WO2025128807A1 - Radiolabeled compounds for the detection of steap1 - Google Patents
Radiolabeled compounds for the detection of steap1 Download PDFInfo
- Publication number
- WO2025128807A1 WO2025128807A1 PCT/US2024/059745 US2024059745W WO2025128807A1 WO 2025128807 A1 WO2025128807 A1 WO 2025128807A1 US 2024059745 W US2024059745 W US 2024059745W WO 2025128807 A1 WO2025128807 A1 WO 2025128807A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- steapl
- fab
- construct
- steap1
- subject
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K51/00—Preparations containing radioactive substances for use in therapy or testing in vivo
- A61K51/02—Preparations containing radioactive substances for use in therapy or testing in vivo characterised by the carrier, i.e. characterised by the agent or material covalently linked or complexing the radioactive nucleus
- A61K51/04—Organic compounds
- A61K51/08—Peptides, e.g. proteins, carriers being peptides, polyamino acids, proteins
- A61K51/10—Antibodies or immunoglobulins; Fragments thereof, the carrier being an antibody, an immunoglobulin or a fragment thereof, e.g. a camelised human single domain antibody or the Fc fragment of an antibody
- A61K51/1045—Antibodies or immunoglobulins; Fragments thereof, the carrier being an antibody, an immunoglobulin or a fragment thereof, e.g. a camelised human single domain antibody or the Fc fragment of an antibody against animal or human tumor cells or tumor cell determinants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K39/00—Medicinal preparations containing antigens or antibodies
- A61K2039/505—Medicinal preparations containing antigens or antibodies comprising antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/50—Immunoglobulins specific features characterized by immunoglobulin fragments
- C07K2317/55—Fab or Fab'
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/92—Affinity (KD), association rate (Ka), dissociation rate (Kd) or EC50 value
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K2317/00—Immunoglobulins specific features
- C07K2317/90—Immunoglobulins specific features characterized by (pharmaco)kinetic aspects or by stability of the immunoglobulin
- C07K2317/94—Stability, e.g. half-life, pH, temperature or enzyme-resistance
Definitions
- the present invention relates to anti-STEAPl constructs for the use of detecting STEAP1 in a human.
- the invention further relates to anti-STEAPl (Fab’)2 fragments that are radiolabeled. Said constructs are useful for imaging STEAP1 expression, for example, in a positron emission tomography scan.
- PCa Prostate cancer
- ADT Androgen deprivation therapies
- the present invention provides an anti-STEAPl construct, wherein the anti-STEAP1 construct is radiolabeled with 64 Cu.
- the anti-STEAPl construct is a (Fab’)2 fragment.
- the anti-STEAPl construct further comprises at least one NOTA.
- the construct comprises one NOTA.
- the construct comprises two NOTAs.
- at least one NOTA comprises 64 Cu.
- each NOTA comprises 64 Cu.
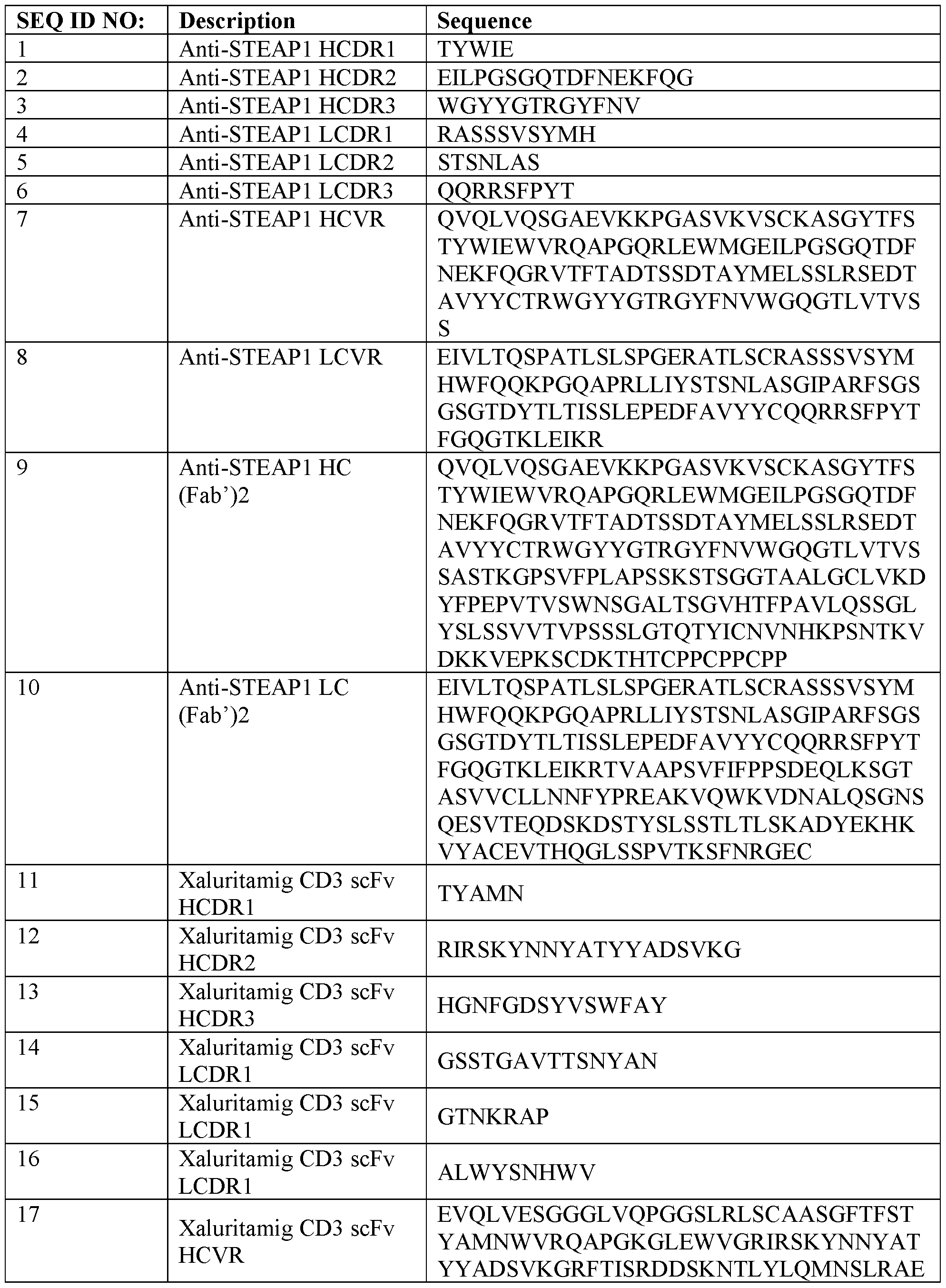
- the anti-STEAPl construct comprises HCDR1 comprising SEQ ID NO: 1, HCDR2 comprising SEQ ID NO: 2, HCDR3 comprising SEQ ID NO: 3, LCDR1 comprising SEQ ID NO: 4, LCDR2 comprising SEQ ID NO: 5, and LCDR3 comprising SEQ ID NO: 6.
- the anti-STEAPl construct comprises HCVR comprising SEQ ID NO: 7, and LCVR comprising SEQ ID NO: 8.
- the anti-STEAPl construct comprises a heavy chain (HC) fragment comprising SEQ ID NO: 9, and a light chain (LC) comprising SEQ ID NO: 10.
- the present invention provides an anti-STEAPl construct, wherein the anti- STEAPl construct is radiolabeled with 64 Cu, and wherein the anti-STEAPl construct is an antibody.
- the anti-STEAPl antibody comprises HCDR1 comprising SEQ ID NO: 1, HCDR2 comprising SEQ ID NO: 2, HCDR3 comprising SEQ ID NO: 3, LCDR1 comprising SEQ ID NO: 4, LCDR2 comprising SEQ ID NO: 5, and LCDR3 comprising SEQ ID NO: 6.
- the anti-STEAPl antibody comprises HCVR comprising SEQ ID NO: 7, and LCVR comprising SEQ ID NO: 8.
- the anti-STEAPl antibody comprises a HC comprising SEQ ID NO: 37, and a LC comprising SEQ ID NO: 10. In an embodiment, the anti-STEAPl antibody comprises a HC comprising SEQ ID NO: 27, and a LC comprising SEQ ID NO: 28.
- the present invention provides an anti-STEAPl construct, wherein the anti- STEAPl construct is radiolabeled with 89 Zr.
- the anti-STEAPl construct is a (Fab’)2 fragment.
- the anti-STEAPl construct further comprises at least one DFO or DFO*.
- the construct comprises one DFO or DFO*.
- the construct comprises two DFO or DFO*.
- the anti-STEAPl construct comprises HCDR1 comprising SEQ ID NO: 1, HCDR2 comprising SEQ ID NO: 2, HCDR3 comprising SEQ ID NO: 3, LCDR1 comprising SEQ ID NO: 4, LCDR2 comprising SEQ ID NO: 5, and LCDR3 comprising SEQ ID NO: 6.
- the anti-STEAPl construct comprises HCVR comprising SEQ ID NO: 7, and LCVR comprising SEQ ID NO: 8.
- the anti-STEAPl construct comprises a HC fragment comprising SEQ ID NO: 9, and a LC comprising SEQ ID NO: 10.
- the present invention provides an anti-STEAPl construct, wherein the anti- STEAPl construct is radiolabeled with 89 Zr, and wherein the anti-STEAPl construct is an antibody.
- the anti-STEAPl antibody comprises HCDR1 comprising SEQ ID NO: 1, HCDR2 comprising SEQ ID NO: 2, HCDR3 comprising SEQ ID NO: 3, LCDR1 comprising SEQ ID NO: 4, LCDR2 comprising SEQ ID NO: 5, and LCDR3 comprising SEQ ID NO: 6.
- the anti-STEAPl antibody comprises HCVR comprising SEQ ID NO: 7, and LCVR comprising SEQ ID NO: 8.
- the anti-STEAPl antibody comprises a HC comprising SEQ ID NO: 37, and a LC comprising SEQ ID NO: 10. In an embodiment, the anti-STEAPl antibody comprises a HC comprising SEQ ID NO: 27, and a LC comprising SEQ ID NO: 28.
- the present invention provides a polynucleotide encoding an amino acid sequence of a construct of the present invention.
- the present invention provides a vector comprising a polynucleotide of the present invention.
- the present invention provides a mammalian cell comprising a vector of the present invention. In an embodiment, the mammalian cell comprises two vectors of the present invention.
- the present invention provides a method of detecting STEAP1 in a subject, wherein the method comprises administering to the subject an anti-STEAPl construct of the present invention.
- the subject has cancer.
- the subject has a solid tumor.
- the subject has a solid tumor wherein the solid tumor expresses STEAP1.
- the subject has prostate cancer.
- the subject has metastatic castration resistant prostate cancer.
- the subject has metastatic castration sensitive prostate cancer.
- the present invention provides an anti-STEAPl construct of the present invention for use in detecting STEAP1 in a subject.
- the subject has cancer.
- the subject has a solid tumor.
- the subject has a solid tumor wherein the solid tumor expresses STEAP1.
- the subject has prostate cancer.
- the subject has metastatic castration resistant prostate cancer.
- the subject has metastatic castration sensitive prostate cancer.
- the present invention provides the use of the anti-STEAPl construct of the present invention for the manufacture of an imaging agent for the detection of cancer.
- the present invention provides the use of the anti-STEAPl construct of the present invention for the manufacture of an imaging agent for the treatment of cancer.
- the cancer is a solid tumor.
- the cancer is a solid tumor wherein the solid tumor expresses STEAP1.
- the cancer is prostate cancer.
- the cancer is metastatic castration resistant prostate cancer.
- the subject has metastatic castration sensitive prostate cancer.
- STEAP1 is detected by positron emission tomography scan.
- STEAP1 is detected in a subject’s prostate. In an embodiment, STEAP1 is detected in metastatic sites.
- the subject is further administered an agent for cancer treatment.
- the subject is further administered an agent for prostate cancer treatment.
- the agent for prostate cancer treatment binds STEAP1.
- the agent for prostate cancer treatment is an inhibitor of STEAP1.
- the agent for prostate cancer treatment results in killing of STEAP1 -expressing cancer cells.
- the agent for prostate cancer treatment is xaluritamig.
- the subject has been diagnosed as having STEAP1 expression.
- the present invention provides an anti-STEAPl construct of the present invention for use as a diagnostic biomarker.
- the present invention provides a diagnostic biomarker comprising an anti- STEAPl construct of the present invention.
- the present invention provides a kit comprising an anti-STEAPl construct of the present invention.
- the present invention provides a method of treating a subject diagnosed with cancer, wherein the method comprises administering to the subject an anti-STEAPl construct of the present invention, imaging STEAP1 expression in the subject, diagnosing the subject with cancer, and administering to the subject an agent for cancer treatment.
- the subject has elevated STEAP1 expression in the subject’s tumor.
- the agent for cancer treatment is xaluritamig.
- the present invention provides a method of treating a subject diagnosed with cancer, wherein the method comprises administering to the subject an anti-STEAPl construct of the present invention, imaging STEAP1 expression in the subject, and administering to the subject an agent for cancer treatment.
- the subject has elevated STEAP1 expression in the subject’s tumor.
- the agent for cancer treatment is xaluritamig.
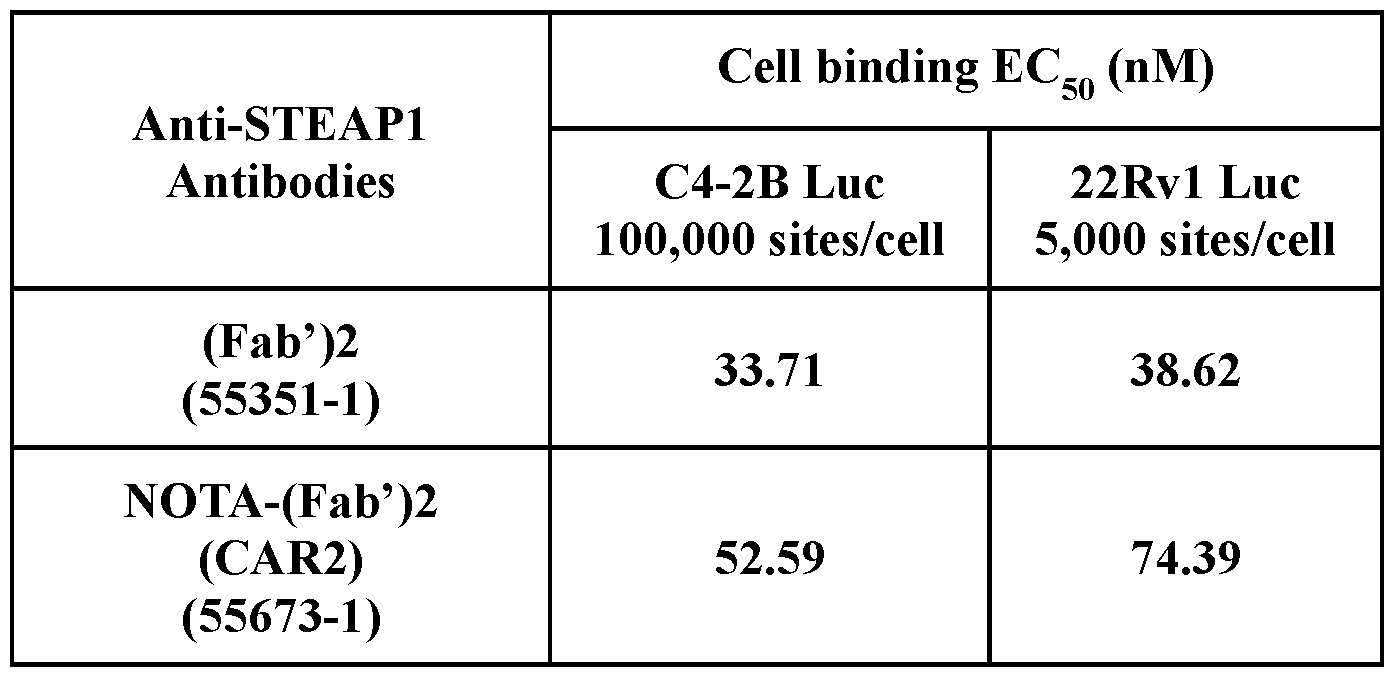
- Figures 1A and IB depict anti-STEAPl (Fab’)2 cell binding after NOTA (CAR1) conjugation.
- C4-2B Luc cells were incubated with increasing concentrations of NOTA-(Fab’)2 (CAR1) ( Figure 1 A), or cold label 63Cu-NOTA-(Fab’)2 (CAR1) ( Figure IB), alongside unconjugated (Fab’)2 up to 1 uM, for 1 hour at 4°C.
- Cell-bound antibodies were detected by flow cytometry after incubation with an AF647-conjugated anti-human IgG, (Fab’)2 specific, secondary antibody and median fluorescence intensity (MFI) of the AF647 channel at increasing concentrations of respective molecules were displayed.
- MFI median fluorescence intensity
- FIGS 2A and 2B depict anti-STEAPl (Fab’)2 cell binding after DFO (CAR1) or DFO* (CAR) conjugation.
- C4-2B Luc cells were incubated with increasing concentrations of DFO-(Fab’)2 (CAR1) ( Figure 2A), or DFO*-(Fab’)2 (CAR1) ( Figure 2B), alongside unconjugated (Fab’)2 up to 3 uM, for 1 hour at 4°C.
- Cell-bound antibodies were detected by flow cytometry after incubation with an AF647-conjugated anti-human IgG, (Fab’)2 specific, secondary antibody and median fluorescence intensity (MFI) of the AF647 channel at increasing concentrations of respective molecules were displayed.
- MFI median fluorescence intensity
- Figures 3A and 3B depict anti-STEAPl (Fab’)2 cell binding after NOTA (CAR2) conjugation.
