescent micrograph images of the photothermal-mediated therapy using AuNP-liposome nanocomposites and laser against MCF-7-fluc2-turboFP tumor cells (absence of red color represents cell death as shown by the arrow). (<b>B</b>) Pre- and post- injecting liposome-AuNP nanocomposite treatment bioluminescence images of mice bearing HT1080-fluc2-turboFP tumor xenograft showing the significant tumor suppression from the combined photothermal and chemothermal treatment. (<b>A</b>,<b>B</b>) Reproduced with permission from [<a href="#B122-jnt-04-00010" class="html-bibr">122</a>], copyright American Chemical Society 2015.</p> Full article ">Figure 11
<p>(<b>A</b>) Thermal images of tumor-bearing mice after exposure to NIR irradiation and injection with wedelolactone (anticancer agent) loaded AuNP-liposome nanocomposites. (<b>B</b>) Changes in the tumor volume in the tumor-bearing mice in different treatment groups. (<b>C</b>) Weights of the tumor in different treatment groups (* means <span class="html-italic">p</span> < 0.05 and ** means <span class="html-italic">p</span> < 0.01). (<b>D</b>) Images showing the removed tumors from the mice bearing S180 tumor after 2 weeks of treatment. All graphs were reproduced with permission from [<a href="#B192-jnt-04-00010" class="html-bibr">192</a>], copyright Elsevier 2019.</p> Full article ">Figure 12
<p>Hepatic uptake and urinary excretion clearances of a variety of macromolecules and nanoparticles in mice after intravenous injection. Nanoparticles include small extracellular vesicles, liposomes, and AuNP [<a href="#B205-jnt-04-00010" class="html-bibr">205</a>,<a href="#B206-jnt-04-00010" class="html-bibr">206</a>,<a href="#B207-jnt-04-00010" class="html-bibr">207</a>,<a href="#B208-jnt-04-00010" class="html-bibr">208</a>,<a href="#B209-jnt-04-00010" class="html-bibr">209</a>]. AuNP (20 nm) and liposomes (150 nm) are shown in red rectangles. Note: Clearances of nanoparticles are approximate values estimated from the figures shown in the references, since raw data were not available. A direct comparison of data from the same reference is possible. However, attention should be paid when absolute clearance values from different references are compared because experimental designs, such as labeling probes and sampling time schedules, were different. The graph was reproduced with permission from [<a href="#B193-jnt-04-00010" class="html-bibr">193</a>], copyright Elsevier 2022.</p> Full article ">
Open AccessReview
Self-Assembled Monolayers Derived from Positively Charged Adsorbates on Plasmonic Substrates for MicroRNA Delivery: A Review
by
Johnson Hoang, Pooria Tajalli, Mina Omidiyan, Maria D. Marquez, Orawan Khantamat, Wirote Tuntiwechapikul, Chien-Hung Li, Arati Kohlhatkar, Hung-Vu Tran, Preethi H. Gunaratne and T. Randall Lee
J. Nanotheranostics 2023, 4(2), 171-200; https://doi.org/10.3390/jnt4020009 - 8 May 2023
Cited by 2
Abstract
MicroRNA (miRNA) has emerged as a promising alternative therapeutic treatment for cancer, but its delivery has been hindered by low cellular uptake and degradation during circulation. In this review, we discuss the various methods of delivering miRNA, including viral and non-viral delivery systems
[...] Read more.
MicroRNA (miRNA) has emerged as a promising alternative therapeutic treatment for cancer, but its delivery has been hindered by low cellular uptake and degradation during circulation. In this review, we discuss the various methods of delivering miRNA, including viral and non-viral delivery systems such as liposomes and nanoparticles. We also examine the use of nanoparticles for miRNA-based diagnostics. We focus specifically on non-viral delivery systems utilizing coinage metals in the form of nanoparticles and the use of self-assembled monolayers (SAMs) as a method of surface modification. We review the use of SAMs for the conjugation and delivery of small noncoding ribonucleic acid (ncRNA), particularly SAMs derived from positively charged adsorbates to generate charged surfaces that can interact electrostatically with negatively charged miRNA. We also discuss the effects of the cellular uptake of gold and other plasmonic nanoparticles, as well as the challenges associated with the degradation of oligonucleotides. Our review highlights the potential of SAM-based systems as versatile and robust tools for delivering miRNA and other RNAs in vitro and in vivo and the need for further research to address the challenges associated with miRNA delivery and diagnostics.
