WO2025126227A1 - A composition for dyeing /lightening of keratin fibres, and methods thereof - Google Patents
A composition for dyeing /lightening of keratin fibres, and methods thereof Download PDFInfo
- Publication number
- WO2025126227A1 WO2025126227A1 PCT/IN2024/052064 IN2024052064W WO2025126227A1 WO 2025126227 A1 WO2025126227 A1 WO 2025126227A1 IN 2024052064 W IN2024052064 W IN 2024052064W WO 2025126227 A1 WO2025126227 A1 WO 2025126227A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- sucrose
- composition
- weight
- combinations
- fatty
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/60—Sugars; Derivatives thereof
- A61K8/604—Alkylpolyglycosides; Derivatives thereof, e.g. esters
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/36—Carboxylic acids; Salts or anhydrides thereof
- A61K8/361—Carboxylic acids having more than seven carbon atoms in an unbroken chain; Salts or anhydrides thereof
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/37—Esters of carboxylic acids
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/37—Esters of carboxylic acids
- A61K8/375—Esters of carboxylic acids the alcohol moiety containing more than one hydroxy group
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K8/00—Cosmetics or similar toiletry preparations
- A61K8/18—Cosmetics or similar toiletry preparations characterised by the composition
- A61K8/30—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds
- A61K8/33—Cosmetics or similar toiletry preparations characterised by the composition containing organic compounds containing oxygen
- A61K8/39—Derivatives containing from 2 to 10 oxyalkylene groups
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/08—Preparations for bleaching the hair
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61Q—SPECIFIC USE OF COSMETICS OR SIMILAR TOILETRY PREPARATIONS
- A61Q5/00—Preparations for care of the hair
- A61Q5/10—Preparations for permanently dyeing the hair
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/40—Chemical, physico-chemical or functional or structural properties of particular ingredients
- A61K2800/59—Mixtures
- A61K2800/596—Mixtures of surface active compounds
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K2800/00—Properties of cosmetic compositions or active ingredients thereof or formulation aids used therein and process related aspects
- A61K2800/80—Process related aspects concerning the preparation of the cosmetic composition or the storage or application thereof
- A61K2800/88—Two- or multipart kits
- A61K2800/882—Mixing prior to application
Definitions
- the dyeing of keratin fibres, such as human hair, permanently via oxidation dyeing is a known practice that is widely used by consumers.
- This dyeing technique consists in applying to the keratin fibres a composition containing dye precursors such as oxidation bases and couplers. These dye precursors are colourless or weakly coloured compounds. When combined with an oxidizing agent, they give rise, by a process of oxidative condensation, to coloured compounds.
- the term “INCI” is an abbreviation of International Nomenclature of Cosmetic Ingredients, which is a system of names provided by the International Nomenclature Committee of the Personal Care Products Council to describe personal care ingredients.
- a percentage range of about 4% to 20% should be interpreted to include not only the explicitly recited limits of about 4% to about 20%, but also to include sub-ranges, such as 5% to 8%, 12% to 18%, and so forth, as well as individual amounts, including fractional amounts, within the specified ranges, such as 5.5%, and 17.25%, for example.
- Embodiments herein provide a composition wherein the composition is used for dyeing/lightening keratin fibres, such as hair.
- the composition has good dyeing performances such as colour uptake, good colour strength, i.e., intensity of colour, uniform colour, long-lasting colour, good lightening, and bleaching effect, and also good conditioning properties such as good nourishment, soft, moisturized hair, and shiny hair.
- the composition also has good working qualities such as, ease of mixing and rinsing.
- the composition incorporates ingredients that are natural and/or have natural origin and are therefore biodegradable and do not bioaccumulate.
- compositions comprising at least one glycerolated nonionic surfactant; at least one sugar-based nonionic surfactant; at least one fatty acid; and at least one additional fatty compound different from the glycerolated nonionic surfactant(s), sugar-based nonionic surfactant(s), and fatty acid(s) function to provide improved cosmeticity, chromaticity, nourishment, and lightening/bleaching effects, while ensuring the composition is environmentally friendly.
- Embodiments herein also provide a method for dyeing/lightening keratin fibres, preferably hair, by mixing the composition as described herein with a composition (B) comprising at least one oxidizing agent to obtain a mixture; and applying the mixture on said fibres.
- a kit comprising the composition as described herein and the composition (B) and the use of the composition as described herein.
- Embodiments herein provide a composition comprising at least one glycerolated nonionic surfactant; at least one sugar-based nonionic surfactant; at least one fatty acid; and at least one additional fatty compound different from the glycerolated nonionic surfactant(s), sugar-based nonionic surfactant(s), and fatty acid(s).
- composition is an environmentally friendly composition and demonstrates good tinctorial performances such as, intense, uniform, and long-lasting colour, good lightening/bleaching effect, and good conditioning properties while also having good working qualities.
- Glycerolated nonionic surfactant is an environmentally friendly composition and demonstrates good tinctorial performances such as, intense, uniform, and long-lasting colour, good lightening/bleaching effect, and good conditioning properties while also having good working qualities.
- composition according to the invention comprises one or more nonionic surfactants.
- glycerolated nonionic surfactant means a nonionic surfactant comprising at least one mole of glycerol, preferably comprising a number of moles of glycerol ranging from 1 to 50, more preferentially from 1 to 20 and even more preferentially from 2 to 10.
- glycerolated nonionic surfactant means mono glycerolated(one mole of glycerol) or poly glycerolated( several moles of glycerol) nonionic surfactant.
- glycerolated nonionic surfactants examples include:
- - monoesters or polyesters of linear or branched Cs to C40 acids and glycerol or polyglycerol comprising from 1 to 50 mol of glycerol, preferably from 1 to 20 or even from 1 to 10 mol of glycerol; in particular monoesters or diesters of linear or branched Cs to C32, better still Cs to C28 or even Cs to C24 acids, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20 or even from 1 to 10 mol of glycerol;
- the glycerolated nonionic surfactant(s) according to the invention do not comprise any oxyalkylenes units, such as oxy ethylene or oxypropylene units.
- monoesters or diesters of linear or branched, Cs to C40, more preferentially Cs to C32, better still Cs to C28 or even Cs to C24 acids and glycerol or polyglycerol, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20 or even from 1 to 10 mol of glycerol, will be preferred most particularly, alone or as a mixture, and better still, alone or as a mixture:
- Ci6 to C24 acids comprising from 1 to 50 mol of glycerol, preferably from 1 to 20 or even from 2 to 10 mol of glycerol; preferentially diesters of branched Ci6 to C24 acids, comprising from 2 to 10 mol of glycerol, among which mention may be made of isostearic acid diester containing 3 mol of glycerol (INCI name: Polyglyceryl-3 diisostearate);
- Cs to Cis acids comprising from 1 to 50 mol of glycerol, preferably from 1 to 20 or even from 1 to 10 mol of glycerol; preferentially monoesters of linear Cs to Cis acids, comprising from 1 to 10 mol of glycerol, among which mention may be made of lauric acid monoester containing 4 mol of glycerol (INCI name: Polyglyceryl-4 laurate), capric acid monoester comprising 4 mol of glycerol (INCI name: Polyglyceryl-4 caprate) or caprylic acid monoester comprising 1 mol of glycerol (INCI name: Glyceryl caprylate), and stearic acid monoester comprising 1 mol of glycerol (INCI name: Glyceryl stearate).
- lauric acid monoester containing 4 mol of glycerol INCI name: Polyglyceryl-4 laurate
- capric acid monoester comprising 4
- the glycerolated nonionic surfactant(s) is/are selected from at least two different glycerolated nonionic surfactants selected from monoesters or polyesters of linear or branched Cs to C40 acids, preferably Cs to C32 acids, more preferably Cs to C28 acids and glycerol or polyglycerol comprising from 1 to 50 mol of glycerol, preferably from 1 to 20, or even from 1 to 10 mol of glycerol, or combinations thereof.
- the glycerolated nonionic surfactant(s), according to embodiments herein, is(are) selected from monoesters of linear or branched, preferably linear, Cs to C24 acids, more preferably Cs to C20 acids, better from Cs to Cis acids, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20, or more preferably from 1 to 10 mol of glycerol.
- glycerolated nonionic surfactant(s), according to embodiments herein, is(are) selected from a lauric acid monoester containing 4 mol of glycerol, a capric acid monoester comprising 4 mol of glycerol, a caprylic acid monoester comprising 1 mol of glycerol, a stearic acid monoester comprising 1 mol of glycerol or combinations thereof.
- the glycerolated nonionic surfactant(s), according to embodiments herein, is(are) in a total amount ranging from 0.1% to 10% by weight, preferably from 0.2% to 8% by weight, and more preferably from 0.5% to 5% by weight, better still from 1% to 5% by weight, relative to the total weight of the composition.
- the glycerolated nonionic surfactant(s) is(are) selected from monoesters of linear, Cs to C24 acids, more preferably Cs to C20 acids, better from Cs to Cis acids, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20, or more preferably from 1 to 10 mol of glycerol, and is(are) in a total amount ranging from 0.1% to 10% by weight, preferably from 0.2% to 8% by weight, and more preferably from 0.5% to 5% by weight, better still from 1% to 5% by weight, relative to the total weight of the composition.
- the glycerolated nonionic surfactant(s) is(are) selected from lauric acid monoester containing 4 mol of glycerol (INCI name: Polyglyceryl- 4 laurate), capric acid monoester comprising 4 mol of glycerol (INCI name: Polyglyceryl-4 caprate), or caprylic acid monoester comprising 1 mol of glycerol (INCI name: Glyceryl caprylate), stearic acid monoester comprising 1 mol of glycerol (INCI name: Glyceryl stearate) or combinations thereof, and is(are) in a total amount ranging from 0.1% to 10% by weight, preferably from 0.2% to 8% by weight, and more preferably from 0.5% to 5% by weight, better still from 1% to 5% by weight, relative to the total weight of the composition.
- the composition according to the invention comprises one or more sugar-based nonionic surfactants.
- sugar-based nonionic surfactant means a nonionic surfactant having a monosaccharide portion (i.e., monosaccharide or oside or simple sugar) or an oligosaccharide portion (short chains formed from the sequence of monosaccharide units, possibly different) or a polysaccharide portion [long chains made up of monosaccharide units, possibly different, i.e., polyholosides or polysosides (homopolysaccharides or heteropolysaccharides)] .
- the sugar-based nonionic surfactant(s) according to the invention do not comprise any oxyalkylene units, such as oxy ethylene or oxypropylene units.
- the sugar-based nonionic surfactant(s) is(are) preferably selected from alkylpolyglucosides, sucrose fatty esters, sorbitan fatty esters, or combinations thereof.
- alkyl(poly)glucoside(s), is(are) represented especially by the following general Formula I:
- Ri represents a linear or branched alkyl or alkenyl radical comprising 6 to 24 carbon atoms and especially 8 to 18 carbon atoms, or an alkylphenyl radical whose linear or branched alkyl radical comprises 6 to 24 carbon atoms and especially 8 to 18 carbon atoms;
- R 2 represents an alkylene radical comprising 2 to 4 carbon atoms
- R 3 represents a sugar unit comprising 5 to 6 carbon atoms
- m denotes a value ranging from 0 to 10 and preferably 0 to 4
- n denotes a value ranging from 1 to 15 and preferably 1 to 4.
- the alkylpoly gluco side surfactants are compounds of the formula described above in which:
- Ri denotes a linear or branched, saturated or unsaturated alkyl radical comprising from 8 to 18 carbon atoms
- R2 represents an alkylene radical comprising 2 to 4 carbon atoms
- m denotes a value ranging from 0 to 3 and preferably equal to 0,
- R3 denotes glucose, fructose or galactose, preferably glucose; the degree of polymerization, i.e. the value of n, possibly ranging from 1 to 15 or from 1 to 4; the mean degree of polymerization more particularly being between 1 and 2.
- Cs/Ci6- alkyl(poly)glucosides 1,4 especially as an aqueous 53% solution, such as those sold by COGNIS under the reference Plantacare® 818 UP.
- the alkylpolyglucoside is selected from decyl glucoside, stearyl glucoside, lauryl glucoside, coco-glucoside, cetearyl glucoside, decyl lauryl glucoside, or combinations thereof.
- the alkylpolyglucoside is selected from decyl glucoside, stearyl glucoside, lauryl glucoside, coco-glucoside, cetearyl glucoside, or combinations thereof.
- An exemplary monoester has the following structure as shown in Formula III: where R is an alkyl group having 3-27 carbons, and typically 7-27 carbons.
- the sucrose fatty esters include blends of sucrose fatty esters, which typically include monoesters, and can also include diesters, triesters and polyesters, which have structures according to Formula (III), above, where two (diesters), three (triesters) or more (polyesters) of X 1 , X 2 , X 3 , X 4 , X 5 , X 6 , X 7 and X 8 , (and typically X 1 and X 8 ) independently, are
- sucrose fatty esters include sucrose fatty ester blends, for example, sucrose ester mixtures containing a specified amount (e.g., percent, by weight) of sucrose monoesters.
- sucrose fatty esters are selected from sucrose fatty monoesters, such as sucrose monocaprylate, sucrose monodecanoate, sucrose monolaurate, sucrose monomyristate, sucrose monopalmitate, sucrose monostearate, sucrose monopelargonate, sucrose monoundecanoate, sucrose monotridecanoate, sucrose monopentadecanoate, and sucrose monoheptadec anoate .
- sucrose monocaprylate sucrose monodecanoate
- sucrose monolaurate sucrose monomyristate
- sucrose monopalmitate sucrose monostearate
- sucrose monopelargonate sucrose monoundecanoate
- sucrose monotridecanoate sucrose monopentadecanoate
- sucrose monopentadecanoate sucrose monopentadecanoate
- sucrose monoheptadec anoate sucrose monoesters
- sucrose fatty esters examples include sucrose cocoate sucrose laurate (and) aqua (and) alcohol (Surfhope(R) C-1215 commercially available from Mitsubishi-Kagaku), sucrose laurate, sucrose myristate, sucrose palmitate, sucrose polystearate, sucrose tristearate, sucrose distearate, sucrose stearate, sucrose dilaurate, sucrose hexaerucate, sucrose oleate, sucrose pentaerucate, sucrose polybehenate, sucrose polycottonseedate, sucrose polylaurate, sucrose polylinoleate, sucrose polypalmate, sucrose polyoleate, sucrose polysoyate, sucrose ricinoleate, sucrose tetraisostearate, sucrose tribehenate, sucrose hexaoleate/hexapalmitate/hexastearate, sucrose hexapalmitate, sucrose trilaurate, or combinations
- the sucrose fatty esters include sucrose oleate, sucrose laurate (and) aqua (and) alcohol, sucrose laurate, sucrose myristate, sucrose palmitate, sucrose polystearate, sucrose tristearate, sucrose distearate, sucrose stearate, and mixtures thereof, all of which are commercially available from Mitsubishi-Kagaku under the tradename Surfhope(R) C.
- the sucrose fatty ester may be used, for example, as a mixture with other ingredient, for example alcohol, such as the products sold, for example, by Mitsubishi-Kagaku under the trade name Surfhope(R) C.
- the sucrose fatty ester may also be used without additives, for example, such as the product Ryoto Sugar Ester S 370 (Ryoto).
- the sucrose fatty ester(s) is(are) selected from sucrose cocoate, sucrose laurate, sucrose myristate, sucrose palmitate, sucrose polystearate, sucrose tristearate, sucrose distearate, sucrose stearate, sucrose dilaurate, sucrose hexaerucate, sucrose oleate, sucrose pentaerucate, sucrose polybehenate, sucrose polycottonseedate, sucrose polylaurate, sucrose polylinoleate, sucrose polypalmate, sucrose polyoleate, sucrose polysoyate, sucrose ricinoleate, sucrose tetraisostearate, sucrose tribehenate, sucrose hexaoleate, sucrose hexastearate, sucrose hexapalmitate, sucrose trilaurate, or combinations thereof.
- sucrose fatty ester(s) is(are) selected from sucrose oleate, sucrose stearate, sucrose distearate, sucrose tristearate, or combinations thereof.
- the sorbitan esters may be selected from the group consisting of sorbitan monostearate, sorbitan tristearate, sorbitan monolaurate, sorbitan monooleate, sorbitan palmitate, or combinations thereof.
- the sugar-based nonionic surfactant(s), according to embodiments herein, is(are) selected from decyl glucoside, stearyl glucoside, lauryl glucoside, coco-glucoside, cetearyl glucoside, sucrose oleate, sucrose stearate, sucrose distearate, sucrose tristearate, or combinations thereof.
- the sugar-based nonionic surfactant(s), according to embodiments herein, is(are) selected from coco-glucoside, sucrose oleate, sucrose stearate, sucrose distearate, sucrose tristearate, or combinations thereof.
- the sugar-based nonionic surfactant(s), according to embodiments herein, is(are) in a total amount ranging from 0.1% to 20% by weight, preferably from 0.5% to 10% by weight, and more preferably from 1% to 5% by weight, relative to the total weight of the composition.
- the sugar-based nonionic surfactant(s) is(are) selected from decyl glucoside, stearyl glucoside, lauryl glucoside, coco-glucoside, cetearyl glucoside, sucrose oleate, sucrose stearate, sucrose distearate, sucrose tristearate, or combinations thereof, and is(are) in a total amount ranging from 0.1% to 20% by weight, preferably from 0.5% to 10% by weight, and more preferably from 1% to 5% by weight, relative to the total weight of the composition.
- composition according to the invention comprises one or more fatty acids.
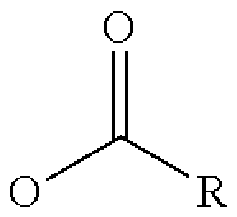
- fatty acid means a long-chain carboxylic acid comprising at least 6 carbon atoms, in particular from 6 to 40 carbon atoms, preferably from 8 to 30 carbon atoms.
- the fatty acids according to the invention preferentially comprise from 10 to 30 carbon atoms and better still from 14 to 22 carbon atoms. They may optionally be hydroxylated.
- the fatty acid of the invention may be present as the free form or as the neutralized, partly or totally, form.
- the fatty acids present in the composition according to the invention include at least one carboxylic acid group and a linear or branched, saturated or unsaturated, in particular unsaturated, alkyl chain comprising from 6 to 40 carbon atoms, preferably from 8 to 30 carbon atoms, more preferably from 10 to 30 carbon atoms and better still from 14 to 22 carbon atoms.
- the fatty acids include at least one carboxylic acid group and a linear or branched, saturated or unsaturated, in particular unsaturated, alkyl chain comprising from 10 to 30 carbon atoms, in particular from 12 to 22 carbon atoms.
- the fatty acids include at least one carboxylic acid group and a linear or branched, unsaturated alkyl chain comprising from 14 to 22 carbon atoms.
- the fatty acids are selected from compounds having the structure Rt-C(O)OH in which R t represents a linear or branched, saturated or unsaturated alkyl group comprising from 5 to 39 carbon atoms, preferably from 7 to 29 carbon atoms, preferentially from 11 to 23 carbon atoms, better still from 13 to 19 carbon atoms.
- the fatty acids present in the composition are neither oxyalkylenated nor glycerolated. Further, they may be present as free form or as the neutralized, partly, or totally, form.
- the fatty acids may be selected from solid fatty acids, liquid fatty acids, or combinations thereof.
- solid fatty acid means a fatty acid with a melting point of greater than 25 °C, preferably greater than or equal to 28°C, more preferentially greater than or equal to 30°C at atmospheric pressure (1.013xl0 5 Pa).
- the solid fatty acids are selected from myristic acid, palmitic acid, arachidic acid, stearic acid, lauric acid, behenic acid, 12-hydroxystearic acid, or combinations thereof.
- the solid fatty acid(s) are selected from lauric acid, myristic acid, palmitic acid, stearic acid, or combinations thereof, more preferably from myristic acid, palmitic acid, stearic acid, or combinations thereof.
- liquid fatty acid means a fatty acid with a melting point of less than or equal to 25°C, preferably less than or equal to 20°C at atmospheric pressure (1.013xl0 5 Pa).
- the liquid fatty acid(s) according to the invention may be selected from oleic acid, linoleic acid, arachidonic acid, isostearic acid, isopalmitic acid, or combinations thereof, more preferably is oleic acid.
- the fatty acid(s), according to embodiments herein, is(are) solid fatty acids, in particular, selected from fatty acids including at least one carboxylic acid group and a linear or branched, unsaturated alkyl chain comprising from 6 to 40 carbon atoms, in particular from 10 to 30 carbon atoms.
- the fatty acid(s) is(are) selected from at least one carboxylic acid comprising 6 to 40 carbon atoms, and preferably from the carboxylic acid comprising 10 to 30 carbon atoms, more preferably from lauric acid, myristic acid, stearic acid, palmitic acid, or combinations thereof.
- the fatty acid(s) is(are) selected from myristic acid, stearic acid, palmitic acid, or combinations thereof.
- the fatty acid(s), according to embodiments herein, is(are) in a total amount ranging from 0.1% to 15% by weight, preferably from 0.2% to 10% by weight, more preferably from 0.4% to 5% by weight, and better from 0.5% to 3% by weight, relative to the total weight of the composition.
- the fatty acid(s) is(are) solid fatty acids, in particular, selected from fatty acids including at least one carboxylic acid comprising 6 to 40 carbon atoms, and preferably from the carboxylic acid comprising 10 to 30 carbon atoms, and is(are) in a total amount ranging from 0.1% to 15% by weight, preferably from 0.2% to 10% by weight, more preferably from 0.4% to 5% by weight, and better from 0.5% to 3% by weight, relative to the total weight of the composition.
- the fatty acid(s) is(are) selected from lauric acid, myristic acid, stearic acid, palmitic acid, or combinations thereof, and is(are) in a total amount ranging from 0.1% to 15% by weight, preferably from 0.2% to 10% by weight, more preferably from 0.4% to 5% by weight, and better from 0.5% to 3% by weight, relative to the total weight of the composition.
- the fatty compounds that are useful may be liquid fatty compounds (or oils) and/or solid fatty compounds.
- liquid fatty compound means a fatty compound with a melting point of less than or equal to 25°C at atmospheric pressure (1.013xl0 5 Pa) and the term “solid fatty compound” means a fatty compound with a melting point of greater than 25 °C at atmospheric pressure (1.013xl0 5 Pa).
- the fatty alcohols and esters contain at least one saturated or unsaturated, linear or branched hydrocarbon-based group, comprising 6 to 40 and better still from 8 to 30 carbon atoms, which is optionally substituted, in particular, with one or more hydroxyl groups (in particular 1 to 4). If they are unsaturated, these compounds may comprise one to three conjugated or unconjugated carbon-carbon double bonds.
