Attorney Docket No.14640.0105-00304 COMPOSITIONS AND METHODS FOR THE TREATMENT OF DISORDERS RELATED TO SYNTAXIN-BINDING PROTEIN 1 DEFICIENCY RELATED APPLICATIONS [01] This application claims the benefit of and priority to US Provisional Application Serial No. 63/606,423, filed December 5, 2023, the contents of which are incorporated herein by reference in their entirety. SEQUENCE LISTING [02] The present application is being filed along with a Sequence Listing in electronic format. The Sequence Listing file, entitled 14640_0105-00304_SL.xml, was created on September 18, 2024, and is 2,039,437 bytes in size. The information in electronic format of the Sequence Listing is incorporated herein by reference in its entirety. FIELD [03] Described herein are compositions and methods relating to adeno-associated virus (AAV) viral particles for the delivery of polynucleotides, e.g., polynucleotides encoding syntaxin-binding protein-1 (STXBP1) proteins and peptides for use in the treatment of an STXBP1-related disorder such as an STXBP1-related neurodegenerative or neuromuscular disorder (e.g., e.g., STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5)). In some embodiments, compositions described herein may be used to treat a subject in need thereof, such as a human subject diagnosed with an STXBP1-related disorder or other condition resulting from a deficiency in the quantity and/or function of STXBP1 protein, or as a research tool in the study of diseases or conditions in cells or animal models of an STXBP1-related disorder. BACKGROUND [04] Syntaxin-binding protein 1 (STXBP1), also known as Munc18-1, is part of the synaptic fusion machinery that enables vesicles to fuse with the plasma membrane. Other names for STXBP1 include P67, DEE4, NSEC1, UNC18, N-Sec1, RBSEC1, unc-18A, and unc18-1. STXBP1 regulates neurotransmitter transmission by interacting with the SNARE complex. The SNARE complex is primarily composed of SNAP-25, vesicular associated membrane protein and syntaxin1. SNAP-25 and syntaxin-1 form the target membrane vesicle protein (T-SNARE), which binds to synaptic vesicle protein (VAMP). STXBP1 has a complex, arched tertiary structure. The arch comprises four closely connected domains, 1, 2, 3a, and 3b. Domains 1 and 3a form an arched gap. STXBP1 primarily regulates vesicle fusion by interacting with syntaxin-1. Domain 3a of STXBP1 is in close contact with the Habc domain of syntaxin-1, and domain 1 of STXBP1 binds to the N-terminal domain of syntaxin-1. STXBP1 regulates
Attorney Docket No.14640.0105-00304 vesicle docking and fusion by interacting with the SNARE complex. STXBP1 affects the released vesicles and participates in the transmission of neurotransmitters. STXBP1 is prominently involved in the early process of neurotransmitter release. [05] STXBP1 is essential for presynaptic vesicle release. It is rapidly phosphorylated by protein kinase C upon neuronal depolarization. [06] STXBP1 is encoded by the STXBP1 gene (Ensembl Gene ID No. ENSG00000136854), which is located on chromosome 9. It is expressed in the brain and spinal cord, and highly enriched in axons. Expression of STXBP1 is highest in the retina and cerebellum. STXBP1 is also found outside the brain. [07] Mutations in the STXBP1 gene are known to cause disease in human subjects. Abnormal expression of STXBP1 plays a role in the pathogenesis of a variety of neurological diseases, including STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), and Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). [08] STXBP1 expression is also abnormal in STXBP1 encephalopathy. There are an estimated 750 known cases of STXBP1 encephalopathy worldwide, and STXBP1 encephalopathy has an estimated incidence of 3.3-3.8 per 100,000 births. [09] Patients with STXBP1 mutations and/or STXBP1 encephalopathy often present with epilepsy. Some patients with STXBP1 mutations present with autistic features, including aggressive behavior, self- mutilation, hyperactivity, compulsive symptoms, episodes of psychosis and/or auditory hallucinations. Phenotypes range from severe neonatal epilepsy to infantile-onset epilepsy. [010] Many patients with STXBP1 mutations also present with non-epileptic movement disorders, including truncal and limb ataxia, generalized tremors, and dystonia. Patients often present with unremitting epileptic activity. [011] Patients with STXBP1 encephalopathy are reliant on caregivers for the duration of their lives. Moreover, 40% of patients become non-ambulatory and lifespan is expected to be significantly reduced to about 30 years. [012] STXBP1 encephalopathy is caused by haploinsufficiency. Thus, disease may occur where there is a mutation to only a single functional copy of the STXBP1 gene. Disease-causing mutations include missense, nonsense, frameshift, splice-site, and whole gene deletions. There are about 135 known pathogenic variants of the STXBP1 gene. [013] Interneurons may be more affected by haploinsufficiency than excitatory neurons. [014] Studies have demonstrated that heterozygous STXBP1 knock-out mice display impaired glutamate and GABA transmission, increased anxiety, increased aggression, and impaired emotional learning, in addition to modest seizure phenotype.
Attorney Docket No.14640.0105-00304 [015] Studies in mice demonstrated that normalizing the excitatory synaptic transmission in STXBP1 heterozygotic knockout mice reduces aggression. This indicated a therapeutic option for managing aggressiveness in patients with STXBP1 mutations. [016] Existing therapies target the symptoms of STXBP1 encephalopathy. Existing first line treatment comprises anti-epileptic drugs such as levetiracetam and phenobarbital. Existing second line treatment comprises further anti-epileptic drugs such as clobazam, topiramate. Existing third line treatment comprises further anti-epileptic drugs and/or interventions. Known interventions for existing third line treatment include adrenocorticotropic hormone, ketogenic diet, and vagal nerve stimulation. [017] No approved therapies target the underlying cause of STXBP1 mutations. There is a high unmet need for such treatments driven by the high seizure rate and shortened lifespan of patients with STXBP1 mutations. [018] Thus, there remains a long-felt need to develop pharmaceutical compositions and methods that can be delivered to the CNS for the treatment of diseases associated with STXBP1 mutations. In particular, a need exists for treatments targeting neurons, including GABAergic and glutamatergic neurons. [019] Adeno-associated viruses (AAVs) have emerged as a widely studied and utilized viral particles for delivery of therapeutically effective polypeptides to mammalian cells. See, e.g., Tratschin et al., Mol. Cell Biol., 5(11):3251-3260 (1985) and Grimm et al., Hum. Gene Ther., 10(15):2445-2450 (1999). [020] The present disclosure provides improved pharmaceutical compositions and methods. In some embodiments, the disclosure provides methods of treatment using AAV capsid variants that are capable of delivering a payload of interest, e.g., human STXBP1, to a target cell or tissue, e.g., a CNS cell or tissue. SUMMARY [021] The present disclosure addresses these challenges by providing AAV-based compositions, AAV-based compositions for use in methods for treating a syntaxin-binding protein-1 (STXBP1)-related disorder in subjects, and methods for treating a STXBP1-related disorder in subjects. Disclosed herein are compositions and methods directed to AAV-based gene delivery of STXBP1 to ameliorate loss-of- function. Disclosed herein are compositions and methods directed to AAV-based gene delivery of STXBP1 to improve neurotransmitter release. The compositions and methods are useful to improve presynaptic vesicle release, and to slow, halt, or reverse neurodegenerative and other symptoms of STXBP1 encephalopathy or a STXBP1-related disorder in a subject (e.g., a subject having one or more mutations in STXBP1). [022] In some aspects, the present disclosure provides an AAV particle comprising an AAV capsid and a nucleotide sequence encoding an STXBP1 protein, also referred to herein as an STXBP1-encoding sequence. In some embodiments, the nucleotide sequence encoding an STXBP1 protein is comprised in a viral genome. In some embodiments, the STXBP1 protein is a human STXBP1 protein. In some
Attorney Docket No.14640.0105-00304 embodiments, the STXBP1 protein is a wildtype human STXBP1 protein. In some embodiments, the AAV particle comprises a viral genome encoding the STXBP1 protein (e.g., a human STXBP1 protein) and the AAV capsid is an AAV capsid variant. In some embodiments, the AAV capsid variant is an AAV9 capsid variant. [023] In some aspects, the present disclosure provides an adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome, wherein the viral genome comprises a syntaxin- binding protein-1 (STXBP1)-encoding sequence and the AAV capsid variant comprises an amino acid sequence having the formula [N1]-[N2]-[N3] (SEQ ID NO: 4681) in loop VIII, wherein [N2] comprises the amino acid sequence of DWHR (SEQ ID NO: 4682); [N1] comprises amino acids X
1, X
2, X
3, and X
4, wherein X
4 is Q, K, E, S, P, R, N, or H; and/or [N3] comprises amino acids X
5, X
6, and X
7, wherein X
5 is I, V, T, M, S, N, L, or F. In some embodiments, the AAV capsid variant is an AAV9 capsid variant. In some embodiments, the AAV capsid variant comprises an amino acid sequence that is at least 95% identical to positions 203-736 of the amino acid sequence of SEQ ID NO: 981. In some embodiments, loop VIII is present at amino acids comprising those corresponding to positions 580-599 of the amino acid sequence of SEQ ID NO: 981. [024] In some embodiments, X
1 is T, S, R, A, I, C, N, K, L, or Q; X
2 is N, T, G, V, S, Y, K, I, H, D, or F; and X
3 is T, N, K, D, I, S, P, A, Y, E, V, L, M, R, H, Q, or C. In some embodiments, X
6 is A, Y, P, N, S, T, G, E, V, W, F, or Q; and X7 is Q, G, N, K, H, R, E, L, P, or M. [025] In some embodiments, [N1] comprises the amino acid sequence of TNTQ (SEQ ID NO: 4688). In some embodiments, [N3] comprises the amino acid sequence of IAQ. In some embodiments, [N1]-N2]-[N3] comprises the amino acid sequence of TNTQDWHRIAQ (SEQ ID NO: 343). [026] In some embodiments, [N1] is present at amino acids comprising those corresponding to positions 580-599 of the amino acid sequence of SEQ ID NO: 981, [N2] is present at amino acids corresponding to positions 586-589 of the amino acid sequence of SEQ ID NO: 981, and [N3] is present at amino acids corresponding to positions 590-592 of the amino acid sequence of SEQ ID NO: 981. [027] In some embodiments, the AAV capsid variant further comprises [N4] comprising amino acids X
8, X
9, X
10, and X
11, wherein: X
8 is T, S, N, P, A, or I; X
9 is G, N, D, R, V, A, S, or Q; X
10 is W, S, C, R, L, or G; and/or X
11 is V, A, S, I, C, G, D, F, L, or T. In some embodiments, [N4] comprises the amino acid sequence of TGWV (SEQ ID NO: 5066). In some embodiments, [N4] is present at amino acids corresponding to positions 593-596 of the amino acid sequence of SEQ ID NO: 981. [028] In some embodiments, [N1]-[N2]-[N3]-[N4] comprises the amino acid sequence of TNTQDWHRIAQTGWV (SEQ ID NO: 201). [029] In some aspects, the present disclosure provides an AAV particle comprising a viral genome comprising an STXBP1-encoding sequence and an AAV capsid variant comprising an amino acid sequence that is at least 95% identical to positions 203-736 of the amino acid sequence of SEQ ID NO: 981, wherein the AAV capsid variant comprises: T at an amino acid corresponding to position 584 of the amino acid sequence of SEQ ID NO: 981; D at an amino acid corresponding to position 586 of the amino
Attorney Docket No.14640.0105-00304 acid sequence of SEQ ID NO: 981; W at an amino acid corresponding to position 587 of the amino acid sequence of SEQ ID NO: 981; H at an amino acid corresponding to position 588 of the amino acid sequence of SEQ ID NO: 981; R at an amino acid corresponding to position 589 of the amino acid sequence of SEQ ID NO: 981; and I at an amino acid corresponding to position 590 of the amino acid sequence of SEQ ID NO: 981. [030] In some aspects, the present disclosure provides an AAV particle comprising a viral genome comprising an STXBP1-encoding sequence and an AAV capsid variant comprising an amino acid sequence that is at least 95% identical to positions 138-736 of the amino acid sequence of SEQ ID NO: 981. In some aspects, the present disclosure provides an AAV particle comprising a viral genome comprising an STXBP1-encoding sequence and an AAV capsid variant comprising an amino acid sequence that is at least 95% identical to positions 138-736 of the amino acid sequence of SEQ ID NO: 981, wherein the AAV capsid variant comprises: T at an amino acid corresponding to position 584 of the amino acid sequence of SEQ ID NO: 981; D at an amino acid corresponding to position 586 of the amino acid sequence of SEQ ID NO: 981; W at an amino acid corresponding to position 587 of the amino acid sequence of SEQ ID NO: 981; H at an amino acid corresponding to position 588 of the amino acid sequence of SEQ ID NO: 981; R at an amino acid corresponding to position 589 of the amino acid sequence of SEQ ID NO: 981; and I at an amino acid corresponding to position 590 of the amino acid sequence of SEQ ID NO: 981. [031] In some aspects, the present disclosure provides an AAV particle comprising a viral genome comprising an STXBP1-encoding sequence and an AAV capsid variant comprising an amino acid sequence that is at least 95% identical to the amino acid sequence of SEQ ID NO: 981. In some aspects, the present disclosure provides an AAV particle comprising a viral genome comprising an STXBP1- encoding sequence and an AAV capsid variant comprising an amino acid sequence that is at least 95% identical to the amino acid sequence of SEQ ID NO: 981, wherein the AAV capsid variant comprises: T at an amino acid corresponding to position 584 of the amino acid sequence of SEQ ID NO: 981; D at an amino acid corresponding to position 586 of the amino acid sequence of SEQ ID NO: 981; W at an amino acid corresponding to position 587 of the amino acid sequence of SEQ ID NO: 981; H at an amino acid corresponding to position 588 of the amino acid sequence of SEQ ID NO: 981; R at an amino acid corresponding to position 589 of the amino acid sequence of SEQ ID NO: 981; and I at an amino acid corresponding to position 590 of the amino acid sequence of SEQ ID NO: 981. [032] In some aspects, the present disclosure provides an AAV particle comprising a viral genome comprising an STXBP1-encoding sequence and an AAV capsid variant, wherein the AAV capsid variant comprises: (i) an amino acid sequence that is at least 99% identical to the amino acid sequence of SEQ ID NO: 981; (ii) an amino acid sequence that is at least 99% identical to positions 138-736 of the amino acid sequence of SEQ ID NO: 981; and/or (iii) an amino acid sequence that is at least 99% identical to positions 203-736 of the amino acid sequence of SEQ ID NO: 981. In some embodiments, the AAV capsid variant comprises the amino acid sequence of TQDWHRI (SEQ ID NO: 941).
Attorney Docket No.14640.0105-00304 [033] In some embodiments, the AAV capsid variant comprises the amino acid sequence of TNTQDWHRIAQ (SEQ ID NO: 343) present at amino acids corresponding to positions 582-592 of the amino acid sequence of SEQ ID NO: 981. In some embodiments, the amino acid sequence of TQDWHRI (SEQ ID NO: 941) or TNTQDWHRIAQ (SEQ ID NO: 343) is present in loop VIII, wherein loop VIII comprises amino acids 580-599 of the amino acid sequence of SEQ ID NO: 981. [034] In some embodiments, the AAV capsid variant comprises: (i) the amino acid sequence of SEQ ID NO: 981; (ii) the amino acid sequence according to positions 138-736 of the amino acid sequence of SEQ ID NO: 981; and/or (iii) the amino acid sequence according to positions 203-736 of the amino acid sequence of SEQ ID NO: 981. [035] In some embodiments, the viral genome encodes a wildtype STXBP1 protein or a fragment thereof. In some embodiments, the viral genome encodes a human STXBP1 protein. In some embodiments, the STXBP1 protein comprises the amino acid sequence of SEQ ID NO: 6413. [036] In some embodiments, the STXBP1-encoding sequence is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to the nucleotide sequence of SEQ ID NO: 6414. In some embodiments, the STXBP1-encoding sequence is at least 95% identical to the nucleotide sequence of SEQ ID NO: 6414. In some embodiments, the STXBP1-encoding sequence is at least 99% identical to the nucleotide sequence of SEQ ID NO: 6414. In some embodiments, the STXBP1-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 6414. In some embodiments, the STXBP1-encoding sequence consists of the nucleotide sequence of SEQ ID NO: 6414. [037] In some embodiments, the viral genome comprises a promoter operably linked to the STXBP1-encoding sequence. In some embodiments, the viral genome further comprises an inverted terminal repeat (ITR) sequence. In some embodiments, viral genome comprises an ITR sequence positioned 5’ relative to the STXBP1-encoding sequence. In some embodiments, the viral genome comprises an ITR sequence positioned 3’ relative to the STXBP1-encoding sequence. In some embodiments, the viral genome comprises an ITR sequence positioned 5’ relative to the STXBP1- encoding sequence, and an ITR sequence positioned 3’ relative to the STXBP1-encoding sequence. [038] In some aspects, the present disclosure provides a cell comprising an AAV particle provided herein. In some embodiments, the cell is a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial cell. [039] In some aspects, the present disclosure provides a method of making an AAV particle provided herein, wherein the method comprises: (i) providing a cell comprising a viral genome comprising an STXBP1-encoding sequence and a nucleic acid encoding an AAV capsid variant; and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant; thereby making the AAV particle. [040] In some embodiments of the method of making an AAV particle, the viral genome comprises the nucleotide sequence of SEQ ID NO: 6414 or a nucleotide sequence that is at least 90% (e.g., at least
Attorney Docket No.14640.0105-00304 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto and the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 981 or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, the amino acid sequence according to positions 138-736 of the amino acid sequence of SEQ ID NO: 981 or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, and/or the amino acid sequence according to positions 203-736 of the amino acid sequence of SEQ ID NO: 981 or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [041] In some embodiments of the method of making an AAV particle, the viral genome comprises the nucleotide sequence of SEQ ID NO: 6414 or a nucleotide sequence that is at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto and the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 981, the amino acid sequence according to positions 138-736 of the amino acid sequence of SEQ ID NO: 981, and/or the amino acid sequence according to positions 203-736 of the amino acid sequence of SEQ ID NO: 981. [042] In some embodiments of the method of making an AAV particle, the viral genome comprises the nucleotide sequence of SEQ ID NO: 6414 and the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 981, the amino acid sequence according to positions 138-736 of the amino acid sequence of SEQ ID NO: 981, and/or the amino acid sequence according to positions 203-736 of the amino acid sequence of SEQ ID NO: 981. [043] In some embodiments, the method of making an AAV particle further comprises, prior to step (i), introducing a nucleic acid molecule comprising the viral genome into the cell. In some embodiments, the method further comprises, prior to step (i), introducing the nucleic acid encoding the AAV capsid variant into the cell. In some embodiments, the cell comprises a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial cell. [044] In some aspects, the present disclosure provides a pharmaceutical composition comprising an AAV particle provided herein and a pharmaceutically acceptable excipient. [045] In some aspects, the present disclosure provides a method of delivering an AAV particle encoding an STXBP1 protein to a cell, comprising administering an effective amount of a pharmaceutical composition or AAV particle provided herein. In some embodiments, the cell is in a subject. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having an STXBP1-related disorder. In some embodiments, the STXBP1-related disorder is a STXBP1-related neurodegenerative or neuromuscular disorder. In some embodiments, the STXBP1-related disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West
Attorney Docket No.14640.0105-00304 syndrome, early myoclonic epileptic encephalopathy, Lennox-Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). [046] In some aspects, the present disclosure provides a method of treating an STXBP1-related disorder in a subject, comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle provided herein, thereby treating the STXBP1-related disorder. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having a STXBP1-related disorder. In some embodiments, the STXBP1-related disorder is a STXBP1-related neurodegenerative or neuromuscular disorder. In some embodiments, the STXBP1-related neurodegenerative or neuromuscular disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox- Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). [047] In some aspects, the present disclosure provides a method of treating an STXBP1-related disorder in a subject, wherein the disorder is STXBP1 encephalopathy, comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle provided herein. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having STXBP1 encephalopathy. [048] In some embodiments, the subject has one or more mutations in the STXBP1 gene. [049] In some embodiments, the subject has lower STXBP1 activity as compared to STXBP1 activity in an individual who does not have an STXBP1-related disorder. [050] In some embodiments, treating results in prevention of progression of an STXBP1-related disorder (e.g., STXBP1 encephalopathy) in the subject. In some embodiments, treating results in amelioration of at least one symptom of an STXBP1-related disorder (e.g., STXBP1 encephalopathy), as indicated by one or more biomarkers. In some embodiments, one or more biomarkers comprise increased release of the neurotransmitters glutamate and/or GABA or reduction in abnormal electroencephalographic activity as evidence of improved STXBP1 activity. In some embodiments, the at least one symptom comprises epilepsy, autistic features, ataxia, generalized tremors, dystonia, or a combination thereof. [051] In some embodiments, the subject is a human. [052] In some embodiments, the AAV particle or pharmaceutical composition is delivered to a cell tissue, or region of the central nervous systems (CNS) of the subject. In some embodiments, the cell, tissue, or region of the CNS is a cell (e.g., a neuron, e.g., a GABAergic neuron and/or a glutamatergic neuron), tissue, or region of the of the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus,
Attorney Docket No.14640.0105-00304 gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region). In some embodiments, the AAV particle or the pharmaceutical composition is delivered to the subject via intravenous administration. [053] In some embodiments, a method of delivery or treating provided herein further comprises evaluating, e.g., measuring, the level of STXBP1 expression, e.g., STXBP1 gene expression, STXBP1 mRNA expression, and/or STXBP1 protein expression, in the subject, e.g., in a cell, tissue, or fluid of the subject. In some embodiments, the level of STXBP1 protein expression is measured by an enzyme-linked immunosorbent assay (ELISA), a Western blot, or an immunohistochemistry assay. In some embodiments, evaluating the level of STXBP1 expression (e.g., STXBP1 gene expression, STXBP1 mRNA expression, and/or STXBP1 protein expression) is performed prior to and/or subsequent to administration of the AAV particle or pharmaceutical composition. In some embodiments, the subject’s level of STXBP1 expression (e.g., STXBP1 gene expression, STXBP1 mRNA expression, and/or STXBP1 protein expression) prior to administration is compared to the subject’s level of STXBP1 expression (e.g., STXBP1 gene expression, STXBP1 mRNA expression, and/or STXBP1 protein expression) subsequent to administration. In some embodiments, the method comprises evaluating the level of STXBP1 expression (e.g., STXBP1 gene expression, STXBP1 mRNA expression, and/or STXBP1 protein expression) in a cell or tissue of the CNS. In some embodiments, the cell or tissue of the CNS is a cell (e.g., a neuron, e.g., a GABAergic neuron and/or a glutamatergic neuron) or tissue of the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region). In some embodiments, the subject’s level of STXBP1 protein expression subsequent to administration is increased relative to the subject’s level of STXBP1 protein expression prior to administration. In some embodiments, a method of delivery or treating provided herein further comprises evaluating, e.g., measuring, the level of STXBP1 activity in the subject, e.g., in a cell or tissue of the subject. [054] In some embodiments, administering a pharmaceutical composition or AAV particle provided herein to the subject results an increase in: (i) the level of STXBP1 activity in a cell or tissue (e.g., a cell or tissue of the CNS, e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, spinal
Attorney Docket No.14640.0105-00304 cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron)), and/or fluid (e.g., CSF and/or serum) of the subject, relative to baseline and/or relative to the level of STXBP1 activity in a cell, tissue, or fluid of an individual with an STXBP1-related disorder who has not been administered the pharmaceutical composition or AAV particle; (ii) the number and/or level of viral genomes (VG) per cell level in a tissue of the CNS (e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region)), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron)) of the subject, relative to the number and/or level of VG per cell in a peripheral tissue of the subject; and/or (iii) the level of STXBP1 protein or STXBP1 gene expression in a cell or tissue (e.g., a cell or tissue of the CNS, e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron)) of the subject relative to baseline and/or relative to the level of STXBP1 protein or STXBP1 gene in a cell or tissue of an individual with a STXBP1-related disorder who has not been administered the pharmaceutical composition or AAV particle. [055] In some embodiments, a method of delivery or treating provided herein further comprises administering to the subject at least one additional agent and/or therapy. In some embodiments, the at least one additional agent and/or therapy comprises an agent and/or therapy suitable for treating the STXBP1-related disorder. In some embodiments, the at least one additional agent and/or therapy comprises one or more anti-epileptic drugs (e.g., bromide, clobazam, felbamate, ganaxolone, lamotrigine, levetiracetam, phenobarbital, topiramate, valproate, or a combination thereof). [056] In some embodiments, a method of delivery or treating provided herein further comprises administering an immunosuppressant to the subject. In some embodiments, the immunosuppressant comprises a corticosteroid (for example, and without limitation, prednisone, prednisolone, methylprednisolone, and/or dexamethasone), adrenocorticotropic hormone, rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine. [057] In some aspects, the present disclosure provides a pharmaceutical composition or AAV particle provided herein for use in a method of treating a disorder provided herein.
Attorney Docket No.14640.0105-00304 [058] In some aspects, the present disclosure provides a pharmaceutical composition or AAV particle provided herein for use in the treatment of an STXBP1-related disorder in a subject. In some embodiments, the STXBP1-related disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). In some embodiments, the subject has, has been diagnosed with having, or is at risk of having the STXBP1-related disorder, e.g., STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). [059] In some aspects, the present disclosure provides a use of a pharmaceutical composition or AAV particle provided herein in the manufacture of a medicament for the treatment of an STXBP1- related disorder. In some embodiments, the STXBP1-related disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). In some embodiments, the subject has, has been diagnosed with having, or is at risk of having the STXBP1-related disorder, e.g., STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). Enumerated Embodiments 1. An adeno-associated virus (AAV) particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)-encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant), wherein the AAV capsid variant comprises an amino acid sequence having the following formula: [N1]-[N2]-[N3] (SEQ ID NO: 4681), wherein [N2] comprises the amino acid sequence of DWHR (SEQ ID NO: 4682) and wherein: (i) [N1] comprises amino acids X
1, X
2, X
3, and X
4, wherein X
4 is Q, K, E, S, P, R, N, H; and/or (ii) [N3] comprises amino acids X
5, X
6, and X
7, wherein X
5 is I, V, T, M, S, N, L, F. 2. The AAV particle of embodiment 1, wherein the AAV capsid variant comprises the amino acid Q at position 585, as numbered according to SEQ ID NO: 138 or 981.
Attorney Docket No.14640.0105-00304 3. The AAV particle of embodiment 1, wherein the AAV capsid variant comprises an amino acid other than Q at position 585, as numbered according to SEQ ID NO: 138 or 981. 4. The AAV particle of embodiment 1 or 3, wherein the AAV capsid variant comprises the amino acid K at position 585, as numbered according to SEQ ID NO: 138 or 981. 5. The AAV particle of any one of embodiments 1-4, wherein the AAV capsid variant comprises one, two, three, or all of: (i) an amino acid other than T at position 582 (e.g., S, R, A, I, C, N, K, L, or Q); (ii) an amino acid other than N at position 583 (e.g., T, G, V, S, Y, K, I, H, D, or F); (iii) an amino acid other than H at position 584 (e.g., T, N, K, D, I, S, P, A, Y, E, V, L, M, R, Q, or C); and/or (iv) an amino acid other than Q at position 585 (e.g., K, E, S, P, R, N, or H); wherein the amino acids are numbered according to SEQ ID NO: 138. 6. The AAV particle of any one of embodiments 1-5, wherein [N1] comprises amino acids X
1, X
2, X
3, and X
4, wherein X
4 is Q, K, E, S, P, R, N, or H. 7. The AAV particle of embodiment 6, wherein X
4 is Q or K. 8. The AAV particle of any one of embodiment 7, wherein X
4 is Q. 9. The AAV particle of embodiment 7, wherein X
4 is K. 10. The AAV particle of any one of embodiments 1-9, wherein the AAV capsid variant comprises an amino acid other than H at position 584 (e.g., T), as numbered according to SEQ ID NO: 138. 11. The AAV particle of any one of embodiments 1-10, wherein the AAV capsid variant comprises the amino acid T at position 584, as numbered according to SEQ ID NO: 138 or 981. 12. The AAV particle of any one of embodiments 1-11, wherein: (i) X
1 is T, S, R, A, I, C, N, K, L, or Q; (ii) X
2 is N, T, G, V, S, Y, K, I, H, D, or F; and/or (iii) X
3 is T, N, K, D, I, S, P, A, Y, E, V, L, M, R, H, Q, or C. 13. The AAV particle of any one of embodiments 1-12, wherein [N1] comprises TN, NT, NK, SN, TT, RN, TG, TV, ST, TS, TY, AN, TK, TI, IN, TH, TD, CN, NN, KN, LN, SG, TF, RT, SY, SS, QN, ND,
Attorney Docket No.14640.0105-00304 NP, GK, TA, VK, NY, TE, SK, NI, YN, GT, TL, TM, YT, TR, NS, IT, NA, KT, GN, HT, DT, NE, NH, YI, HN, NQ, FS, NM, NL, SM, NC, VT, KQ, TQ, DQ, IQ, SQ, PS, KE, AQ, YQ, TP, EQ, VQ, LQ, MQ, KS, IE, RQ, IK, AK, PK, NR, HQ, QQ, or CQ. 14. The AAV particle of any one of embodiments 1-13, wherein [N1] comprises TNT, TNK, TNN, SNN, SNK, SNT, TTN, TND, TTI, RNT, TTK, TTS, TTD, TNP, TTT, TGK, TTA, TVK, TNY, STK, TTE, TSK, TNI, TYN, STI, TTV, TGT, TTL, TTM, ANN, SNI, TKN, TYT, TTR, TNS, TST, TIT, INT, TNA, TKT, STN, ANT, RNN, TGN, TSN, THT, TDT, TNE, CNT, INN, NNN, KNN, LNN, TIN, TNH, STT, SNS, STS, TYI, SGT, THN, TNQ, RNI, TFS, RNS, TNM, RTT, KNT, TNL, TSM, SYT, TNC, SST, TVT, QNT, NTK, NNQ, NKQ, NNE, NTQ, NDQ, TIQ, TKQ, TSQ, TDQ, NPS, NKE, TTQ, GKQ, TAQ, VKQ, NYQ, NTP, TEQ, SKQ, NIQ, YNQ, TVQ, GTQ, NTR, TLQ, TMQ, KNQ, YTQ, NKS, NTE, NIE, TRQ, NSQ, YTK, NIK, NNK, NSK, ITK, NAK, KTK, GNQ, SNQ, HTK, DTK, NEQ, NPK, YTE, NNR, INQ, NHQ, YIQ, HNQ, ITQ, STQ, NSN, NQQ, NNP, ITE, NTN, FSQ, NNH, NMQ, NTS, NLQ, SMQ, NCQ, or VTQ 15. The AAV particle of any one of embodiments 1-14, wherein [N1] is or comprises TNTQ (SEQ ID NO: 4688), TNTK (SEQ ID NO: 4689), TNNQ (SEQ ID NO: 4690), SNNQ (SEQ ID NO: 4691), TNKQ (SEQ ID NO: 4692), TNNE (SEQ ID NO: 4693), SNKQ (SEQ ID NO: 4694), SNTQ (SEQ ID NO: 4695), TTNQ (SEQ ID NO: 4696), TNDQ (SEQ ID NO: 4697), TTIQ (SEQ ID NO: 4698), RNTQ (SEQ ID NO: 4699), TTKQ (SEQ ID NO: 4700), TTSQ (SEQ ID NO: 4701), TTDQ (SEQ ID NO: 4702), TNPS (SEQ ID NO: 4703), TNKE (SEQ ID NO: 4704), TTTQ (SEQ ID NO: 4705), TGKQ (SEQ ID NO: 4706), TTAQ (SEQ ID NO: 4707), TVKQ (SEQ ID NO: 4708), TNYQ (SEQ ID NO: 4709), TNTP (SEQ ID NO: 4710), STKQ (SEQ ID NO: 4711), TTEQ (SEQ ID NO: 4712), TSKQ (SEQ ID NO: 4713), TNIQ (SEQ ID NO: 4714), TYNQ (SEQ ID NO: 4715), STIQ (SEQ ID NO: 4716), TTVQ (SEQ ID NO: 4717), TGTQ (SEQ ID NO: 4718), TNTR (SEQ ID NO: 4719), TTLQ (SEQ ID NO: 4720), TTMQ (SEQ ID NO: 4721), ANNQ (SEQ ID NO: 4722), SNIQ (SEQ ID NO: 4723), TKNQ (SEQ ID NO: 4724), TYTQ (SEQ ID NO: 4725), TNKS (SEQ ID NO: 4726), SNTE (SEQ ID NO: 4727), TNTE (SEQ ID NO: 4728), TNIE (SEQ ID NO: 4729), TTRQ (SEQ ID NO: 4730), TNSQ (SEQ ID NO: 4731), TYTK (SEQ ID NO: 4732), TTTK (SEQ ID NO: 4733), TNIK (SEQ ID NO: 4734), SNTK (SEQ ID NO: 4735), TNNK (SEQ ID NO: 4736), TNSK (SEQ ID NO: 4737), TSTK (SEQ ID NO: 4738), TITK (SEQ ID NO: 4739), INTK (SEQ ID NO: 4740), TNAK (SEQ ID NO: 4741), TKTK (SEQ ID NO: 4742), STNQ (SEQ ID NO: 4743), ANTK (SEQ ID NO: 4744), RNNQ (SEQ ID NO: 4745), TGNQ (SEQ ID NO: 4746), TSNQ (SEQ ID NO: 4747), THTK (SEQ ID NO: 4748), TDTK (SEQ ID NO: 4749), TNEQ (SEQ ID NO: 4750), CNTQ (SEQ ID NO: 4751), TNPK (SEQ ID NO: 4752), INNQ (SEQ ID NO: 4753), TYTE (SEQ ID NO: 4754), NNNQ (SEQ ID NO: 4755), KNNQ (SEQ ID NO: 4756), TNNR (SEQ ID NO: 4757), LNNQ (SEQ ID NO: 4758), TINQ (SEQ ID NO: 4759), TNHQ (SEQ ID NO: 4760), STTQ (SEQ ID NO: 4761), SNSQ (SEQ ID NO: 4762), STSQ (SEQ ID NO: 4763), TYIQ
Attorney Docket No.14640.0105-00304 (SEQ ID NO: 4764), SGTQ (SEQ ID NO: 4765), THNQ (SEQ ID NO: 4766), TITQ (SEQ ID NO: 4767), TSTQ (SEQ ID NO: 4768), TNSN (SEQ ID NO: 4769), TNQQ (SEQ ID NO: 4770), RNIQ (SEQ ID NO: 4771), TNNP (SEQ ID NO: 4772), TITE (SEQ ID NO: 4773), TNTN (SEQ ID NO: 4774), TFSQ (SEQ ID NO: 4775), RNSQ (SEQ ID NO: 4776), INTQ (SEQ ID NO: 4777), RNTE (SEQ ID NO: 4778), TNNH (SEQ ID NO: 4779), TNMQ (SEQ ID NO: 4780), RTTQ (SEQ ID NO: 4781), SNIE (SEQ ID NO: 4782), TNTS (SEQ ID NO: 4783), KNTQ (SEQ ID NO: 4784), TNLQ (SEQ ID NO: 4785), TSMQ (SEQ ID NO: 4786), SYTQ (SEQ ID NO: 4787), TNCQ (SEQ ID NO: 4788), SSTQ (SEQ ID NO: 4789), TVTQ (SEQ ID NO: 4790), or QNTQ (SEQ ID NO: 4791). 16. The AAV particle of embodiment 15, wherein [N1] is or comprises TNTQ (SEQ ID NO: 4688). 17. The AAV particle of embodiment 15, wherein [N1] is or comprises TNTK (SEQ ID NO: 4689). 18. The AAV particle of any one of embodiments 1-17, wherein [N1]-[N2] comprises: (i) TQDWHR (SEQ ID NO: 4686), TKDWHR (SEQ ID NO: 4792), NQDWHR (SEQ ID NO: 4793), KQDWHR (SEQ ID NO: 4794), NEDWHR (SEQ ID NO: 4795), DQDWHR (SEQ ID NO: 4796), IQDWHR (SEQ ID NO: 4797), SQDWHR (SEQ ID NO: 4798), PSDWHR (SEQ ID NO: 4799), KEDWHR (SEQ ID NO: 4800), AQDWHR (SEQ ID NO: 4801), YQDWHR (SEQ ID NO: 4802), TPDWHR (SEQ ID NO: 4803), EQDWHR (SEQ ID NO: 4804), VQDWHR (SEQ ID NO: 4805), TRDWHR (SEQ ID NO: 4806), LQDWHR (SEQ ID NO: 4807), MQDWHR (SEQ ID NO: 4808), KSDWHR (SEQ ID NO: 4809), TEDWHR (SEQ ID NO: 4810), IEDWHR (SEQ ID NO: 4811), RQDWHR (SEQ ID NO: 4812), IKDWHR (SEQ ID NO: 4813), NKDWHR (SEQ ID NO: 4814), SKDWHR (SEQ ID NO: 4815), AKDWHR (SEQ ID NO: 4816), PKDWHR (SEQ ID NO: 4817), NRDWHR (SEQ ID NO: 4818), HQDWHR (SEQ ID NO: 4819), SNDWHR (SEQ ID NO: 4820), QQDWHR (SEQ ID NO: 4821), NPDWHR (SEQ ID NO: 4822), TNDWHR (SEQ ID NO: 4823), NHDWHR (SEQ ID NO: 4824), TSDWHR (SEQ ID NO: 4825), or CQDWHR (SEQ ID NO: 4826); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 19. The AAV particle of any one of embodiments 1-18, wherein [N1]-[N2] comprises: (i) NTQDWHR (SEQ ID NO: 4827), NTKDWHR (SEQ ID NO: 4828), NNQDWHR (SEQ ID NO: 4829), NKQDWHR (SEQ ID NO: 4830), NNEDWHR (SEQ ID NO: 4831), TNQDWHR (SEQ ID NO: 4832), NDQDWHR (SEQ ID NO: 4833), TIQDWHR (SEQ ID NO: 4834), TKQDWHR (SEQ ID
Attorney Docket No.14640.0105-00304 NO: 4835), TSQDWHR (SEQ ID NO: 4836), TDQDWHR (SEQ ID NO: 4837), NPSDWHR (SEQ ID NO: 4838), NKEDWHR (SEQ ID NO: 4839), TTQDWHR (SEQ ID NO: 4840), GKQDWHR (SEQ ID NO: 4841), TAQDWHR (SEQ ID NO: 4842), VKQDWHR (SEQ ID NO: 4843), NYQDWHR (SEQ ID NO: 4844), NTPDWHR (SEQ ID NO: 4845), TEQDWHR (SEQ ID NO: 4846), SKQDWHR (SEQ ID NO: 4847), NIQDWHR (SEQ ID NO: 4848), YNQDWHR (SEQ ID NO: 4849), TVQDWHR (SEQ ID NO: 4850), GTQDWHR (SEQ ID NO: 4851), NTRDWHR (SEQ ID NO: 4852), TLQDWHR (SEQ ID NO: 4853), TMQDWHR (SEQ ID NO: 4854), KNQDWHR (SEQ ID NO: 4855), YTQDWHR (SEQ ID NO: 4856), NKSDWHR (SEQ ID NO: 4857), NTEDWHR (SEQ ID NO: 4858), NIEDWHR (SEQ ID NO: 4859), TRQDWHR (SEQ ID NO: 4860), NSQDWHR (SEQ ID NO: 4861), YTKDWHR (SEQ ID NO: 4862), TTKDWHR (SEQ ID NO: 4863), NIKDWHR (SEQ ID NO: 4864), NNKDWHR (SEQ ID NO: 4865), NSKDWHR (SEQ ID NO: 4866), STKDWHR (SEQ ID NO: 4867), ITKDWHR (SEQ ID NO: 4868), NAKDWHR (SEQ ID NO: 4869), KTKDWHR (SEQ ID NO: 4870), GNQDWHR (SEQ ID NO: 4871), SNQDWHR (SEQ ID NO: 4872), HTKDWHR (SEQ ID NO: 4873), DTKDWHR (SEQ ID NO: 4874), NEQDWHR (SEQ ID NO: 4875), NPKDWHR (SEQ ID NO: 4876), YTEDWHR (SEQ ID NO: 4877), NNRDWHR (SEQ ID NO: 4878), INQDWHR (SEQ ID NO: 4879), NHQDWHR (SEQ ID NO: 4880), YIQDWHR (SEQ ID NO: 4881), HNQDWHR (SEQ ID NO: 4882), ITQDWHR (SEQ ID NO: 4883), STQDWHR (SEQ ID NO: 4884), NSNDWHR (SEQ ID NO: 4885), NQQDWHR (SEQ ID NO: 4886), NNPDWHR (SEQ ID NO: 4887), ITEDWHR (SEQ ID NO: 4888), NTNDWHR (SEQ ID NO: 4889), FSQDWHR (SEQ ID NO: 4890), NNHDWHR (SEQ ID NO: 4891), NMQDWHR (SEQ ID NO: 4892), NTSDWHR (SEQ ID NO: 4893), NLQDWHR (SEQ ID NO: 4894), SMQDWHR (SEQ ID NO: 4895), NCQDWHR (SEQ ID NO: 4896), or VTQDWHR (SEQ ID NO: 4897); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5 or 6 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 20. The AAV particle of any one of embodiments 1-19, wherein [N1]-[N2] is or comprises: (i) TNTQDWHR (SEQ ID NO: 4898), TNTKDWHR (SEQ ID NO: 4899), TNNQDWHR (SEQ ID NO: 4900), SNNQDWHR (SEQ ID NO: 4901), TNKQDWHR (SEQ ID NO: 4902), TNNEDWHR (SEQ ID NO: 4903), SNKQDWHR (SEQ ID NO: 4904), SNTQDWHR (SEQ ID NO: 4905), TTNQDWHR (SEQ ID NO: 4906), TNDQDWHR (SEQ ID NO: 4907), TTIQDWHR (SEQ ID NO: 4908), RNTQDWHR (SEQ ID NO: 4909), TTKQDWHR (SEQ ID NO: 4910), TTSQDWHR (SEQ ID NO: 4911), TTDQDWHR (SEQ ID NO: 4912), TNPSDWHR (SEQ ID NO: 4913), TNKEDWHR (SEQ ID NO: 4914), TTTQDWHR (SEQ ID NO: 4915), TGKQDWHR (SEQ ID NO: 4916), TTAQDWHR (SEQ ID NO: 4917), TVKQDWHR (SEQ ID NO: 4918), TNYQDWHR (SEQ ID NO: 4919),
Attorney Docket No.14640.0105-00304 TNTPDWHR (SEQ ID NO: 4920), STKQDWHR (SEQ ID NO: 4921), TTEQDWHR (SEQ ID NO: 4922), TSKQDWHR (SEQ ID NO: 4923), TNIQDWHR (SEQ ID NO: 4924), TYNQDWHR (SEQ ID NO: 4925), STIQDWHR (SEQ ID NO: 4926), TTVQDWHR (SEQ ID NO: 4927), TGTQDWHR (SEQ ID NO: 4928), TNTRDWHR (SEQ ID NO: 4929), TTLQDWHR (SEQ ID NO: 4930), TTMQDWHR (SEQ ID NO: 4931), ANNQDWHR (SEQ ID NO: 4932), SNIQDWHR (SEQ ID NO: 4933), TKNQDWHR (SEQ ID NO: 4934), TYTQDWHR (SEQ ID NO: 4935), TNKSDWHR (SEQ ID NO: 4936), SNTEDWHR (SEQ ID NO: 4937), TNTEDWHR (SEQ ID NO: 4938), TNIEDWHR (SEQ ID NO: 4939), TTRQDWHR (SEQ ID NO: 4940), TNSQDWHR (SEQ ID NO: 4941), TYTKDWHR (SEQ ID NO: 4942), TTTKDWHR (SEQ ID NO: 4943), TNIKDWHR (SEQ ID NO: 4944), SNTKDWHR (SEQ ID NO: 4945), TNNKDWHR (SEQ ID NO: 4946), TNSKDWHR (SEQ ID NO: 4947), TSTKDWHR (SEQ ID NO: 4948), TITKDWHR (SEQ ID NO: 4949), INTKDWHR (SEQ ID NO: 4950), TNAKDWHR (SEQ ID NO: 4951), TKTKDWHR (SEQ ID NO: 4952), STNQDWHR (SEQ ID NO: 4953), ANTKDWHR (SEQ ID NO: 4954), RNNQDWHR (SEQ ID NO: 4955), TGNQDWHR (SEQ ID NO: 4956), TSNQDWHR (SEQ ID NO: 4957), THTKDWHR (SEQ ID NO: 4958), TDTKDWHR (SEQ ID NO: 4959), TNEQDWHR (SEQ ID NO: 4960), CNTQDWHR (SEQ ID NO: 4961), TNPKDWHR (SEQ ID NO: 4962), INNQDWHR (SEQ ID NO: 4963), TYTEDWHR (SEQ ID NO: 4964), NNNQDWHR (SEQ ID NO: 4965), KNNQDWHR (SEQ ID NO: 4966), TNNRDWHR (SEQ ID NO: 4967), LNNQDWHR (SEQ ID NO: 4968), TINQDWHR (SEQ ID NO: 4969), TNHQDWHR (SEQ ID NO: 4970), STTQDWHR (SEQ ID NO: 4971), SNSQDWHR (SEQ ID NO: 4972), STSQDWHR (SEQ ID NO: 4973), TYIQDWHR (SEQ ID NO: 4974), SGTQDWHR (SEQ ID NO: 4975), THNQDWHR (SEQ ID NO: 4976), TITQDWHR (SEQ ID NO: 4977), TSTQDWHR (SEQ ID NO: 4978), TNSNDWHR (SEQ ID NO: 4979), TNQQDWHR (SEQ ID NO: 4980), RNIQDWHR (SEQ ID NO: 4981), TNNPDWHR (SEQ ID NO: 4982), TITEDWHR (SEQ ID NO: 4983), TNTNDWHR (SEQ ID NO: 4984), TFSQDWHR (SEQ ID NO: 4985), RNSQDWHR (SEQ ID NO: 4986), INTQDWHR (SEQ ID NO: 4987), RNTEDWHR (SEQ ID NO: 4988), TNNHDWHR (SEQ ID NO: 4989), TNMQDWHR (SEQ ID NO: 4990), RTTQDWHR (SEQ ID NO: 4991), SNIEDWHR (SEQ ID NO: 4992), TNTSDWHR (SEQ ID NO: 4993), KNTQDWHR (SEQ ID NO: 4994), TNLQDWHR (SEQ ID NO: 4995), TSMQDWHR (SEQ ID NO: 4996), SYTQDWHR (SEQ ID NO: 4997), TNCQDWHR (SEQ ID NO: 4998), SSTQDWHR (SEQ ID NO: 4999), TVTQDWHR (SEQ ID NO: 5000), or QNTQDWHR (SEQ ID NO: 5001); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, or 7 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions, relative to any one of the amino acid sequences in (i).
Attorney Docket No.14640.0105-00304 21. The AAV particle of embodiment 20, wherein [N1]-[N2] is or comprises TNTQDWHR (SEQ ID NO: 4898). 22. The AAV particle of embodiment 20, wherein [N1]-[N2] is or comprises TNTKDWHR (SEQ ID NO: 4899). 23. The AAV particle of any one of embodiments 1-22, wherein the AAV capsid variant comprises one, two, or all of: an amino acid other than Q at position 590 (e.g., I, V, T, M, S, N, L, or F), an amino acid other than A at position 591 (e.g., Y, P, N, S, T, G, E, V, W, F, Q), and/or an amino acid other than Q at position 592 (e.g., G, N, K, H, R, E, L, P, or M), as numbered according to SEQ ID NO: 138 or 981. 24. The AAV particle of any one of embodiments 1-23, wherein the AAV capsid variant comprises an amino acid other than Q at position 590 (e.g., I, V, T, M, S, N, L, or F), as numbered according to SEQ ID NO: 138 or 981. 25. The AAV particle of any one of embodiments 1-24, wherein the AAV capsid variant comprises the amino acid I at position 590, as numbered according to SEQ ID NO: 138 or 981. 26. The AAV particle of any one of embodiments 1-24, wherein the AAV capsid variant comprises the amino acid V at position 590, as numbered according to SEQ ID NO: 138 or 981. 27. The AAV particle of any one of embodiments 1-26, wherein the AAV capsid variant comprises the amino acid A at position 591 and/or the amino acid Q at position 592, as numbered according to SEQ ID NO: 138 or 981. 28. The AAV particle of any one of embodiments 1-27, wherein [N3] comprises amino acids X
5, X
6, and X
7, wherein X
5 is I, V, T, M, S, N, L, or F. 29. The AAV particle of any one of embodiments 1-28, wherein X
5 is I or V. 30. The AAV particle of embodiment 29, wherein X
5 is I. 31. The AAV particle of any one of embodiments 1-30, wherein: (i) X
6 is A, Y, P, N, S, T, G, E, V, W, F, or Q; and/or (ii) X
7 is Q, G, N, K, H, R, E, L, P, or M.