Full article
(This article belongs to the Topic Advanced Technologies for Drug Delivery, Pathogen Detection and Diagnostics)
►▼
Show Figures
Figure 1
Figure 1
<p>The canonical pathway of miRNA biogenesis. Most miRNA genes are transcribed by RNA polymerase II. The primary miRNAs (pri-miRNAs) are usually long, with a section of ~70 nt that forms a hairpin structure. The pri-miRNAs are cleaved into a miRNA precursor (pre-miRNA) in the nucleus by a microprocessor complex consisting of Drosha and DiGeorge Syndrome Critical Region 8 (DGCR8) protein. The pre-miRNA is then exported to the cytosol by the complex of Exportin-5 (XPO5) and Ran-GTP. In the cytoplasm, Ran-GTP is hydrolyzed to Ran-GDP, and the transport complex releases its pre-miRNA. The pre-miRNA is then cleaved by Dicer, in association with trans-activation response (TAR) RNA-binding protein (TRBP), to generate a mature miRNA duplex. The miRNA duplex-Dicer-TRBP complex then interacts with an Argonaute (AGO) protein to form the RISC-loading complex (RLC). AGO selects a guide strand, and the assembly of the RNA-induced silencing complex (RISC) proceeds. RISC recognizes the mRNA target through base-pairing with the miRNA, which then leads to mRNA degradation or translation inhibition.</p> Full article ">Figure 2
<p>The base pairing of guide strand in RISC and its target mRNA. Schematic represents base pairing between a guide miRNA strand (purple) and a target mRNA (blue). The guide miRNA strand can be divided into four domains from 5′ to 3′. The seed region (g2–g8) is essential for target recognition. The central region (g9–g12) is important for mRNA and passenger strand cleavage. The 3′-supplementary region (g13–g17) stabilizes the complex with the mRNA target. The tail region (g18-3′ end) regulates the turnover of target cleavage and the fate of RISC. The 5′ end and 3′ end of the guide strand are anchored in the MID and PAZ domains of AGO2, respectively. Adapted with permission from reference [<a href="#B42-jnt-04-00009" class="html-bibr">42</a>]. Copyright 2022, Cell Press.</p> Full article ">Figure 3
<p>Schematic illustrations depicting (<b>A</b>) normal growth, proliferation, differentiation, and cell death in normal tissue occurring through proper microRNA transcription; (<b>B</b>) defects at different stages of tumor suppressor miRNA biogenesis leading to the formation of oncoproteins; and (<b>C</b>) overexpression of oncogenic miRNA inhibiting expression of tumor suppressor protein and resulting in tumor formation. Reproduced with permission from reference [<a href="#B51-jnt-04-00009" class="html-bibr">51</a>]. Copyright 2006, Springer Nature.</p> Full article ">Figure 4
<p>Depiction of the “proton sponge” mechanism after endocytosis of gold nanoparticles to escape the endosome. Reproduced with permission from reference [<a href="#B74-jnt-04-00009" class="html-bibr">74</a>]. Copyright 2009, Springer Nature.</p> Full article ">Figure 5
<p>Schematic representation of the three essential parts of the adsorbate (terminal functional group, spacer, and headgroup on the substrate).</p> Full article ">Figure 6
<p>(<b>A</b>) Illustration (Not to Scale) of the Preparation of miRNA−AuNP Conjugates for Delivering miRNAs to Cells. Reproduced with permission from reference [<a href="#B113-jnt-04-00009" class="html-bibr">113</a>]. Copyright 2012 American Chemical Society. (<b>B</b>) Gold-nanocage-based targeted nanocomplex formulation process and (<b>C</b>) NIR-laser-induced targeted gene-photothermal therapy using the nanocomplexes. Adapted with permission from reference [<a href="#B118-jnt-04-00009" class="html-bibr">118</a>]. Copyright 2016 Wiley.</p> Full article ">Figure 7
