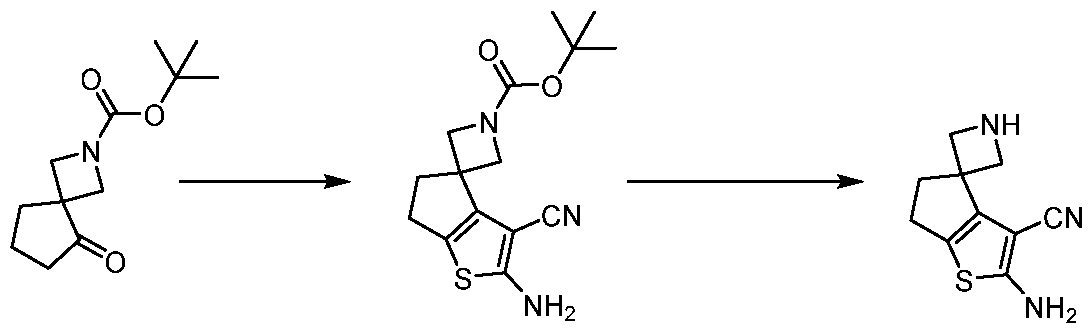
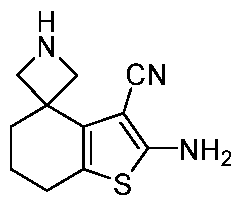
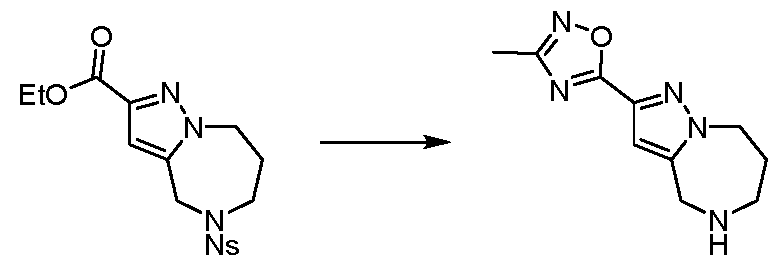
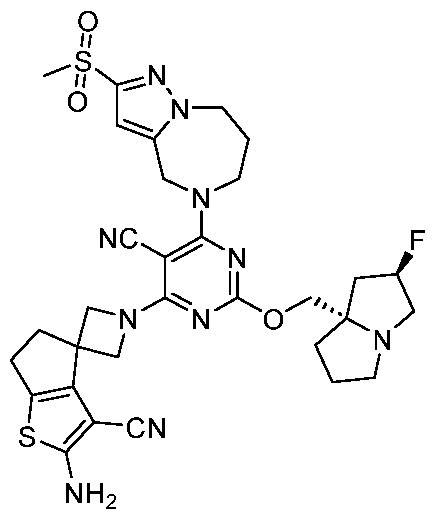
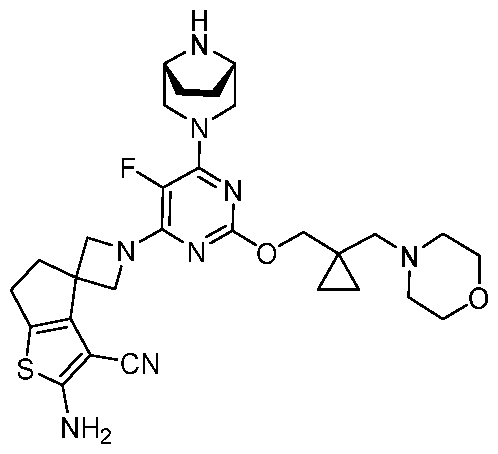
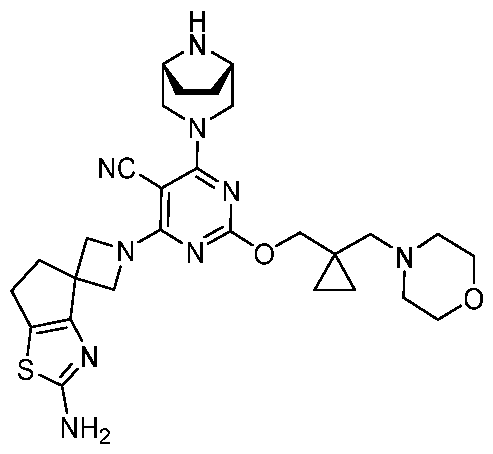
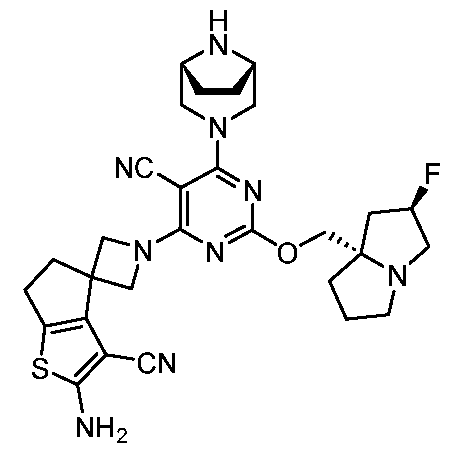
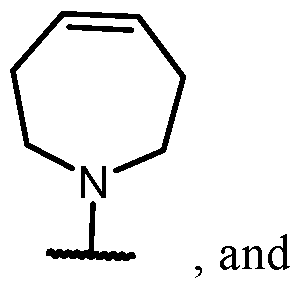WO2025106905A1 - Combination therapies with a kras modulator and an immunomodulator inhibitor - Google Patents
Combination therapies with a kras modulator and an immunomodulator inhibitor Download PDFInfo
- Publication number
- WO2025106905A1 WO2025106905A1 PCT/US2024/056253 US2024056253W WO2025106905A1 WO 2025106905 A1 WO2025106905 A1 WO 2025106905A1 US 2024056253 W US2024056253 W US 2024056253W WO 2025106905 A1 WO2025106905 A1 WO 2025106905A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- compound
- alkyl
- salt
- heterocycle
- optionally substituted
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/495—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having six-membered rings with two or more nitrogen atoms as the only ring heteroatoms, e.g. piperazine or tetrazines
- A61K31/505—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim
- A61K31/506—Pyrimidines; Hydrogenated pyrimidines, e.g. trimethoprim not condensed and containing further heterocyclic rings
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K31/00—Medicinal preparations containing organic active ingredients
- A61K31/33—Heterocyclic compounds
- A61K31/395—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins
- A61K31/55—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole
- A61K31/551—Heterocyclic compounds having nitrogen as a ring hetero atom, e.g. guanethidine or rifamycins having seven-membered rings, e.g. azelastine, pentylenetetrazole having two nitrogen atoms, e.g. dilazep
- A61K31/5513—1,4-Benzodiazepines, e.g. diazepam or clozapine
- A61K31/5517—1,4-Benzodiazepines, e.g. diazepam or clozapine condensed with five-membered rings having nitrogen as a ring hetero atom, e.g. imidazobenzodiazepines, triazolam
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61P—SPECIFIC THERAPEUTIC ACTIVITY OF CHEMICAL COMPOUNDS OR MEDICINAL PREPARATIONS
- A61P35/00—Antineoplastic agents
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K16/00—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies
- C07K16/18—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans
- C07K16/28—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants
- C07K16/2803—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily
- C07K16/2818—Immunoglobulins [IGs], e.g. monoclonal or polyclonal antibodies against material from animals or humans against receptors, cell surface antigens or cell surface determinants against the immunoglobulin superfamily against CD28 or CD152
Definitions
- KRAS The small GTPase protein Kirsten Rat Sarcoma 2 Viral Oncogene Homolog
- MAPK mitogen-activated protein kinase
- Ras has been recognized as a target in cancer for about 40 years, Ras- driven cancers remain among the most difficult to treat due to insensitivity to available targeted therapies.
- Ras encoded by the three major genes KRAS, NRAS and HRAS, has the highest frequency of mutation of any oncogene. All oncogenic Ras mutations drive the switch to accumulate in the active GTP -bound state.
- the most common Ras mutation found across human tumor types is KRAS G12D (e.g., see The AACR Project GENIE Consortium. Cancer Discovery, 2017. 7(8): p. 818-831. Dataset Version 4).
- Activating mutations in codon 12 impair the small GTPases’ ability to perform their role in hydrolyzing GTP. This regulatory impairment is fundamental for initiating and maintaining tumor progression.
- KRAS The small GTPase protein Kirsten Rat Sarcoma 2 Viral Oncogene Homolog
- MAPK mitogen-activated protein kinase
- Ras has been recognized as a target in cancer for about 40 years, Ras-driven cancers remain among the most difficult to treat due to insensitivity to available targeted therapies.
- Ras encoded by the three major genes KRAS, NRAS and HRAS, has the highest frequency of mutation of any oncogene. All oncogenic Ras mutations drive the switch to accumulate in the active GTP -bound state.
- the most common Ras mutation found across human tumor types is KRAS G12D (e.g., see The AACR Project GENIE Consortium. Cancer Discovery, 2017. 7(8): p. 818-831. Dataset Version 4).
- Activating mutations in codon 12 impair the small GTPases’ ability to perform their role in hydrolyzing GTP. This regulatory impairment is fundamental for initiating and maintaining tumor progression.
- GAP GTPase activating protein
- GEF guanine nucleotide exchange factor
- SOS guanine nucleotide exchange factor
- KRAS G12C mutations most common in lung adenocarcinoma, have been clinically shown to be susceptible to direct inhibition by covalent modification with small molecule inhibitors trapping the protein in the inactive GDP -bound state.
- KRAS G12D mutation confers a significantly slower intrinsic rate of GTP hydrolysis than G12C, resulting in more constitutive activation.
- oncogenic KRas mutations create an immunosuppressive microenvironment which can result in resistance to immune checkpoint blockade (ICB) therapy, including anti-PD-1 and anti-PD-Ll inhibitors.
- Activated KRas has been demonstrated to repress the expression of interferon regulatory factor 2 (IRF2), which directly represses CXCL3 expression.
- IRF2 interferon regulatory factor 2
- This KRas-mediated repression of IRF2 leads to increased expression of CXCL3, which binds to CXCR2 on myeloid-derived suppressor cells (MDSC) promoting migration of these cells to the tumor microenvironment.
- MDSC myeloid-derived suppressor cells
- oncogenic KRas signaling has been shown to promote tumor immunoresistance to ICB therapy by stabilizing PD-L1 mRNA via repression of the AU- rich element-binding protein tristetraprolin (TTP), which negatively regulates PD-L1 expression through AU-rich elements in the 3' UTR of PD-L1 mRNA (e.g., see Coelho et al., (2017) Immunity 47(6): 1083-1099).
- TTP AU- rich element-binding protein tristetraprolin
- KRas activating mutations upregulate IL-8 expression in NSCLC
- IL-8 plays a role in cell growth and migration in KRas-associated NSCLC (e.g., see Sunaga et al., (2012) Int. J. Cancer 130(8): 1733-1744).
- activated KRas mutations or other KRas-activating generic alterations expression may modulate many aspects of the immune system and can be responsible for the immunosuppressive tumor microenvironment in KRas mutant or hyperactivated Kras wildtype- associated tumors.
- the direct inhibition of KRas mutant or hyperactivated Kras wildtype-associated-mediated cell activity may reverse this reported immunosuppressive tumor microenvironment thereby improving the clinical activity of immune checkpoint blockade therapy, including the PD-1/PD-L1 pathway.
- the compounds described herein direct potency against tumor cells, compounds described herein may modulate the tumor microenvironment (TME) in favor of antitumor immunity, by modulating tumor cell cytokine/chemokine release.
- TAE tumor microenvironment
- the compounds described herein direct potency against tumor cells, compounds described herein may reprogram the tumor microenvironment (TME) in favor of antitumor immunity, by modulating tumor cell cytokine/chemokine release. Specifically, treatment with compounds described herein reduces immunosuppressive cytokine release while increasing immunostimulatory chemokine release in KRAS mutant cells. Results of our study herein (e.g., example 14) reveal a direct mechanism by which compounds described herein can modify tumor-cell-intrinsic immune signals to enhance antitumor immunity in a synergistic manner with the use of common immune checkpoint inhibitors.
- TEE tumor microenvironment
- the present disclosure provides combination therapies useful for treating a disease or disorder (e.g., cancer).
- a disease or disorder e.g., cancer
- an agent that disrupts Programmed cell death protein 1 (PD-1) and Programmed death-ligand 1 (PD-L1) axis signaling e.g., a PD-1 inhibitor, a PD-Ll inhibitor
- kits comprising the compositions and methods of use thereof.
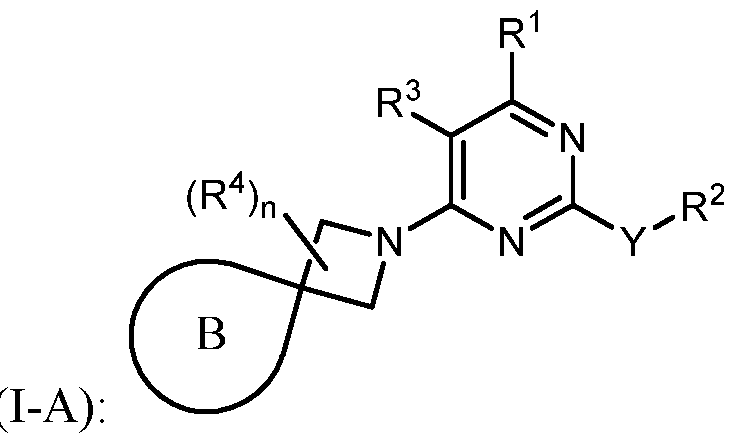
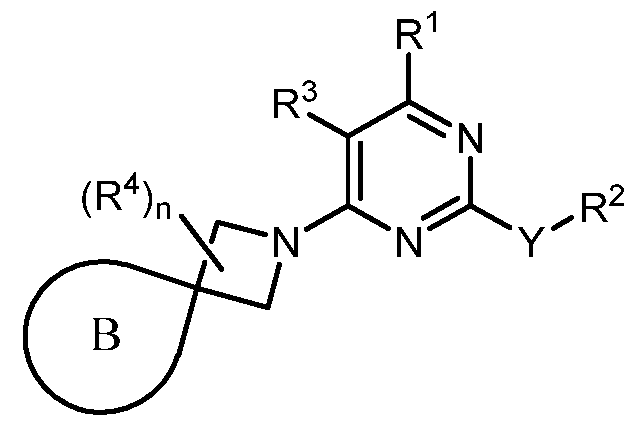
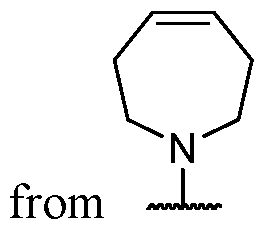
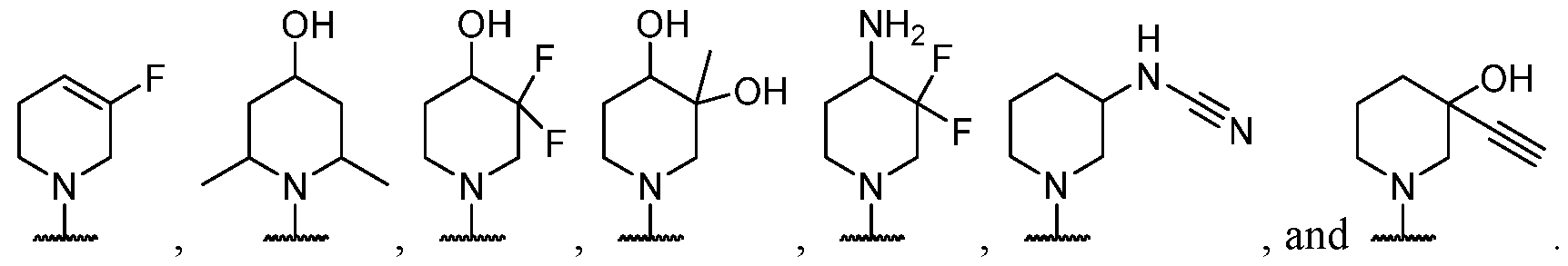
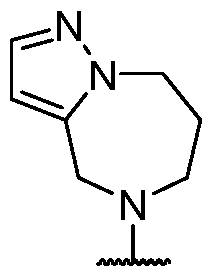
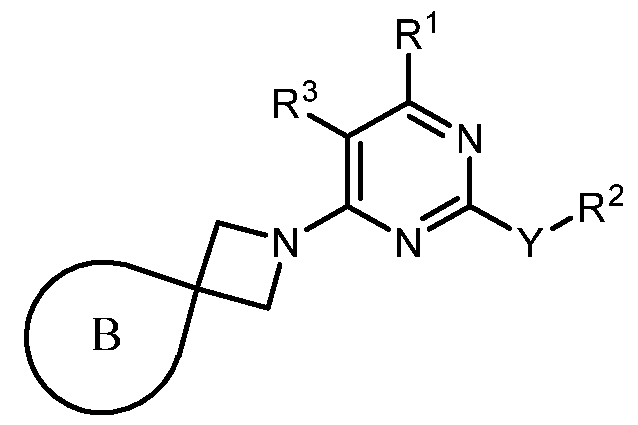
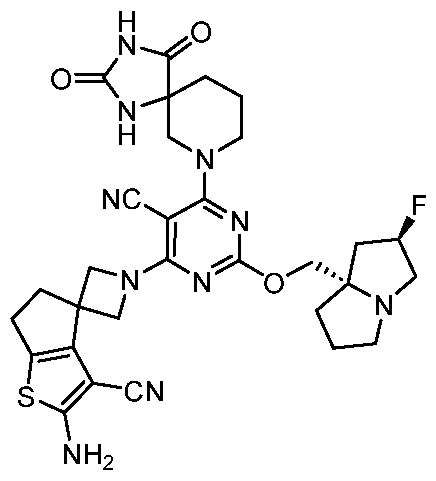
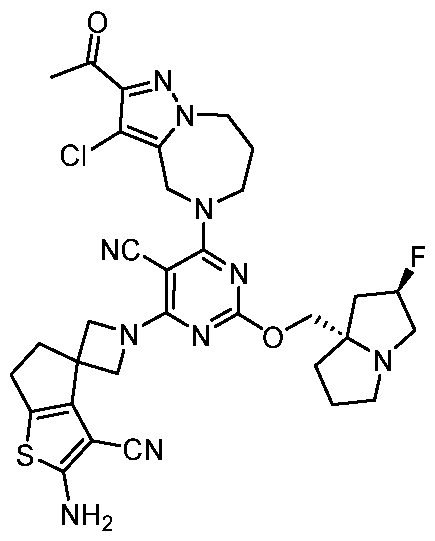
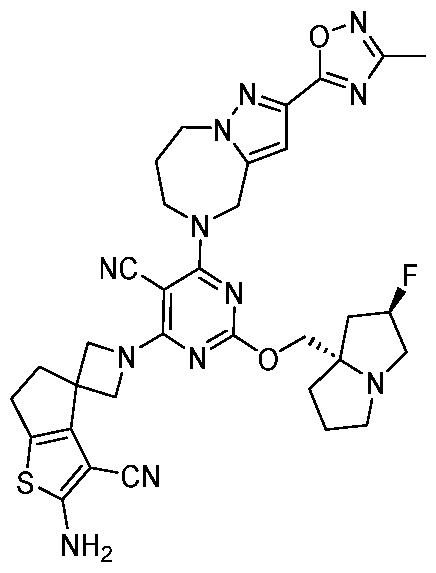
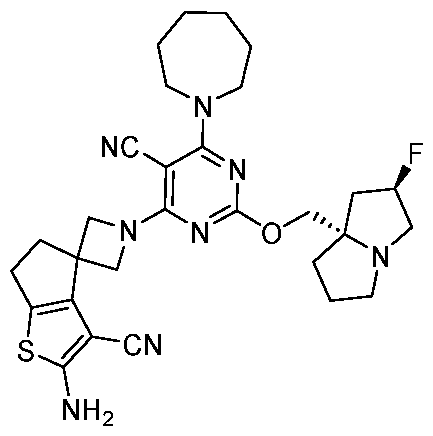
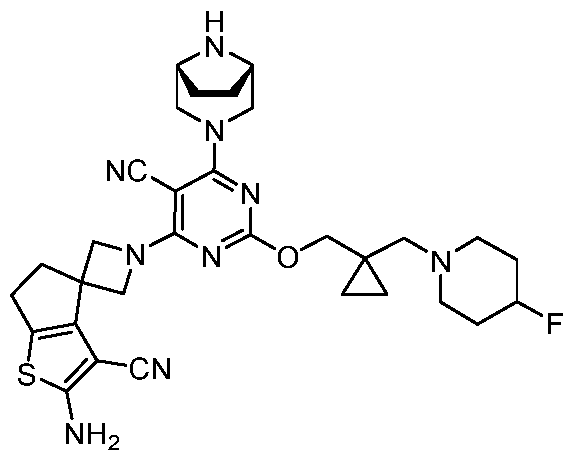
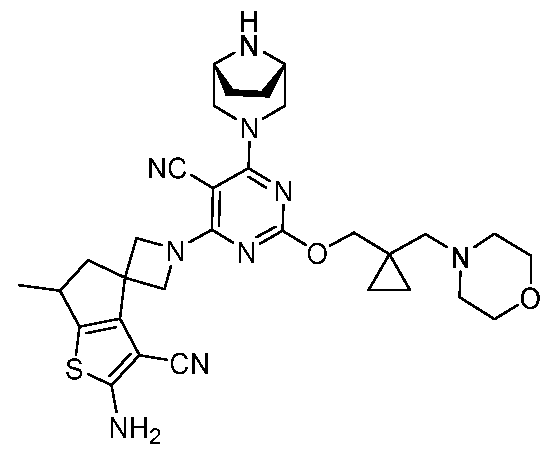
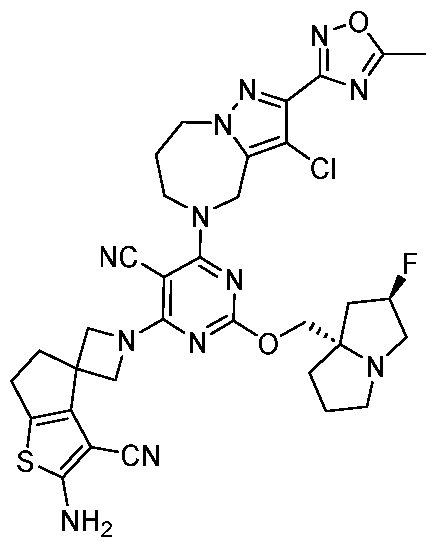
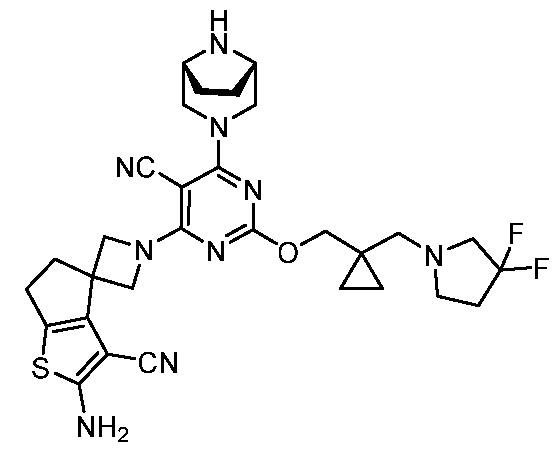
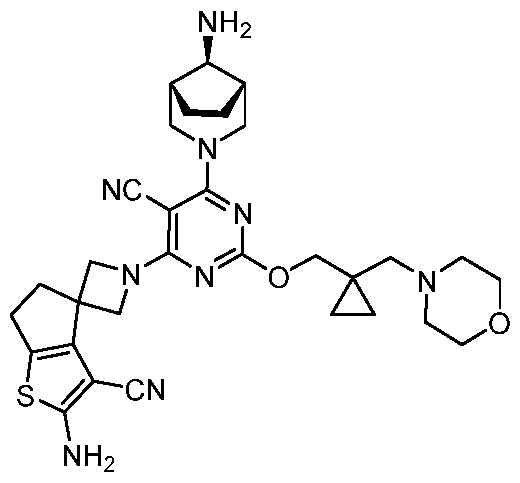
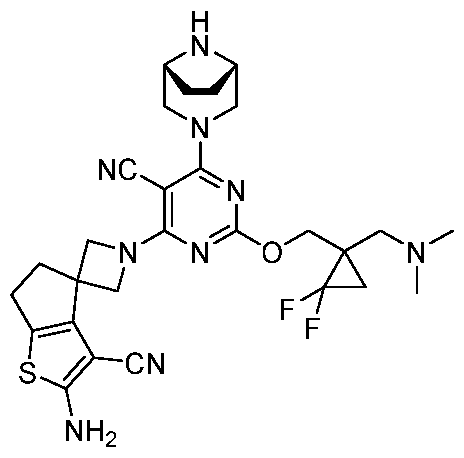
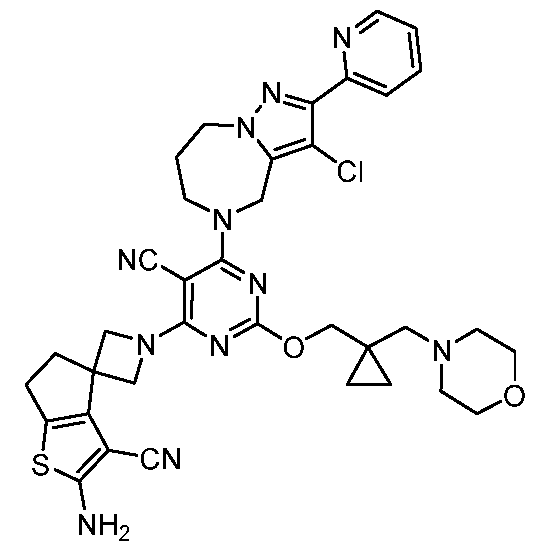
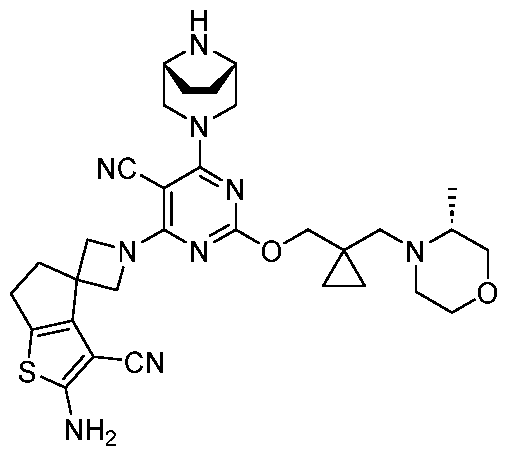
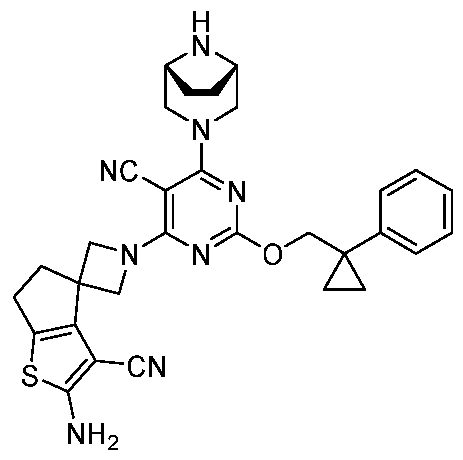
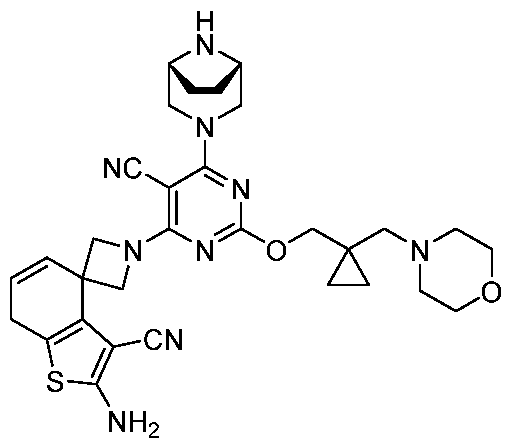
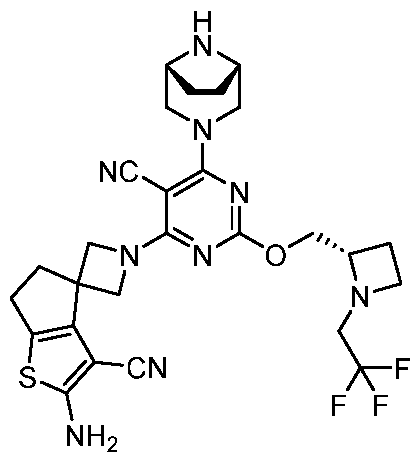
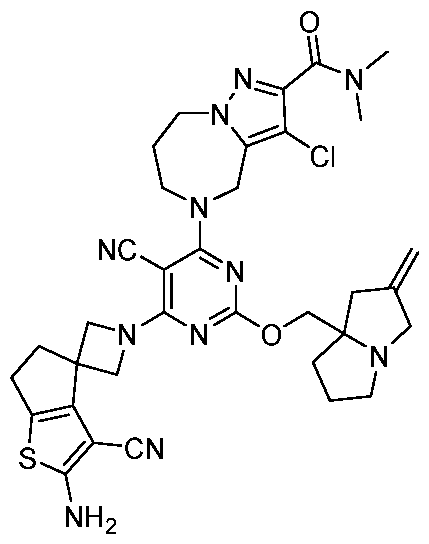
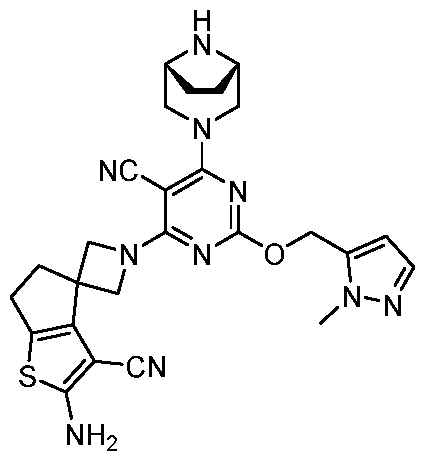
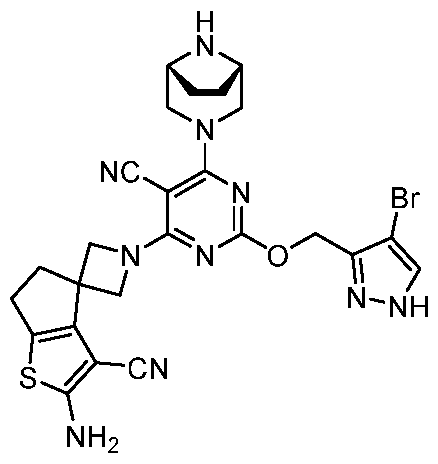
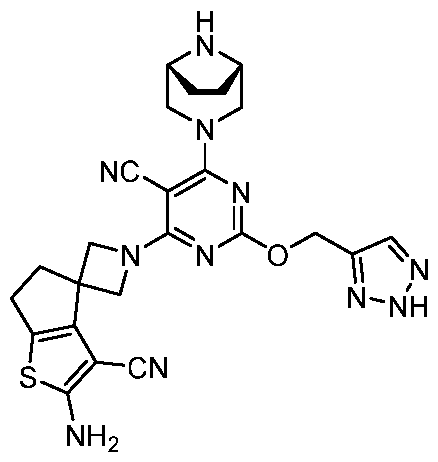
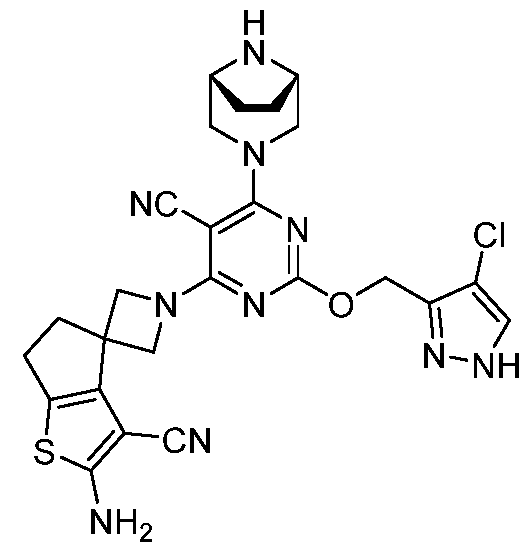
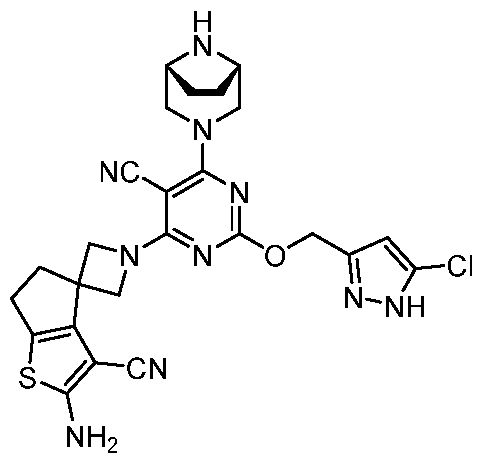
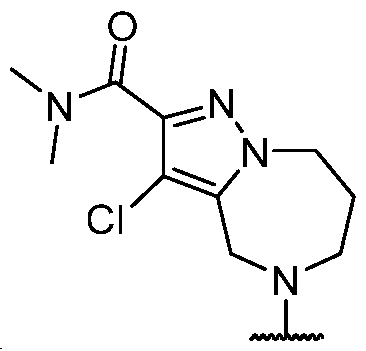
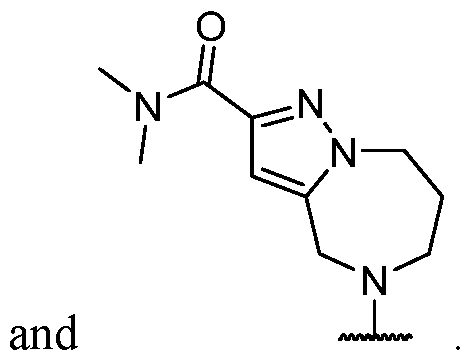
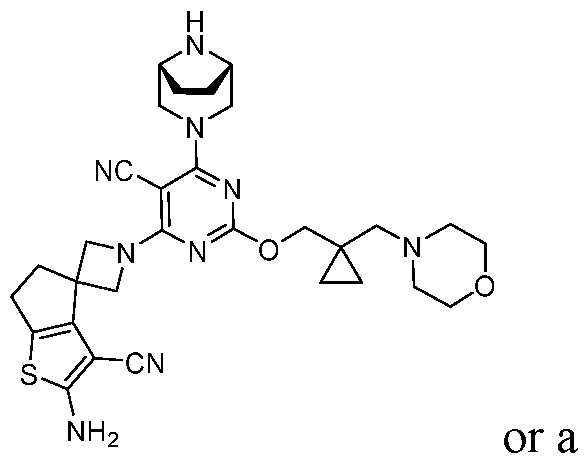
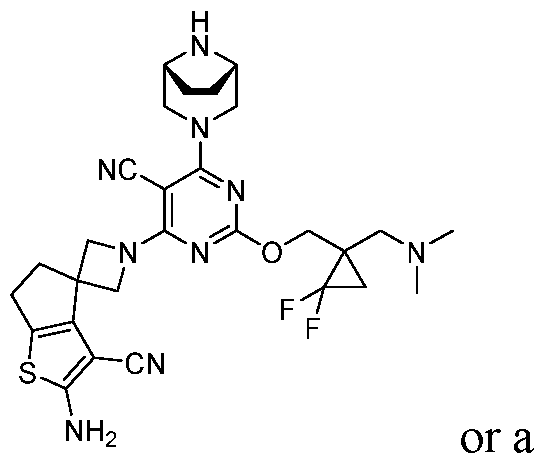
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor and a compound represented by the structure of Formula (I): Formula (I), or a pharmaceutically acceptable salt thereof wherein:
- Y is selected from a bond, -0-, -S-, and -N(R 5 )-;
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor and a compound represented by the structure of
- Y is selected from a bond, and -O-;
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor and a compound represented by the structure of
- Y is -0-
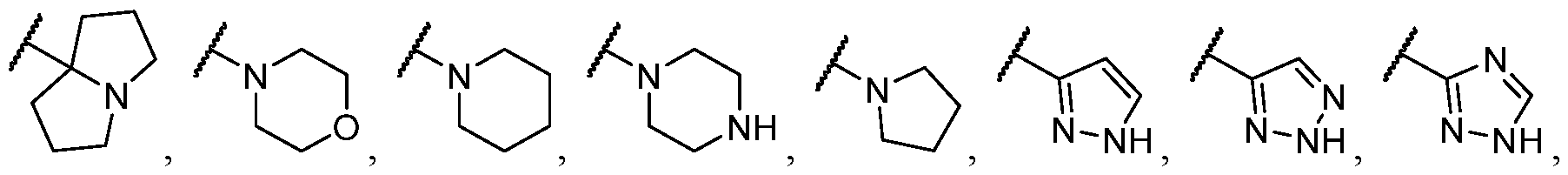
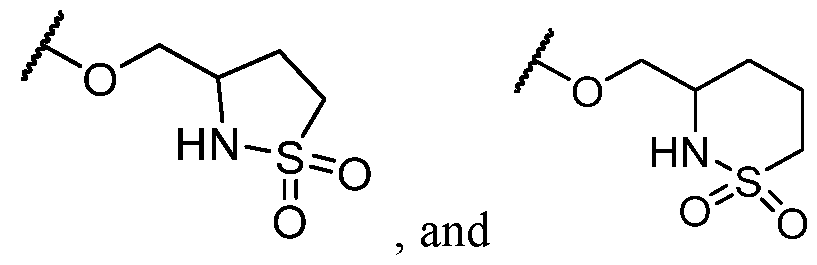
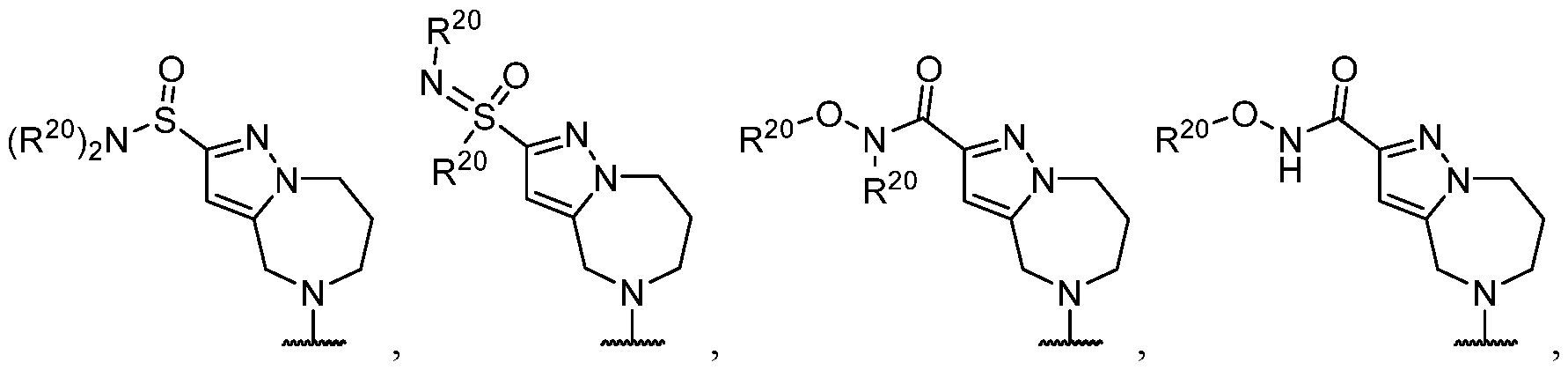
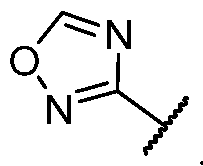
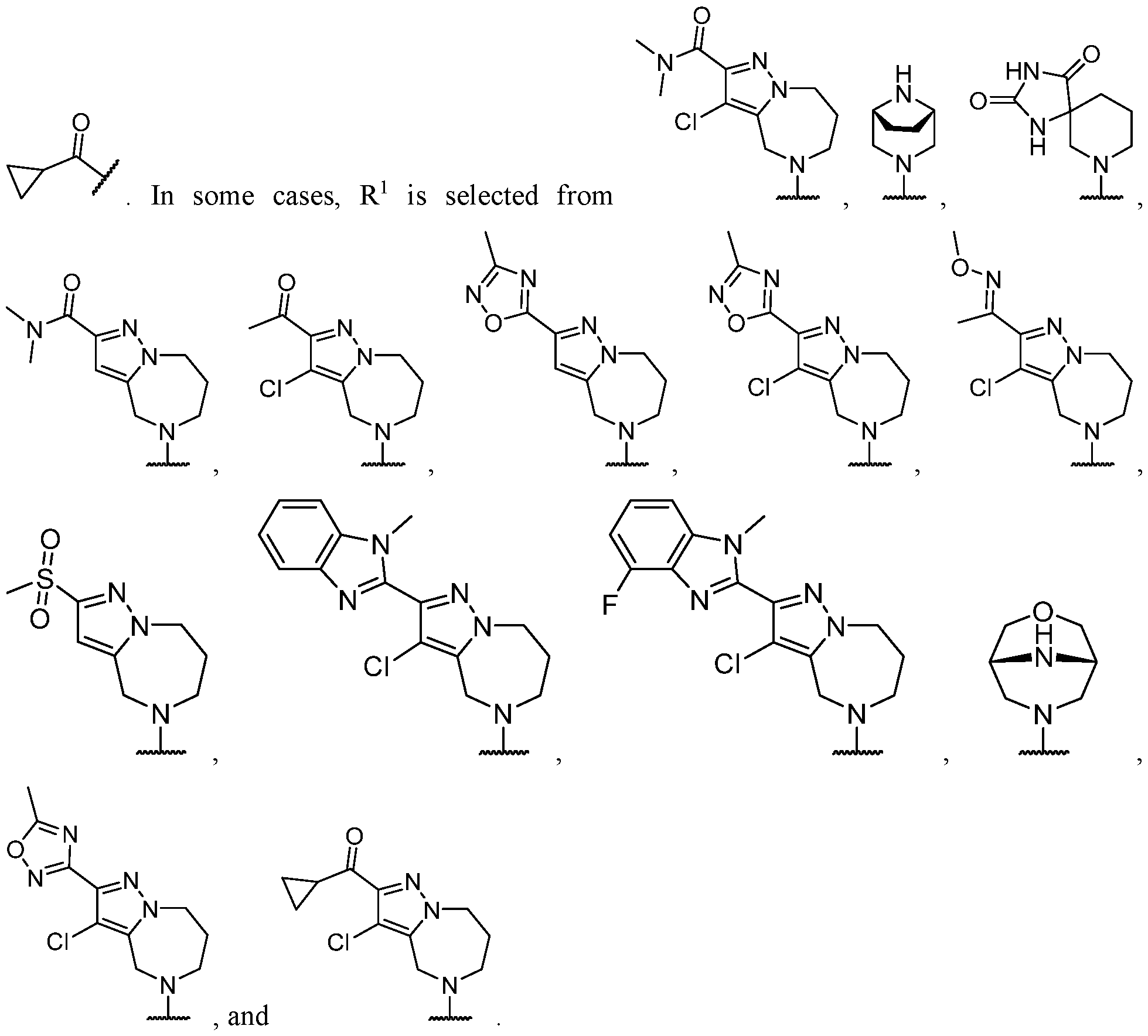
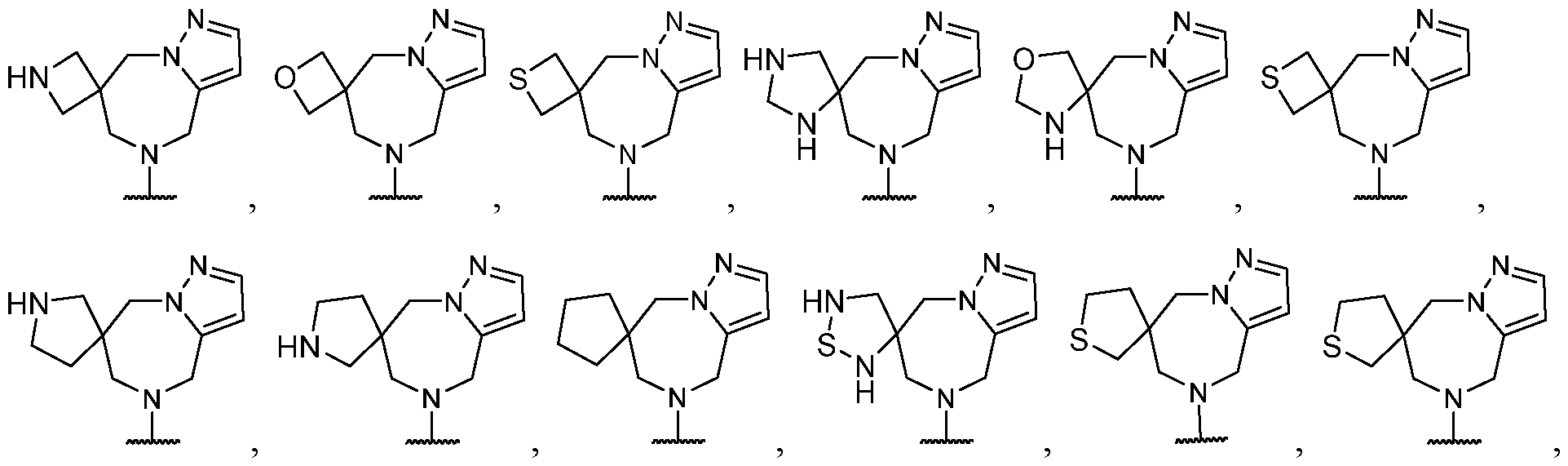
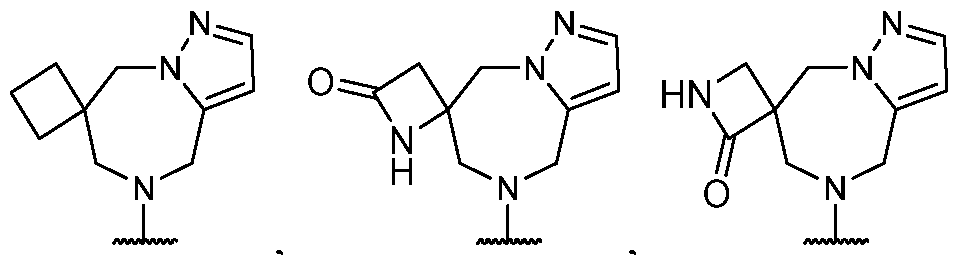
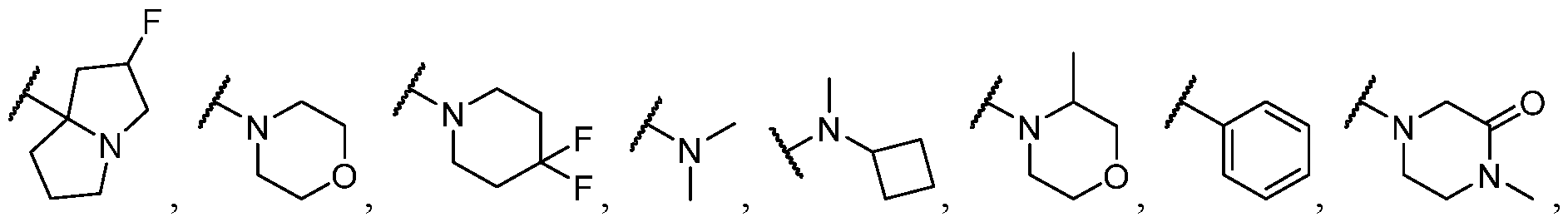
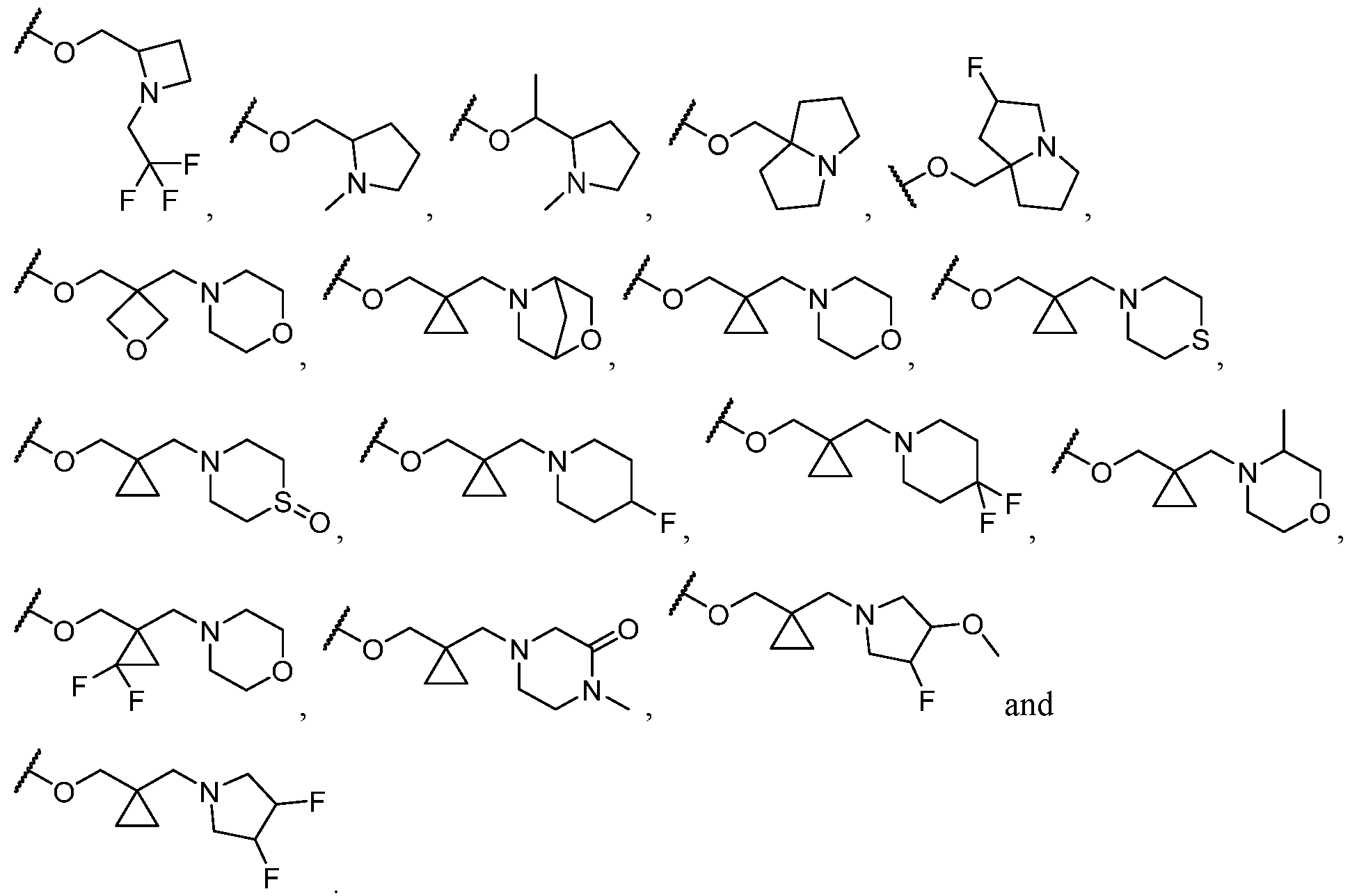
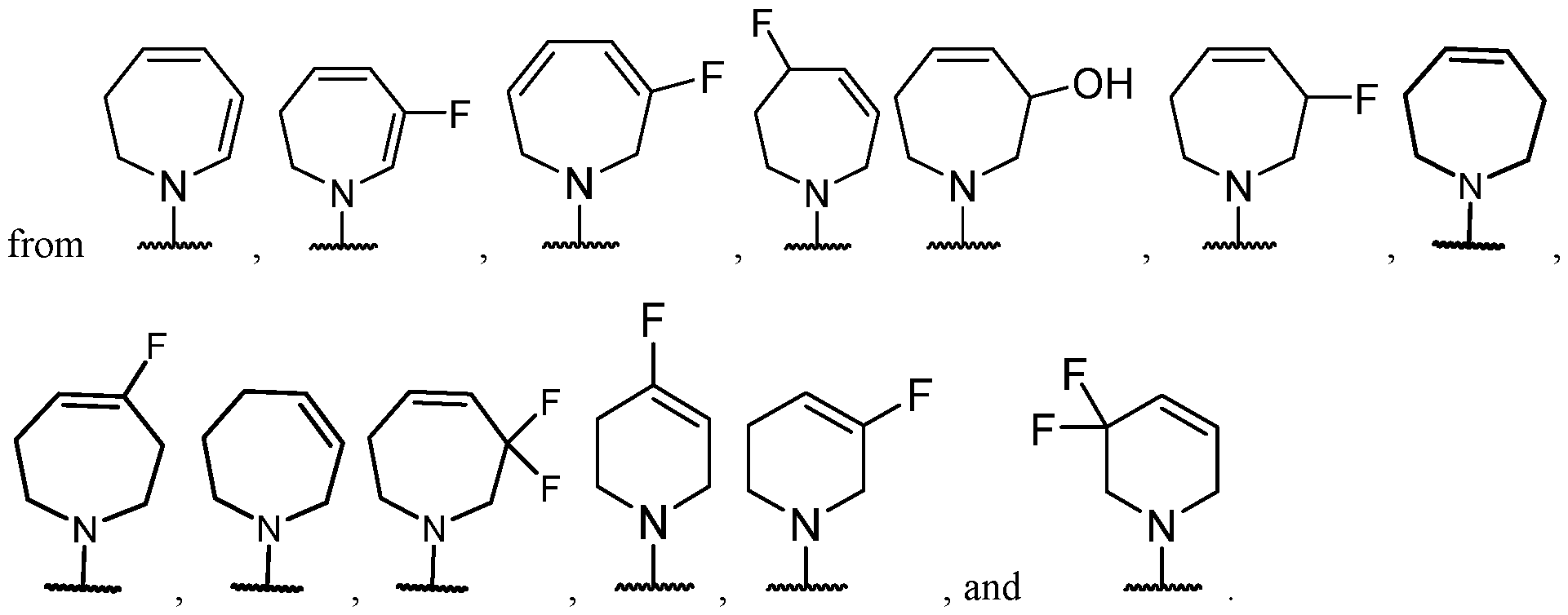
- R 2 is selected from -L-heterocycle, and -L-N(R 23 ) 2 , wherein the heterocycle portion of -L- heterocycle is optionally substituted with one or more R 6 .
- R 3 is selected from hydrogen, halogen, -CN, -N(R 20 ) 2 , -OR 20 , -C(O)R 20 , Ci-6 alkyl-N(R 20 ) 2 , Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, and Ci-6 alkyl; and each R 20 is independently selected from hydrogen; and Ci-6 alkyl, and C3-12 carbocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NH 2 , -N(CI-6 alkyl) 2 , Cnio alkyl, -Ci-io haloalkyl, -O-Ci-io alkyl, and oxo.
- a method of treating a cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, or a pharmaceutical composition thereof; and a compound selected pharmaceutically acceptable salt of any one thereof, or a pharmaceutical composition of any one thereof.
- a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, or a pharmaceutical composition thereof.
- FIG. 1 illustrates that compounds herein reduce immunosuppressive cytokine release while increasing immunostimulatory chemokine release in KRAS-mutant HP AC cells.
- Alkyl refers to a straight or branched hydrocarbon chain radical consisting solely of carbon and hydrogen atoms, containing no unsaturation, and preferably having from one to fifteen carbon atoms (z.e., C1-C15 alkyl).
- an alkyl comprises one to thirteen carbon atoms (z.e., C1-C13 alkyl).
- an alkyl comprises one to eight carbon atoms (z.e., Ci-Cs alkyl).
- an alkyl comprises one to five carbon atoms (z.e., C1-C5 alkyl).
- an alkyl comprises one to four carbon atoms (z.e., C1-C4 alkyl). In other embodiments, an alkyl comprises one to three carbon atoms (z.e., C1-C3 alkyl). In other embodiments, an alkyl comprises one to two carbon atoms (z.e., C1-C2 alkyl). In other embodiments, an alkyl comprises one carbon atom (z.e., Ci alkyl). In other embodiments, an alkyl comprises five to fifteen carbon atoms (z.e., C5-C15 alkyl). In other embodiments, an alkyl comprises five to eight carbon atoms (z.e., Cs-Cs alkyl).
- an alkyl comprises two to five carbon atoms (z.e., C2-C5 alkyl). In other embodiments, an alkyl comprises three to five carbon atoms (z.e., C3-C5 alkyl).
- the alkyl group is selected from methyl, ethyl, 1 -propyl (zz-propyl), 1 -methylethyl (z.w-propyl), 1 -butyl (zz-butyl), 1 -methylpropyl (sec-butyl), 2-methylpropyl (z.w-butyl), 1,1 -dimethylethyl (tert-butyl), 1 -pentyl (zz-pentyl).
- the alkyl is attached to the rest of the molecule by a single bond.
- C x -y when used in conjunction with a chemical moiety, such as alkyl, alkenyl, or alkynyl is meant to include groups that contain from x to y carbons in the chain.
- Ci-ealkyl refers to substituted or unsubstituted saturated hydrocarbon groups, including straight-chain alkyl and branched-chain alkyl groups that contain from 1 to 6 carbons.
- -C x.y alkylene- refers to a substituted or unsubstituted alkylene chain with from x to y carbons in the alkylene chain.
- -Ci-ealkylene- may be selected from methylene, ethylene, propylene, butylene, pentylene, and hexylene, any one of which is optionally substituted.
- Alkoxy refers to a radical bonded through an oxygen atom of the formula -O- alkyl, where alkyl is an alkyl chain as defined above.
- alkenyl refers to a straight or branched hydrocarbon chain radical group consisting solely of carbon and hydrogen atoms, containing at least one carbon-carbon double bond, and preferably having from two to twelve carbon atoms (i.e., C2-C12 alkenyl).
- an alkenyl comprises two to eight carbon atoms (i.e., C2-C8 alkenyl).
- an alkenyl comprises two to six carbon atoms (i.e., C2-C6 alkenyl).
- an alkenyl comprises two to four carbon atoms (i.e., C2-C4 alkenyl).
- alkenyl is attached to the rest of the molecule by a single bond, for example, ethenyl (i.e., vinyl), prop-l-enyl (i.e., allyl), but-l-enyl, pent-l-enyl, penta- 1,4-dienyl, and the like.
- Alkynyl refers to a straight or branched hydrocarbon chain radical group consisting solely of carbon and hydrogen atoms, containing at least one carbon-carbon triple bond, and preferably having from two to twelve carbon atoms (z.e., C2-C12 alkynyl).
- an alkynyl comprises two to eight carbon atoms (z.e., C2-C8 alkynyl).
- an alkynyl comprises two to six carbon atoms (z.e., C2-C6 alkynyl).
- an alkynyl comprises two to four carbon atoms (z.e., C2-C4 alkynyl).
- the alkynyl is attached to the rest of the molecule by a single bond, for example, ethynyl, propynyl, butynyl, pentynyl, hexynyl, and the like.
- C x -yalkenyl and “C x -yalkynyl” refer to substituted or unsubstituted unsaturated aliphatic groups analogous in length and possible substitution to the alkyls described above, but that contain at least one double or triple bond, respectively.
- the term -C x.y alkenylene- refers to a substituted or unsubstituted alkenylene chain with from x to y carbons in the alkenylene chain.
- -C2-ealkenylene- may be selected from ethenylene, propenylene, butenylene, pentenylene, and hexenylene, any one of which is optionally substituted.
- An alkenylene chain may have one double bond or more than one double bond in the alkenylene chain.
- the term -C x.y alkynylene- refers to a substituted or unsubstituted alkynylene chain with from x to y carbons in the alkenylene chain.
- -C2-ealkenylene- may be selected from ethynylene, propynylene, butynylene, pentynylene, and hexynylene, any one of which is optionally substituted.
- An alkynylene chain may have one triple bond or more than one triple bond in the alkynylene chain.
- Alkylene or "alkylene chain” refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing no unsaturation, and preferably having from one to twelve carbon atoms, for example, methylene, ethylene, propylene, ⁇ -butylene, and the like.
- the alkylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond.
- the points of attachment of the alkylene chain to the rest of the molecule and to the radical group may be through any two carbons within the chain.
- an alkylene comprises one to ten carbon atoms (i.e., Ci-Cs alkylene). In certain embodiments, an alkylene comprises one to eight carbon atoms (i.e., Ci-Cs alkylene). In other embodiments, an alkylene comprises one to five carbon atoms (i.e., C1-C5 alkylene). In other embodiments, an alkylene comprises one to four carbon atoms (i.e., C1-C4 alkylene). In other embodiments, an alkylene comprises one to three carbon atoms (i.e., C1-C3 alkylene). In other embodiments, an alkylene comprises one to two carbon atoms (i.e., C1-C2 alkylene).
- an alkylene comprises one carbon atom (i.e., Ci alkylene). In other embodiments, an alkylene comprises five to eight carbon atoms (i.e., Cs-Cs alkylene). In other embodiments, an alkylene comprises two to five carbon atoms (i.e., C2-C5 alkylene). In other embodiments, an alkylene comprises three to five carbon atoms (i.e., C3-C5 alkylene).
- alkenylene or "alkenylene chain” refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing at least one carbon-carbon double bond, and preferably having from two to twelve carbon atoms.
- the alkenylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond.
- the points of attachment of the alkenylene chain to the rest of the molecule and to the radical group may be through any two carbons within the chain.
- an alkenylene comprises two to ten carbon atoms (i.e., C2-C10 alkenylene).
- an alkenylene comprises two to eight carbon atoms (i.e., C2-C8 alkenylene). In other embodiments, an alkenylene comprises two to five carbon atoms (i.e., C2-C5 alkenylene). In other embodiments, an alkenylene comprises two to four carbon atoms (i.e., C2-C4 alkenylene). In other embodiments, an alkenylene comprises two to three carbon atoms (i.e., C2-C3 alkenylene). In other embodiments, an alkenylene comprises two carbon atom (i.e., C2 alkenylene).
- an alkenylene comprises five to eight carbon atoms (i.e., Cs-Cs alkenylene). In other embodiments, an alkenylene comprises three to five carbon atoms (i.e., C3-C5 alkenylene).
- Alkynylene or "alkynylene chain” refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing at least one carbon-carbon triple bond, and preferably having from two to twelve carbon atoms.
- the alkynylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond.
- the points of attachment of the alkynylene chain to the rest of the molecule and to the radical group may be through any two carbons within the chain.
- an alkynylene comprises two to ten carbon atoms (i.e., C2-C10 alkynylene).
- an alkynylene comprises two to eight carbon atoms (i.e., C2-C8 alkynylene). In other embodiments, an alkynylene comprises two to five carbon atoms (i.e., C2-C5 alkynylene). In other embodiments, an alkynylene comprises two to four carbon atoms (i.e., C2-C4 alkynylene). In other embodiments, an alkynylene comprises two to three carbon atoms (i.e., C2-C3 alkynylene). In other embodiments, an alkynylene comprises two carbon atom (i.e., C2 alkynylene).
- an alkynylene comprises five to eight carbon atoms (i.e., C5-C8 alkynylene). In other embodiments, an alkynylene comprises three to five carbon atoms (i.e., C3-C5 alkynylene).
- Aryl refers to a radical derived from an aromatic monocyclic or aromatic multicyclic hydrocarbon ring system by removing a hydrogen atom from a ring carbon atom.
- the aromatic monocyclic or aromatic multicyclic hydrocarbon ring system contains only hydrogen and carbon and from five to eighteen carbon atoms, where at least one of the rings in the ring system is aromatic, i.e., it contains a cyclic, delocalized (4n+2) ⁇ -electron system in accordance with the Hiickel theory.
- the ring system from which aryl groups are derived include, but are not limited to, groups such as benzene, fluorene, indane, indene, tetralin and naphthalene.
- “Aralkyl” refers to a radical of the formula -R c -aryl where R c is an alkylene chain as defined above, for example, methylene, ethylene, and the like.
- alkenyl refers to a radical of the formula -R d -aryl where R d is an alkenylene chain as defined above.
- alkynyl refers to a radical of the formula -R e -aryl, where R e is an alkynylene chain as defined above.
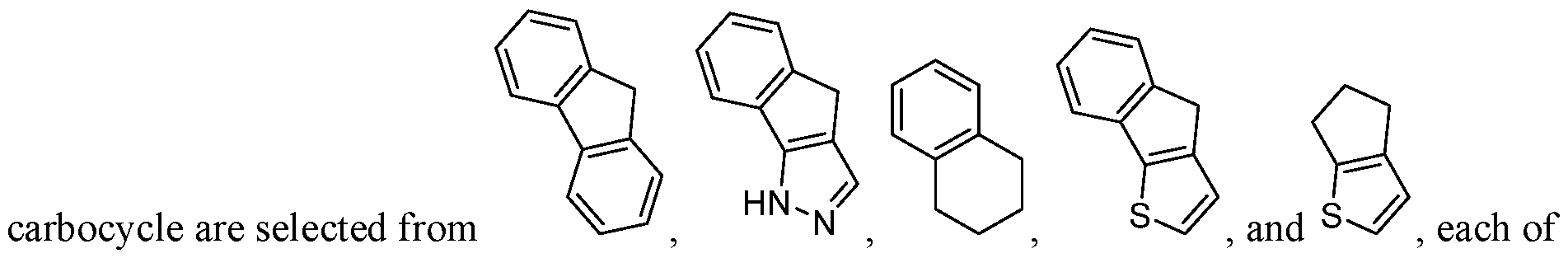
- Carbocycle refers to a saturated, unsaturated or aromatic rings in which each atom of the ring is carbon.
- Carbocycle may include 3- to 10-membered monocyclic rings, 6- to 12- membered bicyclic rings, and 6- to 12-membered bridged rings.
- Each ring of a bicyclic carbocycle may be selected from saturated, unsaturated, and aromatic rings.
- An aromatic ring e.g., phenyl, may be fused to a saturated or unsaturated ring, e.g., cyclohexane, cyclopentane, or cyclohexene.
- unsaturated carbocycle refers to carbocycles with at least one degree of unsaturation and excluding aromatic carbocycles.
- unsaturated carbocycles include cyclohexadiene, cyclohexene, and cyclopentene.
- Cycloalkyl refers to a fully saturated monocyclic or polycyclic hydrocarbon radical consisting solely of carbon and hydrogen atoms, which includes fused or bridged ring systems, and preferably having from three to twelve carbon atoms. In certain embodiments, a cycloalkyl comprises three to ten carbon atoms. In other embodiments, a cycloalkyl comprises five to seven carbon atoms. The cycloalkyl may be attached to the rest of the molecule by a single bond.
- Examples of monocyclic cycloalkyls include, e.g., cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, and cyclooctyl.
- Polycyclic cycloalkyl radicals include, for example, adamantyl, norbornyl (i.e., bicyclo[2.2.1]heptanyl), norbomenyl, decalinyl, 7,7-dimethyl-bicyclo[2.2.1]heptanyl, and the like.
- Cycloalkenyl refers to an unsaturated non-aromatic monocyclic or polycyclic hydrocarbon radical consisting solely of carbon and hydrogen atoms, which includes fused or bridged ring systems, preferably having from three to twelve carbon atoms and comprising at least one double bond.
- a cycloalkenyl comprises three to ten carbon atoms.
- a cycloalkenyl comprises five to seven carbon atoms.
- the cycloalkenyl may be attached to the rest of the molecule by a single bond. Examples of monocyclic cycloalkenyls includes, e.g., cyclopentenyl, cyclohexenyl, cycloheptenyl, and cyclooctenyl.
- Cycloalkylalkyl refers to a radical of the formula -R c -cycloalkyl where R c is an alkylene chain as described above.
- Cycloalkylalkoxy refers to a radical bonded through an oxygen atom of the formula -O-R c -cycloalkyl where R c is an alkylene chain as described above.
- Halo or "halogen” refers to halogen substituents such as bromo, chloro, fluoro and iodo substituents.
- haloalkyl or “haloalkane” refers to an alkyl radical, as defined above, that is substituted by one or more halogen radicals, for example, trifluoromethyl, di chloromethyl, bromomethyl, 2,2,2-trifluoroethyl, l-fluoromethyl-2-fluoroethyl, and the like.
- the alkyl part of the fluoroalkyl radical is optionally further substituted.
- halogen substituted alkanes include halomethane (e.g., chloromethane, bromomethane, fluoromethane, iodomethane), di-and trihalomethane (e.g., tri chloromethane, tribromomethane, trifluoromethane, triiodomethane), 1-haloethane, 2- haloethane, 1,2-dihaloethane, 1-halopropane, 2-halopropane, 3-halopropane, 1,2-dihalopropane, 1,3-dihalopropane, 2,3-dihalopropane, 1,2,3-trihalopropane, and any other suitable combinations of alkanes (or substituted alkanes) and halogens (e.g., Cl, Br, F, I, etc.).
- each halogen may be independently selected
- Fluoroalkyl refers to an alkyl radical, as defined above, that is substituted by one or more fluoro radicals, for example, trifluoromethyl, difluoromethyl, fluoromethyl, 2,2,2-trifluoroethyl, l-fluoromethyl-2-fluoroethyl, and the like.
- Aminoalkyl refers to an alkyl radical, as defined above, that is substituted by one or more amine radicals, for example, propan-2-amine, butane- 1,2-di amine, pentane- 1,2,4- triamine and the like.
- Hydroxyalkyl refers to an alkyl radical, as defined above, that is substituted by one or more hydroxy radicals, for example, propan-l-ol, butane- 1,4-diol, pentane- 1, 2, 4-triol, and the like.
- Alkoxyalkyl refers to an alkyl radical, as defined above, that is substituted by one or more alkoxy radicals, for example, methoxymethane, 1,3 -dimethoxybutane, 1- methoxypropane, 2-ethoxypentane, and the like.
- Cyanoalkyl refers to an alkyl radical, as defined above, that is substituted by one or more cyano radicals, for example, acetonitrile, 2-ethyl-3- methylsuccinonitrile, butyronitrile, and the like.
- Heterocycle refers to a saturated, unsaturated or aromatic ring comprising one or more heteroatoms. Exemplary heteroatoms include N, O, Si, P, B, and S atoms. The heterocycle may be attached to the rest of the molecule through any atom of the heterocycle, valence permitting, such as a carbon or nitrogen atom of the heterocycle.
- Heterocycles include 3- to 10-membered monocyclic rings, 6- to 12-membered bicyclic rings, and 6- to 12-membered bridged rings.
- a bicyclic heterocycle includes any combination of saturated, unsaturated and aromatic bicyclic rings, as valence permits.
- an aromatic ring e.g., pyridyl
- a bicyclic heterocycle includes any combination of ring sizes such as 4-5 fused ring systems, 5-5 fused ring systems, 5- 6 fused ring systems, 6-6 fused ring systems, 5-7 fused ring systems, 6-7 fused ring systems, 5-8 fused ring systems, and 6-8 fused ring systems.
- Bicyclic heterocycles may be fused, bridged, or spiro-ring systems.
- a spiro-ring system may be referred as a “spiroheterocycle”, “spiro heterocycle”, or “spiro-heterocycle”.
- spiro-heterocycles, spiro heterocycles, or spiroheterocycles have at least two molecular rings with only one common atom.
- the spiroheterocycle, spiro heterocycle, or spiroheterocycle comprises one or more heteroatoms.
- Heterocyclene refers to a divalent heterocycle linking the rest of the molecule to a radical group.
- Heteroaryl or “aromatic heterocycle” refers to a radical derived from a heteroaromatic ring radical that comprises one to eleven carbon atoms and at least one heteroatom wherein each heteroatom may be selected from N, O, and S.
- the heteroaryl ring may be selected from monocyclic or bicyclic and fused or bridged ring systems rings wherein at least one of the rings in the ring system is aromatic, z.e., it contains a cyclic, delocalized (4n+2) ⁇ -electron system in accordance with the Hiickel theory.
- the heteroatom(s) in the heteroaryl radical may be optionally oxidized.
- heteroaryl may be attached to the rest of the molecule through any atom of the heteroaryl, valence permitting, such as a carbon or nitrogen atom of the heteroaryl.
- heteroaryls include, but are not limited to, pyridine, pyrimidine, oxazole, furan, pyran, thiophene, isoxazole, benzimidazole, benzthiazole, and imidazopyridine.
- An “X-membered heteroaryl” refers to the number of endocylic atoms, i.e., X, in the ring.
- a 5-membered heteroaryl ring or 5-membered aromatic heterocycle has 5 endocyclic atoms, e.g., triazole, oxazole, thiophene, etc.
- the term “unsaturated heterocycle” refers to heterocycles with at least one degree of unsaturation and excluding aromatic heterocycles.
- unsaturated heterocycles include dihydropyrrole, dihydrofuran, oxazoline, pyrazoline, and dihydropyridine.
- Heterocycles may be optionally substituted by one or more substituents such as those substituents described herein.
- substituted refers to moieties having substituents replacing a hydrogen on one or more carbons or substitutable heteroatoms, e.g., NH, of the structure.
- substitution or “substituted with” includes the implicit proviso that such substitution is in accordance with permitted valence of the substituted atom and the substituent, and that the substitution results in a stable compound, z.e., a compound which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, etc.
- substituted refers to moieties having substituents replacing two hydrogen atoms on the same carbon atom, such as substituting the two hydrogen atoms on a single carbon with an oxo, imino or thioxo group.
- substituted is contemplated to include all permissible substituents of organic compounds.
- the permissible substituents include acyclic and cyclic, branched and unbranched, carbocyclic and heterocyclic, aromatic and non-aromatic substituents of organic compounds.
- the permissible substituents can be one or more and the same or different for appropriate organic compounds.
- the heteroatoms such as nitrogen may have hydrogen substituents and/or any permissible substituents of organic compounds described herein which satisfy the valences of the heteroatoms.
- each R b is independently selected from a direct bond or a straight or branched alkylene, alkenylene, or alkynylene chain, and each R c is a straight or branched alkylene, alkenylene or alkynylene chain.
- electrophile or “electrophilic moiety” is any moiety capable of reacting with a nucleophile (e.g., a moiety having a lone pair of electrons, a negative charge, a partial negative charge and/or an excess of electrons, for example an — SH group).
- Electrophiles typically are electron poor or comprise atoms which are electron poor.
- an electrophile contains a positive charge or partial positive charge, has a resonance structure which contains a positive charge or partial positive charge, or is a moiety in which delocalization or polarization of electrons results in one or more atoms which contains a positive charge or partial positive charge.
- an electrophile comprises a conjugated double bond, for example an a,P-unsaturated carbonyl or a,P-unsaturated thiocarbonyl compound.
- the term “optional” or “optionally” means that the subsequently described event of circumstances may or may not occur, and that the description includes instances where the event or circumstance occurs and instances in which it does not.
- “optionally substituted aryl” means that the aryl group may or may not be substituted and that the description includes both substituted aryl groups and aryl groups having no substitution.
- salt or “pharmaceutically acceptable salt” refers to salts derived from a variety of organic and inorganic counter ions well known in the art.
- Pharmaceutically acceptable acid addition salts can be formed with inorganic acids and organic acids.
- Inorganic acids from which salts can be derived include, for example, hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, and the like.
- Organic acids from which salts can be derived include, for example, acetic acid, propionic acid, glycolic acid, pyruvic acid, oxalic acid, maleic acid, malonic acid, succinic acid, fumaric acid, tartaric acid, citric acid, benzoic acid, cinnamic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, /?-toluenesulfonic acid, salicylic acid, and the like.
- Pharmaceutically acceptable base addition salts can be formed with inorganic and organic bases.
- Inorganic bases from which salts can be derived include, for example, sodium, potassium, lithium, ammonium, calcium, magnesium, iron, zinc, copper, manganese, aluminum, and the like.
- Organic bases from which salts can be derived include, for example, primary, secondary, and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines, basic ion exchange resins, and the like, specifically such as isopropylamine, trimethylamine, diethylamine, triethylamine, tripropylamine, and ethanolamine.
- the pharmaceutically acceptable base addition salt is chosen from ammonium, potassium, sodium, calcium, and magnesium salts.
- parenteral administration and “administered parenterally” as used herein means modes of administration other than enteral and topical administration, usually by injection, and includes, without limitation, intravenous, intramuscular, intraarterial, intrathecal, intracapsular, intraorbital, intracardiac, intradermal, intraperitoneal, transtracheal, subcutaneous, subcuticular, intraarticular, subcapsular, subarachnoid, intraspinal and intrasternal injection and infusion.
- phrases “pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
- phrases “pharmaceutically acceptable excipient” or “pharmaceutically acceptable carrier” as used herein means a pharmaceutically acceptable material, composition or vehicle, such as a liquid or solid filler, diluent, excipient, solvent or encapsulating material. Each carrier must be “acceptable” in the sense of being compatible with the other ingredients of the formulation and not injurious to the patient.
- materials which can serve as pharmaceutically acceptable carriers include: (1) sugars, such as lactose, glucose and sucrose; (2) starches, such as com starch and potato starch; (3) cellulose, and its derivatives, such as sodium carboxymethyl cellulose, ethyl cellulose and cellulose acetate; (4) powdered tragacanth; (5) malt; (6) gelatin; (7) talc; (8) excipients, such as cocoa butter and suppository waxes; (9) oils, such as peanut oil, cottonseed oil, safflower oil, sesame oil, olive oil, com oil and soybean oil; (10) glycols, such as propylene glycol; (11) polyols, such as glycerin, sorbitol, mannitol and polyethylene glycol; (12) esters, such as ethyl oleate and ethyl laurate; (13) agar; (14) buffering agents, such as magnesium hydroxide and aluminum hydrox
- the term “prevent” or “preventing” as related to a disease or disorder may refer to a compound that, in a statistical sample, reduces the occurrence of the disorder or condition in the treated sample relative to an untreated control sample, or delays the onset or reduces the severity of one or more symptoms of the disorder or condition relative to the untreated control sample.
- treat may include alleviating, abating or ameliorating a disease or condition symptoms, preventing additional symptoms, ameliorating or preventing the underlying causes of symptoms, inhibiting the disease or condition, e.g., arresting the development of the disease or condition, relieving the disease or condition, causing regression of the disease or condition, relieving a condition caused by the disease or condition, or stopping the symptoms of the disease or condition either prophylactically and/or therapeutically.
- G12 mutants refers to other oncogenic alleles of KRAS at amino acid position 12 (ie. G12X).
- KRas G12D-associated cancer refers to cancers associated with or mediated by or having a KRas G12D mutation.
- KRas G12V-associated cancer refers to cancers associated with or mediated by or having a KRas G12V mutation.
- KRas wildtype-associated cancer refers to cancers associated with or mediated by or having a KRas wildtype.
- immunomodulator inhibitor refers to an agent that modifies, or modulates, the immune system to help a subject respond to a disease or disorder.
- PD-1 inhibitor refers to an agent that is capable of negatively modulating or inhibiting all or a portion of the PD-1 axis signaling activity and include agents that block PD- 1.
- PD-L1 inhibitor refers to an agent that is capable of negatively modulating or inhibiting all or a portion of the PD-L1 axis signaling activity and include agents that block PD-L1.
- PD-1 binding antagonist is a molecule that decreases, blocks, inhibits, abrogates or interferes with signal transduction resulting from the interaction of PD-1 with one or more of its binding partners, such as PD-L1 and/or PD-L2.
- PD-L1 binding antagonist is a molecule that decreases, blocks, inhibits, abrogates or interferes with signal transduction resulting from the interaction of PD-L1 with either one or more of its binding partners, such as PD-1 and/or B7-1.
- biosimilar means an antibody or antigen-binding fragment that has the same primary amino acid sequence as compared to a reference antibody (e.g., nivolumab or pembrolizumab) and optionally, may have detectable differences in post-translation modifications (e.g., glycosylation and/or phosphorylation) as compared to the reference antibody (e.g., a different glycoform).
- a reference antibody e.g., nivolumab or pembrolizumab
- post-translation modifications e.g., glycosylation and/or phosphorylation
- the terms "subject,” “individual,” and “patient” may be used interchangeably and refer to humans, as well as non-human mammals (e.g., non-human primates, canines, equines, felines, porcines, bovines, ungulates, lagomorphs, and the like).
- the subject can be a human (e.g., adult male, adult female, adolescent male, adolescent female, male child, female child) under the care of a physician or other health worker in a hospital, as an outpatient, or other clinical context.
- the subject may not be under the care or prescription of a physician or other health worker.
- a subject in need thereof' refers to a subject, as described infra, that suffers from, or is at risk for, a pathology to be prophylactically or therapeutically treated with a compound or salt described herein.
- determining means determining if an element is present or not (for example, detection). These terms can include quantitative, qualitative or quantitative and qualitative determinations. Assessing can be relative or absolute. “Detecting the presence of’ can include determining the amount of something present in addition to determining whether it is present or absent depending on the context.
- administer are defined as providing a composition to a subject via a route known in the art, including but not limited to intravenous, intraarterial, oral, parenteral, buccal, topical, transdermal, rectal, intramuscular, subcutaneous, intraosseous, transmucosal, or intraperitoneal routes of administration.
- oral routes of administering a composition can be used.
- administer should be understood to mean providing a compound of the disclosure or a prodrug of a compound of the disclosure to the individual in need.
- the term “effective amount” or “therapeutically effective amount” refers to that amount of a compound or salt described herein that is sufficient to effect the intended application including but not limited to disease treatment, as defined below.
- the therapeutically effective amount may vary depending upon the intended application (in vitro or in vivo), or the subject and disease condition being treated, e.g., the weight and age of the subject, the severity of the disease condition, the manner of administration and the like, which can readily be determined by one of ordinary skill in the art.
- the term can also apply to a dose that can induce a particular response in target cells, e.g., reduction of proliferation or down regulation of activity of a target protein.
- the specific dose can vary depending on the particular compounds chosen, the dosing regimen to be followed, whether it is administered in combination with other compounds, timing of administration, the tissue to which it is administered, and the physical delivery system in which it is carried.
- a "therapeutically effective amount of a combination" of two compounds is an amount that together synergistically increases the activity of the combination in comparison to the therapeutically effective amount of each compound in the combination, i.e., more than merely additive.
- “synergy,” “synergetic,” “synergism,” or “synergistic effect” refer to two or more compounds or compositions, that individually produce an effect, however, together produce a combined effect that is greater than their individual effects.
- the term “about” or “approximately” can mean within an acceptable error range for the particular value as determined by one of ordinary skill in the art, which will depend in part on how the value is measured or determined, i.e., the limitations of the measurement system. For example, “about” can mean within 1 or more than 1 standard deviation, per the practice in the art. Alternatively, “about” can mean a range of up to 20%, up to 15%, up to 10%, up to 5%, or up to 1% of a given value.
- kits for treating a disease or disorder in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of i) an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, ii) and a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, ii) and a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
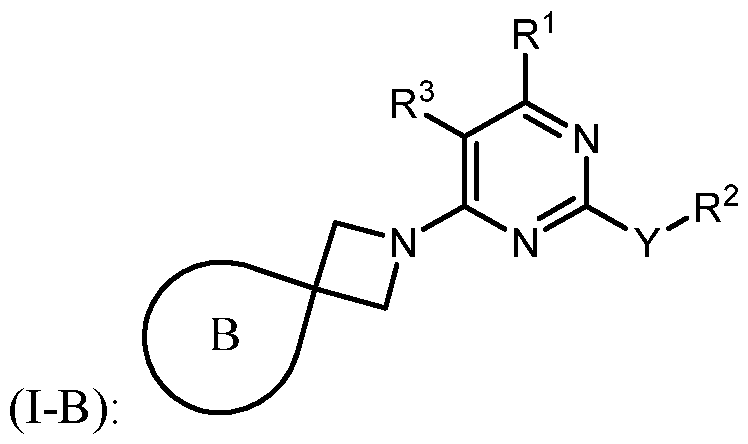
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, ii) and a compound of Formula (I- A), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, ii) and a compound of Formula (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating a disease or disorder in a subject in need thereof comprising administering to the subject a therapeutically effective amount of: i) an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, ii) and a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the immunomodulator inhibitor is selected from a PD-1 inhibitor, a PD-Ll inhibitor, and a CTLA-4 inhibitor. In some cases, the immunomodulator inhibitor is a PD-1 inhibitor. In some cases, the immunomodulator inhibitor is a PD-L1 inhibitor. In some cases, the immunomodulator inhibitor is a CTLA-4 inhibitor. In some embodiments, the immunomodulator inhibitor is selected from a PD-1/PD-L1 checkpoint inhibitor.
- the immunomodulator inhibitor is immune checkpoint inhibitor.
- the immunomodulator inhibitor is pembrolizumab.
- the immunomodulator inhibitor is relatlimab.
- programmed death protein 1 is an immunoinhibitory receptor that is primarily expressed on activated T and B cells.
- PD-1 is a 55 kDa type I transmembrane protein that is part of the Ig gene superfamily (Agata et al. (1996) Int Immunol 8:765-72).
- PD-1 contains a membrane proximal immunoreceptor tyrosine inhibitory motif (ITIM) and a membrane distal tyrosine-based switch motif (ITSM).
- ITIM immunoreceptor tyrosine inhibitory motif
- ITSM membrane distal tyrosine-based switch motif
- Two ligands that bind to PD-1 have been identified, PD-L1 and PD-L2, that have been shown to downregulate T cell activation upon binding to PD-1 (Freeman et al.
- PD-L1 is a ligand for PD-1 and is abundant in a variety of human cancers (Dong et al. (2002) Nat. Med. 8:787-9). In some cases, the interaction between PD-1 and PD-L1 results in a decrease in tumor infiltrating lymphocytes, a decrease in T-cell receptor mediated proliferation, and immune evasion by the cancerous cells (Dong et al. (2003) J. Mol. Med. 81 :281-7).
- immune suppression can be reversed by inhibiting the local interaction of PD-1 with PD-L1, and the effect is additive when the interaction of PD-1 with PD- L2 is blocked as well.
- disruption of the PD-1/PD-L1 interaction has been shown to increase T cell proliferation and cytokine production and block progression of the cell cycle.
- PD-L1 is upregulated in many cancers and contributes to evasion of the host immune system, blocking the interaction between PD-1 and PD-L1 has garnered the attention of the pharmaceutical industry leading to a new break-through class of immune checkpoint therapies for a wide range of cancers.
- the PD-1/PD-L1 pathway is a well-validated target for the development of antibody therapeutics for cancer treatment and several anti -PD-1 and anti-PD-Ll antibodies have undergone human clinical trials for a wide- variety of cancers including NSCLC, renal cell carcinoma, melanoma, head and neck squamous cancer, ureothelial cancer, hepatocellular carcinoma, and other cancers.
- anti-PD-1 antibodies include nivolumab (Opdivo®), pembrolizumab (Keytruda®), cemiplimab (Libtayo®) and tislelizumab, and biosimilars thereof.
- anti-PD-Ll antibodies include atezolizumab (Tecentriq®), avelumab (Bavencio®), and durvalumab (Imfinzi®), and biosimilars thereof.
- agents that disrupt PD-1/PD-L1 signaling axis are well known to those skilled in the art and agents that disrupt PD-1/PD-L1 signaling axis may be obtained from a wide-variety of commercial suppliers, in forms suitable for both research or approved human clinical use.
- suitable agents that disrupt PD-1/PD-L1 signaling for use in the compositions and methods disclosed herein and methods for preparing such agents, and diagnostic and efficacy markers useful for monitoring treatment are disclosed in US Patent Application Publication Nos: 20180327848; 20180237524; 20180148790; 20180111996; 20160305947; 20160304969;
- kits for treating a disease or disorder in a subject in need thereof comprising administering to the subject a combination of: i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating a disease or disorder in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I-A), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating a disease or disorder in a subject in need thereof comprising administering to the subject a therapeutically effective amount of: i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- a PD-1 inhibitor is selected from a PD-1 binding antagonist.
- the PD-1 binding antagonist is selected from anti-PD-1 antibodies, antigen binding fragments thereof, immunoadhesins, aptamers, fusion proteins, and oligopeptides.
- the PD-1 binding antagonist is an anti-PD-1 antibody.
- the PD-1 inhibitor is a molecule that inhibits the binding of PD-1 to its binding partners. In some cases, the PD-1 inhibitor inhibits the binding of PD-1 to PD-L1 and/or PD-L2. In some cases, PD-1 inhibitors include anti-PD-1 antibodies, antigen binding fragments thereof, immunoadhesins, fusion proteins, oligopeptides and other molecules that decrease, block, inhibit, abrogate or interfere with signal transduction resulting from the interaction of PD-1 with PD-L1 and/or PD-L2.
- a PD-1 inhibitor reduces the negative co-stimulatory signal mediated by or through cell surface proteins expressed on T lymphocytes mediated signaling through PD-1 so as render a dysfunctional T-cell less non- dysfunctional.
- the PD-1 inhibitor is an anti-PD-1 antibody.
- the PD- 1 antibody is pembrolizumab, or a biosimilar thereof.
- the PD-1 antibody is cemiplimab, or a biosimilar thereof.
- the PD-1 antibody is tislelizumab, or a biosimilar thereof.
- kits for treating a disease or disorder in a subject in need thereof comprising administering to the subject a combination of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating a disease or disorder in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I- A), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I-B), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
- kits for treating a disease or disorder in a subject in need thereof comprising administering to the subject a therapeutically effective amount of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- a PD-L1 inhibitor is selected from a PD-L1 binding antagonist.
- the PD-L1 binding antagonist is selected from an anti-PD-Ll antibody, antigen binding fragments thereof, immunoadhesins, aptamers, fusion proteins, and oligopeptides.
- the PD-L1 binding antagonist is an anti-PD-Ll antibody.
- a PD-L1 inhibitor is a molecule that inhibits the binding of PD-L1 to its binding partners.
- the PD-L1 inhibitor inhibits binding of PD-L1 to PD-1 and/or B7-1.
- the PD-L1 inhibitors include anti-PD-Ll antibodies, antigen binding fragments thereof, immunoadhesins, fusion proteins, oligopeptides and other molecules that decrease, block, inhibit, abrogate or interfere with signal transduction resulting from the interaction of PD-L1 with one or more of its binding partners, such as PD-1 and/or B7-1.
- a PD-L1 inhibitor reduces the negative co-stimulatory signal mediated by or through cell surface proteins expressed on T lymphocytes mediated signaling through PD-L1 so as render a dysfunctional T-cell less non-dysfunctional.
- a PD-L1 inhibitor is an anti-PD-Ll antibody.
- an anti- PD-L1 antibody is avelumab or a biosimilar thereof.
- an anti-PD-Ll antibody is atezolizumab or a biosimilar thereof.
- an anti-PD- Ll antibody is durvalumab or a biosimilar thereof.
- an anti-PD-Ll antibody is BMS-936559 (MDX-1105) or a biosimilar thereof.
- kits for treating a disease or disorder in a subject in need thereof comprising administering to the subject a combination of: i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating a disease or disorder in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating a disease or disorder in a subject in need thereof comprising administering to the subject a therapeutically effective amount of: i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I- A), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
- kits for treating cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of: i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I-B), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
- the CTLA-4 inhibitor is selected from Ipilimumab. KRAS Inhibitors
- the compounds and salts may be used in combination with at least one other inhibitor (e.g., immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor).
- the compounds and salts e.g., a compound of Formula (I), (I- A), or (I-B)
- may be used in combination with one other inhibitor e.g., immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor.
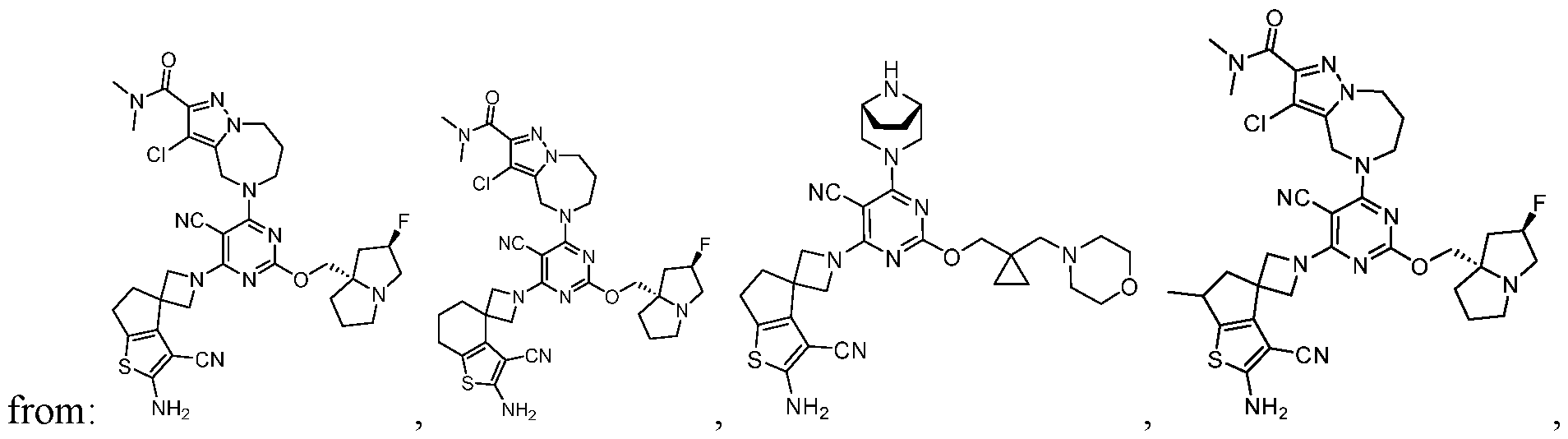
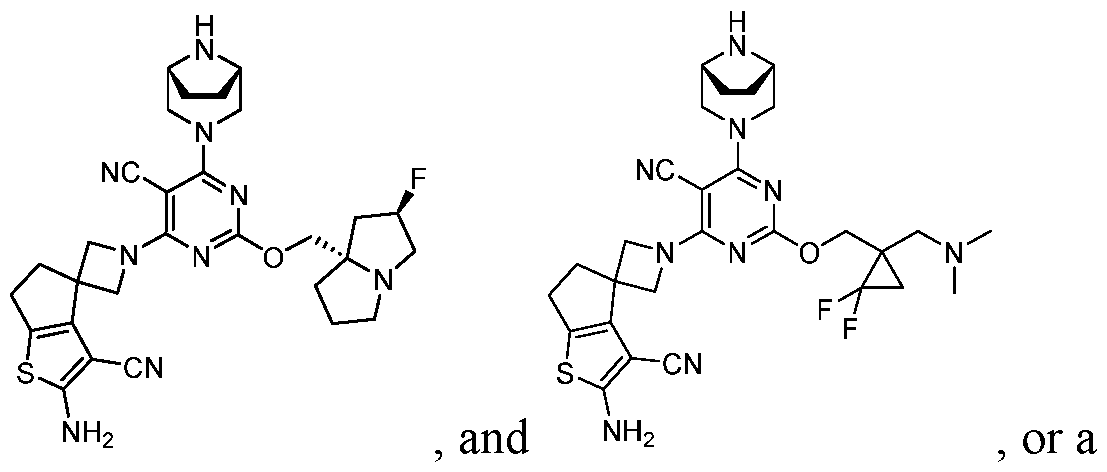
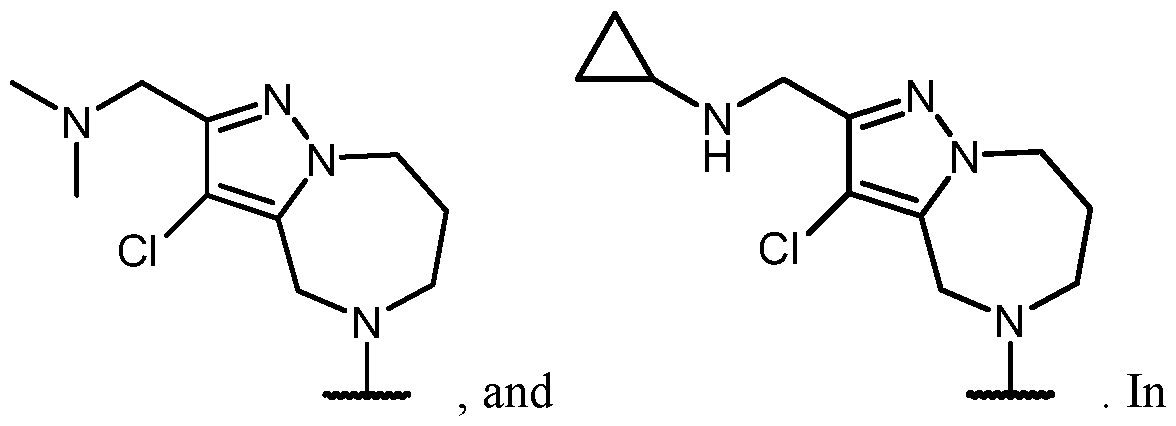
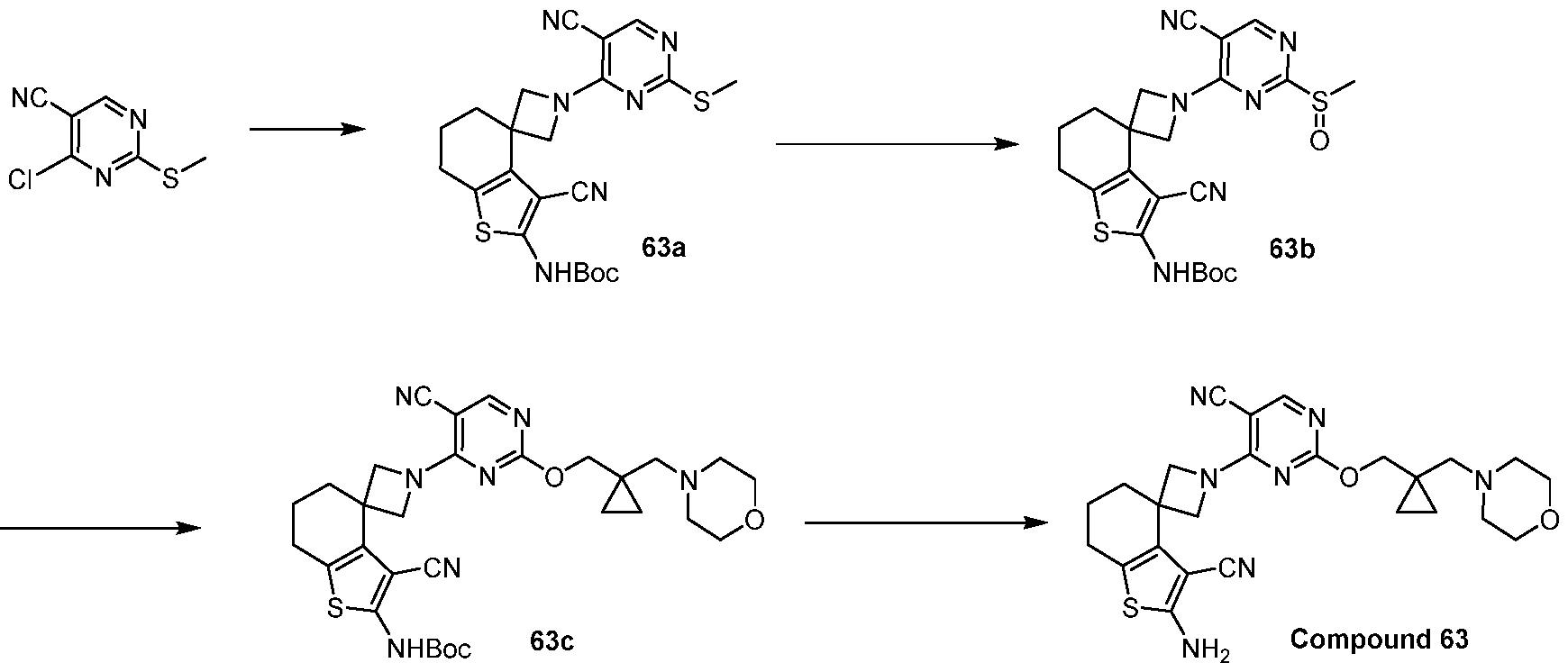
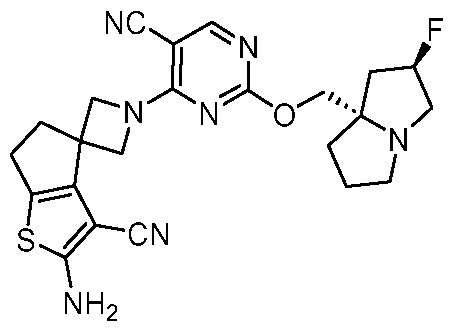
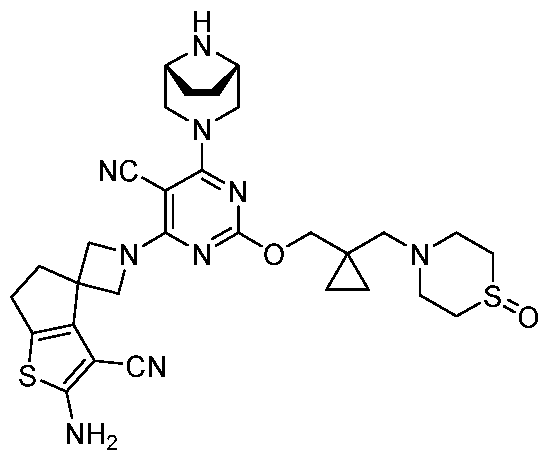
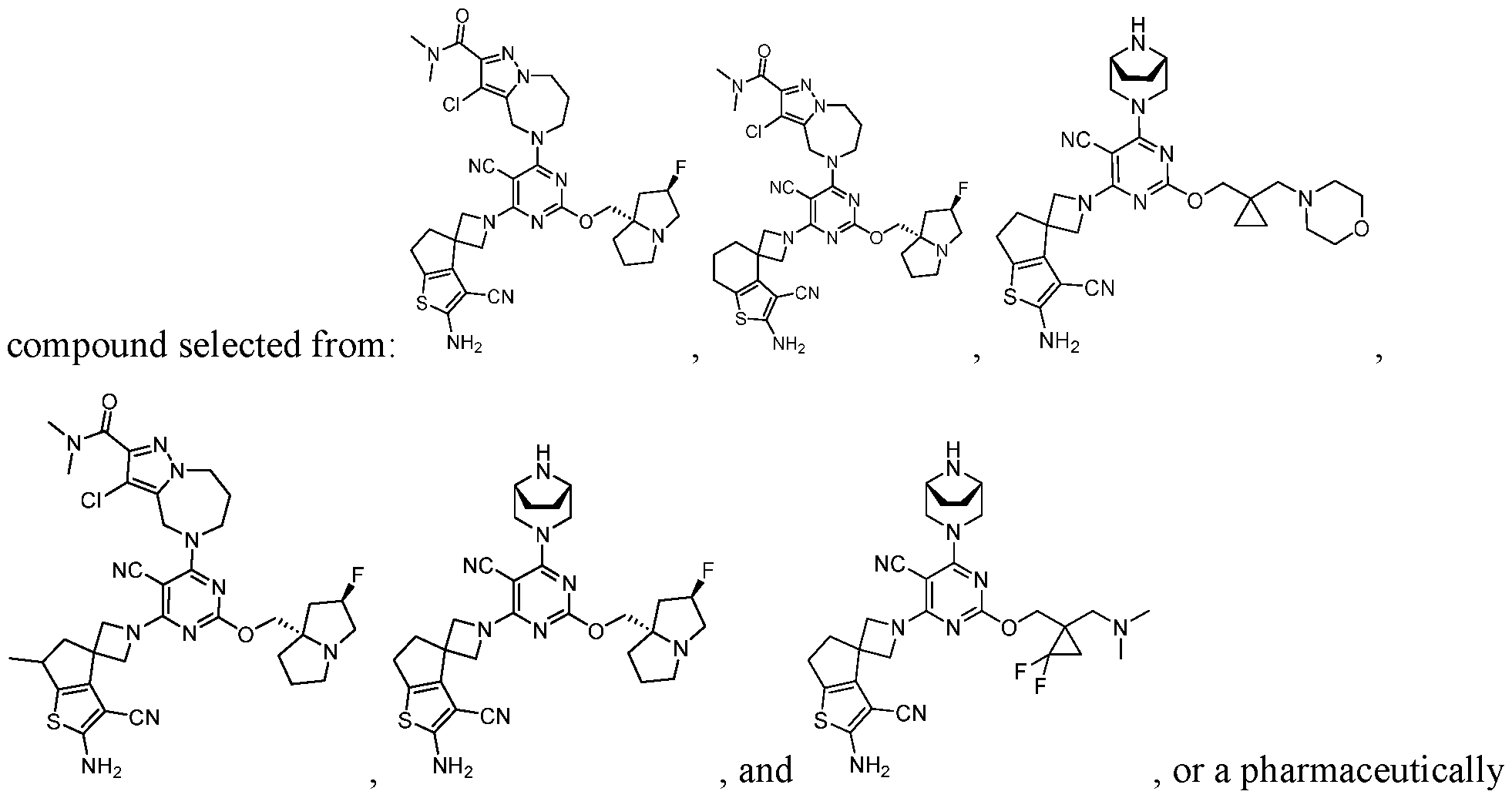
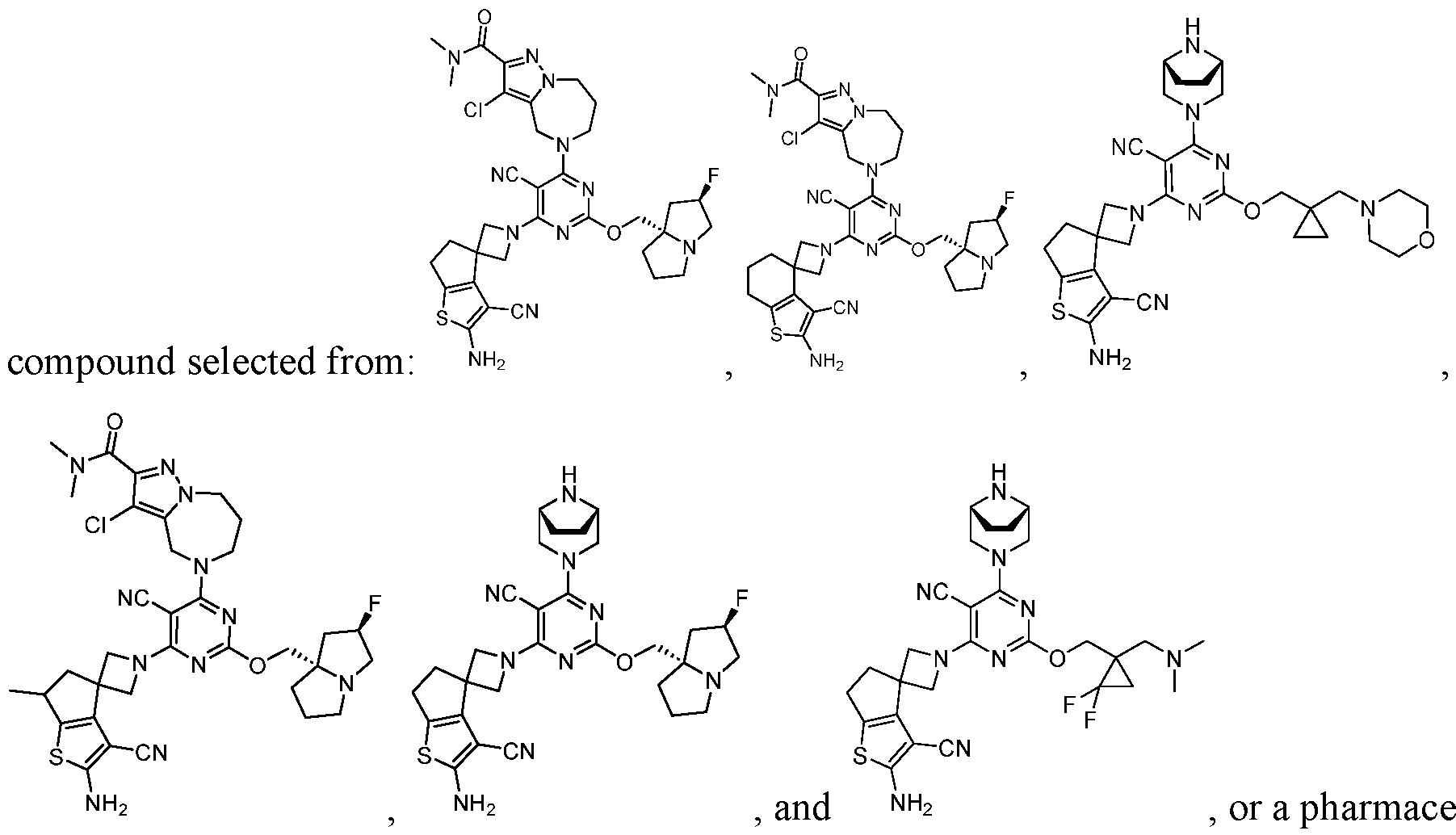
- a compound of Formula (I), (LA), or (LB) may be used in the methods of the disclosure.
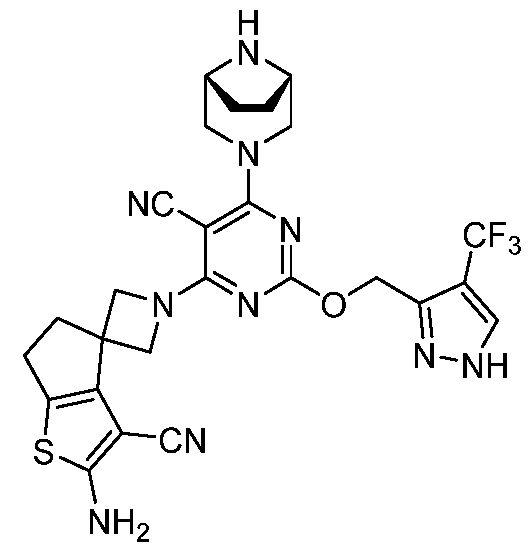
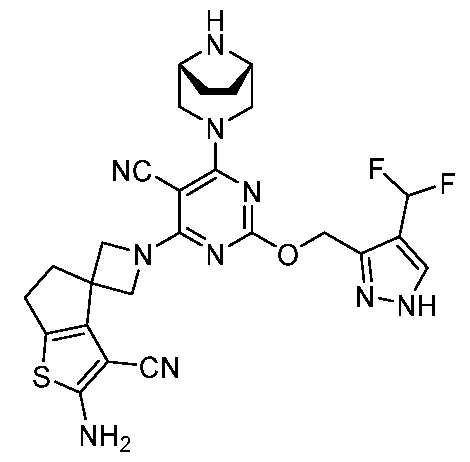
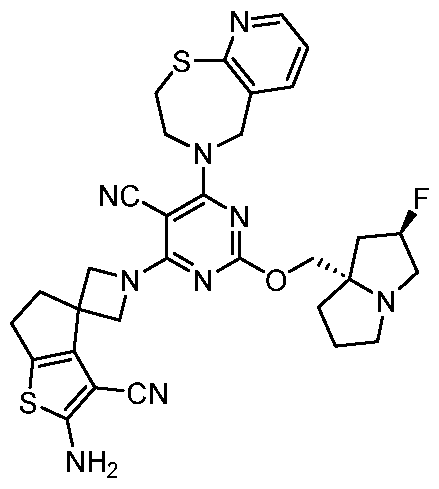
- a compound of Formula (I), (LA), or (LB) may be referred to as a KRAS inhibitor.
- a compound of Formula (I), (LA), or (LB) may be referred to as a KRAS inhibitor.
- Y is selected from a bond, -0-, -S-, and -N(R 5 )-;
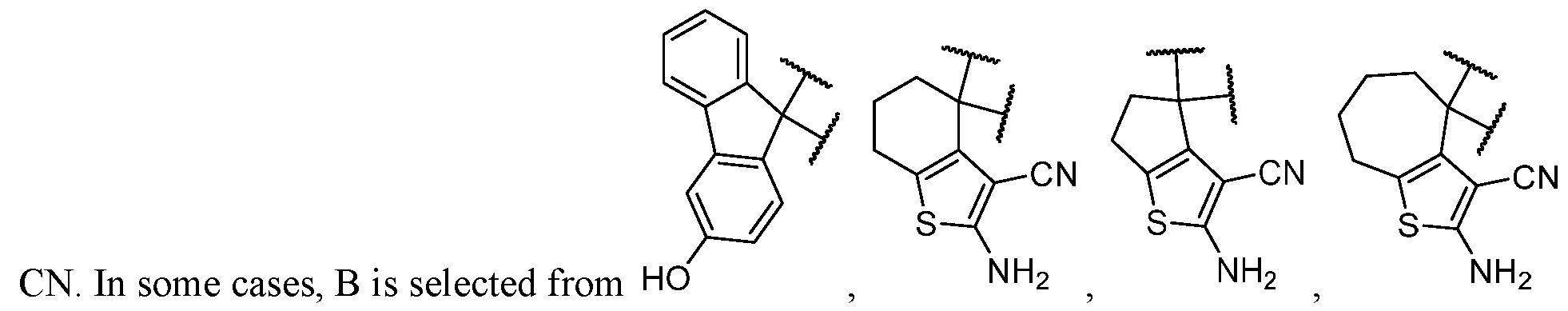
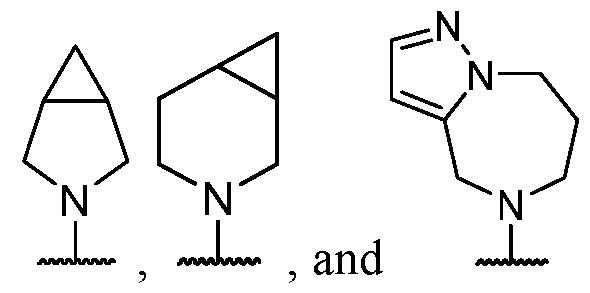
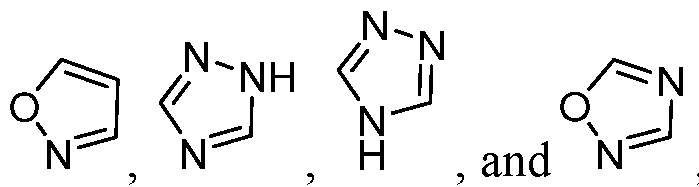
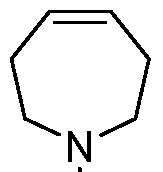
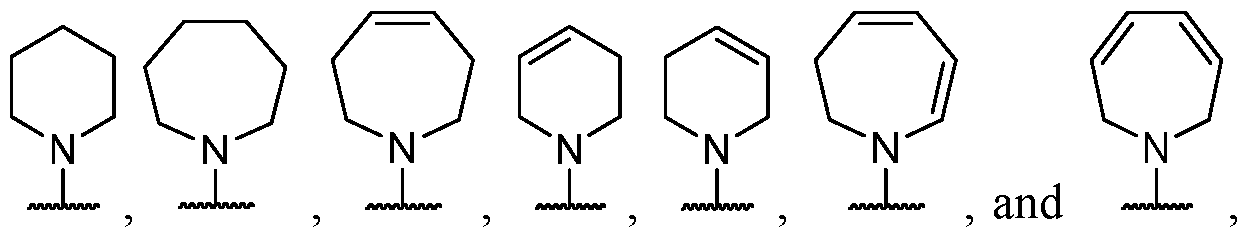
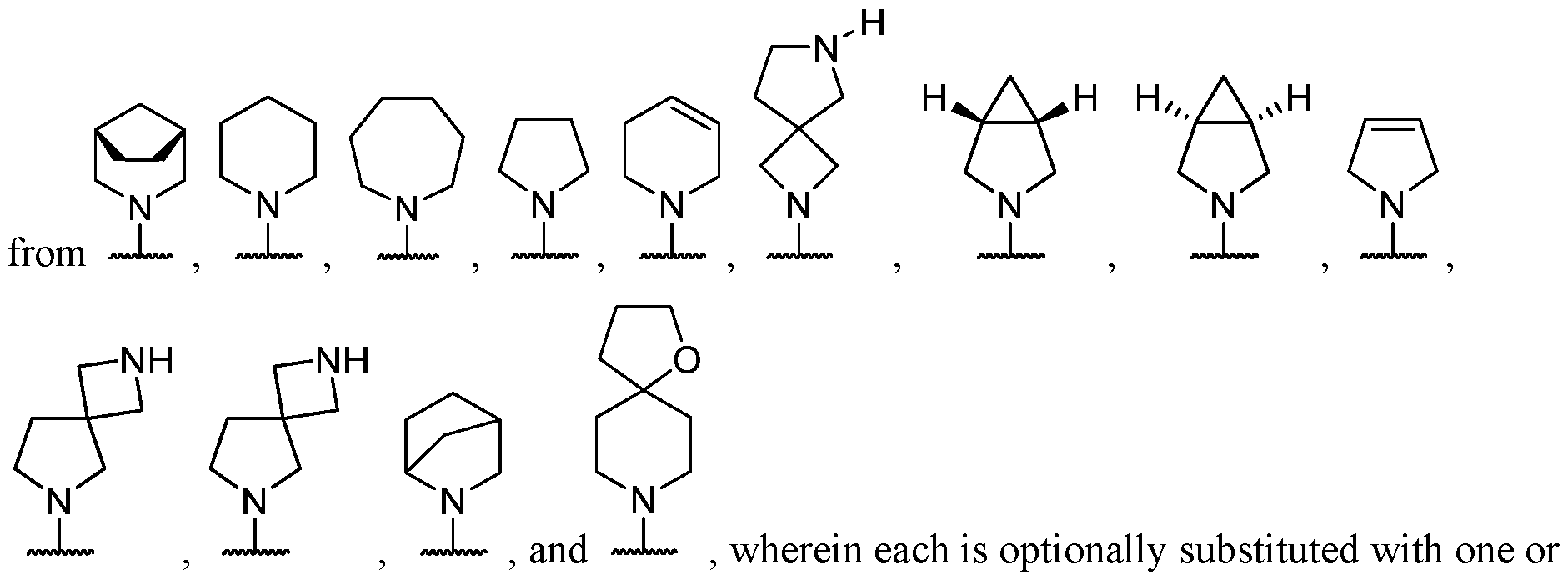
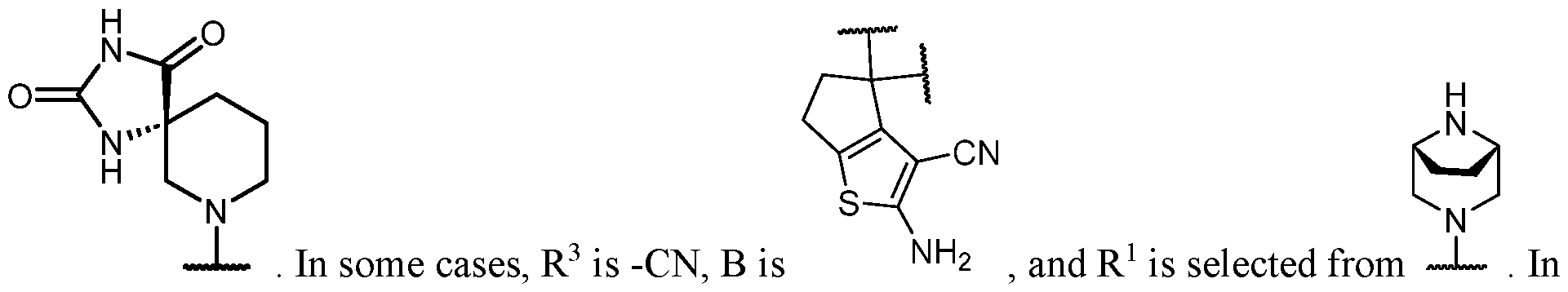
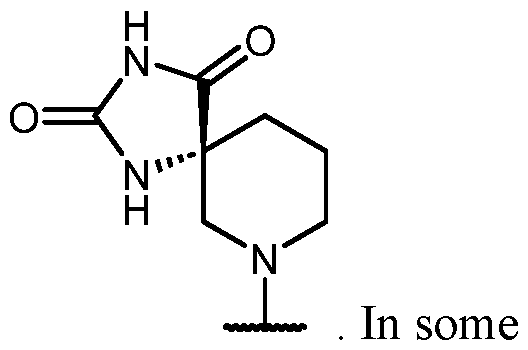
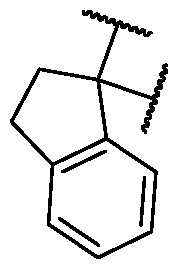
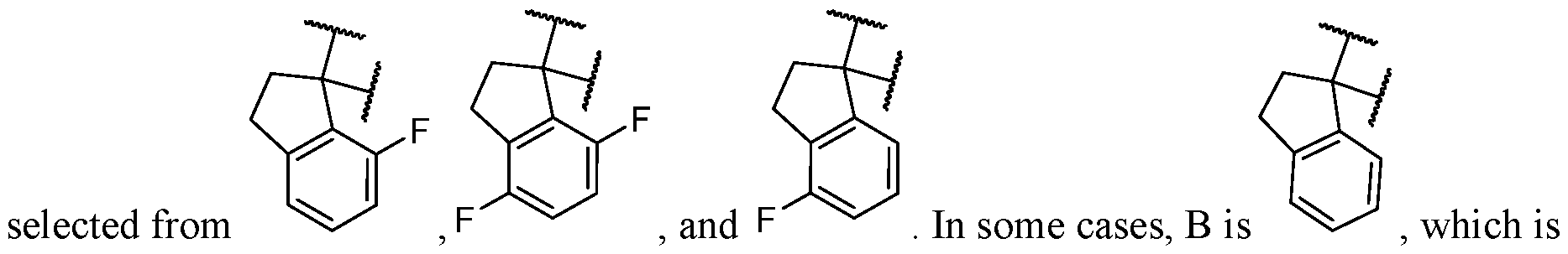
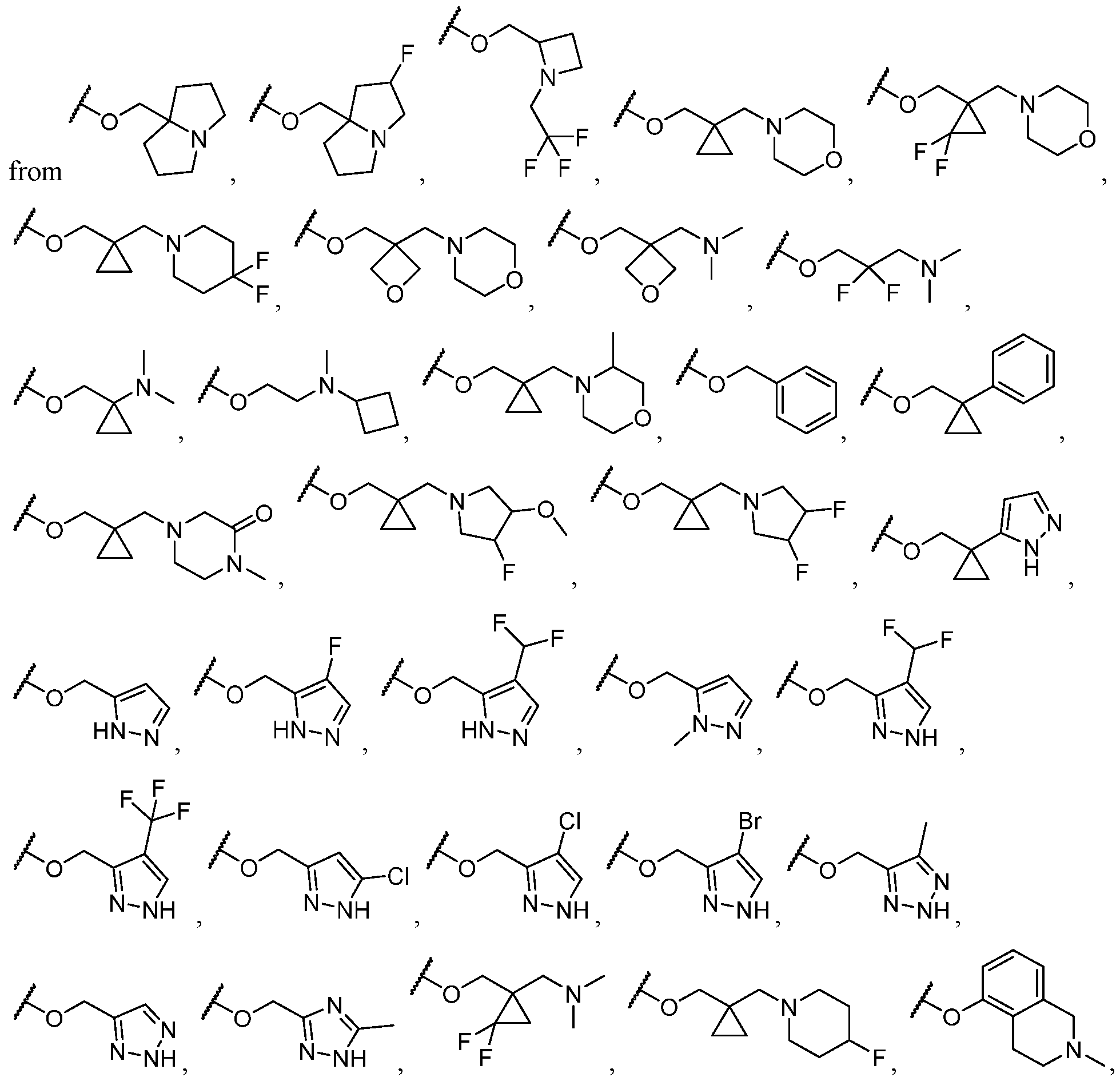
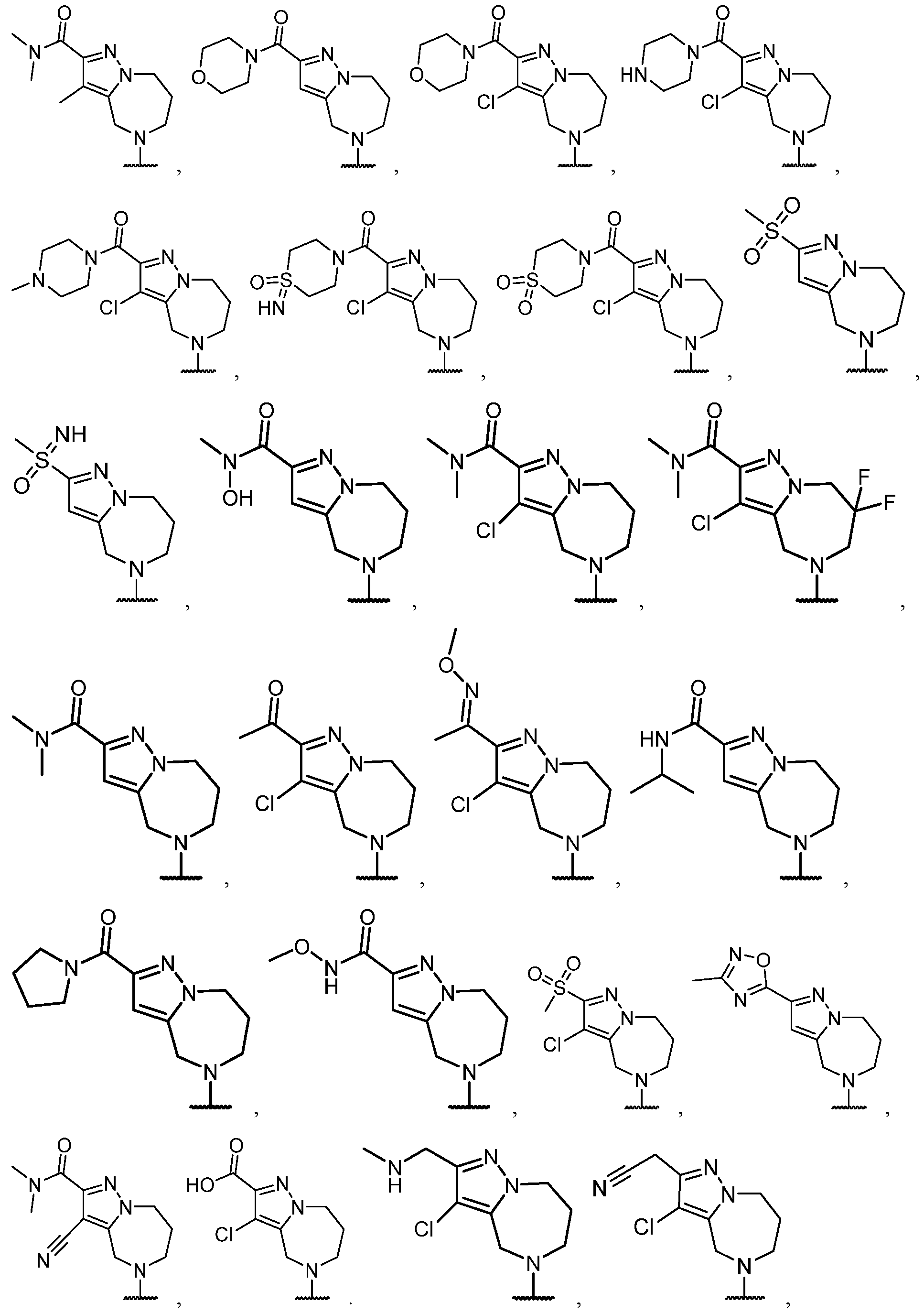
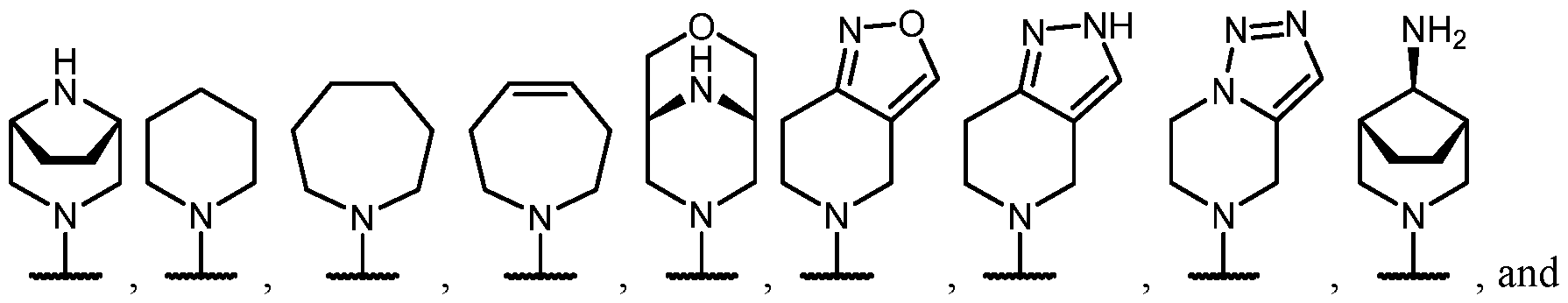
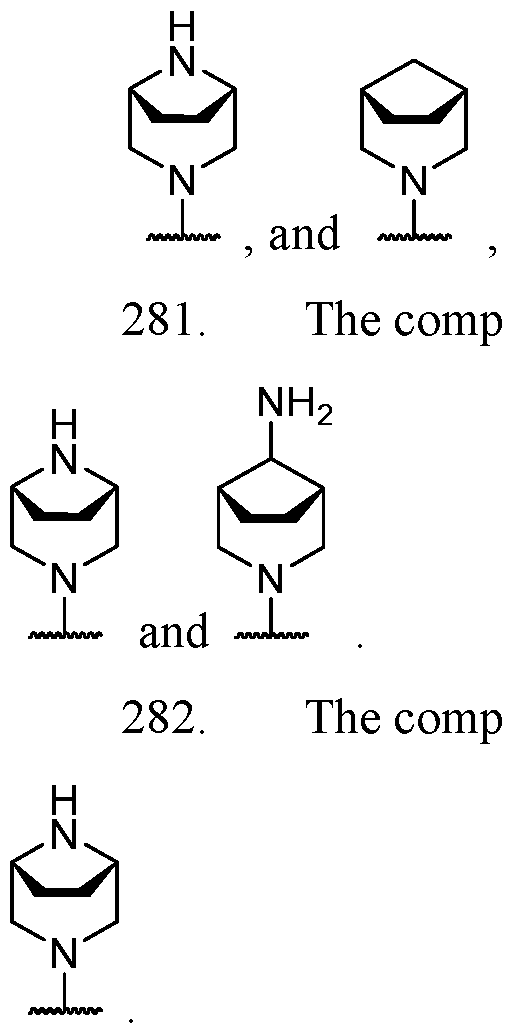
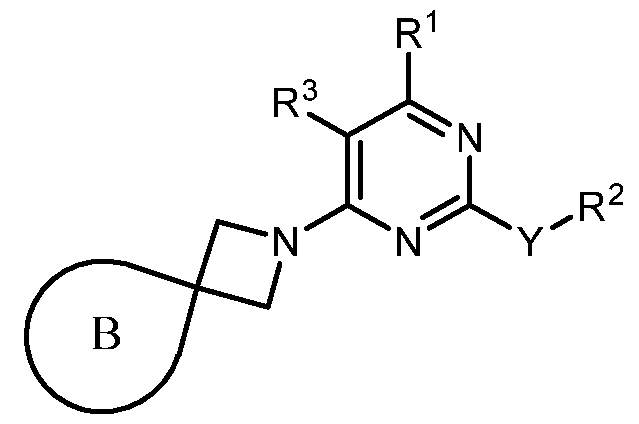
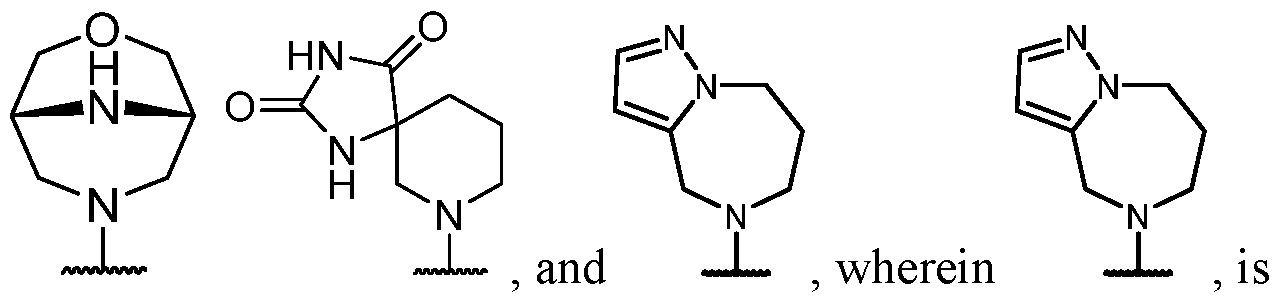
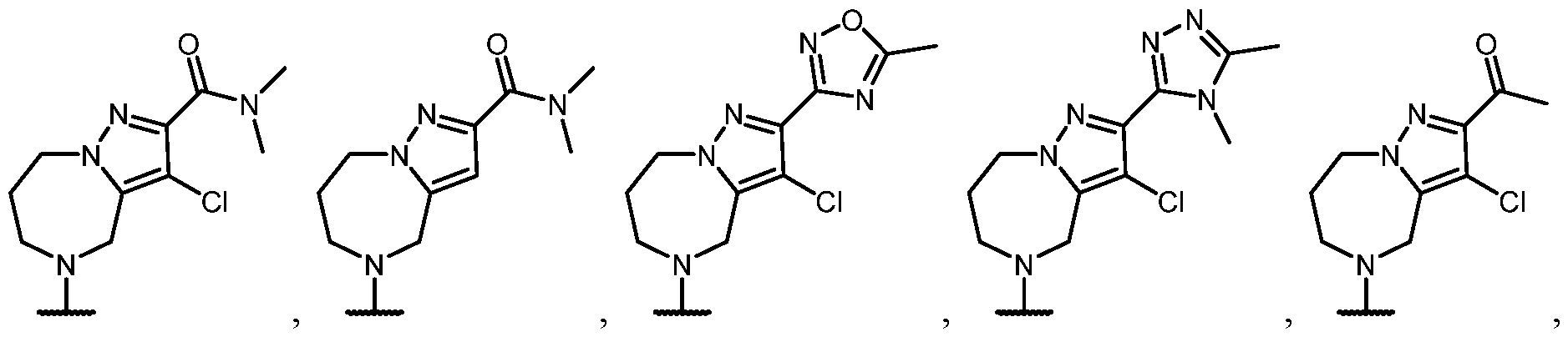
- B is selected from an optionally substituted 7- to 15-membered fused heterocycle and optionally substituted C7-C15 fused carbocycle. In some cases, and optionally substituted C7-C15 fused carbocycle. In some cases, B is an optionally substituted 7- to 15-membered fused heterocycle. In some cases, B is an optionally substituted unsaturated 7- to 15-membered fused heterocycle. In some cases, B is an optionally substituted 7- to 15-membered fused heteroaryl.
