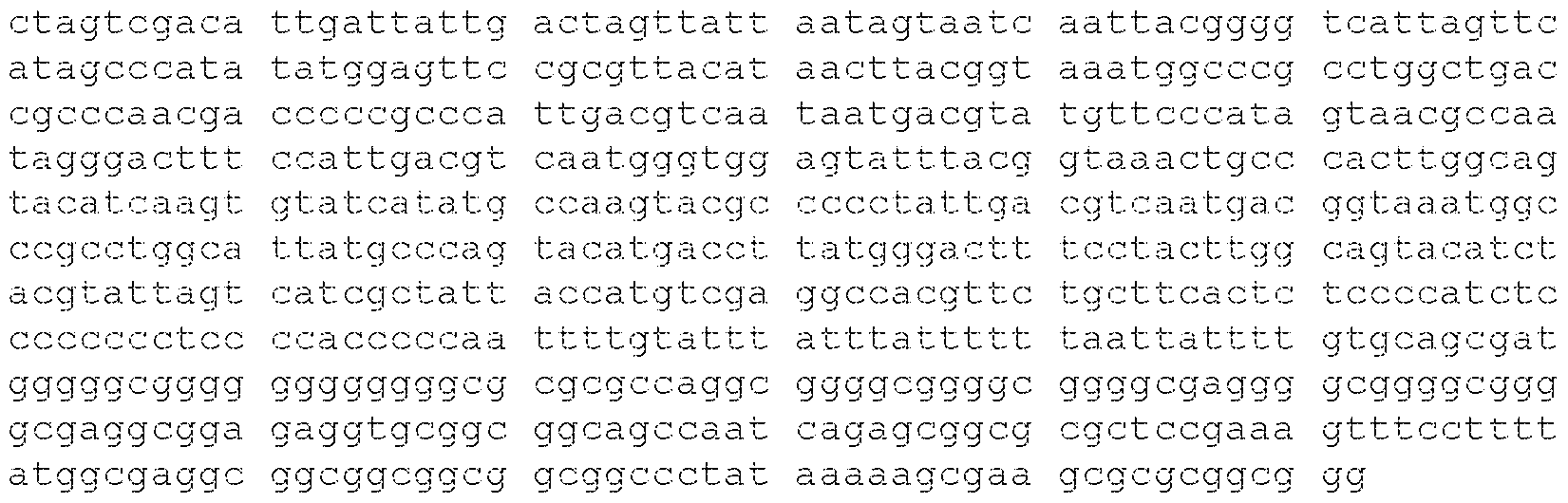WO2025038800A1 - Compositions and methods for the treatment of disorders related to frataxin deficiency - Google Patents
Compositions and methods for the treatment of disorders related to frataxin deficiency Download PDFInfo
- Publication number
- WO2025038800A1 WO2025038800A1 PCT/US2024/042405 US2024042405W WO2025038800A1 WO 2025038800 A1 WO2025038800 A1 WO 2025038800A1 US 2024042405 W US2024042405 W US 2024042405W WO 2025038800 A1 WO2025038800 A1 WO 2025038800A1
- Authority
- WO
- WIPO (PCT)
- Prior art keywords
- seq
- amino acid
- acid sequence
- nucleotide sequence
- sequence
- Prior art date
- Legal status (The legal status is an assumption and is not a legal conclusion. Google has not performed a legal analysis and makes no representation as to the accuracy of the status listed.)
- Pending
Links
Classifications
-
- C—CHEMISTRY; METALLURGY
- C07—ORGANIC CHEMISTRY
- C07K—PEPTIDES
- C07K14/00—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof
- C07K14/005—Peptides having more than 20 amino acids; Gastrins; Somatostatins; Melanotropins; Derivatives thereof from viruses
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
- A61K48/005—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy characterised by an aspect of the 'active' part of the composition delivered, i.e. the nucleic acid delivered
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K38/00—Medicinal preparations containing peptides
-
- A—HUMAN NECESSITIES
- A61—MEDICAL OR VETERINARY SCIENCE; HYGIENE
- A61K—PREPARATIONS FOR MEDICAL, DENTAL OR TOILETRY PURPOSES
- A61K48/00—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy
- A61K48/0075—Medicinal preparations containing genetic material which is inserted into cells of the living body to treat genetic diseases; Gene therapy characterised by an aspect of the delivery route, e.g. oral, subcutaneous
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N15/00—Mutation or genetic engineering; DNA or RNA concerning genetic engineering, vectors, e.g. plasmids, or their isolation, preparation or purification; Use of hosts therefor
- C12N15/09—Recombinant DNA-technology
- C12N15/63—Introduction of foreign genetic material using vectors; Vectors; Use of hosts therefor; Regulation of expression
- C12N15/79—Vectors or expression systems specially adapted for eukaryotic hosts
- C12N15/85—Vectors or expression systems specially adapted for eukaryotic hosts for animal cells
- C12N15/86—Viral vectors
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/14011—Parvoviridae
- C12N2750/14111—Dependovirus, e.g. adenoassociated viruses
- C12N2750/14122—New viral proteins or individual genes, new structural or functional aspects of known viral proteins or genes
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/14011—Parvoviridae
- C12N2750/14111—Dependovirus, e.g. adenoassociated viruses
- C12N2750/14141—Use of virus, viral particle or viral elements as a vector
- C12N2750/14143—Use of virus, viral particle or viral elements as a vector viral genome or elements thereof as genetic vector
-
- C—CHEMISTRY; METALLURGY
- C12—BIOCHEMISTRY; BEER; SPIRITS; WINE; VINEGAR; MICROBIOLOGY; ENZYMOLOGY; MUTATION OR GENETIC ENGINEERING
- C12N—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA
- C12N2750/00—MICROORGANISMS OR ENZYMES; COMPOSITIONS THEREOF; PROPAGATING, PRESERVING, OR MAINTAINING MICROORGANISMS; MUTATION OR GENETIC ENGINEERING; CULTURE MEDIA ssDNA viruses
- C12N2750/00011—Details
- C12N2750/14011—Parvoviridae
- C12N2750/14111—Dependovirus, e.g. adenoassociated viruses
- C12N2750/14141—Use of virus, viral particle or viral elements as a vector
- C12N2750/14145—Special targeting system for viral vectors
Definitions
- AAV adeno-associated virus
- compositions described herein may be used to treat a subject in need thereof, such as a human subject diagnosed with FA, or as a research tool in the study of diseases or conditions in cells or animal models of FA.
- Friedreich’s Ataxia is an autosomal recessive inherited disease that causes progressive damage to the nervous system.
- FA typically results from the degeneration of nervous tissue in the spinal cord due to reduced expression of the mitochondrial protein frataxin (FXN; also known as, e.g., CyaY, FA, FARR, FRDA, and X25) in sensory neurons that direct muscle movement of the arms and legs.
- FXN mitochondrial protein frataxin
- Onset usually occurs at puberty or by age 25. See Campuzano, et al., Science, 271.5254 (Mar 8, 1996): 1423.
- FA initial symptoms include poor coordination such as gait disturbance, poor balance, leg weakness, decreased walking, impaired coordination, dysarthria, nystagmus, impaired sensation, kyphoscoliosis, and foot deformities.
- poor coordination such as gait disturbance, poor balance, leg weakness, decreased walking, impaired coordination, dysarthria, nystagmus, impaired sensation, kyphoscoliosis, and foot deformities.
- FA is also associated with scoliosis, heart disease, and diabetes. The disease generally progresses until a wheelchair is required for mobility.
- Incidence of FA among Caucasian populations is between about 1 in 20,000 and about 1 in 50,000, with a deduced carrier frequency of about 1 in 120 in European populations. See Nageshwaran and Festenstein, Frontiers in Neurology, Vol.
- Omaveloxolone is a semisynthetic oleanane triterpenoid that activates Nrf2, a master transcription factor that regulates genes with antioxidative, anti-inflammatory, and mitochondrial bioenergetic properties.
- Nrf2 a master transcription factor that regulates genes with antioxidative, anti-inflammatory, and mitochondrial bioenergetic properties.
- Adeno-associated viruses AAVs have emerged as a widely studied and utilized viral particles for delivery of therapeutically effective polypeptides to mammalian cells. See, e.g., Tratschin et al., Mol.
- compositions and methods directed to AAV- based gene delivery of FXN e.g., human FXN
- FXN e.g., human FXN
- the compositions and methods can be used to slow, halt, or reverse symptoms of FA.
- the present disclosure provides an AAV particle comprising an AAV capsid and a nucleotide sequence encoding a FXN protein, also referred to herein as a FXN-encoding sequence or as a FXN protein-encoding sequence.
- the nucleotide sequence encoding a FXN protein is comprised in a viral genome.
- the FXN protein is a human FXN protein.
- the FXN protein is a wildtype human FXN protein.
- the AAV particle comprises a viral genome encoding the FXN protein (e.g., a human FXN protein) and the AAV capsid is an AAV capsid variant.
- the viral genome comprises a truncated chicken ⁇ -actin (CBA) promoter operably linked to the FXN-encoding sequence, and the AAV capsid variant is an AAV5 capsid variant.
- CBA truncated chicken ⁇ -actin
- the AAV capsid variant (e.g., an AAV5 capsid variant) comprises a peptide that has replaced amino acid position 577 as numbered according to SEQ ID NO: 138 (in other words, the peptide has replaced the amino acid corresponding to position 577 of SEQ ID NO: 138).
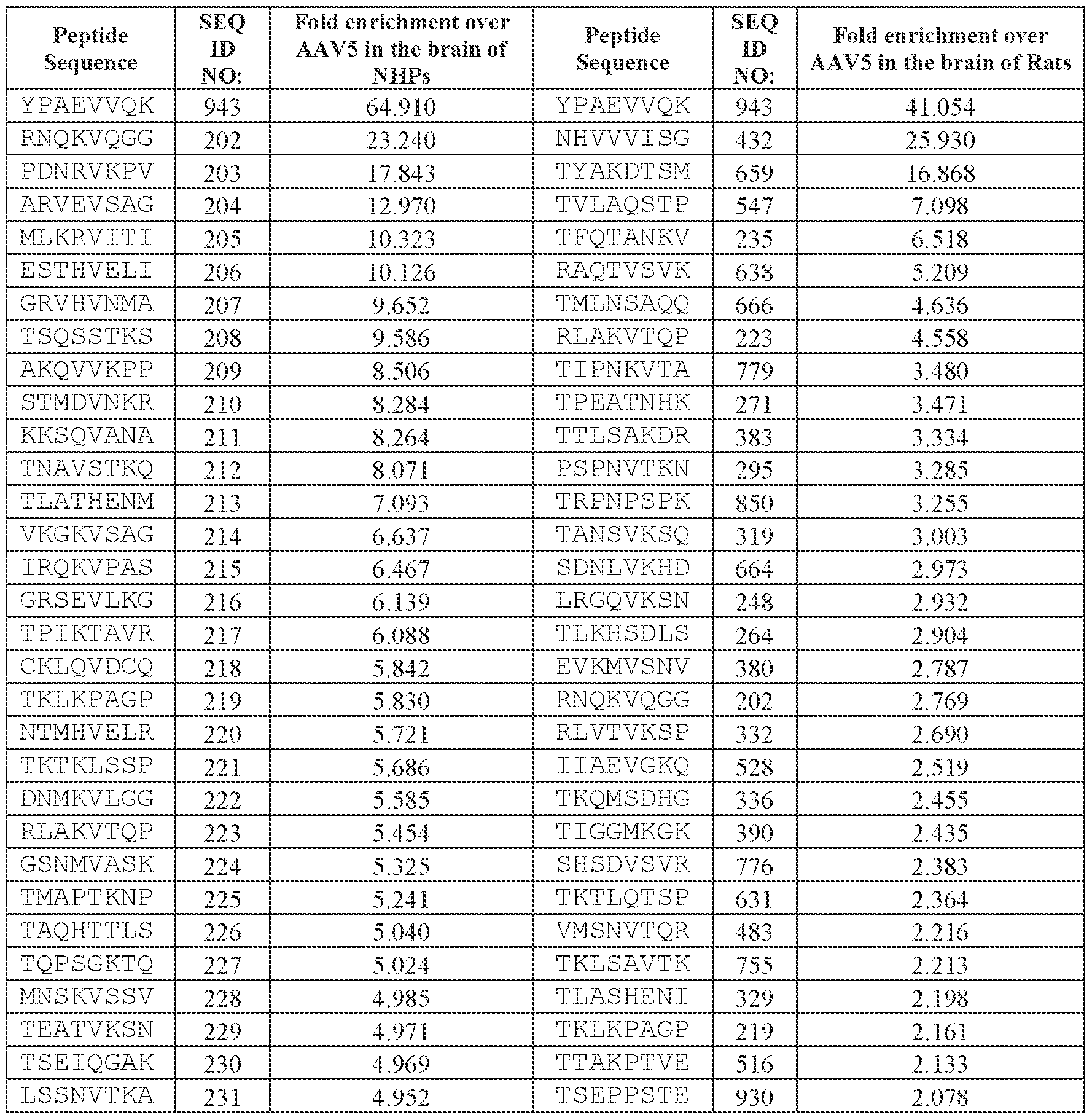
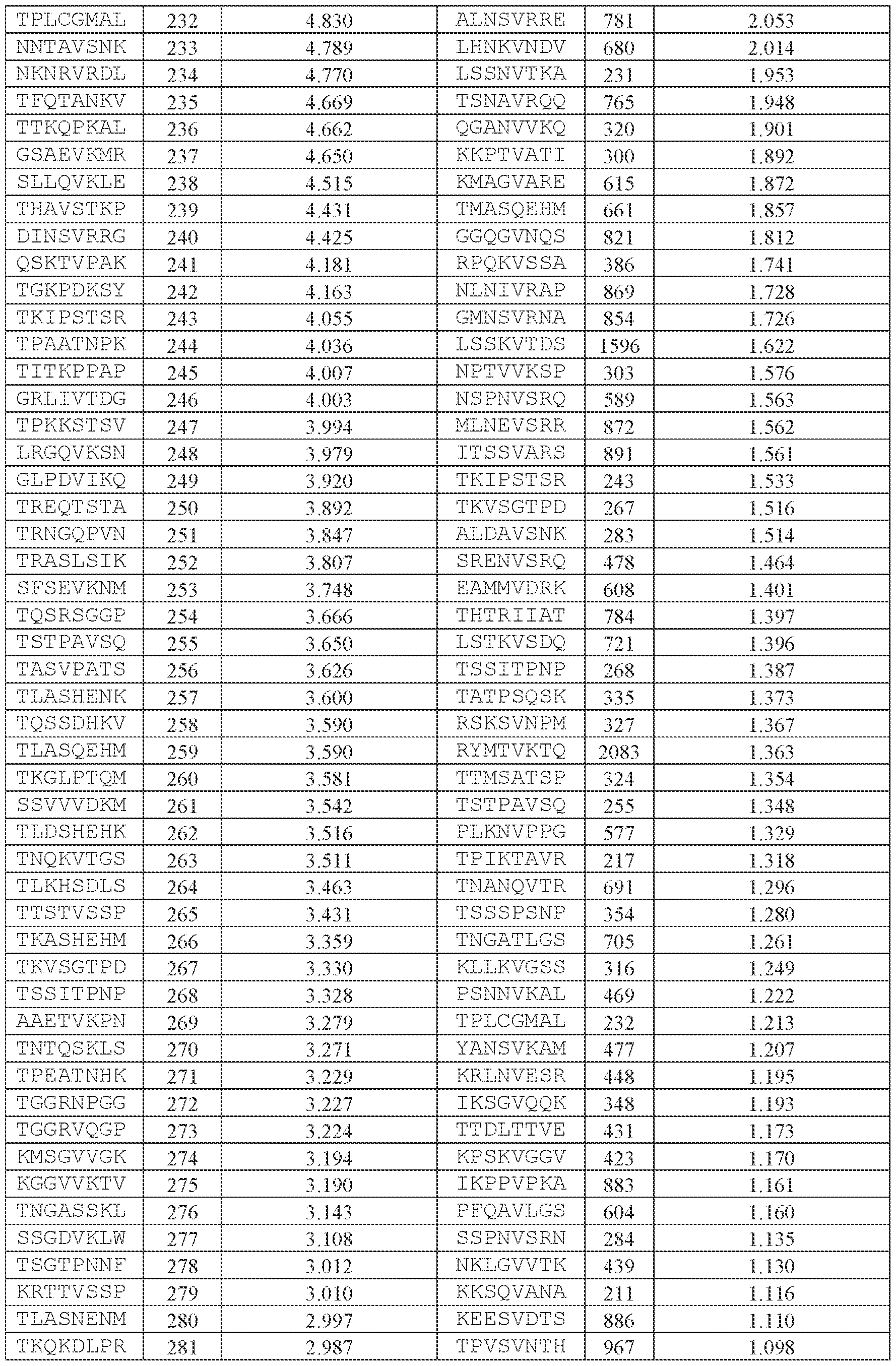
- the AAV capsid variant is an AAV5 capsid variant comprising a peptide comprising the amino acid sequence of VQK in loop VIII.
- the AAV capsid variant comprises a peptide comprising the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) in loop VIII, or a Attorney Docket No.14640.0092-00304 peptide comprising one, two, or three—but no more than three—substitutions relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943).
- the peptide comprising the amino acid sequence of SEQ ID NO: 943 has replaced a threonine at amino acid position 577 as numbered according to SEQ ID NO: 138 (in other words, the peptide comprising the amino acid sequence comprising no more than three substitutions relative to SEQ ID NO: 943 has replaced the amino acid corresponding to position 577 of SEQ ID NO: 138).
- the AAV capsid variant comprises a peptide comprising the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) in loop VIII.
- the peptide comprising the amino acid sequence of SEQ ID NO: 943 has replaced a threonine at amino acid position 577 as numbered according to SEQ ID NO: 138 (in other words, the peptide comprising the amino acid sequence of SEQ ID NO: 943 has replaced the amino acid corresponding to position 577 of SEQ ID NO: 138).
- the peptide is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant. In some embodiments, the peptide is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant.
- the present disclosure provides an adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome, wherein the viral genome comprises a frataxin (FXN)-encoding sequence and the AAV capsid variant comprises an amino acid sequence having the formula [N2]-[N3], wherein: (i) [N2] comprises X1, X2, X3, X4, and X5, wherein X1 is Y, N, or C; X2 is P, K, T, or Q; X 3 is A or P; X 4 is E, S, or A; and X 5 is V, L, or E; and (ii) [N3] comprises the amino acid sequence VQK, EQK, VKK, VHK, VQQ, or LQK; and wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 739, or an amino acid sequence at least 95% identical thereto.
- AAV capsid variant comprises the amino acid sequence of SEQ ID NO
- [N2]-[N3] is present in loop VIII, wherein loop VIII is present at amino acids comprising those corresponding to positions 571-599 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, [N2]-[N3] is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 982.
- the AAV capsid variant is an AAV5 capsid variant comprising [N2]- [N3] in place of an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138.
- [N2] comprises the amino acid sequence YP, YPA, YPAE (SEQ ID NO: 21), or YPAEV (SEQ ID NO: 1).
- [N3] comprises the amino acid sequence VQK.
- [N2]-[N3] comprises the amino acid sequence AEVVQK (SEQ ID NO: 36) or PAEVVQK (SEQ ID NO: 20).
- [N2]-[N3] comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943).
- the AAV capsid variant further comprises [N1] comprising X D , X E , and X F , wherein: X D of [N1] is Q, T, S, A, I, L, or H; X E of [N1] is S, G, A, or R; and X F of [N1] is S, K, L, R, A, or T; wherein [N1] immediately precedes [N2]-[N3].
- [N1] comprises the amino acid sequence QS, SS, or QSS.
- [N1]-[N2] comprises the amino acid sequence QSSYPAEV (SEQ ID NO: 96).
- [N1]-[N2]-[N3] comprises the amino acid sequence SSYPAEVVQ (SEQ ID NO: 121) or QSSYPAEVVQK (SEQ ID NO: 150).
- the AAV capsid variant further comprises [N0] comprising X A , X B , and X C , wherein: X A of [N0] is T, I, or N; X B of [N0] is N; and X C of [N0] is N, T, S, or K; wherein [N0] immediately precedes [N1].
- [N0] comprises the amino acid sequence TN, NN, or TNN.
- [N0]-[N1] comprises the amino acid sequence TNNQSS (SEQ ID NO: 183).
- [N0]-[N1]-[N2]-[N3] comprises the amino acid sequence TNNQSSYPAEVVQK (SEQ ID NO: 500).
- the AAV capsid variant further comprises [N4] comprising X G and X H , wherein: X G of [N4] is T, P, or N; and (b) X H of [N4] is A; wherein [N4] is present immediately subsequent to [N3].
- [N4] comprises the amino acid sequence TA.
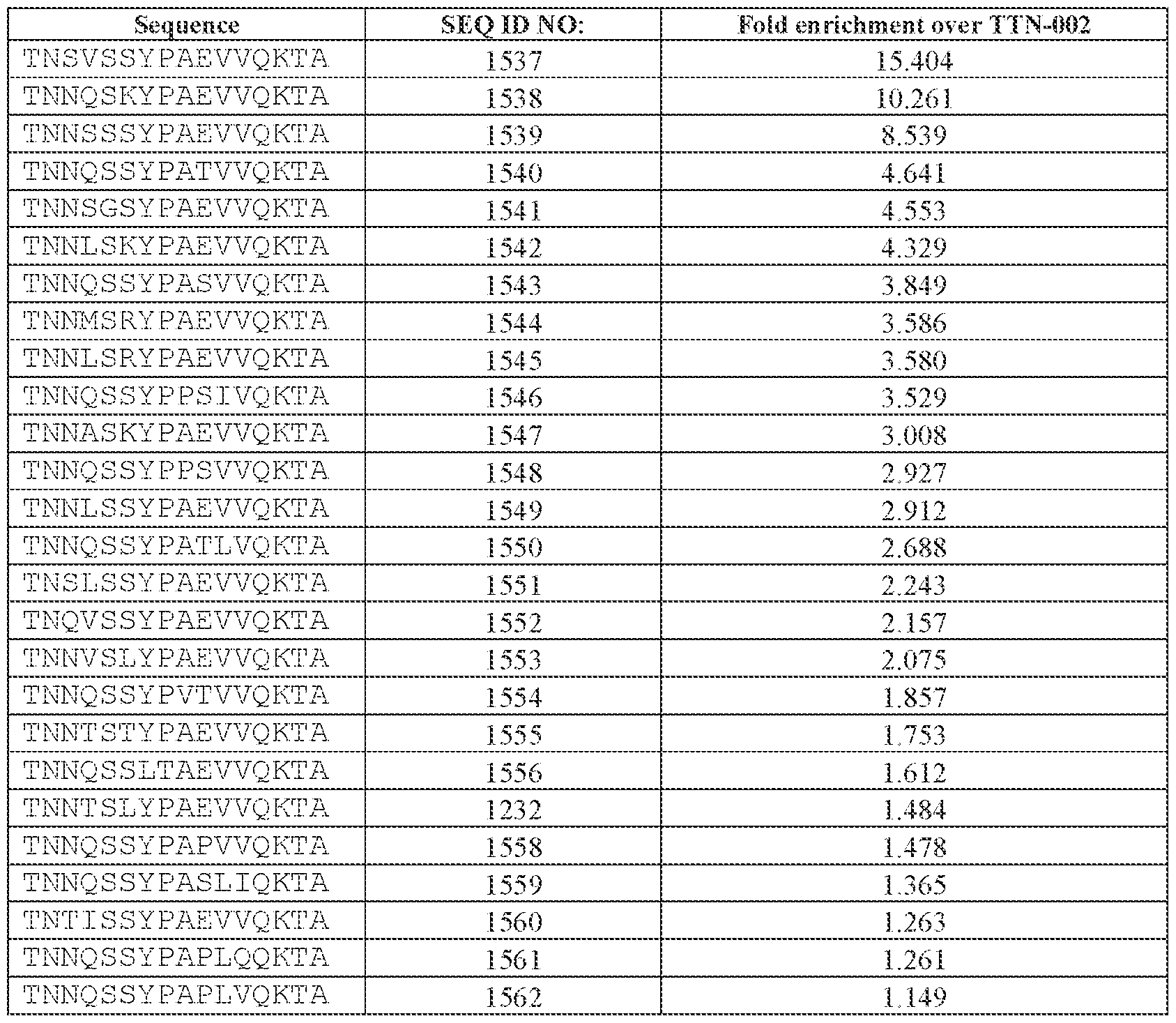
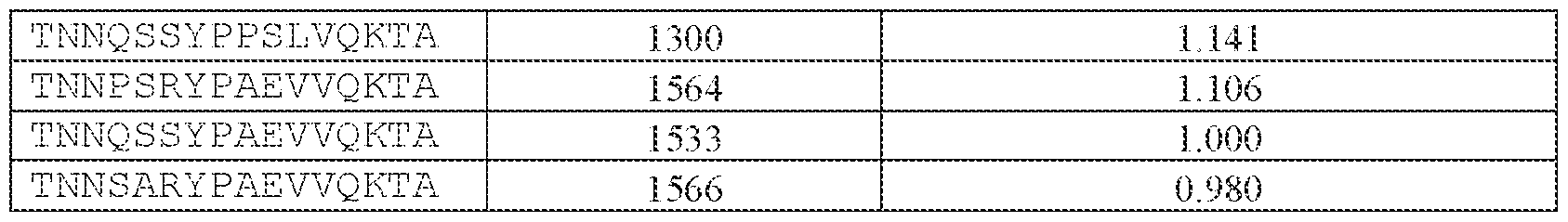
- [N0]-[N1]-[N2]-[N3]-[N4] comprises the amino acid sequence TNNQSSYPAEVVQKTA (SEQ ID NO: 1533).
- [017] In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is present in loop VIII, wherein loop VIII is present at amino acids comprising those corresponding to positions 571-599 of the amino acid sequence of SEQ ID NO: 982.
- the present disclosure provides an adeno-associated virus (AAV) particle comprising: (i) a viral genome comprising a frataxin (FXN)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% identical to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises at least 4, at least 5, at least 6, at least 7, or all 8 consecutive amino acids from the amino acid sequence YPAEVVQK (SEQ ID NO: 943).
- AAV adeno-associated virus
- the at least 4 consecutive amino acids comprise the amino acid sequence YPAE (SEQ ID NO: 21), optionally present at amino acids corresponding to positions 577-580 of the amino acid sequence of SEQ ID NO: 982;
- the at least 5 consecutive amino acids comprise the amino acid sequence YPAEV (SEQ ID NO: 1), optionally present at amino acids corresponding to positions 577-581 of the amino acid sequence of SEQ ID NO: 982;
- the at least 6 consecutive amino acids comprise the amino acid sequence YPAEVV (SEQ ID NO: 725), optionally present at amino acids corresponding to positions 577-582 of the amino acid sequence of SEQ ID NO: 982; or
- the at least 7 consecutive amino acids comprise the amino acid sequence YPAEVVQ (SEQ ID NO: 726), optionally present at amino acids corresponding to positions 577-583 of the amino acid sequence of SEQ ID NO: 982.
- the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943) present at amino acids corresponding to positions 577-584 of the amino acid sequence of SEQ ID NO: 982.
- the present disclosure provides an adeno-associated virus (AAV) particle comprising: (i) a viral genome comprising a frataxin (FXN)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% identical to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943) at amino acids corresponding to positions 577-584 of the amino acid sequence of SEQ ID NO: 982.
- AAV adeno-associated virus
- the AAV capsid variant further comprises (i) an amino acid sequence having at least 95% identity to the amino acid sequence of SEQ ID NO: 738, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); and/or (ii) an amino acid sequence having at least 95% identity to the amino acid sequence of SEQ ID NO: 982, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943).
- the present disclosure provides an adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome, wherein the viral genome comprises a frataxin (FXN)-encoding sequence and the AAV capsid variant comprises (i) an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 982, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); (ii) an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 738, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); and/or (iii) an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943).
- AAV adeno-associated virus
- the AAV capsid variant comprises (i) the amino acid sequence of SEQ ID NO: 982; (ii) the amino acid sequence of SEQ ID NO: 738; and/or (iii) the amino acid sequence of SEQ ID NO: 739.
- the FXN-encoding sequence encodes a FXN protein that is not a cynomolgus monkey FXN protein. In some embodiments, the FXN-encoding sequence encodes a human FXN protein.
- the human FXN protein comprises the amino acid sequence of SEQ ID NO: 1825, or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the human FXN protein is a wildtype human FXN protein.
- the FXN-encoding sequence comprises a nucleotide sequence encoding the amino acid sequence of SEQ ID NO: 1825.
- the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
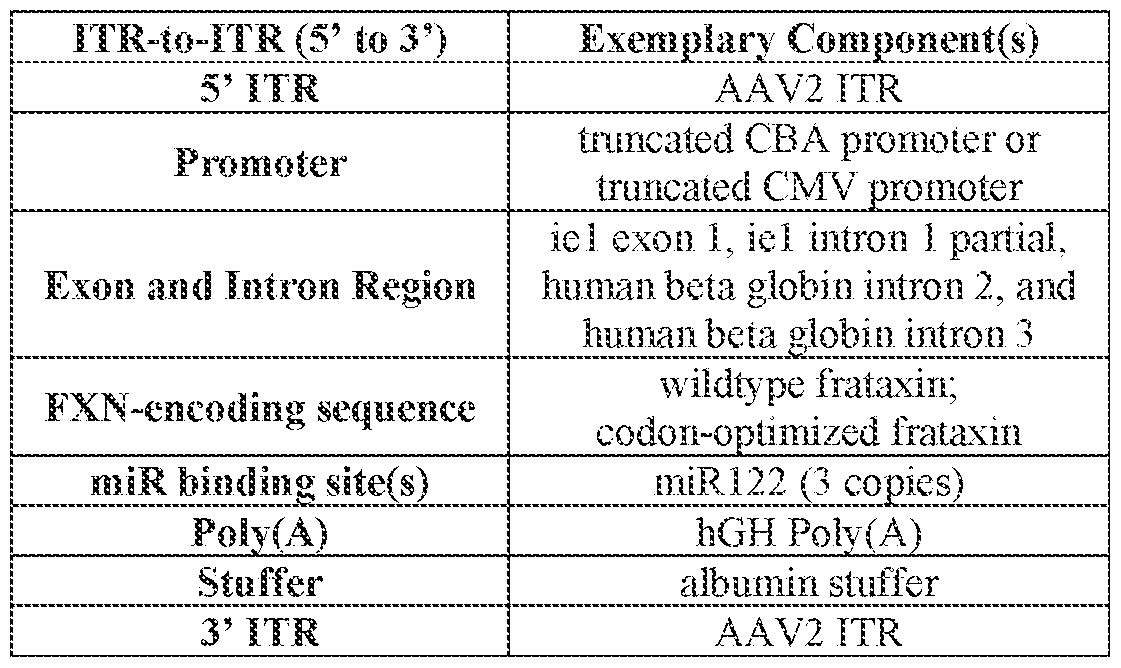
- the viral genome further comprises a promoter operably linked to the FXN-encoding sequence.
- the promoter comprises a human elongation factor 1 ⁇ - subunit (EF1 ⁇ ) promoter, a cytomegalovirus (CMV) immediate-early enhancer and/or promoter, a chicken ⁇ -actin (CBA) promoter, a CAG promoter, a ⁇ glucuronidase (GUSB) promoter, a ubiquitin C Attorney Docket No.14640.0092-00304 (UBC) promoter, a neuron-specific enolase (NSE) promoter, a platelet-derived growth factor (PDGF) promoter, a platelet-derived growth factor B-chain (PDGF- ⁇ ) promoter, a intercellular adhesion molecule 2 (ICAM-2) promoter, a synapsin (Syn) promoter, a methyl-CpG binding protein 2 (MeCP2) promoter, a Ca2+/calmodulin-dependent protein kinase II (CaMKII) promoter, a
- the promoter is a CMV promoter or CBA promoter, or a functional fragment or truncation of a CMV promoter or CBA promoter. [024] In some embodiments, the promoter is a truncated CBA promoter. In some embodiments, the truncated CBA promoter is 50-400 nucleotides in length, e.g., 100-332 nucleotides in length.
- the promoter comprises the nucleotide sequence of any one of SEQ ID NOs: 1738, 1740, and 1742 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- the promoter is a truncated CMV promoter. In some embodiments, the truncated CMV promoter is 50-300 nucleotides in length.
- the promoter comprises the nucleotide sequence of SEQ ID NO: 1750 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
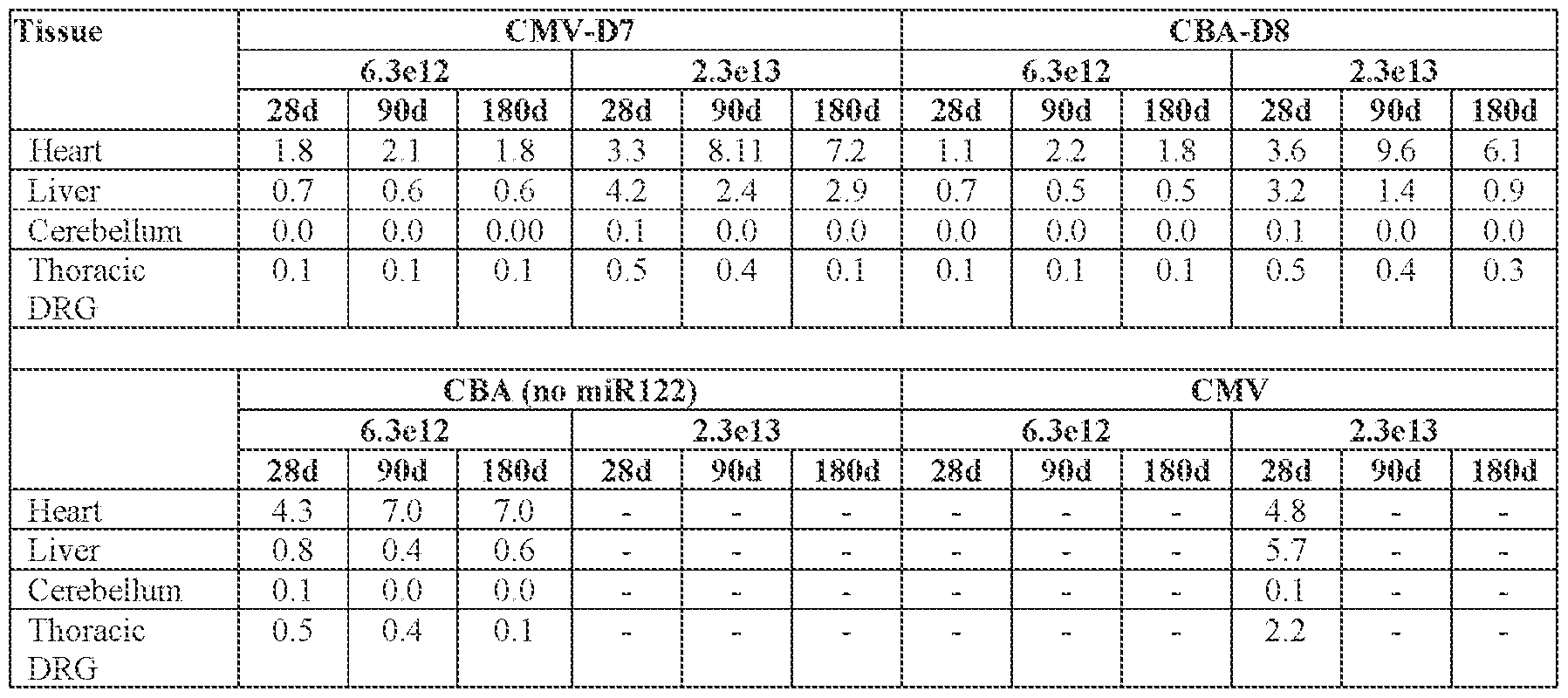
- the viral genome further comprises a microRNA (miR) binding site that modulates expression of the encoded FXN protein in a cell or tissue of the liver.
- the viral genome comprises 3 copies of the miR binding site. In some embodiments, the 3 copies of the miR binding site are identical.
- the 3 copies of the miR binding site are continuous.
- the miR binding site is a miR122 binding site.
- the miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1827 or a sequence having one, two, three, or at most four substitutions relative to the nucleotide sequence of SEQ ID NO: 1827.
- the viral genome comprises 3 copies of a miR122 binding site comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence having one, two, three, four, five, six, seven, eight, nine, or at most ten substitutions relative to the nucleotide sequence of SEQ ID NO: 1826.
- the viral genome further comprises at least one inverted terminal repeat (ITR) region.
- ITR inverted terminal repeat
- the at least one ITR region comprises an AAV2 ITR.
- the viral genome comprises a 5’ ITR region and a 3’ ITR region.
- Attorney Docket No.14640.0092-00304 the 5’ ITR region and 3’ ITR region are each an AAV2 ITR.
- the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
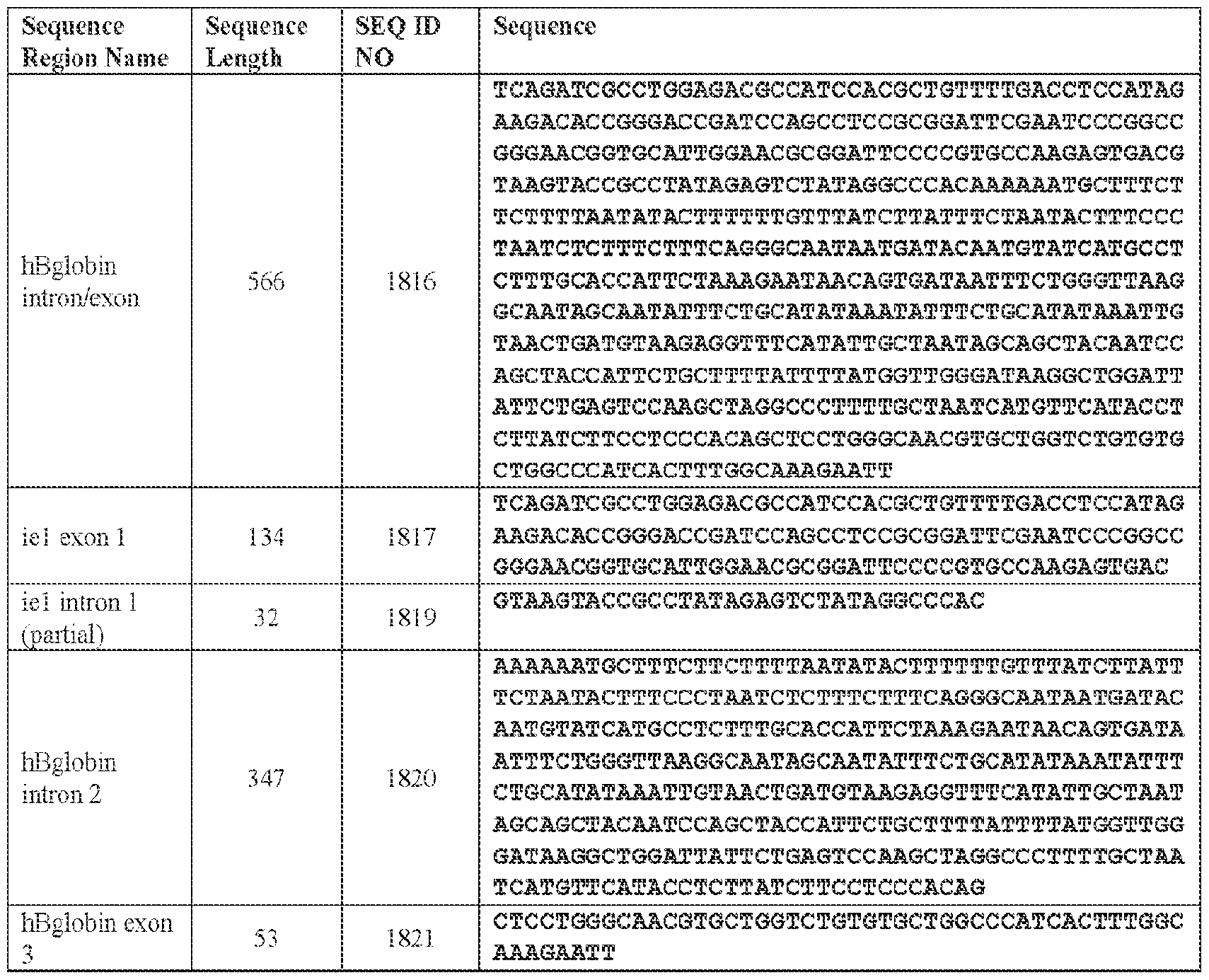
- the viral genome further comprises an intron/exon region comprising an intron region and/or an exon region.
- the intron/exon region comprises: an immediate-early 1 (ie1) intron region and/or a human beta-globin (hBglobin) intron 2 region; and/or an ie1 exon region and/or an hBglobin exon region.
- the intron region comprises: the nucleotide sequence of SEQ ID NO: 1819 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the nucleotide sequence of SEQ ID NO: 1820 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- the nucleotide sequence of SEQ ID NO: 1819 or a sequence that is at least 90% identical e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 9
- the exon region comprises: the nucleotide sequence of SEQ ID NO: 1817 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the nucleotide sequence of SEQ ID NO: 1821 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- the nucleotide sequence of SEQ ID NO: 1817 or a sequence that is at least 90% identical e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 9
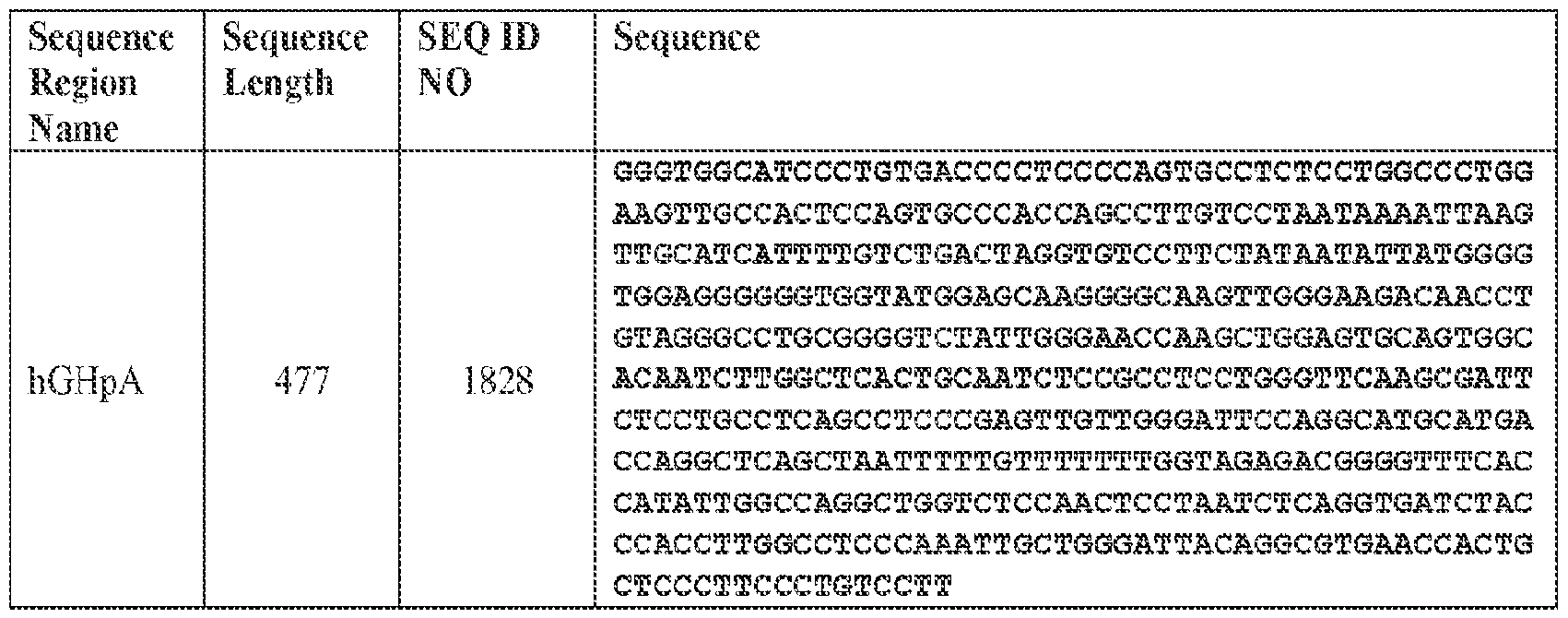
- the viral genome further comprises a polyadenylation (polyA) region.
- the polyA region comprises a human growth hormone (hGH) polyA region.
- the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
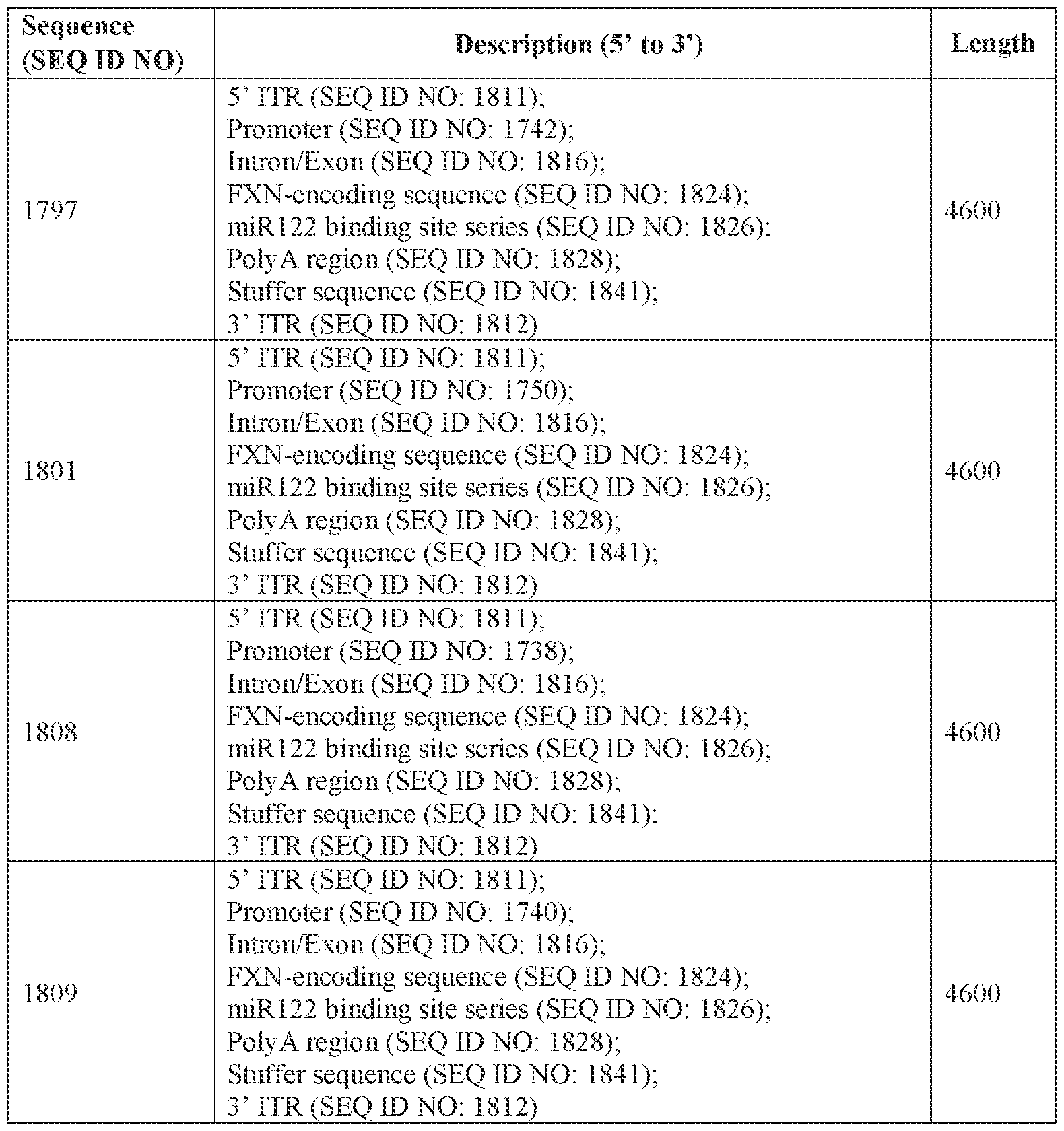
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, Attorney Docket No.14640.0092-00304 at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) the FXN-encoding sequence, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; and (iv) a 3’ ITR region.
- ITR inverted terminal repeat
- the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) the FXN-encoding sequence, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (iv) at least one miR122 binding site; and (v) a 3’ ITR region.
- ITR inverted terminal repeat
- the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron/exon region; (iv) the FXN-encoding sequence, wherein the FXN- encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; and (vi) a 3’ ITR region.
- ITR inverted terminal repeat
- the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron/exon region; (iv) the FXN-encoding sequence, wherein the FXN- encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; and (vii) a 3’ ITR region.
- ITR inverted terminal repeat
- the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron and/or exon region; (iv) the FXN-encoding sequence, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; (vii) a filler sequence; and (viii) a 3’ ITR region.
- ITR inverted terminal repeat
- the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the promoter comprises the nucleotide sequence of SEQ ID NO: 1742 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleot
- the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1841 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region.
- a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1841 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3
- the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the promoter comprises the nucleotide sequence of SEQ ID NO: 1750 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g.
- the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1840 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region.
- a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1840 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3
- the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the promoter comprises the nucleotide sequence of SEQ ID NO: 1738 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g.
- the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: Attorney Docket No.14640.0092-00304 1838 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region.
- a filler sequence comprising the nucleotide sequence of SEQ ID NO: Attorney Docket No.14640.0092-00304 1838 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) there
- the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the promoter comprises the nucleotide sequence of SEQ ID NO: 1740 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g.
- the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1839 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region.
- a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1839 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3
- the viral genome comprises (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, Attorney Docket No.1464
- the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811;
- the promoter comprises the nucleotide sequence of SEQ ID NO: 1742;
- the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816;
- the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824;
- the at least one miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1826;
- the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828; and
- the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812.
- the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1841, wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the viral genome consists of the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides an adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome comprising a frataxin (FXN)-encoding sequence
- the AAV capsid variant comprises: (i) the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); (ii) the amino acid sequence of SEQ ID NO: 738 or an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); or (i) the amino acid sequence of SEQ
- the present disclosure provides an adeno-associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982, the amino acid sequence of SEQ ID NO: 738, and/or the amino acid sequence of SEQ ID NO: 739.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the viral genome is single-stranded.
- the present disclosure provides a cell comprising an AAV particle disclosed herein.
- the cell is a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial cell.
- the present disclosure provides a method of making and AAV particle disclosed herein, wherein the method comprises: (i) providing a cell comprising the viral genome comprising a FXN-encoding sequence and a nucleic acid encoding an AAV capsid variant; and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant; thereby making the AAV particle.
- the viral genome comprises (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 9 least 9
- the viral genome comprises (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%,
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797 and the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982, the amino acid sequence of SEQ ID NO: 738, and/or the amino acid sequence of SEQ ID NO: 739.
- the method further comprises, prior to step (i), introducing a nucleic acid molecule comprising the viral genome into the cell.
- the method further comprises, prior to step (i), introducing the nucleic acid encoding the AAV capsid variant into the cell.
- the cell comprises a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial cell.
- the present disclosure provides a pharmaceutical composition comprising an AAV particle disclosed herein and a pharmaceutically acceptable excipient.
- the present disclosure provides a method of delivering an AAV particle encoding a frataxin (FXN) protein to a cell, comprising administering an effective amount of a pharmaceutical composition or AAV particle described herein.
- the cell is in a subject.
- the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency.
- the disorder is Friedreich’s Ataxia (FA).
- the present disclosure provides a method of delivering an AAV particle encoding a frataxin (FXN) protein to a subject, comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle described herein.
- the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency.
- the disorder is Friedreich’s Ataxia (FA).
- the present disclosure provides a method of treating a subject having or diagnosed with having a disorder associated with frataxin (FXN) deficiency, comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle described herein.
- FXN frataxin
- the subject has, has been diagnosed with having, or is at risk of having Friedreich’s Ataxia (FA).
- FA Friedreich’s Ataxia
- the present disclosure provides a method of treating a subject having or diagnosed with having a disorder, wherein the disorder is Friedreich’s Ataxia (FA), comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle described herein.
- the treating results in prevention of progression of the disorder in the subject. In some embodiments, the treating results in amelioration of at least one symptom of the disorder.
- the at least one symptom comprises impaired sensory functions, impaired motor function (e.g., ataxia and/or involuntary movements), fatigue, chronic pain, seizures, impaired speech, sleep disturbances, metabolic disorders (e.g., diabetes), and/or increased spasticity.
- the treating stabilizes, slows the progression of, or improves the subject’s disorder as determined by the modified Friedreich Ataxia Rating Scale (mFARS), the Scale for the Assessment and Rating of Ataxia (SARA), and/or the International Cooperative Ataxia Rating Scale (ICARS).
- mFARS modified Friedreich Ataxia Rating Scale
- SARA Scale for the Assessment and Rating of Ataxia
- ICARS International Cooperative Ataxia Rating Scale
- the treatment slows the subject’s progression of the disorder as measured by mFARS, SARA, and/or ICARS relative to an individual with the disorder who has not been administered the pharmaceutical composition or the AAV particle.
- the subject is a human.
- the AAV particle or the pharmaceutical composition is delivered to a cell or tissue of the central nervous system (CNS) in the subject.
- CNS central nervous system
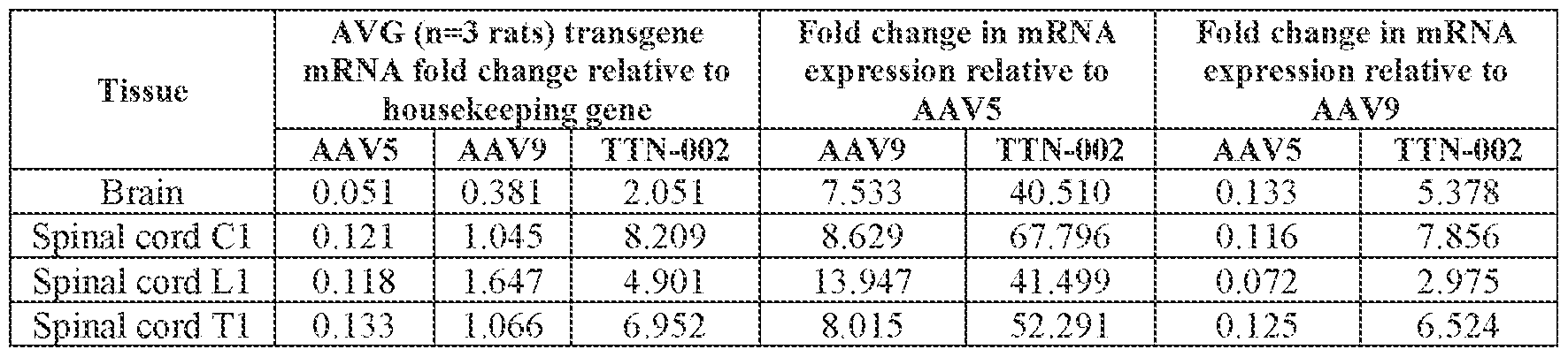
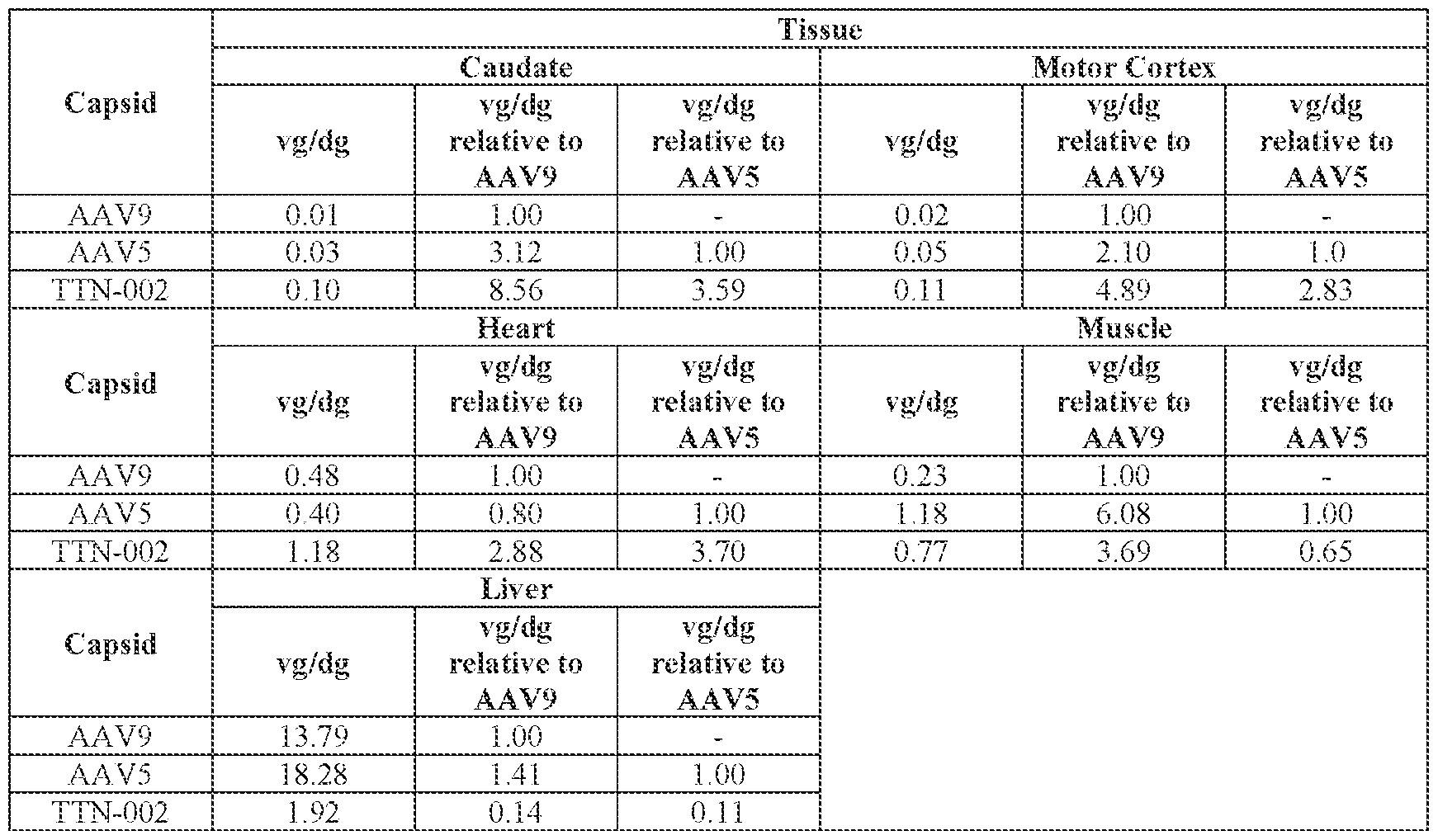
- the cell or tissue of the CNS is a cell or tissue of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum, or a combination thereof.
- the AAV particle or the pharmaceutical composition is delivered to the subject via intravenous administration.
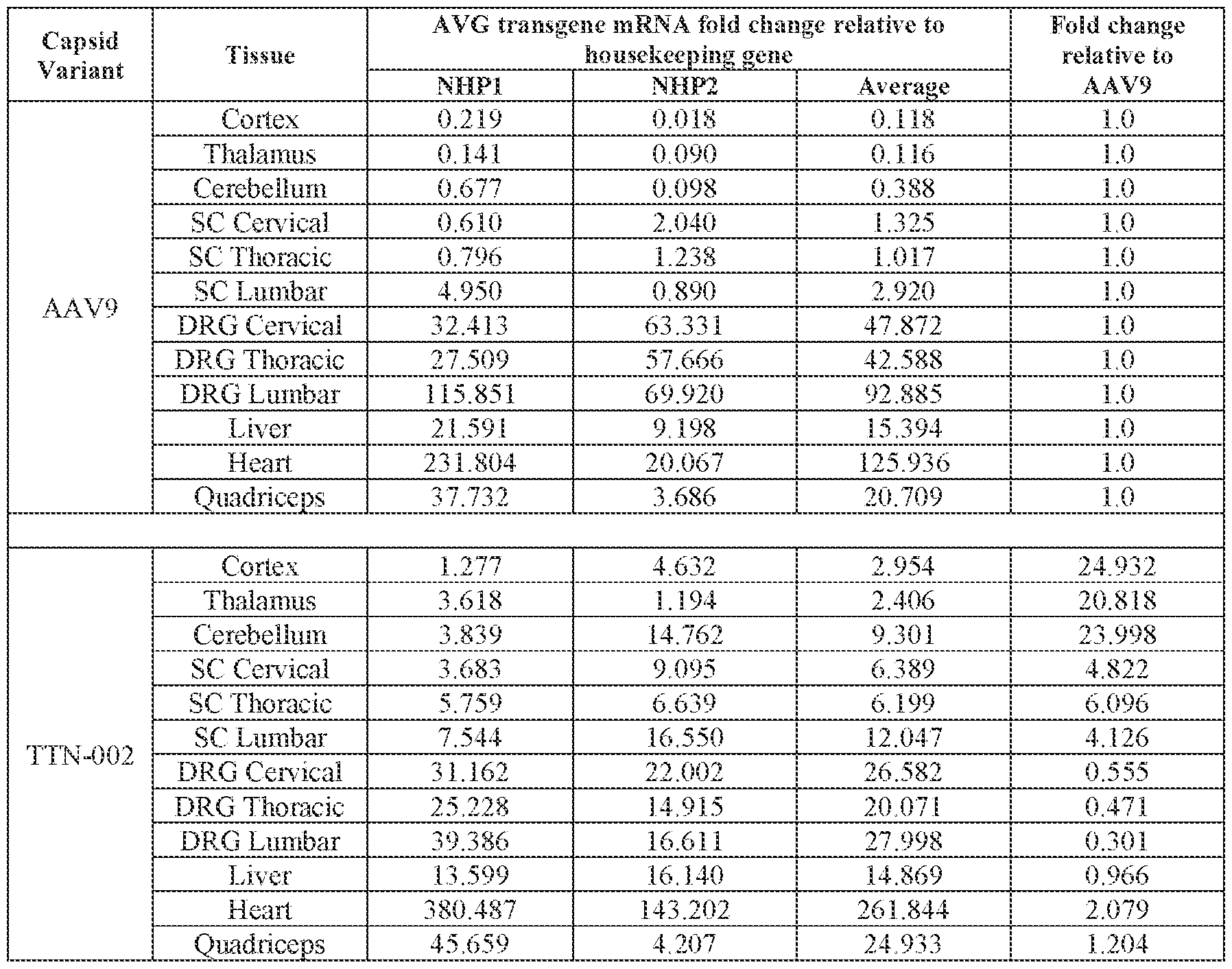
- the method further comprises evaluating, e.g., measuring, the level of FXN expression, e.g., FXN gene expression, FXN mRNA expression, and/or FXN protein expression, in the subject, e.g., in a cell, tissue, or fluid of the subject.
- the level of FXN protein expression is measured by an enzyme-linked immunosorbent assay (ELISA), a Western blot, an immunohistochemistry assay, or a frataxin biofluid assay.
- evaluating the subject’s level of FXN expression is performed before and/or after administration of the pharmaceutical composition or AAV particle.
- the subject’s level of FXN expression before administration is compared to the subject’s level of FXN expression after administration.
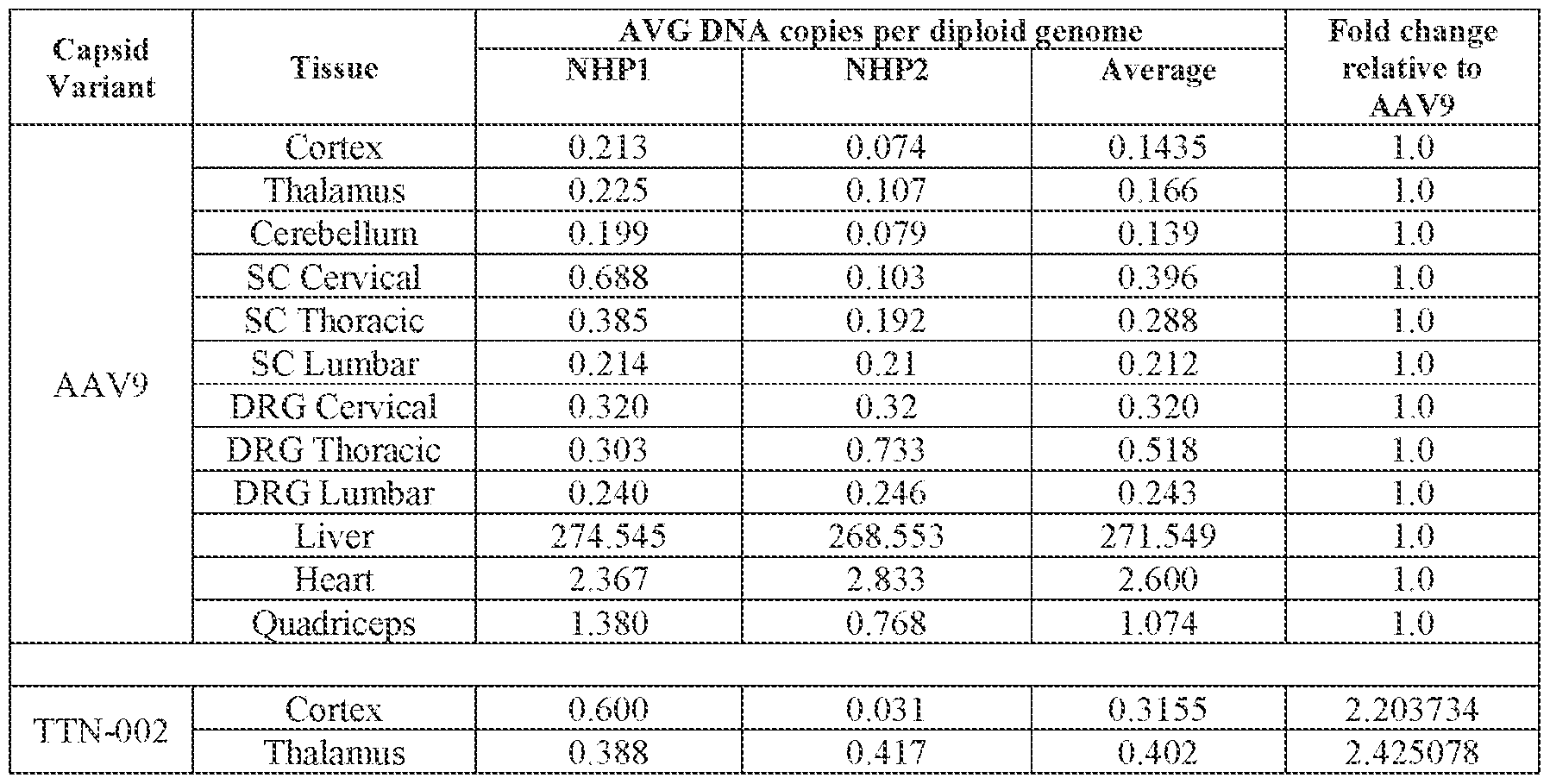
- the method of delivering or treating further comprises evaluating the level of FXN expression in a cell or tissue of the CNS.
- the cell or tissue of the CNS is a cell or tissue of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, Attorney Docket No.14640.0092-00304 caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum.
- the method of delivering or treating further comprises evaluating the level of FXN expression in a peripheral cell or tissue.
- the peripheral cell or tissue is a cell or tissue of the heart and/or muscle.
- the subject’s level of FXN protein expression after administration is increased relative to the subject’s level of FXN protein expression before administration.
- the method of delivering or treating further comprises evaluating, e.g., measuring, the level of FXN protein activity in the subject.
- administering the pharmaceutical composition or AAV particle to the subject results in an increase in: (i) the level of FXN protein or FXN gene expression in a cell, tissue, (e.g., a cell or tissue of the CNS, e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum), and/or fluid (e.g., CSF and/or serum), of the subject relative to baseline; (ii) the number and/or level of viral genomes (VG) per cell in a CNS tissue (e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate
- VG viral genomes
- the method further comprises administering to the subject at least one additional therapeutic agent and/or therapy.
- the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy suitable for treating a disorder associated with FXN deficiency (e.g., Friedreich’s Ataxia).
- the at least one additional therapeutic agent and/or therapy comprises omaveloxolone or idebenone.
- the method further comprises administering an immunosuppressant to the subject.
- the immunosuppressant comprises a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine.
- a corticosteroid e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone
- rapamycin rapamycin
- mycophenolate mofetil tacrolimus
- rituximab rituximab
- eculizumab hydroxychloroquine e.g., eculizumab hydroxychloroquine.
- the present disclosure provides a pharmaceutical composition or AAV particle disclosed herein for use in the treatment of a disorder associated with FXN deficiency in a subject.
- the disorder is Friedreich’s Ataxia (FA).
- the subject has, has been diagnosed with having, or is at risk of having FA.
- the present disclosure provides a use of a pharmaceutical composition or AAV particle disclosed herein in the manufacture of a medicament for the treatment of a disorder associated with FXN deficiency in a subject.
- the disorder is Friedreich’s Ataxia (FA).
- an adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises an amino sequence comprising the following formula: [N2]-[N3], wherein: (i) [N2] comprises X1, X2, X3, X4, and X5, wherein: (a) X 1 is Y, N, C, or T; (b) X 2 is P, E, K, T, or Q; (c) X 3 is A or P; (d) X 4 is E, S, D, or A; and (e) X 5 is V, L, or E; and (ii) [N3] comprises the amino sequence comprising the following formula: [N2]-[N3], wherein: (i) [N2] comprises X1,
- An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises an amino sequence comprising the following formula: [N2]-[N3], wherein: (i) [N2] comprises X1, X2, X3, X4, and X5, wherein: (a) X1 is Y, N, or C; (b) X 2 is P, K, T, or Q; (c) X 3 is A or P; (d) X 4 is E, S, or A; and (e) X 5 is V, L, or E; and (ii) [N3] comprises the amino acid sequence of VQK, EQK, VKK, VHK, VQQ, or LQK.
- FXN frataxin
- An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises one, two, three, four, or all of: (i) an [N0] comprising TNN, TNT, INN, TNS, NNN, or TNK; (ii) an [N1] comprising QSS, QSK, TSL, SSS, QSR, AGA, IGS, QAS, ASS, LGS, QST, HSS, LSS, or QRS; (iii) an [N2] comprising YPAEV (SEQ ID NO: 1), YPPSL (SEQ ID NO: 2), NKAEV (SEQ ID NO: 3), YTAEV (SEQ ID NO: 4),
- AAV particle of embodiment 1 or 2 wherein: (a) X1 is Y or N; (b) X2 is P, T or Q; (c) X 3 is A; (d) X 4 is E or S; and/or (e) X 5 is V or L. 5.
- [N2] comprises YP, NK, YT, YQ, NP, CP, TH, AE, PS, AA, AS, PA, PP, KA, TA, QA, TP, HA, EV, SL, EE, AV, or SH. 6.
- the AAV particle any one of embodiments 1, 2, 4, and 5, wherein [N2] comprises YPA, YPP, NKA, YTA, YQA, YTP, NPA, CPA, THA, PAE, PPS, KAE, TAE, QAE, TPS, PAA, HAS, AEV, PSL, AEE, or AAV. 7.
- the AAV particle of any one of embodiments 1, 2, 4, and 6, wherein [N2] comprises YPAE (SEQ ID NO: 21), YPPS (SEQ ID NO: 22), NKAE (SEQ ID NO: 23), YTAE (SEQ ID NO: 24), YQAE (SEQ ID NO: 25), YTPS (SEQ ID NO: 26), YPAA (SEQ ID NO: 27), NPAE (SEQ ID NO: 28), CPAE (SEQ ID NO: 29), THAS (SEQ ID NO: 30), PAEV (SEQ ID NO: 17), PPSL (SEQ ID NO: 31), KAEV (SEQ ID NO: 32), TAEV (SEQ ID NO: 16), PAEE (SEQ ID NO: 18), QAEV (SEQ ID NO: 15), TPSL (SEQ ID NO: 33), PAAV (SEQ ID NO: 34), or QAEE (SEQ ID NO: 35).
- YPAE SEQ ID NO: 21
- YPPS S
- [N2] is or comprises YPAEV (SEQ ID NO: 1), YPPSL (SEQ ID NO: 2), NKAEV (SEQ ID NO: 3), YTAEV (SEQ ID NO: 4), YPAEE (SEQ ID Attorney Docket No.14640.0092-00304 NO: 5), YQAEV (SEQ ID NO: 6), YTPSL (SEQ ID NO: 7), YPAAV (SEQ ID NO: 8), NPAEV (SEQ ID NO: 9), CPAEV (SEQ ID NO: 10), or YQAEE (SEQ ID NO: 11).
- YPAEV SEQ ID NO: 1
- YPPSL SEQ ID NO: 2
- NKAEV SEQ ID NO: 3
- YTAEV SEQ ID NO: 4
- YPAEE SEQ ID Attorney Docket No.14640.0092-00304 NO: 5
- YQAEV SEQ ID NO: 6
- YTPSL SEQ ID NO: 7
- YPAAV SEQ
- AAV particle of any one of embodiments 1-12 wherein: (i) [N2] is or comprises the amino acid sequence of YTPSL (SEQ ID NO: 7) and [N3] is or comprises the amino acid sequence of VQK; (ii) [N2] is or comprises the amino acid sequence of YPPSL (SEQ ID NO: 2) and [N3] is or comprises the amino acid sequence of VQK; (iii) [N2] is or comprises the amino acid sequence of YPPSL (SEQ ID NO: 2) and [N3] is or comprises the amino acid sequence of EQK; or (iv) [N2] is or comprises the amino acid sequence of YPPSL (SEQ ID NO: 2) and [N3] is or comprises the amino acid sequence of VKK.
- AAV particle of embodiment 1 or 2 or any one of embodiments 4-14, wherein [N2]-[N3] comprises: (i) AEVVQK (SEQ ID NO: 36), PSLVQK (SEQ ID NO: 37), AEVEQK (SEQ ID NO: 38), AEEVQK (SEQ ID NO: 39), PSLEQK (SEQ ID NO: 40), PSLVKK (SEQ ID NO: 41), AEVVKK (SEQ ID NO: 42), AEVVHK (SEQ ID NO: 43), AAVVQK (SEQ ID NO: 44), AEVVQQ (SEQ ID NO: 45), or AEVLQK (SEQ ID NO: 46); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; Attorney Docket No.14640.0092-00304 (iii) an amino acid sequence comprising one, two, or three but no more than four
- [N2]-[N3] comprises: (i) PAEVVQK (SEQ ID NO: 20), PPSLVQK (SEQ ID NO: 47), KAEVVQK (SEQ ID NO: 48), TAEVVQK (SEQ ID NO: 49), PAEVEQK (SEQ ID NO: 50), PAEEVQK (SEQ ID NO: 51), QAEVVQK (SEQ ID NO: 52), TPSLVQK (SEQ ID NO: 53), PPSLEQK (SEQ ID NO: 54), PPSLVKK (SEQ ID NO: 55), PAEVVKK (SEQ ID NO: 56), PAEVVHK (SEQ ID NO: 57), PAAVVQK (SEQ ID NO: 58), PAEVVQQ (SEQ ID NO: 59), TAEVVKK (SEQ ID NO: 60), PAEVLQK (SEQ ID NO: 61), or QAEEVQK (SEQ ID NO: 20), PPSLVQK (SEQ ID NO: 47), KA
- [N2]-[N3] is or comprises: (i) YPAEVVQK (SEQ ID NO: 943), YPPSLVQK (SEQ ID NO: 946), NKAEVVQK (SEQ ID NO: 947), YTAEVVQK (SEQ ID NO: 948), YPAEVEQK (SEQ ID NO: 949), YPAEEVQK (SEQ ID NO: 950), YQAEVVQK (SEQ ID NO: 951), YTPSLVQK (SEQ ID NO: 952), YPPSLEQK (SEQ ID NO: 953), YPPSLVKK (SEQ ID NO: 954), YPAEVVKK (SEQ ID NO: 955), YPAEVVHK (SEQ ID NO: 956), YPAAVVQK (SEQ ID NO: 957), NPAEVVQK (SEQ ID NO: 958), YPAEVV
- AAV particle of any one of embodiments 1-17 which further comprises one, two, three or all of an amino acid other than Q at position 574 (e.g., T, S, A, I, L, or H), an amino acid other than S at Attorney Docket No.14640.0092-00304 position 575 (e.g., G, A, or R), and/or an amino acid other than S at position 576 (e.g., K, L, R, A, or T), numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 19.
- an amino acid other than Q at position 574 e.g., T, S, A, I, L, or H
- an amino acid other than S at Attorney Docket No.14640.0092-00304 position 575 e.g., G, A, or R
- an amino acid other than S at position 576 e.g., K, L, R, A, or T
- the AAV particle of any one of embodiments 1-17 which further comprises: (i) a Q at position 574, an S at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (ii) a T at position 574, an S at position 575, and/or a L at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iii) an S at position 574, an S at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iv) a Q at position 574, an S at position 575, and/or an R at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (v) a Q at position 574, an S at position 575, and/or a K at position 576, numbered according to the amino acid sequence of SEQ ID NO
- AAV particle of any one of embodiments 1-19 which further comprises [N1], wherein [N1] comprises X D , X E , and X F , wherein: (a) X D is Q, T, S, A, I, L, or H; (b) X E is S, G, A, or R; and Attorney Docket No.14640.0092-00304 (c) X F is S, K, L, R, A, or T; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c). 21.
- [N1] comprises X D , X E , and X F , wherein: (a) X D is Q, T, S, A, I, L, or H; (b) X E is S, G, A, or R; and Attorney Docket No.14640.0092-00304 (c) X F is S,
- the AAV particle of embodiment 20, wherein [N1] comprises SK, SL, SS, SR, GA, GS, AS, ST, RS, QS, TS, AG, IG, QA, LG, HS, LS, or QR. 22.
- the AAV particle of any one of embodiments 20-22, wherein [N1]-[N2] comprises: (i) SSYPA (SEQ ID NO: 63), SKYPA (SEQ ID NO: 64), SLYPA (SEQ ID NO: 65), SRYPA (SEQ ID NO: 66), SSYPP (SEQ ID NO: 67), GAYPA (SEQ ID NO: 68), GSYPA (SEQ ID NO: 69), ASYPA (SEQ ID NO: 70), STNKA (SEQ ID NO: 71), SSYTA (SEQ ID NO: 72), SSYQA (SEQ ID NO: 73), SSYTP (SEQ ID NO: 74), SSNPA (SEQ ID NO: 75), SLCPA (SEQ ID NO: 76), RSYTA (SEQ ID NO: 77), or SSTHA (SEQ ID NO: 78); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2,
- AAV particle of any one of embodiments 20-23, wherein [N1]-[N2] comprises: (i) SSYPAE (SEQ ID NO: 79), SKYPAE (SEQ ID NO: 80), SLYPAE (SEQ ID NO: 81), SRYPAE (SEQ ID NO: 82), SSYPPS (SEQ ID NO: 83), GAYPAE (SEQ ID NO: 84), GSYPAE (SEQ ID NO: 85), ASYPAE (SEQ ID NO: 86), STNKAE (SEQ ID NO: 87), SSYTAE (SEQ ID NO: 88), SSYQAE (SEQ ID NO: 89), SSYTPS (SEQ ID NO: 90), SSYPAA (SEQ ID NO: 91), SSNPAE (SEQ ID NO: 92), SLCPAE (SEQ ID NO: 93), RSYTAE (SEQ ID NO: 94), SSTHAS (SEQ ID NO:
- AAV particle of any one of embodiments 20-25, wherein [N1]-[N2]-[N3] comprises: (i) SSYPAEVVQ (SEQ ID NO: 121), SKYPAEVVQ (SEQ ID NO: 122), SLYPAEVVQ (SEQ ID NO: 123), SRYPAEVVQ (SEQ ID NO: 124), SSYPPSLVQ (SEQ ID NO: 125), GAYPAEVVQ (SEQ ID NO: 126), GSYPAEVVQ (SEQ ID NO: 127), ASYPAEVVQ (SEQ ID NO: 128), STNKAEVVQ (SEQ ID NO: 129), SSYTAEVVQ (SEQ ID NO: 130), SKYPAEVEQ (SEQ ID NO: 131), SLYPAEEVQ (SEQ ID NO: 132), SSYQAEVVQ (SEQ ID NO: 133), SSYTPSLVQ (SEQ ID NO: 134), SRYPA
- AAV particle of embodiment 3 or any one of embodiments 20-26, wherein [N1]-[N2]-[N3] is or comprises: (i) QSSYPAEVVQK (SEQ ID NO: 150), QSKYPAEVVQK (SEQ ID NO: 151), TSLYPAEVVQK (SEQ ID NO: 152), SSSYPAEVVQK (SEQ ID NO: 153), QSRYPAEVVQK (SEQ ID NO: 154), QSSYPPSLVQK (SEQ ID NO: 155), AGAYPAEVVQK (SEQ ID NO: 156), IGSYPAEVVQK (SEQ ID NO: 157), QASYPAEVVQK (SEQ ID NO: 158), ASSYPAEVVQK (SEQ ID NO: 159), LGSYPAEVVQK (SEQ ID NO: 160), QSTNKAEVVQK (SEQ ID NO: 161)
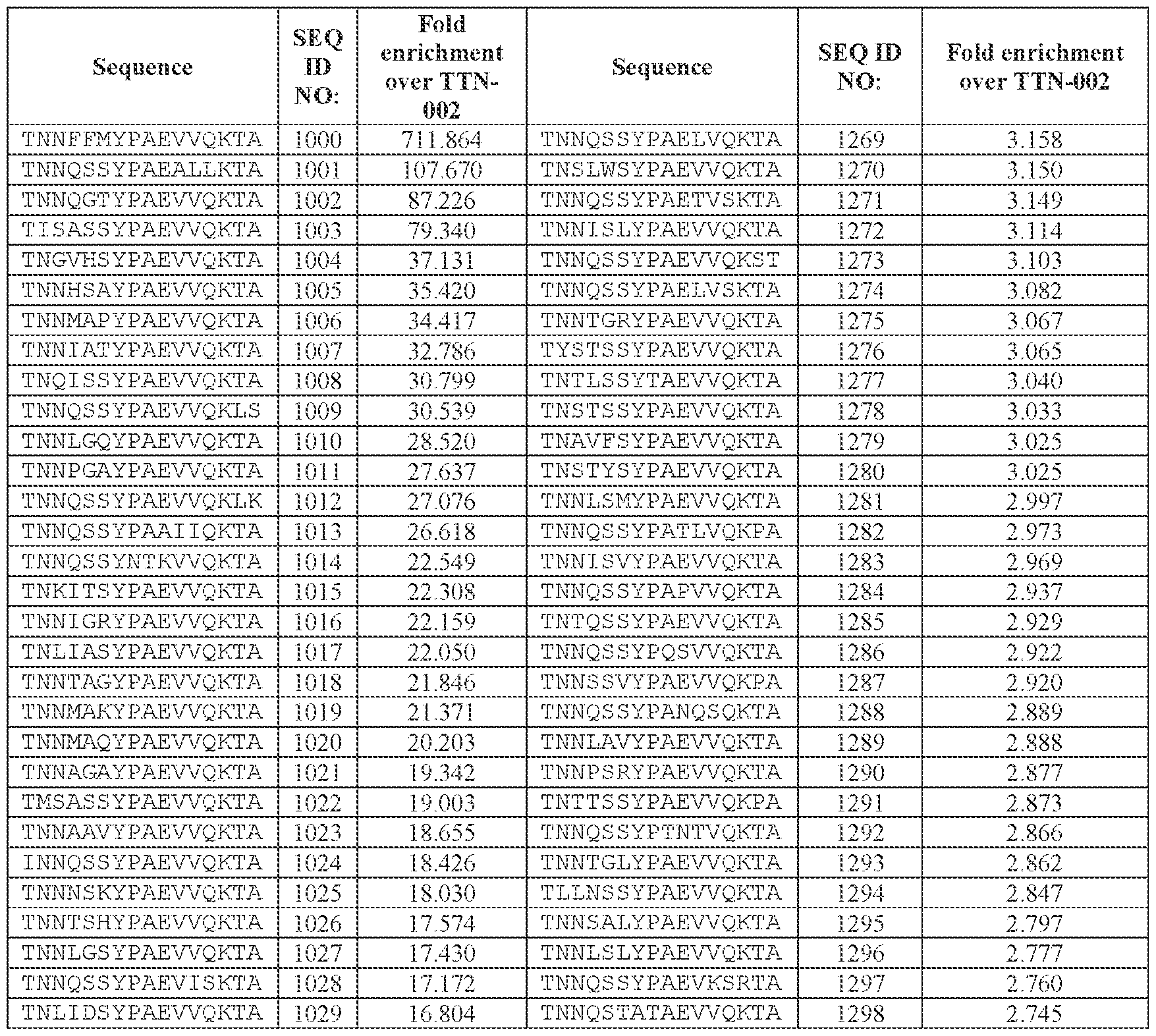
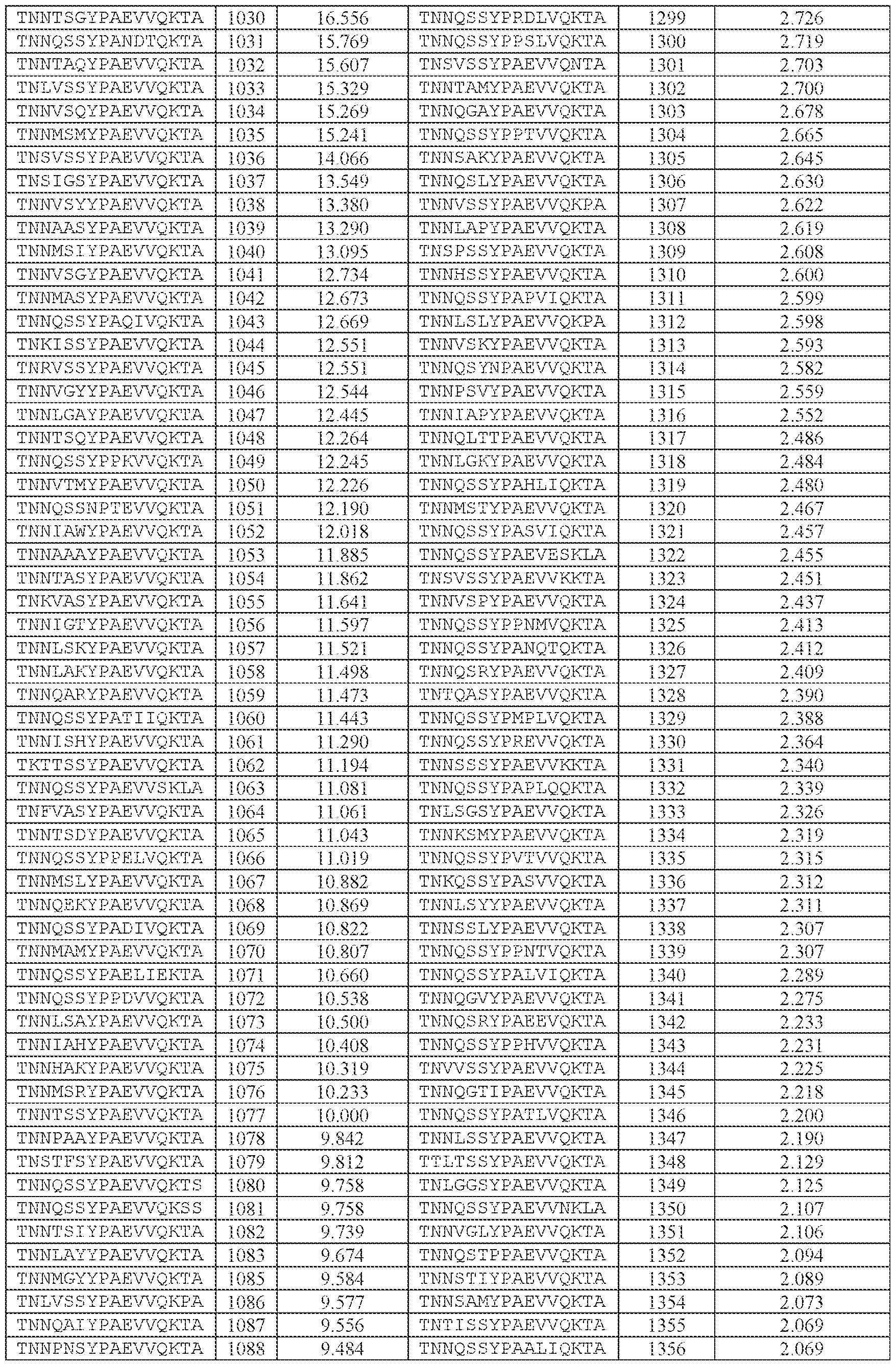
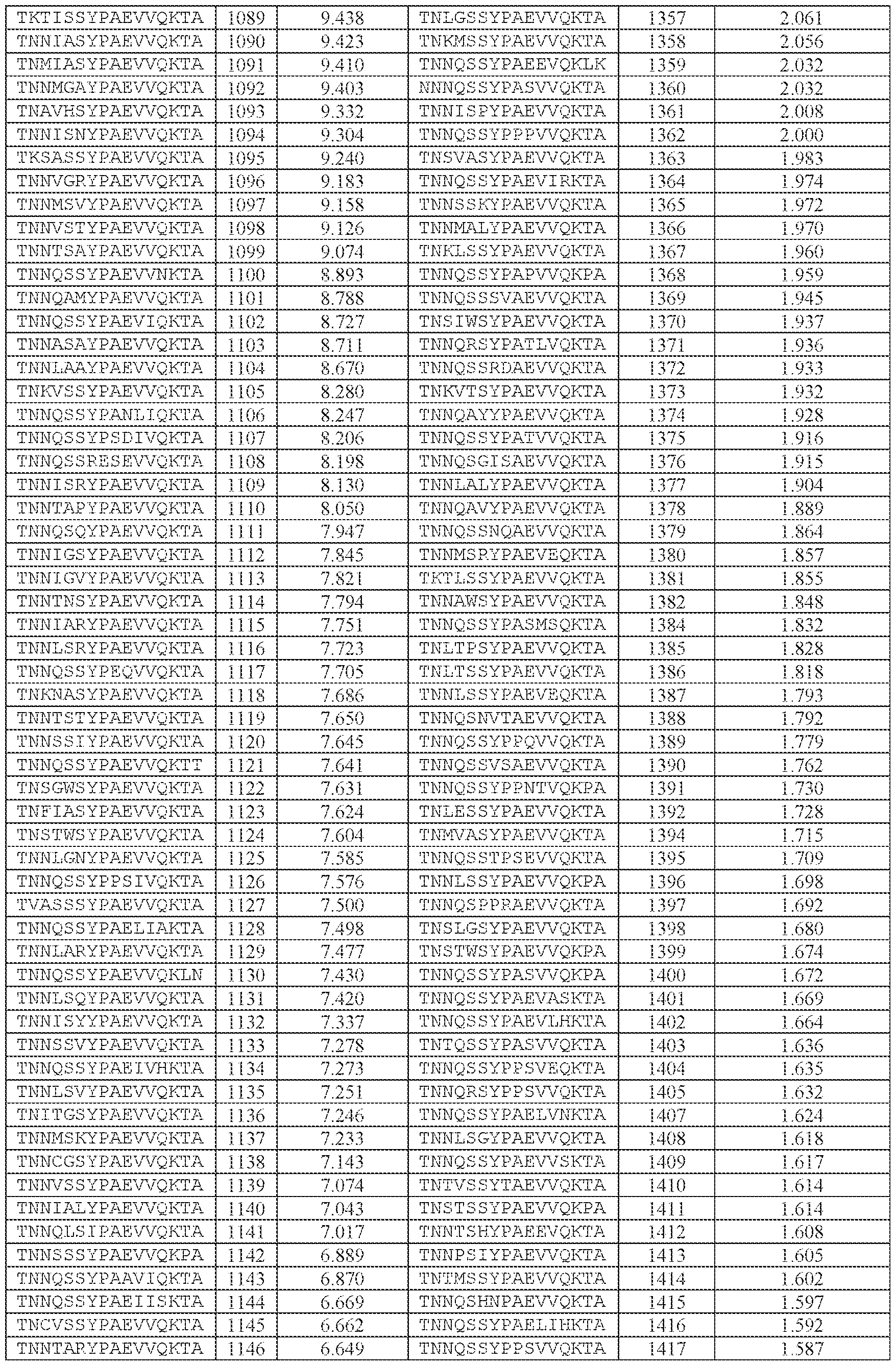
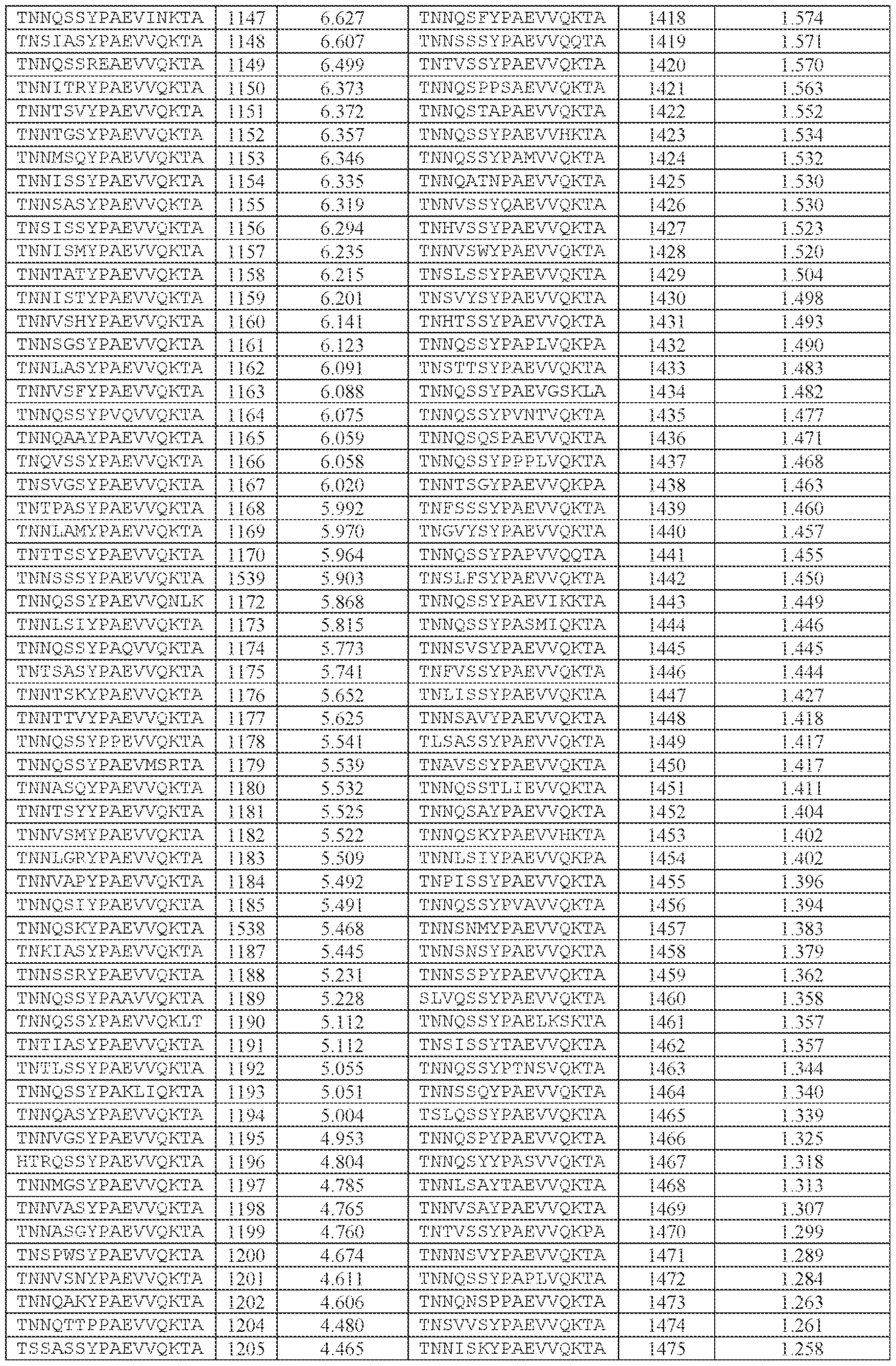
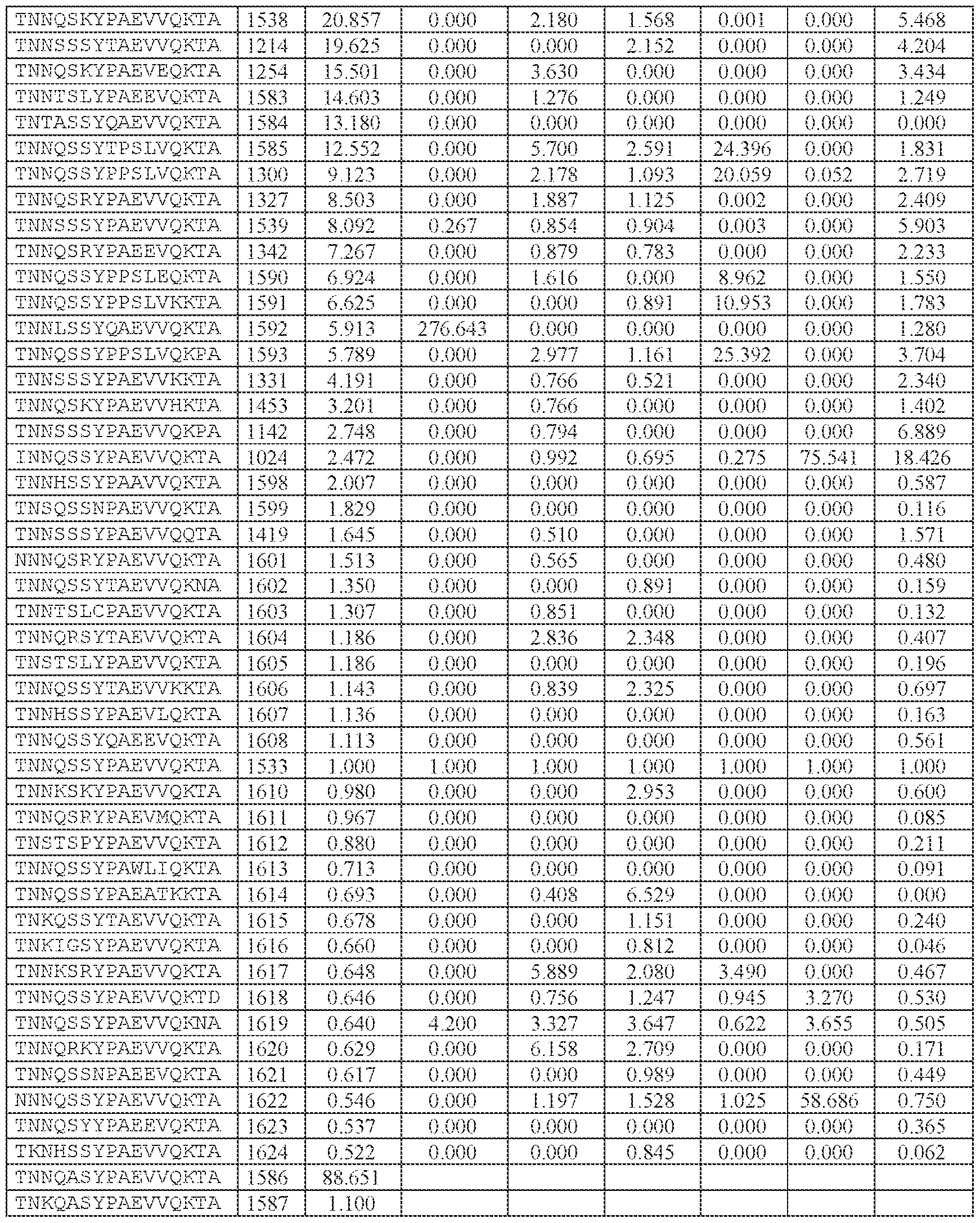
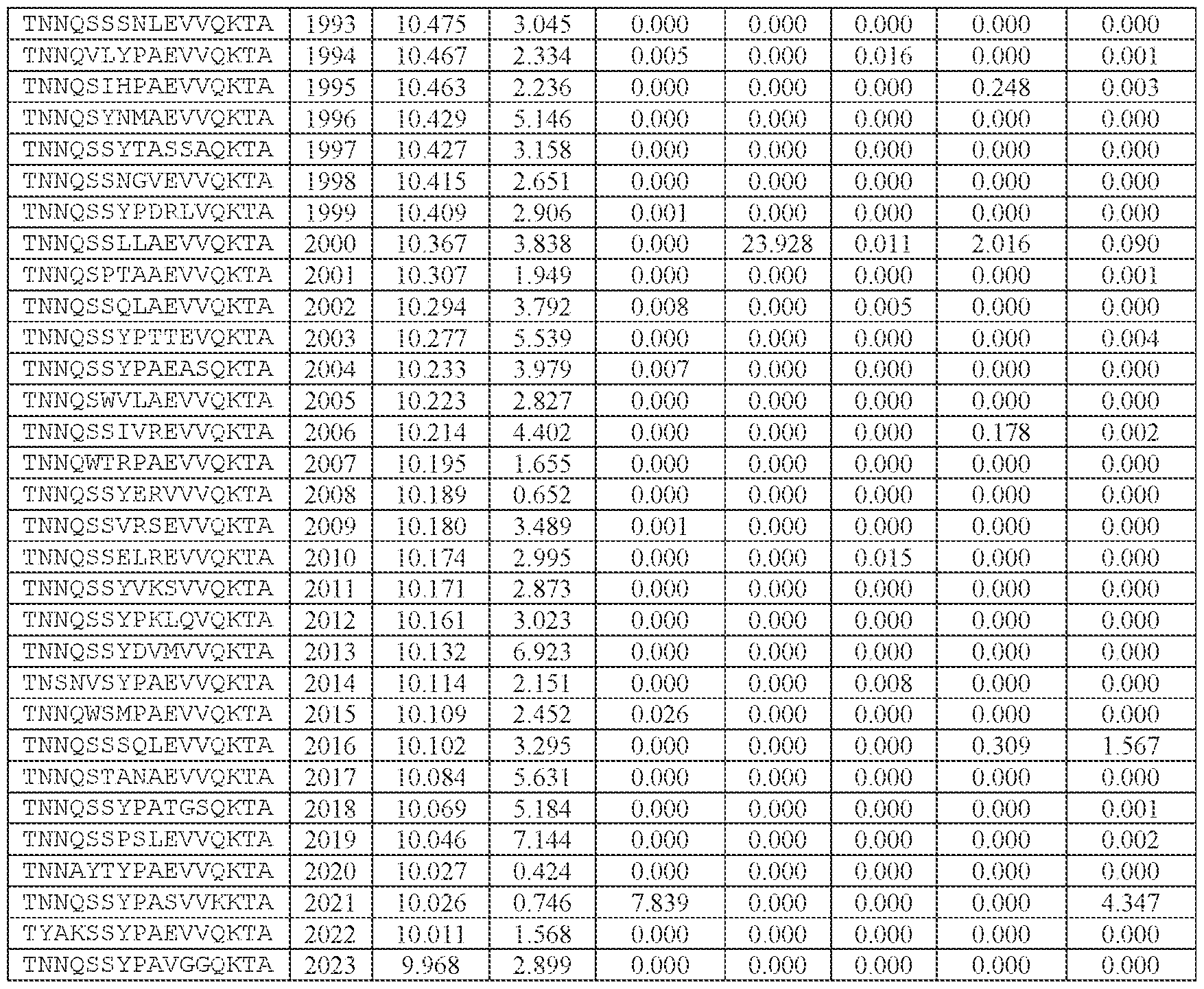
- TNNQSSYPAEVVQK (SEQ ID NO: 500), TNNQSKYPAEVVQK (SEQ ID NO: 503), TNNTSLYPAEVVQK (SEQ ID NO: 506), TNNSSSYPAEVVQK (SEQ ID NO: 508), TNNQSRYPAEVVQK (SEQ ID NO: 510), TNNQSSYPPSLVQK (SEQ ID NO: 512), TNNAGAYPAEVVQK (SEQ ID NO: 513), TNNIGSYPAEVVQK (SEQ ID NO: 514), TNNQASYPAEVVQK (SEQ ID NO: 517), TNTASSYPAEVVQK (SEQ ID NO: 520), TNNLGSYPAEVVQK (SEQ ID NO: 523), TNN
- AAV particle of embodiment 3 or 32, wherein [N0]-[N1]-[N2]-[N3] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500).
- 34. The AAV particle of embodiment 3 or 32, wherein [N0]-[N1]-[N2]-[N3] is or comprises TNNAGAYPAEVVQK (SEQ ID NO: 513), TNNTSLYPAEVVQK (SEQ ID NO: 506), TNNQSKYPAEVVQK (SEQ ID NO: 503), TNNQSSYTPSLVQK (SEQ ID NO: 533), TNNQSSYPPSLVQK (SEQ ID NO: 512), TNNQSRYPAEVVQK (SEQ ID NO: 510), TNNQSSYPPSLEQK (SEQ ID NO: 535), TNNQSSYPPSLVKK (SEQ ID NO: 536), or INNQSSYPAEVVQK (SEQ ID NO:
- [N4] is or comprises TA, PA, or NA.
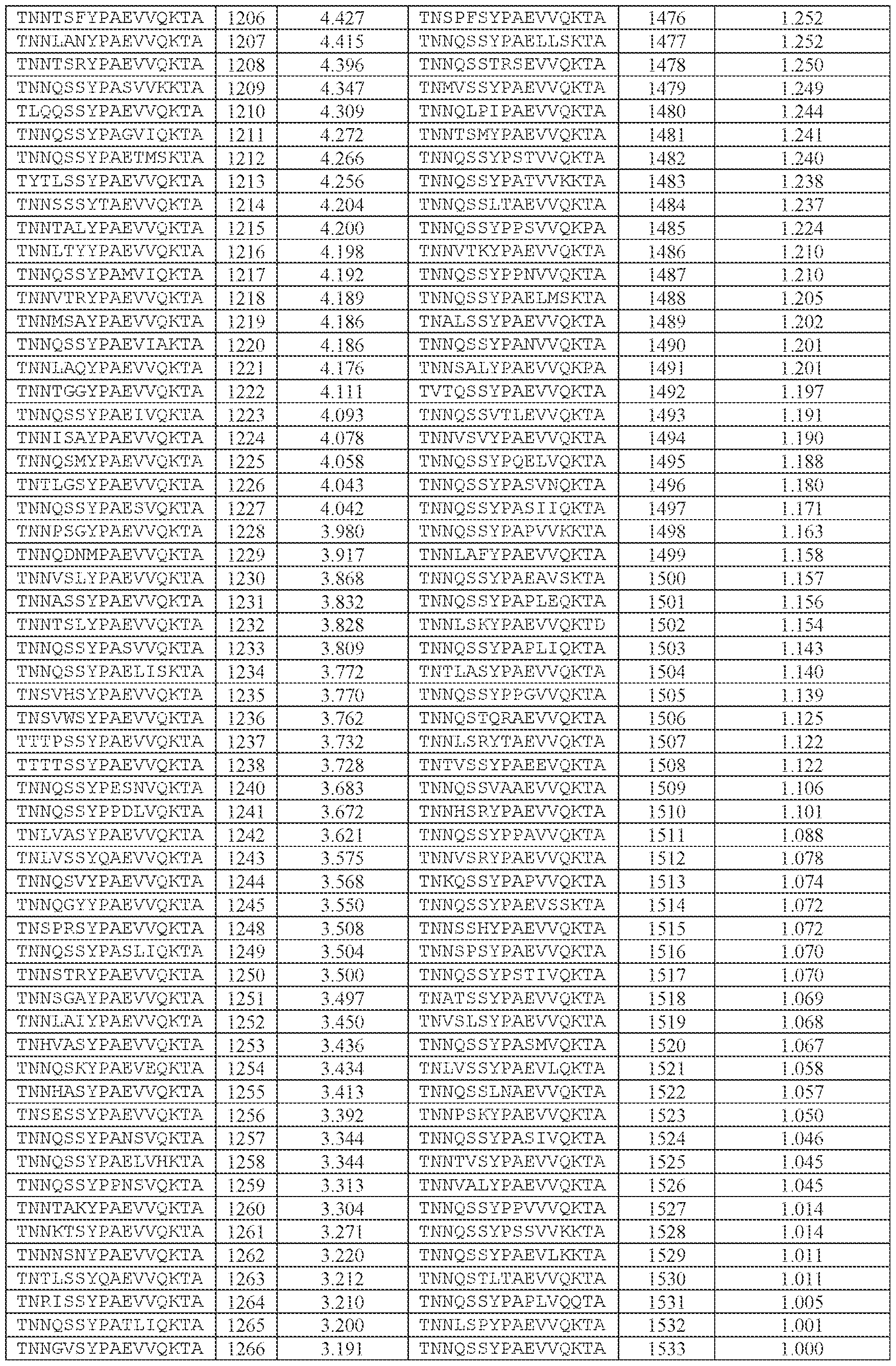
- the AAV particle of embodiment 3 or any one of embodiments 35-37, wherein [N0]-[N1]-[N2]- [N3]-[N4] is or comprises: (i) TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNTSLYPAEVVQKTA (SEQ ID NO: 1232), TNNSSSYPAEVVQKTA (SEQ ID NO: 1539), TNNQSRYPAEVVQKTA (SEQ ID NO: 1327), TNNQSSYPPSLVQKTA (SEQ ID NO: 1300), TNNAGAYPAEVVQKTA (SEQ ID NO: 1021), TNNIGSYPAEVVQKTA (SEQ ID NO: 1112), TNNQASYPAEVVQKTA (SEQ ID NO: 1194), TNTASSYPAEVVQKTA (SEQ ID NO: 1533), TNNQSKYPA
- the AAV particle of embodiment 3 or any one of embodiments 35-38, wherein [N0]-[N1]-[N2]- [N3]-[N4] is or comprises TNNAGAYPAEVVQKTA (SEQ ID NO: 1021), TNNTSLYPAEVVQKTA (SEQ ID NO: 1232), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNQSSYTPSLVQKTA (SEQ ID NO: 1585), TNNQSSYPPSLVQKTA (SEQ ID NO: 1300), TNNQSRYPAEVVQKTA (SEQ ID NO: Attorney Docket No.14640.0092-00304 1327), TNNQSSYPPSLEQKTA (SEQ ID NO: 1590), TNNQSSYPPSLVKKTA (SEQ ID NO: 1591), or INNQSSYPAEVVQKTA (SEQ ID NO: 1024).
- An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises an amino sequence comprising the following formula: [B]-[C] (SEQ ID NO: 2113), wherein (i) [B] comprises X 1 , X 2 , and X 3 , wherein: (a) X 1 is Q, T, S, A, I, L, or H; (b) X 2 is S, G, or A; and (c) X3 is S, K, L, R, or A; and (ii) [C] comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943).