<p>(<b>A</b>) Light-triggered release of miR-34a from nanoshells in TNBC cells. (<b>B</b>) Scheme depicting the process to coat NS with miRNA and Mpegsh. (<b>C</b>) Transmission electron micrograph of miR-co/NS. (<b>D</b>) Plasmon resonant extinction spectra comparing miRNA/NS conjugates to bare NS with peak extinction at 810 nm. (<b>E</b>) Hydrodynamic diameters (black outline) and zeta potential measurements (gray outline) of miRNA/NS conjugates and bare NS. (<b>F</b>) OliGreen analysis of miRNA loading on both miR34a/NS and miRco/NS. Reproduced with permission from reference [<a href="#B116-jnt-04-00009" class="html-bibr">116</a>]. Copyright 2021 American Chemical Society.</p> Full article ">Figure 8
<p>(<b>A</b>) Schematic illustration of gold-nanoparticle-based NIR triggered sequential miR-21 inhibitor/Dox release with precise time interval for optimal combination therapy. (I–III) Formation of HGNP-based co-delivery system. D-P-HGNPs/21i entered cells through endocytosis (IV). Upon entering the tumor cells, miR-21i was released first (V), modulating the intrinsic state to a more chemosensitive state (VI). At the desired time, application of NIR laser triggered collapse of HGNPs and a burst release of doxorubicin (VII), activating two apoptosis signaling pathways, thereby inducing the synergistic apoptosis response (IX). (<b>B</b>) TEM images of HGNPs, D-P-HGNPs, D-P-HGNPs, and D-P-HGNPs-R. (<b>C</b>) Particle size measured by DLS. Adapted with permission from reference [<a href="#B115-jnt-04-00009" class="html-bibr">115</a>]. Copyright 2016 Elsevier.</p> Full article ">Figure 9
<p>(<b>A</b>) Schematic illustration of synthesis of PolyGIONs and fluorescence images of Cy5-labeled miR-100 and antimiR-21-loaded CD-CS complexes. (<b>B</b>) Schematic representation of as-prepared polyGION structure and their various compositions. (<b>C</b>) TEM micrograph of GION. (<b>D</b>) GION coated with miR-100/antimiR-21 encapsulated CD-CS polymer layer, its EDS analysis, and corresponding images. (<b>E</b>) DLS measuring zeta potential and size of nanoparticles after each step of GION surface modification. (<b>F</b>) UV-vis spectra of as-prepared GION NS and its precursor nanoparticles. (<b>G</b>) Gel retardation assay for Cy5-miR-100/antimiR-21 encapsulation in CD-CS hybrid polymer and RNase protection assay for measuring the stability of encapsulated miRNAs in polyGION-CD-CS. Adapted with permission from reference [<a href="#B120-jnt-04-00009" class="html-bibr">120</a>]. Copyright 2019 Elsevier.</p> Full article ">Figure 10
<p>Electrochemical biosensor constructed from DSN-assisted target recycling and TMSD-based catalytic hairpin assembly (CHA) where an amplified “b” and a weak “a” electrochemical signals are measured by differential pulse voltammetry (DPV) in the presence of miR-141 and in the absence of miR-141, repectively. Reprinted (adapted) with permission from [<a href="#B125-jnt-04-00009" class="html-bibr">125</a>]. Copyright 2018 American Chemical Society.</p> Full article ">Figure 11