- B is selected from an optionally substituted 7- to 15-membered fused heteroaryl and optionally substituted C7-C15 fused aryl. In some cases, B is an optionally substituted unsaturated C7-C15 fused carbocycle. In some cases, B is an optionally substituted 7- to 15-membered fused heterocycle, wherein the fused heterocycle is partially unsaturated. In some cases, B is an optionally substituted 7- to 15- membered fused heterocycle, wherein the fused heterocycle is partially saturated.
- B is selected from an optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle. In some cases, and optionally substituted Cs-Cis fused carbocycle. In some cases, B is an optionally substituted 8- to 15-membered fused heterocycle. In some cases, B is an optionally substituted unsaturated 8- to 15-membered fused heterocycle. In some cases, B is an optionally substituted 8- to 15-membered fused heteroaryl.
- B is selected from an optionally substituted 8- to 15-membered fused heteroaryl and optionally substituted Cs-Cis fused aryl. In some cases, B is an optionally substituted unsaturated Cs-Cis fused carbocycle. In some cases, B is an optionally substituted 8- to 15-membered fused heterocycle, wherein the fused heterocycle is partially unsaturated. In some cases, B is an optionally substituted 8- to 15- membered fused heterocycle, wherein the fused heterocycle is partially saturated.
- B is selected from an optionally substituted 8- to 15-membered fused heterocycle, wherein the fused heterocycle is formed by combining three rings (e.g., tricyclic).
- B is selected from an optionally substituted 8- to 15-membered fused heterocycle, wherein the fused heterocycle is formed by combining two rings (e.g., bicyclic).
- the optionally substituted 8- to 15- membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle are each independently bicyclic or tricyclic.
- the optionally substituted 8- to 15- membered fused heterocycle is bicyclic. In some cases, for B the optionally substituted 8- to 15- membered fused heterocycle is tricyclic. [00124] In some embodiments, for a compound or salt for Formula (I), the heterocycle or carbocycle of B is bicyclic. In some cases, the heterocycle or carbocycle of B is tricyclic. In some cases, the tricyclic heterocycle contains three interconnected rings of atoms.
- the heterocycle and carbocycle are each independently selected from bicyclic and tricyclic. In some cases, for B, the heterocycle and carbocycle are each independently tricyclic. In some cases, for B, the heterocycle and carbocycle are each independently bicyclic.
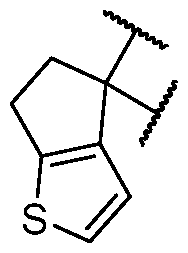
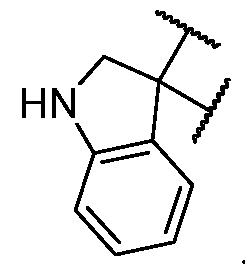
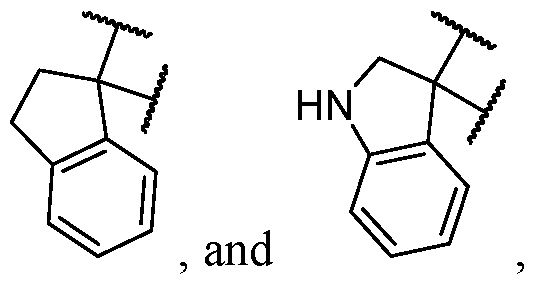
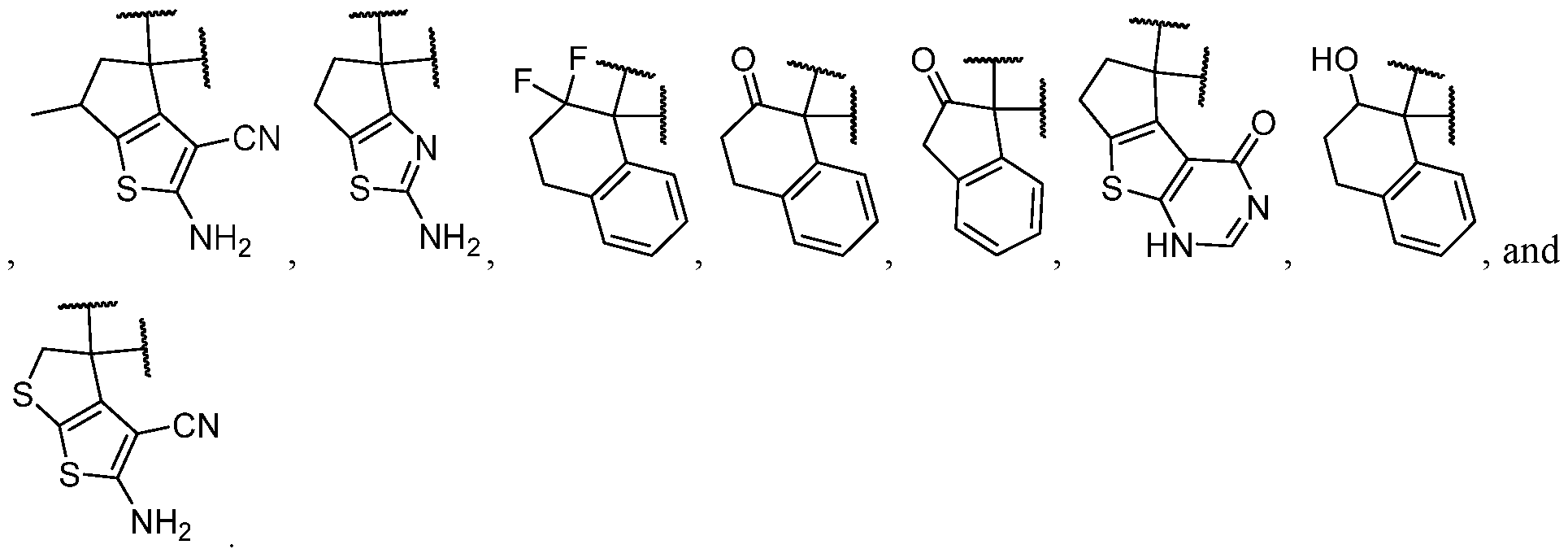
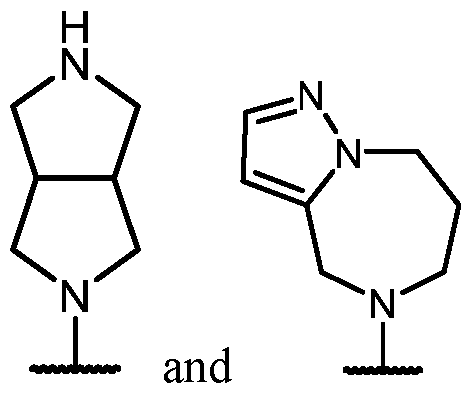
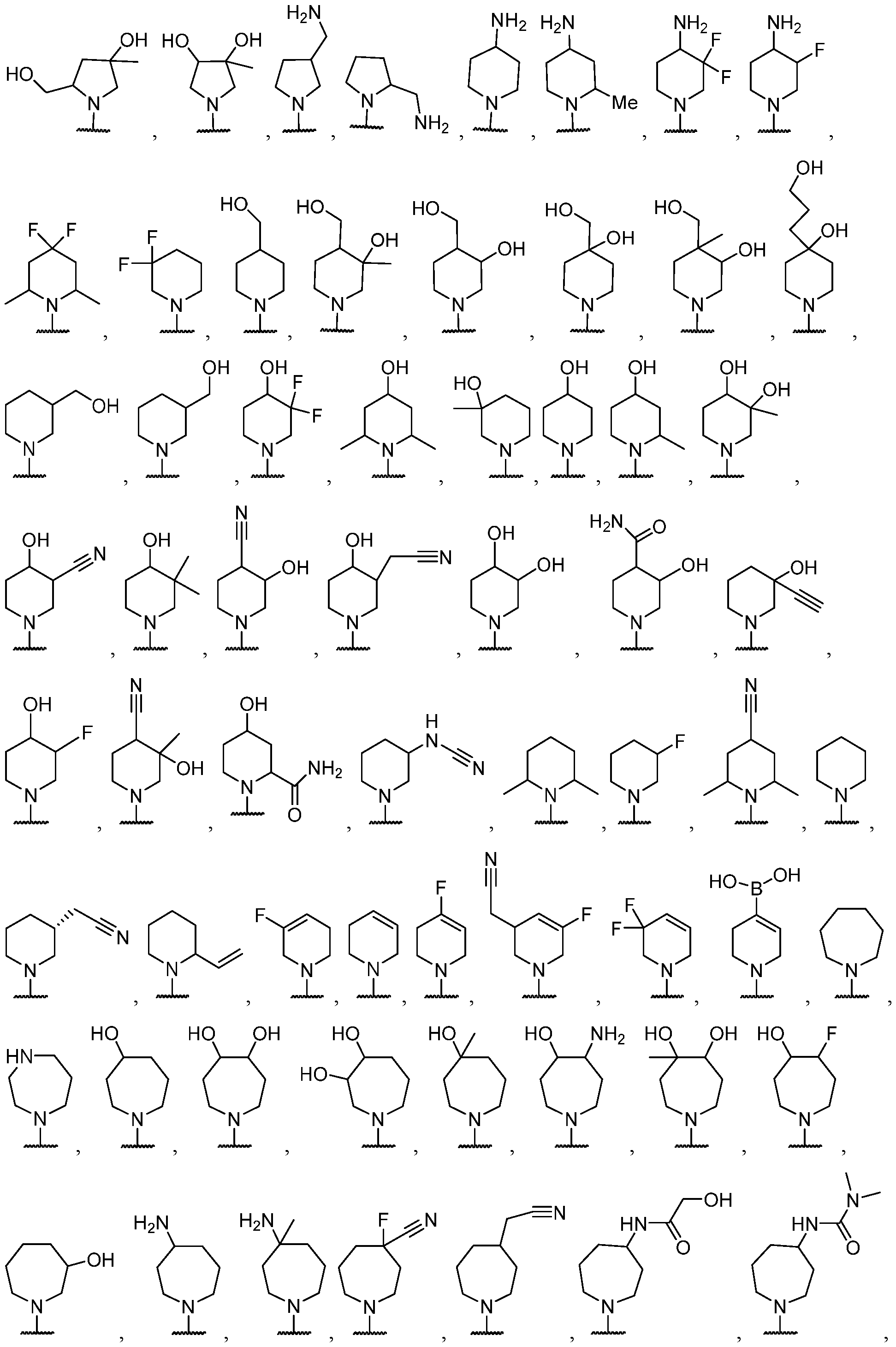
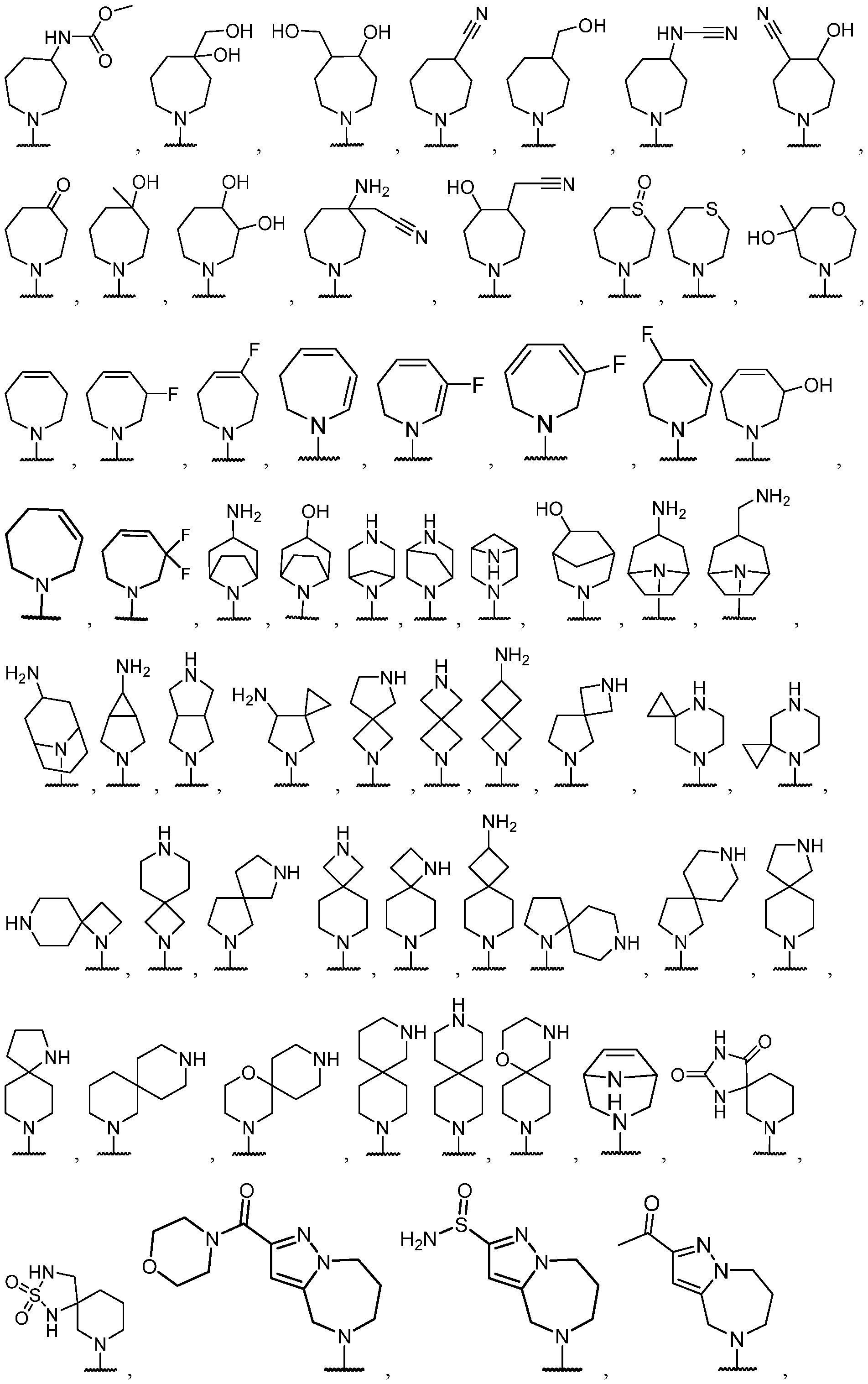
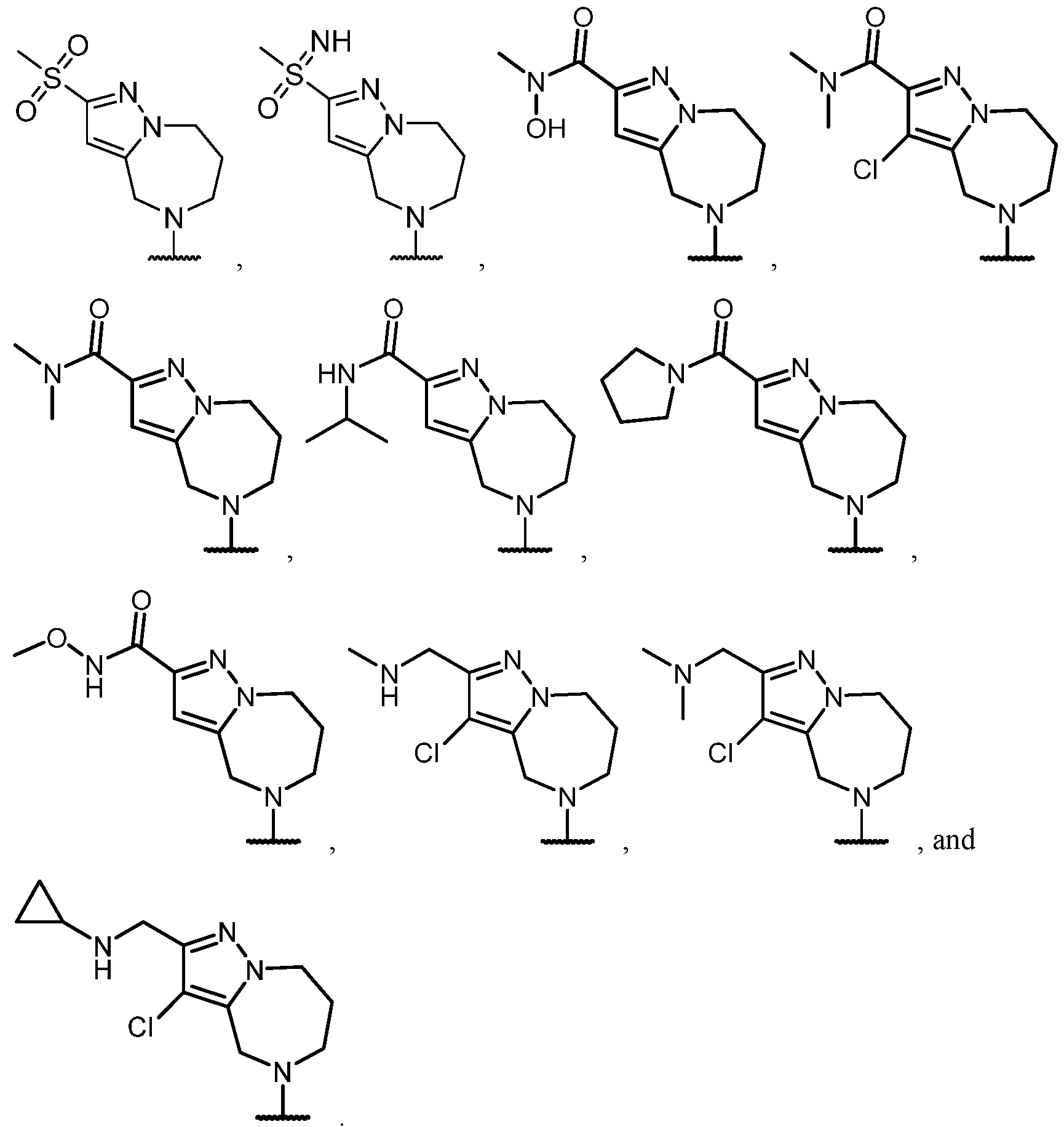
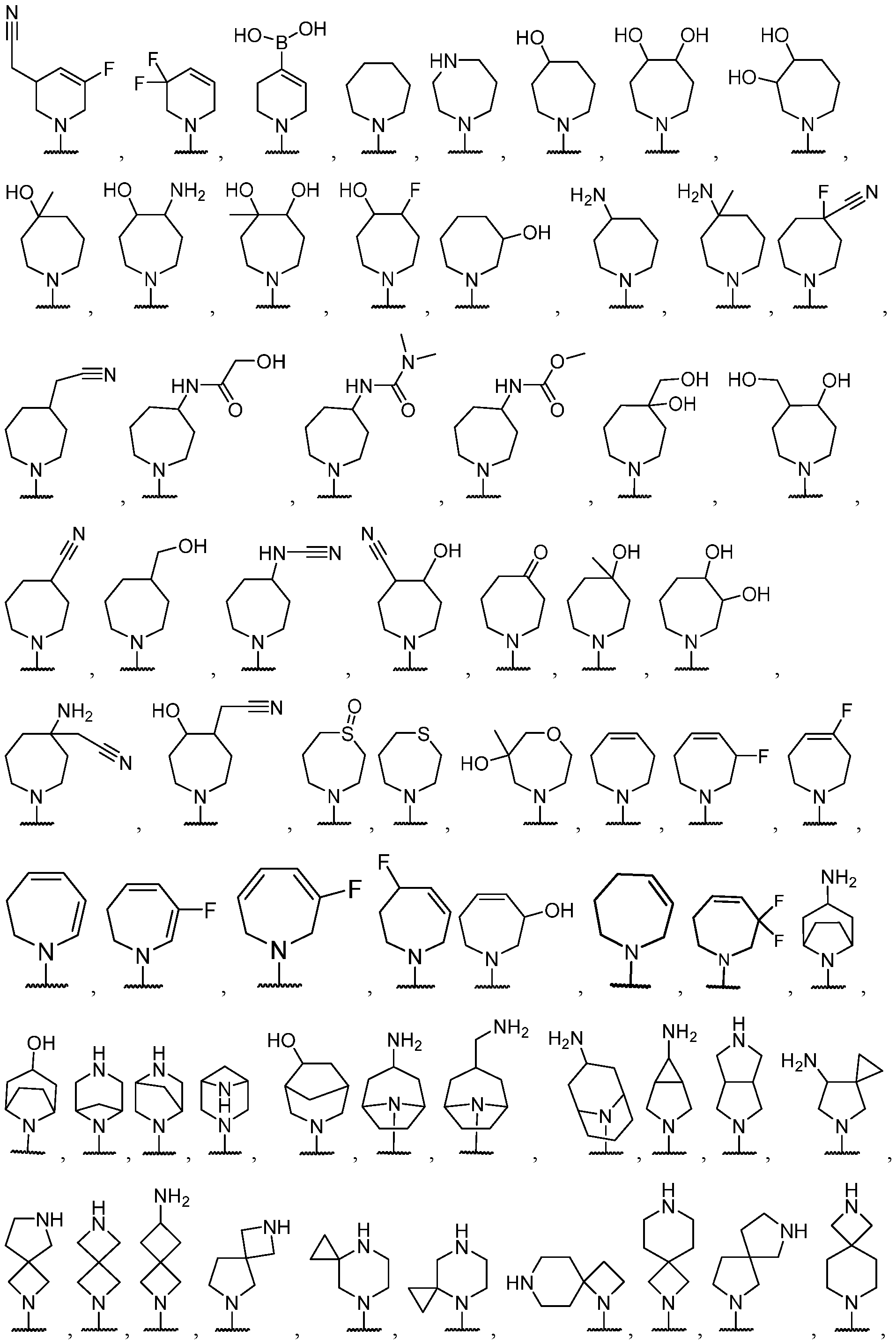
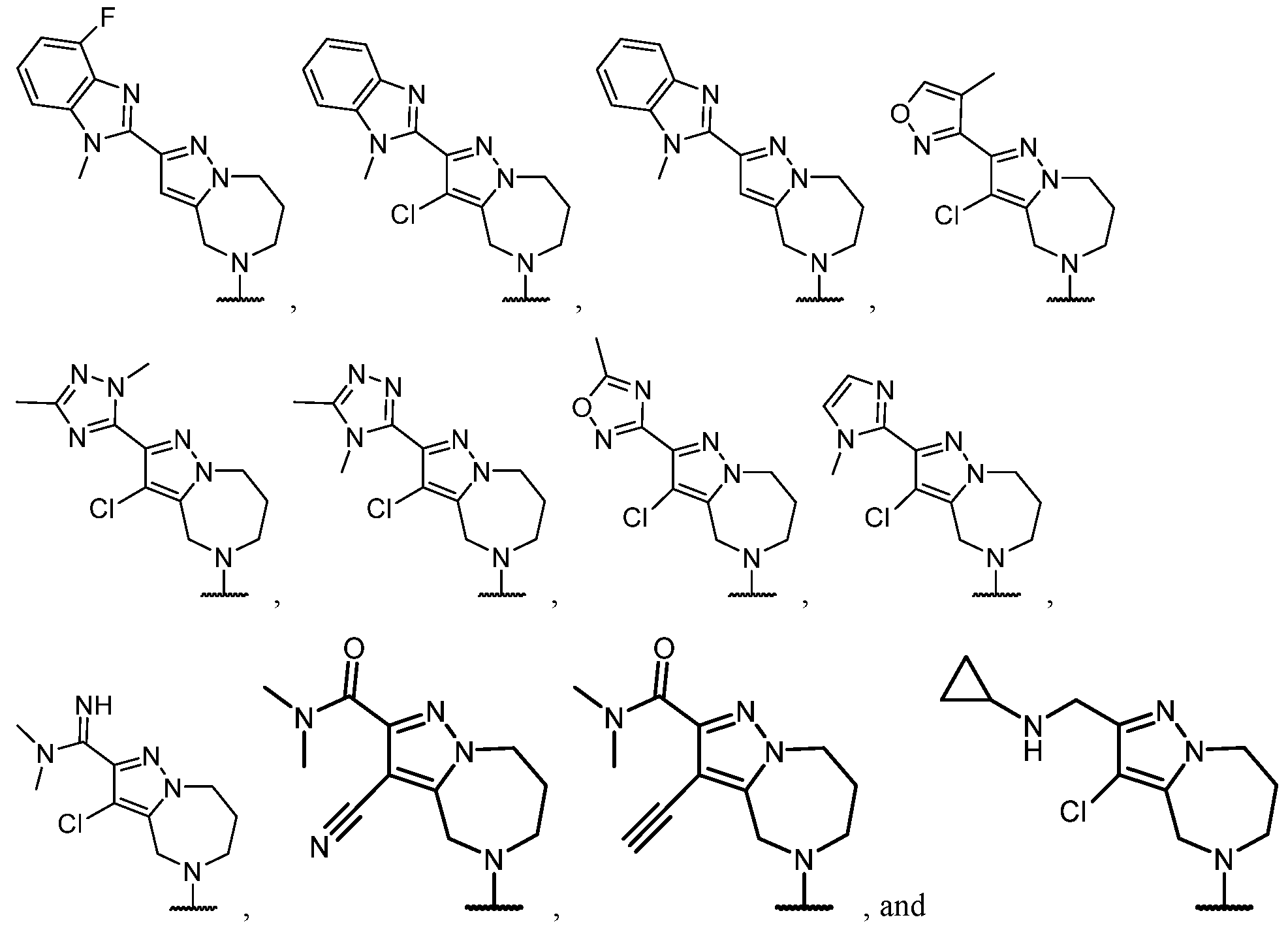
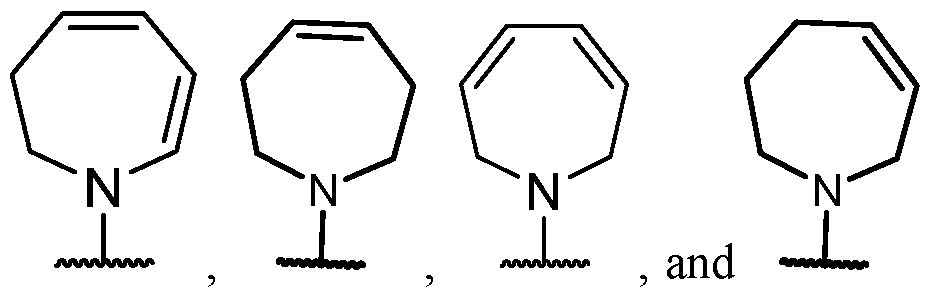
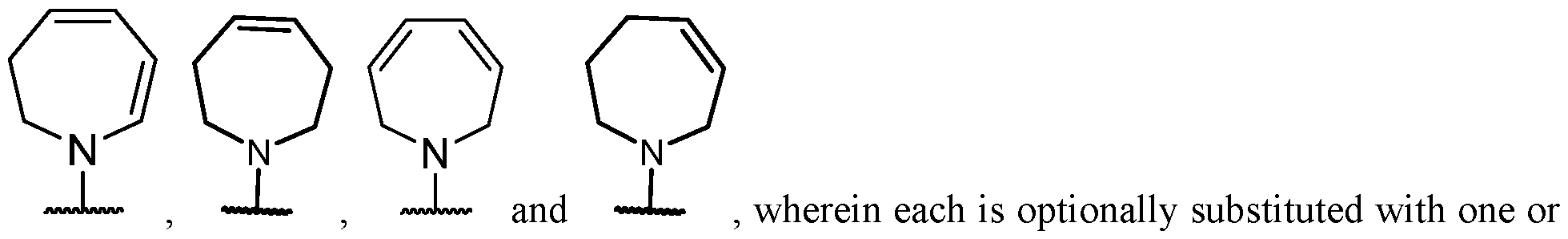
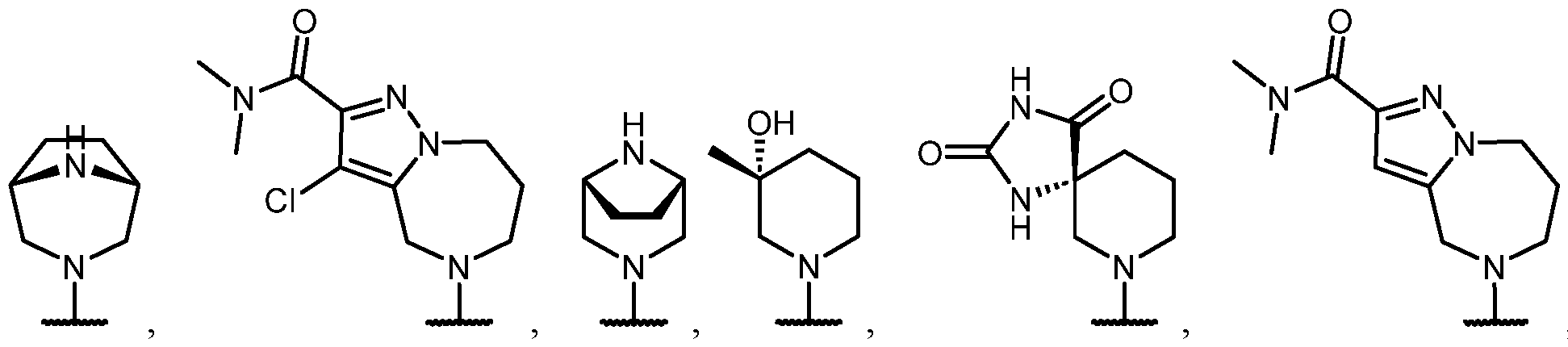
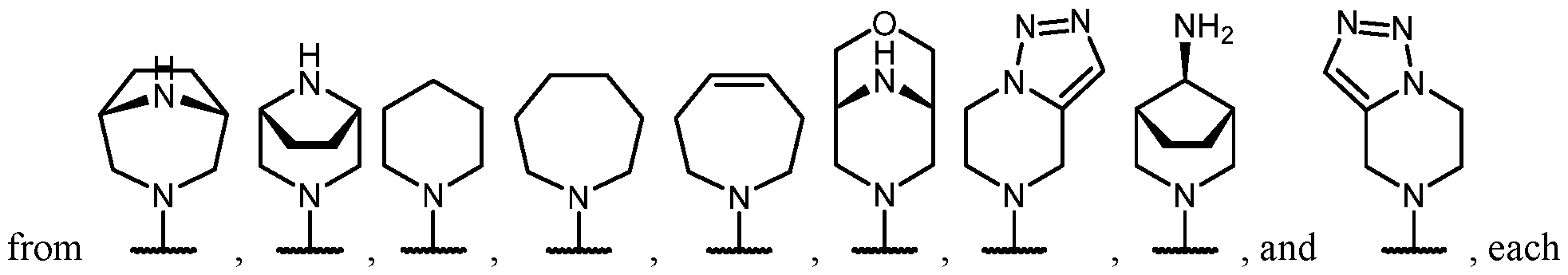
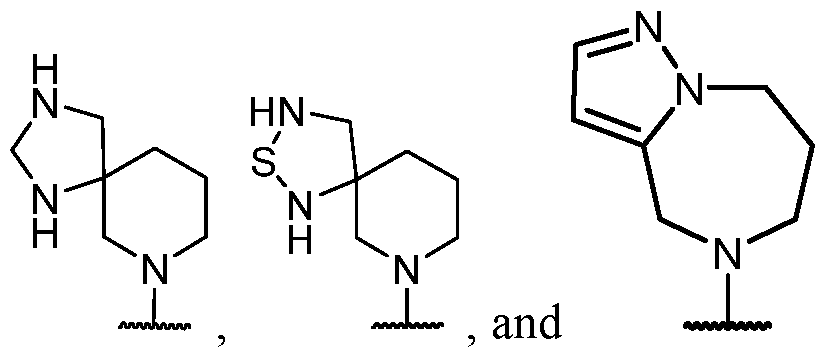
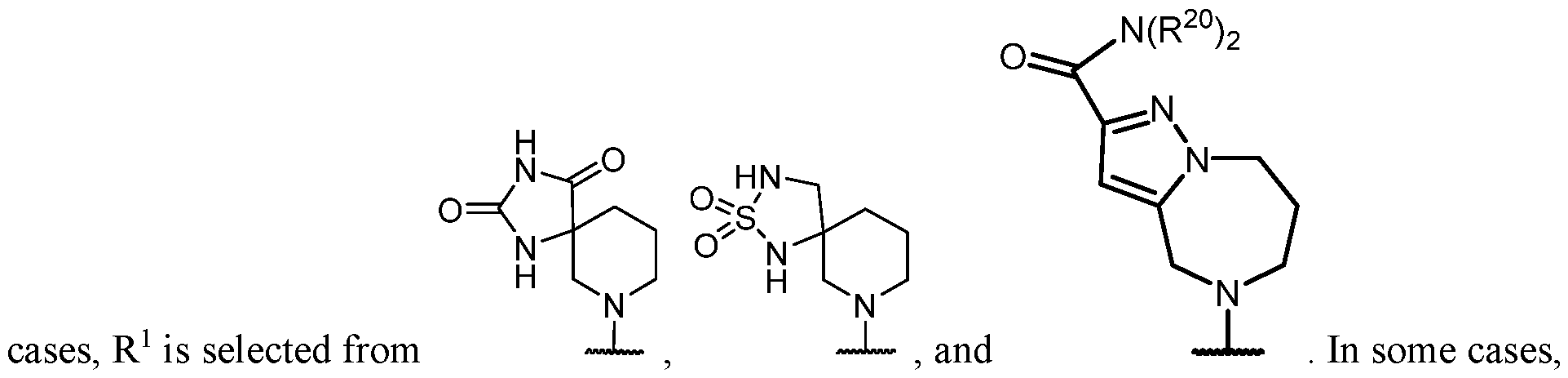
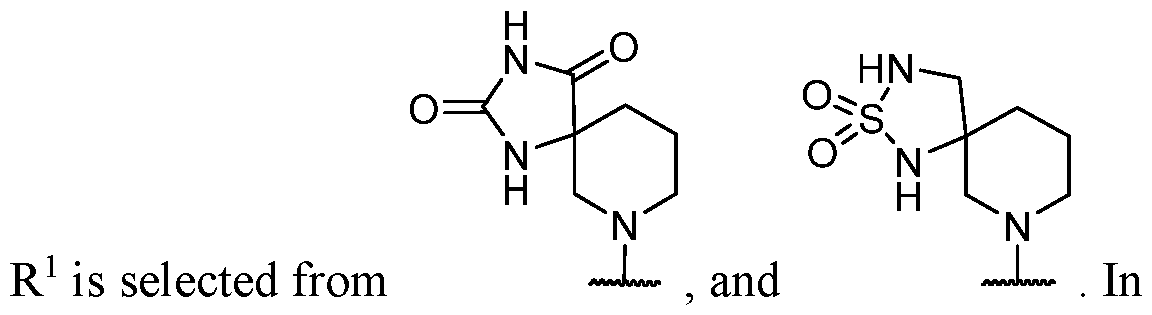
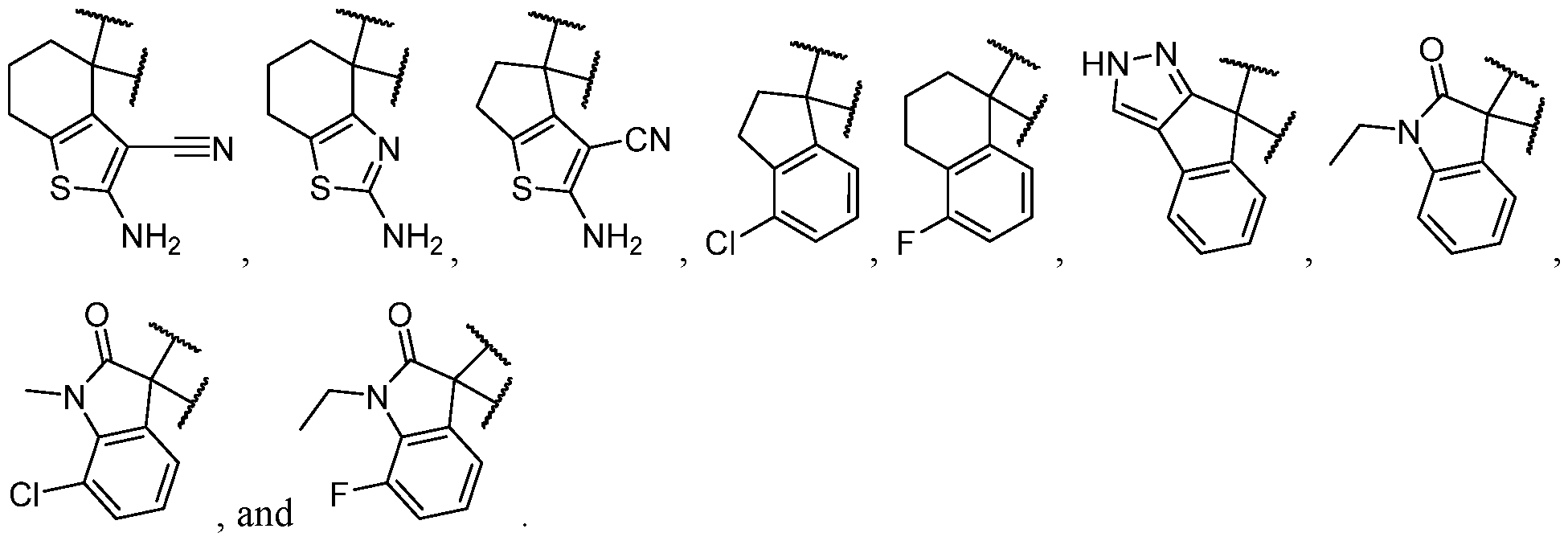
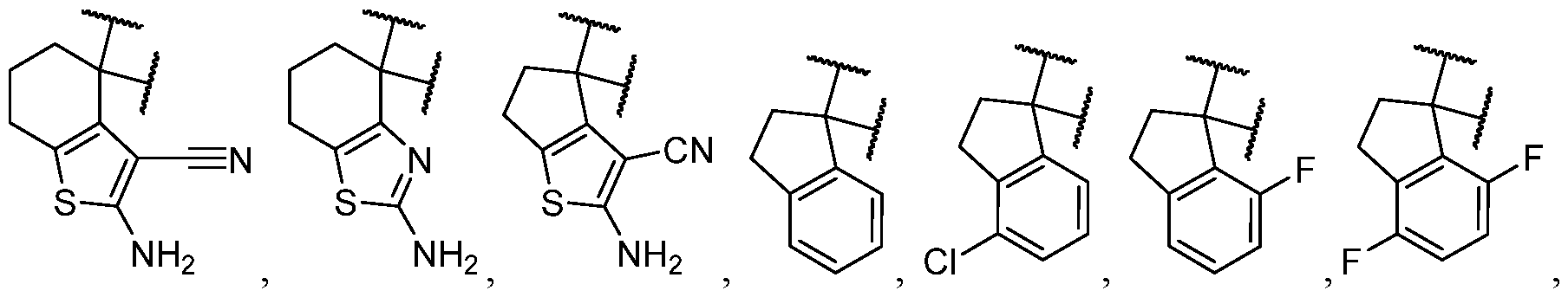
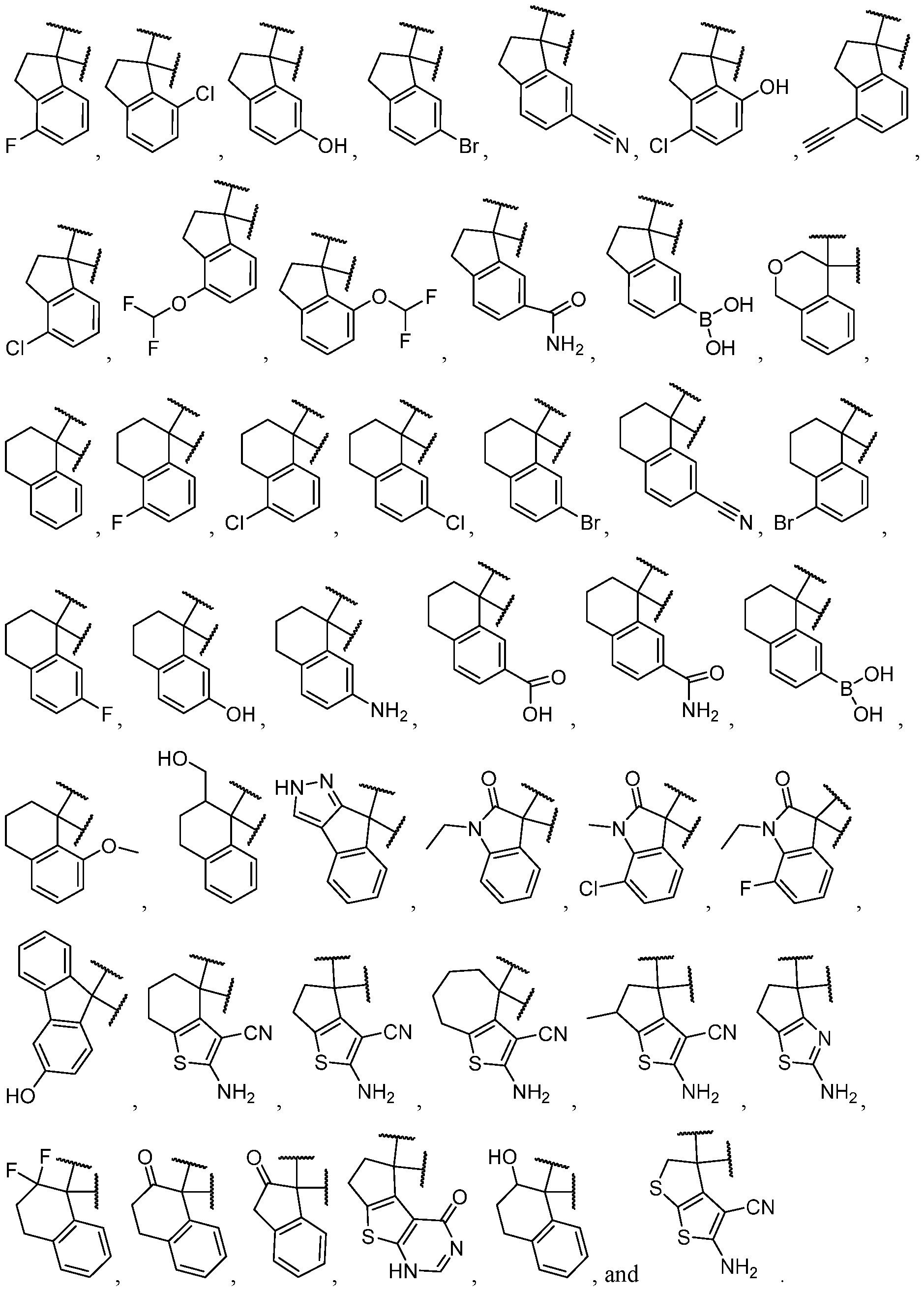
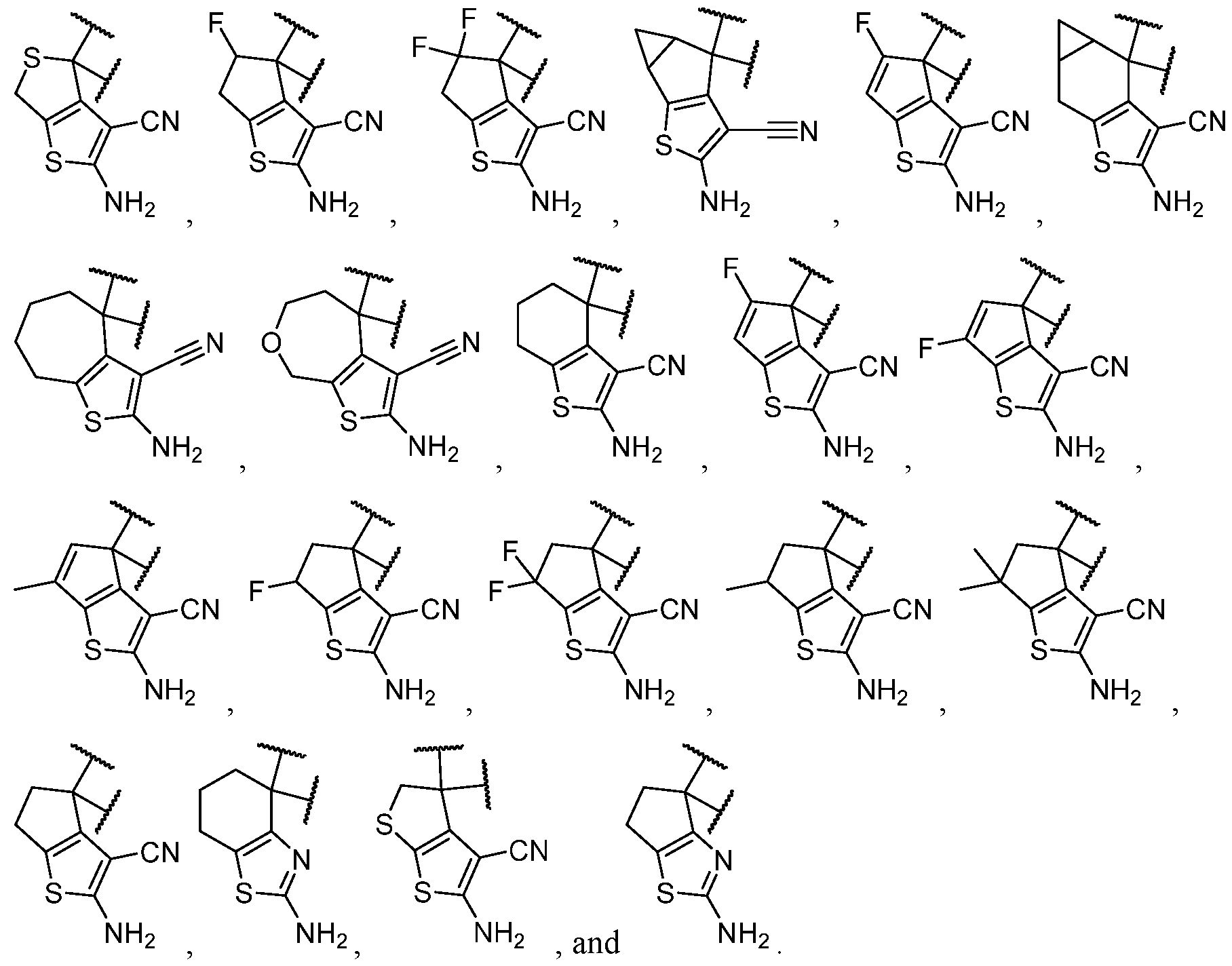
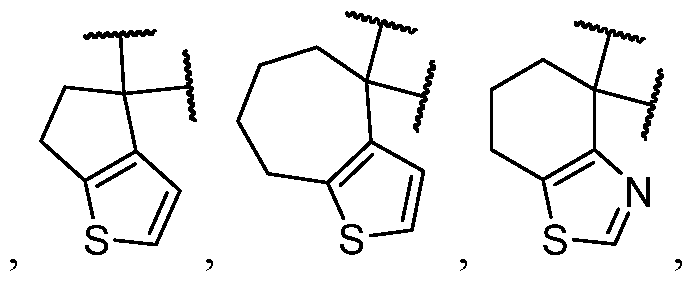
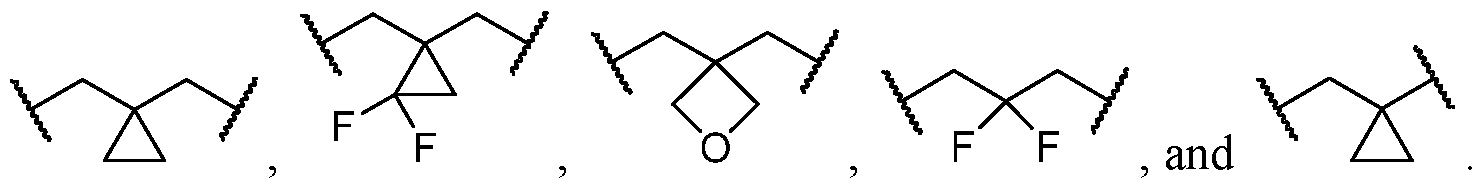
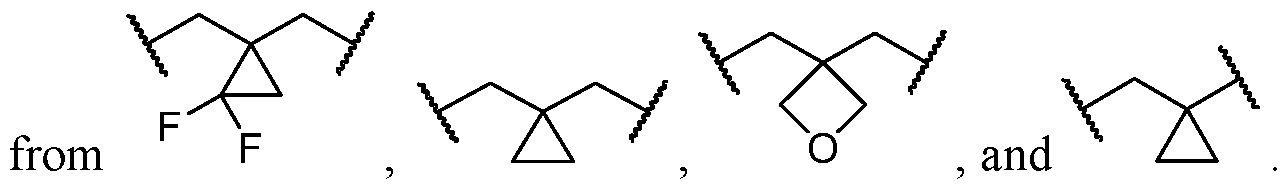
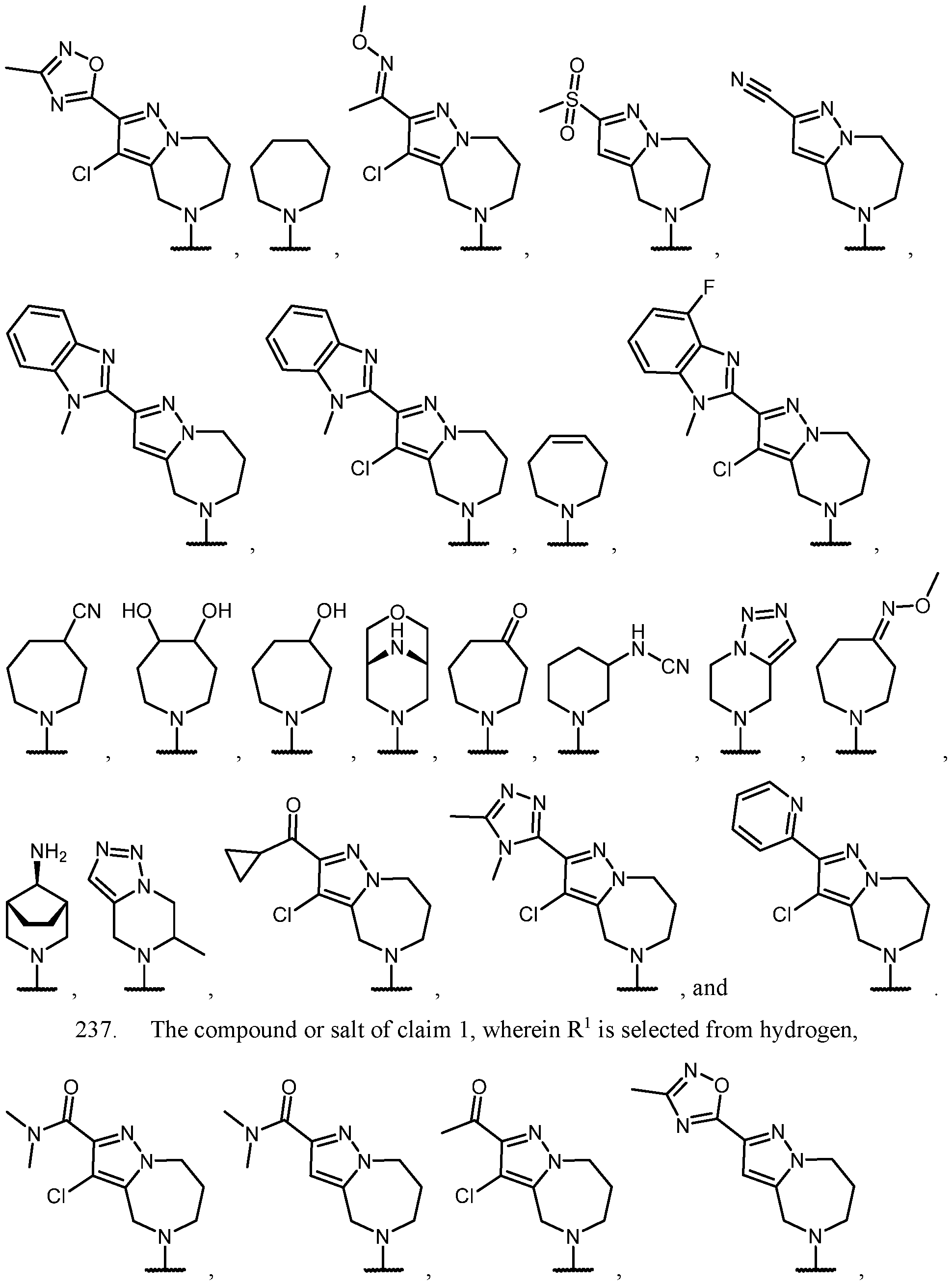
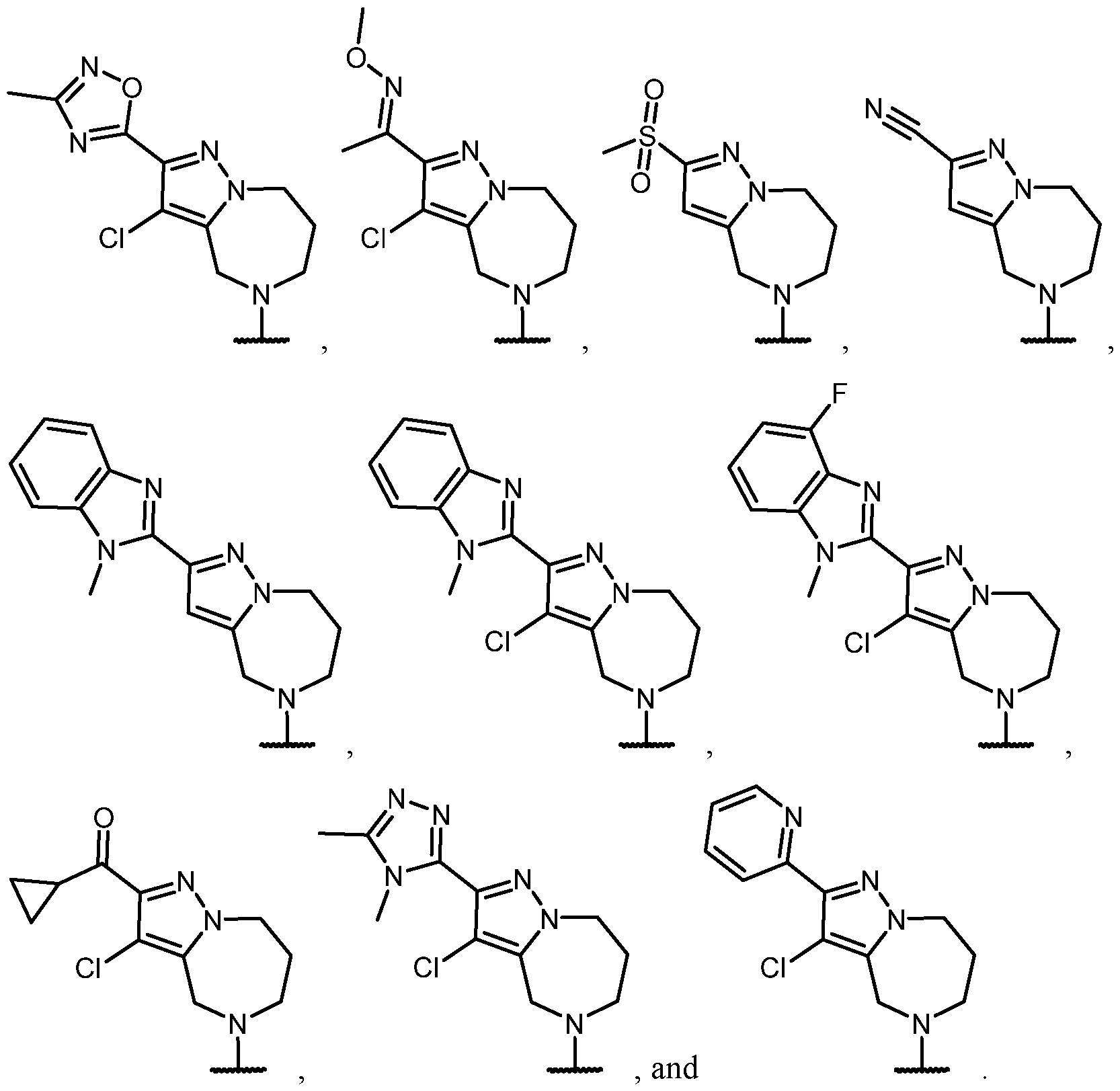
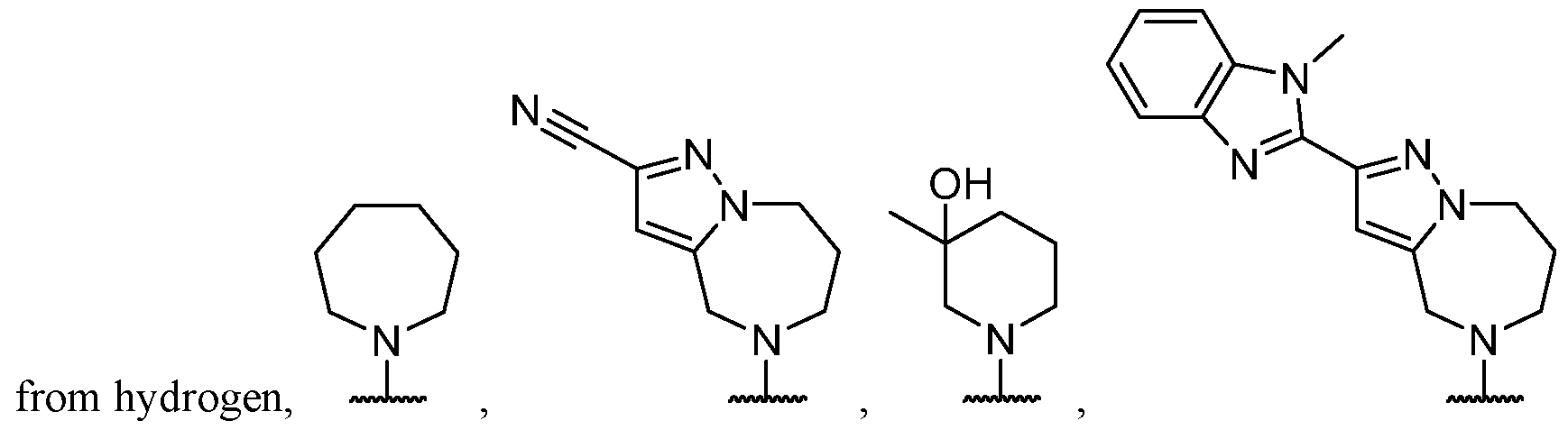
- the optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle are selected from , each of which is optionally substituted with one or more substituents.
- the optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle are selected from , each of which is optionally substituted with one or more substituents.
- B each of which is optionally substituted with one or more substituents.
- B is selected from each of which is optionally substituted with one or more substituents.
- B is selected from substituents.
- the one or more optional substituents of the heterocycle and carbocycle are each independently selected from oxo, -NH 2 , halogen, C1-C3 alkyl.
- B is selected from an optionally substituted 7- to 12-membered fused heterocycle and optionally substituted C9-10 fused carbocycle.
- the heterocycle of B has at least one sulfur atom.
- the heterocycle of B has one or sulfur atoms.
- the heterocycle of B has at least one substituted.
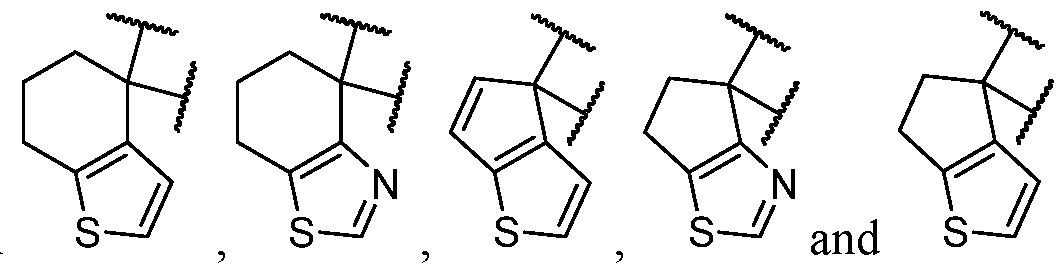
- B is selected from an optionally substituted 8- to 10-membered fused heterocycle having at least one sulfur atom. In is optionally substituted.
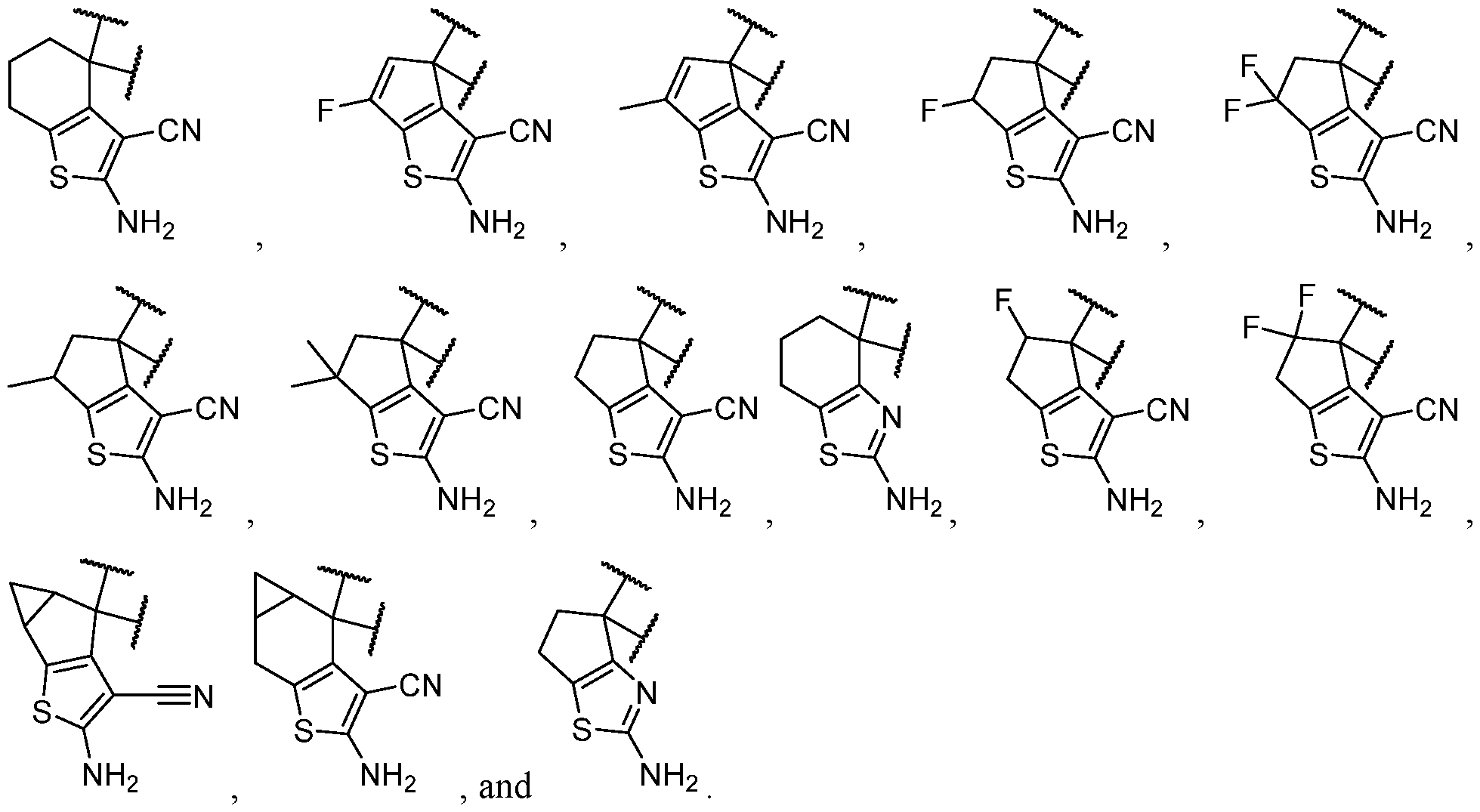
- the one or more optional substituents of B are independently selected at each occurrence from halogen, C1-C3 alkyl, -NH2, and -CN. In some cases, B is substituted. In some cases, B is substituted with at least one -NH2. In some cases, B is selected from some cases, B is substituted with at least one -NH2 at least one -CN. In some cases, B is selected
- B is an optionally substituted 7- to 11 -membered fused heterocycle. In some cases, B is an optionally substituted 8- to 10-membered fused heterocycle. In some cases, B is an optionally substituted 7-membered fused heterocycle. In some cases, B is an optionally substituted 8-membered fused heterocycle. In some cases, B is an optionally substituted 9-membered fused heterocycle. In some cases, B is an optionally substituted 10-membered fused heterocycle. In some cases, the heterocycle of B is an unsaturated heterocycle. In some cases, the heterocycle of B is a non-aromatic heterocycle.
- B has at least one sulfur atom. In some cases, B has at two sulfur atoms. In some cases, B has at least one sulfur atom and at least one nitrogen atom. In some cases, B has at least one sulfur atom and at least one oxygen atom. In some cases, B has only 1 heteroatom. In some cases,
- B has at least 2 heteroatoms. In some cases, B is selected from each of which is optionally substituted. In some cases, B is selected from each of which is optionally substituted. In some cases, B is selected from
- the one or more optional substituents of B are independently selected at each occurrence from halogen, C1-C3 alkyl, -NH2, and -CN.
- B is substituted with at least three substituents.
- B is substituted with at least two substituents. In some cases, B is substituted with at least one substituent. In some cases, B is substituted with at least one substituent selected from halogen, C1-C3 alkyl, -NH2, and -CN. In some cases, B is substituted with at least one substituent selected from halogen. In some cases, B is substituted with at least one substituent selected from -NH2. In some cases, B is substituted with at least one substituent selected from -CN. In some cases, B is selected from ,
- R 3 is selected from hydrogen, halogen, -CN, -NO 2 , -N(R 20 ) 2 , -OR 20 , -SR 20 , -S(O) 2 (R 20 ), -S(O) 2 N(R 20 ) 2 , - NR 20 S(O) 2 R 20 , -C(O)N(R 20 ) 2 , -N(R 20 )C(O)R 20 , -N(R 20 )C(O)N(R 20 ) 2 , -N(R 20 )C(O)OR 20 , - C(O)R 20 , -C(O)OR 20 , -OC(O)R 20 , -OC(O)N(R 20 ) 2 , Ci- 6 aminoalkyl, Ci- 6 alkoxy, Ci- 6 alkoxyalkyl, C1-6 hydroxyalkyl, C 1-6
- R 3 is selected from hydrogen, halogen, -CN, -NO 2 , -N(R 20 ) 2 , -OR 20 , -SR 20 , -C(O)N(R 20 ) 2 , -C(O)R 20 , -C(O)OR 20 , -OC(O)R 20 , Ci- 6 aminoalkyl, Ci- 6 alkoxy, Ci- 6 alkoxyalkyl, Ci- 6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, C 2 -6 alkenyl, C 2 -e alkynyl.
- R 3 is selected from hydrogen, halogen, -CN, -NO 2 , -NH 2 , -N(CI-6 alkyl)H -N(CI-6 alkyl) 2 ,-OH, - C(O)N(R 20 ) 2 , -C(O) R 20 , -C(O)OR 20 , -OC(O) R 20 , CI-6 aminoalkyl, Ci- 6 alkoxy, Ci- 6 alkoxyalkyl,
- R 3 is selected from hydrogen, -CN, -C(O)R 20 , C 1-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, and Ci-6 alkyl. In some cases, R 3 is selected from hydrogen, -CN, -C(O)R 20 , Ci-6 hydroxyalkyl, and Ci-6 alkyl.
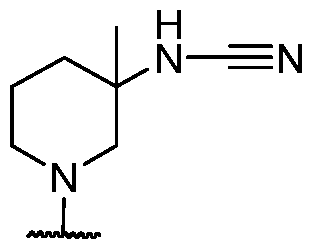
- R 3 is selected from hydrogen, -CN, -C(O)H, Ci hydroxyalkyl, and Ci-6 alkyl. In some cases, R 3 is selected from hydrogen, fluorine, and -CN. In some cases, R 3 is selected from fluorine. In some cases, R 3 is selected from hydrogen. In some cases, R 3 is selected from -CN.
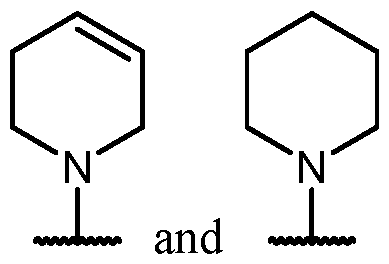
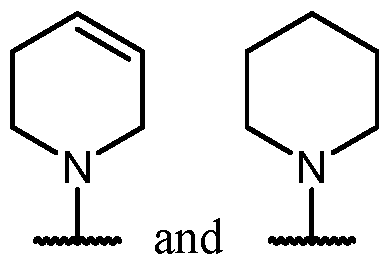
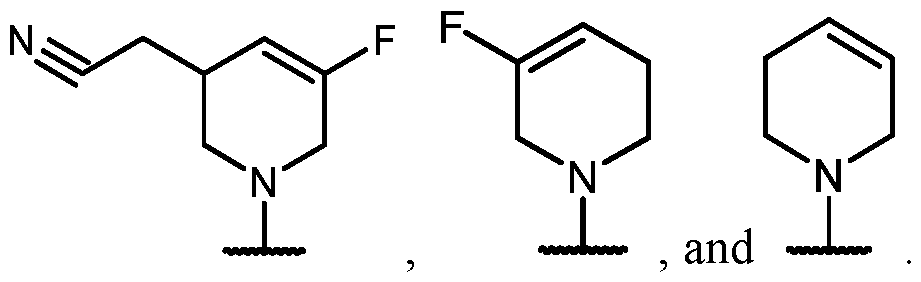
- Y is -O-. In some cases, Y is a bond. In some cases, Y is -S-. In some cases, Y is -N(R 5 )-.
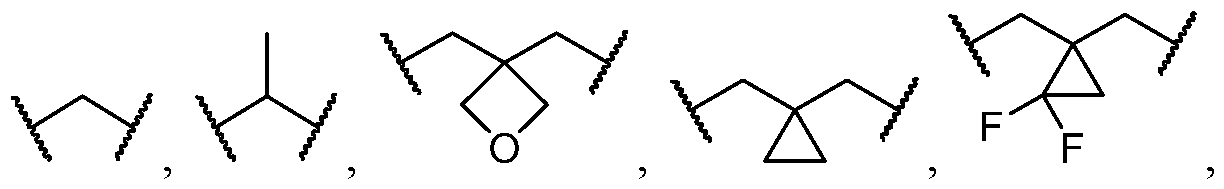
- L is selected from Ci- C4 alkylene. In some cases, L is selected from an unsubstituted C1-C4 alkylene. In some cases, L is selected from an unsubstituted Ci alkylene.
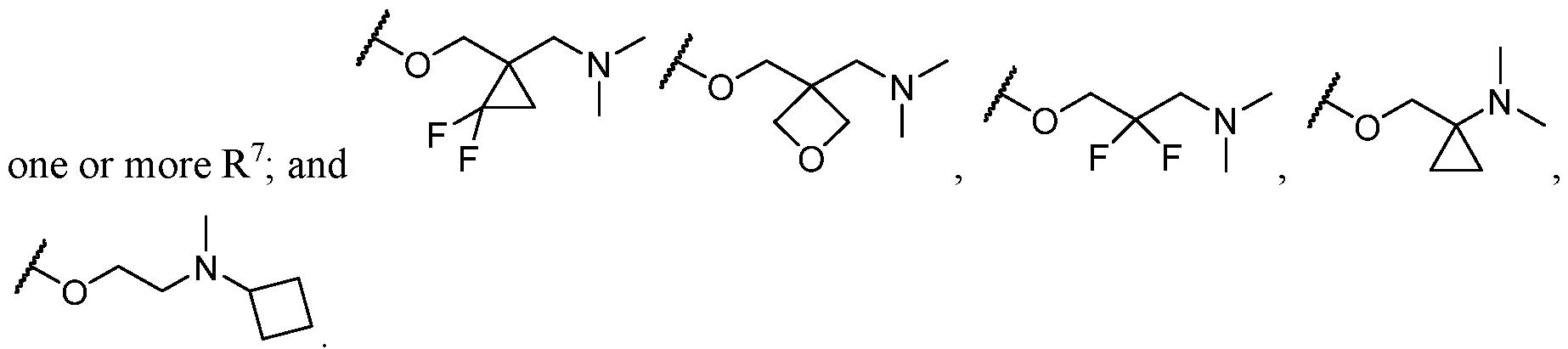
- two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle.
- L is selected from Ci- C4 alkylene. In some cases, L is selected from unsubstituted C1-C4 alkylene.
- the optional substituents of L are selected from C1-C4 hydroxyalkyl, C1-C4 alkyl, C3-C6 carbocycle; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle wherein the C3-C6 carbocycle and 3- to 8- membered heterocycle are optionally substituted with one or more substituents selected from cases, each L is independently selected from a substituted C1-C4 alkylene, and wherein two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle 3- to 5- membered heterocycle.
- each L is independently selected from a substituted C2-3 alkylene, and wherein two substituents on the same carbon atom of L come together to form a C3 carbocycle or 4-membered heterocycle, wherein the C3 carbocycle is optionally substituted with one or more substituents selected from halogen.
- each L is independently selected cases, each L is independently selected from -k .
- each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen and C1-C4 alkyl. In some cases, L is selected from
- each L is independently selected from an unsubstituted C1-C4 alkylene. In some cases, L is selected from ⁇ and .
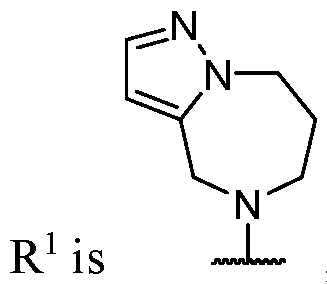
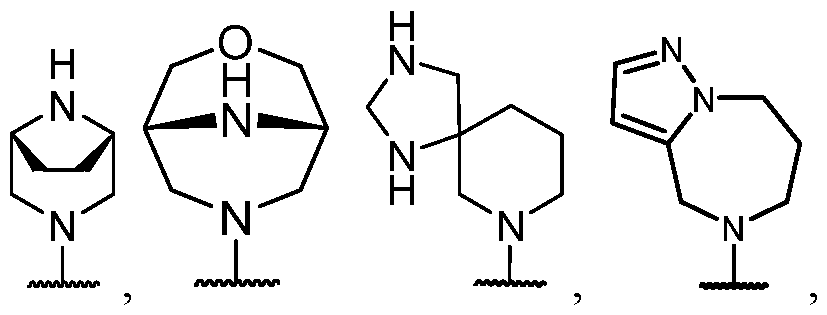
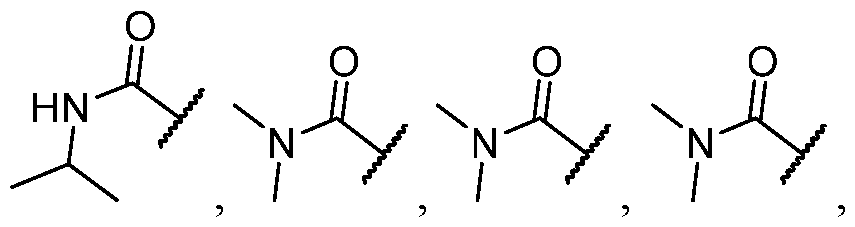
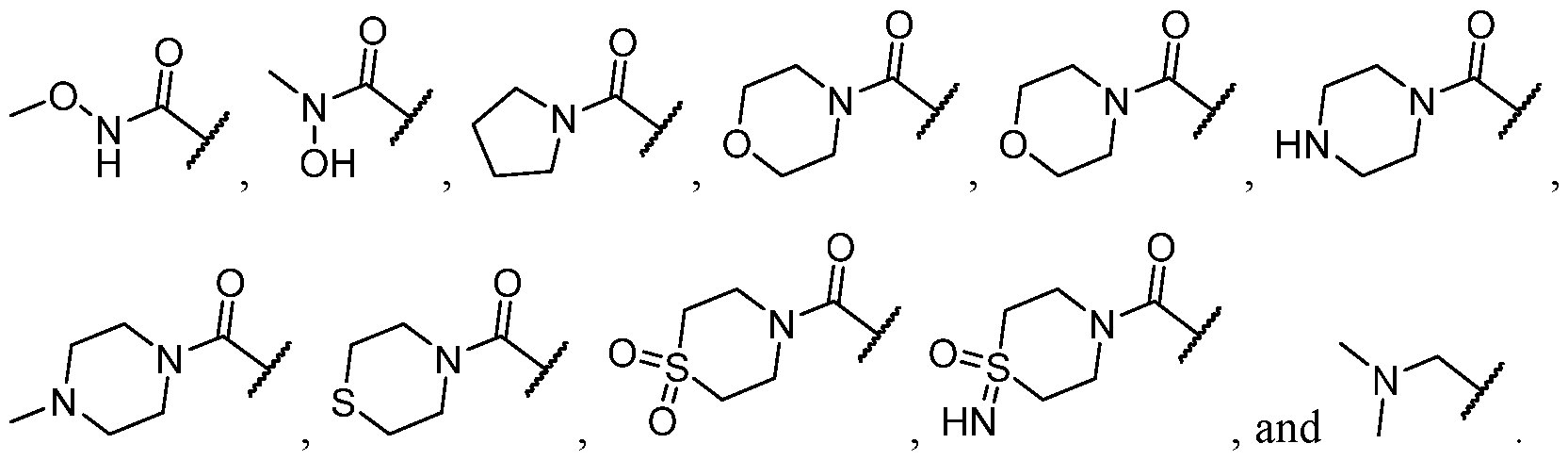
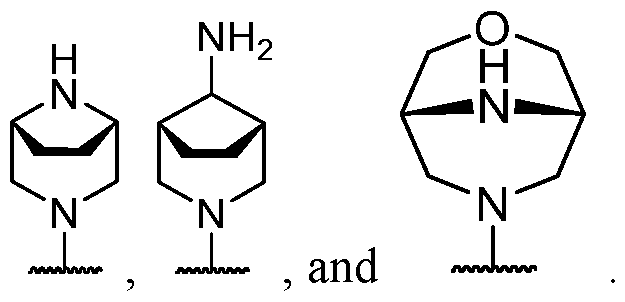
- R 2 is selected from heterocycle, -L-heterocycle, -L-aryl, -L-heteroaryl, and -L-N(R 23 )2, wherein the heterocycle, the heterocycle portion of -L-heterocycle, are each optionally substituted with one or more R 6 , and wherein the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R 7 .
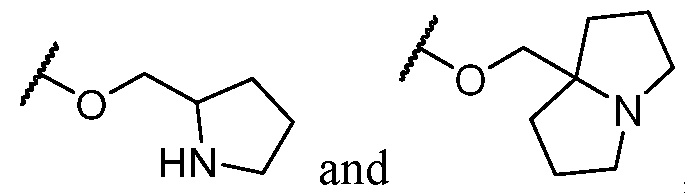
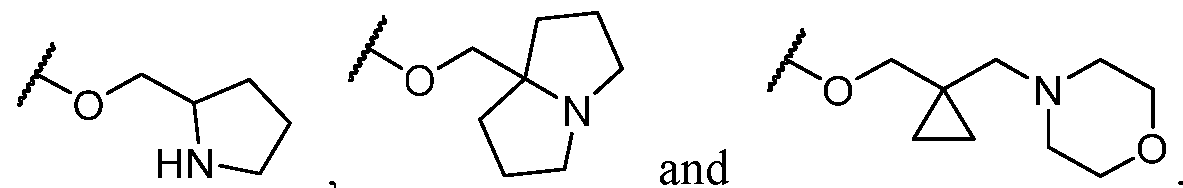
- R 2 is -L-heterocycle, wherein the heterocycle portion is optionally substituted.
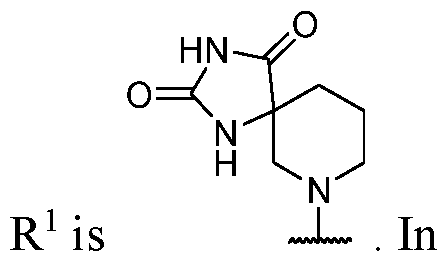
- R 2 is -L-heterocycle, wherein the heterocycle portion is a bicyclic heterocycle.
- R 2 is -L-heterocycle, wherein the heterocycle portion is a monocyclic heterocycle.
- R 2 is -L-heterocycle, wherein the heterocycle portion is a saturated heterocycle.
- R 2 is selected from a - L-5- to 10-membered heterocycle.
- R 2 is selected from a -(C1-C2 alkylene)-5- to 10-membered heterocycle.
- R 2 is selected from a -L-5- to 8-membered heterocycle. In some cases, R 2 is selected from a -L-5- to 8-membered saturated heterocycle. In some cases, R 2 is a -L-5-membered heterocycle. In some cases, R 2 is a -L-8-membered heterocycle. In some cases, the heterocycle contains at least 1 nitrogen atom. In some cases, the heterocycle contains at most 1 nitrogen atom. In some cases, the heterocycle contains 1 nitrogen atom. In some cases, the bicyclic heterocycle contains at least 1 nitrogen atom. In some cases, the bicyclic heterocycle contains at most 1 nitrogen atom. In some cases, the bicyclic heterocycle contains 1 nitrogen atom.
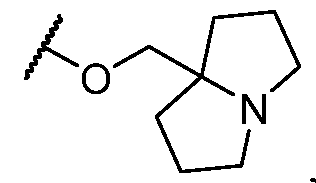
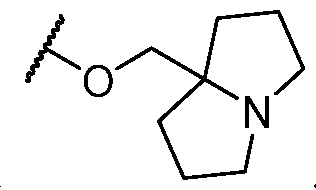
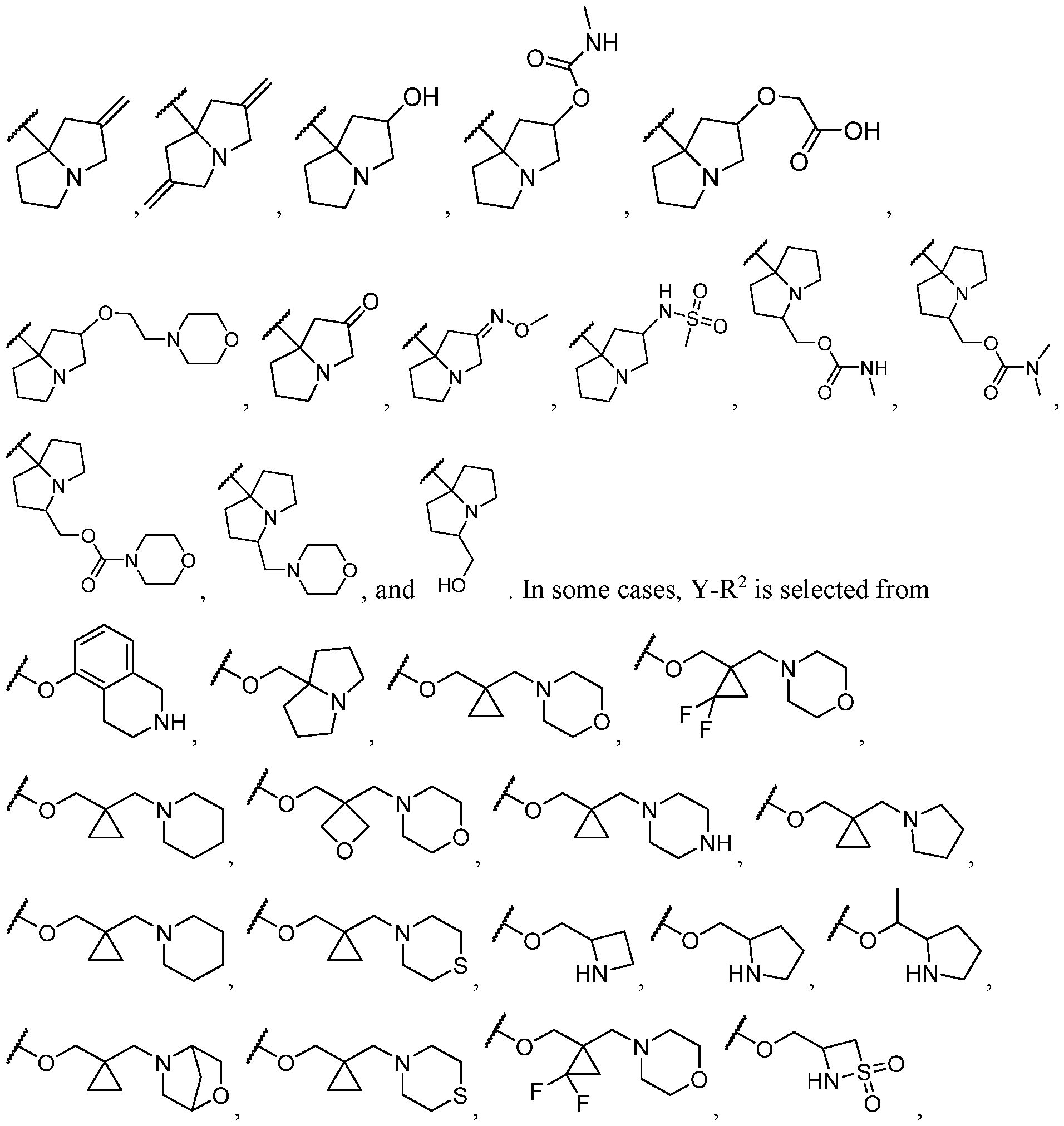
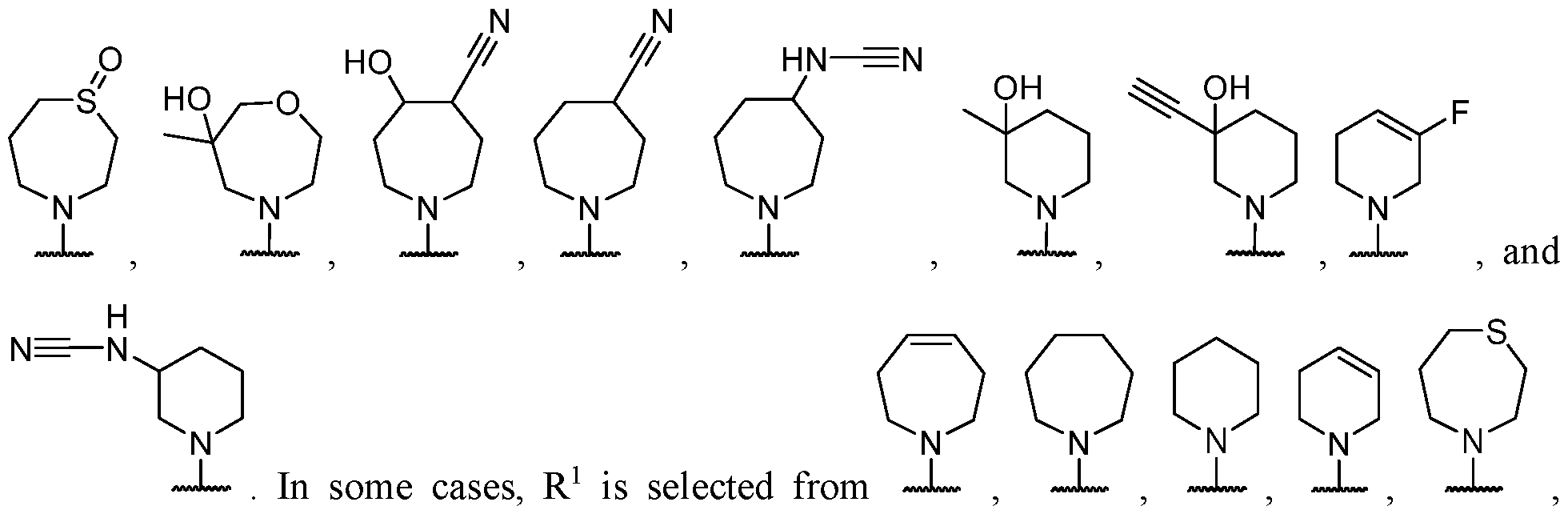
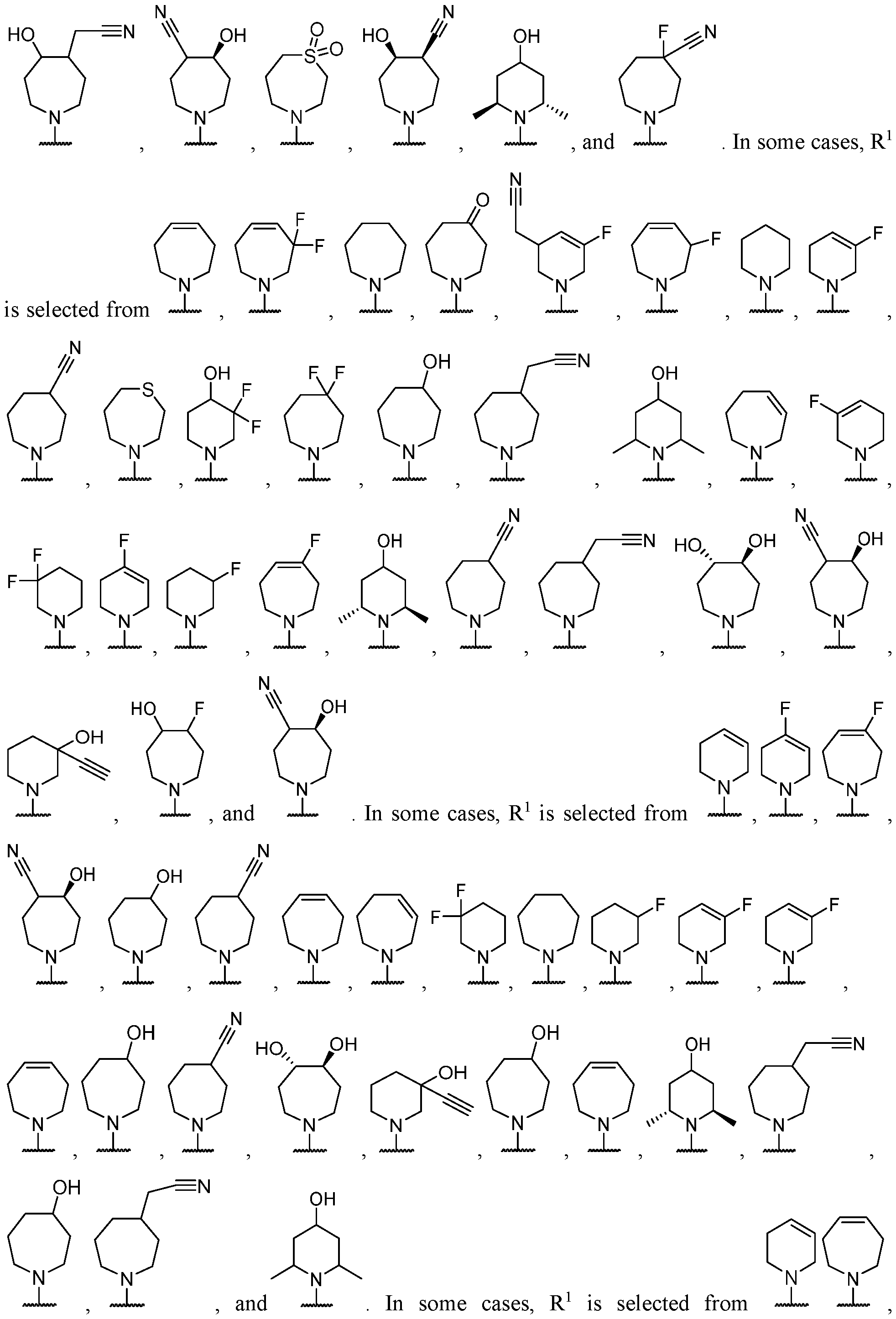
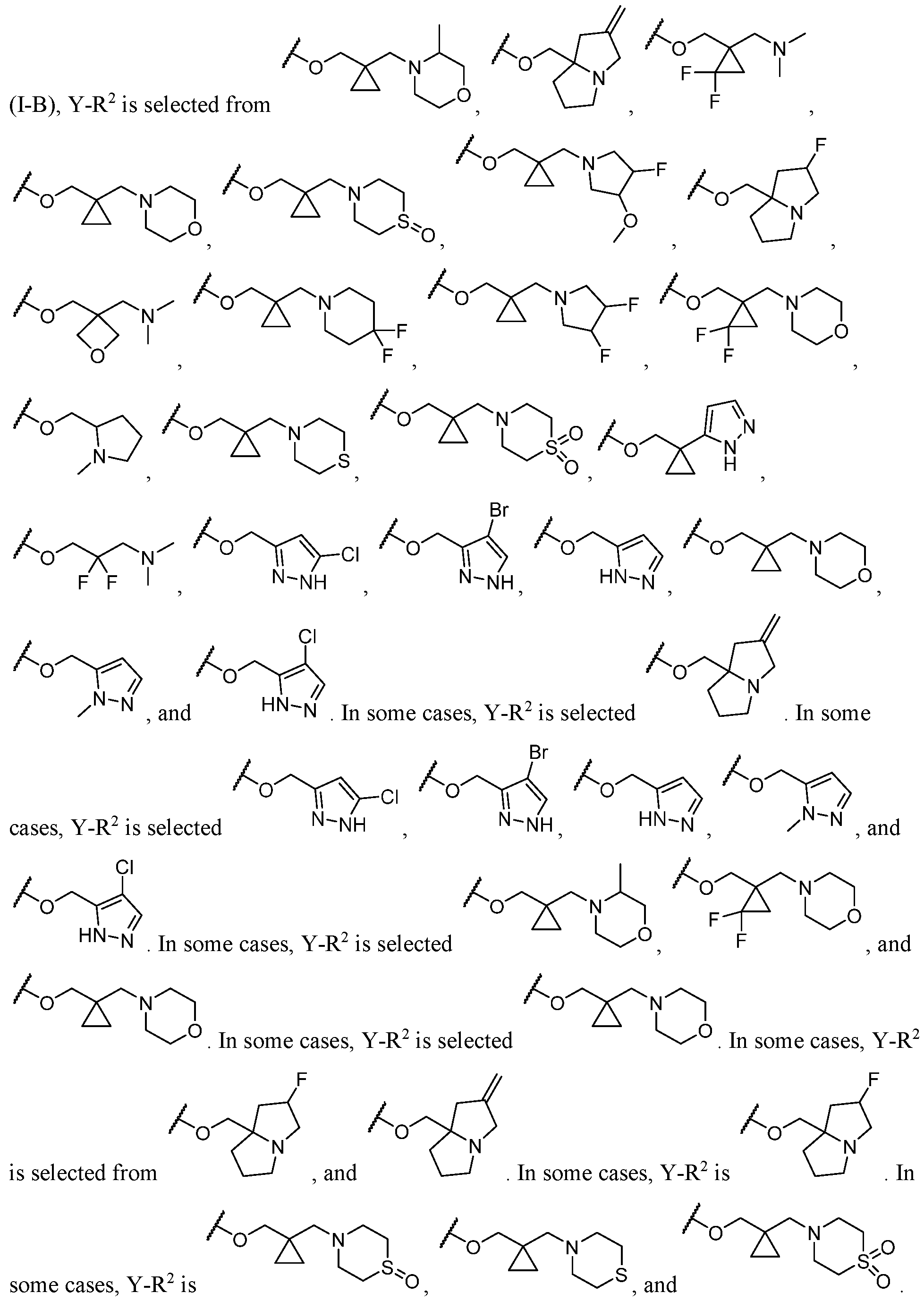
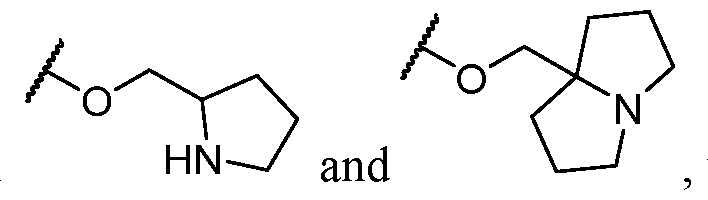
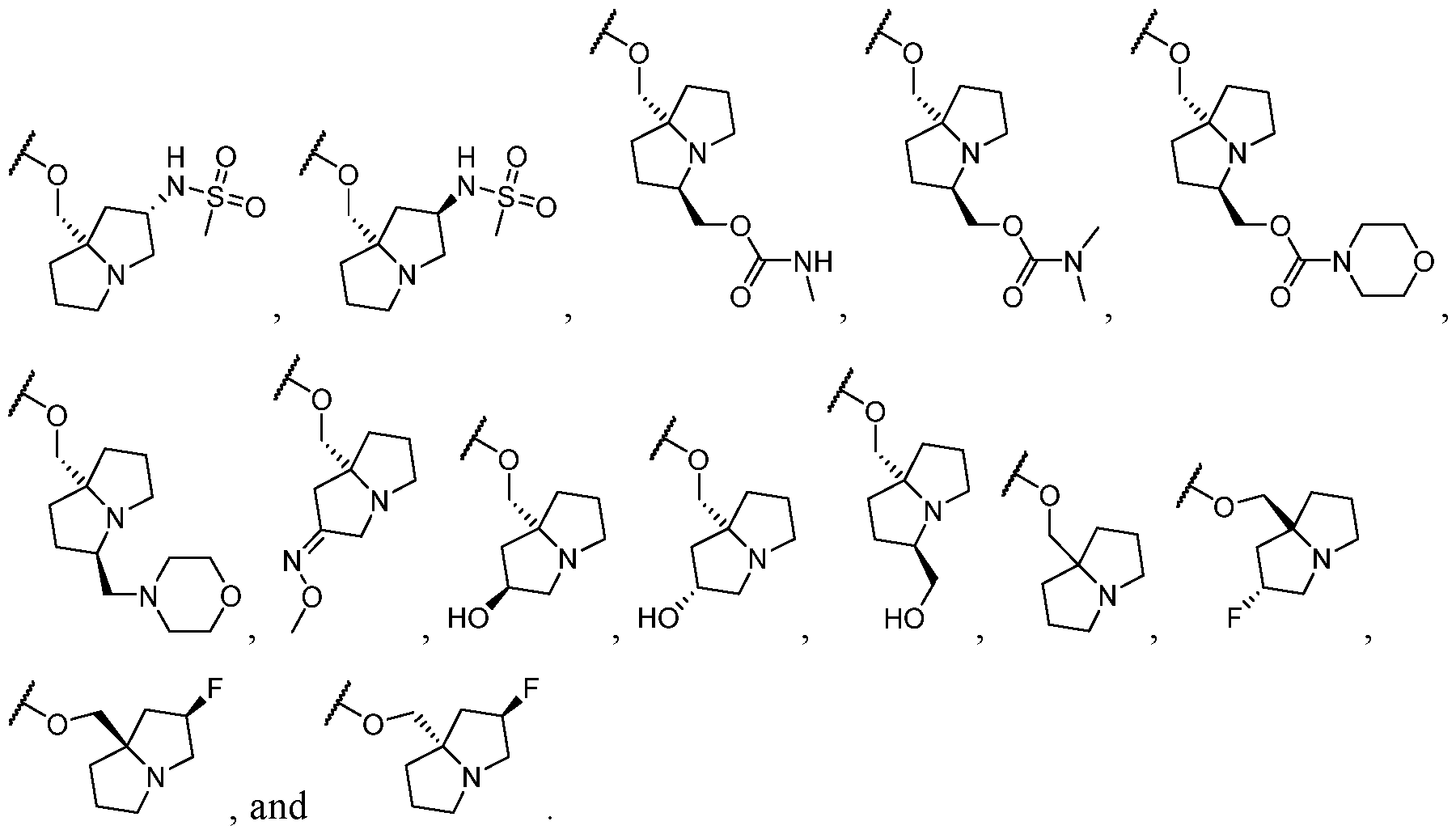
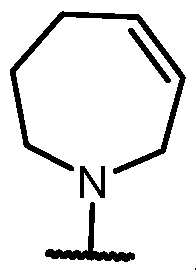
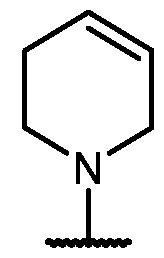
- Y-R 2 is selected from wherein the heterocycle portion is optionally substituted. In some cases, Y-R 2 is selected from , wherein the heterocycle portion is optionally substituted. In some cases, the heterocycle portion is optionally substituted with one or more substituents selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, -CN, and C1-C3 aminoalkyl.
- the heterocycle portion is optionally substituted with one or more substituents selected from halogen, hydroxy, -CN, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, and C1-C3 aminoalkyl.
- the heterocycle portion is optionally substituted with one or more substituents selected from C1-C3 alkyl and halogen.
- Y-R 2 is selected from In some cases, Y-R 2 is
- R 2 is selected from optionally substituted -L-heterocycle.
- the heterocycle is a bicyclic heterocycle.
- the heterocycle is a monocyclic heterocycle.
- the heterocycle has only 1 nitrogen atom.
- the heterocycle has only 1 nitrogen atom and no other heteroatoms.
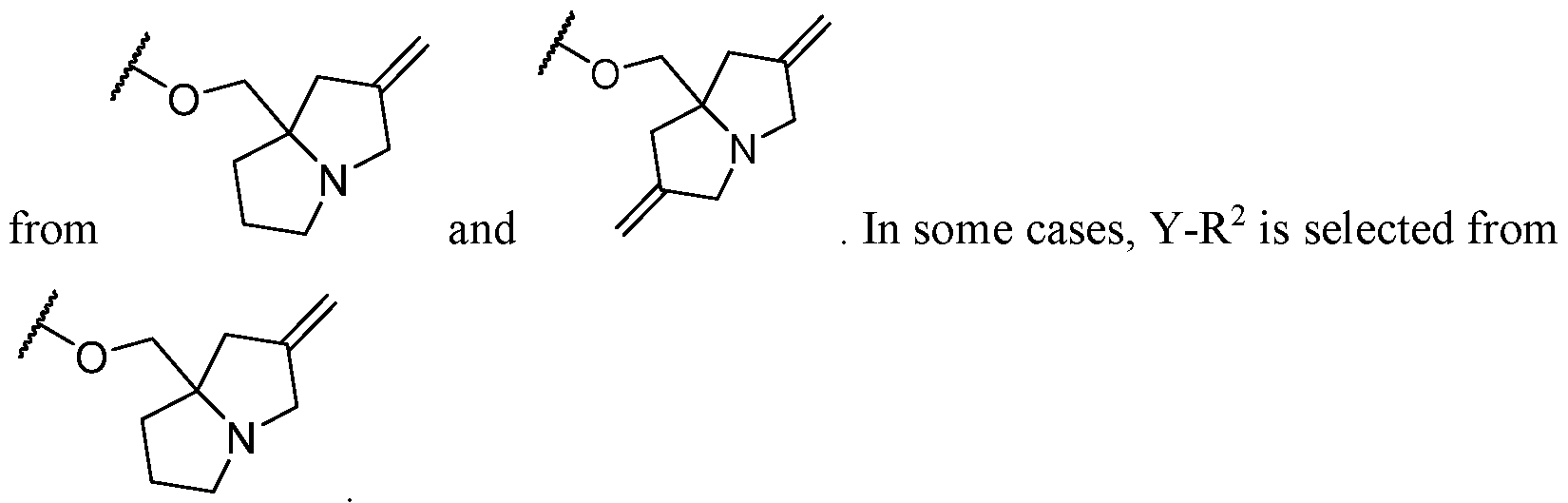
- Y-R 2 is selected from , wherein the heterocycle portion is optionally substituted.
- Y-R 2 is selected from wherein the heterocycle portion is optionally substituted.
- Y-R 2 is selected from , wherein the heterocycle portion is optionally substituted.
- Y-R 2 is selected from , wherein the heterocycle portion is optionally substituted.
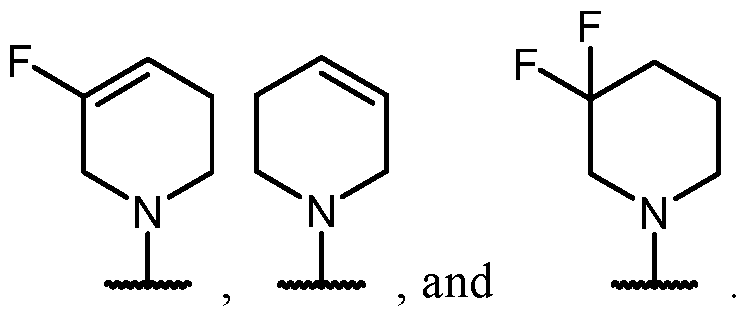
- the heterocycle is substituted with at least one halogen.
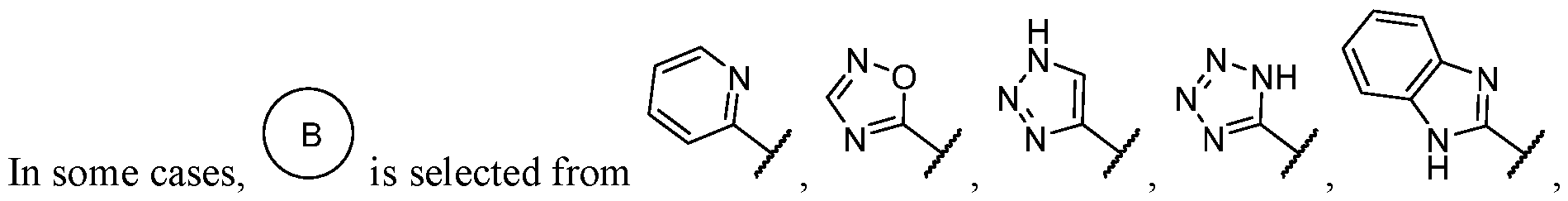
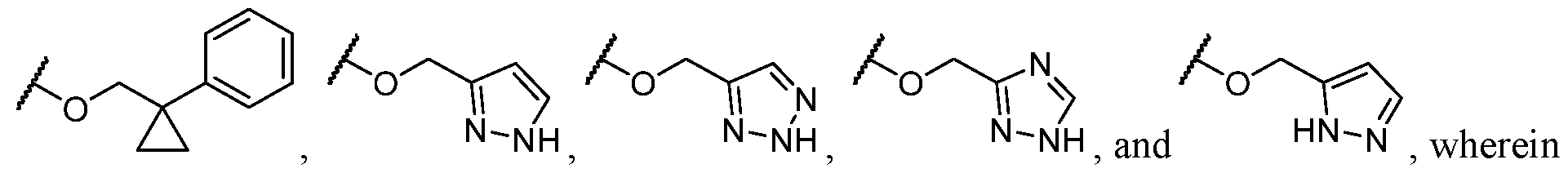
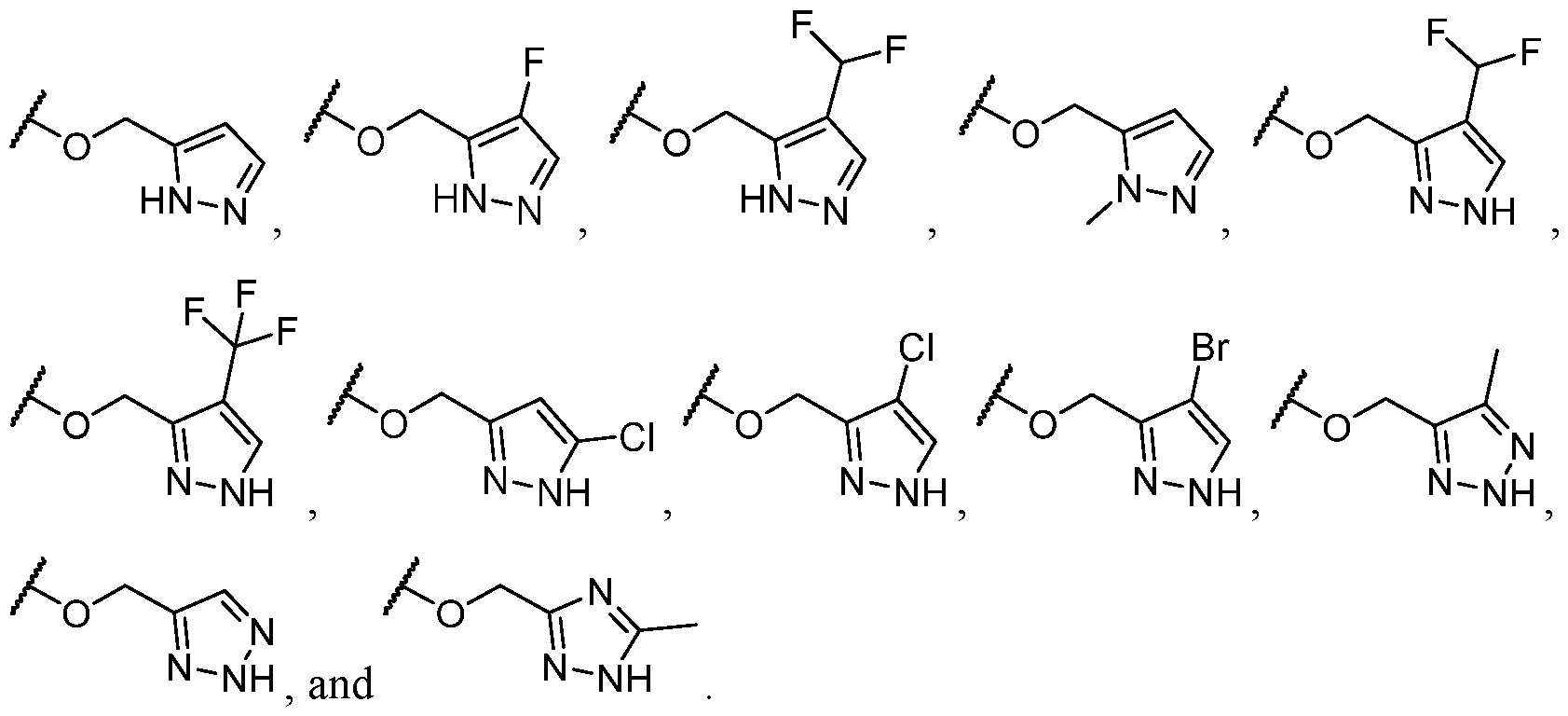
- R 2 is -L-heteroaryl, wherein the heteroaryl portion is optionally substituted with one or more R 7 .
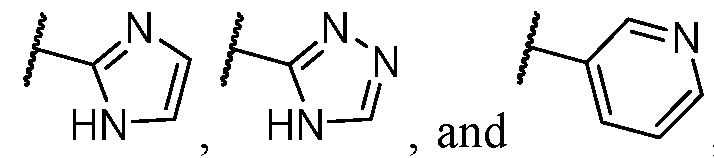
- the heteroaryl is selected from a 5- to 6-membered heteroaryl, wherein the heteroaryl portion is optionally substituted with one or more R 7 .
- the heteroaryl is selected from a 5- membered heteroaryl, wherein the heteroaryl portion is optionally substituted with one or more R 7 .
- the heteroaryl has at least one nitrogen atom.
- the heteroaryl has two nitrogen atoms.
- the heteroaryl has three nitrogen atoms.
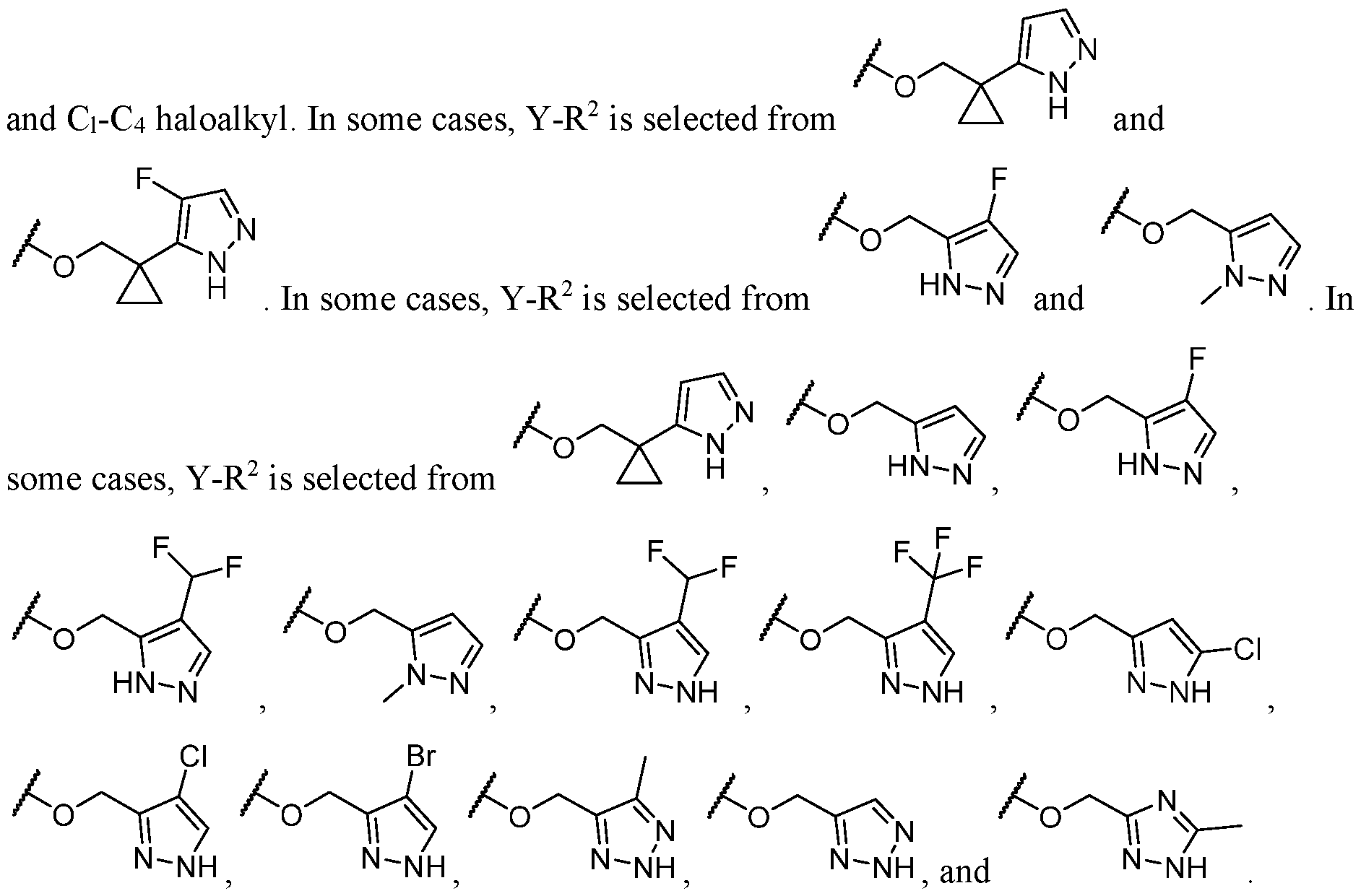
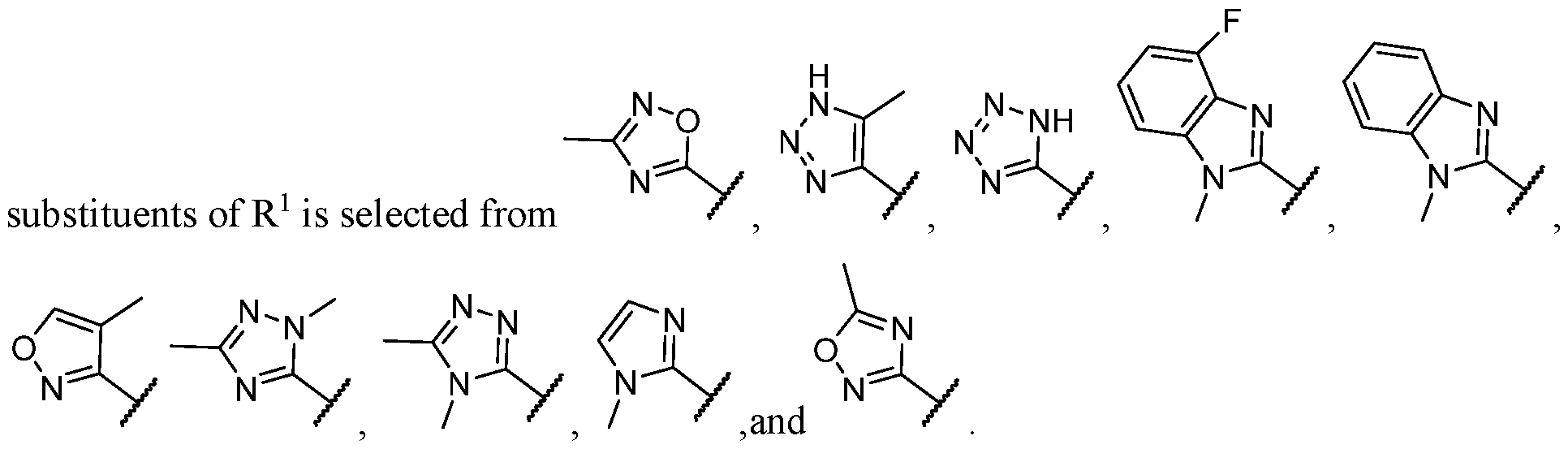
- the heteroaryl is selected from which is optionally substituted. In some cases, the heteroaryl is which is optionally substituted. In some cases, Y-R 2 is selected from , wherein the heteroaryl portion is optionally substituted with one or more R 7 . In some cases, each R 7 is independently selected from C1-C4 alkyl, halogen,
- R 2 is -L-aryl, optionally substituted with one or more R 7 .
- Y-R 2 is selected from wherein the heterocycle portion is optionally substituted with one or more R 7 .
- Y-R 2 is selected from wherein the heterocycle portion is optionally substituted with one or more R 7 .
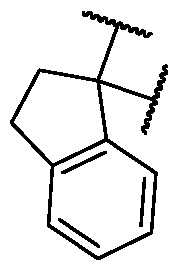
- R 2 is selected from [00142] In some embodiments, for a compound or salt of Formula (I), R 2 is -L-N(R 23 )2. In some cases, selected from
- R 2 is heterocycle, optionally substituted with one or more R 6 .
- the heterocycle which is optionally substituted.
- the heterocycle some cases, Y-R 2 is
- R 2 is selected from heterocycle, -L-heterocycle, wherein the heterocycle, and the heterocycle portion of -L- heterocycle, are each optionally substituted with one or more R 6 ; -L-aryl, and -L-heteroaryl, wherein the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R 7 ; and -L-N(R 23 )2.
- the heterocycle of R 2 is selected from wherein the heterocycle of R 2 is optionally substituted with one or more R 6 ; wherein the aryl and heteroaryl of R 2 is selected from from from
- -L-heterocycle are each optionally substituted with one or more R 6 ; the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with
- L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from hydroxy, C1-C4 hydroxy alkyl and C1-C4 alkyl. In some cases, L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from C1-C4 alkyl. In some cases, L is selected from C1-C4 alkylene. In some cases, L is selected from C1-C2 alkylene. In some cases, L is In some
- the optional substituents of L are selected from C1-C4 hydroxyalkyl, C1-C4 alkyl, C3-C6 carbocycle; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle wherein the C3-C6 carbocycle and 3- to 8- membered heterocycle are optionally substituted with one or more substituents selected from halogen and Ci-6 haloalkyl.
- each L is independently selected from a substituted C1-C4 alkylene, wherein two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle.
- each L is independently selected from a substituted C1-C4 alkylene, and two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle.
- each L is independently selected from a substituted C3 alkylene, and wherein two substituents on the same carbon atom of L come together to form a C3 carbocycle.
- each L is independently selected from
- R 2 is selected from -L- heterocycle, wherein the heterocycle portion of -L-heterocycle is optionally substituted with one or more R 6 .
- the heterocycle is a saturated heterocycle.
- the heterocycle has at least one nitrogen atom and at least one sulfur atom.
- the heterocycle has at least one nitrogen atom.
- the heterocycle has at least one sulfur atom.
- R 2 is selected from , wherein the heterocycle portion is optionally substituted with one or more R 6 .
- Y-R 2 is selected from , wherein the heterocycle portion is optionally substituted with one or more R 6 .
- Y-R 2 is selected from , wherein the heterocycle portion is optionally substituted with one or more R 6 .
- Y-R 2 is selected from , wherein the heterocycle portion is optionally substituted with one or more
- R 2 is selected from -L- saturated heterocycle, wherein the saturated heterocycle portion of the -L-saturated heterocycle is optionally substituted with one or more R 6 , and contains one nitrogen atom and one sulfur atom.
- Y-R 2 is selected from wherein the heterocycle portion is optionally substituted with one or more R 6 . In some cases, Y-R 2 is selected from , wherein the heterocycle portion is optionally substituted with one or more substituents selected from C1-C3 alkyl and oxo. In some cases, Y-R 2 is selected from cases, Y-R 2 is selected from
- Y is a bond.
- R 2 is selected from an optionally substituted heteroaryl and optionally substituted aryl.
- R 2 is selected from an optionally substituted heteroaryl.
- the heteroaryl has at least one nitrogen atom.
- the heteroaryl has at least two nitrogen atoms.
- the heteroaryl only contain nitrogen atom(s).
- the heteroaryl is a 6- membered heteroaryl.
- the heteroaryl is a 5-membered heteroaryl.
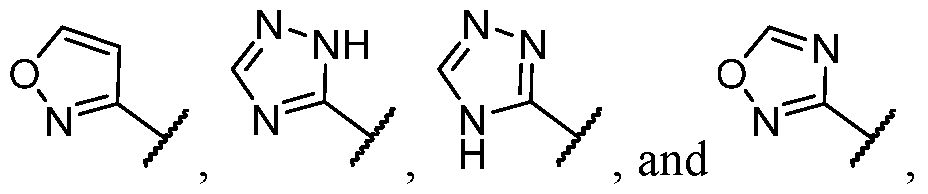
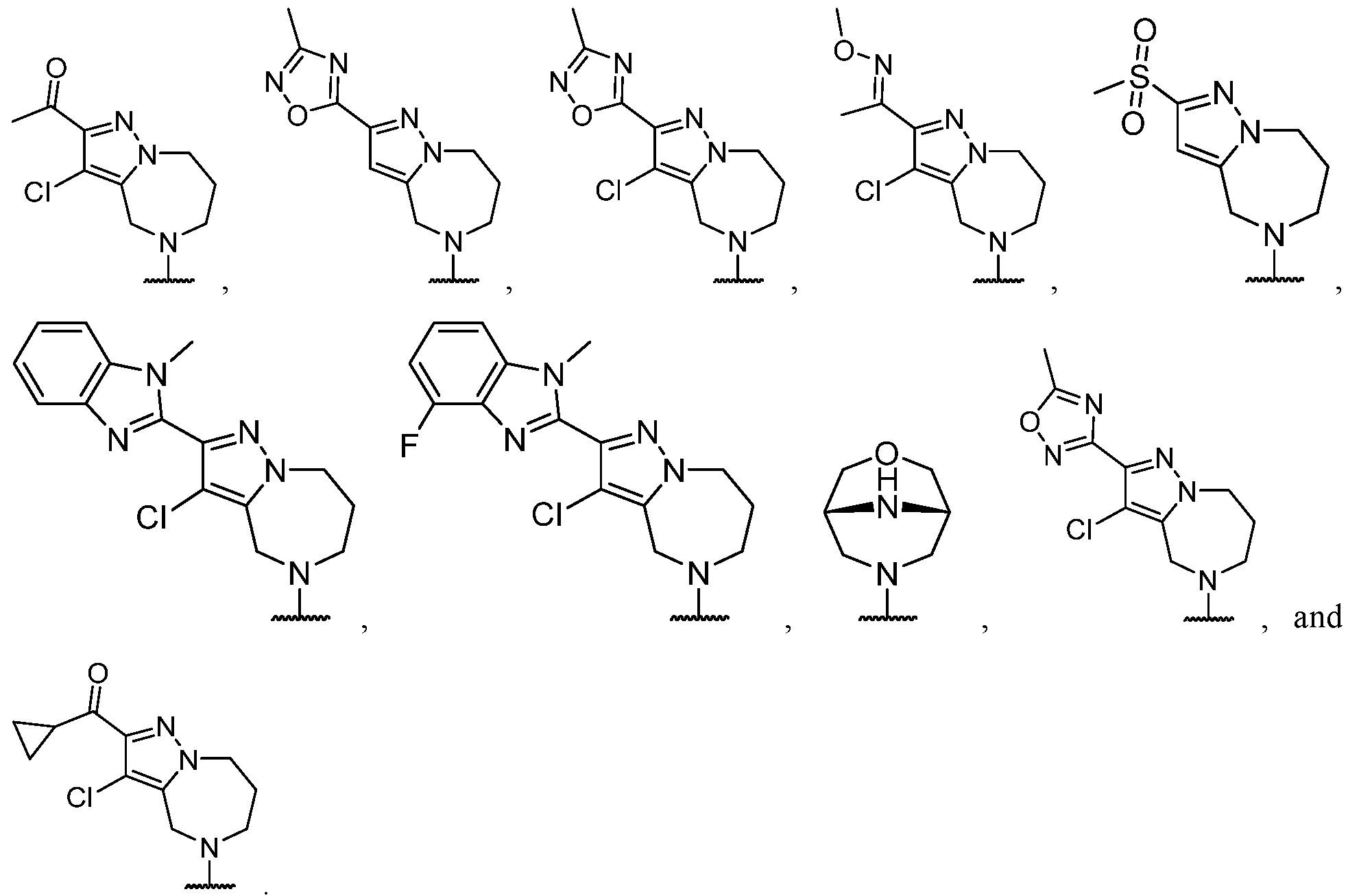
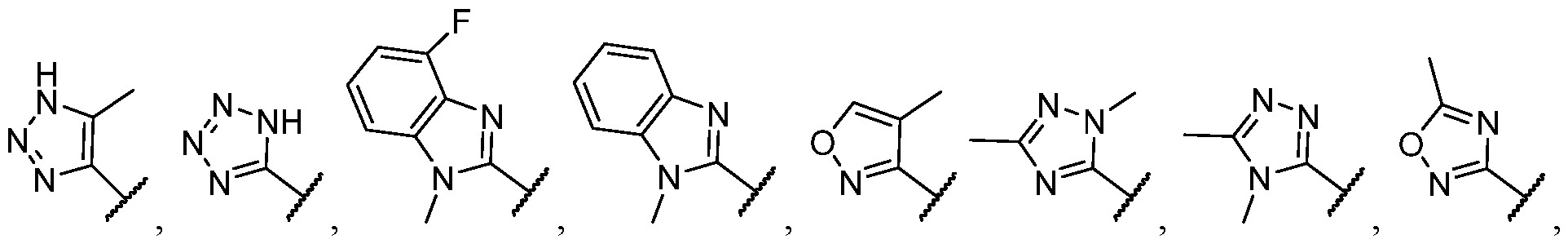
- the heteroaryl is selected from each of which is optionally substituted.
- the heteroaryl is selected from each of which is optionally substituted.
- R 2 is selected from an optionally substituted aryl.
- the aryl is a phenyl.
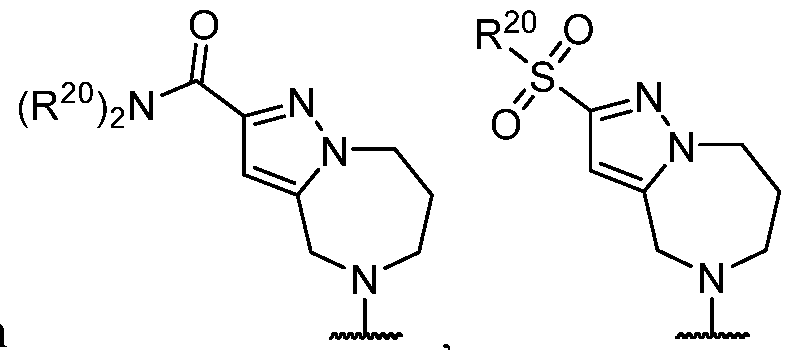
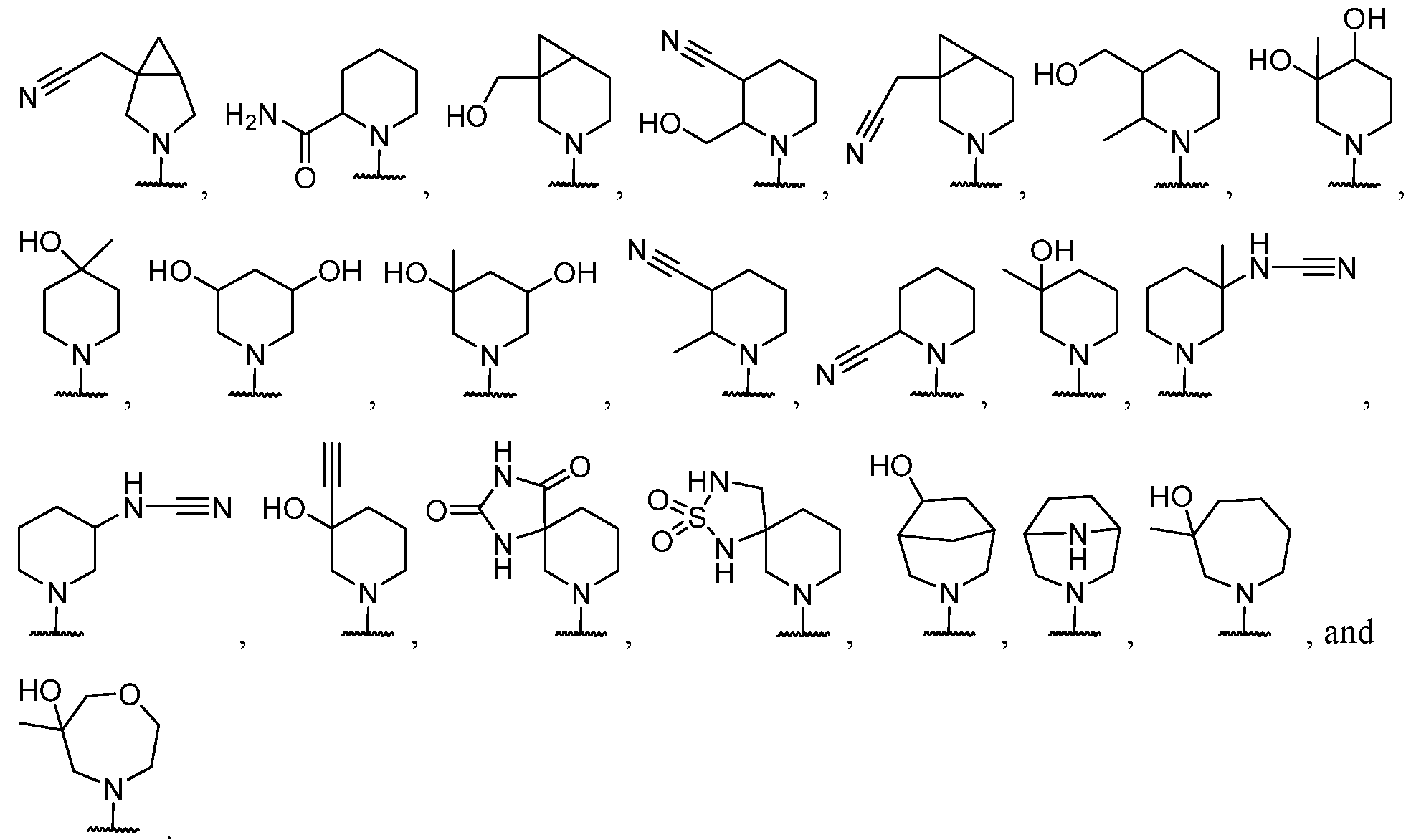
- the heteroaryl is optionally substituted with one or more R 6 , wherein each R 6 is selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, -CHJieterocycle, -C1-C3 alkyl-N(R 5 )2, and -C(O)N(R 5 )2.
- R 6 is selected from C1-C3 alkyl, -CH2heterocycle, and -C(O)N(R 5 )2.
- the aryl is optionally substituted with one or more R 7 .
- Y-R 2 is selected from
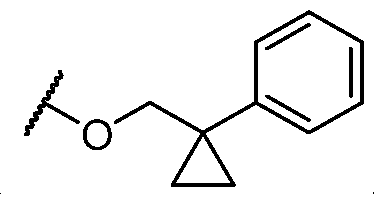
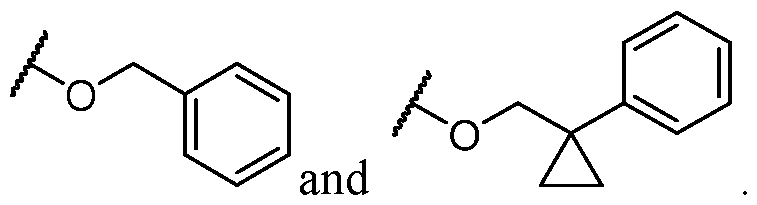
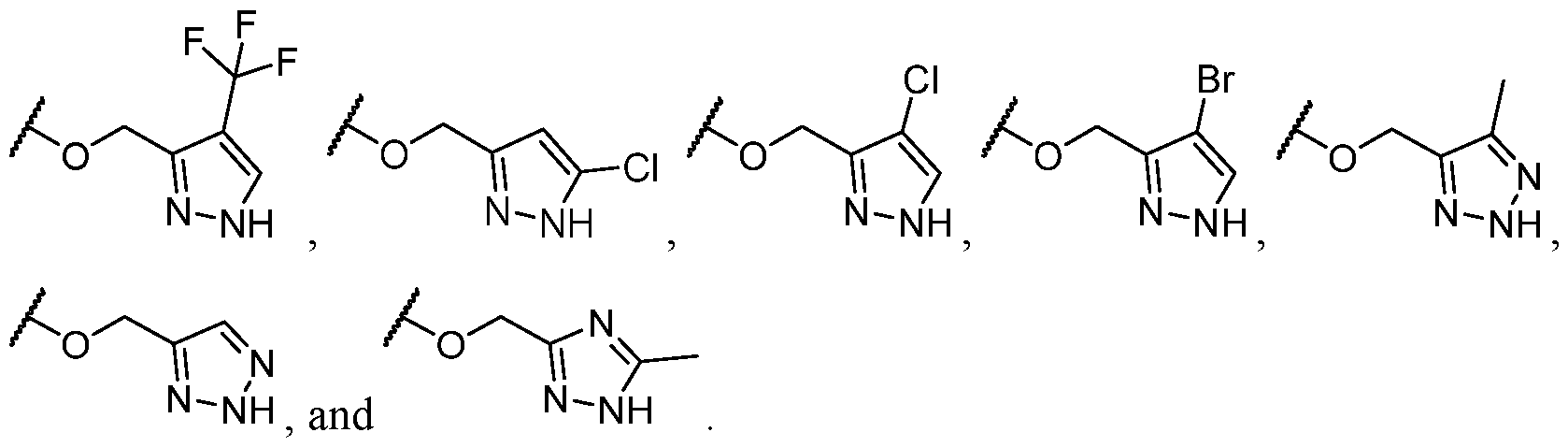
- Y is -O- and R 2 is selected from L-5-membered heteroaryl.