- FXN frataxin
- AAV capsid variant e.g., an AAV5 capsid
- An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises one, two, three, four, or all of: (i) an [A], wherein [A] comprises the amino acid sequence of TNN, TNT, INN, NNN, TNS, or TNK; (ii) a [B], wherein [B] comprises the amino acid sequence of QSS, TSL, SSS, QSR, QSK, AGA, IGS, QAS, ASS, LGS, or HSS; (iii) a [C], wherein [C] comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943); and (iv) a [D], wherein [D] comprises the amino acid sequence of TA or PA; and
- AAV particle of embodiment 41 wherein: (a) X1 is Q, T, S, A, or H; (b) X2 is S or G; and (c) X 3 is S, K, L, or R. 44.
- the AAV particle of embodiment 41 or any one of embodiments 43-45, wherein [B]-[C] comprises: (i) SSYPAEVVQK (SEQ ID NO: 572), SKYPAEVVQK (SEQ ID NO: 573), SLYPAEVVQK (SEQ ID NO: 574), SRYPAEVVQK (SEQ ID NO: 575), GAYPAEVVQK (SEQ ID NO: 576), GSYPAEVVQK (SEQ ID NO: 580), or ASYPAEVVQK (SEQ ID NO: 582); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but
- AAV particle of any one of embodiments 41-46, wherein [B]-[C] is or comprises: (i) QSSYPAEVVQK (SEQ ID NO: 150), QSKYPAEVVQK (SEQ ID NO: 151), TSLYPAEVVQK (SEQ ID NO: 152), SSSYPAEVVQK (SEQ ID NO: 153), QSRYPAEVVQK (SEQ ID NO: 154), AGAYPAEVVQK (SEQ ID NO: 156), IGSYPAEVVQK (SEQ ID NO: 157), QASYPAEVVQK (SEQ ID NO: 158), ASSYPAEVVQK (SEQ ID NO: 159), LGSYPAEVVQK (SEQ ID NO: 160), or HSSYPAEVVQK (SEQ ID NO: 162); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10
- the AAV particle of any one of embodiments 1-49 which further comprises one or both of an amino acid other than T at position 571 (e.g., I or N), and/or an amino acid other than N at position 573 (e.g., T, S, or K), relative to a reference sequence numbered according to the amino acid sequence of SEQ ID NO: 138 or 982.
- Attorney Docket No.14640.0092-00304 51 is a reference sequence numbered according to the amino acid sequence of SEQ ID NO: 138 or 982.
- the AAV particle of any one of embodiments 1-49 which further comprises: (i) a T at position 571, an N at position 572, and/or an N at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (ii) a T at position 571, an N at position 572, and/or a T at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iii) an I at position 571, an N at position 572, and/or an N at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iv) a T at position 571, an N at position 572, and/or an S at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (v) an N at position 571, an N at position 572, and/or an N at position 573, according to the amino acid sequence of SEQ ID NO: 138 or
- AAV particle of any one of embodiments 41-51 which further comprises [A], wherein [A] comprises X A , X B , and X C , wherein: (a) XA is T, I, or N; (b) X B is N; and (c) X C is N, T, S, or K; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c).
- [A] comprises TN, IN, NN, NT, NS, or NK. 54.
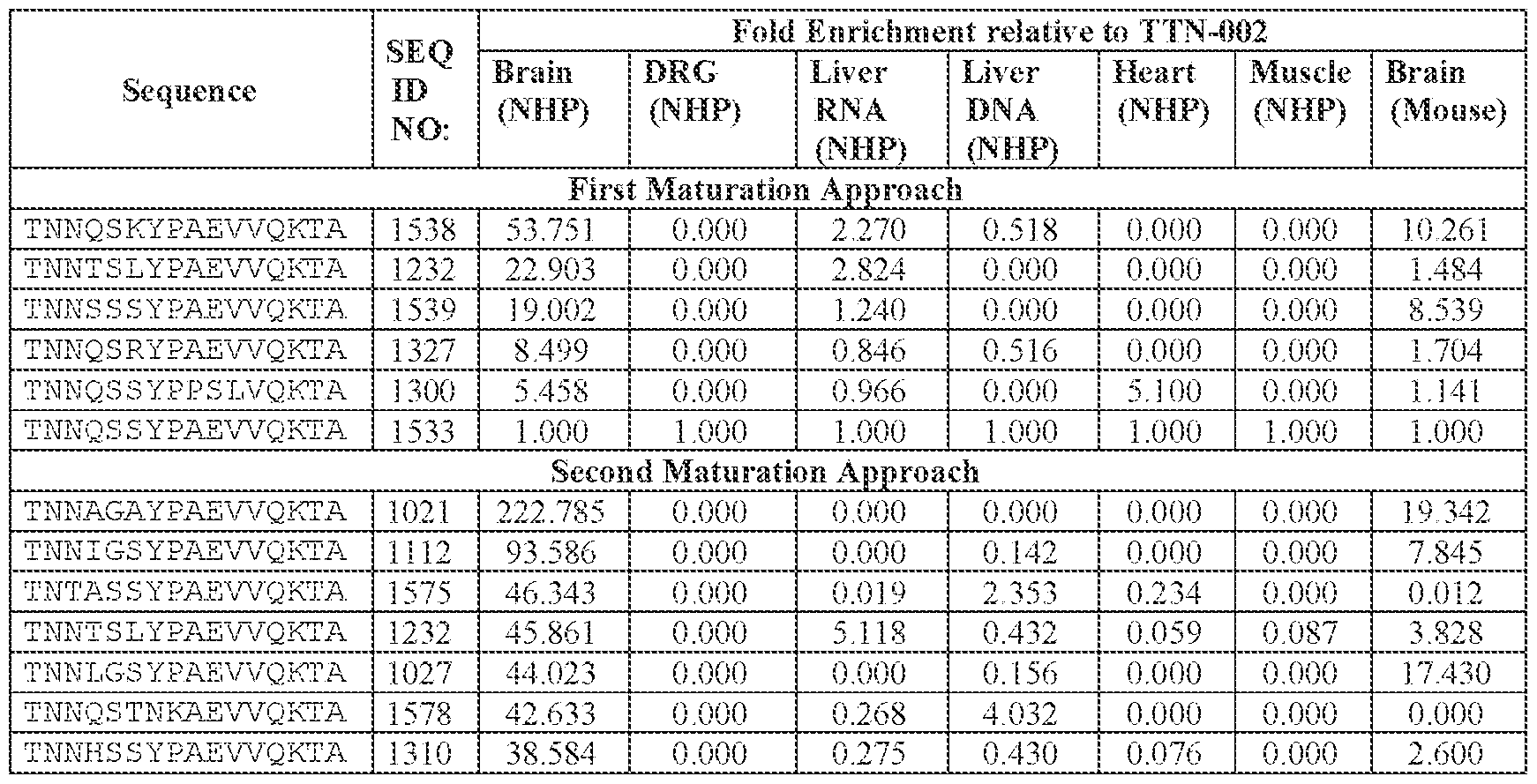
- AAV particle of embodiment 42 or any one of embodiments 52-55, wherein [A]-[B]-[C] is or comprises: (i) TNNQSSYPAEVVQK (SEQ ID NO: 500), TNNQSKYPAEVVQK (SEQ ID NO: 503), TNNTSLYPAEVVQK (SEQ ID NO: 506), TNNSSSYPAEVVQK (SEQ ID NO: 508), TNNQSRYPAEVVQK (SEQ ID NO: 510), TNNAGAYPAEVVQK (SEQ ID NO: 513), TNNIGSYPAEVVQK (SEQ ID NO: 514), TNNQASYPAEVVQK (SEQ ID NO: 517), TNTASSYPAEVVQK (SEQ ID NO: 520), TNNLGSYPAEVVQK (SEQ ID NO: 523), TNNHSSYPAEVVQK (SEQ ID NO: 525), INNQSSYPAEVVQK (S
- TNNAGAYPAEVVQK SEQ ID NO: 513
- TNNTSLYPAEVVQK SEQ ID NO: 506
- TNNQSKYPAEVVQK SEQ ID NO: 503
- TNNQSRYPAEVVQK SEQ ID NO: 510
- the AAV particle of any one of embodiments 1-58 which further comprises: (i) an amino acid other than T at position 578 (e.g., P or N), numbered according to the amino acid sequence of SEQ ID NO: 138; or (ii) an amino acid other than T at position 585 (e.g., P or N), numbered according to the amino acid sequence of SEQ ID NO: 982. 60.
- the AAV particle of any one of embodiments 1-58 which further comprises: Attorney Docket No.14640.0092-00304 (i) a T at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; or a T at position 585 and/or an A at position 586 relative to a reference sequence numbered according to SEQ ID NO: 982; (ii) a P at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; or a P at position 585 and/or an A at position 586 numbered according to SEQ ID NO: 982; or (iii) an N at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; or an N at position 585 and/or an A at position 586 numbered according to SEQ ID NO: 982.
- the AAV particle of any one of embodiments 42, 61, and 62, wherein [C]-[D] is or comprises: (i) YPAEVVQKTA (SEQ ID NO: 584) or YPAEVVQKPA (SEQ ID NO: 586); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
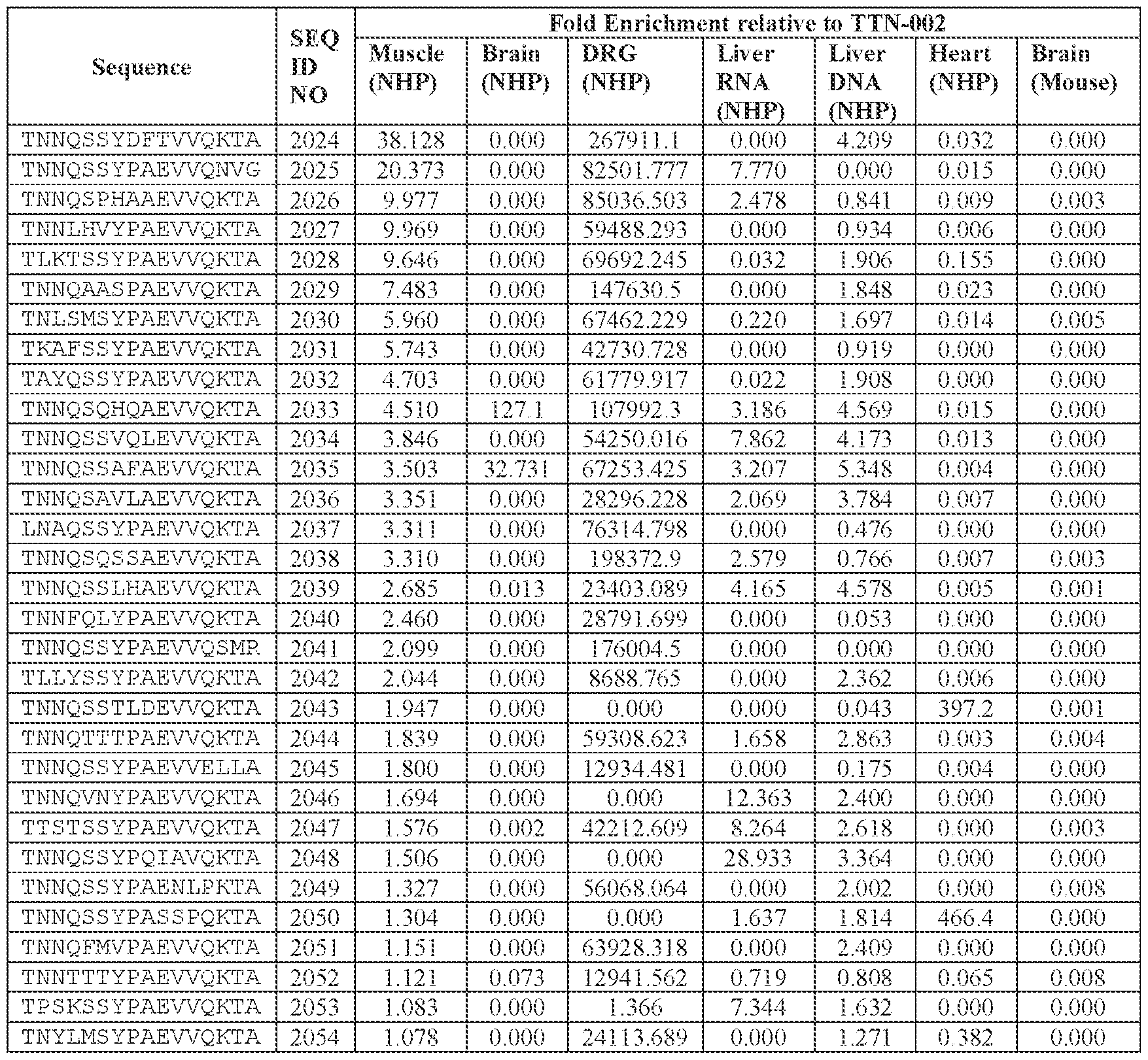
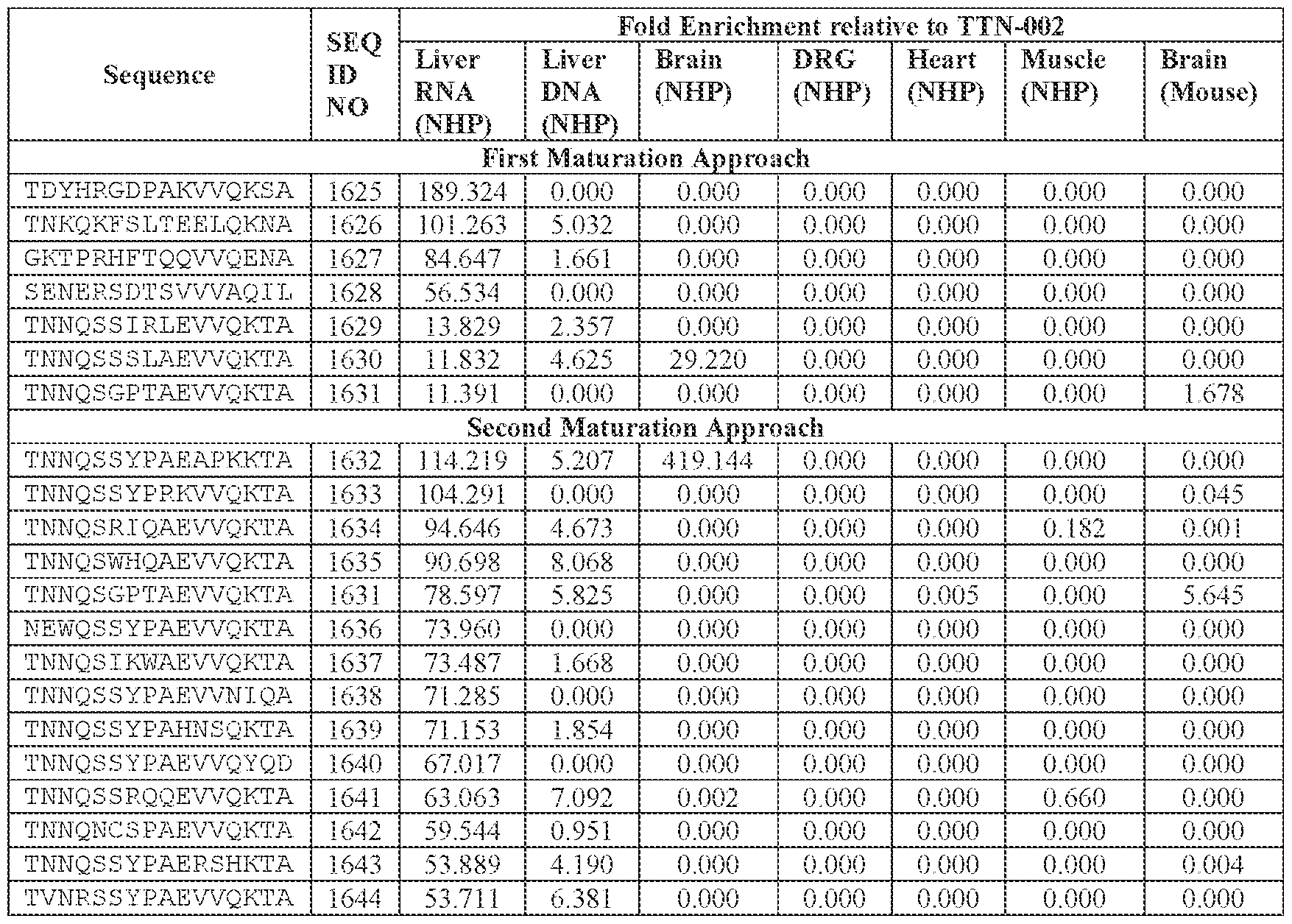
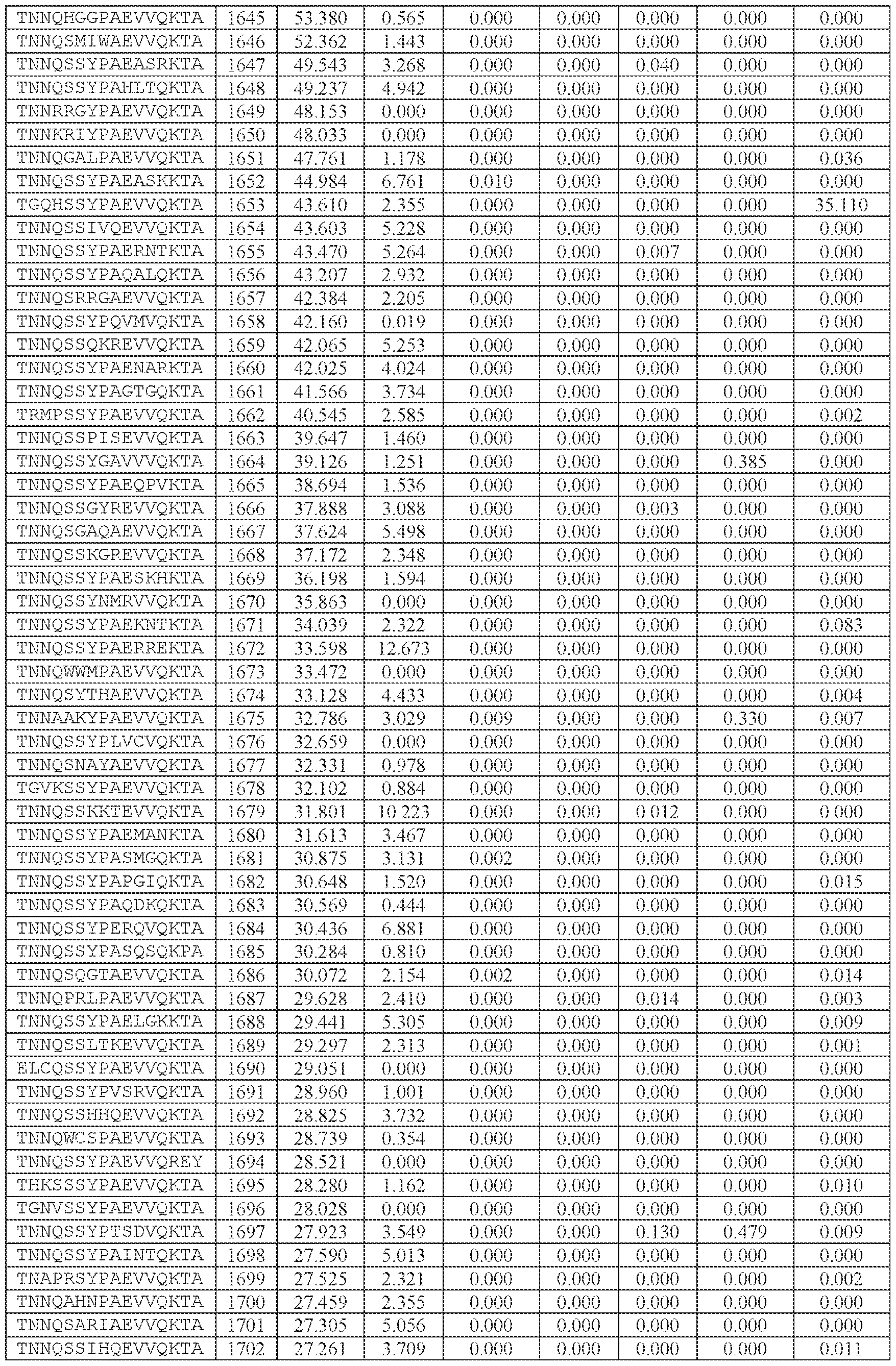
- [A]-[B]-[C]-[D] is or comprises: (i) TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNTSLYPAEVVQKTA (SEQ ID NO: 1232), TNNSSSYPAEVVQKTA (SEQ ID NO: 1539), TNNQSRYPAEVVQKTA (SEQ ID NO: 1327), TNNAGAYPAEVVQKTA (SEQ ID NO: 1021), TNNIGSYPAEVVQKTA (SEQ ID NO: 1112), TNNQASYPAEVVQKTA (SEQ ID NO: 1194), TNTASSYPAEVVQKTA (SEQ ID NO: 1575), TNNLGSYPAEVVQKTA (SEQ ID NO: 1027), TNNHSSYPAEVVQKTA (SEQ ID NO: 1533), TNNQSKYPAEVVQKTA (SEQ
- An AAV particle comprising a viral genome comprising a frataxin (FXN)-encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises an amino sequence comprising the formula [N2]-[N3], wherein: (i) [N2] comprises X 1 , X 2 , X 3 , X 4 , and X 5 , wherein: (a) X1 is Y or T; (b) X2 is Q, T, P, or E; (c) X3 is A; (d) X4 is E or D; and (e) X5 is V or E; and (ii) [N3] comprises the amino acid sequence of VQK or VQN.
- FXN frataxin
- AAV capsid variant comprises an amino sequence comprising the formula [N2]-[N3], wherein: (i)
- An AAV particle comprising a viral genome encoding a frataxin (FXN)-encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises one, two, three, four, or all of: (i) an [N0] comprising TNN, TNS, TNT, or TNK; (ii) an [N1] comprising QSS, SLS, SLY, SAT, or QTS; (iii) an [N2] comprising YPAEV (SEQ ID NO: 1), YQAEV (SEQ ID NO: 6), YTAEV (SEQ ID NO: 4), YPAEE (SEQ ID NO: 5), TEAEV (SEQ ID NO: 12), or YPADV (SEQ ID NO: 13); (iv) an [N3] comprising VQK or VQN; and (v) an [N4]
- AAV particle of embodiment 1 or 67, wherein [N2] comprises YP, YQ, YT, TE, QA, TA, PA, EA, EV, EE, DV, AE, or AD. 70.
- 72. The AAV particle of embodiment 1 or any one of embodiments 67-71, wherein [N2] is or comprises 73.
- AAV particle of embodiment 1 or 67 or any one of embodiments 69-74, wherein [N2]-[N3] comprises: (i) PAEVVQN (SEQ ID NO: 594), QAEVVQK (SEQ ID NO: 52), TAEVVQK (SEQ ID NO: 49), PAEVVQK (SEQ ID NO: 20), PAEEVQK (SEQ ID NO: 51), EAEVVQK (SEQ ID NO: 595), or PADVVQK (SEQ ID NO: 596); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof; Attorney Docket No.14640.0092-00304 (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no
- the AAV particle of embodiment 1 or any one of embodiments 67-78 which further comprises one, two, three or all of an amino acid other than Q at position 574 (e.g., S), an amino acid other than S at position 575 (e.g., L, A, or T), and/or an amino acid other than S at position 576 (e.g., Y or T), numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 80.
- an amino acid other than Q at position 574 e.g., S
- an amino acid other than S at position 575 e.g., L, A, or T
- an amino acid other than S at position 576 e.g., Y or T
- the AAV particle of embodiment 1 or any one of embodiments 67-78 which further comprises: (i) a Q at position 574, an S at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (ii) an S at position 574, an L at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iii) an S at position 574, an L at position 575, and/or a Y at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iv) an S at position 574, an A at position 575, and/or a T at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; or Attorney Docket No.14640.0092-00304 (v) a Q at position 574, a T at position 575, and/or an
- the AAV particle of any one of embodiments 81-83, wherein [N1]-[N2] comprises: (i) SSYPA (SEQ ID NO: 63), LSYQA (SEQ ID NO: 597), LSYTA (SEQ ID NO: 598), LYYPA (SEQ ID NO: 600), ATYPA (SEQ ID NO: 601), LSYPA (SEQ ID NO: 603), or TSTEA (SEQ ID NO: 605); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i);
- AAV particle of any one of embodiments 81-84, wherein [N1]-[N2] comprises: (i) SSYPAE (SEQ ID NO: 79), LSYQAE (SEQ ID NO: 607), LSYTAE (SEQ ID NO: 610), LYYPAE (SEQ ID NO: 611), ATYPAE (SEQ ID NO: 613), LSYPAE (SEQ ID NO: 616), TSTEAE (SEQ ID NO: 619), or LSYPAD (SEQ ID NO: 621); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or Attorney Docket No.14640.0092-00304 (iv) an amino acid sequence comprising one, two,
- [N1]-[N2]-[N3] is or comprises: (i) QSSYPAEVVQK (SEQ ID NO: 150), SLSYQAEVVQK (SEQ ID NO: 635), SLSYTAEVVQK (SEQ ID NO: 637), SLYYPAEVVQK (SEQ ID NO: 639), SATYPAEVVQK (SEQ ID NO: 641), SLSYPAEVVQK (SEQ ID NO: 642), SLSYPAEEVQK (SEQ ID NO: 643), SLSYPAEVVQN (SEQ ID NO: 644), QTSTEAEVVQK (SEQ ID NO: 645), or SLSYPADVVQK (SEQ ID NO: 646); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 amino acids
- AAV particle of embodiment 89 wherein [N0]: (i) comprises TN, NS, NT, NN, or NK; and/or (ii) is or comprises TNS, TNT, TNN, or TNK. 91.
- the AAV particle of any one of embodiments 68, 89, and 90, wherein [N0]-[N1] is or comprises: (i) TNNQSS (SEQ ID NO: 183), TNSSLS (SEQ ID NO: 647), TNSSLY (SEQ ID NO: 648), TNTSAT (SEQ ID NO: 649), TNNQTS (SEQ ID NO: 650), or TNKSAT (SEQ ID NO: 651); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
- the AAV particle of embodiment 68 or any one of embodiments 89-91, wherein [N0]-[N1]-[N2]- [N3] is or comprises: (i) TNNQSSYPAEVVQK (SEQ ID NO: 500), TNSSLSYQAEVVQK (SEQ ID NO: 652), TNSSLSYTAEVVQK (SEQ ID NO: 654), TNSSLYYPAEVVQK (SEQ ID NO: 655), TNTSATYPAEVVQK (SEQ ID NO: 656), TNSSLSYPAEVVQK (SEQ ID NO: 657), TNSSLSYPAEEVQK (SEQ ID NO: 658), TNSSLSYPAEVVQN (SEQ ID NO: 660), TNNQTSTEAEVVQK (SEQ ID NO: 662), TNKSATYPAEVVQK (SEQ ID NO: 663), or TNSSLSYPADVVQK (SEQ ID NO: 6
- the AAV particle of embodiment 68, 94, or 95, wherein [N3]-[N4] is or comprises: (i) VQKTA (SEQ ID NO: 564), EQKTA (SEQ ID NO: 565), VKKTA (SEQ ID NO: 566), VQKPA (SEQ ID NO: 567), VHKTA (SEQ ID NO: 568), VQQTA (SEQ ID NO: 569), VQKNA (SEQ ID NO: 570), or LQKTA (SEQ ID NO: 571); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequence
- the AAV particle of embodiment 68 or any one of embodiments 94-96, wherein [N0]-[N1]-[N2]- [N3]-[N4] is or comprises: (i) TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNSSLSYQAEVVQKTA (SEQ ID NO: 2064), TNSSLSYTAEVVQKTA (SEQ ID NO: 2065), TNSSLYYPAEVVQKTA (SEQ ID NO: 2066), TNTSATYPAEVVQKTA (SEQ ID NO: 2067), TNSSLSYPAEVVQKTA (SEQ ID NO: 2068), TNSSLSYPAEEVQKTA (SEQ ID NO: 2069), TNSSLSYPAEVVQKTD (SEQ ID NO: 2070), TNSSLSYPAEVVQNTA (SEQ ID NO: 2071), TNSSLSYPAEVVQKNA (SEQ ID NO: 2072),
- An AAV particle comprising a viral genome comprising a frataxin (FXN)-encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises the formula [K1]-[K2], wherein: (i) [K1] comprises LSY or LYY; and (ii) [K2] comprises X 1 , X 2 , X 3 , and X 4 , wherein: (a) X 1 is Q, T, or P; (b) X 2 is A; (c) X3 is E or D; and (d) X4 is V or E. 100.
- FXN frataxin
- AAV capsid variant comprises the formula [K1]-[K2], wherein: (i) [K1] comprises LSY or LYY; and (ii) [K2] comprises X 1 , X 2
- An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises one, two, three, four, or all of: (i) an [K0], which comprises TNNS (SEQ ID NO: 14); (ii) an [K1], which comprises LSY or LYY; (iii) an [K2], which comprises QAEV (SEQ ID NO: 15), TAEV (SEQ ID NO: 16), PAEV (SEQ ID NO: 17), PAEE (SEQ ID NO: 18), or PADV (SEQ ID NO: 19); (iv) an [K3], which comprise VQK or VQN; and (v) an [K4], which comprises TA, TD, NA, or PA.
- FXN frataxin
- AAV particle of embodiment 99 or 100, wherein [K1] comprises LSY. 102.
- the AAV particle of embodiment 99 or 101, wherein [K2] comprises QA, TA, PA, EV, EE, DV, AE, or AD.
- Attorney Docket No.14640.0092-00304 105 The AAV particle of embodiment 99 or any one of embodiments 101-104, wherein [K1]-[K2] comprises LSYQA (SEQ ID NO: 597), LSYTA (SEQ ID NO: 598), LYYPA (SEQ ID NO: 600), or LSYPA (SEQ ID NO: 603).
- the AAV particle of embodiment 99 or any one of embodiments 101-105, wherein [K1]-[K2] comprises: (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 107.
- the AAV particle of any one of embodiments 99-106, wherein [K1]-[K2] is or comprises: (i) LSYQAEV (SEQ ID NO: 667), LSYTAEV (SEQ ID NO: 668), LYYPAEV (SEQ ID NO: 669), LSYPAEV (SEQ ID NO: 671), LSYPAEE (SEQ ID NO: 673), or LSYPADV (SEQ ID NO: 674); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
- Attorney Docket No.14640.0092-00304 111 Attorney Docket No.14640.0092-00304 111.
- the AAV particle of embodiment 110, wherein [K0]-[K1] comprises: (i) TNSSLS (SEQ ID NO: 647) or TNSSLY (SEQ ID NO: 648); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 112.
- the AAV particle of any one of embodiments 110-112, wherein [K0]-[K1]-[K2] comprises: (i) TNSSLSYQA (SEQ ID NO: 679), TNSSLSYTA (SEQ ID NO: 681), TNSSLYYPA (SEQ ID NO: 682), or TNSSLSYPA (SEQ ID NO: 683); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, or 8 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
- the AAV particle of any one of embodiments 110-113, wherein [K0]-[K1]-[K2] comprises: (i) TNSSLSYQAE (SEQ ID NO: 684), TNSSLSYTAE (SEQ ID NO: 685), TNSSLYYPAE (SEQ ID NO: 686), TNSSLSYPAE (SEQ ID NO: 687), or TNSSLSYPAD (SEQ ID NO: 689); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or Attorney Docket No.14640.0092-00304 (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequence
- the AAV particle of embodiment 100 or any one of embodiments 110-114, wherein [K0]-[K1]- [K2] is or comprises: (i) TNSSLSYQAEV (SEQ ID NO: 692), TNSSLSYTAEV (SEQ ID NO: 693), TNSSLYYPAEV (SEQ ID NO: 696), TNSSLSYPAEV (SEQ ID NO: 697), TNSSLSYPAEE (SEQ ID NO: 698), or TNSSLSYPADV (SEQ ID NO: 699); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids
- AAV particle of any one of embodiments 99-115 which further comprises [K3], wherein [K3] comprises XA, XB, and XC, wherein: (a) X A is V; (b) X B is Q; and (c) XC is K or N; and optionally wherein the AAV capsid comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c).
- [K3] comprises VQ, QK, or QN; (ii) is or comprises VQK or VQN. 118.
- the AAV particle of embodiment 100, 116, or 117, wherein [K2]-[K3] is or comprises: (i) QAEVVQK (SEQ ID NO: 52), TAEVVQK (SEQ ID NO: 49), PAEVVQK (SEQ ID NO: 20), PAEEVQK (SEQ ID NO: 51), PAEVVQN (SEQ ID NO: 594), or PADVVQK (SEQ ID NO: 596); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
- the AAV particle of embodiment 100 or any one of embodiments 116-118, wherein [K1]-[K2]- [K3] is or comprises: (i) LSYQAEVVQK (SEQ ID NO: 700), LSYTAEVVQK (SEQ ID NO: 701), LYYPAEVVQK (SEQ ID NO: 702), LSYPAEVVQK (SEQ ID NO: 703), LSYPAEEVQK (SEQ ID NO: 704), LSYPAEVVQN (SEQ ID NO: 706), or LSYPADVVQK (SEQ ID NO: 708); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one
- the AAV particle of embodiment 100 or any one of embodiments 116-119, wherein [K0]-[K1]- [K2]-[K3] is or comprises: (i) TNSSLSYQAEVVQK (SEQ ID NO: 652), TNSSLSYTAEVVQK (SEQ ID NO: 654), TNSSLYYPAEVVQK (SEQ ID NO: 655), TNSSLSYPAEVVQK (SEQ ID NO: 657), TNSSLSYPAEEVQK (SEQ ID NO: 658), TNSSLSYPAEVVQN (SEQ ID NO: 660), or TNSSLSYPADVVQK (SEQ ID NO: 665); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three
- An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises [B]-[C] (SEQ ID NO: 2114), wherein: (i) [B] comprises X 1 , X 2 , and X 3 , wherein: (a) X 1 is Q or S; (b) X 2 is S, L, or A; and (c) X3 is S, Y, or T; and (ii) [C] comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943).
- FXN frataxin
- AAV capsid variant e.g., an AAV5 capsid variant
- An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises one, two, three, four, or all of: (i) an [A] comprising TNN, TNS, TNT, or TNK; (ii) a [B] comprising QSS, SLY, SAT, or SLS; (iii) a [C] comprising YPAEVVQK (SEQ ID NO: 943); and (iv) a [D] comprising Attorney Docket No.14640.0092-00304 optionally wherein [C] replaces position 577 numbered according to the amino acid sequence of SEQ ID NO: 138.
- AAV capsid variant comprises one, two, three, four, or all of: (i) an [A]
- the AAV particle of embodiment 125 comprising wherein [B] comprises QS, SL, SA, LY, AT, LS, or SS. 128.
- the AAV particle of any one of embodiments 125, 127, and 128, wherein [B]-[C] comprises: (i) SSYPAEVVQK (SEQ ID NO: 572), LYYPAEVVQK (SEQ ID NO: 702), ATYPAEVVQK (SEQ ID NO: 718), or LSYPAEVVQK (SEQ ID NO: 703); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
- AAV particle of any one of embodiments 125-129, wherein [B]-[C] is or comprises: (i) QSSYPAEVVQK (SEQ ID NO: 150), SLYYPAEVVQK (SEQ ID NO: 639), SATYPAEVVQK (SEQ ID NO: 641), or SLSYPAEVVQK (SEQ ID NO: 642); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
- the AAV particle of any one of embodiments 67-98 or any one of embodiments 125-131 which further comprises: (i) a T at position 571, an N at position 572, and/or an N at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138; (ii) a T at position 571, an N at position 572, and/or a T at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138; (iii) a T at position 571, an N at position 572, and/or a S at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138; or (iv) a T at position 571, an N at position 572, and/or a K at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138.
- AAV particle of any one of embodiments 125-133 which further comprises [A], wherein [A] comprises X A , X B , and X C , wherein: (a) X A is T; (b) X B is N; and (c) X C is N, T, S, or K; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c). 135.
- [A] comprises X A , X B , and X C , wherein: (a) X A is T; (b) X B is N; and (c) X C is N, T, S, or K; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c). 135.
- the AAV particle of embodiment 126 or 134 wherein [A]: (i) comprises TN, NS, NT, NK, or NN; (ii) is or comprises TNN, TNS, TNT, or TNK. 136.
- the AAV particle of any one of embodiments 67-98 or any one of embodiments 125-138 which further comprises: (i) a T at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; (ii) a T at position 578 and/or a D at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; (iii) a P at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; or (iv) an N at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138. 141.
- the AAV particle of embodiment 126, 141, or 142, wherein [C]-[D] is or comprises: (i) YPAEVVQKTA (SEQ ID NO: 584), YPAEVVQKTD (SEQ ID NO: 719), YPAEVVQKNA (SEQ ID NO: 724), or YPAEVVQKPA (SEQ ID NO: 586); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
- the AAV particle of embodiment 126 or any one of embodiments 141-143, wherein [A]-[B]-[C]- [D] is or comprises: (i) TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNSSLYYPAEVVQKTA (SEQ ID NO: 2066), TNTSATYPAEVVQKTA (SEQ ID NO: 2067), TNSSLSYPAEVVQKTA (SEQ ID NO: 2068), TNSSLSYPAEVVQKTD (SEQ ID NO: 2070), TNSSLSYPAEVVQKNA (SEQ ID NO: 2072), TNSSLSYPAEVVQKPA (SEQ ID NO: 2073), or TNKSATYPAEVVQKTA (SEQ ID NO: 2075); (ii) (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids,
- amino acids TNNQSSTTAPATGTYNLQEIVP SEQ ID NO: 736
- positions 571-599 e.g., amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)
- amino acids TNNQSSTTAPATGTYNLQEIVP SEQ ID Attorney Docket No.14640.0092-00304 NO: 736
- positions 571-599 e.g., amino acids TNNQSSYPAEV
- loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)) numbered according to the amino acid sequence of SEQ ID NO: 982.
- positions 571-592 e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)
- positions 571-599 e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)
- AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-155 wherein X 1 of [N2] corresponds to position 577 (e.g., Y577), X 2 of [N2] corresponds to position 578 (e.g., P588), X 3 of [N2] corresponds to position 579 (e.g., A579), X 4 of [N2] corresponds to position 580 (e.g., E580), and X 5 of [N2] corresponds to position 581 (e.g., V581) of the amino acid sequence of SEQ ID NO: 982. 157.
- X 1 of [N2] corresponds to position 577 (e.g., Y577)
- X 2 of [N2] corresponds to position 578 (e.g., P588)
- X 3 of [N2] corresponds to position 579 (e.g., A579)
- [N1]-[N2]-[N3]-[N4] replaces positions 574-579 (e.g., Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 173.
- [N0]-[N1]-[N2]-[N3]-[N4] replaces positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 184.
- positions 571-579 e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and A579
- positions 571-586 e.g., T571, N572, N573, Q574, S575, S576, Y577, P578, A579, E580, V581, V582, Q583, K584, T585, A586
- AAV particle of any one of embodiments 3, 31-40, 68, 94-98, 139, 140, and 146-188 wherein: (i) X A of [N0] is present at position 571, X B of [N0] is present at position 572, and X C of [N0] is present at position 573, numbered according to the amino acid sequence of SEQ ID NO: 982; (ii) XD of [N1] is present at position 574, XE of [N1] is present at position 575, and XF of [N1] is present at position 576, numbered according to the amino acid sequence of SEQ ID NO: 982; Attorney Docket No.14640.0092-00304 (iii) X1 of [N2] is present at position 577, X2 of [N2] is present at position 578, X3 of [N2] is present at position 579, X4 of [N2] is present at position 580, and X5 of [N2]
- loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)), numbered according to the amino acid sequence of SEQ ID NO: 982. 199.
- positions 571-592 e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)
- positions 571-599 e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)
- loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)), numbered according to the amino acid sequence of SEQ ID NO: 982. 200.
- positions 571-592 e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)
- positions 571-599 e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)
- loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)), numbered according to the amino acid sequence of SEQ ID NO: 982. 201.
- positions 571-592 e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)
- positions 571-599 e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)
- Attorney Docket No.14640.0092-00304 202 The AAV particle of any one of embodiments 41-66, 125-145, and 198-201, wherein [B] replaces positions 574-576 (e.g., Q574, S575, and S576), numbered according to the amino acid sequence of SEQ ID NO: 138. 203.
- [B]-[C]-[D] replaces positions 574-579 (e.g., Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 214.
- positions 574-579 e.g., Q574, S575, S576, T577, T578, and A579
- [A]- [B]-[C]-[D] replaces positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 232.
- positions 571-579 e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and A579
- positions 571-586 e.g., T571, N572, N573, Q574, S575, S576, Y577, P578, A579, E580, V581, V582, Q583, K584, T585, A586
- AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-233 wherein: (i) X A of [A] is present at position 571, X B of [A] is present at position 572, and X C of [A] is present at position 573, numbered according to the amino acid sequence of SEQ ID NO: 982; (ii) X1 of [B] is present at position 574, X2 of [B] is present at position 575, and X3 of [B] is present at position 576, numbered according to the amino acid sequence of SEQ ID NO: 982; (iii) [C] is present at positions 577-584, numbered according to the amino acid sequence of SEQ ID NO: 982; and/or (iv) X4 of [D] is present at position 585 and position X5 of [D] is present at position 586, numbered according to the amino acid sequence of SEQ ID NO: 982.
- the AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-237, wherein the capsid variant comprises, from N-terminus to C-terminus, [B]-[C]. 239.
- loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids Attorney Docket No.14640.0092-00304 TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)), numbered according to the amino acid sequence of SEQ ID NO: 982. 244.
- positions 571-592 e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)
- positions 571-599 e.g., the positions of amino acids Attorney Docket No.14640.0092-00304 TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)
- loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)) numbered according to the amino acid sequence of SEQ ID NO: 982. 245.
- positions 571-592 e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)
- positions 571-599 e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)
- An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant) comprising: (a) any one of the amino acid sequences provided in Table 1, 2A, 2B, 14, or 23-28; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of the amino acid sequences provided in Tables 1, 2A, 2B, 14, or 23-28; (c) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences provided in Table 1, 2A, 2B, 14, or 23-28; or (d) an amino sequence comprising one, two, or three but no more than four modifications
- An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant) comprising: (a) the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583-1587, 1590, 1591-1593, 1598-1608, and 1610-1624; Attorney Docket No.14640.0092-00304 (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of
- An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant) comprising: (a) the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080; (c) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080; or (d) an amino sequence comprising one, two, or
- An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant) comprising: (a) the amino acid sequence of any one of SEQ ID NOs: 1021, 1024, 1232, 1300, 1327, 1533, 1538, 1585, 1590, and 1591; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of SEQ ID NOs: 1021, 1024, 1232, 1300, 1327, 1533, 1538, 1585, 1590, and 1591; (c) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one
- the AAV particle of any one of embodiments 267-270, wherein the AAV capsid variant comprises at least 3, at least 4, at least 5, at least 6, or at least 7 consecutive amino acids from SEQ ID NO: 943 or of any one of SEQ ID NOs: 946-966. 272.
- the AAV particle of embodiment 271, wherein the at least 3 consecutive amino acids comprise YPA. 273.
- the AAV particle of embodiment 271, wherein the at least 4 consecutive amino acids comprise YPAE (SEQ ID NO: 21).
- the AAV particle of embodiment 271, wherein the at least 5 consecutive amino acids comprise YPAEV (SEQ ID NO: 1). 275.
- the AAV particle of embodiment 271, wherein the at least 6 consecutive amino acids comprise YPAEVV (SEQ ID NO: 725). 276. The AAV particle of embodiment 271, wherein the at least 7 consecutive amino acids comprise YPAEVVQ (SEQ ID NO: 726). 277. The AAV particle of embodiment 271, wherein the amino acid sequence comprises YPAEVVQK (SEQ ID NO: 943). 278.
- the AAV particle of any one of embodiments 267-277 or embodiment 282, wherein the AAV capsid variant comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). 286.
- the AAV particle of embodiment 267, wherein the AAV capsid variant comprises: (a) the amino acid sequence of any one of SEQ ID NOs: 2024-2063; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of SEQ ID NOs: 2024-2063; Attorney Docket No.14640.0092-00304 (c) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063; or (d) an amino sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063.
- the AAV particle of embodiment 267 or 290 wherein the different amino acids, of the amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063, are present at one or more of the following positions: (i) position 1, wherein the different amino acid is T or L; (ii) position 2, wherein the different amino acid is N, L, K, A, T, or P; (iii) position 3, wherein the different amino acid is N, K, L, A, Y, or S; (iv) position 4, wherein the different amino acid is Q, L, T, S, F, Y, K, or A; (v) position 5, wherein the different amino acid is S, H, A, M, Q, T, V, or F; (vi) position 6, wherein the different amino acid is S, P, V, A, Q, L, T, N, or M; (vii) position 7, wherein the different amino acid is Y, H, S, V, V
- the AAV particle of embodiment 267, wherein the AAV capsid variant comprises: (a) the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842- 2023, and 2087-2112; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798- 1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842-2023, and 2087-2112; (c) an amino acid sequence comprising one, two, or three
- Attorney Docket No.14640.0092-00304 310 Attorney Docket No.14640.0092-00304 310.
- the AAV particle of any one of the embodiments provided herein, wherein the AAV capsid variant further comprises a modification in loop I, II, IV, and/or VI. 322.
- the AAV particle of any one of the embodiments provided herein, wherein the AAV capsid variant comprises a VP1 protein, a VP2 protein, a VP3 protein, or a combination thereof. 329.
- the AAV capsid variant comprises the amino acid sequence corresponding to positions 193-731, e.g., a VP3, of SEQ ID NO: 982, or a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. 331.
- AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, 294-306, 321-328, and 333-335 comprising one, two, or three but no more than four different amino acids, relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 739; or an amino acid sequence at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity to the amino acid sequence of SEQ ID NO: 739. 337.
- the AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 344 which has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. 351.
- the AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 344 which has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982. 352.
- the AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 344 which has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139. 353.
- the AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 344 which transduces a brain region, e.g., a temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum, optionally wherein the level of transduction is at least 1.5, at least 2.2, at least 2.4, at least 2.5, at least 2.6, at least 2.7, at least 3.0, at least 3.2, at least 3.5, at least 3.7, at Attorney Docket No.14640.0092-00304 least 4.0, at least 4.2, at least 4.5, at least 4.7, at least 4.9, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, or at least 35-fold greater as compared to
- a non-human primate and rodent e.g., rat and/or mouse
- a non-human primate and rodent e.g., rat and/or mouse
- AAV particle of embodiment 356 or 357, wherein the at least two to at least three species are Macaca fascicularis, Chlorocebus sabaeus, Callithrix jacchus, rat, and/or mouse (e.g., BALB/c mice). 359.
- the AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 359 which delivers an increased level of a payload to a brain region, optionally wherein the level of the payload is increased by at least 20, at least 25, at least 30, or at least 35-fold, as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139, e.g., when measured by an assay, e.g., a qRT-PCR or a qPCR assay (e.g., as described in Example 2). 361.
- an assay e.g., a qRT-PCR or a qPCR assay (e.g., as described in Example 2).
- the AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 360 which delivers an increased number and/or level of viral genomes to a brain region, optionally wherein the number and/or level of viral genomes is increased by at least 1.5, at least 2.2, at least 2.4, at least 2.5, at least 2.6, at least 2.7, at least 3.0, at least 3.2, at least 3.5, at least 3.7, at least 4.0, at least 4.2, at least 4.5, at least 4.7, at least 4.9, or at least 5-fold as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139, e.g., when measured by an assay, e.g., a qRT-PCR or a qPCR assay (e.g., as described in Example 2).
- an assay e.g., a qRT-PCR or a qPCR assay
- the AAV particle of any one of the embodiments provided herein which shows preferential transduction in a brain region relative to transduction in the dorsal root ganglia (DRG). 366.
- the AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 365 which is capable of transducing neuronal cells. Attorney Docket No.14640.0092-00304 367.
- the AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 366 which is capable of transducing non-neuronal cells, e.g., glial cells (e.g., oligodendrocytes).
- non-neuronal cells e.g., glial cells (e.g., oligodendrocytes).
- the AAV particle of any one of embodiments 67-267, 269, 271-277, 282, 285, 288, 289, 294-306, and 321-349 which has an increased tropism for a heart cell or heart tissue, e.g., a heart ventricle or heart atrium, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. 370.
- the AAV particle of any one of embodiments 67-267, 269, 271-277, 282, 285, 288, 289, 294-306, 321-349, and 369 which has an increased tropism for a heart cell or tissue, e.g., a heart ventricle or heart atrium, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139. 371.
- the AAV particle of any one of embodiments 67-267, 269, 271-277, 282, 285, 288, 289, 294-306, 321-349, 369, and 370 which delivers an increased level of a payload to a heart region, optionally wherein the level of the payload is increased by at least 1.5, at least 2, or at least 2.5-fold, as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139, e.g., when measured by an assay, e.g., an IHC assay or a RT-ddPCR assay (e.g., as described in Example 2). 372.
- an assay e.g., an IHC assay or a RT-ddPCR assay (e.g., as described in Example 2).
- the AAV particle of any one of embodiments 67-267, 269, 271-277, 282, 285, 288, 289, 294-306, 321-349, and 369-371 which has an increased tropism for a heart cell or tissue, e.g., a heart ventricle or heart atrium, relative to the tropism of a an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982. 373.
- the AAV particle of any one of embodiments 67-267, 269, 271-277, 282, 285, 288, 289, 294-306, 321-349, and 369-372 which is enriched at least 4, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, or at least 50-fold in the heart compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982, e.g., when measured by an assay as described in Example 4.
- Attorney Docket No.14640.0092-00304 374 is enriched at least 4, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, or at least 50-fold in the heart compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982, e.g., when measured by an assay as described in Example 4.
- the AAV particle of any one of embodiments 267, 290, 291, 321-328, 331, 332, 344, and 374 which is enriched at least 2, at least 3, at least 4, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, or at least 35-fold in the muscle compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982, e.g., when measured by an assay as described in Example 4.
- the AAV particle of any one of embodiments 267, 292, 293, 321-328, 331, 332, 344, and 377 which is enriched at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 65, at least 70, at least 75, at least 80, at least 85, at least 90, at least 95, at least 100, at least 110, at least 115, at least 120, at least 130, at least 140, at least 150, at least 160, at least 170, at least 180, at least 185, or at least 190-fold in the liver compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982, e.g., when measured by an assay as described in Example 4.
- AAV particle of any one of embodiments 1-378 wherein the AAV particle comprises a viral genome comprising a nucleotide sequence encoding a Rep protein, e.g., a non-structural protein, wherein the Rep protein comprises a Rep78 protein, a Rep68 protein, Rep52 protein, and/or a Rep40 protein (e.g., a Rep78 protein and a Rep52 protein).
- a Rep protein e.g., a non-structural protein
- Rep protein comprises a Rep78 protein, a Rep68 protein, Rep52 protein, and/or a Rep40 protein (e.g., a Rep78 protein and a Rep52 protein).
- a Rep protein e.g., a non-structural protein
- the Rep protein comprises a Rep78 protein, a Rep68 protein, Rep52 protein, and/or a Rep40 protein (e.g., a Rep78 protein and a Rep52 protein).
- Attorney Docket No.14640.0092-00304 [Embodiments 382-445 are intentionally absent.] 446.
- a cell e.g., a host cell, comprising the AAV particle of any one of embodiments 1-381. 447.
- the cell of embodiment 446, wherein the cell is a mammalian cell or an insect cell.
- the cell of any one of embodiments 446-448, wherein the cell is a cell of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum. 450.
- a method of making an AAV particle of embodiment 381, comprising (i) providing a cell comprising a viral genome comprising a frataxin (FXN)-encoding sequence and a nucleic acid encoding an AAV capsid variant; and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant; thereby making the AAV particle. 451.
- the method of embodiment 450 further comprising, prior to step (i), introducing a nucleic acid comprising the viral genome into the host cell. 452.
- a pharmaceutical composition comprising the AAV particle of any one of embodiments 1-381, and a pharmaceutically acceptable excipient.
- a method of delivering a frataxin protein to a subject comprising administering an effective amount of the pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381.
- Attorney Docket No.14640.0092-00304 456 The method of embodiment 455, wherein the frataxin protein is delivered to a cell or tissue of the CNS (e.g., a cell or tissue of a brain or spinal cord region). 457.
- the cell or tissue of the CNS is a cell or tissue of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, or cerebellum.
- Embodiment 458 is intentionally absent.
- the method of embodiment 455 or 456, wherein the cell or tissue of the CNS comprises a neuron, a sensory neuron, a motor neuron, an astrocyte, a glial cell, or an oligodendrocyte. 460.
- a method of treating a disorder associated with FXN deficiency in a subject comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381, optionally wherein the subject has, has been diagnosed with having, or is at risk of having the disorder associated with FXN deficiency. 466.
- a method of treating a neurological disorder e.g., a neurodegenerative disorder, in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381, optionally wherein the subject has, has been diagnosed with having, or is at risk of having the neurological disorder.
- Attorney Docket No.14640.0092-00304 467
- a method of treating a muscular disorder or a neuromuscular disorder in a subject comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381, optionally wherein the subject has, has been diagnosed with having, or is at risk of having the muscular disorder or the neuromuscular disorder.
- 468. The method of any one of embodiments 465-467, wherein the genetic disorder, neurological disorder, neurodegenerative disorder, muscular disorder, or neuromuscular disorder is Friedreich's Ataxia (FA).
- FFA Friedreich's Ataxia
- the method of any one of embodiments 465-468, wherein the treatment comprises prevention of progression of the disease or disorder in the subject.
- 470 The method of any one of embodiments 460-469, wherein the subject is a human.
- Embodiments 474-475 are intentionally absent.] 476.
- the pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381 for use in a method of treating a genetic disorder, a neurological disorder, a neurodegenerative disorder, a muscular disorder, or a neuromuscular disorder.
- the encoded frataxin (FXN) protein comprises an amino acid sequence of SEQ ID NO: 1825, or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the amino acid sequence of SEQ ID NO: 1825, wherein, optionally, the encoded FXN protein is a human FXN protein. 483.
- the FXN-encoding sequence comprises a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to the nucleotide sequence of SEQ ID NO: 1824. 484.
- the frataxin-encoding sequence comprises a nucleotide sequence that is at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to the nucleotide sequence of SEQ ID NO: 1824. 485.
- the AAV particle of embodiment 483 or embodiment 484, wherein the frataxin-encoding sequence comprises a nucleotide sequence at least 95% identical to the nucleotide sequence of SEQ ID NO: 1824. 486.
- the AAV particle of any one of embodiments 483-485, wherein the frataxin-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824. Attorney Docket No.14640.0092-00304 487.
- the AAV particle of any one of embodiments 483-486, wherein the frataxin-encoding sequence consists of the nucleotide sequence of SEQ ID NO: 1824. 488.
- the AAV particle of embodiment 488 wherein: (i) the at least 4 consecutive amino acids comprise YPAE (SEQ ID NO: 21), optionally wherein YPAE (SEQ ID NO: 21) is present at positions 577-580 numbered according to the amino acid sequence of SEQ ID NO: 982; (ii) the at least 5 consecutive amino acids comprise YPAEV (SEQ ID NO: 1), optionally wherein YPAEV (SEQ ID NO: 1) is present at positions 577-581 numbered according to the amino acid sequence of SEQ ID NO: 982; (iii) the at least 6 consecutive amino acids comprise YPAEVV (SEQ ID NO: 725), optionally wherein YPAEVV (SEQ ID NO: 725) is present at positions 577-582 numbered according to the amino acid sequence of SEQ ID NO: 982; or (iv) the at least 7 consecutive amino acids comprise YPAEVVQ (SEQ ID NO: 726), optionally wherein YPAEVVQ (SEQ ID NO: 726)
- the AAV particle of embodiment 488 or 489, wherein the AAV capsid variant comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) present at positions 577-584 numbered according to SEQ ID NO: 982. 491.
- the AAV particle of any one of embodiments 488-490, wherein the AAV capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 95% identity to the amino acid sequence of SEQ ID NO: 982; or (ii) a VP2 protein comprising an amino acid sequence having at least 95% identity to the amino acid sequence of SEQ ID NO: 738. 492.
- the AAV capsid variant comprises (i) a VP1 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 982; (ii) a VP2 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 738; or (iii) a VP3 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 739; or wherein the AAV capsid variant is encoded by the nucleotide sequence of SEQ ID NO: 984 or a sequence at least 90% identical thereto. 494.
- the promoter comprises a human elongation factor 1 ⁇ -subunit (EF1 ⁇ ) promoter, a cytomegalovirus (CMV) immediate-early enhancer and/or promoter, a chicken ⁇ -actin (CBA) promoter, a CAG promoter, a ⁇ glucuronidase (GUSB) promoter, a ubiquitin C (UBC) promoter, a neuron-specific enolase (NSE) promoter, a platelet-derived growth factor (PDGF) promoter, a platelet-derived growth factor B-chain (PDGF- ⁇ ) promoter, a intercellular adhesion molecule 2 (ICAM-2) promoter, a synapsin (Syn) promoter, a methyl-CpG binding protein 2 (Me
- EF1 ⁇ human e
- the AAV particle of embodiment 495, wherein the promoter comprises a CMV promoter. 497.
- the AAV particle of embodiment 495, wherein the promoter comprises a CBA promoter. 498.
- the AAV particle of embodiment 497, wherein the promoter is or comprises a truncated CBA promoter. 499.
- the AAV particle of embodiment 498, wherein the truncated CBA promoter is 50-400 nucleotides in length (e.g., 100-332 nucleotides in length).
- Attorney Docket No.14640.0092-00304 500 is 50-400 nucleotides in length (e.g., 100-332 nucleotides in length).
- the AAV particle of embodiment 499, wherein the truncated CBA promoter comprises or consists of the nucleotide sequence of any one of SEQ ID NOs: 1738, 1740, or 1742 or a nucleotide sequence that is at least 95% identical to any one of SEQ ID NOs: 1738, 1740, or 1742.
- the AAV particle of embodiment 496, wherein the promoter comprises a truncated CMV promoter. 502.
- the AAV particle of embodiment 501, wherein the truncated CMV promoter is 109 nucleotides in length. 503.
- the AAV particle of embodiment 502, wherein the truncated CMV promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1750 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1750. 504.
- the truncated CMV promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1750 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1750. 504.
- a microRNA (miR) binding site e.g., a miR binding site that modulates, e.g., reduces, expression of the FXN protein encoded by the viral genome in a cell or tissue where the corresponding miRNA is expressed.
- the AAV particle of embodiment 510 wherein the viral genome encodes 3 copies of the miR122 binding site, optionally wherein the 3 copies of the miR122 binding site are identical, further optionally wherein the 3 copies of the miR122 binding site are continuous.
- the AAV particle of any one of embodiments 494-513, wherein the viral genome further comprises at least one inverted terminal repeat (ITR) region, optionally wherein the at least one ITR region is an AAV2 ITR. 515.
- ITR inverted terminal repeat
- the AAV particle of embodiment 514 wherein the viral genome comprises a 5’ ITR region and a 3’ ITR region, optionally wherein the 5’ ITR region and the 3’ ITR region is each an AAV2 ITR. 516.
- the AAV particle of embodiment 519, wherein Attorney Docket No.14640.0092-00304 (a) the exon region comprises an ie1 exon region and/or an HB exon region, and/or (b) the intron region comprises: an ie1 intron 1 comprising or consisting of the nucleotide sequence of SEQ ID NO: 1819 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or a hBglobin intron 2 comprising the nucleotide sequence of SEQ ID NO: 1820 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least
- hGH human growth hormone
- the AAV particle of embodiment 523, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1797. 525.
- the AAV particle of embodiment 523 wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1801. 526.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1801. 526.
- the AAV particle of embodiment 523 wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1808.
- Attorney Docket No.14640.0092-00304 527 is the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1808.
- the AAV particle of embodiment 530 wherein the viral genome further comprises a 5’ inverted terminal repeat (ITR) region and/or a 3’ ITR region. 532.
- ITR inverted terminal repeat
- a 5’ ITR region comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucle
- the AAV particle of embodiment 533 wherein the viral genome comprises, e.g., in 5’ to 3’ order: (i) a 5’ ITR region; (ii) a promoter; (iii) an intron and/or exon region; (iv) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; and (vi) a 3’ ITR region, optionally wherein the viral genome further comprises a filler sequence.
- the viral genome comprises, e.g.
- the 5’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1742 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical (e.g., at least 90%,
- the AAV particle of embodiment 535, wherein the promoter consists of the nucleotide sequence of SEQ ID NO: 1742 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 537.
- the 5’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1750 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical (e.g., at least 90%, at least 9
- the AAV particle of embodiment 537, wherein the promoter consists of the nucleotide sequence of SEQ ID NO: 1750 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 539.
- the 5’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1738 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical (e.g., at least 90%, at least 9
- the AAV particle of embodiment 539, wherein the promoter consists of the nucleotide sequence of SEQ ID NO: 1738 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 541.
- the 5’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1740 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
- the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical (e.g., at least 90%, at least 9
- the AAV particle of embodiment 541, wherein the promoter consists of the nucleotide sequence of SEQ ID NO: 1740 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- the promoter consists of the nucleotide sequence of SEQ ID NO: 1740 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- Embodiments 543-696 are intentionally absent.] 697.
- An adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome
- the AAV capsid variant comprises: (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, wherein the VP1 protein comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); (b) a VP2 protein comprising the amino acid sequence of SEQ ID NO: 738 or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto,
- the AAV particle of embodiment 697 wherein: (a) the VP1 protein comprises an amino acid sequence that is at least 95% identical to SEQ ID NO: 982; (b) a VP2 protein comprises an amino acid sequence that is at least 95% identical to SEQ ID NO: 738; and/or (c) a VP3 protein comprises an amino acid sequence that is at least 95% identical to SEQ ID NO: 739. 699.
- the AAV particle of embodiment 698 wherein: (a) the VP1 protein comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 982; (b) a VP2 protein comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 738; and/or (c) a VP3 protein comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 739. 700.
- FXN frataxin
- the viral genome comprises a nucleotide sequence of any one of SEQ ID NOs: 1797, 1801, 1808, and 1809, or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to any one of SEQ ID NOs: 1797, 1801, 1808, and 1809; and wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982. 709.
- the method of embodiment 711, wherein the cell is an HEK293 cell or an Sf9 cell. 713.
- a pharmaceutical composition comprising the AAV particle of any one of embodiments 485-703 and a pharmaceutically acceptable excipient. 714.
- a method of delivering a FXN protein to a subject comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 713 or the AAV particle of any one of embodiments 482-704, thereby delivering the FXN protein. 715.
- a method of treating a disorder associated with frataxin (FXN) deficiency in a subject comprising administering an effective amount of the pharmaceutical composition of embodiment 713 or the AAV particle of any one of embodiments 485-703, thereby treating the disorder, optionally wherein the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency, further optionally wherein the disorder is FA. 717.
- any one of embodiments 716-718 wherein the treatment stabilizes, slows the progression of, or improves the subject’s FA as determined by the modified Friedreich Ataxia Rating Scale (mFARS), the Scale for the Assessment and Rating of Ataxia (SARA), and/or the International Cooperative Ataxia Rating Scale (ICARS).
- mFARS modified Friedreich Ataxia Rating Scale
- SARA Scale for the Assessment and Rating of Ataxia
- ICARS International Cooperative Ataxia Rating Scale
- 723 The method of any one of embodiments 716-718, wherein the treatment slows the subject’s progression of FA as measured by mFARS, SARA, or ICARS relative to an individual with the disorder associated with FXN deficiency (e.g., FA) who has not been administered the pharmaceutical composition or the AAV particle.
- ICARS International Cooperative Ataxia Rating Scale
- AAV particle is administered to the subject intravenously, intracerebrally (IC), via intrathalamic (ITH) administration, intramuscularly, intrathecally, intracerebroventricularly, via intraparenchymal administration, via focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration, or via intra-cisterna magna injection (ICM). 725.
- any one of embodiments 714-724 wherein the AAV particle is delivered to a cell, tissue, or region of the CNS, e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum, or a combination thereof. 726.
- a cell, tissue, or region of the CNS e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum, or a combination thereof. 726.
- any one of embodiments 714-725 further comprising evaluating (e.g., measuring the level of FXN gene, FXN mRNA, and/or FXN protein expression) in the subject (e.g., in a cell, tissue, or fluid of the subject), optionally wherein the level of FXN protein is measured by an assay described herein. 727.
- evaluating e.g., measuring the level of FXN gene, FXN mRNA, and/or FXN protein expression
- the subject e.g., in a cell, tissue, or fluid of the subject
- the level of FXN protein is measured by an assay described herein. 727.
- the method of embodiment 726, wherein the level of FXN protein expression is measured by an enzyme-linked immunosorbent assay (ELISA), a Western blot, an immunohistochemistry assay, or a frataxin biofluid assay. 728.
- ELISA enzyme-linked immunosorbent assay
- the cell or tissue is a cell or tissue of the central nervous system (e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, Attorney Docket No.14640.0092-00304 caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) or a peripheral cell or tissue (e.g., the liver, heart, and/or muscle). 729.
- the central nervous system e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, Attorney Docket No.14640.0092-00304 caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum
- a peripheral cell or tissue e.g., the liver
- a cell, tissue e.g., a cell or tissue of the CNS, e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum), and/or fluid (e.g., CSF and/or serum) of the subject, optionally wherein the level of FXN protein or gene expression is increased by 0.5-3x as compared to baseline; and/or (ii) the number and/or level of viral genomes (VG) per cell in a CNS tissue (e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate
- VG viral genomes
- the at least one additional therapeutic agent and/or therapy comprises an immunosuppressant, optionally wherein the immunosuppressant is a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine. 733.
- the immunosuppressant is a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacroli
- FIGs.1A-1D show immunohistochemistry images from various CNS and peripheral tissues isolated from NHPs (cynomolgus macaques) at 28 days post intravenous administration of AAV particles comprising the TTN-002 capsid variant (top panels) or AAV9 control capsid (bottom panels) and a self- complementary genome encoding a cynomolgus monkey frataxin protein fused to an HA tag driven by a chicken beta actin (CBA) heterologous promoter.
- CBA chicken beta actin
- FIG.1A shows, from left to right, the cerebellum (Purkinje cell layer), spinal cord (cervical), cortex (temporal), and the brainstem.
- FIG.1B shows, from left to right, the globus pallidus, the hippocampus, the thalamus, the putamen, and the dentate.
- FIG.1C shows, from left to right, the whole brain (level H), the whole brain (level K), and the cerebellum.
- FIG. 1D shows, from left to right, the spinal cord (thoracic), the DRG (thoracic), the liver, and the heart.
- FIGs.2A-2D depict frataxin expression and the number of viral genomes per cell in the heart (FIG.2A); cerebellum (FIG.2B); lumbar and DRG (FIG.2C); and liver (FIG.2D).
- FIG.3 shows an exemplary ITR-to-ITR construct encoding FXN.
- FIG.4A shows biodistribution of AAV particles in the cerebellum of wildtype (“WT”) or frataxin-deficient (“FXN-deficient”) mice.
- the AAV particles comprise a TTN-002 capsid encapsulating a viral genome comprising SEQ ID NO: 1797 (“TTN-002_FXN”).
- FIG.4B shows expression of human frataxin (“hFXN”) in the cerebellum of wildtype (“WT”) or frataxin-deficient (“FXN-deficient”) mice after administration of AAV particles comprising a TTN-002 capsid encapsulating a viral genome comprising SEQ ID NO: 1797 (“TTN-002_FXN”).
- AAV particles comprising a TTN-002 capsid encapsulating a viral genome comprising SEQ ID NO: 1797 (“TTN-002_FXN”).
- the present disclosure provides a method for delivering the AAV capsid variant comprising the sequence encoding the FXN protein to a cell or tissue in a subject. In some embodiments, the present disclosure provides a method for delivering the AAV capsid variant, thereby providing a FXN protein, e.g., a human FXN Attorney Docket No.14640.0092-00304 protein, to a cell or tissue in a subject.
- the AAV capsid variants described herein have enhanced CNS tropism compared to other cells or tissues in the body, e.g., liver and/or the DRG.
- AAV have proven to be useful as a biological tool due to their relatively simple structure, their ability to infect a wide range of cells (including quiescent and dividing cells) without integration into the host genome and without replicating, and their relatively benign immunogenic profile.
- Engineered AAV capsids with improved brain tropism represent an attractive solution to the limitations of CNS delivery.
- AAV-derived vectors are promising tools for clinical gene transfer because of their non-pathogenic nature, their low immunogenic profile, low rate of integration into the host genome, and long-term transgene expression in non-dividing cells.
- the transduction efficiency of naturally occurring AAVs in certain organs is too low for clinical applications, and capsid neutralization by pre- existing neutralizing antibodies may prevent treatment of a large proportion of patients.
- the genome of the virus may be modified to contain a minimum of components for the assembly of a functional recombinant virus, or viral particle, which is loaded with or engineered to target a particular tissue and express or deliver FXN.
- the genome of the virus may encode a FXN protein, and the viral particle comprising said genome may be delivered to a target cell, tissue, or organism.
- the genome encodes a human FXN protein, e.g., a wildtype human FXN protein.
- the target cell is a CNS cell.
- the target tissue is a CNS tissue.
- the target CNS tissue may be brain tissue.
- expression vectors e.g., an adeno-associated viral vector (AAVs) or AAV particle, e.g., an AAV particle described herein, can be used to administer and/or deliver a gene encoding FXN (e.g., human FXN) preferentially to the CNS (e.g., the brain or the spinal cord).
- AAVs adeno-associated viral vector
- AAV particle e.g., an AAV particle described herein
- FXN e.g., human FXN
- CNS e.g., the brain or the spinal cord
- compositions and methods which may provide for improved features compared to prior AAV-mediated enzyme replacement approaches, including (i) increased biodistribution of the AAV particle throughout the CNS (e.g., the cortex, striatum, thalamus, cerebellum, brainstem, and/or spinal cord) of the subject, (ii) elevated FXN expression, e.g., FXN mRNA or protein expression, in multiple brain regions (e.g., cortex, thalamus, and brain stem) of the subject; and (iii) reduced biodistribution of the AAV particle in the liver and/or DRG of the subject.
- CNS e.g., the cortex, striatum, thalamus, cerebellum, brainstem, and/or spinal cord
- FXN expression e.g., FXN mRNA or protein expression
- AAV capsid variants e.g., AAV5 capsid variants
- AAV9 AAV capsid variants
- improved properties compared to wildtype AAV5 and/or wildtype AAV9, such as (i) increased penetrance through the blood brain barrier following intravenous administration, (ii) wider distribution throughout the multiple brain regions, e.g., temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, Attorney Docket No.14640.0092-00304 thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum, (iii) elevated FXN expression in multiple brain regions, (iv) wider distribution in one or more peripheral tissues, e.g., the heart and/or muscle, and/or (v) elevated FXN expression in one or more peripheral tissues.
- the AAV capsids described herein enhance the delivery of FXN to multiple regions of the brain including, for example, the temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum.
- the AAV capsids described herein enhance the delivery of FXN to the forebrain.
- the AAV capsids described herein enhance the delivery of FXN to the frontal cortex.
- the AAV capsids described herein enhance the delivery of FXN to the spinal cord.
- the AAV capsids described herein enhance the delivery of FXN to the cerebellum. In some embodiments, the AAV capsids described herein enhance the delivery of FXN to the dentate nucleus. [083] As demonstrated in the Examples, the AAV capsid variants with enhanced brain tropism described herein are capable of significantly increasing FXN mRNA expression in the brain. [084] Thus, the compositions and methods described herein can be used in the treatment of Friedreich’s Ataxia (FA).
- FA Friedreich’s Ataxia
- the disclosure provides an AAV particle comprising one of the AAV capsid variants disclosed herein and an AAV viral genome comprising a nucleotide sequence comprising a truncated promoter region and a sequence encoding a FXN protein (e.g., comprising the nucleotide sequence of any one of SEQ ID NOs: 1797, 1801, 1808, 1809) for use in treating FA.
- AAV Adeno-associated viral particles
- AAVs have a genome of about 5,000 nucleotides in length and contain two open reading frames encoding the proteins responsible for replication (Rep) and the structural protein of the capsid (Cap).
- the open reading frames are flanked by two Inverted Terminal Repeat (ITR) sequences, which serve as the origin of replication of the viral genome.
- ITR Inverted Terminal Repeat
- the wild-type AAV viral genome comprises nucleotide sequences for two open reading frames, one for the four non-structural Rep proteins (Rep78, Rep68, Rep52, Rep40, encoded by Rep genes) and one for the three capsid, or structural, proteins (VP1, VP2, VP3, encoded by capsid genes or Cap genes).
- the Rep proteins are important for replication and packaging, while the capsid proteins are assembled to create the protein shell of the AAV, or AAV capsid.
- VP1 refers to amino acids 1-724
- VP2 refers to amino acids 137-724
- VP3 refers to amino acids 193-724.
- VP1 comprises amino acids 1-731
- VP2 comprises amino acids 137-731
- VP3 comprises amino acids 193-731.
- VP1 is the full-length capsid protein sequence
- VP2 and VP3 are shorter components of the whole.
- changes in the sequence in the VP3 region are also changes to Attorney Docket No.14640.0092-00304 VP1 and VP2, however, the percent difference as compared to the parent sequence will be greatest for VP3 since it is the shortest sequence of the three.
- the nucleic acid sequence encoding these proteins can be similarly described.
- the three capsid proteins assemble to create the AAV capsid.
- the AAV capsid typically comprises a molar ratio of 1:1:10 of VP1:VP2:VP3.
- the AAV vector typically requires a co-helper (e.g., adenovirus) to undergo productive infection in cells. In the absence of such helper functions, the AAV virions essentially enter host cells but do not integrate into the cells’ genome. [087] AAV vectors have been investigated for delivery of gene therapeutics because of several unique features.
- a co-helper e.g., adenovirus
- Non-limiting examples of the features include (i) the ability to infect both dividing and non-dividing cells; (ii) a broad host range for infectivity, including human cells; (iii) wild-type AAV has not been associated with any disease and has not been shown to replicate in infected cells; (iv) the lack of cell-mediated immune response against the vector, and (v) the non-integrative nature in a host chromosome thereby reducing potential for long-term genetic alterations.
- infection with AAV vectors has minimal influence on changing the pattern of cellular gene expression (Stilwell and Samulski et al., Biotechniques, 2003, 34, 148, the contents of which are herein incorporated by reference in their entirety).
- AAV vectors for FXN protein delivery may be recombinant viral vectors which are replication defective as they lack sequences encoding functional Rep and Cap proteins within the viral genome.
- the defective AAV vectors may lack most or all coding sequences and essentially only contain one or two AAV ITR sequences and a sequence encoding a FXN protein.
- the AAV particles of the present disclosure may be introduced into mammalian cells.
- AAV vectors may be modified to enhance the efficiency of delivery. Such modified AAV vectors of the present disclosure can be packaged efficiently and can be used to successfully infect the target cells at high frequency and with minimal toxicity.
- AAV particles of the present disclosure may be used to deliver FXN protein to the central nervous system (see, e.g., U.S. Pat. No.6,180,613; the contents of which are herein incorporated by reference in their entirety) or to specific tissues of the CNS.
- the compositions described herein may have additional conservative or non-essential amino acid substitutions, which do not have a substantial effect on their functions.
- AAV capsid variants disclosed herein comprise one or more modifications (e.g., one or more insertions and/or substitutions) in the loop VIII region of AAV5.
- the AAV5 loop VIII region comprises amino acid positions 571-579, e.g., position 577, numbered according to SEQ ID NO: 138. In some embodiments, the AAV5 loop VIII region is present (is located) at amino acids corresponding to positions 571-579 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV5 loop VIII region (e.g., positions 571-579, e.g., Attorney Docket No.14640.0092-00304 position 577) protrudes above the 3-fold axis of symmetry, e.g., is a surface-exposed location in the AAV5 capsid, e.g., as described in Govindasamy et al.
- loop VIII e.g., as used in loop VIII
- variable region e.g., variable region VIII
- VR e.g., VR-VIII
- loop VIII comprises positions 571-599 (e.g., amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)), numbered according to SEQ ID NO: 982.
- the AAV5 loop VIII is present (is located) at amino acids corresponding to positions 571-599 (e.g., amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)) of the amino acid sequence of SEQ ID NO: 982.
- loop VIII or variable region VIII VR-VIII is as described in Govindasamy et al. (supra) (the contents of which are hereby incorporated by reference in their entirety).
- the AAV particles and payloads of the disclosure may be delivered to one or more target cells, tissues, organs, or organisms.
- the AAV particles of the disclosure demonstrate enhanced tropism for a target cell type, tissue, or organ.
- the AAV particle may have enhanced tropism for cells and tissues of the central or peripheral nervous systems (CNS and PNS, respectively).
- an AAV particle of the disclosure may, in addition, or alternatively, have decreased tropism for a cell-type, tissue or organ.
- AAV are used as a biological tool due to a relatively simple structure, their ability to infect a wide range of cells (including quiescent and dividing cells) without integration into the host genome and without replicating, and their relatively benign immunogenic profile.
- the genome of the virus may be manipulated to contain a minimum of components for the assembly of a functional recombinant virus, or viral particle, which is loaded with or engineered to target a particular tissue and express or deliver a desired payload.
- the AAV is a recombinant AAV.
- the wild-type AAV viral genome is a linear, single-stranded DNA (ssDNA) molecule approximately 5,000 nucleotides (nt) in length.
- ssDNA single-stranded DNA
- ITRs cap the viral genome at both the 5’ and the 3’ end, providing origins of replication for the viral genome.
- an AAV viral genome comprises two ITR sequences.
- the ITRs have a characteristic T-shaped hairpin structure defined by a self-complementary region (145nt in wild-type AAV) at the 5’ and 3’ ends of the ssDNA which form an energetically stable double stranded region.
- the double stranded hairpin structures comprise multiple functions including, but not limited to, acting as an origin for DNA replication by functioning as primers for the endogenous DNA polymerase complex of the host viral replication cell.
- AAV particles described herein may be produced recombinantly and may be based on AAV reference sequences.
- scAAV viral genomes In addition to single stranded AAV viral genomes (e.g., ssAAVs), the present disclosure also provides for self-complementary AAV (scAAV) viral genomes.
- scAAV viral genomes Attorney Docket No.14640.0092-00304 contain DNA strands which anneal together to form double stranded DNA. By skipping second strand synthesis, scAAVs allow for rapid expression in the transduced cell.
- the AAV particle is an scAAV.
- the AAV particle is an ssAAV.
- Methods for producing and/or modifying AAV particles are disclosed in the art such as pseudotyped AAV vectors (PCT Patent Publication Nos.
- the AAV particles of the disclosure comprising an AAV capsid variant, and a viral genome, have enhanced tropism for a cell-type or a tissue, e.g., a CNS cell-type, region, or tissue.
- AAV particles comprising an AAV capsid variant comprising a peptide (e.g., inserted into a wildtype AAV capsid and/or in substitution of an amino acid or peptide in a wildtype AAV capsid) for enhanced or improved transduction of a target tissue (e.g., cells of the CNS or PNS).
- the nucleic acid encoding the peptide is an isolated nucleic acid.
- the nucleic acid encoding the peptide is a recombinant nucleic acid.
- the peptide may increase distribution of an AAV particle to a cell, region, or tissue of the CNS.
- the cell of the CNS may be, but is not limited to, neurons (e.g., excitatory, inhibitory, motor, sensory, autonomic, sympathetic, parasympathetic, Purkinje, Betz, etc.), glial cells (e.g., microglia, astrocytes, oligodendrocytes) and/or supporting cells of the brain such as immune cells (e.g., T cells).
- neurons e.g., excitatory, inhibitory, motor, sensory, autonomic, sympathetic, parasympathetic, Purkinje, Betz, etc.
- glial cells e.g., microglia, astrocytes, oligodendrocytes
- immune cells e.g., T cells
- the tissue of the CNS may be, but is not limited to, the cortex (e.g., frontal, parietal, occipital, temporal), thalamus, hypothalamus, striatum, putamen, caudate nucleus, hippocampus, entorhinal cortex, basal ganglia, or deep cerebellar nuclei.
- the tissue of the CNS is a temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, cerebellum, cervical spinal cord, thoracic spinal cord, or lumbar spinal cord.
- the peptide may modulate distribution of an AAV particle to a cell, region, or tissue of the CNS. In some embodiments, the peptide may decrease distribution of an AAV particle to the DRG. [0103] In some embodiments, the peptide may increase distribution of an AAV particle to the CNS (e.g., the cortex) after intravenous administration. In some embodiments, the peptide may increase distribution of an AAV particle to the CNS (e.g., the cortex) following focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration.
- FUS focused ultrasound
- FUS-MB microbubbles
- the peptide may modulate distribution of an AAV particle to the PNS (e.g., DRG) after intravenous administration.
- the peptide may increase distribution of an AAV particle to non-DRG cells of the PNS following focused ultrasound (FUS), e.g., Attorney Docket No.14640.0092-00304 coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration.
- FUS focused ultrasound
- the peptide may decrease distribution of an AAV particle to the DRG following focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration.
- FUS focused ultrasound
- the peptide may increase distribution of an AAV particle to a cell, region, or tissue of a heart, e.g., a heart atrium or a heart ventricle.
- the peptide may increase distribution of an AAV particle to a heart cell, region, or tissue after intravenous administration.
- the peptide may increase distribution of an AAV particle to a cell, region, or tissue of a muscle.
- the muscle is a heart muscle (e.g., a heart atrium or a heart ventricle) or a quadriceps.
- the peptide may increase distribution of an AAV particle to a muscle cell, region, or tissue after intravenous administration.
- a peptide may comprise a sequence as set forth in Table 1 (e.g., comprising the amino acid sequence of any one of SEQ ID NOs: 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583-1587, 1590- 1593, 1598-1624, or 2064-2079).
- a peptide may comprise a sequence as set forth in Table 2A or 2B.
- the peptide may comprise a sequence set forth in Tables 14 or 23-28. Table 1.
- a peptide comprises an amino acid sequence having the formula [N2]- [N3], wherein [N2] comprises X 1 , X 2 , X 3 , X 4 , and X 5 and [N3] comprises the amino acid sequence of VQK, VQN, EQK, VKK, VHK, VQQ, or LQK.
- X 1 of [N2] is Y, N, C, or T.
- X 2 of [N2] is P, E, K, T, or Q.
- X 3 of [N2] is A or P.
- X 4 of [N2] is E, S, D, or A.
- X 5 of [N2] is V, L, or E.
- X1 of [N2] is Y.
- X2 of [N2] is P.
- X3 of [N2] is A.
- X 4 of [N2] is E.
- X 5 of [N2] is V.
- the amino acid sequence of [N3] comprises VQK.
- the amino acid sequence of [N3] consists of VQK.
- [N2]-[N3] replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138.
- a peptide comprises an amino acid sequence having the formula [N2]- [N3], wherein [N2] comprises X 1 , X 2 , X 3 , X 4 , and X 5 and [N3] comprises the amino acid sequence of VQK, EQK, VKK, VHK, VQQ, or LQK.
- [N3] comprises the amino acid sequence of VQK, EQK, or VKK.
- [N3] comprises the amino acid sequence VQK. In some embodiments, [N3] consists of the amino acid sequence VQK. In some embodiments, X 1 of [N2] is Y, N, or C. In some embodiments, X 1 of [N2] is Y or N. In some embodiments, X 2 of [N2] is P, K, T, or Q. In some embodiments, X 2 of [N2] is P, T, or Q. In some embodiments, X 3 of [N2] is A or P. In some embodiments, X 3 of [N2] is A. In some embodiments, X 4 of [N2] is E, S, or A.
- X 5 of [N2] is V, L, or E. In some embodiments, X 5 of [N2] is V or L. In some embodiments, X 1 of [N2] is Y. In some embodiments, X 2 of [N2] is P. In some embodiments, X 3 of [N2] is A. In some embodiments, X 4 of [N2] is E. In some embodiments, X 5 of [N2] is V.
- [N2] comprises YPA, YPP, NKA, YTA, YQA, YTP, NPA, CPA, THA, PAE, PPS, KAE, TAE, QAE, TPS, PAA, HAS, AEV, PSL, AEE, or AAV.
- [N2] comprises YPAE (SEQ ID NO: 21), YPPS (SEQ ID NO: 22), NKAE (SEQ ID NO: 23), YTAE (SEQ ID NO: 24), YQAE Attorney Docket No.14640.0092-00304 (SEQ ID NO: 25), YTPS (SEQ ID NO: 26), YPAA (SEQ ID NO: 27), NPAE (SEQ ID NO: 28), CPAE (SEQ ID NO: 29), THAS (SEQ ID NO: 30), PAEV (SEQ ID NO: 17), PPSL (SEQ ID NO: 31), KAEV (SEQ ID NO: 32), TAEV (SEQ ID NO: 16), PAEE (SEQ ID NO: 18), QAEV (SEQ ID NO: 15), TPSL (SEQ ID NO: 33), PAAV (SEQ ID NO: 34), or QAEE (SEQ ID NO: 35).
- [N2] is or comprises YPAEV (SEQ ID NO: 1), YPPSL (SEQ ID NO: 2), NKAEV (SEQ ID NO: 3), YTAEV (SEQ ID NO: 4), YPAEE (SEQ ID NO: 5), YQAEV (SEQ ID NO: 6), YTPSL (SEQ ID NO: 7), YPAAV (SEQ ID NO: 8), NPAEV (SEQ ID NO: 9), CPAEV (SEQ ID NO: 10), or YQAEE (SEQ ID NO: 11).
- [N2] comprises the amino acid sequence of YPAEV (SEQ ID NO: 1).
- the amino acid sequence of [N2] consists of YPAEV (SEQ ID NO: 1).
- [N2]-[N3] comprises the amino acid sequence of AEVVQK (SEQ ID NO: 36), PSLVQK (SEQ ID NO: 37), AEVEQK (SEQ ID NO: 38), AEEVQK (SEQ ID NO: 39), PSLEQK (SEQ ID NO: 40), PSLVKK (SEQ ID NO: 41), AEVVKK (SEQ ID NO: 42), AEVVHK (SEQ ID NO: 43), AAVVQK (SEQ ID NO: 44), AEVVQQ (SEQ ID NO: 45), or AEVLQK (SEQ ID NO: 46).
- [N2]-[N3] comprises the amino acid sequence PAEVVQK (SEQ ID NO: 20), PPSLVQK (SEQ ID NO: 47), KAEVVQK (SEQ ID NO: 48), TAEVVQK (SEQ ID NO: 49), PAEVEQK (SEQ ID NO: 50), PAEEVQK (SEQ ID NO: 51), QAEVVQK (SEQ ID NO: 52), TPSLVQK (SEQ ID NO: 53), PPSLEQK (SEQ ID NO: 54), PPSLVKK (SEQ ID NO: 55), PAEVVKK (SEQ ID NO: 56), PAEVVHK (SEQ ID NO: 57), PAAVVQK (SEQ ID NO: 58), PAEVVQQ (SEQ ID NO: 59), TAEVVKK (SEQ ID NO: 60), PAEVLQK (SEQ ID NO: 61), or QAEEVQK (SEQ ID NO: 62).
- [N2]-[N3] is or comprises YPAEVVQK (SEQ ID NO: 943), YPPSLVQK (SEQ ID NO: 946), NKAEVVQK (SEQ ID NO: 947), YTAEVVQK (SEQ ID NO: 948), YPAEVEQK (SEQ ID NO: 949), YPAEEVQK (SEQ ID NO: 950), YQAEVVQK (SEQ ID NO: 951), YTPSLVQK (SEQ ID NO: 952), YPPSLEQK (SEQ ID NO: 953), YPPSLVKK (SEQ ID NO: 954), YPAEVVKK (SEQ ID NO: 955), YPAEVVHK (SEQ ID NO: 956), YPAAVVQK (SEQ ID NO: 957), NPAEVVQK (SEQ ID NO: 958), YPAEVVQQ (SEQ ID NO: 959), CPAEVVQK (S
- [N2]-[N3] is YPAEVVQK (SEQ ID NO: 943). In some embodiments, [N2]-[N3] comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). In some embodiments, [N2]-[N3] (e.g., comprising the amino acid sequence of SEQ ID NO: 943) replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [0110] In some embodiments, the peptide comprising the amino acid sequence comprising the formula of [N2]-[N3] further comprises [N1], which comprises X D , X E , and X F .
- X D of [N1] is Q, T, S, A, I, L, or H.
- X E of [N1] is S, G, A, or R.
- X F of [N1] is S, K, L, R, A, or T.
- [N1] comprises SK, SL, SS, SR, GA, GS, AS, ST, RS, QS, TS, AG, IG, QA, LG, HS, LS, or QR.
- [N1] is or comprises QSS, QSK, TSL, SSS, QSR, AGA, IGS, QAS, ASS, LGS, QST, HSS, LSS, or QRS.
- the amino acid sequence of [N1] is QSS.
- [N1]-[N2] comprises SSYPA (SEQ ID NO: 63), SKYPA (SEQ ID NO: 64), SLYPA (SEQ ID NO: 65), SRYPA (SEQ ID NO: 66), SSYPP (SEQ ID NO: 67), GAYPA (SEQ ID NO: 68), GSYPA (SEQ ID NO: 69), ASYPA (SEQ ID NO: 70), STNKA (SEQ ID NO: 71), SSYTA (SEQ ID NO: 72), SSYQA (SEQ ID NO: 73), SSYTP (SEQ ID NO: 74), SSNPA (SEQ ID NO: 75), SLCPA (SEQ ID NO: 76), RSYTA (SEQ ID NO: 77), or SSTHA (SEQ ID NO: 78).
- [N1]-[N2] comprises SSYPAE (SEQ ID NO: 79), SKYPAE (SEQ ID NO: 80), SLYPAE (SEQ ID NO: 81), SRYPAE (SEQ ID NO: 82), SSYPPS (SEQ ID NO: 83), GAYPAE (SEQ ID NO: 84), GSYPAE (SEQ ID NO: 85), ASYPAE (SEQ ID NO: 86), STNKAE (SEQ ID NO: 87), SSYTAE (SEQ ID NO: 88), SSYQAE (SEQ ID NO: 89), SSYTPS (SEQ ID NO: 90), SSYPAA (SEQ ID NO: 91), SSNPAE (SEQ ID NO: 92), SLCPAE (SEQ ID NO: 93), RSYTAE (SEQ ID NO: 94), SSTHAS (SEQ ID NO: 95).
- [N1]-[N2] is or comprises QSSYPAEV (SEQ ID NO: 96), QSKYPAEV (SEQ ID NO: 97), TSLYPAEV (SEQ ID NO: 98), SSSYPAEV (SEQ ID NO: 99), QSRYPAEV (SEQ ID NO: 100), QSSYPPSL (SEQ ID NO: 101), AGAYPAEV (SEQ ID NO: 102), IGSYPAEV (SEQ ID NO: 103), QASYPAEV (SEQ ID NO: 104), ASSYPAEV (SEQ ID NO: 105), LGSYPAEV (SEQ ID NO: 106), QSTNKAEV (SEQ ID NO: 107), HSSYPAEV (SEQ ID NO: 108), SSSYTAEV (SEQ ID NO: 109), TSLYPAEE (SEQ ID NO: 110), ASSYQAEV (SEQ ID NO: 111), QSSYTPSL (SEQ ID NO: 112), QSRYPAEE
- the amino acid sequence of [N1]-[N2] is QSSYPAEV (SEQ ID NO: 96).
- [N1]-[N2]-[N3] comprises SSYPAEVVQ (SEQ ID NO: 121), SKYPAEVVQ (SEQ ID NO: 122), SLYPAEVVQ (SEQ ID NO: 123), SRYPAEVVQ (SEQ ID NO: 124), SSYPPSLVQ (SEQ ID NO: 125), GAYPAEVVQ (SEQ ID NO: 126), GSYPAEVVQ (SEQ ID NO: 127), ASYPAEVVQ (SEQ ID NO: 128), STNKAEVVQ (SEQ ID NO: 129), SSYTAEVVQ (SEQ ID NO: 130), SKYPAEVEQ (SEQ ID NO: 131), SLYPAEEVQ (SEQ ID NO: 132), SSYQAEVVQ (SEQ ID NO: 133),
- [N1]-[N2]-[N3] is or comprises QSSYPAEVVQK (SEQ ID NO: 150), QSKYPAEVVQK (SEQ ID NO: 151), TSLYPAEVVQK (SEQ ID NO: 152), SSSYPAEVVQK (SEQ ID NO: 153), QSRYPAEVVQK (SEQ ID NO: 154), QSSYPPSLVQK (SEQ ID NO: 155), AGAYPAEVVQK (SEQ ID NO: 156), IGSYPAEVVQK (SEQ ID NO: 157), QASYPAEVVQK (SEQ ID NO: 158), ASSYPAEVVQK (SEQ ID NO: 159), LGSYPAEVVQK (SEQ ID NO: 160), QSTNKAEVVQK (SEQ ID NO: 161), HSSYPAEVVQK (SEQ ID NO: 162), SSSYTAEVVQK (SEQ ID NO: 16
- the amino acid sequence of [N1]-[N2]-[N3] is QSSYPAEVVQK (SEQ ID NO: 150).
- [N2]-[N3] is present immediately subsequent to [N1] and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138.
- the peptide comprising the amino acid sequence comprising the formula [N2]-[N3] further comprises [N0], wherein [N0] comprises X A , X B , and X C .
- X A of [N0] is T, I, or N.
- X B of [N0] is N.
- X C of [N0] is N, T, S, or K.
- [N0] comprises TN, IN, NN, NT, NS, or NK.
- [N0] is or comprises TNN, TNT, INN, TNS, NNN, or TNK.
- the amino acid sequence of [N0] is TNN.
- [N0]-[N1] is or comprises TNNQSS (SEQ ID NO: 183), TNNQSK (SEQ ID NO: 184), TNNTSL (SEQ ID NO: 185), TNNSSS (SEQ ID NO: 186), TNNQSR (SEQ ID NO: 187), TNNAGA (SEQ ID NO: 188), TNNIGS (SEQ ID NO: 189), TNNQAS (SEQ ID NO: 190), TNTASS (SEQ ID NO: 191), TNNLGS (SEQ ID NO: 192), TNNQST (SEQ ID NO: 193), TNNHSS (SEQ ID NO: 194), TNNQSK (SEQ ID NO: 184), TNNLSS (SEQ ID NO: 195), INNQSS (SEQ ID NO: 196), TNSQSS (SEQ ID NO: 197), NNNQSR (SEQ ID NO: 198), TNSTSL (SEQ ID NO: 199), TNNQRS
- [N0]-[N1] is TNNQSS (SEQ ID NO: 183).
- [N0]-[N1]-[N2]-[N3] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500), TNNQSKYPAEVVQK (SEQ ID NO: 503), TNNTSLYPAEVVQK (SEQ ID NO: 506), TNNSSSYPAEVVQK (SEQ ID NO: 508), TNNQSRYPAEVVQK (SEQ ID NO: 510), TNNQSSYPPSLVQK (SEQ ID NO: 512), TNNAGAYPAEVVQK (SEQ ID NO: 513), TNNIGSYPAEVVQK (SEQ ID NO: 514), TNNQASYPAEVVQK (SEQ ID NO: 517), TNTASSYPAEVVQK (SEQ ID NO: 520), TNNLGSYPAEVVQK (SEQ ID NO: 500), TNN
- [N0]-[N1]-[N2]-[N3] is TNNQSSYPAEVVQK (SEQ ID NO: 500).
- [N2]-[N3] is present immediately subsequent to [N0]-[N1] and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138.
- the peptide comprising the amino acid sequence comprising the formula [N2]-[N3] further comprises [N4], which comprises X G and X H .
- X G of [N4] is T, P, or N.
- X G of [N4] is T.
- X H of [N4] is A.
- [N4] is or comprises TA, PA, or NA.
- [N4] is TA.
- [N3]-[N4] is or comprises VQKTA (SEQ ID NO: 564), EQKTA (SEQ ID NO: 565), VKKTA (SEQ ID NO: 566), VQKPA (SEQ ID NO: 567), VHKTA (SEQ ID NO: 568), VQQTA (SEQ ID NO: 569), VQKNA (SEQ ID NO: 570), or LQKTA (SEQ ID NO: 571); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, or 4 amino Attorney Docket No.14640.0092-00304 acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the
- [N3]-[N4] is VQKTA (SEQ ID NO: 564).
- [N0]-[N1]-[N2]-[N3]-[N4] is or comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNTSLYPAEVVQKTA (SEQ ID NO: 1232), TNNSSSYPAEVVQKTA (SEQ ID NO: 1539), TNNQSRYPAEVVQKTA (SEQ ID NO: 1327), TNNQSSYPPSLVQKTA (SEQ ID NO: 1300), TNNAGAYPAEVVQKTA (SEQ ID NO: 1021), TNNIGSYPAEVVQKTA (SEQ ID NO: 1112), TNNQASYPAEVVQKTA (SEQ ID NO: 1194), TNTASSYPAEVVQKTA (SEQ ID NO:
- [N0]-[N1]-[N2]-[N3]-[N4] is TNNQSSYPAEVVQKTA (SEQ ID NO: 1533).
- [N2]-[N3] is present immediately subsequent to [N0]-[N1], immediately precedes [N4], and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138.
- a peptide described herein comprises the formula [N2]-[N3], wherein [N2] comprises X 1 , X 2 , X 3 , X 4 , and X 5 and [N3] comprises the amino acid sequence of VQK or VQN.
- [N3] comprises the amino acid sequence VQK.
- X 1 of [N2] is Y or T.
- X 2 of [N2] is Q, T, P, or E.
- X 3 of [N2] is A.
- X 4 of [N2] is E or D.
- X 4 of [N2] is E or D.
- X 5 of [N2] is V or E.
- X 1 of [N2] is Y.
- X 2 of [N2] is P.
- X3 of [N2] is A.
- X4 of [N2] is E.
- X 5 of [N2] is V.
- [N2] comprises YP, YQ, YT, TE, QA, TA, PA, EA, EV, EE, DV, AE, or AD.
- [N2] comprises YPA, YQA, YTA, TEA, QAE, TAE, PAE, EAE, PAD, AEV, AEE, or ADV.
- [N2] comprises YPAE (SEQ ID NO: 21), YQAE (SEQ ID NO: 25), YTAE (SEQ ID NO: 24), TEAE (SEQ ID NO: 587), YPAD (SEQ ID NO: 588), QAEV (SEQ ID NO: 15), TAEV (SEQ ID NO: 16), PAEV (SEQ ID NO: 17), PAEE (SEQ ID NO: 18), EAEV (SEQ ID NO: 590), or PADV (SEQ ID NO: 19).
- [N2] is or comprises YPAEV (SEQ ID NO: 1), YQAEV (SEQ ID NO: 6), YTAEV (SEQ ID NO: 4), YPAEE (SEQ ID NO: 5), TEAEV (SEQ ID NO: 12), or YPADV (SEQ ID NO: 13).
- [N2] is YPAEV (SEQ ID NO: 1).
- [N2]-[N3] comprises AEVVQK (SEQ ID NO: 36), AEEVQK (SEQ ID NO: 39), AEVVQN (SEQ ID NO: 591), or ADVVQK (SEQ ID NO: 593).
- [N2]-[N3] comprises PAEVVQN (SEQ ID NO: 594), QAEVVQK (SEQ ID NO: 52), TAEVVQK (SEQ ID NO: 49), PAEVVQK (SEQ ID NO: 20), PAEEVQK (SEQ ID NO: 51), EAEVVQK (SEQ ID NO: 595), or PADVVQK (SEQ ID NO: 596).
- [N2]-[N3] is or comprises YPAEVVQK (SEQ ID NO: 943), YQAEVVQK (SEQ ID NO: 951), YTAEVVQK (SEQ ID NO: 948), YPAEEVQK (SEQ ID NO: 950), YPAEVVQN (SEQ ID NO: 964), TEAEVVQK (SEQ ID NO: 965), or YPADVVQK (SEQ ID NO: 966); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, or 7 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequence
- [N2]-[N3] is YPAEVVQK (SEQ ID NO: 943).
- [N2]-[N3] (e.g., comprising the amino acid sequence of SEQ ID NO: 943) replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138.
- the peptide comprising the amino acid sequence comprising the formula of [N2]-[N3] further comprises [N1], which comprises X D , X E , and X F .
- X D of [N1] is Q or S.
- X E of [N1] is S, L, A, or T.
- X F of [N1] is S, Y, or T.
- [N1] comprises QS, SL, SA, QT, LS, LY, AT, TS, or SS.
- [N1] is or comprises QSS, SLS, SLY, SAT, or QTS.
- [N1] is QSS.
- [N1]-[N2] comprises SSYPA (SEQ ID NO: 63), LSYQA (SEQ ID NO: 597), LSYTA (SEQ ID NO: 598), LYYPA (SEQ ID NO: 600), ATYPA (SEQ ID NO: 601), LSYPA (SEQ ID NO: 603), or TSTEA (SEQ ID NO: 605).
- [N1]-[N2] comprises SSYPAE (SEQ ID NO: 79), LSYQAE (SEQ ID NO: 607), LSYTAE (SEQ ID NO: 610), LYYPAE (SEQ ID NO: 611), ATYPAE (SEQ ID NO: 613), LSYPAE (SEQ ID NO: 616), TSTEAE (SEQ ID NO: 619), or LSYPAD (SEQ ID NO: 621).
- [N1]-[N2] is or comprises QSSYPAEV (SEQ ID Attorney Docket No.14640.0092-00304 NO: 96), SLSYQAEV (SEQ ID NO: 622), SLSYTAEV (SEQ ID NO: 623), SLYYPAEV (SEQ ID NO: 624), SATYPAEV (SEQ ID NO: 625), SLSYPAEV (SEQ ID NO: 629), SLSYPAEE (SEQ ID NO: 632), QTSTEAEV (SEQ ID NO: 633), or SLSYPADV (SEQ ID NO: 634); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, or 7 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or
- [N1]-[N2] is QSSYPAEV (SEQ ID NO: 96).
- [N1]-[N2]-[N3] is or comprises QSSYPAEVVQK (SEQ ID NO: 150), SLSYQAEVVQK (SEQ ID NO: 635), SLSYTAEVVQK (SEQ ID NO: 637), SLYYPAEVVQK (SEQ ID NO: 639), SATYPAEVVQK (SEQ ID NO: 641), SLSYPAEVVQK (SEQ ID NO: 642), SLSYPAEEVQK (SEQ ID NO: 643), SLSYPAEVVQN (SEQ ID NO: 644), QTSTEAEVVQK (SEQ ID NO: 645), or SLSYPADVVQK (SEQ ID NO: 646); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6,
- [N1]-[N2]-[N3] is QSSYPAEVVQK (SEQ ID NO: 150).
- [N2]-[N3] is present immediately subsequent to [N1] and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138.
- the peptide comprising the amino acid sequence comprising the formula [N2]-[N3] further comprises [N0], wherein [N0] comprises X A , X B , and X C .
- X A of [N0] is T.
- X B of [N0] is N.
- X C of [N0] is N, T, S, or K.
- [N0] comprises TN, NS, NT, NN, or NK.
- [N0] is or comprises TNS, TNT, TNN, or TNK.
- [N0] is TNN.
- [N0]-[N1] is or comprises TNNQSS (SEQ ID NO: 183), TNSSLS (SEQ ID NO: 647), TNSSLY (SEQ ID NO: 648), TNTSAT (SEQ ID NO: 649), TNNQTS (SEQ ID NO: 650), or TNKSAT (SEQ ID NO: 651); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences.
- TNNQSS SEQ ID NO: 183
- TNSSLS SEQ ID NO: 647
- TNSSLY SEQ ID NO: 6408
- [N0]-[N1] is TNNQSS (SEQ ID NO: 183).
- [N0]-[N1]-[N2]-[N3] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500), TNSSLSYQAEVVQK (SEQ ID NO: 652), TNSSLSYTAEVVQK (SEQ ID NO: 654), TNSSLYYPAEVVQK (SEQ ID NO: 655), TNTSATYPAEVVQK (SEQ ID NO: 656), TNSSLSYPAEVVQK (SEQ ID NO: 657), Attorney Docket No.14640.0092-00304 TNSSLSYPAEEVQK (SEQ ID NO: 658), TNSSLSYPAEVVQN (SEQ ID NO: 660), TNNQTSTEAEVVQK (SEQ ID NO: 662), TNKSATYPAEVVQK (SEQ ID NO: 663),
- [N0]-[N1]-[N2]-[N3] is TNNQSSYPAEVVQK (SEQ ID NO: 500).
- [N2]-[N3] is present immediately subsequent to [N0]-[N1] and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138.
- the peptide comprising the amino acid sequence comprising the formula [N2]-[N3] further comprises [N4], which comprises X G and X H .
- X G of [N4] is T, P, or N.
- X H of [N4] is A or D.
- [N4] is or comprises TA, TD, PA, or NA. In some embodiments, [N4] is TA. In some embodiments, [N3]-[N4] is or comprises VQKTA (SEQ ID NO: 564), EQKTA (SEQ ID NO: 565), VKKTA (SEQ ID NO: 566), VQKPA (SEQ ID NO: 567), VHKTA (SEQ ID NO: 568), VQQTA (SEQ ID NO: 569), VQKNA (SEQ ID NO: 570), or LQKTA (SEQ ID NO: 571); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no
- [N3]-[N4] is VQKTA (SEQ ID NO: 564).
- [N0]-[N1]-[N2]-[N3]-[N4] is or comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNSSLSYQAEVVQKTA (SEQ ID NO: 2064), TNSSLSYTAEVVQKTA (SEQ ID NO: 2065), TNSSLYYPAEVVQKTA (SEQ ID NO: 2066), TNTSATYPAEVVQKTA (SEQ ID NO: 2067), TNSSLSYPAEVVQKTA (SEQ ID NO: 2068), TNSSLSYPAEEVQKTA (SEQ ID NO: 2069), TNSSLSYPAEVVQKTD (SEQ ID NO: 2070), TNSSLSYPAEVVQNTA (SEQ ID NO: 2071), TNSSLSYPAEVVQKNA (SEQ ID NO:
- [N0]-[N1]-[N2]-[N3]-[N4] is TNNQSSYPAEVVQKTA (SEQ ID NO: 1533).
- [N2]-[N3] is present immediately Attorney Docket No.14640.0092-00304 subsequent to [N0]-[N1], immediately precedes [N4], and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [0117]
- [N1] is present immediately subsequent to [N0].
- [N2] is present immediately subsequent to [N1].
- [N3] is present immediately subsequent to [N2].
- [N4] is present immediately subsequent to [N3].
- the peptide comprises, from N-terminus to C-terminus, [N2]-[N3]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3]-[N4]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [N0]-[N1]-[N2]-[N3].
- the peptide comprises, from N-terminus to C-terminus, [N0]-[N1]-[N2]-[N3]-[N4].
- a peptide described herein comprises an amino acid sequence having the formula [B]-[C], wherein [B] comprises X 1 , X 2 , and X 3 , and [C] comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943).
- X 1 of [B] is Q, T, S, A, I, L, or H.
- X 1 of [B] is Q, T, S, A, or H.
- X 2 of [B] is S, G, or A. In some embodiments, X 2 of [B] is S or G. In some embodiments, X 3 of [B] is S, K, L, R, or A. In some embodiments, X 3 of [B] is S, K, L, or R. In some embodiments, [B] comprises Q at X 1 . In some embodiments, [B] comprises S at X2. In some embodiments, [B] comprises S at X3. In some embodiments, [B] comprises QS, TS, SS, AG, IG, QA, AS, LG, HS, SK, SL, SR, GA, or GS.
- [B] is or comprises QSS, TSL, SSS, QSR, QSK, AGA, IGS, QAS, ASS, LGS, or HSS.
- the amino acid sequence of [B] is QSS.
- [B]-[C] comprises SSYPAEVVQK (SEQ ID NO: 572), SKYPAEVVQK (SEQ ID NO: 573), SLYPAEVVQK (SEQ ID NO: 574), SRYPAEVVQK (SEQ ID NO: 575), GAYPAEVVQK (SEQ ID NO: 576), GSYPAEVVQK (SEQ ID NO: 580), or ASYPAEVVQK (SEQ ID NO: 582).
- [B]-[C] is or comprises QSSYPAEVVQK (SEQ ID NO: 150), QSKYPAEVVQK (SEQ ID NO: 151), TSLYPAEVVQK (SEQ ID NO: 152), SSSYPAEVVQK (SEQ ID NO: 153), QSRYPAEVVQK (SEQ ID NO: 154), AGAYPAEVVQK (SEQ ID NO: 156), IGSYPAEVVQK (SEQ ID NO: 157), QASYPAEVVQK (SEQ ID NO: 158), ASSYPAEVVQK (SEQ ID NO: 159), LGSYPAEVVQK (SEQ ID NO: 160), or HSSYPAEVVQK (SEQ ID NO: 162); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids) thereof;
- [B]-[C] is QSSYPAEVVQK (SEQ ID NO: 150).
- a peptide comprising the formula [B]-[C] further comprises [A], which comprises X A , X B , and X C .
- X A of [A] is T, I, or N.
- X B of [A] is N.
- X C of [A] is N, T, S, or K.
- [A] comprises TN, IN, NN, NT, NS, or NK.
- [A] is or comprises TNN, TNT, INN, NNN, TNS, or TNK.
- [A] is TNN.
- [A]-[B] is or comprises TNNQSS (SEQ ID NO: 183), TNNQSK (SEQ ID NO: 184), TNNTSL (SEQ ID NO: 185), TNNSSS (SEQ ID NO: 186), TNNQSR (SEQ ID NO: 187), TNNAGA (SEQ ID NO: 188), TNNIGS (SEQ ID NO: 189), TNNQAS (SEQ ID NO: 190), TNTASS (SEQ ID NO: 191), TNNLGS (SEQ ID NO: 192), TNNHSS (SEQ ID NO: 194), INNQSS (SEQ ID NO: 196), NNNQSR (SEQ ID NO: 198), TNSTSL (SEQ ID NO: 199), or TNKQAS (SEQ ID NO: 201); an amino acid sequence comprising any portion of any one of the aforesaid
- the amino acid sequence of [A]-[B] is TNNQSS (SEQ ID NO: 183).
- [A]-[B]-[C] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500), TNNQSKYPAEVVQK (SEQ ID NO: 503), TNNTSLYPAEVVQK (SEQ ID NO: 506), TNNSSSYPAEVVQK (SEQ ID NO: 508), TNNQSRYPAEVVQK (SEQ ID NO: 510), TNNAGAYPAEVVQK (SEQ ID NO: 513), TNNIGSYPAEVVQK (SEQ ID NO: 514), TNNQASYPAEVVQK (SEQ ID NO: 517), TNTASSYPAEVVQK (SEQ ID NO: 520), TNNLGSYPAEVVQK (SEQ ID NO: 523), TNNHSSYPAEVVQK (SEQ ID NO: 525
- [A]-[B] is TNNQSS (SEQ ID NO: 183). In some embodiments, [A]-[B]-[C] is TNNQSSYPAEVVQK (SEQ ID NO: 500).
- a peptide comprising the formula [B]-[C] further comprises [D], wherein [D] comprises X 4 and X 5 .
- X 4 of [D] is T or N.
- X 5 of [D] is A.
- [D] is or comprises TA or PA.