<p>(<b>A</b>) Purification of miRNA-107 by streptavidin-functionalized (blue part) magnetic beads (Dynabeads) followed by electrochemical measurements of the miRNA-deposited gold-loaded cubic nanoporous ferric oxide on the SPCE (light blue surface). Reprinted from [<a href="#B142-jnt-04-00009" class="html-bibr">142</a>], Copyright 2018, with permission from Elsevier. (<b>B</b>) Photonic resonator absorption microscopy (PRAM) utilizing magneto-plasmonic nanoparticles (MPNPs) on the photonic crystal (PC) surface constructed from (TiO<sub>2</sub>-SiO<sub>2</sub>). Reprinted (adapted) with permission from [<a href="#B141-jnt-04-00009" class="html-bibr">141</a>]. Copyright 2022 American Chemical Society.</p> Full article ">Figure 12
<p>Assembly of AuNRs to create nanogap antennas.Reprinted with permission from Ref. [<a href="#B133-jnt-04-00009" class="html-bibr">133</a>]. Copyright 2020, Wiley-VCH GmbH.</p> Full article ">Figure 13
<p>(<b>A</b>) Purification of target miRNA followed by hybridization to the Au film with immobilized DNA in an enzyme-free SPR biosensor. Reprinted from [<a href="#B123-jnt-04-00009" class="html-bibr">123</a>], Copyright 2017, with permission from Elsevier. (<b>B</b>) SPR biosensor constructed from AuNPs-MoS<sub>2</sub> nanocomposites to enhance SPR signal. Reprinted from [<a href="#B139-jnt-04-00009" class="html-bibr">139</a>], Copyright 2017, with permission from Elsevier.</p> Full article ">Figure 14
<p>AgNPs in an enzyme-free SERS biosensor using mismatched CHA reactions. Reprinted from [<a href="#B87-jnt-04-00009" class="html-bibr">87</a>], Copyright 2019, with permission from Elsevier.</p> Full article ">
<p>The canonical pathway of miRNA biogenesis. Most miRNA genes are transcribed by RNA polymerase II. The primary miRNAs (pri-miRNAs) are usually long, with a section of ~70 nt that forms a hairpin structure. The pri-miRNAs are cleaved into a miRNA precursor (pre-miRNA) in the nucleus by a microprocessor complex consisting of Drosha and DiGeorge Syndrome Critical Region 8 (DGCR8) protein. The pre-miRNA is then exported to the cytosol by the complex of Exportin-5 (XPO5) and Ran-GTP. In the cytoplasm, Ran-GTP is hydrolyzed to Ran-GDP, and the transport complex releases its pre-miRNA. The pre-miRNA is then cleaved by Dicer, in association with trans-activation response (TAR) RNA-binding protein (TRBP), to generate a mature miRNA duplex. The miRNA duplex-Dicer-TRBP complex then interacts with an Argonaute (AGO) protein to form the RISC-loading complex (RLC). AGO selects a guide strand, and the assembly of the RNA-induced silencing complex (RISC) proceeds. RISC recognizes the mRNA target through base-pairing with the miRNA, which then leads to mRNA degradation or translation inhibition.</p> Full article ">Figure 2
<p>The base pairing of guide strand in RISC and its target mRNA. Schematic represents base pairing between a guide miRNA strand (purple) and a target mRNA (blue). The guide miRNA strand can be divided into four domains from 5′ to 3′. The seed region (g2–g8) is essential for target recognition. The central region (g9–g12) is important for mRNA and passenger strand cleavage. The 3′-supplementary region (g13–g17) stabilizes the complex with the mRNA target. The tail region (g18-3′ end) regulates the turnover of target cleavage and the fate of RISC. The 5′ end and 3′ end of the guide strand are anchored in the MID and PAZ domains of AGO2, respectively. Adapted with permission from reference [<a href="#B42-jnt-04-00009" class="html-bibr">42</a>]. Copyright 2022, Cell Press.</p> Full article ">Figure 3