- the heteroaryl has at least 1 nitrogen atom.
- the heteroaryl has at least two nitrogen atoms.
- the heteroaryl has 3 nitrogen atoms.
- L is selected from an optionally substituted C1-C4 alkylene.
- L is independently selected from a C1-C4 alkylene optionally substituted; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents selected from halogen,
- L is selected from In some cases, L is selected from . In some cases, the heteroaryl is optionally substituted with one or more R 7 . In some cases, each R 7 is selected from halogen, C1-C4 alkyl, and C1-C4 haloalkyl. In some cases, Y-R 2 is selected from
- each R 6 is independently selected from halogen, -OH, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, -CN, and C1-C3 aminoalkyl. In some cases, each R 6 is independently selected from halogen, C1-C3 alkyl, and C1-C3 haloalkyl.
- each R 6 is independently selected from halogen, -OH, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 aminoalkyl, C1-C3 haloalkyl, C1-C3 alkoxy, -N(R 5 )2, and oxo. In some cases, each R 6 is independently selected from -OH, Ci- C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 aminoalkyl, C1-C3 alkoxy, and -N(R 5 )2.
- R 6 is selected from halogen, -OH, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, -CN, and C1-C3 aminoalkyl. In some cases, R 6 is selected from halogen and C1-C3 alkyl. In some cases, R 6 is halogen. In some cases, R 6 is C1-C3 alkyl. In some cases, R 6 is selected from halogen and C1-C3 alkyl. In some cases, R 6 is selected from methyl and fluorine.
- R 2 is selected from
- Y-R 2 is selected from
- Y-R 2 is selected from
- Y-R 2 is
- L is selected from unsubstituted C1-C4 alkylene.
- Y-R 2 is selected from , wherein the heterocycle portion is optionally substituted with one or more R 6 .
- R 6 of R 2 is independently selected at each occurrence from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, and C1-C3 aminoalkyl.
- R 6 of R 2 is independently selected at each occurrence from C1-C3 alkyl and halogen.
- Y-R 2 is selected from
- R 1 is selected from an optionally sub stituted 5- to 12-membered heterocycle.
- the heterocycle of R 1 is selected from a 5- to 12-membered heterocycle, 6- to 12-membered heterocycle, 7- to 12- membered heterocycle, and 8- to 12-membered heterocycle.
- the heterocycle of R 1 is selected from a 5- to 11 -membered heterocycle, 5- to 10-membered heterocycle, 5- to 9- membered heterocycle, and 5- to 8-membered heterocycle.
- the heterocycle of R 1 is selected from a 6- to 11 -membered heterocycle, 6- to 10-membered heterocycle, 6- to 9- membered heterocycle, and 6- to 8-membered heterocycle. In some cases, the heterocycle of R 1 is selected from a 7- to 11 -membered heterocycle, 7- to 10-membered heterocycle, 7- to 9- membered heterocycle, and 7- to 8-membered heterocycle. In some cases, the heterocycle of R 1 is selected from a 5- to 6-membered heterocycle and 5- to 9-membered heterocycle. In some cases, the heterocycle of R 1 is selected from an 8- to 9-membered heterocycle.
- R 1 is selected from an optionally substituted 5- to 7-membered heterocycle. In some cases, R 1 is selected from an optionally substituted 6- to 7-membered heterocycle. In some cases, R 1 is selected from an optionally substituted 7-membered heterocycle. In cases, the 5- to 12- membered heterocycle of R 1 is a bridged heterocycle. In cases, the 5- to 12-membered heterocycle of R 1 is not a bridged heterocycle. In some cases, the heterocycle of R 1 is saturated. In some cases, the heterocycle of R 1 is unsaturated. In some cases, the heterocycle of R 1 is an unbridged heterocycle. The heterocycle of R 1 is optionally substituted as described elsewhere herein.
- the heterocycle of R 1 contains at most 1 nitrogen atom. In some embodiments, the heterocycle of R 1 contains at most 1 heteroatom atom. In some cases, the heteroatom is selected from nitrogen, oxygen, and sulfur. In some embodiments, the heterocycle of R 1 contains at most 2 heteroatom atoms. In some cases, the heterocycle of R 1 contains 1 nitrogen atom. In some cases, the heterocycle of R 1 contains only 1 nitrogen atom. In some cases, the heterocycle of R 1 contains only 1 nitrogen atom and no other heteroatoms.
- the 5- to 12-membered heterocycle of R 1 is an unsaturated heterocycle.
- the 5- to 12-membered heterocycle of R 1 is selected from a saturated heterocycle and unsaturated heterocycle.
- the 5- to 12-membered heterocycle of R 1 is a saturated heterocycle.
- the 5- to 12-membered heterocycle of R 1 is an unsaturated heterocycle.
- R 1 is selected from 5- to 15-membered heterocycle, wherein the 5- to 15-membered heterocycle is optionally substituted. In some cases, R 1 is selected from an optionally substituted 6- to 15-membered heterocycle. In some cases, the heterocycle is a spiro heterocycle. In some cases, the heterocycle is a fused heterocycle. In some cases, the heterocycle is a bridged heterocycle. In some cases, the heterocycle is an unsaturated heterocycle.
- R 1 is a 6- to 12- membered fused heterocycle, which is optionally substituted.
- R 1 is a 6- to 12- membered spiroheterocycle, which is optionally substituted. In some cases, R 1 is selected from optionally substituted 7- to 8-membered spiroheterocycle.
- the heterocycle of R 1 is a 5- to 12-membered heterocycle, 6- to 12-membered heterocycle, 7- to 12-membered heterocycle, or 8- to 12-membered heterocycle.
- the heterocycle of R 1 is a 5- to 11 -membered heterocycle, 5- to 10-membered heterocycle, 5- to 9-membered heterocycle, or 5- to 8-membered heterocycle.
- the heterocycle of R 1 is a 6- to 11 -membered heterocycle, 6- to 10-membered heterocycle, 6- to 9-membered heterocycle, or 6- to 8-membered heterocycle.
- the heterocycle of R 1 is a 7- to 11-membered heterocycle, 7- to 10- membered heterocycle, 7- to 9-membered heterocycle, or 7- to 8-membered heterocycle.
- the heterocycle of R 1 is a 5- to 6-membered heterocycle or 5- to 9-membered heterocycle.
- the heterocycle of R 1 is an 8- to 9-membered heterocycle.
- the heterocycle of R 1 is saturated. The heterocycle is optionally substituted as described elsewhere herein.
- R 1 is a 5- to 12- membered monocyclic heterocycle.
- the heterocycle of R 1 is a 5- to 12-membered monocyclic heterocycle, 6- to 12-membered monocyclic heterocycle, 7- to 12-membered monocyclic heterocycle, or 8- to 12-membered monocyclic heterocycle.
- the heterocycle of R 1 is a 5- to 11 -membered monocyclic heterocycle, 5- to 10-membered monocyclic heterocycle, 5- to 9-membered monocyclic heterocycle, or 5- to 8-membered monocyclic heterocycle.
- the heterocycle of R 1 is a 6- to 11 -membered monocyclic heterocycle, 6- to 10-membered monocyclic heterocycle, 6- to 9-membered monocyclic heterocycle, or 6- to 8-membered monocyclic heterocycle.
- the heterocycle of R 1 is a monocyclic 7- to 11 -membered heterocycle, 7- to 10-membered monocyclic heterocycle, 7- to 9-membered monocyclic heterocycle, or 7- to 8-membered monocyclic heterocycle.
- the heterocycle of R 1 is a 5- to 6-membered monocyclic heterocycle or 5- to 9-membered monocyclic heterocycle.
- the heterocycle of R 1 is an 8- to 9-membered monocyclic heterocycle.
- the heterocycle of R 1 is saturated.
- the monocyclic heterocycle is optionally substituted as described elsewhere herein.
- R 1 is a bridged heterocycle.
- the heterocycle of R 1 is a 5- to 12-membered bridged heterocycle, 6- to 12-membered bridged heterocycle, 7- to 12-membered bridged heterocycle, or 8- to 12- membered bridged heterocycle.
- the heterocycle of R 1 is a 5- to 11 -membered bridged heterocycle, 5- to 10-membered bridged heterocycle, 5- to 9-membered bridged heterocycle, or 5- to 8-membered bridged heterocycle.
- the heterocycle of R 1 is a 6- to 11 -membered bridged heterocycle, 6- to 10-membered bridged heterocycle, 6- to 9- membered bridged heterocycle, or 6- to 8-membered bridged heterocycle.
- the heterocycle of R 1 is a bridged 7- to 11 -membered heterocycle, 7- to 10-membered bridged heterocycle, 7- to 9-membered bridged heterocycle, or 7- to 8-membered bridged heterocycle.
- the heterocycle of R 1 is a 5- to 6-membered bridged heterocycle or 5- to 9- membered bridged heterocycle.
- the heterocycle of R 1 is an 8- to 9-membered bridged heterocycle.
- the heterocycle of R 1 is saturated.
- the bridged heterocycle is selected from In some cases, the bridged heterocycle is selected from . Each bridged heterocycle is optionally substituted as described elsewhere herein.
- R 1 is a spiro heterocycle.
- the spiro heterocycle of R 1 is a 7- to 12-membered spiro heterocycle, 7- to 12- membered spiro heterocycle, or 8- to 12-membered spiro heterocycle.
- the spiro heterocycle of R 1 is a 7- to 11 -membered spiro heterocycle, 7- to 10-membered spiro heterocycle, 7- to 9-membered spiro heterocycle, or 7- to 8-membered spiro heterocycle.
- the spiro heterocycle of R 1 is a 7- to 11 -membered spiro heterocycle, 7- to 10-membered spiro heterocycle, 7- to 9-membered spiro heterocycle, or 7- to 8-membered spiro heterocycle.
- the spiro heterocycle of R 1 is a 7- to 11 -membered spiro heterocycle.
- the spiro heterocycle of R 1 is a 7-membered spiro heterocycle.
- the spiro heterocycle of R 1 is an 8-membered spiro heterocycle.
- the spiro heterocycle of R 1 is a 9-membered spiro heterocycle. In some cases, the spiro heterocycle of R 1 is a 10-membered spiro heterocycle. In some cases, the spiro heterocycle of R 1 contains at most 1 nitrogen atom. In some cases, the spiro heterocycle of R 1 contains only 1 nitrogen atom. In some cases, the spiroheterocycle of R 1 contains at most 2 heteroatom atoms. In some cases, the spiro heterocycle of R 1 contains at least 2 heteroatom atoms. In some cases, the spiro heterocycle of R 1 contains at least 3 heteroatom atoms. In some cases, the heteroatom is selected from nitrogen, oxygen, and sulfur. In some cases, the spiroheterocycle of R 1 is bound to the Formula via the nitrogen atom. heterocycle of R 1 is selected from . Each spiro heterocycle is optionally substituted as described elsewhere herein.
- R 1 is a fused heterocycle.
- the fused heterocycle of R 1 is a 6- to 12-membered fused heterocycle, 6- to 12-membered fused heterocycle, 7- to 12-membered fused heterocycle, or 8- to 12-membered fused heterocycle.
- the fused heterocycle of R 1 is a 6- to 11- membered fused heterocycle, 6- to 10-membered fused heterocycle, 6- to 9-membered fused heterocycle, or 6- to 8-membered fused heterocycle.
- the fused heterocycle of R 1 is a 7- to 11 -membered fused heterocycle, 7- to 10-membered fused heterocycle, 7- to 9-membered fused heterocycle, or 7- to 8-membered fused heterocycle.
- the fused heterocycle of R 1 is an 8- to 11-membered fused heterocycle.
- the fused heterocycle of R 1 is a 9-membered fused heterocycle.
- the fused heterocycle of R 1 is a 10-membered fused heterocycle.
- the fused heterocycle of R 1 is an 11 -membered fused heterocycle.
- the fused heterocycle of R 1 is a 6-membered fused heterocycle. In some cases, the fused heterocycle of R 1 is a 7-membered fused heterocycle. In some cases, the fused heterocycle of R 1 is a 10-membered fused heterocycle. In some cases, the fused heterocycle is selected from Each fused heterocycle is optionally substituted as described elsewhere herein.
- R 1 is selected from an optionally substituted 8- to 10-membered fused heterocycle.
- the 8- to 10- membered fused heterocycle is a bicyclic heterocycle.
- the 8- to 10-membered fused heterocycle is a saturated heterocycle.
- the 8- to 10-membered fused heterocycle is an unsaturated heterocycle.
- the 8- to 10-membered heterocycle is a non-aromatic heterocycle.
- R 1 is selected from an optionally substituted 9- membered fused heterocycle.
- R 1 is selected from an optionally substituted 10- membered fused heterocycle.
- the 10-membered fused heterocycle is a bicyclic heterocycle. In some cases, the 10-membered fused heterocycle is a saturated heterocycle. In some cases, the 9-membered heterocycle is a non-aromatic heterocycle. In some cases, the 10- membered heterocycle is a non-aromatic heterocycle. In some cases, the fused heterocycle has one saturated ring and one aromatic ring. In some cases, the fused heterocycle has one saturated ring and one unsaturated ring. In some cases, the fused heterocycle has two saturated rings. In some cases, the 10-membered heterocycle contains at least 1 nitrogen atom. In some cases, the 10-membered heterocycle contains at least 2 nitrogen atoms.
- the 10-membered heterocycle contains at least 3 nitrogen atoms. In some cases, the 9-membered heterocycle contains at least 1 nitrogen atom. In some cases, the 9-membered heterocycle contains at least 2 nitrogen atoms. In some cases, the 9-membered heterocycle contains at least 3 nitrogen atoms. In some cases, R 1 is selected from , each of which is optionally substituted with one or more substituents. In some cases, which is optionally substituted with one or more substituents. In some cases, , which is optionally substituted with one or more substituents.
- Ci-6 alkyl-N(R 20 )2 Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, Ci-
- R 1 6 haloalkyl, C1-6 alkyl, C2-6 alkynyl, and 5- to 12-membered heterocycle, wherein the 5- to 12- membered heterocycle are each optionally substituted independently with one or more R 1 *.
- R 1 is selected from
- the further one or more optional substituents are selected from halogen, -CN, C2 alkenyl, and C1-6 alkyl.
- the further one or more optional substituents are selected from halogen, and C1-6 alkyl.
- the further one or more optional substituents are selected from halogen.
- each R 20 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle. In some cases, each R 20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12- membered heterocycle. In some cases, each R 20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12-membered saturated heterocycle. In some cases, each R 20 is independently selected from 5- to 6-membered saturated heterocycle. In some cases, the heterocycle of R 20 has at least one nitrogen atom. In some cases, the heterocycle of R 20 has at least one sulfur atom. In some cases, the heterocycle of R 20 has at least one oxygen atom. In some cases, the heterocycle of R 20 contains only 1 heteroatom.
- the heterocycle of R 20 has at least two heteroatoms. In some cases, the heterocycle of R 20 contains only 2 heteroatoms. In some cases, the optional one or more substituents of R 1 are independently selected from halogen, -CN, C2 alkenyl, some cases, the optional one or more substituents of R 1 are independently selected from halogen, more substituents of R 1 are independently selected from halogen, and Ci-6 alkyl-N(R 20 )2. In some cases, the optional one or more substituents of R 1 are independently selected from halogen, , hydrogen, Ci-6 alkyl, and C3-6 carbocycle.
- R 1 is selected from a
- R B is selected from a 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted independently with one or more R 1 * ; and R B is selected from hydrogen, halogen, Ci-6 alkyl, Ci-6 haloalkyl, C2-6 alkynyl, and -CN.
- R B is selected from hydrogen, and halogen.
- R B is chloride.
- R B is hydrogen.
- has at least 1 oxygen atom. is a monocyclic heterocycle.
- each R 1 * is independently selected from halogen, -OR 20 , -S(O) 2 (R 20 ), -
- each R 1 * is independently selected from halogen, Ci-6 alkyl-N(R 20 ) 2 , Ci-6 aminoalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, and Ci-6 alkyl.
- each R 1 * is independently selected from halogen, and Ci-6 alkyl. In some cases, is selected from
- R 1 is selected from 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted with one or more substituents.
- R 1 is selected from a saturated 5- to 12-membered heterocycle, which is optionally substituted with one or more substituents. In some cases, the 5- to 12-membered heterocycle of R 1 is bridged. In some cases, the 5- to 12-membered heterocycle of R 1 is not bridged. In some cases, the 5- to 12-membered substituents. [00189] In some embodiments, for a compound or salt of Formula (I), R 1 is selected from , each of which is optionally substituted with one or more substituents.
- R 1 is
- R 1 is selected from an optionally substituted 5- to 12-membered unsaturated heterocycle, wherein the heterocycle has as most one nitrogen atom. In some cases, the 5- to 12-membered unsaturated heterocycle has at least one nitrogen atom. In some cases, the 5- to 12-membered unsaturated heterocycle has at most one nitrogen atom.
- the heterocycle of R 1 contains only 1 nitrogen atom and optionally one or more heteroatoms selected from oxygen, and sulfur.
- the heterocycle is a fused heterocycle or a bridged heterocycle.
- the heterocycle is a monocyclic heterocycle or a bridged heterocycle.
- the heterocycle is a monocyclic heterocycle.
- the heterocycle is a bridged heterocycle.
- the heterocycle is selected from h e t er0C y Cie j s optionally substituted as described elsewhere herein.
- the heterocycle of R 1 has at most 1 nitrogen atom. In some cases, the heterocycle of R 1 has only 1 nitrogen atom and optionally one or more other heteroatoms selected from oxygen and sulfur. In some cases, the heterocycle of R 1 has only 1 nitrogen atom and no other heteroatoms.
- R 1 is selected from an optionally substituted 5- to 12-membered saturated heterocycle, wherein the heterocycle has as most one nitrogen atom.
- the 5- to 12-membered unsaturated heterocycle has at least one nitrogen atom.
- the 5- to 12-membered unsaturated heterocycle has only one nitrogen atom and 0-2 other heteroatoms selected from nitrogen, oxygen, and sulfur.
- the 5- to 12-membered unsaturated heterocycle has only one nitrogen atom and no further heteroatoms.
- the 5- to 12-membered unsaturated heterocycle has three nitrogen atoms and no further heteroatoms.
- R 1 is selected from an optionally substituted 5- to 12-membered unsaturated heterocycle, wherein the heterocycle has as most one nitrogen atom. In some cases, the 5- to 12-membered unsaturated heterocycle has at least one nitrogen atom. In some cases, the 5- to 12-membered unsaturated heterocycle has only one nitrogen atom and no further heteroatoms.
- R 1 is selected from 6- to 7-membered heterocycle. In some cases, R 1 is selected from 7-membered heterocycle. In some cases, R 1 is selected from 6-membered heterocycle. In some cases, the 6- to 7-membered heterocycle contains only 1 nitrogen atom and optionally one or more additional heteroatoms selected from oxygen, and sulfur. In some cases, the optionally one or more additional heteroatoms are selected from sulfur. In some cases, the optionally one or more additional heteroatoms are selected from oxygen. In some cases, the 6- to 7-membered heterocycle contains only 1 nitrogen atom and no further additional heteroatoms. In some cases, the 6- to 7-membered heterocycle is a non-aromatic 6- to 7-membered heterocycle. In some cases, the 6- to 7-membered heterocycle of
- R 1 is bound to Formula (I) via the only 1 nitrogen atom.
- R 1 is selected which is substituted.
- R 1 is selected from selected from each of which is optionally substituted.
- the one or more optional substituents of R 1 are each independently selected from halogen, - OH, -CN, C1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of R 1 are each independently selected from halogen, -OH, and -CN. In some cases, the one or more optional substituents of R 1 are each independently selected from fluorine, -
- R 1 is selected from
- R 1 is selected
- R 1 is selected from an optionally substituted unsaturated 6- to 8-membered heterocycle. In some cases, R 1 is selected from an optionally substituted unsaturated 6-membered heterocycle. In some cases, R 1 is selected from an optionally substituted unsaturated 7-membered heterocycle. In some cases, the heterocycle has 1 or 2 double bonds. In some cases, the heterocycle has only 1 double bond. In some cases, the heterocycle has only 2 double bonds.
- R 1 is selected from wherein each is optionally substituted with one or more substituents independently selected from halogen, -OH, -NH2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl. In some cases, R 1 is selected from wherein each is optionally substituted with one or more substituents independently selected from halogen, -OH, -NH2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl.
- R 1 is selected from more substituents independently selected from halogen, -OH, -NH2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl. In some cases, R 1 is selected from , wherein each is substituted with one or more substituents independently selected from halogen.
- R 1 is selected from an unsaturated 6- to 7-membered heterocycle, wherein the unsaturated 6- to 7-membered heterocycle is substituted with one or more substituents selected from halogen. In some cases, the unsaturated
- 6- to 7-membered heterocycle is substituted with at least one halogen. In some cases, the unsaturated 6- to 7-membered heterocycle is substituted with at only one halogen. In some cases, the unsaturated 7-membered heterocycle is substituted with one fluorine. In some cases, R 1 is selected from an unsaturated 6-membered heterocycle, substituted with at least one halogen. In some cases, R 1 is selected from an unsaturated 7-membered heterocycle, substituted with at least one halogen. In some cases, R 1 is selected from
- R 1 is selected from some cases, R 1 is
- R 1 is selected from an optionally substituted unsaturated 6- to 8-membered heterocycle. In some cases, R 1 is selected from an optionally substituted unsaturated 7-membered heterocycle. In some cases, R 1 is selected from , wherein each is optionally substituted with one or more substituents independently selected from halogen, -OH, -NH2, -NO2, C 1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, Ci-e haloalkyl, and C1-6 alkyl. In some cases, R4S selected from
- R 1 is selected from an optionally substituted 6-membered heterocycle.
- the 6-membered heterocycle contains only 1 nitrogen atom.
- the 6-membered heterocycle of R 1 is bound to
- R 1 is selected from of which is optionally substituted.
- the 6-membered heterocycle is a partially unsaturated 6-membered heterocycle or a saturated 6-membered heterocycle. In some cases, the 6-membered heterocycle is partially unsaturated. In some cases, the 6-membered heterocycle is a saturated 6-membered heterocycle. In some cases, the 6-membered heterocycle is a monocyclic 6- membered heterocycle. In some cases, the 6-membered heterocycle is not a bridged heterocycle.
- R 1 is selected from
- R 1 is selected from an optionally substituted 6-membered unsaturated heterocycle and 6-membered saturated heterocycle.
- R 1 is selected from , wherein each is optionally substituted with one or more substituents independently selected from halogen, -OH, -NH2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl.
- R 1 is selected from , wherein each is optionally substituted with one or more substituents independently selected from halogen, and Ci-e haloalkyl.
- R 1 is selected from [00204] in some embodiments, for a compound or salt of Formula (I), R 1 is selected from [00205] In some embodiments, for a compound or salt of Formula (I), R 1 is selected from , wherein each is optionally substituted two substituents independently selected from halogen, -OH, -NH2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl.
- R 1 is selected from , wherein each is optionally substituted with two substituents independently selected from halogen, and Ci-6 haloalkyl. In some cases,
- R 1 is selected from an optionally substituted 6- to 10-membered heterocycle.
- the 6- to 10-membered heterocycle contains at least 1 nitrogen atom.
- each R 20 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle.
- R 1 is selected from
- R 1 is selected from 6- to 7-membered heterocycle. In some cases, R 1 is selected from 7-membered heterocycle. In some cases, R 1 is selected from 6-membered heterocycle. In some cases, the 6- to 7-membered heterocycle contains only 1 nitrogen atom and optionally one or more additional heteroatoms selected from oxygen, and sulfur. In some cases, the optionally one or more additional heteroatoms are selected from sulfur. In some cases, the optionally one or more additional heteroatoms are selected from oxygen. In some cases, the 6- to 7-membered heterocycle contains only 1 nitrogen atom and no further additional heteroatoms. In some cases, the 6- to 7-membered heterocycle is a non-aromatic 6- to 7-membered heterocycle. In some cases, the 6- to 7-membered heterocycle of
- R 1 is bound to Formula (I) via the only 1 nitrogen atom.
- R 1 is selected from -TML ⁇ - cases, R 1 is selected from each of which is substituted.
- R 1 is selected from selected from each of which is optionally substituted.
- the one or more optional substituents of R 1 are each independently selected from fluorine, -OH, -C(0)NH2, -NH-C(0)-(CI-6 alkoxy), -NH-C(0)-(CI-6 hydroxyalkyl), -NH2, -
- NH(CN), 0, -CN, Ci-e hydroxyalkyl, C1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl.
- the one or more optional substituents of R 1 are each independently selected from halogen, - OH, -CN, C1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl.
- the one or more optional substituents of R 1 are each independently selected from halogen, -OH, and -CN.
- the one or more optional substituents of R 1 are each independently selected from fluorine, - OH, -CN, C1-6 cyanoalkyl, C1-6 alkyl, oxo, and C2-6 alkynyl. In some cases, the one or more optional substituents of R 1 are each independently selected from fluorine, -OH, -CN, C 1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, R 1 is selected from
- the 5- to 12-membered heterocycle of R 1 is unsaturated and a bridged heterocycle.
- R 1 is selected from an optionally substituted 7- to 8-membered unsaturated and bridged heterocycle. In some cases, R 1
- R 1 is selected from an optionally substituted 10-membered heterocycle.
- the 10-membered heterocycle is a bicyclic heterocycle.
- the 10-membered heterocycle is a spiro heterocycle.
- the 10-membered heterocycle is a fused heterocycle.
- the 10- membered heterocycle is a saturated heterocycle.
- the 10-membered heterocycle is a non-aromatic heterocycle.
- the 10-membered heterocycle contains at least 1 nitrogen atom.
- the 10-membered heterocycle contains at least 2 nitrogen atoms.
- R 1 is selected from some cases, R 1 is selected from
- each R 20 is independently selected from hydrogen; and C1-6 alkyl, and C3-12 carbocycle, and each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, C1-10 alkyl, -Ci-io haloalkyl, -O-Ci-10 alkyl, C2- 10 alkenyl, C2-10 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle.
- R 1 is selected from some cases,
- R 1 is selected from an optionally substituted 8- to 10-membered fused heterocycle.
- the 8- to 10- membered fused heterocycle is a bicyclic heterocycle.
- the 8- to 10-membered fused heterocycle is a saturated heterocycle.
- the 8- to 10-membered fused heterocycle is an unsaturated heterocycle.
- the 8- to 10-membered heterocycle is a non-aromatic heterocycle.
- R 1 is selected from an optionally substituted 10- membered fused heterocycle.
- the 10-membered fused heterocycle is a bicyclic heterocycle.
- the 10-membered fused heterocycle is a saturated heterocycle. In some cases, the 10-membered heterocycle is a non-aromatic heterocycle. In some cases, the fused heterocycle has one saturated ring and one aromatic ring. In some cases, the fused heterocycle has one saturated ring and one unsaturated ring. In some cases, the fused heterocycle has two saturated rings. In some cases, the 10-membered heterocycle contains at least 1 nitrogen atom. In some cases, the 10-membered heterocycle contains at least 2 nitrogen atoms. In some cases, the 10-membered heterocycle contains at least 3 nitrogen atoms. In some cases, R 1 is selected from , each of which is optionally substituted with one or more substituents.
- R 1 is selected from hich is optionally substituted with one or more substituents. In some , , which is optionally substituted with one or more substituents. In some cases, the one or more optional substituents of R 1 are independently selected from halogen, -OH,
- R 1 is selected from , each of which is further optionally substituted.
- the further one or more optional substituents are selected from halogen and C1-6 alkyl.
- the further one or more optional substituents are selected from halogen.
- each R 20 is independently selected from hydrogen; and Cn 6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle. In some cases, each R 20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12-membered heterocycle. In some cases, each R 20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12- membered saturated heterocycle. In some cases, each R 20 is independently selected from 5- to 6- membered saturated heterocycle. In some cases, the heterocycle of R 20 has at least one nitrogen atom. In some cases, the heterocycle of R 20 has at least one sulfur atom. In some cases, the heterocycle of R 20 has at least one oxygen atom. In some cases, the heterocycle of R 20 contains only 1 heteroatom.
- the heterocycle of R 20 has at least two heteroatoms. In some cases, the heterocycle of R 20 contains only 2 heteroatoms. In some cases, the optional one or more substituents of R 1 are independently selected from halogen, . In some cases, the optional one or more substituents of R 1 are independently
- R 1 independently selected from halogen, and Ci-6 alkyl-N(R 20 )2.
- the optional one or more substituents of R 1 are independently selected from halogen, , , and .
- R 1 is selected from .
- each R 20 is independently selected from hydrogen, Ci-6 alkyl, and C3-6 carbocycle. In some cases, R 1 is
- R 20 is selected from a 5- to 12-membered heterocycle.
- heterocycle has at least two heteroatoms. In some cases, the heterocycle has at least three heteroatoms. In some cases, the heterocycle has at least four heteroatoms. In some cases, the heterocycle of the one or more optional substituents of R 1 is selected from , each of which is optionally substituted with one or more R 1 *. In some cases, the heterocycle of the one or more optional substituents of R 1 is selected from which is optionally substituted with one or more R 1 *.
- each R 1 * is independently selected from halogen, Ci-6 alkyl-N(R 20 ) 2 , Ci-6 aminoalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, and Ci-6 alkyl. In some cases, each R 1 * is independently selected from halogen, Ci-6 haloalkyl, and Ci-6 alkyl. In some cases, each R 1 * is independently selected from halogen, and Ci-6 alkyl. In some cases, each R 1 * is independently selected from halogen. In some cases, each R 1 * is independently selected from Ci-6 alkyl. In some cases, each R 1 * is independently selected from -OR 20 . In some cases, each R 1 * is independently selected from -OH. In some cases, each R 1 * is independently selected from -OMe. In some cases,
- each R 1 * is independently selected from halogen, C1-6 alkyl-N(R 20 )2, C1-6 aminoalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, and C1-6 alkyl. In some cases, each R 1 * is independently selected from halogen, C1-6 haloalkyl, and C1-6 alkyl. In some cases, each R 1 * is independently selected from halogen, and C1-6 alkyl. In some cases, each R 1 * is independently selected from halogen. In some cases, each R 1 * is independently selected from C1-6 alkyl.
- R 1 is selected from 5- to 15-membered heterocycle (preferably 8- to 10-membered heterocycle or preferably 10- membered heterocycle), each of which are optionally substituted with one or more substituents independently selected from halogen, oxo, -C(O)N(R 20 )2, -C(O)NR 20 OR 20 , -N(R 20 )2, -C(O)R 20 , - C(O)OR 20 , -SO2R 20 , -NHCN, C 1-6 cyanoalkyl, Ci- 6 alkyl, Ci- 6 alkyl-N(R 20 ) 2 , C 2 -6 alkynyl, and 5- to 12-membered heterocycle (preferably 5- to 9-membered heterocycle), wherein the 5- to 12- membered heterocycle are each optionally substituted independently with one or more R 1 *; each R 1 * is independently selected from halogen, C1-6
- the 8- to 10-membered heterocycle is bicyclic. In some cases, the 10-membered heterocycle is substituted. In some cases, R 1 is selected , , , each of which is optionally substituted. In some cases, R 1 is selected -1- , which is optionally substituted. In
- R 1 is selected .
- R 1 is selected from 5- to 15-membered heterocycle (preferably 8- to 10-membered heterocycle or preferably 10- membered heterocycle or preferably 8-membered heterocycle), each of which are optionally substituted with one or more substituents independently selected from halogen, -C(O)N(R 20 )2, - C(O)NR 20 OR 20 , -N(R 20 ) 2 , -C(O)R 20 , -C(O)OR 20 , -NHCN, C I-6 cyanoalkyl, Ci- 6 alkyl, C 2-6 alkynyl, and 5- to 12-membered heterocycle (preferably 5- to 6-membered heterocycle), wherein the 5- to 12-membered heterocycle are each optionally substituted independently with one or more R 1 *; each R 1 * is independently selected from halogen, Ci-6 haloalkyl, and Ci-6 alkyl.
- the 8- to 10-membered heterocycle is bicyclic. In some cases, the 10-membered heterocycle is substituted. In some cases, R is selected -1- , ⁇ L , and -1- , each of which is optionally substituted. In some cases, R is selected -1- , which is optionally substituted. In
- R 1 is selected from
- R 1 is selected from an optionally substituted 7- to 10-membered spiro heterocycle and optionally substituted 7- to 10- membered fused heterocycle.
- the heterocycle of R 1 has at least one nitrogen atom
- the at least one nitrogen at of the heterocycle of R 1 is bound to Formula (I).
- R 1 is selected from an optionally substituted 10-membered spiro heterocycle and optionally substituted 10-membered fused heterocycle.
- R 1 is selected from
- R 1 is selected from hydrogen
- R 1 is selected from an optionally substituted 8- to 10-membered heterocycle.
- the heterocycle is bicyclic.
- the heterocycle has at least one nitrogen atom.
- the heterocycle has which is optionally substituted.
- the optional one or more substituents of R 1 are independently selected from halogen,
- R 1 * is selected from halogen, and Ci-6 alkyl.
- the optional one or more substituents of R 1 are independently selected from chlorine,
- R 1 is selected from an optionally substituted bridged 8- to 9-membered heterocycle.
- the heterocycle of R 1 is selected from more substituents of R 1 are selected from halogen, Ci-6 alkyl, -N(R 20 )2, and Ci-6 aminoalkyl.
- R 1 is selected from an optionally substituted bridged 8-membered heterocycle, wherein the heterocycle contains heteroatoms selected from nitrogen.
- the one or more substituents of R 1 are selected from C i-6 alkyl, -N(R 20 )2, and Ci-6 aminoalkyl.
- the heterocycle of R 1 is selected from each of which is optionally substituted. In some cases, , ,
- R 1 is hydrogen
- R 1 is an optionally substituted 12- to 15-membered heterocycle.
- R 1 is an optionally substituted 12- membered heterocycle.
- R 1 is an optionally substituted 13-membered heterocycle.
- R 1 is an optionally substituted 14-membered heterocycle.
- R 1 is an optionally substituted 15-membered heterocycle.
- the heterocycle of R 1 is tricyclic.
- the heterocycle of R 1 contains a fused heterocycle.
- the heterocycle of R 1 contains a spiro-heterocycle.
- the heterocycle of R 1 contains a fused and spiro-heterocycle. In some cases, the heterocycle of R 1 is an unsaturated heterocycle. In some cases, the heterocycle of R 1 is a non-aromatic heterocycle. In some cases, the heterocycle of R 1 has at least one double bond. In some cases, the heterocycle of R 1 has at least two double bonds. In some cases, the heterocycle of R 1 has at least 2 heteroatoms. In some cases, the heterocycle of R 1 has at least 3 heteroatoms. In some cases, the heterocycle of R 1 has at least 4 heteroatoms. In some cases, the heterocycle of R 1 has at least 5 heteroatoms. In some cases, the heterocycle of R 1 has at least 6 heteroatoms.
- the heterocycle of R 1 has at least 7 heteroatoms. In some cases, the heteroatoms are selected from oxygen, nitrogen, and sulfur. In some cases, the heterocycle of R 1 has at least 3, 4, or 5 nitrogen atoms, and at least 1 sulfur atom. In some cases, the heterocycle of R 1 has at least 3, 4, or 5 nitrogen atoms, and at least 1 oxygen atom. In some cases, the heterocycle of R 1 has at least 3, 4, or 5 nitrogen atoms. In some cases, the heterocycle of R 1 has at least 3, 4, or 5 nitrogen atoms and no other heteroatoms. In some cases, the heteroatoms are selected from nitrogen and sulfur. In some cases, the heteroatoms are selected from nitrogen and oxygen. In some cases, R 1 is selected from
- R 1 is selected from substituted with one or more substituents.
- R 1 is an optionally substituted 12- to 15-membered heterocycle.
- Ring W is an optionally substituted heterocycle and Ring P is an optionally substituted carbocycle or optionally substituted heterocycle, wherein Ring P forms a spirocycle with Ring W.
- Ring W is an optionally substituted fused heterocycle.
- Ring P and Ring W combine to form a heterocycle having at least 12 atoms and most 15 atoms.
- Ring P and Ring W have in total at least 12 atoms and most 15 atoms.
- Ring W is an optionally substituted 10-membered fused heterocycle.
- R 1 is , wherein Ring P is an optionally substituted carbocycle or optionally substituted heterocycle.
- Ring P is an optionally substituted carbocycle.
- Ring P is an optionally substituted heterocycle. In some cases, Ring P forms an optionally substituted C3-C6 carbocycle or optionally substituted 4-to 6- membered heterocycle. In some cases, Ring P forms an optionally substituted C3 carbocycle. In some cases, Ring P forms an optionally substituted C4 carbocycle. In some cases, Ring P forms an optionally substituted C5 carbocycle. In some cases, Ring P forms an optionally substituted 4- membered heterocycle. In some cases, Ring P forms an optionally substituted 5-membered heterocycle. In some cases, Ring P forms an optionally substituted 5-membered heterocycle. In some cases, Ring P has at least 1, 2, or 3 heteroatoms.
- the heteroatoms are selected from oxygen, nitrogen, and sulfur.
- Ring P has 1 sulfur atom.
- Ring P has 1 nitrogen atom.
- Ring P has 1 oxygen atom.
- the one or more optional substituents of Ring W are independently selected from -C(O)R 20 .
- Ring P is substituted.
- Ring W is substituted.
- R 1 is selected from a 5- to 12-membered bridged heterocycle, which is optionally substituted with one or more substituents. In some cases, R 1 is selected from an 8-membered bridged heterocycle, which is optionally substituted with one or more substituents. In some cases, the bridged heterocycle has at least 1 heteroatom. In some cases, the bridged heterocycle has at least 2 heteroatoms. In some cases, the bridged heterocycle has at least 1 nitrogen atom. In some cases, the bridged heterocycle has at least 2 nitrogen atoms. In some cases, the bridged heterocycle has 2 nitrogen atoms. In some cases, R 1 is selected from
- R 1 is selected from — I— , which is optionally substituted with one or more substituents.
- R 1 is selected from an unsaturated 5- to 12-membered heterocycle, which is optionally substituted with one or more substituents.
- the unsaturated 5- to 12-membered heterocycle is selected from each of which is optionally substituted with one or more substituents.
- R 1 for a compound or salt of Formula (I), wherein the 5- to 12- membered heterocycle of R 1 is unsaturated and a bridged heterocycle.
- R 1 is selected from an optionally substituted 7- to 8-membered unsaturated and bridged heterocycle.
- R 1 is selected from -J— .
- R 1 is selected from an optionally substituted 10-membered heterocycle.
- the 10-membered heterocycle is a bicyclic heterocycle.
- the 10-membered heterocycle is a spiro heterocycle.
- the 10-membered heterocycle is a fused heterocycle.
- the 10- membered heterocycle is a saturated heterocycle.
- the 10-membered heterocycle is a non-aromatic heterocycle.
- the 10-membered heterocycle contains at least 1 nitrogen atom.
- the 10-membered heterocycle contains at least 2 nitrogen atoms.
- the 10-membered heterocycle contains at least 3 nitrogen atoms. In some cases, the 10-membered heterocycle contains at least 1 sulfur atom.
- R 1 is selected from an optionally substituted 8- to 10-membered fused heterocycle.
- the 8- to 10- membered fused heterocycle is a bicyclic heterocycle.
- the 8- to 10-membered fused heterocycle is a saturated heterocycle.
- the 8- to 10-membered heterocycle is a non-aromatic heterocycle.
- R 1 is selected from an optionally substituted 10- membered fused heterocycle.
- the 10-membered fused heterocycle is a bicyclic heterocycle.
- the 10-membered fused heterocycle is a saturated heterocycle.
- the 10-membered heterocycle is a non-aromatic heterocycle. In some cases, the fused heterocycle has one saturated ring and one aromatic ring. In some cases, the fused heterocycle has one saturated ring and one unsaturated ring. In some cases, the fused heterocycle has two saturated rings. In some cases, the 10-membered heterocycle contains at least 1 nitrogen atom. In some cases, the 10-membered heterocycle contains at least 2 nitrogen atoms. In some cases, the 10-membered heterocycle contains at least 3 nitrogen atoms. In some cases, R 1 is , each of which is optionally substituted with one or more substituents. In some cases, , which is optionally substituted with one or more substituents.
- the optional one or more substituents are independently selected from -C(O)R 20 , -C(O)N(R 20 )2, and -C(O)NR 20 OR 20 . In some cases, the optional one or more substituents are independently selected from -C(O)R 20 . In some cases, the optional one or more substituents are independently selected from -C(O)N(R 20 )2. In some cases, the optional one or more substituents are independently selected from -C(O)NR 20 OR 20 . In some cases, each R 20 is independently selected from hydrogen; and Ci-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle.
- each R 20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12-membered heterocycle. In some cases, each R 20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12-membered saturated heterocycle. In some cases, the optional one or more substituents of R 1 are independently selected from
- R 1 is selected from an optionally substituted saturated 6- to 7-membered heterocycle. In some cases, R 1 is selected from an optionally substituted saturated 6-membered heterocycle. In some cases, R 1 is selected from —I— , which is optionally substituted. In some cases, the optional one or more substituents are independently selected from halogen, -CN, -NHCN, Ci-6 cyanoalkyl, and Ci-6 alkyl. In some cases, the optional one or more substituents are independently selected from -CN, -NHCN, Ci-6 cyanoalkyl, and Ci-6 alkyl.
- the optional one or more substituents are independently selected from -CN, -NHCN, Ci-6 cyanoalkyl, and Ci-6 alkyl. In some cases, the optional one or more substituents are independently selected from -NHCN, and Ci-6 alkyl. In some cases, R 1 is selected from — i— , which is substituted with one or more substituents selected from -NHCN, and Ci-6 alkyl. In some cases, R 1 is selected from , and
- R 3 is -CN, and R 1 is substituted 8- to 9-membered fused heterocycle, wherein the 8- to 9-membered fused heterocycle has at least one sulfur atom.
- R 3 is -CN, and R 1 is selected from .
- R 3 is -CN, and R 1 is selected from .
- R 3 is -CN, O and R 1 is selected from .
- R 3 is -CN
- R 1 is selected from some cases, selected from optionally substituted 8- to 9-membered fused heterocycle, wherein the 8- to 9-membered fused heterocycle has at least one sulfur atom
- Y-R 2 is selected from -O-L-heterocycle, wherein the heterocycle portion of -O-L-heterocycle is optionally substituted with one or more R 6 .
- B is an optionally substituted 8- to 10-membered fused carbocycle. In some cases, B is a substituted 8- to 10- membered fused carbocycle. In some cases, B is an optionally substituted 9-membered fused carbocycle. In some cases, B is a substituted 9-membered fused carbocycle. In some cases, B is , which is optionally substituted with one or more substituents. In some cases, B is , which is substituted with one or more substituents. In some cases, for B, the one or more substituents are independently selected from halogen, oxo, -NH2, C1-C3 alkyl, -B(OH)2, -
- B is substituted with at least one halogen. In some cases, B is substituted with at least one chlorine. In some cases, B is substituted with at least one fluorine. In some cases, B is selected selected from halogen and Ci-e haloalkyl. In some cases, which is substituted with one or more substituents selected from halogen. In some cases, B is selected from which is substituted with one or more substituents selected from fluorine. In some cases, B is substituted with one or more substituents selected from chlorine. In some cases, B is selected
- R 1 is selected from an optionally substituted unsaturated 10-membered fused heterocycle.
- R 1 is , which is optionally substituted.
- B is an optionally substituted 8- to 10-membered fused heterocycle, wherein the heterocycle each of which is optionally substituted.
- Y-R 2 is selected from , wherein the heterocycle portion is optionally substituted.
- n is 0.
- R 3 is selected from hydrogen, halogen, -CN, -N(R 20 ) 2 , -OH, -S(O) 2 (R 20 ), -C(O)R 20 , Ci- 6 alkyl-N(R 20 ) 2 , Ci- 6 aminoalkyl, Ci- 6 alkoxy, Ci-6 alkoxyalkyl, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, C 1-6 haloalkyl, and Ci-6 alkyl.
- R 3 is selected from hydrogen, halogen, and -CN.
- R 1 is selected from an optionally substituted 11 -membered fused heterocycle. In some cases, R 1 is selected from an optionally substituted unsaturated 11 -membered fused heterocycle. In some cases, the heterocycle contains at least one sulfur atom. In some cases, the heterocycle contains at least one nitrogen atom. In some cases, the heterocycle contains 3 heteroatoms. In some cases, the heterocycle is — L ⁇ , which is optionally substituted. In some cases, R 1 is — L ⁇
- the optional substituents for R 1 are each independently selected from halogen, -OH, -N(R 20 ) 2 , -NO 2 , C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and C1-6 haloalkyl.
- the optional substituents for R 1 are each independently selected from halogen, -CN, -OH, -NH 2 , C 1-6 aminoalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, and C1-6 haloalkyl. . In some cases, the optional substituents for R 1 are each independently selected from -OH, and -NH 2 . In some cases, R 20 is selected from hydrogen and C1-3 alkyl.
- n is selected from 0 to 4. In some cases, n is selected from 0 to 3. In some cases, n is selected from 0 to 2. In some cases, n is selected from 0 and 1. In some cases, n is 0. In some cases, n is 1. In some cases, n is
- each R 20 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(Ci-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci- 10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle.
- each R 20 is independently selected from hydrogen and C1-6 alkyl.
- each R 20 is independently selected from C1-6 alkyl.
- each R 21 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(Ci-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci- 10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle.
- each R 21 is independently selected from hydrogen and C1-6 alkyl.
- each R 21 is independently selected from C1-6 alkyl.
- each R 20 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(Ci-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci- 10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle.
- each R 20 is independently selected from hydrogen and C1-6 alkyl.
- each R 20 is independently selected from C1-6 alkyl.
- each R 23 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O- C1-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle.
- each R 23 is independently selected from hydrogen and C1-6 alkyl.
- each R 23 is independently selected from C1-6 alkyl.
- the present disclosure provides a compound of Formula (I- A) Formula (LA), or a pharmaceutically acceptable salt thereof wherein:
- Y is selected from a bond, -0-, -S-, and -N(R 5 )-;
- Y is -O-
- R 2 is selected from -L-heterocycle, -L-N(R 23 )2, wherein the heterocycle portion of -L- heterocycle is optionally substituted with one or more R 6 .
- R 3 is selected from hydrogen, halogen, -CN, -N(R 20 )2, -OR 20 , -C(O)R 20 , C1-6 alkyl-N(R 20 )2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, and C1-6 alkyl; and each R 20 is independently selected from hydrogen; and C1-6 alkyl, and C3-12 carbocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NH2, -N(Ci-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, and oxo.
- a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), Y is -O-.
- a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), R 2 is selected from optionally substituted -L-heterocycle, and -L-N(R 23 )2.
- R 2 is selected from optionally substituted -L- 5-to 8-membered heterocycle, and -L-N(R 23 )2.
- R 2 is selected from optionally substituted -L-heterocycle.
- R 2 is selected from -L-N(R 23 )2.
- the heterocycle contains at least one nitrogen atom.
- the heterocycle contains at least one sulfur atom.
- the heterocycle contains at least one oxygen atom.
- R 6 when R 2 is a heteroaryl, R 6 is selected from halogen and C1-C3 haloalkyl, C1-C3 alkyl.
- R 2 when R 2 is a heteroaryl, R 6 is selected from a halogen.
- R 23 is selected from hydrogen and C1-C3 alkyl.
- each R 23 is selected from C1-C3 alkyl.
- each R 23 is selected from methyl.
- each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen, and
- each L is selected from ' » [00256]
- a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), B is selected from an 8- to 10-membered heterocycle, wherein the 8- to 10-membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -CN, -NH2, and C1-6 alkyl.
- the heterocycle of B is selected from substituted with one or more substituents independently selected from halogen, -CN, -NH2, and
- a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), R 3 is -CN.
- R 3 is hydrogen.
- R 3 is halogen.
- R 3 is fluorine.
- a compound or salt of Formula (I), Formula (I-A), or Formula which is optionally substituted with one or more substituents.
- the one or more optional substituents of R 1 are not electrophiles.
- R 1 is selected from a compound in the Examples.
- B is selected from a compound in the Examples.
- Y is selected from a compound in the Examples.
- R 2 is selected from a compound in the Examples.
- R 3 is selected from a compound in the Examples.
- each R 20 is selected from hydrogen and C1-3 alkyl. In some cases, each R 20 is selected from hydrogen and Ci alkyl.
- the compounds of Formula (I), (I-A), or (I-B) used in the methods include trifluoroacetic acid salts of the above compounds.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 5 mg to about 500 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 150 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 125 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 100 mg.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 25 mg to about 100 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 50 mg to about 100 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) of is administered to a subject at about 5 mg to about 75 mg.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, or about 150 mg.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 15 mg, about 30 mg, about 45 mg, or about 60 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 15 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 30 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 45 mg.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 60 mg.
- the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
- a compound or salt of Formula (I), (I- A), or (I-B) is administered once daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I- B) is administered twice daily. In some embodiments, the compound or salt of Formula (I), (I- A), or (I-B) is administered 3 times daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered once weekly. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered every other day. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered every 3 days.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 10 mg to 150 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) or a salt thereof is administered to a subject at 10 mg to 125 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 10 mg to 100 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 25 mg to 100 mg.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 50 mg to 100 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 5 mg to 75 mg.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 10 mg, 15 mg, 20 mg, 25 mg, 30 mg, 35 mg, 40 mg, 45 mg, 50 mg, 55 mg, 60 mg, 65 mg, 70 mg, 75 mg, 80 mg, 85 mg, 90 mg, 100 mg, 105 mg, 110 mg, 115 mg, 120 mg, 125 mg, 130 mg, 135 mg, 140 mg, 145 mg, or 150 mg.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 15 mg, 30 mg, 45 mg, or 60 mg.
- the compound or salt of Formula (I) is administered to a subject at 15 mg.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 30 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 45 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 60 mg. In some embodiments, the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 150 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 125 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 100 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 25 mg to about 100 mg, daily.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 50 mg to about 100 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 5 mg to about 75 mg, daily.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, or about 150 mg, daily.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 15 mg, about 30 mg, about 45 mg, or about 60 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 15 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 30 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 45 mg, daily.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 60 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered once daily. In some embodiments, the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 150 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 125 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 100 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 25 mg to about 100 mg, twice daily.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 50 mg to about 100 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 5 mg to about 75 mg, twice daily.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, or about 150 mg, twice daily.
- the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
- the compound or salt of Formula (I), (I- A), or (I-B) is administered to a subject at about 15 mg, about 30 mg, about 45 mg, or about 60 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I- A), or (I-B) is administered to a subject at about 15 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 30 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 45 mg, twice daily.
- the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 60 mg, twice daily.
- the subject is between 12 years old to 18 years old.
- the subject is between greater than or equal 12 years old to less than or equal to 18 years.
- the subject is an adult.
- the subject is greater than or equal to 18 years old.
- a compound of Formula (I), (I-A), or (I-B) is administered as a capsule during the period of time.
- a tablet or capsule formulation of a compound of Formula (I), (I-A), or (I-B) comprises about 10 mg to about 100 mg (e.g., about 10 mg to about 95 mg, about 10 mg to about 90 mg, about 10 mg to about 85 mg, about 10 mg to about 80 mg, about 10 mg to about 75 mg, about 10 mg to about 70 mg, about 10 mg to about 65 mg, about 10 mg to about 60 mg, about 10 mg to about 55 mg, about 10 mg to about 50 mg, about 10 mg to about 45 mg, about 10 mg to about 40 mg, about 10 mg to about 35 mg, about 10 mg to about 30 mg, about 10 mg to about 25 mg, about 10 mg to about 20 mg, about 10 mg to about 15 mg, about 15 mg to about 100 mg, about 15 mg to about 95 mg, about 15 mg to about 90 mg, about 15 mg to about 85 mg, about 15
- a compound of Formula (I), (I- A), or (I-B) is orally administered once a day (QD) on a daily basis during a period of time. In one embodiment, a compound of Formula (I), (I- A), or (I-B) is orally administered twice a day (BID) on a daily basis during a period of time.
- a compound of Formula (I), (I- A), or (I-B) is orally administered in the amount of about 20 mg to about 500 mg (e.g., about 20 mg to about 480 mg, about 20 mg to about 460 mg, about 20 mg to about 440 mg, about 20 mg to about 420 mg, about 20 mg to about 400 mg, about 20 mg to about 380 mg, about 20 mg to about 360 mg, about 20 mg to about 340 mg, about 20 mg to about 320 mg, about 20 mg to about 300 mg, about 20 mg to about 280 mg, about 20 mg to about 260 mg, about 20 mg to about 240 mg, about 20 mg to about 220 mg, about 20 mg to about 200 mg, about 20 mg to about 180 mg, about 20 mg to about 160 mg, about 20 mg to about 140 mg, about 20 mg to about 120 mg, about 20 mg to about 100 mg, about 20 mg to about 80 mg, about 20 mg to about 60 mg, about 20 mg to about 40 mg, about 40 mg to about 500 mg, about 40 mg to about 480 mg
- the combination therapy comprises oral administration of a compound of Formula (I), (I- A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg (e.g., about 10 mg to about 380 mg, about 10 mg to about 360 mg, about 10 mg to about 340 mg, about 10 mg to about 320 mg, about 10 mg to about 300 mg, about 10 mg to about 280 mg, about 10 mg to about 260 mg, about 10 mg to about 240 mg, about 10 mg to about 220 mg, about 10 mg to about 200 mg, about 10 mg to about 180 mg, about 10 mg to about 160 mg, about 10 mg to about 140 mg, about 10 mg to about 120 mg, about 10 mg to about 100 mg, about 10 mg to about 80 mg, about 10 mg to about 60 mg, about 10 mg to about 40 mg, about 10 mg to about 20 mg, about 20 mg to about 400 mg, about 20 mg to about 380 mg,
- CTLA-4 inhibitor or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time).
- the inhibitor is an immunomodulator inhibitor.
- the inhibitor is an PD-1 inhibitor.
- the inhibitor is an PD-L1 inhibitor.
- the inhibitor is an CTLA-4 inhibitor.
- the compound of Formula (I), (I-A), or (I-B) is selected from compounds 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of any one thereof.
- the compound of Formula (I), (I-A), or (I-B) is selected compound2, or a pharmaceutically acceptable salt of any one thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 4, or a pharmaceutically acceptable salt of any one thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 14.
- the compound of Formula (I), (I-A), or (I-B) is compound 25, or a pharmaceutically acceptable salt of any one thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 74 or a pharmaceutically acceptable salt of any one thereof.
- the compound of Formula (I), (I-A), or (I-B) is orally administered once daily. In one embodiment, the compound of Formula (I), (I-A), or (I-B) is orally administered twice daily.
- a compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or pharmaceutical composition thereof is administered as a tablet or capsule.
- a tablet or capsule formulation of a compound of Formula (I), (I- A), or (I-B) comprises about 10 mg to about 100 mg (e.g., about 10 mg to about 95 mg, about 10 mg to about 90 mg, about 10 mg to about 85 mg, about 10 mg to about 80 mg, about 10 mg to about 75 mg, about 10 mg to about 70 mg, about 10 mg to about 65 mg, about 10 mg to about 60 mg, about 10 mg to about 55 mg, about 10 mg to about 50 mg, about 10 mg to about 45 mg, about 10 mg to about 40 mg, about 10 mg to about 35 mg, about 10 mg to about 30 mg, about 10 mg to about 25 mg, about 10 mg to about 20 mg, about 10 mg to about 15 mg, about 15 mg to about 100 mg, about 15 mg to about 95 mg, about 15 mg to about 90 mg, about 15
- a compound is selected from compounds 2, 3, 4, 14, 25, and 74.
- a compound of Formula (I), (I-A), or (I-B) is orally administered once a day (QD) on a daily basis during a period of time.
- a compound of Formula (I), (I-A), or (I-B) is orally administered twice a day (BID) on a daily basis during a period of time.
- a compound of Formula (I), (I-A), or (I-B) is orally administered in the amount of about 20 mg to about 500 mg (e.g., about 20 mg to about 480 mg, about 20 mg to about 460 mg, about 20 mg to about 440 mg, about 20 mg to about 420 mg, about 20 mg to about 400 mg, about 20 mg to about 380 mg, about 20 mg to about 360 mg, about 20 mg to about 340 mg, about 20 mg to about 320 mg, about 20 mg to about 300 mg, about 20 mg to about 280 mg, about 20 mg to about 260 mg, about 20 mg to about 240 mg, about 20 mg to about 220 mg, about 20 mg to about 200 mg, about 20 mg to about 180 mg, about 20 mg to about 160 mg, about 20 mg to about 140 mg, about 20 mg to about 120 mg, about 20 mg to about 100 mg, about 20 mg to about 80 mg, about 20 mg to about 60 mg, about 20 mg to about 40 mg, about 40 mg to about 500 mg, about 40 mg to about 480 mg
- an inhibitor selected from:
- CTLA-4 inhibitor wherein the inhibitor is administered, for example, once a week, once every two weeks, once every three weeks, or once every four weeks, depending on the dosage.
- the compound of Formula (I), (I- A), or (I-B) is selected compound 2, or a pharmaceutically acceptable salt of any one thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 3, or a pharmaceutically acceptable salt of any one thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 4, or a pharmaceutically acceptable salt of any one thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 25, or a pharmaceutically acceptable salt of any one thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 74 or a pharmaceutically acceptable salt of any one thereof. Included in the present disclosure are salts, particularly pharmaceutically acceptable salts, of the compounds described herein.
- the compounds of the present invention that possess a sufficiently acidic, a sufficiently basic, or both functional groups can react with any of a number of inorganic bases, and inorganic and organic acids, to form a salt.
- compounds that are inherently charged, such as those with a quaternary nitrogen can form a salt with an appropriate counterion, e.g., a halide such as bromide, chloride, or fluoride, particularly bromide.
- Chemical entities having carbon-carbon double bonds or carbon-nitrogen double bonds may exist in Z- or E- form (or cis- or trans- form). Furthermore, some chemical entities may exist in various tautomeric forms. Unless otherwise specified, compounds described herein are intended to include all Z-, E- and tautomeric forms as well.
- a “tautomer” refers to a molecule wherein a proton shift from one atom of a molecule to another atom of the same molecule is possible.
- the compounds disclosed herein are used in different enriched isotopic forms, e.g., enriched in the content of 2 H, 3 H, n C, 13 C and/or 14 C.
- the compound is deuterated in at least one position.
- deuterated forms can be made by the procedure described in U.S. Patent Nos. 5,846,514 and 6,334,997. As described in U.S. Patent Nos. 5,846,514 and 6,334,997, deuteration can improve the metabolic stability and or efficacy, thus increasing the duration of action of drugs.
- compounds described herein are intended to include compounds which differ only in the presence of one or more isotopically enriched atoms.
- compounds having the present structures except for the replacement of a hydrogen by a deuterium or tritium, or the replacement of a carbon by 13 C- or 14 C-enriched carbon are within the scope of the present disclosure.
- the compounds of the present disclosure optionally contain unnatural proportions of atomic isotopes at one or more atoms that constitute such compounds.
- the compounds may be labeled with isotopes, such as for example, deuterium ( 2 H), tritium ( 3 H), iodine-125 ( 125 I) or carbon-14 ( 14 C).
- the compounds disclosed herein have some or all of the J H atoms replaced with 2 H atoms.
- the methods of synthesis for deuterium-containing compounds are known in the art and include, by way of non-limiting example only, the following synthetic methods.
- Deuterium substituted compounds are synthesized using various methods such as described in: Dean, Dennis C.; Editor. Recent Advances in the Synthesis and Applications of Radiolabeled Compounds for Drug Discovery and Development. [In: Curr., Pharm. Des., 2000; 6(10)] 2000, 110 pp; George W.; Varma, Rajender S. The Synthesis of Radiolabeled Compounds via Organometallic Intermediates, Tetrahedron, 1989, 45(21), 6601-21; and Evans, E. Anthony. Synthesis of radiolabeled compounds, J. Radioanal. Chem., 1981, 64(1-2), 9-32.
- Deuterated starting materials are readily available and are subjected to the synthetic methods described herein to provide for the synthesis of deuterium-containing compounds.
- Large numbers of deuterium-containing reagents and building blocks are available commercially from chemical vendors, such as Aldrich Chemical Co.
- Compounds of the present invention also include crystalline and amorphous forms of those compounds, pharmaceutically acceptable salts, and active metabolites of these compounds having the same type of activity, including, for example, polymorphs, pseudopolymorphs, solvates, hydrates, unsolvated polymorphs (including anhydrates), conformational polymorphs, and amorphous forms of the compounds, as well as mixtures thereof.
- the compounds described herein may in some cases exist as diastereomers, enantiomers, or other stereoisomeric forms. Where absolute stereochemistry is not specified, the compounds presented herein include all diastereomeric, enantiomeric, and epimeric forms as well as the appropriate mixtures thereof. Separation of stereoisomers may be performed by chromatography or by forming diastereomers and separating by recrystallization, or chromatography, or any combination thereof. (Jean Jacques, Andre Collet, Samuel H. Wilen, “Enantiomers, Racemates and Resolutions”, John Wiley And Sons, Inc., 1981, herein incorporated by reference for this disclosure). Stereoisomers may also be obtained by stereoselective synthesis.
- the methods and compositions described herein include the use of amorphous forms as well as crystalline forms (also known as polymorphs).
- the compounds described herein may be in the form of pharmaceutically acceptable salts.
- active metabolites of these compounds having the same type of activity are included in the scope of the present disclosure.
- the compounds described herein can exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like.
- the solvated forms of the compounds presented herein are also considered to be disclosed herein.
- compounds or salts of the compounds may be prodrugs, e.g., wherein a hydroxyl in the parent compound is presented as an ester or a carbonate, or carboxylic acid present in the parent compound is presented as an ester.
- prodrug is intended to encompass compounds which, under physiologic conditions, are converted into pharmaceutical agents of the present disclosure.
- One method for making a prodrug is to include one or more selected moieties which are hydrolyzed under physiologic conditions to reveal the desired molecule.
- the prodrug is converted by an enzymatic activity of the host animal such as specific target cells in the host animal.
- esters or carbonates e.g., esters or carbonates of alcohols or carboxylic acids and esters of phosphonic acids
- Prodrug forms of the herein described compounds, wherein the prodrug is metabolized in vivo to produce a compound as set forth herein are included within the scope of the claims. In some cases, some of the herein-described compounds may be a prodrug for another derivative or active compound.
- Prodrugs are often useful because, in some situations, they may be easier to administer than the parent drug. They may, for instance, be bioavailable by oral administration whereas the parent is not. Prodrugs may help enhance the cell permeability of a compound relative to the parent drug. The prodrug may also have improved solubility in pharmaceutical compositions over the parent drug. Prodrugs may be designed as reversible drug derivatives, for use as modifiers to enhance drug transport to site-specific tissues or to increase drug residence inside of a cell.
- the design of a prodrug increases the lipophilicity of the pharmaceutical agent. In some embodiments, the design of a prodrug increases the effective water solubility. See, e.g., Fedorak et al., Am. J. Physiol., 269:G210-218 (1995); McLoed et al., Gastroenterol, 106:405-413 (1994); Hochhaus et al., Biomed. Chrom., 6:283-286 (1992); J. Larsen and H. Bundgaard, Int. J. Pharmaceutics, 37, 87 (1987); J. Larsen et al., Int. J.
- the present disclosure provides methods of producing the above-defined compounds.
- the compounds may be synthesized using conventional techniques.
- these compounds are conveniently synthesized from readily available starting materials.
- compositions comprising a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), an immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor, or a pharmaceutically acceptable salt of any one thereof (also referred to herein as “a pharmaceutical agent”).
- compositions may be formulated using one or more physiologically acceptable carriers including excipients and auxiliaries which facilitate processing of the pharmaceutical agent into preparations which are used pharmaceutically. Proper formulation is dependent upon the route of administration chosen.
- a summary of pharmaceutical compositions is found, for example, in Remington: The Science and Practice of Pharmacy, Nineteenth Ed (Easton, Pa., Mack Publishing Company, 1995); Hoover, John E., Remington’s Pharmaceutical Sciences, Mack Publishing Co., Easton, Pennsylvania 1975; Liberman, H.A. and Lachman, L., Eds., Pharmaceutical Dosage Forms, Marcel Decker, New York, N.Y., 1980; and Pharmaceutical Dosage Forms and Drug Delivery Systems, Seventh Ed. (Lippincott Williams & Wilkins, 1999).
- compositions and methods of the present disclosure may be utilized to treat an individual in need thereof.
- the individual is a mammal such as a human, or a non-human mammal.
- the composition or the pharmaceutical agent is preferably administered as a pharmaceutical composition comprising, for example, a pharmaceutical agent and a pharmaceutically acceptable carrier or excipient.
- Pharmaceutically acceptable carriers are well known in the art and include, for example, aqueous solutions such as water or physiologically buffered saline or other solvents or vehicles such as glycols, glycerol, oils such as olive oil, or injectable organic esters.
- the aqueous solution is pyrogen-free, or substantially pyrogen-free.
- the excipients can be chosen, for example, to effect delayed release of an agent or to selectively target one or more cells, tissues or organs.
- the pharmaceutical composition can be in dosage unit form such as tablet, capsule, granule, lyophile for reconstitution, powder, solution, syrup, suppository, injection or the like.
- the composition can also be present in a transdermal delivery system, e.g., a skin patch.
- the composition can also be present in a solution suitable for topical administration, such as an eye drop.
- a pharmaceutically acceptable excipient can contain physiologically acceptable agents that act, for example, to stabilize, increase solubility or to increase the absorption of a compound such as a pharmaceutical agent.
- physiologically acceptable agents include, for example, carbohydrates, such as glucose, sucrose or dextrans, antioxidants, such as ascorbic acid or glutathione, chelating agents, low molecular weight proteins or other stabilizers or excipients.
- the choice of a pharmaceutically acceptable excipient, including a physiologically acceptable agent depends, for example, on the route of administration of the composition.
- the preparation or pharmaceutical composition can be a self emulsifying drug delivery system or a self microemulsifying drug delivery system.
- the pharmaceutical composition also can be a liposome or other polymer matrix, which can have incorporated therein, for example, a compound of the invention.
- Liposomes for example, which comprise phospholipids or other lipids, are nontoxic, physiologically acceptable and metabolizable carriers that are relatively simple to make and administer.
- a pharmaceutical composition can be administered to a subject by any of a number of routes of administration including, for example, orally, for example, drenches as in aqueous or non-aqueous solutions or suspensions, tablets, capsules, including sprinkle capsules and gelatin capsules, boluses, powders, granules, pastes for application to the tongue; absorption through the oral mucosa, e.g., sublingually; anally, rectally or vaginally, for example, as a pessary, cream or foam; parenterally, including intramuscularly, intravenously, subcutaneously or intrathecally as, for example, a sterile solution or suspension; nasally; intraperitoneally; subcutaneously; transdermally, for example, as a patch applied to the skin; and topically, for example, as a cream, ointment or spray applied to the skin, or as an eye drop.
- routes of administration including, for example, orally, for example, drenches as in aque
- a pharmaceutical composition may be a sterile aqueous or non-aqueous solution, suspension or emulsion, e.g., a microemulsion.
- the excipients described herein are examples and are in no way limiting.
- An effective amount or therapeutically effective amount refers to an amount of the one or more pharmaceutical agents administered to a subject, either as a single dose or as part of a series of doses, which is effective to produce a desired therapeutic effect.
- Subjects may generally be monitored for therapeutic effectiveness using assays and methods suitable for the condition being treated, which assays will be familiar to those having ordinary skill in the art and are described herein.
- Pharmacokinetics of a pharmaceutical agent, or one or more metabolites thereof, that is administered to a subject may be monitored by determining the level of the pharmaceutical agent or metabolite in a biological fluid, for example, in the blood, blood fraction, e.g., serum, and/or in the urine, and/or other biological sample or biological tissue from the subject. Any method practiced in the art and described herein to detect the agent may be used to measure the level of the pharmaceutical agent or metabolite during a treatment course.
- the dose of a pharmaceutical agent described herein for treating a disease or disorder may depend upon the subject’s condition, that is, stage of the disease, severity of symptoms caused by the disease, general health status, as well as age, gender, and weight, and other factors apparent to a person skilled in the medical art.
- Pharmaceutical compositions may be administered in a manner appropriate to the disease to be treated as determined by persons skilled in the medical arts.
- suitable duration and frequency of administration of the pharmaceutical agent may also be determined or adjusted by such factors as the condition of the patient, the type and severity of the patient’s disease, the particular form of the active ingredient, and the method of administration.
- Optimal doses of an agent may generally be determined using experimental models and/or clinical trials.
- the optimal dose may depend upon the body mass, weight, or blood volume of the subject. The use of the minimum dose that is sufficient to provide effective therapy is usually preferred. Design and execution of pre- clinical and clinical studies for a pharmaceutical agent, including when administered for prophylactic benefit, described herein are well within the skill of a person skilled in the relevant art.
- the optimal dose of each pharmaceutical agent may be different, such as less than when either agent is administered alone as a single agent therapy.
- two pharmaceutical agents in combination may act synergistically or additively, and either agent may be used in a lesser amount than if administered alone.
- An amount of a pharmaceutical agent that may be administered per day may be, for example, between about 0.01 mg/kg and 100 mg/kg, e.g., between about 0.1 to 1 mg/kg, between about 1 to 10 mg/kg, between about 10-50 mg/kg, between about 50-100 mg/kg body weight. In other embodiments, the amount of a pharmaceutical agent that may be administered per day is between about 0.01 mg/kg and 1000 mg/kg, between about 100-500 mg/kg, or between about 500-1000 mg/kg body weight.
- the optimal dose, per day or per course of treatment may be different for the disease or disorder to be treated and may also vary with the administrative route and therapeutic regimen.
- compositions comprising a pharmaceutical agent can be formulated in a manner appropriate for the delivery method by using techniques routinely practiced in the art.
- the composition may be in the form of a solid, e.g., tablet, capsule, semi-solid, e.g., gel, liquid, or gas, e.g., aerosol.
- the pharmaceutical composition is administered as a bolus infusion.
- compositions are well known in the pharmaceutical art and described, for example, in Rowe et al., Handbook of Pharmaceutical Excipients: A Comprehensive Guide to Uses, Properties, and Safety, 5 th Ed., 2006, and in Remington: The Science and Practice of Pharmacy (Gennaro, 21 st Ed. Mack Pub. Co., Easton, PA (2005)).
- exemplary pharmaceutically acceptable excipients include sterile saline and phosphate buffered saline at physiological pH. Preservatives, stabilizers, dyes, buffers, and the like may be provided in the pharmaceutical composition. In addition, antioxidants and suspending agents may also be used.
- compositions described herein may be formulated as a lyophilizate.
- a composition described herein may be lyophilized or otherwise formulated as a lyophilized product using one or more appropriate excipient solutions for solubilizing and/or diluting the pharmaceutical agent(s) of the composition upon administration.
- the pharmaceutical agent may be encapsulated within liposomes using technology known and practiced in the art.
- a pharmaceutical agent is not formulated within liposomes for application to a stent that is used for treating highly, though not totally, occluded arteries.
- Pharmaceutical compositions may be formulated for any appropriate manner of administration described herein and in the art.
- a pharmaceutical composition e.g., for oral administration or for injection, infusion, subcutaneous delivery, intramuscular delivery, intraperitoneal delivery or other method, may be in the form of a liquid.
- a liquid pharmaceutical composition may include, for example, one or more of the following: a sterile diluent such as water, saline solution, preferably physiological saline, Ringer’s solution, isotonic sodium chloride, fixed oils that may serve as the solvent or suspending medium, polyethylene glycols, glycerin, propylene glycol or other solvents; antibacterial agents; antioxidants; chelating agents; buffers and agents for the adjustment of tonicity such as sodium chloride or dextrose.
- a sterile diluent such as water, saline solution, preferably physiological saline, Ringer’s solution, isotonic sodium chloride, fixed oils that may serve as the solvent or suspending medium, polyethylene glycols, glycerin, propylene glycol or other solvent
- a parenteral composition can be enclosed in ampoules, disposable syringes or multiple dose vials made of glass or plastic.
- physiological saline is preferred, and an injectable pharmaceutical composition is preferably sterile.
- a liquid pharmaceutical composition may be applied to the eye in the form of eye drops.
- a liquid pharmaceutical composition may be delivered orally.
- At least one of the pharmaceutical agents described herein can be used alone or in combination with appropriate additives to make tablets, powders, granules or capsules, and if desired, with diluents, buffering agents, moistening agents, preservatives, coloring agents, and flavoring agents.
- the pharmaceutical agents may be formulated with a buffering agent to provide for protection of the compound from low pH of the gastric environment and/or an enteric coating.
- a pharmaceutical agent included in a pharmaceutical composition may be formulated for oral delivery with a flavoring agent, e.g., in a liquid, solid or semi-solid formulation and/or with an enteric coating.
- a pharmaceutical composition comprising any one of the pharmaceutical agents described herein may be formulated for sustained or slow release, also called timed release or controlled release.
- Such compositions may generally be prepared using well known technology and administered by, for example, oral, rectal, intradermal, or subcutaneous implantation, or by implantation at the desired target site.
- Sustained-release formulations may contain the compound dispersed in a carrier matrix and/or contained within a reservoir surrounded by a rate controlling membrane. Excipients for use within such formulations are biocompatible, and may also be biodegradable; preferably the formulation provides a relatively constant level of active component release.
- the amount of pharmaceutical agent contained within a sustained release formulation depends upon the site of implantation, the rate and expected duration of release, and the nature of the condition, disease or disorder to be treated or prevented.
- the pharmaceutical compositions comprising a pharmaceutical agent are formulated for transdermal, intradermal, or topical administration.
- the compositions can be administered using a syringe, bandage, transdermal patch, insert, or syringelike applicator, as a powder/talc or other solid, liquid, spray, aerosol, ointment, foam, cream, gel, paste.
- This preferably is in the form of a controlled release formulation or sustained release formulation administered topically or injected directly into the skin adjacent to or within the area to be treated, e.g., intradermally or subcutaneously.
- the active compositions can also be delivered via iontophoresis.
- Preservatives can be used to prevent the growth of fungi and other microorganisms. Suitable preservatives include, but are not limited to, benzoic acid, butylparaben, ethyl paraben, methyl paraben, propylparaben, sodium benzoate, sodium propionate, benzalkonium chloride, benzethonium chloride, benzyl alcohol, cetypyridinium chloride, chlorobutanol, phenol, phenylethyl alcohol, thimerosal, and combinations thereof.
- Pharmaceutical compositions comprising a pharmaceutical agent can be formulated as emulsions for topical application. An emulsion contains one liquid distributed in the body of a second liquid.
- the emulsion may be an oil-in-water emulsion or a water-in-oil emulsion.
- Either or both of the oil phase and the aqueous phase may contain one or more surfactants, emulsifiers, emulsion stabilizers, buffers, and other excipients.
- the oil phase may contain other oily pharmaceutically approved excipients.
- Suitable surfactants include, but are not limited to, anionic surfactants, non-ionic surfactants, cationic surfactants, and amphoteric surfactants.
- Compositions for topical application may also include at least one suitable suspending agent, antioxidant, chelating agent, emollient, or humectant.
- Ointments and creams may, for example, be formulated with an aqueous or oily base with the addition of suitable thickening and/or gelling agents.
- Lotions may be formulated with an aqueous or oily base and will in general also contain one or more emulsifying agents, stabilizing agents, dispersing agents, suspending agents, thickening agents, or coloring agents.
- Liquid sprays may be delivered from pressurized packs, for example, via a specially shaped closure.
- Oil-in-water emulsions can also be used in the compositions, patches, bandages and articles. These systems are semisolid emulsions, micro-emulsions, or foam emulsion systems.
- the pharmaceutical agent described herein can be formulated as in inhalant. Inhaled methods can deliver medication directly to the airway.
- the pharmaceutical agent can be formulated as aerosols, microspheres, liposomes, or nanoparticles.
- the pharmaceutical agent can be formulated with solvents, gases, nitrates, or any combinations thereof.
- Compositions described herein are optionally formulated for delivery as a liquid aerosol or inhalable dry powder.
- Liquid aerosol formulations are optionally nebulized predominantly into particle sizes that can be delivered to the terminal and respiratory bronchioles.
- Liquid aerosol and inhalable dry powder formulations are preferably delivered throughout the endobronchial tree to the terminal bronchioles and eventually to the parenchymal tissue.
- Aerosolized formulations described herein are optionally delivered using an aerosol forming device, such as a jet, vibrating porous plate or ultrasonic nebulizer, preferably selected to allow the formation of aerosol particles having with a mass medium average diameter predominantly between 1 to 5 p. Further, the formulation preferably has balanced osmolarity ionic strength and chloride concentration, and the smallest aerosolizable volume able to deliver effective dose of the pharmaceutical agent. Additionally, the aerosolized formulation preferably does not impair negatively the functionality of the airways and does not cause undesirable side effects.
- an aerosol forming device such as a jet, vibrating porous plate or ultrasonic nebulizer
- Aerosolization devices suitable for administration of aerosol formulations described herein include, for example, jet, vibrating porous plate, ultrasonic nebulizers and energized dry powder inhalers, that are able to nebulize the formulation into aerosol particle size predominantly in the size range from 1-5 p. Predominantly in this application means that at least 70% but preferably more than 90% of all generated aerosol particles are within 1-5 p range.
- a jet nebulizer works by air pressure to break a liquid solution into aerosol droplets. Vibrating porous plate nebulizers work by using a sonic vacuum produced by a rapidly vibrating porous plate to extrude a solvent droplet through a porous plate.
- An ultrasonic nebulizer works by a piezoelectric crystal that shears a liquid into small aerosol droplets.
- suitable devices including, for example, AeroNebTM and AeroDoseTM vibrating porous plate nebulizers (AeroGen, Inc., Sunnyvale, California), Sidestream® nebulizers (Medic-Aid Ltd., West Wales, England), Pari LC® and Pari LC Star® jet nebulizers (Pari Respiratory Equipment, Inc., Richmond, Virginia), and AerosonicTM (DeVilbiss Medizinische Kunststoffische Kunststoffische Kunststoffische Kunststoffische Kunststoffische Kunststoffische Kunststoffische Kunststoffische Kunststoffische Kunststoffische Kunststoffische Kunststoffische Kunststoffische Kunststoffische Kunststoffetechnik (Deutschland) GmbH, Heiden, Germany) and UltraAire® (Omron Healthcare, Inc., Vernon Hills, Illinois) ultrasonic nebulizers.
- AeroNebTM and AeroDoseTM vibrating porous plate nebulizers (AeroGen, Inc.
- the pharmaceutical agent(s) can be formulated with oleaginous bases or ointments to form a semisolid composition with a desired shape.
- these semisolid compositions can contain dissolved and/or suspended bactericidal agents, preservatives and/or a buffer system.
- a petrolatum component that may be included may be any paraffin ranging in viscosity from mineral oil that incorporates isobutylene, colloidal silica, or stearate salts to paraffin waxes.
- Absorption bases can be used with an oleaginous system.
- Additives may include cholesterol, lanolin (lanolin derivatives, beeswax, fatty alcohols, wool wax alcohols, low HLB (hydrophobellipophobe balance) emulsifiers, and assorted ionic and nonionic surfactants, singularly or in combination.
- lanolin lanolin derivatives, beeswax, fatty alcohols, wool wax alcohols, low HLB (hydrophobellipophobe balance) emulsifiers, and assorted ionic and nonionic surfactants, singularly or in combination.
- Controlled or sustained release transdermal or topical formulations can be achieved by the addition of time-release additives, such as polymeric structures, matrices, that are available in the art.
- the compositions may be administered through use of hot-melt extrusion articles, such as bioadhesive hot-melt extruded film.
- the formulation can comprise a crosslinked polycarboxylic acid polymer formulation.
- a cross-linking agent may be present in an amount that provides adequate adhesion to allow the system to remain attached to target epithelial or endothelial cell surfaces for a sufficient time to allow the desired release of the compound.
- An insert, transdermal patch, bandage or article can comprise a mixture or coating of polymers that provide release of the pharmaceutical agents at a constant rate over a prolonged period of time.
- the article, transdermal patch or insert comprises water- soluble pore forming agents, such as polyethylene glycol (PEG) that can be mixed with water insoluble polymers to increase the durability of the insert and to prolong the release of the active ingredients.
- PEG polyethylene glycol
- Transdermal devices may also comprise a water insoluble polymer.
- Rate controlling polymers may be useful for administration to sites where pH change can be used to effect release. These rate controlling polymers can be applied using a continuous coating film during the process of spraying and drying with the active compound.
- the coating formulation is used to coat pellets comprising the active ingredients that are compressed to form a solid, biodegradable insert.
- a polymer formulation can also be utilized to provide controlled or sustained release.
- Bioadhesive polymers described in the art may be used.
- a sustained-release gel and the compound may be incorporated in a polymeric matrix, such as a hydrophobic polymer matrix.
- a polymeric matrix include a microparticle. The microparticles can be microspheres, and the core may be of a different material than the polymeric shell.
- the polymer may be cast as a thin slab or film, a powder produced by grinding or other standard techniques, or a gel such as a hydrogel.
- the polymer can also be in the form of a coating or part of a bandage, stent, catheter, vascular graft, or other device to facilitate delivery of the pharmaceutical agent.
- the matrices can be formed by solvent evaporation, spray drying, solvent extraction and other methods known to those skilled in the art.
- Kits with unit doses of one or more of the agents described herein, usually in oral or injectable doses are provided.
- kits may include a container containing the unit dose, an informational package insert describing the use and attendant benefits of the drugs in treating disease, and optionally an appliance or device for delivery of the composition.
- kits and products that include the compound or salt of Formula (I), (I-A), or (I-B) and/or an immunomodulator inhibitor.
- the kit or product can include a package or container with a compound of Formula (I), (I-A), or (I-B) or pharmaceutically acceptable salt.
- kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound or salt of Formula (I), (I-A), or (I-B) in combination with an immunomodulator inhibitor that is separately provided.
- the kits can be used in the methods of treating cancer as described herein.
- kits or products can include both a compound or salt of Formula (I), (I-A), or (I-B) and an immunomodulator inhibitor.
- kits can include one or more containers or packages, which include one or both combination drugs together in a single container and/or package, or in separate packages/containers. In some instances, the two drugs are separately wrapped, but included in a single package, container or box.
- kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound or salt of Formula (I), (I-A), or (I-B) in combination with an immunomodulator inhibitor.
- the kits can be used in the methods of treating cancer as described herein.
- the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
- kits and products that include the compound or salt of Formula (I), (I-A), or (I-B) and/or at least one PD-1 inhibitor.
- the kit or product can include a package or container with a compound or salt of Formula (I), (I-A), or (I-B).
- kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound or salt of Formula (I), (I-A), or (I-B) in combination with an PD-1 inhibitor that is separately provided.
- the kits can be used in the methods of treating cancer as described herein.
- kits or products can include both a compound of Formula (I), (I-A), or (I-B) and at least one PD-1 inhibitor.
- kits can include one or more containers or packages, which include one or both combination drugs together in a single container and/or package, or in separate packages/containers. In some instances, the two drugs are separately wrapped, but included in a single package, container or box.
- kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound or salt of Formula (I), (I-A), or (I-B) in combination with an PD-1 inhibitor.
- the kits can be used in the methods of treating cancer as described herein.
- kits and products that include the compound of Formula (I), (I-A), or (I-B) and/or at least one PD-L1 inhibitor.
- the kit or product can include a package or container with a compound or salt of Formula (I), (I-A), or (I-B) .
- kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound or salt of Formula (I), (I- A), or (I-B) in combination with an PD-L1 inhibitor that is separately provided.
- the kits can be used in the methods of treating cancer as described herein.
- kits or products can include both a compound of Formula (I), (I- A), or (I-B) and at least one PD-L1 inhibitor.
- kits can include one or more containers or packages, which include one or both combination drugs together in a single container and/or package, or in separate packages/containers. In some instances, the two drugs are separately wrapped, but included in a single package, container or box.
- kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound or salt of Formula (I), (I- A), or (I-B) in combination with an PD-L1 inhibitor.
- the kits can be used in the methods of treating cancer as described herein. In some cases, the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
- kits and products that include the compound of Formula (I), (I-A), or (I-B) and/or at least one CTLA-4 inhibitor.
- the kit or product can include a package or container with a compound of Formula (I), (I-A), or (I-B).
- kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound of Formula (I), (I-A), or (I-B) in combination with an CTLA-4 inhibitor that is separately provided.
- the kits can be used in the methods of treating cancer as described herein.
- kits or products can include both a compound or salt of Formula (I), (LA), or (I-B) and at least one CTLA-4 inhibitor.
- kits can include one or more containers or packages, which include one or both combination drugs together in a single container and/or package, or in separate packages/containers. In some instances, the two drugs are separately wrapped, but included in a single package, container or box.
- kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound of Formula (I), (I-A), or (LB) in combination with an CTLA-4 inhibitor.
- the kits can be used in the methods of treating cancer as described herein. In some cases, the compound is selected from compounds 2, 3, 4, 14, 25, and 74. DOSINGS
- the dosage regimen for the compounds herein (e.g, an immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutical composition of any one thereof) will vary depending upon known factors, such as the pharmacodynamic characteristics of the particular agent and its mode and route of administration; the species, age, sex, health, medical condition, and weight of the recipient; the nature and extent of the symptoms; the kind of concurrent treatment; the frequency of treatment; the route of administration, the renal and hepatic function of the patient, and the effect desired.
- an immunomodulator inhibitor e.g., PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor
- a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutical composition of any one thereof will vary depending upon known factors, such as the pharmacodynamic characteristics of the particular agent and its mode and route of administration; the species, age, sex, health
- the dosing frequency for the therapeutic agent may vary, for example, from once per day to six times per day. That is, the dosing frequency may be QD, i.e., once per day, BID, i.e., twice per day; TID, i.e., three times per day; QID, i.e., four times per day; five times per day, or six times per day.
- dosing frequency may be BIW, i.e., twice weekly, TIW, i.e., three times a week, or QIW, i.e. four times a week.
- the treatment cycle may have a period of time where no therapeutic agent is administered.
- val administration refers to administration of the therapeutic agent (e.g, an immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (LA), or (I-B)) followed by void days or void weeks.
- the treatment cycle may be 3 weeks long which includes 2 weeks of dosing of the therapeutic agent(s) followed by 1 week where no therapeutic agent is administered. In some embodiments, the treatment cycle is 4 weeks long which includes 3 weeks of dosing followed by 1 week where no therapeutic agent is administered.
- the daily oral dosage of each active ingredient when used for the indicated effects, will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day.
- active ingredient e.g, immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (I- A), or (LB)
- an active ingredient e.g, an immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (LA), or (LB)
- an active ingredient may be administered at a dose of between about 10 mg/day and about 200 mg/day.
- an active ingredient e.g, an immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (LA), or (LB)
- an active ingredient may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day.
- the dose may be any value or subrange within the recited ranges.
- treatment cycle means a pre-determined period of time for administering the therapeutic agent (e.g, an immunomodulator inhibitor, PD-1 inhibitor, PD- L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (I-A), or (I-B)).
- the patient is examined at the end of each treatment cycle to evaluate the effect of the therapy.
- each of the treatment cycle has about 3 or more days.
- each of the treatment cycle has from about 3 days to about 60 days.
- each of the treatment cycle has from about 5 days to about 50 days.
- each of the treatment cycle has from about 7 days to about 28 days.
- each of the treatment cycle has 28 days.
- the treatment cycle has about 29 days. In some cases, the treatment cycle has about 30 days. In some cases, the treatment cycle has about 31 days. In some cases, the treatment cycle has about a month-long treatment cycle. In some cases, the treatment cycle is any length of time from 3 weeks to 8 weeks. In some cases, the treatment cycle is any length of time from 3 weeks to 6 weeks. In some cases, the treatment cycle is 3 weeks. In some cases, the treatment cycle is one month. In some cases, the treatment cycle is 4 weeks. In some cases, the treatment cycle is 5 weeks. In some cases, the treatment cycle is 6 weeks. In some cases, the treatment cycle is 7 weeks. In some cases, the treatment cycle is 8 weeks. In some cases, the duration of the treatment cycle may include any value or subrange within the recited ranges, including endpoints.
- drugs are administered at the maximum tolerated dose (“MTD”), which is the highest dose of drug that does not cause unacceptable side effects.
- MTD maximum tolerated dose
- the immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof is administered prior to administration of the compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof.
- the immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof is administered after administration of the compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof.
- the immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof is administered at about the same time as administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof or pharmaceutical composition thereof.
- separate administration of each inhibitor/compound, at different times and by different routes may be advantageous.
- the components in the combination i.e.
- a pharmaceutical composition comprises an immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient.
- a pharmaceutical composition comprises a compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient.
- the pharmaceutical compositions may be used in the methods simultaneously, separately, or sequentially.
- a pharmaceutical composition comprises an immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, and/or a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, for use in the methods for simultaneous, separate or sequential use.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at their respective MTDs.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof is dosed at its MTD and the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed in an amount less than its MTD.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof is dosed at an amount less than its MTD and the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD.
- the compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at less than their respective MTDs.
- the administration can be so timed that the peak pharmacokinetic effect of one compound coincides with the peak pharmacokinetic effect of the other.
- the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof is administered QD. In some cases, the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are administered BID. In some cases, the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, of the invention are administered TID.
- a single dose of compound or salt of Formula (I), (I- A), or (I- B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each administered once daily.
- the therapeutically effective amount of the immunomodulator inhibitor of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, the therapeutically effective amount of the immunomodulator inhibitor of the combination will range between about 10 mg/day and about 200 mg/day.
- the therapeutically effective amount of the immunomodulator inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day.
- the therapeutically effective amount of the immunomodulator inhibitor of the combination may be any value or subrange within the recited ranges.
- the therapeutically effective amount of the immunomodulator inhibitor of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, the therapeutically effective amount of the immunomodulator inhibitor of the combination will range between about 0.1 to about 50 mg/kg of body weight. In some embodiments the therapeutically effective amount of the immunomodulator inhibitor of the combination will range between about 10 mg/day and about 200 mg/day.
- the therapeutically effective amount of the immunomodulator inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day.
- the therapeutically effective amount of the immunomodulator inhibitor of the combination may be any value or subrange within the recited ranges.
- the immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, and the compound of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof can be formulated into separate or individual dosage forms which can be co-administered one after the other.
- the route of administration is the same (e.g. oral) two active compounds can be formulated into a single form for coadministration, both methods of co-administration, however, being part of the same therapeutic treatment or regimen.
- the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time).
- the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time).
- the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time).
- the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 5 mg to about 600 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg to about 150 mg. In some embodiments, the immunomodulator inhibitor is administered to a subject at about 10 mg to about 125 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 440 mg to about 520 mg. In some embodiments, the immunomodulator inhibitor is administered to a subject at about 10 mg to about 100 mg. In some embodiments, the immunomodulator inhibitor is administered to a subject at about 25 mg to about 100 mg.
- the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 50 mg to about 100 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof of is administered to a subject at about 5 mg to about 75 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, about 150 mg, about 170 mg, about 190 mg, about 210 mg, about 240 mg, about 300 mg, about 350 mg, about 400 mg, about 420 mg, about 440 mg, about 460 mg, about 480 mg,
- the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg, about 30 mg, about 45 mg, or about 60 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 30 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 45 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 60 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 240 mg.
- the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 480 mg.
- the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
- the immunomodulator inhibitor, or pharmaceutically salt thereof is administered once daily. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered twice daily. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered 3 times daily. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered once weekly. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered every other day. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered every 3 days. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered once a week. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered every two weeks.
- the immunomodulator inhibitor, or pharmaceutically salt thereof is administered every 4 weeks. [00344] In some embodiments, the immunomodulator inhibitor is administered intravenously in the amount of about 800 mg every 2 weeks (Q2W) or about 10 mg/kg every 2 weeks (Q2W). In one embodiment, the immunomodulator inhibitor, is administered intravenously over 60 minutes.
- the immunomodulator inhibitor is administered at a dose of 1200 mg intravenously once every 3 weeks (Q3W) or at a dose of 840 mg intravenously two weeks apart. In some cases, the immunomodulator inhibitor, is administered intravenously over 60 minutes.
- the immunomodulator inhibitor is administered at a dose of 10 mg/kg intravenously once every 2 weeks (Q2W). In one embodiment, the immunomodulator inhibitor, is administered intravenously over 60 minutes.
- a pharmaceutical composition comprises a PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient.
- a pharmaceutical composition comprises a compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient.
- the pharmaceutical compositions may be used in the methods simultaneously, separately, or sequentially.
- a pharmaceutical composition comprises a PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, and/or a compound or salt of Formula (I), (I-A), or (I- B), or a pharmaceutically acceptable salt thereof, for use in the methods which may be for simultaneous, separate or sequential use.
- the PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof is administered prior to administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof.
- the PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof is administered after administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof.
- the PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof is administered at about the same time as administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof or a pharmaceutical composition thereof.
- separate administration of each inhibitor, at different times and by different routes, may be advantageous.
- the components in the combination i.e. compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, and the PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof may need not to be administered at essentially the same time or in any order.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at their respective MTDs.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof is dosed at its MTD and the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed in an amount less than its MTD.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof is dosed at an amount less than its MTD and the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at less than their respective MTDs.
- the administration can be so timed that the peak pharmacokinetic effect of one compound coincides with the peak pharmacokinetic effect of the other.
- the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof is administered QD. In some cases, the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are administered BID. In some cases, the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, of the invention are administered TID.
- a single dose of compound or salt of Formula (I), (I-A), or (I- B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each administered once daily.
- the therapeutically effective amount of the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof) of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day.
- a PD-1 inhibitor the therapeutically effective amount of the PD-1 inhibitor of the combination will range between about 10 mg/day and about 200 mg/day.
- the therapeutically effective amount of the PD-1 inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day.
- the therapeutically effective amount of the PD-1 inhibitor of the combination may be any value or subrange within the recited ranges.
- the combination (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof) of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day.
- the therapeutically effective amount of the PD-1 inhibitor of the combination will range between about 0.1 to about 50 mg/kg of body weight.
- the therapeutically effective amount of the PD-1 inhibitor of the combination will range between about 10 mg/day and about 200 mg/day.
- the therapeutically effective amount of the PD-1 inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day.
- the therapeutically effective amount of the PD-1 inhibitor of the combination may be any value or subrange within the recited ranges.
- the combination (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof) of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per time, and most preferably between about 0.1 to about 20 mg/kg/time.
- the therapeutically effective amount of the PD-1 inhibitor of the combination will range between about 0.1 to about 50 mg/kg of body weight.
- the therapeutically effective amount of the PD-1 inhibitor of the combination will range between about 10 mg/time and about 200 mg/time.
- the therapeutically effective amount of the PD-1 inhibitor of the combination may be administered at a dose of about 10 mg/time, 20 mg/time, 30 mg/time, 40 mg/time, 50 mg/time, 60 mg/time, 70 mg/time, 80 mg/time, 90 mg/time, 100 mg/time, 110 mg/time, 120 mg/time, 130 mg/time, 140 mg/time, 150 mg/time, 160 mg/time, 170 mg/time, 180 mg/time, 190 mg/time, 200 mg/time, 240 mg/time, 480 mg/time, 600 mg/time 1000 mg/time.
- the therapeutically effective amount of the PD-1 inhibitor of the combination may be any value or subrange within the recited ranges.
- the time is 1 day. In some cases, the time is 7 days. In some cases, the time is 14 days. In some cases, the time is 21 days. In some cases, the time is 28 days.
- the PD-1 inhibitor e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof
- the compound of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof can be formulated into separate or individual dosage forms which can be co-administered one after the other.
- the route of administration is the same (e.g. oral) two active compounds can be formulated into a single form for co-administration, both methods of coadministration, however, being part of the same therapeutic treatment or regimen.
- the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of a PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time).
- the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 5 mg to about 600 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg to about 150 mg. In some embodiments, the PD-1 inhibitor is administered to a subject at about 10 mg to about 125 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 440 mg to about 520 mg. In some embodiments the PD-1 inhibitor is administered to a subject at about 10 mg to about 100 mg. In some embodiments, the PD-1 inhibitor is administered to a subject at about 25 mg to about 100 mg.
- the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 50 mg to about 100 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof of is administered to a subject at about 5 mg to about 75 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, about 150 mg, about 170 mg, about 190 mg, about 210 mg, about 240 mg, about 300 mg, about 350 mg, about 400 mg, about 420 mg, about 440 mg, about 460 mg, about 480 mg,
- the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg , about 30 mg, about 45 mg, or about 60 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 30 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 45 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 60 mg. In some embodiments, the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
- the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), or pharmaceutically salt thereof is administered once daily.
- the PD-1 inhibitor ⁇ . g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof is administered twice daily.
- the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), or pharmaceutically salt thereof is administered 3 times daily.
- the PD-1 inhibitor e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof
- the PD-1 inhibitor(e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof) is administered every other day.
- the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), is administered every 3 days. In some embodiments, the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), is administered once a week. In some embodiments, the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), is administered every two weeks. In some embodiments, the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), is administered every 4 weeks.
- the PD-1 inhibitor e.g.,
- the PD-1 inhibitor is nivolumab or a biosimilar thereof.
- nivolumab or a biosimilar thereof is administered at a dose of 240 mg intravenously once every 2 weeks (Q2W).
- nivolumab or a biosimilar thereof is administered at a dose of 480 mg intravenously once every 4 weeks (Q4W).
- nivolumab or a biosimilar thereof is administered intravenously over 30 minutes.
- the PD-1 inhibitor is pembrolizumab or a biosimilar thereof.
- pembrolizumab is administered at a dose of 200 mg intravenously once every 3 weeks (Q3W). In one embodiment, pembrolizumab or a biosimilar thereof is administered intravenously over 60 minutes.
- the PD-1 inhibitor is cemiplimab or a biosimilar thereof. In one embodiment, cemiplimab or a biosimilar thereof is administered at a dose of 350 mg intravenously once every 3 weeks (Q3W). In one embodiment, cemiplimab or a biosimilar thereof is administered intravenously over 30 minutes.
- the PD-1 inhibitor is tislelizumab or a biosimilar thereof.
- tislelizumab or a biosimilar thereof is administered at a dose of 200 mg intravenously once every 3 weeks (Q3W).
- the therapeutically effective amount of pembrolizumab or biosimilar thereof in the combination is about 350 mg administered every three weeks.
- the PD-1 inhibitor is tislelizumab or a biosimilar thereof.
- the therapeutically effective amount of tislelizumab, or biosimilar thereof, in the combination is about 200 mg administered every three weeks.
- the PD-1 inhibitor is atezolizumab or a biosimilar thereof.
- the therapeutically effective amount of atezolizumab or biosimilar thereof in the combination is about 1200 mg administered every three weeks.
- the PD-1 inhibitor is the PD-L1 inhibitor is avelumab or a biosimilar thereof.
- the therapeutically effective amount of avelumab or biosimilar thereof in the combination is about 10 mg/kg administered every two weeks or 800 mg every two weeks.
- the PD-L1 inhibitor is durvalumab or a biosimilar thereof.
- the therapeutically effective amount of durvalumab or biosimilar thereof in the combination is about 10 mg/kg administered every two weeks.
- a pharmaceutical compositions comprise a PD-Ll inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient.
- a pharmaceutical composition comprises a compound of Formula (II), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient.
- the pharmaceutical compositions may be used in the methods simultaneously, separately, or sequentially.
- the pharmaceutical compositions comprise a PD-Ll inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), or a pharmaceutically acceptable salt thereof, and/or a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, for use in the methods may be for simultaneous, separate or sequential use.
- the PD-L1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof is administered prior to administration of the compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof.
- the PD-L1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof is administered after administration of the compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof.
- the PD-L1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof is administered at about the same time as administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof.
- separate administration of each inhibitor, at different times and by different routes, may be advantageous.
- the components in the combination i.e.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, and the PD- L1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, may need not be necessarily administered at essentially the same time or in any order.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at their respective MTDs.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof is dosed at its MTD and the PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed in an amount less than its MTD. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at an amount less than its MTD and the PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at less than their respective MTDs.
- the administration can be so timed that the peak pharmacokinetic effect of one compound coincides with the peak pharmacokinetic effect of the other.
- the PD-L1 inhibitor e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof
- a pharmaceutically acceptable salt or a pharmaceutical composition thereof is administered QD.
- the PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are administered BID.
- the PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, of the invention are administered TID.
- a single dose of compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and PD-L1 inhibitor e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof
- PD-L1 inhibitor e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof
- the therapeutically effective amount of the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof) of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day.
- a PD-L1 inhibitor the therapeutically effective amount of the PD-L1 inhibitor of the combination will range between about 10 mg/day and about 200 mg/day.
- the therapeutically effective amount of the PD-L1 inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day.
- the therapeutically effective amount of the PD-L1 inhibitor of the combination may be any value or subrange within the recited ranges.
- the therapeutically effective amount of the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof) of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per time, and most preferably between about 0.1 to about 20 mg/kg/time. In some embodiments, the therapeutically effective amount of the PD-L1 inhibitor of the combination will range between about 0.1 to about 50 mg/kg of body weight. In some embodiments, the therapeutically effective amount of the PD-L1 inhibitor of the combination will range between about 10 mg/time and about 200 mg/time.
- the PD-L1 inhibitor e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof
- the therapeutically effective amount of the PD-L1 inhibitor of the combination may be administered at a dose of about 10 mg/time, 20 mg/time, 30 mg/time, 40 mg/time, 50 mg/time, 60 mg/time, 70 mg/time, 80 mg/time, 90 mg/time, 100 mg/time, 110 mg/time, 120 mg/time, 130 mg/time, 140 mg/time, 150 mg/time, 160 mg/time, 170 mg/time, 180 mg/time, 190 mg/time, 200 mg/time, 240 mg/time, 300 mg/time, 480 mg/time.
- the therapeutically effective amount of the PD-L1 inhibitor of the combination may be any value or subrange within the recited ranges. In some cases, the time is 1 day.
- the time is 7 days. In some cases, the time is 14 days. In some cases, the time is 21 days. In some cases, the time is 28 days.
- the PD-L1 inhibitor e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof
- the compound of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof can be formulated into separate or individual dosage forms which can be co-administered one after the other. Another option is that if the route of administration is the same (e.g.
- the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of a PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time).
- the PD-L1 inhibitor is administered to a subject at about 200 mg to about 500 mg. In some embodiments, PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 440 mg to about 520 mg. In some embodiments PD-L1 inhibitor is administered to a subject at about 10 mg to about 100 mg. In some embodiments, PD-L1 inhibitor is administered to a subject at about 240 mg to about 480 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 50 mg to about 100 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof of is administered to a subject at about 5 mg to about 75 mg.
- the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, about 150 mg, about 170 mg, about 190 mg, about 210 mg, about 240 mg, about 300 mg, about 350 mg, about 400 mg, about 420 mg, about 440 mg, about 460 mg, about 480 mg, about 500 mg, about 540 mg, about 580 mg, or about 650 mg.
- the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg , about 30 mg, about 45 mg, or about 60 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 30 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 240 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 480 mg. In some embodiments, the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
- the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), or pharmaceutically salt thereof is administered once daily. In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), is administered twice daily. In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), or pharmaceutically salt thereof is administered 3 times daily. In some embodiments, the PD-L1 inhibitor ⁇ .
- the PD-L1 inhibitor e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof
- the PD-L1 inhibitor is administered once weekly.
- the PD-L1 inhibitor e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof
- the PD-L1 inhibitor is administered every other day.
- the PD-L1 inhibitor e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof
- the PD-L1 inhibitor is administered every 3 days.
- the PD-L1 inhibitor e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof
- the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), is administered every two weeks. In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), is administered every 4 weeks.
- the PD-L1 inhibitor is avelumab or a biosimilar thereof. In one embodiment, avelumab or a biosimilar thereof is administered intravenously in the amount of about 800 mg every 2 weeks (Q2W) or about 10 mg/kg every 2 weeks (Q2W). In one embodiment, avelumab or a biosimilar thereof is administered intravenously over 60 minutes.
- the PD-L1 inhibitor is atezolizumab or a biosimilar thereof. In some cases, atezolizumab or a biosimilar thereof is administered at a dose of 1200 mg intravenously once every 3 weeks (Q3W) or at a dose of 840 mg intravenously two weeks apart. In some cases, atezolizumab or a biosimilar thereof is administered intravenously over 60 minutes.
- the PD-L1 inhibitor is durvalumab or a biosimilar thereof.
- durvalumab or a biosimilar thereof is administered at a dose of 10 mg/kg intravenously once every 2 weeks (Q2W). In one embodiment, durvalumab or a biosimilar thereof is administered intravenously over 60 minutes.
- a pharmaceutical composition comprises a CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient.
- a pharmaceutical composition comprises a compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient.
- the pharmaceutical compositions may be used in the methods simultaneously, separately, or sequentially.
- the pharmaceutical compositions comprising a CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, and/or a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, for use in the methods may be for simultaneous, separate or sequential use.
- the CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof is administered prior to administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof.
- the CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof is administered after administration of the compound or salt of Formula (I), (LA), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof.
- the CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof is administered at about the same time as administration of the compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof.
- separate administration of each inhibitor, at different times and by different routes, may be advantageous.
- the components in the combination i.e. compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof and the CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof need not be necessarily administered at essentially the same time or in any order.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at their respective MTDs.
- the compound or salt of Formula (I), (I- A), or (I- B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof is dosed at its MTD and the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed in an amount less than its MTD.
- the compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof is dosed at an amount less than its MTD and the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD.
- the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at less than their respective MTDs.
- the administration can be so timed that the peak pharmacokinetic effect of one compound coincides with the peak pharmacokinetic effect of the other.
- the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof is administered QD. In some cases, the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are administered BID. In some cases, the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, of the invention are administered TID.
- a single dose of compound or salt of Formula (I), (I- A), or (I- B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each administered once daily.
- the therapeutically effective amount of the CTLA-4 inhibitor of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, the therapeutically effective amount of the CTLA-4 inhibitor of the combination will range between about 10 mg/day and about 200 mg/day.
- the therapeutically effective amount of the CTLA-4 inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day.
- the therapeutically effective amount of the CTLA-4 inhibitor of the combination may be any value or subrange within the recited ranges.
- the therapeutically effective amount of the CTLA-4 inhibitor of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, the therapeutically effective amount of the CTLA-4 inhibitor of the combination will range between about 0.1 to about 50 mg/kg of body weight. In some embodiments, the therapeutically effective amount of the CTLA-4 inhibitor of the combination will range between about 10 mg/day and about 200 mg/day.
- the therapeutically effective amount of the CTLA-4 inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day.
- the therapeutically effective amount of the CTLA-4 inhibitor of the combination may be any value or subrange within the recited ranges.
- the CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, and the compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof can be formulated into separate or individual dosage forms which can be co-administered one after the other.
- the route of administration is the same (e.g. oral) two active compounds can be formulated into a single form for co-administration, both methods of coadministration, however, being part of the same therapeutic treatment or regimen.
- the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of a CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time).
- the CTLA-4 inhibitor is administered to a subject at about 200 mg to about 500 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 440 mg to about 520 mg. In some embodiments, the CTLA-4 inhibitor is administered to a subject at about 10 mg to about 100 mg. In some embodiments, the CTLA-4 inhibitor is administered to a subject at about 240 mg to about 480 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 50 mg to about 100 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof of is administered to a subject at about 5 mg to about 75 mg.
- the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, about 150 mg, about 170 mg, about 190 mg, about 210 mg, about 240 mg, about 300 mg, about 350 mg, about 400 mg, about 420 mg, about 440 mg, about 460 mg, about 480 mg, about 500 mg, about 540 mg, about 580 mg, or about 650 mg.
- the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg, about 30 mg, about 45 mg, or about 60 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 30 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 240 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 480 mg. In some embodiments, the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
- the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered once daily. In some embodiments, the CTLA-4 inhibitor, is administered twice daily. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered 3 times daily. In some embodiments, the CTLA-4 inhibitor, is administered once weekly. In some embodiments, the CTLA-4 inhibitor, is administered every other day. In some embodiments, the CTLA-4 inhibitor, is administered every 3 days. In some embodiments, the CTLA-4 inhibitor, is administered once a week. In some embodiments, the CTLA-4 inhibitor, is administered every two weeks. In some embodiments, the CTLA-4 inhibitor, is administered every 4 weeks.
- a single dose of compound or salt of Formula (II), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof is administered per day (i.e., in about 24 hour intervals) (i.e., QD).
- two doses of the compound or salt of Formula (II), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are administered per day (i.e., BID).
- three doses of the compound or salt of Formula (II), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are administered per day (i.e., TID).
- the present disclosure provides methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (LA), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the present disclosure provides methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (LA), or (LB), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the cancer is a KRas G12D-associated cancer. In one embodiment, the cancer is a KRas G12V- associated cancer. In one embodiment, the cancer is a KRas wildtype-associated cancer. In some cases, the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the cancer is a solid tumor cancer. In some cases, the cancer is a solid tumor cancer with a KRAS mutation. In some cases, the cancer is a solid tumor cancer with KRAS wildtype. In some cases, the cancer is a solid tumor cancer with a G12D mutation. In some cases, the cancer is a solid tumor cancer with a G12V mutation.
- the cancer is a KRAS wildtype- associated cancer.
- the KRas G12D-associated cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer.
- the compound of Formula (I), (I- A), or (I-B) is selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of anyone thereof.
- the compound of Formula (I), (I- A), or (I-B) is compound 2.
- the compound of Formula (I), (I- A), or (I-B) is compound 3.
- the compound of Formula (I), (I- A), or (I-B) is compound 4.
- the compound of Formula (I), (I-A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a combination of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (II) selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the compound of Formula (II) is compound 2.
- the compound of Formula (II) is compound 3.
- the compound of Formula (II) is compound 63.
- the compound of Formula (II) is compound 4.
- the compound of Formula (II) is compound 14.
- the compound of Formula (II) is compound 25.
- the compound of Formula (II) is compound 74.
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (I-A), or (I-B) selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 2.
- the compound of Formula (I), (I-A), or (I-B) is compound 3.
- the compound of Formula (I), (I-A), or (I-B) is compound 4.
- the compound of Formula (I), (I-A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
- the present disclosure provides methods for increasing the sensitivity of a cancer cell to a compound or salt of Formula (I), (I-A), or (I-B), comprising contacting the cancer cell with an effective amount of a combination of a compound of Formula (I), (I-A), or (I- B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, wherein the immunomodulator inhibitor synergistically increases the sensitivity of the cancer cell to the compound or salt of Formula (I), (I-A), or (I-B).
- the contacting is in vitro. In one embodiment, the contacting is in vivo.
- the present disclosure provides methods of inhibiting KRas G12 mutants or wildtype in a subject comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the method may inhibit KRas G12 mutants or wildtype activity in a cell.
- inhibiting KRas G12 mutants or wildtype activity in a cell may include contacting the cell in which inhibition of KRas G12 mutants or wildtype activity is desired with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutical compositions containing the compound or pharmaceutically acceptable salt thereof and an immunomodulator inhibitor or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- a cell in which inhibition of KRas G12 mutants or wildtype activity is desired is contacted with an effective amount of a compound of Formula (I), (I-A), or (I-B) or pharmaceutically acceptable salt of any one thereof and an immunomodulator inhibitor to negatively modulate the activity of KRas G12 mutants or wildtype.
- an immunomodulator inhibitor to negatively modulate the activity of KRas G12 mutants or wildtype.
- the methods described herein are designed to inhibit undesired cellular proliferation resulting from enhanced KRas G12 mutants or wildtype activity within the cell.
- the cells may be contacted in a single dose or multiple doses in accordance with a particular treatment regimen to effect the desired negative modulation of KRas G12 mutants or wildtype.
- the ability of compounds to bind KRas G12 mutants or wildtype may be monitored in vitro using well known methods.
- the inhibitory activity of compounds of Formula (I), (I- A), or (I-B) and an immunomodulator inhibitor in cells may be monitored, for example, by measuring the inhibition of KRas G12 mutants or wildtype activity of the amount of phosphorylated ERK.
- methods of treating cancer in a patient in need thereof comprising administering to said patient a therapeutically effective amount of a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt thereof are provided and an immunomodulator inhibitor.
- compositions and methods provided herein may be used for the treatment of a KRas G12 mutants or wildtype-associated cancer in a patient in need thereof, comprising administering to the patient a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and an immunomodulator inhibitor are provided.
- the KRas G12 mutants or wildtype associated cancer is lung cancer.
- the KRas G12 mutants or wildtype associated cancer is solid tumor cancer.
- the KRAS G12 mutants are selected from G12D and G12V.
- the KRAS G12 mutants is G12D.
- the KRAS G12 mutants is G12V.
- the method of treating a cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of a immunomodulator inhibitor and a compound or salt of Formula (I), (I- A), or (I-B), synergistically increases the potency of the compound or salt of Formula (I), (I- A), or (I-B).
- the synergistic increase in potency of the compound or salt of Formula (I), (I- A), or (I-B) results in an improved efficacy of the compound or salt of Formula (I), (I-A), or (I-B).
- the therapeutically effective amount of a combination of two compounds is an amount that together synergistically increases the activity of the combination in comparison to the therapeutically effective amount of each compound in the combination, i.e., more than merely additive.
- the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a pharmaceutical composition thereof and a KRas G12D inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof results in a complete durable response in subjects relative to treatment with only the KRas G12D inhibitor.
- the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a pharmaceutical composition thereof and a KRas G12D inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof results in an increased duration of overall survival (“OS”) in subjects relative to treatment with only the KRas G12D inhibitor.
- OS overall survival
- the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a pharmaceutical composition thereof and a KRas G12D inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof results in an increased duration of progression-free survival (“PFS”) in subjects relative to treatment with only the KRas G12D inhibitor.
- PFS progression-free survival
- the therapeutically effective amount of a combination of two compounds is an amount that together synergistically increases the activity of the combination in comparison to the therapeutically effective amount of each compound in the combination, i.e., more than merely additive.
- the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a pharmaceutical composition thereof and a KRas G12V inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof results in a complete durable response in subjects relative to treatment with only the KRas G12V inhibitor.
- the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a pharmaceutical composition thereof and a KRas G12V inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof results in an increased duration of overall survival (“OS”) in subjects relative to treatment with only the KRas G12V inhibitor.
- OS overall survival
- the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a pharmaceutical composition thereof and a KRas G12V inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof results in an increased duration of progression-free survival (“PFS”) in subjects relative to treatment with only the KRas G12V inhibitor.
- PFS progression-free survival
- the compound and inhibitor are administered on separate days. In some cases, the compound and inhibitor are administered on the same day.
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a PD- 1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer.
- the cancer is a solid tumor cancer.
- the cancer is a solid tumor cancer with a KRAS mutation.
- the cancer is a KRAS wildtype-associated cancer.
- the cancer is a KRas G12D-associated cancer.
- the cancer is a KRas G12V-associated cancer.
- the cancer is a KRas wildtype-associated cancer.
- the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer.
- the cancer is a solid tumor cancer.
- the cancer is a solid tumor cancer with a KRAS mutation. In some cases, the cancer is a solid tumor cancer with KRAS wildtype. In some cases, the cancer is a solid tumor cancer with a G12D mutation. In some cases, the cancer is a solid tumor cancer with a G12V mutation. In some cases, the cancer is a KRAS wildtype-associated cancer. In some cases, the KRas G12D-associated cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the compound of Formula (I), (I-A), or (I-B) is selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of anyone thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 2. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 3. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 4. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (I-A), or (I-B) selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 2.
- the compound of Formula (I), (I-A), or (I-B) is compound 3.
- the compound of Formula (I), (I-A), or (I-B) is compound 4.
- the compound of Formula (I), (I- A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
- the present disclosure provides methods for increasing the sensitivity of a cancer cell to a compound or salt of Formula (I), (I-A), or (I-B), comprising contacting the cancer cell with an effective amount of a combination of a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, wherein the PD-1 inhibitor synergistically increases the sensitivity of the cancer cell to the compound or salt of Formula (I), (I-A), or (I-B).
- the contacting is in vitro. In one embodiment, the contacting is in vivo.
- the present disclosure provides methods of inhibiting KRas G12 mutants in a subject comprising administering to the subject a therapeutically effective amount of a combination of a PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the method may inhibit KRas G12 mutants or wildtype activity in a cell.
- inhibiting KRas G12 mutants or wildtype activity in a cell may include contacting the cell in which inhibition of KRas G12 mutants or wildtype activity is desired with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutical compositions containing the compound or pharmaceutically acceptable salt of any one thereof and a PD-1 inhibitor.
- the contacting is in vitro. In some cases, the contacting is in vivo.
- a cell in which inhibition of KRas G12 mutants or wildtype activity is desired is contacted with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutically acceptable salt of any one thereof and a PD-1 inhibitor to negatively modulate the activity of KRas G12 mutants or wildtype.
- the methods described herein are designed to inhibit undesired cellular proliferation resulting from enhanced KRas G12 mutants or wildtype activity within the cell.
- the cells may be contacted in a single dose or multiple doses in accordance with a particular treatment regimen to effect the desired negative modulation of KRas G12 mutants or wildtype.
- the ability of compounds to bind KRas G12 mutants or wildtype may be monitored in vitro using well known methods.
- the inhibitory activity of exemplary compounds and a PD-1 inhibitor in cells may be monitored, for example, by measuring the inhibition of KRas G12 mutants or wildtype activity of the amount of phosphorylated ERK.
- the present disclosure provides methods of reprogramming a tumor microenvironment (TME) of a subject in favor of antitumor immunity, wherein reprogramming includes modulating tumor cell cytokine/chemokine release in the tumor microenvironment of the subject.
- the method includes providing a compound described herein (a compound of Formula (I, e.g., a compound selected from compounds 2, 3, 4, 14, 25, and 74).
- the method also includes providing an immunomodulator (e.g., checkpoint inhibitors (inhibitors targeting: cytotoxic T lymphocyte associated antigen 4 (CTLA-4), e.g.
- CTLA-4 cytotoxic T lymphocyte associated antigen 4
- the method can also include providing a compound of Formula (I) and an immunomodulator in combination (sequentially or simultaneously).
- the subject has a KRas-associated cancer.
- the present disclosure provides methods of treating a KRas G12D- associated cancer in a subject in need thereof, comprising administering to the subject a combination of an immunomodulator, and a compound of Formula (I) or a pharmaceutically acceptable salt thereof.
- the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
- the present disclosure provides methods of treating a KRas G12V- associated cancer in a subject in need thereof, comprising administering to the subject a combination of an immunomodulator, and a compound of Formula (II) or a pharmaceutically acceptable salt thereof.
- the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
- methods of treating cancer in a patient in need thereof comprising administering to said patient a therapeutically effective amount of a compound or salt of Formula (I), (I-A), or (I-B) or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt thereof are provided and a PD-1 inhibitor.
- compositions and methods provided herein may be used for the treatment of a KRas G12 mutants or wildtype -associated cancer in a patient in need thereof, comprising administering to the patient a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and a PD-1 inhibitor are provided.
- the KRas G12 mutants or wildtype associated cancer is lung cancer.
- compositions and methods provided herein may be used for the treatment of a KRas G12D-associated cancer in a patient in need thereof, comprising administering to the patient a therapeutically effective amount of a compound of Formula (I), (I-A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and a PD-1 inhibitor are provided.
- the method of treating a cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of a PD-1 inhibitor and a compound or salt of Formula (I), (I-A), or (I-B), synergistically increases the potency of the compound or salt of Formula (I), (I-A), or (I-B).
- the synergistic increase in potency of the compound or salt of Formula (I), (I-A), or (I-B) results in an improved efficacy of the compound or salt of Formula (I), (I-A), or (I-B).
- the compound and inhibitor are administered on separate days. In some cases, the compound and inhibitor are administered on the same day.
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer.
- the cancer is a solid tumor cancer.
- the cancer is a solid tumor cancer with a KRAS mutation.
- the cancer is a KRAS wildtype-associated cancer.
- the cancer is a KRas G12D-associated cancer.
- the cancer is a KRas G12V-associated cancer.
- the cancer is a KRas wildtype-associated cancer.
- the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer.
- the cancer is a solid tumor cancer.
- the cancer is a solid tumor cancer with a KRAS mutation. In some cases, the cancer is a solid tumor cancer with KRAS wildtype. In some cases, the cancer is a solid tumor cancer with a G12D mutation. In some cases, the cancer is a solid tumor cancer with a G12V mutation. In some cases, the cancer is a KRAS wildtype-associated cancer. In some cases, the KRas G12D-associated cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the compound of Formula (I), (I-A), or (I-B) is selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of anyone thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 2. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 3. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 4. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (I-A), or (I-B) selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 2.
- the compound of Formula (I), (I-A), or (I-B) is compound 3.
- the compound of Formula (I), (I-A), or (I-B) is compound 4.
- the compound of Formula (I), (I- A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
- the present disclosure provides methods for increasing the sensitivity of a cancer cell to a compound or salt of Formula (I), (I-A), or (I-B), comprising contacting the cancer cell with an effective amount of a combination of a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, wherein the PD-L1 inhibitor synergistically increases the sensitivity of the cancer cell to the compound or salt of Formula (I), (I-A), or (I-B).
- the contacting is in vitro. In one embodiment, the contacting is in vivo.
- the present disclosure provides methods of inhibiting KRas G12 mutants or wildtype in a subject comprising administering to the subject a therapeutically effective amount of a combination of a PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the method may inhibit KRas G12 mutants or wildtype activity in a cell.
- inhibiting KRas G12 mutants or wildtype activity in a cell may include contacting the cell in which inhibition of KRas G12 mutants or wildtype activity is desired with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutical compositions containing the compound or pharmaceutically acceptable salt of any one thereof and a PD-L1 inhibitor.
- the contacting is in vitro. In some cases, the contacting is in vivo.
- a cell in which inhibition of KRas G12 mutants or wildtype activity is desired is contacted with an effective amount of a compound of Formula (I), (I-A), or (I-B) or pharmaceutically acceptable salt of any one thereof and an PD-L1 inhibitor to negatively modulate the activity of KRas G12 mutants or wildtype.
- the methods described herein are designed to inhibit undesired cellular proliferation resulting from enhanced KRas G12 mutants or wildtype activity within the cell.
- the cells may be contacted in a single dose or multiple doses in accordance with a particular treatment regimen to effect the desired negative modulation of KRas G12 mutants or wildtype.
- the ability of compounds to bind KRas G12 mutants or wildtype may be monitored in vitro using well known methods.
- the inhibitory activity of exemplary compounds and an PD-L1 inhibitor in cells may be monitored, for example, by measuring the inhibition of KRAS G12 mutants and wildtype activity of the amount of phosphorylated ERK.
- methods of treating cancer in a patient in need thereof comprising administering to said patient a therapeutically effective amount of a compound or salt of Formula (I), (I-A), or (I-B) or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt thereof are provided and an PD- L1 inhibitor.
- compositions and methods provided herein may be used for the treatment of a KRas G12 mutants or wildtype-associated cancer in a patient in need thereof, comprising administering to the patient a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and a PD-L1 inhibitor are provided.
- the KRas G12 mutants or wildtype associated cancer is lung cancer.
- the method of treating a cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of a PD-L1 inhibitor and a compound or salt of Formula (I), (I-A), or (I-B), synergistically increases the potency of the compound or salt of Formula (I), (I-A), or (I-B).
- the synergistic increase in potency of the compound or salt of Formula (I), (I-A), or (I-B) results in an improved efficacy of the compound or salt of Formula (I), (I-A), or (I-B).
- the compound and inhibitor are administered on separate days. In some cases, the compound and inhibitor are administered on the same day.
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer.
- the cancer is a solid tumor cancer.
- the cancer is a solid tumor cancer with a KRAS mutation.
- the cancer is a KRAS wildtype-associated cancer.
- the cancer is a KRas G12D-associated cancer. In one embodiment, the cancer is a KRas G12V- associated cancer. In one embodiment, the cancer is a KRas wildtype-associated cancer. In some cases, the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the cancer is a solid tumor cancer. In some cases, the cancer is a solid tumor cancer with a KRAS mutation. In some cases, the cancer is a solid tumor cancer with KRAS wildtype. In some cases, the cancer is a solid tumor cancer with a G12D mutation. In some cases, the cancer is a solid tumor cancer with a G12V mutation.
- the cancer is a KRAS wildtype- associated cancer.
- the KRas G12D-associated cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer.
- the compound of Formula (I), (I- A), or (I-B) is selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of anyone thereof.
- the compound of Formula (I), (I- A), or (I-B) is compound 2.
- the compound of Formula (I), (I- A), or (I-B) is compound 3.
- the compound of Formula (I), (I- A), or (I-B) is compound 4.
- the compound of Formula (I), (I-A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
- the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (I-A), or (I-B) selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the compound of Formula (I), (I-A), or (I-B) is compound 2.
- the compound of Formula (I), (I-A), or (I-B) is compound 3.
- the compound of Formula (I), (I-A), or (I-B) is compound 4.
- the compound of Formula (I), (I- A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
- the present disclosure provides methods for increasing the sensitivity of a cancer cell to a compound or salt of Formula (I), (I-A), or (I-B), comprising contacting the cancer cell with an effective amount of a combination of a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, wherein the CTLA-4 inhibitor synergistically increases the sensitivity of the cancer cell to the compound or salt of Formula (I), (I-A), or (I-B).
- the contacting is in vitro. In one embodiment, the contacting is in vivo.
- the present disclosure provides methods of inhibiting KRas G12 mutants or wildtype in a subject comprising administering to the subject a therapeutically effective amount of a combination of a CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
- the method may inhibit KRas G12 mutants or wildtype activity in a cell.
- inhibiting KRas G12 mutants or wildtype activity in a cell may include contacting the cell in which inhibition of KRas G12 mutants activity is desired with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutical compositions containing the compound or pharmaceutically acceptable salt of any one thereof and a CTLA-4 inhibitor.
- the contacting is in vitro. In some cases, the contacting is in vivo.
- a cell in which inhibition of KRas G12 mutants or wildtype activity is desired is contacted with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutically acceptable salt of any one thereof and a CTLA-4 inhibitor to negatively modulate the activity of KRas G12 mutants or wildtype.
- the methods described herein are designed to inhibit undesired cellular proliferation resulting from enhanced KRas G12 mutants or wildtype activity within the cell.
- the cells may be contacted in a single dose or multiple doses in accordance with a particular treatment regimen to effect the desired negative modulation of KRas G12 mutants or wildtype.
- the ability of compounds to bind KRas G12 mutants or wildtype may be monitored in vitro using well known methods.
- the inhibitory activity of exemplary compounds and a CTLA-4 inhibitor in cells may be monitored, for example, by measuring the inhibition of KRas G12 mutants or wildtype activity of the amount of phosphorylated ERK.
- methods of treating cancer in a patient in need thereof comprising administering to said patient a therapeutically effective amount of a compound or salt of Formula (I), (I-A), or (I-B) or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt thereof are provided and a CTLA- 4 inhibitor.
- compositions and methods provided herein may be used for the treatment of a KRas G12 mutants or wildtype-associated cancer in a patient in need thereof, comprising administering to the patient a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and a CTLA-4 inhibitor are provided.
- KRas G12 mutants or wildtype associated cancer is lung cancer.
- compositions and methods provided herein may be used for the treatment of a KRas G12D-associated cancer in a patient in need thereof, comprising administering to the patient a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and a CTLA-4 inhibitor are provided.
- the method of treating a cancer in a subject in need thereof comprising administering to the subject a therapeutically effective amount of a combination of a CTLA-4 inhibitor and a compound or salt of Formula (I), (LA), or (LB), synergistically increases the potency of the compound or salt of Formula (I), (LA), or (LB).
- the synergistic increase in potency of the compound or salt of Formula (I), (LA), or (LB) results in an improved efficacy of the compound or salt of Formula (I), (LA), or (LB).
- contacting refers to the bringing together of indicated moieties in an in vitro system or an in vivo system.
- "contacting" a KRas G12 mutants or wildtype with a compound provided herein and an immunomodulator inhibitor includes the administration of the compound provided herein and an immunomodulator inhibitor to an individual or patient, such as a human, having KRas G12D and/or other G12 mutants, as well as, for example, introducing a compound provided herein and an immunomodulator inhibitor into a sample containing a cellular or purified preparation containing the KRas G12D and/or other G12 mutants.
- compositions and methods provided herein may be used for the treatment of a wide variety of cancers including tumors such as lung, prostate, breast, brain, skin, cervical carcinomas, testicular carcinomas, etc. More particularly, cancers that may be treated by the compositions and methods of the invention include, but are not limited to tumor types such as astrocytic, breast, cervical, colorectal, endometrial, esophageal, gastric, head and neck, hepatocellular, laryngeal, lung, oral, ovarian, prostate and thyroid carcinomas and sarcomas.
- tumor types such as astrocytic, breast, cervical, colorectal, endometrial, esophageal, gastric, head and neck, hepatocellular, laryngeal, lung, oral, ovarian, prostate and thyroid carcinomas and sarcomas.
- these compounds can be used to treat: Cardiac: sarcoma (angiosarcoma, fibrosarcoma, rhabdomyosarcoma, liposarcoma), myxoma, rhabdomyoma, fibroma, lipoma and teratoma; Lung: bronchogenic carcinoma (squamous cell, undifferentiated small cell, undifferentiated large cell, adenocarcinoma), alveolar (bronchiolar) carcinoma, bronchial adenoma, sarcoma, lymphoma, chondromatous hamartoma, mesothelioma; Gastrointestinal: esophagus (squamous cell carcinoma, adenocarcinoma, leiomyosarcoma, lymphoma), stomach (carcinoma, lymphoma, leiomyosarcoma), pancreas (ductal adenocarcinoma, insulinom
- the concentration and route of administration to the patient will vary depending on the cancer to be treated.
- the compounds, pharmaceutically acceptable salts thereof and pharmaceutical compositions comprising such compounds and salts also may be coadministered with other anti-neoplastic compounds, e.g., chemotherapy, or used in combination with other treatments, such as radiation or surgical intervention, either as an adjuvant prior to surgery or post-operatively.
- the compounds described herein can be used in the preparation of medicaments for the prevention or treatment of diseases or conditions.
- a method for treating any of the diseases or conditions described herein in a subject in need of such treatment involves administration of pharmaceutical compositions containing at least one compound described herein, or a pharmaceutically acceptable salt, pharmaceutically acceptable prodrug, or pharmaceutically acceptable solvate thereof, in therapeutically effective amounts to said subject.
- the compositions containing the compound(s) described herein can be administered for prophylactic and/or therapeutic treatments. In therapeutic applications, the compositions are administered to a patient already suffering from a disease or condition, in an amount sufficient to cure or at least partially arrest the symptoms of the disease or condition. Amounts effective for this use will depend on the severity and course of the disease or condition, previous therapy, the patient's health status, weight, and response to the drugs, and the judgment of the treating physician.
- compositions containing the compounds described herein are administered to a patient susceptible to or otherwise at risk of a particular disease, disorder or condition. Such an amount is defined to be a "prophylactically effective amount or dose.”
- a patient susceptible to or otherwise at risk of a particular disease, disorder or condition is defined to be a "prophylactically effective amount or dose.”
- dose a pharmaceutically effective amount or dose.
- the precise amounts also depend on the patient's state of health, weight, and the like.
- effective amounts for this use will depend on the severity and course of the disease, disorder or condition, previous therapy, the patient's health status and response to the drugs, and the judgment of the treating physician.
- the administration of the compounds may be administered chronically, that is, for an extended period of time, including throughout the duration of the patient’s life in order to ameliorate or otherwise control or limit the symptoms of the patient’s disease or condition.
- a maintenance dose is administered if necessary.
- the dosage or the frequency of administration, or both can be reduced, as a function of the symptoms, to a level at which the improved disease, disorder or condition is retained. Patients can, however, require intermittent treatment on a long-term basis upon any recurrence of symptoms.
- the amount of a given agent that will correspond to such an amount will vary depending upon factors such as the particular compound, disease or condition and its severity, the identity (e.g., weight) of the subject or host in need of treatment, but can nevertheless be determined in a manner recognized in the field according to the particular circumstances surrounding the case, including, e.g., the specific agent being administered, the route of administration, the condition being treated, and the subject or host being treated.
- doses employed for adult human treatment will typically be in the range of about 0.02 - about 5000 mg per day, in some embodiments, about 1 - about 1500 mg per day.
- the desired dose may conveniently be presented in a single dose or as divided doses administered simultaneously (or over a short period of time) or at appropriate intervals, for example as two, three, four or more sub -doses per day.
- the pharmaceutical composition described herein may be in unit dosage forms suitable for single administration of precise dosages.
- the formulation is divided into unit doses containing appropriate quantities of one or more compound.
- the unit dosage may be in the form of a package containing discrete quantities of the formulation.
- Nonlimiting examples are packaged tablets or capsules, and powders in vials or ampoules.
- Aqueous suspension compositions can be packaged in single-dose non-reclosable containers.
- multiple-dose reclosable containers can be used, in which case it is typical to include a preservative in the composition.
- formulations for parenteral injection may be presented in unit dosage form, which include, but are not limited to ampoules, or in multi-dose containers, with an added preservative.
- Toxicity and therapeutic efficacy of such therapeutic regimens can be determined by standard pharmaceutical procedures in cell cultures or experimental animals, including, but not limited to, the determination of the LD50 (the dose lethal to 50% of the population) and the ED50 (the dose therapeutically effective in 50% of the population).
- the dose ratio between the toxic and therapeutic effects is the therapeutic index and it can be expressed as the ratio between LD50 and ED50.
- Compounds exhibiting high therapeutic indices are preferred.
- the data obtained from cell culture assays and animal studies can be used in formulating a range of dosage for use in human.
- the dosage of such compounds lies preferably within a range of circulating concentrations that include the ED50 with minimal toxicity.
- the dosage may vary within this range depending upon the dosage form employed and the route of administration utilized.
- the invention provides a method of treating or preventing a disease, state or condition in a patient in need thereof comprising administering to the patient an effective amount of a compound of any one of embodiments of the invention or a pharmaceutically acceptable salt thereof.
- the disease, state or condition may be selected from a group as described elsewhere herein.
- compounds herein can adopt to selectively eliminate an over activated KRas signaling which is induced by KRas mutations by directly binding with the mutated KRas protein, either by stabilizing its GDP bound form (the inactive form) or by blocking the interaction between GTP bound form and its downstream target protein.
- another way is to hijack the protein degradation mechanism in a cell and leverage E3 ligases’ (like VHL, CRBN or IAPS) substrate specificity through a bi-functional molecule called Proteolysis targeting chimera (PROTAC) (Winter GE, Buckley DL, Paulk J, Roberts JM, Souza A, Dhe- Paganon S, Bradner JE.
- a bifunctional compound composed of a target protein (i.e., KRAS G12D)-binding moiety and an E3 ubiquitin ligase-binding moiety, which may induce proteasome- mediated degradation of selected proteins.
- the bifunctional compound comprises a target protein (i.e., KRAS G12D)-binding moiety and an E3 ubiquitin ligase-binding moiety known in the art.