- C4-2B Luc ( Figure 3A) or 22Rvl Luc ( Figure 3B) cells were incubated with increasing concentrations of NOTA-(Fab’)2 (CAR2) in comparison with unconjugated (Fab’)2 up to 1 uM, for 1 hour at 4°C.
- Cell-bound antibodies were detected by flow cytometry after incubation with an AF647-conjugated anti-human IgG, (Fab’)2 specific, secondary antibody and median fluorescence intensity (MFI) of the AF647 channel at increasing concentrations of respective molecules were displayed.
- MFI median fluorescence intensity
- FIGS 4A and 4B depict anti-STEAPl (Fab’)2 cell binding after DFO (CAR2) conjugation.
- C4-2B Luc ( Figure 4A) or 22Rvl Luc ( Figure 4B) cells were incubated with increasing concentrations of DFO-(Fab’)2 (CAR2) in comparison with unconjugated (Fab’)2 up to 1 uM, for 1 hour at 4°C.
- Cell-bound antibodies were detected by flow cytometry after incubation with an AF647-conjugated anti-human IgG, (Fab’)2 specific, secondary antibody and median fluorescence intensity (MFI) of the AF647 channel at increasing concentrations of respective molecules were displayed.
- MFI median fluorescence intensity
- Fig. 5A Unconjugated;
- Fig. 5B NOTA-CAR1;
- Fig. 5C DFO-CAR1;
- Fig. 5D DFO*-CAR1;
- Fig. 5E NOTA-CAR2;
- Fig. 5F DFO-CAR2;
- Fig. 5G 63Cu-NOTA CAR1;
- Fig. 5H buffer blank negative control.
- Figure 6 depicts the mean (and standard deviation) pharmacokinetic profile following 10 mg/kg administration of either NOTA-conjugated anti-STEAPl (Fab’)2 or DFO- conjugated anti-STEAPl (Fab’)2 in female BALB/c nude mice.
- Figure 7 depicts mean (and SD) pharmacokinetic profile following 1.125 mg/kg administration in cynomolgus monkeys.
- FIGS 8A and 8B depict 64 Cu-NOTA-Fab’2 CAR1 vs. CAR2 PET/CT imaging in STEAP1 positive xenografts. 22Rvl STEAPl-high cells were implanted in the right shoulder of the male balb/c nude mice.
- -200 uCi radiotracers 64 Cu-NOTA-Fab’2 CAR1 ( Figure 8 A) or CAR2 ( Figure 8B) were injected via tail vein and images were acquired at 22h (for CAR1) or 24h (for CAR2) post injection. Each tumor uptake of the radiotracer was quantified by calculating the percent injected dose per gram of tissue (%ID/g). Both 64 Cu- NOTA-Fab’2 CAR1 and CAR2 successfully localized to STEAPl-high xenograft tumors with comparable tumor uptake.
- Figures 9A and 9B depict 64 Cu-NOTA-Fab’2 CAR1 vs. CAR2 PET/CT imaging in STEAP1 -negative xenografts. 22Rvl STEAP1 -negative cells were implanted in the right shoulder of the male balb/c nude mice.
- -200 uCi radiotracers 64 Cu-NOTA-Fab’2 CAR1 ( Figure 9A) or CAR2 ( Figure 9B) were injected via tail vein and images were acquired at 22h (for CAR1) or 24h (for CAR2) post injection. Each tumor uptake of the radiotracer was quantified by calculating the percent injected dose per gram of tissue (%ID/g). Very low tumor uptake was observed for both molecules as expected.
- FIGS 10A and 10B depict 89 Zr-DFO-Fab’2 CAR1 vs. CAR2 PET/CT imaging in STEAPl-high xenografts. 22Rvl STEAPl-high cells were implanted in the right shoulder of the male balb/c nude mice.
- -200 uCi radiotracers 89 Zr-DFO-Fab’2 -CAR1 ( Figure 10A) or CAR2 ( Figure 10B) were injected via tail vein and images were acquired at 22h (for CAR1) or 24h (for CAR2) post injection. Each tumor uptake of the radiotracer was quantified by calculating the percent injected dose per gram of tissue (%ID/g). Both 89 Zr-DFO-Fab’2 CAR1 and CAR2 localized to STEAPl-high xenograft tumors with low tumor uptake.
- FIGS 11A and 11B depict 89 Zr-DFO-Fab’2 CAR1 vs. CAR2 PET/CT imaging in STEAP1 -negative xenografts. 22Rvl STEAP1 -negative cells were implanted in the right shoulder of the male balb/c nude mice.
- -200 uCi radiotracers 89 Zr-DFO-Fab’2 -CAR1 ( Figure 11 A) or CAR2 ( Figure 1 IB) were injected via tail vein and images were acquired at 22h (for CAR1) or 24h (for CAR2) post injection. Each tumor uptake of the radiotracer was quantified by calculating the percent injected dose per gram of tissue (%ID/g). No obvious tumor uptake was observed for both 89Zr-DFO-Fab’2-CARl and CAR2.
- FIGS 12A and 12B depict 89 Zr-DFO*-Fab’2 CAR1 PET Imaging in STEAP1 positive and STEAP1 -negative xenografts.
- 22Rvl STEAPl-high (A) and 22Rvl STEAP1-KO cells (B) were implanted in the right shoulder area of the male balb/c nude mice.
- -200 uCi Radiotracers were injected via tail vein and images were acquired at 24h post injection. Each tumor uptake of the radiotracer was indicated by percent injected dose per gram of tissue (%ID/g).
- 89 Zr-DFO*-Fab’2 CAR2 localized to STEAPl-high xenograft tumors in vivo, very low tumor uptake in STEAP1 -negative xenografts.
- Figure 13 depicts tumor uptake of various labeled constructs in vivo.
- STEAPl- high and STEAP1 -negative tumor bearing mice were dosed with -200 uCi radiotracers and imaged 22-24h later. Tumor uptake was quantified by measuring percent injected dose per gram of tissue (%ID/g).
- Figures 14A, B, and C depict an outline of an IRF assay (Fig. 14A), beads used in the assay (Fig. 14B), and an assay readout (Fig. 14C).
- STEAP1 Six Transmembrane Epithelial Antigen of the Prostate-1 (STEAP1) is an emerging therapeutic target for prostate cancer due to its membrane localization and overexpression in mCRPC (Gomes et al., Urol Oncol 2014;32(l):53.e23-9).
- the strategic integration of a molecular imaging technology holds the potential to facilitate decision making and enhance therapeutic outcomes in the dynamic landscape of mCRPC management by enabling patient selection and response monitoring for STEAP1 targeted therapeutics.
- Immunohistochemical (IHC) methods while valuable and widely used, face several shortcomings when compared to radiotracers (e.g. PET tracers) for patient selection.
- IHC sample collection methods are invasive, often causing pain and discomfort to patients.
- IHC assays typically yield information from a limited number of tissue samples takes at a single time point, restricting the dynamic assessment of target antigen expression and therapeutic response.
- decalcification methods can compromise accuracy of IHC results due to potential alterations of epitopes recognized by IHC antibodies.
- imaging agents offer a non-invasive approach to produce real-time whole-body information to guide clinical decisions.
- Copper-64 ( 64 Cu) has a half-life of 12.7 hours, and it decays via electron capture (44%), positron ( + emission) (17%, 0.655 MeV), and beta (0- emission) (39%, 0.573 MeV).
- the positron (0+) emissions are useful for execution of positron emission tomography (PET) imaging studies and the half-life is a good fit for imaging peptides and small proteins as it allows for up to 24h imaging post-injection.
- Zirconium-89 ( 89 Zr) has a half-life of 3.27 days, and it decays via electron capture (77%) and 0+ emission (17%, 0.396 MeV). Its longer halflife makes it a good fit for imaging full length antibodies as it allows for imaging up to 7-10 days post-injection.
- (Fab’)2 molecules demonstrate faster clearance from the body while retaining the essential specificities and affinities of full antibodies, which is desirable for an imaging agent (see e.g. Freise et. al, Molec. Immunology, vol. 67(2), part A (Oct. 2015)).
- anti-STEAPl Fab molecules were tested in comparison to (Fab’)2 molecules, and the Fab molecules had poor binding to STEAP1 whereas the (Fab’)2 molecules retained binding to STEAP1.
- radiolabeled anti-STEAPl antibody constructs are disclosed that are useful as imaging agents (e.g. PET tracer).
- the construct is a ((Fab’)2 fragment.
- the construct comprises 89 Zr.
- the construct comprises 64 Cu.
- an “anti-STEAPl construct” refers to a protein that specifically binds STEAP1.
- the anti-STEAPl construct is an antibody.
- the anti-STEAPl construct is a (Fab’)2 fragment.
- the antibody or (Fab’)2 fragment comprises the heavy chain and light chain CDRs that are in the STEAP1- binding Fabs of xaluritamig.
- the (Fab’)2 fragment is labeled with 64 Cu.
- An antigen binding protein is said to “specifically bind” to its antigen when the antigen binding protein binds its antigen with a dissociation constant (KD) is ⁇ 10' 7 M as measured via a surface plasma resonance technique (e.g., BIACore, GE-Healthcare Uppsala, Sweden) or Kinetic Exclusion Assay (KinExA, Sapidyne, Boise, Idaho).
- KD dissociation constant
- an “antibody” is an immunoglobulin molecule comprising 2 heavy chains (HCs) and 2 light chains (LCs) interconnected by disulfide bonds.
- the amino terminal portion of each LC and HC includes a variable region of about 100-120 amino acids primarily responsible for antigen recognition via the CDRs contained therein.
- the CDRs are separated with regions that are more conserved, termed framework regions (“FR”).
- FR framework regions
- Each LCVR and HCVR is composed of 3 CDRs and 4 FRs, arranged from amino-terminus to carboxyterminus in the following order: FR1, CDR1, FR2, CDR2, FR3, CDR3, FR4.
- the 3 CDRs of the LC are referred to as “LCDR1, LCDR2, and LCDR3,” and the 3 CDRs of the HC are referred to as “HCDR1, HCDR2, and HCDR3.”
- the CDRs contain most of the residues which form specific interactions with the antigen.
- an antibody of the present invention comprises a sequence comprising a C-terminal lysine.
- the antibody comprises a HC without the C-terminal lysine.
- the HC or HCVR N-terminal glutamine and/or the N- terminal glutamic acid may be converted to pyroglutamic acid.
- the HC N-terminal glutamine and/or the N-terminal glutamic acid may be converted to pyroglutamic acid, and the sequence may lack a C-terminal lysine. All forms are envisioned for the antibodies of the present invention.
- a (Fab’)2 fragment comprises an antibody LC, an antibody HCVR and CHI domain, and at least part of the hinge region.
- a (Fab’)2 fragment therefore, is essentially an antibody without an Fc region.
- a (Fab’)2 fragment can be generated by enzymatic digestion of an antibody with pepsin, thereby removing the antibody Fc region. Methods of making (Fab’)2 fragments are known in the art (see e.g., Khawli et. al, Hybrid Hybridomics. 2002 Feb;21(l):l l-8).
- a “chelator” refers to a molecule that is capable of forming a coordination complex with a radioactive metal ion such as 64 Cu or 89 Zr, enabling its stable conjugation to a targeting moiety such as an antibody or antibody fragment (e.g. (Fab’)2).
- a chelator may be conjugated to the construct by methods known in the art.
- a chelator may be conjugated via random lysine conjugation.
- Exemplary random lysine conjugation is described in the Examples herein.
- a chelator may also be conjugated via site-specific conjugation, such as conjugation to a cysteine. Exemplary site-specific conjugation is also described in the Examples herein.
- chelator to antibody ratio generally refers to the number of chelator(s) attached to the construct, which can be, for example, a (Fab’)2 fragment or an antibody.
- CAR is not intended to necessarily mean that the chelator is attached to an antibody, as defined herein, but the chelator may be attached to proteins such as a (Fab’)2 fragment.
- DFO Deroxamine
- DFO* are examples of chelators that can be used for 89 Zr and are known in the art (see e.g. Raave et. al, Eur J Nucl Med Mol Imaging 46, 1966- 1977 (2019); and Chomet et.
- NOTA 2,2',2”-(l,4,7-triazacyclononane-l,4,7-triyl)triacetic acid
- DOTA 2, 2', 2”, 2”’ -(1,4, 7,10- tetraazacyclododecane-l,4,7,10-tetrayl)tetraacetic acid
- CAR1 refers to an average of CAR1, meaning some constructs may have zero chelators attached and some constructs may have two chelators attached, but on average as a distribution, the CAR is one.
- a positron emission tomography (PET) scan is a non-invasive imaging test that detects radioactivity in a subject. It involves administration to a subject a PET tracer, which is a radiolabeled compound with positron-emitting radionuclides that binds a target. The radioactive isotope undergoes decay, emitting positrons. When a positron emitted encounters an electron in the body, they annihilate producing 2 gamma-ray photons traveling in opposite directions. Special detectors surrounding the body capture the paths and timing of these photons. A computer can then use the information to generate 3D images of the source of the annihilation event. PET imaging can be useful in diagnosing, staging, and assessing effectiveness of therapy, and treatment planning in cancer (see e.g. Krarup et. al, Seminars in Nuclear Medicine, Vol. 52;6 (Nov. 2022)).
- Radiolabeled refers to the process of incorporating a radioactive isotope into a compound (e.g. (Fab’2)) to enable their detection during imaging and/or therapy.
- Radioisotopes that may be conjugated to the compounds of the present invention include Copper 64 ( 64 Cu), Zirconium 89 ( 89 Zr), fluorine-18 ( 18 F), Gallium 68 ( 68 Ga), Iodine 123/131 ( 123 I. 131 I), Carbon-11 ( n C), Scandium-44 ( 44 Sc), Iodine-124 ( 124 I) and Yttrium-86 ( 86 Y), and Lutetium 177 ( 177 Lu). Said radiolabels are useful especially for PET imaging.
- Radiolabels that are also useful for imaging and therapy include 3 H, n C, 14 C, 18 F, 32 P, 35 S, 64 Cu, 68 Ga, 86 Y, 89 Zr, "Tc, H 1 In, 123 I, 124 I, 125 I, 131 I, 133 Xe, 177 LU, 211 At, 225 Ac, 212 Pb, and 213 Bi (see e.g. United States Patent Number 10,730,944; United States Patent Number 8,771,966; PCT Publication Number WO/2015055318; and PCT Publication Number WO/2017086021).
- Constructs of the present invention may be administered by parenteral routes, non-limiting examples of which are subcutaneous administration and intravenous administration.
- a construct of the present invention may be administered to a subject with pharmaceutically acceptable carriers, diluents, or excipients in single or multiple doses.
- Pharmaceutical compositions of the present invention can be prepared by methods well known in the art (e.g., Remington: The Science and Practice of Pharmacy, 22nd ed. (2012), A. Loyd et al., Pharmaceutical Press) and comprise a construct, as disclosed herein, and one or more pharmaceutically acceptable carriers, diluents, or excipients.
- Anti-STEAPl constructs of the present invention may be administered as part of a treatment plan for a subject having prostate cancer.
- the anti-STEAPl construct is administered with a therapeutic agent useful for treating cancer.
- the anti-STEAPl construct may be administered before administration of the therapeutic agent.
- the anti- STEAPl construct may be administered after treatment with the therapeutic agent.
- “treatment” and/or “treating” and/or “treat” are intended to refer to all processes wherein there may be a slowing, interrupting, arresting, controlling, stopping, or reversing of the progression of the disorders described herein, but does not necessarily indicate a total elimination of all disorder symptoms.
- Treatment includes administration of a therapeutic agent for treatment of a disease or condition in a human that would benefit from activity of said therapeutic agent, and includes: (a) inhibiting further progression of the disease; and (b) relieving the disease, i.e., causing regression of the disease or disorder or alleviating symptoms or complications thereof.
- a “subject” refers to a human.
- the subject has cancer.
- the subject has prostate cancer.
- the subject has metastatic resistant prostate cancer.
- the subject has metastatic castration sensitive prostate cancer.
- the constructs of the present invention will be useful in detecting STEAP1 in cancer. It is envisioned that the constructs of the present invention will be useful in detecting STEAP1 in a solid tumor. It is envisioned that the constructs of the present invention will be useful in detecting STEAP1 in metastatic sites. In an embodiment, it is envisioned that the constructs of the present invention will be useful in detecting STEAP1 in a solid tumor which has elevated expression of STEAP1. In an embodiment, the expression of STEAP1 is higher in the tumor than it is in non-cancerous cells. In an embodiment, the cancer is lung cancer. In an embodiment, the cancer is non-small cell lung carcinoma (adeno and squamous).
- the cancer is gastric cancer. In an embodiment, the cancer is esophageal cancer. In an embodiment, the cancer is colorectal cancer. In an embodiment, the cancer is hepatocellular carcinoma. In an embodiment, the cancer is pancreatic ductal adenocarcinoma. In an embodiment, the cancer is prostate cancer. In an embodiment, the cancer is metastatic castrate resistant prostate cancer. In an embodiment, the cancer is metastatic castrate sensitive prostate cancer. In an embodiment, the cancer is Ewing’s sarcoma. [0055] A metastatic site is a part of the body to which cancer cells spread outside of the original tumor site. For prostate cancer, for example, common sites of metastasis include bones, lymph nodes, lungs, and liver. Additional sites of metastasis include adrenal glands, brain, breasts, eyes, kidneys, muscles, pancreas, salivary glands, and spleen.
- a construct of the present invention detects STEAP1 in other mammalian species, such as cynomolgus monkey.
- Xaluritamig (AMG 509) is an XmAb® 2+1 T cell engager (TCE) molecule that is designed to direct T effector cells towards STEAP1 expressing cells.
- TCE T cell engager
- the preliminary efficacy results observed with xaluritamig are numerically higher than those reported for other TCEs in prostate cancer.
- Xaluritamig kills tumor cells that express high levels of STEAP1.
- a compound of the present invention can be used as part of a treatment plan with an agent for cancer treatment.