Attorney Docket No.14640.0105-00304 32. The AAV particle of any one of embodiments 1-31, wherein [N3] comprises IA, IY, VP, IN, VN, VY, VA, IS, IT, TA, MA, SA, IG, IE, IV, NA, LA, IP, FA, VS, VT, IW, IF, IQ, VQ, AQ, AG, YQ, PQ, AN, NQ, SG, SQ, TQ, GQ, EQ, AK, AH, AR, AE, AL, AP, TM, SM, WQ, FQ, QQ, FM, AM, or SN. 33. The AAV particle of any one of embodiments 1-32, wherein [N3] is or comprises IAQ, IAG, IYQ, VPQ, IAN, INQ, VNQ, VYQ, VAN, ISG, ISQ, VAQ, ITQ, TAQ, MAQ, SAQ, IGQ, IEQ, IVQ, NAQ, LAQ, IAK, IAH, IPQ, IAR, IAE, IAL, IAP, FAQ, VSQ, VTM, ISM, IWQ, IFQ, IQQ, VQQ, IFM, IAM, or ISN. 34. The AAV particle of any one of embodiments 1-33, wherein [N3] is or comprises IAQ. 35. The AAV particle of any one of embodiments 1-34, wherein [N2]-[N3] comprises: (i) DWHRIA (SEQ ID NO: 5002), DWHRIY (SEQ ID NO: 5003), DWHRVP (SEQ ID NO: 5004), DWHRIN (SEQ ID NO: 5005), DWHRVN (SEQ ID NO: 5006), DWHRVY (SEQ ID NO: 5007), DWHRVA (SEQ ID NO: 5008), DWHRIS (SEQ ID NO: 5009), DWHRIT (SEQ ID NO: 5010), DWHRTA (SEQ ID NO: 5011), DWHRMA (SEQ ID NO: 5012), DWHRSA (SEQ ID NO: 5013), DWHRIG (SEQ ID NO: 5014), DWHRIE (SEQ ID NO: 5015), DWHRIV (SEQ ID NO: 5016), DWHRNA (SEQ ID NO: 5017), DWHRLA (SEQ ID NO: 5018), DWHRIP (SEQ ID NO: 5019), DWHRFA (SEQ ID NO: 5020), DWHRVS (SEQ ID NO: 5021), DWHRVT (SEQ ID NO: 5022), DWHRIW (SEQ ID NO: 5023), DWHRIF (SEQ ID NO: 5024), DWHRIQ (SEQ ID NO: 5025), or DWHRVQ (SEQ ID NO: 5026); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 36. The AAV particle of any one of embodiments 1-35, wherein [N2]-[N3] is or comprises: (i) DWHRIAQ (SEQ ID NO: 5027), DWHRIAG (SEQ ID NO: 5028), DWHRIYQ (SEQ ID NO: 5029), DWHRVPQ (SEQ ID NO: 5030), DWHRIAN (SEQ ID NO: 5031), DWHRINQ (SEQ ID NO: 5032), DWHRVNQ (SEQ ID NO: 5033), DWHRVYQ (SEQ ID NO: 5034), DWHRVAN (SEQ ID NO: 5035), DWHRISG (SEQ ID NO: 5036), DWHRISQ (SEQ ID NO: 5037), DWHRVAQ (SEQ ID NO: 5038), DWHRITQ (SEQ ID NO: 5039), DWHRTAQ (SEQ ID NO: 5040), DWHRMAQ (SEQ ID NO: 5041), DWHRSAQ (SEQ ID NO: 5042), DWHRIGQ (SEQ ID NO: 5043), DWHRIEQ (SEQ ID NO: 5044), DWHRIVQ (SEQ ID NO: 5045), DWHRNAQ (SEQ ID NO: 5046), DWHRLAQ (SEQ ID NO: 5047), DWHRIAK (SEQ ID NO: 5048), DWHRIAH (SEQ ID NO: 5049), DWHRIPQ (SEQ ID
Attorney Docket No.14640.0105-00304 NO: 5050), DWHRIAR (SEQ ID NO: 5051), DWHRIAE (SEQ ID NO: 5052), DWHRIAL (SEQ ID NO: 5053), DWHRIAP (SEQ ID NO: 5054), DWHRFAQ (SEQ ID NO: 5055), DWHRVSQ (SEQ ID NO: 5056), DWHRVTM (SEQ ID NO: 5057), DWHRISM (SEQ ID NO: 5058), DWHRIWQ (SEQ ID NO: 5059), DWHRIFQ (SEQ ID NO: 5060), DWHRIQQ (SEQ ID NO: 5061), DWHRVQQ (SEQ ID NO: 5062), DWHRIFM (SEQ ID NO: 5063), DWHRIAM (SEQ ID NO: 5064), or DWHRISN (SEQ ID NO: 5065); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 37. The AAV particle of any one of embodiments 1-36, wherein [N2]-[N3] is or comprises DWHRIAQ (SEQ ID NO: 5027). 38. The AAV particle of any one of embodiments 1-37, wherein [N1]-[N2]-[N3] is or comprises: (i) the amino acid sequence of any one of SEQ ID NOs: 343-538; (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 39. The AAV particle of embodiment 38, wherein [N1]-[N2]-[N3] is or comprises TNTQDWHRIAQ (SEQ ID NO: 343). 40. The AAV particle of embodiment 38, wherein [N1]-[N2]-[N3] is or comprises TNTKDWHRIAQ (SEQ ID NO: 344). 41. The AAV particle of any one of embodiments 1-40, wherein the AAV capsid variant comprises one, two, three, or all of: an amino acid other than T at position 593 (e.g., S, N, P, A, or I), an amino acid other than G at position 594 (e.g., N, D, R, V, A, S, or Q), an amino acid other than W at position 595 (e.g., S, C, R, L, or G), and/or an amino acid other than V at position 596 (e.g., A, S, I, C, G, D, F, L, or T), as numbered according to SEQ ID NO: 138 or 981.
Attorney Docket No.14640.0105-00304 42. The AAV particle of any one of embodiments 1-40, wherein the AAV capsid variant comprises the amino acid T at position 593, the amino acid G at position 594, the amino acid W at position 595, and the amino acid V at position 596, as numbered according to SEQ ID NO: 138 or 981. 43. The AAV particle of any one of embodiments 1-42, wherein the AAV capsid variant further comprises [N4], wherein [N4] comprises amino acids X
8, X
9, X
10, and X
11, wherein: (i) X
8 is T, S, N, P, A, or I; (ii) X
9 is G, N, D, R, V, A, S, or Q; (iii) X
10 is W, S, C, R, L, or G; and/or (iv) X
11 is V, A, S, I, C, G, D, F, L, or T. 44. The AAV particle of embodiment 43, wherein [N4] comprises TG, TN, SN, NN, SG, PG, TD, AG, IG, NG, TR, TV, TA, TS, SV, TQ, WV, WA, WS, WI, WC, WG, CV, RV, LV, GV, WD, WF, WL, WT, GW, NW, GS, DW, GC, GR, GL, GG, RW, VW, AW, SW, or QW. 45. The AAV particle of embodiment 43 or 44, wherein [N4] comprises TGW, TNW, SNW, NNW, SGW, PGW, TGS, TDW, TGC, TGR, TGL, TGG, AGW, IGW, NGW, TRW, TVW, TAW, TSW, SVW, TQW, GWV, GWA, NWS, NWV, NWI, GWS, GWI, GWC, GWG, GSV, DWV, GCV, GRV, GLV, GGV, GWD, GWF, RWV, VWV, GWL, AWV, SWV, GWT, or QWV. 46. The AAV particle of any one of embodiments 43-45, wherein [N4] is or comprises TGWV (SEQ ID NO: 5066), TGWA (SEQ ID NO: 5067), TNWS (SEQ ID NO: 5068), SNWV (SEQ ID NO: 5069), TNWV (SEQ ID NO: 5070), TNWI (SEQ ID NO: 5071), NNWV (SEQ ID NO: 5072), TGWS (SEQ ID NO: 5073), TGWI (SEQ ID NO: 5074), TGWC (SEQ ID NO: 5075), TGWG (SEQ ID NO: 5076), SGWV (SEQ ID NO: 5077), PGWV (SEQ ID NO: 5078), TGSV (SEQ ID NO: 5079), TDWV (SEQ ID NO: 5080), TGCV (SEQ ID NO: 5081), TGRV (SEQ ID NO: 5082), TGLV (SEQ ID NO: 5083), TGGV (SEQ ID NO: 5084), AGWV (SEQ ID NO: 5085), IGWV (SEQ ID NO: 5086), TGWD (SEQ ID NO: 5087), NGWV (SEQ ID NO: 5088), TGWF (SEQ ID NO: 5089), TRWV (SEQ ID NO: 5090), TVWV (SEQ ID NO: 5091), TGWL (SEQ ID NO: 5092), TAWV (SEQ ID NO: 5093), TSWV (SEQ ID NO: 5094), TGWT (SEQ ID NO: 5095), SVWV (SEQ ID NO: 5096), TQWV (SEQ ID NO: 5097), or PGWG (SEQ ID NO: 5098). 47. The AAV particle of any one of embodiments 43-46, wherein [N4] is or comprises TGWV (SEQ ID NO: 5066). 48. The AAV particle of any one of embodiments 43-47, wherein [N1]-[N2]-[N3]-[N4] is or comprises:
Attorney Docket No.14640.0105-00304 (i) the amino acid sequence of any one of SEQ ID NOs: 201-245, 247-250, 253-255, 257-265, 268-274, 276-286, 288, 290-297, 299-303, 305-309, 311, 313-319, 323-328, 330-337, 339-342, 539-542, 544, 546, 547, 549-557, 559-589, 592, 593, 595, 596, 598, 599, 601-608, 610-614, 616-622, 625, 628, 630, 631, 633, 636, 638, 639-646, 649, 651-657, 667, 669, 670, 672, 673, 679-683, 685-690, 692, 693, 695, 697, 699-701, 703-705, 708-710, 712-717, 719-723, 728-731, 733-738, 740, and 742; (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 49. The AAV particle of embodiment 48, wherein [N1]-[N2]-[N3]-[N4] is or comprises TNTQDWHRIAQTGWV (SEQ ID NO: 201). 50. The AAV particle of any one of embodiment 48, wherein [N1]-[N2]-[N3]-[N4] is or comprises TNTKDWHRIAQTGWV (SEQ ID NO: 202). 51. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising an amino acid sequence having the following formula: [N1]-[N2]-[N3] (SEQ ID NO: 4683), wherein [N2] comprises the amino acid sequence of DWHR (SEQ ID NO: 4682) and wherein: (i) [N1] comprises amino acids X
1, X
2, X
3, and X
4, wherein X
4 is Q, P, or a conservative substitution thereof; and/or (ii) [N3] comprises amino acids X
5, X
6, and X
7, wherein X
5 is I, V, or a conservative substitution thereof. 52. The AAV particle of embodiment 51, wherein the AAV capsid variant comprises the amino acid Q at position 585, as numbered according to SEQ ID NO: 138 or 981. 53. The AAV particle of embodiment 51 or 52, wherein the AAV capsid variant comprises one, two, three, or all of: (i) an amino acid other than T at position 582 (e.g., S); (ii) an amino acid other than N at position 583 (e.g., T, G, S, I, or V); (iii) an amino acid other than H at position 584 (e.g., N, I, S, A, V, or L); and/or (iv) an amino acid other than Q at position 585 (e.g., P),
Attorney Docket No.14640.0105-00304 wherein the amino acids are as numbered according to SEQ ID NO: 138 or 981. 54. The AAV particle of any one of embodiments 51-53, wherein [N1] comprises amino acids X
1, X
2, X
3, and X
4, wherein X
4 is Q or P. 55. The AAV particle of any one of embodiments 51-54, wherein X
4 is Q. 56. The AAV particle of any one of embodiments 51-55, wherein the AAV capsid variant comprises an amino acid other than H at position 584 (e.g., T), as numbered according to SEQ ID NO: 138 or 981. 57. The AAV particle of any one of embodiments 51-56, wherein the AAV capsid variant comprises the amino acid T at position 584, as numbered according to SEQ ID NO: 138 or 981. 58. The AAV particle of any one of embodiments 51-57, wherein: (i) X
1 is T or S; (ii) X
2 is N, T, G, S, I, or V; and/or (iii) X
3 is T, N, I, S, A, V, or L. 59. The AAV particle of any one of embodiments 51-58, wherein [N1] comprises TN, TT, TG, ST, TS, TI, TV, TQ, NQ, IQ, SQ, AQ, VQ, TP, LQ, NT, TA, NI, GT, IT, NN, TL, NS, or VT. 60. The AAV particle of any one of embodiments 51-59, wherein [N1] comprises TNT, TTN, TTI, TTS, TTT, TTA, TNI, TTV, TGT, STT, TST, TIT, TNN, TTL, TNS, TVT, NTQ, TNQ, TIQ, TSQ, TTQ, TAQ, NIQ, TVQ, GTQ, STQ, ITQ, NTP, NNQ, TLQ, NSQ, or VTQ. 61. The AAV particle of any one of embodiments 51-60, wherein [N1] is or comprises TNTQ (SEQ ID NO: 4688), TTNQ (SEQ ID NO: 4696), TTIQ (SEQ ID NO: 4698), TTSQ (SEQ ID NO: 4701), TTTQ (SEQ ID NO: 4705), TTAQ (SEQ ID NO: 4707), TNIQ (SEQ ID NO: 4714), TTVQ (SEQ ID NO: 4717), TGTQ (SEQ ID NO: 4718), STTQ (SEQ ID NO: 4761), TSTQ (SEQ ID NO: 4768), TITQ (SEQ ID NO: 4767), TNTP (SEQ ID NO: 4710), TNNQ (SEQ ID NO: 4690), TTLQ (SEQ ID NO: 4720), TNSQ (SEQ ID NO: 4731), or TVTQ (SEQ ID NO: 4790). 62. The AAV particle of embodiment 61, wherein [N1] is or comprises TNTQ (SEQ ID NO: 4688). 63. The AAV particle of any one of embodiments 51-62, wherein [N1]-[N2] comprises:
Attorney Docket No.14640.0105-00304 (i) TQDWHR (SEQ ID NO: 4686), NQDWHR (SEQ ID NO: 4793), IQDWHR (SEQ ID NO: 4797), SQDWHR (SEQ ID NO: 4798), AQDWHR (SEQ ID NO: 4801), VQDWHR (SEQ ID NO: 4805), TPDWHR (SEQ ID NO: 4803), or LQDWHR (SEQ ID NO: 4807); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 64. The AAV particle of any one of embodiments 51-63, wherein [N1]-[N2] comprises: (i) NTQDWHR (SEQ ID NO: 4827), TNQDWHR (SEQ ID NO: 4832), TIQDWHR (SEQ ID NO: 4834), TSQDWHR (SEQ ID NO: 4836), TTQDWHR (SEQ ID NO: 4840), TAQDWHR (SEQ ID NO: 4842), NIQDWHR (SEQ ID NO: 4848), TVQDWHR (SEQ ID NO: 4850), GTQDWHR (SEQ ID NO: 4851), STQDWHR (SEQ ID NO: 4884), ITQDWHR (SEQ ID NO: 4883), NTPDWHR (SEQ ID NO: 4845), NNQDWHR (SEQ ID NO: 4829), TLQDWHR (SEQ ID NO: 4853), NSQDWHR (SEQ ID NO: 4861), VTQDWHR (SEQ ID NO: 4897); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5 or 6 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 65. The AAV particle of any one of embodiments 51-64, wherein [N1]-[N2] is or comprises: (i) TNTQDWHR (SEQ ID NO: 4898), TTNQDWHR (SEQ ID NO: 4906), TTIQDWHR (SEQ ID NO: 4908), TTSQDWHR (SEQ ID NO: 4911), TTTQDWHR (SEQ ID NO: 4915), TTAQDWHR (SEQ ID NO: 4917), TNIQDWHR (SEQ ID NO: 4924), TTVQDWHR (SEQ ID NO: 4927), TGTQDWHR (SEQ ID NO: 4928), STTQDWHR (SEQ ID NO: 4971), TSTQDWHR (SEQ ID NO: 4978), TITQDWHR (SEQ ID NO: 4977), TNTPDWHR (SEQ ID NO: 4920), TNNQDWHR (SEQ ID NO: 4900), TTLQDWHR (SEQ ID NO: 4930), TNSQDWHR (SEQ ID NO: 4941), TVTQDWHR (SEQ ID NO: 5000); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 56, or 7 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or
Attorney Docket No.14640.0105-00304 (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 66. The AAV particle of embodiment 65, wherein [N1]-[N2] is or comprises TNTQDWHR (SEQ ID NO: 4898). 67. The AAV particle of any one of embodiments 51-66, wherein the AAV capsid variant comprises one, two, or all of: an amino acid other than Q at position 590 (e.g., I or V), an amino acid other than A at position 591 (e.g., P, S, Y, or N), and/or an amino acid other than Q at position 592 (e.g., G or N), as numbered according to SEQ ID NO: 138 or 981. 68. The AAV particle of any one of embodiments 51-67, wherein the AAV capsid variant comprises an amino acid other than Q at position 590 (e.g., I or V), as numbered according to SEQ ID NO: 138 or 981. 69. The AAV particle of any one of embodiments 51-68, wherein the AAV capsid variant comprises the amino acid I at position 590, as numbered according to SEQ ID NO: 138 or 981. 70. The AAV particle of any one of embodiments 51-68, wherein the AAV capsid variant comprises the amino acid V at position 590, as numbered according to SEQ ID NO: 138 or 981. 71. The AAV particle of any one of embodiments 51-70, wherein the AAV capsid variant comprises the amino acid A at position 591 and/or the amino acid Q at position 592, as numbered according to SEQ ID NO: 138 or 981. 72. The AAV particle of any one of embodiments 51-71, wherein [N3] comprises amino acids X
5, X
6, and X
7, wherein X
5 is I or V. 73. The AAV particle of embodiment 72, wherein X
5 is I. 74. The AAV particle of any one of embodiments 51-73, wherein: (i) X
6 is A, P, S, Y, or N; and/or (ii) X
7 is Q, G, or N. 75. The AAV particle of any one of embodiments 51-74, wherein [N3] comprises IA, VP, VA, VS, IY, IN, IS, AQ, AG, PQ, SQ, AN, YQ, or NQ.
Attorney Docket No.14640.0105-00304 76. The AAV particle of any one of embodiments 51-75, wherein [N3] is or comprises IAQ, IAG, VPQ, VAQ, VSQ, IAN, IYQ, INQ, or ISQ. 77. The AAV particle of any one of embodiments 51-76, wherein [N3] is or comprises IAQ. 78. The AAV particle of any one of embodiments 51-77, wherein [N2]-[N3] comprises: (i) DWHRIA (SEQ ID NO: 5002), DWHRVP (SEQ ID NO: 5004), DWHRVA (SEQ ID NO: 5008), DWHRVS (SEQ ID NO: 5021), DWHRIY (SEQ ID NO: 5003), DWHRIN (SEQ ID NO: 5005), or DWHRIS (SEQ ID NO: 5009); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 79. The AAV particle of any one of embodiments 51-78, wherein [N2]-[N3] is or comprises: (i) DWHRIAQ (SEQ ID NO: 5027), DWHRIAG (SEQ ID NO: 5028), DWHRVPQ (SEQ ID NO: 5030), DWHRVAQ (SEQ ID NO: 5038), DWHRVSQ (SEQ ID NO: 5056), DWHRIAN (SEQ ID NO: 5031), DWHRIYQ (SEQ ID NO: 5029), DWHRINQ (SEQ ID NO: 5032), or DWHRISQ (SEQ ID NO: 5037); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 80. The AAV particle of any one of embodiments 51-79, wherein [N2]-[N3] is or comprises DWHRIAQ (SEQ ID NO: 5027). 81. The AAV particle of any one of embodiments 51-80, wherein [N1]-[N2]-[N3] is or comprises: (i) the amino acid sequence of any one of SEQ ID NOs: 343, 350, 352, 355, 359, 361, 364, 367, 370, 371, 373, 374, 376, 377, 378, 381, 395, 420, 454, 457, 460, 464, 481, 482, 488, 493, 494, 516, 525, 536; (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof;
Attorney Docket No.14640.0105-00304 (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 82. The AAV particle of any one of embodiments 51-81, wherein [N1]-[N2]-[N3] is or comprises TNTQDWHRIAQ (SEQ ID NO: 343). 83. The AAV particle of any one of embodiments 51-82, wherein the AAV capsid variant comprises the amino acid W at position 595, as numbered according to SEQ ID NO: 138 or 981. 84. The AAV particle of any one of embodiments 51-83, wherein the AAV capsid variant comprises one, two or all of: an amino acid other than T (e.g., S or N) at position 593, an amino acid other than G (e.g., N) a position 594, and/or an amino acid other than V (e.g., A, I, or S) at position 596, as numbered according to SEQ ID NO: 138 or 981. 85. The AAV particle of any one of embodiments 51-84, wherein the AAV capsid variant comprises: (i) the amino acid T at position 593, the amino acid G at position 594, the amino acid W at position 595, and the amino acid V at position 596, as numbered according to SEQ ID NO: 138 or 981; (ii) the amino acid T at position 593, the amino acid G at position 594, the amino acid W at position 595, and the amino acid A at position 596, as numbered according to SEQ ID NO: 138 or 981; (iii) the amino acid S at position 593, the amino acid N at position 594, the amino acid W at position 595, and the amino acid V at position 596, as numbered according to SEQ ID NO: 138 or 981; (iv) the amino acid N at position 593, the amino acid N at position 594, the amino acid W at position 595, and the amino acid V at position 596, as numbered according to SEQ ID NO: 138 or 981; (v) the amino acid T at position 593, the amino acid G at position 594, the amino acid W at position 595, and the amino acid I at position 596, as numbered according to SEQ ID NO: 138 or 981; or (vi) the amino acid T at position 593, the amino acid G at position 594, the amino acid W at position 595, and the amino acid S at position 596, as numbered according to SEQ ID NO: 138 or 981. 86. The AAV particle of any one of embodiments 51-85, wherein the AAV capsid variant comprises the amino acid T at position 593, the amino acid G at position 594, the amino acid W at position 595, and the amino acid V at position 596, as numbered according to SEQ ID NO: 138 or 981. 87. The AAV particle of any one of embodiments 51-86, wherein the AAV capsid variant further comprises [N4], wherein [N4] comprises amino acids X
8, X
9, X
10, and X
11, wherein X
10 is W.
Attorney Docket No.14640.0105-00304 88. The AAV particle of embodiment 87, wherein: (i) X
8 is T, S, or N; (ii) X
9 is G or N; and/or (iv) X
11 is V, A, I, or S. 89. The AAV particle of embodiment 87 or 88, wherein [N4] comprises TG, SN, NN, WV, WA, WI, WS, GW, or NW. 90. The AAV particle of any one of embodiments 87-89, wherein [N4] comprises TGW, SNW, NNW, GWV, GWA, NWV, GWI, or GWS. 91. The AAV particle of any one of embodiments 87-90, wherein [N4] is or comprises TGWV (SEQ ID NO: 5066), TGWA (SEQ ID NO: 5067), SNWV (SEQ ID NO: 5069), NNWV (SEQ ID NO: 5072), TGWI (SEQ ID NO: 5074), or TGWS (SEQ ID NO: 5073). 92. The AAV particle of any one of embodiments 87-91, wherein [N4] is or comprises TGWV (SEQ ID NO: 5066). 93. The AAV particle of any one of embodiments 87-92, wherein [N1]-[N2]-[N3]-[N4] is or comprises: (i) the amino acid sequence of any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336; (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four substitutions relative to any one of the amino acid sequences in (i). 94. The AAV particle of any one of embodiments 87-93, wherein [N1]-[N2]-[N3]-[N4] is or comprises TNTQDWHRIAQTGWV (SEQ ID NO: 201). 95. The AAV particle of any one of embodiments 1-94, wherein [N1]-[N2]-[N3] is present in loop VIII, optionally wherein loop VIII comprises amino acids 580-599 as numbered according to SEQ ID NO: 138 or 981.
Attorney Docket No.14640.0105-00304 96. The AAV particle of any one of embodiments 43-50 and 87-95, wherein [N4] is present in loop VIII, optionally wherein loop VIII comprises amino acids 580-599 as numbered according to SEQ ID NO: 138 or 981. 97. The AAV particle of any one of embodiments 1-96, wherein [N1] replaces amino acids 582-585 (e.g., T582, N583, H584, Q585) of SEQ ID NO: 138. 98. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, and 85-97, wherein [N1] corresponds to amino acids 582-585 (e.g., T582, N583, T584, Q585) of SEQ ID NO: 981. 99. The AAV particle of any one of embodiments 1-98, wherein [N1] is present at amino acids 582-585, as numbered according to SEQ ID NO: 138 or 981. 100. The AAV particle of any one of embodiments 1-99, wherein [N1] corresponds to positions 582-585 (e.g., T582, N583, T584, Q585) of SEQ ID NO: 981. 101. The AAV particle of any one of embodiments 1-100, wherein [N2] replaces amino acids 586-589 (e.g., S586, A587, Q588, A589) of SEQ ID NO: 138. 102. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101, wherein [N2] corresponds to amino acids 586-589 (e.g., D586, W587, H588, R589) of SEQ ID NO: 981. 103. The AAV particle of any one of embodiments 1-102, wherein [N2] is present at amino acids 586- 589, as numbered according to SEQ ID NO: 138 or 981. 104. The AAV particle of any one of embodiments 1-103, wherein [N1]-[N2] replaces amino acids 582- 589 (e.g., T582, N583, H584, Q585, S586, A587, Q588, A589) of SEQ ID NO: 138. 105. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101-104, wherein [N1]-[N2] corresponds to amino acids 582-589 (e.g., T582, N583, T584, Q585, D586, W587, H588, R589) of SEQ ID NO: 981. 106. The AAV particle of any one of embodiments 1-105, wherein [N1]-[N2] is present at amino acids 582-589, as numbered according to SEQ ID NO: 138 or 981.
Attorney Docket No.14640.0105-00304 107. The AAV particle of any one of embodiments 1-106, wherein [N3] replaces amino acids 590-592 (e.g., Q590, A591, and Q592) of SEQ ID NO: 138. 108. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101-107, wherein [N3] corresponds to amino acids 590-592 (e.g., I590, A591, and Q592) of SEQ ID NO: 981. 109. The AAV particle of any one of embodiments 1-108, wherein [N3] is present at amino acids 590- 592, as numbered according to SEQ ID NO: 138 or 981. 110. The AAV particle of any one of embodiments 1-109, wherein [N2]-[N3] replaces amino acids 586- 592 (e.g., S586, A587, Q588, A589, Q590, A591, and Q592) of SEQ ID NO: 138. 111. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101-110, wherein [N2]-[N3] corresponds to amino acids 586-592 (e.g., D586, W587, H588, R589, I590, A591, and Q592) of SEQ ID NO: 981. 112. The AAV particle of any one of embodiments 1-111, wherein [N2]-[N3] is present at amino acids 586-592, as numbered according to SEQ ID NO: 138 or 981. 113. The AAV particle of any one of embodiments 1-112, wherein [N1]-[N2]-[N3] replaces amino acids 582-592 (e.g., T582, N583, H584, Q585, S586, A587, Q588, A589, Q590, A591, Q592) of SEQ ID NO: 138. 114. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101-113, wherein [N1]-[N2]-[N3] corresponds to amino acids 582-592 (e.g., T582, N583, T584, Q585, D586, W587, H588, R589, I590, A591, Q592) of SEQ ID NO: 981. 115. The AAV particle of any one of embodiments 1-114, wherein [N1]-[N2]-[N3] is present at amino acids 582-592, as numbered according to SEQ ID NO: 138 or 981. 116. The AAV particle of any one of embodiments 43-50 and 87-115, wherein [N4] replaces amino acids 593-596 (e.g., T593, G594, W595, and V596) of SEQ ID NO: 138. 117. The AAV particle of any one of embodiments 43-49 and 87-115, wherein [N4] corresponds to amino acids 593-596 (e.g., T593, G594, W595, and V596) of SEQ ID NO: 138 or 981.
Attorney Docket No.14640.0105-00304 118. The AAV particle of any one of embodiments 43-50 and 87-117, wherein [N4] is present at amino acids 593-596, as numbered according to SEQ ID NO: 138 or 981. 119. The AAV particle of any one of embodiments 43-50 and 87-118, wherein [N2]-[N3]-[N4] replaces amino acids 586-596 (e.g., S586, A587, Q588, A589, Q590, A591, Q592, T593, G594, W595, and V596) of SEQ ID NO: 138. 120. The AAV particle of any one of embodiments 43-49 and 87-119, wherein [N2]-[N3]-[N4] corresponds to amino acids 586-596 (e.g., D586, W587, H588, R589, I590, A591, Q592, T593, G594, W595, and V596) of SEQ ID NO: 981. 121. The AAV particle of any one of embodiments 43-50 and 87-120, wherein [N2]-[N3]-[N4] is present at amino acids 586-596, as numbered according to SEQ ID NO: 138 or 981. 122. The AAV particle of any one of embodiments 43-50 and 87-121, wherein [N1]-[N2]-[N3]-[N4] replaces amino acids 582-596 (e.g., T582, N583, H584, Q585, S586, A587, Q588, A589, Q590, A591, Q592, T593, G594, W595, and V596), as numbered according to SEQ ID NO: 138. 123. The AAV particle of any one of embodiments 43-49 and 87-122, wherein [N1]-[N2]-[N3]-[N4] corresponds to amino acids 582-596 (e.g., T582, N583, T584, Q585, D586, W587, H588, R589, I590, A591, Q592, T593, G594, W595, and V596) of SEQ ID NO: 981. 124. The AAV particle of any one of embodiments 43-50 and 87-123, wherein [N1]-[N2]-[N3]-[N4] is present at amino acids 582-596, as numbered according to SEQ ID NO: 138 or 981. 125. The AAV particle of any one of embodiments 1-124, wherein [N2] is present immediately subsequent to [N1]. 126. The AAV particle of any one of embodiments 1-125, wherein [N3] is present immediately subsequent to [N2]. 127. The AAV particle of any one of embodiments 43-50 and 87-126, wherein [N4] is present immediately subsequent to [N3]. 128. The AAV particle of any one of embodiments 1-127, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3].
Attorney Docket No.14640.0105-00304 129. The AAV particle of any one of embodiments 43-50 and 87-128, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3]-[N4]. 130. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising: (a) the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, 9, 23, 24, or 25; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, or at least 14 consecutive amino acids from any one of the sequences provided in Table 1, 2A, 2B, 9, 23, 24, or 25; or (c) an amino acid sequence comprising at least one, at least two, or at least three but no more than four substitutions relative to any one of the sequences provided in Table 1, 2A, 2B, 9, 23, 24, or 25; or (d) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, 9, 23, 24, or 25. 131. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising: (a) the amino acid sequence of any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, or at least 14 consecutive amino acids from any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336; (c) an amino acid sequence comprising at least one, at least two, or at least three but no more than four substitutions, relative to the amino acid sequence of any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336; or (d) an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336.
Attorney Docket No.14640.0105-00304 132. The AAV particle of embodiment 130 or 131, wherein the AAV capsid variant does not comprise at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, or at least 9 consecutive amino acids from TNHQSAQAQ (SEQ ID NO: 5100), optionally wherein the TNHQSAQAQ (SEQ ID NO: 5100) corresponds to amino acids 582-592 of SEQ ID NO: 138. 133. The AAV particle of any one of embodiments 130-132, wherein the AAV capsid variant does not comprise: TNH, TNHQ (SEQ ID NO: 4760), TNHQS (SEQ ID NO: 5101), TNHQSA (SEQ ID NO: 5102), TNHQSAQ (SEQ ID NO: 5103), TNHQSAQA (SEQ ID NO: 5104), TNHQSAQAQ (SEQ ID NO: 5100), NHQ, NHQS (SEQ ID NO: 5105), NHQSA (SEQ ID NO: 5106), NHQSAQ (SEQ ID NO: 5107), NHQSAQA (SEQ ID NO: 5108), NHQSAQAQ (SEQ ID NO: 5109), HQS, HQSA (SEQ ID NO: 5110), HQSAQ (SEQ ID NO: 5111), HQSAQA (SEQ ID NO: 5112), HQSAQAQ (SEQ ID NO: 5113), QSA, QSAQ (SEQ ID NO: 5114), QSAQA (SEQ ID NO: 5115), QSAQAQ (SEQ ID NO: 5116), SAQA (SEQ ID NO: 5117), or SAQAQ (SEQ ID NO: 5118). 134. The AAV particle of any one of embodiments 130-124, wherein the AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, or at least 14 consecutive amino acids from any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336. 135. The AAV particle of any one of embodiments 130-134, wherein the at least 3 consecutive amino acids comprise TQD. 136. The AAV particle of any one of embodiments 130-135, wherein the at least 4 consecutive amino acids comprise TQDW (SEQ ID NO: 4684). 137. The AAV particle of any one of embodiments 130-136, wherein the at least 5 consecutive amino acids comprise TQDWH (SEQ ID NO: 4685). 138. The AAV particle of any one of embodiments 130-137, wherein the at least 6 consecutive amino acids comprise TQDWHR (SEQ ID NO: 4686). 139. The AAV particle of any one of embodiments 130-138, wherein the at least 7 consecutive amino acids comprise TQDWHRI (SEQ ID NO: 941). 140. The AAV particle of any one of embodiments 130-134, wherein the at least 3 consecutive amino acids comprise TNT.
Attorney Docket No.14640.0105-00304 141. The AAV particle of any one of embodiments 130-134 and 140, wherein the at least 4 consecutive amino acids comprise TNTQ (SEQ ID NO: 4688). 142. The AAV particle of any one of embodiments 130-134, 140, and 141, wherein the at least 5 consecutive amino acids comprise TNTQD (SEQ ID NO: 5119). 143. The AAV particle of any one of embodiments 130-134 and 140-142, wherein the at least 6 consecutive amino acids comprise TNTQDW (SEQ ID NO: 5120). 144. The AAV particle of any one of embodiments 130-134 and 140-143, wherein the at least 7 consecutive amino acids comprise TNTQDWH (SEQ ID NO: 5121). 145. The AAV particle of any one of embodiments 130-134 and 140-144, wherein the at least 8 consecutive amino acids comprise TNTQDWHR (SEQ ID NO: 4898). 146. The AAV particle of any one of embodiments 130-134 and 140-145, wherein the at least 9 consecutive amino acids comprise TNTQDWHRI (SEQ ID NO: 746). 147. The AAV particle of any one of embodiments 130-146, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336. 148. The AAV particle of any one of embodiments 130-147, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of TQDWHRI (SEQ ID NO: 941). 149. The AAV particle of any one of embodiments 130-147, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of TNTQDWHRI (SEQ ID NO: 746). 150. The AAV particle of any one of embodiments 130-149, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four modifications relative to the amino acid sequence of ATNTQDWHRIAQT (SEQ ID NO: 744).
Attorney Docket No.14640.0105-00304 151. The AAV particle of any one of embodiments 130-150, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four substitutions relative to the amino acid sequence of any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336. 152. The AAV particle of any one of embodiments 130-149 and 151, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four substitutions relative to the amino acid sequence of TQDWHRI (SEQ ID NO: 941). 153. The AAV particle of any one of embodiments 130-149, 151, and 152, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four substitutions relative to the amino acid sequence of TNTQDWHRI (SEQ ID NO: 746). 154. The AAV particle of any one of embodiments 130-153, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three but no more than four substitutions relative to the amino acid sequence of ATNTQDWHRIAQT (SEQ ID NO: 744). 155. The AAV particle of any one of embodiments 1-154, wherein the AAV capsid variant comprises the amino acid sequence of any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336. 156. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101-155, wherein the AAV capsid variant comprises the amino acid sequence of TQDWHRI (SEQ ID NO: 941), optionally wherein the amino acid sequence replaces amino acids 584-590 of SEQ ID NO: 138. 157. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101-156, wherein the AAV capsid variant comprises the amino acid sequence of TQDWHRI (SEQ ID NO: 941), optionally wherein the amino acid sequence corresponds to amino acids 584-590 of SEQ ID NO: 981. 158. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, 101-149, 151-153, and 155-157, wherein the AAV capsid variant comprises an amino acid sequence encoded by: (i) the nucleotide sequence of SEQ ID NO: 942;
Attorney Docket No.14640.0105-00304 (ii) a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 942; or (iii) a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 942. 159. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101-158, wherein the AAV capsid variant comprises an amino acid sequence encoded by: (i) the nucleotide sequence of SEQ ID NO: 747; (ii) a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 747; or (iii) a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 747. 160. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101-149, 151-153, and 155-158, wherein the nucleotide sequence encoding the AAV capsid variant comprises: (i) the nucleotide sequence of SEQ ID NO: 942; (ii) a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 942; or (iii) a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 942. 161. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101-160, wherein the nucleotide sequence encoding the AAV capsid variant comprises: (i) the nucleotide sequence of SEQ ID NO: 747; (ii) a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 747; or (iii) a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 747.
Attorney Docket No.14640.0105-00304 162. The AAV particle of any one of embodiments 130-157, wherein the amino acid sequence provided in Table 1, 2A, 2B, 9, 23, 24, or 25 is present in loop VIII, optionally wherein loop VIII comprises amino acids 580-599, as numbered according to SEQ ID NO: 138 or 981. 163. The AAV particle of any one of embodiments 130-157, wherein the amino acid sequence provided in Table 1, 2A, 2B, 9, 23, 24, or 25 replaces amino acids 584, 585, 586, 587, 588, 589, and/or 590 (e.g., H584, Q585, S586, A587, Q588, A589, and/or Q590), as numbered according to the amino acid sequence of SEQ ID NO: 138. 164. The AAV particle of any one of embodiments 130-157, wherein the amino acid sequence provided in Table 1, 2A, 2B, 9, 23, 24, or 25 is present at amino acids 584, 585, 586, 587, 588, 589, and/or 590, as numbered according to the amino acid sequence of SEQ ID NO: 981. 165. The AAV particle of any one of embodiments 130-157, wherein the amino acid sequence provided in Table 1, 2A, 2B, 9, 23, 24, or 25 corresponds to amino acids 584-590 (e.g., T584, Q585, D586, W587, H588, R589, and/or I590), numbered according to the amino acid sequence of SEQ ID NO: 981. 166. The AAV particle of any one of embodiments 121-157, wherein the amino acid sequence provided in Table 1, 2A, 2B, 9, 23, 24, or 25 replaces amino acids 582, 583, 584, 585, 586, 587, 588, 589, and/or 590 (e.g., T582, N583, H584, Q585, S586, A587, Q588, A589, and/or Q590) of SEQ ID NO: 138. 167. The AAV particle of any one of embodiments 121-157, wherein the amino acid sequence provided in Table 1, 2A, 2B, 9, 23, 24, or 25 is present at amino acids 582, 583, 584, 585, 586, 587, 588, 589, and/or 590, as numbered according to the amino acid sequence of SEQ ID NO: 981. 168. The AAV particle of any one of embodiments 121-157, wherein the amino acid sequence provided in Table 1, 2A, 2B, 9, 23, 24, or 25 replaces amino acids 582, 583, 584, 585, 586, 587, 588, 589, 590, 591, 592, 593, 594, 595, and/or 596 (e.g., T582, N583, H584, Q585, S586, A587, Q588, A589, Q590, A591, Q592, T593, G594, W595, and/or V5965) of SEQ ID NO: 138. 169. The AAV particle of any one of embodiments 121-157, wherein the amino acid sequence provided in Table 1, 2A, 2B, 9, 23, 24, or 25 is present at amino acids 582, 583, 584, 585, 586, 587, 588, 589, 590, 591, 592, 593, 594, 595, and/or 596, as numbered according to the amino acid sequence of SEQ ID NO: 981.
Attorney Docket No.14640.0105-00304 170. The AAV particle of any one of embodiments 1-169, wherein the AAV capsid variant comprises one, two, three, four, five, or all of: (i) an amino acid other than H at position 584 (e.g., T); (ii) an amino acid other than S at position 586 (e.g., D); (iii) an amino acid other than A at position 587 (e.g., W); (iv) an amino acid other than Q at position 588 (e.g., H); (v) an amino acid other than A at position 589 (e.g., R); and/or (vi) an amino acid other than Q at position 590 (e.g., I); wherein the amino acids in (i)-(vi) are numbered according to SEQ ID NO: 138. 171. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising one, two, three, four, five, or all of: (i) an amino acid other than H at position 584 (e.g., T); (ii) an amino acid other than S at position 586 (e.g., D); (iii) an amino acid other than A at position 587 (e.g., W); (iv) an amino acid other than Q at position 588 (e.g., H); (v) an amino acid other than A at position 589 (e.g., R); and/or (vi) an amino acid other than Q at position 590 (e.g., I); wherein the amino acids in (i)-(vi) are numbered according to SEQ ID NO: 138. 172. The AAV particle of any one of embodiments 1-171, wherein the AAV capsid variant comprises one, two, three, four, five, or all of: T at position 584, D at position 586, W at position 587, H at position 588, R at position 589, and/or I at position 590, as numbered according to SEQ ID NO: 981. 173. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising one, two, three, four, five, or all of: T at position 584, D at position 586, W at position 587, H at position 588, R at position 589, and/or I at position 590, as numbered according to SEQ ID NO: 981. 174. The AAV particle of any one of embodiments 1-173, wherein the AAV capsid variant comprises one, two, three, four, five, or all of the substitutions H584T, S586D, A587W, Q588H, A589R, and/or Q590I, as numbered according to SEQ ID NO: 138. 175. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9
Attorney Docket No.14640.0105-00304 capsid variant) comprising one, two, three, four, five or all of the substitutions H584T, S586D, A587W, Q588H, A589R, and/or Q590I, as numbered according to SEQ ID NO: 138. 176. The AAV particle of any one of embodiments 1-175, wherein the AAV capsid variant comprises an amino acid other than H at position 584 (e.g., T), an amino acid other than S at position 586 (e.g., D), an amino acid other than A at position 587 (e.g., W), an amino acid other than Q at position 588 (e.g., H), an amino acid other than A at position 589 (e.g., R), and an amino acid other than Q at position 590 (e.g., I), as numbered according to SEQ ID NO: 138. 177. The AAV particle of any one of embodiments 1-176, wherein the AAV capsid variant comprises T at position 584, D at position 586, W at position 587, H at position 588, R at position 589, and I at position 590, as numbered according to SEQ ID NO: 981. 178. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-98, and 100-164, wherein the AAV capsid variant comprises an amino acid at each of positions 584, 586, 587, 588, 589, and 590 that is different from the amino acid at the corresponding positions in SEQ ID NO: 138. 179. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101-178, wherein the AAV capsid variant comprises an amino acid at each of positions 582, 583, 584, 586, 587, 588, and 589 that is different from the amino acid at the corresponding positions in SEQ ID NO: 138. 180. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, and 101-179, wherein the AAV capsid variant comprises an amino acid at each of positions 582, 583, 584, 586, 587, 588, 589, 590, 591, 592, 593, 594, 595, and 596 that is different from the amino acid at the corresponding positions in SEQ ID NO: 138. 181. The AAV particle of any one of embodiments 1-181, wherein the AAV capsid variant comprises the substitutions H584T, S586D, A587W, Q588H, A589R, and Q590I, as numbered according to SEQ ID NO: 138. 182. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising T at position 584, D at position 586, W at position 587, H at position 588, R at position 589, and I at position 590, as numbered according to SEQ ID NO: 981.
Attorney Docket No.14640.0105-00304 183. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising the substitutions H584T, S586D, A587W, Q588H, A589R, and Q590I, as numbered according to SEQ ID NO: 138. 184. The AAV particle of any one of embodiments 1-183, wherein the AAV capsid variant further comprises an amino acid other than A position 581, as numbered according to SEQ ID NO: 138 or 981. 185. The AAV particle of any one of embodiments 1-184, wherein the AAV capsid variant further comprises a T at position 581 or a V at position 581, as numbered according to SEQ ID NO: 138 or 981. 186. The AAV particle of any one of embodiments 1-185, wherein the AAV capsid variant comprises the substitutions A581T or A581V, as numbered according to SEQ ID NO: 138 or 981. 187. The AAV particle of any one of embodiments 1, 3-7, 9-15, 17-20, 22-38, 40-48, 50, 130-134, 147, 151, 155, 162, 163, 166-177, and 181-186, wherein the AAV capsid variant comprises an amino acid other than Q at position 585, as numbered according to SEQ ID NO: 138. 188. The AAV particle of any one of embodiments 1, 3-7, 9-15, 17-20, 22-38, 40-48, 50, 130-134, 147, 151, 155, 162, 163, 166-177, and 181-187, wherein the AAV capsid variant comprises the amino acid K at position 585, as numbered according to SEQ ID NO: 138. 189. The AAV particle of any one of embodiments 1-189, wherein the AAV capsid variant further comprises: (i) a modification in loop I, II, IV, and/or VI; and/or (ii) a substitution at position K449, e.g., a K449R substitution, as numbered according to SEQ ID NO: 138 or 981. 190. The AAV particle of any one of embodiments 1-189, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 138. 191. The AAV particle of any one of embodiments 1-190, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than not more than 30, not more than 20, or not more than 10 substitutions relative to the amino acid sequence of SEQ ID NO: 138.
Attorney Docket No.14640.0105-00304 192. The AAV particle of any one of embodiments 1-191, wherein the AAV capsid variant an amino acid sequence with at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 138. 193. The AAV particle of embodiment 192, wherein the AAV capsid variant an amino acid sequence with at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 138. 194. The AAV particle of any one of embodiments 1-193, wherein the AAV capsid variant comprises an amino acid sequence encoded by a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 137. 195. The AAV particle of embodiment 194, wherein the AAV capsid variant comprises an amino acid sequence encoded by a sequence with at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 137. 196. The AAV particle of any one of embodiments 1-195, wherein the AAV capsid variant comprises a VP1 protein, a VP2 protein, and a VP3 protein in an about 1:1:10 ratio. 197. The AAV particle of any one of embodiments 1-196, wherein the AAV capsid variant comprises amino acids 138-736, e.g., a VP2, of SEQ ID NO: 981, or a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. 198. The AAV particle of any one of embodiments 1-197, wherein the AAV capsid variant comprises amino acids 203-736, e.g., a VP3, of SEQ ID NO: 981, or a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. 199. The AAV particle of any one of embodiments 1-198, wherein the AAV capsid variant comprises an amino acid sequence that is at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to amino acids 138-736, e.g., a VP2, of SEQ ID NO: 138.
Attorney Docket No.14640.0105-00304 200. The AAV particle of any one of embodiments 1-199, wherein the AAV capsid variant comprises an amino acid sequence that is at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to amino acids 203-736, e.g., a VP3, of SEQ ID NO: 138. 201. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, 101-186, and 189-200, wherein the AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, or all 7 consecutive amino acids from the amino acid sequence of TQDWHRI (SEQ ID NO: 941), wherein: (i) the at least 3 consecutive amino acids comprise TQD; (ii) the at least 4 consecutive amino acids comprise TQDW (SEQ ID NO: 4684); (iii) the at least 5 consecutive amino acids comprise TQDWH (SEQ ID NO: 4685); (iv) the at least 6 consecutive amino acids comprise TQDWHR (SEQ ID NO: 4686); or (v) the 7 consecutive amino acids comprise TQDWHRI (SEQ ID NO: 941); wherein the AAV capsid variant comprises: (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 981; (b) a VP2 protein comprising amino acids 138-736 of SEQ ID NO: 981; (c) a VP3 protein comprising amino acids 203-736 of SEQ ID NO: 981; or (d) an amino acid sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to any one of the amino acid sequences in (a)-(c). 202. The AAV particle of embodiment 201, wherein the AAV capsid variant comprises an amino acid sequence with at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to any one of the amino acid sequences in (a)-(c). 203. The AAV particle of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85-99, 101-186, and 189-202, wherein the AAV capsid variant comprises 1, 2, or 3 substitutions relative to the amino acid sequence of TQDWHRI (SEQ ID NO: 941), wherein the AAV capsid variant comprises an amino acid sequence at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. 204. The AAV particle of any one of embodiments 1-203, wherein the AAV capsid variant comprises the amino acid sequence of any one of SEQ ID NO: 981, or an amino acid sequence with at least 90% (e.g.,
