WO2025145091A1 - Crystalline forms of a muscarinic m4 receptor modulator and methods of treating diseases - Google Patents
Crystalline forms of a muscarinic m4 receptor modulator and methods of treating diseases Download PDFInfo
- Publication number
- WO2025145091A1 WO2025145091A1 PCT/US2024/062161 US2024062161W WO2025145091A1 WO 2025145091 A1 WO2025145091 A1 WO 2025145091A1 US 2024062161 W US2024062161 W US 2024062161W WO 2025145091 A1 WO2025145091 A1 WO 2025145091A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- weight percent
- crystalline form
- pharmaceutical composition
- disease
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/28—Drugs for disorders of the nervous system for treating neurodegenerative disorders of the central nervous system, e.g. nootropic agents, cognition enhancers, drugs for treating Alzheimer's disease or other forms of dementia
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P25/00—Drugs for disorders of the nervous system
- A61P25/14—Drugs for disorders of the nervous system for treating abnormal movements, e.g. chorea, dyskinesia
- A61P25/16—Anti-Parkinson drugs
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07D—HETEROCYCLIC COMPOUNDS
- C07D471/00—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00
- C07D471/02—Heterocyclic compounds containing nitrogen atoms as the only ring hetero atoms in the condensed system, at least one ring being a six-membered ring with one nitrogen atom, not provided for by groups C07D451/00 - C07D463/00 in which the condensed system contains two hetero rings
- C07D471/04—Ortho-condensed systems
Definitions
- the disclosure relates to crystalline forms, i.e., polymorphs, of a muscarinic M4 receptor activator, pharmaceutical compositions and combinations comprising those crystalline forms, and their use in treating diseases and disorders, such as Alzheimer's Disease, psychosis in Alzheimer’s disease, schizophrenia, psychotic disorders, pain, addiction, bipolar disorder, tardive dyskinesia, and sleep disorders. Also provided herein are methods for producing the crystalline forms of this disclosure.
- M4 muscarinic acetylcholine receptor activators some researchers have focused on identifying subtype-selective M4 muscarinic acetylcholine receptor activators.
- PAMs positive allosteric modulators
- M4 muscarinic acetylcholine receptor have gained interest as possible agents for treating the behavioral impairments associated with schizophrenia and other neuropsychiatric disorders, e.g., Alzheimer’s Disease.
- PAMs positive allosteric modulators
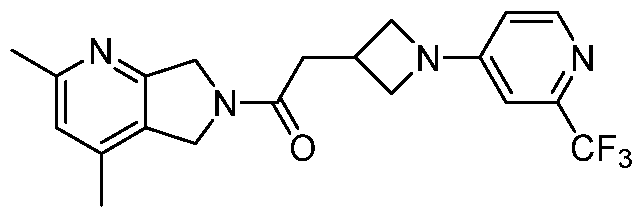
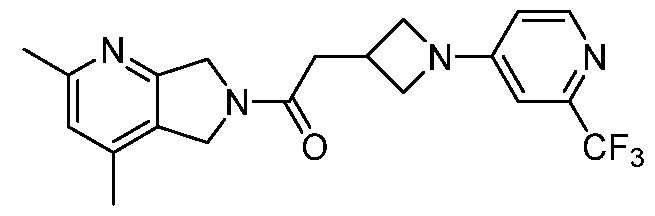
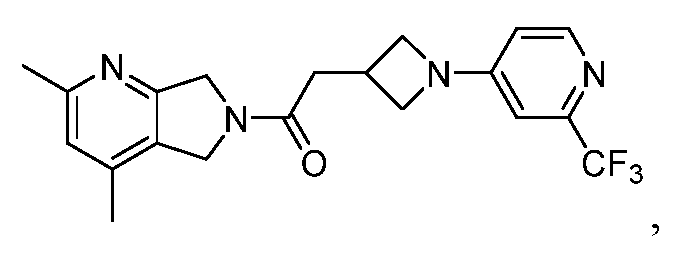
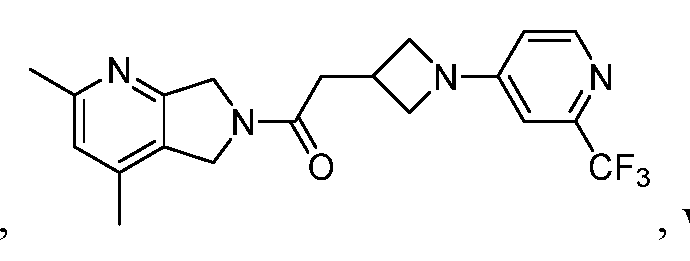
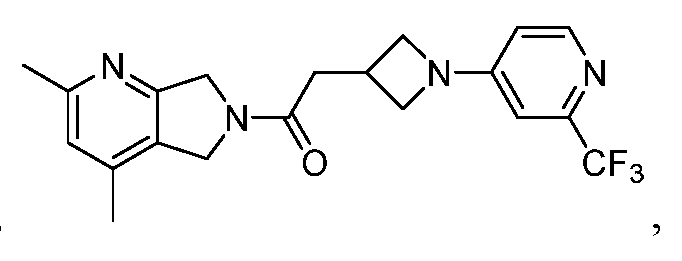
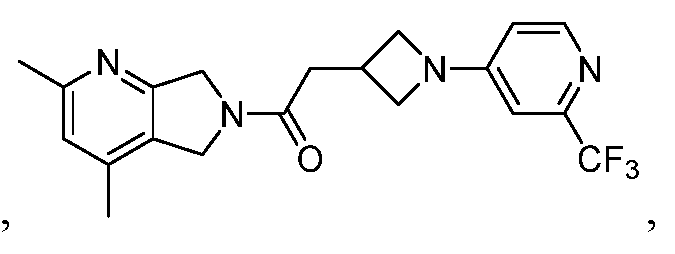
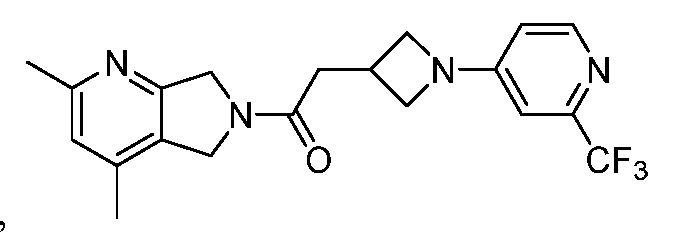
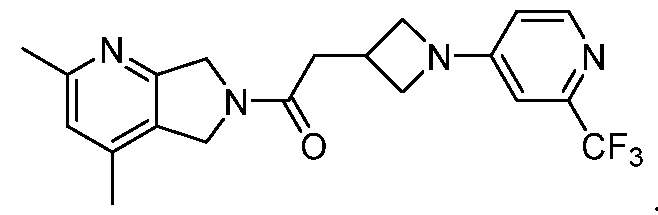
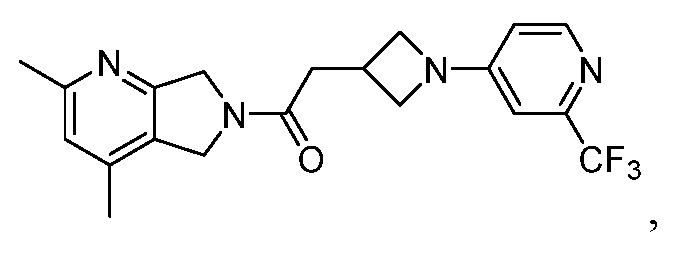
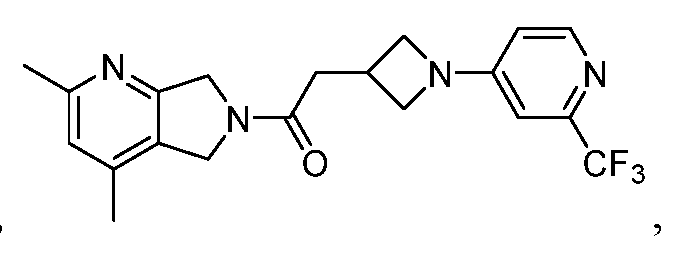
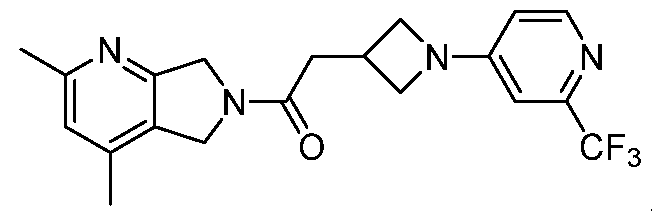
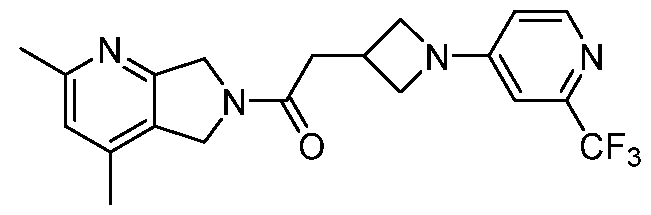
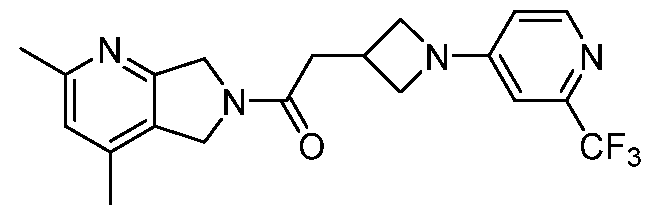
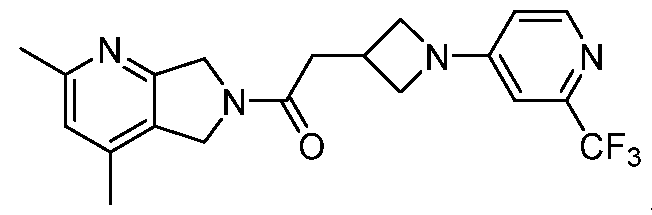
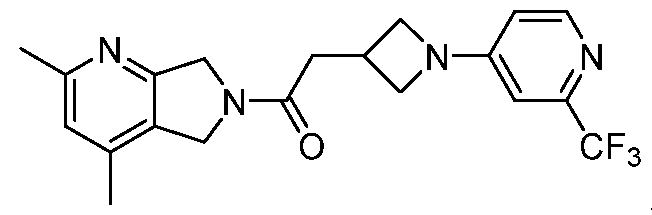
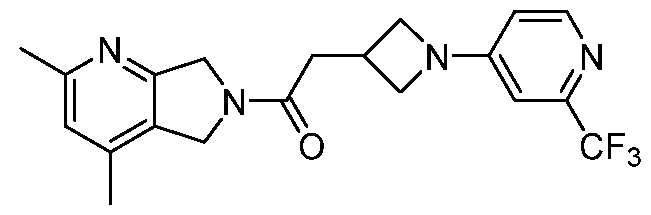
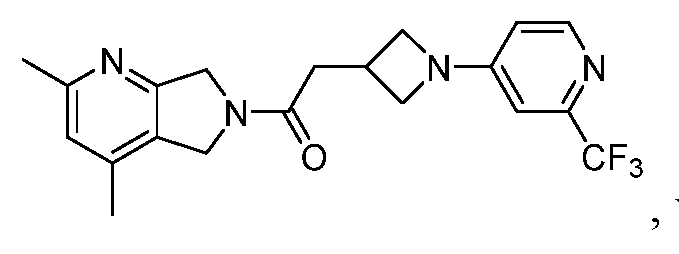
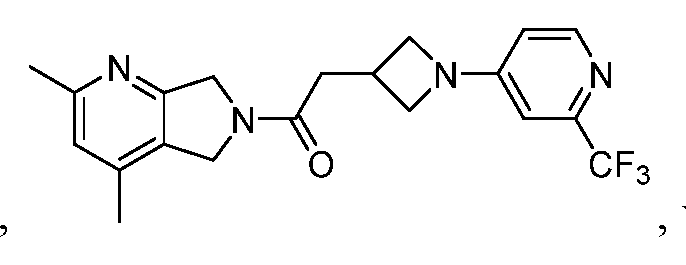
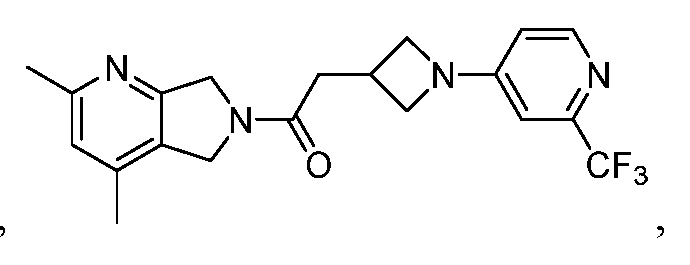
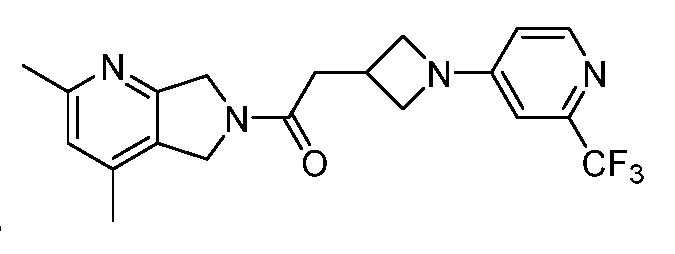
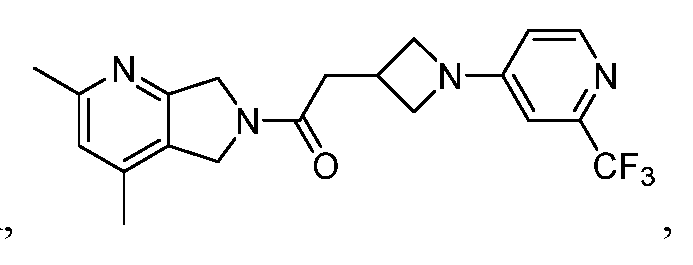
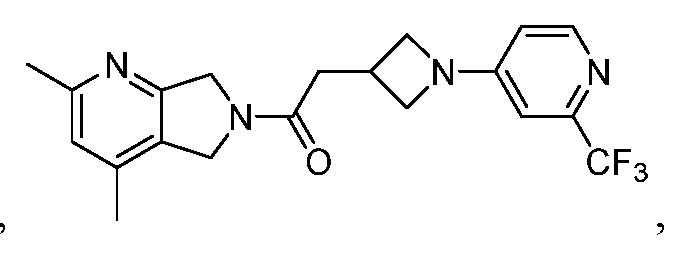
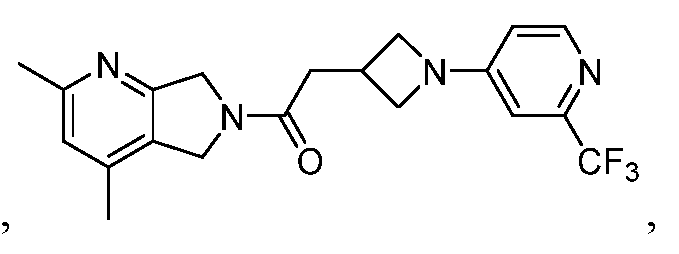
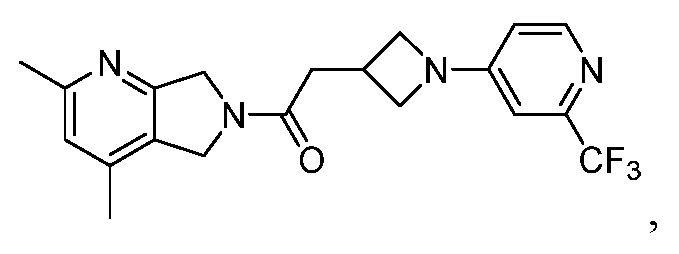
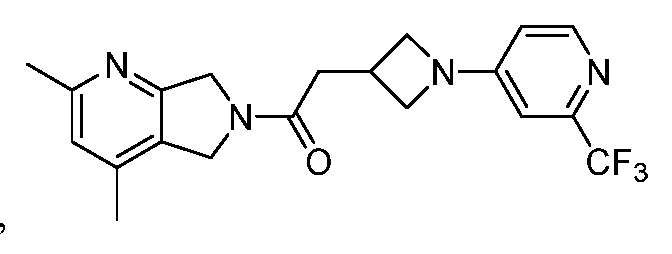
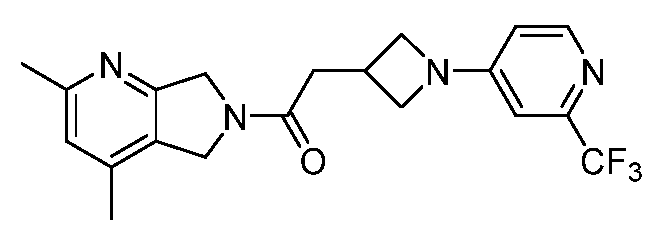
- One such PAM of the M4 muscarinic acetylcholine receptor is Compound A
- thermodynamically stable forms of Compound A or its hydrates forms of Compound A or its hydrates that can be manufactured in high purity and large quantities, and thus be, more suitable for inclusion in pharmaceutical formulations; and/or forms of Compound A or its hydrates that display other advantageous physical properties, such as lack of hygroscopic tendencies, improved solubility, and enhanced rates of dissolution due to different lattice energies.
- the present disclosure provides crystalline forms, i.e., polymorphs, of Compound A, .
- the crystalline forms of Compound A are anhydrous and, in some forms they are hydrates, e.g., hemi-, mono- or di -hydrated (together “crystalline forms of Compound A”).
- Also provided in the embodiments of this disclosure are processes for preparing the crystalline forms of Compound A as well as pharmaceutical compositions and combinations thereof, and methods for using those crystalline forms, compositions, and combinations in the treatment of diseases or disorders, e.g., Alzheimer’s Disease, dementia, including psychosis and agitation associated therewith, schizophrenia, psychotic disorders, pain, addiction, bipolar disorder (bipolar I, bipolar II, bipolar depression, manic or mixed episodes), tardive dyskinesia, and sleep disorders.
- diseases or disorders e.g., Alzheimer’s Disease, dementia, including psychosis and agitation associated therewith, schizophrenia, psychotic disorders, pain, addiction, bipolar disorder (bipolar I, bipolar II, bipolar depression, manic or mixed episodes), tardive dyskinesia, and sleep disorders.
- the present disclosure provides a crystalline form of Compound A, wherein the crystalline form is Form 1 as described herein.
- the present disclosure provides a crystalline form of Compound A, wherein the crystalline form is Form 2 as described herein.
- the present disclosure provides a crystalline form of Compound A, wherein the crystalline form is Form 3 as described herein.
- the present disclosure provides a crystalline form of Compound A, wherein the crystalline form is Form 4 as described herein.
- the present disclosure is directed to a pharmaceutical composition
- a pharmaceutical composition comprising one or more of a crystalline form of Compound A and at least one pharmaceutically acceptable carrier.
- the present disclosure provides a method for treating a disease or disorder in a patient in need thereof, said method comprising administering to the patient a therapeutically effective amount of one or more of a crystalline form of Compound A according to the disclosure, or a pharmaceutical composition or combination of them.
- a crystalline form of Compound A according to the disclosure, or a pharmaceutical composition or combination of them are administered as a mono or adjunctive treatment for the disease or disorder.
- the present disclosure provides a method of producing a crystalline form of Compound A described herein (i.e., Form 1, Form 2, Form 3, and Form 4 of Compound A).
- a crystalline form of Compound A, wherein the crystalline form is Form 1.
- the disease or disorder is selected from Alzheimer’s Disease, psychosis in Alzheimer’s Disease, schizophrenia or psychosis, pain, addiction, a sleep disorder, a cognitive disorder (e.g. mild cognitive impairment), Parkinson’s Disease, Parkinson’s Disease-levodopa-induced dyskinesia, Huntington’s Disease, dyskinesia, dry mouth, pulmonary hypertension, chronic obstructive pulmonary disease (COPD), asthma, urinary incontinence, glaucoma, Trisomy 21 (Down Syndrome), cerebral amyloid angiopathy, dementia, Hereditary Cerebral Hemorrhage with Amyloidosis of the Dutch-Type (HCHWA-D), Creutzfeld-Jakob disease, prion disorders, amyotrophic lateral sclerosis, progressive supranuclear palsy, head trauma, stroke, pancreatitis, inclusion body myositis, other peripheral amyloidoses, diabetes, autism, tardive dyskinesia
- a cognitive disorder e.
- the disease or disorder is selected from Alzheimer’s disease, dementia, a bipolar disorder, schizophrenia, pain, an addiction, a sleep disorder, and conditions associated therewith.
- a method of producing crystalline Form 1 of Compound A comprising the steps of:
- a pharmaceutical composition comprising about 30 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 85 weight percent to about 93 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
- composition according to any one of embodiments 46 to 50, wherein the opacifier is selected from the group consisting of: titanium dioxide, silicon dioxide, zinc oxide, iron oxide, talc, calcium carbonate, calcium phosphate, calcium sulfate, behenic acid, aluminum silicate, aluminum hydroxide, magnesium carbonate, tartrazine, erythrosine, or a mixture thereof.
- composition according to any one of embodiments 46 to 50, wherein the opacifier is titanium dioxide.
- coating comprises the opacifier in an amount of from about 5 weight percent to about 15 weight percent, or about 10 weight percent to about 15 weight percent, or about 15 weight percent to about 20 weight percent of the coating by weight, based upon total weight of the coating composition.
- composition according to any one of embodiments 46 to 56 comprising crystalline Form 2 of Compound A according to any one of embodiments 4 to 6.
- the disease or disorder is selected from Alzheimer’s Disease, psychosis in Alzheimer’s Disease, schizophrenia or psychosis, pain, addiction, a sleep disorder, a cognitive disorder (e.g. mild cognitive impairment), Parkinson’s Disease, Parkinson’s Disease-levodopa-induced dyskinesia, Huntington’s Disease, dyskinesia, dry mouth, pulmonary hypertension, chronic obstructive pulmonary disease (COPD), asthma, urinary incontinence, glaucoma, Trisomy 21 (Down Syndrome), cerebral amyloid angiopathy, dementia, Hereditary Cerebral Hemorrhage with Amyloidosis of the Dutch-Type (HCHWA-D), Creutzfeld-Jakob disease, prion disorders, amyotrophic lateral sclerosis, progressive supranuclear palsy, head trauma, stroke, pancreatitis, inclusion body myositis, other peripheral amyloidoses, diabetes, autism, tardive dyskines
- a cognitive disorder e.g.
- the term “about” includes and describes the value or parameter per se.
- “about x” includes and describes “x” per se.
- the term “about” when used in association with a measurement, or used to modify a value, a unit, a constant, or a range of values refers to variations of 4.10%.
- “about 2:8” in some embodiments includes 1.8-2.2:7 2-8.8.
- ambient temperature refers to the air temperature of any object or environment where the material is stored. In some embodiments, it also refers to an ordinary temperature or the baseline temperature. In some embodiments, it refers to the average temperature of an environment.
- a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof can also, in some embodiments of this disclosure, be introduced by rechargeable or biodegradable polymeric devices or other devices, e.g., patches and pumps, or in formulations, which provide extended, slow, or controlled release of a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof.
- the administration can also be performed, for example, once, a plurality of times, and/or over one or more extended periods.
- the administration includes direct administration, including self-administration, and indirect administration, including the act of prescribing a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof.
- a physician who instructs a patient to self-administer a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof, or to have a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof administered by another and/or who provides a patient with a prescription for a drug is administering a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof to the patient.
- Appropriate methods of administering a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof to a subject will also depend, for example, on the age of the subject, whether the subject is active or inactive at the time of administering, whether the subject is cognitively impaired at the time of administering, the extent of the impairment, and the chemical and biological properties of the crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof (e.g. solubility, digestibility, bioavailability, stability and toxicity).
- a therapeutically effective amount refers to that amount of a crystalline form of Compound A of this disclosure being administered, itself or as part of a pharmaceutical composition or in combination with one of more additional therapeutic agents, which will relieve to some extent one or more of the symptoms of the disorder being treated.
- a therapeutically effective amount refers to that amount which has the effect of relieving to some extent (or, for example, eliminating or reducing) one or more symptoms associated with the disease or disorder (e.g., a positive, negative, or cognitive symptom of schizophrenia; AD; or a psychotic symptom of AD).
- patient refers to warm-blooded animals such as, for example, pigs, cows, chickens, horses, guinea pigs, mice, rats, gerbils, cats, rabbits, dogs, monkeys, chimpanzees, and humans.
- treating means reversing, alleviating, inhibiting the progress of, or preventing, the disorder or condition to which such term applies, or one or more symptoms of such disorder or condition.
- treatment refers to the act of treating as “treating” is defined herein.
- treating also includes monotherapy, as well as adjunctive and neo-adjunctive therapy for the treatment of a subject.
- crystalline form refers to a solid-state form which consists of an orderly arrangement of structural units. Specifically, the use of the term “crystalline form” is intended to refer to a crystalline anhydrate or hydrate of Compound A.
- polymorph refers to a crystalline form of a compound in a particular crystal packing arrangement.
- polymorph includes pseudopolymorphs, which are typically different solvates of a material, and thus their properties differ from one another. Thus, each distinct polymorph and pseudopolymorph disclosed herein is considered to be a distinct single crystalline form herein.
- crystalline form and “polymorph” are used interchangeably unless the context indicates differently.
- Certain polymorphs may exhibit enhanced thermodynamic stability or may be more readily manufactured in high purity in large quantities, and thus be more suitable for inclusion in pharmaceutical formulations. Certain polymorphs may also display other advantageous physical properties such as lack of hygroscopic tendencies, improved solubility, and enhanced rates of dissolution due to different lattice energies.
- any particular peak in an XRPD pattern may appear as a singlet, doublet, triplet, quartet, or multiplet, depending on the type of instrument or the settings, the sensitivity of the instrument, measuring conditions, and/or purity of the crystalline form.
- any particular peak in an XRPD may appear in a symmetric shape or in an asymmetric shape, e.g., having a shoulder.
- instrument variation and other factors can affect the 2-theta values. A skilled artisan understanding these variations is capable of identifying or ascertaining the defining features or characteristics of a particular crystal form using XRPD, as well as using other known physicochemical techniques.
- characteristic peaks when referring to the peaks in an XRPD pattern of a crystalline form of Compound A of this disclosure refers to a collection of certain peaks whose values of 2 ⁇ across a range of 0° - 40° are, as a whole, uniquely assigned to one of the crystalline forms of Compound A.
- hydrate refers to a combination of water with a crystalline form of Compound A according to this disclosure, wherein the water is either absorbed, adsorbed or contained within a crystal lattice of the substrate Compound A.
- this disclosure is directed to crystalline forms of Compound A, .
- these forms in the various embodiments of this disclosure are designated Forms 1, 2, 3, and 4.
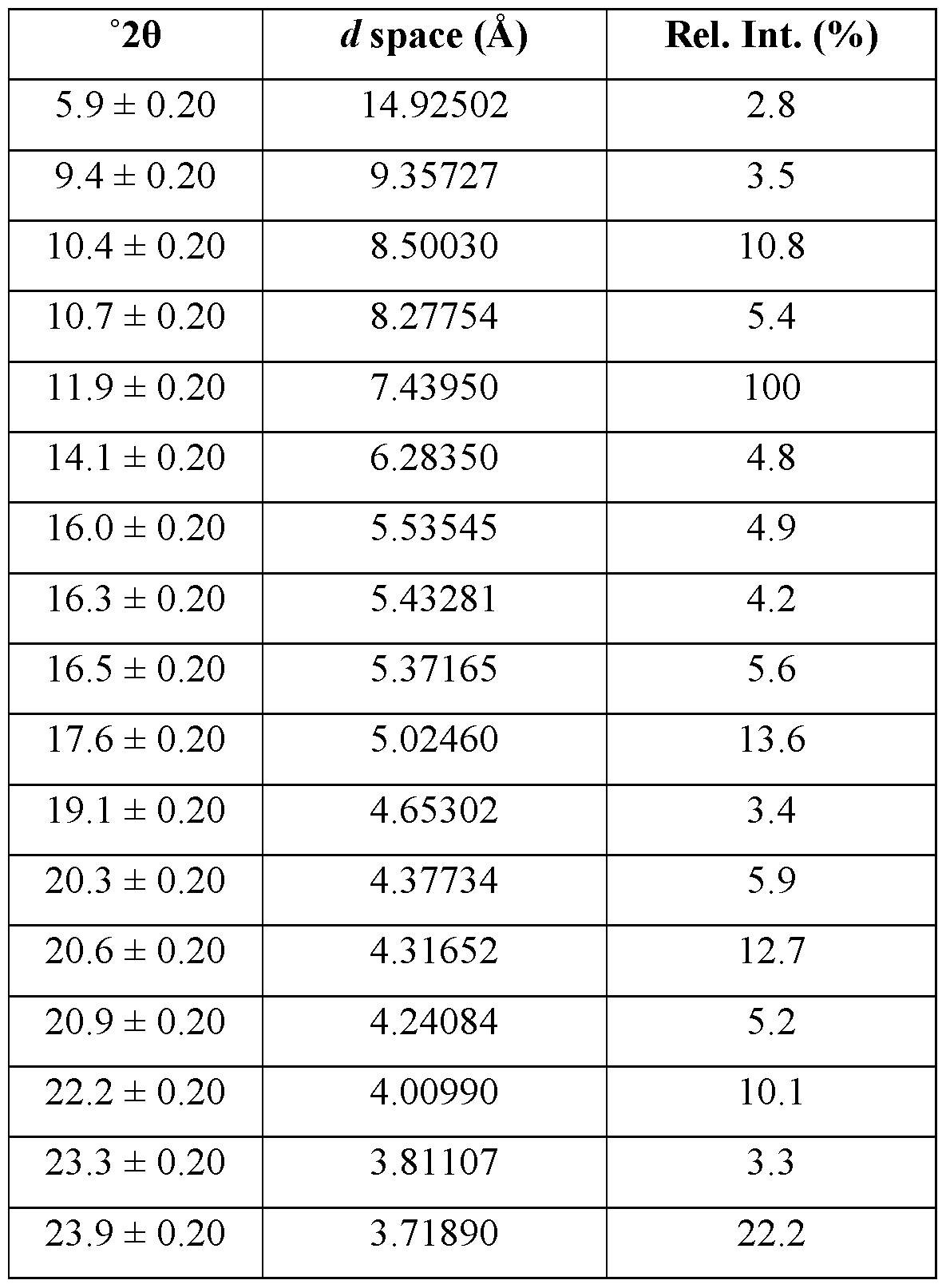
- the crystallized form of Compound A is Form 1. It is anhydrous.