- disclosed herein is the use of the compound disclosed herein in the preparation of degrading a target protein compound by using chemical modification of the compound disclosed herein.
- the target protein-binding moiety is derived from a compound of Formula (I), (I-A), or (I-B).
- the compounds of the present disclosure can generally be prepared in a number of ways well known to those skilled in the art of organic synthesis.
- compounds of the present disclosure can be synthesized using the methods described herein, together with synthetic methods known in the art of synthetic organic chemistry, or variations thereof as appreciated by those skilled in the art.
- the compounds of the present disclosure may be prepared as described in the schemes and examples described elsewhere herein.
- Step 1 Synthesis of 2-nitro-prop-2-ynyl-benzenesulfonamide (Int-la).
- a solution of prop-2-yl-l -amine (1.87 mL, 29.1 mmol) and 7V,7V-diisopropylethylamine (10.15 mL, 58.3 mmol) in DCM (100 mL) was cooled to 0 °C.
- 2-nitrobenzenesulfonyl chloride (6.46 g, 29.15 mmol) was added portion wise.
- the solution was then allowed to warm to rt and was further stirred for 1 hr.
- the mixture was washed with water and brine, dried over ISfeSCh, concentrated.
- Step 2 Synthesis of (3-chloropropyl)-2-nitro-prop-2-ynyl-benzenesulfonamide (Int- 1b).
- a solution of 2-nitro-prop-2-ynyl-benzenesulfonamide (Int-la, 1.0 g, 4.16 mmol) and cesium carbonate (6.764 g, 20.81 mmol) in acetone (30 mL) was added neat l-bromo-3 -chloropropane (6.05 mL, 61.19 mmol) dropwise.
- the reaction mixture was stirred at rt for 2 hrs. The mixture was concentrated, then washed with water and brine, dried over Na2SO4, concentrated.
- Step 3 Synthesis of ethyl 5-(2-nitrophenyl)sulfonyl -4,6,7, 8-tetrahydropyrazolo[ 1,5- a][l,4]diazepine-2-carboxylate (Int-lc).
- Step 4 Synthesis of ethyl 5-(2-nitrophenyl)sulfonyl -4,6,7, 8-tetrahydropyrazolo[ 1,5- a][l,4]diazepine-2-carboxylate (Int-ld).
- Step 5 Synthesis of A,A-dimethyl-5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (Int-le).
- Step 6 Synthesis of 3-chloro- N ,N -dimethyl-5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (Int-lf).
- Step 7 Synthesis of 3-chloro-N ,N -dimethyl-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a][l,4]diazepine-2-carboxamide (Intermediate 1).
- Step 1 Synthesis of diethyl 4-chloro-lH-pyrazole-3,5-dicarboxylate (Int-lg). To a solution of diethyl lH-pyrazole-3,5-dicarboxylate (20 g, 94.25 mmol) in Acetic acid (360mL) was added dropwise NaClO (264 mL, 1980 mmol). After stirring for 3 h at 25 °C, the remaining mixture was diluted with water, and the mixture was extracted with ethyl acetate.
- Step 2 Synthesis of diethyl l-[3-(tert-butoxycarbonylamino)propyl]-4-chloro- pyrazole-3,5-dicarboxylate (Int-lh).
- Diethyl l-[3-(tert- butoxycarbonylamino)propyl]-4-chloro-pyrazole-3,5-dicarboxylate Int-lg, 24.7 g,24.46 mmol, 39.70% yield
- DMF 150mL
- tert-butyl N-(3-bromopropyl)carbamate 16141.77 mg, 67.79 mmol
- Potassium Iodide (204.6 mg, 1.2 3mmol
- CS2CO3 40157.83 mg, 123.25 mmol
- Step 3 Synthesis of diethyl l-(3-aminopropyl)-4-chloro-pyrazole-3,5- dicarboxylate;hydrochloride (Int-li). To a solution of diethyl l-(3-aminopropyl)-4-chloro- pyrazole-3,5-dicarboxylate;hydrochloride (Int-lh, 25.5 g, 23.99 mmol, 98.05% yield) in 1,4- Di oxane (50mL) was added HCl/Dioxane (150 mL, 600 mmol), then the mixture was stirred at 25 °C for Ih.
- Step 4 Synthesis of ethyl 3-chloro-4-oxo-5,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxylate (Int-lj).
- Diethyl l-(3-aminopropyl)-4-chloro- pyrazole-3,5-dicarboxylate;hydrochloride Int-li, 25.5 g, 23.99 mmol
- EtsN (16.74 mL, 119.93 mmol
- Step 5 Synthesis of ethyl 3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a][l,4]diazepine-2-carboxylate (Int-lk).
- ethyl 3-chloro-4-oxo-5, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylate Int-lj, 4.8 g, 18.63 mmol
- THF 60mL
- Borane-Methyl Sulfide Complex 27.94 mL, 55.89 mmol
- Step 6 Synthesis of 05-tert-butyl 02-ethyl 3-chloro-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2,5-di carboxylate (Int-11).
- Step 7 Synthesis of 5-tert-butoxycarbonyl-3-chloro-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxylic acid (Int-lm).
- Step 8 Synthesis of tert-butyl 3-chloro-2-(dimethylcarbamoyl)-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-ln).
- Step 9 Synthesis if 3-chloro-N,N-dimethyl-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a][l,4]diazepine-2-carboxamide (Intermediate 1).
- Step 1 Synthesis of tert-butyl 2-amino-3-cyano-spiro[5,6- dihy drocy cl openta[b]thiophene-4, 3 '-azetidine]- l'-carboxylate (Int-2a).
- Step 2 Synthesis of 2-aminospiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]- 3 -carbonitrile (Intermediate 2).
- Step 1 Preparation of tert-butyl 2-[methoxy(methyl)carbamoyl]-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-5b).
- Step 2 Preparation of tert-butyl 3-chloro-2-[methoxy(methyl)carbamoyl]-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-5c).
- Step 3 Preparation of tert-butyl 2-acetyl-3-chloro-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-5-carboxylate (Int-5d).
- Step 4 Preparation of l-(3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a][l,4]diazepin-2-yl)ethenone (Intermediate 5).
- tert-butyl 2-acetyl-3-chloro-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate Int-5d, 140 mg, 0.45 mmol
- HC1 in Dioxane 56 mL, 224 mmol
- Step 1 Preparation of tert-butyl 7-methyl-5-oxo-2-azaspiro[3.4]octane-2-carboxylate (Int-6b) To a solution of THF (4 mL) was added 1.6M butyllithium in hexane (4.12 mL, 6.59 mmol) at -68 °C. The mixture was stirred at -68 °C for 10 min.
- Step 2 Preparation of tert-butyl 2-amino-3-cyano-6-methyl-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3 '-azetidine]- 1 '-carboxylate (Int-6c).
- Step 3 Preparation of 2-amino-6-methyl-spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3 '-azetidine]-3 -carbonitrile (Intermediate 6).
- Step 1 Preparation of 3-[(E)-(3-methylsulfanyl-lH-pyrazol-5- yl)methyleneamino]propan-l-ol (Int-8a).
- 3-methylsulfanyl-lH-pyrazole-5- carbaldehyde Bioorganic and Medicinal Chemistry, 2012, vol. 20, # 3, p. 1319 1336, 1.1 g, 7.74 mmol
- 3 -aminopropan- l-ol (639.21 mg, 8.51 mmol) in Ethanol (10 mL) was stirred at 80 °C for 16 h. The mixture was concentrated to afford a crude product which was used directly in the next step.
- Step 2 Preparation of 3-[(3-methylsulfanyl-lH-pyrazol-5-yl)methylamino]propan-l- ol (Int-8b)
- To a solution of 3-[(E)-(3-methylsulfanyl-lH-pyrazol-5-yl)methyleneamino]propan- l-ol (Int-8a crude, 1.7 g, 8.53 mmol) in Methanol (30mL) was added Sodium triacetoxyborohydride (3.62 g, 17.06 mmol) at 0 °C. The mixture was stirred at 25 °C for 6 h. The mixture was concentrated directly to lOmL.
- Step 3 Preparation of tert-butyl N-(3-hydroxypropyl)-N-[(3-methylsulfanyl-lH- pyrazol-5-yl)methyl]carbamate (Int-8c).
- Step 4 Preparation of 3-[tert-butoxycarbonyl-[(3-methylsulfanyl-lH-pyrazol-5- yl)methyl]amino]propyl methanesulfonate (Int-8d).
- tert-butyl N-(3- hydroxypropyl)-N-[(3-methylsulfanyl-lH-pyrazol-5-yl)methyl]carbamate (Int-8c, 50 mg, 0.17 mmol) and in DCM (2 mL) was added MsCl (28.5 mg, 0.25 mmol) at 0 °C. The mixture was stirred at 25 °C for 2 h.
- Step 5 Preparation of tert-butyl 2-methylsulfanyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-5-carboxylate (Int-8e).
- Step 6 Preparation of 2-methylsulfanyl-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a][l,4]diazepine (Intermediate 8).
- tert-butyl 2-methylsulfanyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate Int-8e, 120 mg, 0.42 mmol
- DCM 4 mL
- TFA 0.4 mL, 5.23 mmol
- Step 1 Preparation of tert-butyl 6-bromo-5-oxo-2-azaspiro[3.4]octane-2-carboxylate (Int-9a). To the solution of tert-butyl 5-oxo-2-azaspiro[3.4]octane-2-carboxylate (200 mg, 0.89 mmol) in Ether (5 mL) was added 5, 5-dibromohexahydropyrimidine-2, 4, 6-trione (507.6 mg, 1.78 mmol), and the mixture was stirred at 35 °C for 48h.
- Step 2 Preparation of tert-butyl 2-aminospiro[5,6-dihydrocyclopenta[d]thiazole-4,3'- azetidine]-l'-carboxylate (Int-9b).
- tert-butyl 6-bromo-5-oxo-2- azaspiro[3.4]octane-2-carboxylate Int-9a 140 mg, 0.46 mmol
- Ethanol 2 mL
- Thiourea 35.03 mg, 0.46 mmol
- Step 3 Preparation of spiro[5,6-dihydrocyclopenta[d]thiazole-4,3'-azetidine]-2-amine (Intermediate 9).
- the solution of tert-butyl 2-aminospiro[5,6-dihydrocyclopenta[d]thiazole-4,3'- azetidine]-l'-carboxylate (Int-9b, 15 mg, 0.05 mmol) and Trifluoroacetic acid (0.04 mL, 0.53 mmol) in DCM (2 mL) was stirred at 25 °C for Ih.
- Step 2 Synthesis of tert-butyl 2-cyano-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-5-carboxylate (Intermediate 10).
- tert-butyl 2-carbamoyl- 4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate Int-lOa, 300 mg, 1.07 mmol
- Pyridine (0.26 mL, 3.21 mmol
- TFAA 674.36 mg, 3.21 mmol
- Step 1 Synthesis of [5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepin-2-yl]methanol (Int-l la).
- Step 2 Synthesis of 5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carbaldehyde (Int-llb).
- Step 3 Synthesis of 2-(l-methylbenzimidazol-2-yl)-5-(2-nitrophenyl)sulfonyl- 4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine (Int-llc) .
- Step 4 Synthesis of 2-(l-methylbenzimidazol-2-yl)-5,6,7,8-tetrahydro-4H- pyrazolo[l,5-a][l,4]diazepine (Intermediate 11) .
- Step 1 Synthesis of l-(2',2'-difluorospiro[azetidine-3,l'-tetralin]-l-yl)-2,2,2-trifluoro- ethanone (Int-12b). l-(2,2,2-trifluoroacetyl)spiro[azetidine-3,l'-tetralin]-2'-one (Int-12a ref. Angew. Chem. Int. Ed. 2021, 60, 7360-7365, 120 mg, 0.42 mmol) was weighed in a vial and DCM (1.69 mL) was added.
- Step 2 Synthesis of 2',2'-difluorospiro[azetidine-3,r-tetralin].
- Intermediate 12 1- (2',2'-difluorospiro[azetidine-3,r-tetralin]-l-yl)-2,2,2-trifluoro-ethanone (Int-12b, 20 mg, 0.07 mmol) was weighed in a vial and Methanol (0.65 mL) was added. Potassium Carbonate (13.58 mg, 0.1 mmol) was added, and reaction was heated to reflux for 1 hour. Reaction was then evaporated and redissolved in 10% MeOH/DCM.
- Step 1 Synthesis of 5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxamide (Int-14a).
- Step 2 Synthesis of 5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carbonitrile (Int-14b) .
- Step 4 Synthesis of 3-chloro-N-hydroxy-5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamidine (Int-14d).
- Step 5 Synthesis of 3-[3-chloro-5-(2-nitrophenyl)sulfonyl-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-2-yl]-5-methyl-l,2,4-oxadiazole (Int-14e).
- Step 6 Synthesis of 3-(3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin- 2-yl)-5-methyl-l,2,4-oxadiazole.
- Step 1 Synthesis of ethyl 3-chloro-5-(2-nitrophenyl)sulfonyl-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int-17a).
- Step 2 Synthesis of 3-chloro-5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (Int-17b).
- Step 1 Synthesis of tert-butyl 3-chloro-2-[methoxy(methyl)carbamoyl]-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate.
- Step 2 Synthesis of tert-butyl 3-chloro-2-(cyclopropanecarbonyl)-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate.
- Step 3 Synthesis of (3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin-2- yl)-cyclopropyl-methanone.
- Step 1 Synthesis of 3-(l-azabicyclo[1.1.0]butan-3-yl)thietan-3-ol (Int-17a). 2,3- dibromopropan-l-amine;hydrobromide (5000 mg, 16.79 mmol) in THF (lOOmL) at -78 °C, Phenyllithium (50.37 mL, 50.37 mmol) was added, The mixture was stirred at -78 °C for 2h, After this time, the reaction mixture was removed from the cooling bath and warmed to room temperature over 10 minutes with stirring.
- Step 2 Synthesis of 2-(2,2,2-trifluoroacetyl)-6-thia-2-azaspiro[3.4]octan-8-one (Int- 17b). 3-(l-azabicyclo[1.1.0]butan-3-yl)thietan-3-ol (Int-17a, 3000 mg, 20.95 mmol) in DCM at -78 °C, TFAA (2.91 mL, 20.95 mmol) was added. The mixture was stirred at -78 °C for 2h. The mixture was quenched with NaEtCCL (100 mL), extracted with DCM (100 mLx2), dried over Na2SO4, concentrated.
- Step 3 Synthesis of 5-amino-l'-(2,2,2-trifluoroacetyl)spiro[2H-thieno[2,3- b]thiophene-3,3'-azetidine]-4-carbonitrile (Int-17c).
- 2-(2,2,2-trifluoroacetyl)-6- thia-2-azaspiro[3.4]octan-8-one Int-17b, 500 mg, 2.09 mmol
- L-Proline 240.65 mg, 2.09 mmol
- propanedinitrile 207.12 mg, 3.14 mmol
- Sulfur 100.52 mg, 3.14 mmol
- Step 4 Synthesis of tert-butyl N-tert-butoxy carbonyl-N-[4-cyano-l '-(2,2,2- trifluoroacetyl)spiro[2H-thieno[2,3-b]thiophene-3,3'-azetidine]-5-yl]carbamate (Int-17d).
- Step 5 Synthesis of tert-butyl N-(4-cyanospiro[2H-thieno[2,3-b]thiophene-3,3'- azetidine]-5-yl)carbamate (Intermediate 17).
- tert-butyl N-[4-cyano-l'-(2,2,2- trifluoroacetyl)spiro[2H-thieno[2,3-b]thiophene-3,3'-azetidine]-5-yl]carbamate Int-17d, 300 mg, 0.58 mmol) in Methanol (3mL) was added K2CO3 (239.41 mg, 1.73 mmol) at 25 °C.
Landscapes
- Health & Medical Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- General Health & Medical Sciences (AREA)
- Medicinal Chemistry (AREA)
- Organic Chemistry (AREA)
- Pharmacology & Pharmacy (AREA)
- Immunology (AREA)
- Veterinary Medicine (AREA)
- Public Health (AREA)
- Animal Behavior & Ethology (AREA)
- Epidemiology (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Molecular Biology (AREA)
- Genetics & Genomics (AREA)
- Biochemistry (AREA)
- Chemical Kinetics & Catalysis (AREA)
- General Chemical & Material Sciences (AREA)
- Nuclear Medicine, Radiotherapy & Molecular Imaging (AREA)
- Pharmaceuticals Containing Other Organic And Inorganic Compounds (AREA)
Abstract
Provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor and a compound of Formula (I).
Description
COMBINATION THERAPIES WITH A KRAS MODULATOR AND AN IMMUNOMODULATOR INHIBITOR
CROSS-REFERENCE
[0001] This application claims the benefit of U.S. Provisional Patent Application No. 63/600,221 filed on November 17, 2023, the entire content of which is incorporated herein by reference.
BACKGROUND OF THE INVENTION
[0002] The small GTPase protein Kirsten Rat Sarcoma 2 Viral Oncogene Homolog (KRAS) is a member of the Ras family of cell signaling switches, regulating growth and survival of normal and cancerous cells (e.g., see Cully, M. and J. Downward, Snapshot: Ras Signaling. Cell, 2008. 133(7): p. 1292-1292 el). KRAS mutations drive approximately 25% of human cancers by aberrant regulation of the mitogen-activated protein kinase (MAPK) signaling cascade and other effector pathways (e.g., see Stephen, A.G., et al., Dragging ras back in the ring. Cancer Cell, 2014. 25(3): p. 272-81). Though Ras has been recognized as a target in cancer for about 40 years, Ras- driven cancers remain among the most difficult to treat due to insensitivity to available targeted therapies. Ras, encoded by the three major genes KRAS, NRAS and HRAS, has the highest frequency of mutation of any oncogene. All oncogenic Ras mutations drive the switch to accumulate in the active GTP -bound state. The most common Ras mutation found across human tumor types is KRAS G12D (e.g., see The AACR Project GENIE Consortium. Cancer Discovery, 2017. 7(8): p. 818-831. Dataset Version 4). Activating mutations in codon 12 impair the small GTPases’ ability to perform their role in hydrolyzing GTP. This regulatory impairment is fundamental for initiating and maintaining tumor progression.
[0003] The small GTPase protein Kirsten Rat Sarcoma 2 Viral Oncogene Homolog (KRAS) is a member of the Ras family of cell signaling switches, regulating growth and survival of normal and cancerous cells (e.g., see Cully, M. and J. Downward, Snapshot: Ras Signaling. Cell, 2008. 133(7): p. 1292-1292 el). KRAS mutations drive approximately 25% of human cancers by aberrant regulation of the mitogen-activated protein kinase (MAPK) signaling cascade and other effector pathways (e.g., see Stephen, A.G., et al., Dragging ras back in the ring. Cancer Cell, 2014. 25(3): p. 272-81). Though Ras has been recognized as a target in cancer for about 40 years, Ras-driven cancers remain among the most difficult to treat due to insensitivity to available targeted therapies. Ras, encoded by the three major genes KRAS, NRAS and HRAS, has the highest frequency of mutation of any oncogene. All oncogenic Ras mutations drive the switch to accumulate in the active GTP -bound state. The most common Ras mutation found across human tumor types is KRAS G12D (e.g., see The AACR Project GENIE Consortium.
Cancer Discovery, 2017. 7(8): p. 818-831. Dataset Version 4). Activating mutations in codon 12 impair the small GTPases’ ability to perform their role in hydrolyzing GTP. This regulatory impairment is fundamental for initiating and maintaining tumor progression.
[0004] Despite extensive efforts, small molecules have not been identified which block effector binding or restore GTPase activating protein (GAP) sensitivity, though some have been found which block interaction of Ras with the guanine nucleotide exchange factor (GEF), SOS, which activates Ras at the plasma membrane. KRAS G12C mutations, most common in lung adenocarcinoma, have been clinically shown to be susceptible to direct inhibition by covalent modification with small molecule inhibitors trapping the protein in the inactive GDP -bound state. KRAS G12D mutation confers a significantly slower intrinsic rate of GTP hydrolysis than G12C, resulting in more constitutive activation. Thus, pharmacological targeting the of inactive state is unlikely to achieve similar results against G12D, despite the existence of a similar binding pocket in the GDP-state. Additionally, a cysteine present at the site of the activating mutation yields itself to covalent chemistry, while aspartic acid does not provide typical medicinal chemistry approaches for selective covalent modification.
[0005] In order to potentially exploit the accumulation of KRAS G12D and other mutant variants in the GTP -bound state as a vulnerability to achieve selective inhibition of cancer cells while sparing normal Ras function, it is attractive for small molecule inhibitors to bind to the GTP- state and stabilize a conformation that is incompetent for oncogenic signaling interactions with effector proteins. Furthermore, it has been shown that only constitutive activation of Raf, MEK and ERK kinases in the MAPK cascade downstream of Ras can bypass the requirement for Ras proteins in proliferative signaling (e.g., see Drosten, M., et al., Genetic analysis of Ras signalling pathways in cell proliferation, migration and survival. EMBO J, 2010. 29(6): p. 1091-104). As all evidence has indicated that MAPK signaling is essential for the growth effects of Ras in cancer, KRAS-mutant-selective inhibition in this pathway is considered the critical functional readout for potential clinical benefit of novel therapeutic approaches.
SUMMARY OF THE INVENTION
[0006] In some embodiments, oncogenic KRas mutations create an immunosuppressive microenvironment which can result in resistance to immune checkpoint blockade (ICB) therapy, including anti-PD-1 and anti-PD-Ll inhibitors. Activated KRas has been demonstrated to repress the expression of interferon regulatory factor 2 (IRF2), which directly represses CXCL3 expression. This KRas-mediated repression of IRF2 leads to increased expression of CXCL3, which binds to CXCR2 on myeloid-derived suppressor cells (MDSC) promoting migration of these cells to the tumor microenvironment. In some cases, a role for KRas in modulating the
immune microenvironment and primary ICB resistance in advanced colorectal cancer has been established. In colorectal cancer, anti-PD-1 resistance of KRAS-expressing tumors can be overcome by enforced IRF2 expression or by inhibition of CXCR2. (e.g., see Liao et al., (2019) Cancer Cell 35:559 - 572). In addition, oncogenic KRas signaling has been shown to promote tumor immunoresistance to ICB therapy by stabilizing PD-L1 mRNA via repression of the AU- rich element-binding protein tristetraprolin (TTP), which negatively regulates PD-L1 expression through AU-rich elements in the 3' UTR of PD-L1 mRNA (e.g., see Coelho et al., (2017) Immunity 47(6): 1083-1099). In some cases, oncogenic KRas has also been demonstrated to impair antigen presentation by repressing MHC I expression thereby allowing tumor cells to evade cytotoxic T-lymphocytes (e.g., see El-Jawhari et al., (2014) Molecular Immunology 58(2): 160-168), and KRas activating mutations upregulate IL-8 expression in NSCLC, and IL-8 plays a role in cell growth and migration in KRas-associated NSCLC (e.g., see Sunaga et al., (2012) Int. J. Cancer 130(8): 1733-1744).
[0007] Thus, activated KRas mutations or other KRas-activating generic alterations expression may modulate many aspects of the immune system and can be responsible for the immunosuppressive tumor microenvironment in KRas mutant or hyperactivated Kras wildtype- associated tumors. As such, the direct inhibition of KRas mutant or hyperactivated Kras wildtype-associated-mediated cell activity may reverse this reported immunosuppressive tumor microenvironment thereby improving the clinical activity of immune checkpoint blockade therapy, including the PD-1/PD-L1 pathway.
[0008] As so, there is a need to develop combination therapies using KRas mutant or Kras wildtype inhibitors and ICB therapy, including anti-PD-1 and anti-PD-Ll inhibitors, for treating KRas mutant or hyperactivated Kras wildtype-associated cancers that can be resistant to ICB therapy.
[0009] Additionally, the compounds described herein direct potency against tumor cells, compounds described herein may modulate the tumor microenvironment (TME) in favor of antitumor immunity, by modulating tumor cell cytokine/chemokine release.
[0010] Additionally, the compounds described herein direct potency against tumor cells, compounds described herein may reprogram the tumor microenvironment (TME) in favor of antitumor immunity, by modulating tumor cell cytokine/chemokine release. Specifically, treatment with compounds described herein reduces immunosuppressive cytokine release while increasing immunostimulatory chemokine release in KRAS mutant cells. Results of our study herein (e.g., example 14) reveal a direct mechanism by which compounds described herein can modify tumor-cell-intrinsic immune signals to enhance antitumor immunity in a synergistic manner with the use of common immune checkpoint inhibitors. Our preclinical data provides
strong rationale for clinical studies combining compounds described herein (e.g., compounds of Formula (I)) with, and expanding the therapeutic use of, existing checkpoint inhibitors (inhibitors targeting: cytotoxic T lymphocyte associated antigen 4 (CTLA-4), e.g. Yervoy/lpilimumab; programmed death 1/programmed death-ligand 1 (PD-1/PD-L1), e.g. Keytruda/pembrolizumab; and lymphocyte-activation gene 3 (LAG-3), e.g. BMS- 986016/relatlimab) for various types of cancers harboring oncogenic KRAS.
[0011] In an aspect, the present disclosure provides combination therapies useful for treating a disease or disorder (e.g., cancer). In some cases, provided are therapeutically effective combinations of an agent that disrupts Programmed cell death protein 1 (PD-1) and Programmed death-ligand 1 (PD-L1) axis signaling (e.g., a PD-1 inhibitor, a PD-Ll inhibitor) and a compound of Formula (I), kits comprising the compositions and methods of use thereof. In an aspect, the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor and a compound represented by the structure of Formula (I):
Formula (I), or a pharmaceutically acceptable salt thereof wherein:
B is selected from a 7- to 15-membered heterocycle and C7-C15 carbocycle, wherein the 7- to 15-membered heterocycle and C7-C15 carbocycle are each optionally substituted with one or more substituents independently selected from halogen, -CN, -NO2, =0, -N(R21)2, -B(OR21)2, - OR21, -SR21, -S(O)2(R21), -S(O)2N(R21)2, -NR21S(O)2R21, -C(O)N(R21)2, -C(O)NR21OR21, - N(R21)C(O)R21, -N(R21)C(O)N(R21)2, -N(R21)C(O)OR21, -C(0)R21, C(0)0R21, -0C(0)R21, - OC(O)N(R21)2, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-C12 carbocycle and 5- to 12-membered heterocycle;
R1 is selected from hydrogen and 5- to 15-membered heterocycle, wherein the 5- to 15- membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(=NR20)N(R20)2, -C1-6 alkyl(=NOR20), - C(O)NR20OR20, -N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, -C(O)R20, - C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, -N02, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl- N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, C 1-6 haloalkyl, C1-6 alkyl-
SO2R20, Ci-6 alkoxyalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-C 12 carbocycle and 5- to 12- membered heterocycle, wherein the C3-C12 carbocycle and 5- to 12-membered heterocycle are each optionally substituted independently with one or more R1*; each R1* is independently selected from halogen, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(O)NR20OR20, - N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, -C(O)R20, -C(O)OR20, - OC(O)R20, -OC(O)N(R20)2, -NO2, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and C3-C 12 carbocycle;
Y is selected from a bond, -0-, -S-, and -N(R5)-;
R2is selected from heterocycle, aryl, Ci-Ce alkyl, -L-heterocycle, -L-N(R23)2, -L-OR23, -L- aryl, -L-heteroaryl, -L-cycloalkyl, -L-NHC(=NH)NH2, -L-C(O)N(R23)2, -L-Ci-Ce haloalkyl, -L- OR23, -L-NR23C(O)-aryl, -L-C00H, -L-NR23S(O)2(R23), -L-S(O)2N(R23)2, -L-N(R23)C(O)(OR23), -L-OC(O)N(R23)2, and -L-C(=O)OCi-Ce alkyl, wherein the heterocycle, the heterocycle portion of -L-heterocycle, and the cycloalkyl portion of the -L-cycloalkyl are each optionally substituted with one or more R6, and wherein the aryl, aryl portion of -L-NR23C(O)-aryl, the aryl portion of -L- NR23C(O)-aryl, the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R7;
R3 is selected from hydrogen, halogen, -CN, -NO2, -N(R20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -N(R20)C(O)R20, - N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, -C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, CI-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-C12 carbocycle and 5- to 12-membered heterocycle; each R4 is independently selected from halogen, -NO2, =0, =S, -CN, Ci-6 alkyl, C2-6 alkynyl, C 1-6 alkyl -N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, C 1-6 cyanoalkyl, Ci-6 hydroxyalkyl, and Ci-6 haloalkyl; n is selected from 0, 1, 2, 3, and 4; each R5 is independently selected from hydrogen and Ci-Ce alkyl; each R6 is independently selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, oxo, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, =CH2, NMO-C1-C3 alkyl, C1-C3 aminoalkyl, - N(R5)S(O)2(R5), -Q-phenyl, -Q-phenylSCLF, -NHC(O)phenyl, - NHC(O)phenylSO2F, C1-C3 alkyl substituted pyrazolyl, -C1-C3 alkyl-N(R5)2, -C(O)N(R5)2, tert-butyldimethylsilyloxyCFB-, -N(R5)2, (C1-C3 alkoxy)Ci-C3 alkyl-, (C1-C3 alkyl)C(=O), oxo, (C1-C3 haloalkyl)C(=O)-, -SO2F, (C1-C3 alkoxy)Ci-C3 alkoxy, -CH2OC(O)N(R5)2, -CH2NHC(O)OCI-C6 alkyl, -CH2NHC(O)N(R5)2, -
CH2NHC(0)Ci-Ce alkyl, -CH2(pyrazolyl), -CH2NHSO2C1-C6 alkyl, -CH2OC(O)heterocycle, - OC(O)N(R5)2, -OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl), -OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl)phenyl(Ci-C3 alkyl)N(CH3)2, -OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl)phenyl, - OC(O)heterocycle, -O-Ci-C3 alkyl, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), - NR20S(O)2R20, and -CHJieterocycle, wherein the phenyl of -NHC(O)phenyl and -OC(O)NH(Ci- C3 alkyl)(Ci-C3 alkyl)phenyl are optionally substituted with one or more substituents selected from -C(O)H and OH, and wherein the alkyl of -O-Ci-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo and hydroxy; and wherein the heterocycle of -CH2heterocyclyl is optionally substituted with oxo; each Q is selected from a bond, S, and O; each R7 is independently selected from halogen, hydroxy, HC(=O)-, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 hydroxyalkyl, -Ci-C3 alkyl-N(R5)2, -C(O)N(R5)2, and -N(R5)2; each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen, hydroxy, C1-6 alkoxy, C1-C4 hydroxyalkyl, C1-C4 alkyl, C3-Ce carbocycle, and 3- to 8-membered heterocycle, wherein the C3-Ce carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -NO2, =0, =S, -CN, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, Ci-ehydroxyalkyl, C1-6 haloalkyl; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-Ce carbocycle or 3- to 8-membered heterocycle, wherein the C3-Ce carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -NO2, =0, =S, -CN, Ci-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and C1-6 haloalkyl; each R20 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-i2 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-i2 carbocycle, and 3- to 12-membered heterocycle; each R21 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-i2 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-i2 carbocycle, and 3- to 12-membered heterocycle; and each R23 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-i2 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2,
-NH2, -N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle.
[0012] In an aspect, the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor and a compound represented by the structure of
R1 is selected from 8- to 10-membered heterocycle, wherein the 8- to 10-membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -OR20, -S(O)2(R20), -C(O)N(R20)2, -Ci-6 alkyl(=NOR20), -C(O)R20, =0, -CN, -NHCN, C1-6 alkyl-N(R20)2, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, C1-6 alkyl-SO2R20, C1-6 alkoxyalkyl, C1-6 alkyl, C 2-6 alkynyl, and 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle are each optionally substituted independently with one or more R1*; each R1* is independently selected from halogen, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(O)NR20OR20, - N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, -C(O)R20, -C(O)OR20, - OC(O)R20, -OC(O)N(R20)2, -NO2, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl-N(R20)2, Ci- 6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, C 1-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and C3-C12 carbocycle;
B is selected from a 7- to 15-membered heterocycle and C7-C15 carbocycle, wherein the 7- to 15-membered heterocycle and C7-C15 carbocycle are each optionally substituted with one or more substituents independently selected from halogen, -CN, -NO2, =0, -N(R21)2, -B(OR21)2, - OR21, -SR21, -S(O)2(R21), -S(O)2N(R21)2, -NR21S(O)2R21, -C(O)N(R21)2, -C(O)NR21OR21, - N(R21)C(O)R21, -N(R21)C(O)N(R21)2, -N(R21)C(O)OR21, -C(0)R21, C(0)0R21, -0C(0)R21, - OC(O)N(R21)2, CI-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, C 1-6 cyanoalkyl, C 1-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-C12 carbocycle and 5- to 12-membered heterocycle;
Y is selected from a bond, and -O-;
R2 is selected from heterocycle, aryl, Ci-Ce alkyl, -L-heterocycle, -L-N(R23)2, -L-OR23, - L-aryl, -L-heteroaryl, -L-cycloalkyl, -L-NHC(=NH)NH2, -L-C(O)N(R23)2, -L-Ci-Ce haloalkyl, -
L-OR23, -L-NR23C(O)-aryl, -L-COOH, -L-NR23S(O)2(R23), -L-S(O)2N(R23)2, -L- N(R23)C(O)(OR23), -L-OC(O)N(R23)2, and -L-C(=O)OCi-Ce alkyl, wherein the heterocycle, the heterocycle portion of -L-heterocycle, and the cycloalkyl portion of the -L-cycloalkyl are each optionally substituted with one or more R6, and wherein the aryl, aryl portion of -L-NR23C(O)- aryl, the aryl portion of -L-NR23C(O)-aryl, the aryl of the -L-aryl, and the heteroaryl of -L- heteroaryl are each optionally substituted with one or more R7;
R3 is selected from hydrogen, halogen, -CN, -NO2, -N(R20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -N(R20)C(O)R20, - N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, -C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, CI-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-e alkynyl, C3-Ci2 carbocycle and 5- to 12-membered heterocycle; each R4 is independently selected from halogen, -NO2, =0, =S, -CN, Ci-6 alkyl, C2-6 alkynyl, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 cyanoalkyl, Ci-e hydroxyalkyl, and Ci-6 haloalkyl; n is selected from 0, 1, 2, 3, and 4; each R5 is independently selected from hydrogen and Ci-Ce alkyl; each R6 is independently selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, oxo, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, =CH2, NMO-C1-C3 alkyl, C1-C3 aminoalkyl, - N(R5)S(O)2(R5), -Q-phenyl, -Q-phenylSO2F, -NHC(O)phenyl, -NHC(O)phenylSO2F, C1-C3 alkyl substituted pyrazolyl, -C1-C3 alkyl-N(R5)2, -C(O)N(R5)2, tert-butyldimethylsilyloxyCH2-, -N(R5)2, (C1-C3 alkoxy)Ci-C3 alkyl-, (C1-C3 alkyl)C(=O), oxo, (C1-C3 haloalkyl)C(=O)-, -SO2F, (C1-C3 alkoxy)Ci-C3 alkoxy, -CH2OC(O)N(R5)2, -CH2NHC(O)OCI-C6 alkyl, -CH2NHC(O)N(R5)2, - CH2NHC(O)Ci-Ce alkyl, -CH2(pyrazolyl), -CH2NHSO2Ci-Ce alkyl, -CH2OC(O)heterocycle, - OC(O)N(R5)2, -0C(0)NH(CI-C3 alkyl)O(Ci-C3 alkyl), -0C(0)NH(Ci-C3 alkyl)O(Ci-C3 alkyl)phenyl(Ci-C3 alkyl)N(CH3)2, -0C(0)NH(Ci-C3 alkyl)O(Ci-C3 alkyl)phenyl, - OC(O)heterocycle, -O-C1-C3 alkyl, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), - NR20S(O)2R20, and -CH2heterocycle, wherein the phenyl of -NHC(O)phenyl and -0C(0)NH(Ci- C3 alkyl)(Ci-C3 alkyl)phenyl are optionally substituted with one or more substituents selected from -C(0)H and OH, and wherein the alkyl of -O-C1-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo and hydroxy; and wherein the heterocycle of -CH2heterocyclyl is optionally substituted with oxo; each Q is selected from a bond, S, and O; each R7 is independently selected from halogen, hydroxy, HC(=0)-, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 hydroxyalkyl, -C1-C3 alkyl-N(R5)2, -C(O)N(R5)2, and -N(R5)2;
each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen, hydroxy, C1-6 alkoxy, C1-C4 hydroxyalkyl, C1-C4 alkyl, C3-C6 carbocycle, and 3- to 8-membered heterocycle, wherein the C3- Ce carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -NO2, =0, =S, -CN, C1-6 alkyl- N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle, wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, - OH, -NO2, =0, =S, -CN, Ci-6 alkyl-N(R20)2, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and C1-6 haloalkyl; each R20 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle; each R21 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle; each R23 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle.
[0013] In an aspect, the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor and a compound represented by the structure of
Formula
Formula (I-B), or a pharmaceutically acceptable salt thereof wherein:
optionally substituted with one or more substituents independently selected from halogen, - S(O)2(R20), -C(O)N(R20)2, -CI-6 alkyl(=NOR20), -C(O)R20, =0, and 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle are each optionally substituted independently with one or more R1*; each R1* is independently selected from halogen, -OR20, -N(R20)2, -N02, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, and Ci-6 alkyl;
Y is -0-;
R2 is selected from -L-heterocycle, and -L-N(R23)2, wherein the heterocycle portion of -L- heterocycle is optionally substituted with one or more R6.
B is selected from a 7- to 15-membered heterocycle, wherein the 7- to 15-membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -CN, =0, -N(R21)2, -OR21, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, and C2-e alkynyl;
R3 is selected from hydrogen, halogen, -CN, -N(R20)2, -OR20, -C(O)R20, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, and Ci-6 alkyl; and each R20 is independently selected from hydrogen; and Ci-6 alkyl, and C3-12 carbocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NH2, -N(CI-6 alkyl)2, Cnio alkyl, -Ci-io haloalkyl, -O-Ci-io alkyl, and oxo.
[0014] A method of treating a cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, or a pharmaceutical composition thereof; and a compound selected
pharmaceutically acceptable salt of any one thereof, or a pharmaceutical composition of any one thereof.
INCORPORATION BY REFERENCE
[0015] All publications, patents, and patent applications mentioned in this specification are herein incorporated by reference to the same extent as if each individual publication, patent, or patent application was specifically and individually indicated to be incorporated by reference.
BRIEF DESCRIPTION OF THE DRAWINGS
[0016] The novel features of the invention are set forth with particularity in the appended claims. A better understanding of the features and advantages of the present invention will be obtained by reference to the following detailed description that sets forth illustrative embodiments, in which the principles of the invention are utilized, and the accompanying drawings (also “figure” and “FIG.” herein), of which:
[0017] FIG. 1 illustrates that compounds herein reduce immunosuppressive cytokine release while increasing immunostimulatory chemokine release in KRAS-mutant HP AC cells.
DETAILED DESCRIPTION OF THE INVENTION
[0018] The following description sets forth numerous exemplary configurations, methods, parameters, and the like. It should be recognized, however, that such description is not intended as a limitation on the scope of the present disclosure, but is instead provided as a description of exemplary embodiments.
[0019] In the following description, certain specific details are set forth in order to provide a thorough understanding of various embodiments of the disclosure. However, one skilled in the art will understand that the disclosure may be practiced without these details.
Definitions
[0020] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as is commonly understood by one of skill in the art to which this invention belongs. All patents and publications referred to herein are incorporated by reference.
[0021] "Alkyl" refers to a straight or branched hydrocarbon chain radical consisting solely of carbon and hydrogen atoms, containing no unsaturation, and preferably having from one to
fifteen carbon atoms (z.e., C1-C15 alkyl). In certain embodiments, an alkyl comprises one to thirteen carbon atoms (z.e., C1-C13 alkyl). In certain embodiments, an alkyl comprises one to eight carbon atoms (z.e., Ci-Cs alkyl). In other embodiments, an alkyl comprises one to five carbon atoms (z.e., C1-C5 alkyl). In other embodiments, an alkyl comprises one to four carbon atoms (z.e., C1-C4 alkyl). In other embodiments, an alkyl comprises one to three carbon atoms (z.e., C1-C3 alkyl). In other embodiments, an alkyl comprises one to two carbon atoms (z.e., C1-C2 alkyl). In other embodiments, an alkyl comprises one carbon atom (z.e., Ci alkyl). In other embodiments, an alkyl comprises five to fifteen carbon atoms (z.e., C5-C15 alkyl). In other embodiments, an alkyl comprises five to eight carbon atoms (z.e., Cs-Cs alkyl). In other embodiments, an alkyl comprises two to five carbon atoms (z.e., C2-C5 alkyl). In other embodiments, an alkyl comprises three to five carbon atoms (z.e., C3-C5 alkyl). In certain embodiments, the alkyl group is selected from methyl, ethyl, 1 -propyl (zz-propyl), 1 -methylethyl (z.w-propyl), 1 -butyl (zz-butyl), 1 -methylpropyl (sec-butyl), 2-methylpropyl (z.w-butyl), 1,1 -dimethylethyl (tert-butyl), 1 -pentyl (zz-pentyl). The alkyl is attached to the rest of the molecule by a single bond.
[0022] The term “Cx-y” when used in conjunction with a chemical moiety, such as alkyl, alkenyl, or alkynyl is meant to include groups that contain from x to y carbons in the chain. For example, the term “Ci-ealkyl” refers to substituted or unsubstituted saturated hydrocarbon groups, including straight-chain alkyl and branched-chain alkyl groups that contain from 1 to 6 carbons. The term -Cx.yalkylene- refers to a substituted or unsubstituted alkylene chain with from x to y carbons in the alkylene chain. For example -Ci-ealkylene- may be selected from methylene, ethylene, propylene, butylene, pentylene, and hexylene, any one of which is optionally substituted.
[0023] "Alkoxy" refers to a radical bonded through an oxygen atom of the formula -O- alkyl, where alkyl is an alkyl chain as defined above.
[0024] "Alkenyl" refers to a straight or branched hydrocarbon chain radical group consisting solely of carbon and hydrogen atoms, containing at least one carbon-carbon double bond, and preferably having from two to twelve carbon atoms (i.e., C2-C12 alkenyl). In certain embodiments, an alkenyl comprises two to eight carbon atoms (i.e., C2-C8 alkenyl). In certain embodiments, an alkenyl comprises two to six carbon atoms (i.e., C2-C6 alkenyl). In other embodiments, an alkenyl comprises two to four carbon atoms (i.e., C2-C4 alkenyl). The alkenyl is attached to the rest of the molecule by a single bond, for example, ethenyl (i.e., vinyl), prop-l-enyl (i.e., allyl), but-l-enyl, pent-l-enyl, penta- 1,4-dienyl, and the like.
[0025] "Alkynyl" refers to a straight or branched hydrocarbon chain radical group consisting solely of carbon and hydrogen atoms, containing at least one carbon-carbon triple bond, and
preferably having from two to twelve carbon atoms (z.e., C2-C12 alkynyl). In certain embodiments, an alkynyl comprises two to eight carbon atoms (z.e., C2-C8 alkynyl). In other embodiments, an alkynyl comprises two to six carbon atoms (z.e., C2-C6 alkynyl). In other embodiments, an alkynyl comprises two to four carbon atoms (z.e., C2-C4 alkynyl). The alkynyl is attached to the rest of the molecule by a single bond, for example, ethynyl, propynyl, butynyl, pentynyl, hexynyl, and the like.
[0026] The terms “Cx-yalkenyl” and “Cx-yalkynyl” refer to substituted or unsubstituted unsaturated aliphatic groups analogous in length and possible substitution to the alkyls described above, but that contain at least one double or triple bond, respectively. The term -Cx.yalkenylene- refers to a substituted or unsubstituted alkenylene chain with from x to y carbons in the alkenylene chain. For example, -C2-ealkenylene- may be selected from ethenylene, propenylene, butenylene, pentenylene, and hexenylene, any one of which is optionally substituted. An alkenylene chain may have one double bond or more than one double bond in the alkenylene chain. The term -Cx.yalkynylene- refers to a substituted or unsubstituted alkynylene chain with from x to y carbons in the alkenylene chain. For example, -C2-ealkenylene- may be selected from ethynylene, propynylene, butynylene, pentynylene, and hexynylene, any one of which is optionally substituted. An alkynylene chain may have one triple bond or more than one triple bond in the alkynylene chain.
[0027] "Alkylene" or "alkylene chain" refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing no unsaturation, and preferably having from one to twelve carbon atoms, for example, methylene, ethylene, propylene, ^-butylene, and the like. The alkylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond. The points of attachment of the alkylene chain to the rest of the molecule and to the radical group may be through any two carbons within the chain. In certain embodiments, an alkylene comprises one to ten carbon atoms (i.e., Ci-Cs alkylene). In certain embodiments, an alkylene comprises one to eight carbon atoms (i.e., Ci-Cs alkylene). In other embodiments, an alkylene comprises one to five carbon atoms (i.e., C1-C5 alkylene). In other embodiments, an alkylene comprises one to four carbon atoms (i.e., C1-C4 alkylene). In other embodiments, an alkylene comprises one to three carbon atoms (i.e., C1-C3 alkylene). In other embodiments, an alkylene comprises one to two carbon atoms (i.e., C1-C2 alkylene). In other embodiments, an alkylene comprises one carbon atom (i.e., Ci alkylene). In other embodiments, an alkylene comprises five to eight carbon atoms (i.e., Cs-Cs alkylene). In other embodiments, an alkylene comprises two to five carbon atoms (i.e., C2-C5 alkylene). In other embodiments, an alkylene comprises three to five carbon atoms (i.e., C3-C5 alkylene).
[0028] "Alkenylene" or "alkenylene chain" refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing at least one carbon-carbon double bond, and preferably having from two to twelve carbon atoms. The alkenylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond. The points of attachment of the alkenylene chain to the rest of the molecule and to the radical group may be through any two carbons within the chain. In certain embodiments, an alkenylene comprises two to ten carbon atoms (i.e., C2-C10 alkenylene). In certain embodiments, an alkenylene comprises two to eight carbon atoms (i.e., C2-C8 alkenylene). In other embodiments, an alkenylene comprises two to five carbon atoms (i.e., C2-C5 alkenylene). In other embodiments, an alkenylene comprises two to four carbon atoms (i.e., C2-C4 alkenylene). In other embodiments, an alkenylene comprises two to three carbon atoms (i.e., C2-C3 alkenylene). In other embodiments, an alkenylene comprises two carbon atom (i.e., C2 alkenylene). In other embodiments, an alkenylene comprises five to eight carbon atoms (i.e., Cs-Cs alkenylene). In other embodiments, an alkenylene comprises three to five carbon atoms (i.e., C3-C5 alkenylene).
[0029] "Alkynylene" or "alkynylene chain" refers to a straight or branched divalent hydrocarbon chain linking the rest of the molecule to a radical group, consisting solely of carbon and hydrogen, containing at least one carbon-carbon triple bond, and preferably having from two to twelve carbon atoms. The alkynylene chain is attached to the rest of the molecule through a single bond and to the radical group through a single bond. The points of attachment of the alkynylene chain to the rest of the molecule and to the radical group may be through any two carbons within the chain. In certain embodiments, an alkynylene comprises two to ten carbon atoms (i.e., C2-C10 alkynylene). In certain embodiments, an alkynylene comprises two to eight carbon atoms (i.e., C2-C8 alkynylene). In other embodiments, an alkynylene comprises two to five carbon atoms (i.e., C2-C5 alkynylene). In other embodiments, an alkynylene comprises two to four carbon atoms (i.e., C2-C4 alkynylene). In other embodiments, an alkynylene comprises two to three carbon atoms (i.e., C2-C3 alkynylene). In other embodiments, an alkynylene comprises two carbon atom (i.e., C2 alkynylene). In other embodiments, an alkynylene comprises five to eight carbon atoms (i.e., C5-C8 alkynylene). In other embodiments, an alkynylene comprises three to five carbon atoms (i.e., C3-C5 alkynylene).
[0030] "Aryl" refers to a radical derived from an aromatic monocyclic or aromatic multicyclic hydrocarbon ring system by removing a hydrogen atom from a ring carbon atom. The aromatic monocyclic or aromatic multicyclic hydrocarbon ring system contains only hydrogen and carbon and from five to eighteen carbon atoms, where at least one of the rings in the ring system is aromatic, i.e., it contains a cyclic, delocalized (4n+2) ^-electron system in
accordance with the Hiickel theory. The ring system from which aryl groups are derived include, but are not limited to, groups such as benzene, fluorene, indane, indene, tetralin and naphthalene. [0031] "Aralkyl" refers to a radical of the formula -Rc-aryl where Rc is an alkylene chain as defined above, for example, methylene, ethylene, and the like.
[0032] "Aralkenyl" refers to a radical of the formula -Rd-aryl where Rd is an alkenylene chain as defined above. "Aralkynyl" refers to a radical of the formula -Re-aryl, where Re is an alkynylene chain as defined above.
[0033] “Carbocycle” refers to a saturated, unsaturated or aromatic rings in which each atom of the ring is carbon. Carbocycle may include 3- to 10-membered monocyclic rings, 6- to 12- membered bicyclic rings, and 6- to 12-membered bridged rings. Each ring of a bicyclic carbocycle may be selected from saturated, unsaturated, and aromatic rings. An aromatic ring, e.g., phenyl, may be fused to a saturated or unsaturated ring, e.g., cyclohexane, cyclopentane, or cyclohexene. Any combination of saturated, unsaturated and aromatic bicyclic rings, as valence permits, are included in the definition of carbocyclic. Exemplary carbocycles include cyclopentyl, cyclohexyl, cyclohexenyl, adamantyl, phenyl, indanyl, and naphthyl. Bicyclic carbocycles may be fused, bridged or spiro-ring systems. In some cases, spiro-ring carbocycles have at least two molecular rings with only one common atom.
[0034] The term “unsaturated carbocycle” refers to carbocycles with at least one degree of unsaturation and excluding aromatic carbocycles. Examples of unsaturated carbocycles include cyclohexadiene, cyclohexene, and cyclopentene.
[0035] "Cycloalkyl" refers to a fully saturated monocyclic or polycyclic hydrocarbon radical consisting solely of carbon and hydrogen atoms, which includes fused or bridged ring systems, and preferably having from three to twelve carbon atoms. In certain embodiments, a cycloalkyl comprises three to ten carbon atoms. In other embodiments, a cycloalkyl comprises five to seven carbon atoms. The cycloalkyl may be attached to the rest of the molecule by a single bond. Examples of monocyclic cycloalkyls include, e.g., cyclopropyl, cyclobutyl, cyclopentyl, cyclohexyl, cycloheptyl, and cyclooctyl. Polycyclic cycloalkyl radicals include, for example, adamantyl, norbornyl (i.e., bicyclo[2.2.1]heptanyl), norbomenyl, decalinyl, 7,7-dimethyl-bicyclo[2.2.1]heptanyl, and the like.
[0036] "Cycloalkenyl" refers to an unsaturated non-aromatic monocyclic or polycyclic hydrocarbon radical consisting solely of carbon and hydrogen atoms, which includes fused or bridged ring systems, preferably having from three to twelve carbon atoms and comprising at least one double bond. In certain embodiments, a cycloalkenyl comprises three to ten carbon atoms. In other embodiments, a cycloalkenyl comprises five to seven carbon atoms. The cycloalkenyl may be attached to the rest of the molecule by a single bond. Examples of
monocyclic cycloalkenyls includes, e.g., cyclopentenyl, cyclohexenyl, cycloheptenyl, and cyclooctenyl.
[0037] "Cycloalkylalkyl" refers to a radical of the formula -Rc-cycloalkyl where Rc is an alkylene chain as described above.
[0038] "Cycloalkylalkoxy" refers to a radical bonded through an oxygen atom of the formula -O-Rc-cycloalkyl where Rc is an alkylene chain as described above.
[0039] Halo" or "halogen" refers to halogen substituents such as bromo, chloro, fluoro and iodo substituents.
[0040] As used herein, the term "haloalkyl" or “haloalkane” refers to an alkyl radical, as defined above, that is substituted by one or more halogen radicals, for example, trifluoromethyl, di chloromethyl, bromomethyl, 2,2,2-trifluoroethyl, l-fluoromethyl-2-fluoroethyl, and the like. In some embodiments, the alkyl part of the fluoroalkyl radical is optionally further substituted. Examples of halogen substituted alkanes (“haloalkanes”) include halomethane (e.g., chloromethane, bromomethane, fluoromethane, iodomethane), di-and trihalomethane (e.g., tri chloromethane, tribromomethane, trifluoromethane, triiodomethane), 1-haloethane, 2- haloethane, 1,2-dihaloethane, 1-halopropane, 2-halopropane, 3-halopropane, 1,2-dihalopropane, 1,3-dihalopropane, 2,3-dihalopropane, 1,2,3-trihalopropane, and any other suitable combinations of alkanes (or substituted alkanes) and halogens (e.g., Cl, Br, F, I, etc.). When an alkyl group is substituted with more than one halogen radicals, each halogen may be independently selected e.g., l-chloro,2-fluoroethane.
[0041] "Fluoroalkyl" refers to an alkyl radical, as defined above, that is substituted by one or more fluoro radicals, for example, trifluoromethyl, difluoromethyl, fluoromethyl, 2,2,2-trifluoroethyl, l-fluoromethyl-2-fluoroethyl, and the like.
[0042] "Aminoalkyl" refers to an alkyl radical, as defined above, that is substituted by one or more amine radicals, for example, propan-2-amine, butane- 1,2-di amine, pentane- 1,2,4- triamine and the like.
[0043] "Hydroxyalkyl" refers to an alkyl radical, as defined above, that is substituted by one or more hydroxy radicals, for example, propan-l-ol, butane- 1,4-diol, pentane- 1, 2, 4-triol, and the like.
[0044] "Alkoxyalkyl" refers to an alkyl radical, as defined above, that is substituted by one or more alkoxy radicals, for example, methoxymethane, 1,3 -dimethoxybutane, 1- methoxypropane, 2-ethoxypentane, and the like.
[0045] "Cyanoalkyl" as used herein refers to an alkyl radical, as defined above, that is substituted by one or more cyano radicals, for example, acetonitrile, 2-ethyl-3- methylsuccinonitrile, butyronitrile, and the like.
[0046] “Heterocycle” as used herein refers to a saturated, unsaturated or aromatic ring comprising one or more heteroatoms. Exemplary heteroatoms include N, O, Si, P, B, and S atoms. The heterocycle may be attached to the rest of the molecule through any atom of the heterocycle, valence permitting, such as a carbon or nitrogen atom of the heterocycle. Heterocycles include 3- to 10-membered monocyclic rings, 6- to 12-membered bicyclic rings, and 6- to 12-membered bridged rings. A bicyclic heterocycle includes any combination of saturated, unsaturated and aromatic bicyclic rings, as valence permits. In an exemplary embodiment, an aromatic ring, e.g., pyridyl, may be fused to a saturated or unsaturated ring, e.g., cyclohexane, cyclopentane, morpholine, piperidine or cyclohexene. A bicyclic heterocycle includes any combination of ring sizes such as 4-5 fused ring systems, 5-5 fused ring systems, 5- 6 fused ring systems, 6-6 fused ring systems, 5-7 fused ring systems, 6-7 fused ring systems, 5-8 fused ring systems, and 6-8 fused ring systems. Bicyclic heterocycles may be fused, bridged, or spiro-ring systems. A spiro-ring system may be referred as a “spiroheterocycle”, “spiro heterocycle”, or “spiro-heterocycle”. In some cases, spiro-heterocycles, spiro heterocycles, or spiroheterocycles have at least two molecular rings with only one common atom. The spiroheterocycle, spiro heterocycle, or spiroheterocycle comprises one or more heteroatoms.
[0047] “Heterocyclene” refers to a divalent heterocycle linking the rest of the molecule to a radical group.
[0048] "Heteroaryl" or “aromatic heterocycle” refers to a radical derived from a heteroaromatic ring radical that comprises one to eleven carbon atoms and at least one heteroatom wherein each heteroatom may be selected from N, O, and S. As used herein, the heteroaryl ring may be selected from monocyclic or bicyclic and fused or bridged ring systems rings wherein at least one of the rings in the ring system is aromatic, z.e., it contains a cyclic, delocalized (4n+2) ^-electron system in accordance with the Hiickel theory. The heteroatom(s) in the heteroaryl radical may be optionally oxidized. One or more nitrogen atoms, if present, are optionally quatemized. The heteroaryl may be attached to the rest of the molecule through any atom of the heteroaryl, valence permitting, such as a carbon or nitrogen atom of the heteroaryl. Examples of heteroaryls include, but are not limited to, pyridine, pyrimidine, oxazole, furan, pyran, thiophene, isoxazole, benzimidazole, benzthiazole, and imidazopyridine.
[0049] An “X-membered heteroaryl” refers to the number of endocylic atoms, i.e., X, in the ring. For example, a 5-membered heteroaryl ring or 5-membered aromatic heterocycle has 5 endocyclic atoms, e.g., triazole, oxazole, thiophene, etc.
[0050] The term “unsaturated heterocycle” refers to heterocycles with at least one degree of unsaturation and excluding aromatic heterocycles. Examples of unsaturated heterocycles include
dihydropyrrole, dihydrofuran, oxazoline, pyrazoline, and dihydropyridine. Heterocycles may be optionally substituted by one or more substituents such as those substituents described herein. [0051] The term “substituted” refers to moieties having substituents replacing a hydrogen on one or more carbons or substitutable heteroatoms, e.g., NH, of the structure. It will be understood that “substitution” or “substituted with” includes the implicit proviso that such substitution is in accordance with permitted valence of the substituted atom and the substituent, and that the substitution results in a stable compound, z.e., a compound which does not spontaneously undergo transformation such as by rearrangement, cyclization, elimination, etc. In certain embodiments, substituted refers to moieties having substituents replacing two hydrogen atoms on the same carbon atom, such as substituting the two hydrogen atoms on a single carbon with an oxo, imino or thioxo group. As used herein, the term “substituted” is contemplated to include all permissible substituents of organic compounds. In a broad aspect, the permissible substituents include acyclic and cyclic, branched and unbranched, carbocyclic and heterocyclic, aromatic and non-aromatic substituents of organic compounds. The permissible substituents can be one or more and the same or different for appropriate organic compounds. For purposes of this disclosure, the heteroatoms such as nitrogen may have hydrogen substituents and/or any permissible substituents of organic compounds described herein which satisfy the valences of the heteroatoms.
[0052] In some embodiments, substituents may include any substituents described herein, for example: halogen, hydroxy, oxo (=0), thioxo (=S), cyano (-CN), nitro (-NO2), imino (=N-H), oximo (=N-0H), hydrazino (=N-
NH2), -Rb-0Ra, -Rb-OC(O)-Ra, -Rb-OC(O)-ORa, -Rb-0C(0)-N(Ra)2, -Rb-N(Ra)2, -Rb-C(O)Ra, -R b-C(O)ORa, -Rb-C(0)N(Ra)2, -Rb-0-Rc-C(0)N(Ra)2, -Rb-N(Ra)C(0)0Ra, -Rb-N(Ra)C(0)Ra, -Rb- N(Ra)S(O)tRa (where t is 1 or 2), -Rb-S(O)tRa (where t is 1 or 2), -Rb-S(O)tORa (where t is 1 or 2), and -Rb-S(O)tN(Ra)2 (where t is 1 or 2); and alkyl, alkenyl, alkynyl, aryl, aralkyl, aralkenyl, aralkynyl, cycloalkyl, cycloalkylalkyl, and heterocycle, any of which may be optionally substituted by alkyl, alkenyl, alkynyl, halogen, haloalkyl, haloalkenyl, haloalkynyl, oxo (=0), thioxo (=S), cyano (-CN), nitro (-NO2), imino (=N-H), oximo (=N-0H), hydrazine (=N- NH2), -Rb-0Ra, -Rb-0C(0)-Ra, -Rb-0C(0)-0Ra, -Rb-0C(0)-N(Ra)2, -Rb-N(Ra)2, -Rb-C(0)Ra, -R b-C(O)ORa, -Rb-C(0)N(Ra)2, -Rb-0-Rc-C(0)N(Ra)2, -Rb-N(Ra)C(0)0Ra, -Rb-N(Ra)C(0)Ra, -Rb- N(Ra)S(O)tRa (where t is 1 or 2), -Rb-S(O)tRa (where t is 1 or 2), -Rb-S(O)tORa (where t is 1 or 2) and -Rb-S(O)tN(Ra)2 (where t is 1 or 2); wherein each Ra is independently selected from hydrogen, alkyl, cycloalkyl, cycloalkylalkyl, aryl, aralkyl, heterocycloalkyl, heterocycloalkylalkyl, heteroaryl, or heteroarylalkyl, wherein each Ra, valence permitting, may be optionally substituted with alkyl, alkenyl, alkynyl, halogen, haloalkyl, haloalkenyl,
haloalkynyl, oxo (=0), thioxo (=S), cyano (-CN), nitro (-NO2), imino (=N-H), oximo (=N-0H), hydrazine (=N-NH2), -Rb-0Ra, -Rb-OC(O)-Ra, -Rb-OC(O)-ORa, -Rb-OC(O)-N(Ra)2,
-Rb-N(Ra)2, -Rb-C(O)Ra, -Rb-C(O)ORa, -Rb-C(O)N(Ra)2, -Rb-O-Rc-C(O)N(Ra)2, -Rb-N(Ra)C(O)O Ra, -Rb-N(Ra)C(O)Ra, -Rb-N(Ra)S(O)tRa (where t is 1 or 2), -Rb-S(O)tRa (where t is 1 or
2), -Rb-S(O)tORa (where t is 1 or 2) and -Rb-S(O)tN(Ra)2 (where t is 1 or 2); and wherein each Rb is independently selected from a direct bond or a straight or branched alkylene, alkenylene, or alkynylene chain, and each Rc is a straight or branched alkylene, alkenylene or alkynylene chain. [0053] As used herein, the term “electrophile” or “electrophilic moiety” is any moiety capable of reacting with a nucleophile (e.g., a moiety having a lone pair of electrons, a negative charge, a partial negative charge and/or an excess of electrons, for example an — SH group). Electrophiles typically are electron poor or comprise atoms which are electron poor. In certain embodiments, an electrophile contains a positive charge or partial positive charge, has a resonance structure which contains a positive charge or partial positive charge, or is a moiety in which delocalization or polarization of electrons results in one or more atoms which contains a positive charge or partial positive charge. In some embodiments, an electrophile comprises a conjugated double bond, for example an a,P-unsaturated carbonyl or a,P-unsaturated thiocarbonyl compound.
[0054] As used herein, the term “optional” or “optionally” means that the subsequently described event of circumstances may or may not occur, and that the description includes instances where the event or circumstance occurs and instances in which it does not. For example, “optionally substituted aryl” means that the aryl group may or may not be substituted and that the description includes both substituted aryl groups and aryl groups having no substitution.
[0055] As used in the specification and claims, the singular form “a”, “an” and “the” includes plural references unless the context clearly dictates otherwise.
[0056] The term “salt” or “pharmaceutically acceptable salt” refers to salts derived from a variety of organic and inorganic counter ions well known in the art. Pharmaceutically acceptable acid addition salts can be formed with inorganic acids and organic acids. Inorganic acids from which salts can be derived include, for example, hydrochloric acid, hydrobromic acid, sulfuric acid, nitric acid, phosphoric acid, and the like. Organic acids from which salts can be derived include, for example, acetic acid, propionic acid, glycolic acid, pyruvic acid, oxalic acid, maleic acid, malonic acid, succinic acid, fumaric acid, tartaric acid, citric acid, benzoic acid, cinnamic acid, mandelic acid, methanesulfonic acid, ethanesulfonic acid, /?-toluenesulfonic acid, salicylic acid, and the like. Pharmaceutically acceptable base addition salts can be formed with inorganic and organic bases. Inorganic bases from which salts can be derived include, for example, sodium, potassium, lithium, ammonium, calcium, magnesium, iron, zinc, copper, manganese, aluminum, and the like. Organic bases from which salts can be derived include, for example,
primary, secondary, and tertiary amines, substituted amines including naturally occurring substituted amines, cyclic amines, basic ion exchange resins, and the like, specifically such as isopropylamine, trimethylamine, diethylamine, triethylamine, tripropylamine, and ethanolamine. In some embodiments, the pharmaceutically acceptable base addition salt is chosen from ammonium, potassium, sodium, calcium, and magnesium salts.
[0057] The phrases “parenteral administration” and “administered parenterally” as used herein means modes of administration other than enteral and topical administration, usually by injection, and includes, without limitation, intravenous, intramuscular, intraarterial, intrathecal, intracapsular, intraorbital, intracardiac, intradermal, intraperitoneal, transtracheal, subcutaneous, subcuticular, intraarticular, subcapsular, subarachnoid, intraspinal and intrasternal injection and infusion.
[0058] The phrase “pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are, within the scope of sound medical judgment, suitable for use in contact with the tissues of human beings and animals without excessive toxicity, irritation, allergic response, or other problem or complication, commensurate with a reasonable benefit/risk ratio.
[0059] The phrase “pharmaceutically acceptable excipient” or “pharmaceutically acceptable carrier” as used herein means a pharmaceutically acceptable material, composition or vehicle, such as a liquid or solid filler, diluent, excipient, solvent or encapsulating material. Each carrier must be “acceptable” in the sense of being compatible with the other ingredients of the formulation and not injurious to the patient. Some examples of materials which can serve as pharmaceutically acceptable carriers include: (1) sugars, such as lactose, glucose and sucrose; (2) starches, such as com starch and potato starch; (3) cellulose, and its derivatives, such as sodium carboxymethyl cellulose, ethyl cellulose and cellulose acetate; (4) powdered tragacanth; (5) malt; (6) gelatin; (7) talc; (8) excipients, such as cocoa butter and suppository waxes; (9) oils, such as peanut oil, cottonseed oil, safflower oil, sesame oil, olive oil, com oil and soybean oil; (10) glycols, such as propylene glycol; (11) polyols, such as glycerin, sorbitol, mannitol and polyethylene glycol; (12) esters, such as ethyl oleate and ethyl laurate; (13) agar; (14) buffering agents, such as magnesium hydroxide and aluminum hydroxide; (15) alginic acid; (16) pyrogen- free water; (17) isotonic saline; (18) Ringer's solution; (19) ethyl alcohol; (20) phosphate buffer solutions; and (21) other non-toxic compatible substances employed in pharmaceutical formulations.
[0060] In certain embodiments, the term “prevent” or “preventing” as related to a disease or disorder may refer to a compound that, in a statistical sample, reduces the occurrence of the disorder or condition in the treated sample relative to an untreated control sample, or delays the
onset or reduces the severity of one or more symptoms of the disorder or condition relative to the untreated control sample.
[0061] The terms “treat,” “treating” or “treatment,” as used herein, may include alleviating, abating or ameliorating a disease or condition symptoms, preventing additional symptoms, ameliorating or preventing the underlying causes of symptoms, inhibiting the disease or condition, e.g., arresting the development of the disease or condition, relieving the disease or condition, causing regression of the disease or condition, relieving a condition caused by the disease or condition, or stopping the symptoms of the disease or condition either prophylactically and/or therapeutically.
[0062] The term “G12 mutants”, as used herein, refers to other oncogenic alleles of KRAS at amino acid position 12 (ie. G12X).
[0063] The term "KRas G12D-associated cancer" as used herein refers to cancers associated with or mediated by or having a KRas G12D mutation.
[0064] The term "KRas G12V-associated cancer" as used herein refers to cancers associated with or mediated by or having a KRas G12V mutation.
[0065] The term "KRas wildtype-associated cancer" as used herein refers to cancers associated with or mediated by or having a KRas wildtype.
[0066] The term “immunomodulator inhibitor” refers to an agent that modifies, or modulates, the immune system to help a subject respond to a disease or disorder.
[0067] The term “PD-1 inhibitor” refers to an agent that is capable of negatively modulating or inhibiting all or a portion of the PD-1 axis signaling activity and include agents that block PD- 1.
[0068] The term “PD-L1 inhibitor” refers to an agent that is capable of negatively modulating or inhibiting all or a portion of the PD-L1 axis signaling activity and include agents that block PD-L1.
[0069] The term “PD-1 binding antagonist” is a molecule that decreases, blocks, inhibits, abrogates or interferes with signal transduction resulting from the interaction of PD-1 with one or more of its binding partners, such as PD-L1 and/or PD-L2.
[0070] The term “PD-L1 binding antagonist” is a molecule that decreases, blocks, inhibits, abrogates or interferes with signal transduction resulting from the interaction of PD-L1 with either one or more of its binding partners, such as PD-1 and/or B7-1.
[0071] The term “biosimilar” means an antibody or antigen-binding fragment that has the same primary amino acid sequence as compared to a reference antibody (e.g., nivolumab or pembrolizumab) and optionally, may have detectable differences in post-translation
modifications (e.g., glycosylation and/or phosphorylation) as compared to the reference antibody (e.g., a different glycoform).
[0072] The terms "subject," "individual," and "patient" may be used interchangeably and refer to humans, as well as non-human mammals (e.g., non-human primates, canines, equines, felines, porcines, bovines, ungulates, lagomorphs, and the like). In various embodiments, the subject can be a human (e.g., adult male, adult female, adolescent male, adolescent female, male child, female child) under the care of a physician or other health worker in a hospital, as an outpatient, or other clinical context. In certain embodiments, the subject may not be under the care or prescription of a physician or other health worker.
[0073] As used herein, the phrase "a subject in need thereof' refers to a subject, as described infra, that suffers from, or is at risk for, a pathology to be prophylactically or therapeutically treated with a compound or salt described herein.
[0074] The terms “determining,” “measuring,” “evaluating,” “assessing,” “assaying,” and “analyzing” are often used interchangeably herein to refer to forms of measurement. The terms include determining if an element is present or not (for example, detection). These terms can include quantitative, qualitative or quantitative and qualitative determinations. Assessing can be relative or absolute. “Detecting the presence of’ can include determining the amount of something present in addition to determining whether it is present or absent depending on the context.
[0075] The terms “administer”, “administered”, “administers” and “administering” are defined as providing a composition to a subject via a route known in the art, including but not limited to intravenous, intraarterial, oral, parenteral, buccal, topical, transdermal, rectal, intramuscular, subcutaneous, intraosseous, transmucosal, or intraperitoneal routes of administration. In certain embodiments, oral routes of administering a composition can be used. The terms “administer”, “administered”, “administers” and “administering” a compound should be understood to mean providing a compound of the disclosure or a prodrug of a compound of the disclosure to the individual in need.
[0076] The term “effective amount” or “therapeutically effective amount” refers to that amount of a compound or salt described herein that is sufficient to effect the intended application including but not limited to disease treatment, as defined below. The therapeutically effective amount may vary depending upon the intended application (in vitro or in vivo), or the subject and disease condition being treated, e.g., the weight and age of the subject, the severity of the disease condition, the manner of administration and the like, which can readily be determined by one of ordinary skill in the art. The term can also apply to a dose that can induce a particular response in target cells, e.g., reduction of proliferation or down regulation of activity of a target
protein. The specific dose can vary depending on the particular compounds chosen, the dosing regimen to be followed, whether it is administered in combination with other compounds, timing of administration, the tissue to which it is administered, and the physical delivery system in which it is carried.
[0077] As used herein, a "therapeutically effective amount of a combination" of two compounds is an amount that together synergistically increases the activity of the combination in comparison to the therapeutically effective amount of each compound in the combination, i.e., more than merely additive.
[0078] As used herein, “synergy,” “synergetic,” “synergism,” or “synergistic effect” refer to two or more compounds or compositions, that individually produce an effect, however, together produce a combined effect that is greater than their individual effects.
[0079] The term “about” or “approximately” can mean within an acceptable error range for the particular value as determined by one of ordinary skill in the art, which will depend in part on how the value is measured or determined, i.e., the limitations of the measurement system. For example, “about” can mean within 1 or more than 1 standard deviation, per the practice in the art. Alternatively, “about” can mean a range of up to 20%, up to 15%, up to 10%, up to 5%, or up to 1% of a given value.
[0080] It is intended that every maximum numerical limitation given throughout this specification includes every lower numerical limitation, as if such lower numerical limitations were expressly written herein. Every minimum numerical limitation given throughout this specification will include every higher numerical limitation, as if such higher numerical limitations were expressly written herein. Every numerical range given throughout this specification will include every narrower numerical range that falls within such broader numerical range, as if such narrower numerical ranges were all expressly written herein.
[0081] The section headings used herein are for organizational purposes only and are not to be construed as limiting the subject matter described.
[0082] Any aspect or embodiment described herein can be combined with any other aspect or embodiment as disclosed herein.
COMPOSITIONS AND INHIBITORS
Immunomodulator Inhibitors
[0083] In an aspect, provided herein are methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of
i) an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, ii) and a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[0084] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, ii) and a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[0085] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, ii) and a compound of Formula (I- A), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[0086] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, ii) and a compound of Formula (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[0087] In an aspect, provided herein are methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of: i) an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, ii) and a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[0088] In some embodiments, the immunomodulator inhibitor is selected from a PD-1 inhibitor, a PD-Ll inhibitor, and a CTLA-4 inhibitor. In some cases, the immunomodulator inhibitor is a PD-1 inhibitor. In some cases, the immunomodulator inhibitor is a PD-L1 inhibitor.
In some cases, the immunomodulator inhibitor is a CTLA-4 inhibitor. In some embodiments, the immunomodulator inhibitor is selected from a PD-1/PD-L1 checkpoint inhibitor.
[0089] In some embodiments, the immunomodulator inhibitor is immune checkpoint inhibitor.
[0090] In some embodiments, the immunomodulator inhibitor is pembrolizumab.
[0091] In some embodiments, the immunomodulator inhibitor is relatlimab.
PD-1/PD-L1 Inhibitors
[0092] In some embodiments, programmed death protein 1 (PD-1) is an immunoinhibitory receptor that is primarily expressed on activated T and B cells. PD-1 is a 55 kDa type I transmembrane protein that is part of the Ig gene superfamily (Agata et al. (1996) Int Immunol 8:765-72). PD-1 contains a membrane proximal immunoreceptor tyrosine inhibitory motif (ITIM) and a membrane distal tyrosine-based switch motif (ITSM). Two ligands that bind to PD-1 have been identified, PD-L1 and PD-L2, that have been shown to downregulate T cell activation upon binding to PD-1 (Freeman et al. (2000) J Exp Med 192: 1027-34). PD-L1 is a ligand for PD-1 and is abundant in a variety of human cancers (Dong et al. (2002) Nat. Med. 8:787-9). In some cases, the interaction between PD-1 and PD-L1 results in a decrease in tumor infiltrating lymphocytes, a decrease in T-cell receptor mediated proliferation, and immune evasion by the cancerous cells (Dong et al. (2003) J. Mol. Med. 81 :281-7).
[0093] In some embodiments, immune suppression can be reversed by inhibiting the local interaction of PD-1 with PD-L1, and the effect is additive when the interaction of PD-1 with PD- L2 is blocked as well. In some cases, disruption of the PD-1/PD-L1 interaction has been shown to increase T cell proliferation and cytokine production and block progression of the cell cycle.
[0094] In some embodiments, PD-L1 is upregulated in many cancers and contributes to evasion of the host immune system, blocking the interaction between PD-1 and PD-L1 has garnered the attention of the pharmaceutical industry leading to a new break-through class of immune checkpoint therapies for a wide range of cancers. In some cases, the PD-1/PD-L1 pathway is a well-validated target for the development of antibody therapeutics for cancer treatment and several anti -PD-1 and anti-PD-Ll antibodies have undergone human clinical trials for a wide- variety of cancers including NSCLC, renal cell carcinoma, melanoma, head and neck squamous cancer, ureothelial cancer, hepatocellular carcinoma, and other cancers. In some cases, anti-PD-1 antibodies include nivolumab (Opdivo®), pembrolizumab (Keytruda®), cemiplimab (Libtayo®) and tislelizumab, and biosimilars thereof. In some cases, anti-PD-Ll antibodies include atezolizumab (Tecentriq®), avelumab (Bavencio®), and durvalumab (Imfinzi®), and biosimilars thereof.
[0095] In some embodiments, methods for manufacturing agents that disrupt PD-1/PD-L1 signaling axis, including the antibodies described herein, are well known to those skilled in the art and agents that disrupt PD-1/PD-L1 signaling axis may be obtained from a wide-variety of commercial suppliers, in forms suitable for both research or approved human clinical use. In some cases, suitable agents that disrupt PD-1/PD-L1 signaling for use in the compositions and methods disclosed herein and methods for preparing such agents, and diagnostic and efficacy markers useful for monitoring treatment are disclosed in US Patent Application Publication Nos: 20180327848; 20180237524; 20180148790; 20180111996; 20160305947; 20160304969;
20160304606; 20150232555; 20150079109; 20140348743; 20140294852; 20140271684;
20140234296; 20130133091; 20110123550; and 20090217401.
PD-1 inhibitor
[0096] In an aspect, provided herein are methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a combination of: i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[0097] In an aspect, provided herein are methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[0098] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[0099] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of:
i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I-A), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00100] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00101] In an aspect, provided herein are methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of: i) an PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00102] In some embodiments, a PD-1 inhibitor is selected from a PD-1 binding antagonist. In some cases, the PD-1 binding antagonist is selected from anti-PD-1 antibodies, antigen binding fragments thereof, immunoadhesins, aptamers, fusion proteins, and oligopeptides. In some cases, the PD-1 binding antagonist is an anti-PD-1 antibody.
[00103] In some embodiments, the PD-1 inhibitor is a molecule that inhibits the binding of PD-1 to its binding partners. In some cases, the PD-1 inhibitor inhibits the binding of PD-1 to PD-L1 and/or PD-L2. In some cases, PD-1 inhibitors include anti-PD-1 antibodies, antigen binding fragments thereof, immunoadhesins, fusion proteins, oligopeptides and other molecules that decrease, block, inhibit, abrogate or interfere with signal transduction resulting from the interaction of PD-1 with PD-L1 and/or PD-L2. In some cases, a PD-1 inhibitor reduces the negative co-stimulatory signal mediated by or through cell surface proteins expressed on T lymphocytes mediated signaling through PD-1 so as render a dysfunctional T-cell less non- dysfunctional. In some cases, the PD-1 inhibitor is an anti-PD-1 antibody. In some cases, the PD- 1 antibody is pembrolizumab, or a biosimilar thereof. In some cases, the PD-1 antibody is cemiplimab, or a biosimilar thereof. In some cases, the PD-1 antibody is tislelizumab, or a biosimilar thereof.
PD-L1 Inhibitors
[00104] In an aspect, provided herein are methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a combination of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00105] In an aspect, provided herein are methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00106] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
[00107] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I- A), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
[00108] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I-B), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
[00109] In an aspect, provided herein are methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of: i) an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00110] In some embodiments, a PD-L1 inhibitor is selected from a PD-L1 binding antagonist. In some cases, the PD-L1 binding antagonist is selected from an anti-PD-Ll antibody, antigen binding fragments thereof, immunoadhesins, aptamers, fusion proteins, and oligopeptides. In some cases, the PD-L1 binding antagonist is an anti-PD-Ll antibody.
[00111] In some embodiments, a PD-L1 inhibitor is a molecule that inhibits the binding of PD-L1 to its binding partners. In some cases, the PD-L1 inhibitor inhibits binding of PD-L1 to PD-1 and/or B7-1. In some cases, the PD-L1 inhibitors include anti-PD-Ll antibodies, antigen binding fragments thereof, immunoadhesins, fusion proteins, oligopeptides and other molecules that decrease, block, inhibit, abrogate or interfere with signal transduction resulting from the interaction of PD-L1 with one or more of its binding partners, such as PD-1 and/or B7-1. In some cases, a PD-L1 inhibitor reduces the negative co-stimulatory signal mediated by or through cell surface proteins expressed on T lymphocytes mediated signaling through PD-L1 so as render a dysfunctional T-cell less non-dysfunctional. In some cases, a PD-L1 inhibitor is an anti-PD-Ll antibody. In some cases, an anti- PD-L1 antibody is avelumab or a biosimilar thereof. In some cases, an anti-PD-Ll antibody is atezolizumab or a biosimilar thereof. In some cases, an anti-PD- Ll antibody is durvalumab or a biosimilar thereof. In some cases, an anti-PD-Ll antibody is BMS-936559 (MDX-1105) or a biosimilar thereof.
CTLA-4 inhibitor
[00112] In an aspect, provided herein are methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a combination of: i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00113] In an aspect, provided herein are methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of:
i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00114] In an aspect, provided herein are methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of: i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00115] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
[00116] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I- A), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
[00117] In an aspect, provided herein are methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of: i) an CTLA-4 inhibitor, or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof, and ii) a compound of Formula (I-B), or a pharmaceutically acceptable salt, or a pharmaceutical composition thereof.
[00118] In some embodiments, the CTLA-4 inhibitor is selected from Ipilimumab.
KRAS Inhibitors
[00119] The following is a discussion of compounds and salts thereof that may be used in the methods of the disclosure. The compounds and salts (e.g., a compound of Formula (I), (I- A), or (I-B)) may be used in combination with at least one other inhibitor (e.g., immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor). The compounds and salts (e.g., a compound of Formula (I), (I- A), or (I-B)) may be used in combination with one other inhibitor (e.g., immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor). In some cases, a compound of Formula (I), (LA), or (LB), may be used in the methods of the disclosure. In some cases, a compound of Formula (I), (LA), or (LB), may be referred to as a KRAS inhibitor. In some cases, a compound of Formula (I), (LA), or (LB), may be referred to as a KRAS inhibitor.
[00120] In some embodiments, a compound represented by the structure of Formula (I):
Formula (I), or a pharmaceutically acceptable salt thereof wherein:
B is selected from a 7- to 15-membered heterocycle and C7-C15 carbocycle, wherein the 7- to 15-membered heterocycle and C7-C15 carbocycle are each optionally substituted with one or more substituents independently selected from halogen, -CN, -NO2, =0, -N(R21)2, -B(OR21)2, - OR21, -SR21, -S(O)2(R21), -S(O)2N(R21)2, -NR21S(O)2R21, -C(O)N(R21)2, -C(O)NR21OR21, - N(R21)C(O)R21, -N(R21)C(O)N(R21)2, -N(R21)C(O)OR21, -C(0)R21, C(0)0R21, -0C(0)R21, - OC(O)N(R21)2, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-C12 carbocycle and 5- to 12-membered heterocycle;
R1 is selected from hydrogen and 5- to 15-membered heterocycle, wherein the 5- to 15- membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(=NR20)N(R20)2, -C1-6 alkyl(=NOR20), - C(O)NR20OR20, -N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, -C(O)R20, - C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, -NO2, =O, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl- N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, C 1-6 haloalkyl, Ci-e alkyl- SO2R20, C1-6 alkoxyalkyl, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-C 12 carbocycle and 5- to 12-
membered heterocycle, wherein the C3-C12 carbocycle and 5- to 12-membered heterocycle are each optionally substituted independently with one or more R1*; each R1* is independently selected from halogen, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(O)NR20OR20, - N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, -C(O)R20, -C(O)OR20, - OC(O)R20, -OC(O)N(R20)2, -NO2, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-e alkynyl, and C3-C 12 carbocycle;
Y is selected from a bond, -0-, -S-, and -N(R5)-;
R2 is selected from heterocycle, Ci-Ce alkyl, -L-heterocycle, -L-N(R23)2, -L-OR23, -L-aryl, -L-heteroaryl, -L-cycloalkyl, -L-NHC(=NH)NH2, -L-C(O)N(R23)2, -L-CI-C6 haloalkyl, -L-OR23, - L-NR23C(O)-aryl, -L-C00H, -L-NR23S(O)2(R23), -L-S(O)2N(R23)2, -L-N(R23)C(O)(OR23), -L- OC(O)N(R23)2, and -L-C(=O)OCi-Ce alkyl, wherein the heterocycle, the heterocycle portion of - L-heterocycle, and the cycloalkyl portion of the -L-cycloalkyl are each optionally substituted with one or more R6, and wherein the aryl portion of -L-NR23C(O)-aryl, the aryl portion of -L- NR23C(O)-aryl, the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R7;
R3 is selected from hydrogen, halogen, -CN, -NO2, -N(R20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -N(R20)C(O)R20, - N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, -C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, CI-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-C12 carbocycle and 5- to 12-membered heterocycle; each R4 is independently selected from halogen, -NO2, =0, =S, -CN, Ci-6 alkyl, C2-6 alkynyl, C 1-6 alkyl -N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, C 1-6 cyanoalkyl, Ci-6 hydroxyalkyl, and Ci-6 haloalkyl; n is selected from 0, 1, 2, 3, and 4; each R5 is independently selected from hydrogen and Ci-Ce alkyl; each R6 is independently selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, oxo, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, =CH2, NMO-C1-C3 alkyl, C1-C3 aminoalkyl, - N(R5)S(O)2(R5), -Q-phenyl, -Q-phenylSCLF, -NHC(O)phenyl, - NHC(O)phenylSO2F, C1-C3 alkyl substituted pyrazolyl, tert-butyldimethylsilyloxyCFB-, -N(R5)2, (C1-C3 alkoxy)Ci-C3 alkyl-, (Ci- C3 alkyl)C(=O), oxo, (C1-C3 haloalkyl)C(=O)-, -SO2F, (C1-C3 alkoxy)Ci-C3 alkoxy, - CH2OC(O)N(R5)2, -CH2NHC(O)OC1-C6 alkyl, -CH2NHC(O)N(R5)2, -CH2NHC(O)CI-C6 alkyl, - CH2(pyrazolyl), -CH2NHSO2C1-C6 alkyl, -CH2OC(O)heterocycle, -OC(O)N(R5)2,
OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl), -OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl)phenyl(Ci-C3 alkyl)N(CH3)2, -OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl)phenyl, -OC(O)heterocycle, -O-Ci-C3 alkyl, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, and - CH2heterocycle, wherein the phenyl of -NHC(O)phenyl and -OC(O)NH(CI-C3 alkyl)(Ci-C3 alkyl)phenyl are optionally substituted with one or more substituents selected from -C(O)H and OH, and wherein the alkyl of -O-Ci-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo and hydroxy; and wherein the heterocycle of -CH2heterocyclyl is optionally substituted with oxo; each Q is selected from a bond, S, and O; each R7 is independently selected from halogen, hydroxy, HC(=O)-, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 hydroxyalkyl, and -N(R5)2; each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen, hydroxy, C1-6 alkoxy, C1-C4 hydroxyalkyl, C1-C4 alkyl, C3-Ce carbocycle, and 3- to 8-membered heterocycle, wherein the C3-Ce carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -NO2, =0, =S, -CN, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, Ci-ehydroxyalkyl, C1-6 haloalkyl; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-Ce carbocycle or 3- to 8-membered heterocycle, wherein the C3-Ce carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -NO2, =0, =S, -CN, Ci-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and C1-6 haloalkyl; each R20 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-i2 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-i2 carbocycle, and 3- to 12-membered heterocycle; each R21 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-i2 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-i2 carbocycle, and 3- to 12-membered heterocycle; each R23 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-i2 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6
alkyl)2, Ci-io alkyl, -Ci-io haloalkyl, -O-Ci-io alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle.
[00121] In some embodiments, for a compound or salt for Formula (I), B is selected from an optionally substituted 7- to 15-membered fused heterocycle and optionally substituted C7-C15 fused carbocycle. In some cases, and optionally substituted C7-C15 fused carbocycle. In some cases, B is an optionally substituted 7- to 15-membered fused heterocycle. In some cases, B is an optionally substituted unsaturated 7- to 15-membered fused heterocycle. In some cases, B is an optionally substituted 7- to 15-membered fused heteroaryl. In some cases, B is selected from an optionally substituted 7- to 15-membered fused heteroaryl and optionally substituted C7-C15 fused aryl. In some cases, B is an optionally substituted unsaturated C7-C15 fused carbocycle. In some cases, B is an optionally substituted 7- to 15-membered fused heterocycle, wherein the fused heterocycle is partially unsaturated. In some cases, B is an optionally substituted 7- to 15- membered fused heterocycle, wherein the fused heterocycle is partially saturated.
[00122] In some embodiments, for a compound or salt for Formula (I), B is selected from an optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle. In some cases, and optionally substituted Cs-Cis fused carbocycle. In some cases, B is an optionally substituted 8- to 15-membered fused heterocycle. In some cases, B is an optionally substituted unsaturated 8- to 15-membered fused heterocycle. In some cases, B is an optionally substituted 8- to 15-membered fused heteroaryl. In some cases, B is selected from an optionally substituted 8- to 15-membered fused heteroaryl and optionally substituted Cs-Cis fused aryl. In some cases, B is an optionally substituted unsaturated Cs-Cis fused carbocycle. In some cases, B is an optionally substituted 8- to 15-membered fused heterocycle, wherein the fused heterocycle is partially unsaturated. In some cases, B is an optionally substituted 8- to 15- membered fused heterocycle, wherein the fused heterocycle is partially saturated.
[00123] In some embodiments, for a compound or salt for Formula (I), B is selected from an optionally substituted 8- to 15-membered fused heterocycle, wherein the fused heterocycle is formed by combining three rings (e.g., tricyclic). In some cases, B is selected from an optionally substituted 8- to 15-membered fused heterocycle, wherein the fused heterocycle is formed by combining two rings (e.g., bicyclic). In some cases, for B the optionally substituted 8- to 15- membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle are each independently bicyclic or tricyclic. In some cases, for B the optionally substituted 8- to 15- membered fused heterocycle is bicyclic. In some cases, for B the optionally substituted 8- to 15- membered fused heterocycle is tricyclic.
[00124] In some embodiments, for a compound or salt for Formula (I), the heterocycle or carbocycle of B is bicyclic. In some cases, the heterocycle or carbocycle of B is tricyclic. In some cases, the tricyclic heterocycle contains three interconnected rings of atoms.
[00125] In some embodiments, for a compound or salt for Formula (I), for B, the heterocycle and carbocycle are each independently selected from bicyclic and tricyclic. In some cases, for B, the heterocycle and carbocycle are each independently tricyclic. In some cases, for B, the heterocycle and carbocycle are each independently bicyclic.
[00126] In some embodiments, for a compound or salt for Formula (I), for B, the optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle are selected from
, each of which is optionally substituted with one or more substituents. In some cases, for B, the optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle are selected from
, each of which is optionally substituted with one or more substituents In some cases, B
, each of which is optionally substituted with one or more substituents. In some cases,
[00127] In some embodiments, for a compound or salt for Formula (I), B is selected from
substituents.
[00128] In some embodiments, for a compound or salt for Formula (I), for B, the optional substituents of the heterocycle and carbocycle are each independently selected from halogen, - CN, -NO2, =0, -N(R21)2, -B(OR21)2, -OR21, -SR21, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, C1-6 alkyl, C2-6 alkenyl, C2-e alkynyl, C3-C12 carbocycle and 5- to 12-membered heterocycle. In some cases, the optional substituents of the heterocycle and carbocycle are each independently selected from halogen, -CN, -NO2, =O, -
N(R21)2, -B(OR21)2, -OH, -SR21, C 1-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, C2-6 alkenyl, and C2-e alkynyl. In some cases, for B, the one or more optional substituents of the heterocycle and carbocycle are independently selected at each occurrence from halogen, oxo, -NH2, C1-C3 alkyl, -B(0H)2, -OH, -O-C1-C3 haloalkyl, -C(0)NH2, -NH2, =0, -CN, C1-6 alkoxy, C1-6 hydroxyalkyl, and C2-e alkynyl. In some cases, the optional substituents of the heterocycle and carbocycle are each independently selected from halogen, -CN, =0, -NH2, -N(CI-6 alkyl)H -N(CI-6 alkyl)2, -OH, C1-6 aminoalkyl, Ci- 6 alkoxy, C1-6 alkoxyalkyl, Ci-e hydroxyalkyl, C1-6 cyanoalkyl, C 1-6 haloalkyl, C1-6 alkyl, C2-6 alkenyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of the heterocycle and carbocycle are each independently selected from oxo, -NH2, halogen, C1-C3 alkyl. In some cases, for B, the optionally substituted 8- to 15-membered fused heterocycle and optionally
[00129] In some embodiments, for a compound or salt for Formula (I), B is selected from an optionally substituted 7- to 12-membered fused heterocycle and optionally substituted C9-10 fused carbocycle. In some cases, the heterocycle of B has at least one sulfur atom. In some cases, the heterocycle of B has one or sulfur atoms. In some cases, the heterocycle of B has at least one
substituted. In some cases, the one or more optional substituents of B are independently selected at each occurrence from halogen, C1-C3 alkyl, -B(OR20)2, -OR20, -C(O)N(R20)2, -N(R20)2, =0, - CN, -NHCN, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, Ci-e haloalkyl, C2-6 alkenyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of B are independently selected at each occurrence from halogen, C1-C3 alkyl, -OH, -NH2, =0, and -
[00130] In some embodiments, for a compound or salt for Formula (I), B is selected from an optionally substituted 8- to 10-membered fused heterocycle having at least one sulfur atom. In
is optionally substituted. In some cases, the one or more optional substituents of B are independently selected at each occurrence from halogen, C1-C3 alkyl, -OR20, -C(O)N(R20)2, - N(R20)2, =0, -CN, -NHCN, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, C1-6 haloalkyl, C2-6 alkenyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of B are independently selected at each occurrence from halogen, C1-C3 alkyl, -NH2, and -CN. In some cases, B is substituted. In some cases, B is substituted with at least one -NH2. In some cases, B is selected from
some cases, B is substituted with at least one -NH2 at least one -CN. In some cases, B is selected
[00131] In some embodiments, for a compound or salt for Formula (I), B is an optionally substituted 7- to 11 -membered fused heterocycle. In some cases, B is an optionally substituted 8- to 10-membered fused heterocycle. In some cases, B is an optionally substituted 7-membered fused heterocycle. In some cases, B is an optionally substituted 8-membered fused heterocycle. In some cases, B is an optionally substituted 9-membered fused heterocycle. In some cases, B is an optionally substituted 10-membered fused heterocycle. In some cases, the heterocycle of B is an
unsaturated heterocycle. In some cases, the heterocycle of B is a non-aromatic heterocycle. In some cases, B has at least one sulfur atom. In some cases, B has at two sulfur atoms. In some cases, B has at least one sulfur atom and at least one nitrogen atom. In some cases, B has at least one sulfur atom and at least one oxygen atom. In some cases, B has only 1 heteroatom. In some cases,
B has at least 2 heteroatoms. In some cases, B is selected from
each of which is optionally substituted. In some cases, B is selected from
each of which is optionally substituted. In some cases, B is selected from
, each of which is optionally substituted. In some cases, the one or more optional substituents of B, are independently selected at each occurrence from halogen, oxo, -NH2, C1-C3 alkyl, -B(0H)2, -OH, -O-C1-C3 haloalkyl, -C(0)NH2, -NH2, =0, -CN, C1-6 alkoxy, C1-6 hydroxyalkyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of B, are independently selected at each occurrence from halogen, C1-C3 alkyl, -NH2, and -CN. In some cases, B is substituted with at least three substituents. In some cases, B is substituted with at least two substituents. In some cases, B is substituted with at least one substituent. In some cases, B is substituted with at least one substituent selected from halogen, C1-C3 alkyl, -NH2, and -CN. In some cases, B is substituted with at least one substituent selected from halogen. In some cases, B is substituted with at least one substituent selected from -NH2. In some cases, B is substituted with at least one substituent selected from -CN. In some cases, B is selected from
,
[00132] In some embodiments, for a compound or salt for Formula (I), R3 is selected from hydrogen, halogen, -CN, -NO2, -N(R20)2, -OR20, -SR20, -S(O)2(R20), -S(O)2N(R20)2, - NR20S(O)2R20, -C(O)N(R20)2, -N(R20)C(O)R20, -N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, - C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-e alkynyl. In some cases, R3 is selected from hydrogen, halogen, -CN, -NO2, -N(R20)2, -OR20, -SR20, -C(O)N(R20)2, -C(O)R20, -C(O)OR20, -OC(O)R20, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-e alkynyl. In some cases, R3 is selected from hydrogen, halogen, -CN, -NO2, -NH2, -N(CI-6 alkyl)H -N(CI-6 alkyl)2,-OH, - C(O)N(R20)2, -C(O) R20, -C(O)OR20, -OC(O) R20, CI-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl,
C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, C2-6 alkenyl, and C2-e alkynyl. In some cases, R3 is selected from hydrogen, -CN, -C(O)R20, C 1-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, and Ci-6 alkyl. In some cases, R3 is selected from hydrogen, -CN, -C(O)R20, Ci-6 hydroxyalkyl, and Ci-6 alkyl. In some cases, R3 is selected from hydrogen, -CN, -C(O)H, Ci hydroxyalkyl, and Ci-6 alkyl. In some cases, R3 is selected from hydrogen, fluorine, and -CN. In some cases, R3 is selected from fluorine. In some cases, R3 is selected from hydrogen. In some cases, R3 is selected from -CN.
[00133] In some embodiments, for a compound or salt for Formula (I), Y is -O-. In some cases, Y is a bond. In some cases, Y is -S-. In some cases, Y is -N(R5)-.
[00134] In some embodiments, for a compound or salt for Formula (I), L is selected from Ci- C4 alkylene. In some cases, L is selected from an unsubstituted C1-C4 alkylene. In some cases, L is selected from an unsubstituted Ci alkylene. In some cases, two substituents on the same carbon
atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle, wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -NO2, =0, =S, -CN, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl. In some cases, two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle. In some cases, two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle. In some embodiments, for a compound or salt for Formula (I), L is selected from Ci- C4 alkylene. In some cases, L is selected from unsubstituted C1-C4 alkylene. In some cases, each L is independently selected from a C1-C4 alkylene optionally substituted; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents selected from halogen, -OH, -NO2, =0, =S, -CN, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl. In some cases, the optional substituents of L are selected from C1-C4 hydroxyalkyl, C1-C4 alkyl, C3-C6 carbocycle; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle wherein the C3-C6 carbocycle and 3- to 8- membered heterocycle are optionally substituted with one or more substituents selected from
cases, each L is independently selected from a substituted C1-C4 alkylene, and wherein two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle 3- to 5- membered heterocycle. In some cases, each L is independently selected from a substituted C2-3 alkylene, and wherein two substituents on the same carbon atom of L come together to form a C3 carbocycle or 4-membered heterocycle, wherein the C3 carbocycle is optionally substituted with one or more substituents selected from halogen. In some cases, each L is independently selected
cases, each L is independently selected from -k . In some cases, each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen and C1-C4 alkyl. In some cases, L is selected from
[00135] In some embodiments, for a compound or salt for Formula (I), each L is independently selected from an unsubstituted C1-C4 alkylene. In some cases, L is selected from^^^ and
.
[00136] In some embodiments, for a compound or salt for Formula (I), R2 is selected from heterocycle, Ci-Ce alkyl, -L-heterocycle, -L-N(R23)2, -L-OR23, -L-aryl, -L-heteroaryl, -L- cycloalkyl, -L-N(R23)2, -L-NHC(=NH)NH2, -L-C(O)N(R23)2, -L-CI-C6 haloalkyl, -L-OR23, -L- NR23C(O)-aryl, -L-COOH, -L-NR23S(O)2(R23), -L-S(O)2N(R23)2, -L-N(R23)C(O)(OR23), -L- OC(O)N(R23)2, and -L-C(=O)OCi-Ce alkyl, wherein the heterocycle, the heterocycle portion of - L-heterocycle, and the cycloalkyl portion of the -L-cycloalkyl are each optionally substituted with one or more R6, and wherein the aryl portion of -L-NR23C(O)-aryl, the aryl portion of -L- NR23C(O)-aryl, the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R7.
[00137] In some embodiments, for a compound or salt for Formula (I), R2 is selected from heterocycle, -L-heterocycle, -L-aryl, -L-heteroaryl, and -L-N(R23)2, wherein the heterocycle, the heterocycle portion of -L-heterocycle, are each optionally substituted with one or more R6, and wherein the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R7.
[00138] In some embodiments, for a compound or salt for Formula (I), R2 is -L-heterocycle, wherein the heterocycle portion is optionally substituted. In some cases, R2 is -L-heterocycle, wherein the heterocycle portion is a bicyclic heterocycle. In some cases, R2 is -L-heterocycle, wherein the heterocycle portion is a monocyclic heterocycle. In some cases, R2 is -L-heterocycle, wherein the heterocycle portion is a saturated heterocycle. In some cases, R2 is selected from a - L-5- to 10-membered heterocycle. In some cases, R2 is selected from a -(C1-C2 alkylene)-5- to 10-membered heterocycle. In some cases, R2 is selected from a -L-5- to 8-membered heterocycle. In some cases, R2 is selected from a -L-5- to 8-membered saturated heterocycle. In some cases, R2 is a -L-5-membered heterocycle. In some cases, R2 is a -L-8-membered heterocycle. In some cases, the heterocycle contains at least 1 nitrogen atom. In some cases, the heterocycle contains
at most 1 nitrogen atom. In some cases, the heterocycle contains 1 nitrogen atom. In some cases, the bicyclic heterocycle contains at least 1 nitrogen atom. In some cases, the bicyclic heterocycle contains at most 1 nitrogen atom. In some cases, the bicyclic heterocycle contains 1 nitrogen atom. In some cases, Y-R2 is selected from
wherein the heterocycle portion is optionally substituted. In some cases, Y-R2 is selected from
, wherein the heterocycle portion is optionally substituted. In some cases, the heterocycle portion is optionally substituted with one or more substituents selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, -CN, and C1-C3 aminoalkyl. In some cases, the heterocycle portion is optionally substituted with one or more substituents selected from halogen, hydroxy, -CN, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, and C1-C3 aminoalkyl. In some cases, the heterocycle portion is optionally substituted with one or more substituents selected from C1-C3 alkyl and halogen. In
some cases, Y-R2 is selected from
In some cases, Y-R2 is
[00139] In some embodiments, for a compound or salt of Formula (I), R2 is selected from optionally substituted -L-heterocycle. In some cases, the heterocycle is a bicyclic heterocycle. In some cases, the heterocycle is a monocyclic heterocycle. In some cases, the heterocycle has only 1 nitrogen atom. In some cases, the heterocycle has only 1 nitrogen atom and no other heteroatoms. In some cases, Y-R2 is selected from
, wherein the heterocycle portion is optionally substituted. In some cases, Y-R2 is selected from
wherein the heterocycle portion is optionally substituted. In some
cases, Y-R2 is selected from
, wherein the heterocycle portion is optionally substituted. In some cases, Y-R2 is selected from
, wherein the heterocycle portion is optionally substituted. In some cases, the heterocycle is optionally substituted with one or more substituent selected from halogen, hydroxy, C1-C3 alkyl, -N(R5)S(O)2(R5), -OC(O)N(R5)2, oxo, =CH2, =NO-CI-C3 alkyl, -CH2OC(O)heterocycle, -QHfcheterocycle, -CH2OC(O)N(R5)2, and -O-C1-C3 alkyl, wherein the alkyl of -O-C1-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo, and hydroxy. In some cases, the heterocycle is substituted with at least one halogen. In some cases, the heterocycle is substituted with at least one =CH2. In some
[00140] In some embodiments, for a compound or salt of Formula (I), R2 is -L-heteroaryl, wherein the heteroaryl portion is optionally substituted with one or more R7. In some cases, the heteroaryl is selected from a 5- to 6-membered heteroaryl, wherein the heteroaryl portion is optionally substituted with one or more R7. In some cases, the heteroaryl is selected from a 5- membered heteroaryl, wherein the heteroaryl portion is optionally substituted with one or more R7. In some cases, the heteroaryl has at least one nitrogen atom. In some cases, the heteroaryl has two nitrogen atoms. In some cases, the heteroaryl has three nitrogen atoms. In some cases, the
heteroaryl is selected from
which is optionally substituted. In some cases, the heteroaryl is
which is optionally substituted. In some cases, Y-R2 is selected from
, wherein the heteroaryl portion is optionally substituted with one or more R7. In some cases, each R7 is independently selected from C1-C4 alkyl, halogen,
[00141] In some embodiments, for a compound or salt of Formula (I), R2 is -L-aryl, optionally substituted with one or more R7. In some cases, wherein Y-R2 is selected from
wherein the heterocycle portion is optionally substituted with one or more R7. In some cases, Y-
R2 is selected from
[00142] In some embodiments, for a compound or salt of Formula (I), R2 is -L-N(R23)2. In some cases, selected from
In some embodiments, for a compound or salt of Formula (I), R2 is heterocycle, optionally substituted with one or more R6. In some cases, the heterocycle
which is optionally substituted. In some cases, the heterocycle
some cases, Y-R2 is
[00143] In some embodiments, for a compound or salt of Formula (I), R2 is selected from heterocycle, -L-heterocycle, wherein the heterocycle, and the heterocycle portion of -L- heterocycle, are each optionally substituted with one or more R6; -L-aryl, and -L-heteroaryl, wherein the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R7; and -L-N(R23)2. In some cases, the heterocycle of R2 is selected from
wherein the heterocycle of R2 is optionally substituted with one or more R6; wherein the aryl and heteroaryl of R2 is selected from
from
, , wherein the aryl and the heteroaryl are each optionally substituted with one or more R7; and
, In some cases, each R6 is independently selected from halogen, hydroxy, C1-C3 alkyl, C1-C3 haloalkyl, -N(R5)S(O)2(R5), -OC(O)N(R5)2, =CH2, oxo, =NO-CI-C3 alkyl, -CH2OC(O)heterocycle, -CH2heterocycle, -CH2OC(O)N(R5)2, and -O-C1-C3 alkyl, wherein the alkyl of -O-C1-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo, and hydroxy; and wherein each R7 is selected from C1-C3 alkyl, halogen and C1-C3 haloalkyl. In some cases, the heterocycle of R2, the aryl and heteroaryl of R2, and -
-L-heterocycle, are each optionally substituted with one or more R6;
the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with
[00144] In some embodiments, for a compound or salt of Formula (I), L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from hydroxy, C1-C4 hydroxy alkyl and C1-C4 alkyl. In some cases, L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from C1-C4 alkyl. In some cases, L is selected from C1-C4 alkylene. In some cases, L is selected from C1-C2 alkylene. In some cases, L is
In some
[00145] In some embodiments, for a compound or salt of Formula (I), each L is independently selected from an optionally substituted C1-C4 alkylene; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle, wherein the C3-C6 carbocycle is optionally substituted with one or more substituents selected from halogen, -OH, - NO2, =0, =S, -CN, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, Ci-e haloalkyl. In some cases, the optional substituents of L are selected from C1-C4 hydroxyalkyl, C1-C4 alkyl, C3-C6 carbocycle; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle wherein the C3-C6 carbocycle and 3- to 8-
membered heterocycle are optionally substituted with one or more substituents selected from halogen and Ci-6 haloalkyl.
[00146] In some embodiments, for a compound or salt of Formula (I), each L is independently selected from a substituted C1-C4 alkylene, wherein two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle. In some cases, the C3-C6 carbocycle is optionally substituted with one or more substituents selected from halogen, -OH, -NO2, =O, =S, -CN, C1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C 1-6 haloalkyl.
[00147] In some embodiments, for a compound or salt of Formula (I), each L is independently selected from a substituted C1-C4 alkylene, and two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle. In some cases, each L is independently selected from a substituted C3 alkylene, and wherein two substituents on the same carbon atom of L come together to form a C3 carbocycle. In some cases, each L is independently selected from
[00148] In some embodiments, for a compound or salt of Formula (I), R2 is selected from -L- heterocycle, wherein the heterocycle portion of -L-heterocycle is optionally substituted with one or more R6. In some cases, the heterocycle is a saturated heterocycle. In some cases, the heterocycle has at least one nitrogen atom and at least one sulfur atom. In some cases, the heterocycle has at least one nitrogen atom. In some cases, the heterocycle has at least one sulfur atom.
[00149] In some embodiments, for a compound or salt of Formula (I), R2 is selected from
, , wherein the heterocycle portion is optionally substituted with one or more R6.
[00150] In some embodiments, for a compound or salt of Formula (I), Y-R2 is selected from
, wherein the heterocycle portion is optionally substituted with one or more R6.
[00151] In some embodiments, for a compound or salt of Formula (I), Y-R2 is selected from
, wherein the heterocycle portion is optionally substituted with one or more R6.
[00152] In some embodiments, for a compound or salt of Formula (I), Y-R2 is selected from
, wherein the heterocycle portion is optionally substituted with one or more
R6.
[00153] In some embodiments, for a compound or salt of Formula (I), R2 is selected from -L- saturated heterocycle, wherein the saturated heterocycle portion of the -L-saturated heterocycle is optionally substituted with one or more R6, and contains one nitrogen atom and one sulfur atom.
In some cases, Y-R2 is selected from
wherein the heterocycle portion is optionally substituted with one or more R6. In some cases, Y-R2 is selected from
, wherein the heterocycle portion is optionally substituted with one or more substituents selected from C1-C3 alkyl and oxo. In some cases, Y-R2 is selected from
cases, Y-R2 is selected from
[00154] In some embodiments, for a compound or salt of Formula (I), Y is a bond. In some cases, R2 is selected from an optionally substituted heteroaryl and optionally substituted aryl. In some cases, R2 is selected from an optionally substituted heteroaryl. In some cases, the heteroaryl has at least one nitrogen atom. In some cases, the heteroaryl has at least two nitrogen atoms. In some cases, the heteroaryl only contain nitrogen atom(s). In some cases, the heteroaryl is a 6- membered heteroaryl. In some cases, the heteroaryl is a 5-membered heteroaryl. In some cases, the heteroaryl is selected from
each of which is optionally substituted.
In some cases, the heteroaryl is selected from
each of which is optionally substituted. In some cases, R2 is selected from an optionally substituted aryl. In some cases, the aryl is a phenyl. In some cases, the heteroaryl is optionally substituted with one or more
R6, wherein each R6 is selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, -CHJieterocycle, -C1-C3 alkyl-N(R5)2, and -C(O)N(R5)2. In some cases, R6 is selected from C1-C3 alkyl, -CH2heterocycle, and -C(O)N(R5)2. In some cases, the aryl is optionally substituted with one or more R7. In some cases, Y-R2 is selected from
[00155] In some embodiments, for a compound or salt of Formula (I), Y is -O- and R2 is selected from L-5-membered heteroaryl. In some cases, the heteroaryl has at least 1 nitrogen atom. In some cases, the heteroaryl has at least two nitrogen atoms. In some cases, the heteroaryl has 3 nitrogen atoms. In some cases, L is selected from an optionally substituted C1-C4 alkylene. In some cases, L is independently selected from a C1-C4 alkylene optionally substituted; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents selected from halogen,
-OH, -NO2, =0, =S, -CN, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl. In some cases, L is selected from
In some cases, L is selected from
. In some cases, the heteroaryl is optionally substituted with one or more R7. In some cases, each R7 is selected from halogen, C1-C4 alkyl, and C1-C4 haloalkyl. In some cases, Y-R2 is selected from
[00156] In some embodiments, for a compound or salt of Formula (I), each R6 is independently selected from halogen, -OH, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, -CN, C1-C3 aminoalkyl, -Q-phenyl, -Q-phenylSO2F, -NHC(O)phenyl, -NHC(O)phenylSO2F, C1-C3 alkyl substituted pyrazolyl, -N(R5)2, (C1-C3 alkoxy)Ci-C3 alkyl-, (C1-C3 alkyl)C(=O), oxo, (C1-C3 haloalkyl)C(=O)-, -SO2F, (C1-C3 alkoxy)Ci-C3 alkoxy, -CH2OC(O)N(R5)2, -CH2NHC(O)OCI-C6 alkyl, -CH2NHC(O)N(R5)2, -CH2NHC(O)CI-C6 alkyl, -CH2(pyrazolyl), -CH2NHSO2C1-C6 alkyl, -CH2OC(O)heterocycle, -OC(O)N(R5)2, -OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl), -OC(O)NH(Ci- C3 alkyl)O(Ci-C3 alkyl)phenyl(Ci-C3 alkyl)N(CH3)2, -OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl)phenyl, -OC(O)heterocycle, and -CH2heterocycle, wherein the phenyl of -NHC(O)phenyl and -OC(O)NH(CI-C3 alkyl)(Ci-C3 alkyl)phenyl are each optionally substituted with -C(O)H and OH, and wherein the heterocycle of -CH2heterocyclyl is optionally substituted with oxo.
[00157] In some embodiments, for a compound or salt of Formula (I), each R6 is independently selected from halogen, -OH, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, -CN, and C1-C3 aminoalkyl. In some cases, each R6 is independently selected from halogen, C1-C3 alkyl, and C1-C3 haloalkyl.
[00158] In some embodiments, for a compound or salt of Formula (I), each R6 is independently selected from halogen, -OH, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 aminoalkyl, C1-C3 haloalkyl, C1-C3 alkoxy, -N(R5)2, and oxo. In some cases, each R6 is independently selected from -OH, Ci- C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 aminoalkyl, C1-C3 alkoxy, and -N(R5)2. In some cases, each R6 is independently selected from C1-C3 alkyl, C1-C3 alkoxy, and -N(R5)2. In some cases, each R6 is independently selected from halogen, hydroxy, C1-C3 alkyl, C1-C3 haloalkyl, -N(R5)S(O)2(R5), - OC(O)N(R5)2, =CH2, OXO, =NO-CI-C3 alkyl, -CH2OC(O)heterocycle, -CH2heterocycle, - CH2OC(O)N(R5)2, and-O-Ci-Cs alkyl, wherein the alkyl of -O-C1-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo, and hydroxy; and wherein each R7 is selected from C1-C3 alkyl, halogen and C1-C3 haloalkyl.
[00159] In some embodiments, for a compound or salt of Formula (I), R6 is selected from halogen, -OH, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, -CN, and C1-C3 aminoalkyl. In some cases, R6 is selected from halogen and C1-C3 alkyl. In some cases, R6 is
halogen. In some cases, R6 is C1-C3 alkyl. In some cases, R6 is selected from halogen and C1-C3 alkyl. In some cases, R6 is selected from methyl and fluorine.
[00164] In some embodiments, for a compound or salt of Formula (I), L is selected from unsubstituted C1-C4 alkylene. some embodiments, for a compound or salt of Formula (I), Y-R2 is selected from
, wherein the heterocycle portion is optionally substituted with one or more R6.
[00166] In some embodiments, for a compound or salt of Formula (I), R6 of R2 is independently selected at each occurrence from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, and C1-C3 aminoalkyl.
[00167] In some embodiments, for a compound or salt of Formula (I), R6 of R2 is independently selected at each occurrence from C1-C3 alkyl and halogen.
[00169] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally sub stituted 5- to 12-membered heterocycle. In some cases, the heterocycle of R1 is
selected from a 5- to 12-membered heterocycle, 6- to 12-membered heterocycle, 7- to 12- membered heterocycle, and 8- to 12-membered heterocycle. In some cases, the heterocycle of R1 is selected from a 5- to 11 -membered heterocycle, 5- to 10-membered heterocycle, 5- to 9- membered heterocycle, and 5- to 8-membered heterocycle. In some cases, the heterocycle of R1 is selected from a 6- to 11 -membered heterocycle, 6- to 10-membered heterocycle, 6- to 9- membered heterocycle, and 6- to 8-membered heterocycle. In some cases, the heterocycle of R1 is selected from a 7- to 11 -membered heterocycle, 7- to 10-membered heterocycle, 7- to 9- membered heterocycle, and 7- to 8-membered heterocycle. In some cases, the heterocycle of R1 is selected from a 5- to 6-membered heterocycle and 5- to 9-membered heterocycle. In some cases, the heterocycle of R1 is selected from an 8- to 9-membered heterocycle. In some cases, R1 is selected from an optionally substituted 5- to 7-membered heterocycle. In some cases, R1 is selected from an optionally substituted 6- to 7-membered heterocycle. In some cases, R1 is selected from an optionally substituted 7-membered heterocycle. In cases, the 5- to 12- membered heterocycle of R1 is a bridged heterocycle. In cases, the 5- to 12-membered heterocycle of R1 is not a bridged heterocycle. In some cases, the heterocycle of R1 is saturated. In some cases, the heterocycle of R1 is unsaturated. In some cases, the heterocycle of R1 is an unbridged heterocycle. The heterocycle of R1 is optionally substituted as described elsewhere herein.
[00170] In some embodiments, for a compound or salt of Formula (I), the heterocycle of R1 contains at most 1 nitrogen atom. In some embodiments, the heterocycle of R1 contains at most 1 heteroatom atom. In some cases, the heteroatom is selected from nitrogen, oxygen, and sulfur. In some embodiments, the heterocycle of R1 contains at most 2 heteroatom atoms. In some cases, the heterocycle of R1 contains 1 nitrogen atom. In some cases, the heterocycle of R1 contains only 1 nitrogen atom. In some cases, the heterocycle of R1 contains only 1 nitrogen atom and no other heteroatoms.
[00171] In some embodiments, for a compound or salt of Formula (I), R1 is selected from 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -OH, -N(R20)2, - NO2, =0, -CN, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and Ci-e haloalkyl. In some cases, the 5- to 12-membered heterocycle of R1 is an unsaturated heterocycle. In some cases, the 5- to 12-membered heterocycle of R1 is selected from a saturated heterocycle and unsaturated heterocycle. In some cases, the 5- to 12-membered heterocycle of R1 is a saturated heterocycle. In some cases, the 5- to 12-membered heterocycle of R1 is an unsaturated heterocycle.
[00172] In some embodiments, for a compound or salt of Formula (I), R1 is selected from 5- to 15-membered heterocycle, wherein the 5- to 15-membered heterocycle is optionally
substituted. In some cases, R1 is selected from an optionally substituted 6- to 15-membered heterocycle. In some cases, the heterocycle is a spiro heterocycle. In some cases, the heterocycle is a fused heterocycle. In some cases, the heterocycle is a bridged heterocycle. In some cases, the heterocycle is an unsaturated heterocycle.
[00173] In some embodiments, for a compound or salt of Formula (I), R1 is a 6- to 12- membered fused heterocycle, which is optionally substituted.
[00174] In some embodiments, for a compound or salt of Formula (I), R1 is a 6- to 12- membered spiroheterocycle, which is optionally substituted. In some cases, R1 is selected from optionally substituted 7- to 8-membered spiroheterocycle.
[00175] In some embodiments, for a compound or salt of Formula (I), the heterocycle of R1 is a 5- to 12-membered heterocycle, 6- to 12-membered heterocycle, 7- to 12-membered heterocycle, or 8- to 12-membered heterocycle. In some cases, the heterocycle of R1 is a 5- to 11 -membered heterocycle, 5- to 10-membered heterocycle, 5- to 9-membered heterocycle, or 5- to 8-membered heterocycle. In some cases, the heterocycle of R1 is a 6- to 11 -membered heterocycle, 6- to 10-membered heterocycle, 6- to 9-membered heterocycle, or 6- to 8-membered heterocycle. In some cases, the heterocycle of R1 is a 7- to 11-membered heterocycle, 7- to 10- membered heterocycle, 7- to 9-membered heterocycle, or 7- to 8-membered heterocycle. In some cases, the heterocycle of R1 is a 5- to 6-membered heterocycle or 5- to 9-membered heterocycle. In some cases, the heterocycle of R1 is an 8- to 9-membered heterocycle. In some cases, the heterocycle of R1 is saturated. The heterocycle is optionally substituted as described elsewhere herein.
[00176] In some embodiments, for a compound or salt of Formula (I), R1 is a 5- to 12- membered monocyclic heterocycle. In some cases, the heterocycle of R1 is a 5- to 12-membered monocyclic heterocycle, 6- to 12-membered monocyclic heterocycle, 7- to 12-membered monocyclic heterocycle, or 8- to 12-membered monocyclic heterocycle. In some cases, the heterocycle of R1 is a 5- to 11 -membered monocyclic heterocycle, 5- to 10-membered monocyclic heterocycle, 5- to 9-membered monocyclic heterocycle, or 5- to 8-membered monocyclic heterocycle. In some cases, the heterocycle of R1 is a 6- to 11 -membered monocyclic heterocycle, 6- to 10-membered monocyclic heterocycle, 6- to 9-membered monocyclic heterocycle, or 6- to 8-membered monocyclic heterocycle. In some cases, the heterocycle of R1 is a monocyclic 7- to 11 -membered heterocycle, 7- to 10-membered monocyclic heterocycle, 7- to 9-membered monocyclic heterocycle, or 7- to 8-membered monocyclic heterocycle. In some cases, the heterocycle of R1 is a 5- to 6-membered monocyclic heterocycle or 5- to 9-membered monocyclic heterocycle. In some cases, the heterocycle of R1 is an 8- to 9-membered monocyclic
heterocycle. In some cases, the heterocycle of R1 is saturated. The monocyclic heterocycle is optionally substituted as described elsewhere herein.
[00177] In some embodiments, for a compound or salt of Formula (I), R1 is a bridged heterocycle. In some cases, the heterocycle of R1 is a 5- to 12-membered bridged heterocycle, 6- to 12-membered bridged heterocycle, 7- to 12-membered bridged heterocycle, or 8- to 12- membered bridged heterocycle. In some cases, the heterocycle of R1 is a 5- to 11 -membered bridged heterocycle, 5- to 10-membered bridged heterocycle, 5- to 9-membered bridged heterocycle, or 5- to 8-membered bridged heterocycle. In some cases, the heterocycle of R1 is a 6- to 11 -membered bridged heterocycle, 6- to 10-membered bridged heterocycle, 6- to 9- membered bridged heterocycle, or 6- to 8-membered bridged heterocycle. In some cases, the heterocycle of R1 is a bridged 7- to 11 -membered heterocycle, 7- to 10-membered bridged heterocycle, 7- to 9-membered bridged heterocycle, or 7- to 8-membered bridged heterocycle. In some cases, the heterocycle of R1 is a 5- to 6-membered bridged heterocycle or 5- to 9- membered bridged heterocycle. In some cases, the heterocycle of R1 is an 8- to 9-membered bridged heterocycle. In some cases, the heterocycle of R1 is saturated. In some cases, the bridged heterocycle is selected from
In some cases, the bridged heterocycle is selected from
. Each bridged heterocycle is optionally substituted as described elsewhere herein.
[00178] In some embodiments, for a compound or salt of Formula (I), R1 is a spiro heterocycle. The spiro heterocycle of R1 is a 7- to 12-membered spiro heterocycle, 7- to 12- membered spiro heterocycle, or 8- to 12-membered spiro heterocycle. In some cases, the spiro heterocycle of R1 is a 7- to 11 -membered spiro heterocycle, 7- to 10-membered spiro heterocycle, 7- to 9-membered spiro heterocycle, or 7- to 8-membered spiro heterocycle. In some cases, the spiro heterocycle of R1 is a 7- to 11 -membered spiro heterocycle, 7- to 10-membered spiro heterocycle, 7- to 9-membered spiro heterocycle, or 7- to 8-membered spiro heterocycle. In some cases, the spiro heterocycle of R1 is a 7- to 11 -membered spiro heterocycle. In some cases, the spiro heterocycle of R1 is a 7-membered spiro heterocycle. In some cases, the spiro heterocycle of R1 is an 8-membered spiro heterocycle. In some cases, the spiro heterocycle of R1 is a 9-membered spiro heterocycle. In some cases, the spiro heterocycle of R1 is a 10-membered spiro heterocycle. In some cases, the spiro heterocycle of R1 contains at most 1 nitrogen atom. In some cases, the spiro heterocycle of R1 contains only 1 nitrogen atom. In some cases, the spiroheterocycle of R1 contains at most 2 heteroatom atoms. In some cases, the spiro heterocycle
of R1 contains at least 2 heteroatom atoms. In some cases, the spiro heterocycle of R1 contains at least 3 heteroatom atoms. In some cases, the heteroatom is selected from nitrogen, oxygen, and sulfur. In some cases, the spiroheterocycle of R1 is bound to the Formula via the nitrogen atom.
heterocycle of R1 is selected from
. Each spiro heterocycle is optionally substituted as described elsewhere herein.
[00179] In some embodiments, for a compound or salt of Formula (I), R1 is a fused heterocycle. In some cases, the fused heterocycle of R1 is a 6- to 12-membered fused heterocycle, 6- to 12-membered fused heterocycle, 7- to 12-membered fused heterocycle, or 8- to 12-membered fused heterocycle. In some cases, the fused heterocycle of R1 is a 6- to 11- membered fused heterocycle, 6- to 10-membered fused heterocycle, 6- to 9-membered fused heterocycle, or 6- to 8-membered fused heterocycle. In some cases, the fused heterocycle of R1 is a 7- to 11 -membered fused heterocycle, 7- to 10-membered fused heterocycle, 7- to 9-membered fused heterocycle, or 7- to 8-membered fused heterocycle. In some cases, the fused heterocycle of R1 is an 8- to 11-membered fused heterocycle. In some cases, the fused heterocycle of R1 is a 9-membered fused heterocycle. In some cases, the fused heterocycle of R1 is a 10-membered fused heterocycle. In some cases, the fused heterocycle of R1 is an 11 -membered fused heterocycle. In some cases, the fused heterocycle of R1 is a 6-membered fused heterocycle. In some cases, the fused heterocycle of R1 is a 7-membered fused heterocycle. In some cases, the fused heterocycle of R1 is a 10-membered fused heterocycle. In some cases, the fused
heterocycle is selected from
Each fused heterocycle is optionally substituted as described elsewhere herein.
[00180] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 8- to 10-membered fused heterocycle. In some cases, the 8- to 10- membered fused heterocycle is a bicyclic heterocycle. In some cases, the 8- to 10-membered fused heterocycle is a saturated heterocycle. In some cases, the 8- to 10-membered fused heterocycle is an unsaturated heterocycle. In some cases, the 8- to 10-membered heterocycle is a non-aromatic heterocycle. In some cases, R1 is selected from an optionally substituted 9- membered fused heterocycle. In some cases, R1 is selected from an optionally substituted 10- membered fused heterocycle. In some cases, the 10-membered fused heterocycle is a bicyclic heterocycle. In some cases, the 10-membered fused heterocycle is a saturated heterocycle. In some cases, the 9-membered heterocycle is a non-aromatic heterocycle. In some cases, the 10- membered heterocycle is a non-aromatic heterocycle. In some cases, the fused heterocycle has one saturated ring and one aromatic ring. In some cases, the fused heterocycle has one saturated ring and one unsaturated ring. In some cases, the fused heterocycle has two saturated rings. In some cases, the 10-membered heterocycle contains at least 1 nitrogen atom. In some cases, the 10-membered heterocycle contains at least 2 nitrogen atoms. In some cases, the 10-membered heterocycle contains at least 3 nitrogen atoms. In some cases, the 9-membered heterocycle contains at least 1 nitrogen atom. In some cases, the 9-membered heterocycle contains at least 2 nitrogen atoms. In some cases, the 9-membered heterocycle contains at least 3 nitrogen atoms. In some cases, R1 is selected from
, each of which is optionally substituted with one or more substituents. In some cases,
which is optionally substituted
with one or more substituents. In some cases,
, which is optionally substituted with one or more substituents. In some cases, the one or more optional substituents of R1 are independently selected from halogen, -OH, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -C(=NR20)N(R20)2, -C(O)N(R20)2, -C(O)NHOR20, -N(R20)2, -C(O)R20, -NO2,
=0, -CN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, Ci-
6 haloalkyl, C1-6 alkyl, C2-6 alkynyl, and 5- to 12-membered heterocycle, wherein the 5- to 12- membered heterocycle are each optionally substituted independently with one or more R1*. In some cases, the one or more optional substituents of R1 are independently selected from halogen, -OH, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -C(O)N(R20)2, -C(O)NHOR20, -N(R20)2, -C(O)R20, -NO2, =0, -CN, C1-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 hydroxyalkyl, C 1-6 cyanoalkyl, C 1-6 haloalkyl, Ci-6 alkyl, and C2-6 alkynyl. In some cases, the optional one or more substituents are independently selected from halogen, =0, -OH, -CN, - NHCN, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -C(O)R20, -C(O)N(R20)2, - C(O)NR20OR20, CI-6 alkyl -N(R20)2, CI-6 aminoalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, and Ci-6 alkyl. In some cases, the optional one or more substituents are independently selected from halogen, =0, Ci-6 alkyl-N(R20)2, -S(O)2(R20), -S(O)N(R20)2, -S(O)R20(=NR20), - C(O)R20, -C(O)N(R20)2, and -C(O)NR20OR20. In some cases, the optional one or more substituents are independently selected from halogen, =0, -S(O)2(R20), -S(O)N(R20)2, - S(O)R20(=NR20), -C(O)R20, -C(O)N(R20)2, and -C(O)NR20OR20. In some cases, the optional one or more substituents are independently selected from -C(O)R20, -C(O)N(R20)2, and - C(O)NR20OR20. In some cases, the optional one or more substituents are independently selected from -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20). In some cases, the optional one or more substituents are independently selected from -S(O)N(R20)2. In some cases, the optional one or more substituents are independently selected from S(O)2(R20). In some cases, the optional one or more substituents are independently selected from S(O)R20(=NR20). In some cases, the optional one or more substituents are independently selected from -C(O)R20. In some cases, the optional one or more substituents are independently selected from -C(O)N(R20)2. In some cases, the optional one or more substituents are independently selected from -
each of which is further optionally substituted. In some cases, the further one or more optional substituents are selected from halogen, -OH, =0, -CN, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, Ci-e haloalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the further one or more optional substituents are selected from halogen, -CN, C2 alkenyl, and C1-6 alkyl. In some cases, the further one or more optional substituents are selected from halogen, and C1-6 alkyl. In some cases, the further one or more optional substituents are selected from halogen. In some cases, each R20 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle. In some cases, each R20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12- membered heterocycle. In some cases, each R20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12-membered saturated heterocycle. In some cases, each R20 is independently selected from 5- to 6-membered saturated heterocycle. In some cases, the heterocycle of R20 has at least one nitrogen atom. In some cases, the heterocycle of R20 has at least one sulfur atom. In some cases, the heterocycle of R20 has at least one oxygen atom. In some cases, the heterocycle of R20 contains only 1 heteroatom. In some cases, the heterocycle of R20 has at least two heteroatoms. In some cases, the heterocycle of R20 contains only 2 heteroatoms. In some cases, the optional one or more substituents of R1 are independently selected from halogen, -CN, C2 alkenyl,
some cases, the optional one or more substituents of R1 are independently selected from halogen,
more substituents of R1 are independently selected from halogen, and Ci-6 alkyl-N(R20)2. In some cases, the optional one or more substituents of R1 are independently selected from halogen,
, hydrogen, Ci-6 alkyl, and C3-6 carbocycle. In some cases, R1 is selected
some cases, R1 is selected
independently selected from halogen, -OH, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -C(O)N(R20)2, -C(=NR20)N(R20)2, -C(O)OR20, -C(O)NHOR20, -N(R20)2, - C(O)R20, -NO2, =0, -CN, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, C1-6 alkyl, C2-e alkynyl, 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted with one or more substituents selected from halogen, -OR20, and C1-6 alkyl. In some cases, R1 is selected which is optionally substituted with one more substituents independently
selected from halogen and Ci-6 alkyl. In some cases, R1 is selected from
[00181] In some embodiments, for a compound or salt of Formula (I), R1 is selected from a
( B )
, wherein is selected from a 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted independently with one or more R1* ; and RB is selected from hydrogen, halogen, Ci-6 alkyl, Ci-6 haloalkyl, C2-6 alkynyl, and -CN. In some cases, RB is selected from hydrogen, and halogen. In some cases, RB is chloride. In some cases, RB is hydrogen. In some cases,
has at least 1, 2, 3, or 4 heteroatoms. In some cases, 2, 3, or 4 nitrogen atoms. In some cases,
has at least 1 oxygen atom.
, is a monocyclic heterocycle. In some cases,
is a bicyclic heterocycle. In some cases,
is selected from an optionally substituted 5-membered heterocycle. In some cases,
is selected from an optionally substituted 9-membered heterocycle. In some cases,
is selected from
, each of which is optionally substituted with one or more R1*.
each of which is optionally substituted with one or more R1*. In some cases, each R1* is independently selected from halogen, -OR20, -S(O)2(R20), -
S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(O)NR20OR20, -
N(R20)C(O)R20, -N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, -C(O)R20, -C(O)OR20, -
OC(O)R20, -OC(O)N(R20)2, -NO2, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl-N(R20)2, Ci-
6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, and Ci-6 alkyl. In some cases, each R1* is independently selected from halogen, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, and Ci-6 alkyl. In some cases, each R1* is independently selected from halogen, and Ci-6 alkyl. In some cases,
is selected from
[00182] In some embodiments, for a compound or salt of Formula (I), when R1 is substituted with -C(O)R20, R20 is selected from a 5- to 12-membered heterocycle, which is optionally substituted. In some cases, R1 is substituted with -C(O)R20. In some cases, R20 is selected from a 5- to 12-membered unsubstituted heterocycle. In some cases, R20 is selected from a 5- to 6- membered heterocycle, which is optionally substituted. In some cases, the heterocycle has at least one nitrogen atom. In some cases, the heterocycle has at least one sulfur atom. In some cases, the heterocycle has at least one oxygen atom. In some cases, the heterocycle has two heteroatoms. In some cases, the heterocycle of R20 is selected from
and
, each of which is optionally substituted. In some cases, R20 is selected from
some cases, the optional substituents are selected from Ci-io alkyl, oxo, and =NH.
[00183] In some embodiments, for a compound or salt of Formula (I), each R20 is independently selected from hydrogen; and Ci-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O- C1-10 alkyl, oxo, and =NH. In some cases, each R20 is independently selected from hydrogen; and unsubstituted C1-6 alkyl, and 3- to 12-membered heterocycle which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, and =NH.
[00184] In some embodiments, for a compound or salt of Formula (I), each R21 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O- C1-10 alkyl, oxo, and =NH. In some cases, each R21 is independently selected from hydrogen; and unsubstituted C1-6 alkyl, and 3- to 12-membered heterocycle which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, and =NH.
[00185] In some embodiments, for a compound or salt of Formula (I), each R20 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O- C1-10 alkyl, oxo, and =NH. In some cases, each R20 is independently selected from hydrogen; and unsubstituted C1-6 alkyl, and 3- to 12-membered heterocycle which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, and =NH.
[00186] In some embodiments, for a compound or salt of Formula (I), each R23 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O- C1-10 alkyl, oxo, and =NH. In some cases, each R20 is independently selected from hydrogen; and unsubstituted C1-6 alkyl, and 3- to 12-membered heterocycle which is optionally substituted with
one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-io alkyl, -Ci-io haloalkyl, -O-Ci-io alkyl, oxo, and =NH.
[00187] In some embodiments, for a compound or salt for Formula (I), R1 is selected from 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted with one or more substituents. In some cases, the one or more optional substituents are independently selected from halogen, -CN, -NO2, =0, -N(R20)2, -B(OR20)2, -OR20, -SR20, - S(O)2(R20), -S(O)2N(R20)2, -NR20S(O)2R20, -C(O)N(R20)2, -N(R20)C(O)R20, -N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, -C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, C1-6 aminoalkyl, Ci-6 alkoxy, C1-6 alkoxyalkyl, Ci-e hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, C1-6 alkyl, C2-6 alkenyl, and C2-6 alkynyl. In some cases, the one or more optional substituents are independently selected from halogen, -OH, -N(R20)2, -NO2, =0, -CN, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and C1-6 haloalkyl. In some cases, the one or more optional substituents are independently selected from halogen, -OH, -N(R20)2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and C1-6 haloalkyl. In some cases, R20 is selected from hydrogen and C1-3 alkyl.