- the agent for cancer treatment is an agent for prostate cancer treatment.
- the agent for prostate cancer treatment is xaluritamig.
- a compound of the present invention may be used to determine the levels of STEAP1 expression in a subject’s prostate, thereby providing information on STEAP1 expression before and after treatment for prostate cancer with the agent for prostate cancer treatment, such as xaluritamig.
- constructs of the present invention are useful for the detection of STEAP1 in conjunction with administration of an agent for cancer treatment.
- An agent for cancer treatment refers to a drug or molecule that is useful in the treatment of cancer. Depending on the type of cancer and other factors such as stage of disease, a subject’s physician can determine the agent for cancer treatment to be administered to the subject.
- the agent for cancer treatment is a drug or molecule that binds to STEAP1.
- Non-limiting examples include antibody-drug conjugates, T-cell engager molecules, antibodies, and bispecific antibodies.
- an agent for prostate cancer treatment refers to a drug or molecule that is useful in the treatment of prostate cancer.
- the agent for prostate cancer treatment is a drug or molecule that binds STEAP1.
- the agent for prostate cancer treatment is xaluritamig. Xaluritamig is described in described in PCT Publication Number WO/2020010079.
- the agent for prostate cancer treatment comprises a sequence or more than one sequence given by any one of SEQ ID NOs 11-36.
- the agent for prostate cancer treatment is described in PCT Publication Number WO/2020010079, which is hereby incorporated by reference in its entirety.
- a compound of the present invention is useful as an imaging agent for cancer, such as prostate cancer.
- Such imaging agent is useful as a diagnostic biomarker for cancer, such as prostate cancer.
- Said diagnostic biomarker is able to detect and/or confirm disease (e.g. prostate cancer).
- Said diagnostic biomarker can also identify subjects having increased STEAP1 expression. Subjects having increased STEAP1 expression (compared to STEAP1 expression in non-cancer cells) are thought to respond more favorably to molecules that are able to kill cancer cells expressing STEAP1 such as xaluritamig.
- Said diagnostic biomarker may also be used to monitor STEAP1 expression during and after treatment with an anti-cancer therapy such as xaluritamig.
- the imaging agent can be a PET tracer, in which a subject is injected with the PET tracer, and subsequently, the subject undergoes a scan (PET scan) to detect STEAP1 in the subject. Based at least in part on the subject’s STEAP1 expression, a clinician can then recommend a treatment plan.
- Such treatment plan may include administration of an agent for prostate cancer treatment, such as xaluritamig.
- Compounds of the present invention may also be used in the course of a subject’s treatment. In an embodiment, said use is useful to determining a subject’s expression of STEAP1 during or after treatment.
- a clinician is therefore able to determine how well treatment (e.g. with xaluritamig) is working in a subject (e.g. prostate cancer patient). Based on STEAP1 expression, therefore, the clinician may alter or maintain the treatment plan.
- C4-2B Luc cells were incubated with increasing concentrations (up to 1 pM) of NOTA-(Fab’)2 (CAR1), DFO-(Fab’)2 (CAR1), or DFO*-(Fab’)2 (CAR1), or cold label 63 Cu-NOTA-(Fab’)2 (CAR1), or unconjugated (Fab’)2 , for 1 hour at 4 °C.
- C4-2B Luc or 22Rvl Luc cells were incubated with increasing concentrations (up to 1 uM) of NOTA-(Fab’)2 (CAR2) or unconjugated (Fab’)2 for 1 hour at 4°C.
- Cell-bound antibodies were detected by flow cytometry as described above and the results are shown in Figure 3 (NOTA) and Figure 4 (DFO). Binding ECso values of the molecules are shown in Table 3 (NOTA) and Table 4 (DFO).
- Immunoreactive fraction (IRF) assay is a quality control method measuring the bound (to the target) fraction of the compound, which is essential to fulfill the release criteria for compound’s use in the preclinical as well as clinical setting. An outline of the assay is shown in Figure 14. Beads preparation, IRF assay protocol, and data analysis are described below.
- STEAP1 Antigen Coated-beads and control beads preparation To prepare antigen huSTEAPl-flag coated beads (ACB), huSTEAPl-flag, and anti -flag magnetic beads (ThermoFisher) were mixed with a ratio of 120 pg huSTEAPl-flag / 200 pL beads in a final volume of 1 mL ice cold buffer containing TBS-GDN 0.005%. Following constant shaking at 1000 rpm at 4 °C for 1 hour, the beads (200 pL) were saved and washed once with 1 mL of TBS-GDN 0.005% -BSA 1%.
- ACB antigen huSTEAPl-flag coated beads
- huSTEAPl-flag huSTEAPl-flag
- anti -flag magnetic beads ThermoFisher
- the beads were then resuspended in 600 pL of TBS-GDN 0.005% -BSA 1%, and aliquoted to 80 pL slurry/vial.
- the settled beads (20 pL/vial) were stored at -80°C for long term use.
- anti -flag magnetic beads were washed once with buffer containing TBS-GND 0.005%-BSA 1%.
- the settled beads 200 pL were then resuspended in TBS-GDN 0.005%-BSA 1% (600 pL), and aliquoted to 80 pL slurry/vial.
- the settled beads (20 pL/vial) were stored at -80°C for long term use.
- IRF assay protocol Immediately following the end of PET tracer synthesis, each of the radiolabeled test articles was assessed for immunoreactivity using STEAP1 antigen coated beads (ACB). The immunoreactivity fraction >90% for a radiotracer indicates that the targeting functionality of the test article remained intact following radiolabeling.
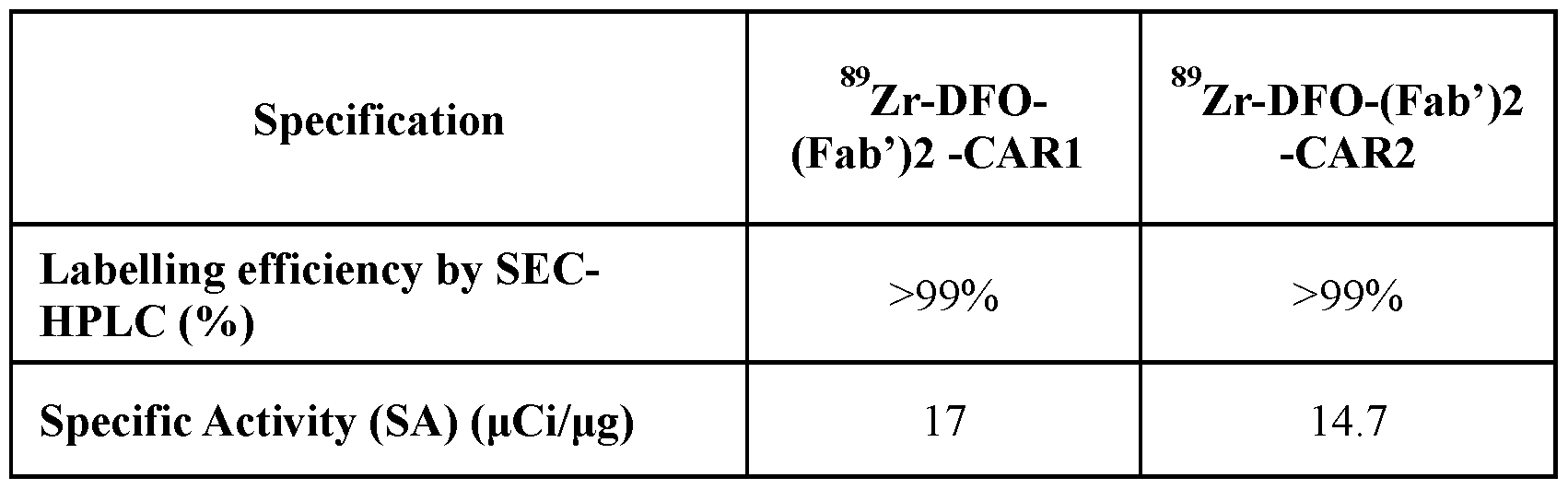
- Test articles for IRF Assay were: 89 Zr-labeled DFO-(Fab’)2 -CAR1, 89 Zr-labeled DFO-(Fab’)2 -CAR2, 89 Zr-labeled DFO*-(Fab’)2 -CAR1, 64 Cu-labeled NOTA-(Fab’)2 -CAR1, or 64 Cu-labeled NOTA-(Fab’)2 -CAR2.
- Negative control (uncoated beads) groups were also included.
- Assay buffer TBS (20 mM Tris-Cl, pH 7.5 + 150 mM NaCl) + GDN 0.005% + BSA 1% were prepared and stored at room temperature. Beads were provided in 20 pL aliquots. Vials were thawed quickly before the assay.
- the vials containing the beads and the radioligand were incubated for 1 hour at 4 °C on a rocker/shaker. After incubation, the vials were centrifuged for 15 seconds at 100 G at 4 °C and then placed on the magnetic rack for 3 minutes. After the supernatant was collected into a ‘supernatant’ vial, the beads were washed with 100 pL assay buffer, and the supernatant was collected into a ‘wash’ vial. The beads were washed a second time with 100 pL assay buffer, and the supernatant was collected into the same ‘wash’ vial. The beads were resuspended in 100 pL assay buffer and moved to a new vial labelled as ‘bound’. Gamma counting the vials: Bound; Supernatant; Wash; Reference (3 reference standards); Background (3 background vials).
- IRF (%) was calculated as following: (Bound / (Bound + supernatant + wash)) *100%
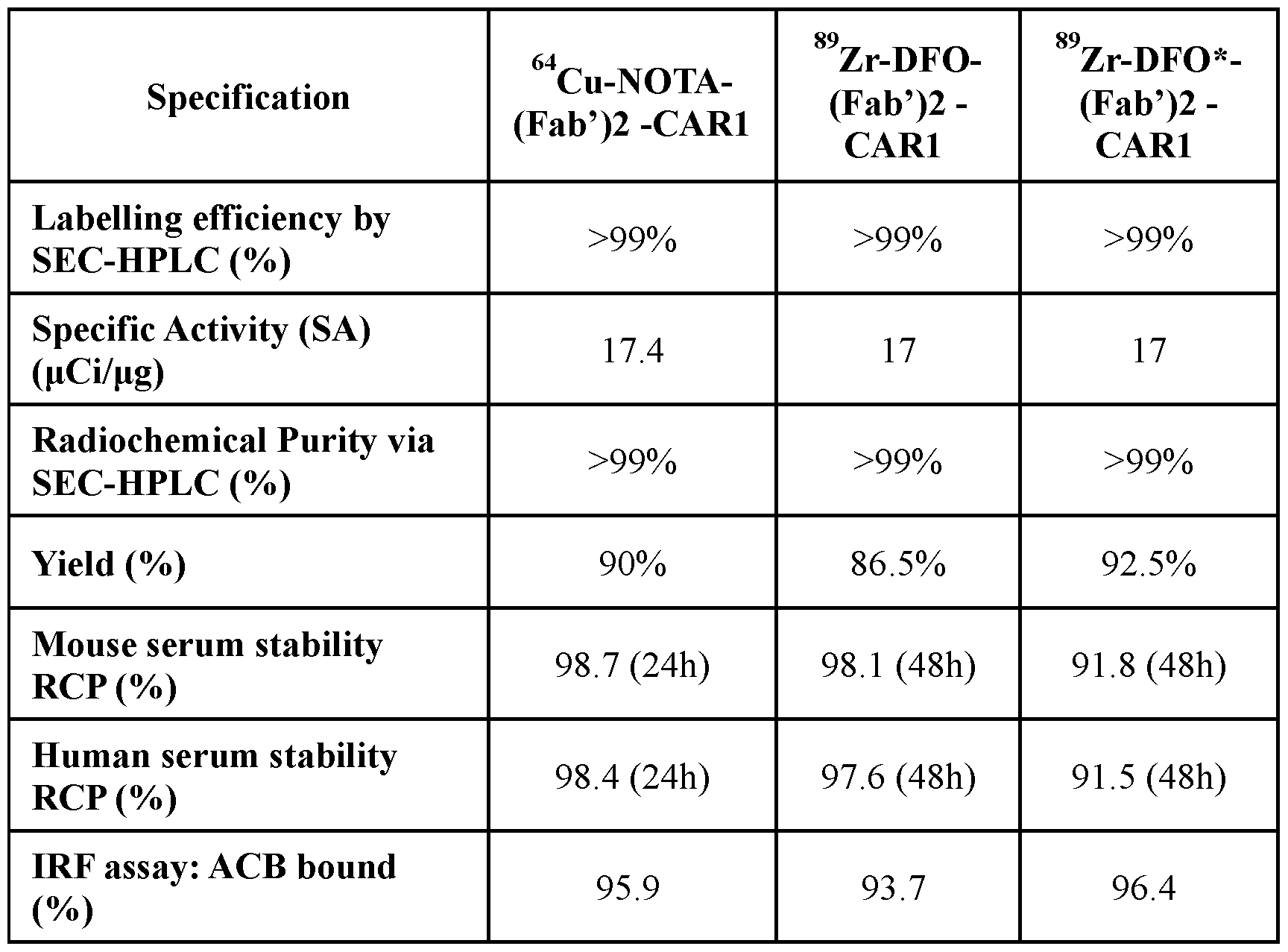
- DFO-Fab'2-CAR1 and DFO*-(Fab’)2 -CAR1 were labeled with 89 Zr with labeling efficiency and radiochemical purity >99%. Stability test showed that both labeled material were stable for up to 48 hours (maximum length of time tested) in PBS at 4 °C (Radiochemical purity (“RCP”) of >98%). Both labeled materials showed a slight decrease in RCP at 48 hours in human and mouse serum at 37 °C (RCP -91% for DFO-Fab'2-CAR1 and -97% for DFO*-(Fab’)2 -CAR1). RF release criteria was met by both radiotracers (IRF of 93.7% for DFO-Fab’2-CAR1 and 96.4% for DFO*-Fab’2-CAR1) (Table 5).
- the NOTA-Fab2-CAR1 conjugate was labeled with 64 Cu with labeling efficiency and radiochemical purity >99%.
- the purified labeled material was stable for up to 24 hours (maximum length of time tested) in PBS at 4°C (RCP of >99%) and in human and mouse serum at 37°C (RCP of >98%) IRF release criterion was met by this radiotracer (IRF of 95.9%) (Table 5).
- a Sartorius Octet HTX instrument with Streptavidin (SAX) biosensors (Sartorius, Inc. Bohemia, NY # 18-5119) was used for binding affinity analysis. All Octet binding experiments, baselines and dilutions were performed with Octet assay buffer (10 mM TRIS pH 7.5, 150 mM NaCl, 1 mM CaCh, 0.08 % GDN, 0.10 mg/mL BSA). Recombinant human STEAPl-FLAG(GDN) protein was produced as described in Nolan-Stevaux et al., Cancer Discov (October 2023).
- (Fab’)2 molecules were immobilized using an anti-human IgG (Fab’)2 goat polyclonal antibody (Jackson ImmunoResearch, West Grove, PA, #109-005-006) that was biotinylated to 2.4 biotin/molecule with the Calbiochem Innolink Biotin 354S (#203119) following the kit directions. Either black flat bottom 384 well plates or black tilted bottom plates were used for method steps. Acquisition rate was set to “High Sensitivity Kinetics” with 96 tip mode.
- Raw data was processed with Genedata Screener vl9 using the SPR affinity package and with standard Octet data processing: for each column of biosensors, the signals from the two reference biosensors were averaged and subtracted from the remaining six biosensors. The Y axis was aligned with the preceding bassline and the interstep correction was made with the dissociation step; High frequency nose was reduced with Savitzky-Golay filtering and the association and dissociation steps were isolated for each interaction pair into a sensorgram. Each grouped processed data was then globally fitted with a 1 : 1 binding model resulting in measurements for the association rate constant (ka) and the dissociation rate constant (kd). The KD affinity was then calculated as a ratio of the rate constants kd/ka.
- Three unlabeled CAR2 molecules were included in this study: CAR2 (Fab’)2 with no conjugation (“unconjugated”), (Fab’)2 CAR2 with DFO conjugation (“DFO”), and (Fab’)2 CAR2 with NOTA conjugation (“NOTA”).
- Unconjugated was administered in 10 mM NaOAc; 9% Sucrose; pH 5.2 (A52Su).
- DFO was administered in 0.1 M NaOAc, pH 5.9.
- NOTA was administered in 0.1 M NH4OAc, pH 6.0.
- Serum samples were taken 5 minutes, 1, 2, 4, 8, 24, 48, 72, 96 and 120 hours post dose. Animals were staggered in sets of 3. All plasma specimens were stored at approximately -70°C ( ⁇ 10°C) until being transferred for subsequent analysis.
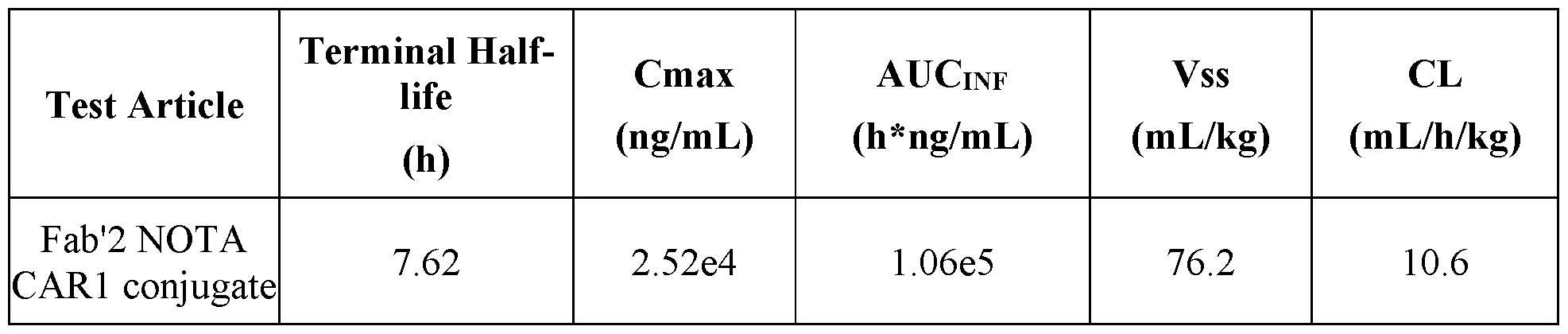
- NOTA CAR1 conjugate when given by intravenous bolus injection to cynomolgus monkeys.