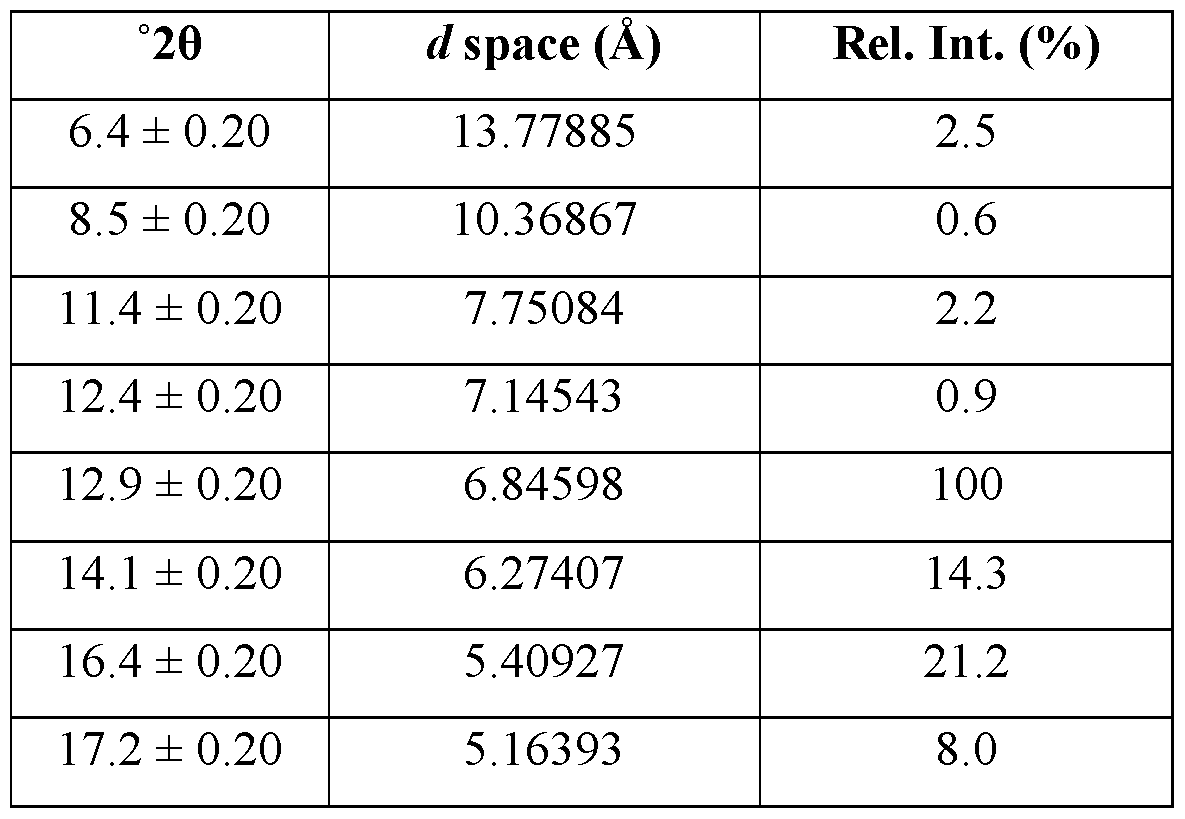
- the crystallized form of Compound A is Form 2. It is a monohydrate.
- the crystallized form of Compound A is Form 3. It is a dihydrate.
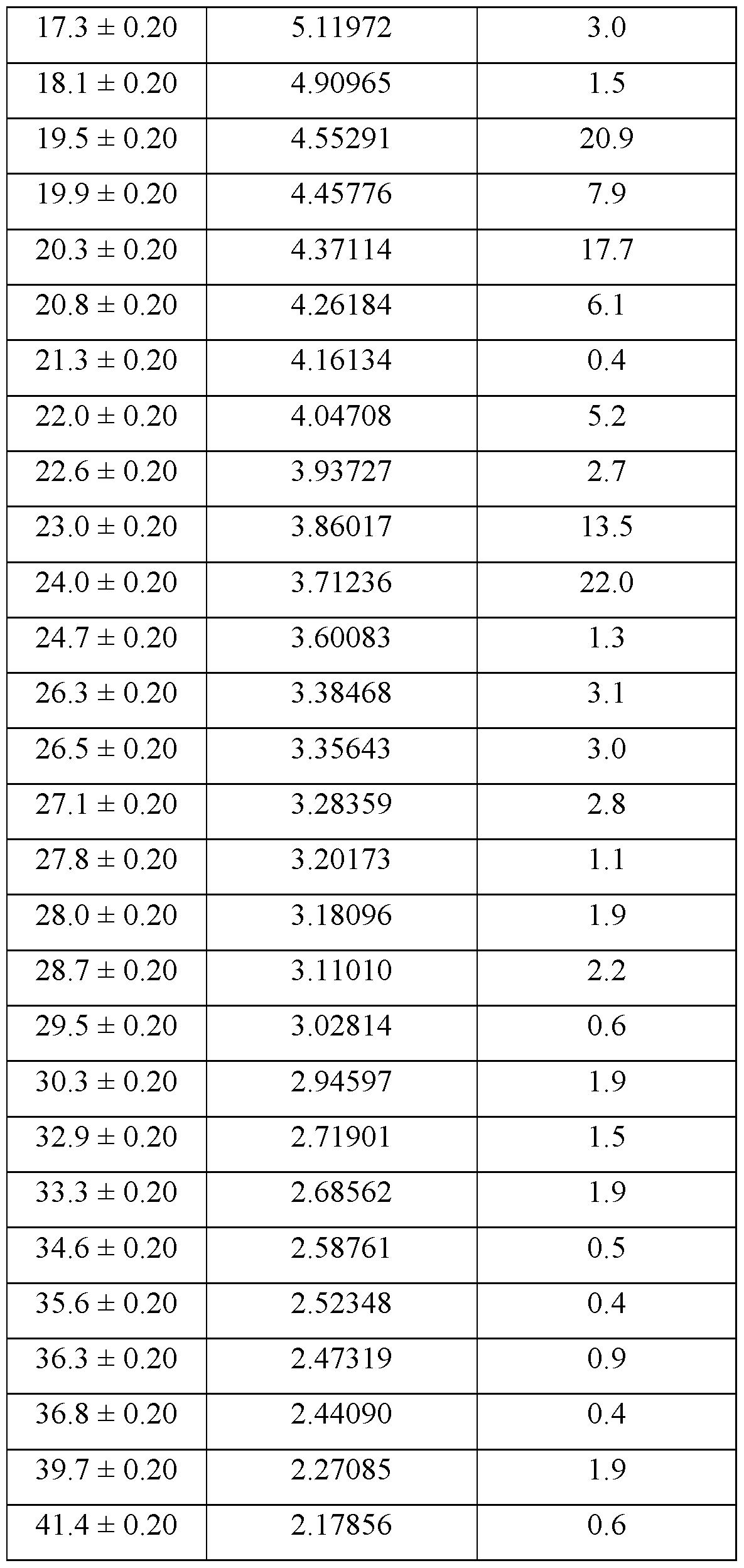
- the crystallized form of Compound A is Form 4. It is anhydrous.
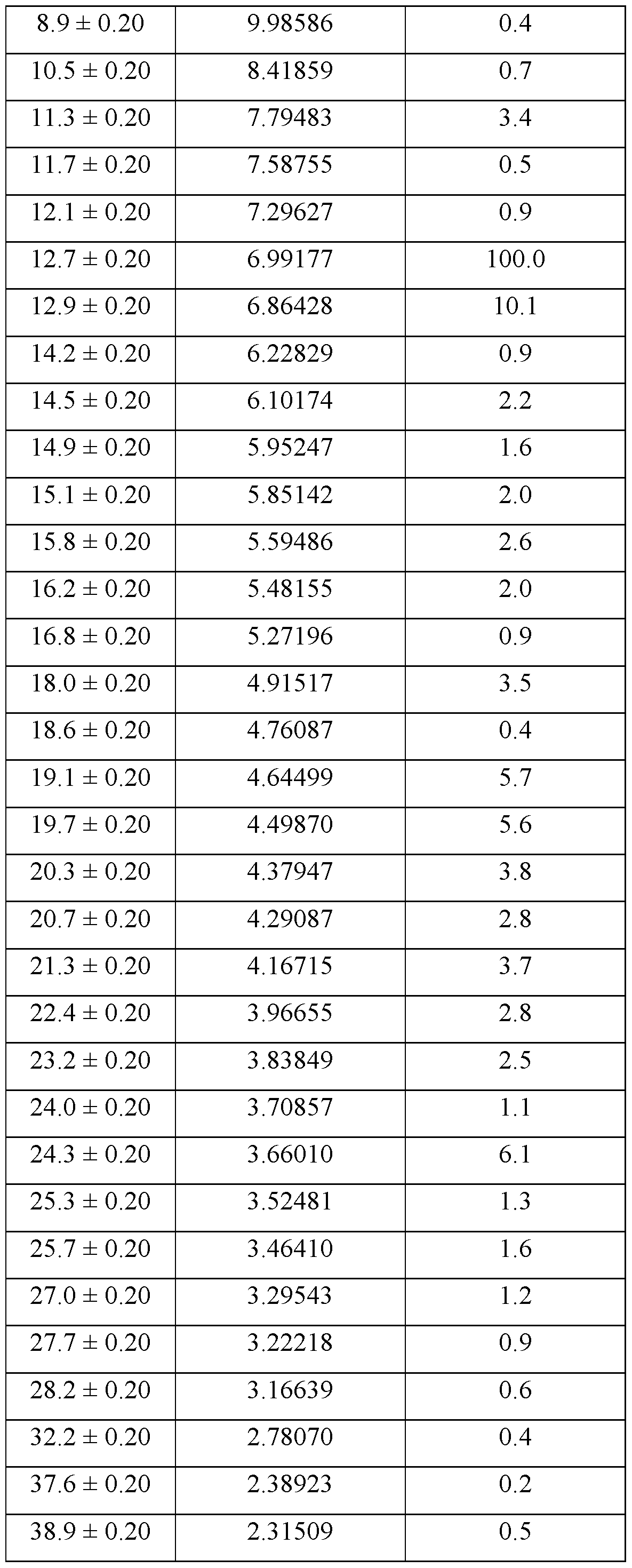
- the crystalline form of Compound A is Form 1, Form 1 being characterized by an XRPD pattern comprising peaks at 12.7, 12.9, 19.1, 19.7, and 24.3 ⁇ 0.2 degrees 2 ⁇ .
- Form 1 is characterized by an XRPD pattern comprising one or more peaks selected from the group consisting of peaks at 12.7, 12.9, 19.1, 19.7, and 24.3 ⁇ 0.2 degrees 2 ⁇ .
- the XRPD pattern further comprises one or more peaks selected from the group consisting of peaks at 11.3, 18.0, 20.3, and 21.3 ⁇ 0.2 degrees 2 ⁇ .
- Form 1 of Compound A is characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 1; b. a TGA thermogram substantially as set forth in Figure 2; c. a DSC thermogram substantially as set forth in Figure 3; d. a DVS plot substantially as set forth in Figure 4; e. a FT Raman spectrum substantially as set forth in Figure 5; and f. a PLM image substantially as set forth in Figure 6.
- Form 1 of Compound A is characterized by a TGA thermogram showing ⁇ 1% weight loss below about 200 °C.
- Form 1 of Compound A is characterized by a DSC thermogram having an endotherm with an onset of about 183 °C.
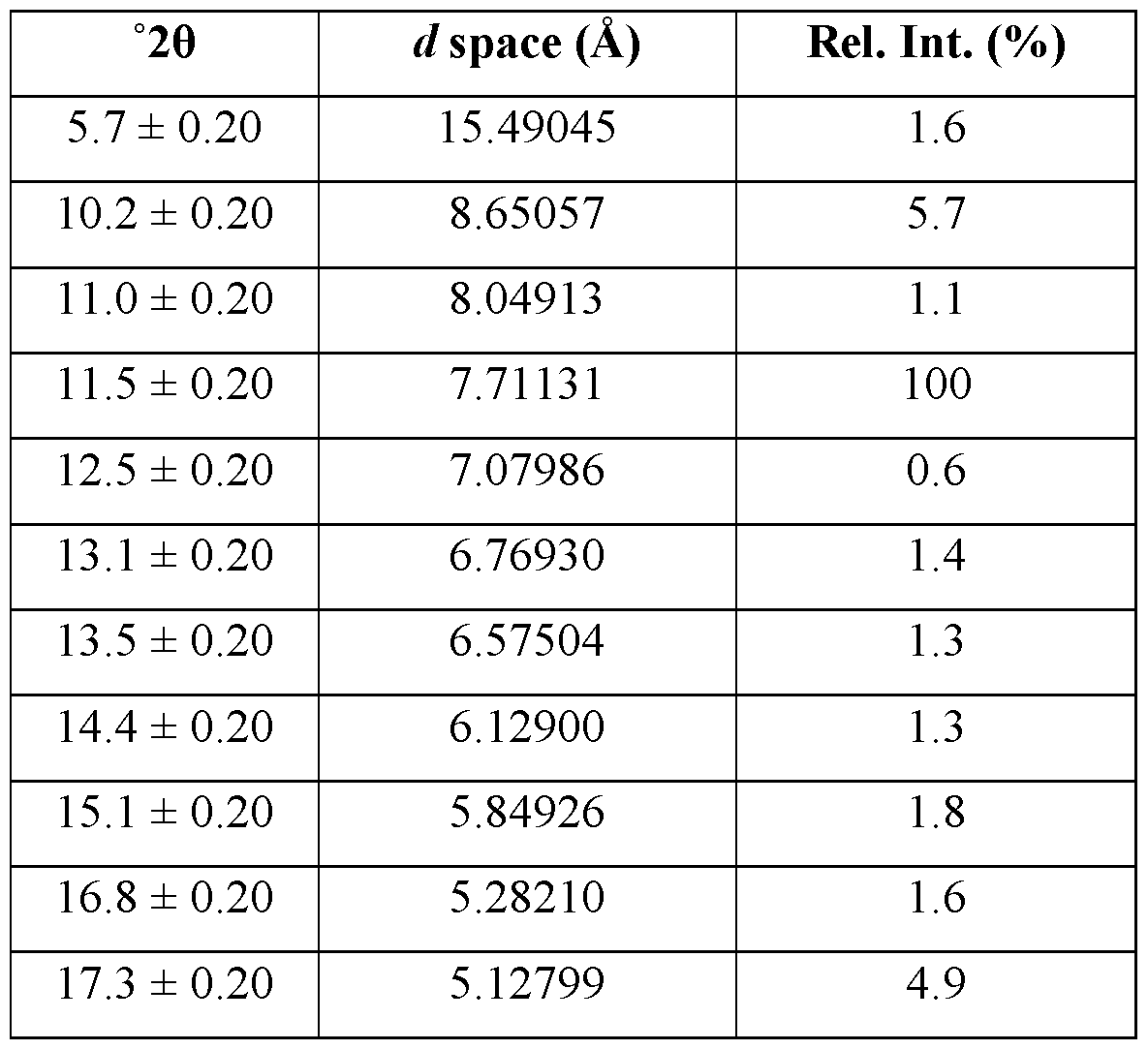
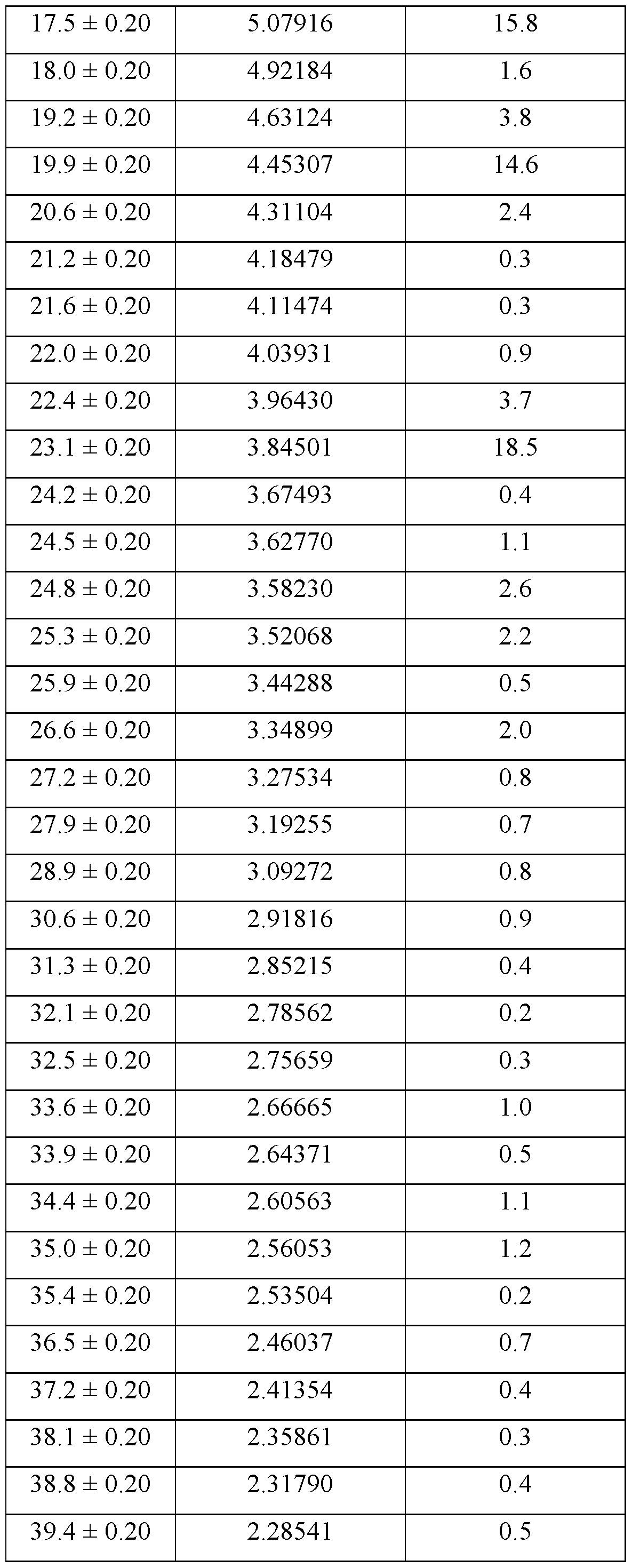
- the crystalline form of Compound A is Form 2, Form 2 being characterized by an XRPD pattern comprising peaks at 10.2, 11.5, 17.5, 19.9, and 23.1 ⁇ 0.2 degrees 2 ⁇ .
- Form 2 is characterized by an XRPD pattern comprising one or more peaks selected from the group consisting of peaks at 10.2, 11.5, 17.5, 19.9, and 23.1 ⁇ 0.2 degrees 2 ⁇ .
- the XRPD pattern further comprises one or more peaks selected from the group consisting of peaks at 17.3, 19.2, 20.6, 22.4, 24.8, 25.3, and 26.6 ⁇ 0.2 degrees 2 ⁇ .
- Form 2 of Compound A is characterized by a TGA thermogram showing about 8% weight loss below about 200 °C.
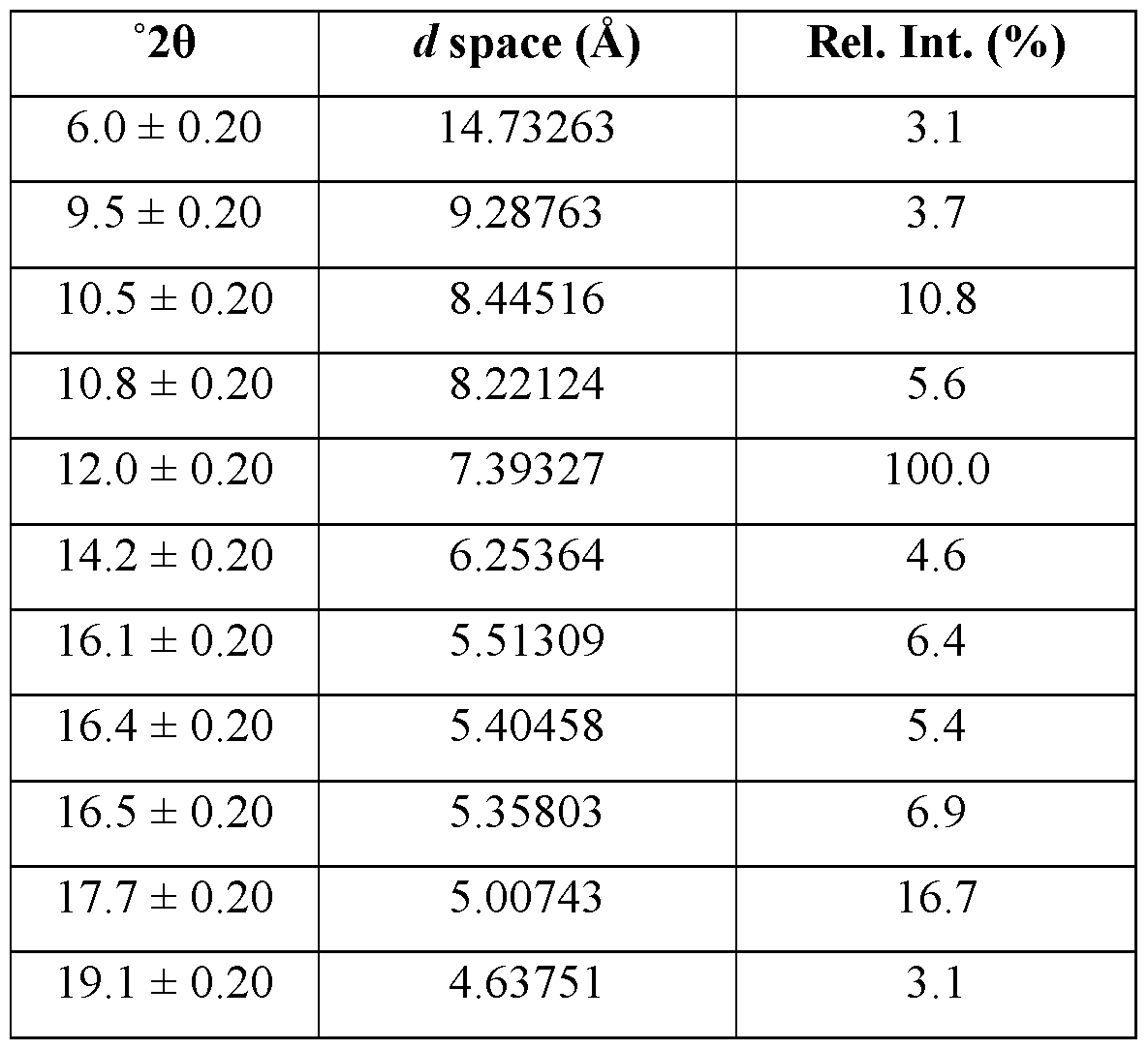
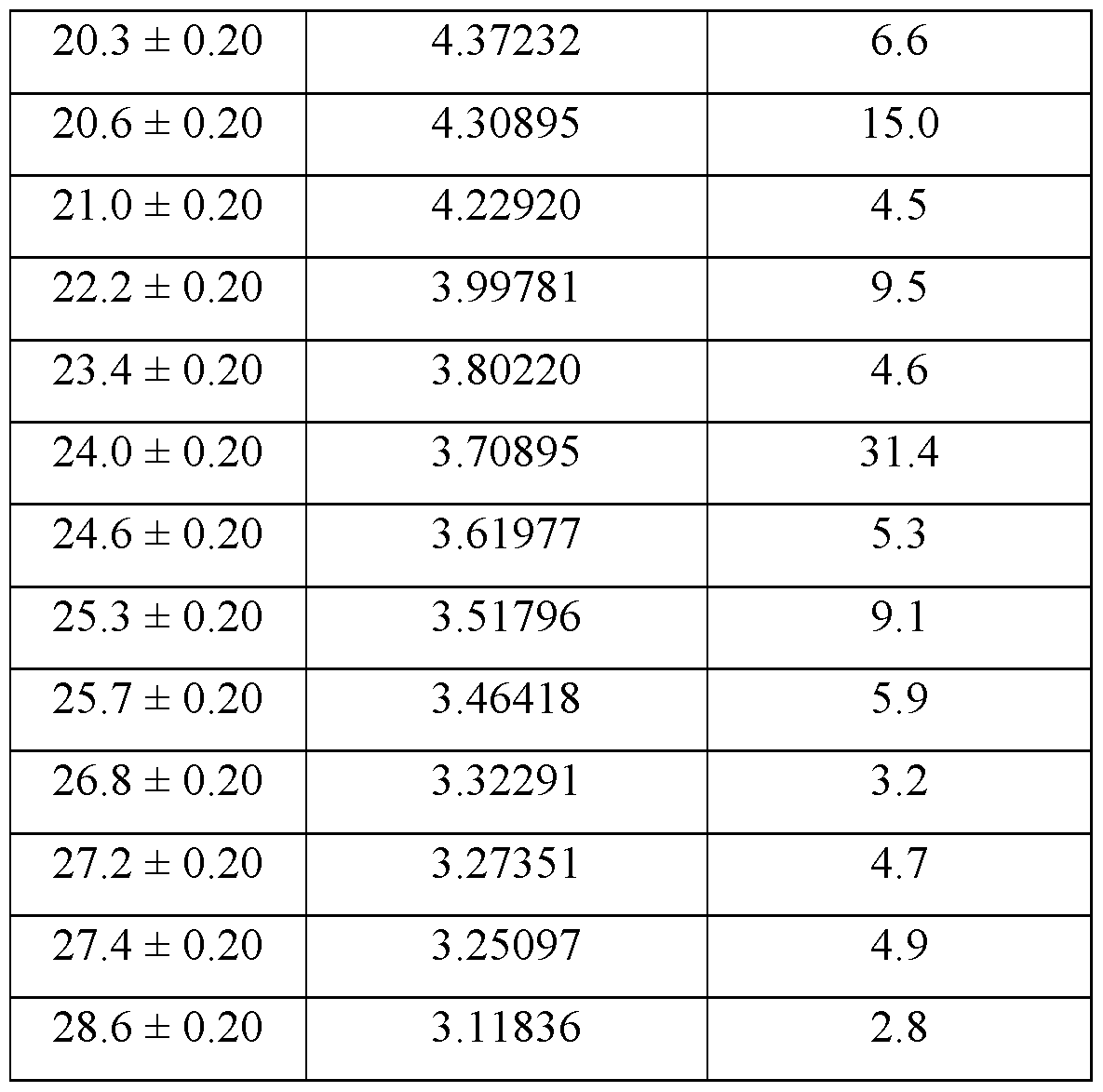
- the crystalline form of Compound A is Form 3, Form 3 being characterized by an XRPD pattern comprising peaks at 10.5, 12.0, 17.7, 20.6, and 24.0 degrees 2 ⁇ ⁇ 0.2 degrees 2 ⁇ .
- Form 3 is characterized by an XRPD pattern comprising one or more peaks selected from the group consisting of peaks at 10.5, 12.0, 17.7, 20.6, and 24.0 degrees 2 ⁇ ⁇ 0.2 degrees 2 ⁇ .
- the XRPD pattern further comprises one or more peaks selected from the group consisting of peaks at 10.8, 16.1, 16.4, 20.3, 22.2, 24.6, 25.3, and 25.7 ⁇ 0.2 degrees 2 ⁇ .
- Form 3 of Compound A is characterized by a DSC thermogram having a broad first endotherm with an onset of about 80 °C followed by a second endotherm with an onset of about 182 °C, and a third endotherm with an onset of about 193 °C.
- Form 3 of Compound A is characterized by a higher thermodynamic stability as compared to Forms 1, 2, and 4 of Compound A.
- Form 3 of Compound A also has limited hygroscopicity, preserving crystallinity, potency, and ease of handling.
- Form 4 of Compound A is characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 15; b. a TGA thermogram substantially as set forth in Figure 16; c. a DSC thermogram substantially as set forth in Figure 17; d. a DVS plot substantially as set forth in Figure 18; e. a FT Raman spectrum substantially as set forth in Figure 19; and f. a PLM image substantially as set forth in Figure 20.
- Form 4 of Compound A is characterized by a TGA thermogram showing ⁇ 1% weight loss below about 200 °C.
- Form 4 of Compound A is characterized by a DSC thermogram having a sharp endotherm with an onset of about 191 °C.
- this disclosure provides pharmaceutical compositions that comprise one of more of a crystalline form of Compound A and a pharmaceutically acceptable carrier, vehicle, or diluent.
- the pharmaceutically acceptable carriers of the pharmaceutical compositions of this disclosure include, but are not limited to ion exchangers, alumina, aluminum stearate, lecithin, serum proteins, such as human serum albumin, buffer substances such as phosphates, glycine, sorbic acid, potassium sorbate, partial glyceride mixtures of saturated vegetable fatty acids, water, salts or electrolytes, such as protamine sulfate, disodium hydrogen phosphate, potassium hydrogen phosphate, sodium chloride, zinc salts, colloidal silica, magnesium trisilicate, polyvinyl pyrrolidone, cellulose-based substances, polyethylene glycol, sodium carboxymethylcellulose, polyacrylates, waxes, polyethylene-polyoxypropylene-block polymers, polyethylene glycol and
- aqueous and non-aqueous carriers examples include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, and injectable organic esters, such as ethyl oleate.
- polyols such as glycerol, propylene glycol, polyethylene glycol, and the like
- vegetable oils such as olive oil
- injectable organic esters such as ethyl oleate.
- Proper fluidity can be maintained, for example, by the use of coating materials, such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants.
- no carrier is used.
- the pharmaceutical compositions described herein may be formulated in any convenient way for use in human medicine.