Attorney Docket No.14640.0105-00304 at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. 205. The AAV particle of any one of embodiments 1-204, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20 or not more than 10 modifications relative to the amino acid sequence of SEQ ID NO: 981. 206. The AAV particle of any one of embodiments, 1-205, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two or at least three, but not more than 30, not more than 20 or not more than 10 substitutions, relative to the amino acid sequence of SEQ ID NO: 981. 207. The AAV particle of any one of embodiments 1-206, wherein the AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 983, or a nucleotide sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. 208. The AAV particle of embodiment 207, wherein the AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 983. 209. The AAV particle of embodiment 207, wherein the AAV capsid variant is encoded by a codon- optimized nucleotide sequence. 210. An AAV particle comprising an AAV capsid variant (e.g., an AAV9 capsid variant) comprising the amino acid sequence of any one of embodiments 1-3, 5-8, 10-16, 18-21, 23-39, 42-49, 51-69, 71-83, 85- 99, 101-186, and 189, and further comprising an amino acid sequence at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to SEQ ID NO: 981. 211. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising the amino acid sequence of SEQ ID NO: 981. 212. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 983, or a nucleotide sequence at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
Attorney Docket No.14640.0105-00304 213. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising amino acids 203-736 of SEQ ID NO: 981, or an amino acid sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto, wherein the AAV capsid variant comprises T at position 584, D at position 586, W at position 587, H at position 588, R at position 589, and I at position 590, as numbered according to SEQ ID NO: 981. 214. The AAV particle of embodiment 213, wherein the AAV capsid variant comprises amino acids 203- 736 of SEQ ID NO: 981. 215. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising amino acids 203-736 of SEQ ID NO: 981. 216. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising amino acids 138-736 of SEQ ID NO: 981, or an amino acid sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto, wherein the AAV capsid variant comprises T at position 584, D at position 586, W at position 587, H at position 588, R at position 589, and I at position 590, as numbered according to SEQ ID NO: 981. 217. The AAV particle any one of embodiments 213-216, wherein the AAV capsid variant comprises amino acids 138-736 of SEQ ID NO: 981. 218. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising amino acids 138-736 of SEQ ID NO: 981. 219. An AAV particle comprising a viral genome comprising a syntaxin-binding protein-1 (STXBP1)- encoding sequence (e.g., encoding a human STXBP1 protein) and an AAV capsid variant (e.g., an AAV9 capsid variant) comprising the amino acid sequence of SEQ ID NO: 981, or an amino acid sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%)identical thereto, wherein the
Attorney Docket No.14640.0105-00304 AAV capsid variant comprises T at position 584, D at position 586, W at position 587, H at position 588, R at position 589, and I at position 590, as numbered according to SEQ ID NO: 981. 220. The AAV particle any one of embodiments 213-219, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 981. 221. The AAV particle of any one of embodiments 1-220, wherein the AAV capsid variant has increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. 222. The AAV particle of any one of embodiments 1-221, wherein the AAV capsid variant transduces a brain region, e.g., sensory cortex, motor cortex, putamen, thalamus, caudate, hippocampus, and/or cerebellum; optionally wherein the level of transduction is at least 39, at least 50, at least 100, at least 120, at least 132, at least 146, at least 150, at least 161, at least 174, at least 175, at least 200, at least 225, at least 250, at least 275, at least 283, at least 300, at least 350, at least 400, at least 450, at least 500, at least 525, at least 528, or at least 550-fold greater as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138, e.g., when measured by an assay, e.g., an immunohistochemistry assay or a qPCR or ddPCR assay, e.g., as described in Example 2. 223. The AAV particle of any one of embodiments 1-222, which is enriched at least 10, at least 14, at least 20, at least 24, at least 50, at least 100, at least 150, at least 200, at least 250, at least 300, at least 350, at least 400, at least 425, at least 450, or at least 460-fold in the brain compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 1 or 3. 224. The AAV particle of any one of embodiments 1-223, which is enriched at least 200, at least 300, at least 400, at least 425, at least 450, or at least 460-fold in the brain compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 1. 225. The AAV particle of any one of embodiments 1-224, which is enriched in the brain of at least two or at least three species, e.g., a non-human primate and rodent (e.g., mouse), e.g., as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. 226. The AAV particle of any one of embodiments 1-225, which is enriched at least 2, at least 3, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50,
Attorney Docket No.14640.0105-00304 at least 55, at least 60, at least 65, at least 70, at least 75, at least 80, at least 85, at least 90, at least 95, at least 100, at least 105, at least 115, at least 120, at least 125, at least 130, at least 135, at least 140, at least 145, at least 150, at least 155, at least 160, at least 165, at least 170, at least 175, at least 180, at least 190, at least 200, at least 205, or at least 210-fold in the brain of at least two or at least three species, e.g., a non-human primate and rodent (e.g., mouse), compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 1 or 4. 227. The AAV particle of embodiment 225 or 226, wherein the at least two or at least three species comprise Macaca fascicularis, Chlorocebus sabaeus, Callithrix jacchus, and/or mouse (e.g., outbred mice). 228. The AAV particle of any one of embodiments 1-227, which is enriched at least 2, at least 3, at least 4, at least 5, at least 10, at least 15, at least 17, at least 20, at least 50, at least 75, at least 100, at least 103, at least 107, at least 125, at least 150, at least 200, at least 250, at least 300, at least 350, at least 400, at least 450, at least 500, at least 750, at least 1000, at least 1200-fold in the brain compared to an AAV particle comprising an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 981, e.g., when measured by an assay as described in Example 3. 229. The AAV particle of any one of embodiments 1-228, which delivers an increased level of STXBP1 to a brain region, optionally wherein the STXBP1 level is increased by at least 39, at least 50, at least 100, at least 120, at least 132, at least 146, at least 150, at least 161, at least 174, at least 175, at least 200, at least 225, at least 250, at least 275, at least 283, at least 300, at least 350, at least 400, at least 450, at least 500, at least 525, at least 528, or at least 550-fold as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138, e.g., when measured by an assay, e.g., a qRT-PCR, a ddPCR, or a qPCR assay (e.g., as described in Example 2). 230. The AAV particle of any one of embodiments 1-229, which delivers an increased level of viral genomes to a brain region, optionally wherein the level of viral genomes is increased by at least 2, at least 5, at least 7, at least 10, at least 15, at least 19, at least 20, at least 22, or at least 25-fold as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138, e.g., when measured by an assay, e.g., a qRT-PCR or a qPCR assay (e.g., as described in Example 2). 231. The AAV particle of embodiment 229 or 230, wherein the brain region is a sensory cortex, motor cortex, putamen, thalamus, caudate, hippocampus, and/or cerebellum.
Attorney Docket No.14640.0105-00304 232. The AAV particle of any one of embodiments 1-231, which is enriched at least 5, at least 10, at least 50, at least 100, at least 115, at least 120, at least 150, at least 175, at least 200, at least 207, at least 225, at least 250, or at least 275-fold in the spinal cord compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 1 or 2. [Embodiments 233-311 are intentionally absent.] 312. The AAV particle of any one of embodiments 1-232, wherein the viral genome is single-stranded. 313. The AAV particle of any one of embodiments 1-232, wherein the viral genome is self- complementary. 314. The AAV particle of any one of embodiments 1-232, embodiment 312, or embodiment 313, wherein the viral genome further comprises a nucleotide sequence encoding a Rep protein, e.g., a non-structural protein, wherein the Rep protein comprises a Rep78 protein, a Rep68 protein, a Rep52 protein, and/or a Rep40 protein (e.g., a Rep78 protein and a Rep52 protein). 315. The AAV particle of any one of embodiments 1-232, embodiment 312, or embodiment 313, wherein the AAV particle further comprises a nucleotide sequence encoding a Rep protein, e.g., a non-structural protein, wherein the Rep protein comprises a Rep78 protein, a Rep68 protein, a Rep52 protein, and/or a Rep40 protein (e.g., a Rep78 protein and a Rep52 protein). 316. The AAV particle of embodiment 314 or 315, wherein the Rep78 protein, the Rep68 protein, the Rep52 protein, and/or the Rep40 protein is encoded by at least one Rep gene. 317. The AAV particle of any one of embodiments 1-232 and 312-316, wherein the AAV particle is an isolated AAV particle. 318. The AAV particle of any one of embodiments 1-232 and 312-317, wherein the AAV particle is a recombinant AAV particle. 319. A cell, e.g., a host cell, comprising the AAV particle of any one of embodiments 1-232 and 312- 318. 320. The cell of embodiment 319, wherein the cell is a mammalian cell or an insect cell.
Attorney Docket No.14640.0105-00304 321. The cell of embodiment 319 or 320, wherein the cell is a cell of a brain region or a spinal cord region, optionally wherein the cell is a cell of the sensory cortex, motor cortex, putamen, thalamus, caudate, hippocampus, or cerebellum. [Embodiments 322-325 are intentionally absent] 326. A pharmaceutical composition comprising the AAV particle of any one of embodiments 1-232 and 312-318, and a pharmaceutically acceptable excipient. 327. A method of delivering STXBP1 to a cell or tissue (e.g., a CNS cell or CNS tissue), comprising administering an effective amount of the pharmaceutical composition of embodiment 326 or the AAV particle of any one of embodiments 1-232 and 312-318. 328. The method of embodiment 327, wherein the cell is a cell of a brain region or a spinal cord region, optionally a cell of the frontal cortex, sensory cortex, motor cortex, caudate, cerebellar cortex, cerebral cortex, brain stem, hippocampus, or thalamus. 329. The method of embodiment 327 or 328, wherein the cell is a neuron, a sensory neuron, and/or a motor neuron. 330. The method of any one of embodiments 327-329, wherein the cell or tissue is within a subject. 331. The method of embodiment 330, wherein the subject has, has been diagnosed with having, or is at risk of having a genetic disorder. 332. The method of embodiment 330 or embodiment 331, wherein the subject has, has been diagnosed with having, or is at risk of having a neurological disorder. 333. The method of embodiment 330 or embodiment 331, wherein the subject has, has been diagnosed with having, or is at risk of having a neurodegenerative disorder. 334. The method of embodiment 330 or embodiment 331, wherein the subject has, has been diagnosed with having, or is at risk of having a muscular disorder or a neuromuscular disorder. 335. A method of treating a subject having or diagnosed with having a genetic disorder, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 326 or the AAV particle of any one of embodiments 1-232 and 312-318.
Attorney Docket No.14640.0105-00304 336. A method of treating a subject having or diagnosed with having a neurological disorder, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 326 or the AAV particle of any one of embodiments 1-232 and 312-318. 337. A method of treating a subject having or diagnosed with having a muscular disorder or a neuromuscular disorder, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 326 or the AAV particle of any one of embodiments 1-232 and 312-318. 338. A method of treating a subject having or diagnosed with having a neurodegenerative disorder, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 326 or the AAV particle of any one of embodiments 1-232 and 312-318. 339. The method of any one of embodiments 327-338, wherein the genetic disorder, neurological disorder, neurodegenerative disorder, muscular disorder, or neuromuscular disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). 340. The method of any one of embodiments 335-339, where treating comprises prevention of progression of the disorder in the subject. 341. The method of any one of embodiments 330-340, wherein the subject is a human. 342. The method of any one of embodiments 330-341, wherein the AAV particle or the pharmaceutical composition is administered to the subject intravenously, via intra-cisterna magna injection (ICM), intracerebrally, intrathecally, intracerebroventricularly, via intraparenchymal administration, intraarterially, or intramuscularly. 343. The method of any one of embodiments 330-342, wherein the AAV particle or pharmaceutical composition is administered to the subject via focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration.
Attorney Docket No.14640.0105-00304 344. The method of any one of embodiments 330-343, wherein the AAV particle or pharmaceutical composition is administered to the subject intravenously. 345. The method of any one of embodiments 330-344, wherein the AAV particle or pharmaceutical composition is administered to the subject via intra-cisterna magna injection (ICM). 346. The method of any one of embodiments 330-345, wherein the AAV particle or pharmaceutical composition is administered to the subject intraarterially. 347. The method of any one of embodiments 342-346, wherein administration of the AAV particle or pharmaceutical composition results in an increased expression of an STXBP1 gene, mRNA, protein, or combination thereof. 348. The method of any one of embodiments 342-346, wherein administration of the AAV particle or pharmaceutical composition results in an increased activity of an STXBP1 gene, mRNA, protein, or a combination thereof. 349. The pharmaceutical composition of embodiment 326 or the AAV particle of any one of embodiments 1-232 and 312-318, for use in a method of delivering STXBP1 to a cell or tissue. 350. The pharmaceutical composition of embodiment 326 or the AAV particle of any one of embodiments 1-232 and 312-318, for use in a method of treating a genetic disorder, a neurological disorder, a neurodegenerative disorder, a muscular disorder, or a neuromuscular disorder. 351. The pharmaceutical composition of embodiment 326 or the AAV particle of any one of embodiments 1-232 and 312-318, for use in the manufacture of a medicament. 352. Use of the pharmaceutical composition of embodiment 326 or the AAV particle of any one of embodiments 1-232 and 312-318 in the manufacture of a medicament. 353. Use of the pharmaceutical composition of embodiment 326 or the AAV particle of any one of embodiments 1-232 and 312-318 in the manufacture of a medicament for treating a genetic disorder, a neurological disorder, a neurodegenerative disorder, a muscular disorder, or a neuromuscular disorder. 354. An adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome, wherein the viral genome comprises a syntaxin-binding protein 1 (STXBP1)-encoding sequence and the
Attorney Docket No.14640.0105-00304 AAV capsid variant comprises an amino acid sequence having the formula [N1]-[N2]-[N3] (SEQ ID NO: 4681) in loop VIII; wherein [N2] comprises the amino acid sequence of DWHR (SEQ ID NO: 4682); and wherein: (i) [N1] comprises amino acids X
1, X
2, X
3, and X
4, wherein X
4 is Q, K, E, S, P, R, N, or H; and/or (ii) [N3] comprises amino acids X
5, X
6, and X
7, wherein X
5 is I, V, T, M, S, N, L, or F. 355. The AAV particle of embodiment 354, wherein the AAV capsid variant is an AAV9 capsid variant. 356. The AAV particle of embodiment 354 or embodiment 355, wherein the AAV capsid variant comprises an amino acid sequence that is at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to amino acids 203-736 of SEQ ID NO: 981. 357. The AAV particle of any one of embodiments 354-356, wherein loop VIII comprises amino acids 580-599 as numbered according to SEQ ID NO: 981. 358. The AAV particle of any one of embodiments 354-357, wherein: (i) X
1 is T, S, R, A, I, C, N, K, L, or Q; (ii) X
2 is N, T, G, V, S, Y, K, I, H, D, or F; and (iii) X
3 is T, N, K, D, I, S, P, A, Y, E, V, L, M, R, H, Q, or C. 359. The AAV particle of any one of embodiments 354-358, wherein: (i) X
6 is A, Y, P, N, S, T, G, E, V, W, F, or Q; and (ii) X
7 is Q, G, N, K, H, R, E, L, P, or M. 360. The AAV particle of any one of embodiments 354-359, wherein [N1] comprises the amino acid sequence of TNTQ (SEQ ID NO: 4688). 361. The AAV particle of any one of embodiments 354-360, wherein [N3] comprises the amino acid sequence of IAQ. 362. The AAV particle of any one of embodiments 354-361, wherein [N1]-N2]-[N3] comprises the amino acid sequence of TNTQDWHRIAQ (SEQ ID NO: 343). 363. The AAV particle of any one of embodiments 354-362, wherein: (i) [N1] is present at amino acids 582-585;
Attorney Docket No.14640.0105-00304 (ii) [N2] is present at amino acids 586-589; and (iii) [N3] is present at amino acids 590-592; wherein the amino acids are numbered according to SEQ ID NO: 981. 364. The AAV particle of any one of embodiments 354-363, further comprising [N4] comprising amino acids X
8, X
9, X
10, and X
11, wherein: (i) X
8 is T, S, N, P, A, or I; (ii) X
9 is G, N, D, R, V, A, S, or Q; (iii) X
10 is W, S, C, R, L, or G; and/or (iv) X
11 is V, A, S, I, C, G, D, F, L, or T. 365. The AAV particle of embodiment 364, wherein [N4] comprises the amino acid sequence of TGWV (SEQ ID NO: 5066). 366. The AAV particle of embodiment 364 or embodiment 365, wherein [N4] is present at amino acids 593-596 as numbered according to SEQ ID NO: 981. 367. The AAV particle of any one of embodiments 354-366, wherein [N1]-[N2]-[N3]-[N4] comprises the amino acid sequence of TNTQDWHRIAQTGWV (SEQ ID NO: 201). 368. An AAV particle comprising: (i) a viral genome comprising a syntaxin-binding protein 1 (STXBP1)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to amino acids 203-736 of SEQ ID NO: 981, wherein the AAV capsid variant comprises: T at position 584, D at position 586, W position 587, H at position 588, R at position 589, and I at position 590; wherein the amino acids are numbered according to SEQ ID NO: 981. 369. The AAV particle of any one of embodiments 354-368, wherein the AAV capsid variant comprises an amino acid sequence having at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identity to amino acids 138-736 of SEQ ID NO: 981.
Attorney Docket No.14640.0105-00304 370. An AAV particle comprising: (i) a viral genome comprising a syntaxin-binding protein 1 (STXBP1)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to amino acids 138-736 of SEQ ID NO: 981, wherein the AAV capsid variant comprises: T at position 584, D at position 586, W position 587, H at position 588, R at position 589, and I at position 590; wherein the amino acids are numbered according to SEQ ID NO: 981. 371. The AAV particle of any one of embodiments 354-370, wherein the AAV capsid variant comprises an amino acid sequence having at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identity to SEQ ID NO: 981. 372. An AAV particle comprising: (i) a viral genome comprising a syntaxin-binding protein 1 (STXBP1)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence that is at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to SEQ ID NO: 981, wherein the AAV capsid variant comprises: T at position 584, D at position 586, W position 587, H at position 588, R at position 589, and I at position 590; wherein the amino acids are numbered according to SEQ ID NO: 981. 373. The AAV particle of embodiment 372, wherein the AAV capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO:981; (ii) a VP2 protein comprising an amino acid sequence having at least 99% identity to amino acids138-736 of SEQ ID NO: 981; and/or (iii) a VP3 protein comprising an amino acid sequence having at least 99% identity to amino acids 203-736 of SEQ ID NO: 981.
Attorney Docket No.14640.0105-00304 374. The AAV particle of any one of embodiments 368-373, wherein the AAV capsid variant comprises the amino acid sequence of TQDWHRI (SEQ ID NO: 941). 375. The AAV particle of any one of embodiments 368-374, wherein the AAV capsid variant comprises at least 8, at least 9, or at least 10 consecutive amino acids from the amino acid sequence of TNTQDWHRIAQ (SEQ ID NO: 343). 376. The AAV particle of any one of embodiments 368-375, wherein the AAV capsid variant comprises the amino acid sequence of TNTQDWHRIAQ (SEQ ID NO: 343) at amino acids 582-592 as numbered according to SEQ ID NO: 981. 377. The AAV particle of any one of embodiments 374-376, wherein the amino acid sequence of TQDWHRI (SEQ ID NO: 941) or TNTQDWHRIAQ (SEQ ID NO: 343) is present in loop VIII, wherein loop VIII comprises amino acids 580-599 as numbered according to SEQ ID NO: 981. 378. The AAV particle of any one of embodiments 354-377, wherein the AAV capsid variant comprises: (i) a VP1 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 981; (ii) a VP2 protein comprising or consisting of the amino acid sequence according to positions 138-736 of SEQ ID NO: 981; and/or (iii) a VP3 protein comprising or consisting of the amino acid sequence according to positions 203-736 of SEQ ID NO: 981. 379. The AAV particle of any one of embodiments 1-232, 312-318, and 354-378, wherein the viral genome encodes a wildtype STXBP1 protein. 380. The AAV particle of any one of embodiments 1-232, 312-318, and 354-379, wherein the viral genome encodes a human STXBP1 protein. 381. The AAV particle of embodiment 379 or embodiment 380, wherein the STXBP1 protein comprises the amino acid sequence of SEQ ID NO: 6413. 382. The AAV particle of any one of embodiments 1-232, 312-318, and 354-381, wherein the STXBP1- encoding sequence is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to SEQ ID NO: 6414.
Attorney Docket No.14640.0105-00304 383. The AAV particle of embodiment 382, wherein the STXBP1-encoding sequence is at least 95% identical to SEQ ID NO: 6414. 384. The AAV particle of embodiment 383, wherein the STXBP1-encoding sequence is at least 99% identical to SEQ ID NO: 6414. 385. The AAV particle of embodiment 384, wherein the STXBP1-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 6414. 386. The AAV particle of embodiment 385, wherein the STXBP1-encoding sequence consists of the nucleotide sequence of SEQ ID NO: 6414. 387. The AAV particle of any one of embodiments 1-232, 312-318, and 354-386, wherein the viral genome comprises a promoter operably linked to the STXBP1-encoding sequence. 388. The AAV particle of any one of embodiments 1-232, 312-318, and 354-387, wherein the viral genome further comprises an inverted terminal repeat (ITR) sequence. 389. The AAV particle of embodiment 388, wherein the viral genome comprises an ITR sequence positioned 5’ relative to the STXBP1-encoding sequence. 390. The AAV particle of embodiment 388 or embodiment 389, wherein the viral genome comprises an ITR sequence positioned 3’ relative to the STXBP1-encoding sequence. 391. The AAV particle of any one of embodiments 388-390, wherein the viral genome comprises an ITR sequence positioned 5’ relative to the STXBP1-encoding sequence, and an ITR sequence positioned 3’ relative to the STXBP1-encoding sequence. 392. A cell comprising the AAV particle of any one of embodiments 1-232, 312-318, and 354-391, optionally wherein the cell is a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial cell. 393. A method of making the AAV particle of any one of embodiments 1-232, 312-318, and 354-391, wherein the method comprises: (i) providing a cell comprising a viral genome comprising an STXBP1-encoding sequence and a nucleic acid encoding an AAV capsid variant; and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV
Attorney Docket No.14640.0105-00304 capsid variant; thereby making the AAV particle. 394. The method of embodiment 393, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 6414 or a nucleotide sequence that is at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto; and the capsid variant comprises the amino acid sequence of SEQ ID NO: 981. 395. The method of embodiment 393 or embodiment 394, further comprising, prior to step (i), introducing a nucleic acid comprising the viral genome into the cell. 396. The method of any one of embodiments 393-395, further comprising, prior to step (i), introducing the nucleic acid encoding the AAV capsid variant into the cell. 397. The method of any one of embodiments 393-396, wherein the cell comprises a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial cell. 398. A pharmaceutical composition comprising the AAV particle of any one of embodiments 1-232, 312- 318, and 354-391 and a pharmaceutically acceptable excipient. 399. A method of delivering an STXBP1 protein to a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 398 or the AAV particle of any one of embodiments 1-232, 312-318, and 354-391, thereby delivering the STXBP1 protein. 400. The method of embodiment 399, wherein the subject has, has been diagnosed with having, or is at risk of having an STXBP1-related disorder, optionally wherein the STXBP1-related disorder is an STXBP1-related neurodegenerative or neuromuscular disorder. 401. The method of embodiment 400, wherein the STXBP1-related neurodegenerative or neuromuscular disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5).
Attorney Docket No.14640.0105-00304 402. A method of treating an STXBP1-related disorder in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 398 or the AAV particle of any one of embodiments 1-232, 312-318, and 354-391, thereby treating the STXBP1-related disorder. 403. The method of embodiment 402, wherein the subject has, has been diagnosed with having, or is at risk of having an STXBP1-related disorder. 404. The method of embodiment 403, wherein the STXBP1-related disorder is an STXBP1-related neurodegenerative or neuromuscular disorder. 405. The method of embodiment 404, wherein the STXBP1-related neurodegenerative or neuromuscular disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). 406. A method of treating an STXBP1-related disorder in a subject, wherein the disorder is STXBP1 encephalopathy, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 398 or the AAV particle of any one of embodiments 1-232, 312-318, and 354-391, thereby treating STXBP1 encephalopathy. 407. The method of embodiment 406, wherein the subject has, has been diagnosed with having, or is at risk of having STXBP1 encephalopathy. 408. The method of any one of embodiments 402-407, wherein the subject has one or more mutations in the STXBP1 gene. 409. The method of any one of embodiments 402-408, wherein the subject has lower STXBP1 activity as compared to STXBP1 activity in an individual who does not have an STXBP1-related disorder. 410. The method of any one of embodiments 402-409, wherein the treating results in prevention of progression of the disorder in the subject. 411. The method of any one of embodiments 402-410, wherein the treating results in amelioration of at least one symptom of the disorder, as indicated by one or more biomarkers.
Attorney Docket No.14640.0105-00304 412. The method of embodiment 411, wherein the one or more biomarkers comprise increased release of the neurotransmitters glutamate and/or GABA or reduction in abnormal electroencephalographic activity as evidence of improved STXBP1 activity. 413. The method of embodiment 411 or embodiment 412, wherein the at least one symptom comprises epilepsy, autistic features, ataxia, generalized tremors, reduced STXBP1 activity, accumulation of glucocerebroside and other glycolipids, e.g., within immune cells (e.g., macrophages), build-up of synuclein aggregates (e.g., Lewy bodies), developmental delay, progressive encephalopathy, progressive dementia, ataxia, myoclonus, oculomotor dysfunction, bulbar palsy, generalized weakness, trembling of a limb, depression, visual hallucinations, cognitive decline, dystonia, or a combination thereof. 414. The method of any one of embodiments 399-413, wherein the subject is a human subject. 415. The method of any one of embodiments 399-414, wherein the AAV particle is delivered to a cell, tissue, or region of the CNS. 416. The method of embodiment 415, wherein the cell, tissue, or region of the CNS is a cell (e.g., a neuron, e.g., a GABAergic neuron and/or a glutamatergic neuron), tissue, or region of the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region). 417. The method of any one of embodiments 399-416, further comprising evaluating, e.g., measuring, the level of STXBP1 expression, e.g., STXBP1 gene expression, STXBP1 mRNA expression, and/or STXBP1 protein expression, in the subject, e.g., in a cell, tissue, or fluid of the subject. 418. The method of embodiment 417, wherein the level of STXBP1 protein expression is measured by an ELISA, a Western blot, or an immunohistochemistry assay. 419. The method of embodiment 417 or embodiment 418, wherein evaluating the level of STXBP1 expression is performed prior to and/or subsequent to administration of the AAV particle, optionally wherein the level of STXBP1 expression prior to treatment is compared to the level of STXBP1 expression subsequent to administration.
Attorney Docket No.14640.0105-00304 420. The method of any one of embodiments 417-419, comprising evaluating the level of STXBP1 expression in a cell or tissue of the CNS. 421. The method of embodiment 420, wherein the cell or tissue of the CNS is a cell (e.g., a neuron, e.g., a GABAergic neuron and/or a glutamatergic neuron) or tissue of the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region). 422. The method of any one of embodiments 417-421, wherein the subject’s level of STXBP1 protein expression subsequent to administration is increased relative to the subject’s level of STXBP1 protein expression prior to administration. 423. The method of any one of embodiments 399-422, further comprising evaluating, e.g., measuring, the level of STXBP1 activity in the subject, e.g., in a cell or tissue of the subject. 424. The method of any one of embodiments 399-423, wherein the administration results in an increase in: (i) the level of STXBP1 activity in a cell or tissue (e.g., a cell or tissue of the CNS, e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron)), and/or fluid (e.g., CSF and/or serum) of the subject, relative to baseline and/or relative to the level of STXBP1 activity in a cell, tissue, or fluid of an individual with an STXBP1-related disorder who has not been administered the pharmaceutical composition or AAV particle; (ii) the number and/or level viral genomes (VG) per cell level in a tissue of the CNS (e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, spinal cord (e.g., cervical spinal cord region, lumbar
Attorney Docket No.14640.0105-00304 spinal cord region, or thoracic spinal cord region)), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron)) of the subject, relative to the number and/or level of VG per cell in a peripheral tissue of the subject; and/or (iii) the level of STXBP1 protein or STXBP1 gene expression in a cell or tissue (e.g., a cell or tissue of the CNS, e.g., amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron)) of the subject relative to baseline and/or relative to the level of STXBP1 protein or STXBP1 gene in a cell or tissue of an individual with an STXBP1-related disorder who has not been administered the pharmaceutical composition or AAV particle. 425. The method of any one of embodiments 399-424, further comprising administering to the subject at least one additional therapeutic agent and/or therapy. 426. The method of embodiment 425, wherein the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy for treating the STXBP1-related disorder. 427. The method of embodiment 426, wherein the at least one additional therapeutic agent and/or therapy comprises one or more anti-epileptic drugs (e.g., bromide, clobazam, felbamate, ganaxolone, lamotrigine, levetiracetam, phenobarbital, topiramate, valproate, or a combination thereof). 428. The method of any one of embodiments 399-427, further comprising administering an immunosuppressant to the subject. 429. The method of embodiment 428, wherein the immunosuppressant comprises a corticosteroid (for example, and without limitation, prednisone, prednisolone, methylprednisolone, and/or dexamethasone), adrenocorticotropic hormone, rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine. 430. The pharmaceutical composition of embodiment 398 or the AAV particle of any one of embodiments 1-232, 312-318, and 354-391 for use in a method of treating a disorder according to any one of embodiments 402-429.
Attorney Docket No.14640.0105-00304 431. The pharmaceutical composition of embodiment 398 or the AAV particle of any one of embodiments 1-232, 312-318, and 354-391, for use in the treatment of an STXBP1-related disorder in a subject; optionally wherein the STXBP1-related disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). 432. The pharmaceutical composition or AAV particle for use of embodiment 431, wherein the subject has, has been diagnosed with having, or is at risk of having the STXBP1-related disorder, optionally wherein the STXBP-related disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). 433. Use of an effective amount of the pharmaceutical composition of embodiment 398 or the AAV particle of any one of embodiments 1-232, 312-318, and 354-391 in the manufacture of a medicament for the treatment of an STXBP1-related disorder in a subject; optionally wherein the STXBP1-related disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). 434. The use of embodiment 433, wherein the subject has, has been diagnosed with having, or is at risk of having the STXBP1-related disorder, optionally wherein the STXBP1-related disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gastaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). [060] The details of various aspects or embodiments of the present disclosure are set forth below. Other features, objects, and advantages of the disclosure will be apparent from the description and the claims. In the description, the singular forms also include the plural unless the context clearly dictates otherwise. Unless defined otherwise, all technical and scientific terms used herein have the same
Attorney Docket No.14640.0105-00304 meaning as commonly understood by one of ordinary skill in the art in the field of this disclosure. In the case of conflict, the present description will control. DETAILED DESCRIPTION Overview [061] Described herein, inter alia, are compositions comprising an AAV capsid variant comprising a sequence encoding a an STXBP1 protein, e.g., a human STXBP1 protein. In some embodiments, the present disclosure provides a method of delivering the AAV capsid variant comprising the sequence encoding the STXBP1 protein to a cell or tissue in a subject. In some embodiments, the present disclosure provides a method for delivering the AAV capsid variant, thereby providing an STXBP1 protein, e.g., a human STXBP1 protein to a cell or tissue in a subject. In some embodiments, the AAV capsid variants described herein have enhanced CNS tropism compared to other cells or tissues in the body, e.g., liver and/or the DRG. [062] AAVs have proven to be useful as a biological tool due to their relatively simple structure, their ability to infect a wide range of cells (including quiescent and dividing cells) without integration into the host genome and without replicating, and their relatively benign immunogenic profile. Engineered adeno-associated virus (AAV) capsids with improved brain tropism represent an attractive solution to the limitations of CNS delivery. AAV-derived vectors are promising tools for clinical gene transfer because of their non-pathogenic nature, their low immunogenic profile, low rate of integration into the host genome, and long-term transgene expression in non-dividing cells. However, the transduction efficiency of naturally occurring AAVs in certain organs is too low for clinical applications, and capsid neutralization by pre-existing neutralizing antibodies may prevent treatment of a large proportion of patients. For these reasons, considerable efforts have been devoted to obtaining capsid variants with enhanced properties. Of many approaches tested so far, significant advances have resulted from directed evolution of AAV capsids using in vitro or in vivo selection of capsid variants created by capsid sequence randomization using either error-prone PCR, shuffling of various parent serotypes, or insertion of fully randomized short peptides at defined positions. [063] The genome of the virus may be modified to contain a minimum of components for the assembly of a functional recombinant virus, or viral particle, which is loaded with or engineered to target a particular tissue and express or deliver STXBP1. The genome of the virus may encode an STXBP1 protein, and the viral particle comprising said genome may be delivered to a target cell, tissue, or organism. In some embodiments, the genome encodes a human STXBP1 protein, e.g., a wildtype human STXBP1 protein. In some embodiments, the target cell is a CNS cell. In some embodiments, the target tissue is a CNS tissue. In some embodiments, the target CNS tissue is brain tissue. [064] Gene therapy presents an alternative approach for treating an STXBP1-related disorder such as an STXBP1-related neurodegenerative or neuromuscular disorder (e.g., STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1
Attorney Docket No.14640.0105-00304 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5)). AAVs are commonly used in gene therapy approaches as a result of a number of advantageous features. Without being bound by theory, it is believed, in some embodiments, an AAV particle described herein can be used to administer and/or deliver a gene encoding STXBP1 protein (e.g., human STXBP1 protein) preferentially to the CNS. In some embodiments, an AAV particle described herein can be used to administer and/or deliver a gene encoding STXBP1 protein (e.g., human STXBP1 protein) preferentially to the brain. In some embodiments, an AAV particle described herein can be used to administer and/or deliver a gene encoding STXBP1 protein (e.g., human STXBP1 protein) preferentially to the spinal cord. [065] Provided herein are compositions and methods which may provide for improved features compared to prior AAV-mediated approaches, including (i) increased biodistribution throughout the CNS (e.g., the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron)), (ii) elevated STXBP1 expression in multiple brain regions (e.g., cortex, thalamus, and brain stem); and (iii) preferential biodistribution in the CNS over the liver and/or DRG, of the subject. [066] Also provided herein are AAV capsid variants (e.g., AAV9 capsid variants) with improved properties compared to wildtype AAV9, such as (i) increased penetrance through the blood brain barrier following intravenous administration, (ii) wider distribution throughout the multiple brain regions, e.g., the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron), (iii) elevated STXBP1 expression in multiple brain regions, (iv) wider distribution in one or more peripheral tissues, e.g., the heart, kidney, muscle (e.g., quadriceps muscle), pancreas, and/or liver, and/or (v) elevated STXBP1 expression in one or more peripheral tissues. In some embodiments, the AAV capsid variants described herein enhance the delivery of an STXBP1 to multiple regions of the CNS, such as the brain, and/or the PNS. Exemplary regions may comprise the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile
Attorney Docket No.14640.0105-00304 nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron). In some embodiments, the AAV capsid variants described herein enhance the delivery of STXBP1 to the forebrain. In some embodiments, the AAV capsid variants described herein enhance the delivery of STXBP1 to the spinal cord. In some embodiments, the AAV capsid variants described herein enhance the delivery of STXBP1 to the dentate nucleus. In some embodiments, the AAV capsid variants described herein enhance the delivery of STXBP1 to the somatosensory cortex. In some embodiments, the AAV capsid variants with enhanced brain tropism described herein increase STXBP1 mRNA expression in the brain. In some embodiments, the AAV capsid variants increase STXBP1 mRNA expression in the forebrain. [067] Thus, the compositions and methods described herein can be used in the treatment of an STXBP1-related disorder such as an STXBP1-related neurodegenerative or neuromuscular disorder (e.g., STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). In some embodiments, the STXBP1-related disorder is STXBP1 encephalopathy. In some embodiments, the disclosure provides an AAV particle comprising one of the AAV capsid variants disclosed herein and an AAV viral genome comprising a nucleotide sequence comprising a promoter and a sequence encoding an STXBP1 protein for use in treating an STXBP1-related disorder such as an STXBP1-related neurodegenerative or neuromuscular disorder (e.g., STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5)). I. Compositions Adeno-associated viral (AAV) Particles [068] AAVs have a genome of about 5,000 nucleotides in length and contains two open reading frames encoding the proteins responsible for replication (Rep) and the structural protein of the capsid (Cap). The open reading frames are flanked by two Inverted Terminal Repeat (ITR) sequences, which serve as the origin of replication of the viral genome. The wild-type AAV viral genome comprises nucleotide sequences for two open reading frames, one for the four non-structural Rep proteins (Rep78, Rep68, Rep52, Rep40, encoded by Rep genes) and one for the three capsid, or structural, proteins (VP1, VP2, VP3, encoded by capsid genes or Cap genes). The Rep proteins are important for replication and packaging, while the capsid proteins are assembled to create the protein shell of the AAV, or AAV capsid. Alternative splicing and alternate initiation codons and promoters result in the generation of four