- mice Male balb/c nude mice were inoculated with 5X10 6 either STEAPl-high or STEAP1 -negative cells subcutaneously in the right shoulder. When tumor size reached 150 — 400 mm 3 , mice were allocated to STEAPl-high or STEAP1 -negative groups, with 4 mice per group.
- Tumor uptake for each imaging reagent was determined by calculating the percent of injected dose per gram (%ID/g) in xenograft tumors at the above indicated time points post injection. Specific tumor uptake was determined by comparing the %ID/g values in STEAPl-high vs. STEAP1 -negative xenografts.
- Tumor uptake was compared between 64 Cu-NOTA-Fab’2-CARl and 64 Cu- NOTA-Fab’2-CAR2 in STEAPl-high ( Figure 8) and STEAP1 -negative ( Figure 9) xenografts.
- Tumor uptake was compared between 89 Zr-DFO-Fab’2-CARl and 89 Zr-DFO- Fab’2-CAR2 in STEAPl-high ( Figure 10) and STEAP1 -negative ( Figure 11) xenografts. While both molecules localized to STEAPl-high xenografts, the tumor uptake was low.
- 89 Zr-DFO*-Fab’2 CAR1 tumor uptake in STEAPl-high vs. STEAP1 -negative xenografts were compared.
- the kidneys had the highest uptake of the imaging agents among all groups. The highest kidney uptake was in the groups dosed with the 89 Zr- labeled imaging agents (18.5 - 65.4 %ID/g at 74 h), and the lowest uptake was in the groups dosed with the 64 Cu-labeled imaging agent (17.1 - 21.4 %ID/g at 46 h). Additionally, the liver uptake was high in the groups dosed with the 64 Cu-labeled imaging agent (12.8 - 15.1 %ID/g at 46 h), while it was relatively low in the groups dosed with the 89 Zr -labeled imaging agents (1.8 - 3.9 %ID/g at 74 h). EXAMPLE 8. MOLECULE ENGINEERING
- STEAP1 (Fab’)2 molecules with various conjugations and radiolabels were engineered and evaluated for activity.
- Engineered formats included STEAP1 (Fab’)2 conjugated by NOTA to 64 Cu, and STEAP1 (Fab’)2 conjugated by DFO or DFO* to 89 Zr. Conjugation was performed by random lysine conjugation.
- STEAP1 (Fab’)2 four amino acid peptide PCPP was inserted to HC of huSTEAPl_21019(VH:N67Q) after HC P230 (EU numbering). Therefore, the hinge region of STEAP1 (Fab’)2 is EPKSCDKTHTCPPCPPCPP.
- a solution of STEAP1 (Fab’)2 (12 mg at 14.759 mg/mL in 10 mM NaOAc, 9% Sucrose, pH 5.2) was buffer exchanged into HEPES buffer (0.1 M HEPES, pH 8.5) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO).
- HEPES buffer 0.1 M HEPES, pH 8.5
- Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO).
- To the resulting solution 6 mL) was added a stock solution of NOTA-NHS in DMSO (15 mM, 5 eq, 0.041 mL, from CheMatech, c 100) and the reaction mixture was heated to 37 °C with agitation for 90 minutes.
- NOTA- NHS solution (2 eq, 0.016 mL) was added and the mixture was heated to 37 °C with agitation for another 90 minutes. The mixture was cooled to room temperature and buffer exchanged into 0.1 M NH4OAc (pH 5.9) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO) to give a solution of NOTA conjugate (4 mL, 2.9 mg/mL). The product was analyzed on an Agilent 6230 LC/TOF and the NOTA:(Fab’)2 ratio was calculated to be 1.13 based on triplicate MS datasets. Data were analyzed using PMI software.
- a solution of STEAP1 (Fab’)2 (6 mg at 14.759 mg/mL in 10 mM NaOAc, 9% Sucrose, pH 5.2) was buffer exchanged into HEPES buffer (0.1 M HEPES, pH 8.5) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO).
- HEPES buffer 0.1 M HEPES, pH 8.5
- Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO).
- To the resulting solution 6 mL) was added a stock solution of NOTA-NHS in DMSO (10 mM, 3 eq, 0.018 mL, from CheMatech, c 100) and the reaction mixture was heated to 37 °C with agitation for 90 minutes.
- NOTA- NHS solution 3 eq, 0.018 mL was added and the mixture was heated to 37 °C with agitation for 1 hour.
- More NOTA-NHS solution (3 eq, 0.018 mL) was added and the mixture was heated to 37 °C with agitation for 1 hour then cooled to 4 °C overnight.
- the mixture was buffer exchanged into 0.1 M NH4OAc (pH 6) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO).
- the reaction was repeated with 6 mg of STEAP1 (Fab’)2 and the products were combined to give a solution of NOTA conjugate (4.2 mL, 2.9 mg/mL).
- the product was analyzed on an Agilent 6230 LC/TOF and the NOTA:(Fab’)2 ratio was calculated to be 2.36 based on triplicate MS datasets. Data were analyzed using PMI software.
- a solution of STEAP1 (Fab’)2 (8 mg at 14.759 mg/mL in 10 mM NaOAc, 9% Sucrose, pH 5.2) was buffer exchanged into HEPES buffer (0.1 M HEPES, pH 8.5) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO).
- HEPES buffer 0.1 M HEPES, pH 8.5
- Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO).
- To the resulting solution ( 5.5 mL) was added a stock solution of p-SCN-Bn-DFO in DMSO (6.67 mM, 3 eq, 0.038 mL, from Macrocyclics, B-705) and the reaction mixture was heated to 37 °C with agitation for 1 hour.
- a solution of STEAP1 (Fab’)2 (6 mg at 14.759 mg/mL in 10 mM NaOAc, 9% Sucrose, pH 5.2) was buffer exchanged into HEPES buffer (0.1 M HEPES, pH 8.5) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO).
- HEPES buffer 0.1 M HEPES, pH 8.5
- Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO).
- To the resulting solution 12 mL) was added a stock solution of p-SCN-Bn-DFO in DMSO (8 mM, 3 eq, 0.023 mL, from Macrocyclics, B-705) and the reaction mixture was heated to 37 °C with agitation for 90 minutes.
- a solution of STEAP1 (Fab’)2 (8 mg at 14.759 mg/mL in 10 mM NaOAc, 9% Sucrose, pH 5.2) was buffer exchanged into HEPES buffer (0.1 M HEPES, pH 8.5) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO).
- HEPES buffer 0.1 M HEPES, pH 8.5
- Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO).
- To the resulting solution ( 5.5 mL) was added a stock solution of DFO*-NCS in DMSO (8 mM, 3 eq, 0.032 mL, from Aber, AB573544) and the reaction mixture was heated to 37 °C with agitation for 1 hour.
- C4-2B Luc cells at 0.25X106 /100 uL were incubated with increasing concentrations (up to 1 pM) of anti-STEAPl mAb, anti-STEAPl (Fab’)2, or anti-STEAPl Fab for 1 hour at 4°C.
- Cell-bound antibodies were detected by flow cytometry after incubation with an AF647-conjugated anti-human IgG (H+L) secondary antibody at 5 ug/mL for 30 min at 4°C and median fluorescence intensity (MFI) of the AF647 channel at increasing concentrations of respective molecules were displayed (Top panel).
- MFI median fluorescence intensity
- the mixture was purified by Cation Exchange Chromatography and the product containing fractions were collected and buffer exchanged into 10 mM NaOAc, 9% Sucrose, pH 5.2 buffer by spin concentration using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO) to give the bis-cysteamine capped Fab’2 (1.5 mL, 10.6 mg/mL).
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Medicinal Chemistry (AREA)
- Organic Chemistry (AREA)
- Immunology (AREA)
- General Health & Medical Sciences (AREA)
- Veterinary Medicine (AREA)
- Pharmacology & Pharmacy (AREA)
- Animal Behavior & Ethology (AREA)
- Public Health (AREA)
- Biochemistry (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Biophysics (AREA)
- Genetics & Genomics (AREA)
- Molecular Biology (AREA)
- Chemical Kinetics & Catalysis (AREA)
- Oncology (AREA)
- Physics & Mathematics (AREA)
- Optics & Photonics (AREA)
- Epidemiology (AREA)
- Medicines Containing Antibodies Or Antigens For Use As Internal Diagnostic Agents (AREA)
Abstract
The present invention relates to anti-STEAP1 constructs for the use of detecting STEAP1 in a subject. The invention further relates to anti-STEAP1 (Fab')2 fragments that are radiolabeled. Said constructs are useful for imaging STEAP1 expression, for example, in a positron emission tomography scan.
Description
RADIOLABELED COMPOUNDS FOR THE DETECTION OF STEAP1
FIELD OF THE INVENTION
[0001] The present invention relates to anti-STEAPl constructs for the use of detecting STEAP1 in a human. The invention further relates to anti-STEAPl (Fab’)2 fragments that are radiolabeled. Said constructs are useful for imaging STEAP1 expression, for example, in a positron emission tomography scan.
DESCRIPTION OF THE TEXT FILE SUBMITTED ELECTRONICALLY
[0002] The present application contains a Sequence Listing, which has been submitted electronically in XML format and is hereby incorporated by reference in its entirety. The computer readable format copy of the Sequence Listing, which was created on December 10, 2024, is named 10289-W001-SEC_ST26.xml and is 41,366 bytes in size.
BACKGROUND OF THE INVENTION
[0003] Prostate cancer (PCa) is the most diagnosed malignancy and the primary contributor to cancer-related deaths among men on a global scale, with more than 1.4 million new cases and about 345,000 deaths in 2020 (Siegel et al., CA Cancer J Clin 2023;73(l): 17-48; and Sung et al., CA Cancer J Clin 2021;71(3):209-249). Androgen deprivation therapies (ADT) have been the cornerstone of treatment for prostate cancer. Although most PCa patients initially respond to ADT, the duration of response varies and about 10-50% of patients eventually progress to metastatic castration resistant prostate cancer (mCRPC), with higher rates of mortality (Akaza et al., J Glob Oncol. 2018 Sep;4: l-12). The current treatment landscape for mCRPC encompasses a range of approaches including androgen receptor targeted therapies, chemotherapies and more recently the PSMA-targeted radioligand therapy (177Lu-PSMA- 617). While these treatments offer benefits, the ongoing quest to improve overall survival and the duration of response necessitates the development of treatment options bolstered with better biomarker and patient selection strategies.
SUMMARY OF THE INVENTION
[0004] The present invention provides an anti-STEAPl construct, wherein the anti- STEAP1 construct is radiolabeled with 64Cu. In an embodiment, the anti-STEAPl construct is a (Fab’)2 fragment. In an embodiment, the anti-STEAPl construct further comprises at least one NOTA. In an embodiment, the construct comprises one NOTA. In an embodiment, the construct comprises two NOTAs. In an embodiment, at least one NOTA comprises 64Cu. In an embodiment, each NOTA comprises 64Cu. In an embodiment, the anti-STEAPl construct comprises HCDR1 comprising SEQ ID NO: 1, HCDR2 comprising SEQ ID NO: 2, HCDR3 comprising SEQ ID NO: 3, LCDR1 comprising SEQ ID NO: 4, LCDR2 comprising SEQ ID NO: 5, and LCDR3 comprising SEQ ID NO: 6. In an embodiment, the anti-STEAPl construct comprises HCVR comprising SEQ ID NO: 7, and LCVR comprising SEQ ID NO: 8. In an embodiment, the anti-STEAPl construct comprises a heavy chain (HC) fragment comprising SEQ ID NO: 9, and a light chain (LC) comprising SEQ ID NO: 10.
[0005] The present invention provides an anti-STEAPl construct, wherein the anti- STEAPl construct is radiolabeled with 64Cu, and wherein the anti-STEAPl construct is an antibody. In an embodiment, the anti-STEAPl antibody comprises HCDR1 comprising SEQ ID NO: 1, HCDR2 comprising SEQ ID NO: 2, HCDR3 comprising SEQ ID NO: 3, LCDR1 comprising SEQ ID NO: 4, LCDR2 comprising SEQ ID NO: 5, and LCDR3 comprising SEQ ID NO: 6. In an embodiment, the anti-STEAPl antibody comprises HCVR comprising SEQ ID NO: 7, and LCVR comprising SEQ ID NO: 8. In an embodiment, the anti-STEAPl antibody comprises a HC comprising SEQ ID NO: 37, and a LC comprising SEQ ID NO: 10. In an embodiment, the anti-STEAPl antibody comprises a HC comprising SEQ ID NO: 27, and a LC comprising SEQ ID NO: 28.
[0006] The present invention provides an anti-STEAPl construct, wherein the anti- STEAPl construct is radiolabeled with 89Zr. In an embodiment, the anti-STEAPl construct is a (Fab’)2 fragment. In an embodiment, the anti-STEAPl construct further comprises at least one DFO or DFO*. In an embodiment, the construct comprises one DFO or DFO*. In an embodiment, the construct comprises two DFO or DFO*. In an embodiment, at least one DFO or at least one DFO*comprises 89Zr. In an embodiment, each DFO or DFO*comprises 89Zr. In an embodiment, the anti-STEAPl construct comprises HCDR1 comprising SEQ ID NO: 1, HCDR2 comprising SEQ ID NO: 2, HCDR3 comprising SEQ ID NO: 3, LCDR1 comprising SEQ ID NO: 4, LCDR2 comprising SEQ ID NO: 5, and LCDR3 comprising SEQ ID NO: 6. In
an embodiment, the anti-STEAPl construct comprises HCVR comprising SEQ ID NO: 7, and LCVR comprising SEQ ID NO: 8. In an embodiment, the anti-STEAPl construct comprises a HC fragment comprising SEQ ID NO: 9, and a LC comprising SEQ ID NO: 10.
[0007] The present invention provides an anti-STEAPl construct, wherein the anti- STEAPl construct is radiolabeled with 89Zr, and wherein the anti-STEAPl construct is an antibody. In an embodiment, the anti-STEAPl antibody comprises HCDR1 comprising SEQ ID NO: 1, HCDR2 comprising SEQ ID NO: 2, HCDR3 comprising SEQ ID NO: 3, LCDR1 comprising SEQ ID NO: 4, LCDR2 comprising SEQ ID NO: 5, and LCDR3 comprising SEQ ID NO: 6. In an embodiment, the anti-STEAPl antibody comprises HCVR comprising SEQ ID NO: 7, and LCVR comprising SEQ ID NO: 8. In an embodiment, the anti-STEAPl antibody comprises a HC comprising SEQ ID NO: 37, and a LC comprising SEQ ID NO: 10. In an embodiment, the anti-STEAPl antibody comprises a HC comprising SEQ ID NO: 27, and a LC comprising SEQ ID NO: 28.
[0008] The present invention provides a polynucleotide encoding an amino acid sequence of a construct of the present invention. The present invention provides a vector comprising a polynucleotide of the present invention. The present invention provides a mammalian cell comprising a vector of the present invention. In an embodiment, the mammalian cell comprises two vectors of the present invention.
[0009] The present invention provides a method of detecting STEAP1 in a subject, wherein the method comprises administering to the subject an anti-STEAPl construct of the present invention. In an embodiment, the subject has cancer. In an embodiment, the subject has a solid tumor. In an embodiment, the subject has a solid tumor wherein the solid tumor expresses STEAP1. In an embodiment, the subject has prostate cancer. In an embodiment, the subject has metastatic castration resistant prostate cancer. In an embodiment, the subject has metastatic castration sensitive prostate cancer.
[0010] The present invention provides an anti-STEAPl construct of the present invention for use in detecting STEAP1 in a subject. In an embodiment, the subject has cancer. In an embodiment, the subject has a solid tumor. In an embodiment, the subject has a solid tumor wherein the solid tumor expresses STEAP1. In an embodiment, the subject has prostate cancer. In an embodiment, the subject has metastatic castration resistant prostate cancer. In an embodiment, the subject has metastatic castration sensitive prostate cancer.
[0011] The present invention provides the use of the anti-STEAPl construct of the present invention for the manufacture of an imaging agent for the detection of cancer. The
present invention provides the use of the anti-STEAPl construct of the present invention for the manufacture of an imaging agent for the treatment of cancer. In an embodiment, the cancer is a solid tumor. In an embodiment, the cancer is a solid tumor wherein the solid tumor expresses STEAP1. In an embodiment, the cancer is prostate cancer. In an embodiment, the cancer is metastatic castration resistant prostate cancer. In an embodiment, the subject has metastatic castration sensitive prostate cancer.
[0012] In an embodiment, STEAP1 is detected by positron emission tomography scan.
[0013] In an embodiment, STEAP1 is detected in a subject’s prostate. In an embodiment, STEAP1 is detected in metastatic sites.
[0014] In an embodiment, the subject is further administered an agent for cancer treatment. In an embodiment, the subject is further administered an agent for prostate cancer treatment. In an embodiment, the agent for prostate cancer treatment binds STEAP1. In an embodiment, the agent for prostate cancer treatment is an inhibitor of STEAP1. In an embodiment, the agent for prostate cancer treatment results in killing of STEAP1 -expressing cancer cells. In an embodiment, the agent for prostate cancer treatment is xaluritamig. In an embodiment, the subject has been diagnosed as having STEAP1 expression.
[0015] The present invention provides an anti-STEAPl construct of the present invention for use as a diagnostic biomarker.
[0016] The present invention provides a diagnostic biomarker comprising an anti- STEAPl construct of the present invention.
[0017] The present invention provides a kit comprising an anti-STEAPl construct of the present invention.