[00188] In some embodiments, for a compound or salt for Formula (I), R1 is selected from a saturated 5- to 12-membered heterocycle, which is optionally substituted with one or more substituents. In some cases, the 5- to 12-membered heterocycle of R1 is bridged. In some cases, the 5- to 12-membered heterocycle of R1 is not bridged. In some cases, the 5- to 12-membered
substituents.
[00189] In some embodiments, for a compound or salt of Formula (I), R1 is selected from
, each of which is optionally substituted with one or more substituents. In some cases, the one or more of the optional substituents are independently selected from halogen, -OH, - N(R20)2, -B(OH)2, -C(O)N(R20)2, -NHCN, -NO2, CI-6 alkoxy, =0, -CN, Ci-6 alkyl, C2.6 alkenyl, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 hydroxyalkyl, and Ci-e haloalkyl. In some cases, R1 is
[00190] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 5- to 12-membered unsaturated heterocycle, wherein the heterocycle has as most one nitrogen atom. In some cases, the 5- to 12-membered unsaturated heterocycle has at least one nitrogen atom. In some cases, the 5- to 12-membered unsaturated heterocycle has at most one nitrogen atom.
[00191] In some embodiments, for a compound or salt of Formula (I), the heterocycle of R1 contains only 1 nitrogen atom and optionally one or more heteroatoms selected from oxygen, and sulfur. In some cases, the heterocycle is a fused heterocycle or a bridged heterocycle. In some cases, the heterocycle is a monocyclic heterocycle or a bridged heterocycle. In some cases, the heterocycle is a monocyclic heterocycle. In some cases, the heterocycle is a bridged heterocycle.
In some cases, the heterocycle is selected from
heter0CyCie js optionally substituted as described elsewhere herein.
[00192] In some embodiments, for a compound or salt of Formula (I), the heterocycle of R1 has at most 1 nitrogen atom. In some cases, the heterocycle of R1 has only 1 nitrogen atom and optionally one or more other heteroatoms selected from oxygen and sulfur. In some cases, the heterocycle of R1 has only 1 nitrogen atom and no other heteroatoms.
[00193] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 5- to 12-membered saturated heterocycle, wherein the heterocycle has as most one nitrogen atom. In some cases, the 5- to 12-membered unsaturated heterocycle has at least one nitrogen atom. In some cases, the 5- to 12-membered unsaturated heterocycle has only one nitrogen atom and 0-2 other heteroatoms selected from nitrogen, oxygen, and sulfur. In some cases, the 5- to 12-membered unsaturated heterocycle has only one nitrogen atom and no further heteroatoms. In some cases, the 5- to 12-membered unsaturated heterocycle has three nitrogen atoms and no further heteroatoms.
[00194] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 5- to 12-membered unsaturated heterocycle, wherein the heterocycle has as most one nitrogen atom. In some cases, the 5- to 12-membered unsaturated heterocycle has at least one nitrogen atom. In some cases, the 5- to 12-membered unsaturated heterocycle has only one nitrogen atom and no further heteroatoms.
[00195] In some embodiments, for a compound or salt of Formula (I), R1 is selected from 6- to 7-membered heterocycle. In some cases, R1 is selected from 7-membered heterocycle. In some cases, R1 is selected from 6-membered heterocycle. In some cases, the 6- to 7-membered heterocycle contains only 1 nitrogen atom and optionally one or more additional heteroatoms selected from oxygen, and sulfur. In some cases, the optionally one or more additional heteroatoms are selected from sulfur. In some cases, the optionally one or more additional heteroatoms are selected from oxygen. In some cases, the 6- to 7-membered heterocycle contains only 1 nitrogen atom and no further additional heteroatoms. In some cases, the 6- to 7-membered heterocycle is a non-aromatic 6- to 7-membered heterocycle. In some cases, the 6- to 7-membered heterocycle of
R1 is bound to Formula (I) via the only 1 nitrogen atom. In some cases, R1 is selected
which is substituted. In some cases, the substituents of R1 are each selected from one or more halogen, -OR20, -SR20, -N(R20)2, -NHCN, -NO2, =0, -CN, C 1-6 fluoroalkyl, and C2-6 alkynyl; and further optionally substituted with one or more substituents independently selected from -C(O)N(R20)2, C1-6 aminoalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkenyl. In some cases, the substituents of R1 are each selected from one or more halogen, -OR20, -N(R20)2, -NHCN, =0, -CN, and C2-6 alkynyl; and further optionally substituted with one or more substituents independently selected from -C(O)N(R20)2, C1-6 aminoalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, and C1-6 alkyl. In some cases, the substituents of R1 are each selected from one or more halogen, -OH, -NHCN, =0, -CN, and C2-6 alkynyl; and further optionally substituted with one or more substituents independently selected from C1-6 alkyl. In some cases, R1 is selected from
selected from
each of which is optionally substituted. In some cases, the one or more optional substituents of R1 are each independently selected from fluorine, -OH, -C(0)NH2, -NH-C(0)-(CI-6 alkoxy), -NH-C(0)-(CI-6 hydroxyalkyl), -NH2, - NH(CN), =0, -CN, Ci-e hydroxyalkyl, C1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of R1 are each independently selected from halogen, - OH, -CN, C1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of R1 are each independently selected from halogen, -OH, and -CN. In some cases, the one or more optional substituents of R1 are each independently selected from fluorine, -
OH, -CN, C1-6 cyanoalkyl, C1-6 alkyl, oxo, and C2-6 alkynyl. In some cases, the one or more optional
substituents of R1 are each independently selected from fluorine, -OH, -CN, C 1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, R1 is selected from
of which is optionally substituted with one or more substituents. In some cases, the one or more
of the optional substituents are independently selected from halogen, -OH, -N(R20)2, -B(0H)2, - C(O)N(R20)2, -NHCN, -NO2, C1-6 alkoxy, =0, -CN, C1-6 alkyl, C2-6 alkenyl, C1-6 aminoalkyl, C1-6 hydroxyalkyl, and Ci-e haloalkyl. In some cases, R1 is selected
[00197] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted unsaturated 6- to 8-membered heterocycle. In some cases, R1 is selected from an optionally substituted unsaturated 6-membered heterocycle. In some cases, R1 is selected from an optionally substituted unsaturated 7-membered heterocycle. In some cases, the heterocycle has 1 or 2 double bonds. In some cases, the heterocycle has only 1 double bond. In some cases, the heterocycle has only 2 double bonds. In some cases, R1 is selected from
wherein each is optionally substituted with one or more substituents independently selected from halogen, -OH, -NH2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl. In some cases, R1 is selected from
wherein each is optionally substituted with one or more substituents independently selected from halogen, -OH, -NH2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl. In some cases, R1 is selected from
more substituents independently selected from halogen, -OH, -NH2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl. In some cases, R1 is selected from
, wherein each is substituted with one or more substituents independently selected from halogen.
[00198] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an unsaturated 6- to 7-membered heterocycle, wherein the unsaturated 6- to 7-membered heterocycle is substituted with one or more substituents selected from halogen. In some cases, the unsaturated
6- to 7-membered heterocycle is substituted with at least one halogen. In some cases, the unsaturated 6- to 7-membered heterocycle is substituted with at only one halogen. In some cases, the unsaturated 7-membered heterocycle is substituted with one fluorine. In some cases, R1 is selected from an unsaturated 6-membered heterocycle, substituted with at least one halogen. In some cases, R1 is selected from an unsaturated 7-membered heterocycle, substituted with at least one halogen. In some cases, R1 is selected from
[00199] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted unsaturated 6- to 8-membered heterocycle. In some cases, R1 is selected from an optionally substituted unsaturated 7-membered heterocycle. In some cases, R1 is selected from
, wherein each is optionally substituted with one or more substituents independently selected from halogen, -OH, -NH2, -NO2, C 1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, Ci-e haloalkyl, and C1-6 alkyl. In some cases, R4S selected from
[00200] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 6-membered heterocycle. In some cases, the 6-membered heterocycle contains only 1 nitrogen atom. In some cases, the 6-membered heterocycle of R1 is bound to
Formula (I) via the only 1 nitrogen atom. In some cases, R1 is selected from of which is optionally substituted. In some cases, the one or more optional
each independently selected from halogen, -OR20, -N(R20)2, =0, -CN, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of R1 are each independently selected from fluorine, -OH, -NH2, -NH(CN), =0, -CN, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of R1 are each independently selected from fluorine, -OH, -NH2, -NH(CN), =0, -CN, C1-6 hydroxyalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the 6-membered heterocycle is a partially
unsaturated 6-membered heterocycle or a saturated 6-membered heterocycle. In some cases, the 6-membered heterocycle is partially unsaturated. In some cases, the 6-membered heterocycle is a saturated 6-membered heterocycle. In some cases, the 6-membered heterocycle is a monocyclic 6- membered heterocycle. In some cases, the 6-membered heterocycle is not a bridged heterocycle.
[00201] In some embodiments, for a compound of Formula (I), R1 is selected from an optionally substituted 6-membered unsaturated heterocycle and 6-membered saturated heterocycle.
[00202] In some embodiments, for a compound or salt of Formula (I), R1 is selected from
, wherein each is optionally substituted with one or more substituents independently selected from halogen, -OH, -NH2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl.
[00203] In some embodiments, for a compound or salt of Formula (I), R1 is selected from
, wherein each is optionally substituted with one or more substituents independently selected from halogen, and Ci-e haloalkyl.
[00204] In some embodiments, for a compound or salt of Formula (I), R1 is selected from
[00205] In some embodiments, for a compound or salt of Formula (I), R1 is selected from
, wherein each is optionally substituted two substituents independently selected from halogen, -OH, -NH2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl.
[00206] In some embodiments, for a compound or salt of Formula (I), R1 is selected from
, wherein each is optionally substituted with two substituents independently selected from halogen, and Ci-6 haloalkyl. In some cases,
[00207] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 6- to 10-membered heterocycle. In some cases, the 6- to 10-membered
heterocycle contains at least 1 nitrogen atom. In some cases, R1 is selected from “-I— ,
optionally substituted with one or more substituents independently selected from halogen, =0, - OH, -CN, -NHCN, -C(O)N(R20)2, C1-6 aminoalkyl, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, and C1-6 alkyl. In some cases, each R20 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle. In some cases, R1 is selected from
[00208] In some embodiments, for a compound or salt of Formula (I), R1 is selected from 6- to 7-membered heterocycle. In some cases, R1 is selected from 7-membered heterocycle. In some cases, R1 is selected from 6-membered heterocycle. In some cases, the 6- to 7-membered heterocycle contains only 1 nitrogen atom and optionally one or more additional heteroatoms selected from oxygen, and sulfur. In some cases, the optionally one or more additional heteroatoms are selected from sulfur. In some cases, the optionally one or more additional heteroatoms are selected from oxygen. In some cases, the 6- to 7-membered heterocycle contains only 1 nitrogen atom and no further additional heteroatoms. In some cases, the 6- to 7-membered heterocycle is a non-aromatic 6- to 7-membered heterocycle. In some cases, the 6- to 7-membered heterocycle of
R1 is bound to Formula (I) via the only 1 nitrogen atom. In some cases, R1 is selected from -™L~-
cases, R1 is selected from
each of which is substituted. In some cases, the substituents of R1 are each selected from one or more halogen, -OR20, -SR20, -N(R20)2, -NHCN, -NO2, =0, -CN, C 1-6 fluoroalkyl, and C2-6 alkynyl; and further optionally substituted with one or more substituents independently selected from -C(O)N(R20)2, CI-6 aminoalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 alkyl, and C2-6 alkenyl. In some cases, the substituents of R1 are each selected from one or more halogen, -OR20, -N(R20)2, -NHCN, =0, -CN, and C2-6 alkynyl; and further optionally substituted with one or more substituents independently selected from -C(O)N(R20)2, C1-6 aminoalkyl, C1-6 hydroxyalkyl, C1-6
cyanoalkyl, and C1-6 alkyl. In some cases, the substituents of R1 are each selected from one or more halogen, -OH, -NHCN, =0, -CN, and C2-6 alkynyl; and further optionally substituted with one or more substituents independently selected from C1-6 alkyl. In some cases, R1 is selected from
selected from
each of which is optionally substituted. In some cases, the one or more optional substituents of R1 are each independently selected from fluorine, -OH, -C(0)NH2, -NH-C(0)-(CI-6 alkoxy), -NH-C(0)-(CI-6 hydroxyalkyl), -NH2, -
NH(CN), =0, -CN, Ci-e hydroxyalkyl, C1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of R1 are each independently selected from halogen, - OH, -CN, C1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of R1 are each independently selected from halogen, -OH, and -CN. In some cases, the one or more optional substituents of R1 are each independently selected from fluorine, - OH, -CN, C1-6 cyanoalkyl, C1-6 alkyl, oxo, and C2-6 alkynyl. In some cases, the one or more optional substituents of R1 are each independently selected from fluorine, -OH, -CN, C 1-6 cyanoalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, R1 is selected from
[00209] In some embodiments, for a compound or salt of Formula (I), the 5- to 12-membered heterocycle of R1 is unsaturated and a bridged heterocycle. In some cases, R1 is selected from an optionally substituted 7- to 8-membered unsaturated and bridged heterocycle. In some cases, R1
[00210] In some embodiments, for a compound or salt of Formula (I), R1 is selected from 5- to 10-membered heterocycle, 7-, 8-, 10-, 11-membered spiro heterocycle, and 6-, 9-, 10-, 11-, and 12-membered fused heterocycle, and wherein each are optionally substituted with one or more substituents independently selected from halogen, -N(R20)2, Ci-6 alkyl, -OR20, - N(R20)C(O)N(R20)2, -B(OR20X CI-6 cyanoalkyl, -N(R20)C(O)N(R20)2, =0, Ci-6 hydroxyalkyl, halogen, -N(R20)C(O)R20, -N(R20) S(O)2(R20), and Ci-6 aminoalkyl. In some cases, R1 is selected
more substituents independently selected from halogen, -N(R20)2, Ci-6 alkyl, -OR20, - N(R20)C(O)N(R20)2, -B(OR20X CI-6 cyanoalkyl, -N(R20)C(O)N(R20)2, =0, Ci-6 hydroxyalkyl, halogen, -N(R20)C(O)R20, -N(R20) S(O)2(R20), and Ci-6 aminoalkyl. In some cases, R1 is selected
[00211] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 10-membered heterocycle. In some cases, the 10-membered heterocycle is a bicyclic heterocycle. In some cases, the 10-membered heterocycle is a spiro heterocycle. In some cases, the 10-membered heterocycle is a fused heterocycle. In some cases, the 10- membered heterocycle is a saturated heterocycle. In some cases, the 10-membered heterocycle is a non-aromatic heterocycle. In some cases, the 10-membered heterocycle contains at least 1 nitrogen atom. In some cases, the 10-membered heterocycle contains at least 2 nitrogen atoms. In some cases, the 10-membered heterocycle contains at least 3 nitrogen atoms. In some cases, the 10-membered heterocycle contains at least 1 sulfur atom. In some cases, R1 is selected from
more substituents independently selected from halogen, =0, -OH, -CN, -NHCN, -C(O)N(R20)2, - C(O)NR20OR20, C I-6 aminoalkyl, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, and Ci-6 alkyl. In some
some cases, R1 is selected from
, which is optionally substituted with one or more substituents independently selected from halogen, -OR20, -SR20, - N(R20)2, -NO2, =0, -CN, C 1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, C1-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, and C2-e alkynyl.
[00212] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted unsaturated 9- to 11 -membered heterocycle. In some cases, R1 is selected from an optionally substituted unsaturated 10-membered heterocycle. In some cases, R1 is selected from an optionally substituted unsaturated 10-membered fused heterocycle. In some cases,
, which is optionally substituted. In some cases, the one or more optional substituents are selected from halogen, -OH, -C(O)N(R20)2, -N(R20)2, -C(O)R20, -NO2, =O, -CN,
C1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C1-6 cyanoalkyl, C 1-6 haloalkyl, C1-6 alkyl, and C2-
6 alkynyl. In some cases,
optionally substituted with one or more substituents selected from -N(R20)2, -C(O)R20, -NO2, =0, -CN, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, C 1-6 haloalkyl, C1-6 alkyl, and C2-e alkynyl. In some cases, each R20 is independently selected from hydrogen; and C1-6 alkyl, and C3-12 carbocycle, and each of which is optionally substituted with one or more substituents independently selected from
halogen, -OH, -CN, -NO2, -NH2, C1-10 alkyl, -Ci-io haloalkyl, -O-Ci-10 alkyl, C2- 10 alkenyl, C2-10 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle.
[00213] In some embodiments, for a compound or salt of Formula (I), R1 is selected from a 7- to 11 -membered spiro heterocycle. In some cases, R1 is selected from a 10-membered spiro heterocycle. In some cases, the spiro heterocycle has at least 3 nitrogen atoms. In some cases, the spiro heterocycle has at least 1 sulfur atom. In some cases, R1 is selected from
, each of which is optionally substituted. In some cases, the one or more optional substituents are independently selected from halogen, -OH, -N(R20)2, -NO2, =0, -CN, -NHCN,
C1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C1-6 cyanoalkyl, Ci-e haloalkyl, C1-6 alkyl, C2-6 alkenyl, and C2-6 alkynyl. In some cases, R1 is selected from
some cases,
[00214] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 8- to 10-membered fused heterocycle. In some cases, the 8- to 10- membered fused heterocycle is a bicyclic heterocycle. In some cases, the 8- to 10-membered fused heterocycle is a saturated heterocycle. In some cases, the 8- to 10-membered fused heterocycle is an unsaturated heterocycle. In some cases, the 8- to 10-membered heterocycle is a non-aromatic heterocycle. In some cases, R1 is selected from an optionally substituted 10- membered fused heterocycle. In some cases, the 10-membered fused heterocycle is a bicyclic heterocycle. In some cases, the 10-membered fused heterocycle is a saturated heterocycle. In some cases, the 10-membered heterocycle is a non-aromatic heterocycle. In some cases, the fused heterocycle has one saturated ring and one aromatic ring. In some cases, the fused heterocycle has one saturated ring and one unsaturated ring. In some cases, the fused heterocycle has two saturated rings. In some cases, the 10-membered heterocycle contains at least 1 nitrogen atom. In some cases, the 10-membered heterocycle contains at least 2 nitrogen atoms. In some
cases, the 10-membered heterocycle contains at least 3 nitrogen atoms. In some cases, R1 is selected from
, each of which is optionally substituted with one or more substituents. In some cases, R1 is selected from
hich is optionally substituted with one or more substituents. In some
, , which is optionally substituted with one or more substituents. In some cases, the one or more optional substituents of R1 are independently selected from halogen, -OH,
-S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -C(O)N(R20)2, -C(O)NHOR20, - N(R20)2, -C(O)R20, -N02, =0, -CN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, and C2-6 alkynyl. In some cases, the optional one or more substituents are independently selected from halogen, =0, -OH, -CN, - NHCN, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -C(O)R20, -C(O)N(R20)2, - C(O)NR20OR20, C 1-6 alkyl -N(R20)2, CI-6 aminoalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, and Ci-6 alkyl. In some cases, the optional one or more substituents are independently selected from halogen, =0, Ci-6 alkyl-N(R20)2, -S(O)2(R20), -S(O)N(R20)2, -S(O)R20(=NR20), - C(O)R20, -C(O)N(R20)2, and -C(O)NR20OR20. In some cases, the optional one or more substituents are independently selected from halogen, =0, -S(O)2(R20), -S(O)N(R20)2, - S(O)R20(=NR20), -C(O)R20, -C(O)N(R20)2, and -C(O)NR20OR20. In some cases, the optional one or more substituents are independently selected from -C(O)R20, -C(O)N(R20)2, and - C(O)NR20OR20. In some cases, the optional one or more substituents are independently selected from -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20). In some cases, the optional one or more substituents are independently selected from -S(O)N(R20)2. In some cases, the optional one or more substituents are independently selected from S(O)2(R20). In some cases, the
optional one or more substituents are independently selected from S(O)R20(=NR20). In some cases, the optional one or more substituents are independently selected from -C(O)R20. In some cases, the optional one or more substituents are independently selected from -C(O)N(R20)2. In some cases, the optional one or more substituents are independently selected from -
C(O)NR20OR20. In some cases, R1 is selected from
, each of which is further optionally substituted. In some cases, the further one or more optional substituents are selected from halogen, -OH, =0, -CN, C 1-6 aminoalkyl, Ci-6 alkoxy, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, and C2-6 alkynyl. In some cases, the further one or more optional substituents are selected from halogen and C1-6 alkyl. In some cases, the further one or more optional substituents are selected from halogen. In some cases, each R20 is independently selected from hydrogen; and Cn 6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle. In some cases, each R20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12-membered heterocycle. In some cases, each R20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12- membered saturated heterocycle. In some cases, each R20 is independently selected from 5- to 6- membered saturated heterocycle. In some cases, the heterocycle of R20 has at least one nitrogen atom. In some cases, the heterocycle of R20 has at least one sulfur atom. In some cases, the heterocycle of R20 has at least one oxygen atom. In some cases, the heterocycle of R20 contains only 1 heteroatom. In some cases, the heterocycle of R20 has at least two heteroatoms. In some cases, the heterocycle of R20 contains only 2 heteroatoms. In some cases, the optional one or more substituents of R1 are independently selected from halogen,
. In some cases, the optional one or more substituents of R1 are independently
independently selected from halogen, and Ci-6 alkyl-N(R20)2. In some cases, the optional one or more substituents of R1 are independently selected from halogen,
, , and
. In some cases, R1 is selected from
. In some cases, each R20 is independently selected from hydrogen, Ci-6 alkyl, and C3-6 carbocycle. In some cases, R1 is
[00215] In some embodiments, for a compound or salt of Formula (I), when R1 is substituted with -C(O)R20, R20 is selected from a 5- to 12-membered heterocycle.
[00216] In some embodiments, for a compound or salt of Formula (I), Formula (I-A), or
Formula
the one or more optional substituents of R1 are independently selected from halogen, -OH, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -C(O)N(R20)2, -C(=NR20)N(R20)2, -C(O)NHOR20, -N(R20)2, -C(O)R20, -NO2, =0, -CN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-e alkynyl, and optionally substituted 5- to 12- membered heterocycle. In some cases, the one or more optional substituents of R1 are independently selected from halogen, -OH, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -C(O)N(R20)2, -C(=NR20)N(R20)2, -C(O)NHOR20, -N(R20)2, -C(O)R20, -NO2, =0, -CN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-e alkynyl, and optionally substituted 5- to 12- membered heterocycle. In some cases, the one or more optional substituents of R1 are
independently selected from halogen, -CN, C2-6 alkynyl, -C(=NR20)N(R20)2, and optionally substituted 5- to 12-membered heterocycle. In some cases, the one or more optional substituents of R1 are independently selected from halogen, -C(=NR20)N(R20)2, and optionally substituted 5- to 12-membered heterocycle. In some cases, the one or more optional substituents of R1 are independently selected from -C(=NR20)N(R20)2, and optionally substituted 5- to 12-membered heterocycle. In some cases, the one or more optional substituents of R1 are independently selected from optionally substituted 5- to 12-membered heterocycle. In some cases, the one or more optional substituents of R1 are independently selected from a 5-membered heterocycle and 9-membered heterocycle, each of which is optionally substituted independently with one or more R1*. In some cases, R1 is substituted with at least one halogen atom and optionally substituted with one or more substituents are independently selected from -CN, C2-6 alkynyl, - C(=NR20)N(R20)2, and 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted independently with one or more R1*. In some cases, the heterocycle has at least one nitrogen atom. In some cases, the heterocycle has at least oxygen atom. In some cases, the heterocycle has at least one nitrogen atom and at least one oxygen atom.
In some cases, heterocycle has at least two heteroatoms. In some cases, the heterocycle has at least three heteroatoms. In some cases, the heterocycle has at least four heteroatoms. In some cases, the heterocycle of the one or more optional substituents of R1 is selected from
, each of which is optionally substituted with one or more R1*. In some cases, the heterocycle of the one or more optional substituents of R1 is selected from
which is optionally substituted with one or more R1*. In some cases, each R1* is independently selected from halogen, -OR20, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -
NR20S(O)2R20, -C(O)N(R20)2, -C(O)NR20OR20, -N(R20)C(O)R20, -N(R20)C(O)N(R20)2, - N(R20)C(O)OR20, -N(R20)2, -C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, -NO2, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, and Ci-6 alkyl. In some cases, each R1* is independently selected from halogen, -OR20, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(O)NR20OR20, -N(R20)C(O)R20, -
N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, -C(O)R20, -C(O)OR20, -OC(O)R20, - OC(O)N(R20)2, -N02, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, and Ci-6 alkyl. In some cases, each R1* is independently selected from halogen, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, and Ci-6 alkyl. In some cases, each R1* is independently selected from halogen, Ci-6 haloalkyl, and Ci-6 alkyl. In some cases, each R1* is independently selected from halogen, and Ci-6 alkyl. In some cases, each R1* is independently selected from halogen. In some cases, each R1* is independently selected from Ci-6 alkyl. In some cases, each R1* is independently selected from -OR20. In some cases, each R1* is independently selected from -OH. In some cases, each R1* is independently selected from -OMe. In some cases,
[00217] In some embodiments, for a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), the one or more optional substituents of R1 are independently selected from - C(=NR20)N(R20)2, and optionally substituted 5- to 12-membered heterocycle. In some cases, the one or more optional substituents of R1 are independently selected from optionally substituted 5-
is optionally substituted with one or more R1*. In some cases, the one or more optional
[00218] In some embodiments, for a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), each R1* is independently selected from halogen, -OR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(O)NR20OR20, - N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, -C(O)R20, -C(O)OR20, - OC(O)R20, -OC(O)N(R20)2, -NO2, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl-N(R20)2, Ci-
6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, and Ci-6 alkyl. In some cases, each R1* is independently selected from halogen, -OR20, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(O)NR20OR20, - N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, -C(O)R20, -C(O)OR20, - OC(O)R20, -OC(O)N(R20)2, -NO2, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl-N(R20)2, Ci- 6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, C 1-6 haloalkyl, and C1-6 alkyl. In some cases, each R1* is independently selected from halogen, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, and C1-6 alkyl. In some cases, each R1* is independently selected from halogen, C1-6 haloalkyl, and C1-6 alkyl. In some cases, each R1* is independently selected from halogen, and C1-6 alkyl. In some cases, each R1* is independently selected from halogen. In some cases, each R1* is independently selected from C1-6 alkyl.
[00219] In some embodiments, for a compound or salt of Formula (I), R1 is selected from 5- to 15-membered heterocycle (preferably 8- to 10-membered heterocycle or preferably 10- membered heterocycle), each of which are optionally substituted with one or more substituents independently selected from halogen, oxo, -C(O)N(R20)2, -C(O)NR20OR20, -N(R20)2, -C(O)R20, - C(O)OR20, -SO2R20, -NHCN, C 1-6 cyanoalkyl, Ci-6 alkyl, Ci-6 alkyl-N(R20)2, C2-6 alkynyl, and 5- to 12-membered heterocycle (preferably 5- to 9-membered heterocycle), wherein the 5- to 12- membered heterocycle are each optionally substituted independently with one or more R1*; each R1* is independently selected from halogen, C1-6 haloalkyl, and C1-6 alkyl. In some cases, the 8- to 10-membered heterocycle is bicyclic. In some cases, the 10-membered heterocycle is substituted. In some cases, R1 is selected
, , , each of which
is optionally substituted. In some cases, R1 is selected -1- , which is optionally substituted. In
[00220] In some embodiments, for a compound or salt of Formula (I), R1 is selected from 5- to 15-membered heterocycle (preferably 8- to 10-membered heterocycle or preferably 10- membered heterocycle or preferably 8-membered heterocycle), each of which are optionally substituted with one or more substituents independently selected from halogen, -C(O)N(R20)2, - C(O)NR20OR20, -N(R20)2, -C(O)R20, -C(O)OR20, -NHCN, C I-6 cyanoalkyl, Ci-6 alkyl, C2-6 alkynyl, and 5- to 12-membered heterocycle (preferably 5- to 6-membered heterocycle), wherein the 5- to 12-membered heterocycle are each optionally substituted independently with one or
more R1*; each R1* is independently selected from halogen, Ci-6 haloalkyl, and Ci-6 alkyl. In some cases, the 8- to 10-membered heterocycle is bicyclic. In some cases, the 10-membered
heterocycle is substituted. In some cases, R is selected -1- , ~L , and -1- , each of which is
optionally substituted. In some cases, R is selected -1- , which is optionally substituted. In
[00222] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 7- to 10-membered spiro heterocycle and optionally substituted 7- to 10- membered fused heterocycle. In some cases, the heterocycle of R1 has at least one nitrogen atom In some cases, the at least one nitrogen at of the heterocycle of R1 is bound to Formula (I). In some cases, R1 is selected from an optionally substituted 10-membered spiro heterocycle and optionally substituted 10-membered fused heterocycle. In some cases, the optional one or more substituents of R1 are independently selected from halogen, -OH, -S(O)2(R20), -S(O)2N(R20)2, - S(O)N(R20)2, -S(O)R20(=NR20), -C(O)N(R20)2, -C(=NR20)N(R20)2, -C(O)OR20, -C(O)NHOR20, -
N(R20)2, -C(O)R20, -N02, =0, -CN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, C2-6 alkynyl, 5- to 12- membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted with one or more substituents selected from halogen, and Ci-6 alkyl. In some cases, R1 is selected from
, which is substituted with one or more substituents independently selected from halogen, -OH, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -C(O)N(R20)2, - C(=NR20)N(R20)2, -C(O)OR20, -C(O)NHOR20, -N(R20)2, -C(O)R20, -NO2, =0, -CN, Ci-6 alkyl- N(R20)2, CI-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-6 alkynyl, 5- to 12-membered heterocycle, wherein the 5- to 12- membered heterocycle is optionally substituted with one or more substituents selected from
[00223] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 6- to 11 -membered heterocycle, wherein the 6- to 11 -membered heterocycle has at least one nitrogen atom. In some cases, the one or more optional substituents of R1 is selected from halogen, -OR20, -C(O)N(R20)2, -C(O)R20, -S(O)2R20, =0, -Ci-6 alkyl(=NOR20), =NO(R20), -
CN, -NHCN, Ci-6 alkyl, and 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted independently with one or more R1*; and wherein each R1* is independently selected from halogen, and Ci-6 alkyl. In some embodiments, R1 is selected from
[00224] In some embodiments, for a compound or salt of Formula (I), R1 is selected from hydrogen and optionally substituted 5- to 15-membered heterocycle. In some cases, R1 is selected
of which is optionally substituted. In some cases, the optional one or more substituents of R1 is selected from -OH, =NO(R20), -NHCN, and Ci-6 alkyl. In some cases, R1 is selected from
[00225] In some embodiments, for a compound or salt of Formula (I), R1 is selected from hydrogen and optionally substituted 7- to 10-membered heterocycle. In some cases, R1 is selected
of which is optionally substituted. In some cases, the optional one or more substituents of R1 are independently selected from halogen, -NH2, -S(O)2(R20), -C(O)R20, -C(O)N(R20)2, =O,=NO(R20),
-CN, -NHCN, C1-6 alkyl, and 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted independently with one or more R1*; and wherein each R1* is independently selected from halogen, and C1-6 alkyl. In some cases, R1 is selected from hydrogen,
[00226] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 8- to 10-membered heterocycle. In some cases, the heterocycle is bicyclic. In some cases, the heterocycle has at least one nitrogen atom. In some cases, the heterocycle has
which is optionally substituted. In some cases, the optional one or more substituents of R1 are independently selected from halogen,
9-membered heteroaryl, wherein the 5- to 9-membered heteroaryl is substituted with at least one R1*, wherein the R1* is selected from halogen, and Ci-6 alkyl. In some cases, the optional one or more substituents of R1 are independently selected from chlorine,
[00227] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted bridged 8- to 9-membered heterocycle. In some cases, the heterocycle of R1 is selected from
more substituents of R1 are selected from halogen, Ci-6 alkyl, -N(R20)2, and Ci-6 aminoalkyl. In
[00228] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted bridged 8-membered heterocycle, wherein the heterocycle contains heteroatoms selected from nitrogen. In some cases, the one or more substituents of R1 are selected
from C i-6 alkyl, -N(R20)2, and Ci-6 aminoalkyl. In some cases, the heterocycle of R1 is selected from
each of which is optionally substituted. In some cases,
, ,
[00229] In some embodiments, for a compound or salt of Formula (I), R1 is hydrogen.
[00230] In some embodiments, for a compound or salt of Formula (I), R1 is an optionally substituted 12- to 15-membered heterocycle. In some cases, R1 is an optionally substituted 12- membered heterocycle. In some cases, R1 is an optionally substituted 13-membered heterocycle. In some cases, R1 is an optionally substituted 14-membered heterocycle. In some cases, R1 is an optionally substituted 15-membered heterocycle. In some cases, the heterocycle of R1 is tricyclic. In some cases, the heterocycle of R1 contains a fused heterocycle. In some cases, the heterocycle of R1 contains a spiro-heterocycle. In some cases, the heterocycle of R1 contains a fused and spiro-heterocycle. In some cases, the heterocycle of R1 is an unsaturated heterocycle. In some cases, the heterocycle of R1 is a non-aromatic heterocycle. In some cases, the heterocycle of R1 has at least one double bond. In some cases, the heterocycle of R1 has at least two double bonds. In some cases, the heterocycle of R1 has at least 2 heteroatoms. In some cases, the heterocycle of R1 has at least 3 heteroatoms. In some cases, the heterocycle of R1 has at least 4 heteroatoms. In some cases, the heterocycle of R1 has at least 5 heteroatoms. In some cases, the heterocycle of R1 has at least 6 heteroatoms. In some cases, the heterocycle of R1 has at least 7 heteroatoms. In some cases, the heteroatoms are selected from oxygen, nitrogen, and sulfur. In some cases, the heterocycle of R1 has at least 3, 4, or 5 nitrogen atoms, and at least 1 sulfur atom. In some cases, the heterocycle of R1 has at least 3, 4, or 5 nitrogen atoms, and at least 1 oxygen atom. In some cases, the heterocycle of R1 has at least 3, 4, or 5 nitrogen atoms. In some cases, the heterocycle of R1 has at least 3, 4, or 5 nitrogen atoms and no other heteroatoms. In some cases, the heteroatoms are selected from nitrogen and sulfur. In some cases, the heteroatoms are selected from nitrogen and oxygen. In some cases, R1 is selected from
each of which is optionally substituted with one or more substituents. In some cases, R1 is selected from
substituted with one or more substituents. In some cases, the optional one or more substituents of R1 are independently selected from halogen, -OH, -NHCN, -S(O)2(R20), -S(O)2N(R20)2, - S(O)N(R20)2, -S(O)R20(=NR20), -C(O)N(R20)2, -C(O)NHOR20, -N(R20)2, -C(O)R20, -NO2, =0, =NH, -CN, C 1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C1-6 cyanoalkyl, Ci-e haloalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the optional one or more substituents of R1 are independently selected from halogen, -OH, -NHCN, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -C(O)N(R20)2, -C(O)NHOR20, -N(R20)2, -C(O)R20, -NO2, =0, -CN, C1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, C1-6 haloalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the optional one or more substituents of R1 are independently selected from halogen, -OH, C1-6 alkyl, and -C(O)N(R20)2. In some cases, R1 is selected from
[00231] In some embodiments, for a compound or salt of Formula (I), R1 is an optionally substituted 12- to 15-membered heterocycle. In some cases,
wherein
Ring W is an optionally substituted heterocycle and Ring P is an optionally substituted carbocycle or optionally substituted heterocycle, wherein Ring P forms a spirocycle with Ring W. In some cases, Ring W is an optionally substituted fused heterocycle. In some cases, Ring P and Ring W combine to form a heterocycle having at least 12 atoms and most 15 atoms. In some cases, Ring P and Ring W have in total at least 12 atoms and most 15 atoms. In some cases, Ring
W is an optionally substituted 10-membered fused heterocycle. In some cases, R1 is
, wherein Ring P is an optionally substituted carbocycle or optionally substituted heterocycle. In some cases,
some cases, Ring P is an optionally substituted carbocycle. In some cases, Ring P is an optionally substituted heterocycle. In some cases, Ring P forms an optionally substituted C3-C6 carbocycle or optionally substituted 4-to 6- membered heterocycle. In some cases, Ring P forms an optionally substituted C3 carbocycle. In some cases, Ring P forms an optionally substituted C4 carbocycle. In some cases, Ring P forms an optionally substituted C5 carbocycle. In some cases, Ring P forms an optionally substituted 4- membered heterocycle. In some cases, Ring P forms an optionally substituted 5-membered heterocycle. In some cases, Ring P forms an optionally substituted 5-membered heterocycle. In some cases, Ring P has at least 1, 2, or 3 heteroatoms. In some cases, the heteroatoms are selected from oxygen, nitrogen, and sulfur. In some cases, Ring P has 1 sulfur atom. In some cases, Ring P has 1 nitrogen atom. In some cases, Ring P has 1 oxygen atom. In some cases, the one or more optional substituents of Ring P are independently selected from halogen, -OH, - NHCN, =0, =NR20, -CN, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of Ring P are independently selected from halogen, -OH, =0, =NH, -CN, and C1-6 alkyl. In some cases, the one or more optional substituents of Ring W are independently selected from halogen, -OH, -NHCN, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -C(O)N(R20)2, - C(O)NHOR20, -N(R20)2, -C(O)R20, -N02, -CN, C1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, C1-6 alkyl, and C2-6 alkynyl. In some cases, the one or more optional substituents of Ring W are independently selected from halogen, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -C(O)N(R20)2, -C(O)NR20OR20, -C(O)NHOR20, - N(R20)2, -C(O)R20, and C1-6 alkyl. In some cases, the one or more optional substituents of Ring W are independently selected from -C(O)R20. In some cases, Ring P is substituted. In some cases, Ring W is substituted.
[00232] In some embodiments, for a compound or salt for Formula (I), R1 is selected from a 5- to 12-membered bridged heterocycle, which is optionally substituted with one or more substituents. In some cases, R1 is selected from an 8-membered bridged heterocycle, which is
optionally substituted with one or more substituents. In some cases, the bridged heterocycle has at least 1 heteroatom. In some cases, the bridged heterocycle has at least 2 heteroatoms. In some cases, the bridged heterocycle has at least 1 nitrogen atom. In some cases, the bridged heterocycle has at least 2 nitrogen atoms. In some cases, the bridged heterocycle has 2 nitrogen atoms. In some cases, R1 is selected from
R1 is selected from — I— , which is optionally substituted with one or more substituents.
[00233] In some embodiments, for a compound or salt for Formula (I), R1 is selected from an unsaturated 5- to 12-membered heterocycle, which is optionally substituted with one or more substituents. In some cases, the unsaturated 5- to 12-membered heterocycle is selected from
each of which is optionally substituted with one or more substituents.
[00234] In some embodiments, for a compound or salt of Formula (I), wherein the 5- to 12- membered heterocycle of R1 is unsaturated and a bridged heterocycle. In some cases, R1 is selected from an optionally substituted 7- to 8-membered unsaturated and bridged heterocycle. In
some cases, R1 is selected from -J— .
[00235] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 10-membered heterocycle. In some cases, the 10-membered heterocycle is a bicyclic heterocycle. In some cases, the 10-membered heterocycle is a spiro heterocycle. In some cases, the 10-membered heterocycle is a fused heterocycle. In some cases, the 10- membered heterocycle is a saturated heterocycle. In some cases, the 10-membered heterocycle is a non-aromatic heterocycle. In some cases, the 10-membered heterocycle contains at least 1 nitrogen atom. In some cases, the 10-membered heterocycle contains at least 2 nitrogen atoms. In some cases, the 10-membered heterocycle contains at least 3 nitrogen atoms. In some cases, the 10-membered heterocycle contains at least 1 sulfur atom. In some cases, R1 is selected from
each of which is optionally substituted with one or more substituents independently selected from halogen, =0, -OH, -CN, -NHCN, -C(O)N(R20)2, - C(O)NR20OR20, C 1-6 aminoalkyl, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, and Ci-6 alkyl. In some
some cases, R1 is selected from
substituted with one or more substituents independently selected from halogen, -OR20, -SR20, - N(R20)2, -NO2, =0, -CN, C 1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, C1-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, and C2-e alkynyl.
[00236] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 8- to 10-membered fused heterocycle. In some cases, the 8- to 10- membered fused heterocycle is a bicyclic heterocycle. In some cases, the 8- to 10-membered
fused heterocycle is a saturated heterocycle. In some cases, the 8- to 10-membered heterocycle is a non-aromatic heterocycle. In some cases, R1 is selected from an optionally substituted 10- membered fused heterocycle. In some cases, the 10-membered fused heterocycle is a bicyclic heterocycle. In some cases, the 10-membered fused heterocycle is a saturated heterocycle. In some cases, the 10-membered heterocycle is a non-aromatic heterocycle. In some cases, the fused heterocycle has one saturated ring and one aromatic ring. In some cases, the fused heterocycle has one saturated ring and one unsaturated ring. In some cases, the fused heterocycle has two saturated rings. In some cases, the 10-membered heterocycle contains at least 1 nitrogen atom. In some cases, the 10-membered heterocycle contains at least 2 nitrogen atoms. In some cases, the 10-membered heterocycle contains at least 3 nitrogen atoms. In some cases, R1 is
, each of which is optionally substituted with one or more substituents. In some cases,
, which is optionally substituted with one or more substituents. In some cases, the optional one or more substituents are independently selected from halogen, =0, -OH, -CN, -NHCN, -C(O)R20, -C(O)N(R20)2, -C(O)NR20OR20, Ci-6 aminoalkyl, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, and Ci-6 alkyl. In some cases, the optional one or more substituents are independently selected from halogen, =0, -C(O)R20, -C(O)N(R20)2, and - C(O)NR20OR20. In some cases, the optional one or more substituents are independently selected from -C(O)R20, -C(O)N(R20)2, and -C(O)NR20OR20. In some cases, the optional one or more substituents are independently selected from -C(O)R20. In some cases, the optional one or more substituents are independently selected from -C(O)N(R20)2. In some cases, the optional one or more substituents are independently selected from -C(O)NR20OR20. In some cases, each R20 is independently selected from hydrogen; and Ci-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle. In some cases, each R20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12-membered heterocycle. In some cases, each R20 is independently selected from hydrogen; and C1-6 alkyl, and 3- to 12-membered saturated heterocycle. In some cases, the optional one or more substituents of R1 are independently selected from
[00237] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted saturated 6- to 7-membered heterocycle. In some cases, R1 is selected from an optionally substituted saturated 6-membered heterocycle. In some cases, R1 is selected from
—I— , which is optionally substituted. In some cases, the optional one or more substituents are independently selected from halogen, -CN, -NHCN, Ci-6 cyanoalkyl, and Ci-6 alkyl. In some cases, the optional one or more substituents are independently selected from -CN, -NHCN, Ci-6 cyanoalkyl, and Ci-6 alkyl. In some cases, the optional one or more substituents are independently selected from -CN, -NHCN, Ci-6 cyanoalkyl, and Ci-6 alkyl. In some cases, the optional one or more substituents are independently selected from -NHCN, and Ci-6 alkyl. In
some cases, R1 is selected from — i— , which is substituted with one or more substituents selected from -NHCN, and Ci-6 alkyl. In some cases, R1 is selected from
, and
[00238] In some embodiments, for a compound or salt of Formula (I), R3 is -CN, and R1 is
substituted 8- to 9-membered fused heterocycle, wherein the 8- to 9-membered fused heterocycle has at least one sulfur atom. In some cases, R3 is -CN, and R1 is selected from
. In some cases, R3 is -CN, and R1 is selected from . In some cases, R3 is -CN,
O and R1 is selected from
. In some cases, R3 is -CN, and R1 is selected from
some cases,
selected from
optionally substituted 8- to 9-membered fused heterocycle, wherein the 8- to 9-membered fused heterocycle has at least one sulfur atom; and Y-R2 is selected from -O-L-heterocycle, wherein the heterocycle portion of -O-L-heterocycle is optionally substituted with one or more R6.
[00239] In some embodiments, for a compound or salt of Formula (I), B is an optionally substituted 8- to 10-membered fused carbocycle. In some cases, B is a substituted 8- to 10- membered fused carbocycle. In some cases, B is an optionally substituted 9-membered fused carbocycle. In some cases, B is a substituted 9-membered fused carbocycle. In some cases, B is
, which is optionally substituted with one or more substituents. In some cases, B is
, which is substituted with one or more substituents. In some cases, for B, the one or more substituents are independently selected from halogen, oxo, -NH2, C1-C3 alkyl, -B(OH)2, -
OH, -C(0)NH2, -NH2, =0, -CN, C1-6 alkoxy, C1-6 hydroxyalkyl, and C2-e alkynyl. In some cases,
B is substituted with at least one halogen. In some cases, B is substituted with at least one chlorine. In some cases, B is substituted with at least one fluorine. In some cases, B is selected
selected from halogen and Ci-e haloalkyl. In some cases,
which is substituted with one or more substituents selected from halogen. In some cases, B is selected from
which is substituted with one or more substituents selected from fluorine. In some cases, B is
substituted with one or more substituents selected from chlorine. In some cases, B is selected
[00240] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted unsaturated 10-membered fused heterocycle. In some cases, R1 is
, which is optionally substituted. In some cases, the optional one or more substituents of R1 are independently selected from halogen, =0, -OH, -CN, -NHCN, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -C(O)R20, -C(O)N(R20)2, -C(O)NR20OR20, CI-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, and Ci-6 alkyl. In some cases, B is an optionally substituted 8- to 10-membered fused heterocycle, wherein the heterocycle
each of which is optionally substituted. In some cases, In some cases, for B, the one or more optional substituents of the heterocycle and carbocycle are independently selected at each occurrence from halogen, oxo, -NH2, C1-C3 alkyl, -OH, -O-C1-C3 haloalkyl, -C(O)NH2, -NH2, =0, -CN, C1-6 alkoxy, C1-6 hydroxyalkyl, and C2-6 alkynyl. In some cases, Y-R2 is selected from
, wherein the heterocycle portion is optionally substituted. In some cases, n is 0. In some cases, R3 is selected from hydrogen, halogen, -CN, -N(R20)2, -OH, -S(O)2(R20), -C(O)R20, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, C 1-6 haloalkyl, and Ci-6 alkyl. In some cases, R3 is selected from hydrogen, halogen, and -CN.
[00241] In some embodiments, for a compound or salt of Formula (I), R1 is selected from an optionally substituted 11 -membered fused heterocycle. In some cases, R1 is selected from an optionally substituted unsaturated 11 -membered fused heterocycle. In some cases, the heterocycle contains at least one sulfur atom. In some cases, the heterocycle contains at least one nitrogen atom. In some cases, the heterocycle contains 3 heteroatoms. In some cases, the
heterocycle is — L~ , which is optionally substituted. In some cases, R1 is — L~
[00242] In some embodiments, for a compound or salt of Formula (I), the one or more optional substituents of R1 are each independently selected from halogen, -CN, -NO2, =0, - N(R20)2, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), -S(O)2N(R20)2, -NR20S(O)2R20, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, Ci-e hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, C1-6 alkyl, C2-6 alkenyl, and C2-6 alkynyl. In some cases, the one or more optional substituents are independently selected from halogen, -OH, -N(R20)2, -NO2, =0, -CN, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and C1-6 haloalkyl. In some cases, the optional substituents for R1 are each independently selected from halogen, -OH, -N(R20)2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and C1-6 haloalkyl. In some cases, the optional substituents for R1 are each independently selected from halogen, -CN, -OH, -NH2, C 1-6 aminoalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, and C1-6 haloalkyl. . In some cases, the optional substituents for R1 are each independently selected from -OH, and -NH2. In some cases, R20 is selected from hydrogen and C1-3 alkyl.
[00243] In some embodiments, for a compound or salt for Formula (I), n is selected from 0 to 4. In some cases, n is selected from 0 to 3. In some cases, n is selected from 0 to 2. In some cases, n is selected from 0 and 1. In some cases, n is 0. In some cases, n is 1. In some cases, n is
[00244] In some embodiments, for a compound or salt for Formula (I), each R4 is independently selected from halogen, -NO2, =0, =S, -CN, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C2-6 alkynyl, C1-6 cyanoalkyl, and C 1-6 haloalkyl. In some cases, R4 is independently selected from halogen, =0, -CN, C1-6 cyanoalkyl, C2-6 alkynyl, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and C1-6 haloalkyl. In some cases, R4 is independently selected from halogen, =0, -CN, C1-6 cyanoalkyl, C2-6 alkynyl, and C 1-6 haloalkyl. In some cases, R4 is independently selected from -CN, C1-6 cyanoalkyl, and C2-6 alkynyl. In some cases, each R4 is independently selected from halogen, -NO2, =0, =S, -CN, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 cyanoalkyl, C 1-6 hydroxyalkyl, and C1-6 haloalkyl.
[00245] In some embodiments, for a compound or salt for Formula (I), Q is a bond. In some cases, Q is a S. In some cases, Q is a O.
[00246] In some embodiments, for a compound or salt for Formula (I), each R7 is independently selected from halogen, hydroxy, HC(=O)-, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 hydroxyalkyl, and -N(R5)2. In some cases, each R7 is independently selected from halogen, hydroxy, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, and C1-C4 hydroxyalkyl.
[00247] In some embodiments, for a compound or salt for Formula (I), each R20 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(Ci-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci- 10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle. In some cases, each R20 is independently selected from hydrogen and C1-6 alkyl. In some cases, each R20 is independently selected from C1-6 alkyl.
[00248] In some embodiments, for a compound or salt for Formula (I), each R21 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(Ci-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci- 10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle. In some cases, each R21 is independently selected from hydrogen and C1-6 alkyl. In some cases, each R21 is independently selected from C1-6 alkyl.
[00249] In some embodiments, for a compound or salt for Formula (I), each R20 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(Ci-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci- 10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle. In some cases, each R20 is independently selected from hydrogen and C1-6 alkyl. In some cases, each R20 is independently selected from C1-6 alkyl.
[00250] In some embodiments, for a compound or salt for Formula (I), each R23 is independently selected from hydrogen; and C1-6 alkyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O- C1-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle. In some cases, each R23 is independently selected from hydrogen and C1-6 alkyl. In some cases, each R23 is independently selected from C1-6 alkyl.
[00251] . In an aspect, the present disclosure provides a compound of Formula (I- A)
Formula (LA), or a pharmaceutically acceptable salt thereof wherein:
R1 is selected from 8- to 10-membered heterocycle, wherein the 8- to 10-membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -OR20, -S(O)2(R20), -C(O)N(R20)2, -Ci-6 alkyl(=NOR20), -C(O)R20 =0, -CN, -NHCN, Ci- 6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Cn 6 alkyl-SO2R20, Ci-6 alkoxyalkyl, Ci-6 alkyl, C2-6 alkynyl, and 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle are each optionally substituted independently with one or more R1*; each R1* is independently selected from halogen, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(O)NR20OR20, - N(R20)C(O)R20, -N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, -C(O)R20, -C(O)OR20, - OC(O)R20, -OC(O)N(R20)2, -N02, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and Cs-Ci2 carbocycle;
B is selected from a 7- to 15-membered heterocycle and C7-C15 carbocycle, wherein the 7- to 15-membered heterocycle and C7-C15 carbocycle are each optionally substituted with one or more substituents independently selected from halogen, -CN, -N02, =0, -N(R21)2, -B(OR21)2, - OR21, -SR21, -S(O)2(R21), -S(O)2N(R21)2, -NR21S(O)2R21, -C(O)N(R21)2, -C(O)NR21OR21, - N(R21)C(O)R21, -N(R21)C(O)N(R21)2, -N(R21)C(O)OR21, -C(0)R21, C(0)0R21, -0C(0)R21, - OC(O)N(R21)2, CI-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, Cs-Ci2 carbocycle and 5- to 12-membered heterocycle;
Y is selected from a bond, -0-, -S-, and -N(R5)-;
R2is selected from heterocycle, aryl, Ci-Ce alkyl, -L-heterocycle, -L-N(R23)2, -L-OR23, -L- aryl, -L-heteroaryl, -L-cycloalkyl, -L-NHC(=NH)NH2, -L-C(O)N(R23)2, -L-Ci-Ce haloalkyl, -L- OR23, -L-NR23C(O)-aryl, -L-C00H, -L-NR23S(O)2(R23), -L-S(O)2N(R23)2, -L-N(R23)C(O)(OR23), -L-OC(O)N(R23)2, and -L-C(=O)OCi-Ce alkyl, wherein the heterocycle, the heterocycle portion of -L-heterocycle, and the cycloalkyl portion of the -L-cycloalkyl are each optionally substituted with one or more R6, and wherein the aryl, aryl portion of -L-NR23C(O)-aryl, the aryl portion of -L-
NR23C(O)-aryl, the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R7;
R3 is selected from hydrogen, halogen, -CN, -NO2, -N(R20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -N(R20)C(O)R20, - N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, -C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-C12 carbocycle and 5- to 12-membered heterocycle; each R4 is independently selected from halogen, -NO2, =0, =S, -CN, C1-6 alkyl, C2-6 alkynyl, C 1-6 alkyl -N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C 1-6 cyanoalkyl, C1-6 hydroxyalkyl, and C1-6 haloalkyl; n is selected from 0, 1, 2, 3, and 4; each R5 is independently selected from hydrogen and Ci-Ce alkyl; each R6 is independently selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, oxo, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, =CH2, NMO-C1-C3 alkyl, C1-C3 aminoalkyl, - N(R5)S(O)2(R5), -Q-phenyl, -Q-phenylSChF, -NHC(O)phenyl, -NHC(O)phenylSO2F, C1-C3 alkyl substituted pyrazolyl, -C1-C3 alkyl-N(R5)2, -C(O)N(R5)2, tert-butyldimethylsilyloxyCFh-, - N(R5)2, (C1-C3 alkoxy)Ci-C3 alkyl-, (C1-C3 alkyl)C(=O), oxo, (C1-C3 haloalkyl)C(=O)-, -SO2F, (C1-C3 alkoxy)Ci-C3 alkoxy, -CH2OC(O)N(R5)2, -CH2NHC(O)OCI-C6 alkyl, - CH2NHC(O)N(R5)2, -CH2NHC(O)CI-C6 alkyl, -CH2(pyrazolyl), -CH2NHSO2C1-C6 alkyl, - CH2OC(O)heterocycle, -OC(O)N(R5)2, -OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl), -0C(0)NH(Ci- C3 alkyl)O(Ci-C3 alkyl)phenyl(Ci-C3 alkyl)N(CH3)2, -OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl)phenyl, -OC(O)heterocycle, -O-C1-C3 alkyl, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -NR20S(O)2R20, and -CFhheterocycle, wherein the phenyl of -NHC(O)phenyl and -0C(0)NH(CI-C3 alkyl)(Ci-C3 alkyl)phenyl are optionally substituted with one or more substituents selected from -C(0)H and OH, and wherein the alkyl of -O-C1-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo and hydroxy; and wherein the heterocycle of -CH2heterocyclyl is optionally substituted with oxo; each Q is selected from a bond, S, and O; each R7 is independently selected from halogen, hydroxy, HC(=0)-, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 hydroxyalkyl, -C1-C3 alkyl-N(R5)2, -C(O)N(R5)2, and -N(R5)2; each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen, hydroxy, C1-6 alkoxy, C1-C4 hydroxyalkyl, C1-C4 alkyl, C3-C6 carbocycle, and 3- to 8-membered heterocycle, wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents
independently selected from halogen, -OH, -N02, =0, =S, -CN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-ehydroxyalkyl, Ci-6 haloalkyl; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle, wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -N02, =0, =S, -CN, Ci-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and C1-6 haloalkyl; each R20 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -N02, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle; each R21 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -N02, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle; each R23 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -N02, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle.
[00252] In some embodiments, Formula (I) or Formula (I-A) is represented by Formula (I-B),
Formula (I-B), or a pharmaceutically acceptable salt thereof wherein: wherein R1 is selected from
each of which is optionally substituted with one or more substituents independently selected from halogen, - S(O)2(R20), -C(O)N(R20)2, -CI-6 alkyl(=NOR20), -C(O)R20, =0, and 5- to 12-membered
heterocycle, wherein the 5- to 12-membered heterocycle are each optionally substituted independently with one or more R1*; each R1* is independently selected from halogen, -OR20, -N(R20)2, -NO2, =O, =N(R20), =NO(R20), -CN, -NHCN, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C 1-6 haloalkyl, and C1-6 alkyl;
Y is -O-;
R2 is selected from -L-heterocycle, -L-N(R23)2, wherein the heterocycle portion of -L- heterocycle is optionally substituted with one or more R6.
B is selected from a 7- to 15-membered heterocycle, wherein the 7- to 15-membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -CN, =0, -N(R21)2, -OR21, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, C 1-6 haloalkyl, C1-6 alkyl, and C2-e alkynyl;
R3 is selected from hydrogen, halogen, -CN, -N(R20)2, -OR20, -C(O)R20, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, and C1-6 alkyl; and each R20 is independently selected from hydrogen; and C1-6 alkyl, and C3-12 carbocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NH2, -N(Ci-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, and oxo.
[00253] In some embodiments, a compound or salt of Formula (I), Formula (LA), or Formula
optionally substituted with one or more substituents independently selected from halogen, - S(O)2(R20), -C(O)N(R20)2, -CI-6 alkyl(=NOR20), -C(O)R20, =0, and 5- to 9-membered heterocycle, wherein the 5- to -membered heterocycle are each optionally substituted independently with one or more R1*; and each R1* is independently selected from halogen, Ci-6 haloalkyl, and Ci-6 alkyl.
[00254] In some embodiments, a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), Y is -O-.
[00255] In some embodiments, a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), R2 is selected from optionally substituted -L-heterocycle, and -L-N(R23)2. In some cases, R2 is selected from optionally substituted -L- 5-to 8-membered heterocycle, and -L-N(R23)2. In some cases, R2 is selected from optionally substituted -L-heterocycle. In some cases, R2 is selected from -L-N(R23)2. In some cases, the heterocycle contains at least one nitrogen atom. In
some cases, the heterocycle contains at least one sulfur atom. In some cases, the heterocycle contains at least one oxygen atom. In some cases, the heterocycle is a heteroaryl. In some cases, the heterocycle is a saturated heterocycle. In some cases, each R6 is independently selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, oxo, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, =CH2, =NO-CI-C3 alkyl, C1-C3 aminoalkyl, C1-C3 alkyl substituted pyrazolyl, -C1-C3 alkyl- N(R5)2, -C(O)N(R5)2, -N(R5)2, (C1-C3 alkoxy)Ci-C3 alkyl-, (C1-C3 alkyl)C(=O), oxo, -O-C1-C3 alkyl. In some cases, each R6 is independently selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, oxo, =CH2, and -O-C1-C3 alkyl. In some cases, when R2 is a heteroaryl, R6 is selected from halogen and C1-C3 haloalkyl, C1-C3 alkyl. In some cases, when R2 is a heteroaryl, R6 is selected from a halogen. In some cases, R23 is selected from hydrogen and C1-C3 alkyl. In some cases, each R23 is selected from C1-C3 alkyl. In some cases, each R23 is selected from methyl. In some cases, each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen, and
C1-C4 alkyl; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 4- to 6-membered heterocycle, wherein the C3-C6 carbocycle and 3- to 6-membered heterocycle are each optionally substituted with one or more
substituents independently selected from halogen. In some cases, each L is selected from ' »
[00256] In some embodiments, a compound or salt of Formula (I), Formula (I-A), or Formula
[00257] In some embodiments, a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), B is selected from an 8- to 10-membered heterocycle, wherein the 8- to 10-membered
heterocycle is optionally substituted with one or more substituents independently selected from halogen, -CN, -NH2, and C1-6 alkyl. In some cases, the heterocycle of B is selected from
substituted with one or more substituents independently selected from halogen, -CN, -NH2, and
[00258] In some embodiments, a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), R3 is -CN. In some cases, R3 is hydrogen. In some cases, R3 is halogen. In some cases, R3 is fluorine.
[00259] In some embodiments, a compound or salt of Formula (I), Formula (I-A), or Formula
which is optionally substituted with one or more substituents. In some cases, the
one or more optional substituents are independently selected from halogen, -S(O)2(R20), - C(O)N(R20)2, -CI-6 alkyl(=NOR20), -C(O)R20 =0, and 5- to 9-membered heterocycle, wherein the 5- to -membered heterocycle are each optionally substituted independently with one or more R1*. In some cases, the one or more optional substituents are independently selected from halogen, - S(O)2(R20), -C(O)N(R20)2, -CI-6 alkyl(=NOR20), -C(O)R20, =0, and 5- to 9-membered heterocycle, wherein the 5- to -membered heterocycle are each optionally substituted independently with one or more R1*. In some cases, the one or more optional substituents are independently selected from halogen, -C(O)N(R20)2, -Ci-6 alkyl(=NOR20), -C(O)R20, and 5- to 9-membered heterocycle, wherein the 5- to -membered heterocycle are each optionally substituted independently with one or more R1*. In some cases,
which is subtituted with at least one substituent. In
independently selected from halogen, Ci-e haloalkyl, and Ci-6 alkyl.
[00260] In some embodiments, for a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), does not contain an electrophile moiety.
[00261] In some embodiments, for a compound or salt of Formula (I), Formula (I-A), or Formula (I-B), the one or more optional substituents of R1 are not electrophiles.
[00262] In some embodiments, for a compound or salt of Formula (I), R1 is selected from a compound in the Examples. In some cases, B is selected from a compound in the Examples. In some cases, Y is selected from a compound in the Examples. In some cases, R2 is selected from a compound in the Examples. In some cases, R3 is selected from a compound in the Examples. [00263] In some embodiments, for a compound or salt of Formula (I), each R20 is selected from hydrogen and C1-3 alkyl. In some cases, each R20 is selected from hydrogen and Ci alkyl. [00264] In some embodiments, the compounds of Formula (I), (I-A), or (I-B) used in the methods include trifluoroacetic acid salts of the above compounds.
[00265] In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 5 mg to about 500 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 150 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 125 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 100 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 25 mg to about 100 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 50 mg to about 100 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) of is administered to a subject at about 5 mg to about 75 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, or about 150 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 15 mg, about 30 mg, about 45 mg, or about 60 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 15 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 30 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 45 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 60 mg. In some embodiments, the subject is between 12 years old to 18 years
old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
[00266] In some embodiments, a compound or salt of Formula (I), (I- A), or (I-B) is administered once daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I- B) is administered twice daily. In some embodiments, the compound or salt of Formula (I), (I- A), or (I-B) is administered 3 times daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered once weekly. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered every other day. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered every 3 days.
[00267] In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 10 mg to 150 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) or a salt thereof is administered to a subject at 10 mg to 125 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 10 mg to 100 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 25 mg to 100 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 50 mg to 100 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 5 mg to 75 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 10 mg, 15 mg, 20 mg, 25 mg, 30 mg, 35 mg, 40 mg, 45 mg, 50 mg, 55 mg, 60 mg, 65 mg, 70 mg, 75 mg, 80 mg, 85 mg, 90 mg, 100 mg, 105 mg, 110 mg, 115 mg, 120 mg, 125 mg, 130 mg, 135 mg, 140 mg, 145 mg, or 150 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 15 mg, 30 mg, 45 mg, or 60 mg. In some embodiments, the compound or salt of Formula (I) is administered to a subject at 15 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 30 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 45 mg. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at 60 mg. In some embodiments, the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
[00268] In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 150 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 125 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B)
is administered to a subject at about 10 mg to about 100 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 25 mg to about 100 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 50 mg to about 100 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 5 mg to about 75 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, or about 150 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 15 mg, about 30 mg, about 45 mg, or about 60 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 15 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 30 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 45 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 60 mg, daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered once daily. In some embodiments, the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
[00269] In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 150 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 125 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg to about 100 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 25 mg to about 100 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 50 mg to about 100 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 5 mg to about 75 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg,
about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, or about 150 mg, twice daily. In some cases, the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
[00270] In some embodiments, the compound or salt of Formula (I), (I- A), or (I-B) is administered to a subject at about 15 mg, about 30 mg, about 45 mg, or about 60 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I- A), or (I-B) is administered to a subject at about 15 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 30 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 45 mg, twice daily. In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B) is administered to a subject at about 60 mg, twice daily. In some embodiments, the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
[00271] In some embodiments, a compound of Formula (I), (I-A), or (I-B) is administered as a capsule during the period of time. In some cases, a tablet or capsule formulation of a compound of Formula (I), (I-A), or (I-B) comprises about 10 mg to about 100 mg (e.g., about 10 mg to about 95 mg, about 10 mg to about 90 mg, about 10 mg to about 85 mg, about 10 mg to about 80 mg, about 10 mg to about 75 mg, about 10 mg to about 70 mg, about 10 mg to about 65 mg, about 10 mg to about 60 mg, about 10 mg to about 55 mg, about 10 mg to about 50 mg, about 10 mg to about 45 mg, about 10 mg to about 40 mg, about 10 mg to about 35 mg, about 10 mg to about 30 mg, about 10 mg to about 25 mg, about 10 mg to about 20 mg, about 10 mg to about 15 mg, about 15 mg to about 100 mg, about 15 mg to about 95 mg, about 15 mg to about 90 mg, about 15 mg to about 85 mg, about 15 mg to about 80 mg, about 15 mg to about 75 mg, about 15 mg to about 70 mg, about 15 mg to about 65 mg, about 15 mg to about 60 mg, about 15 mg to about 55 mg, about 15 mg to about 50 mg, about 15 mg to about 45 mg, about 15 mg to about 40 mg, about 15 mg to about 35 mg, about 15 mg to about 30 mg, about 15 mg to about 25 mg, about 15 mg to about 20 mg, about 20 mg to about 100 mg, about 20 mg to about 95 mg, about 20 mg to about 90 mg, about 20 mg to about 85 mg, about 20 mg to about 80 mg, about 20 mg to about 75 mg, about 20 mg to about 70 mg, about 20 mg to about 65 mg, about 20 mg to about 60 mg, about 20 mg to about 55 mg, about 20 mg to about 50 mg, about 20 mg to about 45 mg, about 20 mg to about 40 mg, about 20 mg to about 35 mg, about 20 mg to about 30 mg, about 20 mg to about 25 mg, about 25 mg to about 100 mg, about 25 mg to about 95 mg, about 25 mg to about 90 mg, about 25 mg to about 85 mg, about 25 mg to about 80 mg, about 25 mg to about 75
mg, about 25 mg to about 70 mg, about 25 mg to about 65 mg, about 25 mg to about 60 mg, about 25 mg to about 55 mg, about 25 mg to about 50 mg, about 25 mg to about 45 mg, about 25 mg to about 40 mg, about 25 mg to about 35 mg, about 25 mg to about 30 mg, about 30 mg to about 100 mg, about 30 mg to about 95 mg, about 30 mg to about 90 mg, about 30 mg to about 85 mg, about 30 mg to about 80 mg, about 30 mg to about 75 mg, about 30 mg to about 70 mg, about 30 mg to about 65 mg, about 30 mg to about 60 mg, about 30 mg to about 55 mg, about 30 mg to about 50 mg, about 30 mg to about 45 mg, about 30 mg to about 40 mg, about 30 mg to about 35 mg, about 35 mg to about 100 mg, about 35 mg to about 95 mg, about 35 mg to about 90 mg, about 35 mg to about 85 mg, about 35 mg to about 80 mg, about 35 mg to about 75 mg, about 35 mg to about 70 mg, about 35 mg to about 65 mg, about 35 mg to about 60 mg, about 35 mg to about 55 mg, about 35 mg to about 50 mg, about 35 mg to about 45 mg, about 35 mg to about 40 mg, about 40 mg to about 100 mg, about 40 mg to about 95 mg, about 40 mg to about 90 mg, about 40 mg to about 85 mg, about 40 mg to about 80 mg, about 40 mg to about 75 mg, about 40 mg to about 70 mg, about 40 mg to about 65 mg, about 40 mg to about 60 mg, about 40 mg to about 55 mg, about 40 mg to about 50 mg, about 40 mg to about 45 mg, about 45 mg to about 100 mg, about 45 mg to about 95 mg, about 45 mg to about 90 mg, about 45 mg to about 85 mg, about 45 mg to about 80 mg, about 45 mg to about 75 mg, about 45 mg to about 70 mg, about 45 mg to about 65 mg, about 45 mg to about 60 mg, about 45 mg to about 55 mg, about 45 mg to about 50 mg, about 50 mg to about 100 mg, about 50 mg to about 95 mg, about 50 mg to about 90 mg, about 50 mg to about 85 mg, about 50 mg to about 80 mg, about 50 mg to about 75 mg, about 50 mg to about 70 mg, about 50 mg to about 65 mg, about 50 mg to about 60 mg, about 50 mg to about 55 mg, about 55 mg to about 100 mg, about 55 mg to about 95 mg, about 55 mg to about 90 mg, about 55 mg to about 85 mg, about 55 mg to about 80 mg, about 55 mg to about 75 mg, about 55 mg to about 70 mg, about 55 mg to about 65 mg, about 55 mg to about 60 mg, about 60 mg to about 100 mg, about 60 mg to about 95 mg, about 60 mg to about 90 mg, about 60 mg to about 85 mg, about 60 mg to about 80 mg, about 60 mg to about 75 mg, about 60 mg to about 70 mg, about 60 mg to about 65 mg, about 65 mg to about 100 mg, about 65 mg to about 95 mg, about 65 mg to about 90 mg, about 65 mg to about 85 mg, about 65 mg to about 80 mg, about 65 mg to about 75 mg, about 65 mg to about 70 mg, about 70 mg to about 100 mg, about 70 mg to about 95 mg, about 70 mg to about 90 mg, about 70 mg to about 85 mg, about 70 mg to about 80 mg, about 70 mg to about 75 mg, about 75 mg to about 100 mg, about 75 mg to about 95 mg, about 75 mg to about 90 mg, about 75 mg to about 85 mg, about 75 mg to about 80 mg, about 80 mg to about 100 mg, about 80 mg to about 95 mg, about 80 mg to about 90 mg, about 80 mg to about 85 mg, about 85 mg to about 100 mg, about 85 mg to about 95 mg, about 85 mg to about 90 mg, about 90 mg to about 100 mg, about 90 mg to about 95 mg, about 95 mg
to about 100 mg, about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 95 mg, or about 100 mg) of a compound of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof. In some cases, the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
[00272] In some embodiments, a compound of Formula (I), (I- A), or (I-B) is orally administered once a day (QD) on a daily basis during a period of time. In one embodiment, a compound of Formula (I), (I- A), or (I-B) is orally administered twice a day (BID) on a daily basis during a period of time. In one embodiment, a compound of Formula (I), (I- A), or (I-B) is orally administered in the amount of about 20 mg to about 500 mg (e.g., about 20 mg to about 480 mg, about 20 mg to about 460 mg, about 20 mg to about 440 mg, about 20 mg to about 420 mg, about 20 mg to about 400 mg, about 20 mg to about 380 mg, about 20 mg to about 360 mg, about 20 mg to about 340 mg, about 20 mg to about 320 mg, about 20 mg to about 300 mg, about 20 mg to about 280 mg, about 20 mg to about 260 mg, about 20 mg to about 240 mg, about 20 mg to about 220 mg, about 20 mg to about 200 mg, about 20 mg to about 180 mg, about 20 mg to about 160 mg, about 20 mg to about 140 mg, about 20 mg to about 120 mg, about 20 mg to about 100 mg, about 20 mg to about 80 mg, about 20 mg to about 60 mg, about 20 mg to about 40 mg, about 40 mg to about 500 mg, about 40 mg to about 480 mg, about 40 mg to about 460 mg, about 40 mg to about 440 mg, about 40 mg to about 420 mg, about 40 mg to about 400 mg, about 40 mg to about 380 mg, about 40 mg to about 360 mg, about 40 mg to about 340 mg, about 40 mg to about 320 mg, about 40 mg to about 300 mg, about 40 mg to about 280 mg, about 40 mg to about 260 mg, about 40 mg to about 240 mg, about 40 mg to about 220 mg, about 40 mg to about 200 mg, about 40 mg to about 180 mg, about 40 mg to about 160 mg, about 40 mg to about 140 mg, about 40 mg to about 120 mg, about 40 mg to about 100 mg, about 40 mg to about 80 mg, about 40 mg to about 60 mg, about 60 mg to about 500 mg, about 60 mg to about 480 mg, about 60 mg to about 460 mg, about 60 mg to about 440 mg, about 60 mg to about 420 mg, about 60 mg to about 400 mg, about 60 mg to about 380 mg, about 60 mg to about 360 mg, about 60 mg to about 340 mg, about 60 mg to about 320 mg, about 60 mg to about 300 mg, about 60 mg to about 280 mg, about 60 mg to about 260 mg, about 60 mg to about 240 mg, about 60 mg to about 220 mg, about 60 mg to about 200 mg, about 60 mg to about 180 mg, about 60 mg to about 160 mg, about 60 mg to about 140 mg, about 60 mg to about 120 mg, about 60 mg to about 100 mg, about 60 mg to about 80 mg, about 80 mg to about 500 mg, about 80 mg to about 480 mg, about 80 mg to about 460 mg, about 80 mg to about 440 mg, about 80 mg to about 420 mg, about 80 mg to about 400 mg, about 80 mg to about 380 mg, about 80 mg to about 360 mg, about 80 mg to about 340 mg, about 80 mg to
about 320 mg, about 80 mg to about 300 mg, about 80 mg to about 280 mg, about 80 mg to about 260 mg, about 80 mg to about 240 mg, about 80 mg to about 220 mg, about 80 mg to about 200 mg, about 80 mg to about 180 mg, about 80 mg to about 160 mg, about 80 mg to about 140 mg, about 80 mg to about 120 mg, about 80 mg to about 100 mg, about 100 mg to about 500 mg, about 100 mg to about 480 mg, about 100 mg to about 460 mg, about 100 mg to about 440 mg, about 100 mg to about 420 mg, about 100 mg to about 400 mg, about 100 mg to about 380 mg, about 100 mg to about 360 mg, about 100 mg to about 340 mg, about 100 mg to about 320 mg, about 100 mg to about 300 mg, about 100 mg to about 280 mg, about 100 mg to about 260 mg, about 100 mg to about 240 mg, about 100 mg to about 220 mg, about 100 mg to about 200 mg, about 100 mg to about 180 mg, about 100 mg to about 160 mg, about 100 mg to about 140 mg, about 100 mg to about 120 mg, about 120 mg to about 500 mg, about 120 mg to about 480 mg, about 120 mg to about 460 mg, about 120 mg to about 440 mg, about 120 mg to about 420 mg, about 120 mg to about 400 mg, about 120 mg to about 380 mg, about 120 mg to about 360 mg, about 120 mg to about 340 mg, about 120 mg to about 320 mg, about 120 mg to about 300 mg, about 120 mg to about 280 mg, about 120 mg to about 260 mg, about 120 mg to about 240 mg, about 120 mg to about 220 mg, about 120 mg to about 200 mg, about 120 mg to about 180 mg, about 120 mg to about 160 mg, about 120 mg to about 140 mg, about 140 mg to about 500 mg, about 140 mg to about 480 mg, about 140 mg to about 460 mg, about 140 mg to about 440 mg, about 140 mg to about 420 mg, about 140 mg to about 400 mg, about 140 mg to about 380 mg, about 140 mg to about 360 mg, about 140 mg to about 340 mg, about 140 mg to about 320 mg, about 140 mg to about 300 mg, about 140 mg to about 280 mg, about 140 mg to about 260 mg, about 140 mg to about 240 mg, about 140 mg to about 220 mg, about 140 mg to about 200 mg, about 140 mg to about 180 mg, about 140 mg to about 160 mg, about 160 mg to about 500 mg, about 160 mg to about 480 mg, about 160 mg to about 460 mg, about 160 mg to about 440 mg, about 160 mg to about 420 mg, about 160 mg to about 400 mg, about 160 mg to about 380 mg, about 160 mg to about 360 mg, about 160 mg to about 340 mg, about 160 mg to about 320 mg, about 160 mg to about 300 mg, about 160 mg to about 280 mg, about 160 mg to about 260 mg, about 160 mg to about 240 mg, about 160 mg to about 220 mg, about 160 mg to about 200 mg, about 160 mg to about 180 mg, about 180 mg to about 500 mg, about 180 mg to about 480 mg, about 180 mg to about 460 mg, about 180 mg to about 440 mg, about 180 mg to about 420 mg, about 180 mg to about 400 mg, about 180 mg to about 380 mg, about 180 mg to about 360 mg, about 180 mg to about 340 mg, about 180 mg to about 320 mg, about 180 mg to about 300 mg, about 180 mg to about 280 mg, about 180 mg to about 260 mg, about 180 mg to about 240 mg, about 180 mg to about 220 mg, about 180 mg to about 200 mg, about 200 mg to about 500 mg, about 200 mg to about 480 mg, about 200 mg to about 460 mg, about 200 mg to
about 440 mg, about 200 mg to about 420 mg, about 200 mg to about 400 mg, about 200 mg to about 380 mg, about 200 mg to about 360 mg, about 200 mg to about 340 mg, about 200 mg to about 320 mg, about 200 mg to about 300 mg, about 200 mg to about 280 mg, about 200 mg to about 260 mg, about 200 mg to about 240 mg, about 200 mg to about 220 mg, about 220 mg to about 500 mg, about 220 mg to about 480 mg, about 220 mg to about 460 mg, about 220 mg to about 440 mg, about 220 mg to about 420 mg, about 220 mg to about 400 mg, about 220 mg to about 380 mg, about 220 mg to about 360 mg, about 220 mg to about 340 mg, about 220 mg to about 320 mg, about 220 mg to about 300 mg, about 220 mg to about 280 mg, about 220 mg to about 260 mg, about 220 mg to about 240 mg, about 240 mg to about 500 mg, about 240 mg to about 480 mg, about 240 mg to about 460 mg, about 240 mg to about 440 mg, about 240 mg to about 420 mg, about 240 mg to about 400 mg, about 240 mg to about 380 mg, about 240 mg to about 360 mg, about 240 mg to about 340 mg, about 240 mg to about 320 mg, about 240 mg to about 300 mg, about 240 mg to about 280 mg, about 240 mg to about 260 mg, about 260 mg to about 500 mg, about 260 mg to about 480 mg, about 260 mg to about 460 mg, about 260 mg to about 440 mg, about 260 mg to about 420 mg, about 260 mg to about 400 mg, about 260 mg to about 380 mg, about 260 mg to about 360 mg, about 260 mg to about 340 mg, about 260 mg to about 320 mg, about 260 mg to about 300 mg, about 260 mg to about 280 mg, about 280 mg to about 500 mg, about 280 mg to about 480 mg, about 280 mg to about 460 mg, about 280 mg to about 440 mg, about 280 mg to about 420 mg, about 280 mg to about 400 mg, about 280 mg to about 380 mg, about 280 mg to about 360 mg, about 280 mg to about 340 mg, about 280 mg to about 320 mg, about 280 mg to about 300 mg, about 300 mg to about 500 mg, about 300 mg to about 480 mg, about 300 mg to about 460 mg, about 300 mg to about 440 mg, about 300 mg to about 420 mg, about 300 mg to about 400 mg, about 300 mg to about 380 mg, about 300 mg to about 360 mg, about 300 mg to about 340 mg, about 300 mg to about 320 mg, about 320 mg to about 500 mg, about 320 mg to about 480 mg, about 320 mg to about 460 mg, about 320 mg to about 440 mg, about 320 mg to about 420 mg, about 320 mg to about 400 mg, about 320 mg to about 380 mg, about 320 mg to about 360 mg, about 320 mg to about 340 mg, about 340 mg to about 500 mg, about 340 mg to about 480 mg, about 340 mg to about 460 mg, about 340 mg to about 440 mg, about 340 mg to about 420 mg, about 340 mg to about 400 mg, about 340 mg to about 380 mg, about 340 mg to about 360 mg, about 360 mg to about 500 mg, about 360 mg to about 480 mg, about 360 mg to about 460 mg, about 360 mg to about 440 mg, about 360 mg to about 420 mg, about 360 mg to about 400 mg, about 360 mg to about 380 mg, about 380 mg to about 500 mg, about 380 mg to about 480 mg, about 380 mg to about 460 mg, about 380 mg to about 440 mg, about 380 mg to about 420 mg, about 380 mg to about 400 mg, about 400 mg to about 500 mg, about 400 mg to about 480 mg, about 400 mg to about 460 mg, about 400 mg to
about 440 mg, about 400 mg to about 420 mg, about 420 mg to about 500 mg, about 420 mg to about 480 mg, about 420 mg to about 460 mg, about 420 mg to about 440 mg, about 440 mg to about 500 mg, about 440 mg to about 480 mg, about 440 mg to about 460 mg, about 460 mg to about 500 mg, about 460 mg to about 480 mg, about 480 mg to about 500 mg, about 25, about 50, about 75, about 100, about 150, about 200, about 250, about 300, about 350, about 400, about 450, or about 500 mg), during a period of time. In some cases, the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
[00273] In some embodiments, the combination therapy comprises oral administration of a compound of Formula (I), (I- A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg (e.g., about 10 mg to about 380 mg, about 10 mg to about 360 mg, about 10 mg to about 340 mg, about 10 mg to about 320 mg, about 10 mg to about 300 mg, about 10 mg to about 280 mg, about 10 mg to about 260 mg, about 10 mg to about 240 mg, about 10 mg to about 220 mg, about 10 mg to about 200 mg, about 10 mg to about 180 mg, about 10 mg to about 160 mg, about 10 mg to about 140 mg, about 10 mg to about 120 mg, about 10 mg to about 100 mg, about 10 mg to about 80 mg, about 10 mg to about 60 mg, about 10 mg to about 40 mg, about 10 mg to about 20 mg, about 20 mg to about 400 mg, about 20 mg to about 380 mg, about 20 mg to about 360 mg, about 20 mg to about 340 mg, about 20 mg to about 320 mg, about 20 mg to about 300 mg, about 20 mg to about 280 mg, about 20 mg to about 260 mg, about 20 mg to about 240 mg, about 20 mg to about 220 mg, about 20 mg to about 200 mg, about 20 mg to about 180 mg, about 20 mg to about 160 mg, about 20 mg to about 140 mg, about 20 mg to about 120 mg, about 20 mg to about 100 mg, about 20 mg to about 80 mg, about 20 mg to about 60 mg, about 20 mg to about 40 mg, about 40 mg to about 400 mg, about 40 mg to about 380 mg, about 40 mg to about 360 mg, about 40 mg to about 340 mg, about 40 mg to about 320 mg, about 40 mg to about 300 mg, about 40 mg to about 280 mg, about 40 mg to about 260 mg, about 40 mg to about 240 mg, about 40 mg to about 220 mg, about 40 mg to about 200 mg, about 40 mg to about 180 mg, about 40 mg to about 160 mg, about 40 mg to about 140 mg, about 40 mg to about 120 mg, about 40 mg to about 100 mg, about 40 mg to about 80 mg, about 40 mg to about 60 mg, about 60 mg to about 400 mg, about 60 mg to about 380 mg, about 60 mg to about 360 mg, about 60 mg to about 340 mg, about 60 mg to about 320 mg, about 60 mg to about 300 mg, about 60 mg to about 280 mg, about 60 mg to about 260 mg, about 60 mg to about 240 mg, about 60 mg to about 220 mg, about 60 mg to about 200 mg, about 60 mg to about 180 mg, about 60 mg to about 160 mg, about 60 mg to about 140 mg, about 60 mg to about 120 mg, about 60 mg to about 100 mg, about 60 mg to about 80 mg, about 80 mg to about 400 mg, about 80 mg to about 380 mg, about 80 mg to about 360 mg, about 80 mg to about 340 mg, about 80 mg to about 320
mg, about 80 mg to about 300 mg, about 80 mg to about 280 mg, about 80 mg to about 260 mg about 80 mg to about 240 mg, about 80 mg to about 220 mg, about 80 mg to about 200 mg, about 80 mg to about 180 mg, about 80 mg to about 160 mg, about 80 mg to about 140 mg, about 80 mg to about 120 mg, about 80 mg to about 100 mg, about 100 mg to about 400 mg, about 100 mg to about 380 mg, about 100 mg to about 360 mg, about 100 mg to about 340 mg, about 100 mg to about 320 mg, about 100 mg to about 300 mg, about 100 mg to about 280 mg, about 100 mg to about 260 mg, about 100 mg to about 240 mg, about 100 mg to about 220 mg, about 100 mg to about 200 mg, about 100 mg to about 180 mg, about 100 mg to about 160 mg, about 100 mg to about 140 mg, about 100 mg to about 120 mg, about 120 mg to about 400 mg, about 120 mg to about 380 mg, about 120 mg to about 360 mg, about 120 mg to about 340 mg, about 120 mg to about 320 mg, about 120 mg to about 300 mg, about 120 mg to about 280 mg, about 120 mg to about 260 mg, about 120 mg to about 240 mg, about 120 mg to about 220 mg, about 120 mg to about 200 mg, about 120 mg to about 180 mg, about 120 mg to about 160 mg, about 120 mg to about 140 mg, about 140 mg to about 400 mg, about 140 mg to about 380 mg, about 140 mg to about 360 mg, about 140 mg to about 340 mg, about 140 mg to about 320 mg, about 140 mg to about 300 mg, about 140 mg to about 280 mg, about 140 mg to about 260 mg, about 140 mg to about 240 mg, about 140 mg to about 220 mg, about 140 mg to about 200 mg, about 140 mg to about 180 mg, about 140 mg to about 160 mg, about 160 mg to about 400 mg, about 160 mg to about 380 mg, about 160 mg to about 360 mg, about 160 mg to about 360 mg, about 160 mg to about 340 mg, about 160 mg to about 320 mg, about 160 mg to about 300 mg, about 160 mg to about 280 mg, about 160 mg to about 260 mg, about 160 mg to about 240 mg, about 160 mg to about 220 mg, about 160 mg to about 200 mg, about 160 mg to about 180 mg, about 180 mg to about 400 mg, about 180 mg to about 380 mg, about 180 mg to about 360 mg, about 180 mg to about 340 mg, about 180 mg to about 320 mg, about 180 mg to about 300 mg, about 180 mg to about 280 mg, about 180 mg to about 260 mg, about 180 mg to about 240 mg, about 180 mg to about 220 mg, about 180 mg to about 200 mg, about 200 mg to about 400 mg, about 200 mg to about 380 mg, about 200 mg to about 360 mg, about 200 mg to about 340 mg, about 200 mg to about 320 mg, about 200 mg to about 300 mg, about 200 mg to about 280 mg, about 200 mg to about 260 mg, about 200 mg to about 240 mg, about 200 mg to about 220 mg, about 220 mg to about 400 mg, about 220 mg to about 380 mg, about 220 mg to about 360 mg, about 220 mg to about 340 mg, about 220 mg to about 320 mg, about 220 mg to about 300 mg, about 220 mg to about 280 mg, about 220 mg to about 260 mg, about 220 mg to about 240 mg, about 240 mg to about 400 mg, about 240 mg to about 380 mg, about 240 mg to about 360 mg, about 240 mg to about 340 mg, about 240 mg to about 320 mg, about 240 mg to about 300 mg, about 240 mg to about 280 mg, about 240 mg to about 260 mg, about 260 mg to about 400 mg,
about 260 mg to about 380 mg, about 260 mg to about 360 mg, about 260 mg to about 340 mg, about 260 mg to about 320 mg, about 260 mg to about 300 mg, about 260 mg to about 280 mg, about 280 mg to about 400 mg, about 280 mg to about 380 mg, about 280 mg to about 360 mg, about 280 mg to about 340 mg, about 280 mg to about 320 mg, about 280 mg to about 300 mg, about 300 mg to about 400 mg, about 300 mg to about 380 mg, about 300 mg to about 360 mg, about 300 mg to about 340 mg, about 300 mg to about 320 mg, about 320 mg to about 400 mg, about 320 mg to about 380 mg, about 320 mg to about 360 mg, about 340 mg to about 360 mg, about 340 mg to about 400 mg, about 340 mg to about 380 mg, about 340 mg to about 360 mg, about 360 mg to about 400 mg, about 360 mg to about 380 mg, about 380 mg to about 400 mg, about 100 mg, about 200 mg, about 300 mg, or about 400 mg), and oral administration of an inhibitor selected from:
1) immunomodulator inhibitor;
2) PD-1 inhibitor;
3) PD-L1 inhibitor; and
4) CTLA-4 inhibitor; or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time). In some cases, the inhibitor is an immunomodulator inhibitor. In some cases, the inhibitor is an PD-1 inhibitor. In some cases, the inhibitor is an PD-L1 inhibitor. In some cases, the inhibitor is an CTLA-4 inhibitor. In some cases, the compound of Formula (I), (I-A), or (I-B) is selected from compounds 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of any one thereof. In some cases, the compound of Formula (I), (I-A), or (I-B)is selected compound2, or a pharmaceutically acceptable salt of any one thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 4, or a pharmaceutically acceptable salt of any one thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25, or a pharmaceutically acceptable salt of any one thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74 or a pharmaceutically acceptable salt of any one thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is orally administered once daily. In one embodiment, the compound of Formula (I), (I-A), or (I-B) is orally administered twice daily.
[00274] In some embodiments, a compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or pharmaceutical composition thereof is administered as a tablet or capsule. In one embodiment, a tablet or capsule formulation of a compound of Formula (I), (I- A), or (I-B) comprises about 10 mg to about 100 mg (e.g., about 10 mg to about 95 mg, about 10 mg to about 90 mg, about 10 mg to about 85 mg, about 10 mg to about 80 mg, about 10 mg to about 75 mg, about 10 mg to about 70 mg, about 10 mg to about 65 mg, about 10 mg to about 60
mg, about 10 mg to about 55 mg, about 10 mg to about 50 mg, about 10 mg to about 45 mg, about 10 mg to about 40 mg, about 10 mg to about 35 mg, about 10 mg to about 30 mg, about 10 mg to about 25 mg, about 10 mg to about 20 mg, about 10 mg to about 15 mg, about 15 mg to about 100 mg, about 15 mg to about 95 mg, about 15 mg to about 90 mg, about 15 mg to about 85 mg, about 15 mg to about 80 mg, about 15 mg to about 75 mg, about 15 mg to about 70 mg, about 15 mg to about 65 mg, about 15 mg to about 60 mg, about 15 mg to about 55 mg, about 15 mg to about 50 mg, about 15 mg to about 45 mg, about 15 mg to about 40 mg, about 15 mg to about 35 mg, about 15 mg to about 30 mg, about 15 mg to about 25 mg, about 15 mg to about 20 mg, about 20 mg to about 100 mg, about 20 mg to about 95 mg, about 20 mg to about 90 mg, about 20 mg to about 85 mg, about 20 mg to about 80 mg, about 20 mg to about 75 mg, about 20 mg to about 70 mg, about 20 mg to about 65 mg, about 20 mg to about 60 mg, about 20 mg to about 55 mg, about 20 mg to about 50 mg, about 20 mg to about 45 mg, about 20 mg to about 40 mg, about 20 mg to about 35 mg, about 20 mg to about 30 mg, about 20 mg to about 25 mg, about 25 mg to about 100 mg, about 25 mg to about 95 mg, about 25 mg to about 90 mg, about 25 mg to about 85 mg, about 25 mg to about 80 mg, about 25 mg to about 75 mg, about 25 mg to about 70 mg, about 25 mg to about 65 mg, about 25 mg to about 60 mg, about 25 mg to about 55 mg, about 25 mg to about 50 mg, about 25 mg to about 45 mg, about 25 mg to about 40 mg, about 25 mg to about 35 mg, about 25 mg to about 30 mg, about 30 mg to about 100 mg, about 30 mg to about 95 mg, about 30 mg to about 90 mg, about 30 mg to about 85 mg, about 30 mg to about 80 mg, about 30 mg to about 75 mg, about 30 mg to about 70 mg, about 30 mg to about 65 mg, about 30 mg to about 60 mg, about 30 mg to about 55 mg, about 30 mg to about 50 mg, about 30 mg to about 45 mg, about 30 mg to about 40 mg, about 30 mg to about 35 mg, about 35 mg to about 100 mg, about 35 mg to about 95 mg, about 35 mg to about 90 mg, about 35 mg to about 85 mg, about 35 mg to about 80 mg, about 35 mg to about 75 mg, about 35 mg to about 70 mg, about 35 mg to about 65 mg, about 35 mg to about 60 mg, about 35 mg to about 55 mg, about 35 mg to about 50 mg, about 35 mg to about 45 mg, about 35 mg to about 40 mg, about 40 mg to about 100 mg, about 40 mg to about 95 mg, about 40 mg to about 90 mg, about 40 mg to about 85 mg, about 40 mg to about 80 mg, about 40 mg to about 75 mg, about 40 mg to about 70 mg, about 40 mg to about 65 mg, about 40 mg to about 60 mg, about 40 mg to about 55 mg, about 40 mg to about 50 mg, about 40 mg to about 45 mg, about 45 mg to about 100 mg, about 45 mg to about 95 mg, about 45 mg to about 90 mg, about 45 mg to about 85 mg, about 45 mg to about 80 mg, about 45 mg to about 75 mg, about 45 mg to about 70 mg, about 45 mg to about 65 mg, about 45 mg to about 60 mg, about 45 mg to about 55 mg, about 45 mg to about 50 mg, about 50 mg to about 100 mg, about 50 mg to about 95 mg, about 50 mg to about 90 mg, about 50 mg to about 85 mg, about 50 mg to about 80 mg, about 50 mg to about 75 mg, about 50 mg to about 70 mg, about 50 mg to about 65 mg, about 50 mg to
about 60 mg, about 50 mg to about 55 mg, about 55 mg to about 100 mg, about 55 mg to about 95 mg, about 55 mg to about 90 mg, about 55 mg to about 85 mg, about 55 mg to about 80 mg, about 55 mg to about 75 mg, about 55 mg to about 70 mg, about 55 mg to about 65 mg, about 55 mg to about 60 mg, about 60 mg to about 100 mg, about 60 mg to about 95 mg, about 60 mg to about 90 mg, about 60 mg to about 85 mg, about 60 mg to about 80 mg, about 60 mg to about 75 mg, about 60 mg to about 70 mg, about 60 mg to about 65 mg, about 65 mg to about 100 mg, about 65 mg to about 95 mg, about 65 mg to about 90 mg, about 65 mg to about 85 mg, about 65 mg to about 80 mg, about 65 mg to about 75 mg, about 65 mg to about 70 mg, about 70 mg to about 100 mg, about 70 mg to about 95 mg, about 70 mg to about 90 mg, about 70 mg to about 85 mg, about 70 mg to about 80 mg, about 70 mg to about 75 mg, about 75 mg to about 100 mg, about 75 mg to about 95 mg, about 75 mg to about 90 mg, about 75 mg to about 85 mg, about 75 mg to about 80 mg, about 80 mg to about 100 mg, about 80 mg to about 95 mg, about 80 mg to about 90 mg, about 80 mg to about 85 mg, about 85 mg to about 100 mg, about 85 mg to about 95 mg, about 85 mg to about 90 mg, about 90 mg to about 100 mg, about 90 mg to about 95 mg, about 95 mg to about 100 mg, about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 95 mg, or about 100 mg. In some cases, a compound is selected from compounds 2, 3, 4, 14, 25, and 74. In some embodiments, a compound of Formula (I), (I-A), or (I-B) is orally administered once a day (QD) on a daily basis during a period of time. In some embodiments, a compound of Formula (I), (I-A), or (I-B) is orally administered twice a day (BID) on a daily basis during a period of time. In one embodiment, a compound of Formula (I), (I-A), or (I-B) is orally administered in the amount of about 20 mg to about 500 mg (e.g., about 20 mg to about 480 mg, about 20 mg to about 460 mg, about 20 mg to about 440 mg, about 20 mg to about 420 mg, about 20 mg to about 400 mg, about 20 mg to about 380 mg, about 20 mg to about 360 mg, about 20 mg to about 340 mg, about 20 mg to about 320 mg, about 20 mg to about 300 mg, about 20 mg to about 280 mg, about 20 mg to about 260 mg, about 20 mg to about 240 mg, about 20 mg to about 220 mg, about 20 mg to about 200 mg, about 20 mg to about 180 mg, about 20 mg to about 160 mg, about 20 mg to about 140 mg, about 20 mg to about 120 mg, about 20 mg to about 100 mg, about 20 mg to about 80 mg, about 20 mg to about 60 mg, about 20 mg to about 40 mg, about 40 mg to about 500 mg, about 40 mg to about 480 mg, about 40 mg to about 460 mg, about 40 mg to about 440 mg, about 40 mg to about 420 mg, about 40 mg to about 400 mg, about 40 mg to about 380 mg, about 40 mg to about 360 mg, about 40 mg to about 340 mg, about 40 mg to about 320 mg, about 40 mg to about 300 mg, about 40 mg to about 280 mg, about 40 mg to about 260 mg, about 40 mg to about 240 mg, about 40 mg to about 220 mg, about 40 mg to about 200 mg, about 40 mg to about 180 mg, about 40 mg to about 160
mg, about 40 mg to about 140 mg, about 40 mg to about 120 mg, about 40 mg to about 100 mg, about 40 mg to about 80 mg, about 40 mg to about 60 mg, about 60 mg to about 500 mg, about 60 mg to about 480 mg, about 60 mg to about 460 mg, about 60 mg to about 440 mg, about 60 mg to about 420 mg, about 60 mg to about 400 mg, about 60 mg to about 380 mg, about 60 mg to about 360 mg, about 60 mg to about 340 mg, about 60 mg to about 320 mg, about 60 mg to about 300 mg, about 60 mg to about 280 mg, about 60 mg to about 260 mg, about 60 mg to about 240 mg, about 60 mg to about 220 mg, about 60 mg to about 200 mg, about 60 mg to about 180 mg, about 60 mg to about 160 mg, about 60 mg to about 140 mg, about 60 mg to about 120 mg, about 60 mg to about 100 mg, about 60 mg to about 80 mg, about 80 mg to about 500 mg, about 80 mg to about 480 mg, about 80 mg to about 460 mg, about 80 mg to about 440 mg, about 80 mg to about 420 mg, about 80 mg to about 400 mg, about 80 mg to about 380 mg, about 80 mg to about 360 mg, about 80 mg to about 340 mg, about 80 mg to about 320 mg, about 80 mg to about 300 mg, about 80 mg to about 280 mg, about 80 mg to about 260 mg, about 80 mg to about 240 mg, about 80 mg to about 220 mg, about 80 mg to about 200 mg, about 80 mg to about 180 mg, about 80 mg to about 160 mg, about 80 mg to about 140 mg, about 80 mg to about 120 mg, about 80 mg to about
100 mg, about 100 mg to about 500 mg, about 100 mg to about 480 mg, about 100 mg to about 460 mg, about 100 mg to about 440 mg, about 100 mg to about 420 mg, about 100 mg to about 400 mg, about 100 mg to about 380 mg, about 100 mg to about 360 mg, about 100 mg to about 340 mg, about 100 mg to about 320 mg, about 100 mg to about 300 mg, about 100 mg to about 280 mg, about 100 mg to about 260 mg, about 100 mg to about 240 mg, about 100 mg to about 220 mg, about 100 mg to about 200 mg, about 100 mg to about 180 mg, about 100 mg to about 160 mg, about 100 mg to about 140 mg, about 100 mg to about 120 mg, about 120 mg to about 500 mg, about 120 mg to about 480 mg, about 120 mg to about 460 mg, about 120 mg to about 440 mg, about 120 mg to about 420 mg, about 120 mg to about 400 mg, about 120 mg to about 380 mg, about 120 mg to about 360 mg, about 120 mg to about 340 mg, about 120 mg to about 320 mg, about 120 mg to about 300 mg, about 120 mg to about 280 mg, about 120 mg to about 260 mg, about 120 mg to about 240 mg, about 120 mg to about 220 mg, about 120 mg to about 200 mg, about 120 mg to about 180 mg, about 120 mg to about 160 mg, about 120 mg to about 140 mg, about 140 mg to about 500 mg, about 140 mg to about 480 mg, about 140 mg to about 460 mg, about 140 mg to about 440 mg, about 140 mg to about 420 mg, about 140 mg to about 400 mg, about 140 mg to about 380 mg, about 140 mg to about 360 mg, about 140 mg to about 340 mg, about 140 mg to about 320 mg, about 140 mg to about 300 mg, about 140 mg to about 280 mg, about 140 mg to about 260 mg, about 140 mg to about 240 mg, about 140 mg to about 220 mg, about 140 mg to about 200 mg, about 140 mg to about 180 mg, about 140 mg to about 160 mg, about 160 mg to about 500 mg, about 160 mg to about 480 mg, about 160 mg to about
mg, about 160 mg to about 440 mg, about 160 mg to about 420 mg, about 160 mg to about mg, about 160 mg to about 380 mg, about 160 mg to about 360 mg, about 160 mg to about mg, about 160 mg to about 320 mg, about 160 mg to about 300 mg, about 160 mg to about mg, about 160 mg to about 260 mg, about 160 mg to about 240 mg, about 160 mg to about mg, about 160 mg to about 200 mg, about 160 mg to about 180 mg, about 180 mg to about mg, about 180 mg to about 480 mg, about 180 mg to about 460 mg, about 180 mg to about mg, about 180 mg to about 420 mg, about 180 mg to about 400 mg, about 180 mg to about mg, about 180 mg to about 360 mg, about 180 mg to about 340 mg, about 180 mg to about mg, about 180 mg to about 300 mg, about 180 mg to about 280 mg, about 180 mg to about mg, about 180 mg to about 240 mg, about 180 mg to about 220 mg, about 180 mg to about mg, about 200 mg to about 500 mg, about 200 mg to about 480 mg, about 200 mg to about mg, about 200 mg to about 440 mg, about 200 mg to about 420 mg, about 200 mg to about mg, about 200 mg to about 380 mg, about 200 mg to about 360 mg, about 200 mg to about mg, about 200 mg to about 320 mg, about 200 mg to about 300 mg, about 200 mg to about mg, about 200 mg to about 260 mg, about 200 mg to about 240 mg, about 200 mg to about mg, about 220 mg to about 500 mg, about 220 mg to about 480 mg, about 220 mg to about mg, about 220 mg to about 440 mg, about 220 mg to about 420 mg, about 220 mg to about mg, about 220 mg to about 380 mg, about 220 mg to about 360 mg, about 220 mg to about mg, about 220 mg to about 320 mg, about 220 mg to about 300 mg, about 220 mg to about mg, about 220 mg to about 260 mg, about 220 mg to about 240 mg, about 240 mg to about mg, about 240 mg to about 480 mg, about 240 mg to about 460 mg, about 240 mg to about mg, about 240 mg to about 420 mg, about 240 mg to about 400 mg, about 240 mg to about mg, about 240 mg to about 360 mg, about 240 mg to about 340 mg, about 240 mg to about mg, about 240 mg to about 300 mg, about 240 mg to about 280 mg, about 240 mg to about mg, about 260 mg to about 500 mg, about 260 mg to about 480 mg, about 260 mg to about mg, about 260 mg to about 440 mg, about 260 mg to about 420 mg, about 260 mg to about mg, about 260 mg to about 380 mg, about 260 mg to about 360 mg, about 260 mg to about mg, about 260 mg to about 320 mg, about 260 mg to about 300 mg, about 260 mg to about mg, about 280 mg to about 500 mg, about 280 mg to about 480 mg, about 280 mg to about mg, about 280 mg to about 440 mg, about 280 mg to about 420 mg, about 280 mg to about mg, about 280 mg to about 380 mg, about 280 mg to about 360 mg, about 280 mg to about mg, about 280 mg to about 320 mg, about 280 mg to about 300 mg, about 300 mg to about mg, about 300 mg to about 480 mg, about 300 mg to about 460 mg, about 300 mg to about mg, about 300 mg to about 420 mg, about 300 mg to about 400 mg, about 300 mg to about mg, about 300 mg to about 360 mg, about 300 mg to about 340 mg, about 300 mg to about
320 mg, about 320 mg to about 500 mg, about 320 mg to about 480 mg, about 320 mg to about
460 mg, about 320 mg to about 440 mg, about 320 mg to about 420 mg, about 320 mg to about
400 mg, about 320 mg to about 380 mg, about 320 mg to about 360 mg, about 320 mg to about
340 mg, about 340 mg to about 500 mg, about 340 mg to about 480 mg, about 340 mg to about
460 mg, about 340 mg to about 440 mg, about 340 mg to about 420 mg, about 340 mg to about
400 mg, about 340 mg to about 380 mg, about 340 mg to about 360 mg, about 360 mg to about
500 mg, about 360 mg to about 480 mg, about 360 mg to about 460 mg, about 360 mg to about
440 mg, about 360 mg to about 420 mg, about 360 mg to about 400 mg, about 360 mg to about
380 mg, about 380 mg to about 500 mg, about 380 mg to about 480 mg, about 380 mg to about
460 mg, about 380 mg to about 440 mg, about 380 mg to about 420 mg, about 380 mg to about
400 mg, about 400 mg to about 500 mg, about 400 mg to about 480 mg, about 400 mg to about
460 mg, about 400 mg to about 440 mg, about 400 mg to about 420 mg, about 420 mg to about
500 mg, about 420 mg to about 480 mg, about 420 mg to about 460 mg, about 420 mg to about
440 mg, about 440 mg to about 500 mg, about 440 mg to about 480 mg, about 440 mg to about
460 mg, about 460 mg to about 500 mg, about 460 mg to about 480 mg, about 480 mg to about
500 mg, about 25, about 50, about 75, about 100, about 150, about 200, about 250, about 300, about 350, about 400, about 450, or about 500 mg), during a period of time, and i.v. administration of an inhibitor, wherein the inhibitor is selected from:
1) immunomodulator inhibitor;
2) PD-1 inhibitor;
3) PD-L1 inhibitor; and
4) CTLA-4 inhibitor; wherein the inhibitor is administered, for example, once a week, once every two weeks, once every three weeks, or once every four weeks, depending on the dosage. In some cases, the compound of Formula (I), (I- A), or (I-B) is selected compound 2, or a pharmaceutically acceptable salt of any one thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 3, or a pharmaceutically acceptable salt of any one thereof. In some cases, the compound of Formula (I), (I-A), or (I-B)is compound 4, or a pharmaceutically acceptable salt of any one thereof. In some cases, the compound of Formula (I), (I-A), or (I-B)is compound 25, or a pharmaceutically acceptable salt of any one thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74 or a pharmaceutically acceptable salt of any one thereof. Included in the present disclosure are salts, particularly pharmaceutically acceptable salts, of the compounds described herein. The compounds of the present invention that possess a sufficiently acidic, a sufficiently basic, or both functional groups, can react with any of a number of inorganic bases, and inorganic and organic acids, to form a salt. Alternatively, compounds that are inherently charged, such as those with a quaternary nitrogen,
can form a salt with an appropriate counterion, e.g., a halide such as bromide, chloride, or fluoride, particularly bromide.
[00275] Chemical entities having carbon-carbon double bonds or carbon-nitrogen double bonds may exist in Z- or E- form (or cis- or trans- form). Furthermore, some chemical entities may exist in various tautomeric forms. Unless otherwise specified, compounds described herein are intended to include all Z-, E- and tautomeric forms as well.
[00276] A “tautomer” refers to a molecule wherein a proton shift from one atom of a molecule to another atom of the same molecule is possible. The compounds presented herein, in certain embodiments, exist as tautomers. In circumstances where tautomerization is possible, a chemical equilibrium of the tautomers will exist. The exact ratio of the tautomers depends on several factors, including physical state, temperature, solvent, and pH. Some examples of tautomeric equilibrium include:
[00277] The compounds disclosed herein, in some embodiments, are used in different enriched isotopic forms, e.g., enriched in the content of 2H, 3H, nC, 13C and/or 14C. In one particular embodiment, the compound is deuterated in at least one position. Such deuterated forms can be made by the procedure described in U.S. Patent Nos. 5,846,514 and 6,334,997. As described in U.S. Patent Nos. 5,846,514 and 6,334,997, deuteration can improve the metabolic stability and or efficacy, thus increasing the duration of action of drugs.
[00278] Unless otherwise stated, compounds described herein are intended to include compounds which differ only in the presence of one or more isotopically enriched atoms. For example, compounds having the present structures except for the replacement of a hydrogen by a deuterium or tritium, or the replacement of a carbon by 13C- or 14C-enriched carbon are within the scope of the present disclosure.
[00279] The compounds of the present disclosure optionally contain unnatural proportions of atomic isotopes at one or more atoms that constitute such compounds. For example, the compounds may be labeled with isotopes, such as for example, deuterium (2H), tritium (3H), iodine-125 (125I) or carbon-14 (14C). Isotopic substitution with 2H, nC, 13C, 14C, 15C, 12N, 13N, 15N, 16N, 16O, 17O, 14F, 15F, 16F, 17F, 18F, 33S, 34S, 35S, 36S, 35C1, 37C1, 79Br, 81Br, and 125I are all contemplated. All isotopic variations of the compounds of the present invention, whether radioactive or not, are encompassed within the scope of the present invention.
[00280] In certain embodiments, the compounds disclosed herein have some or all of the JH atoms replaced with 2H atoms. The methods of synthesis for deuterium-containing compounds are known in the art and include, by way of non-limiting example only, the following synthetic methods.
[00281] Deuterium substituted compounds are synthesized using various methods such as described in: Dean, Dennis C.; Editor. Recent Advances in the Synthesis and Applications of Radiolabeled Compounds for Drug Discovery and Development. [In: Curr., Pharm. Des., 2000; 6(10)] 2000, 110 pp; George W.; Varma, Rajender S. The Synthesis of Radiolabeled Compounds via Organometallic Intermediates, Tetrahedron, 1989, 45(21), 6601-21; and Evans, E. Anthony. Synthesis of radiolabeled compounds, J. Radioanal. Chem., 1981, 64(1-2), 9-32.
[00282] Deuterated starting materials are readily available and are subjected to the synthetic methods described herein to provide for the synthesis of deuterium-containing compounds. Large numbers of deuterium-containing reagents and building blocks are available commercially from chemical vendors, such as Aldrich Chemical Co.
[00283] Compounds of the present invention also include crystalline and amorphous forms of those compounds, pharmaceutically acceptable salts, and active metabolites of these compounds having the same type of activity, including, for example, polymorphs, pseudopolymorphs, solvates, hydrates, unsolvated polymorphs (including anhydrates), conformational polymorphs, and amorphous forms of the compounds, as well as mixtures thereof.
[00284] The compounds described herein may in some cases exist as diastereomers, enantiomers, or other stereoisomeric forms. Where absolute stereochemistry is not specified, the compounds presented herein include all diastereomeric, enantiomeric, and epimeric forms as well as the appropriate mixtures thereof. Separation of stereoisomers may be performed by chromatography or by forming diastereomers and separating by recrystallization, or chromatography, or any combination thereof. (Jean Jacques, Andre Collet, Samuel H. Wilen, “Enantiomers, Racemates and Resolutions”, John Wiley And Sons, Inc., 1981, herein incorporated by reference for this disclosure). Stereoisomers may also be obtained by stereoselective synthesis.
[00285] The methods and compositions described herein include the use of amorphous forms as well as crystalline forms (also known as polymorphs). The compounds described herein may be in the form of pharmaceutically acceptable salts. As well, in some embodiments, active metabolites of these compounds having the same type of activity are included in the scope of the present disclosure. In addition, the compounds described herein can exist in unsolvated as well as solvated forms with pharmaceutically acceptable solvents such as water, ethanol, and the like. The solvated forms of the compounds presented herein are also considered to be disclosed herein.
[00286] In certain embodiments, compounds or salts of the compounds may be prodrugs, e.g., wherein a hydroxyl in the parent compound is presented as an ester or a carbonate, or carboxylic acid present in the parent compound is presented as an ester. The term “prodrug” is intended to encompass compounds which, under physiologic conditions, are converted into pharmaceutical agents of the present disclosure. One method for making a prodrug is to include one or more selected moieties which are hydrolyzed under physiologic conditions to reveal the desired molecule. In other embodiments, the prodrug is converted by an enzymatic activity of the host animal such as specific target cells in the host animal. For example, esters or carbonates (e.g., esters or carbonates of alcohols or carboxylic acids and esters of phosphonic acids) are preferred prodrugs of the present disclosure.
[00287] Prodrug forms of the herein described compounds, wherein the prodrug is metabolized in vivo to produce a compound as set forth herein are included within the scope of the claims. In some cases, some of the herein-described compounds may be a prodrug for another derivative or active compound.
[00288] Prodrugs are often useful because, in some situations, they may be easier to administer than the parent drug. They may, for instance, be bioavailable by oral administration whereas the parent is not. Prodrugs may help enhance the cell permeability of a compound relative to the parent drug. The prodrug may also have improved solubility in pharmaceutical compositions over the parent drug. Prodrugs may be designed as reversible drug derivatives, for use as modifiers to enhance drug transport to site-specific tissues or to increase drug residence inside of a cell.
[00289] In some embodiments, the design of a prodrug increases the lipophilicity of the pharmaceutical agent. In some embodiments, the design of a prodrug increases the effective water solubility. See, e.g., Fedorak et al., Am. J. Physiol., 269:G210-218 (1995); McLoed et al., Gastroenterol, 106:405-413 (1994); Hochhaus et al., Biomed. Chrom., 6:283-286 (1992); J. Larsen and H. Bundgaard, Int. J. Pharmaceutics, 37, 87 (1987); J. Larsen et al., Int. J.
Pharmaceutics, 47, 103 (1988); Sinkula et al., J. Pharm. Sci., 64:181-210 (1975); T. Higuchi and
V. Stella, Pro-drugs as Novel Delivery Systems, Vol. 14 of the A.C.S. Symposium Series; and Edward B. Roche, Bioreversible Carriers in Drug Design, American Pharmaceutical Association and Pergamon Press, 1987, all incorporated herein for such disclosure). According to another embodiment, the present disclosure provides methods of producing the above-defined compounds. The compounds may be synthesized using conventional techniques.
Advantageously, these compounds are conveniently synthesized from readily available starting materials.
[00290] Synthetic chemistry transformations and methodologies useful in synthesizing the compounds described herein are known in the art and include, for example, those described in R. Larock, Comprehensive Organic Transformations (1989); T. W. Greene and P. G. M. Wuts, Protective Groups in Organic Synthesis, 2d. Ed. (1991); L. Fieser and M. Fieser, Fieser and Fieser ’s Reagents for Organic Synthesis (1994); and L. Paquette, ed., Encyclopedia of Reagents for Organic Synthesis (1995).
Pharmaceutical Formulations
[00291] Provided herein, in certain embodiments, are compositions comprising a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), an immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor, or a pharmaceutically acceptable salt of any one thereof (also referred to herein as “a pharmaceutical agent”).
[00292] Pharmaceutical compositions may be formulated using one or more physiologically acceptable carriers including excipients and auxiliaries which facilitate processing of the pharmaceutical agent into preparations which are used pharmaceutically. Proper formulation is dependent upon the route of administration chosen. A summary of pharmaceutical compositions is found, for example, in Remington: The Science and Practice of Pharmacy, Nineteenth Ed (Easton, Pa., Mack Publishing Company, 1995); Hoover, John E., Remington’s Pharmaceutical Sciences, Mack Publishing Co., Easton, Pennsylvania 1975; Liberman, H.A. and Lachman, L., Eds., Pharmaceutical Dosage Forms, Marcel Decker, New York, N.Y., 1980; and Pharmaceutical Dosage Forms and Drug Delivery Systems, Seventh Ed. (Lippincott Williams & Wilkins, 1999). [00293] The compositions and methods of the present disclosure may be utilized to treat an individual in need thereof. In certain embodiments, the individual is a mammal such as a human, or a non-human mammal. When administered to an animal, such as a human, the composition or the pharmaceutical agent, is preferably administered as a pharmaceutical composition comprising, for example, a pharmaceutical agent and a pharmaceutically acceptable carrier or excipient. Pharmaceutically acceptable carriers are well known in the art and include, for
example, aqueous solutions such as water or physiologically buffered saline or other solvents or vehicles such as glycols, glycerol, oils such as olive oil, or injectable organic esters. In a preferred embodiment, when such pharmaceutical compositions are for human administration, particularly for invasive routes of administration, e.g., routes, such as injection or implantation, that circumvent transport or diffusion through an epithelial barrier, the aqueous solution is pyrogen-free, or substantially pyrogen-free. The excipients can be chosen, for example, to effect delayed release of an agent or to selectively target one or more cells, tissues or organs. The pharmaceutical composition can be in dosage unit form such as tablet, capsule, granule, lyophile for reconstitution, powder, solution, syrup, suppository, injection or the like. The composition can also be present in a transdermal delivery system, e.g., a skin patch. The composition can also be present in a solution suitable for topical administration, such as an eye drop.
[00294] A pharmaceutically acceptable excipient can contain physiologically acceptable agents that act, for example, to stabilize, increase solubility or to increase the absorption of a compound such as a pharmaceutical agent. Such physiologically acceptable agents include, for example, carbohydrates, such as glucose, sucrose or dextrans, antioxidants, such as ascorbic acid or glutathione, chelating agents, low molecular weight proteins or other stabilizers or excipients. The choice of a pharmaceutically acceptable excipient, including a physiologically acceptable agent, depends, for example, on the route of administration of the composition. The preparation or pharmaceutical composition can be a self emulsifying drug delivery system or a self microemulsifying drug delivery system. The pharmaceutical composition (preparation) also can be a liposome or other polymer matrix, which can have incorporated therein, for example, a compound of the invention. Liposomes, for example, which comprise phospholipids or other lipids, are nontoxic, physiologically acceptable and metabolizable carriers that are relatively simple to make and administer.
[00295] A pharmaceutical composition (preparation) can be administered to a subject by any of a number of routes of administration including, for example, orally, for example, drenches as in aqueous or non-aqueous solutions or suspensions, tablets, capsules, including sprinkle capsules and gelatin capsules, boluses, powders, granules, pastes for application to the tongue; absorption through the oral mucosa, e.g., sublingually; anally, rectally or vaginally, for example, as a pessary, cream or foam; parenterally, including intramuscularly, intravenously, subcutaneously or intrathecally as, for example, a sterile solution or suspension; nasally; intraperitoneally; subcutaneously; transdermally, for example, as a patch applied to the skin; and topically, for example, as a cream, ointment or spray applied to the skin, or as an eye drop. The compound may also be formulated for inhalation. In certain embodiments, a compound may be simply dissolved or suspended in sterile water.
[00296] A pharmaceutical composition may be a sterile aqueous or non-aqueous solution, suspension or emulsion, e.g., a microemulsion. The excipients described herein are examples and are in no way limiting. An effective amount or therapeutically effective amount refers to an amount of the one or more pharmaceutical agents administered to a subject, either as a single dose or as part of a series of doses, which is effective to produce a desired therapeutic effect. [00297] Subjects may generally be monitored for therapeutic effectiveness using assays and methods suitable for the condition being treated, which assays will be familiar to those having ordinary skill in the art and are described herein. Pharmacokinetics of a pharmaceutical agent, or one or more metabolites thereof, that is administered to a subject may be monitored by determining the level of the pharmaceutical agent or metabolite in a biological fluid, for example, in the blood, blood fraction, e.g., serum, and/or in the urine, and/or other biological sample or biological tissue from the subject. Any method practiced in the art and described herein to detect the agent may be used to measure the level of the pharmaceutical agent or metabolite during a treatment course.
[00298] The dose of a pharmaceutical agent described herein for treating a disease or disorder may depend upon the subject’s condition, that is, stage of the disease, severity of symptoms caused by the disease, general health status, as well as age, gender, and weight, and other factors apparent to a person skilled in the medical art. Pharmaceutical compositions may be administered in a manner appropriate to the disease to be treated as determined by persons skilled in the medical arts. In addition to the factors described herein and above related to use of pharmaceutical agent for treating a disease or disorder, suitable duration and frequency of administration of the pharmaceutical agent may also be determined or adjusted by such factors as the condition of the patient, the type and severity of the patient’s disease, the particular form of the active ingredient, and the method of administration. Optimal doses of an agent may generally be determined using experimental models and/or clinical trials. The optimal dose may depend upon the body mass, weight, or blood volume of the subject. The use of the minimum dose that is sufficient to provide effective therapy is usually preferred. Design and execution of pre- clinical and clinical studies for a pharmaceutical agent, including when administered for prophylactic benefit, described herein are well within the skill of a person skilled in the relevant art. When two or more pharmaceutical agents are administered to treat a disease or disorder, the optimal dose of each pharmaceutical agent may be different, such as less than when either agent is administered alone as a single agent therapy. In certain particular embodiments, two pharmaceutical agents in combination may act synergistically or additively, and either agent may be used in a lesser amount than if administered alone. An amount of a pharmaceutical agent that may be administered per day may be, for example, between about 0.01 mg/kg and 100 mg/kg,
e.g., between about 0.1 to 1 mg/kg, between about 1 to 10 mg/kg, between about 10-50 mg/kg, between about 50-100 mg/kg body weight. In other embodiments, the amount of a pharmaceutical agent that may be administered per day is between about 0.01 mg/kg and 1000 mg/kg, between about 100-500 mg/kg, or between about 500-1000 mg/kg body weight. The optimal dose, per day or per course of treatment, may be different for the disease or disorder to be treated and may also vary with the administrative route and therapeutic regimen.
[00299] Pharmaceutical compositions comprising a pharmaceutical agent can be formulated in a manner appropriate for the delivery method by using techniques routinely practiced in the art. The composition may be in the form of a solid, e.g., tablet, capsule, semi-solid, e.g., gel, liquid, or gas, e.g., aerosol. In other embodiments, the pharmaceutical composition is administered as a bolus infusion.
[00300] Pharmaceutical acceptable excipients are well known in the pharmaceutical art and described, for example, in Rowe et al., Handbook of Pharmaceutical Excipients: A Comprehensive Guide to Uses, Properties, and Safety, 5th Ed., 2006, and in Remington: The Science and Practice of Pharmacy (Gennaro, 21st Ed. Mack Pub. Co., Easton, PA (2005)). Exemplary pharmaceutically acceptable excipients include sterile saline and phosphate buffered saline at physiological pH. Preservatives, stabilizers, dyes, buffers, and the like may be provided in the pharmaceutical composition. In addition, antioxidants and suspending agents may also be used. In general, the type of excipient is selected based on the mode of administration, as well as the chemical composition of the active ingredient(s). Alternatively, compositions described herein may be formulated as a lyophilizate. A composition described herein may be lyophilized or otherwise formulated as a lyophilized product using one or more appropriate excipient solutions for solubilizing and/or diluting the pharmaceutical agent(s) of the composition upon administration. In other embodiments, the pharmaceutical agent may be encapsulated within liposomes using technology known and practiced in the art. In certain particular embodiments, a pharmaceutical agent is not formulated within liposomes for application to a stent that is used for treating highly, though not totally, occluded arteries. Pharmaceutical compositions may be formulated for any appropriate manner of administration described herein and in the art.
[00301] A pharmaceutical composition, e.g., for oral administration or for injection, infusion, subcutaneous delivery, intramuscular delivery, intraperitoneal delivery or other method, may be in the form of a liquid. A liquid pharmaceutical composition may include, for example, one or more of the following: a sterile diluent such as water, saline solution, preferably physiological saline, Ringer’s solution, isotonic sodium chloride, fixed oils that may serve as the solvent or suspending medium, polyethylene glycols, glycerin, propylene glycol or other solvents; antibacterial agents; antioxidants; chelating agents; buffers and agents for the adjustment of
tonicity such as sodium chloride or dextrose. A parenteral composition can be enclosed in ampoules, disposable syringes or multiple dose vials made of glass or plastic. The use of physiological saline is preferred, and an injectable pharmaceutical composition is preferably sterile. In another embodiment, for treatment of an ophthalmological condition or disease, a liquid pharmaceutical composition may be applied to the eye in the form of eye drops. A liquid pharmaceutical composition may be delivered orally.
[00302] For oral formulations, at least one of the pharmaceutical agents described herein can be used alone or in combination with appropriate additives to make tablets, powders, granules or capsules, and if desired, with diluents, buffering agents, moistening agents, preservatives, coloring agents, and flavoring agents. The pharmaceutical agents may be formulated with a buffering agent to provide for protection of the compound from low pH of the gastric environment and/or an enteric coating. A pharmaceutical agent included in a pharmaceutical composition may be formulated for oral delivery with a flavoring agent, e.g., in a liquid, solid or semi-solid formulation and/or with an enteric coating.
[00303] A pharmaceutical composition comprising any one of the pharmaceutical agents described herein may be formulated for sustained or slow release, also called timed release or controlled release. Such compositions may generally be prepared using well known technology and administered by, for example, oral, rectal, intradermal, or subcutaneous implantation, or by implantation at the desired target site. Sustained-release formulations may contain the compound dispersed in a carrier matrix and/or contained within a reservoir surrounded by a rate controlling membrane. Excipients for use within such formulations are biocompatible, and may also be biodegradable; preferably the formulation provides a relatively constant level of active component release. The amount of pharmaceutical agent contained within a sustained release formulation depends upon the site of implantation, the rate and expected duration of release, and the nature of the condition, disease or disorder to be treated or prevented.
[00304] In certain embodiments, the pharmaceutical compositions comprising a pharmaceutical agent are formulated for transdermal, intradermal, or topical administration. The compositions can be administered using a syringe, bandage, transdermal patch, insert, or syringelike applicator, as a powder/talc or other solid, liquid, spray, aerosol, ointment, foam, cream, gel, paste. This preferably is in the form of a controlled release formulation or sustained release formulation administered topically or injected directly into the skin adjacent to or within the area to be treated, e.g., intradermally or subcutaneously. The active compositions can also be delivered via iontophoresis. Preservatives can be used to prevent the growth of fungi and other microorganisms. Suitable preservatives include, but are not limited to, benzoic acid, butylparaben, ethyl paraben, methyl paraben, propylparaben, sodium benzoate, sodium
propionate, benzalkonium chloride, benzethonium chloride, benzyl alcohol, cetypyridinium chloride, chlorobutanol, phenol, phenylethyl alcohol, thimerosal, and combinations thereof. [00305] Pharmaceutical compositions comprising a pharmaceutical agent can be formulated as emulsions for topical application. An emulsion contains one liquid distributed in the body of a second liquid. The emulsion may be an oil-in-water emulsion or a water-in-oil emulsion. Either or both of the oil phase and the aqueous phase may contain one or more surfactants, emulsifiers, emulsion stabilizers, buffers, and other excipients. The oil phase may contain other oily pharmaceutically approved excipients. Suitable surfactants include, but are not limited to, anionic surfactants, non-ionic surfactants, cationic surfactants, and amphoteric surfactants. Compositions for topical application may also include at least one suitable suspending agent, antioxidant, chelating agent, emollient, or humectant.
[00306] Ointments and creams may, for example, be formulated with an aqueous or oily base with the addition of suitable thickening and/or gelling agents. Lotions may be formulated with an aqueous or oily base and will in general also contain one or more emulsifying agents, stabilizing agents, dispersing agents, suspending agents, thickening agents, or coloring agents. Liquid sprays may be delivered from pressurized packs, for example, via a specially shaped closure. Oil-in-water emulsions can also be used in the compositions, patches, bandages and articles. These systems are semisolid emulsions, micro-emulsions, or foam emulsion systems.
[00307] In some embodiments, the pharmaceutical agent described herein can be formulated as in inhalant. Inhaled methods can deliver medication directly to the airway. The pharmaceutical agent can be formulated as aerosols, microspheres, liposomes, or nanoparticles. The pharmaceutical agent can be formulated with solvents, gases, nitrates, or any combinations thereof. Compositions described herein are optionally formulated for delivery as a liquid aerosol or inhalable dry powder. Liquid aerosol formulations are optionally nebulized predominantly into particle sizes that can be delivered to the terminal and respiratory bronchioles. Liquid aerosol and inhalable dry powder formulations are preferably delivered throughout the endobronchial tree to the terminal bronchioles and eventually to the parenchymal tissue.
[00308] Aerosolized formulations described herein are optionally delivered using an aerosol forming device, such as a jet, vibrating porous plate or ultrasonic nebulizer, preferably selected to allow the formation of aerosol particles having with a mass medium average diameter predominantly between 1 to 5 p. Further, the formulation preferably has balanced osmolarity ionic strength and chloride concentration, and the smallest aerosolizable volume able to deliver effective dose of the pharmaceutical agent. Additionally, the aerosolized formulation preferably does not impair negatively the functionality of the airways and does not cause undesirable side effects.
[00309] Aerosolization devices suitable for administration of aerosol formulations described herein include, for example, jet, vibrating porous plate, ultrasonic nebulizers and energized dry powder inhalers, that are able to nebulize the formulation into aerosol particle size predominantly in the size range from 1-5 p. Predominantly in this application means that at least 70% but preferably more than 90% of all generated aerosol particles are within 1-5 p range. A jet nebulizer works by air pressure to break a liquid solution into aerosol droplets. Vibrating porous plate nebulizers work by using a sonic vacuum produced by a rapidly vibrating porous plate to extrude a solvent droplet through a porous plate. An ultrasonic nebulizer works by a piezoelectric crystal that shears a liquid into small aerosol droplets. A variety of suitable devices are available, including, for example, AeroNebTM and AeroDoseTM vibrating porous plate nebulizers (AeroGen, Inc., Sunnyvale, California), Sidestream® nebulizers (Medic-Aid Ltd., West Sussex, England), Pari LC® and Pari LC Star® jet nebulizers (Pari Respiratory Equipment, Inc., Richmond, Virginia), and AerosonicTM (DeVilbiss Medizinische Produkte (Deutschland) GmbH, Heiden, Germany) and UltraAire® (Omron Healthcare, Inc., Vernon Hills, Illinois) ultrasonic nebulizers.
[00310] In some embodiments, the pharmaceutical agent(s) can be formulated with oleaginous bases or ointments to form a semisolid composition with a desired shape. In addition to the pharmaceutical agent, these semisolid compositions can contain dissolved and/or suspended bactericidal agents, preservatives and/or a buffer system. A petrolatum component that may be included may be any paraffin ranging in viscosity from mineral oil that incorporates isobutylene, colloidal silica, or stearate salts to paraffin waxes. Absorption bases can be used with an oleaginous system. Additives may include cholesterol, lanolin (lanolin derivatives, beeswax, fatty alcohols, wool wax alcohols, low HLB (hydrophobellipophobe balance) emulsifiers, and assorted ionic and nonionic surfactants, singularly or in combination.
[00311] Controlled or sustained release transdermal or topical formulations can be achieved by the addition of time-release additives, such as polymeric structures, matrices, that are available in the art. For example, the compositions may be administered through use of hot-melt extrusion articles, such as bioadhesive hot-melt extruded film. The formulation can comprise a crosslinked polycarboxylic acid polymer formulation. A cross-linking agent may be present in an amount that provides adequate adhesion to allow the system to remain attached to target epithelial or endothelial cell surfaces for a sufficient time to allow the desired release of the compound.
[00312] An insert, transdermal patch, bandage or article can comprise a mixture or coating of polymers that provide release of the pharmaceutical agents at a constant rate over a prolonged period of time. In some embodiments, the article, transdermal patch or insert comprises water-
soluble pore forming agents, such as polyethylene glycol (PEG) that can be mixed with water insoluble polymers to increase the durability of the insert and to prolong the release of the active ingredients.
[00313] Transdermal devices (inserts, patches, bandages) may also comprise a water insoluble polymer. Rate controlling polymers may be useful for administration to sites where pH change can be used to effect release. These rate controlling polymers can be applied using a continuous coating film during the process of spraying and drying with the active compound. In one embodiment, the coating formulation is used to coat pellets comprising the active ingredients that are compressed to form a solid, biodegradable insert.
[00314] A polymer formulation can also be utilized to provide controlled or sustained release. Bioadhesive polymers described in the art may be used. By way of example, a sustained-release gel and the compound may be incorporated in a polymeric matrix, such as a hydrophobic polymer matrix. Examples of a polymeric matrix include a microparticle. The microparticles can be microspheres, and the core may be of a different material than the polymeric shell.
Alternatively, the polymer may be cast as a thin slab or film, a powder produced by grinding or other standard techniques, or a gel such as a hydrogel. The polymer can also be in the form of a coating or part of a bandage, stent, catheter, vascular graft, or other device to facilitate delivery of the pharmaceutical agent. The matrices can be formed by solvent evaporation, spray drying, solvent extraction and other methods known to those skilled in the art.
[00315] Kits with unit doses of one or more of the agents described herein, usually in oral or injectable doses, are provided. Such kits may include a container containing the unit dose, an informational package insert describing the use and attendant benefits of the drugs in treating disease, and optionally an appliance or device for delivery of the composition.
KITS AND PRODUCTS
Immunomodulator inhibitor combination Kit
[00316] Some embodiments relate to kits and products that include the compound or salt of Formula (I), (I-A), or (I-B) and/or an immunomodulator inhibitor. For example, the kit or product can include a package or container with a compound of Formula (I), (I-A), or (I-B) or pharmaceutically acceptable salt. Such kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound or salt of Formula (I), (I-A), or (I-B) in combination with an immunomodulator
inhibitor that is separately provided. The kits can be used in the methods of treating cancer as described herein.
[00317] In some embodiments, the kits or products can include both a compound or salt of Formula (I), (I-A), or (I-B) and an immunomodulator inhibitor.
[00318] Such kits can include one or more containers or packages, which include one or both combination drugs together in a single container and/or package, or in separate packages/containers. In some instances, the two drugs are separately wrapped, but included in a single package, container or box. Such kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound or salt of Formula (I), (I-A), or (I-B) in combination with an immunomodulator inhibitor. The kits can be used in the methods of treating cancer as described herein. In some cases, the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
PD-1 inhibitor combination Kit
[00319] Some embodiments relate to kits and products that include the compound or salt of Formula (I), (I-A), or (I-B) and/or at least one PD-1 inhibitor. For example, the kit or product can include a package or container with a compound or salt of Formula (I), (I-A), or (I-B). Such kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound or salt of Formula (I), (I-A), or (I-B) in combination with an PD-1 inhibitor that is separately provided. The kits can be used in the methods of treating cancer as described herein.
[00320] In some embodiments, the kits or products can include both a compound of Formula (I), (I-A), or (I-B) and at least one PD-1 inhibitor.
[00321] Such kits can include one or more containers or packages, which include one or both combination drugs together in a single container and/or package, or in separate packages/containers. In some instances, the two drugs are separately wrapped, but included in a single package, container or box. Such kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound or salt of Formula (I), (I-A), or (I-B) in combination with an PD-1 inhibitor. The kits can be used in the methods of treating cancer as described herein.
PD-L1 inhibitor combination Kit
[00322] Some embodiments relate to kits and products that include the compound of Formula (I), (I-A), or (I-B) and/or at least one PD-L1 inhibitor. For example, the kit or product can include a package or container with a compound or salt of Formula (I), (I-A), or (I-B) . Such kits and products can further include a product insert or label with approved drug administration and
indication information, including how to use the compound or salt of Formula (I), (I- A), or (I-B) in combination with an PD-L1 inhibitor that is separately provided. The kits can be used in the methods of treating cancer as described herein.
[00323] In some embodiments, the kits or products can include both a compound of Formula (I), (I- A), or (I-B) and at least one PD-L1 inhibitor.