Attorney Docket No.14640.0105-00304 different Rep proteins from a single open reading frame and the generation of three capsid proteins from a single open reading frame. Though it varies by AAV serotype, as a non-limiting example, for AAV9/hu.14 (SEQ ID NO: 123 of US 7,906,111, the contents of which are herein incorporated by reference in their entirety), VP1 refers to amino acids 1-736, VP2 refers to amino acids 138-736, and VP3 refers to amino acids 203-736. In some embodiments, with reference to the amino acid sequence of SEQ ID NO: 981, VP1 comprises amino acids 1-736, VP2 comprises amino acids 138-736, and VP3 comprises amino acids 203-736. In other words, VP1 is the full-length capsid protein sequence, while VP2 and VP3 are shorter components of the whole. As a result, changes in the sequence in the VP3 region are also changes to VP1 and VP2, however, the percent difference as compared to the parent sequence will be greatest for VP3 since it is the shortest sequence of the three. Though described here in relation to the amino acid sequence, the nucleic acid sequence encoding these proteins can be similarly described. Together, the three capsid proteins assemble to create the AAV capsid. Without being bound by theory, the AAV capsid typically comprises a molar ratio of 1:1:10 of VP1:VP2:VP3. [069] The AAV particle typically requires a co-helper (e.g., adenovirus) to undergo productive infection in cells. In the absence of such helper functions, the AAV virions essentially enter host cells but do not integrate into the cells’ genome. [070] AAV particles have been investigated for delivery of gene therapeutics because of several unique features. Non-limiting examples of the features include (i) the ability to infect both dividing and non-dividing cells; (ii) a broad host range for infectivity, including human cells; (iii) wild-type AAV has not been associated with any disease and has not been shown to replicate in infected cells; (iv) the lack of cell-mediated immune response against the particle, and (v) the non-integrative nature in a host chromosome thereby reducing potential for long-term genetic alterations. Moreover, infection with AAV particles has minimal influence on changing the pattern of cellular gene expression (Stilwell and Samulski et al., Biotechniques, 2003, 34, 148, the contents of which are herein incorporated by reference in their entirety). [071] Typically, AAV particles for STXBP1 delivery may be recombinant viral particles which are replication defective as they lack sequences encoding functional Rep and Cap proteins within the viral genome. In some cases, the replication defective AAV particles may lack most or all coding sequences and essentially only contain one or two AAV ITR sequences and a nucleic acid sequence encoding an STXBP1 protein. [072] In some embodiments, the AAV particles of the present disclosure may be introduced into mammalian cells. [073] AAV particles may be modified to enhance the efficiency of delivery. Such modified AAV particles of the present disclosure can be packaged efficiently and can be used to successfully infect the target cells at high frequency and with minimal toxicity.
Attorney Docket No.14640.0105-00304 [074] In other embodiments, AAV particles of the present disclosure may be used to deliver STXBP1 to the central nervous system (see, e.g., U.S. Pat. No.6,180,613; the contents of which are herein incorporated by reference in their entirety) or to specific tissues of the CNS. [075] It is understood that the compositions described herein may have additional conservative or non-essential amino acid substitutions, which do not have a substantial effect on their functions. [076] In some embodiments, an AAV capsid variant comprises one or more modifications in loop VIII of AAV9, e.g., at one or more amino acids corresponding to positions 580-599 of VP1 of AAV9. In some embodiments, an AAV capsid variant comprises a substitution at one, two, three, four, five, or all of amino acids corresponding to position(s) 584, 586, 587, 588, 589, and/or 590 of the amino acid sequence of SEQ ID NO: 138 or 981. In some embodiments, the term loop (e.g., loop VIII) is used interchangeably herein with the term variable region (e.g., variable region VIII), or VR (e.g., VR-VIII). In some embodiments, loop VIII comprises amino acids corresponding to positions 580-599 (e.g., the positions of amino acids VATNHQSAQAQAQTGWVQNQ (SEQ ID NO: 5122)) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, loop VIII is present (is located) at amino acids comprising those corresponding to positions 580-599 (e.g., the positions of amino acids VATNHQSAQAQAQTGWVQNQ (SEQ ID NO: 5122)) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, loop VIII comprises amino acids corresponding to positions 582-593 (e.g., the positions of amino acids TNHQSAQAQAQT (SEQ ID NO: 5123)) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, loop VIII comprises amino acids corresponding to positions 587-593 (e.g., the positions of amino acids AQAQAQT (SEQ ID NO: 4687)) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, loop VIII comprises amino acids corresponding to positions 587-590 (e.g., the positions of amino acids AQAQ (SEQ ID NO: 5099)) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, loop VIII is present at amino acids comprising those corresponding to positions 587-590 (e.g., the positions of amino acids AQAQ (SEQ ID NO: 5099)) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, loop VIII or variable region VIII (VR- VIII) is as described in DiMattia et al. “Structural Insights into the Unique Properties of the Adeno- Associated Virus Serotype 9,” Journal of Virology, 12(86):6947-6958 (the contents of which are hereby incorporated by reference in their entirety), e.g., comprising amino acids corresponding to positions 581- 593 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, loop VIII is present at amino acids comprising those corresponding to positions 581-593 of the amino acid sequence of SEQ ID NO: 138. [077] The AAV particles and payloads of the disclosure may be delivered to one or more target cells, tissues, organs, or organisms. In some embodiments, the AAV particles demonstrate enhanced tropism for a target cell type, tissue, or organ. As a non-limiting example, the AAV particle may have enhanced tropism for cells and tissues of the central or peripheral nervous systems (CNS and PNS, respectively). In some embodiments, an AAV particle may, in addition, or alternatively, have decreased tropism for a cell-type, tissue or organ.
Attorney Docket No.14640.0105-00304 [078] In some embodiments, AAV particles are used as a biological tool due to a relatively simple structure, their ability to infect a wide range of cells (including quiescent and dividing cells) without integration into the host genome and without replicating, and their relatively benign immunogenic profile. The genome of the virus may be manipulated to contain a minimum of components for the assembly of a functional recombinant virus, or viral particle, which is loaded with or engineered to target a particular tissue and express or deliver a desired payload. [079] In some embodiments, the AAV particle is a recombinant AAV particle. In some embodiments, the wild-type AAV viral genome is a linear, single-stranded DNA (ssDNA) molecule approximately 5,000 nucleotides (nt) in length. In some embodiments, inverted terminal repeats (ITRs) cap the viral genome at both the 5’ and the 3’ end, providing origins of replication for the viral genome. In some embodiments, an AAV viral genome comprises two ITR sequences. In some embodiments, the ITRs have a characteristic T-shaped hairpin structure defined by a self-complementary region (145nt in wild-type AAV) at the 5’ and 3’ ends of the ssDNA which form an energetically stable double stranded region. In some embodiments, the double stranded hairpin structures comprise multiple functions including, but not limited to, acting as an origin for DNA replication by functioning as primers for the endogenous DNA polymerase complex of the host viral replication cell. [080] AAV particles of the present disclosure may be produced recombinantly and may be based on AAV reference sequences. In addition to single-stranded AAV viral genomes (e.g., ssAAVs), the present disclosure also provides for self-complementary AAV (scAAVs) viral genomes. scAAV viral genomes contain DNA strands that anneal together to form double-stranded DNA. By skipping second strand synthesis, scAAVs allow for rapid expression in the transduced cell. In some embodiments, the AAV particle of the present disclosure is an scAAV. In some embodiments, the AAV particle of the present disclosure is an ssAAV. [081] Methods for producing and/or modifying AAV particles are disclosed in the art such as pseudotyped AAV particles (PCT Patent Publication Nos. WO200028004; WO200123001; WO2004112727; WO2005005610; and WO2005072364, the content of each of which is incorporated herein by reference in its entirety). [082] As described herein, the AAV particles of the disclosure comprising an AAV capsid variant, and a viral genome, have enhanced tropism for a cell-type or a tissue, e.g., a CNS cell-type, region, or tissue. AAV Capsid Variants [083] Disclosed herein are AAV particles comprising an AAV capsid variant comprising one or more modifications (e.g., comprising one or more substitutions relative to a wildtype AAV capsid) for enhanced or improved transduction of a target tissue (e.g., cells, regions, and/or tissues of the CNS and/or PNS). In some embodiments, the peptide (e.g., comprising one or more substitutions relative to a wildtype AAV capsid) is present in VP1, VP2, and/or VP3 proteins of the AAV capsid variant. In some
Attorney Docket No.14640.0105-00304 embodiments, the modification (e.g., comprising one or more substitutions relative to a wildtype AAV capsid) is present in VP1, VP2, and VP3 proteins of the AAV capsid variant. [084] In some embodiments, the modification (e.g., comprising the one or more substitutions relative to a wildtype AAV capsid) is in loop VIII. In some embodiments, the AAV capsid variant is an AAV9 capsid variant. [085] In some embodiments, the one or more substitutions in the AAV capsid variant may increase distribution of an AAV particle to a cell, region, or tissue of the CNS. The cell of the CNS may be, but is not limited to, neurons (e.g., excitatory, inhibitory, motor, sensory, autonomic, sympathetic, parasympathetic, Purkinje, Betz, etc.), glial cells (e.g., microglia, astrocytes, oligodendrocytes) and/or supporting cells of the brain such as immune cells (e.g., T cells). The tissue of the CNS may be, but is not limited to, the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron). [086] In some embodiments, the one or more substitutions may increase distribution of an AAV particle to a cell, region, or tissue of the CNS. In some embodiments, the one or more substitutions may decrease distribution of an AAV particle to the DRG. [087] In some embodiments, the one or more substitutions may increase distribution of an AAV particle to the CNS (e.g., the cortex) after intravenous administration. In some embodiments, the one or more substitutions may increase distribution of an AAV particle to the CNS (e.g., the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron)) following focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration. [088] In some embodiments, the one or more substitutions may increase distribution of an AAV particle to the PNS (e.g., DRG) after intravenous administration. In some embodiments, the one or more substitutions may increase distribution of an AAV particle to non-DRG cells of the PNS following focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration. In some embodiments, the one or more
Attorney Docket No.14640.0105-00304 substitutions may decrease distribution of an AAV particle to the DRG following focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration. In some embodiments, the one or more substitutions may increase distribution of an AAV particle to the PNS (e.g., DRG) following focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration. [089] In some embodiments, the one or more modifications, e.g., substitutions, may increase distribution of an AAV particle to a cell, region, or tissue of a heart, e.g., a heart atrium or a heart ventricle. In some embodiments, the one or more substitutions may increase distribution of an AAV particle to a heart cell, region, or tissue after intravenous administration. [090] In some embodiments, the one or more modifications, e.g., substitutions, may increase distribution of an AAV particle to a cell, region, or tissue of a muscle. In some embodiments, the muscle is a heart muscle (e.g., a heart atrium or a heart ventricle) or a quadriceps muscle. In some embodiments, the one or more substitutions may increase distribution of an AAV particle to a muscle cell, region, or tissue after intravenous administration. [091] In some embodiments, the one or more modifications, e.g., substitutions, may increase distribution an AAV particle to a cell, region, or tissue of the kidney. In some embodiments, the one or more substitutions may increase distribution an AAV particle to a cell, region, or tissue of the pancreas. In some embodiments, the one or more substitutions may increase distribution of an AAV particle to a cell, region, or tissue of the retina. [092] In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) a sequence as set forth in Table 1. In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) a sequence as set forth in Table 2A. In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) a sequence as set forth in Table 2B (e.g., a sequence of any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336). In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) a sequence set forth in Table 9. In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) a sequence as set forth in Table 23. In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) a sequence as set forth in Table 24. In some embodiments, the AAV capsid variant is an AAV9 capsid variant. Table 1. Exemplary Sequences Amino Acid SEQ Amino Ac SEQ SEQ SEQ ID id ID Amino Acid ID Amino Acid ID O: 0
1 2 3
4 5 6
Attorney Docket No.14640.0105-00304 T
TNQDWHRIAQ 350 TNTKDWHRISQ 399 TINQDWHRIAQ 448 TNNPDWHRIAQ 497 T
NDQDWHRIAQ 351 SNTKDWHRIAQ 400 TNNQDWHRIAH 449 TITEDWHRIAQ 498 T
TIQDWHRIAQ 352 TNNKDWHRIAQ 401 TNNQDWHRIGQ 450 TNTNDWHRIAQ 499 0 1 2 3 4 5 6 7 8 9 0 1 2 3 4 5 6 7 8
9 0 1 2 3 4 5
6 7 8 9 0 1 2
3 4 5 6 7 8
SEQ ID Amino Acid Sequence SEQ ID Nucleotide Sequence NO: NO: C
. p y q Amino Acid SEQ ID Amin A id S n SEQ ID Amin A id S n SEQ ID
Attorney Docket No.14640.0105-00304 T
TNQDWHRIAQTGWV 205 STTQDWHRIAQTGWV 230 TTTQDWHRVAQTGWV 290 T
TIQDWHRIAQTGWV 206 TNTQDWHRVAQTGWV 232 TNSQDWHRIAQTGWA 291 T
TSQDWHRIAQTGWV 207 TNTQDWHRVSQTGWV 237 TNSQDWHRVAQTGWV 293 id
sequence having the formula [N1]-[N2]-[N3] (SEQ ID NO: 4681), wherein [N2] comprises the amino acid sequence of DWHR (SEQ ID NO: 4682) and wherein [N1] comprises amino acids X
1, X
2, X
3, and X
4, wherein X
4 is Q, K, E, S, P, R, N, H, or a conservative substitution thereof and/or [N3] comprises X
5, X
6, and X
7, wherein X
5 is I, V, T, M, S, N, L, F, or a conservative substitution thereof. In some embodiments, [N2] comprises the amino acid sequence of DWHR (SEQ ID NO: 4682), [N1] comprises X
1, X
2, X
3, and X
4, wherein X
4 is Q, K, E, S, P, R, N, or H, and/or [N3] comprises X
5, X
6, and X
7, wherein X
5 is I, V, T, M, S, N, L, or F. In some embodiments, X
4 of [N1] is Q. In some embodiments, X
4 of [N1] is K. In some embodiments, X
5 of [N3] is I. In some embodiments, X
1 of [N1] is T, S, R, A, I, C, N, K, L, or Q. In some embodiments, X
2 of [N1] is N, T, G, V, S, Y, K, I, H, D, or F. In some embodiments, X
3 of [N1] is T, N, K, D, I, S, P, A, Y, E, V, L, M, R, H, Q, or C. In some embodiments, [N1] is or comprises TNTQ (SEQ ID NO: 4688). In some embodiments, [N1] is or comprises TNTK (SEQ ID NO: 4689). In some embodiments, [N1]-[N2] is or comprises TNTQDWHR (SEQ ID NO: 4898). In some embodiments, [N1]-[N2] is or comprises TNTKDWHR (SEQ ID NO: 4899). In some embodiments, X
6 of [N3] is A, Y, P, N, S, T, G, E, V, W, F, or Q. In some embodiments, X
7 of [N3] is Q, G, N, K, H, R, E, L, P, or M. In some embodiments, [N3] is or comprises IAQ. In some embodiments, [N2]-[N3] is or comprises DWHRIAQ (SEQ ID NO: 5027). In some embodiments, [N1]-[N2]-[N3] is or comprises TNTQDWHRIAQ (SEQ ID NO: 343). In some embodiments, [N1]-[N2]-[N3] is or comprises TNTKDWHRIAQ (SEQ ID NO: 344). [094] In some embodiments, the amino acid sequence having the formula [N1]-[N2]-[N3] further comprises [N4], wherein [N4] comprises amino acids X
8, X
9, X
10, and X
11, wherein X
8 is T, S, N, P, A, or I; X
9 is G, N, D, R, V, A, S, or Q; X
10 is W, S, C, R, L, or G; and/or X
11 is V, A, S, I, C, G, D, F, L, or T. In some embodiments, [N4] is or comprises TGWV (SEQ ID NO: 5066). In some embodiments, [N1]- [N2]-[N3]-[N4] is or comprises any one of SEQ ID NOs: 201-245, 247-250, 253-255, 257-265, 268-274, 276-286, 288, 290-297, 299-303, 305-309, 311, 313-319, 323-328, 330-337, 339-342, 539-542, 544, 546, 547, 549-557, 559-589, 592, 593, 595, 596, 598, 599, 601-608, 610-614, 616-622, 625, 628, 630, 631, 633, 636, 638, 639-646, 649, 651-657, 667, 669, 670, 672, 673, 679-683, 685-690, 692, 693, 695, 697, 699-701, 703-705, 708-710, 712-717, 719-723, 728-731, 733-738, 740, and 742. In some embodiments, [N1]-[N2]-[N3]-[N4] is or comprises TNTQDWHRIAQTGWV (SEQ ID NO: 201). In some embodiments, [N1]-[N2]-[N3]-[N4] is or comprises TNTKDWHRIAQTGWV (SEQ ID NO: 202).
Attorney Docket No.14640.0105-00304 [095] In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence having the formula [N1]-[N2]-[N3] (SEQ ID NO: 4683), wherein [N2] comprises the amino acid sequence of DWHR (SEQ ID NO: 4682) and wherein [N1] comprises amino acids X
1, X
2, X
3, and X
4, wherein X
4 is Q, P, or a conservative substitution thereof; and/or [N3] comprises amino acids X
5, X
6, and X
7, wherein X
5 is I, V, or a conservative substitution thereof. In some embodiments, [N2] comprises the amino acid sequence of DWHR (SEQ ID NO: 4682); [N1] comprises amino acids X
1, X
2, X
3, and X
4, wherein X
4 is Q or P; and/or [N3] comprises amino acids X
5, X
6, and X
7, wherein X
5 is I or V. In some embodiments, X
4 of [N1] is Q. In some embodiments, X
5 of [N3] is I. In some embodiments, X
5 of [N3] is V. In some embodiments, X
1 of [N1] is T or S. In some embodiments, X
2 of [N1] is N, T, G, S, I, or V. In some embodiments, X
3 of [N1] is T, N, I, S, A, V, or L. In some embodiments, [N1] is or comprises TNTQ (SEQ ID NO: 4688). In some embodiments, [N1]-[N2] is or comprises TNTQDWHR (SEQ ID NO: 4898). In some embodiments, X
6 of [N3] is A, P, S, Y, or N. In some embodiments, X
7 of [N3] is Q, G, or N. In some embodiments, [N3] is or comprises IAQ. In some embodiments, [N2]-[N3] is or comprises DWHRIAQ (SEQ ID NO: 5027). In some embodiments, [N1]-[N2]-[N3] is or comprises TNTQDWHRIAQ (SEQ ID NO: 343). In some embodiments, the amino acid sequence having the formula [N1]-[N2]-[N3] further comprises [N4], wherein [N4] comprises amino acids X
8, X
9, X
10, and X
11, wherein X
10 is W. In some embodiments, X
8 of [N4] is T, S, or N. In some embodiments, X
9 of [N4] is G or N. In some embodiments, X11 of [N4] is V, A, I, or S. In some embodiments, [N4] is or comprises TGWV (SEQ ID NO: 5066). In some embodiments, [N1]-[N2]-[N3]-[N4] is or comprises any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336. In some embodiments, [N1]-[N2]-[N3]-[N4] is or comprises TNTQDWHRIAQTGWV (SEQ ID NO: 201). [096] In some embodiments, [N1]-[N2]-[N3] is present in loop VIII of the AAV capsid variant. In some embodiments, [N4] is present in loop VIII of the AAV capsid variant. In some embodiments, [N1]- [N2]-[N3]-[N4] is present in loop VIII of the AAV capsid variant. In some embodiments, loop VIII is present at amino acids comprising those corresponding to positions 581-593 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. In some embodiments, loop VIII is present at amino acids comprising those corresponding to positions 580-599 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. [097] In some embodiments, [N1] is present at amino acids corresponding to positions 582-585 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. In some embodiments, X
1 of [N1] is present at an amino acid corresponding to position 582 of the amino acid sequence of SEQ ID NO: 138 or 981, X
2 of [N1] is present at an amino acid corresponding to position 583 of the amino acid sequence of SEQ ID NO: 138 or 981, X
3 of [N1] is present at an amino acid corresponding to position 584 of the amino acid sequence of SEQ ID NO: 138 or 981, and X
4 of [N1] is present at an amino acid corresponding to position 585 of the amino acid sequence of SEQ ID NO: 138 or 981. In some
Attorney Docket No.14640.0105-00304 embodiments, [N1] replaces amino acids corresponding to positions 582-585 (e.g., T582, N583, H584, and Q585) of the amino acid sequence of SEQ ID NO: 138. [098] In some embodiments, [N2] is present at amino acids corresponding to positions 586-589 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. In some embodiments, [N2] replaces amino acids corresponding to positions 586-589 of the amino acid sequence of SEQ ID NO: 138 (e.g., S586, A587, Q588, and A589 may be replaced with D586, W587, H588, and R589). [099] In some embodiments, [N1]-[N2] is present at amino acids corresponding to positions 582- 589 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. In some embodiments, [N1]- [N2] replaces amino acids corresponding to positions 582-589 of the amino acid sequence of SEQ ID NO: 138 (e.g., T582, N583, H584, Q585, S586, A587, Q588, and A589 may be replaced with T582, N583, T584, Q585, D586, W587, H588, and R589). [0100] In some embodiments, [N3] is present at amino acids corresponding to positions 590-592 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. In some embodiments, X
5 of [N3] is present at an amino acid corresponding to position 590 of the amino acid sequence of SEQ ID NO: 138 or 981, X
6 of [N3] is present at an amino acid corresponding to position 591 of the amino acid sequence of SEQ ID NO: 138 or 981, and X
7 of [N3] is present at an amino acid corresponding to position 592 of the amino acid sequence of SEQ ID NO: 138 or 981. In some embodiments, [N3] replaces amino acids corresponding to positions 590-592 (e.g., I590, A591, and Q592) of the amino acid sequence of SEQ ID NO: 138. [0101] In some embodiments, [N2]-[N3] is present at amino acids corresponding to positions 586- 592 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. In some embodiments, [N2]- [N3] replaces amino acids corresponding to positions 586-592 of the amino acid sequence of SEQ ID NO: 138 (e.g., S586, A587, Q588, A589, Q590, A591, and Q592 may be replaced with D586, W587, H588, R589, I590, A591, and Q592). [0102] In some embodiments, [N1]-[N2]-[N3] is present at amino acids corresponding to positions 582-592 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. In some embodiments, [N1]-[N2]-[N3] replaces amino acids corresponding to positions 582-592 of the amino acid sequence of SEQ ID NO: 138 (e.g., T582, N583, H584, Q585, S586, A587, Q588, A589, Q590, A591, and Q592 may be replaced with T582, N583, T584, Q585, D586, W587, H588, R589, I590, A591, and Q592). [0103] In some embodiments, [N4] is present at amino acids corresponding to positions 593-596 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. In some embodiments, X
8 of [N4] is present at an amino acid corresponding to position 593 of the amino acid sequence of SEQ ID NO: 138 or 981, X
9 of [N4] is present at an amino acid corresponding to position 594 of the amino acid sequence of SEQ ID NO: 138 or 981, X
10 of [N4] is present at an amino acid corresponding to position 595 of the amino acid sequence of SEQ ID NO: 138 or 981, and X
11 of [N4] is present at an amino acid corresponding to position 596 of the amino acid sequence of SEQ ID NO: 138 or 981. In some
Attorney Docket No.14640.0105-00304 embodiments, [N4] replaces amino acids corresponding to positions 593-596 (e.g., T593, G594, W595, and V596) of the amino acid sequence of SEQ ID NO: 138. [0104] In some embodiments, [N2]-[N3]-[N4] is present at amino acids corresponding to positions 586-596 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. In some embodiments, [N2]-[N3]-[N4] replaces amino acids corresponding to positions 586-596 of the amino acid sequence of SEQ ID NO: 138 (e.g., S586, A587, Q588, A589, Q590, A591, Q592, T593, G594, W595, and V596 may be replaced with D586, W587, H588, R589, I590, A591, Q592, T593, G594, W595, and V596). [0105] In some embodiments, [N1]-[N2]-[N3]-[N4] is present at amino acids corresponding to positions 582-596 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. In some embodiments, [N1]-[N2]-[N3]-[N4] replaces amino acids corresponding to positions 582-596 of the amino acid sequence of SEQ ID NO: 138 (e.g., T582, N583, H584, Q585, S586, A587, Q588, A589, Q590, A591, Q592, T593, G594, W595, and V596 may be replaced with T582, N583, T584, Q585, D586, W587, H588, R589, I590, A591, Q592, T593, G594, W595, and V596). [0106] In some embodiments, [N2] is present immediately subsequent to [N1]. In some embodiments, [N3] is present immediately subsequent to [N2]. In some embodiments, [N4] is present immediately subsequent to [N3]. In some embodiments, loop VIII of the AAV capsid variant comprises an amino acid sequence comprising, from N-terminus to C-terminus, [N1]-[N2]-[N3]. In some embodiments, loop VIII of the AAV capsid variant comprises an amino acid sequence comprising, from N-terminus to C-terminus, [N1]-[N2]-[N3]-[N4]. [0107] In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, or at least 14 consecutive amino acids from any one of the sequences provided in Table 1, 2A, 2B, 9, 23, 24, or 25. In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, or at least 14 consecutive amino acids from any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336. [0108] In some embodiments, the at least 3 consecutive amino acids comprise TQD. In some embodiments, the at least 4 consecutive amino acids comprise TQDW (SEQ ID NO: 4684). In some embodiments, the at least 5 consecutive amino acids comprise TQDWH (SEQ ID NO: 4685). In some embodiments, the at least 6 consecutive amino acids comprise TQDWHR (SEQ ID NO: 4686). In some embodiments, the at least 7 consecutive amino acids comprise TQDWHRI (SEQ ID NO: 941). [0109] In some embodiments, the at least 3 consecutive amino acids comprise TNT. In some embodiments, the at least 4 consecutive amino acids comprise TNTQ (SEQ ID NO: 4688). In some embodiments, the at least 5 consecutive amino acids comprise TNTQD (SEQ ID NO: 5119). In some embodiments, the at least 6 consecutive amino acids comprise TNTQDW (SEQ ID NO: 5120). In some embodiments, the at least 7 consecutive amino acids comprise TNTQDWH (SEQ ID NO: 5121). In some
Attorney Docket No.14640.0105-00304 embodiments, the at least 8 consecutive amino acids comprise TNTQDWHR (SEQ ID NO: 4898). In some embodiments, the at least 9 consecutive amino acids comprise TNTQDWHRI (SEQ ID NO: 746). [0110] In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence comprising at least one, at least two, or at least three modifications, but no more than four modifications, relative to the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, 9, 23, 24, or 25. In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence comprising at least one, at least two, or at least three substitutions, but no more than four substitutions, relative to the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, 9, 23, 24, or 25. In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence comprising at least one, at least two, or at least three modifications, but no more than four modifications, relative to the amino acid sequence of any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336. In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence comprising at least one, at least two, or at least three substitutions, but no more than four substitutions, relative to the amino acid sequence of any one of SEQ ID NOs: 201, 205-209, 211- 214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336. [0111] In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence comprising at least one, at least two, or at least three modifications, but no more than four modifications, relative to the amino acid sequence of TQDWHRI (SEQ ID NO: 941). In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence comprising at least one, at least two, or at least three substitutions, but no more than four substitutions, relative to the amino acid sequence of TQDWHRI (SEQ ID NO: 941). [0112] In some embodiments, the AAV capsid variant comprises the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, 9, 23, 24, or 25. In some embodiments, the AAV capsid variant comprises the amino acid sequence of any one of SEQ ID NOs: 201, 205-209, 211-214, 216, 219, 220, 230, 232, 237, 238, 255, 262-265, 274, 283, 286, 290, 291, 293, 301, 306, 307, 308, 309, 314, and 336. [0113] In some embodiments, the amino acid sequence is present in loop VIII. In some embodiments, loop VIII comprises amino acids corresponding to positions 581-593 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, loop VIII comprises amino acids corresponding to positions 580-599 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, loop VIII is present (is located) at amino acids comprising those corresponding to positions 580-599 (e.g., the positions of amino acids VATNHQSAQAQAQTGWVQNQ (SEQ ID NO: 5122)) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the amino acid sequence replaces 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or all of amino acids corresponding to position(s) 582 (e.g., T582), 583 (e.g., N583), 584 (e.g., H584), 585 (e.g., Q585), 586 (e.g., S586), 587 (e.g., A587), 588 (e.g., Q588), 589 (e.g.,
Attorney Docket No.14640.0105-00304 A589), 590 (e.g., Q590), 591 (e.g., A591), 592 (e.g., Q592), 593 (e.g., T593), 594 (e.g., G594), 595 (e.g., W595), and/or 596 (e.g., V596) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the amino acid sequence is present at 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or all of amino acids corresponding to position(s) 582, 583, 584, 585, 586, 587, 588, 589, 590, 591, 592, 593, 594, 595, and/or 596 of the amino acid sequence of SEQ ID NO: 981. In some embodiments, the AAV capsid variant comprises one or more substitutions at amino acids corresponding to position(s) 582 (e.g., T582), 583 (e.g., N583), 584 (e.g., H584), 585 (e.g., Q585), 586 (e.g., S586), 587 (e.g., A587), 588 (e.g., Q588), 589 (e.g., A589), 590 (e.g., Q590), 591 (e.g., A591), 592 (e.g., Q592), 593 (e.g., T593), 594 (e.g., G594), 595 (e.g., W595), and/or 596 (e.g., V596) of the amino acid sequence of SEQ ID NO: 138. [0114] In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence encoded by a nucleotide sequence described herein, e.g., a nucleotide sequence of Table 2A. In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 942. In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven different substitutions, but not more than ten substitutions, relative to the nucleotide sequence of SEQ ID NO: 942. In some embodiments, the AAV capsid variant comprises (e.g., in loop VIII) an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 942, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. [0115] In some embodiments, the nucleotide sequence encoding an AAV capsid variant described herein, or a portion thereof that is modified relative to a wildtype AAV capsid (e.g., comprising one or more substitutions relative to a wildtype AAV capsid), comprises a nucleotide sequence described herein, e.g., as described in Table 2A. In some embodiments, the nucleotide sequence encoding an AAV capsid variant described herein, or a portion thereof that is modified relative to a wildtype AAV capsid (e.g., comprising one or more substitutions relative to a wildtype AAV capsid), is codon optimized. In some embodiments, the nucleotide sequence is an isolated nucleotide sequence. In some embodiments, the nucleotide sequence is a recombinant nucleotide sequence. [0116] In some embodiments, the nucleotide sequence encoding an AAV capsid variant described herein, or a portion thereof that is modified relative to a wildtype AAV capsid (e.g., comprising one or more substitutions relative to a wildtype AAV capsid), comprises the nucleotide sequence of SEQ ID NO: 942, or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 942. In some embodiments, the nucleotide sequence comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least
Attorney Docket No.14640.0105-00304 six, or at least seven substitutions, but no more than ten substitutions, relative to the nucleotide sequence of SEQ ID NO: 942. In some embodiments, the nucleotide sequence comprises the nucleotide sequence of SEQ ID NO: 942, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. [0117] In some embodiments, the AAV capsid variant further comprises an amino acid other than A at a position corresponding to position 581 of the amino acid sequence of SEQ ID NO: 138 or 981. In some embodiments, the AAV capsid variant further comprises the amino acid T at a position corresponding to position 581 of the amino acid sequence of SEQ ID NO: 138 or 981. In some embodiments, the AAV capsid variant further comprises the amino acid V at a position corresponding to position 581 of the amino acid sequence of SEQ ID NO: 138 or 981. In some embodiments, the AAV capsid variant comprises the substitution A581T or A581V, wherein the substitution is at position that corresponds to position 581 of the amino acid sequence of SEQ ID NO: 138 or 981. [0118] In some embodiments, an AAV capsid variant described herein comprises one, two, three, four, five, or all of an amino acid other than H at a position corresponding to position 584 (e.g., comprises T at an amino acid corresponding to position 584) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than S at a position corresponding to position 586 (e.g., comprises D at an amino acid corresponding to position 586) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A at a position corresponding to position 587 (e.g., comprises W at an amino acid corresponding to position 587) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than Q at a position corresponding to position 588 (e.g., comprises H at an amino acid corresponding to position 588) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A at a position corresponding to position 589 (e.g., comprises R at an amino acid corresponding to position 589) of the amino acid sequence of SEQ ID NO: 138; and/or an amino acid other than Q at a position corresponding to position 590 (e.g., comprises I at an amino acid corresponding to position 590) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises an amino acid other than H at a position corresponding to position 584 (e.g., comprises T at an amino acid corresponding to position 584) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than S at a position corresponding to position 586 (e.g., comprises D at an amino acid corresponding to position 586) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A at a position corresponding to position 587 (e.g., comprises W at an amino acid corresponding to position 587) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than Q at a position corresponding to position 588 (e.g., comprises H at an amino acid corresponding to position 588) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A at a position corresponding to position 589 (e.g., comprises R at an amino acid corresponding to position 589) of the amino acid sequence of SEQ ID NO: 138; and an amino acid other than Q at a position corresponding to position 590 (e.g., comprises I at an amino acid corresponding to position 590) of the amino acid sequence of SEQ ID NO: 138. In some
Attorney Docket No.14640.0105-00304 embodiments, an AAV capsid variant described herein comprises the amino acid T at a position corresponding to position 584 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid D at a position corresponding to position 586 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid W at a position corresponding to position 587 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid H at a position corresponding to position 588 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid R at a position corresponding to position 589 of the amino acid sequence of SEQ ID NO: 138 or 981, and amino acid I at a position corresponding to position 590 of the amino acid sequence of SEQ ID NO: 138 or 981. [0119] In some embodiments, an AAV capsid variant described herein comprises one, two, three, four, five, or all of the amino acid T at a position corresponding to position 584 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid D at a position corresponding to position 586 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid W at a position corresponding to position 587 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid H at a position corresponding to position 588 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid R at a position corresponding to position 589 of the amino acid sequence of SEQ ID NO: 138 or 981, and/or amino acid I at a position corresponding to position 590 of the amino acid sequence of SEQ ID NO: 138 or 981. In some embodiments, the AAV capsid variant comprises the amino acid T at a position corresponding to position 584 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid D at a position corresponding to position 586 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid W at a position corresponding to position 587 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid H at a position corresponding to position 588 of the amino acid sequence of SEQ ID NO: 138 or 981, amino acid R at a position corresponding to position 589 of the amino acid sequence of SEQ ID NO: 138 or 981, and amino acid I at a position corresponding to position 590 of the amino acid sequence of SEQ ID NO: 138 or 981. [0120] In some embodiments, an AAV capsid variant described herein comprises one, two, three, four, five, or all of the substitutions H584T, S586D, A587W, Q588H, A589R, and/or Q590I wherein the substitution(s) is/are at position(s) that correspond to position(s) 584, 586, 587, 588, 589, and/or 590 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises the substitutions H584T, S586D, A587W, Q588H, A589R, and Q590I wherein the substitution(s) is/are at position(s) that correspond to position(s) 584, 586, 587, 588, 589, and/or 590 of the amino acid sequence of SEQ ID NO: 138 or 981. [0121] In some embodiments, an AAV capsid variant described herein comprises the amino acid Q at an amino acid corresponding to position 585 of the amino acid sequence of SEQ ID NO: 138 or 981. [0122] In some embodiments, an AAV capsid variant described herein comprises an amino acid other than Q at an amino acid corresponding to position 585 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, an AAV capsid variant described herein comprises the amino acid K at an amino acid corresponding to position 585 of the amino acid sequence of SEQ ID NO: 138.
Attorney Docket No.14640.0105-00304 [0123] In some embodiments, an AAV capsid variant described herein comprises an amino acid other than Q at an amino acid corresponding to position 590 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises the amino acid I at an amino acid corresponding to position 590 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises the amino acid V at an amino acid corresponding to position 590 of the amino acid sequence of SEQ ID NO: 138. [0124] In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of TQDWHRI (SEQ ID NO: 941), wherein TQDWHRI (SEQ ID NO: 941) is present in the AAV capsid variant at amino acids corresponding to positions 584-590 of the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 981. In some embodiments, TQDWHRI (SEQ ID NO: 941) is present at amino acids corresponding to positions 584-590 of the amino acid sequence of SEQ ID NO: 138 (e.g., H584, S586, A587, Q588, A589, and Q590 may be replaced with T584, D586, W587, H588, R589, and I590). [0125] In some embodiments, an AAV capsid variant described herein comprises the amino acid W at an amino acid corresponding to position 595 of the amino acid sequence of SEQ ID NO: 138 or 981. [0126] In some embodiments, the AAV capsid variant further comprises a substitution corresponding to amino acid K449, (e.g., K449R) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an amino acid other than K at a position corresponding to position 449 (e.g., comprises an R corresponding to position 449) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises an R at amino acid corresponding to position 449 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a modification in loop I, II, IV, and/or VI. [0127] In some embodiments, the AAV capsid variant further comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 138. In some embodiments, the amino acid sequence does not comprise more than 10 modifications. In some embodiments, the AAV capsid variant further comprises an amino acid sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 amino acids that differ from the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises the amino acid sequence of SEQ ID NO: 138, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. [0128] In some embodiments, the AAV capsid variant comprises (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 981 (i.e., comprising amino acids 1-736 of SEQ ID NO: 981); (b) a VP2 protein comprising amino acids 138-736 of SEQ ID NO: 981; (c) a VP3 protein comprising amino acids 203-736 of SEQ ID NO: 981; or (d) an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%,
Attorney Docket No.14640.0105-00304 or at least 99%) sequence identity to any one of the amino acid sequences in (a)-(c), an amino acid sequence comprising at least one, at least two, or at least three substitutions, but not more than 30, not more than 20, or not more than 10 substitutions relative to any one of the amino acid sequences in (a)-(c), or an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 such modifications relative to any one of the amino acid sequences in (a)-(c). [0129] In some embodiments, the AAV capsid variant comprises (a) a VP1 protein comprising an amino acid sequence that is at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to the amino acid sequence of SEQ ID NO: 981; (b) a VP2 protein comprising an amino acid sequence that is at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to amino acids 138-736 of SEQ ID NO: 981; and/or (c) a VP3 protein comprising an amino acid sequence that is at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to amino acids 203-736 of SEQ ID NO: 981, wherein the AAV capsid variant comprises the amino acid sequence of TQDWHRI (SEQ ID NO: 941) or a sequence comprising one, two, or three but not more than three substitutions relative to the amino acid sequence of TQDWHRI (SEQ ID NO: 941). In some embodiments, the AAV capsid variant comprises the amino acid sequence comprising one, two, or three but not more than three substitutions relative to the amino acid sequence of TQDWHRI (SEQ ID NO: 941) in loop VIII. In some embodiments, the AAV capsid variant comprises the amino acid sequence of TQDWHRI (SEQ ID NO: 941) in loop VIII. [0130] In some embodiments, the AAV capsid variant comprises an amino acid sequence as described herein, e.g., an amino acid sequence of TTJ-001, e.g., as described in Tables 3 and 4. In some embodiments, the AAV capsid variant is or comprises TTJ-001. In some embodiments, the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 941 in a loop VIII region. [0131] In some embodiments, the AAV capsid variant comprises a VP1, VP2, and/or VP3 protein comprising an amino acid sequence described herein, e.g., an amino acid sequence of TTJ-001, e.g., as described in Tables 3 and 4. [0132] In some embodiments, the AAV capsid variant comprises an amino acid sequence encoded by a nucleotide sequence as described herein, e.g., a nucleotide sequence encoding TTJ-001, e.g., as described in Tables 3 and 5. [0133] In some embodiments, the polynucleotide or nucleic acid encoding the AAV capsid variant comprises a nucleotide sequence described herein, e.g., a nucleotide sequence encoding TTJ-001, e.g., as described in Tables 3 and 5. Table 3. Exemplary full length capsid sequences VP1 DN VP1 (amino Peptide Name A acid) SEQ ID (amino acid) Peptide DNA SEQ