[0018] The present invention provides a method of treating a subject diagnosed with cancer, wherein the method comprises administering to the subject an anti-STEAPl construct of the present invention, imaging STEAP1 expression in the subject, diagnosing the subject with cancer, and administering to the subject an agent for cancer treatment. In an embodiment, the subject has elevated STEAP1 expression in the subject’s tumor. In an embodiment, the agent for cancer treatment is xaluritamig.
[0019] The present invention provides a method of treating a subject diagnosed with cancer, wherein the method comprises administering to the subject an anti-STEAPl construct of the present invention, imaging STEAP1 expression in the subject, and administering to the subject an agent for cancer treatment. In an embodiment, the subject has elevated STEAP1
expression in the subject’s tumor. In an embodiment, the agent for cancer treatment is xaluritamig.
BRIEF DESCRIPTION OF THE DRAWINGS
[0020] Figures 1A and IB depict anti-STEAPl (Fab’)2 cell binding after NOTA (CAR1) conjugation. C4-2B Luc cells were incubated with increasing concentrations of NOTA-(Fab’)2 (CAR1) (Figure 1 A), or cold label 63Cu-NOTA-(Fab’)2 (CAR1) (Figure IB), alongside unconjugated (Fab’)2 up to 1 uM, for 1 hour at 4°C. Cell-bound antibodies were detected by flow cytometry after incubation with an AF647-conjugated anti-human IgG, (Fab’)2 specific, secondary antibody and median fluorescence intensity (MFI) of the AF647 channel at increasing concentrations of respective molecules were displayed.
[0021] Figures 2A and 2B depict anti-STEAPl (Fab’)2 cell binding after DFO (CAR1) or DFO* (CAR) conjugation. C4-2B Luc cells were incubated with increasing concentrations of DFO-(Fab’)2 (CAR1) (Figure 2A), or DFO*-(Fab’)2 (CAR1) (Figure 2B), alongside unconjugated (Fab’)2 up to 3 uM, for 1 hour at 4°C. Cell-bound antibodies were detected by flow cytometry after incubation with an AF647-conjugated anti-human IgG, (Fab’)2 specific, secondary antibody and median fluorescence intensity (MFI) of the AF647 channel at increasing concentrations of respective molecules were displayed.
[0022] Figures 3A and 3B depict anti-STEAPl (Fab’)2 cell binding after NOTA (CAR2) conjugation. C4-2B Luc (Figure 3A) or 22Rvl Luc (Figure 3B) cells were incubated with increasing concentrations of NOTA-(Fab’)2 (CAR2) in comparison with unconjugated (Fab’)2 up to 1 uM, for 1 hour at 4°C. Cell-bound antibodies were detected by flow cytometry after incubation with an AF647-conjugated anti-human IgG, (Fab’)2 specific, secondary antibody and median fluorescence intensity (MFI) of the AF647 channel at increasing concentrations of respective molecules were displayed.
[0023] Figures 4A and 4B depict anti-STEAPl (Fab’)2 cell binding after DFO (CAR2) conjugation. C4-2B Luc (Figure 4A) or 22Rvl Luc (Figure 4B) cells were incubated with increasing concentrations of DFO-(Fab’)2 (CAR2) in comparison with unconjugated (Fab’)2 up to 1 uM, for 1 hour at 4°C. Cell-bound antibodies were detected by flow cytometry after incubation with an AF647-conjugated anti-human IgG, (Fab’)2 specific, secondary antibody and median fluorescence intensity (MFI) of the AF647 channel at increasing concentrations of respective molecules were displayed.
[0024] Figures 5A-5H sensorgrams depicting the binding kinetics and 1 : 1 model fits for anti-STEAPl (Fab’)2:human STEAP1 interactions. Fig. 5A) Unconjugated; Fig. 5B) NOTA-CAR1; Fig. 5C) DFO-CAR1; Fig. 5D) DFO*-CAR1; Fig. 5E) NOTA-CAR2; Fig. 5F) DFO-CAR2; Fig. 5G) 63Cu-NOTA CAR1; Fig. 5H) buffer blank negative control.
[0025] Figure 6 depicts the mean (and standard deviation) pharmacokinetic profile following 10 mg/kg administration of either NOTA-conjugated anti-STEAPl (Fab’)2 or DFO- conjugated anti-STEAPl (Fab’)2 in female BALB/c nude mice.
[0026] Figure 7 depicts mean (and SD) pharmacokinetic profile following 1.125 mg/kg administration in cynomolgus monkeys.
[0027] Figures 8A and 8B depict 64Cu-NOTA-Fab’2 CAR1 vs. CAR2 PET/CT imaging in STEAP1 positive xenografts. 22Rvl STEAPl-high cells were implanted in the right shoulder of the male balb/c nude mice. -200 uCi radiotracers 64Cu-NOTA-Fab’2 CAR1 (Figure 8 A) or CAR2 (Figure 8B) were injected via tail vein and images were acquired at 22h (for CAR1) or 24h (for CAR2) post injection. Each tumor uptake of the radiotracer was quantified by calculating the percent injected dose per gram of tissue (%ID/g). Both 64Cu- NOTA-Fab’2 CAR1 and CAR2 successfully localized to STEAPl-high xenograft tumors with comparable tumor uptake.
[0028] Figures 9A and 9B depict 64Cu-NOTA-Fab’2 CAR1 vs. CAR2 PET/CT imaging in STEAP1 -negative xenografts. 22Rvl STEAP1 -negative cells were implanted in the right shoulder of the male balb/c nude mice. -200 uCi radiotracers 64Cu-NOTA-Fab’2 CAR1 (Figure 9A) or CAR2 (Figure 9B) were injected via tail vein and images were acquired at 22h (for CAR1) or 24h (for CAR2) post injection. Each tumor uptake of the radiotracer was quantified by calculating the percent injected dose per gram of tissue (%ID/g). Very low tumor uptake was observed for both molecules as expected.
[0029] Figures 10A and 10B depict 89Zr-DFO-Fab’2 CAR1 vs. CAR2 PET/CT imaging in STEAPl-high xenografts. 22Rvl STEAPl-high cells were implanted in the right shoulder of the male balb/c nude mice. -200 uCi radiotracers 89Zr-DFO-Fab’2 -CAR1 (Figure 10A) or CAR2 (Figure 10B) were injected via tail vein and images were acquired at 22h (for CAR1) or 24h (for CAR2) post injection. Each tumor uptake of the radiotracer was quantified by calculating the percent injected dose per gram of tissue (%ID/g). Both 89Zr-DFO-Fab’2 CAR1 and CAR2 localized to STEAPl-high xenograft tumors with low tumor uptake.
[0030] Figures 11A and 11B depict 89Zr-DFO-Fab’2 CAR1 vs. CAR2 PET/CT imaging in STEAP1 -negative xenografts. 22Rvl STEAP1 -negative cells were implanted in the
right shoulder of the male balb/c nude mice. -200 uCi radiotracers 89Zr-DFO-Fab’2 -CAR1 (Figure 11 A) or CAR2 (Figure 1 IB) were injected via tail vein and images were acquired at 22h (for CAR1) or 24h (for CAR2) post injection. Each tumor uptake of the radiotracer was quantified by calculating the percent injected dose per gram of tissue (%ID/g). No obvious tumor uptake was observed for both 89Zr-DFO-Fab’2-CARl and CAR2.
[0031] Figures 12A and 12B depict 89Zr-DFO*-Fab’2 CAR1 PET Imaging in STEAP1 positive and STEAP1 -negative xenografts. 22Rvl STEAPl-high (A) and 22Rvl STEAP1-KO cells (B) were implanted in the right shoulder area of the male balb/c nude mice. -200 uCi Radiotracers were injected via tail vein and images were acquired at 24h post injection. Each tumor uptake of the radiotracer was indicated by percent injected dose per gram of tissue (%ID/g). 89Zr-DFO*-Fab’2 CAR2 localized to STEAPl-high xenograft tumors in vivo, very low tumor uptake in STEAP1 -negative xenografts.
[0032] Figure 13 depicts tumor uptake of various labeled constructs in vivo. STEAPl- high and STEAP1 -negative tumor bearing mice were dosed with -200 uCi radiotracers and imaged 22-24h later. Tumor uptake was quantified by measuring percent injected dose per gram of tissue (%ID/g).
[0033] Figures 14A, B, and C depict an outline of an IRF assay (Fig. 14A), beads used in the assay (Fig. 14B), and an assay readout (Fig. 14C).
DETAILED DESCRIPTION
[0034] Six Transmembrane Epithelial Antigen of the Prostate-1 (STEAP1) is an emerging therapeutic target for prostate cancer due to its membrane localization and overexpression in mCRPC (Gomes et al., Urol Oncol 2014;32(l):53.e23-9). The strategic integration of a molecular imaging technology holds the potential to facilitate decision making and enhance therapeutic outcomes in the dynamic landscape of mCRPC management by enabling patient selection and response monitoring for STEAP1 targeted therapeutics.
[0035] Immunohistochemical (IHC) methods, while valuable and widely used, face several shortcomings when compared to radiotracers (e.g. PET tracers) for patient selection. IHC sample collection methods are invasive, often causing pain and discomfort to patients. Furthermore, IHC assays typically yield information from a limited number of tissue samples takes at a single time point, restricting the dynamic assessment of target antigen expression and therapeutic response. Particularly in bone samples (nearly 90% of men with mCRPC present
with bone metastasis), decalcification methods can compromise accuracy of IHC results due to potential alterations of epitopes recognized by IHC antibodies. In contrast, imaging agents offer a non-invasive approach to produce real-time whole-body information to guide clinical decisions.
[0036] An 89 Zirconium-labeled anti-STEAP-1 antibody has been disclosed (PCT Publication Number WO 2011153346) and an anti-STEAPl PET imaging agent by labeling a monoclonal antibody with zirconium-89 has been studied in a Phase I/II clinical study (see e.g. O'Donoghue et al., Mol Pharm. 2019 Jul l;16(7):3083-3090). However, no further clinical studies with this imaging agent appear to be ongoing at this time. Thus, there is a clinical need for additional imaging methods in mCRPC that are non-invasive, accurate, and efficient at detecting STEAP1 -positive lesions.
[0037] Copper-64 (64Cu) has a half-life of 12.7 hours, and it decays via electron capture (44%), positron ( + emission) (17%, 0.655 MeV), and beta (0- emission) (39%, 0.573 MeV). The positron (0+) emissions are useful for execution of positron emission tomography (PET) imaging studies and the half-life is a good fit for imaging peptides and small proteins as it allows for up to 24h imaging post-injection. Zirconium-89 (89Zr) has a half-life of 3.27 days, and it decays via electron capture (77%) and 0+ emission (17%, 0.396 MeV). Its longer halflife makes it a good fit for imaging full length antibodies as it allows for imaging up to 7-10 days post-injection.
[0038] (Fab’)2 molecules demonstrate faster clearance from the body while retaining the essential specificities and affinities of full antibodies, which is desirable for an imaging agent (see e.g. Freise et. al, Molec. Immunology, vol. 67(2), part A (Oct. 2015)). Interestingly, as part of the present invention, anti-STEAPl Fab molecules were tested in comparison to (Fab’)2 molecules, and the Fab molecules had poor binding to STEAP1 whereas the (Fab’)2 molecules retained binding to STEAP1.
[0039] As the predicted human biological half-life of the (Fab’)2 was modeled as 12 hours, an attempt was made to label an anti-STEAPl (Fab’)2 fragment with 89Zr to allow for imaging out to about ten days. This interval would allow time for the radiolabeled compound (e.g. PET tracer) to clear the blood, and achieve good tumor-to-background ratios and good quantitation. The use of 64Cu might allow imaging up to 24 hours. Interestingly, two 89Zr- labeled (Fab’)2 derivatives, DFO-(Fab’)2 and DFO* (Fab’)2 were successful in imaging the STEAP1+ tumors, however, the tumor uptake was significantly lower than that achieved with 64Cu-NOTA-(Fab’)2.
[0040] Herein, radiolabeled anti-STEAPl antibody constructs are disclosed that are useful as imaging agents (e.g. PET tracer). In an embodiment, the construct is a ((Fab’)2 fragment. In an embodiment, the construct comprises 89Zr. In an embodiment, the construct comprises 64Cu.
[0041] As used herein, an “anti-STEAPl construct” refers to a protein that specifically binds STEAP1. In an embodiment, the anti-STEAPl construct is an antibody. In an embodiment, the anti-STEAPl construct is a (Fab’)2 fragment. In an embodiment, the antibody or (Fab’)2 fragment comprises the heavy chain and light chain CDRs that are in the STEAP1- binding Fabs of xaluritamig. In an embodiment, the (Fab’)2 fragment is labeled with 64Cu. [0042] An antigen binding protein is said to “specifically bind” to its antigen when the antigen binding protein binds its antigen with a dissociation constant (KD) is <10'7 M as measured via a surface plasma resonance technique (e.g., BIACore, GE-Healthcare Uppsala, Sweden) or Kinetic Exclusion Assay (KinExA, Sapidyne, Boise, Idaho).
[0043] As used herein, an “antibody” is an immunoglobulin molecule comprising 2 heavy chains (HCs) and 2 light chains (LCs) interconnected by disulfide bonds. The amino terminal portion of each LC and HC includes a variable region of about 100-120 amino acids primarily responsible for antigen recognition via the CDRs contained therein. The CDRs are separated with regions that are more conserved, termed framework regions (“FR”). Each LCVR and HCVR is composed of 3 CDRs and 4 FRs, arranged from amino-terminus to carboxyterminus in the following order: FR1, CDR1, FR2, CDR2, FR3, CDR3, FR4. The 3 CDRs of the LC are referred to as “LCDR1, LCDR2, and LCDR3,” and the 3 CDRs of the HC are referred to as “HCDR1, HCDR2, and HCDR3.” The CDRs contain most of the residues which form specific interactions with the antigen. The functional ability of an antibody to bind a particular antigen is, thus, largely influenced by the amino acid residues within the six CDRs. Assignment of amino acids to CDR domains within the LCVR and HCVR regions of the antibodies of the present invention is based on the well-known Kabat numbering convention (Kabat, et al., Ann. NY Acad. Sci. 190:382-93 (1971); Kabat et al., Sequences of Proteins of Immunological Interest, Fifth Edition, U.S. Department of Health and Human Services, NIH Publication No. 91-3242 (1991)). It is understand that other numbering conventions may also be used, such as, for example, Chothia (Chothia et al., “Canonical structures for the hypervariable regions of immunoglobulins”, Journal of Molecular Biology, 196, 901-917 (1987); Al-Lazikani et al., “Standard conformations for the canonical structures of immunoglobulins”, Journal of Molecular Biology, 273, 927-948 (1997)), and/or North (North
et al., “A New Clustering of Antibody CDR Loop Conformations”, Journal of Molecular Biology, 406, 228-256 (2011)). The IgG Fc region comprises two CH2 and two CH3 domains. [0044] In an embodiment, an antibody of the present invention comprises a sequence comprising a C-terminal lysine. In alternative aspects, the antibody comprises a HC without the C-terminal lysine. In addition, the HC or HCVR N-terminal glutamine and/or the N- terminal glutamic acid may be converted to pyroglutamic acid. In addition, the HC N-terminal glutamine and/or the N-terminal glutamic acid may be converted to pyroglutamic acid, and the sequence may lack a C-terminal lysine. All forms are envisioned for the antibodies of the present invention.
[0045] A (Fab’)2 fragment comprises an antibody LC, an antibody HCVR and CHI domain, and at least part of the hinge region. A (Fab’)2 fragment, therefore, is essentially an antibody without an Fc region. A (Fab’)2 fragment can be generated by enzymatic digestion of an antibody with pepsin, thereby removing the antibody Fc region. Methods of making (Fab’)2 fragments are known in the art (see e.g., Khawli et. al, Hybrid Hybridomics. 2002 Feb;21(l):l l-8).
[0046] As used herein, a “chelator” refers to a molecule that is capable of forming a coordination complex with a radioactive metal ion such as 64Cu or 89Zr, enabling its stable conjugation to a targeting moiety such as an antibody or antibody fragment (e.g. (Fab’)2).
[0047] A chelator may be conjugated to the construct by methods known in the art. For example, a chelator may be conjugated via random lysine conjugation. Exemplary random lysine conjugation is described in the Examples herein. A chelator may also be conjugated via site-specific conjugation, such as conjugation to a cysteine. Exemplary site-specific conjugation is also described in the Examples herein.
[0048] The phrase “chelator to antibody ratio” or “CAR” generally refers to the number of chelator(s) attached to the construct, which can be, for example, a (Fab’)2 fragment or an antibody. The use of the phrase CAR is not intended to necessarily mean that the chelator is attached to an antibody, as defined herein, but the chelator may be attached to proteins such as a (Fab’)2 fragment. DFO (Deferoxamine) and DFO* are examples of chelators that can be used for 89Zr and are known in the art (see e.g. Raave et. al, Eur J Nucl Med Mol Imaging 46, 1966- 1977 (2019); and Chomet et. al, Eur J Nucl Med Mol Imaging 48, 694-707 (2021)). NOTA (2,2',2”-(l,4,7-triazacyclononane-l,4,7-triyl)triacetic acid) and DOTA (2, 2', 2”, 2”’ -(1,4, 7,10- tetraazacyclododecane-l,4,7,10-tetrayl)tetraacetic acid) are examples of chelators that can be used for 64Cu and are known in the art (see e.g. Lee et. al, Diagnostics 2023, 13, 2649).
“CAR1,” for example, refers to an average of CAR1, meaning some constructs may have zero chelators attached and some constructs may have two chelators attached, but on average as a distribution, the CAR is one.