- acetylcholinesterase inhibitors such as donepezil hydrochloride (ARICEPT, MEMAC), physostigmine salicylate (ANTILIRIUM), physostigmine sulfate (ESERINE), metrifonate, neostigmine, ganstigmine, pyridostigmine (MESTINON), ambenonium (MYTELASE), demarcarium, Debio 9902 (also known as ZT-1; Debiopharm), rivastigmine (EXELON), ladostigil, NP-0361, galantamine hydrobromide (RAZADYNE, RIMINYL, NIVALIN), tacrine (COGNEX), tolserine, velnacrine maleate, memoquin, huperzine A (HUP- A; NeuroHitech), phenserine, edrophonium (ENLON, TENSILON), and INM-176;
- amyloid-B (or fragments thereof), such as A ⁇ 1-15 conjugated to pan HLA DR-binding epitope (PADRE), ACC-001 (Elan/Wyeth), ACL01, ACI-24, AN- 1792, Affitope AD-01, CAD 106, and V-950;
- antibodies to amyloid-B such as ponezumab, solanezumab, bapineuzumab (also known as AAB-001), AAB-002 (Wyeth/Elan), ACI-01-Ab7, BAN-2401, intravenous Ig (GAMMAGARD), LY2062430 (humanized m266; Lilly), R1450 (Roche), ACU- 5A5, huC091, and those disclosed in International Patent Publication Nos WO04/032868, W005/025616, W006/036291, W006/069081, WO06/118959, in US Patent Publication Nos US2003/0073655, US2004/0192898, US2005/0048049, US2005/0019328, in European Patent Publication Nos EP0994728 and 1257584, and in US Patent No 5,750,349;
- amyloid-lowering or -inhibiting agents including those that reduce amyloid production, accumulation and fibrillization
- amyloid-lowering or -inhibiting agents such as dimebon, davunetide, eprodisate, leuprolide, SK-PC- B70M, celecoxib, lovastatin, anapsos, oxiracetam, pramiracetam, varenicline, nicergoline, colostrinin, bisnorcymserine (also known as BNC), NIC5-15 (Humanetics), E-2012 (Eisai), pioglitazone, clioquinol (also known as PBT1), PBT2 (Prana Biotechnology), flurbiprofen (ANS AID, FROBEN) and its R-enantiomer tarenflurbil (FLURIZAN), nitroflurbiprofen, fenoprofen (FENOPRON, NALFON), ibuprofen
- anticonvulsants such as carbamazepine (TEGRETOL, CARBATROL), oxcarbazepine (TRILEPTAL), phenytoin sodium (PHENYTEK), fosphenytoin (CEREBYX, PRODILANTIN), divalproex sodium (DEPAKOTE), gabapentin (NEURONTIN), pregabalin (LYRICA), topirimate (TOPAMAX), valproic acid (DEPAKENE), valproate sodium (DEPACON), 1- benzyl-5-bromouracil, progabide, beclamide, zonisamide (TRERIEF, EXCEGRAN), CP- 465022, retigabine, talampanel, and primidone (MYSOLINE);
- dopamine receptor agonists such as apomorphine (APOKYN), bromocriptine (PARLODEL), cabergoline (DOSTINEX), dihydrexidine, dihydroergocryptine, fenoldopam (CORLOPAM), lisuride (DOPERGIN), terguride spergolide (PERMAX), piribedil (TRIVASTAL, TRASTAL), pramipexole (MIRAPEX), quinpirole, ropinirole (REQUIP), rotigotine (NEUPRO), SKF-82958 (GlaxoSmithKline), cariprazine, pardoprunox and sarizotan;
- APIOKYN apomorphine
- PARLODEL bromocriptine
- DOSTINEX cabergoline
- dihydrexidine dihydroergocryptine
- CORLOPAM fenoldopam
- DOPERGIN terguride spergolide
- PERMAX
- dopamine receptor antagonists such as chlorpromazine, fluphenazine, haloperidol, loxapine, risperidone, thioridazine, thiothixene, trifluoperazine, tetrabenazine (NITOMAN, XENAZINE), 7-hy droxyam oxapine, droperidol (INAPSINE, DRIDOL, DROPLETAN), domperidone (MOTILIUM), L-741742, L-745870, raclopride, SB-277011 A, SCH-23390, ecopipam, SKF-83566, and metoclopramide (REGLAN);
- dopamine reuptake inhibitors such as bupropion, safinamide, nomifensine maleate (MERIT AL), vanoxerine (also known as GBR-12909) and its decanoate ester DBL-583, and aminep
- GABA gamma-amino-butyric acid receptor agonists
- baclofen LIORESAL, KEMSTRO
- siclofen pentobarbital
- GBRENE progabide
- clomethi azole gamma-amino-butyric acid receptor agonists
- histamine 3 (H3) antagonists such as ciproxifan, tiprolisant, S-38093, irdabisant, pitolisant, GSK-239512, GSK-207040, JNJ-5207852, JNJ-17216498, HPP-404, SAR-110894, trans-A-ethyl-3-fluoro-3-[3-fluoro-4-(pyrrolidin-l-ylmethyl)phenyl]-cyclobutanecarboxamide (PF-3654746 and those disclosed in US Patent Publication Nos US2005-0043354, US2005- 0267095, US2005-0256135, US2008-0096955, US2007-1079175, and US2008-0176925;
- immunosuppressants such as methotrexate (TREXALL, RHEUMATREX), mitoxantrone (NOVANTRONE), mycophenolate mofetil (CELLCEPT), mycophenolate sodium (MYFORTIC), azathioprine (AZASAN, IMURAN), mercaptopurine (PURI-NETHOL), cyclophosphamide (NEOSAR, CYTOXAN), chlorambucil (LEUKERAN), cladribine (LEUSTATIN, MYLINAX), alpha-fetoprotein, etanercept (ENBREL), and 4-(benzyloxy)-5-[(5- undecyl-2H-pyrrol-2-ylidene)methyl]- 1H, 1’H-2,2’ -bipyrrole (also known as PNU-156804);
- TREXALL methotrexate
- RHEUMATREX mitoxantrone
- NOVANTRONE mycophenolate mofetil
- interferons including interferon beta-la (AVONEX, REBIF) and interferon beta-lb (BETASERON, BETAFERON);
- muscarinic receptor particularly Ml subtype agonists, such as cevimeline, levetiracetam, bethanechol chloride (DUVOID, URECHOLINE), itameline, pilocarpine (S ALAGEN), NGX267, arecoline, L-687306 (Merck), L-689660 (Merck), furtrethonium iodide (FURAMON, FURANOL), furtrethonium benzensulfonate, furtrethonium p-toluenesulfonate, McN-A-343, oxotremorine, sabcomeline, AC-90222 (Acadia Pharmaceuticals), and carbachol (CARBASTAT, MIOSTAT, CARBOPTIC);
- Ml subtype agonists such as cevimeline, levetiracetam, bethanechol chloride (DUVOID, URECHOLINE), itameline, pilocarpine (S ALAGEN), NGX267, areco
- norepinephrine (noradrenaline) reuptake inhibitors such as atomoxetine (STRATTERA), doxepin (APONAL, ADAPIN, SINEQUAN), nortriptyline (AVENTYL, PAMELOR, NORTRILEN), amoxapine (ASENDIN, DEMOLOX, MOXIDIL), reboxetine (EDRONAX, VESTRA), viloxazine (VIVALAN), maprotiline (DEPRILEPT, LUDIOMIL, PSYMION), bupropion (WELLBUTRIN), and radaxafine;
- atomoxetine STRATTERA
- doxepin APIONAL, ADAPIN, SINEQUAN
- nortriptyline AVENTYL, PAMELOR, NORTRILEN
- amoxapine ASENDIN, DEMOLOX, MOXIDIL
- reboxetine EDRONAX, VESTRA
- viloxazine
- PDE3 inhibitors e.g., anagrelide, cilostazol, milrinone, olprinone, parogrelil, and pimobendan
- PDE4 inhibitors e.g., apremilast, ibudilastroflumilast, rolipram, Ro 20-1724, ibudilast (KETAS), piclamilast (also known as RP73401), CDP840, cilomilast (ARIFLO), roflumilast, tofimilast, oglemilast (also known as GRC 3886), tetomilast (also known as OPC-6535), lirimifast, theophylline (UNIPHYL, THEOLAIR), arofylline (also known as LAS-31025), doxofylline, RPR-122818, or mesembrine), and (e) PDE5 inhibitors (e.g., sil
- (xxxv) serotonin (5-HT) reuptake inhibitors such as alaproclate, citalopram (CELEXA, CIPRAMIL), escitalopram (LEXAPRO, CIPRALEX), clomipramine (ANAFRANIL), duloxetine (CYMBALTA), femoxetine (MALEXIL), fenfluramine (PONDIMIN), norfenfluramine, fluoxetine (PROZAC), fluvoxamine (LUVOX), indalpine, milnacipran (IXEL), paroxetine (PAXIL, SEROXAT), sertraline (ZOLOFT, LUSTRAL), trazodone (DESYREL, MOLIPAXIN), venlafaxine (EFFEXOR), zimelidine (NORMUD, ZELMID), bicifadine, desvenlafaxine (PRISTIQ), brasofensine, vilazodone, cariprazine, 29eurals
- AMPA-type glutamate receptor modulators such as perampanel, mibampator, selurampanel, GSK-729327, N- ⁇ (3S,4S)-4-[4-(5-cyanothiophen-2-yl)phenoxy]tetrahydro-furan- 3-yl ⁇ propane-2-sulfonamide, and the like;
- Interleukin- 1 receptor-associated kinase 4 inhibitors such as, but not limited to, PF-06650833;
- (xli) antidepressants including, but not limited to:
- NDRIs norepinephrine-dopamine reuptake inhibitors
- buproprion WELLBUTRIN, ELONTRIL
- SSRIs selective serotonin reuptake inhibitors
- CELEXA CIPRAMIL
- escitalopram LEXAPRO, CIPRALEX
- fluoxetine PROZAC, SARAFEM
- fluvoxamine LVOX, FAVERIN
- paroxetine PAXIL, SEROXAT
- sertraline ZOLOFT, LUSTRAL
- TCAs tricyclic antidepressants
- TAAs such as, amitriptyline (ELAVIL, ENDEP), amitriptylinoxide (AMIOXID, AMBIVALON, EQUILIBRIN), clomipramine (ANAFRANIL), desipramine (NORPRAMIN, PERTOFRANE), dibenzepin (NOVERIL, VICTORIL), dimetacrine (ISTONIL), dosulepin (PROTHIADEN), doxepin (ADAPIN, SINEQUAN), imipramine (TOFRANIL), lofepramine (LOMONT, GAMANIL), melitracen (DIXERAN, MELIXERAN, TRAUSABUN), nitroxazepine (SINTAMIL), nortriptyline (PAMELOR, AVENTYL), noxiptiline (SINTAMIL), opipramol (INSIDON), pipof ezine (AZAFEN/AZAPHEN), protripty
- the one or more additional therapeutic agents are neuropsychiatric agents (e.g., antipsychotics, including atypical antipsychotics, antischizophrenia agents, and/or antidepressants).
- neuropsychiatric agents e.g., antipsychotics, including atypical antipsychotics, antischizophrenia agents, and/or antidepressants.
- the one or more additional therapeutic agents are antipsychotics, such as risperidone, olanzapine, clozapine, quetiapine, aripiprazole, ziprasidone, paliperidone, asenapine, iloperidone, or bexpiprazole.
- the additional therapeutic agent is bexpiprazole.
- the one or more additional therapeutic agents are antischizophrenia agents, such as ziprasidone, risperidone, olanzapine, quetiapine, aripiprazole, asenapine, blonanserin, or iloperidone, wherein the amounts of the active agent and the combination when taken as a whole are therapeutically effective for treating schizophrenia.
- the one or more additional therapeutic agents are selected from antidepressants, such as the MAOIs, NDRIs, SARIs, SNRIs, SSRIs, TCAs, or TeCAs described herein above.
- the one or more additional therapeutic agents are agents that are able to treat Alzheimer’s Disease and/or psychosis associated with it.
- the one or more additional therapeutic agents are acetylcholinesterase inhibitors and/or muscarinic receptor agonists, partial agonists, or positive allosteric modulators, such as xanomeline, donepezil, galantamine, or rivastigmine.
- the crystalline forms of Compound A and pharmaceutical compositions of them, and the one or more additional therapeutic agents are formulated separately.
- the crystalline forms of Compound A and pharmaceutical compositions of them, and the one or more additional therapeutic agents are packaged together.
- the crystalline forms of Compound A and pharmaceutical compositions of them, and the one or more additional therapeutic agents are packaged separately.
- the crystalline forms of Compound A and pharmaceutical compositions of them, and the one or more additional therapeutic agents are formulated together.
- the crystalline forms of Compound A and pharmaceutical compositions of them, and the one or more additional therapeutic agents in a combination (where they are not formulated together) of this disclosure are administered simultaneously (i.e., with a time separation of no more than about 15 minutes and in some embodiments no more than 10 minutes) or sequentially (i.e., with a time separation of more than about 15 minutes and in some embodiments more than one hour or up to 12-24 hours).
- this disclosure provides pharmaceutical compositions comprising Compound A and one or more pharmaceutically acceptable carriers or excipients.
- the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components are formulated in a solid form such as a capsule, tablet, pill, lozenge, cachet, powder, troche, wafer, or granule.
- the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components may be mixed with one or more pharmaceutically acceptable carriers, such as sodium citrate or dicalcium phosphate, and/or any of the following: (1) fillers or extenders, such as starches, lactose, sucrose, glucose, mannitol, and/or silicic acid; (2) binders, such as, for example, carboxymethylcellulose, alginates, gelatin, polyvinyl pyrrolidone, sucrose, and/or acacia; (3) humectants, such as glycerol; (4) disintegrating agents, such as agar-agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates, and sodium carbonate; (5) solution retarding agents, such as paraffin; (6) absorption accelerators, such as quaternary ammonium compounds; (7) wetting agents, such as, for example, cety
- compositions or combinations according to this disclosure may be mixed with one or more excipients, such as water, saline, dextrose, glycerol, ethanol, or the like and combinations thereof.
- excipients such as water, saline, dextrose, glycerol, ethanol, or the like and combinations thereof.
- the pharmaceutical compositions or combinations (or components thereof) of the disclosure may also comprise buffering agents.
- Solid pharmaceutical compositions or combinations (or components thereof) of a similar type may also be employed as fillers in soft and hard-filled gelatin capsules using such excipients as lactose or milk sugars, as well as high molecular weight polyethylene glycols and the like.
- the pharmaceutical composition or solid formulation of the present invention may be manufactured by a conventional dry granulation manufacturing process that includes blending, milling, blend lubrication, roller compaction and milling, blend lubrication, compression, and aqueous based film coating using equipment commonly available in the pharmaceutical industry.
- the pharmaceutical composition comprises a core and a coating, wherein the core comprises Compound A, or a pharmaceutically acceptable salt or hydrate thereof, and one or more pharmaceutically acceptable excipients, and the coating comprises an opacifier.
- the pharmaceutical composition is a tablet dosage form.
- the core comprises one or more fillers or extenders, binders, humectants, disintegrating agents, and lubricants.
- the pharmaceutical composition comprises a crystalline form of Compound A, including but not limited to Form 1, Form, 2, Form 3, or Form 4 described herein.
- the pharmaceutical composition comprises about 10 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 87 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
- the pharmaceutical composition comprises about 30 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 85 weight percent to about 93 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
- the opacifier comprises from about 4 weight percent to about 20 weight percent of the coating by weight, based upon total weight of the coating composition. In an embodiment, the opacifier comprises about 5 weight percent to about 20 weight percent, of the coating by weight, based upon total weight of the coating composition. In an embodiment, the opacifier comprises about 5 weight percent to about 15 weight percent, or about 10 weight percent to about 15 weight percent, or about 15 weight percent to about 20 weight percent of the coating by weight, based upon total weight of the coating composition. In an embodiment, the opacifier comprises about 6 weight percent, about 14 weight percent, or about 17 weight percent of the coating by weight, based upon total weight of the coating composition.
- kits that are suitable for use in performing the methods of treatment herein.
- the kit contains one of more dosage forms comprising one or more of the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, and one or more containers for the dosage forms, in quantities sufficient to carry out the methods of this disclosure.
- the methods of the present disclosure are directed to a method of producing Form 1 of Compound A, the method comprising slurrying the compound in a solvent selected from acetone, acetonitrile, 1 -butanol, 1,4-di oxane, dimethylformamide, ethanol, ethyl acetate, ethylene glycol, methyl ethyl ketone, methanol, methyl isobutyl ketone, 1- propanol, 2-propanol, water, or a mixture of acetonitrile-water at ambient temperature, 5 °C or 50 °C.
- a solvent selected from acetone, acetonitrile, 1 -butanol, 1,4-di oxane, dimethylformamide, ethanol, ethyl acetate, ethylene glycol, methyl ethyl ketone, methanol, methyl isobutyl ketone, 1- propanol, 2-propanol, water
- the compound is slurried in a solvent selected from acetone, acetonitrile, 1-butanol, 1,4-dioxane, dimethylformamide, ethanol, ethyl acetate, ethylene glycol, methyl ethyl ketone, methanol, methyl isobutyl ketone, 1 -propanol, 2-propanol, water at ambient temperature.
- the compound is slurried in a solvent selected from 1-butanol, ethanol, ethylene glycol, methanol, 1 -propanol, or 2-propanol, at ambient temperature.
- the present disclosure is directed to a method of producing Form 1 of Compound A, the method comprising the steps of: (i) dissolving the compound in a 2- propanol at 65-75 °C; (ii) cooling the mixture to ambient temperature; (iii) filtering the slurry; and (iv) drying the collected solids.
- this disclosure provides a method of producing a monohydrate crystalline form of Compound A (e.g., Form 2), the method comprising slurrying the compound in a mixture selected from: (a) acetonitrile-water at ambient temperature, wherein the mixture comprises about 99.7 % of acetonitrile by volume; (b) acetonitrile-water at 5 °C, wherein the mixture comprises about 99.3% of acetonitrile by volume; or (c) acetonitrile-water at 50 °C, wherein the mixture comprises about 97.2% of acetonitrile by volume.
- a mixture selected from: (a) acetonitrile-water at ambient temperature, wherein the mixture comprises about 99.7 % of acetonitrile by volume; (b) acetonitrile-water at 5 °C, wherein the mixture comprises about 99.3% of acetonitrile by volume; or (c) acetonitrile-water at 50 °C, where
- the compound is dissolved in acetonitrile followed by a slow evaporation. In another embodiment, the compound is dissolved in isopropyl alcohol or acetone followed by a slow evaporation. In another embodiment, the compound is dissolved in ethyl acetate followed by a slow evaporation.
- the compound slurried in the acetonitrile-water mixture at ambient temperature comprises about 99.7% of acetonitrile by volume.
- the compound slurried in the acetonitrile-water mixture at 5 °C comprises about 99.3% of acetonitrile by volume.
- the compound slurried in the acetonitrile-water mixture at 50 °C comprises about 97.2% of acetonitrile by volume.
- the disclosure provides a method of producing a dihydrate crystalline form, Form 3, of Compound A the method comprising slurrying the compound in a solvent selected from 2-methyl tetrahydrofuran, tetrahydrofuran, water, toluene at ambient temperature, 5 °C or 50 °C.
- the disclosure provides a method of producing a dihydrate crystalline form, Form 3, of Compound A the method comprising slurrying the compound in a mixture selected from: (a) acetonitrile-water at ambient temperature, wherein the mixture comprises about 38.4%, 79.2%, 92.1%, 95.6%, 97.2%, 98.1%, 98.8% of acetonitrile by volume; (b) acetonitrile-water at 5°C, wherein the mixture comprises about 79.2% or 97.2% of acetonitrile by volume; or (c) acetonitrile-water at 50 °C, wherein the mixture comprises about 79.2% of acetonitrile by volume.
- a mixture selected from: (a) acetonitrile-water at ambient temperature, wherein the mixture comprises about 38.4%, 79.2%, 92.1%, 95.6%, 97.2%, 98.1%, 98.8% of acetonitrile by volume; (b
- the compound is slurried in a solvent selected from 2-methyl tetrahydrofuran, tetrahydrofuran, water, or toluene at ambient temperature.
- the compound is slurried in 2-methyl tetrahydrofuran or tetrahydrofuran at ambient temperature.
- the compound is slurried in water at ambient temperature.
- the compound is slurried in toluene at ambient temperature.
- the compound is slurried in water at 50 °C.
- the compound is slurried in water at 5 °C.
- the compound is slurried in water at 50 °C and the slurry is filtered after a week.
- the compound is slurried in an acetonitrile-water mixture at ambient temperature wherein the acetonitrile-water mixture comprises about 38.4%, 79.2%, 92.1%, 95.6%, 97.2%, 98.1%, 98.8% of acetonitrile by volume.
- the acetonitrile-water mixture comprises about 38.4% of acetonitrile by volume.
- the acetonitrile-water mixture comprises about 79.2% of acetonitrile by volume.
- the acetonitrile-water mixture comprises about 92.1% of acetonitrile by volume.
- the acetonitrile-water mixture comprises about 79.2% of acetonitrile by volume. In another embodiment, the acetonitrile-water mixture comprises about 97.2% of acetonitrile by volume. In another embodiment, the compound slurried in the acetonitrile-water mixture at 50 °C comprises about 79.2% of acetonitrile by volume.
- the present disclosure is directed to a method of producing Form 3 of Compound A, the method comprising the steps of (i) dissolving the compound in a 2- propanol at 65-75 °C; (ii) cooling the mixture to 50-55 °C; (iii) slowly adding water; (iv) adding a seed crystal; (v) slowly adding water; (vi) cooling the mixture to ambient temperature; (vii) filtering the slurry; and (viii) drying the collected solids.
- the disclosure provides a method of producing an anhydrous crystalline form, Form 4, of Compound A the method comprising slurrying the compound in a mixture of acetonitrile-water at 50 °C, wherein the mixture comprises about 79.2% of acetonitrile by volume.
- the present disclosure is directed to a method of producing Form 4 of Compound A, the method comprising the steps of (i) dissolving the compound in a 2- propanol at 70-80 °C; (ii) cooling the mixture to 45-50 °C; (iii) filtering the slurry; and (iv) drying the collected solids.
- the methods described herein may be adapted and modified as is appropriate for the application being addressed and that the methods described herein may be employed in other suitable applications, and that such other additions and modifications will not depart from the scope hereof.
- the sample was prepared on a polished, zerobackground sample holder by gently pressing onto the flat surface. Measurements were carried out from 3 to 42° (2 ⁇ ), at a scanning speed of 0.05 s/step and step size of 0.02° (2 ⁇ ).
- the diffraction patterns were analyzed using DIFFRAC.EVA (V4.3.1).
- TGA analysis was performed on a Discovery TGA550 (TA Instruments, USA).
- a TA Instruments Discovery TGA was used for the thermal analysis.
- the specific TGA-IR method for the TGA reduces the standard flow rate from 50 mL/minute to 25 mL/minute in order to increase the resonance time of the evolved gas in the gas cell.
- the ramp rate and final temperature for heating were controlled in the TGA software (typically 10 °C/minute). Sample size was typically in the 10-15 mg range.
- the TGA method was set up with the following parameters: Mass flow rate of 25 mL/min, event 1 “on”, ramp 10 °C/min up to desired temperature, event 1 “off”.
- a Thermo 6700 FT-IR spectrometer equipped with a KBr beamsplitter and a d-TGS KBr detector was utilized for TGA-IR analysis.
- the collection range was 4000-400 cm -1 and Happ-Genzel apodization was used for data collection.
- Background spectra were collected at 4 cm -1 resolution with 64 co-added scans.
- the IR background was collected with the TGA furnace closed and purged for 2-3 minutes.
- the sample method was set up for quick spectral collection, as the evolved gases were swept quickly through the gas cell. Each sample spectral data point was a co-added spectrum of five spectra, collected with 4 cm -1 resolution.
- the total IR collection time was adjusted based on the run length of the TGA.
- DSC thermograms were recorded on a Discovery DSC25 (TA Instruments, USA) equipped with a RCS90 chiller. Typically, samples of approximately 0.5 - 5 mg were heated at a rate of 10 °C/min in pin-holed aluminum pans from 30 °C to the final temperature. Measurements were performed at a constant nitrogen flow rate of 50 mL/min. The DSC data were analyzed using TRIOS (V4.4.4.41651).
- the FT Raman spectra depicted in Figure 5 was obtained on a Kaiser Optical Systems dispersive Raman microscope equipped with a thermoelectrically cooled CCD detector.
- the source was a 400 mW diode laser operating at 785 nm, with an average power output of 100 mW through a 50 x 11 mm working distance objective.
- the spot size of the laser beam through the objective was approximately 20 m.
- the microscope was coupled to the spectrometer with multi-mode fiber optic cables.
- the collection range was 3500-50 cm -1 .
- the collection (exposure) time and number of accumulations is adjusted based on the efficiency of the Raman scattering of the sample. For most non-fluorescent samples, an exposure time of 10 seconds with one accumulation proved adequate for spectral collection.
- the polarized light microscopy images depicted in Figures 6, 14, and 20 were captured on an Olympus BX50 or BX60, Olympus BH2, Leitz Orthoplan, or a Nikon Eclipse E600 POL optical microscope, which are considered to be functionally and operationally comparable. Samples were dispersed in silicone oil or analyzed as a dry powder on a flat glass substrate. Samples were viewed under plane and/or cross-polarized light with a 530 nm wave plate.
- Solutions were prepared by adding enough solids to a given solvent so that excess solids were present. The mixture was then agitated in a sealed vial at either ambient or an elevated temperature. After a given amount of time, the solids were isolated for analysis.
- Forms 1 and 4 are anhydrous forms of Compound A; Forms 2 and 3 are hydrates. Crystalline Form 3 (dihydrate) of Compound A was identified as the most thermodynamically stable, relative to the other polymorphic forms of Compound A. Characterization data are discussed in more detail below.
- TGA and DSC thermograms for Form 2 are depicted in Figures 8 and 9, respectively.
- the TGA data showed about 8% weight loss up to ⁇ 200 °C.
- the weight loss was confirmed to be due to water loss by TGA-IR.
- Those data are consistent with a hydrated form.
- the DSC curve showed a prolonged first endotherm followed by two more endothermic events with onsets at ⁇ 83 °C and ⁇ 99 °C, respectively.
- Form 4 was obtained by dissolving Compound A (1.0 eq.) in isopropanol (5.0 vol.) and heating the resulting solution to 70-80 °C. The mixture was then cooled to 45-50 °C and maintained at that temperature overnight, resulting in the precipitation of solids. The solids were collected via vacuum filtration and dried under vacuum at 50 °C to afford Form 4.
- An XRPD pattern for Form 4 is provided in Figure 15, and the observed peaks are summarized in Table 6.
- Embodiment 4 A crystalline form of Compound wherein the crystalline form is Form 2.
- Embodiment 8 The crystalline form of Compound A according to embodiment 7, characterized by an XRPD pattern comprising peaks at 10.5, 12.0, 17.7, 20.6, and 24.0 ⁇ 0.2 degrees 2 ⁇ .
- Embodiment 9 The crystalline form of Compound A according to embodiment 7, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 10; b. a TGA thermogram substantially as set forth in Figure 11; c. a DSC thermogram substantially as set forth in Figure 12; d. a DVS plot substantially as set forth in Figure 13; and e. a PLM image as set forth in Figure 14.
- Embodiment 10 A crystalline form of Compound wherein the crystalline form is Form 4.
- Embodiment 11 The crystalline form of Compound A according to embodiment 10, characterized by an XRPD pattern comprising peaks at 12.9, 16.4, 19.5, 20.3, and 24.0 ⁇ 0.2 degrees 2 ⁇ .
- Embodiment 12 The crystalline form of Compound A according to embodiment 10, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 15; b. a TGA thermogram substantially as set forth in Figure 16; c. a DSC thermogram substantially as set forth in Figure 17; d. a DVS plot substantially as set forth in Figure 18; e. a FT Raman spectrum substantially as set forth in Figure 19; and f. a PLM image as set forth in Figure 20.
- Embodiment 13 A pharmaceutical composition comprising one or more of a crystalline form of Compound A according to any one of embodiments 1-12, and at least one pharmaceutically acceptable carrier or excipient.
- Embodiment 14 The pharmaceutical composition according to embodiment 13, wherein the pharmaceutical composition is formulated as a tablet, capsule, pill, lozenge, powder, granule, solution or suspension.
- Embodiment 15 The pharmaceutical composition according to embodiment 13 or 14, wherein the pharmaceutical composition is in an extended-release form, a non-extended release form, or an immediate release form.
- Embodiment 16 The pharmaceutical composition according to embodiment 15, wherein the pharmaceutical composition is in an extended release form.
- Embodiment 17 The pharmaceutical composition according to embodiment 15, wherein the pharmaceutical composition is in a non-extended release form.
- Embodiment 18 The pharmaceutical composition according to embodiment 15, wherein the pharmaceutical composition is in an immediate release form.
- Embodiment 19 A pharmaceutical combination comprising one or more of a crystalline form of Compound A according to any one of embodiments 1-12, or one or more of a pharmaceutical composition according to any one of embodiments 13-18, and one or more additional therapeutic agents.
- Embodiment 20 The pharmaceutical combination according to embodiment 19, wherein the one or more additional therapeutic agents are selected from risperidone, olanzapine, clozapine, quetiapine, aripiprazole, ziprasidone, paliperidone, asenapine, iloperidone, or bexpiprazole.
- the one or more additional therapeutic agents are selected from risperidone, olanzapine, clozapine, quetiapine, aripiprazole, ziprasidone, paliperidone, asenapine, iloperidone, or bexpiprazole.
- Embodiment 21 The pharmaceutical combination according to embodiment 19, wherein the one or more additional therapeutics agents are selected from ziprasidone, risperidone, olanzapine, quetiapine, aripiprazole, asenapine, blonanserin, or iloperidone.
- Embodiment 22 The pharmaceutical combination according to embodiment 19, wherein the one or more additional therapeutics agents are selected from ziprasidone, risperidone, olanzapine, quetiapine, aripiprazole, asenapine, blonanserin, or iloperidone.
- the one or more additional therapeutic agents are selected from selegiline, selegiline hydrochloride, dimethylselegilene, brofaromine, phenelzine, tranylcypromine, moclobemide, befloxatone, safinamide, isocarboxazid, nialamide, rasagiline, iproniazide, CHF-3381, iproclozide, toloxatone, desoxypeganine, harmine, harmaline, linezolid, pargyline, metralindole, pirlindole, bifemelane, bupropion, nefazodone, trazodone, desvenlafaxine, duloxetine, levomilnacipran, milnacipran, venlafaxine, citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline, amitriptyline, amitripty
- Embodiment 23 The pharmaceutical combination of embodiment 19, wherein the one or more additional therapeutic agents are selected from xanomeline, donepezil, galantamine, or rivastigmine.
- Embodiment 24 A method for treating a disease or disorder in a patient in need thereof, said method comprising administering to the patient a therapeutically effective amount of one or more of a crystalline form of Compound A according to any one of embodiments 1-12, a pharmaceutical composition of them according to any one of embodiments 13-18, or a pharmaceutical combination of them according to any one of embodiments 19-23.
- Embodiment 25 The method according to embodiment 24, wherein the disease or disorder is selected from Alzheimer’s Disease, psychosis in Alzheimer’s Disease, schizophrenia or psychosis, pain, addiction, a sleep disorder, a cognitive disorder (e.g. mild cognitive impairment), Parkinson’s Disease, Parkinson’s Disease-levodopa-induced dyskinesia, Huntington’s Disease, dyskinesia, dry mouth, pulmonary hypertension, chronic obstructive pulmonary disease (COPD), asthma, urinary incontinence, glaucoma, Trisomy 21 (Down Syndrome), cerebral amyloid angiopathy, dementia, Hereditary Cerebral Hemorrhage with Amyloidosis of the Dutch-Type (HCHWA-D), Creutzfeld-Jakob disease, prion disorders, amyotrophic lateral sclerosis, progressive supranuclear palsy, head trauma, stroke, pancreatitis, inclusion body myositis, other peripheral amyloidoses, diabetes, autism
- Embodiment 27 The method according to embodiment 26, wherein the disease or disorder is selected from the group consisting of Alzheimer’s Disease or dementia, psychosis in Alzheimer’s disease or dementia, and agitation in Alzheimer’s disease or dementia.
- Embodiment 28 The method according to embodiment 26, wherein the disease or disorder is schizophrenia.
- Embodiment 29 The method according to embodiment 26, wherein the disease or disorder is a bipolar disorder or depressive episodes associated therewith.
- Embodiment 30 The method according to any one of embodiments 24-29, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered subcutaneously, intravenously, orally, sublingually, buccally, transdermally, arterially, intradermally, intramuscularly, intraperitoneally, ocularly, intranasally, intraspinally or intracerebrally.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- Engineering & Computer Science (AREA)
- Bioinformatics & Cheminformatics (AREA)
- Biomedical Technology (AREA)
- Neurology (AREA)
- Neurosurgery (AREA)
- General Chemical & Material Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- Veterinary Medicine (AREA)
- Medicinal Chemistry (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmacology & Pharmacy (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Psychology (AREA)
- Hospice & Palliative Care (AREA)
- Psychiatry (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Crystalline forms of a muscarinic M4 receptor activator, Compound A, pharmaceutical compositions and combinations comprising those crystalline forms, their use in treating diseases or disorders and methods of producing the crystalline forms. Also provided are pharmaceutical compositions comprising a core comprising Compound A or a pharmaceutically acceptable salt or hydrate thereof, and a coating comprising an opacifier.
Description
CRYSTALLINE FORMS OF A MUSCARINIC M4 RECEPTOR MODULATOR AND METHODS OF TREATING DISEASES
CROSS-REFERENCE TO RELATED APPLICATIONS
[0001] This application claims priority to and benefit of United States Provisional Application No. 63/616,123 filed December 29, 2023, which is incorporated herein by reference in its entirety.
TECHNICAL FIELD OF THE DISCLOSURE
[0002] The disclosure relates to crystalline forms, i.e., polymorphs, of a muscarinic M4 receptor activator, pharmaceutical compositions and combinations comprising those crystalline forms, and their use in treating diseases and disorders, such as Alzheimer's Disease, psychosis in Alzheimer’s disease, schizophrenia, psychotic disorders, pain, addiction, bipolar disorder, tardive dyskinesia, and sleep disorders. Also provided herein are methods for producing the crystalline forms of this disclosure.
BACKGROUND OF THE DISCLOSURE
[0003] Patients with schizophrenia, Alzheimer’s Disease, Parkinson’s Disease, Huntington’s Disease, depression and various other neurological/neurodegenerative diseases frequently suffer from behavioral and cognitive impairments resulting in debilitating disruptions to their daily lives. Over the years many pharmacological treatments have been developed that provide some improvements in behavior and cognitive function. However, the improvements are modest, and as is often the case, the underlying dose-limiting adverse effects associated with these treatments, including extrapy rami dal and metabolic side-effects, lead to partial responsiveness and non-compliance.
[0004] In an effort to discover new and improved pharmacological treatments, researchers have looked to modulation of the muscarinic acetylcholine receptor (mAChR) as a potential viable mechanism. There are five mAChRs subtypes (M1-M5) that have been identified and are part of the G protein-coupled receptor (GPCR) superfamily. These subtypes are widely distributed throughout the periphery and the central nervous system (CNS), with the Ml and M4 subtypes being predominantly expressed in the CNS.