- the amino acid sequence of [D] is TA.
- [C]-[D] is or comprises YPAEVVQKTA (SEQ ID NO: 584) or YPAEVVQKPA (SEQ ID NO: 586); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences.
- amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the a
- the amino acid sequence of [C]-[D] is YPAEVVQKTA (SEQ ID NO: 584).
- [A]-[B]-[C]-[D] is or comprises Attorney Docket No.14640.0092-00304 TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNTSLYPAEVVQKTA (SEQ ID NO: 1232), TNNSSSYPAEVVQKTA (SEQ ID NO: 1539), TNNQSRYPAEVVQKTA (SEQ ID NO: 1327), TNNAGAYPAEVVQKTA (SEQ ID NO: 1021), TNNIGSYPAEVVQKTA (SEQ ID NO: 1112), TNNQASYPAEVVQKTA (SEQ ID NO: 1194), TNTASSYPAEVVQKTA (SEQ ID NO: 1575), TNNLGSYPA
- [C]-[D] is YPAEVVQKTA (SEQ ID NO: 584).
- [A]-[B]-[C]-[D] is TNNQSSYPAEVVQKTA (SEQ ID NO: 1533).
- a peptide described herein comprises an amino acid sequence having the formula [B]-[C], wherein [B] comprises X1, X2, and X3, and [C] comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943).
- X 1 of [B] is Q or S.
- X 2 of [B] is S, L, or A.
- X 3 of [B] is S, Y, or T.
- [B] comprises Q at X1.
- [B] comprises S at X2.
- [B] comprises S at X3.
- [B] comprises QS, SL, SA, LY, AT, LS, or SS.
- [B] is or comprises QSS, SLY, SAT, or SLS.
- [B] is QSS.
- [B]-[C] comprises SSYPAEVVQK (SEQ ID NO: 572), LYYPAEVVQK (SEQ ID NO: 702), ATYPAEVVQK (SEQ ID NO: 718), or LSYPAEVVQK (SEQ ID NO: 703).
- [B]-[C] is or comprises QSSYPAEVVQK (SEQ ID NO: 150), SLYYPAEVVQK (SEQ ID NO: 639), SATYPAEVVQK (SEQ ID NO: 641), or SLSYPAEVVQK (SEQ ID NO: 642); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences.
- QSSYPAEVVQK SEQ ID NO: 150
- SLYYPAEVVQK SEQ ID NO: 639
- SATYPAEVVQK SEQ ID NO: 64
- [B]-[C] is QSSYPAEVVQK (SEQ ID NO: 150).
- a peptide comprising the formula [B]-[C] further comprises [A], which comprises XA, XB, and XC.
- XA of [A] is T.
- XB of [A] is N.
- X C of [A] is N, T, S, or K.
- [A] comprises TN, NS, NT, NK, or NN.
- [A] is or comprises TNN, TNS, TNT, or TNK.
- the amino acid sequence of [A] is TNN.
- [A]-[B] is or comprises Attorney Docket No.14640.0092-00304 TNNQSS (SEQ ID NO: 183), TNSSLY (SEQ ID NO: 648), TNTSAT (SEQ ID NO: 649), TNSSLS (SEQ ID NO: 647), or TNKSAT (SEQ ID NO: 651); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences.
- [A]-[B] is TNNQSS (SEQ ID NO: 183).
- [A]-[B]-[C] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500), TNSSLYYPAEVVQK (SEQ ID NO: 655), TNTSATYPAEVVQK (SEQ ID NO: 656), TNSSLSYPAEVVQK (SEQ ID NO: 657), or TNKSATYPAEVVQK (SEQ ID NO: 663); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any
- [A]-[B] is TNNQSS (SEQ ID NO: 183). In some embodiments, [A]-[B]-[C] is TNNQSSYPAEVVQK (SEQ ID NO: 500).
- a peptide comprising the formula [B]-[C] further comprises [D], wherein [D] comprises X 4 and X 5 .
- X 4 of [D] is T, N, or P.
- X5 of [D] is A or D.
- [D] is or comprises TA, TD, NA, or PA.
- the amino acid sequence of [D] is TA.
- [C]-[D] is or comprises YPAEVVQKTA (SEQ ID NO: 584), YPAEVVQKTD (SEQ ID NO: 719), YPAEVVQKNA (SEQ ID NO: 724), or YPAEVVQKPA (SEQ ID NO: 586); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences.
- amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g.
- [C]-[D] is YPAEVVQKTA (SEQ ID NO: 584).
- [A]-[B]-[C]-[D] is or comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNSSLYYPAEVVQKTA (SEQ ID NO: 2066), TNTSATYPAEVVQKTA (SEQ ID NO: 2067), TNSSLSYPAEVVQKTA (SEQ ID NO: 2068), TNSSLSYPAEVVQKTD (SEQ ID NO: 2070), TNSSLSYPAEVVQKNA (SEQ ID NO: 2072), TNSSLSYPAEVVQKPA (SEQ ID NO: 2073), or TNKSATYPAEVVQKTA (SEQ ID NO: 2075); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 11, 12, 13, 14, or
- [A]-[B]-[C]-[D] is TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). [0124] In some embodiments, [B] is present immediately subsequent to [A]. In some embodiments, [C] is present immediately subsequent to [B]. In some embodiments, [D] is present immediately subsequent to [C]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [B]- [C]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [A]-[B]-[C].
- the peptide comprises, from N-terminus to C-terminus, [B]-[C]-[D]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [A]-[B]-[C]-[D].
- a peptide described herein comprises the formula [K1]-[K2], wherein [K1] comprises LSY or LYY, and [K2] comprises X 1 , X 2 , X 3 , and X 4 . In some embodiments, [K1] comprises LSY. In some embodiments, X 1 of [K2] is Q, T or P.
- X 2 of [K2] is A, in some embodiments, X 3 of [K2] is E or D. In some embodiments, X 4 of [K2] is V or E. In some embodiments, [K2] comprises QA, TA, PA, EV, EE, DV, AE, or AD. In some embodiments, [K2] comprises QAE, TAE, PAE, PAD, AEV, AEE, or ADV. In some embodiments, [K2] is or comprises QAEV (SEQ ID NO: 15), TAEV (SEQ ID NO: 16), PAEV (SEQ ID NO: 17), PAEE (SEQ ID NO: 18), or PADV (SEQ ID NO: 19).
- [K1]-[K2] comprises LSYQA (SEQ ID NO: 597), LSYTA (SEQ ID NO: 598), LYYPA (SEQ ID NO: 600), or LSYPA (SEQ ID NO: 603).
- [K1]-[K2] comprises LSYQAE (SEQ ID NO: 607), LSYTAE (SEQ ID NO: 610), LYYPAE (SEQ ID NO: 611), LSYPAE (SEQ ID NO: 616), or LSYPAD (SEQ ID NO: 621).
- [K1]-[K2] is or comprises LSYQAEV (SEQ ID NO: 667), LSYTAEV (SEQ ID NO: 668), LYYPAEV (SEQ ID NO: 669), LSYPAEV (SEQ ID NO: 671), LSYPAEE (SEQ ID NO: 673), or LSYPADV (SEQ ID NO: 674); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences.
- LSYQAEV SEQ ID NO: 667
- LSYTAEV SEQ ID NO: 668
- the peptide comprising the amino acid sequence comprising the formula of [K1]-[K2] further comprises [K0], which comprises TNNS (SEQ ID NO: 14).
- [K0]-[K1] comprises TNSSLS (SEQ ID NO: 647) or TNSSLY (SEQ ID NO: 648).
- [K0]-[K1] is or comprises TNSSLSY (SEQ ID NO: 676) or TNSSLYY (SEQ ID NO: 678); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences.
- TNSSLSY SEQ ID NO: 676
- TNSSLYY SEQ ID NO: 678
- an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one
- [K0]-[K1]-[K2] comprises TNSSLSYQA (SEQ ID NO: 679), TNSSLSYTA (SEQ ID NO: 681), TNSSLYYPA (SEQ ID NO: 682), or TNSSLSYPA (SEQ ID NO: 683).
- [K0]-[K1]-[K2] comprises TNSSLSYQAE (SEQ ID NO: 684), TNSSLSYTAE (SEQ ID NO: 685), TNSSLYYPAE (SEQ ID NO: 686), TNSSLSYPAE (SEQ ID NO: 687), or TNSSLSYPAD (SEQ ID NO: 689).
- [K0]-[K1]-[K2] is or comprises TNSSLSYQAEV (SEQ ID NO: 692), TNSSLSYTAEV (SEQ ID NO: 693), TNSSLYYPAEV (SEQ ID NO: 696), TNSSLSYPAEV (SEQ ID NO: 697), TNSSLSYPAEE (SEQ ID NO: 698), or TNSSLSYPADV (SEQ ID NO: 699); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences.
- amino acid sequence comprising any portion of any one
- peptide comprising the amino acid sequence comprising the formula of [K1]-[K2] further comprises [K3], wherein [K3] comprises X A , X B , and X C .
- X A of [K3] is V.
- X B of [K3] is Q.
- X C of [K3] is K or N.
- [K3] comprises VQ, QK, or QN.
- [K3] is or comprises VQK or VQN.
- K2]-[K3] is or comprises QAEVVQK (SEQ ID NO: 52), TAEVVQK (SEQ ID NO: 49), PAEVVQK (SEQ ID NO: 20), PAEEVQK (SEQ ID NO: 51) ,PAEVVQN (SEQ ID NO: 594), or PADVVQK (SEQ ID NO: 596); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences.
- amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4,
- [K1]-[K2]-[K3] is or comprises LSYQAEVVQK (SEQ ID NO: 700), LSYTAEVVQK (SEQ ID NO: 701), LYYPAEVVQK (SEQ ID NO: 702), LSYPAEVVQK (SEQ ID NO: 703), LSYPAEEVQK (SEQ ID NO: 704), LSYPAEVVQN (SEQ ID NO: 706), or LSYPADVVQK (SEQ ID NO: 708); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one
- [K0]-[K1]-[K2]-[K3] is or comprises TNSSLSYQAEVVQK (SEQ ID NO: 652), TNSSLSYTAEVVQK (SEQ ID NO: 654), TNSSLYYPAEVVQK (SEQ ID NO: 655), TNSSLSYPAEVVQK (SEQ ID NO: 657), TNSSLSYPAEEVQK (SEQ ID NO: 658), TNSSLSYPAEVVQN (SEQ ID NO: 660), or TNSSLSYPADVVQK (SEQ ID NO: 665); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence Attorney Docket No.14640.0092-00304 comprising one, two, or three but no more than four modifications relative to any one of
- the peptide comprising the amino acid sequence comprising the formula of [K1]-[K2] further comprises [K4], wherein [K4] comprises X D and X E .
- [K4] comprises XD of [K4] is T, P, or N.
- XE of [K4] is A or D.
- [K4] is or comprises TA, TD, PA, or NA.
- [K3]-[K4] is or comprises VQKTA (SEQ ID NO: 564), VQKTD (SEQ ID NO: 714), VQNTA (SEQ ID NO: 715), VQKNA (SEQ ID NO: 570), or VQKPA (SEQ ID NO: 567); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences.
- VQKTA SEQ ID NO: 564
- VQKTD SEQ ID NO: 714
- VQNTA SEQ ID NO: 715
- VQKNA SEQ ID NO: 570
- [N0]-[N1]-[N2]-[N3]-[N4] is or comprises TNSSLSYQAEVVQKTA (SEQ ID NO: 2064), TNSSLSYTAEVVQKTA (SEQ ID NO: 2065), TNSSLYYPAEVVQKTA (SEQ ID NO: 2066), TNSSLSYPAEVVQKTA (SEQ ID NO: 2068), TNSSLSYPAEEVQKTA (SEQ ID NO: 2069), TNSSLSYPAEVVQKTD (SEQ ID NO: 2070), TNSSLSYPAEVVQNTA (SEQ ID NO: 2071), TNSSLSYPAEVVQKNA (SEQ ID NO: 2072), TNSSLSYPAEVVQKPA (SEQ ID NO: 2073), or TNSSLSYPADVVQKTA (SEQ ID NO: 2076); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequence
- [K2] is present immediately subsequent to [K1]. In some embodiments, [K1] is present immediately subsequent to [K0]. In some embodiments, [K3] is present immediately subsequent to [K2]. In some embodiments, [K4] is present immediately subsequent to [K3]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [K1]-[K2]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [K1]-[K2]-[K3].
- the peptide comprises, from N-terminus to C-terminus, [K0]-[K1]-[K2]-[K3]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [K1]-[K2]-[K3]-[K4]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [K0]-[K1]-[K2]-[K3]-[K4].
- a peptide described herein comprises an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of the sequences provided in Table 1, 2A, 2B, 14, or 23-28.
- the peptide comprises an amino acid Attorney Docket No.14640.0092-00304 sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583- 1587, 1590, 1591-1593, 1598-1608, and 1610-1624.
- the peptide comprises an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of SEQ ID NOs: 943 or 2064-2080. In some embodiments, the peptide comprises at least 3, at least 4, at least 5, at least 6, or at least 7 consecutive amino acids from SEQ ID NOs: 943 or any one of SEQ ID NOs: 946-966. [0131] In some embodiments, the at least 3 consecutive amino acids comprise YPA. In some embodiments, the at least 4 consecutive amino acids comprise YPAE (SEQ ID NO: 21).
- the at least 5 consecutive amino acids comprise YPAEV (SEQ ID NO: 1). In some embodiments, the at least 6 consecutive amino acids comprise YPAEVV (SEQ ID NO: 725). In some embodiments, the at least 7 consecutive amino acids comprise YPAEVVQ (SEQ ID NO: 726). In some embodiments, the amino acid sequence comprises YPAEVVQK (SEQ ID NO: 943). In some embodiments, the amino acid sequence consists of YPAEVVQK (SEQ ID NO: 943).
- a peptide described herein comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, 14, or 23-28.
- the peptide comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, 14, or 23-28.
- the peptide comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583- 1587, 1590, 1591-1593, 1598-1608, and 1610-1624.
- the peptide comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583- 1587, 1590, 1591-1593, 1598-1608, and 1610-1624.
- the peptide comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 943 and 2064-2080.
- the peptide comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080. [0133] In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or Attorney Docket No.14640.0092-00304 three but no more than four different amino acids relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943).
- the peptide comprises an amino sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063. In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063.
- the different amino acids of the amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063 are present at one or more of the following positions: (i) position 1, wherein the different amino acid is T or L; (ii) position 2, wherein the different amino acid is N, L, K, A, T, or P; (iii) position 3, wherein the different amino acid is N, K, L, A, Y, or S; (iv) position 4, wherein the different amino acid is Q, L, T, S, F, Y, K, or A; (v) position 5, wherein the different amino acid is S, H, A, M, Q, T, V, or F; (vi) position 6, wherein the different amino acid is S, P, V, A, Q, L, T, N, or M; (vii) position 7, wherein the different amino acid is Y, H, S, V, A, L, or T; (viii) position
- the peptide comprises an amino sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813- 1815, 1818, 1822, 1823, 1829-1837, 1842-2023, and 2087-2112.
- the peptide comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842-2023, and 2087-2112.
- the different amino acids of the amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798- 1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842-2023, and 2087-2112, are present at one or more of the following positions: (i) position 1, wherein the different amino acid is T, G, N, S, E, L, Y, V, or I; (ii) position 2, wherein the different amino acid is D, N, K, E, V, G, R, L, H, F, P, T, A, S, I, or Y; (iii) position 3, wherein the different amino acid is Y, N, K, T, W, Q, M, V, C, A, L, F, H, G, R, S, or P;
- the peptide comprises the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, 14, or 23-28. In some embodiments, the peptide comprises the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583- 1587, 1590, 1591-1593, 1598-1608, and 1610-1624. In some embodiments, the peptide comprises the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080.
- the peptide comprises the amino acid sequence of any one of SEQ ID NOs: 2024-2063. In some embodiments, the peptide comprises the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842- 2023, and 2087-2112. In some embodiments, the peptide comprises the amino acid sequence of SEQ ID NO: 943.
- the peptide comprises an amino acid sequence encoded by a nucleotide sequence described herein, e.g., a nucleotide sequence of Table 2A. In some embodiments, the peptide comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 944.
- the peptide comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 944.
- the peptide comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 944, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto.
- the nucleotide sequence encoding a peptide described herein comprises a nucleotide sequence described herein, e.g., as described in Table 2A.
- the nucleotide sequence encoding a peptide described herein is codon optimized.
- Attorney Docket No.14640.0092-00304 the nucleotide sequence encoding a peptide described herein is isolated.
- the nucleotide sequence encoding a peptide described herein is recombinant.
- the nucleotide sequence encoding a peptide described herein comprises the nucleotide sequence of SEQ ID NO: 944, or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 944.
- the nucleotide sequence encoding a peptide described herein comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 944.
- the nucleic acid encoding a peptide described herein comprises a nucleotide sequence comprising the nucleotide sequence of SEQ ID NO: 944, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto.
- the present disclosure also provides a nucleic acid or polynucleotide encoding any of the peptides described herein and AAV capsid variants, AAV particles, vectors, and cells comprising the same.
- an AAV capsid variant described herein comprises an amino acid other than T corresponding to position 577 (e.g., Y, N, or C) of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant comprises Y corresponding to position 577 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant comprises N corresponding to position 577 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant comprises C corresponding to position 577 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant (e.g., an AAV5 capsid variant) comprises more than one amino acid that replaces the threonine (T) at position 577, numbered according to SEQ ID NO: 138 (e.g., that replaces a T corresponding to T577 of the amino acid sequence of SEQ ID NO: 138).
- an insert of two, three, four, five, six, seven, eight, nine, or ten amino acids replaces the T at position 577, numbered according to SEQ ID NO: 138.
- an insert of eight amino acids replaces the T at position 577, numbered according to SEQ ID NO: 138.
- an AAV particle described herein comprises an AAV capsid variant comprising a peptide or an amino acid sequence described herein.
- the AAV capsid variant comprises a peptide disclosed herein.
- the peptide is present in VP1, VP2, and/or VP3 proteins of the AAV capsid variant.
- the peptide is present in VP1, VP2, and VP3 proteins of the AAV capsid variant.
- the AAV capsid variant comprises a peptide comprising the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, Attorney Docket No.14640.0092-00304 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583- 1587, 1590, 1591-1593, 1598- 1608, and 1610-1624.
- the AAV capsid variant comprises a peptide comprising the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080.
- the AAV capsid variant comprises a peptide comprising the amino acid sequence of any one of SEQ ID NOs: 2024-2063. In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842- 2023, and 2087-2112. In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of SEQ ID NO: 943. In some embodiments, the peptide is present in loop VIII.
- the peptide is present immediately subsequent to an amino acid corresponding to position 570 of the amino acid sequence of SEQ ID NO: 138.
- the peptide replaces amino acids corresponding to positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138.
- the peptide is present immediately subsequent to an amino acid corresponding to position 570, and the peptide replaces amino acids corresponding to positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 571 of the amino acid sequence of SEQ ID NO: 138.
- amino acids corresponding to positions 571-579 e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and T579
- the peptide replaces amino acids corresponding to positions 572-579 (e.g., N572, N573, Q574, S575, S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138.
- the peptide is present immediately subsequent to an amino acid corresponding to position 571, and the peptide replaces amino acids corresponding to positions 572-579 (e.g., N572, N573, Q574, S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138.
- the peptide is present immediately subsequent to an amino acid corresponding to position 572 of the amino acid sequence of SEQ ID NO: 138.
- the peptide replaces amino acids corresponding to positions 573-579 (e.g., N573, Q574, S575, S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 572, and the peptide replaces amino acids corresponding to positions 573-579 (e.g., N573, Q574, S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 573 of the amino acid sequence of SEQ ID NO: 138.
- amino acids corresponding to positions 573-579 e.g., N573, Q574, S575, S576, T577, T578, and T579
- the peptide replaces amino acids corresponding to positions 574-579 (e.g., Q574, S575, S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 573, and the peptide replaces amino acids corresponding to positions 574-579 (e.g., Q574, S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 574 of the amino acid sequence of SEQ ID NO: 138.
- amino acids corresponding to positions 574-579 e.g., Q574, S575, S576, T577, T578, and T579
- Attorney Docket No.14640.0092-00304 the peptide replaces amino acids corresponding to positions 575-579 (e.g., S575, S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138.
- the peptide is present immediately subsequent to an amino acid corresponding to position 574, and the peptide replaces amino acids corresponding to positions 575-579 (e.g., S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138.
- the peptide is present immediately subsequent to an amino acid corresponding to position 575 of the amino acid sequence of SEQ ID NO: 138.
- the peptide replaces amino acids corresponding to positions 576-579 (e.g., S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 575, and the peptide replaces amino acids corresponding to positions 576-579 (e.g., S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 138.
- the peptide replaces an amino acid corresponding to position 577 (e.g., T577) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the peptide replaces amino acids corresponding to positions 577-579 (e.g., T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 576, and the peptide replaces amino acids corresponding to position 577 (e.g., T577), of the amino acid sequence of SEQ ID NO 138.
- the peptide is present immediately subsequent to an amino acid corresponding to position 576, and the peptide replaces amino acids corresponding to positions 577-579 (e.g., T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138.
- the AAV capsid variant comprises a peptide comprising the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 946-966, wherein the peptide replaces an amino acid corresponding to position 577 (e.g., T577) of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant comprises a peptide comprising the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 946-966, wherein the peptide is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant comprises a peptide comprising the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 946-966, wherein the peptide is present immediately subsequent to an amino acid corresponding to position 576, and wherein the peptide replaces an amino acid corresponding to position 577 (e.g., T577), of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant (e.g., an AAV capsid variant described herein), comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 944 or a nucleotide sequence having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity thereto.
- the AAV capsid variant described herein comprises an amino acid sequence encoded by Attorney Docket No.14640.0092-00304 the nucleotide sequence of SEQ ID NO: 944 or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 944.
- the AAV capsid variant comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten, different nucleotides relative to the nucleotide sequence of SEQ ID NO: 944.
- the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 944, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto.
- the nucleic acid sequence encoding the AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 944.
- the nucleotide sequence encoding an AAV capsid variant described herein comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten, different nucleotides relative to the nucleotide sequence of SEQ ID NO: 944.
- an AAV capsid variant described herein comprises a peptide comprising the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the peptide is present in loop VIII.
- the peptide is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant.
- the peptide is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant.
- an AAV capsid variant described herein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the amino acid sequence is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 138.
- an AAV capsid variant described herein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) replaces an amino acid corresponding to position 577 (e.g., T577) of the amino acid sequence of SEQ ID NO: 138.
- an AAV capsid variant described herein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 138, and wherein the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) replaces an amino acid corresponding to position 577 (e.g., T577) of the amino acid sequence of SEQ ID NO: 138.
- an AAV capsid variant described herein comprises the amino acid Y at a position corresponding to amino acid 577 of the amino acid sequence of SEQ ID NO: 138, and further comprises the amino acid sequence of PAEVVQK (SEQ ID NO: 20), which is present immediately Attorney Docket No.14640.0092-00304 subsequent to a position corresponding to amino acid 577 of the amino acid sequence of SEQ ID NO: 138.
- an AAV capsid variant described herein comprises the amino acid Y at a position corresponding to amino acid 577 of the amino acid sequence of SEQ ID NO: 982, and the amino acid sequence of PAEVVQK (SEQ ID NO: 20) at positions corresponding to amino acids 578- 584, of the amino acid sequence of SEQ ID NO: 982.
- an AAV capsid variant described herein comprises the amino acid sequence of TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), which is present in loop VIII.
- the AAV capsid variant comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), which is present immediately subsequent to an amino acid corresponding to position 570 of the amino acid sequence of SEQ ID NO: 138, wherein YPAEVVQK (SEQ ID NO: 943) replaces an amino acid corresponding to position 577 (e.g., replaces T577) of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant comprises: (a) a VP1 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 982; (b) a VP2 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 738; and/or (c) a VP3 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises a peptide comprising the
- the AAV capsid variant comprises: (a) a VP1 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to SEQ ID NO: 982; (b) a VP2 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 738; and/or (c) a VP3 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises a peptide comprising the amino acid sequence of
- the AAV capsid variant comprises: (a) a VP1 protein comprising an amino acid sequence that is at least 98% identical, e.g., at least 98%, at least 99%, or 100% identical, to SEQ ID NO: 982; (b) a VP2 protein comprising an amino acid sequence that is at least 98% identical, e.g., at least 98%, at least 99%, or 100% identical, to SEQ ID NO: 738; and/or (c) a VP3 protein comprising an amino acid sequence that is at least 98% identical, e.g., at least 98%, at least 99%, or 100% identical, to SEQ ID NO: 739, wherein the AAV capsid variant comprises a peptide comprising the Attorney Docket No.14640.0092-00304 amino acid sequence of YPAEVVQK (SEQ ID NO: 943) or a peptide comprising one, two, or three—but no more than three—substitutions relative to the
- the AAV capsid variant comprises: (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982; (b) a VP2 protein comprising the amino acid sequence of SEQ ID NO: 738; and/or (c) a VP3 protein comprising the amino acid sequence of SEQ ID NO: 739.
- the AAV capsid variant is encoded by the nucleotide sequence of SEQ ID NO: 984 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 984.
- the present disclosure provides an AAV particle comprising an AAV capsid variant and a viral genome, wherein the viral genome comprises a frataxin (FXN)-encoding sequence and the AAV capsid variant comprises an amino acid sequence having the formula [N2]-[N3], wherein: (i) [N2] comprises X 1 , X 2 , X 3 , X 4 , and X 5 , wherein: (a) X 1 is Y, N, or C; (b) X 2 is P, K, T, or Q; (c) X 3 is A or P; (d) X 4 is E, S, or A; and (e) X 5 is V, L, or E; and (ii) [N3] comprises the amino acid sequence VQK, EQK, VKK, VHK, VQQ, or LQK; and wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 739, or an amino acid sequence at
- the amino acid sequence [N2]-[N3] is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant. In some embodiments, the amino acid sequence [N2]-[N3] is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant. [0158] In some embodiments, [N2]-[N3] is present in loop VIII of the AAV capsid variant, wherein loop VIII comprises positions 571-599 numbered according to the amino acid sequence of SEQ ID NO: 982.
- [N2]-[N3] is present in loop VIII of the AAV capsid variant, wherein loop VIII is present at amino acids comprising those corresponding to positions 571-599 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, [N2]-[N3] is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, the capsid variant is an AAV5 variant comprising [N2]-[N3] in place of an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138.
- [N2] comprises the amino acid sequence YP, YPA, YPAE (SEQ ID NO: 21), or YPAEV (SEQ ID NO: 1).
- the amino acid sequence is VQK.
- [N2]-[N3] comprises the amino acid sequence AEVVQK (SEQ ID NO: 36) or PAEVVQK (SEQ ID NO: 20).
- [N2]-[N3] comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943).
- [N2]-[N3] is present in loop VIII of a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant. In some embodiments, [N2]-[N3] is present in loop VIII of the VP1, VP2, and VP3 proteins of the AAV capsid variant.
- the AAV capsid variant further comprises [N1] comprising X D , X E , and X F , wherein: (a) X D is Q, T, S, A, I, L, or H; (b) X E is S, G, A, or R; and (c) X F is S, K, L, R, A, or T; Attorney Docket No.14640.0092-00304 and wherein [N1] immediately precedes [N2]-[N3].
- [N1] comprises the amino acid sequence QS, SS, or QSS.
- [N1]-[N2] comprises the amino acid sequence QSSYPAEV (SEQ ID NO: 96).
- [N1]-[N2]-[N3] comprises the amino acid sequence SSYPAEVVQ (SEQ ID NO: 121) or QSSYPAEVVQK (SEQ ID NO: 150).
- [N1]-[N2]-[N3] is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant.
- [N1]-[N2]-[N3] is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant.
- the AAV capsid variant further comprises [N0] comprising X A , X B , and X C , wherein: (a) X A is T, I, or N; (b) X B is N; and (c) X C is N, T, S, or K; and wherein [N0] immediately precedes [N1].
- [N0] comprises the amino acid sequence TN, NN, or TNN.
- [N0]-[N1] comprises the amino acid sequence TNNQSS (SEQ ID NO: 183).
- [N0]-[N1]-[N2]-[N3] comprises the amino acid sequence TNNQSSYPAEVVQK (SEQ ID NO: 500).
- [N0]-[N1]-[N2]-[N3] is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant.
- [N0]-[N1]-[N2]-[N3] is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant.
- the AAV capsid variant further comprises [N4] comprising X G and XH, wherein: (a) XG is T, P, or N; and (b) XH is A; and wherein [N4] is present immediately subsequent to [N3].
- [N4] comprises the amino acid sequence TA.
- [N4] is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant.
- [N4] is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant.
- the AAV capsid variant comprises [N2]-[N3]-[N4], wherein each of [N2], [N3] and [N4] are as described herein. In some embodiments, the AAV capsid variant comprises [N1]-[N2]-[N3]-[N4], wherein each of [N1], [N2], [N3] and [N4] are as described herein. In some embodiments, the AAV capsid variant comprises [N0]-[N1]-[N2]-[N3]-[N4], wherein each of [N0], [N1], [N2], [N3] and [N4] are as described herein.
- [N0]-[N1]-[N2]-[N3]-[N4] comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533).
- the AAV capsid variant is an AAV5 capsid variant that comprises SEQ ID NO: 1533.
- [N0]-[N1]-[N2]-[N3]-[N4] is present in loop VIII, wherein loop VIII comprises positions 571-599 numbered according to the amino acid sequence of SEQ ID NO: 982.
- [N0]-[N1]-[N2]-[N3]-[N4] is present in loop VIII, wherein loop VIII is present at amino acids comprising those corresponding to positions 571-599 of the amino acid sequence of SEQ ID NO: 982.
- the present disclosure provides an AAV particle comprising: (i) a viral genome comprising a frataxin (FXN)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% identical to SEQ ID NO: 739, wherein the AAV capsid variant comprises at least 4, at least 5, at least 6, at least 7, or all 8 consecutive amino acids from the amino acid sequence of YPAEVVQK (SEQ ID NO: 943).
- the at least 4 consecutive amino acids comprise YPAE (SEQ ID NO: 21).
- YPAE (SEQ ID NO: 21) is present at amino acids corresponding to positions 577-580 of the amino acid sequence of SEQ ID NO: 982.
- the at least 5 consecutive amino acids comprise YPAEV (SEQ ID NO: 1).
- YPAEV (SEQ ID NO: 1) is present at amino acids corresponding to positions 577-581 of the amino acid sequence of SEQ ID NO: 982.
- the at least 6 consecutive amino acids comprise YPAEVV (SEQ ID NO: 725).
- YPAEVV (SEQ ID NO: 725) is present at amino acids corresponding to positions 577-582 of the amino acid sequence of SEQ ID NO: 982.
- the at least 7 consecutive amino acids comprise YPAEVVQ (SEQ ID NO: 726).
- YPAEVVQ (SEQ ID NO: 726) is present at amino acids corresponding to positions 577-583 of the amino acid sequence of SEQ ID NO: 982.
- the AAV capsid variant comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) present at amino acids corresponding to positions 577-584 of the amino acid sequence of SEQ ID NO: 982.
- the present disclosure provides an AAV particle comprising: (i) a viral genome comprising a frataxin (FXN)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to the amino acid sequence of SEQ ID NO: 982, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943) at amino acids corresponding to positions 577-584 of the amino acid sequence of SEQ ID NO: 982.
- FXN frataxin
- the AAV capsid variant comprises (i) a VP1 protein comprising an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity) to SEQ ID NO: 982, wherein the VP1 protein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943); (ii) a VP2 protein comprising an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity) to SEQ ID NO: 738, wherein the VP2 protein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943); and/or (iii) a VP3 protein comprising an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least
- the AAV capsid variant comprises (i) a VP1 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO: 982, wherein the VP1 protein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943); (ii) a VP2 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO: 738, wherein the VP2 protein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943); and/or (iii) a VP3 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO: 739, wherein the VP3 protein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943).
- the AAV capsid variant comprises (i) a VP1 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 982; (ii) a VP2 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 738; and/or (iii) a VP3 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 739.
- the AAV capsid variant further comprises one, two, three, or all of (i) an amino acid other than Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138 (e.g., T, S, A, I, L, or H at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138), (ii) an amino acid other than S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138 (e.g., G, A, L, T, or R at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138), and/or (iii) an amino acid other than S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138 (e.g., K, L, R, A, Y, or T at a position corresponding to amino acid 576 of the amino acid sequence of the amino acid sequence of SEQ ID
- the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a T present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an L present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an S present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an R present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises Q present at a position corresponding to amino acid 574 ofthe amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of Attorney Docket No.14640.0092-00304 the amino acid sequence of SEQ ID NO: 138, and/or a K present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an A present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, a G present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an A present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an I present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, a G present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an A present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an A present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an L present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, a G present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or a T present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an H present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an L present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of to the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an R present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an S present at a position Attorney Docket No.14640.0092-00304 corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an L present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an S present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an L present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or a Y present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an S present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an A present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or a T present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, a T present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant comprises an amino acid other than Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant comprises S present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an amino acid other than T present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138 and/or an amino acid other than N present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises I or N present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138 and/or T, S, or K present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an R present at a position corresponding to amino acid 456 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a T present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid sequence of SEQ ID NO: 138, and/or an N present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a T present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid sequence of SEQ ID NO: 138, and/or a T present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an I present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid Attorney Docket No.14640.0092-00304 sequence of SEQ ID NO: 138, and/or an N present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a T present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an N present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid sequence of SEQ ID NO: 138, and/or an N present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a T present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid sequence of SEQ ID NO: 138, and/or a K present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an amino acid other than T present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a P or N present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an amino acid other than T present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or an amino acid other than A present at a position corresponding to amino acid 589 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a P or N present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or a D present at a position corresponding to amino acid 589 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a T present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or an A present at a position corresponding to amino acid 579 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a P present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or an A present at a position corresponding to amino acid 579 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an N present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or an A present at a position corresponding to amino acid 579 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a T present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or a D present at a position corresponding to amino acid 579 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an amino acid other than T present at a position corresponding to amino acid 577 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant comprises a Y present at a position corresponding to amino acid 577 of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises a modification in loop I, II, IV, and/or VI.
- loop I, II, IV, VI, and VIII can be identified as described in Govindasamy et al. Structurally Mapping the Diverse Phenotype of Adeno-Associated Virus Serotype 4. Journal of Virology.2006 Dec.80(23):11556-11570; and Govindasamy et al. Structural Insights into Adeno-Associated Virus Serotype 5. Journal of Virology.2013 Oct.87(20):11187-11199; the contents of which are each hereby incorporated by reference in their entirety.
- additional modifications can be introduced into an AAV capsid variant described herein at positions determined using a structural map of wild-type AAV5, e.g., a structural map described and generated by Govindasamy et al. et al. Structural Insights into Adeno-Associated Virus Serotype 5. Journal of Virology.2013 Oct.87(20):11187-11199 (the contents of which are hereby incorporated herein by reference in their entirety) or Walters et al. “Structure of Adeno-Associated Virus Serotype 5,” Journal of Virology, 2004, 78(7):3361-3371 (the contents of which are hereby incorporated by reference in their entirety).
- an AAV capsid variant described herein comprises a modification as described in Jose et al. “High-Resolution Structural Characterization of a New Adenoassociated Virus Serotype 5 Antibody Epitope toward Engineering Antibody-Resistant Recombinant Gene Delivery Vectors,” Journal of Virology, 2020, 93(1): e01394-18; Qian et al. “Directed Evolution of AAV Serotype 5 for Increased Hepatocyte Transduction and Retained Low Humoral Seroreactivity,” Molecular Therapy: Methods and Clinical Development, 2021, 20:122-132; Afione et al.
- the AAV capsid variant further comprises an amino acid sequence comprising at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, of the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant further comprises an amino acid sequence comprising at least three, but not more than 30, not more than 20, or not more than 10 different amino acids relative to the amino acid sequence of SEQ ID NO: 138.
- the AAV capsid variant is a variant of SEQ ID NO: 138, wherein the AAV capsid variant comprises an amino acid sequence having at least 70% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 138.
- an AAV capsid variant of the present disclosure comprises the amino acid sequence of TTN-002, TTN-003, TTN-004, TTN-005, or TTN-006, e.g., as described in Table 3 or Attorney Docket No.14640.0092-00304 4A.
- an AAV capsid variant of the present disclosure comprises the amino acid sequence of TTN-002, e.g., as described in Table 3 or 4A.
- the AAV capsid variant comprises a VP1, VP2, and/or VP3 protein comprising an amino acid sequence of TTN-002, TTN-003, TTN-004, TTN-005, or TTN-006, e.g., as described in Table 3 or 4A.
- the AAV capsid variant comprises a VP1, VP2, and/or VP3 protein comprising an amino acid sequence of TTN-002, e.g., as described in Table 3 or 4A.
- the AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence of TTN-002, e.g., as described in Table 3 or 4B.
- a polynucleotide or nucleic acid encoding an AAV capsid variantof the present disclosure comprises the nucleotide sequence of TTN-002, e.g., as described in Table 3 or 4B. Table 3.
- Exemplary full-length capsid sequences Table 4A.
- Exemplary full-length capsid amino acid sequences Attorney Docket No.14640.0092-00304 Attorney Docket No.14640.0092-00304 Table 4B.
- an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
- an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 90% sequence identity thereto.
- an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 95% sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 96% sequence identity thereto. In Attorney Docket No.14640.0092-00304 some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 97% sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 98% sequence identity thereto.
- an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 99% sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 982. In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 982.
- an AAV capsid variant described herein comprises the amino acid sequence of any one of SEQ ID NOs: 740-743, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
- an AAV capsid variant described herein comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of any one of SEQ ID NOs: 740-743.
- the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 740-743.
- an AAV capsid variant described herein comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 984, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
- an AAV capsid variant described herein comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 different nucleotides, relative to the amino acid sequence of SEQ ID NO: 984.
- an AAV capsid variant described herein comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the nucleotide sequence of SEQ ID NO: 984.
- the nucleotide sequence encoding an AAV capsid variant, described herein comprises the nucleotide sequence of SEQ ID NO: 984, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
- the Attorney Docket No.14640.0092-00304 nucleotide sequence encoding an AAV capsid variant described herein comprises the nucleotide sequence of SEQ ID NO: 984, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
- 70% e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity thereto.
- the nucleotide sequence encoding an AAV capsid variant described herein comprises a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the nucleotide sequence of SEQ ID NO: 984.
- the nucleotide sequence encoding an AAV capsid variant described herein comprises a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 different nucleotides, relative to the amino acid sequence of SEQ ID NO: 984.
- an AAV capsid variant described herein comprises a VP1, VP2, VP3 protein, or a combination thereof.
- an AAV capsid variant comprises the amino acid sequence corresponding to positions 137-731, e.g., a VP2, of the amino acid sequence of SEQ ID NO: 982, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
- the AAV capsid protein comprises the amino acid sequence corresponding to positions 193-731, e.g., a VP3, of the amino acid sequence of SEQ ID NO: 982, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
- the AAV capsid variant comprises the amino acid sequence corresponding to positions 1- 731, e.g., a VP1, of the amino acid sequence of SEQ ID NO: 982, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto.
- at least 70% e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity thereto.
- an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 739, or an amino acid sequence at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto.
- the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 738, or an amino acid sequence at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto.
- the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982, or an amino acid sequence at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto.
- an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 739 (e.g., VP3).
- the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 738 (e.g., VP2).
- the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982 (e.g., VP1).
- the AAV capsid variant has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord Attorney Docket No.14640.0092-00304 cell, or spinal cord tissue, relative to the tropism of an AAV capsid that does not comprise the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) present at positions corresponding to amino acids 577-584 of the amino acid sequence of SEQ ID NO: 982.
- a CNS cell or tissue e.g., a brain cell, brain tissue, spinal cord Attorney Docket No.14640.0092-00304 cell, or spinal cord tissue
- an AAV capsid variant, described herein has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138.
- an AAV capsid variant described herein has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982.
- an AAV capsid variant described herein has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139.
- an AAV capsid variant described herein transduces a brain region, e.g., a temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, and/or cerebellum.
- the level of transduction is at least 0.5, at least 2.2, at least 2.4, at least 2.5, at least 2.6, at least 2.7, at least 3.0, at least 3.2, at least 3.5, at least 3.7, at least 4.0, at least 4.2, at least 4.5, at least 4.7, at least 4.9, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, or at least 35-fold greater as compared to the level of transduction of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139.
- an AAV capsid variant described herein is enriched at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 12, at least 15, at least 17, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, or at least 65-fold in the brain compared to enrichment in the brain of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138.
- an AAV capsid variant described herein is enriched at least 10, at least 12, at least 15, at least 17, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 61, at least 62, at least 63, at least 64, or at least 65-fold in the brain compared to enrichment in the brain of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138.
- an AAV capsid variant described herein is enriched in the brain of at least two to at least three species, e.g., a non-human primate and rodent (e.g., mouse and/or rat) species, compared to enrichment in the brain of those species of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138.
- a non-human primate and rodent e.g., mouse and/or rat
- an AAV capsid variant described herein is enriched at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 12, at least 15, at least 17, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 70, at least 80, at least 90, or at least 100-fold in the brain of at least two to at least three species, e.g., a non-human primate and rodent (e.g., mouse and/or rat) species, compared to enrichment in the brain of those species of an AAV capsid comprising the amino acid Attorney Docket No.14640.0092-00304 sequence of SEQ ID NO: 138.
- a non-human primate and rodent e.g., mouse and/or rat
- an AAV capsid variant described herein is enriched about 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 70, at least 80, at least 90, or at least 100, at least 125, at least 150, at least 175, at least 200, or at least 225-fold in the brain compared to enrichment in the brain of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982.
- an AAV capsid variant described herein delivers an increased number and/or level of viral genomes to a brain region.
- the number and/or level of viral genomes is increased by at least 1.5, at least 2.2, at least 2.4, at least 2.5, at least 2.6, at least 2.7, at least 3.0, at least 3.2, at least 3.5, at least 3.7, at least 4.0, at least 4.2, at least 4.5, at least 4.7, at least 4.9, or at least 5-fold, as compared to the number and/or level of viral genomes delivered by an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139.
- the brain region comprises a midbrain region (e.g., the hippocampus or thalamus) and/or the brainstem.
- an AAV capsid variant described herein delivers an increased level of frataxin to a brain region.
- the frataxin level is increased by at least 20, at least 25, at least 30, or at least 35-fold, as compared to the frataxin level delivered by an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139.
- the brain region comprises a temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, cerebellum, or a combination thereof.
- an AAV capsid variant described herein is enriched at least 3, at least 3.5, at least 4, at least 4.5, at least 5, at least 5.5, at least 6, or at least 6.5-fold, in a spinal cord region compared to enrichment in a spinal cord region of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139.
- the spinal cord region comprises a cervical spinal cord region, a lumbar spinal cord region, a thoracic spinal cord region, or a combination thereof.
- an AAV capsid variant described herein shows preferential transduction in a brain region relative to the transduction in the dorsal root ganglia (DRG).
- DDG dorsal root ganglia
- an AAV capsid variant described herein shows preferential transduction in a brain region relative to the transduction in the liver.
- an AAV capsid variant described herein is capable of transducing neuronal cells.
- an AAV capsid variant described herein is capable of transducing non- neuronal cells, e.g., glial cells (e.g., oligodendrocytes or astrocytes).
- the AAV capsid variant is capable of transducing neuronal cells and non-neuronal cells, e.g., glial cells (e.g., oligodendrocytes or astrocytes).
- the non-neuronal cells are glial cells (e.g., oligodendrocytes or astrocytes).
- an AAV capsid variant described herein has an increased tropism for a heart cell or heart tissue, e.g., a heart cell or a heart tissue of a heart atrium or a heart ventricle, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138.
- an AAV capsid variant described herein has an increased tropism for a heart cell or heart tissue, e.g., a heart cell or a heart tissue of a heart atrium or a heart ventricle, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139.
- an AAV capsid variant described herein delivers an increased level of frataxin to a heart region.
- the frataxin level is increased by at least 1.5, at least 2, or at least 2.5-fold, as compared to the frataxin level delivered by an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139.
- an AAV capsid variant described herein has decreased tropism for the liver.
- an AAV capsid variant comprises a modification that results in reduced tropism (e.g., de-targeting) and/or activity in the liver.
- the reduced tropism in the liver is compared to an otherwise similar capsid that does not comprise the modification, e.g., a wild-type capsid polypeptide.
- an AAV capsid variant described comprises a modification that results in one or more of the following properties: (1) reduced tropism in the liver; (2) de-targeted expression in the liver; (3) reduced activity in the liver; and/or (4) reduced binding to galactose.
- the reduction in any one, or all, of properties (1)-(3) is compared to an otherwise similar AAV capsid variant that does not comprise the modification.
- the AAV capsid variant e.g., the AAV capsid variant having reduced tropism in the liver, comprises one or more of: an amino acid other than A, G, K, M, N, Q, R, S, and/or T present at a position corresponding to amino acid 581 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A, C, H, I, K, S, T, and/or V present at a position corresponding to amino acid 582 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A, G, H, K, M, N, Q, R, S, T, and/or V present at a position corresponding to amino acid 583 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than L, M, P, Q, R.
- T and/or W present at a position corresponding to amino acid 584 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than F, H, I, K, M, T and/or Y present at a position corresponding to amino acid 585 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than E, G, H, L, M, N, Q, T, and/or W present at a position corresponding to amino acid 586 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A, C, G, H, L, M, R, and/or S present at a position corresponding to amino acid 587 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A, C, D, F, G, H, M, Q, S, V, W, and/or Y present at a position corresponding to amino acid 588 of the amino acid sequence of SEQ ID NO: 138; and/or an amino acid other than A, C, E, G, H,
- an AAV capsid variant of the present disclosure is isolated. In some embodiments, an AAV capsid variant of the present disclosure is recombinant. In some embodiments, a Attorney Docket No.14640.0092-00304 polynucleotide encoding an AAV capsid polypeptide, e.g., an AAV capsid variant, of the present disclosure is isolated. In some embodiments, the polynucleotide is recombinant. [0207] Also provided herein are polynucleotide sequences encoding any of the AAV capsid variants described above and AAV particles, vectors, and cells comprising the same.
- an AAV particle may comprise a capsid protein or variant of any natural or recombinant AAV serotype.
- AAV serotypes may differ in characteristics such as, but not limited to, packaging, tropism, transduction, and immunogenic profiles.
- the AAV capsid protein e.g., an AAV capsid variant
- an AAV capsid variant described herein allows for blood brain barrier penetration following intravenous administration.
- the AAV capsid variant allows for blood brain barrier penetration following intravenous administration, focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration.
- FUS focused ultrasound
- FUS-MB microbubbles
- MRI-guided FUS coupled with intravenous administration.
- the AAV capsid variant allows for increased distribution to a brain region.
- the brain region comprises a temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, cerebellum, frontal cortex, sensory cortex, motor cortex, dentate nucleus, cerebellar cortex, cerebral cortex, brain stem, or a combination thereof.
- the AAV capsid variant allows for preferential transduction in a brain region relative to the transduction in the dorsal root ganglia (DRG).
- DDG dorsal root ganglia
- the AAV capsid variant allows for preferential transduction in a brain region relative to the transduction in the liver.
- the AAV capsid variant allows for transduction in neuronal cells. In some embodiments, the AAV capsid variant allows for transduction in a non-neuronal cell, e.g., a glial cell (e.g., an astrocyte, an oligodendrocyte, or a combination thereof). In some embodiments, the AAV capsid variant allows for transduction in both neuronal cells and non-neuronal cell, e.g., a glial cell (e.g., an astrocyte, an oligodendrocyte, or a combination thereof). [0210] In some embodiments, an AAV capsid variant allows for increased distribution to a spinal cord region.
- the spinal region comprises a cervical spinal cord region, thoracic spinal cord region, and/or lumbar spinal cord region.
- the AAV capsid variant allows for increased distribution to a heart region.
- the AAV capsid variant is suitable for intramuscular administration and/or transduction of muscle fibers.
- the AAV capsid variant allows for increased distribution to a muscle region.
- the muscle region comprises a heart muscle, quadriceps muscle, a diaphragm muscle region, or a combination thereof.
- an AAV capsid described herein comprises a modification as described in Jose et al. High-Resolution Structural Characterization of a New Adenoassociated Virus Serotype 5 Antibody Epitope toward Engineering Antibody-Resistant Recombinant Gene Delivery Vectors. Journal of Virology.2019 Jan.93(1):e01394-18; Qian et al. Directed Evolution of AAV Serotype 5 for Increased Hepatocyte Transduction and Retained Low Humoral Seroreactivity. Molecular Therapy: Methods & Clinical Development.2020 Oct.20:122-132; Afione et al.
- the initiation codon for translation of the AAV VP1 capsid protein e.g., a capsid variant, described herein may be CTG, TTG, or GTG as described in US Patent No. US8163543, the contents of which are herein incorporated by reference in its entirety.
- the present disclosure refers to structural capsid proteins (including VP1, VP2 and VP3) which are encoded by capsid (Cap) genes.
- capsid proteins form an outer protein structural shell (e.g. capsid) of a viral vector such as AAV.
- VP capsid proteins synthesized from Cap polynucleotides generally include a methionine as the first amino acid in the peptide sequence (Met1), which is associated with the start codon (AUG or ATG) in the corresponding Cap nucleotide sequence.
- a first-methionine (Met1) residue or generally any first amino acid (AA1) to be cleaved off after or during polypeptide synthesis by protein processing enzymes such as Met-aminopeptidases.
- Met/AA-clipping This “Met/AA-clipping” process often correlates with a corresponding acetylation of the second amino acid in the polypeptide sequence (e.g., alanine, valine, serine, threonine, etc.). Met-clipping commonly occurs with VP1 and VP3 capsid proteins but can also occur with VP2 capsid proteins. [0216] Where the Met/AA-clipping is incomplete, a mixture of one or more (one, two or three) VP capsid proteins comprising the viral capsid may be produced, some of which may include a Met1/AA1 amino acid (Met+/AA+) and some of which may lack a Met1/AA1 amino acid as a result of Met/AA- clipping (Met-/AA-).
- references to capsid proteins is not limited to either clipped (Met-/AA-) or unclipped (Met+/AA+) and may, in context, refer to independent capsid proteins, viral capsids comprised of a mixture of capsid proteins, and/or polynucleotide sequences (or fragments thereof) which encode, describe, produce or result in capsid proteins of the present disclosure.
- a direct reference to a capsid protein or capsid polypeptide may also comprise VP capsid proteins which include a Met1/AA1 amino acid Attorney Docket No.14640.0092-00304 (Met+/AA+) as well as corresponding VP capsid proteins which lack the Met1/AA1 amino acid as a result of Met/AA-clipping (Met-/AA-).
- a reference to a specific SEQ ID NO: (whether a protein or nucleic acid) which comprises or encodes, respectively, one or more capsid proteins which include a Met1/AA1 amino acid (Met+/AA+) should be understood to teach the VP capsid proteins which lack the Met1/AA1 amino acid as upon review of the sequence, it is readily apparent any sequence which merely lacks the first listed amino acid (whether or not Met1/AA1).
- VP1 polypeptide sequence which is 736 amino acids in length and which includes a “Met1” amino acid (Met+) encoded by the AUG/ATG start codon may also be understood to teach a VP1 polypeptide sequence which is 735 amino acids in length and which does not include the “Met1” amino acid (Met-) of the 736 amino acid Met+ sequence.
- VP1 polypeptide sequence which is 736 amino acids in length and which includes an “AA1” amino acid (AA1+) encoded by any NNN initiator codon may also be understood to teach a VP1 polypeptide sequence which is 735 amino acids in length and which does not include the “AA1” amino acid (AA1-) of the 736 amino acid AA1+ sequence.
- references to viral capsids formed from VP capsid proteins can incorporate VP capsid proteins which include a Met1/AA1 amino acid (Met+/AA1+), corresponding VP capsid proteins which lack the Met1/AA1 amino acid as a result of Met/AA1-clipping (Met-/AA1-), and combinations thereof (Met+/AA1+ and Met-/AA1-).
- an AAV capsid serotype can include VP1 (Met+/AA1+), VP1 (Met-/AA1-), or a combination of VP1 (Met+/AA1+) and VP1 (Met-/AA1-).
- An AAV capsid serotype can also include VP3 (Met+/AA1+), VP3 (Met-/AA1-), or a combination of VP3 (Met+/AA1+) and VP3 (Met-/AA1-); and can also include similar optional combinations of VP2 (Met+/AA1) and VP2 (Met- /AA1-).
- the AAV capsid variant comprises, immediately subsequent to position 570, 571, 572, 573, 574, 575, or 576, numbered according to SEQ ID NO: 138 (in other words, immediately subsequent to an amino acid corresponding to position 570, 571, 572, 573, 574, 575, 576, or 577 of the amino acid sequence of SEQ ID NO: 138), at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, or at least 16 consecutive amino acids of any one of the amino acid sequences provided in Table 1, 2A, 2B, 14, or 23- 28.
- the AAV capsid variant comprises, immediately subsequent to position 570, 571, 572, 573, 574, 575, 576, or 577, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987 (the contents of which Attorney Docket No.14640.0092-00304 are hereby incorporated by reference in their entirety)), at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, or at least 16 consecutive amino acids of any one of the amino acid sequence
- the at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, or at least 16 consecutive amino acids of any one of the amino acid sequences provided in Table 1, 2A, 2B, 14, or 23-28 replaces at least one, at least two, at least three, at least four, at least five, at least six, at least seven, at least eight, or all of positions T571, N572, N573, Q574, S575, S576, T577, T578, and/or A579, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype (e.
- the at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, or at least 16 consecutive amino acids of any one of the amino acid sequences provided in Table 1, 2A, 2B, 14, or 23-28 replaces position T577, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987).
- AAV serotype e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAV
- the AAV capsid variant comprises an amino acid other than the wild-type, e.g., native, amino acid, at one, two, three, four, five, six, seven, eight, or all of positions T571, N572, N573, Q574, S575, S576, T577, T578, and/or A579, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987).
- the AAV capsid variant comprises an amino acid other than the wild-type, e.g., native, amino acid, at position T577, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987).
- AAV serotype e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided
- the AAV capsid variant comprises a modification, e.g., substitution, at one, two, three, four, five, six, seven, eight, or all of positions T571, N572, N573, Q574, S575, S576, T577, T578, and/or A579, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987).
- the AAV capsid variant comprises a modification, e.g., substitution, at position T577, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, Attorney Docket No.14640.0092-00304 AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987).
- AAV1, AAV2, AAV3, AAV3b Attorney Docket No.14640.0092-00304 AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV
- the AAV capsid variant described herein comprises a modification, e.g., substitution, at position 569 (e.g., M569V), 652 (e.g., D652A), 362 (e.g., T362M), 359 (e.g., Q359D), 350 (e.g., E350Q), 533 (e.g., P533S), 585 (e.g., Y585V), 587 (e.g., L587T), 581 (e.g., A581T), 582 (e.g., T582A), 584 (e.g., T584A), or a combination thereof, all numbered according to SEQ ID NO: 138.
- substitution at position 569 (e.g., M569V), 652 (e.g., D652A), 362 (e.g., T362M), 359 (e.g., Q359D), 350 (e.g., E350Q),
- an AAV capsid variant described herein comprises an amino acid from a wild-type AAV5 sequence, e.g., the amino acid sequence of SEQ ID NO: 138, at one or more of positions 581 to 589, numbered according to SEQ ID NO: 138.
- the AAV capsid variant comprises 1, 2, 3, 4, 5, 6, 7, 8, or all of: the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 581 (e.g., comprises the amino acid A at position 581); the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 582 (e.g., comprises the amino acid T at position 582); the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 583 (e.g., comprises the amino acid G at position 583); the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 584 (e.g., comprises the amino acid T at position 584); the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 585 (e.g., comprises the amino acid from a wild-
- an AAV capsid described herein does not comprise a T at position 581, an A at position 582, an A at position 584, a V at position 585, a T at position 585, a V at position 569, an A at position 652, an M at position 362, a Q at position 359, a Q at position 350, an S at position 533, or a combination thereof, all numbered according to SEQ ID NO: 138.
- an AAV capsid described herein does not comprise a modification, e.g., substitution, at positions 581-589 (numbered according to SEQ ID NO: 138), wherein the modification has the amino acid sequence of any of the sequences provided in Tables 2, 7, 8, 10, 12, 14, 16, 18, 20, 22, 24, 26, 28, 30, 32, 34, 36, 39, 41, 43, 45, 47, 49, 51, 53, 55, 57, 59, 61, 63, 65, 67, 69, or 71-86 of WO 2021/242909.
- a position numbered according to SEQ ID NO: 138 (in other words, a position in a peptide or AAV capsid variant corresponding to a position of SEQ ID NO: 138) can be identified by providing an alignment of a reference sequence and a query sequence, wherein the reference sequence is SEQ ID NO: 138, and identifying the residues corresponding to the positions in the query sequence that correspond to positions in the reference sequence.
- the AAV particle of the present disclosure serves as an expression vector comprising a viral genome that encodes a FXN protein (e.g., a human FXN protein, e.g., a wildtype human FXN protein).
- a FXN protein e.g., a human FXN protein, e.g., a wildtype human FXN protein.
- an AAV particle e.g., an AAV particle for the vectorized delivery of a FXN protein described herein, comprises a viral genome, e.g., an AAV viral genome, also referred to as an AAV genome, a vector genome, or an AAV vector genome.
- a viral genome e.g., an AAV viral genome, also referred to as an AAV genome, a vector genome, or an AAV vector genome.
- the viral genome e.g., the AAV viral genome, further comprises an inverted terminal repeat (ITR) region, an enhancer, a promoter, an intron region, an exon region, a nucleic acid encoding a transgene encoding a FXN protein sequence, e.g., any one of the sequences in Table 12 or a sequence that is at least 90% identical to any one of the sequences provided in Table 12), a nucleotide sequence encoding at least one miR binding site (e.g., at least one miR122 binding site), a polyA region, or a combination thereof.
- ITR inverted terminal repeat
- the viral genome may comprise at least one inverted terminal repeat (ITR) region.
- the AAV particles of the present disclosure comprise a viral genome with at least one ITR region and a FXN-encoding sequence.
- the viral genome has two ITRs. These two ITRs flank the FXN-encoding sequence at the 5’ and 3’ ends.
- the ITR functions as an origin of replication comprising a recognition site for replication.
- the ITR comprises a sequence region which can be complementary and symmetrically arranged.
- the ITR incorporated into a viral genome described herein may be comprised of a naturally occurring polynucleotide sequence or a recombinantly derived polynucleotide sequence.
- the AAV viral genome may comprise at least one inverted terminal repeat (ITR) region having a length of 50-250 nucleotides.
- the AAV viral genome comprises a 5’ ITR region that is 50-250 nucleotides in length and a 3’ ITR region that is 50-250 nucleotides in length.
- the 5’ ITR region and the 3’ ITR region may comprise the same length and/or the same sequence.
- the 5’ ITR region and the 3’ ITR region are different in length and/or in sequence.
- ITR sequence regions are described in Table 6.
- the viral genome comprises an ITR comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- the viral genome comprises an ITR comprising the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- the viral genome comprises a 5' ITR region comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the viral genome comprises a 3' ITR region comprising the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
- the viral genome comprises at least one element to enhance the transgene target specificity and expression. See, e.g., Powell et al. Viral Expression Cassette Elements to Enhance Transgene Target Specificity and Expression in Gene Therapy, 2015; the contents of which are herein incorporated by reference in their entirety.
- Non-limiting examples of elements to enhance the transgene target specificity and expression include promoters, endogenous miRNAs, post-transcriptional Attorney Docket No.14640.0092-00304 regulatory elements (PREs), polyadenylation (PolyA) region, upstream enhancers (USEs), CMV enhancers, and introns.
- PREs regulatory elements
- PolyA polyadenylation
- USEs upstream enhancers
- CMV enhancers CMV enhancers
- introns introns.
- expression of the polypeptides in a target cell may be driven by a specific promoter, including but not limited to, a promoter that is species specific, inducible, tissue- specific, or cell cycle-specific (Parr et al., Nat. Med.3:1145-9 (1997); the contents of which are herein incorporated by reference in their entirety).
- the viral genome comprises a promoter that is sufficient for expression, e.g., in a target cell, of a FXN protein, e.g., a human FXN protein, encoded by a transgene.
- the promoter is deemed to be efficient when it drives expression of the FXN encoded in the viral genome of the AAV particle.
- the promoter is deemed to be efficient when it drives expression in the cell or tissue being targeted.
- Promoters may be naturally occurring or non-naturally occurring. Non-limiting examples of promoters include viral promoters, plant promoters, and mammalian promoters. In some embodiments, the promoter may be a human promoter.
- the promoter may be truncated.
- Promoters that promote expression in most tissues include, but are not limited to, human elongation factor 1 ⁇ -subunit (EF1 ⁇ ) promoters, immediate-early cytomegalovirus (CMV) promoters, chicken ⁇ -actin (CBA) promoters, CAG promoters, CAG derivative promoters, ⁇ glucuronidase (GUSB) promoters, ubiquitin C (UBC) promoters, or a fragment of any one of the aforementioned.
- CMV immediate-early cytomegalovirus
- CBA chicken ⁇ -actin
- CAG promoters CAG promoters
- CAG derivative promoters CAG promoters
- GUSB ⁇ glucuronidase
- UBC ubiquitin C
- a CAG promoter typically comprises: (C) the cytomegalovirus early enhancer element; (A) the promoter, the first exon, and the first intron of the chicken beta-actin gene, and (G) the splice acceptor of the rabbit beta-globin gene.
- a derivative of a CAG promoter may comprise (i) a CMVie enhancer and a beta-actin promoter or (ii) a beta-actin promoter and an intron sequence.
- Tissue-specific expression elements can be used to restrict expression to certain cell types such as, but not limited to, nervous system promoters which can be used to restrict expression to neurons, astrocytes, or oligodendrocytes.
- tissue-specific expression elements for neurons include neuron-specific enolase (NSE), platelet-derived growth factor (PDGF), platelet-derived growth factor B-chain (PDGF- ⁇ ), synapsin (Syn), methyl-CpG binding protein 2 (MeCP2), CaMKII, mGluR2, NFL, NFH, n ⁇ 2, PPE, Enk, and EAAT2 promoters.
- tissue-specific expression elements for astrocytes include the glial fibrillary acidic protein (GFAP) and EAAT2 promoters.
- a non-limiting example of a tissue-specific expression element for oligodendrocytes include the myelin basic protein (MBP) promoter.
- the viral genome comprises a ubiquitous promoter.
- ubiquitous promoters include H1, U6, CMV, CBA (including derivatives CAG, CBh, etc.), EF-1 ⁇ , PGK, UBC, GUSB (hGBp), and UCOE (promoter of HNRPA2B1-CBX 3 ).
- the viral genome comprises a CBA promoter.
- the viral genome comprises a truncated CBA promoter, e.g., a CBA promoter that is 50-400 nucleotides Attorney Docket No.14640.0092-00304 in length, e.g., 100-332 nucleotides in length.
- the viral genome comprises a CMV promoter. In some embodiments, the viral genome comprises a truncated CMV promoter, e.g., a CMV promoter that is 50-300 nucleotides in length, e.g., a CMV promoter that is 109 nucleotides in length. [0243] In some embodiments, the AAV vector comprises an enhancer element, a promoter, and/or a 5’UTR intron.
- the enhancer may be, but is not limited to, a CMV enhancer; the promoter may be, but is not limited to, a CMV, CBA, FXN, UBC, GUSB, NSE, Synapsin, MeCP2, or GFAP promoter; and the 5’UTR/intron may be, but is not limited to, SV40, and CBA-MVM.
- the enhancer, promoter, and/or intron used in combination may be: (1) CMV enhancer, CMV promoter, SV405’UTR intron; (2) CMV enhancer, CBA promoter, SV405’UTR intron; (3) CMV enhancer, CBA promoter, CBA-MVM 5’UTR intron; (4) UBC promoter; (5) GUSB promoter; (6) NSE promoter; (7) Synapsin promoter; (8) MeCP2 promoter; (9) GFAP promoter; (10) H1 promoter; and/or (11) U6 promoter.
- the viral genome comprises an engineered promoter.
- the viral genome comprises an enhancer.
- an enhancer may be a separate component of the viral genome than the promoter. In some embodiments, an enhancer may be 5’ to a promoter sequence in a viral genome. In some embodiments, an enhancer may be 3’ to a promoter sequence in a viral genome.
- the viral genome comprises an enhancer, for example an immediate- early “ie” enhancer or a CMV/globin enhancer. In some embodiments, the enhancer comprises ie1 exon 1 and ie1 intron 1 or a fragment thereof. In some embodiments, the enhancer comprises an ie1 exon 1, an ie1 intron 1 or fragment thereof, a human beta-globin intron 2, and a human beta-globin exon 3.
- an enhancer may comprise at least one intron sequence. In some embodiments, an enhancer may comprise at least one exon sequence. In some embodiments, an enhancer comprises one intron sequence and one exon sequence. In some embodiments, an enhancer sequence comprises two intron sequences. In some embodiments, an enhancer sequence comprises two exon sequences. In some embodiments, an enhancer sequence comprises two intron sequences and two exon sequences. [0248] Exemplary promoters are provided in Table 7.
- the promoter comprises or consists of any one of the nucleotide sequences provided in Table 7, or a nucleotide sequence at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto.
- the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1738.
- the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1740.
- the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1742.
- the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1745. Table 7. Exemplary Promoters Promoter SEQ ID NO: Sequence C Attorney Docket No.14640.0092-00304 C C C Viral Genome Component: Introns and Exon Sequences [0249]
- the AAV viral genome comprises at least one intron and/or exon sequence region.
- the AAV viral genome may comprise at least one intron sequence region.
- the intron sequence region(s) may be 10-1200 nucleotides in length.
- the viral genome comprises an intron sequence region that is about 32 nucleotides in length.
- the viral genome comprises an intron sequence region that is about 53 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is about 134 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is about 347 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is about 379 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is about 566 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is about 1016 nucleotides in length.
- the viral genome comprises an intron sequence region that is more than 1016 nucleotides in length.
- the AAV viral genome comprises two intron sequence regions.
- the AAV viral genome comprises three intron sequence regions.
- the AAV viral genome comprises more than three intron sequence regions.
- the AAV viral genome may comprise at least one exon sequence region.
- the exon sequence may be 10-1200 nucleotides in length.
- the viral genome comprises an exon region that is about 32 nucleotides in length.
- the viral genome comprises an exon sequence region that is about 53 nucleotides in length.
- the viral genome comprises an exon sequence region that is about 134 nucleotides in length.
- the viral genome comprises an exon sequence region that is about 347 nucleotides in length.
- the viral genome comprises an exon sequence region that is about 379 nucleotides in length.
- the viral Attorney Docket No.14640.0092-00304 genome comprises an exon sequence region that is about 566 nucleotides in length.
- the viral genome comprises an exon sequence region that is about 1016 nucleotides in length.
- the viral genome comprises an exon sequence region that is more than 1016 nucleotides in length.
- the AAV particle viral genome comprises two exon sequence regions. In some embodiments, the AAV particle viral genome comprises three exon sequence regions. In some embodiments, the AAV particle viral genome comprises more than three exon sequence regions.
- the AAV particle viral genome comprises a hybrid intron/exon sequence region comprising at least one intron and at least one exon. In some embodiments, the hybrid intron/exon sequence region comprises one intron and one exon. In some embodiments, the hybrid intron/exon sequence region comprises two introns and two exons.
- an intron or exon sequence may comprise a full-length intron or exon. In some embodiments, an intron or exon sequence may comprise a fragment or variant of an intron or exon sequence.
- the hybrid intron/exon sequence region(s) may, independently, have a length such as, but not limited to, 15-100, 100-200, 200-300, 300-400, 400-500, 500-600, 600-700, 700-800, 800-900, 900- 1000, 1000-1100, 1100-1200, and more than 1200 nucleotides.
- the viral genome comprises a hybrid intron/exon sequence region that is about 379 nucleotides in length.
- the viral genome comprises a hybrid intron/exon sequence region that is about 566 nucleotides in length. As a non-limiting example, the viral genome comprises a hybrid intron/exon region that is about 379 nucleotides in length.
- the intron/exon sequence region is an enhancer sequence. In some embodiments, the intron/exon sequence region is not an enhancer sequence.
- the intron/exon sequence region is a component of a promoter sequence. In some embodiments, the intron/exon sequence region is not a component of a promoter sequence.
- the AAV particle viral genome comprises at least one intron and/or exon sequence region.
- intron and exon sequence regions are described in Table 8.
- the exon sequence region comprises the nucleotide sequence of SEQ ID NO: 1816, or a nucleotide sequence with at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence identity thereto.
- the exon sequence region comprises the nucleotide sequence of SEQ ID NO: 1817, or a nucleotide sequence with at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence identity thereto. In some embodiments, the exon sequence region comprises the nucleotide sequence of SEQ ID NO: 1819, or a nucleotide sequence with at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence identity thereto.
- the exon sequence region comprises the nucleotide sequence of SEQ ID NO: 1820, or a nucleotide sequence with at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence Attorney Docket No.14640.0092-00304 identity thereto.
- the exon sequence region comprises the nucleotide sequence of SEQ ID NO: 1821, or a nucleotide sequence with at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence identity thereto.
- Tissue- or cell-specific expression of the AAV viral particles of the disclosure can be enhanced by introducing tissue- or cell-specific regulatory sequences, e.g., promoters, enhancers, microRNA binding sites, e.g., a detargeting site.
- tissue- or cell-specific regulatory sequences e.g., promoters, enhancers, microRNA binding sites, e.g., a detargeting site.
- an encoded miR binding site can modulate, e.g., prevent, suppress, or otherwise inhibit, the expression of a gene of interest on the viral genome of the invention, based on the expression of the corresponding endogenous microRNA (miRNA) or a corresponding controlled exogenous miRNA in a tissue or cell, e.g., a non-targeting cell or tissue.
- a miR binding site modulates, e.g., reduces, expression of the FXN-encoding region in a viral genome of an AAV particle described herein in a cell or tissue where the corresponding mRNA is expressed.
- the miR binding site modulates, e.g., reduces, expression of the encoded FXN protein in a cell or tissue of the DRG or liver.
- the viral genome of an AAV particle described herein comprises a nucleotide sequence encoding a microRNA binding site, e.g., a detargeting site.
- the viral genome of an AAV particle described herein comprises a nucleotide sequence encoding a miR Attorney Docket No.14640.0092-00304 binding site, a microRNA binding site series (miR BSs), or a reverse complement thereof.
- the 3' UTR of the viral genome may be engineered to include at least one miRNA binding site.
- the encoded miR binding site series comprise at least 1-5 copies, e.g., at least 1-3, 2-4, 3-5, or at least 1, at least 2, at least 3, at least 4, at least 5 or more copies of a miR binding site (miR BS).
- the encoded miR binding site series comprises 3 copies of a miR binding site. In some embodiments, all copies are identical, e.g., comprise the same miR binding site. In some embodiments, the miR binding sites within the encoded miR binding site series are continuous and not separated by a spacer.
- the miR binding sites within an encoded miR binding site series are separated by a spacer, e.g., a non-coding sequence.
- the viral genome comprises at least one sequence encoding a miRNA binding site to reduce the expression of the transgene in a specific tissue.
- the viral genome may comprise a miR122 binding site (miR122BS) or tandem copies of the miR122BS to reduce the expression of the viral genome in the liver.
- the viral genome may comprise a miR-183 miRNA binding site (miR-183BS) or tandem copies of the miR-183BS to reduce expression of the viral genome in the DRG.
- the miR binding site may be 20-75 nucleotides in length. In some embodiments, the miR binding site is 23 nucleotides in length. In some embodiments, the miR binding site is 71 nucleotides in length. [0264] Non-limiting examples of miR-binding site sequence regions are shown in Table 9. [0265] In some embodiments, the encoded miR binding site is fully complementary to an miR. In some embodiments, the encoded miR binding site is partially complementary to an miR. In some embodiments, the miR is expressed in the liver or in hepatocytes. In some embodiments, the miR is miR122.
- the encoded miR binding site or encoded miR binding site series comprises a miR122 binding site sequence.
- the encoded miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1827, or a nucleotide sequence having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99%sequence identity, or comprising at least one, at least two, at least three, at least four, at least five, or six modifications, but no more than six modifications, relative to the nucleotide sequence of SEQ ID NO: 1827, wherein the modification can result in a mismatch between the encoded miR binding site and the corresponding miRNA.
- the viral genome comprises at least 2, at least 3, at least 4, or at least 5 copies of the encoded miR122 binding site, e.g., an encoded miR122 binding site series, optionally wherein the encoded miR122 binding site series comprises the nucleotide sequence of SEQ ID NO: 1826, or a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence identity, or comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: Attorney Docket No.14640.0092-00304 1826, wherein the modification can result in a mismatch between the encoded miR binding site and the corresponding miRNA.
- the viral genome of the AAV particles of the present disclosure comprises at least one polyadenylation (polyA) region.
- the polyA region is positioned 3’ relative to the nucleic acid encoding a FXN protein described herein.
- the polyA region comprises a length of about 100 to about 500 nucleotides, e.g., about 477 nucleotides. In some embodiments, the polyA region comprises a length of 477 nucleotides.
- polyA region comprises the nucleotide sequence of SEQ ID NO: 1828, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto.
- Table 10 Exemplary PolyA Region S R N G G G T C h T A C C G Viral Genome Component: Filler (Stuffer) Sequence
- shuffer sequence and “filler sequence” are used interchangeably.
- the AAV particle viral genome comprises at least one filler sequence. In some embodiments, the AAV particle viral genome comprises a filler sequence comprising a human albumin sequence. In some embodiments, the AAV particle viral genome comprises the filler sequence of Alb2034. In some embodiments, the AAV particle viral genome comprises the filler sequence of Alb2106. In some embodiments, the AAV particle viral genome comprises the filler sequence of Attorney Docket No.14640.0092-00304 Alb2264. In some embodiments, the AAV particle viral genome comprises the filler sequence of Alb2266 [0270] Non-limiting examples of filler sequences are described in Table 11.
- the filler sequence comprises the nucleotide sequence of SEQ ID NO: 1838, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto.
- the filler sequence comprises the nucleotide sequence of SEQ ID NO: 1839, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto.
- the filler sequence comprises the nucleotide sequence of SEQ ID NO: 1840, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto.
- the filler sequence comprises the nucleotide sequence of SEQ ID NO: 1841, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. Table 11.
- the viral genome comprises the polynucleotide sequence of SEQ ID NO: 1797. In some embodiments, the viral genome comprises a promoter operably linked to a polynucleotide sequence encoding a FXN protein. In some embodiments, the viral genome comprises components as arranged in FIG.3. [0272] In some embodiments, the disclosure herein provides constructs that allow for improved expression of FXN protein (e.g., a human frataxin protein) delivered by gene therapy vectors. [0273] In some embodiments, the disclosure provides constructs that allow for improved biodistribution of FXN protein (e.g., a human frataxin protein) delivered by gene therapy vectors.
- FXN protein e.g., a human frataxin protein
- the present disclosure relates to a composition containing or comprising a nucleic acid sequence encoding a FXN protein (e.g., a human frataxin protein) or a functional fragment or variant thereof and methods of administering the composition in vitro or in vivo in a subject, e.g., a human subject and/or an animal model of disease, e.g., Friedreich’s Ataxia.
- a FXN protein e.g., a human frataxin protein
- the disclosure provides a nucleotide sequence encoding a FXN protein (e.g., a human frataxin protein) for use in an AAV viral genome, wherein the nucleotide sequence comprises any one of the sequences provided in Table 12, or a sequence that is at least 90% identical to any one of the sequences provided in Table 12.
- the AAV viral genome further comprises one or more, e.g., all of, a 5’ ITR sequence, a promoter, an intron sequence, a polyA sequence, at least one miR122 binding site, and a 3’ ITR region.
- the FXN protein encoded by the viral genome is a human FXN protein. Table 12.
- the FXN protein is encoded by the nucleotide sequence of SEQ ID NO: 1824, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto.
- the AAV viral genome comprises a combination of coding and non- coding nucleic acid sequences.
- the FXN-encoding sequence encodes a gene therapy product including, but not limited to, a polypeptide, protein, RNA molecule, or other gene product that, when expressed in a target cell, provides a desired therapeutic effect.
- a gene therapy product may provide a substitute for a non-functional gene or a gene that is absent, expressed in insufficient amounts, or mutated.
- a gene therapy product may comprise a substitute for a non-functional protein or polypeptide or a protein or polypeptide that is absent, expressed in insufficient amounts, misfolded, degraded too rapidly, or mutated.
- a gene therapy product may be encoded by a polynucleotide encoding a FXN protein to treat FA.
- the gene therapy product is encoded by a polynucleotide sequence in an AAV genome, wherein the AAV genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the gene Attorney Docket No.14640.0092-00304 therapy product is encoded by a polynucleotide sequence in an AAV genome, wherein the AAV genome comprises the nucleotide sequence of SEQ ID NO: 1801.
- the gene therapy product is encoded by a polynucleotide sequence in an AAV genome, wherein the AAV genome comprises the nucleotide sequence of SEQ ID NO: 1808. In some embodiments, the gene therapy product is encoded by a polynucleotide sequence in an AAV genome, wherein the AAV genome comprises the nucleotide sequence of SEQ ID NO: 1809. [0282] In some embodiments, the payload encodes a messenger RNA (mRNA).
- mRNA messenger RNA
- mRNA messenger RNA
- FXN e.g., human FXN
- a viral genome may comprise or encode a selectable marker.
- a selectable marker may comprise a gene sequence or a protein or polypeptide encoded by a gene sequence expressed in a host cell that allows for the identification, selection, and/or purification of the host cell from a population of cells that may or may not express the selectable marker.
- the selectable marker provides resistance to survive a selection process that would otherwise kill the host cell, such as treatment with an antibiotic.
- an antibiotic selectable marker may comprise one or more antibiotic resistance factors, including but not limited to neomycin resistance (e.g., neo), hygromycin resistance, kanamycin resistance, and/or puromycin resistance.
- a viral genome may comprise a selectable marker including, but not limited to, ⁇ -lactamase, luciferase, ⁇ -galactosidase, or any other reporter gene as that term is understood in the art.
- a viral genome may encode a selectable marker comprising a fluorescent protein.
- a fluorescent protein as herein described may comprise any fluorescent marker including but not limited to green, yellow, and/or red fluorescent protein (GFP, YFP, and/or RFP).
- GFP green, yellow, and/or red fluorescent protein
- a viral genome may encode a selectable marker comprising a human influenza hemagglutinin (HA) tag.
- a nucleic acid for expression of a FXN protein in a target cell will be incorporated into the viral genome and located between two ITR sequences.
- Exemplary FXN AAV Viral Genome Sequence Regions and ITR-to-ITR Sequences [0286]
- a viral genome described herein comprises a promoter operably linked to a transgene encoding a FXN protein (e.g., a human FXN protein).
- the viral genome further comprises an inverted terminal repeat (ITR) region, a promoter, an intron/exon region, a miR binding site region, a polyA region, or a combination thereof (e.g., all of these elements).
- Exemplary sequence regions within ITR-to-ITR sequences for viral genomes according to the description are provided in Tables 13A and 13B and disclosures below.
- An exemplary arrangement of an ITR-to-ITR sequence is also depicted in FIG.3.
- Attorney Docket No.14640.0092-00304 Table 13A Exemplary Sequence Regions within ITR-to-ITR Sequences Table 13B.
- Exemplary ITR-to-ITR Sequences Attorney Docket No.14640.0092-00304 [0288] SEQ ID NO: 1797:
- the AAV viral genome comprises a 5’ ITR region comprising the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical to the nucleotide sequence of SEQ ID NO: 1811 and/or a 3’ ITR region comprising the nucleotide sequence of SEQ ID NO: 1812 or a nucleotide sequence that is at least 90% identical to SEQ ID NO: 1812.
- the AAV viral genome further comprises or consists of a truncated promoter region of 100-332 nucleotides in length and comprises any one of SEQ ID NOs: 1738, 1740, 1742, or 1750, or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to any one of SEQ ID NOs: 1738, 1740, 1742, or 1750.
- a truncated promoter region of 100-332 nucleotides in length and comprises any one of SEQ ID NOs: 1738, 1740, 1742, or 1750, or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at
- the AAV viral genome further comprises an intron/exon region comprising SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1816.
- the AAV viral genome comprises a miR122 binding site comprising SEQ ID NO: 1827 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1827.
- the AAV viral genome further comprises a miR122 binding site series comprising SEQ ID NO: 1826 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1826.
- a miR122 binding site series comprising SEQ ID NO: 1826 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1826.
- the AAV viral genome further comprises a polyA region comprising SEQ ID NO: 1828 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1828.
- a polyA region comprising SEQ ID NO: 1828 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1828.
- the AAV viral genome further comprises a filler sequence comprising any one of SEQ ID NOs: 1838, 1839, 1840, or 1841, or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to any one of SEQ ID NOs: 1838, 1839, 1840, or 1841.
- the AAV viral genome does not comprise a filler sequence.
- the AAV particle comprises, from 5' to 3', a 5' ITR comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical thereto; a promoter consisting of the nucleotide sequence of SEQ ID NO: 1742 or a sequence that is at least 90% identical thereto; an intron/exon region comprising the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical thereto; a FXN-encoding sequence comprising SEQ ID NO: 1824; a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that is at least 90% identical thereto; a polyA region comprising the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical thereto; and/or a 3' ITR comprising the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical thereto; and/
- the AAV particle comprising the FXN-encoding sequence of SEQ ID NO: 1824 further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1841 or a sequence that is at least 90% identical thereto, wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1809.
- Attorney Docket No.14640.0092-00304 [0301]
- the viral genome is single-stranded.
- the viral genome is capable of forming double-stranded DNA.
- the viral genome is self-complementary.
- the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; and (iv) a 3’ ITR region.
- ITR inverted terminal repeat
- the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (iv) at least one miR122 binding site; and (v) a 3’ ITR region.
- ITR inverted terminal repeat
- the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron and/or exon region; (iv) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; and (vi) a 3’ ITR region.
- ITR inverted terminal repeat
- the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron and/or exon region; (iv) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; and (vii) a 3’ ITR region.
- ITR inverted terminal repeat
- the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron and/or exon region; (iv) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; (vii) a filler sequence; and (vii)
- the AAV viral genome comprises the nucleotide sequence of SEQ ID NO: 1797 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1797.
- the AAV viral genome comprises a truncated promoter comprising or consisting of the nucleotide sequence of SEQ ID NO: 1742, or a sequence that is at least 95% identical to SEQ ID NO: 1742.
- the AAV particle comprises, from 5' to 3', one or more of, e.g., all of, a 5' ITR comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical thereto; a promoter consisting of the nucleotide sequence of SEQ ID NO: 1750 or a sequence that is at least 90% identical thereto; an intron/exon region comprising the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical thereto; a FXN-encoding sequence comprising SEQ ID NO: 1824; a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that
- the AAV particle comprising the FXN-encoding sequence of SEQ ID NO: 1824 further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1840 or a sequence that is at least 90% identical thereto, wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region.
- the AAV viral genome comprises the nucleotide sequence of SEQ ID NO: 1801 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1801.
- the AAV viral genome comprises a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to SEQ ID NO: 1801.
- the AAV viral genome comprises a truncated promoter comprising or consisting of the nucleotide sequence of SEQ ID NO: 1750, or a sequence that is at least 95% identical to SEQ ID NO: 1750.
- the AAV particle comprises, from 5' to 3', a 5' ITR comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical thereto; a promoter consisting of the nucleotide sequence of SEQ ID NO: 1738 or a sequence that is at least 90% identical thereto; an intron/exon region comprising the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical thereto; a FXN-encoding sequence comprising SEQ ID NO: 1824; a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that is at least 90% identical thereto; a polyA region comprising the nucleotide sequence of SEQ ID NO: 1828 or a Attorney Docket No.14640.0092-00304 sequence that is at least 90% identical thereto; and/or a 3' ITR comprising the nucleotide sequence of SEQ ID NO: 1811 or
- the AAV particle comprising the FXN-encoding sequence of SEQ ID NO: 1824 further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1838 or a sequence that is at least 90% identical thereto, wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region.
- the AAV viral genome comprises the nucleotide sequence of SEQ ID NO: 1808 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1808.
- the AAV viral genome comprises a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to SEQ ID NO: 1808.
- the AAV viral genome comprises a truncated promoter comprising or consisting of the nucleotide sequence of SEQ ID NO: 1738, or a sequence that is at least 95% identical to SEQ ID NO: 1738.
- the AAV particle comprises, from 5' to 3', a 5' ITR comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical thereto a promoter consisting of the nucleotide sequence of SEQ ID NO: 1740 or a sequence that is at least 90% identical thereto; an intron/exon region comprising the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical thereto; a FXN-encoding sequence comprising SEQ ID NO: 1824000; a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that is at least 90% identical thereto; a polyA region comprising the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical thereto; and/or a 3' ITR comprising the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical thereto a promote
- the AAV particle comprising the FXN-encoding sequence of SEQ ID NO: 1824 further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1839 or a sequence that is at least 90% identical thereto, wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region.
- the AAV viral genome comprises the nucleotide sequence of SEQ ID NO: 1809 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1809.
- the AAV viral genome comprises a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to SEQ ID NO: 1809.
- the AAV viral genome comprises a truncated promoter comprising or consisting of the nucleotide sequence of SEQ ID NO: 1740, or a sequence that is at least 95% identical to SEQ ID NO: 1740.
- the present disclosure provides an AAV particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides an AAV particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides an AAV particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the FXN-encoding sequences described herein facilitate an intermediate level of expression (e.g., 0.5x-3x relative to baseline) such that the resulting expression alleviates one or more symptoms of FXN deficiency or FA while minimizing or avoiding toxicity.
- this result is facilitated at least by a truncated promoter as described herein.
- the FXN- encoding sequences described herein minimize or avoid overexpression of frataxin in the liver.
- this result is facilitated at least by a miR binding site or series as described herein.
- desirable expression levels or distribution of frataxin are further facilitated by the AAV capsid variants described herein.
- the AAV capsid variants described herein may enhance delivery to the cerebellum. Further, without being bound by theory, the AAV capsid variants described herein may enhance delivery to the dentate nucleus.
- Self-Complementary and Single Stranded Vectors [0319] In some embodiments, the AAV viral genome used in the present disclosure is single- stranded. [0320] In some embodiments, the AAV viral genome is capable of forming double-stranded DNA. In some embodiments, the AAV viral genome is self-complementary. See, e.g., US Patent No.7,465,583. scAAV vectors contain both DNA strands that anneal together to form double stranded DNA.
- scAAVs allow for rapid expression in the cell.
- Methods for producing and/or modifying AAV viral genome and particles are disclosed in the art such as pseudotyped AAV vectors (International Patent Publication Nos. WO200028004; WO200123001; WO2004112727; WO 2005005610 and WO 2005072364, the content of each of which are incorporated herein by reference in their entirety).
- Attorney Docket No.14640.0092-00304 II are disclosed in the art such as pseudotyped AAV vectors (International Patent Publication Nos. WO200028004; WO200123001; WO2004112727; WO 2005005610 and WO 2005072364, the content of each of which are incorporated herein by reference in their entirety).
- Viral production disclosed herein describes processes and methods for producing AAV particles (with enhanced, improved and/or increased tropism for a target tissue), e.g., an AAV particle comprising an AAV capsid variant that may be used to contact a target cell to deliver FXN.
- an AAV particle of the present disclosure e.g., an AAV particle comprising an AAV capsid variant disclosed herein
- the method comprises: (i) providing a cell comprising a viral genome described herein (e.g., a viral genome comprising a FXN-encoding sequence and a nucleic acid encoding an AAV capsid variant disclosed herein) and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in an AAV capsid variant described herein (e.g., an AAV capsid variant listed in Table 3, 4A, or 4B), thereby making the AAV particle.
- a viral genome described herein e.g., a viral genome comprising a FXN-encoding sequence and a nucleic acid encoding an AAV capsid variant disclosed herein
- incubating the cell under conditions suitable to encapsulate the viral genome in an AAV capsid variant described herein (e.g., an AAV capsid variant listed in Table 3,
- the viral genome comprises a nucleotide sequence of any one of SEQ ID NOs: 1797, 1801, 1808, and 1809, or a nucleotide sequence at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to any one of the nucleotide sequences of SEQ ID NOs: 1797, 1801, 1808, and 1809.
- the AAV capsid variant comprises an amino acid sequence of SEQ ID NO: 982.
- the viral genome comprises the nucleotide sequence of any one of SEQ ID NOs: 1797, 1801, 1808, and 1809, or a nucleotide sequence at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to any one of the nucleotide sequences of SEQ ID NOs: 1797, 1801, 1808, and 1809; and the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982.
- the method comprises, prior to step (i), introducing a nucleic acid comprising the viral genome into the cell.
- the method comprises, prior to step (i), introducing the nucleic acid encoding the AAV capsid variant into the cell.
- the AAV particle described herein is an isolated AAV particle.
- the AAV particle described herein is a recombinant AAV particle.
- the present disclosure provides a method of making an AAV particle that comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982.
- the iral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides a method of making an AAV particle that comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides a method of making an AAV particle that comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- AAV particles are produced in mammalian cells (e.g., HEK293 cells). In another embodiment, AAV particles are produced in insect cells (e.g., Sf9 cells). [0328] Methods of making AAV particles are well known in the art and are described in e.g., U.S. Patent Nos.
- the AAV particles are made using the methods described in International Patent Publication WO2015191508, the contents of which are herein incorporated by reference in their entirety.
- the present disclosure provides pharmaceutical compositions of an adeno-associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides pharmaceutical compositions of an adeno-associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides pharmaceutical compositions of an adeno-associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- a composition described herein comprises an AAV polynucleotide or AAV genome or AAV particle and at least one excipient.
- pharmaceutical compositions provided herein e.g., comprising AAV particles comprising a FXN-encoding sequence, are principally directed to pharmaceutical compositions which are suitable for administration to humans, it will be understood by the skilled artisan that such compositions are generally suitable for administration to any other animal, e.g., to non-human animals, e.g., non- human mammals.
- compositions suitable for administration to humans in order to render the compositions suitable for administration to various animals is well understood, and Attorney Docket No.14640.0092-00304 the ordinarily skilled veterinary pharmacologist can design and/or perform such modification with merely ordinary, if any, experimentation.
- Subjects to which administration of the pharmaceutical compositions is contemplated include, but are not limited to, humans and/or other primates; mammals, including commercially relevant mammals such as cattle, pigs, horses, sheep, cats, dogs, mice, and/or rats; and/or birds, including commercially relevant birds such as poultry, chickens, ducks, geese, and/or turkeys.
- compositions are administered to humans, e.g., human patients or human subjects.
- the AAV particle formulations described herein may contain a nucleic acid encoding at least one payload.
- the formulations may contain a nucleic acid encoding 1, 2, 3, 4, or 5 payloads.
- the formulation may contain FXN-encoding sequence, encoding a FXN protein selected from categories such as, but not limited to, human proteins, veterinary proteins, bacterial proteins, biological proteins, antibodies, immunogenic proteins, therapeutic peptides and proteins, secreted proteins, plasma membrane proteins, cytoplasmic proteins, cytoskeletal proteins, intracellular membrane bound proteins, nuclear proteins, proteins associated with human disease, and/or proteins associated with non-human diseases.
- the AAV genome comprises at least one sequence encoding human FXN. In some embodiments, the AAV genome comprises at least one sequence encoding wildtype human FXN.
- a pharmaceutical composition in accordance with the present disclosure may be prepared, packaged, and/or sold in bulk, as a single unit dose, and/or as a plurality of single unit doses.
- a “unit dose” refers to a discrete amount of the pharmaceutical composition comprising a predetermined amount of the active ingredient. The amount of the active ingredient is generally equal to the dosage of the active ingredient which would be administered to a subject and/or a convenient fraction of such a dosage such as, for example, one-half or one-third of such a dosage. IV.
- Formulations of the AAV pharmaceutical compositions described herein may be prepared by any method known or hereafter developed in the art of pharmacology. In general, such preparatory methods include the step of bringing the active ingredient into association with an excipient and/or one or more other accessory ingredients, and then, if necessary and/or desirable, dividing, shaping and/or packaging the product into a desired single- or multi-dose unit. [0338] Relative amounts of the active ingredient, the pharmaceutically acceptable excipient, and/or any additional ingredients in a pharmaceutical composition in accordance with the disclosure will vary, depending upon the identity, size, and/or condition of the subject treated and further depending upon the route by which the composition is to be administered.
- the composition may comprise between 0.1% and 99% (w/w) of the active ingredient.
- the composition may comprise between 0.1% and 100%, e.g., between .5% and 50%, between 1-30%, between 5-80%, or at least 80% (w/w) active ingredient.
- the AAV particles of the disclosure can be formulated using one or more excipients to: (1) increase stability; (2) increase cell transfection or transduction; (3) permit the sustained or delayed release; (4) alter the biodistribution (e.g., target the viral particle to specific tissues or cell types); (5) increase the translation of encoded protein in vivo; (6) alter the release profile of encoded protein in vivo and/or (7) allow for regulatable expression of the FXN.
- Formulations of the present disclosure can include, without limitation, saline, lipidoids, liposomes, lipid nanoparticles, polymers, lipoplexes, core-shell nanoparticles, peptides, proteins, cells transfected with AAV particles (e.g., for transplantation into a subject), nanoparticle mimics and combinations thereof. Further, the AAV particles of the present disclosure may be formulated using self- assembled nucleic acid nanoparticles.
- the present disclosure provides formulations of an adeno-associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982, the amino acid sequence of SEQ ID NO: 738, and/or the amino acid sequence of SEQ ID NO: 739.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the AAV particles encoding FXN may be formulated to optimize baricity and/or osmolality.
- the baricity and/or osmolality of the formulation may be optimized to ensure optimal drug distribution in the central nervous system or a region or component of the central nervous system.
- Excipients [0344]
- the formulations of the disclosure can include one or more excipients, each in an amount that together increases the stability of the AAV particle, increases cell transfection or transduction by the AAV particle, increases the expression of frataxin protein, and/or alters the release profile of frataxin proteins.
- a pharmaceutically acceptable excipient may be at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% pure.
- an excipient is approved for use for humans and for veterinary use.
- an excipient may be approved by United States Food and Drug Administration. In some embodiments, an excipient may be of pharmaceutical grade. In some embodiments, an excipient may meet the standards of the United States Pharmacopoeia (USP), the European Pharmacopoeia (EP), the British Pharmacopoeia, and/or the International Pharmacopoeia.
- Excipients which, as used herein, include, but are not limited to, any and all solvents, dispersion media, diluents, or other liquid vehicles, dispersion or suspension aids, surface active agents, isotonic agents, thickening or emulsifying agents, preservatives, and the like, as suited to the particular dosage form desired.
- excipients for formulating pharmaceutical compositions and techniques for preparing the composition are known in the art (see Remington: The Science and Practice of Pharmacy, 21st Edition, A. R. Gennaro, Lippincott, Williams & Wilkins, Baltimore, MD, 2006; the contents of which are herein incorporated by reference in their entirety).
- the use of a conventional excipient medium Attorney Docket No.14640.0092-00304 may be contemplated within the scope of the present disclosure, except insofar as any conventional excipient medium may be incompatible with a substance or its derivatives, such as by producing any undesirable biological effect or otherwise interacting in a deleterious manner with any other component(s) of the pharmaceutical composition.
- AAV formulations may comprise at least one excipient which is an inactive ingredient.
- the term “inactive ingredient” refers to one or more agents that do not contribute to the activity of the pharmaceutical composition included in formulations. In some embodiments, all, none, or some of the inactive ingredients which may be used in the formulations of the present disclosure may be approved by the US Food and Drug Administration (FDA).
- FDA US Food and Drug Administration
- Formulations of AAV particles disclosed herein may include cations or anions. In one embodiment, the formulations include metal cations such as, but not limited to, Zn 2+ , Ca 2+ , Cu 2+ , Mg + , or combinations thereof.
- formulations may include polymers or polynucleotides complexed with a metal cation (See, e.g., U.S. Pat. Nos.6,265,389 and 6,555,525, the contents of each of which are herein incorporated by reference in their entirety).
- the compositions of the disclosure may be administered to a subject, e.g., to deliver a FXN protein, e.g., to a subject who has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency (e.g., Friedreich’s Ataxia (FA)).
- a disorder associated with FXN deficiency e.g., Friedreich’s Ataxia (FA)
- compositions may similarly be used in the manufacture of a medicament for administration to a subject having a FXN protein deficiency, such as FA.
- the disclosure provides a method of delivering a FXN protein to a subject comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle disclosed herein that comprises a viral genome encoding a FXN protein.
- the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency (e.g., FA).
- the disclosure provides a method for treating a disorder associated with FXN deficiency, such as FA.
- the disclosure provides an AAV particle or pharmaceutical composition according to any one of the embodiments disclosed herein for treating a disorder associated with FXN deficiency, such as FA.
- the present disclosure provides the pharmaceutical composition or the AAV particle of any one the embodiments disclosed herein for use in a method of treating a disorder as disclosed herein, such as FA.
- a pharmaceutical composition or AAV particle disclosed herein that comprises a viral genome encoding FXN may be administered to a subject to treat a disorder associated with FXN deficiency such as FA.
- the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency such as FA.
- the treatment results in a 0.5-3x increase in the subject’s FXN protein level as compared to Attorney Docket No.14640.0092-00304 baseline.
- the treatment may halt or slow progression of FA.
- the treatment improves at least one symptom of FA, e.g., impaired sensory functions, impaired motor function (e.g., ataxia and/or involuntary movements), fatigue, chronic pain, seizures, impaired speech, sleep disturbances, metabolic disorders (e.g., diabetes), and/or increased spasticity.
- the treatment stabilizes, slows the progression of, or improves the subject’s FA as determined by the modified Friedreich Ataxia Rating Scale (mFARS), the Scale for the Assessment and Rating of Ataxia (SARA), and/or the International Cooperative Ataxia Rating Scale (ICARS).
- mFARS modified Friedreich Ataxia Rating Scale
- SARA Scale for the Assessment and Rating of Ataxia
- ICARS International Cooperative Ataxia Rating Scale
- the treatment slows the subject’s progression of FA as measured by mFARS, SARA, and/or ICARS relative to an individual with the disorder associated with FXN deficiency who has not been administered the pharmaceutical composition or the AAV particle.
- the treatment results in an increase in: (i) the level of FXN protein or FXN gene expression in a cell, tissue, (e.g., a cell or tissue of the CNS, e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum), and/or fluid (e.g., CSF and/or serum), of the subject, optionally wherein the level of FXN protein or gene expression is increased by 0.5-3x as compared to baseline; and/or (ii) the number and/or level of viral genomes (VG) per cell in a CNS tissue (e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus
- VG
- AAV particles comprising AAV capsid variants described herein may be desirable for the alleviation or treatment of frataxin deficiency or Friedreich’s Ataxia at least due to their tropism of the dentate nucleus.
- the present disclosure provides delivery of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides delivery of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides delivery of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 Attorney Docket No.14640.0092-00304 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure encompasses the delivery of pharmaceutical, prophylactic, diagnostic, or imaging compositions in combination with agents that may improve their bioavailability, reduce and/or modify their metabolism, and/or modify their distribution within the body.
- the present disclosure encompasses the delivery of pharmaceutical, prophylactic, diagnostic, or imaging compositions in combination with agents that may improve their bioavailability, reduce and/or modify their metabolism, and/or modify their distribution within the body.
- the pharmaceutical compositions described herein are used as research tools, particularly in in vitro investigations using human cell lines such as HEK293T and in vivo testing in nonhuman primates which will occur prior to human clinical trials.
- the present disclosure provides a method for treating a disease, disorder and/or condition in a mammalian subject, including a human subject, comprising administering to the subject any of the viral particles e.g., AAV, AAV particle, or AAV genome that produces FXN protein described herein (e.g, viral genomes or VG) or administering to the subject a particle comprising said AAV particle or AAV genome, or administering to the subject any of the described compositions, including pharmaceutical compositions.
- the viral particles e.g., AAV, AAV particle, or AAV genome that produces FXN protein described herein (e.g, viral genomes or VG)
- administering to the subject a particle comprising said AAV particle or AAV genome e.g, viral genomes or VG
- the present disclosure provides a method of treating a disorder associated with FXN deficiency such as FA comprising administering an effective amount of an adeno- associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982.
- AAV adeno-associated virus
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides a method of treating a disorder associated with FXN deficiency such as FA comprising administering an effective amount of an adeno- associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738.
- AAV adeno-associated virus
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides a method of treating a disorder associated with FXN deficiency such as FA comprising administering an effective amount of an adeno- associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- AAV particles of the present disclosure through delivery of a functional FXN that is a therapeutic product comprising a FXN protein or variant thereof, can modulate the level or function of FXN in the CNS.
- a functional FXN may alleviate or reduce symptoms that result from abnormal level and/or function of the FXN gene product (e.g., an absence or defect in a FXN protein) in a subject in need thereof or that otherwise confers a benefit to a CNS disorder in a subject in need thereof.
- the FXN gene product e.g., an absence or defect in a FXN protein
- companion or combination therapeutic products delivered by AAV particles of the present disclosure may include, but are not limited to, growth and trophic factors, cytokines, hormones, neurotransmitters, enzymes, anti-apoptotic factors, angiogenic factors, FXN polypeptides, and any protein known to be mutated in pathological disorders such as FA (e.g., brain specific Mir-128a, See Adlakha and Saini, Molecular cancer, 2014, 13:33, incorporated herein by reference in its entirety).
- FA pathological disorders
- the neurodegenerative disorder is Friedreich’s Ataxia, e.g., resulting from expansion of an intronic GAA triplet repeat in the FXN gene, which reduces expression of the mitochondrial protein frataxin causing progressive damage to the nervous system.
- the AAV particles of the present disclosure may be used to ameliorate at least one symptom of FA, including, but not limited to, impaired sensory functions, impaired motor function, e.g., ataxia and/or involuntary movements, fatigue, chronic pain, seizures, impaired speech, sleep disturbances, metabolic disorders, e.g., diabetes, and increased spasticity.
- the delivery of the AAV particles may halt or slow the disease progression of Friedreich’s Ataxia by 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or more than 95% using a known analysis method and comparator group for Friedreich’s Ataxia.
- the delivery of the AAV particles may halt or slow progression of Friedreich’s Ataxia as measured by mFARS/SARA by 50% relative to a comparator group.
- the AAV particle encoding FXN may increase the amount of FXN in a tissue by 1%, 2%, 3%, 4%, 5%, 6%, 7%, 8%, 9%, 10%, 11%, 12%, 13%, 14%, 15%, 16%, 17%, 18%, 19%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 97%, 99%, or more than 100%.
- the AAV particle encoding FXN may increase the amount of FXN in a tissue to be comparable to (e.g., approximately the same as) the amount of FXN in the corresponding tissue of a healthy subject. In some embodiments, the AAV particle encoding FXN may increase the amount of FXN in a tissue effective to reduce one or more symptoms of a disease associated with decreased FXN expression or a deficiency in the quantity and/or function of FXN, e.g., FA. VI.
- the present disclosure provides a method of delivering to a cell or tissue any of the above-described AAV particles, comprising contacting the cell or tissue with said AAV particle or contacting the cell or tissue with a formulation comprising said AAV particle, or contacting the cell or tissue with any of the described compositions, including pharmaceutical compositions.
- the method of delivering the AAV particle to a cell or tissue can be accomplished in vitro, ex vivo, or in vivo.
- Attorney Docket No.14640.0092-00304 [0371]
- the AAV particles are delivered to a cell, tissue, or region of the CNS.
- the AAV particles are delivered to a cell or tissue of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum, or a combination thereof. Delivery to Subjects [0372] In some aspects, the present disclosure additionally provides a method of delivering to a subject, including a mammalian subject, any of the above-described AAV particles comprising administering to the subject said AAV particle, or administering to the subject a formulation comprising said AAV particle, or administering to the subject any of the described compositions, including pharmaceutical compositions.
- the AAV particles may be delivered to bypass anatomical blockages (e.g., the blood brain barrier).
- the AAV particles may be formulated and delivered to a subject by a route which increases the speed of drug effect as compared to oral delivery.
- the AAV particles may be delivered using intrathecal infusion.
- a subject may be administered the AAV particles described herein using a bolus infusion.
- the AAV particles encoding FXN may be delivered in a continuous and/or bolus infusion.
- a different dosing regimen may be used for different sites of delivery or the same dosing regimen may be used for each site of delivery.
- the sites of delivery may be in the cervical and the lumbar region.
- the sites of delivery may be in the cervical region.
- the sites of delivery may be in the lumbar region.
- the AAV particles may be delivered to a subject via a single route of administration.
- the AAV particles may be delivered to a subject via a multi-site route of administration. For example, a subject may be administered the AAV particles at 2, 3, 4, 5, or more than 5 sites.
- a subject may be administered the AAV particles described herein using sustained delivery over a period of minutes, hours or days.
- the infusion rate may be changed depending on the subject, distribution, formulation or another delivery parameter known to those in the art.
- the continuous infusion may be for 1 hour, 2, hours, 3 hours, 4 hours, 5 hours, 6 hours, 7 hours, 8 hours, 9 hours, 10 hours, 11 hours, 12 hours, 13 hours, 14 hours, 15 hours, 16 hours, 17 hours, 18 hours, 19 hours, 20 hours, 21 hours, 22 hours, 23 hours, 24 hours, or more than 24 hours.
- the intracranial pressure may be evaluated prior to administration.
- the route, volume, AAV particle concentration, infusion duration and/or vector titer may be optimized based on the intracranial pressure of a subject.
- the AAV particles may be delivered by systemic delivery.
- the systemic delivery may be by intravascular administration.
- the AAV particles may be delivered by injection into the CSF pathway. Non-limiting examples of delivery to the CSF pathway include intrathecal and intracerebroventricular administration.
- the AAV particles may be delivered by direct (intraparenchymal) injection into the substance of an organ, e.g., one or more regions of the brain.
- the AAV particles may be delivered by subpial injection into the spinal cord.
- subjects may be placed into a spinal immobilization apparatus.
- a dorsal laminectomy may be performed to expose the spinal cord.
- Guiding tubes and XYZ manipulators may be used to assist catheter placement.
- Subpial catheters may be placed into the subpial space by advancing the catheter from the guiding tube and AAV particles may be injected through the catheter (Miyanohara et al., Mol Ther Methods Clin Dev.2016; 3: 16046).
- the AAV particles may be injected into the cervical subpial space. In some cases, the AAV particles may be injected into the thoracic subpial space. [0387] In some embodiments, the present disclosure provides delivery by intravenous injection of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- AAV adeno-associated virus
- the present disclosure provides delivery by intravenous injection of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the present disclosure provides delivery by intravenous injection of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738.
- the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
- the AAV particles may be delivered by direct injection to the CNS of a subject.
- direct injection is intracerebral injection, intraparenchymal injection, intrathecal injection, intra-cisterna magna injection, or any combination thereof.
- direct injection to the CNS of a subject comprises convection enhanced delivery (CED).
- CED convection enhanced delivery
- administration comprises peripheral injection.
- peripheral injection is intravenous injection.
- the AAV particles may be delivered to a subject in order to increase a FXN protein level in the CNS (e.g., the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) as compared to a baseline level in the subject.
- a FXN protein level in the CNS e.g., the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum
- the increase may be 0.1x to 5x, 0.5x to 5x, 1x to 5x, 2x to 5x, 3x to 5x, 4x to 5x, 0.1x to 4x, 0.5x to 4x, 1x to 4x, 2x to 4x, 3x to 4x, 0.1x to 3x, 0.5x to 3x, 1x to 3x, 2x to 3x, 0.1x to 2x, 0.5x to 2x, 0.1x to 1x, 0.5x to 1x, 0.1x to 0.5x, 1x to 2x, 0.1x, 0.2x, 0.3x, 0.4x, 0.5x, 0.6x, 0.7x, 0.8x, 0.9x, 1.0x, 1.1x, 1.2x, 1.3x, 1.4x, 1.5x, 1.6x, 1.7x, 1.8x, 1.9x, 2.0x, 2.1x, 2.2x, 2.3x, 2.4x, 2.5x, 2.6x, 2.7x, 2.8x, 2.9x
- the increase may be 0.5x-3x as compared to a baseline level.
- the AAV particles may be delivered to a subject in order to increase a FXN protein level in the CNS (e.g., the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) by transducing cells in these CNS regions. Transduction may also be referred to as the number of cells that are positive for FXN protein.
- the transduction may be greater than or equal to 1%, greater than or equal to 5%, greater than or equal to 10%, greater than or equal to 15%, greater than or equal to 20%, greater than or equal to 25%, greater than or equal to 30%, greater than or equal to 35%, greater than or equal to 40%, greater than or equal to 45%, greater than or equal to 50%, greater than or equal to 55%, greater than or equal to 60%, greater than or equal to 65%, greater than or equal to 70%, greater than or equal to 75%, greater than or equal to 80%, greater than or equal to 85%, greater than or equal to 90%, greater than or equal to 95%, or greater than or equal to 99% of cells in these CNS regions.
- delivery of AAV particles comprising a viral genome encoding FXN as described herein to neurons in the brain may lead to an increased expression of FXN protein in one or more of those neurons.
- the increased FXN protein expression may lead to improved survival and/or function of various cell types in these CNS regions and/or improvement of at least one symptom of Friedreich’s Ataxia.
- the AAV particles may be delivered to a subject in order to establish widespread distribution of the FXN throughout the CNS by administering the AAV particles to the thalamus of the subject.
- Attorney Docket No.14640.0092-00304 [0395]
- the increased expression of FXN protein may lead to improved gait, improved ataxia-associated heart conditions, decreased feeling of exhaustion, improved symptoms relating to metabolic disorders such as diabetes, and/or improved quality of life.
- the present disclosure provides methods comprising administering AAV particles in accordance with the disclosure to a subject in need thereof.
- AAV particle pharmaceutical, diagnostic, or prophylactic compositions thereof may be administered to a subject using any amount and any route of administration effective for treating, or diagnosing a disease, disorder, and/or condition associated with decreased FXN expression or FXN deficiency.
- the disease, disorder, and/or condition is FA.
- the specific therapeutically effective, prophylactically effective, or appropriate imaging dose level for any particular patient will depend upon a variety of factors including the disorder being treated and the severity of the disorder; the activity of the specific compound employed; the specific composition employed; the age, body weight, general health, sex, and diet of the patient; the time of administration, route of administration, and rate of excretion of the specific FXN employed; the duration of the treatment; drugs used in combination or coincidental with the specific compound employed; and like factors well known in the medical arts.
- the desired dosage may be delivered using multiple administrations (e.g., two, three, four, five, six, seven, eight, nine, ten, eleven, twelve, thirteen, fourteen, or more administrations).
- split dosing regimens such as those described herein may be used.
- a “split dose” is the division of single unit dose or total daily dose into two or more doses, e.g., two or more administrations of the single unit dose.
- a “single unit dose” is a dose of any therapeutic composition administered in one dose/at one time/single route/single point of contact, i.e., single administration event.