- the Ce to Ci6 liquid hydrocarbons may be linear, branched, or optionally cyclic, and are preferably selected from alkanes. Examples that may be mentioned include hexane, cyclohexane, undecane, dodecane, isododecane, tridecane or isoparaffins, such as isohexadecane or isodecane, or combinations thereof.
- the fluoro oils may be selected from perfluoromethylcyclopentane and perfluoro- 1,3 -dimethylcyclohexane, sold under the names Flutec® PCI and Flutec® PC3 by the company BNFL Fluorochemicals; perfluoro- 1,2- dimethylcyclobutane; perfluoroalkanes such as dodecafluoropentane and tetradecafluorohexane, sold under the names PF 5050® and PF 5060® by the company 3M, or bromoperfluorooctyl sold under the name Foralkyl® by the company Atochem; nonafluoromethoxybutane and nonafluoroethoxyisobutane; perfluoromorpholine derivatives such as 4-trifluoromethylperfluoromorpholine sold under the name PF 5052® by the company 3M.
- the esters of monoalcohols Preferably, for the esters of monoalcohols, at least one from among the alcohol and the acid is branched.
- the monoesters are selected from dihydroabietyl behenate; isostearyl lactate; lauryl lactate; linoleyl lactate; oleyl lactate; isostearyl octanoate; isocetyl octanoate; isocetyl isostearate; isocetyl laurate; isocetyl stearate; isodecyl octanoate; isodecyl oleate; isononyl isononanoate; isostearyl palmitate; methyl acetyl ricinoleate; octyl isononanoate; 2- ethylhexyl isononate; octyldodecyl erucate; oleyl
- the liquid esters of fatty acids and/or of fatty alcohols is(are) selected from diethyl sebacate; diisopropyl sebacate; diisopropyl adipate; di-n-propyl adipate; dioctyl adipate; diisostearyl adipate; dioctyl maleate; glyceryl undecylenate; octyldodecyl stearoyl stearate; pentaerythrityl monoricinoleate; pentaerythrityl tetraisononanoate; pentaerythrityl tetrapelargonate; pentaerythrityl tetraisostearate; pentaerythrityl tetraoctanoate; propylene glycol; dicaprylate; propylene glycol dicaprate; tridecyl eru
- a liquid ester of a monoacid and of a monoalcohol is preferred.
- the fatty compounds are selected from liquid fatty compounds, preferably from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, liquid fatty alcohols and liquid fatty esters, or combinations thereof, more preferably from plant oils.
- the solid fatty alcohols that may be used are preferably selected from saturated or unsaturated, linear or branched, preferably linear and saturated, (mono)alcohols including from 6 to 40 carbon atoms, better still from 10 to 30, or even from 12 to 24 and even better still from 14 to 22 carbon atoms.
- the waxes that are suitable for use in the invention may be selected from waxes of animal, plant or mineral origin, non-silicone synthetic waxes, or combinations thereof.
- the waxes may be selected from hydrocarbon-based waxes, for instance beeswax, notably of organic origin, lanolin wax and Chinese insect waxes; rice bran wax, carnauba wax, candelilla wax, ouricury wax, esparto grass wax, berry wax, shellac wax, Japan wax and sumac wax; montan wax, orange wax and lemon wax, microcrystalline waxes, paraffins and ozokerite; polyethylene waxes, the waxes obtained by Fischer-Tropsch synthesis and waxy copolymers, and also esters thereof.
- R denotes a hydrogen atom, a (glycosyl)n group, a (galactosyl)m group or a sulfogalactosyl group, in which n is an integer ranging from 1 to 4 and m is an integer ranging from 1 to 8;
- R’ denotes a C15-C26 hydrocarbon-based group, saturated or unsaturated in the alpha position, this group possibly being substituted with one or more C1-C14 alkyl groups; it being understood that in the case of natural ceramides or glycoceramides, R’” may also denote a C15-C26 alpha-hydroxyalkyl group, the hydroxyl group being optionally esterified with a C16-C30 alpha-hydroxy acid.
- Butter may also be used.
- the particular butter(s) are of plant origin, such as those described in Ullmann’s Encyclopedia of Industrial Chemistry (“Fats and Fatty Oils”, A. Thomas, published online: 15 JUN 2000, DOI: 10.1002/14356007. al0_173, point 13.2.2.2. Shea Butter, Borneo Tallow, and Related Fats (Vegetable Butters)).
- the additional fatty compound(s), according to embodiments herein, is(are) selected from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, liquid fatty esters, solid fatty alcohols, solid fatty esters or combinations thereof, more preferably from plant oils, solid fatty alcohols, solid fatty esters or combinations thereof.
- the additional fatty compound(s), is(are) selected from liquid fatty compound, solid fatty compounds or combinations thereof, preferably from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, liquid fatty alcohols, liquid fatty esters, silicone oils, solid fatty alcohols, solid fatty esters, waxes, ceramides, or combinations thereof, and is(are) in a total amount ranging from 5% to 45% by weight, preferably 10% to 40% by weight, and more preferably 15% to 35% by weight, better still from 20% to 32% by weight, relative to the total weight of the composition.
- the additional fatty compound(s), is(are) selected from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, liquid fatty esters, solid fatty alcohols, solid fatty esters or combinations thereof, and is(are) in a total amount ranging from 5% to 45% by weight, preferably 10% to 40% by weight, and more preferably 15 to 35% by weight, better still from 20% to 32% by weight, relative to the total weight of the composition.
- the composition, according to the present invention comprises at least one alkaline agent.
- the alkaline agent(s) that may be used is(are) selected from at least one mineral, organic, or hybrid alkaline agent or combinations thereof.
- the mineral alkaline agents are selected from ammonium hydroxide, alkali metal carbonates or bicarbonates such as sodium (hydrogen)carbonate and potassium (hydrogen)carbonate, alkali metal or alkaline-earth metal silicates or metasilicates such as sodium metasilicate, or combinations thereof.
- the organic alkaline agents are selected from alkanolamines, amino acids, organic amines other than alkanolamines, oxyethylenated and/or oxypropylenated ethylenediamines, 1,3 -diaminopropane, spermine, spermidine, or combinations thereof.
- alkanolamine means an organic amine comprising a primary, secondary, or tertiary amine function, and one or more linear or branched Ci-Cs alkyl groups bearing one or more hydroxyl radicals.
- organic amines selected from alkanolamines such as, monoalkanolamines, dialkanolamines or trialkanolamines comprising one to three identical or different Ci-Cio hydroxy alkyl radicals are in particular suitable for performing the present invention.
- the alkanolamine(s) are selected from monoethanolamine (MEA), diethanolamine, triethanolamine, monoisopropanolamine, diisopropanolamine, N,N-dimethylethanolamine, 2- amino-2-methyl- 1 -propanol, triisopropanolamine, 2-amino-2-methy 1-1,3- propanediol, 3-amino- 1 ,2-propanediol, 3 -dimethylamino- 1 ,2-propanediol, tris(hydroxymethyl)aminomethane, or combinations thereof.
- MEA monoethanolamine
- diethanolamine triethanolamine
- monoisopropanolamine diisopropanolamine
- N,N-dimethylethanolamine 2- amino-2-methyl- 1 -propanol
- 2- amino-2-methyl- 1 -propanol triisopropanolamine
- 2-amino-2-methy 1-1,3- propanediol
- the alkaline agent(s) is(are) selected from alkanolamines and ammonium hydroxide. More preferably, the alkaline agent is selected from alkanolamines, better is, monoethanolamine.
- the total amount of the alkaline agent(s), according to embodiments herein, is(are) in a total amount ranging from 0.1% to 40% by weight, preferably 1% to 30% by weight, more preferably from 2% to 25% by weight, and better from 4% to 20% by weight, relative to the total weight of the composition.
- the alkaline agent(s), according to embodiments herein, is(are) selected from alkanolamines, ammonium hydroxide, or their combinations, and is(are) in a total amount ranging from 0.1% to 40% by weight, preferably 1% to 30% by weight, more preferably from 2% to 25% by weight, and better from 4% to 20% by weight, relative to the total weight of the composition.
- the alkaline agent(s) selected from alkanolamines is(are) in a total amount ranging from 0.1% to 40% by weight, preferably 1% to 30% by weight, more preferably from 2% to 25% by weight, and better from 4% to 20% by weight, relative to the total weight of the composition.
- composition may comprise one or more oxidative dyes.
- the composition according to the invention comprises one or more oxidative dyes.
- the oxidative dye includes at least one oxidation base and/or at least one oxidation coupler.
- the composition according to the present invention comprises one or more oxidation bases in combination with one or more couplers.
- Oxidation bases are generally colourless or weakly coloured compounds which when mixed with oxidizing compounds give rise to coloured compounds or dye. Further, the colour shades obtained with such oxidation bases can be varied by combining them with oxidation couplers or colour modifiers.
- the oxidation base(s), according to the present invention, that may be used, is(are) selected from a group consisting of para-phenylenediamines, bis(phenyl)alkylenediamines, para-aminophenols, ortho-aminophenols, heterocyclic bases, and their addition salts thereof.
- the addition salts of the oxidation bases are preferably selected from the addition salts with an acid, such as, the hydrochlorides, hydrobromides, sulfates, citrates, succinates, tartrates, lactates, tosylates, benzenesulfonates, methanesulfonates, phosphates and acetates, and the addition salts with a base such as, sodium hydroxide, potassium hydroxide, aqueous ammonia, amines, or alkanolamines.
- an acid such as, the hydrochlorides, hydrobromides, sulfates, citrates, succinates, tartrates, lactates, tosylates, benzenesulfonates, methanesulfonates, phosphates and acetates
- a base such as, sodium hydroxide, potassium hydroxide, aqueous ammonia, amines, or alkanolamines.
- the para-phenylenediamines that may be used are selected from para-phenylenediamine, para-tolylenediamine, 2-chloro-para-phenylenediamine, 2,3-dimethyl-para-phenylenediamine, 2,6-dimethyl-para-phenylenediamine, 2,6- diethyl-para-phenylenediamine, 2,5 -dimethyl-para-phenylenediamine, N,N - dimethyl-para-phenylenediamine, N,N -diethyl-para-phenylenediamine, N,N- dipropyl-para-phenylenediamine, 4-amino-N,N-diethyl-3-methylaniline, N,N- bis(P-hydroxyethyl)-para-phenylenediamine, 4-N,N-bis(0-hydroxyethyl)amino-2- methylaniline, 4-N,N-bis(0-hydroxyethyl)a
- the para-phenylenediamines are selected from para-phenylenediamine, para-tolylenediamine, 2-isopropyl-para- phenylenediamine, 2- 0- hydroxycthy I -para-phcny Icncdi am i nc, 2-y-hydroxyethyl- para-phenylenediamine, 2-methoxymethyl -para-phenylenediamine, 2-0- hydroxyethyloxy -para-phenylenediamine, 2,6-dimethyl-para-phenylenediamine, 2,6-diethyl-para-phenylenediamine, 2,3-dimethyl-para-phenylenediamine, N,N- bis(0-hydroxyethyl)-para-phenylenediamine, 2-chloro-para-phenylenediamine, 2- 0-acetylaminoethyloxy-para-phenylenediamine, or,
- the bis(phenyl)alkylenediamines that may be used are selected from N,N’-bis(0-hydroxyethyl)-N,N’-bis(4’ -aminophenyl)- 1,3 -diaminopropanol, N,N’- bis-(0-hydroxyethyl)-N,N’-bis(4’-aminophenyl)ethylenediamine, N,N’-bis(4- aminophenyl)tetramethylenediamine, N,N’-bis(0-hydroxyethyl)-N,N’-bis(4- aminophenyl)tetramethylenediamine, N,N’ -bis(4- methylaminophenyl)tetramethylenediamine, N,N’-bis(ethyl)-N,N’-bis(4 ’-aminos’ -methylphenyl)ethylenediamine, l,8-bis(2,5-diaminophenoxy)-3
- the para-aminophenols may be selected from, para-aminophenol, 4- amino-3-methylphenol, 4-amino-3-fluorophenol, 4-amino-3-chlorophenol, 4- amino-3-hydroxymethylphenol, 4-amino-2-methylphenol, 4-amino-2- hydroxymethylphenol, 4-amino-2-methoxymethylphenol, 4-amino-2- aminomethylphenol, 4-amino-2-(P-hydroxyethylaminomethyl)phenol and 4- amino-2-fluorophenol, or the addition salts thereof with an acid.
- the addition salt is salt of sulphurous acid or sodium metabisulphite salt.
- the ortho-aminophenols that may be used are selected from, for example, 2-aminophenol, 2-amino-5-methylphenol, 2-amino-6-methylphenol and 5-acetamido-2-aminophenol, or the addition salts thereof.
- heterocyclic bases that may be used are selected from pyridine derivatives, pyrimidine derivatives and pyrazole derivatives.
- pyridine derivatives may be selected from the compounds as described, for example, in patents GB 1 026 978 and/or GB 1 153 196, including 2,5-diaminopyridine, 2-(4-methoxyphenyl)amino-3- aminopyridine, 3,4-diaminopyridine, or the addition salts thereof.
- the pyridine derivatives may further be selected from 3-aminopyrazolo[l,5-a]pyridine oxidation bases, or addition salts thereof described, for example, in patent application FR 2 801 308.
- bases include pyrazolo[l,5-a]pyrid-3-ylamine, 2-acetylaminopyrazolo[l,5- a]pyrid-3 -ylamine, 2-morpholin-4-ylpyrazolo [ 1 ,5-a]pyrid-3 -ylamine, 3 - aminopyrazolo[ 1 ,5-a]pyridine-2-carboxylic acid, 2-methoxypyrazolo[l ,5-a]pyrid- 3-ylamine, (3-aminopyrazolo[l,5-a]pyrid-7-yl)methanol, 2-(3-aminopyrazolo[l,5- a]pyrid-5-yl)ethanol, 2-(
- the pyrimidine derivatives may be selected from the compounds described, for example, in the patents DE 2359399; JP 88- 169571; JP 05-63124; EP 0770375 or patent application WO 96/15765, including
- the pyrazole derivatives may be selected from, for example, the compounds described in the patents DE 3843892, DE 4133957 or patent applications WO 94/08969, WO 94/08970, FR-A-2 733 749 or DE 195 43 988, including for example 4,5-diamino-l-methylpyrazole, 4,5- diamino- l-(
- the pyrazole derivatives that may be used also include diamino-N,N-dihydropyrazolo pyrazolones and especially those described in patent application FRA 2886136, such as the following compounds and the addition salts thereof: 2,3-diamino-6,7-dihydro-lH,5H-pyrazolo[l,2- a]pyrazol-l-one, 2-amino-3-ethylamino-6,7-dihydro-lH,5H-pyrazolo[l,2- a]pyrazol-l-one, 2-amino-3-isopropylamino-6,7-dihydro-lH,5H-pyrazolo[l,2- a]pyrazol-l-one, 2-amino-3-(pyrrolidin-l-yl)-6,7-dihydro-lH,5H-pyrazolo[l,2- a]pyrazol-l-l
- the heterocyclic base may be selected from 4,5-diamino- l-(
- the oxidation bases are selected from paraphenylenediamines, bis(phenyl)alkylenediamines, para-aminophenols, orthoaminophenols, heterocyclic bases, the corresponding addition salts, or combinations thereof, preferably from 2-methoxymethyl-para-phenylenediamine, 2-P-hydroxyethyl-para-phenylenediamine, 2-y-hydroxypropyl-para- phenylenediamine, their addition salts thereof, or combinations thereof.
- the oxidation coupler(s), according to the present invention, that may be used, is(are) selected from a group consisting of meta-phenylenediamines, meta- aminophenols, meta-diphenols, naphthalene-based couplers, heterocyclic couplers, and their addition salts.
- the oxidation couplers may be selected from 1,3-dihydroxybenzene, l,3-dihydroxy-2-methylbenzene, 4-chloro-l,3- dihydroxybenzene, 1 -hydroxy-3 -aminobenzene, 1 -methyl-2-hydroxy-4-P- hydroxy ethylaminobenzene, 4-amino-2-hydroxytoluene, 5-amino-6-chl oro-2 - m ethylphenol, 2, 4-diamino-l-(P-hydroxy ethyloxy )benzene, a-naphthol, 6- hydroxyindole, 2-amino-3 -hydroxypyridine, 6-hydroxybenzomorpholine, 3- amino-6-methoxy-2-methylaminopyridine, 2-amino-4-hydroxy ethylaminoanisole, hydroxyethyl-3,4-methylenedioxyaniline, 2-amino-5-ethy
- the oxidation couplers are further selected from 3 -amino-6-m ethoxy -2-methylaminopyri dine, 6-hydroxybenzomorpholine,
- the addition salts of the oxidation couplers are selected from addition salts with an acid, such as, hydrochlorides, hydrobromides, sulfates, citrates, succinates, tartrates, lactates, tosylates, benzenesulfonates, phosphates and acetates, and the addition salts with a base such as, sodium hydroxide, potassium hydroxide, aqueous ammonia, amines or alkanolamines.
- an acid such as, hydrochlorides, hydrobromides, sulfates, citrates, succinates, tartrates, lactates, tosylates, benzenesulfonates, phosphates and acetates
- a base such as, sodium hydroxide, potassium hydroxide, aqueous ammonia, amines or alkanolamines.
- the oxidation couplers are selected from 6- hydroxybenzomorpholine, hydroxy ethyl-3,4-methylenedi oxyaniline, 2-amino-5- ethylphenol, their addition salts, or combinations thereof.
- composition according to embodiments herein is, in some embodiments, free of oxidation couplers chosen from resorcinol, 2- methylresorcinol, 4-chlororesorcinol, or the addition salts thereof.
- oxidation base(s), according to embodiments herein, is(are) in a total amount ranging from 0.0001% to 10% by weight, relative to the total weight of the composition.
- the oxidation coupler(s), according to embodiments herein, is(are) in a total amount ranging from 0.0001% to 10% by weight, relative to the total weight of the composition.
- Examples of cellulose derivatives comprising quaternary ammonium groups are especially described in French patent 1 492597, and mention may be made of the polymers sold under the name UCARE POLYMER “JR” (JR 400 LT, JR 125 and JR 30M) or “LR” (LR 400 or LR 30M) by the company Dow chemical. These polymers are also defined in the CTFA dictionary as quaternary ammoniums of hydroxy ethyl cellulose that have reacted with an epoxide substituted with a trimethylammonium group. The polyquaternium-10 is, for example, one of these polymers.
- the cationic polysaccharide(s), according to embodiments herein, is(are) in a total amount ranging from 0.01% to 8% by weight, preferably from 0.05% to 5% by weight, more preferably from 0.1% to 4%, and better from 0.2% to 3% by weight, relative to the total weight of the composition.
- the cationic polysaccharide(s) is(are) selected from guar gums comprising cationic trialkylammonium groups and is(are) in a total amount ranging from 0.01% to 8% by weight, preferably from 0.05% to 5% by weight, more preferably from 0.1% to 4%, and better from 0.2% to 3% by weight, relative to the total weight of the composition.
- composition may comprise one or more chelating agents.
- the composition comprises one or more chelating agents.
- Rb represents a CH2COOH group when R a represents a hydrogen atom, or Rb represents a hydrogen atom when R a is other than a hydrogen atom;
- R c represents a linear or branched alkyl group comprising from 1 to 14 carbon atoms, preferably 1 to 4 carbon atoms, or cyclic alkyl group comprising from 3 to 30 carbon atoms.
- the carboxylic acids of formula (V) correspond to:
- the carboxylic acids of formula (V) correspond to compounds comprising four carboxylic acid functions when R a represents the -CH(COOH)-(CH2)2-COOH group and Rb represents a hydrogen atom.
- the carboxylic acids of formula (V), according to the present invention may take the form of pure enantiomers, preferably in L configuration, or the form of mixtures, or the form of racemic mixtures.
- the salts of the one or more carboxylic acids of formula (V) are selected from alkali metal salts, alkaline earth metal salts, transition metal salts, organic amine salts, ammonium salts, or combinations thereof.
- alkali metal salts include especially sodium (Na + ) and potassium (K + ) salts
- alkaline earth metal salts include especially calcium (Ca 2+ ) and magnesium (Mg 2+ ) salts.
- a “transition metal” is a metal comprising an incomplete d subshell, and more particularly in the II oxidation state, such as cobalt (Co 2+ ), iron (Fe 2+ ), manganese (Mn 2+ ), zinc (Zn 2+ ) and copper (Cu 2+ ).
- the organic amine salts may be made of the salts of primary, secondary, or tertiary amines, or alternatively of alkanolamines.
- the amines exhibit one or more identical or non-identical radicals, of linear or branched Ci to C20 alkyl type, optionally comprising a heteroatom such as oxygen.
- the carboxylic acids of formula (V), according to embodiments herein, are selected from methylglycinediacetic acid, N- lauroylethylenediamine-N,N',N'-triacetic acid, N,N-dicarboxymethylglutamic acid, iminodisuccinic acid, N,N-bis(carboxymethyl)aspartic acid, alkali metal salts thereof, alkaline earth metal salts thereof, transition metal salts thereof, organic amine salts thereof, ammonium salts thereof, optical isomers thereof, geometric isomers thereof, solvates thereof, or combinations thereof.
- the chelating agents are selected from N,N-dicarboxymethylglutamic acid, N,N-bis(carboxymethyl)aspartic acid, alkali metal salts thereof, alkaline earth metal salts thereof, transition metal salts thereof, organic amine salts thereof, ammonium salts thereof, optical isomers thereof, geometric isomers thereof, solvates thereof, or combinations thereof.
- the chelating agents are selected from N,N-dicarboxymethyl-L-glutamic acid (GLDA), N,N-bis(carboxymethyl)-L- aspartic acid, alkali metal salts thereof, alkaline earth metal salts thereof, transition metal salts thereof, organic amine salts thereof, ammonium salts thereof, solvates thereof, or combinations thereof, and more preferably is N,N-dicarboxymethyl-L- glutamic acid (GLDA), and/or tetrasodium N,N-bis(carboxymethyl)-L-glutamate.
- GLDA N,N-dicarboxymethyl-L-glutamic acid
- GLDA N,N-bis(carboxymethyl)-L-glutamic acid
- N,N-dicarboxymethyl-L-glutamic acid, tetrasodium N,N- bis(carboxymethyl)-L-glutamate and N,N-bis(carboxymethyl)aspartic acid are respectively represented by compounds (II), (III) and (IV) below:
- the chelating agent(s), according to embodiments herein, is (are) in a total amount ranging from 0.01% to 5% by weight, preferably in an amount ranging from 0.05% to 3% by weight, and more preferably from 0.1% to 2% by weight, relative to the total weight of the composition.