Attorney Docket No.14640.0105-00304 Table 4. Exemplary full length capsid amino acid sequences Name and SEQ Amino Acid Sequence Annotation ID NO: P
G GG AK SS TP AN RS LS WP MI QG IT IG
Name and SEQ Nucleotide Sequence Annotation ID NO: T C C A A C T C A C G C G A C A T T T A G T C A G C T C A A A A A T G C A
Attorney Docket No.14640.0105-00304 CAGCAAGCGGTGGAACCCGGAGATCCAGTACACTTCCAACTATTACAAGTCTAATA ATGTTGAATTTGCTGTTAATACTGAAGGTGTATATAGTGAACCCCGCCCCATTGGC ACCAGATACCTGACTCGTAATCTGTAA

nucleotide sequence of SEQ ID NO: 983, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. [0135] In some embodiments, the nucleotide sequence encoding an AAV capsid variant described herein, comprises a nucleotide sequence comprising at least one, at least two, or at least three modifications but not more than 30, not more than 20, or not more than 10 modifications, relative to the nucleotide sequence of SEQ ID NO: 983. In some embodiments, the nucleotide sequence encoding an AAV capsid variant described herein, comprises a nucleotide sequence comprising at least one, at least two, or at least three substitutions, but not more than 30, not more than 20, or not more than 10 substitutions relative to the amino acid sequence of SEQ ID NO: 983. In some embodiments, the nucleic acid sequence encoding an AAV capsid variant described herein is codon optimized. [0136] In some embodiments, the AAV capsid variant comprises one or more substitutions in loop VIII and comprises the amino acid sequence of SEQ ID NO: 981, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 981. In some embodiments, an AAV capsid variant described herein comprises an amino acid sequence comprising at least one, at least two, or at least three substitutions, but not more than 30, not more than 20, or not more than 10 substitutions, relative to the amino acid sequence of SEQ ID NO: 981. [0137] In some embodiments, the AAV capsid variant comprises one or more substitutions in loop VIII and comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 983, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three substitutions, but not more than 30, not more than 20, or not more than 10 substitutions, relative to the amino acid sequence of SEQ ID NO: 983. In some embodiments, an AAV capsid variant described herein comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the nucleotide sequence of SEQ ID NO: 983. [0138] In some embodiments, an AAV capsid variant described herein comprises a VP1, VP2, VP3 protein, wherein at least one of the VP1, VP2, and VP3 comprise one or more substitutions in loop VIII.
Attorney Docket No.14640.0105-00304 In some embodiments, the AAV capsid variant comprises amino acids 138-736, e.g., a VP2, of the amino acid sequence of SEQ ID NO: 981, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid variant comprises amino acids 203-736, e.g., a VP3, of the amino acid sequence of SEQ ID NO: 981, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid variant comprises amino acids 1-736, e.g., a VP1, of the amino acid sequence of SEQ ID NO: 981, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. [0139] In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of amino acids 203-736 of SEQ ID NO: 981, or an amino acid sequence at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical thereto. In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of amino acids 138-736 of SEQ ID NO: 981, or an amino acid sequence at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical thereto. In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 981, or an amino acid sequence at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical thereto. [0140] In some embodiments, an AAV capsid variant described herein has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. [0141] In some embodiments, an AAV capsid variant described herein transduces a brain region, e.g., a midbrain region (e.g., the hippocampus, or thalamus) or the brain stem. In some embodiments, the level of transduction is at least 39, at least 50, at least 100, at least 120, at least 132, at least 146, at least 150, at least 161, at least 174, at least 175, at least 200, at least 225, at least 250, at least 275, at least 283, at least 300, at least 350, at least 400, at least 450, at least 500, at least 525, at least 528, or at least 550- fold greater as compared to an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. [0142] In some embodiments, an AAV capsid variant described herein is enriched at least 10, at least 14, at least 20, at least 24, at least 50, at least 100, at least 150, at least 200, at least 250, at least 300, at least 350, at least 400, at least 425, at least 450, or at least 460-fold in the brain compared to enrichment in the brain of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. In some embodiments, an AAV capsid variant described herein is enriched at least 200, at least 250, at least 300, at least 350, at least 400, at least 425, at least 450, or at least 460-fold in the brain compared to enrichment in the brain of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. [0143] In some embodiments, an AAV capsid variant described herein is enriched in the brain of at least two to three species, e.g., a non-human primate and rodent (e.g., mouse) species, compared to
Attorney Docket No.14640.0105-00304 enrichment in the brain of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. In some embodiments, an AAV capsid variant described herein is enriched at least 2, at least 3, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 65, at least 70, at least 75, at least 80, at least 85, at least 90, at least 95, at least 100, at least 105, at least 115, at least 120, at least 125, at least 130, at least 135, at least 140, at least 145, at least 150, at least 155, at least 160, at least 165, at least 170, at least 175, at least 180, at least 190, at least 200, at least 205, or at least 210-fold in the brain of at least two to three species, e.g., a non- human primate and rodent (e.g., mouse) species, compared to enrichment in the brain of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. In some embodiments, the at least two to three species are Macaca fascicularis, Chlorocebus sabaeus, Callithrix jacchus, and/or mouse (e.g., outbred mice). [0144] In some embodiments, an AAV capsid variant described herein is enriched at least 2, at least 3, at least 4, at least 5, at least 10, at least 15, at least 17, at least 20, at least 50, at least 75, at least 100, at least 103, at least 107, at least 125, at least 150, at least 200, at least 250, at least 300, at least 350, at least 400, at least 450, at least 500, at least 750, at least 1000, or at least 1200-fold in the brain compared to enrichment in the brain of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 981. [0145] In some embodiments, an AAV capsid variant described herein delivers an increased level of viral genomes to a brain region. In some embodiments, the level of viral genomes is increased by at least 2, at least 5, at least 7, at least 10, at least 15, at least 19, at least 20, at least 22, or at least 25-fold, as compared to the level of viral genomes delivered by an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. In some embodiments, the brain region comprises a sensory cortex, motor cortex, putamen, thalamus, caudate, hippocampus, and/or cerebellum. [0146] In some embodiments, an AAV capsid variant delivers an increased level of a payload to a brain region. In some embodiments, the level of the payload is increased by at least 39, at least 50, at least 100, at least 120, at least 132, at least 146, at least 150, at least 161, at least 174, at least 175, at least 200, at least 225, at least 250, at least 275, at least 283, at least 300, at least 350, at least 400, at least 450, at least 500, at least 525, at least 528, or at least 550-fold, as compared to the level of a payload delivered by an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. In some embodiments, the brain region comprises a sensory cortex, motor cortex, putamen, thalamus, caudate, hippocampus, and/or cerebellum. [0147] In some embodiments, an AAV capsid variant described herein is enriched at least 5, at least 10, at least 50, at least 100, at least 115, at least 120, at least 150, at least 175, at least 200, at least 207, at least 225, at least 250, or at least 275-fold in the spinal cord compared to enrichment in the spinal cord of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. [0148] In some embodiments, an AAV capsid variant of the present disclosure has decreased tropism for the liver. In some embodiments, an AAV capsid variant comprises a modification that results in reduced tropism (e.g., de-targeting) and/or activity in the liver. In some embodiments, the reduced
Attorney Docket No.14640.0105-00304 tropism in the liver is compared to an otherwise similar capsid that does not comprise the modification, e.g., a wild-type capsid polypeptide. In some embodiments, an AAV capsid variant comprises a modification that results in one or more of the following properties: (1) reduced tropism in the liver; (2) de-targeted expression in the liver; (3) reduced activity in the liver; and/or (4) reduced binding to galactose. In some embodiments, the reduction in any one or all of properties (1)-(3) is compared to an otherwise similar AAV capsid variant that does not comprise the modification. Exemplary modifications are provided in WO 2018/119330; Pulicherla et al. (2011) Mol. Ther.19(6): 1070-1078; Adachi et al. (2014) Nature Communications 5(3075), DOI: 10.1038/ncomms4075; and Bell et al. (2012) J. Virol. 86(13): 7326-33; the contents of which are hereby incorporated by reference in their entirety. [0149] In some embodiments, the AAV capsid variant comprises a modification at an amino acid corresponding to N470 (e.g., comprises N470A at an amino acid corresponding to N470) of the amino acid sequence of SEQ ID NO: 138; an amino acid corresponding to D271 (e.g., comprises D271A at an amino acid corresponding to D271) of the amino acid sequence of SEQ ID NO: 138; an amino acid corresponding to N272 (e.g., comprises N272A at an amino acid corresponding to N272) of the amino acid sequence of SEQ ID NO: 138; an amino acid corresponding to Y446 (e.g., comprises Y446A at an amino acid corresponding to Y446) of the amino acid sequence of SEQ ID NO: 138; an amino acid corresponding to N498 (e.g., comprises N498Y or N498I at an amino acid corresponding to N498) of the amino acid sequence of SEQ ID NO: 138; an amino acid corresponding to W503 (e.g., comprises W503R or W503A at an amino acid corresponding to W503) of the amino acid sequence of SEQ ID NO: 138; an amino acid corresponding to L620 (e.g., comprises L620F at an amino acid corresponding to L620) of the amino acid sequence of SEQ ID NO: 138; or a combination thereof. In some embodiments, the AAV capsid variant comprises one, two, three, four, five, or all of an amino acid other than N at a position corresponding to position 470 (e.g., comprises A at an amino acid corresponding to position 470) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than D at a position corresponding to position 271 (e.g., comprises A at an amino acid corresponding to position 271) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than N at a position corresponding to position 272 (e.g., comprises A at an amino acid corresponding to position 272) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than Y at a position corresponding to position 446 (e.g., comprises A at an amino acid corresponding to position 446) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than N at a position corresponding to position 498 (e.g., comprises Y or I at an amino acid corresponding to position 498) of the amino acid sequence of SEQ ID NO: 138; an amino acid other than W at a position corresponding to position 503 (e.g., comprises R or A at an amino acid corresponding to position 503) of the amino acid sequence of SEQ ID NO: 138; and an amino acid other than L at a position corresponding to position 620 (e.g., comprises F at an amino acid corresponding to position 620) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises a modification at positions corresponding to amino acid N470 (e.g., comprises N470A at an amino acid corresponding to N470) of the amino acid sequence of SEQ ID NO:
Attorney Docket No.14640.0105-00304 138; corresponding to amino acid D271 (e.g., comprises D271A at an amino acid corresponding to D271) of the amino acid sequence of SEQ ID NO: 138; corresponding to amino acid N272 (e.g., comprises N272A at an amino acid corresponding to N272) of the amino acid sequence of SEQ ID NO: 138; corresponding to amino acid Y446 (e.g., comprises Y446A at an amino acid corresponding to Y446) of the amino acid sequence of SEQ ID NO: 138; and corresponding to amino acid W503 (e.g., comprises W503R or W503A at an amino acid corresponding to W503) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises a modification at a position corresponding to amino acid N498 (e.g., comprises N498Y at an amino acid corresponding to N498) of the amino acid sequence of SEQ ID NO: 138 and corresponding to amino acid L620 (e.g., comprises L620F at an amino acid corresponding to L620) of the amino acid sequence of SEQ ID NO: 138. [0150] In some embodiments, the AAV capsid variant comprises a modification as described in Adachi et al. (2014) Nature Communications 5(3075), DOI: 10.1038/ncomms4075, the contents of which are hereby incorporated by reference in its entirety. Exemplary modifications that alter or do not alter tissue transduction in at least the brain, liver, heart, lung, and/or kidney can be found in Supplementary Data 2 showing the AAV Barcode-Seq data obtained with AAV9-AA-VBCLib of Adachi et al. (supra), the contents of which are hereby incorporated by reference in their entirety. [0151] In some embodiments, the AAV capsid variant is an isolated capsid variant. In some embodiments, the AAV capsid variant is a recombinant capsid variant. In some embodiments, a polynucleotide encoding an AAV capsid polypeptide, e.g., an AAV capsid variant is an isolated and/or a recombinant AAV capsid polypeptide. [0152] Also provided herein are polynucleotide sequences encoding any of the AAV capsid variants described above and AAV particles, vectors, and cells comprising the same. AAV serotypes and capsids [0153] In some embodiments, the AAV particle may comprise a capsid protein or variant of any natural or recombinant AAV serotype. AAV serotypes may differ in characteristics such as, but not limited to, packaging, tropism, transduction and immunogenic profiles. Without being bound by theory, it is believed in some embodiments, that the AAV capsid protein, e.g., an AAV capsid variant can modulate AAV particle tropism in a particular tissue. [0154] In some embodiments, the AAV capsid variant allows for blood brain barrier penetration following intravenous administration. In some embodiments, the AAV capsid variant allows for blood brain barrier penetration following intravenous administration, focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration. In some embodiments, the AAV capsid variant allows for increased distribution to a brain region. In some embodiments, the brain region comprises the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior
Attorney Docket No.14640.0105-00304 olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, and/or vestibular nucleus. In some embodiments, the AAV capsid variant allows for preferential transduction in a brain region relative to the transduction in the dorsal root ganglia (DRG). In some embodiments, the AAV capsid variant allows for preferential transduction in a brain region relative to the transduction in the liver. In some embodiments, the AAV capsid variant allows for transduction in neuronal cells. In some embodiments, the AAV capsid variant allows for transduction in a non-neuronal cell, e.g., a glial cell (e.g., an astrocyte, an oligodendrocyte, or a combination thereof). In some embodiments, the AAV capsid variant allows for transduction in both neuronal cells and non-neuronal cell, e.g., a glial cell (e.g., an astrocyte, an oligodendrocyte, or a combination thereof). [0155] In some embodiments, an AAV capsid variant allows for increased distribution to a spinal cord region. In some embodiments, the spinal region comprises a cervical spinal cord region, thoracic spinal cord region, and/or lumbar spinal cord region. [0156] In some embodiments, the AAV capsid variant allows for increased distribution to a heart region. [0157] In some embodiments, the AAV capsid variant is suitable for intramuscular administration and/or transduction of muscle fibers. In some embodiments, the AAV capsid variant allows for increased distribution to a muscle region. In some embodiments, the muscle region comprises a heart muscle, quadriceps muscle, a diaphragm muscle region, or a combination thereof. In some embodiments, the muscle region comprises a heart muscle region, e.g., a heart atrium muscle region or a heart ventricle muscle region. [0158] In some embodiments, the AAV capsid variant is suitable for increased distribution to a kidney. In some embodiments, the AAV capsid variant is suitable for increased distribution to a pancreas. In some embodiments, the AAV capsid variant is suitable for increased distribution to a retina. [0159] In some embodiments, the initiation codon for translation of the AAV VP1 capsid protein, e.g., a capsid variant, described herein may be CTG, TTG, or GTG as described in US Patent No. US8163543, the contents of which are herein incorporated by reference in its entirety. [0160] The present disclosure refers to structural capsid proteins (including VP1, VP2 and VP3) which are encoded by capsid (Cap) genes. These capsid proteins form an outer protein structural shell (e.g., capsid) of a viral vector such as AAV. VP capsid proteins synthesized from Cap polynucleotides generally include a methionine as the first amino acid in the peptide sequence (Met1), which is associated with the start codon (AUG or ATG) in the corresponding Cap nucleotide sequence. However, it is common for a first-methionine (Met1) residue or generally any first amino acid (AA1) to be cleaved off after or during polypeptide synthesis by protein processing enzymes such as Met-aminopeptidases. This “Met/AA-clipping” process often correlates with a corresponding acetylation of the second amino acid in the polypeptide sequence (e.g., alanine, valine, serine, threonine, etc.). Met-clipping commonly occurs with VP1 and VP3 capsid proteins but can also occur with VP2 capsid proteins.
Attorney Docket No.14640.0105-00304 [0161] Where the Met/AA-clipping is incomplete, a mixture of one or more (one, two or three) VP capsid proteins comprising the viral capsid may be produced, some of which may include a Met1/AA1 amino acid (Met+/AA+) and some of which may lack a Met1/AA1 amino acid as a result of Met/AA- clipping (Met-/AA-). For further discussion regarding Met/AA-clipping in capsid proteins, see Jin, et al. Direct Liquid Chromatography/Mass Spectrometry Analysis for Complete Characterization of Recombinant Adeno-Associated Virus Capsid Proteins. Hum Gene Ther Methods.2017 Oct.28(5):255- 267; Hwang, et al. N-Terminal Acetylation of Cellular Proteins Creates Specific Degradation Signals. Science.2010 February 19.327(5968): 973–977; the contents of which are each incorporated herein by reference in its entirety. [0162] According to the present disclosure, references to capsid proteins, e.g., AAV capsid variants, is not limited to either clipped (Met-/AA-) or unclipped (Met+/AA+) and may, in context, refer to independent capsid proteins, viral capsids comprised of a mixture of capsid proteins, and/or polynucleotide sequences (or fragments thereof) which encode, describe, produce or result in capsid proteins of the present disclosure. A direct reference to a capsid protein or capsid polypeptide (such as VP1, VP2 or VP2) may also comprise VP capsid proteins which include a Met1/AA1 amino acid (Met+/AA+) as well as corresponding VP capsid proteins which lack the Met1/AA1 amino acid as a result of Met/AA-clipping (Met-/AA-). [0163] Further according to the present disclosure, a reference to a specific SEQ ID NO (whether a protein or nucleic acid) which comprises or encodes, respectively, one or more capsid proteins which include a Met1/AA1 amino acid (Met+/AA+) should be understood to teach the VP capsid proteins which lack the Met1/AA1 amino acid as upon review of the sequence, it is readily apparent any sequence which merely lacks the first listed amino acid (whether or not Met1/AA1). [0164] As a non-limiting example, reference to a VP1 polypeptide sequence which is 736 amino acids in length and which includes a “Met1” amino acid (Met+) encoded by the AUG/ATG start codon may also be understood to teach a VP1 polypeptide sequence which is 735 amino acids in length and which does not include the “Met1” amino acid (Met-) of the 736 amino acid Met+ sequence. As a second non-limiting example, reference to a VP1 polypeptide sequence which is 736 amino acids in length and which includes an “AA1” amino acid (AA1+) encoded by any NNN initiator codon may also be understood to teach a VP1 polypeptide sequence which is 735 amino acids in length and which does not include the “AA1” amino acid (AA1-) of the 736 amino acid AA1+ sequence. [0165] References to viral capsids formed from VP capsid proteins (such as reference to specific AAV capsid serotypes), can incorporate VP capsid proteins which include a Met1/AA1 amino acid (Met+/AA1+), corresponding VP capsid proteins which lack the Met1/AA1 amino acid as a result of Met/AA1-clipping (Met-/AA1-), and combinations thereof (Met+/AA1+ and Met-/AA1-). [0166] As a non-limiting example, an AAV capsid serotype can include VP1 (Met+/AA1+), VP1 (Met-/AA1-), or a combination of VP1 (Met+/AA1+) and VP1 (Met-/AA1-). An AAV capsid serotype can also include VP3 (Met+/AA1+), VP3 (Met-/AA1-), or a combination of VP3 (Met+/AA1+) and VP3
Attorney Docket No.14640.0105-00304 (Met-/AA1-); and can also include similar optional combinations of VP2 (Met+/AA1) and VP2 (Met- /AA1-). Additional AAV Capsid Sequences [0167] In some embodiments, the AAV capsid variant comprises at amino acids corresponding to positions 582, 583, 584, 585, 586, 587, 588, 589, and/or 590 of the amino acid sequence of SEQ ID NO: 138, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids of any one of the amino acid sequences provided in Table 1, 2A, 2B, 9, 23, 24, or 25. [0168] In some embodiments, the AAV capsid variant comprises, immediately subsequent to an amino acid corresponding to position 582, 583, 584, 585, 586, 587, 588, 589, and/or 590 of the amino acid sequence of SEQ ID NO: 138 (e.g., corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987 (the contents of which are hereby incorporated by reference in their entirety))), at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids of any one of the amino acid sequences provided in Table 1, 2A, 2B, 9, 14, 15, or 16. In some embodiments, the at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids of any one of the amino acid sequences provided in Table 1, 2A, 2B, 9, 23, 24, or 25 replaces at least one, at least two, at least three, at least four, at least five, at least six, at least seven, at least eight, or all of amino acid(s) corresponding to position(s) 582, 583, 584, 585, 586, 587, 588, 589, and/or 590 (e.g., T582, N583, H584, Q585, S586, A587, Q588, A589, and/or Q590) of the amino acid sequence of SEQ ID NO: 138 (e.g., corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987, which is hereby incorporated by reference in its entirety)). In some embodiments, the AAV capsid variant comprises an amino acid other than the wild-type amino acid, at one, two, three, four, five, six, seven, eight, or all of amino acid(s) corresponding to position(s) 582, 583, 584, 585, 586, 587, 588, 589, and/or 590 of the amino acid sequence of SEQ ID NO: 138 (e.g., corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987, which is hereby incorporated by reference in its entirety). In some embodiments, the AAV capsid variant comprises a modification replacing one, two, three, four, five, six, seven, eight, or all amino acid(s) corresponding to position(s) 582, 583, 584, 585, 586, 587, 588, 589, and/or 590 (e.g., T582, N583, H584, Q585, S586, A587, Q588, A589, and/or Q590) of the amino acid sequence of SEQ ID NO: 138 (e.g., corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8,
Attorney Docket No.14640.0105-00304 AAVrh10, AAVrh32.33, AAVrh74, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987, which is hereby incorporated by reference in its entirety). [0169] In some embodiments, the AAV capsid variant may comprise a variant of a VOY101 capsid polypeptide, an AAVPHP.B (PHP.B) capsid polypeptide, a AAVPHP.N (PHP.N) capsid polypeptide, an AAV1 capsid polypeptide, an AAV2 capsid polypeptide, an AAV5 capsid polypeptide, an AAV9 capsid polypeptide, an AAV9 K449R capsid polypeptide, or an AAVrh10 capsid polypeptide. In some embodiments, the AAV capsid polypeptide, e.g., AAV capsid variant, comprises an amino acid sequence of any one of the AAV capsid polypeptides in Table 6, or an amino acid sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments, the nucleotide sequence encoding the AAV capsid polypeptide comprises any one of the nucleotide sequences in Table 6, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. Table 6. AAV Sequences Serotype SEQ Sequence ID D Q E I R H V P S T P F D Q E I R H V P S G T V D Q E I R H V P