[0049] A positron emission tomography (PET) scan is a non-invasive imaging test that detects radioactivity in a subject. It involves administration to a subject a PET tracer, which is a radiolabeled compound with positron-emitting radionuclides that binds a target. The radioactive isotope undergoes decay, emitting positrons. When a positron emitted encounters an electron in the body, they annihilate producing 2 gamma-ray photons traveling in opposite directions. Special detectors surrounding the body capture the paths and timing of these photons. A computer can then use the information to generate 3D images of the source of the annihilation event. PET imaging can be useful in diagnosing, staging, and assessing effectiveness of therapy, and treatment planning in cancer (see e.g. Krarup et. al, Seminars in Nuclear Medicine, Vol. 52;6 (Nov. 2022)).
[0050] Radiolabeled refers to the process of incorporating a radioactive isotope into a compound (e.g. (Fab’2)) to enable their detection during imaging and/or therapy. Radioisotopes that may be conjugated to the compounds of the present invention include Copper 64 (64Cu), Zirconium 89 (89Zr), fluorine-18 (18F), Gallium 68 (68Ga), Iodine 123/131 (123I. 131I), Carbon-11 (nC), Scandium-44 (44Sc), Iodine-124 (124I) and Yttrium-86 (86Y), and Lutetium 177 (177Lu). Said radiolabels are useful especially for PET imaging. Additional radiolabels that are also useful for imaging and therapy include 3H, nC, 14C, 18F, 32P, 35S, 64Cu, 68Ga, 86Y, 89Zr, "Tc, H 1In, 123I, 124I, 125I, 131I, 133Xe, 177LU, 211At, 225 Ac, 212Pb, and 213Bi (see e.g. United States Patent Number 10,730,944; United States Patent Number 8,771,966; PCT Publication Number WO/2015055318; and PCT Publication Number WO/2016086021).
[0051] Constructs of the present invention, or a pharmaceutical composition comprising the same, may be administered by parenteral routes, non-limiting examples of which are subcutaneous administration and intravenous administration. A construct of the present invention may be administered to a subject with pharmaceutically acceptable carriers, diluents, or excipients in single or multiple doses. Pharmaceutical compositions of the present invention can be prepared by methods well known in the art (e.g., Remington: The Science and Practice of Pharmacy, 22nd ed. (2012), A. Loyd et al., Pharmaceutical Press) and comprise a construct, as disclosed herein, and one or more pharmaceutically acceptable carriers, diluents, or excipients.
[0052] Anti-STEAPl constructs of the present invention may be administered as part of a treatment plan for a subject having prostate cancer. In an embodiment, the anti-STEAPl construct is administered with a therapeutic agent useful for treating cancer. The anti-STEAPl construct may be administered before administration of the therapeutic agent. The anti- STEAPl construct may be administered after treatment with the therapeutic agent. As used interchangeably herein, “treatment” and/or “treating” and/or “treat” are intended to refer to all processes wherein there may be a slowing, interrupting, arresting, controlling, stopping, or reversing of the progression of the disorders described herein, but does not necessarily indicate a total elimination of all disorder symptoms. Treatment includes administration of a therapeutic agent for treatment of a disease or condition in a human that would benefit from activity of said therapeutic agent, and includes: (a) inhibiting further progression of the disease; and (b) relieving the disease, i.e., causing regression of the disease or disorder or alleviating symptoms or complications thereof.
[0053] A “subject” refers to a human. In an embodiment, the subject has cancer. In an embodiment, the subject has prostate cancer. In an embodiment, the subject has metastatic resistant prostate cancer. In an embodiment, the subject has metastatic castration sensitive prostate cancer.
[0054] It is envisioned that the constructs of the present invention will be useful in detecting STEAP1 in cancer. It is envisioned that the constructs of the present invention will be useful in detecting STEAP1 in a solid tumor. It is envisioned that the constructs of the present invention will be useful in detecting STEAP1 in metastatic sites. In an embodiment, it is envisioned that the constructs of the present invention will be useful in detecting STEAP1 in a solid tumor which has elevated expression of STEAP1. In an embodiment, the expression of STEAP1 is higher in the tumor than it is in non-cancerous cells. In an embodiment, the cancer is lung cancer. In an embodiment, the cancer is non-small cell lung carcinoma (adeno and squamous). In an embodiment, the cancer is gastric cancer. In an embodiment, the cancer is esophageal cancer. In an embodiment, the cancer is colorectal cancer. In an embodiment, the cancer is hepatocellular carcinoma. In an embodiment, the cancer is pancreatic ductal adenocarcinoma. In an embodiment, the cancer is prostate cancer. In an embodiment, the cancer is metastatic castrate resistant prostate cancer. In an embodiment, the cancer is metastatic castrate sensitive prostate cancer. In an embodiment, the cancer is Ewing’s sarcoma. [0055] A metastatic site is a part of the body to which cancer cells spread outside of the original tumor site. For prostate cancer, for example, common sites of metastasis include bones,
lymph nodes, lungs, and liver. Additional sites of metastasis include adrenal glands, brain, breasts, eyes, kidneys, muscles, pancreas, salivary glands, and spleen.
[0056] It is also envisioned that a construct of the present invention detects STEAP1 in other mammalian species, such as cynomolgus monkey.
[0057] Xaluritamig (AMG 509) is an XmAb® 2+1 T cell engager (TCE) molecule that is designed to direct T effector cells towards STEAP1 expressing cells. The first in human study of xaluritamig in patients with metastatic castrate resistant prostate cancer (mCRPC), was designed to assess xaluritamig safety, tolerability, pharmacokinetics (PK), and anti-tumor activity, when administered either intravenously or subcutaneously, as monotherapy or in combination with other therapies. The preliminary efficacy results observed with xaluritamig are numerically higher than those reported for other TCEs in prostate cancer. Xaluritamig kills tumor cells that express high levels of STEAP1.
[0058] A compound of the present invention can be used as part of a treatment plan with an agent for cancer treatment. In an embodiment, the agent for cancer treatment is an agent for prostate cancer treatment. In an embodiment, the agent for prostate cancer treatment is xaluritamig. A compound of the present invention may be used to determine the levels of STEAP1 expression in a subject’s prostate, thereby providing information on STEAP1 expression before and after treatment for prostate cancer with the agent for prostate cancer treatment, such as xaluritamig.
[0059] The constructs of the present invention are useful for the detection of STEAP1 in conjunction with administration of an agent for cancer treatment.
[0060] An agent for cancer treatment refers to a drug or molecule that is useful in the treatment of cancer. Depending on the type of cancer and other factors such as stage of disease, a subject’s physician can determine the agent for cancer treatment to be administered to the subject. In an embodiment, the agent for cancer treatment is a drug or molecule that binds to STEAP1. Non-limiting examples include antibody-drug conjugates, T-cell engager molecules, antibodies, and bispecific antibodies.
[0061] By way of example, an agent for prostate cancer treatment refers to a drug or molecule that is useful in the treatment of prostate cancer. In an embodiment, the agent for prostate cancer treatment is a drug or molecule that binds STEAP1. In an embodiment, the agent for prostate cancer treatment is xaluritamig. Xaluritamig is described in described in PCT Publication Number WO/2020010079. In an embodiment, the agent for prostate cancer treatment comprises a sequence or more than one sequence given by any one of SEQ ID NOs
11-36. In an embodiment, the agent for prostate cancer treatment is described in PCT Publication Number WO/2020010079, which is hereby incorporated by reference in its entirety.
[0062] A compound of the present invention is useful as an imaging agent for cancer, such as prostate cancer. Such imaging agent is useful as a diagnostic biomarker for cancer, such as prostate cancer. Said diagnostic biomarker is able to detect and/or confirm disease (e.g. prostate cancer). Said diagnostic biomarker can also identify subjects having increased STEAP1 expression. Subjects having increased STEAP1 expression (compared to STEAP1 expression in non-cancer cells) are thought to respond more favorably to molecules that are able to kill cancer cells expressing STEAP1 such as xaluritamig. Said diagnostic biomarker may also be used to monitor STEAP1 expression during and after treatment with an anti-cancer therapy such as xaluritamig.
[0063] The imaging agent can be a PET tracer, in which a subject is injected with the PET tracer, and subsequently, the subject undergoes a scan (PET scan) to detect STEAP1 in the subject. Based at least in part on the subject’s STEAP1 expression, a clinician can then recommend a treatment plan. Such treatment plan may include administration of an agent for prostate cancer treatment, such as xaluritamig. Compounds of the present invention may also be used in the course of a subject’s treatment. In an embodiment, said use is useful to determining a subject’s expression of STEAP1 during or after treatment. A clinician is therefore able to determine how well treatment (e.g. with xaluritamig) is working in a subject (e.g. prostate cancer patient). Based on STEAP1 expression, therefore, the clinician may alter or maintain the treatment plan.
EXAMPLES
EXAMPLE 1: ANTLSTEAP1 (FAB’)2 (CAR1) CELL BINDING
[0064] To determine if conjugating the anti-STEAPl (Fab’)2 to DFO, DFO* or NOTA chelators or cold-labeling anti-STEAPl (Fab’)2 NOTA molecules with 63Cu impacted binding affinity, the binding ECsos of the conjugated or cold labelled molecules were compared with that of unconjugated anti-STEAPl (Fab’)2 in STEAP1 expressing cell lines.
[0065] To determine the impact of NOTA (CAR1), DFO (CAR1), or DFO* (CAR1) conjugation or cold-labelling on anti-STEAPl (Fab’)2 binding to STEAP1 expressing cells, C4-2B Luc cells were incubated with increasing concentrations (up to 1 pM) of NOTA-(Fab’)2 (CAR1), DFO-(Fab’)2 (CAR1), or DFO*-(Fab’)2 (CAR1), or cold label 63Cu-NOTA-(Fab’)2
(CAR1), or unconjugated (Fab’)2 , for 1 hour at 4 °C. Cell-bound antibodies were detected by flow cytometry after incubation with an AF647-conjugated anti-human IgG, (Fab’)2 specific, secondary antibody and median fluorescence intensity (MFI) of the AF647 channel at increasing concentrations of respective molecules were displayed. Results for NOTA-(Fab’)2 molecules are shown in Figure 1 A and IB. Results for DFO or DFO* (Fab’)2 molecules are shown in Figure 2A and 2B. Binding ECso values of the molecules are shown in Table 1 and Table 2.
[0066] Similar experiments were performed in cell lines with various STEAP1 surface expression levels (determined by DAKO Qifikit).
Table 1. Cell binding with anti-STEAPl NOTA (CARD (Fab’)2 molecules as determined by flow cytometry.
Table 2, Cell binding with anti-STEAPl DFO or DFO* (CARD (Fab’)2 molecules as determined by flow cytometry.
[0067] These data demonstrate that anti-STEAPl (Fab’)2 cell binding was maintained after NOTA (CAR1), DFO (CAR1) or DFO*(CAR1) conjugation or cold label in cell lines with variable STEAP1 surface expression.
EXAMPLE 2: ANTI-STEAP1 (FAB’)2 (CAR2) CELL BINDING
[0068] To determine the impact of NOTA (CAR2) or DFO-(Fab’)2 (CAR2) conjugation on anti-STEAPl (Fab’)2 binding to STEAP1 expressing cells, C4-2B Luc or 22Rvl Luc cells were incubated with increasing concentrations (up to 1 uM) of NOTA-(Fab’)2 (CAR2) or unconjugated (Fab’)2 for 1 hour at 4°C. Cell-bound antibodies were detected by flow cytometry as described above and the results are shown in Figure 3 (NOTA) and Figure 4 (DFO). Binding ECso values of the molecules are shown in Table 3 (NOTA) and Table 4 (DFO). These data demonstrate that anti-STEAPl (Fab’)2 cell binding was maintained after NOTA (CAR2) or DFO (CAR2) conjugation in cell lines.
Table 3, Cell binding with anti-STEAPl NOTA (CAR2) (Fab’)2 molecules as determined by flow cytometry.
Table 4, Cell binding with anti-STEAPl DFO (CAR2) (Fab’)2 molecules as determined by flow cytometry.
EXAMPLE 3: CAR DETERMINATION
[0069] To determine the optimal chelator: antibody ratio (“CAR”), anti-STEAPl (Fab’)2 CAR2 and anti-STEAPl (Fab’)2 CAR1 radiolabeled molecules were assessed for their radiolabeling efficiencies, specific activity, radiochemical purity, yield, serum stability, and retention of immunoreactivity. Retention of immunoreactivity was determined by immunoreactive fraction (IRF) assay.
[0070] Immunoreactive fraction (IRF) assay
[0071] Immunoreactive fraction (IRF) assay is a quality control method measuring the bound (to the target) fraction of the compound, which is essential to fulfill the release criteria for compound’s use in the preclinical as well as clinical setting. An outline of the assay is shown in Figure 14. Beads preparation, IRF assay protocol, and data analysis are described below.
[0072] STEAP1 Antigen Coated-beads and control beads preparation: To prepare antigen huSTEAPl-flag coated beads (ACB), huSTEAPl-flag, and anti -flag magnetic beads (ThermoFisher) were mixed with a ratio of 120 pg huSTEAPl-flag / 200 pL beads in a final volume of 1 mL ice cold buffer containing TBS-GDN 0.005%. Following constant shaking at 1000 rpm at 4 °C for 1 hour, the beads (200 pL) were saved and washed once with 1 mL of TBS-GDN 0.005% -BSA 1%. The beads were then resuspended in 600 pL of TBS-GDN 0.005% -BSA 1%, and aliquoted to 80 pL slurry/vial. The settled beads (20 pL/vial) were stored at -80°C for long term use.
[0073] To prepare negative control (uncoated) beads, anti -flag magnetic beads were washed once with buffer containing TBS-GND 0.005%-BSA 1%. The settled beads (200 pL)
were then resuspended in TBS-GDN 0.005%-BSA 1% (600 pL), and aliquoted to 80 pL slurry/vial. The settled beads (20 pL/vial) were stored at -80°C for long term use.
[0074] IRF assay protocol: Immediately following the end of PET tracer synthesis, each of the radiolabeled test articles was assessed for immunoreactivity using STEAP1 antigen coated beads (ACB). The immunoreactivity fraction >90% for a radiotracer indicates that the targeting functionality of the test article remained intact following radiolabeling.
[0075] Test articles for IRF Assay were: 89Zr-labeled DFO-(Fab’)2 -CAR1, 89Zr-labeled DFO-(Fab’)2 -CAR2, 89Zr-labeled DFO*-(Fab’)2 -CAR1, 64Cu-labeled NOTA-(Fab’)2 -CAR1, or 64Cu-labeled NOTA-(Fab’)2 -CAR2. Negative control (uncoated beads) groups were also included. Assay buffer TBS (20 mM Tris-Cl, pH 7.5 + 150 mM NaCl) + GDN 0.005% + BSA 1% were prepared and stored at room temperature. Beads were provided in 20 pL aliquots. Vials were thawed quickly before the assay.
[0076] The vials containing the beads and the radioligand were incubated for 1 hour at 4 °C on a rocker/shaker. After incubation, the vials were centrifuged for 15 seconds at 100 G at 4 °C and then placed on the magnetic rack for 3 minutes. After the supernatant was collected into a ‘supernatant’ vial, the beads were washed with 100 pL assay buffer, and the supernatant was collected into a ‘wash’ vial. The beads were washed a second time with 100 pL assay buffer, and the supernatant was collected into the same ‘wash’ vial. The beads were resuspended in 100 pL assay buffer and moved to a new vial labelled as ‘bound’. Gamma counting the vials: Bound; Supernatant; Wash; Reference (3 reference standards); Background (3 background vials).
[0077] IRF (%) was calculated as following: (Bound / (Bound + supernatant + wash)) *100%
[0078] DFO-Fab'2-CAR1 and DFO*-(Fab’)2 -CAR1 were labeled with 89Zr with labeling efficiency and radiochemical purity >99%. Stability test showed that both labeled material were stable for up to 48 hours (maximum length of time tested) in PBS at 4 °C (Radiochemical purity (“RCP”) of >98%). Both labeled materials showed a slight decrease in RCP at 48 hours in human and mouse serum at 37 °C (RCP -91% for DFO-Fab'2-CAR1 and -97% for DFO*-(Fab’)2 -CAR1). RF release criteria was met by both radiotracers (IRF of 93.7% for DFO-Fab’2-CAR1 and 96.4% for DFO*-Fab’2-CAR1) (Table 5).
[0079] Similarly, the NOTA-Fab2-CAR1 conjugate was labeled with 64Cu with labeling efficiency and radiochemical purity >99%. The purified labeled material was stable for up to 24
hours (maximum length of time tested) in PBS at 4°C (RCP of >99%) and in human and mouse serum at 37°C (RCP of >98%) IRF release criterion was met by this radiotracer (IRF of 95.9%) (Table 5).
[0080] While a higher CAR may result in an increase in specific activity, target-specific activity was attained with the anti-STEAPl (Fab’)2 CAR1 radiolabeled molecules, rendering an increase in CAR number unnecessary (Table 6 and Table 7).
[0081] These data demonstrate that Zr-89 labelled radiotracers at CAR1 or CAR2 showed comparable good radiolabeling efficiency, specific activity, radiochemical purity, yield, and serum stability. IRF release criteria was met by both radiotracers that were used in vivo biodistribution studies.
[0082] These data demonstrate that 64Cu labelled radiotracers at CAR1 or CAR2 showed comparable good radiolabeling efficiency, specific activity, radiochemical purity, and yield. IRF release criteria was met by both radiotracers that were used in vivo biodistribution studies.
[0083] Overall, target-specific activity was attained with the STEAPl-(Fab’)2 -CAR1 radiolabeled molecules, either 64Cu or 89Zr labelling, rendering an increase in CAR number unnecessary based on in vitro assessment.
EXAMPLE 4: CONJUGATED (FAB’)2 BINDING AFFINITY
[0084] To assess whether conjugating the anti-STEAPl (Fab’)2 to 64Cu or 89Zr impacted binding affinity to STEAP1, the KD binding affinity was determined for the unconjugated anti-STEAPl (Fab’)2 and compared to the affinity of the anti-STEAPl (Fab’)2 conjugated with either DFO, DFO* or NOTA with CAR1 or CAR2. Additionally a NOTA- CAR1 63Cu (cold labeled) was measured as well. All KD binding affinities were measured in a Sartorius Octet HTX instrument.