- the chelating agents are selected from N,N- dicarboxymethyl-L-glutamic acid (GLDA), N,N-bis(carboxymethyl)-L-aspartic acid, alkali metal salts thereof, alkaline earth metal salts thereof, transition metal salts thereof, organic amine salts thereof, ammonium salts thereof, solvates thereof, or combinations thereof, and is (are) in a total amount ranging from 0.01% to 5% by weight, preferably in an amount ranging from 0.05% to 3% by weight, and more preferably from 0.1% to 2% by weight, relative to the total weight of the composition.
- GLDA N,N- dicarboxymethyl-L-glutamic acid
- N,N-bis(carboxymethyl)-L-aspartic acid alkali metal salts thereof, alkaline earth metal salts thereof, transition metal salts thereof, organic amine salts thereof, ammonium salts thereof, solvates thereof, or combinations thereof
- composition according to the invention comprises one or more polyols.
- polyol refers to an organic compound constituted of a hydrocarbon-based chain optionally interrupted with one or more oxygen atoms and bearing at least two free hydroxyl groups (-OH) borne by different carbon atoms, this compound possibly being cyclic or acyclic, linear or branched, and saturated or unsaturated.
- the polyols comprise from 2 to 30 hydroxyl groups, preferably from 2 to 10 hydroxyl groups, more preferably from 2 to 3 hydroxyl groups, and even more preferably comprises 2 hydroxyl groups.
- the polyols are selected from diols, preferably from C3-C6 diols.
- the polyols are selected from propylene glycol, propane- 1,3-diol, 1,3-butylene glycol, pentane- 1,2-diol, dipropylene glycol, hexylene glycol, ethylene glycol, or mixtures thereof, preferably from propylene glycol, propane- 1,3-diol or mixtures thereof.
- the polyols are selected from propylene glycol, propane-
- the polyol is propylene glycol.
- the polyol(s), according to embodiments herein, is(are) in a total amount ranging from 0.5% to 15% by weight, preferably from 1% to 10% by weight, and more preferably 2% to 8% by weight, relative to the total weight of the composition.
- the polyol(s) is(are) selected from C3-C6 diols and is(are) in a total amount ranging from 0.5% to 15% by weight, preferably from 1% to 10% by weight, and more preferably 2% to 8% by weight, relative to the total weight of the composition.
- the polyol(s) is(are) selected from propylene glycol, propane- 1,3 diol, or their mixtures and is(are) in a total amount ranging from 0.5% to 15% by weight, preferably from 1% to 10% by weight, and more preferably 2% to 8% by weight, relative to the total weight of the composition.
- the organic solvents are selected from linear or branched and preferably saturated monoalcohols, comprising 2 to 10 carbon atoms, such as ethanol, isopropanol, aromatic alcohols such as benzyl alcohol or phenylethyl alcohol, polyol ethers, for instance ethylene glycol monomethyl, monoethyl or monobutyl ether, propylene glycol ethers thereof, for instance propylene glycol monomethyl ether, and also diethylene glycol alkyl ethers, especially C1-C4 alkyl ethers, for instance diethylene glycol monoethyl ether or monobutyl ether, alone or as a mixture.
- monoalcohols comprising 2 to 10 carbon atoms, such as ethanol, isopropanol, aromatic alcohols such as benzyl alcohol or phenylethyl alcohol, polyol ethers, for instance ethylene glycol monomethyl, monoethyl or monobutyl ether, propylene glycol ether
- organic solvents when present are in an amount ranging from 0.1% to 20% by weight, preferably ranging from 0.4% to 10% by weight, relative to the total weight of the composition.
- the composition is preferably aqueous.
- water in the composition is in an amount ranging from 30% to 99% by weight, preferably in an amount ranging from 40% to 95% by weight, and more preferably in an amount ranging from 45% to 90% by weight, relative to the total weight of the composition.
- the composition of the invention may comprise at least one oxidizing agent as disclosed below. According to this embodiment, the composition of the invention is a ready to use composition comprising at least one oxidizing agent.
- compositions (A) and (B) may be present in separate sachets or containers or in a single dual compartment sachet, optionally accompanied with suitable one or more applicators which may be identical or different, such as fine brushes, coarse brushes or sponges.
- suitable one or more applicators which may be identical or different, such as fine brushes, coarse brushes or sponges.
- the kit mentioned above may also be equipped with a means for dispensing the desired mixture onto the hair, for instance the devices described in patent FR 2586913.
- Embodiments of composition (B) of the kit provided herein include at least one oxidizing agent.
- the oxidizing agent(s), according to embodiments herein, is(are) in a total amount ranging from 0.1% to 40% by weight, and preferably from 0.5% to 20% by weight, relative to the total weight of the composition (B).
- composition of the present invention may be manufactured using known methods that are generally used in the cosmetics or dermatological field.
- the method for preparation of the composition comprises blending the glycerolated nonionic surfactant(s); sugar-based nonionic surfactant(s); fatty acid(s); and at least one additional fatty compound different from the glycerolated nonionic surfactant(s), sugar-based nonionic surfactant(s), and fatty acid(s), with a suitable solvent(s), and optionally the additive(s) to obtain the composition.
- Embodiments herein also include methods for manufacturing the composition (A) and composition (B) of the kit provided herein.
- Embodiments herein include the use of the composition and/or kit of the present invention.
- composition and/or kit may be used on natural hair and/or artificial hair, including hair extensions.
- the composition and/or kit may be used at different temperatures, including conventionally at room temperature (between 25°C to 30°C) and 80°C and preferably between room temperature and 60 °C.
- Example 1 Composition for dyeing/lightening keratin fibres
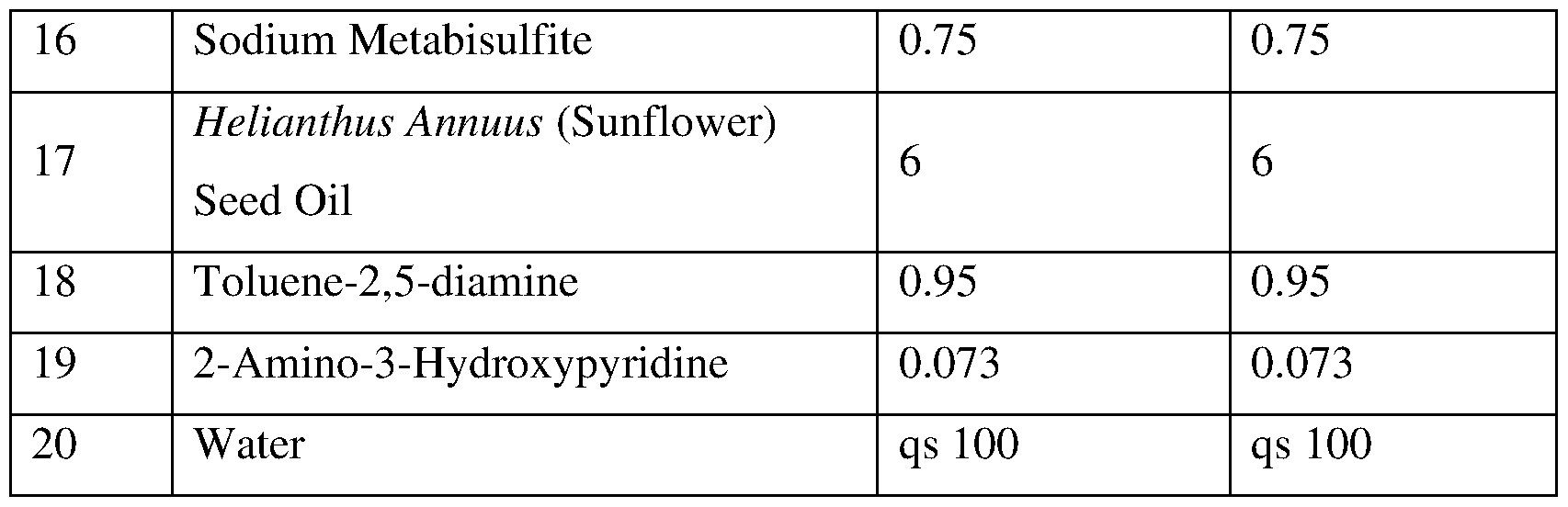
- Composition Al for dyeing and/or lightening keratin fibres was prepared using alkaline agent viz. 7.72% (w/w) of ethanolamine, glycerolated nonionic surfactants viz. 2%(w/w) of glyceryl caprylate, 1% (w/w) of polyglyceryl 4-caprate, 0.6% (w/w) of glyceryl stearate, sugar-based nonionic surfactants viz. 1.4% (w/w) of coco glucoside, 0.4% (w/w) of sucrose stearate, fatty acids viz.
- stearic acid 0.02% (w/w) of myristic acid, 0.26% (w/w) of palmitic acid, fatty compounds viz. 6% (w/w) of Helianthus Annuus (sunflower) seed oil, 18% (w/w) of cetearyl alcohol, polyols viz. 5% (w/w) of propylene glycol, cationic polysaccharides viz. 0.5% (w/w) of guar hydroxypropyltrimonium chloride, reducer/antioxidant viz 0.25 (w/w) of ascorbic acid, 0.75 (w/w) of sodium metabisulfite, and water (qs up to 100 wt%).
- composition Cl comparative composition
- composition Al comparative composition
- glycerolated nonionic surfactants and sugar-based nonionic surfactants that are more natural in origin and environmentally friendly compounds, were replaced with the surfactant oleth-20 which is synthetic and not environmentally friendly.
- Table 1 below provides the ingredients used to prepare the composition Al (according to the present invention), and comparative composition Cl, expressed in % by weight:
- compositions A2 to A8 (according to the invention) and C2 to C8 (comparative) were prepared in the same manner as the compositions Al and Cl, respectively, however different combinations of oxidative bases and couplers were used in place of the combination of toluene-2,5-diamine, and 2-amino-3- hydroxypyridine as provided in Table 1 above.
- compositions A2 and C2 the combination of 0.96% (w/w) toluene-2,5-diamine, and 0.073% (w/w) hydroxybenzo morpholine were used as the oxidative base and coupler.
- compositions A3 and C3 the combination of 0.25% (w/w) N,N-bis(2- hydroxyethyl)-p-phenylenediamine sulfate, and 0.073% (w/w) 2-amino-3- hydroxypyridine were used as the oxidative base and coupler.
- compositions A4 and C4 the combination of 0.25% (w/w) N,N-bis(2-hydroxyethyl)-p- phenylenediamine sulfate, and 0.025% (w/w) 2,4-diamino phenoxyethanol HC1 were used as the oxidative base and coupler.
- compositions A5 and C5 the combination of 0.96% (w/w) toluene-2,5-diamine, and 0.272% (w/w) m- aminophenol were used as the oxidative base and coupler.
- compositions A6 and C6 the combination of 0.96% (w/w) toluene-2,5-diamine, and 0.025% (w/w) 2,4- diamino phenoxyethanol HC1 were used as the oxidative base and coupler.
- compositions A7 and C7 the combination of 0.96% (w/w) Toluene-2,5-diamine, and 0.334% (w/w) hydroxyethyl-3,4-methylenedioxyaniline HC1 were used as the oxidative base and coupler.
- compositions A8 and C8 the combination of 0.25% (w/w) N,N-bis(2-hydroxyethyl)-p-phenylenediamine sulfate, and 0.72% (w/w) hydroxybenzo morpholine were used as the oxidative base and coupler.
- the oxidizing composition B was prepared from the ingredients of which the contents are indicated in Table 2 below, expressed in % by weight:
- Protocol Each of the compositions Al to A8 (according to the invention) and Cl to C8(comparative) were independently mixed with the oxidizing composition B in a 1: 1 weight ratio. Each of the mixtures was applied to locks of hair containing 90% natural white hair, in a proportion of 5 g of mixture per 1 g of hair.
- Results The colouring of the hair was evaluated in the L*a*b* system, using a Konica Minolta CM-3600A spectrocolorimeter (illuminant D65, angle 10°, specular component included) in the CIELab system.
- L* represents the lightness. The lower the value of L*, the darker and more powerful (intense) the colouring obtained.
- the chromaticity is measured by the values a* and b*, a* representing the red/green axis and b* the yellow/blue axis.
- compositions Al to A8 had a lower L* value, with significant AL values (AL >2) and thus showed better colour strength and colour uptake, compared to the comparative compositions Cl to C8.
- the composition of the present disclosure achieves superior colour performances, in particular demonstrating a more intense colour with long lasting colour uptake.
- the composition is environmentally friendly as it incorporates ingredients that are natural and/or are natural in origin, with a better carbon footprint and reliance on more renewable materials, thereby reducing the negative impact on environment.
- the composition contributes to the superior properties of smoother and more nourished hair, along with better shine.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Animal Behavior & Ethology (AREA)
- General Health & Medical Sciences (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Birds (AREA)
- Epidemiology (AREA)
- Emergency Medicine (AREA)
- Cosmetics (AREA)
Abstract
The present disclosure provides a composition comprising at least one glycerolated nonionic surfactant; at least one sugar-based nonionic surfactant; at least one fatty 5 acid; and at least one additional fatty compound different from the glycerolated nonionic surfactant(s), sugar-based nonionic surfactant(s), and fatty acid(s) The present disclosure also provides a kit for dyeing and/or lightening keratin fibres comprising said composition and a method for dyeing and/or lightening keratin fibres.
Description
A COMPOSITION FOR DYEING /LIGHTENING OF KERATIN FIBRES, AND METHODS THEREOF
FIELD OF INVENTION
[0001] The present disclosure relates, in general, to personal care products, and more particularly, to compositions and methods for dyeing/lightening of keratin fibres.
BACKGROUND OF INVENTION
[0002] The dyeing of keratin fibres, such as human hair, permanently via oxidation dyeing is a known practice that is widely used by consumers. This dyeing technique consists in applying to the keratin fibres a composition containing dye precursors such as oxidation bases and couplers. These dye precursors are colourless or weakly coloured compounds. When combined with an oxidizing agent, they give rise, by a process of oxidative condensation, to coloured compounds.
[0003] Oxidation dyeing is generally implemented at an alkaline pH, which produces a dyeing, and at the same time a lightening of the keratin fibre. The lightening of the fibre has the advantageous effect of emphasizing the colour, i.e., making it more visible in the case of naturally pigmented hair. In the case of depigmented hair, it helps in bringing a unified colour prior to the dyeing effect.
[0004] In general, it is preferred to have compositions that have a dyeing and/or lightening effect that impart a uniform, deep colour with a long-lasting effect. Also, consumers increasingly prefer more sustainable compositions that have a low carbon footprint, incorporate fewer compounds of petrochemical origin, and are more environmentally friendly.
[0005] Thus, there is a need for composition for dyeing and/or lightening keratin fibres which is capable of resulting in a good performance, notably in terms of dyeing and/or lightening effect, along with superior colour uptake, color vibrancy, and lastingness, and are environmentally friendly with more ingredients that are natural in origin.
SUMMARY OF THE INVENTION
[0006] In an aspect of the present disclosure, there is provided a composition comprising (a) at least one glycerolated nonionic surfactant; (b) at least one sugar-based nonionic surfactant; (c) at least one fatty acid; and (d) at least one additional fatty compound different from (a), (b) and (c).
[0007] In another aspect of the present disclosure, there is provided a kit comprising a composition (A) that comprises (a) at least one glycerolated nonionic surfactant; (b) at least one sugar-based nonionic surfactant; (c) at least one fatty acid; and (d) at least one additional fatty compound different from (a), (b) and (c); and a composition (B) comprising at least one oxidizing agent, a mixture of composition (A) and composition (B) resulting in a ready to use composition.
[0008] In yet another aspect of the present disclosure, there is provided a method for dyeing and/or lightening keratin fibres, preferably hair, comprising mixing a composition (A) that comprises (a) at least one glycerolated nonionic surfactant; (b) at least one sugar-based nonionic surfactant; (c) at least one fatty acid; and (d) at least one additional fatty compound different from (a), (b) and (c), and a composition (B) comprising at least one oxidizing agent to obtain a mixture; and applying the mixture on said fibres.
[0009] In a further aspect of the present disclosure, there is provided a use of the composition comprising (a) at least one glycerolated nonionic surfactant; (b) at least one sugar-based nonionic surfactant; (c) at least one fatty acid; and (d) at least one additional fatty compound different (a), (b) and (c), or a kit comprising a composition (A) that comprises (a) at least one glycerolated nonionic surfactant; (b) at least one sugar-based nonionic surfactant; (c) at least one fatty acid; and (d) at least one additional fatty compound different (a), (b) and (c); and a composition (B) comprising at least one oxidizing agent, a mixture of composition (A) and composition (B) resulting in a ready to use composition, for dyeing and/or lightening keratin fibres, such as hair.
[0010] These and other features, aspects, and advantages of the present subject matter will be better understood with reference to the following description and appended claims. This summary is provided to introduce a selection of concepts
in a simplified form. This summary is not intended to identify key features or essential features of the claimed subject matter, nor is it intended to be used to limit the scope of the claimed subject matter.
DESCRIPTION OF THE INVENTION
[0011] Those skilled in the art will be aware that the present disclosure is subject to variations and modifications other than those specifically described. It is to be understood that the present disclosure includes all such variations and modifications. The disclosure also includes all such steps, features, compositions, and compounds referred to or indicated in this specification, individually or collectively, and any and all combinations of any or more of such steps or features. Definitions
[0012] For convenience, before further description of the present disclosure, certain terms employed in the specification, and examples are delineated here. These definitions should be read in the light of the remainder of the disclosure and understood as by a person of skill in the art. The terms used herein have the meanings recognized and known to those of skill in the art, however, for convenience and completeness, particular terms and their meanings are set forth below.
[0013] The articles “a”, “an” and “the” are used to refer to one or to more than one (i.e., to at least one) of the grammatical object of the article.
[0014] The terms “comprise” and “comprising” are used in the inclusive, open sense, meaning that additional elements may be included. It is not intended to be construed as “consists of only”.
[0015] The term "at least one" is used to mean one or more and thus includes individual components as well as mixtures/combinations.
[0016] Throughout this specification, unless the context requires otherwise the word “comprise”, and variations such as “comprises” and “comprising”, will be understood to imply the inclusion of a stated element or step or group of element or steps but not the exclusion of any other element or step or group of element or steps.
[0017] The term “including” is used to mean “including but not limited to”. “Including” and “including but not limited to” are used interchangeably.
[0018] The term “INCI” is an abbreviation of International Nomenclature of Cosmetic Ingredients, which is a system of names provided by the International Nomenclature Committee of the Personal Care Products Council to describe personal care ingredients.
[0019] All percentages, parts and ratios are based upon the total weight of the compositions of the present disclosure unless otherwise indicated. Ratios, concentrations, amounts, and other numerical data may be presented herein in a range format. It is to be understood that such range format is used merely for convenience and brevity and should be interpreted flexibly to include not only the numerical values explicitly recited as the limits of the range, but also to include all the individual numerical values or sub-ranges encompassed within that range as if each numerical value and sub-range is explicitly recited. For example, a percentage range of about 4% to 20% should be interpreted to include not only the explicitly recited limits of about 4% to about 20%, but also to include sub-ranges, such as 5% to 8%, 12% to 18%, and so forth, as well as individual amounts, including fractional amounts, within the specified ranges, such as 5.5%, and 17.25%, for example.
[0020] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which this disclosure belongs. Although any methods and materials similar or equivalent to those described herein can be used in the practice or testing of the disclosure, the preferred methods and materials are now described. All publications mentioned herein are incorporated herein by reference.
[0021] The present disclosure is not to be limited in scope by the specific embodiments described herein, which are intended for the purposes of exemplification only. Functionally equivalent products, compositions, and methods are clearly within the scope of the disclosure, as described herein.
[0022] Embodiments herein provide a composition wherein the composition is used for dyeing/lightening keratin fibres, such as hair. The composition has good dyeing performances such as colour uptake, good colour
strength, i.e., intensity of colour, uniform colour, long-lasting colour, good lightening, and bleaching effect, and also good conditioning properties such as good nourishment, soft, moisturized hair, and shiny hair. The composition also has good working qualities such as, ease of mixing and rinsing. In particular, the composition incorporates ingredients that are natural and/or have natural origin and are therefore biodegradable and do not bioaccumulate. These natural ingredients function synergistically in the composition comprising at least one glycerolated nonionic surfactant; at least one sugar-based nonionic surfactant; at least one fatty acid; and at least one additional fatty compound different from the glycerolated nonionic surfactant(s), sugar-based nonionic surfactant(s), and fatty acid(s) function to provide improved cosmeticity, chromaticity, nourishment, and lightening/bleaching effects, while ensuring the composition is environmentally friendly.
[0023] Embodiments herein also provide a method for dyeing/lightening keratin fibres, preferably hair, by mixing the composition as described herein with a composition (B) comprising at least one oxidizing agent to obtain a mixture; and applying the mixture on said fibres. Also provided in some embodiments is a kit comprising the composition as described herein and the composition (B) and the use of the composition as described herein.
Composition
[0024] Embodiments herein provide a composition comprising at least one glycerolated nonionic surfactant; at least one sugar-based nonionic surfactant; at least one fatty acid; and at least one additional fatty compound different from the glycerolated nonionic surfactant(s), sugar-based nonionic surfactant(s), and fatty acid(s).
[0025] The composition, according to embodiments herein, is an environmentally friendly composition and demonstrates good tinctorial performances such as, intense, uniform, and long-lasting colour, good lightening/bleaching effect, and good conditioning properties while also having good working qualities.
Glycerolated nonionic surfactant
[0026] The composition according to the invention comprises one or more nonionic surfactants.
[0027] For the purposes of the invention, the term “glycerolated nonionic surfactant" means a nonionic surfactant comprising at least one mole of glycerol, preferably comprising a number of moles of glycerol ranging from 1 to 50, more preferentially from 1 to 20 and even more preferentially from 2 to 10.
[0028] The term glycerolated nonionic surfactant means mono glycerolated(one mole of glycerol) or poly glycerolated( several moles of glycerol) nonionic surfactant.
[0029] Examples of glycerolated nonionic surfactants that are preferably used, alone or as a mixture, include:
- monoesters or polyesters of linear or branched Cs to C40 acids and glycerol or polyglycerol, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20 or even from 1 to 10 mol of glycerol; in particular monoesters or diesters of linear or branched Cs to C32, better still Cs to C28 or even Cs to C24 acids, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20 or even from 1 to 10 mol of glycerol;
- linear or branched, saturated or unsaturated, monoglycerolated or polyglycerolated Cs to C40, better still C10 to C28, even better still C10 to C24 or even C10 to Cis alcohols, preferably including one or two fatty chains, and comprising from 1 to 50 mol of glycerol, preferably from 1 to 20 or even from 2 to 10 mol of glycerol.