- a single unit dose is provided as a discrete dosage form (e.g., a tablet, capsule, patch, loaded syringe, vial, etc.).
- a “total daily dose” is an amount given or prescribed in 24-hour period. It may be administered as a single unit dose.
- the AAV particles may be formulated in buffer only or in a formulation described herein.
- a pharmaceutical composition described herein can be formulated into a topical, intranasal, pulmonary, intratracheal, or injectable dosage form.
- a pharmaceutical composition described herein can be formulated in a dosage form suitable for intravenous, intraocular, intravitreal, intramuscular, intracardiac, intraperitoneal, and/or subcutaneous administration.
- AAV particles described herein results in minimal serious adverse events (SAEs) as a result of the delivery of the AAV particles.
- SAEs minimal serious adverse events
- the AAV particles may be used in combination with one or more other therapeutic, prophylactic, diagnostic, or imaging agents.
- the phrase “in combination with,” is not intended to require that the agents must be administered at the same time and/or formulated for delivery together, although these methods of delivery are within the scope of the present disclosure.
- Compositions can be administered concurrently with, prior to, or subsequent to, one or more other desired therapeutics or medical procedures. In general, each agent will be administered at a dose and/or on a time schedule determined for that agent.
- the present disclosure encompasses the delivery of pharmaceutical, prophylactic, diagnostic, or imaging compositions in combination with agents that may improve their bioavailability, reduce and/or modify their metabolism, and/or modify their distribution within the body.
- the therapeutic agents may be approved by the US Food and Drug Administration or may be in clinical trial or at the preclinical research stage.
- the therapeutic agents may utilize any therapeutic modality known in the art, with non-limiting examples including gene silencing or interference (i.e., miRNA, siRNA, RNAi, shRNA), gene editing (e.g., TALEN, CRISPR/Cas9 systems, zinc finger nucleases), and gene, protein, or enzyme replacement.
- an AAV particle described herein, or a pharmaceutical composition comprising the AAV particle may be administered in combination with at least one additional therapeutic agent and/or therapy.
- the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy for treating the disorder associated with FXN deficiency (e.g., Friedreich’s Ataxia).
- the at least one additional therapeutic agent and/or therapy comprises omaveloxolone or idebenone.
- the at least one additional therapeutic agent and/or therapy comprises CoQ10, IFN gamma, or a drug to treat comorbidities including diabetes, heart disease, pain.
- the at least one additional therapeutic agent and/or therapy comprises an immunosuppressant.
- the immunosuppressant may be administered to the subject prior to administration of an AAV particle or pharmaceutical composition described herein.
- the immunosuppressant may be administered to the subject simultaneously with administration of an AAV particle or pharmaceutical composition described herein.
- the immunosuppressant may be administered to the subject after administration of an AAV particle or pharmaceutical composition described herein.
- the AAV particle or pharmaceutical composition is administered to a subject who is receiving or has received an immunosuppressant.
- the immunosuppressant comprises a corticosteroid (for example, and without limitation, prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine.
- a corticosteroid for example, and without limitation, prednisone, prednisolone, methylprednisolone, and/or dexamethasone
- rapamycin for example, and without limitation, prednisone, prednisolone, methylprednisolone, and/or dexamethasone
- rapamycin for example, and without limitation, prednisone, prednisolone, methylprednisolone, and/or dexamethasone
- rapamycin for example, and without limitation, rapamycin, mycophenol
- Expression of FXN from viral genomes may be determined using various methods known in the art such as, but not limited to immunochemistry (e.g., IHC), enzyme-linked immunosorbent assay (ELISA), affinity ELISA, ELISPOT, flow cytometry, immunocytology, surface plasmon resonance analysis, kinetic exclusion assay, liquid chromatography-mass spectrometry (LCMS), high-performance liquid chromatography (HPLC), BCA assay, immunoelectrophoresis, Western blot, SDS-PAGE, protein immunoprecipitation, PCR, and/or in situ hybridization (ISH).
- immunochemistry e.g., IHC
- ELISA enzyme-linked immunosorbent assay
- affinity ELISA affinity ELISA
- ELISPOT enzyme-linked immunosorbent assay
- flow cytometry immunocytology
- surface plasmon resonance analysis e.g., surface plasmon resonance analysis
- kinetic exclusion assay e.g., kinetic exclusion assay
- transgenes encoding FXN delivered in different AAV capsids may have different expression levels in dorsal root ganglion (DRG).
- the FXN protein is detectable by an enzyme-linked immunosorbent assay (ELISA).
- ELISA enzyme-linked immunosorbent assay
- the FXN protein is detectable by an immunohistochemistry assay.
- the FXN protein is detectable by Western blot.
- the FXN protein is detectable by a frataxin biofluid assay, such as the assay described in PCT/US2020/045687, the contents of which are hereby incorporated in their entirety.
- a FXN gene, mRNA, and/or protein expression is measured in a cell or tissue of a subject who is receiving or has received an AAV particle described herein.
- the FXN gene, mRNA, and/or protein expression is measured in a cell or tissue of the CNS, such as the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum.
- kits for conveniently and/or effectively carrying out methods of the present disclosure. Typically, kits will comprise sufficient amounts and/or numbers of components to allow a user to perform multiple treatments of a subject(s) and/or to perform multiple experiments.
- Any of the vectors, constructs, or FXN of the present disclosure may be comprised in a kit. In some embodiments, kits may further include reagents and/or instructions for creating and/or synthesizing compounds and/or compositions of the present disclosure.
- kits may also include one or more buffers.
- kits of the disclosure may include components for making protein or nucleic acid arrays or libraries and thus, may include, for example, solid supports.
- kit components may be packaged either in aqueous media or in lyophilized form.
- the container means of the kits will generally include at least one vial, test tube, flask, bottle, syringe or other container means, into which a component may be placed, and suitably aliquoted. Where there is more than one kit component, (labeling reagent and label may be packaged together), kits Attorney Docket No.14640.0092-00304 may also generally contain second, third or other additional containers into which additional components may be separately placed.
- kits may also comprise second container means for containing sterile, pharmaceutically acceptable buffers and/or other diluents.
- various combinations of components may be comprised in one or more vial.
- Kits of the present disclosure may also typically include means for containing compounds and/or compositions of the present disclosure, e.g., proteins, nucleic acids, and any other reagent containers in close confinement for commercial sale. Such containers may include injection or blow-molded plastic containers into which desired vials are retained.
- kit components are provided in one and/or more liquid solutions.
- liquid solutions are aqueous solutions, with sterile aqueous solutions being particularly used.
- kit components may be provided as dried powder(s).
- reagents and/or components are provided as dry powders, such powders may be reconstituted by the addition of suitable volumes of solvent.
- solvents may also be provided in another container means.
- labeling dyes are provided as dried powders.
- 10, 20, 30, 40, 50, 60, 70, 80, 90, 100, 120, 120, 130, 140, 150, 160, 170, 180, 190, 200, 300, 400, 500, 600, 700, 800, 900, 1000 micrograms or at least or at most those amounts of dried dye are provided in kits of the disclosure.
- kits may include instructions for employing kit components as well the use of any other reagent not included in the kit. Instructions may include variations that may be implemented.
- Devices [0416] In some embodiments, compounds and/or compositions of the present disclosure may be combined with, coated onto or embedded in a device. Devices may include, but are not limited to, dental implants, stents, bone replacements, artificial joints, valves, pacemakers and/or other implantable therapeutic device. [0417] The present disclosure provides for devices which may incorporate viral vectors that encode one or more FXN molecules.
- devices contain in a stable formulation the viral vectors which may be immediately delivered to a subject in need thereof, such as a human patient.
- Devices for administration may be employed to deliver the viral vectors encoding FXN of the present disclosure according to single, multi- or split-dosing regimens taught herein.
- Method and devices known in the art for multi-administration to cells, organs and tissues are contemplated for use in conjunction with the methods and compositions disclosed herein as embodiments of the present disclosure. VIII. Definitions [0420] At various places in the present specification, substituents of compounds of the present disclosure are disclosed in groups or in ranges.
- Claims or descriptions that include “or” between one or more members of a group are considered satisfied if one, more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process unless indicated to the contrary or otherwise evident from the context.
- the disclosure includes embodiments in which exactly one member of the group is present in, employed in, or otherwise relevant to a given product or process.
- the disclosure includes embodiments in which more than one, or the entire group members are present in, employed in, or otherwise relevant to a given product or process.
- the term “comprising” is intended to be open and permits but does not require the inclusion of additional elements or steps. [0424] Where ranges are given, endpoints are included.
- Adeno-associated virus As used herein, the term “adeno-associated virus” or “AAV” refers to members of the dependovirus genus or a functional variant thereof. Unless stated otherwise, “AAV” may refer to wildtype (i.e., naturally occurring) AAV or recombinant AAV.
- AAV Particle refers to a particle comprising an AAV capsid, e.g., an AAV capsid variant (such as a parent capsid sequence with at least one peptide insert and/or with at least one substitution), and a polynucleotide, e.g., a viral genome or a vector genome.
- the AAV particle may be capable of delivering a FXN polynucleotide to cells.
- the cells may be mammalian cells, e.g., human cells.
- an AAV particle of the present disclosure may be produced recombinantly.
- an AAV particle may be derived from any serotype, described herein or known in the art, including combinations of serotypes (e.g., “pseudotyped” AAV) or from various genomes (e.g., single stranded or self-complementary).
- the AAV particle may be replication defective and/or targeted.
- the AAV particle may comprise a peptide present in, e.g., inserted into and/or replacing a wildtype amino acid of, the capsid to enhance tropism for a desired target tissue. It is to be understood that reference to the AAV particle of the disclosure also includes pharmaceutical compositions thereof, even if not explicitly recited.
- Administering refers to providing a pharmaceutical agent or composition to a subject.
- Amelioration refers to a lessening of severity of at least one indicator of a condition or disease. For example, in the context of a neurodegenerative disorder, amelioration includes the reduction or stabilization of neuron loss.
- Approximately As used herein, the term “approximately” or “about,” as applied to one or more values of interest, refers to a value that is similar to, i.e., within 10% of, a stated reference value.
- Baseline The term “baseline,” when used to describe a measurement in a subject receiving or about to receive a treatment, refers to a measurement made before starting the treatment.
- Capsid refers to the exterior, e.g., a protein shell, of a virus particle, e.g., an AAV particle, that is substantially (e.g., >50%, >60%, >70%, >80%, >90%, >95%, >99%, or 100%) protein.
- the capsid is an AAV capsid comprising an AAV capsid protein described herein, e.g., a VP1, VP2, and/or VP3 polypeptide.
- the AAV capsid protein can be a wild-type AAV capsid protein or a variant, e.g., a structural and/or functional variant from a wild-type or a reference capsid protein, referred to herein as an “AAV capsid variant.”
- an AAV capsid variant may refer to at least a VP1 protein, a VP2 protein, or a VP3 protein (e.g., all of the VP1, VP2, and VP3 proteins forming the AAV capsid) as will be clear from context.
- the AAV capsid variant described herein may comprise a peptide insertion and/or substitution (i.e., replacement).
- the AAV capsid variant described herein has the ability to encapsulate a viral genome and/or is capable of entry into a cell, e.g., a mammalian cell.
- the AAV capsid variant described herein may have modified tropism compared to that of a wild-type AAV capsid, e.g., the corresponding wild-type capsid.
- Cis-Elements As used herein, cis-elements or the synonymous term “cis-regulatory elements” refer to regions of non-coding DNA which regulate the transcription of nearby genes. The Latin prefix “cis” translates to “on this side.” Cis-elements are found in the vicinity of the gene, or genes, they regulate. Examples of cis-elements include a Kozak sequence, SV40 introns, or a portion of the backbone.
- CNS structures As used herein, “CNS structures” refers to structures of the central nervous system and sub-structures thereof.
- Non-limiting examples of structures in the spinal cord may include ventral horn, dorsal horn, white matter, and nervous system pathways or nuclei within.
- Non-limiting examples of structures in the brain include, forebrain, midbrain, hindbrain, diencephalon, telencephalon, myelencephalon, metencephalon, mesencephalon, prosencephalon, rhombencephalon, cortices, frontal lobe, parietal lobe, temporal lobe, occipital lobe, cerebrum, thalamus, hypothalamus, tectum, tegmentum, cerebellum, pons, medulla, amygdala, hippocampus, basal ganglia, corpus callosum, pituitary gland, putamen, striatum, ventricles and sub-structures thereof.
- CNS cells refers to cells of the central nervous system and sub- structures thereof.
- Non-limiting examples of CNS cells include neurons and sub-types thereof, glia, microglia, oligodendrocytes, ependymal cells and astrocytes.
- Non-limiting examples of neurons include sensory neurons, motor neurons, interneurons, unipolar cells, bipolar cells, multipolar cells, Attorney Docket No.14640.0092-00304 pseudounipolar cells, pyramidal cells, basket cells, stellate cells, Purkinje cells, Betz cells, amacrine cells, granule cell, ovoid cell, medium aspiny neurons, large aspiny neurons, GABAergic neurons, and/or glutamatergic neurons.
- Codon optimization refers to a process of changing codons of a given gene in such a manner that the polypeptide sequence encoded by the gene remains the same.
- Complementary and substantially complementary refers to the ability of polynucleotides to form base pairs with one another. Perfect complementarity or 100% complementarity refers to the situation in which each nucleotide unit of one polynucleotide strand can form a hydrogen bond with a nucleotide unit of a second polynucleotide strand. Less than perfect complementarity refers to the situation in which some, but not all, nucleotide units of two strands can form hydrogen bond with each other. For example, for two 20-mers, if only two base pairs on each strand can form a hydrogen bond with each other, the polynucleotide strands exhibit 10% complementarity.
- the polynucleotide strands exhibit 90% complementarity.
- complementary as used herein can encompass fully complementary or partially (e.g., substantially) complementary. “Fully complementary”, “perfect complementarity”, or “100% complementarity” refers to the situation in which each nucleotide unit of one polynucleotide or oligonucleotide strand can base-pair with a nucleotide unit of a second polynucleotide or oligonucleotide strand.
- the term “substantially complementary” means that >50% of the nucleotide units of a first polynucleotide strand can base pair with nucleotide units on a second polynucleotide strand.
- “substantially complementary” refers to an siRNA that has a sequence (e.g., in the antisense strand) that is sufficient to bind the desired target mRNA and to trigger the RNA silencing of the target mRNA.
- a conservative substitution as applied to an amino acid sequence, also referred to as a “conservative amino acid substitution,” is one in which the amino acid residue is replaced with an amino acid residue having similar biochemical properties.
- conservative substitution refers to a nucleotide replacement that results in an amino acid residue having similar biochemical properties compared to a reference sequence. Families of amino acid residues having similar biochemical properties have been defined in the art.
- amino acids with basic side chains e.g., lysine, arginine, histidine
- acidic side chains e.g., aspartic acid, glutamic acid
- uncharged polar side chains e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine
- nonpolar side chains e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine, tryptophan
- beta-branched side chains e.g., threonine, valine, isoleucine
- aromatic side chains e.g., tyrosine, phenylalanine, tryptophan, histidine
- an amino acid corresponding to position 577 of SEQ ID NO: 138 refers to the amino acid at the 577th position from the N-terminus in SEQ ID NO: 138 or the equivalent position in an aligned sequence.
- an amino acid at a position corresponding to that in a designated sequence may also be referred to as an amino acid at a particular position, numbered according or numbered relative to the designated sequence.
- an amino acid corresponding to position 577 of SEQ ID NO: 138 may also be referred to as an amino acid at position 577, numbered according to SEQ ID NO: 138 or as numbered according to a sequence corresponding to SEQ ID NO: 138 (i.e., relative to a reference sequence of SEQ ID NO: 138).
- a peptide e.g., the peptide [N2]-[N3]
- Derivative refers to a composition (e.g., sequence, compound, formulation, etc.) that is derived from, or finds its basis in, a parent composition.
- a parent composition include a wild-type or original amino acid or nucleic acid sequence, or an undiluted formulation.
- a derivative is a variant of a parent composition.
- a derivative may differ from the parent composition by less than about 1%, less than about 5%, less than about 10%, less than about 15%, less than about 20%, less than about 25%, less than about 30%, less than about 35%, less than about 40%, less than about 45%, or less than about 50%.
- a derivative may differ from a parent composition by more than about 50%. In certain embodiments, a derivative may differ from a parent composition by more than about 75%. In some embodiments, a derivative may be a fragment or truncation of a parent amino acid or nucleotide sequence. As a non-limiting example, a derivative may be a sequence with a nucleotide, amino acid, or peptide substitution and/or insertion as compared to a parent nucleic acid or amino acid sequence (e.g., as compared to AAV5 or AAV9). [0440] Effective amount: As used herein, the term “effective amount” or “therapeutically effective amount” of an agent is that amount sufficient to effect beneficial or desired results.
- Excipient refers to an inactive substance that serves as the vehicle or medium for an active pharmaceutical agent or other active substance.
- Formulation includes at least one active ingredient (e.g., an AAV particle) and at least one inactive ingredient (e.g., a pharmaceutically acceptable excipient).
- Fragment A “fragment,” as used herein, refers to a contiguous portion of a reference sequence.
- a fragment may comprise a functional fragment that retains at least one activity of the reference sequence.
- fragments of proteins may comprise polypeptides obtained by digesting full-length protein isolated from cultured cells.
- a fragment may also refer to a truncation (e.g., an N- terminal and/or C-terminal truncation) of a protein or a truncation (e.g., at the 5’ and/or 3’ end) of a Attorney Docket No.14640.0092-00304 nucleic acid.
- a protein fragment may be obtained by expression of a truncated nucleic acid, such that the nucleic acid encodes a portion of the full-length protein.
- Healthy individual As used herein, the term “healthy individual” refers to an individual who does not have a disease or disorder associated with FXN protein deficiency, e.g., an individual who does not have Friedreich's Ataxia.
- Identity As used herein, the term “identity” refers to the overall relatedness between polymeric molecules, e.g., between oligonucleotide molecules (e.g., DNA molecules and/or RNA molecules) and/or between polypeptide molecules.
- Calculation of the percent identity of two polynucleotide sequences may be performed by aligning the two sequences for optimal comparison purposes (e.g., gaps can be introduced in one or both of a first and a second nucleic acid sequences for optimal alignment and non-identical sequences can be disregarded for comparison purposes).
- the nucleotides at corresponding nucleotide positions are then compared. When a position in the first sequence is occupied by the same nucleotide as the corresponding position in the second sequence, then the molecules are identical at that position.
- the percent identity between the two sequences is a function of the number of identical positions shared by the sequences, taking into account the number of gaps, and the length of each gap, which needs to be introduced for optimal alignment of the two sequences.
- the comparison of sequences and determination of percent identity between two sequences can be accomplished using a mathematical algorithm.
- the percent identity between two nucleotide sequences can be determined using methods such as those described in Computational Molecular Biology, Lesk, A. M., ed., Oxford University Press, New York, 1988; Biocomputing: Informatics and Genome Projects, Smith, D. W., ed., Academic Press, New York, 1993; Sequence Analysis in Molecular Biology, von Heinje, G., Academic Press, 1987; Computer Analysis of Sequence Data, Part I, Griffin, A. M., and Griffin, H. G., eds., Humana Press, New Jersey, 1994; and Sequence Analysis Primer, Gribskov, M.
- the percent identity between two nucleotide sequences can be determined using the algorithm of Myers and Miller (CABIOS, 1989, 4:11-17), which has been incorporated into the ALIGN program (version 2.0) using a PAM120 weight residue table, a gap length penalty of 12 and a gap penalty of 4.
- the percent identity between two nucleotide sequences can, alternatively, be determined using the GAP program in the GCG software package using an NWSgapdna.CMP matrix.
- Methods commonly employed to determine percent identity between sequences include, but are not limited to those disclosed in Carillo, H., and Lipman, D., SIAM J Applied Math., 48:1073 (1988); incorporated herein by reference in its entirety. Techniques for determining identity are codified in publicly available computer programs. Computer software to determine homology between two sequences include, but are not limited to, GCG program package, Devereux, J., et al., Nucleic Acids Research, 12(1), 387 (1984)), the Basic Local Alignment Search Tool (BLAST, which includes, e.g., BLASTP for protein sequences and BLASTN for nucleic acid sequences), and FASTA Altschul, S. F. et al., J.
- BLAST Basic Local Alignment Search Tool
- inverted terminal repeat As used herein, the term “inverted terminal repeat” or “ITR” refers to a cis-regulatory element for the packaging of polynucleotide sequences into viral capsids.
- Isolated refers to a substance or entity that is altered or removed from the natural state, e.g., altered or removed from at least some of component with which it is associated in the natural state.
- a nucleic acid or a peptide naturally present in a living animal is not “isolated,” but the same nucleic acid or peptide partially or completely separated from the coexisting materials of its natural state is “isolated.”
- An isolated nucleic acid or protein can exist in substantially purified form, or can exist in a non-native environment such as, for example, a host cell.
- miRNA binding site refers either to a DNA sequence corresponding to an RNA sequence that is bound by a microRNA, or to the RNA sequence that is bound by the microRNA.
- the miR binding site is capable of binding, or binds, in whole or in part to a microRNA (miRNA, miR) through complete or partial hybridization.
- a miR binding site may be encoded or transcribed in series, also referred to as a “miR binding site series” or “miR BSs”, which includes two or more miR binding sites having the same or a different nucleic acid sequence.
- Modification refers to any substance, compound, or molecule that has been changed in any way.
- a modification in an amino acid sequence may comprise a substitution (e.g., a conservative substitution), an insertion, and/or a deletion of one or more amino acids in the sequence.
- Neurological disease As used herein, a “neurological disease” is any disease associated with the central or peripheral nervous system and components thereof (e.g., neurons).
- Operably linked As used herein, the phrase “operably linked” refers to a functional connection between two or more molecules, constructs, transcripts, entities, moieties or the like.
- Payload As used herein, “payload,” “payload sequence,” or “payload region” refers to one or more polynucleotides or polynucleotide regions encoded by or within a viral genome or an expression product of such polynucleotide or polynucleotide region, e.g., a transgene, a polynucleotide encoding a polypeptide.
- Payload construct As used herein, “payload construct” is one or more polynucleotide regions encoding or comprising a payload that is flanked on one or both sides by an inverted terminal repeat (ITR) sequence.
- ITR inverted terminal repeat
- Payload construct vector is a vector encoding or comprising a payload construct, and regulatory regions for replication and expression in bacterial cells.
- the payload construct vector may also comprise a component for viral expression in a viral replication cell.
- Pharmaceutically acceptable The phrase “pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are suitable for use in contact with the tissues of human beings and animals.
- compositions that can function as vehicles for suspending and/or dissolving active agents.
- pharmaceutically acceptable salts Pharmaceutically acceptable salts of the compounds described herein are forms of the disclosed compounds wherein the acid or base moiety is in its salt form (e.g., as generated by reacting a free base group with a suitable organic acid).
- composition As used herein, the term “pharmaceutical composition” or pharmaceutically acceptable composition” comprises AAV polynucleotides, AAV genomes, or AAV particle and one or more pharmaceutically acceptable excipients, solvents, adjuvants, and/or the like.
- Position The term “position,” as used herein in the context of an amino acid sequence, refers to the location of a particular amino acid or set of amino acids relative to a larger sequence.
- a position or positions of amino acids may interchangeably be referred to by an amino acid number or numbers of a reference sequence.
- positions 1-731, as numbered according to SEQ ID NO: 982 is interchangeable with “amino acids 1-731, as numbered according to SEQ ID NO: 982.”
- amino acids 1-731, as numbered according to SEQ ID NO: 982 is counted from the N-terminus.
- the term “preventing” refers to partially or completely delaying onset of an infection, disease, disorder and/or condition; partially or completely delaying onset of one or more symptoms, features, or clinical manifestations of a particular infection, disease, disorder, and/or condition; partially or completely delaying onset of one or more symptoms, features, or manifestations of a particular infection, disease, disorder, and/or condition; partially or completely delaying progression from an infection, a particular disease, disorder and/or condition; and/or decreasing the risk of developing pathology associated with the infection, the disease, disorder, and/or condition.
- Region refers to a zone or general area. In some embodiments, when referring to a protein or protein module, a region may comprise a linear sequence of amino acids along the protein or protein module or may comprise a three-dimensional area. In some Attorney Docket No.14640.0092-00304 embodiments, regions comprise terminal regions. As used herein, the term “terminal region” refers to regions located at the ends or termini of a given agent.
- terminal regions may comprise N- and/or C-termini.
- N-termini refer to the end of a protein comprising an amino acid with a free amino group.
- C-termini refer to the end of a protein comprising an amino acid with a free carboxyl group.
- N- and/or C-terminal regions may comprise the N- and/or C-termini as well as surrounding amino acids.
- a region may comprise a linear sequence of nucleic acids along the polynucleotide or may comprise a three-dimensional area, secondary structure, or tertiary structure. In some embodiments, regions comprise terminal regions.
- terminal region refers to regions located at the ends or termini of a given agent. When referring to polynucleotides, terminal regions may comprise 5’ and/or 3’ termini.
- sample or “biological sample” refers to a subset of tissues, cells, nucleic acids, or a component or part of the body (e.g., a body fluid, including but not limited to blood, mucus, lymphatic fluid, synovial fluid, cerebrospinal fluid, saliva, amniotic fluid, amniotic cord blood, urine, vaginal fluid, and semen).
- Self-complementary AAV As used herein, the term “self-complementary AAV” refers to an AAV comprising at least a protein capsid and a self-complementary viral genome.
- Serotype As used herein, the term “serotype” refers to distinct variations in a capsid of an AAV based on surface antigens which allow epidemiologic classifications of the AAVs at the sub- species level.
- Silent Mutation As used herein, a “silent mutation” or “silent substitution” refers to a nucleotide replacement that results in the same amino acid residue as a reference sequence.
- Signal Sequence As used herein, the phrase “signal sequence” refers to a sequence which can direct the transport or localization.
- Similarity refers to the overall relatedness between polymeric molecules, e.g., between polynucleotide molecules (e.g., DNA molecules and/or RNA molecules) and/or between polypeptide molecules. Calculation of percent similarity of polymeric molecules to one another can be performed in the same manner as a calculation of percent identity, except that calculation of percent similarity takes into account conservative substitutions as is understood in the art.
- Spacer is generally any selected nucleic acid sequence of, e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 nucleotides in length, which is located between two or more consecutive miR binding site sequences. In some embodiments, spacers may also be more than 10 nucleotides in length, e.g., 20, 30, 40, or 50 or more than 50 nucleotides.
- Subject As used herein, the term “subject” or “patient” refers to any organism to which a composition in accordance with the disclosure may be administered, e.g., for experimental, diagnostic, prophylactic, and/or therapeutic purposes.
- subject or “patient” refers to an organism who may seek, who may require, who is receiving, or who will receive treatment or who is under care by a Attorney Docket No.14640.0092-00304 trained professional for a particular disease or condition.
- Typical subjects include animals (e.g., mammals such as mice, rats, rabbits, non-human primates, and humans).
- a subject or patient may be susceptible to, suspected of having, or have a deficiency in frataxin protein, e.g., may be susceptible to, suspected of having, or have Friedreich’s Ataxia (FA).
- FA Ataxia
- the term “substantially” refers to the qualitative condition of exhibiting total or near-total extent or degree of a characteristic or property of interest.
- One of ordinary skill in the biological arts will understand that biological and chemical phenomena rarely, if ever, go to completion and/or proceed to completeness or achieve or avoid an absolute result. The term “substantially” is therefore used herein to capture the potential lack of completeness inherent in many biological and chemical phenomena.
- Suffering from An individual who is “suffering from” a disease, disorder, and/or condition has been diagnosed with or displays one or more symptoms of a disease, disorder, and/or condition.
- Susceptible to An individual who is “susceptible to” a disease, disorder, and/or condition has not been diagnosed with and/or may not exhibit symptoms of the disease, disorder, and/or condition but harbors a propensity to develop a disease or its symptoms.
- an individual who is susceptible to a disease, disorder, and/or condition may be characterized by one or more of the following: (1) a genetic mutation associated with development of the disease, disorder, and/or condition; (2) a genetic polymorphism associated with development of the disease, disorder, and/or condition; (3) increased and/or decreased expression and/or activity of a protein and/or nucleic acid associated with the disease, disorder, and/or condition; (4) habits and/or lifestyles associated with development of the disease, disorder, and/or condition; (5) a family history of the disease, disorder, and/or condition; and (6) exposure to and/or infection with a microbe associated with development of the disease, disorder, and/or condition.
- Target cells refers to any one or more cells of interest.
- the cells may be found in vitro, in vivo, in situ or in the tissue or organ of an organism.
- the organism may be an animal, preferably a mammal, more preferably a human and most preferably a human patient.
- Target Tissue refers to a tissue of interest that may be found in vitro, in situ, or as part of an animal, preferably a mammal, more preferably a human and most preferably a human patient.
- Therapeutic Agent refers to any agent that, when administered to a subject, elicits a desired biological and/or pharmacological effect.
- therapeutically effective outcome means an outcome that is sufficient in a subject suffering from or susceptible to an infection, Attorney Docket No.14640.0092-00304 disease, disorder, and/or condition, to treat, improve symptoms of, delay progression of symptoms, diagnose, prevent, and/or delay the onset of the infection, disease, disorder, and/or condition.
- Treating refers to partially or completely alleviating, ameliorating, improving, relieving, delaying onset of, inhibiting progression of, reducing severity of, reducing incidence of, and/or preventing one or more symptoms or features of a particular infection, disease, disorder, and/or condition.
- Truncated CBA promoter As used herein, a "truncated CBA promoter" refers to a CBA promoter that is shorter in length than the nucleotide sequence of SEQ ID NO: 1734, provided below.
- Truncated CMV promoter refers to a CMV promoter that is shorter in length than the nucleotide sequence of SEQ ID NO: 1743, as provided below.
- Unmodified may, but does not always, refer to the wild-type or native form of a biomolecule or entity. Molecules or entities may undergo a series of modifications whereby each modified product may serve as the “unmodified” starting molecule or entity for a subsequent modification.
- Variant refers to a polypeptide or polynucleotide that has an amino acid or a nucleotide sequence that has at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, Attorney Docket No.14640.0092-00304 at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to a reference sequence.
- the variant may be a functional variant.
- the term “functional variant” refers to a polypeptide variant or a polynucleotide variant that has at least one activity of the reference sequence.
- Vector As used herein, a “vector” is any molecule or moiety which transports, transduces, or otherwise acts as a carrier of a heterologous molecule. Vectors of the present disclosure may be produced recombinantly and may be based on and/or may comprise adeno-associated virus (AAV) parent or reference sequence(s).
- AAV adeno-associated virus
- Viral genome As used herein, a “viral genome,” “vector genome,” or “VG” is a polynucleotide comprising at least one inverted terminal repeat (ITR) and at least one nucleic acid sequence encoding a payload. A viral genome encodes at least one copy of the payload.
- ITR inverted terminal repeat
- EXAMPLES The present disclosure is further illustrated by the following non-limiting examples. Example 1.
- the initial library was passed three times through non-human primates (NHP), specifically cynomolgus macaques (Macaca fascicularis), rats, or human brain microvascular endothelial cells (hBMVECs). Following the third passage in each system, 572 variants from the NHPs, 80 variants from the rats, and 99 variants from the hBMVECs were pooled into a passage 3 synthetic library of 747 total variants. This library was then passaged in NHPs and rats. After this passage (e.g., one-month post injection into two NHPs and the rats), RNA was extracted from three brain regions.
- NHP non-human primates
- hBMVECs human brain microvascular endothelial cells
- the variant comprising YPAEVVQK (SEQ ID NO: 943) demonstrated a 64.9-fold enrichment in the brain of NHPs.
- Attorney Docket No.14640.0092-00304 [0487] As shown in Table 14, approximately 98 variants were identified with an average fold change greater than wild-type AAV5 in the brain of rats.
- 33 demonstrated a fold-change of greater than 2 compared to wild-type, with one variant demonstrating a fold change of greater than 40.
- the variant comprising YPAEVVQK demonstrated a 41.1-fold enrichment in the brain of rats.
- the variant comprising YPAEVVQK (SEQ ID NO: 943) which demonstrated a high fold enrichment in the brains of NHPs relative to wild-type AAV5 (64.9-fold enrichment), also demonstrated a high fold-change in the brains of rats (41.1-fold enrichment).
- This indicates that this AAV capsid variant comprising SEQ ID NO: 943 is able to cross species, as evidenced by increased expression and tropism in both the NHP and rat brain. Table 14.
- Example 2 Individual capsid characterization in NHPs, rats, and mice [0490] This Example describes the transduction level, tropism, ability to cross the blood brain barrier, and overall spatial distribution in the central nervous system (CNS) of an AAV capsid variant selected from the study described in Example 1, relative to wild-type AAV5, following intravenous injection in cynomolgus macaques (Macaca fascicularis), Norway rats, and BALB/c mice.
- CNS central nervous system
- the capsid variant was TTN-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 943 (encoded by SEQ ID NO: 944)), as outlined in Table 3 above.
- the amino acid and DNA sequence of TTN-002 is provided, e.g., in Tables 4A and 4B respectively.
- AAV particles were generated with this capsid variant encapsulating a luciferase-EGFP transgene or a cynomolgus monkey frataxin payload fused to a hemagglutinin (HA) tag driven by a chicken beta actin (CBA) heterologous promoter.
- HA hemagglutinin
- CBA chicken beta actin
- the in-life period was 28 days for NHPs and mice, and 25 days for rats.
- Various CNS and peripheral tissues were then collected for measuring transgene mRNA, transgene protein, and/or viral DNA (biodistribution).
- the AAV particles administered to the NHPs and rats comprised self-complementary viral genomes and the AAV particles administered to mice comprised a single-stranded viral genome.
- Individual capsid characterization in NHPs [0492] The brains, spinal cord, and peripheral tissues including the heart, liver, and quadriceps, were isolated from NHPs (cynomolgus macaques (Macaca fascicularis)) at 28 days post intravenous administration of the AAV particles comprising the TTN-002 capsid variant and were assayed by qPCR for the presence of transgene RNA as a measure of transgene expression and compared to the AAV9 control.
- TTN-002 capsid variant which is an AAV5 capsid variant
- mRNA transgene expression from the TTN-002 capsid variant was significantly higher in the brain of NHPs relative to the wild-type AAV9 control. More specifically, mRNA expression was approximately 20-25-fold higher from the TTN-002 capsid variant compared to wild-type AAV9 in the brain of NHPs. Additionally, mRNA expression was approximately 4-5-fold higher from the TTN-002 capsid variants compared to wild-type AAV9 in the spinal cord of the NHPs.
- TTN-002 also demonstrated lower mRNA expression in the liver and DRG relative the AAV9 control (Table 15).
- the brains, spinal cord, and peripheral tissues isolated from the NHPs were also assayed for the presence of viral DNA as a measure of viral genome levels. Data are provided in Table 16 as average DNA (viral genome (VG)) copies per diploid genome as well as fold change relative to the AAV9 control.
- VG viral genome
- biodistribution of the AAV5 capsid variant, TTN-002 was significantly higher in the NHP brain relative to the wild-type AAV9 control. Biodistribution of TTN-002 was lower in the NHP liver relative to the wild-type AAV9 control.
- the brain tissues and spinal cords of the NHPs were also subjected to immunohistochemistry staining to evaluate overall CNS tropism and biodistribution in various regions (FIGs.1A-1D). Immunohistochemical staining correlated with the qPCR analysis, as TTN-002 showed significantly stronger staining and payload expression in the brain (e.g., across the entire cerebrum and cerebellum, FIGs.1A-1C) and spinal cord (FIGs.1A and 1D), as compared to the AAV9 control.
- TTN-002 demonstrated localization, strong payload expression, and transduction in both neurons and glial cells in the temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and cerebellum (FIGs.1A-1C). Payload expression was also observed in astrocytes in the dentate nucleus.
- TTN-002 demonstrated localization, strong payload expression, and transduction in the cervical region (e.g., C2), thoracic region (e.g., T10), and lumbar region (e.g., L2) (FIGs.1A and 1D).
- TTN-002 showed less staining in the DRG relative to the wild-type AAV9 control (approximately 2-fold less) (FIG.1D). Both TTN-002 and AAV9 appeared to transduce the liver and heart with similarly high efficiency, by IHC analysis (FIG.1D). Attorney Docket No.14640.0092-00304 Additionally, the histopathology of these samples isolated from the NHPs showed no signs of toxicity in the NHPs, following intravenous administration of AAV particles comprising the TTN-002 capsid variant at a dose of 5e13 VG/kg with a self-complementary viral genome. Table 15. Transgene mRNA expression with the TTN-002 capsid variant in NHPs Table 16.
- mice Viral DNA biodistribution with the TTN-002 capsid variant in NHPs Attorney Docket No.14640.0092-00304 B.
- Individual capsid characterization in rats [0495] The brains and spinal cords were isolated from rats at 25 days post intravenous administration of AAV particles comprising the TTN-002 capsid variant (AAV5 variant) and assayed for the presence of transgene RNA as a measure of transgene expression, relative to a wild-type AAV5 control capsid or a wild-type AAV9 control capsid (Table 17). Data were provided as average mRNA fold change for the transgene relative to a housekeeping gene as well as the fold change relative to the AAV5 and AAV9 controls (Table 17).
- transgene expression from the TTN-002 capsid variant was higher in both the brains and spinal cords relative to the AAV5 and AAV9 controls. More specifically, transgene mRNA expression was approximately 40- to 67-fold higher from the TTN-002 variant in the rat brain and spinal cord regions (cervical, thoracic, and lumbar) compared to wild-type AAV5 and transgene mRNA expression was approximately 5-7-fold higher in the rat brain and spinal cord regions (cervical, thoracic and lumbar) compared to wild-type AAV9.
- TTN-002 showed increased staining relative to both AAV9 and AAV5 in the cortex, hippocampus, cerebellum, and spinal cord of the rat.
- TTN-002 showed increased staining in the heart of the rat relative to AAV5 but decreased staining relative to AAV9.
- Table 17 Transgene mRNA expression with TTN-002 capsid variant in rats C.
- mice Individual capsid characterization in mice [0497] The brains and livers were isolated from BALB/c mice 28 days post intravenous injection of following intravenous administration of AAV particles comprising the TTN-002 capsid variant and were assayed by qPCR for the presence of transgene RNA as a measure of transgene expression and compared to an AAV9 and AAV5 control. Data were provided as average mRNA fold change for the transgene Attorney Docket No.14640.0092-00304 relative to a housekeeping gene (Table 18) and as fold change in transgene mRNA expression relative AAV9 and AAV5 controls (Table 19).
- the AAV5 capsid variant TTN-002 demonstrated similar levels of transgene expression relative to AAV9 in the brain and higher expression than wild-type AAV5.
- Transgene mRNA expression in the mouse brain was 265.9-fold higher with the TTN-002 capsid variant as compared to wild-type AAV5 (Table 19).
- wild-type AAV5 and the AAV5 capsid variant, TTN-002 both resulted in lower transgene expression in the liver, as compared to wild-type AAV9.
- the brains and livers isolated from the mice were also assayed for the presence of viral DNA as a measure of viral genome levels.
- Table 18 Average DNA (viral genome (VG)) copies per diploid genome and in Table 19 as fold change in DNA copies per diploid genome relative AAV9 and AAV5 controls.
- the AAV5 capsid variant TTN-002 demonstrated comparable biodistribution relative to AAV9 in the mouse brain and increased biodistribution and viral genome levels than wild-type AAV5. More specifically, in the brain, the TTN-002 capsid variant led to 9-fold higher DNA (viral genome (VG)) copies per diploid genome relative to the AAV5 control (Table 19).
- wild- type AAV5 and the AAV5 capsid variant, TTN-002 resulted in decreased biodistribution and DNA (viral genome (VG)) copies per diploid genome in the liver relative to AAV9 (Table 19).
- Table 18 Transgene mRNA expression with the TTN-002 capsid variant in mice Attorney Docket No.14640.0092-00304 Table 19. Fold-change in transgene mRNA expression and DNA copies per diploid genome relative to AAV9 and AAV5 in the brain and liver of mice D.
- AAV particles were generated with the TTN-002 capsid variant, the AAV5 capsid control, or the AAV9 capsid control which comprised a self-complementary viral genome encoding a histone H2b protein with an HPC4 tag (TTN-002 capsid variant), T7 tag (AAV5 capsid control), or HA tag (AAV9 control capsid) driven by a ubiquitous CAG promoter.
- the in-life period was 28 days and then various CNS and peripheral tissues were collected for measuring transgene mRNA (expression) by RT-qPCR, protein expression by IHC, and viral DNA (biodistribution) by ddPCR. Data were then normalized to the dose of each viral vector in the dosing solution. Table 20.
- the TTN-002 capsid variant, an AAV5 capsid variant demonstrated increased biodistribution in the caudate and motor cortex in the brain of the marmosets relative to the AAV5 capsid control, as well as the AAV9 control.
- the TTN-002 capsid variant also demonstrated Attorney Docket No.14640.0092-00304 increased transgene expression relative to AAV5 in the caudate and motor cortex, increased transgene expression relative to AAV9 in the caudate, and comparable transgene expression relative to AAV9 in the motor cortex (Tables 21 and 22). Similar expression and distribution was observed by immunohistochemistry. More specifically, staining for TTN-002 was detected in the caudate, putamen, thalamus, and cerebellum, and this staining was increased relative to AAV5. Staining for TTN-002 was also observed in the molecular and granule layer of the cerebellum.
- TTN-002 capsid variant exhibited lower biodistribution (Table 21) and transgene expression (Table 22) relative to AAV5 and AAV9, indicating that the TTN-002 capsid variant was detargeted in the liver relative to AAV5 and AAV9 in marmosets.
- TTN-002 led to increased biodistribution (Table 21) in the heart relative to AAV5 and AAV9, increased transgene expression in the heart relative to AAV5, and decreased transgene expression in the heart relative to AAV9 (Table 22).
- TTN-002 transgene mRNA fold over housekeeping gene
- AAV9 transgene mRNA fold over housekeeping gene relative to AAV9
- AAV5 transgene mRNA fold over housekeeping gene relative to AAV5
- TTN-002 demonstrated the ability to cross species, demonstrating improved CNS tropism and biodistribution in both NHPs and rats relative to AAV5 in mice.
- Example 3 Maturation of the TTN-002 capsid in mice [0504] This Example describes maturation of the TTN-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 943) capsid variant in mice to further enhance its transduction and biodistribution in the central nervous system, evolve the AAV capsid variant further, and to provide cross-species compatibility.
- mutagenic primers were used to introduce point mutations at a low frequency, scattered across the mutagenesis region in the TTN-002 sequence ranging from approximately position 571 to position 586, numbered according to SEQ ID NO: 982.
- sets of three contiguous amino acids were randomized across the mutagenesis region in the TTN-002 sequence, which spanned from approximately position 571 to position 586, numbered according to SEQ ID NO: 982.
- AAV capsid variants arising from each maturation approach for TTN-002 were pooled together, for subsequent testing and characterization in mice.
- the library of pooled matured AAV capsid variants generated from TTN-002 using the first maturation approach and the library of pooled matured AAV capsid variants generated from TTN-002 using the second maturation approach were each injected into three CD-1 Outbred mice. After a period in Attorney Docket No.14640.0092-00304 life, the brains of the mice were isolated and RNA was extracted.
- RNA recovery and RT-PCR amplification Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the TTN-002 control, and the peptides comprised within the matured variants were identified.
- the data from the first maturation approach are provided in Table 23 and the data from the second maturation approach is provided in Table 24.
- the matured capsid variants were filtered based on their coefficient of variance (CV), which was calculated for each peptide across six brain samples taken (two per mouse). Those that had a CV value ⁇ 1 were identified, as these were the peptides that were reliably detected in 5/6 or 6/6 brain samples isolated from the three mice.
- CV coefficient of variance
- Table 23 provides the peptide sequences of these matured capsid variants and the fold enrichment of the matured capsid variant relative to the non-matured TTN-002 control. As shown in Table 23, approximately 28 TTN-002 matured capsid variants demonstrated an increase in expression relative to the non-matured TTN-002 control, with approximately 16 variants demonstrating at least a 2- fold increase in expression. Several variants demonstrated at least an 8-fold to 15-fold increase in expression relative to the non-matured TTN-002 control. Table 23.
- NGS fold-enrichment of TTN-002 matured AAV capsid variants in the brain of outbred mice following first mutagenesis approach Attorney Docket No.14640.0092-00304 [0507]
- the matured capsid variants were filtered for those that were detectable in all samples from all mice injected with the matured capsid variants.
- Table 24 provides the peptide sequences of these matured capsid variants, the fold enrichment of the matured capsid variant relative to the non-matured TTN-002 control, as well as the predicted capsid of origin from which the variant was matured.
- TTN-002 matured capsid variants demonstrated an increase in expression relative to the non-matured TTN-002 control, with approximately 358 variants demonstrated at least a 2-fold increase in expression.
- Several variants demonstrated a 20-711-fold or greater increase in expression relative to the non-matured TTN-002 control. Table 24.
- Example 4 Maturation of the TTN-002 capsid in NHPs
- This Example describes maturation of TTN-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 943 (encoded by SEQ ID NO: 944)) capsid variant in NHPs, specifically cynomolgus macaques (Macaca fascicularis), to further enhance its transduction and biodistribution in the central nervous system and peripheral tissues and to evolve the AAV capsid variant further.
- Two approaches were used to mature the TTN-002 capsid sequences in order to randomize and mutate within and around the peptide insert comprised within loop VIII of the capsid variant.
- mutagenic primers were used to introduce point mutations at a low frequency, scattered across the mutagenesis region in the TTN-002 sequence ranging from approximately position 571 to position 586, numbered according to SEQ ID NO: 982.
- sets of three contiguous amino acids were randomized across the mutagenesis region in the TTN-002 sequence, which spanned from approximately position 571 to position 586, numbered according to SEQ ID NO: 982.
- AAV capsid variants arising from each maturation approach for TTN-002 were pooled together, for subsequent testing and characterization in NHPs.
- the library of pooled matured AAV capsid variants generated from TTN-002 using the first maturation approach and the library of pooled matured AAV capsid variants generated from TTN-002 using the second maturation approach were each injected into two NHPs. After a period in life, the brains, heart, liver, muscle, and DRG of the NHPs were isolated and RNA was extracted. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the TTN-002 control, and the peptides comprised within the variants were identified.
- the matured capsid variants were filtered based on their coefficient of variance (CV), which was calculated for each peptide across the brain, heart, liver, muscle and DRG samples taken from the two NHPs. Those that had a CV value ⁇ 2 were identified, as these were the peptides that were reliably detected in the majority of samples isolated from the brains of the two NHPs.
- CV coefficient of variance
- Table 25 provides the peptide sequences of these matured capsid variants and the fold enrichment of the matured capsid variant relative to the non-matured TTN-002 control that demonstrated the greatest fold-change in expression relative to the non-matured TTN-002 capsid variant in the brain of NHPs, following the first maturation approach and the second maturation approach.
- Table 25 following the first maturation approach, approximately 5 TTN-002 matured capsid variants Attorney Docket No.14640.0092-00304 demonstrated an increase in expression relative to the non-matured TTN-002 control, which demonstrated at least a 5-fold to 53-fold increase in expression in the NHP brain relative to the non- matured TTN-002 control.
- TTN-002 matured capsid variants demonstrated an increase in expression in the NHP brain relative to the non- matured TTN-002 control, with at least 27 demonstrating at least a 2-fold increase in expression (Table 25).
- Several variants demonstrated at least a 12-fold to 222-fold increase in expression in the NHP brain relative to the non-matured TTN-002 control (Table 25).
- Fold-change in expression for the TTN-002 matured variants in Table 25 were also calculated for the DRG, muscle, liver (RNA and DNA), and heart of the NHPs following each maturation approach.
- the matured TTN-002 capsid variant comprising the amino acid sequence TNNQSSYTPSLVQKTA (SEQ ID NO: 1585) demonstrated increased expression in the brain, heart, and liver relative to the non-matured TTN-002 control.
- the matured TTN-002 capsid variants comprising the amino acid sequence TNNQSSYPPSLVKKTA (SEQ ID NO: 1591) and TNNQSSYPPSLVQKPA (SEQ ID NO: 1593), demonstrated increased expression in the brain and heart relative to the non-matured TTN-002 control.
- the matured TTN-002 capsid variant comprising the amino acid sequence INNQSSYPAEVVQKTA (SEQ ID NO: 1024) demonstrated increased expression in the brain and the muscle relative to the non-matured TTN- 002 control. Also, as shown in Table 25, many of the TTN-002 capsid variants that had increased expression in the brain, were de-targeted in the DRG. Therefore, several matured variants demonstrated increased tropism in more than one tissue type in the NHPs, with many showing reduced expression in the DRG. Table 25.
- NGS fold-enrichment of the TTN-002 matured AAV capsid variants in the brain of NHPs following first and second mutagenesis approaches as compared to other NHP and mouse tissues Attorney Docket No.14640.0092-00304 [0514] Furthermore, several of the TTN-002 matured capsid variants demonstrating an increase in expression relative to the non-matured TTN-002 control following the first and second maturation approaches in the brain of NHPs as shown in Table 25, also demonstrated an increase in expression in the brain of mice following the first and second maturation approach in mice.
- the matured TTN-002 capsid variant comprising the amino acid sequence TNNQSKYPAEVVQKTA (SEQ ID NO: 1538) demonstrated a 53.7-fold increase in expression relative to the non-matured TTN-002 following the first maturation approach in brain of NHPs, demonstrated a 20.86-fold increase in expression relative Attorney Docket No.14640.0092-00304 to the non-matured TTN-002 following the second maturation approach in brain of NHPs, a 10.26-fold increase in expression relative to the non-matured TTN-002 following the first maturation approach in brain of mice, and a 5.47-fold increase in increase in expression relative to the non-matured TTN-002 following the second maturation approach in brain of mice.
- the matured TTN-002 capsid variant comprising the amino acid sequence TNNSSSYPAEVVQKTA (SEQ ID NO: 1539) demonstrated an 18.997-fold increase in expression relative to the non-matured TTN-002 following the first maturation approach in brain of NHPs, an 8.093-fold increase in expression relative to the non- matured TTN-002 following the second maturation approach in brain of NHPs, an 8.539-fold increase in expression relative to the non-matured TTN-002 following the first maturation approach in brain of mice, and a 5.903-fold increase in increase in expression relative to the non-matured TTN-002 following the second maturation approach in brain of mice.
- Matured TTN-002 capsid variants comprising the amino acid sequences of SEQ ID NOs: 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, and 1593 also demonstrated an increase in expression in the brain of both NHPs and mice, relative to the non-matured TTN-002 control. Therefore, several matured variants demonstrated increased expression relative to the non-matured controls in at least two different species, indicating cross-species tropism.
- Table 26 provides the peptide sequences of these matured capsid variants, and the fold enrichment of the matured capsid variant relative to the non-matured TTN-002 control that demonstrated increased expression in the heart of NHPs following the second maturation approach.
- approximately 17 TTN-002 matured capsid variants demonstrated an increase in expression in the heart relative to the non-matured TTN-002 control, with at least 13 demonstrating at least a 2-fold increase in expression.
- Several variants demonstrated at least a 10-fold to 47-fold increase in expression in the heart relative to the non-matured TTN-002 control.
- TTN-002 matured capsid variants demonstrated an increase in expression in the muscle relative to the non-matured TTN-002 control, with at least 19 demonstrating at least a 2-fold increase in expression.
- Several variants demonstrated at least a 7-fold to 38-fold increase in expression in the muscle relative to the non-matured TTN-002 control.
- Fold-change in expression for the TTN-002 matured variants in Table 27 were also calculated for the brain, DRG, heart, and liver (RNA and DNA), of the NHPs and in the brains of mice. Table 27.
- Table 28 provides the peptide sequences of these matured capsid variants, and the fold enrichment of the matured capsid variant relative to the non-matured TTN-002 control that demonstrated increased expression in the liver of NHPs following the first and second maturation approach. As shown in Table 28, following the first maturation approach, approximately 7 TTN-002 matured capsid variants demonstrated an 11-fold to 189-fold increase in expression in the liver relative to the non-matured TTN- 002 control.
- TTN-002 matured capsid variants demonstrated an increase in expression in the liver of at least 9-fold relative to the non-matured TTN-002 control (Table 28).
- Several variants demonstrated at least a 50-fold to 114-fold increase in expression in the liver relative to the non-matured TTN-002 control (Table 28).
- Fold-change in expression for the TTN-002 matured variants in Table 28 were also calculated for the brain, DRG, heart, and muscle of the NHPs and in the brains of mice. Table 28.
- NGS fold-enrichment of the TTN-002 matured AAV capsid variants in the liver of NHPs following both mutagenesis approaches Attorney Docket No.14640.0092-00304 Attorney Docket No.14640.0092-00304 Attorney Docket No.14640.0092-00304 Attorney Docket No.14640.0092-00304 Attorney Docket No.14640.0092-00304 Attorney Docket No.14640.0092-00304 [0518]
- TTN-002 AAV capsid variant in diverse primate species [0519] This Example evaluates the tropism and cross-species compatibility of the TTN-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 943) capsid variant in two diverse primate species, marmosets (Callithrix jacchus) and African green monkeys (Chlorocebus sabaeus), as compared to their tropism in cynomolgus macaques (Macaca fascicularis) provided in Example 1 and 2.
- SEQ ID NO: 982 amino acid
- 984 DNA
- the amino acid and DNA sequences of the TTN-002 capsid variant are provided, e.g., in Tables 4 and 5, respectively.
- the TTN-002 capsid variant demonstrated increased CNS tropism in diverse primate species.
- the TTN-002 capsid variant demonstrated a 64.9-fold increase in expression relative to AAV5 in the brain of cynomolgus macaques (Table 14, Example 1), a 7.5-fold increase in expression relative to AAV5 in the brain of African green monkeys, and a 40.4-fold increase in expression relative to AAV5 in the brain of marmosets.
- TTN-002 also resulted in increased expression in the brain of rats (Table 14, Example 1), demonstrating an average fold change in expression relative to AAV5 of 41.1.
- Table 29 NGS-Fold Enrichment of TTN-002 in African green monkeys Table 30. NGS-Fold Enrichment of TTN-002 in Marmosets [0523]
- ITR-to-ITR sequences comprising promoters were packaged into AAV6 capsids and delivered by intrastriatal injection to Sprague Dawley rats at a dose of 1x10 10 VG. Tissue samples were collected at 3 weeks or 10 weeks and frataxin protein levels quantified. Frataxin constructs comprising truncated CBA and CMV promoters were packed into an AAV9 variant capsid (VOY201) and administered by intravenous delivery to Sprague Dawley rats at a dose of either 6.3x10 12 or 2x10 13 VG/kg. After 28 or 90 days, tissue samples were collected and processed for quantification of frataxin expression (ng/mg).
- Viral genomes comprising a CMV-D7 (SEQ ID NO: 1750) or a CBA-D8 (SEQ ID NO: 1742 promoter with a frataxin payload sequence were packaged into another AAV9-variant (VOY101) capsid to generate AAV particles.
- AAV particles were administered by intravenous delivery via the tail vein to male Sprague Dawley rats at one of two doses (6.3x10 12 or 2x10 13 ).
- tissue samples were collected (heart, liver, cerebellum, thoracic and lumbar DRG) and processed for quantification of viral genome per diploid cell and frataxin expression levels based on an Anti-Frataxin SimpleStep ELISA.
- FIGs.2A, 2B, 2C and 2D Data are shown below in Tables 31 and 32, FIGs.2A, 2B, 2C and 2D.
- CMV-D7 SEQ ID NO: 1750
- CBA-D8 SEQ ID NO: 1742
- CBA SEQ ID NO: 1734
- CMV-D7 and CBA-D8 promoters generated FXN expression approximately 50-260-fold lower than CBA-driven frataxin expression.
- the CMV-D7 (SEQ ID NO: 1750) and CBA-D8 (SEQ ID NO: 1742) promoters both drove frataxin expression in the cerebellum, heart and DRG at 180 days after administration of the AAV particles.
- TTN-002 for delivering frataxin in mice
- SEQ ID NO: 982 amino acid
- DNA amino acid
- the amino acid and DNA sequences of TTN-002 are provided, e.g., in Tables 4 and 5, respectively.
- TTN-002_FXN AAV particles were generated with TTN-002 encapsulating a viral genome comprising SEQ ID NO: 1797 (“TTN-002_FXN”).
- the AAV particles or vehicle control (modified PBS) were administered intravenously to wildtype or frataxin-deficient mice (B6.Cg-Pvalb tm1(cre)Arbr Fxn em2Lutzy Fxn em2.1Lutzy /J (Jackson Laboratory Stock#029721)).
- TTN-002_FXN was administered at 1e14 or 2e13 vg/kg.
- mice were euthanized and biodistribution (vg/dc) and frataxin expression (ng/mg) were evaluated from the cerebellum using ddPCR and ELISA, respectively. Results are shown in FIG.4A and FIG.4B.
- Equivalents and Scope [0537] Those skilled in the art will recognize, or be able to ascertain using no more than routine experimentation, many equivalents to the specific embodiments in accordance with the Detailed Attorney Docket No.14640.0092-00304 Description provided herein. The scope of the present disclosure is not intended to be limited to the above Detailed Description, but rather is as set forth in the appended claims. [0538] Where ranges are given, endpoints are included.
- compositions of the disclosure e.g., any, composition, therapeutic or active ingredient; any method of production; any method of use; etc.
- any particular embodiment of the compositions of the disclosure can be excluded from any one or more claims, for any reason, whether or not related to the existence of prior art.
Landscapes
- Health & Medical Sciences (AREA)
- Life Sciences & Earth Sciences (AREA)
- Chemical & Material Sciences (AREA)
- Organic Chemistry (AREA)
- General Health & Medical Sciences (AREA)
- Genetics & Genomics (AREA)
- Medicinal Chemistry (AREA)
- Molecular Biology (AREA)
- Animal Behavior & Ethology (AREA)
- Gastroenterology & Hepatology (AREA)
- Pharmacology & Pharmacy (AREA)
- Biotechnology (AREA)
- Public Health (AREA)
- Veterinary Medicine (AREA)
- Virology (AREA)
- Epidemiology (AREA)
- Biochemistry (AREA)
- Biophysics (AREA)
- Proteomics, Peptides & Aminoacids (AREA)
- Engineering & Computer Science (AREA)
- Medicines Containing Material From Animals Or Micro-Organisms (AREA)
- Micro-Organisms Or Cultivation Processes Thereof (AREA)
- Medicines That Contain Protein Lipid Enzymes And Other Medicines (AREA)
Abstract
The disclosure relates to compositions and methods for altering, e.g., enhancing, the level of frataxin protein via delivery using an adeno-associated viral (AAV) capsid variant. The compositions and methods of the present disclosure are useful in the treatment of subjects who have, have been diagnosed with having, or are at risk of having a disorder associated with frataxin (FXN) deficiency, e.g., Friedreich's Ataxia.
Description
Attorney Docket No.14640.0092-00304 COMPOSITIONS AND METHODS FOR THE TREATMENT OF DISORDERS RELATED TO FRATAXIN DEFICIENCY RELATED APPLICATIONS [01] This application claims the benefit of and priority to US Provisional Application Serial No. 63/519,952, filed August 16, 2023. SEQUENCE LISTING [02] The present application is being filed along with a Sequence Listing in electronic format. The Sequence Listing file, entitled 14640_0092-00304_SL.xml, was created on June 21, 2024, and is 1,753,602 bytes in size. The information in electronic format of the Sequence Listing is incorporated herein by reference in its entirety. FIELD [03] Described herein are compositions and methods relating to adeno-associated virus (AAV) viral particles for the delivery of polynucleotides, e.g., polynucleotides encoding a frataxin (FXN) protein for use in the treatment of Friedreich’s Ataxia (FA). In some embodiments, compositions described herein may be used to treat a subject in need thereof, such as a human subject diagnosed with FA, or as a research tool in the study of diseases or conditions in cells or animal models of FA. BACKGROUND [04] Friedreich’s Ataxia (FA) is an autosomal recessive inherited disease that causes progressive damage to the nervous system. See Parkinson et al., Journal of Neurochemistry, 2013, 126 (Suppl.1), 103-117, the contents of which are herein incorporated by reference in their entirety. FA typically results from the degeneration of nervous tissue in the spinal cord due to reduced expression of the mitochondrial protein frataxin (FXN; also known as, e.g., CyaY, FA, FARR, FRDA, and X25) in sensory neurons that direct muscle movement of the arms and legs. See Koeppen, Arnulf; J Neurol Sci., 2011, April 15; 303(1-2): 1–12. Onset usually occurs at puberty or by age 25. See Campuzano, et al., Science, 271.5254 (Mar 8, 1996): 1423. Initial symptoms of FA include poor coordination such as gait disturbance, poor balance, leg weakness, decreased walking, impaired coordination, dysarthria, nystagmus, impaired sensation, kyphoscoliosis, and foot deformities. See Parkinson et al., Journal of Neurochemistry, 2013, 126 (Suppl.1), 103-117. FA is also associated with scoliosis, heart disease, and diabetes. The disease generally progresses until a wheelchair is required for mobility. Incidence of FA among Caucasian populations is between about 1 in 20,000 and about 1 in 50,000, with a deduced carrier frequency of about 1 in 120 in European populations. See Nageshwaran and Festenstein, Frontiers in Neurology, Vol. 6, Art.262 (2015); Campuzano, et al., Science, 271.5254 (Mar 8, 1996): 1423, the contents of each of which are herein incorporated by reference in their entirety. The expansion of an intronic GAA triplet repeat in the FXN gene is the genetic cause of reduced expression of FXN resulting in FA. See Parkinson
Attorney Docket No.14640.0092-00304 et al., Journal of Neurochemistry, 2013, 126 (Suppl.1), 103-117. Over time, the deficiency causes the aforementioned symptoms, as well as frequent fatigue due to effects on cellular metabolism. Currently, omaveloxolone (Skyclarys®) is the only FDA approved treatment for FA. Omaveloxolone is a semisynthetic oleanane triterpenoid that activates Nrf2, a master transcription factor that regulates genes with antioxidative, anti-inflammatory, and mitochondrial bioenergetic properties. See Reisman et al. (2019) Drug Des Devel Ther.13:1259-1270. While gene therapy constructs for delivering a frataxin protein have been described in the arm, there remains a need to develop improved constructs for better targeting of the appropriate tissues in the body. [05] Adeno-associated viruses (AAVs) have emerged as a widely studied and utilized viral particles for delivery of therapeutically effective polypeptides to mammalian cells. See, e.g., Tratschin et al., Mol. Cell Biol., 5(11):3251-3260 (1985) and Grimm et al., Hum. Gene Ther., 10(15):2445-2450 (1999), the contents of each of which are incorporated herein by reference in their entirety. [06] There remains a need for effective methods of treatment using AAV capsids that are capable of delivering FXN to a target cell or tissue, e.g., a CNS cell or tissue. SUMMARY [07] The present disclosure addresses these challenges by providing AAV-based compositions, AAV-based compositions for use in methods for treating Friedreich’s Ataxia (FA) in subjects, and methods for treating FA in subjects. Disclosed herein are compositions and methods directed to AAV- based gene delivery of FXN (e.g., human FXN) to ameliorate loss-of-function and to improve FXN expression (e.g., in the brain, e.g., in neurons). In some embodiments, the compositions and methods can be used to slow, halt, or reverse symptoms of FA. [08] In some aspects, the present disclosure provides an AAV particle comprising an AAV capsid and a nucleotide sequence encoding a FXN protein, also referred to herein as a FXN-encoding sequence or as a FXN protein-encoding sequence. In some embodiments, the nucleotide sequence encoding a FXN protein is comprised in a viral genome. In some embodiments, the FXN protein is a human FXN protein. In some embodiments, the FXN protein is a wildtype human FXN protein. In some embodiments, the AAV particle comprises a viral genome encoding the FXN protein (e.g., a human FXN protein) and the AAV capsid is an AAV capsid variant. In some embodiments, the viral genome comprises a truncated chicken ȕ-actin (CBA) promoter operably linked to the FXN-encoding sequence, and the AAV capsid variant is an AAV5 capsid variant. [09] In some embodiments, the AAV capsid variant (e.g., an AAV5 capsid variant) comprises a peptide that has replaced amino acid position 577 as numbered according to SEQ ID NO: 138 (in other words, the peptide has replaced the amino acid corresponding to position 577 of SEQ ID NO: 138). In some embodiments, the AAV capsid variant is an AAV5 capsid variant comprising a peptide comprising the amino acid sequence of VQK in loop VIII. In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) in loop VIII, or a
Attorney Docket No.14640.0092-00304 peptide comprising one, two, or three—but no more than three—substitutions relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). In some embodiments, the peptide comprising the amino acid sequence of SEQ ID NO: 943 has replaced a threonine at amino acid position 577 as numbered according to SEQ ID NO: 138 (in other words, the peptide comprising the amino acid sequence comprising no more than three substitutions relative to SEQ ID NO: 943 has replaced the amino acid corresponding to position 577 of SEQ ID NO: 138). In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) in loop VIII. In some embodiments, the peptide comprising the amino acid sequence of SEQ ID NO: 943 has replaced a threonine at amino acid position 577 as numbered according to SEQ ID NO: 138 (in other words, the peptide comprising the amino acid sequence of SEQ ID NO: 943 has replaced the amino acid corresponding to position 577 of SEQ ID NO: 138). In some embodiments, the peptide is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant. In some embodiments, the peptide is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant. [010] In some aspects, the present disclosure provides an adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome, wherein the viral genome comprises a frataxin (FXN)-encoding sequence and the AAV capsid variant comprises an amino acid sequence having the formula [N2]-[N3], wherein: (i) [N2] comprises X1, X2, X3, X4, and X5, wherein X1 is Y, N, or C; X2 is P, K, T, or Q; X3 is A or P; X4 is E, S, or A; and X5 is V, L, or E; and (ii) [N3] comprises the amino acid sequence VQK, EQK, VKK, VHK, VQQ, or LQK; and wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 739, or an amino acid sequence at least 95% identical thereto. [011] In some embodiments, [N2]-[N3] is present in loop VIII, wherein loop VIII is present at amino acids comprising those corresponding to positions 571-599 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, [N2]-[N3] is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 982. [012] In some embodiments, the AAV capsid variant is an AAV5 capsid variant comprising [N2]- [N3] in place of an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [013] In some embodiments, [N2] comprises the amino acid sequence YP, YPA, YPAE (SEQ ID NO: 21), or YPAEV (SEQ ID NO: 1). In some embodiments, [N3] comprises the amino acid sequence VQK. In some embodiments, [N2]-[N3] comprises the amino acid sequence AEVVQK (SEQ ID NO: 36) or PAEVVQK (SEQ ID NO: 20). In some embodiments, [N2]-[N3] comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). [014] In some embodiments, the AAV capsid variant further comprises [N1] comprising XD, XE, and XF, wherein: XD of [N1] is Q, T, S, A, I, L, or H; XE of [N1] is S, G, A, or R; and XF of [N1] is S, K, L, R, A, or T; wherein [N1] immediately precedes [N2]-[N3]. In some embodiments, [N1] comprises the amino acid sequence QS, SS, or QSS. In some embodiments, [N1]-[N2] comprises the amino acid sequence QSSYPAEV (SEQ ID NO: 96). In some embodiments, [N1]-[N2]-[N3] comprises the amino acid sequence SSYPAEVVQ (SEQ ID NO: 121) or QSSYPAEVVQK (SEQ ID NO: 150).
Attorney Docket No.14640.0092-00304 [015] In some embodiments, the AAV capsid variant further comprises [N0] comprising XA, XB, and XC, wherein: XA of [N0] is T, I, or N; XB of [N0] is N; and XC of [N0] is N, T, S, or K; wherein [N0] immediately precedes [N1]. In some embodiments, [N0] comprises the amino acid sequence TN, NN, or TNN. In some embodiments, [N0]-[N1] comprises the amino acid sequence TNNQSS (SEQ ID NO: 183). In some embodiments, [N0]-[N1]-[N2]-[N3] comprises the amino acid sequence TNNQSSYPAEVVQK (SEQ ID NO: 500). [016] In some embodiments, the AAV capsid variant further comprises [N4] comprising XG and XH, wherein: XG of [N4] is T, P, or N; and (b) XH of [N4] is A; wherein [N4] is present immediately subsequent to [N3]. In some embodiments, [N4] comprises the amino acid sequence TA. In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] comprises the amino acid sequence TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). [017] In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is present in loop VIII, wherein loop VIII is present at amino acids comprising those corresponding to positions 571-599 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, [N0] is present at amino acids corresponding to positions 571-573 of the amino acid sequence of SEQ ID NO: 982; [N1] is present at amino acids corresponding to positions 574-576 of the amino acid sequence of SEQ ID NO: 982; [N2] is present at amino acids corresponding to positions 577-581 of the amino acid sequence of SEQ ID NO: 982; [N3] is present at amino acids corresponding to positions 582-584 of the amino acid sequence of SEQ ID NO: 982; and [N4] is present at amino acids corresponding to positions 585-586 of the amino acid sequence of SEQ ID NO: 982. [018] In some aspects, the present disclosure provides an adeno-associated virus (AAV) particle comprising: (i) a viral genome comprising a frataxin (FXN)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% identical to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises at least 4, at least 5, at least 6, at least 7, or all 8 consecutive amino acids from the amino acid sequence YPAEVVQK (SEQ ID NO: 943). In some embodiments, (i) the at least 4 consecutive amino acids comprise the amino acid sequence YPAE (SEQ ID NO: 21), optionally present at amino acids corresponding to positions 577-580 of the amino acid sequence of SEQ ID NO: 982; (ii) the at least 5 consecutive amino acids comprise the amino acid sequence YPAEV (SEQ ID NO: 1), optionally present at amino acids corresponding to positions 577-581 of the amino acid sequence of SEQ ID NO: 982; (iii) the at least 6 consecutive amino acids comprise the amino acid sequence YPAEVV (SEQ ID NO: 725), optionally present at amino acids corresponding to positions 577-582 of the amino acid sequence of SEQ ID NO: 982; or (iv) the at least 7 consecutive amino acids comprise the amino acid sequence YPAEVVQ (SEQ ID NO: 726), optionally present at amino acids corresponding to positions 577-583 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943) present at amino acids corresponding to positions 577-584 of the amino acid sequence of SEQ ID NO: 982.
Attorney Docket No.14640.0092-00304 [019] In some aspects, the present disclosure provides an adeno-associated virus (AAV) particle comprising: (i) a viral genome comprising a frataxin (FXN)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% identical to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943) at amino acids corresponding to positions 577-584 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, the AAV capsid variant further comprises (i) an amino acid sequence having at least 95% identity to the amino acid sequence of SEQ ID NO: 738, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); and/or (ii) an amino acid sequence having at least 95% identity to the amino acid sequence of SEQ ID NO: 982, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). [020] In some aspects, the present disclosure provides an adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome, wherein the viral genome comprises a frataxin (FXN)-encoding sequence and the AAV capsid variant comprises (i) an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 982, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); (ii) an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 738, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); and/or (iii) an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). [021] In some embodiments, the AAV capsid variant comprises (i) the amino acid sequence of SEQ ID NO: 982; (ii) the amino acid sequence of SEQ ID NO: 738; and/or (iii) the amino acid sequence of SEQ ID NO: 739. [022] In some embodiments, the FXN-encoding sequence encodes a FXN protein that is not a cynomolgus monkey FXN protein. In some embodiments, the FXN-encoding sequence encodes a human FXN protein. In some embodiments, the human FXN protein comprises the amino acid sequence of SEQ ID NO: 1825, or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the human FXN protein is a wildtype human FXN protein. In some embodiments, the FXN-encoding sequence comprises a nucleotide sequence encoding the amino acid sequence of SEQ ID NO: 1825. In some embodiments, the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [023] In some embodiments, the viral genome further comprises a promoter operably linked to the FXN-encoding sequence. In some embodiments, the promoter comprises a human elongation factor 1Į- subunit (EF1Į) promoter, a cytomegalovirus (CMV) immediate-early enhancer and/or promoter, a chicken ȕ-actin (CBA) promoter, a CAG promoter, a ȕ glucuronidase (GUSB) promoter, a ubiquitin C
Attorney Docket No.14640.0092-00304 (UBC) promoter, a neuron-specific enolase (NSE) promoter, a platelet-derived growth factor (PDGF) promoter, a platelet-derived growth factor B-chain (PDGF-ȕ) promoter, a intercellular adhesion molecule 2 (ICAM-2) promoter, a synapsin (Syn) promoter, a methyl-CpG binding protein 2 (MeCP2) promoter, a Ca2+/calmodulin-dependent protein kinase II (CaMKII) promoter, a metabotropic glutamate receptor 2 (mGluR2) promoter, a neurofilament light chain (NFL) promoter, a neurofilament heavy chain (NFH) promoter, a ȕ-globin minigene nȕ2 promoter, a preproenkephalin (PPE) promoter, a enkephalin (Enk) and excitatory amino acid transporter 2 (EAAT2) promoter, a glial fibrillary acidic protein (GFAP) promoter, a myelin basic protein (MBP) promoter, a cardiovascular promoter (e.g., ĮMHC, cTnT, and CMV-MLC2k), a liver promoter (e.g., hAAT, TBG), a skeletal muscle promoter (e.g., desmin, MCK, C512), or a functional fragment or truncation of any of the foregoing. In some embodiments, the promoter is a CMV promoter or CBA promoter, or a functional fragment or truncation of a CMV promoter or CBA promoter. [024] In some embodiments, the promoter is a truncated CBA promoter. In some embodiments, the truncated CBA promoter is 50-400 nucleotides in length, e.g., 100-332 nucleotides in length. In some embodiments, the promoter comprises the nucleotide sequence of any one of SEQ ID NOs: 1738, 1740, and 1742 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [025] In some embodiments, the promoter is a truncated CMV promoter. In some embodiments, the truncated CMV promoter is 50-300 nucleotides in length. In some embodiments, the promoter comprises the nucleotide sequence of SEQ ID NO: 1750 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [026] In some embodiments, the viral genome further comprises a microRNA (miR) binding site that modulates expression of the encoded FXN protein in a cell or tissue of the liver. In some embodiments, the viral genome comprises 3 copies of the miR binding site. In some embodiments, the 3 copies of the miR binding site are identical. In some embodiments, the 3 copies of the miR binding site are continuous. [027] In some embodiments, the miR binding site is a miR122 binding site. In some embodiments, the miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1827 or a sequence having one, two, three, or at most four substitutions relative to the nucleotide sequence of SEQ ID NO: 1827. In some embodiments, the viral genome comprises 3 copies of a miR122 binding site comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence having one, two, three, four, five, six, seven, eight, nine, or at most ten substitutions relative to the nucleotide sequence of SEQ ID NO: 1826. [028] In some embodiments, the viral genome further comprises at least one inverted terminal repeat (ITR) region. In some embodiments, the at least one ITR region comprises an AAV2 ITR. In some embodiments, the viral genome comprises a 5’ ITR region and a 3’ ITR region. In some embodiments,
Attorney Docket No.14640.0092-00304 the 5’ ITR region and 3’ ITR region are each an AAV2 ITR. In some embodiments, the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [029] In some embodiments, the viral genome further comprises an intron/exon region comprising an intron region and/or an exon region. In some embodiments, the intron/exon region comprises: an immediate-early 1 (ie1) intron region and/or a human beta-globin (hBglobin) intron 2 region; and/or an ie1 exon region and/or an hBglobin exon region. [030] In some embodiments, the intron region comprises: the nucleotide sequence of SEQ ID NO: 1819 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the nucleotide sequence of SEQ ID NO: 1820 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the exon region comprises: the nucleotide sequence of SEQ ID NO: 1817 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the nucleotide sequence of SEQ ID NO: 1821 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [031] In some embodiments, the viral genome further comprises a polyadenylation (polyA) region. In some embodiments, the polyA region comprises a human growth hormone (hGH) polyA region. In some embodiments, the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [032] In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [033] In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%,
Attorney Docket No.14640.0092-00304 at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [034] In some embodiments, the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) the FXN-encoding sequence, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; and (iv) a 3’ ITR region. [035] In some embodiments, the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) the FXN-encoding sequence, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (iv) at least one miR122 binding site; and (v) a 3’ ITR region. [036] In some embodiments, the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron/exon region; (iv) the FXN-encoding sequence, wherein the FXN- encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; and (vi) a 3’ ITR region. [037] In some embodiments, the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron/exon region; (iv) the FXN-encoding sequence, wherein the FXN- encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; and (vii) a 3’ ITR region. [038] In some embodiments, wherein the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron and/or exon region; (iv) the FXN-encoding sequence, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; (vii) a filler sequence; and (viii) a 3’ ITR region.
Attorney Docket No.14640.0092-00304 [039] In some embodiments, (i) the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises the nucleotide sequence of SEQ ID NO: 1742 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1826 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (vii) the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1841 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. [040] In some embodiments, (i) the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises the nucleotide sequence of SEQ ID NO: 1750 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least
Attorney Docket No.14640.0092-00304 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1826 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (vii) the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1840 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. [041] In some embodiments, (i) the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises the nucleotide sequence of SEQ ID NO: 1738 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1826 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (vii) the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO:
Attorney Docket No.14640.0092-00304 1838 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. [042] In some embodiments, (i) the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises the nucleotide sequence of SEQ ID NO: 1740 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1826 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (vii) the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1839 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. [043] In some embodiments, the viral genome comprises (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%,
Attorney Docket No.14640.0092-00304 at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; or (d) the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [044] In some embodiments, (i) the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811; (ii) the promoter comprises the nucleotide sequence of SEQ ID NO: 1742; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816; (iv) the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824;(v) the at least one miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1826; (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828; and (vii) the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812. In some embodiments, the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1841, wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. In some embodiments, the viral genome consists of the nucleotide sequence of SEQ ID NO: 1797. [045] In some aspects, the present disclosure provides an adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome comprising a frataxin (FXN)-encoding sequence, wherein the AAV capsid variant comprises: (i) the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); (ii) the amino acid sequence of SEQ ID NO: 738 or an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); or (iii) the amino acid sequence of SEQ ID NO: 739 or an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); and wherein the viral genome comprises: (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; or (d) the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least
Attorney Docket No.14640.0092-00304 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [046] In some aspects, the present disclosure provides an adeno-associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982, the amino acid sequence of SEQ ID NO: 738, and/or the amino acid sequence of SEQ ID NO: 739. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [047] In some embodiments, the viral genome is single-stranded. [048] In some aspects, the present disclosure provides a cell comprising an AAV particle disclosed herein. In some embodiments, the cell is a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial cell. [049] In some aspects, the present disclosure provides a method of making and AAV particle disclosed herein, wherein the method comprises: (i) providing a cell comprising the viral genome comprising a FXN-encoding sequence and a nucleic acid encoding an AAV capsid variant; and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant; thereby making the AAV particle. In some embodiments, the viral genome comprises (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; or (d) the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and wherein the AAV capsid variant comprises (i) the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); (ii) the amino acid sequence of SEQ ID NO: 738 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); or (iii) the amino acid sequence of SEQ ID NO: 739 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). In some
Attorney Docket No.14640.0092-00304 embodiments, the viral genome comprises (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; or (d) the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982, the amino acid sequence of SEQ ID NO: 738, and/or the amino acid sequence of SEQ ID NO: 739. [050] In some embodiments of the method of making an AAV particle, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797 and the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982, the amino acid sequence of SEQ ID NO: 738, and/or the amino acid sequence of SEQ ID NO: 739. [051] In some embodiments of the method of making an AAV particle, the method further comprises, prior to step (i), introducing a nucleic acid molecule comprising the viral genome into the cell. In some embodiments, the method further comprises, prior to step (i), introducing the nucleic acid encoding the AAV capsid variant into the cell. In some embodiments, the cell comprises a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial cell. [052] In some aspects, the present disclosure provides a pharmaceutical composition comprising an AAV particle disclosed herein and a pharmaceutically acceptable excipient. [053] In some aspects, the present disclosure provides a method of delivering an AAV particle encoding a frataxin (FXN) protein to a cell, comprising administering an effective amount of a pharmaceutical composition or AAV particle described herein. In some embodiments, the cell is in a subject. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency. In some embodiments, the disorder is Friedreich’s Ataxia (FA). [054] In some aspects, the present disclosure provides a method of delivering an AAV particle encoding a frataxin (FXN) protein to a subject, comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle described herein. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency. In some embodiments, the disorder is Friedreich’s Ataxia (FA). [055] In some aspects, the present disclosure provides a method of treating a subject having or diagnosed with having a disorder associated with frataxin (FXN) deficiency, comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle described herein. In
Attorney Docket No.14640.0092-00304 some embodiments, the subject has, has been diagnosed with having, or is at risk of having Friedreich’s Ataxia (FA). [056] In some aspects, the present disclosure provides a method of treating a subject having or diagnosed with having a disorder, wherein the disorder is Friedreich’s Ataxia (FA), comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle described herein. [057] In some embodiments of a method of treating, the treating results in prevention of progression of the disorder in the subject. In some embodiments, the treating results in amelioration of at least one symptom of the disorder. In some embodiments, the at least one symptom comprises impaired sensory functions, impaired motor function (e.g., ataxia and/or involuntary movements), fatigue, chronic pain, seizures, impaired speech, sleep disturbances, metabolic disorders (e.g., diabetes), and/or increased spasticity. In some embodiments, the treating stabilizes, slows the progression of, or improves the subject’s disorder as determined by the modified Friedreich Ataxia Rating Scale (mFARS), the Scale for the Assessment and Rating of Ataxia (SARA), and/or the International Cooperative Ataxia Rating Scale (ICARS). In some embodiments, the treatment slows the subject’s progression of the disorder as measured by mFARS, SARA, and/or ICARS relative to an individual with the disorder who has not been administered the pharmaceutical composition or the AAV particle. [058] In some embodiments of a method of delivering or treating, the subject is a human. In some embodiments, the AAV particle or the pharmaceutical composition is delivered to a cell or tissue of the central nervous system (CNS) in the subject. In some embodiments, the cell or tissue of the CNS is a cell or tissue of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum, or a combination thereof. [059] In some embodiments of a method of delivering or treating, the AAV particle or the pharmaceutical composition is delivered to the subject via intravenous administration. [060] In some embodiments of a method of delivering or treating, the method further comprises evaluating, e.g., measuring, the level of FXN expression, e.g., FXN gene expression, FXN mRNA expression, and/or FXN protein expression, in the subject, e.g., in a cell, tissue, or fluid of the subject. In some embodiments, the level of FXN protein expression is measured by an enzyme-linked immunosorbent assay (ELISA), a Western blot, an immunohistochemistry assay, or a frataxin biofluid assay. In some embodiments, evaluating the subject’s level of FXN expression is performed before and/or after administration of the pharmaceutical composition or AAV particle. In some embodiments, the subject’s level of FXN expression before administration is compared to the subject’s level of FXN expression after administration. [061] In some embodiments, the method of delivering or treating further comprises evaluating the level of FXN expression in a cell or tissue of the CNS. In some embodiments, the cell or tissue of the CNS is a cell or tissue of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen,
Attorney Docket No.14640.0092-00304 caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum. [062] In some embodiments, the method of delivering or treating further comprises evaluating the level of FXN expression in a peripheral cell or tissue. In some embodiments, the peripheral cell or tissue is a cell or tissue of the heart and/or muscle. [063] In some embodiments, the subject’s level of FXN protein expression after administration is increased relative to the subject’s level of FXN protein expression before administration. [064] In some embodiments, the method of delivering or treating further comprises evaluating, e.g., measuring, the level of FXN protein activity in the subject. [065] In some embodiments of a method of delivering or treating, administering the pharmaceutical composition or AAV particle to the subject results in an increase in: (i) the level of FXN protein or FXN gene expression in a cell, tissue, (e.g., a cell or tissue of the CNS, e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum), and/or fluid (e.g., CSF and/or serum), of the subject relative to baseline; (ii) the number and/or level of viral genomes (VG) per cell in a CNS tissue (e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) of the subject relative to the number and/or level of VG per cell in a peripheral tissue of the subject; and/or (iii) the level of FXN activity in a cell, tissue, (e.g., a cell or tissue of the CNS, e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) of the subject relative to baseline and/or relative to FXN activity in a cell, tissue, or fluid of an individual with a disorder associated with FXN deficiency who has not been administered the pharmaceutical composition or AAV particle. [066] In some embodiments of a method of delivering or treating, the method further comprises administering to the subject at least one additional therapeutic agent and/or therapy. In some embodiments, the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy suitable for treating a disorder associated with FXN deficiency (e.g., Friedreich’s Ataxia). In some embodiments, the at least one additional therapeutic agent and/or therapy comprises omaveloxolone or idebenone. [067] In some embodiments of a method of delivering or treating, the method further comprises administering an immunosuppressant to the subject. In some embodiments, the immunosuppressant comprises a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine. [068] In some aspects, the present disclosure provides a pharmaceutical composition or AAV particle disclosed herein for use in a method of treating a disorder disclosed herein.
Attorney Docket No.14640.0092-00304 [069] In some aspects, the present disclosure provides a pharmaceutical composition or AAV particle disclosed herein for use in the treatment of a disorder associated with FXN deficiency in a subject. In some embodiments, the disorder is Friedreich’s Ataxia (FA). In some embodiments, the subject has, has been diagnosed with having, or is at risk of having FA. [070] In some aspects, the present disclosure provides a use of a pharmaceutical composition or AAV particle disclosed herein in the manufacture of a medicament for the treatment of a disorder associated with FXN deficiency in a subject. In some embodiments, the disorder is Friedreich’s Ataxia (FA). In some embodiments, the subject has, has been diagnosed with having, or is at risk of having FA. Enumerated Embodiments 1. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises an amino sequence comprising the following formula: [N2]-[N3], wherein: (i) [N2] comprises X1, X2, X3, X4, and X5, wherein: (a) X1 is Y, N, C, or T; (b) X2 is P, E, K, T, or Q; (c) X3 is A or P; (d) X4 is E, S, D, or A; and (e) X5 is V, L, or E; and (ii) [N3] comprises the amino acid sequence of VQK, VQN, EQK, VKK, VHK, VQQ, or LQK. 2. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises an amino sequence comprising the following formula: [N2]-[N3], wherein: (i) [N2] comprises X1, X2, X3, X4, and X5, wherein: (a) X1 is Y, N, or C; (b) X2 is P, K, T, or Q; (c) X3 is A or P; (d) X4 is E, S, or A; and (e) X5 is V, L, or E; and (ii) [N3] comprises the amino acid sequence of VQK, EQK, VKK, VHK, VQQ, or LQK.
Attorney Docket No.14640.0092-00304 3. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises one, two, three, four, or all of: (i) an [N0] comprising TNN, TNT, INN, TNS, NNN, or TNK; (ii) an [N1] comprising QSS, QSK, TSL, SSS, QSR, AGA, IGS, QAS, ASS, LGS, QST, HSS, LSS, or QRS; (iii) an [N2] comprising YPAEV (SEQ ID NO: 1), YPPSL (SEQ ID NO: 2), NKAEV (SEQ ID NO: 3), YTAEV (SEQ ID NO: 4), YPAEE (SEQ ID NO: 5), YQAEV (SEQ ID NO: 6), YTPSL (SEQ ID NO: 7), YPAAV (SEQ ID NO: 8), NPAEV (SEQ ID NO: 9), CPAEV (SEQ ID NO: 10), or YQAEE (SEQ ID NO: 11); (iv) an [N3] comprising VQK, EQK, VKK, VHK, VQQ, or LQK; and (v) an [N4] comprising TA, PA, or NA. 4. The AAV particle of embodiment 1 or 2, wherein: (a) X1 is Y or N; (b) X2 is P, T or Q; (c) X3 is A; (d) X4 is E or S; and/or (e) X5 is V or L. 5. The AAV particle of any one of embodiments 1, 2, and 4, wherein [N2] comprises YP, NK, YT, YQ, NP, CP, TH, AE, PS, AA, AS, PA, PP, KA, TA, QA, TP, HA, EV, SL, EE, AV, or SH. 6. The AAV particle any one of embodiments 1, 2, 4, and 5, wherein [N2] comprises YPA, YPP, NKA, YTA, YQA, YTP, NPA, CPA, THA, PAE, PPS, KAE, TAE, QAE, TPS, PAA, HAS, AEV, PSL, AEE, or AAV. 7. The AAV particle of any one of embodiments 1, 2, 4, and 6, wherein [N2] comprises YPAE (SEQ ID NO: 21), YPPS (SEQ ID NO: 22), NKAE (SEQ ID NO: 23), YTAE (SEQ ID NO: 24), YQAE (SEQ ID NO: 25), YTPS (SEQ ID NO: 26), YPAA (SEQ ID NO: 27), NPAE (SEQ ID NO: 28), CPAE (SEQ ID NO: 29), THAS (SEQ ID NO: 30), PAEV (SEQ ID NO: 17), PPSL (SEQ ID NO: 31), KAEV (SEQ ID NO: 32), TAEV (SEQ ID NO: 16), PAEE (SEQ ID NO: 18), QAEV (SEQ ID NO: 15), TPSL (SEQ ID NO: 33), PAAV (SEQ ID NO: 34), or QAEE (SEQ ID NO: 35). 8. The AAV particle of any one of embodiments 1-7, wherein [N2] is or comprises YPAEV (SEQ ID NO: 1), YPPSL (SEQ ID NO: 2), NKAEV (SEQ ID NO: 3), YTAEV (SEQ ID NO: 4), YPAEE (SEQ ID
Attorney Docket No.14640.0092-00304 NO: 5), YQAEV (SEQ ID NO: 6), YTPSL (SEQ ID NO: 7), YPAAV (SEQ ID NO: 8), NPAEV (SEQ ID NO: 9), CPAEV (SEQ ID NO: 10), or YQAEE (SEQ ID NO: 11). 9. The AAV particle of any one of embodiments 1, 2, 4, and 8, wherein [N3] comprises the amino acid sequence of VQK, EQK, or VKK. 10. The AAV particle of embodiment 1 or 2 or any one of embodiments 4-9, wherein [N3] comprises VQK. 11. The AAV particle of embodiment 1 or 2 or any one of embodiments 4-9, wherein [N3] comprises EQK. 12. The AAV particle of embodiment 1 or 2 or any one of embodiments 4-9, wherein [N3] comprises VKK. 13. The AAV particle of any one of embodiments 1-12, wherein [N2] is or comprises the amino acid sequence of YPAEV (SEQ ID NO: 1) and [N3] is or comprises the amino acid sequence of VQK. 14. The AAV particle of any one of embodiments 1-12, wherein: (i) [N2] is or comprises the amino acid sequence of YTPSL (SEQ ID NO: 7) and [N3] is or comprises the amino acid sequence of VQK; (ii) [N2] is or comprises the amino acid sequence of YPPSL (SEQ ID NO: 2) and [N3] is or comprises the amino acid sequence of VQK; (iii) [N2] is or comprises the amino acid sequence of YPPSL (SEQ ID NO: 2) and [N3] is or comprises the amino acid sequence of EQK; or (iv) [N2] is or comprises the amino acid sequence of YPPSL (SEQ ID NO: 2) and [N3] is or comprises the amino acid sequence of VKK. 15. The AAV particle of embodiment 1 or 2 or any one of embodiments 4-14, wherein [N2]-[N3] comprises: (i) AEVVQK (SEQ ID NO: 36), PSLVQK (SEQ ID NO: 37), AEVEQK (SEQ ID NO: 38), AEEVQK (SEQ ID NO: 39), PSLEQK (SEQ ID NO: 40), PSLVKK (SEQ ID NO: 41), AEVVKK (SEQ ID NO: 42), AEVVHK (SEQ ID NO: 43), AAVVQK (SEQ ID NO: 44), AEVVQQ (SEQ ID NO: 45), or AEVLQK (SEQ ID NO: 46); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
Attorney Docket No.14640.0092-00304 (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 16. The AAV particle of embodiment 1 or 2 or any one of embodiments 4-15, wherein [N2]-[N3] comprises: (i) PAEVVQK (SEQ ID NO: 20), PPSLVQK (SEQ ID NO: 47), KAEVVQK (SEQ ID NO: 48), TAEVVQK (SEQ ID NO: 49), PAEVEQK (SEQ ID NO: 50), PAEEVQK (SEQ ID NO: 51), QAEVVQK (SEQ ID NO: 52), TPSLVQK (SEQ ID NO: 53), PPSLEQK (SEQ ID NO: 54), PPSLVKK (SEQ ID NO: 55), PAEVVKK (SEQ ID NO: 56), PAEVVHK (SEQ ID NO: 57), PAAVVQK (SEQ ID NO: 58), PAEVVQQ (SEQ ID NO: 59), TAEVVKK (SEQ ID NO: 60), PAEVLQK (SEQ ID NO: 61), or QAEEVQK (SEQ ID NO: 62); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 17. The AAV particle of any one of embodiments 1-16, wherein [N2]-[N3] is or comprises: (i) YPAEVVQK (SEQ ID NO: 943), YPPSLVQK (SEQ ID NO: 946), NKAEVVQK (SEQ ID NO: 947), YTAEVVQK (SEQ ID NO: 948), YPAEVEQK (SEQ ID NO: 949), YPAEEVQK (SEQ ID NO: 950), YQAEVVQK (SEQ ID NO: 951), YTPSLVQK (SEQ ID NO: 952), YPPSLEQK (SEQ ID NO: 953), YPPSLVKK (SEQ ID NO: 954), YPAEVVKK (SEQ ID NO: 955), YPAEVVHK (SEQ ID NO: 956), YPAAVVQK (SEQ ID NO: 957), NPAEVVQK (SEQ ID NO: 958), YPAEVVQQ (SEQ ID NO: 959), CPAEVVQK (SEQ ID NO: 960), YTAEVVKK (SEQ ID NO: 961), YPAEVLQK (SEQ ID NO: 962), or YQAEEVQK (SEQ ID NO: 963); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, or 7 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 18. The AAV particle of any one of embodiments 1-17, which further comprises one, two, three or all of an amino acid other than Q at position 574 (e.g., T, S, A, I, L, or H), an amino acid other than S at
Attorney Docket No.14640.0092-00304 position 575 (e.g., G, A, or R), and/or an amino acid other than S at position 576 (e.g., K, L, R, A, or T), numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 19. The AAV particle of any one of embodiments 1-17, which further comprises: (i) a Q at position 574, an S at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (ii) a T at position 574, an S at position 575, and/or a L at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iii) an S at position 574, an S at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iv) a Q at position 574, an S at position 575, and/or an R at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (v) a Q at position 574, an S at position 575, and/or a K at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (vi) an A at position 574, a G at position 575, and/or an A at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (vii) an I at position 574, a G at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (viii) a Q at position 574, an A at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (ix) an A at position 574, an S at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (x) an L at position 574, a G at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (xi) a Q at position 574, an S at position 575, and/or a T at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (xii) an H at position 574, an S at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (xiii) an L at position 574, an S at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; or (xiv) a Q at position 574, an R at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 20. The AAV particle of any one of embodiments 1-19, which further comprises [N1], wherein [N1] comprises XD, XE, and XF, wherein: (a) XD is Q, T, S, A, I, L, or H; (b) XE is S, G, A, or R; and
Attorney Docket No.14640.0092-00304 (c) XF is S, K, L, R, A, or T; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c). 21. The AAV particle of embodiment 20, wherein [N1] comprises SK, SL, SS, SR, GA, GS, AS, ST, RS, QS, TS, AG, IG, QA, LG, HS, LS, or QR. 22. The AAV particle of any one of embodiments 3, 20, and 21, wherein [N1] is or comprises QSS, QSK, TSL, SSS, QSR, AGA, IGS, QAS, ASS, LGS, QST, HSS, LSS, or QRS. 23. The AAV particle of any one of embodiments 20-22, wherein [N1]-[N2] comprises: (i) SSYPA (SEQ ID NO: 63), SKYPA (SEQ ID NO: 64), SLYPA (SEQ ID NO: 65), SRYPA (SEQ ID NO: 66), SSYPP (SEQ ID NO: 67), GAYPA (SEQ ID NO: 68), GSYPA (SEQ ID NO: 69), ASYPA (SEQ ID NO: 70), STNKA (SEQ ID NO: 71), SSYTA (SEQ ID NO: 72), SSYQA (SEQ ID NO: 73), SSYTP (SEQ ID NO: 74), SSNPA (SEQ ID NO: 75), SLCPA (SEQ ID NO: 76), RSYTA (SEQ ID NO: 77), or SSTHA (SEQ ID NO: 78); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 24. The AAV particle of any one of embodiments 20-23, wherein [N1]-[N2] comprises: (i) SSYPAE (SEQ ID NO: 79), SKYPAE (SEQ ID NO: 80), SLYPAE (SEQ ID NO: 81), SRYPAE (SEQ ID NO: 82), SSYPPS (SEQ ID NO: 83), GAYPAE (SEQ ID NO: 84), GSYPAE (SEQ ID NO: 85), ASYPAE (SEQ ID NO: 86), STNKAE (SEQ ID NO: 87), SSYTAE (SEQ ID NO: 88), SSYQAE (SEQ ID NO: 89), SSYTPS (SEQ ID NO: 90), SSYPAA (SEQ ID NO: 91), SSNPAE (SEQ ID NO: 92), SLCPAE (SEQ ID NO: 93), RSYTAE (SEQ ID NO: 94), SSTHAS (SEQ ID NO: 95); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
Attorney Docket No.14640.0092-00304 25. The AAV particle of embodiment 3 or any one of embodiments 20-24, wherein [N1]-[N2] is or comprises: (i) QSSYPAEV (SEQ ID NO: 96), QSKYPAEV (SEQ ID NO: 97), TSLYPAEV (SEQ ID NO: 98), SSSYPAEV (SEQ ID NO: 99), QSRYPAEV (SEQ ID NO: 100), QSSYPPSL (SEQ ID NO: 101), AGAYPAEV (SEQ ID NO: 102), IGSYPAEV (SEQ ID NO: 103), QASYPAEV (SEQ ID NO: 104), ASSYPAEV (SEQ ID NO: 105), LGSYPAEV (SEQ ID NO: 106), QSTNKAEV (SEQ ID NO: 107), HSSYPAEV (SEQ ID NO: 108), SSSYTAEV (SEQ ID NO: 109), TSLYPAEE (SEQ ID NO: 110), ASSYQAEV (SEQ ID NO: 111), QSSYTPSL (SEQ ID NO: 112), QSRYPAEE (SEQ ID NO: 113), LSSYQAEV (SEQ ID NO: 114), HSSYPAAV (SEQ ID NO: 115), QSSNPAEV (SEQ ID NO: 116), QSSYTAEV (SEQ ID NO: 117), TSLCPAEV (SEQ ID NO: 118), QRSYTAEV (SEQ ID NO: 119), or QSSYQAEE (SEQ ID NO: 120); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, or 7 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 26. The AAV particle of any one of embodiments 20-25, wherein [N1]-[N2]-[N3] comprises: (i) SSYPAEVVQ (SEQ ID NO: 121), SKYPAEVVQ (SEQ ID NO: 122), SLYPAEVVQ (SEQ ID NO: 123), SRYPAEVVQ (SEQ ID NO: 124), SSYPPSLVQ (SEQ ID NO: 125), GAYPAEVVQ (SEQ ID NO: 126), GSYPAEVVQ (SEQ ID NO: 127), ASYPAEVVQ (SEQ ID NO: 128), STNKAEVVQ (SEQ ID NO: 129), SSYTAEVVQ (SEQ ID NO: 130), SKYPAEVEQ (SEQ ID NO: 131), SLYPAEEVQ (SEQ ID NO: 132), SSYQAEVVQ (SEQ ID NO: 133), SSYTPSLVQ (SEQ ID NO: 134), SRYPAEEVQ (SEQ ID NO: 135), SSYPPSLEQ (SEQ ID NO: 136), SSYPPSLVK (SEQ ID NO: 140), SSYPAEVVK (SEQ ID NO: 141), SKYPAEVVH (SEQ ID NO: 142), SSYPAAVVQ (SEQ ID NO: 143), SSNPAEVVQ (SEQ ID NO: 144), SLCPAEVVQ (SEQ ID NO: 145), RSYTAEVVQ (SEQ ID NO: 146), SSYTAEVVK (SEQ ID NO: 147), SSYPAEVLQ (SEQ ID NO: 148), or SSYQAEEVQ (SEQ ID NO: 149); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, or 8 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
Attorney Docket No.14640.0092-00304 27. The AAV particle of embodiment 3 or any one of embodiments 20-26, wherein [N1]-[N2]-[N3] is or comprises: (i) QSSYPAEVVQK (SEQ ID NO: 150), QSKYPAEVVQK (SEQ ID NO: 151), TSLYPAEVVQK (SEQ ID NO: 152), SSSYPAEVVQK (SEQ ID NO: 153), QSRYPAEVVQK (SEQ ID NO: 154), QSSYPPSLVQK (SEQ ID NO: 155), AGAYPAEVVQK (SEQ ID NO: 156), IGSYPAEVVQK (SEQ ID NO: 157), QASYPAEVVQK (SEQ ID NO: 158), ASSYPAEVVQK (SEQ ID NO: 159), LGSYPAEVVQK (SEQ ID NO: 160), QSTNKAEVVQK (SEQ ID NO: 161), HSSYPAEVVQK (SEQ ID NO: 162), SSSYTAEVVQK (SEQ ID NO: 163), QSKYPAEVEQK (SEQ ID NO: 164), TSLYPAEEVQK (SEQ ID NO: 165), ASSYQAEVVQK (SEQ ID NO: 166), QSSYTPSLVQK (SEQ ID NO: 167), QSRYPAEEVQK (SEQ ID NO: 168), QSSYPPSLEQK (SEQ ID NO: 169), QSSYPPSLVKK (SEQ ID NO: 170), LSSYQAEVVQK (SEQ ID NO: 171), SSSYPAEVVKK (SEQ ID NO: 172), QSKYPAEVVHK (SEQ ID NO: 173), HSSYPAAVVQK (SEQ ID NO: 174), QSSNPAEVVQK (SEQ ID NO: 175), SSSYPAEVVQQ (SEQ ID NO: 176), QSSYTAEVVQK (SEQ ID NO: 177), TSLCPAEVVQK (SEQ ID NO: 178), QRSYTAEVVQK (SEQ ID NO: 179), QSSYTAEVVKK (SEQ ID NO: 180), HSSYPAEVLQK (SEQ ID NO: 181), or QSSYQAEEVQK (SEQ ID NO: 182); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 28. The AAV particle of any one of embodiments 1-27, which further comprises [N0], wherein [N0] comprises XA, XB, and XC, wherein: (a) XA is T, I, or N; (b) XB is N; (c) XC is N, T, S, or K; and optionally, wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c). 29. The AAV particle of embodiment 28, wherein [N0] comprises TN, IN, NN, NT, NS, or NK. 30. The AAV particle of embodiment 3, 28, or 29, wherein [N0] is or comprises TNN, TNT, INN, TNS, NNN, or TNK.
Attorney Docket No.14640.0092-00304 31. The AAV particle of embodiment 3 or any one of embodiments 28-30, wherein [N0]-[N1] is or comprises: (i) TNNQSS (SEQ ID NO: 183), TNNQSK (SEQ ID NO: 184), TNNTSL (SEQ ID NO: 185), TNNSSS (SEQ ID NO: 186), TNNQSR (SEQ ID NO: 187), TNNAGA (SEQ ID NO: 188), TNNIGS (SEQ ID NO: 189), TNNQAS (SEQ ID NO: 190), TNTASS (SEQ ID NO: 191), TNNLGS (SEQ ID NO: 192), TNNQST (SEQ ID NO: 193), TNNHSS (SEQ ID NO: 194), TNNQSK (SEQ ID NO: 184), TNNLSS (SEQ ID NO: 195), INNQSS (SEQ ID NO: 196), TNSQSS (SEQ ID NO: 197), NNNQSR (SEQ ID NO: 198), TNSTSL (SEQ ID NO: 199), TNNQRS (SEQ ID NO: 200), or TNKQAS (SEQ ID NO: 201); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 32. The AAV particle of embodiment 3 or any one of embodiments 28-31, wherein [N0]-[N1]-[N2]-[N3] is or comprises: (i) TNNQSSYPAEVVQK (SEQ ID NO: 500), TNNQSKYPAEVVQK (SEQ ID NO: 503), TNNTSLYPAEVVQK (SEQ ID NO: 506), TNNSSSYPAEVVQK (SEQ ID NO: 508), TNNQSRYPAEVVQK (SEQ ID NO: 510), TNNQSSYPPSLVQK (SEQ ID NO: 512), TNNAGAYPAEVVQK (SEQ ID NO: 513), TNNIGSYPAEVVQK (SEQ ID NO: 514), TNNQASYPAEVVQK (SEQ ID NO: 517), TNTASSYPAEVVQK (SEQ ID NO: 520), TNNLGSYPAEVVQK (SEQ ID NO: 523), TNNQSTNKAEVVQK (SEQ ID NO: 524), TNNHSSYPAEVVQK (SEQ ID NO: 525), TNNSSSYTAEVVQK (SEQ ID NO: 526), TNNQSKYPAEVEQK (SEQ ID NO: 529), TNNTSLYPAEEVQK (SEQ ID NO: 530), TNTASSYQAEVVQK (SEQ ID NO: 531), TNNQSSYTPSLVQK (SEQ ID NO: 533), TNNQSRYPAEEVQK (SEQ ID NO: 534), TNNQSSYPPSLEQK (SEQ ID NO: 535), TNNQSSYPPSLVKK (SEQ ID NO: 536), TNNLSSYQAEVVQK (SEQ ID NO: 539), TNNSSSYPAEVVKK (SEQ ID NO: 540), TNNQSKYPAEVVHK (SEQ ID NO: 542), INNQSSYPAEVVQK (SEQ ID NO: 543), TNNHSSYPAAVVQK (SEQ ID NO: 545), TNSQSSNPAEVVQK (SEQ ID NO: 548), TNNSSSYPAEVVQQ (SEQ ID NO: 551), NNNQSRYPAEVVQK (SEQ ID NO: 552), TNNQSSYTAEVVQK (SEQ ID NO: 553), TNNTSLCPAEVVQK (SEQ ID NO: 554), TNSTSLYPAEVVQK (SEQ ID NO: 556), TNNQRSYTAEVVQK (SEQ ID NO: 557), TNNQSSYTAEVVKK (SEQ ID NO: 558), TNNHSSYPAEVLQK (SEQ ID NO: 560), TNNQSSYQAEEVQK (SEQ ID NO: 562), or TNKQASYPAEVVQK (SEQ ID NO: 563);
Attorney Docket No.14640.0092-00304 (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 33. The AAV particle of embodiment 3 or 32, wherein [N0]-[N1]-[N2]-[N3] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500). 34. The AAV particle of embodiment 3 or 32, wherein [N0]-[N1]-[N2]-[N3] is or comprises TNNAGAYPAEVVQK (SEQ ID NO: 513), TNNTSLYPAEVVQK (SEQ ID NO: 506), TNNQSKYPAEVVQK (SEQ ID NO: 503), TNNQSSYTPSLVQK (SEQ ID NO: 533), TNNQSSYPPSLVQK (SEQ ID NO: 512), TNNQSRYPAEVVQK (SEQ ID NO: 510), TNNQSSYPPSLEQK (SEQ ID NO: 535), TNNQSSYPPSLVKK (SEQ ID NO: 536), or INNQSSYPAEVVQK (SEQ ID NO: 543). 35. The AAV particle of any one of embodiments 1-34, which further comprises [N4], wherein [N4] comprises XG and XH, wherein: (a) XG is T, P, or N; and (b) XH is A; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a) or (b). 36. The AAV particle of embodiment 35, wherein [N4] is or comprises TA, PA, or NA. 37. The AAV particle of any one of embodiments 3, 35, and 36, wherein [N3]-[N4] is or comprises: (i) VQKTA (SEQ ID NO: 564), EQKTA (SEQ ID NO: 565), VKKTA (SEQ ID NO: 566),
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
Attorney Docket No.14640.0092-00304 38. The AAV particle of embodiment 3 or any one of embodiments 35-37, wherein [N0]-[N1]-[N2]- [N3]-[N4] is or comprises: (i) TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNTSLYPAEVVQKTA (SEQ ID NO: 1232), TNNSSSYPAEVVQKTA (SEQ ID NO: 1539), TNNQSRYPAEVVQKTA (SEQ ID NO: 1327), TNNQSSYPPSLVQKTA (SEQ ID NO: 1300), TNNAGAYPAEVVQKTA (SEQ ID NO: 1021), TNNIGSYPAEVVQKTA (SEQ ID NO: 1112), TNNQASYPAEVVQKTA (SEQ ID NO: 1194), TNTASSYPAEVVQKTA (SEQ ID NO: 1575), TNNLGSYPAEVVQKTA (SEQ ID NO: 1027), TNNQSTNKAEVVQKTA (SEQ ID NO: 1578), TNNHSSYPAEVVQKTA (SEQ ID NO: 1310), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNSSSYTAEVVQKTA (SEQ ID NO: 1214), TNNQSKYPAEVEQKTA (SEQ ID NO: 1254), TNNTSLYPAEEVQKTA (SEQ ID NO: 1583), TNTASSYQAEVVQKTA (SEQ ID NO: 1584), TNNQSSYTPSLVQKTA (SEQ ID NO: 1585), TNNQSRYPAEEVQKTA (SEQ ID NO: 1342), TNNQSSYPPSLEQKTA (SEQ ID NO: 1590), TNNQSSYPPSLVKKTA (SEQ ID NO: 1591), TNNLSSYQAEVVQKTA (SEQ ID NO: 1592), TNNQSSYPPSLVQKPA (SEQ ID NO: 1593), TNNSSSYPAEVVKKTA (SEQ ID NO: 1331), TNNQSKYPAEVVHKTA (SEQ ID NO: 1453), TNNSSSYPAEVVQKPA (SEQ ID NO: 1142), INNQSSYPAEVVQKTA (SEQ ID NO: 1024), TNNHSSYPAAVVQKTA (SEQ ID NO: 1598), TNSQSSNPAEVVQKTA (SEQ ID NO: 1599), TNNSSSYPAEVVQQTA (SEQ ID NO: 1419), NNNQSRYPAEVVQKTA (SEQ ID NO: 1601), TNNQSSYTAEVVQKNA (SEQ ID NO: 1602), TNNTSLCPAEVVQKTA (SEQ ID NO: 1603), TNSTSLYPAEVVQKTA (SEQ ID NO: 1605), TNNQRSYTAEVVQKTA (SEQ ID NO: 1604), TNNQSSYTAEVVKKTA (SEQ ID NO: 1606), TNNHSSYPAEVLQKTA (SEQ ID NO: 1607), TNNQSSYQAEEVQKTA (SEQ ID NO: 1608), , TNKQASYPAEVVQKTA (SEQ ID NO: 1587); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 39. The AAV particle of embodiment 3 or any one of embodiments 35-38, wherein [N0]-[N1]-[N2]- [N3]-[N4] is or comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). 40. The AAV particle of embodiment 3 or any one of embodiments 35-38, wherein [N0]-[N1]-[N2]- [N3]-[N4] is or comprises TNNAGAYPAEVVQKTA (SEQ ID NO: 1021), TNNTSLYPAEVVQKTA (SEQ ID NO: 1232), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNQSSYTPSLVQKTA (SEQ ID NO: 1585), TNNQSSYPPSLVQKTA (SEQ ID NO: 1300), TNNQSRYPAEVVQKTA (SEQ ID NO:
Attorney Docket No.14640.0092-00304 1327), TNNQSSYPPSLEQKTA (SEQ ID NO: 1590), TNNQSSYPPSLVKKTA (SEQ ID NO: 1591), or INNQSSYPAEVVQKTA (SEQ ID NO: 1024). 41. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises an amino sequence comprising the following formula: [B]-[C] (SEQ ID NO: 2113), wherein (i) [B] comprises X1, X2, and X3, wherein: (a) X1 is Q, T, S, A, I, L, or H; (b) X2 is S, G, or A; and (c) X3 is S, K, L, R, or A; and (ii) [C] comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). 42. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises one, two, three, four, or all of: (i) an [A], wherein [A] comprises the amino acid sequence of TNN, TNT, INN, NNN, TNS, or TNK; (ii) a [B], wherein [B] comprises the amino acid sequence of QSS, TSL, SSS, QSR, QSK, AGA, IGS, QAS, ASS, LGS, or HSS; (iii) a [C], wherein [C] comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943); and (iv) a [D], wherein [D] comprises the amino acid sequence of TA or PA; and optionally wherein [C] replaces position 577 relative to a reference sequence numbered according to the amino acid sequence of SEQ ID NO: 138. 43. The AAV particle of embodiment 41, wherein: (a) X1 is Q, T, S, A, or H; (b) X2 is S or G; and (c) X3 is S, K, L, or R. 44. The AAV particle of embodiment 41 or 43, wherein [B] comprises QS, TS, SS, AG, IG, QA, AS, LG, HS, SK, SL, SR, GA, or GS. 45. The AAV particle of any one of embodiments 41-44, wherein [B] is or comprises QSS, TSL, SSS, QSR, QSK, AGA, IGS, QAS, ASS, LGS, or HSS.