[0005] In this context, some researchers have focused on identifying subtype-selective M4 muscarinic acetylcholine receptor activators. For example, positive allosteric modulators (PAMs) of the M4 muscarinic acetylcholine receptor have gained interest as possible agents for treating the behavioral impairments associated with schizophrenia and other neuropsychiatric
disorders, e.g., Alzheimer’s Disease. [See: Bubser, Michael, et al., “Selective Activation of M4 Muscarinic Acetylcholine Receptors reverses MK-801 -Induced Behavioral Impairments and
Enhances Associative Learning in Rodents”, American Chemical Society, Chemical Neuroscience (2014); and Bynum, Nellie E., et al., “Antipsychotic Drug-Like Effects of the Selective M4 Muscarinic Acetylcholine Receptor Positive Allosteric Modulator VU0152100”, Neuropsychopharmacology (2014) 1-16],
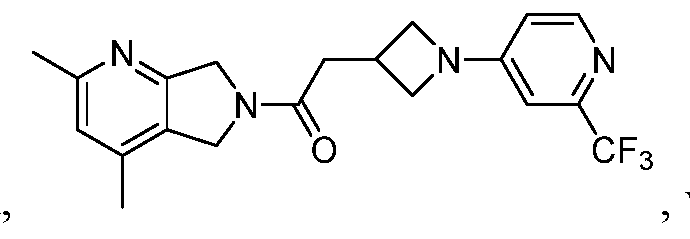
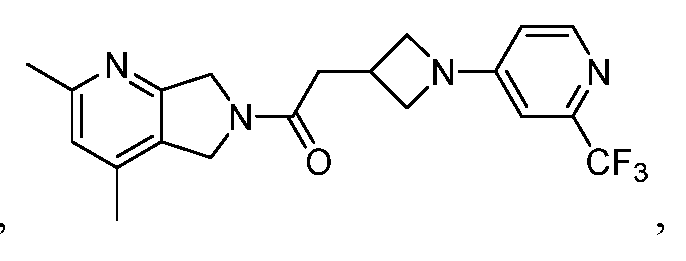
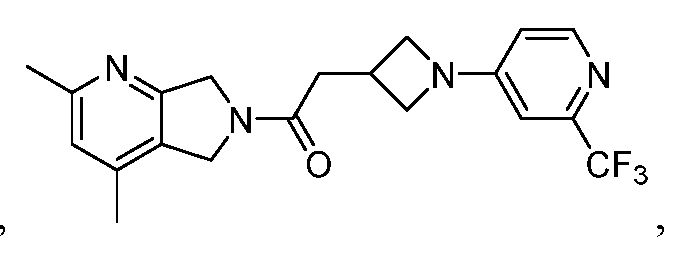
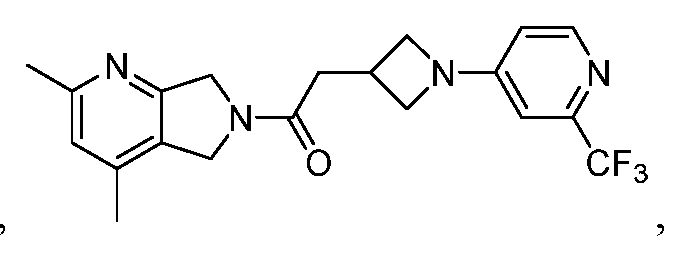
[0006] One such PAM of the M4 muscarinic acetylcholine receptor is Compound A
There is, however, a need for thermodynamically stable forms of Compound A or its hydrates; forms of Compound A or its hydrates that can be manufactured in high purity and large quantities, and thus be, more suitable for inclusion in pharmaceutical formulations; and/or forms of Compound A or its hydrates that display other advantageous physical properties, such as lack of hygroscopic tendencies, improved solubility, and enhanced rates of dissolution due to different lattice energies.
SUMMARY OF THE DISCLOSURE
[0007] The present disclosure provides crystalline forms, i.e., polymorphs, of Compound A,
. In some embodiments, the crystalline forms of Compound A are anhydrous and, in some forms they are hydrates, e.g., hemi-, mono- or di -hydrated (together “crystalline forms of Compound A”). Also provided in the embodiments of this disclosure are processes for preparing the crystalline forms of Compound A as well as pharmaceutical compositions and combinations thereof, and methods for using those crystalline forms, compositions, and combinations in the treatment of diseases or disorders, e.g., Alzheimer’s Disease, dementia, including psychosis and agitation associated therewith, schizophrenia, psychotic disorders, pain, addiction, bipolar disorder (bipolar I, bipolar II, bipolar depression, manic or mixed episodes), tardive dyskinesia, and sleep disorders.
[0008] In a first aspect, the present disclosure provides a crystalline form of Compound A, wherein the crystalline form is Form 1 as described herein.
[0009] In a second aspect, the present disclosure provides a crystalline form of Compound A, wherein the crystalline form is Form 2 as described herein.
[0010] In a third aspect, the present disclosure provides a crystalline form of Compound A, wherein the crystalline form is Form 3 as described herein.
[0011] In a fourth aspect, the present disclosure provides a crystalline form of Compound A, wherein the crystalline form is Form 4 as described herein.
[0012] In a fifth aspect, the present disclosure is directed to a pharmaceutical composition comprising one or more of a crystalline form of Compound A and at least one pharmaceutically acceptable carrier.
[0013] In a sixth aspect, the present disclosure is directed to a pharmaceutical combination comprising one or more of a crystalline form of Compound A and one or more additional therapeutic agents, for example, neuropsychiatric agents (e.g., antipsychotics, including atypical antipsychotics, anti-schizophrenia agents, and/or antidepressants).
[0014] In a seventh aspect, the present disclosure provides a method for treating a disease or disorder in a patient in need thereof, said method comprising administering to the patient a therapeutically effective amount of one or more of a crystalline form of Compound A according to the disclosure, or a pharmaceutical composition or combination of them. In some embodiments, one or more of a crystalline form of Compound A according to the disclosure, or a pharmaceutical composition or combination of them are administered as a mono or adjunctive treatment for the disease or disorder.
[0015] In an eighth aspect, the present disclosure provides a method of producing a crystalline form of Compound A described herein (i.e., Form 1, Form 2, Form 3, and Form 4 of Compound A).
[0016] Particular embodiments of the disclosure are set-forth in the following numbered paragraphs:
2. The crystalline form of Compound A according to embodiment 1, characterized by an XRPD pattern comprising peaks selected from at 12.7, 12.9, 19.1, 19.7, and 24.3 ± 0.2 degrees 2θ.
3. The crystalline form of Compound A according to embodiment 1, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 1; b. a TGA thermogram substantially as set forth in Figure 2; c. a DSC thermogram substantially as set forth in Figure 3;
a DVS plot substantially as set forth in Figure 4; a FT Raman spectrum substantially as set forth in Figure 5; and a PLM image substantially as set forth in Figure 6.
A crystalline form of Compound A,
wherein the crystalline form is Form 2. The crystalline form of Compound A according to embodiment 4, characterized by an XRPD pattern comprising peaks at 10.2, 11.5, 17.5, 19.9, and 23.1 ± 0.2 degrees 2θ. The crystalline form of Compound A according to embodiment 4, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 7; b. a TGA thermogram substantially as set forth in Figure 8; and c. a DSC thermogram substantially as set forth in Figure 9.
A crystalline form of Compound A, wherein the crystalline
form is Form 3. The crystalline form of Compound A according to embodiment 7, characterized by an XRPD pattern comprising peaks at 10.5, 12.0, 17.7, 20.6, and 24.0 ± 0.2 degrees 2θ. The crystalline form of Compound A according to embodiment 7, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 10; b. a TGA thermogram substantially as set forth in Figure 11; c. a DSC thermogram substantially as set forth in Figure 12; d. a DVS plot substantially as set forth in Figure 13; and e. a PLM image as set forth in Figure 14. A crystalline form of Compound
wherein the crystalline form is Form 4.
11. The crystalline form of Compound A according to embodiment 10, characterized by an XRPD pattern comprising peaks at 12.9, 16.4, 19.5, 20.3, and 24.0 ± 0.2 degrees 2θ.
12. The crystalline form of Compound A according to embodiment 10, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 15; b. a TGA thermogram substantially as set forth in Figure 16; c. a DSC thermogram substantially as set forth in Figure 17; d. a DVS plot substantially as set forth in Figure 18; e. a FT Raman spectrum substantially as set forth in Figure 19; and f. a PLM image as set forth in Figure 20.
13. A pharmaceutical composition comprising one or more of a crystalline form of Compound A according to any one of embodiments 1-12, and at least one pharmaceutically acceptable carrier or excipient.
14. The pharmaceutical composition according to embodiment 13, wherein the pharmaceutical composition is formulated as a tablet, capsule, pill, lozenge, powder, granule, solution or suspension.
15. The pharmaceutical composition according to embodiment 13 or 14, wherein the pharmaceutical composition is in an extended-release form, a non-extended release form, or an immediate release form.
16. The pharmaceutical composition according to embodiment 15, wherein the pharmaceutical composition is in an extended release form.
17. The pharmaceutical composition according to embodiment 15, wherein the pharmaceutical composition is in a non-extended release form.
18. The pharmaceutical composition according to embodiment 15, wherein the pharmaceutical composition is in an immediate release form.
19. A pharmaceutical combination comprising one or more of a crystalline form of Compound A according to any one of embodiments 1-12, or one or more of a pharmaceutical composition according to any one of embodiments 13-18, and one or more additional therapeutic agents.
20. The pharmaceutical combination according to embodiment 19, wherein the one or more additional therapeutic agents are selected from risperidone, olanzapine, clozapine, quetiapine, aripiprazole, ziprasidone, paliperidone, asenapine, iloperidone, or bexpiprazole.
21. The pharmaceutical combination according to embodiment 19, wherein the one or more additional therapeutics agents are selected from ziprasidone, risperidone, olanzapine, quetiapine, aripiprazole, asenapine, blonanserin, or iloperidone.
22. The pharmaceutical combination of embodiment 19, wherein the one or more additional therapeutic agents are selected from selegiline, selegiline hydrochloride, dimethylselegilene, brofaromine, phenelzine, tranylcypromine, moclobemide, befloxatone, safinamide, isocarboxazid, nialamide, rasagiline, iproniazide, CHF-3381, iproclozide, toloxatone, desoxypeganine, harmine, harmaline, linezolid, pargyline, metralindole, pirlindole, bifemelane, bupropion, nefazodone, trazodone, desvenlafaxine, duloxetine, levomilnacipran, milnacipran, venlafaxine, citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline, amitriptyline, amitriptylinoxide, clomipramine, desipramine, dibenzepin, dimetacrine, dosulepin, doxepin, imipramine, lofepramine, melitracen, nitroxazepine, nortriptyline, noxiptiline, opipramol, pipofezine, protriptyline, trimiprame, amoxapine, maprotiline, mianserin, mirtazapine, or setiptiline.
23. The pharmaceutical combination of embodiment 19, wherein the one or more additional therapeutic agents are selected from xanomeline, donepezil, galantamine, or rivastigmine.
24. A method for treating a disease or disorder in a patient in need thereof, said method comprising administering to the patient a therapeutically effective amount of one or more of a crystalline form of Compound A according to any one of embodiments 1-12, a pharmaceutical composition according to any one of embodiments 13-18, or a pharmaceutical combination according to any one of embodiments 19-23.
25. The method according to embodiment 24, wherein the disease or disorder is selected from Alzheimer’s Disease, psychosis in Alzheimer’s Disease, schizophrenia or psychosis, pain, addiction, a sleep disorder, a cognitive disorder (e.g. mild cognitive impairment), Parkinson’s Disease, Parkinson’s Disease-levodopa-induced dyskinesia, Huntington’s Disease, dyskinesia, dry mouth, pulmonary hypertension, chronic obstructive pulmonary disease (COPD), asthma, urinary incontinence, glaucoma, Trisomy 21 (Down Syndrome), cerebral amyloid angiopathy, dementia, Hereditary Cerebral Hemorrhage with Amyloidosis
of the Dutch-Type (HCHWA-D), Creutzfeld-Jakob disease, prion disorders, amyotrophic lateral sclerosis, progressive supranuclear palsy, head trauma, stroke, pancreatitis, inclusion body myositis, other peripheral amyloidoses, diabetes, autism, tardive dyskinesia, or atherosclerosis.
26. The method according to embodiment 24, wherein the disease or disorder is selected from Alzheimer’s disease, dementia, a bipolar disorder, schizophrenia, pain, an addiction, a sleep disorder, and conditions associated therewith.
27. The method according to embodiment 26, wherein the disease or disorder is selected from the group consisting of Alzheimer's Disease or dementia, psychosis in Alzheimer’s disease or dementia, and agitation in Alzheimer’s disease or dementia.
28. The method according to embodiment 26, wherein the disease or disorder is schizophrenia.
29. The method according to embodiment 26, wherein the disease or disorder is a bipolar disorder or depressive episodes associated therewith.
30. The method according to any one of embodiments 24-29, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered subcutaneously, intravenously, orally, sublingually, buccally, transdermally, arterially, intradermally, intramuscularly, intraperitoneally, ocularly, intranasally, intraspinally or intracerebrally.
31. The method according to embodiment 24, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered orally.
32. The method according to embodiment 24, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered intramuscularly.
33. The method according to any one of embodiments 24-32, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered once daily.
34. The method according to any one of embodiments 24-32, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination them, is administered twice daily.
35. The method according to any one of embodiments 24-34, wherein the therapeutically effective amount of the crystalline form of Compound A is between 5 mg to 75 mg.
36. The method according to embodiment 35, wherein the therapeutically effective amount of the crystalline form of Compound A is between 10 mg to 30 mg.
(i) dissolving Compound A in 2-propanol at 65-75 °C;
(ii) cooling the mixture to ambient temperature;
(iii) filtering the slurry; and
(iv) drying the collected solids.
(i) dissolving crystalline Form 1 of Compound A in a solvent selected from acetonitrile, isopropyl alcohol, ethyl acetate, or acetone; and
(ii) slow evaporation.
, comprising the steps of:
(i) slurrying crystalline Form 1 of Compound A in a solvent mixture selected from:
(a) acetonitrile-water at ambient temperature, wherein the mixture comprises about 99.7
% of acetonitrile by volume;
(b) acetonitrile-water at 5°C, wherein the mixture comprises about 99.3% of acetonitrile by volume; or
(c) acetonitrile-water at 50°C, wherein the mixture comprises about 97.2% of acetonitrile by volume; and
(ii) filtering the slurry. A method of producing crystalline Form 2 of Compound
comprising the steps of:
(i) dissolving Compound A in 2-propanol at 65-75 °C;
(ii) cooling the mixture to 35-45 °C;
(iii) slowly adding water;
(iv) filtering the slurry; and
(v) drying the collected solids. A method of producing crystalline Form 3 of Compound
comprising the steps of:
(i) slurrying crystalline Form 1 of Compound A in a solvent selected from 2-methyl tetrahydrofuran, tetrahydrofuran, water, or toluene at between 5 °C and 50 °C; and
(ii) filtering the slurry. A method of producing crystalline Form 3 of Compound
comprising the steps of:
(i) slurrying crystalline Form 1 of Compound A in a solvent mixture selected from:
(a) acetonitrile-water at ambient temperature, wherein the mixture comprises about
38.4%, 79.2%, 92.1%, 95.6%, 97.2%, 98.1%, 98.8% of acetonitrile by volume;
(b) acetonitrile-water at 5°C, wherein the mixture comprises about 79.2% or 97.2% of acetonitrile by volume; or
(c) acetonitrile-water at 50°C, wherein the mixture comprises about 79.2% of acetonitrile by volume; and
(ii) filtering the slurry.
43. A method of producing crystalline Form 3 of Compound A,
comprising the steps of:
(i) dissolving Compound A in 2-propanol at 65-75 °C;
(ii) cooling the mixture to 50-55 °C;
(iii) slowly adding water;
(iv) adding a seed crystal;
(v) cooling the mixture to ambient temperature;
(vii) filtering the slurry; and
, comprising the steps of:
(i) slurrying crystalline Form 1 of Compound A in a mixture of acetonitrile-water at 50°C, wherein the mixture comprises about 79.2% of acetonitrile by volume; and
(ii) filtering the slurry. 45. A method of producing crystalline Form 4 of Compound
, comprising the steps of:
(i) dissolving Compound A in 2-propanol at 70-80 °C;
(ii) cooling the mixture to 45-50 °C;
(iii) filtering the slurry; and
(iv) drying the collected solids. 46. A pharmaceutical composition comprising a core and a coating, wherein the core comprises Compound A, or a pharmaceutically acceptable salt or hydrate thereof, and one or more pharmaceutically acceptable carriers or excipients, and wherein the coating comprises an opacifier. 47. A pharmaceutical composition comprising about 5 mg, about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, or about 50 mg of
Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 85 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 5 weight percent of at least one lubricant, and (d) about 1 weight percent to about 8 weight percent of a coating comprising an opacifier, based on the total weight of the pharmaceutical composition.
48. A pharmaceutical composition comprising about 10 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 87 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
49. A pharmaceutical composition comprising about 15 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 87 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
50. A pharmaceutical composition comprising about 30 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 85 weight percent to about 93 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
51. The pharmaceutical composition according to any one of embodiments 46 to 50, wherein the opacifier is selected from the group consisting of: titanium dioxide, silicon dioxide, zinc oxide, iron oxide, talc, calcium carbonate, calcium phosphate, calcium sulfate, behenic acid, aluminum silicate, aluminum hydroxide, magnesium carbonate, tartrazine, erythrosine, or a mixture thereof.
52. The pharmaceutical composition according to any one of embodiments 46 to 50, wherein the opacifier is titanium dioxide.
53. The pharmaceutical composition according to any one of embodiments 46 to 52, wherein coating comprises die opacifier in an amount of from about 4 weight percent to about 25
weight percent of the coating by weight, based upon total weight of the coating composition.
54. The pharmaceutical composition according to any one of embodiments 46 to 53, wherein coating comprises the opacifier in an amount of from about 4 weight percent to about 20 weight percent of the coating by weight, based upon total weight of the coating composition.
55. The pharmaceutical composition according to any one of embodiments 46 to 54, wherein coating comprises the opacifier in an amount of from about 5 weight percent to about 15 weight percent, or about 10 weight percent to about 15 weight percent, or about 15 weight percent to about 20 weight percent of the coating by weight, based upon total weight of the coating composition.
56. The pharmaceutical composition according to any one of embodiments 46 to 55, wherein coating comprises the opacifier in an amount of about 6 weight percent, about 14 weight percent, or about 17 weight percent of the coating by weight, based upon total weight of the coating composition.
57. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising a crystalline form of Compound A.
58. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising one or more of a crystalline form of Compound A according to any one of embodiments 1-12.
59. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising crystalline Form 1 of Compound A according to any one of embodiments 1 to 3.
60. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising crystalline Form 2 of Compound A according to any one of embodiments 4 to 6.
61. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising crystalline Form 3 of Compound A according to any one of embodiments 7 to 9.
62. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising crystalline Form 4 of Compound A according to any one of embodiments 10 to 12.
63. A method for treating a disease or disorder in a patient in need thereof, said method comprising administering to the patient a pharmaceutical composition according to any one of embodiments 46 to 62.
64. The method according to embodiment 63, wherein the disease or disorder is selected from Alzheimer’s Disease, psychosis in Alzheimer’s Disease, schizophrenia or psychosis, pain, addiction, a sleep disorder, a cognitive disorder (e.g. mild cognitive impairment), Parkinson’s Disease, Parkinson’s Disease-levodopa-induced dyskinesia, Huntington’s Disease, dyskinesia, dry mouth, pulmonary hypertension, chronic obstructive pulmonary disease (COPD), asthma, urinary incontinence, glaucoma, Trisomy 21 (Down Syndrome), cerebral amyloid angiopathy, dementia, Hereditary Cerebral Hemorrhage with Amyloidosis of the Dutch-Type (HCHWA-D), Creutzfeld-Jakob disease, prion disorders, amyotrophic lateral sclerosis, progressive supranuclear palsy, head trauma, stroke, pancreatitis, inclusion body myositis, other peripheral amyloidoses, diabetes, autism, tardive dyskinesia, or atherosclerosis.
65. The method according to embodiment 63, wherein the disease or disorder is selected from Alzheimer’s disease, dementia, a bipolar disorder, schizophrenia, pain, an addiction, a sleep disorder, and conditions associated therewith.
66. The method according to embodiment 63, wherein the disease or disorder is selected from the group consisting of Alzheimer's Disease or dementia, psychosis in Alzheimer’s disease or dementia, and agitation in Alzheimer’s disease or dementia.
67. The method according to embodiment 63, wherein the disease or disorder is schizophrenia.
68. The method according to embodiment 63, wherein the disease or disorder is a bipolar disorder or depressive episodes associated therewith.
BRIEF DESCRIPTION OF THE DRAWINGS
[0017] FIGURE 1 depicts an exemplary X-ray powder diffraction (XRPD) pattern for Form 1. [0018] FIGURE 2 depicts an exemplary thermal gravimetric analysis (TGA) thermogram for Form 1.
[0019] FIGURE 3 depicts an exemplary differential scanning calorimetry (DSC) thermogram for Form 1.
[0020] FIGURE 4 depicts an exemplary dynamic vapor sorption (DVS) plot for Form 1.
[0021] FIGURE 5 depicts an exemplary Fourier Transformed (FT) Raman spectrum for Form 1.
[0022] FIGURE 6 depicts an exemplary polarized light microscopy (PLM) image for Form 1.
[0023] FIGURE 7 depcits an exemplary XRPD pattern for Form 2.
[0024] FIGURE 8 depicts an exemplary TGA thermogram for Form 2.
[0025] FIGURE 9 depicts an exemplary DSC thermogram for Form 2.
[0026] FIGURE 10 depicts exemplary XRPD patterns for Form 3.
[0027] FIGURE 11 depicts an exemplary TGA thermogram for Form 3.
[0028] FIGURE 12 depicts an exemplary DSC thermogram for Form 3.
[0029] FIGURE 13 depicts an exemplary DVS plot for Form 3.
[0030] FIGURE 14 depicts an exemplary PLM image for Form 3.
[0031] FIGURE 15 depicts an exemplary XRPD pattern for Form 4.
[0032] FIGURE 16 depicts an exemplary TGA thermogram for Form 4.
[0033] FIGURE 17 depicts an exemplary DSC thermogram for Form 4.
[0034] FIGURE 18 depicts an exemplary DVS plot for Form 4.
[0035] FIGURE 19 depicts an exemplary FT Raman spectrum for Form 4.
[0036] FIGURE 20 depicts an exemplary PLM image for Form 4.
DETAILED DESCRIPTION OF THE DISCLOSURE
Definitions
[0037] Unless otherwise defined herein, scientific and technical terms used in this application shall have the meanings that are commonly understood by those of ordinary skill in the art. Generally, nomenclature used in connection with, and techniques of, chemistry, cell and tissue culture, molecular biology, cell and cancer biology, neurobiology, neurochemistry, virology, immunology, microbiology, pharmacology, genetics and protein and nucleic acid chemistry, described herein, are those well-known and commonly used in the art.
[0038] All of the publications, patents and published patent applications referred to in this application are specifically incorporated by reference herein in their entirety. In case of conflict, the present specification, including its specific definitions, will control.
[0039] Recitation of ranges of values herein are merely intended to serve as a shorthand method of referring individually to each separate value falling within the range and including the endpoints, unless otherwise indicated herein, and each separate value is incorporated into the specification as if it were individually recited herein. All methods described herein can be performed in any suitable order unless otherwise indicated herein or otherwise clearly
contradicted by context. The use of any and all examples, or exemplary language (e.g., “such as”) provided herein, is intended merely to better illuminate the embodiments and does not pose a limitation on the scope of the claims unless otherwise stated. No language in the specification should be construed as indicating any non-claimed element as essential.
[0040] Throughout this specification, the word “comprise” or variations such as “comprises” or “comprising” will be understood to imply the inclusion of a stated integer (or components) or group of integers (or components), but not the exclusion of any other integer (or components) or group of integers (or components).
[0041] The singular forms “a,” “an,” and “the” as used herein include the plurals unless the context clearly dictates otherwise.
[0042] The term “including” means “including but not limited to.” “Including” and “including but not limited to” are used interchangeably.
[0043] The term “or” as used herein should be understood to mean “and/or,” unless the context clearly indicates otherwise.
[0044] The use of the term “about” includes and describes the value or parameter per se. For example, “about x” includes and describes “x” per se. In some embodiments, the term “about” when used in association with a measurement, or used to modify a value, a unit, a constant, or a range of values, refers to variations of 4.10%. For example, “about 2:8” in some embodiments Includes 1.8-2.2:7 2-8.8.
[0045] As used herein, the term “ambient temperature” refers to the air temperature of any object or environment where the material is stored. In some embodiments, it also refers to an ordinary temperature or the baseline temperature. In some embodiments, it refers to the average temperature of an environment.
[0046] “Compound A” as used herein has the structural formula:
Compound A is a muscarinic M4-receptor activator and is useful for treating diseases/disorders in a subject in need or at risk thereof, and/or for slowing the progression of the diseases or disorders in a subject in need or at risk thereof (see, W02018002760A1).
[0047] As used herein, “administering” or “administration of” a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof to a subject can be carried out using one of a variety of methods known to those skilled in the art. For example, a crystalline form of Compound A of this disclosure can be administered itself or in an appropriate
pharmaceutical composition or as part of a combination with one or more additional therapeutic agents intravenously, arterially, intradermally, intramuscularly, intraperitoneally, intravenously, subcutaneously, ocularly, sublingually, orally (by ingestion), intranasally (by inhalation), intraspinally, intracerebrally, and transdermally (by absorption, e.g., through a skin duct). A crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof can also, in some embodiments of this disclosure, be introduced by rechargeable or biodegradable polymeric devices or other devices, e.g., patches and pumps, or in formulations, which provide extended, slow, or controlled release of a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof. The administration can also be performed, for example, once, a plurality of times, and/or over one or more extended periods. In some aspects of this disclosure, the administration includes direct administration, including self-administration, and indirect administration, including the act of prescribing a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof. For example, as used herein, a physician who instructs a patient to self-administer a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof, or to have a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof administered by another and/or who provides a patient with a prescription for a drug is administering a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof to the patient.
[0048] Appropriate methods of administering a crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof to a subject will also depend, for example, on the age of the subject, whether the subject is active or inactive at the time of administering, whether the subject is cognitively impaired at the time of administering, the extent of the impairment, and the chemical and biological properties of the crystalline form of Compound A of this disclosure or a pharmaceutical composition or combination thereof (e.g. solubility, digestibility, bioavailability, stability and toxicity).
[0049] The term “therapeutically effective amount” as used herein refers to that amount of a crystalline form of Compound A of this disclosure being administered, itself or as part of a pharmaceutical composition or in combination with one of more additional therapeutic agents, which will relieve to some extent one or more of the symptoms of the disorder being treated. In reference to the treatment of a disease or disorder (e.g., Alzheimer’s Disease (“AD”), schizophrenia, or psychosis in AD), a therapeutically effective amount refers to that amount which has the effect of relieving to some extent (or, for example, eliminating or reducing) one or
more symptoms associated with the disease or disorder (e.g., a positive, negative, or cognitive symptom of schizophrenia; AD; or a psychotic symptom of AD).
[0050] As used herein, “patient” refers to warm-blooded animals such as, for example, pigs, cows, chickens, horses, guinea pigs, mice, rats, gerbils, cats, rabbits, dogs, monkeys, chimpanzees, and humans.
[0051] As used herein, “pharmaceutically acceptable” indicates that the substance or composition must be compatible, chemically and/or toxicologically, with the other ingredients comprising a formulation, and/or the mammal being treated therewith. In some embodiments, the term includes components, typically non-active, of a pharmaceutical composition that are approved or approvable by a regulatory agency of the Federal or a state government or the corresponding agency in countries other than the United States, or that is listed in the U.S. Pharmacopoeia or other generally recognized pharmacopoeia for use in animals, and more particularly, in humans.
[0052] As used herein “pharmaceutically acceptable carrier” refers to compositions, carriers, diluents, fillers, reagents, and the like, which are pharmaceutically acceptable materials that are capable of administration to or upon a subject. A pharmaceutically acceptable carrier can be involved with carrying or transporting the subject agents from one organ, or portion of the body, to another organ, or portion of the body. The carrier can be in the form of a solid, semi-solid or liquid diluent, cream, or capsule. The active ingredient can be mixed with excipients which are pharmaceutically acceptable and compatible with the active ingredient and in amounts suitable for use in the therapeutic methods described herein. Suitable excipients are, for example, water, saline, dextrose, glycerol, ethanol, or the like and combinations thereof.
[0053] The term “treating” as used herein, unless otherwise indicated, means reversing, alleviating, inhibiting the progress of, or preventing, the disorder or condition to which such term applies, or one or more symptoms of such disorder or condition. The term “treatment” as used herein, unless otherwise indicated, refers to the act of treating as “treating” is defined herein. The term “treating” also includes monotherapy, as well as adjunctive and neo-adjunctive therapy for the treatment of a subject.
[0054] The term “crystalline form” as used herein, refers to a solid-state form which consists of an orderly arrangement of structural units. Specifically, the use of the term “crystalline form” is intended to refer to a crystalline anhydrate or hydrate of Compound A.
[0055] The term “polymorph” as used herein refers to a crystalline form of a compound in a particular crystal packing arrangement. The term “polymorph” includes pseudopolymorphs, which are typically different solvates of a material, and thus their properties differ from one
another. Thus, each distinct polymorph and pseudopolymorph disclosed herein is considered to be a distinct single crystalline form herein. In this disclosure “crystalline form” and “polymorph” are used interchangeably unless the context indicates differently. Certain polymorphs may exhibit enhanced thermodynamic stability or may be more readily manufactured in high purity in large quantities, and thus be more suitable for inclusion in pharmaceutical formulations. Certain polymorphs may also display other advantageous physical properties such as lack of hygroscopic tendencies, improved solubility, and enhanced rates of dissolution due to different lattice energies.
[0056] Crystalline forms are commonly characterized by X-ray powder diffraction (XRPD). An XRPD pattern of reflections (peaks, typically expressed in degrees 2-theta) as well as its major peaks alone, are generally considered fingerprints of a particular crystalline form. The relative intensities of the XRPD peaks may vary depending on, inter alia, the sample preparation technique, crystal size distribution, filters, the sample mounting procedure, and the particular instrument employed. In some instances, more (i.e., new peaks) or fewer (i.e., peaks may disappear) peaks may be present in the XRPD pattern, depending on the type of instrument or the settings. In some instances, any particular peak in an XRPD pattern may appear as a singlet, doublet, triplet, quartet, or multiplet, depending on the type of instrument or the settings, the sensitivity of the instrument, measuring conditions, and/or purity of the crystalline form. In some instances, any particular peak in an XRPD may appear in a symmetric shape or in an asymmetric shape, e.g., having a shoulder. Moreover, instrument variation and other factors can affect the 2-theta values. A skilled artisan understanding these variations is capable of identifying or ascertaining the defining features or characteristics of a particular crystal form using XRPD, as well as using other known physicochemical techniques.
[0057] The term “characteristic peaks” when referring to the peaks in an XRPD pattern of a crystalline form of Compound A of this disclosure refers to a collection of certain peaks whose values of 2θ across a range of 0° - 40° are, as a whole, uniquely assigned to one of the crystalline forms of Compound A.
[0058] The term “ combination” as used herein refers to a product characterized by at least two active agents, one being at least one of a crystalline form of Compound A of this disclosure or a pharmaceutical composition containing them either together (i.e., in one formulation), or separately (i.e., as two separate formulations) with one formulation comprising at least one of a crystalline form of Compound A of this disclosure and the other comprising one of more additional therapeutic agents. The multiple therapeutic agents in a combination of this disclosure can be administered simultaneously or sequentially. The agents can be administered
using the same method, i.e., intraperitoneally, orally, etc., or by different methods. For example, one agent can be administered using one method, e.g., orally, and the other can be administered using a different method, e.g., intraperitoneally.
[0059] As used herein, the term “hydrate” refers to a combination of water with a crystalline form of Compound A according to this disclosure, wherein the water is either absorbed, adsorbed or contained within a crystal lattice of the substrate Compound A.
[0060] As used herein, the term “anhydrous” refers to a form of a crystalline form according to this disclosure that is completely without solvent, e.g., no solvent molecules are contained within the crystal lattice of the crystalline form of Compound A.