[0030] Preferably, in some embodiments, the glycerolated nonionic surfactant(s) according to the invention do not comprise any oxyalkylenes units, such as oxy ethylene or oxypropylene units.
[0031] In some embodiments, monoesters or diesters of linear or branched, Cs to C40, more preferentially Cs to C32, better still Cs to C28 or even Cs to C24 acids
and glycerol or polyglycerol, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20 or even from 1 to 10 mol of glycerol, will be preferred most particularly, alone or as a mixture, and better still, alone or as a mixture:
- diesters of branched C12 to C32, better still C14 to C28, or even Ci6 to C24 acids, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20 or even from 2 to 10 mol of glycerol; preferentially diesters of branched Ci6 to C24 acids, comprising from 2 to 10 mol of glycerol, among which mention may be made of isostearic acid diester containing 3 mol of glycerol (INCI name: Polyglyceryl-3 diisostearate);
- monoesters of linear or branched, preferably linear, Cs to C24, better still Cs to C20, or even Cs to Cis acids, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20 or even from 1 to 10 mol of glycerol; preferentially monoesters of linear Cs to Cis acids, comprising from 1 to 10 mol of glycerol, among which mention may be made of lauric acid monoester containing 4 mol of glycerol (INCI name: Polyglyceryl-4 laurate), capric acid monoester comprising 4 mol of glycerol (INCI name: Polyglyceryl-4 caprate) or caprylic acid monoester comprising 1 mol of glycerol (INCI name: Glyceryl caprylate), and stearic acid monoester comprising 1 mol of glycerol (INCI name: Glyceryl stearate).
[0032] In some embodiments, the glycerolated nonionic surfactant(s) is/are selected from at least two different glycerolated nonionic surfactants selected from monoesters or polyesters of linear or branched Cs to C40 acids, preferably Cs to C32 acids, more preferably Cs to C28 acids and glycerol or polyglycerol comprising from 1 to 50 mol of glycerol, preferably from 1 to 20, or even from 1 to 10 mol of glycerol, or combinations thereof.
[0033] The glycerolated nonionic surfactant(s), according to embodiments herein, is(are) selected from monoesters of linear or branched, preferably linear, Cs to C24 acids, more preferably Cs to C20 acids, better from Cs to Cis acids, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20, or more preferably from 1 to 10 mol of glycerol.
[0034] Preferably, glycerolated nonionic surfactant(s), according to embodiments herein, is(are) selected from a lauric acid monoester containing 4 mol of glycerol, a capric acid monoester comprising 4 mol of glycerol, a caprylic acid monoester comprising 1 mol of glycerol, a stearic acid monoester comprising 1 mol of glycerol or combinations thereof.
[0035] The glycerolated nonionic surfactant(s), according to embodiments herein, is(are) in a total amount ranging from 0.1% to 10% by weight, preferably from 0.2% to 8% by weight, and more preferably from 0.5% to 5% by weight, better still from 1% to 5% by weight, relative to the total weight of the composition.
[0036] Preferably, the glycerolated nonionic surfactant(s) is(are) selected from monoesters of linear, Cs to C24 acids, more preferably Cs to C20 acids, better from Cs to Cis acids, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20, or more preferably from 1 to 10 mol of glycerol, and is(are) in a total amount ranging from 0.1% to 10% by weight, preferably from 0.2% to 8% by weight, and more preferably from 0.5% to 5% by weight, better still from 1% to 5% by weight, relative to the total weight of the composition.
[0037] Preferably, the glycerolated nonionic surfactant(s) is(are) selected from lauric acid monoester containing 4 mol of glycerol (INCI name: Polyglyceryl- 4 laurate), capric acid monoester comprising 4 mol of glycerol (INCI name: Polyglyceryl-4 caprate), or caprylic acid monoester comprising 1 mol of glycerol (INCI name: Glyceryl caprylate), stearic acid monoester comprising 1 mol of glycerol (INCI name: Glyceryl stearate) or combinations thereof, and is(are) in a total amount ranging from 0.1% to 10% by weight, preferably from 0.2% to 8% by weight, and more preferably from 0.5% to 5% by weight, better still from 1% to 5% by weight, relative to the total weight of the composition.
Sugar-based nonionic surfactant
[0038] The composition according to the invention comprises one or more sugar-based nonionic surfactants.
[0039] For the purposes of the invention, the term "sugar-based nonionic surfactant", means a nonionic surfactant having a monosaccharide portion (i.e., monosaccharide or oside or simple sugar) or an oligosaccharide portion (short chains formed from the sequence of monosaccharide units, possibly different) or a polysaccharide portion [long chains made up of monosaccharide units, possibly different, i.e., polyholosides or polysosides (homopolysaccharides or heteropolysaccharides)] .
[0040] Preferably, in some embodiments, the sugar-based nonionic surfactant(s) according to the invention do not comprise any oxyalkylene units, such as oxy ethylene or oxypropylene units.
[0041] The sugar-based nonionic surfactant(s) is(are) preferably selected from alkylpolyglucosides, sucrose fatty esters, sorbitan fatty esters, or combinations thereof.
[0042] The alkyl(poly)glucoside(s), is(are) represented especially by the following general Formula I:
RiO— (R2O)m-(R3)n
(I) wherein:
Ri represents a linear or branched alkyl or alkenyl radical comprising 6 to 24 carbon atoms and especially 8 to 18 carbon atoms, or an alkylphenyl radical whose linear or branched alkyl radical comprises 6 to 24 carbon atoms and especially 8 to 18 carbon atoms;
R2 represents an alkylene radical comprising 2 to 4 carbon atoms,
R3 represents a sugar unit comprising 5 to 6 carbon atoms, m denotes a value ranging from 0 to 10 and preferably 0 to 4, n denotes a value ranging from 1 to 15 and preferably 1 to 4.
[0043] In some embodiments, the alkylpoly gluco side surfactants are compounds of the formula described above in which:
Ri denotes a linear or branched, saturated or unsaturated alkyl radical comprising from 8 to 18 carbon atoms,
R2 represents an alkylene radical comprising 2 to 4 carbon atoms, m denotes a value ranging from 0 to 3 and preferably equal to 0,
R3 denotes glucose, fructose or galactose, preferably glucose; the degree of polymerization, i.e. the value of n, possibly ranging from 1 to 15 or from 1 to 4; the mean degree of polymerization more particularly being between 1 and 2.
[0044] In various embodiments, the glucoside bonds between the sugar units are generally of 1-6 or 1-4 type and preferably of 1-4 type. In some embodiments, the alkyl(poly)glucoside surfactant is an alkyl(poly)glucoside surfactant. Cs/Ci6 alkyl(poly)glucosides 1,4, and especially decyl glucosides and caprylyl/capryl glucosides are also useful.
[0045] In some embodiments, mention may be made of the commercial products sold by the company COGNIS under the names PLANTAREN® (600 CS/U, 1200 and 2000) or PLANTACARE® (818, 1200 and 2000); the products sold by the company SEPPIC under the names ORAMIX CG 110 and ORAMIX NS 10; the products sold by the company BASF under the name LUTENSOL GD 70, or else the products sold by the company CHEM Y under the name AGIO LK.
[0046] In various embodiments, use is made of Cs/Ci6- alkyl(poly)glucosides 1,4, especially as an aqueous 53% solution, such as those sold by COGNIS under the reference Plantacare® 818 UP.
[0047] Preferably, in some embodiments, the alkylpolyglucoside is selected from decyl glucoside, stearyl glucoside, lauryl glucoside, coco-glucoside, cetearyl glucoside, decyl lauryl glucoside, or combinations thereof.
[0048] More preferably, in some embodiments, the alkylpolyglucoside is selected from decyl glucoside, stearyl glucoside, lauryl glucoside, coco-glucoside, cetearyl glucoside, or combinations thereof.
[0049] The sucrose fatty ester(s) include sucrose monoesters, diesters, triesters, polyesters, or combinations thereof, and typically contain sucrose monoesters. The sucrose fatty esters include single fatty acid esters and also include homogeneous mixtures of sucrose esters, containing members with different lengths of fatty acid carbon chain and/or members with different degrees of esterification. For example, the sucrose fatty esters include mixtures of monoesters, diesters, triesters, and/or polyesters.
[0050] Sucrose fatty esters are compounds having the following formula shown in Formula II below:
wherein each of X1, X2, X3, X4, X5, X6, X7 and X8 independently is: a hydroxyl (-
OH) group, or
where: each R is an alkyl group having 3-27 carbon atoms; and when at least one of X1, X2, X3, X4, X5, X6, X7 and X8 is
each R can be a different alkyl group (e.g., having different number of carbon atoms and/or different saturation), or can be the same alkyl group.
[0051] Typically, in the sucrose fatty ester, each R has between 7 and 27 carbon atoms, and typically between 7 and 19 atoms, such as 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, or 19 carbon atoms or between 7 and 17 carbon atoms.
[0052] An alkyl group can be a straight chain or branched alkyl group, can be substituted or unsubstituted, and can be a “saturated alkyl group,” meaning that it does not contain any alkene or alkyne groups; or an “unsaturated alkyl group,” meaning that it contains at least one alkene or alkyne group. An alkyl group that includes at least one carbon-carbon double bond (C — C) also is referred to by the term “alkenyl,” and alkenyl groups optionally can be substituted. An alkyl group that includes at least one carbon-carbon triple bond (C=C) also is referred to by the term “alkynyl,” and alkynyl groups optionally can be substituted.
[0053] An exemplary monoester has the following structure as shown in Formula III:
where R is an alkyl group having 3-27 carbons, and typically 7-27 carbons.
[0054] The sucrose fatty esters include blends of sucrose fatty esters, which typically include monoesters, and can also include diesters, triesters and polyesters, which have structures according to Formula (III), above, where two (diesters), three
(triesters) or more (polyesters) of X1, X2, X3, X4, X5, X6, X7 and X8, (and typically X1 and X8) independently, are
[0055] In some embodiments, the sucrose fatty esters include sucrose fatty ester blends, for example, sucrose ester mixtures containing a specified amount (e.g., percent, by weight) of sucrose monoesters.
[0056] In various embodiments, the sucrose fatty esters are selected from sucrose fatty monoesters, such as sucrose monocaprylate, sucrose monodecanoate, sucrose monolaurate, sucrose monomyristate, sucrose monopalmitate, sucrose monostearate, sucrose monopelargonate, sucrose monoundecanoate, sucrose monotridecanoate, sucrose monopentadecanoate, and sucrose monoheptadec anoate .
[0057] Examples of sucrose fatty esters that may be used include sucrose cocoate sucrose laurate (and) aqua (and) alcohol (Surfhope(R) C-1215 commercially available from Mitsubishi-Kagaku), sucrose laurate, sucrose myristate, sucrose palmitate, sucrose polystearate, sucrose tristearate, sucrose distearate, sucrose stearate, sucrose dilaurate, sucrose hexaerucate, sucrose oleate, sucrose pentaerucate, sucrose polybehenate, sucrose polycottonseedate, sucrose polylaurate, sucrose polylinoleate, sucrose polypalmate, sucrose polyoleate, sucrose polysoyate, sucrose ricinoleate, sucrose tetraisostearate, sucrose tribehenate, sucrose hexaoleate/hexapalmitate/hexastearate, sucrose hexapalmitate, sucrose trilaurate, or combinations thereof.
[0058] In some embodiments, the sucrose fatty esters include sucrose oleate, sucrose laurate (and) aqua (and) alcohol, sucrose laurate, sucrose myristate, sucrose palmitate, sucrose polystearate, sucrose tristearate, sucrose distearate, sucrose stearate, and mixtures thereof, all of which are commercially available from Mitsubishi-Kagaku under the tradename Surfhope(R) C. The sucrose fatty ester may be used, for example, as a mixture with other ingredient, for example alcohol, such as the products sold, for example, by Mitsubishi-Kagaku under the trade name
Surfhope(R) C. Alternatively the sucrose fatty ester may also be used without additives, for example, such as the product Ryoto Sugar Ester S 370 (Ryoto).
[0059] Preferably, in some embodiments, the sucrose fatty ester(s) is(are) selected from sucrose cocoate, sucrose laurate, sucrose myristate, sucrose palmitate, sucrose polystearate, sucrose tristearate, sucrose distearate, sucrose stearate, sucrose dilaurate, sucrose hexaerucate, sucrose oleate, sucrose pentaerucate, sucrose polybehenate, sucrose polycottonseedate, sucrose polylaurate, sucrose polylinoleate, sucrose polypalmate, sucrose polyoleate, sucrose polysoyate, sucrose ricinoleate, sucrose tetraisostearate, sucrose tribehenate, sucrose hexaoleate, sucrose hexastearate, sucrose hexapalmitate, sucrose trilaurate, or combinations thereof.
[0060] More preferably, in some embodiments, the sucrose fatty ester(s) is(are) selected from sucrose oleate, sucrose stearate, sucrose distearate, sucrose tristearate, or combinations thereof.
[0061] The sorbitan esters may be selected from the group consisting of sorbitan monostearate, sorbitan tristearate, sorbitan monolaurate, sorbitan monooleate, sorbitan palmitate, or combinations thereof.
[0062] Preferably, the sugar-based nonionic surfactant(s), according to embodiments herein, is(are) selected from decyl glucoside, stearyl glucoside, lauryl glucoside, coco-glucoside, cetearyl glucoside, sucrose oleate, sucrose stearate, sucrose distearate, sucrose tristearate, or combinations thereof.
[0063] More preferably, the sugar-based nonionic surfactant(s), according to embodiments herein, is(are) selected from coco-glucoside, sucrose oleate, sucrose stearate, sucrose distearate, sucrose tristearate, or combinations thereof.
[0064] The sugar-based nonionic surfactant(s), according to embodiments herein, is(are) in a total amount ranging from 0.1% to 20% by weight, preferably from 0.5% to 10% by weight, and more preferably from 1% to 5% by weight, relative to the total weight of the composition.
[0065] Preferably, the sugar-based nonionic surfactant(s) is(are) selected from decyl glucoside, stearyl glucoside, lauryl glucoside, coco-glucoside, cetearyl glucoside, sucrose oleate, sucrose stearate, sucrose distearate, sucrose tristearate, or combinations thereof, and is(are) in a total amount ranging from 0.1% to 20% by weight, preferably from 0.5% to 10% by weight, and more preferably from 1% to 5% by weight, relative to the total weight of the composition.
Fatty acids
[0066] The composition according to the invention comprises one or more fatty acids.
[0067] The term “fatty acid” means a long-chain carboxylic acid comprising at least 6 carbon atoms, in particular from 6 to 40 carbon atoms, preferably from 8 to 30 carbon atoms. The fatty acids according to the invention preferentially comprise from 10 to 30 carbon atoms and better still from 14 to 22 carbon atoms. They may optionally be hydroxylated.
[0068] The fatty acid of the invention may be present as the free form or as the neutralized, partly or totally, form.
[0069] In some embodiments, the fatty acids present in the composition according to the invention include at least one carboxylic acid group and a linear or branched, saturated or unsaturated, in particular unsaturated, alkyl chain comprising from 6 to 40 carbon atoms, preferably from 8 to 30 carbon atoms, more preferably from 10 to 30 carbon atoms and better still from 14 to 22 carbon atoms.
[0070] In various embodiments, the fatty acids include at least one carboxylic acid group and a linear or branched, saturated or unsaturated, in particular unsaturated, alkyl chain comprising from 10 to 30 carbon atoms, in particular from 12 to 22 carbon atoms.
[0071] Preferably, in some embodiments, the fatty acids include at least one carboxylic acid group and a linear or branched, unsaturated alkyl chain comprising from 14 to 22 carbon atoms.
[0072] In some embodiments, the fatty acids are selected from compounds having the structure Rt-C(O)OH in which Rt represents a linear or branched, saturated or unsaturated alkyl group comprising from 5 to 39 carbon atoms, preferably from 7 to 29 carbon atoms, preferentially from 11 to 23 carbon atoms, better still from 13 to 19 carbon atoms.
[0073] For the purposes of the present invention, the fatty acids present in the composition are neither oxyalkylenated nor glycerolated. Further, they may be present as free form or as the neutralized, partly, or totally, form.
[0074] The fatty acids may be selected from solid fatty acids, liquid fatty acids, or combinations thereof.
[0075] For the purposes of the present invention, the term “solid fatty acid” means a fatty acid with a melting point of greater than 25 °C, preferably greater than or equal to 28°C, more preferentially greater than or equal to 30°C at atmospheric pressure (1.013xl05 Pa).
[0076] The solid fatty acids, in some embodiments, are selected from myristic acid, palmitic acid, arachidic acid, stearic acid, lauric acid, behenic acid, 12-hydroxystearic acid, or combinations thereof.
[0077] Preferably, in some embodiments, the solid fatty acid(s) are selected from lauric acid, myristic acid, palmitic acid, stearic acid, or combinations thereof, more preferably from myristic acid, palmitic acid, stearic acid, or combinations thereof.
[0078] For the purposes of the present invention, the term “liquid fatty acid” means a fatty acid with a melting point of less than or equal to 25°C, preferably less than or equal to 20°C at atmospheric pressure (1.013xl05 Pa).
[0079] The liquid fatty acid(s) according to the invention may be selected from oleic acid, linoleic acid, arachidonic acid, isostearic acid, isopalmitic acid, or combinations thereof, more preferably is oleic acid.
[0080] The fatty acid(s), according to embodiments herein, is(are) solid fatty acids, in particular, selected from fatty acids including at least one carboxylic
acid group and a linear or branched, unsaturated alkyl chain comprising from 6 to 40 carbon atoms, in particular from 10 to 30 carbon atoms.
[0081] Preferably, according to embodiments herein, the fatty acid(s) is(are) selected from at least one carboxylic acid comprising 6 to 40 carbon atoms, and preferably from the carboxylic acid comprising 10 to 30 carbon atoms, more preferably from lauric acid, myristic acid, stearic acid, palmitic acid, or combinations thereof.
[0082] Preferably, according to embodiments herein, the fatty acid(s) is(are) selected from myristic acid, stearic acid, palmitic acid, or combinations thereof.
[0083] The fatty acid(s), according to embodiments herein, is(are) in a total amount ranging from 0.1% to 15% by weight, preferably from 0.2% to 10% by weight, more preferably from 0.4% to 5% by weight, and better from 0.5% to 3% by weight, relative to the total weight of the composition.
[0084] Preferably, the fatty acid(s) is(are) solid fatty acids, in particular, selected from fatty acids including at least one carboxylic acid comprising 6 to 40 carbon atoms, and preferably from the carboxylic acid comprising 10 to 30 carbon atoms, and is(are) in a total amount ranging from 0.1% to 15% by weight, preferably from 0.2% to 10% by weight, more preferably from 0.4% to 5% by weight, and better from 0.5% to 3% by weight, relative to the total weight of the composition.
[0085] Preferably, the fatty acid(s) is(are) selected from lauric acid, myristic acid, stearic acid, palmitic acid, or combinations thereof, and is(are) in a total amount ranging from 0.1% to 15% by weight, preferably from 0.2% to 10% by weight, more preferably from 0.4% to 5% by weight, and better from 0.5% to 3% by weight, relative to the total weight of the composition.
Additional Fatty compounds
[0086] The composition according to the invention comprises one or more additional fatty compounds that are different from fatty acids, glycerolated nonionic surfactants and sugar-based nonionic surfactants previously described. The
additional fatty compounds are other than free fatty acids and neutralized fatty acids.
[0087] The term “fatty compound” means an organic compound that is insoluble in water at 25°C and at atmospheric pressure (1.013xl05 Pa) (solubility of less than 5% by weight, preferably less than 1% by weight, even more preferentially less than 0.1% by weight). They bear in their structure at least one hydrocarbon-based chain including at least 6 carbon atoms and/or a sequence of at least two siloxane groups. In addition, the fatty compounds are generally soluble in organic solvents under the same temperature and pressure conditions, for instance, chloroform, dichloromethane, carbon tetrachloride, ethanol, benzene, toluene, tetrahydrofuran (THF), liquid petroleum jelly or decamethylcyclopentasiloxane.
[0088] The fatty compounds that may be used in the present invention are neither (poly)oxyalkylenated nor glycerolated.
[0089] Preferably, the fatty compounds that may be used according to the invention are non-silicone fatty compounds.
[0090] The term “non-silicone fatty compound” refers to a fatty compound not containing any Si-0 bonds and the term “silicone fatty compound” refers to a fatty compound containing at least one Si-0 bond.
[0091] In some embodiments, the fatty compounds that are useful may be liquid fatty compounds (or oils) and/or solid fatty compounds. The term “liquid fatty compound” means a fatty compound with a melting point of less than or equal to 25°C at atmospheric pressure (1.013xl05 Pa) and the term “solid fatty compound” means a fatty compound with a melting point of greater than 25 °C at atmospheric pressure (1.013xl05 Pa).
[0092] For the purposes of the present invention, the melting point corresponds to the temperature of the most endothermic peak observed on thermal analysis (differential scanning calorimetry or DSC) as described in the standard ISO 11357-3; 1999. The melting point may be measured using a differential scanning calorimeter (DSC), for example the calorimeter sold under the name MDSC 2920
by the company TA Instruments. In the present patent application, all the melting points are determined at atmospheric pressure (1.013xl05 Pa).
[0093] More particularly, the liquid fatty compound(s) may be selected from Ce to Ci6 liquid hydrocarbons, liquid hydrocarbons comprising more than 16 carbon atoms, non-silicone oils of animal origin, oils of triglyceride type of plant (plant oils) or synthetic origin, fluoro oils, liquid fatty alcohols, liquid esters of fatty acid and/or of fatty alcohol other than triglycerides, or combinations thereof.
[0094] In some embodiments, the fatty alcohols and esters contain at least one saturated or unsaturated, linear or branched hydrocarbon-based group, comprising 6 to 40 and better still from 8 to 30 carbon atoms, which is optionally substituted, in particular, with one or more hydroxyl groups (in particular 1 to 4). If they are unsaturated, these compounds may comprise one to three conjugated or unconjugated carbon-carbon double bonds.
[0095] The Ce to Ci6 liquid hydrocarbons may be linear, branched, or optionally cyclic, and are preferably selected from alkanes. Examples that may be mentioned include hexane, cyclohexane, undecane, dodecane, isododecane, tridecane or isoparaffins, such as isohexadecane or isodecane, or combinations thereof.