Attorney Docket No.14640.0105-00304 GPSYRQQRVSTTVTQNNNSEFAWPGASSWALNGRNSLMNPGPAMASHKEGEDRFFPLSGS LIFGKQGTGRDNVDADKVMITNEEEIKTTNPVATESYGQVATNHQSAQAQAQTGWVQNQG ILPGMVWQDRDVYLQGPIWAKIPHTDGNFHPSPLMGGFGMKHPPPQILIKNTPVPADPPT V C C C C C G T C G T A C C C A T C C G C A C G T A T T A G A C G G C G A

AAV Viral Genome [0170] In some embodiments, the AAV particle of the present disclosure serves as an expression vector comprising a viral genome that encodes an STXBP1 protein (e.g., a human STXBP1 protein). [0171] In some embodiments, an AAV particle, e.g., an AAV particle for the vectorized delivery of STXBP1 described herein, comprises a viral genome, e.g., an AAV viral genome, also referred to as an AAV genome, a vector genome, or an AAV vector genome. In some embodiments, the viral genome, e.g., the AAV viral genome, further comprises an inverted terminal repeat (ITR) region, an enhancer, a promoter, an intron region, an exon region, a nucleic acid encoding a transgene encoding an STXBP1 protein, a polyA region, or a combination thereof. In some embodiments, the viral genome, e.g., the AAV viral genome, further comprises a nucleotide sequence encoding at least one miRNA binding site.
Attorney Docket No.14640.0105-00304 Viral Genome Component: Inverted Terminal Repeats (ITRs) [0172] In some embodiments, the viral genome may comprise at least one inverted terminal repeat (ITR) region. The AAV particles of the present disclosure comprise a viral genome with at least one ITR region and a region comprising an STXBP1-encoding sequence. In some embodiments, the viral genome has two ITRs. These two ITRs flank the region comprising the STXBP1-encoding sequence at the 5’ and 3’ ends. In some embodiments, the ITR functions as an origin of replication comprising a recognition site for replication. In some embodiments, the ITR comprises a sequence region which can be complementary and symmetrically arranged. In some embodiments, the ITR incorporated into a viral genome described herein may be comprised of a naturally occurring polynucleotide sequence or a recombinantly derived polynucleotide sequence. [0173] In some embodiments, the ITR is of the same serotype as the capsid, selected from any one of the serotypes described herein, or a derivative thereof. In some embodiments, the ITR is of a different serotype than the capsid. In some embodiments, the AAV particle has more than one ITR. In some embodiments, the AAV particle comprises a viral genome comprising two ITRs. In some embodiments, the ITRs are of the same serotype as one another. In some embodiments, the ITRs are of different serotypes. Non-limiting examples include zero, one, or both of the ITRs having the same serotype as the capsid. Viral Genome Component: Promoters and Enhancers [0174] In some embodiments, the viral genome comprises at least one element to enhance the transgene target specificity and expression. See, e.g., Powell et al. Viral Expression Cassette Elements to Enhance Transgene Target Specificity and Expression in Gene Therapy, 2015; the contents of which are herein incorporated by reference in their entirety. Non-limiting examples of elements to enhance the transgene target specificity and expression include promoters, endogenous miRNAs, post-transcriptional regulatory elements (PREs), polyadenylation (PolyA) region, upstream enhancers (USEs), CMV enhancers, and introns. [0175] In some embodiments, expression of the polypeptides in a target cell may be driven by a specific promoter, including but not limited to, a promoter that is species specific, inducible, tissue- specific, or cell cycle-specific (Parr et al., Nat. Med.3:1145-9 (1997); the contents of which are herein incorporated by reference in their entirety). [0176] In some embodiments, the viral genome comprises a promoter that is sufficient for expression, e.g., in a target cell, of an STXBP1 protein (e.g., a human STXBP1 protein) encoded by a transgene. In some embodiments, the promoter is deemed to be efficient when it drives expression of the STXBP1 encoded in the viral genome of the AAV particle. [0177] In some embodiments, the promoter is a promoter deemed to be efficient when it drives expression in the cell or tissue being targeted.
Attorney Docket No.14640.0105-00304 [0178] Promoters may be naturally occurring or non-naturally occurring. Non-limiting examples of promoters include viral promoters, plant promoters, and mammalian promoters. In some embodiments, the promoters may be human promoters. In some embodiments, the promoters may be truncated. [0179] In some embodiments, the viral genome comprises a promoter that results in expression in one or more cells and/or tissues. In some embodiments, the promoter is a ubiquitous promoter. In some embodiments, a promoter that drives or promotes expression in most mammalian tissues comprises a human elongation factor 1α-subunit (EF1α) promoter, a cytomegalovirus (CMV) immediate-early enhancer and/or promoter, a chicken β-actin (CBA) promoter, a CAG promoter, a β glucuronidase (GUSB) promoter, or a ubiquitin C (UBC) promoter. [0180] In some embodiments, the viral genome comprises a nervous system specific promoter, i.e., a promoter that results in expression of STXBP1 in a neuron, an astrocyte, and/or an oligodendrocyte. Non-limiting examples of tissue-specific expression elements for neurons include synapsin (Syn) or synapsin 1 (Syn1), e.g., human synapsin or synapsin 1. [0181] In some embodiments, the promoter may be less than 1 kb. [0182] In some embodiments, the promoter may be a combination of two or more components of the same or different starting or parental promoters. [0183] In some embodiments, the viral genome comprises an enhancer. [0184] In some embodiments, the viral genome comprises an engineered promoter. Viral Genome Component: Intron and Exon Sequences [0185] In some embodiments, the AAV viral genome comprises at least one intron or a fragment or derivative thereof. In some embodiments, the AAV viral genome comprises at least one exon or a fragment or derivative thereof. [0186] In some embodiments, the intron may be 100-600 nucleotides in length. [0187] In some embodiments, the STXBP1-encoding sequence may be located downstream of an intron in an expression vector such as a beta globin intron or others known in the art. In some embodiments, the STXBP1-encoding sequence may be located within 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, or more than 30 nucleotides downstream from a promoter comprising an intron (e.g., 3’ relative to the promoter comprising an intron) and/or upstream of the polyadenylation sequence (e.g., 5’ relative to the polyadenylation sequence) in an expression vector. In some embodiments, the STXBP1-encoding sequence may be located within 1-5, 1- 10, 1-15, 1-20, 1-25, 1-30, 5-10, 5-15, 5-20, 5-25, 5-30, 10-15, 10-20, 10-25, 10-30, 15-20, 15-25, 15-30, 20-25, 20-30, or 25-30 nucleotides downstream from the intron (e.g., 3’ relative to the intron) and/or upstream of the polyadenylation sequence (e.g., 5’ relative to the polyadenylation sequence) in an expression vector. In some embodiments, the STXBP1-encoding sequence may be located within the first 1%, 2%, 3%, 4%, 5%, 6%, 7%, 8%, 9%, 10%, 15%, 20%, 25%, or more than 25% of the nucleotides downstream from the intron (e.g., 3’ relative to the intron) and/or upstream of the polyadenylation sequence (e.g., 5’ relative to the polyadenylation sequence) in an expression vector. In some
Attorney Docket No.14640.0105-00304 embodiments, the STXBP1-encoding sequence may be located within the first 1-5%, 1-10%, 1-15%, 1- 20%, 1-25%, 5-10%, 5-15%, 5-20%, 5-25%, 10-15%, 10-20%, 10-25%, 15-20%, 15-25%, or 20-25% of the sequence downstream from the intron (e.g., 3’ relative to the intron) and/or upstream of the polyadenylation sequence (e.g., 5’ relative to the polyadenylation sequence) in an expression vector. [0188] In certain embodiments, the intron sequence is not an enhancer sequence. In some embodiments, the intron sequence is not a sub-component of a promoter sequence. In some embodiments, the intron sequence is a sub-component of a promoter sequence. Viral Genome Component: Untranslated Regions (UTRs) [0189] In some embodiments, a wildtype untranslated region (UTR) of a gene is transcribed but not translated. Generally, the 5’ UTR starts at the transcription start site and ends at the start codon and the 3’ UTR starts immediately following the stop codon and continues until the termination signal for transcription. [0190] Features typically found in abundantly expressed genes of specific target organs may be engineered into UTRs to enhance the stability and protein production. [0191] In some embodiments, the viral genome encoding a transgene described herein (e.g., a transgene encoding an STXBP1 protein) comprises a Kozak sequence. Without being bound by theory, wild-type 5′ untranslated regions (UTRs) include features that play roles in translation initiation. Kozak sequences, which are commonly known to be involved in the process by which the ribosome initiates translation of many genes, are usually included in 5’ UTRs. Kozak sequences have the consensus CCR(A/G)CCAUGG, where R is a purine (adenine or guanine) three bases upstream of the start codon (ATG), which is followed by another 'G'. [0192] In some embodiments, the 5’UTR in the viral genome includes a Kozak sequence. [0193] In some embodiments, the 5’UTR in the viral genome does not include a Kozak sequence. [0194] Without being bound by theory, wild-type 3′ UTRs are known to have stretches of adenosines and uridines embedded therein. These AU rich signatures are particularly prevalent in genes with high rates of turnover. Based on their sequence features and functional properties, the AU rich elements (AREs) can be separated into three classes (Chen et al, 1995, the contents of which are herein incorporated by reference in their entirety): Class I AREs, such as, but not limited to, c-Myc and MyoD, contain several dispersed copies of an AUUUA motif within U-rich regions. Class II AREs, such as, but not limited to, GM-CSF and TNF-a, possess two or more overlapping UUAUUUA(U/A)(U/A) nonamers. Class III ARES, such as, but not limited to, c-Jun and Myogenin, are less well defined. These U rich regions do not contain an AUUUA motif. Most proteins binding to the AREs are known to destabilize the messenger, whereas members of the ELAV family, most notably HuR, have been documented to increase the stability of mRNA. HuR binds to AREs of all the three classes. Engineering the HuR specific binding sites into the 3′ UTR of nucleic acid molecules will lead to HuR binding and thus, stabilization of the message in vivo. [0195] Introduction, removal or modification of 3′ UTR AU rich elements (AREs) can be used to
Attorney Docket No.14640.0105-00304 modulate the stability of polynucleotides. When engineering specific polynucleotides, one or more copies of an ARE can be introduced to make polynucleotides less stable and thereby curtail translation and decrease production of the resultant protein. Likewise, AREs can be identified and removed or mutated to increase the intracellular stability and thus increase translation and production of the resultant protein. [0196] In some embodiments, the 3' UTR of the viral genome may include an oligo(dT) sequence for templated addition of a poly-A tail. [0197] Any UTR from any gene known in the art may be incorporated into the viral genome of the AAV particle. These UTRs, or portions thereof, may be placed in the same orientation as in the gene from which they were selected or they may be altered in orientation or location. In some embodiments, the UTR used in the viral genome of the AAV particle may be inverted, shortened, lengthened, or made with one or more other 5′ UTRs or 3′ UTRs known in the art. As used herein, the term “altered,” as it relates to a UTR, means that the UTR has been changed in some way in relation to a reference sequence. For example, a 3′ or 5′ UTR may be altered relative to a wild type or native UTR by the change in orientation or location as taught above or may be altered by the inclusion of additional nucleotides, deletion of nucleotides, swapping or transposition of nucleotides. [0198] In some embodiments, the viral genome of the AAV particle comprises at least one artificial UTR, which is not a variant of a wild type UTR. [0199] In some embodiments, the viral genome of the AAV particle comprises UTRs which have been selected from a family of transcripts whose proteins share a common function, structure, feature, or property. Viral Genome Component: Polyadenylation Region [0200] In some embodiments, the viral genome of the AAV particles of the present disclosure comprises at least one polyadenylation (polyA) sequence. In some embodiments, the polyA signal region is positioned 3’ relative to the nucleic acid comprising the STXBP1-encoding sequence. Viral Genome Component: Filler (Stuffer) Sequence [0201] As used herein, the terms “stuffer sequence” and “filler sequence” are used interchangeably. In some embodiments, the AAV particle viral genome comprises at least one filler sequence. [0202] In some embodiments, the viral genome comprises one or more filler sequences. The filler sequence may be a wild-type sequence or an engineered sequence. A filler sequence may be a variant of a wild-type sequence. [0203] In some embodiments, the viral genome comprises one or more filler sequences in order to have the length of the viral genome be the optimal size for packaging. In some embodiments, the viral genome comprises at least one filler sequence in order to have the length of the viral genome be about 2.3 kb. In some embodiments, the viral genome comprises at least one filler sequence in order to have the length of the viral genome be about 4.6 kb. Viral Genome Component: STXBP1-encoding sequence [0204] In some embodiments, the disclosure provides an AAV particle comprising a viral genome
Attorney Docket No.14640.0105-00304 encoding an STXBP1 protein, e.g., an STXBP1 protein encoded by the nucleotide sequence of SEQ ID NO: 6414. In some embodiments, the viral genome comprises a promoter operably linked to a nucleotide sequence encoding an STXBP1 protein, e.g., SEQ ID NO: 6414. [0205] In some embodiments, the disclosure herein provides constructs that allow for improved expression of STXBP1 protein delivered by gene therapy vectors. [0206] In some embodiments, the disclosure provides constructs that allow for improved biodistribution of STXBP1 protein delivered by gene therapy vectors. [0207] In some embodiments, the disclosure provides constructs that allow for improved sub-cellular distribution or trafficking of STXBP1 protein delivered by gene therapy vectors. [0208] In some embodiments, the disclosure provides constructs that allow for improved trafficking of STXBP1 protein to lysosomal membranes delivered by gene therapy vectors. [0209] In some embodiments, the present disclosure relates to a composition containing or comprising a nucleic acid sequence encoding an STXBP1 protein or a functional fragment or variant thereof and methods of administering the composition in vitro or in vivo in a subject, e.g., a human subject and/or an animal model of disease, e.g., a disease related to expression of STXBP1. [0210] In some embodiments, the nucleotide sequence comprises one or more, e.g., all of, a 5’ ITR sequence, an enhancer sequence, a promoter sequence, an intron sequence, a signal sequence, an STXBP1-encoding sequence, a polyA sequence, and a 3’ ITR sequence. In some embodiments, the STXBP1 protein encoded by the nucleotide sequence has an amino acid sequence that is 100% identical to a wildtype STXBP1 protein. [0211] In some embodiments, the AAV genome comprises a payload construct that comprises a combination of coding and non-coding nucleic acid sequences. [0212] In some embodiments, the viral genome encodes more than one payload. As a non-limiting example, a viral genome encoding more than one payload may be replicated and packaged into a viral particle. A target cell transduced with a viral particle comprising more than one payload may express each of the payloads in a single cell. [0213] In some embodiments, the STXBP1-encoding sequence comprises a gene therapy product including, but not limited to, a polypeptide, protein, RNA molecule, or other gene product that, when expressed in a target cell, provides a desired therapeutic effect. In some embodiments, a gene therapy product may comprise a substitute for a non-functional gene or a gene that is absent, expressed in insufficient amounts, or mutated. In some embodiments, a gene therapy product may comprise a substitute for a non-functional protein or polypeptide or a protein or polypeptide that is absent, expressed in insufficient amounts, misfolded, degraded too rapidly, or mutated. For example, a gene therapy product may comprise a polynucleotide encoding an STXBP1 protein to treat STXBP1 deficiency or STXBP1-related disorders. In some embodiments, the gene therapy product comprises a polynucleotide sequence encoding an STXBP1 protein. [0214] In some embodiments, the payload construct encodes a messenger RNA (mRNA). As used
Attorney Docket No.14640.0105-00304 herein, the term “messenger RNA” (mRNA) refers to any polynucleotide that encodes a polypeptide of interest and that is capable of being translated to produce the encoded polypeptide of interest in vitro, in vivo, in situ, or ex vivo. Certain embodiments provide the mRNA as encoding STXBP1 or a variant thereof. [0215] In some embodiments, an STXBP1-encoding sequence may comprise or encode a selectable marker. In some embodiments, a selectable marker may comprise a gene sequence or a protein or polypeptide encoded by a gene sequence expressed in a host cell that allows for the identification, selection, and/or purification of the host cell from a population of cells that may or may not express the selectable marker. In some embodiments, the selectable marker provides resistance to survive a selection process that would otherwise kill the host cell, such as treatment with an antibiotic. In some embodiments, an antibiotic selectable marker may comprise one or more antibiotic resistance factors, including but not limited to neomycin resistance (e.g., neo), hygromycin resistance, kanamycin resistance, and/or puromycin resistance. [0216] In some embodiments, an STXBP1-encoding sequence may comprise a selectable marker including, but not limited to, β-lactamase, luciferase, β-galactosidase, or any other reporter gene as that term is understood in the art, including cell-surface markers, such as CD4 or the truncated nerve growth factor (NGFR) (for GFP, see WO 96/23810; Heim et al., Current Biology 2:178-182 (1996); Heim et al., Proc. Natl. Acad. Sci. USA (1995); or Heim et al., Science 373:663-664 (1995); for β-lactamase, see WO 96/30540); the contents of each of which are herein incorporated by reference in their entirety. [0217] In some embodiments, an STXBP1-encoding sequence may encode a selectable marker comprising a fluorescent protein. A fluorescent protein as herein described may comprise any fluorescent marker including but not limited to green, yellow, and/or red fluorescent protein (GFP, YFP, and/or RFP). In some embodiments, STXBP1-encoding sequence may encode a selectable marker comprising a human influenza hemagglutinin (HA) tag. [0218] In certain embodiments, a nucleic acid for expression of STXBP1 protein in a target cell as described herein will be incorporated into the viral genome and located between two ITR sequences. Viral Genome Component: Signal Sequence [0219] In some embodiments, the viral genome encoding an STXBP1 protein further comprises a nucleotide sequence encoding a signal sequence. [0220] In some embodiments, the nucleotide sequence encoding the signal sequence is located 5’ relative to the nucleotide sequence encoding the STXBP1 protein. In some embodiments, the encoded STXBP1 protein comprises a signal sequence at the N-terminus, wherein the signal sequence is optionally cleaved during cellular processing and/or localization of the STXBP1 protein. Exemplary STXBP1 Payload [0221] In some embodiments, the encoded STXBP1 protein is a wildtype STXBP1 protein. [0222] Tables 7 and 8 provide exemplary polynucleotide sequences encoding an STXBP1 protein and polypeptide sequences of exemplary STXBP1 proteins that may be used in the viral genomes
Attorney Docket No.14640.0105-00304 disclosed herein and which may constitute an STXBP1 protein payload. In some embodiments, the STXBP1 protein suitable for delivery in an AAV disclosed herein is encoded by the nucleotide sequence of SEQ ID NO: 6414. In some embodiments, the STXBP1 protein suitable for delivery in an AAV disclosed herein is encoded by the nucleotide sequence of SEQ ID NO: 6416, 6418, 6420, 6422, 6424, 6426, 6428, 6430, 6432, 6434, or 6436. Table 7. Exemplary STXBP1 Sequences SEQ ID NO: Type Species Description 6413 Protein Homo sapiens STXBP1 protein NP_001027392.1 “