Compounds According to the Disclosure
[0061] In some aspects, this disclosure is directed to crystalline forms of Compound A,
. In particular, these forms in the various embodiments of this disclosure are designated Forms 1, 2, 3, and 4. In some embodiments of this disclosure, the crystallized form of Compound A is Form 1. It is anhydrous. In some embodiments of this disclosure, the crystallized form of Compound A is Form 2. It is a monohydrate. In some embodiments of this disclosure, the crystallized form of Compound A is Form 3. It is a dihydrate. In some embodiments of this disclosure, the crystallized form of Compound A is Form 4. It is anhydrous.
[0062] In some embodiments, the crystalline form of Compound A is Form 1, Form 1 being characterized by an XRPD pattern comprising peaks at 12.7, 12.9, 19.1, 19.7, and 24.3 ± 0.2 degrees 2θ. In some embodiments, Form 1 is characterized by an XRPD pattern comprising one or more peaks selected from the group consisting of peaks at 12.7, 12.9, 19.1, 19.7, and 24.3 ± 0.2 degrees 2θ. In some embodiments, the XRPD pattern further comprises one or more peaks selected from the group consisting of peaks at 11.3, 18.0, 20.3, and 21.3 ± 0.2 degrees 2θ.
[0063] In some embodiments of this disclosure, Form 1 of Compound A is characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 1; b. a TGA thermogram substantially as set forth in Figure 2; c. a DSC thermogram substantially as set forth in Figure 3; d. a DVS plot substantially as set forth in Figure 4; e. a FT Raman spectrum substantially as set forth in Figure 5; and f. a PLM image substantially as set forth in Figure 6.
[0064] In some embodiments, Form 1 of Compound A is characterized by a TGA thermogram showing <1% weight loss below about 200 °C.
[0065] In one or more embodiments, Form 1 of Compound A is characterized by a DSC thermogram having an endotherm with an onset of about 183 °C.
[0066] In some embodiments of this disclosure, the crystalline form of Compound A is Form 2, Form 2 being characterized by an XRPD pattern comprising peaks at 10.2, 11.5, 17.5, 19.9, and 23.1 ± 0.2 degrees 2θ. In some embodiments, Form 2 is characterized by an XRPD pattern comprising one or more peaks selected from the group consisting of peaks at 10.2, 11.5, 17.5, 19.9, and 23.1 ± 0.2 degrees 2θ. In some embodiments, the XRPD pattern further comprises one or more peaks selected from the group consisting of peaks at 17.3, 19.2, 20.6, 22.4, 24.8, 25.3, and 26.6 ± 0.2 degrees 2θ.
[0067] In some embodiments of this disclosure, Form 2 of Compound A is characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 7; b. a TGA thermogram substantially as set forth in Figure 8; and c. a DSC thermogram substantially as set forth in Figure 9.
[0068] In some embodiments, Form 2 of Compound A is characterized by a TGA thermogram showing about 8% weight loss below about 200 °C.
[0069] In one or more embodiments, Form 2 of Compound A is characterized by a DSC thermogram having a prolonged first endotherm followed by a second endotherm with an onset of about 83 °C, and a third endotherm with an onset of about 99 °C.
[0070] In some embodiments, the crystalline form of Compound A is Form 3, Form 3 being characterized by an XRPD pattern comprising peaks at 10.5, 12.0, 17.7, 20.6, and 24.0 degrees 2θ ± 0.2 degrees 2θ. In some embodiments, Form 3 is characterized by an XRPD pattern comprising one or more peaks selected from the group consisting of peaks at 10.5, 12.0, 17.7, 20.6, and 24.0 degrees 2θ ± 0.2 degrees 2θ. In some embodiments, the XRPD pattern further comprises one or more peaks selected from the group consisting of peaks at 10.8, 16.1, 16.4, 20.3, 22.2, 24.6, 25.3, and 25.7 ± 0.2 degrees 2θ.
[0071] In some embodiments of this disclosure, Form 3 of Compound A is characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 10; b. a TGA thermogram substantially as set forth in Figure 11; c. a DSC thermogram substantially as set forth in Figure 12; d. a DVS plot substantially as set forth in Figure 13; and
e. a PLM image as set forth in Figure 14.
[0072] In some embodiments, Form 3 of Compound A is characterized by a TGA thermogram showing about 8% weight loss below about 200 °C.
[0073] In one or more embodiments, Form 3 of Compound A is characterized by a DSC thermogram having a broad first endotherm with an onset of about 80 °C followed by a second endotherm with an onset of about 182 °C, and a third endotherm with an onset of about 193 °C. [0074] Form 3 of Compound A is characterized by a higher thermodynamic stability as compared to Forms 1, 2, and 4 of Compound A. Form 3 of Compound A also has limited hygroscopicity, preserving crystallinity, potency, and ease of handling.
[0075] In some embodiments, the crystalline form of Compound A is Form 4, Form 4 being characterized by an XRPD pattern comprising at least one peak selected from the group consisting of 12.9, 16.4, 19.5, 20.3, and 24.0 ± 0.2 degrees 2θ. In some embodiments, Form 4 is characterized by an XRPD pattern comprising one or more peaks selected from the group consisting of peaks at 12.9, 16.4, 19.5, 20.3, and 24.0 ± 0.2 degrees 2θ. In some embodiments, the XRPD pattern further comprises one or more peaks selected from the group consisting of peaks at 14.1, 17.2, 19.9, 20.8, 22.0, and 23.0 ± 0.2 degrees 2θ.
[0076] In some embodiments of this disclosure, Form 4 of Compound A is characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 15; b. a TGA thermogram substantially as set forth in Figure 16; c. a DSC thermogram substantially as set forth in Figure 17; d. a DVS plot substantially as set forth in Figure 18; e. a FT Raman spectrum substantially as set forth in Figure 19; and f. a PLM image substantially as set forth in Figure 20.
[0077] In some embodiments, Form 4 of Compound A is characterized by a TGA thermogram showing <1% weight loss below about 200 °C.
[0078] In one or more embodiments, Form 4 of Compound A is characterized by a DSC thermogram having a sharp endotherm with an onset of about 191 °C.
Pharmaceutical Compositions and Combinations
[0079] In one aspect, this disclosure provides pharmaceutical compositions that comprise one of more of a crystalline form of Compound A and a pharmaceutically acceptable carrier, vehicle, or diluent.
[0080] In some embodiments, the pharmaceutically acceptable carriers of the pharmaceutical compositions of this disclosure include, but are not limited to ion exchangers, alumina, aluminum stearate, lecithin, serum proteins, such as human serum albumin, buffer substances such as phosphates, glycine, sorbic acid, potassium sorbate, partial glyceride mixtures of saturated vegetable fatty acids, water, salts or electrolytes, such as protamine sulfate, disodium hydrogen phosphate, potassium hydrogen phosphate, sodium chloride, zinc salts, colloidal silica, magnesium trisilicate, polyvinyl pyrrolidone, cellulose-based substances, polyethylene glycol, sodium carboxymethylcellulose, polyacrylates, waxes, polyethylene-polyoxypropylene-block polymers, polyethylene glycol and wool fat. Examples of suitable aqueous and non-aqueous carriers which may be employed in the pharmaceutical compositions of the disclosure include water, ethanol, polyols (such as glycerol, propylene glycol, polyethylene glycol, and the like), and suitable mixtures thereof, vegetable oils, such as olive oil, and injectable organic esters, such as ethyl oleate. Proper fluidity can be maintained, for example, by the use of coating materials, such as lecithin, by the maintenance of the required particle size in the case of dispersions, and by the use of surfactants. In some embodiments, no carrier is used. The pharmaceutical compositions described herein may be formulated in any convenient way for use in human medicine.
[0081] In some aspects, this disclosure provides pharmaceutical combinations comprising one or more of a crystalline form of Compound A, or a pharmaceutical composition comprising them, and one or more of an additional therapeutic agent.
[0082] In some aspects, the compositions and combinations of this disclosure comprise Form 1 of Compound A. In some aspects, the compositions and combinations of this disclosure comprise Form 2 of Compound A. In some aspects, the compositions and combinations of this disclosure comprise Form 3 of Compound A. In some aspects, the compositions and combinations of this disclosure comprise Form 4 of Compound A.
[0083] In some embodiments, the one or more additional therapeutic agents may be selected for use in conjunction with the crystalline forms of Compound A of this disclosure having regards to the disease, disorder, or condition to be treated.
[0084] In some embodiments, the one or more additional therapeutic agents include, without limitation:
(i) acetylcholinesterase inhibitors, such as donepezil hydrochloride (ARICEPT, MEMAC), physostigmine salicylate (ANTILIRIUM), physostigmine sulfate (ESERINE), metrifonate, neostigmine, ganstigmine, pyridostigmine (MESTINON), ambenonium (MYTELASE), demarcarium, Debio 9902 (also known as ZT-1; Debiopharm), rivastigmine (EXELON),
ladostigil, NP-0361, galantamine hydrobromide (RAZADYNE, RIMINYL, NIVALIN), tacrine (COGNEX), tolserine, velnacrine maleate, memoquin, huperzine A (HUP- A; NeuroHitech), phenserine, edrophonium (ENLON, TENSILON), and INM-176;
(ii) amyloid-B (or fragments thereof), such as Aβ1-15 conjugated to pan HLA DR-binding epitope (PADRE), ACC-001 (Elan/Wyeth), ACL01, ACI-24, AN- 1792, Affitope AD-01, CAD 106, and V-950;
(iii) antibodies to amyloid-B (or fragments thereof), such as ponezumab, solanezumab, bapineuzumab (also known as AAB-001), AAB-002 (Wyeth/Elan), ACI-01-Ab7, BAN-2401, intravenous Ig (GAMMAGARD), LY2062430 (humanized m266; Lilly), R1450 (Roche), ACU- 5A5, huC091, and those disclosed in International Patent Publication Nos WO04/032868, W005/025616, W006/036291, W006/069081, WO06/118959, in US Patent Publication Nos US2003/0073655, US2004/0192898, US2005/0048049, US2005/0019328, in European Patent Publication Nos EP0994728 and 1257584, and in US Patent No 5,750,349;
(iv) amyloid-lowering or -inhibiting agents (including those that reduce amyloid production, accumulation and fibrillization) such as dimebon, davunetide, eprodisate, leuprolide, SK-PC- B70M, celecoxib, lovastatin, anapsos, oxiracetam, pramiracetam, varenicline, nicergoline, colostrinin, bisnorcymserine (also known as BNC), NIC5-15 (Humanetics), E-2012 (Eisai), pioglitazone, clioquinol (also known as PBT1), PBT2 (Prana Biotechnology), flurbiprofen (ANS AID, FROBEN) and its R-enantiomer tarenflurbil (FLURIZAN), nitroflurbiprofen, fenoprofen (FENOPRON, NALFON), ibuprofen (ADVIL, MOTRIN, NUROFEN), ibuprofen lysinate, meclofenamic acid, meclofenamate sodium (MECLOMEN), indomethacin (INDOCIN), diclofenac sodium (VOLTAREN), diclofenac potassium, sulindac (CLINORIL), sulindac sulfide, diflunisal (DOLOBID), naproxen (NAPROSYN), naproxen sodium (ANAPROX, ALEVE), ARC031 (Archer Pharmaceuticals), CAD-106 (Cytos), LY450139 (Lilly), insulin-degrading enzyme (also known as insulysin), the gingko biloba extract Egb-761 (ROKAN, TEBONIN), tramiprosate (CEREBRIL, ALZHEMED), eprodisate (FIBRILLEX, KIACTA), compound W [3,5-bis(4-nitrophenoxy)benzoic acid], NGX-96992, neprilysin (also known as neutral endopeptidase (NEP)), scyllo-inositol (also known as scyllitol), atorvastatin (LIPITOR), simvastatin (ZOCOR), KLVFF-(EEX)3, SKF-74652, ibutamoren mesylate, BACE inhibitors such as ASP-1702, SCH-745966, JNJ-715754, AMG-0683, AZ-12304146, BMS- 782450, GSK-188909, NB-533, E2609 and TTP-854; gamma secretase modulators such as ELND-007; and RAGE (receptor for advanced glycation end-products) inhibitors, such as TTP488 (Transtech) and TTP4000 (Transtech), and those disclosed in US Patent No 7,285,293, including PTI-777;
(v) alpha-adrenergic receptor agonists, such as guanfacine (INTUNIV, TENEX), clonidine (CATAPRES), metaraminol (ARAMINE), methyldopa (ALDOMET, DOPAMET, NOVOMEDOPA), tizanidine (ZANAFLEX), phenylephrine (also known as neosynephrine), methoxamine, cirazoline, guanfacine (INTUNIV), lofexidine, xylazine, modafinil (PRO VIGIL), adrafinil, and armodafinil (NUVIGIL);
(vi) beta-adrenergic receptor blocking agents (beta blockers), such as carteolol, esmolol (BREVIBLOC), labetalol (NORMODYNE, TRANDATE), oxprenolol (LARACOR, TRASACOR), pindolol (VISKEN), propanolol (INDERAL), sotalol (BETAPACE, SOTALEX, SOTACOR), timolol (BLOCADREN, TIMOPTIC), acebutolol (SECTRAL, PRENT), nadolol (CORGARD), metoprolol tartrate (LOPRESSOR), metoprolol succinate (TOPROL-XL), atenolol (TENORMIN), butoxamine, and SR 59230A (Sanofi);
(vii) anticholinergics, such as amitriptyline (ELAVIL, ENDEP), butriptyline, benztropine mesylate (COGENTIN), trihexyphenidyl (ARTANE), diphenhydramine (BENADRYL), orphenadrine (NORFLEX), hyoscyamine, atropine (ATROPEN), scopolamine (TRANSDERMSCOP), scopolamine methylbromide (PARMINE), dicycloverine (BENTYL, BYCLOMINE, DIBENT, DILOMINE), tolterodine (DETROL), oxybutynin (DITROPAN, LYRINEL XL, OXYTROL), penthienate bromide, propantheline (PRO-BANTHINE), cyclizine, imipramine hydrochloride (TOFRANIL), imipramine maleate (SURMONTIL), lofepramine, desipramine (NORPRAMIN), doxepin (SINEQUAN, ZONALON), trimipramine (SURMONTIL), and glycopyrrolate (ROBINUL);
(viii) anticonvulsants, such as carbamazepine (TEGRETOL, CARBATROL), oxcarbazepine (TRILEPTAL), phenytoin sodium (PHENYTEK), fosphenytoin (CEREBYX, PRODILANTIN), divalproex sodium (DEPAKOTE), gabapentin (NEURONTIN), pregabalin (LYRICA), topirimate (TOPAMAX), valproic acid (DEPAKENE), valproate sodium (DEPACON), 1- benzyl-5-bromouracil, progabide, beclamide, zonisamide (TRERIEF, EXCEGRAN), CP- 465022, retigabine, talampanel, and primidone (MYSOLINE);
(ix) antipsychotics, including atypical antipsychotics, such as lurasidone (LATUDA, also known as SM- 13496; Dainippon Sumitomo), aripiprazole (ABILIFY), chlorpromazine (THORAZINE), haloperidol (HALDOL), iloperidone (FANAPTA), flupentixol decanoate (DEPIXOL, FLUANXOL), reserpine (SERPLAN), pimozide (ORAP), fluphenazine decanoate, fluphenazine hydrochloride, prochlorperazine (COMPRO), asenapine (SAPHRIS), loxapine (LOXITANE), molindone (MOBAN), perphenazine, thioridazine, thiothixine, trifluoperazine (STELAZINE), ramelteon, clozapine (CLOZARIL), norclozapine (ACP-104), risperidone (RISPERDAL), paliperidone (INVEGA), melperone, olanzapine (ZYPREXA), quetiapine
(SEROQUEL), talnetant, amisulpride, ziprasidone (GEODON), blonanserin (LONASEN), brexpiprazole (REXULTI), and ACP-103 (Acadia Pharmaceuticals);
(x) calcium channel blockers such as lomerizine, ziconotide, nilvadipine (ESCOR, NIVADIL), diperdipine, amlodipine (NORVASC, ISTIN, AMLODIN), felodipine (PLENDIL), nicardipine (CARDENE), nifedipine (ADALAT, PROCARDIA), MEM 1003 and its parent compound nimodipine (NIMOTOP), nisoldipine (SULAR), nitrendipine, lacidipine (LACIPIL, MOTENS), lercanidipine (ZANIDIP), lifarizine, diltiazem (CARDIZEM), verapamil (CALAN, VERELAN), AR-R 18565 (AstraZeneca), and enecadin;
(xi) catechol O-m ethyl transferase (COMT) inhibitors, such as nitecapone, tolcapone (TASMAR), entacapone (COMTAN), and tropolone;
(xii) central nervous system stimulants, such as atomoxetine, reboxetine, yohimbine, caffeine, phenmetrazine, phendimetrazine, pemoline, fencamfamine (GLUCOENERGAN, REACTIVAN), fenethylline (CAPTAGON), pipradol (MERETRAN), deanol (also known as dimethylaminoethanol), methylphenidate (DAYTRANA), methylphenidate hydrochloride (RITALIN), dexmethylphenidate (FOCALIN), amphetamine (alone or in combination with other CNS stimulants, e.g., ADDERALL (amphetamine aspartate, amphetamine sulfate, dextroamphetamine saccharate, and dextroamphetamine sulfate)), dextroamphetamine sulfate (DEXEDRINE, DEXTROSTAT), methamphetamine (DESOXYN), lisdexamfetamine (VYVANSE), and benzphetamine (DIDREX);
(xiii) corticosteroids, such as prednisone (STERAPRED, DELTASONE), prednisolone (PRELONE), predisolone acetate (OMNIPRED, PRED MILD, PRED FORTE), prednisolone sodium phosphate (ORAPRED ODT), methylprednisolone (MEDROL); methylprednisolone acetate (DEPO-MEDROL), and methylprednisolone sodium succinate (A-METHAPRED, SOLU-MEDROL);
(xiv) dopamine receptor agonists, such as apomorphine (APOKYN), bromocriptine (PARLODEL), cabergoline (DOSTINEX), dihydrexidine, dihydroergocryptine, fenoldopam (CORLOPAM), lisuride (DOPERGIN), terguride spergolide (PERMAX), piribedil (TRIVASTAL, TRASTAL), pramipexole (MIRAPEX), quinpirole, ropinirole (REQUIP), rotigotine (NEUPRO), SKF-82958 (GlaxoSmithKline), cariprazine, pardoprunox and sarizotan;
(xv) dopamine receptor antagonists, such as chlorpromazine, fluphenazine, haloperidol, loxapine, risperidone, thioridazine, thiothixene, trifluoperazine, tetrabenazine (NITOMAN, XENAZINE), 7-hy droxyam oxapine, droperidol (INAPSINE, DRIDOL, DROPLETAN), domperidone (MOTILIUM), L-741742, L-745870, raclopride, SB-277011 A, SCH-23390, ecopipam, SKF-83566, and metoclopramide (REGLAN);
(xvi) dopamine reuptake inhibitors such as bupropion, safinamide, nomifensine maleate (MERIT AL), vanoxerine (also known as GBR-12909) and its decanoate ester DBL-583, and amineptine;
(xvii) gamma-amino-butyric acid (GABA) receptor agonists, such as baclofen (LIORESAL, KEMSTRO), siclofen, pentobarbital (NEMBUTAL), progabide (GABRENE), and clomethi azole;
(xviii) histamine 3 (H3) antagonists such as ciproxifan, tiprolisant, S-38093, irdabisant, pitolisant, GSK-239512, GSK-207040, JNJ-5207852, JNJ-17216498, HPP-404, SAR-110894, trans-A-ethyl-3-fluoro-3-[3-fluoro-4-(pyrrolidin-l-ylmethyl)phenyl]-cyclobutanecarboxamide (PF-3654746 and those disclosed in US Patent Publication Nos US2005-0043354, US2005- 0267095, US2005-0256135, US2008-0096955, US2007-1079175, and US2008-0176925;
International Patent Publication Nos WO2006/136924, W02007/063385, W02007/069053, W02007/088450, W02007/099423, W02007/105053, WO2007/138431, and W02007/088462; and US Patent No 7, 115,600);
(xix) immunomodulators such as glatiramer acetate (also known as copolymer- 1; COPAXONE), MBP-8298 (synthetic myelin basic protein peptide), dimethyl fumarate, fmgolimod (also known as FTY720), roquinimex (LINOMIDE), laquinimod (also known as ABR-215062 and SAIK-MS), ABT-874 (human anti-IL-12 antibody; Abbott), rituximab (RITUXAN), alemtuzumab (CAMPATH), daclizumab (ZENAPAX), and natalizumab (TYSABRI);
(xx) immunosuppressants such as methotrexate (TREXALL, RHEUMATREX), mitoxantrone (NOVANTRONE), mycophenolate mofetil (CELLCEPT), mycophenolate sodium (MYFORTIC), azathioprine (AZASAN, IMURAN), mercaptopurine (PURI-NETHOL), cyclophosphamide (NEOSAR, CYTOXAN), chlorambucil (LEUKERAN), cladribine (LEUSTATIN, MYLINAX), alpha-fetoprotein, etanercept (ENBREL), and 4-(benzyloxy)-5-[(5- undecyl-2H-pyrrol-2-ylidene)methyl]- 1H, 1’H-2,2’ -bipyrrole (also known as PNU-156804);
(xxi) interferons, including interferon beta-la (AVONEX, REBIF) and interferon beta-lb (BETASERON, BETAFERON);
(xxii) levodopa (or its methyl or ethyl ester), alone or in combination with a DOPA decarboxylase inhibitor (e.g., carbidopa (SINEMET, CARBILEV, PARCOP A), benserazide (MADOPAR), a-methyldopa, monofluromethyldopa, difluoromethyldopa, brocresine, or m- hydroxybenzylhydrazine); (xxiii) N-methyl-D-aspartate (NMD A) receptor antagonists, such as memantine (NAMENDA, AXURA, EBIXA), amantadine (SYMMETREL), acamprosate (CAMPRAL), besonprodil, ketamine (KETALAR), delucemine, dexanabinol, dexefaroxan,
dextromethorphan, dextrorphan, traxoprodil, CP-283097, himantane, idantadol, ipenoxazone, L- 701252 (Merck), lancicemine, levorphanol (DROMORAN), LY-233536 and LY-235959 (both Lilly), methadone, (DOLOPHINE), neramexane, perzinfotel, phencyclidine, tianeptine (STABLON), dizocilpine (also known as MK-801), EAB-318 (Wyeth), ibogaine, voacangine, tiletamine, riluzole (RILUTEK), aptiganel (CERESOTAT), gavestinel, and remacimide;
(xxiii) muscarinic receptor (particularly Ml subtype) agonists, such as cevimeline, levetiracetam, bethanechol chloride (DUVOID, URECHOLINE), itameline, pilocarpine (S ALAGEN), NGX267, arecoline, L-687306 (Merck), L-689660 (Merck), furtrethonium iodide (FURAMON, FURANOL), furtrethonium benzensulfonate, furtrethonium p-toluenesulfonate, McN-A-343, oxotremorine, sabcomeline, AC-90222 (Acadia Pharmaceuticals), and carbachol (CARBASTAT, MIOSTAT, CARBOPTIC);
(xxiv) neuroprotective drugs such as bosutinib, condoliase, airmoclomol, lamotrigine, perampanel, aniracetam, minaprime, riluzole, A-hydroxy- 1,2,4, 9-tetrahydro-37/-carbazol-3- imine, desmoteplase, anatibant, astaxanthin, neuropeptide NAP (e.g., AL-108 and AL-208; both Allon Therapeutics), neurostrol, perampenel, ispronicline, bis(4-β-D-glucopyranosyloxybenzyl)- 2-β-D-glucopyranosyl-2-isobutyltartrate (also known as dactylorhin B or DHB), formobactin, xaliproden (XAPRILA), lactacystin, dimeboline hydrochloride (DIMEBON), disufenton (CEROVIVE), arundic acid (ONO-2506, PROGLIA, CEREACT), citicoline (also known as cytidine 5 ’-diphosphocholine), edaravone (RADICUT), AEOL-10113 and AEOL-10150 (both Aeolus Pharmaceuticals), AGY-94806 (also known as SA-450 and Msc-1), granulocyte-colony stimulating factor (also known as AX-200), BAY-38-7271 (also known as KN-387271; Bayer AG), ancrod (VIPRINEX, ARWIN), DP-b99 (D-Pharm Ltd), HF-0220 (17-B- hydroxy epiandrosterone; Newron Pharmaceuticals), HF-0420 (also known as oligotropin), pyridoxal 5’-phosphate (also known as MC-1), microplasmin, S-18986, piclozotan, NP031112, tacrolimus, L-seryl-L-methionyl-L-alanyl-L-lysyl-L-glutamyl-glycyl-L-valine, AC-184897 (Acadia Pharmaceuticals), ADNF-14 (National Institutes of Health), stilbazulenyl nitrone, SUN- N8075 (Daiichi Suntory Biomedical Research), and zonampanel;
(xxv) nicotinic receptor agonists, such as epibatidine, bupropion, CP-601927, varenicline, ABT- 089 (Abbott), ABT-594, AZD-0328 (AstraZeneca), EVP-6124, R3487 (also known as MEM3454; Roche/Memory Pharmaceuticals), R4996 (also known as MEM63908;
Roche/Memory Pharmaceuticals), TC-4959 and TC-5619 (both Targacept), and RJR-2403;
(xxvi) norepinephrine (noradrenaline) reuptake inhibitors, such as atomoxetine (STRATTERA), doxepin (APONAL, ADAPIN, SINEQUAN), nortriptyline (AVENTYL, PAMELOR, NORTRILEN), amoxapine (ASENDIN, DEMOLOX, MOXIDIL), reboxetine (EDRONAX,
VESTRA), viloxazine (VIVALAN), maprotiline (DEPRILEPT, LUDIOMIL, PSYMION), bupropion (WELLBUTRIN), and radaxafine;
(xxvii) phosphodiesterase (PDE) inhibitors, including but not limited to, (a) PDE1 inhibitors (e.g., vinpocetine (CAVINTON, CERACTIN, INTELECTOL) and those disclosed in US Patent No 6,235,742, (b) PDE2 inhibitors (e.g., erythro-9-(2-hydroxy-3-nonyl)adenine (EHNA), BAY 60-7550, and those described in US Patent No. 6,174,884), (c) PDE3 inhibitors (e.g., anagrelide, cilostazol, milrinone, olprinone, parogrelil, and pimobendan), (d) PDE4 inhibitors (e.g., apremilast, ibudilastroflumilast, rolipram, Ro 20-1724, ibudilast (KETAS), piclamilast (also known as RP73401), CDP840, cilomilast (ARIFLO), roflumilast, tofimilast, oglemilast (also known as GRC 3886), tetomilast (also known as OPC-6535), lirimifast, theophylline (UNIPHYL, THEOLAIR), arofylline (also known as LAS-31025), doxofylline, RPR-122818, or mesembrine), and (e) PDE5 inhibitors (e.g., sildenafil (VIAGRA, REVATIO), tadalafil (CIALIS), vardenafil (LEVITRA, VIVANZA), udenafil, avanafil, dipyridamole (PERSANTINE), E-4010, E-4021, E-8010, zaprinast, iodenafil, mirodenafil, DA-8159, and those disclosed in International Patent Applications W02002/020521, W02005/049616, W02006/120552, W02006/126081, W02006/126082, W02006/126083, and
WO2007/122466), (f) PDE7 inhibitors; (g) PDE8 inhibitors; (h) PDE9 inhibitors (e.g., BAY 73- 6691 (Bayer AG) and those disclosed in US Patent Publication Nos US2003/0195205, US2004/0220186, US2006/0111372, US2006/0106035, and USSN 12/118,062 (filed May 9, 2008)), (i) PDE10 inhibitors such as 2-({4-[l-methyl-4-(pyridin-4-yl)- 1H-pyrazol-3- yl]phenoxy}methyl)quinolin-3(4H)-one and SCH-1518291; and (j) PDE11 inhibitors;
(xxviii) quinolines, such as quinine (including its hydrochloride, dihydrochloride, sulfate, bisulfate and gluconate salts), chloroquine, sontoquine, hydroxychloroquine (PLAQUENIL), mefloquine (LARIAM), and amodiaquine (CAMOQUIN, FLAVOQUINE);
(xxix) β-secretase inhibitors, such as ASP-1702, SCH-745966, JNJ-715754, AMG-0683, AZ- 12304146, BMS-782450, GSK-188909, NB-533, LY-2886721, E-2609, HPP-854, (+)- phenserine tartrate (POSIPHEN), LSN-2434074 (also known as LY-2434074), KMI-574, SCH- 745966, Ac-rER (N2-acetyl-D-arginyl-L-arginine), loxistatin (also known as E64d), and CA074Me;
(xxx) γ-secretase inhibitors and modulators, such as BMS-708163 (Avagacest), W020060430064 (Merck), DSP8658 (Dainippon), ITI-009, L-685458 (Merck), ELAN-G, ELAN-Z, 4-chloro-N-[(2S)-3-ethyl-l-hydroxypentan-2-yl]benzenesulfonamide;
(xxxi) serotonin (5-hydroxytryptamine) 1A (5-HTIA) receptor antagonists, such as spiperone, levo-pindolol, BMY 7378, NAD-299, 5-(-)-UH-301, NAN 190, lecozotan;
(xxxii) serotonin (5-hydroxytryptamine) 2C (5-HT2c) receptor agonists, such as vabicaserin and zicronapine;
(xxxiii) serotonin (5-hydroxytryptamine) 4 (5-HT4) receptor agonists, such as PRX-03140 (Epix);
(xxxiv) serotonin (5-hydroxytryptamine) 6 (5-HT6) receptor antagonists, such as A-964324, AVI-101, AVN-211, mianserin (TORVOL, BOLVIDON, NORVAL), methiothepin (also known as metitepine), ritanserin, ALX-1161, ALX-1175, MS-245, LY-483518 (also known as SGS518; Lilly), MS-245, Ro 04-6790, Ro 43-68544, Ro 63-0563, Ro 65-7199, Ro 65-7674, SB- 399885, SB-214111, SB-258510, SB-271046, SB-357134, SB-699929, SB-271046, SB-742457 (GlaxoSmithKline), Lu AE58054 (Lundbeck A/S), and PRX-07034 (Epix);
(xxxv) serotonin (5-HT) reuptake inhibitors such as alaproclate, citalopram (CELEXA, CIPRAMIL), escitalopram (LEXAPRO, CIPRALEX), clomipramine (ANAFRANIL), duloxetine (CYMBALTA), femoxetine (MALEXIL), fenfluramine (PONDIMIN), norfenfluramine, fluoxetine (PROZAC), fluvoxamine (LUVOX), indalpine, milnacipran (IXEL), paroxetine (PAXIL, SEROXAT), sertraline (ZOLOFT, LUSTRAL), trazodone (DESYREL, MOLIPAXIN), venlafaxine (EFFEXOR), zimelidine (NORMUD, ZELMID), bicifadine, desvenlafaxine (PRISTIQ), brasofensine, vilazodone, cariprazine, 29euralstem and tesofensine;
(xxxvi) trophic factors, such as nerve growth factor (NGF), basic fibroblast growth factor (bFGF; ERSOFERMIN), neurotrophin-3 (NT-3), cardiotrophin-1, brain-derived neurotrophic factor (BDNF), neublastin, meteorin, and glial-derived neurotrophic factor (GDNF), and agents that stimulate production of trophic factors, such as propentofylline, idebenone, PYM50028 (COGANE; Phytopharm), and AIT-082 (NEOTROFIN);
(xxxii) Glycine transporter- 1 inhibitors such as paliflutine, ORG-25935, JNJ-17305600, and ORG-26041;
(xxxiii) AMPA-type glutamate receptor modulators such as perampanel, mibampator, selurampanel, GSK-729327, N-{(3S,4S)-4-[4-(5-cyanothiophen-2-yl)phenoxy]tetrahydro-furan- 3-yl}propane-2-sulfonamide, and the like;
(xxxix) Janus kinase inhibitors (JAK) such as, but not limited to, tofacitinib, ruxolitinib, baricitinib, CYT387, GLPG0634, lestaurtinib, pacritinib, and TG101348.