[0096] The liquid hydrocarbons comprising more than 16 carbon atoms may be linear or branched, and of mineral or synthetic origin, and are preferably selected from liquid paraffins or liquid petroleum jelly (or mineral oil), polydecenes, hydrogenated polyisobutene such as Parleam®, or combinations thereof.
[0097] A hydrocarbon-based oil of animal origin that may be mentioned is perhydrosqualene.
[0098] The triglyceride oils of plant (or plant oils) or synthetic origin are preferably selected from liquid fatty acid triglycerides including from 6 to 30 carbon atoms, for instance heptanoic or octanoic acid triglycerides, or alternatively, for example, sunflower oil, maize oil, soybean oil, marrow oil, grapeseed oil, sesame
seed oil, hazelnut oil, apricot oil, macadamia oil, arara oil, castor oil, avocado oil, caprylic/capric acid triglycerides, for instance those sold by the company Stearinerie Dubois or those sold under the names Miglyol® 810, 812 and 818 by the company Dynamit Nobel, jojoba oil and shea butter oil, or combinations thereof.
[0099] The fluoro oils may be selected from perfluoromethylcyclopentane and perfluoro- 1,3 -dimethylcyclohexane, sold under the names Flutec® PCI and Flutec® PC3 by the company BNFL Fluorochemicals; perfluoro- 1,2- dimethylcyclobutane; perfluoroalkanes such as dodecafluoropentane and tetradecafluorohexane, sold under the names PF 5050® and PF 5060® by the company 3M, or bromoperfluorooctyl sold under the name Foralkyl® by the company Atochem; nonafluoromethoxybutane and nonafluoroethoxyisobutane; perfluoromorpholine derivatives such as 4-trifluoromethylperfluoromorpholine sold under the name PF 5052® by the company 3M.
[0100] The liquid fatty alcohols that are suitable are selected from linear or branched, saturated or unsaturated alcohols, preferably unsaturated or branched alcohols, comprising from 6 to 40 carbon atoms, preferably from 8 to 30 carbon atoms. These fatty alcohols are neither oxy alky lenated nor glycerolated. Examples that may be mentioned include octyldodecanol, 2-butyloctanol, 2-hexyldecanol, 2- undecylpentadecanol, isostearyl alcohol, oleyl alcohol, linolenyl alcohol, ricinoleyl alcohol, undecylenyl alcohol and linoleyl alcohol, or combinations thereof. Preferably, oleyl alcohol may be used.
[0101] The liquid esters of fatty acids and/or of fatty alcohols, other than the triglycerides mentioned previously, mention may be made notably of esters of saturated or unsaturated, linear Ci to C26 or branched C3 to C26 aliphatic mono- or poly acids and of saturated or unsaturated, linear Ci to C26 or branched C3 to C26 aliphatic mono- or polyalcohols, the total carbon number of the esters being greater than or equal to 6 and more advantageously greater than or equal to 10.
[0102] Preferably, for the esters of monoalcohols, at least one from among the alcohol and the acid is branched.
[0103] In some embodiments, the monoesters are selected from dihydroabietyl behenate; isostearyl lactate; lauryl lactate; linoleyl lactate; oleyl lactate; isostearyl octanoate; isocetyl octanoate; isocetyl isostearate; isocetyl laurate; isocetyl stearate; isodecyl octanoate; isodecyl oleate; isononyl isononanoate; isostearyl palmitate; methyl acetyl ricinoleate; octyl isononanoate; 2- ethylhexyl isononate; octyldodecyl erucate; oleyl erucate; ethyl palmitate, isopropyl palmitate, 2-ethylhexyl palmitate, 2-octyldecyl palmitate; alkyl myristates such as isopropyl 2-octyldodecyl myristate, isobutyl stearate; 2-hexyldecyl laurate, or combinations thereof.
[0104] Preferably, in some embodiments, among the monoesters of monoacids and of monoalcohols, are selected ethyl palmitate and isopropyl palmitate, alkyl myristates such as isopropyl myristate or ethyl myristate, isocetyl stearate, 2-ethylhexyl isononanoate, isodecyl neopentanoate and isostearyl neopentanoate, or combinations thereof.
[0105] In various embodiments, esters of C4 to C22 dicarboxylic or tricarboxylic acids and of Ci to C22 alcohols and esters of monocarboxylic, dicarboxylic or tricarboxylic acids and of C2 to C26 dihydroxy, trihydroxy, tetrahydroxy or pentahydroxy alcohols may also be used.
[0106] Preferably, in some embodiments, the liquid esters of fatty acids and/or of fatty alcohols is(are) selected from diethyl sebacate; diisopropyl sebacate; diisopropyl adipate; di-n-propyl adipate; dioctyl adipate; diisostearyl adipate; dioctyl maleate; glyceryl undecylenate; octyldodecyl stearoyl stearate; pentaerythrityl monoricinoleate; pentaerythrityl tetraisononanoate; pentaerythrityl tetrapelargonate; pentaerythrityl tetraisostearate; pentaerythrityl tetraoctanoate; propylene glycol; dicaprylate; propylene glycol dicaprate; tridecyl erucate; triisopropyl citrate; triisostearyl citrate; glyceryl trilactate; trioctyldodecyl citrate; trioleyl citrate; propylene glycol dioctanoate; neopentyl glycol diheptanoate; diethylene glycol diisononanoate; and polyethylene glycol distearates, or combinations thereof.
[0107] In some embodiments, fatty esters and diesters of Cf> to C30 and preferably C12 to C22 fatty acids may be included.
[0108] In some embodiments, a liquid ester of a monoacid and of a monoalcohol is preferred.
[0109] In some embodiments, the fatty compounds are selected from liquid fatty compounds, preferably from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, liquid fatty alcohols and liquid fatty esters, or combinations thereof, more preferably from plant oils.
[0110] Preferentially, the liquid fatty compound(s) are selected from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, or combinations thereof, more preferably from plant oils.
[0111] The solid fatty compounds preferably have a viscosity of greater than 2 Pa.s, measured at 25°C and at a shear rate of 1 s’1.
[0112] The solid fatty compound(s) are preferably selected from solid fatty alcohols, solid esters of fatty acids and/or of fatty alcohols, waxes, ceramides, or combinations thereof.
[0113] The term “fatty alcohol” means a long-chain aliphatic alcohol comprising from 6 to 40 carbon atoms, preferably from 8 to 30 carbon atoms, and comprising at least one hydroxyl group OH. These fatty alcohols are neither oxyalkylenated nor glycerolated.
[0114] In some embodiments, the solid fatty alcohols may be saturated or unsaturated, and linear or branched, and include from 6 to 40 carbon atoms, preferably from 8 to 40 carbon atoms. Preferably, the solid fatty alcohols have the structure RV-OH with Rv denoting a linear alkyl group, optionally substituted with one or more hydroxyl groups, comprising from 8 to 40, preferentially from 10 to 30 carbon atoms, better still from 10 to 30, or even from 12 to 24 atoms and even better still from 14 to 22 carbon atoms.
[0115] In various embodiments, the solid fatty alcohols that may be used are preferably selected from saturated or unsaturated, linear or branched, preferably
linear and saturated, (mono)alcohols including from 6 to 40 carbon atoms, better still from 10 to 30, or even from 12 to 24 and even better still from 14 to 22 carbon atoms.
[0116] Examples of the solid fatty alcohols that may be used may be selected, alone or as a mixture, from: myristyl alcohol (or 1 -tetradecanol); cetyl alcohol (or 1 -hexadecanol); stearyl alcohol (or 1 -octadecanol); arachidyl alcohol (or 1-eicosanol); behenyl alcohol (or 1 -docosanol); lignoceryl alcohol (or 1- tetracosanol); ceryl alcohol (or 1-hexacosanol); montanyl alcohol (or 1- octacosanol); myricyl alcohol (or 1-triacontanol).
[0117] Preferably, in some embodiments, the solid fatty alcohol is selected from cetyl alcohol, stearyl alcohol, behenyl alcohol, myristyl alcohol, arachidyl alcohol, or combinations thereof, such as cetylstearyl alcohol or cetearyl alcohol. Particularly preferably, the solid fatty alcohol is selected from cetyl alcohol, stearyl alcohol, or combinations thereof such as cetylstearyl alcohol, or cetearyl alcohol.
[0118] The solid esters of a fatty acid and/or of a fatty alcohol that may be used selected from esters derived from a C9-C26 carboxylic fatty acid and/or from a C9-C26 fatty alcohol.
[0119] Preferably, in some embodiments, these solid fatty esters are esters of a linear or branched, saturated carboxylic acid including at least 10 carbon atoms, preferably from 10 to 30 carbon atoms and more particularly from 12 to 24 carbon atoms, and of a linear or branched, saturated monoalcohol, including at least 10 carbon atoms, preferably from 10 to 30 carbon atoms and more particularly from 12 to 24 carbon atoms. The saturated carboxylic acids may optionally be hydroxylated and are preferably monocarboxylic acids.
[0120] Preferably, in some embodiments, these solid fatty esters are esters of a linear or branched, saturated carboxylic acid including at least 10 carbon atoms, preferably from 10 to 30 carbon atoms and more particularly from 12 to 24 carbon atoms, and of a diol including from 2 to 4 carbon atoms, more preferably glycol distearate.
[0121] In various embodiments, esters of C4-C22 dicarboxylic or tricarboxylic acids and of C1-C22 alcohols and esters of monocarboxylic, dicarboxylic or tricarboxylic acids and of C2-C26 dihydroxy, trihydroxy, tetrahydroxy or pentahydroxy alcohols may also be used.
[0122] In some embodiments, the solid esters of a fatty acid and/or of a fatty alcohol are selected from octyldodecyl behenate, isocetyl behenate, cetyl lactate, stearyl octanoate, octyl octanoate, cetyl octanoate, decyl oleate, hexyl stearate, octyl stearate, myristyl stearate, cetyl stearate, stearyl stearate, octyl pelargonate, cetyl myristate, myristyl myristate, stearyl myristate, octyl palmitate, myristyl palmitate, cetyl palmitate, stearyl palmitate, or combinations thereof.
[0123] Preferably, the solid esters of a fatty acid and/or of a fatty alcohol are selected from C9-C26 alkyl palmitates, notably myristyl palmitate, cetyl palmitate or stearyl palmitate; C9-C26 alkyl myristates, such as cetyl myristate, stearyl myristate and myristyl myristate; and C9-C26 alkyl stearates, notably myristyl stearate, cetyl stearate and stearyl stearate, or combinations thereof.
[0124] For the purposes of the present invention, a wax is a lipophilic compound, which is solid at 25°C and atmospheric pressure, with a reversible solid/liquid change of state, having a melting point greater than about 40°C, which may be up to 200°C, and having in the solid state anisotropic crystal organization. In general, the size of the wax crystals is such that the crystals diffract and/or scatter light, giving the composition that comprises them a more or less opaque cloudy appearance. By bringing the wax to its melting point, it is possible to make it miscible with oils and to form a microscopically homogeneous mixture, but on returning the temperature of the mixture to room temperature, recrystallization of the wax, which is microscopically and macroscopically detectable (opalescence), is obtained.
[0125] In particular, the waxes that are suitable for use in the invention may be selected from waxes of animal, plant or mineral origin, non-silicone synthetic waxes, or combinations thereof.
[0126] In some embodiments, the waxes may be selected from hydrocarbon-based waxes, for instance beeswax, notably of organic origin, lanolin wax and Chinese insect waxes; rice bran wax, carnauba wax, candelilla wax, ouricury wax, esparto grass wax, berry wax, shellac wax, Japan wax and sumac wax; montan wax, orange wax and lemon wax, microcrystalline waxes, paraffins and ozokerite; polyethylene waxes, the waxes obtained by Fischer-Tropsch synthesis and waxy copolymers, and also esters thereof.
[0127] In various embodiments, the waxes are selected from C20 to Ceo microcrystalline waxes, such as Microwax HW. Mention may also be made of the MW 500 polyethylene wax sold under the reference Permalen 50-L polyethylene.
[0128] In some embodiments, the waxes obtained by catalytic hydrogenation of animal or plant oils containing linear or branched Cs to C32 fatty chains may be selected. Among these waxes mention may notably be made of isomerized jojoba oil such as the trans-isomerized partially hydrogenated jojoba oil, notably the product manufactured or sold by the company Desert Whale under the commercial reference Iso-Jojoba-50®, hydrogenated sunflower oil, hydrogenated castor oil, hydrogenated coconut kernel oil, hydrogenated lanolin oil and bis( 1,1,1 - trimethylolpropane) tetrastearate, notably the product sold under the name Hest 2T- 4S® by the company Heterene.
[0129] The waxes obtained by hydrogenation of castor oil esterified with cetyl alcohol, such as those sold under the names Phytowax Castor 16L64® and 22L73® by the company Sophim, may also be used.
[0130] A wax that may also be used is a C20-C40 alkyl (hydroxystearyloxy)stearate (the alkyl group containing from 20 to 40 carbon atoms), alone or as a mixture. Such a wax is notably sold under the names Kester Wax K 82 P®, Hydroxypolyester K 82 P® and Kester Wax K 80 P® by the company Koster Keunen.
[0131] It is also possible to use microwaxes in some embodiments; mention may notably be made of carnauba microwaxes, such as the product sold under the
name MicroCare 350® by the company Micro Powders, synthetic-wax microwaxes, such as the product sold under the name MicroEase 114S® by the company Micro Powders, microwaxes constituted of a mixture of carnauba wax and polyethylene wax, such as the products sold under the names Micro Care 300® and 310® by the company Micro Powders, microwaxes constituted of a mixture of carnauba wax and of synthetic wax, such as the product sold under the name Micro Care 325® by the company Micro Powders, polyethylene microwaxes, such as the products sold under the names Micropoly 200®, 220®, 220L® and 250S® by the company Micro Powders, and polytetrafluoroethylene microwaxes, such as the products sold under the names Microslip 519® and 519 L® by the company Micro Powders.
[0132] In some embodiments, the waxes are preferably selected from mineral waxes, for instance paraffin, petroleum jelly, lignite or ozokerite wax; plant waxes, for instance cocoa butter or cork fibre or sugar cane waxes, olive tree wax, rice wax, hydrogenated jojoba wax, ouricury wax, carnauba wax, candelilla wax, esparto grass wax, or absolute waxes of flowers, such as the essential wax of blackcurrant blossom sold by the company Bertin (France); waxes of animal origin, for instance beeswaxes or modified beeswaxes (cera bellina), spermaceti, lanolin wax and lanolin derivatives; microcrystalline waxes; or combinations thereof.
[0133] The ceramides, or ceramide analogues such as glycoceramides, which may be used in the compositions according to the invention, are known; mention may be made in particular of ceramides of classes I, II, III and V according to the Dawning classification.
[0134] The ceramides or analogues thereof that may be used preferably correspond to the following formula (IV):
R’”CH(OH)CH(CH2OR”)(NHCOR’),
(IV) in which:
R’ denotes a linear or branched, saturated or unsaturated alkyl group, derived from C14-C30 fatty acids, it being possible for this group to be substituted with a hydroxyl group in the alpha position, or a hydroxyl group in the omega position esterified with a saturated or unsaturated C16-C30 fatty acid;
R” denotes a hydrogen atom, a (glycosyl)n group, a (galactosyl)m group or a sulfogalactosyl group, in which n is an integer ranging from 1 to 4 and m is an integer ranging from 1 to 8;
R’” denotes a C15-C26 hydrocarbon-based group, saturated or unsaturated in the alpha position, this group possibly being substituted with one or more C1-C14 alkyl groups; it being understood that in the case of natural ceramides or glycoceramides, R’” may also denote a C15-C26 alpha-hydroxyalkyl group, the hydroxyl group being optionally esterified with a C16-C30 alpha-hydroxy acid.
[0135] In some embodiments, the ceramides that are more particularly preferred are the compounds for which R’ denotes a saturated or unsaturated alkyl derived from C16-C22 fatty acids; R” denotes a hydrogen atom and R’” denotes a saturated linear C15 group.
[0136] Preferentially, use is made of ceramides for which R’ denotes a saturated or unsaturated alkyl group derived from C14-C30 fatty acids; R” denotes a galactosyl or sulfogalactosyl group; and R’” denotes a -CH=CH-(CH2)i2-CH3 group.
[0137] In some embodiments, the ceramides are selected from compounds for which R’ denotes a saturated or unsaturated alkyl radical derived from C12-C22 fatty acids; R” denotes a galactosyl or sulfogalactosyl radical and R’” denotes a saturated or unsaturated C12-C22 hydrocarbon-based radical and preferably a - CH=CH-(CH2)i2-CH3 group.
[0138] In various embodiments, the ceramides that may be used are selected from 2-N-linoleoylaminooctadecane- 1 ,3 -diol, 2-N-oleoylaminooctadecane- 1,3- diol, 2-N-palmitoylaminooctadecane- 1 ,3-diol, 2-N-stearoylaminooctadecane- 1 ,3- diol, 2-N-behenoylaminooctadecane-l,3-diol, 2-N-[2-
hydroxypalmitoyl]aminooctadecane-l,3-diol, 2-N-stearoylaminooctadecane- 1,3,4- triol and in particular N-stearoylphytosphingosine, 2-N- palmitoylaminohexadecane- 1 ,3-diol, N -linoleoyldihydrosphingosine, N - oleoyldihydro sphingosine, N-palmitoyldihydrosphingosine, N- stearoyldihydrosphingosine, and N-behenoyldihydrosphingosine, N-docosanoyl- N-methyl-D-glucamine, cetylic acid N-(2-hydroxyethyl)-N-(3-cetyloxy-2- hydroxypropyl) amide and bis(N-hydroxyethyl-N-cetyl)malonamide, or combinations thereof. N-oleoyldihydrosphingosine will preferably be used.
[0139] The solid fatty compounds are preferably selected from solid fatty alcohols, in particular from cetyl alcohol, stearyl alcohol or combinations thereof such as cetylstearyl or cetearyl alcohol.
[0140] Butter may also be used.
[0141] For the purposes of the present invention, the term “butter” (also referred to as a “pasty fatty compound”) means a lipophilic fatty compound with a reversible solid/liquid change of state, comprising at a temperature of 25°C and at atmospheric pressure (760 mmHg) a liquid fraction and a solid fraction. Preferably, the butter(s) according to the invention have a melting start temperature of more than 25°C and a melting end temperature of less than 60°C.
[0142] Preferably, the particular butter(s) are of plant origin, such as those described in Ullmann’s Encyclopedia of Industrial Chemistry (“Fats and Fatty Oils”, A. Thomas, published online: 15 JUN 2000, DOI: 10.1002/14356007. al0_173, point 13.2.2.2. Shea Butter, Borneo Tallow, and Related Fats (Vegetable Butters)).
[0143] Mention may be made more particularly of shea butter, Karite Nilotica butter (Butyrospermum parkii). galam butter, (Butyrospermum parkii). Borneo butter or fat or tengkawang tallow (Shorea slenoplera). shorea butter, illipe butter, madhuca butter or Bassia madhuca longifolia butter, mowrah butter (Madhuca lai if olio). katiau butter (Madhuca molt ley ana), phulwara butter (M. bulyracea). mango butter (Mangifera indica), murumuru butter (Astrocaryum
murumuru), kokum butter {Garcinia indica), ucuuba butter (Virola sebifera), tucuma butter, painya butter (Kpangnan) Pentadesma butyracea), coffee butter (Coff'ea arabica), apricot butter (Prunus armeniaca). macadamia butter {Macadamia ternifolia), grapeseed butter {Vitis vinifera), avocado butter {Persea gralissima). olive butter (Olea europaea), sweet almond butter {Prunus amygdalus dulcis), cocoa butter and sunflower butter.
[0144] Preferably, the additional fatty compound(s) different from the fatty acids, glycerolated nonionic surfactants and sugar-based nonionic surfactants according to embodiments herein, is(are) selected from liquid fatty compounds, solid fatty compounds, or combinations thereof, preferably from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, liquid fatty alcohols, liquid fatty esters, silicone oils, solid fatty alcohols, solid fatty alcohol esters, waxes, ceramides, or combinations thereof.
[0145] More preferably, the additional fatty compound(s), according to embodiments herein, is(are) selected from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, liquid fatty esters, solid fatty alcohols, solid fatty esters or combinations thereof, more preferably from plant oils, solid fatty alcohols, solid fatty esters or combinations thereof.
[0146] Preferably, the additional fatty compound(s), is(are) selected from liquid fatty compound, solid fatty compounds or combinations thereof, preferably from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, liquid fatty alcohols, liquid fatty esters, silicone oils, solid fatty alcohols, solid fatty esters, waxes, ceramides, or combinations thereof, and is(are) in a total amount ranging from 5% to 45% by weight, preferably 10% to 40% by weight, and more preferably 15% to 35% by weight, better still from 20% to 32% by weight, relative to the total weight of the composition.
[0147] Preferably, the additional fatty compound(s), is(are) selected from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, liquid fatty esters, solid fatty alcohols, solid fatty esters or combinations thereof, and is(are) in a total amount ranging from 5% to 45% by weight, preferably 10% to 40% by
weight, and more preferably 15 to 35% by weight, better still from 20% to 32% by weight, relative to the total weight of the composition.
Alkaline agent
[0148] The composition, according to the invention, may comprise at least one alkaline agent.
[0149] Preferably, the composition, according to the present invention comprises at least one alkaline agent.
[0150] The alkaline agent(s) that may be used is(are) selected from at least one mineral, organic, or hybrid alkaline agent or combinations thereof.
[0151] The mineral alkaline agents are selected from ammonium hydroxide, alkali metal carbonates or bicarbonates such as sodium (hydrogen)carbonate and potassium (hydrogen)carbonate, alkali metal or alkaline-earth metal silicates or metasilicates such as sodium metasilicate, or combinations thereof.
[0152] The organic alkaline agents are selected from alkanolamines, amino acids, organic amines other than alkanolamines, oxyethylenated and/or oxypropylenated ethylenediamines, 1,3 -diaminopropane, spermine, spermidine, or combinations thereof.
[0153] The term “alkanolamine” means an organic amine comprising a primary, secondary, or tertiary amine function, and one or more linear or branched Ci-Cs alkyl groups bearing one or more hydroxyl radicals.
[0154] In some embodiments, organic amines selected from alkanolamines such as, monoalkanolamines, dialkanolamines or trialkanolamines comprising one to three identical or different Ci-Cio hydroxy alkyl radicals are in particular suitable for performing the present invention.