<p>Schematic illustrations depicting (<b>A</b>) normal growth, proliferation, differentiation, and cell death in normal tissue occurring through proper microRNA transcription; (<b>B</b>) defects at different stages of tumor suppressor miRNA biogenesis leading to the formation of oncoproteins; and (<b>C</b>) overexpression of oncogenic miRNA inhibiting expression of tumor suppressor protein and resulting in tumor formation. Reproduced with permission from reference [<a href="#B51-jnt-04-00009" class="html-bibr">51</a>]. Copyright 2006, Springer Nature.</p> Full article ">Figure 4
<p>Depiction of the “proton sponge” mechanism after endocytosis of gold nanoparticles to escape the endosome. Reproduced with permission from reference [<a href="#B74-jnt-04-00009" class="html-bibr">74</a>]. Copyright 2009, Springer Nature.</p> Full article ">Figure 5
<p>Schematic representation of the three essential parts of the adsorbate (terminal functional group, spacer, and headgroup on the substrate).</p> Full article ">Figure 6
<p>(<b>A</b>) Illustration (Not to Scale) of the Preparation of miRNA−AuNP Conjugates for Delivering miRNAs to Cells. Reproduced with permission from reference [<a href="#B113-jnt-04-00009" class="html-bibr">113</a>]. Copyright 2012 American Chemical Society. (<b>B</b>) Gold-nanocage-based targeted nanocomplex formulation process and (<b>C</b>) NIR-laser-induced targeted gene-photothermal therapy using the nanocomplexes. Adapted with permission from reference [<a href="#B118-jnt-04-00009" class="html-bibr">118</a>]. Copyright 2016 Wiley.</p> Full article ">Figure 7
<p>(<b>A</b>) Light-triggered release of miR-34a from nanoshells in TNBC cells. (<b>B</b>) Scheme depicting the process to coat NS with miRNA and Mpegsh. (<b>C</b>) Transmission electron micrograph of miR-co/NS. (<b>D</b>) Plasmon resonant extinction spectra comparing miRNA/NS conjugates to bare NS with peak extinction at 810 nm. (<b>E</b>) Hydrodynamic diameters (black outline) and zeta potential measurements (gray outline) of miRNA/NS conjugates and bare NS. (<b>F</b>) OliGreen analysis of miRNA loading on both miR34a/NS and miRco/NS. Reproduced with permission from reference [<a href="#B116-jnt-04-00009" class="html-bibr">116</a>]. Copyright 2021 American Chemical Society.</p> Full article ">Figure 8
<p>(<b>A</b>) Schematic illustration of gold-nanoparticle-based NIR triggered sequential miR-21 inhibitor/Dox release with precise time interval for optimal combination therapy. (I–III) Formation of HGNP-based co-delivery system. D-P-HGNPs/21i entered cells through endocytosis (IV). Upon entering the tumor cells, miR-21i was released first (V), modulating the intrinsic state to a more chemosensitive state (VI). At the desired time, application of NIR laser triggered collapse of HGNPs and a burst release of doxorubicin (VII), activating two apoptosis signaling pathways, thereby inducing the synergistic apoptosis response (IX). (<b>B</b>) TEM images of HGNPs, D-P-HGNPs, D-P-HGNPs, and D-P-HGNPs-R. (<b>C</b>) Particle size measured by DLS. Adapted with permission from reference [<a href="#B115-jnt-04-00009" class="html-bibr">115</a>]. Copyright 2016 Elsevier.</p> Full article ">Figure 9
<p>(<b>A</b>) Schematic illustration of synthesis of PolyGIONs and fluorescence images of Cy5-labeled miR-100 and antimiR-21-loaded CD-CS complexes. (<b>B</b>) Schematic representation of as-prepared polyGION structure and their various compositions. (<b>C</b>) TEM micrograph of GION. (<b>D</b>) GION coated with miR-100/antimiR-21 encapsulated CD-CS polymer layer, its EDS analysis, and corresponding images. (<b>E</b>) DLS measuring zeta potential and size of nanoparticles after each step of GION surface modification. (<b>F</b>) UV-vis spectra of as-prepared GION NS and its precursor nanoparticles. (<b>G</b>) Gel retardation assay for Cy5-miR-100/antimiR-21 encapsulation in CD-CS hybrid polymer and RNase protection assay for measuring the stability of encapsulated miRNAs in polyGION-CD-CS. Adapted with permission from reference [<a href="#B120-jnt-04-00009" class="html-bibr">120</a>]. Copyright 2019 Elsevier.</p> Full article ">Figure 10