[00324] Such kits can include one or more containers or packages, which include one or both combination drugs together in a single container and/or package, or in separate packages/containers. In some instances, the two drugs are separately wrapped, but included in a single package, container or box. Such kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound or salt of Formula (I), (I- A), or (I-B) in combination with an PD-L1 inhibitor. The kits can be used in the methods of treating cancer as described herein. In some cases, the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
CTLA-4 inhibitor combination Kit
[00325] Some embodiments relate to kits and products that include the compound of Formula (I), (I-A), or (I-B) and/or at least one CTLA-4 inhibitor. For example, the kit or product can include a package or container with a compound of Formula (I), (I-A), or (I-B). Such kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound of Formula (I), (I-A), or (I-B) in combination with an CTLA-4 inhibitor that is separately provided. The kits can be used in the methods of treating cancer as described herein.
[00326] In some embodiments, the kits or products can include both a compound or salt of Formula (I), (LA), or (I-B) and at least one CTLA-4 inhibitor.
[00327] Such kits can include one or more containers or packages, which include one or both combination drugs together in a single container and/or package, or in separate packages/containers. In some instances, the two drugs are separately wrapped, but included in a single package, container or box. Such kits and products can further include a product insert or label with approved drug administration and indication information, including how to use the compound of Formula (I), (I-A), or (LB) in combination with an CTLA-4 inhibitor. The kits can be used in the methods of treating cancer as described herein. In some cases, the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
DOSINGS
[00328] In some embodiments, the dosage regimen for the compounds herein (e.g, an immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutical composition of any one thereof) will vary depending upon known factors, such as the pharmacodynamic characteristics of the particular agent and its mode and route of administration; the species, age, sex, health, medical condition, and weight of the recipient; the nature and extent of the symptoms; the kind of concurrent treatment; the frequency of treatment; the route of administration, the renal and hepatic function of the patient, and the effect desired. In some embodiments, a clinical practitioner can determine and prescribe the effective amount of the drug required to prevent, counter, or arrest the progress of the disease or disorder. In some cases, depending on the patient’s condition and the intended therapeutic effect, the dosing frequency for the therapeutic agent may vary, for example, from once per day to six times per day. That is, the dosing frequency may be QD, i.e., once per day, BID, i.e., twice per day; TID, i.e., three times per day; QID, i.e., four times per day; five times per day, or six times per day. In another embodiment, dosing frequency may be BIW, i.e., twice weekly, TIW, i.e., three times a week, or QIW, i.e. four times a week. In some cases, depending on the patient’s condition and the intended therapeutic effect, the treatment cycle may have a period of time where no therapeutic agent is administered. As used herein, “interval administration” refers to administration of the therapeutic agent (e.g, an immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (LA), or (I-B)) followed by void days or void weeks. For example, the treatment cycle may be 3 weeks long which includes 2 weeks of dosing of the therapeutic agent(s) followed by 1 week where no therapeutic agent is administered. In some embodiments, the treatment cycle is 4 weeks long which includes 3 weeks of dosing followed by 1 week where no therapeutic agent is administered.
[00329] In some embodiments, the daily oral dosage of each active ingredient (e.g, immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (I- A), or (LB)), when used for the indicated effects, will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, an active ingredient (e.g, an immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (LA), or (LB)), may be administered at a dose of between about 10 mg/day and about 200 mg/day. In some embodiments, an active ingredient (e.g, an immunomodulator inhibitor, PD-1 inhibitor, PD-L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (LA), or (LB)), may be
administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day. The dose may be any value or subrange within the recited ranges.
[00330] The term “treatment cycle” as used herein, means a pre-determined period of time for administering the therapeutic agent (e.g, an immunomodulator inhibitor, PD-1 inhibitor, PD- L1 inhibitor, CTLA-4 inhibitor; or a compound or salt of Formula (I), (I-A), or (I-B)). In some embodiments, the patient is examined at the end of each treatment cycle to evaluate the effect of the therapy. In some cases, each of the treatment cycle has about 3 or more days. In some cases, each of the treatment cycle has from about 3 days to about 60 days. In some cases, each of the treatment cycle has from about 5 days to about 50 days. In some cases, each of the treatment cycle has from about 7 days to about 28 days. In some cases, each of the treatment cycle has 28 days. In some cases, the treatment cycle has about 29 days. In some cases, the treatment cycle has about 30 days. In some cases, the treatment cycle has about 31 days. In some cases, the treatment cycle has about a month-long treatment cycle. In some cases, the treatment cycle is any length of time from 3 weeks to 8 weeks. In some cases, the treatment cycle is any length of time from 3 weeks to 6 weeks. In some cases, the treatment cycle is 3 weeks. In some cases, the treatment cycle is one month. In some cases, the treatment cycle is 4 weeks. In some cases, the treatment cycle is 5 weeks. In some cases, the treatment cycle is 6 weeks. In some cases, the treatment cycle is 7 weeks. In some cases, the treatment cycle is 8 weeks. In some cases, the duration of the treatment cycle may include any value or subrange within the recited ranges, including endpoints.
[00331] In some embodiments, drugs are administered at the maximum tolerated dose (“MTD”), which is the highest dose of drug that does not cause unacceptable side effects.
Immunomodulator inhibitor Dosing
[00332] In some embodiments, the immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof is administered prior to administration of the compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof.
[00333] In some embodiments, the immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof, is administered after administration of the compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof.
[00334] In some embodiments, the immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof, is administered at about the same time as administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof or pharmaceutical composition thereof. In some embodiments, separate administration of each inhibitor/compound, at different times and by different routes, may be advantageous. In some cases, the components in the combination i.e. compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof, and the immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, or pharmaceutical composition thereof, need not be necessarily administered at essentially the same time or in any order.
[00335] In some embodiments, a pharmaceutical composition comprises an immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient. In some cases, a pharmaceutical composition comprises a compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient. In some cases, the pharmaceutical compositions may be used in the methods simultaneously, separately, or sequentially. In some cases, a pharmaceutical composition comprises an immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, and/or a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, for use in the methods for simultaneous, separate or sequential use. [00336] In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are each dosed at their respective MTDs. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD and the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed in an amount less than its MTD. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at an amount less than its MTD and the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD. In some cases, the compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at less than their respective MTDs. In some cases, the administration can be so timed that the peak pharmacokinetic effect of one compound coincides with the peak pharmacokinetic effect of the other.
[00337] In some embodiments, the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is administered QD. In some cases, the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are administered BID. In some cases, the immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, of the invention are administered TID.
[00338] In some embodiments, a single dose of compound or salt of Formula (I), (I- A), or (I- B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each administered once daily.
[00339] In some embodiments, the therapeutically effective amount of the immunomodulator inhibitor of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, the therapeutically effective amount of the immunomodulator inhibitor of the combination will range between about 10 mg/day and about 200 mg/day. In some embodiments, the therapeutically effective amount of the immunomodulator inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day. The therapeutically effective amount of the immunomodulator inhibitor of the combination may be any value or subrange within the recited ranges.
[00340] In some embodiments, the therapeutically effective amount of the immunomodulator inhibitor of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, the therapeutically effective amount of the immunomodulator inhibitor of the combination will range between about 0.1 to about 50 mg/kg of body weight. In some embodiments the therapeutically effective amount of the immunomodulator inhibitor of the combination will range between about 10 mg/day and about 200 mg/day. In some embodiments, the therapeutically effective amount of the immunomodulator inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day. The therapeutically effective amount of the
immunomodulator inhibitor of the combination may be any value or subrange within the recited ranges.
[00341] In some embodiments, the immunomodulator inhibitor, or a pharmaceutically acceptable salt thereof, and the compound of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof, can be formulated into separate or individual dosage forms which can be co-administered one after the other. In some embodiments, if the route of administration is the same (e.g. oral) two active compounds can be formulated into a single form for coadministration, both methods of co-administration, however, being part of the same therapeutic treatment or regimen. In some embodiments, the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time). In some embodiments, the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time). In some embodiments, the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time).
[00342] In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 5 mg to about 600 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg to about 150 mg. In some embodiments, the immunomodulator inhibitor is administered to a subject at about 10 mg to about 125 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 440 mg to about 520 mg. In some embodiments, the immunomodulator inhibitor is administered to a subject at about 10 mg to about 100 mg. In some embodiments, the immunomodulator inhibitor is administered to a subject at about 25 mg to about 100 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 50 mg to about 100 mg. In some embodiments, the immunomodulator inhibitor, or
pharmaceutically salt thereof of is administered to a subject at about 5 mg to about 75 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, about 150 mg, about 170 mg, about 190 mg, about 210 mg, about 240 mg, about 300 mg, about 350 mg, about 400 mg, about 420 mg, about 440 mg, about 460 mg, about 480 mg, about 500 mg, about 540 mg, about 580 mg, or about 650 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg, about 30 mg, about 45 mg, or about 60 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 30 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 45 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 60 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 240 mg. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered to a subject at about 480 mg. In some embodiments, the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
[00343] In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered once daily. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered twice daily. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered 3 times daily. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered once weekly. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered every other day. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered every 3 days. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered once a week. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered every two weeks. In some embodiments, the immunomodulator inhibitor, or pharmaceutically salt thereof is administered every 4 weeks.
[00344] In some embodiments, the immunomodulator inhibitor is administered intravenously in the amount of about 800 mg every 2 weeks (Q2W) or about 10 mg/kg every 2 weeks (Q2W). In one embodiment, the immunomodulator inhibitor, is administered intravenously over 60 minutes.
[00345] In some embodiments, the immunomodulator inhibitor, is administered at a dose of 1200 mg intravenously once every 3 weeks (Q3W) or at a dose of 840 mg intravenously two weeks apart. In some cases, the immunomodulator inhibitor, is administered intravenously over 60 minutes.
[00346] In some embodiments, the immunomodulator inhibitor, is administered at a dose of 10 mg/kg intravenously once every 2 weeks (Q2W). In one embodiment, the immunomodulator inhibitor, is administered intravenously over 60 minutes.
PD-1 inhibitor Dosing
[00347] In some embodiments, a pharmaceutical composition comprises a PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient. In some cases, a pharmaceutical composition comprises a compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient. In some cases, the pharmaceutical compositions may be used in the methods simultaneously, separately, or sequentially.
[00348] In some embodiments, a pharmaceutical composition comprises a PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, and/or a compound or salt of Formula (I), (I-A), or (I- B), or a pharmaceutically acceptable salt thereof, for use in the methods which may be for simultaneous, separate or sequential use.
[00349] In some cases, the PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, is administered prior to administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof. In another embodiment, the PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, is administered after administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof. In another embodiment, the PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, is administered at about the same time as administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof or a pharmaceutical composition thereof. In some embodiments, separate administration of each inhibitor, at different times and by different routes, may be advantageous. In some cases, the
components in the combination i.e. compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, and the PD-1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, may need not to be administered at essentially the same time or in any order.
[00350] In some embodiments, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are each dosed at their respective MTDs. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD and the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed in an amount less than its MTD. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at an amount less than its MTD and the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at less than their respective MTDs. In some cases, the administration can be so timed that the peak pharmacokinetic effect of one compound coincides with the peak pharmacokinetic effect of the other.
[00351] In some embodiments, the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is administered QD. In some cases, the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are administered BID. In some cases, the PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, of the invention are administered TID.
[00352] In some embodiments, a single dose of compound or salt of Formula (I), (I-A), or (I- B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each administered once daily.
[00353] In some embodiments, the therapeutically effective amount of the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof) of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, a PD-1 inhibitor the therapeutically effective amount of the PD-1 inhibitor of the combination will range between about 10 mg/day and about 200 mg/day. In
some embodiments, the therapeutically effective amount of the PD-1 inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day. The therapeutically effective amount of the PD-1 inhibitor of the combination may be any value or subrange within the recited ranges.
[00354] In some embodiments, the therapeutically effective amount of the PD-1 inhibitor
(e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof) of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, the therapeutically effective amount of the PD-1 inhibitor of the combination will range between about 0.1 to about 50 mg/kg of body weight. In some embodiments, the therapeutically effective amount of the PD-1 inhibitor of the combination will range between about 10 mg/day and about 200 mg/day. In some embodiments, the therapeutically effective amount of the PD-1 inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day. The therapeutically effective amount of the PD-1 inhibitor of the combination may be any value or subrange within the recited ranges.
[00355] In some embodiments, the therapeutically effective amount of the PD-1 inhibitor
(e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof) of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per time, and most preferably between about 0.1 to about 20 mg/kg/time. In some embodiments, the therapeutically effective amount of the PD-1 inhibitor of the combination will range between about 0.1 to about 50 mg/kg of body weight. In some embodiments, the therapeutically effective amount of the PD-1 inhibitor of the combination will range between about 10 mg/time and about 200 mg/time. In some embodiments, the therapeutically effective amount of the PD-1 inhibitor of the combination may be administered at a dose of about 10 mg/time, 20 mg/time, 30 mg/time, 40 mg/time, 50 mg/time, 60 mg/time, 70 mg/time, 80 mg/time, 90 mg/time, 100 mg/time, 110 mg/time, 120 mg/time, 130 mg/time, 140 mg/time, 150 mg/time, 160 mg/time, 170 mg/time, 180 mg/time, 190 mg/time, 200 mg/time, 240 mg/time, 480 mg/time, 600 mg/time 1000 mg/time. The therapeutically effective amount of the PD-1 inhibitor of the combination may be any value or subrange within the recited ranges. In some cases, the time is 1 day. In some cases, the time is 7
days. In some cases, the time is 14 days. In some cases, the time is 21 days. In some cases, the time is 28 days.
[00356] In some embodiments, the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), or a pharmaceutically acceptable salt thereof, and the compound of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof, can be formulated into separate or individual dosage forms which can be co-administered one after the other. In some cases, if the route of administration is the same (e.g. oral) two active compounds can be formulated into a single form for co-administration, both methods of coadministration, however, being part of the same therapeutic treatment or regimen. In some embodiments, the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of a PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time).
[00357] In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 5 mg to about 600 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg to about 150 mg. In some embodiments, the PD-1 inhibitor is administered to a subject at about 10 mg to about 125 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 440 mg to about 520 mg. In some embodiments the PD-1 inhibitor is administered to a subject at about 10 mg to about 100 mg. In some embodiments, the PD-1 inhibitor is administered to a subject at about 25 mg to about 100 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 50 mg to about 100 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof of is administered to a subject at about 5 mg to about 75 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, about 150 mg, about 170 mg, about 190 mg, about 210 mg, about 240 mg, about 300 mg, about 350 mg, about 400 mg, about 420 mg, about 440 mg, about 460 mg, about 480 mg, about 500 mg, about 540 mg, about 580 mg, or about 650 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg , about 30 mg, about 45 mg, or about 60 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof
is administered to a subject at about 15 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 30 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 45 mg. In some embodiments, the PD-1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 60 mg. In some embodiments, the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
[00358] In some embodiments, the PD-1 inhibitor(e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), or pharmaceutically salt thereof is administered once daily. In some embodiments, the PD-1 inhibitor^. g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), is administered twice daily. In some embodiments, the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), or pharmaceutically salt thereof is administered 3 times daily. In some embodiments, the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), is administered once weekly. In some embodiments, the the PD-1 inhibitor(e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), is administered every other day. In some embodiments, the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), is administered every 3 days. In some embodiments, the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), is administered once a week. In some embodiments, the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), is administered every two weeks. In some embodiments, the PD-1 inhibitor (e.g., nivolumab, pembrolizumab, cemiplimab, tislelizumab, or a biosimilar thereof), is administered every 4 weeks.
[00359] In some embodiments, the PD-1 inhibitor is nivolumab or a biosimilar thereof. In one embodiment, nivolumab or a biosimilar thereof is administered at a dose of 240 mg intravenously once every 2 weeks (Q2W). In one embodiment, nivolumab or a biosimilar thereof is administered at a dose of 480 mg intravenously once every 4 weeks (Q4W). In one embodiment, nivolumab or a biosimilar thereof is administered intravenously over 30 minutes. [00360] In some embodiments, the PD-1 inhibitor is pembrolizumab or a biosimilar thereof. In one embodiment, pembrolizumab is administered at a dose of 200 mg intravenously once every 3 weeks (Q3W). In one embodiment, pembrolizumab or a biosimilar thereof is administered intravenously over 60 minutes.
[00361] In some embodiments, the PD-1 inhibitor is cemiplimab or a biosimilar thereof. In one embodiment, cemiplimab or a biosimilar thereof is administered at a dose of 350 mg intravenously once every 3 weeks (Q3W). In one embodiment, cemiplimab or a biosimilar thereof is administered intravenously over 30 minutes.
[00362] In some embodiments, the PD-1 inhibitor is tislelizumab or a biosimilar thereof. In one embodiment, tislelizumab or a biosimilar thereof is administered at a dose of 200 mg intravenously once every 3 weeks (Q3W).
[00363] In some embodiments, the therapeutically effective amount of pembrolizumab or biosimilar thereof in the combination is about 350 mg administered every three weeks.
[00364] In some embodiments, the PD-1 inhibitor is tislelizumab or a biosimilar thereof. In some embodiments, the therapeutically effective amount of tislelizumab, or biosimilar thereof, in the combination is about 200 mg administered every three weeks.
[00365] In some embodiments, the PD-1 inhibitor is atezolizumab or a biosimilar thereof. In some cases, the therapeutically effective amount of atezolizumab or biosimilar thereof in the combination is about 1200 mg administered every three weeks.
[00366] In some embodiments, the PD-1 inhibitor is the PD-L1 inhibitor is avelumab or a biosimilar thereof. In some cases, the therapeutically effective amount of avelumab or biosimilar thereof in the combination is about 10 mg/kg administered every two weeks or 800 mg every two weeks.
[00367] In some embodiments, the PD-L1 inhibitor is durvalumab or a biosimilar thereof. In some cases, the therapeutically effective amount of durvalumab or biosimilar thereof in the combination is about 10 mg/kg administered every two weeks.
PD-L1 inhibitor Dosing
[00368] In some embodiments, a pharmaceutical compositions comprise a PD-Ll inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient. In some cases, a pharmaceutical composition comprises a compound of Formula (II), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient. In some cases, the pharmaceutical compositions may be used in the methods simultaneously, separately, or sequentially.
[00369] In some embodiments, the pharmaceutical compositions comprise a PD-Ll inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), or a pharmaceutically acceptable salt thereof, and/or a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, for use in the methods may be for simultaneous,
separate or sequential use. In some cases, the PD-L1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, is administered prior to administration of the compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof. In another embodiment, the PD-L1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, is administered after administration of the compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof. In another embodiment, the PD-L1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, is administered at about the same time as administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof. In some embodiments, separate administration of each inhibitor, at different times and by different routes, may be advantageous. Thus, the components in the combination i.e. compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, and the PD- L1 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, may need not be necessarily administered at essentially the same time or in any order. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are each dosed at their respective MTDs. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD and the PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed in an amount less than its MTD. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at an amount less than its MTD and the PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at less than their respective MTDs. In some cases, the administration can be so timed that the peak pharmacokinetic effect of one compound coincides with the peak pharmacokinetic effect of the other.
[00370] In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is administered QD. In some cases, the PD-L1 inhibitor, or a
pharmaceutically acceptable salt or a pharmaceutical composition thereof, are administered BID. In some cases, the PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, of the invention are administered TID.
In some embodiments, a single dose of compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each administered once daily.
[00371] In some embodiments, the therapeutically effective amount of the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof) of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, a PD-L1 inhibitor the therapeutically effective amount of the PD-L1 inhibitor of the combination will range between about 10 mg/day and about 200 mg/day. In some embodiments, the therapeutically effective amount of the PD-L1 inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day. The therapeutically effective amount of the PD-L1 inhibitor of the combination may be any value or subrange within the recited ranges.
[00372] In some embodiments, the therapeutically effective amount of the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof) of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per time, and most preferably between about 0.1 to about 20 mg/kg/time. In some embodiments, the therapeutically effective amount of the PD-L1 inhibitor of the combination will range between about 0.1 to about 50 mg/kg of body weight. In some embodiments, the therapeutically effective amount of the PD-L1 inhibitor of the combination will range between about 10 mg/time and about 200 mg/time. In some embodiments, the therapeutically effective amount of the PD-L1 inhibitor of the combination may be administered at a dose of about 10 mg/time, 20 mg/time, 30 mg/time, 40 mg/time, 50 mg/time, 60 mg/time, 70 mg/time, 80 mg/time, 90 mg/time, 100 mg/time, 110 mg/time, 120 mg/time, 130 mg/time, 140 mg/time, 150 mg/time, 160 mg/time, 170 mg/time, 180 mg/time, 190 mg/time, 200 mg/time, 240 mg/time, 300 mg/time, 480 mg/time. The therapeutically effective amount of the PD-L1 inhibitor of the combination may be any value or subrange within the recited ranges. In some cases, the time is 1 day. In some cases, the time is 7 days. In some cases, the time is 14 days. In some cases, the time is 21 days. In some cases, the time is 28 days.
[00373] In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), or a pharmaceutically acceptable salt thereof, and the compound of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof, can be formulated into separate or individual dosage forms which can be co-administered one after the other. Another option is that if the route of administration is the same (e.g. oral) two active compounds can be formulated into a single form for co-administration, both methods of coadministration, however, being part of the same therapeutic treatment or regimen. In some embodiments, the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of a PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time).
[00374] In some embodiments, the PD-L1 inhibitor is administered to a subject at about 200 mg to about 500 mg. In some embodiments, PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 440 mg to about 520 mg. In some embodiments PD-L1 inhibitor is administered to a subject at about 10 mg to about 100 mg. In some embodiments, PD-L1 inhibitor is administered to a subject at about 240 mg to about 480 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 50 mg to about 100 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof of is administered to a subject at about 5 mg to about 75 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, about 150 mg, about 170 mg, about 190 mg, about 210 mg, about 240 mg, about 300 mg, about 350 mg, about 400 mg, about 420 mg, about 440 mg, about 460 mg, about 480 mg, about 500 mg, about 540 mg, about 580 mg, or about 650 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg , about 30 mg, about 45 mg, or about 60 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 30 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 240 mg. In some embodiments, the PD-L1 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 480 mg. In some embodiments, the subject is between 12 years old to 18
years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
[00375] In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), or pharmaceutically salt thereof is administered once daily. In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), is administered twice daily. In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), or pharmaceutically salt thereof is administered 3 times daily. In some embodiments, the PD-L1 inhibitor^. g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), is administered once weekly. In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), is administered every other day. In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), is administered every 3 days. In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), is administered once a week. In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), is administered every two weeks. In some embodiments, the PD-L1 inhibitor (e.g., atezolizumab, avelumab, durvalumab, or a biosimilar thereof), is administered every 4 weeks.
[00376] In some embodiments, the PD-L1 inhibitor is avelumab or a biosimilar thereof. In one embodiment, avelumab or a biosimilar thereof is administered intravenously in the amount of about 800 mg every 2 weeks (Q2W) or about 10 mg/kg every 2 weeks (Q2W). In one embodiment, avelumab or a biosimilar thereof is administered intravenously over 60 minutes. [00377] In some embodiments, the PD-L1 inhibitor is atezolizumab or a biosimilar thereof. In some cases, atezolizumab or a biosimilar thereof is administered at a dose of 1200 mg intravenously once every 3 weeks (Q3W) or at a dose of 840 mg intravenously two weeks apart. In some cases, atezolizumab or a biosimilar thereof is administered intravenously over 60 minutes.
[00378] In some embodiments, the PD-L1 inhibitor is durvalumab or a biosimilar thereof. In one embodiment, durvalumab or a biosimilar thereof is administered at a dose of 10 mg/kg intravenously once every 2 weeks (Q2W). In one embodiment, durvalumab or a biosimilar thereof is administered intravenously over 60 minutes.
CTLA-4 inhibitor Dosing
[00379] In some embodiments, a pharmaceutical composition comprises a CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient. In
some cases, a pharmaceutical composition comprises a compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, and a pharmaceutically acceptable excipient. In some cases, the pharmaceutical compositions may be used in the methods simultaneously, separately, or sequentially.
[00380] In some embodiments, the pharmaceutical compositions comprising a CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, and/or a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, for use in the methods may be for simultaneous, separate or sequential use.
[00381] In some embodiments, the CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, is administered prior to administration of the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof. In another embodiment, the CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, is administered after administration of the compound or salt of Formula (I), (LA), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof. In another embodiment, the CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof, is administered at about the same time as administration of the compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof. In some embodiments, separate administration of each inhibitor, at different times and by different routes, may be advantageous. In some cases, the components in the combination i.e. compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof and the CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, need not be necessarily administered at essentially the same time or in any order. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are each dosed at their respective MTDs. In some cases, the compound or salt of Formula (I), (I- A), or (I- B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD and the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed in an amount less than its MTD. In some cases, the compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at an amount less than its MTD and the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is dosed at its MTD. In some cases, the compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof and the CTLA-4 inhibitor, or a
pharmaceutically acceptable salt or a pharmaceutical composition thereof are each dosed at less than their respective MTDs. In some cases, the administration can be so timed that the peak pharmacokinetic effect of one compound coincides with the peak pharmacokinetic effect of the other.
[00382] In some embodiments, the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is administered QD. In some cases, the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are administered BID. In some cases, the CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, of the invention are administered TID.
[00383] In some embodiments, a single dose of compound or salt of Formula (I), (I- A), or (I- B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof are each administered once daily.
[00384] In some embodiments, the therapeutically effective amount of the CTLA-4 inhibitor of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, the therapeutically effective amount of the CTLA-4 inhibitor of the combination will range between about 10 mg/day and about 200 mg/day. In some cases, the therapeutically effective amount of the CTLA-4 inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day. The therapeutically effective amount of the CTLA-4 inhibitor of the combination may be any value or subrange within the recited ranges.
[00385] In some embodiments, the therapeutically effective amount of the CTLA-4 inhibitor of the combination will range between about 0.001 to about 1000 mg/kg of body weight, preferably between about 0.01 to about 100 mg/kg of body weight per day, and most preferably between about 0.1 to about 20 mg/kg/day. In some embodiments, the therapeutically effective amount of the CTLA-4 inhibitor of the combination will range between about 0.1 to about 50 mg/kg of body weight. In some embodiments, the therapeutically effective amount of the CTLA-4 inhibitor of the combination will range between about 10 mg/day and about 200 mg/day. In some embodiments, the therapeutically effective amount of the CTLA-4 inhibitor of the combination may be administered at a dose of about 10 mg/day, 20 mg/day, 30 mg/day, 40 mg/day, 50 mg/day, 60 mg/day, 70 mg/day, 80 mg/day, 90 mg/day, 100 mg/day, 110 mg/day, 120 mg/day, 130 mg/day, 140 mg/day, 150 mg/day, 160 mg/day, 170 mg/day, 180 mg/day, 190 mg/day, or 200 mg/day. The
therapeutically effective amount of the CTLA-4 inhibitor of the combination may be any value or subrange within the recited ranges.
[00386] In some embodiments, the CTLA-4 inhibitor, or a pharmaceutically acceptable salt thereof, and the compound of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt thereof, can be formulated into separate or individual dosage forms which can be co-administered one after the other. In some cases, if the route of administration is the same (e.g. oral) two active compounds can be formulated into a single form for co-administration, both methods of coadministration, however, being part of the same therapeutic treatment or regimen. In some embodiments, the combination therapy comprises oral administration of a compound of Formula (I), (I-A), or (I-B) once or twice a day on a daily basis (during a period of time), e.g., in an amount of about 10 mg to about 400 mg and oral administration of a CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof which is administered, for example once a day on a daily basis (during a period of time).
[00387] In some embodiments, the CTLA-4 inhibitor is administered to a subject at about 200 mg to about 500 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 440 mg to about 520 mg. In some embodiments, the CTLA-4 inhibitor is administered to a subject at about 10 mg to about 100 mg. In some embodiments, the CTLA-4 inhibitor is administered to a subject at about 240 mg to about 480 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 50 mg to about 100 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof of is administered to a subject at about 5 mg to about 75 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 10 mg, about 15 mg, about 20 mg, about 25 mg, about 30 mg, about 35 mg, about 40 mg, about 45 mg, about 50 mg, about 55 mg, about 60 mg, about 65 mg, about 70 mg, about 75 mg, about 80 mg, about 85 mg, about 90 mg, about 100 mg, about 105 mg, about 110 mg, about 115 mg, about 120 mg, about 125 mg, about 130 mg, about 135 mg, about 140 mg, about 145 mg, about 150 mg, about 170 mg, about 190 mg, about 210 mg, about 240 mg, about 300 mg, about 350 mg, about 400 mg, about 420 mg, about 440 mg, about 460 mg, about 480 mg, about 500 mg, about 540 mg, about 580 mg, or about 650 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg, about 30 mg, about 45 mg, or about 60 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 15 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 30 mg. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered to a subject at about 240 mg. In some embodiments, the CTLA-4 inhibitor, or
pharmaceutically salt thereof is administered to a subject at about 480 mg. In some embodiments, the subject is between 12 years old to 18 years old. In some embodiments, the subject is between greater than or equal 12 years old to less than or equal to 18 years. In some embodiments, the subject is an adult. In some embodiments, the subject is greater than or equal to 18 years old.
[00388] In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered once daily. In some embodiments, the CTLA-4 inhibitor, is administered twice daily. In some embodiments, the CTLA-4 inhibitor, or pharmaceutically salt thereof is administered 3 times daily. In some embodiments, the CTLA-4 inhibitor, is administered once weekly. In some embodiments, the CTLA-4 inhibitor, is administered every other day. In some embodiments, the CTLA-4 inhibitor, is administered every 3 days. In some embodiments, the CTLA-4 inhibitor, is administered once a week. In some embodiments, the CTLA-4 inhibitor, is administered every two weeks. In some embodiments, the CTLA-4 inhibitor, is administered every 4 weeks.
KRAS Inhibitor Dosing
[00389] In some embodiments, a single dose of compound or salt of Formula (II), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, is administered per day (i.e., in about 24 hour intervals) (i.e., QD). In some cases, two doses of the compound or salt of Formula (II), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are administered per day (i.e., BID). In some cases, three doses of the compound or salt of Formula (II), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, are administered per day (i.e., TID).
COMBINATION THERAPIES
Immunomodulator inhibitor combination therapy
[00390] In an aspect the present disclosure provides methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (LA), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00391] In an aspect the present disclosure provides methods of treating a disease or disorder in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (LA), or (LB), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00392] In an aspect the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In one embodiment, the cancer is a KRas G12D-associated cancer. In one embodiment, the cancer is a KRas G12V- associated cancer. In one embodiment, the cancer is a KRas wildtype-associated cancer. In some cases, the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the cancer is a solid tumor cancer. In some cases, the cancer is a solid tumor cancer with a KRAS mutation. In some cases, the cancer is a solid tumor cancer with KRAS wildtype. In some cases, the cancer is a solid tumor cancer with a G12D mutation. In some cases, the cancer is a solid tumor cancer with a G12V mutation. In some cases, the cancer is a KRAS wildtype- associated cancer. In some cases, the KRas G12D-associated cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the compound of Formula (I), (I- A), or (I-B) is selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of anyone thereof. In some cases, the compound of Formula (I), (I- A), or (I-B) is compound 2. In some cases, the compound of Formula (I), (I- A), or (I-B) is compound 3. In some cases, the compound of Formula (I), (I- A), or (I-B) is compound 4. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
[00393] In an aspect the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a combination of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (II) selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the compound of Formula (II) is compound 2. In some cases, the compound of Formula (II) is compound 3. In some cases, the compound of Formula (II) is compound 63. In some cases, the compound of Formula (II) is compound 4. In some cases, the compound of Formula (II) is compound 14. In some cases, the compound of Formula (II) is compound 25. In some cases, the compound of Formula (II) is compound 74.
[00394] In an aspect the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (I-A), or (I-B) selected
from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 2. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 3. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 4. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
[00395] In an aspect the present disclosure provides methods for increasing the sensitivity of a cancer cell to a compound or salt of Formula (I), (I-A), or (I-B), comprising contacting the cancer cell with an effective amount of a combination of a compound of Formula (I), (I-A), or (I- B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, wherein the immunomodulator inhibitor synergistically increases the sensitivity of the cancer cell to the compound or salt of Formula (I), (I-A), or (I-B). In some cases, the contacting is in vitro. In one embodiment, the contacting is in vivo.
[00396] In an aspect the present disclosure provides methods of inhibiting KRas G12 mutants or wildtype in a subject comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the method may inhibit KRas G12 mutants or wildtype activity in a cell. In some cases, inhibiting KRas G12 mutants or wildtype activity in a cell may include contacting the cell in which inhibition of KRas G12 mutants or wildtype activity is desired with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutical compositions containing the compound or pharmaceutically acceptable salt thereof and an immunomodulator inhibitor or a pharmaceutically acceptable salt or a pharmaceutical composition thereof.
[00397] In some cases, a cell in which inhibition of KRas G12 mutants or wildtype activity is desired is contacted with an effective amount of a compound of Formula (I), (I-A), or (I-B) or pharmaceutically acceptable salt of any one thereof and an immunomodulator inhibitor to negatively modulate the activity of KRas G12 mutants or wildtype. In some cases, by negatively modulating the activity of KRas G12 mutants or wildtype, the methods described herein are designed to inhibit undesired cellular proliferation resulting from enhanced KRas G12 mutants or wildtype activity within the cell. The cells may be contacted in a single dose or multiple doses in accordance with a particular treatment regimen to effect the desired negative modulation of
KRas G12 mutants or wildtype. The ability of compounds to bind KRas G12 mutants or wildtype may be monitored in vitro using well known methods.
[00398] In some embodiments, the inhibitory activity of compounds of Formula (I), (I- A), or (I-B) and an immunomodulator inhibitor in cells may be monitored, for example, by measuring the inhibition of KRas G12 mutants or wildtype activity of the amount of phosphorylated ERK. [00399] In another aspect, methods of treating cancer in a patient in need thereof, comprising administering to said patient a therapeutically effective amount of a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt thereof are provided and an immunomodulator inhibitor.
[00400] The compositions and methods provided herein may be used for the treatment of a KRas G12 mutants or wildtype-associated cancer in a patient in need thereof, comprising administering to the patient a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and an immunomodulator inhibitor are provided. In some cases, the KRas G12 mutants or wildtype associated cancer is lung cancer. In some cases, the KRas G12 mutants or wildtype associated cancer is solid tumor cancer. In some cases, the KRAS G12 mutants are selected from G12D and G12V. In some cases, the KRAS G12 mutants is G12D. In some cases, the KRAS G12 mutants is G12V.
[00401] In some embodiments, the method of treating a cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a immunomodulator inhibitor and a compound or salt of Formula (I), (I- A), or (I-B), synergistically increases the potency of the compound or salt of Formula (I), (I- A), or (I-B). In some cases, the synergistic increase in potency of the compound or salt of Formula (I), (I- A), or (I-B) results in an improved efficacy of the compound or salt of Formula (I), (I-A), or (I-B). [00402] In some embodiments, the therapeutically effective amount of a combination of two compounds is an amount that together synergistically increases the activity of the combination in comparison to the therapeutically effective amount of each compound in the combination, i.e., more than merely additive. For example, in vivo, the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a pharmaceutical composition thereof and a KRas G12D inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, results in a complete durable response in subjects relative to treatment with only the KRas G12D inhibitor. In one embodiment, the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a
pharmaceutical composition thereof and a KRas G12D inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, results in an increased duration of overall survival (“OS”) in subjects relative to treatment with only the KRas G12D inhibitor. In one embodiment, the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a pharmaceutical composition thereof and a KRas G12D inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, results in an increased duration of progression-free survival (“PFS”) in subjects relative to treatment with only the KRas G12D inhibitor.
[00403] In some embodiments, the therapeutically effective amount of a combination of two compounds is an amount that together synergistically increases the activity of the combination in comparison to the therapeutically effective amount of each compound in the combination, i.e., more than merely additive. For example, in vivo, the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a pharmaceutical composition thereof and a KRas G12V inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, results in a complete durable response in subjects relative to treatment with only the KRas G12V inhibitor. In one embodiment, the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a pharmaceutical composition thereof and a KRas G12V inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, results in an increased duration of overall survival (“OS”) in subjects relative to treatment with only the KRas G12V inhibitor. In one embodiment, the therapeutically effective amount of the combination of a PD-1/PD-L1 inhibitor or a pharmaceutical composition thereof and a KRas G12V inhibitor compound of Formula (I), Formula I-A, or Formula I-B or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, results in an increased duration of progression-free survival (“PFS”) in subjects relative to treatment with only the KRas G12V inhibitor.
[00404] In some embodiments, the compound and inhibitor are administered on separate days. In some cases, the compound and inhibitor are administered on the same day.
PD-1 inhibitor combination therapy
[00405] In an aspect the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a PD- 1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a
pharmaceutical composition thereof. In an aspect the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the cancer is a solid tumor cancer. In some cases, the cancer is a solid tumor cancer with a KRAS mutation. In some cases, the cancer is a KRAS wildtype-associated cancer. In one embodiment, the cancer is a KRas G12D-associated cancer. In one embodiment, the cancer is a KRas G12V-associated cancer. In one embodiment, the cancer is a KRas wildtype-associated cancer. In some cases, the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the cancer is a solid tumor cancer. In some cases, the cancer is a solid tumor cancer with a KRAS mutation. In some cases, the cancer is a solid tumor cancer with KRAS wildtype. In some cases, the cancer is a solid tumor cancer with a G12D mutation. In some cases, the cancer is a solid tumor cancer with a G12V mutation. In some cases, the cancer is a KRAS wildtype-associated cancer. In some cases, the KRas G12D-associated cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the compound of Formula (I), (I-A), or (I-B) is selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of anyone thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 2. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 3. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 4. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
[00406] In an aspect the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (I-A), or (I-B) selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 2. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 3. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 4. In some cases, the compound of Formula (I), (I- A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
[00407] In an aspect the present disclosure provides methods for increasing the sensitivity of a cancer cell to a compound or salt of Formula (I), (I-A), or (I-B), comprising contacting the cancer cell with an effective amount of a combination of a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, wherein the PD-1 inhibitor synergistically increases the sensitivity of the cancer cell to the compound or salt of Formula (I), (I-A), or (I-B). In some cases, the contacting is in vitro. In one embodiment, the contacting is in vivo.
[00408] In an aspect the present disclosure provides methods of inhibiting KRas G12 mutants in a subject comprising administering to the subject a therapeutically effective amount of a combination of a PD-1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the method may inhibit KRas G12 mutants or wildtype activity in a cell. In some cases, inhibiting KRas G12 mutants or wildtype activity in a cell may include contacting the cell in which inhibition of KRas G12 mutants or wildtype activity is desired with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutical compositions containing the compound or pharmaceutically acceptable salt of any one thereof and a PD-1 inhibitor. In some cases, the contacting is in vitro. In some cases, the contacting is in vivo. In some cases, a cell in which inhibition of KRas G12 mutants or wildtype activity is desired is contacted with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutically acceptable salt of any one thereof and a PD-1 inhibitor to negatively modulate the activity of KRas G12 mutants or wildtype. In some cases, by negatively modulating the activity of KRas G12 mutants or wildtype, the methods described herein are designed to inhibit undesired cellular proliferation resulting from enhanced KRas G12 mutants or wildtype activity within the cell. The cells may be contacted in a single dose or multiple doses in accordance with a particular treatment regimen to effect the desired negative modulation of KRas G12 mutants or wildtype. The ability of compounds to bind KRas G12 mutants or wildtype may be monitored in vitro using well known methods.
[00409] In some embodiments, the inhibitory activity of exemplary compounds and a PD-1 inhibitor in cells may be monitored, for example, by measuring the inhibition of KRas G12 mutants or wildtype activity of the amount of phosphorylated ERK.
[00410] In an aspect the present disclosure provides methods of reprogramming a tumor microenvironment (TME) of a subject in favor of antitumor immunity, wherein reprogramming includes modulating tumor cell cytokine/chemokine release in the tumor microenvironment of the subject. The method includes providing a compound described herein (a compound of
Formula (I, e.g., a compound selected from compounds 2, 3, 4, 14, 25, and 74). The method also includes providing an immunomodulator (e.g., checkpoint inhibitors (inhibitors targeting: cytotoxic T lymphocyte associated antigen 4 (CTLA-4), e.g. Yervoy/lpilimumab; programmed death 1/programmed death-ligand 1 (PD-1/PD-L1), e.g. Keytruda/pembrolizumab; and lymphocyte-activation gene 3 (LAG-3), e.g. BMS-986016/relatlimab)). The method can also include providing a compound of Formula (I) and an immunomodulator in combination (sequentially or simultaneously). In some cases, the subject has a KRas-associated cancer. [00411] In an aspect the present disclosure provides methods of treating a KRas G12D- associated cancer in a subject in need thereof, comprising administering to the subject a combination of an immunomodulator, and a compound of Formula (I) or a pharmaceutically acceptable salt thereof. In some cases, the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
[00412] In an aspect the present disclosure provides methods of treating a KRas G12V- associated cancer in a subject in need thereof, comprising administering to the subject a combination of an immunomodulator, and a compound of Formula (II) or a pharmaceutically acceptable salt thereof. In some cases, the compound is selected from compounds 2, 3, 4, 14, 25, and 74.
[00413] In another aspect, methods of treating cancer in a patient in need thereof, comprising administering to said patient a therapeutically effective amount of a compound or salt of Formula (I), (I-A), or (I-B) or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt thereof are provided and a PD-1 inhibitor.
[00414] The compositions and methods provided herein may be used for the treatment of a KRas G12 mutants or wildtype -associated cancer in a patient in need thereof, comprising administering to the patient a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and a PD-1 inhibitor are provided. In some cases, the KRas G12 mutants or wildtype associated cancer is lung cancer.
[00415] The compositions and methods provided herein may be used for the treatment of a KRas G12D-associated cancer in a patient in need thereof, comprising administering to the patient a therapeutically effective amount of a compound of Formula (I), (I-A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and a PD-1 inhibitor are provided.
[00416] In some embodiments, the method of treating a cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a PD-1 inhibitor and a compound or salt of Formula (I), (I-A), or (I-B), synergistically increases the potency of the compound or salt of Formula (I), (I-A), or (I-B). In some cases, the synergistic increase in potency of the compound or salt of Formula (I), (I-A), or (I-B) results in an improved efficacy of the compound or salt of Formula (I), (I-A), or (I-B).
[00417] In some embodiments, the compound and inhibitor are administered on separate days. In some cases, the compound and inhibitor are administered on the same day.
PD-L1 inhibitor combination therapy
[00418] In an aspect the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In an aspect the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the cancer is a solid tumor cancer. In some cases, the cancer is a solid tumor cancer with a KRAS mutation. In some cases, the cancer is a KRAS wildtype-associated cancer. In one embodiment, the cancer is a KRas G12D-associated cancer. In one embodiment, the cancer is a KRas G12V-associated cancer. In one embodiment, the cancer is a KRas wildtype-associated cancer. In some cases, the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the cancer is a solid tumor cancer. In some cases, the cancer is a solid tumor cancer with a KRAS mutation. In some cases, the cancer is a solid tumor cancer with KRAS wildtype. In some cases, the cancer is a solid tumor cancer with a G12D mutation. In some cases, the cancer is a solid tumor cancer with a G12V mutation. In some cases, the cancer is a KRAS wildtype-associated cancer. In some cases, the KRas G12D-associated cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the compound of Formula (I), (I-A), or (I-B) is selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of anyone thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 2. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 3. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 4. In
some cases, the compound of Formula (I), (I-A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
[00419] In an aspect the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (I-A), or (I-B) selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 2. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 3. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 4. In some cases, the compound of Formula (I), (I- A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74. [00420] In an aspect the present disclosure provides methods for increasing the sensitivity of a cancer cell to a compound or salt of Formula (I), (I-A), or (I-B), comprising contacting the cancer cell with an effective amount of a combination of a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, wherein the PD-L1 inhibitor synergistically increases the sensitivity of the cancer cell to the compound or salt of Formula (I), (I-A), or (I-B). In some cases, the contacting is in vitro. In one embodiment, the contacting is in vivo.
[00421] In an aspect the present disclosure provides methods of inhibiting KRas G12 mutants or wildtype in a subject comprising administering to the subject a therapeutically effective amount of a combination of a PD-L1 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the method may inhibit KRas G12 mutants or wildtype activity in a cell. In some cases, inhibiting KRas G12 mutants or wildtype activity in a cell may include contacting the cell in which inhibition of KRas G12 mutants or wildtype activity is desired with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutical compositions containing the compound or pharmaceutically acceptable salt of any one thereof and a PD-L1 inhibitor. In some cases, the contacting is in vitro. In some cases, the contacting is in vivo. In some cases, a cell in which inhibition of KRas G12 mutants or wildtype activity is desired is contacted with an effective amount of a compound of Formula (I), (I-A), or (I-B) or pharmaceutically acceptable salt of any one thereof and an PD-L1 inhibitor to negatively modulate the activity of KRas G12
mutants or wildtype.. In some cases, by negatively modulating the activity of KRas G12 mutants or wildtype, the methods described herein are designed to inhibit undesired cellular proliferation resulting from enhanced KRas G12 mutants or wildtype activity within the cell. The cells may be contacted in a single dose or multiple doses in accordance with a particular treatment regimen to effect the desired negative modulation of KRas G12 mutants or wildtype. The ability of compounds to bind KRas G12 mutants or wildtype may be monitored in vitro using well known methods.
[00422] In some embodiments, the inhibitory activity of exemplary compounds and an PD-L1 inhibitor in cells may be monitored, for example, by measuring the inhibition of KRAS G12 mutants and wildtype activity of the amount of phosphorylated ERK.
[00423] In another aspect, methods of treating cancer in a patient in need thereof, comprising administering to said patient a therapeutically effective amount of a compound or salt of Formula (I), (I-A), or (I-B) or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt thereof are provided and an PD- L1 inhibitor.
[00424] The compositions and methods provided herein may be used for the treatment of a KRas G12 mutants or wildtype-associated cancer in a patient in need thereof, comprising administering to the patient a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and a PD-L1 inhibitor are provided. In some cases, the KRas G12 mutants or wildtype associated cancer is lung cancer.
[00425] In some embodiments, the method of treating a cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a PD-L1 inhibitor and a compound or salt of Formula (I), (I-A), or (I-B), synergistically increases the potency of the compound or salt of Formula (I), (I-A), or (I-B). In some cases, the synergistic increase in potency of the compound or salt of Formula (I), (I-A), or (I-B) results in an improved efficacy of the compound or salt of Formula (I), (I-A), or (I-B).
[00426] In some embodiments, the compound and inhibitor are administered on separate days. In some cases, the compound and inhibitor are administered on the same day.
CTLA-4 inhibitor combination therapy
[00427] In an aspect the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical
composition thereof, and a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the cancer is a solid tumor cancer. In some cases, the cancer is a solid tumor cancer with a KRAS mutation. In some cases, the cancer is a KRAS wildtype-associated cancer. In one embodiment, the cancer is a KRas G12D-associated cancer. In one embodiment, the cancer is a KRas G12V- associated cancer. In one embodiment, the cancer is a KRas wildtype-associated cancer. In some cases, the cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the cancer is a solid tumor cancer. In some cases, the cancer is a solid tumor cancer with a KRAS mutation. In some cases, the cancer is a solid tumor cancer with KRAS wildtype. In some cases, the cancer is a solid tumor cancer with a G12D mutation. In some cases, the cancer is a solid tumor cancer with a G12V mutation. In some cases, the cancer is a KRAS wildtype- associated cancer. In some cases, the KRas G12D-associated cancer is pancreatic, colorectal, endometrial, and non-small cell lung cancer. In some cases, the compound of Formula (I), (I- A), or (I-B) is selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of anyone thereof. In some cases, the compound of Formula (I), (I- A), or (I-B) is compound 2. In some cases, the compound of Formula (I), (I- A), or (I-B) is compound 3. In some cases, the compound of Formula (I), (I- A), or (I-B) is compound 4. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74.
[00428] In an aspect the present disclosure provides methods of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound of Formula (I), (I-A), or (I-B) selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 2. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 3. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 4. In some cases, the compound of Formula (I), (I- A), or (I-B) is compound 14. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 25. In some cases, the compound of Formula (I), (I-A), or (I-B) is compound 74. [00429] In an aspect the present disclosure provides methods for increasing the sensitivity of a cancer cell to a compound or salt of Formula (I), (I-A), or (I-B), comprising contacting the cancer cell with an effective amount of a combination of a compound or salt of Formula (I), (I- A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a
CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, wherein the CTLA-4 inhibitor synergistically increases the sensitivity of the cancer cell to the compound or salt of Formula (I), (I-A), or (I-B). In some cases, the contacting is in vitro. In one embodiment, the contacting is in vivo.
[00430] In an aspect the present disclosure provides methods of inhibiting KRas G12 mutants or wildtype in a subject comprising administering to the subject a therapeutically effective amount of a combination of a CTLA-4 inhibitor, or a pharmaceutically acceptable salt or a pharmaceutical composition thereof, and a compound or salt of Formula (I), (I-A), or (I-B), or a pharmaceutically acceptable salt or a pharmaceutical composition thereof. In some cases, the method may inhibit KRas G12 mutants or wildtype activity in a cell. In some cases, inhibiting KRas G12 mutants or wildtype activity in a cell may include contacting the cell in which inhibition of KRas G12 mutants activity is desired with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutical compositions containing the compound or pharmaceutically acceptable salt of any one thereof and a CTLA-4 inhibitor. In some cases, the contacting is in vitro. In some cases, the contacting is in vivo. In some cases, a cell in which inhibition of KRas G12 mutants or wildtype activity is desired is contacted with an effective amount of a compound of Formula (I), (I-A), or (I-B), or pharmaceutically acceptable salt of any one thereof and a CTLA-4 inhibitor to negatively modulate the activity of KRas G12 mutants or wildtype. In some cases, by negatively modulating the activity of KRas G12 mutants or wildtype, the methods described herein are designed to inhibit undesired cellular proliferation resulting from enhanced KRas G12 mutants or wildtype activity within the cell. The cells may be contacted in a single dose or multiple doses in accordance with a particular treatment regimen to effect the desired negative modulation of KRas G12 mutants or wildtype. The ability of compounds to bind KRas G12 mutants or wildtype may be monitored in vitro using well known methods.
[00431] In some embodiments, the inhibitory activity of exemplary compounds and a CTLA-4 inhibitor in cells may be monitored, for example, by measuring the inhibition of KRas G12 mutants or wildtype activity of the amount of phosphorylated ERK.
[00432] In another aspect, methods of treating cancer in a patient in need thereof, comprising administering to said patient a therapeutically effective amount of a compound or salt of Formula (I), (I-A), or (I-B) or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt thereof are provided and a CTLA- 4 inhibitor.
[00433] The compositions and methods provided herein may be used for the treatment of a KRas G12 mutants or wildtype-associated cancer in a patient in need thereof, comprising
administering to the patient a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and a CTLA-4 inhibitor are provided. In some cases, KRas G12 mutants or wildtype associated cancer is lung cancer.
[00434] The compositions and methods provided herein may be used for the treatment of a KRas G12D-associated cancer in a patient in need thereof, comprising administering to the patient a therapeutically effective amount of a compound of Formula (I), (I- A), or (I-B), a pharmaceutically acceptable salt any one thereof, or a pharmaceutical composition comprising the compound or pharmaceutically acceptable salt of any one thereof and a CTLA-4 inhibitor are provided.
[00435] In some embodiments, the method of treating a cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a CTLA-4 inhibitor and a compound or salt of Formula (I), (LA), or (LB), synergistically increases the potency of the compound or salt of Formula (I), (LA), or (LB). In some cases, the synergistic increase in potency of the compound or salt of Formula (I), (LA), or (LB) results in an improved efficacy of the compound or salt of Formula (I), (LA), or (LB).
[00436] As used herein, the term "contacting" refers to the bringing together of indicated moieties in an in vitro system or an in vivo system. For example, "contacting" a KRas G12 mutants or wildtype with a compound provided herein and an immunomodulator inhibitor includes the administration of the compound provided herein and an immunomodulator inhibitor to an individual or patient, such as a human, having KRas G12D and/or other G12 mutants, as well as, for example, introducing a compound provided herein and an immunomodulator inhibitor into a sample containing a cellular or purified preparation containing the KRas G12D and/or other G12 mutants.
[00437] The compositions and methods provided herein may be used for the treatment of a wide variety of cancers including tumors such as lung, prostate, breast, brain, skin, cervical carcinomas, testicular carcinomas, etc. More particularly, cancers that may be treated by the compositions and methods of the invention include, but are not limited to tumor types such as astrocytic, breast, cervical, colorectal, endometrial, esophageal, gastric, head and neck, hepatocellular, laryngeal, lung, oral, ovarian, prostate and thyroid carcinomas and sarcomas. More specifically, these compounds can be used to treat: Cardiac: sarcoma (angiosarcoma, fibrosarcoma, rhabdomyosarcoma, liposarcoma), myxoma, rhabdomyoma, fibroma, lipoma and teratoma; Lung: bronchogenic carcinoma (squamous cell, undifferentiated small cell, undifferentiated large cell, adenocarcinoma), alveolar (bronchiolar) carcinoma, bronchial
adenoma, sarcoma, lymphoma, chondromatous hamartoma, mesothelioma; Gastrointestinal: esophagus (squamous cell carcinoma, adenocarcinoma, leiomyosarcoma, lymphoma), stomach (carcinoma, lymphoma, leiomyosarcoma), pancreas (ductal adenocarcinoma, insulinoma, glucagonoma, gastrinoma, carcinoid tumors, vipoma), small bowel (adenocarcinoma, lymphoma, carcinoid tumors, Kaposi's sarcoma, leiomyoma, hemangioma, lipoma, neurofibroma, fibroma), large bowel (adenocarcinoma, tubular adenoma, villous adenoma, hamartoma, leiomyoma); Genitourinary tract: kidney (adenocarcinoma, Wilm's tumor (nephroblastoma), lymphoma, leukemia), bladder and urethra (squamous cell carcinoma, transitional cell carcinoma, adenocarcinoma), prostate (adenocarcinoma, sarcoma), testis (seminoma, teratoma, embryonal carcinoma, teratocarcinoma, choriocarcinoma, sarcoma, interstitial cell carcinoma, fibroma, fibroadenoma, adenomatoid tumors, lipoma); Liver: hepatoma (hepatocellular carcinoma), cholangiocarcinoma, hepatoblastoma, angiosarcoma, hepatocellular adenoma, hemangioma; Biliary tract: gall bladder carcinoma, ampullary carcinoma, cholangiocarcinoma; Bone: osteogenic sarcoma (osteosarcoma), fibrosarcoma, malignant fibrous histiocytoma, chondrosarcoma, Ewing's sarcoma, malignant lymphoma (reticulum cell sarcoma), multiple myeloma, malignant giant cell tumor chordoma, osteochronfroma (osteocartilaginous exostoses), benign chondroma, chondroblastoma, chondromyxofibroma, osteoid osteoma and giant cell tumors; Nervous system: skull (osteoma, hemangioma, granuloma, xanthoma, osteitis deformans), meninges (meningioma, meningiosarcoma, gliomatosis), brain (astrocytoma, medulloblastoma, glioma, ependymoma, germinoma (pinealoma), glioblastoma multiform, oligodendroglioma, schwannoma, retinoblastoma, congenital tumors), spinal cord neurofibroma, meningioma, glioma, sarcoma); Gynecological: uterus (endometrial carcinoma), cervix (cervical carcinoma, pre-tumor cervical dysplasia), ovaries (ovarian carcinoma (serous cystadenocarcinoma, mucinous cystadenocarcinoma, unclassified carcinoma), granulosa-thecal cell tumors, Sertoli-Leydig cell tumors, dysgerminoma, malignant teratoma), vulva (squamous cell carcinoma, intraepithelial carcinoma, adenocarcinoma, fibrosarcoma, melanoma), vagina (clear cell carcinoma, squamous cell carcinoma, botryoid sarcoma (embryonal rhabdomyosarcoma), fallopian tubes (carcinoma); Hematologic: blood (myeloid leukemia (acute and chronic), acute lymphoblastic leukemia, chronic lymphocytic leukemia, myeloproliferative diseases, multiple myeloma, myelodysplastic syndrome), Hodgkin's disease, non-Hodgkin's lymphoma (malignant lymphoma); Skin: malignant melanoma, basal cell carcinoma, squamous cell carcinoma, Kaposi's sarcoma, moles dysplastic nevi, lipoma, angioma, dermatofibroma, keloids, psoriasis; and Adrenal glands: neuroblastoma. In some cases, the cancer is non-small cell lung cancer, small cell lung cancer, colorectal cancer, rectal cancer or pancreatic cancer. In
some cases, the cancer is non-small cell lung cancer. In some cases, the cancer is a solid tumor cancer.
[00438] In some cases, the concentration and route of administration to the patient will vary depending on the cancer to be treated. The compounds, pharmaceutically acceptable salts thereof and pharmaceutical compositions comprising such compounds and salts also may be coadministered with other anti-neoplastic compounds, e.g., chemotherapy, or used in combination with other treatments, such as radiation or surgical intervention, either as an adjuvant prior to surgery or post-operatively.
[00439] The compounds described herein can be used in the preparation of medicaments for the prevention or treatment of diseases or conditions. In addition, a method for treating any of the diseases or conditions described herein in a subject in need of such treatment, involves administration of pharmaceutical compositions containing at least one compound described herein, or a pharmaceutically acceptable salt, pharmaceutically acceptable prodrug, or pharmaceutically acceptable solvate thereof, in therapeutically effective amounts to said subject. [00440] The compositions containing the compound(s) described herein can be administered for prophylactic and/or therapeutic treatments. In therapeutic applications, the compositions are administered to a patient already suffering from a disease or condition, in an amount sufficient to cure or at least partially arrest the symptoms of the disease or condition. Amounts effective for this use will depend on the severity and course of the disease or condition, previous therapy, the patient's health status, weight, and response to the drugs, and the judgment of the treating physician.
[00441] In prophylactic applications, compositions containing the compounds described herein are administered to a patient susceptible to or otherwise at risk of a particular disease, disorder or condition. Such an amount is defined to be a "prophylactically effective amount or dose." In this use, the precise amounts also depend on the patient's state of health, weight, and the like. When used in a patient, effective amounts for this use will depend on the severity and course of the disease, disorder or condition, previous therapy, the patient's health status and response to the drugs, and the judgment of the treating physician.
[00442] In the case wherein the patient’s condition does not improve, upon the doctor’s discretion the administration of the compounds may be administered chronically, that is, for an extended period of time, including throughout the duration of the patient’s life in order to ameliorate or otherwise control or limit the symptoms of the patient’s disease or condition. [00443] Once improvement of the patient's conditions has occurred, a maintenance dose is administered if necessary. Subsequently, the dosage or the frequency of administration, or both, can be reduced, as a function of the symptoms, to a level at which the improved disease, disorder
or condition is retained. Patients can, however, require intermittent treatment on a long-term basis upon any recurrence of symptoms.
[00444] The amount of a given agent that will correspond to such an amount will vary depending upon factors such as the particular compound, disease or condition and its severity, the identity (e.g., weight) of the subject or host in need of treatment, but can nevertheless be determined in a manner recognized in the field according to the particular circumstances surrounding the case, including, e.g., the specific agent being administered, the route of administration, the condition being treated, and the subject or host being treated. In general, however, doses employed for adult human treatment will typically be in the range of about 0.02 - about 5000 mg per day, in some embodiments, about 1 - about 1500 mg per day. The desired dose may conveniently be presented in a single dose or as divided doses administered simultaneously (or over a short period of time) or at appropriate intervals, for example as two, three, four or more sub -doses per day.
[00445] The pharmaceutical composition described herein may be in unit dosage forms suitable for single administration of precise dosages. In unit dosage form, the formulation is divided into unit doses containing appropriate quantities of one or more compound. The unit dosage may be in the form of a package containing discrete quantities of the formulation. Nonlimiting examples are packaged tablets or capsules, and powders in vials or ampoules. Aqueous suspension compositions can be packaged in single-dose non-reclosable containers. Alternatively, multiple-dose reclosable containers can be used, in which case it is typical to include a preservative in the composition. By way of example only, formulations for parenteral injection may be presented in unit dosage form, which include, but are not limited to ampoules, or in multi-dose containers, with an added preservative.
[00446] Toxicity and therapeutic efficacy of such therapeutic regimens can be determined by standard pharmaceutical procedures in cell cultures or experimental animals, including, but not limited to, the determination of the LD50 (the dose lethal to 50% of the population) and the ED50 (the dose therapeutically effective in 50% of the population). The dose ratio between the toxic and therapeutic effects is the therapeutic index and it can be expressed as the ratio between LD50 and ED50. Compounds exhibiting high therapeutic indices are preferred. The data obtained from cell culture assays and animal studies can be used in formulating a range of dosage for use in human. The dosage of such compounds lies preferably within a range of circulating concentrations that include the ED50 with minimal toxicity. The dosage may vary within this range depending upon the dosage form employed and the route of administration utilized.
[00447] In certain embodiments, the invention provides a method of treating or preventing a disease, state or condition in a patient in need thereof comprising administering to the patient an
effective amount of a compound of any one of embodiments of the invention or a pharmaceutically acceptable salt thereof. The disease, state or condition may be selected from a group as described elsewhere herein.
Bifunctional Compounds
[00448] In some embodiments, compounds herein can adopt to selectively eliminate an over activated KRas signaling which is induced by KRas mutations by directly binding with the mutated KRas protein, either by stabilizing its GDP bound form (the inactive form) or by blocking the interaction between GTP bound form and its downstream target protein. In some embodiments, another way is to hijack the protein degradation mechanism in a cell and leverage E3 ligases’ (like VHL, CRBN or IAPS) substrate specificity through a bi-functional molecule called Proteolysis targeting chimera (PROTAC) (Winter GE, Buckley DL, Paulk J, Roberts JM, Souza A, Dhe- Paganon S, Bradner JE. DRUG DEVELOPMENT. Phthalimide conjugation as a strategy for in vivo target protein degradation. Science. 2015 Jun 19; 348 (6241): 1376-81), which can bind with both mutated KRas protein and E3 ligase, create interactions between those two proteins and induce KRas degradation.
[00449] Disclosed herein is a bifunctional compound composed of a target protein (i.e., KRAS G12D)-binding moiety and an E3 ubiquitin ligase-binding moiety, which may induce proteasome- mediated degradation of selected proteins. In some embodiments, the bifunctional compound comprises a target protein (i.e., KRAS G12D)-binding moiety and an E3 ubiquitin ligase-binding moiety known in the art. In some embodiments, disclosed herein is the use of the compound disclosed herein in the preparation of degrading a target protein compound by using chemical modification of the compound disclosed herein. In some cases, the target protein-binding moiety is derived from a compound of Formula (I), (I-A), or (I-B).
Preparation of Compounds
[00450] The compounds of the present disclosure can generally be prepared in a number of ways well known to those skilled in the art of organic synthesis. By way of example, compounds of the present disclosure can be synthesized using the methods described herein, together with synthetic methods known in the art of synthetic organic chemistry, or variations thereof as appreciated by those skilled in the art. The compounds of the present disclosure may be prepared as described in the schemes and examples described elsewhere herein.
[00451] The following examples further illustrate the invention but, of course, should not be construed as in any way limiting its scope
EXAMPLES
[00452] The following synthetic schemes are provided for purposes of illustration, not limitation. The following examples illustrate the various methods of making compounds described herein. It is understood that one skilled in the art may be able to make these compounds by similar methods or by combining other methods known to one skilled in the art. It is also understood that one skilled in the art would be able to make, in a similar manner as described below by using the appropriate starting materials and modifying the synthetic route as needed. In general, starting materials and reagents can be obtained from commercial vendors or synthesized according to sources known to those skilled in the art or prepared as described herein.
[00453] The present disclosure provides processes for preparing the compounds described herein (described in greater detail below).
Synthesis of Intermediates
Intermediate 1. Synthesis of 3-chloro-N ,N -dimethyl-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a] [ 1 ,4]diazepine-2-carboxamide.
[00454] Step 1. Synthesis of 2-nitro-prop-2-ynyl-benzenesulfonamide (Int-la). A solution of prop-2-yl-l -amine (1.87 mL, 29.1 mmol) and 7V,7V-diisopropylethylamine (10.15 mL, 58.3 mmol) in DCM (100 mL) was cooled to 0 °C. 2-nitrobenzenesulfonyl chloride (6.46 g, 29.15 mmol) was added portion wise. The solution was then allowed to warm to rt and was further stirred for 1 hr. The mixture was washed with water and brine, dried over ISfeSCh, concentrated.
The crude product was purified by silica gel chromatography (eluted with EtOAc in petroleum ether from 20% to 50%) to afford 2-nitro-prop-2-ynyl-benzenesulfonamide (Int-la, 5.98 g, 24.9
mmol, 85.4% yield) as a yellow solid. LCMS calcld for C9H9N2O4S (M+H)+ m/z = 241.0, found: 241.0.
[00455] Step 2. Synthesis of (3-chloropropyl)-2-nitro-prop-2-ynyl-benzenesulfonamide (Int- 1b). A solution of 2-nitro-prop-2-ynyl-benzenesulfonamide (Int-la, 1.0 g, 4.16 mmol) and cesium carbonate (6.764 g, 20.81 mmol) in acetone (30 mL) was added neat l-bromo-3 -chloropropane (6.05 mL, 61.19 mmol) dropwise. Upon complete addition, the reaction mixture was stirred at rt for 2 hrs. The mixture was concentrated, then washed with water and brine, dried over Na2SO4, concentrated. The crude product was purified by silica gel chromatography (eluted with EtOAc in petroleum ether from 20% to 50%). (3-chloropropyl)-2-nitro-prop-2-ynyl- benzenesulfonamide (Int-lb, 2.43 g, 7.67 mmol, 92.1% yield) was obtained as a yellow oil. LCMS calcld for C12H14CIN2O4S (M+H)+ m/z = 317.0, found: 317.0.
[00456] Step 3. Synthesis of ethyl 5-(2-nitrophenyl)sulfonyl -4,6,7, 8-tetrahydropyrazolo[ 1,5- a][l,4]diazepine-2-carboxylate (Int-lc). To solution of (3-chloropropyl)-2-nitro-prop-2-ynyl- benzenesulfonamide (Int-lb, 10.0 g, 31.6 mmol) and ethyl 2-diazoacetate (5.4 g, 47.3 mmol) in chlorobenzene (80 mL) was added A,A-diisopropylethylamine (5.5 mL, 31.57 mmol). Then heated at 140 °C for 1.5 hours. Followed by the addition of cesium carbonate (12.3 g, 37.9 mmol), then heated at 140 °C for 30 minutes. The solvent was concentrated, and the mixture was extracted with EtOAc, washed with brine, dried over Na2SO4, concentrated. The crude product was purified by silica gel chromatography (eluted with EtOAc in petroleum ether from 20% to 90%), then triturated in EtOAc to afford ethyl 5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int-lc, 5.80 g, 14.7 mmol, 46.6% yield) as a yellow solid. LCMS calcld for C16H19N4O6S (M+H)+ m/z = 395.1, found: 395.0.
[00457] Step 4. Synthesis of ethyl 5-(2-nitrophenyl)sulfonyl -4,6,7, 8-tetrahydropyrazolo[ 1,5- a][l,4]diazepine-2-carboxylate (Int-ld). To a solution of ethyl 5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int-lc, 12.6 g, 31.95 mmol) in THF (100 mL) and methanol (25 mL) was added IM LiOH (128 mL, 128 mmol) at 25 °C. The mixture was stirred at 55 °C for 2 h. The mixture was acidified with HC1 (Imol/L in H2O) to pH= 6 and the crude product was triturated in water and filtered. 5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylic acid (Int-ld, 11.5 g, 31.1 mmol, 97.3% yield) was obtained as yellow solid. LCMS calcld for C14H14N4O6S (M+H)+ m/z = 367.1, found: 367.0.
[00458] Step 5. Synthesis of A,A-dimethyl-5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (Int-le). To a solution of 5-(2- nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylic acid (Int-ld, 9.6 mg, 26.2 mmol), DIEA (18.3 mL, 104.8 mmol) and HATU (14.95 g, 39.3 mmol) in DMF (90
mL) was added 2M methylmethanamine in THF (20 mL, 39.3 mmol) at 30 °C. The mixture was stirred at 30 °C for 2h. The mixture was diluted with DCM (300 x 2 mL), washed with water (400 mL) and brine (400 x 2 mL), dried over Na2SO4 and concentrated to afford a crude product N ,N -dimethyl-5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2- carboxamide (Int-le, 10.1 g, 24.4 mmol, 93.1% yield) as yellow oil. LCMS calcld for C16H20N4O3S (M+H)+ m/z =394.1 , found: 394.2.
[00459] Step 6. Synthesis of 3-chloro- N ,N -dimethyl-5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (Int-lf). To a solution of A/A-di methyl - 5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (Int- le, 10 g, 25.67 mmol) in DMF (100 mL) was added Chlorosuccinimide (3.428 g, 25.67mmol) at 0 °C under argon. The mixture was stirred at 45 °C for Ih. The mixture was concentrated to afford a crude product. The crude product was triturated in water and filtered to afford 3-chloro- N ,N -dimethyl-5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2- carb oxami de (Int-lf, 10.2 g, 23.4 mmol, 91.0% yield) was obtained as crude yellow solid.
LCMS calcld for CieHnCMCLS (M+H)+ m/z = 428.1, found: 428.0.
[00460] Step 7. Synthesis of 3-chloro-N ,N -dimethyl-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a][l,4]diazepine-2-carboxamide (Intermediate 1). To a solution of 3-chloro-N ,N -dimethyl-5-(2- nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (Int-lf, 10.2 g, 23.84 mmol), 4-methoxybenzenethiol (8.8 mL, 71.52 mmol) and CS2CO3 (31067.73mg, 95.36 mmol) in acetonitrile (100 mL) . The mixture was stirred at 25 °C for 2 h. The mixture was concentrated, and the crude product was purified by silica gel chromatography (eluted with MeOH in DCM from 3% to 10%) to afford 3-chloro-N ,N -dimethyl-5,6,7,8-tetrahydro-4H- pyrazolo[l,5-a][l,4]diazepine-2-carboxamide (Intermediate 1, 3.2 g, 13.2 mmol, 55.3% yield) as yellow solid. LCMS calcld for C10H15CIN4O (M+H)+ m/z = 243.1, found: 243.0
Alternative synthesis of Intermediate 1. Synthesis of 3-chloro-N ,N -dimethyl-5,6,7,8-tetrahydro- 4H-pyrazolo[ 1 ,5-a] [ 1 ,4]diazepine-2-carboxamide.
Int-1m lnt-1n Intermediate 1
[00461] Step 1. Synthesis of diethyl 4-chloro-lH-pyrazole-3,5-dicarboxylate (Int-lg). To a solution of diethyl lH-pyrazole-3,5-dicarboxylate (20 g, 94.25 mmol) in Acetic acid (360mL) was added dropwise NaClO (264 mL, 1980 mmol). After stirring for 3 h at 25 °C, the remaining mixture was diluted with water, and the mixture was extracted with ethyl acetate. The combined organic layers were concentrated to afford yellowish solid, the solid was dissolved of Petroleum Ether (160 mL) and EtOAc (20 mL), then filtered and collected solids to afford diethyl 4-chloro- lH-pyrazole-3,5-dicarboxylate (Int-lg, 15.20 g, 61.6 mmol, 65.38% yield) as a white solid and mother liquid 7g. LCMS calculated for C9H12CIN2O4 (M+H)+ m/z = 247.6; found: 247. ' H NMR (400 MHz, CD3OD) 5 4.44 - 4.36 (m, 4H), 1.39 (t, J= 7.1 Hz, 6H).
[00462] Step 2. Synthesis of diethyl l-[3-(tert-butoxycarbonylamino)propyl]-4-chloro- pyrazole-3,5-dicarboxylate (Int-lh). To a solution of diethyl l-[3-(tert- butoxycarbonylamino)propyl]-4-chloro-pyrazole-3,5-dicarboxylate (Int-lg, 24.7 g,24.46 mmol, 39.70% yield) in DMF (150mL) was added tert-butyl N-(3-bromopropyl)carbamate (16141.77 mg, 67.79 mmol), Potassium Iodide (204.6 mg, 1.2 3mmol), CS2CO3 (40157.83 mg, 123.25 mmol), then the mixture was stirred at rt for 16h. The reaction was quenched with water, extracted it with EtOAc three times, combined the organic phase, washed it with water for 3 times, dry and filter it, collected the filtrate, and concentrated afford diethyl l-[3-(tert- butoxycarbonylamino)propyl]-4-chloro-pyrazole-3,5-dicarboxylate (Int-lh, 24.70 g, 24.5 mmol, 39.70% yield as reddish brown oil oil. LCMS calculated for C17H27CIN3O6 (M+H)+ m/z =404.8; found: 304.1(M+H-Boc).
[00463] Step 3. Synthesis of diethyl l-(3-aminopropyl)-4-chloro-pyrazole-3,5- dicarboxylate;hydrochloride (Int-li). To a solution of diethyl l-(3-aminopropyl)-4-chloro- pyrazole-3,5-dicarboxylate;hydrochloride (Int-lh, 25.5 g, 23.99 mmol, 98.05% yield) in 1,4- Di oxane (50mL) was added HCl/Dioxane (150 mL, 600 mmol), then the mixture was stirred at 25 °C for Ih. The reaction was concentrated to afford diethyl l-(3-aminopropyl)-4-chloro-
pyrazole-3,5-dicarboxylate;hydrochloride (Int-li, 25.50 g, 24.0 mmol, 98.05% yield) as yellow oil for next step. LCMS calculated for C12H19CIN3O4 (M+H)+ m/z = 304.1; found: 304.3.
[00464] Step 4. Synthesis of ethyl 3-chloro-4-oxo-5,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxylate (Int-lj). To a solution of diethyl l-(3-aminopropyl)-4-chloro- pyrazole-3,5-dicarboxylate;hydrochloride (Int-li, 25.5 g, 23.99 mmol) in Methanol (500mL) was added EtsN (16.74 mL, 119.93 mmol), the mixture was stirred overnight at 80 °C. The reaction was concentrated to a volume of 20 mL, the resulting white solid was filtered, then diluted with MeOH, stirred for 10 min, filtered. Then the solid was washed with water for three times, and extracted by DCM for three times to give ethyl 3-chloro-4-oxo-5, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int-lj, 4.80 g, 18.6 mmol, 77.66 %yield). LCMS calculated for C10H13CIN3O3 (M+H)+ m/z = 258.67; found: 258.1.
[00465] Step 5. Synthesis of ethyl 3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a][l,4]diazepine-2-carboxylate (Int-lk). To a solution of ethyl 3-chloro-4-oxo-5, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int-lj, 4.8 g, 18.63 mmol) in THF (60mL) was added Borane-Methyl Sulfide Complex (27.94 mL, 55.89 mmol) under N2. Thereaction was stirred at rt for 2h and then was stirred at 45 °C for 32h. MeOH to added to quench the reaction at 0°C, then the mixture was concentrated to afford ethyl 3-chloro-5, 6,7,8- tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int-lk, 4.50 g, 18.5 mmol). LCMS calculated for C10H15CIN3O2 (M+H)+ m/z = 244.7; found: 244.3.
[00466] Step 6. Synthesis of 05-tert-butyl 02-ethyl 3-chloro-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2,5-di carboxylate (Int-11). To a solution of ethyl 3-chloro-5,6,7,8-tetrahydro- 4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int-lk, 4.5 g, 18.47 mmol) in DCM (60 mL) was added EtsN (7.73 mL, 55.4 mmol) and DMAP (225.6 mg, 1.85 mmol), then the mixture was stirred at rt for 10 min, BOC2O (8.87 g, 40.63 mmol) was added, the mixture was stirred at 25 °C for 4h. The mixture was concentrated and purified by flash column chromatography (silica gel, eluting with 0% to 6% EtOAc/DCM) to afford 05-tert-butyl 02-ethyl 3-chloro-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2,5-dicarboxylate (Int-11, 4.10 g, 9.54 mmol, 51.66 %yield). LCMS calculated for C15H23CIN3O4 (M+H)+ m/z = 344.8; found: 344.1.
[00467] Step 7. Synthesis of 5-tert-butoxycarbonyl-3-chloro-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxylic acid (Int-lm). To a solution of 05-tert-butyl 02-ethyl 3-chloro- 4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2,5-dicarboxylate (Int-11, 4.1 g, 9.54 mmol) in THF (36mL) and Methanol (9mL) a was added hydroxylithium (913.95 mg, 38.16 mmol) in Water (38.16mL) at 25 °C. The mixture was stirred at 55 °C for 2 h. The mixture was concentrated and acidified with HC1 (Imol/L in H2O) to pH= 1 and the crude product was triturated in water and filtered. 5-tert-butoxycarbonyl-3-chloro-4,6,7,8-tetrahydropyrazolo[l,5-
a][l,4]diazepine-2-carboxylic acid (Int-lm, 2.4 g,7.60 mmol, 79.67% yield) was obtained as white solid. LCMS calculated for C13H19CIN3O4 (M+H)+ m/z = 316.7, found: 316.1.
[00468] Step 8. Synthesis of tert-butyl 3-chloro-2-(dimethylcarbamoyl)-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-ln). The mixture of 5-tert- butoxycarbonyl-3-chloro-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylic acid (Int- lm, 100 mg, 0.32 mmol) in DCM (ImL) was added Oxalyl Chloride (0.32 mL, 0.63 mmol) at 0 °C, the mixture was stirred at 0 °C for 0.5h and rt for 0.5h. Then the mixture was concentrated, diluted it with DCM (ImL), and drop it into Dimethylamine (0.63 mL, 1.27 mmol) (2M in THF), then the mixture was stirred at rt for Ih. The mixture was quenched with NH4CI aqueous, extracted with EtOAc. washed with water and brine, dried over Na2SO4 and concentrated to afford a crude product tert-butyl 3-chloro-2-(dimethylcarbamoyl)-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-ln, 108 mg, 0.315 mmol) as yellow oil. LCMS calcld for C15H24CIN4O3 (M+H)+ m/z =343.8 , found: 343.2. 'H NMR (400 MHz, DMSO) 5 4.45 - 4.58 (m, 2H), 4.33 - 4.43 (m, 2H), 3.69 - 3.78 (m, 2H), 3.09 (s, 6H), 1.92 - 2.01 (m, 2H), 1.44 (s, 9H).
[00469] Step 9. Synthesis if 3-chloro-N,N-dimethyl-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a][l,4]diazepine-2-carboxamide (Intermediate 1). To a solution of tert-butyl 3-chloro-2- (dimethylcarbamoyl)-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (108 mg, 0.32 mmol), tert-butyl 3-chloro-2-(dimethylcarbamoyl)-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-5-carboxylate (199 mg, 0.58 mmol) and tert-butyl 3-chloro-2- (dimethylcarbamoyl)-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (9.96 g, 27.6 mmol) in 1,4-Dioxane (50mL) was added HCl/Dioxane (150 mL, 600 mmol). The mixture was stirred at 20 °C for 2 h. The mixture was concentrated and the crude product was purified by Prep-HPLC on a C18 column (5 uM, 50 x 150 mm) with mobile phase: H2O (0.1% NH4HCO3)/ MeCN at flow rate : 35 mL/min to afford 3-chloro-N,N-dimethyl-5,6,7,8-tetrahydro-4H- pyrazolo[l,5-a][l,4]diazepine-2-carboxamide (6.18 g, 25.46 mmol, 89.36% yield) as white solid. LCMS calcld for CIOHI5C1N40 (M+H)+ m/z = 243.1, found: 243.1. 'H NMR (400 MHz, DMSO) 54.24 - 4.38 (m, 2H), 3.26 - 3.37 (m, 2H), 3.00 - 3.05 (m, 2H), 2.98 (d, J= 15.2 Hz, 6H), 1.66 - L77 (m, 2H).
Intermediate 2
[00470] Step 1. Synthesis of tert-butyl 2-amino-3-cyano-spiro[5,6- dihy drocy cl openta[b]thiophene-4, 3 '-azetidine]- l'-carboxylate (Int-2a). A solution of propanedinitrile (219.93 mg, 3.33 mmol) tert-butyl 5-oxo-2-azaspiro[3.4]octane-2-carboxylate (500 mg, 2.22 mmol) in DMF (5 mL) was added L-proline (255 mg, 2.22 mmol) and sulfur (113.5 mg, 3.33 mmol) at 25 °C under N2. Then the mixture was stirred at 60 °C for 4 h. The mixture was filtered to afford a crude solution. The reaction mixture was purified by prep-HPLC (eluted with CH3CN in H2O from 5.0% to 95%) to afford tert-butyl 2-amino-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3 '-azetidine]- l'-carboxylate (Int-2a, 380 mg, 1.244 mmol, 56.1% yield) as yellow solid. LCMS calcld for C15H19N3O2S (M+H)+ m/z = 306.1, found: 306.1. ‘H NMR (400 MHz, CD3OD) 5 4.19 (s, 2H), 3.95 (d, J= 8.1 Hz, 2H), 2.76 - 2.69 (m, 2H), 2.69 - 2.61 (m, 2H), 1.45 (s, 9H).
[00471] Step 2. Synthesis of 2-aminospiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]- 3 -carbonitrile (Intermediate 2). A solution of tert-butyl 2-amino-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3 '-azetidine]- l'-carboxylate (Int-2a, 380 mg, 1.24 mmol) in DCM (5 mL) was added TFA (1.91 mL, 24.89 mmol) at 25 °C under N2. Then the mixture was stirred at 20 °C for 1 h. The mixture was concentrated and the crude product 2-aminospiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile (Intermediate 2, 380 mg, 0.678 mmol, 54.5% yield) was used directly for the next step. LCMS calcld for C10H11N3S (M+H)+ m/z = 206.2, found: 206.1.
Intermediate 3
[00472] Intermediate 3 was prepared similarly to that of Intermediate 2 from 5-oxo-2- azaspiro[3.5]nonane (ref. Angew. Chem. Int. Ed. 2021, 60, 7360-7365). LCMS calcld for C11H13N3S (M+H)+ m/z =220.1, found: 220.1.
[00473] Intermediate 4. 2-aminospiro[5,6,7,8-tetrahydrocyclohepta[b]thiophene-4,3'- azetidine]-3-carbonitrile
Intermediate 4
[00474] Intermediate 4 was prepared similarly to that of Intermediate 2. LCMS calcld for C12H15N3S (M+H)+ m/z = 234.1, found:234.1.
Intermediate 5. Synthesis of l-(3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin-2- yl)ethanone
lnt-5d Intermediate 5
[00475] Step 1. Preparation of tert-butyl 2-[methoxy(methyl)carbamoyl]-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-5b). To a solution of 5-tert- butoxy carbonyl-4, 6, 7, 8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylic acid (Int 5a prepared similarly to Int-lm, 500 mg, 1.78 mmol) , DIEA (1.24 mL, 7.11 mmol) and HATU (1013.76 mg, 2.67 mmol) in DMF (10 mL) was added N,O-Dimethylhydroxylamine Hydrochloride (260.05 mg, 2.67 mmol) at 25 °C. The mixture was stirred at 25 °C for Ih. The mixture was diluted with EtOAC (50x2 mL), washed with water (50 mL) and brine (50 ml), dried over Na2SO4, concentrated. The crude product was purified by silica gel chromatography (eluted with EtOAc in petroleum ether from 10% to 90%). tert-butyl 2- [methoxy(methyl)carbamoyl]-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-5b, 300 mg, 0.925 mmol, 52.03% yield) was obtained as white solid. LCMS calcld for CI5H24N4O4 (M+H)+ m/z = 325.2, found: 325.1. 'HNMR (400 MHz, CDC13) 8 6.63 (s, IH), 4.47 (dd, J= 14.7, 9.4 Hz, 4H), 3.74 (s, 5H), 3.42 (s, 3H), 1.94 (s, 2H), 1.41 (s, 9H).
[00476] Step 2. Preparation of tert-butyl 3-chloro-2-[methoxy(methyl)carbamoyl]-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-5c). To a solution of tert-butyl 2- [methoxy(methyl)carbamoyl]-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-5b, 400 mg, 1.23 mmol) in Acetic acid (1 mL) was added NCS (246.99 mg, 1.85 mmol) at 60 °C. The mixture was stirred at 60 °C for 3h. The mixture was concentrated to afford a crude product and used without further purification, tert-butyl 3-chloro-2- [methoxy(methyl)carbamoyl]-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-5c, 300 mg, 67.80% yield). LCMS calcld for CI5H24C1N4O4 (M+H)+ m/z = 359.82, found: 359.2.
[00477] Step 3. Preparation of tert-butyl 2-acetyl-3-chloro-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-5-carboxylate (Int-5d). To a solution of tert-butyl 3-chloro-2- [methoxy(methyl)carbamoyl]-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate
(Int-5c, 240 mg, 0.67 mmol) in THF (1 mL) was added chloro(methyl)magnesium (0.67 mL, 2.01 mmol) at -20 °C. The resulting mixture was allowed to rt and stirred for 16 h. The reaction mixture was added sat. NH4CI aq. (30 mL) and extracted with EtOAc (2 x 30 mL) and the combined organic layers were dried over Na2SO4 and concentrated in vacuum to give the residue. The crude product was purified by flash chromatography (eluted with EtOAc in petroleum ether from 10% to 90%). tert-butyl 2-acetyl-3-chloro-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-5-carboxylate (Int-5d, 140 mg, 0.446 mmol, 66.71% yield) was obtained as white solid. LCMS calcld for C14H20CIN3O3 (M+H)+ m/z = 314.1, found: 314.2.
[00478] Step 4. Preparation of l-(3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a][l,4]diazepin-2-yl)ethenone (Intermediate 5). tert-butyl 2-acetyl-3-chloro-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-5d, 140 mg, 0.45 mmol) in HC1 in Dioxane (56 mL, 224 mmol). The mixture was stirred at 25°C. The mixture was concentrated to afford a crude product and used without further purification. l-(3-chloro-5,6,7,8-tetrahydro-4H- pyrazolo[l,5-a][l,4]diazepin-2-yl)ethanone (Intermediate 5, 150 mg) was obtained as white solid. LCMS calcld for C9H13CIN3O (M+H)+ m/z = 214.66, found: 214.2.
Intermediate 6. Synthesis of 2-amino-6-methyl-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'- azetidine]-3-carbonitrile.
ate 6
[00479] Step 1. Preparation of tert-butyl 7-methyl-5-oxo-2-azaspiro[3.4]octane-2-carboxylate (Int-6b) To a solution of THF (4 mL) was added 1.6M butyllithium in hexane (4.12 mL, 6.59 mmol) at -68 °C. The mixture was stirred at -68 °C for 10 min. Then, a solution of tert-butyl 3- cyano-3-(3-iodo-2-methyl-propyl)azetidine-l-carboxylate (Int-6a, CN 11257408, 800 mg, 2.2 mmol) in THF (4 mL) was added at -68 °C . The mixture was stirred at -68 °C for 3 h. The mixture was quenched with Acetic acid (5 mL) at -40 °C, extracted with tert-Butyl methyl ether (60 x 2 mL) and water (60 mL). The solution was washed with NaHCCL (60 mL), Na2S20s (20% 60 mL) and brine (60 mL), dried over Na2SO4, concentrated. The crude product was purified by silica gel chromatography (eluted with EtOAc in petroleum ether from 0% to 5%). tert-butyl 7- methyl-5-oxo-2-azaspiro[3.4]octane-2-carboxylate (Int-6b, 110 mg, 0.45 mmol, 20.93% yield) was obtained as yellow oil.
[00480] Step 2. Preparation of tert-butyl 2-amino-3-cyano-6-methyl-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3 '-azetidine]- 1 '-carboxylate (Int-6c). A solution of propanedinitrile (45.55 mg, 0.69 mmol) tert-butyl 7-methyl-5-oxo-2-azaspiro[3.4]octane-2-
carboxylate (Int-6b, 110 mg, 0.46 mmol) in DMF (1 mL) was added L-Proline (52.92 mg, 0.46 mmol) and Sulfur (23.5 mg, 0.69 mmol) at 25 °C under N2. Then the mixture was stirred at 60 °C for 6 h. The mixture was filtered to afford a crude solution. The reaction mixture was purified by flash chromatography (eluted with CH3CN in H2O from 40% to 60%). tert-butyl 2-amino-3- cyano-6-methyl-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-carboxylate (Int-6c, 30 mg, 0.094 mmol, 20.43% yield) was obtained as yellow solid. LCMS calcld for C16H21N3O2S (M+Na)+ m/z = 342.1, found: 342.1.
[00481] Step 3. Preparation of 2-amino-6-methyl-spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3 '-azetidine]-3 -carbonitrile (Intermediate 6). To a solution of tert-butyl 2-amino-3-cyano-6- methyl-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-carboxylate (Int-6c, 30 mg, 0.09 mmol) in DCM (1 mL) was added TFA (0.14 mL, 1.8 mmol) at rt. The reaction mixture was then stirred at 25 °C for 30 min. The mixture was concentrated to afford a crude product. The crude product 2-amino-6-methyl-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3- carbonitrile (Intermediate 6, 30 mg) was used without purification. LCMS calcld for C11H13N3S (M+H)+ m/z =220.1, found: 220.1.
Intermediate 7. Synthesis of 3-methyl-5-(5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin-2- yl)-l,2,4-oxadiazole
lnt-1 c Intermediate 7
[00482] Preparation of 3-methyl-5-(5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin-2-yl)- 1,2,4-oxadiazole. To a solution of N-hydroxyacetamidine (375.66 mg, 5.07 mmol) and 3A molecular sieves in THF (5 mL) were added NaH (76.06 mg, 3.17 mmol), The mixture was stirred at RT for 15 min. ethyl 5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxylate (Int-lc, 500 mg, 1.27 mmol) was added to the mixture. The mixture was stirred at 25 °C for 3 h. The mixture was filtered over celite and concentrated. The crude product was purified by flash chromatography (eluted with CH3CN in H2O from 5.0% to 95%). The product 3-methyl-5-(5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin-2-yl)-l,2,4- oxadiazole (230 mg, 1.05 mmol, 82.75% yield) was obtained as a yellow solid. LCMS calcld for C10H13N5O (M+H) m/z =220.1, found: 220.1.
Intermediate 8. Synthesis of 2-methylsulfanyl-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a][l,4]diazepine
[00483] Step 1. Preparation of 3-[(E)-(3-methylsulfanyl-lH-pyrazol-5- yl)methyleneamino]propan-l-ol (Int-8a). The solution of 3-methylsulfanyl-lH-pyrazole-5- carbaldehyde (Bioorganic and Medicinal Chemistry, 2012, vol. 20, # 3, p. 1319 1336, 1.1 g, 7.74 mmol) and 3 -aminopropan- l-ol (639.21 mg, 8.51 mmol) in Ethanol (10 mL) was stirred at 80 °C for 16 h. The mixture was concentrated to afford a crude product which was used directly in the next step. 3-[(E)-(3-methylsulfanyl-lH-pyrazol-5-yl)methyleneamino]propan-l-ol (Int-8a, 1.7 g, 3.23 mmol, 41.79% yield) was obtained as crude yellow oil. LCMS calcld for C8HI3N3OS (M-H)- m/z = 199.1, found: 199.1.
[00484] Step 2. Preparation of 3-[(3-methylsulfanyl-lH-pyrazol-5-yl)methylamino]propan-l- ol (Int-8b) To a solution of 3-[(E)-(3-methylsulfanyl-lH-pyrazol-5-yl)methyleneamino]propan- l-ol (Int-8a crude, 1.7 g, 8.53 mmol) in Methanol (30mL) was added Sodium triacetoxyborohydride (3.62 g, 17.06 mmol) at 0 °C. The mixture was stirred at 25 °C for 6 h. The mixture was concentrated directly to lOmL. The crude product was purified by Cl 8 flash chromatography (eluted with CEECN in water from 0% to 7%) to afford 3-[(3-methylsulfanyl- lH-pyrazol-5-yl)methylamino]propan-l-ol (Int-8b, 1.5 g, 7.452 mmol, 87.35% yield). LCMS calcld for C8HI5N3OS (M+H)+ m/z = 202.2, found: 202.2.
[00485] Step 3. Preparation of tert-butyl N-(3-hydroxypropyl)-N-[(3-methylsulfanyl-lH- pyrazol-5-yl)methyl]carbamate (Int-8c). To a solution of 3-[(3-methylsulfanyl-lH-pyrazol-5- yl)methylamino]propan-l-ol (Int-8b, 100 mg, 0.5 mmol) and Et3N (0.14 mL, 0.99 mmol) in DCM (3 mL) was added BOC2O (0.14 mL, 0.6 mmol) at 0 °C. The mixture was stirred at 25 °C for 2 h. The mixture was diluted with DCM (10 mL) and filtered. The solution was washed with water (10 mL) and brine (10 mL), dried over ISfeSCU, concentrated. The crude product was purified by Prep-TLC (eluted with EtOAc 100%). tert-butyl N-(3-hydroxypropyl)-N-[(3-
methylsulfanyl-lH-pyrazol-5-yl)methyl]carbamate (Int-8c, 50 mg, 0.149 mmol, 30.05% yield) was obtained as colorless viscous semi-solid. LCMS calcld for C13H24N3O3S (M+H)+ m/z =
302.1, found: 302.1.
[00486] Step 4. Preparation of 3-[tert-butoxycarbonyl-[(3-methylsulfanyl-lH-pyrazol-5- yl)methyl]amino]propyl methanesulfonate (Int-8d). To a solution of tert-butyl N-(3- hydroxypropyl)-N-[(3-methylsulfanyl-lH-pyrazol-5-yl)methyl]carbamate (Int-8c, 50 mg, 0.17 mmol) and in DCM (2 mL) was added MsCl (28.5 mg, 0.25 mmol) at 0 °C. The mixture was stirred at 25 °C for 2 h. The mixture was quenched with H2O (20 mL), extracted with DCM (10x2 mL), dried over Na2SO4, concentrated. The crude product was purified by Prep-TLC (eluted with EtOAc in petroleum ether 50%). 3-[tert-butoxycarbonyl-[(3-methylsulfanyl-lH- pyrazol-5-yl)methyl]amino]propyl methanesulfonate (Int-8d, 27 mg, 0.069 mmol, 41.60% yield) was obtained as colorless viscous semi-solid. LCMS calcld for C14H25N3O5S2 (M+Na)+ m/z =
402.1, found: 402.0
[00487] Step 5. Preparation of tert-butyl 2-methylsulfanyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-5-carboxylate (Int-8e). To a solution of 3-[tert-butoxycarbonyl-[(3- methylsulfanyl-lH-pyrazol-5-yl)methyl]amino]propyl methanesulfonate (Int-8d 200 mg, 0.53 mmol) in THF (20 mL) was added sodium hydride (63.24 mg, 1.58 mmol) at 0 °C under N2. The mixture was stirred at 25 °C for 3 h. The mixture was quenched with the mixture of NH4CI (20 mL) and iced water (20 mL), extracted with EtOAC (20*3 mL), dried over Na2SO4, concentrated.
[00488] The crude product was purified by flash chromatography (eluted with EtOAc in petroleum ether from 0% to 35%). tert-butyl 2-methylsulfanyl -4,6,7, 8-tetrahydropyrazolo[ 1,5- a][l,4]diazepine-5-carboxylate (Int-8e, 50 mg, 0.1764 mmol, 33.479% yield) was obtained as crude yellow oil.
[00489] Step 6. Preparation of 2-methylsulfanyl-5,6,7,8-tetrahydro-4H-pyrazolo[l,5- a][l,4]diazepine (Intermediate 8). To a solution of tert-butyl 2-methylsulfanyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-8e, 120 mg, 0.42 mmol) in DCM (4 mL) was added TFA (0.4 mL, 5.23 mmol) at 0 °C. The mixture was stirred at 25 °C for 1 h. The mixture was concentrated to afford a crude product and was used without purified. 2- methylsulfanyl-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepine (Intermediate 8, 70 mg) was obtained as crude yellow oil.
Intermediate 9. Synthesis of
[00490] Step 1. Preparation of tert-butyl 6-bromo-5-oxo-2-azaspiro[3.4]octane-2-carboxylate (Int-9a). To the solution of tert-butyl 5-oxo-2-azaspiro[3.4]octane-2-carboxylate (200 mg, 0.89 mmol) in Ether (5 mL) was added 5, 5-dibromohexahydropyrimidine-2, 4, 6-trione (507.6 mg, 1.78 mmol), and the mixture was stirred at 35 °C for 48h. The mixture was extracted with Ether and water, dried with brine and ISfeSC , concentrated and purified by silica gel chromatography (PE:EtOAc = 15: 1) to get tert-butyl 6-bromo-5-oxo-2-azaspiro[3.4]octane-2-carboxylate (Int-9a, 140 mg, 0.460 mmol, 51 ,84%yield).
[00491] Step 2. Preparation of tert-butyl 2-aminospiro[5,6-dihydrocyclopenta[d]thiazole-4,3'- azetidine]-l'-carboxylate (Int-9b). To the solution of tert-butyl 6-bromo-5-oxo-2- azaspiro[3.4]octane-2-carboxylate (Int-9a 140 mg, 0.46 mmol) in Ethanol (2 mL) was added Thiourea (35.03 mg, 0.46 mmol), and the mixture was stirred at 80 °C for 2h. The solvent was removed and purified by prep-TLC (DCM: MeOH=15: 1) to get tert-butyl 2-aminospiro[5,6- dihydrocyclopenta[d]thiazole-4,3'-azetidine]-l'-carboxylate (Int-9b, 20 mg, 0.0711 mmol, 15.44 %yield). LCMS calcld for C13H19N3O2S (M+H)+ m/z =282.37, found: 282.2.
[00492] Step 3. Preparation of spiro[5,6-dihydrocyclopenta[d]thiazole-4,3'-azetidine]-2-amine (Intermediate 9). The solution of tert-butyl 2-aminospiro[5,6-dihydrocyclopenta[d]thiazole-4,3'- azetidine]-l'-carboxylate (Int-9b, 15 mg, 0.05 mmol) and Trifluoroacetic acid (0.04 mL, 0.53 mmol) in DCM (2 mL) was stirred at 25 °C for Ih. The solvent was removed to get spiro[5,6- dihydrocyclopenta[d]thiazole-4,3'-azetidine]-2-amine (Intermediate 9, 15 mg). LCMS calcld for C13H19N3O2S (M+H)+ m/z =182.26, found: 182.2.
Intermediate 10. Synthesis of tert-butyl 2-cyano-4,6,7,8-tetrahydropyrazolo[l,5- a] [ 1 ,4]diazepine-5-carboxylate
[00493] Step 1. Synthesis of tert-butyl 2-carbamoyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-5-carboxylate (Int-lOa). To a solution of 5-tert-butoxycarbonyl-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylic acid (Int-5a, 450 mg, 1.6 mmol) , DIEA (0.84 mL, 4.8 mmol) and HATU (0.91 g, 2.4 mmol) in DMF (45mL) was added NH4CI (0.12 g, 1.92 mmol) at 30 °C. The mixture was stirred at 30 °C for 2h. The mixture was diluted with EtOAc (60 mL), washed with water (40 mL) and brine (40 mL), dried over Na2SC>4, concentrated. The mixture was concentrated to afford a crude product. The crude product was purified by flash chromatography (eluted with CH3CN in H2O (0.1 % TFA) from 5.0% to 95%); tert-butyl 2-carbamoyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-lOa, 350 mg, 1.25 mmol, 78.05% yield) was obtained as yellow solid. LCMS calcld for C13H20N4O3 (M+H)+ m/z =281.0 , found: 281.0.
[00494] Step 2. Synthesis of tert-butyl 2-cyano-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-5-carboxylate (Intermediate 10). To a solution of tert-butyl 2-carbamoyl- 4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Int-lOa, 300 mg, 1.07 mmol) and Pyridine (0.26 mL, 3.21 mmol) in THF (ImL) was added TFAA (674.36 mg, 3.21 mmol) slowly at 25 °C. The mixture was stirred at 25 °C for 48h. The mixture was quenched with H2O (8 mL) at 25 °C, extracted with EtOAC (10 mL), dried over Na2SC>4, concentrated. The crude product was purified by flash chromatography (eluted with CH3CN in H2O (0.1 % TFA) from 5.0% to 95%). tert-butyl 2-cyano-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (Intermediate 10, 145 mg, 0.553 mmol, 51.65% yield) was obtained as yellow solid. LCMS calcld for C13H18N4O2 (M+H)+ m/z = 263.3, found: 263.3.
Intermediate 11. Synthesis of 2-(l-methylbenzimidazol-2-yl)-5,6,7,8-tetrahydro-4H- pyrazolof 1 ,5-a] [ 1 ,4]diazepine
[00495] Step 1. Synthesis of [5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepin-2-yl]methanol (Int-l la). To a solution of ethyl 5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int-lc 1500 mg, 3.8 mmol) in THF (15 mL) was added IM DABAL-H in hexane (11.41 mL, 11.41 mmol) at -68 °C under argon. The
mixture was stirred at 25 °C for 2 h. The reaction was added water and Isopropyl alcohol (l:l,12mL) and extracted with EtOAc (50 ml x 3), and concentrated. The crude product [5-(2- nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepin-2-yl]methanol (Int-l la, 850 mg, 2.39 mmol, 62.79% yield) was used without purification. LCMS calcld for Ci4Hi6N4O5S(M+H)+ m/z =353.1, found:353.1.
[00496] Step 2. Synthesis of 5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carbaldehyde (Int-llb). To a solution of [5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-2-yl]methanol (Int-lla, 850 mg, 2.41 mmol) in DMSO (8 mL) was added IBX (514.6 mg, 3.62 mmol) at 25 °C under argon. The mixture was stirred at 25 °C for 16 h. The mixture was filtered to afford a crude solution. The crude product was purified by flash chromatography (eluted with CH3CN in H2O from 50% to 55%). 5-(2- nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carbaldehyde (Int-llb, 570 mg, 1.59 mmol, 66.10 %yield) was obtained as white solid. LCMS calcld for C14H13CIN4O5S (M+H)+ m/z =351.1, found: 351.1.
[00497] Step 3. Synthesis of 2-(l-methylbenzimidazol-2-yl)-5-(2-nitrophenyl)sulfonyl- 4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine (Int-llc) . The solution of 5-(2- nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carbaldehyde (Int-llb, 250 mg, 1 mmol) and N2-m ethylbenzene- 1,2-diamine (244.34 mg, 2 mmol) in DMF (2 mL) was stirred at 100 °C for 18 h. The mixture was extracted with EtOAc and water, dried with brine and Na2SO4, concentrated and purified by silica gel chromatography (DCM: MeOH= 50: 1) to get the 2-(l-methylbenzimidazol-2-yl)-5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine (Int-llc, 320 mg, 0.707 mmol, 70.72%yield). LCMS calcld for C21H20N6O4S (M+H)+ m/z = 453.49, found: 453.3.
[00498] Step 4. Synthesis of 2-(l-methylbenzimidazol-2-yl)-5,6,7,8-tetrahydro-4H- pyrazolo[l,5-a][l,4]diazepine (Intermediate 11) . A solution of CS2CO3 (230.42 mg, 0.71 mmol), 2-(l-methylbenzimidazol-2-yl)-5-(2-nitrophenyl)sulfonyl-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine (Int-llc, 160 mg, 0.35 mmol) and 4- Methoxy thiophenol (74.36 mg, 0.53 mmol) in CH3CN (5 mL) was stirred at 25 °C for 3h. The mixture was filtered and the solvent was removed and purified by silica gel chromatography (DCM: MeOH=l 1 : 1) to get the 2-(l-methylbenzimidazol-2-yl)-5,6,7,8-tetrahydro-4H- pyrazolo[l,5-a][l,4]diazepine (Intermediate 11, 70 mg, 0.262 mmol, 74.05%yield). LCMS calcld for C15H17N5 (M+H)+ m/z = 268.33, found: 268.3.
Intermediate 12. Synthesis of 2',2'-difluorospiro[azetidine-3,r-tetralin]
[00499] Step 1. Synthesis of l-(2',2'-difluorospiro[azetidine-3,l'-tetralin]-l-yl)-2,2,2-trifluoro- ethanone (Int-12b). l-(2,2,2-trifluoroacetyl)spiro[azetidine-3,l'-tetralin]-2'-one (Int-12a ref. Angew. Chem. Int. Ed. 2021, 60, 7360-7365, 120 mg, 0.42 mmol) was weighed in a vial and DCM (1.69 mL) was added. Diethylaminosulfur Trifluoride (0.11 mL, 0.85 mmol) was added, and mixture stirred at room temperature for 3 hrs. Reaction was purified by silica gel column eluting with 10% Ethyl Acetate/Hexanes to yield l-(2',2'-difluorospiro[azetidine-3,r-tetralin]-l- yl)-2,2,2-trifluoro-ethanone (Int-12b, 20 mg, 0.066 mmol, 15.47% yield). LCMS calculated for C14H12F5NO (M+H)+ m/z =306.08; found: 306.1. ' H NMR (500 MHz, CD3CI) 5 ppm 2.18 - 2.30 (m, 2H) 3.06 (t, 7=6.72 Hz, 2H) 4.17 (d, 7=10.88 Hz, 1H) 4.42 (d, 7=10.09 Hz, 1H) 4.58 (d, 7=10.88 Hz, 1H) 4.79 - 4.88 (m, 1H) 7.17 (d, 7=7.64 Hz, 1H) 7.28 - 7.32 (m, 1H) 7.36 - 7.42 (m, 1H) 7.56 - 7.60 (m, 1H).
[00500] Step 2. Synthesis of 2',2'-difluorospiro[azetidine-3,r-tetralin]. (Intermediate 12). 1- (2',2'-difluorospiro[azetidine-3,r-tetralin]-l-yl)-2,2,2-trifluoro-ethanone (Int-12b, 20 mg, 0.07 mmol) was weighed in a vial and Methanol (0.65 mL) was added. Potassium Carbonate (13.58 mg, 0.1 mmol) was added, and reaction was heated to reflux for 1 hour. Reaction was then evaporated and redissolved in 10% MeOH/DCM. MeOH/DCM was then filtered and evaporated to yield crude product 2',2'-difluorospiro[azetidine-3,r-tetralin] (Intermediate 12, 12 mg, 0.057 mmol, 87.53% yield) which was used without any further purification.
Intermediate 13. Synthesis of spiro[7-thia-9,l l-diazatricyclo[6.4.0.02,6]dodeca-l(8),2(6),10- tri ene-3,3'-azeti dine]- 12-one
[00501] To a solution of tert-butyl 2-amino-3-cyano-spiro[5,6- dihy drocy cl openta[b]thiophene-4, 3 '-azetidine]- l'-carboxylate (Int-2a, 10 mg, 0.03 mmol) in Formic Acid (1 mL, 26.2 mmol) was added 1 drop H2SO4 and stirred at 105 °C for 3 h under argon. Then the reaction mixture was concentrated in vacuum to give the residue. The residue was recrystallized in ethanol to afford spiro[7-thia-9,l l-diazatricyclo[6.4.0.02,6]dodeca-
l(8),2(6),10-triene-3,3'-azetidine]-12-one (Intermediate 13, 7 mg, 0.03 mmol, 91.64% yield) as white solid. LCMS calcld for C11H11N3OS (M+H)+ m/z = 234.1, found: 234.1.
Intermediate 14. Synthesis of 3-(3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin-2- yl)-5-methyl- 1 ,2,4-oxadiazole
[00502] Step 1. Synthesis of 5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxamide (Int-14a). To a solution of 5-(2-nitrophenyl)sulfonyl-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylic acid (Int-ld, 870 mg, 2.37 mmol) , DIEA (1.65 mL, 9.5 mmol) and HATU (1354.48 mg, 3.56 mmol) in DMF (2mL) was added NH4CI (171.13 mg, 2.85 mmol) at 30 °C. The mixture was stirred at 30 °C for 2h. The mixture was diluted with DCM (30 x 2 mL), washed with water (40mL) and brine (40mL), dried over Na2SC>4, concentrated. The mixture was concentrated to afford a crude product. The crude product was purified by flash chromatography (eluted with CH3CN in H2O from 5.0% to 95%) to afford 5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2- carboxamide (Int-14a, 760 mg, 2.08 mmol, 87.59 %yield) as yellow solid. LCMS calcld for C14H15N5O5S (M+H)+ m/z =366.0 , found: 366.0.
[00503] Step 2. Synthesis of 5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carbonitrile (Int-14b) . To a solution of 5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (Int-14a, 1.5 g, 4.11 mmol) and Pyridine (1 mL, 12.32 mmol) in THF (lOmL) was added TFAA (2586.98 mg, 12.32 mmol) slowly at 25 °C. The mixture was stirred at 25 °C for 48 h. The mixture was quenched with H2O (20 mL) at 25 °C, extracted with EtOAC (20 x 3 mL), dried over Na2SO4, concentrated. The crude product was purified by silica gel chromatography (eluted with EtOAc in petroleum ether from 10% to 90%) to afford 5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carbonitrile (Int-14b, 0.8 g, 2.303 mmol, 56.10% yield) as yellow solid. LCMS calcld for C14H14N5O4S (M+H)+ m/z = 348.35, found: 348.0.
[00504] Step 3. Synthesis of 3-chloro-5-(2-nitrophenyl)sulfonyl-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carbonitrile (Int-14c). To a solution of 5-(2- nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carbonitrile (500 mg, 1.44 mmol) in MeCN (5mL) were added NCS (192.21 mg, 1.44 mmol), The mixture was stirred at 60 °C for 4h. The mixture was quenched with H2O (10 mL) at 25 °C, extracted with EtOAC (30 mL), dried over ISfeSCU, concentrated. The crude product was purified by flash chromatography (eluted with EtOAc in petroleum ether from 10% to 90%). The product 3- chloro-5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carbonitrile (Int-14c, 400 mg, 1.05 mmol, 72.78% yield) was obtained as white solid. LCMS calcld for C14H12CIN5O4S (M+H)+ m/z = 382.1, found :382.1.
[00505] Step 4. Synthesis of 3-chloro-N-hydroxy-5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamidine (Int-14d). To a solution of 3-chloro-5- (2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carbonitrile (Int-14c, 400 mg, 1.05mmol) and K2CO3 (433.75 mg, 3.14 mmol) in Ethanol (2 mL) was added HC1 NH2OH (144.58 mg, 2.1 mmol) . The mixture was stirred at 80 °C for 12 h. The solution was concentrated. The crude product was washed with water 10 mL, filtrate to obtain a white solid. The product 3-chloro-N-hydroxy-5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxamidine (Int-14d, 183 mg, 0.441 mmol, 42.11% yield) was obtained as white solid. LCMS calcld for C14H15CIN6O5S (M+H)+ m/z = 415.0, found:415.0.
[00506] Step 5. Synthesis of 3-[3-chloro-5-(2-nitrophenyl)sulfonyl-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-2-yl]-5-methyl-l,2,4-oxadiazole (Int-14e). To a solution of 3-chloro-N-hydroxy-5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxamidine (Int-14d, 183 mg, 0.441 mmol) in Acetic acid (2 mL) were added Acetic anhydride (67.56 mg, 0.66 mmol), The mixture was stirred at 100 °C for 16h. The mixture was filtered to afford a crude product. The crude product was purified by flash chromatography (eluted with CH3CN in H2O from 5.0% to 43%). 3-[3-chloro-5-(2- nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepin-2-yl]-5-methyl-l,2,4- oxadiazole (Int-14e, 163 mg, 0.371 mmol, 84.19% yield) was obtained as white solid. LCMS calcld for Ci6Hi5ClN6O5S(M+H)+ m/z = 439.1, found: 439.1.
[00507] Step 6. Synthesis of 3-(3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin- 2-yl)-5-methyl-l,2,4-oxadiazole. To a solution of 3-[3-chloro-5-(2-nitrophenyl)sulfonyl-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-2-yl]-5-methyl-l,2,4-oxadiazole (Int-14e, 150 mg, 0.34 mmol) ,4-methoxybenzenethiol (0.13 mL, 1.03 mmol) and cesium carbonate (444.34 mg, 1.37 mmol) in CH3CN (4 mL). The mixture was stirred at 20 °C for 1 h. The mixture was filtrated and purified by prep-HPLC on a C18 column with mobile phase: H2O (0.1%NH4HCO3)/CH3CN to
afford 3-(3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin-2-yl)-5-methyl-l,2,4- oxadiazole (Intermediate 14, 80 mg, 0.315 mmol, 92.26% yield) as white solid. LCMS calcld for C10H13ClN5O (M+H)+ m/z = 254.07, found: 254.0.
Intermediate 15. Synthesis of 3-chloro-2-(4,5-dimethyl-l,2,4-triazol-3-yl)-5,6,7,8-tetrahydro-
[00508] Step 1. Synthesis of ethyl 3-chloro-5-(2-nitrophenyl)sulfonyl-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int-17a). A solution of ethyl 5-(2- nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int-lc 1000 mg, 2.54 mmol) in DMF (10 mL) was added NCS (372.42 mg, 2.79 mmol) at 25 °C. Then the mixture was stirred at 80 °C for 1 h. The solution was extracted with EtOAc (10 ml), the organic phase was concentrated. The residue was purified by silica gel chromatography (eluting with EtOAc in PE from 5% to 85%). The product ethyl 3-chloro-5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int-17a, 914 mg, 2.131 mmol, 84.06% yield) was obtained as an oil. LCMS calcld for CieElnCllSkOeS (M+H)+ m/z =429.06, found: 429.2.
[00509] Step 2. Synthesis of 3-chloro-5-(2-nitrophenyl)sulfonyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (Int-17b). A solution of ethyl 3-chloro- 5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylate (Int- 17a, 500 mg, 1.17 mmol) in Ethanol (5 mL) was added NH2NH2.H2O (594.85 mg, 11.66 mmol) at 25 °C. Then the mixture was stirred at 85 °C for 2 h. The solution was extracted with EtOAc (5 ml), the organic phase was concentrated. The residue was purified by silica gel chromatography (eluting with EtOAc in PE from 5% to 95%). The product 3-chloro-5-(2- nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carbohydrazide (Int- 17b, 300 mg, 0.723 mmol, 62.03% yield) was obtained as a white solid. LCMS calcld for Ci4Hi5ClN6O5S (M+H)+ m/z =415.06, found: 415.0.
[00510] Step 3. Synthesis of 3-chloro-2-(4,5-dimethyl-l,2,4-triazol-3-yl)-5-(2- nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine (Int-17c). To a solution of 3-chloro-5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2- carbohydrazide (Int-17b, 150 mg, 0.36 mmol) in 1,4-Dioxane (3 mL) was added 1,1,1- trimethoxyethane (65.17 mg, 0.54 mmol), Methylamine in EtOH (0.17mL, 1.08mmol) and a drop of AcOH (21.7mg, 0.36mmol) at rt. The mixture was stirred at 120 °C for 16 h in a sealed vial. The solution was extracted with EtOAc (2 ml), The water phase was concentrated. The residue was purified by silica gel chromatography (eluting with MeOH in DCM from 1 % to 3%). The product 3-chloro-2-(4,5-dimethyl-l,2,4-triazol-3-yl)-5-(2-nitrophenyl)sulfonyl-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine (Int-17c, 95 mg, 0.210 mmol, 58.14% yield) was obtained as a white solid. LCMS calcld for C17H18CIN7O4S (M+H)+ m/z =452.09, found: 452.2. [00511] Step 4. Synthesis of 3-chloro-2-(4,5-dimethyl-l,2,4-triazol-3-yl)-5,6,7,8-tetrahydro- 4H-pyrazolo[l,5-a][l,4]diazepine (Intermediate 17). To a solution of 3-chloro-2-(4,5-dimethyl- l,2,4-triazol-3-yl)-5-(2-nitrophenyl)sulfonyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine (Int-17c, 75 mg, 0.17 mmol) in CH3CN (0.5 mL) was added 4-Methoxythiophenol (44.21 mg, 0.32 mmol) and CS2CO3 (137.07 mg, 0.42 mmol) at rt. The mixture was stirred at 25 °C for 2 h. The solution was concentrated. The residue was purified by silica gel on chromatography (eluting with MeOH in DCM from 3 % to 11%). The 3-chloro-2-(4,5-dimethyl-l,2,4-triazol-3- yl)-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepine (Intermediate 17, 40 mg, 0.15 mmol, 71.33% yield) was obtained as a white solid. LCMS calcld for C11H15CIN6 (M+H)+ m/z =267.11, found: 267.1.
Intermediate 16. Synthesis of (3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin-2- yl)-cyclopropyl-methanone
[00512] Step 1. Synthesis of tert-butyl 3-chloro-2-[methoxy(methyl)carbamoyl]-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate. The mixture of 5 -tert-butoxy carbonyl-3 - chloro-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxylic acid (350 mg, 1.11 mmol) and HATU (0.41 mL, 1.66 mmol) in DMF (2mL) was added N,O-Dimethylhydroxylamine Hydrochloride (162.18 mg, 1.66 mmol) at 25 °C. The mixture was stirred at 25 °C for 2 h. The mixture was filtered to afford a crude solution. The crude product was purified by flash chromatography (eluted with CH3CN in H2O from51% to 72%). tert-butyl 3-chloro-2-
[methoxy(methyl)carbamoyl]-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (230 mg, 0.641 mmol, 57.83 %yield). LCMS calcld for C15H23CIN4O4 (M+H)+ m/z =359.1 , found: 359.2.
[00513] Step 2. Synthesis of tert-butyl 3-chloro-2-(cyclopropanecarbonyl)-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate. To a solution of tert-butyl 3-chloro-2- [methoxy(methyl)carbamoyl]-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (230 mg, 0.64 mmol) in THF (1.5mL) was added IM bromo(cyclopropyl)magnesium in THF (372.49 mg, 2.56 mmol) at 0 °C under argon. The mixture was stirred at 0 °C for 2 h. The mixture was concentrated to afford a crude product. The crude product was used directly for the next step, tert-butyl 3-chloro-2-(cyclopropanecarbonyl)-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-5-carboxylate (150 mg, 0.441 mmol, 68.86% yield) was obtained as brown oil. LCMS calcld for C16H22CIN3O3 (M+H)+ m/z = 340.1, found: 340.1.
[00514] Step 3. Synthesis of (3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin-2- yl)-cyclopropyl-methanone. To a solution of tert-butyl 3-chloro-2-(cyclopropanecarbonyl)- 4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-5-carboxylate (150 mg, 0.44 mmol) in DCM (ImL) was added Tri fluoroacetic Acid (0.3 mL, 3.89 mmol) at 25 °C under argon. The mixture was stirred at 25 °C for 2 h. The mixture was concentrated to afford a crude product. The crude product was purified by flash chromatography(eluted with CH3CN in H2O from 5.0% to 45%). (3-chloro-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepin-2-yl)-cyclopropyl-methanone (100mg,0.4172mmol, 94.513% yield) was obtained as brown oil. LCMS calcld for C11H14CIN3O (M+H)+ m/z =240.3 , found: 240.3.
Intermediate 17. Synthesis of tert-butyl N-(4-cyanospiro[2H-thieno[2,3-b]thiophene-3,3'- azetidine]-5-yl)carbamate
[00515] Step 1. Synthesis of 3-(l-azabicyclo[1.1.0]butan-3-yl)thietan-3-ol (Int-17a). 2,3- dibromopropan-l-amine;hydrobromide (5000 mg, 16.79 mmol) in THF (lOOmL) at -78 °C, Phenyllithium (50.37 mL, 50.37 mmol) was added, The mixture was stirred at -78 °C for 2h,
After this time, the reaction mixture was removed from the cooling bath and warmed to room temperature over 10 minutes with stirring. After cooling back down to -78 °C, TMEDA (4877.27 mg, 41.97 mmol) was then added followed by s-BuLi (32.29 mL, 41.97 mmol) dropwise, and the resulting solution stirred for 1 hour at -78 °C. After this time, thietan-3-one (1479.63 mg, 16.79 mmol) was added dropwise in THF. The reaction was stirred for a further 1 hour at -78 °C. The mixture was diluted with EtOAC (100 mL x2), washed with water (100 mL), dried over Na2SO4, concentrated. The crude product was used directly for the next step. 3-(l- azabicyclo[1.1.0]butan-3-yl)thietan-3-ol (Int-17a, 4000 mg, 27.93 mmol, 166.36% yield) was obtained as crude yellow oil.
[00516] Step 2. Synthesis of 2-(2,2,2-trifluoroacetyl)-6-thia-2-azaspiro[3.4]octan-8-one (Int- 17b). 3-(l-azabicyclo[1.1.0]butan-3-yl)thietan-3-ol (Int-17a, 3000 mg, 20.95 mmol) in DCM at -78 °C, TFAA (2.91 mL, 20.95 mmol) was added. The mixture was stirred at -78 °C for 2h. The mixture was quenched with NaEtCCL (100 mL), extracted with DCM (100 mLx2), dried over Na2SO4, concentrated. The crude product was purified by flash chromatography (eluted with EtOAc in petroleum ether from 10% to 20%) to afford 2-(2,2,2-trifluoroacetyl)-6-thia-2- azaspiro[3.4]octan-8-one (Int-17b, 525 mg, 2.195 mmol, 10.48% yield) was obtained as crude yellow oil. LCMS calcld for CsHgFsNChS (M+H)+ m/z = 239.21, found: 240.2.
[00517] Step 3. Synthesis of 5-amino-l'-(2,2,2-trifluoroacetyl)spiro[2H-thieno[2,3- b]thiophene-3,3'-azetidine]-4-carbonitrile (Int-17c). To a solution of 2-(2,2,2-trifluoroacetyl)-6- thia-2-azaspiro[3.4]octan-8-one (Int-17b, 500 mg, 2.09 mmol) L-Proline (240.65 mg, 2.09 mmol) propanedinitrile (207.12 mg, 3.14 mmol) in DMF (5mL), Sulfur (100.52 mg, 3.14 mmol) was added. The mixture was stirred at 60 °C for 12h. The mixture was extracted with EtOAC (25 mL), dried over ISfeSCU, concentrated. The crude product was purified by Flash chromatography eluted with CH3CN in H2O from 5.0% to 95%. 5-amino-l'-(2,2,2-trifluoroacetyl)spiro[2H- thieno[2,3-b]thiophene-3,3'-azetidine]-4-carbonitrile (Int-17c, 330 mg, 1.03 mmol, 49.44 % yield) was reddish-brown solid. LCMS calcld for C11H8F3N3OS2 (M+H)+ m/z = 320.1, found: 320.1.
[00518] Step 4. Synthesis of tert-butyl N-tert-butoxy carbonyl-N-[4-cyano-l '-(2,2,2- trifluoroacetyl)spiro[2H-thieno[2,3-b]thiophene-3,3'-azetidine]-5-yl]carbamate (Int-17d). To a solution of 5-amino-l'-(2,2,2-trifluoroacetyl)spiro[2H-thieno[2,3-b]thiophene-3,3'-azetidine]-4- carbonitrile (Int-17c, 250 mg, 0.78 mmol) in MeCN (3 mL) was added BOC2O (341.73 mg, 1.57 mmol) and DMAP (9.56 mg, 0.08 mmol) at 25 °C. The reaction stirred at 25 °C for Ih. The mixture was concentrated. The crude product was purified by silica gel chromatography (eluted with EtOAc in petroleum ether from 10% to 50%). tert-butyl N-tert-butoxycarbonyl-N-[4-cyano- l'-(2,2,2-trifluoroacetyl)spiro[2H-thieno[2,3-b]thiophene-3,3'-azetidine]-5-yl]carbamate (300
mg, 0.577 mmol, 73.75% yield) was obtained as brown solid. LCMS calculated for C16H16F3N3O3S2 (M+Na)+ m/z = 442.2, found: 442.2.
[00519] Step 5. Synthesis of tert-butyl N-(4-cyanospiro[2H-thieno[2,3-b]thiophene-3,3'- azetidine]-5-yl)carbamate (Intermediate 17). To a solution of tert-butyl N-[4-cyano-l'-(2,2,2- trifluoroacetyl)spiro[2H-thieno[2,3-b]thiophene-3,3'-azetidine]-5-yl]carbamate (Int-17d, 300 mg, 0.58 mmol) in Methanol (3mL) was added K2CO3 (239.41 mg, 1.73 mmol) at 25 °C. The reaction stirred at 70 °C for 1 h. The mixture was filtered. The crude product was purified by silica gel chromatography (eluted with MeOH in DCM from 5% to 30%). tert-butyl N-(4-cyanospiro[2H- thieno[2,3-b]thiophene-3,3'-azetidine]-5-yl)carbamate (Intermediate 17, 110 mg, 0.340 mmol, 58.90% yield) was obtained as yellow solid. LCMS calculated for C14H17N3O2S2 (M+H)+ m/z = 324.2, found: 324.2.
[00520] l-(2,2,2-trifluoroacetyl)spiro[azetidine-3,T-tetralin]-2'-one (20 mg, 0.07 mmol) was weighed in a vial and Methanol was added. Potassium Carbonate (19.52 mg, 0.14 mmol) was added, and reaction stirred at 80 °C for 1 hour. Reaction was evaporated and redissolved in 10% MeOH/DCM and then filtered. MeOH/DCM was evaporated to yield product spiro[azetidine-3,l'- tetralin]-2'-one (13 mg, 0.069 mmol, 98.33% yield). *HNMR (500 MHz, CD3CI) 5 ppm 2.62 - 2.69
(m, 2H) 3.00 (t, J=6.66 Hz, 2H) 3.73 (d, =8.07 Hz, 2H) 4.28 (d, J=8.07 Hz, 2H) 7.20 (s, 1H) 7.23
- 7.29 (m, 14H) 7.38 (s, 1H) 7.82 (d, J=7.82 Hz, 1H).
Intermediate 19. Synthesis of 2-aminospiro[6H-thieno[2,3-c]thiophene-4,3'-azetidine]-3- carbonitrile
[00521] Step 1. Preparation of 01-tert-butyl O3-methyl 3-benzylsulfanylazetidine-l,3- dicarboxylate (Int-19a). To a solution of 01-tert-butyl O3-methyl azetidine-l,3-dicarboxylate
(1000 mg, 4.65 mmol) in THF (10 mL) was added IM LDA in THF (4.65 mL, 9.29 mmol) at - 78 °C, the mixture was then stirred at -78 °C for 1 h, then dibenzyldisulfide (2289.34 mg, 9.29 mmol) was added at -78 °C. The mixture was then stirred at -78 °C for 1 h. The reaction mixture was quenched by NH4CI (5 mL), diluted with EtOAc (30 x 2 mL), washed with brine (20 ml), dried over Na2SO4, concentrated. The crude product was purified by silica gel chromatography (eluted with EtOAc in petroleum ether from 0% to 10%). 01-tert-butyl O3-methyl 3- benzylsulfanylazeti dine- 1,3 -dicarboxylate (Int-19a, 610 mg) was obtained as a crude yellow oil. [00522] Step 2. Preparation of tert-butyl 3-benzylsulfanyl-3-(3-methoxy-3-oxo- propanoyl)azetidine-l -carboxylate (Int-19b). To a solution of methyl acetate (0.58 mL, 7.23 mmol) in THF (10 mL) was added IM LiHMDS in THF (7.23 mL, 7.23 mmol) at -78 °C. The mixture was then stirred at -78 °C for 1 h before 01-tert-butyl O3-methyl 3- benzylsulfanylazeti dine- 1,3 -dicarboxylate (Int-19a, 610 mg, 1.81 mmol) was added at -78 °C. The resulting mixture was stirred for Ih at -78 °C and then allowed to warm up to room temperature for 45 min. The reaction mixture was quenched by NH4CI (5 mL), diluted with EtOAc (20 x 2 mL), washed with brine (10 ml), dried over Na2SO4, concentrated.
[00523] The crude product was purified by silica gel chromatography (eluted with EtOAc in petroleum ether from 0% to 20%) to afford tert-butyl 3-benzylsulfanyl-3-(3-methoxy-3-oxo- propanoyl)azetidine-l -carboxylate (Int-19b 270 mg,0.712mmol, 39.36% yield) as a colorless oil.
[00524] Step 3. Preparation of tert-butyl 3-benzylsulfanyl-3-(l,3-dihydroxypropyl)azetidine- 1-carboxylate (Int-19c). To a solution of tert-butyl 3-benzylsulfanyl-3-(3-methoxy-3-oxo- propanoyl)azetidine-l -carboxylate (Int-19b, 1200 mg, 3.16 mmol) in DCM (10 mL) was added IM DIBAL-H in PhMe (12.6 mL, 12.65 mmol) at -78 °C. The mixture was stirred at 25 °C for 16h under argon. The mixture was quenched with MeOH (20 mL) at 0 °C and filtered, extracted with EtOAC (30 x 3 mL), dried over Na2SO4, concentrated. The crude product was purified by silica gel chromatography (eluted with MeOH in DCM from 0% to 10%). The product tert-butyl 3-benzylsulfanyl-3-(l,3-dihydroxypropyl)azetidine-l-carboxylate (Int-19c, 350 mg, 0.99 mmol, 31.31% yield) was obtained as a colorless oil. LCMS calcld for CisH27NO4SNa (M+Na)+ m/z = 376.2; found: 376.2.
[00525] Step 4. Preparation of tert-butyl 8-hydroxy-5-thia-2-azaspiro[3.4]octane-2- carboxylate (Int-19d).
[00526] To a solution of tert-butyl 3-benzylsulfanyl-3-(l,3-dihydroxypropyl)azetidine-l- carboxylate (Int-19c, 300 mg, 0.85 mmol) in pyridine (8 mL) was added TosCl (404.51 mg, 2.12 mmol) at 25 °C. The mixture was stirred at 25 °C for 16h. The mixture was concentrated to afford a crude product. The crude product was purified by silica gel chromatography (eluted with
EtOAc in petroleum ether from 10% to 40%) to afford tert-butyl 8-hydroxy-5-thia-2- azaspiro[3.4]octane-2-carboxylate (Int-19d, 130 mg, 0.530 mmol, 62.43% yield) as a colorless oil. LCMS calcld for C11H20NO3S (M+H)+ m/z = 246.1, found: 190.1 (-tBu).
[00527] Step 5. Preparation of tert-butyl 8-oxo-5-thia-2-azaspiro[3.4]octane-2-carboxylate (Int-19e). To a solution of tert-butyl 8-hydroxy-5-thia-2-azaspiro[3.4]octane-2-carboxylate (Int- 19d, 350 mg, 1.43 mmol) in DCM (8 mL) and DMSO (2 mL) was added TEA (0.99 mL, 7.13 mmol) and pyridine sulfur trioxide (681.17 mg, 4.28 mmol) at 0 °C. The mixture was stirred at 25 °C for 16 h. The mixture was quenched with H2O at 25 °C, extracted with DCM, dried over Na2SO4 and concentrated. The crude product was purified by silica gel chromatography (eluted with EtOAc in petroleum ether from 10% to 25%). Tert-butyl 8-oxo-5-thia-2- azaspiro[3.4]octane-2-carboxylate (Int-19e, 260 mg, 1.07 mmol, 74.90% yield) was obtained as a white solid. LCMS calcld for C11H18NO3S (M+H)+ m/z = 244.1, found: 188.1 (-tBu).
[00528] Step 6. Preparation of tert-butyl 2-amino-3-cyano-spiro[6H-thieno[3,4-b]thiophene- 4,3 '-azetidine]- l'-carboxylate (Int-19f)
[00529] A solution of tert-butyl 8-oxo-5-thia-2-azaspiro[3.4]octane-2-carboxylate (Int-19e, 300 mg, 1.23 mmol), sulfur (63.03 mg, 1.85 mmol), NELOAc (143.88 mg, 1.85 mmol) in ethanol (3 mL) was added propanedinitrile (122.17 mg, 1.85 mmol) slowly at 15 °C. Then the mixture was stirred at 60 °C for 16h. The crude product was purified by flash chromatography (eluted with CH3CN in H2 O from 5.0% to 95%). tert-butyl 2-amino-3-cyano-spiro[6H-thieno[3,4- b]thiophene-4,3'-azetidine]-l'-carboxylate (Int-19f, 150 mg, 0.464 mmol, 37.62% yield) was obtained as a white solid. LCMS calcld for Ci4Hi7N3O2S2Na (M+Na)+ m/z = 346.1, found: 346.1.
[00530] Step 7. Preparation of 2-aminospiro[6H-thieno[2,3-c]thiophene-4,3'-azetidine]-3- carbonitrile;2,2,2-trifluoroacetic acid (Intermediate 19)
[00531] A solution of trifluoroacetic acid (0.31 mL, 4.11 mmol) and tert-butyl 2-amino-3- cyano-spiro[6H-thieno[2,3-c]thiophene-4,3'-azetidine]-l'-carboxylate (Int-19f, 50 mg, 0.15 mmol) in DCM (3 mL) was stirred at 25 °C for Ih. The solvent was removed to get 2- aminospiro[6H-thieno[2,3-c]thiophene-4,3'-azetidine]-3-carbonitrile (Intermediate 19, 52 mg, 99.71% -yield) as a crude brown oil. LCMS calculated for C9H10N3S2 (M+H)+ m/z =224.1, found: 224.1.
Intermediate 20. Synthesis of 2-amino-5-fluoro-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'- azetidine]-3-carbonitrile
[00532] Step 1. Preparation of tert-butyl 3 -(3 -benzyloxy- 1-hy droxy-propyl)-3 -cyano- azetidine-1 -carboxylate (Int-20a). To a solution of tert-butyl 3 -cyanoazetidine- 1 -carboxylate (500 mg, 2.74 mmol) in THF (5 mL) was added LDA (1.92 mL, 3.84mmol) portion wise at -70 °C under N2. Then the mixture was stirred at -70 °C for Ih, and 3-benzyloxypropanal (630.78 mg, 3.84 mmol) was added at -70 °C.
[00533] The resulting mixture was stirred at -70 °C for 30min and warmed to rt for Ih. The reaction mixture was quenched with saturated NH4CI solution, and then extracted with EtOAc. The organic layer was washed with water and brine, dried over Na2SO4, concentrated and purified by flash column chromatography (silica gel, eluting with 0% to 40% EtOAc/PE) to afford tert-butyl 3 -(3 -benzyloxy- l-hydroxy-propyl)-3-cyano-azeti dine- 1 -carboxylate (Int-20a, 450 mg, 1.30 mmol, 47.34% yield) as a colorless oil. LCMS calculated for C19H27N2O4 (M+H)+ m/z =347.20; found: 247.3 (-Boc).
[00534] Step 2. Preparation of tert-butyl 3 -(3 -benzyloxy- l-fluoro-propyl)-3-cyano-azetidine- 1-carboxylate (Int-20b). To a solution of tert-butyl 3 -(3 -benzyloxy- 1-hy droxy-propyl)-3 -cyano- azetidine-1 -carboxylate (Int-20a, 100 mg, 0.29 mmol) in DCM (2 mL) was added diethylaminosulfur trifluoride (0.08 mL, 0.58 mmol) portion-wise at -70 °C under N2. The mixture was stirred at -70 °C for 30min, then warmed to rt for 4h. The reaction mixture was quenched with ice water and extracted with EtOAc. The organic layer was washed with water and brine, dried over Na2SO4, concentrated, and purified by flash column chromatography (silica gel, eluting with 0% to 40% EtOAc/PE) to afford tert-butyl 3 -(3 -benzyloxy- l-fluoro-propyl)-3- cyano-azetidine-1 -carboxylate (Int-20b, 72 mg, 0.207 mmol, 71.59% yield) as a colorless oil. LCMS calculated for C19H26FN2O3 (M+H)+ m/z =349.19; found: 249.3 (-Boc)
[00535] Step 3. Preparation of tert-butyl 3-cyano-3-(l-fluoro-3-hydroxy-propyl)azetidine-l- carboxylate (Int-20c). The mixture of tert-butyl 3 -(3 -benzyloxy- 1 -fluoro-propyl)-3 -cyano- azetidine-1 -carboxylate (Int-20b, 2000 mg, 5.74 mmol), 2,3-dichloro-5,6-dicyano-l,4- benzoquinone (5212.25 mg, 22.96 mmol) in DCM (20 mL) and water (3 mL) was stirred at 45
°C for 20h. The resulting mixture was filtered. The filtrate was treated with saturated NaHCCh, extracted with DCM. The organic layer was washed with brine, dried over Na2SO4, concentrated and purified by flash column chromatography (silica gel, eluting with 0% to 60% EtOAc/PE) to afford tert-butyl 3-cyano-3-(l-fluoro-3-hydroxy-propyl)azetidine-l-carboxylate (Int-20c, 1090 mg, 4.22 mmol, 73.52% yield) as a brown oil. LCMS calculated for C12H20FN2O3 (M+H)+ m/z =259.2; found: 203.2 (-tBu).