[0155] In particular, in some embodiments, the alkanolamine(s) are selected from monoethanolamine (MEA), diethanolamine, triethanolamine, monoisopropanolamine, diisopropanolamine, N,N-dimethylethanolamine, 2- amino-2-methyl- 1 -propanol, triisopropanolamine, 2-amino-2-methy 1-1,3-
propanediol, 3-amino- 1 ,2-propanediol, 3 -dimethylamino- 1 ,2-propanediol, tris(hydroxymethyl)aminomethane, or combinations thereof.
[0156] The alkaline agent(s), according to embodiments herein, is(are) selected from alkanolamines, ammonium hydroxide, metal carbonates, metal bicarbonates, alkali metal silicates, alkaline-earth metal silicates, alkali metal metasilicates, alkaline-earth metal metasilicates, or combinations thereof.
[0157] Preferably, the alkaline agent(s) according to embodiments herein, is(are) selected from alkanolamines and ammonium hydroxide. More preferably, the alkaline agent is selected from alkanolamines, better is, monoethanolamine.
[0158] The total amount of the alkaline agent(s), according to embodiments herein, is(are) in a total amount ranging from 0.1% to 40% by weight, preferably 1% to 30% by weight, more preferably from 2% to 25% by weight, and better from 4% to 20% by weight, relative to the total weight of the composition.
[0159] Preferably, the alkaline agent(s), according to embodiments herein, is(are) selected from alkanolamines, ammonium hydroxide, or their combinations, and is(are) in a total amount ranging from 0.1% to 40% by weight, preferably 1% to 30% by weight, more preferably from 2% to 25% by weight, and better from 4% to 20% by weight, relative to the total weight of the composition.
[0160] Preferably, the alkaline agent(s) selected from alkanolamines is(are) in a total amount ranging from 0.1% to 40% by weight, preferably 1% to 30% by weight, more preferably from 2% to 25% by weight, and better from 4% to 20% by weight, relative to the total weight of the composition.
Oxidative dye
[0161] The composition, according to the invention, may comprise one or more oxidative dyes.
[0162] Preferably, the composition according to the invention comprises one or more oxidative dyes.
[0163] The oxidative dye, according to the present invention, includes at least one oxidation base and/or at least one oxidation coupler. Preferably, the composition according to the present invention comprises one or more oxidation bases in combination with one or more couplers.
[0164] Oxidation bases are generally colourless or weakly coloured compounds which when mixed with oxidizing compounds give rise to coloured compounds or dye. Further, the colour shades obtained with such oxidation bases can be varied by combining them with oxidation couplers or colour modifiers.
[0165] The oxidation base(s), according to the present invention, that may be used, is(are) selected from a group consisting of para-phenylenediamines, bis(phenyl)alkylenediamines, para-aminophenols, ortho-aminophenols, heterocyclic bases, and their addition salts thereof.
[0166] The addition salts of the oxidation bases are preferably selected from the addition salts with an acid, such as, the hydrochlorides, hydrobromides, sulfates, citrates, succinates, tartrates, lactates, tosylates, benzenesulfonates, methanesulfonates, phosphates and acetates, and the addition salts with a base such as, sodium hydroxide, potassium hydroxide, aqueous ammonia, amines, or alkanolamines.
[0167] The para-phenylenediamines that may be used are selected from para-phenylenediamine, para-tolylenediamine, 2-chloro-para-phenylenediamine, 2,3-dimethyl-para-phenylenediamine, 2,6-dimethyl-para-phenylenediamine, 2,6- diethyl-para-phenylenediamine, 2,5 -dimethyl-para-phenylenediamine, N,N - dimethyl-para-phenylenediamine, N,N -diethyl-para-phenylenediamine, N,N- dipropyl-para-phenylenediamine, 4-amino-N,N-diethyl-3-methylaniline, N,N- bis(P-hydroxyethyl)-para-phenylenediamine, 4-N,N-bis(0-hydroxyethyl)amino-2- methylaniline, 4-N,N-bis(0-hydroxyethyl)amino-2-chloroaniline, 2-0- hydroxyethyl-para-phenylenediamine, 2-y-hydroxypropyl-para-phenylenediamine, 2-methoxymethyl-para-phenylenediamine, 2-fluoro-para-phenylenediamine, 2- isopropyl-para-phenylenediamine, N-(0-hydroxypropyl)-para-phenylenediamine,
2-hydroxymethyl-para-phenylenediamine, N,N-dimethyl-3-methyl-para- phenylenediamine, N,N-(ethyl-P-hydroxyethyl)-para- phenylenediamine, N-(0,y- dihydroxypropyl)-para-phenylenediamine, N-(4’-aminophenyl)-para- phenylenediamine, N-phenyl-para-phenylenediamine, 2-0-hydroxyethyloxy-para- phenylenediamine, 2-0-acetylaminoethyloxy -para-phenylenediamine, N-(0- methoxyethyl)-para-phenylenediamine, 2-thienyl-para-phenylenediamine, 4- aminophenylpyrrolidine, 3-hydroxy- 1-(4’ -aminophenyl)pyrrolidine, 2-0- hydroxyethylamino-5-aminotoluene, and/or the addition salts thereof with an acid.
[0168] In some embodiments, the para-phenylenediamines are selected from para-phenylenediamine, para-tolylenediamine, 2-isopropyl-para- phenylenediamine, 2- 0- hydroxycthy I -para-phcny Icncdi am i nc, 2-y-hydroxyethyl- para-phenylenediamine, 2-methoxymethyl -para-phenylenediamine, 2-0- hydroxyethyloxy -para-phenylenediamine, 2,6-dimethyl-para-phenylenediamine, 2,6-diethyl-para-phenylenediamine, 2,3-dimethyl-para-phenylenediamine, N,N- bis(0-hydroxyethyl)-para-phenylenediamine, 2-chloro-para-phenylenediamine, 2- 0-acetylaminoethyloxy-para-phenylenediamine, or, preferably, the addition salts thereof with an acid, or combinations thereof.
[0169] The bis(phenyl)alkylenediamines that may be used are selected from N,N’-bis(0-hydroxyethyl)-N,N’-bis(4’ -aminophenyl)- 1,3 -diaminopropanol, N,N’- bis-(0-hydroxyethyl)-N,N’-bis(4’-aminophenyl)ethylenediamine, N,N’-bis(4- aminophenyl)tetramethylenediamine, N,N’-bis(0-hydroxyethyl)-N,N’-bis(4- aminophenyl)tetramethylenediamine, N,N’ -bis(4- methylaminophenyl)tetramethylenediamine, N,N’-bis(ethyl)-N,N’-bis(4 ’-aminos’ -methylphenyl)ethylenediamine, l,8-bis(2,5-diaminophenoxy)-3,6-dioxaoctane, or their addition salts thereof.
[0170] The para-aminophenols may be selected from, para-aminophenol, 4- amino-3-methylphenol, 4-amino-3-fluorophenol, 4-amino-3-chlorophenol, 4- amino-3-hydroxymethylphenol, 4-amino-2-methylphenol, 4-amino-2- hydroxymethylphenol, 4-amino-2-methoxymethylphenol, 4-amino-2-
aminomethylphenol, 4-amino-2-(P-hydroxyethylaminomethyl)phenol and 4- amino-2-fluorophenol, or the addition salts thereof with an acid. In an example, the addition salt is salt of sulphurous acid or sodium metabisulphite salt.
[0171] The ortho-aminophenols that may be used are selected from, for example, 2-aminophenol, 2-amino-5-methylphenol, 2-amino-6-methylphenol and 5-acetamido-2-aminophenol, or the addition salts thereof.
[0172] The heterocyclic bases that may be used are selected from pyridine derivatives, pyrimidine derivatives and pyrazole derivatives.
[0173] In some embodiments, pyridine derivatives may be selected from the compounds as described, for example, in patents GB 1 026 978 and/or GB 1 153 196, including 2,5-diaminopyridine, 2-(4-methoxyphenyl)amino-3- aminopyridine, 3,4-diaminopyridine, or the addition salts thereof.
[0174] In various embodiments, the pyridine derivatives may further be selected from 3-aminopyrazolo[l,5-a]pyridine oxidation bases, or addition salts thereof described, for example, in patent application FR 2 801 308. Examples of such bases include pyrazolo[l,5-a]pyrid-3-ylamine, 2-acetylaminopyrazolo[l,5- a]pyrid-3 -ylamine, 2-morpholin-4-ylpyrazolo [ 1 ,5-a]pyrid-3 -ylamine, 3 - aminopyrazolo[ 1 ,5-a]pyridine-2-carboxylic acid, 2-methoxypyrazolo[l ,5-a]pyrid- 3-ylamine, (3-aminopyrazolo[l,5-a]pyrid-7-yl)methanol, 2-(3-aminopyrazolo[l,5- a]pyrid-5-yl)ethanol, 2-(3-aminopyrazolo[l,5-a]pyrid-7-yl)ethanol, (3- aminopyrazolo[ 1 ,5-a]pyrid-2-yl)methanol, 3,6-diaminopyrazolo[ 1 ,5-a]pyridine, 3 ,4-diaminopyrazolo [ 1 ,5-a]pyridine, pyrazolo [ 1 ,5-a]pyridine-3 ,7 -diamine, 7 - morpholin-4-ylpyrazolo[l,5-a]pyrid-3-ylamine, pyrazolo[l,5-a]pyridine-3,5- diamine, 5-morpholin-4-ylpyrazolo[l,5-a]pyrid-3-ylamine, 2-[(3- aminopyrazolo[l,5-a]pyrid-5-yl)(2-hydroxyethyl)amino]ethanol, 2-[(3- aminopyrazolo[l,5-a]pyrid-7-yl)(2-hydroxyethyl)amino]ethanol, 3- aminopyrazolo [1,5- a]pyridin- 5 -ol, 3 - aminopyrazolo [ 1 , 5 - a] pyridin-4 -ol, 3 - aminopyrazolo[l,5-a]pyridin-6-ol and 3-aminopyrazolo[l,5-a]pyridin-7-ol, 2-(3- amino-pyrazolo[l,5-a]pyridin-2-yl)oxyethanol, or the addition salts thereof.
[0175] In some embodiments, the pyrimidine derivatives may be selected from the compounds described, for example, in the patents DE 2359399; JP 88- 169571; JP 05-63124; EP 0770375 or patent application WO 96/15765, including
2.4.5.6-tetraaminopyrimidine, 4-hydroxy-2,5,6-triaminopyrimidine, 2 -hydroxy -
4.5.6-triaminopyrimidine, 2,4-dihydroxy-5,6-diaminopyrimidine, 2,5,6- triaminopyrimidine, or their addition salts and their tautomeric forms, when a tautomeric equilibrium exists.
[0176] The pyrazole derivatives, in some embodiments, may be selected from, for example, the compounds described in the patents DE 3843892, DE 4133957 or patent applications WO 94/08969, WO 94/08970, FR-A-2 733 749 or DE 195 43 988, including for example 4,5-diamino-l-methylpyrazole, 4,5- diamino- l-(|3-hydroxyethyl)pyrazole, 3,4-diaminopyrazole, 4,5-diamino- 1-(4’- chlorobenzyl)pyrazole, 4,5-diamino-l,3-dimethylpyrazole, 4,5-diamino-3-methyl- 1-phenylpyrazole, 4,5-diamino-l-methyl-3-phenylpyrazole, 4-amino-l,3- dimethyl-5-hydrazinopyrazole, l-benzyl-4,5-diamino-3-methylpyrazole, 4,5- diamino-3-tert-butyl- 1-methylpyrazole, 4,5-diamino- l-tert-butyl-3-methyl- pyrazole, 4,5-diamino- l-(P-hydroxyethyl)-3-methylpyrazole, 4,5-diamino- 1-ethyl- 3-methylpyrazole, 4,5-diamino- l-ethyl-3-(4’-methoxyphenyl)pyrazole, 4,5- diamino- l-ethyl-3-hydroxymethylpyrazole, 4,5-diamino-3-hydroxymethyl- 1- methylpyrazole, 4,5-diamino-3-hydroxymethyl- 1-isopropylpyrazole, 4,5-diamino- 3-methyl-l-isopropylpyrazole, 4-amino-5-(2’-aminoethyl)amino-l,3- dimethylpyrazole, 3,4,5-triaminopyrazole, l-methyl-3,4,5-triaminopyrazole, 3,5- diamino-l-methyl-4-methylaminopyrazole, 3,5-diamino-4-(P-hydroxyethyl)- amino- 1-methylpyrazole, or their addition salts. 4,5-diamino- l-(P-methoxyethyl)- pyrazole may also be used.
[0177] In various embodiments, mention may be preferably made to 4,5- diaminopyrazole and even more preferably of 4,5-diamino-l-(P- hydroxyethyl)pyrazole, or its salts thereof.
[0178] In a further embodiment, the pyrazole derivatives that may be used, also include diamino-N,N-dihydropyrazolo pyrazolones and especially those
described in patent application FRA 2886136, such as the following compounds and the addition salts thereof: 2,3-diamino-6,7-dihydro-lH,5H-pyrazolo[l,2- a]pyrazol-l-one, 2-amino-3-ethylamino-6,7-dihydro-lH,5H-pyrazolo[l,2- a]pyrazol-l-one, 2-amino-3-isopropylamino-6,7-dihydro-lH,5H-pyrazolo[l,2- a]pyrazol-l-one, 2-amino-3-(pyrrolidin-l-yl)-6,7-dihydro-lH,5H-pyrazolo[l,2- a]pyrazol-l-one, 4,5-diamino-l,2-dimethyl-l,2-dihydropyrazol-3-one, 4,5- diamino- 1 ,2-diethyl- 1 ,2-dihydropyrazol-3 -one, 4,5-diamino- 1 ,2-di-(2- hydroxyethyl)-l,2-dihydropyrazol-3-one, 2-amino-3-(2-hydroxyethyl)amino-6,7- dihydro-lH,5H-pyrazolo[l,2-a]pyrazol-l-one, 2-amino-3-dimethylamino-6,7- dihydro-lH,5H-pyrazolo[l,2-a]pyrazol-l-one, 2,3-diamino-5,6,7,8-tetrahydro- lH,6H-pyridazino[ 1 ,2-a]pyrazol- 1 -one, 4-amino- 1 ,2-diethyl-5-(pyrrolidin- 1 -yl)- l,2-dihydropyrazol-3-one, 4-amino-5-(3-dimethylaminopyrrolidin-l-yl)-l,2- diethyl-l,2-dihydropyrazol-3-one, 2,3-diamino-6-hydroxy-6,7-dihydro-lH,5H- pyrazolo[l,2-a]pyrazol- 1-one, 2,3-diamino-6,7-dihydro-lH,5H-pyrazolo[l,2- a]pyrazol-l-one and/or, preferably, a salt thereof, or combinations thereof. Mention may be preferably made to 2,3-diamino-6,7-dihydro-lH,5H-pyrazolo[l,2- a]pyrazol-l-one, or its salts thereof.
[0179] In some embodiments, the heterocyclic base may be selected from 4,5-diamino- l-(|3-hydroxyethyl)pyrazole, 2,3-diamino-6,7-dihydro-lH,5H- pyrazolo[l,2-a]pyrazol- 1-one, 2-(3-aminopyrazolo[l,5-a]pyridin-2-yl)oxy ethanol, or their salts thereof.
[0180] In various embodiments, the oxidation bases are selected from paraphenylenediamines, bis(phenyl)alkylenediamines, para-aminophenols, orthoaminophenols, heterocyclic bases, the corresponding addition salts, or combinations thereof, preferably from 2-methoxymethyl-para-phenylenediamine, 2-P-hydroxyethyl-para-phenylenediamine, 2-y-hydroxypropyl-para- phenylenediamine, their addition salts thereof, or combinations thereof.
[0181] The oxidation coupler(s), according to the present invention, that may be used, is(are) selected from a group consisting of meta-phenylenediamines,
meta- aminophenols, meta-diphenols, naphthalene-based couplers, heterocyclic couplers, and their addition salts.
[0182] In some embodiments, the oxidation couplers may be selected from 1,3-dihydroxybenzene, l,3-dihydroxy-2-methylbenzene, 4-chloro-l,3- dihydroxybenzene, 1 -hydroxy-3 -aminobenzene, 1 -methyl-2-hydroxy-4-P- hydroxy ethylaminobenzene, 4-amino-2-hydroxytoluene, 5-amino-6-chl oro-2 - m ethylphenol, 2, 4-diamino-l-(P-hydroxy ethyloxy )benzene, 2-amino-4-(P- hydroxy ethylamino)- 1 -methoxybenzene, 1 ,3 -diaminobenzene, 1 ,3 -bis(2,4- diaminophenoxy)propane, 3 -ureidoaniline, 3 -ureido- 1 -dimethylaminobenzene, sesamol, l-P-hydroxyethylamino-3,4-m ethylenedi oxybenzene, a-naphthol, 2- m ethyl- 1 -naphthol, 6-hydroxyindole, 4-hydroxyindole, 4-hydroxy-N- methylindole, 5-methoxy-6-hydroxyindole, 2-amino-3 -hydroxypyridine, 6- hydroxybenzomorpholine, 2-amino-4-hydroxy ethylaminoanisole, 3-amino-6- methoxy-2-methylaminopyridine, 3,5-diamino-2,6-dimethoxypyridine, 1-N-(P- hydroxyethyl)amino-3,4-methylenedi oxybenzene, 2,6-bis(P- hydroxyethylamino)toluene, 6-hydroxyindoline, 2,6-dihydroxy-4-methylpyridine, 2-chl oro-3, 5-diaminopyri dine, 2-chloro-3,5-diamino-6-methoxypyridine, 2- chl oro-3, 5-diamino-6-m ethylpyridine, l-H-3-methylpyrazol-5-one, l-phenyl-3- methylpyrazol-5-one, 4-(3, 5-diaminopyri din-2 -yl)- 1 -(2-hydroxy ethyl)- 1 - methylpiperazin-l-ium chloride, 2,6-dimethylpyrazolo[l,5-b]-l,2,4-triazole, 2,4,6- trimethoxyaniline hydrochloride, 2,6-dimethyl[3,2-c]-l,2,4-triazole, 6- methylpyrazolo[l,5-a]benzimidazole and 2,6-diaminopyrazine, the addition salts thereof with an acid, or combinations thereof.
[0183] In various embodiments, the oxidation couplers may be selected from 1,3-dihydroxybenzene, l,3-dihydroxy-2-methylbenzene, 4-chloro-l,3- dihydroxybenzene, 1 -hydroxy-3 -aminobenzene, 1 -methyl-2-hydroxy-4-P- hydroxy ethylaminobenzene, 4-amino-2-hydroxytoluene, 5-amino-6-chl oro-2 - m ethylphenol, 2, 4-diamino-l-(P-hydroxy ethyloxy )benzene, a-naphthol, 6- hydroxyindole, 2-amino-3 -hydroxypyridine, 6-hydroxybenzomorpholine, 3- amino-6-methoxy-2-methylaminopyridine, 2-amino-4-hydroxy ethylaminoanisole,
hydroxyethyl-3,4-methylenedioxyaniline, 2-amino-5-ethylphenol, their addition salts, or combinations thereof.
[0184] The oxidation couplers, in some embodiments, are further selected from 3 -amino-6-m ethoxy -2-methylaminopyri dine, 6-hydroxybenzomorpholine,
2.4-diamino-l-(P-hydroxy ethyloxy )benzene, 2-amino-3 -hydroxypyridine, 5- amino-6-chl oro-2 -methylphenol, 1 -methyl-2-hydroxy-4-P- hydroxy ethylaminobenzene, 2-amino-4-hydroxy ethylaminoanisole, hy droxy ethyl -
3.4-m ethylenedi oxyaniline, 2-amino-5-ethylphenol, 1 -hydroxy-3 -aminobenzene, their addition salts, or combinations thereof.
[0185] In general, the addition salts of the oxidation couplers, according to the present invention, are selected from addition salts with an acid, such as, hydrochlorides, hydrobromides, sulfates, citrates, succinates, tartrates, lactates, tosylates, benzenesulfonates, phosphates and acetates, and the addition salts with a base such as, sodium hydroxide, potassium hydroxide, aqueous ammonia, amines or alkanolamines.
[0186] In various embodiments, the oxidation couplers are selected from 6- hydroxybenzomorpholine, 2, 4-diamino-l-(P-hydroxy ethyloxy )benzene, 2-amino- 3 -hydroxypyridine, 5-amino-6-chloro-2-methylphenol, l-methyl-2-hydroxy-4-P- hydroxy ethylaminobenzene, 2-amino-4-hydroxy ethylaminoanisole, hy droxy ethyl -
3, 4-m ethylenedi oxyaniline, 2-amino-5-ethylphenol, 1 -hydroxy-3 -aminobenzene, the addition salts thereof, the salts thereof, solvates of the salts thereof, or combinations thereof.
[0187] Preferably, the oxidation couplers are selected from 6- hydroxybenzomorpholine, hydroxy ethyl-3,4-methylenedi oxyaniline, 2-amino-5- ethylphenol, their addition salts, or combinations thereof.
[0188] The composition according to embodiments herein, is, in some embodiments, free of oxidation couplers chosen from resorcinol, 2- methylresorcinol, 4-chlororesorcinol, or the addition salts thereof.
[0189] The oxidation base(s), according to embodiments herein, is(are) in a total amount ranging from 0.0001% to 10% by weight, relative to the total weight of the composition.
[0190] The oxidation coupler(s), according to embodiments herein, is(are) in a total amount ranging from 0.0001% to 10% by weight, relative to the total weight of the composition.
[0191] The oxidation base(s) and coupler(s) may be in suitable amounts such that the total amount of oxidative dye, according to embodiments herein, is in a total amount ranging from 0.0002% to 20% by weight, relative to the total weight of the composition.
Cationic polysaccharide
[0192] The composition, according to the invention, may comprise one or more cationic polysaccharides.
[0193] Preferably, the composition according to the invention, comprises one or more cationic polysaccharides.
[0194] For the purposes of the present invention, the term "cationic polysaccharide" refers to any polysaccharide comprising cationic groups and/or groups that can be ionized to form cationic groups, and not comprising anionic groups and/or groups that can be ionized to anionic groups.
[0195] In some embodiments, the cationic polysaccharide is selected from cationic cellulose derivatives, cationic galactomannan gums, or mixtures thereof, preferably is selected from cationic cellulose derivatives comprising quaternary ammonium groups, cationic cellulose copolymers or cellulose derivatives grafted with a water-soluble quaternary ammonium monomer, cationic galactomannan gums, or combinations thereof.
[0196] Examples of cellulose derivatives comprising quaternary ammonium groups are especially described in French patent 1 492597, and mention may be made of the polymers sold under the name UCARE POLYMER “JR” (JR
400 LT, JR 125 and JR 30M) or “LR” (LR 400 or LR 30M) by the company Dow chemical. These polymers are also defined in the CTFA dictionary as quaternary ammoniums of hydroxy ethyl cellulose that have reacted with an epoxide substituted with a trimethylammonium group. The polyquaternium-10 is, for example, one of these polymers.