Attorney Docket No.14640.0092-00304 46. The AAV particle of embodiment 41 or any one of embodiments 43-45, wherein [B]-[C] comprises: (i) SSYPAEVVQK (SEQ ID NO: 572), SKYPAEVVQK (SEQ ID NO: 573), SLYPAEVVQK (SEQ ID NO: 574), SRYPAEVVQK (SEQ ID NO: 575), GAYPAEVVQK (SEQ ID NO: 576), GSYPAEVVQK (SEQ ID NO: 580), or ASYPAEVVQK (SEQ ID NO: 582); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 47. The AAV particle of any one of embodiments 41-46, wherein [B]-[C] is or comprises: (i) QSSYPAEVVQK (SEQ ID NO: 150), QSKYPAEVVQK (SEQ ID NO: 151), TSLYPAEVVQK (SEQ ID NO: 152), SSSYPAEVVQK (SEQ ID NO: 153), QSRYPAEVVQK (SEQ ID NO: 154), AGAYPAEVVQK (SEQ ID NO: 156), IGSYPAEVVQK (SEQ ID NO: 157), QASYPAEVVQK (SEQ ID NO: 158), ASSYPAEVVQK (SEQ ID NO: 159), LGSYPAEVVQK (SEQ ID NO: 160), or HSSYPAEVVQK (SEQ ID NO: 162); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 48. The AAV particle of any one of embodiments 41-47, wherein [B]-[C] is or comprises QSSYPAEVVQK (SEQ ID NO: 150). 49. The AAV particle of any one of embodiments embodiment 41-47, wherein [B]-[C] is or comprises AGAYPAEVVQK (SEQ ID NO: 156), TSLYPAEVVQK (SEQ ID NO: 152), QSKYPAEVVQK (SEQ ID NO: 151), or QSRYPAEVVQK (SEQ ID NO: 154). 50. The AAV particle of any one of embodiments 1-49, which further comprises one or both of an amino acid other than T at position 571 (e.g., I or N), and/or an amino acid other than N at position 573 (e.g., T, S, or K), relative to a reference sequence numbered according to the amino acid sequence of SEQ ID NO: 138 or 982.
Attorney Docket No.14640.0092-00304 51. The AAV particle of any one of embodiments 1-49, which further comprises: (i) a T at position 571, an N at position 572, and/or an N at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (ii) a T at position 571, an N at position 572, and/or a T at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iii) an I at position 571, an N at position 572, and/or an N at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iv) a T at position 571, an N at position 572, and/or an S at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (v) an N at position 571, an N at position 572, and/or an N at position 573, according to the amino acid sequence of SEQ ID NO: 138 or 982; or (vi) a T at position 571, an N at position 572, and/or a K at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 52. The AAV particle of any one of embodiments 41-51, which further comprises [A], wherein [A] comprises XA, XB, and XC, wherein: (a) XA is T, I, or N; (b) XB is N; and (c) XC is N, T, S, or K; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c). 53. The AAV particle of embodiment 52, wherein [A] comprises TN, IN, NN, NT, NS, or NK. 54. The AAV particle of any one of embodiments 42, 52, and 53, wherein [A] is or comprises TNN, TNT, INN, NNN, TNS, or TNK. 55. The AAV particle of embodiment 42 or any one of embodiments 52-54, wherein [A]-[B] is or comprises: (i) TNNQSS (SEQ ID NO: 183), TNNQSK (SEQ ID NO: 184), TNNTSL (SEQ ID NO: 185), TNNSSS (SEQ ID NO: 186), TNNQSR (SEQ ID NO: 187), TNNAGA (SEQ ID NO: 188), TNNIGS (SEQ ID NO: 189), TNNQAS (SEQ ID NO: 190), TNTASS (SEQ ID NO: 191), TNNLGS (SEQ ID NO: 192), TNNHSS (SEQ ID NO: 194), INNQSS (SEQ ID NO: 196), NNNQSR (SEQ ID NO: 198), TNSTSL (SEQ ID NO: 199), or TNKQAS (SEQ ID NO: 201); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof;
Attorney Docket No.14640.0092-00304 (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 56. The AAV particle of embodiment 42 or any one of embodiments 52-55, wherein [A]-[B]-[C] is or comprises: (i) TNNQSSYPAEVVQK (SEQ ID NO: 500), TNNQSKYPAEVVQK (SEQ ID NO: 503), TNNTSLYPAEVVQK (SEQ ID NO: 506), TNNSSSYPAEVVQK (SEQ ID NO: 508), TNNQSRYPAEVVQK (SEQ ID NO: 510), TNNAGAYPAEVVQK (SEQ ID NO: 513), TNNIGSYPAEVVQK (SEQ ID NO: 514), TNNQASYPAEVVQK (SEQ ID NO: 517), TNTASSYPAEVVQK (SEQ ID NO: 520), TNNLGSYPAEVVQK (SEQ ID NO: 523), TNNHSSYPAEVVQK (SEQ ID NO: 525), INNQSSYPAEVVQK (SEQ ID NO: 543), NNNQSRYPAEVVQK (SEQ ID NO: 552), TNSTSLYPAEVVQK (SEQ ID NO: 556), or TNKQASYPAEVVQK (SEQ ID NO: 563); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 57. The AAV particle of embodiment 42 or any one of embodiments 52-54, wherein [A]-[B]-[C] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500). 58. The AAV particle of embodiment 42 or any one of embodiments 52-54, wherein [A]-[B]-[C] is or comprises TNNAGAYPAEVVQK (SEQ ID NO: 513), TNNTSLYPAEVVQK (SEQ ID NO: 506), TNNQSKYPAEVVQK (SEQ ID NO: 503), TNNQSRYPAEVVQK (SEQ ID NO: 510), or INNQSSYPAEVVQK (SEQ ID NO: 543). 59. The AAV particle of any one of embodiments 1-58, which further comprises: (i) an amino acid other than T at position 578 (e.g., P or N), numbered according to the amino acid sequence of SEQ ID NO: 138; or (ii) an amino acid other than T at position 585 (e.g., P or N), numbered according to the amino acid sequence of SEQ ID NO: 982. 60. The AAV particle of any one of embodiments 1-58, which further comprises:
Attorney Docket No.14640.0092-00304 (i) a T at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; or a T at position 585 and/or an A at position 586 relative to a reference sequence numbered according to SEQ ID NO: 982; (ii) a P at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; or a P at position 585 and/or an A at position 586 numbered according to SEQ ID NO: 982; or (iii) an N at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; or an N at position 585 and/or an A at position 586 numbered according to SEQ ID NO: 982. 61. The AAV particle of any one of embodiments 41-60, which further comprises [D], wherein [D] comprises X4 and X5, wherein: (a) X4 is T or N; and (b) X5 is A. 62. The AAV particle of embodiment 42 or 61, wherein [D] is or comprises TA or PA. 63. The AAV particle of any one of embodiments 42, 61, and 62, wherein [C]-[D] is or comprises: (i) YPAEVVQKTA (SEQ ID NO: 584) or YPAEVVQKPA (SEQ ID NO: 586); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 64. The AAV particle of embodiment 42 or any one of embodiments 61-63, wherein [A]-[B]-[C]-[D] is or comprises: (i) TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNTSLYPAEVVQKTA (SEQ ID NO: 1232), TNNSSSYPAEVVQKTA (SEQ ID NO: 1539), TNNQSRYPAEVVQKTA (SEQ ID NO: 1327), TNNAGAYPAEVVQKTA (SEQ ID NO: 1021), TNNIGSYPAEVVQKTA (SEQ ID NO: 1112), TNNQASYPAEVVQKTA (SEQ ID NO: 1194), TNTASSYPAEVVQKTA (SEQ ID NO: 1575), TNNLGSYPAEVVQKTA (SEQ ID NO: 1027), TNNHSSYPAEVVQKTA (SEQ ID NO: 1310), TNNSSSYPAEVVQKPA (SEQ ID NO: 1142),
Attorney Docket No.14640.0092-00304 (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 65. The AAV particle of embodiment 42 or any one of embodiments 61-64, wherein [A]-[B]-[C]-[D] is or comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). 66. The AAV particle of embodiment 42 or any one of embodiments 61-64, wherein [A]-[B]-[C]-[D] is or comprises TNNAGAYPAEVVQKTA (SEQ ID NO: 1021), TNNTSLYPAEVVQKTA (SEQ ID NO: 1232), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNQSRYPAEVVQKTA (SEQ ID NO: 1327), or INNQSSYPAEVVQKTA (SEQ ID NO: 1024). 67. An AAV particle comprising a viral genome comprising a frataxin (FXN)-encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises an amino sequence comprising the formula [N2]-[N3], wherein: (i) [N2] comprises X1, X2, X3, X4, and X5, wherein: (a) X1 is Y or T; (b) X2 is Q, T, P, or E; (c) X3 is A; (d) X4 is E or D; and (e) X5 is V or E; and (ii) [N3] comprises the amino acid sequence of VQK or VQN. 68. An AAV particle comprising a viral genome encoding a frataxin (FXN)-encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises one, two, three, four, or all of: (i) an [N0] comprising TNN, TNS, TNT, or TNK; (ii) an [N1] comprising QSS, SLS, SLY, SAT, or QTS; (iii) an [N2] comprising YPAEV (SEQ ID NO: 1), YQAEV (SEQ ID NO: 6), YTAEV (SEQ ID NO: 4), YPAEE (SEQ ID NO: 5), TEAEV (SEQ ID NO: 12), or YPADV (SEQ ID NO: 13); (iv) an [N3] comprising VQK or VQN; and (v) an [N4] comprising TA, PA, TD, NA, or PA.
Attorney Docket No.14640.0092-00304 69. The AAV particle of embodiment 1 or 67, wherein [N2] comprises YP, YQ, YT, TE, QA, TA, PA, EA, EV, EE, DV, AE, or AD. 70. The AAV particle of any one of embodiments 1, 67, and 69, wherein [N2] comprises^YPA, YQA, YTA, TEA, QAE, TAE, PAE, EAE, PAD, AEV, AEE, or ADV. 71. The AAV particle of any one of embodiments 1, 67, 69, and 70, wherein [N2] comprises YPAE (SEQ ID NO: 21), YQAE (SEQ ID NO: 25), YTAE (SEQ ID NO: 24), TEAE (SEQ ID NO: 587), YPAD (SEQ ID NO: 588), QAEV (SEQ ID NO: 15), TAEV (SEQ ID NO: 16), PAEV (SEQ ID NO: 17), PAEE (SEQ ID NO: 18), EAEV (SEQ ID NO: 590), or PADV (SEQ ID NO: 19). 72. The AAV particle of embodiment 1 or any one of embodiments 67-71, wherein [N2] is or comprises
73. The AAV particle of embodiment 1 or any one of embodiments 67-72, wherein [N3] comprises the amino acid sequence of VQK. 74. The AAV particle of embodiment 1 or 67 or any one of embodiments 69-73, wherein [N2]-[N3] comprises: (i) AEVVQK (SEQ ID NO: 36), AEEVQK (SEQ ID NO: 39), AEVVQN (SEQ ID NO: 591), or ADVVQK (SEQ ID NO: 593); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 75. The AAV particle of embodiment 1 or 67 or any one of embodiments 69-74, wherein [N2]-[N3] comprises: (i) PAEVVQN (SEQ ID NO: 594), QAEVVQK (SEQ ID NO: 52), TAEVVQK (SEQ ID NO: 49), PAEVVQK (SEQ ID NO: 20), PAEEVQK (SEQ ID NO: 51), EAEVVQK (SEQ ID NO: 595), or PADVVQK (SEQ ID NO: 596); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof;
Attorney Docket No.14640.0092-00304 (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 76. The AAV particle of embodiment 1 or any one of embodiments 57-75, wherein [N2]-[N3] is or comprises: (i) YPAEVVQK (SEQ ID NO: 943), YQAEVVQK (SEQ ID NO: 951), YTAEVVQK (SEQ ID NO: 948), YPAEEVQK (SEQ ID NO: 950), YPAEVVQN (SEQ ID NO: 964), TEAEVVQK (SEQ ID NO: 965), or YPADVVQK (SEQ ID NO: 966); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, or 7 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 77. The AAV particle of embodiment 1 or any one of embodiments 67-76, wherein [N2]-[N3] is or comprises YPAEVVQK (SEQ ID NO: 943). 78. The AAV particle of any one of embodiments 1-77, wherein [N2]-[N3] replaces the amino acid corresponding to position 577, of the amino acid sequence of SEQ ID NO: 138. 79. The AAV particle of embodiment 1 or any one of embodiments 67-78, which further comprises one, two, three or all of an amino acid other than Q at position 574 (e.g., S), an amino acid other than S at position 575 (e.g., L, A, or T), and/or an amino acid other than S at position 576 (e.g., Y or T), numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 80. The AAV particle of embodiment 1 or any one of embodiments 67-78, which further comprises: (i) a Q at position 574, an S at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (ii) an S at position 574, an L at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iii) an S at position 574, an L at position 575, and/or a Y at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; (iv) an S at position 574, an A at position 575, and/or a T at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; or
Attorney Docket No.14640.0092-00304 (v) a Q at position 574, a T at position 575, and/or an S at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 81. The AAV particle of embodiment 1 or any one of embodiments 67-80, which further comprises [N1], wherein [N1] comprises XD, XE, and XF, wherein: (a) XD is Q or S; (b) XE is S, L, A, or T; and (c) XF is S, Y, or T;^and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c). 82. The AAV particle of embodiment 81, wherein [N1] comprises QS, SL, SA, QT, LS, LY, AT, TS, or SS. 83. The AAV particle of any one of embodiments 68, 81, and 82, wherein [N1] is or comprises QSS, SLS, SLY, SAT, or QTS. 84. The AAV particle of any one of embodiments 81-83, wherein [N1]-[N2] comprises: (i) SSYPA (SEQ ID NO: 63), LSYQA (SEQ ID NO: 597), LSYTA (SEQ ID NO: 598), LYYPA (SEQ ID NO: 600), ATYPA (SEQ ID NO: 601), LSYPA (SEQ ID NO: 603), or TSTEA (SEQ ID NO: 605); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 85. The AAV particle of any one of embodiments 81-84, wherein [N1]-[N2] comprises: (i) SSYPAE (SEQ ID NO: 79), LSYQAE (SEQ ID NO: 607), LSYTAE (SEQ ID NO: 610), LYYPAE (SEQ ID NO: 611), ATYPAE (SEQ ID NO: 613), LSYPAE (SEQ ID NO: 616), TSTEAE (SEQ ID NO: 619), or LSYPAD (SEQ ID NO: 621); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or
Attorney Docket No.14640.0092-00304 (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 86. The AAV particle of embodiment 68 or any one of embodiments 81-85, wherein [N1]-[N2] is or comprises: (i)^QSSYPAEV (SEQ ID NO: 96), SLSYQAEV (SEQ ID NO: 622), SLSYTAEV (SEQ ID NO: 623), SLYYPAEV (SEQ ID NO: 624), SATYPAEV (SEQ ID NO: 625), SLSYPAEV (SEQ ID NO: 629), SLSYPAEE (SEQ ID NO: 632), QTSTEAEV (SEQ ID NO: 633), or SLSYPADV (SEQ ID NO: 634); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, or 7 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 87. The AAV particle of embodiment 68 or any one of embodiments 81-86, wherein [N1]-[N2]-[N3] is or comprises: (i) QSSYPAEVVQK (SEQ ID NO: 150), SLSYQAEVVQK (SEQ ID NO: 635), SLSYTAEVVQK (SEQ ID NO: 637), SLYYPAEVVQK (SEQ ID NO: 639), SATYPAEVVQK (SEQ ID NO: 641), SLSYPAEVVQK (SEQ ID NO: 642), SLSYPAEEVQK (SEQ ID NO: 643), SLSYPAEVVQN (SEQ ID NO: 644), QTSTEAEVVQK (SEQ ID NO: 645), or SLSYPADVVQK (SEQ ID NO: 646); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, or 12 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 88. The AAV particle of embodiment 68 or any one of embodiments 81-87, wherein [N1]-[N2]-[N3] is or comprises QSSYPAEVVQK (SEQ ID NO: 150). 89. The AAV particle of embodiment 1 or any one of embodiments 67-88, which further comprises [N0], wherein [N0] comprises XA, XB, and XC, wherein: (a) XA is T; (b) XB is N; and
Attorney Docket No.14640.0092-00304 (c) XC is N, T, S, or K; and optionally wherein the AAV capsid comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c). 90. The AAV particle of embodiment 89, wherein [N0]: (i) comprises TN, NS, NT, NN, or NK; and/or (ii) is or comprises TNS, TNT, TNN, or TNK. 91. The AAV particle of any one of embodiments 68, 89, and 90, wherein [N0]-[N1] is or comprises: (i) TNNQSS (SEQ ID NO: 183), TNSSLS (SEQ ID NO: 647), TNSSLY (SEQ ID NO: 648), TNTSAT (SEQ ID NO: 649), TNNQTS (SEQ ID NO: 650), or TNKSAT (SEQ ID NO: 651); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 92. The AAV particle of embodiment 68 or any one of embodiments 89-91, wherein [N0]-[N1]-[N2]- [N3] is or comprises: (i) TNNQSSYPAEVVQK (SEQ ID NO: 500), TNSSLSYQAEVVQK (SEQ ID NO: 652), TNSSLSYTAEVVQK (SEQ ID NO: 654), TNSSLYYPAEVVQK (SEQ ID NO: 655), TNTSATYPAEVVQK (SEQ ID NO: 656), TNSSLSYPAEVVQK (SEQ ID NO: 657), TNSSLSYPAEEVQK (SEQ ID NO: 658), TNSSLSYPAEVVQN (SEQ ID NO: 660), TNNQTSTEAEVVQK (SEQ ID NO: 662), TNKSATYPAEVVQK (SEQ ID NO: 663), or TNSSLSYPADVVQK (SEQ ID NO: 665); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 93. The AAV particle of embodiment 68 or any one of embodiments 89-92, wherein [N0]-[N1]-[N2]- [N3] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500).
Attorney Docket No.14640.0092-00304 94. The AAV particle of embodiment 1 or any one of embodiments 67-93, which further comprises [N4], wherein [N4] comprises XG and XH, wherein: (a) XG is T, P, or N; and (b) XH is A or D; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a) or (b). 95. The AAV particle of embodiment 94, wherein [N4] is or comprises TA, TD, PA, or NA. 96. The AAV particle of embodiment 68, 94, or 95, wherein [N3]-[N4] is or comprises: (i) VQKTA (SEQ ID NO: 564), EQKTA (SEQ ID NO: 565), VKKTA (SEQ ID NO: 566), VQKPA (SEQ ID NO: 567), VHKTA (SEQ ID NO: 568), VQQTA (SEQ ID NO: 569), VQKNA (SEQ ID NO: 570), or LQKTA (SEQ ID NO: 571); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 97. The AAV particle of embodiment 68 or any one of embodiments 94-96, wherein [N0]-[N1]-[N2]- [N3]-[N4] is or comprises: (i) TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNSSLSYQAEVVQKTA (SEQ ID NO: 2064), TNSSLSYTAEVVQKTA (SEQ ID NO: 2065), TNSSLYYPAEVVQKTA (SEQ ID NO: 2066), TNTSATYPAEVVQKTA (SEQ ID NO: 2067), TNSSLSYPAEVVQKTA (SEQ ID NO: 2068), TNSSLSYPAEEVQKTA (SEQ ID NO: 2069), TNSSLSYPAEVVQKTD (SEQ ID NO: 2070), TNSSLSYPAEVVQNTA (SEQ ID NO: 2071), TNSSLSYPAEVVQKNA (SEQ ID NO: 2072), TNSSLSYPAEVVQKPA (SEQ ID NO: 2073), TNNQTSTEAEVVQKTA (SEQ ID NO: 2074), TNKSATYPAEVVQKTA (SEQ ID NO: 2075), or TNSSLSYPADVVQKTA (SEQ ID NO: 2076); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
Attorney Docket No.14640.0092-00304 98. The AAV particle of embodiment 68 or any one of embodiments 94-97, wherein [N0]-[N1]-[N2]- [N3]-[N4] is or comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). 99. An AAV particle comprising a viral genome comprising a frataxin (FXN)-encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises the formula [K1]-[K2], wherein: (i) [K1] comprises LSY or LYY; and (ii) [K2] comprises X1, X2, X3, and X4, wherein: (a) X1 is Q, T, or P; (b) X2 is A; (c) X3 is E or D; and (d) X4 is V or E. 100. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises one, two, three, four, or all of: (i) an [K0], which comprises TNNS (SEQ ID NO: 14); (ii) an [K1], which comprises LSY or LYY; (iii) an [K2], which comprises QAEV (SEQ ID NO: 15), TAEV (SEQ ID NO: 16), PAEV (SEQ ID NO: 17), PAEE (SEQ ID NO: 18), or PADV (SEQ ID NO: 19); (iv) an [K3], which comprise VQK or VQN; and (v) an [K4], which comprises TA, TD, NA, or PA. 101. The AAV particle of embodiment 99 or 100, wherein [K1] comprises LSY. 102. The AAV particle of embodiment 99 or 101, wherein [K2] comprises QA, TA, PA, EV, EE, DV, AE, or AD. 103. The AAV particle of any one of embodiments 99, 101, and 102, wherein [K2] comprises QAE, TAE, PAE, PAD, AEV, AEE, or ADV. 104. The AAV particle of any one of embodiments 99-103, wherein [K2] is or comprises QAEV (SEQ ID NO: 15), TAEV (SEQ ID NO: 16), PAEV (SEQ ID NO: 17), PAEE (SEQ ID NO: 18), or PADV (SEQ ID NO: 19).
Attorney Docket No.14640.0092-00304 105. The AAV particle of embodiment 99 or any one of embodiments 101-104, wherein [K1]-[K2] comprises LSYQA (SEQ ID NO: 597), LSYTA (SEQ ID NO: 598), LYYPA (SEQ ID NO: 600), or LSYPA (SEQ ID NO: 603). 106. The AAV particle of embodiment 99 or any one of embodiments 101-105, wherein [K1]-[K2] comprises:
(ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 107. The AAV particle of any one of embodiments 99-106, wherein [K1]-[K2] is or comprises: (i) LSYQAEV (SEQ ID NO: 667), LSYTAEV (SEQ ID NO: 668), LYYPAEV (SEQ ID NO: 669), LSYPAEV (SEQ ID NO: 671), LSYPAEE (SEQ ID NO: 673), or LSYPADV (SEQ ID NO: 674); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 108. The AAV particle of any one of embodiments 99-107, which further comprises an amino acid other than Q at position 574 (e.g., S), numbered according to the amino acid sequence of SEQ ID NO: 138. 109. The AAV particle of any one of embodiments 99-108, which further comprises S at position 574, numbered according to the amino acid sequence of SEQ ID NO: 138. 110. The AAV particle of any one of embodiments 99-109, which further comprises [K0], wherein [K0] is or comprises TNNS (SEQ ID NO: 14); an amino acid sequence comprising any portion of an amino acid sequence, e.g., any 2 or 3 amino acids, e.g., consecutive amino acids, thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of TNNS (SEQ ID NO: 14); or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of TNNS (SEQ ID NO: 14).
Attorney Docket No.14640.0092-00304 111. The AAV particle of embodiment 110, wherein [K0]-[K1] comprises: (i) TNSSLS (SEQ ID NO: 647) or TNSSLY (SEQ ID NO: 648); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 112. The AAV particle of any one of embodiments 100, 110, and 111, wherein [K0]-[K1] is or comprises: (i) TNSSLSY (SEQ ID NO: 676) or TNSSLYY (SEQ ID NO: 678); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 113. The AAV particle of any one of embodiments 110-112, wherein [K0]-[K1]-[K2] comprises: (i) TNSSLSYQA (SEQ ID NO: 679), TNSSLSYTA (SEQ ID NO: 681), TNSSLYYPA (SEQ ID NO: 682), or TNSSLSYPA (SEQ ID NO: 683); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, or 8 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 114. The AAV particle of any one of embodiments 110-113, wherein [K0]-[K1]-[K2] comprises: (i) TNSSLSYQAE (SEQ ID NO: 684), TNSSLSYTAE (SEQ ID NO: 685), TNSSLYYPAE (SEQ ID NO: 686), TNSSLSYPAE (SEQ ID NO: 687), or TNSSLSYPAD (SEQ ID NO: 689); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or
Attorney Docket No.14640.0092-00304 (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 115. The AAV particle of embodiment 100 or any one of embodiments 110-114, wherein [K0]-[K1]- [K2] is or comprises: (i) TNSSLSYQAEV (SEQ ID NO: 692), TNSSLSYTAEV (SEQ ID NO: 693), TNSSLYYPAEV (SEQ ID NO: 696), TNSSLSYPAEV (SEQ ID NO: 697), TNSSLSYPAEE (SEQ ID NO: 698), or TNSSLSYPADV (SEQ ID NO: 699); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 116. The AAV particle of any one of embodiments 99-115, which further comprises [K3], wherein [K3] comprises XA, XB, and XC, wherein: (a) XA is V; (b) XB is Q; and (c) XC is K or N; and optionally wherein the AAV capsid comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c). 117. The AAV particle of embodiment 116, wherein [K3]: (i) comprises VQ, QK, or QN; (ii) is or comprises VQK or VQN. 118. The AAV particle of embodiment 100, 116, or 117, wherein [K2]-[K3] is or comprises: (i) QAEVVQK (SEQ ID NO: 52), TAEVVQK (SEQ ID NO: 49), PAEVVQK (SEQ ID NO: 20), PAEEVQK (SEQ ID NO: 51), PAEVVQN (SEQ ID NO: 594), or PADVVQK (SEQ ID NO: 596); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i).
Attorney Docket No.14640.0092-00304 119. The AAV particle of embodiment 100 or any one of embodiments 116-118, wherein [K1]-[K2]- [K3] is or comprises: (i) LSYQAEVVQK (SEQ ID NO: 700), LSYTAEVVQK (SEQ ID NO: 701), LYYPAEVVQK (SEQ ID NO: 702), LSYPAEVVQK (SEQ ID NO: 703), LSYPAEEVQK (SEQ ID NO: 704), LSYPAEVVQN (SEQ ID NO: 706), or LSYPADVVQK (SEQ ID NO: 708); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 120. The AAV particle of embodiment 100 or any one of embodiments 116-119, wherein [K0]-[K1]- [K2]-[K3] is or comprises: (i) TNSSLSYQAEVVQK (SEQ ID NO: 652), TNSSLSYTAEVVQK (SEQ ID NO: 654), TNSSLYYPAEVVQK (SEQ ID NO: 655), TNSSLSYPAEVVQK (SEQ ID NO: 657), TNSSLSYPAEEVQK (SEQ ID NO: 658), TNSSLSYPAEVVQN (SEQ ID NO: 660), or TNSSLSYPADVVQK (SEQ ID NO: 665); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 121. The AAV particle of any one of embodiments 99-120, which further comprises [K4], wherein [K4] comprises XD and XE, wherein: (a) XD is T, P, or N; and (b) XE is A or D; and optionally wherein the AAV capsid variant comprise an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a) or (b). 122. The AAV particle of embodiment 100 or 121, wherein [K4] is or comprises TA, TD, PA, or NA. 123. The AAV particle of embodiment 100, 121, or 122, wherein [K3]-[K4] is or comprises: (i) VQKTA (SEQ ID NO: 564), VQKTD (SEQ ID NO: 714), VQNTA (SEQ ID NO: 715), VQKNA (SEQ ID NO: 570), or VQKPA (SEQ ID NO: 567);
Attorney Docket No.14640.0092-00304 (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 124. The AAV particle of any one of embodiments 100 or any one of embodiments 121-123, wherein [K0]-[K1]-[K2]-[K3]-[K4] is or comprises: (i) TNSSLSYQAEVVQKTA (SEQ ID NO: 2064), TNSSLSYTAEVVQKTA (SEQ ID NO: 2065), TNSSLYYPAEVVQKTA (SEQ ID NO: 2066), TNSSLSYPAEVVQKTA (SEQ ID NO: 2068), TNSSLSYPAEEVQKTA (SEQ ID NO: 2069), TNSSLSYPAEVVQKTD (SEQ ID NO: 2070), TNSSLSYPAEVVQNTA (SEQ ID NO: 2071), TNSSLSYPAEVVQKNA (SEQ ID NO: 2072), TNSSLSYPAEVVQKPA (SEQ ID NO: 2073), or TNSSLSYPADVVQKTA (SEQ ID NO: 2076); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 125. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises [B]-[C] (SEQ ID NO: 2114), wherein: (i) [B] comprises X1, X2, and X3, wherein: (a) X1 is Q or S; (b) X2 is S, L, or A; and (c) X3 is S, Y, or T; and (ii) [C] comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). 126. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant), wherein the AAV capsid variant comprises one, two, three, four, or all of: (i) an [A] comprising TNN, TNS, TNT, or TNK; (ii) a [B] comprising QSS, SLY, SAT, or SLS; (iii) a [C] comprising YPAEVVQK (SEQ ID NO: 943); and (iv) a [D] comprising
Attorney Docket No.14640.0092-00304 optionally wherein [C] replaces position 577 numbered according to the amino acid sequence of SEQ ID NO: 138. 127. The AAV particle of embodiment 125, comprising wherein [B] comprises QS, SL, SA, LY, AT, LS, or SS. 128. The AAV particle of any one of embodiments 125-127, comprising wherein [B] is or comprises QSS, SLY, SAT, or SLS. 129. The AAV particle of any one of embodiments 125, 127, and 128, wherein [B]-[C] comprises: (i) SSYPAEVVQK (SEQ ID NO: 572), LYYPAEVVQK (SEQ ID NO: 702), ATYPAEVVQK (SEQ ID NO: 718), or LSYPAEVVQK (SEQ ID NO: 703); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 130. The AAV particle of any one of embodiments 125-129, wherein [B]-[C] is or comprises: (i) QSSYPAEVVQK (SEQ ID NO: 150), SLYYPAEVVQK (SEQ ID NO: 639), SATYPAEVVQK (SEQ ID NO: 641), or SLSYPAEVVQK (SEQ ID NO: 642); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 131. The AAV particle of embodiment 126 or 130, wherein [B]-[C] is or comprises QSSYPAEVVQK (SEQ ID NO: 150). 132. The AAV particle of any one of embodiments 67-98 or any one of embodiments 125-131, which further comprises an amino acid other than N at position 573 (e.g., T, S, or K), numbered according to the amino acid sequence of SEQ ID NO: 138.
Attorney Docket No.14640.0092-00304 133. The AAV particle of any one of embodiments 67-98 or any one of embodiments 125-131, which further comprises: (i) a T at position 571, an N at position 572, and/or an N at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138; (ii) a T at position 571, an N at position 572, and/or a T at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138; (iii) a T at position 571, an N at position 572, and/or a S at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138; or (iv) a T at position 571, an N at position 572, and/or a K at position 573, numbered according to the amino acid sequence of SEQ ID NO: 138. 134. The AAV particle of any one of embodiments 125-133, which further comprises [A], wherein [A] comprises XA, XB, and XC, wherein: (a) XA is T; (b) XB is N; and (c) XC is N, T, S, or K; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a)-(c). 135. The AAV particle of embodiment 126 or 134, wherein [A]: (i) comprises TN, NS, NT, NK, or NN; (ii) is or comprises TNN, TNS, TNT, or TNK. 136. The AAV particle of any one of embodiments 126, 134, and 135, wherein [A]-[B] is or comprises: (i) TNNQSS (SEQ ID NO: 183), TNSSLY (SEQ ID NO: 648), TNTSAT (SEQ ID NO: 649), TNSSLS (SEQ ID NO: 647), or TNKSAT (SEQ ID NO: 651); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 137. The AAV particle of embodiment 126 or any one of embodiments 134-136, wherein [A]-[B]-[C] is or comprises:
Attorney Docket No.14640.0092-00304 (i) TNNQSSYPAEVVQK (SEQ ID NO: 500), TNSSLYYPAEVVQK (SEQ ID NO: 655), TNTSATYPAEVVQK (SEQ ID NO: 656), TNSSLSYPAEVVQK (SEQ ID NO: 657), or TNKSATYPAEVVQK (SEQ ID NO: 663); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 138. The AAV particle of embodiment 126 or any one of embodiments 134-137, wherein [A]-[B]-[C] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500). 139. The AAV particle of any one of embodiments 67-98 or any one of embodiments 125-138, which further comprises one or both of an amino acid other than T at position 578 (e.g., P or N) and/or an amino acid other than A at position 579 (e.g., D), relative to a reference sequence numbered according to the amino acid sequence of SEQ ID NO: 138. 140. The AAV particle of any one of embodiments 67-98 or any one of embodiments 125-138, which further comprises: (i) a T at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; (ii) a T at position 578 and/or a D at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; (iii) a P at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138; or (iv) an N at position 578 and/or an A at position 579, numbered according to the amino acid sequence of SEQ ID NO: 138. 141. The AAV particle of any one of embodiments 125-140, which further comprises [D], wherein [D] comprises X4 and X5, wherein: (a) X4 is T, N, or P; and (b) X5 is A or D; and optionally wherein the AAV capsid variant comprises an amino acid modification, e.g., a conservative substitution, of any of the aforesaid amino acids in (a) or (b). 142. The AAV particle of embodiment 141, wherein [D] is or comprises TA, TD, NA, or PA.
Attorney Docket No.14640.0092-00304 143. The AAV particle of embodiment 126, 141, or 142, wherein [C]-[D] is or comprises: (i) YPAEVVQKTA (SEQ ID NO: 584), YPAEVVQKTD (SEQ ID NO: 719), YPAEVVQKNA (SEQ ID NO: 724), or YPAEVVQKPA (SEQ ID NO: 586); (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 144. The AAV particle of embodiment 126 or any one of embodiments 141-143, wherein [A]-[B]-[C]- [D] is or comprises: (i) TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNSSLYYPAEVVQKTA (SEQ ID NO: 2066), TNTSATYPAEVVQKTA (SEQ ID NO: 2067), TNSSLSYPAEVVQKTA (SEQ ID NO: 2068), TNSSLSYPAEVVQKTD (SEQ ID NO: 2070), TNSSLSYPAEVVQKNA (SEQ ID NO: 2072), TNSSLSYPAEVVQKPA (SEQ ID NO: 2073), or TNKSATYPAEVVQKTA (SEQ ID NO: 2075); (ii) (ii) an amino acid sequence comprising any portion of an amino acid sequence in (i), e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids, thereof; (iii) an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences in (i); or (iv) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences in (i). 145. The AAV particle of embodiment 126 or any one of embodiments 141-144, wherein [A]-[B]-[C]- [D] is or comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). 146. The AAV particle of any one of the embodiments 1-40, 59, 60, 67-98, 139, and 140, wherein [N2]- [N3] is present in loop VIII, optionally wherein loop VIII is present at amino acids corresponding to positions 571-592 (e.g., amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)) numbered according to the amino acid sequence of SEQ ID NO: 982. 147. The AAV particle of any one of embodiments 2, 15-40, 59, 60, 68, 79-98, 139, 137, and 146 wherein [N0], [N1], and/or [N4] is present in loop VIII, optionally wherein loop VIII is present at amino acids corresponding positions 571-592 (e.g., amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID
Attorney Docket No.14640.0092-00304 NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)) numbered according to the amino acid sequence of SEQ ID NO: 982. 148. The AAV particle of any one of embodiments 2, 15-40, 59, 60, 68, 79-98, 139, 140, 146, and 147, wherein [N0]-[N1]-[N2]-[N3]-[N4] is present in loop VIII, optionally wherein loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)) numbered according to the amino acid sequence of SEQ ID NO: 982. 149. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-148, wherein [N2] is present immediately subsequent to position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 150. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-149, wherein [N2] replaces position 577 (e.g., T577), numbered according to the amino acid sequence of SEQ ID NO: 138. 151. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-150, wherein [N2] is present immediately subsequent to position 576, and wherein [N2] replaces position 577 (e.g., T577), numbered according to the amino acid sequence of SEQ ID NO: 138. 152. The AAV particle of any one of 1-40, 59, 60, 67-98, 139, 140, and 146-151, wherein [N2] corresponds to positions 577-581 (e.g., Y577, P578, A579, E580, V581) of the amino acid sequence of SEQ ID NO: 982. 153. The AAV particle of any one of embodiments 1-152, wherein the AAV capsid variant comprises an amino acid other than T at position 577, numbered according to the amino acid sequence of SEQ ID NO: 138. 154. The AAV particle of any one of embodiments 1-153, wherein the AAV capsid variant comprises a Y at position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 155. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-154, wherein X1 of [N2] is present at position 577 (e.g., T577), and positions X2-X5 of [N2] are present immediately subsequent to position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982.
Attorney Docket No.14640.0092-00304 156. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-155, wherein X1 of [N2] corresponds to position 577 (e.g., Y577), X2 of [N2] corresponds to position 578 (e.g., P588), X3 of [N2] corresponds to position 579 (e.g., A579), X4 of [N2] corresponds to position 580 (e.g., E580), and X5 of [N2] corresponds to position 581 (e.g., V581) of the amino acid sequence of SEQ ID NO: 982. 157. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-156, wherein [N2]-[N3] is present immediately subsequent to position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 158. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-157, wherein [N2]-[N3] replaces position 577 numbered according to the amino acid sequence of SEQ ID NO: 138. 159. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-158, wherein [N2]-[N3] is present immediately subsequent to position 576, and wherein [N2]-[N3] replaces position 577 (e.g., T577), numbered according to the amino acid sequence of SEQ ID NO: 138. 160. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-159, wherein [N2]-[N3] corresponds to positions 577-584 (e.g., Y577, P578, A579, E580, V581, V582, Q583, K584) of the amino acid sequence of SEQ ID NO: 982. 161. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-160, wherein [N2]-[N3]-[N4] is present immediately subsequent to position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 162. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-161, wherein [N2]-[N3]-[N4] replaces positions 577-579 (e.g., T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 163. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-162, wherein [N2]-[N3]-[N4] is present immediately subsequent to position 576, and wherein [N2]-[N3]-[N4] replaces positions 577-579 (e.g., T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 164. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-163, wherein [N2]-[N3]-[N4] corresponds to positions 577-586 (e.g., Y577, P578, A579, E580, V581, V582, Q583, K584, T585, A586) of SEQ ID NO: 982.
Attorney Docket No.14640.0092-00304 165. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-164, wherein [N1] is present immediately subsequent to position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 166. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-165, wherein [N1] replaces positions 574-576 (e.g., Q574, S575, and S576), numbered according to the amino acid sequence of SEQ ID NO: 138. 167. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-166, wherein [N1] is present immediately subsequent to position 573, and wherein [N1] replaces positions 574-576 (e.g., Q574, S575, and S576), relative to a reference sequence numbered according to the amino acid sequence of SEQ ID NO: 138. 168. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-167, wherein [N1] replaces positions 574-576 (e.g., Q574, S575, and S576), numbered according to the amino acid sequence of SEQ ID NO: 982. 169. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-168, wherein [N1] is present immediately subsequent to position 573, and wherein [N1] replaces positions 574-576 (e.g., Q574, S575, and S576), numbered according to the amino acid sequence of SEQ ID NO: 982. 170. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-169, wherein [N1] corresponds to positions 574-576 (e.g., Q574, S575, and S576) of SEQ ID NO: 982. 171. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-170, wherein [N1]-[N2]-[N3]-[N4] is present immediately subsequent to position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 172. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-171, wherein [N1]-[N2]-[N3]-[N4] replaces positions 574-579 (e.g., Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 173. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-171, wherein [N1]-[N2]-[N3]-[N4] is present immediately subsequent to position 573, and wherein [N1]-
Attorney Docket No.14640.0092-00304 [N2]-[N3]-[N4] replaces positions 574-579 (e.g., Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 174. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-173, wherein [N1]-[N2]-[N3]-[N4] corresponds to positions 574-586 (e.g., Q574, S575, S576, Y577, P578, A579, E580, V581, V582, Q583, K584, T585, A586) of SEQ ID NO: 982. 175. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-174 wherein [N0] is present immediately subsequent to position 570, numbered according to the amino acid sequence of SEQ ID NO: 138. 176. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-175, wherein [N0] replaces positions 571-573 (e.g., T571, N572, and N573), numbered according to the amino acid sequence of SEQ ID NO: 138. 177. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-176, wherein [N0] is present immediately subsequent to position 570, and wherein [N0] replaces positions 571-573 (e.g., T571, N572, and N573), numbered according to the amino acid sequence of SEQ ID NO: 138. 178. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-177 wherein [N0] is present immediately subsequent to position 570, numbered according to the amino acid sequence of SEQ ID NO: 982. 179. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-178, wherein [N0] replaces positions 571-573 (e.g., T571, N572, and N573), numbered according to the amino acid sequence of SEQ ID NO: 982. 180. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-179, wherein [N0] is present immediately subsequent to position 570, and wherein [N0] replaces positions 571-573 (e.g., T571, N572, and N573), numbered according to the amino acid sequence of SEQ ID NO: 982. 181. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-181, wherein [N0] corresponds to positions 571-573 (e.g., T571, N572, and N573) of the amino acid sequence of SEQ ID NO: 982.
Attorney Docket No.14640.0092-00304 182. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-181, wherein [N0]-[N1]-[N2]-[N3]-[N4] is present immediately subsequent to position 570, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 183. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-182, wherein [N0]-[N1]-[N2]-[N3]-[N4] replaces positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 184. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-183, wherein [N0]-[N1]-[N2]-[N3]-[N4] is present immediately subsequent to position 570, and wherein [N0]-[N1]-[N2]-[N3]-[N4] replaces positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 185. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-184, wherein [N0]-[N1]-[N2]-[N3]-[N4] corresponds to positions 571-586 (e.g., T571, N572, N573, Q574, S575, S576, Y577, P578, A579, E580, V581, V582, Q583, K584, T585, A586), of the amino acid sequence of SEQ ID NO: 982. 186. The AAV particle of any one of embodiments 3, 31-40, 68, 94-98, 139, 140, and 146-185, wherein [N4] is present immediately subsequent to position 584, numbered according to the amino acid sequence of SEQ ID NO: 982. 187. The AAV particle of any one of embodiments 3, 31-40, 68, 94-98, 139, 140, and 146-186, wherein [N4] replaces position 578 and 579, numbered according to SEQ ID NO: 138; or positions 585 and 586 numbered according to the amino acid sequence of SEQ ID NO: 982. 188. The AAV particle of any one of embodiments 3, 31-40, 68, 94-98, 139, 140, and 146-187, wherein [N4] is present immediately subsequent to position 584 and replaces positions 585 and 586 numbered according to the amino acid sequence of SEQ ID NO: 982. 189. The AAV particle of any one of embodiments 3, 31-40, 68, 94-98, 139, 140, and 146-188, wherein: (i) XA of [N0] is present at position 571, XB of [N0] is present at position 572, and XC of [N0] is present at position 573, numbered according to the amino acid sequence of SEQ ID NO: 982; (ii) XD of [N1] is present at position 574, XE of [N1] is present at position 575, and XF of [N1] is present at position 576, numbered according to the amino acid sequence of SEQ ID NO: 982;
Attorney Docket No.14640.0092-00304 (iii) X1 of [N2] is present at position 577, X2 of [N2] is present at position 578, X3 of [N2] is present at position 579, X4 of [N2] is present at position 580, and X5 of [N2] is present at position 581, numbered according to the amino acid sequence of SEQ ID NO: 982; (iv) [N3] is present at positions 582-584, numbered according to the amino acid sequence of SEQ ID NO: 982; and/or (v) XG of [N4] is present at position 585 and XH of [N4] is present at position 586, numbered according to the amino acid sequence of SEQ ID NO: 982. 190. The AAV particle of any one of embodiments 3, 31-40, 68, 94-98, 139, 140, and 146-189, wherein: (i) [N0] is present at positions 571-573, numbered according to the amino acid sequence of SEQ ID NO: 982; (ii) [N1] is present at positions 574-576, numbered according to the amino acid sequence of SEQ ID NO: 982; (iii) [N2] is present at positions 577-581, numbered according to the amino acid sequence of SEQ ID NO: 982; (iv) [N3] is present at positions 582-584, numbered according to the amino acid sequence of SEQ ID NO: 982; (v) [N4] is present at positions 585-586, numbered according to the amino acid sequence of SEQ ID NO: 982; (vi) [N2]-[N3] is present at positions 577-584, numbered according to the amino acid sequence of SEQ ID NO: 982; and/or (vii) [N0]-[N1]-[N2]-[N3]-[N4] is present at positions 571-586, numbered according to the amino acid sequence of SEQ ID NO: 982. 191. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-190, wherein [N3] is present immediately subsequent to [N2]. 192. The AAV particle of any one of embodiments 3, 31-40, 68, 94-98, 139, 140, and 146-191, wherein [N4] is present immediately subsequent to [N3]. 193. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-192, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [N2]-[N3]. 194. The AAV particle of any one of embodiments 1-40, 59, 60, 67-98, 139, 140, and 146-193, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3].
Attorney Docket No.14640.0092-00304 195. The AAV particle of any one of embodiments 3, 18, 19, 28-40, 59, 60, 68, 89-98, 139, 140, and 146-194, wherein the capsid variant comprises, from N-terminus to C-terminus, [N0]-[N1]-[N2]-[N3]. 196. The AAV particle of any one of embodiments 3, 18-40, 59, 60, 68, 79-98, 139, 140, and 146-195, wherein the capsid variant comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3]-[N4]. 197. The AAV particle of any one of embodiments 3, 18, 19, 28-40, 59, 60, 68, 89-98, 139, 140, and 146-196, wherein the capsid variant comprises, from N-terminus to C-terminus, [N0]-[N1]-[N2]-[N3]- [N4]. 198. The AAV particle of any one of embodiments 41-66 or any one of embodiments 125-145, wherein [B]-[C] is present in loop VIII, optionally wherein loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)), numbered according to the amino acid sequence of SEQ ID NO: 982. 199. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198, wherein [A] and/or [D] is present in loop VIII, optionally wherein loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)), numbered according to the amino acid sequence of SEQ ID NO: 982. 200. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, 198, and 199, wherein [A]- [B]-[C]-[D] is present in loop VIII, optionally wherein loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)), numbered according to the amino acid sequence of SEQ ID NO: 982. 201. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-200, wherein [B] is present immediately subsequent to position 573, numbered according to the amino acid sequence of SEQ ID NO: 138.
Attorney Docket No.14640.0092-00304 202. The AAV particle of any one of embodiments 41-66, 125-145, and 198-201, wherein [B] replaces positions 574-576 (e.g., Q574, S575, and S576), numbered according to the amino acid sequence of SEQ ID NO: 138. 203. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-202, wherein [B] is present immediately subsequent to position 573, and wherein [B] replaces positions 574-576 (e.g., Q574, S575, and S576), numbered according to the amino acid sequence of SEQ ID NO: 138. 204. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-203, wherein [B] is present immediately subsequent to position 573, numbered according to the amino acid sequence of SEQ ID NO: 982. 205. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-204, wherein [B] replaces positions 574-576 (e.g., Q574, S575, and S576), numbered according to the amino acid sequence of SEQ ID NO: 982. 206. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-205, wherein [B] is present immediately subsequent to position 573, and wherein [B] replaces positions 574-576 (e.g., Q574, S575, and S576), numbered according to the amino acid sequence of SEQ ID NO: 982. 207. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-206, wherein [B] corresponds to positions 574-576 (e.g., Q574, S575, and S576) of the amino acid sequence of SEQ ID NO: 982. 208. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-207, wherein [B]-[C] is present immediately subsequent to position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 209. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-208, wherein [B]-[C] replaces positions 574-577 (e.g., Q574, S575, S576, and T577), numbered according to the amino acid sequence of SEQ ID NO: 138. 210. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-209, wherein [B]-[C] is present immediately subsequent to position 573, and
Attorney Docket No.14640.0092-00304 wherein [B]-[C] replaces positions 574-577 (e.g., Q574, S575, S576, and T577), numbered according to the amino acid sequence of SEQ ID NO: 138. 211. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-210, wherein [B]-[C] corresponds to positions 574-584 (e.g., Q574, S575, S576, Y577, P578, A579, E580, V581, V582, Q583, K584) of the amino acid sequence of SEQ ID NO: 982. 212. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-211, wherein [B]-[C]-[D] is present immediately subsequent to position 573, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 213. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-212, wherein [B]-[C]-[D] replaces positions 574-579 (e.g., Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 214. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-213, wherein [B]-[C]-[D] is present immediately subsequent to position 573, and wherein [B]-[C]-[D] replaces positions 574-579 (e.g., Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 215. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-214, wherein [B]-[C]-[D] corresponds to positions 574-586 (e.g., Q574, S575, S576, Y577, P578, A579, E580, V581, V582, Q583, K584, T585, A586) of the amino acid sequence of SEQ ID NO: 982. 216. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-215, wherein [C] is present immediately subsequent to position 576, numbered according to the amino acid sequence of SEQ ID NO: 138. 217. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-216, wherein [C] replaces position 577 (e.g., T577), numbered according to the amino acid sequence of SEQ ID NO: 138. 218. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-217, wherein [C] is present immediately subsequent to position 576, and wherein [C] replaces 577 (e.g., T577), numbered according to the amino acid sequence of SEQ ID NO: 138.
Attorney Docket No.14640.0092-00304 219. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-218, wherein [C] corresponds to positions 577-584 (e.g., Y577, P578, A579, E580, V581, V582, Q583, K584) of the amino acid sequence of SEQ ID NO: 982. 220. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-219, wherein [A] is present immediately subsequent to position 570, numbered according to the amino acid sequence of SEQ ID NO: 138. 221. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-220, wherein [A] replaces positions 571-573 (e.g., T571, N572, and N573), numbered according to the amino acid sequence of SEQ ID NO: 138. 222. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-221, wherein [A] is present immediately subsequent to position 570, and wherein [A] replaces positions 571-573 (e.g., T571, N572, and N573), numbered according to the amino acid sequence of SEQ ID NO: 138. 223. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-222, wherein [A] is present immediately subsequent to position 570, numbered according to the amino acid sequence of SEQ ID NO: 982. 224. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-223, wherein [A] replaces positions 571-573 (e.g., T571, N572, and N573), numbered according to the amino acid sequence of SEQ ID NO: 982. 225. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-224, wherein [A] is present immediately subsequent to position 570, and wherein [A] replaces positions 571-573 (e.g., T571, N572, and N573), numbered according to the amino acid sequence of SEQ ID NO: 982. 226. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-225, wherein [A] corresponds to positions 571-573 (e.g., T571, N572, and N573) of the amino acid sequence of SEQ ID NO: 982. 227. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-226, wherein [D] is present immediately subsequent to position 584, numbered according to the amino acid sequence of SEQ ID NO: 982.
Attorney Docket No.14640.0092-00304 228. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-227, wherein [D] replaces positions 578 and 579 numbered according to the amino acid sequence of SEQ ID NO: 138; or positions 585 and 586, numbered according to the amino acid sequence of SEQ ID NO: 982. 229. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-228, wherein [D] is present immediately subsequent to position 584 and replaces positions 585 and 586, numbered according to the amino acid sequence of SEQ ID NO: 982. 230. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-229, wherein [A]- [B]-[C]-[D] is present immediately subsequent to position 570, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 231. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-230, wherein [A]- [B]-[C]-[D] replaces positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 232. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-231, wherein [A]- [B]-[C]-[D] is present immediately subsequent to position 570, and wherein [A]-[B]-[C]-[D] replaces positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 233. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-232, wherein [A]- [B]-[C]-[D] corresponds to positions 571-586 (e.g., T571, N572, N573, Q574, S575, S576, Y577, P578, A579, E580, V581, V582, Q583, K584, T585, A586) of the amino acid sequence of SEQ ID NO: 982. 234. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-233, wherein: (i) XA of [A] is present at position 571, XB of [A] is present at position 572, and XC of [A] is present at position 573, numbered according to the amino acid sequence of SEQ ID NO: 982; (ii) X1 of [B] is present at position 574, X2 of [B] is present at position 575, and X3 of [B] is present at position 576, numbered according to the amino acid sequence of SEQ ID NO: 982; (iii) [C] is present at positions 577-584, numbered according to the amino acid sequence of SEQ ID NO: 982; and/or (iv) X4 of [D] is present at position 585 and position X5 of [D] is present at position 586, numbered according to the amino acid sequence of SEQ ID NO: 982. 235. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-234, wherein:
Attorney Docket No.14640.0092-00304 (i) [A] is present at positions 571-573, numbered according to the amino acid sequence of SEQ ID NO: 982; (ii) [B] is present at positions 574-576, numbered according to the amino acid sequence of SEQ ID NO: 982; (iii) [C] is present at positions 577-584, numbered according to the amino acid sequence of SEQ ID NO: 982; (iv) [D] is present at positions 585-586, numbered according to the amino acid sequence of SEQ ID NO: 982; and/or (v) [A]-[B]-[C]-[D] is present at positions 571-586, numbered according to the amino acid sequence of SEQ ID NO: 982. 236. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-235, wherein [C] is present immediately subsequent to [B]. 237. The AAV particle of any one of embodiments 42, 61-66, 126, 141-145, and 198-236 wherein [D] is present immediately subsequent to [C]. 238. The AAV particle of any one of embodiments 41-66, any one of embodiments 125-145, or any one of embodiments 198-237, wherein the capsid variant comprises, from N-terminus to C-terminus, [B]-[C]. 239. The AAV particle of any one of embodiments 42, 52-66, 126, 134-145, and 198-238, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [A]-[B]-[C]. 240. The AAV particle of any one of embodiments 42, 61-66, 126, 141-145, and 198-239, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [B]-[C]-[D]. 241. The AAV particle of any one of embodiments 42, 61-66, 126, 141-145, and 198-240, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [A]-[B]-[C]-[D]. 242. The AAV particle of any one of the embodiments 99-124, wherein [K1]-[K2] is present in loop VIII. 243. The AAV particle of any one of embodiments 92-124 or embodiment 242, wherein [K0], [K3], and/or [K4] is present in loop VIII, optionally wherein loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids
Attorney Docket No.14640.0092-00304 TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)), numbered according to the amino acid sequence of SEQ ID NO: 982. 244. The AAV particle of any one of embodiments 92-124 embodiment 242, or embodiment 243, wherein [K0]-[K1]-[K2]-[K3]-[K4] is present in loop VIII, optionally wherein loop VIII comprises positions 571-592 (e.g., the positions of amino acids TNNQSSTTAPATGTYNLQEIVP (SEQ ID NO: 736)) numbered according to the amino acid sequence of SEQ ID NO: 138, or positions 571-599 (e.g., the positions of amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)) numbered according to the amino acid sequence of SEQ ID NO: 982. 245. The AAV particle of any one of embodiments 99-124 or any one of embodiments 242-244, wherein [K1] is present immediately subsequent to position 574, numbered according to the amino acid sequence of SEQ ID NO: 138. 246. The AAV particle of any one of embodiments 99-124 or any one of embodiments 242-245, wherein [K1] replaces positions 575-577 (e.g., S575, S576, and T577), numbered according to the amino acid sequence of SEQ ID NO: 138. 247. The AAV particle of any one of embodiments 99-124 or any one of embodiments 242-246, wherein [K1] is present immediately subsequent to position 574, and wherein [K1] replaces positions 575-577 (e.g., S575, S576, and T577), numbered according to the amino acid sequence of SEQ ID NO: 138. 248. The AAV particle of any one of embodiments 100, 116-124, and 242-247, wherein [K1]-[K2]-[K3] is present immediately subsequent to position 574, numbered according to the amino acid sequence of SEQ ID NO: 138. 249. The AAV particle of any one of embodiments 100, 116-124, and 242-248, wherein [K1]-[K2]-[K3] replaces positions 575-577 (e.g., S575, S576, and T577), numbered according to the amino acid sequence of SEQ ID NO: 138. 250. The AAV particle of any one of embodiments 100, 116-124, and 242-249, wherein [K1]-[K2]-[K3] is present immediately subsequent to position 574, and wherein [K1]-[K2]-[K3] replaces positions 575- 577 (e.g., S575, S576, and T577), numbered according to the amino acid sequence of SEQ ID NO: 138. 251. The AAV particle of any one of embodiments 100, 121-124, and 242-250, wherein [K1]-[K2]-[K3]- [K4] is present immediately subsequent to position 574, numbered according to the amino acid sequence of SEQ ID NO: 138.
Attorney Docket No.14640.0092-00304 252. The AAV particle of any one of embodiments 100, 121-124, and 242-251, wherein [K1]-[K2]-[K3]- [K4] replaces positions 575-579 (e.g., S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 253. The AAV particle of any one of embodiments 100, 121-124, and 242-252, wherein [K1]-[K2]-[K3]- [K4] is present immediately subsequent to position 574, and wherein [K1]-[K2]-[K3]-[K4] replaces positions 575-579 (e.g., S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 254. The AAV particle of any one of embodiments 100, 110-124, and 242-253, wherein [K0] is present immediately subsequent to position 570, numbered according to the amino acid sequence of SEQ ID NO: 138. 255. The AAV particle of any one of embodiments 100, 110-124, and 242-254, wherein [K0] replaces positions 571 to 574 (e.g., T571, N572, N573, and Q574), numbered according to the amino acid sequence of SEQ ID NO: 138. 256. The AAV particle of any one of embodiments 100, 110-124, and 242-255, wherein [K0] is present immediately subsequent to position 570, and wherein [K0] replaces positions 571 to 574 (e.g., T571, N572, N573, and Q574), numbered according to the amino acid sequence of SEQ ID NO: 138. 257. The AAV particle of any one of embodiments 100, 121-124, and 242-256, wherein [K0]-[K1]-[K2]- [K3]-[K4] is present immediately subsequent to position 570, numbered according to the amino acid sequence of SEQ ID NO: 138. 258. The AAV particle of any one of embodiments 100, 121-124, and 242-257, wherein [K0]-[K1]-[K2]- [K3]-[K4] replaces positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 259. The AAV particle of any one of embodiments 100, 121-124, and 242-258, wherein [K0]-[K1]-[K2]- [K3]-[K4] is present immediately subsequent to position 570, and wherein [K0]-[K1]-[K2]-[K3]-[K4] replaces positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 260. The AAV particle of any one of embodiments 100, 116-124, and 242-259, wherein [K3] is present immediately subsequent [K2].
Attorney Docket No.14640.0092-00304 261. The AAV particle of any one of embodiments 100, 121-124, and 242-260, wherein [K4] is present immediately subsequent [K3]. 262. The AAV particle of any one of embodiments 100, 121-124, and 242-261, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [K1]-[K2]. 263. The AAV particle of any one of embodiments 100, 116-124, and 242-262, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [K1]-[K2]-[K3]. 264. The AAV particle of any one of embodiments 100, 116-124, and 242-263, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [K0]-[K1]-[K2]-[K3]. 265. The AAV particle of any one of embodiments 100, 121-124, and 242-264, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [K1]-[K2]-[K3]-[K4]. 266. The AAV particle of any one of embodiments 100, 121-124, and 242-265, wherein the AAV capsid variant comprises, from N-terminus to C-terminus, [K0]-[K1]-[K2]-[K3]-[K4]. 267. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant) comprising: (a) any one of the amino acid sequences provided in Table 1, 2A, 2B, 14, or 23-28; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of the amino acid sequences provided in Tables 1, 2A, 2B, 14, or 23-28; (c) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the amino acid sequences provided in Table 1, 2A, 2B, 14, or 23-28; or (d) an amino sequence comprising one, two, or three but no more than four modifications relative to any one of the amino acid sequences provided in Table 1, 2A, 2B, 14, or 23-28. 268. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant) comprising: (a) the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583-1587, 1590, 1591-1593, 1598-1608, and 1610-1624;
Attorney Docket No.14640.0092-00304 (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583-1587, 1590, 1591-1593, 1598-1608, and 1610-1624; (c) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583-1587, 1590, 1591-1593, 1598-1608, and 1610-1624; or (d) an amino sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583-1587, 1590, 1591- 1593, 1598-1608, and 1610-1624. 269. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant) comprising: (a) the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080; (c) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080; or (d) an amino sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080. 270. An adeno-associated virus (AAV) particle comprising a viral genome comprising a frataxin (FXN)- encoding sequence (e.g., encoding a human FXN protein) and an AAV capsid variant (e.g., an AAV5 capsid variant) comprising: (a) the amino acid sequence of any one of SEQ ID NOs: 1021, 1024, 1232, 1300, 1327, 1533, 1538, 1585, 1590, and 1591; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of SEQ ID NOs: 1021, 1024, 1232, 1300, 1327, 1533, 1538, 1585, 1590, and 1591; (c) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 1021, 1024, 1232, 1300, 1327, 1533, 1538, 1585, 1590, and 1591; or
Attorney Docket No.14640.0092-00304 (d) an amino sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 1021, 1024, 1232, 1300, 1327, 1533, 1538, 1585, 1590, and 1591. 271. The AAV particle of any one of embodiments 267-270, wherein the AAV capsid variant comprises at least 3, at least 4, at least 5, at least 6, or at least 7 consecutive amino acids from SEQ ID NO: 943 or of any one of SEQ ID NOs: 946-966. 272. The AAV particle of embodiment 271, wherein the at least 3 consecutive amino acids comprise YPA. 273. The AAV particle of embodiment 271, wherein the at least 4 consecutive amino acids comprise YPAE (SEQ ID NO: 21). 274. The AAV particle of embodiment 271, wherein the at least 5 consecutive amino acids comprise YPAEV (SEQ ID NO: 1). 275. The AAV particle of embodiment 271, wherein the at least 6 consecutive amino acids comprise YPAEVV (SEQ ID NO: 725). 276. The AAV particle of embodiment 271, wherein the at least 7 consecutive amino acids comprise YPAEVVQ (SEQ ID NO: 726). 277. The AAV particle of embodiment 271, wherein the amino acid sequence comprises YPAEVVQK (SEQ ID NO: 943). 278. The AAV particle of any one of embodiments 267, 268, and 270, wherein the AAV capsid variant comprises: (i) YTP; (ii) YTPS (SEQ ID NO: 26); (iii) YTPSL (SEQ ID NO: 7); (iv) YTPSLV (SEQ ID NO: 727); (v) YTPSLVQ (SEQ ID NO: 728); and/or (vi) YTPSLVQK (SEQ ID NO: 952). 279. The AAV capsid variant of any one of embodiments 267, 268, and 270, wherein: (i) the at least 3 consecutive amino acids comprise YPP;
Attorney Docket No.14640.0092-00304 (ii) the at least 4 consecutive amino acids comprise YPPS (SEQ ID NO: 22); (iii) the at least 5 consecutive amino acids comprise YPPSL (SEQ ID NO: 2); (iv) the at least 6 consecutive amino acids comprise YPPSLV (SEQ ID NO: 729); (v) the at least 7 consecutive amino acids comprise YPPSLVQ (SEQ ID NO: 732); and/or (vi) the amino acid sequence comprises YPPSLVQK (SEQ ID NO: 946). 280. The AAV capsid variant of any one of embodiments 267, 268, and 270, wherein: (i) the at least 3 consecutive amino acids comprise YPP; (ii) the at least 4 consecutive amino acids comprise YPPS (SEQ ID NO: 22); (iii) the at least 5 consecutive amino acids comprise YPPSL (SEQ ID NO: 2); (iv) the at least 6 consecutive amino acids comprise YPPSLE (SEQ ID NO: 733); (v) the at least 7 consecutive amino acids comprise YPPSLEQ (SEQ ID NO: 734); and/or (vi) the amino acid sequence comprises YPPSLEQK (SEQ ID NO: 953). 281. The AAV capsid variant of any one of embodiments 267, 268, and 270, wherein: (i) the at least 3 consecutive amino acids comprise YPP; (ii) the at least 4 consecutive amino acids comprise YPPS (SEQ ID NO: 22); (iii) the at least 5 consecutive amino acids comprise YPPSL (SEQ ID NO: 2); (iv) the at least 6 consecutive amino acids comprise YPPSLV (SEQ ID NO: 729); (v) the at least 7 consecutive amino acids comprise YPPSLVK (SEQ ID NO: 735); and/or (vi) the amino acid sequence comprises YPPSLVKK (SEQ ID NO: 954). 282. The AAV particle of any one of embodiments 267-277, wherein the AAV capsid variant comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). 283. The AAV particle of any one of embodiments 267, 268, 270, and 278-281, wherein the AAV capsid variant comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 1021, 1024, 1232, 1300, 1327, 1533, 1538, 1585, 1590, and 1591. 284. The AAV particle of any one of embodiments 267, 268, 270, and 278-281, wherein the AAV capsid variant comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 946, 952, 953, and 954.
Attorney Docket No.14640.0092-00304 285. The AAV particle of any one of embodiments 267-277 or embodiment 282, wherein the AAV capsid variant comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). 286. The AAV particle of any one of embodiments 267, 268, 270, 278-281, and 283, wherein the AAV capsid variant comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 1021, 1024, 1232, 1300, 1327, 1533, 1538, 1585, 1590, and 1591. 287. The AAV particle of any one of embodiments 267, 268, 270, 278-281, and 284, wherein the AAV capsid variant comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 946, 952, 953, and 954. 288. The AAV particle of any prior embodiment, e.g., any one of embodiments 267-277, 282, and 285, wherein the AAV capsid variant comprises an amino acid sequence encoded by: (i) a nucleotide sequence comprising SEQ ID NO: 944 or a nucleotide sequence that comprises at least one, at least two, at least three, at least four, at least five, at least six, at least seven, at least eight, at least nine, or ten modifications, but no more than ten modifications, , relative to the nucleotide sequence of SEQ ID NO: 944; or (ii) a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, at least seven, at least eight, at least nine, or ten different nucleotides, but no more than ten different nucleotides, relative to the nucleotide sequence of SEQ ID NO: 944. 289. The AAV particle of any one of embodiments 267-277, 282, 285, and 289, wherein the AAV capsid variant is encoded by: (i) a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 944; or (ii) a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 944. 290. The AAV particle of embodiment 267, wherein the AAV capsid variant comprises: (a) the amino acid sequence of any one of SEQ ID NOs: 2024-2063; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of SEQ ID NOs: 2024-2063;
Attorney Docket No.14640.0092-00304 (c) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063; or (d) an amino sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063. 291. The AAV particle of embodiment 267 or 290, wherein the different amino acids, of the amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063, are present at one or more of the following positions: (i) position 1, wherein the different amino acid is T or L; (ii) position 2, wherein the different amino acid is N, L, K, A, T, or P; (iii) position 3, wherein the different amino acid is N, K, L, A, Y, or S; (iv) position 4, wherein the different amino acid is Q, L, T, S, F, Y, K, or A; (v) position 5, wherein the different amino acid is S, H, A, M, Q, T, V, or F; (vi) position 6, wherein the different amino acid is S, P, V, A, Q, L, T, N, or M; (vii) position 7, wherein the different amino acid is Y, H, S, V, A, L, or T; (viii) position 8, wherein the different amino acid is D, P, A, Q, F, L, S, H, or M; (ix) position 9, wherein the different amino acid is F, A, L, D, or Q; (x) position 10, wherein the different amino acid is T, E, I, or S; (xi) position 11, wherein the different amino acid is V, A, N, or S; (xii) position 12, wherein the different amino acid is V, L, or P; (xiii) position 13, wherein the different amino acid is Q, E, or P; (xiv) position 14, wherein the different amino acid is K, N, S, or L; (xv) position 15, wherein the different amino acid is T, V, M, or L; and/or (xvi) position 16, wherein the different amino acid is A, G, or R. 292. The AAV particle of embodiment 267, wherein the AAV capsid variant comprises: (a) the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842- 2023, and 2087-2112; (b) an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798- 1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842-2023, and 2087-2112; (c) an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739,
Attorney Docket No.14640.0092-00304 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842-2023, and 2087-2112; or (d) an amino sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842-2023, and 2087-2112. 293. The AAV particle of embodiment 268 or 292, wherein the different amino acids of the amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842-2023, and 2087-2112, are present at one or more of the following positions: (i) position 1, wherein the different amino acid is T, G, N, S, E, L, Y, V, or I; (ii) position 2, wherein the different amino acid is D, N, K, E, V, G, R, L, H, F, P, T, A, S, I, or Y; (iii) position 3, wherein the different amino acid is Y, N, K, T, W, Q, M, V, C, A, L, F, H, G, R, S, or P; (iv) position 4, wherein the different amino acid is H, Q, P, E, R, K, A, S, V, L, T, D, I, G, M, or N; (v) position 5, wherein the different amino acid is R, S, K, N, H, G, W, A, P, V, Q, Y, L, or F; (vi) position 6, wherein the different amino acid is G, S, F, R, W, H, I, C, M, A, Y, K, N, Q, V, P, E, D, T, or L; (vii) position 7, wherein the different amino acid is D, Y, S, I, H, F, P, K, R, G, L, Q, A, M, T, N, V, W, C, or E; (viii) position 8, wherein the different amino acid is P, L, Q, T, W, V, G, K, I, Y, N, H, R, D, S, M, A, F, or E; (ix) position 9, wherein the different amino acid is A, R, T, Q, S, M, L, E, K, V, G, D, N, H, F, P, or I; (x) position 10, wherein the different amino acid is K, E, Q, H, V, G, R, S, P, I, N, M, A, L, D, or T; (xi) position 11, wherein the different amino acid is V, A, E, N, R, L, M, T, Q, S, K, C, G, D, Y, P, H, F, or I; (xii) position 12, wherein the different amino acid is V, P, L, S, T, N, A, G, K, R, I, H, E, Q, or M; (xiii) position 13, wherein the different amino acid is Q, K, N, A, H, R, T, V, E, I, P, G, S, or L; (xiv) position 14, wherein the different amino acid is K, E, I, Y, Q, R, G, D, L, N, or S; (xv) position 15, wherein the different amino acid is S, T, N, Q, I, P, E, G, K, M, or H; and/or
Attorney Docket No.14640.0092-00304 (xvi) position 16, wherein the different amino acid is A, D, L, Y, Q, or T. 294. The AAV particle of any one of embodiments 267-293, wherein the amino acid sequence is present in loop VIII of the AAV capsid variant. 295. The AAV particle of any one of embodiments 267-294, wherein the amino acid sequence is present immediately subsequent to position 570, 571, 572, 573, 574, 575, or 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 296. The AAV particle of any one of embodiments 267-295, wherein the amino acid sequence replaces one, two, three, four, five or all of positions 571, 572, 573, 574, 575, and/or 576 (e.g., positions T571, N572, N573, Q574, S575, S576, T577, T578, and/or A579), numbered according to the amino acid sequence of SEQ ID NO: 138. 297. The AAV particle of any one of embodiments 267-296, wherein the amino acid sequence is present immediately subsequent to position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 298. The AAV particle of any one of embodiments 267-297, wherein the AAV capsid variant comprises an amino acid residue other than T at position 577, numbered according to the amino acid sequence of SEQ ID NO: 138. 299. The AAV particle of any one of embodiments 267-298, wherein the AAV capsid variant comprises the amino acid Y at position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 300. The AAV particle of any one of embodiments 267-299, wherein the AAV capsid variant comprises the substitution T577Y, numbered according to the amino acid sequence of SEQ ID NO: 138. 301. The AAV particle of embodiment 298 or 299, wherein the amino acid sequence is or comprises YPAEVVQK (SEQ ID NO: 943), and starts at position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 302. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-301, wherein the AAV capsid variant comprises an amino acid other than T at position 577, and further comprises the amino acid sequence of PAEVVQK (SEQ ID NO: 20), which is present immediately subsequent to position 577, numbered according to the amino acid sequence of SEQ ID NO: 138.
Attorney Docket No.14640.0092-00304 303. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-302, wherein the AAV capsid variant comprises the amino acid Y at position 577, and further comprises the amino acid sequence of PAEVVQK (SEQ ID NO: 20), which is present immediately subsequent to position 577, numbered according to the amino acid sequence of SEQ ID NO: 138. 304. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-303, wherein the AAV capsid variant comprises the amino acid Y at position 577, and further comprises the amino acid sequence of PAEVVQK (SEQ ID NO: 20), which is present immediately subsequent to position 577 (e.g., at positions 578-584), numbered according to the amino acid sequence of SEQ ID NO: 982. 305. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-304, wherein the AAV capsid variant comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the amino acid sequence replaces position 577 (e.g., T577), numbered according to the amino acid sequence of SEQ ID NO: 138. 306. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-305, wherein the AAV capsid variant comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the amino acid sequence: (i) is present immediately subsequent to position 576; and (ii) replaces position 577 (e.g., T577), wherein in (i) and (ii) are numbered according to the amino acid sequence of SEQ ID NO: 138. 307. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-306, wherein the AAV capsid variant further comprises an amino acid other than T at position 571 (e.g., I), numbered according to the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 982. 308. The AAV particle of any one of embodiments 267-277, 272, 275, 278, 289, and 294-307, wherein the AAV capsid variant further comprises I at position 571, numbered according to the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 982. 309. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-308, wherein the AAV capsid variant further comprises one, two, or all of an amino acid other than Q at position 574 (e.g., A or T), S at position 575 (e.g., G), and/or S (e.g., A, L, K, or R) at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or SEQ ID NO: 982.
Attorney Docket No.14640.0092-00304 310. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-309, wherein the AAV capsid variant further comprises: (i) A at position 574, G at position 575, and A at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; or (ii) T at position 574 and L at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 311. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-309, wherein the AAV capsid variant further comprises: (i) K at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; or (ii) R at position 576, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 312. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-310, wherein the capsid variant comprises: (i) A at position 574, G at position 575, A at position 576, and Y at position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; and (ii) the amino acid sequence of PAEVVQK (SEQ ID NO: 20), which is present immediately subsequent to position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 313. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-310, wherein the AAV capsid variant comprises: (i) T at position 574, L at position 576, and Y at position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; and (ii) the amino acid sequence of PAEVVQK (SEQ ID NO: 20), which is present immediately subsequent to position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 314. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, 294-309, and 311, wherein the AAV capsid variant comprises: (i) K at position 576 and Y at position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; and (ii) the amino acid sequence of PAEVVQK (SEQ ID NO: 20), which is present immediately subsequent to position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 315. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, 295-309, and 311, wherein the AAV capsid variant comprises:
Attorney Docket No.14640.0092-00304 (i) R at position 576 and Y at position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; and (ii) the amino acid sequence of PAEVVQK (SEQ ID NO: 20), which is present immediately subsequent to position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 316. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, and 294-308, wherein the AAV capsid variant comprises: (i) I at position 571 and Y at position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982; and (ii) the amino acid sequence of PAEVVQK (SEQ ID NO: 20), which is present immediately subsequent to position 577, numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 317. The AAV particle of any one of embodiments 267, 268, 270, 278, 283, 284, 286, and 287, wherein the AAV capsid variant comprises Y at position 577 and the amino acid sequence TPSLVQK (SEQ ID NO: 53), which is present immediately subsequent to position 577, all numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 318. The AAV particle of any one of embodiments 267, 268, 270, 279, 283, 284, 286, and 287, wherein the AAV capsid variant comprises Y at position 577 and the amino acid sequence PPSLVQK (SEQ ID NO: 47), which is present immediately subsequent to position 577, all numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 319. The AAV particle of any one of embodiments 267, 268, 270, 280, 283, 284, 286, and 287, wherein the AAV capsid variant comprises Y at position 577 and the amino acid sequence PPSLEQK (SEQ ID NO: 54), which is present immediately subsequent to position 577, all numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 320. The AAV particle of any one of embodiments 267, 268, 270, 281, 283, 284, 286, and 287, wherein the AAV capsid variant comprises Y at position 577 and the amino acid sequence PPSLVKK (SEQ ID NO: 55), which is present immediately subsequent to position 577, all numbered according to the amino acid sequence of SEQ ID NO: 138 or 982. 321. The AAV particle of any one of the embodiments provided herein, wherein the AAV capsid variant further comprises a modification in loop I, II, IV, and/or VI. 322. The AAV particle of any one of the embodiments provided herein, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications,
Attorney Docket No.14640.0092-00304 but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 138. 323. The AAV particle of any one of the embodiments provided herein, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but no more than 30, no more than 20, or no more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 138. 324. The AAV particle of any one of the embodiments provided herein, wherein the AAV capsid variant comprises an amino acid sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 138. [Embodiments 325-327 are intentionally absent.] 328. The AAV particle of any one of the embodiments provided herein, wherein the AAV capsid variant comprises a VP1 protein, a VP2 protein, a VP3 protein, or a combination thereof. 329. The AAV particle of any one of embodiments 1-98, 125-242, 267-277, 282, 285, 288, 289, 294-306, and 321-328, wherein the AAV capsid variant comprises the amino acid sequence corresponding to positions 137-731, e.g., a VP2, of SEQ ID NO: 982, or a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. 330. The AAV particle of any one of embodiments 1-98, 125-242, 267-277, 282, 285, 288, 289, 294-306, and 321-329, wherein the AAV capsid variant comprises the amino acid sequence corresponding to positions 193-731, e.g., a VP3, of SEQ ID NO: 982, or a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. 331. The AAV particle of any one of embodiments 1-321, wherein the AAV capsid variant comprises a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to the amino acid sequence corresponding to positions 137-724, e.g., a VP2, of SEQ ID NO: 138. 332. The AAV particle of any one of embodiments 1-321, wherein the AAV capsid variant comprises a sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at
Attorney Docket No.14640.0092-00304 least 97%, at least 98%, or at least 99%) sequence identity to the amino acid sequence corresponding to positions 193-724, e.g., a VP3, of SEQ ID NO: 138. 333. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, 294-306, and 321-328, wherein the AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, at least 5, or at least 6 consecutive amino acids from the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein: (i) the at least 3 consecutive amino acids comprise YPA; (ii) the at least 4 consecutive amino acids comprise YPAE (SEQ ID NO: 21); (iii) the at least 5 consecutive amino acids comprise YPAEV (SEQ ID NO: 1); (iv) the at least 6 consecutive amino acids comprise YPAEVV (SEQ ID NO: 725); or (v) the at least 7 consecutive amino acids comprise YPAEVVQ (SEQ ID NO: 726); wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 739, or an amino acid sequence at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical to SEQ ID NO: 739. 334. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, 294-306, 321-328, and 333, wherein the AAV capsid variant comprises an amino acid sequence comprising at least 3, at least 4, at least 5, or at least 6 consecutive amino acids from the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein: (i) the at least 3 consecutive amino acids comprise YPA; (ii) the at least 4 consecutive amino acids comprise YPAE (SEQ ID NO: 21); (iii) the at least 5 consecutive amino acids comprise YPAEV (SEQ ID NO: 1); (iv) the at least 6 consecutive amino acids comprise YPAEVV (SEQ ID NO: 725); or (v) the at least 7 consecutive amino acids comprise YPAEVVQ (SEQ ID NO: 726); wherein the AAV capsid variant comprises: (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982; (b) a VP2 protein comprising the amino acid sequence of positions 137- 731 of SEQ ID NO: 982; (c) a VP3 protein comprising the amino acid sequence of positions 193-731 of SEQ ID NO: 982; or (d) an amino acid sequence with at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to any one of the amino acid sequences in (a)-(c). 335. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, 294-306, 321-328, and 334, wherein the AAV capsid variant comprises one, two, or three but no more than four different amino acids, relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the AAV capsid variant comprises: (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982;
Attorney Docket No.14640.0092-00304 (b) a VP2 protein comprising the amino acid sequence of positions 137-731 of SEQ ID NO: 982; (c) a VP3 protein comprising the amino acid sequence of positions 193-731 of SEQ ID NO: 982; or (d) an amino acid sequence with at least 90% (e.g., at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to any one of the amino acid sequences in (a)- (c). 336. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, 294-306, 321-328, and 333-335, comprising one, two, or three but no more than four different amino acids, relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 739; or an amino acid sequence at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% sequence identity to the amino acid sequence of SEQ ID NO: 739. 337. The AAV particle of any one of embodiments 267-277, 282, 285, 288, 289, 294-306, 321-328, and 333-336, comprising one or two, but no more than three substitutions relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the AAV capsid variant comprises an amino acid sequence at least 90% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to the amino acid sequence of SEQ ID NO: 982. 338. The AAV particle of any one of embodiments 1-98, 125-241, 267-277, 282, 285, 288, 289, 294-306, 321-328, and 333-337, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982, or an amino acid sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. 339. The AAV particle of any one of embodiments 1-98, 125-241, 267-277, 282, 285, 288, 289, 294-306, 321-328, and 333-338, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 95% identity thereto. 340. The AAV particle of any one of embodiments 1-98, 125-241, 267-277, 282, 285, 288, 289, 294-306, 321-328, and 333-339, wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 98% identity thereto. 341. The AAV particle of any one of embodiments 1-98, 125-241, 267-277, 282, 285, 288, 289, 294-306, 321-328, or 333-340, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 982.
Attorney Docket No.14640.0092-00304 342. The AAV particle of any one of embodiments 1-98, 125-241, 267-277, 282, 285, 288, 289, 294-306, 321-328, and 333-341, wherein the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three, but not more than 30, not more than 20, or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 982. 343. The AAV particle of any one of embodiments 1-98, 125-241, 267-277, 282, 285, 288, 289, 294-306, 321-328, and 333-342, wherein the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 984, or a nucleotide sequence with at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. 344. The AAV particle of any one of the embodiments provided herein, wherein the nucleotide sequence encoding the AAV capsid variant is codon-optimized. [Embodiments 345-349 are intentionally absent.] 350. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 344, which has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. 351. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 344, which has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982. 352. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 344, which has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139. 353. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 344, which transduces a brain region, e.g., a temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum, optionally wherein the level of transduction is at least 1.5, at least 2.2, at least 2.4, at least 2.5, at least 2.6, at least 2.7, at least 3.0, at least 3.2, at least 3.5, at least 3.7, at
Attorney Docket No.14640.0092-00304 least 4.0, at least 4.2, at least 4.5, at least 4.7, at least 4.9, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, or at least 35-fold greater as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139, e.g., when measured by an assay, e.g., an immunohistochemistry assay or a qPCR assay, e.g., as described in Example 2. 354. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 353, which is enriched at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 12, at least 15, at least 17, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, or at least 65-fold in the brain compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 1. 355. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 354, which is enriched at least 10, at least 12, at least 15, at least 17, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 61, at least 62, at least 63, at least 64, or at least 65-fold in the brain compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138, e.g., when measured by an assay as described in Example 1. 356. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 355, which is enriched in the brain of at least two to at least three species, e.g., a non-human primate and rodent (e.g., rat and/or mouse), e.g., as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. 357. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 356, which is enriched at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 12, at least 15, at least 17, at least 20, at least 25, at least 30, at least 35, at least 40, or at least 45-fold in the brain of at least two to at least three species, e.g., a non-human primate and rodent (e.g., rat and/or mouse), compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138, e.g., when measured by an assay as described in Examples 1, 2, 4, and 5. 358. The AAV particle of embodiment 356 or 357, wherein the at least two to at least three species are Macaca fascicularis, Chlorocebus sabaeus, Callithrix jacchus, rat, and/or mouse (e.g., BALB/c mice). 359. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 358, which is enriched at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at
Attorney Docket No.14640.0092-00304 least 40, at least 45, at least 50, at least 55, at least 60, at least 65, at least 70, at least 75, at least 80, at least 85, at least 90, at least 100, at least 125, at least 150, at least 175, at least 200, or at least 225-fold, in the brain compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982, e.g., when measured by an assay as described in Example 4. 360. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 359, which delivers an increased level of a payload to a brain region, optionally wherein the level of the payload is increased by at least 20, at least 25, at least 30, or at least 35-fold, as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139, e.g., when measured by an assay, e.g., a qRT-PCR or a qPCR assay (e.g., as described in Example 2). 361. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 360, which delivers an increased number and/or level of viral genomes to a brain region, optionally wherein the number and/or level of viral genomes is increased by at least 1.5, at least 2.2, at least 2.4, at least 2.5, at least 2.6, at least 2.7, at least 3.0, at least 3.2, at least 3.5, at least 3.7, at least 4.0, at least 4.2, at least 4.5, at least 4.7, at least 4.9, or at least 5-fold as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139, e.g., when measured by an assay, e.g., a qRT-PCR or a qPCR assay (e.g., as described in Example 2). 362. The AAV particle of any one of embodiments 350-361, wherein the brain region is a temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, cerebellum, or a combination thereof. 363. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 362, which is enriched at least 3, at least 3.5, at least 4, at least 4.5, at least 5, at least 5.5, at least 6, or at least 6.5-fold in a spinal cord region compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139, e.g., when measured by an assay as described in Example 2. 364. The AAV particle of any one of embodiments 363, wherein the spinal cord region is a cervical region, a lumbar region, a thoracic region, or a combination thereof. 365. The AAV particle of any one of the embodiments provided herein, which shows preferential transduction in a brain region relative to transduction in the dorsal root ganglia (DRG). 366. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 365, which is capable of transducing neuronal cells.
Attorney Docket No.14640.0092-00304 367. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 366, which is capable of transducing non-neuronal cells, e.g., glial cells (e.g., oligodendrocytes). 368. The AAV particle of any one of embodiments 1-66, 139, 140, 146-241, 267, 268, 270-289, and 294- 367, which shows preferential transduction in a brain region relative to transduction in the liver. 369. The AAV particle of any one of embodiments 67-267, 269, 271-277, 282, 285, 288, 289, 294-306, and 321-349, which has an increased tropism for a heart cell or heart tissue, e.g., a heart ventricle or heart atrium, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. 370. The AAV particle of any one of embodiments 67-267, 269, 271-277, 282, 285, 288, 289, 294-306, 321-349, and 369, which has an increased tropism for a heart cell or tissue, e.g., a heart ventricle or heart atrium, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139. 371. The AAV particle of any one of embodiments 67-267, 269, 271-277, 282, 285, 288, 289, 294-306, 321-349, 369, and 370, which delivers an increased level of a payload to a heart region, optionally wherein the level of the payload is increased by at least 1.5, at least 2, or at least 2.5-fold, as compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139, e.g., when measured by an assay, e.g., an IHC assay or a RT-ddPCR assay (e.g., as described in Example 2). 372. The AAV particle of any one of embodiments 67-267, 269, 271-277, 282, 285, 288, 289, 294-306, 321-349, and 369-371, which has an increased tropism for a heart cell or tissue, e.g., a heart ventricle or heart atrium, relative to the tropism of a an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982. 373. The AAV particle of any one of embodiments 67-267, 269, 271-277, 282, 285, 288, 289, 294-306, 321-349, and 369-372, which is enriched at least 4, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, or at least 50-fold in the heart compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982, e.g., when measured by an assay as described in Example 4.
Attorney Docket No.14640.0092-00304 374. The AAV particle of any one of embodiments 267, 290, 291, 321-328, 331, 332, and 344, which has an increased tropism for a muscle cell or tissue, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982. 375. The AAV particle of any one of embodiments 267, 290, 291, 321-328, 331, 332, 344, and 374, which is enriched at least 2, at least 3, at least 4, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, or at least 35-fold in the muscle compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982, e.g., when measured by an assay as described in Example 4. 376.^The AAV particle of embodiment 374 or 375, wherein the muscle cell or tissue is a heart muscle (e.g., a heart ventricle or a heart atrium, or both), a quadriceps muscle, or both. 377. The AAV particle of any one of embodiments 267, 292, 293, 321-328, 331, 332, and 344, which has an increased tropism for a liver cell or tissue, relative to the tropism of an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982. 378. The AAV particle of any one of embodiments 267, 292, 293, 321-328, 331, 332, 344, and 377, which is enriched at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 65, at least 70, at least 75, at least 80, at least 85, at least 90, at least 95, at least 100, at least 110, at least 115, at least 120, at least 130, at least 140, at least 150, at least 160, at least 170, at least 180, at least 185, or at least 190-fold in the liver compared to an AAV particle comprising an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982, e.g., when measured by an assay as described in Example 4. 379. The AAV particle of any one of embodiments 1-378, wherein the AAV particle comprises a viral genome comprising a nucleotide sequence encoding a Rep protein, e.g., a non-structural protein, wherein the Rep protein comprises a Rep78 protein, a Rep68 protein, Rep52 protein, and/or a Rep40 protein (e.g., a Rep78 protein and a Rep52 protein). 380. The AAV particle of any one of the embodiments provided herein, wherein the AAV particle comprises a nucleotide sequence encoding a Rep protein, e.g., a non-structural protein, wherein the Rep protein comprises a Rep78 protein, a Rep68 protein, Rep52 protein, and/or a Rep40 protein (e.g., a Rep78 protein and a Rep52 protein). 381. The AAV particle of embodiment 379 or 380, wherein the Rep78 protein, the Rep68 protein, the Rep52 protein, and/or the Rep40 protein are encoded by at least one Rep gene.
Attorney Docket No.14640.0092-00304 [Embodiments 382-445 are intentionally absent.] 446. A cell, e.g., a host cell, comprising the AAV particle of any one of embodiments 1-381. 447. The cell of embodiment 446, wherein the cell is a mammalian cell or an insect cell. 448. The cell of embodiment 446 or 447, wherein the cell is a cell of the CNS and/or the spinal cord. 449. The cell of any one of embodiments 446-448, wherein the cell is a cell of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum. 450. A method of making an AAV particle of embodiment 381, comprising (i) providing a cell comprising a viral genome comprising a frataxin (FXN)-encoding sequence and a nucleic acid encoding an AAV capsid variant; and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant; thereby making the AAV particle. 451. The method of embodiment 450, further comprising, prior to step (i), introducing a nucleic acid comprising the viral genome into the host cell. 452. The method of embodiment 450 or 451, further comprising, prior to step (i), introducing the nucleic acid encoding the AAV capsid variant into the cell. 453. The method of embodiment 452, wherein the second nucleic acid molecule is introduced into the host cell prior to, concurrently with, or after the first nucleic acid molecule. 454. A pharmaceutical composition comprising the AAV particle of any one of embodiments 1-381, and a pharmaceutically acceptable excipient. 455. A method of delivering a frataxin protein to a subject, comprising administering an effective amount of the pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381.
Attorney Docket No.14640.0092-00304 456. The method of embodiment 455, wherein the frataxin protein is delivered to a cell or tissue of the CNS (e.g., a cell or tissue of a brain or spinal cord region). 457. The method of embodiment 456, wherein the cell or tissue of the CNS is a cell or tissue of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, or cerebellum. [Embodiment 458 is intentionally absent.] 459. The method of embodiment 455 or 456, wherein the cell or tissue of the CNS comprises a neuron, a sensory neuron, a motor neuron, an astrocyte, a glial cell, or an oligodendrocyte. 460. The method of any one of embodiments 455-459, wherein the subject is a human. 461. The method of embodiment 460, wherein the subject has, has been diagnosed with having, or is at risk of having a genetic disorder. 462. The method of embodiment 460 or 461, wherein the subject has, has been diagnosed with having, or is at risk of having a neurological (e.g., a neurodegenerative disorder) or a neuromuscular disorder. 463. The method of embodiment 460 or 461, wherein the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency. 464. The method of embodiment 460 or 461, wherein the subject has, has been diagnosed with having, or is at risk of having Friedreich's Ataxia. 465. A method of treating a disorder associated with FXN deficiency in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381, optionally wherein the subject has, has been diagnosed with having, or is at risk of having the disorder associated with FXN deficiency. 466. A method of treating a neurological disorder, e.g., a neurodegenerative disorder, in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381, optionally wherein the subject has, has been diagnosed with having, or is at risk of having the neurological disorder.
Attorney Docket No.14640.0092-00304 467. A method of treating a muscular disorder or a neuromuscular disorder in a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381, optionally wherein the subject has, has been diagnosed with having, or is at risk of having the muscular disorder or the neuromuscular disorder. 468. The method of any one of embodiments 465-467, wherein the genetic disorder, neurological disorder, neurodegenerative disorder, muscular disorder, or neuromuscular disorder is Friedreich's Ataxia (FA). 469. The method of any one of embodiments 465-468, wherein the treatment comprises prevention of progression of the disease or disorder in the subject. 470. The method of any one of embodiments 460-469, wherein the subject is a human. 471. The method of any one of embodiments 465-470, wherein the AAV particle is administered to the subject intravenously, via intra-cisterna magna injection (ICM), intracerebrally, intrathecally, intracerebroventricularly, via intraparenchymal administration, or intramuscularly. 472. The method of any one of embodiments 465-471, wherein the AAV particle is administered to the subject via focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration. 473. The method of any one of embodiments 465-471, wherein the AAV particle is administered to the subject intravenously. [Embodiments 474-475 are intentionally absent.] 476. The method of any one of embodiments 465-474, wherein administration of the AAV particle results in an increased presence, level, and/or activity of a frataxin gene, mRNA, protein, or a combination thereof. 477. The pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381, for use in a method of delivering a payload to a cell or tissue. 478. The pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381, for use in a method of treating a genetic disorder, a neurological disorder, a neurodegenerative disorder, a muscular disorder, or a neuromuscular disorder.
Attorney Docket No.14640.0092-00304 479. The pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381, for use in the manufacture of a medicament. 480. Use of the pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381 in the manufacture of a medicament. 481. Use of the pharmaceutical composition of embodiment 454 or the AAV particle of any one of embodiments 1-381 in the manufacture of a medicament for treating a genetic disorder, a neurological disorder, a neurodegenerative disorder, a muscular disorder, or a neuromuscular disorder. 482. The AAV particle, pharmaceutical composition, cell, method, or use of any one of the embodiments provided herein, wherein the encoded frataxin (FXN) protein comprises an amino acid sequence of SEQ ID NO: 1825, or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the amino acid sequence of SEQ ID NO: 1825, wherein, optionally, the encoded FXN protein is a human FXN protein. 483. The AAV particle, pharmaceutical composition, cell, method, or use of any one of the embodiments provided herein, wherein the FXN-encoding sequence comprises a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to the nucleotide sequence of SEQ ID NO: 1824. 484. The AAV particle, pharmaceutical composition, cell, method, or use of any one of the embodiments provided herein, wherein the frataxin-encoding sequence comprises a nucleotide sequence that is at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to the nucleotide sequence of SEQ ID NO: 1824. 485. The AAV particle of embodiment 483 or embodiment 484, wherein the frataxin-encoding sequence comprises a nucleotide sequence at least 95% identical to the nucleotide sequence of SEQ ID NO: 1824. 486. The AAV particle of any one of embodiments 483-485, wherein the frataxin-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824.
Attorney Docket No.14640.0092-00304 487. The AAV particle of any one of embodiments 483-486, wherein the frataxin-encoding sequence consists of the nucleotide sequence of SEQ ID NO: 1824. 488. The AAV particle of any one of embodiments 482-487, wherein the AAV capsid variant comprises at least 4, at least 5, at least 6, at least 7, or 8 consecutive amino acids from the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), and wherein the AAV capsid variant comprises an amino acid sequence that is at least 95% identical to the amino acid sequence of SEQ ID NO: 739. 489. The AAV particle of embodiment 488, wherein: (i) the at least 4 consecutive amino acids comprise YPAE (SEQ ID NO: 21), optionally wherein YPAE (SEQ ID NO: 21) is present at positions 577-580 numbered according to the amino acid sequence of SEQ ID NO: 982; (ii) the at least 5 consecutive amino acids comprise YPAEV (SEQ ID NO: 1), optionally wherein YPAEV (SEQ ID NO: 1) is present at positions 577-581 numbered according to the amino acid sequence of SEQ ID NO: 982; (iii) the at least 6 consecutive amino acids comprise YPAEVV (SEQ ID NO: 725), optionally wherein YPAEVV (SEQ ID NO: 725) is present at positions 577-582 numbered according to the amino acid sequence of SEQ ID NO: 982; or (iv) the at least 7 consecutive amino acids comprise YPAEVVQ (SEQ ID NO: 726), optionally wherein YPAEVVQ (SEQ ID NO: 726) is present at positions 577-583 numbered according to the amino acid sequence of SEQ ID NO: 982. 490. The AAV particle of embodiment 488 or 489, wherein the AAV capsid variant comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) present at positions 577-584 numbered according to SEQ ID NO: 982. 491. The AAV particle of any one of embodiments 488-490, wherein the AAV capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 95% identity to the amino acid sequence of SEQ ID NO: 982; or (ii) a VP2 protein comprising an amino acid sequence having at least 95% identity to the amino acid sequence of SEQ ID NO: 738. 492. The AAV particle of any one of embodiments 488-491, wherein the AAV capsid variant comprises: (i) a VP1 protein comprising an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 982; (ii) a VP2 protein comprising an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 738; or
Attorney Docket No.14640.0092-00304 (iii) a VP3 protein comprising an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 739. 493. The AAV particle of any one of embodiments 488-492, wherein the AAV capsid variant comprises (i) a VP1 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 982; (ii) a VP2 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 738; or (iii) a VP3 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 739; or wherein the AAV capsid variant is encoded by the nucleotide sequence of SEQ ID NO: 984 or a sequence at least 90% identical thereto. 494. The AAV particle of any one of the embodiments provided herein, wherein the viral genome further comprises a promoter operably linked to the FXN-encoding sequence. 495. The AAV particle of embodiment 494, wherein the promoter comprises a human elongation factor 1Į-subunit (EF1Į) promoter, a cytomegalovirus (CMV) immediate-early enhancer and/or promoter, a chicken ȕ-actin (CBA) promoter, a CAG promoter, a ȕ glucuronidase (GUSB) promoter, a ubiquitin C (UBC) promoter, a neuron-specific enolase (NSE) promoter, a platelet-derived growth factor (PDGF) promoter, a platelet-derived growth factor B-chain (PDGF-ȕ) promoter, a intercellular adhesion molecule 2 (ICAM-2) promoter, a synapsin (Syn) promoter, a methyl-CpG binding protein 2 (MeCP2) promoter, a Ca2+/calmodulin-dependent protein kinase II (CaMKII) promoter, a metabotropic glutamate receptor 2 (mGluR2) promoter, a neurofilament light chain (NFL) or neurofilament heavy chain (NFH) promoter, a ȕ-globin minigene nȕ2 promoter, a preproenkephalin (PPE) promoter, a enkephalin (Enk) and excitatory amino acid transporter 2 (EAAT2) promoter, a glial fibrillary acidic protein (GFAP) promoter, a myelin basic protein (MBP) promoter, a cardiovascular promoter (e.g., ĮMHC, cTnT, and CMV-MLC2k), a liver promoter (e.g., hAAT, TBG), a skeletal muscle promoter (e.g., desmin, MCK, C512), or a functional fragment or truncation of any of the foregoing. 496. The AAV particle of embodiment 495, wherein the promoter comprises a CMV promoter. 497. The AAV particle of embodiment 495, wherein the promoter comprises a CBA promoter. 498. The AAV particle of embodiment 497, wherein the promoter is or comprises a truncated CBA promoter. 499. The AAV particle of embodiment 498, wherein the truncated CBA promoter is 50-400 nucleotides in length (e.g., 100-332 nucleotides in length).
Attorney Docket No.14640.0092-00304 500. The AAV particle of embodiment 499, wherein the truncated CBA promoter comprises or consists of the nucleotide sequence of any one of SEQ ID NOs: 1738, 1740, or 1742 or a nucleotide sequence that is at least 95% identical to any one of SEQ ID NOs: 1738, 1740, or 1742. 501. The AAV particle of embodiment 496, wherein the promoter comprises a truncated CMV promoter. 502. The AAV particle of embodiment 501, wherein the truncated CMV promoter is 109 nucleotides in length. 503. The AAV particle of embodiment 502, wherein the truncated CMV promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1750 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1750. 504. The AAV particle of any one of embodiments 494-593, wherein the viral genome further comprises a microRNA (miR) binding site, e.g., a miR binding site that modulates, e.g., reduces, expression of the FXN protein encoded by the viral genome in a cell or tissue where the corresponding miRNA is expressed. 505. The AAV particle of embodiment 504, wherein the miR binding site is fully or partially complementary to a miRNA expressed in a cell or tissue of the liver. 506. The AAV particle of embodiment 504 or embodiment 505, wherein the viral genome comprises at least 1, 2, 3, 4, or 5 copies of the miR binding site. 507. The AAV particle of embodiment 506, wherein the viral genome comprises at least 3 copies of the miR binding site, optionally wherein the viral genome has 3 copies of the miR binding site. 508. The AAV particle of embodiment 507, wherein the 3 copies of the miR binding site are identical, optionally wherein the 3 copies of the miR binding site are continuous. 509. The AAV particle of any one of embodiments 504-508, wherein the miR binding site is or comprises a miR122 binding site. 510. The AAV particle of any one of embodiments 504-509, wherein the viral genome encodes at least 1- 5 copies, e.g., 3 copies, of the miR122 binding site.
Attorney Docket No.14640.0092-00304 511. The AAV particle of embodiment 510, wherein the viral genome encodes 3 copies of the miR122 binding site, optionally wherein the 3 copies of the miR122 binding site are identical, further optionally wherein the 3 copies of the miR122 binding site are continuous. 512. The AAV particle of embodiment 510 or embodiment 511, wherein each copy of the miR122 binding site comprises or consists of the nucleotide sequence of SEQ ID NO: 1827. 513. The AAV particle of any one of embodiments 504-512, wherein the viral genome comprises a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence having one, two, three, four, five, six, seven, eight, nine, or at most ten substitutions relative to the nucleotide sequence of SEQ ID NO: 1826. 514. The AAV particle of any one of embodiments 494-513, wherein the viral genome further comprises at least one inverted terminal repeat (ITR) region, optionally wherein the at least one ITR region is an AAV2 ITR. 515. The AAV particle of embodiment 514, wherein the viral genome comprises a 5’ ITR region and a 3’ ITR region, optionally wherein the 5’ ITR region and the 3’ ITR region is each an AAV2 ITR. 516. The AAV particle of embodiment 515, wherein: the 5’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the 3’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 517. The AAV particle of any one of embodiments 494-517, wherein the viral genome further comprises an intron region. 518. The AAV particle of embodiment 517, wherein the intron region comprises an immediate-early 1 (ie1) intron region and/or a hemoglobin-beta (HB) intron region. 519. The AAV particle of any one of embodiments 494-518, wherein the viral genome further comprises an exon region. 520. The AAV particle of embodiment 519, wherein
Attorney Docket No.14640.0092-00304 (a) the exon region comprises an ie1 exon region and/or an HB exon region, and/or (b) the intron region comprises: an ie1 intron 1 comprising or consisting of the nucleotide sequence of SEQ ID NO: 1819 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or a hBglobin intron 2 comprising the nucleotide sequence of SEQ ID NO: 1820 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 521. The AAV particle of any one of embodiments 494-510, wherein the viral genome further comprises a polyadenylation (polyA) region. 522. The AAV particle of embodiment 521, wherein the polyA region comprises a human growth hormone (hGH) polyA region, optionally wherein the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 523. The AAV particle of any one of embodiments 494-522, wherein the encoded FXN protein is a wildtype frataxin protein, optionally wherein the encoded FXN protein is not a cynomolgus monkey FXN protein, further optionally wherein the encoded FXN protein is a human frataxin protein. 524. The AAV particle of embodiment 523, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1797. 525. The AAV particle of embodiment 523, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1801. 526. The AAV particle of embodiment 523, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1808.
Attorney Docket No.14640.0092-00304 527. The AAV particle of any one of embodiment 523, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1809. 528. The AAV particle of any one of embodiments 494-527, wherein the viral genome is single-stranded. 529. The AAV particle of any one of embodiments 494-527, wherein the viral genome is self- complementary. 530. The AAV particle of any one of the embodiments provided herein, wherein the viral genome comprises: (i) a promoter; and (ii) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824. 531. The AAV particle of embodiment 530, wherein the viral genome further comprises a 5’ inverted terminal repeat (ITR) region and/or a 3’ ITR region. 532. The AAV particle of embodiment 530 or embodiment 531, wherein the viral genome comprises, e.g., in 5’ to 3’ order: (i) a 5’ ITR region; (ii) a promoter; (iii) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (iv) at least one miR122 binding site; and (v) a 3’ ITR region. 533. The AAV particle of embodiment 532, wherein the viral genome comprises, e.g., in 5’ to 3’ order: (i) a 5’ ITR region; (ii) a promoter; (iii) an intron and/or exon region;
Attorney Docket No.14640.0092-00304 (iv) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; and (vi) a 3’ ITR region. 534. The AAV particle of embodiment 533, wherein the viral genome comprises, e.g., in 5’ to 3’ order: (i) a 5’ ITR region; (ii) a promoter; (iii) an intron and/or exon region; (iv) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; and (vi) a 3’ ITR region, optionally wherein the viral genome further comprises a filler sequence. 535. The AAV particle of embodiment 534, wherein: (i) the 5’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1742 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding region comprises the nucleotide sequence of SEQ ID NO: 1824 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
Attorney Docket No.14640.0092-00304 (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (vii) the 3’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; optionally wherein the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1841 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, further optionally wherein the filler region is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. 536. The AAV particle of embodiment 535, wherein the promoter consists of the nucleotide sequence of SEQ ID NO: 1742 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 537. The AAV particle of embodiment 534, wherein: (i) the 5’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1750 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding region comprises the nucleotide sequence of SEQ ID NO: 1824 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
Attorney Docket No.14640.0092-00304 (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (vii) the 3’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; optionally wherein the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1840 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, further optionally wherein the filler region is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. 538. The AAV particle of embodiment 537, wherein the promoter consists of the nucleotide sequence of SEQ ID NO: 1750 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 539. The AAV particle of embodiment 534, wherein: (i) the 5’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1738 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding region comprises the nucleotide sequence of SEQ ID NO: 1824 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
Attorney Docket No.14640.0092-00304 (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (vii) the 3’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; optionally wherein the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1838 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, further optionally wherein the filler region is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. 540. The AAV particle of embodiment 539, wherein the promoter consists of the nucleotide sequence of SEQ ID NO: 1738 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 541. The AAV particle of embodiment 534, wherein: (i) the 5’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1740 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding region comprises the nucleotide sequence of SEQ ID NO: 1824 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto;
Attorney Docket No.14640.0092-00304 (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical (e.g at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (vii) the 3’ ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; optionally wherein the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1839 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, further optionally wherein the filler region is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. 542. The AAV particle of embodiment 541, wherein the promoter consists of the nucleotide sequence of SEQ ID NO: 1740 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. [Embodiments 543-696 are intentionally absent.] 697. An adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome, wherein the AAV capsid variant comprises: (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, wherein the VP1 protein comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); (b) a VP2 protein comprising the amino acid sequence of SEQ ID NO: 738 or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, wherein the VP2 protein comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); and/or (c) a VP3 protein comprising the amino acid sequence of SEQ ID NO: 739 or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, wherein the VP3 protein comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); and wherein the viral genome comprises: (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least
Attorney Docket No.14640.0092-00304 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1797; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1801; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1808; or (d) the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1809. 698. The AAV particle of embodiment 697, wherein: (a) the VP1 protein comprises an amino acid sequence that is at least 95% identical to SEQ ID NO: 982; (b) a VP2 protein comprises an amino acid sequence that is at least 95% identical to SEQ ID NO: 738; and/or (c) a VP3 protein comprises an amino acid sequence that is at least 95% identical to SEQ ID NO: 739. 699. The AAV particle of embodiment 698, wherein: (a) the VP1 protein comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 982; (b) a VP2 protein comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 738; and/or (c) a VP3 protein comprises an amino acid sequence that is at least 99% identical to SEQ ID NO: 739. 700. The AAV particle of any one of embodiments 697-699, wherein: (a) the VP1 protein comprises the amino acid sequence of SEQ ID NO: 982; (b) the VP2 protein comprises the amino acid sequence of SEQ ID NO: 738; and/or (c) the VP3 protein comprises the amino acid sequence of SEQ ID NO: 739.
Attorney Docket No.14640.0092-00304 701. The AAV particle of embodiment 701, wherein the AAV capsid variant is encoded by the nucleotide sequence of SEQ ID NO: 984 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 702. The AAV particle of embodiment 701, wherein the AAV capsid variant is encoded by the nucleotide sequence of SEQ ID NO: 984. 703. The AAV particle of any one of embodiments 482-702, further comprising a nucleic acid encoding a Rep protein, wherein the Rep protein comprises a Rep78 protein, a Rep68 protein, a Rep52 protein, and/or a Rep40 protein. 704. A vector encoding the AAV particle of any one of embodiments 660-703. 705. A cell comprising the AAV particle of any one of embodiments 660-703 or the vector of embodiment 704. 706. The cell of embodiment 705, which is a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial cell. 707. A method of making an AAV particle of any one of embodiments 482-706, wherein the method comprises: (i) providing a cell comprising a viral genome comprising a frataxin (FXN)-encoding sequence and a nucleic acid encoding an AAV capsid variant; and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant; thereby making the AAV particle. 708. The method of embodiment 707, wherein: the viral genome comprises a nucleotide sequence of any one of SEQ ID NOs: 1797, 1801, 1808, and 1809, or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to any one of SEQ ID NOs: 1797, 1801, 1808, and 1809; and wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982. 709. The method of embodiment 708, further comprising, prior to step (i), introducing a nucleic acid comprising the viral genome into the cell.