[00536] Step 4. Preparation of tert-butyl 3 -cyano-3-(l -fluoro-3 -methyl sulfonyloxy - propyl)azetidine-l -carboxylate (Int-20d). To a solution of tert-butyl 3 -cyano-3-(l -fluoro-3 - hydroxy-propyl)azetidine-l -carboxylate (Int-20c, 100 mg, 0.39 mmol) in DCM (2 mL) were added methanesulfonyl chloride (0.05 mL, 0.58 mmol) and TEA (0.11 mL, 0.77 mmol) at 0 °C. Then the mixture was stirred at rt for Ih. The resulting mixture was quenched with water, and then extracted with EtOAc. The organic layer was washed with brine, dried over ISfeSCh, concentrated to afford tert-butyl 3-cyano-3-(l-fluoro-3-methylsulfonyloxy-propyl)azetidine-l- carboxylate (Int-20d, 130 mg, 0.387 mmol, 99.82% yield) as a green oil. LCMS calculated for C13H22FN2O5S (M+H)+ m/z =337.13; found: 281.0 (-tBu).
[00537] Step 5. Preparation of tert-butyl 3-cyano-3-(l-fluoro-3-iodo-propyl)azetidine-l- carboxylate (Int-20e). The mixture of tert-butyl 3-cyano-3-(l-fluoro-3-methylsulfonyloxy- propyl)azetidine-l -carboxylate (Int-20d, 130 mg, 0.39 mmol) and Nal (173.78 mg, 1.16 mmol) in acetone (2 mL) was stirred at rt overnight. The reaction mixture was stirred at 60 °C for 5h. The mixture was cooled to room temperature and the solvent was removed under reduced pressure. The mixture was diluted with water, extracted with EtOAc, washed with brine, dried over Na2SO4, concentrated and purified by flash column chromatography (silica gel, eluting with 0% to 70% EtOAc/PE) to afford tert-butyl 3 -cyano-3-(l -fluoro-3 -iodo-propyl)azeti dine- 1- carboxylate (Int-20e, 103 mg, 0.28 mmol, 72.39% yield) as a colorless oil. LCMS calculated for C12H19FIN2O2 (M+H)+ m/z =369.05; found: 313.0 (-tBu).
[00538] Step 6. Preparation of tert-butyl 5-fluoro-8-oxo-2-azaspiro[3.4]octane-2-carboxylate (Int-20f) To a solution of tert-butyl 3-cyano-3-(l-fluoro-3-iodo-propyl)azetidine-l-carboxylate (Int-20e, 1450 mg, 3.94 mmol) in THF (15 mL) was added n-butyllithium (504.56 mg, 7.88 mmol) at -78 °C under N2. Then the mixture was stirred at -78 °C for 30min. After completion of the reaction, HOAc (0.68 mL, 11.81 mmol) was added dropwise at -78 °C. The resulting mixture was diluted with EtOAc and water was added. The organic layer was washed with brine, dried over Na2SO4, concentrated and purified by flash column chromatography (silica gel, eluting with 0% to 70% EtOAc/PE) to afford tert-butyl 5-fluoro-8-oxo-2-azaspiro[3.4]octane-2-carboxylate (Int-20f, 655 mg, 2.69 mmol, 68.37% yield) as a yellow oil. LCMS calculated for C12H19FNO3 (M+H)+ m/z =244.14; found: 188.0 (-tBu).
[00539] Step 7. Preparation of tert-butyl 2-amino-3-cyano-5-fluoro-spiro[5,6- dihy drocy cl openta[b]thiophene-4, 3 '-azetidine]- l'-carboxylate (Int-20g)
[00540] To a solution of tert-butyl 5-fluoro-8-oxo-2-azaspiro[3.4]octane-2-carboxylate (Int- 20f, 600 mg, 2.47 mmol), sulfur (118.68 mg, 3.7 mmol), NE OAc (287.83 mg, 3.7 mmol) in ethanol (12mL) was added propanedinitrile (244.4 mg, 3.7 mmol) at rt. Then the mixture was stirred at 30 °C for 3h. Diluted with water and extracted with EtOAc. The combined extracts were dried, filtered and concentrated and the crude product was purified to afford tert-butyl 2- amino-3-cyano-5-fluoro-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-r-carboxylate (Int-20g, 407 mg, 1.26 mmol, 51.03% yield) as a light-yellow solid. LCMS calculated for C15H19FN3O2S (M+H)+ m/z =324.12; found: 224.0 (-tBu).
[00541] Step 8. Preparation of 2-amino-5-fluoro-spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3'-azetidine]-3-carbonitrile;hydrochloride (Intermediate 20). To a solution of tert-butyl 2- amino-3-cyano-5-fluoro-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-r-carboxylate (Int-20g, 360 mg, 1.11 mmol) in DCM (15mL) was added HC1 in dioxane (3 mL, 12 mmol). Then the mixture was stirred at rt overnight. The resulting mixture was diluted with DCM, concentrated to afford 2-amino-5-fluoro-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'- azetidine]-3-carbonitrile; hydrochloride (Intermediate 20, 322 mg, 1.103 mmol, 99.12% yield) as a brown solid. LCMS calculated for C10H11FN3S (M+H)+ m/z =224.07; found: 224.2.
Intermediate 21. Synthesis of 2-aminospiro[4a,5,5a,6-tetrahydrocyclopropa[f]benzothiophene-
[00542] Step 1. Preparation of bicyclo[3.1.0]hexan-2-one (Int-21a). NaH (4872.11 mg, 121.8 mmol) in DMSO (50mL) was added trimethyl sulfoxonium iodide (26805.12 mg, 121.8 mmol) at 25 °C. Then the mixture was stirred at 25 °C for 1 h. The cyclopent-2-en-l-one (10.2 mL, 121.8 mmol) was added to the mixture at 25 °C for 30 mins. After 30 mins, the mixture was stirred at 50 °C for 2 h. The solution was extracted with MTBE (100 ml), the organic phase was concentrated. The residue was purified by silica gel on chromatography (eluting with 100% DCM). The product bicyclo[3.1.0]hexan-2-one (Int-21a, 2000 mg, 20.81 mmol, 17.08% yield) was obtained as a colorless oil. LCMS calculated for CLFEO (M+H)+ m/z =97.07, found: 97.2.
[00543] Step 2. Preparation of 2-(l-azabicyclo[1.1.0]butan-3-yl)bicyclo[3.1.0]hexan-2-ol (Int-21b). To a solution of 2, 3 -dibromopropan- 1 -amine; hydrobromide (4000 mg, 13.43 mmol) in THF (80 mL) at -78 °C, PhLi (3386.32 mg, 40.29 mmol) was added. The mixture was stirred at -78 °C for 2h. The reaction mixture was then removed from the cooling bath and warmed to rt for over 10 mins with stirring. After cooling back down to -78 °C, TMDEA (3901.82 mg, 33.58 mmol) was added, followed by the dropwise addition of s-BuLi (6703.94 mg, 33.58 mmol). The resulting solution was stirred for 1 hour at -78 °C. Then, bicyclo[3.1.0]hexan-2-one (Int-21a, 1291.16 mg, 13.43 mmol) was added dropwise in THF. The reaction was stirred for another 1 hour at -78 °C. The mixture was diluted with EtOAc (40 mL), washed with water (200 mL), dried over Na2SO4, concentrated to afford 2-(l-azabicyclo[1.1.0]butan-3-yl)bicyclo[3.1.0]hexan- 2-ol (Int-21b, 3500 mg) as a crude yellow oil.
[00544] Step 3. Preparation of l-(2,2,2-trifluoroacetyl)spiro[azetidine-3,2'-norcarane]-3'-one (Int-21c). A solution of 2-(l-azabicyclo[1.1.0]butan-3-yl)bicyclo[3.1.0]hexan-2-ol (Int-21b, 3500 mg, 23.15 mmol) in DCM (66 mL) was added TFAA (6.44 mL, 46.29 mmol) at -78 °C. Then the mixture was stirred at -78 °C for 1 h. The solution was added EtOAc (20 ml), washed with aqueous NaHCOs until pH=7. Then the organic phase was concentrated. The residue was purified by silica gel chromatography (eluting with EtOAc in PE from 17% to 50%). The product l-(2,2,2-trifluoroacetyl)spiro[azetidine-3,2'-norcarane]-3'-one (Int-21c, 1000 mg, 4.05 mmol, 17% yield) was obtained as a brown oil. LCMS calculated for C11H13F3NO2 (M+H)+ m/z =248.09, found:248.1.
[00545] Step 4. Preparation of 2-amino-l'-(2,2,2-trifluoroacetyl)spiro[4a,5,5a,6- tetrahydrocyclopropa[f]benzothiophene-4,3'-azetidine]-3-carbonitrile (Int-21d)
[00546] To a solution of l-(2,2,2-trifluoroacetyl)spiro[azetidine-3,2'-norcarane]-3'-one (Int- 21c, 180 mg, 0.73 mmol) in ethanol (2mL) was added NH4OAC (84.97 mg, 1.09 mmol), propanedinitrile (72.15 mg, 1.09 mmol) and S8 (34.95 mg, 1.09 mmol) at 25 °C. The reaction stirred at 55 °C for 16 h.
[00547] The solution was concentrated and purified by prep-HPLC (eluting with CH3CN in H2O from 5% to 95%). The product of 2-amino-l'-(2,2,2-trifhioroacetyl)spiro[4a,5,5a,6- tetrahydrocyclopropa[f]benzothiophene-4,3'-azetidine]-3-carbonitrile (Int-21d, 100 mg, 0.306 mmol, 41.96% yield) was obtained as a yellow solid. LCMS calculated for C14H13F3N3OS (M+H)+ m/z =328.1, found:328.0.
[00548] Step 5. Preparation of 2-aminospiro[4a,5,5a,6- tetrahydrocyclopropa[f]benzothiophene-4,3'-azetidine]-3-carbonitrile (Intermediate 21) [00549] To a solution of 2-amino-l'-(2,2,2-trifhioroacetyl)spiro[4a,5,5a,6- tetrahydrocyclopropa[f]benzothiophene-4,3'-azetidine]-3-carbonitrile (Int-21d, 80 mg, 0.24
mmol) in methanol (5mL) was added K2CO3 (67.56 mg, 0.49 mmol) at 25 °C. The reaction stirred at 80 °C for Ih. The solution was concentrated, and the crude product was purified by silica gel chromatography (eluting with MeOH in DCM from 0% to 20%). The product of 2- aminospiro[4a,5,5a,6-tetrahydrocyclopropa[f]benzothiophene-4,3'-azetidine]-3-carbonitrile (Intermediate 21, 52 mg, 0.225 mmol, 91.98% yield) was obtained as a yellow solid. LCMS calculated for C12H14N3S (M+H)+ m/z =232.1, found:232.1.
Intermediate 22. Synthesis of 2,3,4, 5-tetrahydropyrido[3,2-f][l,4]thiazepine
lnt-22a lnt-22b Intermediate 22
[00550] Step 1 : Synthesis of 2-(2-aminoethylsulfanyl)pyridine-3-carboxamide (Int-22a)To the suspension of 2-aminoethanethiol;hydrochloride (4.25 g, 37.41 mmol) and 2-chloropyridine- 3-carboxamide (3.91 g, 24.97 mmol) in Ethanol (50mL) was added the solution of KOH (4.2 g, 70 mmol) in Ethanol (25mL) at reflux dropwise over 0.5 h. The mixture was stirred for 1 h and cooled to rt. It was diluted with Na2COs solution (100 mL) and extracted with EtOAc (100 mL). It was dried over Na2SO4 and concentrated in vacuo to give the crude product 2-(2- aminoethylsulfanyl)pyridine-3-carboxamide (Int-22a, 6.00 g) as a white solid. LCMS calculated for C8H12N3OS (M+H)+ m/z =198.26; found: 198.4. 'H NMR (400 MHz, DMSO) 5 8.48 (dd, J= 4.8, 1.7 Hz, 1H), 8.00 (s, 1H), 7.81 (dd, J= 7.6, 1.7 Hz, 1H), 7.54 (s, 1H), 7.16 (dd, J= 7.6, 4.8 Hz, 1H), 3.18 - 3.06 (m, 2H), 2.74 (t, J= 6.9 Hz, 2H), 1.56 (br, 2H).
[00551] Step 2. Synthesis of 3,4-dihydro-2H-pyrido[3,2-f][l,4]thiazepin-5-one (Int-22b). The suspension of 2-(2-aminoethylsulfanyl)pyridine-3-carboxamide (Int-22a, 2 g, 8.11 mmol) and Benzoic Acid (0.2 g, 1.64 mmol) in m-Xylene (lOOmL) was heated to 120 °C for 16 h. It was cooled to rt and the solvent was removed in vacuo and the residue was diluted with DMSO (10 mL) and purified using reverse phase FC (eluant with MeCN in base water 5 to 20%), concentrated in vacuo to give the product 3,4-dihydro-2H-pyrido[3,2-f][l,4]thiazepin-5-one (Int- 22b, 420 mg, 2.33 mmol, 28.73 %yield) as a white solid. LCMS calculated for C8H8N2OS (M+H)+ m/z =181.04; found: 181.4. 'HNMR (400 MHz, DMSO) 5 8.48 (dd, J= 4.8, 1.7 Hz, 1H), 8.00 (s, 1H), 7.81 (dd, J= 7.6, 1.7 Hz, 1H), 7.54 (s, 1H), 7.16 (dd, J= 7.6, 4.8 Hz, 1H), 3.18 - 3.06 (m, 2H), 2.74 (t, J= 6.9 Hz, 2H), 1.56 (br, 2H).
[00552] Step 3: Synthesis of 2,3,4,5-tetrahydropyrido[3,2-f][l,4]thiazepine (Intermediate 22). A solution of LiAlH4 (84.23 mg, 2.22 mmol) in THF (30mL) was cooled 0 °C, then a solution of 3,4-dihydro-2H-pyrido[3,2-f][l,4]thiazepin-5-one (Int-22b, 200 mg, 1.11 mmol) in
THF (30 mL) was added dropwise. The reaction mixture was stirred at rt overnight and quenched with lOH2O.Na2SO4.The mixture was filtrated, and the filtrate was concentrated under reduced pressure. The crude residue was purified by silica gel column chromatography (DCM/MeOH, 20: 1) to afford the title product 2,3,4,5-tetrahydropyrido[3,2-f][l,4]thiazepine, 82 mg, 44.45% yield as a colorless oil. LCMS calculated for C8H10N2S (M+H)+ m/z =167.06; found: 167.1.
Example 1: Exemplary synthesis of l-[6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]-2- [[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4- yl]spiro[azetidine-3,9'-fluorene]-3'-ol (Compound 1).
[00553] Step 1 : Synthesis of tert-butyl (lA,55)-3-(2,6-dichloropyrimidin4-yl)-3,8- diazabicyclo[3.2.1]octane-8-carboxylate (la). To a solution of tert-butyl (lR,5S)-3,8- diazabicyclo[3.2.1]octane-8-carboxylate (1.157g , 5.45 mmol) in DCM (20 mL) at -60 °C were added 2,4,6-trichloropyrimidine (1 g, 5.45 mmol) and DIEA (2.7 mL, 16.36 mmol) under N2. Then the mixture was stirred at -60 °C for Ih. The resulting mixture was quenched with saturated aqueous NH4CI (60 mL), and then extracted with DCM (60 mL x 2). The organic layer was washed with brine (50 mL), dried over ISfeSCL, concentrated and purified by flash column chromatography (silica gel, eluting with 0% to 50% petroleum ether/EtOAc) to afford crude tertbutyl (lR,5S)-3-(2,6-dichloropyrimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (la, 1.566 g, 4.359 mmol, 79.9% yield) as a light white solid. LCMS calcld for C15H21Q2N4O2 (M+H)+ m/z = 359.1; found: 359.2.
[00554] Step 2: Synthesis of tert-butyl (lA,55)-3-(6-chloro-2-(((2A,7a5)-2-fluorotetrahydro- U/-pyrrolizin-7a(5rt)-yl)methoxy)pyrimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (lb). The mixture of tert-butyl (lR,5S)-3-(2,6-dichloropyrimidin-4-yl)-3,8-
diazabicyclo[3.2.1]octane-8-carboxylate (la, 700 mg, 1.95 mmol) and [(2R,8S)-2-fluoro- l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methanol (931 mg, 5.85 mmol) in 1,4-dioxane (1 mL) was added DIEA (1.02 mL, 5.85 mmol) and it was stirred at 90 °C overnight. After the reaction was completed, the reaction mixture diluted with water, and extracted with DCM. The combined organic layers were washed with brine, dried over anhydrous Na2SO4, filtered, and concentrated to give the crude product. The crude product was purified by column chromatography on a silica gel column (DCM / MeOH = 20/1 to 10/1) to afford tert-butyl (lR,5S)-3-[6-chloro-2-[[(2R,8S)- 2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3,8- diazabicyclo[3.2.1]octane-8-carboxylate (lb, 501 mg, 1.04 mmol, 53.3% yield) and recovered starting material (198 mg). LCMS calcld for C23H34CIFN5O3 (M+H)+ m/z =482.23; found: 482.4.
[00555] Step 3 : Synthesis of 3-(methoxymethoxy)-9H-fluorene (1c). To a solution of 9H- fluoren-3-ol (1.70 g, 9.33 mmol) and bromo(m ethoxy )m ethane (1.17 mL, 14.0 mmol) in acetonitrile (30 mL) was added DIEA (4.87 mL, 28.0 mmol). The mixture was stirred at rt for 1 h. The mixture was diluted with EtOAc (30 mL), washed with water (50 mL) and saturated brine (30 mL), dried over Na2SO4, concentrated, and purified by flash column chromatography (silica gel, eluting with EtOAc in petroleum ether 10% to 20%) to afford 3-(methoxymethoxy)-9H- fluorene (1c, 1.34 g, 5.92 mmol, 63.5% yield) as a white solid. LCMS calcld for C15H15O2 (M+H)+ m/z = 227.1; found: 227.1.
[00556] Step 4: Synthesis of (9-(hydroxymethyl)-3-(methoxymethoxy)fluoren-9-yl)methanol (Id). To a solution of paraformaldehyde (497mg, 5.52mmol, 2.5 eq) in DMF (3 mL) was added MeONa (29.8mg, 0.550mmol, 0.25 eq) at 0 °C. Then the mixture was stirred at 0 °C for 10 min, and 3-(methoxymethoxy)-9H-fluorene (1c, 500 mg, 2.21 mmol) in DMSO (1 mL) was added drop wise at 0 °C. The mixture was allowed to warm to rt and stirred for 2 h. The mixture was quenched with IN HC1, diluted with water (30 mL), extracted with EtOAc (30 mL). The organic phase was dried and purified by flash column chromatography (silica gel, eluting with 5% MeOH in DCM) to afford (9-(hydroxymethyl)-3-(methoxymethoxy)fluoren-9-yl)methanol (Id, 400 mg, 1.40 mmol, 63.2% yield) as a yellow oil. LCMS calcld for C17H19O4 (M+H)+ m/z = 287.1; found: 287.1.
[00557] Step 5: Synthesis of (3-(methoxymethoxy)-9-(methylsulfonyloxymethyl)fluoren-9- yl)methyl methanesulfonate (le). To the solution of (9-(hydroxymethyl)-3- (methoxymethoxy)fluoren-9-yl)methanol (Id, 100 mg, 0.35 mmol, 1 eq) and Eh N (0.22 mL, 1.57 mmol) in DCM (3 mL) was added MsCl (160 mg, 1.40 mmol, 4 eq) at 0 °C and the mixture was stirred for 2 h. Then the mixture was partitioned between with EtOAc/H2O. The combined organic layer was washed with brine, dried over Na2SO4 and concentrated. The residue was
purified by FC (silica gel, eluting EtOAc in petroleum ether 0 to 50%) to afford (3- (methoxymethoxy)-9-(methylsulfonyloxymethyl)fluoren-9-yl)methyl methanesulfonate (le, 150 mg, 0.339 mmol, 97.1% yield) as a colorless oil. LCMS calcld for C19H23O8S2 (M+H)+ m/z = 443.1; found: 459.8.
[00558] Step 6: Synthesis of l-(3,4-dimethylbenzyl)-3'-(methoxymethoxy)spiro[azetidine- 3,9'-fluorene] (If). A mixture of (3-(methoxymethoxy)-9-(methylsulfonyloxymethyl)fluoren-9- yl)methyl methanesulfonate (le, 150 mg, 0.34 mmol) and 2,4-Dimethoxybenzylamine (1 mL) was stirred at 110 °C for 16 h. Then the mixture was cooled to rt and NaHCCh (5 mL) aqueous solution was added to quench the reaction. The mixture was extracted with EtOAc (10 mL). The combined organic layer was washed with brine, dried over Na2SO4 and concentrated. The residue was purified by FC (silica gel, eluting with EtOAc in petroleum ether 0 to 22%) to afford l-(3,4-dimethylbenzyl)-3'-(methoxymethoxy)spiro[azetidine-3,9'-fluorene] (If, 110 mg, 0.264 mmol, 77.7% yield) as a yellow oil. LCMS calcld for C26H28NO4 (M+H)+ m/z = 418.2; found: 417.8.
[00559] Step 7: Synthesis of spiro[azetidine-3,9'-fluorene]-3'-ol (1g). To a solution of 1 -(3,4- dimethylbenzyl)-3'-(methoxymethoxy)spiro[azetidine-3,9'-fluorene] (450 mg, 1.08 mmol) in DCE (5 mL) was added 1 -chloroethyl carbonochloridate (462 mg, 3.23 mmol) and the reaction was stirred at 100 °C for 6h. Then the mixture was concentrated in vacuo to dry. Methanol (5 mL) was added and the mixture was heated to 70 °C for 4 h. The mixture was filtered and purified by prep-HPLC on a C18 column (5 um, 50 x 150 mm) with mobile phase: H2O (0.1% NH4HCO3 / MeOH) at flow rate: 35 mL/min to afford spiro[azetidine-3,9'-fluorene]-3'-ol (148 mg, 0.663 mmol, 61.5% yield) as a yellow solid. LCMS calcld for C15H14NO (M+H)+ m/z =224.1; found: 224.0.
[00560] Step 8: Synthesis of tert-butyl (lR,5S)-3-[6-(3'-hydroxyspiro[azetidine-3,9'-fluorene]- l-yl)-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3,8- diazabicyclo[3.2.1]octane-8-carboxylate (Ih). The mixture of tert-butyl (lR,5S)-3-[6-chloro-2- [[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3,8- diazabicyclo[3.2.1]octane-8-carboxylate (30.0 mg, 0.06 mmol), spiro[azetidine-3,9'-fluorene]-3'- ol (69.5 mg, 0.31 mmol) and cesium carbonate (121 mg, 0.370 mmol) in NMP (1.5 mL) was irritated to 135 °C using microwave for 3 h under Ar. Then the mixture was purified by prep- HPLC on a C18 column (5 um, 50 x 150 mm) with mobile phase: H2O (0.1% NH4HCO3 / MeOH) at flow rate: 35 mL/min to afford tert-butyl (lR,5S)-3-[6-(3'-hydroxyspiro[azetidine- 3,9'-fhiorene]-l-yl)-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizi8-yl]methoxy]pyrimidi4- yl]-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (Ih, 16 mg, 0.0239 mmol, 38.4% yield) as a white solid. LCMS calcld for C38H46FN6O4 (M+H)+ m/z = 669.4; found: 669.0.
[00561] Step 9: Synthesis of l-[6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octa3-yl]-2-[[(2R,8S)-2- fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[azetidine-3,9'- fluorene]-3'-ol (compound 1). To a solution of tert-butyl (lR,5S)-3-[6-(3'- hydroxyspiro[azetidine-3,9'-fluorene]-l-yl)-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (Ih, 24.0 mg, 0.04 mmol) in 1,4-dioxane (1 mL) was added HC1 in 1,4-dioxane (1 mL, 4.0 mmol) at 0 °C and the reaction was stirred at rt for 2 h. Then the mixture was dried by N2 and then purified by prep-HPLC on a C18 column (5 um, 50 x 150 mm) with mobile phase: H2O (0.1% NH4HCO3 / MeOH) at flow rate: 35 mL/min to afford l-[6-[(lR,5S)-3,8- diazabicyclo[3.2.1]octa3-yl]-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methoxy]pyrimidin-4-yl]spiro[azetidine-3,9'-fluorene]-3'-ol (1, 12.3 mg, 0.0214 mmol, 59.7% yield) as a white solid. LCMS calcld for C33H38FN6O2 (M+H)+ m/z = 569.3; found: 569.0. JH NMR (400 MHz, CD3OD) 5 7.64 - 7.71 (m, 2 H), 7.51 (d, J= 8.0 Hz, 1 H), 7.33 - 7.40 (m, 2 H), 7.15 (d, J= 2.4 Hz, 1 H), 6.81 (dd, J= 8.0, 2.4 Hz, 1 H), 5.34 (s, 0.5 H), 5.27 (s, 1 H), 5.20 (s, 0.5 H), 4.36 (s, 4 H), 4.15 (d, J= 10.4 Hz, 1 H), 3.97 - 4.07 (m, 3 H), 3.59 (s, 2 H), 3.14 - 3.27 (m, 3 H), 3.00 - 3.06 (m, 2 H), 2.94 - 3.00 (m, 1 H), 2.15 - 2.34 (m, 2 H), 2.08 - 2.14 (m, 1 H), 1.89 - 2.00 (m, 2 H), 1.79 - 1.88 (m, 3 H), 1.72 - 1.79 (m, 2 H).
Example 2. Exemplary synthesis of 5-[6-(2-amino-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahy dropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3-chloro-N ,N -dimethyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (compound 2)
[00562] Step 1. Synthesis of 3-chloro-5-(6-chloro-5-cyano-2-methylsulfanyl-pyrimidin-4-yl)- N ,N -dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (2a). The mixture
of 3-chloro-7V,A-dimethyl-5,6,7,8-tetrahydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxamide (Intermediate 1, 1.7 g, 7 mmol), 4,6-dichloro-2-methylsulfanyl-pyrimidine-5-carbonitrile (1.7 g, 7.7 mmol) and DIEA (2.71 mg, 21.0 mmol) in DCM (3 mL)was stirred at 25 °C for 0.5 h under argon. The mixture was concentrated, diluted with DCM (50 mL), washed with water (50 x 2 mL) and brine (50 ml), dried over Na2SO4, concentrated. The crude product 3-chloro-5-(6- chloro-5-cyano-2-methylsulfanyl-pyrimidin-4-yl)-A,7V-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxamide (2a) was used without purification. LCMS calcld for CieHisChNvOS (M+H)+ m/z = 426.1, found: 426.3.
[00563] Step 2. Synthesis of (2b). The mixture of 3-chloro-5-(6-chloro-5-cyano-2- methylsulfanyl-pyrimidin-4-yl)-N ,N -dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine- 2-carboxamide (2a, 2.9 g, 6.8 mmol), oxone (6.272 g, 10.2 mmol) , THF (20 mL) and water (10 mL) was stirred at 45 °C for 2 h under argon. The mixture was diluted with DCM (100 mL), washed with H2O (2 x 100 mL) and brine (100 mL), dried over Na2SO4 and concentrated to afford crude product 3-chloro-5-(6-chloro-5-cyano-2-methylsulfonyl-pyrimidin-4-yl)-7V,A- dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (2b, 2.35 g, 5.13 mmol, 75.4% yield). LCMS calcld for C16H18CI2N7O3S (M+H)+ m/z = 458.1, found: 458.1.
[00564] Step 3. Synthesis of 3-chloro-5-[6-chloro-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-7V,A-dimethyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (2c). The mixture of 3-chloro-5-(6- chloro-5-cyano-2-methylsulfonyl-pyrimidin-4-yl)-7V,A-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxamide (2b, 1.9 g, 4.15 mmol) and [(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methanol (1.98 g, 12.44 mmol) in DCM (20 mL) was stirred at 25 °C for 1 h under argon. The mixture was concentrated to afford a crude product. The crude product was purified by silica gel chromatography (eluted with MeOH in DCM from 3% to 10%) to afford 3-chloro-5-[6-chloro-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methoxy]pyrimidin-4-yl]-7V,A-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2- carboxamide (2c, 1.2 g, 1.85 mmol, 44.7% yield) LCMS m/z calcld for C23H28CI2FN8O2 (M+H)+ m/z = 537.2, found: 537.2.
[00565] Step 4. Synthesis of 5-[6-(2-amino-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahy dropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3-chloro-A,A-dimethyl-4, 6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (compound 2). The mixture of 3-chloro- 5-[6-chloro-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizi8- yl]methoxy]pyrimidin-4-yl]-7V,A-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2- carboxamide (2c, 200 mg, 0.37 mmol), 2-aminospiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-
azetidine]-3-carbonitrile (Intermediate 2, 244 mg, 0.6 mmol), DIEA (192 g, 1.49 mmol) in 1,4- di oxane (5 mL) was stirred at 90 °C for 1.5 h under argon. The mixture was diluted with EtOAc (50 mL), washed with H2O (2 x 20 mL) and brine (50 mL), dried over ISfeSCU and concentrated. The crude product was purified by prep-HPLC (eluted with CH3CN in H2O from 5% to 95%) to afford 5-[6-(2-amino-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5- cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3- chloro-A,A-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide (compound 2), 89 mg, 0.12 mmol, 32% yield) as yellow solid. LCMS calcld for C33H38CIFN11O2S (M+H)+ m/z = 706.2, found: 706.2. *HNMR (400 MHz, CDCI3) 8 8.05 - 7.94 (s, 2H), 5.17 (d, J= 52.4 Hz, 1H), 4.93 (s, 1H), 4.79 (d, J= 21.4 Hz, 2H), 4.61 (s, 2H), 4.54 (s, 1H), 4.35 (s, 2H), 4.17 - 3.87 (m, 4H), 3.13 (d, J= 14.8 Hz, 2H), 3.03 (d, J= 5.7 Hz, 6H), 2.87 (t, .7= 7.2 Hz, 1H), 2.77 - 2.59 (m, 4H), 2.22 - 1.91 (m, 5H), 1.79 (ddd, J= 20.3, 19.1, 11.6 Hz, 4H).
[00566] Compound 3. 5-[6-(2-amino-3-cyano-spiro[6,7-dihydro-5H-benzothiophene-4,3'- azetidine]-l'-yl)-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methoxy]pyrimidin-4-yl]-3-chloro-A,A-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5- a] [ 1 ,4]diazepine-2-carboxamide
[00567] Compound 3 was prepared similarly to that of Ex. 2 as a TFA salt using Intermediate 3. LCMS calcld for C34H40CIFN11O2S (M+H)+ m/z = 720.27; found: 720.2. 1 H NMR (400 MHz, CD3OD) 5 5.55 (d, J= 52.0 Hz, 1H), 4.96 (s, 3H), 4.33 (m, 8H), 3.96 - 3.73 (m, 3H), 3.43 (m, 1H), 3.10 (m, 6H), 2.75 - 2.45 (m, 4H), 2.41 - 2.24 (m, 5H), 2.24 - 2.00 (m, 4H), 1.81 (s, 2H).
Example 3. Exemplary synthesis of2-amino-l'-[5-cyano-2-[[l- (morpholinomethyl)cyclopropyl]methoxy]-6-[-(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile (Compound 4).
[00568] Step 1. Preparation of tert-butyl (lR,5S)-3-(6-chloro-5-cyano-2-methylsulfanyl- pyrimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (4a). To a solution of tert-butyl (lR,5S)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (1.93 g, 9.09 mmol) and 4,6-dichloro-2- methylsulfanyl-pyrimidine-5-carbonitrile (2.0 g, 9.09 mmol) in DCM (20 mL) was added DIEA (4.75 mL, 27.26 mmol) at 0 °C. The mixture was stirred at 0 °C for 1 h. The mixture was diluted with EtOAc (50 mL), washed with H2O (2x30 mL) and brine (30 mL), dried over ISfeSCL and concentrated. The crude product was purified by silica gel chromatography (PE: EtOAc = 20: 1 to 5: 1). The product tert-butyl (lR,5S)-3-(6-chloro-5-cyano-2-methylsulfanyl-pyrimidin-4-yl)-3,8- diazabicyclo[3.2.1]octane-8-carboxylate (4a, 3.35 g, 8.46 mmol, 93.11% yield) was obtained as white solid. LCMS calcld for C17H23CIN5O2S (M+H)+ m/z = 396.1, found: 396.0.
[00569] Step 2. Preparation of tert-butyl (lR,5S)-3-(6-chloro-5-cyano-2-methylsulfonyl- pyrimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (4b). To a solution of tert-butyl (lR,5S)-3-(6-chloro-5-cyano-2-methylsulfanyl-pyrimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8- carboxylate (4a, 3.35 g, 8.46 mmol) in THF (35 mL)/Water (35 mL) was added oxone (5.2 g, 16.92 mmol) at 25°C. The mixture was stirred at 25°C for 3 h. The mixture was diluted with EtOAc (40 mL), washed with H2O (2x30 mL) and brine (30 mL), dried over Na2SO4 and concentrated. The crude product was used without purification. The product tert-butyl (lR,5S)-3- (6-chloro-5-cyano-2-methylsulfonyl-pyrimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8- carboxylate (4b, 3.40 g, 7.95 mmol, 93.90 % yield) was obtained as crude white solid. LCMS calcld for C17H23CIN5O4S (M+H)+ m/z = 428.1, found: 428.2.
[00570] Step 3. Preparation of tert-butyl (lR,5S)-3-[5-cyano-2-methylsulfonyl-6- [[l(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]-3,8-diazabicyclo[3.2.1]octane-8- carboxylate (4c). To a solution of tert-butyl (lR,5S)-3-(6-chloro-5-cyano-2-methylsulfonyl- pyrimidin-4-yl)-3,8-diazabicyclo[3.2.1]octane-8-carboxylate (4b, 3.4 g, 7.95 mmol) in DCM (34 mL) was added [l-(morpholinomethyl)cyclopropyl]methanol (2.72 g, 15.89 mmol) at 25°C. The mixture was stirred at 25°C for 3 h. The mixture was concentrated to afford a crude product. The
crude product was purified by silica gel chromatography (PE: EtOAc = 10: 1 to 1 : 1). The product tert-butyl (lR,5S)-3-[6-chloro-5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]-3,8-diazabicyclo[3.2.1]octane-8- carboxylate (4c, 2.10 g, 4.05 mmol, 50.92% yield) was obtained as yellow oil. LCMS calcld for C25H36CIN6O4 (M+H)+ m/z = 519.2, found: 519.2.
[00571] Step 4. Preparation of tert-butyl (lR,5S)-3-[6-(2-amino-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2-[[l- (morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]-3,8-diazabicyclo[3.2.1]octane-8- carboxylate (4d). To a solution of 2-aminospiro[5,6-dihydrocyclopenta[b]thiophene-4,3'- azetidine]-3-carbonitrile (Intermediate 2, 1.0 g, 4.88 mmol) and tert-butyl (lR,5S)-3-[6-chloro- 5-cyano-2-[[l-(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]-3,8- diazabicyclo[3.2.1]octane-8-carboxylate (4c, 1.95 g, 3.76 mmol) in 1,4-Dioxane (20 mL) was added DIEA (3.27 mL, 18.78 mmol) at 25°C. The mixture was stirred at 90°C for 2 h. The mixture was concentrated to afford a crude product. The crude product was purified by silica gel chromatography (PE: EtOAc = 1:1 to 1:5). The product tert-butyl (lR,5S)-3-[6-(2-amino-3- cyano-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2-[[l- (morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]-3,8-diazabicyclo[3.2.1]octane-8- carboxylate (4d, 1.80 g, 2.62 mmol, 69.65 % yield) was obtained as yellow oil. LCMS calcld for C35H46N9O4S (M+H)+ m/z = 688.3, found: 688.2.
[00572] Step 5. Preparation of 2-amino-l'-[5-cyano-2-[[l- (morpholinomethyl)cyclopropyl]methoxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile (4e). To a solution of tert-butyl (lR,5S)-3-[6-(2-amino-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2-[[l- (morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]-3,8-diazabicyclo[3.2.1]octane-8- carboxylate (4d, 1.8 g, 2.62 mmol) and TFA (6.0 mL, 78.35 mmol) in THF (18 mL) at 25°C. The mixture was stirred at 25°C for 1 h. The mixture was concentrated to afford a crude product. The crude product was purified by Prep-HPLC (eluted with CH3CN in H2O (0.1 % NH3) from 5.0% to 95%). 2-amino-l'-[5-cyano-2-[[l-(morpholinomethyl)cyclopropyl]methoxy]-6-[(lR,5S)- 3,8-diazabicyclo[3.2.1]octan-3-yl]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'- azetidine]-3-carbonitrile (Compound 4, 544.5 mg, 0.92 mmol, 35.16% yield) was obtained as white solid. LCMS calcld for C30H38N9O2S (M+H)+ m/z = 588.3, found: 588.3. *HNMR (400 MHz, CD3OD) 5 4.68 - 4.13 (m, 8H), 3.68 (t, J= 4.6 Hz, 4H), 3.56 (s, 2H), 3.32 - 3.20 (m, 2H), 2.86 - 2.65 (m, 4H), 2.49 (s, 4H), 2.39 (s, 2H), 1.82 (s, 4H), 0.66 (t, J= 5.2 Hz, 2H), 0.46 (t, J= 5.2 Hz, 2H)
[00573] Compound 5. 5-[6-(2-amino-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene-
4,3'-azetidine]-r-yl)-5-cyano-2-[[l-(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]-3- chloro-N,N-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide
[00574] Compound 5 was prepared similarly to that of Ex. 2. LCMS calcld for C34H41CIN11O3S (M+H)+ m/z =718.3 found 718.2. *HNMR (400 MHz, CD3OD) 5 5.08-5.01 (m, 2H), 4.62-4.40 (m, 5H), 4.29-4.14 (m, 5H), 4.81-471 (m, 4H), 3.15-3.03 (m, 6H), 2.88-2.64 (m, 10H), 2.31-2.21 (m, 2H), 0.76-0.53 (m, 4H).
Example 4. Exemplary synthesis of 2-amino-l'-[5-fluoro-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-[(3R)-3-hydroxy-3-methyl-l-piperidyl]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile (Compound 6).
[00575] Step 1. Synthesis of (3R)-l-(6-chloro-5-fluoro-2-methylsulfanyl-pyrimidin-4-yl)-3- methyl-piperi din-3 -ol (6a). A solution of 4,6-dichloro-5-fluoro-2-methylsulfanyl-pyrimidine (200 mg, 0.94 mmol) in DCM (8mL) was added (3R)-3-methylpiperi din-3 -ol (108.11 mg, 0.94 mmol) and DIEA (0.33 mL, 1.88 mmol) at 0 °C. Then the mixture was stirred at 0 °C for 1 h. The solution was extracted with EtOAc (20 ml x3), dried over ISfeSC and concentrated. The residue was purified by silica gel chromatography (DCM:MeOH=30: l). The product of (3R)-1- (6-chloro-5-fluoro-2-methylsulfanyl-pyrimidin-4-yl)-3-methyl-piperidin-3-ol (210 mg, 0.720
mmol, 76.67% yield) was obtained. LCMS calculated for C11H16CIFN3OS (M+H)+ m/z =292.3, found: 292.3.
[00576] Step 2. Synthesis of (3R)-l-(6-chloro-5-fluoro-2-methylsulfmyl-pyrimidin-4-yl)-3- methyl-piperi din-3 -ol (6b). A solution of (3R)-l-(6-chloro-5-fluoro-2-methylsulfanyl-pyrimidin- 4-yl)-3-methyl-piperi din-3 -ol (6a, 170 mg, 0.58 mmol) in THF (5mL) and Water (2.5mL) was added oxone (302.4 mg, 0.87 mmol) at 25 °C. Then the mixture was stirred at 25 °C for 1 h. The solution was extracted with EtOAc (20 ml x3), dried over Na2SO4 and concentrated. The residue was purified by silica gel chromatography (eluting with MeOH in DCM from 3% to 6%). The product (3R)-l-(6-chloro-5-fluoro-2-methylsulfmyl-pyrimidin-4-yl)-3-methyl-piperidin-3-ol (180 mg, 0.585 mmol, quantitative yield) was obtained as a white solid. LCMS calculated for C11H16CIFN3O2S (M+H)+ m/z =308.0, found: 308.0.
[00577] Step 3. Synthesis of 2-amino-l'-[5-fluoro-2-methylsulfmyl-6-[(3R)-3-hydroxy-3- methyl-l-piperidyl]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3- carbonitrile (6c). The mixture of DIEA (0.12 mL, 0.71 mmol) 2-aminospiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile (Intermediate 2, 146.74 mg, 0.71 mmol) and (3R)-l-(6-chloro-5-fluoro-2-methylsulfmyl-pyrimidin-4-yl)-3-methyl-piperidin-3-ol (6b, 110 mg, 0.36 mmol) in 1,4-Dioxane (1 mL) was stirred at 90 °C for 2 h under argon. The mixture was filtered to afford a crude solution. The crude product was purified by flash chromatography (eluted with CH3CN in H2O from 5.0% to 95%). The product 2-amino-l'-[5- fluoro-2-methylsulfmyl-6-[(3R)-3-hydroxy-3-methyl-l-piperidyl]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile (80 mg, 0.168 mmol, 46.97% yield) was obtained as a white solid. LCMS calcld for C21H26FN6O2S2 (M+H)+ m/z =477.2, found: 477.2.
[00578] Step 4. Synthesis of tert-butyl N-tert-butoxycarbonyl-N-[3-cyano-l'-[5-fluoro-2- methylsulfinyl-6-[(3S)-3-hydroxy-3-methyl-l-piperidyl]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2-yl]carbamate (6d). The mixture of 2-amino-l'- [5-fluoro-2-methylsulfmyl-6-[(3S)-3-hydroxy-3-methyl-l-piperidyl]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile (6c, 70 mg, 0.15 mmol) BOC2O (70.52 mg, 0.32 mmol), DMAP (1.79 mg, 0.01 mmol) in CH3CN (0.5mL) was stirred at 25 °C for 0.2 h under argon. The mixture was concentrated to afford a crude product. The crude product was purified by flash chromatography EhCFCEECN = 90: 10 to 50:50. tert-butyl N-[3- cyano-l'-[5-fluoro-2-methylsulfmyl-6-[(3S)-3-hydroxy-3-methyl-l-piperidyl]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2-yl]carbamate (80 mg, 0.139 mmol, 94.45% yield)) was obtained as yellow oil. LCMS calcld for C26H34N6O4S2 (M+H)+ m/z = 577.2, found: 577.2.
[00579] Step 5. Synthesis of tert-butyl N-[3-cyano-l'-[5-fluoro-2-[[(2R,8S)-2-fluoro- 1,2, 3,5,6, 7-hexahy dropyrrolizin-8-yl]methoxy]-6-[(3R)-3-hy droxy-3 -methyl- 1 - piperidyl]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2- yl]carbamate (6e). The mixture of [(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methanol (44.17 mg, 0.28 mmol) and NaH (8.32 mg, 0.21 mmol) in DMF (0.5mL) was stirred at 25 °C for 0.2h under argon. Then, tert-butyl N-[3-cyano-l'-[5-fluoro-6-[(3R)-3-hydroxy-3- methyl-l-piperidyl]-2-methylsulfinyl-pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3'-azetidine]-2-yl]carbamate (6d, 40 mg, 0.07 mmol) was added at 25 °C . The mixture was stirred at 25 °C for 0.2 h. The mixture was acidified with Acetic acid (Imol/L in H2O) to pH=5, extracted with EtOAc (3 x lOmL), dried over ISfeSCU and concentrated. The crude product was used directly for the next step, tert-butyl N-[3-cyano-l'-[5-fluoro-2-[[(2R,8S)-2-fluoro- 1,2, 3, 5, 6, 7-hexahy dropyrrolizin-8-yl]methoxy]-6-[(3R)-3-hy droxy-3 -methyl- 1- piperidyl]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2- yl]carbamate (6e, 40 mg, 0.060 mmol) was obtained as yellow oil. LCMS calcld for C33H44F2N7O4 (M+H)+ m/z =672.2, found: 672.4.
[00580] Step 6. Synthesis of 2-amino-T-[5-fhioro-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahy dropyrrolizin-8-yl]methoxy]-6-[(3R)-3-hy droxy-3 -methyl- 1-piperi dyl]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile (Compound 6) . The mixture of tert-butyl N-[3-cyano-l'-[5-fluoro-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahy dropyrrolizin-8-yl]methoxy]-6-[(3R)-3-hy droxy-3 -methyl- 1-piperi dyl]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2-yl]carbamate (6e, 30 mg, 0.04 mmol) and TFA (0.1 mL, 1.31 mmol) in DCM (ImL) was stirred at 25 °C for 0.2h under argon. The mixture was stirred at 25 °C for 0.2 h. The mixture was concentrated to afford a crude product.
[00581] The crude product was purified by prep-HPLC (eluted with CH3CN in H2O (0.1 % TFA) from 5.0% to 95%). 2-amino-l'-[5-fhioro-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahy dropyrrolizin-8-yl]methoxy]-6-[(3R)-3-hy droxy-3 -methyl- 1-piperi dyl]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile (Compound 6, 8.16 mg, 0.0089 mmol, 19.83% yield). LCMS calcld for C28H36F2N7OS (M+H)+ m/z =572.3, found: 572.2. 1H NMR (400 MHz, CD3OD) 5 5.64 - 5.44 (m, 1H), 4.51 - 4.41 (m, 2H), 4.41 - 4.36 (m, 1H), 4.28 - 4.16 (m, 2H), 4.06 - 3.71 (m, 4H), 3.69 - 3.60 (m, 1H), 3.52 - 3.35 (m, 4H), 3.35 - 3.33 (m, 2H), 2.82 - 2.66 (m, 4H), 2.63 - 2.44 (m, 1H), 2.40 - 2.03 (m, 4H), 1.92 - 1.78 (m, 1H), 1.73 - 1.62 (m, 2H), 1.62 - 1.46 (m, 1H), 1.20 (s, 3H).
Compound 7. 2-amino-l'-[6-[(5R)-2,4-dioxo-l,3,9-triazaspiro[4.5]decan-9-yl]-5-fluoro-2-
[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00582] Compound 7 was prepared similarly to that of Ex. 4 as a TFA salt. LCMS calcld for C29H34F2N9O3S (M+H)+ m/z = 626.2, found: 626.3. *HNMR (400 MHz, CD3OD) 5 5.56 - 5.38 (m, 1H), 4.48 - 4.15 (m, 6H), 3.87 - 3.62 (m, 2H), 3.40 - 3.33 (m, 2H), 3.26 - 3.17 (m, 1H), 2.82 - 2.67 (m, 3H), 2.60 - 2.36 (m, 2H), 2.33 - 1.98 (m, 9H), 1.90 - 1.75 (m, 3H), 1.70 - 1.56 (m, 1H).
Compound 8. 5-[6-(2-amino-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]- l'-yl)-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4- yl]-N,N-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide
[00583] Compound 8 was prepared similarly to that of Ex. 2 as a formate salt. LCMS calcld for C33H39FNnO2S(M+H)+ m/z =672.3, found: 672.5. 'H NMR (400 MHz, CD3OD) 5 6.67 (s, 1H), 5.29 (d, J= 53.1 Hz, 1H), 5.07 (s, 2H), 4.52 - 4.46 (m, 5H), 4.22 - 4.07 (m, 5H), 3.26 - 3.23 (m, 6H), 3.17 - 2.97 (m, 4H), 2.77 - 2.70 (m, 4H), 2.28 - 1.86 (m, 8H).
Compound 9. 5-[6-(2-amino-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]- r-yl)-5-fhioro-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4- yl]-3-chloro-N,N-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide
[00584] Compound 9 was prepared similarly to that of Ex. 4 as a TFA salt. LCMS calcld for
C32H38CIF2N10O2S (M+H)+ m/z =699.2, found: 699.3. *HNMR (400 MHz, CD3OD) 5 5.54 (d, J
= 52.0 Hz, 1H), 4.92 - 4.88 (m, 1H), 4.83 - 4.75 (m, 1H), 4.55 - 4.40 (m, 4H), 4.38 (s, J= 6.1 Hz, 2H), 4.30 - 4.18 (m, 2H), 4.14 - 3.73 (m, 5H), 3.41 (dd, J= 16.2, 11.2 Hz, 1H), 3.09 (d, J= 6.8 Hz, 6H), 2.83 - 2.69 (m, 4H), 2.69 - 2.42 (m, 2H), 2.31 (td, J= 9.9, 4.1 Hz, 3H), 2.19 - 1.99 (m, 3H).
Compound 10. 5-[6-(2-amino-3-cyano-spiro[5,6,7,8-tetrahydrocyclohepta[b]thiophene-4,3'- azetidine]-l'-yl)-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methoxy]pyrimidin-4-yl]-3-chloro-N,N-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5- a] [ 1 ,4]diazepine-2-carboxamide
[00585] Compound 10 was prepared similarly to that of Ex. 2. LCMS calcld for C35H42CIFN11O2S (M+H)+ m/z =734.28; found: 734.3. 'H NMR (CDCI3, 500 MHz) 5 7.22 - 7.29 (m, 5H), 7.18 (d, J= 7.2 Hz, 1H), 5.39 - 5.52 (m, 1H), 5.23 - 5.39 (m, 1H), 4.67 - 5.09 (m, 4H), 4.42 (br. s., 6H), 3.98 - 4.22 (m, 3H), 3.50 - 3.83 (m, 2H), 3.20 - 3.48 (m, 1H), 2.94 - 3.18 (m, 6H), 2.49 - 2.66 (m, 3H), 1.30 - 2.40 (m, 31H), 0.65 - 0.92 (m, 3H).
[00586] Compound 11. 2-amino-l'-[5-cyano-6-[(5R)-2,4-dioxo-l,3,9- triazaspiro[4.5]decan-9-yl]-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3- carbonitrile
[00587] Compound 11 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calcld for C30H34FN10O3S (M+H)+ m/z = 633.2, found: 633.2. 'HNMR (400 MHz, CD3OD) 5 5.71 - 5.42 (m, 1H), 4.84 - 4.76 (m, 1H), 4.64 - 4.27 (m, 7H), 4.05 - 3.71 (m, 3H), 3.60 - 3.36 (m, 3H), 2.85 - 2.44 (m, 6H), 2.39 - 2.06 (m, 5H), 2.02 - 1.71 (m, 3H).
[00588] Compound 12. 2-amino-l'-[5-cyano-6-(2,4-dioxo-l,3,9-triazaspiro[4.5]decan-9- yl)-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00589] Compound 12 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calcld for C30H34FN10O3S (M+H)+ m/z = 633.2, found: 633.4. ‘HNMR (400 MHz, CD3OD) 5 5.65 - 5.39 (m, 1H), 4.65 - 4.25 (m, 8H), 4.03 - 3.75 (m, 3H), 3.60 - 3.37 (m, 3H), 2.89 - 2.47 (m, 6H), 2.39 - 2.22 (m, 3H), 2.21 - 2.08 (m, 2H), 2.02 - 1.73 (m, 3H).
[00590] Compound 13. l'-[6-(2-acetyl-3-chloro-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepin-5-yl)-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methoxy]pyrimidin-4-yl]-2-amino-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3- carbonitrile
[00591] Compound 13 was prepared similarly to that of Ex. 2 using Intermediate 5. LCMS calcld for C32H35CIFN10O2S (M+H)+ m/z = 677.2, found: 677.2. 'H NMR (DMSO-d6, 400 MHz)
5 7.22 (s, 2H), 5.29 (d, J= 52.8 Hz, 1H), 5.03 (s, 2H), 4.51 (s, 2H), 4.46-4.25 (m, 4H), 4.11 (s, 2H), 3.98 (d, J= 10.4 Hz, 1H), 3.90 (d, J= 10.4 Hz, 1H), 3.02 (br, 2H), 2.96-2.94 (m, 1H), 2.82- 2.76 (m, 1H), 2.67-2.65 (m, 4H), 2.44 (s, 3H), 2.15 (br, 2H), 2.03 (s, 1H), 1.96 (s, 1H), 1.89-1.85 (m, 1H), 1.83-1.79 (m, 1H), 1.78-1.67 (m, 2H).
[00592] Compound 14. 5-[6-(2-amino-3-cyano-6-methyl-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahy dropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3-chloro-N,N-dimethyl-4, 6,7,8- tetrahydropyrazolof 1 ,5-a] [ 1 ,4]diazepine-2-carboxamide
[00593] Compound 14 was prepared similarly to that of Ex. 2 as a formate salt, using Intermediate 6. LCMS calcld for C34H40CIFN11O2S (M+H)+ m/z =720.3 , found: 720.2. 'H NMR (400 MHz, CD3OD) 5 8.66 - 8.46 (s, 1H), 5.38 - 5.19 (m, 1H), 5.10 - 5.01 (m, 2H), 4.62 - 4.41 (m, 4H), 4.25 - 4.07 (m, 4H), 3.27 - 3.11 (m, 4H), 3.09 (d, J= 4.1 Hz, 6H), 3.06 - 2.97 (m, 2H), 2.97 - 2.88 (m, 1H), 2.41 - 1.69 (m, 10H), 1.17 (d, J= 6.8 Hz, 3H).
[00594] Compound 15. 2-amino-l'-[6-(3,8-diazabicyclo[3.2.1]octan-3-yl)-5-fluoro-2- [[(2R,8S)-2-fhioro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00595] Compound 15 was prepared similarly to that of Ex. 4 as a TFA salt. LCMS calcld for C28H35F2N8OS (M+H)+ m/z = 569.3, found: 569.3. *HNMR (400 MHz, CD3OD) 5 5.51 - 5.29 (m, 1H), 4.51 - 4.41 (m, 2H), 4.30 - 4.12 (m, 6H), 4.08 - 3.99 (m, 2H), 3.66 - 3.46 (m, 3H), 3.38 - 3.32 (m, 2H), 3.27 - 3.15 (m, 1H), 2.83 - 2.63 (m, 4H), 2.53 - 1.91 (m, 10H).
[00596] Compound 16. 2-amino-l'-[5-cyano-6-[2-(3-methyl-l,2,4-oxadiazol-5-yl)-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-5-yl]-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-
hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-
[00597] Compound 16 was prepared similarly to that of Ex. 2 as a formate salt using Intermediate 7. LCMS calcld for C33H36FN12O2S (M+H)+ m/z =683.3, found: 683.3. *H NMR (400 MHz, CD3OD) 5 7.03 (s, 1H), 5.39-5.07 (m, 3H), 4.65-4.02 (m, 10H), 3.29-3.09 (m, 3H), 3.08-2.97 (m, 1H), 2.78-2.68 (m, 4H), 2.40 (s, 3H), 2.28-1.80 (m, 8H).
[00598] Compound 17. 2-amino-l'-[6-[3-chloro-2-(3-methyl-l,2,4-oxadiazol-5-yl)-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3 '-azetidine]-3 -carbonitrile.
[00599] Compound 17 was prepared similarly to Compound 16 as a formate salt. LCMS calcld for C33H35CIFN12O2S (M+H)+ m/z =717.2, found: 717.1. *HNMR (400 MHz, CD3OD) 5 5.46-5.26 (m, 1H), 5.16-4.97 (m, 2H), 4.59-4.14 (m, 10H), 3.55-3.36 (m, 3H), 3.20-3.08 (m, 1H), 2.82-2.66 (m, 4H), 2.44 (s, 3H), 2.40-2.00 (m, 7H), 1.95-1.85 (m, 1H).
[00600] Compound 18. 5-[6-(2-amino-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3'-azetidine]-r-yl)-5-cyano-2-(l,2,3,5,6,7-hexahydropyrrolizin-8-ylmethoxy)pyrimidin-4-yl]- 3-chloro-N,N-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide
[00601] Compound 18 was prepared similarly to that of Ex. 2 as a formate salt. LCMS calcld for C33H39CIN11O2S (M+H)+ m/z = 688.3; found : 688.2. *H NMR (400 MHz, CD3OD) 5 5.01 (s, 2H), 4.62 - 4.47 (m, 2H), 4.47 - 4.40 (m, 2H), 4.35 (s, 2H), 4.21 (dd, J = 3.5, 2.9 Hz, 2H), 3.53 -
3.44 (m, 2H), 3.37 - 3.33 (m, 2H), 3.15 - 3.10 (m, 2H), 3.09 (dd, J = 5.5, 2.1 Hz, 6H), 2.82 - 2.65 (m, 4H), 2.37 - 1.85 (m, 10H).
[00602] Compound 19. 2-amino-l'-[6-(azepan-l-yl)-5-cyano-2-[[(2R)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-
[00603] Compound 19 was prepared similarly to that of Ex. 2 as a formate salt. LCMS calcld for C29H36FN8OS (M+H)+ m/z = 563.3; found :563.4. 'H NMR (400 MHz, CD3OD) 5 5.27 (d, J = 33.1, 23.2 Hz, 1H), 4.69 - 4.45 (m, 2H), 4.51 - 4.26 (m, 2H), 4.14 (dd, J= 46.1, 10.8 Hz, 2H), 3.92 - 3.75 (m, 4H), 3.30 - 3.19 (m, 3H), 3.10 - 2.97 (m, 1H), 2.84 - 2.62 (m, 4H), 2.41 - 1.92 (m, 5H), 1.86 (d, J= 5.3 Hz, 5H), 1.67 - 1.50 (m, 4H).
Example 5. Exemplary synthesis of 2-amino-l'-[6-[3-chloro-2-[(E)-N-methoxy-C-methyl- carbonimidoyl]-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-[[(2R,8S)-2- fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile (Compound 20)
[00604] Preparation of 2-amino-l'-[6-[3-chloro-2-[(E)-N-methoxy-C-methyl-carbonimidoyl]- 4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3 '-azetidine]-3 -carbonitrile (Compound 20). To a solution of Methoxyammonium chloride (1.23 mg, 0.01 mmol) and l'-[6-(2-acetyl-3-chloro-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepin-5-yl)-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methoxy]pyrimidin-4-yl]-2-amino-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3- carbonitrile (Compound 13, 5 mg, 0.01 mmol) in Ethanol (1 mL) AcONa (1.21 mg, 0.01 mmol) was added. The mixture was stirred at rt for 3h. The mixture was diluted with EtOAC (5 mL), washed with water (5 mL), dried over Na2SO4, concentrated. The crude product was purified by Prep-HPLC (eluted with CH3CN in H2O (0.1 % NH4HCO3) from 5.0% to 95%). 2-amino-l'-[6- [3-chloro-2-[(E)-N-methoxy-C-methyl-carbonimidoyl]-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepin-5-yl]-5-cyano-2-[[(2R,8S)-2-fhioro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3- carbonitrile (Compound 20, 3.12 mg, 0.0038 mmol, 51.09% yield) was obtained as white solid. LCMS calcld for C33H38CIFN11O2S (M+H)+ m/z = 706.3, found: 706.1. XH NMR (DMSO-d6, 400 MHz) 5 7.22 (s, 2H), 5.24 (d, J= 56.0 Hz, 1H), 5.00 (br, 2H), 4.43-4.41 (m, 2H), 4.37-4.17 (m, 2H), 4.09 (m, 2H), 4.00 (m, 1H), 3.91 (m, 1H), 3.88 (s, 3H), 3.04 (br, 2H), 2.95 (br, 1H), 2.82-2.76 (m, 1H), 2.68-2.63 (m, 5H), 2.15-2.12 (m, 1H), 2.11 (s, 3H), 2.05-2.03 (m, 1H), 2.01- 1.96 (m, 3H), 1.90-1.87 (m, 1H), 1.82-1.78 (m, 1H), 1.74-1.70 (m, 2H).
[00605] Compound 21. 2-amino-l'-[5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-(2-methylsulfonyl-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepin-5-yl)pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3- carbonitrile
[00606] Compound 21 was prepared similarly to that of Ex. 2 using Intermediate 8. LCMS calcld for C31H36FN10O3S2 (M+H)+ m/z = 679.2, found: 679.1. 'HNMR (400 MHz, CD3OD) 5 6.87 (s, 1H), 5.36 - 5.15 (m, 1H), 5.15 - 4.97 (m, 2H), 4.64 - 4.42 (m, 6H), 4.29 - 4.21 (m, 2H), 4.13 - 4.01 (m, 2H), 3.21 (m, 2H), 3.16 (s, 3H), 3.15 (s, 2H), 2.75 (dd, J = 9.1, 5.2 Hz, 4H), 2.23 - 2.11 (m, 4H), 2.10 - 1.77 (m, 4H).
[00607] Compound 22. 2-amino-l'-[6-(3,8-diazabicyclo[3.2.1]octan-3-yl)-5-fluoro-2-[[l- (morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00608] Compound 22 was prepared similarly to that of Ex. 4 as a TFA salt. LCMS calcld for C29H38FN8O2S (M+H)+ m/z = 581.3, found: 581.4. ‘HNMR (400 MHz, CD3OD) 5 4.49 - 4.41 (m, 2H), 4.29 - 4.22 (m, 4H), 4.19 (d, J= 12.4 Hz, 2H), 4.14 - 4.07 (m, 2H), 3.91 - 3.78 (m, 4H), 3.39 - 3.33 (m, 2H), 3.18 - 2.89 (m, 6H), 2.82 - 2.70 (m, 4H), 2.15 - 2.05 (m, 4H), 0.80 (s, 2H), 0.69 (s, 2H).
[00609] Compound 23. 4-(2-aminospiro[5,6-dihydrocyclopenta[d]thiazole-4,3'-azetidine]-l'- yl)-2-[[l-(morpholinomethyl)cyclopropyl]methoxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]pyrimidine-5-carbonitrile
[00610] Compound 23 was prepared similarly to that of Ex. 3 using Intermediate 9. LCMS calcld for C28H38N9O2S (M+H)+ m/z =564.3, found: 564.2. 'H NMR (400 MHz, CD3OD) 5 4.45
(s, 6H), 4.24 (s, 2H), 3.98 (s, 2H), 3.67 (t, J= 4.5 Hz, 4H), 3.42 (d, J= 13.9 Hz, 2H), 2.89 - 2.69 (m, 4H), 2.51 (s, 4H), 2.41 (s, 2H), 2.01 (s, 4H), 0.64 (t, J= 5.2 Hz, 2H), 0.47 (t, J= 5.2 Hz, 1H). [00611] Compound 24. 2 -amino-l'-[5-cyano-6-(2-cyano-4,6,7,8-tetrahydropyrazolo[l,5- a][l,4]diazepin-5-yl)-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3- carbonitrile
[00612] Compound 24 was prepared similarly to that of Ex. 2 using Intermediate 10. LCMS calcld for C31H33FN11OS (M+H)+ m/z = 626.3, found: 626.2. 'H NMR (400 MHz, CD3OD) 5 6.87 (s, 1H), 5.26 (d, J= 53.8 Hz, 1H), 5.05 (s, 2H), 4.55 (m, 5H), 4.22 (s, 2H), 4.06 (m, 2H), 3.23 (s, 2H), 3.15 (m, 1H), 2.99 (s, 1H), 2.83 - 2.64 (m, 4H), 2.25 - 1.78 (m, 9H).
[00613] Compound 25. 2-amino-l'-[5-cyano-6-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2- [[(2R,8S)-2-fhioro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00614] Compound 25 was prepared similarly to that of Ex. 3 as a formate salt. LCMS calcld for C29H35FN9OS (M+H)+ m/z = 576.3, found: 576.3. *HNMR (400 MHz, CD3OD) 5 8.43 (s, 1H), 5.55 - 5.29 (m, 1H), 4.77 - 4.18 (m, 8H), 4.12 - 4.01 (m, 2H), 3.76 - 3.39 (m, 5H), 3.29 - 3.16 (m, 1H), 2.82 - 2.70 (m, 4H), 2.55 - 1.96 (m, 10H).
[00615] Compound 26. 2-amino-l'-[5-cyano-6-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-[3- (dimethylamino)-2,2-difluoro-propoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3 '-azetidine]-3 -carbonitrile
[00616] Compound 26 was prepared similarly to that of Ex. 3 as a formate salt. LCMS calcld for C26H32F2N9OS (M+H)+ m/z = 556.2, found: 556.3. ‘H NMR (400 MHz, CD3OD) 5 8.40 (s, 1H), 4.75-4.20 (m, 8H), 4.11 (s, 2H), 3.54-3.44 (m, 2H), 2.95-2.68 (m, 5H), 2.35 (s, 6H), 2.07 (s, 4H).
[00617] Compound 27. 2-amino-l'-[5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-[(3R)-3-hydroxy-3-methyl-l-piperidyl]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00618] Compound 27 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS m/z calcd for C29H36FN8O2S (M+H)+ m/z = 579.3, found 579.2. 'HNMR (400 MHz, CD3OD) 5 5.63 - 5.38 (m, 1H), 4.93 - 4.87 (m, 1H), 4.67 - 4.32 (m, 5H), 4.12 - 3.79 (m, 5H), 3.60 - 3.37 (m, 3H), 2.86 - 2.70 (m, 4H), 2.69 - 2.46 (m, 2H), 2.41 - 2.23 (m, 3H), 2.21 - 2.06 (m, 1H), 2.00 - 1.85 (m, 1H), 1.81 - 1.55 (m, 3H), 1.21 (s, 3H).
[00619] Compound 28. 2-amino-l'-[5-cyano-6-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-[[l-[(4- fluoro-l-piperidyl)methyl]cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00620] Compound 28 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C31H39FN9OS (M+H)+ m/z = 604.3, found:604.2. ‘H NMR (400 MHz, CD3OD) 5 5.00 (d, J=
41.6 Hz, 1H), 4.82 - 4.33 (m, 6H), 4.28 (s, 2H), 4.16 (s, 2H), 3.79 - 3.61 (m, 2H), 3.56 - 3.44
(m, 2H), 3.29 - 3.22 (m, 2H), 2.86 - 2.65 (m, 4H), 2.42 - 2.13 (m, 4H), 2.14 - 1.94 (m, 6H), 0.90 (d, J= 3.5 Hz, 2H), 0.86 - 0.78 (m, 2H).
[00621] Compound 29. 2-amino-l'-[5-cyano-6-[2-(l-methylbenzimidazol-2-yl)-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-5-yl]-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-
[00622] Compound 29 was prepared similarly to that of Ex. 2 as a formate salt using
Intermediate 11. LCMS calcld for C38H40FN12OS (M+H)+ m/z = 731.3, found: 731.4. *H NMR (400 MHz, CD3OD) 5 7.66 (d, J= 7.6 Hz, 1H), 7.55 (d, J= 7.6 Hz, 1H), 7.42 - 7.26 (m, 2H), 7.05 (s, 1H), 5.43 - 5.25 (m, 1H), 5.24 - 5.13 (m, 2H), 4.76 - 4.38 (m, 5H), 4.34 - 3.99 (m, 8H), 3.32 - 3.19 (m, 3H), 3.10 - 2.98 (m, 1H), 2.82 - 2.68 (m, 4H), 2.40 - 1.83 (m, 8H).
[00623] Compound 30. 2-amino-l'-[5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-(2,3,6,7-tetrahydroazepin-l-yl)pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00624] Compound 30 was prepared similarly to Compound 29 as a formate salt. LCMS calcld for C38H39FN12OS (M+H)+ m/z = 765.3, found: 765.5. 'H NMR (400 MHz, CD3OD) 5 8.47 (s, 2H), 5.80 - 5.71 (m, 2H), 5.55 - 5.37 (m, 1H), 4.72 - 4.21 (m, 8H), 4.02 - 3.93 (m, 2H), 3.76 - 3.58 (m, 3H), 3.27 - 3.18 (m, 1H), 2.84 - 2.71 (m, 4H), 2.57 - 2.00 (m, 10H).
[00625] Compound 31. 2-amino-l'-[5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-(2,3,6,7-tetrahydroazepin-l-yl)pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00626] Compound 31 was prepared similarly to that of Ex. 2 as a formate salt. LCMS calcld for C29H34FN8OS (M+H)+ m/z = 561.3, found: 561.2. *HNMR (400 MHz, CD3OD) 5 8.47 (s, 2H), 5.80 - 5.71 (m, 2H), 5.55 - 5.37 (m, 1H), 4.72 - 4.21 (m, 8H), 4.02 - 3.93 (m, 2H), 3.76 - 3.58 (m, 3H), 3.27 - 3.18 (m, 1H), 2.84 - 2.71 (m, 4H), 2.57 - 2.00 (m, 10H).
[00627] Compound 32. 2-amino-l'-[6-[3-chloro-2-(4-fluoro-l-methyl-benzimidazol-2-yl)- 4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3 '-azetidine]-3 -carbonitrile
[00628] Compound 32 was prepared similarly Compound 29 as a formate salt. LCMS calcld for C38H38CIF2N12OS (M+H)+ m/z = 783.3, found:783.3. 'H NMR (400 MHz, CD3OD) 5 7.46- 7.29 (m, 2H), 7.11-7.01 (m, 1H), 5.67-5.47 (m, 1H), 5.18-4.90 (m, 3H), 4.61-4.22 (m, 9H), 4.00- 3.76 (m, 6H), 3.50-3.36 (m, 1H), 2.83-2.47 (m, 6H), 2.43-2.10 (m, 6H).
[00629] Compound 33. 2-amino-l'-[5-cyano-6-(4-cyanoazepan-l-yl)-2-[[(2R,8S)-2-fluoro- l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00630] Compound 33 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calcld for C30H35FN9OS (M+H)+ m/z =588.3, found: 588.3. *H NMR (400 MHz, CD3OD) 5: 5.55 (d, 7=51.6 Hz, 1H), 4.55-4.36 (m, 6H), 4.02-3.81 (m, 7H), 3.48-3.40 (m, 1H), 3.08-3.04 (m, 1H), 2.80-2.70 (m, 4H), 2.68-2.49 (m, 2H), 2.37- 1.91 (m, 10H).
[00631] Compound 34. 2-amino-l'-[5-cyano-6-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-[[l-
[(4,4-difluoro-l-piperidyl)methyl]cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00632] Compound 34 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C31H38F2N9OS (M+H)+ m/z =622.3 , found: 622.2. *HNMR (400 MHz, CD3OD) 5 4.69 - 4.27 (m, 6H), 4.26 - 4.19 (m, 2H), 3.59 - 3.48 (m, 2H), 3.29 - 3.13 (m, 2H), 2.81 - 2.37 (m, 10H), 1.99 - 1.88 (m, 4H), 1.85 - 1.75 (m, 4H), 0.69 - 0.61 (m, 2H), 0.50 - 0.41 (m, 2H).
[00633] Compound 35. 3-chloro-5-[5-cyano-6-(2',2'-difluorospiro[azetidine-3,l'-tetralin]-l- yl)-2-[[(2R,8S)-2-fluoro- 1,2, 3,5,6, 7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-N,N- dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2-carboxamide
[00634] Compound 35 was prepared similarly to that of Ex. 2 using Intermediate 12. LCMS calcld for C35H40CIF3N9O2 (M+H)+ m/z =710.3; found: 710.3. 'H NMR (500 MHz, CDCI3) 5 ppm 2.07 - 2.67 (m, 12 H) 2.99 - 3.07 (m, 3 H) 3.10 (d, 7=10.03 Hz, 6 H) 3.14 - 3.24 (m, 1 H)
3.35 - 3.47 (m, 2 H) 3.78 - 4.01 (m, 2 H) 4.01 - 4.32 (m, 3 H) 4.32 - 4.49 (m, 4 H) 4.54 (d,
7=11.55 Hz, 2 H) 4.59 - 5.14 (m, 5 H) 5.32 - 5.57 (m, 1 H) 7.13 (d, 7=7.64 Hz, 1 H) 7.22 - 7.26 (m, 1 H) 7.32 - 7.37 (m, 1 H) 7.65 (d, 7=7.89 Hz, 1 H)
[00635] Compound 36. 3-chloro-5-[5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-(2'-oxospiro[azetidine-3,r-tetralin]-l-yl)pyrimidin-4-yl]-
[00636] Compound 36 was prepared similarly to that of Ex. 2 using Intermediate 18. LCMS calcld for C35H4OC1FN902(M+H)+ m/z =688.3; found: 688.3. 'H NMR (500 MHz, CDCI3) 5 2.02
- 2.13 (m, 3 H) 2.19 - 2.29 (m, 3 H) 2.30 - 2.47 (m, 2 H) 2.71 (t, 7=6.79 Hz, 2 H) 3.06 (t, 7=6.79
Hz, 2 H) 3.10 (s, 3 H) 3.11 (s, 3 H) 3.27 - 3.43 (m, 1 H) 3.59 - 3.80 (m, 2 H) 4.04 - 4.21 (m, 2 H)
4.32 (d, 7=11.19 Hz, 1 H) 4.37 - 4.49 (m, 3 H) 4.69 - 5.12 (m, 4 H) 5.31 - 5.48 (m, 1 H) 7.20 - 7.24 (m, 1 H) 7.29 (td, 7=7.47, 0.76 Hz, 1 H) 7.38 (t, 7=7.27 Hz, 1 H) 7.63 (d, 7=7.64 Hz, 1 H) [00637] Compound 37. 3-chloro-5-[5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-(2'-oxospiro[azetidine-3,r-indane]-l-yl)pyrimidin-4-yl]-
[00638] Compound 36 was prepared similarly to that of Ex. 2 using 5-oxo-2- azaspiro[3.4]octane (Angew. Chem. Int. Ed. 2021, 60, 7360-7365). LCMS calcld for C34H38CIFN9O3 (M+H)+ m/z =674.3; found: 674.3. 'H NMR (CDCI3) d: 7.79 (d, 7= 7.6 Hz, 1H), 7.64 (t, 7= 7.5 Hz, 1H), 7.48 (d, 7= 7.6 Hz, 1H), 7.39 - 7.44 (t, 7= 7.5 Hz, 1H), 5.32 - 5.49 (d, 7= 55 Hz, 1H), 4.80 - 5.08 (dd, 7= 15 Hz , 1H), 3.99 - 4.72 (m, 10H), 3.73 - 3.94 (m, 2H), 3.54 (s, 2H), 3.46 (d, J = 1.4 Hz, 1H), 3.38 (t, 7= 16.0 Hz, 1H), 3.12 (s, 2H), 3.09 (s, 3H), 2.32 - 2.56 (m, 2H), 2.20 - 2.31 (m, 3H), 2.13 (br. s., 3H).
[00639] Compound 38. 2-amino-l'-[5-cyano-6-(4,5-dihydroxyazepan-l-yl)-2-[[(2R,8S)-2- fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00640] Compound 38 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calcld for C29H36FN8O3S (M+H)+ m/z = 595.3, found: 595.3. *HNMR (400 MHz, CD3OD) 5 5.55 (d, J = 51.6 Hz, 1H), 4.61 - 4.22 (m, 6H), 4.03 - 3.55 (m, 9H), 3.50 - 3.39 (m, 1H), 2.82 - 2.48 (m, 6H), 2.40 - 2.10 (m, 6H), 1.97 - 1.84 (m, 2H).
[00641] Compound 39. 2-[[l-(morpholinomethyl)cyclopropyl]methoxy]-4-(12-oxospiro[7- thia-9,l l-diazatricyclo[6.4.0.02,6]dodeca-l(8),2(6),10-triene-3,3'-azetidine]-r-yl)-6-[(lR,5S)- 3,8-diazabicyclo[3.2.1]octan-3-yl]pyrimidine-5-carbonitrile
[00642] Compound 39 was prepared similarly to that of Ex. 3 using Intermediate 13 as a TFA salt. LCMS calcld for C31H38N9O3S (M+H)+ m/z = 616.3, found: 616.2. *HNMR (400 MHz, CD3OD) 5 8.01 (s, 1H), 4.75-4.58 (m, 3H), 4.44-4.14 (m, 6H), 4.10-4.00 (m, 2H), 3.88- 3.63 (m, 4H), 3.60-3.31 (m, 4H), 3.29-3.04 (m, 5H), 2.99-2.90 (m, 2H), 2.15-2.03 (m, 4H), 0.95- 0.80 (m, 4H).
[00643] Compound 40. 2-amino-l'-[5-cyano-6-(4-hydroxyazepan-l-yl)-2-[[(2R,8S)-2-fluoro- l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00644] Compound 40 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calcld for C29H36FN8O2S (M+H)+ m/z = 579.3, found: 579.3. *HNMR (400 MHz, CD3OD) 5 5.66 - 5.40 (m, 1H), 4.64 - 4.29 (m, 6H), 4.02 - 3.72 (m, 8H), 3.52 - 3.38 (m, 1H), 2.82 - 2.47 (m, 6H), 2.41 - 2.24 (m, 3H), 2.18 - 2.01 (m, 3H), 1.93 - 1.60 (m, 4H).
[00645] Compound 41. 2-amino-l'-[5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]pyrimidin-4-yl]spiro[6,7-dihydro-5H-benzothiophene-4,3'-azetidine]-3-carbonitrile
[00646] Compound 41 was prepared similarly to that of Ex. 3. LCMS calcld for C31H40N9O2S (M+H)+ m/z =602.3, found: 602.2. *H NMR (400 MHz, CD3OD) 5: 4.75-4.72 (m, 1H), 4.51-4.21 (m, 6H), 4.03-4.00 (m, 1H), 3.69-3.67 (m, 4H), 3.56-3.56 (m, 2H), 3.28-3.20 (m, 2H), 2.54- 2.35 (m, 8H), 2.17-2.10 (m, 2H), 1.85-1.78 (m, 6H), 0.69-0.65 (m, 2H), 0.51-0.45 (m, 2H).
[00647] Compound 42. 2-amino-l'-[5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]-6-(3-oxa-7,9-diazabicyclo[3.3.1]nonan-7- yl)pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00648] Compound 42 was prepared similarly to that of Ex. 3. LCMS calcld for C30H38N9O3S (M+H)+ m/z = 604.3, found: 604.4. 'H NMR (400 MHz, CD3OD) 5 4.78 (d, J= 13.2 Hz, 2H), 4.65 - 4.47 (m, 2H), 4.48 - 4.31 (m, 2H), 4.25 (s, 2H), 4.03 - 3.93 (m, 2H), 3.92 - 3.82 (m, 2H),
3.66 (t, J= 4.4 Hz, 4H), 3.52 (d, J= 13.4 Hz, 2H), 2.91 (s, 2H), 2.81 - 2.67 (m, 4H), 2.48 (s, 4H), 2.38 (s, 2H), 0.64 (t, J= 5.2 Hz, 2H), 0.44 (t, J= 5.2 Hz, 2H).
[00649] Compound 43. 2-amino-l'-[5-cyano-2-[[(2S,8R)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-(3-oxa-7,9-diazabicyclo[3.3.1]nonan-7-yl)pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00650] Compound 43 was prepared similarly to that of Ex. 3. LCMS calcld for C29H35FN9O2S (M+H)+ m/z = 592.3, found: 592.2. 'H NMR (DMSO-d6, 400 MHz) 5 7.23 (s, 2H), 5.25 (d, J= 52.0 Hz, 1H), 4.60-4.57 (m, 2H), 4.34 (br, 4H), 3.97 (d, J= 12.0 Hz, 1H), 3.84 (d, J= 12.0 Hz, 1H), 3.80 (d, J= 8.0 Hz, 2H), 3.70 (d, J= 8.0 Hz, 2H), 3.41-3.89 (m, 2H), 3.05 (br, 2H), 3.01-2.93 (m, 1H), 2.84-2.79 (m, 3H), 2.67 (br, 4H), 2.07-2.05 (m, 1H), 1.99-1.98 (m, 1H), 1.95-1.93 (m, 1H), 1.83-1.80 (m, 1H), 1.75-1.71 (m, 2H).
[00651] Compound 44. 3-chloro-5-[5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-(12-oxospiro[7-thia-9,l l-diazatricyclo[6.4.0.02,6]dodeca- 1(8), 2(6), 10-triene-3, 3'-azeti dine]-l'-yl)pyrimidin-4-yl]-N,N-dimethyl-4, 6,7,8- tetrahydropyrazolo[ 1 ,5-a] [ 1 ,4]diazepine-2-carboxamide
[00652] Compound 44 was prepared similarly to that of Ex. 2 as a formate salt using Intermediate 13. LCMS calcld for C34H38CIFN11O3S (M+H)+ m/z = 734.3, found: 734.2. 'H NMR (400 MHz, CD3OD) 5 8.52 (s, 2H), 7.99 (s, 1H), 5.26 (d, J= 56.2 Hz, 2H), 4.74 - 4.41 (m, 4H), 4.15 (m, 5H), 3.17 (m, 3H), 3.08 (m, 8H), 3.01 - 2.88 (m, 3H), 2.30 - 1.76 (m, 10H).
[00653] Compound 45. 2-amino-l'-[5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]pyrimidin-4-yl]-6-methyl-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3- carbonitrile
[00654] Compound 45 was prepared similarly to that of Ex. 2 as a TFA salt using Intermediate 6 (Compound 14). LCMS calcld for C31H40N9O2S (M+H)+ m/z = 602.2, found: 602.3. ‘HNMR (400 MHz, CD3OD) 5 4.66-4.01 (m, 12H), 3.93-3.68 (m, 4H), 3.57-3.42 (m, 2H), 3.30-3.10 (m, 3H), 3.01-2.89 (m, 1H), 2.29-2.19 (m, 1H), 2.14-2.02 (m, 4H), 1.18 (d, J= 6.8 Hz, 3H), 0.95-0.78 (m, 4H).
[00655] Compound 46. 2-amino-l'-[6-[3-chloro-2-(5-methyl-l,2,4-oxadiazol-3-yl)-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3 '-azetidine]-3 -carbonitrile
[00656] Compound 46 was prepared similarly to Ex. 2 as a formate salt using Intermediate 14. LCMS calcld for C33H35CIFN12O2S (M+H)+ m/z = 717.2, found: 717.1. 'HNMR (400 MHz, CD3OD) 5 5.34 (dd, J= 35.3, 18.5 Hz, 1H), 5.12 - 5.02 (m, 2H), 4.74 - 4.44 (m, 6H), 4.29 - 4.09 (m, 5H), 3.47 - 3.33 (m, 2H), 3.15 - 3.00 (m, 1H), 2.79 - 2.68 (m, 4H), 2.66 (s, 3H), 2.43 - 1.78 (m, 9H).
[00657] Compound 47. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]- 2-[[l-(dimethylamino)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00658] Compound 47 was prepared similarly to that of Ex. 3 as a TFA. LCMS calcld for C27H34N9OS (M+H)+ m/z =532.3, found:532,2. 'H NMR (400 MHz, CD3OD) 5 4.66 - 4.53 (m, 4H), 4.44 - 4.37 (s, 2H), 4.35 - 4.16 (m, 2H), 3.62 - 3.48 (m, 2H), 3.29 - 3.10 (m, 2H), 2.83 - 2.67 (m, 4H), 2.46 (s, 6H), 1.89 - 1.71 (m, 4H), 0.80 - 0.66 (m, 4H).
[00659] Compound 48. 2-amino-l'-[5-cyano-2-[2-[cyclobutyl(methyl)amino]ethoxy]-6- [(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00660] Compound 48 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C28H36N9OS (M+H)+ m/z = 546.3, found: 546.2. 1 H NMR (400 MHz, CD3OD) 5 4.75 - 4.33 (m, 7H), 4.23 - 4.15 (m, 2H), 3.89 - 3.78 (m, 1H), 3.66 - 3.43 (m, 3H), 3.43 - 3.34 (m, 2H), 2.85 (s, 3H), 2.83 - 2.70 (m, 4H), 2.45 - 2.17 (m, 4H), 2.11 (s, 4H), 1.96 - 1.79 (m, 2H).
[00661] Compound 49. 2-amino-l'-[5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-(4-oxoazepan-l-yl)pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00662] Compound 49 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calcld for C29H34FN8O2S (M+H)+ m/z =577.2, found: 577.1. XH NMR (400 MHz, CD3OD) 5 5.57 (d, ./=52 Hz, 1H), 4.57-4.39 (m, 6H), 4.21-4.18 (m, 2H), 4.12-4.01 (m, 2H), 4.00-3.83 (m, 4H), 3.50-3.41 (m, 2H), 2.81-2.51 (m, 10H), 2.40- 2.31 (m, 3H), 2.29-1.89 (m, 3H).
[00663] Compound 50. 4-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]-2-[[l- (morpholinomethyl)cyclopropyl]methoxy]-6-(2'-oxospiro[azetidine-3,r-tetralin]-l- yl)pyrimidine-5-carbonitrile
[00664] Compound 50 was prepared similarly to that of Ex. 3 using Intermediate 18. LCMS calcld for C32H40N7O3 (M+H)+ m/z = 570.3, found: 570.2. 1 H NMR (400 MHz, DMSO-d6) 5 7.65 (d, J= 8.0 Hz, 1H), 7.39-7.35 (m, 1H), 7.31-7.30 (m, 2H), 4.69 (br, 2H), 4.23-4.20 (m, 2H), 4.15 (s, 4H), 3.58 (br, 2H), 3.53 (br, 5H), 3.23 (d, J= 12.0 Hz, 2H), 3.04 (m, 2H), 2.70 (m, 2H), 2.35 (br, 4H), 2.24 (s, 2H), 1.69-1.62 (m, 4H), 0.57(s, 2H), 0.38 (s, 2H).
[00665] Compound 51. 2-amino-l'-[6-(4-cyanoazepan-l-yl)-5-fluoro-2-[[(2R,8S)-2-fluoro- l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00666] Compound 51 was prepared similarly to that of Ex. 4 as a TFA salt. LCMS calcld for C29H35F2N8OS (M+H)+ m/z =581.3, found:581.1. *HNMR (400 MHz, CD3OD) 5 5.54 (dt, J= 51.6, 3.5 Hz, 1H), 4.52 - 4.30 (m, 4H), 4.24 (d, J= 8.8 Hz, 2H), 4.04 - 3.86 (m, 2H), 3.85 - 3.68 (m, 5H), 3.49 - 3.39 (m, 1H), 3.04 (d, J= 11.3 Hz, 1H), 2.82 - 2.71 (m, 4H), 2.70 - 2.45 (m, 2H), 2.39 - 2.24 (m, 3H), 2.22 - 1.90 (m, 7H).
[00667] Compound 52. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]- 2-[[3-[(dimethylamino)methyl]oxetan-3-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00668] Compound 52 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C28H36N9O2S (M+H)+ m/z =562.3, found:562.2. 1H NMR (400 MHz, CD3OD) 5 4.83 - 4.61 (m,
6H), 4.60 - 4.35 (m, 6H), 4.24 - 4.10 (m, 2H), 3.79 - 3.62 (m, 2H), 3.60 - 3.42 (m, 2H), 2.91 (s, 6H), 2.81 - 2.68 (m, 4H), 2.19 - 1.99 (m, 4H).
[00669] Compound 53. 4-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]-6-(2'- hydroxy spiro[azetidine-3 , 1 '-tetralin]- 1 -yl)-2-[[ 1 -
[00670] Compound 53 was prepared similarly to that of Ex. 3 as a TFA salt using Intermediate 18. LCMS calcld for C32H42N7O7 (M+H)+ m/z = 572.3, found: 572.2. *HNMR (DMSO-d6, 400 MHz) 5 7.58 (d, J= 8.0 Hz, 1H), 7.26 (t, J= 8.0 Hz, 1H), 7.19 (t, J= 8.0 Hz, 1H), 7.10 (m, 1H), 5.38 (m, 1H), 4.34 (br, 3H), 4.16 (s, 2H), 4.06 (br, 2H), 3.93-3.90 (m, 1H), 3.54 (m, 5H), 3.43-3.41 (m, 3H), 2.91-2.84 (m, 1H), 2.78-2.63 (m, 1H), 2.37-2.33 (m, 5H), 2.25 (br, 2H), 1.91-1.82 (m, 6H), 1.66 (br, 1H) 0.57 (s, 2H), 0.40 (s, 2H).
[00671] Compound 54. [l-[6-(2-amino-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3'-azetidine]-l'-yl)-5-fluoro-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8- yl]methoxy]pyrimidin-4-yl]-3-piperidyl]cyanamide
[00672] Compound 54 was prepared similarly to that of Ex. 4 as a TFA salt. LCMS calcd for C28H34F2N9OS (M+H)+ m/z: 582.3, found: 582.4. *HNMR (400 MHz, CD3OD) 5 5.54 (d, J= 52.1 Hz, 1H), 4.51 - 4.33 (m, 4H), 4.26 - 4.15 (m, 3H), 4.03 - 3.70 (m, 4H), 3.46 - 3.39 (m, 1H), 3.27 - 3.22 (m, 3H), 2.90 - 1.98 (m, 12H), 1.82 (s, 1H), 1.60 (s, 1H).
[00673] Compound 55. 2-amino-l'-[5-cyano-6-(6,7-dihydro-4H-triazolo[l,5-a]pyrazin-5-yl)- 2-[[(2R,8S)-2-fhioro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00674] Compound 55 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calculated for C28H31FN11OS (M+H)+ m/z =588.2, found: 588.2. 1 H NMR (400 MHz, CD3OD) 5 7.65 (s, 1H), 5.65-5.46 (m, 1H), 5.12 (s, 2H), 4.75-4.20 (m, 9H), 4.00-3.82 (m, 3H), 3.50-3.40 (m, 1H), 2.82-2.50 (m, 6H), 2.41-2.26 (m, 3H), 2.20-2.10 (m, 1H).
[00675] Compound 56. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]- 2-[[l-[(4,4-difluoro-l-piperidyl)methyl]cyclopropyl]methoxy]pyrimidin-4-yl]spiro[6,7-dihydro- 5H-benzothiophene-4,3'-azetidine]-3-carbonitrile
[00676] Compound 56 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C32H40F2N9OS (M+H)+ m/z =636.3 , found: 636.2.. 'H NMR (400 MHz, CD3OD) 5 4.71 - 4.56 (m, 2H), 4.52 (s, 1H), 4.40 (s, 2H), 4.29 (d, J= 16.0 Hz, 2H), 4.16 (s, 2H), 4.00 (s, 1H), 3.61 - 3.38 (m, 3H), 2.49 (m, 6H), 2.10 (m, 7H), 1.82 (s, 2H), 1.30 (s, 4H), 0.91 (s, 2H), 0.83 (s, 2H).
[00677] Compound 57. 2-amino-l'-[5-cyano-2-[(2-methyl-3,4-dihydro-lH-isoquinolin-5- yl)oxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00678] Compound 57 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C31H34N9OS (M+H)+ m/z: 580.3, found: 580.4. XH NMR (400 MHz, CD3OD) 5 7.36 (t, J= 7.9 Hz, 1H), 7.13 (dd, J= 10.9, 7.9 Hz, 2H), 4.73 - 4.17 (m, 7H), 4.10 (s, 3H), 3.62 - 3.33 (m, 4H), 3.04 (s, 5H), 2.74 - 2.70 (m, 4H), 2.04 (s, 4H).
[00679] Compound 58. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]-
2-[[l-[(3,3-difluoropyrrolidin-l-yl)methyl]cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00680] Compound 58 was prepared similarly to that of Ex. 3 as a formate salt. LCMS calcld for C30H36F2N9OS (M+H)+ m/z = 608.3, found: 608.4. *HNMR (400 MHz, CD3OD) 54.58 (s, 6H), 4.24 (s, 2H), 3.63 (s, 2H), 2.88 (d, J= 13.3 Hz, 2H), 2.79 - 2.67 (m, 6H), 2.47 (s, 2H), 2.22 - 2.15 (m, 3H), 2.03 - 1.98 (m, 2H), 1.84 (s, 3H), 0.63 (t, J= 5.2 Hz, 2H), 0.45 (t, J= 5.3 Hz, 2H).
[00681] Compound 59. 2-amino-l'-[5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-[(4Z)-4-methoxyiminoazepan-l-yl]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00682] Compound 59 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calcld for C30H37FN9O2S (M+H)+ m/z =606.3, found: 606.2. *H NMR (400 MHz, CD3OD) 5: 5.54 (d, .7=51.6 Hz, 1 H), 4.57-4.39 (m, 6 H), 4.07-3.84 (m, 7 H), 3.77-3.76 (m, 3 H), 3.48-3.41 (m, 1 H), 2.80-2.57 (m, 8 H), 2.38- 2.29 (m, 3 H), 2.27-2.10 (m, 1 H), 1.91-1.78 (m, 1H).
[00683] Compound 60. 2-amino-l'-[6-[3-chloro-2-(4,5-dimethyl-l,2,4-triazol-3-yl)-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3 '-azetidine]-3 -carbonitrile
[00684] Compound 60 was prepared similarly to that of Ex. 2 as a TFA salt using Intermediate 15. LCMS calcld for C34H38CIFN13OS (M+H)+ m/z =730.3, found: 730.4. 1 H NMR (400 MHz, CD3OD) 5: 5.58 (d, ./=52 Hz, 1H), 5.14-5.01 (m, 2H), 4.59-4.43 (m, 8H), 4.34- 4.25 (m, 2H), 3.98-3.85 (m, 3H), 3.808 (s, 3H), 3.50-3.41 (m, 1H), 2.79- 2.72 (m, 4H), 2.69-2.50 (m, 5H), 2.41 -2.11 (m, 6H).
[00685] Compound 61. 2-amino-l'-[6-[(lR,5S)-8-amino-3-azabicyclo[3.2.1]octan-3-yl]-5- cyano-2-[[l-(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00686] Compound 61 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calcld for C31H40N9O2S (M+H)+ m/z =602.2, found: 602.3. *HNMR (400 MHz, CD3OD) 5: 4.67 - 4.22 (m, 10H), 4.17 - 4.00 (m, 3H), 3.98 - 3.65 (m, 5H), 3.62 - 3.55 (m, 1H), 3.24 - 3.09 (m, 2H), 2.86 - 2.72 (m, 4H), 2.56 - 2.45 (m, 2H), 1.98 - 1.76 (m, 4H), 0.98 - 0.91 (m, 2H), 0.89 - 0.81 (m, 2H).
[00687] Compound 62. 2-amino-l'-[6-[3-chloro-2-(cyclopropanecarbonyl)-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3 '-azetidine]-3 -carbonitrile
[00688] Compound 62 was prepared similarly to that of Ex. 2 as a formate salt using Intermediate 16. LCMS calcld for C34H37CIFN10O2S (M+H)+ m/z =703.2, found:703.4. 'H NMR (400 MHz, CD3OD) 5 8.51 (s, 1H), 5.37 (d, J= 51.0 Hz, 1H), 5.07 - 4.97 (m, 2H), 4.68 - 4.38 (m, 6H), 4.29 - 4.17 (m, 4H), 3.45 (m, 3H), 3.18 - 3.09 (m, 1H), 3.01 (m, 1H), 2.74 (m, 4H), 2.34 (m, 4H), 2.18 - 1.96 (m, 3H), 1.92 (s, 1H), 1.09 (m, 2H), 1.02 (m, 2H).
Example 6. Exemplary synthesis of 2-amino-l'-[5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[6,7-dihydro-5H-benzothiophene-
[00689] Step 1. Synthesis of tert-butyl N-[3-cyano-l'-(5-cyano-2-methylsulfanyl-pyrimidin-4- yl)spiro[6,7-dihydro-5H-benzothiophene-4,3'-azetidine]-2-yl]carbamate (63a). The solution of 4- chloro-2-methylsulfanyl-pyrimidine-5-carbonitrile (43.59 mg, 0.23 mmol), K2CO3 (108.01 mg, 0.7 8mmol) and tert-butyl N-(3-cyanospiro[6,7-dihydro-5H-benzothiophene-4,3'-azetidine]-2- yl)carbamate (50 mg, 0.16 mmol) in IPA (5mL) was stirred at 50 °C for Ih. The solvent was removed and purified by silica gel chromatography (PE: EtOAc=3: l) to get tert-butyl N-[3- cyano-r-(5-cyano-2-methylsulfanyl-pyrimidin-4-yl)spiro[6,7-dihydro-5H-benzothiophene-4,3'- azetidine]-2-yl]carbamate (63a, 45 mg, 0.0960 mmol, 61.35 %yield) (crude, white solid). LCMS calculated for C22H25N6O2S2 (M+H)+ m/z =469.1, found: 469.3.
[00690] Step 2. Synthesis of tert-butyl N-[3-cyano-r-(5-cyano-2-methylsulfmyl-pyrimidin-4- yl)spiro[6,7-dihydro-5H-benzothiophene-4,3'-azetidine]-2-yl]carbamate (63b). The solution of
oxone (118.07 mg, 0.19 mmol) and tert-butyl N-[3-cyano-l'-(5-cyano-2-methylsulfanyl- pyrimidin-4-yl)spiro[6,7-dihydro-5H-benzothiophene-4,3'-azetidine]-2-yl]carbamate (45 mg, 0.1 mmol) in Water (2mL) and THF (2mL) was stirred at 25 °C for 18h. The mixture was extracted with EtOAc and water, dried with brine and Na2SO4, concentrated to get tert-butyl N-[3-cyano- T-(5-cyano-2-methylsulfinyl-pyrimidin-4-yl)spiro[6,7-dihydro-5H-benzothiophene-4,3'- azetidine]-2-yl]carbamate (63b, 25 mg, 0.0516 mmol, 53.72%yield) (crude, white solid) LCMS calculated for C22H24NeNaO3S2 (M+Na)+ m/z =507.1, found: 507.3.
[00691] Step 3. Synthesis of tert-butyl N-[3-cyano-l'-(5-cyano-2-hydroxy-pyrimidin-4- yl)spiro[6,7-dihydro-5H-benzothiophene-4,3'-azetidine]-2-yl]carbamate (63c). The NaH (12 mg, 0.3 mmol) was put into the solution of [l-(morpholinomethyl)cyclopropyl]methanol (51.37 mg, 0.3 mmol) in DMSO (1.5mL) at 25 °C. After stirring for Ih, tert-butyl N-[3-cyano-l'-(5-cyano- 2-methylsulfinyl-pyrimidin-4-yl)spiro[6,7-dihydro-5H-benzothiophene-4,3'-azetidine]-2- yl]carbamate (63b, 25 mg) and tert-butyl N-[3-cyano-l'-(5-cyano-2-methylsulfonyl-pyrimidin-4- yl)spiro[6,7-dihydro-5H-benzothiophene-4,3'-azetidine]-2-yl]carbamate (25 mg) in DMSO was added, and the mixture was stirred at rt for Ih. The mixture was quenched with water, extracted with EtOAc, dried with brine and Na2SO4, concentrated and purified by prep-HPLC (DCM: MeOH= 20:1) to get tert-butyl N-[3-cyano-l'-[5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[6,7-dihydro-5H-benzothiophene- 4,3'-azetidine]-2-yl]carbamate (63c, 30 mg, 0.0507 mmol, 98.27 %yield) (light yellow oil). LCMS calculated for C30H38N7O4S (M+H)+ m/z =592.3, found: 592.4.
[00692] Step 4. Synthesis of 2-amino-l'-[5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[6,7-dihydro-5H-benzothiophene- 4,3 '-azetidine]-3 -carbonitrile (Compound 63). The solution of Trifluoroacetic acid (0.06 mL, 0.51 mmol) and tert-butyl N-[3-cyano-l'-[5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[6,7-dihydro-5H-benzothiophene- 4,3'-azetidine]-2-yl]carbamate (30 mg, 0.05 mmol) in DCM (3mL) was stirred at rt for 5h. The solvent was removed and purified by prep-HPLC (0.1% TFA) to get 2-amino-l'-[5-cyano-2-[[l- (morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[6,7-dihydro-5H-benzothiophene- 4,3 '-azetidine]-3 -carbonitrile (Compound 63, 10 mg, 0.0201 mmol, 39.72 %yield). LCMS calculated for C25H30N7O2S (M+H)+ m/z =492.2, found: 492.2. 'H NMR (400 MHz, CD3OD) 5 8.39 (s, IH), 4.87 - 4.85 (m, IH), 4.50 (dd, J= 22.9, 9.9 Hz, 2H), 4.32 (s, 2H), 4.07 (m, 3H), 3.83 (m, 2H), 3.71 (m, 2H), 3.32 - 3.26 (m, 2H), 3.19 - 3.05 (m, 2H), 2.51 (t, J = 6.1 Hz, 2H), 2.15 (m, 2H), 1.98 - 1.75 (m, 2H), 1.02 - 0.76 (m, 4H).
[00693] Compound 64. 2-amino-l'-[5-cyano-2-[[3-(morpholinomethyl)oxetan-3- yl]methoxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00694] Compound 64 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C30H38N9O3S (M+H)+ m/z =604.3, found:604.1. 'H NMR (400 MHz, CD3OD) 5 4.61 (s, 4H), 4.54 (dd, J= 14.8, 6.2 Hz, 6H), 3.61 (s, 4H), 3.56 (s, 2H), 2.76 (m, 6H), 2.38 (s, 4H), 1.81 (s, 4H), 1.31 (d, J= 17.6 Hz, 4H).
[00695] Compound 65. 2-amino-l'-[5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3 '-azetidine]-3 -carbonitrile
[00696] Compound 65 was prepared similarly to that of Ex. 6 as a TFA salt. LCMS calcld for C23H25FN7OS (M+H)+ m/z = 466.2, found: 466.3. XH NMR (400 MHz, CDCI3) 6 8.28 (s, 1H), 5.39 - 5.26 (m, 1H), 4.84 (s, 2H), 4.59 (s, 2H), 4.27 (s, 2H), 3.53 (s, 2H), 3.33 - 3.25 (m, 1H), 3.05 (s, 1H), 2.82 (d, J= 6.4 Hz, 2H), 2.74 (s, 2H), 2.38 - 2.15 (m, 5H), 2.08 - 1.98 (m, 3H).
[00697] Compound 66. 2-amino-l'-[5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00698] Compound 66 was prepared similarly to that of Ex. 6 as a TFA salt. LCMS calcld for C24H28N7O2S (M+H)+ m/z =478.2, found: 478.2. *HNMR (400 MHz, CD3OD) 5 8.40 (s, 1H), 4.75 - 4.67 (m, 2H), 4.33 (s, 6H), 4.07 (s, 2H), 3.82 (s, 2H), 3.73 - 3.66 (m, 2H), 3.13 (s, 2H), 2.78 (s, 4H), 0.95 (s, 2H), 0.84 (s, 2H).
[00699] Compound 67. 2-amino-l'-[5-cyano-6-(6-methyl-6,7-dihydro-4H-triazolo[l,5- a]pyrazin-5-yl)-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00700] Compound 67 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calculated for C29H33FN11OS (M+H)+ m/z =602.3, found: 602.2. 1 H NMR (400 MHz, CD3OD) 5 7.64 (s, 1H), 5.66-5.46 (m, 1H), 5.33-5.22 (m, 1H), 4.86-4.75 (m, 1H), 4.64-4.40 (m, 6H), 4.04-3.81 (m, 4H), 3.51-3.38 (m, 1H), 2.84-2.50 (m, 7H), 2.41-2.24 (m, 3H), 2.20-2.09 (m, 1H), 1.69 (d, J= 6.8 Hz, 3H).
[00701] Compound 68. 2-amino-l'-[5-cyano-2-[[2,2-difluoro-l- (morpholinomethyl)cyclopropyl]methoxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00702] Compound 68 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcd for C30H36F2N9O2S (M+H)+ m/z = 624.3. found: 624.4. 1HNMR (400 MHz, CD3OD) 54.66 - 4.39 (m, 8H), 4.16 (s, 2H), 3.95 (s, 4H), 3.71 - 3.44 (m, 4H), 3.36 - 3.31 (m, 4H), 2.76 (d, J= 5.9 Hz, 4H), 2.09 (s, 4H), 1.95 - 1.65 (m, 2H).
[00703] Compound 69. 5-[3-(5-amino-4-cyano-spiro[4,5-dihydro-2H-thieno[2,3- b]thiophene-3,3'-azetidine]-l'-yl)-2-cyano-5-[[(2R,8S)-2-fluoro-l,2,3,5,6,7-hexahydropyrrolizin- 8-yl]methoxy]phenyl]-3-chloro-N,N-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5-a][l,4]diazepine- 2-carboxamide
[00704] Compound 69 was prepared similarly to that of Ex. 2 using Intermediate 17. LCMS calcld for C32H36CIFN11O2S2 (M+H)+ m/z = 724.2, found: 724.5. JH NMR (400 MHz, CD3OD) 5 5.25 (d, J= 54.6 Hz, 1H), 5.12 - 5.03 (m, 2H), 4.64 - 4.52 (m, 2H), 4.53 - 4.35 (m, 4H), 4.23 - 4.06 (m, 4H), 4.03 (s, 2H), 3.24 - 3.12 (m, 3H), 3.09 (s, 3H), 3.08 (s, 3H), 3.02 - 2.93 (m, 1H), 2.28 - 1.80 (m, 8H).
[00705] Compound 70. 2-amino-l'-[5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]pyrimidin-4-yl]spiro[5,6,7,8-tetrahydrocyclohepta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00706] Compound 70 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C32H42N9O2S (M+H)+ m/z = 616.3, found: 616.6. 'H NMR (400 MHz, CD3OD) 5 4.80 - 4.24 (m, 7H), 4.19 - 4.00 (m, 5H), 3.93 - 3.67 (m, 4H), 3.60 - 3.43 (m, 2H), 3.39 - 3.31 (m, 2H), 3.21 - 3.06 (m, 2H), 2.85 - 2.58 (m, 2H), 2.38 - 2.23 (m, 2H), 2.15 - 1.99 (m, 4H), 1.93 - 1.72 (m, 4H), 0.98 - 0.79 (m, 4H).
[00707] Compound 71. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]- 2-[[l-[(l-oxo-l,4-thiazinan-4-yl)methyl]cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00708] Compound 71 was prepared similarly to that of Ex. 3. LCMS calcld for C30H36N9O2S (M-H)- m/z = 618.3, found:618.2.
[00709] Compound 72. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]-
2-[[l-(thiomorpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00710] Compound 72 was prepared similarly to that of Ex. 3. LCMS calcld for C30H38N9OS2 (M+H)+ m/z = 604.3, found:604.1.
[00711] Compound 73. 5-amino-l'-[5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]pyrimidin-4-yl]spiro[2H-thieno[2,3-b]thiophene-3,3'-azetidine]-4-carbonitrile
[00712] Compound 73 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C29H36N9O2S2 (M+H)+ m/z = 606.2, found: 606.1. *HNMR (400 MHz, CD3OD) 54.95 - 4.86 (m, 2H), 4.74 - 4.44 (m, 5H), 4.30 - 4.15 (m, 4H), 4.12 - 3.97 (m, 4H), 3.93 - 3.62 (m, 4H), 3.55 - 3.38 (m, 3H), 3.26 - 3.01 (m, 2H), 2.13 - 2.03 (m, 4H), 0.95 - 0.77 (m, 4H).
[00713] Compound 74 2 -amino-l'-[5-cyano-6-(3,8-diazabicyclo[3.2.1]octan-3-yl)-2-[[l- [(dimethylamino)methyl]-2,2-difluoro-cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00714] Compound 74 was prepared similarly to that of Ex. 3. LCMS calculated for
C28H34F2N9OS (m/z) =582.3; found:582.3. 'H NMR (400 MHz, CD3OD) 5 4.16- 4.67 (m, 8H),
3.55 (s, 2H), 3.23 - 3.29 (m, 2H), 2.65-2.85 (m, 5H), 2.38 (d, J= 12.8 Hz, 1H), 2.24 (s, 6H), 1.73- 1.86 (m, 4H), 1.53 - 1.66 (m, 1H), 1.27 - 1.40 (m, 1H).
[00715] Compound 74A and 74B. 2 -amino-l'-[5-cyano-6-(3,8-diazabicyclo[3.2.1]octan-3- yl)-2-[[(l*)-l-[(dimethylamino)methyl]-2,2-difluoro-cyclopropyl]methoxy]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00716] Compound 74 was purified on an DAICELCHIRALCEL®AD (250*25 mm 10 mm) column on a Waters SFC 150 system (Mobile Phase A: Supercritical CO2, Mobile Phase B : EtOH [0.5%NH3(7M in MeOH)] =75/25; Flow: lOOml/min to give faster eluting Pl (74A) and slower eluting P2 (74B).
[00717] Compound 75. 2-amino-l'-[6-[3-chloro-2-(2-pyridyl)-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-[[l- (morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00718] Compound 75 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calcld for C36H39CIN11O2S (M+H)+ m/z = 724.3, found: 724.5. 'H NMR (400 MHz, CD3OD) 5 8.68 (m, 1H), 8.22 (m, 2H), 7.65 (m, 1H), 4.63 - 4.41 (m, 5H), 4.39 - 4.13 (m, 5H), 4.10 - 3.91 (m, 2H), 3.88 - 3.51 (m, 4H), 3.29 - 3.22 (m, 4H), 3.18 - 2.98 (m, 2H), 2.82 - 2.65 (m, 4H), 2.37 - 2.22 (m, 2H), 0.94 - 0.72 (m, 4H).
[00719] Compound 76. 2-amino-l'-[2-benzyloxy-5-cyano-6-[(lR,5S)-3,8- diazabicyclo[3.2.1]octan-3-yl]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'- azetidine]-3-carbonitrile
[00720] Compound 76 was prepared similarly to that of Ex. 3. LCMS calcld for C28H29N8OS (M+H)+ m/z = 525.2, found: 525.2. 'H NMR (400 MHz, CD3OD) 5 7.45 - 7.37 (m, 2H), 7.37 -
7.32 (m, 2H), 7.32 - 7.26 (m, 1H), 5.33 (s, 2H), 4.73 - 4.11 (m, 6H), 3.63 - 3.44 (m, 2H), 3.25 (m, 2H), 2.81 - 2.63 (m, 4H), 1.89 - 1.55 (m, 4H).
[00721] Compound 77. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]-
2-[[l-[[(3S)-3-methylmorpholin-4-yl]methyl]cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00722] Compound 77 was prepared similarly to that of Ex. 3. LCMS calcld for C31H40N9O2S (M+H)+ m/z = 602.3, found: 602.2. 'H NMR (400 MHz, CD3OD) 5 4.51 (d, J= 10.7 Hz, 4H), 4.32 (s, 3H), 3.92 (d, J= 10.9 Hz, 1H), 3.72 (m, 3.1 Hz, 1H), 3.67 - 3.51 (m, 4H), 3.26 (s, 3H), 3.18 (m, 1H), 3.00 (m, 1H), 2.80 - 2.67 (m, 4H), 2.34 (m, 1H), 2.19 (m, 1H), 1.80 (s, 4H), 1.63 (d, J= 12.6 Hz, 1H), 0.91 (d, J= 6.3 Hz, 3H), 0.65 (m, 1H), 0.60 - 0.47 (m, 2H), 0.36 (m, 1H).
[00723] Compound 78. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]-
2-[[l-[[(3S,4R)-3,4-difluoropyrrolidin-l-yl]methyl]cyclopropyl]methoxy]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00724] Compound 78 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C30H36F2N9OS (M+H)+ m/z = 608.3, found: 608.3. *HNMR (400 MHz, CD3OD) 5 5.56-5.33 (m,
2H), 4.75-4.20 (m, 5H), 4.27 (s, 2H), 4.19-3.93 (m, 4H), 3.89-3.63 (m, 2H), 3.57-3.32 (m, 5H), 2.83-2.68 (m, 4H), 2.08 (s, 4H), 0.94-0.79 (m, 4H).
[00725] Compound 79. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]-
2-(lH-pyrazol-5-ylmethoxy)pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'- azetidine]-3-carbonitrile
[00726] Compound 79 was prepared similarly to that of Ex. 3. LCMS calcld for C25H27N10OS (M+H)+ m/z = 515.2, found: 515.1. 'HNMR (400 MHz, CD3OD) 5 7.59 (s, 1H), 6.39 (d, J= 2.0 Hz, 1H), 5.35 (s, 2H), 4.46 (m, 6H), 3.54 (s, 2H), 3.26 (m, 2H), 2.82 - 2.62 (m, 4H), 1.87 - 1.64 (m, 4H), 1.30 (s, 1H).
[00727] Compound 80. 2-amino-l'-[5-cyano-2-[[l-[(4-methyl-3-oxo-piperazin-l- yl)methyl]cyclopropyl]methoxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00728] Compound 80 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for
C31H39N10O2S (M+H)+ m/z = 615.3, found: 615.2. ‘HNMR (400 MHz, CD3OD) 5 4.61 (s, 5H), 4.31 (s, 3H), 4.15 (s, 2H), 3.64 (s, 2H), 3.48 (t, J = 15.8 Hz, 2H), 2.99 (s, 3H), 2.94 - 2.84 (m, 2H), 2.82 - 2.70 (m, 2H), 2.09 (s, 4H), 0.83 - 0.77 (m, 2H), 0.77 - 0.71 (m, 2H).
[00729] Compound 81. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]- 2-[[l-[[(3S,4S)-3-fluoro-4-methoxy-pyrrolidin-l-yl]methyl]cyclopropyl]methoxy]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00730] Compound 81 was prepared similarly to that of Ex. 3 as a TFA salt of a mixture of diastereomers. LCMS calcld for C31H39N9O2S (M+H)+ m/z = 620.3, found: 602.3. *HNMR (400 MHz, CD3OD) 5 5.35 (d, J= 51.6 Hz, 1H), 4.72-4.40 (m, 5H), 4.38-4.12 (m, 4H), 3.57-3.42 (m, 6H), 3.60-3.42 (m, 7H), 3.33-3.20 (m, 4H), 2.81-2.70 (m, 4H), 0.96-0.76 (m, 4H).
[00731] Compound 82. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]-
2-[[l-[[(3R)-3-methylmorpholin-4-yl]methyl]cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00732] Compound 82 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C31H40N9O2S (M+H)+ m/z = 602.3, found: 602.2. 'H NMR (400 MHz, CD3OD) 5 4.76 - 4.39 (m, 5H), 4.38 - 4.21 (m, 3H), 4.16 (s, 2H), 4.12 - 3.77 (m, 5H), 3.67 - 3.55 (m, 1H), 3.56 - 3.44 (m, 2H), 3.49 - 3.35 (m, 2H), 3.27 - 3.09 (m, 1H), 2.81 - 2.69 (m, 4H), 2.08 (s, 3H), 2.07 - 1.98 (m, 1H), 1.49 - 1.32 (m, 3H), 1.07 - 0.67 (m, 4H).
[00733] Compound 83. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]-
2-[[(2S)-l-methylpyrrolidin-2-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00734] Compound 83 was prepared similarly to that of Ex. 3 as a formate salt. LCMS calcld for C27H34N9OS (M+H)+ m/z = 532.3, found: 532.2. *HNMR (400 MHz, CD3OD) 5 8.44 (s, 2H), 4.68 - 4.41 (m, 7H), 4.06 (s, 2H), 3.68 - 3.53 (m, 2H), 3.52 - 3.41 (m, 2H), 3.15 - 3.01 (m, 1H), 2.92 (s, 3H), 2.81 - 2.70 (m, 4H), 2.37 - 2.24 (m, 1H), 2.15 - 1.90 (m, 7H).
[00735] Compound 84. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]- 2-[(l-phenylcyclopropyl)methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'- azetidine]-3-carbonitrile
[00736] Compound 84 was prepared similarly to that of Ex. 3. LCMS calcld for C3iH33N8OS(M+H)+ m/z =565.2 , found:565.2. 'H NMR (400 MHz, CD3OD) 5 7.35 (d, J= 13 Hz, 2H), 7.25 (t, J= 7.5 Hz, 2H), 7.15 (t, J= 7.2 Hz, 1H), 4.66 - 4.47 (m, 2H), 4.47 - 4.37 (m, 3H), 4.36 - 4.09 (m, 3H), 3.56 (s, 2H), 3.23 (m, 2H), 2.79 - 2.65 (m, 4H), 1.89 - 1.73 (m, 4H), 1.05 - 0.87 (m, 4H).
[00737] Compound 85. 2-amino-l'-[6-[3-chloro-2-(2,5-dimethyl-l,2,4-triazol-3-yl)-4,6,7,8- tetrahydropyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3 '-azetidine]-3 -carbonitrile
[00738] Compound 85 was prepared similarly to that of Ex. 2 as a TFA salt. LCMS calculated for C34H38CIFN13OS (M+H)+ m/z =730.3, found: 730.2. 'H NMR (400 MHz, CD3OD) 5 5.55 (d, J= 51.6 Hz, 1H), 5.17-4.98 (m, 2H), 4.70-4.17 (m, 10H), 3.99 (s, 3H), 3.98-3.79 (m, 3H), 3.51- 3.35 (m, 1H), 2.82-2.44 (m, 6H), 2.40-2.22 (m, 8H), 2.17-2.04 (m, 1H).
[00739] Compound 86. 2-amino-l'-[5-cyano-2-[[l-
(morpholinomethyl)cyclopropyl]methoxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]pyrimidin-4-yl]spiro[7H-benzothiophene-4,3'-azetidine]-3 -carbonitrile
[00740] Compound 86 was prepared similarly to that of Ex. 3. LCMS calculated for C31H38N9O2S (M+H)+ m/z =600.3, found: 600.2. ‘HNMR (400 MHz, CD3OD) 5: 6.18-5.98 ( m, 2H), 4.84-4.77 (m, 1H), 4.60-4.57 (m, 1H), 4.49-4.40 (m, 1H), 4.24-4.18 (m, 2H), 4.17-4.12 (m, 1H), 3.66-3.57 (m, 8H), 3.27- 3.18 (m, 4H), 2.47-2.37 (m, 6H), 1.87- 1.80 (m, 4H), 0.65- 0.61 (m, 2H), 0.46- 0.43 (m, 2H).
[00741] Compound 87. 5-[6-(2-amino-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3'-azetidine]-l'-yl)-5-cyano-2-[[l-[(dimethylamino)methyl]-2,2-difluoro- cyclopropyl]methoxy]pyrimidin-4-yl]-3-chloro-N,N-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5- a] [ 1 ,4]diazepine-2-carboxamide
[00742] Compound 87 was prepared similarly to that of Ex. 2. LCMS calculated for C32H37CIF2N11O2S (M+H)+ m/z =712.2; found:712.3. 'HNMR (400 MHz, CD3OD) 5 5.08 (s, 2H), 4.58 (s, 2H), 4.45 - 4.49 (m, 2H), 4.42 (s, 2H), 4.19 (s, 2H), 3.08 (d, J= 2.8 Hz, 6H), 2.69 - 2.79 (m, 4H), 2.45 (s, 1H), 2.28 (s, 7H), 1.60 (s, 2H), 1.26 - 1.38 (m, 5H), 0.86 - 0.99 (m, 1H).
[00743] Compound 88. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]-
2-[(lS)-l-[(2S)-l-methylpyrrolidin-2-yl]ethoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00744] Compound 88 was prepared similarly to that of Ex. 3 as a formate salt. LCMS calcld for C28H36N9OS (M+H)+ m/z = 546.3, found: 546.2. ‘HNMR (400 MHz, CD3OD) 5 8.46 (s,
2H), 5.26 - 5.14 (m, 1H), 4.69 - 4.34 (m, 5H), 4.01 (s, 2H), 3.58 - 3.40 (m, 4H), 3.14 - 3.02 (m, 1H), 2.95 (s, 3H), 2.83 - 2.69 (m, 4H), 2.37 - 2.24 (m, 1H), 2.16 - 2.07 (m, 1H), 2.06 - 1.96 (m, 6H), 1.92 - 1.82 (m, 1H), 1.41 (d, J = 6.1 Hz, 3H).
[00745] Compound 89. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]- 2-[[l-[[(lS,4S)-2-oxa-5-azabicyclo[2.2.1]heptan-5-yl]methyl]cyclopropyl]methoxy]pyrimidin-4- yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3-carbonitrile
[00746] Compound 89 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C31H38N9O2S (M+H)+ m/z = 600.3, found: 600.3. 'H NMR (400 MHz, CD3OD) 5 5.05 - 4.88 (m, 2H), 4.74 - 4.44 (m, 6H), 4.40 - 4.07 (m, 5H), 4.01 - 3.75 (m, 2H), 3.67 - 3.41 (m, 3H), 3.22 - 2.94 (m, 2H), 2.85 - 2.62 (m, 4H), 2.32 - 1.98 (m, 6H), 1.10 - 0.74 (m, 4H).
[00747] Compound 90. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-[(4-fluoro-lH-pyrazol-5-yl)methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00748] Compound 90 was prepared similarly to that of Ex. 3. LCMS calcld for C25H26FN10OS (M+H)+ m/z = 533.2, found: 533.3. 'H NMR (400 MHz, CD3OD) 5 8.46 (d, J = 4.6 Hz, 1H), 4.67 (s, 2H), 4.66 - 4.60 (m, 2H), 4.59 - 4.26 (m, 4H), 3.63 - 3.53 (m, 2H), 3.39 - 3.32 (m, 2H), 2.89 - 2.65 (m, 4H), 1.87 - 1.74 (m, 4H).
[00749] Example 7. preparation of 5-[6-(2-amino-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3-chloro-7,7-difluoro-N,N-dimethyl-6,8- dihydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxamide TFA salt Compound 91.
[00750] Step 1. Preparation of tert-butyl N-(2,2-difluoro-3-hydroxy-propyl)carbamate (91a)
[00751] To a solution of 3-amino-2,2-difluoro-propan-l-ol (2 g, 18 mmol) in THF (20 mL) and water (20 mL), was added BOC2O (3.93 g, 18 mmol), and the reaction mixture was stirred at 25 °C for 16 h. The mixture was evaporated under reduced pressure to give crude product, and then the mixture was extracted by EtOAc (10 ml x3), washed by brine (10 ml x 2), dried over Na2SO4, concentrated. The crude product was washed with hexane to afford tert-butyl N-(2,2- difhroro-3-hydroxy-propyl)carbamate (3.10 g, 14.7 mmol, 81.53%) yield as white solid.
[00752] Step 2. Preparation of diethyl l-[3-(tert-butoxycarbonylamino)-2,2-difluoro- propy 1 ] -4-chl oro-py razol e-3 , 5 -di carb oxy 1 ate (91 b)
[00753] To a solution of diethyl 4-chloro-lH-pyrazole-3,5-dicarboxylate (1 g, 4.05 mmol) and PPh3 (2.12 g, 8.11 mmol) in THF (5 mL), was added tert-butyl N-(2,2-difluoro-3 -hydroxy - propyl)carbamate (91a, 1.71 g, 8.11 mmol) and BTB AD (1867.1 mg, 8.11 mmol) , and the reaction mixture was stirred at 120 °C for Ih in microwave. The mixture was concentrated to afford a crude product diethyl l-[3-(tert-butoxycarbonylamino)-2,2-difluoro-propyl]-4-chloro-pyrazole-3,5- dicarboxylate (91b, 1.50 g) as a yellow oil. LCMS calcld for C17H25CIF2N3O6 (M+H)+ m/z = 440.1, found: 384.0 (-tBu).
[00754] Step 3. Preparation of diethyl l-(3-amino-2,2-difluoro-propyl)-4-chloro-pyrazole- 3,5-dicarboxylate; hydrochloride (91c)
[00755] To a solution of diethyl l-[3-(tert-butoxycarbonylamino)-2,2-difluoro-propyl]-4- chloro-pyrazole-3,5-dicarboxylate (91b, 3 g, 6.82 mmol) in 1,4-dioxane (45 mL), was added HCl/di oxane (4713.42 mg, 34.1 mmol), and the reaction mixture was stirred at 25 °C for 2h. The mixture was concentrated to afford the crude product diethyl l-(3-amino-2,2-difluoro-propyl)-4- chloro-pyrazole-3,5-dicarboxylate;hydrochloride (91c, 2.50 g) as a yellow solid. LCMS calcld for C12H17CIF2N3O4 (M+H)+ m/z = 340.1, found:340.1.
[00756] Step 4. Preparation of N,N-diethylethanamine; ethyl 3-chloro-7,7-difluoro-4-oxo- 6,8-dihydro-5H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylate; hydrochloride (91d)
[00757] To a solution of diethyl l-(3-amino-2,2-difluoro-propyl)-4-chloro-pyrazole-3,5- dicarboxylate; hydrochloride (91c, 2.77 g, 7.36 mmol) in ethanol (42 mL), was added EtsN (20.54 mL, 147.18 mmol), and the reaction mixture was stirred at 70 °C for 24h. The mixture was concentrated and then purified by silica gel chromatography (eluted with CH3CN in H2O from 5.0% to 95%) to afford N,N-diethylethanamine;ethyl 3-chloro-7,7-difluoro-4-oxo-6,8-dihydro- 5H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylate;hydrochloride (91d, 1.80 g, 2.24 mmol, 30.40% yield) as a white solid. LCMS calcld for C10H11CIF2N3O3 (M+H)+ m/z = 294.6, found: 294.6.
[00758] Step 5. Preparation of ethyl 3-chloro-7,7-difluoro-4,5,6,8-tetrahydropyrazolo[l,5- a] [ 1 ,4]diazepine-2-carboxylate (91e)
[00759] To a solution of ethyl 3-chloro-7,7-difluoro-4-oxo-6,8-dihydro-5H-pyrazolo[l,5- a][l,4]diazepine-2-carboxylate (91d, 200 mg, 0.37 mmol) in THF (5mL), was added BH3 in SMe2 (55.42 mg, 1.46 mmol), and the reaction mixture was stirred at 60 °C for 3h. The mixture was diluted with CH3OH (5 mL), and then the mixture was stirred at 60 °C for 3h. The crude product was not purified. Ethyl 3-chloro-7,7-difluoro-4,5,6,8-tetrahydropyrazolo[l,5-a][l,4]diazepine-2- carboxylate (91e, 35 mg) was obtained as a colorless oil. LCMS calcld for CioHi3ClF2N302(M+H)+ m/z = 280.1, found: 280.1.
[00760] Step 6. Preparation of ethyl 3-chloro-5-(6-chloro-5-cyano-2-methylsulfanyl- pyrimidin-4-yl)-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylate (91f) [00761] To a solution of ethyl 3-chloro-7,7-difluoro-4,5,6,8-tetrahydropyrazolo[l,5- a][l,4]diazepine-2-carboxylate (200 mg, 0.72 mmol) in THF (10 mL), was added 4,6-dichloro-2- methylsulfanyl-pyrimidine-5-carbonitrile (91e, 157.39 mg, 0.72 mmol) and DIEA (138.63 mg, 1.07 mmol), and the reaction mixture was stirred at 40 °C for 3h . The mixture was concentrated to afford a crude product. The crude product was purified by silica gel chromatography/flash chromatography (eluted with CH3CN in H2O from 5.0% to 95%); ethyl 3-chloro-5-(6-chloro-5- cyano-2-methylsulfanyl-pyrimidin-4-yl)-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5- a][l,4]diazepine-2-carboxylate (91f, 270 mg, 0.583 mmol, 81.49% yield) was obtained as white solid. LCMS calcld for C16H15CI2F2N6O2S (M+H)+ m/z = 463.02, found: 463.02.
[00762] Step 7. Preparation of ethyl 5-[6-(2-amino-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2-methylsulfanyl-pyrimidin-4-yl]-
3-chloro-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylate (91g)
[00763] To a solution of ethyl 3-chloro-5-(6-chloro-5-cyano-2-methylsulfanyl-pyrimidin-
4-yl)-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylate (91f, 100 mg, 0.22 mmol) in 1,4-dioxane (ImL) was added DIEA (55.79 mg, 0.43 mmol) and 2-aminospiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile (44.31 mg, 0.22 mmol) at 25 °C. The mixture was stirred at 50 °C for 1 h. The mixture was concentrated, then purified by silica gel on chromatography (eluting with EtOAc in PE from 0 % to 50 %). The product ethyl 5-[6-(2- amino-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2- methylsulfanyl-pyrimidin-4-yl]-3-chloro-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5- a][l,4]diazepine-2-carboxylate (91g, 135 mg, 0.214 mmol, 98.95% yield) was obtained as a white solid. LCMS calcld for C26H25C1F2N9O2S2(M+H)+ m/z = 632.1, found: 632.0.
[00764] Step 8. Preparation of ethyl 5-[6-[2-(tert-butoxycarbonylamino)-3-cyano- spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl]-5-cyano-2-methylsulfanyl- pyrimidin-4-yl]-3-chl oro-7, 7-difluoro-6, 8-dihy dro-4H-pyrazolo[l, 5-a][l, 4]diazepine-2- carb oxy late (91h)
[00765] To a solution of ethyl 5-[6-(2-amino-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2-methylsulfanyl-pyrimidin-4-yl]- 3-chloro-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylate (91g, 135 mg, 0.21 mmol) in MeCN (1 mL) was added DMAP (2.61 mg, 0.02 mmol) and BOC2O (16.21 mg, 0.43 mmol) at 25 °C , The mixture was stirred at 25 °C for 1 h. The mixture was purified by silica gel on chromatography (eluting with EtOAc in PE from 0 % to 50 %). The mixture of product ethyl 5-[6-[2-(tert-butoxycarbonylamino)-3-cyano-spiro[5,6-
dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl]-5-cyano-2-methylsulfanyl-pyrimidin-4-yl]- 3-chloro-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylate (91h, 145 mg, 0.198 mmol, 92.72% yield). LCMS calcld for C31H33CIF2N9O4S2 (M+H)+ m/z = 732.2, found: 732.0.
[00766] Step 9. Preparation of 5-[6-[2-(tert-butoxycarbonylamino)-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl]-5-cyano-2-methylsulfanyl-pyrimidin-4-yl]- 3-chloro-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylic acid (91i) [00767] To a mixture of ethyl 5-[6-[2-(tert-butoxycarbonylamino)-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl]-5-cyano-2-methylsulfanyl-pyrimidin-4-yl]- 3-chloro-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylate
[00768] (91h, 128.2 mg, 0.16 mmol) in THF (1 mL) and Water (1 mL) was added
LiOH.EEO (10.31 mg, 0.25 mmol) and the mixture was stirred at 25 °C for Ih. The mixture was acidified with HC1 (1 mol/L in H2O) to PH=6, extracted with EtOAc (3 x lOmL), dried over Na2SO4, concentrated. The mixture was not purified and used in the next step directly. The mixture of 5-[6-[2-(tert-butoxycarbonylamino)-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'- azetidine]-l'-yl]-5-cyano-2-methylsulfanyl-pyrimidin-4-yl]-3-chloro-7,7-difluoro-6,8-dihydro- 4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylic acid (91i, 110 mg) was obtained as a white solid. LCMS calcld for C29H29CIF2N9O4S2 (M+H)+ m/z = 704.1, found:704.2.
[00769] Step 10. Preparation of tert-butyl N-[l'-[6-[3-chloro-2-(dimethylcarbamoyl)-7,7- difluoro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-methylsulfanyl- pyrimidin-4-yl]-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2- yl]carbamate (91j)
[00770] To a solution of 5-[6-[2-(tert-butoxycarbonylamino)-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl]-5-cyano-2-methylsulfanyl-pyrimidin-4-yl]- 3-chloro-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxylic acid (91i, 100 mg, 0.13 mmol) in DCM (2mL) was added Dimethylamine (0.01 mL, 0.15 mmol), DIEA (19.28 mg, 0.15 mmol) and HATU (56.73 mg, 0.15 mmol) at 25 °C , The mixture was stirred at 25 °C for 1 h. The mixture was extracted DCM (3 x lOmL), dried over Na2SO4, concentrated. The mixture was purified by silica gel (eluting with CH3OH in DCM from 0% to 10%). The mixture of tertbutyl N-[l'-[6-[3-chloro-2-(dimethylcarbamoyl)-7,7-difhioro-6,8-dihydro-4H-pyrazolo[l,5- a][l,4]diazepin-5-yl]-5-cyano-2-methylsulfanyl-pyrimidin-4-yl]-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2-yl]carbamate (91j, 100 mg, 0.120 mmol, 97% yield) was obtained as a white solid. LCMS calcld for C31H34CIF2N10O3S2 (M+H)+ m/z = 731.2, found: 731.1.
[00771] Step 11. Preparation of tert-butyl N-[l'-[6-[3-chloro-2-(dimethylcarbamoyl)-7,7- difluoro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-methylsulfmyl- pyrimidin-4-yl]-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2- yl]carbamate (91k)
[00772] To a solution of tert-butyl N-[l'-[6-[3-chloro-2-(dimethylcarbamoyl)-7,7-difluoro- 6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-methylsulfanyl-pyrimidin-4-yl]-3- cyano-spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2-yl]carbamate (91j, 30 mg, 0.04 mmol) in THF (1 mL) and water (1 mL) was added oxone (18.92 mg, 0.06 mmol) at 25 °C , The mixture was stirred at 25 °C for 4 h. The mixture was extracted with EtOAc (3 ml x3), dried over Na2SO4, concentrated. The mixture was not purified and used to next step directly. Tert-butyl N- [l'-[6-[3-chloro-2-(dimethylcarbamoyl)-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5- a][l,4]diazepin-5-yl]-5-cyano-2-methylsulfmyl-pyrimidin-4-yl]-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2-yl]carbamate (91k, 30 mg) was obtained as a white solid. LCMS calcld for C3iH34ClF2Nio04S2(M+H)+ m/z = 747.2, found:747.1.
[00773] Step 12. Preparation of tert-butyl N-[l'-[6-[3-chloro-2-(dimethylcarbamoyl)-7,7- difhioro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-[[(2R,8S)-2-fluoro- l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2-yl]carbamate (911)To a solution of tert-butyl N- [l'-[6-[3-chloro-2-(dimethylcarbamoyl)-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5- a][l,4]diazepin-5-yl]-5-cyano-2-methylsulfanyl-pyrimidin-4-yl]-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2-yl]carbamate (91k, 80 mg, 0.0 mmol) in THF (ImL) was added [(2R,8S)-2-fhioro-l,2,3,5,6,7-hexahydropyrrolizin-8-yl]methanol (6.39 mg, 0.04 mmol) at 0 °C , The mixture was added LiHDMS (0.04 mL, 0.04 mmol) at 0 °C for 1 h. The mixture was quenched with aqueous NH4CI and extracted with EtOAc (3 x 5mL), dried over Na2SO4 and concentrated. The mixture was purified by silica gel chromatography (eluting with MeOH in DCM from 0% to 10%). The mixture of tert-butyl N-[l'-[6-[3-chloro-2- (dimethylcarbamoyl)-7,7-difluoro-6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano- 2-[[(2R,8S)-2-fluoro- 1,2, 3,5,6, 7-hexahy dropyrrolizin-8-yl]methoxy]pyrimi din-4-yl]-3-cy ano- spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2-yl]carbamate (911, 85 mg, 0.101 mmol, 94% yield) was obtained as a white solid. LCMS calcld for C38Hj4ClF3NnO4S(M+H)+ m/z = 842.3, found: 842.4.