Attorney Docket No.14640.0105-00304 6423 Protein Homo sapiens STXBP1 protein NP_001361239.1 “syntaxin-binding protein 1 isoform e [Homo sapiens]”, National Center for
Attorney Docket No.14640.0105-00304 “syntaxin-binding protein 1 isoform a [Homo sapiens]”, National Center for Biotechnology Information, Mar 18, 2023 Table 8
Description Sequence SEQ ID NO: T
XBP1 t AT ATT T AAA T TT T A A AA ATTAT AT 414
Attorney Docket No.14640.0105-00304 HIAEVSQEVTRSLKDFSSSKRMNTGEKTTMRDLSQMLKKMPQYQKELSK YSTHLHLAEDCMKHYQGTVDKLCRVEQDLAMGTDAEGEKIKDPMRAIVP ILLDANVSTYDKIRIILLYIFLKNGITEENLNKLIQHAQIPPEDSEIIT

Information cited in Tables 7 and 8 is hereby incorporated by reference in its entirety. [0224] In some embodiments, the present disclosure provides a viral genome comprising an STXBP1-encoding sequence comprising SEQ ID NO: 6414 or a nucleotide sequence having at least 75%, at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity to SEQ ID NO: 6414. In some embodiments, the STXBP1-encoding sequence is at least 95% identical to SEQ ID NO: 6414. In some embodiments, the STXBP1-encoding sequence is at least 99% identical to SEQ ID NO: 6414. In some embodiments, the STXBP1-encoding sequence comprises SEQ ID NO: 6414. In some embodiments, the STXBP1-encoding sequence consists of SEQ ID NO: 6414. [0225] In some embodiments, the AAV viral genome further comprises a nucleic acid encoding a capsid protein, e.g., a structural protein. In some embodiments, the capsid protein comprises a VP1 polypeptide, a VP2 polypeptide, and/or a VP3 polypeptide. In some embodiments, the VP1 polypeptide, the VP2 polypeptide, and/or the VP3 polypeptide are encoded by at least one Cap gene. In some embodiments, the AAV viral genome further comprises a nucleic acid encoding a Rep protein, e.g., a non-structural protein. In some embodiments, the Rep protein comprises a Rep78 protein, a Rep68, Rep52 protein, and/or a Rep40 protein. In some embodiments, the Rep78 protein, the Rep68 protein, the Rep52 protein, and/or the Rep40 protein are encoded by at least one Rep gene. [0226] In some embodiments, the capsid comprises the amino acid sequence of SEQ ID NO: 981. [0227] In some embodiments, the AAV particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 6414, or a sequence having at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%) sequence identity thereto, comprises a capsid comprising an amino acid sequence selected from Table 3 or Table 4. [0228] In some embodiments, the AAV particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 6414, or a sequence having at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%) sequence identity thereto, comprises a capsid comprising (i) a VP1 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 981 or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) a VP2 protein comprising or consisting of the amino acid sequence according to positions 138-736 of SEQ ID NO: 981 or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least
Attorney Docket No.14640.0105-00304 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (iii) a VP3 protein comprising or consisting of the amino acid sequence according to positions 203-736 of SEQ ID NO: 981 or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, wherein the capsid in (i), (ii), or (iii) comprises T at amino acid 584, D at amino acid 586, W at amino acid 587, H at amino acid 588, R at amino acid 589, and/or I at amino acid 590 of the amino acid sequence of SEQ ID NO: 981. [0229] In some embodiments, the AAV particle comprises a viral genome that is packaged in a capsid comprising the amino acid T at position 584, D at position 586, W at position 587, H at position 588, R at position 589, and I at position 590, numbered according to SEQ ID NO: 981. In some embodiments, the AAV capsid variant comprises the amino acid sequence of amino acids 203-736 of SEQ ID NO: 981, or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence of amino acids 138-736 of SEQ ID NO: 981, or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence SEQ ID NO: 981, or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [0230] In some embodiments, the AAV particle comprises a viral genome that is packaged in a capsid comprising the amino acid T at a position corresponding to position 584 of SEQ ID NO: 981, D at a position corresponding to position 586 of SEQ ID NO: 981, W at a position corresponding to position 587 of SEQ ID NO: 981, H at a position corresponding to position 588 of SEQ ID NO: 981, R at a position corresponding to position 589 of SEQ ID NO: 981, and I at a position corresponding to position 590 of SEQ ID NO: 981. In some embodiments, the AAV capsid variant comprises the amino acid sequence of amino acids 203-736 of SEQ ID NO: 981, or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence of amino acids 138-736 of SEQ ID NO: 981, or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence SEQ ID NO: 981, or an amino acid sequence that is at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [0231] In some embodiments, the present disclosure provides an AAV particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 6414 and an AAV capsid variant comprising the amino acid sequence of amino acids 138-736 of SEQ ID NO: 981. [0232] In some embodiments, the present disclosure provides an AAV particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 6414 and an AAV capsid variant comprising the amino acid sequence of amino acids 203-736 of SEQ ID NO: 981.
Attorney Docket No.14640.0105-00304 [0233] In some embodiments, the present disclosure provides an AAV particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 6414 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 981. [0234] The present disclosure provides in some embodiments, vectors, cells, and/or AAV particles comprising the above identified viral genomes. Self-Complementary and Single Stranded Viral Genomes [0235] In some embodiments, the AAV viral genome used in the present disclosure is single- stranded (ssAAV). [0236] In some embodiments, the AAV viral genome is capable of forming double-stranded DNA. In some embodiments, the AAV viral genome is self-complementary. See, e.g., US Patent No.7,465,583. scAAV particles contain both DNA strands that anneal together to form double stranded DNA. By skipping second strand synthesis, scAAVs allow for rapid expression in the cell. [0237] Methods for producing and/or modifying AAV viral genome and particles are disclosed in the art such as pseudotyped AAV particles (International Patent Publication Nos. WO200028004; WO200123001; WO2004112727; WO 2005005610 and WO 2005072364, the content of each of which are incorporated herein by reference in their entirety). II. AAV Production [0238] Viral production disclosed herein describes processes and methods for producing AAV particles (with enhanced, improved and/or increased tropism for a target tissue), e.g., an AAV particle comprising an AAV capsid variant that may be used to contact a target cell to deliver STXBP1. [0239] In some embodiments, disclosed herein is a method of making an AAV particle of the present disclosure, e.g., an AAV particle comprising an AAV capsid variant disclosed herein, wherein the method comprises: (i) providing a cell comprising a viral genome described herein (e.g., a viral genome comprising an STXBP1-encoding sequence and a nucleic acid encoding an AAV capsid variant disclosed herein) and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant, e.g., an AAV capsid variant described herein (e.g., an AAV capsid variant listed in Tables 3, 4, or 5), thereby making the AAV particle. In some embodiments, the viral genome comprises an STXBP1-encoding sequence comprising SEQ ID NO: 6414. In some embodiments, the AAV capsid variant comprises an amino acid sequence of SEQ ID NO: 981. In some embodiments, the viral genome comprises an STXBP1-encoding sequence comprising SEQ ID NO: 6414 and the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 981. In some embodiments, the method comprises, prior to step (i), introducing a nucleic acid comprising the viral genome into the cell. In some embodiments, the method comprises, prior to step (i), introducing the nucleic acid encoding the AAV capsid variant into the cell. In some embodiments, the AAV particle described herein is an isolated AAV particle. In some embodiments, the AAV particle described herein is a recombinant AAV particle.
Attorney Docket No.14640.0105-00304 [0240] Any method known in the art may be used for the preparation of AAV particles. In some embodiments, AAV particles are produced in mammalian cells (e.g., HEK293 cells). In some embodiments, AAV particles are produced in insect cells (e.g., Sf9 cells). [0241] Methods of making AAV particles are well known in the art and are described in e.g., U.S. Patent Nos. US6204059, US5756283, US6258595, US6261551, US6270996, US6281010, US6365394, US6475769, US6482634, US6485966, US6943019, US6953690, US7022519, US7238526, US7291498 and US7491508, US5064764, US6194191, US6566118, US8137948; or International Publication Nos. WO1996039530, WO1998010088, WO1999014354, WO1999015685, WO1999047691, WO2000055342, WO2000075353 and WO2001023597; Methods In Molecular Biology, ed. Richard, Humana Press, NJ (1995); O'Reilly et al., Baculovirus Expression Vectors, A Laboratory Manual, Oxford Univ. Press (1994); Samulski et al., J. Vir.63:3822-8 (1989); Kajigaya et al., Proc. Nat'l. Acad. Sci. USA 88: 4646-50 (1991); Ruffing et al., J. Vir.66:6922-30 (1992); Kimbauer et al., Vir., 219:37-44 (1996); Zhao et al., Vir.272:382-93 (2000); the contents of each of which are herein incorporated by reference in their entirety. In some embodiments, the AAV particles are made using the methods described in International Patent Publication WO2015191508, the contents of which are herein incorporated by reference in their entirety. III. Pharmaceutical Compositions [0242] In some embodiments, the present disclosure provides pharmaceutical compositions of an adeno-associated virus (AAV) particle comprising a viral genome comprising an STXBP1-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 6414 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 981. [0243] In some embodiments, a composition described herein comprises an AAV polynucleotide or AAV genome or AAV particle, and at least one excipient. [0244] Although pharmaceutical compositions provided herein, e.g., comprising AAV particles comprising an STXBP1-encoding sequence, are principally directed to pharmaceutical compositions that are suitable for administration to humans, it will be understood by the skilled artisan that such compositions may be suitable for administration to any other animal, e.g., non-human mammals. Modification of pharmaceutical compositions suitable for administration to humans in order to render the compositions suitable for administration to various non-human animals is well understood, and the ordinarily skilled veterinary pharmacologist can design and/or perform such modification with merely ordinary, if any, experimentation. Subjects to which administration of the pharmaceutical compositions is contemplated include, but are not limited to, humans and/or other primates; mammals, including commercially relevant mammals such as cattle, pigs, horses, sheep, cats, dogs, mice, and/or rats; and/or birds, including commercially relevant birds such as poultry, chickens, ducks, geese, and/or turkeys. [0245] In some embodiments, compositions are administered to humans, e.g., human patients or human subjects.
Attorney Docket No.14640.0105-00304 [0246] In some embodiments, the AAV particle formulations described herein may contain a nucleic acid encoding at least one STXBP1-encoding sequence. In some embodiments, the formulations may contain a nucleic acid encoding 1, 2, 3, 4, or 5 STXBP1-encoding sequences. In some embodiments, the formulation may contain STXBP1-encoding sequences encoding STXBP1 proteins selected from categories such as, but not limited to, human proteins, veterinary proteins, bacterial proteins, biological proteins, antibodies, immunogenic proteins, therapeutic peptides and proteins, secreted proteins, plasma membrane proteins, cytoplasmic proteins, cytoskeletal proteins, intracellular membrane bound proteins, nuclear proteins, proteins associated with human disease, and/or proteins associated with non-human diseases. In some embodiments, the AAV formulation comprises at least one sequence encoding human STXBP1. In some embodiments, the AAV formulation comprises at least one sequence encoding wildtype human STXBP1. [0247] A pharmaceutical composition in accordance with the present disclosure may be prepared, packaged, and/or sold in bulk, as a single unit dose, and/or as a plurality of single unit doses. As used herein, a “unit dose” refers to a discrete amount of the pharmaceutical composition comprising a predetermined amount of the active ingredient. The amount of the active ingredient is generally equal to the dosage of the active ingredient which would be administered to a subject and/or a convenient fraction of such a dosage such as, for example, one-half or one-third of such a dosage. IV. Formulations [0248] Formulations of the AAV pharmaceutical compositions described herein may be prepared by any method known or hereafter developed in the art of pharmacology. In general, such preparatory methods include the step of bringing the active ingredient into association with an excipient and/or one or more other accessory ingredients, and then, if necessary and/or desirable, dividing, shaping and/or packaging the product into a desired single- or multi-dose unit. [0249] Relative amounts of the active ingredient, the pharmaceutically acceptable excipient, and/or any additional ingredients in a pharmaceutical composition in accordance with the disclosure will vary, depending upon the identity, size, and/or condition of the subject treated and further depending upon the route by which the composition is to be administered. [0250] For example, the composition may comprise about 0.1% to about 99% (w/w) of the active ingredient. By way of example, the composition may comprise about 0.1% to about 100%, e.g., about 0.5% to about 50%, about 1% to about 30%, about 5% to about 80%, or at least 80% (w/w) active ingredient. [0251] The AAV particles of the disclosure can be formulated using one or more excipients to: (1) increase stability; (2) increase cell transfection or transduction; (3) permit the sustained or delayed release; (4) alter the biodistribution (e.g., target the viral particle to specific tissues or cell types); (5) increase the translation of encoded protein in vivo; (6) alter the release profile of encoded protein in vivo and/or (7) allow for regulatable expression of STXBP1.
Attorney Docket No.14640.0105-00304 [0252] Formulations of the present disclosure can include, without limitation, saline, lipidoids, liposomes, lipid nanoparticles, polymers, lipoplexes, core-shell nanoparticles, peptides, proteins, cells transfected with viral vectors (e.g., for transplantation into a subject), nanoparticle mimics and combinations thereof. Further, the viral vectors of the present disclosure may be formulated using self- assembled nucleic acid nanoparticles. [0253] In some embodiments, the viral vectors encoding STXBP1 may be formulated to optimize baricity and/or osmolality. In some embodiments, the baricity and/or osmolality of the formulation may be optimized to ensure optimal drug distribution in the central nervous system or a region or component of the central nervous system. Excipients [0254] The formulations of the disclosure can include one or more excipients, each in an amount that together increases the stability of the AAV particle, increases cell transfection or transduction by the viral particle, increases the expression of viral particle encoded protein, and/or alters the release profile of AAV particle encoded proteins. In some embodiments, a pharmaceutically acceptable excipient may be at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% pure. In some embodiments, an excipient is approved for use for humans and for veterinary use. In some embodiments, an excipient may be approved by United States Food and Drug Administration. In some embodiments, an excipient may be of pharmaceutical grade. In some embodiments, an excipient may meet the standards of the United States Pharmacopoeia (USP), the European Pharmacopoeia (EP), the British Pharmacopoeia, and/or the International Pharmacopoeia. [0255] Excipients, which, as used herein, include, but are not limited to, any and all solvents, dispersion media, diluents, or other liquid vehicles, dispersion or suspension aids, surface active agents, isotonic agents, thickening or emulsifying agents, preservatives, and the like, as suited to the particular dosage form desired. Various excipients for formulating pharmaceutical compositions and techniques for preparing the composition are known in the art (see Remington: The Science and Practice of Pharmacy, 21st Edition, A. R. Gennaro, Lippincott, Williams & Wilkins, Baltimore, MD, 2006; the contents of which are herein incorporated by reference in their entirety). The use of a conventional excipient medium may be contemplated within the scope of the present disclosure, except insofar as any conventional excipient medium may be incompatible with a substance or its derivatives, such as by producing any undesirable biological effect or otherwise interacting in a deleterious manner with any other component(s) of the pharmaceutical composition. Inactive Ingredients [0256] In some embodiments, AAV formulations may comprise at least one excipient which is an inactive ingredient. As used herein, the term “inactive ingredient” refers to one or more agents that do not contribute to the activity of the pharmaceutical composition included in formulations. In some embodiments, all, none, or some of the inactive ingredients which may be used in the formulations of the present disclosure may be approved by the US Food and Drug Administration (FDA).
Attorney Docket No.14640.0105-00304 [0257] Formulations of AAV particles may include cations or anions. In one embodiment, the formulations include metal cations such as, but not limited to, Zn
2+, Ca
2+, Cu
2+, Mg
+, or combinations thereof. In some embodiments, formulations may include polymers or polynucleotides complexed with a metal cation (See, e.g., U.S. Pat. Nos.6,265,389 and 6,555,525, the contents of each of which are herein incorporated by reference in their entirety). V. Uses and Applications [0258] The compositions of the disclosure may be administered to a subject, e.g., to deliver STXBP1, e.g., to a subject who has, has been diagnosed with having, or is at risk of having an STXBP1-related disorder. A STXBP-related disorder refers to any disease, disorder, or condition associated with or caused by STXBP1 protein deficiency, such as an STXBP1-related neurodegenerative or neuromuscular disorder (e.g., STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5)). The compositions may similarly be used in the manufacture of a medicament for administration to a subject having an STXBP1-related disorder (e.g., an STXBP1-related neurodegenerative or neuromuscular disorder). In some embodiments, the STXBP1-related disorder is STXBP1 encephalopathy. [0259] In some embodiments, the disclosure provides a method of delivering an STXBP1 protein to a subject comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle disclosed herein that comprises a viral genome encoding an STXBP1 protein, thereby delivering the STXBP1 protein. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having an STXBP1-related disorder. The STXBP1-related disorder may be an STXBP1- related disorder such as an STXBP1-related neurodegenerative or neuromuscular disorder (e.g., STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5)). In some embodiments, the STXBP1-related disorder is STXBP1 encephalopathy. [0260] In some embodiments, the disclosure provides a method for treating an STXBP1-related disorder such as an STXBP1-related neurodegenerative or neuromuscular disorder. In some embodiments, the disclosure provides an AAV particle or pharmaceutical composition according to any one of the embodiments disclosed herein for treating an STXBP1-related disorder, such as STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). In some
Attorney Docket No.14640.0105-00304 embodiments, the present disclosure provides the pharmaceutical composition or the AAV particle of any one the embodiments disclosed herein for use in a method of treating a disorder as disclosed herein, such as STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). In some embodiments, the disorder is STXBP1 encephalopathy. [0261] In certain embodiments, a pharmaceutical composition or AAV particle disclosed herein that comprises a viral genome encoding STXBP1 may be administered to a subject to treat an STXBP1- related disorder such as an STXBP1-related neurodegenerative or neuromuscular disorder, thereby treating the STXBP1-related disorder. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having an STXBP1-related disorder such as an STXBP1-related neurodegenerative or neuromuscular disorder (e.g., STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox- Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5)). In some embodiments, the treatment results in an increase in the subject’s STXBP1 protein level as compared to baseline. [0262] In some embodiments, the STXBP1-related disorder is STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). In some embodiments, the STXBP1-related disorder is STXBP1 encephalopathy. [0263] In some embodiments, the disclosure provides a method of treating STXBP1 encephalopathy in a subject. In certain embodiments, a pharmaceutical composition or AAV particle disclosed herein that comprises a viral genome encoding STXBP1 may be administered to a subject to treat STXBP1 encephalopathy. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having STXBP1 encephalopathy. [0264] A subject may have one or more mutations in the STXBP1 gene. In some embodiments, the subject has lower STXBP1 activity as compared to STXBP1 activity in an individual who does not have an STXBP1-related disorder. [0265] In some embodiments, the treatment may result in prevention of progression of the STXBP1- related disorder. For example, the treatment may result in in amelioration of at least one symptom of the disorder, as indicated by one or more biomarkers. In some embodiments, the one or more biomarkers comprises increased release of the neurotransmitters glutamate and/or GABA or reduction in abnormal electroencephalographic activity as evidence of improved STXBP1 activity.
Attorney Docket No.14640.0105-00304 [0266] In certain embodiments, the treatment improves at least one symptom of an STXBP1-related disorder, e.g., epilepsy, autistic features, ataxia, generalized tremors, reduced STXBP1 activity, accumulation of glucocerebroside and other glycolipids, e.g., within immune cells (e.g., macrophages), build-up of synuclein aggregates (e.g., Lewy bodies), developmental delay, progressive encephalopathy, progressive dementia, ataxia, myoclonus, oculomotor dysfunction, bulbar palsy, generalized weakness, trembling of a limb, depression, visual hallucinations, cognitive decline, dystonia, or a combination thereof. [0267] In some embodiments, the methods disclosed herein further comprise evaluating, e.g., measuring, the level of STXBP1 expression, e.g., STXBP1 gene expression, STXBP1 mRNA expression, and/or STXBP1 protein expression, in the subject, e.g., in a cell, tissue, or fluid of the subject. STXBP1 protein expression may be measured by an ELISA, a Western blot, or an immunohistochemistry assay. In some embodiments, evaluating the level of STXBP1 expression is performed prior to and/or subsequent to administration of the AAV particle, optionally wherein the level of STXBP1 expression prior to treatment is compared to the level of STXBP1 expression subsequent to administration. In some embodiments, the level of STXBP1 expression may be evaluated in a cell or tissue of the central nervous system. In some embodiments, the level of STXBP1 expression may be evaluated in a cell of a peripheral tissue (e.g., liver, heart, muscle, or spleen). In some embodiments, the cell of a peripheral tissue is a muscle cell. In some embodiments, the subject’s level of STXBP1 protein expression subsequent to administration is increased relative to the subject’s level of STXBP1 protein expression prior to administration. [0268] In some embodiments, the administration of the effective amount of a pharmaceutical composition or AAV particle disclosed herein that comprises a viral genome encoding an STXBP1 protein may be a treatment that results in an increase in: (i) the level of STXBP1 activity in a cell, tissue, (e.g., a cell or tissue of the CNS and/or the PNS, e.g., the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron), and/or fluid (e.g., CSF and/or serum), of the subject, relative to baseline and/or relative to the level of STXBP1 activity in a cell, tissue, or fluid of an individual with an STXBP1-related disorder who has not been administered the pharmaceutical composition or AAV particle; and/or (ii) the level and/or number of viral genomes (VG) per cell in a tissue of the CNS and/or the PNS, e.g., the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior
Attorney Docket No.14640.0105-00304 olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron) of the subject relative to the number and/or level of VG per cell in a peripheral tissue of the subject; and/or (iii) the level of STXBP1 mRNA expression in a cell or tissue (e.g., a cell or tissue of the CNS and/or the PNS, e.g., the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron)) of the subject relative to baseline and/or relative to the level of STXBP1 mRNA expression in a cell or tissue of an individual with an STXBP1-related disorder who has not been administered the pharmaceutical composition or AAV particle. [0269] In some embodiments, at least one additional agent suitable for treatment of an STXBP1- related disorder may be administered together with the effective amount of a pharmaceutical composition or AAV particle disclosed herein (comprising a viral genome encoding an STXBP1 protein). The at least one additional agent may comprise one or more anti-epileptic drugs (e.g., bromide, clobazam, felbamate, ganaxolone, lamotrigine, levetiracetam, phenobarbital, topiramate, valproate, or a combination thereof). [0270] In some embodiments, the present disclosure encompasses the delivery of pharmaceutical, prophylactic, diagnostic, or imaging compositions in combination with agents that may improve their bioavailability, reduce and/or modify their metabolism, and/or modify their distribution within the body. [0271] In certain embodiments, the pharmaceutical compositions described herein are used as research tools, particularly in in vitro investigations using human cell lines such as HEK293T and in vivo testing in nonhuman primates which will occur prior to human clinical trials. [0272] The present disclosure provides a method for treating a disease, disorder and/or condition in a mammalian subject, including a human subject, comprising administering to the subject any of the viral particles, e.g., AAV, AAV particles, or AAV genome that produces STXBP1 protein described herein (i.e., viral genomes or “VG”), administering to the subject a composition or formulation comprising said AAV particle. [0273] Delivery of a payload construct comprising an STXBP1-encoding sequence may alleviate or reduce symptoms that result from abnormal level and/or function of a gene product (e.g., an absence or defect in a protein) in a subject in need thereof or that otherwise confers a benefit to a CNS disorder in a subject in need thereof.
Attorney Docket No.14640.0105-00304 VI. Delivery of AAV Particles Delivery to Cells [0274] In some aspects, the present disclosure provides a method of delivering to a cell or tissue any of the above-described AAV particles, comprising contacting the cell or tissue with said AAV particle or contacting the cell or tissue with a formulation comprising said AAV particle, or contacting the cell or tissue with any of the described compositions, including pharmaceutical compositions. The method of delivering the AAV particle to a cell or tissue can be accomplished in vitro, ex vivo, or in vivo. [0275] In some embodiments, the AAV particles are delivered to a cell, tissue, or region of the CNS. In some embodiments, the AAV particles are delivered to a cell or tissue of the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron). Delivery to Subjects [0276] In some aspects, the present disclosure additionally provides a method of delivering to a subject, including a mammalian subject, any of the above-described AAV particles comprising administering to the subject said AAV particle, or administering to the subject a formulation comprising said AAV particle, or administering to the subject any of the described compositions, including pharmaceutical compositions. [0277] In some embodiments, the AAV particles may be delivered to bypass anatomical blockages (e.g., the blood brain barrier). [0278] In some embodiments, the AAV particles may be formulated and delivered to a subject by a route which increases the speed of drug effect as compared to oral delivery. [0279] In some embodiments, the AAV particles may be delivered using intrathecal infusion. [0280] In some embodiments, a subject may be administered the AAV particles described herein using a bolus infusion. [0281] In some embodiments, the AAV particles may be delivered in a continuous and/or bolus infusion. Each site of delivery may use a different dosing regimen or the same dosing regimen may be used for each site of delivery. As a non-limiting example, the sites of delivery may be in the cervical and the lumbar region. As another non-limiting example, the sites of delivery may be in the cervical region. As another non-limiting example, the sites of delivery may be in the lumbar region. [0282] In some embodiments, the AAV particles may be delivered to a subject via a single route of administration.
Attorney Docket No.14640.0105-00304 [0283] In some embodiments, the AAV particles may be delivered to a subject via a multi-site route of administration. For example, a subject may be administered the AAV particles at 2, 3, 4, 5, or more than 5 sites. [0284] In some embodiments, a subject may be administered the AAV particles described herein using sustained delivery over a period of minutes, hours, or days. The infusion rate may be changed depending on the subject, distribution, formulation, or another delivery parameter known to those in the art. [0285] In some embodiments, if continuous delivery (continuous infusion) of the AAV particles is used, the continuous infusion may be for 1 hour, 2, hours, 3 hours, 4 hours, 5 hours, 6 hours, 7 hours, 8 hours, 9 hours, 10 hours, 11 hours, 12 hours, 13 hours, 14 hours, 15 hours, 16 hours, 17 hours, 18 hours, 19 hours, 20 hours, 21 hours, 22 hours, 23 hours, 24 hours, or more than 24 hours. [0286] In some embodiments, the intracranial pressure may be evaluated prior to administration. The route, volume, AAV particle concentration, infusion duration and/or vector titer may be optimized based on the intracranial pressure of a subject. [0287] In some embodiments, the AAV particles may be delivered by systemic delivery. In some embodiments, the systemic delivery may be by intravascular administration. In some embodiments, the systemic delivery may be by intravenous administration. [0288] In some embodiments, the AAV particles may be delivered by injection into the CSF pathway. Non-limiting examples of delivery to the CSF pathway include intrathecal and intracerebroventricular administration. [0289] In some embodiments, an AAV particle described herein is administered intravenously. [0290] In some embodiments, the AAV particles may be delivered by direct (intraparenchymal) injection into the substance of an organ, e.g., one or more regions of the brain. [0291] In some embodiments, the AAV particles may be delivered by subpial injection into the spinal cord. For example, subjects may be placed into a spinal immobilization apparatus. A dorsal laminectomy may be performed to expose the spinal cord. Guiding tubes and XYZ manipulators may be used to assist catheter placement. Subpial catheters may be placed into the subpial space by advancing the catheter from the guiding tube and AAV particles may be injected through the catheter (Miyanohara et al., Mol Ther Methods Clin Dev.2016; 3: 16046). In some cases, the AAV particles may be injected into the cervical subpial space. In some cases, the AAV particles may be injected into the thoracic subpial space. [0292] In some embodiments, the AAV particles may be delivered by direct injection to the CNS of a subject. In some embodiments, direct injection is intracerebral injection, intraparenchymal injection, intrathecal injection, intra-cisterna magna injection, or any combination thereof. In some embodiments, direct injection to the CNS of a subject comprises convection enhanced delivery (CED). In some embodiments, administration comprises peripheral injection. In some embodiments, peripheral injection is intravenous injection.
Attorney Docket No.14640.0105-00304 [0293] In some embodiments, the AAV particles may be delivered to a subject in order to increase an STXBP1 protein level in the CNS and/or PNS (e.g., the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron)) as compared to a baseline level in the subject. [0294] In some embodiments, the AAV particles may be delivered to a subject in order to increase an STXBP1 protein level in the CNS and/or PNS (e.g., the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron)) by transducing cells in these CNS regions. Transduction may also be referred to as the number of cells that are positive for STXBP1 protein. [0295] In some embodiments, delivery of AAV particles comprising a viral genome encoding STXBP1 as described herein to neurons (e.g., GABAergic neurons and/or glutamatergic neurons) in the brain (e.g., the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus, oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, and/or vestibular nucleus) may lead to an increased expression of STXBP1 protein in one or more of those neurons. In some embodiments, the increased STXBP1 protein expression may lead to improved survival and/or function of various cell types in these CNS regions and/or improvement of at least one symptom of an STXBP1-related disorder, such as an STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5). [0296] In some embodiments, the AAV particles may be delivered to a subject in order to establish widespread distribution of STXBP1 throughout the CNS, e.g., by administering the AAV particles to the thalamus of the subject. In some embodiments, the increased expression of STXBP1 protein may lead to
Attorney Docket No.14640.0105-00304 a reduction in at least one symptom of an STXBP1-related disorder such as epilepsy, autistic features, ataxia, generalized tremors, reduced STXBP1 activity, accumulation of glucocerebroside and other glycolipids, e.g., within immune cells (e.g., macrophages), build-up of synuclein aggregates (e.g., Lewy bodies), developmental delay, progressive encephalopathy, progressive dementia, ataxia, myoclonus, oculomotor dysfunction, bulbar palsy, generalized weakness, trembling of a limb, depression, visual hallucinations, cognitive decline, dystonia, or a combination thereof. Administration [0297] In some embodiments, the present disclosure provides methods comprising administering viral vectors in accordance with the disclosure to a subject in need thereof. Viral vector pharmaceutical, diagnostic, or prophylactic compositions thereof, may be administered to a subject using any amount and any route of administration effective for treating, or diagnosing a disease, disorder, and/or condition associated with decreased STXBP1 expression or STXBP1 deficiency. In some embodiments, the disease, disorder, and/or condition is an STXBP1-related disorder, such as an STXBP1-related neurodegenerative or neuromuscular disorder (e.g., STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5)). [0298] Compositions in accordance with the disclosure may be formulated in unit dosage form for ease of administration and uniformity of dosage. It will be understood, however, that the total daily usage of the compositions of the present disclosure may be decided by the attending physician within the scope of sound medical judgment. The specific therapeutically effective, prophylactically effective, or appropriate imaging dose level for any particular patient will depend upon a variety of factors including the disorder being treated and the severity of the disorder; the activity of the specific compound employed; the specific composition employed; the age, body weight, general health, sex, and diet of the patient; the time of administration, route of administration, and rate of excretion of the specific protein employed; the duration of the treatment; drugs used in combination or coincidental with the specific compound employed; and like factors well known in the medical arts. [0299] In certain embodiments, the desired dosage may be delivered using multiple administrations (e.g., two, three, four, five, six, seven, eight, nine, ten, eleven, twelve, thirteen, fourteen, or more administrations). When multiple administrations are employed, split dosing regimens such as those described herein may be used. As used herein, a “split dose” is the division of single unit dose or total daily dose into two or more doses, e.g., two or more administrations of the single unit dose. As used herein, a “single unit dose” is a dose of any therapeutic composition administered in one dose/at one time/single route/single point of contact, e.g., single administration event. In some embodiments, a single unit dose is provided as a discrete dosage form (e.g., a tablet, capsule, patch, loaded syringe, vial, etc.). As used herein, a “total daily dose” is an amount given or prescribed in 24-hour period. It may be
Attorney Docket No.14640.0105-00304 administered as a single unit dose. The viral particles may be formulated in buffer only or in a formulation described herein. [0300] In some embodiments, a pharmaceutical composition described herein can be formulated into a topical, intranasal, pulmonary, intratracheal, or injectable dosage form. In some embodiments, a pharmaceutical composition described herein can be formulated in a dosage form suitable for intravenous, intraocular, intravitreal, intramuscular, intracardiac, intraperitoneal, and/or subcutaneous administration. [0301] In some embodiments, an AAV particle described herein is administered via intravenous administration. In some embodiments, an AAV particle described herein is formulated for intravenous administration. [0302] In some embodiments, delivery of the AAV particles described herein results in minimal serious adverse events (SAEs) as a result of the delivery of the AAV particles. Combinations [0303] The AAV particles may be used in combination with one or more other therapeutic, prophylactic, diagnostic, or imaging agents. The phrase “in combination with,” is not intended to require that the agents must be administered at the same time and/or formulated for delivery together, although these methods of delivery are within the scope of the present disclosure. Compositions can be administered concurrently with, prior to, or subsequent to, one or more other desired therapeutics or medical procedures. In general, each agent will be administered at a dose and/or on a time schedule determined for that agent. In some embodiments, the present disclosure encompasses the delivery of pharmaceutical, prophylactic, diagnostic, or imaging compositions in combination with agents that may improve their bioavailability, reduce and/or modify their metabolism, and/or modify their distribution within the body. [0304] The therapeutic agents may be approved by the US Food and Drug Administration or may be in clinical trial or at the preclinical research stage. The therapeutic agents may utilize any therapeutic modality known in the art, with non-limiting examples including gene silencing or interference (e.g., miRNA, siRNA, RNAi, shRNA), gene editing (e.g., TALEN, CRISPR/Cas9 systems, zinc finger nucleases), and gene, protein, or enzyme replacement. [0305] In some embodiments, an AAV particle described herein, or a pharmaceutical composition comprising the AAV particle, may be administered in combination with at least one additional therapeutic agent and/or therapy. In some embodiments, the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy for treating an STXBP1-related disorder such as an STXBP1-related neurodegenerative or neuromuscular disorder (e.g., STXBP1 encephalopathy, epileptic encephalopathy, Ohtahara syndrome, developmental encephalopathy, West syndrome, early myoclonic epileptic encephalopathy, Lennox-Gaustaut syndrome, autism (e.g., autism with STXBP1 mutations and optionally further mutations), Dravet syndrome (not caused by mutations in SCN1A), or Rett syndrome phenotype (not caused by mutation of MECP2 or CDKL5)).
Attorney Docket No.14640.0105-00304 [0306] In some embodiments, the at least one additional therapeutic agent and/or therapy comprises one or more anti-epileptic drugs (e.g., bromide, clobazam, felbamate, ganaxolone, lamotrigine, levetiracetam, phenobarbital, topiramate, valproate, or a combination thereof). [0307] In some embodiments, the at least one additional therapeutic agent and/or therapy comprises an immunosuppressant. In some embodiments, the immunosuppressant may be administered to the subject prior to administration of an AAV particle or pharmaceutical composition described herein. In some embodiments, the immunosuppressant may be administered to the subject simultaneously with administration of an AAV particle or pharmaceutical composition described herein. In some embodiments, the immunosuppressant may be administered to the subject after administration of an AAV particle or pharmaceutical composition described herein. In some embodiments, the AAV particle or pharmaceutical composition is administered to a subject who is receiving or has received an immunosuppressant. In some embodiments, the immunosuppressant comprises a corticosteroid (for example, and without limitation, prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine. In some embodiments, the corticosteroid comprises prednisone, prednisolone, methylprednisolone, and/or dexamethasone. In some embodiments, the immunosuppressant comprises adrenocorticotropic hormone. Measurement of Expression [0308] Expression of STXBP1 from viral genomes may be determined using various methods known in the art such as, but not limited to immunochemistry (e.g., IHC), enzyme-linked immunosorbent assay (ELISA), affinity ELISA, ELISPOT, flow cytometry, immunocytology, surface plasmon resonance analysis, kinetic exclusion assay, liquid chromatography-mass spectrometry (LCMS), high-performance liquid chromatography (HPLC), BCA assay, immunoelectrophoresis, Western blot, SDS-PAGE, protein immunoprecipitation, PCR, and/or in situ hybridization (ISH). In some embodiments, transgenes encoding STXBP1 delivered in different AAV capsid variants may have different expression levels in dorsal root ganglion (DRG). [0309] In certain embodiments, the STXBP1 protein is detectable by an enzyme-linked immunosorbent assay (ELISA). [0310] In certain embodiments, the STXBP1 protein is detectable by an immunohistochemistry assay. [0311] In certain embodiments, the STXBP1 protein is detectable by Western blot. [0312] In certain embodiments, expression of an STXBP1 gene, mRNA, and/or protein is measured in a cell or tissue of a subject who is receiving or has received an AAV particle described herein. In certain embodiments, the STXBP1 gene, mRNA, and/or protein expression is measured in a cell or tissue of the CNS and/or PNS comprises the amygdala, brainstem, caudate, central grey, cerebellum (e.g., Purkinje cell layer and/or deep cerebellar nuclei), cortex (e.g., frontal cortex, motor cortex, perirhinal cortex, sensory cortex, and/or temporal cortex), external cuneate nucleus, geniculate nucleus, globus pallidus, gracile nucleus, hippocampus, inferior colliculus, inferior olivary complex, nucleus ambiguus,
Attorney Docket No.14640.0105-00304 oculomotor nucleus, putamen, substantia nigra, thalamus, ventral palladium, vestibular nucleus, and/or spinal cord (e.g., cervical spinal cord region, lumbar spinal cord region, and/or thoracic spinal cord region), and/or a neuron (e.g., a GABAergic neuron and/or a glutamatergic neuron). In certain embodiments, the STXBP1 gene, mRNA, and/or protein expression is measured in a peripheral cell or tissue, such as the liver, heart, kidney, pancreas, and/or muscle. In some embodiments, the STXBP1 gene, mRNA, and/or protein expression is measured in the retina. VII. Kits and Devices Kits [0313] In some aspects, the present disclosure provides a variety of kits for conveniently and/or effectively carrying out methods of the present disclosure. Typically, kits will comprise sufficient amounts and/or numbers of components to allow a user to perform multiple treatments of a subject(s) and/or to perform multiple experiments. [0314] Any of the vectors, constructs, or STXBP1 sequences (polypeptides or nucleotides) of the present disclosure may be comprised in a kit. In some embodiments, kits may further include reagents and/or instructions for creating and/or synthesizing compounds and/or compositions of the present disclosure. In some embodiments, kits may also include one or more buffers. In some embodiments, kits of the disclosure may include components for making protein or nucleic acid arrays or libraries and thus, may include, for example, solid supports. [0315] In some embodiments, kit components may be packaged either in aqueous media or in lyophilized form. The container means of the kits will generally include at least one vial, test tube, flask, bottle, syringe or other container means, into which a component may be placed, and suitably aliquoted. Where there is more than one kit component, (labeling reagent and label may be packaged together), kits may also generally contain second, third or other additional containers into which additional components may be separately placed. In some embodiments, kits may also comprise second container means for containing sterile, pharmaceutically acceptable buffers and/or other diluents. In some embodiments, various combinations of components may be comprised in one or more vial. Kits of the present disclosure may also typically include means for containing compounds and/or compositions of the present disclosure, e.g., proteins, nucleic acids, and any other reagent containers in close confinement for commercial sale. Such containers may include injection or blow-molded plastic containers into which desired vials are retained. [0316] In some embodiments, kit components are provided in one and/or more liquid solutions. In some embodiments, liquid solutions are aqueous solutions, with sterile aqueous solutions being particularly used. In some embodiments, kit components may be provided as dried powder(s). When reagents and/or components are provided as dry powders, such powders may be reconstituted by the addition of suitable volumes of solvent. In some embodiments, it is envisioned that solvents may also be provided in another container means. In some embodiments, labeling dyes are provided as dried powders.
Attorney Docket No.14640.0105-00304 In some embodiments, it is contemplated that 10, 20, 30, 40, 50, 60, 70, 80, 90, 100, 120, 120, 130, 140, 150, 160, 170, 180, 190, 200, 300, 400, 500, 600, 700, 800, 900, 1000 micrograms or at least or at most those amounts of dried dye are provided in kits of the disclosure. In such embodiments, dye may then be resuspended in any suitable solvent, such as DMSO. [0317] In some embodiments, kits may include instructions for employing kit components as well the use of any other reagent not included in the kit. Instructions may include variations that may be implemented. Devices [0318] In some embodiments, compounds and/or compositions of the present disclosure may be combined with, coated onto or embedded in a device. Devices may include, but are not limited to, dental implants, stents, bone replacements, artificial joints, valves, pacemakers and/or other implantable therapeutic device. [0319] The present disclosure provides for devices which may incorporate viral vectors that encode one or more STXBP1 molecules. These devices contain in a stable formulation the viral vectors which may be immediately delivered to a subject in need thereof, such as a human patient. [0320] Devices for administration may be employed to deliver the viral vectors encoding STXBP1 of the present disclosure according to single, multi- or split-dosing regimens taught herein. [0321] Method and devices known in the art for multi-administration to cells, organs and tissues are contemplated for use in conjunction with the methods and compositions disclosed herein as embodiments of the present disclosure. VIII. Definitions [0322] At various places in the present specification, substituents of compounds of the present disclosure are disclosed in groups or in ranges. It is specifically intended that the present disclosure include each and every individual sub-combination of the members of such groups and ranges. The following is a non-limiting list of term definitions. [0323] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which the invention pertains. [0324] The articles “a,” “an,” and “the” may mean one or more than one unless indicated to the contrary or otherwise evident from the context. Claims or descriptions that include “or” between one or more members of a group are considered satisfied if one, more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process unless indicated to the contrary or otherwise evident from the context. The disclosure includes embodiments in which exactly one member of the group is present in, employed in, or otherwise relevant to a given product or process. The disclosure includes embodiments in which more than one, or the entire group members are present in, employed in, or otherwise relevant to a given product or process. [0325] The term “comprising” is intended to be open and permits but does not require the inclusion of additional elements or steps.
Attorney Docket No.14640.0105-00304 [0326] Where ranges are given, endpoints are included. Furthermore, it is to be understood that unless otherwise indicated or otherwise evident from the context and understanding of one of ordinary skill in the art, values that are expressed as ranges can assume any specific value or subrange within the stated ranges in different embodiments of the disclosure, to the tenth of the unit of the lower limit of the range, unless the context clearly dictates otherwise. [0327] Adeno-associated virus: As used herein, the term “adeno-associated virus” or “AAV” refers to members of the dependovirus genus or a functional variant thereof. Unless stated otherwise, “AAV” may refer to wildtype (i.e., naturally occurring) AAV or recombinant AAV. [0328] AAV Particle: As used herein, an “AAV particle” refers to a particle comprising an AAV capsid, e.g., an AAV capsid variant (such as a parent capsid sequence with at least one peptide insertion and/or with at least one substitution), and a polynucleotide, e.g., a viral genome or a vector genome. The AAV particle may be capable of delivering an STXBP1 polynucleotide to cells. The cells may be mammalian cells, e.g., human cells. In some embodiments, an AAV particle of the present disclosure may be produced recombinantly. In some embodiments, an AAV particle may be derived from any serotype, described herein or known in the art, including combinations of serotypes (e.g., “pseudotyped” AAV) or from various genomes (e.g., single stranded or self-complementary). In some embodiments, the AAV particle may be replication defective and/or targeted. In some embodiments, the AAV particle may comprise a peptide present in, e.g., inserted into and/or replacing a wildtype amino acid of, the capsid to enhance tropism for a desired target tissue. It is to be understood that reference to the AAV particle of the disclosure also includes pharmaceutical compositions thereof, even if not explicitly recited. [0329] Administering: As used herein, the term “administering” refers to providing a pharmaceutical agent or composition to a subject. [0330] Amelioration: As used herein, the term “amelioration” or “ameliorating” refers to a lessening of severity of at least one indicator of a condition or disease. For example, in the context of a neurodegenerative disorder, amelioration includes the reduction or stabilization of neuron loss. [0331] Approximately: As used herein, the term “approximately” or “about,” as applied to one or more values of interest, refers to a value that is similar to, i.e., within 10% of, a stated reference value. [0332] Baseline: The term “baseline,” when used to describe a measurement in a subject receiving or about to receive a treatment, refers to a measurement made before starting the treatment. [0333] Capsid: As used herein, the term “capsid” refers to the exterior, e.g., a protein shell, of a virus particle, e.g., an AAV particle, that is substantially (e.g., >50%, >60%, >70%, >80%, >90%, >95%, >99%, or 100%) protein. In some embodiments, the capsid is an AAV capsid comprising an AAV capsid protein described herein, e.g., a VP1, VP2, and/or VP3 polypeptide. The AAV capsid protein can be a wild-type AAV capsid protein or a variant, e.g., a structural and/or functional variant from a wild-type or a reference capsid protein, referred to herein as an “AAV capsid variant.” For example, and without limitation, an AAV capsid variant may refer to at least a VP1 protein, a VP2 protein, or a VP3 protein (e.g., all of the VP1, VP2, and VP3 proteins forming the AAV capsid) as will be clear from context. In
Attorney Docket No.14640.0105-00304 some embodiments, the AAV capsid variant described herein may comprise a peptide and/or amino acid insertion and/or substitution. The terms “substitution” and “replacement” are used interchangeably in this context. In some embodiments, the AAV capsid variant described herein has the ability to encapsulate a viral genome and/or is capable of entry into a cell, e.g., a mammalian cell. In some embodiments, the AAV capsid variant described herein may have modified tropism compared to that of a wild-type AAV capsid, e.g., the corresponding wild-type capsid. [0334] CNS Cells: As used herein, “CNS cells” refers to cells of the central nervous system and sub- structures thereof. Non-limiting examples of CNS cells include neurons and sub-types thereof, glia, microglia, oligodendrocytes, ependymal cells, and astrocytes. Non-limiting examples of neurons include sensory neurons, motor neurons, interneurons, unipolar cells, bipolar cells, multipolar cells, pseudounipolar cells, pyramidal cells, basket cells, stellate cells, Purkinje cells, Betz cells, amacrine cells, granule cell, ovoid cell, medium aspiny neurons and large aspiny neurons, GABAergic neurons and/or glutamatergic neurons. [0335] Codon optimization: As used herein, the term “codon optimization” refers to a process of changing codons of a given gene in such a manner that the polypeptide sequence encoded by the gene remains the same. [0336] Complementary and substantially complementary: As used herein, the term “complementary” refers to the ability of polynucleotides to form base pairs with one another. Perfect complementarity or 100% complementarity refers to the situation in which each nucleotide unit of one polynucleotide strand can form a hydrogen bond with a nucleotide unit of a second polynucleotide strand. Less than perfect complementarity refers to the situation in which some, but not all, nucleotide units of two strands can form hydrogen bond with each other. For example, for two 20-mers, if only two base pairs on each strand can form a hydrogen bond with each other, the polynucleotide strands exhibit 10% complementarity. In the same example, if 18 base pairs on each strand can form hydrogen bonds with each other, the polynucleotide strands exhibit 90% complementarity. The term “complementary” as used herein can encompass fully complementary or partially (e.g., substantially) complementary. “Fully complementary”, “perfect complementarity”, or “100% complementarity” refers to the situation in which each nucleotide unit of one polynucleotide or oligonucleotide strand can base-pair with a nucleotide unit of a second polynucleotide or oligonucleotide strand. As used herein, the term “substantially complementary” means that >50% of the nucleotide units of a first polynucleotide strand can base pair with nucleotide units on a second polynucleotide strand. When used in the context of RNA silencing, “substantially complementary” refers to an siRNA that has a sequence (e.g., in the antisense strand) that is sufficient to bind the desired target mRNA and to trigger the RNA silencing of the target mRNA. [0337] Conservative substitution: As used herein, a conservative substitution, as applied to an amino acid sequence, also referred to as a “conservative amino acid substitution,” is one in which the amino acid residue is replaced with an amino acid residue having similar biochemical properties. When used in reference to a nucleic acid sequence, the term “conservative substitution” refers to a nucleotide
Attorney Docket No.14640.0105-00304 replacement that results in an amino acid residue having similar biochemical properties compared to a reference sequence. Families of amino acid residues having similar biochemical properties have been defined in the art. These families include amino acids with basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine), nonpolar side chains (e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine, tryptophan), beta-branched side chains (e.g., threonine, valine, isoleucine) and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan, histidine). [0338] Corresponding to: As used herein, the phrase “corresponding to” in the context of an amino acid sequence refers to the location of an amino acid in a reference sequence or the equivalent position in a modified sequence when aligned. For example, an amino acid corresponding to position 585 of the amino acid sequence of SEQ ID NO: 138 refers to the amino acid at the 585th position from the N- terminus in the amino acid sequence of SEQ ID NO: 138 or the equivalent position in an aligned sequence. As used herein, an amino acid at a position corresponding to that in a designated sequence may also be referred to as an amino acid at a particular position, numbered according or numbered relative to the designated sequence. For instance, an amino acid corresponding to position 585 of the amino acid sequence of SEQ ID NO: 138 may also be referred to as an amino acid at position 585 (or amino acid 585), numbered according to the amino acid sequence of SEQ ID NO: 138 or as numbered according to a sequence corresponding to the amino acid sequence of SEQ ID NO: 138 (i.e., relative to a reference sequence of the amino acid sequence of SEQ ID NO: 138). [0339] Derivative: As used herein, the term “derivative” refers to a composition (e.g., sequence, compound, formulation, etc.) that is derived from, or finds its basis in, a parent composition. Non- limiting examples of a parent composition include a wild-type or original amino acid or nucleic acid sequence, or an undiluted formulation. In some embodiments, a derivative is a variant of a parent composition. A derivative may differ from the parent composition by less than about 1%, less than about 5%, less than about 10%, less than about 15%, less than about 20%, less than about 25%, less than about 30%, less than about 35%, less than about 40%, less than about 45%, or less than about 50%. In certain embodiments, a derivative may differ from a parent composition by more than about 50%. In certain embodiments, a derivative may differ from a parent composition by more than about 75%. In some embodiments, a derivative may be a fragment or truncation of a parent amino acid or nucleotide sequence. As a non-limiting example, a derivative may be a sequence with a nucleotide, amino acid, or peptide substitution and/or insertion as compared to a parent nucleic acid or amino acid sequence (e.g., as compared to AAV9). [0340] Effective amount: As used herein, the term “effective amount” or “therapeutically effective amount” of an agent is that amount sufficient to effect beneficial or desired results. An effective amount is provided in a single dose or multiple doses to treat, improve symptoms of, delay progression of symptoms, diagnose, prevent, and/or delay the onset of the infection, disease, disorder, and/or condition.
Attorney Docket No.14640.0105-00304 [0341] Fragment: A “fragment,” as used herein, refers to a contiguous portion of a reference sequence. A fragment may comprise a functional fragment that retains at least one activity of the reference sequence. For example, fragments of proteins may comprise polypeptides obtained by digesting full-length protein isolated from cultured cells. A fragment may also refer to a truncation (e.g., an N- terminal and/or C-terminal truncation) of a protein or a truncation (e.g., at the 5’ and/or 3’ end) of a nucleic acid. A protein fragment may be obtained by expression of a truncated nucleic acid, such that the nucleic acid encodes a portion of the full-length protein. [0342] Healthy individual: As used herein, the term “healthy individual” refers to an individual who does not have a disease or disorder associated with STXBP1 protein deficiency, e.g., an individual who does not have an STXBP1-related disorder. [0343] Identity: As used herein, the term “identity” refers to the overall relatedness between polymeric molecules, e.g., between oligonucleotide molecules (e.g., DNA molecules and/or RNA molecules) and/or between polypeptide molecules. Calculation of the percent identity of two polynucleotide sequences, for example, may be performed by aligning the two sequences for optimal comparison purposes (e.g., gaps can be introduced in one or both of a first and a second nucleic acid sequences for optimal alignment and non-identical sequences can be disregarded for comparison purposes). The nucleotides at corresponding nucleotide positions are then compared. When a position in the first sequence is occupied by the same nucleotide as the corresponding position in the second sequence, then the molecules are identical at that position. The percent identity between the two sequences is a function of the number of identical positions shared by the sequences, taking into account the number of gaps, and the length of each gap, which needs to be introduced for optimal alignment of the two sequences. The comparison of sequences and determination of percent identity between two sequences can be accomplished using a mathematical algorithm. For example, the percent identity between two nucleotide sequences can be determined using methods such as those described in Computational Molecular Biology, Lesk, A. M., ed., Oxford University Press, New York, 1988; Biocomputing: Informatics and Genome Projects, Smith, D. W., ed., Academic Press, New York, 1993; Sequence Analysis in Molecular Biology, von Heinje, G., Academic Press, 1987; Computer Analysis of Sequence Data, Part I, Griffin, A. M., and Griffin, H. G., eds., Humana Press, New Jersey, 1994; and Sequence Analysis Primer, Gribskov, M. and Devereux, J., eds., M Stockton Press, New York, 1991; each of which is incorporated herein by reference in its entirety. For example, the percent identity between two nucleotide sequences can be determined using the algorithm of Myers and Miller (CABIOS, 1989, 4:11-17), which has been incorporated into the ALIGN program (version 2.0) using a PAM120 weight residue table, a gap length penalty of 12 and a gap penalty of 4. The percent identity between two nucleotide sequences can, alternatively, be determined using the GAP program in the GCG software package using an NWSgapdna.CMP matrix. Methods commonly employed to determine percent identity between sequences include, but are not limited to those disclosed in Carillo, H., and Lipman, D., SIAM J Applied Math., 48:1073 (1988); incorporated herein by reference in its entirety. Techniques for
Attorney Docket No.14640.0105-00304 determining identity are codified in publicly available computer programs. Computer software to determine homology between two sequences include, but are not limited to, GCG program package, Devereux, J., et al., Nucleic Acids Research, 12(1), 387 (1984)), the Basic Local Alignment Search Tool (BLAST, which includes, e.g., BLASTP for protein sequences and BLASTN for nucleic acid sequences), and FASTA Altschul, S. F. et al., J. Molecular Biol., 215, 403 (1990)), EMBOSS Needle, Clustal Omega, Benchling, and Geneious. In preferred embodiments, sequence identity may be determined using BLAST, Clustal Omega, or EMBOSS Needle. [0344] Inverted terminal repeat: As used herein, the term “inverted terminal repeat” or “ITR” refers to a cis-regulatory element for the packaging of polynucleotide sequences into viral capsids. [0345] Isolated: As used herein, the term “isolated” refers to a substance or entity that is altered or removed from the natural state, e.g., altered or removed from at least some of component with which it is associated in the natural state. For example, a nucleic acid or a peptide naturally present in a living animal is not “isolated,” but the same nucleic acid or peptide partially or completely separated from the coexisting materials of its natural state is “isolated.” An isolated nucleic acid or protein can exist in substantially purified form, or can exist in a non-native environment such as, for example, a host cell. Such polynucleotides could be part of a vector and/or such polynucleotides or polypeptides could be part of a composition, and still be isolated in that such vector or composition is not part of the environment in which it is found in nature. In some embodiments, an isolated nucleic acid is recombinant, e.g., incorporated into a vector. [0346] miRNA binding site: As used herein, a “miRNA binding site” or “miR binding site” refers to an RNA sequence that is bound by a microRNA, or to the RNA sequence that is bound by the microRNA. The miR binding site is capable of binding, or binds, in whole or in part to a microRNA (miRNA, miR) through complete or partial hybridization. A miR binding site may be encoded or transcribed in series, also referred to as a “miR binding site series” or “miR BSs”, which includes two or more miR binding sites having the same or a different nucleic acid sequence. [0347] Modification: As used herein, the term “modification” or “modified,” refers to any substance, compound, or molecule that has been changed in any way. For example, a modification in an amino acid sequence may comprise a substitution (e.g., a conservative substitution), an insertion, and/or a deletion of one or more amino acids in the sequence. [0348] Neurological disease: As used herein, a “neurological disease” is any disease associated with the central or peripheral nervous system and components thereof (e.g., neurons). [0349] Operably linked: As used herein, the phrase “operably linked” refers to a functional connection between two or more molecules, constructs, transcripts, entities, moieties or the like. [0350] Payload: As used herein, “payload,” “payload sequence,” or “payload region” refers to one or more polynucleotides or polynucleotide regions encoded by or within a viral genome or an expression product of such polynucleotide or polynucleotide region, e.g., a transgene, a polynucleotide encoding a polypeptide.
Attorney Docket No.14640.0105-00304 [0351] Pharmaceutical Composition: As used herein, the term “pharmaceutical composition” or pharmaceutically acceptable composition” comprises AAV polynucleotides, AAV genomes, or AAV particle and one or more pharmaceutically acceptable excipients, solvents, adjuvants, and/or the like. [0352] Position: The term “position,” as used herein in the context of an amino acid sequence, refers to the location of a particular amino acid or set of amino acids relative to a larger sequence. A position or positions of amino acids may interchangeably be referred to by an amino acid number or numbers of a reference sequence. For example, and unless otherwise specified, “positions 1-736 as numbered according to SEQ ID NO: 981” is interchangeable with “amino acids 1-736, as numbered according to SEQ ID NO: 981” and “one or more modifications at one or more of positions 580-599, as numbered according to SEQ ID NO: 138” is interchangeable with “one or more modifications at one or more of amino acids 580-599, as numbered according to SEQ ID NO: 138.” Within a sequence, an amino acid position is counted from the N-terminus. [0353] Preventing: As used herein, the term “preventing” refers to partially or completely delaying onset of an infection, disease, disorder and/or condition; partially or completely delaying onset of one or more symptoms, features, or clinical manifestations of a particular infection, disease, disorder, and/or condition; partially or completely delaying onset of one or more symptoms, features, or manifestations of a particular infection, disease, disorder, and/or condition; partially or completely delaying progression from an infection, a particular disease, disorder and/or condition; and/or decreasing the risk of developing pathology associated with the infection, the disease, disorder, and/or condition. The term “prevention” or “preventing” of an infection, disease, disorder and/or condition may be considered a subset within the meaning with the term “treatment” or “treating” of the infection, disease, disorder and/or condition. [0354] Region: As used herein, the term “region” refers to a zone or general area. In some embodiments, when referring to a protein or protein module, a region may comprise a linear sequence of amino acids along the protein or protein module or may comprise a three-dimensional area. In some embodiments, regions comprise terminal regions. As used herein, the term “terminal region” refers to regions located at the ends or termini of a given agent. When referring to proteins, terminal regions may comprise N- and/or C-termini. N-termini refer to the end of a protein comprising an amino acid with a free amino group. C-termini refer to the end of a protein comprising an amino acid with a free carboxyl group. N- and/or C-terminal regions may comprise the N- and/or C-termini as well as surrounding amino acids. When referring to a polynucleotide, a region may comprise a linear sequence of nucleic acids along the polynucleotide or may comprise a three-dimensional area, secondary structure, or tertiary structure. In some embodiments, regions comprise terminal regions. As used herein, the term “terminal region” refers to regions located at the ends or termini of a given agent. When referring to polynucleotides, terminal regions may comprise 5’ and/or 3’ termini. [0355] Sample: As used herein, the term “sample” or “biological sample” refers to a subset of tissues, cells, nucleic acids, or a component or part of the body (e.g., a body fluid, including but not
Attorney Docket No.14640.0105-00304 limited to blood, mucus, lymphatic fluid, synovial fluid, cerebrospinal fluid, saliva, amniotic fluid, amniotic cord blood, urine, vaginal fluid, and semen). [0356] Self-complementary AAV: As used herein, the term “self-complementary AAV” refers to an AAV comprising at least a protein capsid and a self-complementary viral genome. [0357] Serotype: As used herein, the term “serotype” refers to distinct variations in a capsid of an AAV based on surface antigens which allow epidemiologic classifications of the AAVs at the sub- species level. [0358] Silent Mutation: As used herein, a “silent mutation” or “silent substitution” refers to a nucleotide replacement that results in the same amino acid residue as a reference sequence. [0359] Signal Sequence: As used herein, the phrase “signal sequence” refers to a sequence which can direct the transport or localization. [0360] Similarity: As used herein, the term “similarity” refers to the overall relatedness between polymeric molecules, e.g., between polynucleotide molecules (e.g., DNA molecules and/or RNA molecules) and/or between polypeptide molecules. Calculation of percent similarity of polymeric molecules to one another can be performed in the same manner as a calculation of percent identity, except that calculation of percent similarity takes into account conservative substitutions as is understood in the art. [0361] Spacer: As used herein, a “spacer” is generally any selected nucleic acid sequence of, e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 nucleotides in length, which is located between two or more consecutive miR binding site sequences. In some embodiments, spacers may also be more than 10 nucleotides in length, e.g., 20, 30, 40, or 50 or more than 50 nucleotides. [0362] STXBP1-related disorder: As used herein, an “STXBP1-related disorder” refers to a disease, disorder, or condition in which one or more symptoms is caused by or associated with a deficiency of syntaxin-binding protein-1 (STXBP1) in a subject. [0363] Subject: As used herein, the term “subject” or “patient” refers to any organism to which a composition in accordance with the disclosure may be administered, e.g., for experimental, diagnostic, prophylactic, and/or therapeutic purposes. Similarly, “subject” or “patient” refers to an organism who may seek, who may require, who is receiving, or who will receive treatment or who is under care by a trained professional for a particular disease or condition. Typical subjects include animals (e.g., mammals such as mice, rats, rabbits, non-human primates, and humans). As used herein, a subject or patient may be susceptible to, suspected of having, or have a deficiency in STXBP1 protein, e.g., human STXBP1 protein, and, e.g., may be susceptible to, suspected of having, or have an STXBP1-related disorder. [0364] Substantially: As used herein, the term “substantially” refers to the qualitative condition of exhibiting total or near-total extent or degree of a characteristic or property of interest. One of ordinary skill in the biological arts will understand that biological and chemical phenomena rarely, if ever, go to completion and/or proceed to completeness or achieve or avoid an absolute result. The term
Attorney Docket No.14640.0105-00304 “substantially” is therefore used herein to capture the potential lack of completeness inherent in many biological and chemical phenomena. [0365] Suffering from: An individual who is “suffering from” a disease, disorder, and/or condition has been diagnosed with or displays one or more symptoms of a disease, disorder, and/or condition. [0366] Susceptible to: An individual who is “susceptible to” a disease, disorder, and/or condition has not been diagnosed with and/or may not exhibit symptoms of the disease, disorder, and/or condition but harbors a propensity to develop a disease or its symptoms. In some embodiments, an individual who is susceptible to a disease, disorder, and/or condition may be characterized by one or more of the following: (1) a genetic mutation associated with development of the disease, disorder, and/or condition; (2) a genetic polymorphism associated with development of the disease, disorder, and/or condition; (3) increased and/or decreased expression and/or activity of a protein and/or nucleic acid associated with the disease, disorder, and/or condition; (4) habits and/or lifestyles associated with development of the disease, disorder, and/or condition; (5) a family history of the disease, disorder, and/or condition; and (6) exposure to and/or infection with a microbe associated with development of the disease, disorder, and/or condition. In some embodiments, an individual who is susceptible to a disease, disorder, and/or condition will develop the disease, disorder, and/or condition. In some embodiments, an individual who is susceptible to a disease, disorder, and/or condition will not develop the disease, disorder, and/or condition. [0367] Target Cells: As used herein, “target cells” refers to any one or more cells of interest. The cells may be found in vitro, in vivo, in situ or in the tissue or organ of an organism. The organism may be an animal, preferably a mammal, more preferably a human and most preferably a human patient. [0368] Target Tissue: As used herein, “target tissue” refers to a tissue of interest that may be found in vitro, in situ, or as part of an animal, preferably a mammal, more preferably a human and most preferably a human patient. [0369] Therapeutic Agent: The term “therapeutic agent” refers to any agent that, when administered to a subject, elicits a desired biological and/or pharmacological effect. [0370] Therapeutically Effective Outcome: As used herein, the term “therapeutically effective outcome” means an outcome that is sufficient in a subject suffering from or susceptible to an infection, disease, disorder, and/or condition, to treat, improve symptoms of, delay progression of symptoms, diagnose, prevent, and/or delay the onset of the infection, disease, disorder, and/or condition. [0371] Treating: As used herein, the term “treating” refers to partially or completely alleviating, ameliorating, improving, relieving, delaying onset of, inhibiting progression of, reducing severity of, reducing incidence of, and/or preventing one or more symptoms or features of a particular infection, disease, disorder, and/or condition. Treatment may be administered to a subject who does not exhibit signs of a disease, disorder, and/or condition and/or to a subject who exhibits only early signs of a disease, disorder, and/or condition for the purpose of decreasing the risk of developing pathology associated with the disease, disorder, and/or condition.
Attorney Docket No.14640.0105-00304 [0372] Unmodified: As used herein, “unmodified” refers to any substance, compound or molecule prior to being changed in any way. Unmodified may, but does not always, refer to the wild-type or native form of a biomolecule or entity. Molecules or entities may undergo a series of modifications whereby each modified product may serve as the “unmodified” starting molecule or entity for a subsequent modification. [0373] Variant: The term “variant” refers to a polypeptide or polynucleotide that has an amino acid or a nucleotide sequence that has at least 90% (at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to a reference sequence. The variant may be a functional variant. As used herein, the term “functional variant” refers to a polypeptide variant or a polynucleotide variant that has at least one activity of the reference sequence. [0374] Vector: As used herein, a “vector” is any molecule or moiety which transports, transduces, or otherwise acts as a carrier of a heterologous molecule. Vectors of the present disclosure may be produced recombinantly and may be based on and/or may comprise adeno-associated virus (AAV) parent or reference sequence(s). [0375] Viral genome: As used herein, a “viral genome,” “vector genome,” or “VG” is a polynucleotide comprising at least one inverted terminal repeat (ITR) and at least one nucleic acid sequence encoding a payload. A viral genome encodes at least one copy of the payload. EXAMPLES [0376] The present disclosure is further illustrated by the following non-limiting examples. Example 1. High-throughput screen of TRACER AAV library in non-human primates (NHPs) [0377] A TRACER based method as described in WO2020072683, WO 2021/202651, and WO2021230987, the contents of which are herein incorporated by reference in their entirety, was used to generate the AAV capsid variants described herein. An orthogonal evolution approach was combined with a high throughput screening by NGS. Briefly, the library of AAV capsid variants was generated using a sliding window approach, where amino acid modifications, e.g., substitutions, were randomly introduced into various positions across loop VIII of AAV9, including between positions 581-593, relative to a reference sequence numbered according to SEQ ID NO: 138. The initial library (passage 1) was passed first through cynomolgus macaques (Macaca fascicularis, n=2, 2-4 years of age) or human brain microvascular endothelial cells (hBMVECs) followed by cynomolgus macaques (n=2). These first passage libraries were then pooled and screened a second time through cynomolgus macaques (passage 2). After the second passage (e.g., 28 days post injection into two NHPs), RNA was extracted from multiple brain and spinal cord regions. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate fold enrichment relative to an AAV9 wild-type control. The top variants were selected for the creation of a synthetic library. [0378] After creation of the synthetic library with the sub-selected variants, the synthetic library was screened (passage 3) in two NHPs (2-4 years of age). The animals were injected intravenously with the
Attorney Docket No.14640.0105-00304 synthetic library. After a period in vivo, (e.g., 28-days) RNA was extracted from nervous tissue, e.g., brain, spinal cord, and DRG of the NHPs. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed, and the peptides comprised within the variants were identified and the capsid enrichment ratio for each variant compared to the wild-type AAV9 control was calculated (fold enrichment relative to wild-type AAV9). Values above 1 indicate an increase in expression relative to AAV9. All animals were dosed intravenously at 2e13 VG/kg across the screen. [0379] Following these three passages, approximately 1542 variants were identified with an average fold change greater than wild-type AAV9 in the brain of the NHPs (Macaca fascicularis). Of the 1542 variants, 422 demonstrated a fold-change of greater than 5 and 118 demonstrated a fold-change greater than 10 compared to wild-type and were detected across all brain regions investigated. The capsid variant that demonstrated the greatest fold-change in expression relative to AAV9 in the brain of the NHP comprised the amino acid sequence TQDWHRI (SEQ ID NO: 941), resulting in a fold-change in expression of 460 relative to the AAV9 control (Table 9). Table 9. NGS fold-enrichment of AAV capsid variants in NHPs (Macaca fascicularis) Peptide Sequence SEQ ID Fold enrichment over AAV9 NO: Brain Spinal cord DRG

AV9 variant library with loop VIII modifications in NHPs (cynomolgus macaques (Macaca fascicularis)), many AAV capsid variants outperformed the wild-type AAV9, for example, in penetration of the blood brain barrier (BBB) and spinal cord expression. Example 2. Individual Capsid Characterization in NHPs [0381] The goal of these experiments was to determine the transduction level, tropism, ability to cross the blood brain barrier, and overall spatial distribution in the central nervous system (CNS) of a capsid variant selected from the study described in Example 1 relative to AAV9 following intravenous injection in NHPs (cynomolgus macaques (Macaca fascicularis)) and marmosets (Callithrix jacchus). The capsid variant was TTJ-001 (SEQ ID NO: 981 (amino acid) and 983 (DNA), comprising SEQ ID NO: 941 or 201), as outlined in Table 3 above. The amino acid and DNA sequences of TTJ-001 are provided, e.g., in Tables 4 and 5, respectively. A. Evaluation of TTJ-001 in Cynomolgus macaques [0382] AAV particles were generated with this capsid variant encapsulating a sequence encoding a cynomolgus frataxin protein tagged with hemagglutinin (cyno-FXN-HA) driven by a CBA promoter. The capsid variant and AAV9 control were tested by intravenously administering the AAV particle formulation at 2e13 VG/kg to NHPs (n=2). The in-life period was 14 days and the brain, spinal cord, and peripheral tissues including the heart, liver, and quadriceps were collected for measuring transgene mRNA, transgene protein, and viral DNA (biodistribution). The AAV particles administered to the NHPs comprised single stranded viral genomes.
Attorney Docket No.14640.0105-00304 [0383] The brains isolated from the NHPs injected with the AAV particles encapsulated in the TTJ- 001 capsid variant (AAV_TTJ-001) were assayed by ddPCR for the presence of transgene RNA as a measure of transgene expression, and the presence of viral DNA as a measure of viral genome levels. Data were provided as average mRNA fold change for the transgene relative to a housekeeping gene as well as the fold change relative to the AAV9 control (Table 10). As shown in Table 10, mRNA transgene expression from the TTJ-001 capsid variant, which is an AAV9 capsid variant, was significantly higher in the brain of NHPs relative to the wild-type AAV9 control. More specifically, mRNA expression was approximately 40-280-fold higher from the TTJ-001 capsid variant compared to wild-type AAV9 in the brain of NHPs. Additionally, mRNA expression was approximately 3-274-fold higher from the TTJ-001 capsid variants compared to wild-type AAV9 in the spinal cord of the NHPs. Comparable results were obtained when mRNA levels were measured by qPCR. Table 10. Transgene mRNA expression with the TTJ-001 capsid variant in NHPs Capsid Tissue AVG transgene mRNA fold Fold change relative to AAV9 Variant change relative to housekee in ene

Attorney Docket No.14640.0105-00304 [0384] The brains, spinal cord, and peripheral tissues isolated from the NHPs were also assayed for the presence of viral DNA as a measure of viral genome levels by ddPCR. Data are provided in Table 11 as average DNA (viral genome (VG)) copies per diploid genome as well as fold change relative to the AAV9 control. As shown in Table 11, biodistribution of the AAV9 capsid variant, TTJ-001, was significantly higher in the NHP brain relative to the wild-type AAV9 control. Biodistribution of TTJ-001 was lower in the NHP liver relative to the wild-type AAV9 control. Comparable results were obtained when average DNA levels were measured by qPCR. Table 11. Viral DNA biodistribution with the TTJ-001 capsid variant in NHPs Capsid Tissue AVG DNA copies per diploid Fold change relative to AAV9 Variant genome AAV9 Sensor Cortex 0199017 10

[0385] The brain tissues and spinal cords of the mice were also subjected to immunohistochemistry staining to evaluate overall CNS tropism and biodistribution in various regions. Immunohistochemical staining correlated with the qPCR analysis, as TTJ-001 showed significantly stronger staining and more
Attorney Docket No.14640.0105-00304 widespread payload expression in the brain and spinal cord, as compared to the AAV9 control. More specifically, TTJ-001 demonstrated moderately to strongly positive staining in the globus pallidus, thalamus, substantia nigra, oculomotor nucleus, geniculate nucleus, central grey, inferior colliculus, external cuneate nucleus, gracile nucleus, nucleus ambiguus, ventral horn of the spinal cord, vestibular nucleus, inferior olivary complex, deep cerebellar nuclei, and the DRG. TTJ-001 demonstrated weakly positive to moderately positive staining in the ventral pallidum, amygdala, caudate, putamen, hippocampus, temporal cortex, auditory cortex, and somatosensory cortex. [0386] The peripheral tissues were also subjected to immunohistochemistry staining to evaluate tropism and biodistribution. TTJ-001 resulted in widespread transduction of multiple peripheral tissues, as compared to the AAV9 control. TTJ-001 demonstrated weakly positive to moderately positive staining in the pancreas and quadriceps and moderately to strongly positive staining in the heart, kidney, and liver. [0387] Taken together, these data demonstrate that TTJ-001 is an enhanced CNS-tropic capsid variant in NHPs. Additionally, this capsid variant was able to successfully penetrate the blood brain barrier following intravenous injection. B. Evaluation of TTJ-001 in Marmosets (Callithrix jacchus) [0388] AAV particles were generated with the TTJ-001 capsid variant or the AAV9 capsid control which comprised a self-complementary viral genome encoding a histone H2b protein with a VSV tag (TTJ-001 capsid variant) or HA tag (AAV9 control capsid) driven by a ubiquitous CAG promoter. The AAV particles comprising the TTJ-001 capsid variant or the AAV9 capsid control were administered to the marmosets (Callithrix jacchus) (n=3) intravenously in a single solution, at the doses indicated in Table 12. The in-life period was 28 days and then various CNS and peripheral tissues were collected for measuring transgene mRNA (expression) by RT-qPCR, protein expression by IHC, and viral DNA (biodistribution) by ddPCR. Data were then normalized to the dose of each viral vector in the dosing solution. Table 12. Titer of the AAV particles comprising the TTJ-001 or AAV9 capsid in the solution dosed in marmosets Capsid Variant Actual Titer Dosed Ratio of Capsid Variant to AAV9 TTJ-001 2.23 x 10
11 vg/mL 0.56