Attorney Docket No.14640.0092-00304 710. The method of embodiment 708 or embodiment 709, further comprising, prior to step (i), introducing the nucleic acid encoding the AAV capsid variant into the cell. 711. The method of any one of embodiments 707-710, wherein the cell comprises a mammalian cell, an insect cell, or a bacterial cell. 712. The method of embodiment 711, wherein the cell is an HEK293 cell or an Sf9 cell. 713. A pharmaceutical composition comprising the AAV particle of any one of embodiments 485-703 and a pharmaceutically acceptable excipient. 714. A method of delivering a FXN protein to a subject, comprising administering to the subject an effective amount of the pharmaceutical composition of embodiment 713 or the AAV particle of any one of embodiments 482-704, thereby delivering the FXN protein. 715. The method of embodiment 714, wherein the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency, optionally wherein the disorder is Friedreich’s Ataxia (FA). 716. A method of treating a disorder associated with frataxin (FXN) deficiency in a subject, comprising administering an effective amount of the pharmaceutical composition of embodiment 713 or the AAV particle of any one of embodiments 485-703, thereby treating the disorder, optionally wherein the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency, further optionally wherein the disorder is FA. 717. The method of any one of embodiments 714-716, wherein the subject has a reduced level of FXN protein or gene expression as compared to a healthy individual. 718. The method of embodiment 716 or embodiment 717, wherein the treatment results in a 0.5-3x increase in the subject’s FXN protein level as compared to baseline. 719. The method of any one of embodiments 716-718, wherein the treatment results in amelioration of at least one symptom of FA. 720. The method of embodiment 719, wherein the at least one symptom of FA includes impaired sensory functions, impaired motor function, e.g., ataxia and/or involuntary movements, fatigue, chronic pain,
Attorney Docket No.14640.0092-00304 seizures, impaired speech, sleep disturbances, metabolic disorders, e.g., diabetes, and increased spasticity. 721. The method of any one of embodiments 716-718, wherein the treatment stabilizes, slows the progression of, or improves the subject’s FA as determined by the modified Friedreich Ataxia Rating Scale (mFARS), the Scale for the Assessment and Rating of Ataxia (SARA), and/or the International Cooperative Ataxia Rating Scale (ICARS). 722. The method of any one of embodiments 716-718, wherein the treatment slows the subject’s progression of FA as measured by mFARS, SARA, or ICARS relative to an individual with the disorder associated with FXN deficiency (e.g., FA) who has not been administered the pharmaceutical composition or the AAV particle. 723. The method of any one of embodiments 714-722, wherein the subject is a human. 724. The method of any one of embodiments 714-723, wherein the AAV particle is administered to the subject intravenously, intracerebrally (IC), via intrathalamic (ITH) administration, intramuscularly, intrathecally, intracerebroventricularly, via intraparenchymal administration, via focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration, or via intra-cisterna magna injection (ICM). 725. The method of any one of embodiments 714-724, wherein the AAV particle is delivered to a cell, tissue, or region of the CNS, e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum, or a combination thereof. 726. The method of any one of embodiments 714-725, further comprising evaluating (e.g., measuring the level of FXN gene, FXN mRNA, and/or FXN protein expression) in the subject (e.g., in a cell, tissue, or fluid of the subject), optionally wherein the level of FXN protein is measured by an assay described herein. 727. The method of embodiment 726, wherein the level of FXN protein expression is measured by an enzyme-linked immunosorbent assay (ELISA), a Western blot, an immunohistochemistry assay, or a frataxin biofluid assay. 728. The method of embodiment 726 or embodiment 727, wherein the cell or tissue is a cell or tissue of the central nervous system (e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen,
Attorney Docket No.14640.0092-00304 caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) or a peripheral cell or tissue (e.g., the liver, heart, and/or muscle). 729. The method of any one of embodiments 716-728, wherein the treatment results in an increase in: (i) the level of FXN protein or FXN gene expression in a cell, tissue, (e.g., a cell or tissue of the CNS, e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum), and/or fluid (e.g., CSF and/or serum) of the subject, optionally wherein the level of FXN protein or gene expression is increased by 0.5-3x as compared to baseline; and/or (ii) the number and/or level of viral genomes (VG) per cell in a CNS tissue (e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) of the subject relative to the number and/or level of VG per cell in a peripheral tissue of the subject. 730. The method of any one of embodiments 714-729, further comprising administering to the subject at least one additional therapeutic agent and/or therapy. 731. The method of embodiment 730, wherein the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy for treating the disorder associated with FXN deficiency (e.g., Friedreich’s ataxia), optionally wherein the at least one additional therapeutic agent and/or therapy comprises omaveloxolone or idebenone. 732. The method of embodiment 730, wherein the at least one additional therapeutic agent and/or therapy comprises an immunosuppressant, optionally wherein the immunosuppressant is a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine. 733. The pharmaceutical composition of embodiment 713 or the AAV particle of any one of embodiments 485-703 for use in the treatment of a disorder associated with FXN deficiency in a subject, optionally wherein the disorder is Friedreich’s Ataxia (FA). 734. Use of an effective amount of the pharmaceutical composition of embodiment 713 or the AAV particle of any one of embodiments 485-703 in the manufacture of a medicament for the treatment of a disorder associated with FXN deficiency in a subject, optionally wherein the disorder Friedreich’s Ataxia (FA).
Attorney Docket No.14640.0092-00304 735. The pharmaceutical composition or use of embodiment 733 or embodiment 734, wherein the subject has, has been diagnosed with having, or is at risk of having FA. [071] The details of various aspects or embodiments of the present disclosure are set forth below. Other features, objects, and advantages of the disclosure will be apparent from the description and the claims. In the description, the singular forms also include the plural unless the context clearly dictates otherwise. Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art in the field of this disclosure. In the case of conflict, the present description will control. BRIEF DESCRIPTION OF THE DRAWINGS [072] FIGs.1A-1D show immunohistochemistry images from various CNS and peripheral tissues isolated from NHPs (cynomolgus macaques) at 28 days post intravenous administration of AAV particles comprising the TTN-002 capsid variant (top panels) or AAV9 control capsid (bottom panels) and a self- complementary genome encoding a cynomolgus monkey frataxin protein fused to an HA tag driven by a chicken beta actin (CBA) heterologous promoter. FIG.1A shows, from left to right, the cerebellum (Purkinje cell layer), spinal cord (cervical), cortex (temporal), and the brainstem. FIG.1B shows, from left to right, the globus pallidus, the hippocampus, the thalamus, the putamen, and the dentate. FIG.1C shows, from left to right, the whole brain (level H), the whole brain (level K), and the cerebellum. FIG. 1D shows, from left to right, the spinal cord (thoracic), the DRG (thoracic), the liver, and the heart. [073] FIGs.2A-2D depict frataxin expression and the number of viral genomes per cell in the heart (FIG.2A); cerebellum (FIG.2B); lumbar and DRG (FIG.2C); and liver (FIG.2D). [074] FIG.3 shows an exemplary ITR-to-ITR construct encoding FXN. [075] FIG.4A shows biodistribution of AAV particles in the cerebellum of wildtype (“WT”) or frataxin-deficient (“FXN-deficient”) mice. The AAV particles comprise a TTN-002 capsid encapsulating a viral genome comprising SEQ ID NO: 1797 (“TTN-002_FXN”). [076] FIG.4B shows expression of human frataxin (“hFXN”) in the cerebellum of wildtype (“WT”) or frataxin-deficient (“FXN-deficient”) mice after administration of AAV particles comprising a TTN-002 capsid encapsulating a viral genome comprising SEQ ID NO: 1797 (“TTN-002_FXN”). DETAILED DESCRIPTION Overview [077] Described herein, inter alia, are compositions comprising an AAV capsid variant comprising a sequence encoding a FXN protein, e.g., a human FXN protein. In some embodiments, the present disclosure provides a method for delivering the AAV capsid variant comprising the sequence encoding the FXN protein to a cell or tissue in a subject. In some embodiments, the present disclosure provides a method for delivering the AAV capsid variant, thereby providing a FXN protein, e.g., a human FXN
Attorney Docket No.14640.0092-00304 protein, to a cell or tissue in a subject. The AAV capsid variants described herein have enhanced CNS tropism compared to other cells or tissues in the body, e.g., liver and/or the DRG. [078] AAV have proven to be useful as a biological tool due to their relatively simple structure, their ability to infect a wide range of cells (including quiescent and dividing cells) without integration into the host genome and without replicating, and their relatively benign immunogenic profile. Engineered AAV capsids with improved brain tropism represent an attractive solution to the limitations of CNS delivery. AAV-derived vectors are promising tools for clinical gene transfer because of their non-pathogenic nature, their low immunogenic profile, low rate of integration into the host genome, and long-term transgene expression in non-dividing cells. However, the transduction efficiency of naturally occurring AAVs in certain organs is too low for clinical applications, and capsid neutralization by pre- existing neutralizing antibodies may prevent treatment of a large proportion of patients. For these reasons, considerable efforts have been devoted to obtaining capsid variants with enhanced properties. Of many approaches tested so far, significant advances have resulted from directed evolution of AAV capsids using in vitro or in vivo selection of capsid variants created by capsid sequence randomization using either error-prone PCR, shuffling of various parent serotypes, or insertion of fully randomized short peptides at defined positions. [079] The genome of the virus may be modified to contain a minimum of components for the assembly of a functional recombinant virus, or viral particle, which is loaded with or engineered to target a particular tissue and express or deliver FXN. The genome of the virus may encode a FXN protein, and the viral particle comprising said genome may be delivered to a target cell, tissue, or organism. In some embodiments, the genome encodes a human FXN protein, e.g., a wildtype human FXN protein. In some embodiments, the target cell is a CNS cell. In some embodiments, the target tissue is a CNS tissue. The target CNS tissue may be brain tissue. [080] Without being bound by theory, it is believed, in some embodiments, that expression vectors, e.g., an adeno-associated viral vector (AAVs) or AAV particle, e.g., an AAV particle described herein, can be used to administer and/or deliver a gene encoding FXN (e.g., human FXN) preferentially to the CNS (e.g., the brain or the spinal cord). [081] Provided herein are compositions and methods which may provide for improved features compared to prior AAV-mediated enzyme replacement approaches, including (i) increased biodistribution of the AAV particle throughout the CNS (e.g., the cortex, striatum, thalamus, cerebellum, brainstem, and/or spinal cord) of the subject, (ii) elevated FXN expression, e.g., FXN mRNA or protein expression, in multiple brain regions (e.g., cortex, thalamus, and brain stem) of the subject; and (iii) reduced biodistribution of the AAV particle in the liver and/or DRG of the subject. [082] Also provided herein are AAV capsid variants (e.g., AAV5 capsid variants) with improved properties compared to wildtype AAV5 and/or wildtype AAV9, such as (i) increased penetrance through the blood brain barrier following intravenous administration, (ii) wider distribution throughout the multiple brain regions, e.g., temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate,
Attorney Docket No.14640.0092-00304 thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum, (iii) elevated FXN expression in multiple brain regions, (iv) wider distribution in one or more peripheral tissues, e.g., the heart and/or muscle, and/or (v) elevated FXN expression in one or more peripheral tissues. In some embodiments, the AAV capsids described herein enhance the delivery of FXN to multiple regions of the brain including, for example, the temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum. In some embodiments, the AAV capsids described herein enhance the delivery of FXN to the forebrain. In some embodiments, the AAV capsids described herein enhance the delivery of FXN to the frontal cortex. In some embodiments, the AAV capsids described herein enhance the delivery of FXN to the spinal cord. In some embodiments, the AAV capsids described herein enhance the delivery of FXN to the cerebellum. In some embodiments, the AAV capsids described herein enhance the delivery of FXN to the dentate nucleus. [083] As demonstrated in the Examples, the AAV capsid variants with enhanced brain tropism described herein are capable of significantly increasing FXN mRNA expression in the brain. [084] Thus, the compositions and methods described herein can be used in the treatment of Friedreich’s Ataxia (FA). In some embodiments, the disclosure provides an AAV particle comprising one of the AAV capsid variants disclosed herein and an AAV viral genome comprising a nucleotide sequence comprising a truncated promoter region and a sequence encoding a FXN protein (e.g., comprising the nucleotide sequence of any one of SEQ ID NOs: 1797, 1801, 1808, 1809) for use in treating FA. I. Compositions Adeno-associated viral (AAV) particles [085] AAVs have a genome of about 5,000 nucleotides in length and contain two open reading frames encoding the proteins responsible for replication (Rep) and the structural protein of the capsid (Cap). The open reading frames are flanked by two Inverted Terminal Repeat (ITR) sequences, which serve as the origin of replication of the viral genome. The wild-type AAV viral genome comprises nucleotide sequences for two open reading frames, one for the four non-structural Rep proteins (Rep78, Rep68, Rep52, Rep40, encoded by Rep genes) and one for the three capsid, or structural, proteins (VP1, VP2, VP3, encoded by capsid genes or Cap genes). The Rep proteins are important for replication and packaging, while the capsid proteins are assembled to create the protein shell of the AAV, or AAV capsid. Alternative splicing and alternate initiation codons and promoters result in the generation of four different Rep proteins from a single open reading frame and the generation of three capsid proteins from a single open reading frame. Though it varies by AAV serotype, as a non-limiting example, for AAV5 (SEQ ID NO: 138) VP1 refers to amino acids 1-724, VP2 refers to amino acids 137-724, and VP3 refers to amino acids 193-724. In some embodiments, e.g., for the amino acid sequence of SEQ ID NO: 982, VP1 comprises amino acids 1-731, VP2 comprises amino acids 137-731, and VP3 comprises amino acids 193-731. In other words, VP1 is the full-length capsid protein sequence, while VP2 and VP3 are shorter components of the whole. As a result, changes in the sequence in the VP3 region, are also changes to
Attorney Docket No.14640.0092-00304 VP1 and VP2, however, the percent difference as compared to the parent sequence will be greatest for VP3 since it is the shortest sequence of the three. Though described here in relation to the amino acid sequence, the nucleic acid sequence encoding these proteins can be similarly described. Together, the three capsid proteins assemble to create the AAV capsid. Without being bound by theory, the AAV capsid typically comprises a molar ratio of 1:1:10 of VP1:VP2:VP3. [086] The AAV vector typically requires a co-helper (e.g., adenovirus) to undergo productive infection in cells. In the absence of such helper functions, the AAV virions essentially enter host cells but do not integrate into the cells’ genome. [087] AAV vectors have been investigated for delivery of gene therapeutics because of several unique features. Non-limiting examples of the features include (i) the ability to infect both dividing and non-dividing cells; (ii) a broad host range for infectivity, including human cells; (iii) wild-type AAV has not been associated with any disease and has not been shown to replicate in infected cells; (iv) the lack of cell-mediated immune response against the vector, and (v) the non-integrative nature in a host chromosome thereby reducing potential for long-term genetic alterations. Moreover, infection with AAV vectors has minimal influence on changing the pattern of cellular gene expression (Stilwell and Samulski et al., Biotechniques, 2003, 34, 148, the contents of which are herein incorporated by reference in their entirety). [088] Typically, AAV vectors for FXN protein delivery may be recombinant viral vectors which are replication defective as they lack sequences encoding functional Rep and Cap proteins within the viral genome. In some cases, the defective AAV vectors may lack most or all coding sequences and essentially only contain one or two AAV ITR sequences and a sequence encoding a FXN protein. [089] In some embodiments, the AAV particles of the present disclosure may be introduced into mammalian cells. [090] AAV vectors may be modified to enhance the efficiency of delivery. Such modified AAV vectors of the present disclosure can be packaged efficiently and can be used to successfully infect the target cells at high frequency and with minimal toxicity. [091] In other embodiments, AAV particles of the present disclosure may be used to deliver FXN protein to the central nervous system (see, e.g., U.S. Pat. No.6,180,613; the contents of which are herein incorporated by reference in their entirety) or to specific tissues of the CNS. [092] It is understood that the compositions described herein may have additional conservative or non-essential amino acid substitutions, which do not have a substantial effect on their functions. [093] In some embodiments, AAV capsid variants disclosed herein comprise one or more modifications (e.g., one or more insertions and/or substitutions) in the loop VIII region of AAV5. In some embodiments, the AAV5 loop VIII region comprises amino acid positions 571-579, e.g., position 577, numbered according to SEQ ID NO: 138. In some embodiments, the AAV5 loop VIII region is present (is located) at amino acids corresponding to positions 571-579 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV5 loop VIII region (e.g., positions 571-579, e.g.,
Attorney Docket No.14640.0092-00304 position 577) protrudes above the 3-fold axis of symmetry, e.g., is a surface-exposed location in the AAV5 capsid, e.g., as described in Govindasamy et al. “Structural Insights into Adeno-Associated Virus Serotype 5,” Journal of Virology, 2013, 87(20):11187-11199 (the contents of which are hereby incorporated by reference in their entirety). In some embodiments, the term "loop" (e.g., as used in loop VIII) is used interchangeably herein with the term "variable region" (e.g., variable region VIII), or "VR" (e.g., VR-VIII). In some embodiments, loop VIII (e.g., VR-VIII) comprises positions 571-599 (e.g., amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)), numbered according to SEQ ID NO: 982. In some embodiments, the AAV5 loop VIII is present (is located) at amino acids corresponding to positions 571-599 (e.g., amino acids TNNQSSYPAEVVQKTAPATGTYNLQEIVP (SEQ ID NO: 756)) of the amino acid sequence of SEQ ID NO: 982. In some embodiments, loop VIII or variable region VIII (VR-VIII) is as described in Govindasamy et al. (supra) (the contents of which are hereby incorporated by reference in their entirety). [094] The AAV particles and payloads of the disclosure may be delivered to one or more target cells, tissues, organs, or organisms. In some embodiments, the AAV particles of the disclosure demonstrate enhanced tropism for a target cell type, tissue, or organ. As a non-limiting example, the AAV particle may have enhanced tropism for cells and tissues of the central or peripheral nervous systems (CNS and PNS, respectively). In some embodiments, an AAV particle of the disclosure may, in addition, or alternatively, have decreased tropism for a cell-type, tissue or organ. [095] In some embodiments, AAV are used as a biological tool due to a relatively simple structure, their ability to infect a wide range of cells (including quiescent and dividing cells) without integration into the host genome and without replicating, and their relatively benign immunogenic profile. The genome of the virus may be manipulated to contain a minimum of components for the assembly of a functional recombinant virus, or viral particle, which is loaded with or engineered to target a particular tissue and express or deliver a desired payload. [096] In some embodiments, the AAV is a recombinant AAV. In some embodiments, the wild-type AAV viral genome is a linear, single-stranded DNA (ssDNA) molecule approximately 5,000 nucleotides (nt) in length. In some embodiments, ITRs cap the viral genome at both the 5’ and the 3’ end, providing origins of replication for the viral genome. In some embodiments, an AAV viral genome comprises two ITR sequences. In some embodiments, the ITRs have a characteristic T-shaped hairpin structure defined by a self-complementary region (145nt in wild-type AAV) at the 5’ and 3’ ends of the ssDNA which form an energetically stable double stranded region. In some embodiments, the double stranded hairpin structures comprise multiple functions including, but not limited to, acting as an origin for DNA replication by functioning as primers for the endogenous DNA polymerase complex of the host viral replication cell. [097] AAV particles described herein may be produced recombinantly and may be based on AAV reference sequences. In addition to single stranded AAV viral genomes (e.g., ssAAVs), the present disclosure also provides for self-complementary AAV (scAAV) viral genomes. scAAV viral genomes
Attorney Docket No.14640.0092-00304 contain DNA strands which anneal together to form double stranded DNA. By skipping second strand synthesis, scAAVs allow for rapid expression in the transduced cell. In some embodiments, the AAV particle is an scAAV. In some embodiments, the AAV particle is an ssAAV. [098] Methods for producing and/or modifying AAV particles are disclosed in the art such as pseudotyped AAV vectors (PCT Patent Publication Nos. WO200028004; WO200123001; WO2004112727; WO2005005610; and WO2005072364, the content of each of which is incorporated herein by reference in its entirety). [099] As described herein, the AAV particles of the disclosure comprising an AAV capsid variant, and a viral genome, have enhanced tropism for a cell-type or a tissue, e.g., a CNS cell-type, region, or tissue. Capsid Peptides [0100] Disclosed herein are AAV particles comprising an AAV capsid variant comprising a peptide (e.g., inserted into a wildtype AAV capsid and/or in substitution of an amino acid or peptide in a wildtype AAV capsid) for enhanced or improved transduction of a target tissue (e.g., cells of the CNS or PNS). In some embodiments, the nucleic acid encoding the peptide is an isolated nucleic acid. In some embodiments, the nucleic acid encoding the peptide is a recombinant nucleic acid. [0101] In some embodiments, the peptide may increase distribution of an AAV particle to a cell, region, or tissue of the CNS. The cell of the CNS may be, but is not limited to, neurons (e.g., excitatory, inhibitory, motor, sensory, autonomic, sympathetic, parasympathetic, Purkinje, Betz, etc.), glial cells (e.g., microglia, astrocytes, oligodendrocytes) and/or supporting cells of the brain such as immune cells (e.g., T cells). The tissue of the CNS may be, but is not limited to, the cortex (e.g., frontal, parietal, occipital, temporal), thalamus, hypothalamus, striatum, putamen, caudate nucleus, hippocampus, entorhinal cortex, basal ganglia, or deep cerebellar nuclei. In some embodiments, the tissue of the CNS is a temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, cerebellum, cervical spinal cord, thoracic spinal cord, or lumbar spinal cord. [0102] In some embodiments, the peptide may modulate distribution of an AAV particle to a cell, region, or tissue of the CNS. In some embodiments, the peptide may decrease distribution of an AAV particle to the DRG. [0103] In some embodiments, the peptide may increase distribution of an AAV particle to the CNS (e.g., the cortex) after intravenous administration. In some embodiments, the peptide may increase distribution of an AAV particle to the CNS (e.g., the cortex) following focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration. [0104] In some embodiments, the peptide may modulate distribution of an AAV particle to the PNS (e.g., DRG) after intravenous administration. In some embodiments, the peptide may increase distribution of an AAV particle to non-DRG cells of the PNS following focused ultrasound (FUS), e.g.,
Attorney Docket No.14640.0092-00304 coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration. In some embodiments, the peptide may decrease distribution of an AAV particle to the DRG following focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration. [0105] In some embodiments, the peptide may increase distribution of an AAV particle to a cell, region, or tissue of a heart, e.g., a heart atrium or a heart ventricle. In some embodiments, the peptide may increase distribution of an AAV particle to a heart cell, region, or tissue after intravenous administration. [0106] In some embodiments, the peptide may increase distribution of an AAV particle to a cell, region, or tissue of a muscle. In some embodiments, the muscle is a heart muscle (e.g., a heart atrium or a heart ventricle) or a quadriceps. In some embodiments, the peptide may increase distribution of an AAV particle to a muscle cell, region, or tissue after intravenous administration. [0107] In some embodiments a peptide may comprise a sequence as set forth in Table 1 (e.g., comprising the amino acid sequence of any one of SEQ ID NOs: 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583-1587, 1590- 1593, 1598-1624, or 2064-2079). In some embodiments a peptide may comprise a sequence as set forth in Table 2A or 2B. In some embodiments, the peptide may comprise a sequence set forth in Tables 14 or 23-28. Table 1. Exemplary Peptide Sequences
Attorney Docket No.14640.0092-00304 Table 2A. Exemplary Peptide Sequences
Table 2B. Exemplary Peptide Sequences
[0108] In some embodiments, a peptide comprises an amino acid sequence having the formula [N2]- [N3], wherein [N2] comprises X1, X2, X3, X4, and X5 and [N3] comprises the amino acid sequence of VQK, VQN, EQK, VKK, VHK, VQQ, or LQK. In some embodiments, X1 of [N2] is Y, N, C, or T. In some embodiments, X2 of [N2] is P, E, K, T, or Q. In some embodiments, X3 of [N2] is A or P. In some embodiments, X4 of [N2] is E, S, D, or A. In some embodiments, X5 of [N2] is V, L, or E. In some embodiments, X1 of [N2] is Y. In some embodiments, X2 of [N2] is P. In some embodiments, X3 of [N2] is A. In some embodiments, X4 of [N2] is E. In some embodiments, X5 of [N2] is V. In some embodiments, the amino acid sequence of [N3] comprises VQK. In some embodiments, the amino acid sequence of [N3] consists of VQK. In some embodiments, [N2]-[N3] replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [0109] In some embodiments, a peptide comprises an amino acid sequence having the formula [N2]- [N3], wherein [N2] comprises X1, X2, X3, X4, and X5 and [N3] comprises the amino acid sequence of VQK, EQK, VKK, VHK, VQQ, or LQK. In some embodiments, [N3] comprises the amino acid sequence of VQK, EQK, or VKK. In some embodiments, [N3] comprises the amino acid sequence VQK. In some embodiments, [N3] consists of the amino acid sequence VQK. In some embodiments, X1 of [N2] is Y, N, or C. In some embodiments, X1 of [N2] is Y or N. In some embodiments, X2 of [N2] is P, K, T, or Q. In some embodiments, X2 of [N2] is P, T, or Q. In some embodiments, X3 of [N2] is A or P. In some embodiments, X3 of [N2] is A. In some embodiments, X4 of [N2] is E, S, or A. In some embodiments, X5 of [N2] is V, L, or E. In some embodiments, X5 of [N2] is V or L. In some embodiments, X1 of [N2] is Y. In some embodiments, X2 of [N2] is P. In some embodiments, X3 of [N2] is A. In some embodiments, X4 of [N2] is E. In some embodiments, X5 of [N2] is V. In some embodiments, [N2] comprises YPA, YPP, NKA, YTA, YQA, YTP, NPA, CPA, THA, PAE, PPS, KAE, TAE, QAE, TPS, PAA, HAS, AEV, PSL, AEE, or AAV. In some embodiments, [N2] comprises YPAE (SEQ ID NO: 21), YPPS (SEQ ID NO: 22), NKAE (SEQ ID NO: 23), YTAE (SEQ ID NO: 24), YQAE
Attorney Docket No.14640.0092-00304 (SEQ ID NO: 25), YTPS (SEQ ID NO: 26), YPAA (SEQ ID NO: 27), NPAE (SEQ ID NO: 28), CPAE (SEQ ID NO: 29), THAS (SEQ ID NO: 30), PAEV (SEQ ID NO: 17), PPSL (SEQ ID NO: 31), KAEV (SEQ ID NO: 32), TAEV (SEQ ID NO: 16), PAEE (SEQ ID NO: 18), QAEV (SEQ ID NO: 15), TPSL (SEQ ID NO: 33), PAAV (SEQ ID NO: 34), or QAEE (SEQ ID NO: 35). In some embodiments, [N2] is or comprises YPAEV (SEQ ID NO: 1), YPPSL (SEQ ID NO: 2), NKAEV (SEQ ID NO: 3), YTAEV (SEQ ID NO: 4), YPAEE (SEQ ID NO: 5), YQAEV (SEQ ID NO: 6), YTPSL (SEQ ID NO: 7), YPAAV (SEQ ID NO: 8), NPAEV (SEQ ID NO: 9), CPAEV (SEQ ID NO: 10), or YQAEE (SEQ ID NO: 11). In some embodiments, [N2] comprises the amino acid sequence of YPAEV (SEQ ID NO: 1). In some embodiments, the amino acid sequence of [N2] consists of YPAEV (SEQ ID NO: 1). In some embodiments, [N2]-[N3] comprises the amino acid sequence of AEVVQK (SEQ ID NO: 36), PSLVQK (SEQ ID NO: 37), AEVEQK (SEQ ID NO: 38), AEEVQK (SEQ ID NO: 39), PSLEQK (SEQ ID NO: 40), PSLVKK (SEQ ID NO: 41), AEVVKK (SEQ ID NO: 42), AEVVHK (SEQ ID NO: 43), AAVVQK (SEQ ID NO: 44), AEVVQQ (SEQ ID NO: 45), or AEVLQK (SEQ ID NO: 46). In some embodiments, [N2]-[N3] comprises the amino acid sequence PAEVVQK (SEQ ID NO: 20), PPSLVQK (SEQ ID NO: 47), KAEVVQK (SEQ ID NO: 48), TAEVVQK (SEQ ID NO: 49), PAEVEQK (SEQ ID NO: 50), PAEEVQK (SEQ ID NO: 51), QAEVVQK (SEQ ID NO: 52), TPSLVQK (SEQ ID NO: 53), PPSLEQK (SEQ ID NO: 54), PPSLVKK (SEQ ID NO: 55), PAEVVKK (SEQ ID NO: 56), PAEVVHK (SEQ ID NO: 57), PAAVVQK (SEQ ID NO: 58), PAEVVQQ (SEQ ID NO: 59), TAEVVKK (SEQ ID NO: 60), PAEVLQK (SEQ ID NO: 61), or QAEEVQK (SEQ ID NO: 62). In some embodiments, [N2]-[N3] is or comprises YPAEVVQK (SEQ ID NO: 943), YPPSLVQK (SEQ ID NO: 946), NKAEVVQK (SEQ ID NO: 947), YTAEVVQK (SEQ ID NO: 948), YPAEVEQK (SEQ ID NO: 949), YPAEEVQK (SEQ ID NO: 950), YQAEVVQK (SEQ ID NO: 951), YTPSLVQK (SEQ ID NO: 952), YPPSLEQK (SEQ ID NO: 953), YPPSLVKK (SEQ ID NO: 954), YPAEVVKK (SEQ ID NO: 955), YPAEVVHK (SEQ ID NO: 956), YPAAVVQK (SEQ ID NO: 957), NPAEVVQK (SEQ ID NO: 958), YPAEVVQQ (SEQ ID NO: 959), CPAEVVQK (SEQ ID NO: 960), YTAEVVKK (SEQ ID NO: 961), YPAEVLQK (SEQ ID NO: 962), or YQAEEVQK (SEQ ID NO: 963); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, or 7 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N2]-[N3] is YPAEVVQK (SEQ ID NO: 943). In some embodiments, [N2]-[N3] comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). In some embodiments, [N2]-[N3] (e.g., comprising the amino acid sequence of SEQ ID NO: 943) replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [0110] In some embodiments, the peptide comprising the amino acid sequence comprising the formula of [N2]-[N3] further comprises [N1], which comprises XD, XE, and XF. In some embodiments, XD of [N1] is Q, T, S, A, I, L, or H. In some embodiments, XE of [N1] is S, G, A, or R. In some
Attorney Docket No.14640.0092-00304 embodiments, XF of [N1] is S, K, L, R, A, or T. In some embodiments, [N1] comprises SK, SL, SS, SR, GA, GS, AS, ST, RS, QS, TS, AG, IG, QA, LG, HS, LS, or QR. In some embodiments, [N1] is or comprises QSS, QSK, TSL, SSS, QSR, AGA, IGS, QAS, ASS, LGS, QST, HSS, LSS, or QRS. In some embodiments, the amino acid sequence of [N1] is QSS. In some embodiments, [N1]-[N2] comprises SSYPA (SEQ ID NO: 63), SKYPA (SEQ ID NO: 64), SLYPA (SEQ ID NO: 65), SRYPA (SEQ ID NO: 66), SSYPP (SEQ ID NO: 67), GAYPA (SEQ ID NO: 68), GSYPA (SEQ ID NO: 69), ASYPA (SEQ ID NO: 70), STNKA (SEQ ID NO: 71), SSYTA (SEQ ID NO: 72), SSYQA (SEQ ID NO: 73), SSYTP (SEQ ID NO: 74), SSNPA (SEQ ID NO: 75), SLCPA (SEQ ID NO: 76), RSYTA (SEQ ID NO: 77), or SSTHA (SEQ ID NO: 78). In some embodiments, [N1]-[N2] comprises SSYPAE (SEQ ID NO: 79), SKYPAE (SEQ ID NO: 80), SLYPAE (SEQ ID NO: 81), SRYPAE (SEQ ID NO: 82), SSYPPS (SEQ ID NO: 83), GAYPAE (SEQ ID NO: 84), GSYPAE (SEQ ID NO: 85), ASYPAE (SEQ ID NO: 86), STNKAE (SEQ ID NO: 87), SSYTAE (SEQ ID NO: 88), SSYQAE (SEQ ID NO: 89), SSYTPS (SEQ ID NO: 90), SSYPAA (SEQ ID NO: 91), SSNPAE (SEQ ID NO: 92), SLCPAE (SEQ ID NO: 93), RSYTAE (SEQ ID NO: 94), SSTHAS (SEQ ID NO: 95). In some embodiments, [N1]-[N2] is or comprises QSSYPAEV (SEQ ID NO: 96), QSKYPAEV (SEQ ID NO: 97), TSLYPAEV (SEQ ID NO: 98), SSSYPAEV (SEQ ID NO: 99), QSRYPAEV (SEQ ID NO: 100), QSSYPPSL (SEQ ID NO: 101), AGAYPAEV (SEQ ID NO: 102), IGSYPAEV (SEQ ID NO: 103), QASYPAEV (SEQ ID NO: 104), ASSYPAEV (SEQ ID NO: 105), LGSYPAEV (SEQ ID NO: 106), QSTNKAEV (SEQ ID NO: 107), HSSYPAEV (SEQ ID NO: 108), SSSYTAEV (SEQ ID NO: 109), TSLYPAEE (SEQ ID NO: 110), ASSYQAEV (SEQ ID NO: 111), QSSYTPSL (SEQ ID NO: 112), QSRYPAEE (SEQ ID NO: 113), LSSYQAEV (SEQ ID NO: 114), HSSYPAAV (SEQ ID NO: 115), QSSNPAEV (SEQ ID NO: 116), QSSYTAEV (SEQ ID NO: 117), TSLCPAEV (SEQ ID NO: 118), QRSYTAEV (SEQ ID NO: 119), or QSSYQAEE (SEQ ID NO: 120); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, or 7 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, the amino acid sequence of [N1]-[N2] is QSSYPAEV (SEQ ID NO: 96). In some embodiments, [N1]-[N2]-[N3] comprises SSYPAEVVQ (SEQ ID NO: 121), SKYPAEVVQ (SEQ ID NO: 122), SLYPAEVVQ (SEQ ID NO: 123), SRYPAEVVQ (SEQ ID NO: 124), SSYPPSLVQ (SEQ ID NO: 125), GAYPAEVVQ (SEQ ID NO: 126), GSYPAEVVQ (SEQ ID NO: 127), ASYPAEVVQ (SEQ ID NO: 128), STNKAEVVQ (SEQ ID NO: 129), SSYTAEVVQ (SEQ ID NO: 130), SKYPAEVEQ (SEQ ID NO: 131), SLYPAEEVQ (SEQ ID NO: 132), SSYQAEVVQ (SEQ ID NO: 133), SSYTPSLVQ (SEQ ID NO: 134), SRYPAEEVQ (SEQ ID NO: 135), SSYPPSLEQ (SEQ ID NO: 136), SSYPPSLVK (SEQ ID NO: 140), SSYPAEVVK (SEQ ID NO: 141), SKYPAEVVH (SEQ ID NO: 142), SSYPAAVVQ (SEQ ID NO: 143), SSNPAEVVQ (SEQ ID NO: 144), SLCPAEVVQ (SEQ ID NO: 145), RSYTAEVVQ (SEQ ID NO: 146), SSYTAEVVK (SEQ ID NO: 147), SSYPAEVLQ
Attorney Docket No.14640.0092-00304 (SEQ ID NO: 148), or SSYQAEEVQ (SEQ ID NO: 149). In some embodiments, [N1]-[N2]-[N3] is or comprises QSSYPAEVVQK (SEQ ID NO: 150), QSKYPAEVVQK (SEQ ID NO: 151), TSLYPAEVVQK (SEQ ID NO: 152), SSSYPAEVVQK (SEQ ID NO: 153), QSRYPAEVVQK (SEQ ID NO: 154), QSSYPPSLVQK (SEQ ID NO: 155), AGAYPAEVVQK (SEQ ID NO: 156), IGSYPAEVVQK (SEQ ID NO: 157), QASYPAEVVQK (SEQ ID NO: 158), ASSYPAEVVQK (SEQ ID NO: 159), LGSYPAEVVQK (SEQ ID NO: 160), QSTNKAEVVQK (SEQ ID NO: 161), HSSYPAEVVQK (SEQ ID NO: 162), SSSYTAEVVQK (SEQ ID NO: 163), QSKYPAEVEQK (SEQ ID NO: 164), TSLYPAEEVQK (SEQ ID NO: 165), ASSYQAEVVQK (SEQ ID NO: 166), QSSYTPSLVQK (SEQ ID NO: 167), QSRYPAEEVQK (SEQ ID NO: 168), QSSYPPSLEQK (SEQ ID NO: 169), QSSYPPSLVKK (SEQ ID NO: 170), LSSYQAEVVQK (SEQ ID NO: 171), SSSYPAEVVKK (SEQ ID NO: 172), QSKYPAEVVHK (SEQ ID NO: 173), HSSYPAAVVQK (SEQ ID NO: 174), QSSNPAEVVQK (SEQ ID NO: 175), SSSYPAEVVQQ (SEQ ID NO: 176), QSSYTAEVVQK (SEQ ID NO: 177), TSLCPAEVVQK (SEQ ID NO: 178), QRSYTAEVVQK (SEQ ID NO: 179), QSSYTAEVVKK (SEQ ID NO: 180), HSSYPAEVLQK (SEQ ID NO: 181), or QSSYQAEEVQK (SEQ ID NO: 182); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, the amino acid sequence of [N1]-[N2]-[N3] is QSSYPAEVVQK (SEQ ID NO: 150). In some embodiments, [N2]-[N3] is present immediately subsequent to [N1] and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [0111] In some embodiments, the peptide comprising the amino acid sequence comprising the formula [N2]-[N3] further comprises [N0], wherein [N0] comprises XA, XB, and XC. In some embodiments, XA of [N0] is T, I, or N. In some embodiments, XB of [N0] is N. In some embodiments, XC of [N0] is N, T, S, or K. In some embodiments, [N0] comprises TN, IN, NN, NT, NS, or NK. In some embodiments, [N0] is or comprises TNN, TNT, INN, TNS, NNN, or TNK. In some embodiments, the amino acid sequence of [N0] is TNN. In some embodiments, [N0]-[N1] is or comprises TNNQSS (SEQ ID NO: 183), TNNQSK (SEQ ID NO: 184), TNNTSL (SEQ ID NO: 185), TNNSSS (SEQ ID NO: 186), TNNQSR (SEQ ID NO: 187), TNNAGA (SEQ ID NO: 188), TNNIGS (SEQ ID NO: 189), TNNQAS (SEQ ID NO: 190), TNTASS (SEQ ID NO: 191), TNNLGS (SEQ ID NO: 192), TNNQST (SEQ ID NO: 193), TNNHSS (SEQ ID NO: 194), TNNQSK (SEQ ID NO: 184), TNNLSS (SEQ ID NO: 195), INNQSS (SEQ ID NO: 196), TNSQSS (SEQ ID NO: 197), NNNQSR (SEQ ID NO: 198), TNSTSL (SEQ ID NO: 199), TNNQRS (SEQ ID NO: 200), or TNKQAS (SEQ ID NO: 201); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or
Attorney Docket No.14640.0092-00304 an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N0]-[N1] is TNNQSS (SEQ ID NO: 183). In some embodiments, [N0]-[N1]-[N2]-[N3] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500), TNNQSKYPAEVVQK (SEQ ID NO: 503), TNNTSLYPAEVVQK (SEQ ID NO: 506), TNNSSSYPAEVVQK (SEQ ID NO: 508), TNNQSRYPAEVVQK (SEQ ID NO: 510), TNNQSSYPPSLVQK (SEQ ID NO: 512), TNNAGAYPAEVVQK (SEQ ID NO: 513), TNNIGSYPAEVVQK (SEQ ID NO: 514), TNNQASYPAEVVQK (SEQ ID NO: 517), TNTASSYPAEVVQK (SEQ ID NO: 520), TNNLGSYPAEVVQK (SEQ ID NO: 523), TNNQSTNKAEVVQK (SEQ ID NO: 524), TNNHSSYPAEVVQK (SEQ ID NO: 525), TNNSSSYTAEVVQK (SEQ ID NO: 526), TNNQSKYPAEVEQK (SEQ ID NO: 529), TNNTSLYPAEEVQK (SEQ ID NO: 530), TNTASSYQAEVVQK (SEQ ID NO: 531), TNNQSSYTPSLVQK (SEQ ID NO: 533), TNNQSRYPAEEVQK (SEQ ID NO: 534), TNNQSSYPPSLEQK (SEQ ID NO: 535), TNNQSSYPPSLVKK (SEQ ID NO: 536), TNNLSSYQAEVVQK (SEQ ID NO: 539), TNNSSSYPAEVVKK (SEQ ID NO: 540), TNNQSKYPAEVVHK (SEQ ID NO: 542), INNQSSYPAEVVQK (SEQ ID NO: 543), TNNHSSYPAAVVQK (SEQ ID NO: 545), TNSQSSNPAEVVQK (SEQ ID NO: 548), TNNSSSYPAEVVQQ (SEQ ID NO: 551), NNNQSRYPAEVVQK (SEQ ID NO: 552), TNNQSSYTAEVVQK (SEQ ID NO: 553), TNNTSLCPAEVVQK (SEQ ID NO: 554), TNSTSLYPAEVVQK (SEQ ID NO: 556), TNNQRSYTAEVVQK (SEQ ID NO: 557), TNNQSSYTAEVVKK (SEQ ID NO: 558), TNNHSSYPAEVLQK (SEQ ID NO: 560), TNNQSSYQAEEVQK (SEQ ID NO: 562) or TNKQASYPAEVVQK (SEQ ID NO: 563); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N0]-[N1]-[N2]-[N3] is TNNQSSYPAEVVQK (SEQ ID NO: 500). In some embodiments, [N2]-[N3] is present immediately subsequent to [N0]-[N1] and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [0112] In some embodiments, the peptide comprising the amino acid sequence comprising the formula [N2]-[N3] further comprises [N4], which comprises XG and XH. In some embodiments, XG of [N4] is T, P, or N. In some embodiments, XG of [N4] is T. In some embodiments, XH of [N4] is A. In some embodiments, [N4] is or comprises TA, PA, or NA. In some embodiments, [N4] is TA. In some embodiments, [N3]-[N4] is or comprises VQKTA (SEQ ID NO: 564), EQKTA (SEQ ID NO: 565), VKKTA (SEQ ID NO: 566), VQKPA (SEQ ID NO: 567), VHKTA (SEQ ID NO: 568), VQQTA (SEQ ID NO: 569), VQKNA (SEQ ID NO: 570), or LQKTA (SEQ ID NO: 571); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, or 4 amino
Attorney Docket No.14640.0092-00304 acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N3]-[N4] is VQKTA (SEQ ID NO: 564). In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is or comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNTSLYPAEVVQKTA (SEQ ID NO: 1232), TNNSSSYPAEVVQKTA (SEQ ID NO: 1539), TNNQSRYPAEVVQKTA (SEQ ID NO: 1327), TNNQSSYPPSLVQKTA (SEQ ID NO: 1300), TNNAGAYPAEVVQKTA (SEQ ID NO: 1021), TNNIGSYPAEVVQKTA (SEQ ID NO: 1112), TNNQASYPAEVVQKTA (SEQ ID NO: 1194), TNTASSYPAEVVQKTA (SEQ ID NO: 1575), TNNLGSYPAEVVQKTA (SEQ ID NO: 1027), TNNQSTNKAEVVQKTA (SEQ ID NO: 1578), TNNHSSYPAEVVQKTA (SEQ ID NO: 1310), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNSSSYTAEVVQKTA (SEQ ID NO: 1214), TNNQSKYPAEVEQKTA (SEQ ID NO: 1254), TNNTSLYPAEEVQKTA (SEQ ID NO: 1583), TNTASSYQAEVVQKTA (SEQ ID NO: 1584), TNNQSSYTPSLVQKTA (SEQ ID NO: 1585), TNNQSRYPAEEVQKTA (SEQ ID NO: 1342), TNNQSSYPPSLEQKTA (SEQ ID NO: 1590), TNNQSSYPPSLVKKTA (SEQ ID NO: 1591), TNNLSSYQAEVVQKTA (SEQ ID NO: 1592), TNNQSSYPPSLVQKPA (SEQ ID NO: 1593), TNNSSSYPAEVVKKTA (SEQ ID NO: 1331), TNNQSKYPAEVVHKTA (SEQ ID NO: 1453), TNNSSSYPAEVVQKPA (SEQ ID NO: 1142), INNQSSYPAEVVQKTA (SEQ ID NO: 1024), TNNHSSYPAAVVQKTA (SEQ ID NO: 1598), TNSQSSNPAEVVQKTA (SEQ ID NO: 1599), TNNSSSYPAEVVQQTA (SEQ ID NO: 1419), NNNQSRYPAEVVQKTA (SEQ ID NO: 1601), TNNQSSYTAEVVQKNA (SEQ ID NO: 1602), TNNTSLCPAEVVQKTA (SEQ ID NO: 1603), TNSTSLYPAEVVQKTA (SEQ ID NO: 1605), TNNQRSYTAEVVQKTA (SEQ ID NO: 1604), TNNQSSYTAEVVKKTA (SEQ ID NO: 1606), TNNHSSYPAEVLQKTA (SEQ ID NO: 1607), TNNQSSYQAEEVQKTA (SEQ ID NO: 1608), or TNKQASYPAEVVQKTA (SEQ ID NO: 1587); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). In some embodiments, [N2]-[N3] is present immediately subsequent to [N0]-[N1], immediately precedes [N4], and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [0113] In some embodiments, a peptide described herein comprises the formula [N2]-[N3], wherein [N2] comprises X1, X2, X3, X4, and X5 and [N3] comprises the amino acid sequence of VQK or VQN. In some embodiments, [N3] comprises the amino acid sequence VQK. In some embodiments, X1 of [N2] is Y or T. In some embodiments, X2 of [N2] is Q, T, P, or E. In some embodiments, X3 of [N2] is A. In
Attorney Docket No.14640.0092-00304 some embodiments, X4 of [N2] is E or D. In some embodiments, X4 of [N2] is E or D. In some embodiments, X5 of [N2] is V or E. In some embodiments, X1 of [N2] is Y. In some embodiments, X2 of [N2] is P. In some embodiments, X3 of [N2] is A. In some embodiments, X4 of [N2] is E. In some embodiments, X5 of [N2] is V. In some embodiments, [N2] comprises YP, YQ, YT, TE, QA, TA, PA, EA, EV, EE, DV, AE, or AD. In some embodiments, [N2] comprises YPA, YQA, YTA, TEA, QAE, TAE, PAE, EAE, PAD, AEV, AEE, or ADV. In some embodiments, [N2] comprises YPAE (SEQ ID NO: 21), YQAE (SEQ ID NO: 25), YTAE (SEQ ID NO: 24), TEAE (SEQ ID NO: 587), YPAD (SEQ ID NO: 588), QAEV (SEQ ID NO: 15), TAEV (SEQ ID NO: 16), PAEV (SEQ ID NO: 17), PAEE (SEQ ID NO: 18), EAEV (SEQ ID NO: 590), or PADV (SEQ ID NO: 19). In some embodiments, [N2] is or comprises YPAEV (SEQ ID NO: 1), YQAEV (SEQ ID NO: 6), YTAEV (SEQ ID NO: 4), YPAEE (SEQ ID NO: 5), TEAEV (SEQ ID NO: 12), or YPADV (SEQ ID NO: 13). In some embodiments, [N2] is YPAEV (SEQ ID NO: 1). In some embodiments, [N2]-[N3] comprises AEVVQK (SEQ ID NO: 36), AEEVQK (SEQ ID NO: 39), AEVVQN (SEQ ID NO: 591), or ADVVQK (SEQ ID NO: 593). In some embodiments, [N2]-[N3] comprises PAEVVQN (SEQ ID NO: 594), QAEVVQK (SEQ ID NO: 52), TAEVVQK (SEQ ID NO: 49), PAEVVQK (SEQ ID NO: 20), PAEEVQK (SEQ ID NO: 51), EAEVVQK (SEQ ID NO: 595), or PADVVQK (SEQ ID NO: 596). In some embodiments, [N2]-[N3] is or comprises YPAEVVQK (SEQ ID NO: 943), YQAEVVQK (SEQ ID NO: 951), YTAEVVQK (SEQ ID NO: 948), YPAEEVQK (SEQ ID NO: 950), YPAEVVQN (SEQ ID NO: 964), TEAEVVQK (SEQ ID NO: 965), or YPADVVQK (SEQ ID NO: 966); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, or 7 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N2]-[N3] is YPAEVVQK (SEQ ID NO: 943). In some embodiments, [N2]-[N3] (e.g., comprising the amino acid sequence of SEQ ID NO: 943) replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [0114] In some embodiments, the peptide comprising the amino acid sequence comprising the formula of [N2]-[N3] further comprises [N1], which comprises XD, XE, and XF. In some embodiments, XD of [N1] is Q or S. In some embodiments, XE of [N1] is S, L, A, or T. In some embodiments, XF of [N1] is S, Y, or T. In some embodiments, [N1] comprises QS, SL, SA, QT, LS, LY, AT, TS, or SS. In some embodiments, [N1] is or comprises QSS, SLS, SLY, SAT, or QTS. In some embodiments, [N1] is QSS. In some embodiments, [N1]-[N2] comprises SSYPA (SEQ ID NO: 63), LSYQA (SEQ ID NO: 597), LSYTA (SEQ ID NO: 598), LYYPA (SEQ ID NO: 600), ATYPA (SEQ ID NO: 601), LSYPA (SEQ ID NO: 603), or TSTEA (SEQ ID NO: 605). In some embodiments, [N1]-[N2] comprises SSYPAE (SEQ ID NO: 79), LSYQAE (SEQ ID NO: 607), LSYTAE (SEQ ID NO: 610), LYYPAE (SEQ ID NO: 611), ATYPAE (SEQ ID NO: 613), LSYPAE (SEQ ID NO: 616), TSTEAE (SEQ ID NO: 619), or LSYPAD (SEQ ID NO: 621). In some embodiments, [N1]-[N2] is or comprises QSSYPAEV (SEQ ID
Attorney Docket No.14640.0092-00304 NO: 96), SLSYQAEV (SEQ ID NO: 622), SLSYTAEV (SEQ ID NO: 623), SLYYPAEV (SEQ ID NO: 624), SATYPAEV (SEQ ID NO: 625), SLSYPAEV (SEQ ID NO: 629), SLSYPAEE (SEQ ID NO: 632), QTSTEAEV (SEQ ID NO: 633), or SLSYPADV (SEQ ID NO: 634); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, or 7 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N1]-[N2] is QSSYPAEV (SEQ ID NO: 96). In some embodiments, [N1]-[N2]-[N3] is or comprises QSSYPAEVVQK (SEQ ID NO: 150), SLSYQAEVVQK (SEQ ID NO: 635), SLSYTAEVVQK (SEQ ID NO: 637), SLYYPAEVVQK (SEQ ID NO: 639), SATYPAEVVQK (SEQ ID NO: 641), SLSYPAEVVQK (SEQ ID NO: 642), SLSYPAEEVQK (SEQ ID NO: 643), SLSYPAEVVQN (SEQ ID NO: 644), QTSTEAEVVQK (SEQ ID NO: 645), or SLSYPADVVQK (SEQ ID NO: 646); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N1]-[N2]-[N3] is QSSYPAEVVQK (SEQ ID NO: 150). In some embodiments, [N2]-[N3] is present immediately subsequent to [N1] and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [0115] In some embodiments, the peptide comprising the amino acid sequence comprising the formula [N2]-[N3] further comprises [N0], wherein [N0] comprises XA, XB, and XC. In some embodiments, XA of [N0] is T. In some embodiments, XB of [N0] is N. In some embodiments, XC of [N0] is N, T, S, or K. In some embodiments, [N0] comprises TN, NS, NT, NN, or NK. In some embodiments, [N0] is or comprises TNS, TNT, TNN, or TNK. In some embodiments, [N0] is TNN. In some embodiments, [N0]-[N1] is or comprises TNNQSS (SEQ ID NO: 183), TNSSLS (SEQ ID NO: 647), TNSSLY (SEQ ID NO: 648), TNTSAT (SEQ ID NO: 649), TNNQTS (SEQ ID NO: 650), or TNKSAT (SEQ ID NO: 651); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N0]-[N1] is TNNQSS (SEQ ID NO: 183). In some embodiments, [N0]-[N1]-[N2]-[N3] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500), TNSSLSYQAEVVQK (SEQ ID NO: 652), TNSSLSYTAEVVQK (SEQ ID NO: 654), TNSSLYYPAEVVQK (SEQ ID NO: 655), TNTSATYPAEVVQK (SEQ ID NO: 656), TNSSLSYPAEVVQK (SEQ ID NO: 657),
Attorney Docket No.14640.0092-00304 TNSSLSYPAEEVQK (SEQ ID NO: 658), TNSSLSYPAEVVQN (SEQ ID NO: 660), TNNQTSTEAEVVQK (SEQ ID NO: 662), TNKSATYPAEVVQK (SEQ ID NO: 663), or TNSSLSYPADVVQK (SEQ ID NO: 665); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N0]-[N1]-[N2]-[N3] is TNNQSSYPAEVVQK (SEQ ID NO: 500). In some embodiments, [N2]-[N3] is present immediately subsequent to [N0]-[N1] and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [0116] In some embodiments, the peptide comprising the amino acid sequence comprising the formula [N2]-[N3] further comprises [N4], which comprises XG and XH. In some embodiments, XG of [N4] is T, P, or N. In some embodiments, XH of [N4] is A or D. In some embodiments, [N4] is or comprises TA, TD, PA, or NA. In some embodiments, [N4] is TA. In some embodiments, [N3]-[N4] is or comprises VQKTA (SEQ ID NO: 564), EQKTA (SEQ ID NO: 565), VKKTA (SEQ ID NO: 566), VQKPA (SEQ ID NO: 567), VHKTA (SEQ ID NO: 568), VQQTA (SEQ ID NO: 569), VQKNA (SEQ ID NO: 570), or LQKTA (SEQ ID NO: 571); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N3]-[N4] is VQKTA (SEQ ID NO: 564). In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is or comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNSSLSYQAEVVQKTA (SEQ ID NO: 2064), TNSSLSYTAEVVQKTA (SEQ ID NO: 2065), TNSSLYYPAEVVQKTA (SEQ ID NO: 2066), TNTSATYPAEVVQKTA (SEQ ID NO: 2067), TNSSLSYPAEVVQKTA (SEQ ID NO: 2068), TNSSLSYPAEEVQKTA (SEQ ID NO: 2069), TNSSLSYPAEVVQKTD (SEQ ID NO: 2070), TNSSLSYPAEVVQNTA (SEQ ID NO: 2071), TNSSLSYPAEVVQKNA (SEQ ID NO: 2072), TNSSLSYPAEVVQKPA (SEQ ID NO: 2073), TNNQTSTEAEVVQKTA (SEQ ID NO: 2074), TNKSATYPAEVVQKTA (SEQ ID NO: 2075), or TNSSLSYPADVVQKTA (SEQ ID NO: 2076); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). In some embodiments, [N2]-[N3] is present immediately
Attorney Docket No.14640.0092-00304 subsequent to [N0]-[N1], immediately precedes [N4], and replaces an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. [0117] In some embodiments, [N1] is present immediately subsequent to [N0]. In some embodiments, [N2] is present immediately subsequent to [N1]. In some embodiments, [N3] is present immediately subsequent to [N2]. In some embodiments, [N4] is present immediately subsequent to [N3]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [N2]-[N3]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [N1]-[N2]-[N3]-[N4]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [N0]-[N1]-[N2]-[N3]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [N0]-[N1]-[N2]-[N3]-[N4]. [0118] In some embodiments, a peptide described herein comprises an amino acid sequence having the formula [B]-[C], wherein [B] comprises X1, X2, and X3, and [C] comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). In some embodiments, X1 of [B] is Q, T, S, A, I, L, or H. In some embodiments, X1 of [B] is Q, T, S, A, or H. In some embodiments, X2 of [B] is S, G, or A. In some embodiments, X2 of [B] is S or G. In some embodiments, X3 of [B] is S, K, L, R, or A. In some embodiments, X3 of [B] is S, K, L, or R. In some embodiments, [B] comprises Q at X1. In some embodiments, [B] comprises S at X2. In some embodiments, [B] comprises S at X3. In some embodiments, [B] comprises QS, TS, SS, AG, IG, QA, AS, LG, HS, SK, SL, SR, GA, or GS. In some embodiments, [B] is or comprises QSS, TSL, SSS, QSR, QSK, AGA, IGS, QAS, ASS, LGS, or HSS. In some embodiments, the amino acid sequence of [B] is QSS. In some embodiments, [B]-[C] comprises SSYPAEVVQK (SEQ ID NO: 572), SKYPAEVVQK (SEQ ID NO: 573), SLYPAEVVQK (SEQ ID NO: 574), SRYPAEVVQK (SEQ ID NO: 575), GAYPAEVVQK (SEQ ID NO: 576), GSYPAEVVQK (SEQ ID NO: 580), or ASYPAEVVQK (SEQ ID NO: 582). In some embodiments, [B]-[C] is or comprises QSSYPAEVVQK (SEQ ID NO: 150), QSKYPAEVVQK (SEQ ID NO: 151), TSLYPAEVVQK (SEQ ID NO: 152), SSSYPAEVVQK (SEQ ID NO: 153), QSRYPAEVVQK (SEQ ID NO: 154), AGAYPAEVVQK (SEQ ID NO: 156), IGSYPAEVVQK (SEQ ID NO: 157), QASYPAEVVQK (SEQ ID NO: 158), ASSYPAEVVQK (SEQ ID NO: 159), LGSYPAEVVQK (SEQ ID NO: 160), or HSSYPAEVVQK (SEQ ID NO: 162); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [B]-[C] is QSSYPAEVVQK (SEQ ID NO: 150). [0119] In some embodiments, a peptide comprising the formula [B]-[C] further comprises [A], which comprises XA, XB, and XC. In some embodiments, XA of [A] is T, I, or N. In some embodiments, XB of [A] is N. In some embodiments, XC of [A] is N, T, S, or K. In some embodiments, [A] comprises TN, IN, NN, NT, NS, or NK. In some embodiments, [A] is or comprises TNN, TNT, INN, NNN, TNS, or TNK.
Attorney Docket No.14640.0092-00304 In some embodiments, [A] is TNN. In some embodiments, [A]-[B] is or comprises TNNQSS (SEQ ID NO: 183), TNNQSK (SEQ ID NO: 184), TNNTSL (SEQ ID NO: 185), TNNSSS (SEQ ID NO: 186), TNNQSR (SEQ ID NO: 187), TNNAGA (SEQ ID NO: 188), TNNIGS (SEQ ID NO: 189), TNNQAS (SEQ ID NO: 190), TNTASS (SEQ ID NO: 191), TNNLGS (SEQ ID NO: 192), TNNHSS (SEQ ID NO: 194), INNQSS (SEQ ID NO: 196), NNNQSR (SEQ ID NO: 198), TNSTSL (SEQ ID NO: 199), or TNKQAS (SEQ ID NO: 201); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences. In some embodiments, the amino acid sequence of [A]-[B] is TNNQSS (SEQ ID NO: 183). In some embodiments, [A]-[B]-[C] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500), TNNQSKYPAEVVQK (SEQ ID NO: 503), TNNTSLYPAEVVQK (SEQ ID NO: 506), TNNSSSYPAEVVQK (SEQ ID NO: 508), TNNQSRYPAEVVQK (SEQ ID NO: 510), TNNAGAYPAEVVQK (SEQ ID NO: 513), TNNIGSYPAEVVQK (SEQ ID NO: 514), TNNQASYPAEVVQK (SEQ ID NO: 517), TNTASSYPAEVVQK (SEQ ID NO: 520), TNNLGSYPAEVVQK (SEQ ID NO: 523), TNNHSSYPAEVVQK (SEQ ID NO: 525), INNQSSYPAEVVQK (SEQ ID NO: 543), NNNQSRYPAEVVQK (SEQ ID NO: 552), TNSTSLYPAEVVQK (SEQ ID NO: 556), or TNKQASYPAEVVQK (SEQ ID NO: 563); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences. In some embodiments, [A]-[B] is TNNQSS (SEQ ID NO: 183). In some embodiments, [A]-[B]-[C] is TNNQSSYPAEVVQK (SEQ ID NO: 500). [0120] In some embodiments, a peptide comprising the formula [B]-[C] further comprises [D], wherein [D] comprises X4 and X5. In some embodiments, X4 of [D] is T or N. In some embodiments, X5 of [D] is A. In some embodiments [D] is or comprises TA or PA. In some embodiments, the amino acid sequence of [D] is TA. In some embodiments, [C]-[D] is or comprises YPAEVVQKTA (SEQ ID NO: 584) or YPAEVVQKPA (SEQ ID NO: 586); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences. In some embodiments, the amino acid sequence of [C]-[D] is YPAEVVQKTA (SEQ ID NO: 584). In some embodiments, [A]-[B]-[C]-[D] is or comprises
Attorney Docket No.14640.0092-00304 TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNNQSKYPAEVVQKTA (SEQ ID NO: 1538), TNNTSLYPAEVVQKTA (SEQ ID NO: 1232), TNNSSSYPAEVVQKTA (SEQ ID NO: 1539), TNNQSRYPAEVVQKTA (SEQ ID NO: 1327), TNNAGAYPAEVVQKTA (SEQ ID NO: 1021), TNNIGSYPAEVVQKTA (SEQ ID NO: 1112), TNNQASYPAEVVQKTA (SEQ ID NO: 1194), TNTASSYPAEVVQKTA (SEQ ID NO: 1575), TNNLGSYPAEVVQKTA (SEQ ID NO: 1027), TNNHSSYPAEVVQKTA (SEQ ID NO: 1310), TNNSSSYPAEVVQKPA (SEQ ID NO: 1142), INNQSSYPAEVVQKTA (SEQ ID NO: 1024), NNNQSRYPAEVVQKTA (SEQ ID NO: 1601), TNSTSLYPAEVVQKTA (SEQ ID NO: 1605), or TNKQASYPAEVVQKTA (SEQ ID NO: 1587); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences. In some embodiments, [C]-[D] is YPAEVVQKTA (SEQ ID NO: 584). In some embodiments, [A]-[B]-[C]-[D] is TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). [0121] In some embodiments, a peptide described herein comprises an amino acid sequence having the formula [B]-[C], wherein [B] comprises X1, X2, and X3, and [C] comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). In some embodiments, X1 of [B] is Q or S. In some embodiments, X2 of [B] is S, L, or A. In some embodiments, X3 of [B] is S, Y, or T. In some embodiments, [B] comprises Q at X1. In some embodiments, [B] comprises S at X2. In some embodiments, [B] comprises S at X3. In some embodiments, [B] comprises QS, SL, SA, LY, AT, LS, or SS. In some embodiments, [B] is or comprises QSS, SLY, SAT, or SLS. In some embodiments, [B] is QSS. In some embodiments, [B]-[C] comprises SSYPAEVVQK (SEQ ID NO: 572), LYYPAEVVQK (SEQ ID NO: 702), ATYPAEVVQK (SEQ ID NO: 718), or LSYPAEVVQK (SEQ ID NO: 703). In some embodiments, [B]-[C] is or comprises QSSYPAEVVQK (SEQ ID NO: 150), SLYYPAEVVQK (SEQ ID NO: 639), SATYPAEVVQK (SEQ ID NO: 641), or SLSYPAEVVQK (SEQ ID NO: 642); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids, relative to any one of the aforesaid amino acid sequences. In some embodiments, [B]-[C] is QSSYPAEVVQK (SEQ ID NO: 150). [0122] In some embodiments, a peptide comprising the formula [B]-[C] further comprises [A], which comprises XA, XB, and XC. In some embodiments, XA of [A] is T. In some embodiments, XB of [A] is N. In some embodiments, XC of [A] is N, T, S, or K. In some embodiments, [A] comprises TN, NS, NT, NK, or NN. In some embodiments, [A] is or comprises TNN, TNS, TNT, or TNK. In some embodiments, the amino acid sequence of [A] is TNN. In some embodiments, [A]-[B] is or comprises
Attorney Docket No.14640.0092-00304 TNNQSS (SEQ ID NO: 183), TNSSLY (SEQ ID NO: 648), TNTSAT (SEQ ID NO: 649), TNSSLS (SEQ ID NO: 647), or TNKSAT (SEQ ID NO: 651); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, or 5 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [A]-[B] is TNNQSS (SEQ ID NO: 183). In some embodiments, [A]-[B]-[C] is or comprises TNNQSSYPAEVVQK (SEQ ID NO: 500), TNSSLYYPAEVVQK (SEQ ID NO: 655), TNTSATYPAEVVQK (SEQ ID NO: 656), TNSSLSYPAEVVQK (SEQ ID NO: 657), or TNKSATYPAEVVQK (SEQ ID NO: 663); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [A]-[B] is TNNQSS (SEQ ID NO: 183). In some embodiments, [A]-[B]-[C] is TNNQSSYPAEVVQK (SEQ ID NO: 500). [0123] In some embodiments, a peptide comprising the formula [B]-[C] further comprises [D], wherein [D] comprises X4 and X5. In some embodiments, X4 of [D] is T, N, or P. In some embodiments, X5 of [D] is A or D. In some embodiments, [D] is or comprises TA, TD, NA, or PA. In some embodiments, the amino acid sequence of [D] is TA. In some embodiments, [C]-[D] is or comprises YPAEVVQKTA (SEQ ID NO: 584), YPAEVVQKTD (SEQ ID NO: 719), YPAEVVQKNA (SEQ ID NO: 724), or YPAEVVQKPA (SEQ ID NO: 586); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [C]-[D] is YPAEVVQKTA (SEQ ID NO: 584). In some embodiments, [A]-[B]-[C]-[D] is or comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), TNSSLYYPAEVVQKTA (SEQ ID NO: 2066), TNTSATYPAEVVQKTA (SEQ ID NO: 2067), TNSSLSYPAEVVQKTA (SEQ ID NO: 2068), TNSSLSYPAEVVQKTD (SEQ ID NO: 2070), TNSSLSYPAEVVQKNA (SEQ ID NO: 2072), TNSSLSYPAEVVQKPA (SEQ ID NO: 2073), or TNKSATYPAEVVQKTA (SEQ ID NO: 2075); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any
Attorney Docket No.14640.0092-00304 one of the aforesaid amino acid sequences. In some embodiments, [A]-[B]-[C]-[D] is TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). [0124] In some embodiments, [B] is present immediately subsequent to [A]. In some embodiments, [C] is present immediately subsequent to [B]. In some embodiments, [D] is present immediately subsequent to [C]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [B]- [C]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [B]-[C]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [A]-[B]-[C]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [B]-[C]-[D]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [A]-[B]-[C]-[D]. [0125] In some embodiments, a peptide described herein comprises the formula [K1]-[K2], wherein [K1] comprises LSY or LYY, and [K2] comprises X1, X2, X3, and X4. In some embodiments, [K1] comprises LSY. In some embodiments, X1 of [K2] is Q, T or P. In some embodiments, X2 of [K2] is A, in some embodiments, X3 of [K2] is E or D. In some embodiments, X4 of [K2] is V or E. In some embodiments, [K2] comprises QA, TA, PA, EV, EE, DV, AE, or AD. In some embodiments, [K2] comprises QAE, TAE, PAE, PAD, AEV, AEE, or ADV. In some embodiments, [K2] is or comprises QAEV (SEQ ID NO: 15), TAEV (SEQ ID NO: 16), PAEV (SEQ ID NO: 17), PAEE (SEQ ID NO: 18), or PADV (SEQ ID NO: 19). In some embodiments, [K1]-[K2] comprises LSYQA (SEQ ID NO: 597), LSYTA (SEQ ID NO: 598), LYYPA (SEQ ID NO: 600), or LSYPA (SEQ ID NO: 603). In some embodiments, [K1]-[K2] comprises LSYQAE (SEQ ID NO: 607), LSYTAE (SEQ ID NO: 610), LYYPAE (SEQ ID NO: 611), LSYPAE (SEQ ID NO: 616), or LSYPAD (SEQ ID NO: 621). In some embodiments, [K1]-[K2] is or comprises LSYQAEV (SEQ ID NO: 667), LSYTAEV (SEQ ID NO: 668), LYYPAEV (SEQ ID NO: 669), LSYPAEV (SEQ ID NO: 671), LSYPAEE (SEQ ID NO: 673), or LSYPADV (SEQ ID NO: 674); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. [0126] In some embodiments, the peptide comprising the amino acid sequence comprising the formula of [K1]-[K2] further comprises [K0], which comprises TNNS (SEQ ID NO: 14). In some embodiments, [K0]-[K1] comprises TNSSLS (SEQ ID NO: 647) or TNSSLY (SEQ ID NO: 648). In some embodiments, [K0]-[K1] is or comprises TNSSLSY (SEQ ID NO: 676) or TNSSLYY (SEQ ID NO: 678); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some
Attorney Docket No.14640.0092-00304 embodiments, [K0]-[K1]-[K2] comprises TNSSLSYQA (SEQ ID NO: 679), TNSSLSYTA (SEQ ID NO: 681), TNSSLYYPA (SEQ ID NO: 682), or TNSSLSYPA (SEQ ID NO: 683). In some embodiments, [K0]-[K1]-[K2] comprises TNSSLSYQAE (SEQ ID NO: 684), TNSSLSYTAE (SEQ ID NO: 685), TNSSLYYPAE (SEQ ID NO: 686), TNSSLSYPAE (SEQ ID NO: 687), or TNSSLSYPAD (SEQ ID NO: 689). In some embodiments, [K0]-[K1]-[K2] is or comprises TNSSLSYQAEV (SEQ ID NO: 692), TNSSLSYTAEV (SEQ ID NO: 693), TNSSLYYPAEV (SEQ ID NO: 696), TNSSLSYPAEV (SEQ ID NO: 697), TNSSLSYPAEE (SEQ ID NO: 698), or TNSSLSYPADV (SEQ ID NO: 699); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, or 10 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. [0127] In some embodiments, peptide comprising the amino acid sequence comprising the formula of [K1]-[K2] further comprises [K3], wherein [K3] comprises XA, XB, and XC. In some embodiments, XA of [K3] is V. In some embodiments, XB of [K3] is Q. In some embodiments, XC of [K3] is K or N. In some embodiments, [K3] comprises VQ, QK, or QN. In some embodiments, [K3] is or comprises VQK or VQN. In some embodiments, K2]-[K3] is or comprises QAEVVQK (SEQ ID NO: 52), TAEVVQK (SEQ ID NO: 49), PAEVVQK (SEQ ID NO: 20), PAEEVQK (SEQ ID NO: 51) ,PAEVVQN (SEQ ID NO: 594), or PADVVQK (SEQ ID NO: 596); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, or 6 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [K1]-[K2]-[K3] is or comprises LSYQAEVVQK (SEQ ID NO: 700), LSYTAEVVQK (SEQ ID NO: 701), LYYPAEVVQK (SEQ ID NO: 702), LSYPAEVVQK (SEQ ID NO: 703), LSYPAEEVQK (SEQ ID NO: 704), LSYPAEVVQN (SEQ ID NO: 706), or LSYPADVVQK (SEQ ID NO: 708); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, or 9 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [K0]-[K1]-[K2]-[K3] is or comprises TNSSLSYQAEVVQK (SEQ ID NO: 652), TNSSLSYTAEVVQK (SEQ ID NO: 654), TNSSLYYPAEVVQK (SEQ ID NO: 655), TNSSLSYPAEVVQK (SEQ ID NO: 657), TNSSLSYPAEEVQK (SEQ ID NO: 658), TNSSLSYPAEVVQN (SEQ ID NO: 660), or TNSSLSYPADVVQK (SEQ ID NO: 665); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, or 13 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence
Attorney Docket No.14640.0092-00304 comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. [0128] In some embodiments, the peptide comprising the amino acid sequence comprising the formula of [K1]-[K2] further comprises [K4], wherein [K4] comprises XD and XE. In some embodiments, XD of [K4] is T, P, or N. In some embodiments, XE of [K4] is A or D. In some embodiments, [K4] is or comprises TA, TD, PA, or NA. In some embodiments, [K3]-[K4] is or comprises VQKTA (SEQ ID NO: 564), VQKTD (SEQ ID NO: 714), VQNTA (SEQ ID NO: 715), VQKNA (SEQ ID NO: 570), or VQKPA (SEQ ID NO: 567); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, or 4 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is or comprises TNSSLSYQAEVVQKTA (SEQ ID NO: 2064), TNSSLSYTAEVVQKTA (SEQ ID NO: 2065), TNSSLYYPAEVVQKTA (SEQ ID NO: 2066), TNSSLSYPAEVVQKTA (SEQ ID NO: 2068), TNSSLSYPAEEVQKTA (SEQ ID NO: 2069), TNSSLSYPAEVVQKTD (SEQ ID NO: 2070), TNSSLSYPAEVVQNTA (SEQ ID NO: 2071), TNSSLSYPAEVVQKNA (SEQ ID NO: 2072), TNSSLSYPAEVVQKPA (SEQ ID NO: 2073), or TNSSLSYPADVVQKTA (SEQ ID NO: 2076); an amino acid sequence comprising any portion of any one of the aforesaid amino acid sequences (e.g., any 2, 3, 4, 5, 6, 7, 8, 9, 10, 11, 12, 13, 14, or 15 amino acids, e.g., consecutive amino acids) thereof; an amino acid sequence comprising one, two, or three but no more than four modifications relative to any one of the aforesaid amino acid sequences; or an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to any one of the aforesaid amino acid sequences. [0129] In some embodiments, [K2] is present immediately subsequent to [K1]. In some embodiments, [K1] is present immediately subsequent to [K0]. In some embodiments, [K3] is present immediately subsequent to [K2]. In some embodiments, [K4] is present immediately subsequent to [K3]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [K1]-[K2]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [K0]-[K1]-[K2]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [K1]-[K2]-[K3]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [K0]-[K1]-[K2]-[K3]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [K1]-[K2]-[K3]-[K4]. In some embodiments, the peptide comprises, from N-terminus to C-terminus, [K0]-[K1]-[K2]-[K3]-[K4]. [0130] In some embodiments, a peptide described herein comprises an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of the sequences provided in Table 1, 2A, 2B, 14, or 23-28. In some embodiments, the peptide comprises an amino acid
Attorney Docket No.14640.0092-00304 sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583- 1587, 1590, 1591-1593, 1598-1608, and 1610-1624. In some embodiments, the peptide comprises an amino acid sequence comprising at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, or at least 15 consecutive amino acids from any one of SEQ ID NOs: 943 or 2064-2080. In some embodiments, the peptide comprises at least 3, at least 4, at least 5, at least 6, or at least 7 consecutive amino acids from SEQ ID NOs: 943 or any one of SEQ ID NOs: 946-966. [0131] In some embodiments, the at least 3 consecutive amino acids comprise YPA. In some embodiments, the at least 4 consecutive amino acids comprise YPAE (SEQ ID NO: 21). In some embodiments, the at least 5 consecutive amino acids comprise YPAEV (SEQ ID NO: 1). In some embodiments, the at least 6 consecutive amino acids comprise YPAEVV (SEQ ID NO: 725). In some embodiments, the at least 7 consecutive amino acids comprise YPAEVVQ (SEQ ID NO: 726). In some embodiments, the amino acid sequence comprises YPAEVVQK (SEQ ID NO: 943). In some embodiments, the amino acid sequence consists of YPAEVVQK (SEQ ID NO: 943). [0132] In some embodiments, a peptide described herein comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, 14, or 23-28. In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, 14, or 23-28. In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583- 1587, 1590, 1591-1593, 1598-1608, and 1610-1624. In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583- 1587, 1590, 1591-1593, 1598-1608, and 1610-1624. In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 943 and 2064-2080. In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080. [0133] In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or
Attorney Docket No.14640.0092-00304 three but no more than four different amino acids relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). [0134] In some embodiments, the peptide comprises an amino sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063. In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063. In some embodiments, the different amino acids of the amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 2024-2063 are present at one or more of the following positions: (i) position 1, wherein the different amino acid is T or L; (ii) position 2, wherein the different amino acid is N, L, K, A, T, or P; (iii) position 3, wherein the different amino acid is N, K, L, A, Y, or S; (iv) position 4, wherein the different amino acid is Q, L, T, S, F, Y, K, or A; (v) position 5, wherein the different amino acid is S, H, A, M, Q, T, V, or F; (vi) position 6, wherein the different amino acid is S, P, V, A, Q, L, T, N, or M; (vii) position 7, wherein the different amino acid is Y, H, S, V, A, L, or T; (viii) position 8, wherein the different amino acid is D, P, A, Q, F, L, S, H, or M; (ix) position 9, wherein the different amino acid is F, A, L, D, or Q; (x) position 10, wherein the different amino acid is T, E, I, or S; (xi) position 11, wherein the different amino acid is V, A, N, or S; (xii) position 12, wherein the different amino acid is
L, or P; (xiii) position 13, wherein the different amino acid is Q, E, or P; (xiv) position 14, wherein the different amino acid is K, N, S, or L; (xv) position 15, wherein the different amino acid is T, V, M, or L; and/or (xvi) position 16, wherein the different amino acid is A, G, or R. [0135] In some embodiments, the peptide comprises an amino sequence comprising one, two, or three but no more than four modifications relative to the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813- 1815, 1818, 1822, 1823, 1829-1837, 1842-2023, and 2087-2112. In some embodiments, the peptide comprises an amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842-2023, and 2087-2112. In some embodiments, the different amino acids of the amino acid sequence comprising one, two, or three but no more than four different amino acids relative to the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798- 1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842-2023, and 2087-2112, are present at one or more of the following positions: (i) position 1, wherein the different amino acid is T, G, N, S, E, L, Y, V, or I; (ii) position 2, wherein the different amino acid is D, N, K, E, V, G, R, L, H, F, P, T, A, S, I, or Y; (iii) position 3, wherein the different amino acid is Y, N, K, T, W, Q, M, V, C, A, L, F, H, G, R, S, or P; (iv) position 4, wherein the different amino acid is H, Q, P, E, R, K, A, S, V, L, T, D, I, G, M, or N; (v) position 5, wherein the different amino acid is R, S, K, N, H, G, W, A, P, V, Q, Y, L, or F; (vi) position 6, wherein the different amino acid is G, S, F, R, W, H, I, C, M, A, Y, K, N, Q, V, P, E,
Attorney Docket No.14640.0092-00304 D, T, or L; (vii) position 7, wherein the different amino acid is D, Y, S, I, H, F, P, K, R, G, L, Q, A, M, T, N, V, W, C, or E; (viii) position 8, wherein the different amino acid is P, L, Q, T, W, V, G, K, I, Y, N, H, R, D, S, M, A, F, or E; (ix) position 9, wherein the different amino acid is A, R, T, Q, S, M, L, E, K, V, G, D, N, H, F, P, or I; (x) position 10, wherein the different amino acid is K, E, Q, H, V, G, R, S, P, I, N, M, A, L, D, or T; (xi) position 11, wherein the different amino acid is V, A, E, N, R, L, M, T, Q, S, K, C, G, D, Y, P, H, F, or I; (xii) position 12, wherein the different amino acid is V, P, L, S, T, N, A, G, K, R, I, H, E, Q, or M; (xiii) position 13, wherein the different amino acid is Q, K, N, A, H, R, T, V, E, I, P, G, S, or L; (xiv) position 14, wherein the different amino acid is K, E, I, Y, Q, R, G, D, L, N, or S; (xv) position 15, wherein the different amino acid is S, T, N, Q, I, P, E, G, K, M, or H; and/or (xvi) position 16, wherein the different amino acid is A, D, L, Y, Q, or T. [0136] In some embodiments, the peptide comprises the amino acid sequence of any one of the sequences provided in Table 1, 2A, 2B, 14, or 23-28. In some embodiments, the peptide comprises the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583- 1587, 1590, 1591-1593, 1598-1608, and 1610-1624. In some embodiments, the peptide comprises the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080. In some embodiments, the peptide comprises the amino acid sequence of any one of SEQ ID NOs: 2024-2063. In some embodiments, the peptide comprises the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842- 2023, and 2087-2112. In some embodiments, the peptide comprises the amino acid sequence of SEQ ID NO: 943. [0137] In some embodiments, the peptide comprises an amino acid sequence encoded by a nucleotide sequence described herein, e.g., a nucleotide sequence of Table 2A. In some embodiments, the peptide comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 944. In some embodiments, the peptide comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 944. In some embodiments, the peptide comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 944, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. [0138] In some embodiments, the nucleotide sequence encoding a peptide described herein comprises a nucleotide sequence described herein, e.g., as described in Table 2A. In some embodiments, the nucleotide sequence encoding a peptide described herein is codon optimized. In some embodiments,
Attorney Docket No.14640.0092-00304 the nucleotide sequence encoding a peptide described herein is isolated. In some embodiments, the nucleotide sequence encoding a peptide described herein is recombinant. [0139] In some embodiments, the nucleotide sequence encoding a peptide described herein comprises the nucleotide sequence of SEQ ID NO: 944, or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 944. In some embodiments, the nucleotide sequence encoding a peptide described herein comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten different nucleotides relative to the nucleotide sequence of SEQ ID NO: 944. In some embodiments, the nucleic acid encoding a peptide described herein comprises a nucleotide sequence comprising the nucleotide sequence of SEQ ID NO: 944, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. [0140] The present disclosure also provides a nucleic acid or polynucleotide encoding any of the peptides described herein and AAV capsid variants, AAV particles, vectors, and cells comprising the same. AAV Capsid Variants [0141] In some embodiments, an AAV capsid variant described herein (e.g., an AAV5 capsid variant) comprises an amino acid other than T corresponding to position 577 (e.g., Y, N, or C) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises Y corresponding to position 577 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises N corresponding to position 577 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises C corresponding to position 577 of the amino acid sequence of SEQ ID NO: 138. [0142] In some embodiments, the AAV capsid variant (e.g., an AAV5 capsid variant) comprises more than one amino acid that replaces the threonine (T) at position 577, numbered according to SEQ ID NO: 138 (e.g., that replaces a T corresponding to T577 of the amino acid sequence of SEQ ID NO: 138). In some embodiments, an insert of two, three, four, five, six, seven, eight, nine, or ten amino acids replaces the T at position 577, numbered according to SEQ ID NO: 138. In some embodiments, an insert of eight amino acids replaces the T at position 577, numbered according to SEQ ID NO: 138. [0143] In some embodiments, an AAV particle described herein comprises an AAV capsid variant comprising a peptide or an amino acid sequence described herein. In some embodiments, the AAV capsid variant comprises a peptide disclosed herein. In some embodiments, the peptide is present in VP1, VP2, and/or VP3 proteins of the AAV capsid variant. In some embodiments, the peptide is present in VP1, VP2, and VP3 proteins of the AAV capsid variant. [0144] In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of any one of SEQ ID NOs: 943, 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300,
Attorney Docket No.14640.0092-00304 1310, 1327, 1331, 1342, 1419, 1453, 1533, 1538, 1539, 1575, 1578, 1583- 1587, 1590, 1591-1593, 1598- 1608, and 1610-1624. In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 2064-2080. In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of any one of SEQ ID NOs: 2024-2063. In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of any one of SEQ ID NOs: 1632-1733, 1735-1737, 1739, 1741, 1744-1749, 1751-1796, 1798-1800, 1802-1807, 1810, 1813-1815, 1818, 1822, 1823, 1829-1837, 1842- 2023, and 2087-2112. In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of SEQ ID NO: 943. In some embodiments, the peptide is present in loop VIII. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 570 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the peptide replaces amino acids corresponding to positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 570, and the peptide replaces amino acids corresponding to positions 571-579 (e.g., T571, N572, N573, Q574, S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 571 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the peptide replaces amino acids corresponding to positions 572-579 (e.g., N572, N573, Q574, S575, S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 571, and the peptide replaces amino acids corresponding to positions 572-579 (e.g., N572, N573, Q574, S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 572 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the peptide replaces amino acids corresponding to positions 573-579 (e.g., N573, Q574, S575, S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 572, and the peptide replaces amino acids corresponding to positions 573-579 (e.g., N573, Q574, S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 573 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the peptide replaces amino acids corresponding to positions 574-579 (e.g., Q574, S575, S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 573, and the peptide replaces amino acids corresponding to positions 574-579 (e.g., Q574, S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 574 of the amino acid sequence of SEQ ID NO: 138. In some embodiments,
Attorney Docket No.14640.0092-00304 the peptide replaces amino acids corresponding to positions 575-579 (e.g., S575, S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 574, and the peptide replaces amino acids corresponding to positions 575-579 (e.g., S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 575 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the peptide replaces amino acids corresponding to positions 576-579 (e.g., S576, T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 575, and the peptide replaces amino acids corresponding to positions 576-579 (e.g., S575, S576, T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the peptide replaces an amino acid corresponding to position 577 (e.g., T577) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the peptide replaces amino acids corresponding to positions 577-579 (e.g., T577, T578, and T579) of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 576, and the peptide replaces amino acids corresponding to position 577 (e.g., T577), of the amino acid sequence of SEQ ID NO 138. In some embodiments, the peptide is present immediately subsequent to an amino acid corresponding to position 576, and the peptide replaces amino acids corresponding to positions 577-579 (e.g., T577, T578, and T579), of the amino acid sequence of SEQ ID NO 138. [0145] In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 946-966, wherein the peptide replaces an amino acid corresponding to position 577 (e.g., T577) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 946-966, wherein the peptide is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises a peptide comprising the amino acid sequence of SEQ ID NOs: 943 or any one of SEQ ID NOs: 946-966, wherein the peptide is present immediately subsequent to an amino acid corresponding to position 576, and wherein the peptide replaces an amino acid corresponding to position 577 (e.g., T577), of the amino acid sequence of SEQ ID NO: 138. [0146] In some embodiments, the AAV capsid variant (e.g., an AAV capsid variant described herein), comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 944 or a nucleotide sequence having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity thereto. In some embodiments, the AAV capsid variant described herein comprises an amino acid sequence encoded by
Attorney Docket No.14640.0092-00304 the nucleotide sequence of SEQ ID NO: 944 or a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 944. In some embodiments, the AAV capsid variant comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten, different nucleotides relative to the nucleotide sequence of SEQ ID NO: 944. [0147] In some embodiments, the nucleotide sequence encoding the AAV capsid variant comprises the nucleotide sequence of SEQ ID NO: 944, or a nucleotide sequence substantially identical (e.g., having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 92%, at least 95%, at least 97%, at least 98%, or at least 99% sequence identity) thereto. In some embodiments, the nucleic acid sequence encoding the AAV capsid variant comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO: 944. In some embodiments, the nucleotide sequence encoding an AAV capsid variant described herein comprises a nucleotide sequence comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven, but no more than ten, different nucleotides relative to the nucleotide sequence of SEQ ID NO: 944. [0148] In some embodiments, an AAV capsid variant described herein comprises a peptide comprising the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the peptide is present in loop VIII. In some embodiments, the peptide is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant. In some embodiments, the peptide is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant. [0149] In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the amino acid sequence is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) replaces an amino acid corresponding to position 577 (e.g., T577) of the amino acid sequence of SEQ ID NO: 138. In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943), wherein the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 138, and wherein the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) replaces an amino acid corresponding to position 577 (e.g., T577) of the amino acid sequence of SEQ ID NO: 138. [0150] In some embodiments, an AAV capsid variant described herein comprises the amino acid Y at a position corresponding to amino acid 577 of the amino acid sequence of SEQ ID NO: 138, and further comprises the amino acid sequence of PAEVVQK (SEQ ID NO: 20), which is present immediately
Attorney Docket No.14640.0092-00304 subsequent to a position corresponding to amino acid 577 of the amino acid sequence of SEQ ID NO: 138. [0151] In some embodiments, an AAV capsid variant described herein comprises the amino acid Y at a position corresponding to amino acid 577 of the amino acid sequence of SEQ ID NO: 982, and the amino acid sequence of PAEVVQK (SEQ ID NO: 20) at positions corresponding to amino acids 578- 584, of the amino acid sequence of SEQ ID NO: 982. [0152] In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), which is present in loop VIII. In some embodiments, the AAV capsid variant comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533), which is present immediately subsequent to an amino acid corresponding to position 570 of the amino acid sequence of SEQ ID NO: 138, wherein YPAEVVQK (SEQ ID NO: 943) replaces an amino acid corresponding to position 577 (e.g., replaces T577) of the amino acid sequence of SEQ ID NO: 138. [0153] In some embodiments, the AAV capsid variant comprises: (a) a VP1 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 982; (b) a VP2 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 738; and/or (c) a VP3 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises a peptide comprising the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) or a peptide comprising one, two, or three—but no more than three—substitutions relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). [0154] In some embodiments, the AAV capsid variant comprises: (a) a VP1 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to SEQ ID NO: 982; (b) a VP2 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 738; and/or (c) a VP3 protein comprising an amino acid sequence that is at least 95% identical, e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical, to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises a peptide comprising the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). [0155] In some embodiments, the AAV capsid variant comprises: (a) a VP1 protein comprising an amino acid sequence that is at least 98% identical, e.g., at least 98%, at least 99%, or 100% identical, to SEQ ID NO: 982; (b) a VP2 protein comprising an amino acid sequence that is at least 98% identical, e.g., at least 98%, at least 99%, or 100% identical, to SEQ ID NO: 738; and/or (c) a VP3 protein comprising an amino acid sequence that is at least 98% identical, e.g., at least 98%, at least 99%, or 100% identical, to SEQ ID NO: 739, wherein the AAV capsid variant comprises a peptide comprising the
Attorney Docket No.14640.0092-00304 amino acid sequence of YPAEVVQK (SEQ ID NO: 943) or a peptide comprising one, two, or three—but no more than three—substitutions relative to the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). [0156] In some embodiments, the AAV capsid variant comprises: (a) a VP1 protein comprising the amino acid sequence of SEQ ID NO: 982; (b) a VP2 protein comprising the amino acid sequence of SEQ ID NO: 738; and/or (c) a VP3 protein comprising the amino acid sequence of SEQ ID NO: 739. In some embodiments, the AAV capsid variant is encoded by the nucleotide sequence of SEQ ID NO: 984 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 984. [0157] In some embodiments, the present disclosure provides an AAV particle comprising an AAV capsid variant and a viral genome, wherein the viral genome comprises a frataxin (FXN)-encoding sequence and the AAV capsid variant comprises an amino acid sequence having the formula [N2]-[N3], wherein: (i) [N2] comprises X1, X2, X3, X4, and X5, wherein: (a) X1 is Y, N, or C; (b) X2 is P, K, T, or Q; (c) X3 is A or P; (d) X4 is E, S, or A; and (e) X5 is V, L, or E; and (ii) [N3] comprises the amino acid sequence VQK, EQK, VKK, VHK, VQQ, or LQK; and wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 739, or an amino acid sequence at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the amino acid sequence [N2]-[N3] is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant. In some embodiments, the amino acid sequence [N2]-[N3] is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant. [0158] In some embodiments, [N2]-[N3] is present in loop VIII of the AAV capsid variant, wherein loop VIII comprises positions 571-599 numbered according to the amino acid sequence of SEQ ID NO: 982. In some embodiments, [N2]-[N3] is present in loop VIII of the AAV capsid variant, wherein loop VIII is present at amino acids comprising those corresponding to positions 571-599 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, [N2]-[N3] is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, the capsid variant is an AAV5 variant comprising [N2]-[N3] in place of an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, [N2] comprises the amino acid sequence YP, YPA, YPAE (SEQ ID NO: 21), or YPAEV (SEQ ID NO: 1). In some embodiments, the amino acid sequence is VQK. In some embodiments, [N2]-[N3] comprises the amino acid sequence AEVVQK (SEQ ID NO: 36) or PAEVVQK (SEQ ID NO: 20). In some embodiments, [N2]-[N3] comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). In some embodiments, [N2]-[N3] is present in loop VIII of a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant. In some embodiments, [N2]-[N3] is present in loop VIII of the VP1, VP2, and VP3 proteins of the AAV capsid variant. [0159] In some embodiments, the AAV capsid variant further comprises [N1] comprising XD, XE, and XF, wherein: (a) XD is Q, T, S, A, I, L, or H; (b) XE is S, G, A, or R; and (c) XF is S, K, L, R, A, or T;
Attorney Docket No.14640.0092-00304 and wherein [N1] immediately precedes [N2]-[N3]. In some embodiments, [N1] comprises the amino acid sequence QS, SS, or QSS. In some embodiments, [N1]-[N2] comprises the amino acid sequence QSSYPAEV (SEQ ID NO: 96). In some embodiments, [N1]-[N2]-[N3] comprises the amino acid sequence SSYPAEVVQ (SEQ ID NO: 121) or QSSYPAEVVQK (SEQ ID NO: 150). In some embodiments, [N1]-[N2]-[N3] is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant. In some embodiments, [N1]-[N2]-[N3] is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant. [0160] In some embodiments, the AAV capsid variant further comprises [N0] comprising XA, XB, and XC, wherein: (a) XA is T, I, or N; (b) XB is N; and (c) XC is N, T, S, or K; and wherein [N0] immediately precedes [N1]. In some embodiments, [N0] comprises the amino acid sequence TN, NN, or TNN. In some embodiments, [N0]-[N1] comprises the amino acid sequence TNNQSS (SEQ ID NO: 183). In some embodiments, [N0]-[N1]-[N2]-[N3] comprises the amino acid sequence TNNQSSYPAEVVQK (SEQ ID NO: 500). In some embodiments, [N0]-[N1]-[N2]-[N3] is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant. In some embodiments, [N0]-[N1]-[N2]-[N3] is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant. [0161] In some embodiments, the AAV capsid variant further comprises [N4] comprising XG and XH, wherein: (a) XG is T, P, or N; and (b) XH is A; and wherein [N4] is present immediately subsequent to [N3]. In some embodiments, [N4] comprises the amino acid sequence TA. In some embodiments, [N4] is present in a VP1 protein, a VP2 protein, and/or a VP3 protein of the AAV capsid variant. In some embodiments, [N4] is present in the VP1, VP2, and VP3 proteins of the AAV capsid variant. In some embodiments, the AAV capsid variant comprises [N2]-[N3]-[N4], wherein each of [N2], [N3] and [N4] are as described herein. In some embodiments, the AAV capsid variant comprises [N1]-[N2]-[N3]-[N4], wherein each of [N1], [N2], [N3] and [N4] are as described herein. In some embodiments, the AAV capsid variant comprises [N0]-[N1]-[N2]-[N3]-[N4], wherein each of [N0], [N1], [N2], [N3] and [N4] are as described herein. [0162] In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] comprises TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). In some embodiments, the AAV capsid variant is an AAV5 capsid variant that comprises SEQ ID NO: 1533. In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is present in loop VIII, wherein loop VIII comprises positions 571-599 numbered according to the amino acid sequence of SEQ ID NO: 982. In some embodiments, [N0]-[N1]-[N2]-[N3]-[N4] is present in loop VIII, wherein loop VIII is present at amino acids comprising those corresponding to positions 571-599 of the amino acid sequence of SEQ ID NO: 982. [0163] In some embodiments, [N0] is present at amino acids corresponding to positions 571-573 of the amino acid sequence of SEQ ID NO: 982; [N1] is present at amino acids corresponding to positions 574-576 of the amino acid sequence of SEQ ID NO: 982; [N2] is present at amino acids corresponding to positions 577- 581 of the amino acid sequence of SEQ ID NO: 982; [N3] is present at amino acids corresponding to positions 582-584 of the amino acid sequence of SEQ ID NO: 982; and [N4] is present
Attorney Docket No.14640.0092-00304 at amino acids corresponding to positions 585-586 of the amino acid sequence of SEQ ID NO: 982 of SEQ ID NO: 982. [0164] In some embodiments, the present disclosure provides an AAV particle comprising: (i) a viral genome comprising a frataxin (FXN)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% identical to SEQ ID NO: 739, wherein the AAV capsid variant comprises at least 4, at least 5, at least 6, at least 7, or all 8 consecutive amino acids from the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). In some embodiments, the at least 4 consecutive amino acids comprise YPAE (SEQ ID NO: 21). In some embodiments, YPAE (SEQ ID NO: 21) is present at amino acids corresponding to positions 577-580 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, the at least 5 consecutive amino acids comprise YPAEV (SEQ ID NO: 1). In some embodiments, YPAEV (SEQ ID NO: 1) is present at amino acids corresponding to positions 577-581 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, the at least 6 consecutive amino acids comprise YPAEVV (SEQ ID NO: 725). In some embodiments, YPAEVV (SEQ ID NO: 725) is present at amino acids corresponding to positions 577-582 of the amino acid sequence of SEQ ID NO: 982. In some embodiments, the at least 7 consecutive amino acids comprise YPAEVVQ (SEQ ID NO: 726). In some embodiments, YPAEVVQ (SEQ ID NO: 726) is present at amino acids corresponding to positions 577-583 of the amino acid sequence of SEQ ID NO: 982. [0165] In some embodiments, the AAV capsid variant comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) present at amino acids corresponding to positions 577-584 of the amino acid sequence of SEQ ID NO: 982. [0166] In some embodiments, the present disclosure provides an AAV particle comprising: (i) a viral genome comprising a frataxin (FXN)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% identical (e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identical) to the amino acid sequence of SEQ ID NO: 982, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943) at amino acids corresponding to positions 577-584 of the amino acid sequence of SEQ ID NO: 982. [0167] In some embodiments, the AAV capsid variant comprises (i) a VP1 protein comprising an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity) to SEQ ID NO: 982, wherein the VP1 protein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943); (ii) a VP2 protein comprising an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity) to SEQ ID NO: 738, wherein the VP2 protein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943); and/or (iii) a VP3 protein comprising an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% identity) to SEQ ID NO: 739, wherein the VP3 protein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943).
Attorney Docket No.14640.0092-00304 [0168] In some embodiments, the AAV capsid variant comprises (i) a VP1 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO: 982, wherein the VP1 protein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943); (ii) a VP2 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO: 738, wherein the VP2 protein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943); and/or (iii) a VP3 protein comprising an amino acid sequence having at least 99% identity to SEQ ID NO: 739, wherein the VP3 protein comprises the amino acid sequence of YPAEVVQK (SEQ ID NO: 943). [0169] In some embodiments, the AAV capsid variant comprises (i) a VP1 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 982; (ii) a VP2 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 738; and/or (iii) a VP3 protein comprising or consisting of the amino acid sequence of SEQ ID NO: 739. [0170] In some embodiments, the AAV capsid variant further comprises one, two, three, or all of (i) an amino acid other than Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138 (e.g., T, S, A, I, L, or H at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138), (ii) an amino acid other than S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138 (e.g., G, A, L, T, or R at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138), and/or (iii) an amino acid other than S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138 (e.g., K, L, R, A, Y, or T at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138). In some embodiments, the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a T present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an L present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an S present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an R present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises Q present at a position corresponding to amino acid 574 ofthe amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of
Attorney Docket No.14640.0092-00304 the amino acid sequence of SEQ ID NO: 138, and/or a K present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an A present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, a G present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an A present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an I present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, a G present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an A present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an A present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an L present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, a G present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or a T present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an H present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an L present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an S present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of to the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an R present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an S present at a position
Attorney Docket No.14640.0092-00304 corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an L present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an S present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an L present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or a Y present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an S present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, an A present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or a T present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138, a T present at a position corresponding to amino acid 575 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 576 of the amino acid sequence of SEQ ID NO: 138. [0171] In some embodiments, the AAV capsid variant comprises an amino acid other than Q present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises S present at a position corresponding to amino acid 574 of the amino acid sequence of SEQ ID NO: 138. [0172] In some embodiments, the AAV capsid variant further comprises an amino acid other than T present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138 and/or an amino acid other than N present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises I or N present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138 and/or T, S, or K present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an R present at a position corresponding to amino acid 456 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a T present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid sequence of SEQ ID NO: 138, and/or an N present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a T present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid sequence of SEQ ID NO: 138, and/or a T present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an I present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid
Attorney Docket No.14640.0092-00304 sequence of SEQ ID NO: 138, and/or an N present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a T present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid sequence of SEQ ID NO: 138, and/or an S present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an N present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid sequence of SEQ ID NO: 138, and/or an N present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a T present at a position corresponding to amino acid 571 of the amino acid sequence of SEQ ID NO: 138, an N present at a position corresponding to amino acid 572 of the amino acid sequence of SEQ ID NO: 138, and/or a K present at a position corresponding to amino acid 573 of the amino acid sequence of SEQ ID NO: 138. [0173] In some embodiments, the AAV capsid variant further comprises an amino acid other than T present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a P or N present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an amino acid other than T present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or an amino acid other than A present at a position corresponding to amino acid 589 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a P or N present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or a D present at a position corresponding to amino acid 589 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a T present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or an A present at a position corresponding to amino acid 579 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a P present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or an A present at a position corresponding to amino acid 579 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an N present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or an A present at a position corresponding to amino acid 579 of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises a T present at a position corresponding to amino acid 578 of the amino acid sequence of SEQ ID NO: 138 and/or a D present at a position corresponding to amino acid 579 of the amino acid sequence of SEQ ID NO: 138. [0174] In some embodiments, the AAV capsid variant further comprises an amino acid other than T present at a position corresponding to amino acid 577 of the amino acid sequence of SEQ ID NO: 138. In
Attorney Docket No.14640.0092-00304 some embodiments, the AAV capsid variant comprises a Y present at a position corresponding to amino acid 577 of the amino acid sequence of SEQ ID NO: 138. [0175] In some embodiments, the AAV capsid variant further comprises a modification in loop I, II, IV, and/or VI. In some embodiments, loop I, II, IV, VI, and VIII can be identified as described in Govindasamy et al. Structurally Mapping the Diverse Phenotype of Adeno-Associated Virus Serotype 4. Journal of Virology.2006 Dec.80(23):11556-11570; and Govindasamy et al. Structural Insights into Adeno-Associated Virus Serotype 5. Journal of Virology.2013 Oct.87(20):11187-11199; the contents of which are each hereby incorporated by reference in their entirety. [0176] In some embodiments, additional modifications can be introduced into an AAV capsid variant described herein at positions determined using a structural map of wild-type AAV5, e.g., a structural map described and generated by Govindasamy et al. et al. Structural Insights into Adeno-Associated Virus Serotype 5. Journal of Virology.2013 Oct.87(20):11187-11199 (the contents of which are hereby incorporated herein by reference in their entirety) or Walters et al. “Structure of Adeno-Associated Virus Serotype 5,” Journal of Virology, 2004, 78(7):3361-3371 (the contents of which are hereby incorporated by reference in their entirety). [0177] In some embodiments, an AAV capsid variant described herein comprises a modification as described in Jose et al. “High-Resolution Structural Characterization of a New Adenoassociated Virus Serotype 5 Antibody Epitope toward Engineering Antibody-Resistant Recombinant Gene Delivery Vectors,” Journal of Virology, 2020, 93(1): e01394-18; Qian et al. “Directed Evolution of AAV Serotype 5 for Increased Hepatocyte Transduction and Retained Low Humoral Seroreactivity,” Molecular Therapy: Methods and Clinical Development, 2021, 20:122-132; Afione et al. “Identification and Mutagenesis of the Adeno-Associated Virus 5 Sialic Acid Binding Region,” Journal of Virology, 2015, 89(3):1660-1672; and/or Wang et al. “Directed evolution of adeno-associated virus 5 capsid enables specific liver tropism,” Mol Ther Nucleic Acids, 2022, 28:293-306; the contents of each of which are hereby incorporated by reference in their entirety. [0178] In some embodiments, the AAV capsid variant, further comprises an amino acid sequence comprising at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, of the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant further comprises an amino acid sequence comprising at least three, but not more than 30, not more than 20, or not more than 10 different amino acids relative to the amino acid sequence of SEQ ID NO: 138. In some embodiments, the AAV capsid variant is a variant of SEQ ID NO: 138, wherein the AAV capsid variant comprises an amino acid sequence having at least 70% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to SEQ ID NO: 138. [0179] In some embodiments, an AAV capsid variant of the present disclosure comprises the amino acid sequence of TTN-002, TTN-003, TTN-004, TTN-005, or TTN-006, e.g., as described in Table 3 or
Attorney Docket No.14640.0092-00304 4A. In some embodiments, an AAV capsid variant of the present disclosure comprises the amino acid sequence of TTN-002, e.g., as described in Table 3 or 4A. [0180] In some embodiments, the AAV capsid variant comprises a VP1, VP2, and/or VP3 protein comprising an amino acid sequence of TTN-002, TTN-003, TTN-004, TTN-005, or TTN-006, e.g., as described in Table 3 or 4A. In some embodiments, the AAV capsid variant comprises a VP1, VP2, and/or VP3 protein comprising an amino acid sequence of TTN-002, e.g., as described in Table 3 or 4A. [0181] In some embodiments, the AAV capsid variant comprises an amino acid sequence encoded by the nucleotide sequence of TTN-002, e.g., as described in Table 3 or 4B. [0182] In some embodiments, a polynucleotide or nucleic acid encoding an AAV capsid variantof the present disclosure comprises the nucleotide sequence of TTN-002, e.g., as described in Table 3 or 4B. Table 3. Exemplary full-length capsid sequences
Table 4A. Exemplary full-length capsid amino acid sequences
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304 Table 4B. Exemplary full length capsid nucleic acid sequences
[0183] In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 90% sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 95% sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 96% sequence identity thereto. In
Attorney Docket No.14640.0092-00304 some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 97% sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 98% sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence with at least 99% sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of SEQ ID NO: 982. In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 different amino acids, relative to the amino acid sequence of SEQ ID NO: 982. [0184] In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of any one of SEQ ID NOs: 740-743, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the amino acid sequence of any one of SEQ ID NOs: 740-743. In some embodiments, the AAV capsid variant comprises an amino acid sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 different amino acids, relative to the amino acid sequence of any one of SEQ ID NOs: 740-743. [0185] In some embodiments, an AAV capsid variant described herein comprises an amino acid sequence encoded by the nucleotide sequence of SEQ ID NO: 984, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, an AAV capsid variant described herein comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 different nucleotides, relative to the amino acid sequence of SEQ ID NO: 984. In some embodiments, an AAV capsid variant described herein comprises an amino acid sequence encoded by a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the nucleotide sequence of SEQ ID NO: 984. [0186] In some embodiments, the nucleotide sequence encoding an AAV capsid variant, described herein comprises the nucleotide sequence of SEQ ID NO: 984, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the
Attorney Docket No.14640.0092-00304 nucleotide sequence encoding an AAV capsid variant described herein comprises the nucleotide sequence of SEQ ID NO: 984, or a nucleotide sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the nucleotide sequence encoding an AAV capsid variant described herein, comprises a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 modifications, relative to the nucleotide sequence of SEQ ID NO: 984. In some embodiments, the nucleotide sequence encoding an AAV capsid variant described herein, comprises a nucleotide sequence comprising at least one, at least two, or at least three modifications, but not more than 30, not more than 20, or not more than 10 different nucleotides, relative to the amino acid sequence of SEQ ID NO: 984. In some embodiments, the nucleic acid sequence encoding an AAV capsid variant described herein is codon optimized. [0187] In some embodiments, an AAV capsid variant described herein comprises a VP1, VP2, VP3 protein, or a combination thereof. In some embodiments, an AAV capsid variant comprises the amino acid sequence corresponding to positions 137-731, e.g., a VP2, of the amino acid sequence of SEQ ID NO: 982, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid protein comprises the amino acid sequence corresponding to positions 193-731, e.g., a VP3, of the amino acid sequence of SEQ ID NO: 982, or a sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence corresponding to positions 1- 731, e.g., a VP1, of the amino acid sequence of SEQ ID NO: 982, or an amino acid sequence with at least 70% (e.g., at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity thereto. [0188] In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 739, or an amino acid sequence at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 738, or an amino acid sequence at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. In some embodiments, the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982, or an amino acid sequence at least 95% (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. [0189] In some embodiments, an AAV capsid variant described herein comprises the amino acid sequence of SEQ ID NO: 739 (e.g., VP3). In some embodiments, the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 738 (e.g., VP2). In some embodiments, the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982 (e.g., VP1). In some embodiments, the AAV capsid variant has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord
Attorney Docket No.14640.0092-00304 cell, or spinal cord tissue, relative to the tropism of an AAV capsid that does not comprise the amino acid sequence of YPAEVVQK (SEQ ID NO: 943) present at positions corresponding to amino acids 577-584 of the amino acid sequence of SEQ ID NO: 982. [0190] In some embodiments, an AAV capsid variant, described herein has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. [0191] In some embodiments, an AAV capsid variant described herein has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982. [0192] In some embodiments, an AAV capsid variant described herein has an increased tropism for a CNS cell or tissue, e.g., a brain cell, brain tissue, spinal cord cell, or spinal cord tissue, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139. [0193] In some embodiments, an AAV capsid variant described herein transduces a brain region, e.g., a temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, and/or cerebellum. In some embodiments, the level of transduction is at least 0.5, at least 2.2, at least 2.4, at least 2.5, at least 2.6, at least 2.7, at least 3.0, at least 3.2, at least 3.5, at least 3.7, at least 4.0, at least 4.2, at least 4.5, at least 4.7, at least 4.9, at least 5, at least 10, at least 15, at least 20, at least 25, at least 30, or at least 35-fold greater as compared to the level of transduction of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139. [0194] In some embodiments, an AAV capsid variant described herein is enriched at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 12, at least 15, at least 17, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, or at least 65-fold in the brain compared to enrichment in the brain of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. In some embodiments, an AAV capsid variant described herein is enriched at least 10, at least 12, at least 15, at least 17, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 61, at least 62, at least 63, at least 64, or at least 65-fold in the brain compared to enrichment in the brain of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. [0195] In some embodiments, an AAV capsid variant described herein is enriched in the brain of at least two to at least three species, e.g., a non-human primate and rodent (e.g., mouse and/or rat) species, compared to enrichment in the brain of those species of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. In some embodiments, an AAV capsid variant described herein is enriched at least 2, at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 12, at least 15, at least 17, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 70, at least 80, at least 90, or at least 100-fold in the brain of at least two to at least three species, e.g., a non-human primate and rodent (e.g., mouse and/or rat) species, compared to enrichment in the brain of those species of an AAV capsid comprising the amino acid
Attorney Docket No.14640.0092-00304 sequence of SEQ ID NO: 138. In some embodiments, the at least two to at least three species are Macaca fascicularis, Chlorocebus sabaeus, Callithrix jacchus, rat and/or mouse (e.g., BALB/c mice). [0196] In some embodiments, an AAV capsid variant described herein is enriched about 5, at least 10, at least 15, at least 20, at least 25, at least 30, at least 35, at least 40, at least 45, at least 50, at least 55, at least 60, at least 70, at least 80, at least 90, or at least 100, at least 125, at least 150, at least 175, at least 200, or at least 225-fold in the brain compared to enrichment in the brain of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 982. [0197] In some embodiments, an AAV capsid variant described herein delivers an increased number and/or level of viral genomes to a brain region. In some embodiments, the number and/or level of viral genomes is increased by at least 1.5, at least 2.2, at least 2.4, at least 2.5, at least 2.6, at least 2.7, at least 3.0, at least 3.2, at least 3.5, at least 3.7, at least 4.0, at least 4.2, at least 4.5, at least 4.7, at least 4.9, or at least 5-fold, as compared to the number and/or level of viral genomes delivered by an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139. In some embodiments, the brain region comprises a midbrain region (e.g., the hippocampus or thalamus) and/or the brainstem. [0198] In some embodiments, an AAV capsid variant described herein delivers an increased level of frataxin to a brain region. In some embodiments, the frataxin level is increased by at least 20, at least 25, at least 30, or at least 35-fold, as compared to the frataxin level delivered by an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139. In some embodiments, the brain region comprises a temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, cerebellum, or a combination thereof. [0199] In some embodiments an AAV capsid variant described herein is enriched at least 3, at least 3.5, at least 4, at least 4.5, at least 5, at least 5.5, at least 6, or at least 6.5-fold, in a spinal cord region compared to enrichment in a spinal cord region of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139. In some embodiments, the spinal cord region comprises a cervical spinal cord region, a lumbar spinal cord region, a thoracic spinal cord region, or a combination thereof. [0200] In some embodiments, an AAV capsid variant described herein shows preferential transduction in a brain region relative to the transduction in the dorsal root ganglia (DRG). In some embodiments an AAV capsid variant described herein shows preferential transduction in a brain region relative to the transduction in the liver. [0201] In some embodiments, an AAV capsid variant described herein is capable of transducing neuronal cells. [0202] In some embodiments, an AAV capsid variant described herein is capable of transducing non- neuronal cells, e.g., glial cells (e.g., oligodendrocytes or astrocytes). In some embodiments, the AAV capsid variant is capable of transducing neuronal cells and non-neuronal cells, e.g., glial cells (e.g., oligodendrocytes or astrocytes). In some embodiments, the non-neuronal cells are glial cells (e.g., oligodendrocytes or astrocytes).