(xl) Interleukin- 1 receptor-associated kinase 4 inhibitors (IRAK4) such as, but not limited to, PF-06650833;
(xli) antidepressants, including, but not limited to:
(a) monoamine oxidase inhibitors (MAOIs), such as, selegiline (EMSAM), selegiline
hydrochloride (1-deprenyl, ELDEPRYL, ZELAPAR), dimethylselegilene, brofaromine, phenelzine (NARDIL), tranylcypromine (PARNATE), moclobemide (AURORIX, MANERIX), befloxatone, safinamide, isocarboxazid (MARPLAN), nialamide (NIAMID), rasagiline (AZILECT), iproniazide (MARSILID, IPROZID, IPRONID), CHF-3381 (Chiesi Farmaceutici), iproclozide, toloxatone (HUMOR YL, PERENUM), desoxypeganine, harmine (also known as telepathine or banasterine), harmaline, linezolid (ZYVOX, ZYVOXID), pargyline (EUDATIN, SUPIRDYL), metralindole (INKAZAN), pirlindole (PIRAZIDOL), and bifemelane (ALNERT, CELEPORT);
(b) norepinephrine-dopamine reuptake inhibitors (NDRIs), such as, buproprion (WELLBUTRIN, ELONTRIL);
(c) serotonin antagonist and reuptake inhibitors (SARIs), such as, nefazodone (DUTONIN, NEFADAR, SERZONE) and trazodone (DESYREL);
(d) serotonin-norepinephrine reuptake inhibitors (SNRIs), such as, desvenlafaxine (PRISTIQ), duloxetine (CYMBALTA), levomilnacipran (FETZIMA), milnacipran (IXEL, SAVELLA, MILNANEURAX), and venlafaxine (EFFEXOR, TREVILOR);
(e) selective serotonin reuptake inhibitors (SSRIs), such as, citalopram (CELEXA, CIPRAMIL), escitalopram (LEXAPRO, CIPRALEX), fluoxetine (PROZAC, SARAFEM), fluvoxamine (LUVOX, FAVERIN), paroxetine (PAXIL, SEROXAT), and sertraline (ZOLOFT, LUSTRAL);
(f) tricyclic antidepressants (TCAs), such as, amitriptyline (ELAVIL, ENDEP), amitriptylinoxide (AMIOXID, AMBIVALON, EQUILIBRIN), clomipramine (ANAFRANIL), desipramine (NORPRAMIN, PERTOFRANE), dibenzepin (NOVERIL, VICTORIL), dimetacrine (ISTONIL), dosulepin (PROTHIADEN), doxepin (ADAPIN, SINEQUAN), imipramine (TOFRANIL), lofepramine (LOMONT, GAMANIL), melitracen (DIXERAN, MELIXERAN, TRAUSABUN), nitroxazepine (SINTAMIL), nortriptyline (PAMELOR, AVENTYL), noxiptiline (SINTAMIL), opipramol (INSIDON), pipof ezine (AZAFEN/AZAPHEN), protriptyline (VIVACTIL), and trimiprame (SURMONTIL); and
(g) tetracyclic antidepressants (TeCAs), such as, am oxapine (ASENDIN), maprotiline (LUDIOMIL), mianserin (TOLVON), mirtazapine (REMERON), and setiptiline (TECIPUL). [0085] In some embodiments of this disclosure, the one or more additional therapeutic agents are neuropsychiatric agents (e.g., antipsychotics, including atypical antipsychotics, antischizophrenia agents, and/or antidepressants). In some embodiments of this disclosure, the one or more additional therapeutic agents are antipsychotics, such as risperidone, olanzapine, clozapine, quetiapine, aripiprazole, ziprasidone, paliperidone, asenapine, iloperidone, or
bexpiprazole. In some embodiments, the additional therapeutic agent is bexpiprazole. In some embodiments of this disclosure, the one or more additional therapeutic agents are antischizophrenia agents, such as ziprasidone, risperidone, olanzapine, quetiapine, aripiprazole, asenapine, blonanserin, or iloperidone, wherein the amounts of the active agent and the combination when taken as a whole are therapeutically effective for treating schizophrenia. In some embodiments of this disclosure, the one or more additional therapeutic agents are selected from antidepressants, such as the MAOIs, NDRIs, SARIs, SNRIs, SSRIs, TCAs, or TeCAs described herein above.
[0086] In other embodiments, the one or more additional therapeutic agents, are agents that are able to treat Alzheimer’s Disease and/or psychosis associated with it. In some such embodiments, the one or more additional therapeutic agents are acetylcholinesterase inhibitors and/or muscarinic receptor agonists, partial agonists, or positive allosteric modulators, such as xanomeline, donepezil, galantamine, or rivastigmine.
[0087] In some embodiments of the combinations of this disclosure, the crystalline forms of Compound A and pharmaceutical compositions of them, and the one or more additional therapeutic agents are formulated separately. In certain such embodiments, the crystalline forms of Compound A and pharmaceutical compositions of them, and the one or more additional therapeutic agents are packaged together. In some of such embodiments, the crystalline forms of Compound A and pharmaceutical compositions of them, and the one or more additional therapeutic agents are packaged separately. In some embodiments of the combinations of this disclosure, the crystalline forms of Compound A and pharmaceutical compositions of them, and the one or more additional therapeutic agents are formulated together.
[0088] In some embodiments of this disclosure, the crystalline forms of Compound A and pharmaceutical compositions of them, and the one or more additional therapeutic agents in a combination (where they are not formulated together) of this disclosure are administered simultaneously (i.e., with a time separation of no more than about 15 minutes and in some embodiments no more than 10 minutes) or sequentially (i.e., with a time separation of more than about 15 minutes and in some embodiments more than one hour or up to 12-24 hours).
Formulations and Types of Administration
[0089] In some embodiments, this disclosure provides pharmaceutical compositions comprising Compound A and one or more pharmaceutically acceptable carriers or excipients. In some embodiments, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, are
formulated in a solid form such as a capsule, tablet, pill, lozenge, cachet, powder, troche, wafer, or granule. In solid dosage forms for oral administration, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, may be mixed with one or more pharmaceutically acceptable carriers, such as sodium citrate or dicalcium phosphate, and/or any of the following: (1) fillers or extenders, such as starches, lactose, sucrose, glucose, mannitol, and/or silicic acid; (2) binders, such as, for example, carboxymethylcellulose, alginates, gelatin, polyvinyl pyrrolidone, sucrose, and/or acacia; (3) humectants, such as glycerol; (4) disintegrating agents, such as agar-agar, calcium carbonate, potato or tapioca starch, alginic acid, certain silicates, and sodium carbonate; (5) solution retarding agents, such as paraffin; (6) absorption accelerators, such as quaternary ammonium compounds; (7) wetting agents, such as, for example, cetyl alcohol and glycerol monostearate; (8) absorbents, such as kaolin and bentonite clay; (9) lubricants, such a talc, calcium stearate, magnesium stearate, solid polyethylene glycols, sodium lauryl sulfate, and mixtures thereof; and (10) coloring agents. The crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, may be mixed with one or more excipients, such as water, saline, dextrose, glycerol, ethanol, or the like and combinations thereof. In the case of capsules, tablets and pills, the pharmaceutical compositions or combinations (or components thereof) of the disclosure may also comprise buffering agents. Solid pharmaceutical compositions or combinations (or components thereof) of a similar type may also be employed as fillers in soft and hard-filled gelatin capsules using such excipients as lactose or milk sugars, as well as high molecular weight polyethylene glycols and the like. In an embodiment of the present disclosure, the pharmaceutical composition or solid formulation of the present invention may be manufactured by a conventional dry granulation manufacturing process that includes blending, milling, blend lubrication, roller compaction and milling, blend lubrication, compression, and aqueous based film coating using equipment commonly available in the pharmaceutical industry.
[0090] In some embodiments, the pharmaceutical composition comprises a core and a coating, wherein the core comprises Compound A, or a pharmaceutically acceptable salt or hydrate thereof, and one or more pharmaceutically acceptable excipients, and the coating comprises an opacifier. In some embodiments, the pharmaceutical composition is a tablet dosage form. In some embodiments, the core comprises one or more fillers or extenders, binders, humectants, disintegrating agents, and lubricants. In some embodiments, the pharmaceutical composition comprises a crystalline form of Compound A, including but not limited to Form 1, Form, 2, Form 3, or Form 4 described herein.
[0091] In some embodiments, the pharmaceutical composition comprises about 5 mg, about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, or about 50 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 85 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 5 weight percent of at least one lubricant, and (d) about 1 weight percent to about 8 weight percent of a coating, based on the total weight of the pharmaceutical composition. In some embodiments, the pharmaceutical composition comprises about 10 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 87 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition. In some embodiments, the pharmaceutical composition comprises about 15 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 87 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition. In some embodiments, the pharmaceutical composition comprises about 30 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 85 weight percent to about 93 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
[0092] In some embodiments, the coating can advantageously provide protection from light, moisture, and oxidation, as well as improving mechanical strength and making odors or taste. In some aspects, the coating comprises an opacifier, and may also incorporate further functional ingredients such as glidants, flavours and viscosity modifiers all of which are well known in the art. General details on pharmaceutical coatings can be found in Aulton’s Pharmaceutics, 5th Edition, 2018, Elsevier, at e.g. page 580-596. In some embodiments, the coatings contain opacifiers that may prevent or reduce light or UV-induced formation of impurities or degradants, by, for example, physically blocking the damaging wavelength or by providing chemical protection, such as, by scavenging oxidizing radicals. Examples of opacifiers include, bu t are
not limited to, titanium dioxide, silicon dioxide, zinc oxide, iron oxide, talc, calcium carbonate, calcium phosphate, calcium sulfate, behenic acid, aluminum silicate, aluminum hydroxide, magnesium carbonate, tartrazine, erythrosine, or a mixture thereof. In some embodiments, the opacifier may comprise from about 4 weight percent to about 25 weight percent of the coating by weight, based upon total weight of the coating composition. In an embodiment, the opacifier comprises from about 4 weight percent to about 20 weight percent of the coating by weight, based upon total weight of the coating composition. In an embodiment, the opacifier comprises about 5 weight percent to about 20 weight percent, of the coating by weight, based upon total weight of the coating composition. In an embodiment, the opacifier comprises about 5 weight percent to about 15 weight percent, or about 10 weight percent to about 15 weight percent, or about 15 weight percent to about 20 weight percent of the coating by weight, based upon total weight of the coating composition. In an embodiment, the opacifier comprises about 6 weight percent, about 14 weight percent, or about 17 weight percent of the coating by weight, based upon total weight of the coating composition. In an embodiment, the opacifier comprises from about 17 weight percent of the coating by weight, based upon total weight of the coating composition. In some embodiments, the coating comprises OPADRY or OPADRY II coating compositions commercially available from Colorcon, Inc., in Harleysville, PA, inducting but not limited to OPADRY II Blue (85F10944), OPADRY II Green, (85F91159), OPADRY II Red (85F150131-CN), OPADRY II Yellow, OPADRY II Grau. In some embodiments, the coating comprises polyvinyl alcohol, titanium dioxide, polyethylene glycol, talc, and a coloring agent. [0093] In some embodiments, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, are formulated in an aqueous or non-aqueous liquid dosage form, inducting solution, emulsion, microemulsion, suspension, syrup, pastille, or elixir. In some embodiments, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, of the disclosure are formulated in an aqueous solution. Liquid dosage forms may also comprise inert diluents commonly used in the art, such as water or other solvents, solubilizing agents and emulsifiers, such as ethyl alcohol (ethanol), isopropyl alcohol, ethyl carbonate, ethyl acetate, benzyl alcohol, benzyl benzoate, propylene glycol, 1,3-butylene glycol, oils (in particular, cottonseed, groundnut, corn, germ, olive, castor, and sesame oils), glycerol, tetrahydrofuryl alcohol, polyethylene glycols and fatty acid esters of sorbitan, and mixtures thereof.
[0094] In some embodiments, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their
components, are formulated in a suspension form. In some embodiments, the suspensions of this disclosure may comprise suspending agents such as ethoxylated isostearyl alcohols, polyoxyethylene sorbitol, and sorbitan esters, microcrystalline cellulose, aluminum metahydroxide, bentonite, agar-agar and tragacanth, and mixtures thereof.
[0095] In some embodiments, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, are formulated in a unit dosage form. In some embodiments, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, are formulated in a capsule or tablet form. [0096] The crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, may be formulated for administration by any suitable route as described herein and known in the art. In certain embodiments, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, are formulated for oral administration (including sublingual and buccal administration), In such cases, they may be in the form of, inter alia, bioadhesive polymers, tablets, patches, thin films, liquids and semisolids. Besides inert diluents, oral dosage forms of this disclosure may also include adjuvants such as wetting agents, emulsifying and suspending agents, sweetening, flavoring, coloring, perfuming, and preservative agents.
[0097] In some embodiments, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, are formulated for intraperenteral administration. In some embodiments, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, are formulated for parental administration (e.g., subcutaneously, intravenously, arterially, intradermally, intramuscularly, intraperitoneally) or intraspinal or intracerebral administration include sterile aqueous and nonaqueous injectable solutions, dispersions, suspensions or emulsions as well as sterile powders to be reconstituted in sterile injectable solutions or dispersions prior to use, which may contain antioxidants, buffers, bacteriostats, solutes which render the pharmaceutical composition or combination (or a component thereof) isotonic with the blood of the intended recipient, or suspending or thickening agents. When administered parenterally, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, may be in a pyrogen-free, physiologically
acceptable form. Techniques and formulations generally may be found in Remington’s Pharmaceutical Sciences, Meade Publishing Co., Easton, PA.
[0098] The crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, may also be formulated for respiratory administration (pulmonary and nasal delivery). Such formulations may include, but are not limited to, a variety of pressurized metered dose inhalers, dry powder inhalers, nebulizers, aqueous mist inhalers, drops, solutions, suspensions, sprays, powders, gels, ointments, and specialized systems such as liposomes and microspheres (see e.g. Owens et al, “Alternative Routes of Insulin Delivery” and Martini et al).
[0099] The crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, may also be formulated for transdermal delivery. Such formulations may include, but are not limited to, colloids, patches, and microemulsions.
[0100] Other suitable administration forms for the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, include, but are not limited to, depot injectable formulations, long-acting injectable formulations, suppositories, sprays, ointments, cremes, gels, inhalants, dermal patches, implants, devices, and formulations for ocular administration.
[0101] Where appropriate, the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, may be prepared with coatings such as enteric coatings or they may be formulated so as to provide extended release (e.g., a controlled release, a prolonged release, a sustained release, a delayed release, or a slow release) according to methods well known in the art.
[0102] Formulations of the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, may also comprise adjuvants, such as preservatives, wetting agents, emulsifying agents and dispersing agents. Prevention of the action of microorganisms may be ensured by the inclusion of various antibacterial and antifungal agents, for example, paraben, chlorobutanol, phenol sorbic acid, and the like. It may also be desirable to include isotonic agents, such as sugars, sodium chloride, and the like into the pharmaceutical compositions or combinations or components. In addition, prolonged absorption of the injectable formulation may be brought about by the inclusion of agents which delay absorption, such as aluminum monostearate and gelatin.
[0103] Pharmaceutical compositions or combinations of the disclosure can be prepared by methods well known in the art of pharmacy, see, e.g., Goodman et al., 2001; Ansel, et al., 2004; Stoklosa et al., 2001; and Bustamante, et al., 1993.
[0104] In some embodiments, formulations of the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure comprise a crystalline form of Compound A (e.g., Form 1, Form 2, Form 3, or Form 4) in an amount of 0.05 mg to 5000 mg or 5 mg to 1000 mg. In some embodiments, formulations of the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, comprise about 5 mg to 100 mg, about 10 to about 75 mg, about 10 mg to about 50 mg, about 10 mg to about 30 mg, about 10 mg to about 15 mg, and about 15 mg to about 30 mg of the crystalline forms of Compound A of this disclosure. In some embodiments, formulations of the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, comprise about 0.5 mg, about 1 mg, about 2 mg, about 2.5 mg, about 3 mg, about 5 mg, about 7.5 mg, about 10 mg, about 12.5 mg, about 15 mg, about 20 mg, about 25 mg, about 30mg, about 40 mg, about 50 mg, about 60 mg, about 75 mg, about 100 mg, about 150 mg, about 250 mg, about 500 mg, about 750 mg, about 1000 mg, about 1250 mg, about 2500 mg, about 3500 mg, or about 5000 mg of the crystalline forms of Compound A of this disclosure. In some embodiments, formulations of the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, comprise between 5 mg to 75 mg of the crystalline forms of Compound A. In some embodiments, formulations of the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, comprise between 10 mg to 30 mg of the crystalline forms of Compound A.
[0105] The present disclosure further comprises kits that are suitable for use in performing the methods of treatment herein. In one embodiment, the kit contains one of more dosage forms comprising one or more of the crystalline forms of Compound A, the pharmaceutical compositions and the combinations according to this disclosure, or one or more of their components, and one or more containers for the dosage forms, in quantities sufficient to carry out the methods of this disclosure.
Methods o f Treating Using Crystalline Forms o f Compound A
[0106] In one aspect, the present disclosure provides a method for treating a disease or disorder in a patient in need thereof, said method comprising administering to the patient a
therapeutically effective amount of at least one of a crystalline form of Compound A according to the disclosure, or a pharmaceutical composition or combination of them.
[0107] In some aspects, the present disclosure provides at least one of a crystalline form of Compound A according to the disclosure, or a pharmaceutical composition or combination of them, for use in treating a disease or disorder.
[0108] In some aspects, the present disclosure provides for the use of at least one of a crystalline form of Compound A according to the disclosure, or a pharmaceutical composition or combination of them, in the manufacture of a medicament for treating a disease or disorder.
[0109] In some embodiments, the disease or disorder is Alzheimer’s Disease, psychosis in Alzheimer’s Disease, schizophrenia or psychosis, pain, addiction, a sleep disorder, a cognitive disorder (e.g. mild cognitive impairment), Parkinson’s Disease, Parkinson’s Disease-levodopa- induced dyskinesia, Huntington’s Disease, dyskinesia (e.g., tardive dyskinesia), dry mouth, pulmonary hypertension, chronic obstructive pulmonary disease (COPD), asthma, urinary incontinence, glaucoma, Trisomy 21 (Down Syndrome), cerebral amyloid angiopathy, dementia, Hereditary Cerebral Hemorrhage with Amyloidosis of the Dutch-Type (HCHWA-D), Creutzfeld- Jakob disease, prion disorders, amyotrophic lateral sclerosis, progressive supranuclear palsy, head trauma, stroke, pancreatitis, inclusion body myositis, other peripheral amyloidoses, diabetes, autism, or atherosclerosis.
[0110] In some embodiments, the disease or disorder is Alzheimer’s disease, dementia, a bipolar disorder, schizophrenia, pain, an addiction, a sleep disorder, or a condition associated therewith. For example, in some embodiments, the disease or disorder is Alzheimer’s disease or dementia, psychosis in Alzheimer’s disease or dementia, or agitation in Alzheimer’s disease or dementia. In certain other embodiments, the disease or disorder is a bipolar disorder or depressive episodes associated therewith. When the disease or disorder is a bipolar disorder or depressive episodes associated therewith, treatment may include, for example, maintenance treatment or acute treatment of manic or mixed episodes.
[0111] The present disclosure further provides methods of treating a disease or disorder in a patient in need thereof, said method comprising administering to the patient a therapeutically effective amount of at least one of a crystalline form of Compound A according to the disclosure, or a pharmaceutical composition or combination of them, wherein no dose titration is required. In some embodiments, the disease or disorder is selected from the group consisting of: schizophrenia, psychosis (including Alzheimer’s disease psychosis), tardive dyskinesia, bipolar disorder (bipolar I or bipolar II), and bipolar depression. In some embodiments, the
therapeutically effective amount of the crystalline forms of Compound A of this disclosure is about 5 mg, about 10 mg, about 15 mg, about 30 mg, about 50 mg, or about 60 mg daily.
[0112] The present disclosure provides methods of treating schizophrenia in a patient in need thereof, said method comprising administering to the patient a total daily dose of about 5 mg, 10 mg, about 15 mg, about 30 mg, or about 50 mg of the crystalline forms of Compound A of this disclosure, or a pharmaceutical composition or combination of them, once daily or multiple times daily, wherein in some embodiments, no dose titration is required.
[0113] The present disclosure provides methods of treating Alzheimer’s disease psychosis in a patient in need thereof, said method comprising administering to the patient a total daily dose of about 1 mg, about 2 mg, about 2.5 mg, about 5 mg, 10 mg, about 15 mg, about 30 mg, or about 50 mg, once daily or multiple times daily, of a crystalline form of Compound A according to the disclosure, or a pharmaceutical composition or combination thereof, wherein in some embodiments, no dose titration is required.
[0114] The present disclosure provides methods of treating tardive dyskinesia in a patient in need thereof, said method comprising administering to the patient a total daily dose of about 1 mg, about 2 mg, about 2.5 mg, about 5 mg, 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 50 mg, or about 60 mg, once daily or multiple times daily, of a crystalline form of Compound A according to the disclosure, or a pharmaceutical composition or combination thereof, wherein in some embodiments, no dose titration is required.
[0115] The present disclosure provides methods of treating bipolar disorder (such as bipolar I or bipolar II) in a patient in need thereof, said method comprising administering to the patient a total daily dose of about 1 mg, about 2 mg, about 2.5 mg, about 5 mg, 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 50 mg, or about 60 mg, once daily or multiple times daily, of a crystalline form of Compound A, or a pharmaceutical composition or combination thereof, wherein in some embodiments, no dose titration is required.
Methods of Producing Crystalline Forms of Compound A
[0116] Various methods may be used to produce the crystalline forms (including Forms 1, 2, 3 and 4) of Compound A. Accordingly, the crystalline Forms 1, 2, 3, and 4 of Compound A of this disclosure are not limited by any particular process by which they are produced. Rather, they are characterized by their physical properties, including, but not limited to, one or more of their XRPD pattern and major peaks therein, TGA thermograms, DSC thermograms, DVS plots, FT Raman spectra, and PLM images of this disclosure.
[0117] In some embodiments, the methods of the present disclosure are directed to a method of producing Form 1 of Compound A, the method comprising slurrying the compound in a solvent selected from acetone, acetonitrile, 1 -butanol, 1,4-di oxane, dimethylformamide, ethanol, ethyl acetate, ethylene glycol, methyl ethyl ketone, methanol, methyl isobutyl ketone, 1- propanol, 2-propanol, water, or a mixture of acetonitrile-water at ambient temperature, 5 °C or 50 °C.
[0118] In some embodiments according to the disclosure, the solvent for slurrying the compound is selected from acetone, acetonitrile, 1-butanol, 1,4-dioxane, dimethylformamide, ethanol, ethyl acetate, ethylene glycol, methyl ethyl ketone, methanol, methyl isobutyl ketone, 1- propanol, 2-propanol, water, or a mixture of acetonitrile-water at ambient temperature, 5 °C or 50 °C In some embodiments, the compound is slurried in a solvent selected from acetonitrile or 2-propanol at 5 °C. In some embodiments, the compound is slurried in a solvent selected from acetonitrile or 2-propanol at 50 °C. In some embodiments, the solvent is acetonitrile. In some embodiments, the solvent is 2-propanol.
[0119] In some embodiments of the disclosure, the compound is slurried in a mixture of acetonitrile-water at ambient temperature, wherein the mixture comprises about 99.3% of acetonitrile by volume.
[0120] In some embodiments of the disclosure, the compound is slurried in a solvent selected from acetone, acetonitrile, 1-butanol, 1,4-dioxane, dimethylformamide, ethanol, ethyl acetate, ethylene glycol, methyl ethyl ketone, methanol, methyl isobutyl ketone, 1 -propanol, 2-propanol, water at ambient temperature. In some embodiments, the compound is slurried in a solvent selected from 1-butanol, ethanol, ethylene glycol, methanol, 1 -propanol, or 2-propanol, at ambient temperature. In some embodiments, the compound is slurried in a solvent selected from acetone, ethyl acetate, methyl ethyl ketone or methyl isobutyl ketone at ambient temperature. In some embodiments, the compound is slurried in a solvent selected from 1,4-dioxane or ethyl acetate, at ambient temperature. In some embodiments, the compound is slurried in a solvent selected from acetonitrile, dimethylformamide or water at ambient temperature. In some embodiments, the compound is slurried in a solvent selected from acetonitrile, dimethylformamide or water at ambient temperature.
[0121] In some embodiments, the present disclosure is directed to a method of producing Form 1 of Compound A, the method comprising the steps of: (i) dissolving the compound in a 2- propanol at 65-75 °C; (ii) cooling the mixture to ambient temperature; (iii) filtering the slurry; and (iv) drying the collected solids.
[0122] In another embodiment, this disclosure provides a method of producing a monohydrate crystalline form of Compound A (e.g., Form 2), the method comprising slurrying the compound in a mixture selected from: (a) acetonitrile-water at ambient temperature, wherein the mixture comprises about 99.7 % of acetonitrile by volume; (b) acetonitrile-water at 5 °C, wherein the mixture comprises about 99.3% of acetonitrile by volume; or (c) acetonitrile-water at 50 °C, wherein the mixture comprises about 97.2% of acetonitrile by volume.
[0123] In another embodiment, the compound is dissolved in acetonitrile followed by a slow evaporation. In another embodiment, the compound is dissolved in isopropyl alcohol or acetone followed by a slow evaporation. In another embodiment, the compound is dissolved in ethyl acetate followed by a slow evaporation.
[0124] In another embodiment, the compound slurried in the acetonitrile-water mixture at ambient temperature comprises about 99.7% of acetonitrile by volume. In another embodiment, the compound slurried in the acetonitrile-water mixture at 5 °C comprises about 99.3% of acetonitrile by volume. In another embodiment, the compound slurried in the acetonitrile-water mixture at 50 °C comprises about 97.2% of acetonitrile by volume.
[0125] In some embodiments, the present disclosure is directed to a method of producing Form 2 of Compound A, the method comprising the steps of: (i) dissolving the compound in a 2- propanol at 65-75 °C; (ii) cooling the mixture to 35-45 °C; (iii) slowly adding water; (iv) filtering the slurry; and (v) drying the collected solids.
[0126] In another embodiment, the disclosure provides a method of producing a dihydrate crystalline form, Form 3, of Compound A the method comprising slurrying the compound in a solvent selected from 2-methyl tetrahydrofuran, tetrahydrofuran, water, toluene at ambient temperature, 5 °C or 50 °C. In another embodiment, the disclosure provides a method of producing a dihydrate crystalline form, Form 3, of Compound A the method comprising slurrying the compound in a mixture selected from: (a) acetonitrile-water at ambient temperature, wherein the mixture comprises about 38.4%, 79.2%, 92.1%, 95.6%, 97.2%, 98.1%, 98.8% of acetonitrile by volume; (b) acetonitrile-water at 5°C, wherein the mixture comprises about 79.2% or 97.2% of acetonitrile by volume; or (c) acetonitrile-water at 50 °C, wherein the mixture comprises about 79.2% of acetonitrile by volume.
[0127] In another embodiment, the compound is slurried in a solvent selected from 2-methyl tetrahydrofuran, tetrahydrofuran, water, or toluene at ambient temperature. In another embodiment, the compound is slurried in 2-methyl tetrahydrofuran or tetrahydrofuran at ambient temperature. In another embodiment, the compound is slurried in water at ambient temperature. In another embodiment, the compound is slurried in toluene at ambient temperature. In another
embodiment, the compound is slurried in water at 50 °C. In another embodiment, the compound is slurried in water at 5 °C. In another embodiment, the compound is slurried in water at 50 °C and the slurry is filtered after a week.
[0128] In another embodiment, the compound is slurried in an acetonitrile-water mixture at ambient temperature wherein the acetonitrile-water mixture comprises about 38.4%, 79.2%, 92.1%, 95.6%, 97.2%, 98.1%, 98.8% of acetonitrile by volume. In another embodiment, the acetonitrile-water mixture comprises about 38.4% of acetonitrile by volume. In another embodiment, the acetonitrile-water mixture comprises about 79.2% of acetonitrile by volume. In another embodiment, the acetonitrile-water mixture comprises about 92.1% of acetonitrile by volume. In another embodiment, the acetonitrile-water mixture comprises about 95.6% of acetonitrile by volume. In another embodiment, the acetonitrile-water mixture comprises about 97.2% of acetonitrile by volume. In another embodiment, the acetonitrile-water mixture comprises about 98.1% of acetonitrile by volume. In another embodiment, the acetonitrile-water mixture comprises about 98.8% of acetonitrile by volume. In another embodiment, the compound slurried in the acetonitrile-water mixture at 5 °C comprises about 79.2% or 97.2% of acetonitrile by volume. In another embodiment, the acetonitrile-water mixture comprises about 79.2% of acetonitrile by volume. In another embodiment, the acetonitrile-water mixture comprises about 97.2% of acetonitrile by volume. In another embodiment, the compound slurried in the acetonitrile-water mixture at 50 °C comprises about 79.2% of acetonitrile by volume.
[0129] In some embodiments, the present disclosure is directed to a method of producing Form 3 of Compound A, the method comprising the steps of (i) dissolving the compound in a 2- propanol at 65-75 °C; (ii) cooling the mixture to 50-55 °C; (iii) slowly adding water; (iv) adding a seed crystal; (v) slowly adding water; (vi) cooling the mixture to ambient temperature; (vii) filtering the slurry; and (viii) drying the collected solids.
[0130] In another embodiment, the disclosure provides a method of producing an anhydrous crystalline form, Form 4, of Compound A the method comprising slurrying the compound in a mixture of acetonitrile-water at 50 °C, wherein the mixture comprises about 79.2% of acetonitrile by volume.
[0131] In some embodiments, the present disclosure is directed to a method of producing Form 4 of Compound A, the method comprising the steps of (i) dissolving the compound in a 2- propanol at 70-80 °C; (ii) cooling the mixture to 45-50 °C; (iii) filtering the slurry; and (iv) drying the collected solids.
[0132] It will be understood by one of ordinary skill in the art that the methods described herein may be adapted and modified as is appropriate for the application being addressed and that the methods described herein may be employed in other suitable applications, and that such other additions and modifications will not depart from the scope hereof.
[0133] This disclosure will be better understood from the Examples and experimental details which follow. However, one skilled in the art will readily appreciate that the specific methods and results discussed are merely illustrative of the invention as described more fully in the embodiments which follow thereafter.
EXAMPLES
[0134] This invention will be better understood from the Examples which follow. However, one skilled in the art will readily appreciate that the specific methods and results discussed are merely illustrative of the invention as described more fully in the embodiments described herein above and those which follow thereafter.
[0135] A synthetic procedure has been described for Compound A (see, WO2018/002760).
Example 1. Instruments and Methodology
A. X-Ray Powder Diffraction (XRPD)
[0136] XRPD patterns (see, Figures 1, 7, 10, and 15) were collected in reflection mode using a D8 ADVANCE diffractometer (Bruker, Germany) with Cu Kα (λ = 1.5406 Å, generator settings: 40 kV and 40 mA) radiation at RT. The sample was prepared on a polished, zerobackground sample holder by gently pressing onto the flat surface. Measurements were carried out from 3 to 42° (2θ), at a scanning speed of 0.05 s/step and step size of 0.02° (2θ). The diffraction patterns were analyzed using DIFFRAC.EVA (V4.3.1).
B. Thermogravimetric Analysis
[0137] TGA analysis was performed on a Discovery TGA550 (TA Instruments, USA).
Typically, 1 - 5 mg of each sample was loaded onto a pre-tared aluminium oxide pan and heated at 10 °C/min from room temperature to 300 °C. A nitrogen purge at 60 mL/min was maintained over the sample. The TGA data were analyzed using TRIOS. (V4.4.4.41651).