[0085] A Sartorius Octet HTX instrument with Streptavidin (SAX) biosensors (Sartorius, Inc. Bohemia, NY # 18-5119) was used for binding affinity analysis. All Octet binding experiments, baselines and dilutions were performed with Octet assay buffer (10 mM TRIS pH 7.5, 150 mM NaCl, 1 mM CaCh, 0.08 % GDN, 0.10 mg/mL BSA). Recombinant human STEAPl-FLAG(GDN) protein was produced as described in Nolan-Stevaux et al., Cancer Discov (October 2023).
[0086] (Fab’)2 molecules were immobilized using an anti-human IgG (Fab’)2 goat polyclonal antibody (Jackson ImmunoResearch, West Grove, PA, #109-005-006) that was biotinylated to 2.4 biotin/molecule with the Calbiochem Innolink Biotin 354S (#203119) following the kit directions. Either black flat bottom 384 well plates or black tilted bottom plates were used for method steps. Acquisition rate was set to “High Sensitivity Kinetics” with 96 tip mode. Each interaction tested arose from a column of eight biosensors; each (Fab’)2 molecule was immobilized to a fixed level on eight biosensors in a column then the first six biosensors were incubated with the soluble human STEAP1 dilution series and the remaining two biosensors were incubated with assay buffer and used for reference subtraction. Testing seven (Fab’)2 molecules and buffer blank corresponds to eight columns of eight biosensors or 64 SAX biosensors which were dipped in various plate wells in a single experiment.
[0087] The Octet binding experiments following a seven step method: (1) assay buffer, 60 seconds; (2) 5 nM biotinylated anti-human IgG (Fab’)2 goat polyclonal antibody (2.4 biotin/molecule) for 2000 seconds and reached 3.8 nm loading level; (3) assay buffer, 60 seconds; (4) 10 nM anti-STEAPl (Fab’)2 molecules or a negative control antibody (one column of eight SAX biosensors per (FAB’)2 molecule or buffer blank control); final (Fab’)2 loading levels were 1.1 - 1.2 nm; (5) assay buffer, 60 seconds; (6) association step: each column of eight biosensors: a six-point, three-fold serial dilution series of human STEAP1(1- 339)-FLAG(GDN) 100 nM, 33.3 nM, 11.1 nM, 3.70 nM, 1.23 nM, 0.41 nM with the remaining
two reference biosensors just assay buffer, 1200 seconds; (7) dissociation step: assay buffer, 5400 seconds.
[0088] Raw data was processed with Genedata Screener vl9 using the SPR affinity package and with standard Octet data processing: for each column of biosensors, the signals from the two reference biosensors were averaged and subtracted from the remaining six biosensors. The Y axis was aligned with the preceding bassline and the interstep correction was made with the dissociation step; High frequency nose was reduced with Savitzky-Golay filtering and the association and dissociation steps were isolated for each interaction pair into a sensorgram. Each grouped processed data was then globally fitted with a 1 : 1 binding model resulting in measurements for the association rate constant (ka) and the dissociation rate constant (kd). The KD affinity was then calculated as a ratio of the rate constants kd/ka.
[0089] In this assay format, the unconjugated anti-STEAPl (Fab’)2 bound human STEAP1 with KD affinity 2.6 nM. All conjugated anti-STEAPl (Fab’)2 molecules bound human STEAP1 with KD affinities within 2-fold of the unconjugated form indicating the conjugation did not significantly affect the binding affinity of the anti-STEAPl (Fab’)2 to human STEAP1 receptor (Table 8). Additionally, the sensorgrams depicting the binding kinetics and 1 : 1 model fits for these interactions were indistinguishable between the unconjugated and conjugated (Fab’)2 molecules again demonstrating the NOTA, DFO, DFO* in CAR1 and CAR2 conjugations have no significant effect on the binding kinetics (Figure 5).
Table 8, Measured kinetic and affinity values for the association rate constant (ka), dissociation rate constant (kd) and calculated equilibrium dissociation constant (KD),
EXAMPLE 5: PHARMACOKINETICS AND IN VIVO STABILITY
[0090] The objective of this study was to characterize the pharmacokinetics and assess the in vivo stability of the STEAP1 compounds following a single intravenous bolus administration (5 mL/kg of 2 mg/mL dose of unlabeled CAR2 (Fab’)2 molecules for a dose of 10 mg/kg) in female BALB/c nude mice (n=9 mice per group). Three unlabeled CAR2 molecules were included in this study: CAR2 (Fab’)2 with no conjugation (“unconjugated”), (Fab’)2 CAR2 with DFO conjugation (“DFO”), and (Fab’)2 CAR2 with NOTA conjugation (“NOTA”). Unconjugated was administered in 10 mM NaOAc; 9% Sucrose; pH 5.2 (A52Su). DFO was administered in 0.1 M NaOAc, pH 5.9. NOTA was administered in 0.1 M NH4OAc, pH 6.0.
[0091] Serum samples were taken 5 minutes, 1, 2, 4, 8, 24, 48, 72, 96 and 120 hours post dose. Animals were staggered in sets of 3. All plasma specimens were stored at approximately -70°C (±10°C) until being transferred for subsequent analysis.
[0092] Quantitation of the (Fab’)2 CAR2 molecules in mouse serum was performed using electrochemiluminescent immunoassays with a biotinylated anti-idiotype mAb that interacts with the anti-STEAPl binding domain as the capture reagent and a mouse anti-human IgG Fab detection reagent. Analyte concentrations were interpolated from standard curves using the protein of interest and data analyses were performed in non-GLP Watson L.
[0093] Analysis was performed using Phoenix Winonlin. Data for serum drug concentration vs. relative nominal time post dose were used for all pharmacokinetic analyses. Mean (and standard deviation) profiles are shown in Figure 6. Noncompartmental analyses were performed. AUC values were calculated using the linear log-linear trapezoidal interpolation method and used to estimate clearance and volume of distribution as shown in Table 9. Terminal half-life is also reported in Table 9. Pharmacokinetic profiles as well as pharmacokinetic parameters show distinct exposures for each of the three molecules evaluated in this study.
Table 9, Summary pharmacokinetic parameters following 10 mg/kg IV administration of (Fab’)2 CAR2 to female BALB/c mouse.
EXAMPLE 6: PHARMACOKINETIC ANALYSIS IN CYNOMOLGUS MONKEYS
[0094] The objective of this study was to determine the pharmacokinetics of a Fab'2
NOTA CAR1 conjugate when given by intravenous bolus injection to cynomolgus monkeys. This study included 3 naive male cynomolgus monkeys of Asian origin (2.7 to 2.9 years of age) weighing between 1.8 to 2.0 kg and at time of dosing. All monkeys received a single bolus intravenous dose of 1.5 mL/kg of 0.75 mg/mL unlabeled Fab'2 NOTA CAR1 conjugate for a dose of 1.125 mg/kg in 0.1 M NH4OAc buffer, pH 5.9. Serum samples were taken 5 minutes, 1, 2, 4, 8, 12, 16, 24, 48, 72, 96, 120, and 168 hours post dose.
[0095] Quantitation of the Fab'2 NOTA CAR1 conjugate in cynomolgus monkey serum was performed using an electrochemiluminescent immunoassay with a biotinylated antiidiotype mAb that interacts with the anti-STEAPl binding domain as the capture reagent and a mouse anti-human IgG Fab detection reagent. Analyte concentrations were interpolated from a standard curve using the protein of interest and data analyses were performed in non-GLP Watson LIMS.
[0096] Analysis was also performed. Data for serum drug concentration versus relative nominal time post dose were used for all pharmacokinetic analyses. Noncompartmental analyses were performed for each individual animal. AUC values were calculated using the linear log-linear trapezoidal interpolation method and used to estimate clearance and volume of distribution as shown in Error! Reference source not found.10. Terminal half-life is also reported in 9. Mean (and standard deviation) profiles are shown in Figure 7.
Table 10. Mean pharmacokinetic parameters following administration of 1,125 mg/kg of Fab'2 NOTA CAR1 conjugate to cynomolgus monkeys.
EXAMPLE 7. IN VIVO IMAGING
[0097] To determine the uptake of 64Cu labelled anti-STEAPl Fab’2 and 89Zr labelled anti-STEAPl Fab’2 molecules in vivo in tumor bearing mice, the biodistribution of each molecule was assessed in mice with STEAPl-high (STEAP1 overexpressing 22Rvl Luc cells) and STEAP1 -negative (STEAP1 knock out 22Rvl Luc cells) tumors.
[0098] Male balb/c nude mice were inoculated with 5X106 either STEAPl-high or STEAP1 -negative cells subcutaneously in the right shoulder. When tumor size reached 150 — 400 mm3, mice were allocated to STEAPl-high or STEAP1 -negative groups, with 4 mice per group.
[0099] Five constructs were included: 64Cu-NOTA-Fab’2-CARl, 64Cu-NOTA-Fab’2- CAR2, 89Zr-DFO-Fab’2-CARl, 89Zr-DFO-Fab’2-CAR2, and 89Zr-DFO*-Fab’2-CARl.
[00100] Each construct was labeled on the day of injection and all productions met the release criteria for radiochemical purity (>90%).
[00101] On the day of injection, mice were injected intravenously with one of the constructs at -200 uCi per mouse. All groups were imaged by PET/CT in a 4-bed mouse hotel. Thirty-minute static acquisitions were acquired at 2, 22-24, 46, and 74 h for 89Zr-labeled imaging agents, and at 2 and 22 -24 h and 46 h for 64Cu-labeled imaging agent. Following each PET acquisition, a CT was taken for anatomic co-regi strati on and attenuation and scatter corrections. A blood sample was taken for gamma counting after the 22-24 hour imaging timepoint. Immediately following the last imaging timepoint, all animals on study were humanely euthanized and tissues were collected for ex vivo radioactivity analysis by gamma counting.
[00102] Tumor uptake for each imaging reagent was determined by calculating the percent of injected dose per gram (%ID/g) in xenograft tumors at the above indicated time
points post injection. Specific tumor uptake was determined by comparing the %ID/g values in STEAPl-high vs. STEAP1 -negative xenografts.
[00103] Tumor uptake was compared between 64Cu-NOTA-Fab’2-CARl and 64Cu- NOTA-Fab’2-CAR2 in STEAPl-high (Figure 8) and STEAP1 -negative (Figure 9) xenografts. 64Cu-NOTA-Fab’2-CARl and 64Cu-NOTA-Fab’2-CAR2 successfully localized to STEAPl- high xenograft tumors with comparable tumor uptake (Average %ID/g = 11.9 for CAR1 and 11.8 for CAR2). Very low tumor uptake was observed in STEAP1 -negative xenografts (Average %ID/g = 3.5 for CAR1 and 3.3 for CAR2) consistent with specific tumor uptake in STEAPl-high tumors.
[00104] Tumor uptake was compared between 89Zr-DFO-Fab’2-CARl and 89Zr-DFO- Fab’2-CAR2 in STEAPl-high (Figure 10) and STEAP1 -negative (Figure 11) xenografts. While both molecules localized to STEAPl-high xenografts, the tumor uptake was low.
[00105] 89Zr-DFO*-Fab’2 CAR1 tumor uptake in STEAPl-high vs. STEAP1 -negative xenografts were compared. 89Zr-DFO*-Fab’2 CAR1 successfully localized to STEAPl-high tumors (%ID/g =7.9 for STEAPl-high and 2.7 for STEAP1 -negative).
[00106] In summary, 64Cu-NOTA-Fab’2-CARl and CAR2 were successful in imaging STEAPl-high tumors, the tumor uptake for both molecules were almost identical. 89Zr-labeled Fab’2 derivatives, DFO-Fab’2 and DFO*-Fab’2 was successful in imaging the STEAP1+ tumors, however, the tumor uptake was significantly lower than that achieved with 64Cu- NOTA-Fab’2. DFO*-Fab’2 is superior to DFO-Fab’2. Quantified results are shown in Figure 13.
[00107] In addition to tumor update, all five imaging agents showed rapid clearance from the blood as observed from the low concentration of the imaging agents at 22 h in blood from ex-vivo data. For example, in the STEAPl-high groups the blood had 1.32 ± 0.04 %ID/g for 64Cu-NOTA-Fab’2, 0.48± 0.13 % ID/g for 89Zr-DFO-Fab’2, and 1.1 ± 0.04 %ID/g for 89Zr- DFO*-Fab’2.
[00108] Among clearance organs, the kidneys had the highest uptake of the imaging agents among all groups. The highest kidney uptake was in the groups dosed with the 89Zr- labeled imaging agents (18.5 - 65.4 %ID/g at 74 h), and the lowest uptake was in the groups dosed with the 64Cu-labeled imaging agent (17.1 - 21.4 %ID/g at 46 h). Additionally, the liver uptake was high in the groups dosed with the 64Cu-labeled imaging agent (12.8 - 15.1 %ID/g at 46 h), while it was relatively low in the groups dosed with the 89Zr -labeled imaging agents (1.8 - 3.9 %ID/g at 74 h).
EXAMPLE 8. MOLECULE ENGINEERING
[00109] STEAP1 (Fab’)2 molecules with various conjugations and radiolabels were engineered and evaluated for activity. Engineered formats included STEAP1 (Fab’)2 conjugated by NOTA to 64Cu, and STEAP1 (Fab’)2 conjugated by DFO or DFO* to 89Zr. Conjugation was performed by random lysine conjugation. To enable recombinant production of STEAP1 (Fab’)2 , four amino acid peptide PCPP was inserted to HC of huSTEAPl_21019(VH:N67Q) after HC P230 (EU numbering). Therefore, the hinge region of STEAP1 (Fab’)2 is EPKSCDKTHTCPPCPPCPP.
[00110] Synthesis of STEAP1 (Fab’)2 -NOTA CAR1 conjugate:
[00111] A solution of STEAP1 (Fab’)2 (12 mg at 14.759 mg/mL in 10 mM NaOAc, 9% Sucrose, pH 5.2) was buffer exchanged into HEPES buffer (0.1 M HEPES, pH 8.5) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO). To the resulting solution ( 6 mL) was added a stock solution of NOTA-NHS in DMSO (15 mM, 5 eq, 0.041 mL, from CheMatech, c 100) and the reaction mixture was heated to 37 °C with agitation for 90 minutes. More NOTA- NHS solution (2 eq, 0.016 mL) was added and the mixture was heated to 37 °C with agitation for another 90 minutes. The mixture was cooled to room temperature and buffer exchanged into 0.1 M NH4OAc (pH 5.9) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO) to give a solution of NOTA conjugate (4 mL, 2.9 mg/mL). The product was analyzed on an Agilent 6230 LC/TOF and the NOTA:(Fab’)2 ratio was calculated to be 1.13 based on triplicate MS datasets. Data were analyzed using PMI software.
[00112] Synthesis of STEAP1 (Fab’)2 -NOTA CAR2 conjugate:
[00113] A solution of STEAP1 (Fab’)2 (6 mg at 14.759 mg/mL in 10 mM NaOAc, 9% Sucrose, pH 5.2) was buffer exchanged into HEPES buffer (0.1 M HEPES, pH 8.5) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO). To the resulting solution ( 6 mL) was added a stock solution of NOTA-NHS in DMSO (10 mM, 3 eq, 0.018 mL, from CheMatech, c 100) and the reaction mixture was heated to 37 °C with agitation for 90 minutes. More NOTA- NHS solution (3 eq, 0.018 mL) was added and the mixture was heated to 37 °C with agitation for 1 hour. More NOTA-NHS solution (3 eq, 0.018 mL) was added and the mixture was heated to 37 °C with agitation for 1 hour then cooled to 4 °C overnight. The mixture was buffer
exchanged into 0.1 M NH4OAc (pH 6) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO). The reaction was repeated with 6 mg of STEAP1 (Fab’)2 and the products were combined to give a solution of NOTA conjugate (4.2 mL, 2.9 mg/mL). The product was analyzed on an Agilent 6230 LC/TOF and the NOTA:(Fab’)2 ratio was calculated to be 2.36 based on triplicate MS datasets. Data were analyzed using PMI software.
[00114] Synthesis of STEAP1 (Fab’)2 -DFO CAR1 conjugate:
[00115] A solution of STEAP1 (Fab’)2 (8 mg at 14.759 mg/mL in 10 mM NaOAc, 9% Sucrose, pH 5.2) was buffer exchanged into HEPES buffer (0.1 M HEPES, pH 8.5) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO). To the resulting solution ( 5.5 mL) was added a stock solution of p-SCN-Bn-DFO in DMSO (6.67 mM, 3 eq, 0.038 mL, from Macrocyclics, B-705) and the reaction mixture was heated to 37 °C with agitation for 1 hour. More p-SCN-Bn-DFO in DMSO (3 eq, 0.038 mL) was added and the mixture was heated to 37 °C with agitation for another 90 minutes. The mixture was cooled to room temperature for 2 hours and buffer exchanged into 0.1 M NaOAc (pH 5.9) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO) to give a solution of DFO conjugate (2.5 mL, 2.7 mg/mL). The product was analyzed on an Agilent 6230 LC/TOF and the DFO:(Fab’)2 ratio was calculated to be 1.2 based on triplicate MS datasets. Data were analyzed using PMI software.