Attorney Docket No.14640.0092-00304 [0203] In some embodiments, an AAV capsid variant described herein has an increased tropism for a heart cell or heart tissue, e.g., a heart cell or a heart tissue of a heart atrium or a heart ventricle, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 138. In some embodiments, an AAV capsid variant described herein has an increased tropism for a heart cell or heart tissue, e.g., a heart cell or a heart tissue of a heart atrium or a heart ventricle, relative to the tropism of an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139. [0204] In some embodiments, an AAV capsid variant described herein delivers an increased level of frataxin to a heart region. In some embodiments, the frataxin level is increased by at least 1.5, at least 2, or at least 2.5-fold, as compared to the frataxin level delivered by an AAV capsid comprising the amino acid sequence of SEQ ID NO: 139. [0205] In some embodiments, an AAV capsid variant described herein has decreased tropism for the liver. In some embodiments, an AAV capsid variant comprises a modification that results in reduced tropism (e.g., de-targeting) and/or activity in the liver. In some embodiments, the reduced tropism in the liver is compared to an otherwise similar capsid that does not comprise the modification, e.g., a wild-type capsid polypeptide. In some embodiments, an AAV capsid variant described comprises a modification that results in one or more of the following properties: (1) reduced tropism in the liver; (2) de-targeted expression in the liver; (3) reduced activity in the liver; and/or (4) reduced binding to galactose. In some embodiments, the reduction in any one, or all, of properties (1)-(3) is compared to an otherwise similar AAV capsid variant that does not comprise the modification. In some embodiments, the AAV capsid variant, e.g., the AAV capsid variant having reduced tropism in the liver, comprises one or more of: an amino acid other than A, G, K, M, N, Q, R, S, and/or T present at a position corresponding to amino acid 581 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A, C, H, I, K, S, T, and/or V present at a position corresponding to amino acid 582 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A, G, H, K, M, N, Q, R, S, T, and/or V present at a position corresponding to amino acid 583 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than L, M, P, Q, R. T and/or W present at a position corresponding to amino acid 584 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than F, H, I, K, M, T and/or Y present at a position corresponding to amino acid 585 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than E, G, H, L, M, N, Q, T, and/or W present at a position corresponding to amino acid 586 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A, C, G, H, L, M, R, and/or S present at a position corresponding to amino acid 587 of the amino acid sequence of SEQ ID NO: 138; an amino acid other than A, C, D, F, G, H, M, Q, S, V, W, and/or Y present at a position corresponding to amino acid 588 of the amino acid sequence of SEQ ID NO: 138; and/or an amino acid other than A, C, E, G, H, M, N, P, Q, S, V, and/or W present at a position corresponding to amino acid 589 of the amino acid sequence of SEQ ID NO: 138. [0206] In some embodiments, an AAV capsid variant of the present disclosure is isolated. In some embodiments, an AAV capsid variant of the present disclosure is recombinant. In some embodiments, a
Attorney Docket No.14640.0092-00304 polynucleotide encoding an AAV capsid polypeptide, e.g., an AAV capsid variant, of the present disclosure is isolated. In some embodiments, the polynucleotide is recombinant. [0207] Also provided herein are polynucleotide sequences encoding any of the AAV capsid variants described above and AAV particles, vectors, and cells comprising the same. Exemplary Properties of AAV Capsids [0208] In some embodiments, an AAV particle may comprise a capsid protein or variant of any natural or recombinant AAV serotype. AAV serotypes may differ in characteristics such as, but not limited to, packaging, tropism, transduction, and immunogenic profiles. Without being bound by theory, it is believed in some embodiments that the AAV capsid protein, e.g., an AAV capsid variant, can modulate AAV particle tropism in a particular tissue. [0209] In some embodiments, an AAV capsid variant described herein allows for blood brain barrier penetration following intravenous administration. In some embodiments, the AAV capsid variant allows for blood brain barrier penetration following intravenous administration, focused ultrasound (FUS), e.g., coupled with the intravenous administration of microbubbles (FUS-MB), or MRI-guided FUS coupled with intravenous administration. In some embodiments, the AAV capsid variant allows for increased distribution to a brain region. In some embodiments, the brain region comprises a temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, cerebellum, frontal cortex, sensory cortex, motor cortex, dentate nucleus, cerebellar cortex, cerebral cortex, brain stem, or a combination thereof. In some embodiments, the AAV capsid variant allows for preferential transduction in a brain region relative to the transduction in the dorsal root ganglia (DRG). In some embodiments, the AAV capsid variant allows for preferential transduction in a brain region relative to the transduction in the liver. In some embodiments, the AAV capsid variant allows for transduction in neuronal cells. In some embodiments, the AAV capsid variant allows for transduction in a non-neuronal cell, e.g., a glial cell (e.g., an astrocyte, an oligodendrocyte, or a combination thereof). In some embodiments, the AAV capsid variant allows for transduction in both neuronal cells and non-neuronal cell, e.g., a glial cell (e.g., an astrocyte, an oligodendrocyte, or a combination thereof). [0210] In some embodiments, an AAV capsid variant allows for increased distribution to a spinal cord region. In some embodiments, the spinal region comprises a cervical spinal cord region, thoracic spinal cord region, and/or lumbar spinal cord region. [0211] In some embodiments, the AAV capsid variant allows for increased distribution to a heart region. [0212] In some embodiments, the AAV capsid variant is suitable for intramuscular administration and/or transduction of muscle fibers. In some embodiments, the AAV capsid variant, allows for increased distribution to a muscle region. In some embodiments, the muscle region comprises a heart muscle, quadriceps muscle, a diaphragm muscle region, or a combination thereof.
Attorney Docket No.14640.0092-00304 [0213] In some embodiments, an AAV capsid described herein comprises a modification as described in Jose et al. High-Resolution Structural Characterization of a New Adenoassociated Virus Serotype 5 Antibody Epitope toward Engineering Antibody-Resistant Recombinant Gene Delivery Vectors. Journal of Virology.2019 Jan.93(1):e01394-18; Qian et al. Directed Evolution of AAV Serotype 5 for Increased Hepatocyte Transduction and Retained Low Humoral Seroreactivity. Molecular Therapy: Methods & Clinical Development.2020 Oct.20:122-132; Afione et al. Identification and Mutagenesis of the Adeno-Associated Virus 5 Sialic Binding Region. Journal of Virology.2015 Feb. 89(3):1660-1672; the contents of which are each hereby incorporated by reference in their entirety. [0214] In some embodiments, the initiation codon for translation of the AAV VP1 capsid protein, e.g., a capsid variant, described herein may be CTG, TTG, or GTG as described in US Patent No. US8163543, the contents of which are herein incorporated by reference in its entirety. [0215] The present disclosure refers to structural capsid proteins (including VP1, VP2 and VP3) which are encoded by capsid (Cap) genes. These capsid proteins form an outer protein structural shell (e.g. capsid) of a viral vector such as AAV. VP capsid proteins synthesized from Cap polynucleotides generally include a methionine as the first amino acid in the peptide sequence (Met1), which is associated with the start codon (AUG or ATG) in the corresponding Cap nucleotide sequence. However, it is common for a first-methionine (Met1) residue or generally any first amino acid (AA1) to be cleaved off after or during polypeptide synthesis by protein processing enzymes such as Met-aminopeptidases. This “Met/AA-clipping” process often correlates with a corresponding acetylation of the second amino acid in the polypeptide sequence (e.g., alanine, valine, serine, threonine, etc.). Met-clipping commonly occurs with VP1 and VP3 capsid proteins but can also occur with VP2 capsid proteins. [0216] Where the Met/AA-clipping is incomplete, a mixture of one or more (one, two or three) VP capsid proteins comprising the viral capsid may be produced, some of which may include a Met1/AA1 amino acid (Met+/AA+) and some of which may lack a Met1/AA1 amino acid as a result of Met/AA- clipping (Met-/AA-). For further discussion regarding Met/AA-clipping in capsid proteins, see Jin, et al. Direct Liquid Chromatography/Mass Spectrometry Analysis for Complete Characterization of Recombinant Adeno-Associated Virus Capsid Proteins. Hum Gene Ther Methods.2017 Oct.28(5):255- 267; Hwang, et al. N-Terminal Acetylation of Cellular Proteins Creates Specific Degradation Signals. Science.2010 February 19.327(5968): 973–977; the contents of which are each incorporated herein by reference in its entirety. [0217] According to the present disclosure, references to capsid proteins, e.g., AAV capsid variants, is not limited to either clipped (Met-/AA-) or unclipped (Met+/AA+) and may, in context, refer to independent capsid proteins, viral capsids comprised of a mixture of capsid proteins, and/or polynucleotide sequences (or fragments thereof) which encode, describe, produce or result in capsid proteins of the present disclosure. A direct reference to a capsid protein or capsid polypeptide (such as VP1, VP2 or VP2) may also comprise VP capsid proteins which include a Met1/AA1 amino acid
Attorney Docket No.14640.0092-00304 (Met+/AA+) as well as corresponding VP capsid proteins which lack the Met1/AA1 amino acid as a result of Met/AA-clipping (Met-/AA-). [0218] Further according to the present disclosure, a reference to a specific SEQ ID NO: (whether a protein or nucleic acid) which comprises or encodes, respectively, one or more capsid proteins which include a Met1/AA1 amino acid (Met+/AA+) should be understood to teach the VP capsid proteins which lack the Met1/AA1 amino acid as upon review of the sequence, it is readily apparent any sequence which merely lacks the first listed amino acid (whether or not Met1/AA1). [0219] As a non-limiting example, reference to a VP1 polypeptide sequence which is 736 amino acids in length and which includes a “Met1” amino acid (Met+) encoded by the AUG/ATG start codon may also be understood to teach a VP1 polypeptide sequence which is 735 amino acids in length and which does not include the “Met1” amino acid (Met-) of the 736 amino acid Met+ sequence. As a second non-limiting example, reference to a VP1 polypeptide sequence which is 736 amino acids in length and which includes an “AA1” amino acid (AA1+) encoded by any NNN initiator codon may also be understood to teach a VP1 polypeptide sequence which is 735 amino acids in length and which does not include the “AA1” amino acid (AA1-) of the 736 amino acid AA1+ sequence. [0220] References to viral capsids formed from VP capsid proteins (such as reference to specific AAV capsid serotypes), can incorporate VP capsid proteins which include a Met1/AA1 amino acid (Met+/AA1+), corresponding VP capsid proteins which lack the Met1/AA1 amino acid as a result of Met/AA1-clipping (Met-/AA1-), and combinations thereof (Met+/AA1+ and Met-/AA1-). [0221] As a non-limiting example, an AAV capsid serotype can include VP1 (Met+/AA1+), VP1 (Met-/AA1-), or a combination of VP1 (Met+/AA1+) and VP1 (Met-/AA1-). An AAV capsid serotype can also include VP3 (Met+/AA1+), VP3 (Met-/AA1-), or a combination of VP3 (Met+/AA1+) and VP3 (Met-/AA1-); and can also include similar optional combinations of VP2 (Met+/AA1) and VP2 (Met- /AA1-). Additional AAV Capsid Sequences [0222] In some embodiments, the AAV capsid variant comprises, immediately subsequent to position 570, 571, 572, 573, 574, 575, or 576, numbered according to SEQ ID NO: 138 (in other words, immediately subsequent to an amino acid corresponding to position 570, 571, 572, 573, 574, 575, 576, or 577 of the amino acid sequence of SEQ ID NO: 138), at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, or at least 16 consecutive amino acids of any one of the amino acid sequences provided in Table 1, 2A, 2B, 14, or 23- 28. [0223] In some embodiments, the AAV capsid variant comprises, immediately subsequent to position 570, 571, 572, 573, 574, 575, 576, or 577, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987 (the contents of which
Attorney Docket No.14640.0092-00304 are hereby incorporated by reference in their entirety)), at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, or at least 16 consecutive amino acids of any one of the amino acid sequence provided in Table 1, 2A, 2B, 14, or 23- 28. In some embodiments, the at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, or at least 16 consecutive amino acids of any one of the amino acid sequences provided in Table 1, 2A, 2B, 14, or 23-28 replaces at least one, at least two, at least three, at least four, at least five, at least six, at least seven, at least eight, or all of positions T571, N572, N573, Q574, S575, S576, T577, T578, and/or A579, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987). In some embodiments, the at least 3, at least 4, at least 5, at least 6, at least 7, at least 8, at least 9, at least 10, at least 11, at least 12, at least 13, at least 14, at least 15, or at least 16 consecutive amino acids of any one of the amino acid sequences provided in Table 1, 2A, 2B, 14, or 23-28 replaces position T577, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987). In some embodiments, the AAV capsid variant comprises an amino acid other than the wild-type, e.g., native, amino acid, at one, two, three, four, five, six, seven, eight, or all of positions T571, N572, N573, Q574, S575, S576, T577, T578, and/or A579, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987). In some embodiments, the AAV capsid variant comprises an amino acid other than the wild-type, e.g., native, amino acid, at position T577, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987). In some embodiments, the AAV capsid variant comprises a modification, e.g., substitution, at one, two, three, four, five, six, seven, eight, or all of positions T571, N572, N573, Q574, S575, S576, T577, T578, and/or A579, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b, AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987). In some embodiments, the AAV capsid variant comprises a modification, e.g., substitution, at position T577, numbered according to SEQ ID NO: 138 or corresponding to equivalent positions in any other AAV serotype (e.g., AAV1, AAV2, AAV3, AAV3b,
Attorney Docket No.14640.0092-00304 AAV4, AAV6, AAV7, AAV8, AAV9, AAVrh8, AAVrh10, AAVrh32.33, AAVrh74, SEQ ID NO: 139, PHP.N, PHP.B, or an AAV serotype as provided in Table 6 of WO 2021/230987). [0224] In some embodiments, the AAV capsid variant described herein comprises a modification, e.g., substitution, at position 569 (e.g., M569V), 652 (e.g., D652A), 362 (e.g., T362M), 359 (e.g., Q359D), 350 (e.g., E350Q), 533 (e.g., P533S), 585 (e.g., Y585V), 587 (e.g., L587T), 581 (e.g., A581T), 582 (e.g., T582A), 584 (e.g., T584A), or a combination thereof, all numbered according to SEQ ID NO: 138. [0225] In some embodiments, an AAV capsid variant described herein comprises an amino acid from a wild-type AAV5 sequence, e.g., the amino acid sequence of SEQ ID NO: 138, at one or more of positions 581 to 589, numbered according to SEQ ID NO: 138. In some embodiments, the AAV capsid variant comprises 1, 2, 3, 4, 5, 6, 7, 8, or all of: the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 581 (e.g., comprises the amino acid A at position 581); the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 582 (e.g., comprises the amino acid T at position 582); the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 583 (e.g., comprises the amino acid G at position 583); the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 584 (e.g., comprises the amino acid T at position 584); the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 585 (e.g., comprises the amino acid Y at position 585); the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 586 (e.g., comprises the amino acid N at position 586); the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 587 (e.g., comprises the amino acid L at position 587); the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 588 (e.g., comprises the amino acid Q at position 588); and/or the amino acid from a wild-type AAV5 sequence (e.g., SEQ ID NO: 138) at position 589 (e.g., comprises the amino acid E at position 589). [0226] In certain embodiments, an AAV capsid described herein does not comprise a T at position 581, an A at position 582, an A at position 584, a V at position 585, a T at position 585, a V at position 569, an A at position 652, an M at position 362, a Q at position 359, a Q at position 350, an S at position 533, or a combination thereof, all numbered according to SEQ ID NO: 138. [0227] In some embodiments, an AAV capsid described herein does not comprise a modification, e.g., substitution, at positions 581-589 (numbered according to SEQ ID NO: 138), wherein the modification has the amino acid sequence of any of the sequences provided in Tables 2, 7, 8, 10, 12, 14, 16, 18, 20, 22, 24, 26, 28, 30, 32, 34, 36, 39, 41, 43, 45, 47, 49, 51, 53, 55, 57, 59, 61, 63, 65, 67, 69, or 71-86 of WO 2021/242909. [0228] In any of the embodiments described herein, a position numbered according to SEQ ID NO: 138 (in other words, a position in a peptide or AAV capsid variant corresponding to a position of SEQ ID NO: 138) can be identified by providing an alignment of a reference sequence and a query sequence, wherein the reference sequence is SEQ ID NO: 138, and identifying the residues corresponding to the positions in the query sequence that correspond to positions in the reference sequence.
Attorney Docket No.14640.0092-00304 Tabl 5 AAV S S A R A ( I a P R V S G A D T L A G D ( V a T Y p E 1 Q o A N K A S K ( N a D T p E 1 P o E N A g a ( a g g c c c c a c c c c g c g c c c t c c g g
Attorney Docket No.14640.0092-00304 c c c C C C C C C C T A D 1 Q ( E a I R H V P S G T V
AAV Viral Genome [0229] In some embodiments, the AAV particle of the present disclosure serves as an expression vector comprising a viral genome that encodes a FXN protein (e.g., a human FXN protein, e.g., a wildtype human FXN protein). [0230] In some embodiments, an AAV particle, e.g., an AAV particle for the vectorized delivery of a FXN protein described herein, comprises a viral genome, e.g., an AAV viral genome, also referred to as an AAV genome, a vector genome, or an AAV vector genome. In some embodiments, the viral genome, e.g., the AAV viral genome, further comprises an inverted terminal repeat (ITR) region, an enhancer, a promoter, an intron region, an exon region, a nucleic acid encoding a transgene encoding a FXN protein sequence, e.g., any one of the sequences in Table 12 or a sequence that is at least 90% identical to any one of the sequences provided in Table 12), a nucleotide sequence encoding at least one miR binding site (e.g., at least one miR122 binding site), a polyA region, or a combination thereof. Viral Genome Component: Inverted Terminal Repeats (ITRs) [0231] In some embodiments, the viral genome may comprise at least one inverted terminal repeat (ITR) region. The AAV particles of the present disclosure comprise a viral genome with at least one ITR region and a FXN-encoding sequence. In some embodiments, the viral genome has two ITRs. These two ITRs flank the FXN-encoding sequence at the 5’ and 3’ ends. In some embodiments, the ITR functions as an origin of replication comprising a recognition site for replication. In some embodiments, the ITR comprises a sequence region which can be complementary and symmetrically arranged. In some embodiments, the ITR incorporated into a viral genome described herein may be comprised of a naturally occurring polynucleotide sequence or a recombinantly derived polynucleotide sequence.
Attorney Docket No.14640.0092-00304 [0232] In some embodiments, the AAV viral genome may comprise at least one inverted terminal repeat (ITR) region having a length of 50-250 nucleotides. In some embodiments, the AAV viral genome comprises a 5’ ITR region that is 50-250 nucleotides in length and a 3’ ITR region that is 50-250 nucleotides in length. In some embodiments, the 5’ ITR region and the 3’ ITR region may comprise the same length and/or the same sequence. In some embodiments, the 5’ ITR region and the 3’ ITR region are different in length and/or in sequence. [0233] Non-limiting examples of ITR sequence regions are described in Table 6. In some embodiments, the viral genome comprises an ITR comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the viral genome comprises an ITR comprising the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. In some embodiments, the viral genome comprises a 5' ITR region comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the viral genome comprises a 3' ITR region comprising the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. Table 6. Exemplary Inverted Terminal Repeat (ITR) Sequence Regions S R N I I C A G T
GCAGG Viral Genome Component: Promoters and Enhancers [0234] In some embodiments, the viral genome comprises at least one element to enhance the transgene target specificity and expression. See, e.g., Powell et al. Viral Expression Cassette Elements to Enhance Transgene Target Specificity and Expression in Gene Therapy, 2015; the contents of which are herein incorporated by reference in their entirety. Non-limiting examples of elements to enhance the transgene target specificity and expression include promoters, endogenous miRNAs, post-transcriptional
Attorney Docket No.14640.0092-00304 regulatory elements (PREs), polyadenylation (PolyA) region, upstream enhancers (USEs), CMV enhancers, and introns. [0235] In some embodiments, expression of the polypeptides in a target cell may be driven by a specific promoter, including but not limited to, a promoter that is species specific, inducible, tissue- specific, or cell cycle-specific (Parr et al., Nat. Med.3:1145-9 (1997); the contents of which are herein incorporated by reference in their entirety). [0236] In some embodiments, the viral genome comprises a promoter that is sufficient for expression, e.g., in a target cell, of a FXN protein, e.g., a human FXN protein, encoded by a transgene. In some embodiments, the promoter is deemed to be efficient when it drives expression of the FXN encoded in the viral genome of the AAV particle. [0237] In some embodiments, the promoter is deemed to be efficient when it drives expression in the cell or tissue being targeted. [0238] Promoters may be naturally occurring or non-naturally occurring. Non-limiting examples of promoters include viral promoters, plant promoters, and mammalian promoters. In some embodiments, the promoter may be a human promoter. In some embodiments, the promoter may be truncated. [0239] Promoters that promote expression in most tissues include, but are not limited to, human elongation factor 1Į-subunit (EF1Į) promoters, immediate-early cytomegalovirus (CMV) promoters, chicken ȕ-actin (CBA) promoters, CAG promoters, CAG derivative promoters, ȕ glucuronidase (GUSB) promoters, ubiquitin C (UBC) promoters, or a fragment of any one of the aforementioned. A CAG promoter typically comprises: (C) the cytomegalovirus early enhancer element; (A) the promoter, the first exon, and the first intron of the chicken beta-actin gene, and (G) the splice acceptor of the rabbit beta-globin gene. In some embodiments, a derivative of a CAG promoter may comprise (i) a CMVie enhancer and a beta-actin promoter or (ii) a beta-actin promoter and an intron sequence. [0240] Tissue-specific expression elements can be used to restrict expression to certain cell types such as, but not limited to, nervous system promoters which can be used to restrict expression to neurons, astrocytes, or oligodendrocytes. Non-limiting examples of tissue-specific expression elements for neurons include neuron-specific enolase (NSE), platelet-derived growth factor (PDGF), platelet-derived growth factor B-chain (PDGF-ȕ), synapsin (Syn), methyl-CpG binding protein 2 (MeCP2), CaMKII, mGluR2, NFL, NFH, nȕ2, PPE, Enk, and EAAT2 promoters. Non-limiting examples of tissue-specific expression elements for astrocytes include the glial fibrillary acidic protein (GFAP) and EAAT2 promoters. A non-limiting example of a tissue-specific expression element for oligodendrocytes include the myelin basic protein (MBP) promoter. [0241] In some embodiments, the viral genome comprises a ubiquitous promoter. Non-limiting examples of ubiquitous promoters include H1, U6, CMV, CBA (including derivatives CAG, CBh, etc.), EF-1Į, PGK, UBC, GUSB (hGBp), and UCOE (promoter of HNRPA2B1-CBX3). [0242] In some embodiments, the viral genome comprises a CBA promoter. In some embodiments, the viral genome comprises a truncated CBA promoter, e.g., a CBA promoter that is 50-400 nucleotides
Attorney Docket No.14640.0092-00304 in length, e.g., 100-332 nucleotides in length. In some embodiments, the viral genome comprises a CMV promoter. In some embodiments, the viral genome comprises a truncated CMV promoter, e.g., a CMV promoter that is 50-300 nucleotides in length, e.g., a CMV promoter that is 109 nucleotides in length. [0243] In some embodiments, the AAV vector comprises an enhancer element, a promoter, and/or a 5’UTR intron. The enhancer may be, but is not limited to, a CMV enhancer; the promoter may be, but is not limited to, a CMV, CBA, FXN, UBC, GUSB, NSE, Synapsin, MeCP2, or GFAP promoter; and the 5’UTR/intron may be, but is not limited to, SV40, and CBA-MVM. In some embodiments, the enhancer, promoter, and/or intron used in combination may be: (1) CMV enhancer, CMV promoter, SV405’UTR intron; (2) CMV enhancer, CBA promoter, SV405’UTR intron; (3) CMV enhancer, CBA promoter, CBA-MVM 5’UTR intron; (4) UBC promoter; (5) GUSB promoter; (6) NSE promoter; (7) Synapsin promoter; (8) MeCP2 promoter; (9) GFAP promoter; (10) H1 promoter; and/or (11) U6 promoter. [0244] In some embodiments, the viral genome comprises an engineered promoter. [0245] In some embodiments, the viral genome comprises an enhancer. In some embodiments, an enhancer may be a separate component of the viral genome than the promoter. In some embodiments, an enhancer may be 5’ to a promoter sequence in a viral genome. In some embodiments, an enhancer may be 3’ to a promoter sequence in a viral genome. [0246] In some embodiments, the viral genome comprises an enhancer, for example an immediate- early “ie” enhancer or a CMV/globin enhancer. In some embodiments, the enhancer comprises ie1 exon 1 and ie1 intron 1 or a fragment thereof. In some embodiments, the enhancer comprises an ie1 exon 1, an ie1 intron 1 or fragment thereof, a human beta-globin intron 2, and a human beta-globin exon 3. [0247] In some embodiments, an enhancer may comprise at least one intron sequence. In some embodiments, an enhancer may comprise at least one exon sequence. In some embodiments, an enhancer comprises one intron sequence and one exon sequence. In some embodiments, an enhancer sequence comprises two intron sequences. In some embodiments, an enhancer sequence comprises two exon sequences. In some embodiments, an enhancer sequence comprises two intron sequences and two exon sequences. [0248] Exemplary promoters are provided in Table 7. In some embodiments, the promoter comprises or consists of any one of the nucleotide sequences provided in Table 7, or a nucleotide sequence at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. In some embodiments, the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1738. In some embodiments, the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1740. In some embodiments, the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1742. In some embodiments, the promoter comprises or consists of the nucleotide sequence of SEQ ID NO: 1745. Table 7. Exemplary Promoters Promoter SEQ ID NO: Sequence C
Attorney Docket No.14640.0092-00304 C C C
Viral Genome Component: Introns and Exon Sequences [0249] In some embodiments, the AAV viral genome comprises at least one intron and/or exon sequence region. [0250] In some embodiments, the AAV viral genome may comprise at least one intron sequence region. The intron sequence region(s) may be 10-1200 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is about 32 nucleotides in length. As a non- limiting example, the viral genome comprises an intron sequence region that is about 53 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is about 134 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is about 347 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is about 379 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is about 566 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is about 1016 nucleotides in length. As a non-limiting example, the viral genome comprises an intron sequence region that is more than 1016 nucleotides in length. [0251] In some embodiments, the AAV viral genome comprises two intron sequence regions. In some embodiments, the AAV viral genome comprises three intron sequence regions. In some embodiments, the AAV viral genome comprises more than three intron sequence regions. [0252] In some embodiments, the AAV viral genome may comprise at least one exon sequence region. In some embodiments, the exon sequence may be 10-1200 nucleotides in length. As a non- limiting example, the viral genome comprises an exon region that is about 32 nucleotides in length. As a non-limiting example, the viral genome comprises an exon sequence region that is about 53 nucleotides in length. As a non-limiting example, the viral genome comprises an exon sequence region that is about 134 nucleotides in length. As a non-limiting example, the viral genome comprises an exon sequence region that is about 347 nucleotides in length. As a non-limiting example, the viral genome comprises an exon sequence region that is about 379 nucleotides in length. As a non-limiting example, the viral
Attorney Docket No.14640.0092-00304 genome comprises an exon sequence region that is about 566 nucleotides in length. As a non-limiting example, the viral genome comprises an exon sequence region that is about 1016 nucleotides in length. As a non-limiting example, the viral genome comprises an exon sequence region that is more than 1016 nucleotides in length. [0253] In some embodiments, the AAV particle viral genome comprises two exon sequence regions. In some embodiments, the AAV particle viral genome comprises three exon sequence regions. In some embodiments, the AAV particle viral genome comprises more than three exon sequence regions. [0254] In some embodiments, the AAV particle viral genome comprises a hybrid intron/exon sequence region comprising at least one intron and at least one exon. In some embodiments, the hybrid intron/exon sequence region comprises one intron and one exon. In some embodiments, the hybrid intron/exon sequence region comprises two introns and two exons. In some embodiments, an intron or exon sequence may comprise a full-length intron or exon. In some embodiments, an intron or exon sequence may comprise a fragment or variant of an intron or exon sequence. [0255] The hybrid intron/exon sequence region(s) may, independently, have a length such as, but not limited to, 15-100, 100-200, 200-300, 300-400, 400-500, 500-600, 600-700, 700-800, 800-900, 900- 1000, 1000-1100, 1100-1200, and more than 1200 nucleotides. As a non-limiting example, the viral genome comprises a hybrid intron/exon sequence region that is about 379 nucleotides in length. As a non-limiting example, the viral genome comprises a hybrid intron/exon sequence region that is about 566 nucleotides in length. As a non-limiting example, the viral genome comprises a hybrid intron/exon region that is about 379 nucleotides in length. [0256] In some embodiments, the intron/exon sequence region is an enhancer sequence. In some embodiments, the intron/exon sequence region is not an enhancer sequence. [0257] In some embodiments, the intron/exon sequence region is a component of a promoter sequence. In some embodiments, the intron/exon sequence region is not a component of a promoter sequence. [0258] In some embodiments, the AAV particle viral genome comprises at least one intron and/or exon sequence region. Non-limiting examples of intron and exon sequence regions are described in Table 8. In some embodiments, the exon sequence region comprises the nucleotide sequence of SEQ ID NO: 1816, or a nucleotide sequence with at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence identity thereto. In some embodiments, the exon sequence region comprises the nucleotide sequence of SEQ ID NO: 1817, or a nucleotide sequence with at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence identity thereto. In some embodiments, the exon sequence region comprises the nucleotide sequence of SEQ ID NO: 1819, or a nucleotide sequence with at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence identity thereto. In some embodiments, the exon sequence region comprises the nucleotide sequence of SEQ ID NO: 1820, or a nucleotide sequence with at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence
Attorney Docket No.14640.0092-00304 identity thereto. In some embodiments, the exon sequence region comprises the nucleotide sequence of SEQ ID NO: 1821, or a nucleotide sequence with at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence identity thereto. Table 8 Exemplary Intron and Exon Sequence Regions S R G C G T C T h in G G C T T G G ie C ie ( T C A h T in T G A h C 3
Viral Genome Component: miR Binding Site [0259] Tissue- or cell-specific expression of the AAV viral particles of the disclosure can be enhanced by introducing tissue- or cell-specific regulatory sequences, e.g., promoters, enhancers, microRNA binding sites, e.g., a detargeting site. Without being bound by theory, it is believed that an encoded miR binding site can modulate, e.g., prevent, suppress, or otherwise inhibit, the expression of a gene of interest on the viral genome of the invention, based on the expression of the corresponding endogenous microRNA (miRNA) or a corresponding controlled exogenous miRNA in a tissue or cell, e.g., a non-targeting cell or tissue. In some embodiments, a miR binding site modulates, e.g., reduces, expression of the FXN-encoding region in a viral genome of an AAV particle described herein in a cell or tissue where the corresponding mRNA is expressed. In some embodiments, the miR binding site modulates, e.g., reduces, expression of the encoded FXN protein in a cell or tissue of the DRG or liver. [0260] In some embodiments, the viral genome of an AAV particle described herein comprises a nucleotide sequence encoding a microRNA binding site, e.g., a detargeting site. In some embodiments, the viral genome of an AAV particle described herein comprises a nucleotide sequence encoding a miR
Attorney Docket No.14640.0092-00304 binding site, a microRNA binding site series (miR BSs), or a reverse complement thereof. In some embodiments, the 3' UTR of the viral genome may be engineered to include at least one miRNA binding site. [0261] In some embodiments, the encoded miR binding site series comprise at least 1-5 copies, e.g., at least 1-3, 2-4, 3-5, or at least 1, at least 2, at least 3, at least 4, at least 5 or more copies of a miR binding site (miR BS). In some embodiments, the encoded miR binding site series comprises 3 copies of a miR binding site. In some embodiments, all copies are identical, e.g., comprise the same miR binding site. In some embodiments, the miR binding sites within the encoded miR binding site series are continuous and not separated by a spacer. In some embodiments, the miR binding sites within an encoded miR binding site series are separated by a spacer, e.g., a non-coding sequence. [0262] In some embodiments, the viral genome comprises at least one sequence encoding a miRNA binding site to reduce the expression of the transgene in a specific tissue. In some embodiments, the viral genome may comprise a miR122 binding site (miR122BS) or tandem copies of the miR122BS to reduce the expression of the viral genome in the liver. In some embodiments, the viral genome may comprise a miR-183 miRNA binding site (miR-183BS) or tandem copies of the miR-183BS to reduce expression of the viral genome in the DRG. [0263] In some embodiments, the miR binding site may be 20-75 nucleotides in length. In some embodiments, the miR binding site is 23 nucleotides in length. In some embodiments, the miR binding site is 71 nucleotides in length. [0264] Non-limiting examples of miR-binding site sequence regions are shown in Table 9. [0265] In some embodiments, the encoded miR binding site is fully complementary to an miR. In some embodiments, the encoded miR binding site is partially complementary to an miR. In some embodiments, the miR is expressed in the liver or in hepatocytes. In some embodiments, the miR is miR122. In some embodiments, the encoded miR binding site or encoded miR binding site series comprises a miR122 binding site sequence. In some embodiments, the encoded miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1827, or a nucleotide sequence having at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99%sequence identity, or comprising at least one, at least two, at least three, at least four, at least five, or six modifications, but no more than six modifications, relative to the nucleotide sequence of SEQ ID NO: 1827, wherein the modification can result in a mismatch between the encoded miR binding site and the corresponding miRNA. In some embodiments, the viral genome comprises at least 2, at least 3, at least 4, or at least 5 copies of the encoded miR122 binding site, e.g., an encoded miR122 binding site series, optionally wherein the encoded miR122 binding site series comprises the nucleotide sequence of SEQ ID NO: 1826, or a nucleotide sequence having at least 50%, at least 55%, at least 60%, at least 65%, at least 70%, at least 75%, at least 80%, at least 85%, at least 90%, at least 95%, or at least 99% sequence identity, or comprising at least one, at least two, at least three, at least four, at least five, at least six, or at least seven modifications, but no more than ten modifications, relative to the nucleotide sequence of SEQ ID NO:
Attorney Docket No.14640.0092-00304 1826, wherein the modification can result in a mismatch between the encoded miR binding site and the corresponding miRNA. Table 9 Exem lar miR Bindin Site Se uence Re ions S N m s S b
n ng s te Viral Genome Component: Polyadenylation Region [0266] In some embodiments, the viral genome of the AAV particles of the present disclosure comprises at least one polyadenylation (polyA) region. In some embodiments, the polyA region is positioned 3’ relative to the nucleic acid encoding a FXN protein described herein. [0267] In some embodiments, the polyA region comprises a length of about 100 to about 500 nucleotides, e.g., about 477 nucleotides. In some embodiments, the polyA region comprises a length of 477 nucleotides. [0268] A non-limiting example of a polyA region is described in Table 10. In some embodiments, the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. Table 10. Exemplary PolyA Region S R N G G G T C h T A C C G
Viral Genome Component: Filler (Stuffer) Sequence [0269] As used herein, the terms “stuffer sequence” and “filler sequence” are used interchangeably. In some embodiments, the AAV particle viral genome comprises at least one filler sequence. In some embodiments, the AAV particle viral genome comprises a filler sequence comprising a human albumin sequence. In some embodiments, the AAV particle viral genome comprises the filler sequence of Alb2034. In some embodiments, the AAV particle viral genome comprises the filler sequence of Alb2106. In some embodiments, the AAV particle viral genome comprises the filler sequence of
Attorney Docket No.14640.0092-00304 Alb2264. In some embodiments, the AAV particle viral genome comprises the filler sequence of Alb2266 [0270] Non-limiting examples of filler sequences are described in Table 11. In some embodiments, the filler sequence comprises the nucleotide sequence of SEQ ID NO: 1838, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. In some embodiments, the filler sequence comprises the nucleotide sequence of SEQ ID NO: 1839, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. In some embodiments, the filler sequence comprises the nucleotide sequence of SEQ ID NO: 1840, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. In some embodiments, the filler sequence comprises the nucleotide sequence of SEQ ID NO: 1841, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. Table 11. Exemplary Filler Sequences S R N T T C G A C A A C C T G T C C C A A G G A A T C C T G A T A A A T A C
Attorney Docket No.14640.0092-00304 A A G A T T G T A G G A A T T T C T C T C C T A C C A A T C A C A T T A G A T G C C T A T A T T G T C G T T A A T G A G A A
Attorney Docket No.14640.0092-00304 C T G C T T C C A T A T A T C C C T T T T A T C T A A T A C A A C T A G C T T A G C C A T A A A C T T A T A T T T A A T A T
Attorney Docket No.14640.0092-00304 A A G T T A A T T T C A G A T A T
Viral Genome Component: FXN-Encoding Sequences [0271] In some embodiments, the disclosure provides an AAV particle comprising a viral genome encoding a FXN protein, e.g., a human frataxin protein, wherein the viral genome comprises the polynucleotide sequence of any one of SEQ ID NOs: 1797, 1801, 1808, or 1809. In some embodiments, the viral genome comprises the polynucleotide sequence of SEQ ID NO: 1797. In some embodiments, the viral genome comprises a promoter operably linked to a polynucleotide sequence encoding a FXN protein. In some embodiments, the viral genome comprises components as arranged in FIG.3. [0272] In some embodiments, the disclosure herein provides constructs that allow for improved expression of FXN protein (e.g., a human frataxin protein) delivered by gene therapy vectors. [0273] In some embodiments, the disclosure provides constructs that allow for improved biodistribution of FXN protein (e.g., a human frataxin protein) delivered by gene therapy vectors. [0274] In some embodiments, the present disclosure relates to a composition containing or comprising a nucleic acid sequence encoding a FXN protein (e.g., a human frataxin protein) or a functional fragment or variant thereof and methods of administering the composition in vitro or in vivo in a subject, e.g., a human subject and/or an animal model of disease, e.g., Friedreich’s Ataxia. [0275] In some embodiments, the disclosure provides a nucleotide sequence encoding a FXN protein (e.g., a human frataxin protein) for use in an AAV viral genome, wherein the nucleotide sequence comprises any one of the sequences provided in Table 12, or a sequence that is at least 90% identical to any one of the sequences provided in Table 12. In some embodiments, the AAV viral genome further comprises one or more, e.g., all of, a 5’ ITR sequence, a promoter, an intron sequence, a polyA sequence, at least one miR122 binding site, and a 3’ ITR region. In some embodiments, the FXN protein encoded by the viral genome is a human FXN protein. Table 12. Exemplary Frataxin Sequences Type Species Description
Attorney Docket No.14640.0092-00304
[0276] In some embodiments, the FXN protein is encoded by the nucleotide sequence of SEQ ID NO: 1824, or a nucleotide sequence at least 80% (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. [0277] SEQ ID NO: 1824: atgtggactc tcgggcgccg cgcagtagcc ggcctcctgg cgtcacccag cccagcccag gcccagaccc tcacccgggt cccgcggccg gcagagttgg ccccactctg cggccgccgt ggcctgcgca ccgacatcga tgcgacctgc acgccccgcc gcgcaagttc gaaccaacgt ggcctcaacc agatttggaa tgtcaaaaag cagagtgtct atttgatgaa tttgaggaaa tctggaactt tgggccaccc aggctctcta gatgagacca cctatgaaag actagcagag gaaacgctgg actctttagc agagtttttt gaagaccttg cagacaagcc atacacgttt gaggactatg atgtctcctt tgggagtggt gtcttaactg tcaaactggg tggagatcta ggaacctatg tgatcaacaa gcagacgcca aacaagcaaa tctggctatc ttctccatcc agtggaccta agcgttatga ctggactggg aaaaactggg tgtactccca cgacggcgtg tccctccatg agctgctggc cgcagagctc actaaagcct taaaaaccaa actggacttg tcttccttgg cctattccgg aaaagatgct tga [0278] In some embodiments, the encoded FXN protein comprises the amino acid sequence of SEQ ID NO: 1825, or an amino acid sequence at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical thereto. [0279] SEQ ID NO: 1825: MWTLGRRAVAGLLASPSPAQAQTLTRVPRPAELAPLCGRRGLRTDIDATCTPRRASSNQRGLNQIWNVKK QSVYLMNLRKSGTLGHPGSLDETTYERLAEETLDSLAEFFEDLADKPYTFEDYDVSFGSGVLTVKLGGDL GTYVINKQTPNKQIWLSSPSSGPKRYDWTGKNWVYSHDGVSLHELLAAELTKALKTKLDLSSLAYSGKDA [0280] In some embodiments, the AAV viral genome comprises a combination of coding and non- coding nucleic acid sequences. [0281] In some embodiments, the FXN-encoding sequence encodes a gene therapy product including, but not limited to, a polypeptide, protein, RNA molecule, or other gene product that, when expressed in a target cell, provides a desired therapeutic effect. In some embodiments, a gene therapy product may provide a substitute for a non-functional gene or a gene that is absent, expressed in insufficient amounts, or mutated. In some embodiments, a gene therapy product may comprise a substitute for a non-functional protein or polypeptide or a protein or polypeptide that is absent, expressed in insufficient amounts, misfolded, degraded too rapidly, or mutated. For example, a gene therapy product may be encoded by a polynucleotide encoding a FXN protein to treat FA. In some embodiments, the gene therapy product is encoded by a polynucleotide sequence in an AAV genome, wherein the AAV genome comprises the nucleotide sequence of SEQ ID NO: 1797. In some embodiments, the gene
Attorney Docket No.14640.0092-00304 therapy product is encoded by a polynucleotide sequence in an AAV genome, wherein the AAV genome comprises the nucleotide sequence of SEQ ID NO: 1801. In some embodiments, the gene therapy product is encoded by a polynucleotide sequence in an AAV genome, wherein the AAV genome comprises the nucleotide sequence of SEQ ID NO: 1808. In some embodiments, the gene therapy product is encoded by a polynucleotide sequence in an AAV genome, wherein the AAV genome comprises the nucleotide sequence of SEQ ID NO: 1809. [0282] In some embodiments, the payload encodes a messenger RNA (mRNA). As used herein, the term “messenger RNA” (mRNA) refers to any polynucleotide that encodes a polypeptide of interest and that is capable of being translated to produce the encoded polypeptide of interest in vitro, in vivo, in situ, or ex vivo. Certain embodiments provide the mRNA as encoding FXN (e.g., human FXN) or a variant thereof. [0283] In some embodiments, a viral genome may comprise or encode a selectable marker. In some embodiments, a selectable marker may comprise a gene sequence or a protein or polypeptide encoded by a gene sequence expressed in a host cell that allows for the identification, selection, and/or purification of the host cell from a population of cells that may or may not express the selectable marker. In some embodiments, the selectable marker provides resistance to survive a selection process that would otherwise kill the host cell, such as treatment with an antibiotic. In some embodiments, an antibiotic selectable marker may comprise one or more antibiotic resistance factors, including but not limited to neomycin resistance (e.g., neo), hygromycin resistance, kanamycin resistance, and/or puromycin resistance. In some embodiments, a viral genome may comprise a selectable marker including, but not limited to, ȕ-lactamase, luciferase, ȕ-galactosidase, or any other reporter gene as that term is understood in the art. [0284] In some embodiments, a viral genome may encode a selectable marker comprising a fluorescent protein. A fluorescent protein as herein described may comprise any fluorescent marker including but not limited to green, yellow, and/or red fluorescent protein (GFP, YFP, and/or RFP). In some embodiments, a viral genome may encode a selectable marker comprising a human influenza hemagglutinin (HA) tag. [0285] In certain embodiments, a nucleic acid for expression of a FXN protein in a target cell will be incorporated into the viral genome and located between two ITR sequences. Exemplary FXN AAV Viral Genome Sequence Regions and ITR-to-ITR Sequences [0286] In some embodiments, a viral genome described herein comprises a promoter operably linked to a transgene encoding a FXN protein (e.g., a human FXN protein). In some embodiments, the viral genome further comprises an inverted terminal repeat (ITR) region, a promoter, an intron/exon region, a miR binding site region, a polyA region, or a combination thereof (e.g., all of these elements). [0287] Exemplary sequence regions within ITR-to-ITR sequences for viral genomes according to the description are provided in Tables 13A and 13B and disclosures below. An exemplary arrangement of an ITR-to-ITR sequence is also depicted in FIG.3.
Attorney Docket No.14640.0092-00304 Table 13A. Exemplary Sequence Regions within ITR-to-ITR Sequences
Table 13B. Exemplary ITR-to-ITR Sequences
Attorney Docket No.14640.0092-00304 [0288] SEQ ID NO: 1797:
Attorney Docket No.14640.0092-00304 tcttaaaagt tctatgctag gtggctcata tattaagtct taaataatta caaattgata aaaaaaatca gtcaataaaa attacttacg cattctggaa tttgtactct ccaagctgct caaaaagctc acaattttgt ttgattaaat tctgaggctc ttccacaaga ggtttaaatt catcgaactg aaacataaaa gaattgtgtt aatagtatta tgcctcagga tcagatgcag gttgtttttc acccctctgg tcagaatgat atcctttctt cttcctactc aaagccacat gaatagatga atatcaatgc cgctctgtca caatacattc gaactattta tttggtgagc ttgcatcatg taagtggtta agaatattca ggctctggag ttcaatgacc agtatttgag ccaagtcatg taactcattc ttcagttgtg caattttact taccatctcc acatttcaga atccttatcc ctaaaatagg ggcaaagatg gcactcatct catagagctg ctctgtaaac caggtaagct aatgaaatta aacagtgcct tgcatataat aaaagtttaa taaatgatgg ctatgattgt acatattatt attattagtt cacctttcca gttagattcc aggctctcca aaatgagaga ttttacgttg gtcattgttt gtgcatggtg cctggcatac agtagcttct tgaaagcagg aaaaatgagt tttctctgat tctgaatagt tcagaaacca ttacatatgc tagtgggaac cctaaaaatc ctccagaaca gatttcctct cctaacttaa tttcttgtaa aactattcct aaacatcctc aaaaagattt gaaaatattc taaagggata ggttctataa tttgccataa gatactaatt ctagtacttg attaatcctg gaatcaggtt aactcacttt acatctaagt taaatatctt ctaattaaca tttaaattta attttttttt gttctcagga ttgtgaaaaa gagaaaaaaa gatcaaaatt ttttagagat tgctctattc agatctttct attctaacta gtctaaattt tgtcctaggt atcgatgcta cgtagataag tagcatggcg ggttaatcat taactacaga ggaaccccta gtgatggagt tggccactcc ctctctgcgc gctcgctcgc tcactgaggc cgggcgacca aaggtcgccc gacgcccggg ctttgcccgg gcggcctcag tgagcgagcg agcgcgcagc tgcctgcagg [0289] SEQ ID NO: 1801:
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304 gcaagcttcg tttagtgaac cgtcagatcg cctggagacg ccatccacgc tgttttgacc tccatagaag acaccgggac cgatccagcc tccgcggatt cgaatcccgg ccgggaacgg tgcattggaa cgcggattcc ccgtgccaag agtgacgtaa gtaccgccta tagagtctat aggcccacaa aaaatgcttt cttcttttaa tatacttttt tgtttatctt atttctaata ctttccctaa tctctttctt tcagggcaat aatgatacaa tgtatcatgc ctctttgcac cattctaaag aataacagtg ataatttctg ggttaaggca atagcaatat ttctgcatat aaatatttct gcatataaat tgtaactgat gtaagaggtt tcatattgct aatagcagct acaatccagc taccattctg cttttatttt atggttggga taaggctgga ttattctgag tccaagctag gcccttttgc taatcatgtt catacctctt atcttcctcc cacagctcct gggcaacgtg ctggtctgtg tgctggccca tcactttggc aaagaattgg gattcgaacc ggtatgtgga ctctcgggcg ccgcgcagta gccggcctcc tggcgtcacc cagcccagcc caggcccaga ccctcacccg ggtcccgcgg ccggcagagt tggccccact ctgcggccgc cgtggcctgc gcaccgacat cgatgcgacc tgcacgcccc gccgcgcaag ttcgaaccaa cgtggcctca accagatttg gaatgtcaaa aagcagagtg tctatttgat gaatttgagg aaatctggaa ctttgggcca cccaggctct ctagatgaga ccacctatga aagactagca gaggaaacgc tggactcttt agcagagttt tttgaagacc ttgcagacaa gccatacacg tttgaggact atgatgtctc ctttgggagt ggtgtcttaa ctgtcaaact gggtggagat ctaggaacct atgtgatcaa caagcagacg ccaaacaagc aaatctggct atcttctcca tccagtggac ctaagcgtta tgactggact gggaaaaact gggtgtactc ccacgacggc gtgtccctcc atgagctgct ggccgcagag ctcactaaag ccttaaaaac caaactggac ttgtcttcct tggcctattc cggaaaagat gcttgactcg agccattgac tagtacaaac accattgtca cactccacac aaacaccatt gtcacactcc acacaaacac cattgtcaca ctccactgca gtcaggtcta tcctgaggat gggtggcatc cctgtgaccc ctccccagtg cctctcctgg ccctggaagt tgccactcca gtgcccacca gccttgtcct aataaaatta agttgcatca ttttgtctga ctaggtgtcc ttctataata ttatggggtg gaggggggtg gtatggagca aggggcaagt tgggaagaca acctgtaggg cctgcggggt ctattgggaa ccaagctgga gtgcagtggc acaatcttgg ctcactgcaa tctccgcctc ctgggttcaa gcgattctcc tgcctcagcc tcccgagttg ttgggattcc aggcatgcat gaccaggctc agctaatttt tgtttttttg gtagagacgg ggtttcacca tattggccag gctggtctcc aactcctaat ctcaggtgat ctacccacct tggcctccca aattgctggg attacaggcg tgaaccactg ctcccttccc tgtccttggc ctagtctcct gaacaactag ttaaacttgg ctttgagttc cacctgtacc acttgcataa tcttgggaaa gtgagttgcc taattcagtg acattaataa atttattaat ttcttctttc aataaaacct ggagagagct tcatatgtat cagcatatgc taaacttgaa agatacaagt agaaaatgga aggaaatata tctgactcaa tagggatagt tcaagggtta aattaaaagt agtaaagtat tataattaat ctgacatggt acctaatata taataatcat gtattaagaa tgccagtcac cattaaaagt caatgtatga ctttaatcta ctcgaggaaa gaaactatgt cttgttcact gttattatct ctaaaatcca taatcagaag agcaccatgt gtatgagcca cacaataaat atctactgta taatatgtct cttcttgttt ttaaccttca tagataagac tctattgaat tgggacatta gtccagcaag ccattctgtc tctgtctctt ctatggaggg aaaggtttaa ccatcaaaga ctaggtgcat ctcccaaaca acctgaattt aatattcaaa tatgtatcta aattcatttg ttacattttt gtgttcagct tacatattac tttttgagcg acatctattc aaggcctact acttgctgct ctacaaaata ttgccatgct ctatttgccc attaactatt tcttaacctt caagggacat gctcagttct gatataccaa gatttggtat ttaccctccc agcctacata cttccaatct taagagaaca atttttagac tacattcaaa tatagacctc tccaccccat caactatttt atctctcctc tcctatcttt cttgacaaag agtgattaga aatatgcaat gtattttcat tcttaaaagt tctatgctag gtggctcata tattaagtct taaataatta caaattgata aaaaaaatca gtcaataaaa attacttacg cattctggaa tttgtactct ccaagctgct caaaaagctc acaattttgt ttgattaaat tctgaggctc ttccacaaga ggtttaaatt catcgaactg aaacataaaa gaattgtgtt aatagtatta tgcctcagga tcagatgcag gttgtttttc acccctctgg tcagaatgat atcctttctt cttcctactc aaagccacat gaatagatga atatcaatgc cgctctgtca caatacattc gaactattta tttggtgagc ttgcatcatg taagtggtta agaatattca ggctctggag ttcaatgacc agtatttgag ccaagtcatg taactcattc ttcagttgtg caattttact taccatctcc acatttcaga atccttatcc ctaaaatagg ggcaaagatg gcactcatct catagagctg ctctgtaaac caggtaagct aatgaaatta aacagtgcct tgcatataat aaaagtttaa taaatgatgg
Attorney Docket No.14640.0092-00304 ctatgattgt acatattatt attattagtt cacctttcca gttagattcc aggctctcca aaatgagaga ttttacgttg gtcattgttt gtgcatggtg cctggcatac agtagcttct tgaaagcagg aaaaatgagt tttctctgat tctgaatagt tcagaaacca ttacatatgc tagtgggaac cctaaaaatc ctccagaaca gatttcctct cctaacttaa tttcttgtaa aactattcct aaacatcctc aaaaagattt gaaaatattc taaagggata ggttctataa tttgccataa gatactaatt ctagtacttg attaatcctg gaatcaggtt aactcacttt acatctaagt taaatatctt ctaattaaca tttaaattta attttttttt gttctcagga ttgtgaaaaa gagaaaaaaa gatcaaaatt ttttagagat tgctctattc agatctttct attctaacta gtctaaattt tgtcctaggt atcgatgcta cgtagataag tagcatggcg ggttaatcat taactacaga ggaaccccta gtgatggagt tggccactcc ctctctgcgc gctcgctcgc tcactgaggc cgggcgacca aaggtcgccc gacgcccggg ctttgcccgg gcggcctcag tgagcgagcg agcgcgcagc tgcctgcagg [0291] SEQ ID NO: 1809: cctgcaggca gctgcgcgct cgctcgctca ctgaggccgc ccgggcaaag cccgggcgtc gggcgacctt tggtcgcccg gcctcagtga gcgagcgagc gcgcagagag ggagtggcca actccatcac taggggttcc ttgtagttaa tgattaaccc gccatgctac ttatctacgt agccatgcgt cgacataacg cgtccacgtt ctgcttcact ctccccatct cccccccctc cccaccccca attttgtatt tatttatttt ttaattattt tgtgcagcga tgggggcggg gggggggggc gcgcgccagg cggggcgggg cggggcgagg ggcggggcgg ggcgaggcgg agaggtgcgg cggcagccaa tcagagcggc gcgctccgaa agtttccttt tatggcgagg cggcggcggc ggcggcccta taaaaagcga agcgcgcggc gggcgggagc aagcttcgtt tagtgaaccg tcagatcgcc tggagacgcc atccacgctg ttttgacctc catagaagac accgggaccg atccagcctc cgcggattcg aatcccggcc gggaacggtg cattggaacg cggattcccc gtgccaagag tgacgtaagt accgcctata gagtctatag gcccacaaaa aatgctttct tcttttaata tacttttttg tttatcttat ttctaatact ttccctaatc tctttctttc agggcaataa tgatacaatg tatcatgcct ctttgcacca ttctaaagaa taacagtgat aatttctggg ttaaggcaat agcaatattt ctgcatataa atatttctgc atataaattg taactgatgt aagaggtttc atattgctaa tagcagctac aatccagcta ccattctgct tttattttat ggttgggata aggctggatt attctgagtc caagctaggc ccttttgcta atcatgttca tacctcttat cttcctccca cagctcctgg gcaacgtgct ggtctgtgtg ctggcccatc actttggcaa agaattggga ttcgaaccgg tatgtggact ctcgggcgcc gcgcagtagc cggcctcctg gcgtcaccca gcccagccca ggcccagacc ctcacccggg tcccgcggcc ggcagagttg gccccactct gcggccgccg tggcctgcgc accgacatcg atgcgacctg cacgccccgc cgcgcaagtt cgaaccaacg tggcctcaac cagatttgga atgtcaaaaa gcagagtgtc tatttgatga atttgaggaa atctggaact ttgggccacc caggctctct agatgagacc acctatgaaa gactagcaga ggaaacgctg gactctttag cagagttttt tgaagacctt gcagacaagc catacacgtt tgaggactat gatgtctcct ttgggagtgg tgtcttaact gtcaaactgg gtggagatct aggaacctat gtgatcaaca agcagacgcc aaacaagcaa atctggctat cttctccatc cagtggacct aagcgttatg actggactgg gaaaaactgg gtgtactccc acgacggcgt gtccctccat gagctgctgg ccgcagagct cactaaagcc ttaaaaacca aactggactt gtcttccttg gcctattccg gaaaagatgc ttgactcgag ccattgacta gtacaaacac cattgtcaca ctccacacaa acaccattgt cacactccac acaaacacca ttgtcacact ccactgcagt caggtctatc ctgaggatgg gtggcatccc tgtgacccct ccccagtgcc tctcctggcc ctggaagttg ccactccagt gcccaccagc cttgtcctaa taaaattaag ttgcatcatt ttgtctgact aggtgtcctt ctataatatt atggggtgga ggggggtggt atggagcaag gggcaagttg ggaagacaac ctgtagggcc tgcggggtct attgggaacc aagctggagt gcagtggcac aatcttggct cactgcaatc tccgcctcct gggttcaagc gattctcctg cctcagcctc ccgagttgtt gggattccag gcatgcatga ccaggctcag ctaatttttg tttttttggt agagacgggg tttcaccata ttggccaggc tggtctccaa ctcctaatct caggtgatct acccaccttg gcctcccaaa ttgctgggat tacaggcgtg aaccactgct cccttccctg tccttggcct aggatgttgc tatgttccat tcatcatatt atctccatct gcagagtagt gggttagtgg agggtagaaa acattctcct gaacaactag ttaaacttgg ctttgagttc cacctgtacc acttgcataa tcttgggaaa gtgagttgcc taattcagtg acattaataa atttattaat ttcttctttc aataaaacct ggagagagct tcatatgtat
Attorney Docket No.14640.0092-00304
[0292] In some embodiments, the AAV viral genome comprises a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1824. In some embodiments, the AAV viral genome comprises a 5’ ITR region comprising the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical to the nucleotide sequence of SEQ ID NO: 1811 and/or a 3’ ITR region comprising the nucleotide sequence of SEQ ID NO: 1812 or a nucleotide sequence that is at least 90% identical to SEQ ID NO: 1812. [0293] In some embodiments, the AAV viral genome further comprises or consists of a truncated promoter region of 100-332 nucleotides in length and comprises any one of SEQ ID NOs: 1738, 1740, 1742, or 1750, or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to any one of SEQ ID NOs: 1738, 1740, 1742, or 1750.
Attorney Docket No.14640.0092-00304 [0294] In some embodiments, the AAV viral genome further comprises an intron/exon region comprising SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1816. In some embodiments, the AAV viral genome comprises a miR122 binding site comprising SEQ ID NO: 1827 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1827. [0295] In some embodiments, the AAV viral genome further comprises a miR122 binding site series comprising SEQ ID NO: 1826 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1826. [0296] In some embodiments, the AAV viral genome further comprises a polyA region comprising SEQ ID NO: 1828 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1828. [0297] In some embodiments, the AAV viral genome further comprises a filler sequence comprising any one of SEQ ID NOs: 1838, 1839, 1840, or 1841, or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to any one of SEQ ID NOs: 1838, 1839, 1840, or 1841. [0298] In some embodiments, the AAV viral genome does not comprise a filler sequence. [0299] In some embodiments, the AAV particle comprises, from 5' to 3', a 5' ITR comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical thereto; a promoter consisting of the nucleotide sequence of SEQ ID NO: 1742 or a sequence that is at least 90% identical thereto; an intron/exon region comprising the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical thereto; a FXN-encoding sequence comprising SEQ ID NO: 1824; a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that is at least 90% identical thereto; a polyA region comprising the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical thereto; and/or a 3' ITR comprising the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical thereto. In some embodiments, the AAV particle comprising the FXN-encoding sequence of SEQ ID NO: 1824 further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1841 or a sequence that is at least 90% identical thereto, wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. [0300] In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to the nucleotide sequence of SEQ ID NO: 1809.
Attorney Docket No.14640.0092-00304 [0301] In some embodiments, the viral genome is single-stranded. [0302] In some embodiments, the viral genome is capable of forming double-stranded DNA. In some embodiments, the viral genome is self-complementary. [0303] In some embodiments, the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; and (iv) a 3’ ITR region. In some embodiments, features (i), (ii), and/or (iv) may be as described herein. [0304] In some embodiments, the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (iv) at least one miR122 binding site; and (v) a 3’ ITR region. In some embodiments, features (i), (ii), (iv), and/or (v) may be as described stated elsewhere herein. [0305] In some embodiments, the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron and/or exon region; (iv) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; and (vi) a 3’ ITR region. In some embodiments, features (i), (ii), (iii), (v), and/or (vi) may be as described herein. [0306] In some embodiments, the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron and/or exon region; (iv) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; and (vii) a 3’ ITR region. In some embodiments, features (i), (ii), (iii), (v), (vi), and/or (vii) may be as described herein. [0307] In some embodiments, the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron and/or exon region; (iv) a FXN-encoding sequence comprising the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; (vii) a filler sequence; and (viii) a 3’ ITR
Attorney Docket No.14640.0092-00304 region. In some embodiments, features (i), (ii), (iii), (v), (vi), (vii), and/or (viii) may be as described herein. [0308] In some embodiments, the AAV viral genome comprises the nucleotide sequence of SEQ ID NO: 1797 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1797. In some embodiments, the AAV viral genome comprises a truncated promoter comprising or consisting of the nucleotide sequence of SEQ ID NO: 1742, or a sequence that is at least 95% identical to SEQ ID NO: 1742. [0309] In some embodiments, the AAV particle comprises, from 5' to 3', one or more of, e.g., all of, a 5' ITR comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical thereto; a promoter consisting of the nucleotide sequence of SEQ ID NO: 1750 or a sequence that is at least 90% identical thereto; an intron/exon region comprising the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical thereto; a FXN-encoding sequence comprising SEQ ID NO: 1824; a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that is at least 90% identical thereto; a polyA region comprising the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical thereto; and/or a 3' ITR comprising the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical thereto. In some embodiments, the AAV particle comprising the FXN-encoding sequence of SEQ ID NO: 1824 further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1840 or a sequence that is at least 90% identical thereto, wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. [0310] In some embodiments, the AAV viral genome comprises the nucleotide sequence of SEQ ID NO: 1801 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1801. In some embodiments, the AAV viral genome comprises a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to SEQ ID NO: 1801. In some embodiments, the AAV viral genome comprises a truncated promoter comprising or consisting of the nucleotide sequence of SEQ ID NO: 1750, or a sequence that is at least 95% identical to SEQ ID NO: 1750. [0311] In some embodiments, the AAV particle comprises, from 5' to 3', a 5' ITR comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical thereto; a promoter consisting of the nucleotide sequence of SEQ ID NO: 1738 or a sequence that is at least 90% identical thereto; an intron/exon region comprising the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical thereto; a FXN-encoding sequence comprising SEQ ID NO: 1824; a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that is at least 90% identical thereto; a polyA region comprising the nucleotide sequence of SEQ ID NO: 1828 or a
Attorney Docket No.14640.0092-00304 sequence that is at least 90% identical thereto; and/or a 3' ITR comprising the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical thereto. In some embodiments, the AAV particle comprising the FXN-encoding sequence of SEQ ID NO: 1824 further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1838 or a sequence that is at least 90% identical thereto, wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. [0312] In some embodiments, the AAV viral genome comprises the nucleotide sequence of SEQ ID NO: 1808 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1808. In some embodiments, the AAV viral genome comprises a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to SEQ ID NO: 1808. In some embodiments, the AAV viral genome comprises a truncated promoter comprising or consisting of the nucleotide sequence of SEQ ID NO: 1738, or a sequence that is at least 95% identical to SEQ ID NO: 1738. [0313] In some embodiments, the AAV particle comprises, from 5' to 3', a 5' ITR comprising the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical thereto a promoter consisting of the nucleotide sequence of SEQ ID NO: 1740 or a sequence that is at least 90% identical thereto; an intron/exon region comprising the nucleotide sequence of SEQ ID NO: 1816 or a sequence that is at least 90% identical thereto; a FXN-encoding sequence comprising SEQ ID NO: 1824000; a miR122 binding site series comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence that is at least 90% identical thereto; a polyA region comprising the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical thereto; and/or a 3' ITR comprising the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical thereto. In some embodiments, the AAV particle comprising the FXN-encoding sequence of SEQ ID NO: 1824 further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1839 or a sequence that is at least 90% identical thereto, wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. [0314] In some embodiments, the AAV viral genome comprises the nucleotide sequence of SEQ ID NO: 1809 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1809. In some embodiments, the AAV viral genome comprises a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to SEQ ID NO: 1809. In some embodiments, the AAV viral genome comprises a truncated promoter comprising or consisting of the nucleotide sequence of SEQ ID NO: 1740, or a sequence that is at least 95% identical to SEQ ID NO: 1740.
Attorney Docket No.14640.0092-00304 [0315] In some embodiments, the present disclosure provides an AAV particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0316] In some embodiments, the present disclosure provides an AAV particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0317] In some embodiments, the present disclosure provides an AAV particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0318] Without being bound by theory, the FXN-encoding sequences described herein may provide a desirable level of frataxin expression in disease-relevant cells or tissues. For example, in some embodiments, the FXN-encoding sequences described herein facilitate an intermediate level of expression (e.g., 0.5x-3x relative to baseline) such that the resulting expression alleviates one or more symptoms of FXN deficiency or FA while minimizing or avoiding toxicity. In some embodiments, this result is facilitated at least by a truncated promoter as described herein. In some embodiments, the FXN- encoding sequences described herein minimize or avoid overexpression of frataxin in the liver. In some embodiments, this result is facilitated at least by a miR binding site or series as described herein. In some embodiments, desirable expression levels or distribution of frataxin are further facilitated by the AAV capsid variants described herein. For example, without being bound by theory, the AAV capsid variants described herein may enhance delivery to the cerebellum. Further, without being bound by theory, the AAV capsid variants described herein may enhance delivery to the dentate nucleus. Self-Complementary and Single Stranded Vectors [0319] In some embodiments, the AAV viral genome used in the present disclosure is single- stranded. [0320] In some embodiments, the AAV viral genome is capable of forming double-stranded DNA. In some embodiments, the AAV viral genome is self-complementary. See, e.g., US Patent No.7,465,583. scAAV vectors contain both DNA strands that anneal together to form double stranded DNA. By skipping second strand synthesis, scAAVs allow for rapid expression in the cell. [0321] Methods for producing and/or modifying AAV viral genome and particles are disclosed in the art such as pseudotyped AAV vectors (International Patent Publication Nos. WO200028004; WO200123001; WO2004112727; WO 2005005610 and WO 2005072364, the content of each of which are incorporated herein by reference in their entirety).
Attorney Docket No.14640.0092-00304 II. AAV Production [0322] Viral production disclosed herein describes processes and methods for producing AAV particles (with enhanced, improved and/or increased tropism for a target tissue), e.g., an AAV particle comprising an AAV capsid variant that may be used to contact a target cell to deliver FXN. [0323] In some embodiments, disclosed herein is a method of making an AAV particle of the present disclosure, e.g., an AAV particle comprising an AAV capsid variant disclosed herein, wherein the method comprises: (i) providing a cell comprising a viral genome described herein (e.g., a viral genome comprising a FXN-encoding sequence and a nucleic acid encoding an AAV capsid variant disclosed herein) and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in an AAV capsid variant described herein (e.g., an AAV capsid variant listed in Table 3, 4A, or 4B), thereby making the AAV particle. In some embodiments, the viral genome comprises a nucleotide sequence of any one of SEQ ID NOs: 1797, 1801, 1808, and 1809, or a nucleotide sequence at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to any one of the nucleotide sequences of SEQ ID NOs: 1797, 1801, 1808, and 1809. In some embodiments, the AAV capsid variant comprises an amino acid sequence of SEQ ID NO: 982. In some embodiments, the viral genome comprises the nucleotide sequence of any one of SEQ ID NOs: 1797, 1801, 1808, and 1809, or a nucleotide sequence at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) identical to any one of the nucleotide sequences of SEQ ID NOs: 1797, 1801, 1808, and 1809; and the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982. In some embodiments, the method comprises, prior to step (i), introducing a nucleic acid comprising the viral genome into the cell. In some embodiments, the method comprises, prior to step (i), introducing the nucleic acid encoding the AAV capsid variant into the cell. In some embodiments, the AAV particle described herein is an isolated AAV particle. In some embodiments, the AAV particle described herein is a recombinant AAV particle. [0324] In some embodiments, the present disclosure provides a method of making an AAV particle that comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982. In some embodiments, the iral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0325] In some embodiments, the present disclosure provides a method of making an AAV particle that comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0326] In some embodiments, the present disclosure provides a method of making an AAV particle that comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797.
Attorney Docket No.14640.0092-00304 [0327] Any method known in the art may be used for the preparation of AAV particles. In some embodiments, AAV particles are produced in mammalian cells (e.g., HEK293 cells). In another embodiment, AAV particles are produced in insect cells (e.g., Sf9 cells). [0328] Methods of making AAV particles are well known in the art and are described in e.g., U.S. Patent Nos. US6204059, US5756283, US6258595, US6261551, US6270996, US6281010, US6365394, US6475769, US6482634, US6485966, US6943019, US6953690, US7022519, US7238526, US7291498 and US7491508, US5064764, US6194191, US6566118, US8137948; or International Publication Nos. WO1996039530, WO1998010088, WO1999014354, WO1999015685, WO1999047691, WO2000055342, WO2000075353 and WO2001023597; Methods In Molecular Biology, ed. Richard, Humana Press, NJ (1995); O'Reilly et al., Baculovirus Expression Vectors, A Laboratory Manual, Oxford Univ. Press (1994); Samulski et al., J. Vir.63:3822-8 (1989); Kajigaya et al., Proc. Nat'l. Acad. Sci. USA 88: 4646-50 (1991); Ruffing et al., J. Vir.66:6922-30 (1992); Kimbauer
(1996); Zhao et al., Vir.272:382-93 (2000); the contents of each of which are herein incorporated by reference in their entirety. In some embodiments, the AAV particles are made using the methods described in International Patent Publication WO2015191508, the contents of which are herein incorporated by reference in their entirety. III. Pharmaceutical Compositions [0329] In some embodiments, the present disclosure provides pharmaceutical compositions of an adeno-associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0330] In some embodiments, the present disclosure provides pharmaceutical compositions of an adeno-associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0331] In some embodiments, the present disclosure provides pharmaceutical compositions of an adeno-associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0332] In some embodiments, a composition described herein comprises an AAV polynucleotide or AAV genome or AAV particle and at least one excipient. [0333] Although pharmaceutical compositions provided herein, e.g., comprising AAV particles comprising a FXN-encoding sequence, are principally directed to pharmaceutical compositions which are suitable for administration to humans, it will be understood by the skilled artisan that such compositions are generally suitable for administration to any other animal, e.g., to non-human animals, e.g., non- human mammals. Modification of pharmaceutical compositions suitable for administration to humans in order to render the compositions suitable for administration to various animals is well understood, and
Attorney Docket No.14640.0092-00304 the ordinarily skilled veterinary pharmacologist can design and/or perform such modification with merely ordinary, if any, experimentation. Subjects to which administration of the pharmaceutical compositions is contemplated include, but are not limited to, humans and/or other primates; mammals, including commercially relevant mammals such as cattle, pigs, horses, sheep, cats, dogs, mice, and/or rats; and/or birds, including commercially relevant birds such as poultry, chickens, ducks, geese, and/or turkeys. [0334] In some embodiments, compositions are administered to humans, e.g., human patients or human subjects. [0335] In some embodiments, the AAV particle formulations described herein may contain a nucleic acid encoding at least one payload. In some embodiments, the formulations may contain a nucleic acid encoding 1, 2, 3, 4, or 5 payloads. In some embodiments, the formulation may contain FXN-encoding sequence, encoding a FXN protein selected from categories such as, but not limited to, human proteins, veterinary proteins, bacterial proteins, biological proteins, antibodies, immunogenic proteins, therapeutic peptides and proteins, secreted proteins, plasma membrane proteins, cytoplasmic proteins, cytoskeletal proteins, intracellular membrane bound proteins, nuclear proteins, proteins associated with human disease, and/or proteins associated with non-human diseases. In some embodiments, the AAV genome comprises at least one sequence encoding human FXN. In some embodiments, the AAV genome comprises at least one sequence encoding wildtype human FXN. [0336] A pharmaceutical composition in accordance with the present disclosure may be prepared, packaged, and/or sold in bulk, as a single unit dose, and/or as a plurality of single unit doses. As used herein, a “unit dose” refers to a discrete amount of the pharmaceutical composition comprising a predetermined amount of the active ingredient. The amount of the active ingredient is generally equal to the dosage of the active ingredient which would be administered to a subject and/or a convenient fraction of such a dosage such as, for example, one-half or one-third of such a dosage. IV. Formulations [0337] Formulations of the AAV pharmaceutical compositions described herein may be prepared by any method known or hereafter developed in the art of pharmacology. In general, such preparatory methods include the step of bringing the active ingredient into association with an excipient and/or one or more other accessory ingredients, and then, if necessary and/or desirable, dividing, shaping and/or packaging the product into a desired single- or multi-dose unit. [0338] Relative amounts of the active ingredient, the pharmaceutically acceptable excipient, and/or any additional ingredients in a pharmaceutical composition in accordance with the disclosure will vary, depending upon the identity, size, and/or condition of the subject treated and further depending upon the route by which the composition is to be administered. [0339] For example, the composition may comprise between 0.1% and 99% (w/w) of the active ingredient. By way of example, the composition may comprise between 0.1% and 100%, e.g., between .5% and 50%, between 1-30%, between 5-80%, or at least 80% (w/w) active ingredient.
Attorney Docket No.14640.0092-00304 [0340] The AAV particles of the disclosure can be formulated using one or more excipients to: (1) increase stability; (2) increase cell transfection or transduction; (3) permit the sustained or delayed release; (4) alter the biodistribution (e.g., target the viral particle to specific tissues or cell types); (5) increase the translation of encoded protein in vivo; (6) alter the release profile of encoded protein in vivo and/or (7) allow for regulatable expression of the FXN. [0341] Formulations of the present disclosure can include, without limitation, saline, lipidoids, liposomes, lipid nanoparticles, polymers, lipoplexes, core-shell nanoparticles, peptides, proteins, cells transfected with AAV particles (e.g., for transplantation into a subject), nanoparticle mimics and combinations thereof. Further, the AAV particles of the present disclosure may be formulated using self- assembled nucleic acid nanoparticles. [0342] In some embodiments, the present disclosure provides formulations of an adeno-associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982, the amino acid sequence of SEQ ID NO: 738, and/or the amino acid sequence of SEQ ID NO: 739. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0343] In some embodiments, the AAV particles encoding FXN may be formulated to optimize baricity and/or osmolality. In some embodiments, the baricity and/or osmolality of the formulation may be optimized to ensure optimal drug distribution in the central nervous system or a region or component of the central nervous system. Excipients [0344] The formulations of the disclosure can include one or more excipients, each in an amount that together increases the stability of the AAV particle, increases cell transfection or transduction by the AAV particle, increases the expression of frataxin protein, and/or alters the release profile of frataxin proteins. In some embodiments, a pharmaceutically acceptable excipient may be at least 95%, at least 96%, at least 97%, at least 98%, at least 99%, or 100% pure. In some embodiments, an excipient is approved for use for humans and for veterinary use. In some embodiments, an excipient may be approved by United States Food and Drug Administration. In some embodiments, an excipient may be of pharmaceutical grade. In some embodiments, an excipient may meet the standards of the United States Pharmacopoeia (USP), the European Pharmacopoeia (EP), the British Pharmacopoeia, and/or the International Pharmacopoeia. [0345] Excipients, which, as used herein, include, but are not limited to, any and all solvents, dispersion media, diluents, or other liquid vehicles, dispersion or suspension aids, surface active agents, isotonic agents, thickening or emulsifying agents, preservatives, and the like, as suited to the particular dosage form desired. Various excipients for formulating pharmaceutical compositions and techniques for preparing the composition are known in the art (see Remington: The Science and Practice of Pharmacy, 21st Edition, A. R. Gennaro, Lippincott, Williams & Wilkins, Baltimore, MD, 2006; the contents of which are herein incorporated by reference in their entirety). The use of a conventional excipient medium
Attorney Docket No.14640.0092-00304 may be contemplated within the scope of the present disclosure, except insofar as any conventional excipient medium may be incompatible with a substance or its derivatives, such as by producing any undesirable biological effect or otherwise interacting in a deleterious manner with any other component(s) of the pharmaceutical composition. Inactive Ingredients [0346] In some embodiments, AAV formulations may comprise at least one excipient which is an inactive ingredient. As used herein, the term “inactive ingredient” refers to one or more agents that do not contribute to the activity of the pharmaceutical composition included in formulations. In some embodiments, all, none, or some of the inactive ingredients which may be used in the formulations of the present disclosure may be approved by the US Food and Drug Administration (FDA). [0347] Formulations of AAV particles disclosed herein may include cations or anions. In one embodiment, the formulations include metal cations such as, but not limited to, Zn2+, Ca2+, Cu2+, Mg+, or combinations thereof. In some embodiments, formulations may include polymers or polynucleotides complexed with a metal cation (See, e.g., U.S. Pat. Nos.6,265,389 and 6,555,525, the contents of each of which are herein incorporated by reference in their entirety). V. Uses and Applications [0348] The compositions of the disclosure may be administered to a subject, e.g., to deliver a FXN protein, e.g., to a subject who has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency (e.g., Friedreich’s Ataxia (FA)). The compositions may similarly be used in the manufacture of a medicament for administration to a subject having a FXN protein deficiency, such as FA. [0349] In some embodiments, the disclosure provides a method of delivering a FXN protein to a subject comprising administering to the subject an effective amount of a pharmaceutical composition or AAV particle disclosed herein that comprises a viral genome encoding a FXN protein. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency (e.g., FA). [0350] In some embodiments, the disclosure provides a method for treating a disorder associated with FXN deficiency, such as FA. In some embodiments, the disclosure provides an AAV particle or pharmaceutical composition according to any one of the embodiments disclosed herein for treating a disorder associated with FXN deficiency, such as FA. In some embodiments, the present disclosure provides the pharmaceutical composition or the AAV particle of any one the embodiments disclosed herein for use in a method of treating a disorder as disclosed herein, such as FA. [0351] In certain embodiments, a pharmaceutical composition or AAV particle disclosed herein that comprises a viral genome encoding FXN may be administered to a subject to treat a disorder associated with FXN deficiency such as FA. In some embodiments, the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency such as FA. In some embodiments, the treatment results in a 0.5-3x increase in the subject’s FXN protein level as compared to
Attorney Docket No.14640.0092-00304 baseline. In some embodiments, the treatment may halt or slow progression of FA. In certain embodiments, the treatment improves at least one symptom of FA, e.g., impaired sensory functions, impaired motor function (e.g., ataxia and/or involuntary movements), fatigue, chronic pain, seizures, impaired speech, sleep disturbances, metabolic disorders (e.g., diabetes), and/or increased spasticity. In some embodiments, the treatment stabilizes, slows the progression of, or improves the subject’s FA as determined by the modified Friedreich Ataxia Rating Scale (mFARS), the Scale for the Assessment and Rating of Ataxia (SARA), and/or the International Cooperative Ataxia Rating Scale (ICARS). In some embodiments, the treatment slows the subject’s progression of FA as measured by mFARS, SARA, and/or ICARS relative to an individual with the disorder associated with FXN deficiency who has not been administered the pharmaceutical composition or the AAV particle. In some embodiments, the treatment results in an increase in: (i) the level of FXN protein or FXN gene expression in a cell, tissue, (e.g., a cell or tissue of the CNS, e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum), and/or fluid (e.g., CSF and/or serum), of the subject, optionally wherein the level of FXN protein or gene expression is increased by 0.5-3x as compared to baseline; and/or (ii) the number and/or level of viral genomes (VG) per cell in a CNS tissue (e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) of the subject relative to the number and/or level of VG per cell in a peripheral tissue of the subject. [0352] Without being bound by theory, AAV particles comprising AAV capsid variants described herein (e.g., AAV5 variants comprising a peptide comprising the amino acid sequence of SEQ ID NO: 943 or SEQ ID NO: 1533, e.g., in loop VIII) may be desirable for the alleviation or treatment of frataxin deficiency or Friedreich’s Ataxia at least due to their tropism of the dentate nucleus. [0353] In some embodiments, the present disclosure provides delivery of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0354] In some embodiments, the present disclosure provides delivery of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0355] In some embodiments, the present disclosure provides delivery of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824
Attorney Docket No.14640.0092-00304 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0356] In some embodiments, the present disclosure encompasses the delivery of pharmaceutical, prophylactic, diagnostic, or imaging compositions in combination with agents that may improve their bioavailability, reduce and/or modify their metabolism, and/or modify their distribution within the body. [0357] In some embodiments, the present disclosure encompasses the delivery of pharmaceutical, prophylactic, diagnostic, or imaging compositions in combination with agents that may improve their bioavailability, reduce and/or modify their metabolism, and/or modify their distribution within the body. [0358] In certain embodiments, the pharmaceutical compositions described herein are used as research tools, particularly in in vitro investigations using human cell lines such as HEK293T and in vivo testing in nonhuman primates which will occur prior to human clinical trials. [0359] The present disclosure provides a method for treating a disease, disorder and/or condition in a mammalian subject, including a human subject, comprising administering to the subject any of the viral particles e.g., AAV, AAV particle, or AAV genome that produces FXN protein described herein (e.g, viral genomes or VG) or administering to the subject a particle comprising said AAV particle or AAV genome, or administering to the subject any of the described compositions, including pharmaceutical compositions. [0360] In some embodiments, the present disclosure provides a method of treating a disorder associated with FXN deficiency such as FA comprising administering an effective amount of an adeno- associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0361] In some embodiments, the present disclosure provides a method of treating a disorder associated with FXN deficiency such as FA comprising administering an effective amount of an adeno- associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0362] In some embodiments, the present disclosure provides a method of treating a disorder associated with FXN deficiency such as FA comprising administering an effective amount of an adeno- associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0363] In some embodiments, AAV particles of the present disclosure, through delivery of a functional FXN that is a therapeutic product comprising a FXN protein or variant thereof, can modulate the level or function of FXN in the CNS.
Attorney Docket No.14640.0092-00304 [0364] A functional FXN may alleviate or reduce symptoms that result from abnormal level and/or function of the FXN gene product (e.g., an absence or defect in a FXN protein) in a subject in need thereof or that otherwise confers a benefit to a CNS disorder in a subject in need thereof. [0365] As non-limiting examples, companion or combination therapeutic products delivered by AAV particles of the present disclosure may include, but are not limited to, growth and trophic factors, cytokines, hormones, neurotransmitters, enzymes, anti-apoptotic factors, angiogenic factors, FXN polypeptides, and any protein known to be mutated in pathological disorders such as FA (e.g., brain specific Mir-128a, See Adlakha and Saini, Molecular cancer, 2014, 13:33, incorporated herein by reference in its entirety). [0366] In some embodiments, the neurodegenerative disorder is Friedreich’s Ataxia, e.g., resulting from expansion of an intronic GAA triplet repeat in the FXN gene, which reduces expression of the mitochondrial protein frataxin causing progressive damage to the nervous system. [0367] In some embodiments, the AAV particles of the present disclosure may be used to ameliorate at least one symptom of FA, including, but not limited to, impaired sensory functions, impaired motor function, e.g., ataxia and/or involuntary movements, fatigue, chronic pain, seizures, impaired speech, sleep disturbances, metabolic disorders, e.g., diabetes, and increased spasticity. [0368] In some embodiments, the delivery of the AAV particles may halt or slow the disease progression of Friedreich’s Ataxia by 10%, 20%, 30%, 40%, 50%, 60%, 70%, 80%, 90%, 95%, or more than 95% using a known analysis method and comparator group for Friedreich’s Ataxia. As a non- limiting example, the delivery of the AAV particles may halt or slow progression of Friedreich’s Ataxia as measured by mFARS/SARA by 50% relative to a comparator group. [0369] In some embodiments, the AAV particle encoding FXN may increase the amount of FXN in a tissue by 1%, 2%, 3%, 4%, 5%, 6%, 7%, 8%, 9%, 10%, 11%, 12%, 13%, 14%, 15%, 16%, 17%, 18%, 19%, 20%, 25%, 30%, 35%, 40%, 45%, 50%, 55%, 60%, 65%, 70%, 75%, 80%, 85%, 90%, 95%, 97%, 99%, or more than 100%. In some embodiments, the AAV particle encoding FXN may increase the amount of FXN in a tissue to be comparable to (e.g., approximately the same as) the amount of FXN in the corresponding tissue of a healthy subject. In some embodiments, the AAV particle encoding FXN may increase the amount of FXN in a tissue effective to reduce one or more symptoms of a disease associated with decreased FXN expression or a deficiency in the quantity and/or function of FXN, e.g., FA. VI. Delivery of AAV Particles Delivery to Cells [0370] In some aspects, the present disclosure provides a method of delivering to a cell or tissue any of the above-described AAV particles, comprising contacting the cell or tissue with said AAV particle or contacting the cell or tissue with a formulation comprising said AAV particle, or contacting the cell or tissue with any of the described compositions, including pharmaceutical compositions. The method of delivering the AAV particle to a cell or tissue can be accomplished in vitro, ex vivo, or in vivo.
Attorney Docket No.14640.0092-00304 [0371] In some embodiments, the AAV particles are delivered to a cell, tissue, or region of the CNS. In some embodiments, the AAV particles are delivered to a cell or tissue of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum, or a combination thereof. Delivery to Subjects [0372] In some aspects, the present disclosure additionally provides a method of delivering to a subject, including a mammalian subject, any of the above-described AAV particles comprising administering to the subject said AAV particle, or administering to the subject a formulation comprising said AAV particle, or administering to the subject any of the described compositions, including pharmaceutical compositions. [0373] In some embodiments, the AAV particles may be delivered to bypass anatomical blockages (e.g., the blood brain barrier). [0374] In some embodiments, the AAV particles may be formulated and delivered to a subject by a route which increases the speed of drug effect as compared to oral delivery. [0375] In some embodiments, the AAV particles may be delivered using intrathecal infusion. [0376] In some embodiments, a subject may be administered the AAV particles described herein using a bolus infusion. [0377] In some embodiments, the AAV particles encoding FXN may be delivered in a continuous and/or bolus infusion. A different dosing regimen may be used for different sites of delivery or the same dosing regimen may be used for each site of delivery. As a non-limiting example, the sites of delivery may be in the cervical and the lumbar region. As another non-limiting example, the sites of delivery may be in the cervical region. As another non-limiting example, the sites of delivery may be in the lumbar region. [0378] In some embodiments, the AAV particles may be delivered to a subject via a single route of administration. [0379] In some embodiments, the AAV particles may be delivered to a subject via a multi-site route of administration. For example, a subject may be administered the AAV particles at 2, 3, 4, 5, or more than 5 sites. [0380] In some embodiments, a subject may be administered the AAV particles described herein using sustained delivery over a period of minutes, hours or days. The infusion rate may be changed depending on the subject, distribution, formulation or another delivery parameter known to those in the art. [0381] In some embodiments, if continuous delivery (continuous infusion) of the AAV particles is used, the continuous infusion may be for 1 hour, 2, hours, 3 hours, 4 hours, 5 hours, 6 hours, 7 hours, 8 hours, 9 hours, 10 hours, 11 hours, 12 hours, 13 hours, 14 hours, 15 hours, 16 hours, 17 hours, 18 hours, 19 hours, 20 hours, 21 hours, 22 hours, 23 hours, 24 hours, or more than 24 hours.
Attorney Docket No.14640.0092-00304 [0382] In some embodiments, the intracranial pressure may be evaluated prior to administration. The route, volume, AAV particle concentration, infusion duration and/or vector titer may be optimized based on the intracranial pressure of a subject. [0383] In some embodiments, the AAV particles may be delivered by systemic delivery. In some embodiments, the systemic delivery may be by intravascular administration. [0384] In some embodiments, the AAV particles may be delivered by injection into the CSF pathway. Non-limiting examples of delivery to the CSF pathway include intrathecal and intracerebroventricular administration. [0385] In some embodiments, the AAV particles may be delivered by direct (intraparenchymal) injection into the substance of an organ, e.g., one or more regions of the brain. [0386] In some embodiments, the AAV particles may be delivered by subpial injection into the spinal cord. For example, subjects may be placed into a spinal immobilization apparatus. A dorsal laminectomy may be performed to expose the spinal cord. Guiding tubes and XYZ manipulators may be used to assist catheter placement. Subpial catheters may be placed into the subpial space by advancing the catheter from the guiding tube and AAV particles may be injected through the catheter (Miyanohara et al., Mol Ther Methods Clin Dev.2016; 3: 16046). In some cases, the AAV particles may be injected into the cervical subpial space. In some cases, the AAV particles may be injected into the thoracic subpial space. [0387] In some embodiments, the present disclosure provides delivery by intravenous injection of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0388] In some embodiments, the present disclosure provides delivery by intravenous injection of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 739. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0389] In some embodiments, the present disclosure provides delivery by intravenous injection of an adeno-associated virus (AAV) particle to a subject for treating a disorder associated with FXN deficiency such as FA, wherein the AAV particle comprises a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 738. In some embodiments, the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. [0390] In some embodiments, the AAV particles may be delivered by direct injection to the CNS of a subject. In some embodiments, direct injection is intracerebral injection, intraparenchymal injection, intrathecal injection, intra-cisterna magna injection, or any combination thereof. In some embodiments, direct injection to the CNS of a subject comprises convection enhanced delivery (CED). In some
Attorney Docket No.14640.0092-00304 embodiments, administration comprises peripheral injection. In some embodiments, peripheral injection is intravenous injection. [0391] In some embodiments, the AAV particles may be delivered to a subject in order to increase a FXN protein level in the CNS (e.g., the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) as compared to a baseline level in the subject. The increase may be 0.1x to 5x, 0.5x to 5x, 1x to 5x, 2x to 5x, 3x to 5x, 4x to 5x, 0.1x to 4x, 0.5x to 4x, 1x to 4x, 2x to 4x, 3x to 4x, 0.1x to 3x, 0.5x to 3x, 1x to 3x, 2x to 3x, 0.1x to 2x, 0.5x to 2x, 0.1x to 1x, 0.5x to 1x, 0.1x to 0.5x, 1x to 2x, 0.1x, 0.2x, 0.3x, 0.4x, 0.5x, 0.6x, 0.7x, 0.8x, 0.9x, 1.0x, 1.1x, 1.2x, 1.3x, 1.4x, 1.5x, 1.6x, 1.7x, 1.8x, 1.9x, 2.0x, 2.1x, 2.2x, 2.3x, 2.4x, 2.5x, 2.6x, 2.7x, 2.8x, 2.9x, 3.0x, 3.1x, 3.2x, 3.3x, 3.4x, 3.5x, 3.6x, 3.7x, 3.8x, 3.9x, 4.0x, 4.1x, 4.2x, 4.3x, 4.4x, 4.5x, 4.6x, 4.7x, 4.8x, 4.9x or more than 5x as compared to a baseline level. In some embodiments, the increase may be 0.5x-3x as compared to a baseline level. [0392] In some embodiments, the AAV particles may be delivered to a subject in order to increase a FXN protein level in the CNS (e.g., the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) by transducing cells in these CNS regions. Transduction may also be referred to as the number of cells that are positive for FXN protein. The transduction may be greater than or equal to 1%, greater than or equal to 5%, greater than or equal to 10%, greater than or equal to 15%, greater than or equal to 20%, greater than or equal to 25%, greater than or equal to 30%, greater than or equal to 35%, greater than or equal to 40%, greater than or equal to 45%, greater than or equal to 50%, greater than or equal to 55%, greater than or equal to 60%, greater than or equal to 65%, greater than or equal to 70%, greater than or equal to 75%, greater than or equal to 80%, greater than or equal to 85%, greater than or equal to 90%, greater than or equal to 95%, or greater than or equal to 99% of cells in these CNS regions. [0393] In some embodiments, delivery of AAV particles comprising a viral genome encoding FXN as described herein to neurons in the brain (e.g., neurons in the temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) may lead to an increased expression of FXN protein in one or more of those neurons. In some embodiments, the increased FXN protein expression may lead to improved survival and/or function of various cell types in these CNS regions and/or improvement of at least one symptom of Friedreich’s Ataxia. [0394] In particular embodiments, the AAV particles may be delivered to a subject in order to establish widespread distribution of the FXN throughout the CNS by administering the AAV particles to the thalamus of the subject.
Attorney Docket No.14640.0092-00304 [0395] In some embodiments, the increased expression of FXN protein may lead to improved gait, improved ataxia-associated heart conditions, decreased feeling of exhaustion, improved symptoms relating to metabolic disorders such as diabetes, and/or improved quality of life. Administration [0396] In some embodiments, the present disclosure provides methods comprising administering AAV particles in accordance with the disclosure to a subject in need thereof. AAV particle pharmaceutical, diagnostic, or prophylactic compositions thereof, may be administered to a subject using any amount and any route of administration effective for treating, or diagnosing a disease, disorder, and/or condition associated with decreased FXN expression or FXN deficiency. In some embodiments, the disease, disorder, and/or condition is FA. [0397] Compositions in accordance with the disclosure may be formulated in unit dosage form for ease of administration and uniformity of dosage. It will be understood, however, that the total daily usage of the compositions of the present disclosure may be decided by the attending physician within the scope of sound medical judgment. The specific therapeutically effective, prophylactically effective, or appropriate imaging dose level for any particular patient will depend upon a variety of factors including the disorder being treated and the severity of the disorder; the activity of the specific compound employed; the specific composition employed; the age, body weight, general health, sex, and diet of the patient; the time of administration, route of administration, and rate of excretion of the specific FXN employed; the duration of the treatment; drugs used in combination or coincidental with the specific compound employed; and like factors well known in the medical arts. [0398] In certain embodiments, the desired dosage may be delivered using multiple administrations (e.g., two, three, four, five, six, seven, eight, nine, ten, eleven, twelve, thirteen, fourteen, or more administrations). When multiple administrations are employed, split dosing regimens such as those described herein may be used. As used herein, a “split dose” is the division of single unit dose or total daily dose into two or more doses, e.g., two or more administrations of the single unit dose. As used herein, a “single unit dose” is a dose of any therapeutic composition administered in one dose/at one time/single route/single point of contact, i.e., single administration event. In some embodiments, a single unit dose is provided as a discrete dosage form (e.g., a tablet, capsule, patch, loaded syringe, vial, etc.). As used herein, a “total daily dose” is an amount given or prescribed in 24-hour period. It may be administered as a single unit dose. The AAV particles may be formulated in buffer only or in a formulation described herein. [0399] In some embodiments, a pharmaceutical composition described herein can be formulated into a topical, intranasal, pulmonary, intratracheal, or injectable dosage form. In some embodiments, a pharmaceutical composition described herein can be formulated in a dosage form suitable for intravenous, intraocular, intravitreal, intramuscular, intracardiac, intraperitoneal, and/or subcutaneous administration.
Attorney Docket No.14640.0092-00304 [0400] In some embodiments, delivery of the AAV particles described herein results in minimal serious adverse events (SAEs) as a result of the delivery of the AAV particles. Combinations [0401] The AAV particles may be used in combination with one or more other therapeutic, prophylactic, diagnostic, or imaging agents. The phrase “in combination with,” is not intended to require that the agents must be administered at the same time and/or formulated for delivery together, although these methods of delivery are within the scope of the present disclosure. Compositions can be administered concurrently with, prior to, or subsequent to, one or more other desired therapeutics or medical procedures. In general, each agent will be administered at a dose and/or on a time schedule determined for that agent. In some embodiments, the present disclosure encompasses the delivery of pharmaceutical, prophylactic, diagnostic, or imaging compositions in combination with agents that may improve their bioavailability, reduce and/or modify their metabolism, and/or modify their distribution within the body. [0402] The therapeutic agents may be approved by the US Food and Drug Administration or may be in clinical trial or at the preclinical research stage. The therapeutic agents may utilize any therapeutic modality known in the art, with non-limiting examples including gene silencing or interference (i.e., miRNA, siRNA, RNAi, shRNA), gene editing (e.g., TALEN, CRISPR/Cas9 systems, zinc finger nucleases), and gene, protein, or enzyme replacement. [0403] In some embodiments, an AAV particle described herein, or a pharmaceutical composition comprising the AAV particle, may be administered in combination with at least one additional therapeutic agent and/or therapy. In some embodiments, the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy for treating the disorder associated with FXN deficiency (e.g., Friedreich’s Ataxia). In some embodiments, the at least one additional therapeutic agent and/or therapy comprises omaveloxolone or idebenone. In some embodiments, the at least one additional therapeutic agent and/or therapy comprises CoQ10, IFN gamma, or a drug to treat comorbidities including diabetes, heart disease, pain. [0404] In some embodiments, the at least one additional therapeutic agent and/or therapy comprises an immunosuppressant. In some embodiments, the immunosuppressant may be administered to the subject prior to administration of an AAV particle or pharmaceutical composition described herein. In some embodiments, the immunosuppressant may be administered to the subject simultaneously with administration of an AAV particle or pharmaceutical composition described herein. In some embodiments, the immunosuppressant may be administered to the subject after administration of an AAV particle or pharmaceutical composition described herein. In some embodiments, the AAV particle or pharmaceutical composition is administered to a subject who is receiving or has received an immunosuppressant. In some embodiments, the immunosuppressant comprises a corticosteroid (for example, and without limitation, prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine.
Attorney Docket No.14640.0092-00304 Measurement of Expression [0405] Expression of FXN from viral genomes may be determined using various methods known in the art such as, but not limited to immunochemistry (e.g., IHC), enzyme-linked immunosorbent assay (ELISA), affinity ELISA, ELISPOT, flow cytometry, immunocytology, surface plasmon resonance analysis, kinetic exclusion assay, liquid chromatography-mass spectrometry (LCMS), high-performance liquid chromatography (HPLC), BCA assay, immunoelectrophoresis, Western blot, SDS-PAGE, protein immunoprecipitation, PCR, and/or in situ hybridization (ISH). In some embodiments, transgenes encoding FXN delivered in different AAV capsids may have different expression levels in dorsal root ganglion (DRG). [0406] In certain embodiments, the FXN protein is detectable by an enzyme-linked immunosorbent assay (ELISA). [0407] In certain embodiments, the FXN protein is detectable by an immunohistochemistry assay. [0408] In certain embodiments, the FXN protein is detectable by Western blot. [0409] In certain embodiments, the FXN protein is detectable by a frataxin biofluid assay, such as the assay described in PCT/US2020/045687, the contents of which are hereby incorporated in their entirety. [0410] In certain embodiments, a FXN gene, mRNA, and/or protein expression is measured in a cell or tissue of a subject who is receiving or has received an AAV particle described herein. In certain embodiments, the FXN gene, mRNA, and/or protein expression is measured in a cell or tissue of the CNS, such as the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum. In certain embodiments, the FXN gene, mRNA, and/or protein expression is measured in a peripheral cell or tissue, such as the liver, heart, and/or muscle. VII. Kits and Devices Kits [0411] In some aspects, the present disclosure provides a variety of kits for conveniently and/or effectively carrying out methods of the present disclosure. Typically, kits will comprise sufficient amounts and/or numbers of components to allow a user to perform multiple treatments of a subject(s) and/or to perform multiple experiments. [0412] Any of the vectors, constructs, or FXN of the present disclosure may be comprised in a kit. In some embodiments, kits may further include reagents and/or instructions for creating and/or synthesizing compounds and/or compositions of the present disclosure. In some embodiments, kits may also include one or more buffers. In some embodiments, kits of the disclosure may include components for making protein or nucleic acid arrays or libraries and thus, may include, for example, solid supports. [0413] In some embodiments, kit components may be packaged either in aqueous media or in lyophilized form. The container means of the kits will generally include at least one vial, test tube, flask, bottle, syringe or other container means, into which a component may be placed, and suitably aliquoted. Where there is more than one kit component, (labeling reagent and label may be packaged together), kits
Attorney Docket No.14640.0092-00304 may also generally contain second, third or other additional containers into which additional components may be separately placed. In some embodiments, kits may also comprise second container means for containing sterile, pharmaceutically acceptable buffers and/or other diluents. In some embodiments, various combinations of components may be comprised in one or more vial. Kits of the present disclosure may also typically include means for containing compounds and/or compositions of the present disclosure, e.g., proteins, nucleic acids, and any other reagent containers in close confinement for commercial sale. Such containers may include injection or blow-molded plastic containers into which desired vials are retained. [0414] In some embodiments, kit components are provided in one and/or more liquid solutions. In some embodiments, liquid solutions are aqueous solutions, with sterile aqueous solutions being particularly used. In some embodiments, kit components may be provided as dried powder(s). When reagents and/or components are provided as dry powders, such powders may be reconstituted by the addition of suitable volumes of solvent. In some embodiments, it is envisioned that solvents may also be provided in another container means. In some embodiments, labeling dyes are provided as dried powders. In some embodiments, it is contemplated that 10, 20, 30, 40, 50, 60, 70, 80, 90, 100, 120, 120, 130, 140, 150, 160, 170, 180, 190, 200, 300, 400, 500, 600, 700, 800, 900, 1000 micrograms or at least or at most those amounts of dried dye are provided in kits of the disclosure. In such embodiments, dye may then be resuspended in any suitable solvent, such as DMSO. [0415] In some embodiments, kits may include instructions for employing kit components as well the use of any other reagent not included in the kit. Instructions may include variations that may be implemented. Devices [0416] In some embodiments, compounds and/or compositions of the present disclosure may be combined with, coated onto or embedded in a device. Devices may include, but are not limited to, dental implants, stents, bone replacements, artificial joints, valves, pacemakers and/or other implantable therapeutic device. [0417] The present disclosure provides for devices which may incorporate viral vectors that encode one or more FXN molecules. These devices contain in a stable formulation the viral vectors which may be immediately delivered to a subject in need thereof, such as a human patient. [0418] Devices for administration may be employed to deliver the viral vectors encoding FXN of the present disclosure according to single, multi- or split-dosing regimens taught herein. [0419] Method and devices known in the art for multi-administration to cells, organs and tissues are contemplated for use in conjunction with the methods and compositions disclosed herein as embodiments of the present disclosure. VIII. Definitions [0420] At various places in the present specification, substituents of compounds of the present disclosure are disclosed in groups or in ranges. It is specifically intended that the present disclosure
Attorney Docket No.14640.0092-00304 include each and every individual sub-combination of the members of such groups and ranges. The following is a non-limiting list of term definitions. [0421] Unless defined otherwise, all technical and scientific terms used herein have the same meaning as commonly understood by one of ordinary skill in the art to which the invention pertains. [0422] The articles “a,” “an,” and “the” may mean one or more than one unless indicated to the contrary or otherwise evident from the context. Claims or descriptions that include “or” between one or more members of a group are considered satisfied if one, more than one, or all of the group members are present in, employed in, or otherwise relevant to a given product or process unless indicated to the contrary or otherwise evident from the context. The disclosure includes embodiments in which exactly one member of the group is present in, employed in, or otherwise relevant to a given product or process. The disclosure includes embodiments in which more than one, or the entire group members are present in, employed in, or otherwise relevant to a given product or process. [0423] The term “comprising” is intended to be open and permits but does not require the inclusion of additional elements or steps. [0424] Where ranges are given, endpoints are included. Furthermore, it is to be understood that unless otherwise indicated or otherwise evident from the context and understanding of one of ordinary skill in the art, values that are expressed as ranges can assume any specific value or subrange within the stated ranges in different embodiments of the disclosure, to the tenth of the unit of the lower limit of the range, unless the context clearly dictates otherwise. [0425] Adeno-associated virus: As used herein, the term “adeno-associated virus” or “AAV” refers to members of the dependovirus genus or a functional variant thereof. Unless stated otherwise, “AAV” may refer to wildtype (i.e., naturally occurring) AAV or recombinant AAV. [0426] AAV Particle: As used herein, an “AAV particle” refers to a particle comprising an AAV capsid, e.g., an AAV capsid variant (such as a parent capsid sequence with at least one peptide insert and/or with at least one substitution), and a polynucleotide, e.g., a viral genome or a vector genome. The AAV particle may be capable of delivering a FXN polynucleotide to cells. The cells may be mammalian cells, e.g., human cells. In some embodiments, an AAV particle of the present disclosure may be produced recombinantly. In some embodiments, an AAV particle may be derived from any serotype, described herein or known in the art, including combinations of serotypes (e.g., “pseudotyped” AAV) or from various genomes (e.g., single stranded or self-complementary). In some embodiments, the AAV particle may be replication defective and/or targeted. In some embodiments, the AAV particle may comprise a peptide present in, e.g., inserted into and/or replacing a wildtype amino acid of, the capsid to enhance tropism for a desired target tissue. It is to be understood that reference to the AAV particle of the disclosure also includes pharmaceutical compositions thereof, even if not explicitly recited. [0427] Administering: As used herein, the term “administering” refers to providing a pharmaceutical agent or composition to a subject.