[0197] The cationic cellulose copolymers or cellulose derivatives grafted with a water-soluble quaternary ammonium monomer are described especially in US patent 4 131 576, and mention may be made of hydroxyalkyl celluloses, for instance hydroxymethyl-, hydroxyethyl- or hydroxypropyl celluloses grafted, in particular, with a methacryloyl ethyl trimethylammonium, methacrylamidopropyl trimethyl ammonium or dimethyldiallyl ammonium salt. The commercial products corresponding to this definition are more particularly the products sold under the names CELQUAT L 200 and CELQUAT H 100 by the company National Starch.
[0198] The cationic galactomannan gums are described more particularly in US patents 3 589 578 and 4 031 307, and mention may be made of guar gums comprising cationic trialkylammonium groups, preferably Ci-Ce trialkylammonium groups. Use is made, for example, of guar gums modified with a 2,3-epoxypropyltrimethylammonium salt (for example, chloride). Such products are sold under the names JAGUAR C13 S, JAGUAR C 15, JAGUAR C 17 or JAGUAR Cl 62 by the company Rhodia.
[0199] In some embodiments, the cationic polysaccharides are selected from cationic cellulose derivatives comprising quaternary ammonium groups, cationic galactomannan gums comprising cationic trialkylammonium groups, or combinations thereof.
[0200] Preferably, the cationic polysaccharides are selected from cationic galactomannan gums comprising cationic trialkylammonium groups, and better from guar gums comprising cationic trialkylammonium groups. More preferably, the cationic polysaccharides are selected from polymers with INCI names, such as guar hydroxypropyltrimonium chloride, hydroxypropyl guar
hydroxypropyltrimonium chloride, or their mixtures, better is guar hydroxypropyltrimonium chloride.
[0201] The cationic polysaccharide(s), according to embodiments herein, is(are) in a total amount ranging from 0.01% to 8% by weight, preferably from 0.05% to 5% by weight, more preferably from 0.1% to 4%, and better from 0.2% to 3% by weight, relative to the total weight of the composition.
[0202] Preferably, the cationic polysaccharide(s) is(are) selected from cationic galactomannan gums comprising cationic trialkylammonium groups and is(are) in a total amount ranging from 0.01% to 8% by weight, preferably from 0.05% to 5% by weight, more preferably from 0.1% to 4%, and better from 0.2% to 3% by weight, relative to the total weight of the composition.
[0203] More preferably, the cationic polysaccharide(s) is(are) selected from guar gums comprising cationic trialkylammonium groups and is(are) in a total amount ranging from 0.01% to 8% by weight, preferably from 0.05% to 5% by weight, more preferably from 0.1% to 4%, and better from 0.2% to 3% by weight, relative to the total weight of the composition.
Chelating Agents
[0204] The composition, according to the invention, may comprise one or more chelating agents.
[0205] Preferably, the composition, according to the invention, comprises one or more chelating agents.
[0206] In some embodiments, the chelating agents are selected from at least one carboxylic acid of formula (V) below, its optical isomers, geometric isomers, salts, solvates, or combinations thereof:
Ra-N-(CH(Rb)COOH)2
(V) in which:
Ra represents hydrogen atom, or a -CH(COOH)-(CH2)2-COOH, -CH2CH2OH, -CH(CH3)COOH, -(CH2)2N(CORC)-CH2-COOH, or -CH(COOH)- CH2-COOH group; and
- Rb represents a CH2COOH group when Ra represents a hydrogen atom, or Rb represents a hydrogen atom when Ra is other than a hydrogen atom; and
- Rc represents a linear or branched alkyl group comprising from 1 to 14 carbon atoms, preferably 1 to 4 carbon atoms, or cyclic alkyl group comprising from 3 to 30 carbon atoms.
[0207] Preferably, the carboxylic acids of formula (V) correspond to:
- compounds comprising four carboxylic acid functions, when Ra represents a hydrogen atom and Rb represents a -CH2-COOH group, or when Ra represents the -CH(COOH)-(CH2)2-COOH group and Rb represents a hydrogen atom, or when Ri represents the -CH(COOH)-CH2-COOH group and Rb represents a hydrogen atom;
- compounds comprising three carboxylic acid functions, when Ra represents the - CH(CH3)-COOH group and Rb represents a hydrogen atom, or when Ra represents a -(CH2)2-N(CORC)-CH2-COOH group and Rb represents a hydrogen atom; and to
- compounds comprising two carboxylic acid functions, when Ri represents the - CH2CH2OH group and Rb represents a hydrogen atom.
[0208] More preferably, according to embodiments herein, the carboxylic acids of formula (V) correspond to compounds comprising four carboxylic acid functions when Ra represents the -CH(COOH)-(CH2)2-COOH group and Rb represents a hydrogen atom.
[0209] The carboxylic acids of formula (V), according to the present invention, may take the form of pure enantiomers, preferably in L configuration, or the form of mixtures, or the form of racemic mixtures.
[0210] In an embodiment, the salts of the one or more carboxylic acids of formula (V) are selected from alkali metal salts, alkaline earth metal salts, transition metal salts, organic amine salts, ammonium salts, or combinations thereof.
[0211] Examples of alkali metal salts include especially sodium (Na+) and potassium (K+) salts, whereas examples of alkaline earth metal salts include especially calcium (Ca2+) and magnesium (Mg2+) salts.
[0212] For the purposes of the present invention, a “transition metal” is a metal comprising an incomplete d subshell, and more particularly in the II oxidation state, such as cobalt (Co2+), iron (Fe2+), manganese (Mn2+), zinc (Zn2+) and copper (Cu2+).
[0213] The organic amine salts, according to the present invention, may be made of the salts of primary, secondary, or tertiary amines, or alternatively of alkanolamines. The amines exhibit one or more identical or non-identical radicals, of linear or branched Ci to C20 alkyl type, optionally comprising a heteroatom such as oxygen.
[0214] Preferably, the carboxylic acids of formula (V), according to embodiments herein, are selected from methylglycinediacetic acid, N- lauroylethylenediamine-N,N',N'-triacetic acid, N,N-dicarboxymethylglutamic acid, iminodisuccinic acid, N,N-bis(carboxymethyl)aspartic acid, alkali metal salts thereof, alkaline earth metal salts thereof, transition metal salts thereof, organic amine salts thereof, ammonium salts thereof, optical isomers thereof, geometric isomers thereof, solvates thereof, or combinations thereof.
[0215] Preferably, in some embodiments, the chelating agents are selected from N,N-dicarboxymethylglutamic acid, N,N-bis(carboxymethyl)aspartic acid, alkali metal salts thereof, alkaline earth metal salts thereof, transition metal salts thereof, organic amine salts thereof, ammonium salts thereof, optical isomers thereof, geometric isomers thereof, solvates thereof, or combinations thereof.
[0216] Preferably, in some embodiments, the chelating agents are selected from N,N-dicarboxymethyl-L-glutamic acid (GLDA), N,N-bis(carboxymethyl)-L-
aspartic acid, alkali metal salts thereof, alkaline earth metal salts thereof, transition metal salts thereof, organic amine salts thereof, ammonium salts thereof, solvates thereof, or combinations thereof, and more preferably is N,N-dicarboxymethyl-L- glutamic acid (GLDA), and/or tetrasodium N,N-bis(carboxymethyl)-L-glutamate.
[0217] N,N-dicarboxymethyl-L-glutamic acid, tetrasodium N,N- bis(carboxymethyl)-L-glutamate and N,N-bis(carboxymethyl)aspartic acid are respectively represented by compounds (II), (III) and (IV) below:
[0218] The chelating agent(s), according to embodiments herein, is (are) in a total amount ranging from 0.01% to 5% by weight, preferably in an amount ranging from 0.05% to 3% by weight, and more preferably from 0.1% to 2% by weight, relative to the total weight of the composition.
[0219] Preferably, the chelating agents are selected from N,N- dicarboxymethyl-L-glutamic acid (GLDA), N,N-bis(carboxymethyl)-L-aspartic acid, alkali metal salts thereof, alkaline earth metal salts thereof, transition metal salts thereof, organic amine salts thereof, ammonium salts thereof, solvates thereof, or combinations thereof, and is (are) in a total amount ranging from 0.01% to 5% by weight, preferably in an amount ranging from 0.05% to 3% by weight, and more preferably from 0.1% to 2% by weight, relative to the total weight of the composition.
Polyols
[0220] The composition, according to the invention, may comprise one or more polyols.
[0221] Preferably the composition according to the invention, comprises one or more polyols.
[0222] For the purposes of the present invention, the term “polyol” refers to an organic compound constituted of a hydrocarbon-based chain optionally interrupted with one or more oxygen atoms and bearing at least two free hydroxyl groups (-OH) borne by different carbon atoms, this compound possibly being cyclic or acyclic, linear or branched, and saturated or unsaturated.
[0223] The polyols comprise from 2 to 30 hydroxyl groups, preferably from 2 to 10 hydroxyl groups, more preferably from 2 to 3 hydroxyl groups, and even more preferably comprises 2 hydroxyl groups.
[0224] In some embodiments, the polyols are selected from diols, preferably from C3-C6 diols.
[0225] In some embodiments, the polyols, are selected from propylene glycol, propane- 1,3-diol, 1,3-butylene glycol, pentane- 1,2-diol, dipropylene glycol, hexylene glycol, ethylene glycol, or mixtures thereof, preferably from propylene glycol, propane- 1,3-diol or mixtures thereof.
[0226] Preferably, the polyols are selected from propylene glycol, propane-
1.3-diol, 1,3-butylene glycol, pentane- 1,2-diol, dipropylene glycol, hexylene glycol, ethylene glycol, or mixtures thereof, better from propylene glycol, propane-
1.3-diol or mixtures thereof. More preferably the polyol is propylene glycol.
[0227] The polyol(s), according to embodiments herein, is(are) in a total amount ranging from 0.5% to 15% by weight, preferably from 1% to 10% by weight, and more preferably 2% to 8% by weight, relative to the total weight of the composition.
[0228] Preferably, the polyol(s) is(are) selected from C3-C6 diols and is(are) in a total amount ranging from 0.5% to 15% by weight, preferably from 1% to 10% by weight, and more preferably 2% to 8% by weight, relative to the total weight of the composition.
[0229] Preferably, the polyol(s) is(are) selected from propylene glycol, propane- 1,3 diol, or their mixtures and is(are) in a total amount ranging from 0.5% to 15% by weight, preferably from 1% to 10% by weight, and more preferably 2% to 8% by weight, relative to the total weight of the composition.
Additives
[0230] The composition according to the present invention optionally comprises one or more additives, different from the compounds previously described and among which mention may be made of mineral thickening agents, solvents other than polyols, antidandruff agents, anti-seborrheic agents, agents for preventing hair loss and/or for promoting hair regrowth, vitamins and provitamins, sunscreens, mineral or organic pigments, plasticizers, solubilizers, opacifiers or pearlescent agents, antioxidants, hydroxy acids, fragrances, and preservatives.
[0231] Each of the additives, in various embodiments herein, may be present in an amount ranging from 0.01% and 20% by weight, relative to the total weight of the composition.
[0232] The solvents other than the polyols mentioned herein of the composition, according to the present invention, may comprise water and/or one or more organic solvents.
[0233] In an embodiment, the organic solvents are selected from linear or branched and preferably saturated monoalcohols, comprising 2 to 10 carbon atoms, such as ethanol, isopropanol, aromatic alcohols such as benzyl alcohol or phenylethyl alcohol, polyol ethers, for instance ethylene glycol monomethyl, monoethyl or monobutyl ether, propylene glycol ethers thereof, for instance propylene glycol monomethyl ether, and also diethylene glycol alkyl ethers,
especially C1-C4 alkyl ethers, for instance diethylene glycol monoethyl ether or monobutyl ether, alone or as a mixture.
[0234] The organic solvents, according to embodiments herein, when present are in an amount ranging from 0.1% to 20% by weight, preferably ranging from 0.4% to 10% by weight, relative to the total weight of the composition.
[0235] The composition, according to the present invention, is preferably aqueous. In an embodiment, water in the composition, according to embodiments herein, is in an amount ranging from 30% to 99% by weight, preferably in an amount ranging from 40% to 95% by weight, and more preferably in an amount ranging from 45% to 90% by weight, relative to the total weight of the composition.
[0236] When the composition comprises water, the pH preferably ranges from 8 to 12, more preferably from 9 to 11.
[0237] In an embodiment, the composition of the invention may comprise at least one oxidizing agent as disclosed below. According to this embodiment, the composition of the invention is a ready to use composition comprising at least one oxidizing agent.
[0238] In another embodiment, the composition of the invention is free of oxidizing agent. According to this embodiment, the composition is preferably mixed with a composition comprising at least one oxidizing agent as disclosed below immediately before use.
Kit
[0239] Embodiments provided herein also include a kit comprising a composition (A) as described in the embodiments herein above at least one glycerolated nonionic surfactant; at least one sugar-based nonionic surfactant; at least one fatty acid; and at least one additional fatty compound different from the glycerolated nonionic surfactant(s), sugar-based nonionic surfactant(s), and fatty acid(s), and a composition (B) comprising at least one oxidizing agent. The compositions (A) and (B) are intended to be mixed together at the time of use to
obtain a ready to use composition which is then applied to keratin fibres for dyeing and/or lightening of keratin fibres. Preferably, the pH of the ready-to-use composition is between 8 and 11, preferably between 9.0 and 10.5.
[0240] The compositions (A) and (B) may be present in separate sachets or containers or in a single dual compartment sachet, optionally accompanied with suitable one or more applicators which may be identical or different, such as fine brushes, coarse brushes or sponges. The kit mentioned above may also be equipped with a means for dispensing the desired mixture onto the hair, for instance the devices described in patent FR 2586913.
Oxidizing agent
[0241] Embodiments of composition (B) of the kit provided herein include at least one oxidizing agent.
[0242] The oxidizing agents may be selected from, hydrogen peroxide, urea peroxide, alkali metal bromates or ferricyanides, peroxygenated salts, such as persulfates, perborates, peracids and/or precursors thereof, or percarbonates of alkali metals or alkaline-earth metals.
[0243] In some embodiments, the oxidizing agent is hydrogen peroxide.
[0244] The oxidizing agent(s), according to embodiments herein, is(are) in a total amount ranging from 0.1% to 40% by weight, and preferably from 0.5% to 20% by weight, relative to the total weight of the composition (B).
Method
[0245] The composition of the present invention may be manufactured using known methods that are generally used in the cosmetics or dermatological field. Typically, the method for preparation of the composition comprises blending the glycerolated nonionic surfactant(s); sugar-based nonionic surfactant(s); fatty acid(s); and at least one additional fatty compound different from the glycerolated nonionic surfactant(s), sugar-based nonionic surfactant(s), and fatty acid(s), with a suitable solvent(s), and optionally the additive(s) to obtain the composition.
[0246] Embodiments herein also include methods for manufacturing the composition (A) and composition (B) of the kit provided herein. In an embodiment, the method of preparation of the composition (A) is according to the method described herein above comprising blending the glycerolated nonionic surfactant(s); sugar-based nonionic surfactant(s); fatty acid(s); additional fatty compound(s) different from the glycerolated nonionic surfactant(s), sugar-based nonionic surfactant(s), and fatty acid(s), with suitable solvent(s), and optionally the additive(s) to obtain the composition (A). The method of preparation of the composition (B) comprises blending oxidizing agent with suitable solvent(s) and optionally, comprising the additive(s) to obtain the composition (B).
[0247] Embodiments herein further include a method for dyeing of keratin fibres. The method includes dyeing and/or lightening of keratin fibres, particularly hair. In an embodiment, the method comprises mixing the composition (A) and the composition (B) of the kit as provided herein to obtain a mixture and applying the mixture on said fibre. In some embodiments, the mixture is allowed to stand for a pause time in a range of 5 to 45 minutes. In some embodiments, the mixture is allowed to stand for a pause time in a range of 20 to 40 minutes.
[0248] The temperature during the method is conventionally between room temperature (from 25 °C to 30°C) and 80°C and preferably between room temperature and 60°C.
Use
[0249] Embodiments herein include the use of the composition and/or kit of the present invention.
[0250] The composition and/or the kit, according to embodiments herein, may be used for dyeing and/or lightening or colour keratin fibres, particularly hair. The composition may be used for the purpose of colour change, colour lightening/bleaching of hair and/or coverage of greying hair.
[0251] The composition and/or kit may be used on natural hair and/or artificial hair, including hair extensions.
[0252] The composition and/or kit may be used at different temperatures, including conventionally at room temperature (between 25°C to 30°C) and 80°C and preferably between room temperature and 60 °C.
[0253] Although the subject matter has been described in considerable detail with reference to certain examples and implementations thereof, it is understood that other implementations are possible and included within the scope of the present invention.
Examples
[0254] The disclosure will now be illustrated with working examples, which is intended to illustrate the working of disclosure and not intended to take restrictively to imply any limitations on the scope of the present disclosure. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood to one of ordinary skill in the art to which this disclosure belongs. Although methods and materials similar or equivalent to those described herein can be used in the practice of the disclosed methods and compositions, the exemplary methods, devices, and materials are described herein. It is to be understood that this disclosure is not limited to particular methods, and experimental conditions described, as such methods and conditions may apply.
Example 1: Composition for dyeing/lightening keratin fibres
[0255] Composition Al (according to the present invention) for dyeing and/or lightening keratin fibres was prepared using alkaline agent viz. 7.72% (w/w) of ethanolamine, glycerolated nonionic surfactants viz. 2%(w/w) of glyceryl caprylate, 1% (w/w) of polyglyceryl 4-caprate, 0.6% (w/w) of glyceryl stearate, sugar-based nonionic surfactants viz. 1.4% (w/w) of coco glucoside, 0.4% (w/w) of sucrose stearate, fatty acids viz. 0.32% (w/w) stearic acid, 0.02% (w/w) of myristic acid, 0.26% (w/w) of palmitic acid, fatty compounds viz. 6% (w/w) of Helianthus Annuus (sunflower) seed oil, 18% (w/w) of cetearyl alcohol, polyols viz. 5% (w/w) of propylene glycol, cationic polysaccharides viz. 0.5% (w/w) of guar
hydroxypropyltrimonium chloride, reducer/antioxidant viz 0.25 (w/w) of ascorbic acid, 0.75 (w/w) of sodium metabisulfite, and water (qs up to 100 wt%).
[0256] Composition Cl (comparative composition) was prepared in the same manner as the composition Al however, the glycerolated nonionic surfactants and sugar-based nonionic surfactants, that are more natural in origin and environmentally friendly compounds, were replaced with the surfactant oleth-20 which is synthetic and not environmentally friendly.
[0257] Table 1 below provides the ingredients used to prepare the composition Al (according to the present invention), and comparative composition Cl, expressed in % by weight:
[0258] The compositions A2 to A8 (according to the invention) and C2 to C8 (comparative) were prepared in the same manner as the compositions Al and Cl, respectively, however different combinations of oxidative bases and couplers were used in place of the combination of toluene-2,5-diamine, and 2-amino-3- hydroxypyridine as provided in Table 1 above. In compositions A2 and C2, the combination of 0.96% (w/w) toluene-2,5-diamine, and 0.073% (w/w) hydroxybenzo morpholine were used as the oxidative base and coupler. In compositions A3 and C3, the combination of 0.25% (w/w) N,N-bis(2- hydroxyethyl)-p-phenylenediamine sulfate, and 0.073% (w/w) 2-amino-3- hydroxypyridine were used as the oxidative base and coupler. In compositions A4 and C4, the combination of 0.25% (w/w) N,N-bis(2-hydroxyethyl)-p- phenylenediamine sulfate, and 0.025% (w/w) 2,4-diamino phenoxyethanol HC1 were used as the oxidative base and coupler. In compositions A5 and C5, the combination of 0.96% (w/w) toluene-2,5-diamine, and 0.272% (w/w) m- aminophenol were used as the oxidative base and coupler. In compositions A6 and C6, the combination of 0.96% (w/w) toluene-2,5-diamine, and 0.025% (w/w) 2,4- diamino phenoxyethanol HC1 were used as the oxidative base and coupler. In compositions A7 and C7, the combination of 0.96% (w/w) Toluene-2,5-diamine, and 0.334% (w/w) hydroxyethyl-3,4-methylenedioxyaniline HC1 were used as the oxidative base and coupler. In compositions A8 and C8, the combination of 0.25% (w/w) N,N-bis(2-hydroxyethyl)-p-phenylenediamine sulfate, and 0.72% (w/w) hydroxybenzo morpholine were used as the oxidative base and coupler.
[0259] The oxidizing composition B was prepared from the ingredients of which the contents are indicated in Table 2 below, expressed in % by weight:
[0260] Protocol: Each of the compositions Al to A8 (according to the invention) and Cl to C8(comparative) were independently mixed with the oxidizing composition B in a 1: 1 weight ratio. Each of the mixtures was applied to locks of hair containing 90% natural white hair, in a proportion of 5 g of mixture per 1 g of hair.
[0261] After a leave-on time of 30 minutes on a hot plate at 27°C, the hair was rinsed, washed and dried.
[0262] Results: The colouring of the hair was evaluated in the L*a*b* system, using a Konica Minolta CM-3600A spectrocolorimeter (illuminant D65, angle 10°, specular component included) in the CIELab system.
[0263] In this system, L* represents the lightness. The lower the value of L*, the darker and more powerful (intense) the colouring obtained. The chromaticity is measured by the values a* and b*, a* representing the red/green axis and b* the yellow/blue axis.
[0264] The results for the Compositions Al to A8 and Cl to C8 are indicated in Table 3 below:
[0265] Compositions Al to A8 (according to the invention) had a lower L* value, with significant AL values (AL >2) and thus showed better colour strength and colour uptake, compared to the comparative compositions Cl to C8.
ADVANTAGES OF THE PRESENT DISCLOSURE
[0266] The composition of the present disclosure achieves superior colour performances, in particular demonstrating a more intense colour with long lasting colour uptake. The composition is environmentally friendly as it incorporates ingredients that are natural and/or are natural in origin, with a better carbon footprint and reliance on more renewable materials, thereby reducing the negative impact on environment. In addition, the composition contributes to the superior properties of smoother and more nourished hair, along with better shine.
Claims
1. A composition comprising: a) at least one glycerolated nonionic surfactant; b) at least one sugar-based nonionic surfactant; c) at least one fatty acid; and d) at least one additional fatty compound different from (a), (b) and (c).