C. Thermogravimetric Analysis - TGA-IR method
[0138] A TA Instruments Discovery TGA was used for the thermal analysis. The specific TGA-IR method for the TGA reduces the standard flow rate from 50 mL/minute to 25 mL/minute in order to increase the resonance time of the evolved gas in the gas cell. The ramp rate and final temperature for heating were controlled in the TGA software (typically 10
°C/minute). Sample size was typically in the 10-15 mg range. The TGA method was set up with the following parameters: Mass flow rate of 25 mL/min, event 1 “on”, ramp 10 °C/min up to desired temperature, event 1 “off”.
[0139] A Thermo 6700 FT-IR spectrometer equipped with a KBr beamsplitter and a d-TGS KBr detector was utilized for TGA-IR analysis. The collection range was 4000-400 cm-1 and Happ-Genzel apodization was used for data collection. Background spectra were collected at 4 cm-1 resolution with 64 co-added scans. The IR background was collected with the TGA furnace closed and purged for 2-3 minutes. The sample method was set up for quick spectral collection, as the evolved gases were swept quickly through the gas cell. Each sample spectral data point was a co-added spectrum of five spectra, collected with 4 cm-1 resolution. The total IR collection time was adjusted based on the run length of the TGA.
D. Differential Scanning Calorimetry
[0140] DSC thermograms were recorded on a Discovery DSC25 (TA Instruments, USA) equipped with a RCS90 chiller. Typically, samples of approximately 0.5 - 5 mg were heated at a rate of 10 °C/min in pin-holed aluminum pans from 30 °C to the final temperature. Measurements were performed at a constant nitrogen flow rate of 50 mL/min. The DSC data were analyzed using TRIOS (V4.4.4.41651).
E. Dynamic vapor sorption (DVS)
[0141] DVS data was collected on a TGA Q5000 V4.5A by TA Instruments or a VTI SGE-CX by TA Instruments (see, Figures 4, 13, and 18). The microbalance was calibrated using a 100 mg standard weight. The relative humidity (RH) sensor was calibrated at 5.0, 11.3, 32.8, 52.8, 75.3, and 84.3% RH (25 °C) using saturated salt solutions, as well as 80% RH (25 °C) using polyvinylpyrrolidone. 8-10 mg of the powder sample was placed in the platinum sample pan. The RH was first maintained at 30% RH, then decreased to 10% RH, and then 3%RH with a maximum hold time of 120 min at each step. The attainment of equilibrium at each RH step was assumed when the weight change of the sample was < 0.00 lwt% in 5 min. The RH was then progressively increased to 90% in increments of 10% followed by a decreased to a final RH of 10% in 10% RH decrements with a maximum hold time of 120 min at each step. The attainment of equilibrium was assumed when the weight change of the sample was < 0.001wt% in 5 min. The weight gain at the end of the sorption cycle (90% RH) was calculated on the basis of the initial weight at 30% RH.
F. Fourier Transformed (FT) Raman Spectroscopy
[0142] The FT Raman spectra depicted in Figure 5 was obtained on a Kaiser Optical Systems dispersive Raman microscope equipped with a thermoelectrically cooled CCD detector. The
source was a 400 mW diode laser operating at 785 nm, with an average power output of 100 mW through a 50 x 11 mm working distance objective. The spot size of the laser beam through the objective was approximately 20 m. The microscope was coupled to the spectrometer with multi-mode fiber optic cables. The collection range was 3500-50 cm-1. The collection (exposure) time and number of accumulations is adjusted based on the efficiency of the Raman scattering of the sample. For most non-fluorescent samples, an exposure time of 10 seconds with one accumulation proved adequate for spectral collection.
[0143] The FT Raman spectrum depicted in Figure 19 was collected on a Nicolet NXR FT- Rama accessory attached to the FT-IR bench. The spectrometer was equipped with a 1064 nm Nd: YV04 laser and an indium gallium arsenide (InGaAs) detector. Prior to data acquisition, instrument performance and calibration verifications were conducted using polystyrene.
Samples were analyzed in glass NMR tubes that were spun during spectral collection. The spectra were collected using 0.5 W of laser power and 128 co-added scans. The collection range was 3500-50 cm-1. The sample spectra were recorded using 4 cm-1 resolution, and Happ-Genzel apodization was utilized for all of the spectra. Multiple spectra were recorded and the reported spectrum is an average of four spots.
G. Polarized Light Microscopy (PLM)
[0144] The polarized light microscopy images depicted in Figures 6, 14, and 20 were captured on an Olympus BX50 or BX60, Olympus BH2, Leitz Orthoplan, or a Nikon Eclipse E600 POL optical microscope, which are considered to be functionally and operationally comparable. Samples were dispersed in silicone oil or analyzed as a dry powder on a flat glass substrate. Samples were viewed under plane and/or cross-polarized light with a 530 nm wave plate.
Example 2. Polymorph Screening
[0145] Polymorph screening experiments were performed using different crystallization or solid transition methods. The methods and solvents utilized, and crystalline forms identified therefrom are summarized in Table 1.
[0146] Compound A free base was used as received (ETS purity) for the enabling polymorph screen. Approximately 50 mg of Form 1 was added to 1.5 mL HPLC vials followed by addition of ~1.0 mL solvent or binary solvent/water mixtures. The slurry experiments were set up at room temperature (RT), 5 °C and 50 °C. Additional sample was added as needed to ensure saturation. After 2 weeks of stirring with a magnetic stir bar, small aliquots from each slurry were filtered using 0.45 pM nylon centrifuge filters. The solids were analyzed via powder X-ray diffractometry. Gravimetric solubility was measured only for samples that were slurried at RT.
The final form was evaluated by powder X-ray diffractometry. The results of these screening experiments yielded a total of four unique forms: Form 1 (anhydrate), Form 2 (monohydrate), Form 3 (dihydrate), and Form 4 (anhydrate). Table 1. Polymorph Screening Experiments for Compound A
Slow Evaporation
[0147] Solutions were prepared in selected solvents and agitated between aliquot additions to assist in dissolution. Once a mixture reached complete dissolution, as judged by visual observation, the solution was filtered through a 0.2-pm nylon filter into a sample vial. The vial opening was covered with foil and pierced 3x to allow slow evaporation at ambient. The solids that formed were isolated for evaluation.
Slurry
[0148] Solutions were prepared by adding enough solids to a given solvent so that excess solids were present. The mixture was then agitated in a sealed vial at either ambient or an elevated temperature. After a given amount of time, the solids were isolated for analysis.
Solubility Estimation
[0149] Approximate Solubility Assessment performed in connection with the polymorph screen was conducted using ~ 27 solvents. 2-3 mg of a given material was added into each vial. Solvents were added in the following increments: 10μl, 10μl, 20μl, 20 μl, 40μl, 100μl, 100μl, 200μl, and 500μl. Between each addition, visual observations were made to determine if the compound has completely dissolved into the given amount of solvents. Based on the amount of the given material added and the total amount of solvents added, approximate solubility values were generated.
[0150] Forms 1 and 4 are anhydrous forms of Compound A; Forms 2 and 3 are hydrates. Crystalline Form 3 (dihydrate) of Compound A was identified as the most thermodynamically stable, relative to the other polymorphic forms of Compound A. Characterization data are discussed in more detail below.
Form 1, Anhydrate
[0151] Form 1 is a crystalline anhydrate of Compound A that exhibits birefringence with extinction and very small plate shaped particles.
[0152] Form 1 was obtained by dissolving Compound A (1.0 eq.) in 2-propanol (8.0 vol.) and heating the resulting solution to 65-75 °C. The mixture was then cooled to ambient temperature, resulting in the precipitation of a solid. The solids were collected via vacuum filtration and dried under vacuum at 50 °C to afford Form 1.
[0153] An XRPD pattern for Form 1 is provided in Figure 1, and the observed peaks are summarized in Table 2.
Table 2. Observed XRPD peaks for Form 1 of Compound A.
[0154] TGA and DSC thermograms of Form 1 are depicted in Figures 2 and 3, respectively. The TGA thermogram shows minimal (<1%) weight loss up to ~200 °C, consistent with an anhydrous form. The DSC thermogram exhibits an endotherm with an onset of ~183 °C assigned to a melt event.
[0155] The DVS plot suggests that Form 1 exhibits low hygroscopicity (see, Figure 4).
Specifically, the weight change through the sorption/desorption cycle was negligible (<0.3 wt% at 90% RH) with no hysteresis. This data indicates that hydrate formation is not favorable at high relative humidity.
[0156] Additional techniques were utilized to characterize crystalline Form 1. Specifically, Form 1 was further characterized using Raman spectroscopy (Figure 5) and microscopy (Figure 6).
Form 2, Monohydrate
[0157] Form 2 is a crystalline monohydrate of Compound A.
[0158] Form 2 was obtained by dissolving Compound A (1.0 eq.) in 2-propanol (8.0 vol.) and heating the resulting solution to 65-75 °C. The mixture was then cooled to 35-45 °C, and water (0.5 vol.) was added drop-wise to the mixture. The mixture was maintained at 35-45 °C, resulting in the precipitation of solids. The solids were collected via vacuum filtration and dried under vacuum at 50 °C to afford Form 2.
[0159] An XRPD pattern for Form 2 is provided in Figure 7, and the observed peaks are summarized in Table 3.
[0160] TGA and DSC thermograms for Form 2 are depicted in Figures 8 and 9, respectively. The TGA data showed about 8% weight loss up to ~200 °C. The weight loss was confirmed to be due to water loss by TGA-IR. Those data are consistent with a hydrated form. The DSC curve showed a prolonged first endotherm followed by two more endothermic events with onsets at ~83 °C and ~99 °C, respectively.
Form 3, Dihydrate
[0161] Form 3 is a crystalline dihydrate of Compound A that exhibits birefringence with extinction and very small plate shaped particles. [0162] Form 3 was obtained by dissolving Compound A (1.0 eq.) in 2-propanol (13.0 vol.) and heating the resulting solution to 65-75 °C. The mixture was then cooled to 50-55 °C and water (3.9 vol.) was added drop-wise to the mixture. A seed crystal of Form 3 (0.5% w/w) was then added to the mixture, which was maintained at 50-55 °C for 1-2 hours. Water (12.1 vol.) was again added to the mixture drop-wise and the mixture was cooled to ambient temperature, resulting in solid formation. The solids were collected via vacuum filtration and dried under wet nitrogen gas (RH = 40-60%) at 50 °C to afford Form 3.
[0163] Exemplary XRPD patterns for Form 3 are provided in Figure 10, and the observed peaks are summarized in Tables 4 & 5.
0164] TGA and DSC thermograms of Form 3 are depicted in Figures 11 and 12, respectively.
The TGA data showed about 8% weight loss up to ~200 °C. The weight loss was confirmed to be due to water loss by TGA-IR. Those data are consistent with a hydrated form. The DSC curve showed a broad endotherm with an onset at ~80 °C assigned to a dehydration event, followed by two more endothermic events with onsets at ~182 °C and ~193 °C, respectively. [0165] Additional techniques were utilized to characterize crystalline Form 3. Specifically, Form 3 was further characterized using DVS (Figure 13) and microscopy (Figure 14).
Form 4, Anhydrate
[0166] Form 4 is an anhydrate of Compound A that exhibits birefringence with extinction and irregular shaped particles with soft agglomerates.
[0167] Form 4 was obtained by dissolving Compound A (1.0 eq.) in isopropanol (5.0 vol.) and heating the resulting solution to 70-80 °C. The mixture was then cooled to 45-50 °C and maintained at that temperature overnight, resulting in the precipitation of solids. The solids were collected via vacuum filtration and dried under vacuum at 50 °C to afford Form 4. [0168] An XRPD pattern for Form 4 is provided in Figure 15, and the observed peaks are summarized in Table 6.
[0169] TGA and DSC thermograms of Form 4 are depicted in Figures 16 and 17, respectively. The TGA thermogram shows minimal (<1%) weight loss up to ~200 °C, consistent with an anhydrous form. The DSC thermogram exhibits a sharp endotherm with an onset of ~191 °C assigned to a melt event.
[0170] The DVS plot suggests that Form 4 exhibits low hygroscopicity (see, Figure 18). Specifically, the weight change through the sorption/desorption cycle was negligible (<0.15 wt% at 90% RH) with no hysteresis. This data indicates that hydrate formation is not favorable at high relative humidity.
[0171] Additional techniques were utilized to characterize crystalline Form 4. Specifically, Form 4 was further characterized using Raman spectroscopy (Figure 19) and microscopy (Figure 20).
Example 3. Relative Thermodynamic Stability
[0172] Based on the water activity and slurry experiments conducted to date, Form 3 is the most thermodynamically stable form. Form 3 is stable over a wide range of temperatures and water vapor pressures. This form is chemically and physically stable at 70 °C/75% RH for 1 week. It is also stable in 0.5% methocel suspension formulation (up to 14 days) and from aqueous solubility studies. Form 3 is stable at and above 30% relative humidity.
[0173] Water activity slurries utilizing Form 3 were conducted in two solvent systems to understand form conversion propensity. Approximately 50 mg of Form 3 was added to 1.5 mL HPLC vials followed by addition of ~1.0 mL solvent or binary solvent/water mixtures. Based on the comprehensive screening, Form 3 is the most stable form at normal manufacturing and storage conditions identified to date.
Example 4. Photostability of Drug Product [0174] Coating Effect on Light Exposure Uncoated tablets comprising 30 mg of Compound A (Form 3) were exposed to ICH Photostability conditions and tested to provide baseline information for comparison. Impurities/degradants/related compounds were detected at Relative Retention Time (RRT) = 1.09 and RRT = 1.16, and they were each in an amount of 0.05% or higher at time zero and 0.08% or higher at 7 days, under ICH Option 2 guidelines (UV/25°C/60% RH) in a quartz petri dish. Tablets coated with a coating composition comprising titanium dioxide (Opadry II Red, 85F150131-CN Red) were tested under similar conditions (ICH Option 2 guidelines (UV/25°C/60% RH) in a quartz petri dish), and the following results were found.
[0175] It will be apparent to those skilled in the art that various modifications and variations can be made in the present invention without departing form the scope or spirit of the invention. Other embodiments of the invention will be apparent to those skilled in the art from consideration of the specification and practice of the invention disclosed herein. It is intended that the specification, including its embodiments and examples, be considered as exemplary only.
ENUMERATED EMBODIMENTS
[0177] Embodiment 2. The crystalline form of Compound A according to embodiment 1, characterized by an XRPD pattern comprising peaks selected from at 12.7, 12.9, 19.1, 19.7, and 24.3 ± 0.2 degrees 2θ.
[0178] Embodiment 3. The crystalline form of Compound A according to embodiment 1, characterized by one or more of a. an XRPD pattern substantially as set forth in Figure 1; b. a TGA thermogram substantially as set forth in Figure 2; c. a DSC thermogram substantially as set forth in Figure 3; d. a DVS plot substantially as set forth in Figure 4; e. a FT Raman spectrum substantially as set forth in Figure 5; and f. a PLM image substantially as set forth in Figure 6.
[0180] Embodiment 5. The crystalline form of Compound A according to embodiment 4, characterized by an XRPD pattern comprising peaks at 10.2, 11.5, 17.5, 19.9, and 23.1 ± 0.2 degrees 2θ.
[0181] Embodiment 6. The crystalline form of Compound A according to embodiment 4, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 7; b. a TGA thermogram substantially as set forth in Figure 8; and c. a DSC thermogram substantially as set forth in Figure 9.
[0183] Embodiment 8. The crystalline form of Compound A according to embodiment 7, characterized by an XRPD pattern comprising peaks at 10.5, 12.0, 17.7, 20.6, and 24.0 ± 0.2 degrees 2θ.
[0184] Embodiment 9. The crystalline form of Compound A according to embodiment 7, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 10; b. a TGA thermogram substantially as set forth in Figure 11; c. a DSC thermogram substantially as set forth in Figure 12; d. a DVS plot substantially as set forth in Figure 13; and e. a PLM image as set forth in Figure 14.
[0186] Embodiment 11. The crystalline form of Compound A according to embodiment 10, characterized by an XRPD pattern comprising peaks at 12.9, 16.4, 19.5, 20.3, and 24.0 ± 0.2 degrees 2θ.
[0187] Embodiment 12. The crystalline form of Compound A according to embodiment 10, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 15; b. a TGA thermogram substantially as set forth in Figure 16; c. a DSC thermogram substantially as set forth in Figure 17; d. a DVS plot substantially as set forth in Figure 18;
e. a FT Raman spectrum substantially as set forth in Figure 19; and f. a PLM image as set forth in Figure 20.
[0188] Embodiment 13. A pharmaceutical composition comprising one or more of a crystalline form of Compound A according to any one of embodiments 1-12, and at least one pharmaceutically acceptable carrier or excipient.
[0189] Embodiment 14. The pharmaceutical composition according to embodiment 13, wherein the pharmaceutical composition is formulated as a tablet, capsule, pill, lozenge, powder, granule, solution or suspension.
[0190] Embodiment 15. The pharmaceutical composition according to embodiment 13 or 14, wherein the pharmaceutical composition is in an extended-release form, a non-extended release form, or an immediate release form.
[0191] Embodiment 16. The pharmaceutical composition according to embodiment 15, wherein the pharmaceutical composition is in an extended release form.
[0192] Embodiment 17. The pharmaceutical composition according to embodiment 15, wherein the pharmaceutical composition is in a non-extended release form.
[0193] Embodiment 18. The pharmaceutical composition according to embodiment 15, wherein the pharmaceutical composition is in an immediate release form.
[0194] Embodiment 19. A pharmaceutical combination comprising one or more of a crystalline form of Compound A according to any one of embodiments 1-12, or one or more of a pharmaceutical composition according to any one of embodiments 13-18, and one or more additional therapeutic agents.
[0195] Embodiment 20. The pharmaceutical combination according to embodiment 19, wherein the one or more additional therapeutic agents are selected from risperidone, olanzapine, clozapine, quetiapine, aripiprazole, ziprasidone, paliperidone, asenapine, iloperidone, or bexpiprazole.
[0196] Embodiment 21. The pharmaceutical combination according to embodiment 19, wherein the one or more additional therapeutics agents are selected from ziprasidone, risperidone, olanzapine, quetiapine, aripiprazole, asenapine, blonanserin, or iloperidone.
[0197] Embodiment 22. The pharmaceutical combination of embodiment 19, wherein the one or more additional therapeutic agents are selected from selegiline, selegiline hydrochloride, dimethylselegilene, brofaromine, phenelzine, tranylcypromine, moclobemide, befloxatone, safinamide, isocarboxazid, nialamide, rasagiline, iproniazide, CHF-3381, iproclozide, toloxatone, desoxypeganine, harmine, harmaline, linezolid, pargyline, metralindole, pirlindole, bifemelane, bupropion, nefazodone, trazodone, desvenlafaxine, duloxetine, levomilnacipran, milnacipran, venlafaxine, citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline, amitriptyline, amitriptylinoxide, clomipramine, desipramine, dibenzepin, dimetacrine, dosulepin, doxepin, imipramine, lofepramine, melitracen, nitroxazepine, nortriptyline, noxiptiline, opipramol, pipofezine, protriptyline, trimiprame, amoxapine, maprotiline, mianserin, mirtazapine, or setiptiline.
[0198] Embodiment 23. The pharmaceutical combination of embodiment 19, wherein the one or more additional therapeutic agents are selected from xanomeline, donepezil, galantamine, or rivastigmine.
[0199] Embodiment 24. A method for treating a disease or disorder in a patient in need thereof, said method comprising administering to the patient a therapeutically effective amount of one or more of a crystalline form of Compound A according to any one of embodiments 1-12, a pharmaceutical composition of them according to any one of embodiments 13-18, or a pharmaceutical combination of them according to any one of embodiments 19-23.
[0200] Embodiment 25. The method according to embodiment 24, wherein the disease or disorder is selected from Alzheimer’s Disease, psychosis in Alzheimer’s Disease, schizophrenia or psychosis, pain, addiction, a sleep disorder, a cognitive disorder (e.g. mild cognitive impairment), Parkinson’s Disease, Parkinson’s Disease-levodopa-induced dyskinesia, Huntington’s Disease, dyskinesia, dry mouth, pulmonary hypertension, chronic obstructive pulmonary disease (COPD), asthma, urinary incontinence, glaucoma, Trisomy 21 (Down Syndrome), cerebral amyloid angiopathy, dementia, Hereditary Cerebral Hemorrhage with Amyloidosis of the Dutch-Type (HCHWA-D), Creutzfeld-Jakob disease, prion disorders, amyotrophic lateral sclerosis, progressive supranuclear palsy, head trauma, stroke, pancreatitis, inclusion body myositis, other peripheral amyloidoses, diabetes, autism, tardive dyskinesia, or atherosclerosis.
[0201] Embodiment 26. The method according to embodiment 24, wherein the disease or disorder is selected from Alzheimer’s disease, dementia, a bipolar disorder, schizophrenia, pain, an addiction, a sleep disorder, and conditions associated therewith.
[0202] Embodiment 27. The method according to embodiment 26, wherein the disease or disorder is selected from the group consisting of Alzheimer’s Disease or dementia, psychosis in Alzheimer’s disease or dementia, and agitation in Alzheimer’s disease or dementia.
[0203] Embodiment 28. The method according to embodiment 26, wherein the disease or disorder is schizophrenia.
[0204] Embodiment 29. The method according to embodiment 26, wherein the disease or disorder is a bipolar disorder or depressive episodes associated therewith.
[0205] Embodiment 30. The method according to any one of embodiments 24-29, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered subcutaneously, intravenously, orally, sublingually, buccally, transdermally, arterially, intradermally, intramuscularly, intraperitoneally, ocularly, intranasally, intraspinally or intracerebrally.
[0206] Embodiment 31. The method according to embodiment 24, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered orally.
[0207] Embodiment 32. The method according to embodiment 24, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered intramuscularly.
[0208] Embodiment 33. The method according to any one of embodiments 24-32, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered once daily.
[0209] Embodiment 34. The method according to any one of embodiments 24-32, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination them, is administered twice daily.
[0210] Embodiment 35. The method according to any one of embodiments 24-34, wherein the therapeutically effective amount of the crystalline form of Compound A is between 5 mg to 75 mg.
[0211] Embodiment 36. The method according to embodiment 35, wherein the therapeutically effective amount of the crystalline form of Compound A is between 10 mg to 30 mg.
[0212] Embodiment 37. A method of producing crystalline Form 1 of Compound A,
, comprising the steps of:
(i) dissolving Compound A in 2-propanol at 65-75 °C;
(ii) cooling the mixture to ambient temperature;
(iii) filtering the slurry; and
(iv) drying the collected solids.
[0213] Embodiment 38. A method of producing crystalline Form 2 of Compound A,
, comprising the steps of:
(i) dissolving crystalline Form 1 of Compound A in a solvent selected from acetonitrile, isopropyl alcohol, ethyl acetate, or acetone; and
(ii) slow evaporation.
[0214] Embodiment 39. A method of producing crystalline Form 2 of Compound A,
, comprising the steps of:
(i) slurrying crystalline Form 1 of Compound A in a solvent mixture selected from:
(a) acetonitrile-water at ambient temperature, wherein the mixture comprises about 99.7 % of acetonitrile by volume;
(b) acetonitrile-water at 5°C, wherein the mixture comprises about 99.3% of acetonitrile by volume; or
(c) acetonitrile-water at 50°C, wherein the mixture comprises about 97.2% of acetonitrile by volume; and
(ii) filtering the slurry.
[0215] Embodiment 40. A method of producing crystalline Form 2 of Compound A,
, comprising the steps of:
(i) dissolving Compound A in 2-propanol at 65-75 °C;
(ii) cooling the mixture to 35-45 °C;
(iii) slowly adding water;
(iv) filtering the slurry; and
(v) drying the collected solids.
[0216] Embodiment 41. A method of producing crystalline Form 3 of Compound A,
, comprising the steps of:
(i) slurrying crystalline Form 1 of Compound A in a solvent selected from 2-methyl tetrahydrofuran, tetrahydrofuran, water, or toluene at between 5 °C and 50 °C; and
(ii) filtering the slurry.
[0217] Embodiment 42. A method of producing crystalline Form 3 of Compound A,
, comprising the steps of:
(i) slurrying crystalline Form 1 of Compound A in a solvent mixture selected from:
(a) acetonitrile-water at ambient temperature, wherein the mixture comprises about 38.4%, 79.2%, 92.1%, 95.6%, 97.2%, 98.1%, 98.8% of acetonitrile by volume;
(b) acetonitrile-water at 5°C, wherein the mixture comprises about 79.2% or 97.2% of acetonitrile by volume; or
(c) acetonitrile-water at 50°C, wherein the mixture comprises about 79.2% of acetonitrile by volume; and
(ii) filtering the slurry.
[0218] Embodiment 43. A method of producing crystalline Form 3 of Compound A,
, comprising the steps of:
(i) dissolving Compound A in 2-propanol at 65-75 °C;
(ii) cooling the mixture to 50-55 °C;
(iii) slowly adding water;
(iv) adding a seed crystal;
(v) cooling the mixture to ambient temperature;
(vii) filtering the slurry; and
(viii) drying the collected solids.
[0219] Embodiment 44. A method of producing crystalline Form 4 of Compound A,
, comprising the steps of:
(i) slurrying crystalline Form 1 of Compound A in a mixture of acetonitrile-water at 50°C, wherein the mixture comprises about 79.2% of acetonitrile by volume; and
(ii) filtering the slurry.
[0220] Embodiment 45. A method of producing crystalline Form 4 of Compound A,
, comprising the steps of:
(i) dissolving Compound A in 2-propanol at 70-80 °C;
(ii) cooling the mixture to 45-50 °C;
(iii) filtering the slurry; and
(iv) drying the collected solids.
[0221] Embodiment 46. A pharmaceutical composition comprising a core and a coating, wherein the core comprises Compound A or a salt thereof and one or more pharmaceutically acceptable carriers or excipients, and wherein the coating comprises an opacifier.
[0222] Embodiment 47. A pharmaceutical composition comprising about 5 mg, about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, or about 50 mg of Compound A or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 85 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 5 weight percent of at least one lubricant, and (d) about 1 weight percent to about 8 weight percent of a coating comprising an opacifier, based on the total weight of the pharmaceutical composition.
[0223] Embodiment 48. A pharmaceutical composition comprising about 10 mg of Compound A or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 87 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
[0224] Embodiment 49. A pharmaceutical composition comprising about 15 mg of Compound A or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 87 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
[0225] Embodiment 50. A pharmaceutical composition comprising about 30 mg of Compound A or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 85 weight percent to about 93 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
[0226] Embodiment 51. The pharmaceutical composition according to any one of embodiments 46 to 50, wherein the opacifier is selected from the group consisting of: titanium dioxide, silicon dioxide, zinc oxide, iron oxide, talc, calcium carbonate, calcium phosphate, calcium sulfate, behenic acid, aluminum silicate, aluminum hydroxide, magnesium carbonate, tartrazine, erythrosine, or a mixture thereof.
[0227] Embodiment 52. The pharmaceutical composition according to any one of embodiments 46 to 50, wherein the opacifier is titanium dioxide.
[0228] Embodiment 53. The pharmaceutical composition according to any one of embodiments 46 to 52, wherein coating comprises the opacifier in an amount of from about 4 weight percent to about 25 weight percent of the coating by weight, based upon total weight of the coating composition.
[0229] Embodiment 54. The pharmaceutical composition according to any one of embodiments 46 to 53, wherein coating comprises the opacifier in an amount of from about 4 weight percent to about 20 weight percent of the coating by weight, based upon total weight of the coating composition.
[0230] Embodiment 55. The pharmaceutical composition according to any one of embodiments 46 to 54, wherein coating comprises the opacifier in an amount of from about 5 weight percent to about 15 weight percent, or about 10 weight percent to about 15 weight percent, or about 15 weight percent to about 20 weight percent of the coating by weight, based upon total weight of the coating composition.
[0231] Embodiment 56. The pharmaceutical composition according to any one of embodiments 46 to 55, wherein coating comprises the opacifier in an amount of about 6 weight percent, about 14 weight percent, or about 17 weight percent of the coating by weight, based upon total weight of the coating composition.
[0232] Embodiment 57. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising a crystalline form of Compound A.
[0233] Embodiment 58. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising one or more of a crystalline form of Compound A according to any one of embodiments 1-12.
[0234] Embodiment 59. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising crystalline Form 1 of Compound A.
[0235] Embodiment 60. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising crystalline Form 2 of Compound A.
[0236] Embodiment 61. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising crystalline Form 3 of Compound A.
[0237] Embodiment 62. The pharmaceutical composition according to any one of embodiments 46 to 56, comprising crystalline Form 4 of Compound A.
[0238] Embodiment 63. A method for treating a disease or disorder in a patient in need thereof, said method comprising administering to the patient a pharmaceutical composition according to any one of embodiments 46 to 62.
[0239] Embodiment 64. The method according to embodiment 63, wherein the disease or disorder is selected from Alzheimer’s Disease, psychosis in Alzheimer’s Disease, schizophrenia or psychosis, pain, addiction, a sleep disorder, a cognitive disorder (e.g. mild cognitive impairment), Parkinson’s Disease, Parkinson’s Disease-levodopa-induced dyskinesia, Huntington’s Disease, dyskinesia, dry mouth, pulmonary hypertension, chronic obstructive pulmonary disease (COPD), asthma, urinary incontinence, glaucoma, Trisomy 21 (Down
Syndrome), cerebral amyloid angiopathy, dementia, Hereditary Cerebral Hemorrhage with Amyloidosis of the Dutch-Type (HCHWA-D), Creutzfeld-Jakob disease, prion disorders, amyotrophic lateral sclerosis, progressive supranuclear palsy, head trauma, stroke, pancreatitis, inclusion body myositis, other peripheral amyloidoses, diabetes, autism, tardive dyskinesia, or atherosclerosis.
[0240] Embodiment 65. The method according to embodiment 63, wherein the disease or disorder is selected from Alzheimer’s disease, dementia, a bipolar disorder, schizophrenia, pain, an addiction, a sleep disorder, and conditions associated therewith.
[0241] Embodiment 66. The method according to embodiment 63, wherein the disease or disorder is selected from the group consisting of Alzheimer's Disease or dementia, psychosis in
Alzheimer’s disease or dementia, and agitation in Alzheimer’s disease or dementia.
[0242] Embodiment 67. The method according to embodiment 63, wherein the disease or disorder is schizophrenia.
[0243] Embodiment 68. The method according to embodiment 63, wherein the disease or disorder is a bipolar disorder or depressive episodes associated therewith.
Claims
2. The crystalline form of Compound A according to claim 1, characterized by an XRPD pattern comprising peaks selected from at 12.7, 12.9, 19.1, 19.7, and 24.3 ± 0.2 degrees 2θ.
3. The crystalline form of Compound A according to claim 1, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 1; b. a TGA thermogram substantially as set forth in Figure 2; c. a DSC thermogram substantially as set forth in Figure 3; d. a DVS plot substantially as set forth in Figure 4; e. a FT Raman spectrum substantially as set forth in Figure 5; and f. a PLM image substantially as set forth in Figure 6.
5. The crystalline form of Compound A according to claim 4, characterized by an XRPD pattern comprising peaks at 19.2, 11.5, 17.5, 19.9, and 23.1 ± 9.2 degrees 2θ.
6. The crystalline form of Compound A according to claim 4, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 7; b. a TGA thermogram substantially as set forth in Figure 8; and c. a DSC thermogram substantially as set forth in Figure 9.
8. The crystalline form of Compound A according to claim 7, characterized by an XRPD pattern comprising peaks at 10.5, 12.0, 17.7, 20.6, and 24.0 ± 0.2 degrees 2θ.
9. The crystalline form of Compound A according to claim 7, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 10; b. a TGA thermogram substantially as set forth in Figure 11; c. a DSC thermogram substantially as set forth in Figure 12; d. a DVS plot substantially as set forth in Figure 13; and e. a PLM image as set forth in Figure 14.
11. The crystalline form of Compound A according to claim 10, characterized by an XRPD pattern comprising peaks at 12.9, 16.4, 19.5, 20.3, and 24.0 ± 0.2 degrees 2θ.