Attorney Docket No.14640.0092-00304 [0428] Amelioration: As used herein, the term “amelioration” or “ameliorating” refers to a lessening of severity of at least one indicator of a condition or disease. For example, in the context of a neurodegenerative disorder, amelioration includes the reduction or stabilization of neuron loss. [0429] Approximately: As used herein, the term “approximately” or “about,” as applied to one or more values of interest, refers to a value that is similar to, i.e., within 10% of, a stated reference value. [0430] Baseline: The term “baseline,” when used to describe a measurement in a subject receiving or about to receive a treatment, refers to a measurement made before starting the treatment. [0431] Capsid: As used herein, the term “capsid” refers to the exterior, e.g., a protein shell, of a virus particle, e.g., an AAV particle, that is substantially (e.g., >50%, >60%, >70%, >80%, >90%, >95%, >99%, or 100%) protein. In some embodiments, the capsid is an AAV capsid comprising an AAV capsid protein described herein, e.g., a VP1, VP2, and/or VP3 polypeptide. The AAV capsid protein can be a wild-type AAV capsid protein or a variant, e.g., a structural and/or functional variant from a wild-type or a reference capsid protein, referred to herein as an “AAV capsid variant.” For example, and without limitation, an AAV capsid variant may refer to at least a VP1 protein, a VP2 protein, or a VP3 protein (e.g., all of the VP1, VP2, and VP3 proteins forming the AAV capsid) as will be clear from context. In some embodiments, the AAV capsid variant described herein may comprise a peptide insertion and/or substitution (i.e., replacement). In some embodiments, the AAV capsid variant described herein has the ability to encapsulate a viral genome and/or is capable of entry into a cell, e.g., a mammalian cell. In some embodiments, the AAV capsid variant described herein may have modified tropism compared to that of a wild-type AAV capsid, e.g., the corresponding wild-type capsid. [0432] Cis-Elements: As used herein, cis-elements or the synonymous term “cis-regulatory elements” refer to regions of non-coding DNA which regulate the transcription of nearby genes. The Latin prefix “cis” translates to “on this side.” Cis-elements are found in the vicinity of the gene, or genes, they regulate. Examples of cis-elements include a Kozak sequence, SV40 introns, or a portion of the backbone. [0433] CNS structures: As used herein, “CNS structures” refers to structures of the central nervous system and sub-structures thereof. Non-limiting examples of structures in the spinal cord may include ventral horn, dorsal horn, white matter, and nervous system pathways or nuclei within. Non-limiting examples of structures in the brain include, forebrain, midbrain, hindbrain, diencephalon, telencephalon, myelencephalon, metencephalon, mesencephalon, prosencephalon, rhombencephalon, cortices, frontal lobe, parietal lobe, temporal lobe, occipital lobe, cerebrum, thalamus, hypothalamus, tectum, tegmentum, cerebellum, pons, medulla, amygdala, hippocampus, basal ganglia, corpus callosum, pituitary gland, putamen, striatum, ventricles and sub-structures thereof. [0434] CNS Cells: As used herein, “CNS cells” refers to cells of the central nervous system and sub- structures thereof. Non-limiting examples of CNS cells include neurons and sub-types thereof, glia, microglia, oligodendrocytes, ependymal cells and astrocytes. Non-limiting examples of neurons include sensory neurons, motor neurons, interneurons, unipolar cells, bipolar cells, multipolar cells,
Attorney Docket No.14640.0092-00304 pseudounipolar cells, pyramidal cells, basket cells, stellate cells, Purkinje cells, Betz cells, amacrine cells, granule cell, ovoid cell, medium aspiny neurons, large aspiny neurons, GABAergic neurons, and/or glutamatergic neurons. [0435] Codon optimization: As used herein, the term “codon optimization” refers to a process of changing codons of a given gene in such a manner that the polypeptide sequence encoded by the gene remains the same. [0436] Complementary and substantially complementary: As used herein, the term “complementary” refers to the ability of polynucleotides to form base pairs with one another. Perfect complementarity or 100% complementarity refers to the situation in which each nucleotide unit of one polynucleotide strand can form a hydrogen bond with a nucleotide unit of a second polynucleotide strand. Less than perfect complementarity refers to the situation in which some, but not all, nucleotide units of two strands can form hydrogen bond with each other. For example, for two 20-mers, if only two base pairs on each strand can form a hydrogen bond with each other, the polynucleotide strands exhibit 10% complementarity. In the same example, if 18 base pairs on each strand can form hydrogen bonds with each other, the polynucleotide strands exhibit 90% complementarity. The term “complementary” as used herein can encompass fully complementary or partially (e.g., substantially) complementary. “Fully complementary”, “perfect complementarity”, or “100% complementarity” refers to the situation in which each nucleotide unit of one polynucleotide or oligonucleotide strand can base-pair with a nucleotide unit of a second polynucleotide or oligonucleotide strand. As used herein, the term “substantially complementary” means that >50% of the nucleotide units of a first polynucleotide strand can base pair with nucleotide units on a second polynucleotide strand. When used in the context of RNA silencing, “substantially complementary” refers to an siRNA that has a sequence (e.g., in the antisense strand) that is sufficient to bind the desired target mRNA and to trigger the RNA silencing of the target mRNA. [0437] Conservative substitution: As used herein, a conservative substitution, as applied to an amino acid sequence, also referred to as a “conservative amino acid substitution,” is one in which the amino acid residue is replaced with an amino acid residue having similar biochemical properties. When used in reference to a nucleic acid sequence, the term “conservative substitution” refers to a nucleotide replacement that results in an amino acid residue having similar biochemical properties compared to a reference sequence. Families of amino acid residues having similar biochemical properties have been defined in the art. These families include amino acids with basic side chains (e.g., lysine, arginine, histidine), acidic side chains (e.g., aspartic acid, glutamic acid), uncharged polar side chains (e.g., glycine, asparagine, glutamine, serine, threonine, tyrosine, cysteine), nonpolar side chains (e.g., alanine, valine, leucine, isoleucine, proline, phenylalanine, methionine, tryptophan), beta-branched side chains (e.g., threonine, valine, isoleucine) and aromatic side chains (e.g., tyrosine, phenylalanine, tryptophan, histidine). [0438] Corresponding to: As used herein, the phrase “corresponding to” in the context of an amino acid sequence refers to the location of an amino acid in a reference sequence or the equivalent position in
Attorney Docket No.14640.0092-00304 a modified sequence when aligned. For example, an amino acid corresponding to position 577 of SEQ ID NO: 138 refers to the amino acid at the 577th position from the N-terminus in SEQ ID NO: 138 or the equivalent position in an aligned sequence. As used herein, an amino acid at a position corresponding to that in a designated sequence may also be referred to as an amino acid at a particular position, numbered according or numbered relative to the designated sequence. For instance, an amino acid corresponding to position 577 of SEQ ID NO: 138 may also be referred to as an amino acid at position 577, numbered according to SEQ ID NO: 138 or as numbered according to a sequence corresponding to SEQ ID NO: 138 (i.e., relative to a reference sequence of SEQ ID NO: 138). For further example, in some embodiments, a peptide (e.g., the peptide [N2]-[N3]) replaces an amino acid at a position corresponding to position 577 of SEQ ID NO: 138, i.e., said peptide (e.g., the peptide [N2]-[N3]) replaces position 577 numbered according to SEQ ID NO: 138. [0439] Derivative: As used herein, the term “derivative” refers to a composition (e.g., sequence, compound, formulation, etc.) that is derived from, or finds its basis in, a parent composition. Non- limiting examples of a parent composition include a wild-type or original amino acid or nucleic acid sequence, or an undiluted formulation. In some embodiments, a derivative is a variant of a parent composition. A derivative may differ from the parent composition by less than about 1%, less than about 5%, less than about 10%, less than about 15%, less than about 20%, less than about 25%, less than about 30%, less than about 35%, less than about 40%, less than about 45%, or less than about 50%. In certain embodiments, a derivative may differ from a parent composition by more than about 50%. In certain embodiments, a derivative may differ from a parent composition by more than about 75%. In some embodiments, a derivative may be a fragment or truncation of a parent amino acid or nucleotide sequence. As a non-limiting example, a derivative may be a sequence with a nucleotide, amino acid, or peptide substitution and/or insertion as compared to a parent nucleic acid or amino acid sequence (e.g., as compared to AAV5 or AAV9). [0440] Effective amount: As used herein, the term “effective amount” or “therapeutically effective amount” of an agent is that amount sufficient to effect beneficial or desired results. An effective amount is provided in a single dose or multiple doses to treat, improve symptoms of, delay progression of symptoms, diagnose, prevent, and/or delay the onset of the infection, disease, disorder, and/or condition. [0441] Excipient: As used herein, the term “excipient” refers to an inactive substance that serves as the vehicle or medium for an active pharmaceutical agent or other active substance. [0442] Formulation: As used herein, a “formulation” includes at least one active ingredient (e.g., an AAV particle) and at least one inactive ingredient (e.g., a pharmaceutically acceptable excipient). [0443] Fragment: A “fragment,” as used herein, refers to a contiguous portion of a reference sequence. A fragment may comprise a functional fragment that retains at least one activity of the reference sequence. For example, fragments of proteins may comprise polypeptides obtained by digesting full-length protein isolated from cultured cells. A fragment may also refer to a truncation (e.g., an N- terminal and/or C-terminal truncation) of a protein or a truncation (e.g., at the 5’ and/or 3’ end) of a
Attorney Docket No.14640.0092-00304 nucleic acid. A protein fragment may be obtained by expression of a truncated nucleic acid, such that the nucleic acid encodes a portion of the full-length protein. [0444] Healthy individual: As used herein, the term “healthy individual” refers to an individual who does not have a disease or disorder associated with FXN protein deficiency, e.g., an individual who does not have Friedreich's Ataxia. [0445] Identity: As used herein, the term “identity” refers to the overall relatedness between polymeric molecules, e.g., between oligonucleotide molecules (e.g., DNA molecules and/or RNA molecules) and/or between polypeptide molecules. Calculation of the percent identity of two polynucleotide sequences, for example, may be performed by aligning the two sequences for optimal comparison purposes (e.g., gaps can be introduced in one or both of a first and a second nucleic acid sequences for optimal alignment and non-identical sequences can be disregarded for comparison purposes). The nucleotides at corresponding nucleotide positions are then compared. When a position in the first sequence is occupied by the same nucleotide as the corresponding position in the second sequence, then the molecules are identical at that position. The percent identity between the two sequences is a function of the number of identical positions shared by the sequences, taking into account the number of gaps, and the length of each gap, which needs to be introduced for optimal alignment of the two sequences. The comparison of sequences and determination of percent identity between two sequences can be accomplished using a mathematical algorithm. For example, the percent identity between two nucleotide sequences can be determined using methods such as those described in Computational Molecular Biology, Lesk, A. M., ed., Oxford University Press, New York, 1988; Biocomputing: Informatics and Genome Projects, Smith, D. W., ed., Academic Press, New York, 1993; Sequence Analysis in Molecular Biology, von Heinje, G., Academic Press, 1987; Computer Analysis of Sequence Data, Part I, Griffin, A. M., and Griffin, H. G., eds., Humana Press, New Jersey, 1994; and Sequence Analysis Primer, Gribskov, M. and Devereux, J., eds., M Stockton Press, New York, 1991; each of which is incorporated herein by reference in its entirety. For example, the percent identity between two nucleotide sequences can be determined using the algorithm of Myers and Miller (CABIOS, 1989, 4:11-17), which has been incorporated into the ALIGN program (version 2.0) using a PAM120 weight residue table, a gap length penalty of 12 and a gap penalty of 4. The percent identity between two nucleotide sequences can, alternatively, be determined using the GAP program in the GCG software package using an NWSgapdna.CMP matrix. Methods commonly employed to determine percent identity between sequences include, but are not limited to those disclosed in Carillo, H., and Lipman, D., SIAM J Applied Math., 48:1073 (1988); incorporated herein by reference in its entirety. Techniques for determining identity are codified in publicly available computer programs. Computer software to determine homology between two sequences include, but are not limited to, GCG program package, Devereux, J., et al., Nucleic Acids Research, 12(1), 387 (1984)), the Basic Local Alignment Search Tool (BLAST, which includes, e.g., BLASTP for protein sequences and BLASTN for nucleic acid sequences), and FASTA Altschul, S. F. et al., J. Molecular Biol., 215, 403 (1990)), EMBOSS Needle, Clustal
Attorney Docket No.14640.0092-00304 Omega, Benchling, and Geneious. In preferred embodiments, sequence identity may be determined using BLAST, Clustal Omega, or EMBOSS Needle. [0446] Inverted terminal repeat: As used herein, the term “inverted terminal repeat” or “ITR” refers to a cis-regulatory element for the packaging of polynucleotide sequences into viral capsids. [0447] Isolated: As used herein, the term “isolated” refers to a substance or entity that is altered or removed from the natural state, e.g., altered or removed from at least some of component with which it is associated in the natural state. For example, a nucleic acid or a peptide naturally present in a living animal is not “isolated,” but the same nucleic acid or peptide partially or completely separated from the coexisting materials of its natural state is “isolated.” An isolated nucleic acid or protein can exist in substantially purified form, or can exist in a non-native environment such as, for example, a host cell. Such polynucleotides could be part of a vector and/or such polynucleotides or polypeptides could be part of a composition, and still be isolated in that such vector or composition is not part of the environment in which it is found in nature. In some embodiments, an isolated nucleic acid is recombinant, e.g., incorporated into a vector. [0448] miRNA binding site: As used herein, a “miRNA binding site” or “miR binding site” refers either to a DNA sequence corresponding to an RNA sequence that is bound by a microRNA, or to the RNA sequence that is bound by the microRNA. The miR binding site is capable of binding, or binds, in whole or in part to a microRNA (miRNA, miR) through complete or partial hybridization. A miR binding site may be encoded or transcribed in series, also referred to as a “miR binding site series” or “miR BSs”, which includes two or more miR binding sites having the same or a different nucleic acid sequence. [0449] Modification: As used herein, the term “modification” or “modified” refers to any substance, compound, or molecule that has been changed in any way. For example, a modification in an amino acid sequence may comprise a substitution (e.g., a conservative substitution), an insertion, and/or a deletion of one or more amino acids in the sequence. [0450] Neurological disease: As used herein, a “neurological disease” is any disease associated with the central or peripheral nervous system and components thereof (e.g., neurons). [0451] Operably linked: As used herein, the phrase “operably linked” refers to a functional connection between two or more molecules, constructs, transcripts, entities, moieties or the like. [0452] Payload: As used herein, “payload,” “payload sequence,” or “payload region” refers to one or more polynucleotides or polynucleotide regions encoded by or within a viral genome or an expression product of such polynucleotide or polynucleotide region, e.g., a transgene, a polynucleotide encoding a polypeptide. [0453] Payload construct: As used herein, “payload construct” is one or more polynucleotide regions encoding or comprising a payload that is flanked on one or both sides by an inverted terminal repeat (ITR) sequence. The payload construct is a template that is replicated in a viral production cell to produce a viral genome.
Attorney Docket No.14640.0092-00304 [0454] Payload construct vector: As used herein, “payload construct vector” is a vector encoding or comprising a payload construct, and regulatory regions for replication and expression in bacterial cells. The payload construct vector may also comprise a component for viral expression in a viral replication cell. [0455] Pharmaceutically acceptable: The phrase “pharmaceutically acceptable” is employed herein to refer to those compounds, materials, compositions, and/or dosage forms which are suitable for use in contact with the tissues of human beings and animals. [0456] Pharmaceutically acceptable excipients: As used herein, the term “pharmaceutically acceptable excipient,” as used herein, refers to any ingredient other than active agents (e.g., as described herein) present in pharmaceutical compositions that can function as vehicles for suspending and/or dissolving active agents. [0457] Pharmaceutically acceptable salts: Pharmaceutically acceptable salts of the compounds described herein are forms of the disclosed compounds wherein the acid or base moiety is in its salt form (e.g., as generated by reacting a free base group with a suitable organic acid). Examples of pharmaceutically acceptable salts include, but are not limited to, mineral or organic acid salts of basic residues such as amines; alkali or organic salts of acidic residues such as carboxylic acids; and the like. [0458] Pharmaceutical Composition: As used herein, the term “pharmaceutical composition” or pharmaceutically acceptable composition” comprises AAV polynucleotides, AAV genomes, or AAV particle and one or more pharmaceutically acceptable excipients, solvents, adjuvants, and/or the like. [0459] Position: The term “position,” as used herein in the context of an amino acid sequence, refers to the location of a particular amino acid or set of amino acids relative to a larger sequence. A position or positions of amino acids may interchangeably be referred to by an amino acid number or numbers of a reference sequence. For example, and unless otherwise specified, “positions 1-731, as numbered according to SEQ ID NO: 982” is interchangeable with “amino acids 1-731, as numbered according to SEQ ID NO: 982.” Within a sequence, an amino acid position is counted from the N-terminus. [0460] Preventing: As used herein, the term “preventing” refers to partially or completely delaying onset of an infection, disease, disorder and/or condition; partially or completely delaying onset of one or more symptoms, features, or clinical manifestations of a particular infection, disease, disorder, and/or condition; partially or completely delaying onset of one or more symptoms, features, or manifestations of a particular infection, disease, disorder, and/or condition; partially or completely delaying progression from an infection, a particular disease, disorder and/or condition; and/or decreasing the risk of developing pathology associated with the infection, the disease, disorder, and/or condition. The term “prevention” or “preventing” of an infection, disease, disorder and/or condition may be considered a subset within the meaning with the term “treatment” or “treating” of the infection, disease, disorder and/or condition. [0461] Region: As used herein, the term “region” refers to a zone or general area. In some embodiments, when referring to a protein or protein module, a region may comprise a linear sequence of amino acids along the protein or protein module or may comprise a three-dimensional area. In some
Attorney Docket No.14640.0092-00304 embodiments, regions comprise terminal regions. As used herein, the term “terminal region” refers to regions located at the ends or termini of a given agent. When referring to proteins, terminal regions may comprise N- and/or C-termini. N-termini refer to the end of a protein comprising an amino acid with a free amino group. C-termini refer to the end of a protein comprising an amino acid with a free carboxyl group. N- and/or C-terminal regions may comprise the N- and/or C-termini as well as surrounding amino acids. When referring to a polynucleotide, a region may comprise a linear sequence of nucleic acids along the polynucleotide or may comprise a three-dimensional area, secondary structure, or tertiary structure. In some embodiments, regions comprise terminal regions. As used herein, the term “terminal region” refers to regions located at the ends or termini of a given agent. When referring to polynucleotides, terminal regions may comprise 5’ and/or 3’ termini. [0462] Sample: As used herein, the term “sample” or “biological sample” refers to a subset of tissues, cells, nucleic acids, or a component or part of the body (e.g., a body fluid, including but not limited to blood, mucus, lymphatic fluid, synovial fluid, cerebrospinal fluid, saliva, amniotic fluid, amniotic cord blood, urine, vaginal fluid, and semen). [0463] Self-complementary AAV: As used herein, the term “self-complementary AAV” refers to an AAV comprising at least a protein capsid and a self-complementary viral genome. [0464] Serotype: As used herein, the term “serotype” refers to distinct variations in a capsid of an AAV based on surface antigens which allow epidemiologic classifications of the AAVs at the sub- species level. [0465] Silent Mutation: As used herein, a “silent mutation” or “silent substitution” refers to a nucleotide replacement that results in the same amino acid residue as a reference sequence. [0466] Signal Sequence: As used herein, the phrase “signal sequence” refers to a sequence which can direct the transport or localization. [0467] Similarity: As used herein, the term “similarity” refers to the overall relatedness between polymeric molecules, e.g., between polynucleotide molecules (e.g., DNA molecules and/or RNA molecules) and/or between polypeptide molecules. Calculation of percent similarity of polymeric molecules to one another can be performed in the same manner as a calculation of percent identity, except that calculation of percent similarity takes into account conservative substitutions as is understood in the art. [0468] Spacer: As used herein, a “spacer” is generally any selected nucleic acid sequence of, e.g., 1, 2, 3, 4, 5, 6, 7, 8, 9, or 10 nucleotides in length, which is located between two or more consecutive miR binding site sequences. In some embodiments, spacers may also be more than 10 nucleotides in length, e.g., 20, 30, 40, or 50 or more than 50 nucleotides. [0469] Subject: As used herein, the term “subject” or “patient” refers to any organism to which a composition in accordance with the disclosure may be administered, e.g., for experimental, diagnostic, prophylactic, and/or therapeutic purposes. Similarly, “subject” or “patient” refers to an organism who may seek, who may require, who is receiving, or who will receive treatment or who is under care by a
Attorney Docket No.14640.0092-00304 trained professional for a particular disease or condition. Typical subjects include animals (e.g., mammals such as mice, rats, rabbits, non-human primates, and humans). As used herein, a subject or patient may be susceptible to, suspected of having, or have a deficiency in frataxin protein, e.g., may be susceptible to, suspected of having, or have Friedreich’s Ataxia (FA). [0470] Substantially: As used herein, the term “substantially” refers to the qualitative condition of exhibiting total or near-total extent or degree of a characteristic or property of interest. One of ordinary skill in the biological arts will understand that biological and chemical phenomena rarely, if ever, go to completion and/or proceed to completeness or achieve or avoid an absolute result. The term “substantially” is therefore used herein to capture the potential lack of completeness inherent in many biological and chemical phenomena. [0471] Suffering from: An individual who is “suffering from” a disease, disorder, and/or condition has been diagnosed with or displays one or more symptoms of a disease, disorder, and/or condition. [0472] Susceptible to: An individual who is “susceptible to” a disease, disorder, and/or condition has not been diagnosed with and/or may not exhibit symptoms of the disease, disorder, and/or condition but harbors a propensity to develop a disease or its symptoms. In some embodiments, an individual who is susceptible to a disease, disorder, and/or condition may be characterized by one or more of the following: (1) a genetic mutation associated with development of the disease, disorder, and/or condition; (2) a genetic polymorphism associated with development of the disease, disorder, and/or condition; (3) increased and/or decreased expression and/or activity of a protein and/or nucleic acid associated with the disease, disorder, and/or condition; (4) habits and/or lifestyles associated with development of the disease, disorder, and/or condition; (5) a family history of the disease, disorder, and/or condition; and (6) exposure to and/or infection with a microbe associated with development of the disease, disorder, and/or condition. In some embodiments, an individual who is susceptible to a disease, disorder, and/or condition will develop the disease, disorder, and/or condition. In some embodiments, an individual who is susceptible to a disease, disorder, and/or condition will not develop the disease, disorder, and/or condition. [0473] Target Cells: As used herein, “target cells” refers to any one or more cells of interest. The cells may be found in vitro, in vivo, in situ or in the tissue or organ of an organism. The organism may be an animal, preferably a mammal, more preferably a human and most preferably a human patient. [0474] Target Tissue: As used herein, “target tissue” refers to a tissue of interest that may be found in vitro, in situ, or as part of an animal, preferably a mammal, more preferably a human and most preferably a human patient. [0475] Therapeutic Agent: The term “therapeutic agent” refers to any agent that, when administered to a subject, elicits a desired biological and/or pharmacological effect. [0476] Therapeutically Effective Outcome: As used herein, the term “therapeutically effective outcome” means an outcome that is sufficient in a subject suffering from or susceptible to an infection,
Attorney Docket No.14640.0092-00304 disease, disorder, and/or condition, to treat, improve symptoms of, delay progression of symptoms, diagnose, prevent, and/or delay the onset of the infection, disease, disorder, and/or condition. [0477] Treating: As used herein, the term “treating” refers to partially or completely alleviating, ameliorating, improving, relieving, delaying onset of, inhibiting progression of, reducing severity of, reducing incidence of, and/or preventing one or more symptoms or features of a particular infection, disease, disorder, and/or condition. Treatment may be administered to a subject who does not exhibit signs of a disease, disorder, and/or condition and/or to a subject who exhibits only early signs of a disease, disorder, and/or condition for the purpose of decreasing the risk of developing pathology associated with the disease, disorder, and/or condition. [0478] Truncated CBA promoter: As used herein, a "truncated CBA promoter" refers to a CBA promoter that is shorter in length than the nucleotide sequence of SEQ ID NO: 1734, provided below. SEQ ID NO: 1734
[0479] Truncated CMV promoter: As used herein, a “truncated CMV promoter” refers to a CMV promoter that is shorter in length than the nucleotide sequence of SEQ ID NO: 1743, as provided below. SEQ ID NO: 1743 gttgacattg attattgact agttattaat agtaatcaat tacggggtca ttagttcata gcccatatat ggagttccgc gttacataac ttacggtaaa tggcccgcct ggctgaccgc ccaacgaccc ccgcccattg acgtcaataa tgacgtatgt tcccatagta acgccaatag ggactttcca ttgacgtcaa tgggtggagt atttacggta aactgcccac ttggcagtac atcaagtgta tcatatgcca agtacgcccc ctattgacgt caatgacggt aaatggcccg cctggcatta tgcccagtac atgaccttat gggactttcc tacttggcag tacatctacg tattagtcat cgctattacc atggtgatgc ggttttggca gtacatcaat gggcgtggat agcggtttga ctcacgggga tttccaagtc tccaccccat tgacgtcaat gggagtttgt tttggcacca aaatcaacgg gactttccaa aatgtcgtaa caactccgcc ccattgacgc aaatgggcgg taggcgtgta cggtgggagg tctatataag cagagctc [0480] Unmodified: As used herein, “unmodified” refers to any substance, compound or molecule prior to being changed in any way. Unmodified may, but does not always, refer to the wild-type or native form of a biomolecule or entity. Molecules or entities may undergo a series of modifications whereby each modified product may serve as the “unmodified” starting molecule or entity for a subsequent modification. [0481] Variant: The term “variant” refers to a polypeptide or polynucleotide that has an amino acid or a nucleotide sequence that has at least 90% (e.g., at least 90%, at least 91%, at least 92%, at least 93%,
Attorney Docket No.14640.0092-00304 at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99%) sequence identity to a reference sequence. The variant may be a functional variant. As used herein, the term “functional variant” refers to a polypeptide variant or a polynucleotide variant that has at least one activity of the reference sequence. [0482] Vector: As used herein, a “vector” is any molecule or moiety which transports, transduces, or otherwise acts as a carrier of a heterologous molecule. Vectors of the present disclosure may be produced recombinantly and may be based on and/or may comprise adeno-associated virus (AAV) parent or reference sequence(s). [0483] Viral genome: As used herein, a “viral genome,” “vector genome,” or “VG” is a polynucleotide comprising at least one inverted terminal repeat (ITR) and at least one nucleic acid sequence encoding a payload. A viral genome encodes at least one copy of the payload. EXAMPLES [0484] The present disclosure is further illustrated by the following non-limiting examples. Example 1. High-throughput screen of TRACER AAV library in NHP and Rats [0485] A TRACER based method as described in WO 2020/072683, WO 2021/202651, and WO 2021/230987, the contents of which are herein incorporated by reference in their entirety, was used to generate the AAV capsid variants described herein. An orthogonal evolution approach was combined with high throughput screening by NGS. Briefly, the library of AAV capsid variants was generated using a mutagenesis approach, where sequences of 7 to 8 amino acids in length were inserted into different positions across loop VIII of AAV5, including between residues 570-584, relative to a reference sequence numbered according to SEQ ID NO: 138. The initial library was passed three times through non-human primates (NHP), specifically cynomolgus macaques (Macaca fascicularis), rats, or human brain microvascular endothelial cells (hBMVECs). Following the third passage in each system, 572 variants from the NHPs, 80 variants from the rats, and 99 variants from the hBMVECs were pooled into a passage 3 synthetic library of 747 total variants. This library was then passaged in NHPs and rats. After this passage (e.g., one-month post injection into two NHPs and the rats), RNA was extracted from three brain regions. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate fold enrichment relative to an AAV5 wild-type control in NHPs (Table 14, left column) and rats (Table 14, right column) and the peptides comprised within the variants were identified in both animals. Fold enrichment values above 1 indicate an increase in expression relative to AAV5. All libraries of AAV5 variants generated and passaged in the different hosts were under the control of the synapsin promoter. [0486] As shown in Table 14, approximately 288 variants were identified with an average fold change greater than wild-type AAV5 in the brain of NHPs. Of the 288 NHP variants, 27 demonstrated a fold-change of greater than 5 compared to wild-type, with 1 variant demonstrating a fold change of greater than 60. For instance, the variant comprising YPAEVVQK (SEQ ID NO: 943) demonstrated a 64.9-fold enrichment in the brain of NHPs.
Attorney Docket No.14640.0092-00304 [0487] As shown in Table 14, approximately 98 variants were identified with an average fold change greater than wild-type AAV5 in the brain of rats. Of the 98 variants, 33 demonstrated a fold-change of greater than 2 compared to wild-type, with one variant demonstrating a fold change of greater than 40. For instance, the variant comprising YPAEVVQK (SEQ ID NO: 943) demonstrated a 41.1-fold enrichment in the brain of rats. [0488] The variant comprising YPAEVVQK (SEQ ID NO: 943) which demonstrated a high fold enrichment in the brains of NHPs relative to wild-type AAV5 (64.9-fold enrichment), also demonstrated a high fold-change in the brains of rats (41.1-fold enrichment). This indicates that this AAV capsid variant comprising SEQ ID NO: 943 is able to cross species, as evidenced by increased expression and tropism in both the NHP and rat brain. Table 14. NGS fold-enrichment of AAV capsid variants in NHPs and rats
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
[0489] Taken together, these results demonstrate that after 3 rounds of screening of this AAV5 variant library with loop VIII modifications in NHP and rats, many AAV capsid variants outperformed the wild-type AAV5, for example, in penetration of the blood brain barrier (BBB) and expression in the brain. Also, capsid variants were identified that could infect both rats and NHPs, indicating cross-species tropism and compatibility. Example 2. Individual capsid characterization in NHPs, rats, and mice [0490] This Example describes the transduction level, tropism, ability to cross the blood brain barrier, and overall spatial distribution in the central nervous system (CNS) of an AAV capsid variant selected from the study described in Example 1, relative to wild-type AAV5, following intravenous injection in cynomolgus macaques (Macaca fascicularis), Norway rats, and BALB/c mice. The capsid variant was TTN-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 943 (encoded by SEQ ID NO: 944)), as outlined in Table 3 above. The amino acid and DNA sequence of TTN-002 is provided, e.g., in Tables 4A and 4B respectively. [0491] AAV particles were generated with this capsid variant encapsulating a luciferase-EGFP transgene or a cynomolgus monkey frataxin payload fused to a hemagglutinin (HA) tag driven by a chicken beta actin (CBA) heterologous promoter. Each capsid variant and control AAV5 and/or AAV9 capsids were tested by intravenously administering the AAV particle formulation at 5e13 VG/kg to NHPs (n=2), 1e13 VG per rat (n=3; 3e13 VG/kg), and/or 5e11 VG per mouse (n=3; 2e13 VG/kg). The in-life period was 28 days for NHPs and mice, and 25 days for rats. Various CNS and peripheral tissues were then collected for measuring transgene mRNA, transgene protein, and/or viral DNA (biodistribution). The AAV particles administered to the NHPs and rats comprised self-complementary viral genomes and the AAV particles administered to mice comprised a single-stranded viral genome.
Attorney Docket No.14640.0092-00304 A. Individual capsid characterization in NHPs [0492] The brains, spinal cord, and peripheral tissues including the heart, liver, and quadriceps, were isolated from NHPs (cynomolgus macaques (Macaca fascicularis)) at 28 days post intravenous administration of the AAV particles comprising the TTN-002 capsid variant and were assayed by qPCR for the presence of transgene RNA as a measure of transgene expression and compared to the AAV9 control. Data were provided as average mRNA fold change for the transgene relative to a housekeeping gene as well as the fold change relative to the AAV9 control (Table 15). As shown in Table 15, mRNA transgene expression from the TTN-002 capsid variant, which is an AAV5 capsid variant, was significantly higher in the brain of NHPs relative to the wild-type AAV9 control. More specifically, mRNA expression was approximately 20-25-fold higher from the TTN-002 capsid variant compared to wild-type AAV9 in the brain of NHPs. Additionally, mRNA expression was approximately 4-5-fold higher from the TTN-002 capsid variants compared to wild-type AAV9 in the spinal cord of the NHPs. TTN-002 also demonstrated lower mRNA expression in the liver and DRG relative the AAV9 control (Table 15). [0493] The brains, spinal cord, and peripheral tissues isolated from the NHPs were also assayed for the presence of viral DNA as a measure of viral genome levels. Data are provided in Table 16 as average DNA (viral genome (VG)) copies per diploid genome as well as fold change relative to the AAV9 control. As shown in Table 16, biodistribution of the AAV5 capsid variant, TTN-002, was significantly higher in the NHP brain relative to the wild-type AAV9 control. Biodistribution of TTN-002 was lower in the NHP liver relative to the wild-type AAV9 control. [0494] The brain tissues and spinal cords of the NHPs were also subjected to immunohistochemistry staining to evaluate overall CNS tropism and biodistribution in various regions (FIGs.1A-1D). Immunohistochemical staining correlated with the qPCR analysis, as TTN-002 showed significantly stronger staining and payload expression in the brain (e.g., across the entire cerebrum and cerebellum, FIGs.1A-1C) and spinal cord (FIGs.1A and 1D), as compared to the AAV9 control. More specifically, TTN-002 demonstrated localization, strong payload expression, and transduction in both neurons and glial cells in the temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and cerebellum (FIGs.1A-1C). Payload expression was also observed in astrocytes in the dentate nucleus. Additionally, quantification of co-expression of the payload and the pan-neuronal marker SMI- 311 showed payload expression in approximately 73.4% of the large neurons in the dentate nucleus of the deep cerebral nucleus in the cerebellum at 28-days post intravenous administration the AAV particles comprising the TTN-002 capsid variant. In the spinal cord, TTN-002 demonstrated localization, strong payload expression, and transduction in the cervical region (e.g., C2), thoracic region (e.g., T10), and lumbar region (e.g., L2) (FIGs.1A and 1D). Additionally, TTN-002 showed less staining in the DRG relative to the wild-type AAV9 control (approximately 2-fold less) (FIG.1D). Both TTN-002 and AAV9 appeared to transduce the liver and heart with similarly high efficiency, by IHC analysis (FIG.1D).
Attorney Docket No.14640.0092-00304 Additionally, the histopathology of these samples isolated from the NHPs showed no signs of toxicity in the NHPs, following intravenous administration of AAV particles comprising the TTN-002 capsid variant at a dose of 5e13 VG/kg with a self-complementary viral genome. Table 15. Transgene mRNA expression with the TTN-002 capsid variant in NHPs
Table 16. Viral DNA biodistribution with the TTN-002 capsid variant in NHPs
Attorney Docket No.14640.0092-00304
B. Individual capsid characterization in rats [0495] The brains and spinal cords were isolated from rats at 25 days post intravenous administration of AAV particles comprising the TTN-002 capsid variant (AAV5 variant) and assayed for the presence of transgene RNA as a measure of transgene expression, relative to a wild-type AAV5 control capsid or a wild-type AAV9 control capsid (Table 17). Data were provided as average mRNA fold change for the transgene relative to a housekeeping gene as well as the fold change relative to the AAV5 and AAV9 controls (Table 17). As shown in Table 17, mRNA transgene expression from the TTN-002 capsid variant was higher in both the brains and spinal cords relative to the AAV5 and AAV9 controls. More specifically, transgene mRNA expression was approximately 40- to 67-fold higher from the TTN-002 variant in the rat brain and spinal cord regions (cervical, thoracic, and lumbar) compared to wild-type AAV5 and transgene mRNA expression was approximately 5-7-fold higher in the rat brain and spinal cord regions (cervical, thoracic and lumbar) compared to wild-type AAV9. [0496] The brain and spinal cord tissues, as well as the heart peripheral tissue were subject to immunohistochemistry staining to evaluate overall tropism and biodistribution. Immunohistochemical staining correlated with the qPCR analysis, as TTN-002 showed increased staining relative to both AAV9 and AAV5 in the cortex, hippocampus, cerebellum, and spinal cord of the rat. TTN-002 showed increased staining in the heart of the rat relative to AAV5 but decreased staining relative to AAV9. Table 17. Transgene mRNA expression with TTN-002 capsid variant in rats
C. Individual capsid characterization in mice [0497] The brains and livers were isolated from BALB/c mice 28 days post intravenous injection of following intravenous administration of AAV particles comprising the TTN-002 capsid variant and were assayed by qPCR for the presence of transgene RNA as a measure of transgene expression and compared to an AAV9 and AAV5 control. Data were provided as average mRNA fold change for the transgene
Attorney Docket No.14640.0092-00304 relative to a housekeeping gene (Table 18) and as fold change in transgene mRNA expression relative AAV9 and AAV5 controls (Table 19). As shown in Table 18 and Table 19, the AAV5 capsid variant TTN-002 demonstrated similar levels of transgene expression relative to AAV9 in the brain and higher expression than wild-type AAV5. Transgene mRNA expression in the mouse brain was 265.9-fold higher with the TTN-002 capsid variant as compared to wild-type AAV5 (Table 19). Additionally, wild-type AAV5 and the AAV5 capsid variant, TTN-002, both resulted in lower transgene expression in the liver, as compared to wild-type AAV9. [0498] The brains and livers isolated from the mice were also assayed for the presence of viral DNA as a measure of viral genome levels. Data are provided in Table 18 as average DNA (viral genome (VG)) copies per diploid genome and in Table 19 as fold change in DNA copies per diploid genome relative AAV9 and AAV5 controls. The AAV5 capsid variant TTN-002 demonstrated comparable biodistribution relative to AAV9 in the mouse brain and increased biodistribution and viral genome levels than wild-type AAV5. More specifically, in the brain, the TTN-002 capsid variant led to 9-fold higher DNA (viral genome (VG)) copies per diploid genome relative to the AAV5 control (Table 19). Furthermore, wild- type AAV5 and the AAV5 capsid variant, TTN-002, resulted in decreased biodistribution and DNA (viral genome (VG)) copies per diploid genome in the liver relative to AAV9 (Table 19). Table 18. Transgene mRNA expression with the TTN-002 capsid variant in mice
Attorney Docket No.14640.0092-00304 Table 19. Fold-change in transgene mRNA expression and DNA copies per diploid genome relative to AAV9 and AAV5 in the brain and liver of mice
D. Individual capsid characterization in Marmosets (Callithrix jacchus) [0499] AAV particles were generated with the TTN-002 capsid variant, the AAV5 capsid control, or the AAV9 capsid control which comprised a self-complementary viral genome encoding a histone H2b protein with an HPC4 tag (TTN-002 capsid variant), T7 tag (AAV5 capsid control), or HA tag (AAV9 control capsid) driven by a ubiquitous CAG promoter. The AAV particles comprising the TTN-002 capsid variant, the AAV5 capsid control, or the AAV9 capsid control were administered to the marmosets (Callithrix jacchus) (n=3) intravenously in a single solution, at the doses indicated in Table 20. The in-life period was 28 days and then various CNS and peripheral tissues were collected for measuring transgene mRNA (expression) by RT-qPCR, protein expression by IHC, and viral DNA (biodistribution) by ddPCR. Data were then normalized to the dose of each viral vector in the dosing solution. Table 20. Titer of the AAV particles comprising the various capsids in solution dosed in marmosets
[0500] Table 21 shows the quantification of viral genome copies per diploid genome (biodistribution) by ddPCR following intravenous administration of AAV particles comprising a TTN- 002 capsid, which is normalized to the actual titer of the viral vector in the dosing solution (vg/dg = viral genome copies/ diploid genome). Table 22 shows the quantification of transgene mRNA by RT-qPCR following intravenous administration of AAV particles comprising a TTN-002 capsid, normalized to the actual titer of the viral vector in the dosing solution (mRNA = transgene mRNA fold over housekeeping gene; rel. to AAV9= transgene mRNA fold over housekeeping gene relative to AAV9; rel. to AAV5 transgene mRNA fold over housekeeping gene relative to AAV5). [0501] As shown in Table 21, the TTN-002 capsid variant, an AAV5 capsid variant, demonstrated increased biodistribution in the caudate and motor cortex in the brain of the marmosets relative to the AAV5 capsid control, as well as the AAV9 control. The TTN-002 capsid variant also demonstrated
Attorney Docket No.14640.0092-00304 increased transgene expression relative to AAV5 in the caudate and motor cortex, increased transgene expression relative to AAV9 in the caudate, and comparable transgene expression relative to AAV9 in the motor cortex (Tables 21 and 22). Similar expression and distribution was observed by immunohistochemistry. More specifically, staining for TTN-002 was detected in the caudate, putamen, thalamus, and cerebellum, and this staining was increased relative to AAV5. Staining for TTN-002 was also observed in the molecular and granule layer of the cerebellum. [0502] Biodistribution and transgene expression were also measured in the peripheral tissues of the liver, heart, and quadriceps, as shown in Table 21 and Table 22, respectively. In the liver, the TTN-002 capsid variant exhibited lower biodistribution (Table 21) and transgene expression (Table 22) relative to AAV5 and AAV9, indicating that the TTN-002 capsid variant was detargeted in the liver relative to AAV5 and AAV9 in marmosets. TTN-002 led to increased biodistribution (Table 21) in the heart relative to AAV5 and AAV9, increased transgene expression in the heart relative to AAV5, and decreased transgene expression in the heart relative to AAV9 (Table 22). In the muscle, the TTN-002 capsid variant exhibited higher biodistribution (Table 21) and transgene expression (Table 22) relative to AAV9. The TTN-002 capsid variant also demonstrated decreased biodistribution (Table 21) but increased transgene expression (Table 22) in the muscle relative to AAV5. Table 21. Quantification of viral genome copies per diploid genome (biodistribution) by ddPCR following intravenous administration of AAV particles comprising a TTN-002 capsid normalized to the actual titer of the viral vector in the dosing solution (vg/dg = viral genome copies/ diploid genome)
Attorney Docket No.14640.0092-00304 Table 22. Quantification of transgene mRNA by RT-qPCR following intravenous administration of AAV particles comprising a TTN-002 capsid normalized to the actual titer of the viral vector in the dosing solution (mRNA = transgene mRNA fold over housekeeping gene; rel. to AAV9= transgene mRNA fold over housekeeping gene relative to AAV9; rel. to AAV5 transgene mRNA fold over housekeeping gene relative to AAV5)
Conclusions [0503] Taken together, these data demonstrate that TTN-002, which is an AAV5 capsid variant, is an enhanced CNS tropic capsid, in both NHPs and rodents (e.g., rats and mice), and was able to successfully penetrate the blood brain barrier. More specifically, TTN-002 demonstrated the ability to cross species, demonstrating improved CNS tropism and biodistribution in both NHPs and rats relative to AAV5 in mice. Example 3. Maturation of the TTN-002 capsid in mice [0504] This Example describes maturation of the TTN-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 943) capsid variant in mice to further enhance its transduction and biodistribution in the central nervous system, evolve the AAV capsid variant further, and to provide cross-species compatibility. Two approaches were used to mature the TTN-002 capsid sequence in order to randomize and mutate within and around the peptide insert comprised within loop VIII of the capsid variant. In the first maturation approach, mutagenic primers were used to introduce point mutations at a low frequency, scattered across the mutagenesis region in the TTN-002 sequence ranging from approximately position 571 to position 586, numbered according to SEQ ID NO: 982. In the second maturation approach, sets of three contiguous amino acids were randomized across the mutagenesis region in the TTN-002 sequence, which spanned from approximately position 571 to position 586, numbered according to SEQ ID NO: 982. AAV capsid variants arising from each maturation approach for TTN-002 were pooled together, for subsequent testing and characterization in mice. [0505] The library of pooled matured AAV capsid variants generated from TTN-002 using the first maturation approach and the library of pooled matured AAV capsid variants generated from TTN-002 using the second maturation approach were each injected into three CD-1 Outbred mice. After a period in
Attorney Docket No.14640.0092-00304 life, the brains of the mice were isolated and RNA was extracted. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the TTN-002 control, and the peptides comprised within the matured variants were identified. The data from the first maturation approach are provided in Table 23 and the data from the second maturation approach is provided in Table 24. [0506] Following the RNA recovery and NGS analysis from the first maturation approach, the matured capsid variants were filtered based on their coefficient of variance (CV), which was calculated for each peptide across six brain samples taken (two per mouse). Those that had a CV value <1 were identified, as these were the peptides that were reliably detected in 5/6 or 6/6 brain samples isolated from the three mice. Table 23 provides the peptide sequences of these matured capsid variants and the fold enrichment of the matured capsid variant relative to the non-matured TTN-002 control. As shown in Table 23, approximately 28 TTN-002 matured capsid variants demonstrated an increase in expression relative to the non-matured TTN-002 control, with approximately 16 variants demonstrating at least a 2- fold increase in expression. Several variants demonstrated at least an 8-fold to 15-fold increase in expression relative to the non-matured TTN-002 control. Table 23. NGS fold-enrichment of TTN-002 matured AAV capsid variants in the brain of outbred mice following first mutagenesis approach
Attorney Docket No.14640.0092-00304
[0507] Following the RNA recovery and NGS analysis from the second maturation approach, the matured capsid variants were filtered for those that were detectable in all samples from all mice injected with the matured capsid variants. Table 24 provides the peptide sequences of these matured capsid variants, the fold enrichment of the matured capsid variant relative to the non-matured TTN-002 control, as well as the predicted capsid of origin from which the variant was matured. As shown in Table 24, approximately 526 TTN-002 matured capsid variants demonstrated an increase in expression relative to the non-matured TTN-002 control, with approximately 358 variants demonstrated at least a 2-fold increase in expression. Several variants demonstrated a 20-711-fold or greater increase in expression relative to the non-matured TTN-002 control. Table 24. NGS fold-enrichment of TTN-002 matured AAV capsid variants in the brain of outbred mice following second mutagenesis approach
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
[0508] These data demonstrate that following two maturation approaches, matured TTN-002 capsid variants with loop VIII modifications were generated with significantly enhanced CNS tropism in mice compared to the corresponding non-matured capsid variants, which already exhibited a significant fold enrichment over AAV5 and/or AAV9 in the brain of mice, rats, and/or NHPs. Example 4. Maturation of the TTN-002 capsid in NHPs [0509] This Example describes maturation of TTN-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 943 (encoded by SEQ ID NO: 944)) capsid variant in NHPs, specifically cynomolgus macaques (Macaca fascicularis), to further enhance its transduction and biodistribution in the central nervous system and peripheral tissues and to evolve the AAV capsid variant further. Two approaches were used to mature the TTN-002 capsid sequences in order to randomize and mutate within and around the peptide insert comprised within loop VIII of the capsid variant. In the first maturation approach, mutagenic primers were used to introduce point mutations at a low frequency, scattered across the mutagenesis region in the TTN-002 sequence ranging from approximately position 571 to position 586, numbered according to SEQ ID NO: 982. In the second maturation approach, sets of three contiguous amino acids were randomized across the mutagenesis region in the TTN-002 sequence, which spanned from approximately position 571 to position 586, numbered according to SEQ ID NO: 982. AAV capsid variants arising from each maturation approach for TTN-002 were pooled together, for subsequent testing and characterization in NHPs. [0510] The library of pooled matured AAV capsid variants generated from TTN-002 using the first maturation approach and the library of pooled matured AAV capsid variants generated from TTN-002 using the second maturation approach were each injected into two NHPs. After a period in life, the brains, heart, liver, muscle, and DRG of the NHPs were isolated and RNA was extracted. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the TTN-002 control, and the peptides comprised within the variants were identified. [0511] Following the RNA recovery and NGS analysis from the second maturation approach, the matured capsid variants were filtered based on their coefficient of variance (CV), which was calculated for each peptide across the brain, heart, liver, muscle and DRG samples taken from the two NHPs. Those that had a CV value <2 were identified, as these were the peptides that were reliably detected in the majority of samples isolated from the brains of the two NHPs. [0512] Table 25 provides the peptide sequences of these matured capsid variants and the fold enrichment of the matured capsid variant relative to the non-matured TTN-002 control that demonstrated the greatest fold-change in expression relative to the non-matured TTN-002 capsid variant in the brain of NHPs, following the first maturation approach and the second maturation approach. As shown in Table 25, following the first maturation approach, approximately 5 TTN-002 matured capsid variants
Attorney Docket No.14640.0092-00304 demonstrated an increase in expression relative to the non-matured TTN-002 control, which demonstrated at least a 5-fold to 53-fold increase in expression in the NHP brain relative to the non- matured TTN-002 control. Following the second maturation approach, approximately 38 TTN-002 matured capsid variants demonstrated an increase in expression in the NHP brain relative to the non- matured TTN-002 control, with at least 27 demonstrating at least a 2-fold increase in expression (Table 25). Several variants demonstrated at least a 12-fold to 222-fold increase in expression in the NHP brain relative to the non-matured TTN-002 control (Table 25). [0513] Fold-change in expression for the TTN-002 matured variants in Table 25 were also calculated for the DRG, muscle, liver (RNA and DNA), and heart of the NHPs following each maturation approach. Several variants that led to increased expression relative to the non-matured TTN-002 variant in the brain of the NHP also led to increased expression in other tissues. For instance, the matured TTN-002 capsid variant comprising the amino acid sequence TNNQSSYTPSLVQKTA (SEQ ID NO: 1585) demonstrated increased expression in the brain, heart, and liver relative to the non-matured TTN-002 control. Similarly, the matured TTN-002 capsid variants comprising the amino acid sequence TNNQSSYPPSLVKKTA (SEQ ID NO: 1591) and TNNQSSYPPSLVQKPA (SEQ ID NO: 1593), demonstrated increased expression in the brain and heart relative to the non-matured TTN-002 control. Additionally, the matured TTN-002 capsid variant comprising the amino acid sequence INNQSSYPAEVVQKTA (SEQ ID NO: 1024) demonstrated increased expression in the brain and the muscle relative to the non-matured TTN- 002 control. Also, as shown in Table 25, many of the TTN-002 capsid variants that had increased expression in the brain, were de-targeted in the DRG. Therefore, several matured variants demonstrated increased tropism in more than one tissue type in the NHPs, with many showing reduced expression in the DRG. Table 25. NGS fold-enrichment of the TTN-002 matured AAV capsid variants in the brain of NHPs following first and second mutagenesis approaches as compared to other NHP and mouse tissues
Attorney Docket No.14640.0092-00304
[0514] Furthermore, several of the TTN-002 matured capsid variants demonstrating an increase in expression relative to the non-matured TTN-002 control following the first and second maturation approaches in the brain of NHPs as shown in Table 25, also demonstrated an increase in expression in the brain of mice following the first and second maturation approach in mice. For instance, the matured TTN-002 capsid variant comprising the amino acid sequence TNNQSKYPAEVVQKTA (SEQ ID NO: 1538) demonstrated a 53.7-fold increase in expression relative to the non-matured TTN-002 following the first maturation approach in brain of NHPs, demonstrated a 20.86-fold increase in expression relative
Attorney Docket No.14640.0092-00304 to the non-matured TTN-002 following the second maturation approach in brain of NHPs, a 10.26-fold increase in expression relative to the non-matured TTN-002 following the first maturation approach in brain of mice, and a 5.47-fold increase in increase in expression relative to the non-matured TTN-002 following the second maturation approach in brain of mice. Similarly, the matured TTN-002 capsid variant comprising the amino acid sequence TNNSSSYPAEVVQKTA (SEQ ID NO: 1539) demonstrated an 18.997-fold increase in expression relative to the non-matured TTN-002 following the first maturation approach in brain of NHPs, an 8.093-fold increase in expression relative to the non- matured TTN-002 following the second maturation approach in brain of NHPs, an 8.539-fold increase in expression relative to the non-matured TTN-002 following the first maturation approach in brain of mice, and a 5.903-fold increase in increase in expression relative to the non-matured TTN-002 following the second maturation approach in brain of mice. Matured TTN-002 capsid variants comprising the amino acid sequences of SEQ ID NOs: 1021, 1024, 1027, 1112, 1142, 1214, 1232, 1254, 1300, 1310, 1327, 1331, 1342, and 1593 also demonstrated an increase in expression in the brain of both NHPs and mice, relative to the non-matured TTN-002 control. Therefore, several matured variants demonstrated increased expression relative to the non-matured controls in at least two different species, indicating cross-species tropism. [0515] Table 26 provides the peptide sequences of these matured capsid variants, and the fold enrichment of the matured capsid variant relative to the non-matured TTN-002 control that demonstrated increased expression in the heart of NHPs following the second maturation approach. As shown in Table 26, approximately 17 TTN-002 matured capsid variants demonstrated an increase in expression in the heart relative to the non-matured TTN-002 control, with at least 13 demonstrating at least a 2-fold increase in expression. Several variants demonstrated at least a 10-fold to 47-fold increase in expression in the heart relative to the non-matured TTN-002 control. Fold-change in expression for the TTN-002 matured variants in Table 26 were also calculated for the brain, DRG, muscle, and liver (RNA and DNA), of the NHPs and in the brains of mice. Table 26. NGS fold-enrichment of the TTN-002 matured AAV capsid variants in the heart of NHPs following the second mutagenesis approach as compared to other NHP and mouse tissues
Attorney Docket No.14640.0092-00304
[0516] Table 27 provides the peptide sequences of these matured capsid variants, and the fold enrichment of the matured capsid variant relative to the non-matured TTN-002 control that demonstrated increased expression in the muscle of NHPs following the second maturation approach. As shown in Table 27, approximately 33 TTN-002 matured capsid variants demonstrated an increase in expression in the muscle relative to the non-matured TTN-002 control, with at least 19 demonstrating at least a 2-fold increase in expression. Several variants demonstrated at least a 7-fold to 38-fold increase in expression in the muscle relative to the non-matured TTN-002 control. Fold-change in expression for the TTN-002 matured variants in Table 27 were also calculated for the brain, DRG, heart, and liver (RNA and DNA), of the NHPs and in the brains of mice. Table 27. NGS fold-enrichment of the TTN-002 matured AAV capsid variants in the muscle of NHPs following the second mutagenesis approach
Attorney Docket No.14640.0092-00304
[0517] Table 28 provides the peptide sequences of these matured capsid variants, and the fold enrichment of the matured capsid variant relative to the non-matured TTN-002 control that demonstrated increased expression in the liver of NHPs following the first and second maturation approach. As shown in Table 28, following the first maturation approach, approximately 7 TTN-002 matured capsid variants demonstrated an 11-fold to 189-fold increase in expression in the liver relative to the non-matured TTN- 002 control. Following the second maturation approach, approximately 395 TTN-002 matured capsid variants demonstrated an increase in expression in the liver of at least 9-fold relative to the non-matured TTN-002 control (Table 28). Several variants demonstrated at least a 50-fold to 114-fold increase in expression in the liver relative to the non-matured TTN-002 control (Table 28). Fold-change in expression for the TTN-002 matured variants in Table 28 were also calculated for the brain, DRG, heart, and muscle of the NHPs and in the brains of mice. Table 28. NGS fold-enrichment of the TTN-002 matured AAV capsid variants in the liver of NHPs following both mutagenesis approaches
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
Attorney Docket No.14640.0092-00304
[0518] These data demonstrate that following two maturation approaches, matured TTN-002 capsid variants (AAV5 capsid variants) with loop VIII modifications were generated with significantly enhanced CNS, heart, muscle, and liver tropism in NHPs compared to the corresponding non-matured capsid variants, which already exhibited a significant fold enrichment over AAV5 and/or AAV9 in the brain of mice, rats, and/or NHPs. Also, several of the resulting matured variants demonstrated cross- species CNS tropism in both NHPs and mice. Example 5. Evaluation of TTN-002 AAV capsid variant in diverse primate species [0519] This Example evaluates the tropism and cross-species compatibility of the TTN-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 943) capsid variant in two diverse primate species, marmosets (Callithrix jacchus) and African green monkeys (Chlorocebus sabaeus), as compared to their tropism in cynomolgus macaques (Macaca fascicularis) provided in Example 1 and 2. The amino acid and DNA sequences of the TTN-002 capsid variant are provided, e.g., in Tables 4 and 5, respectively. [0520] To investigate tropism in African green monkeys, AAV particles comprising the TTN-002 capsid variant or an AAV5 control under the control of a synapsin promoter, were intravenously injected into the African green monkeys (n=2, 3-12 years of age) at a dose of 2E13 vg/kg. After 14-days in life, the brains and tissues (liver, DRG, quadriceps, and heart) of the NHPs were collected and RNA was
Attorney Docket No.14640.0092-00304 extracted. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the AAV5 wild-type control. [0521] To investigate tropism in marmoset monkeys, AAV particles comprising the TTN-002 capsid variant, or an AAV5 control, were intravenously injected into marmosets (n=2, >10 months of age) at a dose of 2E13 vg/kg. After 28-days in life, the brains and tissues (liver quadriceps, and heart) of the NHPs were collected and RNA was extracted. Following RNA recovery and RT-PCR amplification, a systematic NGS enrichment analysis was performed to calculate the fold enrichment ratio relative to the AAV5 wild-type control. [0522] As provided in Table 29 (African green monkeys) and Table 30 (marmosets), the TTN-002 capsid variant demonstrated increased CNS tropism in diverse primate species. The TTN-002 capsid variant demonstrated a 64.9-fold increase in expression relative to AAV5 in the brain of cynomolgus macaques (Table 14, Example 1), a 7.5-fold increase in expression relative to AAV5 in the brain of African green monkeys, and a 40.4-fold increase in expression relative to AAV5 in the brain of marmosets. Furthermore, TTN-002 also resulted in increased expression in the brain of rats (Table 14, Example 1), demonstrating an average fold change in expression relative to AAV5 of 41.1. Table 29. NGS-Fold Enrichment of TTN-002 in African green monkeys
Table 30. NGS-Fold Enrichment of TTN-002 in Marmosets
[0523] Taken together, these data demonstrate that the AAV5 capsid variant TTN-002 demonstrated increased CNS tropism relative to the AAV5 control in the CNS across three diverse primate species and rats, providing evidence of strong cross-species capacity. Example 6. In vivo Promoter Selection Studies [0524] ITR-to-ITR sequences comprising promoters were packaged into AAV6 capsids and delivered by intrastriatal injection to Sprague Dawley rats at a dose of 1x1010 VG. Tissue samples were collected at 3 weeks or 10 weeks and frataxin protein levels quantified. Frataxin constructs comprising truncated CBA and CMV promoters were packed into an AAV9 variant capsid (VOY201) and administered by intravenous delivery to Sprague Dawley rats at a dose of either 6.3x1012 or 2x1013 VG/kg. After 28 or 90 days, tissue samples were collected and processed for quantification of frataxin expression (ng/mg). The sequence for the VOY201 capsid is disclosed in, e.g., PCT/US2019/053681, the contents of which are incorporated herein in their entirety.
Attorney Docket No.14640.0092-00304 [0525] Promoters CMV-D7 (SEQ ID NO: 1750) and CBA-D8 (SEQ ID NO: 1742) demonstrated the target moderate frataxin expression as compared to the other constructs and were therefore selected for further study. [0526] To test the durability and persistence of frataxin expression driven by the CMV-D7 (SEQ ID NO: 1750) and CBA-D8 (SEQ ID NO: 1742) promoters, a time course study was conducted. Viral genomes comprising a CMV-D7 (SEQ ID NO: 1750) or a CBA-D8 (SEQ ID NO: 1742 promoter with a frataxin payload sequence were packaged into another AAV9-variant (VOY101) capsid to generate AAV particles. These AAV particles were administered by intravenous delivery via the tail vein to male Sprague Dawley rats at one of two doses (6.3x1012 or 2x1013). At 28, 90, or 180 days after administration, tissue samples were collected (heart, liver, cerebellum, thoracic and lumbar DRG) and processed for quantification of viral genome per diploid cell and frataxin expression levels based on an Anti-Frataxin SimpleStep ELISA. Data are shown below in Tables 31 and 32, FIGs.2A, 2B, 2C and 2D. Table 31. Frataxin expression (ng/mg)
Table 32. Viral genome/diploid cell (VG/dc)
[0527] In tissue collected from the heart ventricle, driving frataxin expression using the CMV-D7 (SEQ ID NO: 1750) promoter enhanced frataxin expression 0.2-2.5x, while driving frataxin expression using the CBA-D8 (SEQ ID NO: 1742) promoter enhanced frataxin expression 0.3-7.8x (FIG.2A).
Attorney Docket No.14640.0092-00304 [0528] In tissue collected from the cerebellum, driving frataxin expression using the CMV-D7 (SEQ ID NO: 1750) promoter enhanced frataxin expression 0.01-0.31x, while driving frataxin expression using the CBA-D8 (SEQ ID NO: 1742) promoter enhanced frataxin expression 0.01-0.28x (FIG.2B). [0529] In tissue collected from the lumbar DRG, driving frataxin expression using the CMV-D7 (SEQ ID NO: 1750) promoter enhanced frataxin expression 1.6-6.2x, while driving frataxin expression using the CBA-D8 (SEQ ID NO: 1742) promoter enhanced frataxin expression 1.1-5.2x (FIG.2C). [0530] Immunohistochemical analysis was performed on 30μm tissue samples collected 28 days after AAV particle administration. An anti-hFXN antibody (1/50,000) was used. Frataxin expression driven by the CMV-D7 (SEQ ID NO: 1750) and CBA-D8 (SEQ ID NO: 1742) promoters was detected in the dentate nucleus of treated rats. [0531] Each of CMV-D7 (SEQ ID NO: 1750), CBA-D8 (SEQ ID NO: 1742) and CBA (SEQ ID NO: 1734) promoters showed similar distribution and expression patterns in the DRG and brain. In the heart, CMV-D7 and CBA-D8 promoters generated FXN expression approximately 50-260-fold lower than CBA-driven frataxin expression. [0532] The CMV-D7 (SEQ ID NO: 1750) and CBA-D8 (SEQ ID NO: 1742) promoters both drove frataxin expression in the cerebellum, heart and DRG at 180 days after administration of the AAV particles. At this time point, expression in the cerebellum was approximately 3-fold greater than that achieved using a reference CBA promoter, indicating that the CMV-D7 and CBA-D8 promoters are active in target cells of the cerebellum. Example 7. Evaluation of TTN-002 for delivering frataxin in mice [0533] The goal of these experiments was to determine the ability of the capsid variant TTN-002 (SEQ ID NO: 982 (amino acid) and 984 (DNA), comprising SEQ ID NO: 943) with a frataxin-encoding transgene to increase the expression of frataxin in mice, and particularly in mouse cerebellum. The amino acid and DNA sequences of TTN-002 are provided, e.g., in Tables 4 and 5, respectively. [0534] AAV particles were generated with TTN-002 encapsulating a viral genome comprising SEQ ID NO: 1797 (“TTN-002_FXN”). [0535] The AAV particles or vehicle control (modified PBS) were administered intravenously to wildtype or frataxin-deficient mice (B6.Cg-Pvalbtm1(cre)Arbr Fxnem2Lutzy Fxnem2.1Lutzy/J (Jackson Laboratory Stock#029721)). TTN-002_FXN was administered at 1e14 or 2e13 vg/kg. [0536] At 16-weeks post-administration, the mice were euthanized and biodistribution (vg/dc) and frataxin expression (ng/mg) were evaluated from the cerebellum using ddPCR and ELISA, respectively. Results are shown in FIG.4A and FIG.4B. Equivalents and Scope [0537] Those skilled in the art will recognize, or be able to ascertain using no more than routine experimentation, many equivalents to the specific embodiments in accordance with the Detailed
Attorney Docket No.14640.0092-00304 Description provided herein. The scope of the present disclosure is not intended to be limited to the above Detailed Description, but rather is as set forth in the appended claims. [0538] Where ranges are given, endpoints are included. Furthermore, it is to be understood that unless otherwise indicated or otherwise evident from the context and understanding of one of ordinary skill in the art, values that are expressed as ranges can assume any specific value or subrange within the stated ranges in different embodiments of the disclosure, to the tenth of the unit of the lower limit of the range, unless the context clearly dictates otherwise. [0539] In addition, it is to be understood that any particular embodiment of the present disclosure that falls within the prior art may be explicitly excluded from any one or more of the claims. Since such embodiments are deemed to be known to one of ordinary skill in the art, they may be excluded even if the exclusion is not set forth explicitly herein. Any particular embodiment of the compositions of the disclosure (e.g., any, composition, therapeutic or active ingredient; any method of production; any method of use; etc.) can be excluded from any one or more claims, for any reason, whether or not related to the existence of prior art. [0540] It is to be understood that the words which have been used are words of description rather than limitation, and that changes may be made within the purview of the appended claims without departing from the true scope and spirit of the disclosure in its broader aspects. [0541] While the present disclosure has been described at some length and with some particularity with respect to the several described embodiments, it is not intended that it should be limited to any such particulars or embodiments or any particular embodiment, but it is to be construed with references to the appended claims so as to provide the broadest possible interpretation of such claims in view of the prior art and, therefore, to effectively encompass the intended scope of the disclosure. [0542] In addition, section headings, the materials, methods, and examples are illustrative only and not intended to be limiting.
Claims
Attorney Docket No.14640.0092-00304 CLAIMS What is claimed is: 1. An adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome, wherein the viral genome comprises a frataxin (FXN)-encoding sequence and the AAV capsid variant comprises an amino acid sequence having the formula [N2]-[N3], wherein: (i) [N2] comprises X1, X2, X3, X4, and X5, wherein: (a) X1 is Y, N, or C; (b) X2 is P, K, T, or Q; (c) X3 is A or P; (d) X4 is E, S, or A; and (e) X5 is V, L, or E; and (ii) [N3] comprises the amino acid sequence VQK, EQK, VKK, VHK, VQQ, or LQK; and wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 739, or an amino acid sequence at least 95% identical thereto. 2. The AAV particle of claim 1, wherein [N2]-[N3] is present in loop VIII, wherein loop VIII is present at amino acids comprising those corresponding to positions 571-599 of the amino acid sequence of SEQ ID NO: 982. 3. The AAV particle of claim 1 or claim 2, wherein [N2]-[N3] is present immediately subsequent to an amino acid corresponding to position 576 of the amino acid sequence of SEQ ID NO: 982. 4. The AAV particle of any one of claims 1-3, wherein the AAV capsid variant is an AAV5 capsid variant comprising [N2]-[N3] in place of an amino acid corresponding to T577 of the amino acid sequence of SEQ ID NO: 138. 5. The AAV particle of any one of claims 1-4, wherein [N2] comprises the amino acid sequence YP, YPA, YPAE (SEQ ID NO: 21), or YPAEV (SEQ ID NO: 1). 6. The AAV particle of any one of claims 1-5, wherein [N3] comprises the amino acid sequence VQK. 7. The AAV particle of any one of claims 1-6, wherein [N2]-[N3] comprises the amino acid sequence AEVVQK (SEQ ID NO: 36) or PAEVVQK (SEQ ID NO: 20). 8. The AAV particle of any one of claims 1-7, wherein [N2]-[N3] comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943).
Attorney Docket No.14640.0092-00304 9. The AAV particle of any one of claims 1-8, wherein the AAV capsid variant further comprises [N1] comprising XD, XE, and XF, wherein: (a) XD of [N1] is Q, T, S, A, I, L, or H; (b) XE of [N1] is S, G, A, or R; and (c) XF of [N1] is S, K, L, R, A, or T; wherein [N1] immediately precedes [N2]-[N3]. 10. The AAV particle of claim 9, wherein [N1] comprises the amino acid sequence QS, SS, or QSS. 11. The AAV particle of claim 9 or claim 10, wherein [N1]-[N2] comprises the amino acid sequence QSSYPAEV (SEQ ID NO: 96). 12. The AAV particle of any one of claims 9-11, wherein [N1]-[N2]-[N3] comprises the amino acid sequence SSYPAEVVQ (SEQ ID NO: 121) or QSSYPAEVVQK (SEQ ID NO: 150). 13. The AAV particle of any one of claims 9-12, wherein the AAV capsid variant further comprises [N0] comprising XA, XB, and XC, wherein: (a) XA of [N0] is T, I, or N; (b) XB of [N0] is N; and (c) XC of [N0] is N, T, S, or K; wherein [N0] immediately precedes [N1]. 14. The AAV particle of claim 13, wherein [N0] comprises the amino acid sequence TN, NN, or TNN. 15. The AAV particle of claim 13 or claim 14, wherein [N0]-[N1] comprises the amino acid sequence TNNQSS (SEQ ID NO: 183). 16. The AAV particle of any one of claims 13-15, wherein [N0]-[N1]-[N2]-[N3] comprises the amino acid sequence TNNQSSYPAEVVQK (SEQ ID NO: 500). 17. The AAV particle of any one of claims 13-16, wherein the AAV capsid variant further comprises [N4] comprising XG and XH, wherein: (a) XG of [N4] is T, P, or N; and (b) XH of [N4] is A; wherein [N4] is present immediately subsequent to [N3].
Attorney Docket No.14640.0092-00304 18. The AAV particle of claim 17, wherein [N4] comprises the amino acid sequence TA. 19. The AAV particle of claim 17 or claim 18, wherein [N0]-[N1]-[N2]-[N3]-[N4] comprises the amino acid sequence TNNQSSYPAEVVQKTA (SEQ ID NO: 1533). 20. The AAV particle of any one of claims 16-18, wherein [N0]-[N1]-[N2]-[N3]-[N4] is present in loop VIII, wherein loop VIII is present at amino acids comprising those corresponding to positions 571-599 of the amino acid sequence of SEQ ID NO: 982. 21. The AAV particle of claim 20, wherein: (i) [N0] is present at amino acids corresponding to positions 571-573 of the amino acid sequence of SEQ ID NO: 982; (ii) [N1] is present at amino acids corresponding to positions 574-576 of the amino acid sequence of SEQ ID NO: 982; (iii) [N2] is present at amino acids corresponding to positions 577-581 of the amino acid sequence of SEQ ID NO: 982; (iv) [N3] is present at amino acids corresponding to positions 582-584 of the amino acid sequence of SEQ ID NO: 982; and (v) [N4] is present at amino acids corresponding to positions 585-586 of the amino acid sequence of SEQ ID NO: 982. 22. An adeno-associated virus (AAV) particle comprising: (i) a viral genome comprising a frataxin (FXN)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% identical to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises at least 4, at least 5, at least 6, at least 7, or all 8 consecutive amino acids from the amino acid sequence YPAEVVQK (SEQ ID NO: 943). 23. The AAV particle of claim 22, wherein: (i) the at least 4 consecutive amino acids comprise the amino acid sequence YPAE (SEQ ID NO: 21), optionally present at amino acids corresponding to positions 577-580 of the amino acid sequence of SEQ ID NO: 982; (ii) the at least 5 consecutive amino acids comprise the amino acid sequence YPAEV (SEQ ID NO: 1), optionally present at amino acids corresponding to positions 577-581 of the amino acid sequence of SEQ ID NO: 982;
Attorney Docket No.14640.0092-00304 (iii) the at least 6 consecutive amino acids comprise the amino acid sequence YPAEVV (SEQ ID NO: 725), optionally present at amino acids corresponding to positions 577-582 of the amino acid sequence of SEQ ID NO: 982; or (iv) the at least 7 consecutive amino acids comprise the amino acid sequence YPAEVVQ (SEQ ID NO: 726), optionally present at amino acids corresponding to positions 577-583 of the amino acid sequence of SEQ ID NO: 982. 24. The AAV particle of claim 22 or claim 23, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943) present at amino acids corresponding to positions 577-584 of the amino acid sequence of SEQ ID NO: 982. 25. An adeno-associated virus (AAV) particle comprising: (i) a viral genome comprising a frataxin (FXN)-encoding sequence; and (ii) an AAV capsid variant comprising an amino acid sequence at least 95% identical to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943) at amino acids corresponding to positions 577-584 of the amino acid sequence of SEQ ID NO: 982. 26. The AAV particle of claim 25, wherein the AAV capsid variant further comprises (i) an amino acid sequence having at least 95% identity to the amino acid sequence of SEQ ID NO: 738, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); and/or (ii) an amino acid sequence having at least 95% identity to the amino acid sequence of SEQ ID NO: 982, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). 27. An adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome, wherein the viral genome comprises a frataxin (FXN)-encoding sequence and the AAV capsid variant comprises (i) an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 982, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); (ii) an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 738, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); and/or
Attorney Docket No.14640.0092-00304 (iii) an amino acid sequence having at least 99% identity to the amino acid sequence of SEQ ID NO: 739, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). 28. The AAV particle of any one of claims 1-27, wherein the AAV capsid variant comprises (i) the amino acid sequence of SEQ ID NO: 982; (ii) the amino acid sequence of SEQ ID NO: 738; and/or (iii) the amino acid sequence of SEQ ID NO: 739. 29. The AAV particle of any one of claims 1-28, wherein the FXN-encoding sequence encodes a FXN protein that is not a cynomolgus monkey FXN protein. 30. The AAV particle of any one of claims 1-29, wherein the FXN-encoding sequence encodes a human FXN protein. 31. The AAV particle of claim 30, wherein the human FXN protein comprises the amino acid sequence of SEQ ID NO: 1825, or an amino acid sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical thereto, optionally wherein the human FXN protein is a wildtype human FXN protein. 32. The AAV particle of any one of claims 1-31, wherein the FXN-encoding sequence comprises a nucleotide sequence encoding the amino acid sequence of SEQ ID NO: 1825. 33. The AAV particle of any one of claims 1-32, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 34. The AAV particle of any one of claims 1-33, wherein the viral genome further comprises a promoter operably linked to the FXN-encoding sequence. 35. The AAV particle of claim 34, wherein the promoter comprises a human elongation factor 1Į-subunit (EF1Į) promoter, a cytomegalovirus (CMV) immediate-early enhancer and/or promoter, a chicken ȕ- actin (CBA) promoter, a CAG promoter, a ȕ glucuronidase (GUSB) promoter, a ubiquitin C (UBC) promoter, a neuron-specific enolase (NSE) promoter, a platelet-derived growth factor (PDGF) promoter, a platelet-derived growth factor B-chain (PDGF-ȕ) promoter, a intercellular adhesion molecule 2 (ICAM-
Attorney Docket No.14640.0092-00304 2) promoter, a synapsin (Syn) promoter, a methyl-CpG binding protein 2 (MeCP2) promoter, a Ca2+/calmodulin-dependent protein kinase II (CaMKII) promoter, a metabotropic glutamate receptor 2 (mGluR2) promoter, a neurofilament light chain (NFL) promoter, a neurofilament heavy chain (NFH) promoter, a ȕ-globin minigene nȕ2 promoter, a preproenkephalin (PPE) promoter, a enkephalin (Enk) and excitatory amino acid transporter 2 (EAAT2) promoter, a glial fibrillary acidic protein (GFAP) promoter, a myelin basic protein (MBP) promoter, a cardiovascular promoter (e.g., ĮMHC, cTnT, and CMV-MLC2k), a liver promoter (e.g., hAAT, TBG), a skeletal muscle promoter (e.g., desmin, MCK, C512), or a functional fragment or truncation of any of the foregoing. 36. The AAV particle of claim 32 or claim 33, wherein the promoter is a CMV promoter or CBA promoter, or a functional fragment or truncation of a CMV promoter or CBA promoter. 37. The AAV particle of claim 36, wherein the promoter is a truncated CBA promoter. 38. The AAV particle of claim 37, wherein the truncated CBA promoter is 50-400 nucleotides in length, e.g., 100-332 nucleotides in length. 39. The AAV particle of any one of claims 36-38, wherein the promoter comprises the nucleotide sequence of any one of SEQ ID NOs: 1738, 1740, and 1742 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 40. The AAV particle of claim 36, wherein the promoter is a truncated CMV promoter. 41. The AAV particle of claim 40, wherein the truncated CMV promoter is 50-300 nucleotides in length. 42. The AAV particle of claim 40 or claim 41, wherein the promoter comprises the nucleotide sequence of SEQ ID NO: 1750 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 43. The AAV particle of any one of claims 1-42, wherein the viral genome further comprises a microRNA (miR) binding site that modulates expression of the encoded FXN protein in a cell or tissue of the liver. 44. The AAV particle of claim 43, wherein the viral genome comprises 3 copies of the miR binding site.
Attorney Docket No.14640.0092-00304 45. The AAV particle of claim 44, wherein the 3 copies of the miR binding site are identical. 46. The AAV particle of claim 44 or claim 45, wherein the 3 copies of the miR binding site are continuous. 47. The AAV particle of any one of claims 43-46, wherein the miR binding site is a miR122 binding site, optionally wherein: the miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1827 or a sequence having one, two, three, or at most four substitutions relative to the nucleotide sequence of SEQ ID NO: 1827; or the viral genome comprises 3 copies of a miR122 binding site comprising the nucleotide sequence of SEQ ID NO: 1826 or a sequence having one, two, three, four, five, six, seven, eight, nine, or at most ten substitutions relative to the nucleotide sequence of SEQ ID NO: 1826. 48. The AAV particle of any one of claims 1-47, wherein the viral genome further comprises at least one inverted terminal repeat (ITR) region. 49. The AAV particle of claim 48, wherein the at least one ITR region comprises an AAV2 ITR. 50. The AAV particle of claim 48 or claim 49, wherein the viral genome comprises a 5’ ITR region and a 3’ ITR region. 51. The AAV particle of claim 50, wherein the 5’ ITR region and 3’ ITR region are each an AAV2 ITR. 52. The AAV particle of claim 51, wherein the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 53. The AAV particle of any one of claims 1-52, wherein the viral genome further comprises an intron/exon region comprising an intron region and/or an exon region. 54. The AAV particle of claim 53, wherein the intron/exon region comprises: an immediate-early 1 (ie1) intron region and/or a human beta-globin (hBglobin) intron 2 region; and/or
Attorney Docket No.14640.0092-00304 an ie1 exon region and/or an hBglobin exon region. 55. The AAV particle of claim 53 or claim 54, wherein the intron region comprises: the nucleotide sequence of SEQ ID NO: 1819 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the nucleotide sequence of SEQ ID NO: 1820 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 56. The AAV particle of claim 55, wherein the exon region comprises: the nucleotide sequence of SEQ ID NO: 1817 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or the nucleotide sequence of SEQ ID NO: 1821 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 57. The AAV particle of any one of claims 1-56, wherein the viral genome further comprises a polyadenylation (polyA) region. 58. The AAV particle of claim 57, wherein the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 59. The AAV particle of any one of claims 1-30, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 60. The AAV particle of any one of claims 1-30, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto.
Attorney Docket No.14640.0092-00304 61. The AAV particle of any one of claims 1-30, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 62. The AAV particle of any one of claims 1-30, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 63. The AAV particle of any one of claims 1-30, wherein the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) the FXN-encoding sequence, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; and (iv) a 3’ ITR region. 64. The AAV particle of any one of claims 1-30, wherein the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) the FXN-encoding sequence, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (iv) at least one miR122 binding site; and (v) a 3’ ITR region. 65. The AAV particle of any one of claims 1-30, wherein the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron/exon region; (iv) the FXN-encoding sequence, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824;
Attorney Docket No.14640.0092-00304 (v) at least one miR122 binding site; and (vi) a 3’ ITR region. 66. The AAV particle of any one of claims 1-30, wherein the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron/exon region; (iv) the FXN-encoding sequence, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; and (vii) a 3’ ITR region. 67. The AAV particle of any one of claims 1-30, wherein the viral genome comprises: (i) a 5’ inverted terminal repeat (ITR) region; (ii) a promoter; (iii) an intron and/or exon region; (iv) the FXN-encoding sequence, wherein the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 80% identical (e.g., at least 80%, at least 85%, at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) to SEQ ID NO: 1824; (v) at least one miR122 binding site; (vi) a polyadenylation (polyA) region; (vii) a filler sequence; and (viii) a 3’ ITR region. 68. The AAV particle of claim 66 or claim 67, wherein: (i) the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises the nucleotide sequence of SEQ ID NO: 1742 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least
Attorney Docket No.14640.0092-00304 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1826 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (vii) the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 69. The AAV particle of claim 68, wherein the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1841 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. 70. The AAV particle of claim 66 or claim 67, wherein: (i) the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises the nucleotide sequence of SEQ ID NO: 1750 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least
Attorney Docket No.14640.0092-00304 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1826 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (vii) the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 71. The AAV particle of claim 70, wherein the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1840 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. 72. The AAV particle of claim 66 or claim 67, wherein: (i) the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises the nucleotide sequence of SEQ ID NO: 1738 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1826 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at
Attorney Docket No.14640.0092-00304 least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or (vii) the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 73. The AAV particle of claim 72, wherein the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1838 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. 74. The AAV particle of claim 66 or claim 67, wherein: (i) the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (ii) the promoter comprises the nucleotide sequence of SEQ ID NO: 1740 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (iv) the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (v) the at least one miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1826 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and/or
Attorney Docket No.14640.0092-00304 (vii) the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 75. The AAV particle of claim 74, wherein the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1839 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto, optionally wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region. 76. The AAV particle of claim 66 or claim 67, wherein the viral genome comprises (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; or (d) the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 77. The AAV particle of any one of claims 66-68, wherein: (i) the 5' ITR region comprises the nucleotide sequence of SEQ ID NO: 1811; (ii) the promoter comprises the nucleotide sequence of SEQ ID NO: 1742; (iii) the intron/exon region comprises the nucleotide sequence of SEQ ID NO: 1816; (iv) the FXN-encoding sequence comprises the nucleotide sequence of SEQ ID NO: 1824; (v) the at least one miR122 binding site comprises the nucleotide sequence of SEQ ID NO: 1826; (vi) the polyA region comprises the nucleotide sequence of SEQ ID NO: 1828; and (vii) the 3' ITR region comprises the nucleotide sequence of SEQ ID NO: 1812. 78. The AAV particle of claim 77, wherein the viral genome further comprises a filler sequence comprising the nucleotide sequence of SEQ ID NO: 1841, wherein the filler sequence is positioned 3’ to the polyA region and 5’ to the 3’ ITR region.
Attorney Docket No.14640.0092-00304 79. The AAV particle of any one of claims 66-68 or any one of claims 76-78, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. 80. The AAV particle of any one of claims 66-68 or any one of claims 76-79, wherein the viral genome consists of the nucleotide sequence of SEQ ID NO: 1797. 81. An adeno-associated virus (AAV) particle comprising an AAV capsid variant and a viral genome comprising a frataxin (FXN)-encoding sequence, wherein the AAV capsid variant comprises: (i) the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); (ii) the amino acid sequence of SEQ ID NO: 738 or an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); or (iii) the amino acid sequence of SEQ ID NO: 739 or an amino acid sequence having at least 95% identity (e.g., at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); and wherein the viral genome comprises: (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical thereto; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; or (d) the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence that is at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto. 82. An adeno-associated virus (AAV) particle comprising a viral genome comprising the nucleotide sequence of SEQ ID NO: 1824 and an AAV capsid variant comprising the amino acid sequence of SEQ ID NO: 982, the amino acid sequence of SEQ ID NO: 738, and/or the amino acid sequence of SEQ ID NO: 739.
Attorney Docket No.14640.0092-00304 83. The AAV particle of claim 82, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797. 84. The AAV particle of any one of claims 1-83, wherein the viral genome is single-stranded. 85. A cell comprising the AAV particle of any one of claims 1-84, optionally wherein the cell is a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial cell. 86. A method of making the AAV particle of any one of claims 1-84, wherein the method comprises: (i) providing a cell comprising the viral genome comprising a FXN-encoding sequence and a nucleic acid encoding an AAV capsid variant; and (ii) incubating the cell under conditions suitable to encapsulate the viral genome in the AAV capsid variant; thereby making the AAV particle. 87. The method of claim 86, wherein the viral genome comprises (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; or (d) the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and wherein the AAV capsid variant comprises (i) the amino acid sequence of SEQ ID NO: 982 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); (ii) the amino acid sequence of SEQ ID NO: 738 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least
Attorney Docket No.14640.0092-00304 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943); or (iii) the amino acid sequence of SEQ ID NO: 739 or an amino acid sequence having at least 90% identity (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identity) thereto, wherein the AAV capsid variant comprises the amino acid sequence YPAEVVQK (SEQ ID NO: 943). 88. The method of claim 86, wherein the viral genome comprises (a) the nucleotide sequence of SEQ ID NO: 1797 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (b) the nucleotide sequence of SEQ ID NO: 1801 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; (c) the nucleotide sequence of SEQ ID NO: 1808 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; or (d) the nucleotide sequence of SEQ ID NO: 1809 or a nucleotide sequence at least 90% identical (e.g., at least 90%, at least 91%, at least 92%, at least 93%, at least 94%, at least 95%, at least 96%, at least 97%, at least 98%, or at least 99% identical) thereto; and wherein the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982, the amino acid sequence of SEQ ID NO: 738, and/or the amino acid sequence of SEQ ID NO: 739. 89. The method of claim 86, wherein the viral genome comprises the nucleotide sequence of SEQ ID NO: 1797 and the AAV capsid variant comprises the amino acid sequence of SEQ ID NO: 982, the amino acid sequence of SEQ ID NO: 738, and/or the amino acid sequence of SEQ ID NO: 739. 90. The method of any one of claims 86-89, further comprising, prior to step (i), introducing a nucleic acid molecule comprising the viral genome into the cell. 91. The method of any one of claims 86-90, further comprising, prior to step (i), introducing the nucleic acid encoding the AAV capsid variant into the cell. 92. The method of any one of claims 86-91, wherein the cell comprises a mammalian cell (e.g., an HEK293 cell), an insect cell (e.g., an Sf9 cell), or a bacterial cell.
Attorney Docket No.14640.0092-00304 93. A pharmaceutical composition comprising the AAV particle of any one of claims 1-84 and a pharmaceutically acceptable excipient. 94. A method of delivering an AAV particle encoding a frataxin (FXN) protein to a cell, comprising administering an effective amount of the pharmaceutical composition of claim 93 or the AAV particle of any one of claims 1-84. 95. The method of claim 94, wherein the cell is in a subject. 96. The method of claim 95, wherein the subject has, has been diagnosed with having, or is at risk of having a disorder associated with FXN deficiency, optionally wherein the disorder is Friedreich’s Ataxia (FA). 97. A method of treating a subject having or diagnosed with having a disorder associated with frataxin (FXN) deficiency, comprising administering to the subject an effective amount of the pharmaceutical composition of claim 93 or the AAV particle of any one of claims 1-84. 98. The method of claim 97, wherein the subject has, has been diagnosed with having, or is at risk of having Friedreich’s Ataxia (FA). 99. A method of treating a subject having or diagnosed with having a disorder, wherein the disorder is Friedreich’s Ataxia (FA), comprising administering to the subject an effective amount of the pharmaceutical composition of claim 93 or the AAV particle of any one of claims 1-84. 100. The method of any one of claims 97-99, wherein the treating results in prevention of progression of the disorder in the subject. 101. The method of any one of claims 97-100, wherein the treating results in amelioration of at least one symptom of the disorder. 102. The method of claim 101, wherein the at least one symptom comprises impaired sensory functions, impaired motor function (e.g., ataxia and/or involuntary movements), fatigue, chronic pain, seizures, impaired speech, sleep disturbances, metabolic disorders (e.g., diabetes), and/or increased spasticity. 103. The method of any one of claims 97-102, wherein the treating stabilizes, slows the progression of, or improves the subject’s disorder as determined by the modified Friedreich Ataxia Rating Scale
Attorney Docket No.14640.0092-00304 (mFARS), the Scale for the Assessment and Rating of Ataxia (SARA), and/or the International Cooperative Ataxia Rating Scale (ICARS). 104. The method of claim 103, wherein the treatment slows the subject’s progression of the disorder as measured by mFARS, SARA, and/or ICARS relative to an individual with the disorder who has not been administered the pharmaceutical composition or the AAV particle. 105. The method of any one of claims 95-104, wherein the subject is a human. 106. The method of any one of claims 95-105, wherein the AAV particle or the pharmaceutical composition is delivered to a cell or tissue of the central nervous system (CNS) in the subject. 107. The method of claim 106, wherein the cell or tissue of the CNS is a cell or tissue of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, cerebellum, or a combination thereof. 108. The method of any one of claims 95-107, wherein the AAV particle or the pharmaceutical composition is delivered to the subject via intravenous administration. 109. The method of any one of claims 95-108, further comprising evaluating, e.g., measuring, the level of FXN expression, e.g., FXN gene expression, FXN mRNA expression, and/or FXN protein expression, in the subject, e.g., in a cell, tissue, or fluid of the subject. 110. The method of claim 109, wherein the level of FXN protein expression is measured by an enzyme- linked immunosorbent assay (ELISA), a Western blot, an immunohistochemistry assay, or a frataxin biofluid assay. 111. The method of claim 109 or claim 110, wherein evaluating the subject’s level of FXN expression is performed before and/or after administration of the pharmaceutical composition or AAV particle, optionally wherein the subject’s level of FXN expression before administration is compared to the subject’s level of FXN expression after administration. 112. The method of any one of claims 109-111, comprising evaluating the level of FXN expression in a cell or tissue of the CNS.
Attorney Docket No.14640.0092-00304 113. The method of claim 112, wherein the cell or tissue of the CNS is a cell or tissue of the spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum. 114. The method of any one of claims 109-111, comprising evaluating the level of FXN expression in a peripheral cell or tissue. 115. The method of claim 114, wherein the peripheral cell or tissue is a cell or tissue of the heart and/or muscle. 116. The method of any one of claims 109-115, wherein the subject’s level of FXN protein expression after administration is increased relative to the subject’s level of FXN protein expression before administration. 117. The method of any one of claims 95-116, further comprising evaluating, e.g., measuring, the level of FXN protein activity in the subject. 118. The method of any one of claims 95-117, wherein administering the pharmaceutical composition or AAV particle to the subject results in an increase in: (i) the level of FXN protein or FXN gene expression in a cell, tissue, (e.g., a cell or tissue of the CNS, e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum), and/or fluid (e.g., CSF and/or serum), of the subject relative to baseline; (ii) the number and/or level of viral genomes (VG) per cell in a CNS tissue (e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) of the subject relative to the number and/or level of VG per cell in a peripheral tissue of the subject; and/or (iii) the level of FXN activity in a cell, tissue, (e.g., a cell or tissue of the CNS, e.g., spinal cord, temporal cortex, perirhinal cortex, globus pallidus, putamen, caudate, thalamus, hippocampus, geniculate nucleus, Purkinje cell layer, deep cerebellar nuclei, dentate nucleus, brainstem, and/or cerebellum) of the subject relative to baseline and/or relative to FXN activity in a cell, tissue, or fluid of an individual with a disorder associated with FXN deficiency who has not been administered the pharmaceutical composition or AAV particle. 119. The method of any one of claims 95-116, further comprising administering to the subject at least one additional therapeutic agent and/or therapy.
Attorney Docket No.14640.0092-00304 120. The method of claim 119, wherein the at least one additional therapeutic agent and/or therapy comprises an agent and/or therapy suitable for treating a disorder associated with FXN deficiency (e.g., Friedreich’s Ataxia). 121. The method of claim 119 or claim 120, wherein the at least one additional therapeutic agent and/or therapy comprises omaveloxolone or idebenone. 122. The method of any one of claims 95-121, further comprising administering an immunosuppressant to the subject. 123. The method of claim 122, wherein the immunosuppressant comprises a corticosteroid (e.g., prednisone, prednisolone, methylprednisolone, and/or dexamethasone), rapamycin, mycophenolate mofetil, tacrolimus, rituximab, and/or eculizumab hydroxychloroquine. 124. The pharmaceutical composition of claim 93 or the AAV particle of any one of claims 1-84 for use in a method of treating a disorder according to any one of claims 97-123. 125. The pharmaceutical composition of claim 93 or the AAV particle of any one of claims 1-84 for use in the treatment of a disorder associated with FXN deficiency in a subject, optionally wherein the disorder is Friedreich’s Ataxia (FA). 126. The pharmaceutical composition or the AAV particle of claim 125, wherein the subject has, has been diagnosed with having, or is at risk of having FA. 127. Use of the pharmaceutical composition of claim 93 or the AAV particle of any one of claims 1-84 in the manufacture of a medicament for the treatment of a disorder associated with FXN deficiency in a subject, optionally wherein the disorder is Friedreich’s Ataxia (FA). 128. The use of claim 127, wherein the subject has, has been diagnosed with having, or is at risk of having FA.
Applications Claiming Priority (2)
| Application Number | Priority Date | Filing Date | Title |
|---|---|---|---|
| US202363519952P | 2023-08-16 | 2023-08-16 | |
| US63/519,952 | 2023-08-16 |
Publications (1)
| Publication Number | Publication Date |
|---|---|
| WO2025038800A1 true WO2025038800A1 (en) | 2025-02-20 |
Family
ID=92672204
Family Applications (1)
| Application Number | Title | Priority Date | Filing Date |
|---|---|---|---|
| PCT/US2024/042405 Pending WO2025038800A1 (en) | 2023-08-16 | 2024-08-15 | Compositions and methods for the treatment of disorders related to frataxin deficiency |
Country Status (3)
| Country | Link |
|---|---|
| AR (1) | AR133562A1 (en) |
| TW (1) | TW202523846A (en) |
| WO (1) | WO2025038800A1 (en) |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12419969B2 (en) | 2021-11-02 | 2025-09-23 | Voyager Therapeutics, Inc. | AAV capsid variants and uses thereof |
| US12467046B2 (en) | 2018-10-02 | 2025-11-11 | Voyager Therapeutics, Inc. | Redirection of tropism of AAV capsids |
Citations (36)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5064764A (en) | 1988-12-20 | 1991-11-12 | Commissariat A L'energie Atomique | Mineral hollow fiber bioreactor for the cultivation of animal cells |
| WO1996039530A2 (en) | 1995-06-05 | 1996-12-12 | The Trustees Of The University Of Pennsylvania | Recombinant adenovirus and adeno-associated virus, cell lines, and methods of production and use thereof |
| WO1998010088A1 (en) | 1996-09-06 | 1998-03-12 | Trustees Of The University Of Pennsylvania | An inducible method for production of recombinant adeno-associated viruses utilizing t7 polymerase |
| US5756283A (en) | 1995-06-05 | 1998-05-26 | The Trustees Of The University Of Pennsylvania | Method for improved production of recombinant adeno-associated viruses for gene therapy |
| WO1999014354A1 (en) | 1997-09-19 | 1999-03-25 | The Trustees Of The University Of The Pennsylvania | Methods and vector constructs useful for production of recombinant aav |
| WO1999015685A1 (en) | 1997-09-19 | 1999-04-01 | The Trustees Of The University Of Pennsylvania | Methods and cell line useful for production of recombinant adeno-associated viruses |
| WO1999047691A1 (en) | 1998-03-20 | 1999-09-23 | Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| WO2000028004A1 (en) | 1998-11-10 | 2000-05-18 | The University Of North Carolina At Chapel Hill | Virus vectors and methods of making and administering the same |
| WO2000075353A1 (en) | 1999-06-02 | 2000-12-14 | Trustees Of The University Of Pennsylvania | Compositions and methods useful for production of recombinant viruses which require helper viruses |
| US6194191B1 (en) | 1996-11-20 | 2001-02-27 | Introgen Therapeutics, Inc. | Method for the production and purification of adenoviral vectors |
| US6204059B1 (en) | 1994-06-30 | 2001-03-20 | University Of Pittsburgh | AAV capsid vehicles for molecular transfer |
| WO2001023001A2 (en) | 1999-09-29 | 2001-04-05 | The Trustees Of The University Of Pennsylvania | Rapid peg-modification |
| WO2001023597A2 (en) | 1999-09-29 | 2001-04-05 | The Trustees Of The University Of Pennsylvania | Cell lines and constructs useful in production of e1-deleted adenoviruses in absence of replication competent adenovirus |
| US6258595B1 (en) | 1999-03-18 | 2001-07-10 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| US6265389B1 (en) | 1995-08-31 | 2001-07-24 | Alkermes Controlled Therapeutics, Inc. | Microencapsulation and sustained release of oligonucleotides |
| US6485966B2 (en) | 1999-03-18 | 2002-11-26 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| US6566118B1 (en) | 1997-09-05 | 2003-05-20 | Targeted Genetics Corporation | Methods for generating high titer helper-free preparations of released recombinant AAV vectors |
| WO2004112727A2 (en) | 2003-06-19 | 2004-12-29 | Avigen, Inc. | Aav virions with decreased immunoreactivity and uses therefor |
| WO2005005610A2 (en) | 2003-06-30 | 2005-01-20 | The Regents Of The University Of California | Mutant adeno-associated virus virions and methods of use thereof |
| WO2005072364A2 (en) | 2004-01-27 | 2005-08-11 | University Of Florida | A modified baculovirus expression system for production of pseudotyped raav vector |
| US6953690B1 (en) | 1998-03-20 | 2005-10-11 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| US7291498B2 (en) | 2003-06-20 | 2007-11-06 | The Trustees Of The University Of Pennsylvania | Methods of generating chimeric adenoviruses and uses for such chimeric adenoviruses |
| US7465583B2 (en) | 2000-06-01 | 2008-12-16 | The University Of North Carolina At Chapel Hill | Duplexed parvovirus vectors |
| US7491508B2 (en) | 2003-06-20 | 2009-02-17 | The Trustees Of The University Of Pennsylvania | Methods of generating chimeric adenoviruses and uses for such chimeric adenoviruses |
| US8137948B2 (en) | 2003-05-21 | 2012-03-20 | Genzyme Corporation | Methods for producing preparations of recombinant AAV virions substantially free of empty capsids |
| US8163543B2 (en) | 2005-10-20 | 2012-04-24 | Amsterdam Molecular Therapeutics B.V. | AAV vectors produced in insect cells |
| WO2015191508A1 (en) | 2014-06-09 | 2015-12-17 | Voyager Therapeutics, Inc. | Chimeric capsids |
| WO2020055342A1 (en) | 2018-09-10 | 2020-03-19 | Ozyegin Universitesi | Robotic manipulator including pneumatic artificial muscle |
| WO2020072683A1 (en) | 2018-10-02 | 2020-04-09 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
| US20210277418A1 (en) * | 2018-08-03 | 2021-09-09 | Voyager Therapeutics, Inc. | Aav variants with enhanced tropism |
| WO2021202651A1 (en) | 2020-04-01 | 2021-10-07 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
| WO2021230987A1 (en) | 2020-05-13 | 2021-11-18 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
| WO2021242909A1 (en) | 2020-05-26 | 2021-12-02 | Shape Therapeutics Inc. | High throughput engineering of functional aav capsids |
| WO2023091948A1 (en) * | 2021-11-17 | 2023-05-25 | Voyager Therapeutics, Inc. | Aav capsid variants and uses thereof |
| WO2023154693A1 (en) * | 2022-02-08 | 2023-08-17 | Voyager Therapeutics, Inc. | Aav capsid variants and uses thereof |
| WO2023240236A1 (en) * | 2022-06-10 | 2023-12-14 | Voyager Therapeutics, Inc. | Compositions and methods for the treatment of spinal muscular atrophy related disorders |
-
2024
- 2024-08-15 AR ARP240102170A patent/AR133562A1/en unknown
- 2024-08-15 TW TW113130648A patent/TW202523846A/en unknown
- 2024-08-15 WO PCT/US2024/042405 patent/WO2025038800A1/en active Pending
Patent Citations (47)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US5064764A (en) | 1988-12-20 | 1991-11-12 | Commissariat A L'energie Atomique | Mineral hollow fiber bioreactor for the cultivation of animal cells |
| US6204059B1 (en) | 1994-06-30 | 2001-03-20 | University Of Pittsburgh | AAV capsid vehicles for molecular transfer |
| US6261551B1 (en) | 1995-06-05 | 2001-07-17 | The Trustees Of The University Of Pennsylvania | Recombinant adenovirus and adeno-associated virus, cell lines, and methods of production and use thereof |
| WO1996039530A2 (en) | 1995-06-05 | 1996-12-12 | The Trustees Of The University Of Pennsylvania | Recombinant adenovirus and adeno-associated virus, cell lines, and methods of production and use thereof |
| US5756283A (en) | 1995-06-05 | 1998-05-26 | The Trustees Of The University Of Pennsylvania | Method for improved production of recombinant adeno-associated viruses for gene therapy |
| US6281010B1 (en) | 1995-06-05 | 2001-08-28 | The Trustees Of The University Of Pennsylvania | Adenovirus gene therapy vehicle and cell line |
| US6270996B1 (en) | 1995-06-05 | 2001-08-07 | The Trustees Of The University Of Pennsylvania | Recombinant adenovirus and adeno-associated virus, cell lines and methods of production and use thereof |
| US6265389B1 (en) | 1995-08-31 | 2001-07-24 | Alkermes Controlled Therapeutics, Inc. | Microencapsulation and sustained release of oligonucleotides |
| US6555525B2 (en) | 1995-08-31 | 2003-04-29 | Alkermes Controlled Therapeutics, Inc. | Microencapsulation and sustained release of oligonucleotides |
| WO1998010088A1 (en) | 1996-09-06 | 1998-03-12 | Trustees Of The University Of Pennsylvania | An inducible method for production of recombinant adeno-associated viruses utilizing t7 polymerase |
| US6194191B1 (en) | 1996-11-20 | 2001-02-27 | Introgen Therapeutics, Inc. | Method for the production and purification of adenoviral vectors |
| US6566118B1 (en) | 1997-09-05 | 2003-05-20 | Targeted Genetics Corporation | Methods for generating high titer helper-free preparations of released recombinant AAV vectors |
| WO1999014354A1 (en) | 1997-09-19 | 1999-03-25 | The Trustees Of The University Of The Pennsylvania | Methods and vector constructs useful for production of recombinant aav |
| US6482634B1 (en) | 1997-09-19 | 2002-11-19 | The Trustees Of The University Of Pennsylvania | Methods and vector constructs useful for production of recombinant AAV |
| US7238526B2 (en) | 1997-09-19 | 2007-07-03 | The Trustees Of The University Of Pennsylvania | Methods and cell line useful for production of recombinant adeno-associated viruses |
| US6943019B2 (en) | 1997-09-19 | 2005-09-13 | The Trustees Of The University Of Pennsylvania | Methods and vector constructs useful for production of recombinant AAV |
| WO1999015685A1 (en) | 1997-09-19 | 1999-04-01 | The Trustees Of The University Of Pennsylvania | Methods and cell line useful for production of recombinant adeno-associated viruses |
| US6475769B1 (en) | 1997-09-19 | 2002-11-05 | The Trustees Of The University Of Pennsylvania | Methods and cell line useful for production of recombinant adeno-associated viruses |
| WO1999047691A1 (en) | 1998-03-20 | 1999-09-23 | Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| US6953690B1 (en) | 1998-03-20 | 2005-10-11 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| WO2000028004A1 (en) | 1998-11-10 | 2000-05-18 | The University Of North Carolina At Chapel Hill | Virus vectors and methods of making and administering the same |
| US7022519B2 (en) | 1999-03-18 | 2006-04-04 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| US6258595B1 (en) | 1999-03-18 | 2001-07-10 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| US6485966B2 (en) | 1999-03-18 | 2002-11-26 | The Trustees Of The University Of Pennsylvania | Compositions and methods for helper-free production of recombinant adeno-associated viruses |
| WO2000075353A1 (en) | 1999-06-02 | 2000-12-14 | Trustees Of The University Of Pennsylvania | Compositions and methods useful for production of recombinant viruses which require helper viruses |
| WO2001023001A2 (en) | 1999-09-29 | 2001-04-05 | The Trustees Of The University Of Pennsylvania | Rapid peg-modification |
| US6365394B1 (en) | 1999-09-29 | 2002-04-02 | The Trustees Of The University Of Pennsylvania | Cell lines and constructs useful in production of E1-deleted adenoviruses in absence of replication competent adenovirus |
| WO2001023597A2 (en) | 1999-09-29 | 2001-04-05 | The Trustees Of The University Of Pennsylvania | Cell lines and constructs useful in production of e1-deleted adenoviruses in absence of replication competent adenovirus |
| US7465583B2 (en) | 2000-06-01 | 2008-12-16 | The University Of North Carolina At Chapel Hill | Duplexed parvovirus vectors |
| US8137948B2 (en) | 2003-05-21 | 2012-03-20 | Genzyme Corporation | Methods for producing preparations of recombinant AAV virions substantially free of empty capsids |
| WO2004112727A2 (en) | 2003-06-19 | 2004-12-29 | Avigen, Inc. | Aav virions with decreased immunoreactivity and uses therefor |
| US7291498B2 (en) | 2003-06-20 | 2007-11-06 | The Trustees Of The University Of Pennsylvania | Methods of generating chimeric adenoviruses and uses for such chimeric adenoviruses |
| US7491508B2 (en) | 2003-06-20 | 2009-02-17 | The Trustees Of The University Of Pennsylvania | Methods of generating chimeric adenoviruses and uses for such chimeric adenoviruses |
| WO2005005610A2 (en) | 2003-06-30 | 2005-01-20 | The Regents Of The University Of California | Mutant adeno-associated virus virions and methods of use thereof |
| WO2005072364A2 (en) | 2004-01-27 | 2005-08-11 | University Of Florida | A modified baculovirus expression system for production of pseudotyped raav vector |
| US8163543B2 (en) | 2005-10-20 | 2012-04-24 | Amsterdam Molecular Therapeutics B.V. | AAV vectors produced in insect cells |
| WO2015191508A1 (en) | 2014-06-09 | 2015-12-17 | Voyager Therapeutics, Inc. | Chimeric capsids |
| US20210277418A1 (en) * | 2018-08-03 | 2021-09-09 | Voyager Therapeutics, Inc. | Aav variants with enhanced tropism |
| WO2020055342A1 (en) | 2018-09-10 | 2020-03-19 | Ozyegin Universitesi | Robotic manipulator including pneumatic artificial muscle |
| WO2020072683A1 (en) | 2018-10-02 | 2020-04-09 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
| WO2021202651A1 (en) | 2020-04-01 | 2021-10-07 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
| WO2021230987A1 (en) | 2020-05-13 | 2021-11-18 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
| US20220042044A1 (en) * | 2020-05-13 | 2022-02-10 | Voyager Therapeutics, Inc. | Redirection of tropism of aav capsids |
| WO2021242909A1 (en) | 2020-05-26 | 2021-12-02 | Shape Therapeutics Inc. | High throughput engineering of functional aav capsids |
| WO2023091948A1 (en) * | 2021-11-17 | 2023-05-25 | Voyager Therapeutics, Inc. | Aav capsid variants and uses thereof |
| WO2023154693A1 (en) * | 2022-02-08 | 2023-08-17 | Voyager Therapeutics, Inc. | Aav capsid variants and uses thereof |
| WO2023240236A1 (en) * | 2022-06-10 | 2023-12-14 | Voyager Therapeutics, Inc. | Compositions and methods for the treatment of spinal muscular atrophy related disorders |
Non-Patent Citations (39)
| Title |
|---|
| "Biocomputing: Informatics and Genome Projects", 1993, ACADEMIC PRESS |
| "Computer Analysis of Sequence Data. Part 1", 1994, HUMANA PRESS |
| "Methods In Molecular Biology", 1995, HUMANA PRESS |
| "Sequence Analysis Primer", 1991, M STOCKTON PRESS |
| A. R. GENNARO: "Remington: The Science and Practice of Pliamiac. .", 2006, LIPPINCOTT. WILLIAMS & WILKINS |
| ADLAKHA, SAINI, MOLECULAR CANCER, vol. 13, 2014, pages 33 |
| AFIONE ET AL.: "Identification and Mutagenesis of the Adeno-Associated Virus 5 Sialic Acid Binding Region", JOURNAL OF \ ''IROLOGY, vol. 89, no. 3, 2015, pages 1660 - 1672 |
| AFIONE ET AL.: "Identification and Mutagenesis of the Adeno-Associated Virus 5 Sialic Binding Region", JOURNAL F VIROLOGY, vol. 9, no. 3, 8 February 2015 (2015-02-08), pages 1660 - 1672 |
| ALTSCHUL. S. F. ET AL., J. MOLECULAR BIOL., vol. 215, 1990, pages 403 |
| CAMPUZANO ET AL., SCIENCE, vol. 271, no. 5254, 8 March 1996 (1996-03-08), pages 1423 |
| CARILLO. H.LIPMAN, D., SIAM J APPLIED MATH., vol. 48, no. 1073, 1988 |
| DEVEREUX, J. ET AL., NUCLEIC ACIDS RESEARCH, vol. 12, no. 1, 1984, pages 387 |
| GOVINDASAMY ET AL.: "Structural Insights into Adeno-Associated Virus Serotype 5", JOURNAL OF VIROLOG, vol. 87, no. 20, 2013, pages 11187 - 11199, XP055865805, DOI: 10.1128/JVI.00867-13 |
| GOVINDASAMY ET AL.: "Structural Insights into Adeno-Associated Virus Serotype 5", JOURNAL OF VIROLOGY, vol. 7, no. 20, 8 October 2013 (2013-10-08), pages 11187 - 11199, XP055865805, DOI: 10.1128/JVI.00867-13 |
| GOVINDASAMY ET AL.: "Structurally Mapping the Diverse Phenotype of Adeno-Associated Virus Serotype 4", JOURNAL OF VIROLOGY, vol. 0, no. 23, 8 December 2006 (2006-12-08), pages 11556 - 11570 |
| GRIMM ET AL., HUM. GENE THER., vol. 10, no. 15, 1999, pages 2445 - 2450 |
| HWANG ET AL.: "N-Terminal Acetylation of Cellular Proteins Creates Specific Degradation Signals", SCIENCE, vol. 327, no. 5968, 19 February 2010 (2010-02-19), pages 973 - 977, XP055369420, DOI: 10.1126/science.1183147 |
| JIN ET AL.: "Direct Liquid Chromatography/Mass Spectrometry Analysis for Complete Characterization of Recombinant Adeno-Associated Virus Capsid Proteins", ILCRM GENE TITER METHOD, vol. 28, no. 5, October 2017 (2017-10-01), pages 255 - 267 |
| JOSE ET AL.: "High-Resolution Structural Characterization of a New Adenoassociated Virus Serotype 5 Antibody Epitope toward Engineering Antibody-Resistant Recombinant Gene Delivery Vectors", JOURNAL OF V IROLOGY, vol. 93, no. 1, January 2019 (2019-01-01), pages e01394 - 18 |
| JOSE ET AL.: "High-Resolution Structural Characterization of a New Adenoassociated Virus Serotype 5 Antibody Epitope toward Engineering Antibody-Resistant Recombinant Gene Delivery Vectors", JOURNAL OF VIROLOGY, vol. 93, no. 1, 2020, pages e01394 - 18 |
| KAJIGAYA ET AL., PROC. ,\'AT'L. ACAD. SCI. USA, vol. 88, 1991, pages 4646 - 50 |
| KIMBAUER ET AL., VIR., vol. 219, 1996, pages 37 - 44 |
| KOEPPENAMULF, J NEUROL SCI., vol. 303, no. 1-2, 15 April 2011 (2011-04-15), pages 1 - 12 |
| MIYANOHARA ET AL., MOL THER METHODS CLIN DEV, vol. 3, 2016, pages 16046 |
| MYERSMILLER, CABIOS, vol. 4, 1989, pages 11 - 17 |
| NAGESHWARANFESTENSTEIN, FRONTIERS IN NEUROLOGY, vol. 6, 2015 |
| O'REILLY ET AL.: "A Laboratory Manual", 1994, OXFORD UNIV. PRESS, article "Baculovirus Expression Vectors" |
| PARKINSON ET AL., JOURNAL OF NEUROCHEMISTRY, vol. 126, 2013, pages 103 - 117 |
| POWELL ET AL., VIRAL EXPRESSION CASSETTE ELEMENTS TO EN ANCE TRANSGENE TARGET SPECIFICITY AND EXPRESSION IN GENE THERAPY, 2015 |
| QIAN ET AL.: "Directed Evolution of AAV Serotype 5 for Increased Hepatocyte Transduction and Retained Low Humoral Seroreactivity", MOLECULAR THERAPY: METHODS AND CLINICAL DEVELOPMENT, vol. 20, 2021, pages 122 - 132, XP055864144, DOI: 10.1016/j.omtm.2020.10.010 |
| QIAN ET AL.: "Directed Evolution of AAV Serotype 5 for Increased Hepatocyte Transduction and Retained Low Humoral Seroreactivity. Molecular Therapy", METHODS & CLINICAL DEVELOPMENT, vol. 20, October 2020 (2020-10-01), pages 122 - 132 |
| REISMAN, DRUG DES DEVEL THER, vol. 13, 2019, pages 1259 - 1270 |
| RUFFING ET AL., J. VIR., vol. 66, 1992, pages 6922 - 30 |
| SAMULSKI ET AL., J. VIR., vol. 63, 1989, pages 3822 - 8 |
| TRATSCHIN ET AL., MOL. CELL BIOL., vol. 5, no. 11, 1985, pages 3251 - 3260 |
| VON HEINJE, G.: "Sequence Analysis in Molecular Biology", 1987, ACADEMIC PRESS |
| WALTERS ET AL.: "Structure of Adeno-Associated Virus Serotype 5", JOURNAL OF V IROLOGY, vol. 78, no. 7, 2004, pages 3361 - 3371, XP002297595 |
| WANG ET AL.: "Directed evolution of adeno-associated virus 5 capsid enables specific liver tropism", MOL THER NUCLEIC ACIDS, vol. 28, 2022, pages 293 - 306, XP055980992, DOI: 10.1016/j.omtn.2022.03.017 |
| ZHAO ET AL., TIR, vol. 272, 2000, pages 382 - 93 |
Cited By (2)
| Publication number | Priority date | Publication date | Assignee | Title |
|---|---|---|---|---|
| US12467046B2 (en) | 2018-10-02 | 2025-11-11 | Voyager Therapeutics, Inc. | Redirection of tropism of AAV capsids |
| US12419969B2 (en) | 2021-11-02 | 2025-09-23 | Voyager Therapeutics, Inc. | AAV capsid variants and uses thereof |
Also Published As
| Publication number | Publication date |
|---|---|
| TW202523846A (en) | 2025-06-16 |
| AR133562A1 (en) | 2025-10-08 |
Similar Documents
| Publication | Publication Date | Title |
|---|---|---|
| JP7761686B2 (en) | Frataxin expression constructs with engineered promoters and methods of use thereof | |
| JP7619958B2 (en) | Recombinant adeno-associated viruses and uses thereof | |
| Klein et al. | Dose and promoter effects of adeno-associated viral vector for green fluorescent protein expression in the rat brain | |
| KR102116378B1 (en) | Compositions and methods for treating spinal muscular atrophy | |
| WO2025038800A1 (en) | Compositions and methods for the treatment of disorders related to frataxin deficiency | |
| KR102629418B1 (en) | Optimized CLN1 gene and expression cassette and uses thereof | |
| US20170007720A1 (en) | Methods and compositions for gene delivery to on bipolar cells | |
| CN116438311A (en) | Compositions useful in the treatment of Charcot-Marie-Tooth disease | |
| WO2025038802A1 (en) | Compositions and methods for the treatment of disorders related to ataxin-2 | |
| WO2025038796A1 (en) | Compositions and methods for the treatment of disorders related to cdkl5 deficiency | |
| WO2025038805A1 (en) | Compositions and methods for the treatment of disorders related to glucosylceramidase beta 1 deficiency | |
| WO2025038795A1 (en) | Compositions and methods for the treatment of disorders related to dystrophia myotonica protein kinase | |
| WO2024229125A2 (en) | Compositions and methods for the treatment of disorders related to frataxin deficiency | |
| WO2022222869A1 (en) | Recombinant adeno-associated virus and application thereof | |
| CN121241145A (en) | Compositions and methods for treating conditions associated with a synapse fusion protein binding protein 1 deficiency | |
| TW202509226A (en) | Compositions and methods for the treatment of disorders related to ataxin-2 | |
| CN113121653A (en) | Novel adeno-associated virus capsid protein specific to muscle and retina | |
| US20230285596A1 (en) | Compositions and methods for the treatment of niemann-pick type c1 disease | |
| WO2022232575A1 (en) | Compositions comprising adeno-associated virus chimera capsid proteins and methods of using the same | |
| KR20220145838A (en) | Compositions useful for treating GM1 gangliosiosis | |
| CN119971081B (en) | Compositions for treating intellectual disability, their applications, and drug screening methods | |
| WO2025122531A1 (en) | Compositions and methods for the treatment of disorders related to frataxin deficiency | |
| WO2025137219A1 (en) | Compositions and methods for the treatment of disorders related to dystrophia myotonica protein kinase | |
| WO2024125530A1 (en) | Aav vector for infecting retina, adalimumab, and use thereof | |
| TW202124416A (en) | Frataxin expression constructs having engineered promoters and methods of use thereof |
Legal Events
| Date | Code | Title | Description |
|---|---|---|---|
| 121 | Ep: the epo has been informed by wipo that ep was designated in this application |
Ref document number: 24765854 Country of ref document: EP Kind code of ref document: A1 |