[00116] Synthesis of STEAP1 (Fab’)2 -DFO CAR2 conjugate:
[00117] A solution of STEAP1 (Fab’)2 (6 mg at 14.759 mg/mL in 10 mM NaOAc, 9% Sucrose, pH 5.2) was buffer exchanged into HEPES buffer (0.1 M HEPES, pH 8.5) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO). To the resulting solution ( 12 mL) was added a stock solution of p-SCN-Bn-DFO in DMSO (8 mM, 3 eq, 0.023 mL, from Macrocyclics, B-705) and the reaction mixture was heated to 37 °C with agitation for 90 minutes. More p-SCN-Bn-DFO in DMSO (3 eq, 0.046 mL) was added every hour and the mixture was heated to 37 °C with agitation for another 4 hours. The mixture was cooled to 4 °C overnight and buffer exchanged into 0.1 M NaOAc (pH 5.9) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO). The reaction was repeated with 12 mg of STEAP1 (Fab’)2 and the products were combined to give a solution of DFO conjugate (4.2 mL, 5.5 mg/mL). The product was analyzed on an Agilent 6230 LC/TOF and the DFO:(Fab’)2 ratio was calculated to be 2.63 based on triplicate MS datasets. Data was analyzed using PMI software.
[00118] Synthesis of STEAP1 (Fab’)2 -DFO* conjugate
[00119] A solution of STEAP1 (Fab’)2 (8 mg at 14.759 mg/mL in 10 mM NaOAc, 9% Sucrose, pH 5.2) was buffer exchanged into HEPES buffer (0.1 M HEPES, pH 8.5) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO). To the resulting solution ( 5.5 mL) was added a stock solution of DFO*-NCS in DMSO (8 mM, 3 eq, 0.032 mL, from Aber, AB573544) and the reaction mixture was heated to 37 °C with agitation for 1 hour. More DFO*-NCS in DMSO (3 eq, 0.032 mL) was added and the mixture was heated to 37 °C with agitation for another 90 minutes. More DFO*-NCS in DMSO (1 eq, 0.010 mL) was added every hour for four hours. The mixture was buffer exchanged into 0.1 M NaOAc (pH 5.9) using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO) to give a solution of DFO* conjugate (2.5 mL, 2.6 mg/mL). The product was analyzed on an Agilent 6230 LC/TOF and the DFO*:(Fab’)2 ratio was calculated to be 1.03 based on triplicate MS datasets. Data were analyzed using PMI software.
EXAMPLE 10: CELL BINDING
[00120] C4-2B Luc cells at 0.25X106 /100 uL were incubated with increasing concentrations (up to 1 pM) of anti-STEAPl mAb, anti-STEAPl (Fab’)2, or anti-STEAPl Fab for 1 hour at 4°C. Cell-bound antibodies were detected by flow cytometry after incubation with an AF647-conjugated anti-human IgG (H+L) secondary antibody at 5 ug/mL for 30 min at 4°C and median fluorescence intensity (MFI) of the AF647 channel at increasing concentrations of respective molecules were displayed (Top panel). MFI (AF647) from primary antibodies at 300 nM were plotted in histograms and compared to the secondary only.
[00121] The results demonstrate EC50 values of 15.68 pM for the mAb, 4.322 pM for the (Fab’)2, and 207,907 pM for the Fab. These data indicate that the Fab was not able to bind STEAP1, whereas the mAb and (Fab’)2 bound STEAP1.
EXAMPLE 11 : SITE-SPECIFIC CONJUGATION
[00122] To prepare constructs via site-specific conjugation, anti-STEAPl (Fab’)2 was engineered with a cysteine mutation on the LC for site-specific conjugation.
[00123] Synthesis of STEAP1 (Fab’)2 D88C-NOTA CAR2 conjugate:
[00124] To a solution of STEAP1 (Fab’)2 D88C (20 mg in 10 mM NaOAc, 9% Sucrose, pH 5.2) was added 15 mL solution of 1 mM cysteamine, 2.5 mM cystamine in 40 mM HEPES buffer, pH 8.5. The mixture was kept at room temperature for 1 hour and the solution was diluted with 100 mM NaOAc buffer, pH 5.0. The mixture was purified by Cation Exchange Chromatography and the product containing fractions were collected and buffer exchanged into 10 mM NaOAc, 9% Sucrose, pH 5.2 buffer by spin concentration using Amicon Ultra-15 Centrifugal Filter Unit (30K MWCO) to give the bis-cysteamine capped Fab’2 (1.5 mL, 10.6 mg/mL).
[00125] To a solution of the bis-cysteamine capped Fab’2 (4.5 mg in 10 mM NaOAc, 9% Sucrose, pH 5.2 ) was added a TPPTS solution in water ( 3.85 mM, 4 eq, 0.049 mL) and the mixture was kept at room temperature for 1 hour. The reaction mixture was then buffer exchanged into PBS by spin concentration using Amicon Ultra- 15 Centrifugal Filter Unit (3 OK MWCO). To the resulting solution was added a DHAA solution in water ( 4 mM, 10 eq., 0.112 mL) and the mixture was kept at room temperature for 2 h. The mixture was then divided evenly between two vials. A stock solution of Maleimido-mono-amide-NOTA in DMSO (10 mM, 4 equivalents, 0.008 mL, from Macrocyclics, B-622) was added to one of the vials containing the solution after DHAA oxidation. The resulting mixture was then agitated at room temperature for 1 hour. The reaction mixture was buffer exchanged into 0.1 M NaOAc, pH 5.9 buffer to give the Fab’2 D88C -NOTA CAR2 conjugate (0.8 mL, 2.3 mg/mL).
[00126] Synthesis of STEAP1 (Fab’)2 D88C-DFO CAR2 conjugate:
[00127] To one of the vials containing Fab’2 solution after DHAA oxidation was added a stock solution of DFO-Mal eimide in DMSO (10 mM, 4 eq., 0.008 mL, From Macrocyclics, B-772) and the mixture was kept at room temperature with agitation for 1 hour. The reaction mixture was buffer exchanged into 0.1 M NaOAc, pH 5.9 buffer to give the Fab’2 D88C-DFO CAR2 conjugate (0.8 mL, 2.36 mg/mL).
[00128] Both NOTA and DFO conjugates were prepared and tested in binding assays to determine if conjugation impacts binding to cells. Binding assays evaluated binding of constructs to C4-2B luc cells (STEAP1 high cells) and 22Rvl luc cells (STEAP1 low cells).
[00129] These data indicate that conjugation of Fab'2 to either NOTA or DFO does not impact binding to cells.
Claims
1. An anti-STEAPl construct, wherein the anti-STEAPl construct is radiolabeled with 64Cu.
2. The anti-STEAPl construct of Claim 1, wherein the anti-STEAPl construct is a (Fab’)2 fragment.
3. The anti-STEAPl construct of Claim 1 or Claim 2, wherein the anti-STEAPl construct further comprises at least one NOTA.
4. The anti-STEAPl construct of Claim 3, wherein the construct comprises one NOTA.
5. The anti-STEAPl construct of Claim 3, wherein the construct comprises two NOTAs.
6. The anti-STEAPl construct of any one of Claims 3-5, wherein at least one NOTA comprises 64Cu.
7. The anti-STEAPl construct of any one of Claims 1-6, wherein the anti-STEAPl construct comprises HCDR1 comprising SEQ ID NO: 1, HCDR2 comprising SEQ ID NO: 2, HCDR3 comprising SEQ ID NO: 3, LCDR1 comprising SEQ ID NO: 4, LCDR2 comprising SEQ ID NO: 5, and LCDR3 comprising SEQ ID NO: 6.
8. The anti-STEAPl construct of any one of Claims 1-7, wherein the anti-STEAPl construct comprises HCVR comprising SEQ ID NO: 7, and LCVR comprising SEQ ID NO: 8.
9. The anti-STEAPl construct of any one of Claims 1-8, wherein the anti-STEAPl construct comprises a HC fragment comprising SEQ ID NO: 9, and a LC comprising SEQ ID NO: 10.
10. An anti-STEAPl construct, wherein the anti-STEAPl construct is radiolabeled with 64Cu, and wherein the anti-STEAPl construct is an antibody that comprises a HC comprising SEQ ID NO: 37, and a LC comprising SEQ ID NO: 10.
11. A method of detecting STEAP1 in a subject, wherein the method comprises administering to the subject an anti-STEAPl construct of any one of Claims 1-10.
12. The method of Claim 11, further comprising detecting STEAP1 by positron emission tomography scan.
13. The method of Claim 11 or 12, wherein STEAP1 is detected in the subject’s prostate.
14. The method of Claim 11 or 12, wherein STEAP1 is detected in metastatic sites.
15. The method of any one of Claims 11-14, wherein the subject is further administered an agent for cancer treatment.
16. The method of Claim 15, wherein the agent for cancer treatment is xaluritamig.
17. The anti-STEAPl construct of any one of Claims 1-10 for use in detecting STEAP1 in a subject.
18. Use of the anti-STEAPl construct of any one of Claims 1-10 for the manufacture of an imaging agent for the treatment of cancer.
19. The use of Claim 17 or 18„ wherein the subject has cancer.
20. The use of Claim 17 or 18, wherein the subject has prostate cancer.
21. The anti-STEAPl construct of any one of Claims 1-10 for use as a diagnostic biomarker.
22. A diagnostic biomarker comprising the anti-STEAPl construct of any one of Claims 1-10.
23. A kit comprising the anti-STEAPl construct of any one of Claims 1-10.
24. A method of treating a subject diagnosed with cancer, wherein the method comprises administering to the subject the anti-STEAPl construct of any one of Claims 1-10, imaging STEAP1 expression in the subject, and administering to the subject an agent for cancer treatment.
25. The method of Claim 24, wherein the subject has elevated STEAP1 expression in the subject’s cancer.
26. The method of Claim 24 or 25, wherein the agent for cancer treatment is xaluritamig.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363609667P | 2023-12-13 | 2023-12-13 | |
| US63/609,667 | 2023-12-13 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025128807A1 true WO2025128807A1 (en) | 2025-06-19 |
Family
ID=94283586
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/059745 Pending WO2025128807A1 (en) | 2023-12-13 | 2024-12-12 | Radiolabeled compounds for the detection of steap1 |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025128807A1 (en) |
Citations (8)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011153346A1 (en) | 2010-06-03 | 2011-12-08 | Genentech, Inc. | Immuno-pet imaging of antibodies and immunoconjugates and uses therefor |
| WO2015055318A1 (en) | 2013-10-18 | 2015-04-23 | Deutsches Krebsforschungszentrum | Labeled inhibitors of prostate specific membrane antigen (psma), their use as imaging agents and pharmaceutical agents for the treatment of prostate cancer |
| WO2016086021A1 (en) | 2014-11-25 | 2016-06-02 | Bristol-Myers Squibb Company | Novel pd-l1 binding polypeptides for imaging |
| US20170043034A1 (en) * | 2014-01-24 | 2017-02-16 | Genentech, Inc. | Methods of using anti-steap1 antibodies and immunoconjugates |
| WO2019157340A1 (en) * | 2018-02-08 | 2019-08-15 | Amgen Inc. | Low ph pharmaceutical antibody formulation |
| WO2020010079A2 (en) | 2018-07-02 | 2020-01-09 | Amgen Inc. | Anti-steap1 antigen-binding protein |
| US10730944B2 (en) | 2017-07-24 | 2020-08-04 | Regeneron Pharmaceuticals, Inc. | Anti-CD8 antibodies and uses thereof |
| US20220348686A1 (en) * | 2019-09-05 | 2022-11-03 | Memorial Sloan Kettering Cancer Center | Anti-steap1 antibodies and uses thereof |
-
2024
- 2024-12-12 WO PCT/US2024/059745 patent/WO2025128807A1/en active Pending
Patent Citations (9)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2011153346A1 (en) | 2010-06-03 | 2011-12-08 | Genentech, Inc. | Immuno-pet imaging of antibodies and immunoconjugates and uses therefor |
| US8771966B2 (en) | 2010-06-03 | 2014-07-08 | Genentech, Inc. | Immuno-PET imaging of antibodies and immunoconjugates and uses therefor |
| WO2015055318A1 (en) | 2013-10-18 | 2015-04-23 | Deutsches Krebsforschungszentrum | Labeled inhibitors of prostate specific membrane antigen (psma), their use as imaging agents and pharmaceutical agents for the treatment of prostate cancer |
| US20170043034A1 (en) * | 2014-01-24 | 2017-02-16 | Genentech, Inc. | Methods of using anti-steap1 antibodies and immunoconjugates |
| WO2016086021A1 (en) | 2014-11-25 | 2016-06-02 | Bristol-Myers Squibb Company | Novel pd-l1 binding polypeptides for imaging |
| US10730944B2 (en) | 2017-07-24 | 2020-08-04 | Regeneron Pharmaceuticals, Inc. | Anti-CD8 antibodies and uses thereof |
| WO2019157340A1 (en) * | 2018-02-08 | 2019-08-15 | Amgen Inc. | Low ph pharmaceutical antibody formulation |
| WO2020010079A2 (en) | 2018-07-02 | 2020-01-09 | Amgen Inc. | Anti-steap1 antigen-binding protein |
| US20220348686A1 (en) * | 2019-09-05 | 2022-11-03 | Memorial Sloan Kettering Cancer Center | Anti-steap1 antibodies and uses thereof |
Non-Patent Citations (17)
| Title |
|---|
| A. LOYD ET AL.: "Remington: The Science and Practice of Pharmacy", 2012, PHARMACEUTICAL PRESS |
| AKAZA ET AL., J GLOB ONCOL., vol. 4, September 2018 (2018-09-01), pages 1 - 12 |
| AL-LAZIKANI ET AL.: "Standard conformations for the canonical structures of immunoglobulins", JOURNAL OF MOLECULAR BIOLOGY, vol. 273, 1997, pages 927 - 948, XP004461383, DOI: 10.1006/jmbi.1997.1354 |
| CHOMET, EUR J NUCL MED MOL IMAGING, vol. 48, 2021, pages 694 - 707 |
| CHOTHIA ET AL.: "Canonical structures for the hypervariable regions of immunoglobulins", JOURNAL OF MOLECULAR BIOLOGY, vol. 196, 1987, pages 901 - 917, XP024010426, DOI: 10.1016/0022-2836(87)90412-8 |
| FREISE, MOLEC. IMMUNOLOGY, vol. 67, no. 2, October 2015 (2015-10-01) |
| GOMES ET AL., UROL ONCOL, vol. 32, no. 1, 2014, pages 23 - 9 |
| KABAT ET AL., ANN. NY ACAD. SCI., vol. 190, 1971, pages 382 - 93 |
| KABAT ET AL.: "Sequences of Proteins of Immunological Interest", 1991, NIH PUBLICATION |
| KHAWLI, HYBRID HYBRIDOMICS., vol. 21, no. 1, February 2002 (2002-02-01), pages 11 - 8 |
| KRARUP, SEMINARS IN NUCLEAR MEDICINE, vol. 52, no. 6, November 2022 (2022-11-01) |
| NOLAN-STEVAUX ET AL., CANCER DISCOV, October 2023 (2023-10-01) |
| NORTH ET AL.: "A New Clustering of Antibody CDR Loop Conformations", JOURNAL OF MOLECULAR BIOLOGY, vol. 406, 2011, pages 228 - 256, XP028129711, DOI: 10.1016/j.jmb.2010.10.030 |
| O'DONOGHUE ET AL., MOL PHARM., vol. 16, no. 7, 1 July 2019 (2019-07-01), pages 3083 - 3090 |
| RAAVÉ, EUR J NUCL MED MOL IMAGING, vol. 46, 2019, pages 1966 - 1977 |
| SIEGEL ET AL., CA CANCER J CLIN, vol. 73, no. 1, 2023, pages 17 - 48 |
| SUNG ET AL., CA CANCER J CLIN, vol. 71, no. 3, 2021, pages 209 - 249 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| US12404343B2 (en) | Humanised anti kallikrein-2 antibody | |
| US20250345466A1 (en) | Dota-hapten compositions for anti-dota/anti-tumor antigen bispecific antibody pretargeted radioimmunotherapy | |
| CA2853669C (en) | Therapeutic agents and uses thereof | |
| Steis et al. | Toxicity, immunogenicity, and tumor radioimmunodetecting ability of two human monoclonal antibodies in patients with metastatic colorectal carcinoma. | |
| CN111465414B (en) | Melanin antibody and application thereof | |
| CA2665957A1 (en) | Anti-met monoclonal antibody, fragments and derivatives thereof for use in tumor diagnosis, corresponding compositions and kits | |
| WO2025128807A1 (en) | Radiolabeled compounds for the detection of steap1 | |
| US11396538B2 (en) | Antibodies to centrin-1, methods of making, and uses thereof | |
| US20180193455A1 (en) | Antibodies to tumor associated complex n-glycans with terminal glcnacbeta residues and methods of use thereof | |
| CN119499408A (en) | Molecular probes targeting GPC3 and uses thereof | |
| CN117813326A (en) | Radiation-Based Detection, Companion Testing, and Treatment of Directin-1 | |
| US20240417457A1 (en) | Antibodies directed against alpha-folate receptor | |
| Blumenthal et al. | Experimental model systems for antibody targeting and radioimmunodetection | |
| WO2025207909A1 (en) | Radiolabeled glypican-3 (gpc3)-specific single domain antibody and use thereof as an imaging agent for detection of gpc3-positive tumors | |
| TW202517674A (en) | Anti-gpc3 antibodies and radioconjugates thereof | |
| EA044225B1 (en) | APPLICATION OF A CLEANING AGENT BASED ON DENDRONS N-ACETYLGALACTOSAMINE IN PRE-TARGETED RADIOIMMUNOTHERAPY WITH DOTA CHELATOR | |
| BR122020023278B1 (en) | USE OF AN ANTIBODY POLYPEPTIDE FOR THE MANUFACTURE OF A MEDICINE FOR THE TREATMENT AND/OR DIAGNOSIS OF PROSTATE CANCER | |
| BR112018000672B1 (en) | KIT AND USE OF A RADIOACTIVELY LABELED HEAVY CHAIN VARIABLE DOMAIN DERIVED FROM A HEAVY CHAIN ANTIBODY (VHH) OR A FUNCTIONAL FRAGMENT THEREOF IN THE TREATMENT OF CANCER |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24837760 Country of ref document: EP Kind code of ref document: A1 |