<p>Electrochemical biosensor constructed from DSN-assisted target recycling and TMSD-based catalytic hairpin assembly (CHA) where an amplified “b” and a weak “a” electrochemical signals are measured by differential pulse voltammetry (DPV) in the presence of miR-141 and in the absence of miR-141, repectively. Reprinted (adapted) with permission from [<a href="#B125-jnt-04-00009" class="html-bibr">125</a>]. Copyright 2018 American Chemical Society.</p> Full article ">Figure 11
<p>(<b>A</b>) Purification of miRNA-107 by streptavidin-functionalized (blue part) magnetic beads (Dynabeads) followed by electrochemical measurements of the miRNA-deposited gold-loaded cubic nanoporous ferric oxide on the SPCE (light blue surface). Reprinted from [<a href="#B142-jnt-04-00009" class="html-bibr">142</a>], Copyright 2018, with permission from Elsevier. (<b>B</b>) Photonic resonator absorption microscopy (PRAM) utilizing magneto-plasmonic nanoparticles (MPNPs) on the photonic crystal (PC) surface constructed from (TiO<sub>2</sub>-SiO<sub>2</sub>). Reprinted (adapted) with permission from [<a href="#B141-jnt-04-00009" class="html-bibr">141</a>]. Copyright 2022 American Chemical Society.</p> Full article ">Figure 12
<p>Assembly of AuNRs to create nanogap antennas.Reprinted with permission from Ref. [<a href="#B133-jnt-04-00009" class="html-bibr">133</a>]. Copyright 2020, Wiley-VCH GmbH.</p> Full article ">Figure 13
<p>(<b>A</b>) Purification of target miRNA followed by hybridization to the Au film with immobilized DNA in an enzyme-free SPR biosensor. Reprinted from [<a href="#B123-jnt-04-00009" class="html-bibr">123</a>], Copyright 2017, with permission from Elsevier. (<b>B</b>) SPR biosensor constructed from AuNPs-MoS<sub>2</sub> nanocomposites to enhance SPR signal. Reprinted from [<a href="#B139-jnt-04-00009" class="html-bibr">139</a>], Copyright 2017, with permission from Elsevier.</p> Full article ">Figure 14
<p>AgNPs in an enzyme-free SERS biosensor using mismatched CHA reactions. Reprinted from [<a href="#B87-jnt-04-00009" class="html-bibr">87</a>], Copyright 2019, with permission from Elsevier.</p> Full article ">
Highly Accessed Articles
Latest Books
E-Mail Alert
News
Topics
Topic in
Biomedicines, JNT, Pharmaceutics, Polymers, Nanomaterials, Pharmaceuticals
Applications of Polymers and Polymer Nanomaterials in Drug Delivery and Nanomedicine
Topic Editors: Stanislav Rangelov, Emi HaladjovaDeadline: 31 May 2025

Conferences
Special Issues
Special Issue in
JNT
Biomimetic Nanodelivery: Recent Developments and Future Prospects
Guest Editor: Qinqin HuangDeadline: 31 August 2024
Special Issue in
JNT
Nanozymes: New Advances in the Next Generation of Enzyme-Like Nanosystems
Guest Editors: Herman Sander Mansur, Alexandra Ancelmo Piscitelli MansurDeadline: 30 September 2024
Special Issue in
JNT
Nanoengineered Solutions: Advancements in Targeted Drug Delivery and Theranostics
Guest Editor: Tamer ElbayoumiDeadline: 1 December 2024
Special Issue in
JNT
Interventional Nanotheranostics for Translational Nano-Immunotherapy
Guest Editor: Dong-Hyun KimDeadline: 31 December 2024