12. The crystalline form of Compound A according to claim 10, characterized by one or more of: a. an XRPD pattern substantially as set forth in Figure 15; b. a TGA thermogram substantially as set forth in Figure 16; c. a DSC thermogram substantially as set forth in Figure 17; d. a DVS plot substantially as set forth in Figure 18; e. a FT Raman spectrum substantially as set forth in Figure 19; and f. a PLM image as set forth in Figure 20.
13. A pharmaceutical composition comprising one or more of a crystalline form of Compound A according to any one of claims 1-12, and at least one pharmaceutically acceptable carrier or excipient.
14. The pharmaceutical composition according to claim 13, wherein the pharmaceutical composition is formulated as a tablet, capsule, pill, lozenge, powder, granule, solution or suspension.
15. The pharmaceutical composition according to claim 13 or 14, wherein the pharmaceutical composition is in an extended-release form, a non-extended release form, or an immediate release form.
16. The pharmaceutical composition according to claim 15, wherein the pharmaceutical composition is in an extended release form.
17. The pharmaceutical composition according to claim 15, wherein the pharmaceutical composition is in a non-extended release form.
18. The pharmaceutical composition according to claim 15, wherein the pharmaceutical composition is in an immediate release form.
19. A pharmaceutical combination comprising one or more of a crystalline form of Compound A according to any one of claims 1-12, or one or more of a pharmaceutical composition according to any one of claims 13-18, and one or more additional therapeutic agents.
20. The pharmaceutical combination according to claim 19, wherein the one or more additional therapeutic agents are selected from risperidone, olanzapine, clozapine, quetiapine, aripiprazole, ziprasidone, paliperidone, asenapine, iloperidone, or bexpiprazole.
21. The pharmaceutical combination according to claim 19, wherein the one or more additional therapeutics agents are selected from ziprasidone, risperidone, olanzapine, quetiapine, aripiprazole, asenapine, blonanserin, or iloperidone.
22. The pharmaceutical combination of claim 19, wherein the one or more additional therapeutic agents are selected from selegiline, selegiline hydrochloride, dimethylselegilene, brofaromine, phenelzine, tranylcypromine, moclobemide, befloxatone, safinamide, isocarboxazid, nialamide, rasagiline, iproniazide, CHF-3381, iproclozide, toloxatone, desoxypeganine, harmine, harmaline, linezolid, pargyline, metralindole, pirlindole, bifemelane, bupropion, nefazodone, trazodone, desvenlafaxine, duloxetine, levomilnacipran, milnacipran, venlafaxine, citalopram, escitalopram, fluoxetine, fluvoxamine, paroxetine, sertraline, amitriptyline, amitriptylinoxide, clomipramine, desipramine, dibenzepin, dimetacrine, dosulepin, doxepin, imipramine, lofepramine, melitracen, nitroxazepine, nortriptyline, noxiptiline, opipramol, pipofezine, protriptyline, trimiprame, amoxapine, maprotiline, mianserin, mirtazapine, or setiptiline.
23. The pharmaceutical combination of claim 19, wherein the one or more additional therapeutic agents are selected from xanomeline, donepezil, galantamine, or rivastigmine.
24. A method for treating a disease or disorder in a patient in need thereof, said method comprising administering to the patient a therapeutically effective amount of one or more of
a crystalline form of Compound A according to any one of claims 1-12, a pharmaceutical composition according to any one of claims 13-18, or a pharmaceutical combination according to any one of claims 19-23.
25. The method according to claim 24, wherein the disease or disorder is selected from Alzheimer’s Disease, psychosis in Alzheimer’s Disease, schizophrenia or psychosis, pain, addiction, a sleep disorder, a cognitive disorder (e.g. mild cognitive impairment), Parkinson’s Disease, Parkinson’s Disease-levodopa-induced dyskinesia, Huntington’s Disease, dyskinesia, dry mouth, pulmonary hypertension, chronic obstructive pulmonary disease (COPD), asthma, urinary incontinence, glaucoma, Trisomy 21 (Down Syndrome), cerebral amyloid angiopathy, dementia, Hereditary Cerebral Hemorrhage with Amyloidosis of the Dutch-Type (HCHWA-D), Creutzfeld-Jakob disease, prion disorders, amyotrophic lateral sclerosis, progressive supranuclear palsy, head trauma, stroke, pancreatitis, inclusion body myositis, other peripheral amyloidoses, diabetes, autism, tardive dyskinesia, or atherosclerosis.
26. The method according to claim 24, wherein the disease or disorder is selected from Alzheimer’s disease, dementia, a bipolar disorder, schizophrenia, pain, an addiction, a sleep disorder, and conditions associated therewith.
27. The method according to claim 26, wherein the disease or disorder is selected from the group consisting of Alzheimer’s Disease or dementia, psychosis in Alzheimer’s disease or dementia, and agitation in Alzheimer’s disease or dementia.
28. The method according to claim 26, wherein the disease or disorder is schizophrenia.
29. The method according to claim 26, wherein the disease or disorder is a bipolar disorder or depressive episodes associated therewith.
30. The method according to any one of claims 24-29, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered subcutaneously, intravenously, orally, sublingually, buccally, transdermally, arterially, intradermally, intramuscularly, intraperitoneally, ocularly, intranasally, intraspinally or intracerebrally.
31. The method according to claim 24, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered orally.
32. The method according to claim 24, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered intramuscularly.
33. The method according to any one of claims 24-32, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination of them, is administered once daily.
34. The method according to any one of claims 24-32, wherein the crystalline form of Compound A, the pharmaceutical composition of them, or the pharmaceutical combination them, is administered twice daily.
35. The method according to any one of claims 24-34, wherein the therapeutically effective amount of the crystalline form of Compound A is between 5 mg to 75 mg.
36. The method according to claim 35, wherein the therapeutically effective amount of the crystalline form of Compound A is between 10 mg to 30 mg.
(i) dissolving Compound A in 2-propanol at 65-75 °C;
(ii) cooling the mixture to ambient temperature;
(iii) filtering the slurry; and
(iv) drying the collected solids.
(i) dissolving crystalline Form 1 of Compound A in a solvent selected from acetonitrile, isopropyl alcohol, ethyl acetate, or acetone; and
(ii) slow evaporation.
, comprising the steps of:
(i) slurrying crystalline Form 1 of Compound A in a solvent mixture selected from:
(a) acetonitrile-water at ambient temperature, wherein the mixture comprises about 99.7 % of acetonitrile by volume;
(b) acetonitrile-water at 5°C, wherein the mixture comprises about 99.3% of acetonitrile by volume; or
(c) acetonitrile-water at 50°C, wherein the mixture comprises about 97.2% of acetonitrile by volume; and
(ii) filtering the slurry.
(i) dissolving Compound A in 2-propanol at 65-75 °C;
(ii) cooling the mixture to 35-45 °C;
(iii) slowly adding water;
(iv) filtering the slurry; and
(v) drying the collected solids.
(i) slurrying crystalline Form 1 of Compound A in a solvent selected from 2-methyl tetrahydrofuran, tetrahydrofuran, water, or toluene at between 5 °C and 50 °C; and
(ii) filtering the slurry.
(i) slurrying crystalline Form 1 of Compound A in a solvent mixture selected from:
(a) acetonitrile-water at ambient temperature, wherein the mixture comprises about 38.4%, 79.2%, 92.1%, 95.6%, 97.2%, 98.1%, 98.8% of acetonitrile by volume;
(b) acetonitrile-water at 5°C, wherein the mixture comprises about 79.2% or 97.2% of acetonitrile by volume; or
(c) acetonitrile-water at 50°C, wherein the mixture comprises about 79.2% of acetonitrile by volume; and
(ii) filtering the slurry.
(i) dissolving Compound A in 2-propanol at 65-75 °C;
(ii) cooling the mixture to 50-55 °C;
(iii) slowly adding water;
(iv) adding a seed crystal;
(v) cooling the mixture to ambient temperature;
(vii) filtering the slurry; and
(viii) drying the collected solids.
(i) slurrying crystalline Form 1 of Compound A in a mixture of acetonitrile-water at 50°C, wherein the mixture comprises about 79.2% of acetonitrile by volume; and
(ii) filtering the slurry.
, comprising the steps of:
(i) dissolving Compound A in 2-propanol at 70-80 °C;
(ii) cooling the mixture to 45-50 °C;
(iii) filtering the slurry; and
(iv) drying the collected solids.
46. A pharmaceutical composition comprising a core and a coating, wherein the core comprises Compound A, or a pharmaceutically acceptable salt or hydrate thereof, and one or more pharmaceutically acceptable carriers or excipients, and wherein the coating comprises an opacifier.
47. A pharmaceutical composition comprising about 5 mg, about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, or about 50 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 85 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 5 weight percent of at least one lubricant, and (d) about 1 weight percent to about 8 weight percent of a coating comprising an opacifier, based on the total weight of the pharmaceutical composition.
48. A pharmaceutical composition comprising about 10 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 87 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
49. A pharmaceutical composition comprising about 15 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 87 weight percent to about 95 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
50. A pharmaceutical composition comprising about 30 mg of Compound A, or a pharmaceutically acceptable salt or hydrate thereof; and (a) about 85 weight percent to about 93 weight percent of at least one filler, (b) about 2 weight percent to about 5 weight percent of at least one disintegrant, (c) about 0.25 weight percent to about 3 weight percent of at least one lubricant, and (d) about 2 weight percent to about 5 weight percent of a coating, based on the total weight of the pharmaceutical composition.
51. The pharmaceutical composition according to any one of claims 46 to 50, wherein the opacifier is selected from the group consisting of: titanium dioxide, silicon dioxide, zinc oxide, iron oxide, talc, calcium carbonate, calcium phosphate, calcium sulfate, behenic acid, aluminum silicate, aluminum hydroxide, magnesium carbonate, tartrazine, erythrosine, or a mixture thereof.
52. The pharmaceutical composition according to any one of claims 46 to 50, wherein the opacifier is titanium dioxide.
53. The pharmaceutical composition according to any one of claims 46 to 52, wherein coating comprises the opacifier in an amount of from about 4 weight percent to about 25 weight percent of the coating by weight, based upon total weight of the coating composition.
54. The pharmaceutical composition according to any one of claims 46 to 53, wherein coating comprises the opacifier in an amount of from about 4 weight percent to about 20 weight percent of the coating by weight, based upon total weight of the coating composition.
55. The pharmaceutical composition according to any one of claims 46 to 54, wherein coating comprises the opacifier in an amount of from about 5 weight percent to about 15 weight percent, or about 10 weight percent to about 15 weight percent, or about 15 weight percent to about 20 weight percent of the coating by weight, based upon total weight of the coating composition.
56. The pharmaceutical composition according to any one of claims 46 to 55, wherein coating comprises the opacifier in an amount of about 6 weight percent, about 14 weight percent, or about 17 weight percent of the coating by weight, based upon total weight of the coating composition.
57. The pharmaceutical composition according to any one of claims 46 to 56, comprising a crystalline form of Compound A.
58. The pharmaceutical composition according to any one of claims 46 to 56, comprising one or more of a crystalline form of Compound A according to any one of claims 1-12.
59. The pharmaceutical composition according to any one of claims 46 to 56, comprising crystalline Form 1 of Compound A.
60. The pharmaceutical composition according to any one of claims 46 to 56, comprising crystalline Form 2 of Compound A.
61. The pharmaceutical composition according to any one of claims 46 to 56, comprising crystalline Form 3 of Compound A.
62. The pharmaceutical composition according to any one of claims 46 to 56, comprising crystalline Form 4 of Compound A.
63. A method for treating a disease or disorder in a patient in need thereof, said method comprising administering to the patient a pharmaceutical composition according to any one of claims 46 to 62.
64. The method according to claim 63, wherein the disease or disorder is selected from Alzheimer’s Disease, psychosis in Alzheimer’s Disease, schizophrenia or psychosis, pain, addiction, a sleep disorder, a cognitive disorder (e.g. mild cognitive impairment), Parkinson’s Disease, Parkinson’s Disease-levodopa-induced dyskinesia, Huntington’s Disease, dyskinesia, dry mouth, pulmonary hypertension, chronic obstructive pulmonary disease (COPD), asthma, urinary incontinence, glaucoma, Trisomy 21 (Down Syndrome), cerebral amyloid angiopathy, dementia, Hereditary Cerebral Hemorrhage with Amyloidosis of the Dutch-Type (HCHWA-D), Creutzfeld-Jakob disease, prion disorders, amyotrophic lateral sclerosis, progressive supranuclear palsy, head trauma, stroke, pancreatitis, inclusion body myositis, other peripheral amyloidoses, diabetes, autism, tardive dyskinesia, or atherosclerosis.
65. The method according to claim 63, wherein the disease or disorder is selected from Alzheimer’s disease, dementia, a bipolar disorder, schizophrenia, pain, an addiction, a sleep disorder, and conditions associated therewith.
66. The method according to claim 63, wherein the disease or disorder is selected from the group consisting of Alzheimer's Disease or dementia, psychosis in Alzheimer’s disease or dementia, and agitation in Alzheimer’s disease or dementia.
67. The method according to claim 63, wherein the disease or disorder is schizophrenia.
68. The method according to claim 63, wherein the disease or disorder is a bipolar disorder or depressive episodes associated therewith.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363616123P | 2023-12-29 | 2023-12-29 | |
| US63/616,123 | 2023-12-29 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025145091A1 true WO2025145091A1 (en) | 2025-07-03 |
Family
ID=94383728
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/062161 Pending WO2025145091A1 (en) | 2023-12-29 | 2024-12-27 | Crystalline forms of a muscarinic m4 receptor modulator and methods of treating diseases |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025145091A1 (en) |
Citations (44)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5750349A (en) | 1993-01-25 | 1998-05-12 | Takeda Chemical Industries Ltd. | Antibodies to β-amyloids or their derivatives and use thereof |
| EP0994728A1 (en) | 1997-04-09 | 2000-04-26 | Mindset, Ltd. | Recombinant antibodies specific for beta-amyloid ends, dna encoding and methods of use thereof |
| US6174884B1 (en) | 1997-03-11 | 2001-01-16 | Bayer Aktiengesellschaft | 1,5-dihydro-pyrazolo[34-D]-pyrimidinone derivatives |
| US6235742B1 (en) | 1997-10-24 | 2001-05-22 | Pfizer Inc. | 5-substituted pyrazolo[4,3-D]pyrimidin-7-ones |
| WO2002020521A1 (en) | 2000-09-11 | 2002-03-14 | Merck Patent Gmbh | Use of indole derivatives for treating illnesses of the central nervous system |
| EP1257584A2 (en) | 2000-02-24 | 2002-11-20 | Washington University St. Louis | Humanized antibodies that sequester amyloid beta peptide |
| US20030073655A1 (en) | 1997-04-09 | 2003-04-17 | Chain Daniel G. | Specific antibodies to amyloid beta peptide, pharmaceutical compositions and methods of use thereof |
| US20030195205A1 (en) | 2001-11-02 | 2003-10-16 | Pfizer Inc. | PDE9 inhibitors for treating cardiovascular disorders |
| WO2004032868A2 (en) | 2002-10-09 | 2004-04-22 | Rinat Neuroscience Corp. | Methods of treating alzheimer's disease using antibodies directed against amyloid beta peptide and compositions thereof |
| US20040192898A1 (en) | 2001-08-17 | 2004-09-30 | Jia Audrey Yunhua | Anti-abeta antibodies |
| US20040220186A1 (en) | 2003-04-30 | 2004-11-04 | Pfizer Inc. | PDE9 inhibitors for treating type 2 diabetes,metabolic syndrome, and cardiovascular disease |
| US20050019328A1 (en) | 1997-12-02 | 2005-01-27 | Neuralab Limited | Prevention and treatment of amyloidogenic disease |
| US20050043354A1 (en) | 2003-06-19 | 2005-02-24 | Pfizer Inc | NK1 antagonist |
| US20050048049A1 (en) | 1997-12-02 | 2005-03-03 | Neuralab Limited | Prevention and treatment of amyloidogenic disease |
| WO2005025616A1 (en) | 2003-09-09 | 2005-03-24 | Takeda Pharmaceutical Company Limited | Use of antibody |
| WO2005049616A1 (en) | 2003-11-24 | 2005-06-02 | Pfizer Limited | 5,7-DIAMINOPYRAZOLO [4,3-d] PYRIMIDINES WITH PDE-5 INHIBITING ACTIVITY |
| US20050256135A1 (en) | 2004-05-12 | 2005-11-17 | Graham Lunn | Tetrahydronaphthyridine derivatives |
| US20050267095A1 (en) | 2004-05-07 | 2005-12-01 | Patrick Bernardelli | 3- or 4-monosubstituted phenol and thiophenol derivatives useful as H3 ligands |
| WO2006036291A2 (en) | 2004-07-30 | 2006-04-06 | Rinat Neuroscience Corp. | Antibodies directed against amyloid-beta peptide and methods using same |
| US20060106035A1 (en) | 2002-08-23 | 2006-05-18 | Martin Hendrix | Phenyl-substituted pyrazolopyrimidines |
| US20060111372A1 (en) | 2002-08-23 | 2006-05-25 | Bayer Healthcare Ag | Alkyl-substituted pyrazolopyrimidines |
| WO2006069081A2 (en) | 2004-12-22 | 2006-06-29 | Washington University In St. Louis | USE OF ANTI-Aβ ANTIBODY TO TREAT TRAUMATIC BRAIN INJURY |
| US7115600B2 (en) | 2004-02-02 | 2006-10-03 | Pfizer Inc. | Histamine-3 receptor modulators |
| WO2006118959A2 (en) | 2005-04-29 | 2006-11-09 | Rinat Neuroscience Corp. | Antibodies directed against amyloid-beta peptide and methods using same |
| WO2006120552A2 (en) | 2005-05-12 | 2006-11-16 | Pfizer Inc. | ANHYDROUS CRYSTALLINE FORMS OF N-[1-(2-ETHOXYETHYL)-5-(N-ETHYL-N-METHYLAMINO)-7-(4-METHYLPYRIDIN-2-yl-AMINO)-1H-PYRAZOLO[4,3-d]PYRIMIDINE-3-CARBONYL]METHANESULFONAMIDE |
| WO2006126083A1 (en) | 2005-05-24 | 2006-11-30 | Pharmacia & Upjohn Company Llc | Pyridine [3 , 4-b] pyrazinone compounds as pde-5 inhibitors |
| WO2006126081A2 (en) | 2005-05-24 | 2006-11-30 | Pharmacia & Upjohn Company Llc | Pyridino [2 , 3-b] pyrazinones as pde-5 inhibitors |
| WO2006126082A2 (en) | 2005-05-24 | 2006-11-30 | Pharmacia & Upjohn Company Llc | Pyridine [3,4-b] pyrazinones as pde-5 inhibitors |
| WO2006136924A1 (en) | 2005-06-22 | 2006-12-28 | Pfizer Products Inc. | Histamine-3 receptor antagonists |
| US20070079175A1 (en) | 2005-09-30 | 2007-04-05 | Hon Hai Precision Industry Co., Ltd. | System, portable electronic apparatus and method for timely receiving and displaying electronic files |
| WO2007063385A2 (en) | 2005-12-01 | 2007-06-07 | Pfizer Products Inc. | Spirocyclic amine histamine-3 receptor antagonists |
| WO2007069053A1 (en) | 2005-12-14 | 2007-06-21 | Pfizer Products Inc. | Benzimidazole antagonists of the h-3 receptor |
| WO2007088450A2 (en) | 2006-02-01 | 2007-08-09 | Pfizer Products Inc. | Chromane antagonist of the h-3 receptor |
| WO2007088462A1 (en) | 2006-02-01 | 2007-08-09 | Pfizer Products Inc. | Spirochromane antagonists of the h-3 receptor |
| WO2007099423A1 (en) | 2006-03-02 | 2007-09-07 | Pfizer Products Inc. | 1-pyrrolidine indane derivatives as histamine-3 receptor antagonists |
| WO2007105053A2 (en) | 2006-03-13 | 2007-09-20 | Pfizer Products Inc. | Tetralines antagonists of the h-3 receptor |
| US7285293B2 (en) | 2000-11-03 | 2007-10-23 | Gerardo Castillo | Methods of isolating amyloid-inhibiting compounds and use of compounds isolated from Uncaria tomentosa and related plants |
| WO2007122466A1 (en) | 2006-04-21 | 2007-11-01 | Pfizer Products Inc. | Pyridine[3,4-b]pyrazinones |
| WO2007138431A2 (en) | 2006-05-30 | 2007-12-06 | Pfizer Products Inc. | Azabicyclic ether histamine-3 antagonists |
| US20080096955A1 (en) | 2005-10-27 | 2008-04-24 | Pfizer Inc | Histamine-3 receptor antagonists |
| US20080176925A1 (en) | 2007-01-22 | 2008-07-24 | Pfizer Inc | Toluene sulfonic acid salt of a therapeutic compound and pharmaceutical compositions thereof |
| WO2018002760A1 (en) | 2016-07-01 | 2018-01-04 | Pfizer Inc. | 5,7-dihydro-pyrrolo-pyridine derivatives for treating neurological and neurodegenerative diseases |
| US11806208B2 (en) | 2014-03-21 | 2023-11-07 | Align Technology, Inc. | Orthodontic appliances with shell segments and elastic segments |
| WO2024061253A1 (en) * | 2022-09-23 | 2024-03-28 | 苏州科睿思制药有限公司 | Crystal form of emraclidine, preparation method therefor, and use thereof |
-
2024
- 2024-12-27 WO PCT/US2024/062161 patent/WO2025145091A1/en active Pending
Patent Citations (44)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5750349A (en) | 1993-01-25 | 1998-05-12 | Takeda Chemical Industries Ltd. | Antibodies to β-amyloids or their derivatives and use thereof |
| US6174884B1 (en) | 1997-03-11 | 2001-01-16 | Bayer Aktiengesellschaft | 1,5-dihydro-pyrazolo[34-D]-pyrimidinone derivatives |
| US20030073655A1 (en) | 1997-04-09 | 2003-04-17 | Chain Daniel G. | Specific antibodies to amyloid beta peptide, pharmaceutical compositions and methods of use thereof |
| EP0994728A1 (en) | 1997-04-09 | 2000-04-26 | Mindset, Ltd. | Recombinant antibodies specific for beta-amyloid ends, dna encoding and methods of use thereof |
| US6235742B1 (en) | 1997-10-24 | 2001-05-22 | Pfizer Inc. | 5-substituted pyrazolo[4,3-D]pyrimidin-7-ones |
| US20050019328A1 (en) | 1997-12-02 | 2005-01-27 | Neuralab Limited | Prevention and treatment of amyloidogenic disease |
| US20050048049A1 (en) | 1997-12-02 | 2005-03-03 | Neuralab Limited | Prevention and treatment of amyloidogenic disease |
| EP1257584A2 (en) | 2000-02-24 | 2002-11-20 | Washington University St. Louis | Humanized antibodies that sequester amyloid beta peptide |
| WO2002020521A1 (en) | 2000-09-11 | 2002-03-14 | Merck Patent Gmbh | Use of indole derivatives for treating illnesses of the central nervous system |
| US7285293B2 (en) | 2000-11-03 | 2007-10-23 | Gerardo Castillo | Methods of isolating amyloid-inhibiting compounds and use of compounds isolated from Uncaria tomentosa and related plants |
| US20040192898A1 (en) | 2001-08-17 | 2004-09-30 | Jia Audrey Yunhua | Anti-abeta antibodies |
| US20030195205A1 (en) | 2001-11-02 | 2003-10-16 | Pfizer Inc. | PDE9 inhibitors for treating cardiovascular disorders |
| US20060106035A1 (en) | 2002-08-23 | 2006-05-18 | Martin Hendrix | Phenyl-substituted pyrazolopyrimidines |
| US20060111372A1 (en) | 2002-08-23 | 2006-05-25 | Bayer Healthcare Ag | Alkyl-substituted pyrazolopyrimidines |
| WO2004032868A2 (en) | 2002-10-09 | 2004-04-22 | Rinat Neuroscience Corp. | Methods of treating alzheimer's disease using antibodies directed against amyloid beta peptide and compositions thereof |
| US20040220186A1 (en) | 2003-04-30 | 2004-11-04 | Pfizer Inc. | PDE9 inhibitors for treating type 2 diabetes,metabolic syndrome, and cardiovascular disease |
| US20050043354A1 (en) | 2003-06-19 | 2005-02-24 | Pfizer Inc | NK1 antagonist |
| WO2005025616A1 (en) | 2003-09-09 | 2005-03-24 | Takeda Pharmaceutical Company Limited | Use of antibody |
| WO2005049616A1 (en) | 2003-11-24 | 2005-06-02 | Pfizer Limited | 5,7-DIAMINOPYRAZOLO [4,3-d] PYRIMIDINES WITH PDE-5 INHIBITING ACTIVITY |
| US7115600B2 (en) | 2004-02-02 | 2006-10-03 | Pfizer Inc. | Histamine-3 receptor modulators |
| US20050267095A1 (en) | 2004-05-07 | 2005-12-01 | Patrick Bernardelli | 3- or 4-monosubstituted phenol and thiophenol derivatives useful as H3 ligands |
| US20050256135A1 (en) | 2004-05-12 | 2005-11-17 | Graham Lunn | Tetrahydronaphthyridine derivatives |
| WO2006036291A2 (en) | 2004-07-30 | 2006-04-06 | Rinat Neuroscience Corp. | Antibodies directed against amyloid-beta peptide and methods using same |
| WO2006069081A2 (en) | 2004-12-22 | 2006-06-29 | Washington University In St. Louis | USE OF ANTI-Aβ ANTIBODY TO TREAT TRAUMATIC BRAIN INJURY |
| WO2006118959A2 (en) | 2005-04-29 | 2006-11-09 | Rinat Neuroscience Corp. | Antibodies directed against amyloid-beta peptide and methods using same |
| WO2006120552A2 (en) | 2005-05-12 | 2006-11-16 | Pfizer Inc. | ANHYDROUS CRYSTALLINE FORMS OF N-[1-(2-ETHOXYETHYL)-5-(N-ETHYL-N-METHYLAMINO)-7-(4-METHYLPYRIDIN-2-yl-AMINO)-1H-PYRAZOLO[4,3-d]PYRIMIDINE-3-CARBONYL]METHANESULFONAMIDE |
| WO2006126081A2 (en) | 2005-05-24 | 2006-11-30 | Pharmacia & Upjohn Company Llc | Pyridino [2 , 3-b] pyrazinones as pde-5 inhibitors |
| WO2006126082A2 (en) | 2005-05-24 | 2006-11-30 | Pharmacia & Upjohn Company Llc | Pyridine [3,4-b] pyrazinones as pde-5 inhibitors |
| WO2006126083A1 (en) | 2005-05-24 | 2006-11-30 | Pharmacia & Upjohn Company Llc | Pyridine [3 , 4-b] pyrazinone compounds as pde-5 inhibitors |
| WO2006136924A1 (en) | 2005-06-22 | 2006-12-28 | Pfizer Products Inc. | Histamine-3 receptor antagonists |
| US20070079175A1 (en) | 2005-09-30 | 2007-04-05 | Hon Hai Precision Industry Co., Ltd. | System, portable electronic apparatus and method for timely receiving and displaying electronic files |
| US20080096955A1 (en) | 2005-10-27 | 2008-04-24 | Pfizer Inc | Histamine-3 receptor antagonists |
| WO2007063385A2 (en) | 2005-12-01 | 2007-06-07 | Pfizer Products Inc. | Spirocyclic amine histamine-3 receptor antagonists |
| WO2007069053A1 (en) | 2005-12-14 | 2007-06-21 | Pfizer Products Inc. | Benzimidazole antagonists of the h-3 receptor |
| WO2007088450A2 (en) | 2006-02-01 | 2007-08-09 | Pfizer Products Inc. | Chromane antagonist of the h-3 receptor |
| WO2007088462A1 (en) | 2006-02-01 | 2007-08-09 | Pfizer Products Inc. | Spirochromane antagonists of the h-3 receptor |
| WO2007099423A1 (en) | 2006-03-02 | 2007-09-07 | Pfizer Products Inc. | 1-pyrrolidine indane derivatives as histamine-3 receptor antagonists |
| WO2007105053A2 (en) | 2006-03-13 | 2007-09-20 | Pfizer Products Inc. | Tetralines antagonists of the h-3 receptor |
| WO2007122466A1 (en) | 2006-04-21 | 2007-11-01 | Pfizer Products Inc. | Pyridine[3,4-b]pyrazinones |
| WO2007138431A2 (en) | 2006-05-30 | 2007-12-06 | Pfizer Products Inc. | Azabicyclic ether histamine-3 antagonists |
| US20080176925A1 (en) | 2007-01-22 | 2008-07-24 | Pfizer Inc | Toluene sulfonic acid salt of a therapeutic compound and pharmaceutical compositions thereof |
| US11806208B2 (en) | 2014-03-21 | 2023-11-07 | Align Technology, Inc. | Orthodontic appliances with shell segments and elastic segments |
| WO2018002760A1 (en) | 2016-07-01 | 2018-01-04 | Pfizer Inc. | 5,7-dihydro-pyrrolo-pyridine derivatives for treating neurological and neurodegenerative diseases |
| WO2024061253A1 (en) * | 2022-09-23 | 2024-03-28 | 苏州科睿思制药有限公司 | Crystal form of emraclidine, preparation method therefor, and use thereof |
Non-Patent Citations (4)
| Title |
|---|
| "Aulton's Pharmaceutics", 2018, ELSEVIER, pages: 580 - 596 |
| "Remington's Pharmaceutical Sciences", MEADE PUBLISHING CO. |
| BUBSER, MICHAEL ET AL.: "Selective Activation of M4 Muscarinic Acetylcholine Receptors reverses MK-801-Induced Behavioral Impairments and Enhances Associative Learning in Rodents", AMERICAN CHEMICAL SOCIETY, CHEMICAL NEUROSCIENCE, 2014 |
| BYNUM, NELLIE E. ET AL.: "Antipsychotic Drug-Like Effects of the Selective M4 Muscarinic Acetylcholine Receptor Positive Allosteric Modulator VUO 152 100", NEUROPSYCHOPHARMACOLOGY, 2014, pages 1 - 16 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| TWI404721B (en) | Amino-heterocyclic compounds | |
| TWI500616B (en) | Novel bicyclic pyridinones | |
| TWI568733B (en) | Novel chromene and 1,1a,2,7b-tetrahydrocyclopropa[c]chromene pyridopyrazinediones | |
| TWI629276B (en) | Imidazolium compound | |
| JP7497294B2 (en) | Piperazine azaspiro derivatives | |
| TW201326182A (en) | Hexahydropyrano[3,4-d][1,3]thiazin-2-amine compounds | |
| TWI616445B (en) | Novel cyclopropabenzofuranyl pyridopyrazinediones | |
| ES2742078T3 (en) | Novel bicyclic pyridones as gamma-secretase modulators | |
| EP4634193A1 (en) | Substituted dihydropyrrolo[3, 4-d]pyrimidine compounds and their use in treating medical conditions | |
| EP4634182A1 (en) | Substituted tetrahydropyrrolo-pyridinone compounds and their use in treating medical conditions | |
| WO2025145091A1 (en) | Crystalline forms of a muscarinic m4 receptor modulator and methods of treating diseases | |
| KR20250121123A (en) | Substituted tetrahydropyrrolo-pyridinone compounds and their use in treating medical conditions | |
| US20160229847A1 (en) | Novel bicyclic pyridinones as gamma-secretase modulators | |
| HK1242315B (en) | Novel cyclopropabenzofuranyl pyridopyrazinediones | |
| OA18058A (en) | Chromene and 1,1 A,2,7B-tetrahydrocyclopropa[C]chromene pyridopyrazinediones as gamma-secretase modulators. | |
| HK1262295B (en) | 6,7-dihydro-5h-pyrazolo[5,1-b][1,3]oxazine-2-carboxamide compounds | |
| WO2012172449A1 (en) | Lactams as beta secretase inhibitors | |
| HK1231481B (en) | Chromene and 1,1a,2,7b-tetrahydrocyclopropa[c]chromene pyridopyrazinediones as gamma-secretase modulators |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24847124 Country of ref document: EP Kind code of ref document: A1 |