[00774] Step 13. Preparation of 5-[6-(2-amino-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3-chloro-7,7-difluoro-N,N-dimethyl-6,8-
dihydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxamide;2,2,2-trifluoroacetic acid (Compound 91)
[00775] To a solution of tert-butyl N-[T-[6-[3-chloro-2-(dimethylcarbamoyl)-7,7-difluoro- 6,8-dihydro-4H-pyrazolo[l,5-a][l,4]diazepin-5-yl]-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-2-yl]carbamate (911, 65 mg, 0.08 mmol) in DCM (1 mL) was added TFA (0.2 mL, 2.61 mmol) at 25 °C , The mixture was stirred at 25 °C for Ih. The mixture was concentrated. The mixture was purified by prep-HPLC (eluting with CH3CN (0.1%) in H2O from 5% to 95%). The product of 5-[6-(2-amino-3-cyano-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-l'-yl)-5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4-yl]-3-chloro-7,7-difluoro-N,N-dimethyl-6,8- dihydro-4H-pyrazolo[l,5-a][l,4]diazepine-2-carboxamide;2,2,2-trifluoroacetic acid (Compound 91, 6.75 mg, 0.008 mmol, 10.19% yield) was obtained as a white solid. LCMS calcld for C33H36C1F3NIIO2S(M+H)+ m/z = 742.2., found:742.1. 'H NMR (400 MHz, CD3OD) 5: 5.57 ( d, J= 52 Hz, IH), 4.95 (s, 2H), 4.88-4.67 (m, 2H), 4.82-4.82 (m, 2H), 4.75-4.69 (m, 2H), 4.61-4.56 (m, 2H), 4.45 (s, 2H), 3.96-3.83 (m, 3H), 3.50-3.39 (m, IH), 3.13-3.10 (m, 6H), 2.78- 2.69 (m, 4H), 2.68- 2.48 (m, 2H), 2.39- 2.25 (m, 3H), 2.19- 2.09 (m, IH).
[00776] Compound 92 2 -amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-[[(2S)-l-(2,2,2-trifluoroethyl)azetidin-2-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00777] Compound 92 was prepared similarly to that of Ex. 3. LCMS calcld for C27H31F3N9OS (M+H)+ m/z = 586.2, found: 586.3. 'H NMR (400 MHz, CD3OD) 5 4.64 - 4.42 (m, 3H), 4.40 - 4.17 (m, 5H), 3.78 - 3.66 (m, IH), 3.57 - 3.45 (m, 3H), 3.45 - 3.32 (m, IH), 3.26 (m, 2H), 3.18 - 2.99 (m, 2H), 2.80 - 2.68 (m, 4H), 2.22 - 2.03 (m, 2H), 1.88 - 1.75 (m, 4H).
[00778] Compound 93. 5-[6-(2-amino-3-cyano-spiro[5,6-dihydrocyclopenta[b]thiophene- 4,3'-azetidine]-l'-yl)-5-cyano-2-[(6-methylene-2,3,5,7-tetrahydro-lH-pyrrolizin-8- yl)methoxy]pyrimidin-4-yl]-3-chloro-N,N-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5- a] [ 1 ,4]diazepine-2-carboxamide
[00779] Compound 93 was prepared similarly to that of Ex. 2. LCMS calculated for C34H39CIN11O2S (M+H)+ m/z =700.3; found: 700.0/702.0. ‘HNMR (400 MHz, CD3OD) 5 5.08 (s, 2H), 4.97 (s, 3H), 4.42 - 4.67 (m, 6H), 4.16 (m, 5H), 3.71 (d, J= 14.8 Hz, 1H), 3.10 - 3.16 (m, 1H), 3.08 (s, 6H), 2.65 - 2.79 (m, 6H), 2.43 (d, J= 15.6 Hz, 1H), 2.15 - 2.28 (m, 2H), 1.73 - 2.10 (m, 5H)
[00780] Compound 94. 2-amino-l'-[5-cyano-2-[(6-methylene-2,3,5,7-tetrahydro-lH- pyrrolizin-8-yl)methoxy]-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00781] Compound 94 was prepared similarly to that of Ex. 3. LCMS calculated for C30H36N9OS (M+H)+ m/z = 570.3, found: 570.3. 'HNMR (400 MHz, CD3OD) 54.96 (s, 2H), 4.43 (m, 6H), 4.02 - 4.11 (m, 2H), 3.65 (d, J = 14.0 Hz, 1H), 3.53 (s, 2H), 3.18 - 3.30 (m, 3H), 3.05 - 3.13 (m, 1H), 2.60 - 2.82 (m, 6H), 2.41 (d, J = 15.6 Hz, 1H), 1.72 - 2.11 (m, 8H).
[00782] Compound 95. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-[[l-[(l,l-dioxo-l,4-thiazinan-4-yl)methyl]cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00783] Compound 95 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C30H38N9O3S2 (M+H)+ m/z = 636.3, found: 636.1. ‘HNMR (400 MHz, CD3OD) 54.55 - 4.15
(m, 10H), 3.49 - 3.10 (m, 8H), 2.84 - 2.75 (m, 6H), 2.09 (s, 4H), 1.33 - 1.28 (d, 2H), 0.76 (s, 2H), 0.61 (s, 2H).
[00784] Compound 96. 5-[6-(2-amino-3-cyano-spiro[6,7-dihydro-5H-benzothiophene-
4,3'-azetidine]-l'-yl)-5-cyano-2-[[l-[(dimethylamino)methyl]-2,2-difluoro- cyclopropyl]methoxy]pyrimidin-4-yl]-3-chloro-N,N-dimethyl-4,6,7,8-tetrahydropyrazolo[l,5- a] [ 1 ,4]diazepine-2-carboxamide
[00785] Compound 96 was prepared similarly to that of Ex. 2. LCMS calculated for C33H39CIF2N11O2S (M+H)+ m/z =726.3, found: 726.3/728.3. 'H NMR (400 MHz, CD3OD) 5 5.11 - 5.26 (m, 1H), 5.01 (m, 1H), 4.59 (s, 6H), 4.47 (m, 4H), 4.36 (m, 1H), 3.08 (d, J= 3.2 Hz, 6H),
2.75 (s, 1H), 2.49 (t, J = 6.0 Hz, 2H), 2.31 - 2.41 (m, 1H), 2.20 - 2.28 (m, 8H), 2.08 - 2.14 (m, 2H), 1.76 - 1.85 (m, 2H), 1.53 - 1.62 (m, 1H), 1.27 - 1.39 (m, 2H).
[00786] Compound 97. 2-amino-l'-[5-cyano-2-[(2-methylpyrazol-3-yl)methoxy]-6- [(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00787] Compound 97 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C26H29N10OS (M+H)+ m/z = 529.2, found: 529.6. *HNMR (400 MHz, CD3OD) 5 7.41 (d, J= 1.8 Hz, 1H), 6.38 (d, J= 1.8 Hz, 1H), 5.43 (s, 2H), 4.60 (m, 5H), 4.34 (s, 1H), 4.11 (m, 2H), 3.89 (m, 3H), 3.51 (s, 2H), 2.77 (dd, J= 11.6, 8.0 Hz, 4H), 2.07 (s, 4H).
[00788] Compound 98. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-[[l-(lH-pyrazol-5-yl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00789] Compound 98 was prepared similarly to that of Ex. 3. LCMS calcld for C28H31N10OS (M+H)+ m/z = 555.2, found: 555.6. 'H NMR (400 MHz, CD3OD) 5 7.47 (s, 1H), 6.18 (s, 1H), 4.39 (m, 8H), 3.55 (s, 2H), 3.25 (m, 2H), 2.79 - 2.67 (m, 4H), 1.85 - 1.72 (m, 4H), 1.05 (s, 4H).
[00790] Compound 99. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-[[(2R,8S)-2-fluoro- 1,2, 3,5,6, 7-hexahydropyrrolizin-8-yl]methoxy]pyrimidin-4- yl]spiro[4a,5,5a,6-tetrahydrocyclopropa[f]benzothiophene-4,3'-azetidine]-3-carbonitrile
[00791] Compound 99 was prepared similarly to that of Ex. 3 as a TFA salt using Intermediate 21. LCMS calcld for C31H37FN9OS (M+H)+ m/z =602.3, found: 602.3. 1HNMR (4OO MHz, CD3OD) 5: 5.54 (d, .7=52 Hz, 1H), 5.03-4.99 ( m, 1H), 4.75-4.28 ( m, 7H), 4.16 ( s, 2H), 3.97-3.80 ( m, 3H), 3.63-3.40 (m, 3H), 2.91-2.80 (m, 2H), 2.71-2.49 (m, 2H), 2.41-2.27 (m, 3H), 2.18-2.03 (m, 5H), 1.71-1.66 (m, 1H), 1.55-1.51 ( m, 1H), 0.80-0.74 (m, 1H), 0.19-0.16 (m, 1H).
[00792] Compound 100. 2 -amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-[[l-(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]spiro[6H-thieno[2,3- c]thiophene-4,3'-azetidine]-3-carbonitrile
[00793] Compound 100 was prepared similarly to that of Ex. 3 as a formate salt using
Intermediate 19.
[00794] LCMS calcld for C29H36N9O2S2 (M+H)+ m/z =606.2, found: 606.2. 1 H NMR (400
MHz, CD3OD) 5 4.84 - 4.30 (m, 6H), 4.25 (s, 2H), 4.10 (s, 2H), 4.07 (s, 2H), 3.71 (s, 4H), 3.58 - 3.38 (m, 2H), 2.66 (s, 4H), 2.56 (s, 2H), 2.06 (s, 4H), 0.69 (s, 2H), 0.53 (s, 2H).
[00795] Compound 101. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-[[l-(morpholinomethyl)cyclopropyl]methoxy]pyrimidin-4-yl]-5-fluoro-spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00796] Compound 101 was prepared similarly to that of Ex. 3 using Intermediate 20. LCMS calculated for C30H37FN9O2S (M+H)+ m/z = 606.6; found: 606.5. 1H NMR (400 MHz, CD3OD) 5 5.60 (d, J= 52.3 Hz, 1H), 4.27 - 4.55 (m, 6H), 4.24 (s, 2H), 3.66 (m, 4H), 3.54 (s, 2H), 3.14 - 3.29 (m, 3H), 2.89 (m, 1H), 2.47 (s, 4H), 2.37 (s, 2H), 1.75 - 1.85 (m, 4H), 0.64 (m, 2H), 0.44 (m, 2H).
[00797] Compound 102. 2 -amino-l'-[2-[(4-bromo-lH-pyrazol-3-yl)methoxy]-5-cyano-6- [(lS,5R)-3,8-diazabicyclo[3.2.1]octan-3-yl]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00798] Compound 102 was prepared similarly to that of Ex. 3 as a TFA and formate salt. LCMS cal cl d for C25H26BrNioOS (M+H)+ m/z = 593.2, found: 593.2. 'H NMR (400 MHz, CD3OD) 5 8.53 - 8.45 (m, 1H), 7.79 - 7.60 (m, 1H), 5.34 (s, 2H), 4.74 - 4.18 (m, 6H), 4.15 - 3.98 (m, 2H), 3.60 - 3.38 (m, 2H), 2.82 - 2.69 (m, 4H), 2.03 (s, 4H).
[00799] Compound 103. 2 -amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-(2H-triazol-4-ylmethoxy)pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'- azetidine]-3-carbonitrile
[00800] Compound 103 was prepared similarly to that of Ex. 3 as a formate salt. LCMS calcld for C24H26N11OS (M+H)+ m/z = 516.5, found: 516.5. 'H NMR (DMSO-d6, 400 MHz) 5 8.34 (s, 2H), 7.90 (s, 1H), 7.23 (s, 2H), 5.33 (s, 2H), 4.19 (d, J= 12.0 Hz, 2H), 3.48 (s, 3H), 3.17 (d, J= 12.0 Hz, 2H), 2.68 (s, 4H), 1.62-1.56 (m, 4H).
[00801] Compound 104. 2 -amino-l'-[2-[(4-chloro-lH-pyrazol-3-yl)methoxy]-5-cyano-6- [(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00802] Compound 104 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C25H26CIN10OS (M+H)+ m/z = 549.1, found: 549.2. *H NMR (400 MHz, CD3OD) 5 7.68 (s, 1H), 5.35 (s, 2H), 4.76 - 4.21 (m, 6H), 4.14 (s, 2H), 3.60 - 3.38 (m, 2H), 2.82 - 2.67 (m, 4H), 2.08 (s, 4H).
[00803] Compound 105. 2-amino-l'-[2-[(5-chloro-lH-pyrazol-3-yl)methoxy]-5-cyano-6- [(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3-yl]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00804] Compound 105 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C25H26CIN10OS (M+H)+ m/z = 549.2, found: 549.3. *H NMR (400 MHz, CD3OD) 5 6.31 (s,
1H), 5.34 (s, 2H), 4.27 (s, 2H), 4.75-4.18 (m, 6H), 4.14 (s, 2H), 3.58-3.38 (m, 2H), 2.83-2.68 (m, 4H), 2.05 (s, 4H).
[00805] Compound 106. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-[[4-(trifluoromethyl)-lH-pyrazol-3-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00806] Compound 106 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C26H26F3N10OS (M+H)+ m/z = 583.2, found: 583.2. 'H NMR (400 MHz, CD3OD) 5 8.05 (s, 1H), 5.42 (s, 2H), 4.58 (s, 5H), 4.27 (s, 1H), 4.13 (s, 2H), 3.48 (s, 2H), 2.76 (dt, J= 12.0, 6.8 Hz, 4H), 2.06 (s, 4H).
[00807] Compound 107 2 -amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-[(5-methyl-lH-l,2,4-triazol-3-yl)methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00808] Compound 107 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C25H28N11OS (M+H)+ m/z = 530.3, found: 530.4. XH NMR (400 MHz, CD3OD) 5 5.35 (s, 2H), 4.75 - 4.18 (m, 6H), 4.17 - 4.09 (m, 2H), 3.56 - 3.42 (m, 2H), 2.85 - 2.66 (m, 4H), 2.43 (s, 3H), 2.12 - 1.97 (m, 4H).
[00809] Compound 108. 2-amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-[[4-(trifluoromethyl)-lH-pyrazol-3-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00810] Compound 108 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C25H28N11OS (M+H)+ m/z = 530.6, found: 530.3. 'HNMR (400 MHz, CD3OD) 5 5.43 (s, 2H), 4.74 - 4.20 (m, 6H), 4.14 (s, 2H), 3.61 - 3.37 (m, 2H), 2.85 - 2.66 (m, 4H), 2.36 (s, 3H), 2.07 (s, 4H).
[00811] Compound 109. 2 -amino-l'-[5-cyano-6-[(lR,5S)-3,8-diazabicyclo[3.2.1]octan-3- yl]-2-[[4-(difluoromethyl)-lH-pyrazol-3-yl]methoxy]pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00812] Compound 109 was prepared similarly to that of Ex. 3 as a TFA salt. LCMS calcld for C26H27F2N10OS (M+H)+ m/z = 565.2, found: 565.2 H NMR (400 MHz, CD3OD) 5 7.86 (s, 1H), 6.92 (t, J= 55.9 Hz, 1H), 5.44 (s, 2H), 4.56 (s, 6H), 4.12 (s, 2H), 3.60 - 3.36 (m, 2H), 2.84 - 2.68 (m, 4H), 2.04 (s, 4H).
[00813] Compound 110. 2-amino-l'-[5-cyano-2-[[(2R,8S)-2-fluoro-l,2,3,5,6,7- hexahydropyrrolizin-8-yl]methoxy]-6-(l,4-oxazepan-4-yl)pyrimidin-4-yl]spiro[5,6- dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3 -carbonitrile
[00814] Compound 110 was prepared similarly to that of Ex. 3. LCMS calculated for C28H34FN8O2S (M+H)+ m/z = 565.24; found: 565.3. ‘HNMR (400 MHz, CD3OD) 5 5.25 (d, J = 53.6 Hz, 1H), 4.20 - 4.59 (m, 4H), 3.94 - 4.15 (m, 6H), 3.82 - 3.88 (m, 2H), 3.70 - 3.78 (m, 2H), 3.10 - 3.24 (m, 3H), 2.92 - 3.02 (m, 1H), 2.69 - 2.81 (m, 4H), 1.77 - 2.28 (m, 8H).
[00815] Compound 111. 2-amino-l'-[5-cyano-6-(3,5-dihydro-2H-pyrido[3,2- f][ l,4]thiazepin-4-yl)-2-[[(2R, 8S)-2-fluoro- 1,2, 3,5,6, 7-hexahy dropyrrolizin-8- yl]methoxy]pyrimidin-4-yl]spiro[5,6-dihydrocyclopenta[b]thiophene-4,3'-azetidine]-3- carbonitrile
[00816] Compound 111 was prepared similarly to that of Ex. 3 using intermediate 22. LCMS calculated for C31H33FN9OS2 (M+H)+ m/z =630.22; found:630.3. ' H NMR (400 MHz, DMSO) 5 8.23 (dd, = 4.7, 1.6 Hz, 1H), 7.70 (dd, J= 7.6, 1.4 Hz, 1H), 7.22 (s, 2H), 7.13 (dd, J = 7.6, 4.7 Hz, 1H), 5.21 (d, J= 54.4 Hz, 1H), 4.88 (s, 2H), 4.42 (s, 2H), 4.18 (tm, 3H), 3.65 (m, 2H), 3.45 (s, 2H), 3.03 (m, 2H), 2.93 (m, 1H), 2.82 - 2.74 (m, 1H), 2.66 (s, 4H), 1.98 (s, 1H), 1.94 - 1.87 (m, 1H), 1.86 - 1.75 (m, 2H), 1.68 (m, 2H).
Example 8: Nucleotide Exchange Assay:
[00817] Ras proteins cycle between an active, GTP bound state, and an inactive GDP-bound state. This activity is tightly regulated by GTPase activating proteins (GAPs) and guanine nucleotide exchange factors (GEFs). GEFs, such as SOS 1/2, activate Ras proteins by exchanging GDP for GTP, thus returning Ras to its active conformation (Simanshu, Nissley, & McCormick, 2017). Therefore, a small molecule that binds K-Ras in a manner that prevents SOS-mediated nucleotide exchange locks KRas in its inactive state. Homogenous time resolved fluorescence (HTRF) was used to detect SOS-mediated binding of a fluorescent GTP analog, GTP-DY-647P1 (Jena Biosciences NU-820-647P1) to GST-tagged KRAS-G12D (2-169, Reaction Biology, MSC- 11-539) or to GST-tagged KRAS-G12V (2-169, Reaction Biology, MSC-11-540).
[00818] GST-tagged KRAS-G12D (2-169) and anti-GST MAb Tb Cryptate Gold (CisBio 61GSTTLB) were diluted into assay buffer (20 mM HEPES, pH 7.3, 150 mMNaCl, 5 mM MgC12, 0.05% BSA 0.0025% NP40, 1 mM DTT) to prepare a 2.5X donor solution. 5X compound was added to the protein mixture and incubated for 1 h at RT. 2.5X acceptor solution containing SOSlcat (564-1049, Reaction Biology MSC-11-502) and GTP-DY-647P1 were then added to the donor KRAS mixture such that the final concentration of the reaction contained 5 nM GST-tagged KRAS-G12D (2-169), 20 nM SOScat, and 150 nM GTP. The reaction was monitored using at RT with the Envision multimode plate reader (Ex/Em 337/665, 620 nM) up to 90 minutes at 5 minute
intervals. To monitor KRAS-G12V Sos-mediated nucleotide exchange, 80 nM SOS was added to reaction instead of 20 nM. All other components were the same as previously described. Data was blanked to reactions without S0S1 and % inhibition was calculated such that DMSO only = 0% and blank = 100%. Curve fitting was done using a 4 parameter fit. Reported IC50 values were extracted at 30 min and 90 min for the KRAS-G12D and KRAS-G12V assays, respectively. [00819] Curve fitting was done using a 4 parameter fit. NEA KRAS G12D IC50 (uM) values of selected compounds are depicted in Table 2 with compounds having a value <0.01 uM as ++++; > 0.01 uM to 0.1 uM as +++; > 0.1 uM to 1 uM as ++; > 1 uM to 20 uM as +; and >20 uM as NA.
Example 9: Protein Constructs for Protein-Protein Interaction:
Example 10. Recombinant Protein Production:
[00820] Biotinylated KRAS wt and KRAS G12D/V proteins were expressed and purified in conditions similar to those previously reported (Tran, et al., 2021 ) (Zhang, et al., 2020). Briefly, KRAS (1-169) proteins were expressed in E. coli at 18°C with an upstream TEV cleavage site (ENLFYQS) followed an Avi tag sequence (GLNDIFEAQKIEWHE). KRAS expression
constructs contained both a His6 and maltose-binding protein (MBP) tags at the N-terminus for Ni-NTA column purification prior to overnight TEV cleavage and MBP column purification. The avi-tagged NBAS expression construct contained both a His6 tag and SUMO cleavage sige at the N-terminus for Ni-NTA column purification followed by His-ULPl digestion overnight. All avi-tagged RAS proteins were dialyzed into buffer containing ATP, biotin, and BirA followed by purification over a second Ni-NTA column and then run over a size exclusion HiLoad™ 26/600 Superdex™ column in 20 mM HEPES, pH 7.5, 300 mM NaCl, 5 mM MgCh, and 1 mM TCEP. Fractions containing the protein of interest were pooled, concentrated, and confirmed by intact mass spectrometry. To prepare ‘GTP’ loaded KRAS and NRAS, biotinylated KRAS or NRAS was nucleotide exchanged from GDP -bound protein to GppNHp-bound (Jena Biosciences, NU-401-50) protein in the presence of alkaline phosphatase and excess GppNHp as previously described and the resulting nucleotide content was confirmed by HPLC reverse phase analytical chromatography (Donohue, et al., 2019) (Tran, et al., 2021 ).
[00821] Elis-tagged RAFI (52-131) was similarly expressed in E. coli at 18°C overnight with an upstream TEV cleavage site. His-tagged RAFI expression construct contained both a His6 and MBP tags at the N-terminus for Ni-NTA column purification followed by MBP-tagged TEV digestion overnight. RAFI protein samples were further purified over a MBP column followed by a Ni-NTA column and a second MBP column. The fractions containing the protein of interest were pooled, concentrated, and further purified over a HiLoad™ 16/600 Superdex™ 75 pg size exclusion column into 20mM HEPES, pH8.0, 200mM NaCl, 5mM TCEP.
Example 11: Protein-Protein Interaction (PPI) Assay:
[00822] When RAS proteins are in the active GTP -bound conformation, they bind the effector protein RAFI at the N-terminus Ras-binding domain (RBD, residues 52-131) (Tran, et al., 2021). Homogenous time resolved fluorescence (HTRF) was used to monitor the interaction between wt or mutant KRAS and RAFI or wt NRAS and RAFI. Compounds were assayed in the presence of KRAS G12D/V and RAFI versus wt KRAS to assess activity against mutant and w.t. KRAS. Similarly, compounds were then assayed in the presence of w.t. NRAS and RAFI to assess RAS isoform selectivity. In all assay formats, His-tagged RAFI protein was incubated with the HTRF donor, anti-6His Tb Cryptate gold (Cisbio 61DB10RDF), and biotinylated RAS proteins were incubated with the HTRF acceptor, streptavidin-d2 (CisBio 610SADLA). The intensity of the fluorescence signal emitted is proportional to binding between the two proteins. The donor solution was prepared by mixing 16 nM His-tagged RAFI in protein dilution buffer with 1 : 100 anti-6His Tb cryptate in PPI-Terbium detection buffer. 16 nM biotinylated RAS protein was diluted into protein dilution buffer (50 mM Tris-HCl pH 7.5, 150 mM NaCl, 0.1 mM
MgC12, 1 mM TCEP, 0.005% Tween20) and mixed with 1 :2000 Streptavidin-d2 diluted in PPI- Terbium detection buffer (CisBio 61DB10RDF). 50X compound in DMSO was mixed with 16 nM KRAS-acceptor solution and incubated for 30 minutes at room temperature. After compound pre-incub ati on with KRAS, the RAFI donor solution was added to the KRAS-acceptor solution and incubated for 1 hour at room temperature. The fluorescence signal emitted was monitored at 665 nm and 615 nm using an Envision multimode plate reader. The HTRF ratio (665/615) was calculated and normalized to 0% inhibition in the absence of compound and 100% inhibition in the presence of untagged RAFI protein. PPI KRAS G12D/RAF1, KRAS G12V/RAF1, w.t.KRAS/RAFl and NRAS/RAF1 IC50 (uM) values of selected compounds are depicted in Table 3 with compounds having a value <0.1 uM as ++++; > 0.1 uM to 1 uM as +++; > 1 uM to 10 uM as ++; > 10 uM to 100 uM as +; and >100 uM as NA.
[00823] Compounds described herein are active against KRAS G12 mutant and other alleles representative by PPI-G12D, PPI-G12V and PPI-w.t.KRAS potency for broad activity against mutant KRAS and wtKRAS amplification driven malignancies. Compounds described herein are selective for the KRAS isoform representative by lack of activity in the PPI-NRAS assay.
Example 12. pERK Inhibition cellular HTRF assay in AGS Cell Lines (Method A) [00824] The Phospho-ERK cellular HTRF assay measures ERK protein phosphorylated at Thr202/Tyr204 as a readout of MAPK pathway activation (Cisbio 64ERKPEH). AGS cells (ATCC CRL-1739) are cultured in the complete medium containing 10% fetal bovine serum and lx Penicillin/Streptomycin at 37°C in a humid atmosphere of 5% CO2 in the air (AGS cells: RPMI 1640 medium).
[00825] On day 1, the cells are plated in tissue-culture treated 96-well plates at the specified densities and allowed to attach for overnight (AGS: 30,000 cells/well). On day 2, the cells are treated with the serially diluted compound solutions in a final concentration of 0.5 % DMSO. After the treatment for the specified time (AGS cells: 3 hours), the supernatant is removed, and the cells are lysed by the lysis buffer supplied with the kit. Then, the cell lysates are treated with the detection reagents overnight at 4°C in darkness. On day 3, the fluorescence intensities at the wavelengths 665 and 620 nm are measured by the Envision plate reader (Perkin Elmer). The data are processed and fitted to a 4-parameter logistic model for IC50 calculations (GraphPad Prism 9).
Example 13. pERK In Cell Western (ICW) assay (Method B) [00826] pERK ICW is a high throughput screening assay to evaluate the cellular potency of mutant KRAS small molecule inhibitors. KRAS mutant cell lines AGS and GP2D (KRASG12D)
were purchased from ATCC and maintained respectively in RPMI and DMEM medium supplemented with 10% fetal bovine serum and Penicillin/Streptomycin.
[00827] Cells grown in exponential phase were trypsinized, resuspended in fresh media, and viable cells were counted using a cell counter with Trypan Blue (BioRad TC20). Cells were seeded into 384-well plate (Greiner 781091) at density of 5,000 cells/well for AGS and 10,000 cells/well for GP2D and allowed to grow overnight in a 37°C CO2 incubator. The next day, compounds were dispensed into wells with a ’A log, 10-point serial dilution and top concentration of 10 pM using Tecan D300e dispenser and incubated for 3 hours in a 37 °C CO2 incubator. Cells were then fixed with paraformaldehyde (Electron Microscopy Sciences, 15710, 4% final concentration) for 30 min, permeabilized with wash buffer (IX PBS + 0.1% Triton X-100) for 30 min and blocked with Odyssey blocking buffer (Li-COR 927-70001) for 1 hour, all at room temperature (RT). Phospho- ERK antibody (CST 4370L) was diluted 1 :500 in Odyssey blocking + 0.2% Tween 20 and incubated with cells overnight at 4 °C. The next day, plates were washed 5x with wash buffer, incubated with IRDye 800 CW, Goat anti -Rabbit secondary antibody (Li-COR 926-32211, 1 :500) and DRAQ5 (CST 4084L, 1 :5,000) diluted in in Odyssey blocking + 0.2% Tween 20 for 1 hour, washed 5x, and imaged on an Odyssey CLx imaging system.
[00828] For data analysis, signal intensities from 800 (phosphor-ERK) and 700 (DRAQ5) channels were extracted, and phospho-ERK signals were normalized to DRAQ5 signals for each well and percent of DMSO control values were computed. Data were then imported into Graphpad Prism to compute half-maximal inhibitory concentrations (IC50) using a 4-parameter variable slope model. Z-factor for each plate was computed from signals derived from wells treated with either DMSO or 5 pM of Trametinib. AGS pERK ICW (Method B) IC50 (uM) values of selected compounds are depicted in Table 2 with compounds having a value 0.001 uM to 0.01 uM as ++++; > 0.01 uM to 0.1 uM as +++; > 0.1 uM to 1 uM as ++; 1 uM to 10 uM as + and > 10 uM as NA.
[00829] Table 2 includes NEA KRAS G12D IC50 (uM) values (<0.01 uM as ++++; > 0.01 uM to 0.1 uM as +++; > 0.1 uM to 1 uM as ++; > 1 uM to 20 uM as +; and >20 uM as NA), PPI KRAS G12D/RAF1 IC50 (uM) values (<0.1 uM as ++++; > 0.1 uM to 1 uM as +++; > 1 uM to 10 uM as ++; > 10 uM to 100 uM as +; and >100 uM as NA), AGS pERK HTRF (Method A) IC5o (uM) values (<0.01 uM as ++++; > 0.01 uM to 0.1 uM as +++; 0.1 uM to 1 uM as ++; 1 uM to 20 uM as + and >20 uM as NA), and AGS pERK ICW (Method B) IC50 (uM) values (0.001 uM to 0.01 uM as ++++; > 0.01 uM to 0.1 uM as +++; > 0.1 uM to 1 uM as ++; 1 uM to 10 uM as + and > 10 uM as NA) of selected compounds. ND indicates not determined.
Table ) values for various assays
[00830] Table 3 includes KRASG12V/RAF1, wtKRAS/RAFl and wtNRAS/RAFl PPI IC50 (uM) values of selected compounds; with compounds having a value <0.1 uM as ++++; 0.1 uM to 1 uM as +++; >1 uM to 10 uM as ++; >10 uM to 100 uM as +; and >100 uM as NA.
[00831] Table 4 includes NEA KRAS G12D IC5o (uM) values (<0.01 uM as ++++; > 0.01 uM to 0.1 uM as +++; > 0.1 uM to 1 uM as ++; > 1 uM to 20 uM as +; and >20 uM as NA), PPI KRAS G12D/RAF1 IC50 (uM) values (<0.1 uM as ++++; > 0.1 uM to 1 uM as +++; > 1 uM to 10 uM as ++; > 10 uM to 100 uM as +; and >100 uM as NA), AGS and GP2D pERK ICW IC50 (uM) values (0.001 uM to 0.01 uM as ++++; > 0.01 uM to 0.1 uM as +++; > 0.1 uM to 1 uM as ++; 1 uM to 10 uM as + and > 10 uM as NA) of selected compounds. ND indicates not determined.
Example 14. Quantification of cytokine/chemokine release under treatment in
HP AC cells with a KRAS inhibitor
[00832] Previous studies have shown KRAS-mutation-mediated MAPK signaling activation in tumor cells leads to immunosuppressive tumor microenvironment partially by increasing tumor cell immunosuppressive cytokine (e.g. GM-CSF) while reducing immunostimulatory chemokine (e.g. CXCL10 and CCL20) release (reference article, Huang et al., 2021, KRAS mutation: from undruggable to druggable in cancer). This assay aims to determine whether mutant KRAS inhibitor treatment can reverse tumor cell cytokine/chemokine release by inhibiting KRAS/MAPK signaling.
[00833] Cytokine/chemokine levels in tumor cell-conditioned medium can be measured by ELISA (Enzyme-Linked Immunosorbent Assay) kit. Endpoint relative cell numbers are also measured by CTG (CellTiter-Glo) assay as cytokine/chemokine release is directly affected by the number of assayed tumor cells.
[00834] KRAS mutant cell line HP AC (KRASG12D) was purchased from ATCC (ATCC
CRL-2119) and maintained in DMEM medium supplemented with 10% fetal bovine serum and Penicillin/Streptomycin (complete DMEM medium, thereafter).
[00835] HP AC cells grown in exponential phase were trypsinized, resuspended in fresh media, and viable cells were counted using a cell counter with Trypan Blue (BioRad TC20). Cells were seeded into 6-well plate (Corning 3506) at density of 0.75 million cells/well in 1.5 mL complete DMEM medium and allowed to grow overnight in a 37°C CO2 incubator. The next day, the medium was replaced by the same volume of fresh complete DMEM medium with 500 nM of each compound (e.g., compound 2, 3, 4, 14, 25, or 74,) or DMSO control, triplet samples for each compound treatment or control. The cells were then cultured for another 48 hours in 37°C CO2 incubator.
[00836] By the end of drug treatment, 1 mL of supernatant from 1.2 mL conditioned medium of each sample after centrifugation (6000 rpm) was collected and analyzed by R&D Systems’ GM-CSF (R&D DGM00RD), CXCL10 (R&D DIP 100), and CCL20 (R&D DM3A00) standard curve-based ELISA kits following the manufacturers’ protocols. Fresh complete DMEM, not conditioned, was used as BLK control of the assay.
[00837] To determine relative cell numbers by the end of assay, cells were initially seeded in a 384-well plate in 40 pL complete DMEM medium at the same density (0.5 million cells/mL), treated with the same concentration of a compound (e.g., compound 2, 3, 4, 14, 25, or 74,) or control, and incubated the same time courses in parallel in 6-well plates.
Relative cell numbers were assayed by dispensing 40 pL of CellTiter-Glo (Promega G7573) reagent for luminance reading following manufacturers’ protocol.
[00838] For data analysis, corrected ELISA OD reading (OD450 subtracts OD570 of each sample) was used to determine cytokine/chemokine level using the formula generated from a standard curve of each ELISA assay. The cytokine/chemokine level was normalized by relative cell number determined by CTG assay of each sample.
[00839] FIG. 1 shows levels of immunosuppressive cytokine granulocyte-macrophage colony-stimulating factor (GM-CSF) (left chart) and immunostimulatory chemokines C-X-C motif chemokine ligand 10 (CXCL10) (middle chart) and Chemokine (C-C motif) ligand 20 (CCL20) (right chart) in KRAS G12D mutant HP AC cell conditioned medium were measured by each cytokine/chemokine specific Enzyme-Linked-Immunosorbent-Assay (ELISA), and normalized by tumor cell numbers of each sample. * p <0.05, ** p <0.01, *** p<0.001, **** p <0.0001.
[00840] As shown in FIG. 1, exemplified by compounds 2, 3, 4, 14, 25, and 74, compounds described herein may reprogram the tumor microenvironment (TME) in favor of antitumor immunity, by modulating tumor cell cytokine/chemokine release. Specifically, compound treatment reduces immunosuppressive cytokine release while increasing immunostimulatory chemokine release in KRAS mutant cells (refer to histograms in FIG. 1). These data support clinical studies combining compounds described herein (e.g., Formula (I), Formula (LA), etc.) with existing checkpoint inhibitors (inhibitors targeting: cytotoxic T lymphocyte associated antigen 4 (CTLA-4), e.g. Yervoy/lpilimumab; programmed death 1/programmed death-ligand 1 (PD-1/PD-L1), e.g. Keytruda/pembrolizumab; and lymphocyteactivation gene 3 (LAG-3), e.g. BMS-986016/relatlimab) for various types of cancers harboring KRAS G12D (e.g., tumors harboring KRAS G12D).
[00841] While preferred embodiments of the present invention have been shown and described herein, it will be obvious to those skilled in the art that such embodiments are provided by way of example only. Numerous variations, changes, and substitutions will now occur to those skilled in the art without departing from the invention. It should be understood that various alternatives to the embodiments of the invention described herein may be employed in practicing the invention. It is intended that the following claims define the scope of the invention and that methods and structures within the scope of these claims and their equivalents be covered thereby.
Claims
WHAT IS CLAIMED IS:
1. A method of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor and a compound of Formula (I):
Formula (I), or a pharmaceutically acceptable salt thereof wherein:
B is selected from a 7- to 15-membered heterocycle and C7-C15 carbocycle, wherein the 7- to 15- membered heterocycle and C7-C15 carbocycle are each optionally substituted with one or more substituents independently selected from halogen, -CN, -NO2, =0, -N(R21)2, -B(OR21)2, -OR21, -SR21, -S(O)2(R21), -S(O)2N(R21)2, -NR21S(O)2R21, -C(O)N(R21)2, -C(O)NR21OR21, - N(R21)C(O)R21, -N(R21)C(O)N(R21)2, -N(R21)C(O)OR21, -C(0)R21, C(0)0R21, -0C(0)R21, - OC(O)N(R21)2, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, C1-6 alkyl, C2-6 alkenyl, C2-e alkynyl, C3-C12 carbocycle and 5- to 12-membered heterocycle;
R1 is selected from hydrogen and 5- to 15-membered heterocycle, wherein the 5- to 15- membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(=NR20)N(R20)2, -C1-6 alkyl(=NOR20), - C(O)NR20OR20, -N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, - C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, -N02, =0, =N(R20), =NO(R20), -CN, - NHCN, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, C1-6 alkyl-SO2R20, C1-6 alkoxyalkyl, C1-6 alkyl, C2-6 alkenyl, C2-e alkynyl, C3- C12 carbocycle and 5- to 12-membered heterocycle, wherein the C3-C12 carbocycle and 5- to 12-membered heterocycle are each optionally substituted independently with one or more R1*; each R1* is independently selected from halogen, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -
C(O)NR20OR20, -N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, - C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, -NO2, =0, =N(R20), =NO(R20), -CN, - NHCN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-e alkynyl, and Cs-Ci2 carbocycle;
Y is selected from a bond, -O-, -S-, and -N(R5)-;
R2 is selected from heterocycle, aryl, Ci-Ce alkyl, -L-heterocycle, -L-N(R23)2, -L-OR23, -L-aryl, - L-heteroaryl, -L-cycloalkyl, -L-NHC(=NH)NH2, -L-C(O)N(R23)2, -L-Ci-Ce haloalkyl, -L- OR23, -L-NR23C(O)-aryl, -L-COOH, -L-NR23S(O)2(R23), -L-S(O)2N(R23)2, -L- N(R23)C(O)(OR23), -L-OC(O)N(R23)2, and -L-C(=O)OCI-C6 alkyl, wherein the heterocycle, the heterocycle portion of -L-heterocycle, and the cycloalkyl portion of the -L-cycloalkyl are each optionally substituted with one or more R6, and wherein the aryl, the aryl portion of -L- NR23C(O)-aryl, the aryl portion of -L-NR23C(O)-aryl, the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R7;
R3 is selected from hydrogen, halogen, -CN, -NO2, -N(R20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, - N(R20)C(O)R20, -N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, -C(O)R20, -C(O)OR20, -OC(O)R20, - OC(O)N(R20)2, CI-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-e alkynyl, Cs-Ci2 carbocycle and 5- to 12-membered heterocycle; each R4 is independently selected from halogen, -NO2, =0, =S, -CN, Ci-6 alkyl, C2-6 alkynyl, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 cyanoalkyl, Ci-e hydroxyalkyl, and Ci-6 haloalkyl; n is selected from 0, 1, 2, 3, and 4; each R5 is independently selected from hydrogen and Ci-Ce alkyl; each R6 is independently selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, oxo, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, =CH2, =NO-CI-C3 alkyl, C1-C3 aminoalkyl, - N(R5)S(O)2(R5), -Q-phenyl, -Q-phenylSO2F, -NHC(O)phenyl, - NHC(O)phenylSO2F, C1-C3 alkyl substituted pyrazolyl, -C1-C3 alkyl-N(R5)2, -C(O)N(R5)2, -C1-C3 alkyl-N(R5)2, - C(O)N(R5)2, tert-butyldimethylsilyloxyCH2-, -N(R5)2, (C1-C3 alkoxy)Ci-C3 alkyl-, (C1-C3 alkyl)C(=O), oxo, (C1-C3 haloalkyl)C(=O)-, -SO2F, (C1-C3 alkoxy)Ci-C3 alkoxy, - CH2OC(O)N(R5)2, -CH2NHC(O)OC1-C6 alkyl, -CH2NHC(O)N(R5)2, -CH2NHC(O)CI-C6 alkyl, -CH2(pyrazolyl), -CH2NHSO2Ci-Ce alkyl, -CH2OC(O)heterocycle, -OC(O)N(R5)2, - 0C(0)NH(CI-C3 alkyl)O(Ci-C3 alkyl), -0C(0)NH(Ci-C3 alkyl)O(Ci-C3 alkyl)phenyl(Ci-C3 alkyl)N(CH3)2, -0C(0)NH(Ci-C3 alkyl)O(Ci-C3 alkyl)phenyl, -OC(O)heterocycle, -O-C1-C3 alkyl, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, and -
CH2heterocycle, wherein the phenyl of -NHC(O)phenyl and -0C(0)NH(CI-C3 alkyl)(Ci-C3 alkyl)phenyl are optionally substituted with one or more substituents selected from -C(O)H and OH, and wherein the alkyl of -O-C1-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo and hydroxy; and wherein the heterocycle of - CH2heterocyclyl is optionally substituted with oxo; each Q is selected from a bond, S, and O; each R7 is independently selected from halogen, hydroxy, HC(=O)-, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 hydroxyalkyl, -C1-C3 alkyl-N(R5)2, -C(O)N(R5)2, and -N(R5)2; each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen, hydroxy, C1-6 alkoxy, C1-C4 hydroxyalkyl, C1-C4 alkyl, C3-C6 carbocycle, and 3- to 8-membered heterocycle, wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -NO2, =0, =S, -CN, C1-6 alkyl- N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle, wherein the C3-C6 carbocycle and 3- to 8- membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -N02, =0, =S, -CN, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, and C1-6 haloalkyl; each R20 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -N02, -NH2, - N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12- membered heterocycle; each R21 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -N02, -NH2, - N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12- membered heterocycle; each R23 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -N02, -NH2, - N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12- membered heterocycle.
2. The method of claim 1, wherein the immunomodulator inhibitor is a PD-1 inhibitor.
3. The method of claim 2, wherein the PD-1 inhibitor is an antibody or antigenbinding fragment.
4. The method of claim 2, wherein the PD-1 inhibitor is selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, and a biosimilar thereof.
5. The method of claim 2, wherein the PD-1 inhibitor is nivolumab or a biosimilar thereof.
6. The method of claims 4 or 5, wherein the therapeutically effective amount of nivolumab or biosimilar thereof in the combination is about 240 mg administered every two weeks.
7. The method of claims 4 or 5, wherein the therapeutically effective amount of nivolumab, or biosimilar thereof, in the combination is about 480 mg administered every four weeks.
8. The method of claim 4, wherein the PD-1 inhibitor is pembrolizumab or a biosimilar thereof.
9. The method of claim 4, wherein the therapeutically effective amount of pembrolizumab or biosimilar thereof in the combination is about 200 mg administered every three weeks.
10. The method of claim 4, wherein the wherein the PD-1 inhibitor is cemiplimab or a biosimilar thereof.
11. The method of claim 1, wherein the immunomodulator inhibitor is a PD-L1 inhibitor.
12. The method of claim 11, wherein the PD-L1 inhibitor is selected from the group consisting of atezolizumab, avelumab, durvalumab, and a biosimilar thereof.
13. The method of claim 12, wherein the PD-L1 inhibitor is atezolizumab or a biosimilar thereof.
14. The method of claim 1, wherein the immunomodulator inhibitor is a CTLA-4 inhibitor.
15. The method of any one of claims 1 to 14, wherein the immunomodulator inhibitor is administered orally.
16. The method of any one of claims 1 to 15, wherein the cancer is selected from: Cardiac: sarcoma (angiosarcoma, fibrosarcoma, rhabdomyosarcoma, liposarcoma), myxoma, rhabdomyoma, fibroma, lipoma and teratoma;
Lung: bronchogenic carcinoma (squamous cell, undifferentiated small cell, undifferentiated large cell, adenocarcinoma), alveolar (bronchiolar) carcinoma, bronchial adenoma, sarcoma, lymphoma, chondromatous hamartoma, mesothelioma;
Gastrointestinal: esophagus (squamous cell carcinoma, adenocarcinoma, leiomyosarcoma, lymphoma), stomach (carcinoma, lymphoma, leiomyosarcoma), pancreas (ductal adenocarcinoma, insulinoma, glucagonoma, gastrinoma, carcinoid tumors, vipoma), small bowel (adenocarcinoma, lymphoma, carcinoid tumors, Kaposi's sarcoma, leiomyoma, hemangioma, lipoma, neurofibroma, fibroma), large bowel (adenocarcinoma, tubular adenoma, villous adenoma, hamartoma, leiomyoma);
Genitourinary tract: kidney (adenocarcinoma, Wilm's tumor (nephroblastoma), lymphoma, leukemia), bladder and urethra (squamous cell carcinoma, transitional cell carcinoma, adenocarcinoma), prostate (adenocarcinoma, sarcoma), testis (seminoma, teratoma, embryonal carcinoma, teratocarcinoma, choriocarcinoma, sarcoma, interstitial cell carcinoma, fibroma, fibroadenoma, adenomatoid tumors, lipoma);
Liver: hepatoma (hepatocellular carcinoma), cholangiocarcinoma, hepatoblastoma, angiosarcoma, hepatocellular adenoma, hemangioma;
Biliary tract: gall bladder carcinoma, ampullary carcinoma, cholangiocarcinoma;
Bone: osteogenic sarcoma (osteosarcoma), fibrosarcoma, malignant fibrous histiocytoma, chondrosarcoma, Ewing's sarcoma, malignant lymphoma (reticulum cell sarcoma), multiple myeloma, malignant giant cell tumor chordoma, osteochronfroma (osteocartilaginous exostoses), benign chondroma, chondroblastoma, chondromyxofibroma, osteoid osteoma and giant cell tumors;
Nervous system: skull (osteoma, hemangioma, granuloma, xanthoma, osteitis deformans), meninges (meningioma, meningiosarcoma, gliomatosis), brain (astrocytoma, medulloblastoma, glioma, ependymoma, germinoma (pinealoma), glioblastoma multiform, oligodendroglioma, schwannoma, retinoblastoma, congenital tumors), spinal cord neurofibroma, meningioma, glioma, sarcoma);
Gynecological: uterus (endometrial carcinoma), cervix (cervical carcinoma, pre-tumor cervical dysplasia), ovaries (ovarian carcinoma (serous cystadenocarcinoma, mucinous cystadenocarcinoma, unclassified carcinoma), granulosa-thecal cell tumors, Sertoli-Leydig cell tumors, dysgerminoma, malignant teratoma), vulva (squamous cell carcinoma, intraepithelial carcinoma, adenocarcinoma, fibrosarcoma, melanoma), vagina (clear cell carcinoma, squamous cell carcinoma, botryoid sarcoma (embryonal rhabdomyosarcoma), fallopian tubes (carcinoma);
Hematologic: blood (myeloid leukemia (acute and chronic), acute lymphoblastic leukemia, chronic lymphocytic leukemia, myeloproliferative diseases, multiple myeloma,
myelodysplastic syndrome), Hodgkin's disease, non-Hodgkin's lymphoma (malignant lymphoma);
Skin: malignant melanoma, basal cell carcinoma, squamous cell carcinoma, Kaposi's sarcoma, moles dysplastic nevi, lipoma, angioma, dermatofibroma, keloids, psoriasis; and
Adrenal glands: neuroblastoma.
17. The method of any one of claims 1 to 16, wherein the cancer is non-small cell lung cancer, small cell lung cancer, colorectal cancer, rectal cancer or pancreatic cancer.
18. The method of any one of claims 1 to 16, wherein the cancer is non-small cell lung cancer.
19. The method of any one of claims 1 to 16, wherein the cancer is small cell lung cancer.
20. The method of any one of claims 1 to 16, wherein the cancer is colorectal cancer.
21. The method of any one of claims 1 to 16, wherein the cancer is rectal cancer.
22. The method of any one of claims 1 to 16, wherein the cancer is pancreatic cancer.
23. The method of any one of claims 1 to 16, wherein the cancer is a solid tumor cancer.
24. The method of any one of claims 1 to 16, wherein the cancer is selected from a KRas mutant-associated cancer.
25. The method of any one of claims 1 to 16, wherein the cancer is selected from a KRas wildtype-associated cancer.
26. The method of any one of claims 1 to 16, wherein the cancer is selected from a KRas G12D-associated cancer, a KRas G12V-associated cancer, and a KRas wildtype-associated cancer.
27. The method of any one of claims 1 to 16, wherein the cancer is a KRas G12D- associated cancer.
28. The method of any one of claims 1 to 16, wherein the cancer is a KRas G12V- associated cancer.
29. The method of any one of claims 25 to 26, wherein the KRas wildtype is hyperactivated.
30. The method of any one of claims 1 to 29, wherein the immunomodulator inhibitor synergistically increases the sensitivity of cancer cells to the compound or salt of Formula (I).
31. The method of claim 1, wherein the combination exhibits synergy.
32. The method of any one of claims 1 to 31, wherein the therapeutically effective amount of the combination of the immunomodulator inhibitor and the compound or salt of Formula (I) results in an increased duration of overall survival, an increased duration of
progression free survival, an increase in tumor growth regression, an increase in tumor growth inhibition, an increased duration of stable disease in the subjects relative to treatment with only the compound or salt of Formula (I), or any combination thereof.
33. The method of any one of claims 1 to 32, wherein the therapeutically effective amount of the compound or salt of Formula (I) of the combination is between about 0.01 to 100 mg/kg per day.
34. The method of any one of claims 1 to 33, wherein the therapeutically effective amount of the compound or salt of Formula (I) in the combination is between about 0.1 to 50 mg/kg per day.
35. The method of any one of claims 1 to 34, wherein the therapeutically effective amount of the immunomodulator inhibitor of the combination is between about 0.01 to 100 mg/kg per day.
36. The method of any one of claims 1 to 35, wherein the therapeutically effective amount of the immunomodulator inhibitor of the combination is between about 0.1 to 50 mg/kg per day.
37. The method of any one of claims 1 to 36, wherein the immunomodulator inhibitor and the compound or salt of Formula (I) are administered on different days.
38. The method of any one of claims 1 to 37, wherein the compound or salt of Formula (I) is administered at a maximum tolerated dose.
39. The method of any one of claims 1 to 38, wherein the immunomodulator inhibitor is administered at a maximum tolerated dose.
40. The method of any one of claims 1 to 39, wherein the immunomodulator inhibitor and the compound or salt of Formula (I) are each administered at a maximum tolerated dose.
41. The compound or salt of any one of claims 1 to 40, wherein B is selected from an optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle.
42. The compound or salt of any one of claims 1 to 41, wherein B is an optionally substituted 8- to 15-membered fused heterocycle.
43. The compound or salt of any one of claims 1 to 42, wherein B is an optionally substituted Cs-Cis fused carbocycle.
44. The compound or salt of any one of claims 1 to 43, wherein for B, the optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle are each independently bicyclic or tricyclic.
45. The compound or salt of any one of claims 1 to 44, wherein the heterocycle or carbocycle of B is bicyclic.
46. The compound or salt of any one of claims 1 to 45, wherein the heterocycle or carbocycle of B is tricyclic.
47. The compound or salt of any one of claims 1 to 46, wherein for B, the optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused
which is optionally substituted with one or more substituents.
48. The compound or salt of claim 47, wherein for B, the optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle are selected
optionally substituted with one or more substituents.
49. The compound or salt of any one of claims 1 to 48, wherein for B, the one or more optional substituents of the heterocycle and carbocycle are each independently selected from oxo, -NH2, halogen, and C1-C3 alkyl.
50. The compound or salt of any one of claims 1 to 49, wherein for B, the optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle are selected from
51. The compound or salt of any one of claims 1 to 46, wherein B is selected from an optionally substituted 5- to 15-membered heterocycle and optionally substituted C3-C15 carbocycle.
52. The compound or salt of claim 51, wherein B is selected from an optionally substituted 8- to 15-membered fused heterocycle and optionally substituted Cs-Cis fused carbocycle.
53. The compound or salt of claim 52, wherein B is an optionally substituted 8- to 15- membered fused heterocycle.
54. The compound or salt of claim 52, wherein B is an optionally substituted unsaturated Cs-Cis fused carbocycle.
55. The compound or salt of any one of claims 51 to 54, wherein for B, the heterocycle and carbocycle are each independently bicyclic or tricyclic.
56. The compound or salt of claim 55, wherein for B, the heterocycle and carbocycle are each independently bicyclic.
57. The compound or salt of claim 55, wherein for B, the heterocycle and carbocycle are each independently tricyclic.
58. The compound or salt of any one of claims 51 to 55, wherein B is selected from
optionally substituted with one or more substituents.
59. The compound or salt of claim 58, wherein B is selected from
each of which is optionally substituted with one or more substituents.
60. The compound or salt of claim 59, wherein B is selected from
, and
, each of which is optionally substituted with one or more substituents.
61. The compound or salt of any of claims 51 to 60, wherein for B, the one or more optional substituents are independently selected from oxo, -NH2, halogen, C1-C3 alkyl.
63. The compound or salt of any of claims 1 to 60, wherein for B, the one or more optional substituents of the heterocycle and carbocycle are independently selected at each occurrence from halogen, C1-C3 alkyl, -B(OR20)2, -OR20, -C(O)N(R20)2, -N(R20)2, =0, -CN, - NHCN, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, C2-6 alkenyl, and C2-6 alkynyl.
64. The compound or salt of claim 63, wherein for B, the one or more optional substituents of the heterocycle and carbocycle are independently selected at each occurrence from halogen, oxo, -NH2, C1-C3 alkyl, -B(0H)2, -OH, -O-C1-C3 haloalkyl, -C(0)NH2, -NH2, =0, -CN, C1-6 alkoxy, Ci-e hydroxyalkyl, and C2-6 alkynyl.
66. The compound or salt of any one of claims 1 to 46, wherein B is an optionally substituted 7- to 12-membered fused heterocycle.
67. The compound or salt of any one of claims 1 to 46, wherein B is an optionally substituted 7- to 11 -membered fused heterocycle.
68. The compound or salt of claims 66 or 67, wherein B is an optionally substituted
8- to 10-membered fused heterocycle.
69. The compound or salt of claim 68, wherein B is an optionally substituted 8- to 9- membered fused heterocycle.
70. The compound or salt of claims 66 or 67, wherein the heterocycle is an unsaturated heterocycle.
71. The compound or salt of any one of claims 66 to 70, wherein B has at least one sulfur atom.
72. The compound or salt of any one of claims 66 to 71, wherein B has at least one sulfur atom and at least one nitrogen atom.
73. The compound or salt of any one of claims 66 to 71, wherein B has at least one sulfur atom and at least one oxygen atom.
74. The compound or salt of any one of claims 66 to 71, wherein B has at two sulfur atoms.
78. The compound or salt of any one of claims 66 to 77, wherein the one or more optional substituents of B, are independently selected at each occurrence from halogen, oxo, - NH2, C1-C3 alkyl, -B(OH)2, -OH, -O-C1-C3 haloalkyl, -C(O)NH2, -NH2, =0, -CN, Ci-6 alkoxy,
C1-6 hydroxyalkyl, and C2-e alkynyl.
79. The compound or salt of claim 78, wherein the one or more optional substituents of B, are independently selected at each occurrence from halogen, C1-C3 alkyl, -NH2, and -CN.
81. The compound or salt of any one of claims 1 to 46, wherein B is selected from an optionally substituted 7- to 12-membered fused heterocycle and optionally substituted C9-10 fused carbocycle.
82. The compound or salt of claim 81, wherein the heterocycle of B has at least one sulfur atom.
83. The compound or salt of claim 81 or 82, wherein the heterocycle of B has one or sulfur atoms.
84. The compound or salt of claim 83, wherein the heterocycle of B has at least one nitrogen atom.
86. The compound or salt of any one of claims 81 to 85, wherein the one or more optional substituents of B are independently selected at each occurrence from halogen, C1-C3 alkyl, -B(OR20)2, -OR20, -C(O)N(R20)2, -N(R20)2, =0, -CN, -NHCN, Ci-6 aminoalkyl, Ci-6 alkoxy,
C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, C2-6 alkenyl, and C2-e alkynyl.
87. The compound or salt of any one of claims 81 to 85, wherein the one or more optional substituents of B are independently selected at each occurrence from halogen, C1-C3 alkyl, -OH, -NH2, =0, and -CN.
89. The compound or salt of any one of claims 1 to 46, wherein B is selected from an optionally substituted 8- to 10-membered fused heterocycle having at least one sulfur atom.
90. The compound or salt of claim 89, wherein B is selected from
each of which is optionally substituted.
91. The compound or salt of claims 89 or 90, wherein the one or more optional substituents of B are independently selected at each occurrence from halogen, C1-C3 alkyl, - OR20, -C(O)N(R20)2, -N(R20)2, =0, -CN, -NHCN, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, C2-6 alkenyl, and C2-e alkynyl.
92. The compound or salt of claim 91, wherein the one or more optional substituents of B are independently selected at each occurrence from halogen, C1-C3 alkyl, -NH2, and -CN.
93. The compound or salt of any one of claims 89 to 92, wherein B is substituted.
94. The compound or salt of any one of claims 89 to 92, wherein B is substituted with at least one -NH2.
96. The compound or salt of any one of claims 89 to 94, wherein B is substituted with at least one -NH2 at least one -CN.
98. The compound or salt of any one of claims 1 to 97, wherein R3 is selected from hydrogen, halogen, -CN, -C(O)R22, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-e haloalkyl, and Ci-6 alkyl.
99. The compound or salt of claim 98, wherein R3 is selected from hydrogen, fluorine, -CN, -C(O)H, Ci hydroxyalkyl, and Ci-6 alkyl.
100. The compound or salt of claims 98 or 99, wherein R3 is selected from hydrogen, fluorine, and -CN.
101. The compound or salt of claim 100, wherein R3 is selected from fluorine.
102. The compound or salt of claim 100, wherein R3 is selected from hydrogen.
103. The compound or salt of claim 100, wherein R3 is selected from -CN.
104. The compound or salt of any one of claims 1 to 103, wherein Y is -O-.
105. The compound or salt of any one of claims 1 to 104, wherein L is selected from
C1-C4 alkylene.
106. The compound or salt of claim 105, wherein L is selected from unsubstituted Ci- C4 alkylene.
107. The compound or salt of claim 105, wherein each L is independently selected from a C1-C4 alkylene optionally substituted; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents selected from halogen, -OH, -NO2, =O, =S, -CN, C1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C 1-6 haloalky 1.
108. The compound or salt of claim 107, wherein the optional substituents of L are selected from C1-C4 hydroxyalkyl, C1-C4 alkyl, C3-C6 carbocycle; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8- membered heterocycle wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are optionally substituted with one or more substituents selected from halogen and Ci-e haloalkyl.
111. The compound or salt of claim 107 or 108, wherein each L is independently selected from a substituted C1-C4 alkylene, and wherein two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle 3- to 5-membered heterocycle.
112. The compound or salt of claim 111, wherein each L is independently selected from a substituted C2-3 alkylene, and wherein two substituents on the same carbon atom of L come together to form a C3 carbocycle or 4-membered heterocycle, wherein the C3 carbocycle is optionally substituted with one or more substituents selected from halogen.
116. The compound or salt of claim 105, wherein each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen and C1-C4 alkyl.
118. The compound or salt of claim 105, wherein each L is independently selected from an unsubstituted C1-C4 alkylene.
121. The compound or salt of any one of claims 1 to 120, wherein R2 is selected from heterocycle, -L-heterocycle, -L-aryl, -L-heteroaryl, and -L-N(R23)2, wherein the heterocycle, the heterocycle portion of -L-heterocycle, are each optionally substituted with one or more R6, and wherein the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R7.
122. The compound or salt of claim 121, wherein R2 is -L-heterocycle, wherein the heterocycle portion of R2 is an optionally substituted heterocycle.
123. The compound or salt of claim 121 or 122, wherein R2 is -L-heterocycle, optionally substituted with one or more R6.
124. The compound or salt of any one of claims 1 to 123, wherein Y-R2 is selected from
wherein the heterocycle portion is optionally substituted with one or more R6.
125. The compound or salt of any one of claims 121 to 124, wherein the heterocycle portion of R2 is a heterocycle optionally substituted with one or more substituents selected from halogen, hydroxy, -CN, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, and Ci- C3 aminoalkyl.
126. The compound or salt of claim 125, wherein the heterocycle portion of R2 is a heterocycle optionally substituted with one or more substituents selected from C1-C3 alkyl and halogen.
128. The compound or salt of any one of claims 1 to 124, wherein Y-R2 is selected from
wherein the heterocycle portion is optionally substituted with one or more R6.
129. The compound or salt of claim 128, wherein R6 of R2 is independently selected at each occurrence from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, and C1-C3 aminoalkyl.
130. The compound or salt of claim 129, wherein R6 of R2 is independently selected at each occurrence from C1-C3 alkyl and halogen.
132. The compound or salt of any one of claims 1 to 121, wherein Y-R2 is selected
, eterocycle portion is optionally substituted with one or more R6.
, wherein the heterocycle is optionally substituted.
135. The compound or salt of claim 134, wherein the heterocycle is optionally substituted with one or more substituents selected from halogen, hydroxy, C1-C3 alkyl, - N(R5)S(O)2(R5), -OC(O)N(R5)2, =CH2, OXO, =NO-CI-C3 alkyl, -CH2OC(O)heterocycle, - CH2heterocycle, -CH2OC(O)N(R5)2, and -O-C1-C3 alkyl, wherein the alkyl of -O-C1-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo, and hydroxy.
137. The compound or salt of claim 121, wherein R2 is -L-heteroaryl, wherein the heteroaryl portion is optionally substituted with one or more R7.
138. The compound or salt of claim 137, wherein each R7 is independently selected from halogen, and C1-C4 haloalkyl;
140. The compound or salt of claim 121, wherein R2 is -L-aryl, optionally substituted with one or more R7.
141. The compound or salt of claim 140, wherein Y-R2 is selected from erein the heterocycle portion is optionally substituted with one or more R7. mpound or salt of claim 141, wherein Y-R2 is selected from
143. The compound or salt of claim 121, wherein R2 is -L-heteroaryl, optionally substituted with one or more R7.
O'
144. The compound or salt of claim 143, wherein Y-R2 is selected from HN-^ , wherein the heteroaryl portion is optionally substituted with one or more
146. The compound or salt of claim 121, wherein R2 is -L-N(R23)2.
148. The compound or salt of claim 147, wherein R2 is heterocycle, optionally substituted with one or more R6.
151. The compound or salt of any one of claims 1 to 120, wherein R2 is selected from heterocycle, -L-heterocycle, wherein the heterocycle, and the heterocycle portion of -L- heterocycle, are each optionally substituted with one or more R6; -L-aryl, and -L-heteroaryl, wherein the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R7; and -L-N(R23)2.
152. The compound or salt of claim 151, wherein the heterocycle of R2 is selected
wherein the heterocycle of R2 is optionally
substituted with one or more R6; wherein the aryl and heteroaryl of R2 is selected from
, wherein the aryl and the heteroaryl are each optionally substituted with one or more
153. The compound or salt of claim 152, wherein the heterocycle of R2 is selected
, wherein the heterocycle is optionally substituted with one or more R6; wherein the aryl and heteroaryl of R2 is selected from
154. The compound or salt of any one of claims 151 to 153, wherein each R6 is independently selected from halogen, hydroxy, C1-C3 alkyl, C1-C3 haloalkyl, -N(R5)S(O)2(R5), - OC(O)N(R5)2, =CH2, OXO, =N0-CI-C3 alkyl, -CH2OC(O)heterocycle, -Ckhheterocycle, - CH2OC(O)N(R5)2, and -O-Ci-Cs alkyl, wherein the alkyl of -O-C1-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo, and hydroxy; and wherein each R7 is selected from C1-C3 alkyl, halogen and C1-C3 haloalkyl.
155. The compound or salt of any one of claims 151-154, wherein the heterocycle of
R2, the aryl and heteroaryl of R2, and -N(R20)2 of R2 is selected from is selected from
-L-heterocycle, are each optionally substituted with one or more R6;
the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with
159. The compound or salt of any one of claims 1, wherein Y is -O- and R2 is selected from L-5-membered heteroaryl, wherein L is selected from an optionally substituted C1-C4 alkylene, and wherein the heteroaryl is optionally substituted with one or more R7, wherein each R7 is preferably selected from halogen, C1-C4 alkyl, and C1-C4 haloalkyl.
161. The compound or salt of any one of claims 1 to 158, wherein R1 is selected from 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -OH, -N(R20)2, - NO2, =0, -CN, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and Ci-e haloalkyl.
162. The compound or salt of claim 159, wherein R1 is selected from 5- to 12- membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -OH, -N(R20)2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, and Ci-e haloalkyl.
163. The compound or salt of any one of claims 1 to 161, wherein R1 is an optionally substituted 5- to 12-membered heterocycle.
164. The compound or salt of claim 163, wherein the 5- to 12-membered heterocycle of R1 is unsaturated.
165. The compound or salt of claim 163, wherein the 5- to 12-membered heterocycle of R1 is saturated.
166. The compound or salt of claim 163, wherein the 5- to 12-membered heterocycle of R1 is bridged.
167. The compound or salt of claim 163, wherein the 5- to 12-membered heterocycle of R1 is a spiro heterocycle.
168. The compound or salt of claim 163, wherein the 5- to 12-membered heterocycle of R1 is non-aromatic.
170. The compound or salt of claim 169, wherein the optional one or more substituents are each independently selected from halogen, -OH, -N(R20)2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, =0, -CN, C1-6 hydroxyalkyl, and C1-6 haloalkyl.
171. The compound or salt of claim 170, wherein R1 is selected from
f which is optionally substituted with one or more substituents independently selected from halogen, -OH, -N(R20)2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, =0, -CN, Ci-e hydroxyalkyl, and C 1-6 haloalkyl.
173. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from 6- to 8-membered heterocycle, which is optionally substituted.
174. The compound or salt of claim 173, wherein R1 is selected from 7-membered saturated heterocycle, 8-membered bridged heterocycle, and 6- to 7-membered unsaturated heterocycle, each of which is optionally substituted.
175. The compound or salt of claim 174, wherein R1 is selected from
each of which is optionally substituted.
176. The compound or salt of claim 175, wherein the one or more optional substituents are independently selected from -OH, -CN, oxo, Ci-6 cyanoalkyl.
178. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from an optionally substituted 6- to 7-membered heterocycle.
179. The compound or salt of claim 178, wherein the 6- to 7-membered heterocycle contains only 1 nitrogen atom, and wherein the 6- to 7-membered heterocycle is optionally substituted.
180. The compound or salt of 179, wherein the 6- to 7-membered heterocycle of R1 is bound to Formula (I) via the only 1 nitrogen atom.
181. The compound or salt of any one of claims 178 to 180, wherein R1 is selected from an optionally substituted unsaturated 6-membered heterocycle.
182. The compound or salt of any one of claims 178 to 181, wherein R1 is selected from an optionally substituted unsaturated 7-membered heterocycle.
183. The compound or salt of any one of claims 178 to 182, wherein R1 is selected from
, any of which is optionally substituted.
184. The compound or salt of any one of claims 178, wherein R1 is selected from
, any of which is optionally substituted.
185. The compound or salt of any one of claims 178 to 184, wherein the one or more optional substituents of R1 are each independently selected from halogen, -OR20, -N(R20)2, =0, -CN, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, C1-6 alkyl, -NHCN, and C2-e alkynyl.
186. The compound or salt of any one of claims 178 to 185, each is optionally substituted with one or more substituents independently selected from halogen, -OH, -NH2, -
NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl.
187. The compound or salt of any one of claims 178 to 186, wherein the one or more optional substituents of R1 are each independently selected from halogen.
189. The compound or salt of any one of claims 178 to 184, wherein R1 is selected from
, wherein each is optionally substituted with one or more substituents independently selected from halogen, -OH, -NH2, -NO2, C 1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C 1-6 haloalkyl, and C1-6 alkyl.
190. The compound or salt of claim 189, wherein R1 is selected from
, wherein each is optionally substituted with one or more substituents independently selected from halogen, -OH, -NH2, -NO2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl, and C1-6 alkyl.
191. The compound or salt of claim 190, wherein R1 is selected from
, wherein each is optionally substituted with one or more substituents independently selected from halogen, and C1-6 haloalkyl.
193. The compound or salt of claim 184, wherein R1 is selected from —J-— , which is optionally substituted with one or more substituents independently selected from halogen, -OH, - NH2, -NO2, C 1-6 cyanoalkyl, C 1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, C 1-6 haloalkyl, and C1-6 alkyl.
194. The compound or salt of claim 193, wherein R1 is selected from
, which is optionally substituted with one or more substituents independently selected from halogen.
196. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from an optionally substituted 6- to 7-membered heterocycle.
197. The compound or salt of claim 196, wherein the 6- to 7-membered heterocycle contains only 1 nitrogen atom and optionally one or more additional heteroatoms selected from oxygen, and sulfur.
198. The compound or salt of claim 197, wherein the optionally one or more additional heteroatoms are selected from sulfur.
199. The compound or salt of any one of claims 196 to 198, wherein the 6- to 7- membered heterocycle contains only 1 nitrogen atom and no further additional heteroatoms.
200. The compound or salt of any one of claims 196 to 199, wherein the 6- to 7- membered heterocycle is a non-aromatic 6- to 7-membered heterocycle.
201. The compound or salt of any one of claims 196 to 200, wherein the 6- to 7- membered heterocycle of R1 is bound to Formula (I) via the only 1 nitrogen atom.
202. The compound or salt of any one of claims 196 to 201, wherein R1 is selected from
, each of which is optionally substituted.
203. The compound or salt of any one of claims 196 to 202, wherein the one or more optional substituents of R1 are each independently selected from halogen, -OH, -CN, Ci-6 cyanoalkyl, -NHCN, Ci-6 alkyl, oxo, and C2-e alkynyl.
204. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from an optionally substituted 6- to 10-membered heterocycle.
, each of which is optionally substituted.
206. The compound or salt of claim 205, wherein the one or more optional substituents are independently selected from halogen, =0, -OH, -C(O)N(R20)2, C2-6 alkynyl, -NHCN, -CN, C1-6 aminoalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, and C1-6 alkyl.
208. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from a 7- to 11 -membered spiro heterocycle.
209. The compound or salt of claim 208, wherein R1 is selected from a 10-membered spiro heterocycle.
210. The compound or salt of claims 208 or 209, wherein the spiro heterocycle has at least 3 nitrogen atoms.
H
211. The compound or salt of claim 210, wherein R1 is selected from
, and
, each of which is optionally substituted.
H
212. The compound or salt of claim 211, wherein R1 is selected from
, and
, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -N(R20)2, -NO2, =0, -CN, -NHCN, C1-6 aminoalkyl, C1-6 alkoxy, Ci-
6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, C1-6 alkyl, C2-6 alkenyl, and C2-e alkynyl.
214. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from an optionally substituted unsaturated 9- to 11 -membered heterocycle.
215. The compound or salt of claim 214, wherein R1 is selected from an optionally substituted unsaturated 10-membered heterocycle.
217. The compound or salt of claim 216, wherein
, which is optionally substituted with one or more substituents selected from halogen, -OH, -C(O)N(R20)2, - C(O)NHOR20, -N(R20)2, -C(O)R20, -N02, =0, -CN, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, and C2-e alkynyl.
218. The compound or salt of claim 217, wherein R1 is selected from
each of which is further optionally substituted with one or more substituents selected from halogen, -OH, -N(R20)2, =0, -CN, C i-6 aminoalkyl, Ci-6 alkoxy, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl, and C2-e alkynyl.
219. The compound or salt of any one of claims 214 to 216, wherein the one or more optional substituents of R1 are independently selected from halogen, -OH, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -C(O)N(R20)2, -C(O)NHOR20, -N(R20)2, - C(O)R20, -NO2, =0, -CN, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, C1-6 alkyl, and C2-e alkynyl.
222. The compound or salt of claim 216, wherein the one or more optional substituents of R1 are independently selected from halogen, -OH, -N(R20)2, =0, -CN, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-e haloalkyl, -C(O)N(R20)2, Ci-6 alkyl, and C2-6 alkynyl.
223. The compound or salt of claim 222, wherein the one or more optional substituents of R1 are independently selected from halogen, and -C(O)N(R20)2.
227. The compound or salt of claim 216, wherein the one or more optional substituents of R1 are independently selected from halogen, Ci-6 alkyl, Ci-6 alkyl-N(R20)2, -
C(O)NR20OR20, -C(O)N(R20)2, and -C(O)R20.
228. The compound or salt of claims 227, wherein the one or more optional substituents of R1 are selected from chlorine,
230. The compound or salt of any one of claims 214 to 216, wherein the one or more optional substituents of R1 are independently selected from halogen, -N(R20)2, -CN, Ci-6 alkyl, Ci-6 cyanoalkyl, Ci-6 alkyl-N(R20)2, Ci-6 alkyl-SCh-Ci-e alkyl, C2-6 alkenyl, - C(O)NR20OR20, -C(O)N(R20)2, -C(O)R20, and 5- to 10-membered heterocycle, wherein the 5- to 10-membered heterocycle is optionally substituted independently with one or more R1*, wherein each R1* is independently selected from halogen, -OR20, and C1-6 alkyl.
231. The compound or salt of claims 216 or 230, wherein the one or more optional substituents of R1 are selected from chlorine, -NH2, -CN, Ci alkyl, C2 alkenyl,
o o o o o
233. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from an optionally substituted 6- to 11 -membered heterocycle, wherein the 6- to 11 -membered heterocycle has at least one nitrogen atom.
234. The compound or salt of claim 233, wherein the one or more optional substituents of R1 is selected from halogen, -OR20, -C(O)N(R20)2, -C(O)R20, -S(O)2R20, =0, -Ci-6 alkyl(=NOR20), =NO(R20), -CN, -NHCN, Ci-6 alkyl, and 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted independently with one or more R1*; and wherein each R1* is independently selected from halogen, and Ci-6 alkyl.
238. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from hydrogen and optionally substituted 5- to 15-membered heterocycle.
239. The compound or salt of claims 238, wherein R1 is selected from
each of which is optionally substituted.
240. The compound or salt of claim 239, wherein the optional one or more substituents ofR1 is selected from -OH, =NO(R20), -NHCN, and Ci-6 alkyl.
242. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from hydrogen and optionally substituted 7- to 10-membered heterocycle.
243. The compound or salt of claim 242, wherein R1 is selected from hydrogen
optionally substituted.
244. The compound or salt of claims 242 or 243, wherein the optional one or more substituents of R1 are independently selected from halogen, -NH2, -S(O)2(R20), -C(O)R20, - C(O)N(R20)2, =O,=NO(R20), -CN, -NHCN, C1-6 alkyl, and 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle is optionally substituted independently with one or more R1*; and wherein each R1* is independently selected from halogen, and C1-6 alkyl.
246. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from an optionally substituted 8- to 10-membered heterocycle.
247. The compound or salt of claim 246, wherein the heterocycle is bicyclic.
248. The compound or salt of claims 246 or 247, wherein the heterocycle has at least one nitrogen atom.
249. The compound or salt of claims 246 or 247, wherein the heterocycle has at least two nitrogen atoms.
251. The compound or salt of any one of claims 246 or 250, wherein the optional one or more substituents of R1 are independently selected from halogen,
-membered heteroaryl, wherein the 5- to 9-membered heteroaryl is substituted with at least one R1*, wherein the R1* is selected from halogen, and Ci-6 alkyl.
252. The compound or salt of any one of claims 246 or 251, wherein the optional one or more substituents of R1 are independently selected from chlorine,
254. The compound or salt of any one of claims 1 to 168, wherein R1 is an optionally substituted 12- to 15-membered heterocycle.
255. The compound or salt of claim 254, wherein R1 is an optionally substituted 12- membered heterocycle.
256. The compound or salt of claim 255, wherein R1 is an optionally substituted 13- membered heterocycle.
257. The compound or salt of claim 256, wherein R1 is an optionally substituted 14- membered heterocycle.
258. The compound or salt of claim 257, wherein R1 is an optionally substituted 15- membered heterocycle.
259. The compound or salt of any one of claims 254 to 258, wherein the heterocycle of R1 is tricyclic.
260. The compound or salt of any one of claims 254 to 259, wherein the heterocycle of R1 contains a fused heterocycle.
261. The compound or salt of any one of claims 254 to 260, wherein the heterocycle of R1 contains a spiro heterocycle.
262. The compound or salt of any one of claims 254 to 261, wherein the heterocycle of R1 contains a fused and spiro-heterocycle.
263. The compound or salt of any one of claims 254 to 262, wherein the heterocycle of R1 is an unsaturated heterocycle.
264. The compound or salt of any one of claims 254 to 263, wherein the heterocycle of R1 is a non-aromatic heterocycle.
265. The compound or salt of any one of claims 254 to 264, wherein the heterocycle of R1 has at least one double bond.
266. The compound or salt of any one of claims 254 to 265, wherein the heterocycle of R1 has at least two double bonds.
267. The compound or salt of any one of claims 254 to 266, wherein the heterocycle of R1 has two double bonds.
269. The compound or salt of claim 268, wherein the optional one or more substituents are selected from halogen, -OH, -NHCN, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -C(O)N(R20)2, -C(O)NHOR20, -N(R20)2, -C(O)R20, -NO2, =0, -CN, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, and C2-6 alkynyl.
270. The compound or salt of claim 269, wherein the optional one or more substituents are selected from halogen, -OH, Ci-6 alkyl, and -C(O)N(R20)2.
272. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from substituted 6- to 7-membered heterocycle, wherein the 6- to 7-membered heterocycle is substituted with at least one -NHCN, and further optionally substituted with one or more Ci-6 alkyl.
274. The compound or salt of any one of claims 1 to 168, wherein R1 is selected from an optionally substituted bridged 8- to 9-membered heterocycle.
275. The compound or salt of claim 274, wherein the heterocycle of R1 is selected from
each of which is optionally substituted.
276. The compound or salt of claims 274 or 275, wherein the one or more substituents of R1 are selected from halogen, Ci-6 alkyl, -N(R20)2, and Ci-6 aminoalkyl.
278. The compound or salt of claim 274, wherein R1 is selected from an optionally substituted bridged 8-membered heterocycle, wherein the heterocycle contains heteroatoms selected from nitrogen.
279. The compound or salt of claim 278, wherein the one or more substituents of R1 are selected from Ci-6 alkyl, -N(R20)2, and Ci-6 aminoalkyl.
280. The compound or salt of claim 279, wherein the heterocycle of R1 is selected from each of which is optionally substituted. ound or salt of any one of claims 278 to 280, wherein R1 is selected ound or salt of claims 1 to 168, or 281, wherein R1 is selected from
283. The compound or salt of any one of claims 1 to 163, wherein R1 is hydrogen.
284. The compound or salt of any one of claims 1 to 283, wherein n is 0.
285. A method of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor and a compound of Formula (I- A):
Formula (I- A), or a pharmaceutically acceptable salt thereof wherein:
R1 is selected from 8- to 10-membered heterocycle, wherein the 8- to 10-membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -OR20, -S(O)2(R20), -C(O)N(R20)2, -Ci-6 alkyl(=NOR20), -C(O)R20, =0, -CN, -NHCN, Ci- 6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Cn 6 alkyl-SO2R20, Ci-6 alkoxyalkyl, Ci-6 alkyl, C2-6 alkynyl, and 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle are each optionally substituted independently with one or more R1*; each R1* is independently selected from halogen, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(O)NR20OR20, - N(R20)C(O)R20, -N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, -C(O)R20, -C(O)OR20, - OC(O)R20, -OC(O)N(R20)2, -N02, =0, =N(R20), =NO(R20), -CN, -NHCN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-e hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, and Cs-Ci2 carbocycle;
B is selected from a 7- to 15-membered heterocycle and C7-C15 carbocycle, wherein the 7- to 15-membered heterocycle and C7-C15 carbocycle are each optionally substituted with one or more substituents independently selected from halogen, -CN, -N02, =0, -N(R21)2, -B(OR21)2, - OR21, -SR21, -S(O)2(R21), -S(O)2N(R21)2, -NR21S(O)2R21, -C(O)N(R21)2, -C(O)NR21OR21, - N(R21)C(O)R21, -N(R21)C(O)N(R21)2, -N(R21)C(O)OR21, -C(0)R21, C(0)0R21, -0C(0)R21, - OC(O)N(R21)2, CI-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 alkoxyalkyl, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-6 haloalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, Cs-Ci2 carbocycle and 5- to 12-membered heterocycle;
Y is selected from a bond, and -0-;
R2is selected from heterocycle, aryl, Ci-Ce alkyl, -L-heterocycle, -L-N(R23)2, -L-OR23, -L- aryl, -L-heteroaryl, -L-cycloalkyl, -L-NHC(=NH)NH2, -L-C(O)N(R23)2, -L-Ci-Ce haloalkyl, -L- OR23, -L-NR23C(O)-aryl, -L-C00H, -L-NR23S(O)2(R23), -L-S(O)2N(R23)2, -L-N(R23)C(O)(OR23), -L-OC(O)N(R23)2, and -L-C(=O)OCi-Ce alkyl, wherein the heterocycle, the heterocycle portion of
-L-heterocycle, and the cycloalkyl portion of the -L-cycloalkyl are each optionally substituted with one or more R6, and wherein the aryl, aryl portion of -L-NR23C(O)-aryl, the aryl portion of -L- NR23C(O)-aryl, the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R7;
R3 is selected from hydrogen, halogen, -CN, -NO2, -N(R20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -N(R20)C(O)R20, - N(R20)C(O)N(R20)2, -N(R20)C(O)OR20, -C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-C12 carbocycle and 5- to 12-membered heterocycle; each R4 is independently selected from halogen, -NO2, =0, =S, -CN, C1-6 alkyl, C2-6 alkynyl, C 1-6 alkyl -N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C 1-6 cyanoalkyl, C1-6 hydroxyalkyl, and C1-6 haloalkyl; n is selected from 0, 1, 2, 3, and 4; each R5 is independently selected from hydrogen and Ci-Ce alkyl; each R6 is independently selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, oxo, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, =CH2, NMO-C1-C3 alkyl, C1-C3 aminoalkyl, - N(R5)S(O)2(R5), -Q-phenyl, -Q-phenylSChF, -NHC(O)phenyl, -NHC(O)phenylSO2F, C1-C3 alkyl substituted pyrazolyl, -C1-C3 alkyl-N(R5)2, -C(O)N(R5)2, tert-butyldimethylsilyloxyCFh-, - N(R5)2, (C1-C3 alkoxy)Ci-C3 alkyl-, (C1-C3 alkyl)C(=O), oxo, (C1-C3 haloalkyl)C(=O)-, -SO2F, (C1-C3 alkoxy)Ci-C3 alkoxy, -CH2OC(O)N(R5)2, -CH2NHC(O)OCI-C6 alkyl, - CH2NHC(O)N(R5)2, -CH2NHC(O)CI-C6 alkyl, -CH2(pyrazolyl), -CH2NHSO2C1-C6 alkyl, - CH2OC(O)heterocycle, -OC(O)N(R5)2, -0C(0)NH(Ci-C3 alkyl)O(Ci-C3 alkyl), -0C(0)NH(Ci- C3 alkyl)O(Ci-C3 alkyl)phenyl(Ci-C3 alkyl)N(CH3)2, -0C(0)NH(Ci-C3 alkyl)O(Ci-C3 alkyl)phenyl, -OC(O)heterocycle, -O-C1-C3 alkyl, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -NR20S(O)2R20, and -CFhheterocycle, wherein the phenyl of -NHC(O)phenyl and -0C(0)NH(CI-C3 alkyl)(Ci-C3 alkyl)phenyl are optionally substituted with one or more substituents selected from -C(0)H and OH, and wherein the alkyl of -O-C1-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo and hydroxy; and wherein the heterocycle of -CH2heterocyclyl is optionally substituted with oxo; each Q is selected from a bond, S, and O; each R7 is independently selected from halogen, hydroxy, HC(=0)-, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 hydroxyalkyl, -C1-C3 alkyl-N(R5)2, -C(O)N(R5)2, and -N(R5)2; each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen, hydroxy, C1-6 alkoxy, C1-C4 hydroxyalkyl,
C1-C4 alkyl, C3-C6 carbocycle, and 3- to 8-membered heterocycle, wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -NO2, =0, =S, -CN, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, Ci-ehydroxyalkyl, C1-6 haloalkyl; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle, wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -NO2, =0, =S, -CN, Ci-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, and C1-6 haloalkyl; each R20 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle; each R21 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle; each R23 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -N(CI-6 alkyl)2, Ci-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12-membered heterocycle.
286. The compound or salt of claim 285, wherein Formula (I-A) is represented by Formula (I-B),
Formula (I-B), or a pharmaceutically acceptable salt thereof wherein:
wherein R1 is selected from
each of which is optionally substituted with one or more substituents independently selected from halogen, - S(O)2(R20), -C(O)N(R20)2, -CI-6 alkyl(=NOR20), -C(O)R20, =0, and 5- to 12-membered heterocycle, wherein the 5- to 12-membered heterocycle are each optionally substituted independently with one or more R1*; each R1* is independently selected from halogen, -OR20, -N(R20)2, -NO2, =0, =N(R20), =NO(R20), -CN, -NHCN, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C 1-6 haloalkyl, and C1-6 alkyl;
Y is -0-;
R2 is selected from -L-heterocycle, and -L-N(R23)2, wherein the heterocycle portion of -L- heterocycle is optionally substituted with one or more R6.
B is selected from a 7- to 15-membered heterocycle, wherein the 7- to 15-membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -CN, =0, -N(R21)2, -OR21, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, Ci-e hydroxyalkyl, C 1-6 cyanoalkyl, C 1-6 haloalkyl, C1-6 alkyl, and C2-e alkynyl;
R3 is selected from hydrogen, halogen, -CN, -N(R20)2, -OR20, -C(O)R20, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C1-6 cyanoalkyl, C1-6 haloalkyl, and C1-6 alkyl; and each R20 is independently selected from hydrogen; and C1-6 alkyl, and C3-12 carbocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NH2, -N(Ci-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, and oxo.
287. The compound or salt of claims 285 or 286, wherein R1 is selected from
optionally substituted with one or more substituents independently selected from halogen, -S(O)2(R20), -C(O)N(R20)2, -Ci-6 alkyl(=NOR20), -C(O)R20, =0, and 5- to 9-membered heterocycle, wherein the 5- to -membered heterocycle are each optionally substituted independently with one or more R1*; and each R1* is independently selected from halogen, Ci-6 haloalkyl, and Ci-6 alkyl.
288. The compound or salt of any one of claims 285 to 287, wherein Y is -O-.
289. The compound or salt of any one of claims 285 to 288, wherein each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen, and C1-C4 alkyl; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 4- to 6- membered heterocycle, wherein the C3-C6 carbocycle and 3- to 6-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen.
291. The compound or salt of any one of claims 285 to 290, wherein Y-R2 is selected
The compound or salt of any one of claims 285 to 291, wherein B is selected from an optionally substituted 8- to 10-membered heterocycle.
293. The compound or salt of claim 292, wherein the 8- to 10-membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -CN, -
NH2, and C1-6 alkyl.
294. The compound or salt of any one of claims 285 to 293, wherein the heterocycle of
295. The compound or salt of any one of claims 285 to 294, wherein
each of which is optionally substituted with one or more substituents independently selected from halogen, -CN, -NH2, and C1-6 alkyl.
297. The compound or salt of any one of claims 285 to 296, wherein R3 is -CN.
298. The compound or salt of any one of claims 285 to 297, wherein
which is optionally substituted with one or more substituents.
302. The compound or salt of any one of claims 285 to 301, wherein the one or more optional substituents of R1 are independently selected from halogen, -S(O)2(R20), -C(O)N(R20)2, -
Ci-6 alkyl(=NOR20), -C(O)R20 =0, and 5- to 9-membered heterocycle, wherein the 5- to - membered heterocycle are each optionally substituted independently with one or more R1*.
303. The compound or salt of any one of claims 285 to 301, wherein the one or more optional substituents of R1 are independently selected from halogen, -S(O)2(R20), -C(O)N(R20)2, - Ci-6 alkyl(=NOR20), -C(O)R20, =0, and 5- to 9-membered heterocycle, wherein the 5- to - membered heterocycle are each optionally substituted independently with one or more R1*.
304. The compound or salt of any one of claims 285 to 301, wherein the one or more optional substituents of R1 are independently selected from halogen, -C(O)N(R20)2, -Ci-6 alkyl(=NOR20), -C(O)R20, and 5- to 9-membered heterocycle, wherein the 5- to -membered heterocycle are each optionally substituted independently with one or more R1*.
305. The compound or salt of claim 304, wherein each R1* is independently selected from halogen, Ci-6 haloalkyl, and Ci-6 alkyl.
306. The compound or salt of any one of claims 285 to 298, wherein
which is subtituted with at least one substituent.
309. A pharmaceutical composition comprising the compound of any one of claims 1 to 308 and a pharmaceutically acceptable excipient.
310. A method of treating a disease or disorder, using a compound or salt of any one of claims 1 to 308 or a pharmaceutical composition of claim 309.
311. A method of inhibiting KRas G12D and/or other G12 mutants, using a compound or salt of any one of claims 1 to 308 or a pharmaceutical composition of claim 309.
312. A method of inhibiting KRas G12D and/or other G12 mutants, using a compound or salt of any one of claims 1 to 308 or a pharmaceutical composition of claim 309.
313. A method of inhibiting KRas G12D and/or other G12 alleles, using a compound or salt of any one of claims 1 to 308 or a pharmaceutical composition of claim 309.
314. A method of inhibiting KRas G12D and/or other alleles, using a compound or salt of any one of claims 1 to 308 or a pharmaceutical composition of claim 309.
315. Use of a compound of any one of claims 1 to 308 thereof in the preparation of a target protein degrading compound by using chemical modification of compound of any one of claims 1-308.
316. A bifunctional compound composed of a target protein-binding moiety and an E3 ubiquitin ligase-binding moiety, wherein the target protein-binding moiety is derived from the compound of any of claims 1 to 308.
317. A method of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of an immunomodulator inhibitor and a compound selected from compound 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36,
37, 38, 39, 40, 41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62,
63, 64, 65, 66, 67, 68, 69, 70, 71, 72, 73, 74, 74A, 74B, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85,
86, 87, 88, 89, 90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107, 108,
109, 110, and 111, or a pharmaceutically acceptable salt of anyone thereof.
318. The method of claims 285 or 317, wherein the immunomodulator inhibitor is a PD-1 inhibitor.
319. The method of claim 318, wherein the PD-1 inhibitor is an antibody or antigenbinding fragment.
320. The method of claim 318, wherein the PD-1 inhibitor is selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, and a biosimilar thereof.
321. The method of claim 318, wherein the PD-1 inhibitor is nivolumab or a biosimilar thereof.
322. The method of claims 320 or 321, wherein the therapeutically effective amount of nivolumab or biosimilar thereof in the combination is about 240 mg administered every two weeks.
323. The method of claims 320 or 321, wherein the therapeutically effective amount of nivolumab, or biosimilar thereof, in the combination is about 480 mg administered every four weeks.
324. The method of claim 320, wherein the PD-1 inhibitor is pembrolizumab or a biosimilar thereof.
325. The method of claim 324, wherein the therapeutically effective amount of pembrolizumab or biosimilar thereof in the combination is about 200 mg administered every three weeks.
326. The method of claim 320, wherein the wherein the PD-1 inhibitor is cemiplimab or a biosimilar thereof.
327. The method of claims 285 or 317, wherein the immunomodulator inhibitor is a PD-L1 inhibitor.
328. The method of claim 327, wherein the PD-L1 inhibitor is selected from the group consisting of atezolizumab, avelumab, durvalumab, and a biosimilar thereof.
329. The method of claim 328, wherein the PD-L1 inhibitor is atezolizumab or a biosimilar thereof.
330. The method of claims 285 or 317, wherein the immunomodulator inhibitor is a CTLA-4 inhibitor.
331. The method of any one of claims 285 to 330, wherein the immunomodulator inhibitor is administered orally.
332. The method of any one of claims 285 to 331, wherein the cancer is selected from: Cardiac: sarcoma (angiosarcoma, fibrosarcoma, rhabdomyosarcoma, liposarcoma), myxoma, rhabdomyoma, fibroma, lipoma and teratoma;
Lung: bronchogenic carcinoma (squamous cell, undifferentiated small cell, undifferentiated large cell, adenocarcinoma), alveolar (bronchiolar) carcinoma, bronchial adenoma, sarcoma, lymphoma, chondromatous hamartoma, mesothelioma;
Gastrointestinal: esophagus (squamous cell carcinoma, adenocarcinoma, leiomyosarcoma, lymphoma), stomach (carcinoma, lymphoma, leiomyosarcoma), pancreas (ductal adenocarcinoma, insulinoma, glucagonoma, gastrinoma, carcinoid tumors, vipoma), small bowel (adenocarcinoma, lymphoma, carcinoid tumors, Kaposi's sarcoma, leiomyoma, hemangioma, lipoma, neurofibroma, fibroma), large bowel (adenocarcinoma, tubular adenoma, villous adenoma, hamartoma, leiomyoma);
Genitourinary tract: kidney (adenocarcinoma, Wilm's tumor (nephroblastoma), lymphoma, leukemia), bladder and urethra (squamous cell carcinoma, transitional cell carcinoma, adenocarcinoma), prostate (adenocarcinoma, sarcoma), testis (seminoma, teratoma, embryonal carcinoma, teratocarcinoma, choriocarcinoma, sarcoma, interstitial cell carcinoma, fibroma, fibroadenoma, adenomatoid tumors, lipoma);
Liver: hepatoma (hepatocellular carcinoma), cholangiocarcinoma, hepatoblastoma, angiosarcoma, hepatocellular adenoma, hemangioma;
Biliary tract: gall bladder carcinoma, ampullary carcinoma, cholangiocarcinoma;
Bone: osteogenic sarcoma (osteosarcoma), fibrosarcoma, malignant fibrous histiocytoma, chondrosarcoma, Ewing's sarcoma, malignant lymphoma (reticulum cell sarcoma), multiple myeloma, malignant giant cell tumor chordoma, osteochronfroma (osteocartilaginous exostoses), benign chondroma, chondroblastoma, chondromyxofibroma, osteoid osteoma and giant cell tumors;
Nervous system: skull (osteoma, hemangioma, granuloma, xanthoma, osteitis deformans), meninges (meningioma, meningiosarcoma, gliomatosis), brain (astrocytoma, medulloblastoma, glioma, ependymoma, germinoma (pinealoma), glioblastoma multiform, oligodendroglioma, schwannoma, retinoblastoma, congenital tumors), spinal cord neurofibroma, meningioma, glioma, sarcoma);
Gynecological: uterus (endometrial carcinoma), cervix (cervical carcinoma, pre-tumor cervical dysplasia), ovaries (ovarian carcinoma (serous cystadenocarcinoma, mucinous cystadenocarcinoma, unclassified carcinoma), granulosa-thecal cell tumors, Sertoli-Leydig cell tumors, dysgerminoma, malignant teratoma), vulva (squamous cell carcinoma, intraepithelial carcinoma, adenocarcinoma, fibrosarcoma, melanoma), vagina (clear cell carcinoma, squamous cell carcinoma, botryoid sarcoma (embryonal rhabdomyosarcoma), fallopian tubes (carcinoma);
Hematologic: blood (myeloid leukemia (acute and chronic), acute lymphoblastic leukemia, chronic lymphocytic leukemia, myeloproliferative diseases, multiple myeloma, myelodysplastic syndrome), Hodgkin's disease, non-Hodgkin's lymphoma (malignant lymphoma);
Skin: malignant melanoma, basal cell carcinoma, squamous cell carcinoma, Kaposi's sarcoma, moles dysplastic nevi, lipoma, angioma, dermatofibroma, keloids, psoriasis; and
Adrenal glands: neuroblastoma.
333. The method of any one of claims 285 to 332, wherein the cancer is non-small cell lung cancer, small cell lung cancer, colorectal cancer, rectal cancer or pancreatic cancer.
334. The method of any one of claims 285 to 332, wherein the cancer is non-small cell lung cancer.
335. The method of any one of claims 285 to 332, wherein the cancer is small cell lung cancer.
336. The method of any one of claims 285 to 332, wherein the cancer is colorectal cancer.
337. The method of any one of claims 285 to 332, wherein the cancer is rectal cancer.
338. The method of any one of claims 285 to 332, wherein the cancer is pancreatic cancer.
339. The method of any one of claims 285 to 332, wherein the cancer is a solid tumor cancer.
340. The method of any one of claims 285 to 339, wherein the cancer is selected from a KRas mutant-associated cancer.
341. The method of any one of claims 285 to 339, wherein the cancer is selected from a KRas wildtype-associated cancer.
342. The method of any one of claims 285 to 341, wherein the cancer is selected from a KRas G12D-associated cancer, a KRas G12V-associated cancer, and a KRas wildtype- associated cancer.
343. The method of any one of claims 285 to 342, wherein the cancer is a KRas G12D- associated cancer.
344. The method of any one of claims 285 to 342, wherein the cancer is a KRas G12V- associated cancer.
345. The method of any one of claims 341 to 344, wherein the KRas wildtype is hyperactivated.
346. The method of any one of claims 285 to 345, wherein the immunomodulator inhibitor synergistically increases the sensitivity of cancer cells to the compound or salt of Formula (I).
347. The method of any one of claims 317 to 346, wherein the compound is selected from compounds 2, 3, 4, 14, 25, and 74, or a pharmaceutical salt thereof.
348. The method of any one of claims 317 to 346, wherein the compound is compound 74, or a pharmaceutical salt thereof.
349. The method of any one of claims 317 to 346, wherein the compound is compound 25, or a pharmaceutical salt thereof.
350. The method of any one of claims 317 to 346, wherein the compound is selected from compound 14, or a pharmaceutical salt thereof.
351. The method of any one of claims 317 to 346, wherein the compound is selected from compound 4, or a pharmaceutical salt thereof.
352. The method of any one of claims 317 to 346, wherein the compound is selected from compound 3, or a pharmaceutical salt thereof.
353. The method of any one of claims 317 to 346, wherein the compound is selected from compound 2, or a pharmaceutical salt thereof.
354. A method of treating a subject identified or diagnosed as having a KRas associated cancer, comprising (a) administering a KRas inhibitor as a monotherapy until disease progression, and (b) after (a), administering to the subject a combination of an immunomodulator, or a pharmaceutical composition thereof, and a compound of Formula (II), or a pharmaceutically acceptable salt thereof, or a pharmaceutical composition thereof.
355. The method of claim 354, wherein the KRas associated cancer is a KRas G12D- associated cancer.
356. The method of claim 354, wherein the KRas associated cancer is a KRas G12V- associated cancer.
357. The method of claim 354, wherein the KRas associated cancer is a KRas wildtype-associated cancer.
358. The method of claim 354, wherein the compound is selected from compound 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of anyone thereof.
359. The method of any one of claims 317 to 358, wherein the immunomodulator is selected from a checkpoint inhibitor.
360. The method of any one of claims 317 to 358, wherein the immunomodulator is selected from a PD-l/PD-Ll pathway inhibitor.
361. A method of treating cancer in a subject in need thereof, comprising administering to the subject a combination of an immunomodulator and a compound of Formula (II), wherein the compound is selected from: compound 1, 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, 15, 16, 17, 18, 19, 20, 21, 22, 23, 24, 25, 26, 27, 28, 29, 30, 31, 32, 33, 34, 35, 36, 37, 38, 39, 40,
41, 42, 43, 44, 45, 46, 47, 48, 49, 50, 51, 52, 53, 54, 55, 56, 57, 58, 59, 60, 61, 62, 63, 64, 65, 66,
67, 68, 69, 70, 71, 72, 73, 74, 74A, 74B, 75, 76, 77, 78, 79, 80, 81, 82, 83, 84, 85, 86, 87, 88, 89,
90, 91, 92, 93, 94, 95, 96, 97, 98, 99, 100, 101, 102, 103, 104, 105, 106, 107, 108, 109, 110, and
111, or a pharmaceutically acceptable salt of anyone thereof.
362. A method of treating cancer in a subject in need thereof, comprising administering to the subject a combination of an immunomodulator and a compound of Formula (II), wherein the compound is selected from: compounds 2, 3, 4, 14, 25, and 74, or a pharmaceutically acceptable salt of any one thereof.
363. The method of claim 316, wherein the compound is compound 2.
364. The method of claim 316, wherein the compound is compound 3.
365. The method of claim 316, wherein the compound is compound 4.
366. The method of claim 316, wherein the compound is compound 14.
367. The method of claim 316, wherein the compound is compound 25.
368. The method of claim 316, wherein the compound is compound 74.
369. The method of any one of claims 362 to 368, wherein the immunomodulator inhibitor is a PD-1 inhibitor.
370. The method of claim 369, wherein the PD-1 inhibitor is an antibody or antigenbinding fragment.
371. The method of claim 369, wherein the PD-1 inhibitor is selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, and a biosimilar thereof.
372. The method of claim 369, wherein the PD-1 inhibitor is nivolumab or a biosimilar thereof.
373. The method of claim 369, wherein the PD-1 inhibitor is pembrolizumab or a biosimilar thereof.
374. The method of claim 369, wherein the wherein the PD-1 inhibitor is cemiplimab or a biosimilar thereof.
375. The method of any one of claims 362 to 368, wherein the immunomodulator inhibitor is a PD-L1 inhibitor.
376. The method of claim 375, wherein the PD-L1 inhibitor is selected from the group consisting of atezolizumab, avelumab, durvalumab, and a biosimilar thereof.
377. The method of claim 376, wherein the PD-L1 inhibitor is atezolizumab or a biosimilar thereof.
378. The method of any one of claims 362 to 368, wherein the immunomodulator inhibitor is a CTLA-4 inhibitor.
379. The method of any one of claims 361 to 378, wherein the combination exhibits synergy.
380. A method of treating cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of an immunomodulator inhibitor and a compound of Formula (I):
Formula (I), or a pharmaceutically acceptable salt thereof wherein:
B is selected from a 7- to 15-membered heterocycle and C7-C15 carbocycle, wherein the 7- to 15- membered heterocycle and C7-C15 carbocycle are each optionally substituted with one or more substituents independently selected from halogen, -CN, -NO2, =0, -N(R21)2, -B(OR21)2, -OR21, -SR21, -S(O)2(R21), -S(O)2N(R21)2, -NR21S(O)2R21, -C(O)N(R21)2, -C(O)NR21OR21, - N(R21)C(O)R21, -N(R21)C(O)N(R21)2, -N(R21)C(O)OR21, -C(0)R21, C(0)0R21, -0C(0)R21, - OC(O)N(R21)2, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, C1-6 alkyl, C2-6 alkenyl, C2-e alkynyl, C3-C12 carbocycle and 5- to 12-membered heterocycle;
R1 is selected from hydrogen and 5- to 15-membered heterocycle, wherein the 5- to 15- membered heterocycle is optionally substituted with one or more substituents independently selected from halogen, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, - S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, -C(=NR20)N(R20)2, -C1-6 alkyl(=NOR20), - C(O)NR20OR20, -N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, - C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, -N02, =0, =N(R20), =NO(R20), -CN, -
NHCN, Ci-6 alkyl-N(R20)2, Ci-6 aminoalkyl, Ci-6 alkoxy, Ci-6 hydroxyalkyl, Ci-6 cyanoalkyl, Ci-e haloalkyl, Ci-6 alkyl-SCLR20, Ci-6 alkoxyalkyl, Ci-6 alkyl, C2-6 alkenyl, C2-e alkynyl, C3- C12 carbocycle and 5- to 12-membered heterocycle, wherein the C3-C12 carbocycle and 5- to 12-membered heterocycle are each optionally substituted independently with one or more R1*; each R1* is independently selected from halogen, -B(OR20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, - C(O)NR20OR20, -N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -N(R20)2, - C(O)R20, -C(O)OR20, -OC(O)R20, -OC(O)N(R20)2, -NO2, =0, =N(R20), =NO(R20), -CN, - NHCN, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, Ci-e haloalkyl, C1-6 alkyl, C2-6 alkenyl, C2-e alkynyl, and C3-C12 carbocycle;
Y is selected from a bond, -0-, -S-, and -N(R5)-;
R2 is selected from heterocycle, aryl, Ci-Ce alkyl, -L-heterocycle, -L-N(R23)2, -L-OR23, -L-aryl, - L-heteroaryl, -L-cycloalkyl, -L-NHC(=NH)NH2, -L-C(O)N(R23)2, -L-Ci-Ce haloalkyl, -L- OR23, -L-NR23C(O)-aryl, -L-C00H, -L-NR23S(O)2(R23), -L-S(O)2N(R23)2, -L- N(R23)C(O)(OR23), -L-OC(O)N(R23)2, and -L-C(=O)OCI-C6 alkyl, wherein the heterocycle, the heterocycle portion of -L-heterocycle, and the cycloalkyl portion of the -L-cycloalkyl are each optionally substituted with one or more R6, and wherein the aryl, the aryl portion of -L- NR23C(O)-aryl, the aryl portion of -L-NR23C(O)-aryl, the aryl of the -L-aryl, and the heteroaryl of -L-heteroaryl are each optionally substituted with one or more R7;
R3 is selected from hydrogen, halogen, -CN, -NO2, -N(R20)2, -OR20, -SR20, -S(O)2(R20), - S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, -C(O)N(R20)2, - N(R20)C(O)R20, -N(R20)C(0)N(R20)2, -N(R20)C(O)OR20, -C(O)R20, -C(O)OR20, -OC(O)R20, - OC(O)N(R20)2, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 alkoxyalkyl, C1-6 hydroxyalkyl, C 1-6 cyanoalkyl, C 1-6 haloalkyl, C1-6 alkyl, C2-6 alkenyl, C2-e alkynyl, C3-C12 carbocycle and 5- to 12-membered heterocycle; each R4 is independently selected from halogen, -NO2, =0, =S, -CN, C1-6 alkyl, C2-6 alkynyl, C1-6 alkyl-N(R20)2, C 1-6 aminoalkyl, C1-6 alkoxy, C1-6 cyanoalkyl, Ci-e hydroxyalkyl, and C1-6 haloalkyl; n is selected from 0, 1, 2, 3, and 4; each R5 is independently selected from hydrogen and Ci-Ce alkyl; each R6 is independently selected from halogen, hydroxy, C1-C3 hydroxyalkyl, C1-C3 alkyl, oxo, C1-C3 haloalkyl, C1-C3 alkoxy, cyano, =CH2, NMO-C1-C3 alkyl, C1-C3 aminoalkyl, - N(R5)S(O)2(R5), -Q-phenyl, -Q-phenylSCLF, -NHC(O)phenyl, - NHC(O)phenylSO2F, C1-C3 alkyl substituted pyrazolyl, -C1-C3 alkyl-N(R5)2, -C(O)N(R5)2, -C1-C3 alkyl-N(R5)2, -
C(O)N(R5)2, tert-butyldimethylsilyloxyCH2-, -N(R5)2, (C1-C3 alkoxy)Ci-C3 alkyl-, (C1-C3 alkyl)C(=O), oxo, (C1-C3 haloalkyl)C(=O)-, -SO2F, (C1-C3 alkoxy)Ci-C3 alkoxy, - CH2OC(O)N(R5)2, -CH2NHC(O)OC1-C6 alkyl, -CH2NHC(O)N(R5)2, -CH2NHC(O)CI-C6 alkyl, -CH2(pyrazolyl), -CH2NHSO2C1-C6 alkyl, -CH2OC(O)heterocycle, -OC(O)N(R5)2, - OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl), -OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl)phenyl(Ci-C3 alkyl)N(CH3)2, -OC(O)NH(CI-C3 alkyl)O(Ci-C3 alkyl)phenyl, -OC(O)heterocycle, -O-C1-C3 alkyl, -S(O)2(R20), -S(O)2N(R20)2, -S(O)N(R20)2, -S(O)R20(=NR20), -NR20S(O)2R20, and - Cltheterocycle, wherein the phenyl of -NHC(O)phenyl and -0C(0)NH(Ci-C3 alkyl)(Ci-C3 alkyl)phenyl are optionally substituted with one or more substituents selected from -C(O)H and OH, and wherein the alkyl of -O-C1-C3 alkyl is optionally substituted with substituents selected from heterocycle, oxo and hydroxy; and wherein the heterocycle of - CH2heterocyclyl is optionally substituted with oxo; each Q is selected from a bond, S, and O; each R7 is independently selected from halogen, hydroxy, HC(=O)-, C1-C4 alkyl, C1-C4 alkoxy, C1-C4 haloalkyl, C1-C4 hydroxyalkyl, -C1-C3 alkyl-N(R5)2, -C(O)N(R5)2, and -N(R5)2; each L is independently selected from a C1-C4 alkylene optionally substituted with one or more substituents independently selected from halogen, hydroxy, C1-6 alkoxy, C1-C4 hydroxyalkyl, C1-C4 alkyl, C3-C6 carbocycle, and 3- to 8-membered heterocycle, wherein the C3-C6 carbocycle and 3- to 8-membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -NO2, =0, =S, -CN, C1-6 alkyl- N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, C1-6 hydroxyalkyl, C1-6 haloalkyl; and wherein optionally two substituents on the same carbon atom of L come together to form a C3-C6 carbocycle or 3- to 8-membered heterocycle, wherein the C3-C6 carbocycle and 3- to 8- membered heterocycle are each optionally substituted with one or more substituents independently selected from halogen, -OH, -NO2, =0, =S, -CN, C1-6 alkyl-N(R20)2, C1-6 aminoalkyl, C1-6 alkoxy, Ci-e hydroxyalkyl, and C1-6 haloalkyl; each R20 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, - N(CI-6 alkyl)2, C1-10 alkyl, -C1-10 haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12- membered heterocycle; each R21 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, -
N(CI-6 alkyl)2, Ci-io alkyl, -Ci-io haloalkyl, -O-Ci-io alkyl, oxo, C3-12 carbocycle, and 3- to 12- membered heterocycle; each R23 is independently selected from hydrogen; and C1-6 alkyl, C2-6 alkenyl, C2-6 alkynyl, C3-12 carbocycle, and 3- to 12-membered heterocycle, each of which is optionally substituted with one or more substituents independently selected from halogen, -OH, -CN, -NO2, -NH2, - N(CI-6 alkyl)2, C1-10 alkyl, -Ci-io haloalkyl, -O-Ci-10 alkyl, oxo, C3-12 carbocycle, and 3- to 12- membered heterocycle.
381. A method of treating a cancer in a subj ect in need thereof, comprising administering to the subject a therapeutically effective amount of a combination of a PD-l/PD- L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, or a pharmaceutical composition thereof; and a compound selected from:
pharmaceutically acceptable salt of any one thereof, or a pharmaceutical composition of any one thereof.
382. A method of treating a subject identified or diagnosed as having a cancer that include (a) detecting resistance of the cancer in the subject to treatment with a PD-1/PD-L1 inhibitor that was previously administered to the patient; and (b) after (a), administering to the subject a therapeutically effective amount of a combination of a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab,
atezolizumab, avelumab, and durvalumab, or a pharmaceutical composition thereof; and a
acceptable salt of any one thereof, or a pharmaceutical composition of any one thereof.
383. A method of treating a subject identified or diagnosed as having a cancer and determined to have previously developed resistance to treatment with a KRAS G12D inhibitor that include administering to the subject a therapeutically effective amount of a combination of a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, or a pharmaceutical
pharmaceutically acceptable salt of any one thereof, or a pharmaceutical composition of any one thereof.
384. A method of treating a cancer in a subject in need thereof, comprising administering to the subject a therapeutically effective amount of a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, or a pharmaceutical composition thereof; and a
eutically acceptable salt of any one thereof, or a pharmaceutical composition of any one thereof.
385. A method of treating a subject identified or diagnosed as having a cancer that include (a) detecting resistance of the cancer in the subject to treatment with a PD-1/PD-L1 inhibitor that was previously administered to the patient; and (b) after (a), administering to the subject a therapeutically effective amount of a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, or a pharmaceutical composition thereof, and a compound selected from:
one thereof, or a pharmaceutical composition of any one thereof.
386. A method of treating a subject identified or diagnosed as having a cancer and determined to have previously developed resistance to treatment with a KRAS G12D inhibitor that include administering to the subject a therapeutically effective amount of a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, or a pharmaceutical composition
pharmaceutically acceptable salt of any one thereof, or a pharmaceutical composition of any one thereof.
387. The method of any one of claims 380 to 386, wherein the compound is pharmaceutically acceptable salt thereof. hod of any one of claims 380 to 387, wherein the compound is
harmaceutically acceptable salt thereof.
393. The method of any one of claims 380 to 387, wherein the PD-1/PD-L1 inhibitor is a PD-1 inhibitor.
394. The method of any one of claims 380 to 387, wherein the PD-1 inhibitor is selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, and tislelizumab.
395. The method of any one of claims 380 to 387, wherein the PD-1 inhibitor is nivolumab.
396. The method of any one of claims 380 to 387, wherein the PD-1 inhibitor is pembrolizumab.
397. The method of any one of claims 380 to 387, wherein the wherein the PD-1 inhibitor is cemiplimab.
398. The method of any one of claims 380 to 387, wherein the PD-1 inhibitor is tislelizumab.
399. The method of any one of claims 380 to 387, wherein the PD-1/PD-L1 inhibitor is a PD-Ll inhibitor.
400. The method of any one of claims 380 to 387, wherein the PD-L1 inhibitor is selected from the group consisting of atezolizumab, avelumab, and durvalumab.
401. The method of any one of claims 380 to 387, wherein the PD-L1 inhibitor is atezolizumab.
402. The method of any one of claims 380 to 387, wherein the PD-L1 inhibitor is avelumab.
403. The method of any one of claims 380 to 387, wherein the PD-L1 inhibitor is durvalumab.
404. The method of any one of claims 380 to 387, wherein the PD-1/PD-L1 inhibitor and the compound, or a pharmaceutically acceptable salt thereof are administered on the same day.
405. The method of any one of claims 380 to 387, wherein the PD-1/PD-L1 inhibitor and the compound, or a pharmaceutically acceptable salt thereof are administered on different days.
406. The method of any one of claims 380 to 387, wherein the cancer is a KRas G12D- associated cancer.
407. The method of any one of claims 380 to 387, wherein the cancer is a KRas G12V- associated cancer.
408. The method of any one of claims 380 to 407, wherein the cancer is selected from:
Cardiac: sarcoma (angiosarcoma, fibrosarcoma, rhabdomyosarcoma, liposarcoma), myxoma, rhabdomyoma, fibroma, lipoma and teratoma;
Lung: bronchogenic carcinoma (squamous cell, undifferentiated small cell, undifferentiated large cell, adenocarcinoma), alveolar (bronchiolar) carcinoma, bronchial adenoma, sarcoma, lymphoma, chondromatous hamartoma, mesothelioma;
Gastrointestinal: esophagus (squamous cell carcinoma, adenocarcinoma, leiomyosarcoma, lymphoma), stomach (carcinoma, lymphoma, leiomyosarcoma), pancreas (ductal adenocarcinoma, insulinoma, glucagonoma, gastrinoma, carcinoid tumors, vipoma), small bowel (adenocarcinoma, lymphoma, carcinoid tumors, Kaposi's sarcoma, leiomyoma, hemangioma, lipoma, neurofibroma, fibroma), large bowel (adenocarcinoma, tubular adenoma, villous adenoma, hamartoma, leiomyoma);
Genitourinary tract: kidney (adenocarcinoma, Wilm's tumor (nephroblastoma), lymphoma, leukemia), bladder and urethra (squamous cell carcinoma, transitional cell carcinoma, adenocarcinoma), prostate (adenocarcinoma, sarcoma), testis (seminoma, teratoma, embryonal carcinoma, teratocarcinoma, choriocarcinoma, sarcoma, interstitial cell carcinoma, fibroma, fibroadenoma, adenomatoid tumors, lipoma);
Liver: hepatoma (hepatocellular carcinoma), cholangiocarcinoma, hepatoblastoma, angiosarcoma, hepatocellular adenoma, hemangioma;
Biliary tract: gall bladder carcinoma, ampullary carcinoma, cholangiocarcinoma;
Bone: osteogenic sarcoma (osteosarcoma), fibrosarcoma, malignant fibrous histiocytoma, chondrosarcoma, Ewing's sarcoma, malignant lymphoma (reticulum cell sarcoma), multiple myeloma, malignant giant cell tumor chordoma, osteochronfroma (osteocartilaginous exostoses), benign chondroma, chondroblastoma, chondromyxofibroma, osteoid osteoma and giant cell tumors;
Nervous system: skull (osteoma, hemangioma, granuloma, xanthoma, osteitis deformans), meninges (meningioma, meningiosarcoma, gliomatosis), brain (astrocytoma, medulloblastoma, glioma, ependymoma, germinoma (pinealoma), glioblastoma multiform, oligodendroglioma, schwannoma, retinoblastoma, congenital tumors), spinal cord neurofibroma, meningioma, glioma, sarcoma);
Gynecological: uterus (endometrial carcinoma), cervix (cervical carcinoma, pre-tumor cervical dysplasia), ovaries (ovarian carcinoma (serous cystadenocarcinoma, mucinous cystadenocarcinoma, unclassified carcinoma), granulosa-thecal cell tumors, Sertoli-Leydig cell tumors, dysgerminoma, malignant teratoma), vulva (squamous cell carcinoma, intraepithelial carcinoma, adenocarcinoma, fibrosarcoma, melanoma), vagina (clear cell carcinoma, squamous cell carcinoma, botryoid sarcoma (embryonal rhabdomyosarcoma), fallopian tubes (carcinoma);
Hematologic: blood (myeloid leukemia (acute and chronic), acute lymphoblastic leukemia, chronic lymphocytic leukemia, myeloproliferative diseases, multiple myeloma, myelodysplastic syndrome), Hodgkin's disease, non-Hodgkin's lymphoma (malignant lymphoma);
Skin: malignant melanoma, basal cell carcinoma, squamous cell carcinoma, Kaposi's sarcoma, moles dysplastic nevi, lipoma, angioma, dermatofibroma, keloids, psoriasis; and
Adrenal glands: neuroblastoma.
409. The method of any one of claims 380 to 408, wherein the cancer is non-small cell lung cancer, small cell lung cancer, colorectal cancer, rectal cancer or pancreatic cancer.
410. The method of any one of claims 380 to 408, wherein the cancer is non-small cell lung cancer.
411. The method of any one of claims 380 to 408, wherein the cancer is small cell lung cancer.
412. The method of any one of claims 380 to 408, wherein the cancer is colorectal cancer.
413. The method of any one of claims 380 to 408, wherein the cancer is rectal cancer.
414. The method of any one of claims 380 to 408, wherein the cancer is pancreatic cancer.
415. The method of any one of claims 380 to 408, wherein the cancer is a solid tumor cancer.
416. A kit comprising: a) a pharmaceutical composition comprising a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, and b) a pharmaceutical composition
comprising
pharmaceutically acceptable salt thereof, for treating a cancer in a subject.
417. A kit comprising: a) a pharmaceutical composition comprising a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, and b) a pharmaceutical composition comprising
pharmaceutically acceptable salt thereof, for treating a cancer in a subject.
418. A kit comprising: a) a pharmaceutical composition comprising a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, and b) a pharmaceutical composition comprising
pharmaceutically acceptable salt thereof, for treating a cancer in a subject.
419. A kit comprising: a) a pharmaceutical composition comprising a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, and b) a pharmaceutical composition
comprising
pharmaceutically acceptable salt thereof, for treating a cancer in a subject.
420. A kit comprising: a) a pharmaceutical composition comprising a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, and b) a pharmaceutical composition comprising
pharmaceutically acceptable salt thereof, for treating a cancer in a subject.
421. A kit comprising: a) a pharmaceutical composition comprising a PD-1/PD-L1 inhibitor selected from the group consisting of nivolumab, pembrolizumab, cemiplimab, tislelizumab, atezolizumab, avelumab, and durvalumab, and b) a pharmaceutical composition comprising
pharmaceutically acceptable salt thereof, for treating a cancer in a subject.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363600221P | 2023-11-17 | 2023-11-17 | |
| US63/600,221 | 2023-11-17 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025106905A1 true WO2025106905A1 (en) | 2025-05-22 |
Family
ID=93843623
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/056253 Pending WO2025106905A1 (en) | 2023-11-17 | 2024-11-15 | Combination therapies with a kras modulator and an immunomodulator inhibitor |
Country Status (1)
| Country | Link |
|---|---|
| WO (1) | WO2025106905A1 (en) |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025255438A1 (en) | 2024-06-07 | 2025-12-11 | Revolution Medicines, Inc. | Methods of treating a ras protein-related disease or disorder |
| WO2025265060A1 (en) | 2024-06-21 | 2025-12-26 | Revolution Medicines, Inc. | Therapeutic compositions and methods for managing treatment-related effects |
| WO2026006747A1 (en) | 2024-06-28 | 2026-01-02 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2026015796A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015790A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015801A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015825A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Use of ras inhibitor for treating pancreatic cancer |
Citations (16)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5846514A (en) | 1994-03-25 | 1998-12-08 | Isotechnika, Inc. | Enhancement of the efficacy of nifedipine by deuteration |
| US6334997B1 (en) | 1994-03-25 | 2002-01-01 | Isotechnika, Inc. | Method of using deuterated calcium channel blockers |
| US20090217401A1 (en) | 2005-05-09 | 2009-08-27 | Medarex, Inc | Human Monoclonal Antibodies To Programmed Death 1(PD-1) And Methods For Treating Cancer Using Anti-PD-1 Antibodies Alone or in Combination with Other Immunotherapeutics |
| US20110123550A1 (en) | 2008-07-04 | 2011-05-26 | Ono Pharmaceutical Co., Ltd. | Use of an efficacy marker for optimizing therapeutic efficacy of an anti-human pd-1 antibody on cancers |
| US20140234296A1 (en) | 2011-03-31 | 2014-08-21 | Merck Sharp & Dohme Corp. | Stable formulations of antibodies to human programmed death receptor pd-1 and related treatments |
| US20140271684A1 (en) | 2007-07-23 | 2014-09-18 | Medarex, Inc. | Pd-1 antibodies in combination with a cytokine-secreting cell and methods of use thereof |
| US20150079109A1 (en) | 2013-09-13 | 2015-03-19 | Beigene, Ltd. | Anti-PD1 Antibodies and their Use as Therapeutics and Diagnostics |
| US20150232555A1 (en) | 2007-06-18 | 2015-08-20 | Merck Sharp & Dohme B.V. | Antibodies to human programmed death receptor pd-1 |
| US20160304969A1 (en) | 2013-12-17 | 2016-10-20 | Merck Sharp & Dohme Corp. | Ifn-gamma gene signature biomarkers of tumor response to pd-1 antagonists |
| US20160305947A1 (en) | 2013-12-10 | 2016-10-20 | Merck Sharp & Dohme Corp. | Immunohistochemical proximity assay for pd-1 positive cells and pd-ligand positive cells in tumor tissue |
| US20180148790A1 (en) | 2015-04-17 | 2018-05-31 | Merck Sharp & Dohme Corp. | Blood-based biomarkers of tumor sensitivity to pd-1 antagonists |
| WO2018098352A2 (en) * | 2016-11-22 | 2018-05-31 | Jun Oishi | Targeting kras induced immune checkpoint expression |
| US20180237524A1 (en) | 2015-02-27 | 2018-08-23 | Merck Sharp & Dohme Corp. | Crystals of anti-human pd-1 monoclonal antibodies |
| US20180327848A1 (en) | 2014-12-09 | 2018-11-15 | Merck Sharp & Dohme Corp. | System and methods for deriving gene signature biomarkers of response to pd-1 antagonists |
| WO2022241265A1 (en) * | 2021-05-14 | 2022-11-17 | Syndax Pharmaceuticals, Inc. | Inhibitors of the menin-mll interaction |
| WO2023230190A1 (en) * | 2022-05-25 | 2023-11-30 | Quanta Therapeutics, Inc. | Pyrimidine based modulators and uses thereof |
-
2024
- 2024-11-15 WO PCT/US2024/056253 patent/WO2025106905A1/en active Pending
Patent Citations (21)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US6334997B1 (en) | 1994-03-25 | 2002-01-01 | Isotechnika, Inc. | Method of using deuterated calcium channel blockers |
| US5846514A (en) | 1994-03-25 | 1998-12-08 | Isotechnika, Inc. | Enhancement of the efficacy of nifedipine by deuteration |
| US20090217401A1 (en) | 2005-05-09 | 2009-08-27 | Medarex, Inc | Human Monoclonal Antibodies To Programmed Death 1(PD-1) And Methods For Treating Cancer Using Anti-PD-1 Antibodies Alone or in Combination with Other Immunotherapeutics |
| US20130133091A1 (en) | 2005-05-09 | 2013-05-23 | Ono Pharmaceutical Co., Ltd. | Human Monoclonal Antibodies To Programmed Death 1 (PD-1) And Methods For Treating Cancer Using Anti-PD-1 Antibodies Alone Or In Combination With Other Immunotherapeutics |
| US20140294852A1 (en) | 2005-05-09 | 2014-10-02 | Ono Pharmaceutical Co., Ltd. | Monoclonal antibodies to programmed death 1 (pd-1) |
| US20140348743A1 (en) | 2005-05-09 | 2014-11-27 | Ono Pharmaceutical Co., Ltd. | Monoclonal antibodies to programmed death 1 (pd-1) |
| US20160304606A9 (en) | 2007-06-18 | 2016-10-20 | Merck Sharp & Dohme B.V. | Antibodies to human programmed death receptor pd-1 |
| US20180111996A1 (en) | 2007-06-18 | 2018-04-26 | Merck Sharp & Dohme B.V. | Antibodies to human programmed death receptor pd-1 |
| US20150232555A1 (en) | 2007-06-18 | 2015-08-20 | Merck Sharp & Dohme B.V. | Antibodies to human programmed death receptor pd-1 |
| US20140271684A1 (en) | 2007-07-23 | 2014-09-18 | Medarex, Inc. | Pd-1 antibodies in combination with a cytokine-secreting cell and methods of use thereof |
| US20110123550A1 (en) | 2008-07-04 | 2011-05-26 | Ono Pharmaceutical Co., Ltd. | Use of an efficacy marker for optimizing therapeutic efficacy of an anti-human pd-1 antibody on cancers |
| US20140234296A1 (en) | 2011-03-31 | 2014-08-21 | Merck Sharp & Dohme Corp. | Stable formulations of antibodies to human programmed death receptor pd-1 and related treatments |
| US20150079109A1 (en) | 2013-09-13 | 2015-03-19 | Beigene, Ltd. | Anti-PD1 Antibodies and their Use as Therapeutics and Diagnostics |
| US20160305947A1 (en) | 2013-12-10 | 2016-10-20 | Merck Sharp & Dohme Corp. | Immunohistochemical proximity assay for pd-1 positive cells and pd-ligand positive cells in tumor tissue |
| US20160304969A1 (en) | 2013-12-17 | 2016-10-20 | Merck Sharp & Dohme Corp. | Ifn-gamma gene signature biomarkers of tumor response to pd-1 antagonists |
| US20180327848A1 (en) | 2014-12-09 | 2018-11-15 | Merck Sharp & Dohme Corp. | System and methods for deriving gene signature biomarkers of response to pd-1 antagonists |
| US20180237524A1 (en) | 2015-02-27 | 2018-08-23 | Merck Sharp & Dohme Corp. | Crystals of anti-human pd-1 monoclonal antibodies |
| US20180148790A1 (en) | 2015-04-17 | 2018-05-31 | Merck Sharp & Dohme Corp. | Blood-based biomarkers of tumor sensitivity to pd-1 antagonists |
| WO2018098352A2 (en) * | 2016-11-22 | 2018-05-31 | Jun Oishi | Targeting kras induced immune checkpoint expression |
| WO2022241265A1 (en) * | 2021-05-14 | 2022-11-17 | Syndax Pharmaceuticals, Inc. | Inhibitors of the menin-mll interaction |
| WO2023230190A1 (en) * | 2022-05-25 | 2023-11-30 | Quanta Therapeutics, Inc. | Pyrimidine based modulators and uses thereof |
Non-Patent Citations (36)
| Title |
|---|
| "Curr., Pharm. Des.", vol. 6, 2000, article "Recent Advances in the Synthesis and Applications of Radiolabeled Compounds for Drug Discovery and Development", pages: 110 |
| "Pharmaceutical Dosage Forms and Drug Delivery Systems", 1999, LIPPINCOTT WILLIAMS & WILKINS |
| "Pharmaceutical Dosage Forms", 1980, MARCEL DECKER |
| "The AACR Project GENIE Consortium", CANCER DISCOVERY, vol. 7, no. 8, 2017, pages 818 - 831 |
| AGATA ET AL., INT IMMUNOL, vol. 8, 1996, pages 765 - 72 |
| ANGEW. CHEM. INT. ED., vol. 60, 2021, pages 7360 - 7365 |
| BIOORGANIC AND MEDICINAL CHEMISTRY, vol. 20, no. 3, 2012, pages 1319 - 1336 |
| COELHO ET AL., IMMUNITY, vol. 47, no. 6, 2017, pages 1083 - 1099 |
| CULLY, M.J. DOWNWARD: "SnapShot: Ras Signaling", CELL, vol. 133, no. 7, 2008, pages 1292 - 1292 |
| DONG ET AL., J. MOL. MED., vol. 81, 2003, pages 281 - 7 |
| DONG ET AL., NAT. MED., vol. 8, 2002, pages 787 - 9 |
| DROSTEN, M. ET AL.: "Genetic analysis of Ras signalling pathways in cell proliferation, migration and survival", EMBO J, vol. 29, no. 6, 2010, pages 1091 - 104, XP093208809, DOI: 10.1038/emboj.2010.7 |
| EDWARD B. ROCHE: "Bioreversible Carriers in Drug Design", 1987, AMERICAN PHARMACEUTICAL ASSOCIATION AND PERGAMON PRESS |
| EL-JAWHARI ET AL., MOLECULAR IMMUNOLOGY, vol. 58, no. 2, 2014, pages 160 - 168 |
| EVANS, E. ANTHONY: "Synthesis of radiolabeled compounds", J. RADIOANAL. CHEM., vol. 64, no. 1-2, 1981, pages 9 - 32 |
| FEDORAK ET AL., AM. J. PHYSIOL., vol. 269, 1995, pages 210 - 218 |
| FREEMAN ET AL., J EXP MED, vol. 192, 2000, pages 1027 - 34 |
| GEORGE W.VARMA, RAJENDER S: "The Synthesis of Radiolabeled Compounds via Organometallic Intermediates", TETRAHEDRON, vol. 45, no. 21, 1989, pages 6601 - 21 |
| HOCHHAUS ET AL., BIOMED. CHROM., vol. 6, 1992, pages 283 - 286 |
| HOOVER, JOHN E.: "Remington's Pharmaceutical Sciences", 1975, MACK PUBLISHING CO. |
| J. LARSEN ET AL., INT. J. PHARMACEUTICS, vol. 47, 1988, pages 103 |
| J. LARSENH. BUNDGAARD, INT. J. PHARMACEUTICS, vol. 37, 1987, pages 87 |
| JEAN JACQUESANDRE COLLETSAMUEL H. WILEN: "Enantiomers, Racemates and Resolutions", 1981, JOHN WILEY AND SONS, INC. |
| L. FIESERM. FIESER, FIESER AND FIESER'S REAGENTS FOR ORGANIC SYNTHESIS, 1994 |
| LIAO ET AL., CANCER CELL, vol. 35, 2019, pages 559 - 572 |
| MCLOED ET AL., GASTROENTEROL, vol. 106, 1994, pages 405 - 413 |
| R. LAROCK, COMPREHENSIVE ORGANIC TRANSFORMATIONS, 1989 |
| REMINGTON: "Encyclopedia of Reagents for Organic Synthesis", 1995, MACK PUBLISHING COMPANY |
| REMINGTON: "The Science and Practice of Pharmacy", 2005, MACK PUB. CO. |
| ROWE ET AL.: "Handbook of Pharmaceutical Excipients: A Comprehensive Guide to Uses, Properties, and Safety", 2006 |
| SINKULA ET AL., J. PHARM. SCI., vol. 64, 1975, pages 181 - 210 |
| STEPHEN, A.G. ET AL.: "Dragging ras back in the ring", CANCER CELL, vol. 25, no. 3, 2014, pages 272 - 81, XP028633347, DOI: 10.1016/j.ccr.2014.02.017 |
| SUNAGA ET AL., INT. J. CANCER, vol. 130, no. 8, 2012, pages 1733 - 1744 |
| T. HIGUCHI, V. STELLA: "A.C.S. Symposium Series", PRO-DRUGS AS NOVEL DELIVERY SYSTEMS, vol. 14 |
| T. W. GREENE, P. G. M. WUTS: "Protective Groups in Organic Synthesis", 1991 |
| WINTER GEBUCKLEY DLPAULK JROBERTS JMSOUZA ADHE-PAGANON SBRADNER JE: "Phthalimide conjugation as a strategy for in vivo target protein degradation. Science", DRUG DEVELOPMENT, vol. 348, no. 6241, 19 June 2015 (2015-06-19), pages 1376 - 81, XP055328122, DOI: 10.1126/science.aab1433 |
Cited By (7)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| WO2025255438A1 (en) | 2024-06-07 | 2025-12-11 | Revolution Medicines, Inc. | Methods of treating a ras protein-related disease or disorder |
| WO2025265060A1 (en) | 2024-06-21 | 2025-12-26 | Revolution Medicines, Inc. | Therapeutic compositions and methods for managing treatment-related effects |
| WO2026006747A1 (en) | 2024-06-28 | 2026-01-02 | Revolution Medicines, Inc. | Ras inhibitors |
| WO2026015796A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015790A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015801A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Methods of treating a ras related disease or disorder |
| WO2026015825A1 (en) | 2024-07-12 | 2026-01-15 | Revolution Medicines, Inc. | Use of ras inhibitor for treating pancreatic cancer |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7791361B2 (en) | Pyrimidine-based modulators and uses thereof | |
| JP7713603B2 (en) | KRAS MODULATORS AND USES THEREOF | |
| WO2025106905A1 (en) | Combination therapies with a kras modulator and an immunomodulator inhibitor | |
| US12421254B2 (en) | KRAS modulators and uses thereof | |
| EP4010080B1 (en) | Heterocyclic compounds for use in the treatment of cancer | |
| WO2022256459A1 (en) | Kras modulators and uses thereof | |
| AU2015359626B2 (en) | 1,3-thiazol-2-yl substituted benzamides | |
| TWI723511B (en) | Highly active sting protein agonist compound | |
| CA3185888A1 (en) | Brm targeting compounds and associated methods of use | |
| WO2025034883A1 (en) | Combination therapies with kras modulators | |
| WO2024259169A1 (en) | Pyrimidine based modulators and uses thereof | |
| KR20210089662A (en) | Spiro-sulfonamide derivatives as inhibitors of myeloid cell leukemia-1 (MCL-1) protein | |
| WO2025106901A1 (en) | Combination cancer therapies with a kras modulator and an rtk-mapk pathway inhibitor | |
| US12263171B2 (en) | 7-, 8-, and 10-substituted amino triazolo quinazoline derivatives as adenosine receptor antagonists, pharmaceutical compositions and their use | |
| WO2025049402A1 (en) | Kras modulators and uses thereof | |
| CA3080249A1 (en) | Novel pyrazolo-pyrrolo-pyrimidine-dione derivatives as p2x3 inhibitors | |
| CN115697972A (en) | Receptor-interacting protein 1 inhibitors including piperazine heterocyclic amide ureas | |
| WO2025034919A1 (en) | Combination therapies with kras modulators |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24821213 Country of ref document: EP Kind code of ref document: A1 |