2. The composition as claimed in claim 1, wherein the glycerolated nonionic surfactant(s) is(are) selected from monoesters or polyesters of linear or branched, Cs to C40 acids, preferably Cs to C32 acids, more preferably Cs to C28 acids and glycerol or polyglycerol, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20, or even from 1 to 10 mol of glycerol, or combinations thereof.
3. The composition as claimed in claim 1, comprising at least two different glycerolated nonionic surfactants selected from monoesters or polyesters of linear or branched, Cs to C40 acids, preferably Cs to C32 acids, more preferably Cs to C28 acids and glycerol or polyglycerol, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20, or even from 1 to 10 mol of glycerol, or combinations thereof.
4. The composition as claimed in any one of the preceding claims, wherein the glycerolated nonionic surfactant(s) is(are) selected from monoesters of linear or branched, preferably linear, Cs to C24 acids, more preferably Cs to C20 acids, better from Cs to Cis acids, comprising from 1 to 50 mol of glycerol, preferably from 1 to 20, or more preferably from 1 to 10 mol of glycerol.
5. The composition as claimed in any one of the preceding claims, wherein the glycerolated nonionic surfactant(s) is(are) selected from a lauric acid monoester containing 4 mol of glycerol, a capric acid monoester comprising 4 mol of glycerol, a caprylic acid monoester comprising 1 mol of glycerol, a stearic acid monoester comprising 1 mol of glycerol, or combinations thereof.
6. The composition as claimed in any one of the preceding claims, wherein the glycerolated nonionic surfactant(s) is(are) in a total amount ranging from 0.1% to 10% by weight, preferably from 0.2% to 8% by weight, and more preferably from 0.5% to 5% by weight, better still from 1% to 5% by weight, relative to the total weight of the composition.
7. The composition as claimed in any one of the preceding claims, wherein the sugar-based nonionic surfactant(s) is(are) selected from alkylpolyglucosides, sucrose fatty esters, sorbitan fatty esters, or combinations thereof.
8. The composition as claimed in any one of the preceding claims, comprising at least two different sugar-based nonionic surfactant(s) selected from alkylpoly gluco sides, sucrose fatty esters, sorbitan fatty esters, or combinations thereof.
9. The composition as claimed in claim 7 or 8, wherein the alkylpolyglucoside(s) is(are) selected from decyl glucoside, stearyl glucoside, lauryl glucoside, coco- glucoside, cetearyl glucoside, decyl lauryl glucoside, or combinations thereof, the sucrose fatty ester(s) is(are) selected from sucrose cocoate, sucrose laurate, sucrose myristate, sucrose palmitate, sucrose polystearate, sucrose tristearate, sucrose distearate, sucrose stearate, sucrose dilaurate, sucrose hexaerucate, sucrose oleate, sucrose pentaerucate, sucrose polybehenate, sucrose polycottonseedate, sucrose polylaurate, sucrose polylinoleate, sucrose
polypalmate, sucrose polyoleate, sucrose polysoyate, sucrose ricinoleate, sucrose tetraisostearate, sucrose tribehenate, sucrose hexaoleate, sucrose hexastearate, sucrose hexapalmitate, sucrose trilaurate, or combinations thereof, and the sorbitan fatty ester(s) is(are) selected from sorbitan monostearate, sorbitan tristearate, sorbitan monolaurate, sorbitan monooleate, sorbitan palmitate, or combinations thereof.
10. The composition as claimed in any one of the preceding claims, wherein the sugar-based nonionic surfactant(s) is(are) selected from decyl glucoside, stearyl glucoside, lauryl glucoside, coco-glucoside, cetearyl glucoside, sucrose oleate, sucrose stearate, sucrose distearate, sucrose tristearate, or combinations thereof, preferably from coco-glucoside, sucrose oleate, sucrose stearate, sucrose distearate, sucrose tristearate, or combinations thereof, and more preferably from coco-glucoside, sucrose stearate, or combinations thereof.
11. The composition as claimed in any one of the preceding claims, wherein the sugar-based nonionic surfactant(s) is(are) in a total amount ranging from 0.1% to 20% by weight, preferably from 0.5% to 10% by weight, and more preferably from 1% to 5% by weight, relative to the total weight of the composition.
12. The composition as claimed in any one of the preceding claims, wherein the fatty acid(s) is(are) selected from at least one carboxylic acid comprising 6 to 40 carbon atoms, preferably from the carboxylic acid comprising 10 to 30 carbon atoms, more preferably from lauric acid, myristic acid, stearic acid, palmitic acid, or combinations thereof, and better from myristic acid, stearic acid, palmitic acid, or combinations thereof.
13. The composition as claimed in any one of the preceding claims, wherein the fatty acid(s) is(are) in a total amount ranging from 0.1% to 15% by weight, preferably
from 0.2% to 10% by weight, more preferably from 0.4% to 5% by weight, and better from 0.5% to 3% by weight, relative to the total weight of the composition.
14. The composition as claimed in any one of the preceding claims, wherein the additional fatty compound(s) is(are) selected from liquid fatty compound, solid fatty compounds, or combinations thereof, preferably from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, liquid fatty alcohols, liquid fatty esters, silicone oils, solid fatty alcohols, solid fatty esters, waxes, ceramides, or combinations thereof, and more preferably from liquid hydrocarbons containing more than 16 carbon atoms, plant oils, liquid fatty esters, solid fatty alcohols, solid fatty esters, or combinations thereof.
15. The composition as claimed in any one of the preceding claims, wherein the additional fatty compound(s) is(are) in a total amount ranging from 5% to 45% by weight, preferably 10% to 40% by weight, and more preferably 15% to 35% by weight, better still from 20% to 32% by weight, relative to the total weight of the composition.
16. The composition as claimed in any one of the preceding claims, wherein the composition comprises at least one alkaline agent, preferably selected from alkanolamines, ammonium hydroxide, metal carbonates, metal bicarbonates, alkali metal silicates, alkaline-earth metal silicates, alkali metal metasilicates, alkaline-earth metal metasilicates, or combinations thereof, more preferably alkanolamines, ammonium hydroxide, or combinations thereof, even more preferably alkanolamines, and better is monoethanolamine.
17. The composition as claimed in any one of the preceding claims, wherein the alkaline agent(s) is(are) in a total amount ranging from 0.1% to 40% by weight, preferably from 1% to 30% by weight, more preferably from 2% to 25% by
weight, and better from 4% to 20% by weight, relative to the total weight of the composition.
18. The composition as claimed in any one of the preceding claims, wherein the composition comprises at least one oxidative dye, preferably comprises at least one oxidation base and/or at least one oxidation coupler: the oxidation base being preferably selected from a group consisting of para-phenylenediamines, bis(phenyl)alkylenediamines, para-aminophenols, ortho-aminophenols, heterocyclic bases, and their addition salts; and the oxidation coupler being preferably selected from a group consisting of meta-phenylenediamines, meta-aminophenol, meta-diphenols, naphthalene- based couplers, heterocyclic couplers, and their addition salts, the amount of each of which is in a weight range of 0.0001% to 10%, relative to the total weight of the composition.
19. A kit comprising:
(a) a composition (A) as claimed in any one of the preceding claims; and
(b) a composition (B) comprising at least one oxidizing agent, a mixture of composition (A) and composition (B) resulting in a ready to use composition.
20. The kit as claimed in claim 19, wherein the oxidizing agent is in an amount ranging from 0.1% to 40% by weight, and preferably from 0.5% to 20% by weight, relative to the total weight of the composition (B).
21. A method for dyeing and/or lightening keratin fibres, preferably hair, comprising:
mixing the composition (A) as claimed in any one of claims 1 to 18 and the composition (B) comprising at least one oxidizing agent to obtain a mixture; and applying the mixture on said fibres.
22. Use of the composition as claimed in claims 1 to 18 or the kit as claimed in claims 19 to 20 for dyeing and/or lightening keratin fibres, such as hair.
Applications Claiming Priority (4)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| IN202321085967 | 2023-12-15 | ||
| IN202321085967 | 2023-12-15 | ||
| FR2401902A FR3159513A3 (en) | 2024-02-27 | 2024-02-27 | COMPOSITION FOR DYEING/LIGHTENING KERATIN FIBERS, AND RELATED METHODS |
| FRFR2401902 | 2024-02-27 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025126227A1 true WO2025126227A1 (en) | 2025-06-19 |
Family
ID=93379218
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/IN2024/052064 Pending WO2025126227A1 (en) | 2023-12-15 | 2024-10-14 | A composition for dyeing /lightening of keratin fibres, and methods thereof |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025126227A1 (en) |
Citations (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1026978A (en) | 1962-03-30 | 1966-04-20 | Schwarzkopf Verwaltung G M B H | Method of dyeing hair |
| FR1492597A (en) | 1965-09-14 | 1967-08-18 | Union Carbide Corp | New cellulose ethers containing quaternary nitrogen |
| GB1153196A (en) | 1965-07-07 | 1969-05-29 | Schwarzkopf Verwaltung G M B H | Method of Dyeing Hair |
| US3589578A (en) | 1968-01-20 | 1971-06-29 | Monforts Fa A | Tension-relieving device for stretchable sheet material |
| DE2359399A1 (en) | 1973-11-29 | 1975-06-12 | Henkel & Cie Gmbh | Tetraaminopyrimidines as developers in oxidation hair dyes - esp. used with meta aminophenol couplers for blue shading dyes |
| US4031307A (en) | 1976-05-03 | 1977-06-21 | Celanese Corporation | Cationic polygalactomannan compositions |
| US4131576A (en) | 1977-12-15 | 1978-12-26 | National Starch And Chemical Corporation | Process for the preparation of graft copolymers of a water soluble monomer and polysaccharide employing a two-phase reaction system |
| FR2586913A1 (en) | 1985-09-10 | 1987-03-13 | Oreal | PROCESS FOR FORMING IN IN SITU A COMPOSITION CONSISTING OF TWO PARTS CONDITIONED SEPARATELY AND DISPENSING ASSEMBLY FOR IMPLEMENTING SAID METHOD |
| DE3843892A1 (en) | 1988-12-24 | 1990-06-28 | Wella Ag | OXIDATION HAIR AGENTS CONTAINING DIAMINOPYRAZOL DERIVATIVES AND NEW DIAMINOPYRAZOLE DERIVATIVES |
| JPH0563124A (en) | 1991-09-03 | 1993-03-12 | Mitsubishi Electric Corp | Hybrid integrated circuit device |
| DE4133957A1 (en) | 1991-10-14 | 1993-04-15 | Wella Ag | HAIR DYE CONTAINING AMINOPYRAZOLE DERIVATIVES AND NEW PYRAZOLE DERIVATIVES |
| WO1994008970A1 (en) | 1992-10-16 | 1994-04-28 | Wella Aktiengesellschaft | Oxidation hair dye containing 4,5-diaminopyrazole derivatives and novel 4,5-diaminopyrazole derivatives and process for their production |
| WO1994008969A1 (en) | 1992-10-16 | 1994-04-28 | Wella Aktiengesellschaft | Process for producing 4,5-diamino pyrazole derivatives, their use for colouring hair and novel pyrazole derivatives |
| WO1996015765A1 (en) | 1994-11-17 | 1996-05-30 | Henkel Kommanditgesellschaft Auf Aktien | Oxidation dyes |
| FR2733749A1 (en) | 1995-05-05 | 1996-11-08 | Oreal | COMPOSITIONS FOR DYEING KERATINIC FIBERS CONTAINING DIAMINO PYRAZOLES, DYEING PROCESS, NOVEL DIAMINO PYRAZOLES, AND PREPARATION METHOD THEREOF |
| EP0770375A1 (en) | 1995-10-21 | 1997-05-02 | GOLDWELL GmbH | Hair dyeing composition |
| DE19543988A1 (en) | 1995-11-25 | 1997-05-28 | Wella Ag | Oxidative hair dye composition |
| EP0955037A1 (en) * | 1998-03-30 | 1999-11-10 | Cognis Deutschland GmbH | Skin care compositions |
| FR2801308A1 (en) | 1999-11-19 | 2001-05-25 | Oreal | KERATIN FIBER DYEING COMPOSITIONS CONTAINING 3-AMINO PYRAZOLO- [1, (- a] -PYRIDINES, DYEING PROCESS, NOVEL 3-AMINO PYRAZOLO- [1,5-a] -PYRIDINES |
| JP2013169571A (en) | 2012-02-21 | 2013-09-02 | Nippon Steel & Sumitomo Metal Corp | Method of manufacturing forged steel roll |
| US20170360662A1 (en) * | 2016-06-15 | 2017-12-21 | Zotos International, Inc. | Cosmetic composition capable of simultaneously coloring hair and relaxing curls |
| US20210085584A1 (en) * | 2016-12-05 | 2021-03-25 | Zotos International, Inc. | Composition For Providing High Lift And Color Deposit |
| WO2023275205A1 (en) * | 2021-06-30 | 2023-01-05 | L'oreal | Cosmetic composition comprising at least one alkyl (poly)glycoside, n,n-dicarboxymethylglutamic acid, propane-1,3-diol, at least one fatty substance other than fatty acids, at least one dye |
-
2024
- 2024-10-14 WO PCT/IN2024/052064 patent/WO2025126227A1/en active Pending
Patent Citations (23)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| GB1026978A (en) | 1962-03-30 | 1966-04-20 | Schwarzkopf Verwaltung G M B H | Method of dyeing hair |
| GB1153196A (en) | 1965-07-07 | 1969-05-29 | Schwarzkopf Verwaltung G M B H | Method of Dyeing Hair |
| FR1492597A (en) | 1965-09-14 | 1967-08-18 | Union Carbide Corp | New cellulose ethers containing quaternary nitrogen |
| US3589578A (en) | 1968-01-20 | 1971-06-29 | Monforts Fa A | Tension-relieving device for stretchable sheet material |
| DE2359399A1 (en) | 1973-11-29 | 1975-06-12 | Henkel & Cie Gmbh | Tetraaminopyrimidines as developers in oxidation hair dyes - esp. used with meta aminophenol couplers for blue shading dyes |
| US4031307A (en) | 1976-05-03 | 1977-06-21 | Celanese Corporation | Cationic polygalactomannan compositions |
| US4131576A (en) | 1977-12-15 | 1978-12-26 | National Starch And Chemical Corporation | Process for the preparation of graft copolymers of a water soluble monomer and polysaccharide employing a two-phase reaction system |
| FR2586913A1 (en) | 1985-09-10 | 1987-03-13 | Oreal | PROCESS FOR FORMING IN IN SITU A COMPOSITION CONSISTING OF TWO PARTS CONDITIONED SEPARATELY AND DISPENSING ASSEMBLY FOR IMPLEMENTING SAID METHOD |
| DE3843892A1 (en) | 1988-12-24 | 1990-06-28 | Wella Ag | OXIDATION HAIR AGENTS CONTAINING DIAMINOPYRAZOL DERIVATIVES AND NEW DIAMINOPYRAZOLE DERIVATIVES |
| JPH0563124A (en) | 1991-09-03 | 1993-03-12 | Mitsubishi Electric Corp | Hybrid integrated circuit device |
| DE4133957A1 (en) | 1991-10-14 | 1993-04-15 | Wella Ag | HAIR DYE CONTAINING AMINOPYRAZOLE DERIVATIVES AND NEW PYRAZOLE DERIVATIVES |
| WO1994008970A1 (en) | 1992-10-16 | 1994-04-28 | Wella Aktiengesellschaft | Oxidation hair dye containing 4,5-diaminopyrazole derivatives and novel 4,5-diaminopyrazole derivatives and process for their production |
| WO1994008969A1 (en) | 1992-10-16 | 1994-04-28 | Wella Aktiengesellschaft | Process for producing 4,5-diamino pyrazole derivatives, their use for colouring hair and novel pyrazole derivatives |
| WO1996015765A1 (en) | 1994-11-17 | 1996-05-30 | Henkel Kommanditgesellschaft Auf Aktien | Oxidation dyes |
| FR2733749A1 (en) | 1995-05-05 | 1996-11-08 | Oreal | COMPOSITIONS FOR DYEING KERATINIC FIBERS CONTAINING DIAMINO PYRAZOLES, DYEING PROCESS, NOVEL DIAMINO PYRAZOLES, AND PREPARATION METHOD THEREOF |
| EP0770375A1 (en) | 1995-10-21 | 1997-05-02 | GOLDWELL GmbH | Hair dyeing composition |
| DE19543988A1 (en) | 1995-11-25 | 1997-05-28 | Wella Ag | Oxidative hair dye composition |
| EP0955037A1 (en) * | 1998-03-30 | 1999-11-10 | Cognis Deutschland GmbH | Skin care compositions |
| FR2801308A1 (en) | 1999-11-19 | 2001-05-25 | Oreal | KERATIN FIBER DYEING COMPOSITIONS CONTAINING 3-AMINO PYRAZOLO- [1, (- a] -PYRIDINES, DYEING PROCESS, NOVEL 3-AMINO PYRAZOLO- [1,5-a] -PYRIDINES |
| JP2013169571A (en) | 2012-02-21 | 2013-09-02 | Nippon Steel & Sumitomo Metal Corp | Method of manufacturing forged steel roll |
| US20170360662A1 (en) * | 2016-06-15 | 2017-12-21 | Zotos International, Inc. | Cosmetic composition capable of simultaneously coloring hair and relaxing curls |
| US20210085584A1 (en) * | 2016-12-05 | 2021-03-25 | Zotos International, Inc. | Composition For Providing High Lift And Color Deposit |
| WO2023275205A1 (en) * | 2021-06-30 | 2023-01-05 | L'oreal | Cosmetic composition comprising at least one alkyl (poly)glycoside, n,n-dicarboxymethylglutamic acid, propane-1,3-diol, at least one fatty substance other than fatty acids, at least one dye |
Non-Patent Citations (4)
| Title |
|---|
| A. THOMASSHEA BUTTERBORNEO TALLOW: "Ullmann's Encyclopedia of Industrial Chemistry", 15 June 2000, article "Fats and Fatty Oils" |
| DATABASE GNPD [online] MINTEL; 13 October 2023 (2023-10-13), ANONYMOUS: "Intense Care & Shine Permanent Colourant", XP093244370, retrieved from https://www.gnpd.com/sinatra/recordpage/11186070/ Database accession no. 11186070 * |
| DATABASE GNPD [online] MINTEL; 18 May 2022 (2022-05-18), ANONYMOUS: "Hair Color Cream", XP093244368, retrieved from https://www.gnpd.com/sinatra/recordpage/9579308/ Database accession no. 9579308 * |
| DATABASE GNPD [online] MINTEL; 2 May 2023 (2023-05-02), ANONYMOUS: "Permanent Crème Color", XP093244369, retrieved from https://www.gnpd.com/sinatra/recordpage/10770914/ Database accession no. 10770914 * |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| WO2023275205A1 (en) | Cosmetic composition comprising at least one alkyl (poly)glycoside, n,n-dicarboxymethylglutamic acid, propane-1,3-diol, at least one fatty substance other than fatty acids, at least one dye | |
| EP4444264A1 (en) | Composition comprising two particular oxidation dye precursors and a phosphoric surfactant | |
| EP4444258A1 (en) | Composition comprising a particular oxidation dye precursor, an oxyalkylenated fatty alcohol and a polysaccharide | |
| EP4362894A1 (en) | Composition comprising n,n-dicarboxymethylglutamic acid, at least one fatty alcohol, at least one fatty acid, at least one polyol, at least one alkaline agent and optionally at least one dye | |
| EP4362896A1 (en) | Composition comprising n,n-dicarboxymethylglutamic acid, 1,3-propanediol, at least one nonionic surfactant, at least one alkaline agent and/or at least one dye | |
| WO2025126227A1 (en) | A composition for dyeing /lightening of keratin fibres, and methods thereof | |
| EP4362891A1 (en) | Composition comprising at least one particular coupler, at least one particular oxidation base, at least one fatty substance and at least one anionic polysaccharide | |
| WO2025126228A1 (en) | A composition for dyeing /lightening of keratin fibres | |
| WO2023275208A1 (en) | Composition comprising at least nonionic surfactant, 1,3-propanediol, at least one fatty substance, at least one colouring agent | |
| EP4362906A1 (en) | Composition comprising shea, an alkyl(poly)glycoside, a polysaccharide and a dye | |
| WO2022129379A1 (en) | Composition comprising two particular oxidation dye precursors, and a particular carboxylic acid | |
| WO2025069042A1 (en) | Compositions and methods for dyeing and/or lightening keratin fibres | |
| WO2025168477A1 (en) | Composition comprising an oxidation dye, an alkaline agent, a chemical oxidizing agent, an anionic acrylic polymer, a fatty acid, a nonionic surfactant and a fatty substance | |
| FR3159514A3 (en) | KERATIN FIBERS DYEING/LIGHTENING COMPOSITION | |
| WO2024241333A1 (en) | Product containing a dye composition comprising a fatty acid, a non-ionic surfactant, a non-associative polymer, at least 10% by weight of one or more fatty substances, a polyol, two specific dye precursors and an oxidizing composition | |
| FR3159513A3 (en) | COMPOSITION FOR DYEING/LIGHTENING KERATIN FIBERS, AND RELATED METHODS | |
| WO2024261770A1 (en) | Product containing a dye composition comprising a fatty acid, a non-ionic surfactant, a non- associative polymer | |
| WO2024241332A1 (en) | Product containing a dye composition comprising a fatty acid, a non-ionic surfactant, a non-associative polymer, at least 10% by weight of one or more fatty substances, a polyol, a specific dye precursor and an oxidizing composition | |
| WO2025069043A1 (en) | Sustainable compositions and methods for dyeing and/or lightening keratin fibres | |
| WO2025003368A1 (en) | Composition comprising a particular polycarboxylic acid or a salt thereof, a (bi)carbonate in a particular content, an additional alkaline agent, a fatty substance and a dye | |
| WO2025003347A1 (en) | Composition comprising hyaluronic acid and/or a derivative thereof, an anionic surfactant, an alkaline agent, a dye, a solid fatty alcohol and a liquid fatty alcohol | |
| EP4362901A1 (en) | Composition comprising at least one particular oxidation dye, at least one shea-derived fatty substance and at least one alkaline agent | |
| WO2025003366A1 (en) | Composition comprising a particular coupler ammonium bicarbonate and fatty acid | |
| WO2025003345A1 (en) | Composition comprising a particular base ammonium bicarbonate and fatty acid | |
| FR3163845A1 (en) | Composition obtained by mixing at least one coloring composition comprising at least one fatty acid, at least one alkali and at least one oxidizing dye, and at least one oxidizing composition |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24801687 Country of ref document: EP Kind code of ref document: A1 |