, n the caudate and motor cortex in the brain of the marmosets relative to the AAV9 control. The TTJ-001 capsid variant also led to increased transgene expression (Table 14) in the caudate and motor cortex in the brain of the marmosets. In fact, biodistribution and transgene expression were increased over 5-7 fold and 15-17 fold, respectively, for TTJ-001 in the brain of marmosets relative to AAV9. Similar expression and biodistribution was observed by immunohistochemistry. More specifically, staining for TTJ-001 was detected in the mid-brain, caudate, putamen, thalamus, and cerebellum, and this staining was increased
Attorney Docket No.14640.0105-00304 relative to AAV9. Staining for TTJ-001 was also observed in the molecular and granule layer of the cerebellum. [0390] Biodistribution and transgene expression were also measured in the peripheral tissues of the liver, heart, and quadriceps. In the liver, the TTJ-001 capsid variant exhibited lower biodistribution (Table 13) and transgene expression (Table 14) relative to AAV9, indicating that the TTJ-001 capsid variant was detargeted in the liver relative to AAV9 in marmosets. Additionally, the TTJ-001 capsid variant resulted in increased transduction of the heart and muscle, as compared to the AAV9 control (Table 13 and Table 14). Table 13. Quantification of viral genome copies per diploid genome (biodistribution) by ddPCR following intravenous administration of AAV particles comprising the TTJ-001 capsid variant normalized to the actual titer of the viral vector in the dosing solution (vg/dg = viral genome copies/ diploid genome) Tissue Caudate Motor Cortex Heart Muscle Liver d d d d dg e V9

f AAV particles comprising the TTJ-001 capsid variant normalized to the actual titer of the viral vector in the dosing solution (mRNA = transgene mRNA fold over housekeeping gene; rel. to AAV9= transgene mRNA fold over housekeeping gene relative to AAV9) Tissue Capsid Caudate Motor Cortex Heart Muscle Liver o 9

macaques, demonstrating cross-species compatibility of the TTJ-001 capsid variant. [0392] Taken together, these data demonstrate that TTJ-001 is an enhanced CNS tropic capsid in marmosets. Additionally, the TTJ-001 capsid variant was able to successfully penetrate the blood brain barrier following intravenous injection in the marmosets. C. Further Evaluation of TTJ-001 in Cynomolgus Macaques (Macaca fascicularis) [0393] AAV particles were generated with the TTJ-001 capsid variant comprising a single-stranded viral genome encapsulating codon-optimized nucleotide sequence encoding GBA1, which is tagged with hemagglutinin (GBA1-HA). The GBA1-HA payload is driven by a CBA promoter. The TTJ-001 capsid variant and AAV9 control were tested by intravenously administering the AAV particle formulation at 1e13 vg/kg (n=3) or 3e12 vg/kg (n=3) to the NHPs. The in-life period was 28 days and the brain, spinal cord, and peripheral tissues including the heart, liver, and quadriceps were collected for measuring transgene mRNA, transgene protein, and viral DNA (biodistribution).
Attorney Docket No.14640.0105-00304 [0394] As shown in Tables 15 and 16, the TTJ-001 capsid variant demonstrated increased biodistribution and transduction as well as transgene expression in the brain (putamen, motor cortex, frontal cortex, substantia nigra, and dentate nucleus) and spinal cord (cervical C3 ventral horn) relative to AAV9. When dosed at 1e13 vg/kg, TTJ-001 led to at least a 6-9-fold increase in biodistribution in the brain and at least an 8-fold increase in biodistribution in the spinal cord relative to AAV9. With respect to transgene expression, when dosed at 1e13 kg/vg, TTJ-001 demonstrated at least a 25-61-fold increase in expression in the brain and at least a 20-fold increase in the spinal cord relative to AAV9. TTJ-001 also demonstrated comparable biodistribution and transgene expression in the DRG relative to AAV9 (Tables 15, 16A, and 16B). [0395] By immunohistochemistry (IHC), when dosed at 1e13 vg/kg, TTJ-001 demonstrated largely neuronal tropism, as evidenced by positive co-staining for HA/SMI 331 (neuronal marker) at least in the putamen and substantia nigra. No staining was observed in astrocytes in the putamen and substantia nigra. Additionally, by IHC, increased staining and transduction by AAV particles comprising the TTJ- 001 capsid variant was also observed in multiple brain regions (substantia nigra, putamen, motor cortex, frontal cortex) and the spinal cord regions (cervical region, thoracic region, Clarke’s column) of the NHPs, relative to AAV9, when administered at a dose of 1e13 vg/kg. Staining and therefore transduction was also observed in the brain and spinal cord regions even when the AAV particles comprising the TTJ- 001 capsid variant were administered at the lower dose of 3e12 vg/kg. Comparable transduction and expression were observed by IHC in the DRG following administration of AAV particles comprising the TTJ-001 capsid variant and AAV particles comprising the AAV9 control capsid. IHC staining of the payload-HA positive cells of various brain regions following transduction with the AAV particles comprising the TTJ-001 capsid variant or AAV particles comprising an AAV9 control capsid was also quantified (Table 17). At a dose of 1e13 vg/kg, TTJ-001 was capable of transducing greater than 2-9% of cells across the various brain regions investigated (Table 17). Even at the low dose of 3e12 vg/kg, TTJ- 001 was capable of transducing greater than 0.9 to 5% cells in the brain regions tested (Table 17). The percent HA+ cells in Table 17 may underestimate true positivity for the TTJ-001 capsid variant, as it demonstrated neuronal tropism. [0396] Biodistribution and transgene expression were also measured in the peripheral tissues of the liver and heart. In the liver, at a dose of 1e13 vg/kg, the TTJ-001 capsid variant exhibited lower biodistribution (Table 15) and transgene expression (Tables 16A and 16B) relative to AAV9, indicating that the TTJ-001 capsid variant was detargeted in the liver relative to AAV9. At the 1e13 vg/kg dose, the TTJ-001 demonstrated comparable biodistribution but decreased transgene expression in the heart relative to AAV9 (Tables 15, 16A, and 16B). Similar transduction and expression were observed in the heart by IHC following transduction with AAV particles comprising the TTJ-001 capsid variant and AAV particles comprising the AAV9 capsid control at a dose of 1e13 vg/kg.
Attorney Docket No.14640.0105-00304 Table 15. Quantification of viral genome copies per diploid genome (biodistribution) by ddPCR following intravenous administration of AAV particles comprising the TTJ-001 capsid variant (vg/dg = viral genome copies/ diploid genome) Tissues Putamen Motor Cortex Frontal Cortex Dose vg/dg relative vg/dg relative vg/dg relative e e tration
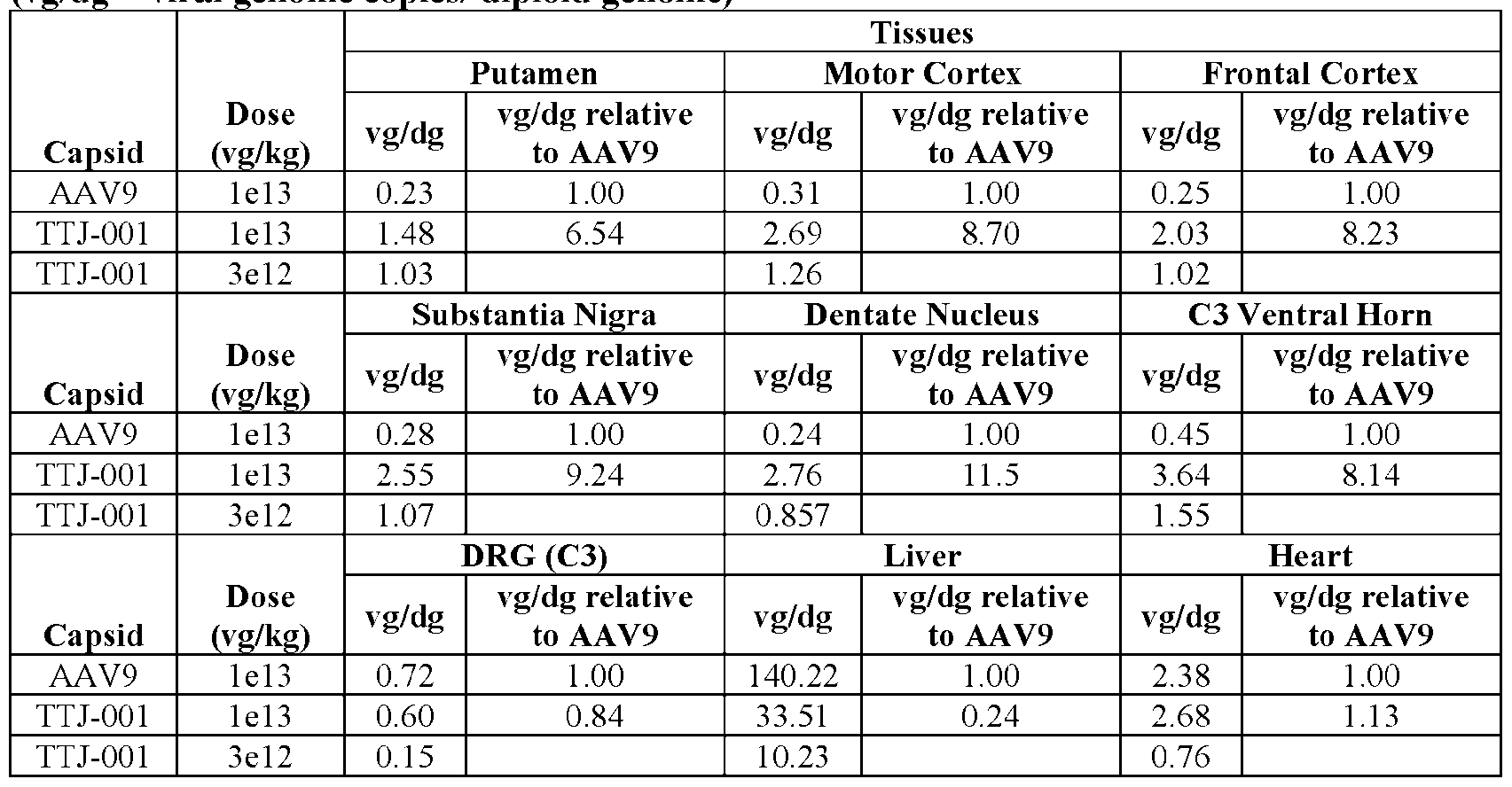
o AAV part c es compr s ng t e TTJ-001 caps d var ant (mRNA = transgene mRNA o d over housekeeping gene; rel. to AAV9= transgene mRNA fold over housekeeping gene relative to AAV9) Tissues Frontal Cortex Motor Cortex Putamen
ration of AAV particles comprising the TTJ-001 capsid variant (mRNA = transgene mRNA fold over endogenous GBA mRNA; rel. to AAV9= transgene mRNA fold change over endogenous GBA mRNA relative to fold change over AAV9) Tissues Frontal Cortex Motor Cortex Putamen
Attorney Docket No.14640.0105-00304 Dose mRNA rel. to mRNA rel. to mR rel. to (vg/kg) AAV9 AAV9 NA AAV9 AAV9 1e13 0.10 0.58 14.51
Frontal Cortex Motor Cortex Putamen Substantia Nigra AAV9 1e13 05 01 06 13 blood

brain barrier following intravenous administration in NHPs. D. Further Evaluation of TTJ-001 in Cynomolgus Macaques (Macaca fascicularis) [0398] AAV particles were generated with the TTJ-001 capsid variant or the AAV9 capsid control, each of which comprised a self-complementary viral genome encoding a histone H2b protein driven by a ubiquitous CAG promoter. The AAV particles comprising the TTJ-001 capsid control were administered to a first group of male cynomolgus macaques (Macaca fascicularis; 4-6 kg body weight; over 2 years old) intravenously at a dose per capsid of 4e12 VG/kg. The AAV particles comprising the AAV9 capsid control were administered to a second group of male cynomolgus macaques (Macaca fascicularis; 4-6 kg body weight; over 2 years old) intravenously at a dose per capsid of 4e12 VG/kg. The in-life period was 28 days for both groups, and then various CNS and peripheral tissues were collected for measuring transgene mRNA (expression) by RT-qPCR; protein expression by IHC/chromogenic staining (e.g., DAB staining for percent of DAB+ cells indicating the percent of cells transduced); percent positive cells (e.g., neurons, motor neurons, and astrocytes) in brain and spinal cord regions by immunofluorescence microscopy; and viral DNA (biodistribution) by ddPCR. [0399] As shown in Table 18, TTJ-001 demonstrated increased CNS transduction and/or biodistribution in several regions of the brain (greater than 10-20% of cells transduced observed in several regions) and spinal cord of the cynomolgus macaques after intravenous administration at a relatively low dose of 4e12 vg/kg. More specifically, TTJ-001 was capable of transducing up to 11% of cells in the putamen; up to 19% of cells in the caudate, and up to 23% of cells in the putamen. TTJ-001 also showed improved delivery to the spinal cord relative to AAV9, at a dose of 4e12 vg/kg. [0400] TTJ-001 also demonstrated increased transduction and/or biodistribution in the peripheral tissues of the muscle, specifically the vastus lateralis muscle and the gastrocnemius muscle relative to AAV9, and comparable transduction and/or biodistribution in the heart relative to AAV9 (Table 19 and Table 20). TTJ-001 also appeared to exhibit partial de-targeting in the liver (Table 19 and Table 20).
Attorney Docket No.14640.0105-00304 Table 18. Quantification of viral genome copies per diploid genome (vg/dg) (biodistribution) by ddPCR, transgene mRNA by RT-qPCR (mRNA = transgene mRNA fold over housekeeping gene), and percent of DAB+ cells in tissues of the CNS of cynomolgus macaques Putamen Capsid vg/dg mRNA %DAB+ Cells AAV9 0.1 0.03 2

tion) by ddPCR, transgene mRNA by RT-qPCR (mRNA = transgene mRNA fold over housekeeping gene), and percent of DAB+ cells in peripheral tissues of cynomolgus macaques Liver Capsid vg/dg mRNA %DAB+ Cells
Table 20. Ratio of viral genome copies per diploid genome (vg/dg) (biodistribution) or transgene mRNA fold over housekeeping gene (mRNA) in the muscle and motor cortex relative to the liver and/or heart Gastrocnemius to Liver Ca sid Biodistribution mRNA
Attorney Docket No.14640.0105-00304 Gastrocnemius to Heart Capsid Biodistribution mRNA AAV9 0.25 0.083 d CNS tropism

but also increased tropism in the heart and muscle in cynomolgus macaques. Example 3. Evaluation of TTJ-001 AAV9 capsid variant in Diverse Primate Species [0402] This Example evaluates the tropism and cross-species compatibility of the TTJ-001 (SEQ ID NO: 981 (amino acid) and 983 (DNA), comprising SEQ ID NO: 941 or 201) capsid variant in two diverse primate species, marmosets (Callithrix jacchus) and African green monkeys (Chlorocebus sabaeus), as compared to their tropism in cynomolgus macaques (Macaca fascicularis) provided in Example 1 and 2. [0403] To investigate tropism in African green monkeys, AAV particles comprising the TTJ-001 capsid variant or an AAV9 control, under the control of a synapsin promoter, were intravenously injected into NHPs (n=2, 3-12 years of age) at a dose of 2E13 vg/kg. After 14-days in life, the brains and tissues (liver, DRG, quadriceps, and heart) of the NHPs were collected and RNA was extracted. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the AAV9 wild-type control. [0404] To investigate tropism in marmoset monkeys, AAV particles comprising the TTJ-001 capsid variant or an AAV9 control, were intravenously injected into the NHPs (n=2, >10 months of age) at a dose of 2E13 vg/kg (8.75E12 vg/mL). After 28-days in life, the brains and tissues (liver quadriceps, and heart) of the NHPs were collected and RNA was extracted. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the AAV9 wild-type control. [0405] As provided in Table 21 (African green monkeys) and Table 22 (marmosets), the TTJ-001 capsid variant demonstrated increased CNS tropism in diverse primate species relative to the AAV9 control. The TTJ-001 capsid variant demonstrated a 460.2-fold increase in expression relative to AAV9 in the brain of cynomolgus macaques (Table 9, Example 1), a 14.67-fold increase in expression relative to AAV9 in the brain of African green monkeys, and a 24.2-fold increase in expression relative to AAV9 in the brain of marmosets. Table 21. NGS fold-enrichment of TTJ-001 AAV capsid variant comprising SEQ ID NO: 941 in African green monkeys SEQ Fold enrichment relative to AAV9 Sequence ID NO B i DR H Li DNA Li RNA M l DNA

Attorney Docket No.14640.0105-00304 Table 22. NGS-fold enrichment of TTJ-001 (comprises SEQ ID NO: 941) in marmosets S
equence SEQ ID Fold enrichment relative to AAV9 NO: Brain Heart Liver DNA Liver RNA Muscle DNA T
QDWHRI 941 24.2163 1.5775 0.0720 0.1706 3.6998 ated

increased CNS tropism relative to the AAV9 control in the CNS across three diverse primate species, providing evidence of strong cross-species capacity. Example 4. Maturation of TTJ-001 Capsid in NHPs and Mice [0407] This Example describes maturation of the AAV9 capsid variant, TTJ-001 (SEQ ID NO: 981 (amino acid) and 983 (DNA), comprising SEQ ID NO: 941 (encoded by SEQ ID NO: 942)) or 201) in two species of NHPs, cynomolgus macaques (Macaca fascicularis) and marmosets (Callithrix jacchus) to further enhance their transduction and biodistribution in the central nervous system as well as other tissues, and evolve the AAV capsid variants to provide further cross-species compatibility. The TTJ-001 capsid variant was also matured in mice. Two approaches were used to mature the TTJ-001 capsid sequence in order to randomize and mutate within and around the modifications comprised within loop VIII of the capsid variant. In the first maturation approach, sets of three contiguous amino acids were randomized across the mutagenesis region in the TTJ-001 sequence, which spanned from position 582 to position 593, numbered according to SEQ ID NO: 981. In the second maturation approach, mutagenic primers were used to introduce point mutations at a low frequency, scattered across the mutagenesis region in the TTJ-001 sequence ranging from position 582 to position 593, numbered according to SEQ ID NO: 981. [0408] The library of pooled matured AAV capsid variants generated from TTJ-001 using the first maturation approach and the library of pooled matured AAV capsid variants generated from TTJ-001 using the second maturation approach were each injected into two cynomolgus macaques (Macaca fascicularis), two marmosets (Callithrix jacchus), and three outbred mice. After a 14- or 28-day period in life, the brains of the NHPs and mice were isolated and RNA was extracted. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the TTJ-001 non-matured control or an AAV9 control, and the peptides comprised within the variants were identified. [0409] Following the RNA recovery and NGS analysis from the second maturation approach, the matured capsid variants were filtered based on their coefficient of variation (CV), which was calculated for each peptide across the brain samples taken from the NHPs and mice. Those that had a CV value <1 were identified, as these were the peptides that were reliably detected in the majority of samples isolated from the brains of the two NHPs. The peptides were also filtered based on a read per million (RPM) greater than or equal to 1 in the original virus stock. [0410] Table 23 provides the peptide sequences of the matured capsid variants from the first maturation approach having an RPM greater than or equal to 1 in the original virus stock, a CV of less
Attorney Docket No.14640.0105-00304 than 1 for the brain samples isolated, and that also demonstrated a 24-fold or greater fold-increase in expression in the brain relative to the AAV9 control in the brain of cynomolgus macaques (Macaca fascicularis) and/or a 50-fold or greater fold-increase in expression in the brain relative to the AAV9 control in the brain of marmosets (Callithrix jacchus), that were also detected in the maturation screen in mice, as these variants demonstrate cross-species compatibility. For example, the TTJ-001 capsid variants comprising SEQ ID NOs: 205, 208, 212, 219, 286, or 307 generated using the first maturation approach demonstrated a fold change greater than the non-matured TTJ-001 control and the AAV9 control in both species of NHPs and mice. TTJ-001 capsid variants comprising any one of SEQ ID NOs: 262-265 generated using the first maturation approach demonstrated a fold change greater than the non- matured TTJ-001 control and the AAV9 control in both species of NHPs. Table 23. NGS fold-enrichment of the TTJ-001 matured AAV capsid variants in the brain of cynomolgus macaques, marmosets, and mice generated using the first maturation approach Brain of cynomolgus macaques Brain of marmosets Brain of mice SEQ F ld F ld F ld F ld F ld F ld to 1
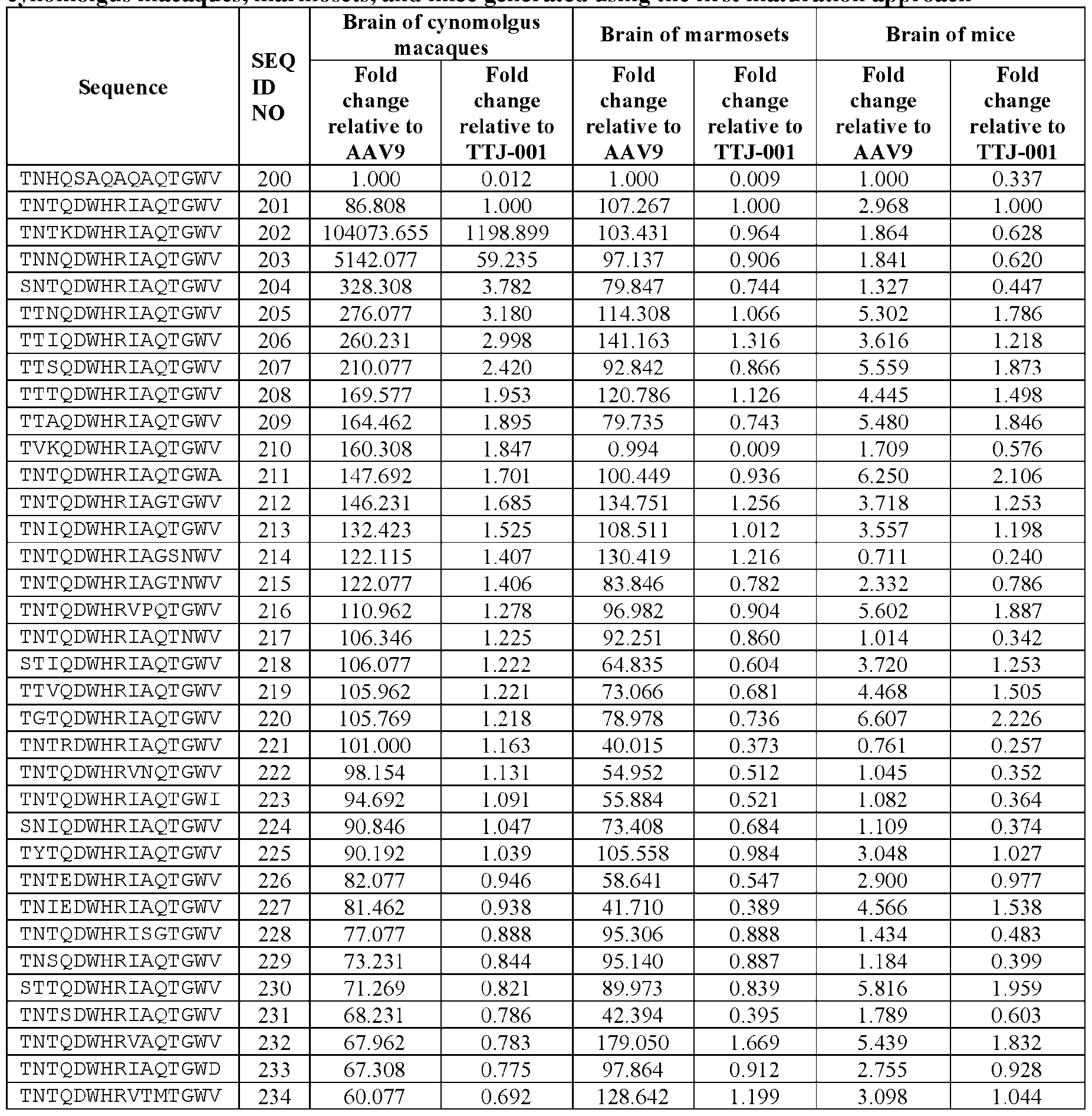
Attorney Docket No.14640.0105-00304 T
NTQDWHRISMTGWV 235 55.808 0.643 64.425 0.601 2.134 0.719 T
NTQDWHRISQTGWV 236 55.808 0.643 106.204 0.990 1.941 0.654 T
NTQDWHRVSQTGWV 237 53.538 0.617 149.383 1.393 3.516 1.185 8 8 9 9 4

maturation approach having an RPM greater than or equal to 1 in the original virus stock, a CV of less than 1 for the brain samples isolated, and that also demonstrated a 36-fold or greater fold-increase in expression in the brain relative to the AAV9 control in the brain of cynomolgus macaques (Macaca fascicularis) and/or about a 10-fold or greater fold-increase in expression in the brain relative to the AAV9 control in the brain of marmosets (Callithrix jacchus), that were also detected in the maturation screen in mice, as these variants demonstrate cross-species compatibility. For example, the TTJ-001 capsid variants comprising SEQ ID NOs: 283, 286, 290, 291, and 293 generated using the second maturation approach demonstrated a fold change greater than the non-matured TTJ-001 control and the AAV9 control in both species of NHPs and mice. The TTJ-001 capsid variants comprising SEQ ID NOs: 205, 208, 212, 213, or 307 generated using the second maturation approach demonstrated a fold change greater than the non-matured TTJ-001 control in cynomolgus macaques and mice and a fold-change greater than the AAV9 control in both species of NHPs and mice. The TTJ-001 capsid variants comprising SEQ ID NOs: 219 and 265 generated using the second maturation approach demonstrated a fold change greater than the non-matured TTJ-001 control and the AAV9 control in both species of NHPs and the TTJ-001 capsid variant comprising SEQ ID NO: 263 generated using the second
Attorney Docket No.14640.0105-00304 maturation approach lead to a fold-change greater than AAV9 in both species of NHPs and a fold-change greater than TTJ-001 in marmosets. Table 24. NGS fold-enrichment of the TTJ-001 matured AAV capsid variants in the brain of cynomolgus macaques, marmosets, and mice generated using the second maturation approach Brain of cynomolgus Brain of marmosets macaque Brain of mice SEQ s (Callithrix jacchus) Fold Fold Fold Fold Fold Fold to 1

Attorney Docket No.14640.0105-00304 T
NTQDWHRVAQTGWV 232 67.265 0.914 15.332 1.261 2.838 1.566 S
NSQDWHRIAQTGWV 249 66.347 0.901 3.312 0.272 0.374 0.207 T
NTQDWHRIAQTGWC 302 66.163 0.899 10.852 0.893 2.340 1.292

Attorney Docket No.14640.0105-00304 [0412] Table 25 provides the sequences of TTJ-001 capsid variants with an RPM greater than or equal to 1 in the original virus stock, a CV value of less than or equal to 1 and a fold-change in the brain of cynomolgus macaques of greater than 50 relative to AAV9. Approximately 59 variants were identified using the first maturation approach with a fold change above the TTJ-001 non-matured control in the brain of cynomolgus macaques. Approximately 181 variants were identified using the second maturation approach with a fold change above the TTJ-001 non-matured control in the brain of cynomolgus macaques. Many of the variants with increased expression in the brain of cynomolgus macaques relative to AAV9 and the non-matured TTJ-001 controls comprised a K at position 585 numbered according to SEQ ID NO: 981 or 138. Table 25. NGS fold-enrichment of TTJ-001 in the brain of cynomolgus macaques by both maturation approaches First Maturation Approach Sequence SEQ ID NO: Fold change relative to AAV9 Fold change relative to TTJ-001

Attorney Docket No.14640.0105-00304 T
NTQDWHRVPQTGWV 216 110.9615 1.278245 T
NTQDWHRIANNNWV 262 109.0385 1.256092 T
NTQDWHRIAQTNWV 217 106.3462 1.225078 01
Attorney Docket No.14640.0105-00304 T
NTKDWHRIGQTGWV 585 1618.429 21.986 T
NTKDWHRIAQTGSV 586 1599.571 21.730 T
NTKDWHRIEQTGWV 587 1590.959 21.613
Attorney Docket No.14640.0105-00304 T
NNQDWHRITQTGWV 628 238.796 3.244 T
NRKTALHLAQTGWV 629 234.857 3.190 N
NNQDWHRIAQTGWV 630 227.776 3.094
Attorney Docket No.14640.0105-00304 T
NIQDWHRIAQTGWV 213 89.224 1.212 T
NTQDWHRVPQTGWV 216 89.041 1.210 S
NTQDWHRISQTGWV 292 88.245 1.199
Attorney Docket No.14640.0105-00304 T
NNQDWQRIAQTGWV 304 65.449 0.889 T
NIQDWHRIPQTGWV 305 64.918 0.882 T
TTQDWHRIAQTGWI 306 64.755 0.880
Attorney Docket No.14640.0105-00304 T
ITQDWHRTAQTGWV 317 50.143 0.681 R
NTQDWHRIAQTGWA 742 50.082 0.680 T
NNQDGHRIAQTGSV 743 50.061 0.680

psid variants (AAV9 capsid variants) with loop VIII modifications were generated with significantly enhanced CNS tropism over wild-type AAV9 controls in two species of NHPs (cynomolgus macaques (Macaca fascicularis) and marmosets (Callithrix jacchus)) and mice, as well as cross-species compatibility. Example 5. Individual Capsid Characterization in Mice [0414] The goal of these experiments was to determine the transduction level, tropism, ability to cross the blood brain barrier, and overall spatial distribution in the central nervous system (CNS) of a capsid variant selected from the study described in Example 1 relative to AAV9 following intravenous injection in BALB/c mice. The capsid variant was TTJ-001 (SEQ ID NO: 981 (amino acid) and 983 (DNA), comprising SEQ ID NO: 941 or 201), as outlined in Table 3 above. The amino acid and DNA sequences of TTJ-001 are provided, e.g., in Tables 4 and 5, respectively. [0415] AAV particles were generated with this capsid variant encapsulating a luciferase-EGFP-HA tagged reporter gene driven by a CBA promoter encoded by a single stranded viral genome. The TTJ-001 capsid variant and AAV9 control were tested by intravenously administering the AAV particle formulation at 2e13 VG/kg (5e11/dose) to mice (n=3). The in-life period was 28 days and the brain and the liver were collected for measuring transgene mRNA, transgene protein, and viral DNA (biodistribution). [0416] At 27 days post-injection of the AAV particles encapsulated in the TTJ-001 capsid variant, mice were injected with luciferin and imaged on the ventral and dorsal side by the IVIS imager. Robust luciferase signal was observed in mice injected with AAV particles encapsulated in the AAV9 control capsid but less luciferase signal was observed in mice injected with the AAV particles encapsulated in the TTJ-001 capsid variant. [0417] The brains and livers were isolated on day 28 and subsequently assayed by qPCR for the presence of transgene mRNA, as a measure of transgene expression. Data were provided as average mRNA fold change for the transgene relative to a housekeeping gene as well as the fold change relative to the AAV9 control (Table 26). As shown in Table 26, mRNA expression from the TTJ-001 capsid variant was slightly increased in the brain of the mice relative to the AAV9 control. More specifically, mRNA expression was approximately 1.3-fold higher from the TTJ-001 capsid variant compared to wild- type AAV9 in the brain of the mice. In the liver, mRNA expression from the TTJ-001 capsid variant was decreased relative to the AAV9 control. Table 26. Transgene mRNA expression with the TTJ-001 capsid variant in the brain and liver of mice Capsid Variant Tissue AVG transgene mRNA fold change Fold change relative to AAV9 l ti t h k i

Attorney Docket No.14640.0105-00304 AAV9 Brain 0.544 1.0 Liver 8.938 1.0 TTJ-001 Brain 0.734 1.349 esence of
viral DNA by qPCR as a measure of viral genome levels. Data are provided in Table 27 as average DNA (viral genome (VG) copies per diploid genome (dg) as well as fold change relative to the AAV9 control. As shown in Table 27, biodistribution of TTJ-001, was slightly higher in the mouse brain relative to the wild-type AAV9 control. Biodistribution of TTJ-001 was lower in the mouse liver relative to the wild- type AAV9 control. Table 27. Viral DNA biodistribution with the TTJ-001 capsid variant in the brain and liver of mice Capsid Variant Tissue AVG VG per Fold change relative to AAV9 diploid cell AAV9 Brain 0141 10

jected to immunohistochemistry staining to evaluate overall tropism in the CNS and peripheral tissues and transgene expression. TTJ-001 showed comparable staining in the brain, muscle, and liver, as compared to the AAV9 control. Additionally, TTJ-001 demonstrated decreased staining in the heart relative to AAV9. Example 6. Evaluation of additional TTJ-001 capsid variants [0420] Matured variants of the TTJ-001 capsid variant generated using the methods described in Example 4 were injected into cynomolgus macaques (Macaca fascicularis), marmosets (Callithrix jacchus), or mice. The matured AAV capsid variants generated from the TTJ-001 non-matured capsid variant (SEQ ID NO: 981 (amino acid) and SEQ ID NO: 983 (DNA), comprising SEQ ID NO: 941 or SEQ ID NO: 201)) were injected into two NHPs (cynomolgus macaques or marmosets) or mice. After a period in life, the brains, heart, muscles, and/or livers of the NHPs or mice were isolated and DNA/RNA was extracted. Following DNA/RNA recovery and amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the TTJ-001 control or an AAV9 control, and the peptides comprised within the AAV capsid variants were identified. [0421] Table 28 provides the sequences of the matured capsid variants having a counts per million greater than or equal to 10, a CV of less than or equal to 1, and an average fold-change in cDNA levels in the brain relative to the cDNA levels of TTJ-001 that is greater than or equal to 1 in cynomolgus macaques. These variants demonstrated increased tropism in the brain of cynomolgus macaques. Table 28. NGS fold-change in cDNA or viral DNA (vDNA) levels of TTJ-001 matured AAV capsid variants in the brains and livers of cynomolgus macaques (changes in the peptide sequence relative to TTJ-001 are bolded and underlined) (Fc rel. to AAV9= fold change relative to AAV9; Fc. rel. to TTJ-001= fold change relative to TTJ-001) Sequence Brain cDNA Brain vDNA Liver cDNA
Attorney Docket No.14640.0105-00304 SEQ Fc rel. Fc r Fc rel. Fc rel. ID to el. to to Fc rel. to t Fc rel. to TTJ-001 AAV9 TTJ-00 o NO: AAV9 1 AAV9 TTJ-001 er million

greater than or equal to 10, a CV of less than or equal to 1, and an average fold-change in cDNA levels in the brain relative to the cDNA levels of TTJ-001 that is greater than or equal to 1 in marmosets. These variants demonstrated increased tropism in the marmoset brain. AAV capsid variants comprising SEQ ID NO: 216 or SEQ ID NO: 943, were able to cross the blood brain barrier and demonstrate increased expression in the brains of both cynomolgus macaques (Table 28) and marmosets (Table 29) relative to AAV9, and comparable or increased expression relative to TTJ-001 in the same tissues. Table 29. NGS fold-change in cDNA levels of TTJ-001 matured AAV capsid variants in the brains, hearts, and muscles of marmosets (changes in the peptide sequence relative to TTJ-001 are bolded and underlined) (Fc rel. to AAV9= fold change relative to AAV9; Fc. rel. to TTJ-001= fold change relative to TTJ-001) Brain cDNA Heart cDNA Muscle cDNA SEQ Sequence ID Fc rel. Fc r Fc rel. Fc rel. t el. to t Fc rel. to t Fc rel. to 1

ounts per million greater than or equal to 10, a CV across the brain tissues of less than or equal to 1, and an average fold-change in cDNA levels in the brain relative to the cDNA levels of TTJ-001 that is greater than or equal to 1 in marmosets that were generated using two different maturation approaches. These variants demonstrated increased or comparable tropism in the marmoset brain relative to TTJ-001. Table 30. NGS fold-change in cDNA levels of TTJ-001 matured AAV capsid variants in the brains, liver, hearts, and muscles of marmosets (changes in the peptide sequence relative to TTJ-001 are bolded and underlined) Mutational Approach 1 SEQ ID Fold change relative to TTJ-001 Fold change relative to AAV9 9

p p g lso isolated from the brains of mice following two maturation approaches, as they demonstrated increased tropism in the brain relative to TTJ-001 (Table 31). Table 31 provides data that was filtered by a counts per million greater than or equal to 10, a CV across the brain tissues of less than or equal to 1, and an
Attorney Docket No.14640.0105-00304 average fold-change in cDNA levels in the brain relative to the cDNA levels of TTJ-001 that is greater than or equal to 1 in mice. Table 31. NGS fold-change in cDNA levels of TTJ-001 matured AAV capsid variants in the brains, hearts, and muscles of mice (changes in the peptide sequence relative to TTJ-001 are bolded and underlined) Mutational Approach 1 SEQ Fold change relative to TTJ-001 Fold change relative to AAV9 Sequence ID NO: Brain Liver Heart 9 n

greater than or equal to 10, a CV of less than or equal to 1, and an average fold-change in cDNA levels in the heart relative to the cDNA levels of AAV9 that is greater than or equal to 2 in cynomolgus macaques. These variants demonstrated increased tropism in the heart and muscles (intercostal, diaphragm, and quadriceps) relative to AAV9 and comparable expression to TTJ-001 in these same tissues in cynomolgus macaques. Table 32. NGS fold-change in cDNA levels of TTJ-001 matured AAV capsid variants in the muscle, heart, and liver of cynomolgus macaques (changes in the peptide sequence relative to TTJ-001 are bolded and underlined) Diaphragm Quadriceps Intercostal Muscle Heart Liver g. C to J- 1 0
0 55 51 95

, demonstrated increased expression in the heart and brain relative to AAV9 and TTJ-001 and in the muscle relative to TTJ-001 in marmosets (Table 33). The AAV capsid variants comprising SEQ ID NO: 241 or SEQ ID NO: 984 also demonstrated detargeting in the liver relative to AAV9 and TTJ-001 (Table 33). Table 33 provides data that was filtered by a counts per million greater than or equal to 10, a CV of less than or equal to 1, and an average fold-change in cDNA levels in the heart relative to the cDNA levels of AAV9 that is greater than or equal to 2 in marmosets. Table 33. NGS fold-change in cDNA levels of TTJ-001 matured AAV capsid variants in the brains, hearts, livers, and muscles of marmosets (changes in the peptide sequence relative to TTJ-001 are
Attorney Docket No.14640.0105-00304 bolded and underlined) (Fc rel. to AAV9= fold change relative to AAV9; Fc. rel. to TTJ-001= fold change relative to TTJ-001) Heart cDNA Muscle cDNA Brain cDNA Liver cDNA SEQ uence ID F Fc rel. Fc Seq c rel. Fc rel. Fc rel. Fc rel. Fc rel. Fc rel. t t TT t to t rel. to t t TTJ- 9
0 1 9

with loop VIII modifications were generated with enhanced CNS, heart, and/or muscle tropism over wild-type AAV9 controls and/or the non-matured TTJ-001 capsid in two species of NHPs (cynomolgus macaques (Macaca fascicularis) and marmosets (Callithrix jacchus)) and mice. Equivalents and Scope [0428] Those skilled in the art will recognize, or be able to ascertain using no more than routine experimentation, many equivalents to the specific embodiments in accordance with the Detailed Description provided herein. The scope of the present disclosure is not intended to be limited to the above Detailed Description, but rather is as set forth in the appended claims. [0429] Where ranges are given, endpoints are included. Furthermore, it is to be understood that unless otherwise indicated or otherwise evident from the context and understanding of one of ordinary skill in the art, values that are expressed as ranges can assume any specific value or subrange within the stated ranges in different embodiments of the disclosure, to the tenth of the unit of the lower limit of the range, unless the context clearly dictates otherwise. [0430] In addition, it is to be understood that any particular embodiment of the present disclosure that falls within the prior art may be explicitly excluded from any one or more of the claims. Since such embodiments are deemed to be known to one of ordinary skill in the art, they may be excluded even if the exclusion is not set forth explicitly herein. Any particular embodiment of the compositions of the disclosure (e.g., any, composition, therapeutic or active ingredient; any method of production; any method of use; etc.) can be excluded from any one or more claims, for any reason, whether or not related to the existence of prior art. [0431] It is to be understood that the words which have been used are words of description rather than limitation, and that changes may be made within the purview of the appended claims without departing from the true scope and spirit of the disclosure in its broader aspects. [0432] While the present disclosure has been described at some length and with some particularity with respect to the several described embodiments, it is not intended that it should be limited to any such particulars or embodiments or any particular embodiment, but it is to be construed with references to the appended claims so as to provide the broadest possible interpretation of such claims in view of the prior art and, therefore, to effectively encompass the intended scope of the disclosure.
Attorney Docket No.14640.0105-00304 [0433] All publications, patent applications, patents, and other references mentioned herein are incorporated by reference in their entirety. In case of conflict, the present specification, including definitions, will control. In addition, section headings, the materials, methods, and examples are illustrative only and not intended to be limiting.


















































